Heart valve repair and replacement
Braido , et al. December 8, 2
U.S. patent number 10,856,974 [Application Number 16/217,869] was granted by the patent office on 2020-12-08 for heart valve repair and replacement. This patent grant is currently assigned to St. Jude Medical, Cardiology Division, Inc.. The grantee listed for this patent is St. Jude Medical, Cardiology Division, Inc.. Invention is credited to Thomas M. Benson, Peter N. Braido, Theodore Paul Dale, Mina S. Fahim, Mathias Charles Glimsdale, Mark Krans, Andrea N. Para.






| United States Patent | 10,856,974 |
| Braido , et al. | December 8, 2020 |
| **Please see images for: ( Certificate of Correction ) ** |
Heart valve repair and replacement
Abstract
A prosthetic heart valve having an inflow end and an outflow end includes a collapsible and expandable stent having a plurality of commissure features, a plurality of first struts and a plurality of second struts. The plurality of first struts define a substantially cylindrical portion and the plurality of second struts have first ends attached to the cylindrical portion and free ends projecting radially outward from the cylindrical portion and configured to couple to adjacent heart tissue to anchor the stent. A collapsible and expandable valve assembly disposed within the stent has a plurality of leaflets coupled to the commissure features.
| Inventors: | Braido; Peter N. (Wyoming, MN), Fahim; Mina S. (Shoreview, MN), Benson; Thomas M. (Minneapolis, MN), Dale; Theodore Paul (Corcoran, MN), Para; Andrea N. (Centennial, TX), Krans; Mark (Hopkins, MN), Glimsdale; Mathias Charles (St. Michael, MN) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | St. Jude Medical, Cardiology
Division, Inc. (St. Paul, MN) |
||||||||||
| Family ID: | 56236094 | ||||||||||
| Appl. No.: | 16/217,869 | ||||||||||
| Filed: | December 12, 2018 |
Prior Publication Data
| Document Identifier | Publication Date | |
|---|---|---|
| US 20190110894 A1 | Apr 18, 2019 | |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | Issue Date | ||
|---|---|---|---|---|---|
| 15177598 | Jun 9, 2016 | 10179042 | |||
| 62174690 | Jun 12, 2015 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61F 2/2469 (20130101); A61F 2/2409 (20130101); A61F 2/2418 (20130101); A61F 2220/0008 (20130101); A61F 2230/0006 (20130101); A61F 2230/0069 (20130101); A61F 2220/0016 (20130101) |
| Current International Class: | A61F 2/24 (20060101) |
References Cited [Referenced By]
U.S. Patent Documents
| 3657744 | April 1972 | Ersek |
| 4275469 | June 1981 | Gabbay |
| 4491986 | January 1985 | Gabbay |
| 4759758 | July 1988 | Gabbay |
| 4878906 | November 1989 | Lindemann et al. |
| 4922905 | May 1990 | Strecker |
| 4994077 | February 1991 | Dobben |
| 5411552 | May 1995 | Andersen et al. |
| 5415664 | May 1995 | Pinchuk |
| 5480423 | January 1996 | Ravenscroft et al. |
| 5843167 | December 1998 | Dwyer et al. |
| 5855601 | January 1999 | Bessler et al. |
| 5935163 | August 1999 | Gabbay |
| 5961549 | October 1999 | Nguyen et al. |
| 6077297 | June 2000 | Robinson et al. |
| 6083257 | July 2000 | Taylor et al. |
| 6090140 | July 2000 | Gabbay |
| 6214036 | April 2001 | Letendre et al. |
| 6264691 | July 2001 | Gabbay |
| 6267783 | July 2001 | Letendre et al. |
| 6368348 | April 2002 | Gabbay |
| 6419695 | July 2002 | Gabbay |
| 6458153 | October 2002 | Bailey |
| 6468660 | October 2002 | Ogle et al. |
| 6488702 | December 2002 | Besselink |
| 6517576 | February 2003 | Gabbay |
| 6533810 | March 2003 | Hankh et al. |
| 6582464 | June 2003 | Gabbay |
| 6610088 | August 2003 | Gabbay |
| 6623518 | September 2003 | Thompson et al. |
| 6685625 | February 2004 | Gabbay |
| 6719789 | April 2004 | Cox |
| 6730118 | May 2004 | Spenser et al. |
| 6783556 | August 2004 | Gabbay |
| 6790230 | September 2004 | Beyersdorf et al. |
| 6814746 | November 2004 | Thompson et al. |
| 6830584 | December 2004 | Seguin |
| 6869444 | March 2005 | Gabbay |
| 6893460 | May 2005 | Spenser et al. |
| 6908481 | June 2005 | Cribier |
| 7018406 | March 2006 | Seguin et al. |
| 7025780 | April 2006 | Gabbay |
| 7137184 | November 2006 | Schreck |
| 7160322 | January 2007 | Gabbay |
| 7247167 | July 2007 | Gabbay |
| 7267686 | September 2007 | DiMatteo et al. |
| 7311730 | December 2007 | Gabbay |
| 7374573 | May 2008 | Gabbay |
| 7381218 | June 2008 | Schreck |
| 7452371 | November 2008 | Pavcnik et al. |
| 7510572 | March 2009 | Gabbay |
| 7524331 | April 2009 | Birdsall |
| RE40816 | June 2009 | Taylor et al. |
| 7585321 | September 2009 | Cribier |
| 7682390 | March 2010 | Seguin |
| 7731742 | June 2010 | Schlick et al. |
| 7803185 | September 2010 | Gabbay |
| 7846203 | December 2010 | Cribier |
| 7846204 | December 2010 | Letac et al. |
| 7914569 | March 2011 | Nguyen et al. |
| D648854 | November 2011 | Braido |
| D652926 | January 2012 | Braido |
| D652927 | January 2012 | Braido et al. |
| D653341 | January 2012 | Braido et al. |
| D653342 | January 2012 | Braido et al. |
| D653343 | January 2012 | Ness et al. |
| D654169 | February 2012 | Braido |
| D654170 | February 2012 | Braido et al. |
| D660432 | May 2012 | Braido |
| D660433 | May 2012 | Braido et al. |
| D660967 | May 2012 | Braido et al. |
| 8449599 | May 2013 | Chau |
| 8579964 | November 2013 | Lane |
| 8591570 | November 2013 | Revuelta |
| 8840663 | September 2014 | Salahieh et al. |
| 9289291 | March 2016 | Gorman, III |
| 9480556 | November 2016 | Revuelta |
| 9585747 | March 2017 | Quadri |
| 9629721 | April 2017 | McKinnis et al. |
| 9763657 | September 2017 | Hacohen |
| 9763778 | September 2017 | Eidenschink et al. |
| 9763780 | September 2017 | Morriss |
| D800908 | October 2017 | Hariton |
| D815744 | April 2018 | Ratz |
| 9974651 | May 2018 | Hariton |
| 10010414 | July 2018 | Cooper |
