Treatment of Lung Diseases Using Pharmaceutical Agents that Eliminate Senescent Cells
Vlahakis; Nick ; et al.
U.S. patent application number 16/636299 was filed with the patent office on 2020-06-25 for treatment of lung diseases using pharmaceutical agents that eliminate senescent cells. The applicant listed for this patent is Unity Biotechnology, Inc. Buck Institute for Research on Aging. Invention is credited to Scott Armstrong, Anne-Marie Beausoleil, Jamie Dananberg, Nathaniel David, Ryan Hudson, Remi-Martin Laberge, Nick Vlahakis.
| Application Number | 20200199103 16/636299 |
| Document ID | / |
| Family ID | 68841874 |
| Filed Date | 2020-06-25 |




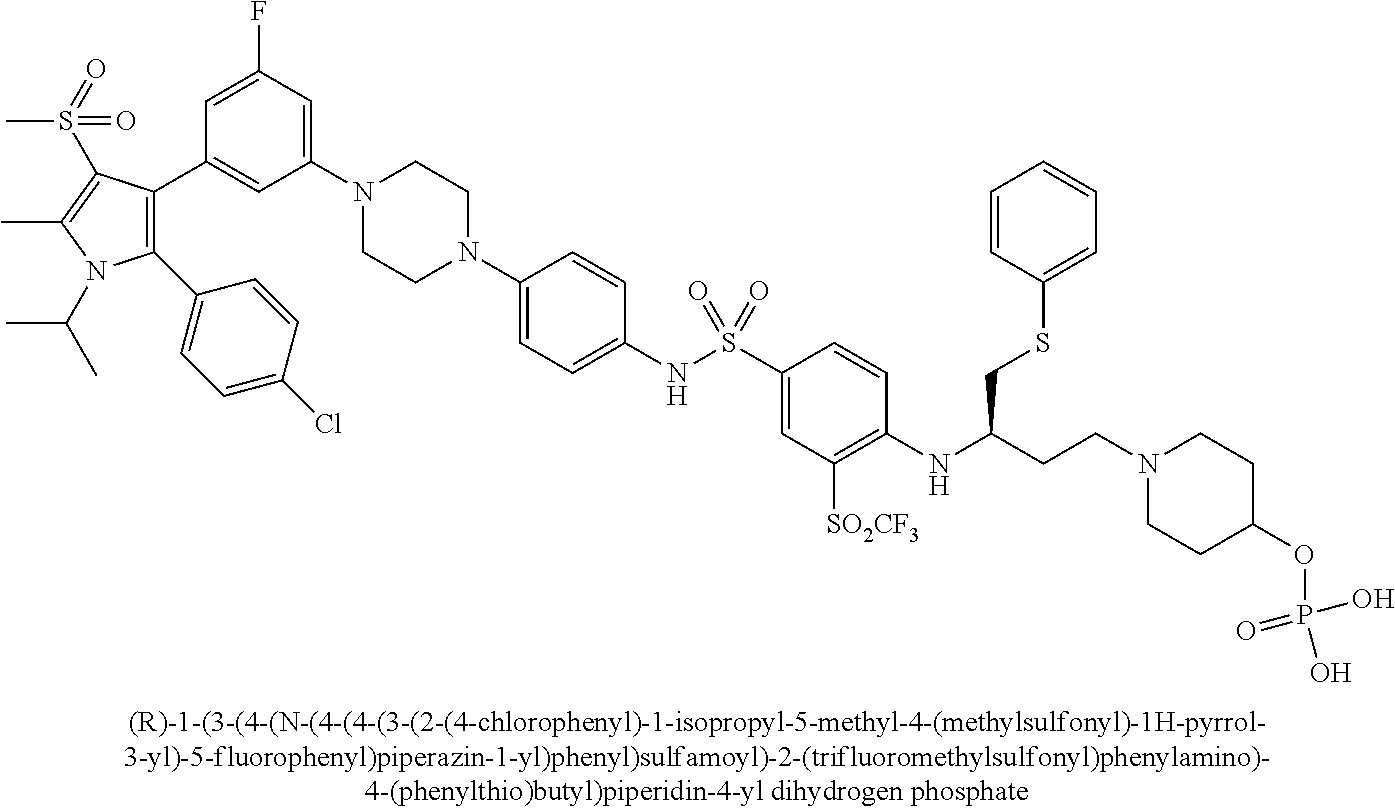
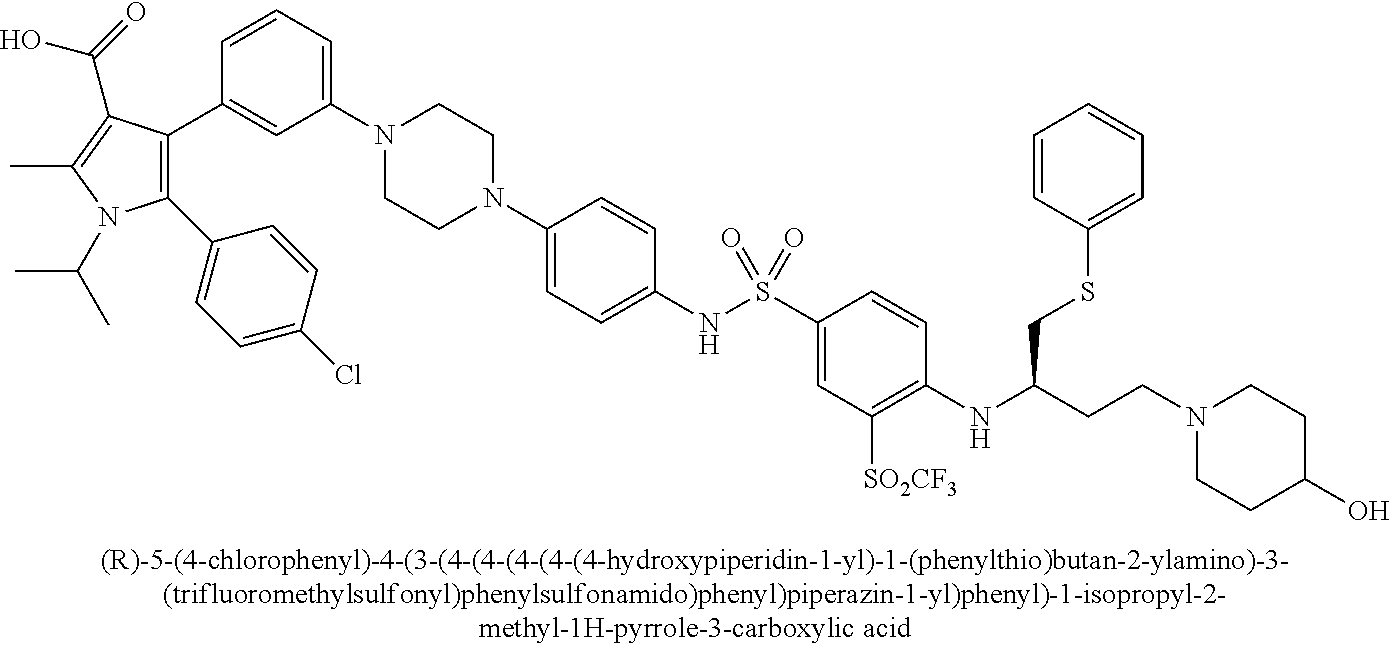
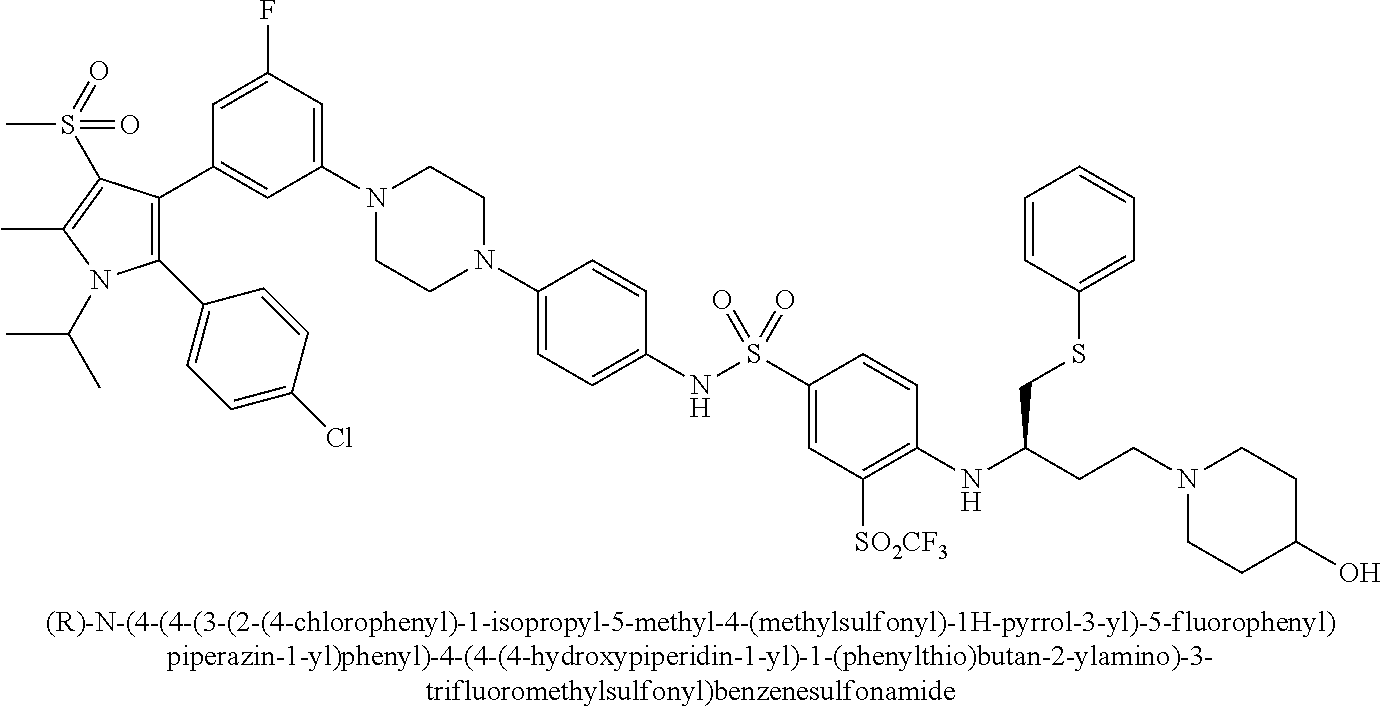
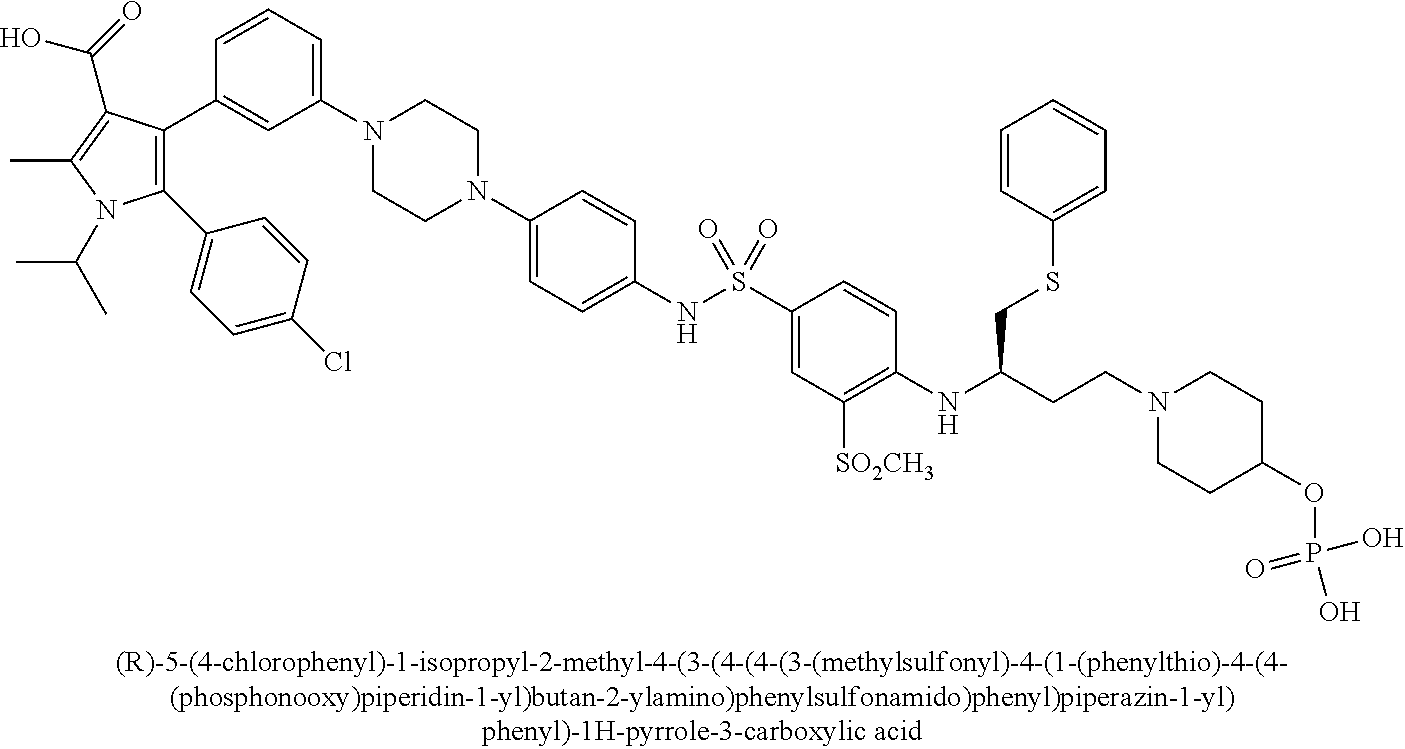
View All Diagrams
| United States Patent Application | 20200199103 |
| Kind Code | A1 |
| Vlahakis; Nick ; et al. | June 25, 2020 |
Treatment of Lung Diseases Using Pharmaceutical Agents that Eliminate Senescent Cells
Abstract
This invention is based on the discovery that many lung diseases associated with aging are mediated at least in part by cells bearing a senescent phenotype. Senescent cells accumulate with age, and express factors that contribute to the pathophysiology of age related conditions. The severity of age-related conditions typically correlates with the abundance of senescent cells: thus, clearing senescent cells can help abrogate the condition: providing symptomatic relief, and potentially inhibiting disease progression. In accordance with this invention, a family of Bcl protein inhibitors has been developed for the treatment of lung diseases. These senolytic agents have an appropriate dose and specificity profile to be effective in the clinical management of previously intractable pulmonary diseases.
| Inventors: | Vlahakis; Nick; (Brisbane, CA) ; Armstrong; Scott; (Brisbane, CA) ; Dananberg; Jamie; (Brisbane, CA) ; Hudson; Ryan; (Brisbane, CA) ; Beausoleil; Anne-Marie; (Brisbane, CA) ; David; Nathaniel; (Brisbane, CA) ; Laberge; Remi-Martin; (Brisbane, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 68841874 | ||||||||||
| Appl. No.: | 16/636299 | ||||||||||
| Filed: | August 13, 2018 | ||||||||||
| PCT Filed: | August 13, 2018 | ||||||||||
| PCT NO: | PCT/US2018/046567 | ||||||||||
| 371 Date: | February 3, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62684681 | Jun 13, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07D 207/34 20130101; C07F 9/65583 20130101; A61P 9/10 20180101; C07D 401/12 20130101; A61P 43/00 20180101; A61P 19/02 20180101; A61P 27/00 20180101; A61P 11/00 20180101 |
| International Class: | C07D 401/12 20060101 C07D401/12; A61P 11/00 20060101 A61P011/00 |
Claims
1. A compound according to Formula (II): ##STR00097## wherein: X.sup.1 is --Cl; X.sup.2 is --COOH or --SO.sub.2CH.sub.3; X.sup.3 is --SO.sub.2CF.sub.3, --SO.sub.2CH.sub.3, or --NO.sub.2; X.sup.5 is --F or --H; R.sup.1 is --CH(CH.sub.3).sub.2; R.sup.2 is --CH.sub.3; R.sup.3 and R.sup.4 are both --H; n is 2; R.sup.6 is selected from --OH, --OR.sup.7, ##STR00098## and R.sup.7 is --PO(OH).sub.2, or a salt or a stereoisomer thereof.
2. The compound of claim 1, wherein X.sup.2 is --COOH.
3. The compound of claim 1, wherein X.sup.2 is --SO.sub.2CH.sub.3.
4. The compound of claim 1, wherein X.sup.3 is --SO.sub.2CF.sub.3.
5. The compound of claim 1, wherein X.sup.3 is --SO.sub.2CH.sub.3.
6. The compound of claim 1, wherein X.sup.3 is --NO.sub.2.
7. The compound of any of claims 1 to 6, wherein X.sup.5 is --F.
8. The compound of any of claims 1 to 6, wherein X.sup.5 is --H.
9. The compound of any of claims 1 to 8, wherein X.sup.6 is --OH.
10. The compound of any of claims 1 to 8, wherein X.sup.6 is --OR.sup.7.
11. The compound of any of claims 1 to 8, wherein X.sup.6 is ##STR00099##
12. The compound of any of claims 1 to 8, wherein X.sup.6 is ##STR00100##
13. The compound of any of claims 1 to 8, wherein X.sup.6 is ##STR00101##
14. The compound of any of claims 1 to 13, wherein the carboxyl group in X.sup.2 is phosphorylated.
15. The compound of claim 1, wherein the compound is selected from the group consisting of: ##STR00102##
16. The compound of any preceding claim, which has pro-apoptotic activity.
17. The compound of any preceding claim, which specifically kills senescent cells compared with non-senescent cells, said senescent cells being defined as non-cancerous cells that express p16.
18. The compound of any preceding claim, which specifically kills cancer cells compared with non-cancer cells of the same tissue type.
19. The compound of any preceding claim, which has an IC.sub.50 for Bcl-xL of 1 nM or less.
20. The compound of any preceding claim, which has an IC.sub.50 for Bcl-2 of 10 nM or less.
21. The compound of any preceding claim, which has an IC.sub.50 for Bcl-xL of 1 nM or less and an IC.sub.50 for Bcl-2 of 10 nM or less.
22. A pharmaceutical composition comprising a compound according to any preceding claim in a pharmaceutically compatible excipient.
23. A method of selectively removing senescent cells and/or cancer cells from a mixed cell population or tissue, comprising contacting a cell, a cell population or a tissue with a compound according to any of claims 1 to 21 or a pharmaceutical composition according to claim 22.
24. A method of treating a senescence related condition in a tissue in a subject, wherein the senescence related condition is characterized as being caused or mediated at least in part by senescent cells, or is characterized as having an overabundance of senescent cells in or around the tissue, in comparison with unaffected tissue, the method comprising: administering to a tissue of a subject in need thereof, an amount of a compound according to any of claims 1 to 21 or a pharmaceutical composition according to claim 22 that is effective to selectively remove senescent cells from the tissue, thereby relieving or ameliorating one or more signs or symptoms of a senescence related condition in the subject.
25. A unit dose of a pharmaceutical composition comprising: an amount of a compound that inhibits Bcl function configured for use in the treatment of a senescence associated condition that is caused or mediated at least in part by senescent cells, wherein the compound is a compound according to any of claims 1 to 21, wherein the pharmaceutical composition contains a formulation of the compound configured for administration to a target tissue in a subject that manifests the senescence associated condition, and wherein the formulation and the amount of the compound in the unit dose configure the unit dose to be effective in selectively removing senescent cells in or around the tissue in the subject, thereby decreasing the severity of one or more signs or symptoms of the condition without causing adverse effects in the subject when administered to the tissue as a single dose.
26. The unit dose of claim 25, packaged with an informational insert describing the use and attendant benefits of the drugs in treating the senescent cell associated condition.
27. A compound according to any of claims 1 to 21 or a pharmaceutical composition according to claim 24 for use in selectively eliminating senescent cells from a tissue or mixed cell population or for use in treating a senescence-related condition.
28. Use of a compound according to any of claims 1 to 21 in the manufacture of a medicament for treating a senescence-related condition.
29. The method, unit dose, or use of any of claims 24 to 28, wherein the condition is osteoarthritis.
30. The method, unit dose, or use of any of claims 24 to 28, wherein the condition is an ophthalmic condition.
31. The method, unit dose, or use of any of claims 24 to 28, wherein the condition is a pulmonary disease.
32. A method of treating cancer, comprising administering to a tissue of a subject in need thereof an amount of a compound according to any of claims 1 to 21 or a pharmaceutical composition according to claim 22 effective to selectively remove cancer cells from the tissue.
33. A compound according to any of claims 1 to 21 or a pharmaceutical composition according to claim 22 for use in selectively eliminating cancer cells from a tissue or mixed cell population or for use in treating cancer.
34. A method of treating a pulmonary disease in a subject, comprising administering to the subject in need thereof a therapeutically effective amount of a pharmaceutical composition comprising: a compound of Formula (I): ##STR00103## wherein: X.sup.1 is --Cl; X.sup.2 is --COOH or --SO.sub.2CH.sub.3; X.sup.3 is --SO.sub.2CF.sub.3; --SO.sub.2CH.sub.3; or --NO.sub.2; X.sup.5 is --F or --H; R.sup.1 is --CH(CH.sub.3).sub.2; R.sup.2 is --CH.sub.3; R.sup.3 and R.sup.4 are both --H; n is 2; and R.sup.6 is selected from --OR.sup.7, ##STR00104## and R.sup.7 is --H or --PO(OH).sub.2, or a salt or a stereoisomer thereof; and a pharmaceutically compatible excipient.
35. The method of claim 34, wherein X.sup.2 is --COOH.
36. The method of claim 34, wherein X.sup.2 is --SO.sub.2CH.sub.3.
37. The method of claim 34, wherein X.sup.3 is --SO.sub.2CF.sub.3.
38. The method of claim 34, wherein X.sup.3 is --SO.sub.2CH.sub.3.
39. The method of claim 34, wherein X.sup.3 is --NO.sub.2.
40. The method of any of claims 34 to 39, wherein X.sup.5 is --F.
41. The method of any of claims 34 to 39, wherein X.sup.5 is --H.
42. The method of any of claims 34 to 39, wherein R.sup.6 is --OR.sup.7.
43. The method of any of claims 34 to 39, wherein R.sup.6 is ##STR00105##
44. The method of any of claims 34 to 39, wherein R.sup.6 is ##STR00106##
45. The method of any of claims 34 to 44, wherein R.sup.7 is --H.
46. The method of any of claims 34 to 44, wherein R.sup.7 is --PO(OH).sub.2.
47. The method of any of claims 34 to 46, wherein the carboxyl group in X.sup.2 is phosphorylated.
48. The method of claim 34, wherein the compound is selected from the group consisting of: ##STR00107## ##STR00108##
49. The method of claim 34, wherein the pulmonary disease is idiopathic pulmonary fibrosis (IPF).
50. The method of claim 34, wherein the pulmonary disease is chronic obstructive pulmonary disease (COPD).
51. The method of claim 48, wherein the pulmonary disease is idiopathic pulmonary fibrosis (IPF).
52. The method of claim 48, wherein the pulmonary disease is chronic obstructive pulmonary disease (COPD).
53. The method of claim 34, wherein the administration of the pharmaceutical composition is by inhalation as an aerosol.
54. The method of claim 48, wherein the administration of the pharmaceutical composition is by inhalation as an aerosol.
Description
RELATED APPLICATIONS
[0001] This application claims priority to U.S. patent application Ser. No. 15/675,171, filed Aug. 11, 2017, which is a continuation-in-part of U.S. patent application Ser. No. 15/611,589, filed Jun. 1, 2017, which is a continuation of International patent application no. PCT/US16/16894, filed Feb. 5, 2016, which claims priority to U.S. provisional patent application No. 62/113,227, filed Feb. 6, 2015. This application also claims priority to U.S. provisional patent application No. 62/684,681, filed Jun. 13, 2018. The disclosures of each of the above-referenced applications are incorporated herein by reference in their entireties.
FIELD OF THE INVENTION
[0002] The technology disclosed and claimed below relates generally to the field of lung disease leading to impaired respiration capacity. This disclosure provides a family of compounds and techniques that can be used for treating pulmonary disease by eliminating senescent cells implicated in the underlying pathophysiology and symptomatology.
BACKGROUND
[0003] Recent WHO data in 2015 show that respiratory diseases make up three of the top five causes of death worldwide (http://www.who.int/mediacentre/factsheets/fs310/en/). Chronic obstructive pulmonary disease (COPD) and lower respiratory infections are third and fourth on this list and both are diseases most prevalent in the elderly. Furthermore, the NHLBI published a white paper in 2017 highlighting the association of age with lung disease, including inflammatory pulmonary fibrosis (IPF) and COPD, and underscoring the potential for understanding and developing therapeutics related to ageing biology. Historically, therapies for these diseases have been non-specific in their mode of action, either anti-inflammatory (e.g. corticosteroids) or immunosuppressive (e.g., cyclophosphamide) or purely supportive in nature e.g., supplemental oxygen, anti-tussives and diuretics.
[0004] There is growing evidence of senescent cells playing a role in such non-oncologic pulmonary diseases, including data disclosed herein. Thus, the goal of the compounds of the invention is to not just interrupt specific pathogenic pathways but specifically target senescent cells and in turn inhibit multiple pathogenic pathways.
[0005] The invention provided herein provides novel compounds for the treatment of respiratory disease (primary or secondary etiology), and extra-pulmonary effects arising from or associated with such lung diseases, through the elimination of senescent cells implicated in the pathophysiology of diseases of the pulmonary system. The disclosure that follows outlines its implementation and use and describes many of the ensuing benefits.
SUMMARY
[0006] This invention is based in part on the discovery that many pulmonary diseases and conditions associated with aging are mediated at least in part by cells bearing a senescent phenotype. Senescent cells accumulate with age, which is why conditions mediated by senescent cells occur more frequently in older adults. Senescent cells express factors that contribute to the pathophysiology of the age related and senescence-associated conditions. Different types of stress on pulmonary tissues may promote the emergence of senescent cells and the phenotype they express. Cell stressors include oxidative stress, metabolic stress, DNA damage (for example, because of environmental ultraviolet light exposure or genetic cause), oncogene activation, and telomere shortening (resulting, for example, from hyperproliferation).
[0007] This invention is also based in part on new acyl sulfonamides that are Bcl inhibitors. Some of the Bcl inhibitors in this family are particularly effective senolytic agents for lung diseases. Contacting senescent cells in vitro or in vivo with the compounds and compositions of the invention selectively modulates or eliminates such cells. These inhibitors can be used for administration to a diseased lung tissue in a subject having an age-related lung disease, thereby selectively eliminating senescent cells in or around the diseased lung tissue and relieving one or more symptoms or signs of the disease. Selected compounds from the family can be formulated and marketed as chemotherapeutic agents.
BRIEF DESCRIPTION OF THE DRAWINGS
[0008] FIG. 1 shows a general synthetic scheme for chemically synthesizing exemplary compounds according to this invention.
[0009] FIGS. 2A and 2B show immunohistochemical staining for p16 in human IPF lung tissue to demonstrate the presence of senescent cells. FIG. 2A shows a macro-view of the IPF lung tissue having p16 positive staining FIG. 2B is an enlarged view of an area of the IPF lung tissue of FIG. 2A. The senescent cells were predominately epithelial in origin and located in areas of fibrosis and at the leading edge of the disease. See Example 1.
[0010] FIG. 3 shows the quantification of p16 positive cells in all normal human lung tissues sampled as compared to all human IPF lung tissues sampled. Increased presence of p16 positive cells in human lung tissue with significant fibrotic area was indicative of a significant role in disease progression (****p<0.0001 for group difference among means by one-way ANOVA). See Example 1.
[0011] FIGS. 4A, 4B and 4C show immunohistochemical staining for p16 in human scleroderma lung tissue to demonstrate the presence of senescent cells. FIG. 4A shows a macro-view of the human scleroderma lung tissue having p16 positive staining. FIG. 4B is an enlarged view of an area of the scleroderma lung tissue of FIG. 4A. FIG. 4C is a further enlarged view of an area of the scleroderma lung tissue of FIG. 4B. The p16 positive senescent cells were fibrotic in origin and located in honeycomb areas of the lung. See Example 1.
[0012] FIG. 5 shows the quantification of p16 positive cells in normal human lung tissue as compared to human scleroderma lung tissue. Increased presence of p16 positive cells in human lung tissue with significant fibrotic area was indicative of a significant role in disease progression (****p<0.0001 for group difference among means by one-way ANOVA). See Example 1.
[0013] FIG. 6 shows a concentration-response curve demonstrating the selectivity of Compound 1 for senescent lung epithelial cells (SnC--solid lines) in contrast to non-senescent lung epithelial cells (NsC--dashed lines). See Example 2.
[0014] FIG. 7 shows a concentration-response curve, using one-way ANOVA, of relative p16 gene expression changes of 8%, 14% and 27% upon treatment with 0.1 mg/ml, 0.3 mg/ml and 1.0 mg/ml of Compound 1, respectively, in mice challenged with bleomycin (+) to induce senescence in the lung. See Example 4.
[0015] FIG. 8 shows the ability of Compound 1 to eliminate p16 positive lung epithelial cells in mice challenged with bleomycin (+) to induce senescence in the lung. See Example 4.
DETAILED DESCRIPTION
Definitions
[0016] The terms "enantiomerically enriched" and "stereoisomerically enriched" denote that the compound of the invention comprises 75%, 80%, 85%, 90%, 95%, 98%, or 99% or more by weight of the enantiomer or the stereoisomer.
[0017] The term "ex vivo" refers to experimentation or manipulation done in or on living tissue in an artificial environment outside the organism.
[0018] The term "pharmaceutically acceptable carrier" or "pharmaceutically acceptable excipient" includes any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents and the like. The use of such media and agents for pharmaceutically active substances is well known in the art. Except insofar as any conventional media or agent is incompatible with the active ingredient, its use in the therapeutic compositions is contemplated. Supplementary active ingredients can also be incorporated into the compositions.
[0019] The term "pharmaceutically acceptable salt" refers to salts that retain the biological effectiveness and properties of the compounds of this invention and, which are not biologically or otherwise undesirable. In many cases, the compounds of this invention are capable of forming acid and/or base salts by virtue of the presence of amino, phosphate, and/or carboxyl groups or groups similar thereto. Pharmaceutically acceptable acid addition salts can be formed with inorganic acids and organic acids. Inorganic acids from which salts can be derived include, for example, hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like. Organic acids from which salts can be derived include, for example, acetic acid, propionic acid, naphtoic acid, oleic acid, palmitic acid, pamoic (emboic) acid, stearic acid, glycolic acid, pyruvic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, ascorbic acid, glucoheptonic acid, glucuronic acid, lactic acid, lactobioic acid, tartaric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, p-toluenesulfonic acid, salicylic acid, and the like. Pharmaceutically acceptable base addition salts can be formed with inorganic and organic bases. Inorganic bases from which salts can be derived include, for example, sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminum, and the like; particularly preferred are the ammonium, potassium, sodium, calcium and magnesium salts. Organic bases from which salts can be derived include, for example, primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, basic ion exchange resins, and the like, specifically such as isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, histidine, arginine, lysine, benethamine, N-methyl-glucamine, and ethanolamine Other acids include dodecylsufuric acid, naphthalene-1,5-disulfonic acid, naphthalene-2-sulfonic acid, and saccharin.
[0020] A "phosphorylated" form of a compound is a compound in which one or more --OH or --COOH groups have been substituted with a phosphate group which is either --OPO(OH).sub.2 or -alkyl-OPO(OH).sub.2 (where alkyl is C.sub.1-6 alkyl), such that the phosphate group may be removed in vivo (for example, by enzymolysis). A non-phosphorylated or dephosphorylated form has no such phosphate group.
[0021] "Prodrug" refers to a derivative of an active agent that requires a transformation within the body to release the active agent. The transformation can be an enzymatic transformation. Prodrugs are frequently, although not necessarily, pharmacologically inactive until converted to the active agent.
[0022] A "senescent cell" is generally thought to be derived from a cell type that typically replicates, but as a result of aging or other event that causes a change in cell state, can no longer replicate. It remains metabolically active and commonly adopts a senescence associated secretory phenotype (SASP) that includes chemokines, cytokines and extracellular matrix and fibrosis modifying proteins and enzymes. The nucleus of senescent cells is often characterized by senescence-associated heterochromatin foci and DNA segments with chromatin alterations reinforcing senescence. Without implying any limitation on the practice of what is claimed in this disclosure that is not explicitly stated or required, the invention is premised on the hypothesis that senescent cells cause or mediate certain conditions associated with tissue damage or aging. For the purpose of practicing aspects of this invention, senescent cells can be identified as expressing at least one marker selected from p16, senescence-associated .beta.-galactosidase, and lipofuscin; sometimes two or more of these markers, and other markers of SASP such as but not limited to, interleukin 6 (IL-6), and inflammatory, angiogenic and extracellular matrix modifying proteins.
[0023] A "senescence associated", "senescence related" or "age related" disease, disorder, or condition is a physiological condition that presents with one or more symptoms or signs, wherein a subject having the condition needs or would benefit from a lessening of such symptoms or signs. The condition is senescence associated if it is caused or mediated in part by senescent cells, which may be induced by multiple etiologic factors including age, DNA damage, oxidative stress, genetic defects, etc. Lists of senescence associated disorders that can potentially be treated or managed using the methods and products taught in this disclosure include those discussed in this disclosure and the previous disclosures to which this application claims priority.
[0024] A compound of the invention is typically referred to as "senolytic" if it eliminates senescent cells, compared with replicative cells of the same tissue type, or quiescent cells lacking SASP markers. Alternatively, or in addition, compounds of the invention may effectively be used according to this invention if it decreases the release of pathological soluble factors or mediators as part of the senescence associated secretory phenotype that play a role in the initial presentation or ongoing pathology of a condition or inhibit its resolution. In this respect, the term "senolytic" is exemplary, with the understanding that compounds that work primarily by inhibiting rather than eliminating senescent cells (senescent cell inhibitors) can be used in a similar fashion with ensuing benefits.
[0025] "Small molecule" Bcl inhibitors according to this invention have molecular weights less than 20,000 daltons, and are often less than 10,000, 5,000, or 2,000 daltons. Small molecule inhibitors are not antibody molecules or oligonucleotides, and typically have no more than five hydrogen bond donors (the total number of nitrogen-hydrogen and oxygen-hydrogen bonds), and no more than 10 hydrogen bonds.
[0026] Successful "treatment" of a lung disease according to this invention may have any effect that is beneficial to the subject being treated. This includes decreasing severity, duration, or progression of a condition, or of any adverse signs or symptoms resulting therefrom. In some circumstances, senolytic agents can also be used to prevent or inhibit presentation of a condition for which a subject is susceptible, for example, because of an inherited susceptibility of because of medical history.
[0027] A "therapeutically effective amount" is an amount of a compound of the present disclosure that (i) treats the particular disease, condition, or disorder, (ii) attenuates, ameliorates, or eliminates one or more symptoms of the particular disease, condition, or disorder, (iii) prevents or delays the onset of one or more symptoms of the particular disease, condition, or disorder described herein, (iv) prevents or delays progression of the particular disease, condition or disorder, or (v) at least partially reverses damage caused by the condition prior to treatment.
[0028] Unless otherwise stated or required, all the compound structures referred to in the invention include conjugate acids and bases having the same structure, crystalline and amorphous forms of those compounds, pharmaceutically acceptable salts, and dissolved and solid forms thereof, including, for example, polymorphs, solvates, hydrates, unsolvated polymorphs (including anhydrates), conformational polymorphs, and amorphous forms of the compounds, as well as mixtures thereof. Except where otherwise stated or required, other terms used in the specification have their ordinary meaning.
[0029] The half maximal inhibitory concentration (IC.sub.50) is a measure of the potency of a compound in inhibiting a specific biological or biochemical function. Specifically, for compounds of the invention, IC.sub.50 is the measure of the amount of a compound required to achieve 50% inhibition of the activity of the target Bcl. For example, compounds of the invention have a demonstrated IC.sub.50 for Bcl-xL of less than 10 nM, less than 5 nM, or less than 1 nM. Compounds of the invention have a demonstrated IC.sub.50 for Bcl-xL of between 1 nM to 10 nM, between 1 nM and 5 nM, between 5 nM to 10 nM, or between 0.1 nM to 1 nM. Compounds of the invention have also demonstrated an IC.sub.50 for Bcl-2 of less than 15 nM, less than 10 nM, less than 5 nM, or less than 1 nM. Compounds of the invention have also demonstrated an IC.sub.50 for Bcl-2 of between 1 nM to 10 nM, between 1 nM and 5 nM, between 5 nM to 10 nM, or between 0.1 nM to 1 nM.
[0030] "Alkyl" refers to monovalent saturated aliphatic hydrocarbyl groups having from 1 to 10 carbon atoms and preferably 1 to 6 carbon atoms. This term includes, by way of example, linear and branched hydrocarbyl groups such as methyl (--CH.sub.3), ethyl (--CH.sub.2CH.sub.3, n-propyl (--CH.sub.2CH.sub.2CH.sub.3), isopropyl (--CH(CH.sub.3).sub.2), n-butyl (--CH.sub.2CH.sub.2CH.sub.2CH.sub.3), isobutyl (--CH.sub.2CH(CH.sub.3).sub.2), sec-butyl (--CH(CH.sub.2CH.sub.3)(CH).sub.3), t-butyl (--C(CH.sub.3).sub.3), n-pentyl (--CH.sub.2CH.sub.2CH.sub.2CH.sub.2CH.sub.3), and neopentyl (--CH.sub.2C(CH.sub.3).sub.3), etc. In certain embodiments, an "alkyl" group can be substituted, where the term "substituted," when used to modify a specified group or radical, means that one or more hydrogen atoms of the specified group or radical are each independently replaced with the same or different substituent groups as defined below.
[0031] Substituent groups for substituting for one or more hydrogens (any two hydrogens on a single carbon can be replaced with .dbd.O, .dbd.NR.sup.50, .dbd.N--OR.sup.50, .dbd.N.sub.2 or .dbd.S) on carbon atoms in the specified group or radical are, unless otherwise specified, --R.sup.60, halo, .dbd.O, --OR.sup.50, --SR.sup.50, --NR.sup.50R.sup.50, trihalomethyl, --CN, --OCN, --SCN, --NO, --NO.sub.2, .dbd.N.sub.2, --N.sub.3, --SO.sub.2R.sup.50, --SO.sub.2OR.sup.50, --OSO.sub.2R.sup.50, --OSO.sub.2OR.sup.50, --P(O)(OH).sub.2, --P(O)(OR.sup.50)OH, --P(O)(OR.sup.50).sub.2, --C(O)R.sup.50, --C(S)R.sup.50)--C(NR.sup.50)R.sup.50, --C(O)OR.sup.50, --C(S)OR.sup.50, --C(O)NR.sup.50R.sup.50, --C(NR.sup.50)NR.sup.50R.sup.50, --OC(O)R.sup.50, --OC(S)R.sup.50, --OC(O)OR.sup.50, --OC(S)OR.sup.50, --NR.sup.50C(O)R.sup.50, --NR.sup.50 C(S)R.sup.50, --NR.sup.50CO.sub.2R.sup.50, --NR.sup.50C(S)OR.sup.50, --N R.sup.50C(O)NR.sup.50R.sup.50, --NR.sup.50C(NR.sup.50)R.sup.50 and --NR.sup.50C(NR.sup.50)NR.sup.50R.sup.50, where R.sup.60 is selected from optionally substituted alkyl, cycloalkyl, heteroalkyl, heterocycloalkylalkyl, cycloalkylalkyl, aryl, arylalkyl, heteroaryl and heteroarylalkyl, and each R.sup.50 is independently hydrogen or R.sup.60.
[0032] "Heteroalkyl" refers to a saturated or unsaturated group having a single ring or multiple condensed rings, including fused bridged and spiro ring systems, and having from 3 to 20 ring atoms, including 1 to 10 hetero atoms. These ring atoms are selected from nitrogen, sulfur, or oxygen, where, in fused ring systems, one or more of the rings can be cycloalkyl, aryl, or heteroaryl. In certain embodiments, a "heteroalkyl" group can be substituted, as defined above.
DESCRIPTION OF THE INVENTION
[0033] It is a premise of this disclosure that many or most lung diseases that are age-related or are characterized by a pathogenic senescent biology that may be present at any age, are caused or mediated at least in part by senescent cells, which accumulate with age or in a disease-specific manner, and with deleterious impact on respiratory system pulmonary tissues. Senescent cells are typically cells that no longer have replicative capacity, but remain in the tissue of origin, eliciting a senescence-associated secretory phenotype (SASP). Senescent cells are thought to derive from proliferative cells of a variety of tissue types, including cells that reside in and around the lung. SASP factors include molecules that are angiogenic, inflammatory, proliferative, fibrotic, and extracellular matrix modifying molecules (Acosta et al., 2013). Some factors implicated in pulmonary pathologies are part of the constellation of factors produced by senescent cells. For this reason, elimination or control of senescent cells provides a means by which to treat lung disease, not only through the elimination of senescent cells but also through reduction of their associated SASP factors and impact on surrounding cells.
[0034] Different lung diseases present in the clinic with different signs and symptoms and have different types of pathophysiologic mechanisms. The heterogeneity of lung disease is consistent with the role of senescent cells in the disease pathology because senescent cells may be from different cell lineages, induced by different stressors, reside in different pulmonary tissues, and interact with surrounding cells in a different fashion. Nevertheless, senescent cells in the various tissues of the lung have a related secretory phenotype that contributes to disorders throughout the respiratory system.
[0035] The role of senescent cells in promoting or mediating the spectrum of lung diseases provides an approach to treatment with a number of advantages for the managing clinician. Since senescent cells are non-proliferative, eliminating senescent cells has the potential for a clinically beneficial effect that persists for an extended time between episodes of treatment. Features of the condition mediated by senescent cells can resolve at least until senescent cells re-accumulate. Since senescent cells and the burden of their pathogenic effect are likely to accumulate slowly, as the nature of age related diseases is to evolve over a period of many years, the effects of a single treatment or treatment cycle may last for weeks, months, or years.
[0036] The specific clearance of senescent cells from tissue is referred to in this disclosure as senolysis. Small molecule compounds capable of senolysis are referred to as senolytic agents, and clear senescent cells irrespective of mechanism of senescence induction, SASP profile or cell lineage. To the extent that senescent cells exacerbate the underlying extracellular pathogenic mechanisms, the long-lasting effect of senolysis provides a window in which such pathology is held at bay, potentially giving the tissue a chance for repair. This means that senescent cell medicine has the potential not just to halt progression of pulmonary diseases and conditions but allow some degree of reversal of the disease and its symptoms for the benefit of the patient.
[0037] Since senescent cells in different parts of the lung respond to the same senolytic agents, several different lung diseases can be treated in the same patient at the same time. For example, a patient may present to the clinician with several concurrent active disease processes already under way: such as fibrosis and chronic obstructive lung disease. It may be possible to administer a single senolytic agent in a treatment protocol that addresses the disease and its symptoms of each of the multiple conditions. Beyond the convenience of this approach, it has the added benefit of lowering the risk of side effects that may result from multiple drugs being given in combination to treat each of the conditions individually. Furthermore, it is possible that factors elicited by cells in one part of the lung may impact other parts of the lung such that treating senescence in two locations in the lung may have a beneficial effect on both lung diseases.
[0038] Senolytic medicines can be an important adjunct to other types of therapies, such as for example, standard of care, to relieve the symptoms that result from the condition(s). The two modes of therapy can work synergistically to reduce the burden, frequency and side effects of either mode administered separately.
[0039] Bcl Inhibitors
[0040] The technology described and claimed below represents the first description of a new class of Bcl inhibitors that can be used to selectively eliminate senescent cells from a target tissue for purposes of treatment of age-related conditions.
[0041] The Bcl protein family (TC#1.A.21) includes evolutionarily-conserved proteins that share Bcl-2 homology (BH) domains. Bcl proteins are most notable for their ability to up- or down-regulate apoptosis, a form of programmed cell death, at the mitochondrion. The following explanation is provided to assist the user in understanding some of the scientific underpinnings of the compounds of this invention. These concepts are not needed to practice the invention, nor do they limit the use of the compounds and methods described here in any manner beyond that which is expressly stated or required. In the context of this invention, the Bcl proteins of particular interest are those that downregulate apoptosis. Anti-apoptotic Bcl proteins contain BH1 and BH2 domains, some of them contain an additional N-terminal BH4 domain (Bcl-2, Bcl-x(L) and Bcl-w (Bcl-2L2), Inhibiting these proteins increases the rate or susceptibility of cells to apoptosis. Thus, an inhibitor of such proteins can be used to help eliminate cells in which the proteins are expressed.
[0042] In the mid-2000s, Abbott Laboratories developed a novel inhibitor of Bcl-2, Bcl-xL and Bch w, known as ABT-737 (Navitoclax). This compound is part of a group of BH3 mimetic small molecule inhibitors (SMI) that target these Bcl-2 family proteins, but not Al or Mcl-1. ABT-737 is superior to previous BCL-2 inhibitors given its higher affinity for Bcl-2, Bcl-xL and Bcl-w. In vitro studies showed that primary cells from patients with B-cell malignancies are sensitive to ABT-737. In human patients, ABT-737 is effective against some types of cancer cells but is subject to dose-limiting thrombocytopenia.
[0043] U.S. Application Publication No. 2016/0339019 (Laberge et al.) describes treatment of certain age-related conditions using MDM2 inhibitors, Bcl inhibitors, and Akt inhibitors. U.S. Application Publication No. 2017/0266211 (David et al.) describes the use of particular Bcl inhibitors for treatment of age-related conditions. U.S. Pat. Nos. 8,691,184, 9,096,625, and 9,403,856 (Wang et al.) describe Bcl inhibitors in a small-molecule library. It has now been discovered that the compounds described here fit into the active site of Bcl protein to provide strong Bcl inhibition and/or promote apoptosis of target cells. These compounds can be developed as highly potent and specific drugs to target senescent cells and cancer cells, as described in the sections that follow.
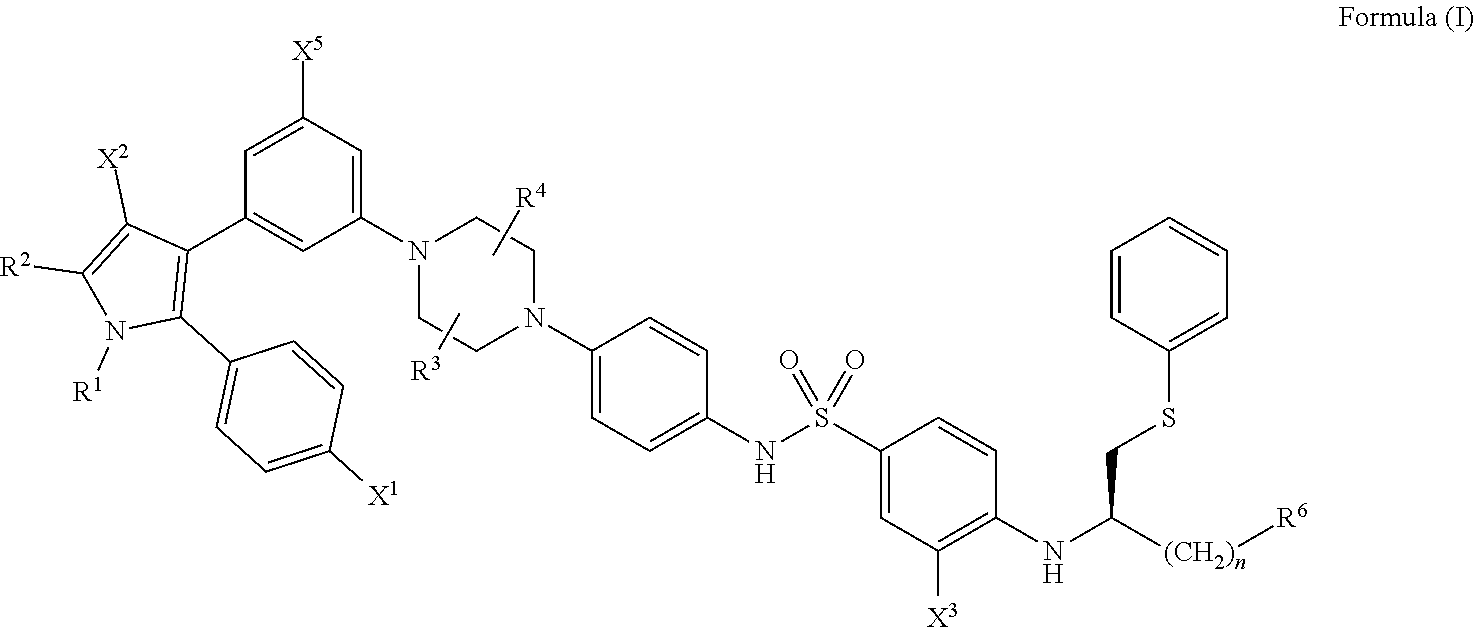
[0044] Generally, the compounds of the present disclosure have a structure that falls within the scope of the structure according to Formula (I) shown below.
##STR00001##
wherein: [0045] X.sup.1 is --Cl; [0046] X.sup.2 is --COOH or --SO.sub.2CH.sub.3; [0047] X.sup.3 is --SO.sub.2CF.sub.3; --SO.sub.2CH.sub.3; or --NO.sub.2; [0048] X.sup.5 is --F or --H; [0049] R.sup.1 is .sup.--CH(CH.sub.3).sub.2; [0050] R.sup.2 is --CH.sub.3; [0051] R.sup.3 and R.sup.4 are both --H; [0052] n is 2;
##STR00002##
[0052] and [0053] R.sup.6 is selected from --OR.sup.7, and [0054] R.sup.7 is --H or --PO(OH).sub.2, [0055] or a salt or a stereoisomer thereof; and [0056] a pharmaceutically compatible excipient.
[0057] As described above, in compounds of Formula (I), X.sup.1 is --Cl, R.sup.1 is --CH(CH.sub.3).sub.2, R.sup.2 is --CH.sub.3, R.sup.3 is H, R.sup.4 is --H, and n is 2.
[0058] In certain embodiments,X.sup.2 is --COOH or --SO.sub.2CH.sub.3. For example, X.sup.2 can be --COOH, or X.sup.2 can be --SO.sub.2CH.sub.3. As used herein, the dash symbol ("-") indicates the point of attachment of the moiety of interest to the remainder of the compound being described.
[0059] In certain embodiments, X.sup.3 is --SO.sub.2CF.sub.3, --SO.sub.2CH.sub.3, or --NO.sub.2. For example, X.sup.3 can be --SO.sub.2CF.sub.3, or X.sup.3 can be --SO.sub.2CH.sub.3, or X.sup.3 can be --NO.sub.2.
[0060] In certain embodiments, X.sup.5 is --F or --H. For example, X.sup.5 can be --F, or X.sup.5 can be --H.
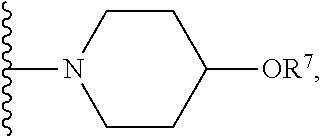
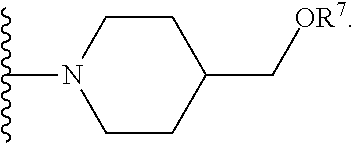
[0061] In certain embodiments, R.sup.6 is selected from --OR.sup.7,
##STR00003##
For example, R.sup.6 can be
##STR00004##
or R.sup.6 can be
##STR00005##
[0062] or R.sup.6 can be
[0063] As used herein, a wavy line ("") indicates the point of attachment or the bond where the moiety of interest is attached to the remainder of the compound being described.
[0064] In certain embodiments, R.sup.7 is --H or --PO(OH).sub.2. For example, R.sup.7 can be --H, or R.sup.7 can be --PO(OH).sub.2.
[0065] Any of the various combinations of the X.sup.2, X.sup.3, X.sup.5, R.sup.6, and R.sup.7 substituents are possible for compounds of Formula (I).
[0066] In combination with any of the aforelisted options, the --COOH group of X.sup.2 may be phosphorylated as well as or instead of the hydroxyl group at the R.sup.6 position, at the user's option.
[0067] Examples of the compounds of Formula (I) are shown in TABLE 1.
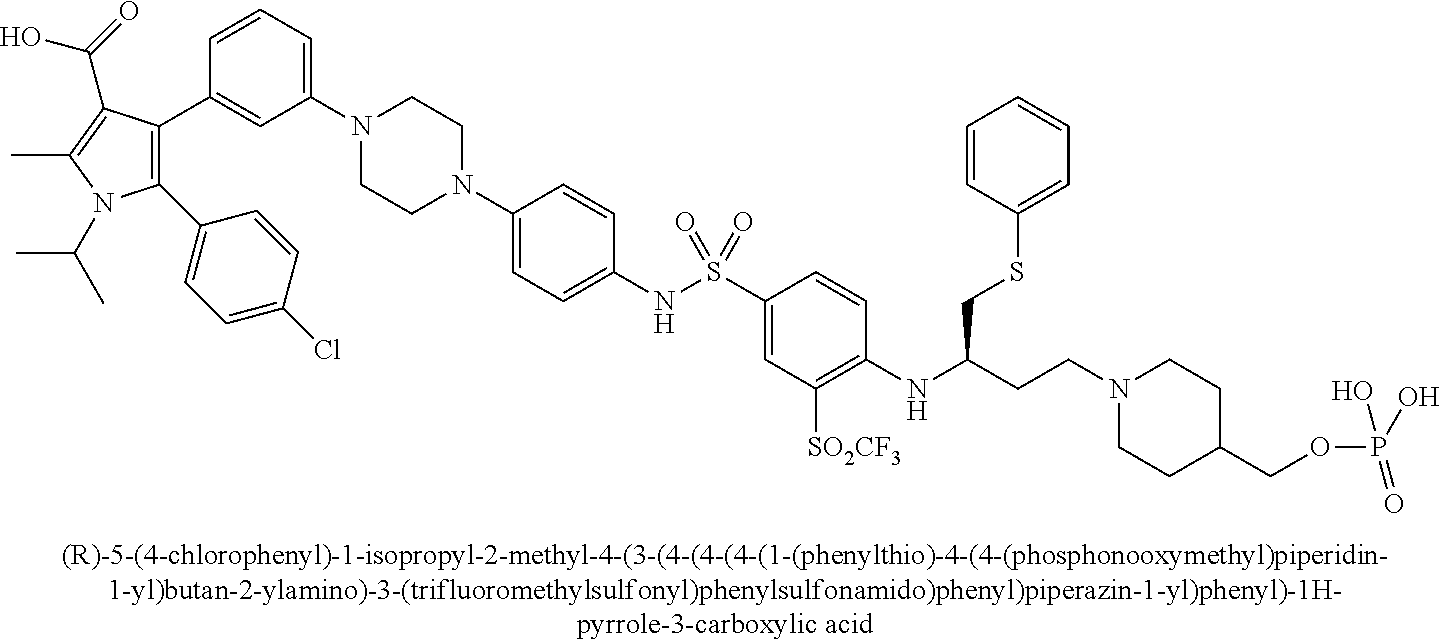
TABLE-US-00001 TABLE 1 Com- pound No. Compound Structure and Name 1 ##STR00006## 2 ##STR00007## 3 ##STR00008## 4 ##STR00009## 5 ##STR00010## 6 ##STR00011## 7 ##STR00012## 8 ##STR00013## 9 ##STR00014## 10 ##STR00015## 11 ##STR00016## 12 ##STR00017## 13 ##STR00018## 14 ##STR00019## 15 ##STR00020## 16 ##STR00021## 17 ##STR00022## 18 ##STR00023## 19 ##STR00024## 20 ##STR00025## 21 ##STR00026## 22 ##STR00027## 23 ##STR00028## 24 ##STR00029## 25 ##STR00030## 26 ##STR00031## 27 ##STR00032## 28 ##STR00033## 29 ##STR00034## 30 ##STR00035## 31 ##STR00036## 32 ##STR00037## 33 ##STR00038## 34 ##STR00039## 35 ##STR00040## 36 ##STR00041##
[0068] Compounds of Formula (I) find use in methods of treating a pulmonary disease in a subject as described herein. For example, the pulmonary disease can be idiopathic pulmonary fibrosis (IPF), or the pulmonary disease can be chronic obstructive pulmonary disease (COPD).
[0069] In certain embodiments, the compounds of the present disclosure have a structure that falls within the scope of the structure according to Formula (II) shown below.
##STR00042##
wherein: [0070] X.sup.1 is --Cl; [0071] X.sup.2 is --COOH or --SO.sub.2CH.sub.3; [0072] X.sup.3 is --SO.sub.2CF.sub.3, --SO.sub.2CH.sub.3, or --NO.sub.2; [0073] X.sup.5 is --F or --H; [0074] R.sup.1 is --CH(CH.sub.3).sub.2; [0075] R.sup.2 is --CH.sub.3; [0076] R.sup.3 and R.sup.4 are both --H; [0077] n is 2; [0078] R.sup.6 is selected from --OH, --OR.sup.7,
##STR00043##
[0078] and [0079] R.sup.7 is --PO(OH).sub.2, [0080] or a salt or a stereoisomer thereof.
[0081] As described above, in compounds of Formula (II), X.sup.1 is --Cl, R.sup.1 is --CH(CH.sub.3).sub.2, R.sup.2 is --CH.sub.3, R.sup.3 is H, R.sup.4 is --H, n is 2, and R.sup.7 is --PO(OH).sub.2.
[0082] In certain embodiments, X.sup.2 is --COOH or --SO.sub.2CH.sub.3. For example, X.sup.2 can be --COOH, or X.sup.2 can be --SO.sub.2CH.sub.3.
[0083] In certain embodiments, X.sup.3 is --SO.sub.2CF.sub.3, --SO.sub.2CH.sub.3, or --NO.sub.2. For example, X.sup.3 can be --SO.sub.2CF.sub.3, or X.sup.3 can be --SO.sub.2CH.sub.3, or X.sup.3 can be --NO.sub.2.
[0084] In certain embodiments, X.sup.5 is --F or --H. For example, X.sup.5 can be --F, or X.sup.5 can be --H.
[0085] In certain embodiments, R.sup.6 is selected from --OH, --OR.sup.7,
##STR00044##
For example, R.sup.6 can be --OH, or R.sup.6 can be --, or R.sup.6 can be
##STR00045##
or R.sup.6 can be
##STR00046##
[0086] or R.sup.6 can be
##STR00047##
[0088] Any of the various combinations of the X.sup.2, X.sup.3, X.sup.5, and R.sup.6 substituents are possible for compounds of Formula (II).
[0089] In combination with any of the aforelisted options, the --COOH group of X.sup.2 may be phosphorylated as well as or instead of the hydroxyl group at the R.sup.6 position, at the user's option.
[0090] Examples of the compounds of Formula (II) are shown in TABLE 2.
TABLE-US-00002 TABLE 2 Com- pound No. Compound Structure and Name 1 ##STR00048## 2 ##STR00049## 3 ##STR00050## 4 ##STR00051## 5 ##STR00052## 6 ##STR00053## 7 ##STR00054## 8 ##STR00055## 9 ##STR00056## 10 ##STR00057## 11 ##STR00058## 12 ##STR00059## 13 ##STR00060## 14 ##STR00061## 15 ##STR00062## 16 ##STR00063## 17 ##STR00064## 18 ##STR00065## 19 ##STR00066## 20 ##STR00067## 21 ##STR00068## 26 ##STR00069## 27 ##STR00070## 28 ##STR00071## 29 ##STR00072## 30 ##STR00073## 31 ##STR00074## 32 ##STR00075## 33 ##STR00076## 34 ##STR00077## 35 ##STR00078## 36 ##STR00079##
[0091] In certain embodiments, the compounds of the present disclosure have a structure that falls within the scope of the structure according to Formula (III) shown below.
##STR00080##
wherein: [0092] R.sup.1 and R.sup.2 are independently C.sub.1 to C.sub.4 alkyl; [0093] R.sup.3, R.sup.4 and R.sup.5 are independently --H or --CH.sub.3; [0094] R.sup.8 is --OH or --N(R.sup.6)(R.sup.7), wherein R.sup.6 and R.sup.7 are independently alkyl or heteroalkyl, and are optionally cyclized; [0095] X.sup.1 is --F, --Cl, --Br, or --OCH.sub.3; [0096] X.sup.2 is --SO.sub.2R' or --CO.sub.2R', where R' is --H, --CH.sub.3, or --CH.sub.2CH.sub.3; [0097] X.sup.3 is --SO.sub.2CF.sub.3; --SO.sub.2CH.sub.3; or --NO.sub.2; and [0098] X.sup.5 is --F, --Br, --Cl, --H, or --OCH.sub.3.
[0099] In certain embodiments, R.sup.1 and R.sup.2 are independently C.sub.1 to C.sub.4 alkyl. For example, R.sup.1 can be C.sub.1 to C.sub.4 alkyl, and R.sup.2 can be C.sub.1 to C.sub.4 alkyl.
[0100] In certain embodiments, R.sup.3, R.sup.4 and R.sup.5 are independently --H or --CH.sub.3. For example, R.sup.3 can be --H or --CH.sub.3. In some instances, R.sup.4 is --H or --CH.sub.3. In some instances, R.sup.5 is --H or --CH.sub.3.
[0101] In certain embodiments, R.sup.8 is --OH or --N(R.sup.6)(R.sup.7), where R.sup.6 and R.sup.7 are independently alkyl or heteroalkyl, and are optionally cyclized. For example, R.sup.8 can be --OH. In some instances, R.sup.8 is --N(R.sup.6)(R.sup.7). In these instances, R.sup.6 can be alkyl or heteroalkyl, and R.sup.7 can be alkyl or heteroalkyl. In some instances, R.sup.6 and R.sup.7 together with the nitrogen to which they are attached are cyclized. For example, in embodiments where R.sup.6 and R.sup.7 together with the nitrogen to which they are attached are cyclized, the resulting R.sup.8 group can be a heterocyclyl. For instance, in embodiments where R.sup.6 and R.sup.7 together with the nitrogen to which they are attached are cyclized, the resulting R.sup.8 group can be
##STR00081##
where m is an integer selected from 1, 2, and 3, and X.sup.4 is --OH, --COOH, or --CH.sub.2OH.
[0102] In some instances, the R.sup.8 heterocyclyl is selected from a group such as, but not limited to, pyrrolidinyl, pyrazolidinyl, imidazolidinyl, piperidinyl, piperazinyl, morpholinyl, and the like. In some cases, where R.sup.6 and R.sup.7 together with the nitrogen to which they are attached are cyclized, the resulting R.sup.8 group is piperidinyl. In some cases, where R.sup.6 and R.sup.7 together with the nitrogen to which they are attached are cyclized, the resulting R.sup.8 group is morpholinyl. In some cases, where R.sup.6 and R.sup.7 together with the nitrogen to which they are attached are cyclized, the resulting R.sup.8 group is optionally substituted with one or more substituent groups. For example, the optional substituent on the R.sup.8 group can be --OH, --COOH, or --CH.sub.2OH.
[0103] In certain embodiemnts, X.sup.1 is --F, --Cl, --Br, or --OCH.sub.3. For example, X.sup.1 can be --F, or X.sup.1 can be --Cl, or X.sup.1 can be --Br, or X.sup.1 can be --OCH.sub.3.
[0104] In certain embodiments, X.sup.2 is --SO.sub.2R' or --CO.sub.2R', where R' is --H, --CH.sub.3, or --CH.sub.2CH.sub.3. For example, X.sup.2 can be --SO.sub.2R', or X.sup.2 can be --CO.sub.2R'. In some instances, R' is --H, --CH.sub.3, or --CH.sub.2CH.sub.3. For example, R' can be --H, or R' can be --CH.sub.3, or R' can be --CH.sub.2CH.sub.3.
[0105] In certain embodiments, X.sup.3 is --SO.sub.2CF.sub.3, --SO.sub.2CH.sub.3, or --NO.sub.2. For example, X.sup.3 can be --SO.sub.2CF.sub.3, or X.sup.3 can be --SO.sub.2CH.sub.3, or X.sup.3 can be --NO.sub.2.
[0106] In certain embodiments, X.sup.5 is --F, --Br, --Cl, --H, or --OCH.sub.3. For example, X.sup.5 can be --F, or X.sup.5 can be --Br, or X.sup.5 can be --Cl, or X.sup.5 can be --H, or X.sup.5 can be --OCH.sub.3.
[0107] In certain embodiments, the compound of Formula (III) is phosphorylated. For example, compounds of Formula (III) can be phosphorylated on the R.sup.8 group.
[0108] In certain embodiments, the compounds of Formula (III) also include salts or stereoisomers thereof.
[0109] Compounds of Formula (III) find use in methods of treating a pulmonary disease in a subject as described herein. For example, the pulmonary disease can be idiopathic pulmonary fibrosis (IPF), or the pulmonary disease can be chronic obstructive pulmonary disease (COPD).
Evaluating Compounds for Senolytic Activity
[0110] These and other compounds can be evaluated on the molecular level for their ability to perform as candidate senolytic agents for use according to this invention. For example, where the therapy includes triggering apoptosis of senescent cells by way of Bcl-2, Bcl-xL, Bcl-w, or other Bcl family proteins, compounds of the invention can be tested for their ability to inhibit binding between one or more Bcl proteins and their respective cognate ligand and thereby cause senolysis.
[0111] For example, a suitable assay can be a homogeneous assay (an assay that does not require a separation step) for purposes of determining binding to the Bcl isoforms, which is based on oxygen channeling that is marketed by PerkinElmer Inc., Waltham, Massachusetts: see Eglin et al., Current Chemical Genomics, 2008, 1, 2-10. The test compound is combined with the target Bcl protein and a peptide representing the corresponding cognate ligand, labeled with biotin. The mixture is then combined with streptavidin bearing luminescent donor beads and luminescent acceptor beads, which proportionally reduces luminescence if the compound has inhibited the peptide from binding to the Bcl protein.
[0112] Alternatively, or in addition, compounds of the invention can be evaluated for an ability to kill senescent cells specifically, as described herein in Example 2. Compounds can be screened for biological activity in an assay using senescent cells. Cultured cells are contacted with the compound, and the degree of cytotoxicity or inhibition of the cells is determined. The ability of the compound to kill or inhibit senescent cells can be compared with the effect of the compound on normal cells that are freely dividing at low density, and normal cells that are in a quiescent state at high density. By way of example, cultured cells, such as, for example, human target tissue fibroblast IMR90 cell lines and HUVEC cells, are contacted with the test compound, and the degree of cytotoxicity or inhibition of the cells is determined using, for example, a thermostable luciferase to enable reaction conditions that generate a stable luminescent signal while simultaneously inhibiting endogenous ATPase released during cell lysis.
Lung Diseases --Classification of Lung Disease According to Underlying Pathophysiology
[0113] As a guide to treating pulmonary diseases in accordance with this invention, the diseases can be classified according to the primary underlying pathophysiology. Diseases that fall within the same classification are amenable to applying senolytic medicine with the same principles and with similar objectives.
[0114] Pulmonary diseases suitable for treatment are discussed in more detail below, within the following classifications: [0115] TYPE 1: Restrictive: result from diseases that cause a reduction in lung compliance and in turn reduction in lung vital capacity and total lung volume commonly a result of thickening of the lung interstitium exemplified by fibrotic diseases. [0116] TYPE 2: Obstructive: result from diseases that cause air trapping in the lung and in turn a reduction in expiratory volume and increased total lung volume commonly a result of airway obstruction or destruction exemplified by diseases such as COPD and asthma. [0117] TYPE 3: Vascular: result from a disruption of the normal functioning of the blood vessels resulting from disorders affecting the cellular components of the vessel including but not limited to the endothelium. These disruptions may result in diseases characterized by vascular inflammation and increased vessel tone (e.g. ANCA-vasculitis and pulmonary hypertension) ultimately leading to dysfunction of the physiologic function of the lung and heart. [0118] TYPE 4: Genetic: result from genetic abnormalities that result in lung disease that affects multiple anatomic components of the lung and extra-pulmonary organs. These include but are not limited to cystic fibrosis and alpha-1 antitrypsin. [0119] TYPE 5: Infections: result from pathogenic microorganisms, such as bacteria, viruses, parasites or fungi, may infect any anatomical location including the airways, alveoli and pleura. They may result in symptomatic (e.g. pneumonia) or asymptomatic latent disease (e.g. tuberculosis).
[0120] This classification is provided to assist in understanding and applying the invention to a particular patient and is not meant to limit application of this technology. Certain conditions may invoke several of these categories: for example, an inflammatory process may contribute to pathological processes having other underlying causes. Similarly, the SASP may trigger additional pathologic processes regardless of the primary insult.
Pulmonary Diseases Suitable for Treatment with the Compounds of the Invention:
[0121] Provided in the sections that follow is a discussion of specific lung diseases arranged by broad etiologic category as discussed above, that are candidates for treatment with a senolytic agent in accordance with this invention. The degree to which a particular pulmonary disease will be amenable to treatment with a senolytic agent will depend on the degree and extent senescent cells play a role in disease pathology or symptomatology. The treatment protocol and patient management are within the judgment of the managing clinician. The efficacy of the therapy can be determined with clinical, physiological and radiological evaluation.
[0122] In certain embodiments, compounds of Formula (I) as described herein find use in methods of treating a pulmonary disease in a subject as described herein. In certain embodiments, compounds of Formula (II) as described herein find use in methods of treating a pulmonary disease in a subject as described herein. In certain embodiments, compounds of Formula (III) as described herein find use in methods of treating a pulmonary disease in a subject as described herein.
TYPE 1: Restrictive
[0123] Physiologic restriction of the lung result from diseases that cause a reduction in lung compliance and in turn reduction in lung vital capacity and total lung volume commonly a result of thickening of the lung interstitium exemplified by fibrotic diseases, such as, for example, idiopathic pulmonary fibrosis (IPF) and connective tissue disease-associated lung fibrosis such as systemic sclerosis (SSc).
[0124] IPF is a chronic and progressive fibrotic lung disease characterized by stiffening and scarring of the lung, which can lead to respiratory failure, pulmonary hypertension and increases the risk for lung cancer, and heart failure. Fibrosis is associated with dysfunctional repair of the lung interstitium epithelium. Fibroblasts are activated, production of extracellular matrix proteins is increased, and transdifferentiation to contractile myofibroblasts contribute to wound contraction. A provisional matrix plugs the injured epithelium and provides a scaffold for epithelial cell migration, involving an epithelial-mesenchymal transition (EMT). Blood loss associated with epithelial injury induces platelet activation, production of growth factors, and an acute inflammatory response. Normally, the epithelial barrier heals and the inflammatory response resolves. However, in fibrotic disease the fibroblast response continues, resulting in unresolved wound healing. Formation of fibroblastic foci is a feature of the disease, reflecting locations of ongoing fibrogenesis.
[0125] The general approach and objectives of senolytic therapy for restrictive conditions are based on elimination of senescent cells from the area of the lung tissue that is central to the fibrogenic process. For example, based on Unity data in human IPF lung tissue, this includes the distal lung epithelium which has been demonstrated to express the senescence marker p16.
