Antibodies Targeting Cdh19 For Melanoma
Xiao; Shouhua ; et al.
U.S. patent application number 17/326717 was filed with the patent office on 2022-04-28 for antibodies targeting cdh19 for melanoma. The applicant listed for this patent is AMGEN INC.. Invention is credited to Brian Mingtung Chan, M. Shawn Jeffries, Chadwick Terence King, Zheng Pan, Dineli Wickramasinghe, Shouhua Xiao.
| Application Number | 20220127354 17/326717 |
| Document ID | / |
| Family ID | 1000006078522 |
| Filed Date | 2022-04-28 |










View All Diagrams
| United States Patent Application | 20220127354 |
| Kind Code | A1 |
| Xiao; Shouhua ; et al. | April 28, 2022 |
ANTIBODIES TARGETING CDH19 FOR MELANOMA
Abstract
The present disclosure provides a human antibody or antigen binding fragment thereof or an antibody construct comprising a human binding domain or antigen binding fragment thereof capable of binding to human CDH19 on the surface of a target cell. The disclosure relates to a nucleic acid sequence encoding the antibody or antigen binding fragment thereof contained in the antibody construct, a vector comprising the nucleic acid sequence and a host cell transformed or transfected with the vector. Furthermore, the disclosure relates to a process for the production of the antibody construct of the disclosure, a medical use or a method of treatment using the antibody construct and a kit comprising the antibody or antigen binding fragment thereof or the antibody construct.
| Inventors: | Xiao; Shouhua; (Foster City, CA) ; Pan; Zheng; (Fremont, CA) ; Wickramasinghe; Dineli; (San Francisco, CA) ; Jeffries; M. Shawn; (Indianapolis, IN) ; King; Chadwick Terence; (North Vancouver, CA) ; Chan; Brian Mingtung; (Port Coquitlam, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000006078522 | ||||||||||
| Appl. No.: | 17/326717 | ||||||||||
| Filed: | May 21, 2021 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15878047 | Jan 23, 2018 | 11053311 | ||
| 17326717 | ||||
| 14762053 | Jul 20, 2015 | 9920121 | ||
| PCT/EP2014/051551 | Jan 27, 2014 | |||
| 15878047 | ||||
| 61756977 | Jan 25, 2013 | |||
| 61785119 | Mar 14, 2013 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 16/3053 20130101; A61K 47/6803 20170801; A61K 47/6849 20170801; C07K 16/28 20130101; C07K 2317/34 20130101; C07K 2317/33 20130101; C07K 2317/92 20130101; C07K 2317/21 20130101; C07K 2317/73 20130101; C07K 2317/77 20130101; A61K 2039/505 20130101; A61K 47/6865 20170801; C07K 2317/55 20130101 |
| International Class: | C07K 16/28 20060101 C07K016/28; A61K 47/68 20060101 A61K047/68; C07K 16/30 20060101 C07K016/30 |
Claims
1. An isolated human antibody or antigen binding fragment thereof capable of binding to human cadherin 19 (CDH19) on the surface of a target cell, comprising a VH region comprising CDR-H1, CDR-H2 and CDR-H3 and a VL region comprising CDR-L1, CDR-L2 and CDR-L3 selected from the group consisting of: (a) a VH region comprising CDR-H1 as set forth in SEQ ID NO: 94, CDR-H2 as set forth in SEQ ID NO: 95, CDR-H3 as set forth in SEQ ID NO: 96, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 262, CDR-L2 as set forth in SEQ ID NO: 263 and CDR-L3 as set forth in SEQ ID NO: 264, (b) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 100, CDR-H2 as set forth in SEQ ID NO: 101, CDR-H3 as set forth in SEQ ID NO: 102, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 268, CDR-L2 as set forth in SEQ ID NO: 269 and CDR-L3 as set forth in SEQ ID NO: 270, (c) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 118, CDR-H2 as set forth in SEQ ID NO: 119, CDR-H3 as set forth in SEQ ID NO: 120, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 286, CDR-L2 as set forth in SEQ ID NO: 287 and CDR-L3 as set forth in SEQ ID NO: 288, (d) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 154, CDR-H2 as set forth in SEQ ID NO: 155, CDR-H3 as set forth in SEQ ID NO: 156, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 322, CDR-L2 as set forth in SEQ ID NO: 323 and CDR-L3 as set forth in SEQ ID NO: 324, (e) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 100, CDR-H2 as set forth in SEQ ID NO: 101, CDR-H3 as set forth in SEQ ID NO: 912, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 268, CDR-L2 as set forth in SEQ ID NO: 269 and CDR-L3 as set forth in SEQ ID NO: 270, (f) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 100, CDR-H2 as set forth in SEQ ID NO: 101, CDR-H3 as set forth in SEQ ID NO: 913, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 268, CDR-L2 as set forth in SEQ ID NO: 269 and CDR-L3 as set forth in SEQ ID NO: 270, (g) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 94, CDR-H2 as set forth in SEQ ID NO: 95, CDR-H3 as set forth in SEQ ID NO: 910, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 262, CDR-L2 as set forth in SEQ ID NO: 263 and CDR-L3 as set forth in SEQ ID NO: 264, (h) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 94, CDR-H2 as set forth in SEQ ID NO: 95, CDR-H3 as set forth in SEQ ID NO: 911, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 262, CDR-L2 as set forth in SEQ ID NO: 263 and CDR-L3 as set forth in SEQ ID NO: 264, (i) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 118, CDR-H2 as set forth in SEQ ID NO: 119, CDR-H3 as set forth in SEQ ID NO: 120, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 286, CDR-L2 as set forth in SEQ ID NO: 287 and CDR-L3 as set forth in SEQ ID NO: 288, (j) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 118, CDR-H2 as set forth in SEQ ID NO: 914, CDR-H3 as set forth in SEQ ID NO: 120, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 286, CDR-L2 as set forth in SEQ ID NO: 287 and CDR-L3 as set forth in SEQ ID NO: 288, and (k) a VH region comprising CDR-H1 as set forth in CDR-H1 as set forth in SEQ ID NO: 154, CDR-H2 as set forth in SEQ ID NO: 155, CDR-H3 as set forth in SEQ ID NO: 920, and a VL region comprising CDR-L1 as set forth in SEQ ID NO: 322, CDR-L2 as set forth in SEQ ID NO: 323 and CDR-L3 as set forth in SEQ ID NO: 324.
2. The human antibody or antigen binding fragment thereof according to claim 1, wherein the antibody or antigen binding fragment thereof is a monoclonal antibody or a fragment thereof.
3. (canceled)
4. The human antibody or antigen binding fragment thereof according to claim 1, comprising a VH region comprising the amino acid sequence selected from the group consisting of: SEQ ID NO: 338, SEQ ID NO: 354, SEQ ID NO: 378, SEQ ID NO: 356, SEQ ID NO: 476, SEQ ID NO: 477, SEQ ID NO: 478, SEQ ID NO: 479, SEQ ID NO: 480, SEQ ID NO: 481, SEQ ID NO: 482, SEQ ID NO: 483, SEQ ID NO: 484, SEQ ID NO: 501, SEQ ID NO: 502, SEQ ID NO: 503, SEQ ID NO: 504, SEQ ID NO: 505, SEQ ID NO: 506, SEQ ID NO: 517, and SEQ ID NO: 518.
5. The human antibody or antigen binding fragment thereof according to claim 1, comprising a VL region comprising the amino acid sequence selected from the group consisting of: SEQ ID NO: 394, SEQ ID NO: 410, SEQ ID NO: 434, SEQ ID NO: 412, SEQ ID NO: 571, SEQ ID NO: 572, SEQ ID NO: 573, SEQ ID NO: 574, SEQ ID NO: 575, SEQ ID NO: 576, SEQ ID NO: 577, SEQ ID NO: 578, SEQ ID NO: 579, SEQ ID NO: 596, SEQ ID NO: 597, SEQ ID NO: 598, SEQ ID NO: 599, SEQ ID NO: 600, SEQ ID NO: 601, SEQ ID NO: 612, and SEQ ID NO: 613.
