Alarm suspend system
Kiani , et al. Feb
U.S. patent number RE47,249 [Application Number 15/583,948] was granted by the patent office on 2019-02-19 for alarm suspend system. This patent grant is currently assigned to MASIMO CORPORATION. The grantee listed for this patent is MASIMO CORPORATION. Invention is credited to Steve L. Cebada, Massi Joe E. Kiani, Gregory A. Olsen.



| United States Patent | RE47,249 |
| Kiani , et al. | February 19, 2019 |
Alarm suspend system
Abstract
An alarm suspend system utilizes an alarm trigger responsive to physiological parameters and corresponding limits on those parameters. The parameters are associated with both fast and slow treatment times corresponding to length of time it takes for a person to respond to medical treatment for out-of-limit parameter measurements. Audible and visual alarms respond to the alarm trigger. An alarm silence button is pressed to silence the audible alarm for a predetermined suspend time. The audible alarm is activated after the suspend time has lapsed. Longer suspend times are associated with slow treatment parameters and shorter suspend times are associated with fast treatment parameters.
| Inventors: | Kiani; Massi Joe E. (Laguna Niguel, CA), Cebada; Steve L. (Mission Viejo, CA), Olsen; Gregory A. (Trabuco Canyon, CA) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | MASIMO CORPORATION (Irvine,
CA) |
||||||||||
| Family ID: | 41351506 | ||||||||||
| Appl. No.: | 15/583,948 | ||||||||||
| Filed: | May 1, 2017 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | Issue Date | ||
|---|---|---|---|---|---|
| 14036496 | Sep 30, 2014 | 8847740 | |||
| 13476725 | Oct 1, 2013 | 8547209 | |||
| 12510982 | Jun 19, 2012 | 8203438 | |||
| 61084615 | Jul 29, 2008 | ||||
| Reissue of: | 14469426 | Aug 26, 2014 | 9153121 | Oct 6, 2015 | |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | G08B 25/001 (20130101); A61B 5/1455 (20130101); A61B 5/1455 (20130101); G08B 25/001 (20130101); A61B 5/746 (20130101); A61B 5/6838 (20130101); A61B 5/746 (20130101); A61B 5/6838 (20130101); A61B 5/6826 (20130101); A61B 5/6826 (20130101); A61B 2560/0276 (20130101); G08B 5/22 (20130101); G08B 5/22 (20130101); A61B 2560/0276 (20130101) |
| Current International Class: | G08B 5/22 (20060101); G08B 25/00 (20060101); A61B 5/1455 (20060101); A61B 5/00 (20060101) |
| Field of Search: | ;340/511,517,573.1,539.12,286.07,527 ;600/310,316,322-324,336,344,507,301,320 |
References Cited [Referenced By]
U.S. Patent Documents
| 4639718 | January 1987 | Gasper |
| 4653498 | March 1987 | New et al. |
| 4960128 | October 1990 | Gordon et al. |
| 4964408 | October 1990 | Hink et al. |
| 5041187 | August 1991 | Hink et al. |
| 5069213 | December 1991 | Polczynski |
| 5111817 | May 1992 | Clark et al. |
| 5163438 | November 1992 | Gordon et al. |
| 5226417 | July 1993 | Swedlow et al. |
| 5253645 | October 1993 | Friedman et al. |
| 5253646 | October 1993 | Delpy et al. |
| 5319355 | June 1994 | Russek |
| 5319363 | June 1994 | Welch |
| 5337744 | August 1994 | Branigan |
| 5341805 | August 1994 | Stavridi et al. |
| D353195 | December 1994 | Savage et al. |
| D353196 | December 1994 | Savage et al. |
| 5377676 | January 1995 | Vari et al. |
| D359546 | June 1995 | Savage et al. |
| 5431170 | July 1995 | Mathews |
| D361840 | August 1995 | Savage et al. |
| D362063 | September 1995 | Savage et al. |
| 5452717 | September 1995 | Branigan et al. |
| D363120 | October 1995 | Savage et al. |
| 5456252 | October 1995 | Vari et al. |
| 5479934 | January 1996 | Imran |
| 5482036 | January 1996 | Diab et al. |
| 5490505 | February 1996 | Diab et al. |
| 5494043 | February 1996 | O'Sullivan et al. |
| 5533511 | July 1996 | Kaspari et al. |
| 5534851 | July 1996 | Russek |
| 5561275 | October 1996 | Savage et al. |
| 5562002 | October 1996 | Lalin |
| 5590649 | January 1997 | Caro et al. |
| 5602924 | February 1997 | Durand et al. |
| 5632272 | May 1997 | Diab et al. |
| 5638816 | June 1997 | Kiani-Azarbayjany et al. |
| 5638818 | June 1997 | Diab et al. |
| 5645440 | July 1997 | Tobler et al. |
| 5685299 | November 1997 | Diab et al. |
| D393830 | April 1998 | Tobler et al. |
| 5743262 | April 1998 | Lepper, Jr. et al. |
| 5758644 | June 1998 | Diab et al. |
| 5760910 | June 1998 | Lepper, Jr. et al. |
| 5769785 | June 1998 | Diab et al. |
| 5782757 | July 1998 | Diab et al. |
| 5785659 | July 1998 | Caro et al. |
| 5791347 | August 1998 | Flaherty et al. |
| 5810734 | September 1998 | Caro et al. |
| 5823950 | October 1998 | Diab et al. |
| 5830131 | November 1998 | Caro et al. |
| 5833618 | November 1998 | Caro et al. |
| 5860919 | January 1999 | Kiani-Azarbayjany et al. |
| 5876348 | March 1999 | Sugo et al. |
| 5890929 | April 1999 | Mills et al. |
| 5904654 | May 1999 | Wohltmann et al. |
| 5919134 | July 1999 | Diab |
| 5934925 | August 1999 | Tobler et al. |
| 5940182 | August 1999 | Lepper, Jr. et al. |
| 5995855 | November 1999 | Kiani et al. |
| 5997343 | December 1999 | Mills et al. |
| 6002952 | December 1999 | Diab et al. |
| 6005658 | December 1999 | Kaluza et al. |
| 6011986 | January 2000 | Diab et al. |
| 6027452 | February 2000 | Flaherty et al. |
| 6036642 | March 2000 | Diab et al. |
| 6045509 | April 2000 | Caro et al. |
| 6050951 | April 2000 | Friedman et al. |
| 6067462 | May 2000 | Diab et al. |
| 6081735 | June 2000 | Diab et al. |
| 6088607 | July 2000 | Diab et al. |
| 6110522 | August 2000 | Lepper, Jr. et al. |
| 6124597 | September 2000 | Shehada |
| 6128521 | October 2000 | Marro et al. |
| 6129675 | October 2000 | Jay |
| 6144868 | November 2000 | Parker |
| 6151516 | November 2000 | Kiani-Azarbayjany et al. |
| 6152754 | November 2000 | Gerhardt et al. |
| 6157850 | December 2000 | Diab et al. |
| 6165005 | December 2000 | Mills et al. |
| 6184521 | February 2001 | Coffin, IV et al. |
| 6190325 | February 2001 | Narimatsu |
| 6206830 | March 2001 | Diab et al. |
| 6229856 | May 2001 | Diab et al. |
| 6232609 | May 2001 | Snyder et al. |
| 6236872 | May 2001 | Diab et al. |
| 6241683 | June 2001 | Macklem et al. |
| 6253097 | June 2001 | Aronow et al. |
| 6256523 | July 2001 | Diab et al. |
| 6263222 | July 2001 | Diab et al. |
| 6278522 | August 2001 | Lepper, Jr. et al. |
| 6280213 | August 2001 | Tobler et al. |
| 6285896 | September 2001 | Tobler et al. |
| 6301493 | October 2001 | Marro et al. |
| 6317627 | November 2001 | Ennen et al. |
| 6321100 | November 2001 | Parker |
| 6325761 | December 2001 | Jay |
| 6334065 | December 2001 | Al-Ali et al. |
| 6343224 | January 2002 | Parker |
| 6349228 | February 2002 | Kiani et al. |
| 6360114 | March 2002 | Diab et al. |
| 6368283 | April 2002 | Xu et al. |
| 6371921 | April 2002 | Caro et al. |
| 6377829 | April 2002 | Al-Ali |
| 6388240 | May 2002 | Schulz et al. |
| 6397091 | May 2002 | Diab et al. |
| 6430437 | August 2002 | Marro |
| 6430525 | August 2002 | Weber et al. |
| 6463311 | October 2002 | Diab |
| 6470199 | October 2002 | Kopotic et al. |
| 6501975 | December 2002 | Diab et al. |
| 6505059 | January 2003 | Kollias et al. |
| 6515273 | February 2003 | Al-Ali |
| 6519487 | February 2003 | Parker |
| 6525386 | February 2003 | Mills et al. |
| 6526300 | February 2003 | Kiani et al. |
| 6541756 | April 2003 | Schulz et al. |
| 6542764 | April 2003 | Al-Ali et al. |
| 6553242 | April 2003 | Sarussi |
| 6580086 | June 2003 | Schulz et al. |
| 6584336 | June 2003 | Ali et al. |
| 6595316 | July 2003 | Cybulski et al. |
| 6597932 | July 2003 | Tian et al. |
| 6597933 | July 2003 | Kiani et al. |
| 6606511 | August 2003 | Ali et al. |
| 6632181 | October 2003 | Flaherty et al. |
| 6639668 | October 2003 | Trepagnier |
| 6640116 | October 2003 | Diab |
| 6643530 | November 2003 | Diab et al. |
| 6645155 | November 2003 | Inukai et al. |
| 6650917 | November 2003 | Diab et al. |
| 6654624 | November 2003 | Diab et al. |
| 6658276 | December 2003 | Kianl et al. |
| 6661161 | December 2003 | Lanzo et al. |
| 6671531 | December 2003 | Al-Ali et al. |
| 6678543 | January 2004 | Diab et al. |
| 6684090 | January 2004 | Ali et al. |
| 6684091 | January 2004 | Parker |
| 6697656 | February 2004 | Al-Ali |
| 6697657 | February 2004 | Shehada et al. |
| 6697658 | February 2004 | Al-Ali |
| RE38476 | March 2004 | Diab et al. |
| 6699194 | March 2004 | Diab et al. |
| 6714804 | March 2004 | Al-Ali et al. |
| RE38492 | April 2004 | Diab et al. |
| 6721582 | April 2004 | Trepagnier et al. |
| 6721585 | April 2004 | Parker |
| 6725075 | April 2004 | Al-Ali |
| 6728560 | April 2004 | Kollias et al. |
| 6735459 | May 2004 | Parker |
| 6745060 | June 2004 | Diab et al. |
| 6754516 | June 2004 | Mannheimer |
| 6760607 | July 2004 | Al-Ali |
| 6770028 | August 2004 | Ali et al. |
| 6771994 | August 2004 | Kiani et al. |
| 6792300 | September 2004 | Diab et al. |
| 6813511 | November 2004 | Diab et al. |
| 6816741 | November 2004 | Diab |
| 6822564 | November 2004 | Al-Ali |
| 6826419 | November 2004 | Diab et al. |
| 6830711 | December 2004 | Mills et al. |
| 6850787 | February 2005 | Weber et al. |
| 6850788 | February 2005 | Al-Ali |
| 6852083 | February 2005 | Caro et al. |
| 6861639 | March 2005 | Al-Ali |
| 6898452 | May 2005 | Al-Ali et al. |
| 6920345 | July 2005 | Al-Ali et al. |
| 6931268 | August 2005 | Kiani-Azarbayjany et al. |
| 6934570 | August 2005 | Kiani et al. |
| 6939305 | September 2005 | Flaherty et al. |
| 6943348 | September 2005 | Coffin, IV |
| 6950687 | September 2005 | Al-Ali |
| 6961598 | November 2005 | Diab |
| 6970792 | November 2005 | Diab |
| 6979812 | December 2005 | Al-Ali |
| 6985764 | January 2006 | Mason et al. |
| 6993371 | January 2006 | Kiani et al. |
| 6996427 | February 2006 | Ali et al. |
| 6999904 | February 2006 | Weber et al. |
| 7003338 | February 2006 | Weber et al. |
| 7003339 | February 2006 | Diab et al. |
| 7015451 | March 2006 | Dalke et al. |
| 7024233 | April 2006 | Ali et al. |
| 7027849 | April 2006 | Al-Ali |
| 7030749 | April 2006 | Al-Ali |
| 7039449 | May 2006 | Al-Ali |
| 7041060 | May 2006 | Flaherty et al. |
| 7044918 | May 2006 | Diab |
