Heat Insulator
HINO; HIROHISA ; et al.
U.S. patent application number 16/198236 was filed with the patent office on 2019-06-13 for heat insulator. The applicant listed for this patent is Panasonic Intellectual Property Management Co., Ltd.. Invention is credited to HIROHISA HINO, TAICHI NAKAMURA, KAZUMA OIKAWA, SHIGEAKI SAKATANI.
| Application Number | 20190177911 16/198236 |
| Document ID | / |
| Family ID | 66629275 |
| Filed Date | 2019-06-13 |

| United States Patent Application | 20190177911 |
| Kind Code | A1 |
| HINO; HIROHISA ; et al. | June 13, 2019 |
HEAT INSULATOR
Abstract
A heat insulator is provided that has a coating structure preventing separation of silica aerogel in a composite sheet. The heat insulator includes: a composite layer having silica aerogel enclosed in a nonwoven fabric; and a coating film containing a hydrophilic resin and a lipophilic resin and coating a surface of the composite layer. The nonwoven fabric resides in the coating film. In the coating film, the heat insulator includes the lipophilic resin which is present in the hydrophilic resin in the form of a plurality of islands.
| Inventors: | HINO; HIROHISA; (Osaka, JP) ; NAKAMURA; TAICHI; (Osaka, JP) ; OIKAWA; KAZUMA; (Osaka, JP) ; SAKATANI; SHIGEAKI; (Osaka, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 66629275 | ||||||||||
| Appl. No.: | 16/198236 | ||||||||||
| Filed: | November 21, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | D06M 15/507 20130101; D06N 3/12 20130101; D06N 2209/065 20130101; D06N 3/0011 20130101; D06N 3/0059 20130101; D06M 2400/02 20130101; D06M 15/55 20130101; F16L 59/06 20130101; D06M 11/79 20130101; F16L 59/00 20130101; B32B 5/16 20130101 |
| International Class: | D06N 3/00 20060101 D06N003/00; D06N 3/12 20060101 D06N003/12 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Dec 8, 2017 | JP | 2017-235639 |
Claims
1. A heat insulator comprising: a composite layer having silica aerogel enclosed in a nonwoven fabric; and a coating film containing a hydrophilic resin and a lipophilic resin and coating a surface of the composite layer.
2. The heat insulator according to claim 1, wherein the nonwoven fabric resides in the coating film.
3. The heat insulator according to claim 1, wherein the lipophilic resin is present in the hydrophilic resin in the form of a plurality of islands.
4. The heat insulator according to claim 3, wherein the nonwoven fabric is lipophilic, and resides on the lipophilic resin present in the form of islands.
5. The heat insulator according to claim 1, wherein the hydrophilic resin is a hydrophilic coating material-base resin.
6. The heat insulator according to claim 1, wherein the lipophilic resin is a thermosetting resin.
7. The heat insulator according to claim 1, wherein the hydrophilic resin is a hydrophilic polyester resin, and the lipophilic resin is an epoxy resin.
8. The heat insulator according to claim 1, wherein the coating film has a thickness of 1 to 100 .mu.m.
9. The heat insulator according to claim 3, wherein the plurality of islands each have a diameter of 0.1 to 50 .mu.m.
10. The heat insulator according to claim 1, wherein the lipophilic resin is 5 to 50 weight % of the coating film.
Description
TECHNICAL FIELD
[0001] The present disclosure relates to a heat insulator, particularly to a heat insulator having a surface coating film.
BACKGROUND
[0002] There is an ongoing trend for lighter, thinner, and higher performance mobile devices. A user of a mobile device usually holds and operates the device with a hand for certain lengths of time. It is accordingly important for a mobile device to keep low surface temperature.
[0003] A method is available that prevents temperature increase of a mobile device surface with a heat insulator installed directly above a heat generating component of a mobile device. Silica aerogel, one of many heat insulators currently available, is a material having high heat insulating performance.