[0126] Other subjects at risk of developing fibrotic diseases, for example, include but are not limited to, those exposed to environmental or occupational pollutants, such as asbestosis and silicosis, those who have a connective tissue diseases such as RA, SLE, scleroderma, sarcoidosis, those who take certain medications, including, for example, amiodarone, bleomycin, busufan, methotrexate, and nitrofurantoin; those subject to radiation therapy to the chest; and those whose family member have pulmonary fibrosis.
TYPE 2: Obstructive
[0127] Physiologic obstruction of the lung result from diseases that cause air trapping in the lung and in turn a reduction in expiratory volume and increased total lung volume commonly a result of airway obstruction or destruction exemplified by diseases such as, for example, chronic obstructive pulmonary diseases (COPD) and asthma.
[0128] COPD is a lung disease defined by persistently poor airflow resulting from the breakdown of lung tissue, emphysema, and the dysfunction of the small airways, obstructive bronchiolitis. Primary symptoms of COPD include shortness of breath, wheezing, chest tightness, chronic cough, and excess sputum production. Elastase from cigarette smoke-activated neutrophils and macrophages can disintegrate the extracellular matrix of alveolar structures, resulting in enlarged air spaces and loss of respiratory capacity. COPD can be caused by, for example, tobacco smoke, cigarette smoke, cigar smoke, secondhand smoke, pipe smoke, occupational exposure, exposure to dust, smoke, fumes, and pollution, occurring over decades thereby implicating aging as a risk factor for developing COPD.
[0129] The processes that cause lung damage include, for example, oxidative stress produced by the high concentrations of free radicals in tobacco smoke, cytokine release due to the inflammatory response to irritants in the airway, and impairment of anti-protease enzymes by tobacco smoke and free radicals, allowing proteases to damage the lungs. Genetic susceptibility can also contribute to the disease. In about 1% percent of people with COPD, the disease results from a genetic disorder that causes low level production of alpha-1-antitrypsin in the liver. Alpha-1-antitrypsin is normally secreted into the bloodstream to help protect the lungs.
[0130] Symptoms of COPD can include any one of shortness of breath, wheezing, chest tightness, having to clear one's throat first thing in the morning because of excess mucus in the lungs, a chronic cough that produces sputum that can be clear, white, yellow or greenish, cyanosis, frequent respiratory infections, lack of energy, and unintended weight loss. Subjects with COPD can also experience exacerbations, during which symptoms worsen and persist for days or longer. Symptoms of pulmonary fibrosis include, for example, shortness of breath, particularly during exercise; dry, hacking cough; fast, shallow breathing; gradual, unintended weight loss; fatigue; aching joints and muscles; and clubbing of the fingers or toes.
TYPE 3: Vascular
[0131] These conditions are characterized by a disruption of the normal functioning of the blood vessels resulting from disorders affecting the cellular components of the vessel including, but not limited to, the endothelium. These disruptions may result in diseases characterized by such findings as vascular inflammation, increased vessel tone (vasoconstriction) and restricted blood flow ultimately leading to damage and physiologic dysfunction of the lung and heart. It also includes local deficiencies that arise in a given part of a body resulting from issues affecting blood flow but not the vessel itself, such as vasoconstriction, thrombosis, or embolism. Examples of vascular pulmonary diseases include, but are not limited to, pulmonary hypertension and vasculitis such as Wegener's granulomatosis.
[0132] The general approach and objectives of senolytic therapy for ischemic or vascular conditions are based on elimination of senescent cells from the vasculature and decrease the associated SASP factor impact on surrounding cells or area of the lung tissue that is central to the effects of vascular dysfunction. Thus, the senolytic agent can be delivered either systemically or directly in the vasculature.
[0133] Pulmonary hypertension (PH) is a pathophysiological disorder that may involve multiple clinical conditions and can complicate many pulmonary and cardiovascular diseases. It is defined physiologically by the resulting hemodynamic change of a mean pulmonary artery pressure (mPAP) at rest greater than 25 mmHg The clinical pathogenic categories of PH are described based on broad etiologies: pulmonary arteries (including pulmonary veno-occlusion and pulmonary capillary dysfunction), left heart disease, lung disease/hypoxia, artery obstruction and from unclear or multi-factorial causes (2015 ESC/ERS Guidelines; Galie N. et al., Eur Respir J 2015; 46:903). The overall treatment goal in patients with PH is to maintain good exercise capacity, good quality of life, good Right Ventricular function and a low mortality risk. Specifically, this means bringing and/or keeping the patient in WHO-FC II whenever possible.
[0134] TYPE 4: Genetic
[0135] Genetic respiratory conditions are characterized as a disease that is caused by a mutation, deletion, or insertion in an individual's DNA sequence. Genetic disorders can be grouped into three main categories: (1) Single gene disorders: disorders caused by defects in one particular gene, often with simple and predictable inheritance patterns such as dominant, recessive and x-linked; (2) Chromosome disorders: disorders resulting from changes in the number or structure of the chromosomes; and (3) Multifactorial disorders (complex diseases): disorders caused by changes in multiple genes, often in a complex interaction with environmental and lifestyle factors such as diet or cigarette smoke. Examples of genetic pulmonary diseases include cystic fibrosis (CF) and alpha-1 antitrypsin deficiency (A1AT).
[0136] CF is a monogenic autosomal recessive disease that is caused by mutations in CFTR, located on chromosome 7. The CFTR protein is an ion channel that regulates transport of chloride ions (Cl--) in epithelial cells in the airways, as well as in the pancreas, liver, intestine and skin. The various CFTR mutations cause different CFTR protein defects, which impair transport of chloride and sodium across epithelial surfaces, leading to thick viscous secretions (e.g. mucus or phlegm).
[0137] Consequently, dysfunction of the exocrine glands throughout the body leads to elevated sweat chloride, pancreatic insufficiency, recurrent pulmonary infection, hepatobiliary disease, and infertility. The diagnosis of CF relies primarily on clinical evidence and is confirmed by elevated sweat chloride or CFTR mutations in two alleles. CFTR modulators and potentiators are drugs that modify the function of CFTR to improve lung function and reduce symptoms and pulmonary exacerbations.
[0138] The general approach and objectives of senolytic therapy for genetic conditions are based on the following: Genetic disorders of the pulmonary system can affect all anatomic layers and are associated with cellular defects that may lead to an accelerated aging phenotype, caused or mediated at least in part by senescent cells. An inheritable susceptibility to certain lung diseases suggests that the accumulation of disease-mediating senescent cells may directly or indirectly be influenced by genetic components, which again may lead to earlier presentation. Genetic disorders demonstrate a multifactorial cascade with senescent cells and SASP production contributing to ongoing cell dysfunction and degeneration/death. These disorders can benefit from senolytic therapy because senescent cells and their associated SASP factors mediate associated contributions to ongoing cell dysfunction, cell loss, and disease progression via blockage of the angiogenic, inflammatory, fibrotic, and extracellular matrix-modifying proteins present in the pathophysiology.
TYPE 5: Infectious Pulmonary Diseases
[0139] These are diseases caused by pathogenic microorganisms, such as bacteria, viruses, parasites or fungi; the diseases can be spread, directly or indirectly, from one person to another. Infectious pulmonary diseases can be caused by numerous infectious agents, including but not limited to Streptococcus, Myocobacteria, Pneumocystis, Blastomyces, Paragonimus and human immunodeficiency virus (HIV). These infections present with either or both acute and chronic clinical features including cough, lung infiltrates, hemoptysis and respiratory distress which may become life threatening and in many cases require long term anti-infective therapies with significant associated comorbidities.
[0140] The general approach and objectives of senolytic therapy for infections conditions can be based upon the following: Infectious disorders of the pulmonary system may impact all anatomic locations of the lung and appear to occur more frequently in the elderly, suggesting senescence. Infectious agents may contribute to the induction of senescence and a multifactorial cascade with senescent cells and SASP production contributing to ongoing cell dysfunction. Once present, senescent cells may in turn impact the ability to fight infection.
[0141] Senescent cells have an impaired ability to control viral replication (Kim et al., Enhanced Viral Replication by Cellular Replicative Senescence., Immune Network., 2016 October; 16(5):286-295), which is in line with the known increased susceptibility to infection that occurs with age. Senescence and the ability to respond to infectious agents are a category of lung disease that can be significantly impacted by senolytic therapy. Elimination of senescent cells and their associated SASP factors can ameliorate damage to the cellular microenvironment.
Routes of Administration
[0142] In one embodiment, the pharmaceutical senolytic compositions of the invention are formulated for administration by inhalation. Suitable pharmaceutical compositions for administration by inhalation will typically be in the form of an aerosol or a powder. [0143] Aerosol Delivery:
[0144] Compounds of the invention may be directly administered as an aerosol to a site of pulmonary pathology as described above. The aerosol may also be delivered to the pulmonary compartment for absorption into the pulmonary vasculature for therapy or prophylaxis of extra-pulmonary pathologies such as fibrosis for example, or pulmonary or intra-nasal delivery for extra-pulmonary or extra-nasal cavity diseases.
[0145] When administered by inhalation using a pressurized container, the pharmaceutical compositions of the invention will typically comprise the active ingredient and a suitable propellant, such as, for example, dichlorodifluoromethane, trichlorofluoromethane, dichlorotetrafluoroethane, carbon dioxide or other suitable gas. Additionally, the pharmaceutical composition may be in the form of a capsule or cartridge (made, for example, from gelatin) comprising a compound of the invention and a powder suitable for use in a powder inhaler. Suitable powder bases include, by way of example, lactose or starch.
[0146] Compositions of the invention may be administered using well-known delivery devices, such as a metered-dose inhaler, a dry powder inhaler, a nebulizer, a vaporizer, or a similar delivery device. [0147] A. Meter Dose Inhaler (MDI):
[0148] A propellant driven inhaler (pMDI) releases a metered dose of medicine upon each actuation. The medicine is formulated as a suspension or solution of a drug substance in a suitable propellant such as a halogenated hydrocarbon. pMDIs are described in, for example, Newman, S. P., Aerosols and the Lung, Clarke et al., eds., pp. 197-224 (Butterworths, London, England, 1984). [0149] B. Dry Powder Inhaler (DPI):
[0150] There are two major designs of dry powder inhalers. One design is the metering device in which a reservoir for the drug is placed within the device and the patient adds a dose of the drug into the inhalation chamber. The second is a factory-metered device in which each individual dose has been manufactured in a separate container. Both systems depend upon the formulation of drug into small particles of mass median diameters from about 1 to about 5 micron, and usually involve co-formulation with larger excipient particles (typically 100 micron diameter lactose particles). Drug powder is placed into the inhalation chamber (either by device metering or by breakage of a factory-metered dosage) and the inspiratory flow of the patient accelerates the powder out of the device and into the oral cavity. Non-laminar flow characteristics of the powder path cause the excipient-drug aggregates to decompose, and the mass of the large excipient particles causes their impaction at the back of the throat, while the smaller drug particles are deposited deep in the lungs. [0151] C. Nebulizers:
[0152] Any known inhalation nebulizer suitable to provide delivery of a medicament as described herein may be used in the various embodiments and methods described herein. Such nebulizers include, e.g., jet nebulizers, ultrasonic nebulizers, pulsating membrane nebulizers, nebulizers with a vibrating mesh or plate with multiple apertures, and nebulizers comprising a vibration generator and an aqueous chamber (e.g., ParieFlow.RTM.). [0153] D. Vaporizers:
[0154] Any known vaporizer suitable to provide delivery of a medicament as described herein may be used in the various embodiments and methods described herein. For example, a vaporizer may be used to vaporize a pharmaceutical composition as described herein, such that the vaporized components of the pharmaceutical composition can be inhaled by a user. In some instances, the vaporizer applies sufficient heat to the pharmaceutical composition to vaporize one or more components of the pharmaceutical composition, such as the active agent or drug substance. The pharmaceutical composition for use in a vaporizer may be provided in solid form or liquid form prior to vaporization.
Formulation of Medicaments
[0155] A pulmonary preparation can be prepared by mixing a senolytic agent with a pharmaceutically acceptable base or carrier and as needed one or more pharmaceutically acceptable excipients.
[0156] Ingredients acceptable in a pulmonary formulation are excipients or carriers that cause little to no pulmonary irritation, provide suitable preservation if needed, and deliver one or more agents in a suitable volume. Examples of a base or carrier include water; an aqueous solvent such as a polar solvent; a polyalcohol; a vegetable oil; and an oily base. Examples of the base or carrier for an intrapulmonary injection include water for injection and physiological saline.
[0157] For pulmonary delivery, a senolytic agent may be combined with acceptable excipients for use in and around the lung, such as a surfactant, preservatives, co-solvents, a flavor or cooling agent, an antiseptic, a bactericide or antibacterial agent, a pH adjusting agent, a tonicity agent, a chelating agent, a buffering agent, a stabilizer, an antioxidant, viscosity enhancers, penetration enhancers, sodium chloride and a thickening agent. In some cases, a composition for intrapulmonary injection may contain one or more of a solubilizing agent, a suspending agent, a tonicity agent, a buffering agent, a soothing agent, a stabilizer, and an antiseptic. The pulmonary composition carrier and excipients can be combined to form an aqueous, sterile pulmonary suspension, solution, or viscous or semi-viscous gels or other types of solid or semisolid composition such as an ointment.
[0158] Exemplary excipients and additives that can be used include surfactants (for example, polyoxyethylene and block copolymers); buffers and pH adjusting agents (for example, hydrochloric acid, sodium hydroxide, phosphate, citrate, and sodium cyanide); tonicity agents (for example, sodium bisulfite, sodium sulfite, glycerin, and propylene glycol); chelating agents (for example, ascorbic acid, sodium edetate, and citric acid); flavors; coloring agents; antiseptics; bactericides; antibacterial agents; and the like.
[0159] Pulmonary solution formulations may be prepared by dissolving the agent in a physiologically acceptable isotonic aqueous buffer. Further, the pulmonary solution may include an acceptable surfactant to assist in dissolving the agent. Viscosity building compounds, such as hydroxymethyl cellulose, hydroxyethyl cellulose, methylcellulose, polyvinylpyrrolidone may be added to improve the retention of the compound.
[0160] Sterile pulmonary gel formulations may be prepared by suspending the agent in a hydrophilic base prepared from the combination of, for example, CARBOPOL.RTM.-940. VISCOAT.RTM. (Alcon Laboratories, Inc., Fort Worth, Tex.) may be used for intrapulmonary injection. Other compositions of the present invention may contain penetration enhancing materials such as CREMOPHOR.RTM. (Sigma Aldrich, St. Louis, Mo.) and TWEEN.RTM. 80 (polyoxyethylene sorbitan monolaureate, Sigma Aldrich), in the event the agents of the present invention are less penetrating in the lung.
[0161] This invention provides commercial products that are kits that enclose one or more unit doses of one or more of the agents or compositions described in this disclosure. Such kits typically comprise a pharmaceutical preparation in one or more containers. The preparatoins may be provided as one or more unit doses (either combined or separate). The kit may contain a device such as a syringe for administration of the agent or composition in or around the lung of a subject in need thereof. The product may also contain or be accompanied by an informational package insert describing the use and attendant benefits of the drugs in treating the senescent cell associated lung disease, and optionally an appliance or device for delivery of the composition.
[0162] A unit dose refers to a physically discrete unit suitable as a single dosage for a subject in need thereof, where each unit dose contains a predetermined quantity of a compound of the invention in an amount sufficient to produce the desired therapeutic effect. The compound may be provided in the usit dose in association with a pharmaceutically acceptable diluent, carrier and/or vehicle. The amount of the compound in each unit dose may depend on the particular compound employed and the effect to be achieved, and the pharmacodynamics associated with the compound in the user.
Combination of Senolytic Agents with Approved Standard-of-Care Therapies
[0163] Senolytic agents for treating pulmonary diseases and conditions can be combined with other pharmaceutical agents that are approved for clinical use. Since the removal of senescent cells works by a different mechanism from current therapies, the two agents can operate synergistically or additively to minimize the administration schedule and improve outcomes. The senolytic agent will remove senolytic cells in the lung that are promoting persistence and progression of disease-related pathophysiology.
[0164] The methods of this invention for treating or reducing the likelihood of a pulmonary disease or condition can also be used for treating a subject who is aging and has loss of pulmonary function, or degeneration of pulmonary tissue. The respiratory system can undergo various anatomical, physiological and immunological changes with age. The structural changes include chest wall and thoracic spine deformities that can impair the total respiratory system compliance resulting in increased effort to breathe. The respiratory system undergoes structural, physiological, and immunological changes with age. An increased proportion of neutrophils and lower percentage of macrophages can be found in bronchoalveolar lavage (BAL) of older adults compared with younger adults. Persistent low grade inflammation in the lower respiratory tract can cause proteolytic and oxidant-mediated injury to the lung matrix resulting in loss of alveolar unit and impaired gas exchange across the alveolar membrane seen with aging. Sustained inflammation of the lower respiratory tract can predispose older adults to increased susceptibility to toxic environmental exposure and accelerated lung function decline. Oxidative stress exacerbates inflammation during aging. Alterations in redox balance and increased oxidative stress during aging precipitate the expression of cytokines, chemokines, and adhesion molecules, and enzymes. Constitutive activation and recruitment of macrophages, T cells, and mast cells foster release of proteases leading to extracellular matrix degradation, cell death, remodeling, and other events that can cause tissue and organ damage during chronic inflammation.
[0165] The effects of the treatment can be determined using techniques that evaluate mechanical functioning of the lung, for example, techniques that measure lung capacitance, elastance, and airway hypersensitivity can be performed. To determine lung function and to monitor lung function throughout treatment, any one of numerous measurements can be obtained, for example, expiratory reserve volume (ERV), forced vital capacity (FVC), forced expiratory volume (FEV) (e.g., FEV in one second, FEV1), FEV1/FEV ratio, forced expiratory flow 25% to 75%, and maximum voluntary ventilation (MVV), peak expiratory flow (PEF), slow vital capacity (SVC). Total lung volumes include total lung capacity (TLC), vital capacity (VC), residual volume (RV), and functional residual capacity (FRC). Gas exchange across alveolar capillary membrane can be measured using diffusion capacity for carbon monoxide (DLCO). Peripheral capillary oxygen saturation (SpO2) can also be measured; normal oxygen levels are typically between 95% and 100%. An SpO2 level below 90% suggests the subject has hypoxemia. Values below 80% are considered critical and require intervention to maintain brain and cardiac function and avoid cardiac or respiratory arrest.
Treatment Design
[0166] Senescent cells accumulate with age, which is why conditions mediated by senescent cells occur more frequently in older adults. In addition, different types of stress on pulmonary tissues may promote the emergence of senescent cells and the phenotype they express. Cell stressors include oxidative stress, metabolic stress, DNA damage (for example, as a result of environmental ultraviolet light exposure or genetic disorder), oncogene activation, and telomere shortening (resulting, for example, from hyperproliferation). Pulmonary tissues that are subject to such stressors may have a higher prevalence of senescent cells, which in turn may lead to presentation of certain lung diseases at an earlier age, or in a more severe form. An inheritable susceptibility to certain lung diseases suggests that the accumulation of disease-mediating senescent cells may directly or indirectly be influenced by genetic components, which can lead to earlier presentation.
[0167] To treat a particular pulmonary disease with a senolytic agent according to this invention, the therapeutic regimen will depend on the location of the senescent cells, and the pathophysiology of the disease.
[0168] In some embodiments, one or more doses of a compound or pharmaceutical composition of the invention are administered to a subject in need thereof. The frequency of administration of the compound or pharmaceutical composition can vary depending on any of a variety of factors, e.g., severity of the symptoms, condition of the subject, etc. For example, in some embodiments, the compound or pharmaceutical composition is administered once per month, twice per month, three times per month, every other week (qow), once per week (qw), twice per week (biw), three times per week (tiw), four times per week, five times per week, six times per week, every other day (qod), daily (qd), twice a day (qid), or three times a day (tid), or the like.
[0169] Compounds that may be useful for clearing senescent cells in or around the lung for purposes of treating pulmonary diseases according to this invention include Bcl-2 inhibitors, Bcl-xL inhibitors, MDM2 inhibitors, and Akt inhibitors. See U.S. Pat. Nos. 8,691,184, 9,096,625, and 9,403,856; published applications WO 2015/017159, WO 2015/116740, WO 2016/127135, WO 2017/008060, and WO/2017/101851.
[0170] Candidate senolytic agents that act as Bcl 2, Bcl w, and Bcl xL inhibitors can be characterized as a benzothiazole-hydrazone, an amino pyridine, a benzimidazole, a tetrahydroquinolin, or a phenoxyl compound. Examples of compounds that inhibit Bcl isoforms include WEHI 539, A 1155463, ABT 737, and ABT 263 (Navitoclax).
INCORPORATION BY REFERENCE
[0171] For all purposes in the United States and in other jurisdictions where effective, each and every publication and patent document cited in this disclosure is hereby incorporated herein by reference in its entirety for all purposes to the same extent as if each such publication or document was specifically and individually indicated to be incorporated herein by reference.
[0172] U.S. Application Publication No. 2016/0339019 (Laberge et al.) and International application publication no. WO 2016127135 (David et al.) are hereby incorporated herein by reference in their entireties for all purposes, including but not limited to the identification, formulation, and use of compounds capable of eliminating or reducing the activity of senescent cells and treating pulmonary diseases and conditions. U.S. Pat. Nos. 8,691,184, 9,096,625, and 9,403,856 (Wang et al.) are hereby incorporated herein by reference in their entireties for all purposes, including the features of compounds in the Bcl library, their preparation and use.
EMBODIMENTS OF THE INVENTION
[0173] Embodiments of the invention of the present disclosure can be described by the following clauses.
[0174] Clause 1. A compound according to Formula (II):
##STR00082##
wherein: [0175] X.sup.1 is --Cl; [0176] X.sup.2 is --COOH or --SO.sub.2CH.sub.3; [0177] X.sup.3 is --SO.sub.2CF.sub.3, --SO.sub.2CH.sub.3, or --NO.sub.2; [0178] X.sup.5 is --F or --H; [0179] R.sup.1 is --CH(CH.sub.3).sub.2; [0180] R.sup.2 is --CH.sub.3; [0181] R.sup.3 and R.sup.4 are both --H; [0182] n is 2; [0183] R.sup.6 is selected from --OH, --OR.sup.7,
##STR00083##
[0183] and [0184] R.sup.7 is --PO(OH).sub.2, [0185] or a salt or a stereoisomer thereof.
[0186] Clause 2. The compound of Clause 1, wherein X.sup.2 is --COOH.
[0187] Clause 3. The compound of Clause 1, wherein X.sup.2 is --SO.sub.2CH.sub.3.
[0188] Clause 4. The compound of Clause 1, wherein X.sup.3 is --SO.sub.2CF.sub.3.
[0189] Clause 5. The compound of Clause 1, wherein X.sup.3 is --SO.sub.2CH.sub.3.
[0190] Clause 6. The compound of Clause 1, wherein X.sup.3 is --NO.sub.2.
[0191] Clause 7. The compound of any of Clauses 1 to 6, wherein X.sup.5 is --F.
[0192] Clause 8. The compound of any of Clauses 1 to 6, wherein X.sup.5 is --H.
[0193] Clause 9. The compound of any of Clauses 1 to 8, wherein X.sup.6 is --OH.
[0194] Clause 10. The compound of any of Clauses 1 to 8, wherein X.sup.6 is --OR.sup.7.
[0195] Clause 11. The compound of any of Clauses 1 to 8, wherein X.sup.6 is
##STR00084##
[0196] Clause 12. The compound of any of Clauses 1 to 8, wherein X.sup.6 is
##STR00085##
[0197] Clause 13. The compound of any of Clauses 1 to 8, wherein X.sup.6 is
##STR00086##
[0198] Clause 14. The compound of any of Clauses 1 to 13, wherein the carboxyl group in X.sup.2 is phosphorylated.
[0199] Clause 15. The compound of Clause 1, wherein the compound is selected from the group consisting of:
##STR00087##
[0200] Clause 16. The compound of any preceding Clause, which has pro-apoptotic activity.
[0201] Clause 17. The compound of any preceding Clause, which specifically kills senescent cells compared with non-senescent cells, said senescent cells being defined as non-cancerous cells that express p16.
[0202] Clause 18. The compound of any preceding Clause, which specifically kills cancer cells compared with non-cancer cells of the same tissue type.
[0203] Clause 19. The compound of any preceding Clause, which has an IC.sub.50 for Bcl-xL of 1 nM or less.
[0204] Clause 20. The compound of any preceding Clause, which has an IC.sub.50 for Bcl-2 of 10 nM or less.
[0205] Clause 21. The compound of any preceding Clause, which has an IC.sub.50 for Bcl-xL of 1 nM or less and an IC.sub.50 for Bcl-2 of 10 nM or less.
[0206] Clause 22. A pharmaceutical composition comprising a compound according to any preceding Clause in a pharmaceutically compatible excipient.
[0207] Clause 23. A method of selectively removing senescent cells and/or cancer cells from a mixed cell population or tissue, comprising contacting a cell, a cell population or a tissue with a compound according to any of Clauses 1 to 21 or a pharmaceutical composition according to Clause 22.
[0208] Clause 24. A method of treating a senescence related condition in a tissue in a subject, wherein the senescence related condition is characterized as being caused or mediated at least in part by senescent cells, or is characterized as having an overabundance of senescent cells in or around the tissue, in comparison with unaffected tissue, the method comprising: [0209] administering to a tissue of a subject in need thereof, an amount of a compound according to any of Clauses 1 to 21 or a pharmaceutical composition according to Clause 22 that is effective to selectively remove senescent cells from the tissue, thereby relieving or ameliorating one or more signs or symptoms of a senescence related condition in the subject.
[0210] Clause 25. A unit dose of a pharmaceutical composition comprising: [0211] an amount of a compound that inhibits Bcl function configured for use in the treatment of a senescence associated condition that is caused or mediated at least in part by senescent cells, [0212] wherein the compound is a compound according to any of Clauses 1 to 21, [0213] wherein the pharmaceutical composition contains a formulation of the compound configured for administration to a target tissue in a subject that manifests the senescence associated condition, and [0214] wherein the formulation and the amount of the compound in the unit dose configure the unit dose to be effective in selectively removing senescent cells in or around the tissue in the subject, thereby decreasing the severity of one or more signs or symptoms of the condition without causing adverse effects in the subject when administered to the tissue as a single dose.
[0215] Clause 26. The unit dose of Clause 25, packaged with an informational insert describing the use and attendant benefits of the drugs in treating the senescent cell associated condition.
[0216] Clause 27. A compound according to any of Clauses 1 to 21 or a pharmaceutical composition according to claim 24 for use in selectively eliminating senescent cells from a tissue or mixed cell population or for use in treating a senescence-related condition.
[0217] Clause 28. Use of a compound according to any of Clauses 1 to 21 in the manufacture of a medicament for treating a senescence-related condition.
[0218] Clause 29. The method, unit dose, or use of any of Clauses 24 to 28, wherein the condition is osteoarthritis.
[0219] Clause 30. The method, unit dose, or use of any of Clauses 24 to 28, wherein the condition is an ophthalmic condition.
[0220] Clause 31. The method, unit dose, or use of any of Clauses 24 to 28, wherein the condition is a pulmonary disease.
[0221] Clause 32. A method of treating cancer, comprising administering to a tissue of a subject in need thereof an amount of a compound according to any of Clauses 1 to 21 or a pharmaceutical composition according to Clause 22 effective to selectively remove cancer cells from the tissue.
[0222] Clause 33. A compound according to any of Clauses 1 to 21 or a pharmaceutical composition according to claim 22 for use in selectively eliminating cancer cells from a tissue or mixed cell population or for use in treating cancer.
[0223] Clause 34. A method of treating a pulmonary disease in a subject, comprising administering to the subject in need thereof a therapeutically effective amount of a pharmaceutical composition comprising: [0224] a compound of Formula (I):
##STR00088##
[0224] wherein: [0225] X.sup.1 is --Cl; [0226] X.sup.2 is --COOH or --SO.sub.2CH.sub.3; [0227] X.sup.3 is --SO.sub.2CF.sub.3; --SO.sub.2CH.sub.3; or --NO.sub.2; [0228] X.sup.5 is --F or --H; [0229] R.sup.1 is --CH(CH.sub.3).sub.2; [0230] R.sup.2 is --CH.sub.3; [0231] R.sup.3 and R.sup.4 are both --H; [0232] n is 2; and [0233] R.sup.6 is selected from --OR.sup.7,
##STR00089##
[0233] and [0234] R.sup.7 is --H or --PO(OH).sub.2, [0235] or a salt or a stereoisomer thereof; and [0236] a pharmaceutically compatible excipient.
[0237] Clause 35. The method of Clause 34, wherein X.sup.2 is --COOH.
[0238] Clause 36. The method of Clause 34, wherein X.sup.2 is --SO.sub.2CH.sub.3.
[0239] Clause 37. The method of Clause 34, wherein X.sup.3 is --SO.sub.2CF.sub.3.
[0240] Clause 38. The method of Clause 34, wherein X.sup.3 is --SO.sub.2CH.sub.3.
[0241] Clause 39. The method of Clause 34, wherein X.sup.3 is --NO.sub.2.
[0242] Clause 40. The method of any of Clauses 34 to 39, wherein X.sup.5 is --F.
[0243] Clause 41. The method of any of Clauses 34 to 39, wherein X.sup.5 is --H.
[0244] Clause 42. The method of any of Clauses 34 to 39, wherein R.sup.6 is --OR.sup.7.
[0245] Clause 43. The method of any of Clauses 34 to 39, wherein R.sup.6 is
##STR00090##
[0246] Clause 44. The method of any of Clauses 34 to 39, wherein R.sup.6 is
##STR00091##
[0247] Clause 45. The method of any of Clauses 34 to 44, wherein R.sup.7 is --H.
[0248] Clause 46. The method of any of Clauses 34 to 44, wherein R.sup.7 is --PO(OH).sub.2.
[0249] Clause 47. The method of any of Clauses 34 to 46, wherein the carboxyl group in X.sup.2 is phosphorylated.
[0250] Clause 48. The method of Clause 34, wherein the compound is selected from the group consisting of:
##STR00092## ##STR00093##
[0251] Clause 49. The method of Clause 34, wherein the pulmonary disease is idiopathic pulmonary fibrosis (IPF).
[0252] Clause 50. The method of Clause 34, wherein the pulmonary disease is chronic obstructive pulmonary disease (COPD).
[0253] Clause 51. The method of Clause 48, wherein the pulmonary disease is idiopathic pulmonary fibrosis (IPF).
[0254] Clause 52. The method of Clause 48, wherein the pulmonary disease is chronic obstructive pulmonary disease (COPD).
[0255] Clause 53. The method of Clause 34, wherein the administration of the pharmaceutical composition is by inhalation as an aerosol.
[0256] Clause 54. The method of Clause 48, wherein the administration of the pharmaceutical composition is by inhalation as an aerosol.
[0257] Clause 55. The method of Clause 34, wherein the pulmonary disease is a restrictive pulmonary disease.
[0258] Clause 56. The method of Clause 55, wherein the restrictive pulmonary disease is idiopathic pulmonary fibrosis (IPF) or systemic sclerosis (SSc).
[0259] Clause 57. The method of Clause 34, wherein the pulmonary disease is an obstructive pulmonary disease.
[0260] Clause 58. The method of Clause 57, wherein the obstructive pulmonary disease is chronic obstructive pulmonary diseases (COPD) or asthma.
[0261] Clause 59. The method of Clause 34, wherein the pulmonary disease is a vascular pulmonary disease.
[0262] Clause 60. The method of Clause 59, wherein the vascular pulmonary disease is pulmonary hypertension or vasculitis.
[0263] Clause 61. The method of Clause 34, wherein the pulmonary disease is a genetic pulmonary disease.
[0264] Clause 62. The method of Clause 61, wherein the genetic pulmonary disease is cystic fibrosis (CF) or alpha-1 antitrypsin deficiency (A1AT).
[0265] Clause 63. The method of Clause 34, wherein the pulmonary disease is an infectious pulmonary disease.
[0266] Clause 64. The method of Clause 63, wherein the infectious pulmonary disease is pneumonia or tuberculosis.
[0267] Clause 65. A method of treating a pulmonary disease in a subject, comprising administering to the subject in need thereof a therapeutically effective amount of a compound of Formula (III) or a phosphorylated form thereof:
##STR00094##
wherein: [0268] R.sup.1 and R.sup.2 are independently C.sub.1 to C.sub.4 alkyl; [0269] R.sup.3, R.sup.4 and R.sup.5 are independently --H or --CH.sub.3; [0270] R.sup.8 is --OH or --N(R.sup.6)(R.sup.7), wherein R.sup.6 and R.sup.7 are independently alkyl or heteroalkyl, and are optionally cyclized; [0271] X.sup.1 is --F, --Cl, --Br, or --OCH.sub.3; [0272] X.sup.2 is --SO.sub.2R' or --CO.sub.2R', where R' is --H, --CH.sub.3, or --CH.sub.2CH.sub.3; [0273] X.sup.3 is --SO.sub.2CF.sub.3; --SO.sub.2CH.sub.3; or --NO.sub.2; and [0274] X.sup.5 is --F, --Br, --Cl, --H, or --OCH.sub.3.
[0275] Clause 66. The method of Clause 65, wherein: [0276] R.sup.1 and R.sup.2 are independently C.sub.1 to C.sub.4 alkyl; [0277] R.sup.3 and R.sup.4 are independently --H or --CH.sub.3; [0278] R.sup.5 is --H; [0279] R.sup.8 is --OH or
[0279] ##STR00095## [0280] X.sup.1 is --F, --Cl, --Br, or --OCH.sub.3; [0281] X.sup.2 is --SO.sub.2R' or --CO.sub.2R', where R' is --H, --CH.sup.3, or --CH.sup.2CH.sup.3; [0282] X.sup.3 is --SO.sub.2CF.sub.3, --SO.sub.2CH.sub.3, or --NO.sub.2; [0283] X.sup.4 is --OH, --COOH or --CH.sub.2OH; [0284] X.sup.5 is --F, --Cl, or --H; and [0285] m is 1, 2, or 3.
[0286] Clause 67. The method of Clause 65, wherein: [0287] X.sup.3 is --SO.sub.2CF.sub.3 or --NO.sub.2; and [0288] R.sup.8 is --N(R.sup.6)(R.sup.7), wherein R.sup.6 and R.sup.7 are independently alkyl or heteroalkyl, and are optionally cyclized.
[0289] Clause 68. The method of Clause 66, wherein: [0290] X.sup.3 is --SO.sub.2CF.sub.3 or --NO.sub.2; and [0291] R.sup.8 is
##STR00096##
[0291] wherein X.sup.4 is --OH or --COOH.
EXAMPLES
Example 1
Cellular Senescence Burden in Diseased Human Lung Tissue
[0292] Senescent cells associated with areas of active disease in lung tissue was tested in human idiopathic pulmonary fibrosis (IPF) tissues and human scleroderma tissues taken from afflicted human patients. Human IPF tissues were procured from the University of Michigan, courtesy of Dr. Eric White. Human scleroderma tissues were procured from the Medical University of South Carolina, courtesy of Dr. Carol Browstick. The samples included both upper and lower lung lobe sections from 9 normal, 12 human IPF patients and 7 human scleroderma patients.
[0293] Immunohistochemistry staining for p16 was performed as follows. All slides were processed using a standard p16 substrate chromogen, 3,3'-diaminobenzidine tetrahydrochloride hydrate (DAB) protocol and a Leica Bond Refine Detection kit which contains a peroxide block, post primary, polymer reagent, DAB chromogen and hematoxylin counterstain. Specifically, specimen slides were incubated with hydrogen peroxide for 5 minutes to quench endogenous peroxidase activity, and then washed. A p16 primary antibody (ClNtec.RTM. p16, Roche Diagnostics) was applied for 15 minutes and then washed. A Post Primary IgG linker reagent was applied for 8 minutes, and subsequently a polymer-HRP IgG reagent was applied, before washing. The substrate chromogen, 3,3'-diaminobenzidine tetrahydrochloride hydrate (DAB) was applied for 10 minutes to visualize the complex via a brown precipitate before washing. Hematoxylin (blue) counterstaining for 1 minute allowed the visualization of cell nuclei.
[0294] Immunohistochemistry staining for p16 in human IPF lung tissue demonstrated the presence of senescent cells, see FIGS. 2A and 2B. These cells were predominantly epithelial in origin and located in areas of fibrosis and at the leading edge of the disease, which indicated accessibility by inhalation therapeutics.
[0295] Next, quantitation of the extent of fibrosis in the lung in human IPF was measured relative to normal lung tissue. Assignment of percent of observed fibrosis was made as follows: <25% fibrosis, 25-49% fibrosis, 50-74% fibrosis, and 75-100% fibrosis. A p<0.0001 for group difference among means by one-way ANOVA was performed. All human IPF samples tested positive for p16 expression, therefore confirming that an increase in p16 correlates with the progression of IPF, see FIG. 3.
[0296] Immunohistochemistry staining for p16 in human scleroderma lung tissue demonstrated the presence of senescent cells, particularly in honeycomb areas, see FIGS. 4A, 4B and 4C, as compared with normal lung, which had no p16 staining detected. Furthermore, the extent of fibrosis in the lung in human scleroderma was compared to normal lung tissue. All human scleroderma samples tested positive for p16 expression, therefore confirming that the presence of p16 correlates with scleroderma, see FIG. 5.
Example 2
Effect of A Senolytic Agent on Senescent Airway Epithelial Cells
[0297] The ability of candidate agents to eliminate senescent cells or senescent-like lung epithelial cells was measured directly in the following assay. Primary human small airway epithelial cells (SAEC) and bronchial epithelial cells (BEC) were obtained from Lonza.RTM., ATCC.RTM., and Promocell.RTM.. Cells were maintained and propagated at <75% confluency in Airway Epithelial Cell Growth Medium or Small Airway Epithelial Cell Growth Medium (Promocell.RTM.; Heidelberg, Germany) at 20% O.sub.2, 5% CO.sub.2, and .about.95% humidity. To make primary cells senescent, x-ray irradiation was employed. On day 0, SAEC/BEC cells were covered with TrypLE trypsin-containing reagent (Thermofisher Scientific, Waltham, Mass.) and incubated for 8 min until the cells rounded up and began to detach from the plate. Cells were dispersed, counted, and prepared in medium at a concentration of 188,800, 94,400, 47,200, and 23,600 cells per mL. This cell suspension was plated in 384-well plates at a volume of 25 .mu.L per well (4720, 2360, 1180, and 590 cells/well (c/w) respectively). Within 24-hours after cell plating, the 384-well plates were irradiated at 12 Gy to generate senescent cells (SnC). In addition, control 384-well plates were processed in parallel that were not irradiated and served as a control and represent normal, non-senescent cells (NsC). On day 3 the medium in each well was aspirated and replaced with 25 .mu.L fresh medium. On day 7, an exemplary test compound of the invention, Compound 1 was combined with the cells as follows. A DMSO dilution series of the test compound was prepared at 1000 times the final desired concentration in a 384-well plate. Immediately before use, the DMSO stocks were diluted 1:1000 into prewarmed complete medium. Medium was aspirated from the cells in each well, and 25 .mu.L/well of the compound containing medium was added. Compound 1 was cultured with the cells for 3 days. The assay system used the properties of a thermostable luciferase to enable reaction conditions that generated a stable luminescent signal while simultaneously inhibiting endogenous ATPase released during cell lysis. On day 10, the end of the culture period, the plates were removed from the incubator and allowed to equilibrate at room temperature for 15 minutes, then 25 .mu.L of CellTiter-Glo.RTM. reagent (Promega.RTM. Corp., Madison, Wis.) was added to each of the wells. The cell plates were placed for 30 seconds on an orbital shaker and then allowed to stand at room temperature for 30 minutes before measuring luminescence. The luminescence readings were normalized to determine % cell survival/growth and plotted against test compound concentrations, and potencies (EC50 values) for Compound 1 were determined by non-linear curve fitting in Graphpad Prism.RTM..
[0298] FIG. 6 shows the results. The concentration-response curve demonstrated sensitivity of senescent lung epithelial cell (SnC) survival to incubation with different amounts of Compound 1, whereas non-senescent cells (NsC) have very limited potency. These data show that compounds of the invention are capable of selectively eliminating senescent lung airway cells in culture.
Example 3
Measuring Senolysis in PCLS
[0299] Precision-cut lung slices (PCLS) are functional 3D organ models that can be used to ex-vivo determine effective senolysis in tissue slices obtained from normal and IPF human patients. Human PCLS will be prepared as follows. Lungs from normal, healthy individuals or IPF patients will be gently inflated with warm 1.5% agarose-DMEM mix. Afterwards, lung explants will be macroscopically assessed by an experienced pulmopathologist to identify regions of interest and exclude previously unknown medical conditions (e.g. neoplasias or infections). Next, sections (.0.4-8 mm) will be sliced in cold EBSS using a Krumdieck Tissue Slicer (Alabama Research and Development.sup.SM, Munford, Ala., USA) into approx. 250-300 .mu.m thin slices. PCLS will be washed thoroughly before cultivation in DMEM (2 slice per 500 .mu.l) under normal immersion culture conditions (37.degree. C., 5% CO2, and >95% air humidity) for up to 15 days. PCLS will be treated for 1 h with 1% Triton X-100 to serve as a dead, negative control reference.
[0300] As shown above in Example 1 and FIG. 2, senescence marker p16 expression is upregulated in idiopathic pulmonary fibrosis (IPF), therefore any significant reduction of p16 expression as measured by qPCR or abundance of p16 as measured by IHC in IPF PCLS following administration of compounds of the invention will indicate effective senolysis. Determination of the beneficial effects of compounds of the invention on reducing the senescence burden in PCLS obtained from IPF patients will be performed as follows. In each experiment three dose levels (10 .mu.M, 1 .mu.M and 0.1 .mu.M) of a DMSO formulation of compounds of the invention will be tested, in addition to a vehicle control sample. PCLSs will be exposed to such compounds for three days, followed by a media wash-out and a three-day recovery period. Upon harvest, PCLSs will be collected for staining and analysis of p16/senescence and fibrosis, or flash frozen for RNA-seq/qPCR detection of relevant markers of senescence, fibrosis and epithelial regeneration. Supernatants will be harvested for studying changes in SASP factors through a Luminex.RTM. or MSD.RTM. analysis. Upon observing senolysis through p16 reduction, markers of fibrosis will be evaluated using established methodologies, including collagen level determination (picrosirius red staining) and gene expression changes of fibrosis markers, such as FN1, SERPINE1, COL1A1, CTGF, MMPI, and ACTA2, via qPCR.
Example 4
Effect of senolytic Agents In an In Vivo Pharmacodynamic Model
[0301] The ability of candidate agents to eliminate senescent mouse epithelial-enriched cells induced by locally administered bleomycin was measured directly in the following assay. On day 0, male C57BL/6 mice (The Jackson Laboratory) were administered 2.2 U/kg of bleomycin or its vehicle (PBS) by oral aspiration (50 mL). On day 11, each mouse received either vehicle (2.5% glycerin in PBS) or increasing concentrations of Compound 1 via oral aspiration (50 mL). On day 14, mice were euthanized, exsanguinated, and perfused PBS, 1 mL of dispase, and 0.2 mL of a 1% low melt agarose. Individual lobes of the lungs were collected for either single cell isolation, cell enrichment, and qPCR or for fixing, staining, and IHC analysis.
[0302] Left lung lobes were collected for epithelial cell enrichment as follows. Lung lobes were placed in 2 mL of dispase on a rocker for 45 minutes. To obtain isolated cells, a serological pipet was then used to dissociate the lung tissue prior to adding 10 mL of sort buffer containing 50U/ml of DNase. Next, samples were incubated on a rocker for 10 minutes at 37.degree. C. and then passed through both 100 mm and 70 mm cell strainers. The collected suspension was then spun at 550 g for 5 minutes at 4.degree. C. to pellet the cells. The supernatant was then removed, and 1 mL cold RBC lysis buffer was added at room temperature for 20 seconds. Next, 5 mL of sort buffer was added, and the sample was spun at 550 g for 5 minutes at 4.degree. C. The supernatant was then removed and 5 mL MACS buffer was added. The sample was next passed through a 40 mm cell strainer. Cells were counted using a cell counter then re-spun (550 g for 5 minutes) and suspended in MACS buffer containing 10% CD45+ microbeads at a volume of 100 uL per 1.times.10.sup.7 cells. Cells were mixed and incubated for 15 minutes at 4.degree. C. Next, 2 mL of MACS buffer was added and then the cells spun at 350 g for 5 minutes at 4.degree. C. The supernatant was removed, cells were resuspended in 500 uL MACS buffer and added to a MACS separator cell column. The column was washed 4 times with 500 uL MACS buffer and the effluent (CD45-) collected. Cells were then counted with a cell counter. The sample was then spun at 350 g for 5 minutes at 4.degree. C. The supernatant was removed, and the cells resuspended in MACS buffer containing 10% CD326+microbeads at a volume of 100 uL per 1.times.10.sup.7 cells. Cells were mixed and incubated for 15 minutes at 4.degree. C. Next, 2 mL of MACS buffer was added and then the cells spun at 350 g for 5 minutes at 4.degree. C. The supernatant was removed, cells were resuspended in 500 uL MACS buffer and added to a MACS separator cell column. The column was washed 4 times with 500 uL MACS buffer and the effluent (EpCAM-) collected. The column was then removed from the magnet and placed on a new collection tube. Finally, 3 mL of MACS buffer was added to the column through to collect remaining cells (CD45-, EpCAM+). The cells were then spun down (550 g for 5 minutes) and resuspended in 500 uL TRIzol.RTM.(Thermo Fisher Scientific) for storage at -80.degree. C.
[0303] Quantification of p16 mRNA by qPCR was performed as follows. Desired frozen cells (CD45-, EpCAM+) were thawed, 20% chloroform added, and sample vortexed until milky in appearance. Cells were then spun at 15,000 g for 15 minutes. The supernatant was removed, collected into a new tube, diluted with equal volume 100% ethanol, and added to the Zymo column (Zymo Research Direct-zol RNA MicroPrep kit; Cat #R2063). Column was spun at 15,000 g for 1 minute. Next, 400 uL wash buffer was added to each sample and respun at 15,000 g for 1 minute. Next, 40 uL DNase reaction mix (5 uL DNase I+35uL DNA digestion buffer) was added to the column matrix and incubated for 15 minutes at room temperature. Next, 400 uL pre-wash buffer was added to each sample and respun at 15,000 g for 1 minute. This step was repeated and then the cell pellet was resuspended in 700 uL wash buffer followed by another spin. The flow-through was discarded and the column was respun at 15,000 g for 2 minutes. Next, the column was transferred to a Rnase-free tube and 20 uL of DNase/RNase free water was added to the column. The column was then respun at 15,000g for 1 minute and samples were analyzed for RNA quality on the Agilent TapeStation.RTM. 2200 (Agilent Technologies.RTM.).
[0304] The extracted RNA was then used to generate single-stranded cDNA using the kit and protocol available from Applied Biosystems.RTM. (High Capacity cDNA Reverse Transcription kit; Cat #4368813). Finally, Real-time PCR was performed using the QuantStudio.RTM. 7 PCR System with Taqman.RTM. gene-specific primers for p16.sup.INK4a. All signals were normalized to .beta.-actin. Relative gene expression was calculated by the .DELTA..DELTA.Ct method where the ACt was calculated using the .beta.-actin reference gene. AACt was calculated relative to the PBS/Vehicle control group.
[0305] FIG. 7 shows the results. The concentration-response curve for Compound 1, demonstrates sensitivity of mouse lung epithelial cell p16 mRNA induced by bleomycin to a senolytic molecule following local (OA) administration. These data show that senolytic agents are capable of selectively eliminating senescent lung airway epithelial cells in vivo.
[0306] Right lung lobes were collected for p16 immunohistochemistry analysis was performed as follows. Lung lobes were fixed in 4% paraformaldehyde overnight, dehydrated in increasing concentrations of ethanol, and embedded in paraffin. Five micron sections were cut from the paraffin block and collected on glass slides. Slides were dewaxed using BOND Dewaxing Solution at 70.degree. C. for 30 seconds and washed with TBST 3 times. Antigen retrieval was done using BOND ER2 solution (EDTA buffer pH 9.0) for 20 minutes at 95.degree. C. and then 3% H.sub.2O.sub.2 was added for 10 minutes at room temperature followed by 3 successive washes. Slides were then incubated with SEA BLOCK.RTM. blocking buffer (Thermo Fisher Scientific Cat #37527) for 30 minutes at room temperature and washed with TBST 3 times. Slides were then incubated in mouse IgG block for 20 minutes and then incubated in primary p16 antibody (BD Biosciences Cat #550834) at a 1:50 dilution in TBST for 1 hour. Next, slides were washed with TBST 3 times and incubated with goat anti-mouse poly-HRP secondary antibody (Thermo Fisher Scientific Cat #B40961) for 30 minutes at room temperature. Next, slides were washed with TBST 3 times and a DAB substrate solution was added and incubated for 10 min at room temperature. Next, slides were washed with dH.sub.2O 3 times and then placed in hematoxylin for 1 minute. Slides were then washed thoroughly in dH.sub.2O for several exchanges, placed in a bluing reagent for 30 seconds, and then rinsed in tap water for 5 minutes. Finally, slides were mounted using water-based mounting medium and left to dry overnight. Quantification was performed with the number of p16+ foci determined in reference to the total number of cells.
[0307] FIG. 8 demonstrates sensitivity of mouse lung epithelial cell p16+ cells, induced to senesce by bleomycin, to Compound 1 following local (OA) administration. These data show that compounds of the invention are capable of selectively eliminating senescent lung airway epithelial cells in vivo.
Example 5
Synthesis
[0308] Compounds of this invention were prepared by using or adapting the synthetic scheme shown in FIG. 1.
[0309] The several hypotheses presented in this disclosure provide a premise by way of which the reader may understand the invention. This premise is provided for the enrichment and appreciation of the reader. Practice of the invention does not require detailed understanding or application of the hypothesis. Except where stated otherwise, features of the hypothesis presented in this disclosure do not limit application or practice of the claimed invention. For example, except where the elimination of senescent cells is explicitly required, the compounds of this invention may be used for treating the conditions described regardless of their effect on senescent cells. Although many of the pulmonary diseases and conditions referred to in this disclosure occur predominantly in older patients, the invention may be practiced on patients of any age having the condition indicated, unless otherwise explicitly indicated or required.
[0310] While the invention has been described with reference to the specific examples and illustrations, changes can be made and equivalents can be substituted to adapt to a particular context or intended use as a matter of routine development and optimization and within the purview of one of ordinary skill in the art, thereby achieving benefits of the invention without departing from the scope of what is claimed.
* * * * *
References