| 7067893 | June 2006 | Mills et al. |
| 7096052 | August 2006 | Mason et al. |
| 7096054 | August 2006 | Abdul-Hafiz et al. |
| 7123950 | October 2006 | Mannheimer |
| 7132641 | November 2006 | Schulz et al. |
| 7142901 | November 2006 | Kiani et al. |
| 7149561 | December 2006 | Diab |
| 7186966 | March 2007 | Al-Ali |
| 7190261 | March 2007 | Al-Ali |
| 7215984 | May 2007 | Diab |
| 7215986 | May 2007 | Diab |
| 7221971 | May 2007 | Diab |
| 7225006 | May 2007 | Al-Ali et al. |
| 7225007 | May 2007 | Al-Ali |
| RE39672 | June 2007 | Shehada et al. |
| 7239905 | July 2007 | Kiani-Azarbayjany et al. |
| 7245953 | July 2007 | Parker |
| 7254429 | August 2007 | Schurman et al. |
| 7254431 | August 2007 | Al-Ali |
| 7254433 | August 2007 | Diab et al. |
| 7254434 | August 2007 | Schulz et al. |
| 7272425 | September 2007 | Al-Ali |
| 7274955 | September 2007 | Kiani et al. |
| D554263 | October 2007 | Al-Ali |
| 7280858 | October 2007 | Al-Ali et al. |
| 7289835 | October 2007 | Mansfield et al. |
| 7292883 | November 2007 | De Felice et al. |
| 7295866 | November 2007 | Al-Ali |
| 7328053 | February 2008 | Diab et al. |
| 7332784 | February 2008 | Mills et al. |
| 7340287 | March 2008 | Mason et al. |
| 7341559 | March 2008 | Schulz et al. |
| 7343186 | March 2008 | Lamego et al. |
| D566282 | April 2008 | Al-Ali et al. |
| 7355512 | April 2008 | Al-Ali |
| 7356365 | April 2008 | Schurman |
| 7371981 | May 2008 | Abdul-Hafiz |
| 7373193 | May 2008 | Al-Ali et al. |
| 7373194 | May 2008 | Weber et al. |
| 7376453 | May 2008 | Diab et al. |
| 7377794 | May 2008 | Al-Ali et al. |
| 7377899 | May 2008 | Weber et al. |
| 7383070 | June 2008 | Diab et al. |
| 7415297 | August 2008 | Al-Ali et al. |
| 7428432 | September 2008 | Ali et al. |
| 7438683 | October 2008 | Al-Ali et al. |
| 7440787 | October 2008 | Diab |
| 7454240 | November 2008 | Diab et al. |
| 7467002 | December 2008 | Weber et al. |
| 7469157 | December 2008 | Diab et al. |
| 7471969 | December 2008 | Diab et al. |
| 7471971 | December 2008 | Diab et al. |
| 7483729 | January 2009 | Al-Ali et al. |
| 7483730 | January 2009 | Diab et al. |
| 7489958 | February 2009 | Diab et al. |
| 7496391 | February 2009 | Diab et al. |
| 7496393 | February 2009 | Diab et al. |
| D587657 | March 2009 | Al-Ali et al. |
| 7499741 | March 2009 | Diab et al. |
| 7499835 | March 2009 | Weber et al. |
| 7500950 | March 2009 | Al-Ali et al. |
| 7509154 | March 2009 | Diab et al. |
| 7509494 | March 2009 | Al-Ali |
| 7510849 | March 2009 | Schurman et al. |
| 7526328 | April 2009 | Diab et al. |
| 7530942 | May 2009 | Diab |
| 7530949 | May 2009 | Al Ali et al. |
| 7530955 | May 2009 | Diab et al. |
| 7563110 | July 2009 | Al-Ali et al. |
| 7596398 | September 2009 | Al-Ali et al. |
| 7618375 | November 2009 | Flaherty et al. |
| D606659 | December 2009 | Kiani et al. |
| 7647083 | January 2010 | Al-Ali et al. |
| D609193 | February 2010 | Al-Ali et al. |
| D614305 | April 2010 | Al-Ali et al. |
| RE41317 | May 2010 | Parker |
| 7729733 | June 2010 | Al-Ali et al. |
| 7734320 | June 2010 | Al-Ali |
| 7761127 | July 2010 | Al-Ali et al. |
| 7761128 | July 2010 | Al-Ali et al. |
| 7764982 | July 2010 | Dalke et al. |
| D621516 | August 2010 | Kiani et al. |
| 7791155 | September 2010 | Diab |
| 7801581 | September 2010 | Diab |
| 7822452 | October 2010 | Schurman et al. |
| RE41912 | November 2010 | Parker |
| 7844313 | November 2010 | Kiani et al. |
| 7844314 | November 2010 | Al-Ali |
| 7844315 | November 2010 | Al-Ali |
| 7865222 | January 2011 | Weber et al. |
| 7873497 | January 2011 | Weber et al. |
| 7880606 | February 2011 | Al-Ali |
| 7880626 | February 2011 | Al-Ali et al. |
| 7891355 | February 2011 | Al-Ali et al. |
| 7894868 | February 2011 | Al-Ali et al. |
| 7899507 | March 2011 | Al-Ali et al. |
| 7899518 | March 2011 | Trepagnier et al. |
| 7904132 | March 2011 | Weber et al. |
| 7909772 | March 2011 | Popov et al. |
| 7910875 | March 2011 | Al-Ali |
| 7919713 | April 2011 | Al-Ali et al. |
| 7937128 | May 2011 | Al-Ali |
| 7937129 | May 2011 | Mason et al. |
| 7937130 | May 2011 | Diab et al. |
| 7941199 | May 2011 | Kiani |
| 7951086 | May 2011 | Flaherty et al. |
| 7957780 | June 2011 | Lamego et al. |
| 7962188 | June 2011 | Kiani et al. |
| 7962190 | June 2011 | Diab et al. |
| 7976472 | July 2011 | Kiani |
| 7988637 | August 2011 | Diab |
| 7990382 | August 2011 | Kiani |
| 7991446 | August 2011 | Ali et al. |
| 8000761 | August 2011 | Al-Ali |
| 8008088 | August 2011 | Bellott et al. |
| RE42753 | September 2011 | Kiani-Azarbayjany et al. |
| 8019400 | September 2011 | Diab et al. |
| 8028701 | October 2011 | Al-Ali et al. |
| 8029765 | October 2011 | Bellott et al. |
| 8036727 | October 2011 | Schurman et al. |
| 8036728 | October 2011 | Diab et al. |
| 8046040 | October 2011 | Ali et al. |
| 8046041 | October 2011 | Diab et al. |
| 8046042 | October 2011 | Diab et al. |
| 8048040 | November 2011 | Kiani |
| 8050728 | November 2011 | Al-Ali et al. |
| RE43169 | February 2012 | Parker |
| 8118620 | February 2012 | Al-Ali et al. |
| 8126528 | February 2012 | Diab et al. |
| 8128572 | March 2012 | Diab et al. |
| 8130105 | March 2012 | Al-Ali et al. |
| 8145287 | March 2012 | Diab et al. |
| 8150487 | April 2012 | Diab et al. |
| 8175672 | May 2012 | Parker |
| 8180420 | May 2012 | Diab et al. |
| 8182443 | May 2012 | Kiani |
| 8185180 | May 2012 | Diab et al. |
| 8190223 | May 2012 | Al-Ali et al. |
| 8190227 | May 2012 | Diab et al. |
| 8203438 | June 2012 | Kiani et al. |
| 8203704 | June 2012 | Merritt et al. |
| 8204566 | June 2012 | Schurman et al. |
| 8219172 | July 2012 | Schurman et al. |
| 8224411 | July 2012 | Al-Ali et al. |
| 8228181 | July 2012 | Al-Ali |
| 8229533 | July 2012 | Diab et al. |
| 8233955 | July 2012 | Al-Ali et al. |
| 8244325 | August 2012 | Al-Ali et al. |
| 8255026 | August 2012 | Al-Ali |
| 8255027 | August 2012 | Al-Ali et al. |
| 8255028 | August 2012 | Al-Ali et al. |
| 8260577 | September 2012 | Weber et al. |
| 8265723 | September 2012 | McHale et al. |
| 8274360 | September 2012 | Sampath et al. |
| 8301217 | October 2012 | Al-Ali et al. |
| 8306596 | November 2012 | Schurman et al. |
| 8310336 | November 2012 | Muhsin et al. |
| 8315683 | November 2012 | Al-Ali et al. |
| RE43860 | December 2012 | Parker |
| 8337403 | December 2012 | Al-Ali et al. |
| 8346330 | January 2013 | Lamego |
| 8353842 | January 2013 | Al-Ali et al. |
| 8355766 | January 2013 | MacNeish, III et al. |
| 8359080 | January 2013 | Diab et al. |
| 8364223 | January 2013 | Al-Ali et al. |
| 8364226 | January 2013 | Diab et al. |
| 8374665 | February 2013 | Lamego |
| 8385995 | February 2013 | Al-Ali et al. |
| 8385996 | February 2013 | Smith et al. |
| 8388353 | March 2013 | Kiani et al. |
| 8399822 | March 2013 | Al-Ali |
| 8401602 | March 2013 | Kiani |
| 8401607 | March 2013 | Mannheimer |
| 8405608 | March 2013 | Al-Ali et al. |
| 8414499 | April 2013 | Al-Ali et al. |
| 8418524 | April 2013 | Al-Ali |
| 8423106 | April 2013 | Lamego et al. |
| 8428967 | April 2013 | Olsen et al. |
| 8430817 | April 2013 | Al-Ali et al. |
| 8437825 | May 2013 | Dalvi et al. |
| 8455290 | June 2013 | Siskavich |
| 8457703 | June 2013 | Al-Ali |
| 8457707 | June 2013 | Kiani |
| 8463349 | June 2013 | Diab et al. |
| 8466286 | June 2013 | Bellott et al. |
| 8471713 | June 2013 | Poeze et al. |
| 8473020 | June 2013 | Kiani et al. |
| 8483787 | July 2013 | Al-Ali et al. |
| 8489364 | July 2013 | Weber et al. |
| 8498684 | July 2013 | Weber et al. |
| 8504128 | August 2013 | Blank et al. |
| 8509867 | August 2013 | Workman et al. |
| 8515509 | August 2013 | Bruinsma et al. |
| 8523781 | September 2013 | Al-Ali |
| 8529301 | September 2013 | Al-Ali et al. |
| 8532727 | September 2013 | Ali et al. |
| 8532728 | September 2013 | Diab et al. |
| D692145 | October 2013 | Al-Ali et al. |
| 8547209 | October 2013 | Kiani et al. |
| 8548548 | October 2013 | Al-Ali |
| 8548549 | October 2013 | Schurman et al. |
| 8548550 | October 2013 | Al-Ali et al. |
| 8554297 | October 2013 | Moon et al. |
| 8560032 | October 2013 | Al-Ali et al. |
| 8560034 | October 2013 | Diab et al. |
| 8570167 | October 2013 | Al-Ali |
| 8570503 | October 2013 | Vo et al. |
| 8571617 | October 2013 | Reichgott et al. |
| 8571618 | October 2013 | Lamego et al. |
| 8571619 | October 2013 | Al-Ali et al. |
| 8577431 | November 2013 | Lamego et al. |
| 8581732 | November 2013 | Al-Ali et al. |
| 8584345 | November 2013 | Al-Ali et al. |
| 8588880 | November 2013 | Abdul-Hafiz et al. |
| 8600467 | December 2013 | Al-Ali et al. |
| 8606342 | December 2013 | Diab |
| 8626255 | January 2014 | Al-Ali et al. |
| 8630691 | January 2014 | Lamego et al. |
| 8634889 | January 2014 | Al-Ali et al. |
| 8641631 | February 2014 | Sierra et al. |
| 8652060 | February 2014 | Al-Ali |
| 8663107 | March 2014 | Kiani |
| 8666468 | March 2014 | Al-Ali |
| 8667967 | March 2014 | Al-Ali et al. |
| 8670811 | March 2014 | O'Reilly |
| 8670814 | March 2014 | Diab et al. |
| 8676286 | March 2014 | Weber et al. |
| 8682407 | March 2014 | Al-Ali |
| RE44823 | April 2014 | Parker |
| RE44875 | April 2014 | Kiani et al. |
| 8690799 | April 2014 | Telfort et al. |
| 8700112 | April 2014 | Kiani |
| 8702627 | April 2014 | Telfort et al. |
| 8706179 | April 2014 | Parker |
| 8712494 | April 2014 | MacNeish, III et al. |
| 8715206 | May 2014 | Telfort et al. |
| 8718735 | May 2014 | Lamego et al. |
| 8718737 | May 2014 | Diab et al. |
| 8718738 | May 2014 | Blank et al. |
| 8720249 | May 2014 | Al-Ali |
| 8721541 | May 2014 | Al-Ali et al. |
| 8721542 | May 2014 | Al-Ali et al. |
| 8723677 | May 2014 | Kiani |
| 8740792 | June 2014 | Kiani et al. |
| 8754776 | June 2014 | Poeze et al. |
| 8755535 | June 2014 | Telfort et al. |
| 8755856 | June 2014 | Diab et al. |
| 8755872 | June 2014 | Marinow |
| 8761850 | June 2014 | Lamego |
| 8764671 | July 2014 | Kiani |
| 8768423 | July 2014 | Shakespeare et al. |
| 8771204 | July 2014 | Telfort et al. |
| 8777634 | July 2014 | Kiani et al. |
| 8781543 | July 2014 | Diab et al. |