6. The human antibody or antigen binding fragment thereof according to claim 1, comprising a VH region and a VL region comprising a pair of amino acid sequences selected from the group consisting of: SEQ ID NOs: 338 and 394, SEQ ID NOs: 354 and 410, SEQ ID NOs: 378 and 434, SEQ ID NOs: 356 and 412, SEQ ID NOs: 476 and 571, SEQ ID NOs: 477 and 572, SEQ ID NOs: 478 and 573, SEQ ID NOs: 479 and 574, SEQ ID NOs: 480 and 575, SEQ ID NOs: 481 and 576, SEQ ID NOs: 482 and 577, SEQ ID NOs: 483 and 578, SEQ ID NOs: 484 and 579, SEQ ID NOs: 501 and 596, SEQ ID NOs; 502 and 597, SEQ ID NOs: 503 and 598, SEQ ID NOs: 504 and 599, SEQ ID NOs: 505 and 600, SEQ ID NOs: 506 and 601, SEQ ID NOs: 517 and 612, and SEQ ID NOs: 518 and 613.
7. The human antibody or antigen binding fragment thereof according to claim 6, wherein the human binding domain or antigen binding fragment thereof comprising a heavy and light chain amino acid sequence selected from the group consisting of: SEQ ID NOs: 656 and 692, SEQ ID NOs: 654 and 690, SEQ ID NOs: 664 and 700, SEQ ID NOs: 670 and 706, SEQ ID NOs: 738 and 833, SEQ ID NOs: 739 and 834, SEQ ID NOs: 740 and 835, SEQ ID NOs: 741 and 836, SEQ ID NOs: 742 and 837, SEQ ID NOs: 743 and 838, SEQ ID NOs: 744 and 839, SEQ ID NOs: 745 and 840, SEQ ID NOs: 746 and 841, SEQ ID NOs: 763 and 858, SEQ ID NOs: 764 and 859, SEQ ID NOs: 765 and 860, SEQ ID NOs: 766 and 861, SEQ ID NOs: 767 and 862, SEQ ID NOs: 768 and 863, SEQ ID NOs: 779 and 874, and SEQ ID NOs: 780 and 875.
8. An antibody construct comprising the human antibody or antigen binding fragment thereof according to claim 1 conjugated to a chemotherapeutic agent.
9. The antibody construct according to claim 8, further comprising a linker, wherein the linker conjugates the chemotherapeutic agent to the human antibody or antigen binding fragment thereof.
10. The antibody construct according to claim 9, wherein the linker is a non-cleavable linker.
11. The antibody construct according to claim 10, wherein the linker comprises N-succinimidyl 4-(N-maleimidomethyl) cyclohexane-1 carboxylate (MCC).
12. The antibody construct of claim 8, wherein the chemotherapeutic agent is conjugated to one or more lysines contained in the human antibody or antigen binding fragment thereof.
13. The antibody construct of claim 8, wherein the chemotherapeutic agent is maytansinoid (DM1).
14. The antibody construct of claim 13, wherein the average number of DM1 molecules per antibody construct is between 1 and 10.
15. The antibody construct of claim 13, wherein the average number of DM1 molecules per antibody construct is between 3 and 7.
16. The antibody construct of claim 13, wherein the average number of DM1 molecules per antibody construct is between 4 and 6.
17. The antibody construct of claim 13, wherein the average number of DM1 molecules per antibody construct is about 4.0, about 4.1, about 4.2, about 4.3, about 4.4, about 4.5, about 4.6, about 4.7, about 4.8, about 4.9, about 5.0, about 5.1, about 5.2, about 5.3, about 5.4, about 5.5, about 5.6, about 5.7, about 5.8, about 5.9, or about 6.0.
18. An isolated nucleic acid molecule encoding the human antibody or antigen binding fragment thereof of claim 1.
19. A vector comprising the nucleic acid molecule of claim 18.
20. A host cell transformed or transfected with the nucleic acid molecule of claim 18.
21. A process for producing a human antibody or antigen binding fragment thereof, said process comprising culturing the host cell of claim 16 under conditions allowing the expression of the antibody or antigen binding fragment thereof.
22. The process of claim 21 further comprising recovering the produced antibody or antigen binding fragment thereof, and conjugating a chemotherapeutic agent to the recovered antibody or antigen binding fragment thereof to produce an antibody conjugate.
23. A composition comprising the human antibody or antigen binding fragment thereof according to claim 1 and a pharmaceutically acceptable carrier.
24-25. (canceled)
26. A method for treating or ameliorating a melanoma disease or metastatic melanoma disease, comprising administering to a subject in need thereof the antibody or antigen binding fragment thereof of claim 1.
27. The method according to claim 26, wherein the melanoma disease or metastatic melanoma disease is selected from the group consisting of superficial spreading melanoma, lentigo maligna, lentigo maligna melanoma, acral lentiginous melanoma and nodular melanoma.
