Polyamic acid resin and polyamideimide film
Kim , et al. October 27, 2
U.S. patent number 10,815,378 [Application Number 16/127,671] was granted by the patent office on 2020-10-27 for polyamic acid resin and polyamideimide film. This patent grant is currently assigned to SK Global Chemical Co., Ltd., SK IE Technology Co., Ltd., SK Innovation Co., Ltd.. The grantee listed for this patent is SK Global Chemical Co., Ltd., SK IE Technology Co., Ltd., SK Innovation Co., Ltd.. Invention is credited to Tae Sug Jang, Hyeon Jeong Kim, Jin Hyung Park, Sang Yoon Park.










| United States Patent | 10,815,378 |
| Kim , et al. | October 27, 2020 |
Polyamic acid resin and polyamideimide film
Abstract
Provided are a polyamic acid resin derived from an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride, and a polyamideimide film including polyamideimide derived from an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride.
| Inventors: | Kim; Hyeon Jeong (Daejeon, KR), Park; Sang Yoon (Daejeon, KR), Jang; Tae Sug (Daejeon, KR), Park; Jin Hyung (Daejeon, KR) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | SK Innovation Co., Ltd. (Seoul,
KR) SK Global Chemical Co., Ltd. (Seoul, KR) SK IE Technology Co., Ltd. (Seoul, KR) |
||||||||||
| Family ID: | 1000005141137 | ||||||||||
| Appl. No.: | 16/127,671 | ||||||||||
| Filed: | September 11, 2018 |
Prior Publication Data
| Document Identifier | Publication Date | |
|---|---|---|
| US 20190077960 A1 | Mar 14, 2019 | |
Foreign Application Priority Data
| Sep 12, 2017 [KR] | 10-2017-0116249 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C08G 73/1007 (20130101); C08G 73/1067 (20130101); C08G 73/1042 (20130101); C08G 73/14 (20130101); C08G 73/1039 (20130101); G02F 1/0063 (20130101); C08L 79/08 (20130101); C08L 2203/16 (20130101); G02F 2203/01 (20130101) |
| Current International Class: | C08G 73/14 (20060101); G02F 1/00 (20060101); C08G 73/10 (20060101); C08L 79/08 (20060101) |
References Cited [Referenced By]
U.S. Patent Documents
| 10370496 | August 2019 | Park |
| 2016/0319076 | November 2016 | Ju |
| 2017/0329062 | November 2017 | Nakajima |
| 2002161136 | Jun 2002 | JP | |||
Attorney, Agent or Firm: The Webb Law Firm
Claims
What is claimed is:
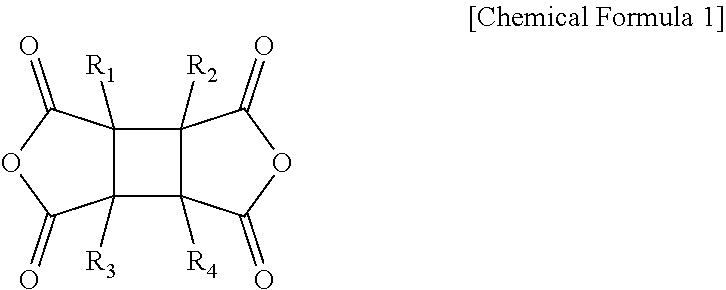
1. A polyamideimide film comprising polyamideimide derived from an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride, wherein the cycloaliphatic dianhydride includes a compound represented by the following Chemical Formula 1, wherein a content of the aromatic dianhydride is 5 to 25 mol of 4,4'-hexafluoroisopropylidene diphthalic anhydride and 5 to 25 mol of biphenyltetracarboxylic dianhydride, based on 100 mol of the aromatic diamine, and the polyamideimide film has a total light transmittance of 88% or more, and a light transmittance measured at 388 nm of 40% or less: ##STR00011## wherein R.sub.1 to R.sub.4 are independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
2. The polyamideimide film of claim 1, wherein the aromatic diamine includes 2,2'-bis(trifluoromethyl)-benzidine.
3. The polyamideimide film of claim 1, wherein the aromatic diacid dichloride includes terephthaloyl dichloride.
4. The polyamideimide film of claim 1, wherein a content of the aromatic diacid dichloride is more than 50 mol, based on 100 mol of the aromatic diamine.
5. The polyamideimide film of claim 1, wherein a content of the cycloaliphatic dianhydride is 5 to 30 mol, based on 100 mol of the aromatic diamine.
6. The polyamideimide film of claim 1, wherein the polyamideimide film has a modulus of 5.0 GPa or more, as measured at an extension speed of 25 mm/min using UTM 3365 available from Instron.
7. The polyamideimide film of claim 1, wherein the polyamideimide film has a yellow index of 3.0 or less, as measured in accordance with the ASTM E313 standard.
8. The polyamideimide film of claim 6, wherein the polyamideimide film has a modulus of 5.0 GPa or more, as measured at an extension speed of 25 mm/min using UTM 3365 available from Instron, for a specimen having a thickness of 55 .mu.m, a length of 50 mm and a width of 10 mm.
9. The polyamideimide film of claim 1, wherein the polyamideimide film has a total light transmittance of 88% or more, based on a thickness of 55 .mu.m.
10. The polyamideimide film of claim 7, wherein the polyamideimide film has a yellow index of 3.0 or less, as measured in accordance with the ASTM E313 standard, based on a thickness of 55 .mu.m.
11. The polyamideimide film of claim 1, wherein the polyamideimide film has a light transmittance measured at 388 nm of 40% or less, based on a thickness of 55 .mu.m.
12. An image display device comprising the polyamideimide film of claim 1.
Description
CROSS-REFERENCE TO RELATED APPLICATION
This application claims priority to Korean Patent Application No. 10-2017-0116249 filed Sep. 12, 2017, the disclosure of which is hereby incorporated in its entirety by reference.
TECHNICAL FIELD
The following disclosure relates to a polyamic acid resin and a polyamideimide film. More particularly, the following disclosure relates to a polyamic acid resin derived from a combination of specific components, and a polyamideimide film including polyamideimide derived therefrom, having adjusted optical properties and being capable of implementing a high modulus.