| 8781544 | July 2014 | Al-Ali et al. |
| 8781549 | July 2014 | Al-Ali et al. |
| 8788003 | July 2014 | Schurman et al. |
| 8790268 | July 2014 | Al-Ali |
| 8801613 | August 2014 | Al-Ali et al. |
| 8821397 | September 2014 | Al-Ali et al. |
| 8821415 | September 2014 | Al-Ali et al. |
| 8830449 | September 2014 | Lamego et al. |
| 8831700 | September 2014 | Schurman et al. |
| 8838196 | September 2014 | Mannheimer |
| 8840549 | September 2014 | Al-Ali et al. |
| 8847740 | September 2014 | Kiani et al. |
| 8849365 | September 2014 | Smith et al. |
| 8852094 | October 2014 | Al-Ali et al. |
| 8852115 | October 2014 | Muir |
| 8852994 | October 2014 | Wojtczuk et al. |
| 8868147 | October 2014 | Stippick et al. |
| 8868150 | October 2014 | Al-Ali et al. |
| 8870792 | October 2014 | Al-Ali et al. |
| 8886271 | November 2014 | Kiani et al. |
| 8888539 | November 2014 | Al-Ali et al. |
| 8888708 | November 2014 | Diab et al. |
| 8892180 | November 2014 | Weber et al. |
| 8897847 | November 2014 | Al-Ali |
| 8909310 | December 2014 | Lamego et al. |
| 8911377 | December 2014 | Al-Ali |
| 8912909 | December 2014 | Al-Ali et al. |
| 8920317 | December 2014 | Al-Ali et al. |
| 8921699 | December 2014 | Al-Ali et al. |
| 8922382 | December 2014 | Al-Ali et al. |
| 8929964 | January 2015 | Al-Ali et al. |
| 8942777 | January 2015 | Diab et al. |
| 8948834 | February 2015 | Diab et al. |
| 8948835 | February 2015 | Diab |
| 8956294 | February 2015 | McCombie et al. |
| 8965471 | February 2015 | Lamego |
| 8983564 | March 2015 | Al-Ali |
| 8989831 | March 2015 | Al-Ali et al. |
| 8996085 | March 2015 | Kiani et al. |
| 8998809 | April 2015 | Kiani |
| 9028407 | May 2015 | Bennett-Guerrero |
| 9028429 | May 2015 | Telfort et al. |
| 9037207 | May 2015 | Al-Ali et al. |
| 9060721 | June 2015 | Reichgott et al. |
| 9066666 | June 2015 | Kiani |
| 9066680 | June 2015 | Al-Ali et al. |
| 9072474 | July 2015 | Al-Ali et al. |
| 9078560 | July 2015 | Schurman et al. |
| 9084569 | July 2015 | Weber et al. |
| 9095316 | August 2015 | Welch et al. |
| 9106038 | August 2015 | Telfort et al. |
| 9107625 | August 2015 | Telfort et al. |
| 9107626 | August 2015 | Al-Ali et al. |
| 9113831 | August 2015 | Al-Ali |
| 9113832 | August 2015 | Al-Ali |
| 9119595 | September 2015 | Lamego |
| 9131881 | September 2015 | Diab et al. |
| 9131882 | September 2015 | Al-Ali et al. |
| 9131883 | September 2015 | Al-Ali |
| 9131917 | September 2015 | Telfort et al. |
| 9138180 | September 2015 | Coverston et al. |
| 9138182 | September 2015 | Al-Ali et al. |
| 9138192 | September 2015 | Weber et al. |
| 9142117 | September 2015 | Muhsin et al. |
| 9153112 | October 2015 | Kiani et al. |
| 9153121 | October 2015 | Kiani et al. |
| 9161696 | October 2015 | Al-Ali et al. |
| 9161713 | October 2015 | Al-Ali et al. |
| 9167995 | October 2015 | Lamego et al. |
| 9176141 | November 2015 | Al-Ali et al. |
| 9186102 | November 2015 | Bruinsma et al. |
| 9192312 | November 2015 | Al-Ali |
| 9192329 | November 2015 | Al-Ali |
| 9192351 | November 2015 | Telfort et al. |
| 9195385 | November 2015 | Al-Ali et al. |
| 9211072 | December 2015 | Kiani |
| 9211095 | December 2015 | Al-Ali |
| 9218454 | December 2015 | Kiani et al. |
| 9226696 | January 2016 | Kiani |
| 9241662 | January 2016 | Al-Ali et al. |
| 9245668 | January 2016 | Vo et al. |
| 9259185 | February 2016 | Abdul-Hafiz et al. |
| 9267572 | February 2016 | Barker et al. |
| 9277880 | March 2016 | Poeze et al. |
| 9289167 | March 2016 | Diab et al. |
| 9295421 | March 2016 | Kiani et al. |
| 9307928 | April 2016 | Al-Ali et al. |
| 9323894 | April 2016 | Kiani |
| D755392 | May 2016 | Hwang et al. |
| 9326712 | May 2016 | Kiani |
| 9333316 | May 2016 | Kiani |
| 9339220 | May 2016 | Lamego et al. |
| 9341565 | May 2016 | Lamego et al. |
| 9351673 | May 2016 | Diab et al. |
| 9351675 | May 2016 | Al-Ali et al. |
| 9364181 | June 2016 | Kiani et al. |
| 9368671 | June 2016 | Wojtczuk et al. |
| 9370325 | June 2016 | Al-Ali et al. |
| 9370326 | June 2016 | McHale et al. |
| 9370335 | June 2016 | Al-ali et al. |
| 9375185 | June 2016 | Ali et al. |
| 9386953 | July 2016 | Al-Ali |
| 9386961 | July 2016 | Al-Ali et al. |
| 9392945 | July 2016 | Al-Ali et al. |
| 9397448 | July 2016 | Al-Ali et al. |
| 9408542 | August 2016 | Kinast et al. |
| 9436645 | September 2016 | Al-Ali et al. |
| 9445759 | September 2016 | Lamego et al. |
| 9466919 | October 2016 | Kiani et al. |
| 9474474 | October 2016 | Lamego et al. |
| 9480422 | November 2016 | Al-Ali |
| 9480435 | November 2016 | Olsen |
| 9492110 | November 2016 | Al-Ali et al. |
| 9510779 | December 2016 | Poeze et al. |
| 9517024 | December 2016 | Kiani et al. |
| 9532722 | January 2017 | Lamego et al. |
| 9538949 | January 2017 | Al-Ali et al. |
| 9538980 | January 2017 | Telfort et al. |
| 9549696 | January 2017 | Lamego et al. |
| 9554737 | January 2017 | Schurman et al. |
| 9560996 | February 2017 | Kiani |
| 9560998 | February 2017 | Al-Ali et al. |
| 9566019 | February 2017 | Al-Ali et al. |
| 9579039 | February 2017 | Jansen et al. |
| 9591975 | March 2017 | Dalvi et al. |
| 9622693 | April 2017 | Diab |
| 2002/0161291 | October 2002 | Kianl et al. |
| 2003/0137423 | July 2003 | Al-Ali |
| 2003/0191358 | October 2003 | MacKin et al. |
| 2004/0162499 | August 2004 | Nagai et al. |
| 2005/0027182 | February 2005 | Siddiqui |
| 2005/0177096 | August 2005 | Bollish |
| 2005/0240091 | October 2005 | Lynn |
| 2006/0220881 | October 2006 | Al-Ali |
| 2007/0040692 | February 2007 | Smith et al. |
| 2007/0282478 | December 2007 | Al-Ali et al. |
| 2008/0103375 | May 2008 | Kiani |
| 2008/0244425 | October 2008 | Kikin-Gil et al. |
| 2008/0255438 | October 2008 | Saidara |
| 2008/0281168 | November 2008 | Gibson |
| 2009/0040874 | February 2009 | Rooney |
| 2009/0247848 | October 2009 | Baker |
| 2009/0247849 | October 2009 | McCutcheon et al. |
| 2009/0247984 | October 2009 | Lamego et al. |
| 2009/0275813 | November 2009 | Davis |
| 2009/0275844 | November 2009 | Al-Ali |
| 2010/0004518 | January 2010 | Vo et al. |
| 2010/0030040 | February 2010 | Poeze et al. |
| 2010/0113904 | May 2010 | Batchelder et al. |
| 2010/0331656 | December 2010 | Mensinger et al. |
| 2011/0082711 | April 2011 | Poeze et al. |
| 2011/0105854 | May 2011 | Kiani et al. |
| 2011/0125060 | May 2011 | Telfort et al. |
| 2011/0208015 | August 2011 | Welch et al. |
| 2011/0213212 | September 2011 | Al-Ali |
| 2011/0230733 | September 2011 | Al-Ali |
| 2011/0237969 | September 2011 | Eckerbom et al. |
| 2011/0288383 | November 2011 | Diab |
| 2012/0041316 | February 2012 | Al-Ali et al. |
| 2012/0046557 | February 2012 | Kiani |
| 2012/0059267 | March 2012 | Lamego et al. |
| 2012/0088984 | April 2012 | Al-Ali et al. |
| 2012/0105233 | May 2012 | Bobey et al. |
| 2012/0165629 | June 2012 | Merritt et al. |
| 2012/0209082 | August 2012 | Al-Ali |
| 2012/0209084 | August 2012 | Olsen et al. |
| 2012/0232366 | September 2012 | Kiani |
| 2012/0283524 | November 2012 | Kiani et al. |
| 2012/0296178 | November 2012 | Lamego et al. |
| 2012/0303085 | November 2012 | Vitense et al. |
| 2012/0319816 | December 2012 | Al-Ali |
| 2013/0023775 | January 2013 | Lamego et al. |
| 2013/0041591 | February 2013 | Lamego |
| 2013/0046204 | February 2013 | Lamego et al. |
| 2013/0060147 | March 2013 | Welch et al. |
| 2013/0096405 | April 2013 | Garfio |
| 2013/0096936 | April 2013 | Sampath et al. |
| 2013/0243021 | September 2013 | Siskavich |
| 2013/0253334 | September 2013 | Al-Ali et al. |
| 2013/0267804 | October 2013 | Al-Ali |
| 2013/0274572 | October 2013 | Al-Ali et al. |
| 2013/0296672 | November 2013 | O'Neil et al. |
| 2013/0296713 | November 2013 | Al-Ali et al. |
| 2013/0324808 | December 2013 | Al-Ali et al. |
| 2013/0331660 | December 2013 | Al-Ali et al. |
| 2013/0331670 | December 2013 | Kiani |
| 2014/0012100 | January 2014 | Al-Ali et al. |
| 2014/0034353 | February 2014 | Al-Ali et al. |
| 2014/0051953 | February 2014 | Lamego et al. |
| 2014/0066783 | March 2014 | Kiani et al. |
| 2014/0077956 | March 2014 | Sampath et al. |
| 2014/0081100 | March 2014 | Muhsin et al. |
| 2014/0081175 | March 2014 | Telfort |
| 2014/0100434 | April 2014 | Diab et al. |
| 2014/0114199 | April 2014 | Lamego et al. |
| 2014/0120564 | May 2014 | Workman et al. |
| 2014/0121482 | May 2014 | Merritt et al. |
| 2014/0127137 | May 2014 | Bellott et al. |
| 2014/0129702 | May 2014 | Lamego et al. |
| 2014/0135588 | May 2014 | Al-Ali et al. |
| 2014/0142401 | May 2014 | Al-Ali et al. |
| 2014/0163344 | June 2014 | Al-Ali |
| 2014/0163402 | June 2014 | Lamego et al. |
| 2014/0166076 | June 2014 | Kiani et al. |
| 2014/0171763 | June 2014 | Diab |
| 2014/0180038 | June 2014 | Kiani |
| 2014/0180154 | June 2014 | Sierra et al. |
| 2014/0180160 | June 2014 | Brown et al. |
| 2014/0187973 | July 2014 | Brown et al. |
| 2014/0213864 | July 2014 | Abdul-Hafiz et al. |
| 2014/0266790 | September 2014 | Al-Ali et al. |
| 2014/0275808 | September 2014 | Poeze et al. |
| 2014/0275835 | September 2014 | Lamego et al. |
| 2014/0275871 | September 2014 | Lamego et al. |
| 2014/0275872 | September 2014 | Merritt et al. |
| 2014/0276115 | September 2014 | Dalvi et al. |
| 2014/0288400 | September 2014 | Diab et al. |
| 2014/0309507 | October 2014 | Baker, Jr. |
| 2014/0316217 | October 2014 | Purdon et al. |
| 2014/0316218 | October 2014 | Purdon et al. |
| 2014/0316228 | October 2014 | Blank et al. |
| 2014/0323825 | October 2014 | Al-Ali et al. |
| 2014/0323897 | October 2014 | Brown et al. |
| 2014/0323898 | October 2014 | Purdon et al. |
| 2014/0330092 | November 2014 | Al-Ali et al. |
| 2014/0330098 | November 2014 | Merritt et al. |
| 2014/0330099 | November 2014 | Al-Ali et al. |
| 2014/0336481 | November 2014 | Shakespeare et al. |
| 2014/0357966 | December 2014 | Al-Ali et al. |
| 2015/0005600 | January 2015 | Blank et al. |
| 2015/0011907 | January 2015 | Purdon et al. |
| 2015/0012231 | January 2015 | Poeze et al. |
| 2015/0025406 | January 2015 | Al-Ali |
| 2015/0032029 | January 2015 | Al-Ali et al. |