[0004] Silica aerogel is known as a nanoporous material having a porosity of 90% or more. In terms of aging and heat resistance, silica aerogel is more desirable than other heat insulators, and has a desirable thermal conductivity of about 15 mW/mK. However, silica aerogel lacks high mechanical strength because of its network structure of fine point-contact silica particles of several tens of nanometers. In order to overcome the weakness of silica aerogel, Oikawa et al. has proposed to improve strength by forming a sheet of silica aerogel with other materials such as a fiber, a nonwoven fabric, and a resin.
[0005] The network structure of fine silica aerogel particles is intrinsically weak, and there are cases where the individual particles break away from the network structure of silica aerogel. These free silica aerogel particles float inside a mobile device, and cause malfunctions in the device.
[0006] A common approach to preventing this drawback is to enclose and pack a silica aerogel composite sheet by covering it with laminate films (hereinafter, referred to as "laminate packing"), as disclosed in, for example, Japanese Patent Number 6064149.
[0007] However, the method of related art involves the following problem. Laminate packing is essentially a custom-made process made to individual shapes and sizes, and greatly increases cost. This pushes up the cost of the product mobile device, and the product loses some competitiveness in the market.
SUMMARY
[0008] The present disclosure is intended to provide a solution to the foregoing problem of related art, and it is an object of the present disclosure to provide a silica aerogel-containing heat insulator without using laminate packing.
[0009] According to an aspect of the disclosure, a heat insulator is used that includes: a composite layer having silica aerogel enclosed in a nonwoven fabric; and a coating film containing a hydrophilic resin and a lipophilic resin and coating a surface of the composite layer.
[0010] With the coating structure of the nonwoven fabric-silica aerogel composite layer of the aspect of the disclosure, a protective film can be formed on the silica aerogel composite layer by applying a coating material to a surface of the silica aerogel composite layer, regardless of the shape of the coating applied portion. The film has high adhesion, and can provide a heat insulator having excellent heat insulation. A device using such a heat insulator, and a method for forming a coating structure for such a heat insulator also can be provided.
BRIEF DESCRIPTION OF THE DRAWINGS
[0011] FIG. 1A is a cross sectional view of a heat insulator formed by applying a coating material of an embodiment.
[0012] FIG. 1B is a plan view of a coating film of the embodiment.
[0013] FIG. 2 is a diagram showing an appearance of the coating film of the embodiment.