BACKGROUND
In general, polyimide has excellent mechanical and thermal properties, thereby being applied to various fields including an insulating substrate field for forming circuits and devices. However, since a charge transfer complex is formed between aromatic rings at the time of polymerization to cause polyimide to be brown or yellow colored, leading to low light transmittance in the visible light region, it is difficult to apply the polyimide to display materials.
As a method of improving the light transmittance of the polyimide to make the polyimide colorless and transparent, a method of using alicyclic diamine or aliphatic diamine as a diamine component to inhibit formation of an intramolecular charge transfer complex is known in the art. Japanese Patent Laid-Open Publication No. 2002-161136 (Patent Document 1) discloses polyimide obtained by imidizing a polyimide precursor formed by aromatic acid dianhydride such as pyromellitic dianhydride and trans-1,4-diaminocyclohexane, however, which represents high transparency but has deteriorated mechanical physical properties.
As such, as a method for converting the yellow color of polyimide to be colorless and transparent, an attempt has been made to use various functional monomers. However, since a polyimide film having a high light transmittance in the visible light region also has a high light transmittance value in a short wavelength region of 400 nm or less, when the film is exposed to ultraviolet rays, damage due to ultraviolet rays occurred in a lower laminate structure of a display including the polyimide film. In order to solve the problem, it was intended to use a widely used ultraviolet ray absorber or ultraviolet ray stabilizer, however, polyimide is processed at high temperature so that it is hard to use an additive, and even in the case of using the additive, a yellow index was increased.
Thus, it is currently needed to develop a technology for polyimide having excellent inherent mechanical physical properties which is not deteriorated by implementing a high modulus so that the polyimide may be applied to various display material fields, and capable of preventing damage of a lower laminate structure of a display including a polyimide film due to ultraviolet rays by decreasing a light transmittance in a short wavelength region of 400 nm or less, so that the coverage may be further broadened.
RELATED ART DOCUMENT
Patent Document
(Patent Document 1) Japanese Patent Laid-Open Publication No. 2002-161136 (Jun. 4, 2002)
SUMMARY
An embodiment of the present invention is directed to providing a polyamic acid resin and a polyamideimide film which may have excellent mechanical physical properties simultaneously with a low yellow index, and have an excellent light transmittance in an entire wavelength region of visible light and implement a low light transmittance in a short wavelength region.
In particular, an embodiment of the present invention is directed to providing a polyamideimide film capable of implementing a high modulus.
In addition, an embodiment of the present invention is directed to providing a polyamideimide film capable of preventing ultraviolet ray-induced damage by ultraviolet ray exposure to a lower structure of a display including the polyamideimide film.
In one general aspect, a polyamideimide film includes polyamideimide derived from an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride, wherein
the cycloaliphatic dianhydride includes a compound represented by the following Chemical Formula 1, and
the polyamideimide film may have a total light transmittance of 88% or more, and a light transmittance measured at 388 nm of 40% or less, as measured in accordance with the ASTM D1003 standard:
##STR00001##
wherein
R.sub.1 to R.sub.4 are independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
According to an exemplary embodiment of the present invention, the aromatic diamine may include 2,2'-bis(trifluoromethyl)-benzidine.
According to an exemplary embodiment of the present invention, the aromatic dianhydride may include 4,4'-hexafluoroisopropylidene diphthalic anhydride and biphenyltetracarboxylic dianhydride.
According to an exemplary embodiment of the present invention, the aromatic diacid dichloride may include terephthaloyl dichloride.
According to an exemplary embodiment of the present invention, a content of the aromatic diacid dichloride may be more than 50 mol, based on 100 mol of the aromatic diamine.
According to an exemplary embodiment of the present invention, a content of the aromatic dianhydride may be 10 to 50 mol, based on 100 mol of the aromatic diamine.
According to an exemplary embodiment of the present invention, a content of the aromatic dianhydride may be 5 to 25 mol of 4,4'-hexafluoroisopropylidene diphthalic anhydride and 5 to 25 mol of biphenyltetracarboxylic dianhydride, based on 100 mol of the aromatic diamine.
According to an exemplary embodiment of the present invention, a content of the cycloaliphatic dianhydride may be 5 to 30 mol, based on 100 mol of the aromatic diamine.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a modulus of 5.0 GPa or more, as measured at an extension speed of 25 mm/min using UTM 3365 available from Instron.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a yellow index of 3.0 or less, as measured in accordance with the ASTM E313 standard.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a modulus of 5.0 GPa or more, as measured at an extension speed of 25 mm/min using UTM 3365 available from Instron for a specimen having a thickness of 55 .mu.m, a length of 50 mm and a width of 10 mm.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a total light transmittance of 88% or more, as measured in accordance with the ASTM D1003 standard, based on a film having a thickness of 55 .mu.m.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a yellow index of 3.0 or less, as measured in accordance with the ASTM E313 standard, based on a film having a thickness of 55 .mu.m.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a light transmittance of 40% or less, as measured at 388 nm, based on a film having a thickness of 55 .mu.m.
In another general aspect, an image display device includes the polyamideimide film as described above.
In still another general aspect, a polyamic acid resin is derived from an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride, wherein
the cycloaliphatic dianhydride is represented by the following Chemical Formula 1:
##STR00002##
wherein
R.sub.1 to R.sub.4 are independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
According to an exemplary embodiment of the present invention, the aromatic diamine may include 2,2'-bis(trifluoromethyl)-benzidine.
According to an exemplary embodiment of the present invention, the aromatic dianhydride may include 4,4'-hexafluoroisopropylidene diphthalic anhydride and biphenyltetracarboxylic dianhydride.
According to an exemplary embodiment of the present invention, the aromatic diacid dichloride may include terephthaloyl dichloride.
Other features and aspects will be apparent from the following detailed description, the drawings, and the claims.
DETAILED DESCRIPTION OF EMBODIMENTS
Hereinafter, the present invention will be described in more detail with reference to the exemplary embodiments and Examples including the accompanying drawings. However, the following exemplary embodiments and Examples are only a reference for describing the present invention in detail, and the present invention is not limited thereto, and may be implemented in various forms.