| 2015/0038859 | February 2015 | Dalvi et al. |
| 2015/0045637 | February 2015 | Dalvi |
| 2015/0051462 | February 2015 | Olsen |
| 2015/0080754 | March 2015 | Purdon et al. |
| 2015/0087936 | March 2015 | Al-Ali et al. |
| 2015/0094546 | April 2015 | Al-Ali |
| 2015/0097701 | April 2015 | Al-Ali et al. |
| 2015/0099950 | April 2015 | Al-Ali et al. |
| 2015/0099951 | April 2015 | Al-Ali et al. |
| 2015/0099955 | April 2015 | Al-Ali et al. |
| 2015/0101844 | April 2015 | Al-Ali et al. |
| 2015/0106121 | April 2015 | Muhsin et al. |
| 2015/0112151 | April 2015 | Muhsin et al. |
| 2015/0116076 | April 2015 | Al-Ali et al. |
| 2015/0126830 | May 2015 | Schurman et al. |
| 2015/0133755 | May 2015 | Smith et al. |
| 2015/0141781 | May 2015 | Weber et al. |
| 2015/0165312 | June 2015 | Kiani |
| 2015/0196237 | July 2015 | Lamego |
| 2015/0216459 | August 2015 | Al-Ali et al. |
| 2015/0230755 | August 2015 | Al-Ali et al. |
| 2015/0233722 | August 2015 | Al-Ali |
| 2015/0245773 | September 2015 | Lamego et al. |
| 2015/0245794 | September 2015 | Al-Ali |
| 2015/0257689 | September 2015 | Al-Ali et al. |
| 2015/0272514 | October 2015 | Kiani et al. |
| 2015/0351697 | December 2015 | Weber et al. |
| 2015/0351704 | December 2015 | Kiani et al. |
| 2015/0359429 | December 2015 | Al-Ali et al. |
| 2015/0366472 | December 2015 | Kiani |
| 2015/0366507 | December 2015 | Blank |
| 2015/0374298 | December 2015 | Al-Ali et al. |
| 2015/0380875 | December 2015 | Coverston et al. |
| 2016/0000362 | January 2016 | Diab et al. |
| 2016/0007930 | January 2016 | Weber et al. |
| 2016/0029932 | February 2016 | Al-Ali |
| 2016/0045118 | February 2016 | Kiani |
| 2016/0051205 | February 2016 | Al-Ali et al. |
| 2016/0058338 | March 2016 | Schurman et al. |
| 2016/0058347 | March 2016 | Reichgott et al. |
| 2016/0066823 | March 2016 | Kind et al. |
| 2016/0066824 | March 2016 | Al-Ali et al. |
| 2016/0066879 | March 2016 | Telfort et al. |
| 2016/0072429 | March 2016 | Kiani et al. |
| 2016/0081552 | March 2016 | Wojtczuk et al. |
| 2016/0095543 | April 2016 | Telfort et al. |
| 2016/0095548 | April 2016 | Al-Ali et al. |
| 2016/0103598 | April 2016 | Al-Ali et al. |
| 2016/0113527 | April 2016 | Al-Ali |
| 2016/0143548 | May 2016 | Al-Ali |
| 2016/0166182 | June 2016 | Al-Ali et al. |
| 2016/0166183 | June 2016 | Poeze et al. |
| 2016/0166188 | June 2016 | Bruinsma et al. |
| 2016/0166210 | June 2016 | Al-Ali |
| 2016/0192869 | July 2016 | Kiani et al. |
| 2016/0196388 | July 2016 | Lamego |
| 2016/0197436 | July 2016 | Barker et al. |
| 2016/0213281 | July 2016 | Eckerbom et al. |
| 2016/0228043 | August 2016 | O'Neil et al. |
| 2016/0233632 | August 2016 | Scruggs et al. |
| 2016/0234944 | August 2016 | Schmidt et al. |
| 2016/0270735 | September 2016 | Diab et al. |
| 2016/0283665 | September 2016 | Sampath et al. |
| 2016/0287090 | October 2016 | Al-Ali et al. |
| 2016/0287786 | October 2016 | Kiani |
| 2016/0296169 | October 2016 | McHale et al. |
| 2016/0310052 | October 2016 | Al-Ali et al. |
| 2016/0314260 | October 2016 | Kiani |
| 2016/0324486 | November 2016 | Al-Ali et al. |
| 2016/0324488 | November 2016 | Olsen |
| 2016/0327984 | November 2016 | Al-Ali et al. |
| 2016/0328528 | November 2016 | Al-Ali et al. |
| 2016/0331332 | November 2016 | Al-Ali |
| 2016/0367173 | December 2016 | Dalvi et al. |
| 2017/0007134 | January 2017 | Al-Ali et al. |
| 2017/0007190 | January 2017 | Al-Ali et al. |
| 2017/0007198 | January 2017 | Al-Ali et al. |
| 2017/0014084 | January 2017 | Al-Ali et al. |
| 2017/0021099 | January 2017 | Al-Ali et al. |
| 2017/0027456 | February 2017 | Kinast et al. |
| 2017/0042488 | February 2017 | Muhsin |
| 2017/0055847 | March 2017 | Kiani et al. |
| 2017/0055851 | March 2017 | Al-Ali |
| 2017/0055882 | March 2017 | Al-Ali et al. |
| 2017/0055887 | March 2017 | Al-Ali |
| 2017/0055896 | March 2017 | Al-Ali et al. |
| 2017/0079594 | March 2017 | Telfort et al. |
| 2017/0086723 | March 2017 | Al-Ali et al. |
| WO 2005/087097 | Sep 2005 | WO | |||
| WO 2010/014743 | Feb 2010 | WO | |||
Other References
|
US 9,579,050, 02/2017, Al-Ali (withdrawn) cited by applicant . Rheineck-Leyssius, MD, et al., "Advanced Pulse Oximeter Signal Processing Technology Compared to Simple Averaging. I. Effect on Frequency of Alarms in the Operating Room", Journal of Clinical Anesthesia, The Netherlands, May 1999, vol. 11, pp. 192-195. cited by applicant . Cust, AE, et al., "Alarm settings for the Marquette 8000 pulse oximeter to prevent hyperoxic and hypoxic episodes", Journal Paediatric Child Health, 1999, vol. 35, pp. 159-162. cited by applicant . Lawless, Stephen T., "Crying wolf: False alarms in a pediatric intensive care unit", Critical Care Medicine, 1994, vol. 22, No. 6, pp. 981-985. cited by applicant . Informal Expert Report of Dr. Stephen Barker, Lead Case No. 16-05968-LT11, executed Feb. 10, 2017. cited by applicant . Impact of Clinical Alarms on Patient Safety, ACCE Healthcare Technology Foundation, 2006, 20 pages. cited by applicant . Taenzer, M.D., Andreas, "Impact of Pulse Oximetry Surveillance on Rescue Events and Intensive Care Unit Transfers", The American Society of Anesthesiologists, Inc., Feb. 2010, vol. 112, No. 2, pp. 282-287. cited by applicant . Stefanescu, MD MSc, Beatrice, "Improved Filtering of Pulse Oximeter Monitoring Alarms in the Neonatal ICU: Bedside Significance", Respiratory Care, Jan. 2016, vol. 61, No. 1, pp. 85-89. cited by applicant . Rheineck-Leyssius, A.T., et al., "Influence of pulse oximeter lower alarm limit on the incidence of hypoxaemia in the recovery room", British Journal of Anaesthesia, 1997, vol. 79, pp. 460-464. cited by applicant . Rheineck-Leyssius, MD, Aart, et al., "Influence of Pulse Oximeter Settings on the Frequency of Alarms, and Detection of Hypoxemia", Journal of Clinical Monitoring and Computing, 1998, vol. 14, pp. 151-156. cited by applicant . Pan, MD., Peter, "Intraoperative Pulse Oximetry: Frequency and Distribution of Discrepant Data", Journal of Clinical Monitoring and Computing, Nov./Dec. 1994, vol. 6, pp. 491-495. cited by applicant . Graham, Kelly, et al., Monitor Alarm Fatigue: Standardizing Use of Physiological Monitors and Decreasing Nuisance Alarms, American Journal of Critical Care, Jan. 2010, vol. 19, No. 1, pp. 27-37. cited by applicant . Barker, PhD, MD, Steven, "`Motion-Resistant` Pulse Oximetry: A Comparison of New and Old Models" Department of Anesthesiology, 2002, vol. 95, pp. 967-972. cited by applicant . National Patient Safety Goals Effective Jan. 1, 2015, Hospital Accreditation Program, The Joint Commission, Jan. 2015, pp. 1-17. cited by applicant . Brostowicz, Heather, et al. Oxygen saturation monitoring in the Neonatal Intensive Care Unit (NICU): Evaluation of a new alarm management, Journal of Neonatal-Perinatal Medicine 3, 2010, pp. 135-139. cited by applicant . Kowalczyk, Liz, "Patient alarms often unheard, unheeded" The Boston Globe, Feb. 13, 2011, in 5 pages. cited by applicant . Taenzer, Andreas, et al., "Postoperative Monitoring--The Dartmouth Experience", The Official Journal of the Anesthesia Patient Safety Foundation, Spring-Summer 2012, vol. 27, No. 1, pp. 1-28. cited by applicant . Tobin, Martin, "Principles and Practice of Intensive Care Monitoring", 1998, in 30 pages. cited by applicant . Severinghaus, M.D., John, et al., Pulse Oximeter Failure Thresholds in Hypotension and Vasoconstriction, Anesthesiology, Sep. 1990, vol. 73, No. 3, pp. 532-537. cited by applicant . Pologe, Jonas A., "Pulse Oximetry: Technical Aspects of Machine Design", International Anesthesiology Clinics, Advances in Oxygen Monitoring, Fall, 1987, vol. 25, No. 3, pp. 137-153. cited by applicant . Tremper, Ph.D., Kevin et al., "Pulse Oximetry", Medical Intelligence Article, Anesthesiology, Jan. 1989, vol. 70, No. 1, pp. 98-108. cited by applicant . Bosque, PhD, Elena, "Symbiosis of nurse and machine through fuzzy logic: Improved specificity of a neonatal pulse oximeter alarm", Dec. 1995, vol. 18, Issue 2, pp. 67-75. cited by applicant . Paine, MPH, Christine, et al., "Systematic review of physiologic monitor alarm characteristics and pragmatic interventions to reduce alarm frequency" J Hosp Med., Feb. 2017, vol. 11, No. 2, pp. 136-144. cited by applicant . Top 10 Technology Hazards for 2012, Guidance Article, Nov. 2011, www.ecri.org, pp. 358-373. cited by applicant . Top 10 Technology Hazards for 2013, Health Devices, ECRI Institute, Nov. 2012, reprinted from vol. 41, Issue 11, in 25 pages. cited by applicant . Top 10 Health Technology Hazards for 2014, Health Devices, ECRI Institute, Nov. 2013, www.ecri.org/2014hazards, 1 page. cited by applicant . Masimo Advanced Alarm Performance: An Evidence-Based Approach to Reduce False Alarms and Nuisance Alarms, 2010, in 8 pages. cited by applicant . International Search Report for PCT/US2009/052146, dated Dec. 15, 2009. cited by applicant . International Preliminary Report on Patentability and Written Opinion for PCT/US2009/052146 dated Feb. 10, 2011. cited by applicant. |
Primary Examiner: Escalante; Ovidio
Attorney, Agent or Firm: Knobbe, Martens, Olson & Bear, LLP
Parent Case Text
CROSS-REFERENCE TO RELATED APPLICATIONS
This .[.application.]. is .Iadd.an application for reissue of U.S. Pat. No. 9,153,121, issued on Oct. 6, 2015 and titled "Alarm Suspend System," which is .Iaddend.a continuation of U.S. patent application Ser. No. 14/036,496, filed Sep. 25, 2013 and titled "Alarm Suspend System," which is a continuation of U.S. patent application Ser. No. 13/476,725, filed May 21, 2012 and titled "Alarm Suspend System," which is a continuation of U.S. patent application Ser. No. 12/510,982 filed Jul. 28, 2009 and titled "Alarm Suspend System," which claims priority benefit under 35 U.S.C. .sctn. 119(e) to U.S. Provisional Patent Application Ser. No. 61/084,615, filed Jul. 29, 2008, titled "Alarm Management System.Iadd.;" more than one reissue application has been filed for the reissue of U.S. Pat. No. 9,153,121, including U.S. patent application Ser. No. 15/583,948 (the present application), U.S. patent application Ser. No. 15/583,922, and U.S. patent application Ser. No. 15/583,935.Iaddend...[.".]. All of the above-referenced applications are hereby incorporated by reference herein in their entireties.