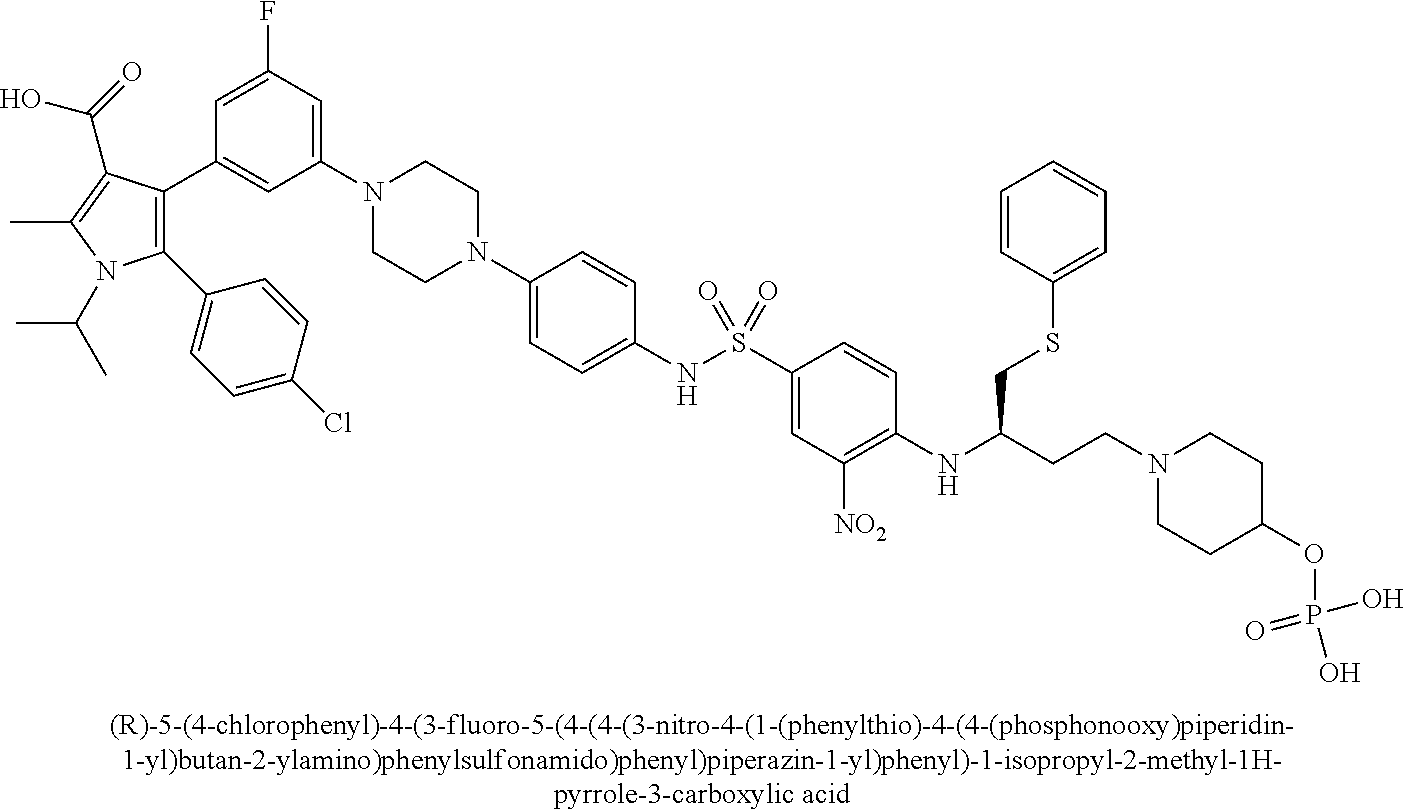
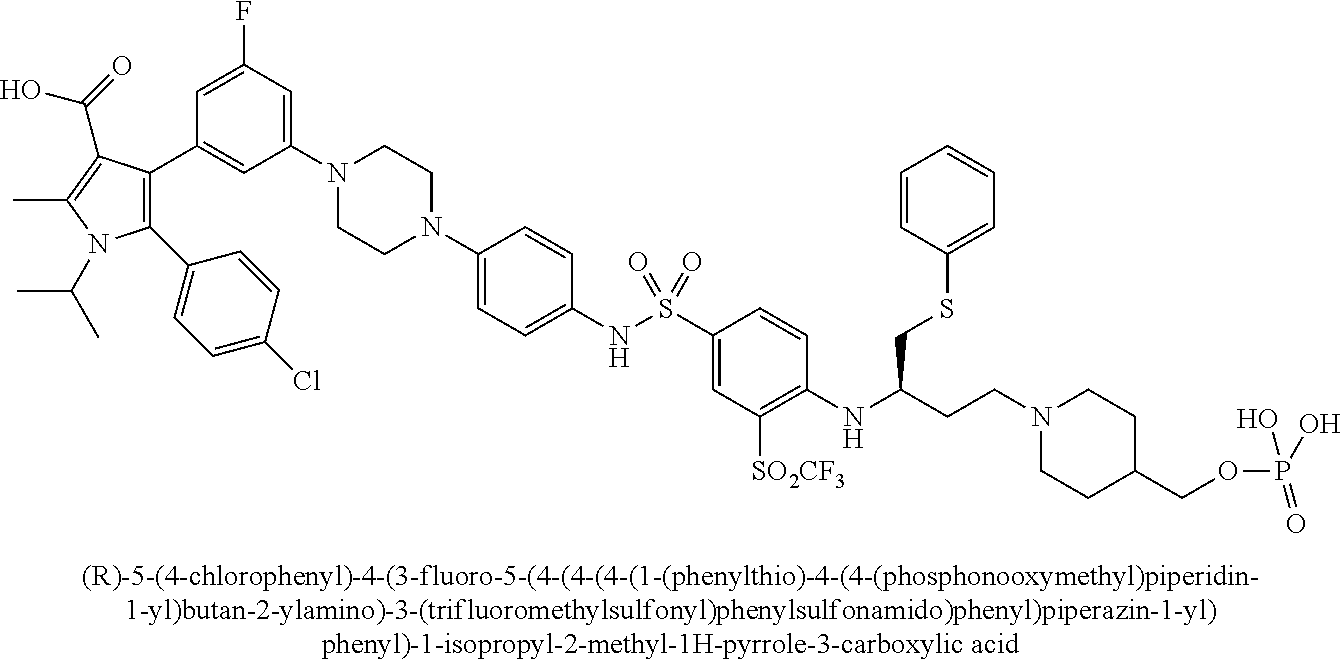
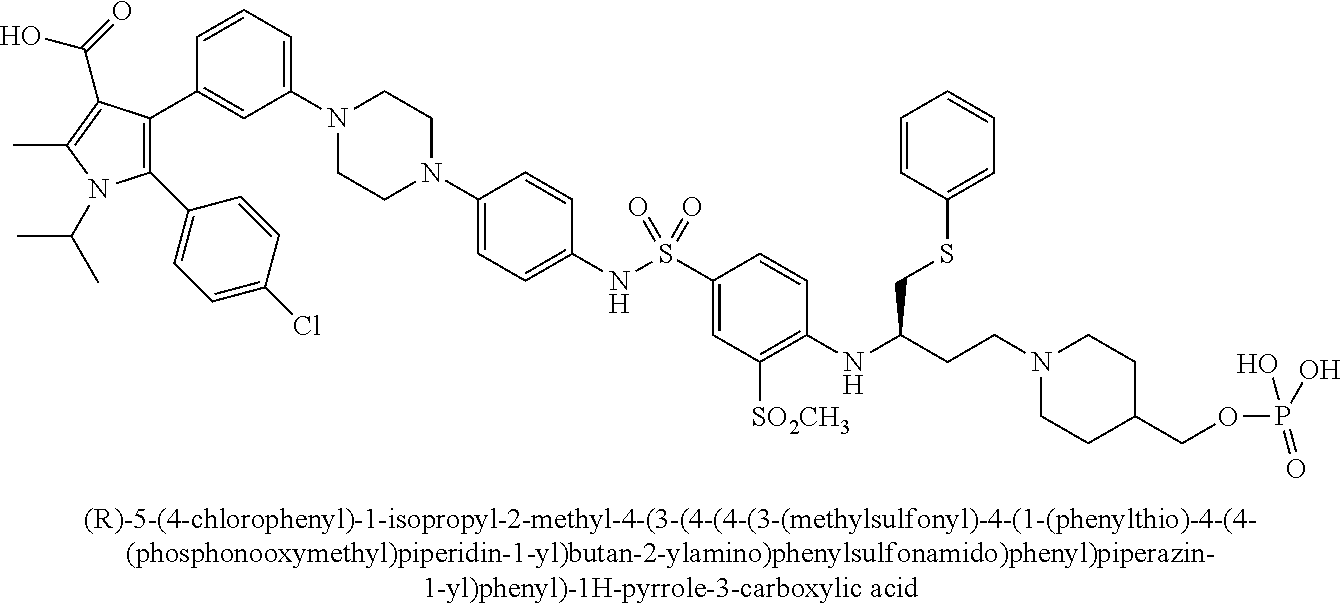
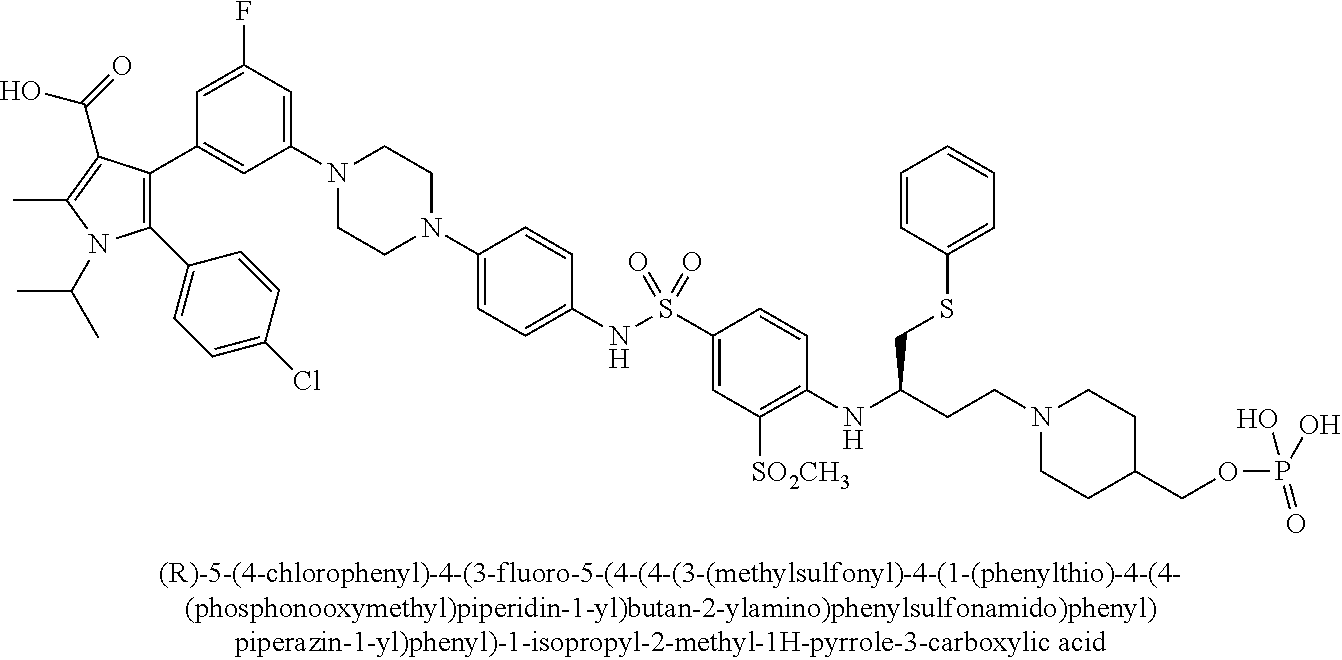
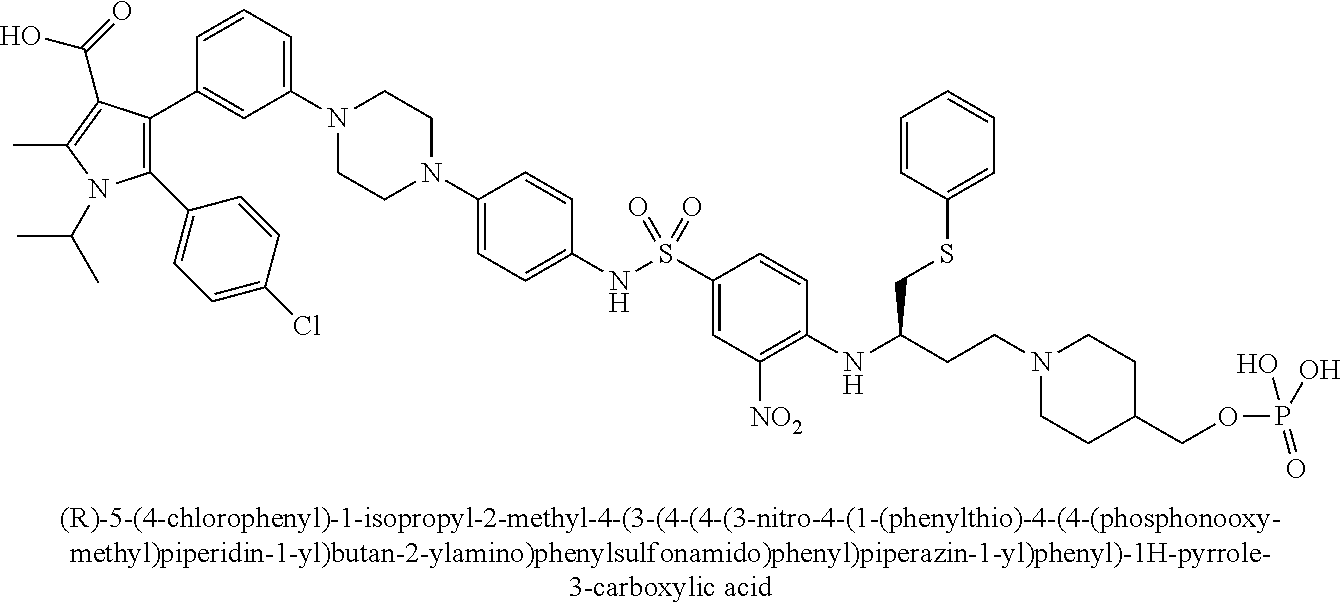
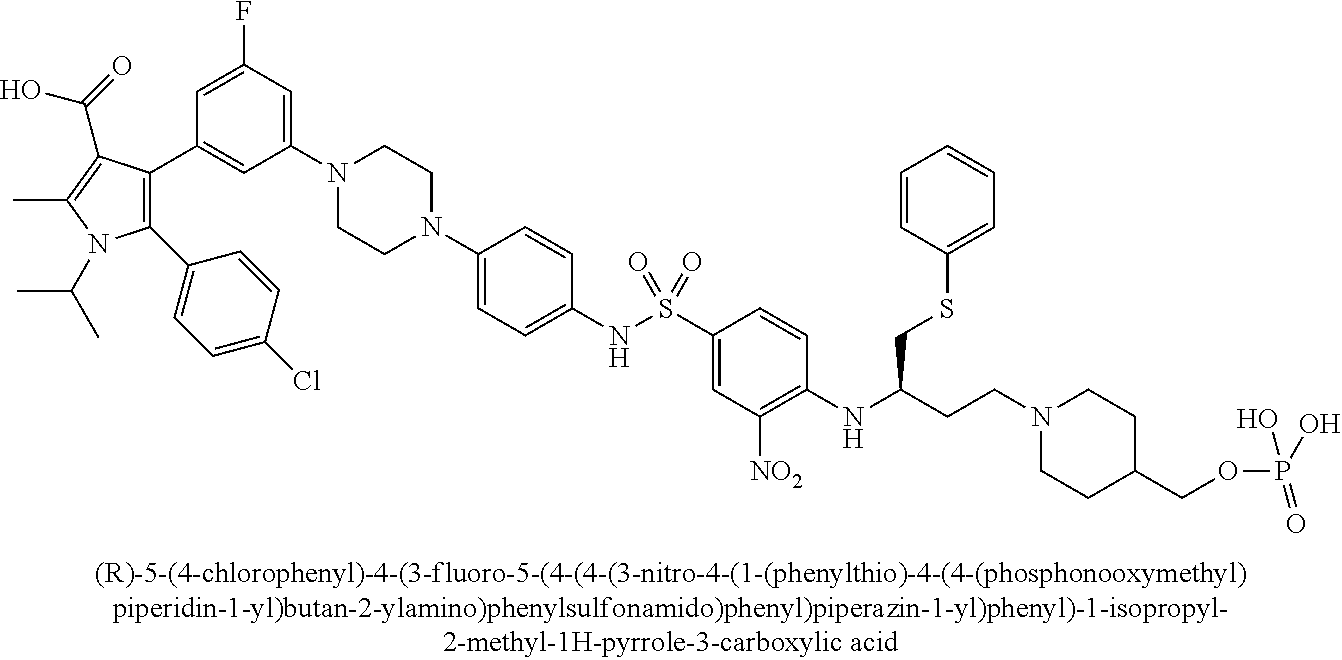
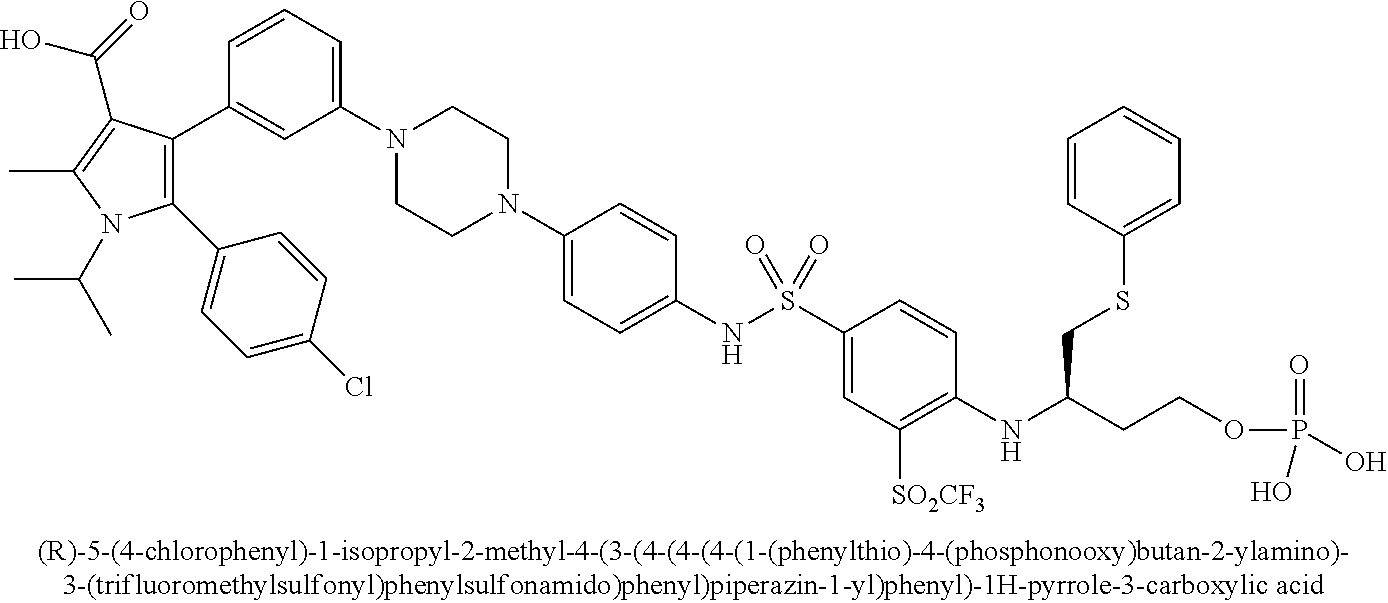
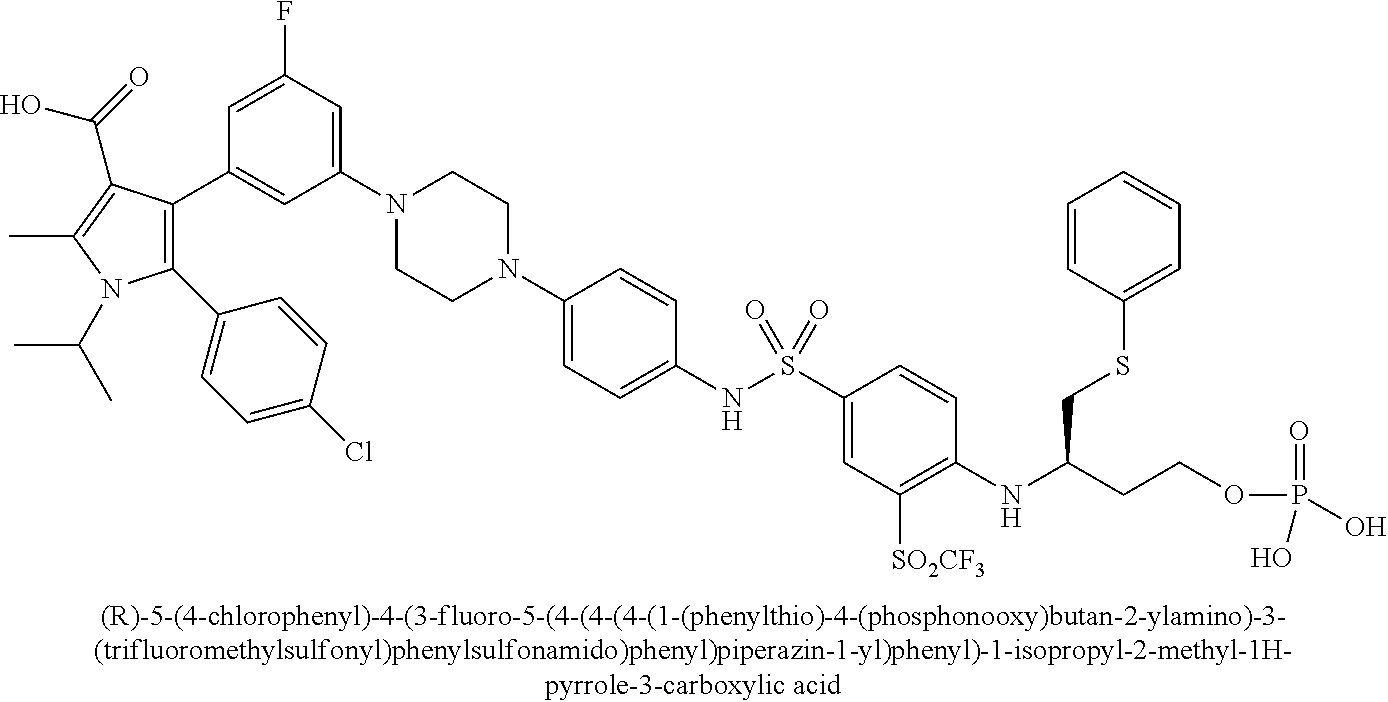
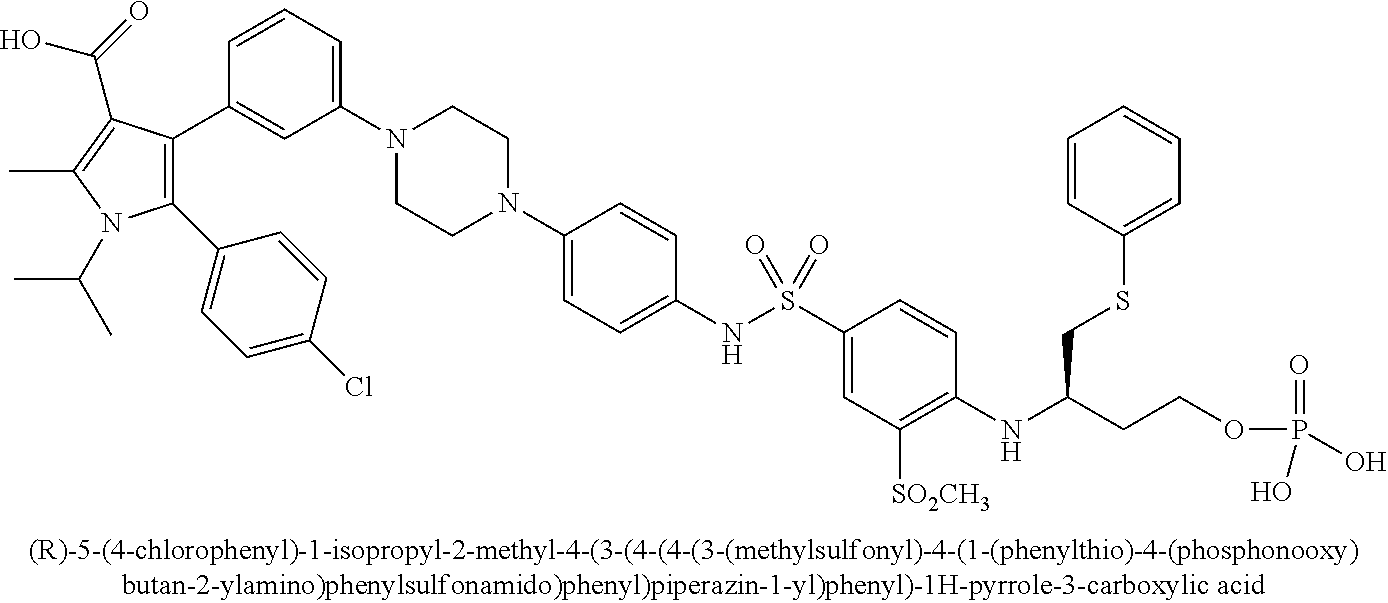
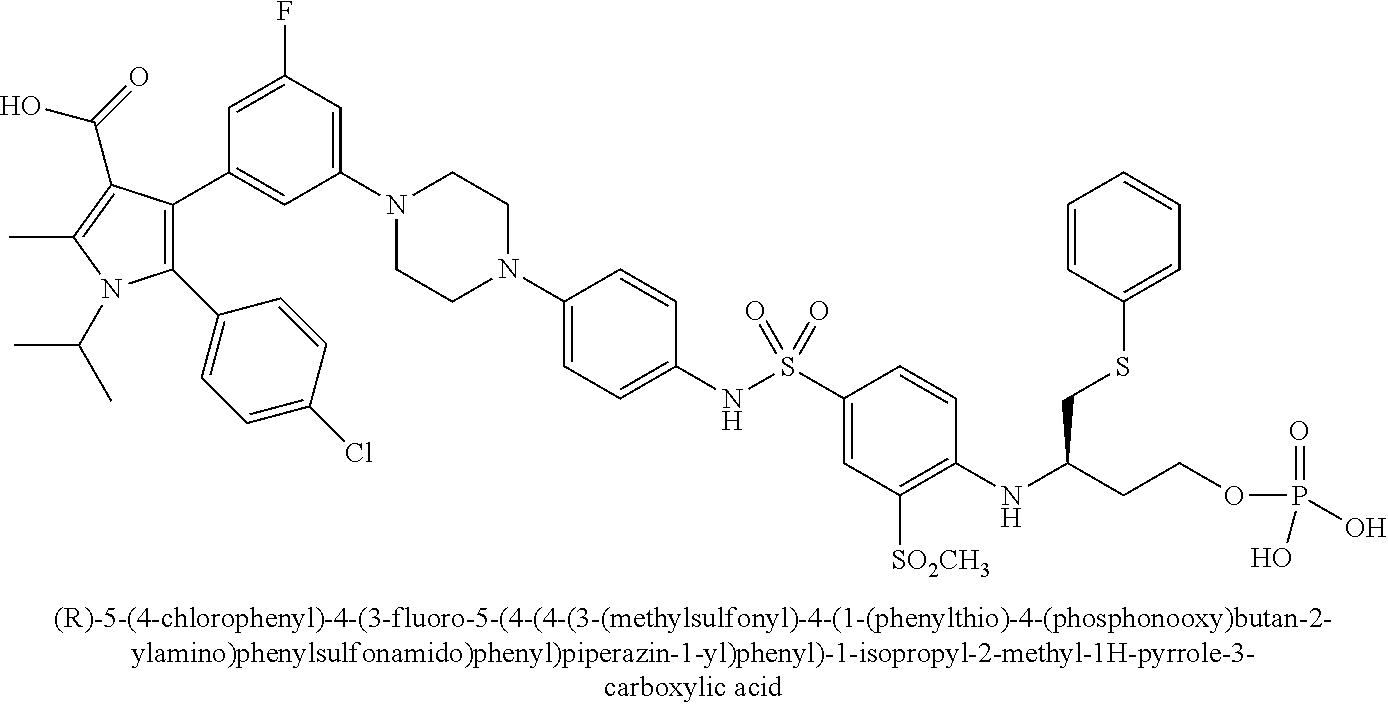