| 10117744 | November 2018 | Ratz |
| 10149756 | December 2018 | Quadri |
| 10179042 | January 2019 | Braido |
| 10179044 | January 2019 | Ratz |
| 10390952 | August 2019 | Hariton |
| 2002/0036220 | March 2002 | Gabbay |
| 2003/0023303 | January 2003 | Palmaz et al. |
| 2003/0050694 | March 2003 | Yang et al. |
| 2003/0130726 | July 2003 | Thorpe et al. |
| 2004/0049262 | March 2004 | Obermiller et al. |
| 2004/0093075 | May 2004 | Kuehne |
| 2004/0210304 | October 2004 | Seguin et al. |
| 2005/0096726 | May 2005 | Sequin et al. |
| 2005/0137690 | June 2005 | Salahieh et al. |
| 2005/0137695 | June 2005 | Salahieh et al. |
| 2005/0137697 | June 2005 | Salahieh et al. |
| 2005/0240200 | October 2005 | Bergheim |
| 2005/0256566 | November 2005 | Gabbay |
| 2006/0008497 | January 2006 | Gabbay |
| 2006/0052867 | March 2006 | Revuelta |
| 2006/0074484 | April 2006 | Huber |
| 2006/0122692 | June 2006 | Gilad et al. |
| 2006/0149360 | July 2006 | Schwammenthal et al. |
| 2006/0173532 | August 2006 | Flagle et al. |
| 2006/0178740 | August 2006 | Stacchino et al. |
| 2006/0190074 | August 2006 | Hill et al. |
| 2006/0195180 | August 2006 | Kheradvar et al. |
| 2006/0206202 | September 2006 | Bonhoeffer et al. |
| 2006/0241744 | October 2006 | Beith |
| 2006/0241745 | October 2006 | Solem |
| 2006/0259120 | November 2006 | Vongphakdy et al. |
| 2006/0259136 | November 2006 | Nguyen et al. |
| 2006/0259137 | November 2006 | Artof et al. |
| 2006/0265056 | November 2006 | Nguyen et al. |
| 2006/0271166 | November 2006 | Thill et al. |
| 2006/0276813 | December 2006 | Greenberg |
| 2007/0010876 | January 2007 | Salahieh et al. |
| 2007/0027534 | February 2007 | Bergheim et al. |
| 2007/0043435 | February 2007 | Seguin et al. |
| 2007/0055358 | March 2007 | Krolik et al. |
| 2007/0067029 | March 2007 | Gabbay |
| 2007/0093890 | April 2007 | Eliasen et al. |
| 2007/0100435 | May 2007 | Case et al. |
| 2007/0118210 | May 2007 | Pinchuk |
| 2007/0213813 | September 2007 | Von Segesser et al. |
| 2007/0233228 | October 2007 | Eberhardt et al. |
| 2007/0244545 | October 2007 | Birdsall et al. |
| 2007/0244552 | October 2007 | Salahieh et al. |
| 2007/0288087 | December 2007 | Fearnot et al. |
| 2008/0021552 | January 2008 | Gabbay |
| 2008/0039934 | February 2008 | Styrc |
| 2008/0071361 | March 2008 | Tuval et al. |
| 2008/0071362 | March 2008 | Tuval et al. |
| 2008/0071366 | March 2008 | Tuval et al. |
| 2008/0071369 | March 2008 | Tuval et al. |
| 2008/0082164 | April 2008 | Friedman |
| 2008/0097595 | April 2008 | Gabbay |
| 2008/0114452 | May 2008 | Gabbay |
| 2008/0125853 | May 2008 | Bailey et al. |
| 2008/0140189 | June 2008 | Nguyen et al. |
| 2008/0147183 | June 2008 | Styrc |
| 2008/0154355 | June 2008 | Benichou et al. |
| 2008/0154356 | June 2008 | Obermiller et al. |
| 2008/0221672 | September 2008 | Lamphere |
| 2008/0243245 | October 2008 | Thambar et al. |
| 2008/0255660 | October 2008 | Guyenot et al. |
| 2008/0255662 | October 2008 | Stacchino et al. |
| 2008/0262602 | October 2008 | Wilk et al. |
| 2008/0269879 | October 2008 | Sathe et al. |
| 2009/0054976 | February 2009 | Tuval et al. |
| 2009/0112309 | April 2009 | Jaramillo et al. |
| 2009/0138079 | May 2009 | Tuval et al. |
| 2009/0157175 | June 2009 | Benichou |
| 2009/0287299 | November 2009 | Tabor et al. |
| 2010/0004740 | January 2010 | Seguin et al. |
| 2010/0036484 | February 2010 | Hariton et al. |
| 2010/0049306 | February 2010 | House et al. |
| 2010/0082094 | April 2010 | Quadri et al. |
| 2010/0087907 | April 2010 | Lattouf |
| 2010/0131055 | May 2010 | Case et al. |
| 2010/0168778 | July 2010 | Braido |
| 2010/0168839 | July 2010 | Braido et al. |
| 2010/0185277 | July 2010 | Braido et al. |
| 2010/0191326 | July 2010 | Alkhatib |
| 2010/0204781 | August 2010 | Alkhatib |
| 2010/0204785 | August 2010 | Alkhatib |
| 2010/0217382 | August 2010 | Chau et al. |
| 2010/0249911 | September 2010 | Alkhatib |
| 2010/0249923 | September 2010 | Alkhatib et al. |
| 2010/0256723 | October 2010 | Murray |
| 2010/0286768 | November 2010 | Alkhatib |
| 2010/0298931 | November 2010 | Quadri et al. |
| 2010/0312333 | December 2010 | Navia |
| 2011/0029072 | February 2011 | Gabbay |
| 2011/0098802 | April 2011 | Braido et al. |
| 2011/0137397 | June 2011 | Chau et al. |
| 2011/0218620 | September 2011 | Meiri et al. |
| 2011/0238168 | September 2011 | Pellegrini et al. |
| 2011/0264206 | October 2011 | Tabor |
| 2011/0313515 | December 2011 | Quadri |
| 2012/0035722 | February 2012 | Tuval |
| 2012/0078347 | March 2012 | Braido et al. |
| 2012/0078353 | March 2012 | Quadri |
| 2012/0215303 | August 2012 | Quadri |
| 2012/0303116 | November 2012 | Gorman, III |
| 2013/0150956 | June 2013 | Yohanan et al. |
| 2013/0184813 | July 2013 | Quadri et al. |
| 2013/0211508 | August 2013 | Lane et al. |
| 2013/0274870 | October 2013 | Lombardi et al. |
| 2013/0274873 | October 2013 | Delaloye et al. |
| 2014/0005771 | January 2014 | Braido et al. |
| 2014/0018911 | January 2014 | Zhou et al. |
| 2014/0018915 | January 2014 | Biadillah et al. |
| 2014/0046433 | February 2014 | Kovalsky |
| 2014/0163668 | June 2014 | Rafiee |
| 2014/0228940 | August 2014 | McKinnis et al. |
| 2014/0236292 | August 2014 | Braido |
| 2014/0243965 | August 2014 | Benson et al. |
| 2014/0249621 | September 2014 | Eidenschink |
| 2014/0277390 | September 2014 | Ratz |
| 2014/0277425 | September 2014 | Dakin |
| 2014/0324164 | October 2014 | Gross et al. |
| 2014/0330371 | November 2014 | Gloss et al. |
| 2015/0351904 | December 2015 | Cooper |
| 2016/0030165 | February 2016 | Mitra et al. |
| 2016/0030171 | February 2016 | Quijano et al. |
| 2016/0067038 | March 2016 | Park et al. |
| 2016/0074160 | March 2016 | Christianson et al. |