In addition, unless otherwise defined, all technical terms and scientific terms have the same meanings as those commonly understood by a person skilled in the art to which the present invention pertains, the terms used herein is only for effectively describing a certain exemplary embodiment, and not intended to limit the present invention.
Throughout the present specification describing the present invention, unless explicitly described to the contrary, "comprising" any elements will be understood to imply further inclusion of other elements rather than the exclusion of any other elements.
In addition, the singular form used in the specification and claims appended thereto may be intended to also include a plural form, unless otherwise indicated in the context.
In addition, "a polyamic acid solution", which is a solution formed by mixing monomers in an organic solvent, refers to containing a polyamic acid resin obtained by copolymerization of monomers in the organic solvent.
The inventors of the present invention provide a polyamideimide film including an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride. That is, a modulus may be surprisingly excellent, a yellow index may be significantly lowered, and a light transmittance over an entire region of visible light may be raised, by deriving polyamideimide from a polyamic acid resin having a combination of specific aromatic diamine, dianhydride and aromatic diacid dichloride. At the same time, it was found that a polyamideimide film preventing damage due to ultraviolet rays in a lower structure of a display including the polyamideimide film may be provided by significantly decreasing a light transmittance in a short wavelength region, thereby completing the present invention.
Specifically, in order to provide a resin and a film which may be applied various display fields by improving optical properties as well as mechanical and thermal properties, the present invention provides a polyamic acid resin including specific aromatic diamine, aromatic dianhydride, cycloaliphatic dianhydride and aromatic diacid dichloride, and a polyamideimide film derived therefrom.
According to an exemplary embodiment of the present invention, the present invention relates to a polyamideimide film including polyamideimide derived from an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride, wherein
the cycloaliphatic dianhydride includes a compound represented by the following Chemical Formula 1, and
the polyamideimide film may have a total light transmittance of 88% or more, and a light transmittance measured at 388 nm of 40% or less, as measured in accordance with the ASTM D1003 standard:
##STR00003##
wherein
R.sub.1 to R.sub.4 are independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
Preferably, according to an exemplary embodiment of the present invention, the present invention relates to a polyamideimide film including polyamideimide derived from an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride, wherein
the aromatic diamine includes 2,2'-bis(trifluoromethyl)-benzidine,
the aromatic dianhydride may include 4,4'-hexafluoroisopropylidene diphthalic anhydride and biphenyltetracarboxylic dianhydride,
the cycloaliphatic dianhydride includes a compound represented by the following Chemical Formula 1, and
the aromatic diacid dichloride includes terephthaloyl dichloride:
##STR00004##
wherein
R.sub.1 to R.sub.4 are independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a total light transmittance of 88% or more, as measured in accordance with the ASTM D1003 standard. Preferably the total light transmittance may be 89% or more. Specifically, the total light transmittance may be 88 to 99%. More preferably, the total light transmittance may be 89 to 95%. Here, the total light transmittance may be measured, based on a specimen having a thickness of 55 .mu.m.
In addition, according to an exemplary embodiment of the present invention, the polyamideimide film may have a light transmittance of 40% or less, as measured at 388 nm using UV-3600 available from Shimadzu. Preferably, the light transmittance measured at 388 nm may be 30% or less. Specifically, the light transmittance measured at 388 nm may be 1 to 40%. Preferably, the light transmittance measured at 388 nm may be 5 to 30%. Here, the light transmittance measured at 388 nm may be measured, based on a specimen having a thickness of 55 .mu.m.
The polyamideimide film including the polyamideimide by the combination of the present invention as described above has an excellent modulus, while significantly lowering a yellow index and raising a light transmittance over an entire wavelength region of visible light. At the same time, as the light transmittance is significantly decreased in a short wavelength region, damage due to ultraviolet rays in a lower structure of a display including the polyamideimide film may be prevented without an ultraviolet additive. In addition, since the ultraviolet additive is not included, deterioration of physical properties of polyamideimide may be prevented, and thus, the film is preferred.
According to an exemplary embodiment of the present invention, the cycloaliphatic dianhydride may include a compound represented by the following Chemical Formula 1:
##STR00005##
wherein
R.sub.1 to R.sub.4 may be independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
Preferably, in Chemical Formula 1,
R.sub.1 to R.sub.4 may be independently of one another hydrogen or a C.sub.1 to C.sub.10 alkyl group.
More preferably, in Chemical Formula 1,
R.sub.1 to R.sub.4 may be independently of one another hydrogen or a C.sub.1 to C.sub.5 alkyl group.
Most preferably, specifically for example, 1,2,3,4-cyclobutanetetracarboxylic dianhydride (CBDA) may be included.
When the cycloaliphatic dianhydride is included, a low yellow index may be implemented, and a light transmittance in a visible light region may be improved, in the polyamideimide film prepared by the combination of the aromatic diamine, the aromatic dianhydride and the aromatic diacid dichloride as described above, which is thus preferred.
According to an exemplary embodiment of the present invention, a content of the cycloaliphatic dianhydride may be to 30 mol, based on 100 mol of the aromatic diamine. Preferably, the content may be 5 to 20 mol. When the cycloaliphatic dianhydride is included at the content, a yellow index is significantly decreased, and a light transmittance over an entire wavelength region of visible light may be raised, which is thus preferred.
The aromatic diamine according to an exemplary embodiment of the present invention may include 2,2'-bis(trifluoromethyl)-benzidine. The aromatic diamine may impart excellent optical properties by a charge transfer effect of fluorine substituents, and 2,2'-bis(trifluoromethyl)-benzidine may provide polyamideimide which is an imidized product of a polyamic acid resin with a combination of an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride, thereby implementing optical properties to be desired depending on a wavelength region, and also excellent mechanical physical properties.
Here, the aromatic diamine may be used by mixing 2,2'-bis(trifluoromethyl)-benzidine and other known aromatic diamine components, however, it is more preferred to use 2,2'-bis(trifluoromethyl)-benzidine alone for implementing the effect to be achieved.
The dianhydride according to an exemplary embodiment of the present invention includes an aromatic dianhydride and a cycloaliphatic dianhydride.