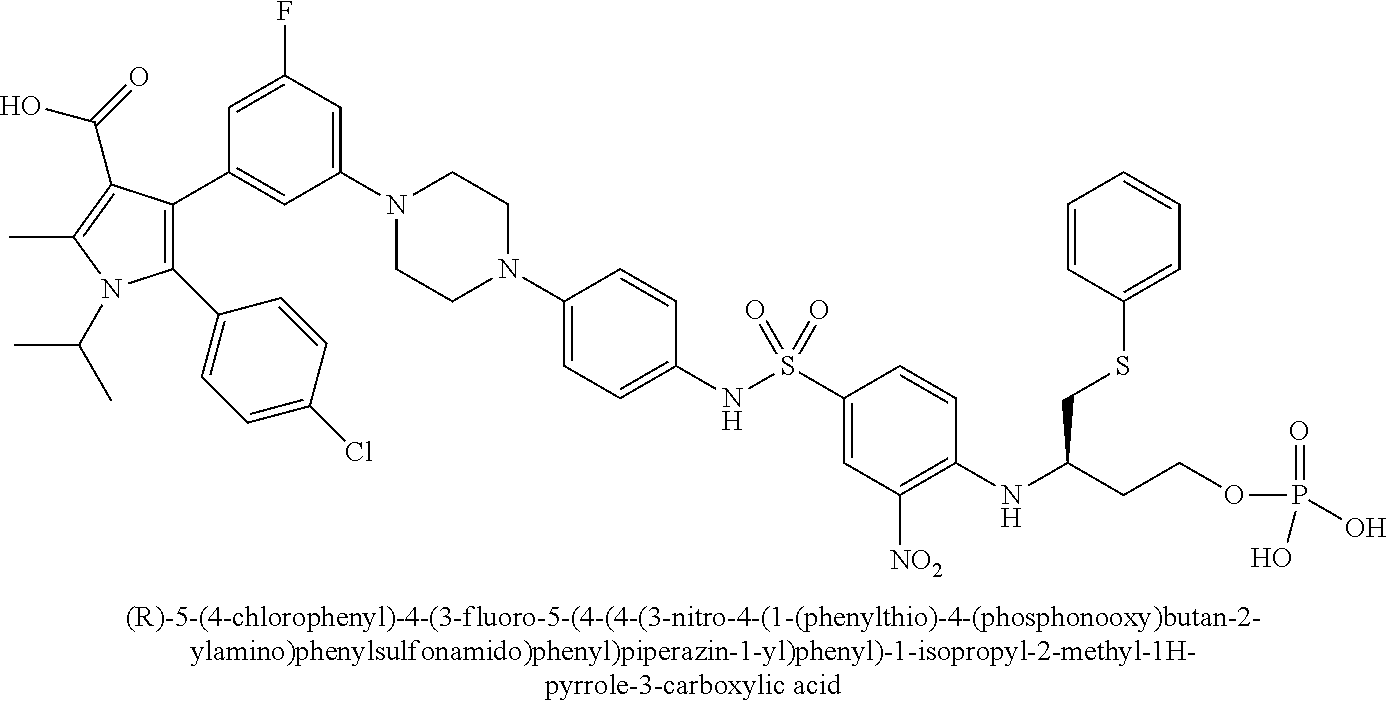
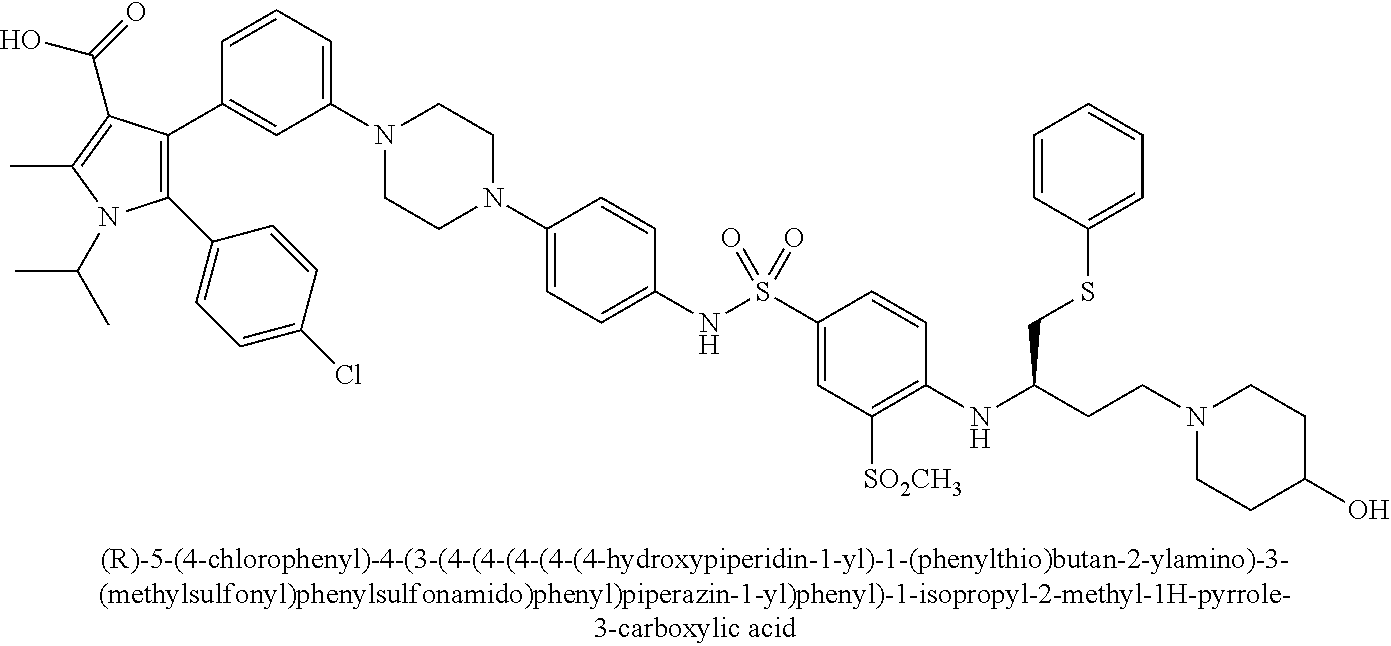