28. A kit comprising the antibody or antigen binding fragment thereof of claim 1 in a vial or a syringe.
Description
RELATED APPLICATIONS
[0001] This application is related to a U.S. provisional application entitled "Antibody constructs for CDH19 and CD3," filed on Mar. 15, 2013, the same day as the present application is filed. This related application is incorporated in its entirety by reference.
FIELD OF THE INVENTION
[0002] The present invention relates to compositions of antigen binding proteins including antibodies capable of binding to human CDH19 on the surface of a target cell, as well as related methods. Moreover, the invention provides a nucleic acid sequence encoding the antibody construct, a vector comprising the nucleic acid sequence and a host cell transformed or transfected with the nucleic acid sequence or a vector comprising the nucleic acid sequence. Furthermore, the invention provides a process for the production of the antibody of the invention, a method of treatment using the antibody and a kit comprising the antibody.
BACKGROUND OF THE INVENTION
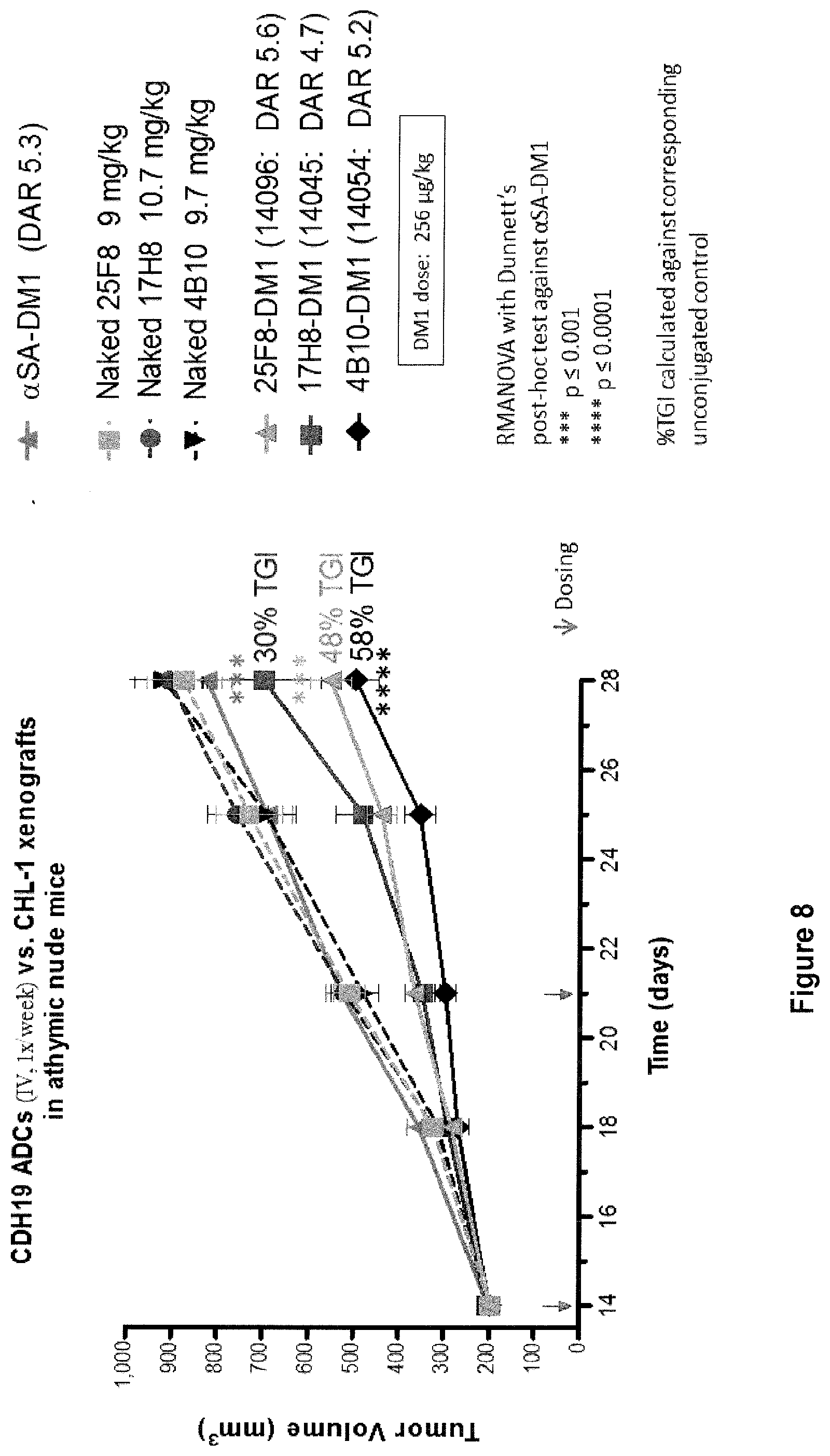
[0003] Melanoma is a skin cancer that is caused by the oncogenic transformation of melanocytes, which are pigment producing skin cells. As of 2009, Melanoma had a prevalence of more than 870,000 cases in the US alone (US National Institutes of Health). Each year, over 75,000 new cases of melanoma are diagnosed in the US, and approximately 25% of patients have advanced disease at the time of diagnosis. Despite the fact that cases of primary melanoma can be cured by surgery if they are detected early enough, melanoma is the leading cause of death from skin disease in the US, responsible for about 10,000 deaths per year in the US. Once the disease has spread and became metastatic, the prognosis is poor, with a 5 year relative survival of 15%.