DESCRIPTION OF EMBODIMENTS
[0014] An embodiment of the present disclosure is described below, with reference to the accompanying drawings.
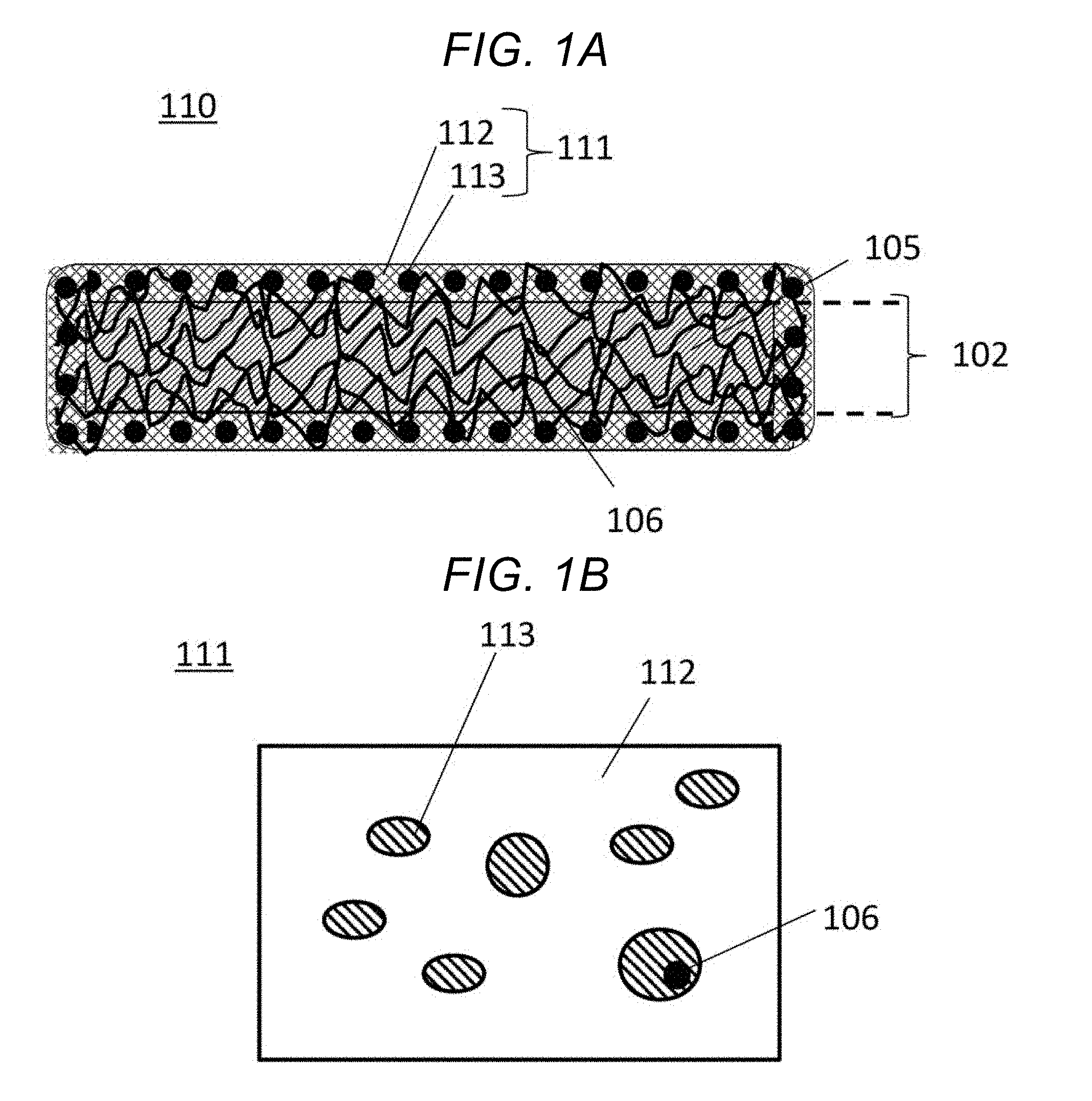
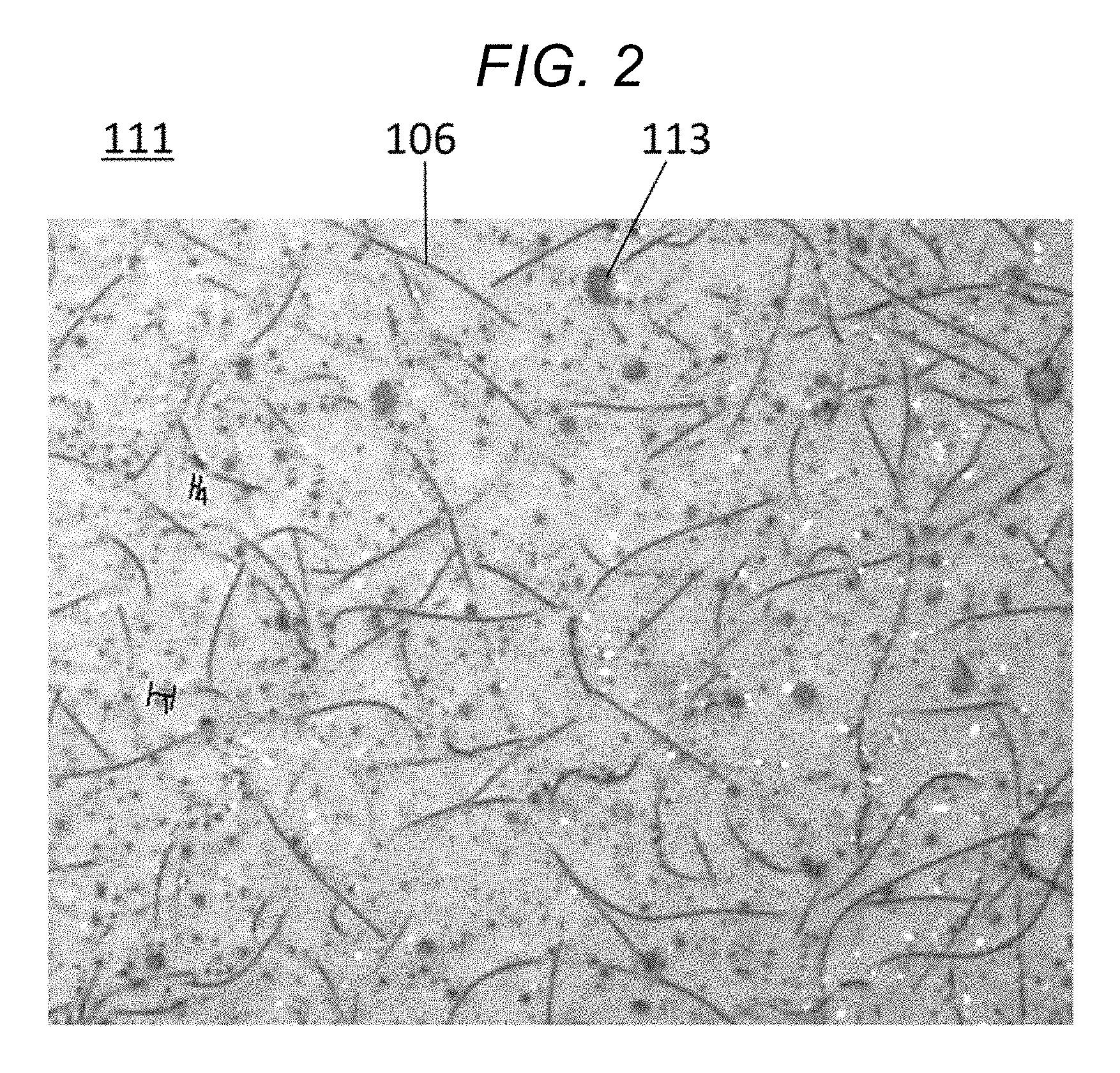
[0015] FIG. 1A shows a cross section of a heat insulator 110 with an applied coating according to an embodiment. FIG. 1B is a plan view of a coating film of the embodiment. FIG. 2 is a diagram showing an appearance of the coating film of the embodiment.
[0016] Heat insulator 110 has a structure in which coating film 111 is formed around composite layer 102 of nonwoven fabric 106 and silica aerogel 105. The fibers of nonwoven fabric 106 sticking out of composite layer 102 are adhering to hydrophilic resin 112 and reactive lipophilic resin 113 in coating film 111.
[0017] Composite Layer 102
[0018] Composite layer 102 is a sheet enclosing silica aerogel 105 of a nanosize porous structure in the nonwoven fabric 106 having a thickness of 0.05 to 1.0 mm. The thermal conductivity is 0.01 to 0.1 W/mK.
[0019] The thermal conductivity of nonwoven fabric 106 is typically 0.030 to 0.060 W/mK, and this value can be regarded as about the same as the sum of the thermal conductivity of the solid component of nonwoven fabric 106, and the thermal conductivity of the air (nitrogen molecules) component present in the spaces of the nonwoven fabric.