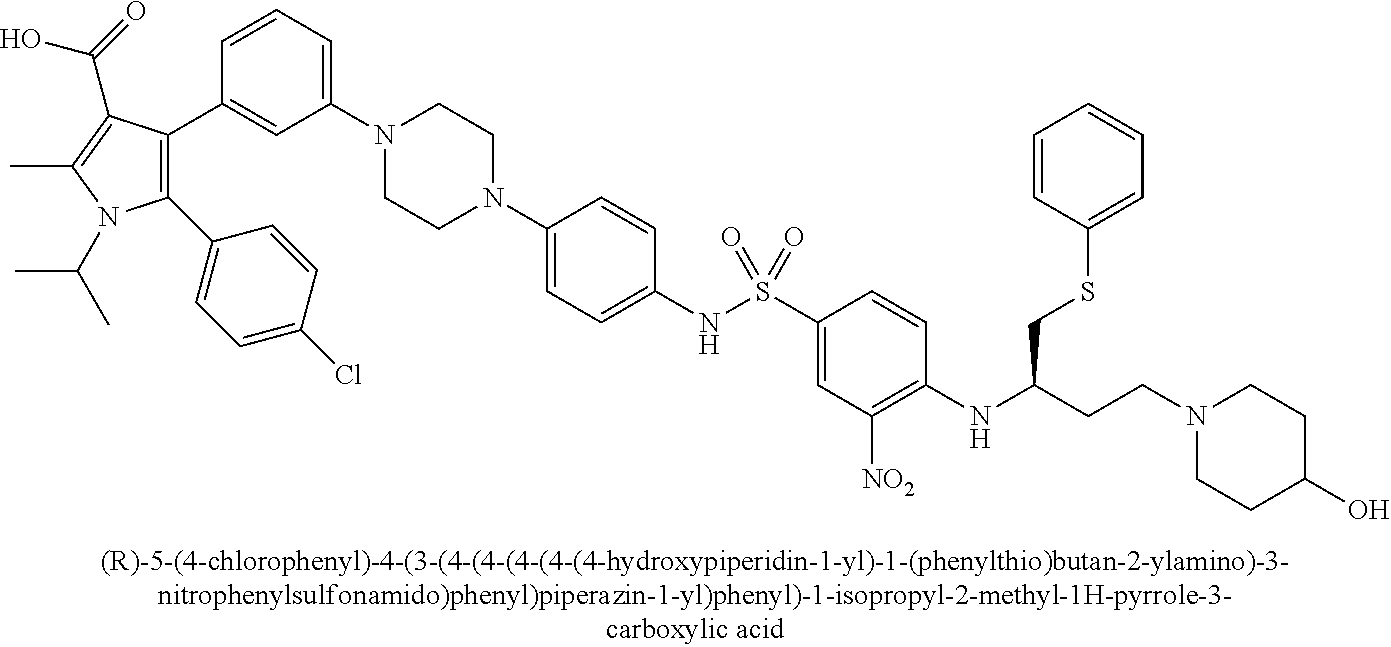
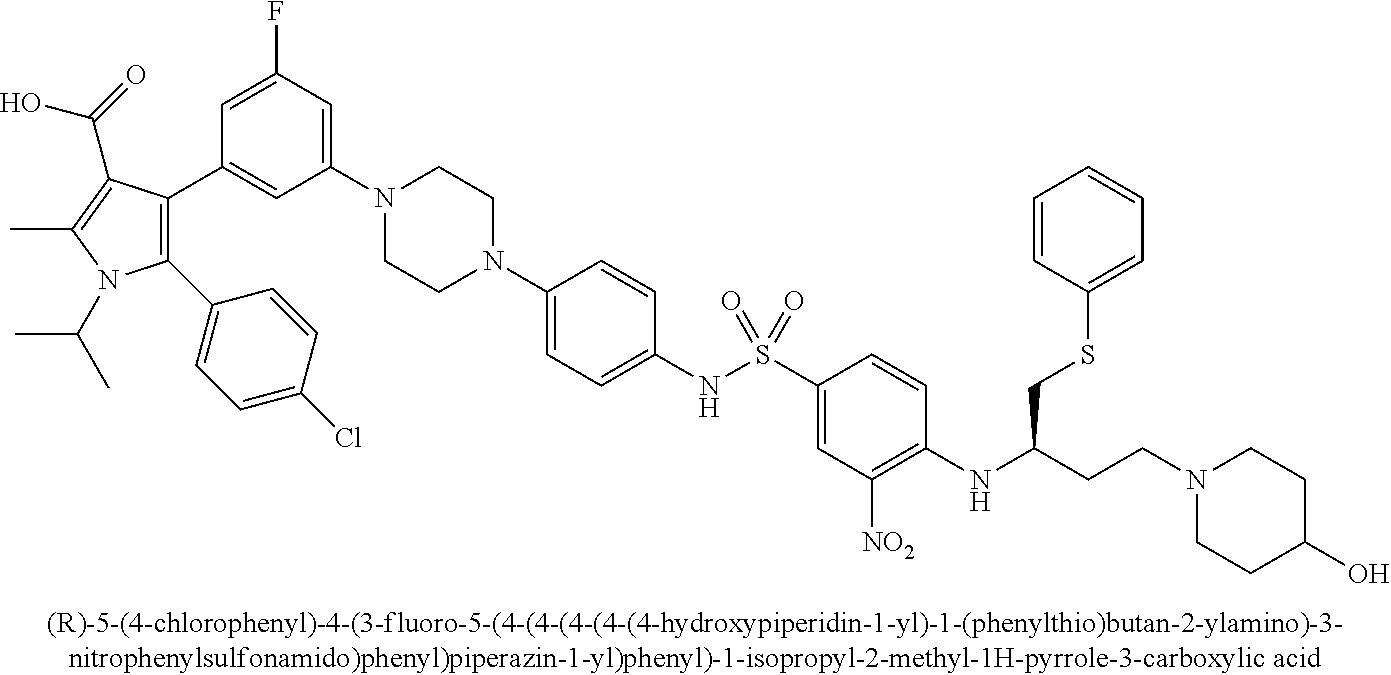
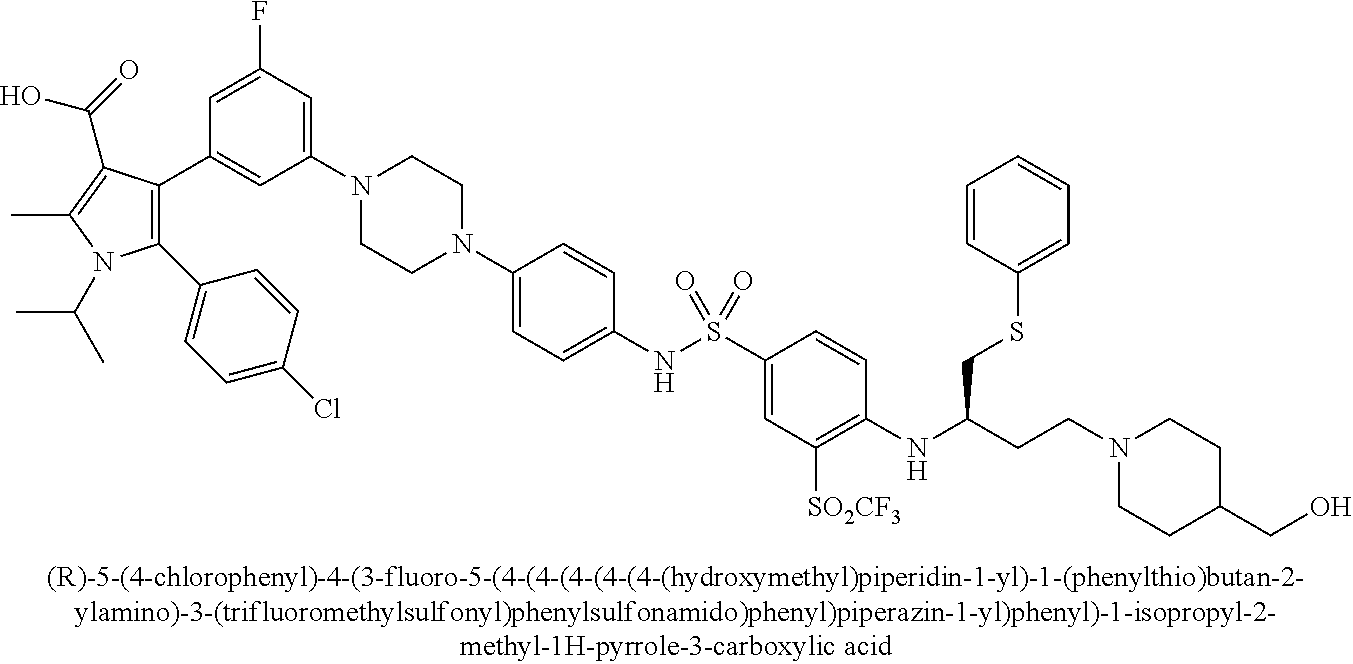
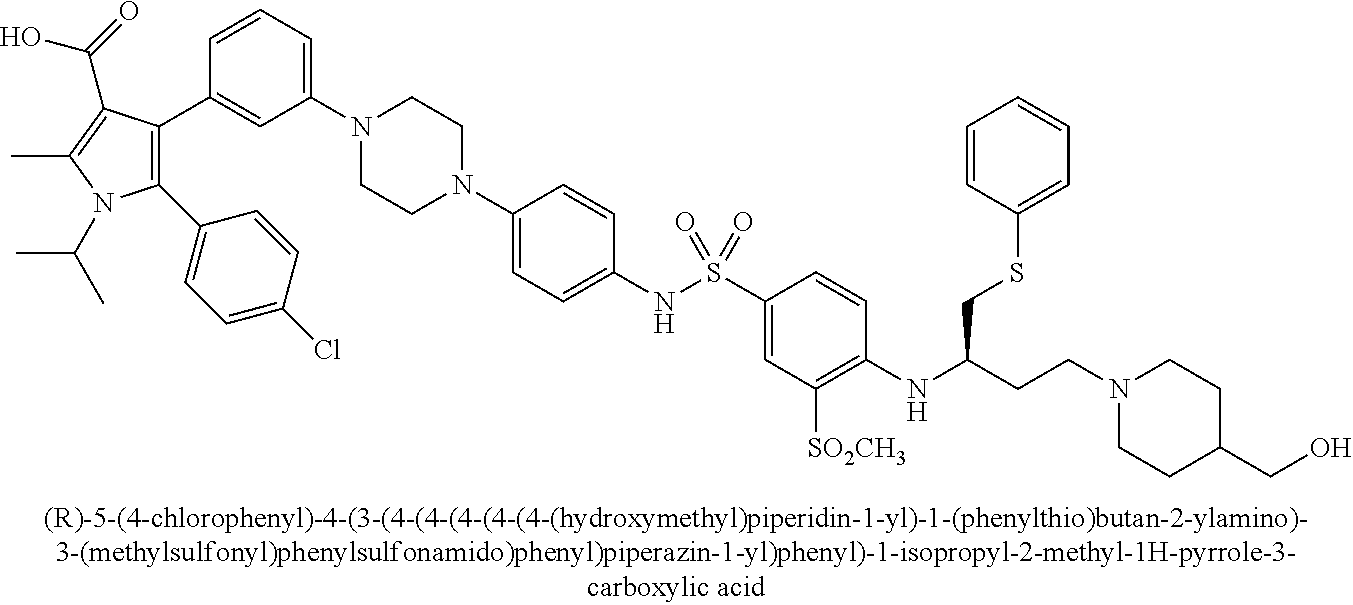
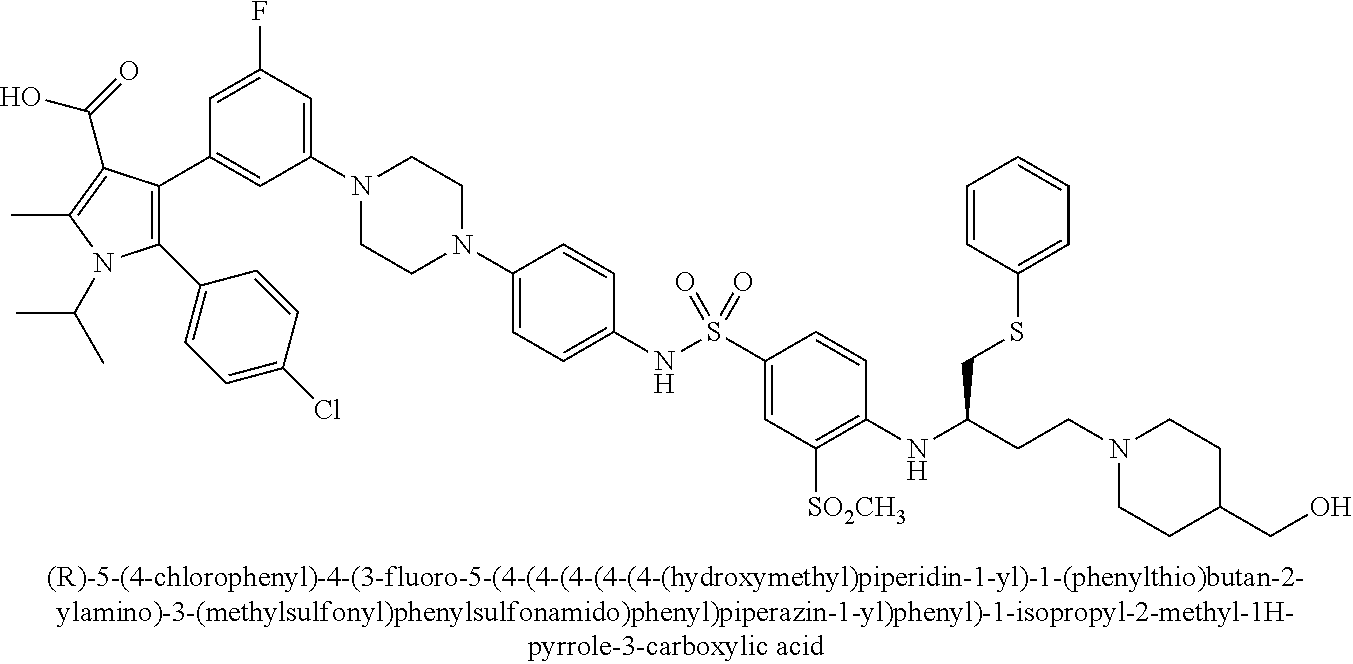
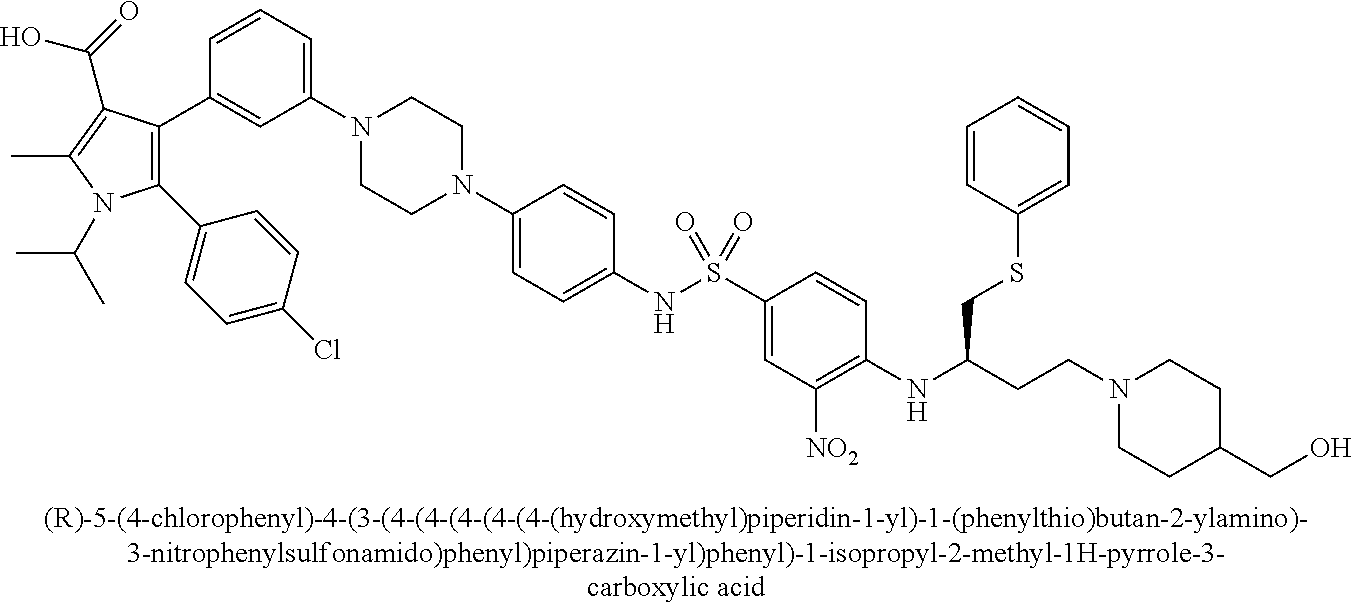


