Reduced Sodium Food Products
Van Lengerich; Bernhard H. ; et al.
U.S. patent application number 16/877126 was filed with the patent office on 2020-09-10 for reduced sodium food products. The applicant listed for this patent is General Mills, Inc.. Invention is credited to Olaf Gruess, Joachim Hans, Lars Ole Haustedt, Andreas Hochheimer, Michael Krohn, Jens-Peter Muller, Christine M. Nowakowski, Suzanne Denise Pecore, Candace Michelle Rathjen-Nowak, Lia Scarabottolo, Karsten Siems, Bernhard H. Van Lengerich.
| Application Number | 20200281236 16/877126 |
| Document ID | / |
| Family ID | 1000004843087 |
| Filed Date | 2020-09-10 |
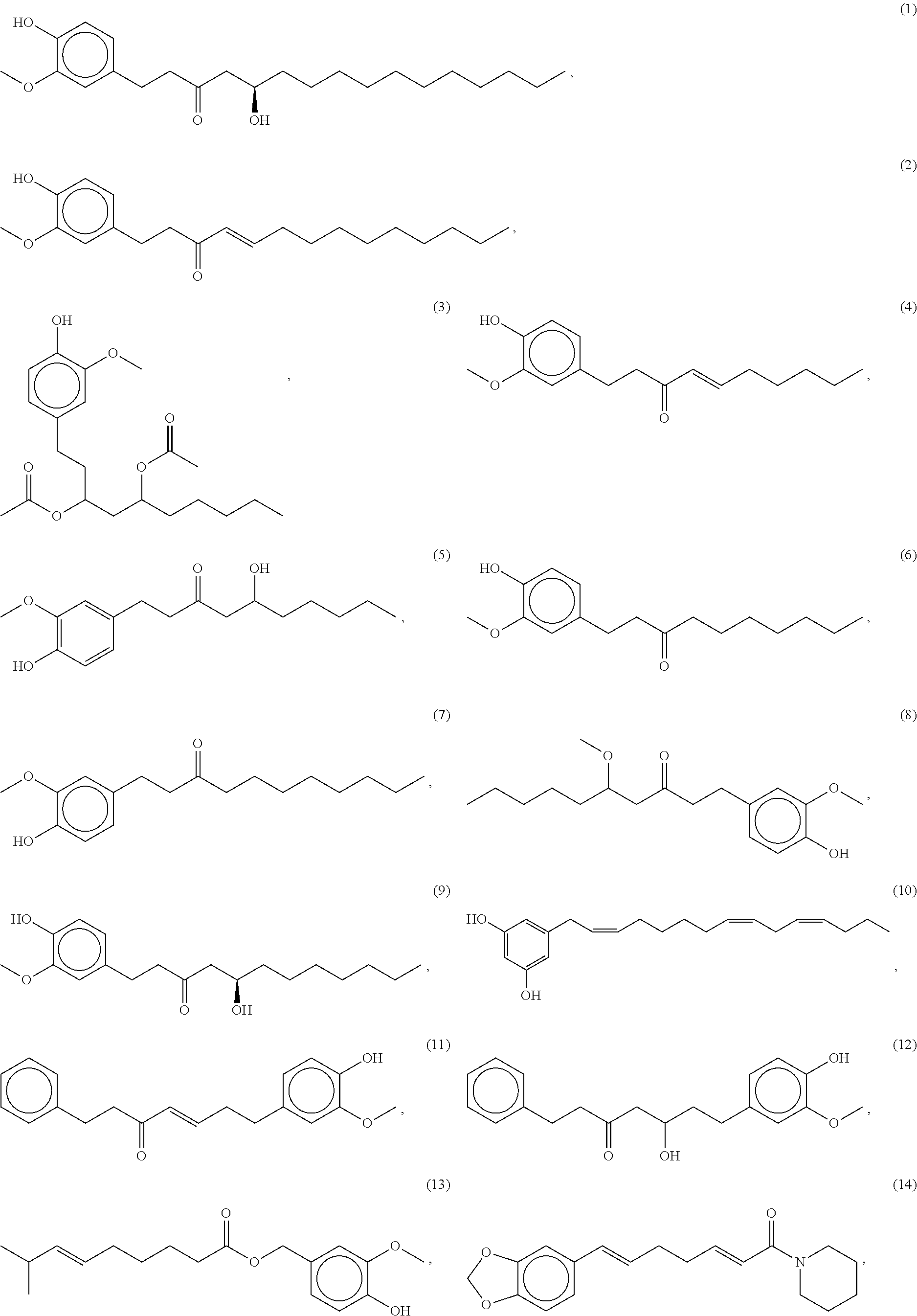
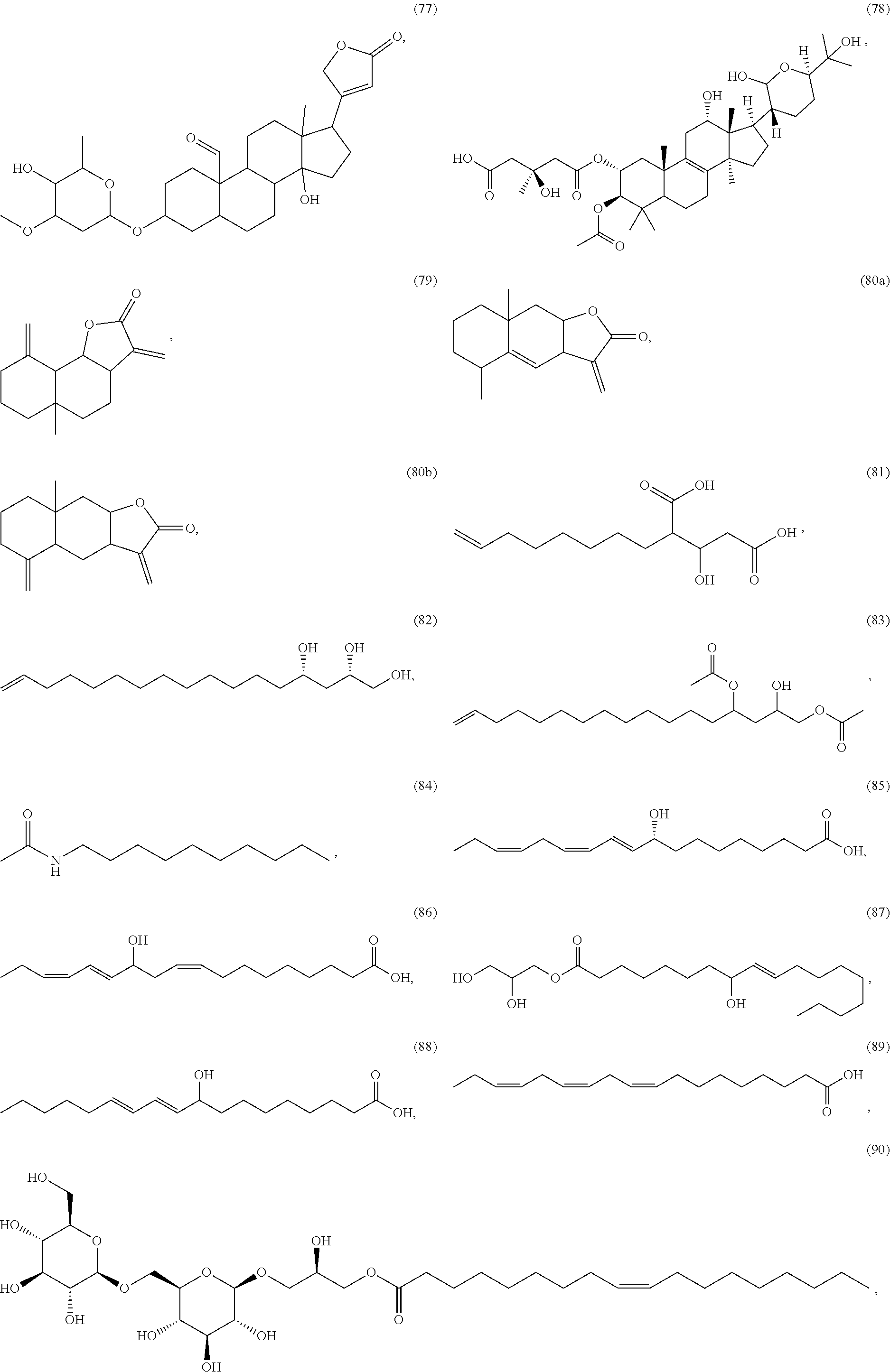
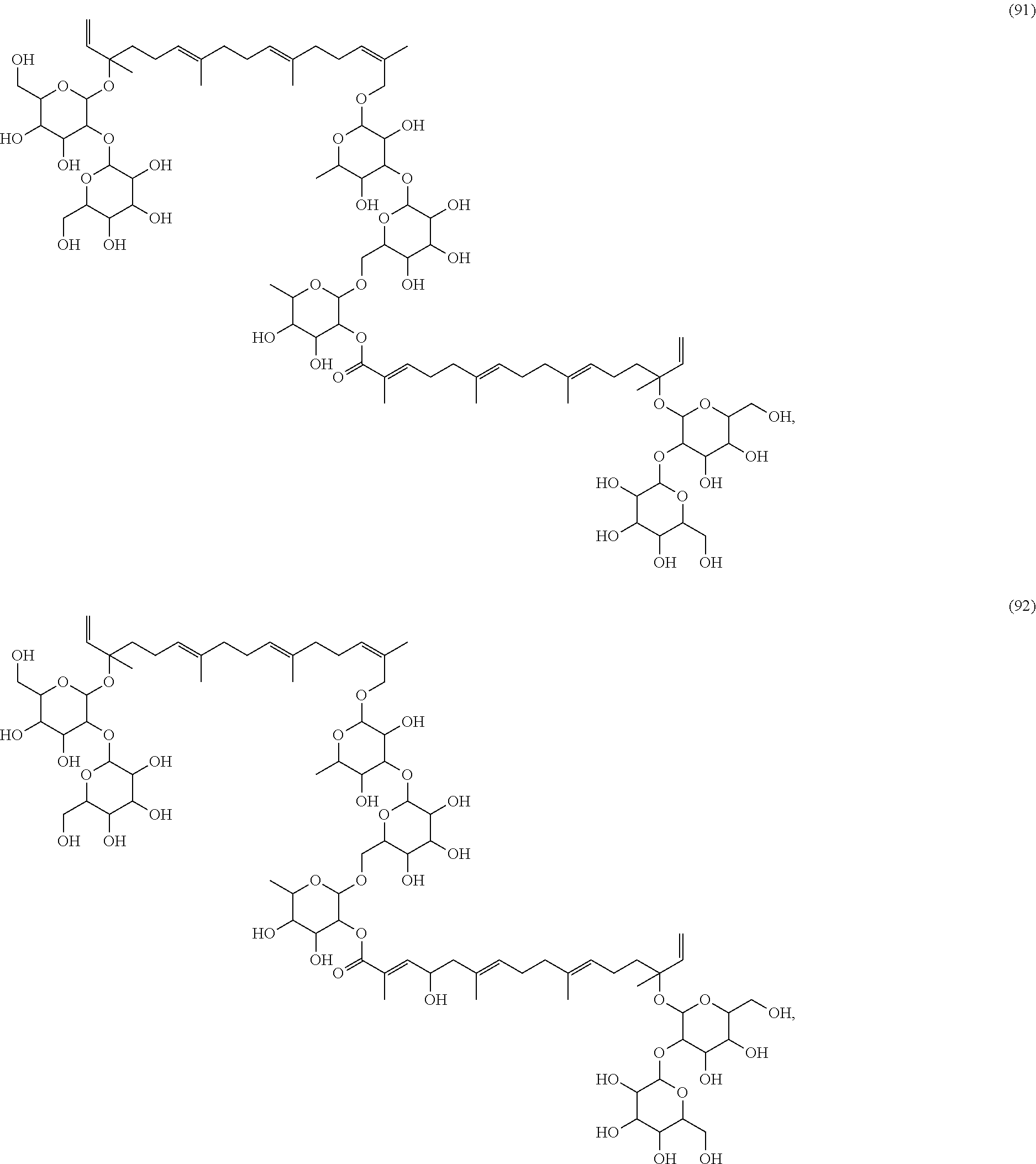
View All Diagrams
| United States Patent Application | 20200281236 |
| Kind Code | A1 |
| Van Lengerich; Bernhard H. ; et al. | September 10, 2020 |
REDUCED SODIUM FOOD PRODUCTS
Abstract
Naturally-derived compounds having various structures elicit the perception of salty taste, enhance the perception of salty taste of a salt, or act at one or more sodium channels. Food products may include such naturally-derived compounds, which may be used to reduce the sodium content, while imparting a similar level of saltiness.
| Inventors: | Van Lengerich; Bernhard H.; (Plymouth, MN) ; Gruess; Olaf; (Maple Grove, MN) ; Hans; Joachim; (Holzminden, DE) ; Haustedt; Lars Ole; (Potsdam, DE) ; Hochheimer; Andreas; (Regensburg, DE) ; Krohn; Michael; (Lorsch, DE) ; Muller; Jens-Peter; (Berlin, DE) ; Nowakowski; Christine M.; (Plymouth, MN) ; Pecore; Suzanne Denise; (Pasadena, CA) ; Rathjen-Nowak; Candace Michelle; (St. Michael, MN) ; Scarabottolo; Lia; (Peschiera Borromeo, IT) ; Siems; Karsten; (Michendorf, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004843087 | ||||||||||
| Appl. No.: | 16/877126 | ||||||||||
| Filed: | May 18, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16197550 | Nov 21, 2018 | |||
| 16877126 | ||||
| 14820891 | Aug 7, 2015 | 10159268 | ||
| 16197550 | ||||
| PCT/US2014/015230 | Feb 7, 2014 | |||
| 14820891 | ||||
| PCT/US2014/015192 | Feb 7, 2014 | |||
| 14820891 | ||||
| PCT/US2014/015200 | Feb 7, 2014 | |||
| PCT/US2014/015192 | ||||
| PCT/US2014/015207 | Feb 7, 2014 | |||
| PCT/US2014/015200 | ||||
| PCT/US2014/015211 | Feb 7, 2014 | |||
| PCT/US2014/015207 | ||||
| PCT/US2014/015216 | Feb 7, 2014 | |||
| PCT/US2014/015211 | ||||
| PCT/US2014/015220 | Feb 7, 2014 | |||
| PCT/US2014/015216 | ||||
| PCT/US2014/015227 | Feb 7, 2014 | |||
| PCT/US2014/015220 | ||||
| PCT/US2014/015227 | Feb 7, 2014 | |||
| PCT/US2014/015227 | ||||
| PCT/US2014/015239 | Feb 7, 2014 | |||
| PCT/US2014/015227 | ||||
| PCT/US2014/015240 | Feb 7, 2014 | |||
| PCT/US2014/015239 | ||||
| PCT/US2014/015244 | Feb 7, 2014 | |||
| PCT/US2014/015240 | ||||
| 61762781 | Feb 8, 2013 | |||
| 61762792 | Feb 8, 2013 | |||
| 61762798 | Feb 8, 2013 | |||
| 61762804 | Feb 8, 2013 | |||
| 61763244 | Feb 11, 2013 | |||
| 61763274 | Feb 11, 2013 | |||
| 61763300 | Feb 11, 2013 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A23L 27/45 20160801; A23L 7/117 20160801; A23L 23/00 20160801; A23V 2002/00 20130101; A23L 27/40 20160801 |
| International Class: | A23L 7/117 20060101 A23L007/117; A23L 27/40 20060101 A23L027/40; A23L 23/00 20060101 A23L023/00 |
Claims
1-78. (canceled)
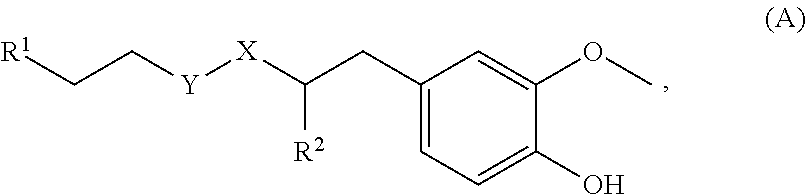
79. A food product comprising: a first ingredient; a salt that imparts a salty taste; and a compound having the following structure: ##STR00205## where: R.sup.1 is H or C.sub.1-C.sub.10 alkyl; R.sup.2 is H or C.sub.1-C.sub.3 alkyl; X is CHOR.sup.3 or C.dbd.O; R.sup.3 is H, C.sub.1-C.sub.3 alkyl, or ##STR00206## R.sup.4 is H or C.sub.1-C.sub.3 alkyl; Y is CR.sup.5.dbd.CH or CHR.sup.5--CH.sub.2; R.sup.5 is H, OH, --OCH.sub.3, --OCH.sub.2CH.sub.3, --O--OCH.sub.2CH.sub.2CH.sub.3, or ##STR00207## and R.sup.6 is H or C.sub.1-C.sub.3 alkyl, wherein the compound of formula A is present in the food product in an amount sufficient to enhance a perception of saltiness of the food product, and wherein the compound of formula A is isolated or purified before being included in the food product.
80. The food product of claim 79, wherein R.sup.1 is C.sub.2-C.sub.8 alkyl.
81. The food product of claim 79, wherein R.sup.2 is H.
82. The food product of claim 79, wherein X is C.dbd.O or ##STR00208## where R4 is CH.sub.3.
83. The food product of claim 79, wherein Y is CR.sup.5.dbd.CH and R.sup.5 is H.
84. The food product of claim 79, wherein Y is CHR.sup.5--CH.sub.2 and R.sup.5 is OH or --OCH.sub.3.
85. The food product of claim 79, wherein R.sup.6 is CH.sub.3.
86. The food product of claim 79, wherein the compound of formula A is one of the following compounds: ##STR00209## ##STR00210##
87. The food product of claim 79, wherein the compound of formula A is compound 1.
88. The food product of claim 79, wherein the compound of formula A is compound 2.
89. The food product of claim 79, wherein the compound of formula A is compound 3.
90. The food product of claim 79, wherein the compound of formula A is compound 4.
91. The food product of claim 79, wherein the compound of formula A is compound 5.
92. The food product of claim 79, wherein the compound of formula A is compound 6.
93. The food product of claim 79, wherein the compound of formula A is compound 7.
94. The food product of claim 79, wherein the compound of formula A is compound 8.
95. The food product of claim 79, wherein the compound of formula A is compound 9.
96. The food product of claim 79, wherein the compound of formula A is present in the food product from 0.01 to 2 percent by weight.
Description
RELATED APPLICATIONS
[0001] This application claims the benefit of each of the following Provisional Patent Applications: US 61/762,781, filed on Feb. 8, 2013; US 61/762,792, filed on Feb. 8, 2013; US 61/762,798, filed on Feb. 8, 2013; US 61/762,804, filed on Feb. 8, 2013; U.S. 61/763,244, filed on Feb. 11, 2013; US 61/763,274, filed on Feb. 11, 2013; and US 61/763,300, filed on Feb. 11, 2013. Each of the above-referenced US Provisional Patent Applications is hereby incorporated herein by reference in its respective entirety to the extent that it does not conflict with the disclosure presented herein.
FIELD
[0002] This disclosure generally relates to, among other things, food products having a compound that modifies or enhances the taste of the food product, for example, the saltiness of the food product.
BACKGROUND
[0003] Sodium chloride, ordinary table salt, is the prototypical compound for eliciting the perception of salty taste. However, attempts to reduce sodium consumption have led investigators to find suitable substitutes for sodium chloride or to reduce sodium chloride amounts, without sacrificing salty taste.
[0004] Salts can elicit complex tastes, including mixtures of sweet, bitter, sour, umami, and salty perceptual components. It is believed that the cations of salts impart the perceptual taste component, while the anions, in addition to contributing to tastes of their own, modify the perception of the taste of the cations. By way of example, sodium and lithium are believed to impart only salty tastes, while potassium and other alkaline earth cations produce both salty and bitter tastes. Among the anions commonly found in foods, the chloride ion is considered to be the least inhibitory to the salty taste, while the citrate anion is more inhibitory.
[0005] Many attempts have been made to provide salty tasting compositions as a substitute for table salt which will give the same or a similar seasoning effect and which are comprised of substantially reduced quantities of sodium chloride. To this end, potassium chloride, ammonium chloride, and similar compounds have been suggested. The use of such salts, and combinations of such salts, leaves much to be desired as to taste. None of them individually or in combination positively affects other taste modalities and tastes like sodium chloride. Each alone has a disagreeable taste, as do mixtures of such salts. For example, potassium chloride has a strong aftertaste that is characterized as "bitter" by most people. Ammonium chloride also has a bitter aftertaste.
SUMMARY
[0006] This disclosure describes, among other things, compounds that elicit or enhance the perception of salty taste, or another taste associated with consumption of sodium chloride or other salts, or that interact with a receptor or ion channel associated with the perception of salty taste or another complex taste associated with consumption of sodium chloride or other salts. In embodiments, the compounds are naturally derived taste modulating compounds used as ingredients in food products to elicit or enhance perception of salty taste. In embodiments, the food products contain lower amounts of sodium than normal.
[0007] As described herein, a number of derived compounds were screened for their ability to modulate activity of a sodium channel in vitro. Many of the identified compounds were found to enhance the saltiness of a composition containing sodium chloride.
[0008] One or more embodiments of the compounds, compositions, food products or methods described herein provide one or more advantages over prior compounds, compositions, food products or methods. For example, food products that include one or more taste modulating or salty taste modulating compounds described herein may have lower sodium content relative to food products that do not include such taste modulating or salty compounds while imparting a similar level of saltiness. This and other advantages will be readily understood from the following detailed description.
BRIEF DESCRIPTION OF THE DRAWINGS
[0009] FIG. 1 is a table that provides results of DAP score testing regarding the perception of saltiness of various combinations of compounds in sodium chloride solution.
[0010] FIG. 2 is a table that provides results of DAP score testing regarding the perception of saltiness of various combinations of compounds in combination in broth solution.
DETAILED DESCRIPTION
[0011] This disclosure describes, among other things, compounds that elicit or enhance the perception of salty taste or another taste associated with consumption of sodium chloride. In embodiments, the compounds are taste modulating compounds used as ingredients in food products to elicit or enhance perception of salty taste. In embodiments, the food products are food products that contain reduced amounts of sodium, while imparting a salty taste typically associated with higher amounts of sodium.
[0012] In embodiments, a food product includes (i) a taste modulating or salty taste modulating compound, or derivatives thereof, or (ii) a composition that comprises a taste modulating or salty taste modulating compound, or derivatives thereof. The taste modulating, or salty taste modulating compound may be derived from a natural product, may be synthesized, or may be isolated or purified.
[0013] As used herein, a "food product" is a food in a form that does not exist in nature. In embodiments, a food product includes at least two edible ingredients that do not exist together in nature. A "food" is a nutritious substance that animals, including humans, pets and livestock, eat or drink. A "nutritious substance" is a macronutrient such as a fat, carbohydrate or protein, or a micronutrient such as an essential or non-essential vitamin or mineral.
[0014] One or more taste modulating or salty taste modulating compounds described herein or derivatives thereof, alone or in combination, may be incorporated into a food product. The one or more compounds may elicit a perception of saltiness when the food product is consumed. In embodiments, the one or more compounds are included in a food product that contains a salt that imparts a salty taste. Preferably, at least one of the one or more compounds is a taste modulating compound or salty taste modulating compound.
[0015] In embodiments, a food product includes an ingredient, a salt that imparts a salty taste, and a taste modulating or salty taste modulating compound. The ingredient may be a nutritious ingredient; that is, an ingredient that is a nutritious substance. The taste modulating or salty taste modulating compound may be present in the food product in an amount sufficient to enhance the salty taste of the food product. In embodiments, the ingredient, the salt and the taste modulating or salty taste modulating compound are present in the food product in amounts or concentrations not found in naturally existing food products, such as bananas, peppers, avocados, wheat, or the like.
[0016] In embodiments, at least one of the one or more compounds is a salty taste modulating compound and is present in the food product in an amount or concentration sufficient to elicit or enhance the perception of saltiness. In embodiments, the one or more salty taste modulating compounds are present in the food product in an amount or concentration sufficient to elicit or enhance the perception of salty taste such that less salt may be included in the food product to elicit a similar perception of saltiness as a substantially similar food product that does not include the one or more salty taste modulating compounds. Preferably, the reduced salt food product elicits the same or a similar perception of saltiness as a substantially similar food product that does not include the one or more salty taste modulating compounds.
[0017] In embodiments, the one or more taste modulating or salty taste modulating compounds are present in a food product in an amount or concentration sufficient to elicit or enhance the perception of salty taste such that the amount of sodium may be reduced by about 10 mg or more per serving relative to a substantially similar food product that does not have the one or more taste modulating or salty taste modulating compounds while having a similar salty taste. In embodiments, the one or more taste modulating or salty taste modulating compounds are present in a food product in an amount or concentration sufficient to elicit or enhance the perception of salty taste such that the amount of sodium in a serving of a food product may be reduced to about 150 mg or less, more particularly to about 100 mg or less, more particularly to about 75 mg or less, more particularly to about 25 mg or less, more particularly to about 10 mg or less. By way of example, it may be desirable to reduce sodium by about 10 mg or more in cereals or snacks per serving relative to a substantially similar food product that does not have the one or more taste modulating or salty taste modulating compounds while having a similar salty taste. It may be desirable to reduce sodium to about 150 mg or less, more particularly to about 100 mg or less, more particularly to about 75 mg or less, more particularly to about 25 mg or less, more particularly to about 10 mg or less in cereals or snacks per serving. For cereal, a typical serving size is 50 grams. Of course, cereals may have other serving sizes.
[0018] In embodiments, the one or more taste modulating or salty taste modulating compounds are present in a food product in an amount or concentration sufficient to elicit or enhance the perception of salty taste such that the amount of sodium may be reduced by about 20 mg or more per serving relative to a substantially similar food product that does not have the one or more taste modulating or salty taste modulating compounds while having a similar salty taste. In embodiments, the one or more taste modulating or salty taste modulating compounds are present in a serving of a food product in an amount or concentration sufficient to elicit or enhance the perception of salty taste such that the amount of sodium may be reduced to about 800 mg or less, more particularly to about 500 mg or less, more particularly to about 300 mg or less, more particularly to about 100 mg or less, more particularly to about 20 mg or less. By way of example, it may be desirable to reduce sodium by about 20 mg or more in meals per serving. It may be desirable to reduce sodium to about 800 mg or less, more particularly to about 500 mg or less, more particularly to about 300 mg or less, more particularly to about 100 mg or less, more particularly to about 20 mg or less in meals per serving.
[0019] In embodiments, the one or more taste modulating or salty taste modulating compounds are present in a food product in an amount or concentration sufficient to elicit or enhance the perception of salty taste such that the amount of sodium may be reduced by about 100 mg or more per serving relative to a substantially similar food product that does not have the one or more taste modulating or salty taste modulating compounds while having a similar salty taste. In embodiments, the one or more taste modulating or salty taste modulating compounds are present in a serving of a food product in an amount or concentration sufficient to elicit or enhance the perception of salty taste such that the amount of sodium may be reduced to about 800 mg or less, more particularly to about 500 mg or less, more particularly to about 300 mg or less, more particularly to about 200 mg or less, more particularly to about 100 mg or less relative to a substantially similar food product that does not have the one or more taste modulating or salty taste modulating compounds while having a similar salty taste. By way of example, it may be desirable to reduce sodium by about 100 mg or more in soups per serving. It may be desirable to reduce sodium to about 800 mg or less, more particularly to about 500 mg or less, more particularly to about 300 mg or less, more particularly to about 200 mg or less, more particularly to about 100 mg or less in soups per serving. For soup, a typical serving size is 250 grams. Of course, soups may have other serving sizes.
[0020] Any suitable combination of compounds described herein, or derivatives thereof, may be included in a food product. In embodiments, a food product includes a combination of compounds such that the combination includes at least two structurally diverse taste modulating or salty taste modulating compounds.
[0021] A food product or composition may include one or more compounds described herein, or derivatives thereof, in any suitable concentration. By way of example, a compound described herein, or a derivate thereof, such as a taste modulating or salty taste modulating compound may be present in a food product at a concentration of about 0.01% by weight or greater, about 2% by weight or less, or from about 0.01% by weight to about 2% by weight. It will be understood that the concentration of the salt or salts in the food product may affect the desired concentration of a taste modulating or salty taste modulating compound. For example, if more salt is present, less taste modulating or salty taste modulating compound may be desired. In addition, it will be understood that the presence of more than one taste modulating or salty taste modulating compound may affect the desired concentration of other taste modulating or salty taste modulating compounds, particularly if the effects of the taste modulating or salty taste modulating compounds are additive or synergistic.
[0022] Any salt that imparts a salty taste may be present or incorporated into a food product that contains a bioactive, taste modulating, or salty taste modulating compound. The most commonly used salt for food applications is sodium chloride (typically referred to as common table salt). Other illustrative sources of sodium salts that may be present of incorporated into a food product include sodium phosphates, mono sodium glutamate, sodium nitrite, sodium nitrate, sodium bicarbonate, sodium lactate, sodium citrate, and sodium stearoyl lactylate. Similar lithium, potassium, ammonium or other alkali earth salts may be present or included in addition or as an alternative to one or more sodium salts.
[0023] In embodiments, a food product includes sodium chloride as a salt that imparts a salty taste. Sodium chloride may be present in the food product at any suitable amount or concentration. In embodiments, sodium chloride is present in the food product in an amount up to about 10.0 weight percent, more particularly, up to about 5.0 weight percent, even more particularly up to about 1.2 weight percent, or in the range of about 0.017 to about 1.2 weight percent, or about 0.1 to about 1, or about 0.4 to about 0.6 weight percent. In embodiments, a food product that includes one or more bioactive, taste modulating, or salty taste modulating compounds comprises no more than 0.04 weight percent, no more than 0.1 weight percent sodium, no more than 0.2 weight percent, no more than 0.25 weight percent sodium, no more than 0.3 weight percent, no more than 0.4 weight percent, no more than 0.5 weight percent sodium, no more than 0.75 weight percent sodium, no more than 1 weight percent sodium, no more than 5 weight percent sodium, or no more than 10 weight percent sodium. It will be understood that a desired weight percent of sodium may vary depending on the type of food product. For example, it may be desirable for a seasoning to have a higher weight percent sodium than a soup or a breakfast cereal. In embodiments, a food product that includes one or more taste modulating or salty taste modulating compounds comprises no more than 100 mg sodium per serving, no more than 250 mg sodium per serving, no more than 500 mg sodium per serving.
[0024] One or more taste modulating or salty taste modulating compounds may be utilized in connection with virtually any food product for which it is desired to elicit or enhance the perception of a salty taste or other taste associated with consumption of a salt. The taste modulating or salty taste modulating compounds can find application for imparting saltiness to beverages or food dishes or as an ingredient in snack foods or other food products in which saltiness is desired.
[0025] Examples of food products that may incorporate one or more taste modulating or salty taste modulating compound include a confectionary, a gum, a bakery product, an ice cream, a dairy product, a fruit snack, a chip or crisp, an extruded snack, a tortilla chip or corn chip, a popcorn, a pretzel, a nut, a snack bar, a meal replacement, a ready meal, a soup, a pasta, a canned food, a frozen processed food, a dried processed food, an instant noodle, a chilled processed food, an oil or fat, a sauce dressing or condiment, a dip, a pickled product, a seasoning, a baby food, a spread, a chip or a crisp such as chips or crisps comprising potato, corn, rice, vegetable (including raw, pickled, cooked and dried vegetables), a fruit, a grain, a soup, a seasoning, a baked product such as a ready-to-eat breakfast cereal, hot cereal or dough, an ice cream such as a frozen yogurt, a dairy products such as a yogurt or cheese, ready meal, a soup, a pasta, a canned food, a frozen processed food, a dried processed food, an instant noodle, or a chilled processed food, a beverage including beverages that include fiber or protein a meat or a meat substitute, a pet food, an animal product, a medical food, a nutritional supplement, a vitamin supplement, and an infant formula product.
[0026] In embodiments, one or more bioactive, taste modulating, or salty taste modulating compounds are incorporated into a medicinal or pharmaceutical product, or the like.
[0027] In embodiments, a food product is a processed food product. Food processing includes the transformation of raw ingredients into food or transforming forms of food into other forms of food. Food processing often includes using harvested crops or animal products to produce marketable products sold to consumers at stores, restaurants and the like. Processed food products include products for which additional processing by a consumer occurs after purchase but prior to consumption (e.g., heating, cooking, baking, or the like).
[0028] Particularly suitable food products including soup, meal kits, grain products such as ready-to-eat cereals, snacks, bars and baked dough, and dairy products such as ice cream, yogurt and cheese. In some aspects, a bioactive, taste modulating, or salty taste modulating compound is used to reduce the amount of sodium salt that is typically included in soups, including (but not limited to) chicken or poultry broth, chicken- or poultry-based soups (such as chicken noodle soup), tomato-based soups, and the like. In some aspects, a taste modulating or salty taste modulating compound is used to reduce sodium salt in meal kits, such as kits that include ingredients to be combined with meat to prepare a meal. Such meal kits can include dried components (such as noodles, rice, dried potatoes, or the like) and seasoning packages. In some aspects, a taste modulating or salty taste modulating compound is used to reduce the sodium chloride that is typically added to a snack food to enhance its flavor. Exemplary snack foods include potato chips, corn chips, pretzels, fruit-type snacks, and snack mixes including any mixes of any of these foods with other ingredients (such as cereals).
[0029] In some aspects, a taste modulating or salty taste modulating compound is used to reduce the amount of sodium salt that is typically included in a ready-to-eat cereal or other grain-based food products, such as dough, baked goods, grain snacks, grain bars, or the like. In some aspects, a taste modulating or salty taste modulating compound is used to reduce the amount of sodium salt that is typically included in dairy-based food products, such as fresh or frozen dairy products, which may include yogurt, ice cream, or the like. In some aspects, a taste modulating or salty taste modulating compound is used to reduce the amount of sodium salt that is typically included in packaged meal food products, such as packaged meals that contain rice, potatoes, or vegetables, dry packaged meals, frozen packaged meals, or the like.
[0030] For the purposes of the present disclosure "grain" includes grain and pseudograin. Examples of food grains include corn; sorghum; fonio; millet such as pearl millet, proso millet, finger millet, foxtail millet, Japanese millet, kodo millet and the like; Job's tears; wheat; rice; rye; barley; oat; triticale; wild rice; teff; amaranth; quinoa; buckwheat; and the like.
[0031] A taste modulating or salty taste modulating compound can also be used in connection with soup, broth, sauce (such as basting sauce), various seasoning sauces, ketchup, dressings, and other like foods.
[0032] In embodiments, a food product into which a taste modulating or salty taste modulating compound or composition is included has a water content of about 30% or more by weight. For example, the food product may have a water content of about 35% or more, or about 40% or more by weight. Non-limiting examples of food products that typically have water contents of about 30% or more by weight include soups, beverages, batters and dough.
[0033] In embodiments, a food product into which a taste modulating or salty taste modulating compound or composition is included has a water content of about 50% or more by weight. For example, the food product may have a water content of about 60% or more, or about 70% or more by weight. Non-limiting examples of food products that typically have water contents of about 50% or more by weight include soups and beverages. For example, a soup containing a taste modulating or salty taste modulating compound or composition may contain from about 50% water by weight to about 90% water by weight.
[0034] In embodiments, a food product into which a taste modulating or salty taste modulating composition is included has a water content of about 20% or less by weight. For example, the food product may be incorporated into dry food products that having low water contents. In embodiments, a taste modulating or salty taste modulating food product is included in a dried for as a seasoning. In embodiments, the seasoning comprises, consists essentially of, or consists of one or more taste modulating or salty taste modulating compounds, one or more carriers, and one or more salts.
[0035] A taste modulating or salty taste modulating compound can be employed to elicit the perception of salty taste or enhance the perceived salt taste of any salts used in food or beverage products. The preferred salt taste to be elicited or enhanced by the salty compounds is that of sodium chloride.
[0036] Moreover, a taste modulating or salty taste modulating compound described herein can be used to elicit or enhance the perceived salt taste of known salty tasting compounds that may be used as salt substitutes. Such compounds include amino acids such as cationic amino acids and low molecular weight peptides such as dipeptides and tripeptides. Specific examples of these compounds include arginine hydrochloride, lysine hydrochloride, and lysine-ornithine hydrochloride. These compounds exhibit a salty taste but are typically useful only at low concentrations since they exhibit a bitter flavor at higher concentrations. Ordinarily, these salt-tasting compounds will be used in concentrations in the range of about 1 to about 40 mM, or about 10 to about 30 mM. Thus, it is feasible to reduce the sodium chloride content of a food or beverage product by first formulating a food or beverage with less sodium chloride than is necessary to achieve a desired salt taste and then adding to the food or beverage a taste modulating or salty taste modulating compound described herein in an amount sufficient to enhance the salt taste of the salted food or beverage to reach the desired taste. In addition, sodium chloride content may be further reduced by substituting a salt-tasting cationic amino acid, a low molecular eight dipeptide, or mixtures thereof, for at least a portion of the salt.
[0037] In embodiments, a method includes setting a target salty taste of a food product, including an amount of a salt that imparts a salty taste in the food product, where the amount of the salt does not achieve the target level of salty taste, and including an amount of a salty taste enhancing compound (or more than one salty taste enhancing compounds) to achieve the desired salty taste. In embodiments, a method includes setting a target salty taste of a food product, including an amount of a salty-taste imparting sodium salt in the food product that does not achieve the target level of salty taste, including an amount of a non-sodium salt that imparts a salty taste and an amount of a salty taste enhancing compound (or more than one salty taste enhancing compounds) to achieve the desired salty taste.
[0038] Processing
[0039] A taste modulating or salty taste modulating compound, or derivative thereof, described herein can be added to food products in dry or liquid form. For example, a taste modulating or salty taste modulating compound that is in the liquid form can be prepared by simply dissolving or suspending the compound in an appropriate relative amount in an aqueous liquid. Useful aqueous liquids include water, alcohol-water mixtures, triacetin, propylene glycol, and triglycerides and other known organic solvents. Depending upon the concentration of the taste modulating or salty taste modulating compound, it can be desirable to heat the mixture to dissolve the compound.
[0040] Taste modulating or salty taste modulating compounds that exist in a dry state, such as powders or granules, can be prepared by either mixing or blending the compounds with other components in the dry state. The dry blending or mixing can be carried out in any conventional suitable apparatus. In some aspects, the taste modulating or salty taste modulating compounds described herein can be prepared into dry compositions by commonly used methods of granulation from mixtures of the several ingredients, preferably initially conveniently smaller than forty mesh. Such starting mixtures can be wetted in known manner, granulated, and their granulations dried as usual and screened to give a product approximately the typical size of common table salt, for example, by taking the fraction passing through the thirty mesh screen and retained on the forty mesh screen.
[0041] Taste modulating or salty taste modulating compounds that exist in a dry composition state can be alternatively prepared by first forming a solution, emulsion or suspension of the compounds and other individual components, and then extruding or drying the solution or suspension. The preparation of the solution or suspension of the components can be carried out as described above in the context of preparing the liquid flavoring agents. The thus-prepared solution, emulsion or suspension can then be dried using any conventional suitable apparatus, such as a rotary drier, a drum drier, or a fluidized bed drier or spray drier.
[0042] Taste modulating or salty taste modulating compounds described herein can be prepared by thoroughly mixing the compounds with other components in the indicated proportions until a suitably mixed (for example, homogeneous) product is attained.
[0043] Compositions or formulations containing the taste modulating or salty taste modulating compounds can then be combined with a food product.
[0044] Perception of Saltiness
[0045] In embodiments, a composition that includes a salty taste modulating compound is perceived as imparting a quantity of saltiness equal to a substantially similar composition that does not include the salty taste modulating compound but that has a higher concentration of the salt. Preferably, the composition that includes the salty taste modulating compound imparts a perception of saltiness equal to the substantially similar composition that does not have the salty taste modulating compound when the composition has less salt than the substantially similar composition (e.g., salt reduced by about 1% or more). For example, the composition that includes the salty taste modulating compound may impart a perception of saltiness equal to the substantially similar composition that does not have the salty compound when the composition that includes the salty taste modulating compound has a salt concentration reduced by about 2% or more, about 5% or more, about 7% or more, about 8% or more, about 9% or more, about 10% or more, about 11% or more, about 15% or more, about 20% or more, about 30% or more, about 35% or more, about 40% or more, or about 50% or more, relative to the substantially similar composition. In embodiments, one or more salty taste modulating compounds may be present in a food product in an amount sufficient to reduce the amount of a salt, such as sodium chloride, by about 1% or more, about 2% or more, about 5% or more, about 7% or more, about 8% or more, about 10% or more, about 11% or more, about 12% or more, about 15% or more, about 20% or more, about 22% or more, about 25% or more, about 30% or more, about 35% or more, about 40% or more, about 45% or more, about 50% or more, about 55% or more, about 60% or more, about 65% or more, about 70% or more, about 75% or more, about 80% or more, about 85% or more, about 90% or more, about 95% or more, or the like. Preferably, the reduced salt food product elicits the same or similar perception of saltiness as a substantially similar food product that does not include the one or more salty taste modulating compounds.
[0046] Perception of saltiness may be evaluated in any suitable manner. In embodiments, saltiness is determined by a trained analytical sensory panel. In embodiments, the trained sensory panel determines the saltiness of a composition having a salty taste modulating compound relative to a substantially similar composition having increased sodium chloride content.