The aromatic dianhydride of the present invention is specifically for example, any one or a mixture of two or more selected from the group consisting of 4,4'-hexafluoroisopropylidene diphthalic anhydride (6-FDA), biphenyltetracarboxylic dianhydride (BPDA), 1,2,4,5-benzenetetracarboxylic dianhydride (PMDA), benzophenonetetracarboxylic dianhydride (BTDA), 4,4'-oxydiphthalic dianhydride (ODPA), bisdicarboxyphenoxy diphenylsulfide dianhydride (BDSDA), IBIS-BAN (CAS No. 867350-98-9), TBIS-MPN (CAS No. 933041-59-9) and the like. It is more preferred to use only 4,4'-hexafluoroisopropylidene diphthalic anhydride and biphenyltetracarboxylic dianhydride for implementing the physical property effect to be desired.
A content of the aromatic dianhydride according to an exemplary embodiment of the present invention may be 10 to 50 mol, based on 100 mol of the aromatic diamine. Preferably, the content may be 10 to 40 mol. More preferably, the content may be 20 to 40 mol.
More preferably, a content of the aromatic dianhydride according to an exemplary embodiment of the present invention may be 5 to 25 mol of 4,4'-hexafluoroisopropylidene diphthalic anhydride and 5 to 25 mol of biphenyltetracarboxylic dianhydride, based on 100 mol of the aromatic diamine. Preferably, the content may be 10 to 20 mol of 4,4'-hexafluoroisopropylidene diphthalic anhydride and 10 to 25 mol of biphenyltetracarboxylic dianhydride. Within the range of the content, when the polyamideimide film is prepared, the light transmittance is significantly decreased in a short wavelength region, and damage due to ultraviolet rays in a lower structure of a display including the polyamideimide film may be prevented, which is preferred.
According to an exemplary embodiment of the present invention, the aromatic diacid dichloride includes terephthaloyl dichloride for being reacted with the aromatic diamine to form an amide structure in a high molecular chain. Besides, other known aromatic diacid dichloride may be further included. Specifically for example, any one or a mixture of two or more selected from the group consisting of 1,4-naphthalene dicarboxylic dichloride, 2,6-naphthalene dicarboxylic dichloride, 1,5-naphthalene dicarboxylic dichloride and the like may be used. It is preferred to use terephthaloyl dichloride alone for adjusting optical properties depending on the range of the polyamideimide film, and also significantly improving the modulus.
According to an exemplary embodiment of the present invention, a content of the aromatic diacid dichloride may be more than 50 mol, based on 100 mol of the aromatic diamine. Preferably, the content may be 55 mol or more. Specifically, the content may be 55 to 90 mol, preferably 55 to 80 mol, more preferably 55 to 75 mol.
When the content satisfies the range, the physical property balance to be desired, that is, a high light transmittance over an entire region of visible light without deteriorating mechanical and thermal physical properties may be implemented, with the combination of other components. Besides, by implementing a low light transmittance even in a short wavelength region of 400 nm or less, damage due to ultraviolet rays in a lower structure of a display including the polyamideimide film may be prevented. In addition, a yellow index may be further decreased, and a synergistic effect of dramatically improving a modulus may be implemented.
By copolymerizing a high content of the aromatic diacid dichloride as described above, the polyamideimide film prepared therefrom has dramatically improved optical properties, and at the same time may implement a high modulus, which are preferred.
In particular, there was difficulty in using the aromatic diacid dichloride at a high content more than 50 mol, based on the diamine due to gelation and the like during a polymerization reaction. Thus, conventionally, in order to use the aromatic diacid dichloride at a high content, lithium chloride, calcium chloride or the like was used together, however, these compounds leave a chloride ion to have a bad influence on the environment or deteriorate the physical properties of the film. In order to solve the problems, in the present invention, a compositional ratio depending on a combination with other components including the aromatic diamine, the aromatic dianhydride and the cycloaliphatic dianhydride, and a reaction order and a polymerization concentration of the reaction components may be adjusted to dramatically increase the content of the aromatic diacid dichloride, thereby achieving the physical property effect to be desired.
Since the specific example, content and the like of the above-described aromatic diamine, aromatic dianhydride, cycloaliphatic dianhydride and aromatic diacid dichloride are described above, detailed description will be omitted.
In the present invention, the polyamic acid resin which is a precursor before preparing polyamideimide may be preferably a resin produced by copolymerizing an aromatic diamine, an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride.
Specifically, a polyamic acid resin wherein the aromatic diamine includes 2,2'-bis(trifluoromethyl)-benzidine, and the aromatic dianhydride includes 4,4'-hexafluoroisopropylidene diphthalic anhydride and biphenyltetracarboxylic dianhydride, the cycloaliphatic dianhydride includes a compound represented by the following Chemical Formula 1, and the aromatic diacid dichloride includes terephthaloyl dichloride, may be provided.
##STR00006##
wherein
R.sub.1 to R.sub.4 may be independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
In the present invention, an equivalent ratio of the aromatic diamine and a mixture of the aromatic dianhydride, the cycloaliphatic dianhydride and the aromatic diacid dichloride is preferably 0.9:1 to 1.1:1, and more preferably 1:1. When the range is satisfied, the physical properties of the film including the film forming properties are improved when forming a film from polyamideimide which is obtained by imidizing the polyamic acid resin derived from the monomer, which is more preferred.
The polyamic acid resin may be provided as a polyamic acid solution dissolved in a solvent, and the polyamic acid solution is a solution of the above-described monomers, and may include an organic solvent for a solution polymerization reaction. The kind of the organic solvent is not particularly limited, and specifically for example, it is preferred to use any one or two or more selected from the group consisting of dimethylacetamide (DMAc), N-methyl-pyrrolidone (NMP), dimethylformamide (DMF), dimethylformsulfoxide (DMSO), acetone, diethylacetamide, m-cresol and the like.
In the present invention, the polyamic acid solution including the polyamic acid resin and the solvent may be imidized by further including any one or two or more selected from the group consisting of an imidization catalyst and a dehydrating agent, in addition to the polyamic acid resin.
As the imidization catalyst, any one or more selected from the group consisting of pyridine, isoquinoline and .beta.-quinoline may be used. In addition, as the dehydrating agent, any one or more selected from the group consisting of an acetic anhydride, a phthalic anhydride and a maleic anhydride may be used, but not necessarily limited thereto.