| 2016/0081799 | March 2016 | Leo |
| 2016/0095701 | April 2016 | Dale et al. |
| 2016/0113768 | April 2016 | Ganesan et al. |
| 2016/0158003 | June 2016 | Wallace et al. |
| 2016/0158007 | June 2016 | Centola |
| 2016/0199180 | July 2016 | Zeng et al. |
| 2016/0206423 | July 2016 | O'Connor |
| 2016/0228249 | August 2016 | Mantanus et al. |
| 2016/0235529 | August 2016 | Ma |
| 2016/0256271 | September 2016 | Backus |
| 2016/0278922 | September 2016 | Braido et al. |
| 2016/0278923 | September 2016 | Krans et al. |
| 2016/0302918 | October 2016 | Keidar |
| 2016/0302919 | October 2016 | Scorsin et al. |
| 2016/0310268 | October 2016 | Oba |
| 2016/0317301 | November 2016 | Quadri et al. |
| 2016/0331525 | November 2016 | Straubinger et al. |
| 2016/0361161 | December 2016 | Braido et al. |
| 2017/0000604 | January 2017 | Conklin et al. |
| 2017/0042673 | February 2017 | Vietmeier |
| 2017/0049565 | February 2017 | Bailey et al. |
| 2017/0056169 | March 2017 | Johnson et al. |
| 2017/0071733 | March 2017 | Ghione et al. |
| 2017/0079786 | March 2017 | Li et al. |
| 2017/0095326 | April 2017 | Lim et al. |
| 2017/0095328 | April 2017 | Cooper et al. |
| 2017/0112621 | April 2017 | Salahieh et al. |
| 2017/0128204 | May 2017 | Morriss |
| 2017/0128207 | May 2017 | Tuval et al. |
| 2017/0143481 | May 2017 | Morriss et al. |
| 2017/0143482 | May 2017 | Kveen et al. |
| 2017/0143484 | May 2017 | Braido |
| 2017/0143485 | May 2017 | Gorman, III et al. |
| 2018/0116790 | May 2018 | Ratz |
| 2018/0116798 | May 2018 | Perszyk |
| 2019/0060070 | February 2019 | Groothuis |
| 2019/0167423 | June 2019 | Hariton |
| 2019/0262129 | August 2019 | Cooper |
| 2019/0321171 | October 2019 | Morriss |
| 19857887 | Jul 2000 | DE | |||
| 10121210 | Nov 2002 | DE | |||
| 102005003632 | Aug 2006 | DE | |||
| 202008009610 | Dec 2008 | DE | |||
| 0850607 | Jul 1998 | EP | |||
| 1000590 | May 2000 | EP | |||
| 1360942 | Nov 2003 | EP | |||
| 1584306 | Oct 2005 | EP | |||
| 1598031 | Nov 2005 | EP | |||
| 1926455 | Jun 2008 | EP | |||
| 2777616 | Sep 2014 | EP | |||
| 2847800 | Jun 2004 | FR | |||
| 2850008 | Jul 2004 | FR | |||
| 9117720 | Nov 1991 | WO | |||
| 9716133 | May 1997 | WO | |||
| 9832412 | Jul 1998 | WO | |||
| 9913801 | Mar 1999 | WO | |||
| 01028459 | Apr 2001 | WO | |||
| 0149213 | Jul 2001 | WO | |||
| 01054625 | Aug 2001 | WO | |||
| 01056500 | Aug 2001 | WO | |||
| 01076510 | Oct 2001 | WO | |||
| 0236048 | May 2002 | WO | |||
| 0247575 | Jun 2002 | WO | |||
| 02067782 | Sep 2002 | WO | |||
| 03047468 | Jun 2003 | WO | |||
| 2005070343 | Aug 2005 | WO | |||
| 06073626 | Jul 2006 | WO | |||
| 07071436 | Jun 2007 | WO | |||
| 08070797 | Jun 2008 | WO | |||
| 2010008548 | Jan 2010 | WO | |||
| 2010008549 | Jan 2010 | WO | |||
| 2010096176 | Aug 2010 | WO | |||
| 2010098857 | Sep 2010 | WO | |||
| 2012068377 | May 2012 | WO | |||
Other References
|
"Direct-Access Valve Replacement", Christoph H. Huber, et al., Journal of the American College of Cardiology, vol. 46, No. 2, (Jul. 19, 2005). cited by applicant . "Minimally invasive cardiac surgery", M. J. Mack, Surgical Endoscopy, 2006, 20:S488-S492, DOI: 10.1007/s00464-006-0110-8 (presented Apr. 24, 2006). cited by applicant . "Percutaneous Aortic Valve Implantation Retrograde From the Femoral Artery", John G. Webb et al., Circulation, 2006; 113:842-850 (Feb. 6, 2006). cited by applicant . "Transapical aortic valve implantation: an animal feasibility study"; Todd M. Dewey et al., the annals of thoracic surgery 2006; 82: 110-6 (Feb. 13, 2006). cited by applicant . "Transapical approach for sutureless stent-fixed aortic valve implantation: experimental results"; Th. Walther et al., European Journal of Cardio-thoracic Surgery 29 (2006) 703-708 (Jan. 30, 2006). cited by applicant . "Transapical Transcatheter Aortic Valve Implantation in Humans", Samuel V. Lichtenstein et al., Circulation. 2006; 114: 591-596 (Jul. 31, 2006). cited by applicant . Braido et al., U.S. Appl. No. 29/375,243, filed Sep. 20, 2010, titled "Surgical Stent Assembly". cited by applicant . Catheter-implanted prosthetic heart valves, Knudsen, L.L., et al., The International Journal of Artificial Organs, vol. 16, No. 5 1993, pp. 253-262. cited by applicant . International Search Report for Application No. PCT/US2016/036560 dated Aug. 18, 2016. cited by applicant . Is It Reasonable to Treat All Calcified Stenotic Aortic Valves With a Valved Stent?, 579-584, Zegdi, Rachid, MD, PhD at al., J. of the American College of Cardiology, vol. 51, No. 5, Feb. 5, 2008. cited by applicant . Moazami, N. et al., "Transluminal Aortic Valve Placement," ASAIO Journal, Sep./Oct. 1996, pp. M381-M385, vol. 42. cited by applicant . Percutaneous aortic valve replacement: resection before implantation, 836-840, Quaden, Rene et al., European J. of Cardio-thoracic Surgery, 27 (2005). cited by applicant . Textbook "Transcatheter Valve Repair", 2006, pp. 165-186. cited by applicant . Transluminal Catheter Implanted Prosthetic Heart Valves, Andersen, Henning Rud, International Journal of Angiology 7:102-106 (1998). cited by applicant . Transluminal implantation of artificial heart valves, Andersen, H. R., et al., European Heart Joumal (1992) 13, 704-708. cited by applicant . U.S. Appl. No. 29/375,260, filed Sep. 20, 2010. cited by applicant. |
Primary Examiner: Stewart; Alvin J
Attorney, Agent or Firm: Lerner, David, Littenberg, Krumholz & Mentlik, LLP
Parent Case Text
CROSS-REFERENCE TO RELATED APPLICATIONS
This application is a continuation of U.S. application Ser. No. 15/177,598, filed on Jun. 9, 2016, and claims the benefit of the filing date of U.S. Provisional Patent Application No. 62/174,690 filed Jun. 12, 2015, the disclosures of which are hereby incorporated herein by reference.