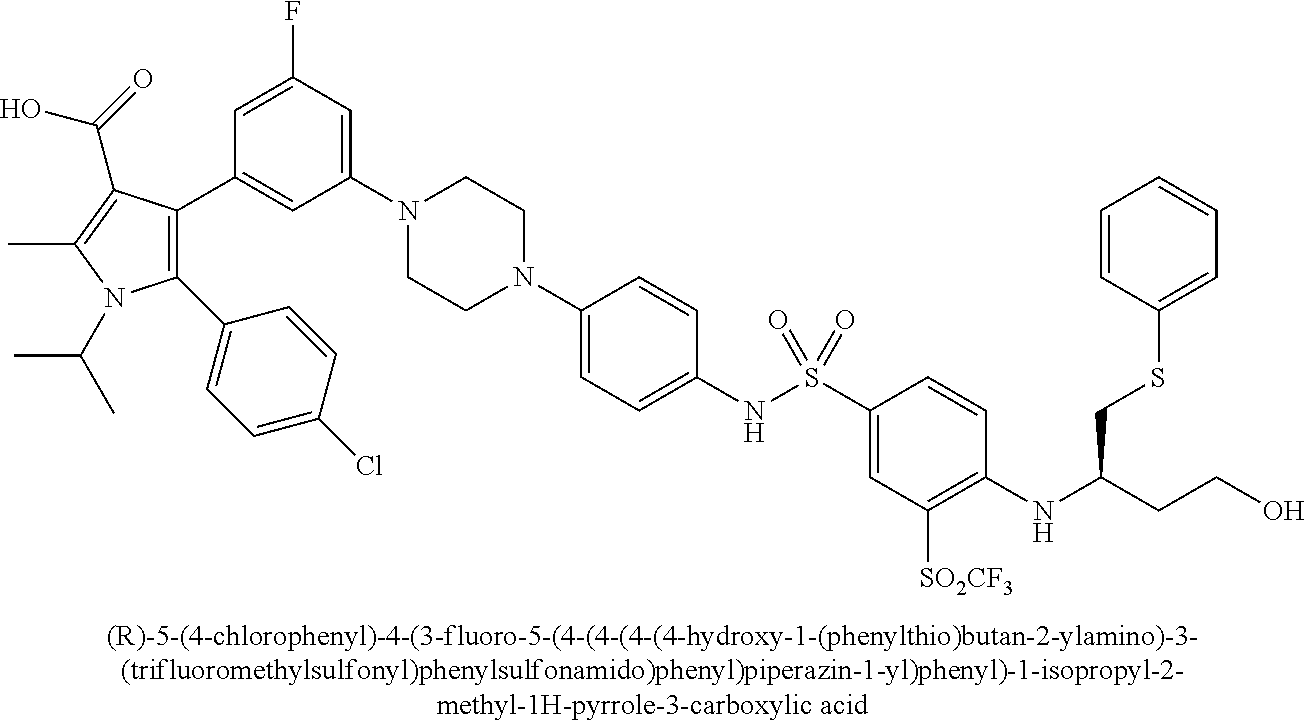


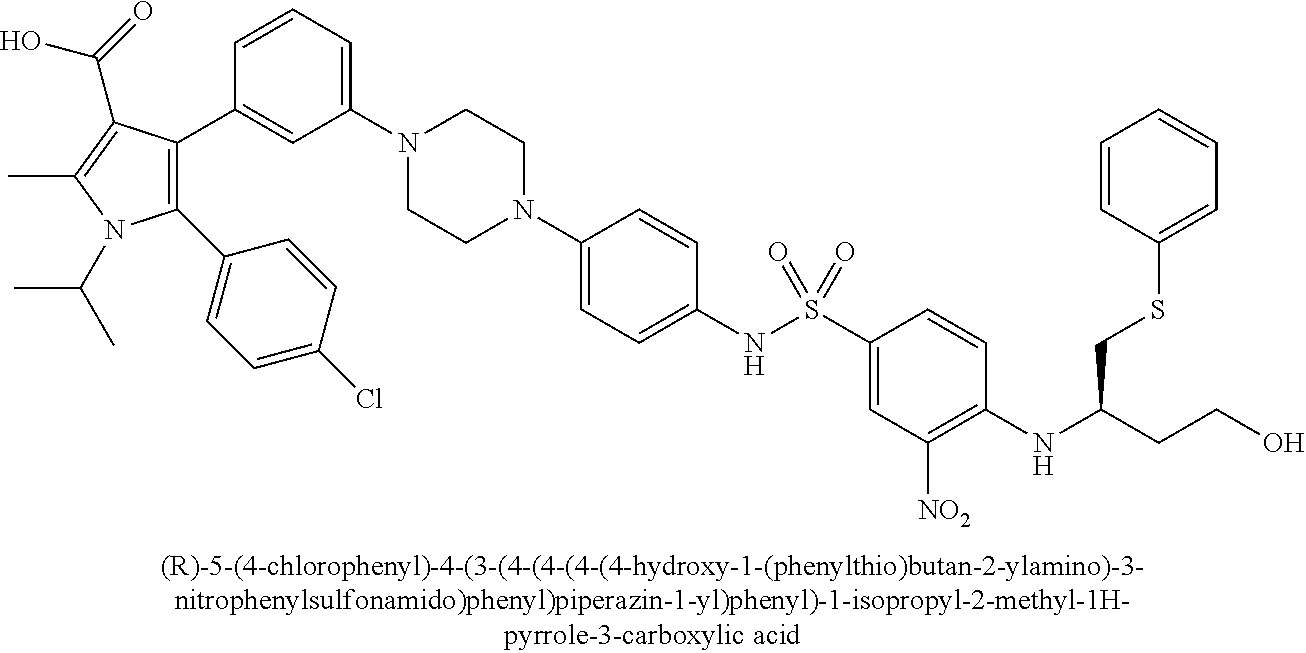

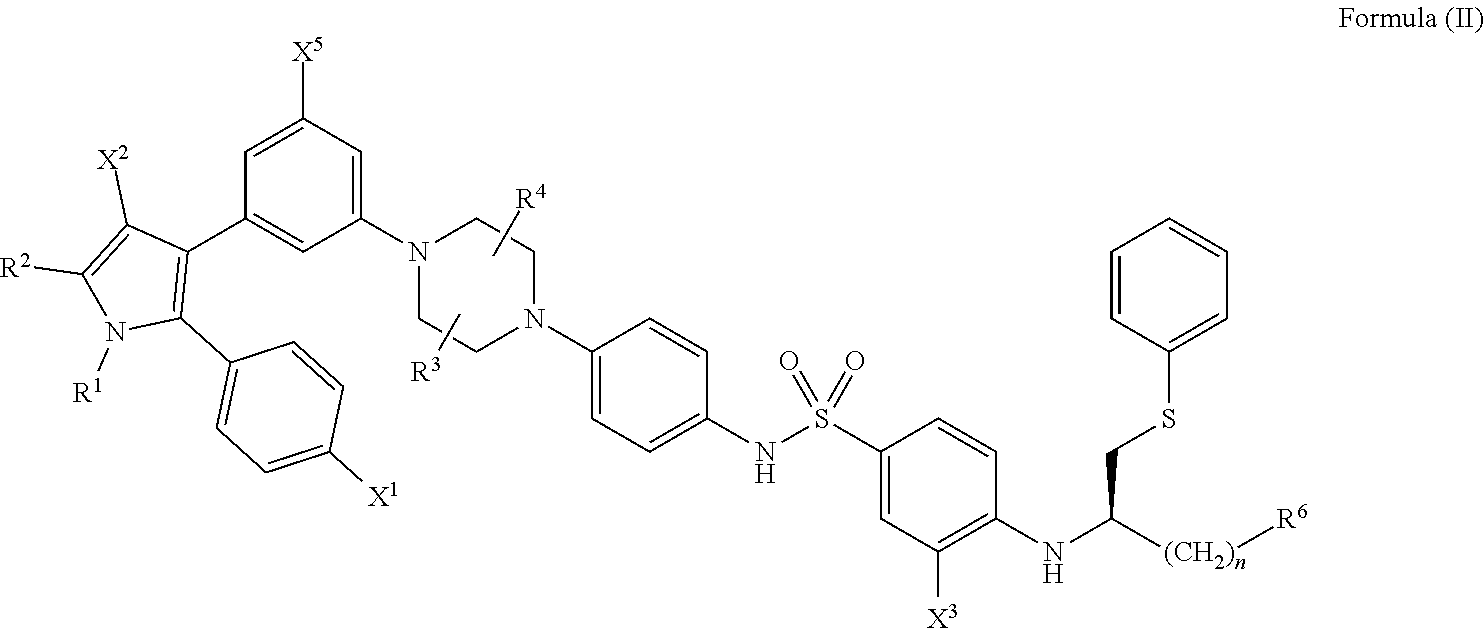
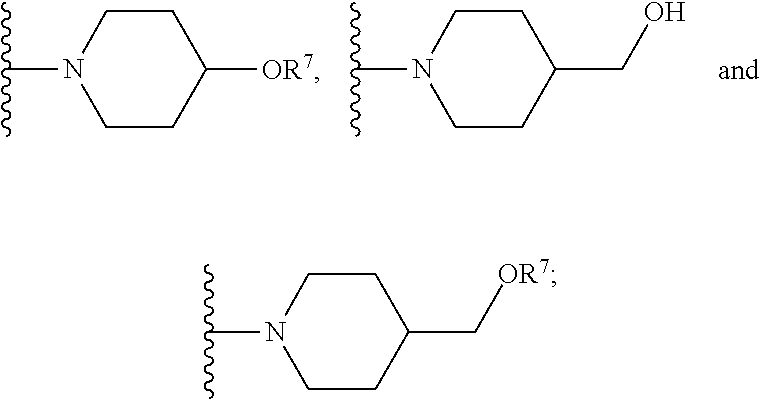
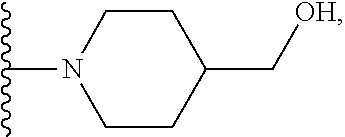
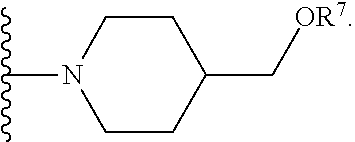




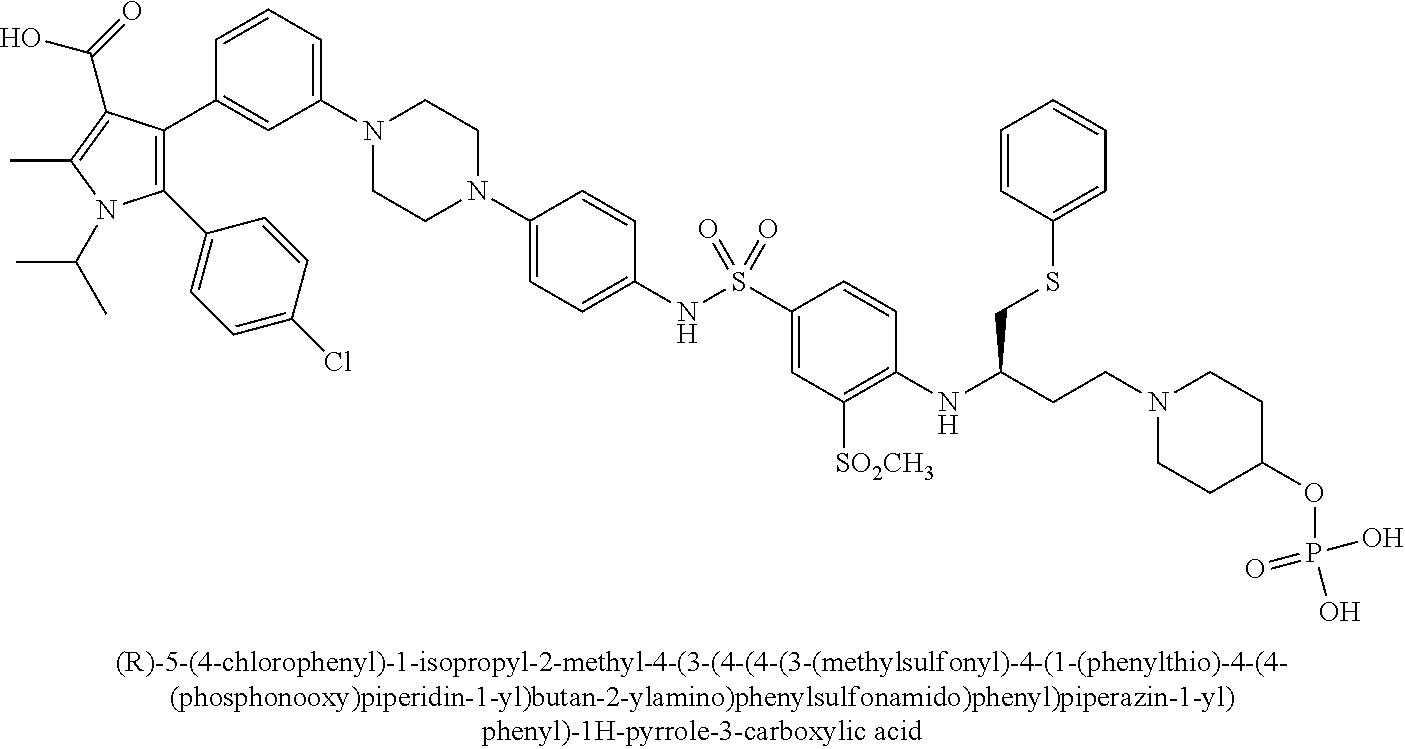
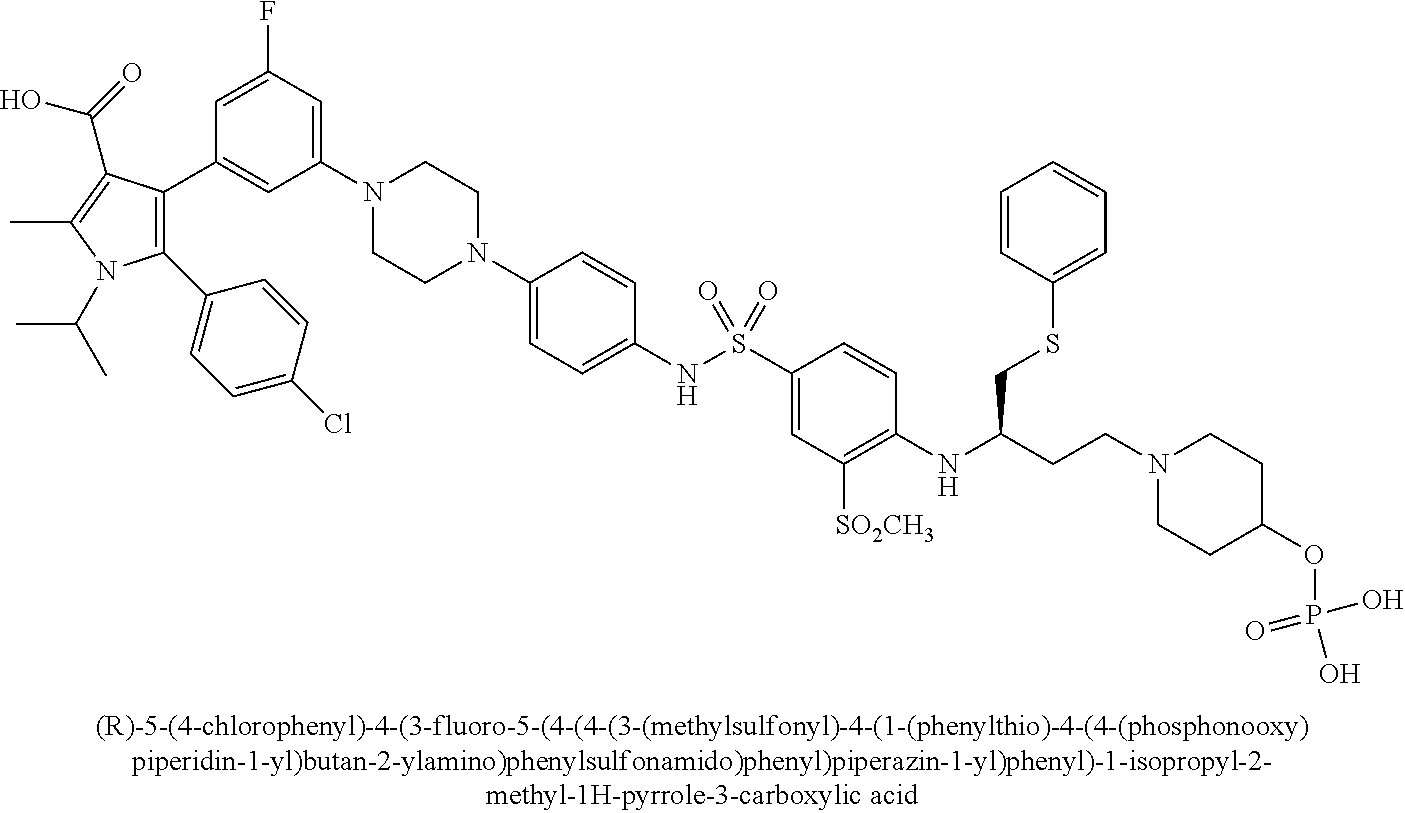
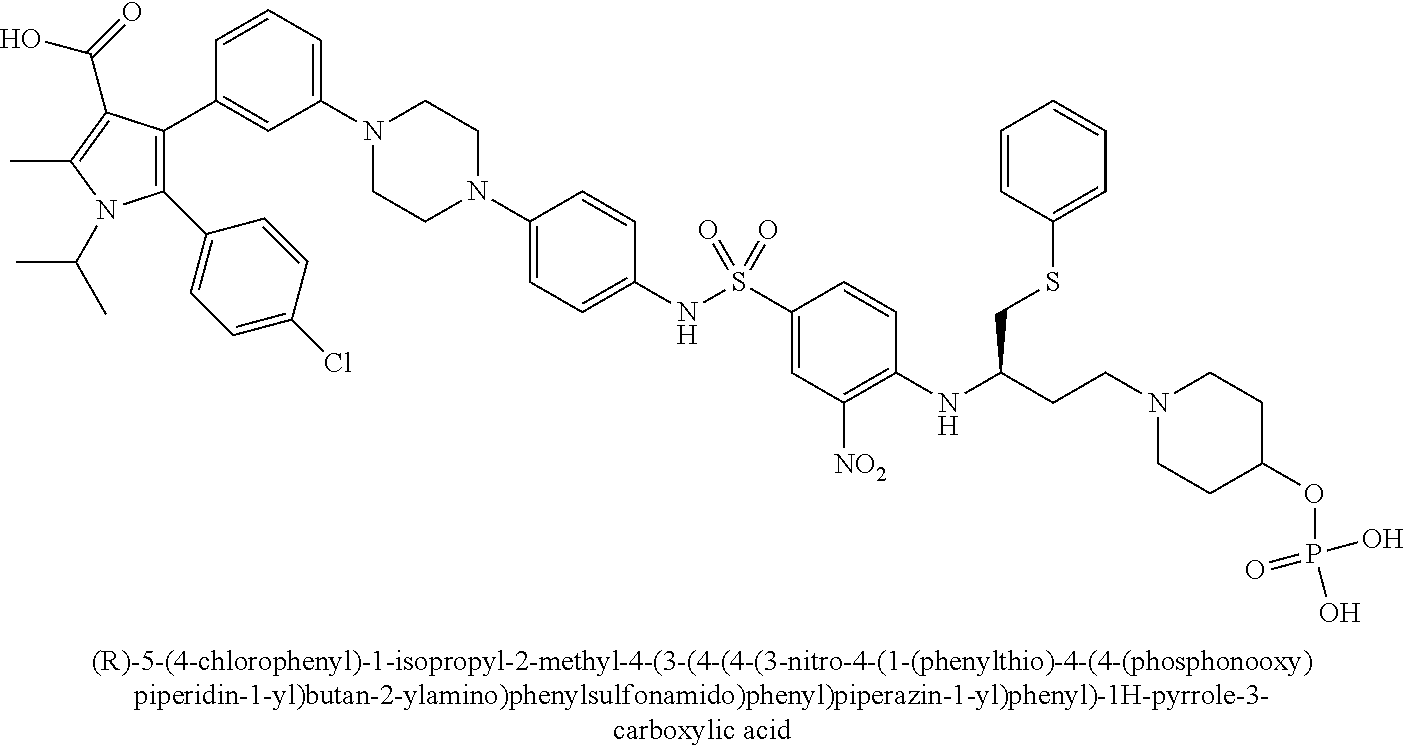




