Crystalline metallophosphates, their method of preparation, and use
Yuhas , et al.
U.S. patent number 10,662,069 [Application Number 16/015,115] was granted by the patent office on 2020-05-26 for crystalline metallophosphates, their method of preparation, and use. The grantee listed for this patent is UOP LLC. Invention is credited to Mark A. Miller, John P. S. Mowat, Mimoza Sylejmani-Rekaliu, Kristine N. Wilson, Benjamin D. Yuhas.
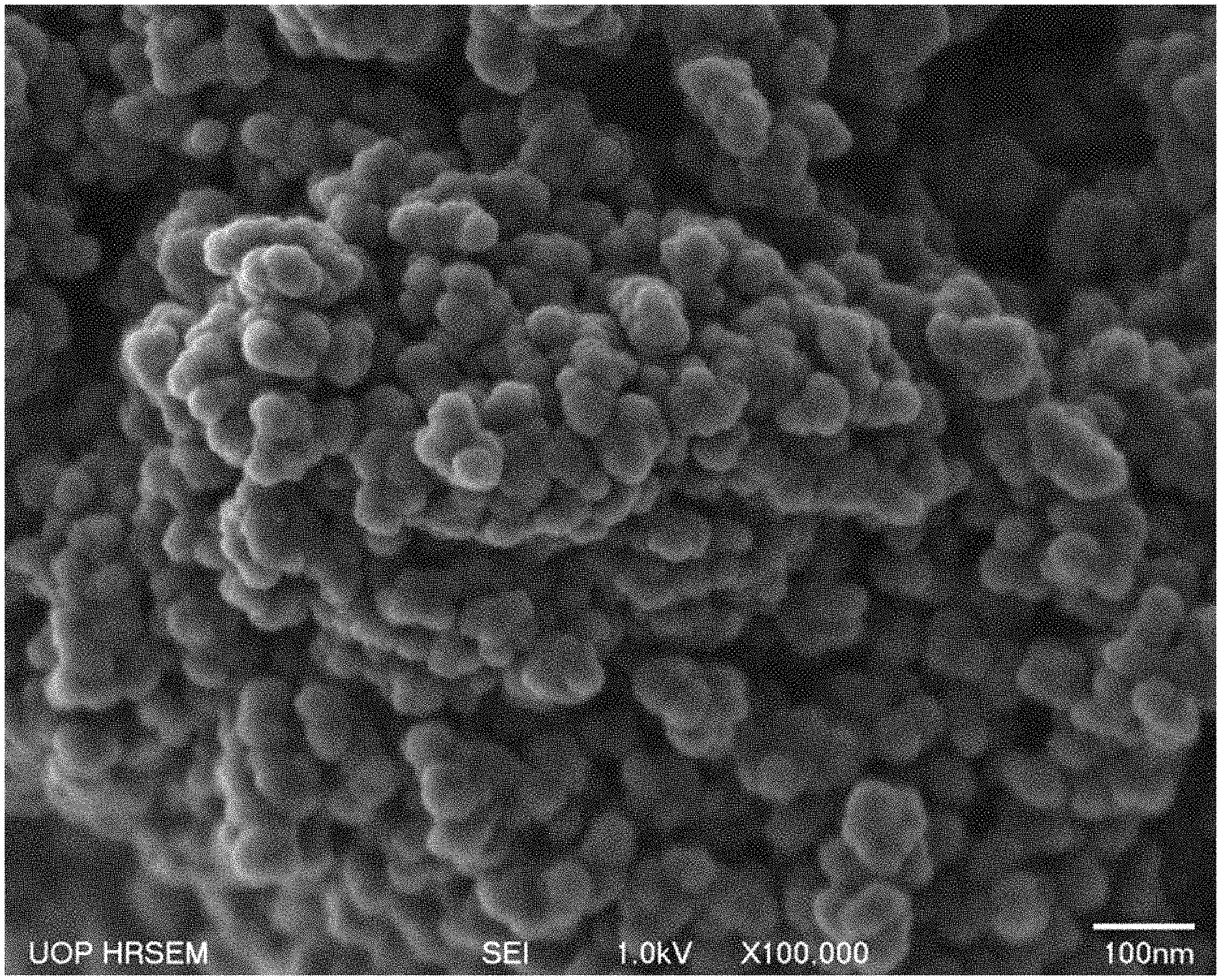

| United States Patent | 10,662,069 |
| Yuhas , et al. | May 26, 2020 |
Crystalline metallophosphates, their method of preparation, and use
Abstract
A new family of crystalline microporous metallophosphates designated AlPO-90 has been synthesized. These metallophosphates are represented by the empirical formula R.sup.+.sub.rM.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where R is an organoammonium cation, M is a framework metal alkaline earth or transition metal of valence +2, and E is a trivalent framework element such as aluminum or gallium. The AlPO-90 compositions are characterized by a new unique ABC-6 net structure, and have catalytic properties suitable for carrying out various hydrocarbon conversion processes, as well as characteristics suitable for the efficient adsorption of water vapor in a variety of applications, such as adsorption heat pumps.
| Inventors: | Yuhas; Benjamin D. (Evanston, IL), Wilson; Kristine N. (Elgin, IL), Miller; Mark A. (Niles, IL), Sylejmani-Rekaliu; Mimoza (Bensenville, IL), Mowat; John P. S. (Arlington Heights, IL) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Family ID: | 68980555 | ||||||||||
| Appl. No.: | 16/015,115 | ||||||||||
| Filed: | June 21, 2018 |
Prior Publication Data
| Document Identifier | Publication Date | |
|---|---|---|
| US 20190389733 A1 | Dec 26, 2019 | |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | B01J 20/3085 (20130101); B01J 23/8913 (20130101); B01J 37/0018 (20130101); B01J 37/031 (20130101); B01J 29/85 (20130101); C01B 39/54 (20130101); B01J 37/036 (20130101); B01D 53/261 (20130101); B01J 37/10 (20130101); B01J 23/74 (20130101); B01J 35/002 (20130101); B01D 53/04 (20130101); B01J 20/18 (20130101); B01D 53/02 (20130101); B01J 29/83 (20130101); B01J 37/04 (20130101); B01J 20/3057 (20130101); Y02P 30/42 (20151101); B01D 2253/116 (20130101); Y02P 20/129 (20151101); B01D 2257/80 (20130101) |
| Current International Class: | C01B 39/54 (20060101); B01J 29/85 (20060101); B01J 37/04 (20060101); B01J 37/10 (20060101); B01D 53/26 (20060101); B01J 20/30 (20060101); B01J 20/18 (20060101); B01D 53/04 (20060101); B01J 37/00 (20060101) |
References Cited [Referenced By]
U.S. Patent Documents
| 4310440 | January 1982 | Wilson et al. |
| 4440871 | April 1984 | Lok et al. |
| 4567029 | January 1986 | Wilson et al. |
| 4973785 | November 1990 | Lok et al. |
| 5126308 | June 1992 | Barger et al. |
| 7422993 | September 2008 | Takewaki et al. |
| 8323747 | December 2012 | Dunne et al. |
| 8569557 | October 2013 | Lewis et al. |
| 8747807 | June 2014 | Jan et al. |
| 8906225 | September 2014 | Lewis et al. |
| 8911614 | December 2014 | Lewis et al. |
| 8936776 | January 2015 | Lewis |
| 8937776 | January 2015 | Munz |
| 9517942 | December 2016 | Chen et al. |
| 9522896 | December 2016 | Nicholas et al. |
| 2867166 | Mar 2017 | EP | |||
| WO-2017205091 | May 2017 | WO | |||
Other References
|
Smith et al., Enumeration of 4-connected 3-dimensional nets and classification of framework silicates: the infinite set of ABC-6 nets; the Archimedian and .sigma.-related nets; American Mineralogist, vol. 66, pp. 777-788, 1981. cited by applicant . Xie et al., "SSZ-52, a Zeolite with an 18-Layer Aluminosilicate Framework Structure RElated to That of the DeNOx Catalyst Cu-SSZ-13", Journal of the American Chemical Society, 2013, 135, 10519-10524, 2013. cited by applicant . Van Heyden, Kinetics of Water Adsorption in Microporous Aluminophosphate layers for regenerative heat exchangers, Applied Thermal Engineering, 29 (2009) 1514-1522. cited by applicant . Wright et al., Cation-directed syntheses of novel zeolite-like metalloaluminophosphates STA-6 and STA-7 in the presence of azamacrocycle templates, J. Chem. Sc=oc., Dalton Trans., 2000, pp. 1243-1248. cited by applicant . Schreyeck et al., The diaza-polyoxa-macrocycle `Kryptofix222` as a new template for the synth esis of LTA-type-AIPO4 Co-templating role of F and/or (CH3)4N+ ions, Micorporous and Mesoporous Materials 22 (1998) 87-106. cited by applicant . De Lange et al., Adsorption-Driven Heat Pumps: The Potential of Metal-Organic Frameworks, Chem. Rev. 2015, 115, 12205-12250, American Chemical Society. cited by applicant . Meier et al., Zeolite Structure Type EAB: Crystal Structure and Mechanism for the Topotactic Transformation of the Na, TMA Form, Journal of Solid State Chemistry, 37, 204-218 (1981), Academic Press. cited by applicant . Turrina et al., STA-20: An ABC-6 Zeotype Structure Prepared by Co-Templating and Solved via a Hypothetical Structure Database and STEM-ADF Imaging, Chemistry of Materials, ACS Publications, 2017. cited by applicant . Li et al.,Nature Communications, DOI: 10.1038/ncomms9328, 2015, Macmillan Publishers Limited. cited by applicant. |
Primary Examiner: Brooks; Clinton A
Claims
The invention claimed is:
1. A microporous crystalline material that has an empirical composition in an as-synthesized form and on an anhydrous basis expressed by an empirical formula: C.sub.c.sup.+A.sub.a.sup.+M.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one framework divalent cation and is selected from the group consisting of alkaline earth and transition metals, wherein M is a cation selected from the group consisting of beryllium, magnesium, cobalt (II), manganese, zinc, iron(II), nickel and mixtures thereof, C is a cyclic organoammonium cation, A is an acyclic organoammonium cation, the ratio (c/a) having a value from 0.01 to about 100, and the sum (c+a) representing the mole ratio of (C+A) to E and has a value of about 0.1 to about 2.0, "m" is the mole ratio of M to E and varies from 0 to about 1.0, "x" is a mole ratio of P to E and varies from 0.5 to about 2.0, a ratio of silicon to E is represented by "y" which varies from about 0 to about 1.0, E is a trivalent element which is tetrahedrally coordinated, is present in the framework, and is selected from the group consisting of aluminum, gallium, iron(III) and boron and "z" is a mole ratio of O to E and is given by an equation: z=(2m+c+a+3+5x+4y)/2 and is characterized by a following x-ray diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 1: TABLE-US-00018 TABLE 1 2-theta (.degree.) d (.ANG.) Intensity 9.86-9.91 8.96-8.91 w-m 13.97-14.10 6.34-6.28 m 17.21-17.26 5.15-5.13 vw-w 18.79-18.91 4.72-4.68 vw-w 19.78-19.87 4.49-4.46 m-s 22.19-22.33 4.01-3.97 w-m 23.57-23.63 3.78-3.76 w 24.36-24.50 3.66-3.63 vs 27.55-27.61 3.24-3.22 w-m 28.16-28.37 3.17-3.14 m 31.51-31.69 2.84-2.82 w-m 33.26-33.37 2.70-2.68 vw 34.32-34.86 2.62-2.57 w-m 42.59-42.91 2.13-2.10 vw-w 47.70-47.90 1.91-1.89 vw-w 51.92-52.37 1.76-1.74 w-m.