Claims
The invention claimed is:
1. A prosthetic heart valve having an inflow end and an outflow end, comprising: a collapsible and expandable stent including a plurality of first struts forming a substantially cylindrical portion and a plurality of second struts that are curled when the stent is in an expanded condition, each of the second struts having a first end attached to the cylindrical portion, a free end projecting radially outward from the cylindrical portion, and an anchoring member at the free end for engaging adjacent heart tissue; and a collapsible and expandable valve assembly coupled to the stent and including a plurality of leaflets, wherein the anchoring member comprises an elongated pivot head including a substantially straight strut attached to the free end of the second strut at a pivot point approximately in the middle of the substantially straight strut, the elongated pivot head being rotatable about the pivot point and within a plane from a first position to a second position.
2. The prosthetic heart valve of claim 1, wherein at least some of the first ends are attached to the stent adjacent the outflow end.
3. The prosthetic heart valve of claim 1, wherein at least some of the first ends are attached to the stent adjacent the inflow end.
4. The prosthetic heart valve of claim 1, wherein at least some of the first ends are attached to the stent adjacent the outflow end and others of the first ends are attached to the stent adjacent the inflow end.
5. The prosthetic heart valve of claim 4, wherein each of the free ends projects toward a midsection of the stent between the inflow end and the outflow end.
6. The prosthetic heart valve of claim 1, wherein the first position is orthogonal to the free end of the second strut and the second position is oblique to the free end of the second strut.
7. The prosthetic heart valve of claim 1, wherein the elongated pivot head is shape-set to the second position.
8. The prosthetic heart valve of claim 1, wherein the valve assembly and the stent are capable of replacing the function of at least one of a native mitral valve or a native tricuspid valve.
9. A prosthetic heart valve having an inflow end and an outflow end, comprising: a collapsible and expandable stent including a plurality of first struts forming a substantially cylindrical portion and a plurality of second struts that are curled when the stent is in an expanded condition, each of the second struts having a first end attached to the cylindrical portion at the outflow end, a free end projecting radially outward from the cylindrical portion, and an anchoring member at the free end; and a collapsible and expandable valve assembly coupled to the stent and including a plurality of leaflets, wherein the anchoring member is bell-shaped and comprises a base having a first width attached to the free end of the second strut having a second width less than the first width and two arcuate atraumatic ends extending from the base radially outward and away from the first end of the second strut.
10. The prosthetic heart valve of claim 9, wherein the anchoring member defines a depressed portion for capturing chordae tendineae between the two arcuate ends.
11. The prosthetic heart valve of claim 10, wherein the two arcuate atraumatic ends are convex relative to the base.
Description
BACKGROUND OF THE INVENTION
The present disclosure relates to heart valve repair and, in particular, to collapsible prosthetic heart valves. More particularly, the present disclosure relates to devices and methods for repairing and/or replacing the functionality of native valve leaflets.
Diseased and/or defective heart valves may lead to serious health complications. One method of addressing this condition is to replace a non-functioning heart valve with a prosthetic valve. Prosthetic heart valves that are collapsible to a relatively small circumferential size can be delivered into a patient less invasively than valves that are not collapsible. For example, a collapsible valve may be delivered into a patient via a tube-like delivery apparatus such as a catheter, a trocar, a laparoscopic instrument, or the like. This collapsibility can avoid the need for a more invasive procedure such as full open-chest, open-heart surgery.
Collapsible prosthetic heart valves typically take the form of a valve structure mounted on a stent. There are two types of stents on which the valve structures are ordinarily mounted: a self-expanding stent and a balloon-expandable stent. To place such valves into a delivery apparatus and ultimately into a patient, the valve must first be collapsed or crimped to reduce its circumferential size.
When a collapsed prosthetic valve has reached the desired implant site in the patient (e.g., at or near the annulus of the patient's heart valve that is to be replaced by the prosthetic valve), the prosthetic valve can be deployed or released from the delivery apparatus and re-expanded to full operating size. For balloon-expandable valves, this generally involves releasing the entire valve, assuring its proper location, and then expanding a balloon positioned within the valve stent. For self-expanding valves, on the other hand, the stent automatically expands as the sheath covering the valve is withdrawn.
SUMMARY OF THE INVENTION
In some embodiments, a prosthetic heart valve having an inflow end and an outflow end, includes a collapsible and expandable stent having a plurality of commissure features, a plurality of first struts and a plurality of second struts. The plurality of first struts define a substantially cylindrical portion of the stent and the plurality of second struts have first ends attached to the cylindrical portion and free ends projecting radially outward of the cylindrical portion and configured to couple to adjacent heart tissue to anchor the stent. A collapsible and expandable valve assembly is disposed within the stent and has a plurality of leaflets coupled to the commissure features.
BRIEF DESCRIPTION OF THE DRAWINGS
Various embodiments of the present disclosure are disclosed herein with reference to the drawings, wherein:
FIG. 1 is a schematic cutaway representation of a human heart showing a transapical delivery approach;
FIG. 2A is a schematic representation of a native mitral valve and associated structures during normal operation;
FIG. 2B is a schematic representation of a native mitral valve having a prolapsed leaflet;
FIG. 3 is a schematic longitudinal cross-section of one embodiment of a prosthetic heart valve having a stent, a valve assembly, and a frame;
FIG. 4 is a schematic longitudinal cross-section of one embodiment of a prosthetic heart valve having a stent, a valve assembly, and curling struts;
FIG. 5 is a schematic longitudinal cross-section of one embodiment of a prosthetic heart valve having a stent, a valve assembly, and curved struts;
FIGS. 6A-L are schematic representations of several variations of the terminal ends of selected struts; and
FIG. 7 is a schematic longitudinal cross-section of one embodiment of a prosthetic heart valve having a stent, a valve assembly, and curling struts confined to a region adjacent the outflow section.
Various embodiments of the present disclosure will now be described with reference to the appended drawings. It is to be appreciated that these drawings depict only some embodiments of the disclosure and are therefore not to be considered limiting of its scope.
DETAILED DESCRIPTION
In conventional collapsible prosthetic heart valves, the stent is usually anchored within the native valve annulus via radial forces exerted by the expanding stent against the native valve annulus. If the radial force is too high, damage may occur to heart tissue. If, instead, the radial force is too low, the heart valve may move from its implanted position, for example, into the left ventricle. Because such anchoring partly depends on the presence of calcification or plaque in the native valve annulus, it may be difficult to properly anchor the valve in locations where plaque is lacking (e.g., the mitral valve annulus).
In view of the foregoing, there is a need for further improvements to the devices, systems, and methods for restoring and/or replacing the function of a native heart valve, such as a mitral valve, a tricuspid valve, an aortic valve, or a pulmonary valve. Among other advantages, the present disclosure may address one or more of these needs. While many of the examples disclosed herein are described with reference to a specific valve (e.g., a mitral valve or a tricuspid valve), it will be understood that many of the examples are not so limited and that the concepts described apply equally to other heart valves unless expressly limited herein.