[0047] Sensory panelists may be trained in any suitable manner. Preferably, the panelists are trained to discern salty taste or other attributes without reference to liking or acceptability. The panelists are also preferably trained to accurately quantify salty taste or other attributes according to an intensity scale. General information that may be helpful in understanding beneficial training protocols can be found in, for example, Sensory Evaluation Techniques, 4.sup.th Ed by Meilgaard M., Civille G. V. and Can B. T (2007), CRC Press, pages 147-152. Prescreening, selection, and training of panelists may be occur as described in one or more standards, such as Hootman R C, Manual 13 MNL13 Manual on Descriptive Analysis Testing for Sensory Evaluation, ASTM (1992); STP758 Guidelines for the Selection and Training of Sensory Panel Members, ASTM (1981); and Munoz A. M and Civille, G. V., MLN13: The Spectrum Descriptive Analysis Method, ASTM (1992). Preferably panelists are trained according to the Spectrum Method (Munoz A. M and Civille, G. V., MLN13: The Spectrum Descriptive Analysis Method, ASTM 1992).
[0048] Preferably, average scores regarding saltiness from more than one panelist trained to discern salty taste or other attributes using the same training are considered in determining whether a reduced salt food product elicits the same or similar perception of saltiness as a substantially similar food product that does not include the one or more salty taste modulating compounds. For example, a panel may contain three or more trained panelists, 5 or more trained panelists, 7 or more trained panelists, 10 or more trained panelists, or the like.
[0049] A taste modulating or a salty taste modulating compound may be a compound that directly acts to elicit or enhance the perception of salty taste of a salt or may be a compound that is converted, when ingested, into a compound that directly acts to elicit enhance the perception of salty taste of the salt.
[0050] Taste Modulating, and Salty Taste Modulating Compounds
[0051] A variety of naturally-derived compounds were tested in vitro for their ability to activate or facilitate activation of a TrpML3 channel, a TrpV1 channel or an ENaC channel.
[0052] The TrpML3 (transient receptor potential cation channel, mucolipin subfamily, member 3) channel, also known as Mucolipin-3 is a protein that, in humans, is encoded by the MCOLN3 gene. The TrpV1 (transient receptor potential cation channel subfamily V member 1) channel, also known as the capsaicin receptor and the vanilloid receptor 1, is a protein that, in humans, is encoded by the TrpV1 gene. The ENaC (epithelial sodium channel), also known as sodium channel non-neuronal 1 (SCNN1) or amiloride sensitive sodium channel (ASSC) is a membrane-bound ion-channel that is permeable for Li+-ions, protons and especially Na+-ions.
[0053] Any compound that interacts with one or more of the TrpML3 channel, the TrpV1 channel and the ENaC channel may be useful for modulating taste or saltiness of a food product into which the compound is incorporated.
[0054] It is estimated that natural products, extracts, and isolated compounds that collectively contained about 2,000,000 potential taste modulating or salty taste modulating compounds were tested for sodium channel activity. About 600 of the 2,000,000 compounds had some level of sodium channel activity. About 300 of the 600 compounds had an increased threshold level of activity. Further analysis, including structure based toxicological analysis, resulted in 99 initial compounds being selected as candidates for taste modulating or salty taste modulating compounds. Presented herein are naturally-derived compounds and compound classes that have been identified as acting at one or more of these channels, or that otherwise may function as bioactive, taste modulating, or salty taste modulating compounds.
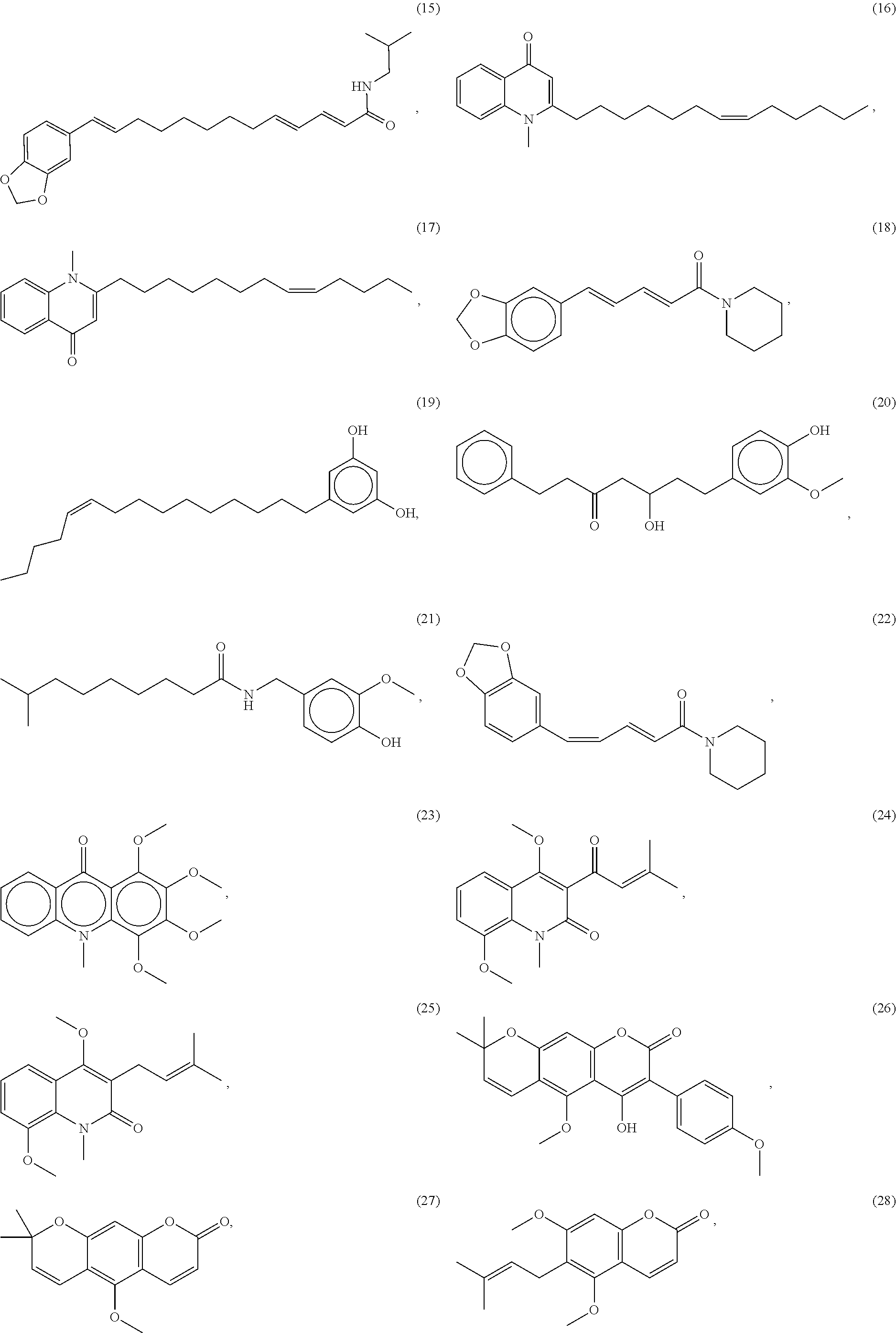
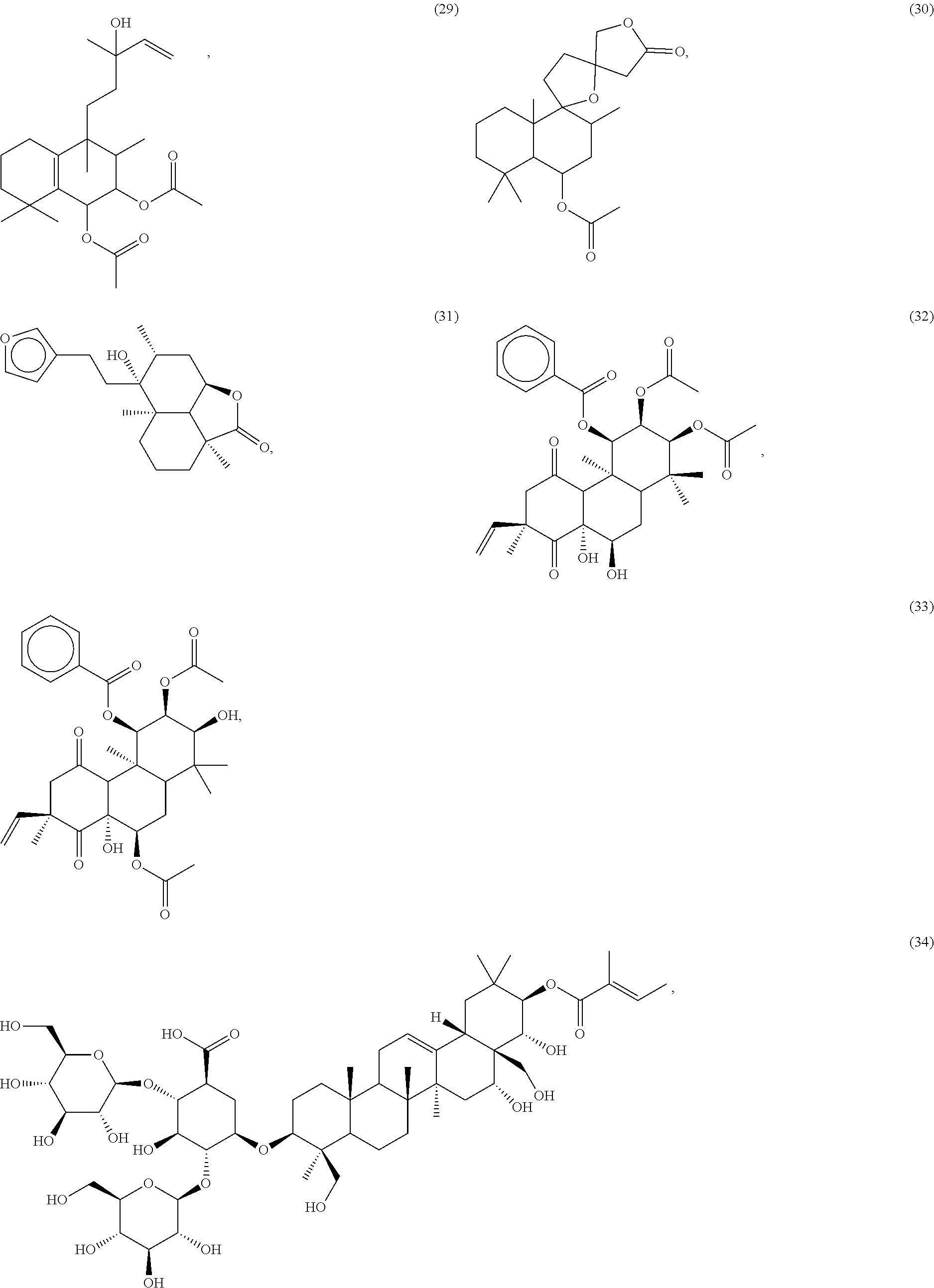
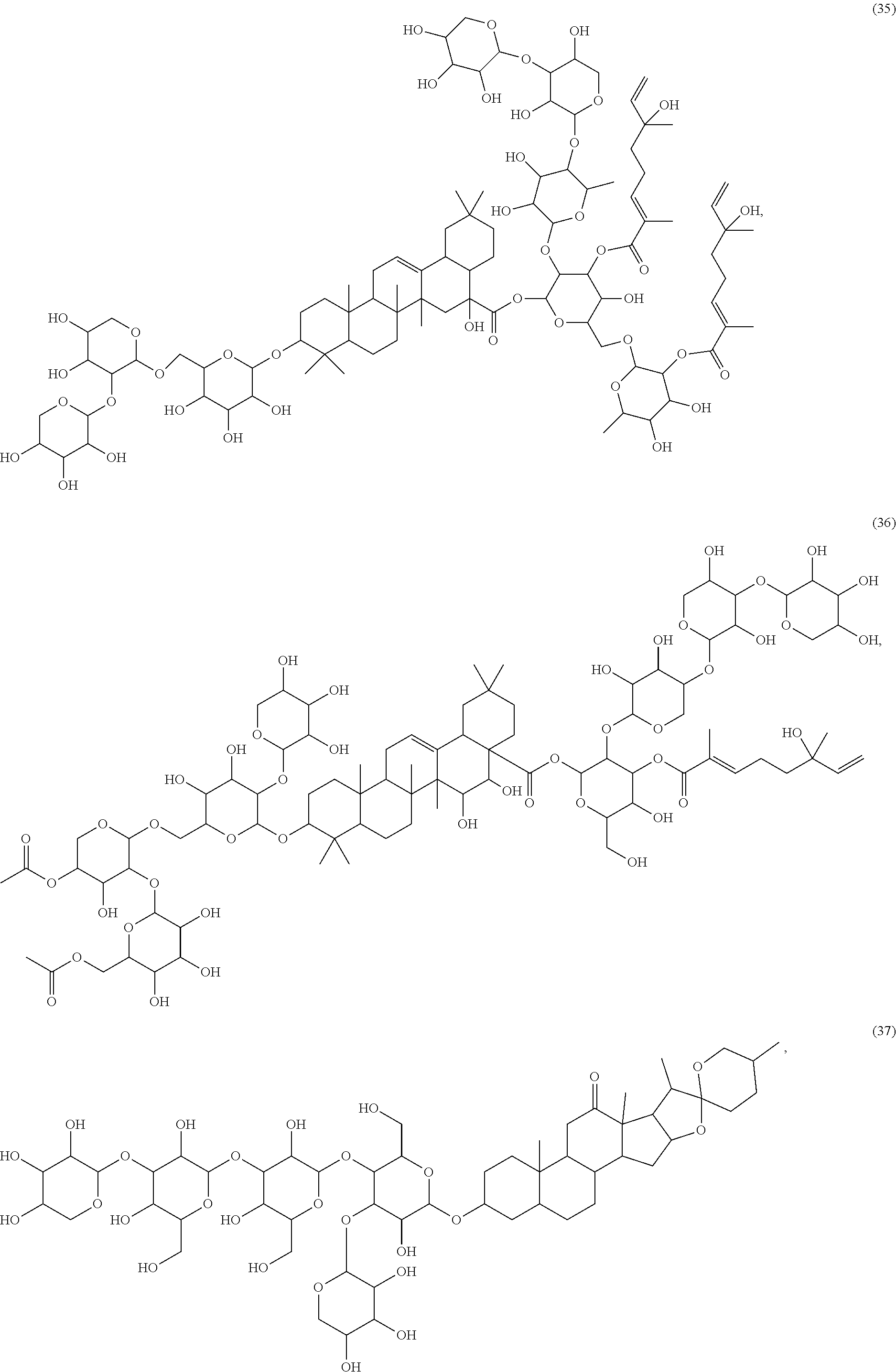
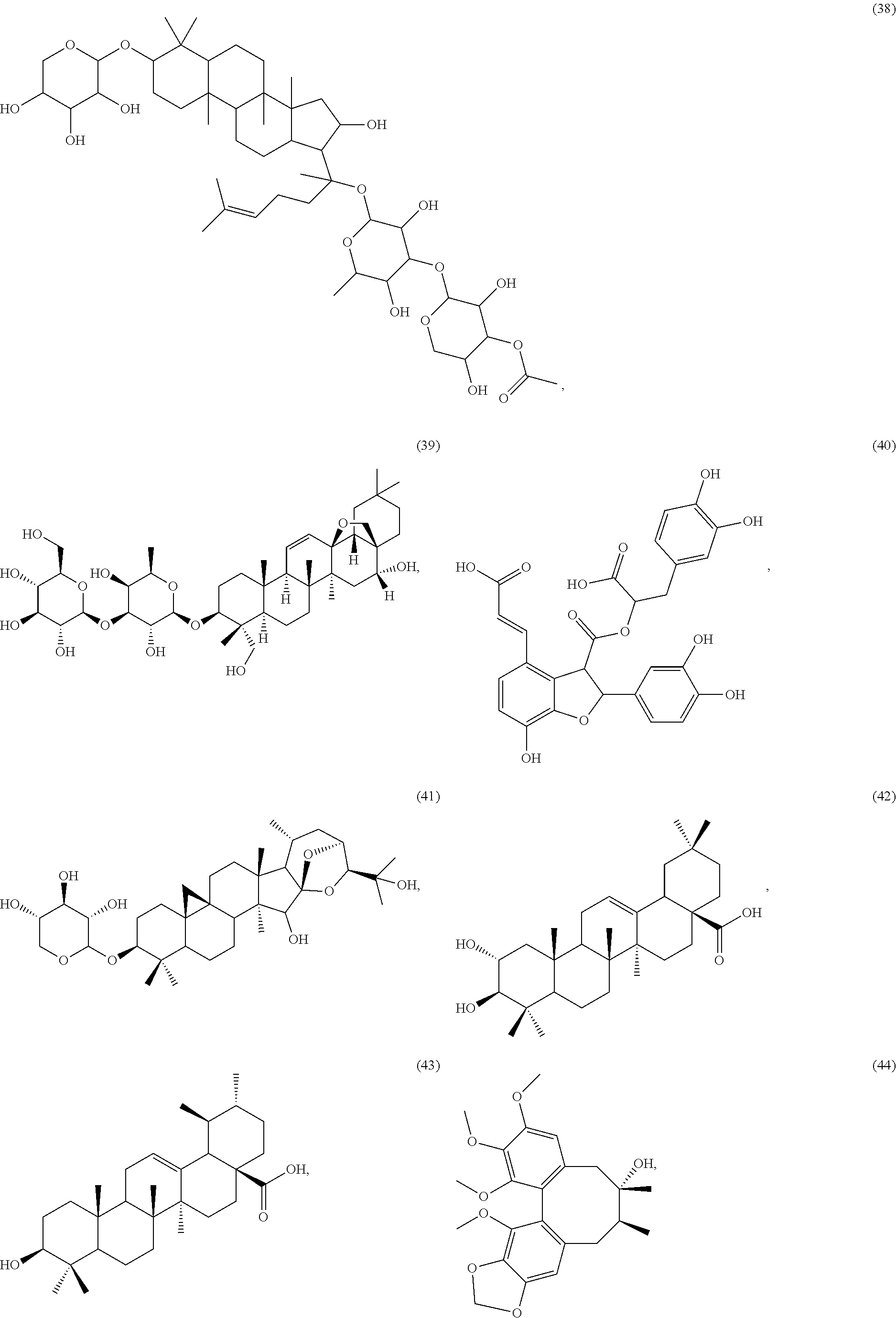
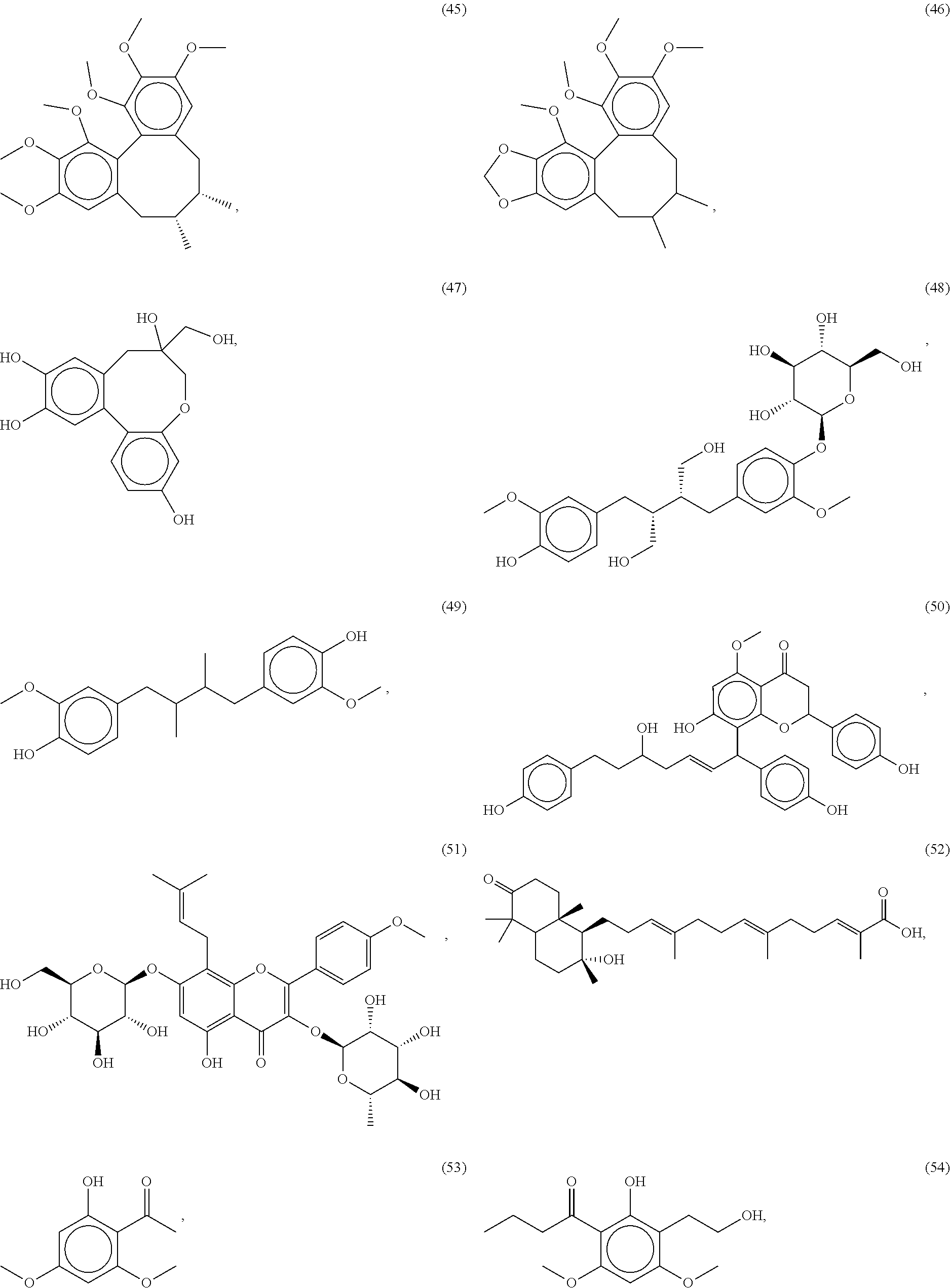
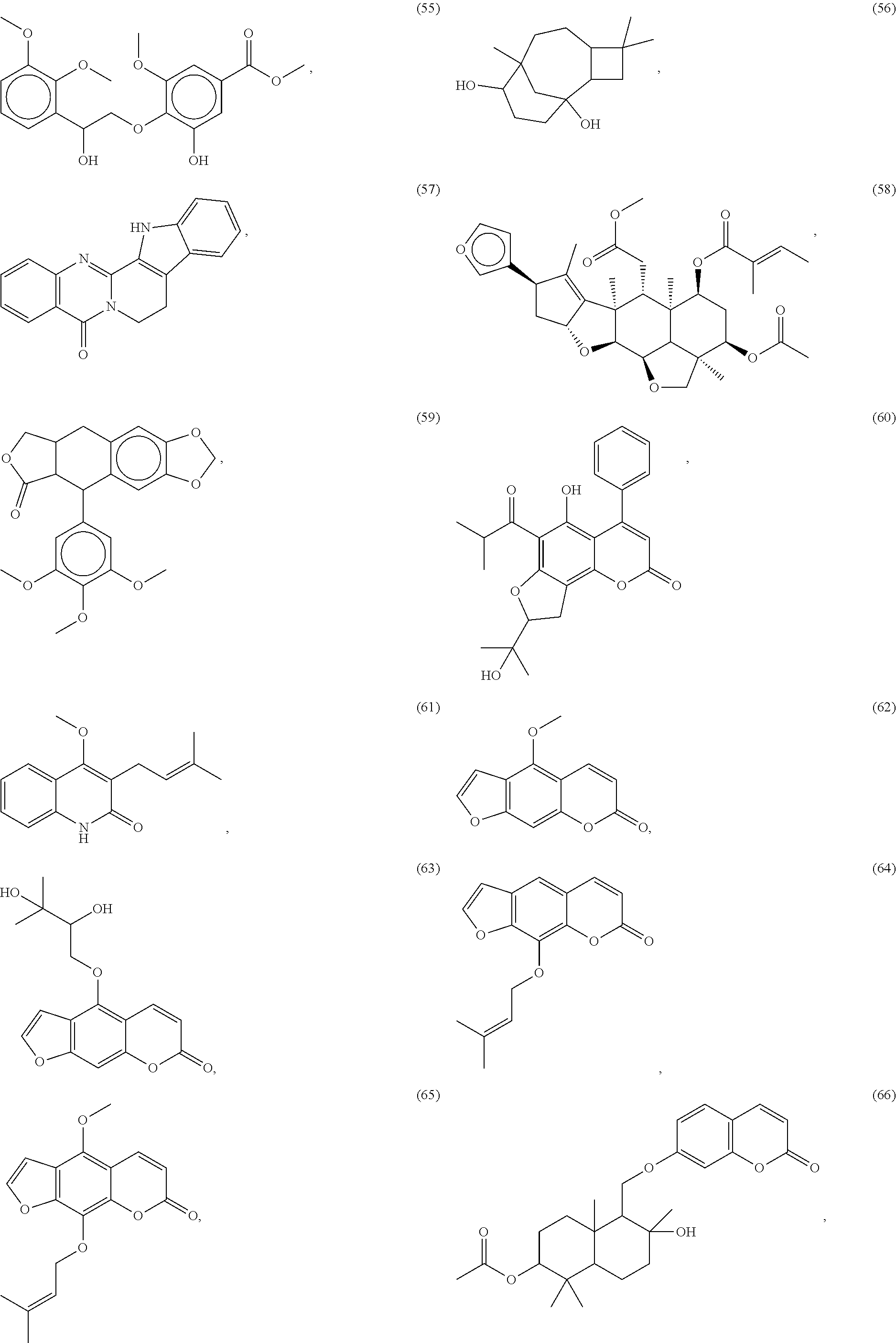
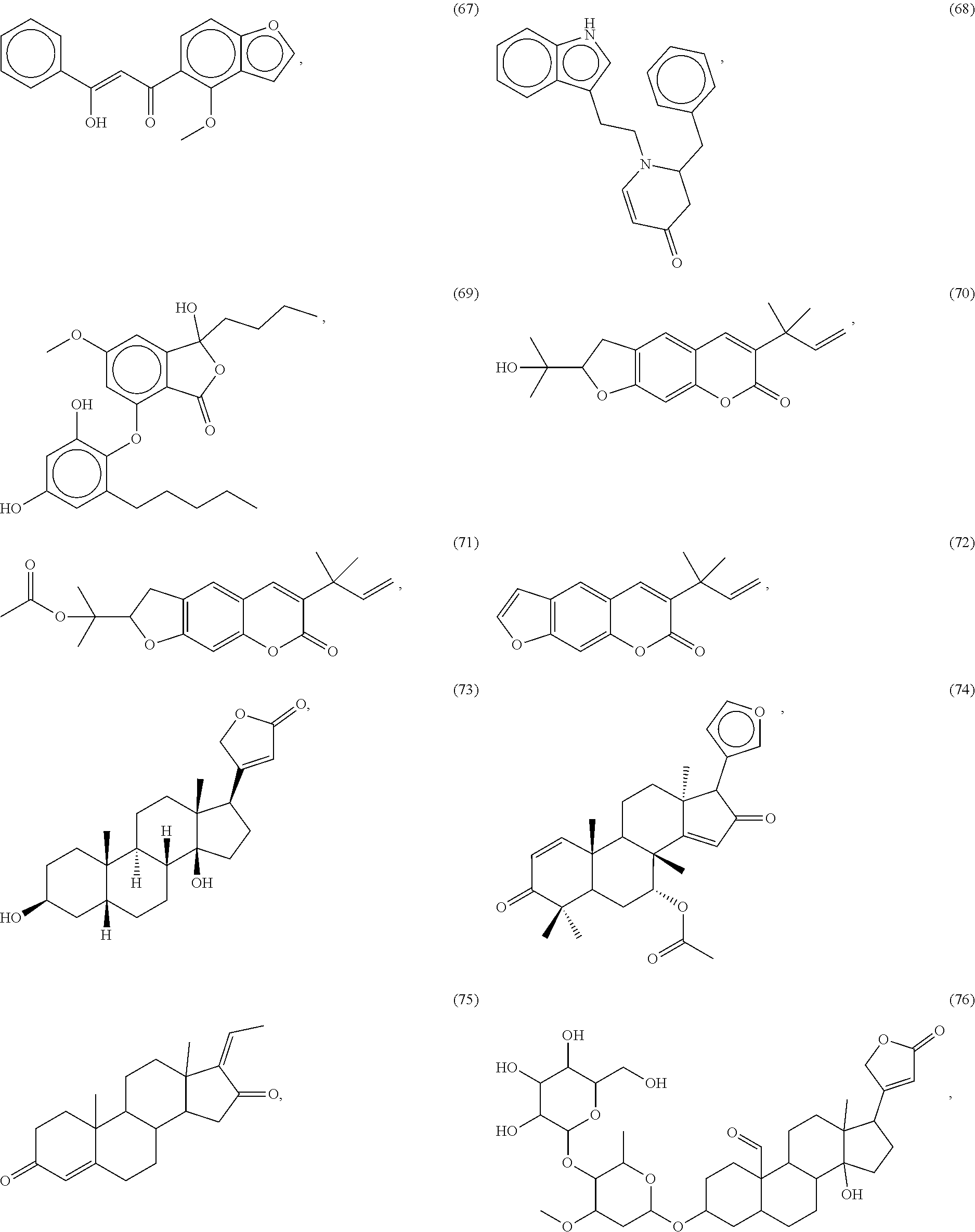
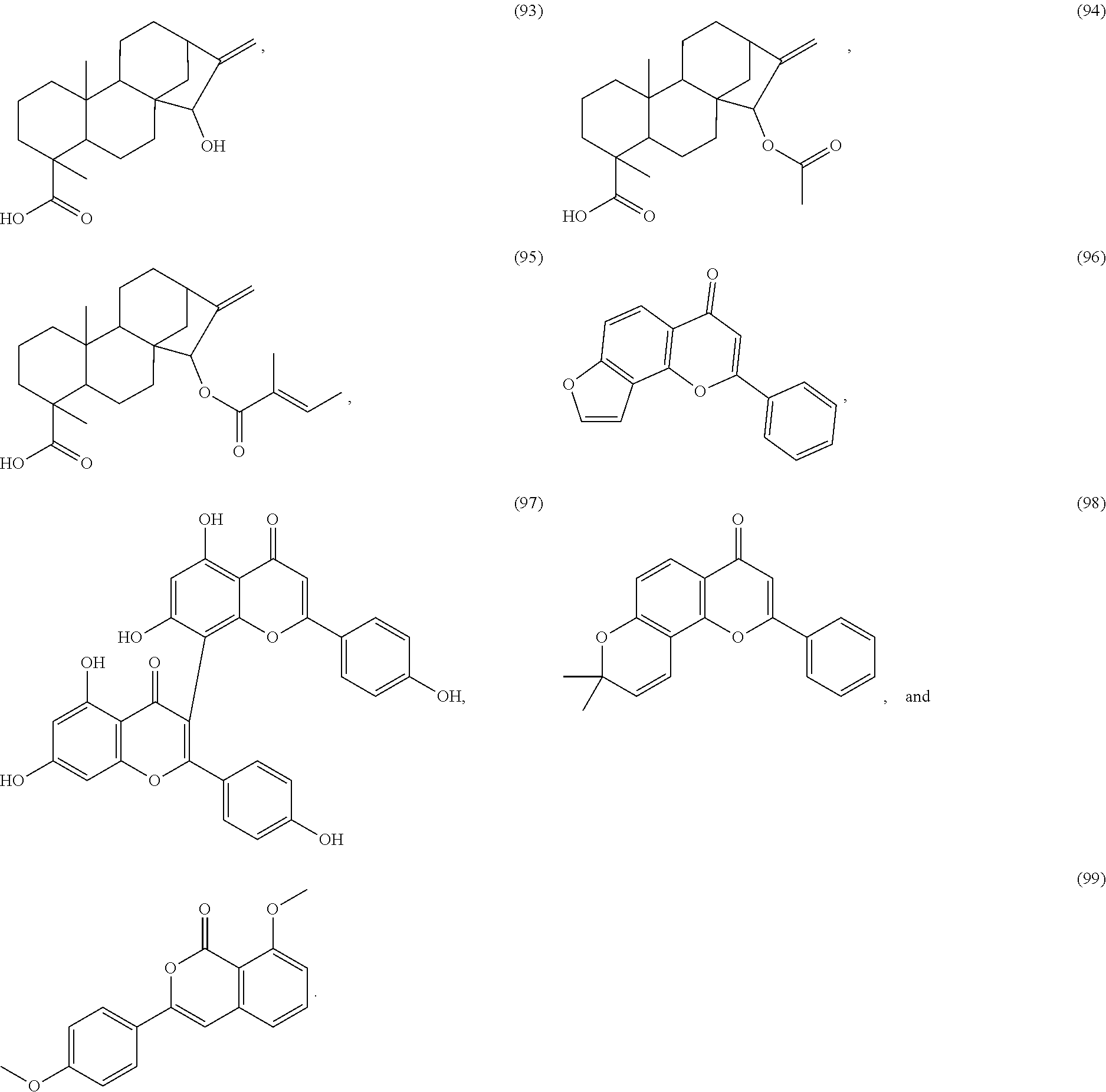
[0055] A listing of the 99 initially selected compounds (Cmpd), as well as the common names (where known), Chemical Abstract Service (CAS) Registry numbers where known (CAS-RN), Sources/Taxons (where known) from which the compounds were isolated (Source/Taxon) and common name of the sources (Common Name), is presented in Table 1 below. The structures of the compounds are also presented herein. To the extent the structures conflict with other information provided, the structures of the 99 initially selected compounds should be considered determinative.
TABLE-US-00001 TABLE 1 Information regarding initially selected compounds Cmpd CAS-RN Common name Source/Taxon Common Name 1 104264-55-3 12-Gingerol Zingiber officinalis Ginger 2 36752-54-2 [10]-Shogaol Zingiber officinalis Ginger 3 143615-75-2 [6]-Gingerdiacetate Zingiber officinalis Ginger 4 555-66-8 6-Shogaol Aframomum meleguata, Grains of paradise; Zingiber officinalis Ginger 5 39886-76-5 [6]-Gingerol Zingiber officinalis Ginger 6 53172-04-6 [7]-Paradol Aframomum meleguata, Grains of paradise; Zingiber officinalis Ginger 7 27113-22-0 [6]-Paradol or Aframomum meleguata, Grains of paradise; [6]-Gingerone Zingiber officinalis Ginger 8 626200-64-4 5-methoxy-[6]-Gingerol Aframomum meleguata, Grains of paradise; Zingiber officinalis Ginger 9 23513-08-8 8-Gingerol Zingiber officinalis Ginger 10 1083195-12-3 a pentadecatrienyl-1,3-benzenediol Embelia ribes False Black Pepper, white-flowered Embelia 11 79559-60-7 a 1-Ph-4-hepten-3-one Kaempferia galanga Kencur, aromatic ginger, sand ginger, cutcherry, resurrection lily 12 79559-61-8 a 1-Ph-5-OH-3-heptanone Kaempferia galanga Kencur, aromatic ginger, sand ginger, cutcherry, resurrection lily 13 205687-01-0 Capsiate Capsiate 14 147030-09-9 Pipersintenamide Piper longum Long pepper, Indian long pepper 15 55038-30-7 Guineensine Piper longum Long pepper, Indian long pepper 16 182056-19-5 Evodia rutaecarpa Evodia fruit 17 15266-38-3 Evocarpine Evodia rutaecarpa Evodia fruit 18 94-62-2 Piperine Piper longum Long pepper, Indian long pepper 19 52483-20-2 Irisresorcinol Ardisia silvestris 20 79559-61-8 a 1-Ph-5-OH-3-heptanone Alpinia officinarum Lesser galangal (ginger family) 21 19408-84-5 Dihydrocapsaicin Capsicum annuum Serrano pepper 22 30511-77-4 Isochavicine Piper longum, Long pepper, Piper nigrum Indian long pepper; black pepper 23 517-73-7 Melicopicine Teclea trichocarpa 24 Zanthoxylum esquirolii 25 41303-25-7 O-Methylglycosolone Zanthoxylum esquirolii 26 5307-59-5 Robustic acid Derris robusta 27 84-99-1 Xanthoxyletin Toddalia asiatica; Orange climber Millettia pulchra 28 4335-12-0 Toddaculin Toddalia asiatica Orange climber 29 351427-18-4 Vitetrifolin D Vitex agnus Vitex, Chaste Tree, Chasteberry, Abraham's Balm, Monk's Pepper 30 61263-52-3 Vitex agnus Vitex, Chaste Tree, Chasteberry, Abraham's Balm, Monk's Pepper 31 465-92-9 Marrubin Marrubium vulgare White Horehound, Common Horehound 32 238088-78-3 Orthosiphol I Orthosiphon stamineus Cat whiskers 33 345905-36-4 Orthosiphol M Orthosiphon stamineus Cat whiskers 34 254896-53-2 Aesculioside A Aesculus hippocastaneum Horse-chestnut, conker tree 35 Gleditschia australis Locust tree 36 1383715-41-0 Pithecoctenium echinatum Monkey comb 37 Yucca gloriosa Spanish dagger 38 Nephelium cuspidatum Bayong 39 20874-52-6 Saikosaponin D Bupleurum falcatum Chinese throroughwax; Sickle hare's ear 40 1217879-76-9 a 3-benzofurancarboxylic Salvia miltiorrhiza Red sage, Chinese ester sage, tan shen, or danshen 41 27994-11-2 Cimigoside; Cimicifuga racemosa Black cohosh, b-D-Xylopyranoside black bugbane, black snakeroot, fairy candle 42 4373-41-5 Maslinic acid, Alchemilla xanthochlora Lady's Mantle Crategolic acid 43 77-52-1 Ursolic acid Lavandula officinalis Lavender, English lavender, common lavender, true lavender, narrow-leaved lavender 44 58546-54-6 Schizandrol B; Schisandra chinensis Five flavor berry Gomisin A 45 61281-38-7 Schisandrin A, Schisandra chinensis Five flavor berry Deoxyschizandrin 46 61281-37-6 Schizandrin B Schisandra chinensis Five flavor berry 47 102036-29-3 Protosappanin B 48 129102-89-2 Secoisolariciresinol; Angelica archangelica Garden Angelica, Secoisolariciresin-4-yl Holy Ghost, Wild b-D-glucopyranoside Celery, Norwegian angelica 49 66322-34-7 Dihydroguaiaretic acid Schisandra chinensis Five flavor berry 50 193816-85-2 Epicalyxin C Alpinia katsumadai Greater galangal 51 181490-70-0 Icariin Angelica sinensis dong quai or female ginseng 52 446030-43-9 Myrrhanone B Commiphora mukul Guggul, Indian Bdellium 53 936499-55-7 Brevifolin Zanthoxylum piperitum Japanese pepper, Japanese pricklyash 54 1083202-45-2 Fungus Strain code: 01469fxxx000005 55 Erythrina variegata Tiger's claw or Indian coral tree 56 155485-76-0 Senecrassidiol Psidium guajava Apple guava or common guava 57 84-26-4 Rutaecarpine Evodia rutaecarpa Evodia fruit 58 992-20-1 Salannin Azadirachta indica Neem, Nimtree and Indian Eilac 59 69222-20-4 Isoanthricin Podophyllum peltatum May Apple, hogapple, Indian apple, mayflower, umbrella plant, wild mandrake, American mandrake 60 850494-43-8 Mammea A/AD cyclo Mesua ferrea Ceylon ironwood, F Indian rose chestnut, or Cobra's saffron 61 7282-19-1 Atanine Zanthoxylum piperitum Japanese pepper, Japanese pricklyash 62 484-20-8 Bergapten Petroselinum stativum Parsley 63 133164-11-1 Prangol Petroselinum stativum Parsley 64 482-44-0 Imperatorin Petroselinum stativum Parsley 65 2543-94-4 Phellopterin Petroselinum stativum Parsley 66 497226-80-9 terpenoid coumarins Ferula assa-foetida Giant fennel, asant, food of the gods, jowani badian, stinking gum, Devil's dung 67 484-33-3 Pongamol Millettia pulchra 68 artificial 69 1176891-50-1 Sakisacaulon A Lichen 70 1268481-32-8 Chalepin 71 1092383-76-0 Rutamarin 72 13164-03-9 Halepensin 73 143-62-4 Digitoxigenin Xysmalobium undulatum Uzara 74 26241-51-0 Azadiradione Azadirachta indica Neem, Nimtree, Indian Lilac 75 95975-55-6 (Z)-Guggulsterone Commiphora mukul Guggul, Indian Bdellium 76 1941-73-7 Apobioside Apocynum cannabinum Dogbane, Amy Root, Hemp Dogbane, Indian Hemp, Rheumatism Root, Wild Cotton 77 3751-87-9 Apocannoside Apocynum cannabinum Dogbane, Amy Root, Hemp Dogbane, Indian Hemp, Rheumatism Root, Wild Cotton 78 86894-26-0 Hebelomic acid A Hebeloma senescens mushroom methyl ester 79 2221-82-1 .beta.-Cyclocostunolide Critonia morifolia 80a 546-43-0 Alantolactone Inula helenium Elecampane, horse-heal, marchalan 80b 470-17-7 Isoalantolactone Inula helenium Elecampane, horse-heal, marchalan 81 a 2-octenyl-3-hydroxy-1,5-pentanedioic Fungus Strain code acid 02295fxxx000001 82 83797-45-9 16-Heptadecene-1,2,4-triol; Persea gratissima Avocado Avocadene 83 1356361-43-7 16-Heptadecene-1,2,4-triol, 1,4-diacetate Persea gratissima Avocado 84 16423-52-2 N-Decyl acetamide Bacteria Strain code 0172axxx000002 85 21402-68-6 9-hydroxy-10,12,15-Octadecatrienoic Marrubium vulgare White Horehound, acid Common Horehound 86 167936-49-4 12-hydroxy-9,13,15-Octadecatrienoic Petroselinum stativum Parsley acid 87 Ricinus communis Castor oil plant 88 15514-85-9 Dimorphecolic acid Podophyllum peltatum May Apple, hogapple, Indian apple, mayflower, umbrella plant, wild mandrake, American mandrake 89 463-40-1 Linolenic acid Mesua ferrea Ceylon ironwood, Indian rose chestnut, Cobra's saffron 90 35949-86-1 Gingerglycolipid C Zingiber officinalis Ginger 91 187218-23-1 Capsianoside E Capsicum annuum Serrano pepper 92 131580-15-9 Capsianoside D Capsicum annuum Serrano pepper 93 22338-69-8 Grandifloric acid Aralia cordata, Spikenard, udo Espeletia spp. 94 6619-97-2 Xylopic acid Xylopia aethiopica Bitterwood 95 32381-03-6 Angeloylgrandifloric acid Sideritis hirsuta Hairy ironwort 96 482-00-8 Lanceolatin B 97 101140-06-1 Biapigenin Fagopyrum esculentum, Hypericum perforatum 98 64125-32-2 Millettia pulchra 99 36640-12-7 Lichen
[0056] Compounds 12 and 20 are the same compound isolated from different sources.