[0004] There are four basic types of melanomas. Three types are found in the top layers of the skin and the fourth one is invasive and has penetrated deeper into the skin and may have spread to other areas of the body.
[0005] Superficial spreading melanoma is the most common type of melanoma which accounts for about 70% of all cases. It grows along the top layer of the skin for a fairly long time before penetrating more deeply. It first appears as a flat or slightly raised discolored patch that has irregular borders and may be somewhat asymmetrical in form. The color varies, and you may see areas of tan, brown, black, red, blue or white. This type of melanoma can occur in a previously benign mole and is found most often in young people.
[0006] Lentigo maligna is similar to the superficial spreading type, as it also remains close to the skin surface for quite a while, and usually appears as a flat or mildly elevated mottled tan, brown or dark brown discoloration. It is found most often in the elderly. When this cancer becomes invasive, it is referred to as lentigo maligna melanoma.
[0007] Acral lentiginous melanoma also spreads superficially before penetrating more deeply. It is quite different from the others, though, as it usually appears as a black or brown discoloration under the nails or on the soles of the feet or palms of the hands. This type of melanoma is sometimes found on dark-skinned people, and can often advance more quickly than superficial spreading melanoma and lentigo maligna.
[0008] Nodular melanoma is usually invasive at the time it is first diagnosed. The malignancy is recognized when it becomes a bump. It is usually black, but occasionally is blue, gray, white, brown, tan, red or skin tone. This is the most aggressive of the melanomas, and is found in 10 to 15 percent of cases.
[0009] Common treatments for metastatic melanoma include chemotherapy, targeted therapies for eligible patients (e.g. BRAF inhibitor treatment for patients with BRAF mutations) and immunotherapy. Metastatic melanoma is a tumor type where immunotherapy has been demonstrated to not only slow disease progression, but to lead to cures in late stage patients. Interleukin-2 was approved for the use in metastatic melanoma in 1998, and in 2011 an antibody targeting CTLA4, a member of a new generation of immune checkpoint inhibitors, gained approval by the FDA.
[0010] CDH19 is a type II cadherin transmembrane protein of unknown function. The human gene was cloned in 2000 based on its sequence similarity to CDH7 (Kools, P. et al. Genomics. 2000). Expressed Sequence Tags (ESTs) for CDH19 were isolated from melanocyte cDNA libraries, indicating that expression of CDH19 may be limited to cells of neural crest origin (Kools, P. et al. Genomics. 2000). In support of this notion, rat CDH19 was found to be expressed primarily in nerve ganglia and in Schwann cells during rat embryonic development (Takahashi, M. and Osumi, O. Devl Dynamics. 2005).
[0011] Diagnostic antibodies detecting CDH19 in Western Blot, immunohistochemitstry or flow cytometry are known in the art and commercially available. Those antibodies comprise poly- and monoclonal antibodies generated in animal hosts.
SUMMARY OF THE INVENTION
[0012] The present invention provides an isolated human antibody or antigen binding fragment thereof capable of binding to human CDH19 on the surface of a target cell. In a preferred embodiment the antibody or antigen binding fragment thereof comprises a monoclonal antibody or a fragment thereof.