Blood flows through the mitral valve from the left atrium to the left ventricle. As used herein, the term "inflow," when used in connection with a prosthetic mitral heart valve, refers to the end of the heart valve closest to the left atrium when the heart valve is implanted in a patient, whereas the term "outflow," when used in connection with a prosthetic mitral heart valve, refers to the end of the heart valve closest to the left ventricle when the heart valve is implanted in a patient. When used in connection with a prosthetic aortic valve, "inflow" refers to the end closest to the left ventricle and "outflow" refers to the end closest to the aorta. The same convention is applicable for other valves wherein "inflow" and "outflow" are defined by the direction of blood flow therethrough. "Trailing" is to be understood as relatively close to the user, and "leading" is to be understood as relatively farther away from the user. As used herein, the terms "proximal," "distal," "leading" and "trailing" are to be taken as relative to a user using the disclosed delivery devices. "Proximal" or "trailing end" are to be understood as relatively close to the user and "distal" or "leading end" are to be understood as relatively farther away from the user. Also, as used herein, the words "substantially," "approximately," "generally" and "about" are intended to mean that slight variations from absolute are included within the scope of the structure or process recited.
FIG. 1 is a schematic representation of a human heart 100. The human heart includes two atria and two ventricles: a right atrium 112 and a left atrium 122, and a right ventricle 114 and a left ventricle 124. As illustrated in FIG. 1, the heart 100 further includes an aorta 110, and an aortic arch 120. Disposed between the left atrium and the left ventricle is the mitral valve 130. The mitral valve 130, also known as the bicuspid valve or left atrioventricular valve, is a dual-flap that opens as a result of increased pressure in the left atrium as it fills with blood. As atrial pressure increases above that of the left ventricle, the mitral valve opens and blood passes toward the left ventricle. Blood flows through heart 100 in the direction shown by arrows "B".
A dashed arrow, labeled "TA", indicates a transapical approach for repairing or replacing heart valves, such as a mitral valve. In transapical delivery, a small incision is made between the ribs and into the apex of the left ventricle 124 at position "P1" in heart wall 150 to deliver a prosthesis or device to the target site.
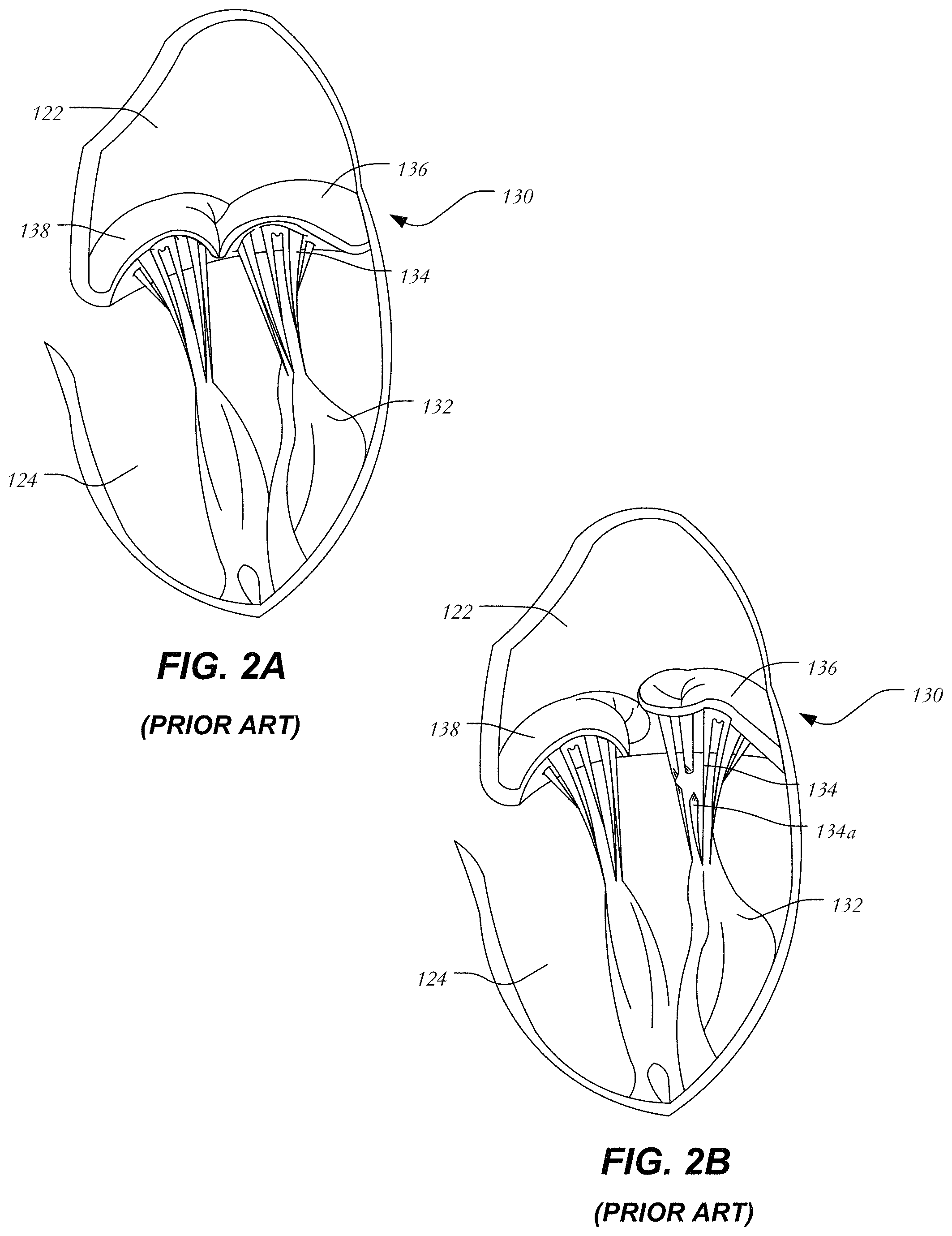
FIG. 2A is a more detailed schematic representation of a native mitral valve 130 and its associated structures. Mitral valve 130 includes two flaps or leaflets, a posterior leaflet 136 and an anterior leaflet 138, disposed between left atrium 122 and left ventricle 124. Cord-like tendons known as chordae tendineae 134 connect the two leaflets 136, 138 to the medial and lateral papillary muscles 132. During atrial systole, blood flows from the left atrium to the left ventricle down the pressure gradient. When the left ventricle contracts in ventricular systole, the increased blood pressure in the chamber pushes the mitral valve to close, preventing backflow of blood into the left atrium. Since the blood pressure in the left atrium is much lower than that in the left ventricle, the flaps attempt to evert to the low pressure regions. The chordae tendineae prevent the eversion by becoming tense, thus pulling the flaps and holding them in the closed position.
FIG. 2B is a schematic representation of mitral valve prolapse as discussed above. Posterior leaflet 136 has prolapsed into left atrium 122. Moreover, certain chordae tendineae have stretched and others have ruptured. Because of damaged chordae 134a, even if posterior leaflet 136 returns to its intended position, it will eventually resume the prolapsed position due to being inadequately secured. Thus, mitral valve 130 is incapable of functioning properly and blood is allowed to return to the left atrium and the lungs. It will be understood that in addition to chordae damage, other abnormalities or failures may be responsible for mitral valve insufficiency.