[0057] The CAS registry numbers presented in Table 1 above reflect a compound or an isomer thereof. It will be understood that other isomers may have other CAS registry numbers. Further, the structures presented herein, to the extent that they show stereochemistry may not match the particular isomer of the CAS registry number presented in Table 1.
[0058] Those compounds for which no CAS registry numbers are provided in Table 1, as well as those for which registry numbers are provided, may be isolated or purified in any suitable manner. For example, the natural source of the compound, which is presented in Table 1, may be fractionated and the fractions subjected to chromatography, such as gas chromatography or HPLC, or other suitable separation process to isolate or purify the compound. The selection of, for example, a chromatography column and parameters can be readily identified based on the chemical structure of the compound. To facilitate isolation or purification or for verification, obtained fractions, subfractions, or individual compounds may be tested for ability to activate a sodium channel, for example, expressed in cells in culture, cell membrane, or the like and employing an appropriate assay, such as an electrophysiological assay, a colorimetric assay, or the like.
[0059] Alternatively or in addition, the compounds listed in Table 1 may be synthesized. Alternatively or in addition, companies that have access to the appropriate natural sources or the ability to test for sodium channel activity may be contracted to isolate the compounds. Companies that have access to natural products or natural product libraries that may include sources presented in Table 1 or that have expertise in development of assays for identification of compounds or fractions containing compounds capable of activating a sodium channel include Biotechnology Research And Information Network AG (Zwingengerg, Germany); AnalytiCon Discovery, GmbH (Potsdam, Germany); Albany Molecular Research, Inc. (Albany, N.Y., USA); Axxam SpA (Milan, Italy); Boulder BioPharmaceuticals, LLC, Boulder, Colo.; ChromaDex (Irvine, Calif., USA); Enzo Life Sciences, Inc. (Farmingdale, N.Y., USA); IMD Natural Solutions GmbH (Dortmund, Germany); TimTec LLC (Newark, Del., USA); and The Natural Products Discovery Institute (Doylestown, Pa., USA).
[0060] The structures of the initially selected compounds are as follows:
##STR00001## ##STR00002## ##STR00003## ##STR00004## ##STR00005## ##STR00006## ##STR00007## ##STR00008## ##STR00009## ##STR00010## ##STR00011##
[0061] Presented below are chemical compound structures based on groupings of structural relationships of the 99 initially selected compounds. The compounds are grouped into 21 categories (A through U) containing one or more of the 99 initially selected compounds. Within some categories, sub-categories are described.
[0062] In some instances, a compound may be in more than one category due to its structural similarity to compounds in more than one category. It will be understood that structural similarities of the various compounds other than those presented herein exist and that groupings into categories other than those presented herein are possible and contemplated.
[0063] Each of the 22 categories of compounds presented herein is discussed independently. That is, discussion of substituents with regard to one category should not be construed to limit discussion of substituents with regard to another category. For example, R.sup.1 for the group A compounds is independently defined relative to R.sup.1 for the group B compounds. In addition, discussion of substituents with regard to subgroups is independently defined. For example, R.sup.1 for the group J1 compounds is independently defined relative to R.sup.1 for the group J2 compounds, unless otherwise stated.
[0064] Group a Compounds
[0065] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00012## [0066] where: [0067] R.sup.1 is H or C.sub.1-C.sub.10 alkyl; [0068] R.sup.2 is H or C.sub.1-C.sub.3 alkyl; [0069] X is CHOR.sup.3 or C.dbd.O;
[0069] ##STR00013## [0070] R.sup.3 is H, C.sub.1-C.sub.3 alkyl, or [0071] R.sup.4 is H or C.sub.1-C.sub.3 alkyl; [0072] Y is CR.sup.5.dbd.CH or CHR.sup.5--CH.sub.2; [0073] R.sup.5 is H, OH, --OCH.sub.3, --OCH.sub.2CH.sub.3, --O--OCH.sub.2CH.sub.2CH.sub.3, or
[0073] ##STR00014## [0074] and [0075] R.sup.6 is H or C.sub.1-C.sub.3 alkyl.
[0076] In embodiments, R.sup.1 is C.sub.2-C.sub.8 alkyl. In embodiments, R.sup.2 is H. In embodiments, X is C.dbd.O or
##STR00015##
where R.sup.4 is CH.sub.3. In embodiments, when Y is CR.sup.5.dbd.CH, R.sup.5 is H. In embodiments, when Y is CHR.sup.5--CH.sub.2, R.sup.5 is OH or --OCH.sub.3. In embodiments, R.sup.6 is CH.sub.3.
[0077] Group B Compounds
[0078] Group B1 Compounds
[0079] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00016##
[0080] where: [0081] R.sup.1 and R.sup.2 are each independently OH or C.sub.1-C.sub.3 alkoxy or where R.sup.1 and R.sup.2 together with the carbons to which they are bound form a five-membered ring having two oxygen heteroatoms to form a compound having the following structure
##STR00017## ##STR00018##
[0082] Group B2 Compounds
[0083] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00019##
[0084] where R.sup.1 is C.sub.10-C.sub.15 alkyl or alkenyl.
[0085] Group B3 Compounds
[0086] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00020##
[0087] where: [0088] R.sup.1 and R.sup.2 are each independently OH or C.sub.1-C.sub.3 alkoxy, and [0089] R.sup.3 is selected from the group consisting of [0090] (i) C.sub.10-C.sub.20 unsubstituted straight or branched chain alkenyl with one or more double bonds; and [0091] (ii)
[0091] ##STR00021## [0092] where X, Y and Z are independently CH, CH.sub.2, CO or CHOR.sup.5 where R.sup.5 is H or C.sub.1-C.sub.3 alkyl, provided that if one of X or Y are CH then Z is also CH, and where R.sup.4 is C.sub.1-C.sub.8 straight or branched chain unsubstituted alkyl or
[0092] ##STR00022## [0093] where n is 1-5, provided that if R.sup.4 is C.sub.1-C.sub.8 straight or branched chain alkyl, then Y is CHOR.sup.5 where R.sup.5 is C.sub.1-C.sub.3 alkyl.
[0094] In embodiments, R.sup.1 and R.sup.2 are OH. In such embodiments, R.sup.1 may be substituted at the 5 position, R.sup.2 may be substituted at the 3 position, and R.sup.3 may be substituted at the 1 position.
[0095] In embodiments, R.sup.3 is C.sub.10-C.sub.20 unsubstituted straight or branched chain alkenyl with one or more double bonds. In such embodiments, R.sup.3 may be C.sub.12-C.sub.18 unsubstituted straight or branched chain alkenyl. For example, R.sup.3 may be C.sub.13-C.sub.17 unsubstituted straight or branched chain alkenyl, such as C.sub.15 unsubstituted straight or branched chain alkenyl. In embodiments, R.sup.3 has 1-5 double bonds. For example, R.sup.3 may have 1-4 double bonds, such as 1-3 double bonds. In embodiments, R.sup.3 is a straight chain alkenyl. By way of example, R.sup.3 may be
##STR00023##
[0096] In embodiments, R.sup.1 is C.sub.1-C.sub.3 alkoxy, such as methoxy, and R.sup.2 is OH. In such embodiments, R.sup.1 may be substituted at the 3 position, R.sup.2 may substituted at the 4 position, and R.sup.3 may substituted at the 1 position.
[0097] In embodiments, R.sup.3 is
##STR00024##
In such embodiments, R.sup.4 may be
##STR00025##
In embodiments, n is 2. In embodiments, Z is CH or CH.sub.2, X and Z are each independently CH or CH.sub.2, depending on whether Z is CH or CH.sub.2. In embodiments, X is CHOR.sup.5. R.sup.5 may be H. In embodiments, Y is CO. In embodiments, X is CO. By way of example, R.sup.3 may be
##STR00026##
[0098] In embodiments, Y is CHOR.sup.5. R.sup.5 may be C.sub.1-C.sub.3 alkyl. For example, R.sup.5 may be methyl. In some embodiments, where Y is CHOR.sup.5, R.sup.4 is C.sub.1-C.sub.8 straight or branched chain alkyl. For example, R.sup.4 may be C.sub.4-C.sub.6 straight or branched chain alkyl, such as C.sub.5 straight or branched chain alkyl. IN embodiments, R.sup.4 is a straight chain alkyl.
[0099] In embodiments, R.sup.3 is
##STR00027##
[0100] In embodiments, R.sup.3 is selected from the group consisting of:
##STR00028##
[0101] Group B4 Compounds
[0102] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00029## [0103] where: [0104] A and B are each independently NCH.sub.3 or C(O), with the proviso that one of A and B is NCH.sub.3 and the other of A and B is C(O); and [0105] R.sub.1 and R.sub.2 are independently selected from H or C.sub.8-C.sub.16 unsaturated alkyl, with the proviso that if one of R.sub.1 and R.sub.2 is H, then the other of R.sub.1 and R.sub.2 is C.sub.8-C.sub.16 unsaturated alkyl.
[0106] In embodiments, R.sub.1 and R.sub.2 are independently selected from H or C.sub.8-C.sub.16 unsaturated alkyl, wherein the C.sub.8-C.sub.16 unsaturated alkyl contains 1 to 3 double bonds. In embodiments, R.sub.1 and R.sub.2 are independently selected from H or C.sub.8-C.sub.16 unsaturated alkyl, wherein the C.sub.8-C.sub.16 unsaturated alkyl contains only 1 double bond.
[0107] In embodiments, B is NCH.sub.3 and R.sub.2 is C.sub.8-C.sub.16 unsaturated alkyl.
[0108] In embodiments, R.sub.1 and R.sub.2 are independently selected from H or C.sub.11-C.sub.15 unsaturated alkyl, such as C.sub.13 unsaturated alkyl.
[0109] Group C Compounds
[0110] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00030##
[0111] where: [0112] X is C or N; [0113] R.sup.1 is H, OH, C.sub.1-C.sub.3 alkoxy, or C.sub.1-C.sub.6 alkyl; [0114] R.sup.2 and R.sup.3 are each independently selected from H; OH; C.sub.1-C.sub.3 alkoxy; straight or branched chain, saturated or unsaturated C.sub.1-C.sub.6 alkyl or alkenyl; or R.sup.1 and R.sup.2 together with the carbons to which they are bound form a part of a five or six membered ring structure; [0115] R.sup.4 is H or C.sub.1-C.sub.3 alkyl; [0116] R.sup.5 is H, OH, C.sub.1-C.sub.3 alkoxy or C.sub.1-C.sub.3 alkyl; [0117] A and B are each independently selected from CH, C.dbd.O, C-benzyl methoxy, C--CH.sub.2--R.sup.6 or C--C(O)R.sup.6 where R.sup.6 is straight or branched chain, saturated or unsaturated C.sub.1-C.sub.6 alkyl, or A and B together are part of an aromatic six-membered ring structure sharing a side with the remainder of the structure of Formula (C); and [0118] Y is O, CH, C.dbd.O, or C--O--R.sup.7, where R.sup.7 is H or C.sub.1-C.sub.3 alkyl.
[0119] In embodiments, when X is N, R.sup.5 is C.sub.1-C.sub.3 alkyl, such as methyl. In embodiments, when X is N, Y is C.dbd.O or C--O--R.sup.7, such as C--O-Me. In embodiments when X is C, R.sup.5 is H or OH. In embodiments when X is C, Y is O. In embodiments, R.sup.1 is H or methoxy. In embodiments, one of A or B is C.dbd.O and the other is H, C-benzyl methoxy, C--CH.sub.2CHC(CH.sub.3).sub.2 or C--C(O)CHC(CH.sub.3).sub.2.
[0120] In embodiments, R.sup.2 and R.sup.3 together with the carbons to which they are bound form a part of a six membered ring structure. In embodiments, the six membered ring structure includes an oxygen or nitrogen heteroatom. In embodiments, the six membered ring structure contains one or more carbon atoms substituted with one or more C.sub.1-C.sub.6 alkyl, such as methyl. In embodiments, one carbon atom of the ring structure is substituted with two methyl groups. In embodiments, the ring structure is an unsubstituted six carbon aromatic ring structure.
[0121] In embodiments, a compound according to Formula (C) has the following structure:
##STR00031##
where A, B, X, Y, R', R.sup.4 and R.sup.5 are as described above for Formula (C).
[0122] In embodiments, a compound according to Formula (C) has the following structure:
##STR00032## [0123] where: [0124] R.sup.1, R.sup.2 and R.sup.3 are each independently H, OH or C.sub.1-C.sub.3 alkoxy; [0125] R.sup.4 and R.sup.5 are each independently H or C.sub.1-C.sub.3 alkyl; [0126] Y is C.dbd.O or C--O--R.sup.7, where R.sup.7 is H or C.sub.1-C.sub.3 alkyl; and [0127] A is C--CH.sub.2--R.sup.6 or C--C(O)R.sup.6 where R.sup.6 is straight or branched chain, saturated or partially unsaturated C.sub.1-C.sub.6 alkyl, B is C.dbd.O, or A and B together are part of an aromatic six-carbon membered ring structure sharing a side with the remainder of the structure of Formula (C'').
[0128] In embodiments of a compound according to Formula (C''), R.sup.1, R.sup.2 and R.sup.3 are H.
[0129] In embodiments, A is C--CH.sub.2--R.sup.6 or C--C(O)R.sup.6 where R.sup.6 is straight or branched chain, saturated or partially unsaturated C.sub.1-C.sub.6 alkyl and B is C.dbd.O.
[0130] In embodiments, R.sup.1, R.sup.2 and R.sup.3 are each independently OH or C.sub.1-C.sub.3 alkoxy. In some embodiments, R.sup.1, R.sup.2 and R.sup.3 are the same.
[0131] In embodiments, A and B together are part of an aromatic six-carbon membered unsubstituted ring structure.
[0132] In embodiments, a compound according to Formula (C) has the following structure:
##STR00033## [0133] where: [0134] R.sup.1, R.sup.2 and R.sup.3 are each independently H, OH or C.sub.1-C.sub.3 alkoxy; [0135] R.sup.4 and R.sup.5 are each independently H or C.sub.1-C.sub.3 alkyl; [0136] Y is C.dbd.O or C--O--R.sup.7, where R.sup.7 is H or C.sub.1-C.sub.3 alkyl; and [0137] A is C--CH.sub.2--R.sup.6 or C--C(O)R.sup.6 where R.sup.6 is straight or branched chain, saturated or partially unsaturated C.sub.1-C.sub.6 alkyl, B is C.dbd.O, or A and B together are part of an aromatic six-carbon membered ring structure sharing a side with the remainder of the structure of Formula (C'').
[0138] In embodiments of a compound according to Formula (C'''), R.sup.1, R.sup.2 and R.sup.3 are H. In embodiments, A is C--CH.sub.2--R.sup.6 or C--C(O)R.sup.6 where R.sup.6 is straight or branched chain, saturated or partially unsaturated C.sub.1-C.sub.6 alkyl and B is C.dbd.O. In embodiments, R.sup.1, R.sup.2 and R.sup.3 are each independently OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.1, R.sup.2 and R.sup.3 are the same. In embodiments, A and B together are part of an aromatic six-carbon membered unsubstituted ring structure.
[0139] In embodiments, a compound according to Formula (C) has the following structure:
##STR00034## [0140] where: [0141] X is N; [0142] R.sup.1 is H or C.sub.1-C.sub.3 alkyl; [0143] R.sup.2 and R.sup.3 are each independently selected from H and straight or branched chain, saturated or unsaturated C.sub.1-C.sub.3 alkyl or alkenyl; [0144] R.sup.4 is H or C.sub.1-C.sub.3 alkyl; [0145] R.sup.5 is H or C.sub.1-C.sub.3 alkyl; [0146] A and B are each independently selected from C.dbd.O and C--C(O)R.sup.6 where R.sup.6 is straight or branched chain, saturated or partially unsaturated C.sub.1-C.sub.6 alkyl; and [0147] Y is C--O--R.sup.7, where R.sup.7 is H or C.sub.1-C.sub.3 alkyl.
[0148] In embodiments of a compound according to Formula (C''''), one or more of R.sup.1, R.sup.2 and R.sup.3 are H. In embodiments, each of R.sup.1, R.sup.2 and R.sup.3 are H. In embodiments, R.sup.4 and R.sup.5 are independently C.sub.1-C.sub.3 alkyl. In embodiments, R.sup.4 and R.sup.5 are methyl. In embodiments, wherein A is C--C(O)R.sup.6. In embodiments, B is C.dbd.O.
[0149] Group D Compounds
[0150] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00035## [0151] where: [0152] R.sup.1 is H, methyl, OCOCH.sub.3 or forms together with R.sup.6 a five membered ring structure in which R.sup.1 and R.sup.6 together are C.dbd.O or CH.sub.2; [0153] R.sup.6 is H, C.dbd.OCH.sub.3, or together forms a five membered ring structure in which R.sup.1 and R.sup.6 together are C.dbd.O or CH.sub.2; [0154] R.sup.7 is OCOCH.sub.3; [0155] A and B are C, CH or CCH.sub.3, wherein when A and B are both C a double bond is formed between A and B; and [0156] R.sup.4 and R.sup.5 are independently selected from OH, methyl,
[0156] ##STR00036## [0157] or [0158] R.sup.4 and R.sup.5 together with the carbon to which they are bound form
[0158] ##STR00037## [0159] to form a compound of the following formula
[0159] ##STR00038## [0160] where R.sup.1, R.sup.6 and R.sup.7 are as described above.
[0161] Group E Compounds
[0162] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00039##
[0163] where R.sup.1, R.sup.2 and R.sup.3 are independently selected from the group consisting of H and COCH.sub.3.
[0164] Group F Compounds
[0165] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00040## [0166] where: [0167] R.sup.42 is H or OH; [0168] R.sup.43 are R.sup.44 each independently H or CH.sub.3; [0169] R.sup.1 and R.sup.2 are each independently OH, C.sub.1-C.sub.3 alkyl or C.sub.1-C.sub.3 hydroxyl; [0170] R.sup.3 and R.sup.4 are each independently H, OH or C.sub.1-C.sub.3 hydroxyl; [0171] R.sup.5 is H or --OC(O)R.sup.8, where R.sup.8 is C.sub.1-C.sub.8 straight or branched chain, saturated or unsaturated alkyl; [0172] W is CH; [0173] Y is CH, X is CH or CH.sub.2, and Z is C or CR.sup.9, provided that Z is C when X is CH.sub.2, where R.sup.9 is H or together with R.sup.6 and the carbons to which they are bound and the intervening carbon of W form a five membered ring with an oxygen heteroatom; [0174] R.sup.6 is C.sub.1-C.sub.3 hydroxyl, C(O)R.sup.10, (CH.sub.2)p'R.sup.10 where p' is zero or 1, or together with R.sup.9 and the carbons to which they are bound and the intervening carbon of W form a five membered ring with an oxygen heteroatom, [0175] where R.sup.10 is H, OH, or saccharidyl; and [0176] R.sup.7 is H, OH, C.sub.1-C.sub.3 hydroxyl, or (CH.sub.2).sub.pR.sup.44, where p is zero or 1 and R.sup.44 is saccharidyl.
[0177] In embodiments, R.sup.10 is saccharidyl and is
##STR00041## [0178] where a and b are each independently zero or 1; [0179] where R.sup.11 is H or
[0179] ##STR00042## [0180] where d is zero or 1, [0181] R.sup.14 is H, OH, CH.sub.3 or C.sub.1-C.sub.3 hydroxyl, and [0182] R.sup.15 is H, C(O)R.sup.16 where R.sup.16 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, [0183] where R.sup.12 and R.sup.13 are each independently H, C(O)R.sup.41 where R.sup.41 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0183] ##STR00043## [0184] where e is zero or 1, [0185] R.sup.17 is H, OH or CH.sub.3, and [0186] R.sup.18 is H, C(O)R.sup.42 where R.sup.42 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0186] ##STR00044## [0187] where f is zero or 1, [0188] R.sup.19 is H, OH or CH.sub.3, and [0189] R.sup.20 and R.sup.21 are each independently H, C(O)R.sup.43 where R.sup.43 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0189] ##STR00045## [0190] where [0191] g is zero or 1, and [0192] R.sup.22, R.sup.23 and R.sup.24 are each independently H, OH or CH.sub.3.
[0193] In embodiments, R.sup.44 is saccharidyl and is
##STR00046## [0194] where [0195] i is zero or 1, [0196] R.sub.25, R.sub.26, R.sup.27 and R.sup.28 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2)--OC(O)R.sup.29 where n is zero, 1, 2 or 3 and R.sup.29 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.mOR.sup.30 where m is zero or 1 and R.sup.30 is
[0196] ##STR00047## [0197] where [0198] j is zero or 1, [0199] R.sup.31, R.sup.32, R.sup.33 and R.sup.34 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.qOC(O)R.sup.35 where q is zero, 1, 2 or 3 and R.sup.35 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.tOR.sup.36 where t is zero or 1 and R.sup.36 is
[0199] ##STR00048## [0200] where [0201] k is zero or 1, and [0202] R.sup.37, R.sup.38, R.sup.39 and R.sup.40 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.uOC(O)R.sup.41 where u is zero, 1, 2 or 3 and R.sup.41 is H or C.sub.1-C.sub.3 alkyl.
[0203] In embodiments, Z is C. In embodiments, Z is CR.sup.9 and wherein R.sup.9 together with R.sup.6 and the carbons to which they are bound and the intervening carbon of W for a five membered ring with an oxygen heteroatom. In embodiments, R.sup.6 is C(O)R.sup.10. In embodiments, R.sup.10 is H or OH. In embodiments, R.sup.7 is OH. In embodiments, R.sup.1 and R.sup.2 are each independently C.sub.1-C.sub.3 alkly or C.sub.1-C.sub.3 hydroxyl. In embodiments, R.sup.3 is H or OH. In embodiments, R.sup.4 is H. In embodiments, R.sup.5 is H. In embodiments, one or more of R.sup.16, if present, R.sup.42 and R.sup.43 are
##STR00049##
[0204] In embodiments, a compound according to Formula (F) has the following structure:
##STR00050##
[0205] where: [0206] R.sup.1 and R.sup.2 are each independently OH, C.sub.1-C.sub.3 alkyl or C.sub.1-C.sub.3 hydroxyl; [0207] R.sup.3 and R.sup.4 are each independently H, OH or C.sub.1-C.sub.3 hydroxyl; [0208] R.sup.5 is H; [0209] W is CH; [0210] Y is CH, X is CH or CH.sub.2, and Z is C or CR.sup.9, provided that Z is C when X is CH.sub.2, where R.sup.9 is H or together with R.sup.6 and the carbons to which they are bound and the intervening carbon of W form a five membered ring with an oxygen heteroatom; [0211] R.sup.6 is C.sub.1-C.sub.3 hydroxyl, C(O)R.sup.10, (CH.sub.2)p'R.sup.10 where p' is zero or 1, or together with R.sup.9 and the carbons to which they are bound and the intervening carbon of W form a five membered ring with an oxygen heteroatom, [0212] where R.sup.10 is H or saccharidyl; and [0213] R.sup.7 is H, OH, C.sub.1-C.sub.3 hydroxyl, or (CH.sub.2).sub.pOR.sup.44, where p is zero or 1 and R.sup.44 is saccharidyl.
[0214] In embodiments of a compound according to Formula F', R.sup.10 is saccharidyl and is
##STR00051## [0215] where a and b are each independently zero or 1; [0216] R.sup.11 is H or
[0216] ##STR00052## [0217] where d is zero or 1, [0218] R.sup.14 is H, OH, CH.sub.3 or C.sub.1-C.sub.3 hydroxyl, and [0219] R.sup.15 is H, C(O)R.sup.16 where R.sup.16 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, [0220] where R.sup.12 and R.sup.13 are each independently H, C(O)R.sup.41 where R.sup.41 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0220] ##STR00053## [0221] where e is zero or 1, [0222] R.sup.17 is H, OH or CH.sub.3 and [0223] R.sup.18 is H, C(O)R.sup.42 where R.sup.42 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0223] ##STR00054## [0224] where f is zero or 1, [0225] R.sup.19 is H, OH or CH.sub.3, and [0226] R.sup.20 and R.sup.21 are each independently H, C(O)R.sup.43 where R.sup.43 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0226] ##STR00055## [0227] where [0228] g is zero or 1, and [0229] R.sup.22, R.sup.23 and R.sup.24 are each independently H, OH or CH.sub.3.
[0230] In embodiments of a compound according to Formula F', R.sup.44 is saccharidyl and is
##STR00056## [0231] where [0232] i is zero or 1, and [0233] R.sup.25, R.sup.26, R.sup.27 and R.sup.28 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2)--OC(O)R.sup.29 where n is zero, 1, 2 or 3 and R.sup.29 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.mOR.sup.30 where m is zero or 1 and R.sup.30 is
[0233] ##STR00057## [0234] where [0235] j is zero or 1, and [0236] R.sup.31, R.sup.32, R.sup.33 and R.sup.34 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.qOC(O)R.sup.35 where q is zero, 1, 2 or 3 and R.sup.35 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.tOR.sup.36 where t is zero or 1 and R.sup.36 is
[0236] ##STR00058## [0237] where [0238] k is zero or 1, and [0239] R.sup.37, R.sup.38, R.sup.39 and R.sup.40 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.uOC(O)R.sup.41 where u is zero, 1, 2 or 3 and R.sup.41 is H or C.sub.1-C.sub.3 alkyl.
[0240] In embodiments of a compound according to Formula F', Z is C. In embodiments, Z is CR.sup.9 and wherein R.sup.9 together with R.sup.6 and the carbons to which they are bound and the intervening carbon of W for a five membered ring with an oxygen heteroatom. In embodiments, R.sup.6 is C(O)R.sup.10. In embodiments, R.sup.1 and R.sup.2 are each independently C.sub.1-C.sub.3 alkly or C.sub.1-C.sub.3 hydroxyl. In embodiments, R.sup.3 is H or OH. In embodiments, R.sup.4 is H. In embodiments, R.sup.16, if present, is
##STR00059##
[0241] In embodiments, a compound according to Formula (F) has the following structure:
##STR00060## [0242] where: [0243] R.sup.1 and R.sup.2 are each independently OH, C.sub.1-C.sub.3 alkyl or C.sub.1-C.sub.3 hydroxyl; [0244] R.sup.3 is H; [0245] R.sup.4 is H, OH or C.sub.1-C.sub.3 hydroxyl; [0246] R.sup.5 is H; [0247] W is CH; [0248] Y is CH, X is CH or CH.sub.2, and Z is C or CR.sup.9, provided that Z is C when X is CH.sub.2, where R.sup.9 is H or together with R.sup.6 and the carbons to which they are bound and the intervening carbon of W form a five membered ring with an oxygen heteroatom; [0249] R.sup.6 is, together with R.sup.9 and the carbons to which they are bound and the intervening carbon of W form a five membered ring with an oxygen heteroatom, or C(O)R.sup.10 [0250] where R.sup.10 is H or
[0250] ##STR00061## [0251] where a and b are each independently zero or 1; [0252] where R.sup.11 is H, OH, or
[0252] ##STR00062## [0253] where d is zero or 1, [0254] R.sup.14 is H, OH, CH.sub.3 or C.sub.1-C.sub.3 hydroxyl, and [0255] R.sup.15 is H, C(O)R.sup.16 where R.sup.16 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, [0256] where R.sup.12 and R.sup.13 are each independently H, C(O)R.sup.41 where R.sup.41 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0256] ##STR00063## [0257] where e is zero or 1, [0258] R.sup.17 is H, OH or CH.sub.3 and [0259] R.sup.18 is H, C(O)R.sup.42 where R.sup.42 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0259] ##STR00064## [0260] where f is zero or 1, [0261] R.sup.19 is H, OH or CH.sub.3, and [0262] R.sup.20 and R.sup.21 are each independently H, C(O)R.sup.43 where R.sup.43 is C.sub.1-C.sub.15 straight or branched chain, saturated or unsaturated alkyl unsubstituted or substituted with hydroxyl, or
[0262] ##STR00065## where g is zero or 1, and R.sub.22, R.sub.23 and R.sup.24 are each independently H, OH or CH.sub.3; and [0263] R.sup.7 is H, OH, C.sub.1-C.sub.3 hydroxyl, or (CH.sub.2).sub.pR.sup.44, where p is zero or 1 and R.sup.44 is
[0263] ##STR00066## [0264] where [0265] i is zero or 1, and [0266] R.sup.25, R.sup.26, R.sup.27 and R.sup.28 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2)--OC(O)R.sup.29 where n is zero, 1, 2 or 3 and R.sup.29 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.mOR.sup.30 where m is zero or 1 and R.sup.30 is
[0266] ##STR00067## [0267] where [0268] j is zero or 1, and [0269] R.sup.31, R.sup.32, R.sup.33 and R.sup.34 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.qOC(O)R.sup.35 where q is zero, 1, 2 or 3 and R.sup.35 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.tOR.sup.36 where t is zero or 1 and R.sup.36 is
[0269] ##STR00068## [0270] where [0271] k is zero or 1, and [0272] R.sup.37, R.sup.38, R.sup.39 and R.sup.40 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2)--OC(O)R.sup.41 where u is zero, 1, 2 or 3 and R.sup.41 is H or C.sub.1-C.sub.3 alkyl.
[0273] In embodiments of a compound according to Formula (F''), R.sup.4 is H. In embodiments, R.sup.1 is CH.sub.3. In embodiments, R.sup.2 is CH.sub.3.
[0274] Group G Compound
[0275] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00069##
[0276] Group H Compounds
[0277] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00070##
[0278] where: [0279] R.sup.1 is H or saccharidyl; [0280] R.sup.2 is H or OH; [0281] R.sup.3 and R.sup.4 are independently selected from H or methyl or together form CH.sub.2; [0282] R.sup.5 is CH.sub.2 or CH; [0283] R.sup.6 is CH or C, provided that when R.sup.5 is CH, R.sup.6 is C; [0284] R.sup.7 and R.sup.8 together with the carbons to which they are bound form
[0284] ##STR00071## [0285] to form a compound having the following structure
[0285] ##STR00072## [0286] and [0287] R.sup.9, R.sup.10, and R.sup.11 are independently H or methyl.
[0288] In embodiments, R.sup.1 is saccharidyl and is
##STR00073##
[0289] Group I Compounds
[0290] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00074## [0291] where: [0292] X is O or CH.sub.2; [0293] R.sup.1 and R.sup.9 are independently selected from H, OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 alkyl, and (CH.sub.2)--OH, where n is an integer from 1 to 3; [0294] R.sup.2 and R.sup.10 are independently selected from H, OH and C.sub.1-C.sub.3 alkyl; [0295] R.sup.3 and R.sup.4 are independently selected from H and C.sub.1-C.sub.3 alkyl or together form CH.sub.2; [0296] R.sup.5, R.sup.6 and R.sup.7 are independently selected from H and C.sub.1-C.sub.3 alkyoxy; and [0297] R.sup.8 is H, OH or C.sub.1-C.sub.3 alkyoxy.
[0298] In embodiments, R.sup.5, R.sup.6, R.sup.7 and R.sup.8 are independently C.sub.1-C.sub.3 alkyoxy. In embodiments, R.sup.5, R.sup.6, R.sup.7 and R.sup.8 are methoxy. In embodiments, R.sup.3 and R.sup.4 together form CH.sub.2. In embodiments, R.sup.3 and R.sup.4 are independently C.sub.1-C.sub.3 alkyl. In embodiments, R.sup.3 and R.sup.4 are methyl. In embodiments, at least two of R.sup.1, R.sup.2, R.sup.9 and R.sup.10 are independently C.sub.1-C.sub.3 alkyl. In embodiments, at least two of R.sup.1, R.sup.2, R.sup.9 and R.sup.10 are methyl. In embodiments, X is CH.sub.2.
[0299] In embodiments, X is O. In some embodiments where X is O, R.sup.2 and R.sup.10 are H. In some embodiments where X is O, two or more or R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7, and R.sup.10 are H. In some embodiments where X is O, each of R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7, and R.sup.10 are H. In some embodiments where X is O, R.sup.8 is OH or C.sub.1-C.sub.3 alkoxy. In some embodiments where X is O, R.sup.1 and R.sup.9 are independently selected from OH, C.sub.1-C.sub.3 alkoxy, and (CH.sub.2).sub.n--OH. In some embodiments where X is O, one of R.sup.1 and R.sup.9 is selected from OH and C.sub.1-C.sub.3 alkoxy, and the other of R.sup.1 and R.sup.9 is (CH.sub.2).sub.n--OH.
[0300] In embodiments, a compound according to Formula (I) has the following structure:
##STR00075## [0301] where: [0302] X is O; [0303] R.sup.1 and R.sup.9 are independently selected from H, OH, C.sub.1-C.sub.3 alkoxy, C.sub.1-C.sub.3 alkyl, and [0304] (CH.sub.2)--OH, where n is an integer from 1 to 3; [0305] R.sup.2 and R.sup.10 are independently selected from H, OH and C.sub.1-C.sub.3 alkyl; [0306] R.sup.3 and R.sup.4 are independently selected from H and C.sub.1-C.sub.3 alkyl; [0307] R.sup.5, R.sup.6 and R.sup.7 are H; and [0308] R.sup.8 is H, OH or C.sub.1-C.sub.3 alkyoxy.
[0309] In embodiments of a compound according to Formula (I'), R.sup.2 and R.sup.10 are H. In embodiments, each of R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7, and R.sup.10 are H. In embodiments, R.sup.8 is OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.1 and R.sup.9 are independently selected from OH, C.sub.1-C.sub.3 alkoxy, and (CH.sub.2)--OH. In embodiments, one of R.sup.1 and R.sup.9 is selected from OH and C.sub.1-C.sub.3 alkoxy, and the other of R.sup.1 and R.sup.9 is (CH.sub.2).sub.n--OH.
[0310] Group J Compounds
[0311] Group J1 Compounds
[0312] In embodiments, a bioactive, taste modulating, or salty taste modulating is a compound having the following structure:
##STR00076##
[0313] where: [0314] R.sup.1 and R.sup.6 are each independently OH, C.sub.1-C.sub.3 hydroxyl, C.sub.1-C.sub.3 alkoxy or saccharidyl; [0315] R.sup.2, R.sup.3, R.sup.7 and R.sup.8 are each independently H, OH, C.sub.1-C.sub.3 hydroxyl, C.sub.1-C.sub.3 alkoxy; [0316] R.sup.4 and R.sup.5 are each independently H, OH, C.sub.1-C.sub.3 alkyl, C.sub.1-C.sub.3 alkoxy, or C.sub.1-C.sub.3 hydroxyl; [0317] Y is CHCH and n is zero or 1; and [0318] X is CHR.sup.9 and m is zero or 1, where R.sup.9 is
[0318] ##STR00077## [0319] where R.sup.10, R.sup.11 and R.sup.12 are each independently OH, C.sub.1-C.sub.3 alkoxy, or C.sub.1-C.sub.3 hydroxyl, [0320] provided that if n is 1, then m is 1.
[0321] In embodiments of a compound according to Formula J1, R.sup.1, R.sup.6, or R.sup.1 and R.sup.6 are saccharidyl and are
##STR00078##
[0322] In embodiments of a compound according to Formula J1, R.sup.1 is OH. In embodiments, R.sup.2, R.sup.3, R.sup.7 and R.sup.8 are each independently H or C.sub.1-C.sub.3 alkoxy. In embodiments, where one of wherein R.sup.2 and R.sup.3 is H and the other is C.sub.1-C.sub.3 alkoxy, one of R.sup.7 and R.sup.8 is H and the other is C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.2, R.sup.3, R.sup.7 and R.sup.8 are each H. In embodiments, R.sup.10 is present and is OH. In embodiments, R.sup.11 is present and is C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.12 is present and is OH. In embodiments, R.sup.9 is present and is
##STR00079##
where R.sup.10, R.sup.11, and R.sup.12 are as defined above.
[0323] In embodiments of a compound according to Formula J1, the compound has the following structure:
##STR00080## [0324] where R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7, R.sup.8, and R.sup.9 are as defined above.
[0325] In embodiments of a compound according to Formula J1, the compound has the following structure:
##STR00081## [0326] where R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7, and R.sup.8 are as defined above.
[0327] In embodiments of a compound according to Formula J1, the compound has the following structure:
##STR00082## [0328] where:
[0329] R.sup.1 and R.sup.6 are each independently OH, C.sub.1-C.sub.3 hydroxyl, C.sub.1-C.sub.3 alkoxy or saccharidyl; [0330] R.sup.2, R.sup.3, R.sup.7 and R.sup.8 are each independently H, OH, C.sub.1-C.sub.3 hydroxyl, C.sub.1-C.sub.3 alkoxy; [0331] R.sup.4 and R.sup.5 are each independently H, OH, C.sub.1-C.sub.3 alkoxy, or C.sub.1-C.sub.3 hydroxyl; [0332] Y is CHCH and n is zero or 1; and [0333] X is CHR.sup.9 and m is zero or 1, where R.sup.9 is
[0333] ##STR00083## [0334] where R.sup.10, R.sup.11 and R.sup.12 are each independently OH, C.sub.1-C.sub.3 alkoxy, or C.sub.1-C.sub.3 hydroxyl, [0335] provided that if n is 1, then m is 1.
[0336] In embodiments, R.sup.1, R.sup.6 or R.sup.1 and R.sup.6 are saccharidyl and are
##STR00084##
In embodiments, R.sup.1 is OH. In embodiments, R.sup.2, R.sup.3, R.sup.7 and R.sup.8 are each independently H or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.2 and R.sup.3 is H and the other is C.sub.1-C.sub.3 alkoxy, and one of R.sup.7 and R.sup.8 is H and the other is C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.2, R.sup.3, R.sup.7 and R.sup.8 are each H. In embodiments, R.sup.10 is present and is OH. In embodiments, R.sup.11 is present and is C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.12 is present and is OH. In embodiments, R.sup.9 is present and is
##STR00085## [0337] where R.sup.10, R.sup.11, and R.sup.12 are as defined above.
[0338] Group J2 Compounds
[0339] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00086##
[0340] where [0341] R.sup.1 is H, OH, or saccharidyl; [0342] R.sup.2 and R.sup.3 are each independently H or C.sub.1-C.sub.3 alkyl; [0343] R.sup.4 and R.sup.5 are each independently H,
[0343] ##STR00087## [0344] or CHC(R.sup.10)R.sup.11, where [0345] R.sup.10 and R.sup.11 are each independently H or C.sub.1-5 alkyl unsubstituted or substituted with one or more of OH and
[0345] ##STR00088## [0346] X is C.dbd.O or O; [0347] Y is C.dbd.O or O, provided that when X is O, Y is C.dbd.O, or when X is C.dbd.O, Y is O; [0348] A is CHR.sup.12 or CR.sup.12, where [0349] R.sup.12 is H, OH, or saccharidyl; [0350] B is C or CH, provided that if B is C then A is CR.sup.12.
[0351] In embodiments of a compound according to Formula J2, R.sup.1 is saccharidyl and is
##STR00089## [0352] where [0353] a is zero or 1, and
[0354] R.sup.6, R.sup.7, R.sup.8, and R.sup.9 are each independently H, OH, CH.sub.3, or CH.sub.2OH.
[0355] In embodiments of a compound according to Formula J2, R.sup.12 is saccharidyl and is
##STR00090## [0356] where [0357] b is zero or 1, and [0358] R.sup.13, R.sup.14, R.sup.15, and R.sup.16 are each independently H, OH, CH.sub.3, or CH.sub.2OH.
[0359] In embodiments of a compound according to Formula J2, R.sup.4 is CHC(CH.sub.3).sub.2 or
##STR00091##
[0360] In embodiments of a compound according to Formula J2, R.sup.5 is H or
##STR00092##
[0361] Group K Compound
[0362] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00093##
[0363] Group L Compounds
[0364] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00094##
[0365] where: [0366] R.sup.1, R.sup.3, R.sup.4, R.sup.5 and R.sup.6 are each independently H, OH, C.sub.1-C.sub.3 hydroxyl or C.sub.1-C.sub.3 alkoxy; and [0367] R.sup.2 is H, C.sub.1-C.sub.3 alkyl, or
[0367] ##STR00095## [0368] where R.sup.7, R.sup.8 and R.sup.9 are each independently OH, C.sub.1-C.sub.3 hydroxyl or C.sub.1-C.sub.3 alkoxy.