[0013] In one embodiment the human antibody or antigen binding fragment thereof of the invention comprises a human binding domain or antigen binding fragment thereof comprising a VH region comprising CDR-H1, CDR-H2 and CDR-H3 and a VL region comprising CDR-L1, CDR-L2 and CDR-L3 selected from the group consisting of: [0014] (a) CDR-H1 as depicted in SEQ ID NO: 52, CDR-H2 as depicted in SEQ ID NO: 53, CDR-H3 as depicted in SEQ ID NO: 54, CDR-L1 as depicted in SEQ ID NO: 220, CDR-L2 as depicted in SEQ ID NO: 221 and CDR-L3 as depicted in SEQ ID NO: 222, CDR-H1 as depicted in SEQ ID NO: 82, CDR-H2 as depicted in SEQ ID NO: 83, CDR-H3 as depicted in SEQ ID NO: 84, CDR-L1 as depicted in SEQ ID NO: 250, CDR-L2 as depicted in SEQ ID NO: 251 and CDR-L3 as depicted in SEQ ID NO: 252, CDR-H1 as depicted in SEQ ID NO: 82, CDR-H2 as depicted in SEQ ID NO: 83, CDR-H3 as depicted in SEQ ID NO: 84, CDR-L1 as depicted in SEQ ID NO: 250, CDR-L2 as depicted in SEQ ID NO: 251 and CDR-L3 as depicted in SEQ ID NO: 927, CDR-H1 as depicted in SEQ ID NO: 82, CDR-H2 as depicted in SEQ ID NO: 83, CDR-H3 as depicted in SEQ ID NO: 909, CDR-L1 as depicted in SEQ ID NO: 250, CDR-L2 as depicted in SEQ ID NO: 251 and CDR-L3 as depicted in SEQ ID NO: 927, CDR-H1 as depicted in SEQ ID NO: 52, CDR-H2 as depicted in SEQ ID NO: 53, CDR-H3 as depicted in SEQ ID NO: 54, CDR-L1 as depicted in SEQ ID NO: 220, CDR-L2 as depicted in SEQ ID NO: 221 and CDR-L3 as depicted in SEQ ID NO: 926, and CDR-H1 as depicted in SEQ ID NO: 52, CDR-H2 as depicted in SEQ ID NO: 53, CDR-H3 as depicted in SEQ ID NO: 904, CDR-L1 as depicted in SEQ ID NO: 220, CDR-L2 as depicted in SEQ ID NO: 221 and CDR-L3 as depicted in SEQ ID NO: 926; [0015] (b) CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 126, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 294, CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 132, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 300, CDR-H1 as depicted in SEQ ID NO: 136, CDR-H2 as depicted in SEQ ID NO: 137, CDR-H3 as depicted in SEQ ID NO: 138, CDR-L1 as depicted in SEQ ID NO: 304, CDR-L2 as depicted in SEQ ID NO: 305 and CDR-L3 as depicted in SEQ ID NO: 306, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 144, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 312, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 318, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 336, CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 915, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 294, CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 915, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 928, CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 915, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 929, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 336, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 942, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 943, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 318, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 937, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 938, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 919, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 938, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 144, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 935, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 918, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 935, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 918, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 936, CDR-H1 as depicted in SEQ ID NO: 136, CDR-H2 as depicted in SEQ ID NO: 137, CDR-H3 as depicted in SEQ ID NO: 138, CDR-L1 as depicted in SEQ ID NO: 304, CDR-L2 as depicted in SEQ ID NO: 305 and CDR-L3 as depicted in SEQ ID NO: 933, CDR-H1 as depicted in SEQ ID NO: 