Any one or a mixture of two or more selected from the group consisting of the imidization catalyst and the dehydrating agent according to an exemplary embodiment of the present invention may be included at 1 to 5 mol, based on 1 mol of the aromatic dianhydride and the cycloaliphatic dianhydride. Preferably, 1.5 to 3 mol may be included, but not limited thereto.
In the present invention, the polyamic acid solution may be imidized to obtain the polyamideimide film. Here, imidization may be carried out using a known imidization method. Chemical imidization is preferred. Chemical imidization of the polyamic acid solution by including pyridine and an acetic anhydride is more preferred.
The polyamic acid solution according to the present invention may further include various forms of additives. As the additive, any one or two or more selected from the group consisting of a flame retardant, an adhesion improver, inorganic particle, an antioxidant, an ultraviolet ray inhibitor, a plasticizer, an antistatic agent and the like may be further included, but not necessarily limited thereto.
In addition, the present invention provides an image display device including the above-described polyamideimide film.
The present invention provides a method for preparing a polyamideimide film including the following steps:
(a) dissolving an aromatic diamine in an organic solvent, then adding an aromatic dianhydride, a cycloaliphatic dianhydride and an aromatic diacid dichloride thereto, and performing a reaction to prepare a polyamic acid solution;
(b) imidizing the polyamic acid solution to prepare polyamideimide; and
(c) coating a polyamide imide solution in which the polyamideimide is dissolved in an organic solvent.
In the present invention, the method for preparing a polyamideimide film is not particularly limited, however, it is preferred to perform the method using a reactor equipped with a stirrer, a nitrogen injection apparatus, a dropping apparatus, a temperature controller and a cooler.
According to an exemplary embodiment of the present invention, step (a) of preparing a polyamic acid solution may be to add an organic solvent in a reactor, dissolve an aromatic diamine therein, then react an aromatic dianhydride and a cycloaliphatic dianhydride, and then add an aromatic diacid dichloride to perform the reaction.
In addition, another exemplary embodiment of (a) preparing a polyamic acid solution may be to add an organic solvent to a reactor, dissolve an aromatic diamine therein, then perform a reaction with an aromatic diacid dichloride, and then react an aromatic dianhydride and a cycloaliphatic dianhydride. This is more preferred, since the content of the aromatic diacid dichloride in a finally obtained polymer may be increased, that is, polymerization reaction uniformity is excellent in spite of a high solid content, and a high modulus may be implemented, in addition to excellent optical properties.
According to an exemplary embodiment of the present invention, when preparing the polyamic acid solution, the reaction is performed by adding the aromatic diamine not altogether, but stepwisely to the organic solvent, for increasing reactivity. In addition, it is preferred to firstly add the aromatic diamine to the organic solvent, and then sufficiently dissolve the aromatic diamine. Here, the organic solvent to be used is as described above, and it is preferred to use dimethyl acetamide or N-methyl-2-pyrrolidone. In addition, the content of the organic solvent may be properly selected considering the molecular weight of polyamideimide which is a copolymerized product derived from the monomers, and may be 80 to 97 wt % in the entire composition. The content is preferably 85 to 95 wt %, more preferably 87 to 95 wt %. That is, the solid content is 3 to 20 wt %, preferably 5 to 15 wt %, and more preferably 5 to 13 wt %.
When the content of the organic solvent is less than 80 wt %, gelation may occur during a polymerization process, a uniform solution may be difficult to be obtained, a solution having a high viscosity out of an available range is formed, so that purification using the solvent may not be easy. As such, when the purification is not done well, optical properties such as a light transmittance and a yellow index may be deteriorated when forming a film. In addition, when the content of the organic solvent is more than 97 wt %, solution formation is possible, but the yield of polyamideimide may be decreased.
Step (a) is performed under an inert gas atmosphere, specifically for example, with nitrogen or argon gas refluxed in the reactor. In addition, a reaction temperature range is from room temperature to 80.degree. C., specifically 20 to 80.degree. C., and a reaction time is 30 minutes to 24 hours, but not limited thereto.
Step (b) of imidization is to imidize the polyamic acid solution prepared in step (a) to obtain polyamideimide, and a known imidization method, for example, a thermal imidization method, a chemical imidization method, and a combination of a thermal imidization method and a chemical imidization method may be applied. It is preferred to subject the solution to chemical imidization, but not limited thereto.
In addition, the imidization may be performed before or after coating a polyamideimide solution, and applied by the known various methods, and thus, is not limited.
In the present invention, the chemical imidization may be performed by further including any one or two or more selected from the group consisting of an imidization catalyst and a dehydrating agent in the prepared polyamic acid solution. It is preferred that the chemical imidization is performed by adding any one or two or more selected from the group consisting of an imidization catalyst and a dehydrating agent to the polyamic acid solution prepared in step (a), in terms of the physical properties of the obtained polyamideimide. More preferably, any one or two or more selected from the group consisting of an imidization catalyst and a dehydrating agent are added to the polyamic acid solution to perform imidization, and then purification using a solvent is performed to obtain a solid content, which is dissolved in the solvent to obtain the polyamideimide solution.
Here, as the dehydrating agent, any one or more selected from the group consisting of acetic anhydride, phthalic anhydride and maleic anhydride may be used, and as the imidization catalyst, any one or more selected from the group consisting of pyridine, isoquinoline and .beta.-quinoline may be used, but not limited thereto.
According to an exemplary embodiment of the present invention, it is preferred to use a solvent which may significantly decrease the solubility of the solid content of polyamideimide, specifically for example, any one or more selected from the group consisting of water and alcohol, as the solvent used in the purification after imidizing the polyamic acid solution.
When the solid content of polyamideimide is obtained by purification, and then dissolved in the organic solvent to obtain imidized polyamideimide, as the organic solvent to be used, a solvent identical to or different from the organic solvent used when preparing the polyamic acid solution may be used. Here, the content of the solvent may be 70 to 95 wt %. The content is preferably 75 to 95 wt %, more preferably 80 to 90 wt %.