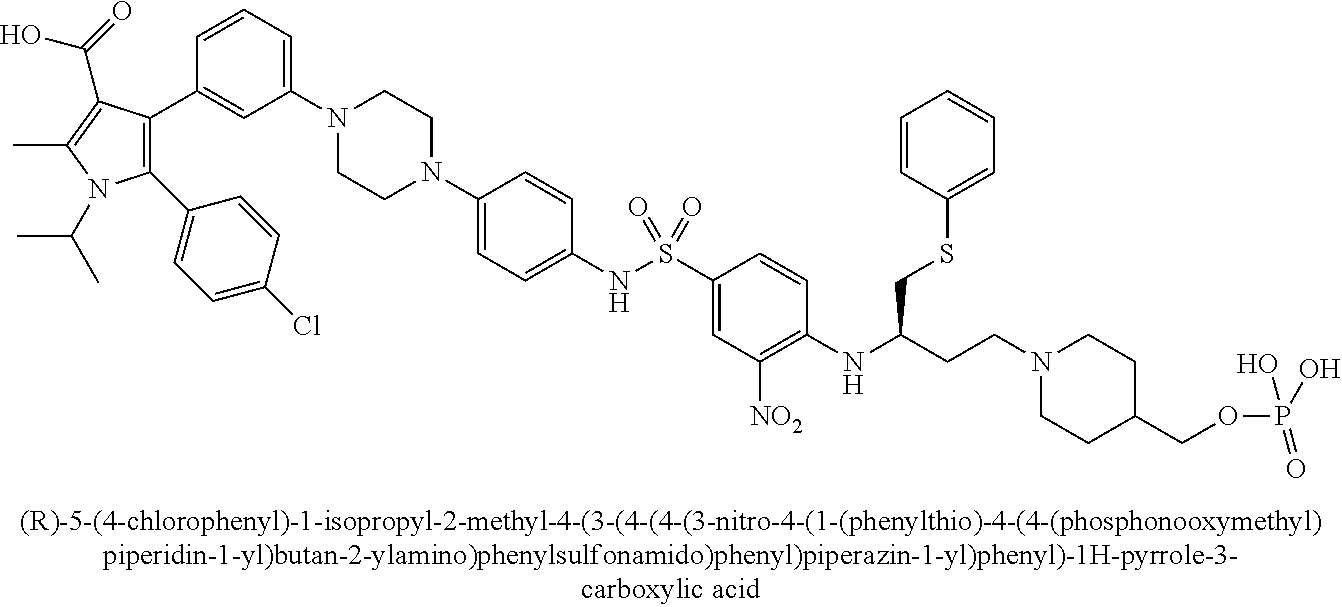

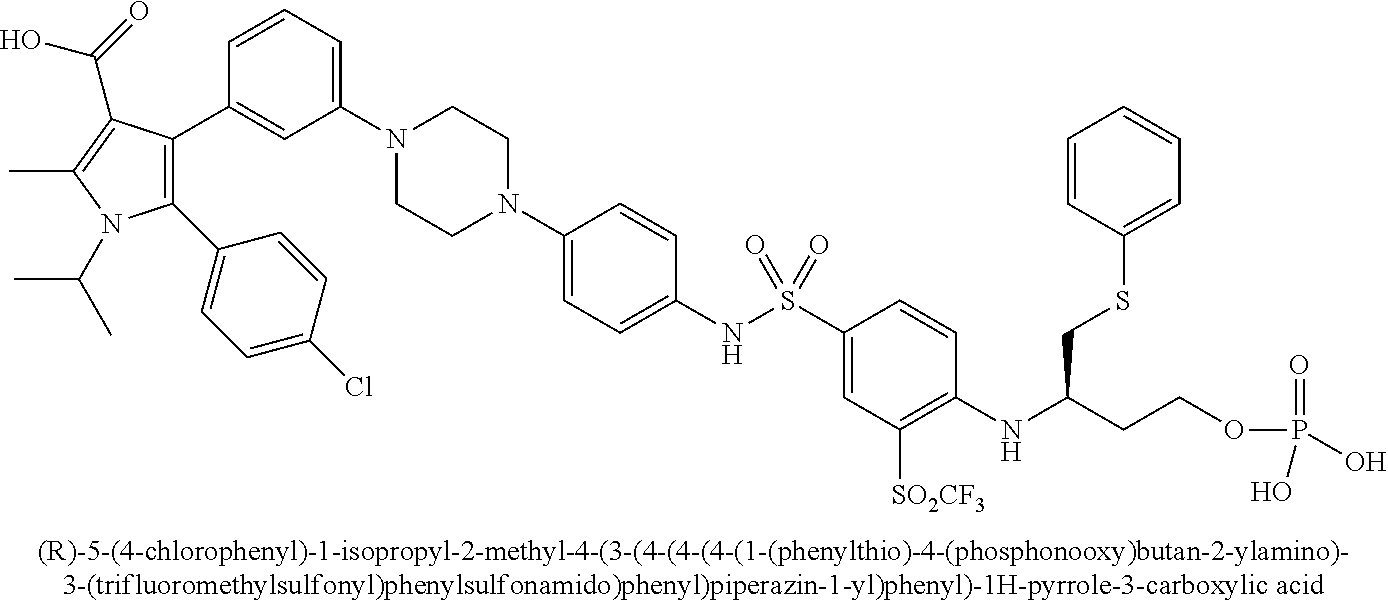
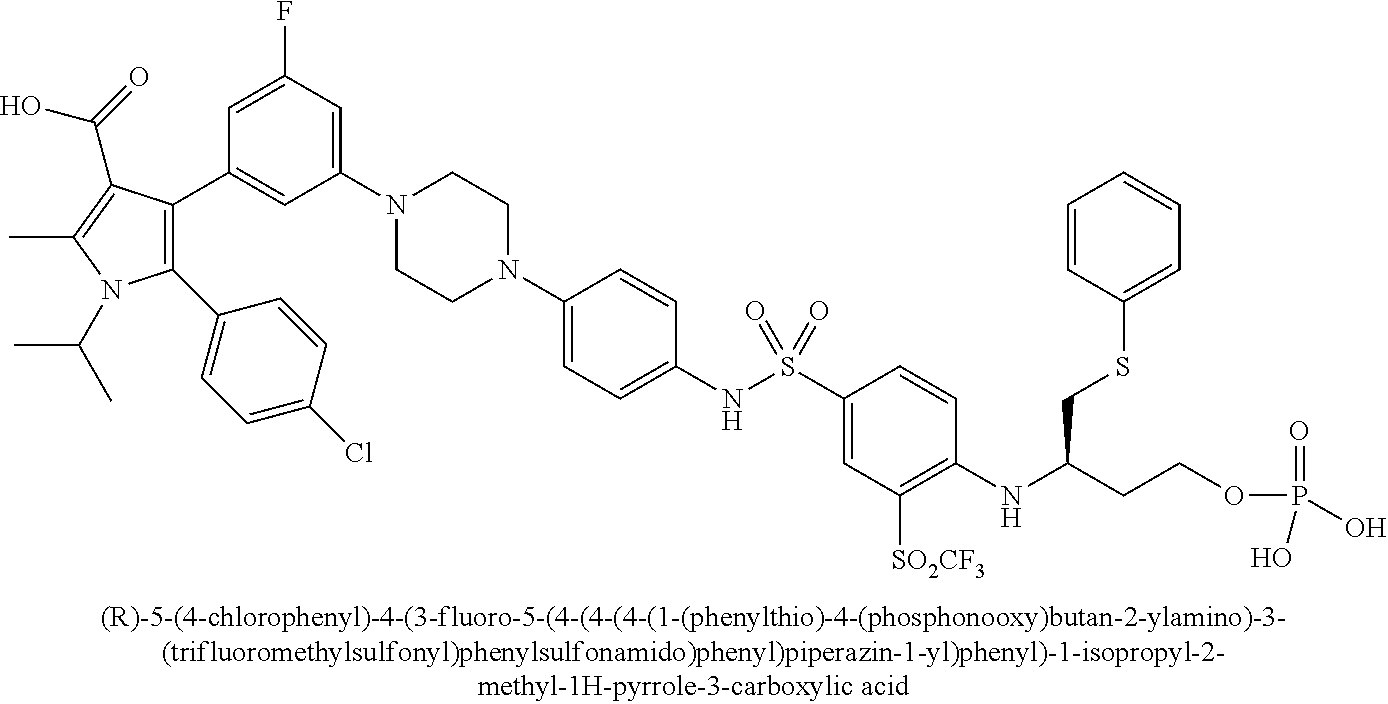
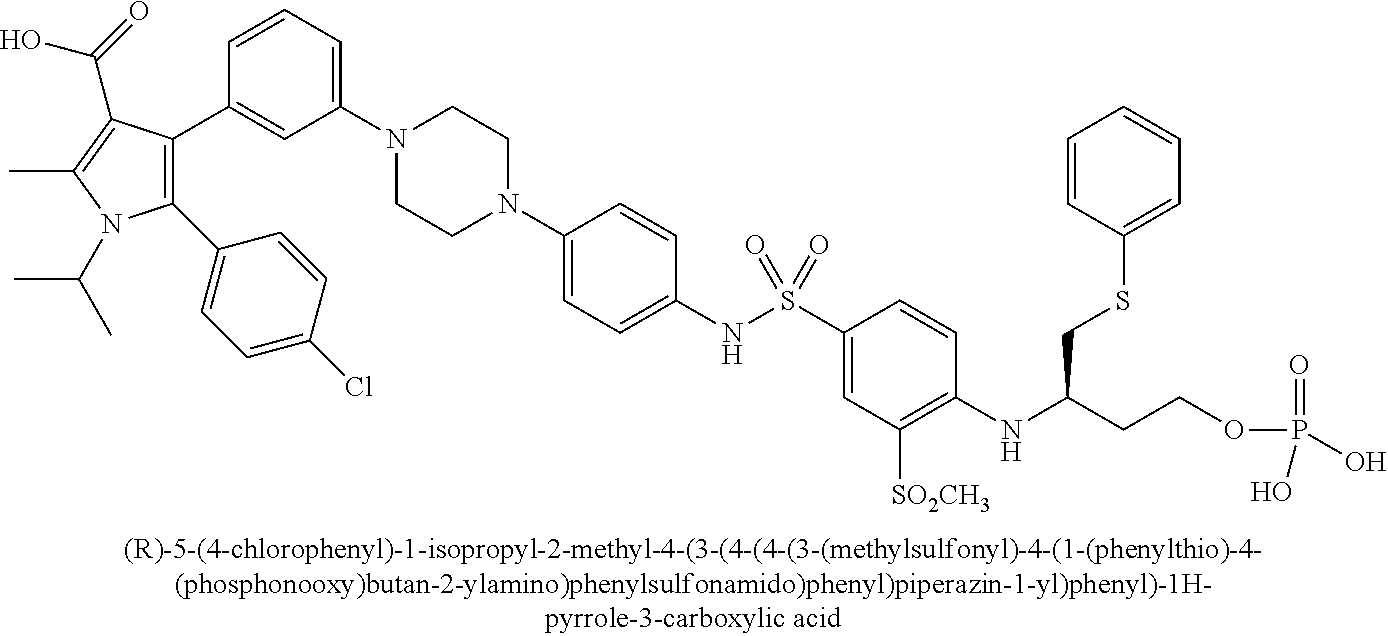
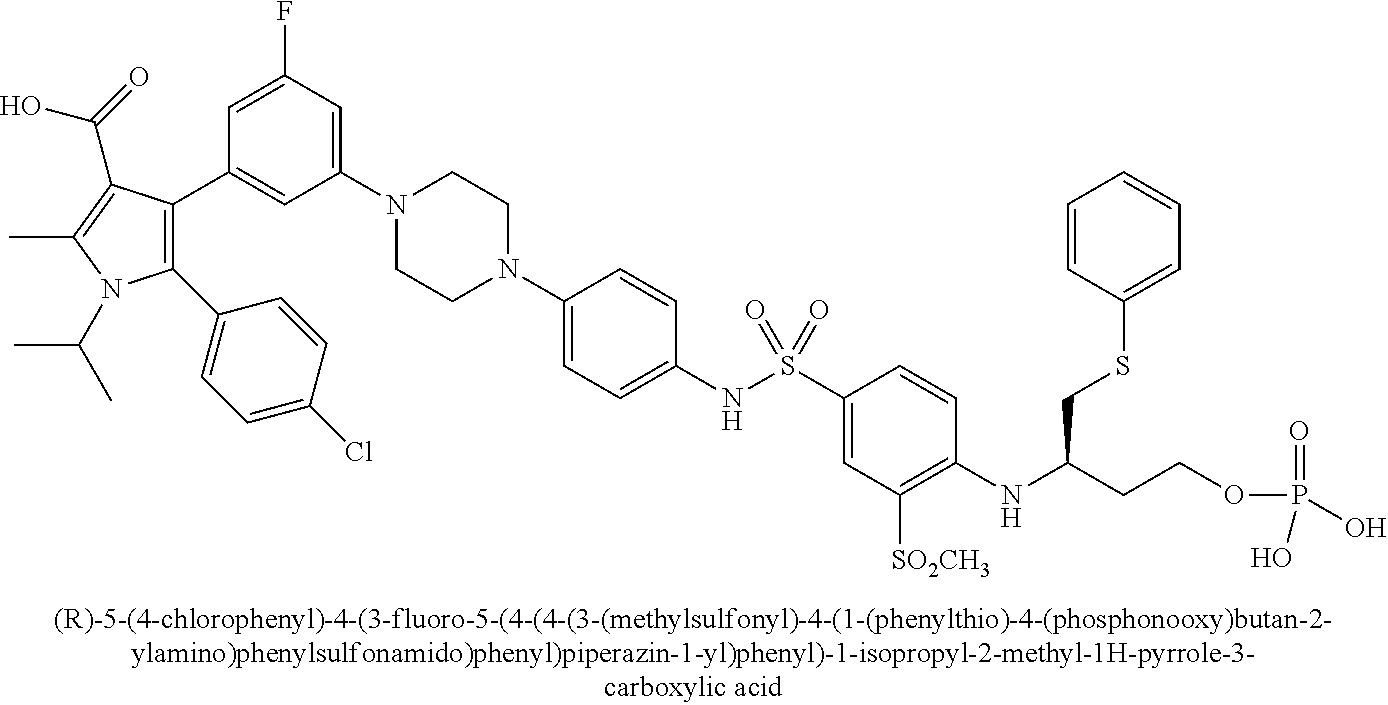

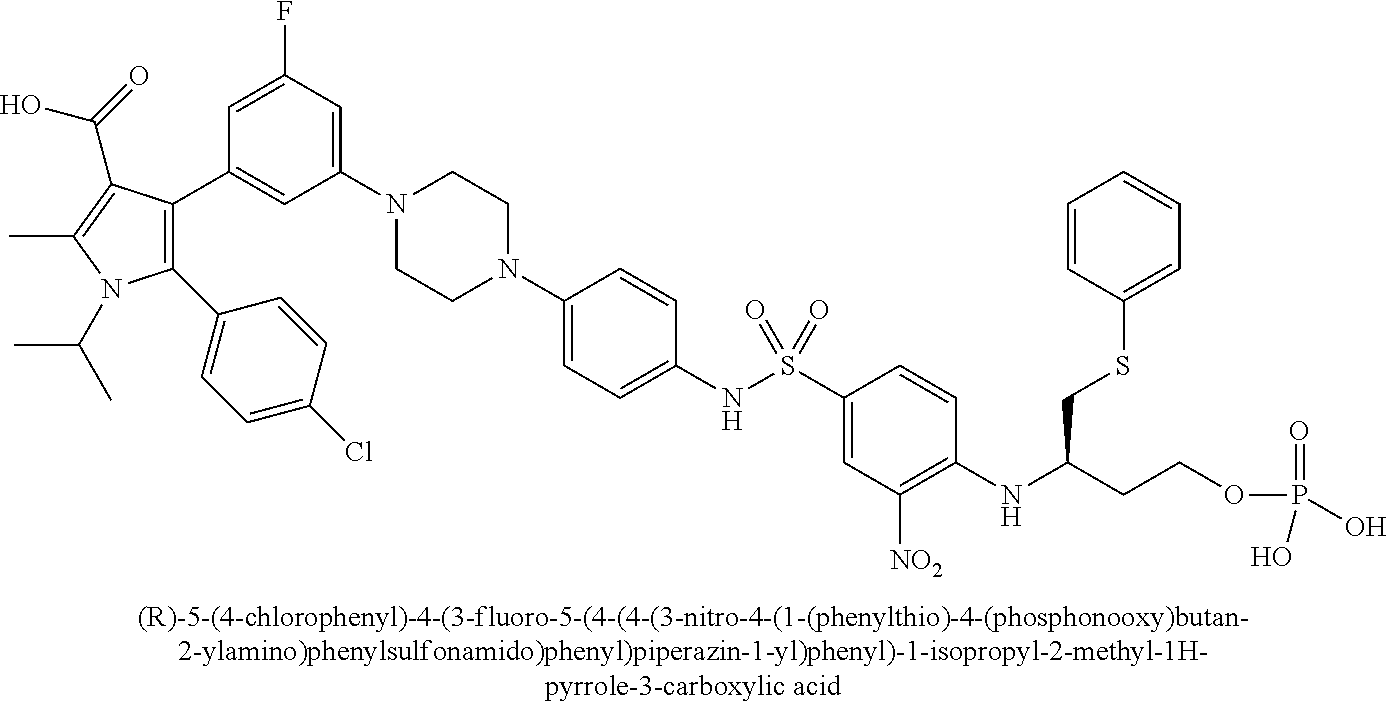
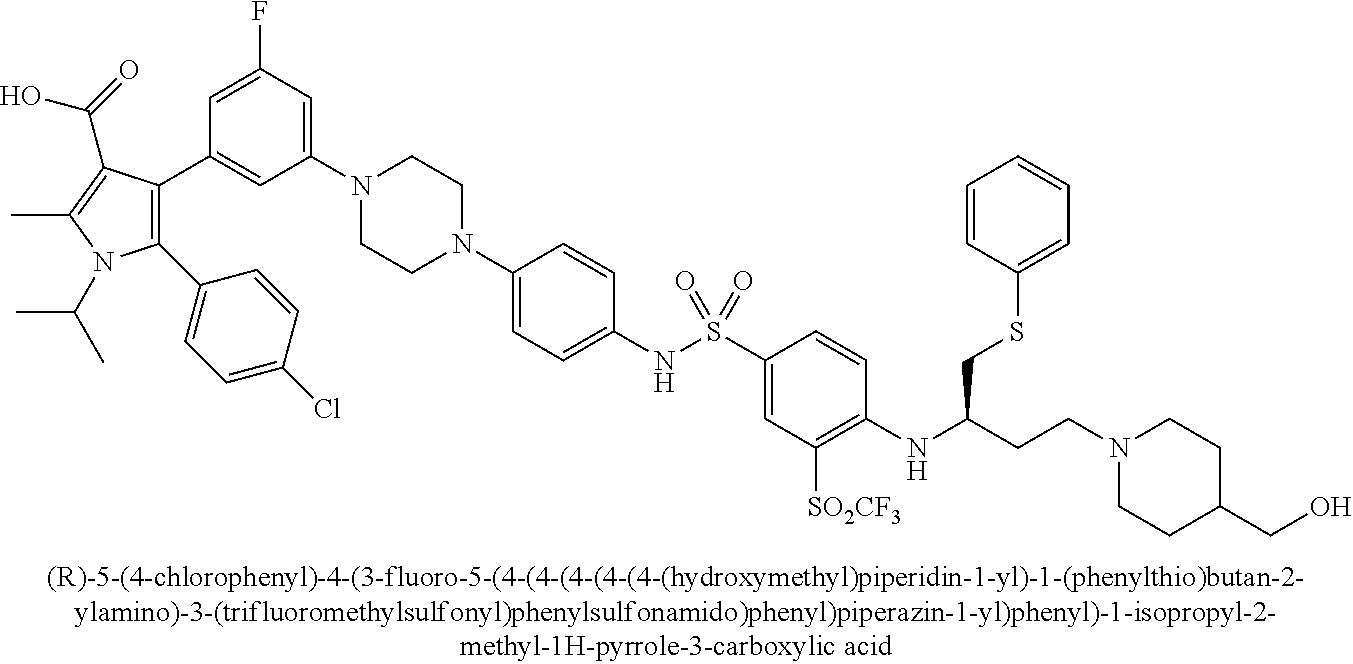
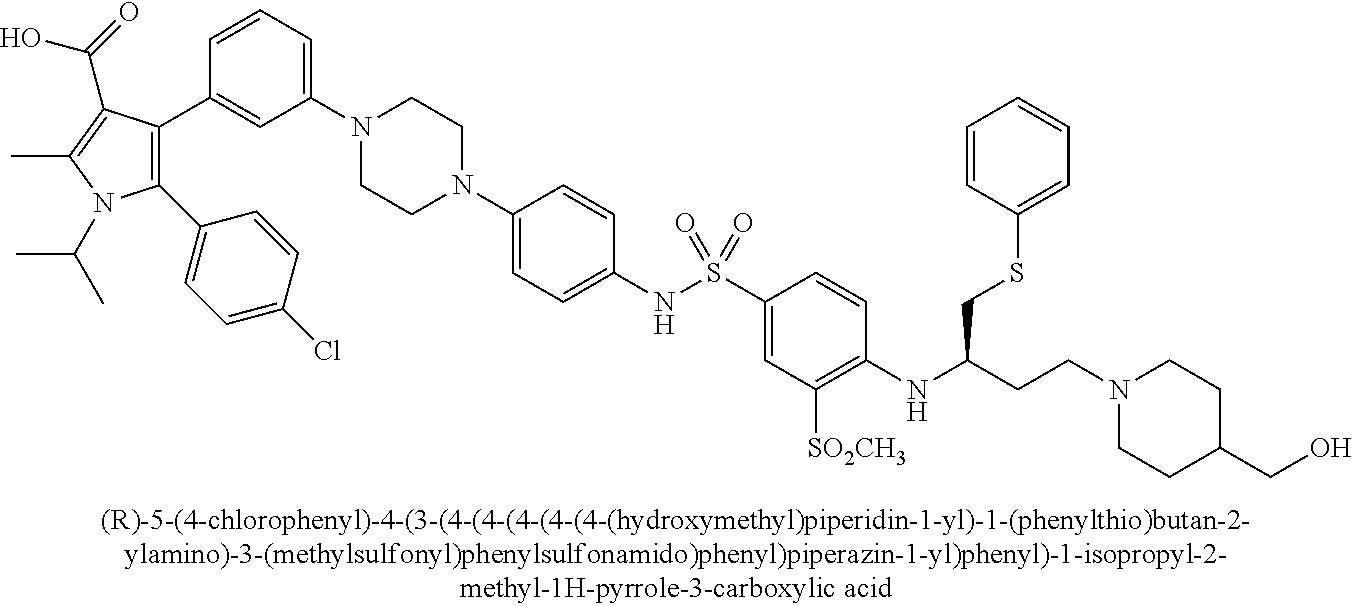




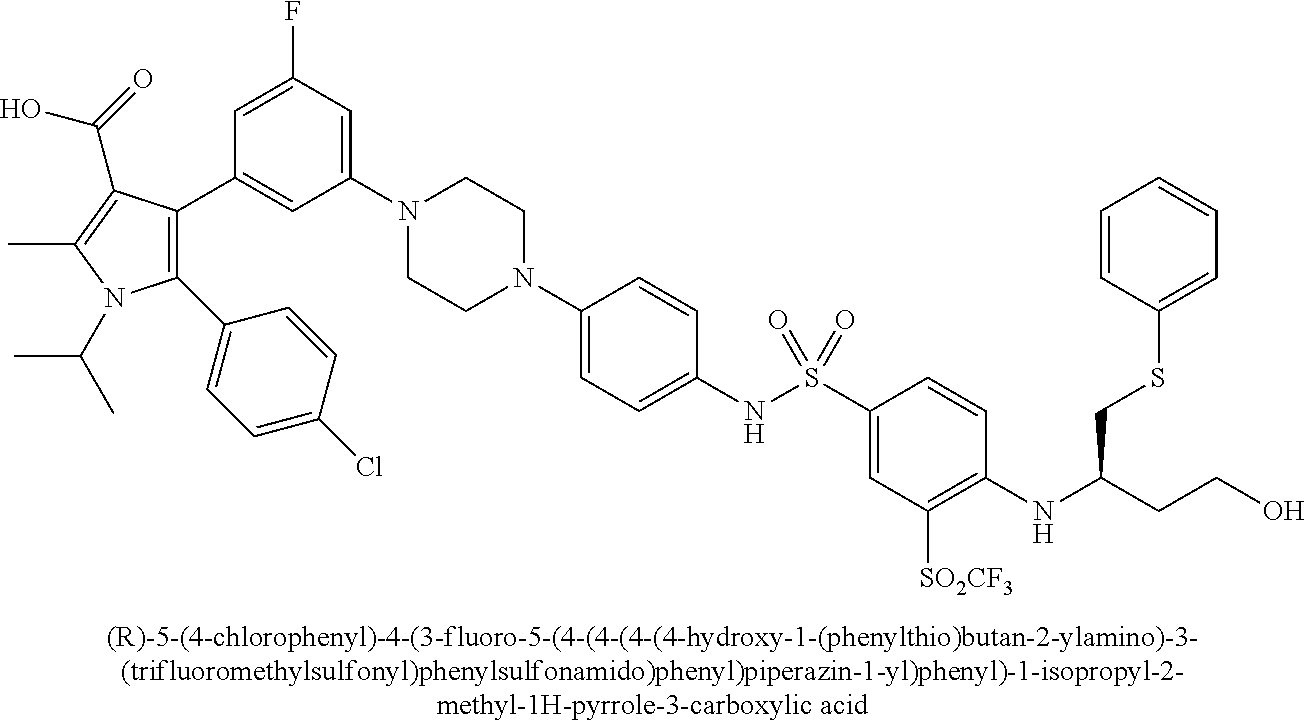




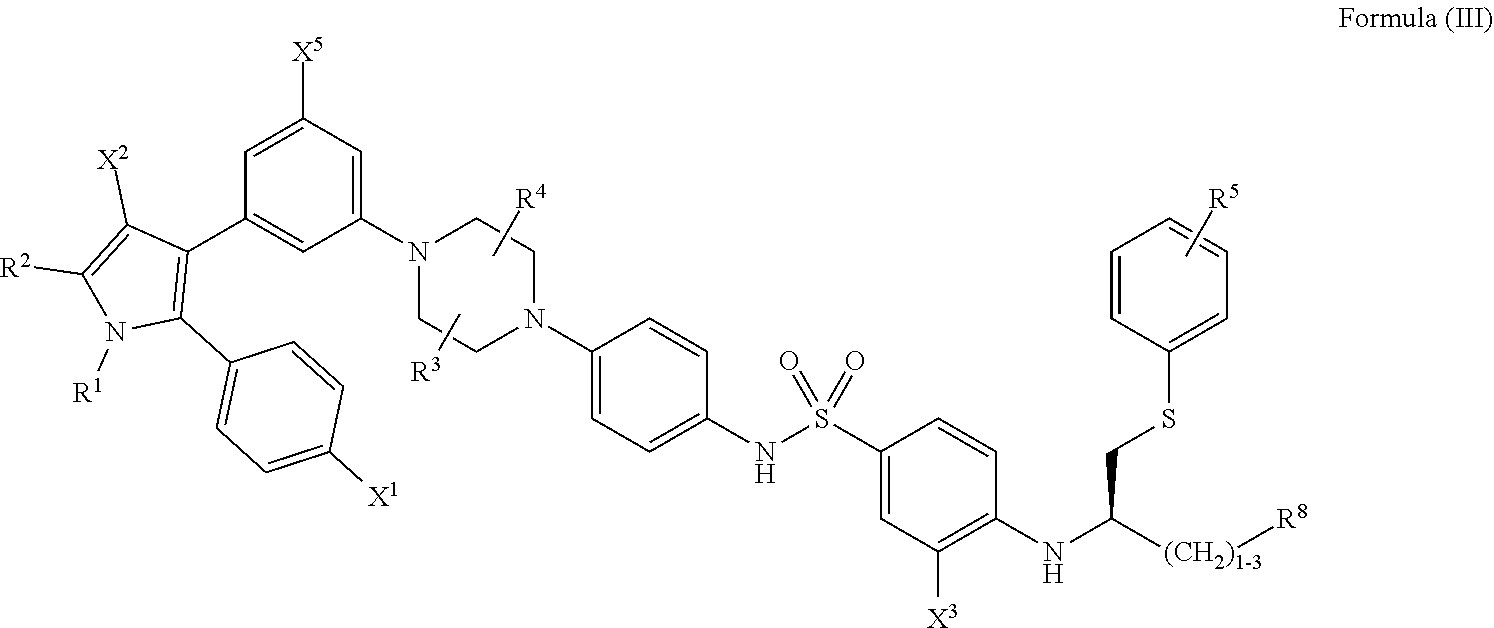
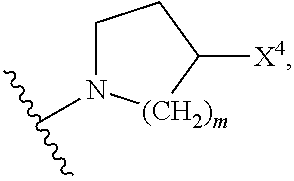
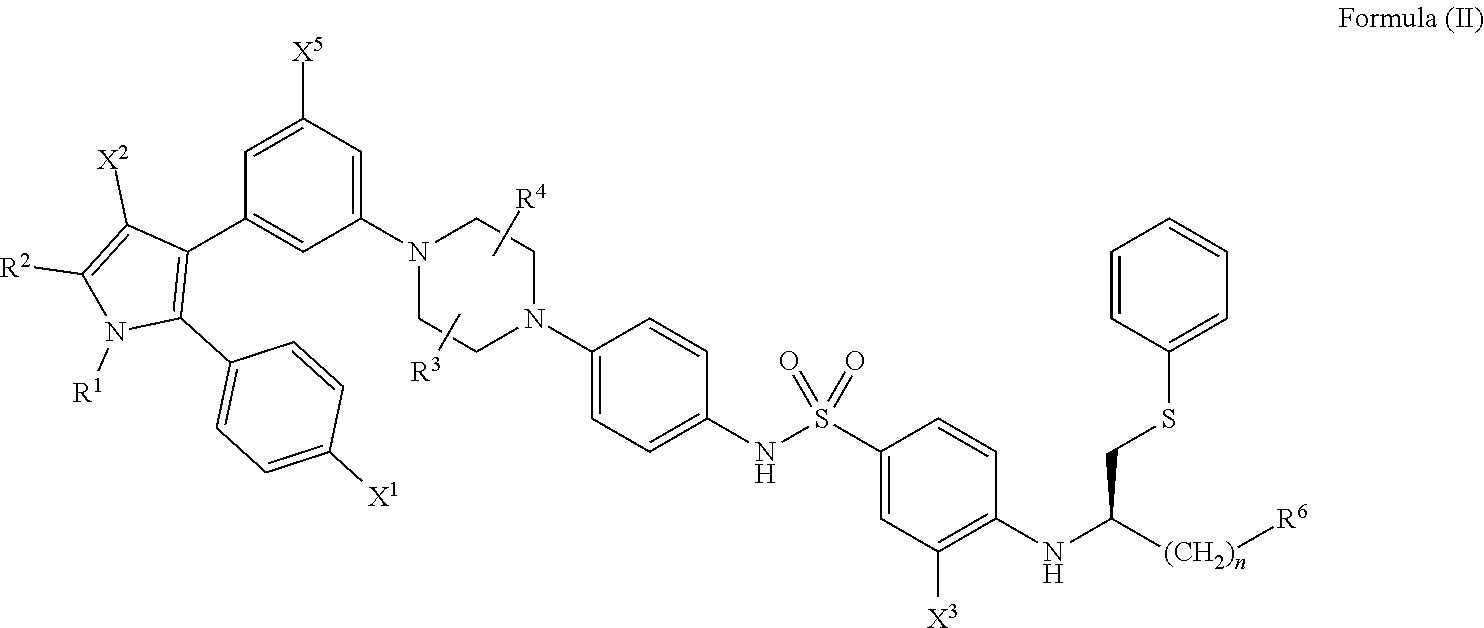