Claims
What is claimed is:
1. A physiological measurement system comprising: a .Iadd.noninvasive .Iaddend.physiological sensor .[.including: a plurality of light emitting diodes.]. configured to .[.transmit wavelengths of light onto a tissue site of a patient; and at least one detector configured to measure an indication of the wavelengths of light after attenuation by tissue of the patient and.]. .Iadd.be positioned on a patient and .Iaddend.output a signal responsive .[.of the attenuated light.]. .Iadd.to a physiological condition of the patient.Iaddend.; and one or more processors in communication with the .Iadd.noninvasive .Iaddend.physiological sensor, the one or more processors configured to .Iadd.electronically.Iaddend.: determine a measurement of a physiological parameter based at least in part upon the signal; .Iadd.determine whether an alarm condition exists by determining whether an activation threshold has been satisfied by the measurement of the physiological parameter;.Iaddend. .[.receive, from a user, an indication of.]. .Iadd.access an alarm hold initiator for .Iaddend.a parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time corresponding to the physiological parameter, the parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time being .[.selected from.]. .Iadd.one of .Iaddend.a plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time, the parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time being different from at least one other parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time corresponding to at least one other physiological parameter .Iadd.for which the one or more processors are configured to determine at least one measurement.Iaddend.; .[.activate an alarm in response to determining that an alarm activation threshold has been satisfied by the physiological parameter measurement; receive.]. .Iadd.determine that the alarm hold initiator indicates to hold an indication of .Iaddend.an alarm .[.suspension indication.]. .Iadd.for the alarm condition.Iaddend.; .[.and.]. in response to .[.receiving.]. .Iadd.determining that the alarm hold initiator indicates to hold the indication of .Iaddend.the alarm .[.suspension indication.]., .[.suspend.]. .Iadd.hold the indication of .Iaddend.the alarm for the .[.indicated.]. parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time.Iadd.; and subsequent to the parameter-specific alarm hold period of time passing, activate the indication of the alarm while the measurement of the physiological parameter satisfies the activation threshold.Iaddend..
2. The physiological measurement system of claim 1, wherein the one or more processors are further configured to: provide a user interface to the user including at least a plurality of user-selectable elements, each of the .Iadd.plurality of user-.Iaddend.selectable elements corresponding to one of the plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.
3. The physiological measurement system of claim 2, wherein providing the user interface further includes: constructing a pop-up window for a display; and displaying the plurality of user-selectable elements in the pop-up window.
4. The physiological measurement system of claim 3, wherein the plurality of user-selectable elements are configured to allow a user to select a specific one of the plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.
5. The physiological measurement system of claim 4, wherein .[.the.]. .Iadd.a .Iaddend.selected parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time is selected by selection of one of the plurality of user-selectable elements.
6. The physiological measurement system of claim 1, wherein the one or more processors are further configured to: associate .[.the.]. .Iadd.a .Iaddend.selected parameter-specific .Iadd.alarm hold .Iaddend.period of time .[.is.]. with the physiological parameter.
7. The physiological measurement system of claim 6, wherein the selected parameter-specific .Iadd.alarm hold .Iaddend.period of time is stored in a memory device in communication with the one or more processors.
8. The physiological measurement system of claim 1, wherein the one or more processors are further configured to: determine a .Iadd.measurement of a .Iaddend.second physiological parameter .[.measurement.]. based at least in part upon the signal.Iadd., the second physiological parameter being different from the physiological parameter.Iaddend.; .Iadd.determine whether a second alarm condition exists by determining whether a second activation threshold has been satisfied by the measurement of the second physiological parameter;.Iaddend. .[.receive, from the user,.]. .Iadd.access .Iaddend.a second .[.indication of.]. .Iadd.alarm hold initiator for .Iaddend.a second parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time corresponding to the second physiological parameter, the second parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time being .[.selected from.]. .Iadd.one of .Iaddend.a second plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.Iadd., the second parameter-specific alarm hold period of time being different from the parameter-specific alarm hold period of time corresponding to the physiological parameter.Iaddend.; .[.activate a second alarm in response to determining a second alarm activation threshold has been satisfied by the second physiological parameter measurement.]. .Iadd.determine that the second alarm hold initiator indicates to hold an indication of a second alarm for the second alarm condition.Iaddend.; .[.and.]. in response to .[.receiving.]. .Iadd.determining that the second alarm hold initiator indicates to hold the indication of .Iaddend.the .Iadd.second .Iaddend.alarm .[.suspension indication.]., .[.suspend.]. .Iadd.hold the indication of .Iaddend.the second alarm for the .[.indicated.]. second parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time.Iadd.; and subsequent to the second parameter-specific alarm hold period of time passing, activate the indication of the second alarm while the measurement of the second physiological parameter satisfies the second activation threshold.Iaddend..
9. The physiological measurement system of claim 8, wherein the one or more processors are further configured to: provide a user interface to the user including at least a first plurality of user-selectable elements and a second plurality of user-selectable elements, wherein each of the first plurality of user-selectable elements corresponds to one of the plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time, and each of the second plurality of user-selectable element corresponds to one of the second plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.
10. The physiological measurement system of claim 9, wherein the one or more processors are further configured to: construct a pop-up window for a display; and display both the first and second plurality of user-selectable elements in the pop-up window.
11. The physiological measurement system of claim 10, wherein .[.the.]. .Iadd.a .Iaddend.selected .Iadd.first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time is selected by selection of one of the first plurality of user-selectable elements, and .[.the.]. .Iadd.a .Iaddend.selected second parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time is selected by selection of one of the second plurality of user-selectable elements.
12. The physiological measurement system of claim 11, wherein the at least one of the .[.first.]. plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time is different from any of the second plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.
13. .[.An.]. .Iadd.A .Iaddend.method comprising: measuring a .Iadd.first .Iaddend.physiological parameter .Iadd.and a second physiological parameter .Iaddend.using a patient monitoring device, the patient monitoring device including a processor and a memory device .[.configured to store a parameter-specific alarm suspension period of time.].; .[.receiving.]. .Iadd.accessing.Iaddend., from .[.a user.]. .Iadd.the memory device.Iaddend., .[.an indication of.]. .Iadd.a first alarm hold initiator for .Iaddend.a .Iadd.first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time corresponding to the .Iadd.first .Iaddend.physiological parameter .Iadd.and a second alarm hold initiator for a second parameter-specific alarm hold period of time corresponding to the second physiological parameter.Iaddend., .[.the parameter-specific alarm suspension period of time being selected from a plurality of parameter-specific alarm suspension periods of time,.]. the .Iadd.first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time being different from .[.at least one other.]. .Iadd.the second .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time .[.corresponding to at least one other physiological parameter; activating an alarm in response to determining an alarm activation threshold has been satisfied by the physiological parameter measurement.].; .[.receiving an.]. .Iadd.determining that the first alarm hold initiator indicates to hold a first indication of a first .Iaddend.alarm .[.suspension indication.]. .Iadd.for a first alarm condition for the first physiological parameter.Iaddend.; .[.and.]. in response to .[.receiving.]. .Iadd.determining that the first alarm hold initiator indicates to hold the first indication of .Iaddend.the .Iadd.first .Iaddend.alarm .[.suspension indication.]., .[.suspending.]. .Iadd.holding the first indication of .Iaddend.the .Iadd.first .Iaddend.alarm for the .[.indicated.]. .Iadd.first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time.Iadd.; and subsequent to the first parameter-specific alarm hold period of time passing, activating the first indication of the first alarm.Iaddend..