[0020] The low thermal conductivity specified above can be achieved by enclosing silica aerogel 105 as a low thermal conductivity material having a commonly agreed thermal conductivity of 0.010 to 0.015 W/mK--in these spaces.
[0021] The thermal conductivity of still air at ordinary temperature is said to be typically about 0.026 W/mK, and a value thereof is smaller than the thermal conductivity of nonwoven fabric 106.
[0022] A characteristic of composite layer 102 is that it is a sheet having a smaller thermal conductivity than still air.
[0023] Composite layer 102 has water repellency and sound absorbability, in addition to heat insulation. Composite layer 102 also can be imparted with heat resistance and flame retardancy by appropriately selecting the type of nonwoven fabric 106.
[0024] In the present embodiment, an oxidized acryl is used as nonwoven fabric 106 to impart heat resistance and flame retardancy. It is, however, possible to use glass fiber paper.
Thermal Conductivity of Composite Layer 102
[0025] Composite layer 102 used in the present embodiment has a thermal conductivity of 0.01 to 0.1 W/mK. The heat insulating effect of composite layer 102 increases as the thermal conductivity becomes smaller, and the thickness needed for composite layer 102 to show the same heat insulating effect can be reduced when the thermal conductivity is smaller.
[0026] The heat insulating effect becomes smaller when the thermal conductivity increases above 0.1 W/mK. This is not desirable as it necessitates thicker composite layer 102 to obtain the necessary heat insulating effect.
Thickness of Composite Layer 102
[0027] Composite layer 102 has a thickness of 0.05 mm to 2 mm, preferably 0.5 mm to 1 mm. When composite layer 102 has a thickness of less than 0.05 mm, the heat insulating effect becomes small in thickness direction, and the heat conduction across the thickness from one side to the other side of the layer cannot be desirably reduced unless a low heat conductive material of extremely low thermal conductivity (even lower than the lowest thermal conductivity currently available) is selected.
Method of Production of Composite Layer 102
[0028] An example of a method for producing composite layer 102 is described below.
(1) Mixing Raw Materials
[0029] A sol solution is prepared by adding 1.4 wt % of concentrated hydrochloric acid (12 N) as a catalyst to a high-molar sodium silicate (silicate aqueous solution; Si concentration of 14%), and stirring the mixture.
(2) Impregnation
[0030] The sol solution is poured onto nonwoven fabric 106 (the material is oxidized acryl; the thickness is 0.4 .mu.m; the basis weight is 50 g/m.sup.2; and the dimensions are 12 mm.times.12 mm), and nonwoven fabric 106 is impregnated with the sol solution under the pressure of rollers.
(3) The nonwoven fabric impregnated with the sol solution is sandwiched between two PP films (each having a thickness of 40 .mu.m), and allowed to stand at room temperature (23.degree. C.) for about 20 minutes to transform the sol into a gel.
(4) Thickness Control
[0031] After checking that the gel has formed, nonwoven fabric 106 with the films is passed through a preset gap of 650 .mu.m (including the film thickness) between two-axis rollers to squeeze out the excessive gel from nonwoven fabric 106 and achieve a target thickness of 700 .mu.m.
(5) Curing
[0032] The gel sheet with the films is put in a container, and kept in a 85.degree. C./85 RH % constant temperature and humidity vessel for 3 hours to prevent drying, and silica particles are allowed to grow (through dehydrocondensation reaction of silanol) and form a porous structure.
(6) Removing Films
[0033] The sheet is taken out of the curing container, and the films are removed.
(7) Hydrophobization 1 (Dipping in Hydrochloric Acid)
[0034] The gel sheet is dipped in hydrochloric acid (6 to 12 N), and allowed to stand at ordinary temperature (23.degree. C.) for 1 hour to incorporate hydrochloric acid in the gel sheet.