FIG. 3 is a longitudinal cross-section of prosthetic heart valve 200 in accordance with one embodiment of the present disclosure. Prosthetic heart valve 200 is a collapsible prosthetic heart valve designed to replace the function of the native mitral valve of a patient. (See native mitral valve 130 of FIGS. 1-2.) Generally, prosthetic valve 200 has inflow end 210 and outflow end 212. Prosthetic valve 200 may be substantially cylindrically shaped and may include features for anchoring, as will be discussed in more detail below. When used to replace native mitral valve 130, prosthetic valve 200 may have a low profile so as not to interfere with atrial function.
Prosthetic heart valve 200 includes stent 250, which may be formed from biocompatible materials that are capable of self-expansion, such as, for example, shape-memory alloys including nitinol. Alternatively, stent 250 may be formed of a material suitable for forming a balloon-expandable stent. Stent 250 may include a plurality of struts 252 that form closed cells 254 connected to one another in one or more annular rows around the stent. Cells 254 may all be of substantially the same size around the perimeter and along the length of stent 250. Alternatively, cells 254 near inflow end 210 may be larger than the cells near outflow end 212. Stent 250 may be expandable to provide a radial force to assist with positioning and stabilizing prosthetic heart valve 200 within the native mitral valve annulus.
Prosthetic heart valve 200 may also include valve assembly 260, including a pair of leaflets 262 attached to a cylindrical cuff 264. Leaflets 262 replace the function of native mitral valve leaflets 136 and 138 described above with reference to FIG. 2. That is, leaflets 262 coapt with one another to function as a one-way valve. It will be appreciated, however, that prosthetic heart valve 200 may have more than two leaflets when used to replace a mitral valve or other cardiac valves within a patient. Valve assembly 260 of prosthetic heart valve 200 may be substantially cylindrical, or may taper outwardly from outflow end 212 to inflow end 210. Both cuff 264 and leaflets 262 may be wholly or partly formed of any suitable biological material, such as bovine or porcine pericardium, or polymers, such as PTFE, urethanes and the like.
When used to replace a native mitral valve, valve assembly 260 may be sized in the range of about 20 mm to about 40 mm in diameter. Valve assembly 260 may be secured to stent 250 by suturing to struts 252 or by using tissue glue, ultrasonic welding or other suitable methods.
An optional frame 300 may surround and house valve assembly 260 and stent 250. Frame 300 may be formed of a braided material in various configurations to create shapes and/or geometries for engaging tissue and filling the spaces between valve assembly 260 and the native valve annulus. As shown in FIG. 3, frame 300 includes a plurality of braided strands or wires 305 arranged in three-dimensional shapes. In one example, wires 305 form a braided metal fabric that is both resilient and capable of heat treatment to substantially set a desired preset shape. One class of materials which meets these qualifications is shape-memory alloys. One example of a suitable shape-memory alloy is nitinol. It is also contemplated that wires 305 may comprise various materials other than nitinol that have elastic and/or memory properties, such as spring stainless steel, alloys such as Elgiloy.RTM., Hastelloy.RTM., and MP35N.RTM., CoCrNi alloys (e.g., trade name Phynox), CoCrMo alloys, or a mixture of metal and polymer fibers. Depending on the individual material selected, the strand diameter, number of strands, and pitch may be altered to achieve desired properties for frame 300.
In its simplest configuration, shown in FIG. 3, frame 300 may be formed in a cylindrical or tubular configuration having inlet end 310, outlet end 312 and lumen 315 extending between inlet end 310 and outlet end 312 for housing stent 250 and valve assembly 260. However, in certain embodiments stent 250 may be omitted, and valve assembly 260 may be directly attached to frame 300 using any of the techniques described above for attaching valve assembly 260 to stent 250. Frame 300 may be radially collapsed from a relaxed or preset configuration to a compressed or reduced configuration for delivery into the patient. Once released after delivery, the shape-memory properties of frame 300 may cause it to re-expand to its relaxed or preset configuration. Frame 300 may also be locally compliant in a radial direction such that a force exerted in the direction of arrow F deforms a portion of the frame. In this manner, irregularities in the native valve annulus may be filled by frame 300, thereby preventing paravalvular leakage. Moreover, portions of frame 300 may endothelialize and in-grow into the heart wall over time, providing permanent stability and a low thrombus surface.
FIG. 4 illustrates a variation in which prosthetic heart valve 400 includes outwardly curling struts to aid in its fixation to heart tissue. Prosthetic heart valve 400 may extend between inflow end 210 and outflow end 212 and include all the elements disclosed above including stent 250 formed of struts 252 defining cells 254, and valve assembly 260 having leaflets 262 and cuff 264. Stent 250 may be substantially cylindrical as shown and may further include two rows of curling struts 410a, 410b that project radially outward from the general stent body to anchor the stent at a predetermined location in the native valve annulus. A first row 420 of curling struts 410a is disposed adjacent inflow end 210 of prosthetic heart valve 400 and a second row 422 of curling struts 410b is disposed adjacent outflow end 212. Each curling strut 410a, 410b has a first end 412a connected to stent 250 and a free end 412b, with a curled configuration between these ends. Curling struts 410a, 410b may be formed of the same material as struts 252 and may be formed integrally with stent 250 by laser cutting from the same tube that forms stent 250 or separately formed and attached to stent 250 using welding techniques or other suitable methods. As shown in FIG. 4, the first end 412a of each curling strut 410a is connected to stent 250 at fixation points 430 at the bottom of the first full row of cells 254 adjacent inflow end 210 of prosthetic heart valve 400 and the first end 412a of each curling strut 410b is connected to stent 250 at fixation points 432 at the top of the last (i.e., bottom-most) full row of cells 254 adjacent outflow end 212 of the prosthetic heart valve. It will be understood that other fixation points (e.g., closer to inflow end 210 or further from inflow end 210) are possible.
As noted above, each curling strut 410a, 410b has a curled configuration between its ends. Curling struts 410a initially extend upward from fixation points 430 toward inflow end 210 before bending outwardly and downwardly toward outflow end 212 to form a substantially "fiddlehead" shape. Likewise, each curling strut 410b initially extends downward from a fixation point 432 toward outflow end 212 before bending outwardly and upwardly toward inflow end 210. Curling struts 410a,410b may be subjected to heat treatment to substantially preset their desired curled shape. During the delivery of prosthetic heart valve 400 into a patient, curling struts 410a,410b may be distorted to a substantially linear configuration within the sheath of a delivery device and may return to their curled configuration when released from the sheath.
When heart valve 400 is implanted, first row 420 of curling struts 410a may engage upper portions of the native mitral valve (i.e., portions of the native mitral valve in left atrium 122) or the atrial wall itself, while second row 422 of curling struts 410b may engage lower portions of the native mitral valve (i.e., portions of the native mitral valve in left ventricle 124) or the ventricular wall itself. The engagement of curling struts 410a and 410b with the surrounding native tissue may help to affix heart valve 400 in the proper position in the native mitral valve annulus.
FIG. 5 illustrates another variation in which prosthetic heart valve 500 includes projecting curved struts to aid in its fixation to heart tissue. Prosthetic heart valve 500 may extend between inflow end 210 and outflow end 212 and include all the elements described above in connection with heart valve 400, including stent 250 formed of struts 252 defining cells 254, and valve assembly 260 having leaflets 262 and cuff 264. As shown, prosthetic heart valve 500 includes two rows of curved struts 510a, 510b.