14. The method of claim 13, wherein the .Iadd.first .Iaddend.alarm includes an audible component and a visual component, and wherein .[.suspending.]. .Iadd.holding the first indication of .Iaddend.the .Iadd.first .Iaddend.alarm comprises .[.suspending.]. .Iadd.holding .Iaddend.the audible component and not .[.suspending.]. .Iadd.holding .Iaddend.the visual component.
15. The method of claim 13 further comprising: providing a user interface to the user including at least a plurality of user-selectable elements, each of the .Iadd.plurality of user-.Iaddend.selectable elements corresponding to one of .[.the.]. .Iadd.a .Iaddend.plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.Iadd., wherein the plurality of parameter-specific alarm hold periods of time comprise the first parameter specific alarm hold period of time.Iaddend..
16. The method of claim 15 further comprising: constructing a pop-up window for a display; and displaying the plurality of user-selectable elements in the pop-up window.
17. The method of claim 16, wherein .[.the.]. .Iadd.a .Iaddend.selected parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time is selected by selection of one of the plurality of user-selectable elements.
18. A physiological measurement system comprising: a physiological sensor .[.means for outputting.]. .Iadd.configured to output .Iaddend.a signal responsive to a .[.noninvasive measurement of attenuated light transmitted through a tissue site.]. .Iadd.physiological condition .Iaddend.of a patient; .Iadd.a memory configured to store a first alarm activation threshold; .Iaddend.and .[.a processing means.]. .Iadd.one or more processors .Iaddend.in communication with the physiological sensor .[.means.]. and configured to: determine a .Iadd.first .Iaddend.measurement of .[.a.]. .Iadd.the first measured .Iaddend.physiological parameter based at least in part upon the signal; .Iadd.determine whether a first alarm condition exists by determining whether the first alarm activation threshold has been satisfied by the first measurement of the first measured physiological parameter; .Iaddend. .[.receive, from a user, an indication of a.]. .Iadd.access a first alarm hold initiator for a first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time corresponding to the .Iadd.first measured .Iaddend.physiological parameter, .[.the parameter-specific alarm suspension period of time being selected from a plurality of parameter-specific alarm suspension periods of time,.]. the .Iadd.first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time being different from .[.at least one other.]. .Iadd.a second .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time corresponding to .[.at least one other.]. .Iadd.a second measured .Iaddend.physiological parameter .Iadd.for which the one or more processors are configured to determine a second measurement.Iaddend.; .[.activate an alarm in response to determining an alarm activation threshold has been satisfied by the physiological parameter measurement; receive an.]. .Iadd.determine that the first alarm hold initiator indicates to hold activation of a first .Iaddend.alarm .[.suspension indication.]. .Iadd.for the first alarm condition.Iaddend.; .[.and.]. in response to .[.receiving.]. .Iadd.determining that the first alarm hold initiator indicates to hold activation of .Iaddend.the .Iadd.first .Iaddend.alarm .[.suspension indication.]., .[.suspend.]. .Iadd.hold activation of .Iaddend.the .Iadd.first .Iaddend.alarm for the .[.indicated.]. .Iadd.first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time.Iadd.; and activate the first alarm.Iaddend..
19. The physiological measurement system of claim 18, wherein the .[.processing means is.]. .Iadd.one or more processors are .Iaddend.further configured to: determine .[.a.]. .Iadd.the .Iaddend.second .[.physiological parameter.]. measurement based at least in part upon the signal; .Iadd.determine whether a second alarm condition exists by determining whether a second alarm activation threshold has been satisfied by the second measurement; .Iaddend. .[.receive, from the user,.]. .Iadd.access .Iaddend.a second .[.indication of a.]. .Iadd.alarm hold initiator for the .Iaddend.second parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time .[.corresponding to the second physiological parameter, the second parameter-specific alarm suspension period of time being selected from a second plurality of parameter-specific alarm suspension periods of time; activate a second alarm in response to determining a second alarm activation threshold has been satisfied by the second physiological parameter measurement.].; .Iadd.determine that the second alarm hold initiator indicates to hold activation of a second alarm for the second alarm condition; .Iaddend.and in response to .[.receiving.]. .Iadd.determining that the second alarm hold initiator indicates to hold activation of .Iaddend.the .Iadd.second .Iaddend.alarm .[.suspension indication.]., .[.suspend.]. .Iadd.hold activation of .Iaddend.the second alarm for the .[.indicated.]. second parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time.
20. The physiological measurement system of claim 19, wherein the .[.processing means is.]. .Iadd.one or more processors are .Iaddend.further configured to: provide a user interface to the user including at least a first plurality of user-selectable elements and a second plurality of user-selectable elements, wherein each of the first plurality of user-selectable elements corresponds to one of .[.the.]. .Iadd.a first .Iaddend.plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time, and each of the second plurality of user-selectable .[.element.]. .Iadd.elements .Iaddend.corresponds to one of .[.the.]. .Iadd.a .Iaddend.second plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.Iadd., wherein the first plurality of parameter-specific alarm hold periods of time comprise the first parameter-specific alarm hold period of time, and the second plurality of parameter-specific alarm hold periods of time comprise the second parameter-specific alarm hold period of time.Iaddend..
21. The physiological measurement system of claim 20, wherein .[.the.]. .Iadd.a .Iaddend.selected .Iadd.first .Iaddend.parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time is selected by selection of one of the first plurality of user-selectable elements, and .[.the.]. .Iadd.a .Iaddend.selected second parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.period of time is selected by selection of one of the second plurality of user-selectable elements.
22. The physiological measurement system of claim 21, wherein at least one of the first plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time is different from any of the second plurality of parameter-specific alarm .[.suspension.]. .Iadd.hold .Iaddend.periods of time.
.Iadd.23. The physiological measurement system of claim 1, wherein the one or more processors are further configured to modify a visual indicator for the parameter-specific alarm hold period of time..Iaddend.
.Iadd.24. The physiological measurement system of claim 1, wherein the physiological parameter comprises one of an oxygen saturation, a pulse rate, a perfusion index, a pleth variability index, a carboxyhemoglobin, a methemoglobin, or a total hemoglobin..Iaddend.
Description
BACKGROUND
Pulse oximetry for measuring constituents of circulating blood has achieved acceptance in a wide variety of medical applications, including surgical wards, intensive care and neonatal units, general wards, home care, physical training, and virtually all types of monitoring scenarios. A pulse oximeter generally includes a two-wavelength optical sensor applied to a patient, a monitor for processing sensor signals and displaying results and a patient cable electrically interconnecting the sensor and the monitor. The monitor typically provides a numerical readout of physiological parameters such as oxygen saturation (SpO.sub.2) and pulse rate (PR). Advanced physiological monitors utilize multiple wavelength sensors and enhanced measurement capabilities to provide readouts of additional parameters, such as carboxyhemoglobin (HbCO), methemoglobin (HbMet) and total hemoglobin (Hbt).
Pulse oximeters capable of reading through motion induced noise are disclosed in at least U.S. Pat. Nos. 6,770,028, 6,658,276, 6,650,917, 6,157,850, 6,002,952, 5,769,785 and 5,758,644; low noise pulse oximetry sensors are disclosed in at least U.S. Pat. Nos. 6,088,607 and 5,782,757; all of which are assigned to Masimo Corporation, Irvine, Calif. ("Masimo") and are incorporated by reference herein.
Physiological monitors and corresponding multiple wavelength optical sensors are described in at least U.S. patent application Ser. No. 11/367,013, filed Mar. 1, 2006 and titled Multiple Wavelength Sensor Emitters and U.S. patent application Ser. No. 11/366,208, filed Mar. 1, 2006 and titled Noninvasive Multi-Parameter Patient Monitor, both assigned to Masimo Laboratories, Irvine, Calif. (Masimo Labs) and both incorporated by reference herein.
Further, physiological monitoring systems that include low noise optical sensors and pulse oximetry monitors, such as any of LNOP.RTM. adhesive or reusable sensors, SofTouch.TM. sensors, Hi-Fi Trauma.TM. or Blue.TM. sensors; and any of Radical.RTM., SatShare.TM., Rad-9.TM., Rad-5.TM., Rad-5v.TM. or PPO+.TM. Masimo SET.RTM. pulse oximeters, are all available from Masimo. Physiological monitoring systems including multiple wavelength sensors and corresponding noninvasive blood parameter monitors, such as Rainbow.TM. adhesive and reusable sensors and RAD-57.TM. and Radical-7.TM. monitors for measuring SpO.sub.2, pulse rate (PR), perfusion index (PI), pleth variability index (PVI), signal quality, HbCO and HbMet among other parameters are also available from Masimo.
SUMMARY OF THE INVENTION
Monitor alarms are triggered by out-of-limit parameters and system failures, the latter including monitor or sensor failures or improper sensor placement, to name a few. Alarms can be visual, audible or both. Alarms can also have different levels of priority, which are reflected in the type of visual and audible alarms. In an embodiment, parameters exceeding limits such as low SpO.sub.2, high HbCO, high HbMet and low and high BPM trigger high priority alarms. System failures due to sensor off, no sensor or defective sensor also trigger high priority alarms. Parameters exceeding limits such as high SpO.sub.2, low and high PI, low and high PVI, for example, trigger medium priority alarms. Parameters exceeding limits such as low HbCO and low HbMet along with a system low battery indication are examples of low priority alarms.
An audible alarm may be temporarily suspended by pressing an alarm silence button so as to prevent unnecessary disturbance to the patient and distraction of the caregiver. During alarm suspension, visual alarms remain active. If an alarm condition persists after a predetermined alarm suspend period, the audible alarm resumes. The alarm suspend period is typically long enough to give a caregiver sufficient time to intervene with appropriate patient treatment yet short enough to ensure that patient health is not endangered if intervention is ineffective. For conventional pulse oximetry, an alarm suspend may be, for example, a maximum of 120 seconds.
Alarm suspension on advanced blood parameter monitors is problematic. With conventional pulse oximetry, treatment for abnormal parameter measurements can be quickly applied and a patient response is typically fast. For example, a treatment for low oxygen saturation is the application of an oxygen mask or an increase in oxygen flow. By contrast, the duration of treatment for parameters measured by advanced monitors is highly dependent on the alarm-triggering parameter. For example, the treatment for high methemoglobin is the injection of methylene blue, and the patient response to such an injection is slow. When patient treatment time exceeds the maximum alarm suspend period, an audible alarm will constantly reactivate. Thus, a single alarm suspend duration for all parameters is inadequate to cope with the many different types of parameters measured by advanced monitors.
One aspect of an alarm suspend system for silencing the alarms is an alarm trigger responsive to any of various parameters and predetermined limits corresponding to the parameters, where the parameters are partitioned according to treatment time, i.e. the relative length of time it takes for a person to respond to medical treatment for a parameter measurement outside of the predetermined limits. An audible alarm is responsive to the alarm trigger. An alarm silence button is actuated so as to suspend the audible alarm. A timer tracks the duration of the suspended alarm and is initiated by actuation of an alarm silence button. The timer retriggers the audible alarm after the timed duration has lapsed/expired. In an embodiment, a long duration suspend time is associated with slow treatment parameters and a short duration suspend time is associated with fast treatment parameters. Fast treatment parameters may include, for example, parameters relating to normal blood hemoglobin constituents and slow treatment parameters may include parameters relating to abnormal blood hemoglobin constituents.
In various embodiments, a short duration suspend time is less than or equal to about two minutes and a long duration suspended time is greater than about two minutes. A default duration associated with the fast treatment parameters is about two minutes and a default duration associated with the slow treatment parameters is about fifteen minutes. The alarm suspend system may also have an alarm suspend override responsive to a predetermined unit change in the parameter triggering a suspended alarm. The override results in reactivation of the suspended alarm. A physiological monitor having an alarm suspend system may also have a pop-up window that appears on the monitor display in response to actuation of the silence button, where the pop-up window presents a choice of alarm suspend durations.