(8) Hydrophobization 2 (Siloxane Treatment)
[0035] The gel sheet is dipped in, for example, a mixture of octamethyltrisiloxane (silylation agent) and 2-propanol (IPA; amphiphatic solvent), and placed in a 55.degree. C. thermostat bath to allow reaction for 2 hours. As soon as the trimethylsiloxane bond starts to form, the gel sheet releases hydrochloric acid water, and the solution separates into two layers (the silylation agent is on the top, and the hydrochloric acid water is at the bottom).
(9) Drying
[0036] The gel sheet is transferred to a 150.degree. C. thermostat bath, and dried for 2 hours.
Coating of Composite Layer 102
[0037] Composite layer 102 is a composite of nonwoven fabric 106 and silica aerogel 105, with some of the fibers of nonwoven fabric 106 sticking out of the surface and end portions. Any method may be used to form the structure in which the fibers of the nonwoven fabric 106 are sticking out of composite layer 102, and the method is not limited.
[0038] As an example, the surface and end portions of composite layer 102 obtained after the steps (1) to (9) of producing composite layer 102 above are roughened with, for example, an adhesive roller or a brush to expose fibers.
[0039] Alternatively, composite layer 102 with sticking fibers may be obtained without grinding by optimizing the thicknesses of silica aerogel 105 and nonwoven fabric 106 in such a way as to expose fibers of nonwoven fabric 106 in the steps of producing the gel sheet.
Coating Material
[0040] The coating material that coats composite layer 102 is configured from at least a hydrophilic base coating material, and thermosetting lipophilic resin 113.
[0041] The mainstream base coating material is a hydrophilic coating material containing particulate hydrophilic resin 112 dispersed in water solvent. Because of the unique structure resulting from the hydrophobization, silica aerogel 105 of the present embodiment, unlike its original form, blends well with lipophilic resin, and the network structure is destroyed by blending with lipophilic resin. It is accordingly necessary that the coating material be basically a hydrophilic material.
[0042] The hydrophilic coating material is required to blend well with water solvent, and can be classified into a self-emulsion type having a hydrophilic functional group attached to the skeleton of hydrophilic resin 112, and a forced-emulsion type in which the resin is forcibly dispersed with the use of an emulsifier. Examples of hydrophilic resin 112 as a base material include acrylic resins, polyurethane resins, polyester resins, epoxy resins, silicone resins, and fluororesins.
[0043] As to the characteristics of each type of hydrophilic resin 112, acrylic resins typically have desirable properties including lightfastness, weather resistance, monomer variety, relatively low cost, colorlessness and transparency, and glossiness.
[0044] Polyurethane resins have different types of bonds within the molecule, such as urethane bonds and urea bonds, and are configured from a strongly aggregated hard segment and a flexible soft segment. This makes polyurethane resins desirable in properties such as adhesion for a substrate, coating hardness, abrasion resistance with high elasticity, durability, waterfastness, and chemical resistance.
[0045] The polyester resins used in the embodiment are alkyd resins, which have a fatty acid side chain linked by ester linkage to the main chain of a copolyester or a polyester of reduced crystallinity. Because the backbone of the main chain has the ester bond formed by reaction of a carboxyl group with a hydroxyl group, the polyester resins have high adhesion for a substrate, high coating strength, and desirable heat resistance.
[0046] The coating materials containing hydrophilic resin 112 as a base are inferior to common solvent-based coating materials with respect to the following, for example.
[0047] (1) The coating properties are poor because of the relatively low molecular weight (mesh structure).
[0048] (2) Because the base of the coating is a hydrophilic group, the waterfastness is intrinsically poor.
[0049] (3) Because of a lack of crosslinking reaction, the coating has a small Tg, and the adhesion is poor.
[0050] As a countermeasure against these shortcomings, the coating material used in the present embodiment has a unique structure in which thermosetting lipophilic resin 113 is added and finely dispersed in the form of islands ("island-in-sea structure") in hydrophilic resin 112.