2. The microporous crystalline material of claim 1 wherein after being calcined said AlPO-90 material is characterized by the x-ray diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 2 below: TABLE-US-00019 TABLE 2 2-theta (.degree.) d (.ANG.) Intensity 9.96-10.05 8.87-8.79 w 14.13-14.17 6.27-6.24 vs 19.23-19.32 4.62-4.59 vw-m 20.06-20.10 4.43-4.41 w-m 22.41-22.49 3.97-3.95 vw-m 24.02-24.65 3.71-3.60 m-vs 27.88-27.98 3.20-3.18 w-m 28.38-28.54 3.15-3.12 m 31.89-31.98 2.81-2.79 w 35.05-35.12 2.56-2.55 w 52.60-52.81 1.74-1.73 vw.
3. The microporous crystalline material of claim 1 characterized on an anhydrous basis by the empirical formula: H.sub.wM.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one metal cation of valence+2 selected from the group consisting of Be.sup.2+, Mg.sup.2+, Zn.sup.2+, Co.sup.2+, Mn.sup.2+, Fe.sup.2+, Ni.sup.2+, "m" is the mole ratio of M to E and varies from 0 to about 1.0, H is a proton, w'' is the mole ratio of H to E and varies from 0 to 2.5, E is a trivalent element selected from the group consisting of aluminum, gallium, iron, boron and mixtures thereof, "x" is mole ratio of P to E and varies from 0.5 to about 2.0, "y" is the mole ratio of Si to E and varies from 0.05 to about 1.0, "m"+"y".gtoreq.0.1, and "z" is the mole ratio of O to E and has a value determined by the equation: z=(w+2m+3+5x+4y)/2.
4. The microporous crystalline material of claim 1 that indexes on a unit cell with hexagonal axes with lattice parameters a=12.768 .ANG. and c=15.333 .ANG. and has an ABC-6 net structure with the stacking sequence repeating every 6 layers along the c-axis (p=15.333/2.5=6.13).
5. The microporous crystalline material of claim 4 that can be described as a combination of two zeotypes with stacking sequences of AABCBC and ABACBC.
6. The microporous crystalline material of claim 5 where the percentage of AABCBC zeotype can be defined as x and the percentage of ABACBC zeotype can be defined as (100-x), where x ranges from 0-100, inclusive.
Description
BACKGROUND OF THE INVENTION
This invention relates to a novel family of metallophosphates, collectively designated AlPO-90. They are represented by the empirical formula: C.sub.c.sup.+A.sub.a.sup.+M.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is a divalent framework metal such as magnesium or zinc, C is a cyclic organoammonium cation, A is an acyclic organoammonium cation, and E is a trivalent framework element such as aluminum or gallium.
Classes of molecular sieves include crystalline aluminophosphate, silicoaluminophosphate, or metalloaluminophosphate compositions which are microporous and which are formed from corner sharing AlO.sub.4/2 and PO.sub.4/2 tetrahedra. In 1982, Wilson et al. first reported aluminophosphate molecular sieves, the so-called AlPOs, which are microporous materials that have many of the same properties as zeolites, although they do not contain silica (See U.S. Pat. No. 4,310,440). Subsequently, charge was introduced to the neutral aluminophosphate frameworks via the substitution of SiO.sub.4/2 tetrahedra for PO.sub.4/2.sup.+ tetrahedra to produce the SAPO molecular sieves as described by Lok et al. (See U.S. Pat. No. 4,440,871). Another way to introduce framework charge to neutral aluminophosphates is to substitute [Me.sup.2+O.sub.4/2].sup.2- tetrahedra for AlO.sub.4/2.sup.- tetrahedra, which yields the MeAPO molecular sieves (see U.S. Pat. No. 4,567,029). It is furthermore possible to introduce framework charge on AlPO-based molecular sieves via the simultaneous introduction of SiO.sub.4/2 and [M.sup.2+O.sub.4/2].sup.2- tetrahedra to the framework, giving MeAPSO molecular sieves (See U.S. Pat. No. 4,973,785).
Numerous molecular sieves, both naturally occurring and synthetically prepared, are used in various industrial processes. Synthetically, these molecular sieves are prepared via hydrothermal synthesis employing suitable sources of Si, Al, P, metals, and structure directing agents such as amines or organoammonium cations. The structure directing agents reside in the pores of the molecular sieve and are largely responsible for the particular structure that is ultimately formed. These species may balance the framework charge associated with silicon or other metals such as Zn or Mg in the aluminophosphate compositions, and can also serve as space fillers to stabilize the tetrahedral framework. A particular synthetic scheme utilizes multiple structure-directing agents in the same gel in order to direct the formation of multiple cages or cavities. This has been demonstrated for aluminosilicates, such as UZM-5 (U.S. Pat. No. 8,747,807), as well as silicoaluminophosphates, such as STA-20 (Turrina et al., Chem. Mater., 29, 2180 (2017)).
Molecular sieves are characterized by having pore openings of uniform dimensions, having a significant ion exchange capacity, and being capable of reversibly desorbing an adsorbed phase which is dispersed throughout the internal voids of the crystal without significantly displacing any atoms which make up the permanent molecular sieve crystal structure. Molecular sieves can be used for separation applications, in which certain species of a mixed liquid or vapor stream are captured within the pores of the molecular sieve, and others are excluded. Molecular sieves can also be used as catalysts for hydrocarbon conversion reactions, which can take place on outside surfaces as well as on internal surfaces within the pore.
As stated above, molecular sieves are capable of reversibly adsorbing and desorbing certain molecules depending on the adsorbate's size and the molecular sieve's internal pore structure. There are many applications where it is desired to adsorb water vapor, preferably in a reversible manner. One such application is an adsorption heat pump, which is a device that can be used to recover energy from exhaust or waste heat. As such, adsorption heat pumps can be utilized to maximize energy efficiency in an environmentally friendly manner. Molecular sieves can be useful materials to act as water vapor adsorbents in an adsorption heat pump due to their high capacity for water vapor. A description of the use of adsorbents in adsorption heat pumps can be found in U.S. Pat. No. 8,323,747, incorporated by reference herein in its entirety.
The type of molecular sieves used in adsorption heat pumps must meet certain requirements for optimal performance. A high overall capacity for water vapor is important, but most critically, they should fully desorb all adsorbed water at no greater than 100.degree. C. Otherwise, too much heat must be applied to fully remove the adsorbed water from the micropores (i.e., the regeneration temperature is too high), thus requiring too high of an energy input. The majority of aluminosilicates (i.e., zeolites) have rapid uptake of water vapor at very low pressures (P/P.sub.0), which conversely leads to an unacceptably high regeneration temperature, despite a high overall capacity for water vapor. Aluminophosphates and silicoaluminophosphates have been shown to have more favorable adsorption characteristics for water vapor (see, for example, M. F. de Lange et al. Chem. Rev. 115, 12205 (2015); H. van Heyden et al. Appl. Therm. Eng. 29, 1514 (2009). In particular, the materials SAPO-34 and SAPO-5 (zeotypes CHA and AFI, respectively) have been shown to have particular utility as adsorbent materials in adsorption heat pumps (see U.S. Pat. Nos. 7,422,993, 9,517,942).