[0369] In embodiments, R.sup.2 is C.sub.1-C.sub.3 alkyl, such as methyl. In embodiments, R.sup.5 is OH or C.sub.1-C.sub.3 hydroxyl. In embodiments, R.sup.3 is H. In embodiments, R.sup.4 is C.sub.1-C.sub.3 alkoxy, such as methoxy. In embodiments, R.sup.6 is C.sub.1-C.sub.3 alkyl, such as methyl or propyl. In embodiments, R.sup.1 is H. In embodiments, R.sup.1 is C.sub.1-C.sub.3 hydroxyl, such as C.sub.2 hydroxyl. In embodiments, R.sup.2 is
##STR00096##
In embodiments, R.sup.7, R.sup.8 and R.sup.9 are each independently OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.7 is OH and R.sup.8 and R.sup.9 are each independently C.sub.1-C.sub.3 alkoxy, such as methoxy.
[0370] In embodiments, R.sup.3 is OH, C.sub.1-C.sub.3 hydroxyl, or C.sub.1-C.sub.3 alkoxy.
[0371] In embodiments, R.sup.5 is H.
[0372] In embodiments, R.sup.6 is C.sub.1-C.sub.3 alkoxy, such as methoxy.
[0373] In embodiments, R.sup.1 is C.sub.1-C.sub.3 alkoxy, such as methoxy.
[0374] In embodiments, R.sup.4 is H.
[0375] In embodiments of a compound according to Formula L, (i) R.sup.1 is OH, C.sub.1-C.sub.3 hydroxyl or C.sub.1-C.sub.3 alkoxy; (ii) R.sup.2 is H, C.sub.1-C.sub.3 alkyl, or
##STR00097##
where R.sup.7, R.sup.8 and R.sup.9 are each independently OH, C.sub.1-C.sub.3 hydroxyl or C.sub.1-C.sub.3 alkoxy; and (iii) R.sup.3, R.sup.4, R.sup.5 and R.sup.6 are each independently H, OH, C.sub.1-C.sub.3 hydroxyl or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.2 is C.sub.1-C.sub.3 alkyl, such as methyl. In embodiments, R.sup.5 is OH or C.sub.1-C.sub.3 hydroxyl. In embodiments, R.sup.3 is H. In embodiments, R.sup.4 is C.sub.1-C.sub.3 alkoxy, such as methoxy. In embodiments, R.sup.6 is C.sub.1-C.sub.3 alkyl, such as butyl. In embodiments, R.sup.1 is C.sub.1-C.sub.3 hydroxyl. In some embodiments where R.sup.2 is
##STR00098##
R.sup.7, R.sup.8 and R.sup.9 may each independently be OH or C.sub.1-C.sub.3 alkoxy. For example, R.sup.7 may be OH and R.sup.8 and R.sup.9 may each independently be C.sub.1-C.sub.3 alkoxy, such as methoxy. In some embodiments where R.sup.2 is
##STR00099##
R.sup.3 is OH, C.sub.1-C.sub.3 hydroxyl, or C.sub.1-C.sub.3 alkoxy. In some embodiments where R.sup.2 is
##STR00100##
R.sup.5 is H. In some embodiments where R.sup.2 is
##STR00101##
R.sup.6 is C.sub.1-C.sub.3 alkoxy, such as methoxy. In some embodiments where R.sup.2 is
##STR00102##
R.sup.1 is C.sub.1-C.sub.3 alkoxy, such as methoxy. In some embodiments where R.sup.2 is
##STR00103##
R.sup.4 is H.
[0376] In embodiments, a compound according to Formula L is a compound having the following structure:
##STR00104##
[0377] where: [0378] R.sup.1, R.sup.3, R.sup.4, R.sup.5 and R.sup.6 are each independently H, OH, C.sub.1-C.sub.3 hydroxyl or C.sub.1-C.sub.3 alkoxy; and [0379] R.sup.7, R.sup.8 and R.sup.9 are each independently OH, C.sub.1-C.sub.3 hydroxyl or C.sub.1-C.sub.3 alkoxy.
[0380] In embodiments of a compounds according to Formula L', R.sup.7, R.sup.8 and R.sup.9 are each independently OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.7 is OH and R.sup.8 and R.sup.9 are each independently C.sub.1-C.sub.3 alkoxy, such as methoxy. In embodiments, R.sup.3 is OH, C.sub.1-C.sub.3 hydroxyl, or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.5 is H. In embodiments, R.sup.6 is C.sub.1-C.sub.3 alkoxy, such as methoxy. In embodiments, R.sup.1 is C.sub.1-C.sub.3 alkoxy, such as methoxy. In embodiments, R.sup.4 is H.
[0381] Group M Compound
[0382] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00105##
[0383] Group N Compound
[0384] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00106##
[0385] Group O Compound
[0386] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00107##
[0387] Group P Compounds
[0388] Group P1 Compounds
[0389] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00108##
[0390] where: [0391] X is CH or CH.sub.2; [0392] Y is CH or C, with the caveat that if Y is C, then X is CH; [0393] R.sup.1 is H, or
[0393] ##STR00109## [0394] where R.sup.5 is OH, C(O)OH, or OC(O)R.sup.6, where R.sup.6 is a C.sub.1-C.sub.3 alkyl; [0395] R.sup.2 and R.sup.3 are independently H, or substituted with hydroxyl or unsubstituted saturated or unsaturated branched or straight C.sub.1-C.sub.6 alkoxy; and [0396] R.sup.4 is H, or a saturated or unsaturated branched or straight C.sub.1-C.sub.6 alkyl.
[0397] In embodiments, at least one of R.sup.2 or R.sup.3 is H. In embodiments, both R.sup.2 and R.sup.3 are H. In embodiments, at least one of R.sup.2 and R.sup.3 is a substituted saturated or unsaturated branched or straight C.sub.1-C.sub.6 alkoxy substituted with at least one hydroxyl group. In embodiments, both R.sup.2 and R.sup.3 are independently selected from a substituted saturated or unsaturated branched or straight C.sub.1-C.sub.6 alkoxy substituted with at least one hydroxyl group. For example, at least one of R.sup.2 and R.sup.3 can be methoxy. In embodiments, at least one of R.sup.2 and R.sup.3 is unsaturated. In embodiments, at least one of R.sup.2 and R.sup.3 is branched. In embodiments, R.sup.2 is a straight unsubstituted saturated C.sub.1-C.sub.3 alkoxy; and R.sup.3 is a branched unsubstituted, unsaturated C.sub.3-C.sub.6 alkoxy.
[0398] In embodiments, R.sup.4 is saturated or unsaturated branched or straight C.sub.1-C.sub.6 alkyl. For example, R.sup.4 can be C.sub.5 alkyl. In embodiments, R.sup.4 is unsaturated. In embodiments, R.sup.4 is CH(CH.sub.3).sub.2CHCH.sub.2.
[0399] Group P2 Compounds
[0400] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00110##
[0401] where [0402] X is selected from NH, O, C(O), CR.sup.5 or CR.sup.5R.sup.6, where R.sup.5 and R.sup.6 are independently selected from H, OH, a straight or branched C.sub.1 to C.sub.6 hydroxyl, or a C.sub.1 to C.sub.6 alkyl; and [0403] Y, and Z are independently selected from O, CR.sup.5, CR.sup.5R.sup.6, where R.sup.5 and R.sup.6 are independently selected from H, OH, a straight or branched C.sub.1 to C.sub.6 hydroxyl, or a C.sub.1 to C.sub.6 alkyl, or
[0403] ##STR00111## [0404] with the proviso that (i) no more than two of X, Y and Z and are O, and (ii) if Y is [0405] CR.sup.5 then X or Z is also CR.sup.5; [0406] R.sup.1 is H, C.sub.1-C.sub.3 alkoxy, C(O)R.sup.7, where R.sup.7 is a C.sub.1 to C.sub.6 straight or branched alkyl, or
[0406] ##STR00112## [0407] where R.sup.8, R.sup.9 and R.sup.10 are independently selected from: H, OH, C.sub.1 to C.sub.3 hydroxyl, C.sub.1 to C.sub.3 alkoxy, or C.sub.3 to C.sub.8 alkyl; [0408] R.sup.2 is H, OH, or
[0408] ##STR00113## [0409] where R.sup.11 is H, OH, or C.sub.1 to C.sub.3 hydroxyl; [0410] R.sup.3 is H, C.sub.1-C.sub.3 alkoxy, or together with R.sup.4 form a six membered ring having an oxygen heteroatom to form a compound having a structure of:
[0410] ##STR00114## [0411] R.sup.4 is H, C.sub.1 to C.sub.3 alkoxy, or together with R.sup.3 forms the structure above.
[0412] In embodiments, X is CR.sup.5R.sup.6. In embodiments, X is C(OH)(CH.sub.2CH.sub.2CH.sub.2CH.sub.3). In embodiments, X is O. In embodiments, X is CH.sub.2.
[0413] In embodiments, Y is O. In embodiments, Y is CR.sup.5R.sup.6, where R.sup.5 and R.sup.6 are H and a branched C.sub.1 to C.sub.6 hydroxyl, such as C(CH.sub.3)(CH.sub.3)OH. In embodiments, Y is CH.
[0414] In embodiments, Z is C(O). In embodiments, Z is CH.sub.2. In embodiments, Z is O.
[0415] In embodiments, R.sup.1 is
##STR00115##
In embodiments,
[0416] R1 is a C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.1 is C(O)R.sup.7, where R.sup.7 is a C.sub.1 to C.sub.6 straight or branched alkyl.
[0417] In embodiments, R.sup.7 is a branched C.sub.3 alkyl.
[0418] In embodiments, R.sup.2 is H. In embodiments, R.sup.2 is OH. In embodiments, R.sup.2 is
##STR00116##
[0419] In embodiments, R.sup.3 together with R.sup.4 form a six membered ring having an oxygen heteroatom to form a compound having the structure:
##STR00117##
[0420] In embodiments, R.sup.3 is H. In embodiments, R.sup.3 is a C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.4 is H.
[0421] In embodiments, X is NH.
[0422] In embodiments, Z is
##STR00118##
[0423] Group P3 Compounds
[0424] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00119## [0425] where [0426] Y is independently selected from CR.sup.5, CR.sup.5R.sup.6, where R.sup.5 and R.sup.6 are independently selected from H, OH, a straight or branched C.sub.1 to C.sub.6 hydroxyl, or a C.sub.1 to C.sub.6 alkyl; [0427] R.sup.1 is H, C.sub.1-C.sub.3 alkoxy, C(O)R.sup.7, where R.sup.7 is a C.sub.1 to C.sub.6 straight or branched alkyl, or
[0427] ##STR00120## [0428] where R.sup.8, R.sup.9 and R.sup.10 are independently selected from: H, OH, C.sub.1 to C.sub.3 hydroxyl, C.sub.1 to C.sub.3 alkoxy, or C.sub.3 to C.sub.8 alkyl; [0429] R.sup.2 is H or
[0429] ##STR00121## [0430] where R.sup.11 is H, OH, or C.sub.1 to C.sub.3 hydroxyl; [0431] R.sup.3 is H, C.sub.1-C.sub.3 alkoxy, or together with R.sup.4 form a six membered ring having an oxygen heteroatom to form a compound having a structure of:
[0431] ##STR00122## [0432] R.sup.4 is H, C.sub.1 to C.sub.3 alkoxy, or together with R.sup.3 forms the structure above.
[0433] In embodiments, R.sup.1 is C.sub.1 to C.sub.3 alkoxy. In embodiments, R.sup.1 is H. In embodiments, R.sup.2 is H. In embodiments, R.sup.3 is H. In embodiments, R.sup.4 is H.
[0434] Group P4 Compounds
[0435] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00123##
[0436] where [0437] R.sup.1 is H or a saturated or unsaturated C.sub.1 to C.sub.6 alkyl; [0438] R.sup.2 is H or a C.sub.1 to C.sub.3 alkoxy; and [0439] R.sup.3 is H or
[0439] ##STR00124## [0440] where R.sup.4, R.sup.5, [0441] R.sup.6, R.sup.7, and R.sup.8 are independently selected from: H, OH, C.sub.1 to C.sub.3 hydroxyl, C.sub.1 to C.sub.3 alkoxy, and C.sub.1 to C.sub.3 alkyl; and [0442] X is NH or O.
[0443] In embodiments, R.sup.1 is CH.sub.2CHC.dbd.(CH.sub.3).sub.2. In embodiments, R.sup.1 is H. In embodiments, R.sup.2 is methoxy. In embodiments, R.sup.2 is H. In embodiments, R.sup.3 is H. In embodiments, R.sup.3 is
##STR00125##
[0444] Group P5 Compound
[0445] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00126##
[0446] Group Q Compounds
[0447] Group Q1 Compounds
[0448] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00127## [0449] where A is: [0450] a. CR.sup.9 where R.sup.9 is H, saturated or unsaturated C.sub.1 to C.sub.6 alkyl, C.sub.1 to C.sub.3 hydroxyl or CHR.sup.10, where R.sup.10 is C.sub.1 to C.sub.6 alkyl;
[0451] b. CCR.sup.64R.sup.65R.sup.66, where R.sup.64, R.sup.65, and R.sup.66 are independently selected from: C.sub.1 to C.sub.10 saturated or unsaturated, branched or straight chain alkyl, or (CH.sub.2).sub.aR.sup.11 where a is zero or 1 and R.sup.11 is H, OH, or saccharidyl, [0452] c. CR.sup.23, where R.sup.23 is saturated or unsaturated, substituted with an oxygen or unsubstituted five membered ring including at least one oxygen heteroatom, [0453] d. CR.sup.24, where R.sup.24 is
[0453] ##STR00128## [0454] where R.sup.25 and R.sup.26 are independently selected from H, OH, or C.sub.1 to C.sub.6 branched or straight hydroxyl, [0455] e. or along with Q and the atoms attached thereto form a ring structure of formula (Q1A) below,
[0455] ##STR00129## [0456] where R.sup.27 and R.sup.28 are H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 alkoxy, or C.sub.1 to C.sub.3 hydroxyl; [0457] B is CH.sub.2, or CH, with the caveat that if B is CH then D is C; [0458] D, E, F, and G are independently selected from: C, CR.sup.29, where R.sup.29 is H, OH, C.sub.1 to C.sub.3 alkyl, or C.sub.1 to C.sub.3 hydroxyl, with the caveat that if D is C, then B is CH; if E is C, then G is C; and if F is C, then u is zero; [0459] J is C(R.sup.30).sub.vR.sup.31, where v is zero or 1 and R.sup.30 and R.sup.31 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 hydroxyl, or
##STR00130##
[0459] where T and X are CH.sub.2 and where f and g are independently selected from zero (0), 1, or 2, and Z is selected from CR.sup.32R.sup.33, where R.sup.32 and R.sup.33 are independently selected from H, OH, C.sub.1 to C.sub.3 hydroxyl, or C.sub.1 to C.sub.3 alkyl, with the caveat that is v is zero then k is also zero; [0460] K is CR.sup.5.sub.uR.sup.6, where u is zero or 1 and R.sup.5 and R.sup.6 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 hydroxyl, C.sub.1 to C.sub.3 alkoxy, with the caveat that if u is zero than F is C; [0461] L is CR.sup.34.sub.kR.sup.35 where k is zero or 1 and R.sup.34 and R.sup.35 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 hydroxyl, with the caveat that if v is zero then k is zero; [0462] M is C(O), COC(O)R.sup.36 where R.sup.36 is C.sub.1 to C.sub.3 alkyl, CH(OH), CH(CH.sub.2).sub.jR.sup.37 where j is zero, 1, or 2 and R.sup.37 is H, OH, or saccharidyl; [0463] Q is C(O), or CR.sup.61R.sup.62, where R.sup.61 and R.sup.62 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 hydroxyl or along with A form a five membered ring containing an oxygen hetero atom, or structure Q1A; [0464] R.sup.1, R.sup.3, and R.sup.7 are independently selected from H, OH or C.sub.1 to C.sub.3 alkyl; [0465] R.sup.2, is selected from H, O, OH or C.sub.1 to C.sub.3 alkyl; and [0466] R.sup.4 and R.sup.8 are independently selected from H, C(O), OH, C.sub.1 to C.sub.3 alkyl, C(O)R.sup.63 where R.sup.63 is H or C.sub.1 to C.sub.3 alkyl, or OC(O)R.sup.64 where R.sup.64 is C.sub.1 to C.sub.3 alkyl, or H.
[0467] In embodiments, A is CR.sup.23, wherein R.sup.23 is saturated or unsaturated, substituted with an oxygen or unsubstituted five membered ring including at least one oxygen heteroatom.
[0468] In embodiments, R.sup.23 is
##STR00131##
[0469] In embodiments, A is CR.sup.9, where R.sup.9 is H, saturated or unsaturated C.sub.1 to C.sub.6 alkyl, C.sub.1 to C.sub.3 hydroxyl or CHR.sup.10, where R.sup.10 is C.sub.1 to C.sub.6 alkyl.
[0470] In embodiments, R.sup.9 is CHR.sup.10. In embodiments, R.sup.10 is CH.sub.3.
[0471] In embodiments, A along with Q and the atoms attached thereto form a ring structure of formula (Q1A) below,
##STR00132##
[0472] where R.sup.27 and R.sup.28 are H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 alkoxy, or C.sub.1 to C.sub.3 hydroxyl. In embodiments, R.sup.27 and R.sup.28 are each CH.sub.3.
[0473] In embodiments, A is CCR.sup.64R.sup.65R.sup.66, where R.sup.64, R.sup.65, and R.sup.66 are independently selected from: C.sub.1 to C.sub.10 saturated or unsaturated, branched or straight chain alkyl, or (CH.sub.2).sub.aR.sup.11 where a is zero or 1 and R.sup.11 is H, OH, or
##STR00133## [0474] where a' is zero or 1, R.sup.12, R.sup.13, R.sup.14, and R.sup.15 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.bOC(O)R.sup.16 where b is zero, 1, 2 or 3 and R.sup.16 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.dO(CH.sub.2).sub.d'R.sup.17, where d and d' are independently zero (0) or 1 and R.sup.17 is H or
##STR00134##
[0475] where R.sup.18, R.sup.19, R.sup.20 and R.sup.21 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.eOC(O)R.sup.22 where e is zero, 1, 2 or 3 and R.sup.22 is H or C.sub.1-C.sub.3 alkyl.
[0476] In embodiments, R.sup.64 is CH.sub.3, R.sup.65 is a C.sub.3 to C.sub.8 unsaturated, branched or chain alkyl, and R.sup.66 is (CH.sub.2).sub.aO(CH.sub.2).sub.a'R.sup.11 where a and a' are both zero and R.sup.11 is H or
##STR00135## [0477] where R.sup.12, R.sup.13, and R.sup.15 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, and R.sub.14 is (CH.sub.2).sub.dO(CH.sub.2).sub.d'R.sup.17, where d and d' are independently zero (0) or 1 and R.sup.17 is H or
##STR00136##
[0478] where R.sup.18, R.sup.19, R.sup.29 and R.sup.21 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, OC(O)R.sup.22 where R.sup.22 is H or C.sub.1-C.sub.3 alkyl.
[0479] In embodiments, R.sup.12, R.sup.13, and R.sup.15 are each independently OH or CH.sub.3.
[0480] In embodiments, R.sup.18, R.sup.19, and R.sup.21 are each independently OH or H and R.sup.20 is OC(O)CH.sub.3.
[0481] In embodiments, A is CR.sup.24, where R.sup.24 is
##STR00137##
where R.sup.25 and R.sup.26 are independently selected from H, OH, or C.sub.1 to C.sub.6 branched or straight hydroxyl. In embodiments, R.sup.26 is OH and R.sup.25 is C(CH.sub.3).sub.2OH.
[0482] In embodiments, Q is CR.sup.61R.sup.62, where R.sup.61 and R.sup.62 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 hydroxyl. In embodiments, Q is CH.sub.2. In embodiments, Q is CHOH. In embodiments, Q is C(O).
[0483] In embodiments, B is CH.sub.2. In embodiments, B is CH and D is C.
[0484] In embodiments, D is CR.sup.29, where R.sup.29 is H, OH, C.sub.1 to C.sub.3 alkyl, or C.sub.1 to C.sub.3 hydroxyl.
[0485] In embodiments, R.sup.29 is OH. In embodiments, R.sup.29 is H. In embodiments, R.sup.29 is CH.sub.3.
[0486] In embodiments, E is CR.sup.29, where R.sup.29 is H, OH, C.sub.1 to C.sub.3 alkyl, or C.sub.1 to C.sub.3 hydroxyl. In embodiments, R.sup.29 is H. In embodiments, R.sup.29 is CH.sub.3. In embodiments, E is C and G is C.
[0487] In embodiments, G is CH.
[0488] In embodiments, F is CH. In embodiments, K is CH and F is C.
[0489] In embodiments, K is CR.sup.5.sub.uR.sup.6, where u is 1 and R.sup.5 and R.sup.6 are independently selected from C.sub.1 to C.sub.3 alkyl. In embodiments, R.sup.5 and R.sup.6 are both CH.sub.3. In embodiments, R.sup.5 and R.sup.6 are both H.
[0490] In embodiments, both J and L are CH. In embodiments, J is CH.sub.2. In embodiments, J is C(R.sup.30).sub.vR.sup.31, where vi s 1, R.sup.30 and R.sup.31 is
##STR00138##
where T and X are CH.sub.2 and where f and g are independently selected from zero, 1, or 2, and Z is CR.sup.32R.sup.33, where R.sup.32 and R.sup.33 are independently selected from H, OH, C.sub.1 to C.sub.3 hydroxyl, or C.sub.1 to C.sub.3 alkyl. In embodiments, f and g are both zero and R.sup.32 and R.sup.33 are OH and C.sub.1 to C.sub.3 alkyl.
[0491] In embodiments, L is CH.sub.2.
[0492] In embodiments, M is CHOH. In embodiments, M is CH(CH.sub.2).sub.jR.sup.37 where j is zero, 1, or 2 and R.sup.37 is H or
##STR00139## [0493] where j' is zero or 1, R.sup.38, R.sup.39, R.sup.40, and R.sup.41 are each independently selected from H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, C.sub.1-C.sub.3 alkoxy, COOH, (CH.sub.2).sub.mOC(O)R.sup.42 where m is zero, 1, 2 or 3 and R.sup.42 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.nO(CH.sub.2).sub.n'R.sup.43, where n and n' are independently zero or 1 and R.sup.43 is H or
[0493] ##STR00140## [0494] where R.sup.44, R.sup.45, R.sup.46 and R.sup.47 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.pOC(O)R.sup.48 where p is zero, 1, 2 or 3 and R.sup.48 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.qO(CH.sub.2).sub.q'R.sup.49 where q and q' are independently zero or 1 and R.sup.49 is H or
[0494] ##STR00141## [0495] where R.sup.50, R.sup.51, R.sup.52 and R.sup.53 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.rOC(O)R.sup.54 where r is zero, 1, 2 or 3 and R.sup.54 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.sO(CH.sub.2).sub.s'R.sup.55 where s and s' are independently zero or 1 and R.sup.55 is H or
[0495] ##STR00142## [0496] where R.sup.56, R.sup.57, R.sup.58 and R.sup.59 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.tOC(O)R.sup.60 where t is zero, 1, 2 or 3 and R.sup.60 is H or C.sub.1-C.sub.3 alkyl.
[0497] In embodiments, j and j' are both zero and R.sup.38 is CH.sub.3; R.sup.39 is OH; R.sup.40 is OCH.sub.3; and R.sup.41 is H. In embodiments, j and j' are both zero and R.sup.39 is (CH.sub.2)--O(CH.sub.2).sub.n'R.sup.43, where n and n' are independently zero or 1 and R.sup.43 is H or
##STR00143## [0498] where R.sup.44, R.sup.45, R.sup.46 and R.sup.47 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.pOC(O)R.sup.48 where p is zero, 1, 2 or 3 and R.sup.48 is H or C.sub.1-C.sub.3 alkyl.
[0499] In embodiments, R.sup.44 is CH.sub.2OH; and R.sup.45, R.sup.46, and R.sup.47 are all OH.
[0500] In embodiments, M is CH(CH.sub.2).sub.jR.sup.37 where R.sup.37 is H, OH, or
##STR00144## [0501] where j' is zero or 1, R.sup.38 and R.sup.41 are each independently selected from H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, C.sub.1-C.sub.3 alkoxy, COOH, (CH.sub.2).sub.mOC(O)R.sup.42 where m is zero, 1, 2 or 3 and R.sup.42 is H or C.sub.1-C.sub.3 alkyl; [0502] where R.sup.39 is OR.sup.43, where R.sup.43 is H or
[0502] ##STR00145## [0503] where R.sup.44, R.sup.45 and R.sup.47 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.pOC(O)R.sup.48 where p is zero, 1, 2 or 3 and R.sup.48 is H or C.sub.1-C.sub.3 alkyl; and R.sup.46 is (CH.sub.2).sub.qO(CH.sub.2).sub.q'R.sup.49 where q and q' are independently zero or 1 and R.sup.49 is H or
[0503] ##STR00146## [0504] where R.sup.50, R.sup.51 and R.sup.53 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.rOC(O)R.sup.54 where r is zero, 1, 2 or 3 and R.sup.54 is H or C.sub.1-C.sub.3 alkyl; and R.sup.52 is OR.sup.55 where R.sup.55 is H or
[0504] ##STR00147## [0505] where R.sup.56, R.sup.57, R.sup.58 and R.sup.59 are each independently H, OH, CH.sub.3 or C.sub.1-C.sub.3 hydroxyl; and [0506] R.sup.40 is OR.sup.43, and R.sup.43 is H or
[0506] ##STR00148## [0507] where [0508] R.sup.44, R.sup.45, R.sup.46 and R.sup.47 are each independently OH, or C.sub.1-C.sub.3 hydroxyl.
[0509] In embodiments, M is CO.
[0510] In embodiments, R.sup.1 is H or C.sub.1-C.sub.3 alkyl. In embodiments R.sup.1 is CH.sub.3.
[0511] In embodiments, R.sup.2 is H, OH, O, or C.sub.1-C.sub.3 alkyl.
[0512] In embodiments, R.sup.3 is H or C.sub.1-C.sub.3 alkyl.
[0513] In embodiments, R.sup.4 is CH.sub.3. In embodiments, R.sup.4 is CHO.
[0514] In embodiments, R.sup.7 and R.sup.8 are both H. In embodiments, R.sup.8 is OCOCH.sub.3.
[0515] In embodiments, F is CR.sup.29, where R.sup.29 is H, OH, C.sub.1-C.sub.3 alkyl, or C.sub.1-C.sub.3 hydroxyl.
[0516] In embodiments, K is CR.sup.5R.sup.6.
[0517] In embodiments, M is C(O), COC(O)R.sup.36, where R.sup.36 is C.sub.1-C.sub.3 alkyl, CH(CH.sub.2).sub.jR.sup.37 where j is zero or 1.
[0518] Group Q1B Compounds
[0519] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00149## [0520] where A is: [0521] a. CCR.sup.64R.sup.65R.sup.66, where R.sup.64, R.sup.65, and R.sup.66 are independently selected from: C.sub.1 to C.sub.10 saturated or unsaturated, branched or straight chain alkyl, or (CH.sub.2).sub.aR.sup.11 where a is zero or 1 and R.sup.11 is H, OH, or saccharidyl, [0522] b. or along with Q and the atoms attached thereto form a ring structure of formula (Q1B') below,
[0522] ##STR00150## [0523] where R.sup.27 and R.sup.28 are H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 alkoxy, or C.sub.1 to C.sub.3 hydroxyl; [0524] D, E, F, and G are independently selected from: CR.sup.29, where R.sup.29 is H, OH, C.sub.1 to C.sub.3 alkyl, or C.sub.1 to C.sub.3 hydroxyl; [0525] K is CR.sup.5R.sup.6, where R.sup.5 and R.sup.6 are independently selected from H, or C.sub.1 to C.sub.3 alkyl; [0526] M is CH(CH.sub.2).sub.jR.sup.37 where j is zero, 1, or 2 and R.sup.37 is H, OH, or saccharidyl; [0527] Q is CHOH, or along with A form a five membered ring containing an oxygen heteroatom, or structure II; [0528] R.sup.1, R.sup.2 R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7 and R.sup.8 are independently selected from H, OH, 0, or C.sub.1 to C.sub.3 alkyl.
[0529] In embodiments, A along with Q and the atoms attached thereto form a ring structure of formula (Q1B') below,
##STR00151## [0530] where R.sup.27 and R.sup.28 are H, OH, C.sub.1 to C.sub.3 alkyl, C.sub.1 to C.sub.3 alkoxy, or C.sub.1 to C.sub.3 hydroxyl. In embodiments R.sup.27 and R.sup.28 are each CH.sub.3.
[0531] In embodiments, A is CCR.sup.64R.sup.65R.sup.66, where R.sup.64, R.sup.65, and R.sup.66 are independently selected from: C.sub.1 to C.sub.10 saturated or unsaturated, branched or straight chain alkyl, or (CH.sub.2).sub.aO(CH.sub.2).sub.a'R.sup.11 where a and a' are independently zero or 1 and R.sup.11 is saccharidyl and is:
##STR00152## [0532] where a' is zero or 1, R.sup.12, R.sup.13, R.sup.14, and R.sup.15 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.bOC(O)R.sup.16 where b is zero, 1, 2 or 3 and R.sup.16 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.dO(CH.sub.2).sub.d'R.sup.17, where d and d' are independently zero (0) or 1 and R.sup.17 is H or
[0532] ##STR00153## [0533] where R.sup.18, R.sup.19, R.sup.20 and R.sup.21 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.eOC(O)R.sup.22 where e is zero, 1, 2 or 3 and R.sup.22 is H or C.sub.1-C.sub.3 alkyl.
[0534] In embodiments, R.sup.64 is CH.sub.3, R.sup.65 is a C.sub.3 to C.sub.8 unsaturated, branched or chain alkyl, and R.sup.66 is (CH.sub.2).sub.aO(CH.sub.2).sub.a'R.sup.11 where a and a' are both zero and R.sup.11 is saccharidyl and is:
##STR00154##
[0535] where R.sup.12, R.sup.13, and R.sup.15 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, and R.sup.14 is (CH.sub.2).sub.dO(CH.sub.2).sub.d'R.sup.17, where d and d' are independently zero (0) or 1 and R.sup.17 is H or
##STR00155##
[0536] where R.sup.18, R.sup.19, R.sup.20 and R.sup.21 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, OC(O)R.sup.22 where R.sup.22 is H or C.sub.1-C.sub.3 alkyl.
[0537] In embodiments, R.sup.12, R.sup.13, and R.sup.15 are each independently OH or CH.sub.3.
[0538] In embodiments, R.sup.18, R.sup.19, and R.sup.21 are each independently OH or H and R.sup.20 is OC(O)CH.sub.3.
[0539] In embodiments, M is CH(CH.sub.2).sub.jR.sup.37 where j is zero, 1, or 2 and R.sup.37 is saccharidyl and is:
##STR00156##
[0540] where j' is zero or 1, R.sup.38, R.sup.39, R.sup.40, and R.sup.41 are each independently selected from H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, C.sub.1-C.sub.3 alkoxy, COOH, (CH.sub.2).sub.mOC(O)R.sup.42 where m is zero, 1, 2 or 3 and R.sup.42 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.nO(CH.sub.2).sub.n'R.sup.43, where n and n' are independently zero (0) or 1 and R.sup.43 is H or
##STR00157##
[0541] where R.sub.44, R.sup.45, R.sup.46 and R.sup.47 each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.pOC(O)R.sup.48 where p is zero, 1, 2 or 3 and R.sup.48 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.qO(CH.sub.2).sub.q'R.sup.49 where q and q' are independently zero or 1 and R.sup.49 is H or
##STR00158##
[0542] where R.sup.50, R.sup.51, R.sup.52 and R.sup.53 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.rOC(O)R.sup.54 where r is zero, 1, 2 or 3 and R.sup.54 is H or C.sub.1-C.sub.3 alkyl, or (CH.sub.2).sub.sO(CH.sub.2).sub.s'R.sup.55 where s and s' are independently zero or 1 and R.sup.55 is H or
##STR00159##
[0543] where R.sup.56, R.sup.57, R.sup.58 and R.sup.59 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.tOC(O)R.sup.60 where t is zero, 1, 2 or 3 and R.sup.60 is H or C.sub.1-C.sub.3 alkyl.
[0544] In embodiments, j and j' are both zero and R.sup.38 is CH.sub.3; R.sup.39 is OH; R.sup.40 is OCH.sub.3; and R.sup.41 is H.
[0545] In embodiments, j and j' are both zero and R.sup.39 is (CH.sub.2).sub.nO(CH.sub.2).sub.n'R.sup.43, where n and n' are independently zero or 1 and R.sup.43 is saccharidyl and is:
##STR00160## [0546] where R.sup.44, R.sup.45, R.sup.46 and R.sup.47 are each independently H, OH, CH.sub.3, C.sub.1-C.sub.3 hydroxyl, COOH, (CH.sub.2).sub.pOC(O)R.sup.48 where p is zero, 1, 2 or 3 and R.sup.48 is H or C.sub.1-C.sub.3 alkyl.
[0547] In embodiments, R.sup.44 is CH.sub.2OH; and R.sup.45, R.sup.46, and R.sup.47 are all OH.
[0548] In embodiments, D is CR.sup.29, where R.sup.29 is C.sub.1 to C.sub.3 alkyl.
[0549] In embodiments, E is CR.sup.29, where R.sup.29 is C.sub.1 to C.sub.3 alkyl.
[0550] In embodiments, G is CH.
[0551] In embodiments, F is CH.
[0552] In embodiments, K is CH and F is C.
[0553] In embodiments, K is CR.sup.5.sub.uR.sup.6, where u is 1 and R.sup.5 and R.sup.6 are independently selected from C.sub.1 to C.sub.3 alkyl. In embodiments, R.sup.5 and R.sup.6 are both CH.sub.3.
[0554] In embodiments, R.sup.2, R.sup.4, R.sup.8, and R.sup.9 are all H.
[0555] In embodiments, R.sup.1 is C.sub.1-C.sub.3 alkyl. In embodiments, R.sup.1 is H.
[0556] In embodiments, R.sup.3 is O. In embodiments, R.sup.3 is H.
[0557] Group Q2 Compounds
[0558] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00161## [0559] where A is C or CR.sup.4, where R.sub.4 is H, OH, C.sub.1 to C.sub.3 alkyl, with the caveat that if A is C, then B is CR.sup.6; [0560] B is CR.sup.5.sub.aR.sup.6, where a is zero (0) or 1, and R.sup.5 and R.sup.6 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl, with the caveat that if A is C, then B is CR.sup.6; [0561] X is H, or along with Y and the atoms attached thereto form a ring structure having formula (Q2A) below,
[0561] ##STR00162## [0562] where R.sup.9 and R.sup.10 are independently selected from O and CH.sub.2; [0563] Z is H or along with Y and the atoms attached thereto form a ring structure having formula (Q2B) below,
[0563] ##STR00163## [0564] where R.sup.13 and R.sup.14 are independently selected from O and CH.sub.2; [0565] Y together with either X or Z forms a ring structure of formula Q2A or Q2B above; and [0566] R.sup.1, R.sup.2 and R.sup.3 are independently selected from H, CH.sub.2, C.sub.1 to C.sub.3 alkyl.
[0567] In embodiments, A is CH.sub.3. In embodiments, R.sup.1 is CH.sub.2. In embodiments, R.sup.2 is H. In embodiments, R.sup.3 is H. In embodiments, A is CR.sup.4, where R.sup.4 is C.sub.1 to C.sub.3 alkyl. In embodiments, B is CH.sub.2.
[0568] In embodiments, X and Y along with the atoms attached thereto form a ring structure having formula (Q2A) below,
##STR00164##
In embodiments, R.sup.9 is O and R.sup.10 is CH.sub.2.
[0569] In embodiments, Y and Z along with the atoms attached thereto form a ring structure having formula (Q2B) below,
##STR00165## [0570] where R.sup.13 and R.sup.14 are independently selected from O and CH.sub.2. In embodiments, R.sup.3 is CH.sub.3. In embodiments, R.sup.1 is H. In embodiments, R.sup.2 is CH.sub.2. In embodiments, R.sup.2 is CH.sub.3.
[0571] Group Q3 Compounds
[0572] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00166## [0573] where A is C or CR.sup.4, where R.sub.4 is H, OH, C.sub.1 to C.sub.3 alkyl, with the caveat that if A is C, then B is CR.sup.6; [0574] B is CR.sup.5.sub.aR.sup.6, where a is zero or 1, and R.sup.5 and R.sup.6 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl; [0575] Z is H or CR.sup.11R.sup.12 where R.sup.11 and R.sup.12 are independently selected from H, OH, C.sub.1 to C.sub.3 alkyl; [0576] R.sup.1, R.sup.2 and R.sup.3 are independently selected from H, CH.sub.2, or a saturated or unsaturated C.sub.1 to C.sub.6 alkyl; and [0577] R.sup.9 and R.sup.10 are independently selected from O or CH.sub.2.
[0578] In embodiments, A is CCH.sub.3. In embodiments, B is CH.sub.2. In embodiments, Z is H. In embodiments, R.sup.1 is CH.sub.2. In embodiments, R.sub.2 is H. In embodiments, R.sub.3 is H. In embodiments, R.sup.9 is O. In embodiments, R.sup.10 is CH.sub.2.
[0579] Group R Compounds
[0580] Group R1 Compounds
[0581] In embodiments a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00167##
[0582] where R.sup.1 is OH or saccharidyl;
[0583] R.sup.2 and R.sup.3 are independently H, OH, and COOH; and
[0584] R.sup.4 is C.sub.3-C.sub.14 saturated or unsaturated alkyl optionally substituted with one or more hydroxyls.
[0585] In embodiments R.sup.1 is OH. In embodiments R.sup.2 is H. In embodiments, R.sup.2 is OH. In embodiments, R.sup.3 is H. In embodiments, R.sup.3 is COOH. In embodiments, R.sup.4 is C.sub.9 to C.sub.13 unsaturated alkyl. In embodiments, R.sup.4 is C.sub.9 to C.sub.13 unsaturated alkyl with at least one hydroxyl. In embodiments, R.sup.4 is C.sub.10 to C.sub.12 alkyl with at least two double bonds, such as C.sub.11 with at least two double bonds. In embodiments, R.sup.4 is C.sub.11 with three double bonds. In embodiments, R.sup.4 is C.sub.11 with three double bonds and one hydroxyl. In embodiments, R.sup.4 is C.sub.3 to C.sub.7 with at least one double bond.
[0586] In embodiments of a compound according to Formula R.sub.1 is a compound where R.sup.2 and R.sup.3 are independently H, OH, and COOH; and R.sup.4 is C.sub.3-C.sub.10 saturated or unsaturated alkyl optionally substituted with one or more hydroxyls. In embodiments, R.sup.2 is OH. In embodiments, R.sup.3 is COOH. In embodiments, R.sup.4 is C.sub.3 to C.sub.7 with at least one double bond.
[0587] Group R2 Compounds
[0588] In embodiments a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00168##
[0589] where
[0590] A is (CH.sub.2).sub.x, where x is 0 or 1;
[0591] R.sup.5 is saccharidyl;
[0592] R.sup.2 and R.sup.3 are independently H, OH, and COOH; and
[0593] R.sup.4 is C.sub.8-C.sub.16 saturated or unsaturated alkyl optionally substituted with one or more hydroxyls.
[0594] In embodiments, a compound of formula R.sub.2 the following structure:
##STR00169##
[0595] where
[0596] A, D, E and G are independently (CH.sub.2).sub.x, where x is 0 or 1;
[0597] R.sup.1 is H or saccharidyl;
[0598] R.sup.2 and R.sup.3 are independently H, OH, and COOH; and
[0599] R.sup.4 is C.sub.8-C.sub.16 saturated or unsaturated alkyl optionally substituted with one or more hydroxyls.
[0600] In embodiments, R.sup.1 is H. In embodiments, R.sup.2 is H. In embodiments, R.sup.3 is H. In embodiments, R.sup.4 is C.sub.8 to C.sub.14 unsaturated alkyl optionally substituted with one or more hydroxyls, such as C.sub.10 to C.sub.13 unsaturated alkyl optionally substituted with one or more hydroxyls. In embodiments, R.sup.4 is C.sub.11 unsaturated alkyl optionally substituted with one or more hydroxyls. In embodiments, R.sup.4 is C.sub.ii unsaturated alkyl with one double bond optionally substituted with one or more hydroxyls. In embodiments, x is one in A and E; and x is zero in D and E.
[0601] Group R3 Compounds
[0602] In embodiments a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00170##
[0603] where [0604] R.sup.1 is H, or OH; [0605] R.sup.2 and R.sup.3 are independently H, OH, or CH.sub.2OH; [0606] X is O or CHOH; [0607] Y is C.dbd.O or CH.sub.2; and [0608] R.sup.4 is C.sub.3-C.sub.12 unsaturated alkyl.
[0609] In embodiments, R.sup.1 is OH. In embodiments, R.sup.2 is H. In embodiments, X is CHOH. In embodiments, Y is CH.sub.2. In embodiments, R.sup.3 is H.
[0610] In embodiments, R.sup.4 is a C.sub.3 to C.sub.7 unsaturated alkyl. In embodiments, R.sup.4 is a C.sub.5 unsaturated alkyl. In embodiments, R.sup.4 is a C.sub.5 alkyl with one double bond.
[0611] In embodiments, R.sup.3 is OH.
[0612] In embodiments, R.sup.4 is a C.sub.8 to C.sub.12 unsaturated alkyl. In embodiments, R.sup.4 is a C.sub.10 unsaturated alkyl. In embodiments, R.sup.4 is a C.sub.10 alkyl with one double bond.