[0051] Thermosetting lipophilic resin 113 blends well with fibers of water repellent nonwoven fabric 106, and was found to exhibit strong adhesion upon being joined to nonwoven fabric 106 and thermally cured. Various surfactants and alcohols capable of improving blendability for both hydrophilic resin 112 and lipophilic resin 113 were found to be effective at finely dispersing thermosetting lipophilic (=water repellent) resin 113 in base coating material hydrophilic resin 112. 2-Propanol (IPA) is one such alcohol. Being an amphiphatic solvent, 2-propanol was found to be capable of controlling the particle size of the thermosetting lipophilic resin 113, and the adhesion for fibers of nonwoven fabric 106 improved when thermosetting lipophilic resin 113 had a smaller particle size.
[0052] Suited as hydrophilic resin 112 used to disperse lipophilic resin 113 of the present embodiment in the form of islands (hereinafter, such a structure will be referred to as "island-in-sea structure") are water-based acrylic resins, water-based urethane resins, and water-based polyester resins, of which the water-based polyester resins are most suited.
[0053] A reactive one-component epoxy resin was found to be suitable as lipophilic resin 113. When adding a lipophilic resin to a water-based coating material, it is typical to use a hydrophilic resin having high compatibility with the water-based coating material. The present embodiment, however, is intended to improve adhesion for coating film 111 by causing lipophilic resin 113 to adhere to fibers of nonwoven fabric 106 sticking out of composite layer 102, and, in order to enable pin-point bonding and reinforcement of fibers, it is important that lipophilic resin 113 does not dissolve into the water-based coating material. To this end, a lipophilic one-component epoxy resin needs to be selected, and finely dispersed in the water-based coating material in the form of an island-in-sea structure.
Island-in-Sea Structure
[0054] Heat insulator 110 formed upon application of the coating material has a structure in which fibers of nonwoven fabric 106 are sticking out of the surface layer of composite layer 102 formed by attachment and integration of silica aerogel 105 into nonwoven fabric 106 through aggregation.
[0055] In heat insulator 110, nonwoven fabric 106 binds to coating film 111, and lipophilic resin 113 is present in coating film 111 by being dispersed in a scattered fashion in the form of an island-in-sea structure. Coating film 111 has a thickness of preferably 1 to 100 .mu.m, more preferably 10 to 30 .mu.m. When the thickness is less than 1 .mu.m, the film strength weakens, and the film easily breaks. When coating film 111 is thicker than 100 .mu.m, the heat insulating performance deteriorates.
[0056] Lipophilic resin 113 has a particle size of preferably 0.1 .mu.m to 50 .mu.m, and the epoxy content in the coating film is preferably 5 to 50 weight %.
[0057] When the particle size is less than 0.1 .mu.m, hydrophilicity increases, and the film becomes weaker. When the particle size is larger than 50 .mu.m, lipophilic resin 113 adheres to fibers of nonwoven fabric 106 at fewer points, and the adhesion weakens.
[0058] When the epoxy content (lipophilic resin 113) in coating film 111 is less than 5 weight %, lipophilic resin 113 contacts fibers of nonwoven fabric 106 at fewer contact points, and the adhesion weakens.
[0059] When the epoxy content is higher than 50 weight %, lipophilic resin 113 readily blends with lipophilic silica aerogel 105, and destroys the fine structure of silica aerogel 105. This leads to poor heat insulation. The epoxy content is further preferably 10 to 30 weight %.
EXAMPLES
[0060] Examples of hydrophilic resin 112 and thermosetting lipophilic resin 113 used in the embodiment are described below. It is to be noted that the heat insulator structure presented in the embodiment is not limited to the following exemplary materials.
[0061] A hydrophilic polyester resin Pluscoat Z-880 (Goo Chemical Co., Ltd.) was used as hydrophilic resin 112. A Novacure HX3941HP (Asahi Kasei) was used as thermosetting lipophilic resin 113. 2-Propanol (IPA) (amphiphatic solvent) was used as a compatibilizing agent.