In addition, when a viscosity of the polyamideimide solution prepared by dissolving the finally obtained polyamideimide in the solvent is measured, the viscosity may be 5,000 to 500,000 cps, preferably 8,000 to 300,000 cps, more preferably 10,000 to 200,000 cps. Here, the viscosity is measured using a Brookfield viscometer at 25.degree. C.
The polyamideimide obtained in the present invention has a weight average molecular weight of 50,000 to 1,000,000 g/mol, preferably 50,000 to 800,000 g/mol, and more preferably 50,000 to 500,000 g/mol. Here, the weight average molecular weight is measured with polystyrene as a standard sample using 1260 Infinity available from Agilent Technologies, in which PL gel Olexis was used as a column, and 4 mg contained in 100 ml of LiCl at 0.5 wt % with DMAc as a solvent was used as the sample. In addition, the polyamideimide may have a glass transition temperature of 200 to 400.degree. C., preferably 320 to 390.degree. C.
Step (c) of coating a polyamideimide solution in which the polyamideimide is dissolved in the solvent and subjecting the coated solution to heat treatment may be further included. The heat treatment step is to cast the polyamideimide solution on a substrate such as a glass substrate and subject the cast solution to heat treatment to form a film. Here, the term "polyamideimide solution` described in step (c) refers to a coating composition for preparing a polyamideimide film containing polyamideimide.
According to an exemplary embodiment of the present invention, it is preferred that the heat treatment is stepwisely performed as an example. Preferably, the heat treatment may be stepwisely performed at 80 to 100.degree. C. for 1 minute to 2 hours, at 100 to 200.degree. C. for 1 minute to 2 hours, or at 250 to 300.degree. C. for 1 minute to 2 hours. More preferably, the stepwise heat treatment depending on each temperature range is performed for 30 minutes to 2 hours. Here, it is more preferred to perform the stepwise heat treatment by heating in a range of preferably 1 to 20.degree. C./min when moving to each step. In addition, the heat treatment may be performed in a separate vacuum oven, but not necessarily limited thereto.
According to an exemplary embodiment of the present invention, the coating may be performed using an applicator considering a thickness of the prepared film to form the film on the substrate, and the film thickness may be 10 to 100 .mu.m, preferably 20 to 90 .mu.m, but not limited thereto.
According to an exemplary embodiment of the present invention, the polyamideimide film may have a modulus of 5.0 GPa or more, as measured at an extension speed of 25 mm/min using UTM 3365 available from Instron. Specifically, the modulus may be 5.0 to 10 GPa, preferably 5.0 to 8.0 GPa. Here, the modulus may be measured, based on a specimen having a thickness of 55 .mu.m, a length of 50 mm and a width of 10 mm.
In addition, according to an exemplary embodiment of the present invention, the polyamideimide film may have a total light transmittance of 88% or more, as measured in accordance with the ASTM D1003 standard. Preferably the light transmittance may be 89% or more. Specifically, the total light transmittance may be 88 to 99%. Preferably, the total light transmittance may be 89 to 95%. Here, the total light transmittance may be measured, based on a specimen having a thickness of 55 .mu.m.
In addition, according to an exemplary embodiment of the present invention, the polyamideimide film may have a light transmittance of 40% or less, as measured at 388 nm using UV-3600 available from Shimadzu. Preferably, the light transmittance may be 30% or less. Specifically, the light transmittance measured at 388 nm may be 1 to 40%. Preferably, the light transmittance may be 5 to 30%. Here, the light transmittance measured at 388 nm may be measured, based on a specimen having a thickness of 55 .mu.m.
In addition, the polyamideimide film may have a yellow index of 3.00 or less, preferably 2.60 or less, as measured in accordance with the ASTM E313 standard. Specifically, the yellow index may be 1.00 to 3.00, preferably 1.00 to 2.60. Here, the yellow index is measured using a ColorQuest XE measuring device available from HunterLab. In addition, the yellow index may be measured, based on a specimen having a thickness of 55 .mu.m.
As described above, the polyamideimide film capable of implementing excellent physical properties may be derived from the aromatic diamine, the aromatic dianhydride, the cycloaliphatic dianhydride and the aromatic diacid dichloride. Specifically, the film may be provided from polyamideimide derived from the polyamic acid resin wherein the aromatic diamine includes 2,2'-bis(trifluoromethyl)-benzidine, and the aromatic dianhydride includes 4,4'-hexafluoroisopropylidene diphthalic anhydride and biphenyltetracarboxylic dianhydride, the cycloaliphatic dianhydride includes a compound represented by the following Chemical Formula 1, and the aromatic diacid dichloride includes terephthaloyl dichloride.
##STR00007##
wherein
R.sub.1 to R.sub.4 are independently of one another, selected from the group consisting of hydrogen, halogen, a C.sub.1 to C.sub.10 alkyl group or a C.sub.1 to C.sub.10 alkoxy group.
The polyamideimide film prepared by the combination may implement optical properties such as a high total light transmittance and a low yellow index as described above, and also has an excellent modulus, and may decrease a light transmittance in a short wavelength region.
The present invention may manufacture various forms of molded articles using the polyamideimide. As an example, the present invention may be applied to a printed wiring board, a flexible circuit board and the like including a film, a protective film or an insulating film, but not limited thereto. Preferably, the present invention may be applied to a protective film which may replace cover glass, and has a wide application range in various industrial fields including a display.
Hereinafter, the preferred Examples and Comparative Examples of the present invention will be described. However, the following Examples are only a preferred exemplary embodiment, and the present invention is not limited thereto.
The physical properties of the present invention were measured as follows:
(1) Light Transmittance (Unit: %)
Total light transmittance of the films prepared in the Examples and the Comparative Examples were measured using COH-400 available from Nippon Denshoku in accordance with the ASTM D1003 standard. The light transmittance at 388 nm of the films prepared in the Examples and the Comparative Examples were measured using UV-3600 available from Shimadzu.
(2) Yellow Index
The yellow index of the films prepared in the Examples and the Comparative Examples was measured in accordance with the ASTM E313 standard using ColorQuest XE (Mode type: Total transmission, Area view: 0.375 in., UV filter: Nominal) available from HunterLab.