[0613] Group R4 Compounds
[0614] In embodiments a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00171## [0615] where [0616] X is O or NH; [0617] R.sup.1 is H, OH, or CH.sub.2OH; [0618] R.sup.2 is H or OCOR.sup.5, wherein R.sup.5 is a C.sub.1-C.sub.3 alkyl; and [0619] R.sup.3 is H or C.sub.1-C.sub.10 saturated or unsaturated alkyl.
[0620] In embodiments, X is O. In embodiments, R.sup.1 is OH. In embodiments, R.sup.2 is OCOR.sup.5. In some embodiments where R.sup.2 is OCOR.sup.5, R.sup.5 is CH.sub.3. In embodiments, R.sup.3 is C.sub.5 to C.sub.9 saturated or unsaturated alkyl, such as C.sub.7 unsaturated alkyl. In embodiments, R.sup.3 is C.sub.7 alkyl with one double bond.
[0621] In embodiments, X is NH. In some embodiments where X is NH, R.sup.1 and R.sup.2 are both H. In some embodiments where X is NH, R.sup.3 is H.
[0622] Group S Compounds
[0623] In embodiments a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00172## [0624] where: [0625] X and Y are each independently C.sub.7-C.sub.20 straight or branched chain unsaturated alkyl unsubstituted or substituted with one or more OH; [0626] A is saccharidyl; and [0627] R.sup.10 and R.sup.11 are each independently H, OH or saccharidyl.
[0628] In embodiments a compound according to Formula S has the following structure:
##STR00173## [0629] where: [0630] a, b, d, e, f and g are each independently zero or 1; [0631] X and Y are each independently C.sub.7-C.sub.20 straight or branched chain unsaturated alkyl unsubstituted or substituted with one or more OH; [0632] R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7, R.sup.8, and R.sup.9 are each independently H, OH, CH.sub.3, CH.sub.2CH.sub.3, and CH.sub.2OH; and [0633] R.sup.10 and R.sup.11 are each independently H, OH or saccharidyl.
[0634] In embodiments, one or both of R.sup.10 and R.sup.11 are saccharidyl and are independently
##STR00174## [0635] where [0636] i, j and k are each independently zero or 1, and [0637] R.sup.12, R.sup.13, R.sup.14, R.sup.15, R.sup.16, R.sup.17 and R.sup.18 are each independently H, OH, CH.sub.3, CH.sub.2CH.sub.3 and CH.sub.2OH.
[0638] In embodiments, X and Y are each independently C.sub.8-C.sub.16 straight or branched chain unsaturated alkyl unsubstituted or substituted with one or more OH and having 2 to 6 double bounds. In embodiments, X and Y are each independently C.sub.10-C.sub.15 straight or branched chain unsaturated alkyl unsubstituted or substituted with one or more OH and having 2 to 4 double bounds.
[0639] In embodiments a compound according to Formula S has the following structure:
##STR00175## [0640] where a, b, d, e, f and g, and R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.5, R.sup.6, R.sup.7, R.sup.8, R.sup.9, R.sup.10 and R.sup.11 are as defined above; and wherein [0641] R.sup.19, R.sup.20, R.sup.21, R.sup.22 and R.sup.23 are each independently H, CH.sub.3 or OH.
[0642] In embodiments a compound according to Formula S has the following structure:
##STR00176## [0643] where R.sup.19, R.sup.20, R.sup.23 and R.sup.24 are each independently H, CH.sub.3 or OH.
[0644] Group T Compounds
[0645] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00177## [0646] where: [0647] R.sup.1 is H, or COR.sup.4 where R.sup.4 is H, or saturated or unsaturated C.sub.1-C.sub.6 straight or branched alkyl; and [0648] R.sup.2, R.sup.3, R.sup.a, R.sup.b, R.sup.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, and R.sup.j are independently selected from H, or saturated or unsaturated C.sub.1-C.sub.3 alkyl.
[0649] In embodiments, R.sup.2, R.sup.3, R.sup.a, R.sup.b, R.sup.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, and R.sup.j are independently selected from H, or saturated or unsaturated C.sub.1-C.sub.3 alkyl with the caveat that saturated or unsaturated C.sub.1-C.sub.3 alkyl substituents are not directly adjacent each other.
[0650] In embodiments, R.sup.2 is a saturated or unsaturated C.sub.1-C.sub.3 alkyl, such as methyl. In embodiments, R.sup.3 is a saturated or unsaturated C.sub.1-C.sub.3 alkyl, such as methyl. In embodiments, R.sup.2 is CH.sub.3 and R.sup.3 is CH.sub.3.
[0651] In embodiments, R.sup.1 is H. In embodiments, R.sup.1 is COCH.sub.3. In embodiments, R.sup.1 is COR.sup.4 and R.sup.4 is
##STR00178##
where R.sup.5 and R.sup.6 are independently selected from H, or saturated or unsaturated C.sub.1-C.sub.4.
[0652] In embodiments, R.sup.5 and R.sup.6 are both CH.sub.3.
[0653] In embodiments a compound according to Formula T is a compound where (i) R.sup.2, R.sup.3, R.sup.a, R.sup.b, R.sup.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, and R.sup.j are independently selected from H, or saturated or unsaturated C.sub.1-C.sub.3 alkyl; and (ii) R.sup.7 is H or saturated or unsaturated C.sub.1-C.sub.3 alkyl. In such embodiments, R.sup.7 may be CH.sub.3. R.sup.2, R.sup.3, R.sup.a, R.sup.b, R.sup.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, and R.sup.j may independently be selected from H, or saturated or unsaturated C.sub.1-C.sub.3 alkyl with the caveat that saturated or unsaturated C.sub.1-C.sub.3 alkyl substituents are not directly adjacent each other. R.sup.2 may be a saturated or unsaturated C.sub.1-C.sub.3 alky, such as methyl. R.sup.3 may be a saturated or unsaturated C.sub.1-C.sub.3 alkyl, such as methyl. In some of such embodiment, R.sup.2 is CH.sub.3 and R.sup.3 is CH.sub.3.
[0654] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00179##
[0655] where [0656] R.sup.2, R.sup.3, R.sup.a, R.sup.b, R.sup.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, and R.sup.j are independently selected from H, or saturated or unsaturated C.sub.1-C.sub.3 alkyl; and [0657] R.sup.7 is H or saturated or unsaturated C.sub.1-C.sub.3 alkyl.
[0658] Group U Compounds
[0659] In embodiments, a bioactive, taste modulating, or salty taste modulating compound is a compound having the following structure:
##STR00180##
[0660] where: [0661] R.sup.1 is H, OH, or C.sub.1-C.sub.3 alkoxy; [0662] R.sup.a, R.sup.b, R.sup.c, R.sup.d, and R.sup.e are each independently selected from: H, OH, or C.sub.1-C.sub.3 alkoxy; [0663] R.sup.2 is H, OH, C.sub.1-C.sub.3 alkoxy, or R.sup.2 and R.sup.3 together form
[0663] ##STR00181## [0664] to form a compound of the following structure
[0664] ##STR00182## [0665] R.sup.3 is H, OH, C.sub.1-C.sub.3 alkoxy, or R.sup.2 and R.sup.3 together form a ring structure as indicated above; [0666] R.sup.4 is H, OH, or C.sub.1-C.sub.3 alkoxy; [0667] X is O or CH; and [0668] Y is O or CR.sup.5, where R.sup.5 is H or
[0668] ##STR00183## [0669] where [0670] R.sup.6, R.sup.7, and R.sup.8 are each independently selected from: H, OH, or C.sub.1-C.sub.3 alkoxy; [0671] R.sup.f, R.sup.g, R.sup.h, R.sup.i, R.sup.j, and R.sup.k are each independently selected from: H, OH, or C.sub.1-C.sub.3 alkoxy, [0672] wherein the at least one compound of formula U is present in the food product in an amount sufficient to enhance a perception of saltiness of the food product.
[0673] In embodiments, R.sup.1 is H. In embodiments, R.sup.1 is --OCH.sub.3. In embodiments, R.sup.2 is H. In embodiments, R.sup.3 is H. In embodiments, both R.sup.2 and R.sup.3 are H.
[0674] In embodiments where R.sup.2 and R.sup.3 together form
##STR00184##
to form a compound of the following structure
##STR00185##
R.sup.4 is H.
[0675] In embodiments where R.sup.2 and R.sup.3 form
##STR00186##
to form a compound of the following structure
##STR00187##
R.sup.4 is H.
[0676] In embodiments, R.sup.a, R.sup.b, R.sup.c, R.sup.d, and R.sup.e are independently H or OH.
[0677] In embodiments, Y is O. In embodiments, Y is CR.sup.5, where CR.sup.5 is
##STR00188##
R.sup.6 may be OH. R.sup.7 may be OH. R.sup.8 may be OH. R.sup.f, R.sup.g, R.sup.h, R.sup.i, R.sup.j, or R.sup.k may independently be H or OH.
[0678] In embodiments, a compound according to Formula U is a compound having the following structure:
##STR00189##
[0679] where: [0680] R.sup.1, R.sup.2, R.sup.3, R.sup.4, R.sup.6, R.sup.7, and R.sup.8 are each independently H, OH, or C.sub.1-C.sub.3 alkoxy; [0681] X and Z are each independently O or CH; and [0682] R.sup.a, R.sup.b, R.sup.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, R.sup.j, and R.sup.k are each independently H, OH, or C.sub.1-C.sub.3 alkoxy.
[0683] In embodiments, a compound of Formula UA is a compound where R.sup.1 is OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.2 is H. In embodiments, R.sup.3 is OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.4 is OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.6 is OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.7 is OH or C.sub.1-C.sub.3 alkoxy. In embodiments, R.sup.a, R.sup.b, R.sub.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, R.sup.j, and R.sup.k are each independently H or OH. In embodiments, R.sup.a, R.sup.b, R.sub.c, R.sup.d, R.sup.e, R.sup.f, R.sup.g, R.sup.h, R.sup.i, R.sup.j, and R.sup.k are each H. In embodiments, X is O. In embodiments, Z is O.
[0684] For purpose of convenience, Table 2A lists the initially selected 99 compounds (with compounds 12 and 20 being the same compound isolated from different sources) and indicates which categories and subcategories, if appropriate, into which each of the compounds falls. Table 2B lists the categories and the compound that fall within each category.
TABLE-US-00002 TABLE 2A Listing of compounds according to category Compound Category Sub-Categories 1 A -- 2 -- 3 -- 4 -- 5 -- 6 -- 7 -- 8 A and B B1, B3 9 A -- 10 B B1, B3 11 B1, B3 12 B1, B3 13 B1 14 B1' 15 B1' 16 B2, B4 17 B2, B4 18 B1' 19 B1, B3 20 B1, B3 21 B1 22 B1' 23 C C'', C''' 24 C'', C''', C'''' 25 C'', C''' 26 C', C''' 27 C' 28 -- 29 D -- 30 -- 31 -- 32 E -- 33 -- 34 F -- 35 F', F'' 36 F' 37 Q Q1, Q1A, Q1B 38 Q1, Q1B 39 F F' 40 G -- 41 H -- 42 F -- 43 -- 44 I -- 45 -- 46 -- 47 I' 48 J J1, J1'' 49 J1, J1'' 50 J1, J1', J2 51 J2 52 K -- 53 L 54 55 L' 56 M -- 57 N -- 58 O -- 59 P P5 60 P2, P2' 61 P4 62 P1 63 P1 64 P1 65 P1 66 P4 67 P2 68 P2, P3 69 P2 70 P1 71 P1 72 P1 73 Q Q1 74 Q1 75 Q1 76 Q1 77 Q1 78 Q1 79 Q2, Q2A, Q3 80a Q2, Q2B 80b Q2, Q2B 81 R R1 82 R3 83 R4 84 R4 85 R1 86 R1 87 R3 88 R1 89 R1 90 R2 91 S -- 92 -- 93 T -- 94 T2 95 -- 96 U U' 97 UA 98 U'' 99 --
TABLE-US-00003 TABLE 2B Listing of compounds according to category Category Compounds A 1-9 B 8, 10-22 C 23-28 D 29-31 E 32, 33 F 34-36, 39, 42, 43 G 40 H 41 I 44-47 J 48-51 K 52 L 53-55 M 56 N 57 O 58 P 59-72 Q 37, 38, 73-79, 80a, 80b R 81-90 S 91, 92 T 93-95 U 96-99
[0685] As can be seen from the structures of the compounds provided above, many of the compounds have structural similarities. Accordingly, it is believed that structural derivatives of the specific compounds presented above would also have the ability to elicit the perception of saltiness or enhance saltiness. Combinations of the compounds could also serve to elicit the perception of saltiness or enhance saltiness. In addition or alternatively, one or more of the compounds may elicit the perception of other simple or complex flavors, other than or in addition to saltiness.
[0686] Many of the structural similarities between the compounds are reflected in the compounds of Formulas A through U, as well as subclasses thereof, presented above. It will be further understood, based on the compounds identified herein, that one or more of gingerols, alkyl substituted phenols, acridone alcaloids, labdanes, primaranes, saponines, neolignans, pentacyclic triterpenes, 2,2'-cyclolignans, dibenzylbutane lignans, bicyclic triterpenes, phloroglucines, carylophyllenes, beta-carbolines, limnoids, cumarines, cardanolide steroids, fatty acids, and derivatives may be candidates for taste modulating compounds. It will be further understood that other structural similarities of the compounds presented herein may be exploited to develop taste modulating compounds.
[0687] By way of example, many of the compounds presented herein have unsaturated carbon chains of at least 11 carbons with attached hydroxyl groups and may be amphiphilic, with hydrophobic head portions and hydrophobic tails. Other compounds having, for example C.sub.5-C.sub.20 alkane or alkene tails may elicit or enhance saltiness. Similarly, other compounds with differently substituted carboxyl or hydroxyl groups may elicit or enhance the perception of saltiness.
[0688] Many compounds presented herein have large numbers of cyclic groups having a central portion that may be hydrophobic and peripheral regions that may be hydrophilic. More specifically, some compounds include pentacyclohexane with hydroxyl groups, attached sugars and at least one ester linkage. Substitution of the central hydrophobic ring structure with, for example, C.sub.5-C.sub.20 hydrophobic alkyl or alkene groups, may result in compounds that may elicit or enhance saltiness. Alternative substitution of hydroxyl groups at the peripheral regions or substitution with carboxylic groups may also result in compounds that elicit or enhance the perception of saltiness.
[0689] Many of the compounds presented above have one or more aromatic ring structures, with some being substituted and some being unsubstituted. Similar substitution or unsubstitution of such compounds may result in compounds that enhance or elicit saltiness.
[0690] A plurality of compounds presented herein include saturated carbon chains of at least 9 carbons and one oxygen containing group such as hydroxyl, carbonyl, carboxyl, or ester. Other compounds having unsaturated carbon chains of, for example, 5 to 15 carbons and an oxygen group may have similar effects with regard to salty taste.
[0691] Many compounds presented herein have at least one phenol group with an ether group and a carbon side chain comprising at least seven carbons. Other similar compounds may have similar effects with regard to salty taste.
[0692] A number of compounds presented herein have a benzyl heterocyclic furan with various attached groups containing unsaturated carbon linkages and at least one carbonyl group. Other similar compounds may have similar activity with regard to salty taste.
[0693] A plurality of compounds presented above contain a cyclopentaphenthrene group. Other compounds having a cyclopentaphenthrene and similar substituents may have similar activity with regard to salty taste.
[0694] A number of the compounds presented herein include a benzopyranone group. Other compounds having a benzopyranone and similar substituents may have similar activity with regard to salty taste.
[0695] Some compounds presented above have unsubstituted carbon chain with a minimum of 13 carbons and at least one carbonyl group. Other similar compounds may have similar activity with regard to salty taste.
[0696] A plurality of compounds presented herein have a methoxymethyltetrahydrobenzo-cyclooctabenzo-dioxole or -annulene group. Other compounds having such groups may have similar activity with regard to salty taste.
[0697] A number of compounds presented above have tetracyclohexane with an attached ester or carbonyl moiety. Other similar compounds may have similar activity with regard to salty taste.
[0698] Many of the compounds can be classified as lactones, lignol-like compounds, oxylipins, polyisoprene glycosides, triterpenoid glycosides, alkylamides, or gamma pyrenes. Other similar compounds may also elicit or enhance salty taste perception.
[0699] It will be understood that derivations of the compounds discussed above are provided for purposes of example and that other derivatives or derivations may be made based on structural similarities between the various compounds, resulting in compounds that elicit or enhance perception of saltiness.
[0700] Evaluation of Salty Taste or Salty Taste Enhancement
[0701] Many of the identified compounds were tested by tasters and rated for perception of saltiness in combination with reduced amounts of sodium chloride and were assigned a rating (DAP score) for saltiness. Briefly, each individual tested compound was placed in water and in sodium solution to test saltiness and saltiness enhancement potential. Tests in water were executed in a compound concentration of 10 ppm. Tests in sodium solution were executed in compound concentrations of 0.1, 1 and 10 ppm. Two control sodium solutions with known organoleptic salt intensities were provided as references for each test. The test for individual compounds was also conducted using simple broth instead of sodium solution. A number of compound combinations identified from the Na-solution DAP test were used for the broth DAP test. Tests were executed with a trained panel of 9-12 assessors. For Na-solution DAP tests, a DAP score of greater than 3.1 indicates saltiness or salt enhancement. The DAP score can be correlated with a sodium reduction potential by subtracting 3.1 from the DAP score. For example, a DAP score of 4.0 would result in a 9% sodium reduction potential ((4.0-3.1)*10=9%), which means that 9% less sodium may be present in a food product having the salty compound relative to a substantially similar food product that does not have the salty compound while producing a similar saltiness. For broth DAP tests, a DAP score of greater than 7.6 indicates saltiness or salt enhancement. The DAP score can be correlated with a sodium reduction potential by subtracting 7.6 from the DAP score. For example, a DAP score of 8.5 would result in a 9% sodium reduction potential ((8.5-7.6)*10=9%), which means that 9% less sodium may be present in a food product having the salty compound relative to a substantially similar food product that does not have the salty compound while producing a similar saltiness.
[0702] A summary of the compounds, the channels for which in vitro thresholds were identified, and the DAP scores are provided below in Table 3.
TABLE-US-00004 TABLE 3 Activity of identified compounds in Na-Solution and in simple broth DAP in DAP in Compound Na-Sol. Broth 1 3.6 7.8 2 3.5 7.8 3 4.1 8.8 4 3.5 N.T. 5 3.6 N.T. 6 3.8 N.T. 7 3.7 N.T. 8 4 N.T. 9 3.8 N.T. 10 3.9 7.9 11 2.6 8.1 12 4 7.6 13 3.8 7.9 14 3.6 N.T. 15 3.5 8.1 16 3.3 8.0 17 3.6 8.2 18 3.8 8.1 19 N.T. N.T. 20 4 N.T. 21 4 N.T. 22 N.T. N.T. 23 2.9 8.3 24 N.T. N.T. 25 3.9 N.T. 26 3.4 N.T. 27 3.4 N.T. 28 3.6 N.T. 29 3.5 8.0 30 3.1 N.T. 31 3.2 N.T. 32 3.5 8.1 33 3.4 7.7 34 3.9 7.7 35 3.7 8.1 36 3.9 8.2 37 3.7 8.1 38 3.9 8.1 39 N.T. N.T. 40 3.5 7.8 41 3.9 7.5 42 3.1 8.1 43 3.7 8.0 44 3.8 8.6 45 3.5 8.2 46 3.6 7.8 47 N.T. N.T. 48 3.7 7.8 49 3.6 8.2 50 4 N.T. 51 N.T. N.T. 52 3.6 N.T. 53 3.7 8.5 54 3.4 8.1 55 N.T. N.T. 56 4 7.7 57 3.1 N.T. 58 3.3 8.0 59 3.3 7.9 60 3.8 8.1 61 3.6 N.T. 62 3.9 8.0 63 3.5 8.0 64 3.7 7.8 65 3.3 7.9 66 3.2 7.8 67 N.T. N.T. 68 N.T. N.T. 69 N.T. N.T. 70 N.T. N.T. 71 N.T. N.T. 72 N.T. N.T. 73 3.5 8.0 74 3.2 7.7 75 3.6 N.T. 76 3.2 8.0 77 3.4 8.1 78 N.T. N.T. 79 N.T. N.T. 80a 3.9 N.T. 80b 3.9 N.T. 81 3.2 7.8 82 3.8 8.3 83 4.2 8.1 84 3.8 8.1 85 2.9 N.T. 86 3.9 8.0 87 3.9 8.0 88 3.2 8.0 89 3.7 7.6 90 N.T. N.T. 91 3.7 N.T. 92 4 N.T. 93 N.T. N.T. 94 4.2 N.T. 95 N.T. N.T. 96 N.T. N.T. 97 N.T. N.T. 98 N.T. N.T. 99 4.1 N.T.
[0703] In Table 3 above, "N.T.," with regard to a DAP score, means the compound was not taste tested.
[0704] The data in Table 3 above reflect the best DAP score for the tested concentrations of 0.1 parts per million (ppm), 1 ppm and 10 ppm. The highest concentrations did not always result in the highest DAP scores.
[0705] Compounds in water with no sodium were also tested. The compounds in water with no sodium elicited no appreciably discernable salty taste, even at the 10 ppm concentration (data not shown).
[0706] DAP score testing results for various pairs of compounds are presented in FIG. 1 (sodium solution) and FIG. 2 (broth). Certain combinations were tested twice. For these combinations, two DAP scores are shown in the tables presented in FIGS. 1-2. As shown in the results presented in FIGS. 1-2, certain combinations of compounds can enhance the perception of saltiness. Some combinations resulted in DAP scores as high as 4.5 in some tests. See, for example, the combination of compound 83 and 13 in FIG. 1 (sodium solution) and the combination of compound 12 and 18 in FIG. 2 (broth). Such DAP scores may result in a sodium reduction potential of about 14%. The combinations tested in FIGS. 1-2 are representative of the combinations that may be used in a food product to enhance the perception of saltiness or reduce sodium content. It will be understood that any other suitable combination of compounds may be employed.
[0707] Additional testing of combinations of pairs of compounds was performed in sodium solution. The DAP scores from this additional testing is shown below in Table 4.
TABLE-US-00005 TABLE 4 Activity of a combination of bioactive compounds in sodium solution Compound 66: Compound 29: Compound 16: Compound 33: Compound 73: (0.1 ppm) (10 ppm) (1 ppm) (1 ppm) (1 ppm) Compound 83: (1 ppm) 3.7 4.0 3.8 3.6 4.0 Compound 10: (1 ppm) 3.3 3.7 3.8 2.9 3.6 Compound 45: (10 ppm) 3.2 3.3 3.8 3.7 4.2
[0708] In addition, more than two bioactive, taste modulating or salty taste modulating compounds described herein may be included in a food product. By way of example, Table 5 below shows DAP scores obtained from testing sodium solutions and chicken broth containing a combination of compounds 12, 13 and 83. As shown in Table 5, such a combination resulted in a DAP score of 5.3 when tested in a sodium solution. Accordingly, such a combination may result in a sodium reduction potential of about 22%. Of course, other suitable combinations of three or more compounds may be used or included in a food product to enhance the perception of saltiness or to reduce sodium content.
TABLE-US-00006 TABLE 5 Activity of a combination of bioactive compounds in sodium solution and broth In Na In Compound Conc. Solution Broth 12 1 ppm 5.3 8.1 13 10 ppm 83 1 ppm
[0709] Some illustrative examples of combinations of compounds that may produce desired or beneficial effect, for example when incorporated in a food product, include combinations that include at least one compound selected from the group consisting of compounds 3, 10, 12, 13, 16, 18, 29, 33, 36, 37, 41, 43, 44, 45, 48, 53, 56, 62, 66, 73, 82, 83, and 84. Another illustrative example is a combination that includes at least one compound selected from the group consisting of compounds 10, 12, 13, 18, 36, 45, 56, 82, and 83. Yet another illustrative example is a combination that includes compounds 12, 13 and 83. Of course, any other suitable or desirable combination may be used.
[0710] DAP scores for combinations of three different compounds in broth are shown in Table 6 below.
TABLE-US-00007 TABLE 6 Activity of a combination of bioactive compounds in broth Compound (concentration) DAP score 3 (0.1 ppm) 7.8 36 (0.1 ppm) 44 (10 ppm) 3 (0.1 ppm) 7.7 36 (0.1 ppm) 53 (1 ppm) 3 (0.1 ppm) 7.9 36 (0.1 ppm) 18 (10 ppm) 13 (10 ppm) 8.1 84 (1 ppm) 44 (10 ppm) 13 (10 ppm) 7.9 84 (1 ppm) 53 (1 ppm) 13 (10 ppm) 8.0 84 (1 ppm) 3 (0.1 ppm) 18 (10 ppm) 8.1 12 (1 ppm) 44 (10 ppm) 18 (10 ppm) 8.2 12 (1 ppm) 53 (1 ppm) 18 (10 ppm) 8.3 12 (1 ppm) 3 (0.1 ppm)
[0711] Sourcing
[0712] Natural sources of the mentioned taste modulating or salty taste modulating compounds can be extracted by a variety of methods such as, but not exclusive to, water, solvent extractions (ethanol/water combinations), or supercritical carbon dioxide or other volatilization methods. These concentrated extracts or isolates could be stabilized physically by encapsulation, for example, or chemical reaction to non-reactive compounds such as simple sugars or small chain fatty acids. Compounds may be altered for their solubility in aqueous solutions by hybridization to larger sized molecules and additionally processed or reacted to create an impacting ingredient in either a dry or aqueous form.
[0713] In embodiments, a composition comprises a bioactive, taste modulating or salty taste modulating compound described herein. The composition may be included in a food product. In embodiments, the composition comprises one or more natural extracts. In another embodiment, the extract is selected from a plant or microbial (e.g., fungi or bacterial) source. Examples of suitable natural extracts include extracts derived from Aesculus hippocastaneum; Alchemilla xanthochlora; Angelica archangelica; Apocynum cannabinum; Azadirachta indica; Actinomycete bacteria (Strain code: 01702axxx000002); Capsicum annuum; Cimicifuga racemosa; Commiphora mukul; Embelia ribes; Evodia rutaecarpa; Ferula assa-foetida; Fungi (Strain code: 02295fxxx000001; Strain code: 01469fxxx000005); Gleditschia australis; Kaempferia galanga; Lavandula officinalis; Marrubium vulgare; Mesua ferrea; Nephelium cuspidatum; Orthosiphon stamineus; Persea gratissima; Petroselinum stativum; Piper longum; Pithecoctenium echinatum; Podophyllum peltatum; Psidium guajava; Ricinus communis; Salvia miltiorrhiza; Schisandea chinensis; Teclea trichocarpa; Vitex agnus; Xysmalobium undulatum; Yucca gloriosa; Zanthoxylum piperitum; Zingiber officinalis and others. The composition may be in a dry or liquid form. The liquid composition may be a solution, suspension, colloidal suspension, microencapsulated suspension, emulsion, or the like, or combinations thereof. The dry composition may be a microencapsulation solid, agglomeration, or the like or combinations thereof.
[0714] In embodiments, a bioactive, taste modulating or salty taste modulating compound described herein is included in a composition comprising a carrier. The composition comprising the carrier may be incorporated into a food product. Any suitable carrier may be used. Examples of suitable carriers include propylene glycol, ethanol, water, or oil. In embodiments, the carrier is a starch, such as a starch comprising carbohydrate, a maltodextrin, a cyclodextrin or another dextrin, or a liposome. In embodiments, the carrier is an encaspulant or the carrier may comprise an embedded bioactive, taste modulating or salty taste modulating compound.
Definitions
[0715] All scientific and technical terms used herein have meanings commonly used in the art unless otherwise specified. The definitions provided herein are to facilitate understanding of certain terms used frequently herein and are not meant to limit the scope of the present disclosure.
[0716] As used in this specification and the appended claims, the singular forms "a", "an", and "the" encompass embodiments having plural referents, unless the content clearly dictates otherwise.
[0717] As used in this specification and the appended claims, the term "or" is generally employed in its sense including "and/or" unless the content clearly dictates otherwise. The term "and/or" means one or all of the listed elements or a combination of any two or more of the listed elements.
[0718] As used herein, "have", "having", "include", "including", "comprise", "comprising" or the like are used in their open ended sense, and generally mean "including, but not limited to". It will be understood that "consisting essentially of", "consisting of", and the like are subsumed in "comprising" and the like. As used herein, "consisting essentially of," as it relates to an composition, product, method or the like, means that the components of the composition, product, method or the like are limited to the enumerated components and any other components that do not materially affect the basic and novel characteristic(s) of the composition, product, method or the like.
[0719] The words "preferred" and "preferably" refer to embodiments of the invention that may afford certain benefits, under certain circumstances. However, other embodiments may also be preferred, under the same or other circumstances. Furthermore, the recitation of one or more preferred embodiments does not imply that other embodiments are not useful, and is not intended to exclude other embodiments from the scope of the disclosure, including the claims.
[0720] Also herein, the recitations of numerical ranges by endpoints include all numbers subsumed within that range (e.g., 1 to 5 includes 1, 1.5, 2, 2.75, 3, 3.80, 4, 5, etc. or 10 or less includes 10, 9.4, 7.6, 5, 4.3, 2.9, 1.62, 0.3, etc.). Where a range of values is "up to" a particular value, that value is included within the range.
[0721] As used herein, a "bioactive compound" is a compound that alters the flow of ions through one or more channels associated with the perception of salty taste or another taste associated with consumption of sodium chloride.
[0722] As used herein, a "taste modulating compound" is a compound that modifies the taste of a food product. By way of example, a taste modulating compound may modify the taste of a food product due to a particular taste imparted by the taste modulating compound, due to a modification of the perceived taste of the food product, or a component thereof, or the like. In embodiments, a taste modulating compound is a salty taste modulating compound.
[0723] As used herein a "salty taste modulating compound" is a compound that, when ingested, elicits or enhances a perception of salty taste alone or in the presence of a salt, such as sodium chloride.
[0724] As used herein, a composition that is "substantially similar" to another composition contains substantially the same concentration of components (e.g., within about 5%) except for the specifically enumerated components that make the compositions different. For example, a composition that includes a salty compound may be substantially similar to a composition that does not have the salty compound, if the components of the compositions, other than the salt and the salty compound, are present in a substantially similar concentration.
[0725] As used herein, a compound "derived" from a natural product is a compound that exists in a natural product, whose identity is verified. The compound derived from the natural product may be extracted from, for example, a plant or microbial source as opposed to being produced synthetically. Extraction or isolation of the naturally-derived compound may be facilitated by simple chemical reactions such as acidification, basification, ion exchange, hydrolysis, and salt formation as well as microbial fermentation, and the like. In embodiments, a taste modulating or salty taste modulating compound is derived from natural sources such as natural plant, fungi, and bacterial sources. Examples of such natural sources include, but are not limited to Aesculus hippocastaneum; Alchemilla xanthochlora; Angelica archangelica; Apocynum cannabinum; Azadirachta indica; Actinomycete bacteria (Strain code: 01702axxx000002); Capsicum annuum; Cimicifuga racemosa; Commiphora mukul; Embelia ribes; Evodia rutaecarpa; Ferula assa-foetida; Fungi (Strain code: 02295fxxx000001; Strain code: 01469fxxx000005); Gleditschia australis; Kaempferia galanga; Lavandula officinalis; Marrubium vulgare; Mesua ferrea; Nephelium cuspidatum; Orthosiphon stamineus; Persea gratissima; Petroselinum stativum; Piper longum; Pithecoctenium echinatum; Podophyllum peltatum; Psidium guajava; Ricinus communis; Salvia miltiorrhiza; Schisandea chinensis; Teclea trichocarpa; Vitex agnus; Xysmalobium undulatum; Yucca gloriosa; Zanthoxylum piperitum; Zingiber officinalis; and others. In embodiments, one or more compounds derived from Persea gratissima are combined with one or more compounds derived from Kaempferia galanga or one or more compounds derived from Capsicum annuum; and others.
[0726] As used herein, an "isolated" or "purified" compound is a compound that is substantially separated from other components of the source of the compound. For example, if the source of the compound is a natural product, an isolated or purified compound may be a compound that is separated from its naturally occurring environment. If the compound is synthesized, the compound may be separated from unreacted reagents, reaction byproducts, solvents, or the like.
[0727] As used herein a "synthetic compound" is a compound that is synthesized via chemical reaction in vitro. A compound that is "synthesized" is a synthetic compound. A synthesized compound may be identical to a compound derived from a natural product.
[0728] For the purposes of this disclosure, reference to a compound includes reference to salts of the compound, hydrates of the compound, polymorphs of the compound, isomers of the compound (including constitutional isomers and stereoisomers such as enantiomers and diasteriomers), and the like.
[0729] For the purposes of the present disclosure, it will be understood that a ring structure having a structure of
##STR00190##
or the like, will be considered to be aromatic.
[0730] It will be understood that compounds disclosed herein may be glycosylated or may be substituted with one or more saccharides. In various embodiments, specific or generic compounds substituted with one or more saccharides are disclosed. However, it will be understood that other saccharide substitutions are possible and are contemplated.
[0731] As used herein, a "saccharide" is a monosaccharide or an oligosaccharide. A monosaccharide may be a diose, a triose, a tetrose, a pentose, a hexose, a heptose, and so one. Monosacharides include aldoses and ketoses. Examples of monosaccharides include glyceraldehyde, dihydroxyacetone, erythrose, threose, erythrulose, arabinose, lyxose, ribose, xylose, ribulose, xylulose, allose, altrose, galactose, glucose, gulose, idose, nannose, talose, fructose, psicose, sorbose, tagatose, mannoheptulose, sedoheptulose, 2-keto-3-deoxy-manno-actonate, and sialose. Monosaccharides may be acyclic or cyclic. Cyclic isomers include furanoses and pyranoses.
[0732] As used herein, "monosaccharide" includes deoxygenated variants of monosaccharides that are deoxygenated at one or more positions. As used herein, "monosaccharide" includes monosaccharides having carbon atoms of a monosaccharide chain or ring that are substituted with one or more of the following: H(H), CH.sub.2OH, OH, COOH, OCOR.sup.100, CH.sub.3, OCH.sub.3, C(CH.sub.3).sub.2OH, and
##STR00191##
where R.sup.100 is selected from the group consisting of
##STR00192##
[0733] An "oligosaccharide" is a chain of two or more monosaccharides where each monosaccharide is bound by a glycosidic bond.
[0734] As used herein, "saccharidyl" means a monosaccharide or oligosaccharide substituent. The monosaccharide or oligosaccharide substituent may be a terminal substituent or an internal substituent. That is, a saccharidyl group may be bound to one or more parent compounds (e.g., parent structure 1-saccharidyl or parent structure 1-saccharidyl-parent structure 2).
[0735] For purposes of example, a generic structure representing a monosaccharide is presented below:
##STR00193##
where one of A', B', D', E', F' and G' is O and where each of the rest of A', B', D', E', F' and G' is independently selected from the group consisting of CH.sub.2, CHOH, CHCH.sub.2OH, CHCH.sub.3, CHCOOH, CHOCOR.sup.101, CHOCH.sub.3, CHC(CH.sub.3).sub.2OH, and
##STR00194##
where R.sup.101 is selected from the group consisting of
##STR00195##
[0736] For purposes of Example, a generic structure representing an embodiment of an oligosaccharide that is a disaccharide is presented below:
##STR00196##
(i) where (a) one of A', B', D', E', and F' is O, (b) one of A'', B'', D'', E'', and F'' is O, and (c) each of the rest of A', B', D', E', F', A'', B'', D'', E'', and F'' is independently selected from the group consisting of CH.sub.2, CHOH, CHCH.sub.2OH, CHCH.sub.3, CHCOOH, CHOCOR.sup.102, CHOCH.sub.3, CHC(CH.sub.3).sub.2OH, and
##STR00197##
where R.sup.102 is selected from the group consisting of
##STR00198##
and (ii) where x' and y' are independently selected from zero or 1. Typically, the oxygen heteroatom of a monosaccharide ring will be in an ortho or meta position relative to a saccharide substitution.
[0737] For purposes of example, a generic structure representing an embodiment of an oligosaccharide that is a trisaccharide is presented below:
##STR00199##
[0738] (i) where (a) one of A''', B''', C''', E''', and F''' is O, (b) one of A'', B'', D'', E'', and F'' is O, and (c) each of the rest of A', B', D', A'', B'', D'', E'', F'', A''', B''', D''', E''', and F''' is independently selected from the group consisting of CH.sub.2, CHOH, CHCH.sub.2OH, CHCH.sub.3, CHCOOH, CHOCOR.sup.103, CHOCH.sub.3, CHC(CH.sub.3).sub.2O H, and
##STR00200##
where R.sup.103 is selected from the group consisting of
##STR00201##
and (ii) where f', g', x' and y' are independently selected from zero or 1. In the structure depicted above, the oxygen heteroatom of a monosaccharide ring is in an ortho position relative to the other saccharide substitutions. Of course, trisaccharides (or tetra-, penta-, etc-saccharides) where the where substitutions of the other saccharides are ortho/meta, ortho/para, meta/para or meta/meta relative to the oxygen of the central ring are also possible. Typically, trisaccharides (or tetra-, penta-, etc-saccharides) will be ortho/ortho, meta/meta, or ortho/meta regarding the saccharide substitutions relative to the oxygen heteroatom of a central ring.
[0739] For purposes of example, a generic structure representing an embodiment of an oligosaccharide that is a tetrasaccharide is presented below:
##STR00202##
[0740] (i) where (a) one of A''', B''', D''', E''', and F''' is O, (b) one of A''', B''', D''', E''', and F''' is O, (c) one of A'', B'', D'', E'', and F'' is O, and (d) each of the rest of A', B', A'', B'', D'', E'', F'', A''', B''', D''', E''', F''', A'''', B'''', D'''', E'''', and F'''' is independently selected from the group consisting of CH.sub.2, CHOH, CHCH.sub.2OH, CHCH.sub.3, CHCOOH, CHOCOR.sup.104, CHOCH.sub.3, CHC(CH.sub.3).sub.2OH, and
##STR00203##
where R.sup.104 is selected from the group consisting of
##STR00204##
and (ii) where f', g', p', q', x' and y' are independently selected from zero or 1. In the structure depicted above, the oxygen heteroatom of a monosaccharide ring is in an ortho position relative to the other saccharide substitutions. The example of a tetrasaccharide depicted below is a branched chain saccharide. However, tetrasaccharides (or penta-, hexa, etc-saccharides) may have linear chains.
[0741] It will be understood that the structures of the saccharides presented above are examples of saccharides and that other saccharides are contemplated herein. Any compound described herein may be optionally saccharidyl substituted at any suitable position. Examples of some saccharidyl substituents are presented herein below (e.g., with regard to compounds 34-39, 41, 48, 51, 76, 77, and 90-92). However, it will be understood that similar compounds with different saccharide substitutions are contemplated herein.
INCORPORATION BY REFERENCE
[0742] Any patent or non-patent literature cited herein is hereby incorporated herein by reference in its entirety to the extent that it does not conflict with the disclosure presented herein.
[0743] In the detailed description above several specific embodiments of compounds, compositions, products and methods are disclosed. It is to be understood that other embodiments are contemplated and may be made without departing from the scope or spirit of the present disclosure. The detailed description, therefore, is not to be taken in a limiting sense.