A first row 520 of curved struts 510a is disposed adjacent inflow end 210 of prosthetic heart valve 500 and a second row 522 of curved struts 510b is disposed adjacent outflow end 212. Each curved strut 510a, 510b has a first end 512a connected to stent 250, a free end 512b and a bowed configuration between these ends. Curved struts 510a, 510b may be formed of the same material as struts 252 and may be formed integrally with stent 250 by laser cutting from the same tube that forms stent 250 or separately and attached to stent 250 using welding or another suitable method. Curved struts 510a, 510b may be between about 3.0 mm and about 8.0 mm in length. In at least some examples, curved struts 510a, 510b are approximately 5.0 mm in length to aid in fixation. Additionally, curved struts 510a, 510b may apply a small radial force on the surrounding tissue. For example, the applied force may be enough to maintain contact to avoid thrombus, but not enough damage the tissue. In at least some examples, a radial force of between about 0.1 N and about 2.0 N may be exerted by the curved struts on the surrounding tissue. The force applied by curved struts on surrounding tissues may also be selected by adjusting the thickness and/or width of the curved struts. In some examples, curved struts 510a, 510b may have a width that is between about 20% to about 50% of struts 252. In some examples, curved struts 510a, 510b may have a wall thickness that is between about 20% to about 50% of struts 252.
In the example shown, the first end 512a of each curved strut 510a is connected to stent 250 at the top of the first full row of cells 254 adjacent inflow end 210 of prosthetic heart valve 500 and the first end 512a of each curved strut 510b is connected to stent 250 at the bottom of the last full row of cells 254 adjacent outflow end 212 of the prosthetic heart valve. Each curved strut 510a extends from its connection to stent 250 downwardly towards a midsection M of heart valve 500 and radially outwardly from the stent. Likewise, each curved strut 510b extends from its connection to stent 250 upwardly toward midsection M and radially outwardly from the stent. The connection of the curved struts to cells 254 does not have to be at the junction of two struts 252. Rather, as shown in FIG. 5, prosthetic heart valve 500 may include curved struts 510c that are coupled to selected struts 252a, 252b at spaced distances from the junction between the two. In this example, two curved struts 510c originate from one cell 254. This configuration of two curved struts 510c per cell 254 may be termed a "double takeoff" configuration and may be repeated at inflow end 210, outflow end 212, or both the inflow end and the outflow end. This "double takeoff" configuration may also alternate with the single curved struts 510a,510b, or replace all of the single curved struts 510a,510b.
Each curved strut 510a, 510b, 510c may terminate in a substantially circular eyelet 530 that forms a smooth and blunted shape at its free end 512b to prevent trauma to heart tissue. As shown in greater detail in FIG. 6A, strut 610a may terminate in circular eyelet 611, having an aperture 612. Aperture 612 may be useful to mate with portions of a delivery device for maneuvering and positioning heart valve 500 during deployment. Instead of round eyelets, curved struts 510a, 510b, 510c may have other smoothly curved eyelets on their free ends, such as oval or elliptical eyelets. Further, these smoothly curved structures need not include an aperture, but may be in the form of a solid disk, oval or ellipse. Alternatively, one or more of curved struts 510a, 510b 510c may include an anchoring feature at its free end as will be described with reference to FIGS. 6B-L. In the following examples, reference may be made to anchoring to heart tissue. It is intended by this that the features described may couple to at least one of an atrial wall, a ventricular wall, a native valve leaflet, heart muscle, papillary muscles, tendons, chordae tendineae or any other tissue adjacent a heart valve, such as a mitral valve or a tricuspid valve. Unless otherwise noted, each of the features shown in FIGS. 6A-6L and described below (or described above in the case of the feature of FIG. 6A) may be provided on any one or more of the curved struts of prosthetic heart valve 500.
FIG. 6B illustrates a variation in which strut 610b terminates in a bell-shaped end 620 having a broad base 621 composed of two convex ends 622 that are curved and disposed on either side of a middle depressed portion 623. Without being bound to any particular theory, it is believed that broad base 621 provides a larger surface area for pushing against native tissue and reduces the risk of trauma to heart tissue, and that depressed portion 623 may provide a region to which chordae tendineae may attach. FIG. 6C illustrates another variation in which strut 610c terminates in a rounded end 625 having a narrowed neck 626 and a bulbous crown 627. Narrowed neck 626 may add flexibility to strut 610c while bulbous crown 627 provides an atraumatic contact point with body tissue. FIG. 6D illustrates another variation in which strut 610d includes a pivoting head 630 that is capable of rotating at pivot 631 to alternate between a first position R1 and a second position R2 shown in phantom lines at an oblique angle to the strut. It will be understood that pivoting head 630 may be heat set or otherwise shape set so as to be disposed in position R2 during delivery of prosthetic heart valve 500 into the patient, and may then return to position R1 after deployment for anchoring. In FIG. 6E, strut 610e terminates in arrow-shaped end 635 having two outwardly extending wings 636 defining a cavity 637 between strut 610e and each wing. Cavities 637 may capture certain portions of the heart tissue such as, for example, chordae tendineae. FIG. 6F illustrates strut 610f which terminates in corkscrew 640 formed of a helical member 641 that progressively narrows to a point 642. Corkscrew 640 may be configured to engage certain heart tissue by having the tissue wrap around the progressively narrowing member or by piercing the tissue with point 642. A similar configuration, shown in FIG. 6G, illustrates strut 610g having opposing teeth-like barbs 645,646 which capture heart tissue. Each barb 645, 646 is substantially triangular and angled slightly away from the free end of strut 610g such that the teeth are capable of grasping onto heart tissue when implanted. FIG. 6H illustrates another example for anchoring a strut to heart tissue. Strut 610h includes an energy-excitable region 650, for example, having bio-glues like cyanoacrylates that bonds to heart tissue when excited by an energy source (e.g., laser energy, ultrasound, etc.). Instead of an energy-excitable region, strut 610i of FIG. 6I includes a chemical bonding portion 655, which includes a coating on strut 610i to aid in attachment to heart tissue. In one example, chemical bonding region 655 includes a biocompatible adhesive 656 that is coated onto one or more surfaces of strut 610i. In FIG. 6J, strut 610j includes a region 660 having pores 661. Porous region 660 may be formed from a different material than the remainder of strut 610j and may be biodegradable. Additionally, an adhesive 662 or the like may be added to pores 661 to aid in anchoring. FIG. 6K illustrates two struts 610k1,610k2 each having a polarized region 665,666, respectively. Polarized regions 665,666 may be magnetic and may have opposite polarities such that, when brought close together, struts 610k1,610k2 will be attracted to one another and magnetically clamp onto tissue. In some examples, certain struts that are used for anchoring may be divided into a first group of struts having a first polarized region 610k1 and a second group of struts having a second polarized region 610k2, the first and second polarized regions being of opposite polarities. In at least some examples, struts 610k1 having polarized regions 665 may be in a first row and struts 610k2 having polarized regions 666 may be in a second row, and the two rows may be disposed on opposite sides of heart tissue. In FIG. 6L, strut 610l includes clamp 670 having opposed clamps 671 defining a receiving portion 672 therebetween for receiving a portion of heart tissue.