SUMMARY OF THE INVENTION
As stated, the present invention relates to a new family of metallophosphate molecular sieves, collectively designated AlPO-90. Accordingly, one embodiment of the invention is a microporous crystalline material having a three-dimensional framework of at least EO.sub.4/2 and PO.sub.4/2.sup.+ tetrahedral units and optionally, at least one of [M.sup.2+O.sub.4/2].sup.2- and SiO.sub.4/2 tetrahedral units and an empirical composition in the as-synthesized form and anhydrous basis expressed by an empirical formula of: C.sub.c.sup.+A.sub.a.sup.+M.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one metal cation of valence +2 selected from the group consisting of Be.sup.2+, Mg.sup.2+, Zn.sup.2+, Co.sup.2+, Mn.sup.2+, Fe.sup.2+, N.sup.2+, "m" is the mole ratio of M to E and varies from 0 to about 1.0, C is a cyclic organoammonium cation, and A is an acyclic organoammonium cation. The ratio c/a can have a value from 0.01 to about 100, and the sum (c+a) represents the mole ratio of (C+A) to E and has a value of about 0.1 to about 2.0. E is a trivalent element selected from the group consisting of aluminum, gallium, iron, boron and mixtures thereof, "x" is mole ratio of P to E and varies from 0.5 to about 2.0, "y" is the mole ratio of Si to E and varies from 0 to about 1.0, and "z" is the mole ratio of 0 to E and has a value determined by the equation: z=(2m+c+a+3+5x+4y)/2
The invention is characterized in that it has the x-ray diffraction pattern having at least the d-spacings and intensities set forth in Table 1:
TABLE-US-00001 TABLE 1 2-theta (.degree.) d (.ANG.) Intensity 9.86-9.91 8.96-8.91 w-m 13.97-14.10 6.34-6.28 m 17.21-17.26 5.15-5.13 vw-w 18.79-18.91 4.72-4.68 vw-w 19.78-19.87 4.49-4.46 m-s 22.19-22.33 4.01-3.97 w-m 23.57-23.63 3.78-3.76 w 24.36-24.50 3.66-3.63 vs 27.55-27.61 3.24-3.22 w-m 28.16-28.37 3.17-3.14 m 31.51-31.69 2.84-2.82 w-m 33.26-33.37 2.70-2.68 vw 34.32-34.86 2.62-2.57 w-m 42.59-42.91 2.13-2.10 vw-w 47.70-47.90 1.91-1.89 vw-w 51.92-52.37 1.76-1.74 w-m
Another embodiment of the invention is a microporous crystalline material having a three-dimensional framework of at least EO.sub.4/2.sup.- and PO.sub.4/2.sup.+ tetrahedral units and optionally, at least one of [M.sup.2+O.sub.4/2].sup.2- and SiO.sub.4/2 tetrahedral units and an empirical composition in the calcined form and anhydrous basis expressed by an empirical formula of: H.sub.wM.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z
where "m", "x", "y" are as described above, H is a proton, "w" is the mole ratio of H to E and varies from 0 to 2.5, and "z" is the mole ratio of 0 to E and has a value determined by the equation: z=(w+2m+3+5x+4y)/2 and the invention is characterized in that it has the x-ray diffraction pattern having at least the d-spacings and intensities set forth in Table 2:
TABLE-US-00002 TABLE 2 2-theta (.degree.) d (.ANG.) Intensity 9.96-10.05 8.87-8.79 w 14.13-14.17 6.27-6.24 vs 19.23-19.32 4.62-4.59 vw-m 20.06-20.10 4.43-4.41 w-m 22.41-22.49 3.97-3.95 vw-m 24.02-24.65 3.71-3.60 m-vs 27.88-27.98 3.20-3.18 w-m 28.38-28.54 3.15-3.12 m 31.89-31.98 2.81-2.79 w 35.05-35.12 2.56-2.55 w 52.60-52.81 1.74-1.73 vw
Another embodiment of the invention is a process for preparing the crystalline microporous metallophosphate molecular sieve described above. The process comprises forming a reaction mixture containing reactive sources of C, A, E, P, one or both of M and Si, and heating the reaction mixture at a temperature of about 60.degree. C. to about 200.degree. C. for a time sufficient to form the molecular sieve, the reaction mixture having a composition expressed in terms of mole ratios of the oxides of: aA.sub.2O:bC.sub.2O:cMO:E.sub.2O.sub.3:dP.sub.2O.sub.5:eSiO.sub.2:fH.sub.- 2O where "a" has a value of about 0.01 to about 5, "b" has a value of about 0.01 to about 5, "c" has a value of about 0 to about 2, "d" has a value of about 0.5 to about 8, "e" has a value of about 0 to about 4, and "f" has a value from 30 to 1000.
Yet another embodiment of the invention is a hydrocarbon conversion process using the above-described molecular sieve as a catalyst. The process comprises contacting at least one hydrocarbon with the molecular sieve at conversion conditions to generate at least one converted hydrocarbon.
Still another embodiment of the invention is an adsorption process using the crystalline AlPO-90 material. The process may involve the adsorption and desorption of water vapor over AlPO-90 in an adsorption heat pump-type apparatus. Separation of molecular species can be based either on the molecular size (kinetic diameter) or on the degree of polarity of the molecular species. Removing contaminants may be by ion exchange with the molecular sieve.
BRIEF DESCRIPTION OF THE DRAWINGS
FIG. 1 is a scanning electron microscope (SEM) image of an exemplary AlPO-90 material according to an embodiment described herein.
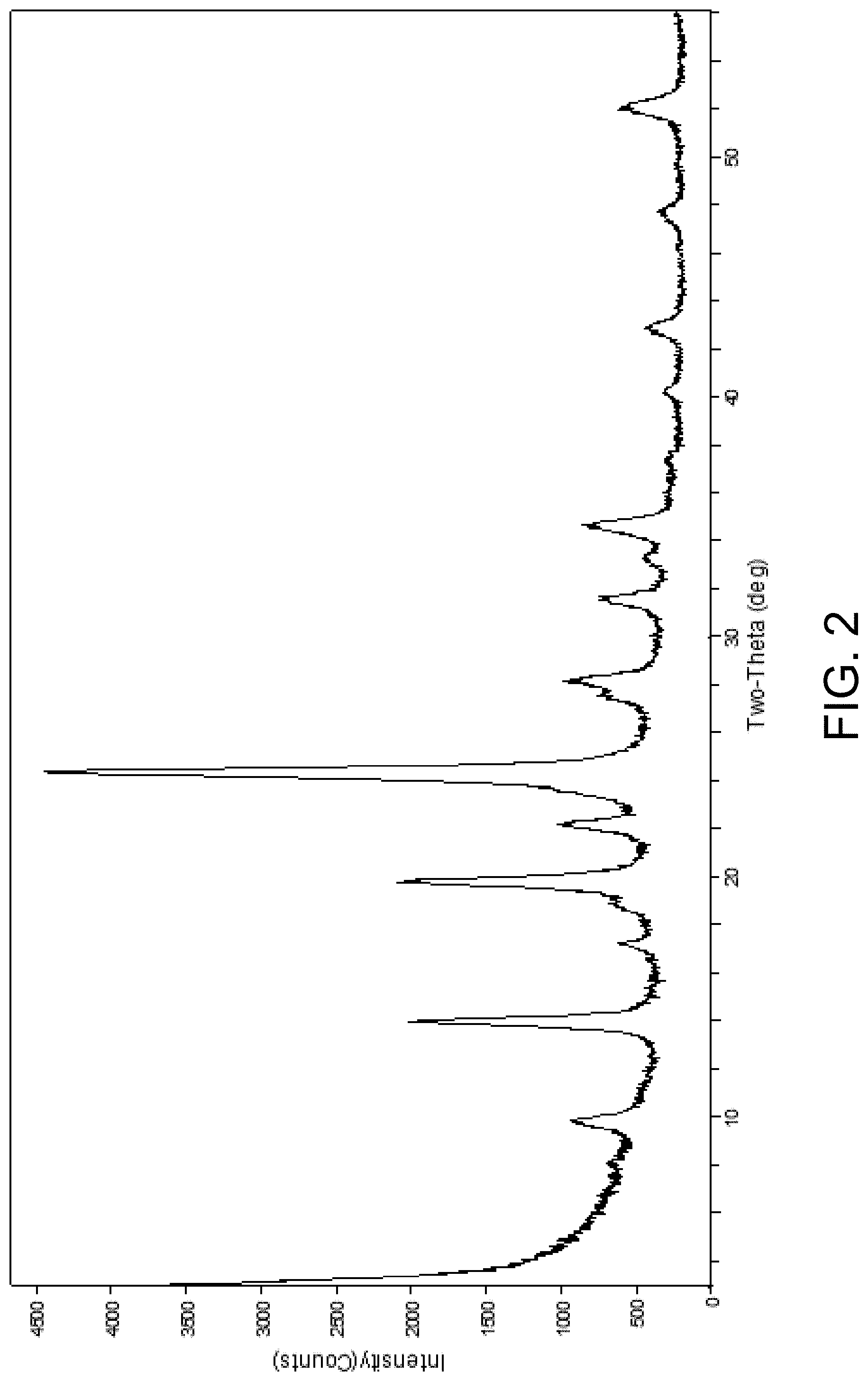
FIG. 2 is an x-ray diffraction pattern of an exemplary AlPO-90 material in the as-synthesized form.