Another aspect of an alarm suspend system is a partition of measured parameters into at least a first group and a second group. An audible alarm is triggered if at least one parameter is outside of predetermined limits. The audible alarm is suspended in response to a silence request. A first duration is associated with the first group and a second duration is associated with the second group. The audible alarm is reactivated after at least one of the first duration and the second duration. The first duration may be set so as to generally correspond to a first range of treatment times for the first group of parameters. Likewise, the second duration may be set so as to generally correspond to a second range of treatment times for the second group of parameters, where the first range of treatment times and the second range of treatment times are non-overlapping.
In various embodiments, suspended audible alarms are overridden if the triggering parameter has greater than a predetermined unit change before the suspended alarm expires according to either the first duration or the second duration. The first and second groups are defined in relation to normal hemoglobin measurements abnormal hemoglobin measurements, respectively. The first duration is set to be less than or equal to two minutes and the second duration is set to be greater than two minutes, with default durations of about two minutes corresponding to the first group and about fifteen minutes corresponding to the second group. In an embodiment, a pop-up window for a monitor display is constructed and the first duration and the second duration are selected from a range of durations presented within the pop-up window.
A further aspect of an alarm suspend system deactivates an audible alarm for one of a short duration and a long duration according to the alarm-triggering parameter. A first group of parameters is associated with the short duration and a second group of parameters is associated with the long duration. The first group and the second group are partitioned according to a fast treatment time and a short treatment time associated with the parameters. An override reactivates the audible alarm if the trigger parameter changes more than a predetermine amount during the corresponding duration. In various embodiments, the first group comprises parameters related to the measurement of normal hemoglobin and the second group comprises parameters related to the measurement of abnormal hemoglobin. The long duration is greater than about 120 seconds and the short duration is less than or equal to about 120 seconds. A pop-up window for the display allows selection of the long duration and the short duration in response to the silence button.
BRIEF DESCRIPTION OF THE DRAWINGS
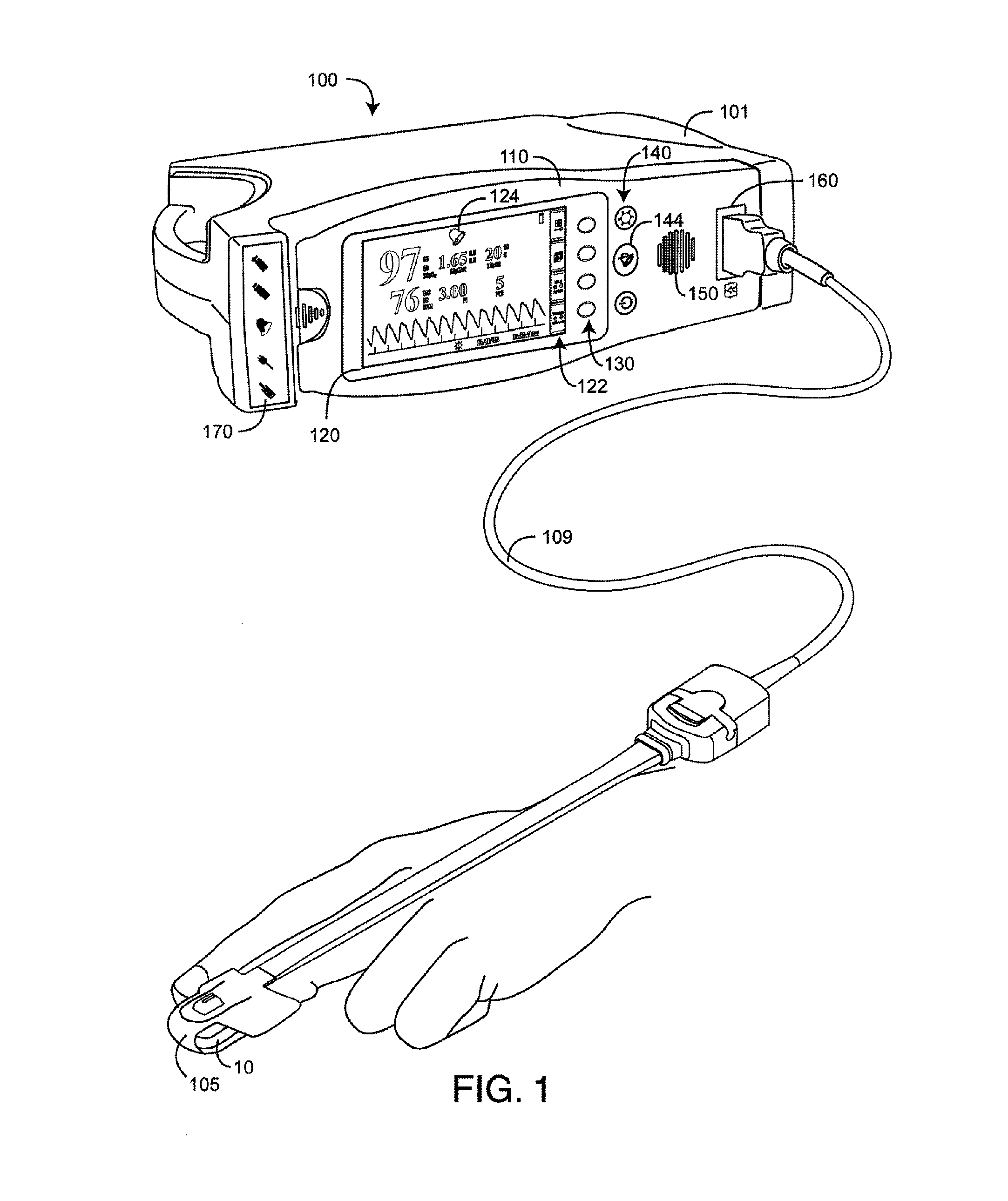
FIG. 1 is a perspective view of a physiological measurement system utilizing an alarm suspend system;
FIG. 2 is a detailed block diagram of a physiological measurement system utilizing an alarm suspend system;
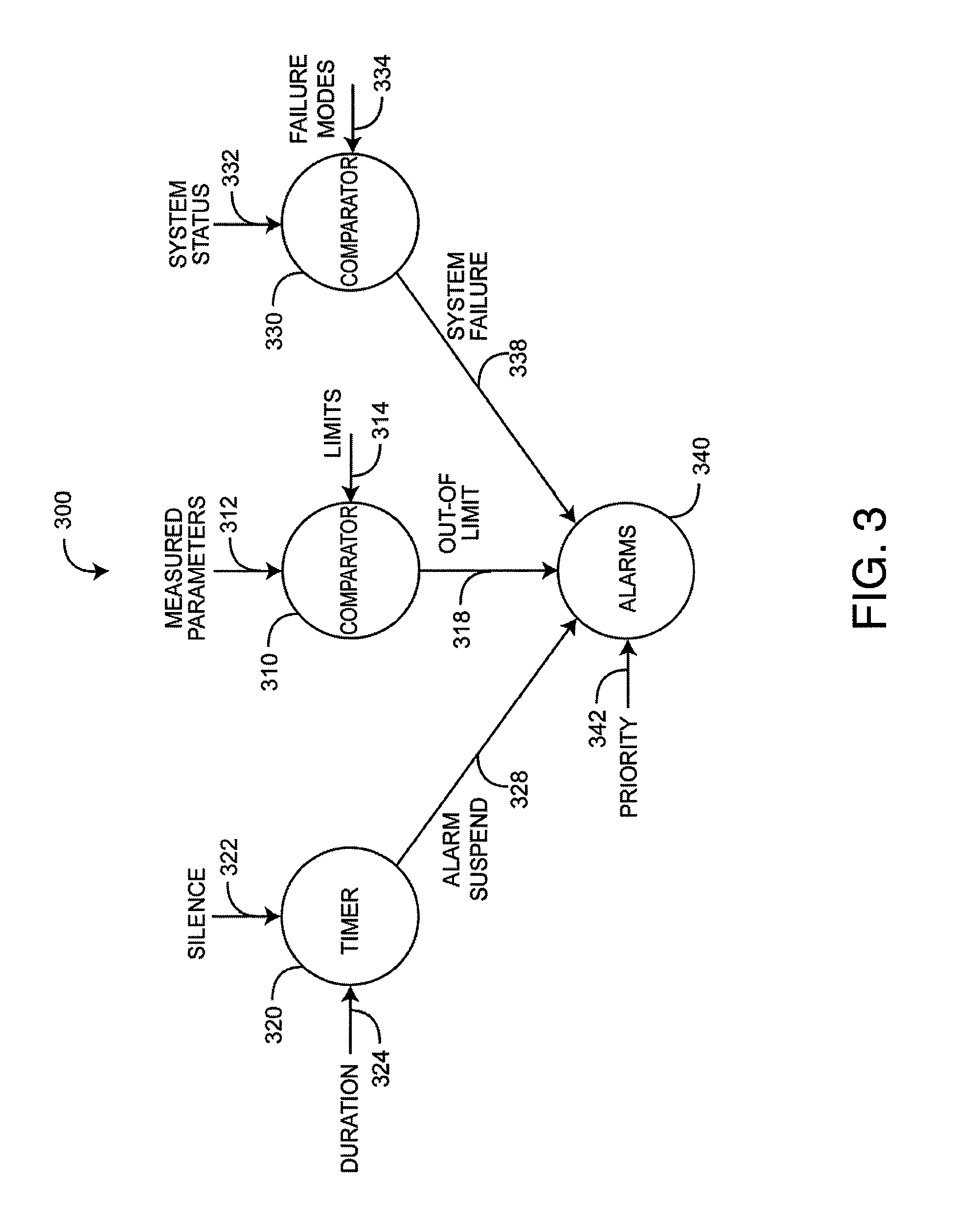
FIG. 3 is a flow diagram of an alarm suspend system embodiment;
FIG. 4 is a state diagram of an alarm suspend system embodiment; and
FIG. 5 is an illustration of an alarm suspend pop-up window.
DETAILED DESCRIPTION
FIG. 1 illustrates a physiological measurement system 100 that utilizes an alarm suspend system. The physiological measurement system 100 has a noninvasive sensor 105 attached to a tissue site 10, a physiological monitor 101, and an interface cable 109 interconnecting the monitor 101 and the sensor 105. The physiological measurement system 100 may incorporate pulse oximetry in addition to advanced features, such as a multiple wavelength sensor and advanced processes for determining physiological parameters other than or in addition to those of pulse oximetry, such as carboxyhemoglobin, methemoglobin and total hemoglobin, as a few examples.
The monitor 101 has a front panel 110 providing a display 120, touch keys 130, controls 140, a speaker 150, a sensor port 160 and status indicators 170. The display 120 shows parameter readouts, limits and waveforms among other items. The display 120 also has touch key icons 122 that indicate touch key 130 functions. The speaker 150 provides an audible alarm in response to physiological measurements that violate preset conditions, such as an out-of-limit parameter, as well as system failures, such as a low battery condition. The controls 140 include an alarm silence button 144 that is pressed to temporarily suspend out-of-limit parameter alarms and system alarms, such as low battery. The display 120 provides visual alarms, which include a bell-shaped alarm status indicator 124 that illuminates during an alarm condition and parameter readouts 210 and limits 220 that flash when parameters are out-of-limit. Status indicators 170 also provide visual alarms. When there are multiple alarm conditions, the parameter displays 202 indicate parameters with the highest alarm priority. Touch keys 130 and corresponding icons 122 include an alarm menu access button for setting alarm conditions, such as high or low alarm limits for SpO.sub.2, HbCO, HbMet, PR and PI. The alarm silence button 144 is pressed to temporarily suspend audible alarms. Advantageously, an alarm suspend system provides a parameter-dependent variation in the alarm suspend duration, as described below, utilizing a common silence button or other suspend initiator.