136, CDR-H2 as depicted in SEQ ID NO: 137, CDR-H3 as depicted in SEQ ID NO: 917, CDR-L1 as depicted in SEQ ID NO: 304, CDR-L2 as depicted in SEQ ID NO: 305 and CDR-L3 as depicted in SEQ ID NO: 934, CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 132, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 930, CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 916, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 931, and CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 916, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 932; [0016] (c) CDR-H1 as depicted in SEQ ID NO: 94, CDR-H2 as depicted in SEQ ID NO: 95, CDR-H3 as depicted in SEQ ID NO: 96, CDR-L1 as depicted in SEQ ID NO: 262, CDR-L2 as depicted in SEQ ID NO: 263 and CDR-L3 as depicted in SEQ ID NO: 264, CDR-H1 as depicted in SEQ ID NO: 100, CDR-H2 as depicted in SEQ ID NO: 101, CDR-H3 as depicted in SEQ ID NO: 102, CDR-L1 as depicted in SEQ ID NO: 268, CDR-L2 as depicted in SEQ ID NO: 269 and CDR-L3 as depicted in SEQ ID NO: 270, CDR-H1 as depicted in SEQ ID NO: 118, CDR-H2 as depicted in SEQ ID NO: 119, CDR-H3 as depicted in SEQ ID NO: 120, CDR-L1 as depicted in SEQ ID NO: 286, CDR-L2 as depicted in SEQ ID NO: 287 and CDR-L3 as depicted in SEQ ID NO: 288, CDR-H1 as depicted in SEQ ID NO: 154, CDR-H2 as depicted in SEQ ID NO: 155, CDR-H3 as depicted in SEQ ID NO: 156, CDR-L1 as depicted in SEQ ID NO: 322, CDR-L2 as depicted in SEQ ID NO: 323 and CDR-L3 as depicted in SEQ ID NO: 324, CDR-H1 as depicted in SEQ ID NO: 100, CDR-H2 as depicted in SEQ ID NO: 101, CDR-H3 as depicted in SEQ ID NO: 912, CDR-L1 as depicted in SEQ ID NO: 268, CDR-L2 as depicted in SEQ ID NO: 269 and CDR-L3 as depicted in SEQ ID NO: 270, CDR-H1 as depicted in SEQ ID NO: 100, CDR-H2 as depicted in SEQ ID NO: 101, CDR-H3 as depicted in SEQ ID NO: 913, CDR-L1 as depicted in SEQ ID NO: 268, CDR-L2 as depicted in SEQ ID NO: 269 and CDR-L3 as depicted in SEQ ID NO: 270, CDR-H1 as depicted in SEQ ID NO: 94, CDR-H2 as depicted in SEQ ID NO: 95, CDR-H3 as depicted in SEQ ID NO: 910, CDR-L1 as depicted in SEQ ID NO: 262, CDR-L2 as depicted in SEQ ID NO: 263 and CDR-L3 as depicted in SEQ ID NO: 264, CDR-H1 as depicted in SEQ ID NO: 94, CDR-H2 as depicted in SEQ ID NO: 95, CDR-H3 as depicted in SEQ ID NO: 911, CDR-L1 as depicted in SEQ ID NO: 262, CDR-L2 as depicted in SEQ ID NO: 263 and CDR-L3 as depicted in SEQ ID NO: 264, CDR-H1 as depicted in SEQ ID NO: 118, CDR-H2 as depicted in SEQ ID NO: 119, CDR-H3 as depicted in SEQ ID NO: 120, CDR-L1 as depicted in SEQ ID NO: 286, CDR-L2 as depicted in SEQ ID NO: 287 and CDR-L3 as depicted in SEQ ID NO: 288, CDR-H1 as depicted in SEQ ID NO: 118, CDR-H2 as depicted in SEQ ID NO: 914, CDR-H3 as depicted in SEQ ID NO: 120, CDR-L1 as depicted in SEQ ID NO: 286, CDR-L2 as depicted in SEQ ID NO: 287 and CDR-L3 as depicted in SEQ ID NO: 288, and CDR-H1 as depicted in SEQ ID NO: 154, CDR-H2 as depicted in SEQ ID NO: 155, CDR-H3 as depicted in SEQ ID NO: 920, CDR-L1 as depicted in SEQ ID NO: 322, CDR-L2 as depicted in SEQ ID NO: 323 and CDR-L3 as depicted in SEQ ID NO: 324; [0017] (d) CDR-H1 as depicted in SEQ ID NO: 4, CDR-H2 as depicted in SEQ ID NO: 5, CDR-H3 as depicted in SEQ ID NO: 6, CDR-L1 as depicted in SEQ ID NO: 172, CDR-L2 as depicted in SEQ ID NO: 173 and CDR-L3 as depicted in SEQ ID NO: 174, CDR-H1 as depicted in SEQ ID NO: 10, CDR-H2 as depicted in SEQ ID NO: 11, CDR-H3 as depicted in SEQ ID NO: 12, CDR-L1 as depicted in SEQ ID NO: 178, CDR-L2 as depicted in SEQ ID NO: 179 and CDR-L3 as depicted in SEQ ID NO: 180, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 196, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 198, CDR-H1 as depicted in SEQ ID NO: 34, CDR-H2 as depicted in SEQ ID NO: 35, CDR-H3 as depicted in SEQ ID NO: 36, CDR-L1 as depicted in SEQ ID NO: 202, CDR-L2 