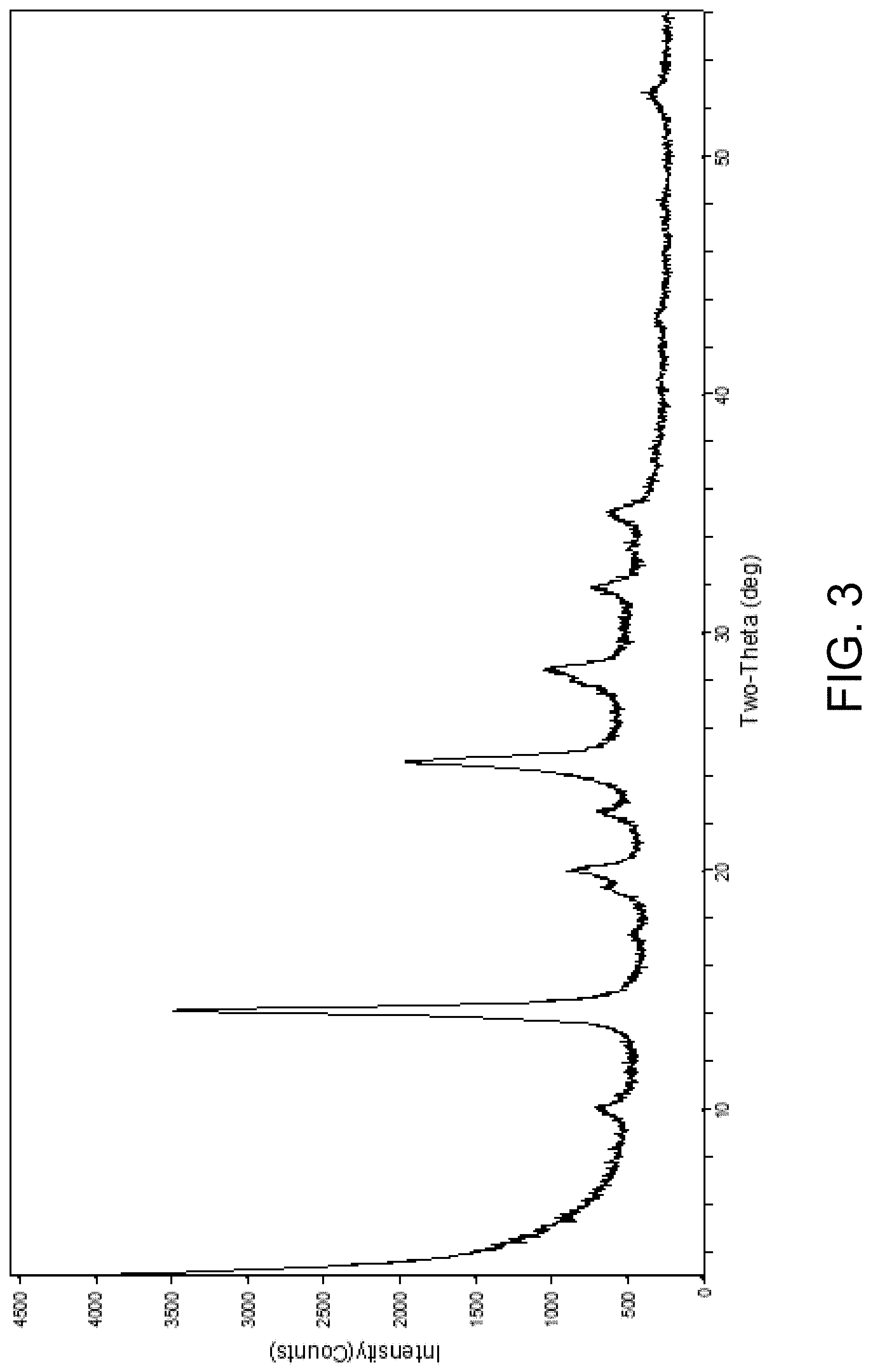
FIG. 3 is an x-ray diffraction pattern of an exemplary AlPO-90 material in the calcined form.
DETAILED DESCRIPTION OF THE INVENTION
Applicants have prepared a family of metallophosphate materials whose topological structure is unique. In their paper "Enumeration of 4-connected 3-dimensional nets and classification of framework silicates: the infinite set of ABC-6 nets; the Archimedean and .sigma.-related nets," Smith and Bennett state "To a first approximation, all silicates belonging to the ABC-6net family have x-ray diffraction patterns which can be indexed on a hexagonal prismatic unit cell with lattice parameters a .about.13.0.+-.0.3 .ANG. and c.about.p.times.(2.6.+-.0.1 .ANG.)." (See American Mineralogist, 66, 777-788 (1981)). This finding has subsequently been confirmed by others (see, for example, D. Xie et al. J. Am. Chem. Soc. 135, 10519 (2013)) as the ABC-6 family has expanded.
One particular composition of AlPO-90 indexes on a unit cell with hexagonal axes with lattice parameters a=12.559 .ANG. and c=15.333 .ANG., which is suggests an ABC-6 net structure with the stacking sequence repeating every 6 layers along the c-axis (p=15.333/2.5=6.13). In the prior art, Meier and Groner enumerated the 10 possible stacking sequences for a 6-layer molecular sieve with hexagonal symmetry in 1981 (J. Solid State Chem., 37, 204 (1981)). This finding was later confirmed by Li et al. in 2015 (Nat. Commun. 2015, 6, 8328). At the time of these publications, only 4 of the 10 possible 6-layer stacking sequences had been experimentally realized in the prior art (zeotypes CHA, ERI, LIO, and EAB). Because there are many potential stacking sequences possible for a given ABC-6 material, the fact that a material has similar lattice parameters to a known material in the prior art does not automatically imply that the two materials are identical. Through a combination of x-ray diffraction experimental techniques as well as modeling, applicants have determined that AlPO-90 can be described as a combination of two novel zeotypes, which have stacking sequences of AABCBC and ABACBC. Although these zeotypes have been theoretically predicted to exist, the instant material AlPO-90, with its unique topological connectivity, represents the first experimental realization of these structures.
The instant microporous crystalline material (AlPO-90) has an empirical composition in the as-synthesized form and on an anhydrous basis expressed by the empirical formula: C.sub.c.sup.+A.sub.a.sup.+M.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one framework divalent cation and is selected from the group consisting of alkaline earth and transition metals. Specific examples of the M cations include but are not limited to beryllium, magnesium, cobalt (II), manganese, zinc, iron(II), nickel and mixtures thereof. C is a cyclic organoammonium cation, and A is an acyclic organoammonium cation. The ratio c/a can have a value from 0.01 to about 100, and the sum (c+a) represents the mole ratio of (C+A) to E and has a value of about 0.1 to about 2.0. The value of "m" is the mole ratio of M to E and varies from 0 to about 1.0, "x" is mole ratio of P to E and varies from 0.5 to about 2.0. The ratio of silicon to E is represented by "y" which varies from about 0 to about 1.0. E is a trivalent element which is tetrahedrally coordinated, is present in the framework, and is selected from the group consisting of aluminum, gallium, iron(III) and boron. Lastly, "z" is the mole ratio of 0 to E and is given by the equation: z=(2m+r+3+5x+4y)/2.
Synthesis of molecular sieve materials often relies on the use of organoamino or organoammonium templates known as organic structure directing agents (OSDAs). While simple OSDAs such as tetramethylammonium, tetraethylammonium and tetrapropylammonium are commercially available, oftentimes OSDAs are complicated molecules that are difficult and expensive to synthesize. However, their importance lies in their ability to impart aspects of their structural features to the molecular sieve to yield a desirable pore structure. For example, the use of 1,4,7,10,13,16-hexamethyl-1,4,7,10,13,16-hexaazacyclooctadecane as OSDA has been shown to allow synthesis of STA-7, an aluminophosphate based material of the SAV zeotype (Wright et. al. J. Chem. Soc., Dalton Trans., 2000, 1243-1248); the use of 4,7,13,16,21,24-hexaoxa-1,10-diazabicyclo[8.8.8]hexacosane (`Kryptofix 222`) led to the synthesis of AlPO4-42 (Schreyeck et. al. Micro. Meso. Mater. 1998, 22, 87-106); MAPO-35, a magnesium aluminophosphate material with the LEV topology, is disclosed in U.S. Pat. No. 4,567,029 in which quinuclidine is employed as a structure directing agent; and in U.S. Pat. No. 4,973,785, the MeAPSO composition CoAPSO-35 is disclosed, which contains both cobalt and silicon in the framework in addition to Al and P and uses methylquinuclidine as the structure directing agent.
The art clearly shows that use of complex organoammonium SDAs often results in new molecular sieve materials. However, the synthesis of these complicated organoammonium compounds is quite lengthy and requires many steps, often in an organic solvent, thereby hindering development of the new molecular sieve material. Frequently, even for simple, commercially available OSDAs, the OSDA is the most costly ingredient used in synthesizing molecular sieve materials. Consequently, it would be economically advantageous to synthesize new molecular sieves from either commercially available organoammonium SDAs or SDAs which may be readily synthesized from commercially available starting materials. This has recently been demonstrated in an elegant fashion using simple aqueous chemistry to generate a novel family of organo-1-oxa-4-azonium cyclohexane compounds (U.S. Pat. No. 9,522,896), derived from morpholino-based compounds. The procedures described in U.S. Pat. No. 9,522,896 can be extended to the family of piperidine-based compounds as well. This procedure thereby allows the preparation of SDAs, such as unusual quaternary ammonium salts, from readily available starting reagents in a facile and practical manner. OSDAs prepared by the methods of the present invention are in aqueous solution and do not pose odor and flashpoint concerns. The result is the unprecedented ability to remove the cooling step typically required in the preparation of in-situ zeolite reaction mixtures and to avoid purification steps such as evaporation of organic solvent typically required in ex-situ preparation methods. The obtained organoammonium bromide salt can be ion-exchanged, either by reaction with Ag.sub.2O or by anion exchange resins to yield the hydroxide form of the organoammonium compound, or used as the halogen salt directly. Finally, the resultant organoammonium compound can be used for the synthesis of a zeolite or molecular sieve.