* * * * *
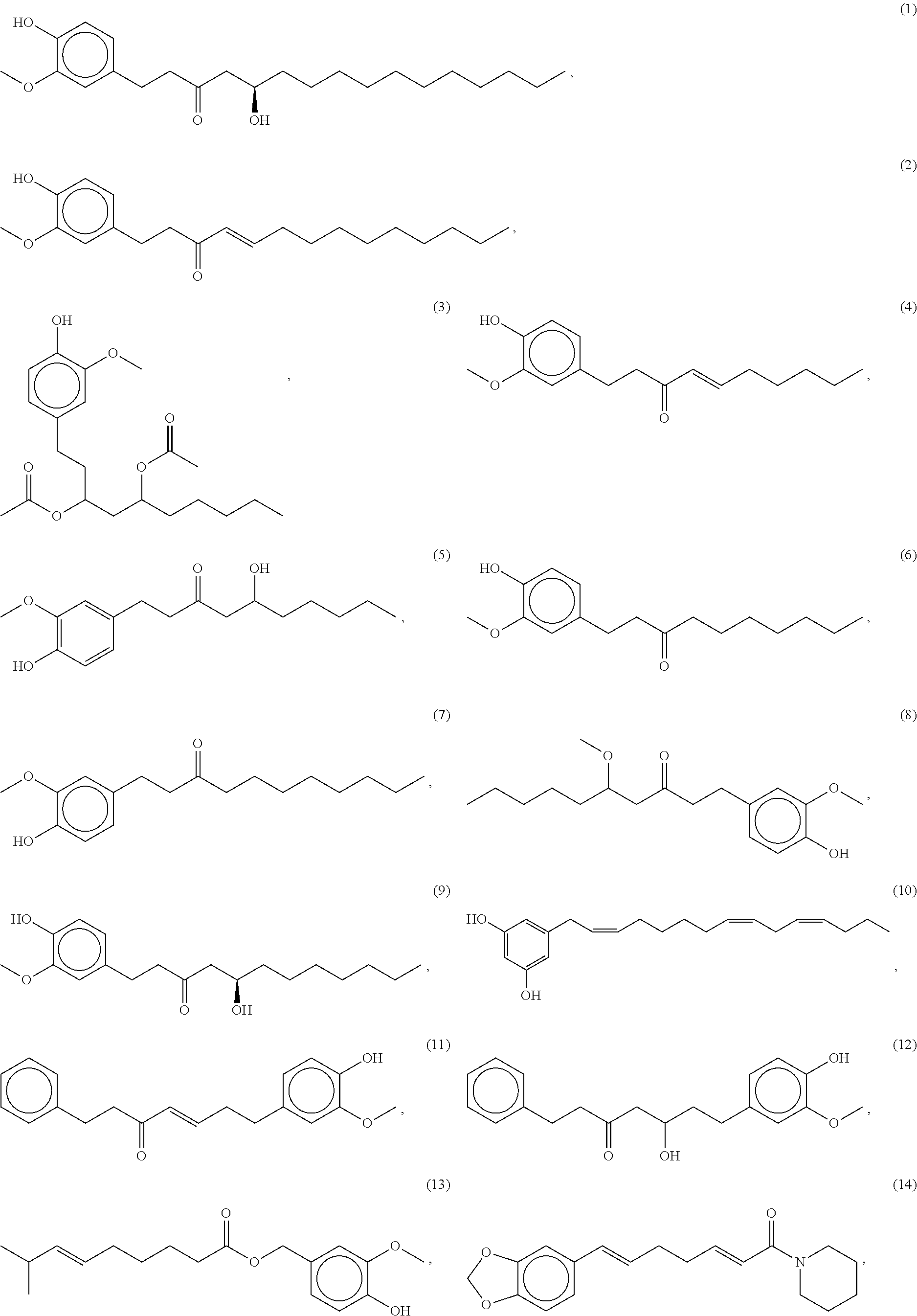







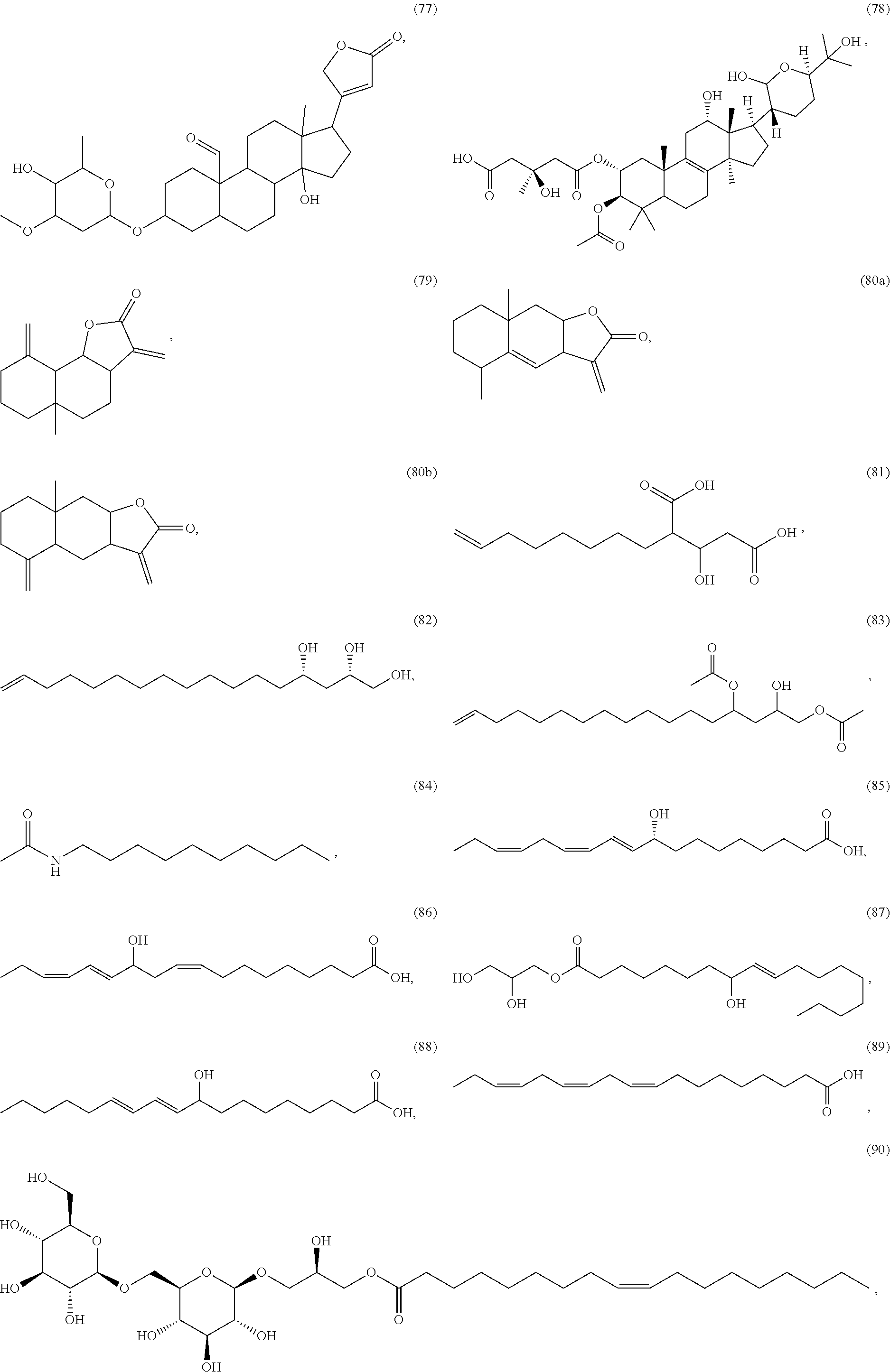
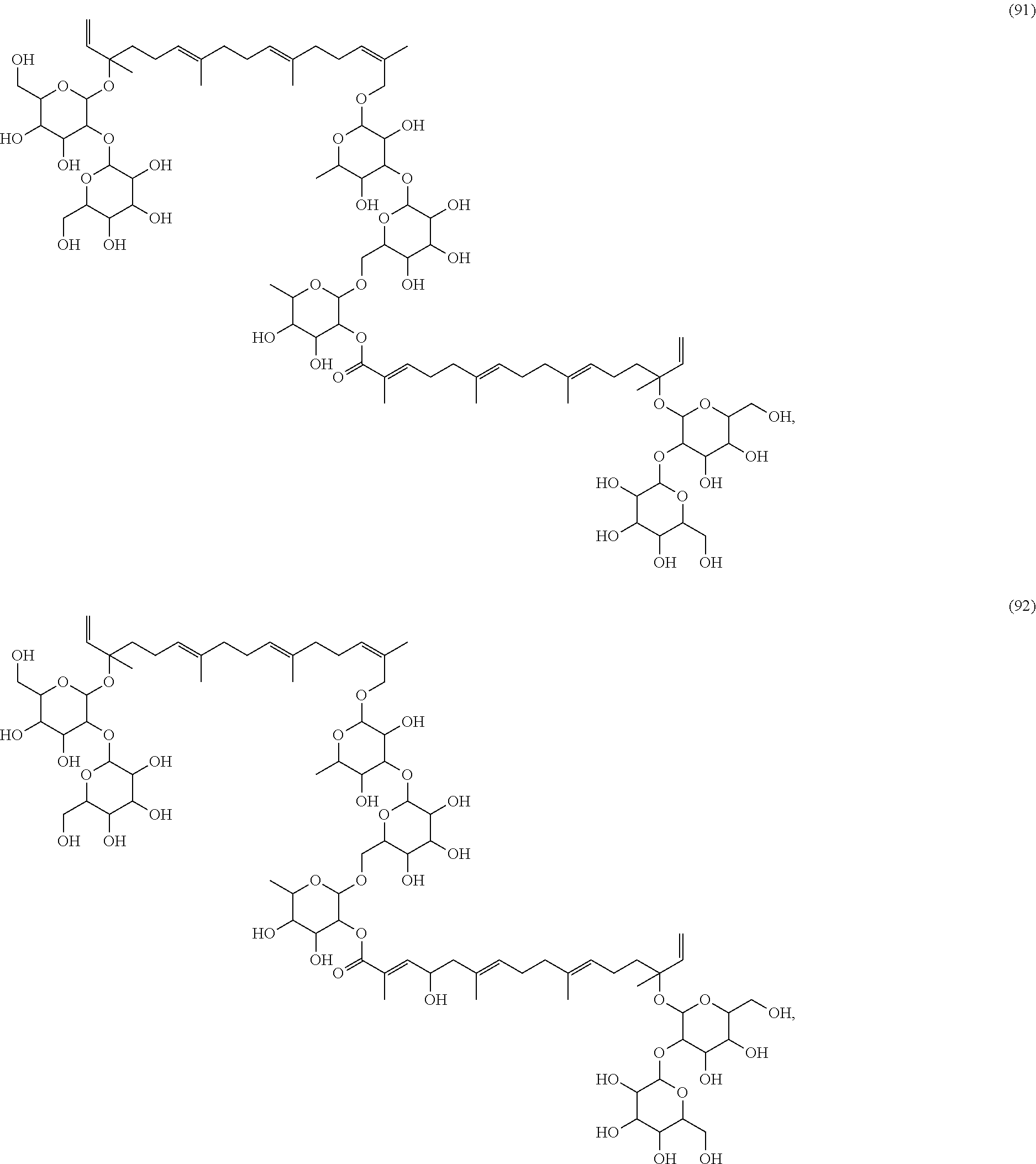


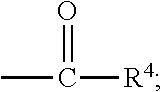
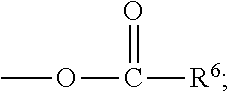
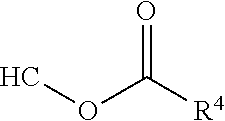

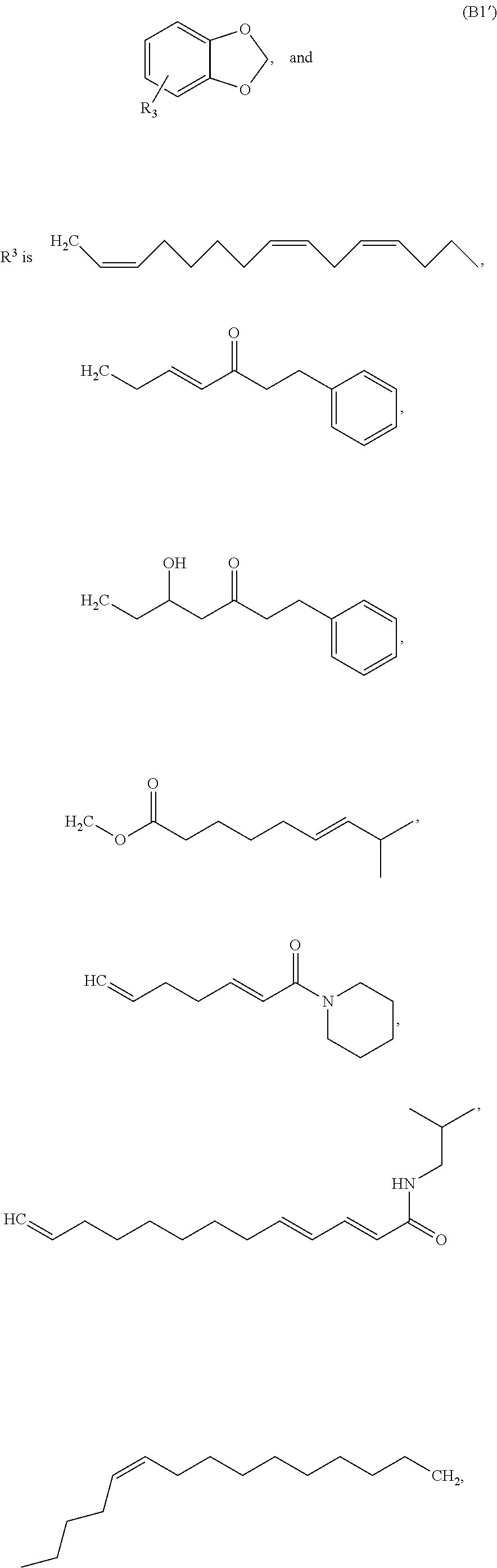
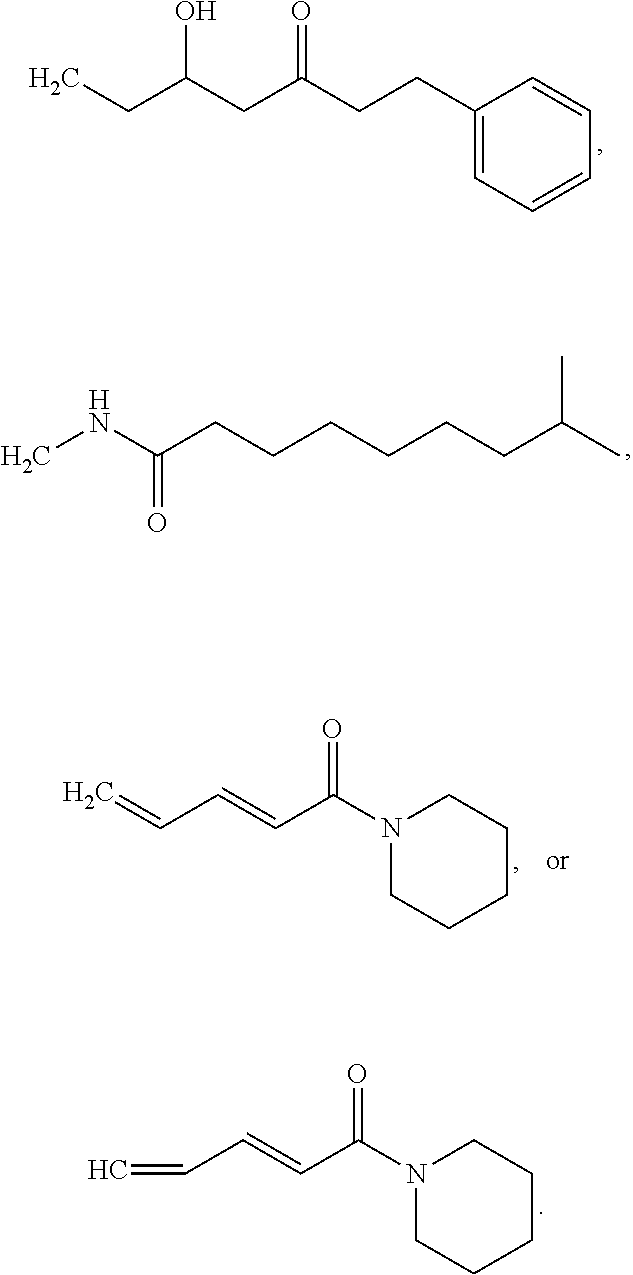
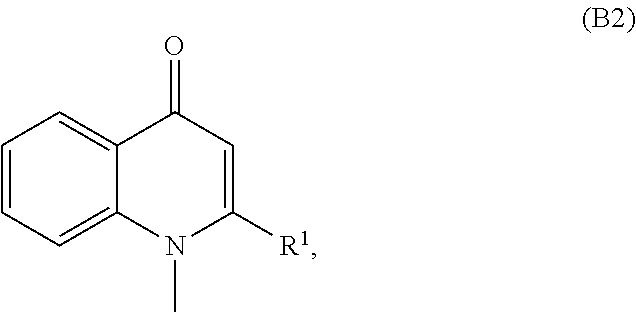
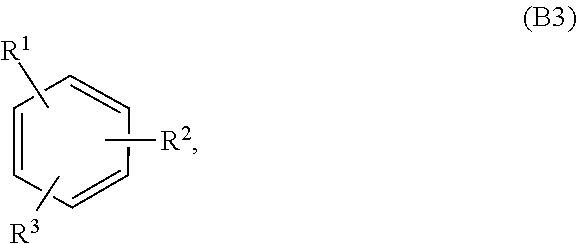
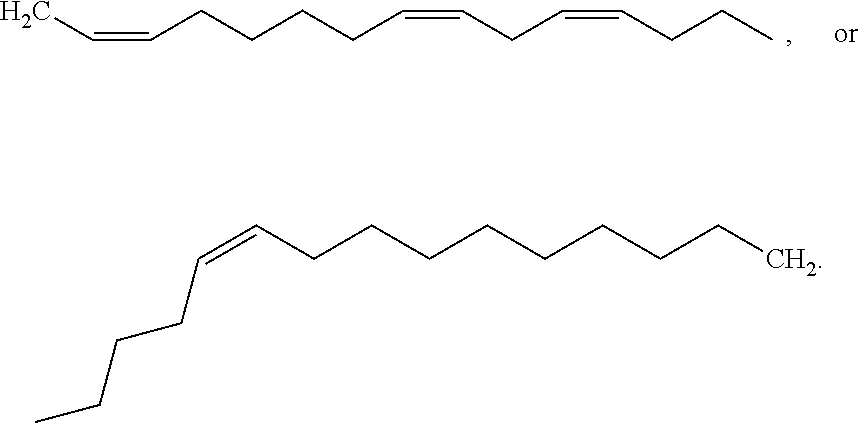
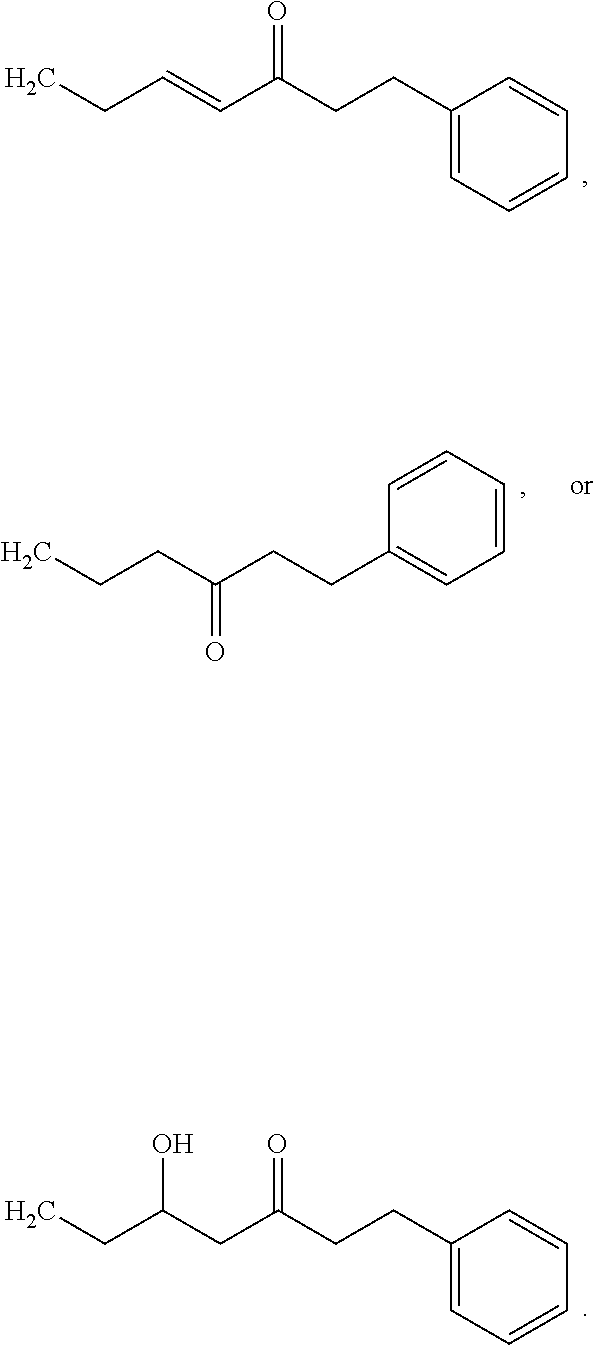

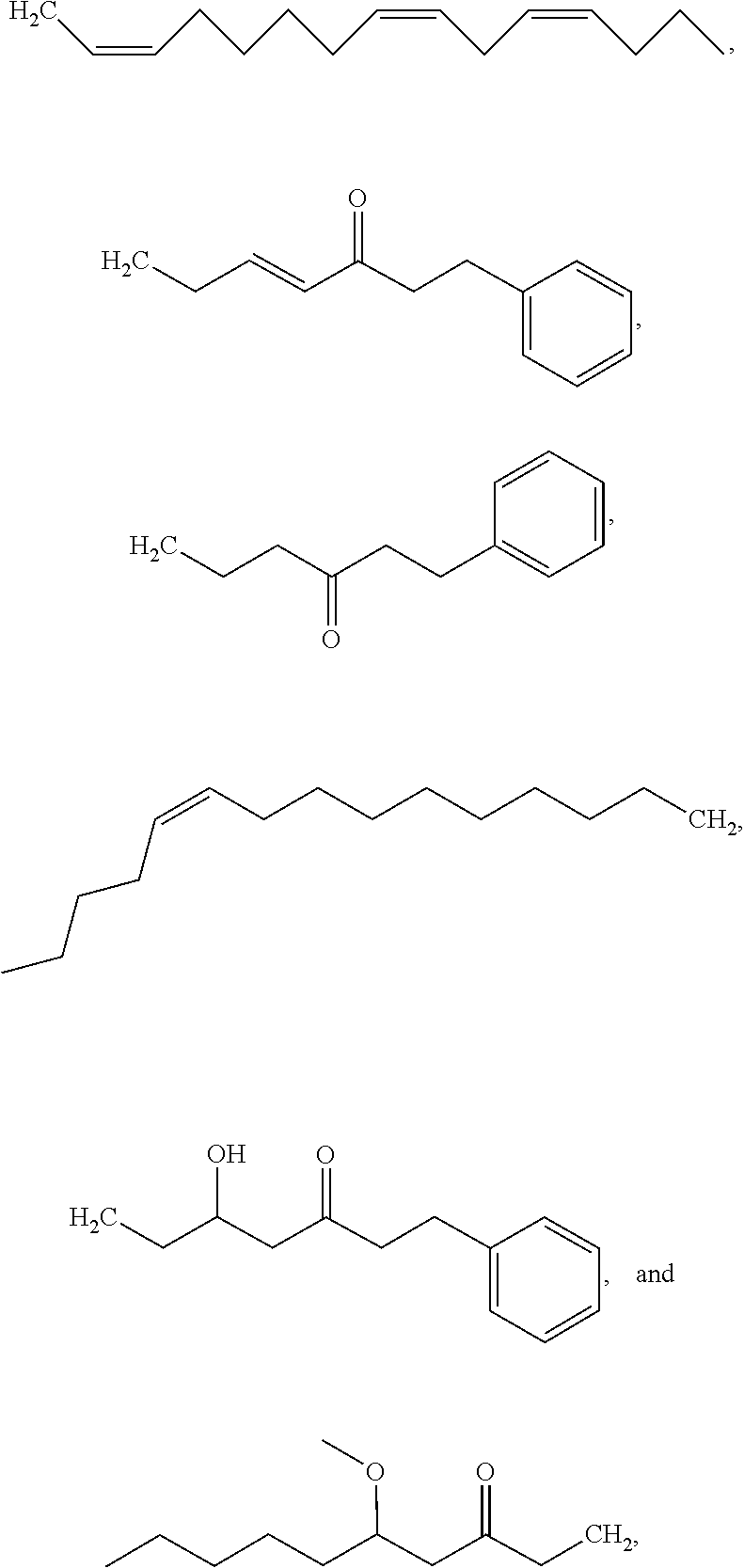
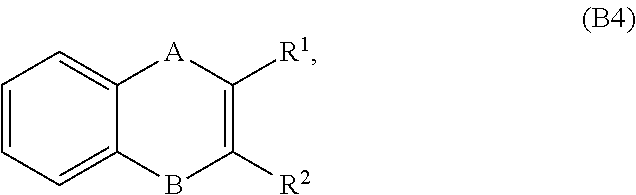
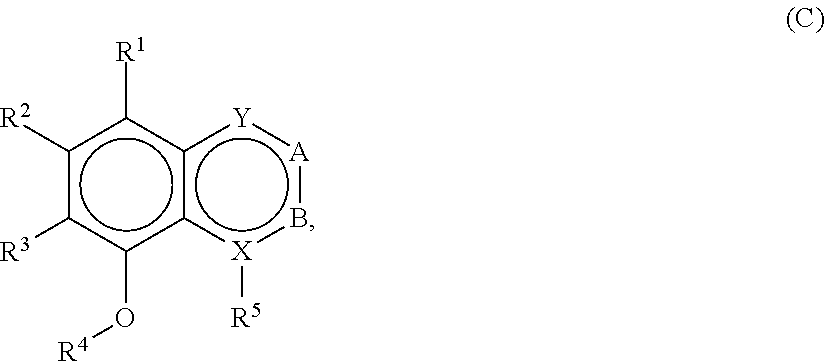
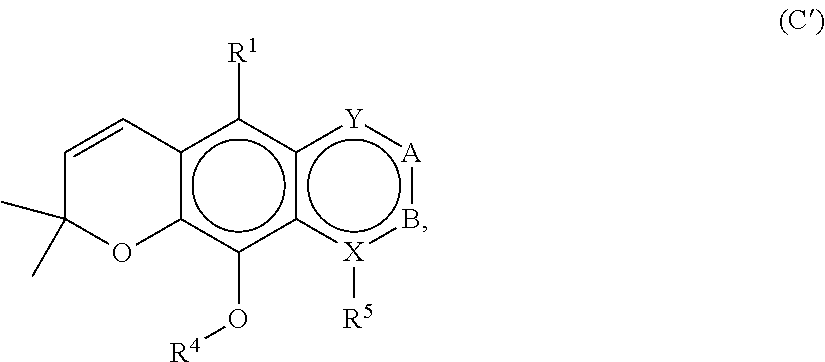
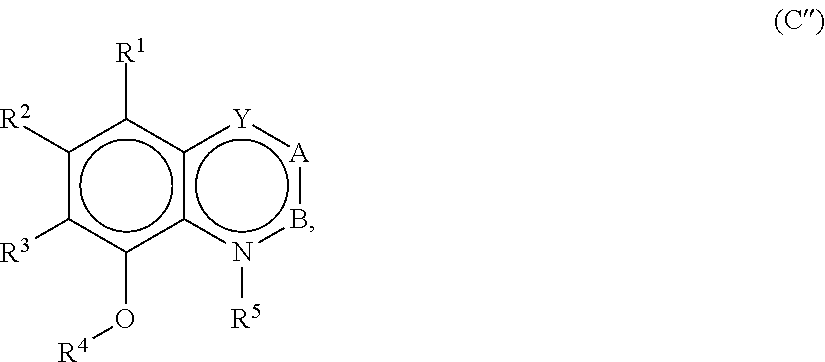
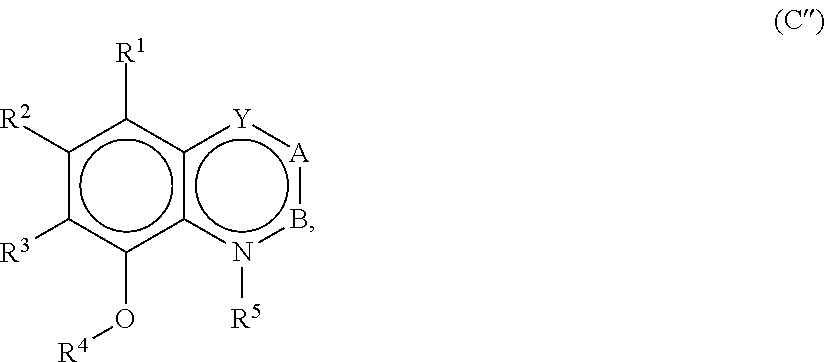
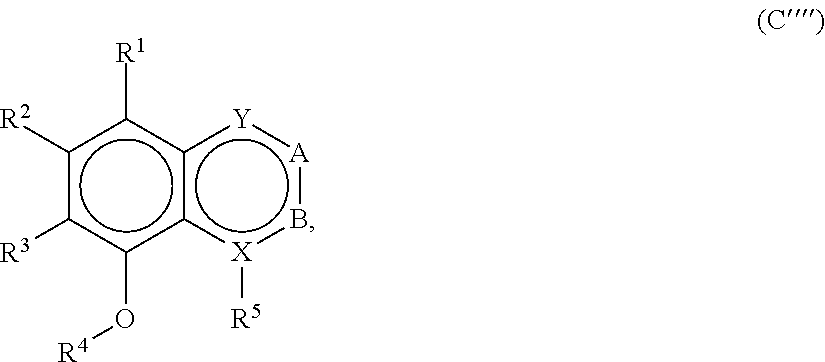
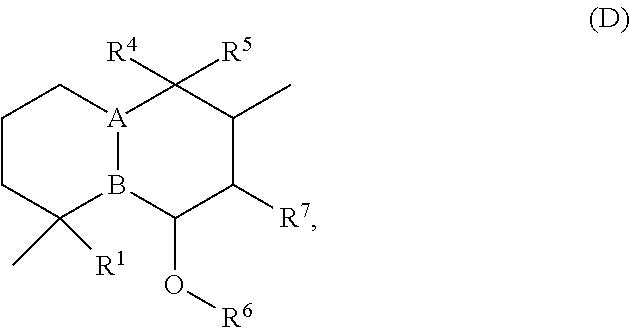
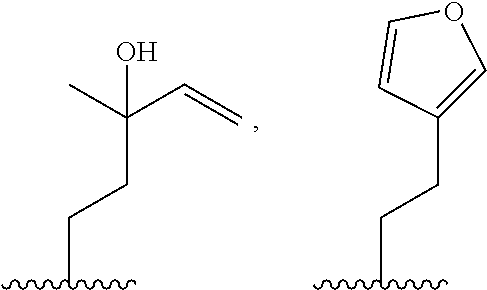
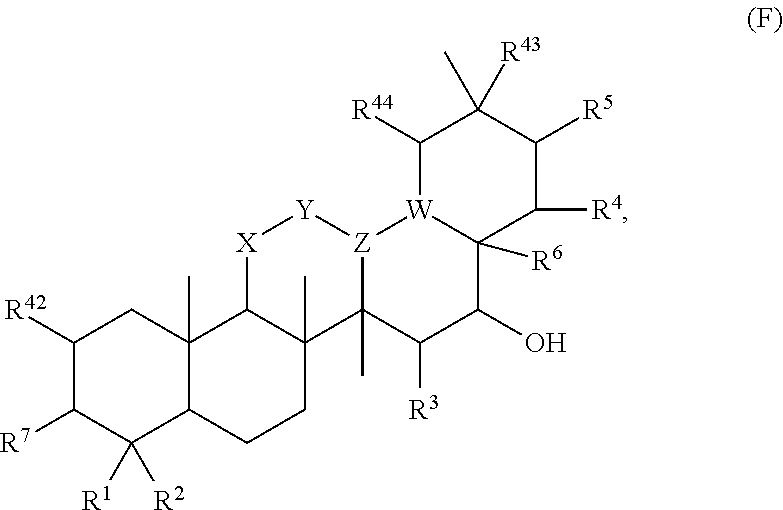
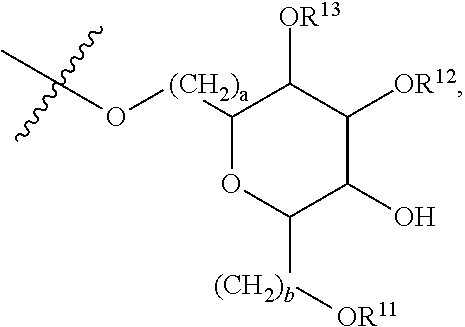
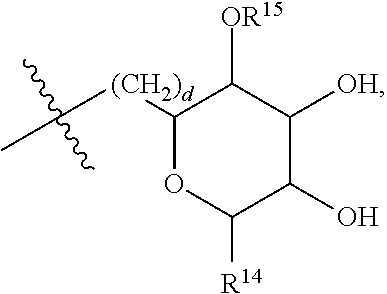
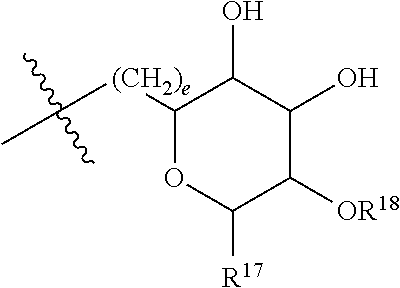
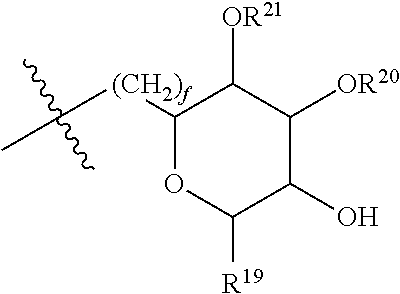
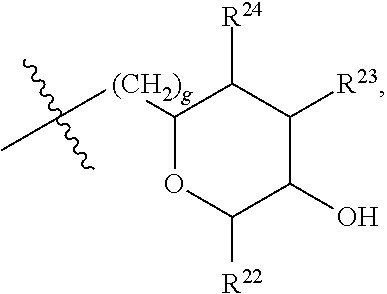
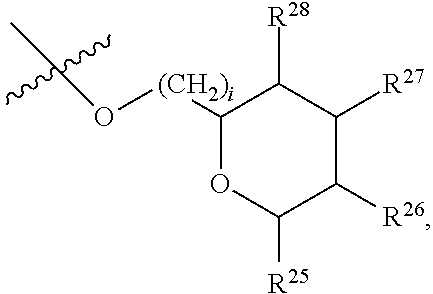
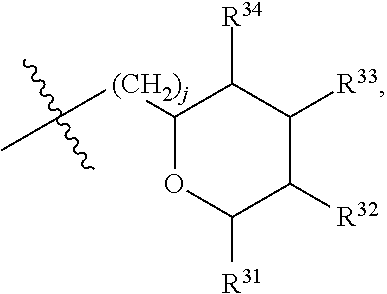
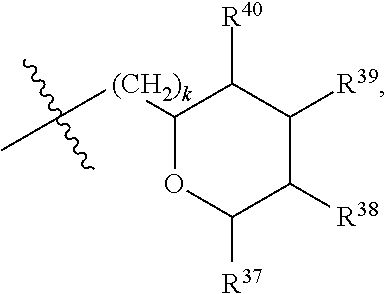
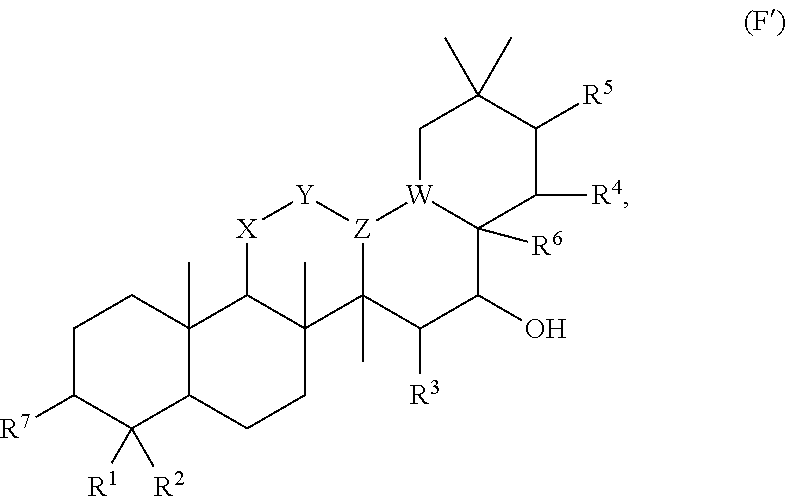
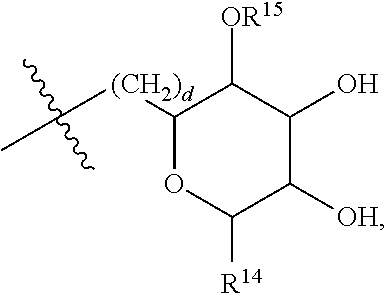
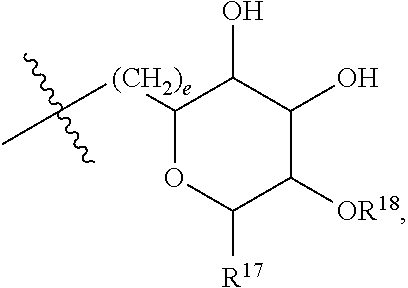
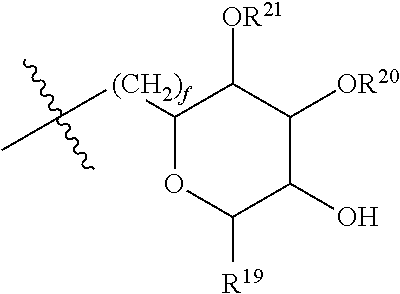

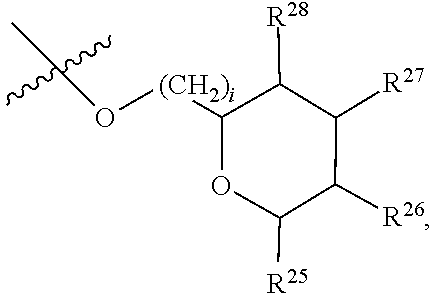
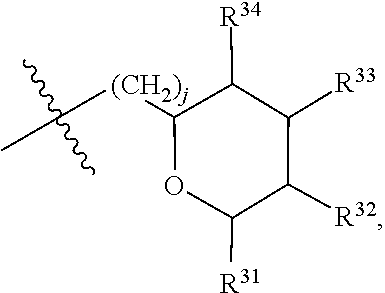
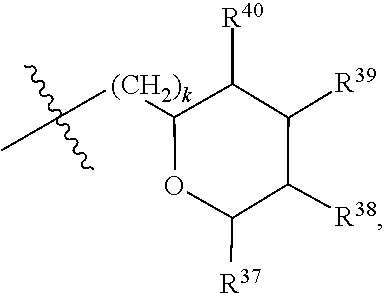
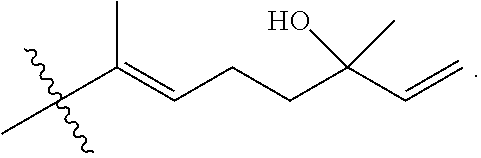
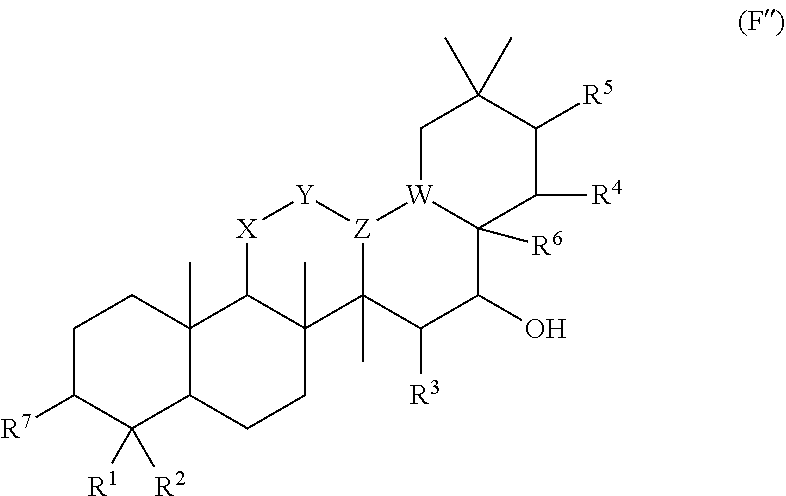
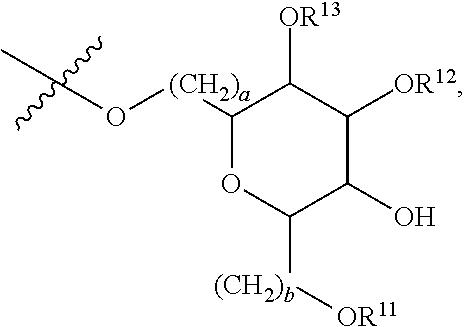
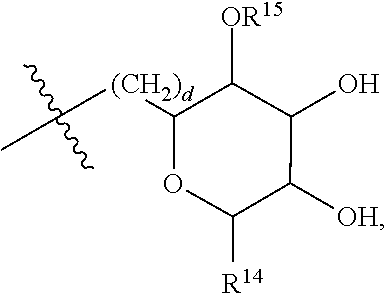
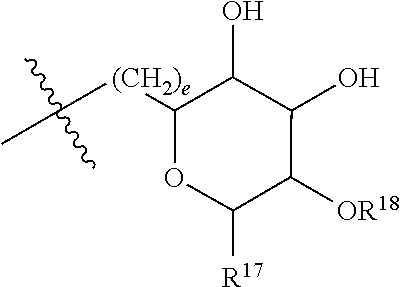
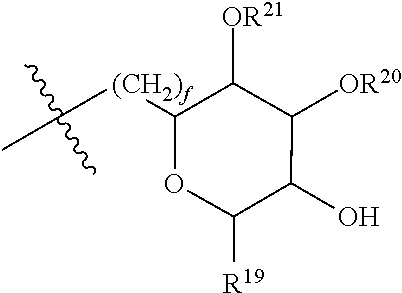
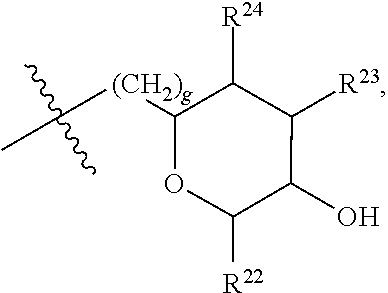
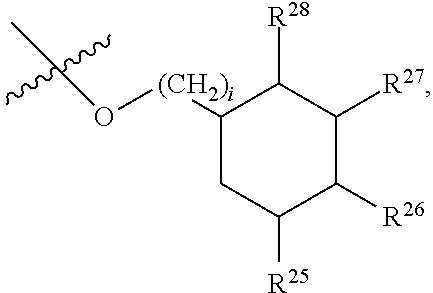
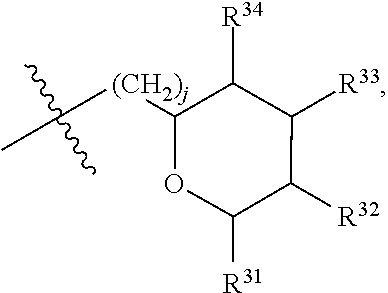
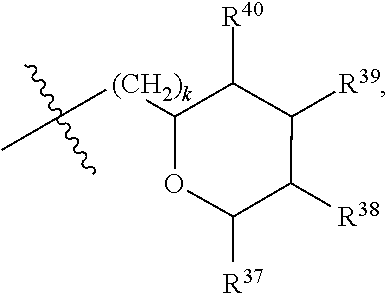