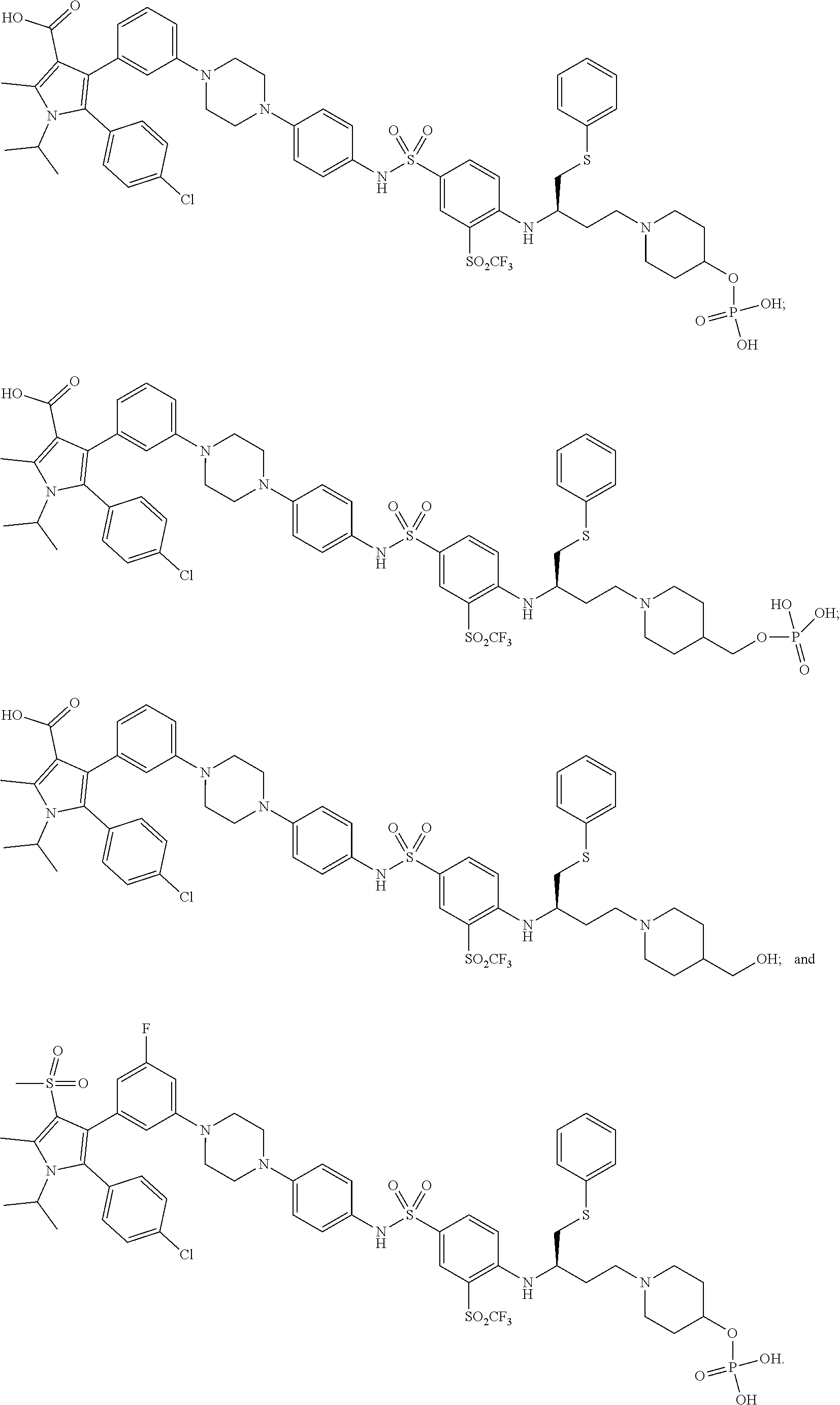
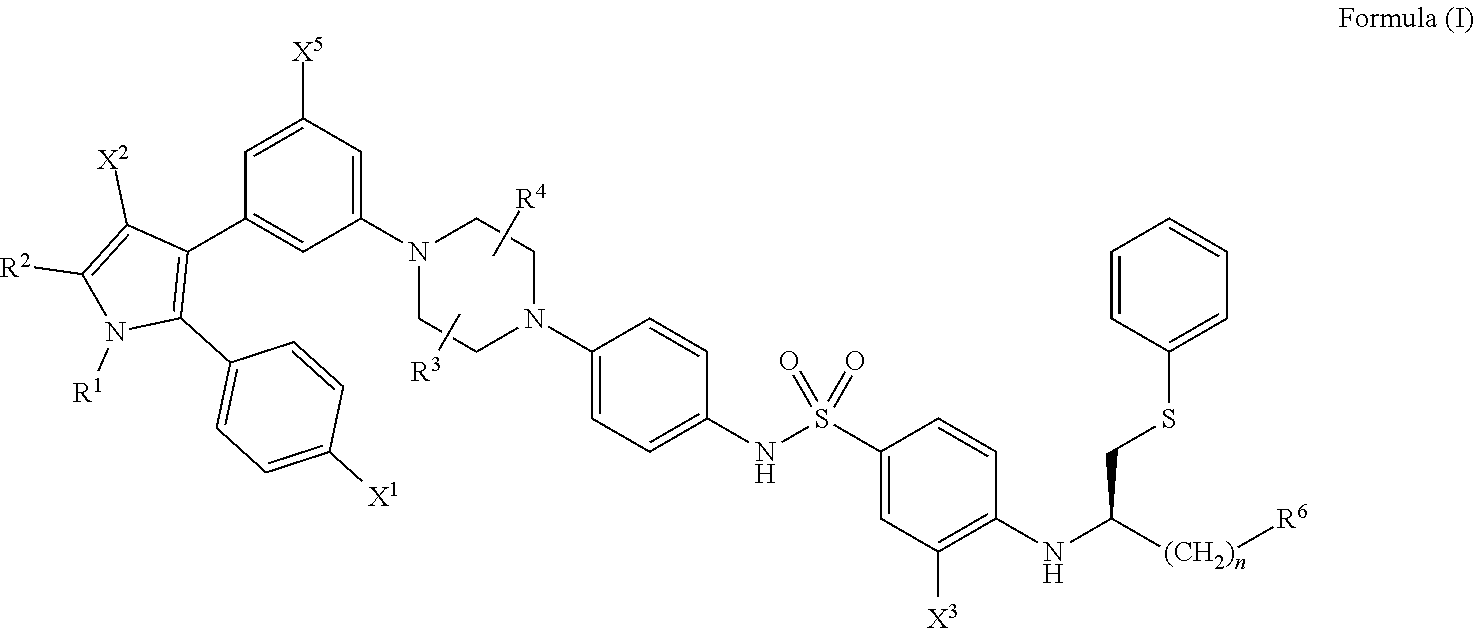

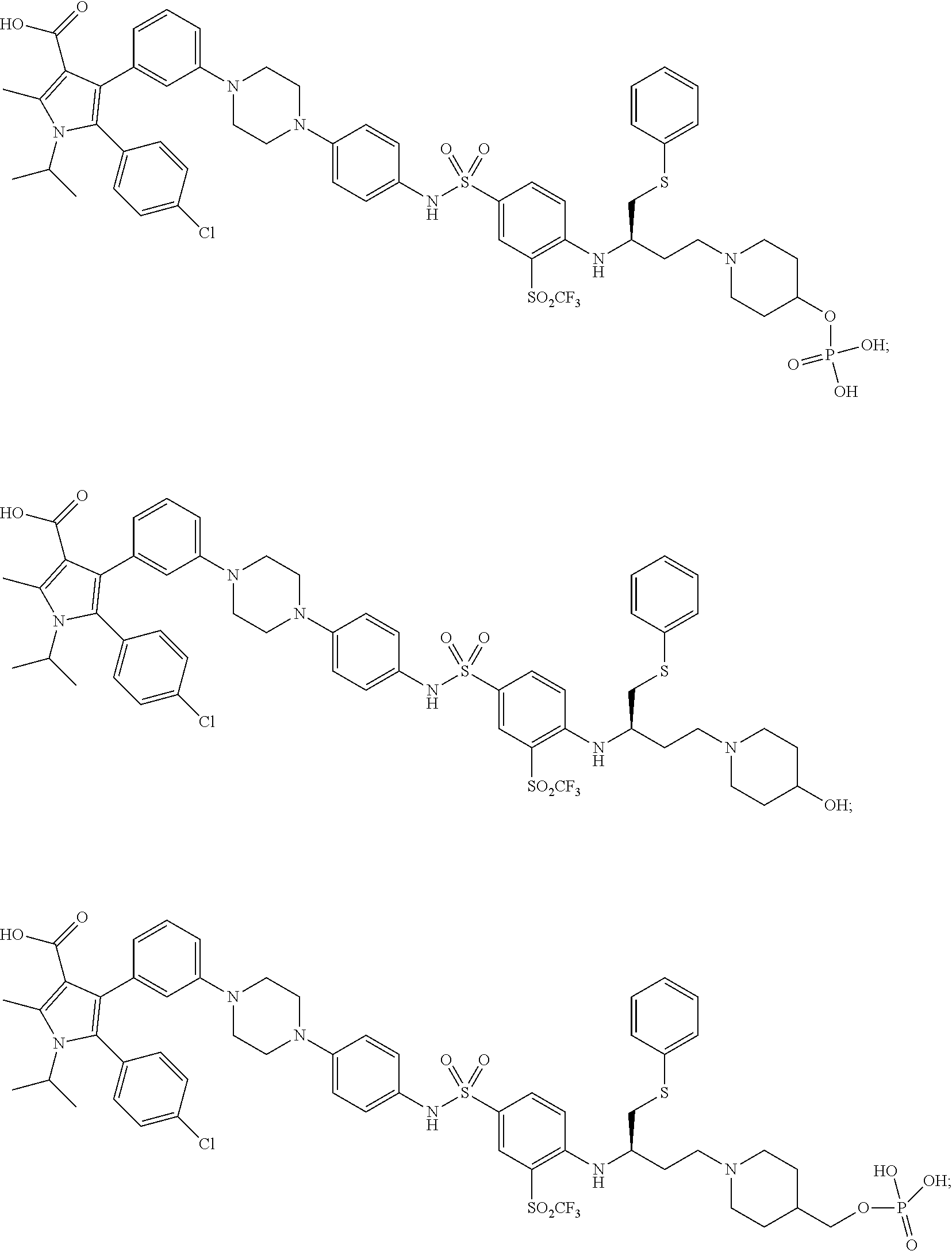




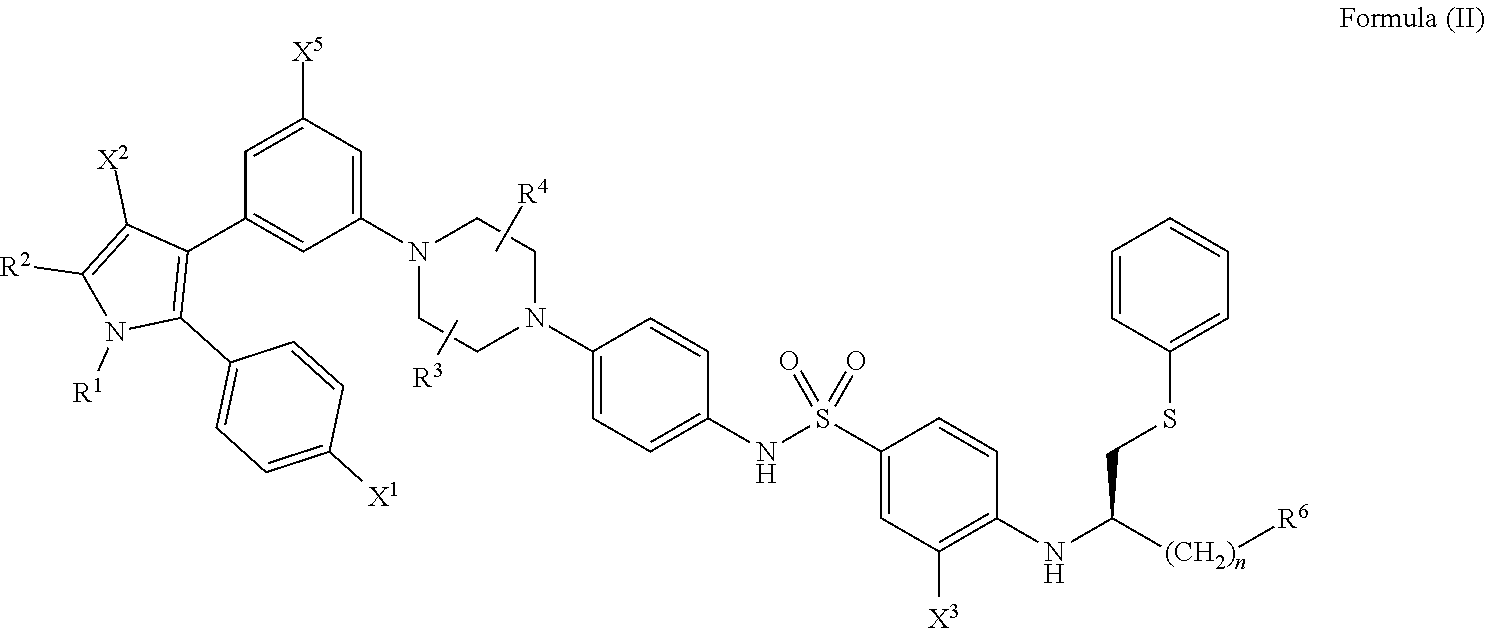
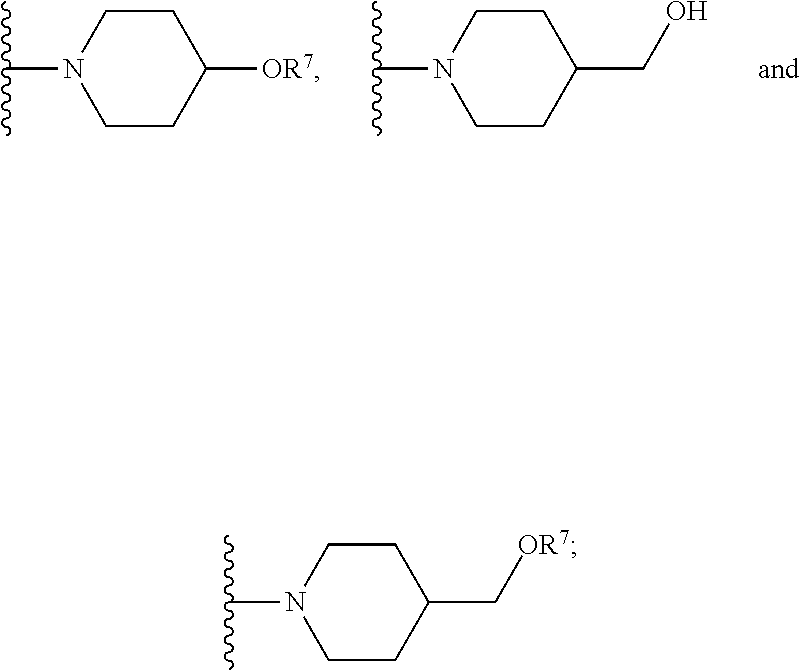
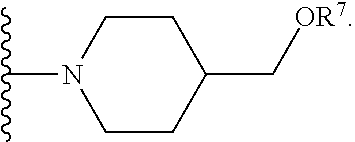
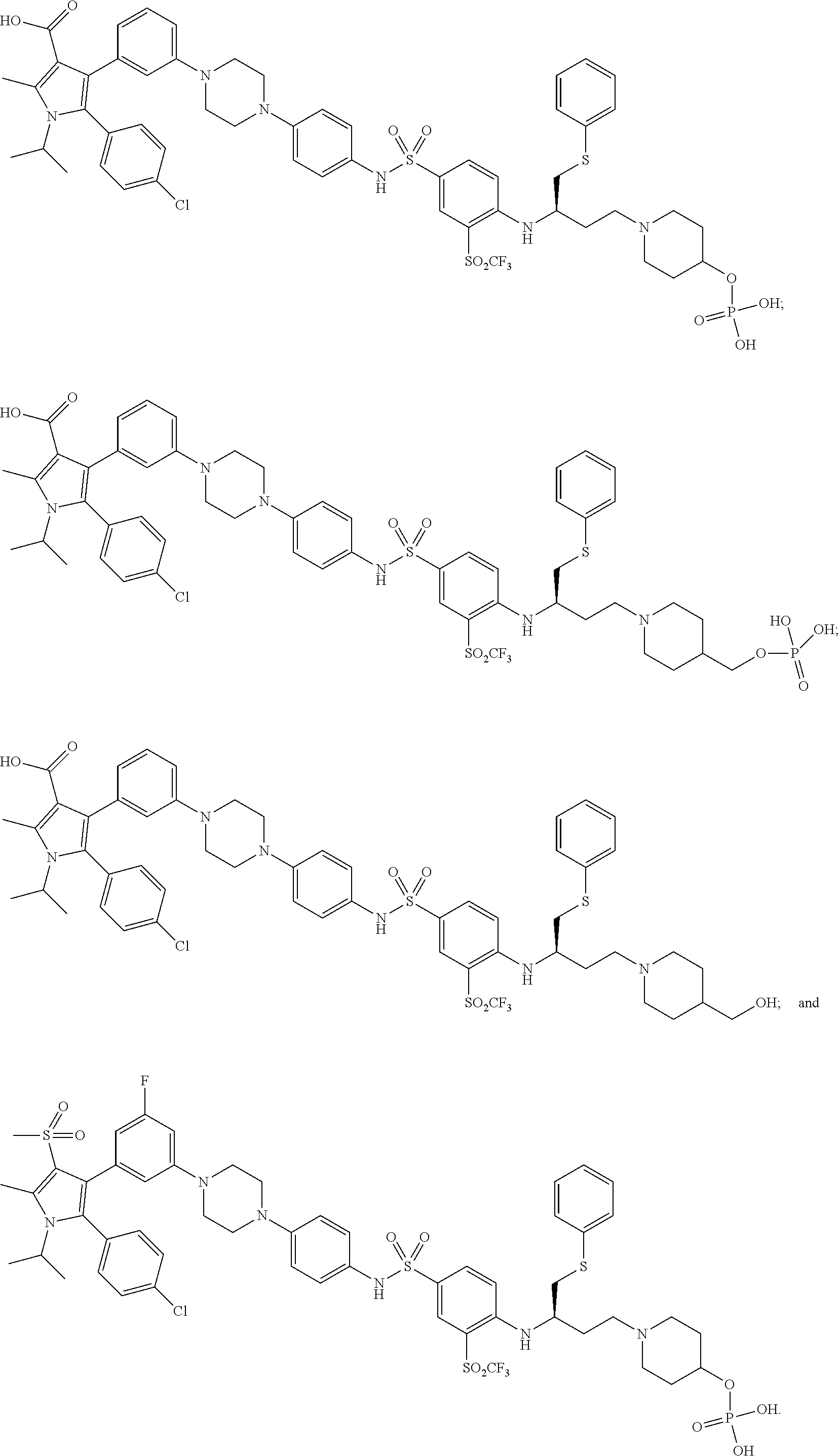

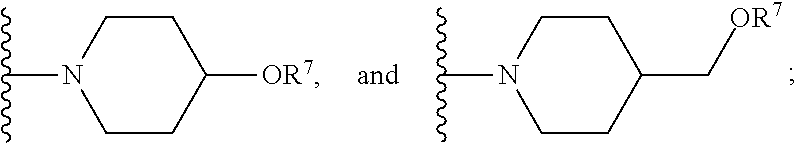

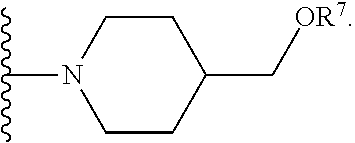


D00000

D00001

D00002

D00003
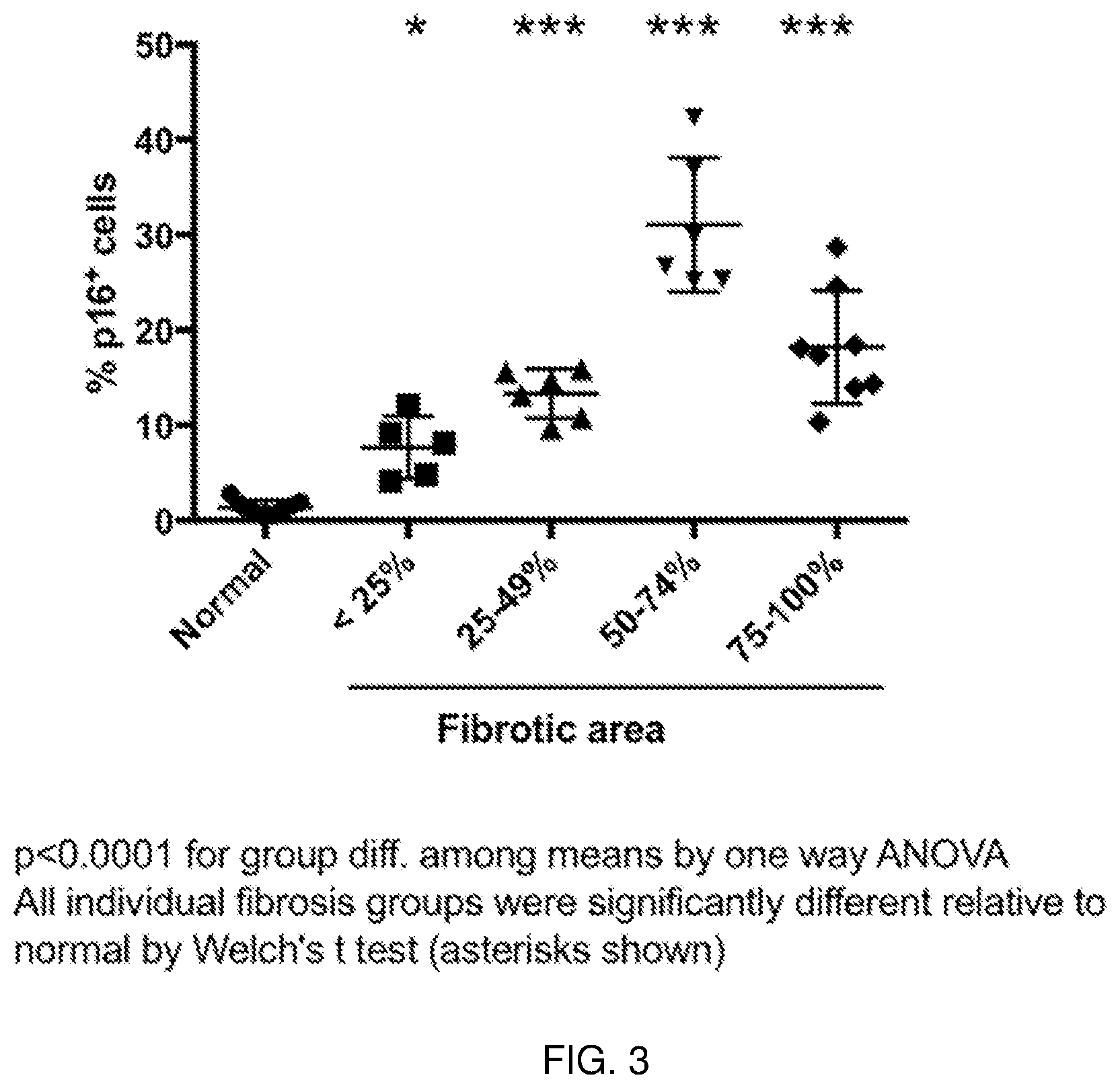
D00004
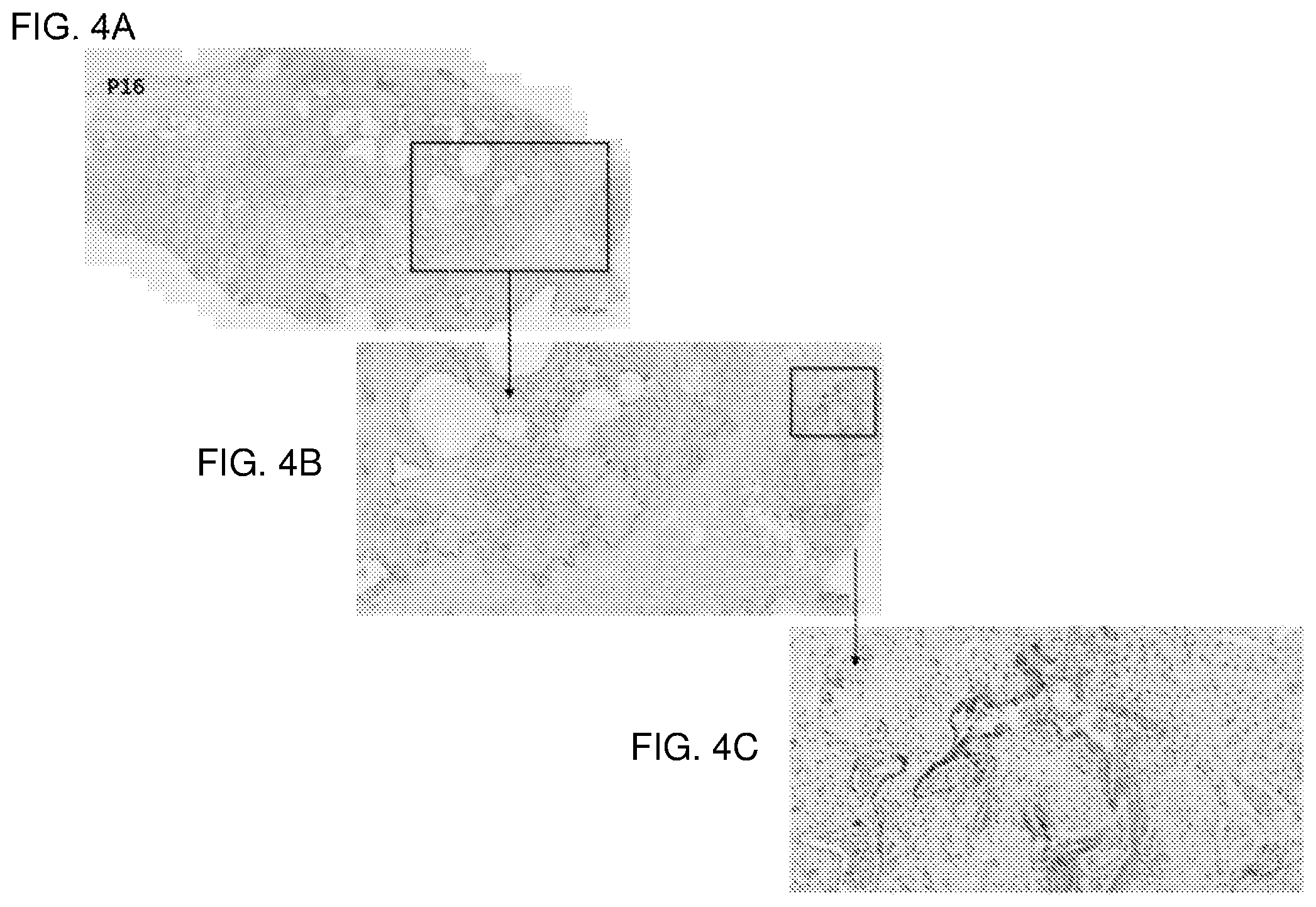
D00005

D00006

D00007

D00008

P00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.