The microporous crystalline metallophosphate AlPO-90 is prepared by a hydrothermal crystallization of a reaction mixture prepared by combining reactive sources of C, A, E, phosphorus, and one or both of M and silicon. A preferred form of the AlPO-90 materials is when E is Al. The sources of aluminum include but are not limited to aluminum alkoxides, precipitated aluminas, aluminum metal, aluminum hydroxide, aluminum salts and alumina sols. Specific examples of aluminum alkoxides include, but are not limited to aluminum ortho sec-butoxide and aluminum ortho isopropoxide. Sources of phosphorus include, but are not limited to, orthophosphoric acid, phosphorus pentoxide, and ammonium dihydrogen phosphate. Sources of silica include but are not limited to tetraethylorthosilicate, colloidal silica, and precipitated silica. Sources of the other E elements include but are not limited to organoammonium borates, boric acid, precipitated gallium oxyhydroxide, gallium sulfate, ferric sulfate, and ferric chloride. Sources of the M metals include the halide salts, nitrate salts, acetate salts, and sulfate salts of the respective alkaline earth and transition metals.
One component of C may be an organoammonium cation prepared from the reaction of an aqueous mixture of a cyclic secondary amine and an organic dihalide. Specific examples of cyclic secondary amines include, without limitation, piperidine, homopiperidine, pyrrolidine, and morpholine. Specific examples of organic dihalides include, without limitation, 1,4-dibromobutane, 1,5-dibromopentane, and 1,6-dibromohexane.
In one embodiment, the cyclic secondary amine is piperidine and the organic dihalide is 1,4-dibromobutane. In another embodiment, the cyclic secondary amine is piperidine and the organic dihalide is 1,4-dibromopentane. In another embodiment, the cyclic secondary amine is piperidine and the organic dihalide is 1,5-dibromopentane. In another embodiment, the cyclic secondary amine is pyrrolidine and the organic dihalide is 1,4-dibromobutane.
One component of A may be a tetraalkylammonium cation, represented as NR.sub.4.sup.+. The R groups are chosen such that is the cation A is acyclic, and there may be multiple distinct R groups on the same quaternary nitrogen. A particular R group may have the formula C.sub.nH.sub.2n+1, where n is a whole number ranging from 1 to 4, inclusive. Non-limiting examples of component A include tetramethylammonium, ethyltrimethylammonium, diethyldimethylammonium, methyltriethylammonium, tetraethylammonium, tetrapropylammonium, propyltrimethylammonium, butyltrimethylammonium, dipropyldimethylammonium, and methylethyldipropylammonium.
The reaction mixture containing reactive sources of the desired components can be described in terms of molar ratios of the oxides by the formula: aA.sub.2O:bC.sub.2O:cMO:E.sub.2O.sub.3:dP.sub.2O.sub.5:eSiO.sub.- 2:fH.sub.2O
where "a" has a value of about 0.01 to about 5, "b" has a value of about 0.01 to about 5, "c" has a value of about 0 to about 2, "d" has a value of about 0.5 to about 8, "e" has a value of about 0 to about 4, and "f" has a value from 30 to 1000. If alkoxides are used, it is preferred to include a distillation or evaporative step to remove the alcohol hydrolysis products.
The reaction mixture is reacted at a temperature of about 60.degree. C. to about 200.degree. C. and preferably from about 125.degree. C. to about 175.degree. C. for a period of about 1 day to about 21 days and preferably for a time of about 2 days to about 10 days in a sealed reaction vessel at autogenous pressure. The reaction vessel may be heated with stirring, heated while tumbling, or heated quiescently. After crystallization is complete, the solid product is isolated from the heterogeneous mixture by means such as filtration or centrifugation, and then washed with deionized water and dried in air at ambient temperature up to about 100.degree. C. AlPO-90 seeds can optionally be added to the reaction mixture in order to accelerate the formation of the desired microporous composition.
The AlPO-90 aluminophosphate-based material, which is obtained from the above-described process, is characterized by the x-ray following diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 1:
TABLE-US-00003 TABLE 1 2-theta (.degree.) d (.ANG.) Intensity 9.86-9.91 8.96-8.91 w-m 13.97-14.10 6.34-6.28 m 17.21-17.26 5.15-5.13 vw-w 18.79-18.91 4.72-4.68 vw-w 19.78-19.87 4.49-4.46 m-s 22.19-22.33 4.01-3.97 w-m 23.57-23.63 3.78-3.76 w 24.36-24.50 3.66-3.63 vs 27.55-27.61 3.24-3.22 w-m 28.16-28.37 3.17-3.14 m 31.51-31.69 2.84-2.82 w-m 33.26-33.37 2.70-2.68 vw 34.32-34.86 2.62-2.57 w-m 42.59-42.91 2.13-2.10 vw-w 47.70-47.90 1.91-1.89 vw-w 51.92-52.37 1.76-1.74 w-m
The AlPO-90 material may be calcined in either air or nitrogen to remove the occluded template. In one embodiment of the invention, the AlPO-90 is calcined at a temperature of at least 550.degree. C. In another embodiment of the invention, the AlPO-90 is calcined at a temperature of at least 600.degree. C. In another embodiment of the invention, the AlPO-90 is calcined at a temperature of at least 650.degree. C. The AlPO-90 is thermally stable upon calcination, and may be characterized by the x-ray diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 2 below:
TABLE-US-00004 TABLE 2 2-theta (.degree.) d (.ANG.) Intensity 9.96-10.05 8.87-8.79 w 14.13-14.17 6.27-6.24 vs 19.23-19.32 4.62-4.59 vw-m 20.06-20.10 4.43-4.41 w-m 22.41-22.49 3.97-3.95 vw-m 24.02-24.65 3.71-3.60 m-vs 27.88-27.98 3.20-3.18 w-m 28.38-28.54 3.15-3.12 m 31.89-31.98 2.81-2.79 w 35.05-35.12 2.56-2.55 w 52.60-52.81 1.74-1.73 vw
The stable calcined AlPO-90 material can be characterized on an anhydrous basis by the empirical formula: H.sub.wM.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one metal cation of valence +2 selected from the group consisting of Be.sup.2+, Mg.sup.2+, Zn.sup.2+, Co.sup.2+, Mn.sup.2+, Fe.sup.2+, Ni.sup.2+, "m" is the mole ratio of M to E and varies from 0 to about 1.0, H is a proton, w" is the mole ratio of H to E and varies from 0 to 2.5, E is a trivalent element selected from the group consisting of aluminum, gallium, iron, boron and mixtures thereof, "x" is mole ratio of P to E and varies from 0.5 to about 2.0, "y" is the mole ratio of Si to E and varies from 0 to about 1.0, and "z" is the mole ratio of 0 to E and has a value determined by the equation: z=(2m+c+a+3+5x+4y)/2
The crystalline AlPO-90 materials of this invention can be used for separating mixtures of molecular species, removing contaminants through ion exchange and catalyzing various hydrocarbon conversion processes. Separation of molecular species can be based either on the molecular size (kinetic diameter) or on the degree of polarity of the molecular species.
The AlPO-90 compositions of this invention can also be used as a catalyst or catalyst support in various hydrocarbon conversion processes. Hydrocarbon conversion processes are well known in the art and include cracking, hydrocracking, alkylation of both aromatics and isoparaffin, isomerization, polymerization, reforming, hydrogenation, dehydrogenation, transalkylation, dealkylation, hydration, dehydration, hydrotreating, hydrodenitrogenation, hydrodesulfurization, methanol to olefins, methanation and syngas shift process. Specific reaction conditions and the types of feeds which can be used in these processes are set forth in U.S. Pat. Nos. 4,310,440, 4,440,871 and 5,126,308, which are incorporated by reference.
The AlPO-90 materials may also be used as a catalyst for the conversion of methanol to olefins. The methanol can be in the liquid or vapor phase with the vapor phase being preferred. Contacting the methanol with the AlPO-90 catalyst can be done in a continuous mode or a batch mode with a continuous mode being preferred. The amount of time that the methanol is in contact with the AlPO-90 catalyst must be sufficient to convert the methanol to the desired light olefin products. When the process is carried out in a batch process, the contact time varies from about 0.001 hrs to about 1 hr and preferably from about 0.01 hr to about 1.0 hr. The longer contact times are used at lower temperatures while shorter times are used at higher temperatures. When the process is carried out in a continuous mode, the Weight Hourly Space Velocity (WHSV) based on methanol can vary from about 1 hr-1 to about 1000 hr-1 and preferably from about 1 hr-1 to about 100 hr-1.
Generally, the process must be carried out at elevated temperatures in order to form light olefins at a fast enough rate. Thus, the process should be carried out at a temperature of about 300.degree. C. to about 600.degree. C., preferably from about 400.degree. C. to about 550.degree. C. and most preferably from about 435.degree. C. to about 525.degree. C. The process may be carried out over a wide range of pressure including autogenous pressure. Thus, the pressure can vary from about 0 kPa (0 psig) to about 1724 kPa (250 psig) and preferably from about 34 kPa (5 psig) to about 345 kPa (50 psig).
Optionally, the methanol feedstock may be diluted with an inert diluent in order to more efficiently convert the methanol to olefins. Examples of the diluents which may be used are helium, argon, nitrogen, carbon monoxide, carbon dioxide, hydrogen, steam, paraffinic hydrocarbons, e.g., methane, aromatic hydrocarbons, e.g., benzene, toluene and mixtures thereof. The amount of diluent used can vary considerably and is usually from about 5 to about 90 mole percent of the feedstock and preferably from about 25 to about 75 mole percent.