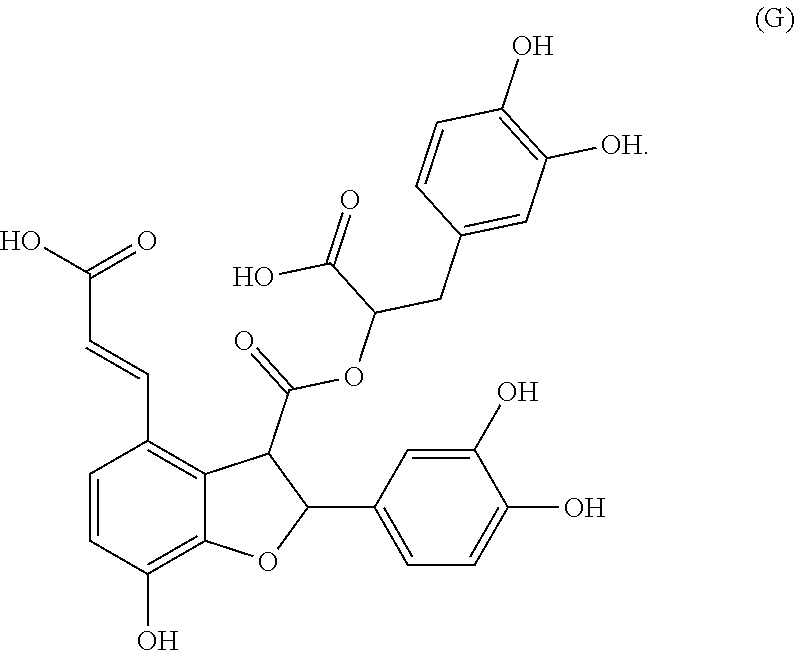
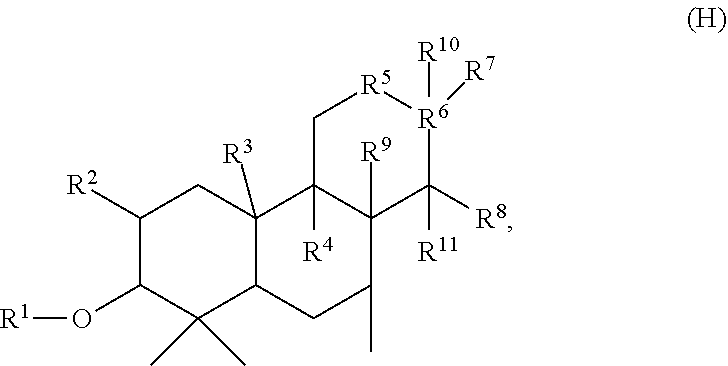
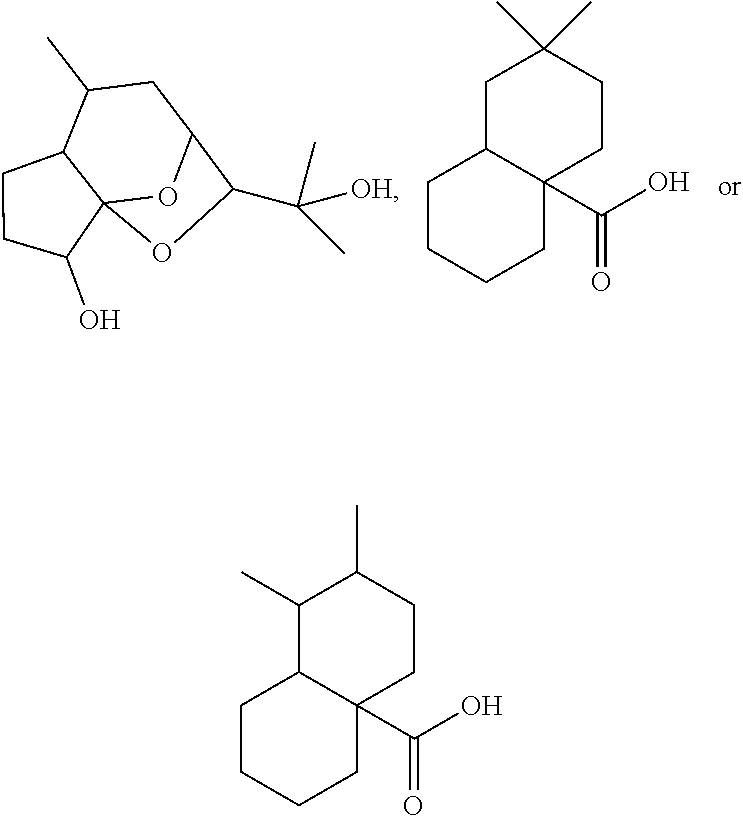
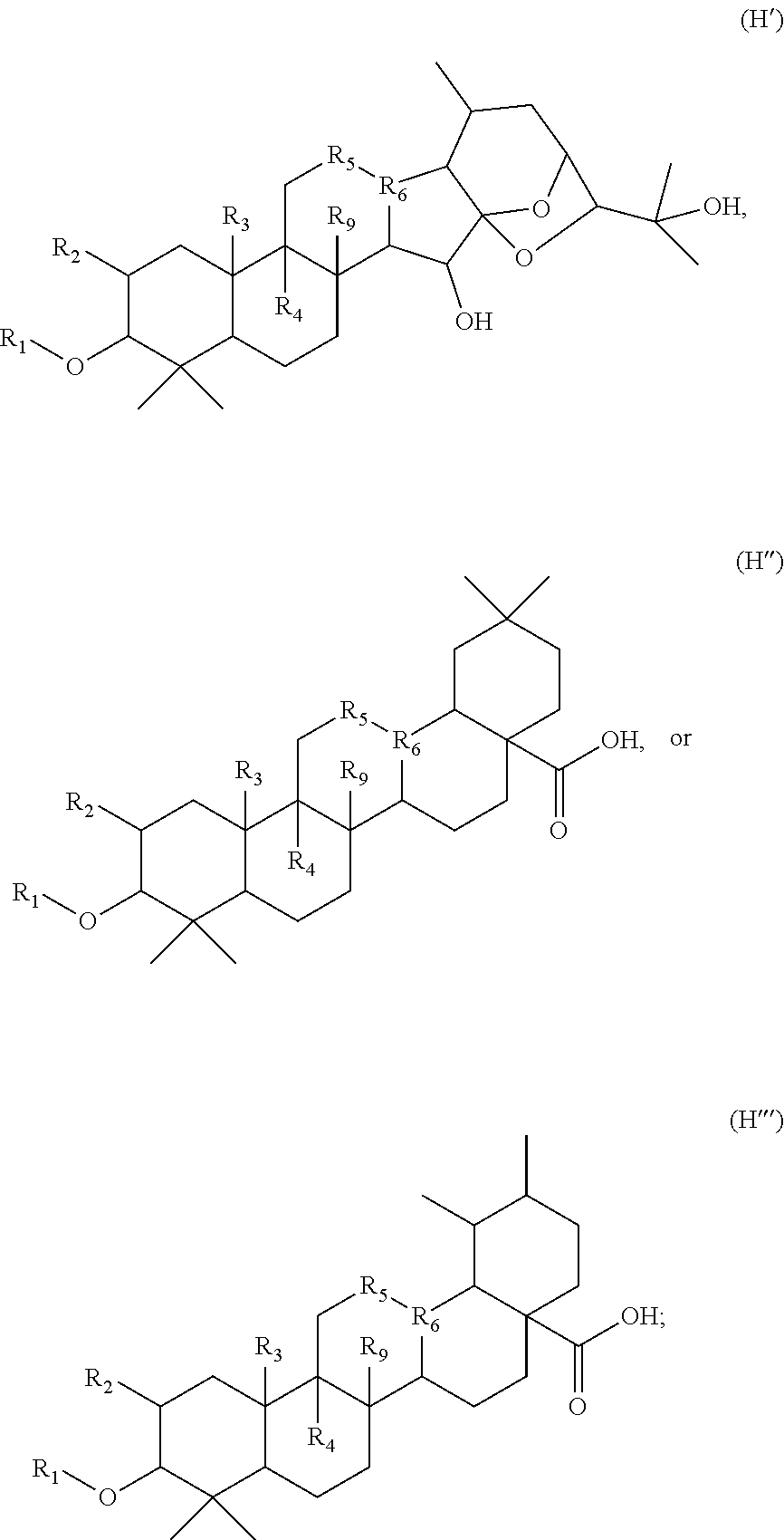
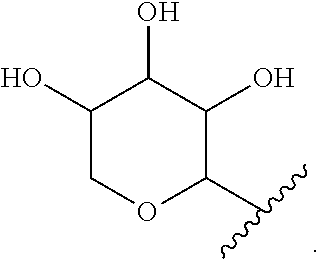
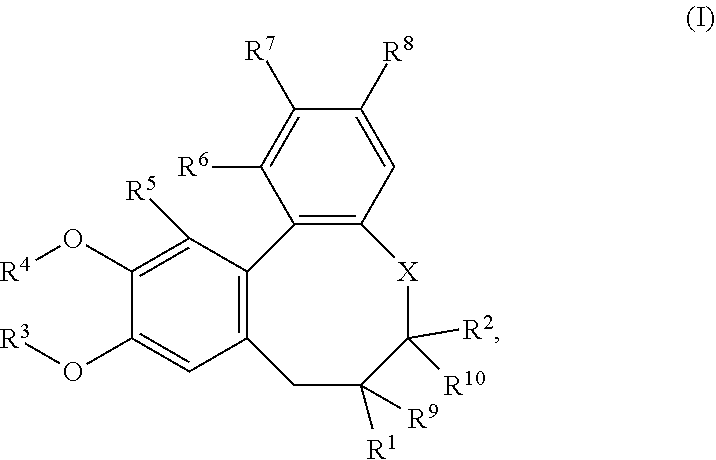
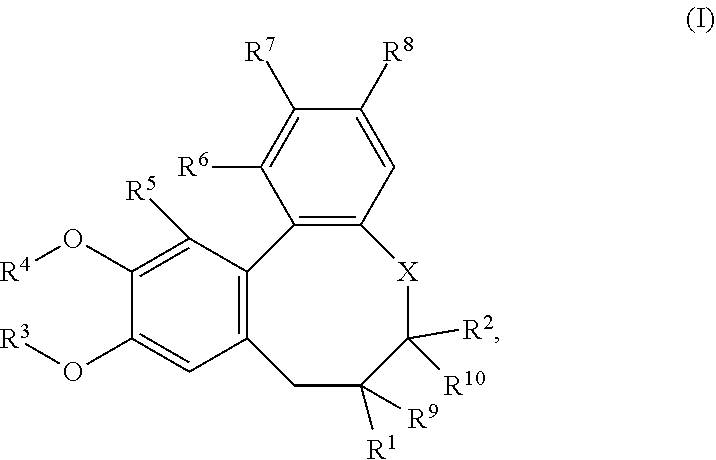
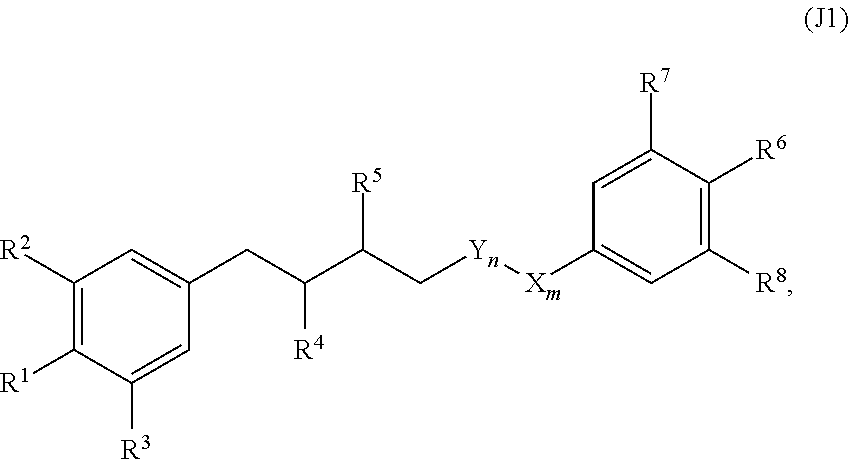
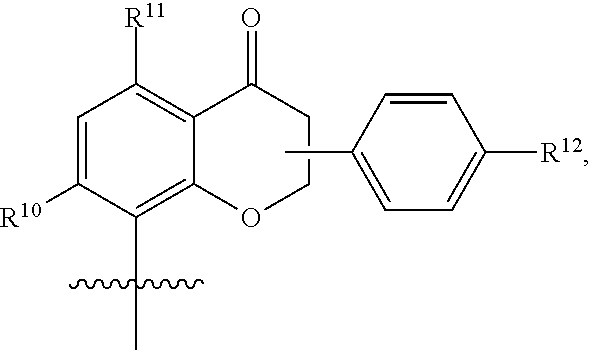
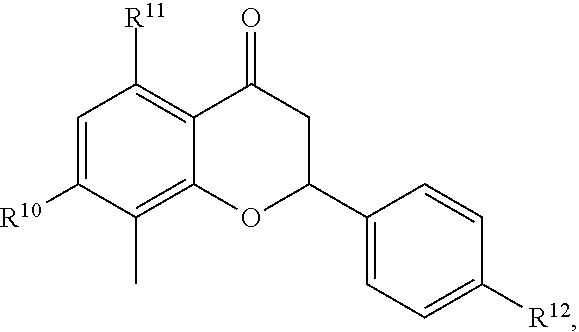
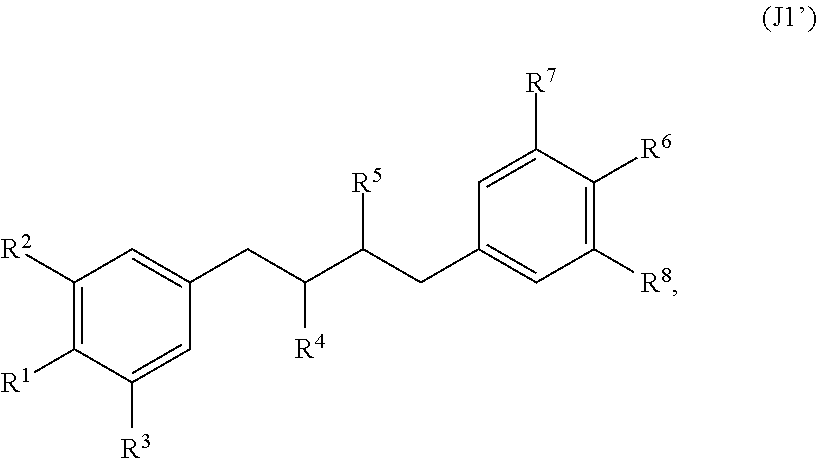

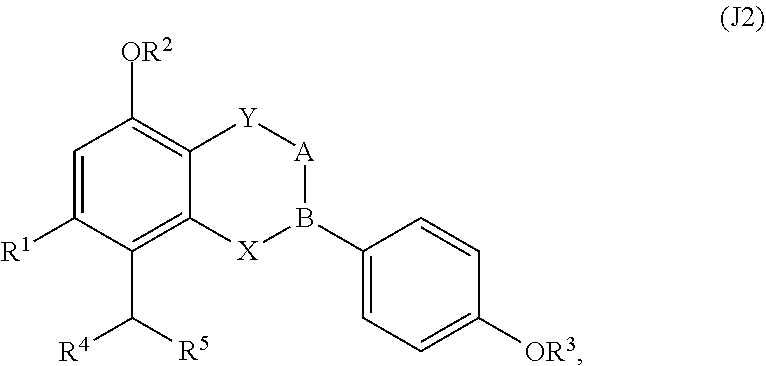
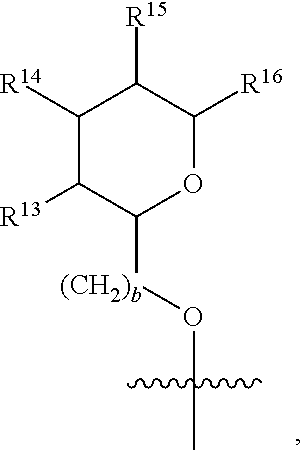
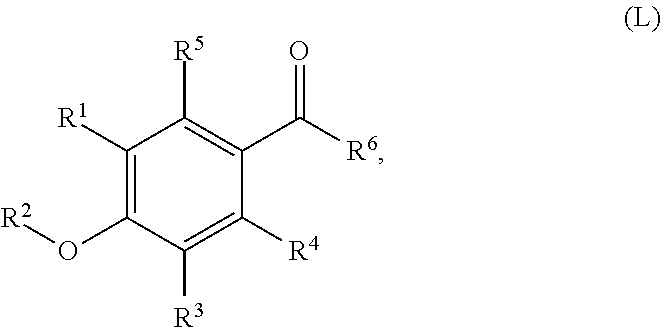
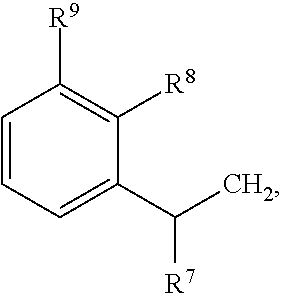
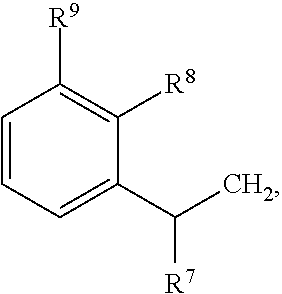
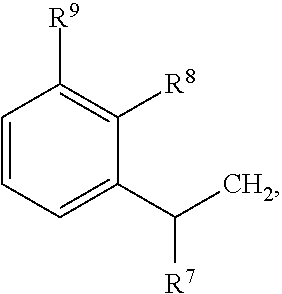
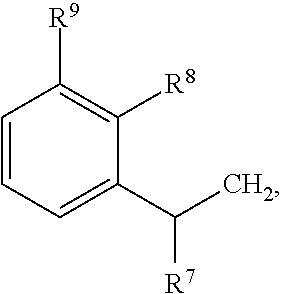
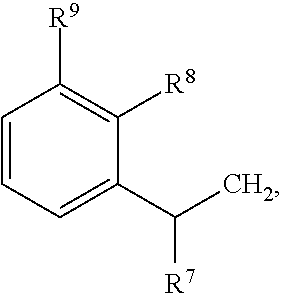
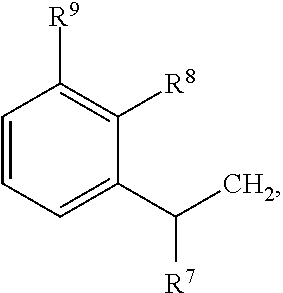

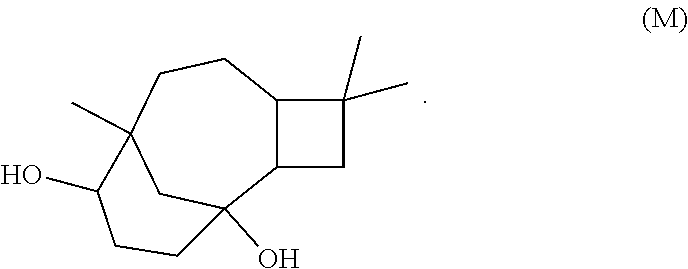
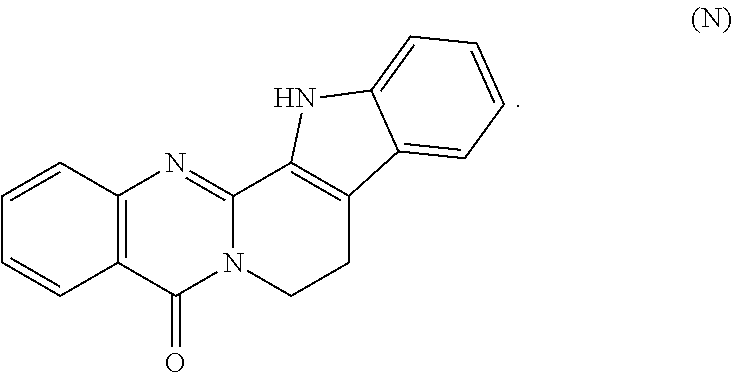
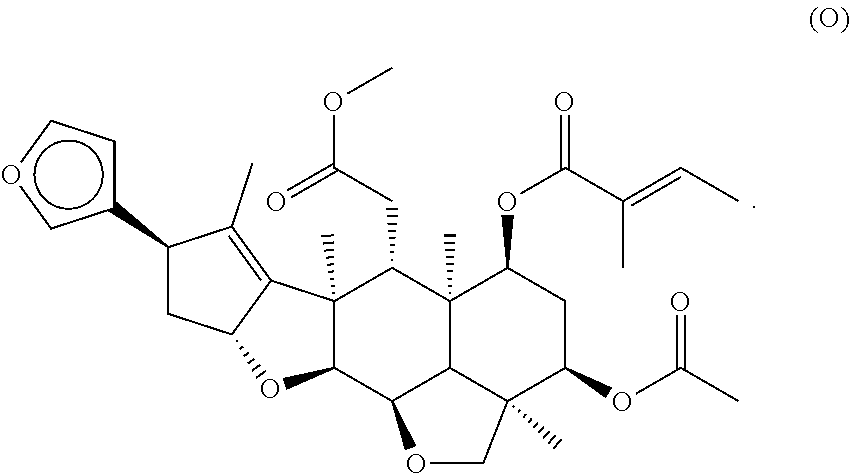
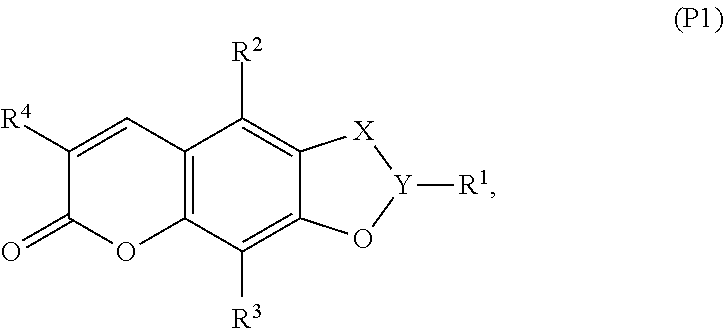
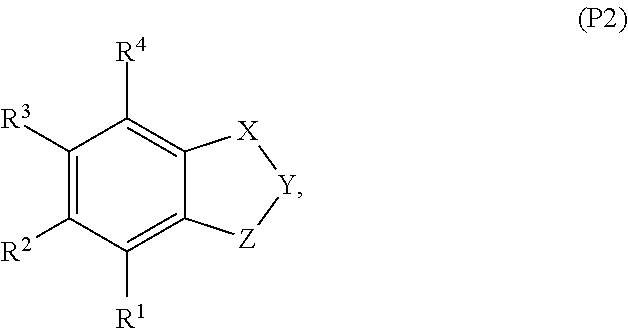
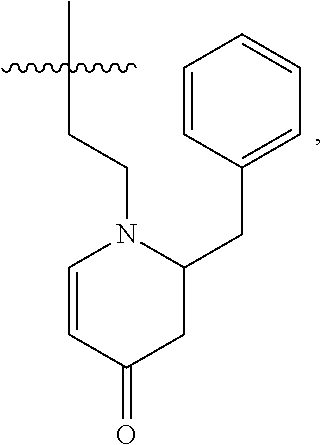
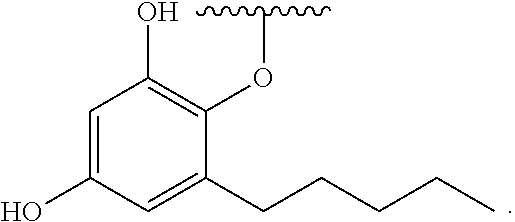
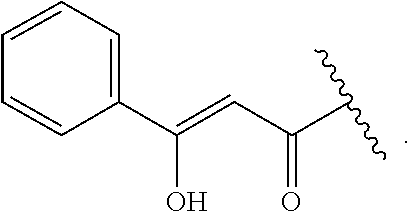
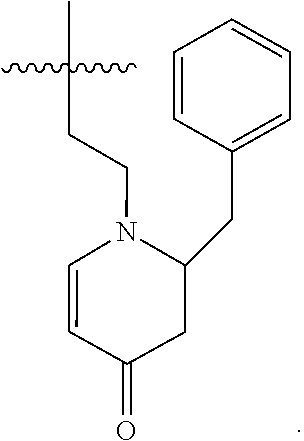
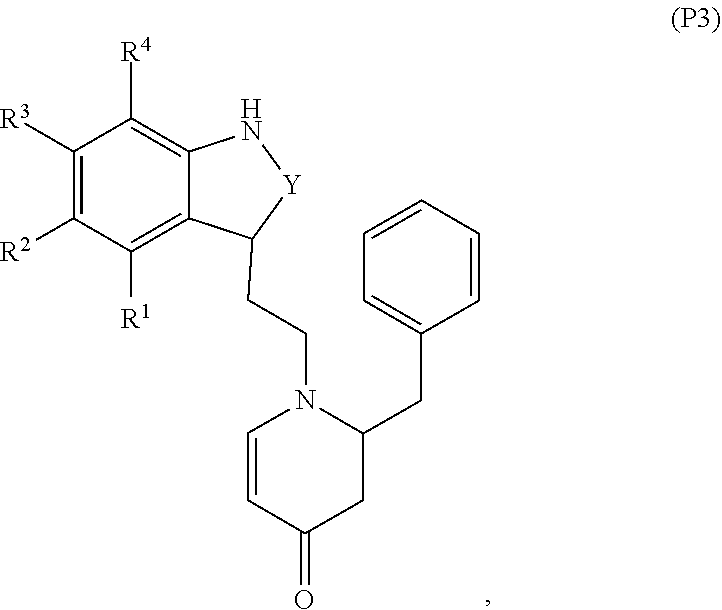
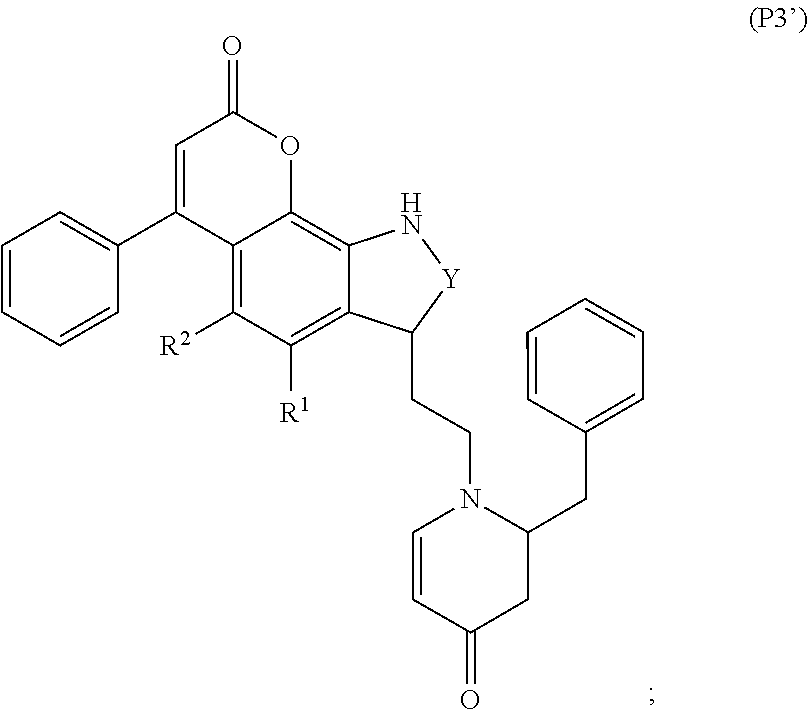
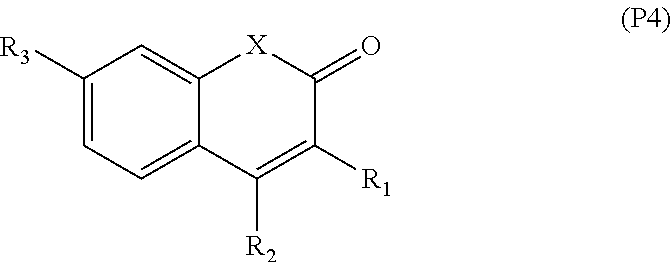
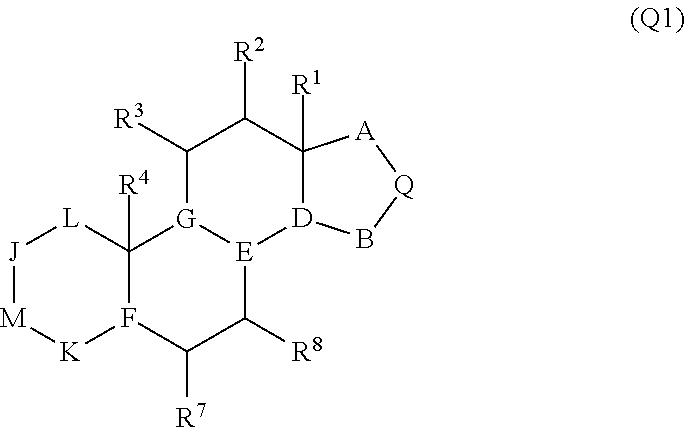
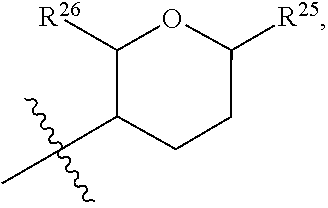
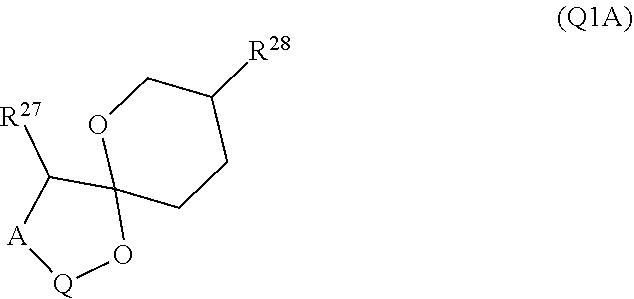
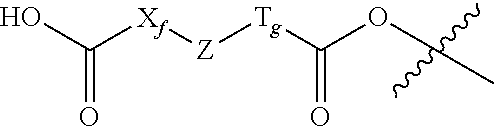
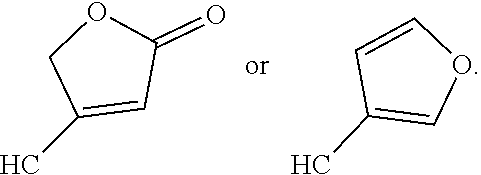
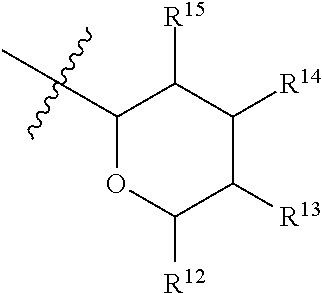
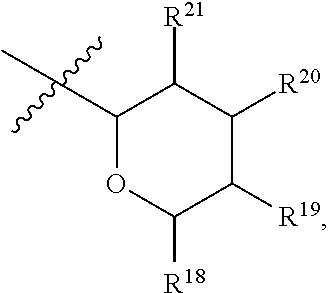
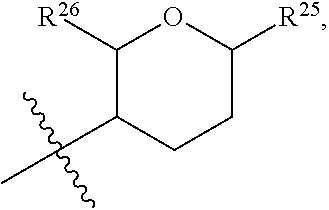
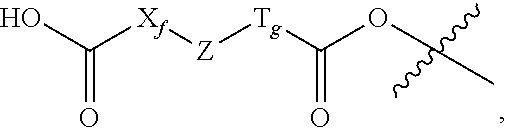
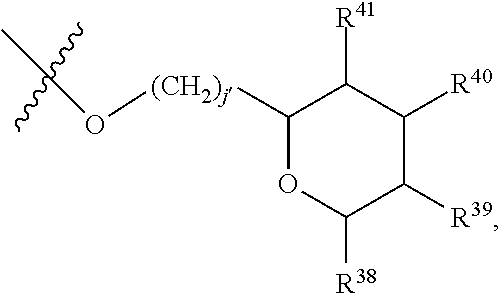
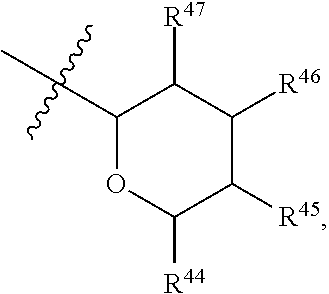
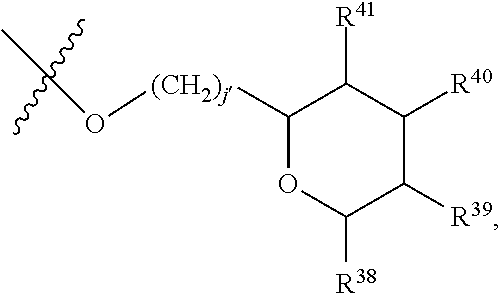
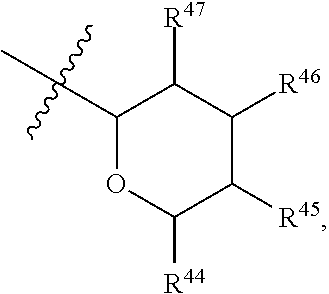
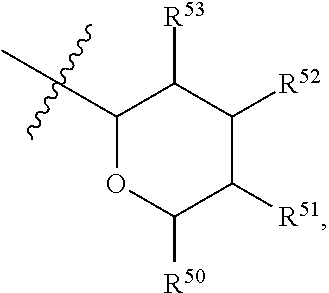
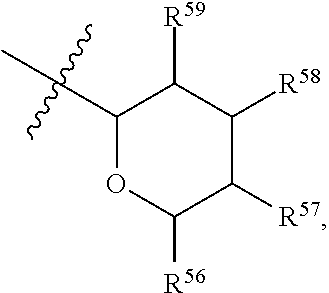
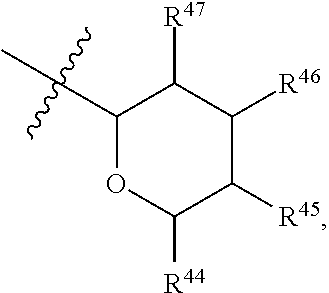