(1) Exemplary Composition of Coating Material Forming Island-in-Sea Structure
(a) Hydrophilic Resin 112
[0062] Water-based polyester resin Pluscoat Z-880 (solid content of 25 wt %): 100 parts
(b) Lipophilic Resin 113
[0063] One-component epoxy resin Novacure HX3941HP (imidazole fraction=1/3 wt %, epoxy fraction=2/3 wt %): 10 parts
(c) Compatibilizing Agent
[0064] 2-Propanol (IPA): 10 parts
(2) Production of Coating Material
[0065] The components (a) to (c) were weighed, and mixed and stirred with a disper for 15 minutes to make a coating material for forming an island-in-sea structure.
(3) Application of Coating Material
[0066] The coating material is applied by printing on composite layer 102 having fibers of nonwoven fabric 106 sticking out of the surface portion, using a printing mask and a squeegee. After application, the coating material is heat cured to form coating film 111.
[0067] Coating film 111 may cover the whole composite layer 102 with the coating material applied to not only the surface portion but end portions of composite layer 102. All surfaces of the composite layer 102 can be coated by applying the coating material to both surfaces and end portions in the same fashion.
[0068] When the area to be coated is small, composite layer 102 may be completely dipped in the coating material using a dipping method.
(4) Curing of Coating Material
[0069] Composite layer 102 after the application of the coating material to both surfaces is dried in a 120.degree. C. thermostat bath for 15 minutes. This causes water to evaporate, and the particles of the epoxy resin (lipophilic resin 113) to cure in a curing reaction, forming coating film 111.
(5) Shape of Coating Film 111
[0070] Coating film 111 had a thickness of 30 .mu.m, and thermosetting lipophilic resin 113 had a particle size of 5 .mu.m. The particle fraction of thermosetting lipophilic resin 113 was about 20%.
Structure and Properties of Composite Layer 102 Coated with Coating Material
[0071] Nonwoven fabric 106 of composite layer 102 had some of its fibers sticking out of the surface layer of composite layer 102, and these fibers, which are lipophilic, blended well with lipophilic resin 113 of coating film 111 in the outermost layer, and strongly bonded itself to lipophilic resin 113 in a reaction that provided excellent adhesion.
[0072] In coating film 111, reactive lipophilic resin 113 is dispersed in the form of islands in an island-in-sea structure, and strong adhesion occurs as lipophilic resin 113 chemically reacts with fibers of nonwoven fabric 106.
[0073] In some applications, the heat insulating effect of heat insulator 110 is exploited by placing heat insulator 110 over a curved surface of a heat generating component. Here, the base polyester resin is flexible, and the foregoing structure provides high adhesion. This prevents detachment of coating film 111 from composite layer 102, and heat insulator 110 can exhibit desirable heat insulating performance. The fine particles of silica aerogel 105 enclosed in composite layer 102 also can be prevented from being exposed on the surface.
Adhesion Force Between Composite Layer 102 and Coating Resin Film
[0074] Coating film 111 measuring 10 mm in width and 30 .mu.m in thickness was formed on composite layer 102, and tensile strength was measured using a 90-degree peeling method.
(a) 0.7 N with no sticking nonwoven fabric 106 (b) 5.5 N with sticking nonwoven fabric 106 (c) 2.3 N with no thermosetting resin in (b)
Thermal Conductivity of Heat Insulator 110
[0075] Heat insulator 110 formed by coating a 30 .mu.m-thick coating film 111 on composite layer 102 had a thermal conductivity of 0.07 W/mK, confirming that heat insulator 110 had excellent heat insulating performance comparable to that of composite layer 102 alone.
[0076] The heat insulator of the embodiment can prevent separation of silica aerogel particles, and, because the heat insulating performance is maintained, the heat insulator can be used for heat insulating purposes in applications such as in mobile devices.
* * * * *
D00000

D00001

D00002

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.