The actual configuration of the reaction zone may be any well known catalyst reaction apparatus known in the art. Thus, a single reaction zone or a number of zones arranged in series or parallel may be used. In such reaction zones the methanol feedstock is flowed through a bed containing the AlPO-90 catalyst. When multiple reaction zones are used, one or more AlPO-90 catalysts may be used in series to produce the desired product mixture. Instead of a fixed bed, a dynamic bed system, (e.g., fluidized bed or moving bed), may be used. Such a dynamic system would facilitate any regeneration of the AlPO-90 catalyst that may be required. If regeneration is required, the AlPO-90 catalyst can be continuously introduced as a moving bed to a regeneration zone where it can be regenerated by means such as oxidation in an oxygen containing atmosphere to remove carbonaceous materials.
The AlPO-90 materials of this invention can also be used as an adsorbent for water vapor. The adsorbent may be a component of an adsorption heat pump apparatus. Adsorbents used for adsorption heat pump purposes are desired to have a high capacity for water vapor, as well as a large crystallographic density. The crystallographic density of microporous crystalline materials is conveniently expressed in units of T-atom/1000 .ANG..sup.3. Generally speaking, adsorbents with a low density can be problematic since they would require a large volume of material to take up a given quantity of adsorbate. This can be troublesome if space is limited in the application. It is thus of interest to consider uptake capacity on a volumetric basis as opposed to a gravimetric basis.
The following examples are presented in illustration of this invention and are not intended as undue limitations on the generally broad scope of the invention as set out in the appended claims. The products will be designated with names that contain the suffix "-90" to indicate the "-90" structure and prefix that reflects the compositional nature of the product, such as "SAPO" for a silicoaluminophosphate, "ZnAPO" for a zinc aluminophosphate, and "MgAPSO" for a magnesium silicoaluminophosphate, etc.
The structure of the AlPO-90 compositions of this invention was determined by x-ray analysis. The x-ray patterns presented in the following examples were obtained using standard x-ray powder diffraction techniques. The radiation source was a high-intensity, x-ray tube operated at 45 kV and 35 mA. The diffraction pattern from the copper K-alpha radiation was obtained by appropriate computer based techniques. Flat compressed powder samples were continuously scanned at 2.degree. to 56.degree. (2.theta.). Interplanar spacings (d) in Angstrom units were obtained from the position of the diffraction peaks expressed as .theta. where .theta. is the Bragg angle as observed from digitized data. Intensities were determined from the integrated area of diffraction peaks after subtracting background, "I.sub.o" being the intensity of the strongest line or peak, and "I" being the intensity of each of the other peaks.
As will be understood by those skilled in the art, the determination of the parameter 2.theta. is subject to both human and mechanical error, which in combination can impose an uncertainty of about .+-.0.4.degree. on each reported value of 2.theta.. This uncertainty is, of course, also manifested in the reported values of the d-spacings, which are calculated from the 2.theta. values. This imprecision is general throughout the art and is not sufficient to preclude the differentiation of the present crystalline materials from each other and from the compositions of the prior art. In some of the x-ray patterns reported, the relative intensities of the d-spacings are indicated by the notations vs, s, m, w, and vw which represent very strong, strong, medium, weak, and very weak respectively. In terms of 100.times.I/I.sub.o, the above designations are defined as: vw=0-5; w=5-15; m=15-40: s=40-75 and vs=75-100
In certain instances the purity of a synthesized product may be assessed with reference to its x-ray powder diffraction pattern. Thus, for example, if a sample is stated to be pure, it is intended only that the x-ray pattern of the sample is free of lines attributable to crystalline impurities, not that there are no amorphous materials present.
In order to more fully illustrate the invention, the following examples are set forth. It is to be understood that the examples are only by way of illustration and are not intended as an undue limitation on the broad scope of the invention as set forth in the appended claims.
EXAMPLE 1
468.1 g of water was added to a three-necked 2-liter round bottom flask equipped with a condenser, overhead mixer, thermocouple, and a nitrogen blanket over the top of the condenser. The flask was placed in an ice bath. 261.7 g of 1,4-dibromobutane (99%) was added to the flask. The temperature of the mixture reached 8.degree. C. before 206.4 g of piperidine (99%) was slowly added. The temperature of the mixture was 29.degree. C. after addition of the piperidine, then steadily rose to a peak temperature of 70.degree. C. Once the temperature started dropping from its peak, the cloudy white mixture became clear, and was mixed for an additional 2 hours.
EXAMPLE 2
885 g of the product from Example 1 was added to a three-necked 2 liter round bottom flask equipped with an overhead mixer. 283.9 g of Ag.sub.2O was added to the flask and stirred at room temperature for 1 day. The mixture is grey in color. The mixture was then filtered to remove precipitated silver bromide. The remaining mixture was then analyzed for water content, which showed it was 70.5% water.
EXAMPLE 3
20.11 g of the product from Example 1 was combined with 2.37 g of tetramethylammonium hydroxide (TMAOH; Sigma-Aldrich, 25%). 1.94 g of Al(OH).sub.3 (Pfaltz & Bauer) was then added to the mixture followed by 0.22 g of Ludox AS-40 (Sigma-Aldrich, 40% SiO.sub.2). 5.36 g of phosphoric acid (Fisher; 85%) was then slowly added. The material was then mixed for 30 minutes. The mixture was then transferred to a 45 cc autoclave and held at 170.degree. C. in a tumble oven for 3 days. After cooling to room temperature, the material was isolated via centrifugation and dried at 100.degree. C. overnight. ICP analysis showed a composition of 1.70% Si, 21.2% Al, 24.6% P (weight percent). CHN analysis showed 12.6% C, 2.89% H, 2.45% N (weight percent). XRD analysis of the material gave the following peaks:
TABLE-US-00005 2-Theta d (.ANG.) Intensity 9.8883 8.9377 w 13.9716 6.3334 m 17.2166 5.1463 vw 18.7904 4.7187 w 19.7852 4.4836 s 22.1931 4.0023 w 23.5708 3.7714 w 24.3648 3.6503 vs 27.5523 3.2348 w 28.1685 3.1654 m 31.5141 2.8366 m 33.2654 2.6911 vw 34.6595 2.586 w 40.246 2.239 vw 42.9014 2.1064 w 47.7024 1.905 w 51.9222 1.7596 m
This material was determined to be SAPO-90 by XRD.
EXAMPLE 4
20.26 g of the product from Example 1 was combined with 2.39 g of tetramethylammonium hydroxide (TMAOH; Sigma-Aldrich, 25%). 1.96 g of Al(OH).sub.3 (Pfaltz & Bauer) was then added to the mixture followed by 5.4 g of phosphoric acid (Fisher; 85%) slowly. The material was mixed for 30 minutes. The mixture was then transferred to a 45 cc autoclave and held at 160.degree. C. in a tumble oven for 2 days. After cooling to room temperature, the material was isolated via centrifugation and dried at 100.degree. C. overnight. XRD analysis of the material gave the following peaks:
TABLE-US-00006 2-Theta d (.ANG.) Intensity 9.9092 8.9189 m 14.0904 6.2803 m 17.2562 5.1346 w 18.9086 4.6895 vw 19.8646 4.4659 m 22.3234 3.9792 m 23.6282 3.7624 w 24.4941 3.6313 vs 27.6099 3.2282 m 28.3668 3.1437 m 31.6819 2.8219 w 33.3349 2.6857 vw 34.3206 2.6108 w 34.8282 2.5739 w 42.6232 2.1195 vw 43.1211 2.0961 w 47.8905 1.8979 vw 52.3698 1.7456 w
The product was determined to be AlPO-90 by XRD.
EXAMPLE 5
20.26 g of the product from Example 1 was combined with 2.39 g of tetramethylammonium hydroxide (TMAOH; Sigma-Aldrich, 25%). 1.96 g of Al(OH).sub.3 (Pfaltz & Bauer) was then added to the mixture followed by 5.4 g of phosphoric acid (Fisher; 85%) slowly. The material was mixed for 30 minutes. The mixture was then transferred to a 45 cc autoclave and held at 170.degree. C. in a tumble oven for 2 days. After cooling to room temperature, the material was isolated via centrifugation and dried at 100.degree. C. overnight. XRD analysis of the material gave the following peaks:
TABLE-US-00007 2-Theta d (.ANG.) Intensity 9.8694 8.9548 m 14.0805 6.2847 m 17.2366 5.1404 w 18.8794 4.6966 w 19.8548 4.4681 m 22.2939 3.9844 m 23.6083 3.7655 w 24.4742 3.6342 vs 27.6004 3.2293 m 28.3469 3.1459 m 31.6522 2.8245 w 33.3643 2.6834 vw 34.3506 2.6086 w 34.8576 2.5718 w 42.5946 2.1208 vw 43.0913 2.0975 w 47.82 1.9005 vw 52.3299 1.7469 w
The product was determined to be AlPO-90 by XRD.
EXAMPLE 6
13.34 g of water was combined with 93.89 g of the product from Example 1 followed by 12.66 g of tetramethylammonium hydroxide (TMAOH; Sigma-Aldrich, 25%). 10.37 g of Al(OH).sub.3 (Pfaltz & Bauer) was then added to the mixture followed by 1.17 g of Ludox AS-40 (Sigma-Aldrich, 40% SiO.sub.2) and 28.60 g of phosphoric acid (Fisher; 85%) slowly. The material was mixed for 30 minutes. The mixture was then transferred to a 300 cc stirred autoclave and held at 170.degree. C. for 2 days with a stir rate of 300 rpm. After cooling to room temperature, the material was isolated via centrifugation and dried at 100.degree. C. overnight. ICP analysis showed a composition of 1.66% Si, 20.7% Al, 25.4% P (weight percent). XRD analysis of the material gave the following peaks:
TABLE-US-00008 2-Theta d (.ANG.) Intensity 9.8692 8.955 w 14.0209 6.3113 m 17.2355 5.1407 vw 19.8051 4.4792 m 22.1942 4.0021 w 24.3648 3.6503 vs 27.561 3.2338 w 28.2375 3.1578 m 31.6023 2.8289 w 33.2637 2.6913 vw 34.6887 2.5839 m 40.2936 2.2365 vw 42.7826 2.1119 w 47.7112 1.9046 vw 52.0912 1.7543 w
The product was determined to be SAPO-90 by XRD.