(3) Modulus
The modulus of the films having a length of 50 mm and a width of 10 mm prepared in the Examples and the Comparative Examples was measured using UTM 3365 available from Instron, under the condition of pulling at 25 mm/min at 25.degree. C.
(4) Viscosity
The viscosity was measured using a Brookfield viscometer (Dv2TRV-cone&plate, CPA-52Z) at 25.degree. C.
(5) Molecular Weight
The weight average molecular weight was measured using 1260 Infinity available from Agilent Technologies, with polystyrene as a standard sample, in which PL gel Olexis was used as the column, and 4 mg contained in 100 ml of LiCl at a concentration of 0.5 wt % with DMAc as a solvent was used as the sample.
Example 1
Under a nitrogen atmosphere, methylene chloride and 2,2'-bis(trifluoromethyl)-benzidine (TFMB) were added to a reactor and sufficiently stirred, and then terephthaloyl dichloride (TPC) was added thereto and stirred for 6 hours to be dissolved and reacted. Thereafter, an excessive amount of methanol was used for precipitation and filtration to obtain a reaction product, which was dried under vacuum at 50.degree. C. for 6 hours or more, and added again with DMAc to the reactor under the nitrogen atmosphere, and 4,4'-hexafluoroisopropyllidene diphthalic anhydride (6FDA) was added thereto and sufficiently stirred until dissolved, and then biphenyltetracarboxylic dianhydride (BPDA) was added and stirred until dissolved, and then cyclobutanetetracarboxylic dianhydride (CBDA) was added and stirred until dissolved. Subsequently, pyridine and acetic anhydride were added to the solution at 2.5-fold molar amount of the total added amount of dianhydrides, and stirred at 60.degree. C. for 1 hour. Here, the amount of each monomer was such that the mole ratio of TFMB:BPDA:CBDA:6FDA:TPC was 100:10:20:15:55, and the solution was adjusted to have a solid content of 12 wt %. The viscosity of the finally obtained polyamideimide was 33,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
The obtained solution was solution-cast on a glass substrate using an applicator. Thereafter, heat treatment was performed in a vacuum oven at 100.degree. C. for 30 minutes, at 200.degree. C. for 30 minutes and at 300.degree. C. for 30 minutes, and then cooling was performed at room temperature, and the film formed on the glass substrate was separated from the substrate to obtain a polyamideimide film having a thickness of 55 .mu.m. The result of measuring the weight average molecular weight of the film was 205,000 g/mol.
Example 2
A polyamideimide film having a thickness of 56 .mu.m was prepared in the same manner as in Example 1, except that the mole ratio of TFMB:BPDA:CBDA:6FDA:TPC was 100:20:10:15:55. The weight average molecular weight of the prepared polyamideimide was 195,000 g/mol, and the viscosity of the finally obtained polyamideimide was 27,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
Example 3
A polyamideimide film having a thickness of 56 .mu.m was prepared in the same manner as in Example 1, except that the mole ratio of TFMB:BPDA:CBDA:6FDA:TPC was 100:10:5:15:70. The weight average molecular weight of the prepared polyamideimide was 210,000 g/mol, and the viscosity of the finally obtained polyamideimide was 30,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
Example 4
A polyamideimide film having a thickness of 55 .mu.m was prepared in the same manner as in Example 1, except that the mole ratio of TFMB:BPDA:CBDA:6FDA:TPC was 100:15:10:15:60. The weight average molecular weight of the prepared polyamideimide was 190,000 g/mol, and the viscosity of the finally obtained polyamideimide was 28,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
Example 5
A polyamideimide film having a thickness of 60 .mu.m was prepared in the same manner as in Example 1, except that biphenyltetracarboxylic dianhydride (BPDA) was not used, and the compound represented by the following Chemical Formula 2 was further included, so that the mole ratio of TFMB:TBIS-MPN:CBDA:6FDA:TPC was 100:5:25:15:55. The weight average molecular weight of the prepared polyamideimide was 195,000 g/mol, and the viscosity of the finally obtained polyamideimide was 28,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
##STR00008##
Example 6
A polyamideimide film having a thickness of 58 .mu.m was prepared in the same manner as in Example 1, except that biphenyltetracarboxylic dianhydride (BPDA) was not used, and the compound represented by the following Chemical Formula 3 was further included, so that the mole ratio of TFMB:TBIS-BAN:CBDA:6FDA:TPC was 100:5:25:15:55. The weight average molecular weight of the prepared polyamideimide was 170,000 g/mol, and the viscosity of the finally obtained polyamideimide was 21,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
##STR00009##
Example 7
A polyamideimide film having a thickness of 50 .mu.m was prepared in the same manner as in Example 1, except that biphenyltetracarboxylic dianhydride (BPDA) was not used, the mole ratio of TFMB:CBDA:6FDA:TPC was 100:30:15:55, and 0.15 wt % of a UV additive (TBO, 2,5-Bis(5-tert-butyl-2-benzoxazolyl)thiophene) was further included, based on the total amount of the composition. The weight average molecular weight of the prepared polyamideimide was 220,000 g/mol, and the viscosity of the finally obtained polyamideimide was 58,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
Comparative Example 1
A polyamideimide film having a thickness of 50 .mu.m was prepared in the same manner as in Example 1, except that biphenyltetracarboxylic dianhydride (BPDA) was not used, and the mole ratio of TFMB:CBDA:6FDA:TPC was 100:20:25:55. The weight average molecular weight of the prepared polyamideimide was 190,000 g/mol, and the viscosity of the finally obtained polyamideimide was 35,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
Comparative Example 2
A polyamideimide film having a thickness of 58 .mu.m was prepared in the same manner as in Example 1, except that cyclobutanetetracarboxylic dianhydride (CBDA) was not used, and the mole ratio of TFMB:BPDA:6FDA:TPC was 100:20:25:55. The weight average molecular weight of the prepared polyamideimide was 180,000 g/mol, and the viscosity of the finally obtained polyamideimide was 32,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
Comparative Example 3
The process was performed in the same manner as in Example 1, except that 4,4'-hexafluoroisopropylidene diphthalic anhydride (6FDA) was not used, and the mole ratio of TFMB:BPDA:CBDA:TPC was 100:20:20:60, however, the solution was hardened in a cloudy opaque state, so that the viscosity and the weight average molecular weight was not able to be measured.