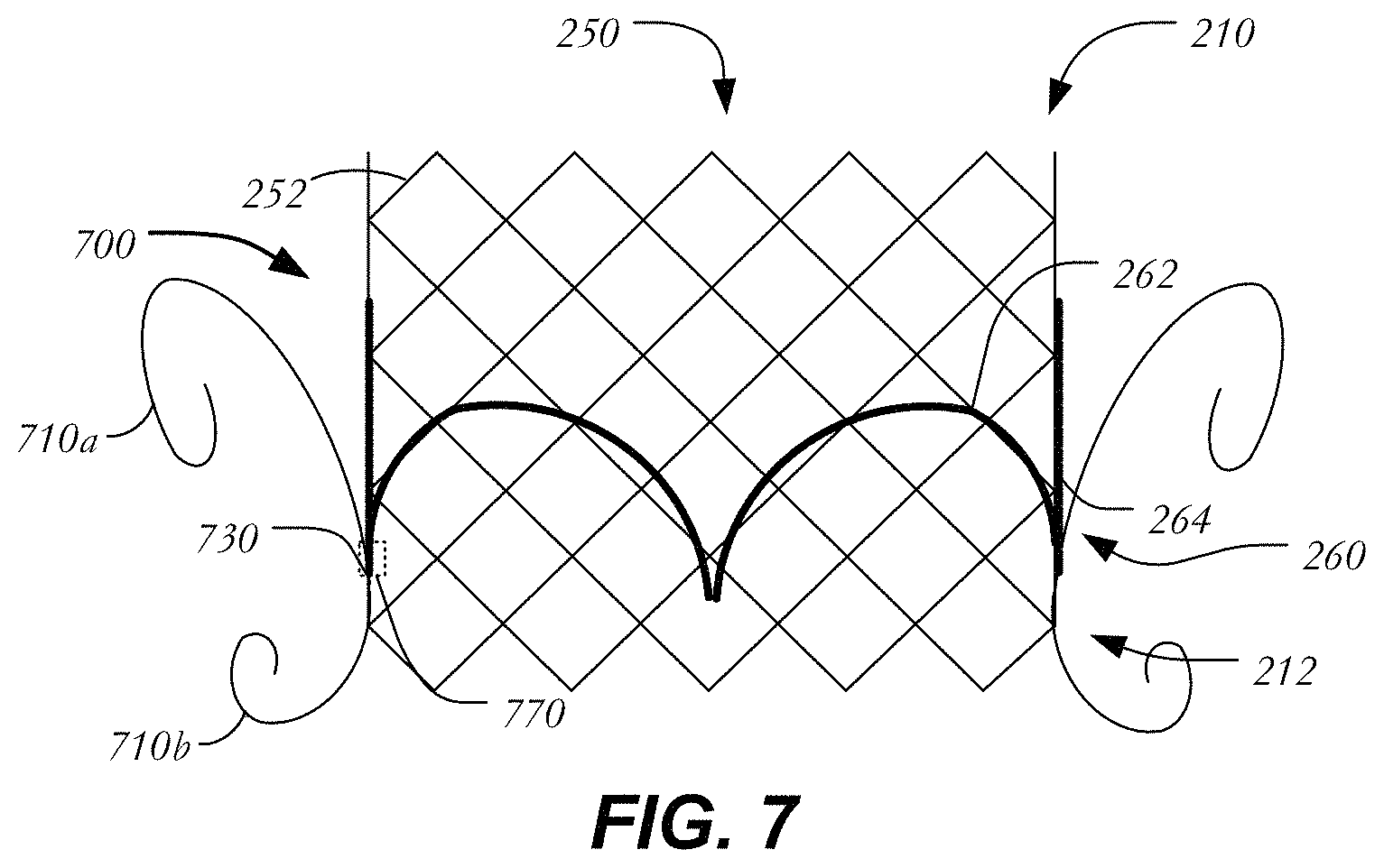
FIG. 7 illustrates another variation in which prosthetic heart valve 700 includes projecting curved struts to aid in its fixation to heart tissue. Prosthetic heart valve 700 may extend between inflow end 210 and outflow end 212 and include all the elements described above in connection with heart valve 400, including stent 250 formed of struts 252 defining cells 254, and valve assembly 260 having leaflets 262 and cuff 264. Similar to prosthetic heart valve 400 of FIG. 4, prosthetic heart valve 700 includes upper curling struts 710a and lower curling struts 710b. Curling struts 710a, 710b may be between about 10 mm and about 20 mm. In contrast to prosthetic heart valve 400, however, curling struts 710a-b are arranged in pairs, with each pair originating at a single fixation point 730. In this case, fixation points 730 are disposed close to inflow end 210 (e.g., closer to the atrium when prosthetic heart valve 700 is implanted) to minimize protrusion into the left ventricular outflow tract. In some examples, fixation points 730 may be disposed equidistant between inflow end 210 and outflow end 212. Alternatively, fixation points 730 may be disposed closer to outflow end 212 than to inflow end 210. In at least some examples, curling struts 710a are longer than curling struts 710b and fixation point 730 is disposed closer to outflow end 212 than inflow end 210. Curling struts may apply a radial force on surrounding tissue in the range described above with respect to cured struts. In some examples, curling struts 710a, 710b may have a width that is between about 20% to about 50% of struts 252. In some examples, curling struts 710a, 710b may have a wall thickness that is between about 20% to about 50% of struts 252. Though leaflets 262 and stent 250 are illustrated in a simplified manner, it will be appreciated that leaflets 262 may be attached to stent 250 at commissure features 770 and that fixation points 730 may be disposed adjacent to or at the same longitudinal position as the commissure features as shown, or anywhere between commissure features 770 and outflow end 212.
According to the disclosure, a prosthetic heart valve has an inflow end and an outflow end, and may include a collapsible and expandable stent including a plurality of commissure features, a plurality of first struts and a plurality of second struts, the plurality of first struts defining a substantially cylindrical projection and the plurality of second struts projecting radially outward from the cylindrical portion and configured to couple to adjacent heart tissue to anchor the stent, and a collapsible and expandable valve assembly disposed within the stent and having a plurality of leaflets coupled to the commissure features; and/or
the plurality of second struts may include curved struts, the free ends of the curved struts projecting toward a midsection of the stent; and/or
the plurality of second struts may include curling struts, the curling struts forming a fiddlehead shape between the first ends and the second ends; and/or
the first ends may be disposed adjacent the outflow end; and/or
the first ends may be disposed at a longitudinal position between the plurality of commissure features and the outflow end; and/or
the free ends may terminate in at least one of a bell-shaped base, a rounded end having a narrowed neck or a bulbous crown; and/or
the free ends may terminate in a pivoting head; and/or
the free ends may terminate in at least one of an arrow-shaped end, a corkscrew, or a plurality of barbs; and/or
the free ends may terminate in at least one of a porous region, a chemical bonding region, or an electrically excitable region; and/or
the free ends may terminate in a region coated with a biocompatible adhesive; and/or
the free ends of a first group of the second struts may have a first polarity and the free ends of a second group of the second struts may have a second polarity opposite a first polarity; and/or
the valve assembly and the stent may be capable of replacing the function of at least one of a native mitral valve and a native tricuspid valve.
Although the invention herein has been described with reference to particular embodiments, it is to be understood that these embodiments are merely illustrative of the principles and applications of the present invention. It is therefore to be understood that numerous modifications may be made to the illustrative embodiments and that other arrangements may be devised without departing from the spirit and scope of the present invention as defined by the appended claims.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.