EXAMPLE 7
The product from Example 3 was calcined in air at 600.degree. C. for 4 hours in a muffle furnace. The temperature was ramped up to 600.degree. C. at a rate of 2.degree. C./min. The material was then cooled to room temperature. XRD analysis of the material gave the following peaks:
TABLE-US-00009 2-Theta d (.ANG.) Intensity 10.0499 8.7944 w 14.1305 6.2626 vs 19.2316 4.6114 vw 20.0628 4.4222 w 22.4897 3.9502 vw 24.5834 3.6183 s 27.8886 3.1965 w 28.3884 3.1414 m 31.8925 2.8038 w 35.0573 2.5576 w 52.6388 1.7373 vw
The calcined SAPO-90 product was pressed into a pellet and loaded in a McBain gravimetric balance for adsorption studies. It was observed that the SAPO-90 took up 14.3% water by weight and 3.0% n-butane by weight.
EXAMPLE 8
The product from Example 4 was calcined in air at 650.degree. C. for 8 hours in a muffle furnace. The temperature was ramped up to 650.degree. C. at a rate of 1-2.degree. C./min. The material was then cooled to room temperature. XRD analysis of the material gave the following peaks:
TABLE-US-00010 2-Theta d (.ANG.) Intensity 10.0089 8.8303 w 14.1602 6.2495 vs 17.2659 5.1317 vw 19.3174 4.5911 m 20.0739 4.4198 m 22.4733 3.953 m 24.0265 3.7009 m 24.6434 3.6096 vs 27.9788 3.1864 m 28.5356 3.1255 m 31.9707 2.7971 w 33.6032 2.6648 vw 35.1167 2.5534 w 52.8083 1.7322 vw
EXAMPLE 9
The product from Example 5 was calcined in air at 650.degree. C. for 8 hours in a muffle furnace. The temperature was ramped up to 650.degree. C. at a rate of 1-2.degree. C./min. The material was then cooled to room temperature. XRD analysis of the material gave the following peaks:
TABLE-US-00011 2-Theta d (.ANG.) Intensity 9.9987 8.8393 w 14.1601 6.2495 vs 17.3757 5.0996 vw 19.3271 4.5888 m 20.0938 4.4155 m 22.4628 3.9548 w 24.0262 3.7009 m 24.6534 3.6082 s 27.9588 3.1887 m 28.5061 3.1287 m 31.9908 2.7954 w 33.5239 2.671 vw 35.1566 2.5506 w 52.8279 1.7316 vw
EXAMPLE 10
The product from Example 6 was calcined in air at 650.degree. C. for 8 hours in a muffle furnace. The temperature was ramped up to 650.degree. C. at a rate of 1-2.degree. C./min. The material was then cooled to room temperature. XRD analysis of the material gave the following peaks:
TABLE-US-00012 2-Theta d (.ANG.) Intensity 9.9696 8.865 w 14.1502 6.2539 vs 20.0939 4.4154 w 22.4128 3.9636 vw 24.6037 3.6154 s 27.9481 3.1899 w 28.4562 3.134 m 31.9111 2.8022 w 35.0572 2.5576 w 52.6095 1.7382 vw
COMPARATIVE EXAMPLE 1
9.49 g of water was combined with 12.58 g of tetramethylammonium hydroxide (TMAOH; Sigma-Aldrich, 25%). 2.07 g of Al(OH).sub.3 (Pfaltz & Bauer) was then added to the mixture followed 0.25 g of Ludox AS-40 (Sigma-Aldrich, 40% SiO.sub.2). 5.70 g of phosphoric acid (Fisher; 85%) was then slowly added. The material was mixed for 30 minutes. The mixture was then transferred to a 45 cc autoclave and held at 170.degree. C. in a tumble oven for 3 days. After cooling to room temperature, the material was isolated via centrifugation and dried at 100.degree. C. overnight. XRD analysis of the material showed that it was AlPO-20 (SOD structure).
SPECIFIC EMBODIMENTS
While the following is described in conjunction with specific embodiments, it will be understood that this description is intended to illustrate and not limit the scope of the preceding description and the appended claims.
A first embodiment of the invention is a microporous crystalline material that has an empirical composition in an as-synthesized form and on an anhydrous basis expressed by an empirical formula C.sup.+.sub.cA.sup.+.sub.aM.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one framework divalent cation and is selected from the group consisting of alkaline earth and transition metals, wherein M is a cation selected from the group consisting of beryllium, magnesium, cobalt (II), manganese, zinc, iron(II), nickel and mixtures thereof, C is a cyclic organoammonium cation, A is an acyclic organoammonium cation, the ratio (c/a) having a value from 0.01 to about 100, and the sum (c+a) representing the mole ratio of (C+A) to E and has a value of about 0.1 to about 2.0, "m" is the mole ratio of M to E and varies from 0 to about 1.0, "x" is a mole ratio of P to E and varies from 0.5 to about 2.0, a ratio of silicon to E is represented by "y" which varies from about 0 to about 1.0, E is a trivalent element which is tetrahedrally coordinated, is present in the framework, and is selected from the group consisting of aluminum, gallium, iron(III) and boron and "z" is a mole ratio of 0 to E and is given by an equation z=(2m+c+a+3+5x+4y)/2 and is characterized by a following x-ray diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 1.
TABLE-US-00013 TABLE 1 2-theta (.degree.) d (.ANG.) Intensity 9.86-9.91 8.96-8.91 w-m 13.97-14.10 6.34-6.28 m 17.21-17.26 5.15-5.13 vw-w 18.79-18.91 4.72-4.68 vw-w 19.78-19.87 4.49-4.46 m-s 22.19-22.33 4.01-3.97 w-m 23.57-23.63 3.78-3.76 w 24.36-24.50 3.66-3.63 vs 27.55-27.61 3.24-3.22 w-m 28.16-28.37 3.17-3.14 m 31.51-31.69 2.84-2.82 w-m 33.26-33.37 2.70-2.68 vw 34.32-34.86 2.62-2.57 w-m 42.59-42.91 2.13-2.10 vw-w 47.70-47.90 1.91-1.89 vw-w 51.92-52.37 1.76-1.74 w-m
An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein after being calcined the AlPO-90 material is characterized by the x-ray diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 2 below.
TABLE-US-00014 TABLE 2 2-theta (.degree.) d (.ANG.) Intensity 9.96-10.05 8.87-8.79 w 14.13-14.17 6.27-6.24 vs 19.23-19.32 4.62-4.59 vw-m 20.06-20.10 4.43-4.41 w-m 22.41-22.49 3.97-3.95 vw-m 24.02-24.65 3.71-3.60 m-vs 27.88-27.98 3.20-3.18 w-m 28.38-28.54 3.15-3.12 m 31.89-31.98 2.81-2.79 w 35.05-35.12 2.56-2.55 w 52.60-52.81 1.74-1.73 vw
An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the AlPO-90 material is characterized on an anhydrous basis by the empirical formula H.sub.wM.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one metal cation of valence +2 selected from the group consisting of Be.sup.2+, Mg.sup.2+, Zn.sup.2+, Co.sup.2+, Mn.sup.2+, Fe.sup.2+, Ni.sup.2+, "m" is the mole ratio of M to E and varies from 0 to about 1.0, H is a proton, w" is the mole ratio of H to E and varies from 0 to 2.5, E is a trivalent element selected from the group consisting of aluminum, gallium, iron, boron and mixtures thereof, "x" is mole ratio of P to E and varies from 0.5 to about 2.0, "y" is the mole ratio of Si to E and varies from 0.05 to about 1.0, "m"+"y">0.1, and "z" is the mole ratio of 0 to E and has a value determined by the equation z=(w+2m+3+5x+4y)/2. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the microporous crystalline material indexes on a unit cell with hexagonal axes with lattice parameters a=12.768 .ANG. and c=15.333 .ANG. and has an ABC-6 net structure with the stacking sequence repeating every 6 layers along the c-axis (p=15.333/2.5=6.13). An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the microporous crystalline material can be described as a combination of two zeotypes with stacking sequences of AABCBC and ABACBC. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the microporous crystalline material has a percentage of AABCBC zeotype that can be defined as x and the percentage of ABACBC zeotype can be defined as (100-x), where x ranges from 0-100, inclusive.