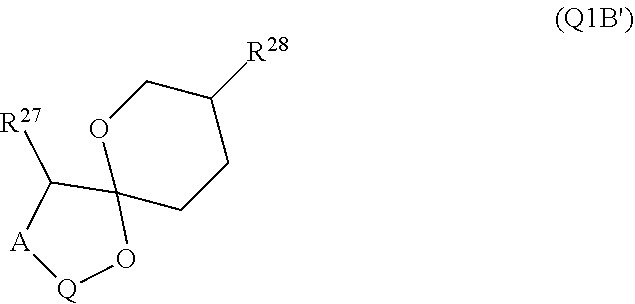
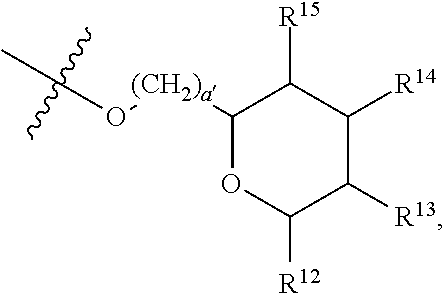
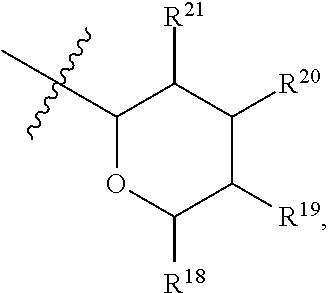
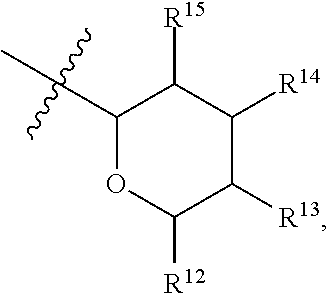
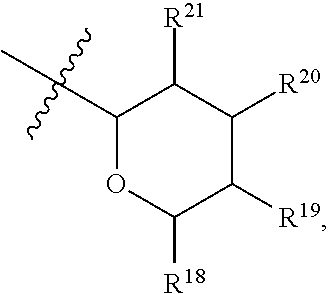
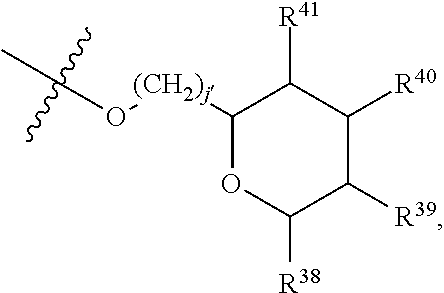
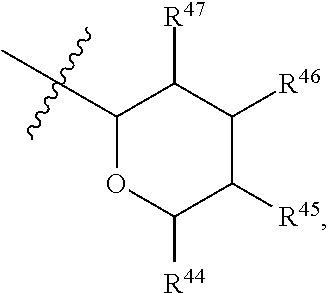
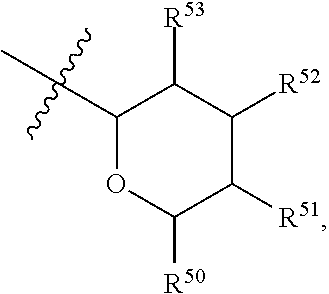
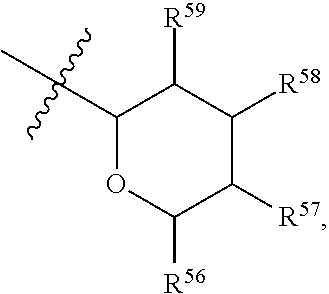
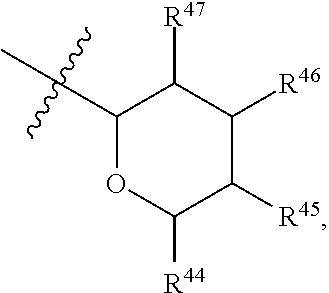
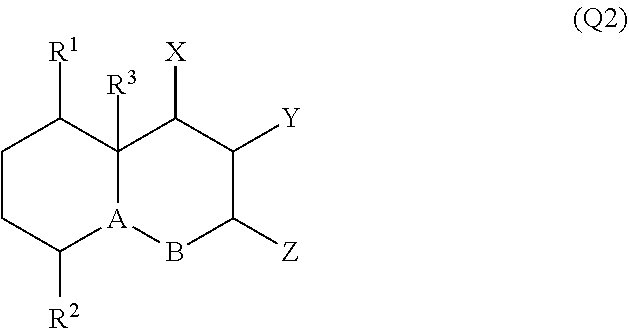
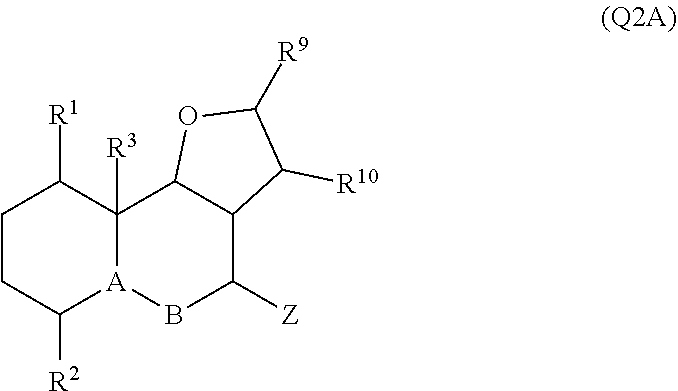

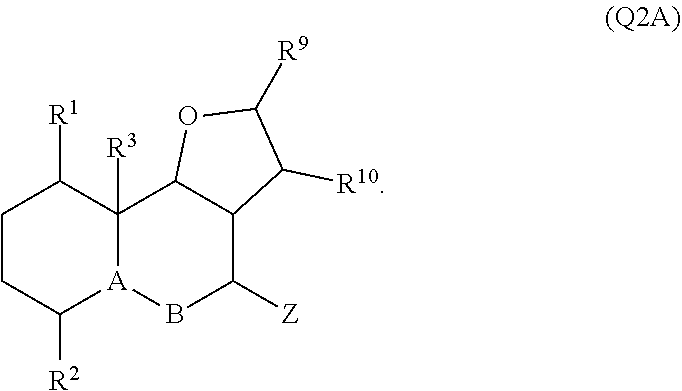
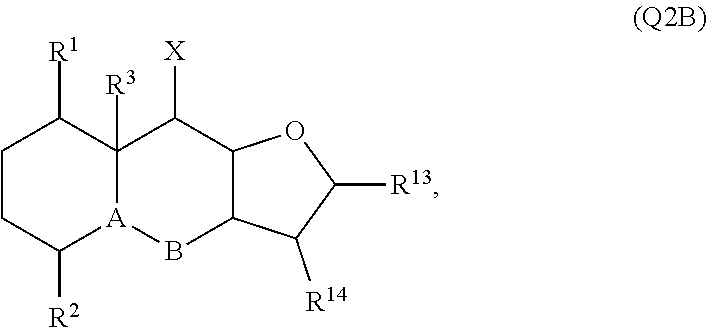
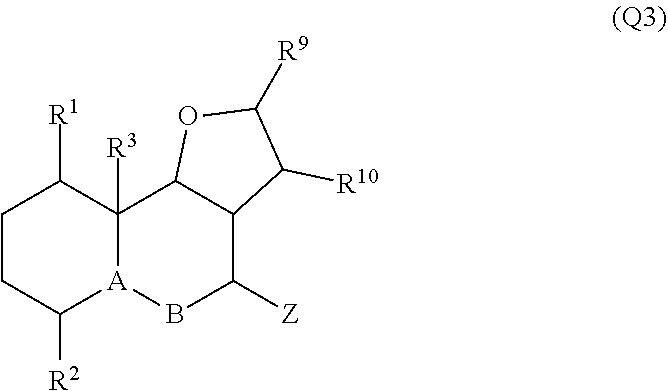
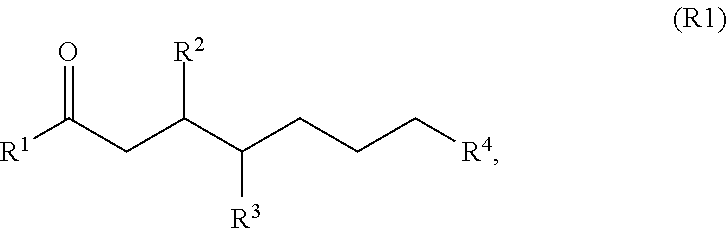

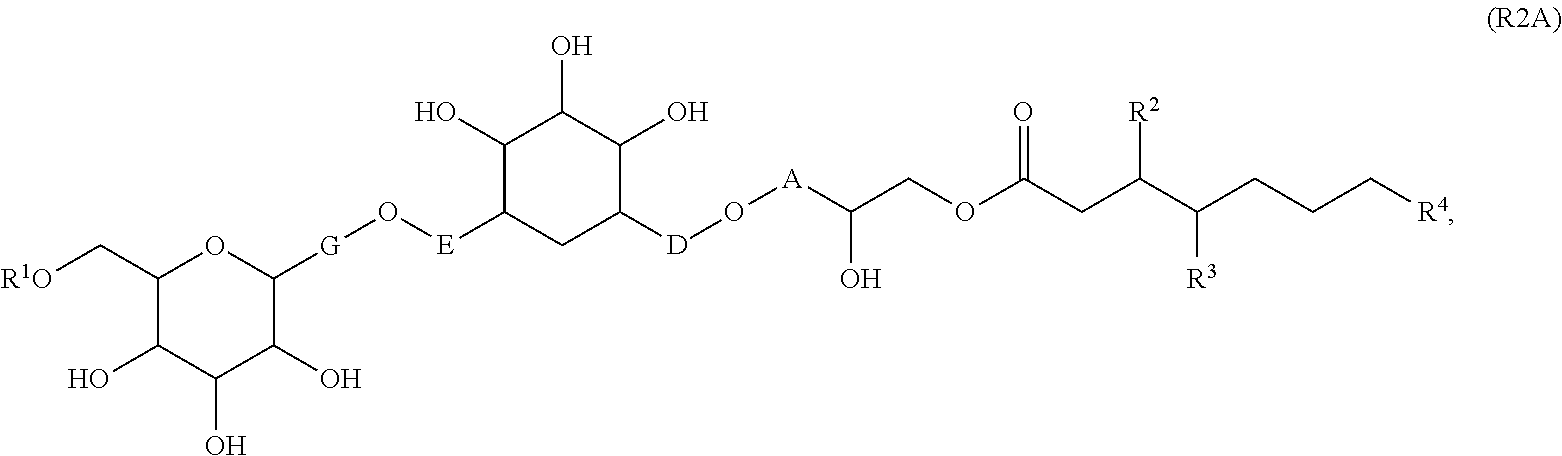
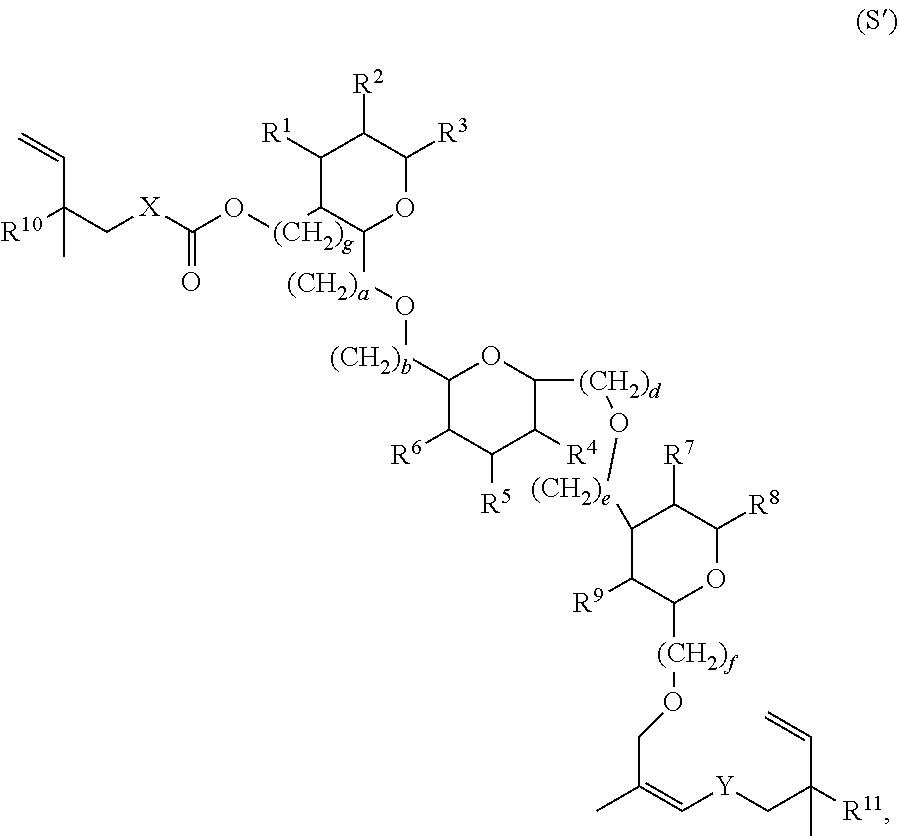
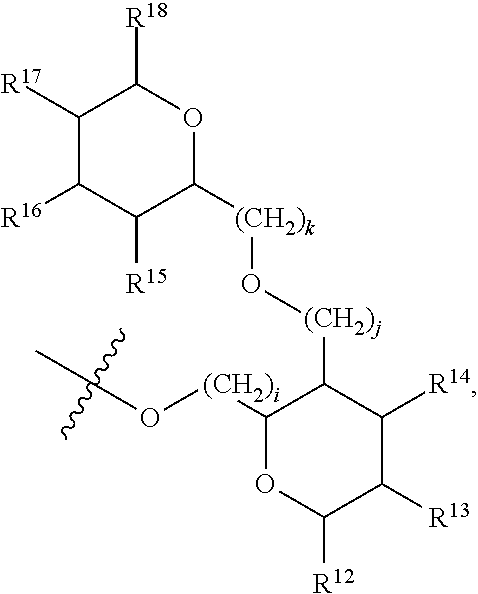
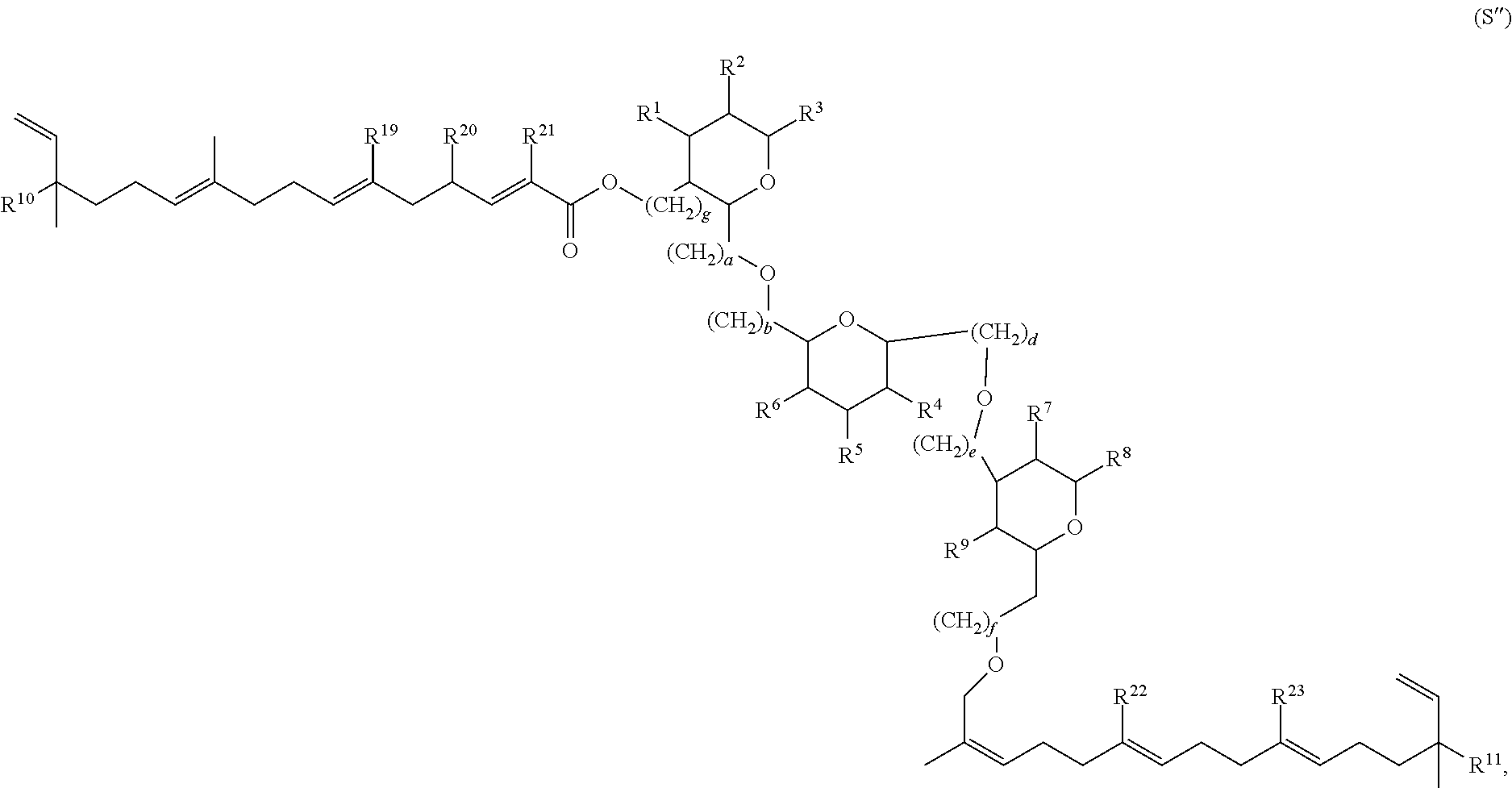
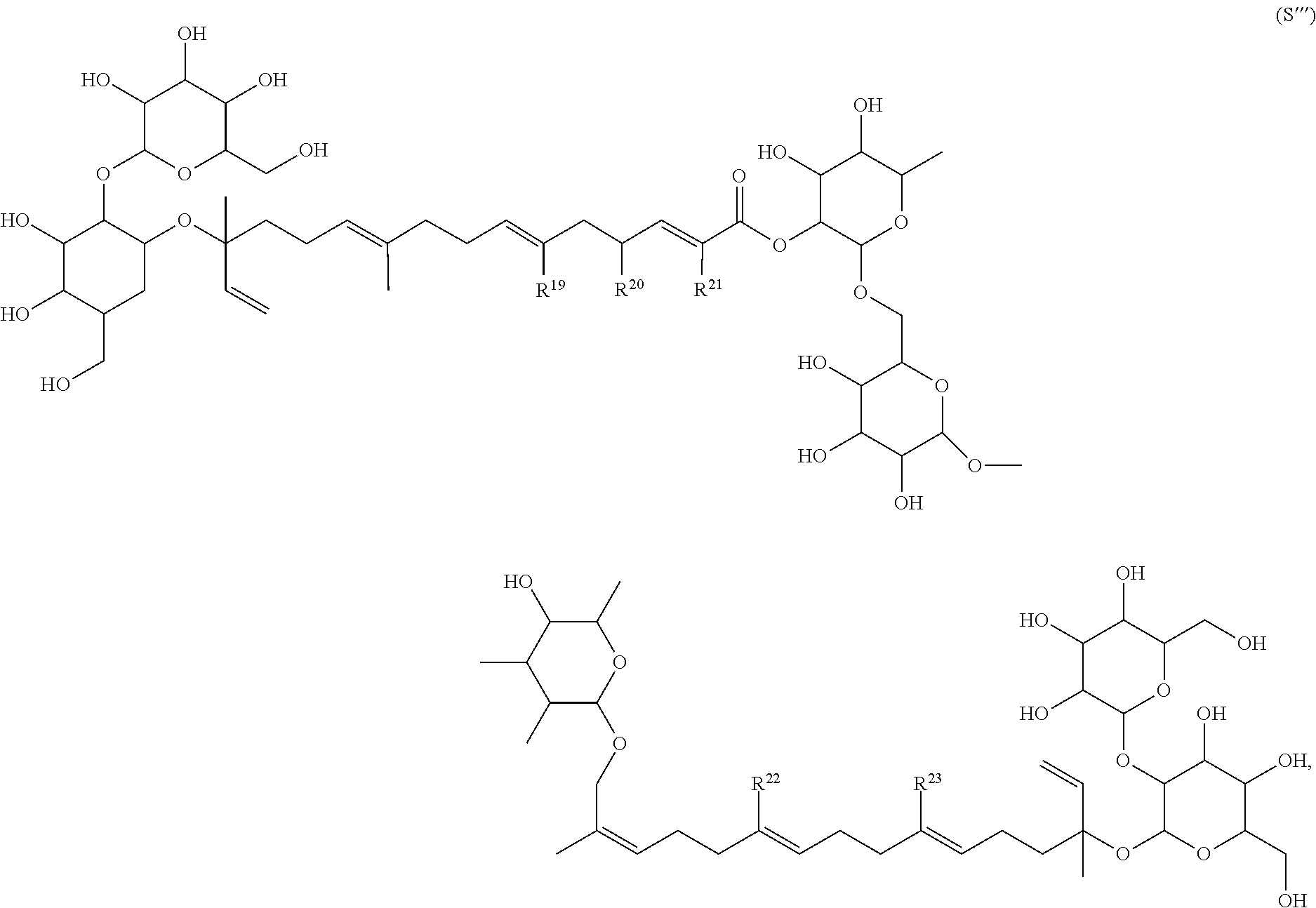
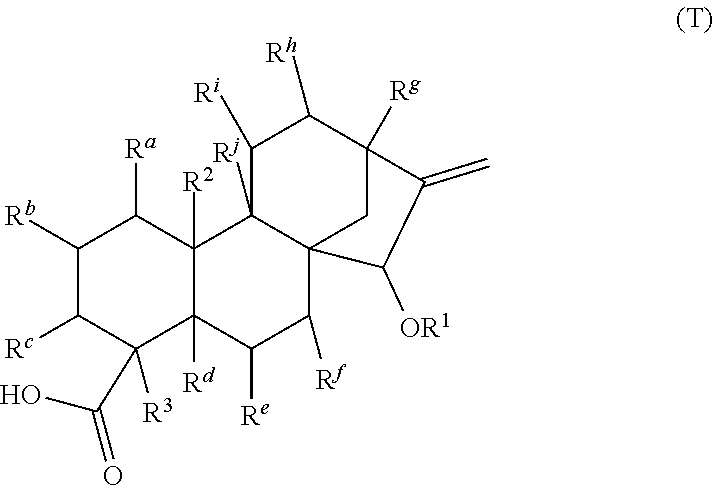
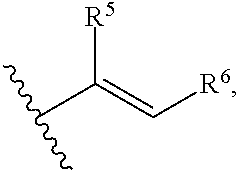
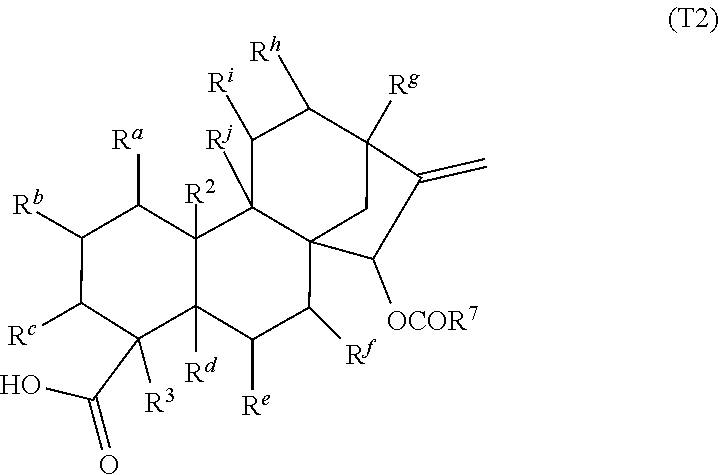
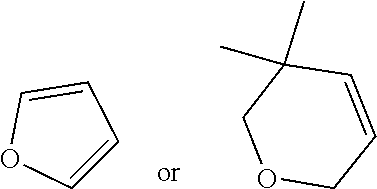
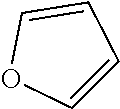
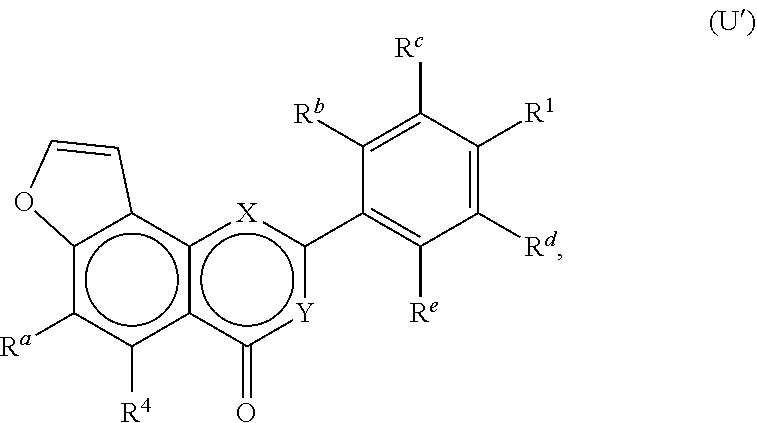
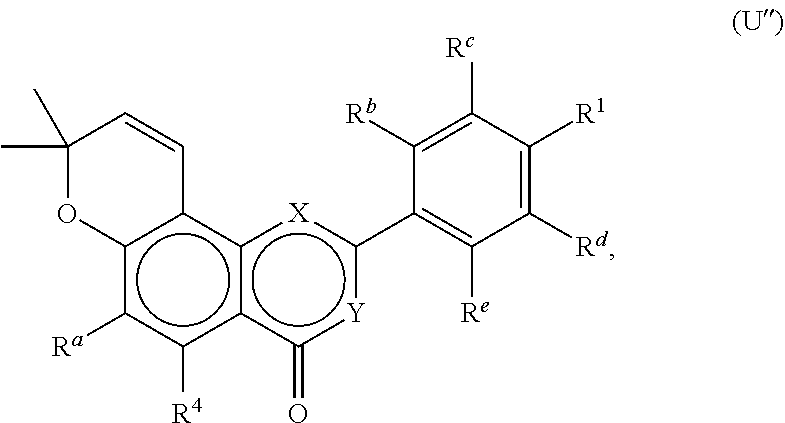
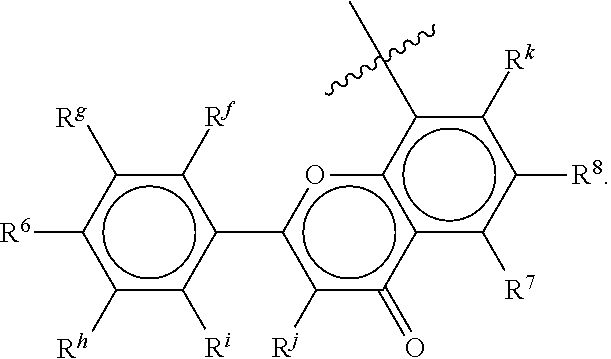
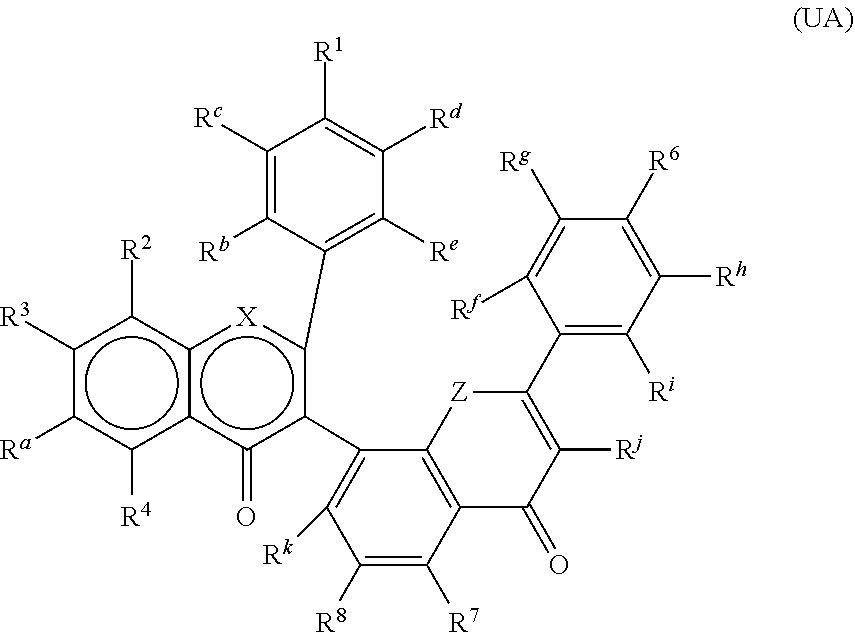
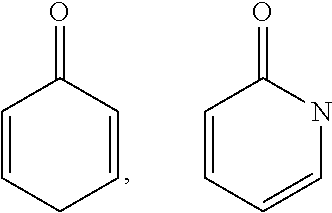
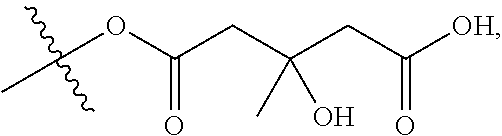
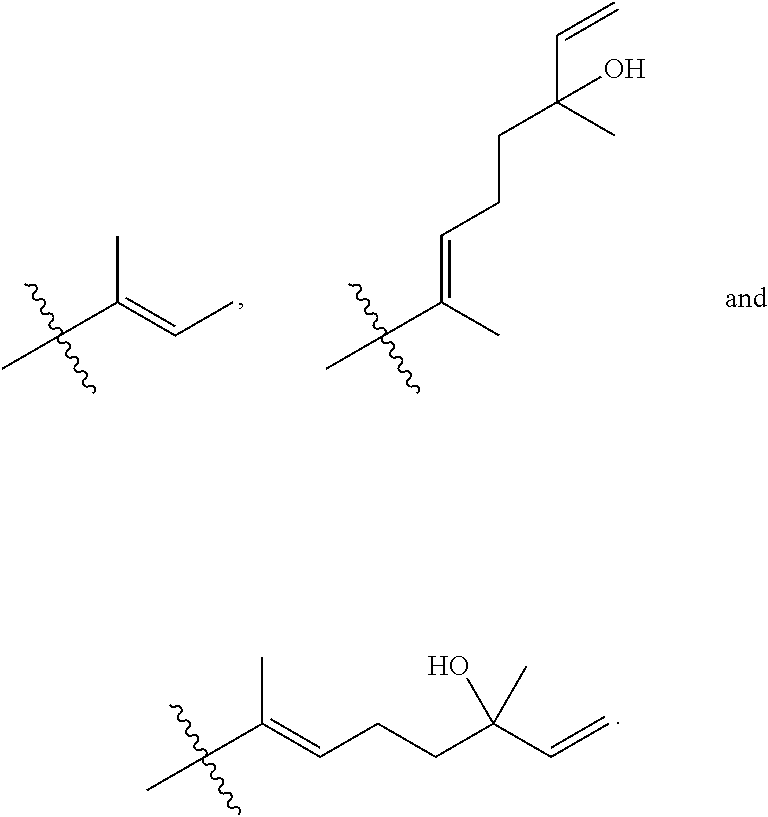
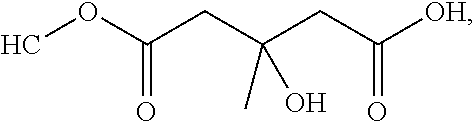
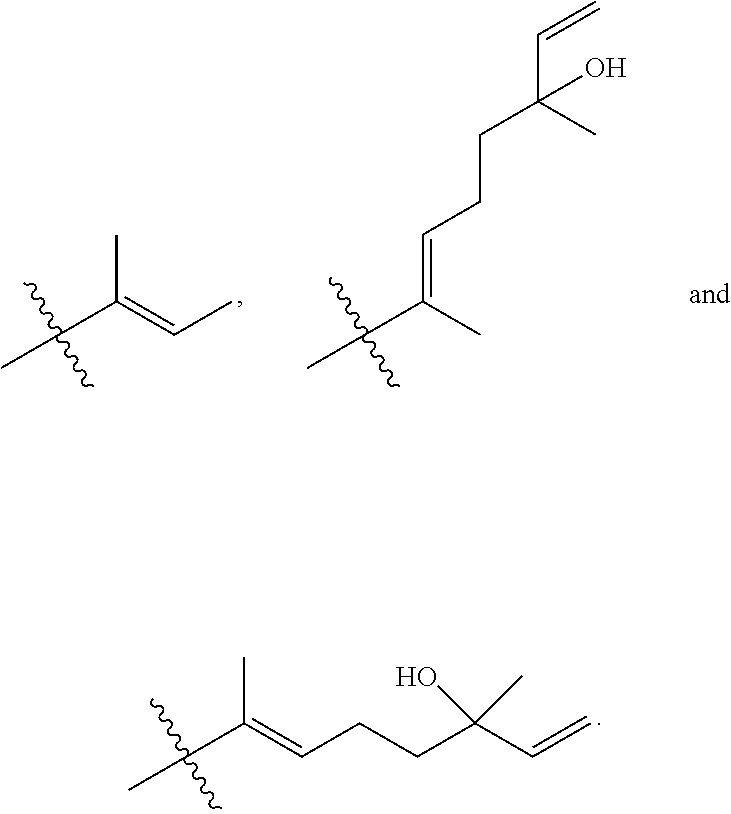
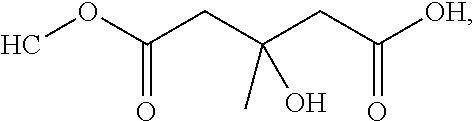
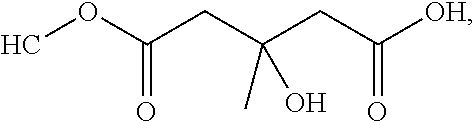
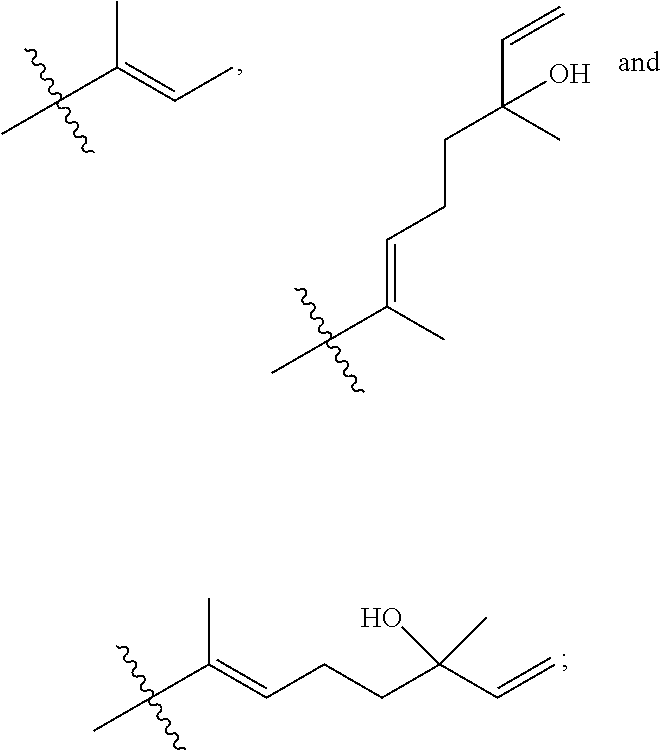
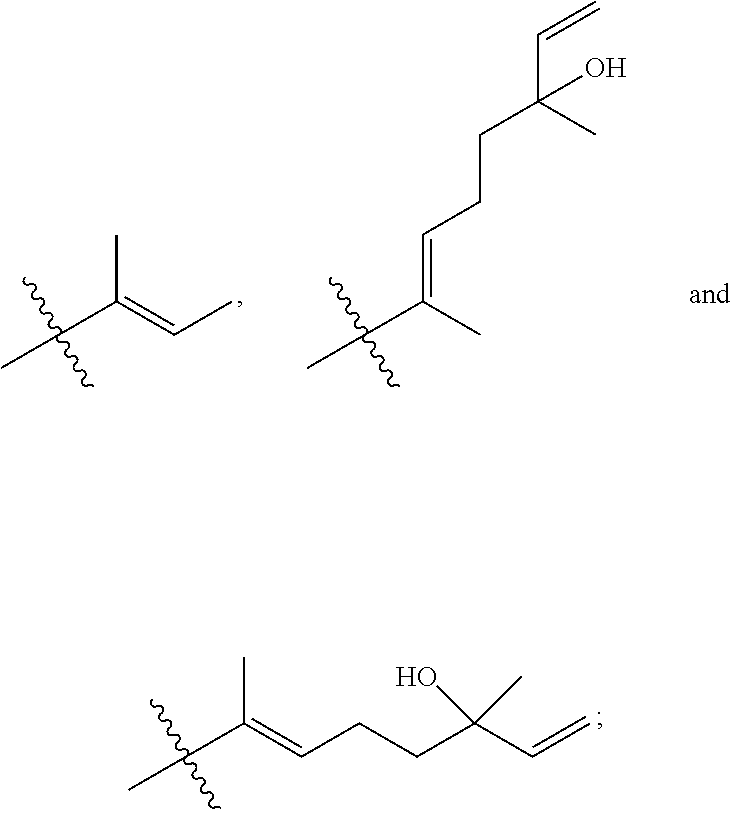
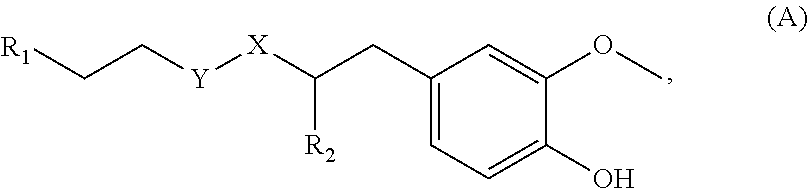
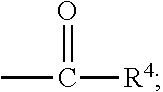
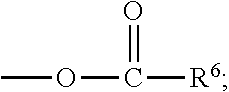
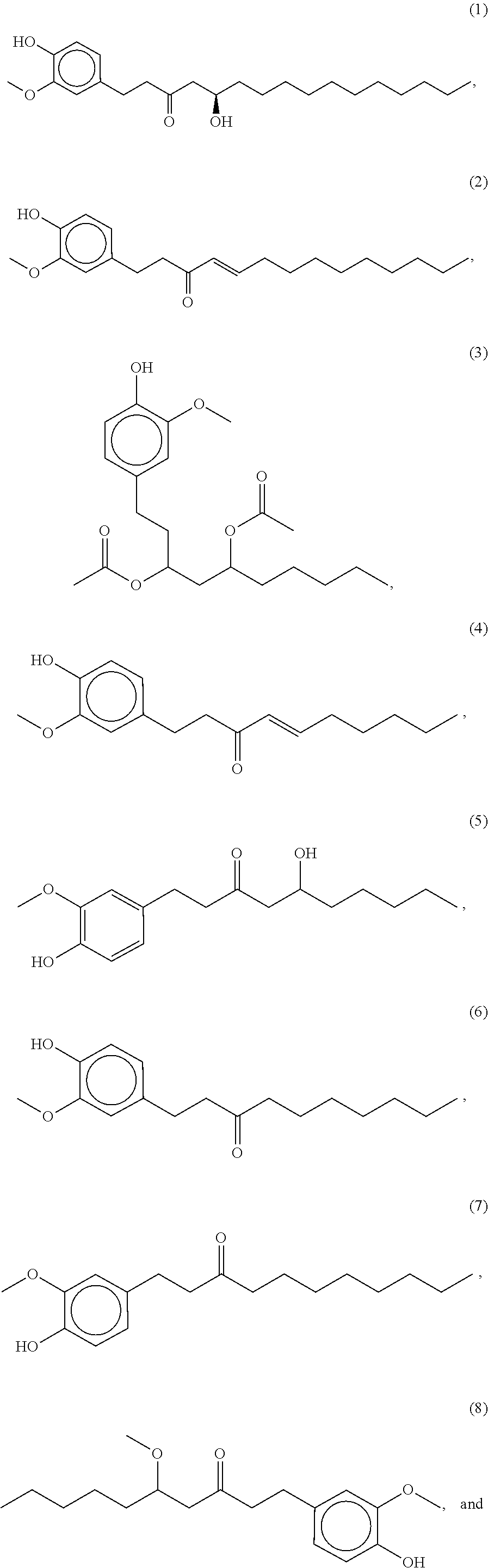
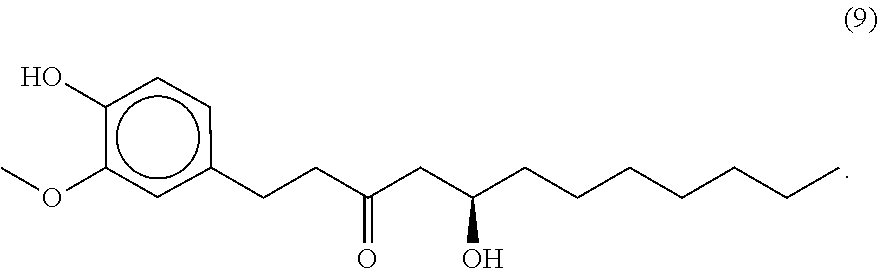
D00001
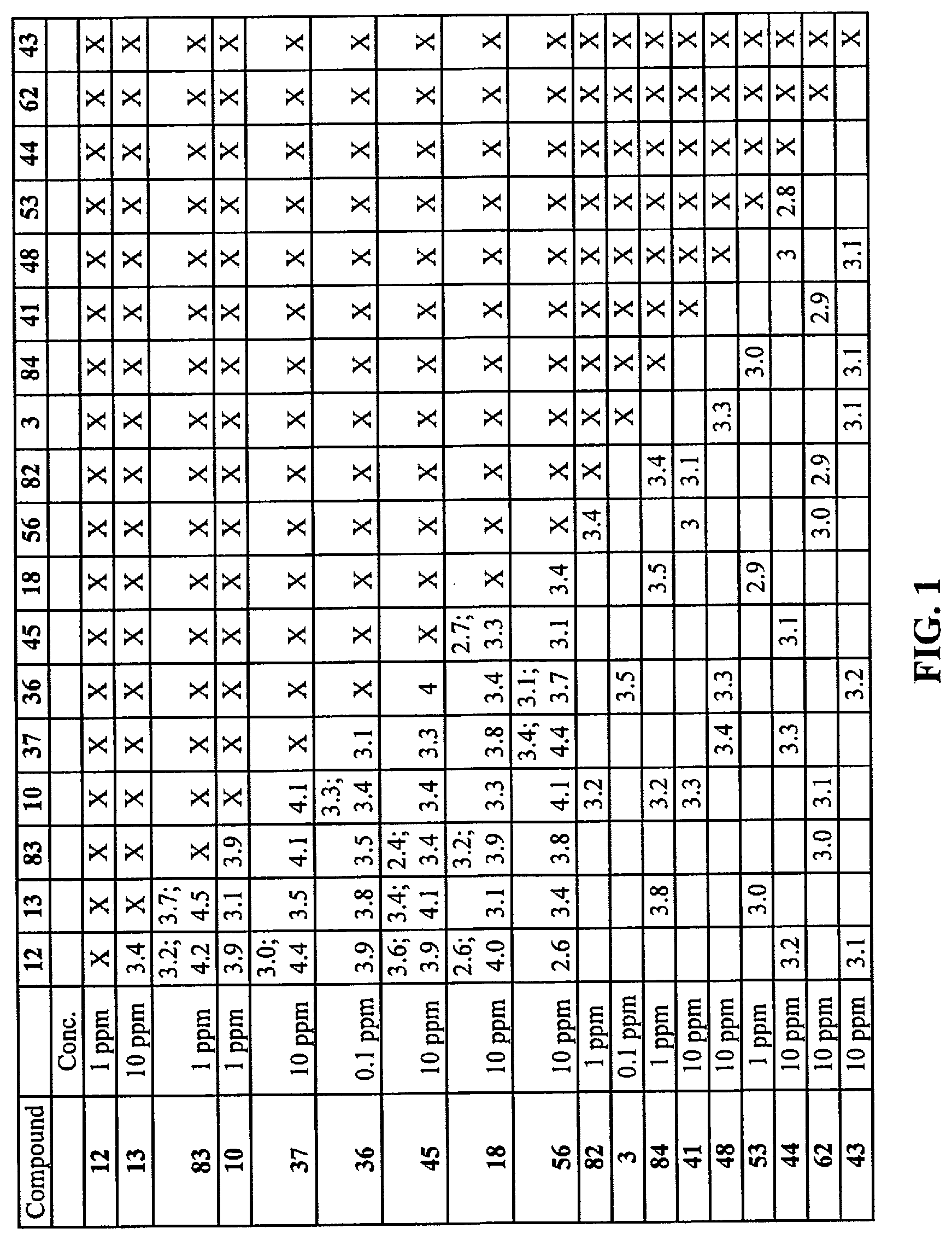
D00002
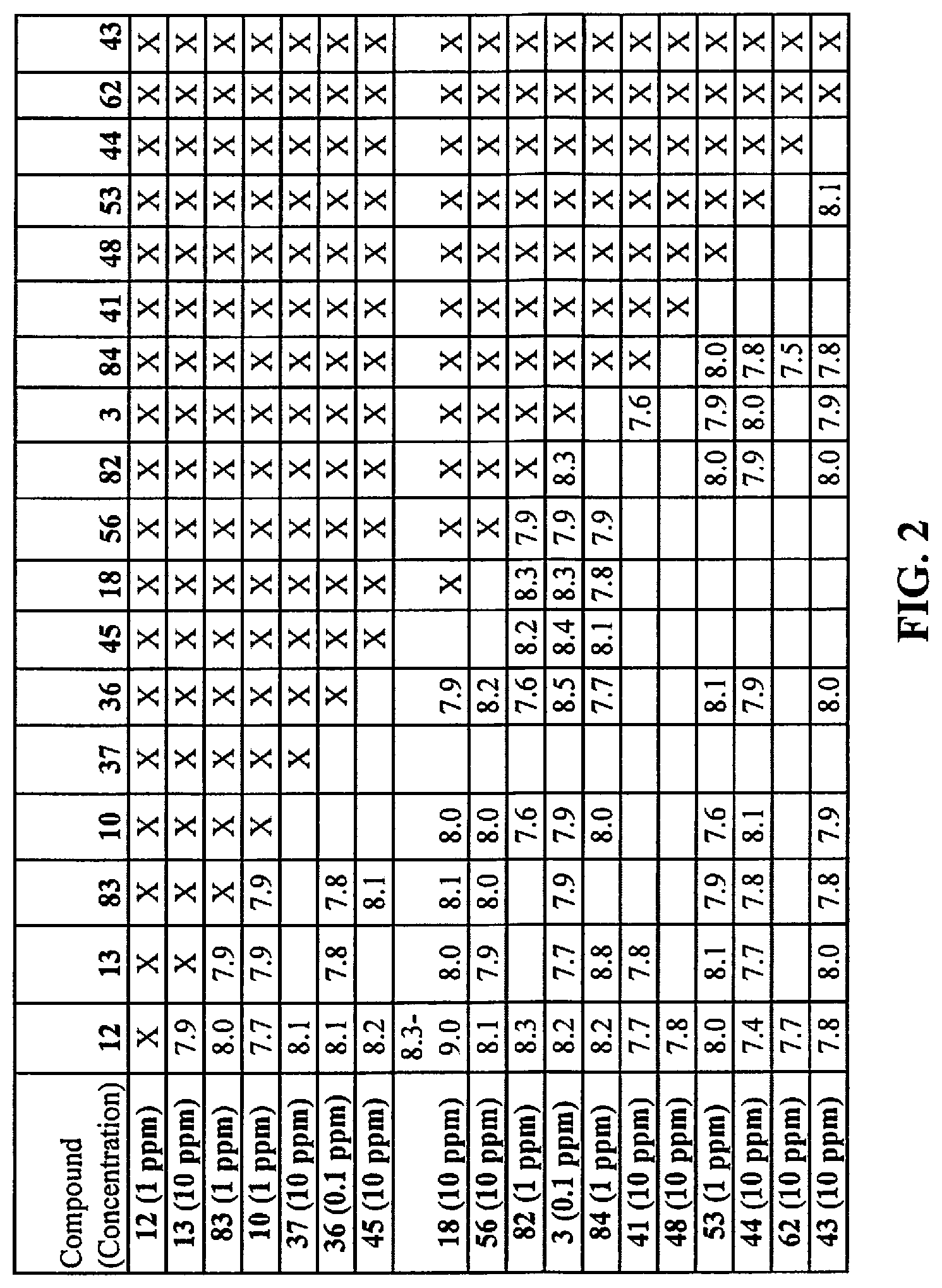
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.