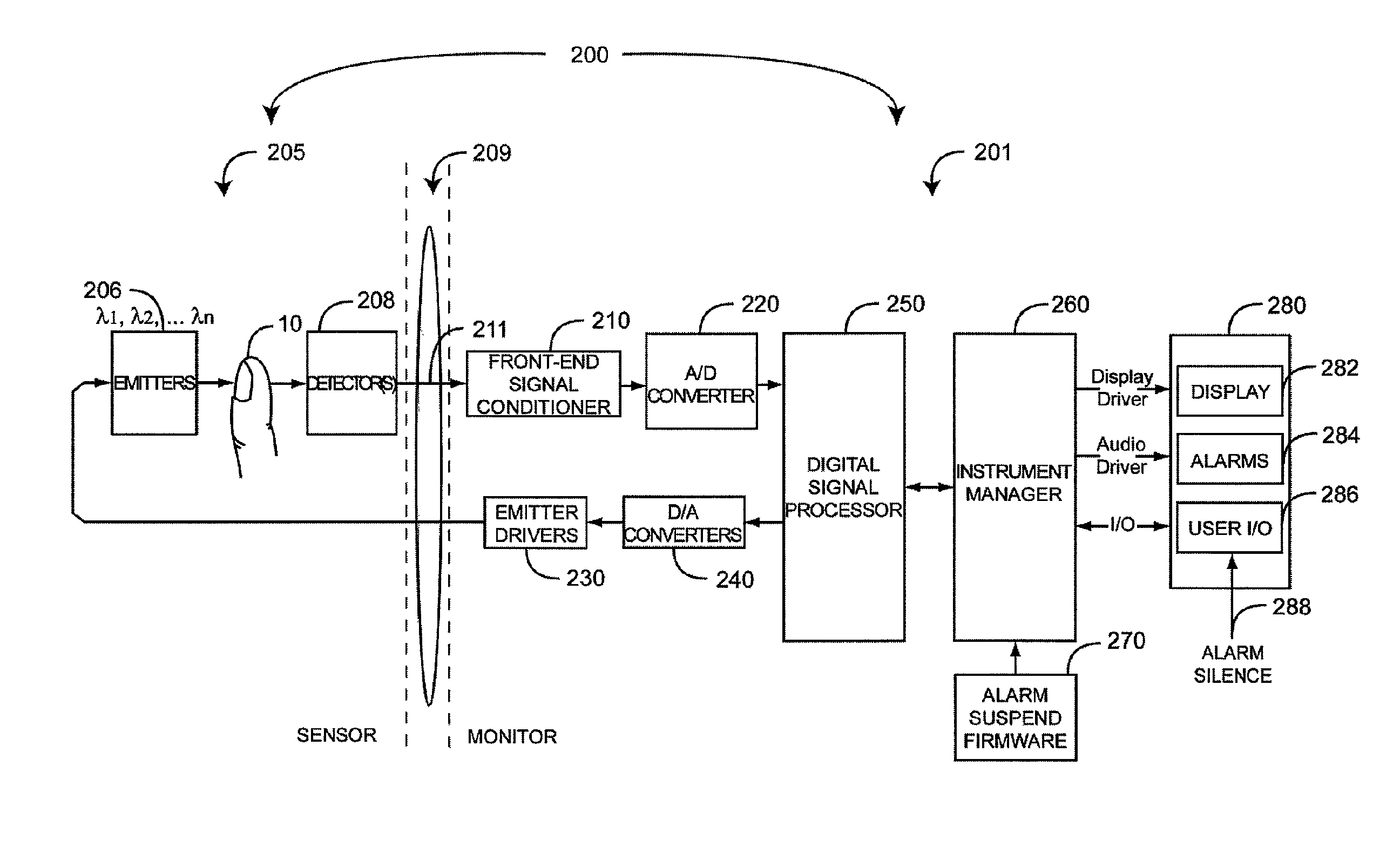
FIG. 2 illustrates a physiological measurement system 200 including a physiological monitor 201, a sensor 205 and an interface cable 209. The sensor 205 is attached to a tissue site, such as a finger 10, and includes a plurality of emitters 206 irradiating the tissue site 10 with multiple wavelengths of light. The sensor 205 also includes one or more detectors 208 capable of detecting the light after attenuation by the tissue site 10. The sensor 205 transmits optical radiation at wavelengths other than or including the red and infrared wavelengths utilized in pulse oximeters. The monitor 201 inputs a corresponding sensor signal 211 and determines the relative concentrations of blood constituents other than or in addition to the "normal" blood hemoglobin constituents HbO.sub.2 and Hb, including "abnormal" blood hemoglobin constituents HbCO, HbMet and blood related parameters such as fractional oxygen saturation, total hemoglobin and blood glucose to name a few.
As shown in FIG. 2, the monitor 201 has a front-end signal conditioner 210, an A/D converter 220, emitter drivers 230, D/A converters 240 and a digital signal processor ("DSP") 250. In general, the emitter drivers 230 convert digital control signals, via the D/A converters 240, into analog drive signals capable of driving the sensor emitters 206. The front-end signal conditioner 210 converts, via the A/D converter 220, composite analog intensity signal(s) from light sensitive detector(s) 208 into digital data input to the DSP 250. The emitter drivers 230 and front-end signal conditioner 210 communicate with the sensor 205 via the interface cable 209.
Also shown in FIG. 2, the monitor 201 has an instrument manager 260 and a user interface 280. The user interface 280 includes one or more displays 282, alarms 284 and user input/output (I/O) 286. The instrument manager 260 communicates with the DSP 250 to receive parameter data and to present that data on the display 282. The instrument manager 260 may also store and display historical or trending data related to one or more of the measured parameters or combinations of the measured parameters. The instrument manager 260 also controls audible and visual alarms and indicators 284. The instrument manager 260 responds to user-actuated keys and communicates with external devices via various I/O ports 286. Further, the instrument manager 260 executes alarm suspend firmware 270 so as to respond to an alarm silence button press 288, as described in detail with respect to FIGS. 3-4.
FIG. 3 generally illustrates an alarm suspend system 300. Alarm triggers include system failures 338 and out-of-limit parameters 318. Triggered alarms 340 may be audible, visual or both, and may vary according to priority 342. Audible alarms may be generated by a monitor front-panel-mounted speaker 150 (FIG. 1) and may vary in loudness, pitch and sound pattern. Visual alarms may include parameter labels, parameter numerics, symbols and status lights, which can flash and vary in color.
As shown in FIG. 3, measured parameters 312 are compared 310 to default or user-specified limits 314. An out-of-limit condition 318 triggers an alarm 340. An alarm suspend 328 is user-initiated by a silence request 322. This may be a press of a silence button 144 (FIG. 1) on a monitor front panel 110 (FIG. 1). In an embodiment, the alarm suspend 328 silences audible alarms and modifies the display of visual alarms. The alarm suspend 328 is based on a timer 320, which ends the alarm suspend 328 after a predetermined duration 324. The duration 324 may be a function of the out-of-limit parameter 312. In an advantageous embodiment, the duration 324 relates to, or is a function of, the treatment time for the alarm-triggering parameter so as to avoid nuisance alarms while maintaining alarm integrity.
FIG. 4 illustrates an alarm suspend embodiment 400 that operates independently for each measured parameter that can trigger an alarm. An alarm is initially off 410. The alarm remains off as long as the parameter is within its set limits 412. If a parameter is measured outside of its set limits 414, an alarm is triggered 420. The alarm may audible, visual or both audible and visual. A user can request to silence the alarm by pressing an alarm silence button 144 (FIG. 1), for example. The silence request 422 suspends the alarm 430 which turns off audible alarms but, in an embodiment, does not deactivate visual alarms. The audible alarm remains suspended 430 for a predetermined duration 432. When the suspend duration has passed, the alarm suspend expires 434 and audible alarms are once again activated 420. The alarm remains on 428 until the triggering parameter is within limits 424 or a user once again requests silence 422. The alarm suspend 430 deactivates if the measured parameter becomes within limits 438, such as when the patient condition improves, or if no physiological data is detected 439, such as no sensor, sensor off, no cable or malfunctioning sensor situations, to name a few. Also, if the measured parameter changes during the alarm suspend 430 by a sufficient out-of-limit amount, an override 436 reactivates the audible alarms 420.
In an alarm suspend system embodiment, parameters are classified according to the typical time it takes for medical treatment to transition an out-of-limit measurement to a within-limit measurement. Suspend durations 324 (FIG. 3) are set accordingly. For example, in a two-tier embodiment, relatively slow treatment parameters, such as HbMet, HbCO, Hbt and PVI, are assigned relatively long suspend durations. Similarly, relatively fast treatment parameters, such as SpO.sub.2 and PR, are assigned relatively short suspend durations. In an embodiment, the alarm suspend duration is adjustable for each individual parameter, including 2, 5, 10, 15, 20, 25 and 30 minutes for slow treatment parameters, with a default of 15 minutes; and 30, 60, 90 and 120 seconds for fast treatment parameters, with a default of 120 seconds. These alarm features are only active when alarm limits have been set. Other alarm features apply to both slow treatment and fast treatment parameters. For example, an alarm delay of 0, 5, 10 or 15 seconds applies to all enabled parameters.
In an embodiment, an override 436 occurs if slow treatment parameters such as HbCO, HbMet or PVI increase or Hbt decreases by a certain unit change during the alarm suspend duration. The unit change is adjustable for each parameter, such as from 1-15 in increments of 1. TABLE 1 shows a default embodiment of override unit changes for these parameters.
TABLE-US-00001 TABLE 1 Override Unit Changes for Selected Parameters Parameter Unit Change Direction HbCO 5 Increase HbMet 2 Increase Hbt 2 Decrease PVI OFF Increase
FIG. 5 illustrates an alarm suspend window 500 that provides a "pop-up" display so that a monitor user may manually enter an alarm suspend duration. The alarm suspend window 500 appears as a portion of a monitor display 501, such as the front panel display 120 (FIG. 1) described above. The pop-up window 500 responds to a suspend request, such as a silence button 144 (FIG. 1) press. The alarm suspend window 500 has a window identifier 502 and one or more parameter subsections 510, 520. Each parameter subsection 510, 520 has a parameter identifier 512, 522 and corresponding suspend duration options 514, 524. In an embodiment, specific suspend times are selected via monitor touch keys 130 (FIG. 1) as guided by corresponding touch key icons 560. Selected suspend times are highlighted or otherwise identified and entered, also via a touch key 130 (FIG. 1). In an alternative embodiment, the monitor display is a touch screen and alarm suspend times are directly entered by a finger press on a specific duration "virtual button" 514, 524. Once one or more suspend durations are entered, the pop-up window 500 disappears from the display 501. The alarm suspend window 500 advantageously allows a user to quickly choose an appropriate alarm suspend duration for the situation at hand, rather than relying on a predetermined or default duration.
An alarm suspend system is described above with respect to alarms triggered by measured parameters and limits associated with those measured parameters. Limits may correspond to levels of a measured parameter, such as a percentage oxygen saturation to name but one example. Limits may also correspond to trends of a measured parameter, such as a rate-of-change of oxygen saturation, for example. Limits may also correspond to patterns in a measured parameter or a comparison of one measured parameter with another measured parameter, as further examples.
An alarm suspend system is described above with respect to a two-tier grouping of parameters, such as slow treatment and fast treatment parameters and alarm suspend durations associated with those groups. Groupings of parameters with respect to alarm suspend durations may be multi-tier, such as slow, medium and fast treatment parameters, to name but one example.
An alarm suspend system has been disclosed in detail in connection with various embodiments. These embodiments are disclosed by way of examples only and are not to limit the scope of the claims that follow. One of ordinary skill in the art will appreciate many variations and modifications.
* * * * *
References
D00000

D00001

D00002

D00003

D00004

D00005

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.