A second embodiment of the invention is a method of making a AlPO-90 microporous crystalline material comprising preparing a reaction mixture containing reactive sources described in terms of molar ratios of the oxides by a formula aA.sub.2O bC.sub.2O cMO E.sub.2O.sub.3 dP.sub.2O.sub.5 eSiO.sub.2 fH.sub.2O where "a" has a value of about 0.01 to about 5, "b" has a value of about 0.01 to about 5, "c" has a value of about 0 to about 2, "d" has a value of about 0.5 to about 8, "e" has a value of about 0 to about 4, and "f" has a value from 30 to 1000, wherein reactive sources of C, A, E, phosphorus and one or both M and silicon; reacting the reaction mixture at a temperature from about 60.degree. C. to about 200.degree. C. for a period of about 1 day to about 21 days; and isolating a solid product from a heterogeneous mixture wherein the AlPO-90 microporous material, is characterized by the x-ray following diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 1:
TABLE-US-00015 TABLE 1 2-theta (.degree.) d (.ANG.) Intensity 9.86-9.91 8.96-8.91 w-m 13.97-14.10 6.34-6.28 m 17.21-17.26 5.15-5.13 vw-w 18.79-18.91 4.72-4.68 vw-w 19.78-19.87 4.49-4.46 m-s 22.19-22.33 4.01-3.97 w-m 23.57-23.63 3.78-3.76 w 24.36-24.50 3.66-3.63 vs 27.55-27.61 3.24-3.22 w-m 28.16-28.37 3.17-3.14 m 31.51-31.69 2.84-2.82 w-m 33.26-33.37 2.70-2.68 vw 34.32-34.86 2.62-2.57 w-m 42.59-42.91 2.13-2.10 vw-w 47.70-47.90 1.91-1.89 vw-w 51.92-52.37 1.76-1.74 w-m
An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the AlPO-90 is calcined at a temperature of at least 550.degree. C. and is characterized by the x-ray diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 2 below:
TABLE-US-00016 TABLE 2 2-theta (.degree.) d (.ANG.) Intensity 9.96-10.05 8.87-8.79 w 14.13-14.17 6.27-6.24 vs 19.23-19.32 4.62-4.59 vw-m 20.06-20.10 4.43-4.41 w-m 22.41-22.49 3.97-3.95 vw-m 24.02-24.65 3.71-3.60 m-vs 27.88-27.98 3.20-3.18 w-m 28.38-28.54 3.15-3.12 m 31.89-31.98 2.81-2.79 w 35.05-35.12 2.56-2.55 w 52.60-52.81 1.74-1.73 vw
An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the sources of aluminum are selected from the group consisting of aluminum alkoxides, precipitated aluminas, aluminum metal, aluminum hydroxide, aluminum salts and alumina sols. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein sources of phosphorus are selected from the group consisting of orthophosphoric acid, phosphorus pentoxide, and ammonium dihydrogen phosphate. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein sources of silica are selected from the group consisting of tetraethylorthosilicate, colloidal silica, and precipitated silica. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein sources of E elements are selected from the group consisting of organoammonium borates, boric acid, precipitated gallium oxyhydroxide, gallium sulfate, ferric sulfate, and ferric chloride. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein sources of the M metals are selected from the group consisting of halide salts, nitrate salts, acetate salts, and sulfate salts of the respective alkaline earth and transition metals. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein C is an organoammonium cation prepared from a reaction of an aqueous mixture of a cyclic secondary amine and an organic dihalide. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the cyclic secondary amines are selected from the group consisting of piperidine, homopiperidine, pyrrolidine, and morpholine. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein A is an acyclic organoammonium cation represented as NR.sub.4.sup.+. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the R groups are, independently, aliphatic carbon chains of the formula C.sub.nH.sub.2n+1, where n is a whole number ranging from 1 to 4, inclusive. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the AlPO-90 microporous crystalline material is calcined at a temperature of at least 600.degree. C. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the first embodiment in this paragraph wherein the AlPO-90 microporous crystalline material is calcined at a temperature of at least 650.degree. C.
A third embodiment of the invention is a process of separating mixtures of molecular species, removing contaminants or catalyzing hydrocarbon conversion processes comprising contacting a feed stream with a microporous crystalline material that has an empirical composition in a calcined form and on an anhydrous basis expressed by an empirical formula H.sub.wM.sub.m.sup.2+EP.sub.xSi.sub.yO.sub.z where M is at least one framework divalent cation and is selected from the group consisting of alkaline earth and transition metals, wherein M is a cation selected from the group consisting of beryllium, magnesium, cobalt (II), manganese, zinc, iron(II), nickel and mixtures thereof. H is a proton, w" is the mole ratio of H to E and varies from 0 to 2.5, "m" is the mole ratio of M to E and varies from 0 to about 1.0, "x" is a mole ratio of P to E and varies from 0.5 to about 2.0, a ratio of silicon to E is represented by "y" which varies from about 0 to about 1.0, E is a trivalent element which is tetrahedrally coordinated, is present in the framework, and is selected from the group consisting of aluminum, gallium, iron(III) and boron and "z" is a mole ratio of O to E and is given by an equation z=(2m+r+3+5x+4y)/2 and is characterized by an x-ray following diffraction pattern, having at least the d-spacings and relative intensities set forth in Table 2
TABLE-US-00017 TABLE 2 2-theta (.degree.) d (.ANG.) Intensity 9.96-10.05 8.87-8.79 w 14.13-14.17 6.27-6.24 vs 19.23-19.32 4.62-4.59 vw-m 20.06-20.10 4.43-4.41 w-m 22.41-22.49 3.97-3.95 vw-m 24.02-24.65 3.71-3.60 m-vs 27.88-27.98 3.20-3.18 w-m 28.38-28.54 3.15-3.12 m 31.89-31.98 2.81-2.79 w 35.05-35.12 2.56-2.55 w 52.60-52.81 1.74-1.73 vw
An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the second embodiment in this paragraph wherein the separation of molecular species is in an operation of an adsorption heat pump wherein water vapor is adsorbed by the microporous crystalline material. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the second embodiment in this paragraph wherein the hydrocarbon conversion processes are selected from the group consisting of cracking, hydrocracking, alkylation of both aromatics and isoparaffin, isomerization, polymerization, reforming, hydrogenation, dehydrogenation, transalkylation, dealkylation, hydration, dehydration, hydrotreating, hydrodenitrogenation, hydrodesulfurization, methanol to olefins, methanation and a syngas shift process. An embodiment of the invention is one, any or all of prior embodiments in this paragraph up through the second embodiment in this paragraph wherein the separation of molecular species is based on the molecular size (kinetic diameter) or on the degree of polarity of the molecular species.
* * * * *
D00000
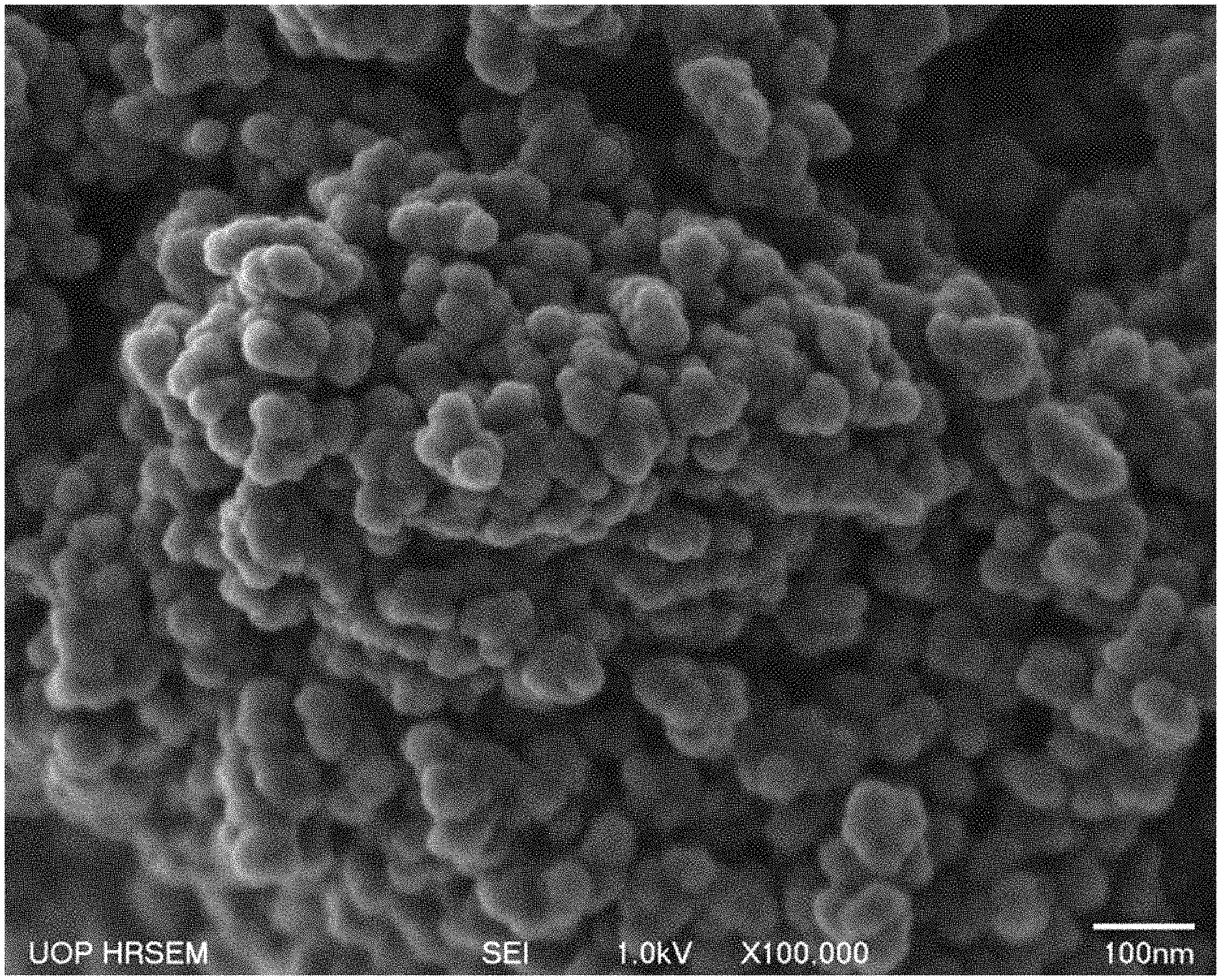
D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.