Comparative Example 4
A polyamideimide film having a thickness of 55 .mu.m was prepared in the same manner as in Example 1, except that biphenyltetracarboxylic dianhydride (BPDA) and cyclobutanetetracarboxylic dianhydride (CBDA) were not used, and the mole ratio of TFMB:6FDA:TPC was 100:30:70. The weight average molecular weight of the prepared polyamideimide was 150,000 g/mol, and the viscosity of the finally obtained polyamideimide was 15,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
Comparative Example 5
A polyamideimide film having a thickness of 59 .mu.m was prepared in the same manner as in Example 1, except that biphenyltetracarboxylic dianhydride (BPDA) was not used, and the compound represented by the following Chemical Formula 4 was further included, so that the mole ratio of TFMB:BPAF:CBDA:6FDA:TPC was 100:10:20:15:55. The weight average molecular weight of the prepared polyamideimide was 160,000 g/mol, and the viscosity of the finally obtained polyamideimide was 18,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
##STR00010##
Comparative Example 6
A polyamideimide film having a thickness of 52 .mu.m was prepared in the same manner as in Example 1, except that biphenyltetracarboxylic dianhydride (BPDA) was not used, and aromatic 3,4-oxydianiline (3,4-ODA) was included, so that the mole ratio of TFMB:3,4-ODA:CBDA:6FDA:TPC was 90:10:30:15:55. The weight average molecular weight of the prepared polyamideimide was 195,000 g/mol, and the viscosity of the finally obtained polyamideimide was 30,000 cps, as measured using a Brookfield viscometer at 25.degree. C.
TABLE-US-00001 TABLE 1 Compositional ratio (mole ratio) TFMB 3,4-ODA BPDA BPAF TBIS-MPN TBIS-BAN CBDA 6FDA TPC Example 1 100 -- 10 -- -- -- 20 15 55 Example 2 100 -- 20 -- -- -- 10 15 55 Example 3 100 -- 10 -- -- -- 5 15 70 Example 4 100 -- 15 -- -- -- 10 15 60 Example 5 100 -- -- -- 5 -- 25 15 55 Example 6 100 -- -- -- -- 5 25 15 55 Example 7 100 -- -- -- -- -- 30 15 55 Comparative 100 -- -- -- -- -- 20 25 55 Example 1 Comparative 100 -- 20 -- -- -- -- 25 55 Example 2 Comparative 100 -- 20 -- -- -- 20 -- 60 Example 3 Comparative 100 -- -- -- -- -- -- 30 70 Example 4 Comparative 100 -- -- 10 -- -- 20 15 55 Example 5 Comparative 90 10 -- -- -- -- 30 15 55 Example 6 Light UV Total light transmittance Yellow additive (wt %) Thickness (.mu.m) transmittance (%) at 388 nm (%) index Modulus (GPa) Example 1 -- 55 89.2 28.1 2.6 5.3 Example 2 -- 56 89.0 11.8 2.9 5.1 Example 3 -- 56 89.2 26.7 2.7 5.2 Example 4 -- 55 89.0 17.5 2.5 5.0 Example 5 -- 60 90.0 36.0 3.9 4.2 Example 6 -- 58 89.5 1.5 14.4 4.3 Example 7 0.15 50 89.7 12.0 4.3 5.2 Comparative -- 50 89.8 69.5 2.4 4.8 Example 1 Comparative -- 58 89.2 11.3 3.6 4.5 Example 2 Comparative -- -- -- -- -- -- Example 3 Comparative -- 55 90.0 70.2 3.2 4.5 Example 4 Comparative -- 59 90.0 58.0 2.6 4.3 Example 5 Comparative -- 52 83.93 0.7 18.7 4.8 Example 6
As shown in Table 1, it was confirmed that the Examples according to the present invention had an excellent total light transmittance, a low yellow index, and a high modulus. In particular, it was confirmed that the light transmittance measured at 388 nm was low. In particular, preferably, when the aromatic diamine includes 2,2'-bis(trifluoromethyl)-benzidine, and the aromatic dianhydride includes 4,4'-hexafluoroisopropylidene diphthalic anhydride and biphenyltetracarboxylic dianhydride, the cycloaliphatic dianhydride includes cyclobutanetetracarboxylic dianhydride (CBDA), and the aromatic diacid dichloride includes terephthaloyl dichloride, all of the above-described physical properties may be well-implemented.
However, Comparative Example 1 had a high light transmittance in a short wavelength region which was measured at 388 nm, and a low modulus, as compared with the Examples. In addition, Comparative Example 2 had a high yellow index, and Comparative Example 4 had a high light transmittance in a short wavelength region which was measured at 388 nm, and a low modulus, as compared with the Examples. In addition, Comparative Examples 5 and 6 had a high light transmittance in a short wavelength region which was measured at 388 nm, or a very high yellow index, and a low modulus, as compared with the Examples.
The polyamideimide film according to the present invention may have excellent mechanical physical properties, simultaneously with a low yellow index and an excellent light transmittance in an entire wavelength region of visible light, and implement a low light transmittance in a short wavelength region.
In particular, as the polyamideimide film may implement a high modulus, and have a low light transmittance in a short wavelength region, ultraviolet ray-induced damage by ultraviolet ray exposure to a lower structure of a display including the polyamideimide film may be prevented, and thus, the polyamideimide film may be applied to various display fields.
Simultaneously, the polyamideimide film may significantly improve a modulus, and implement excellent mechanical strength, thereby being applied various fields including a display.
Hereinabove, although the present invention has been described by the specific matters and specific exemplary embodiments, they have been provided only for assisting in the entire understanding of the present invention. Therefore, the present invention is not limited to the exemplary embodiments, and various modifications and changes may be made by those skilled in the art to which the present invention pertains from this description.
Therefore, the spirit of the present invention should not be limited to the above-described exemplary embodiments, and the following claims as well as all modified equally or equivalently to the claims are intended to fall within the scope and spirit of the invention.
* * * * *
C00001

C00002

C00003

C00004

C00005

C00006

C00007

C00008

C00009

C00010

C00011

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.