as depicted in SEQ ID NO: 203 and CDR-L3 as depicted in SEQ ID NO: 204, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 48, CDR-L1 as depicted in SEQ ID NO: 214, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 58, CDR-H2 as depicted in SEQ ID NO: 59, CDR-H3 as depicted in SEQ ID NO: 60, CDR-L1 as depicted in SEQ ID NO: 226, CDR-L2 as depicted in SEQ ID NO: 227 and CDR-L3 as depicted in SEQ ID NO: 228, CDR-H1 as depicted in SEQ ID NO: 64, CDR-H2 as depicted in SEQ ID NO: 65, CDR-H3 as depicted in SEQ ID NO: 66, CDR-L1 as depicted in SEQ ID NO: 232, CDR-L2 as depicted in SEQ ID NO: 233 and CDR-L3 as depicted in SEQ ID NO: 234, CDR-H1 as depicted in SEQ ID NO: 70, CDR-H2 as depicted in SEQ ID NO: 71, CDR-H3 as depicted in SEQ ID NO: 72, CDR-L1 as depicted in SEQ ID NO: 238, CDR-L2 as depicted in SEQ ID NO: 239 and CDR-L3 as depicted in SEQ ID NO: 240, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 161, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 328, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 48, CDR-L1 as depicted in SEQ ID NO: 924, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 902, CDR-L1 as depicted in SEQ ID NO: 924, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 903, CDR-L1 as depicted in SEQ ID NO: 924, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 48, CDR-L1 as depicted in SEQ ID NO: 925, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 70, CDR-H2 as depicted in SEQ ID NO: 907, CDR-H3 as depicted in SEQ ID NO: 72, CDR-L1 as depicted in SEQ ID NO: 238, CDR-L2 as depicted in SEQ ID NO: 239 and CDR-L3 as depicted in SEQ ID NO: 240, CDR-H1 as depicted in SEQ ID NO: 70, CDR-H2 as depicted in SEQ ID NO: 907, CDR-H3 as depicted in SEQ ID NO: 908, CDR-L1 as depicted in SEQ ID NO: 238, CDR-L2 as depicted in SEQ ID NO: 239 and CDR-L3 as depicted in SEQ ID NO: 240, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 901, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 922, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, CDR-H1 as depicted in SEQ ID NO: 58, CDR-H2 as depicted in SEQ ID NO: 905, CDR-H3 as depicted in SEQ ID NO: 906, CDR-L1 as depicted in SEQ ID NO: 226, CDR-L2 as depicted in SEQ ID NO: 227 and CDR-L3 as depicted in SEQ ID NO: 228, CDR-H1 as depicted in SEQ ID NO: 58, CDR-H2 as depicted in SEQ ID NO: 905, CDR-H3 as depicted in SEQ ID NO: 60, CDR-L1 as depicted in SEQ ID NO: 226, CDR-L2 as depicted in SEQ ID NO: 227 and CDR-L3 as depicted in SEQ ID NO: 228, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 161, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 939, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 921, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 939, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 940, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 161, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 941, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 196, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 922, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 901, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 922, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, and CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 939, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330; and
[0018] (e) CDR-H1 as depicted in SEQ ID NO: 76, CDR-H2 as depicted in SEQ ID NO: 77, CDR-H3 as depicted in SEQ ID NO: 78, CDR-L1 as depicted in SEQ ID NO: 244, CDR-L2 as depicted in SEQ ID NO: 245 and CDR-L3 as depicted in SEQ ID NO: 246, CDR-H1 as depicted in SEQ ID NO: 88, CDR-H2 as depicted in SEQ ID NO: 89, CDR-H3 as depicted in SEQ ID NO: 90, CDR-L1 as depicted in SEQ ID NO: 256, CDR-L2 as depicted in SEQ ID NO: 257 and CDR-L3 as depicted in SEQ ID NO: 258, CDR-H1 as depicted in SEQ ID NO: 106, CDR-H2 as depicted in SEQ ID NO: 107, CDR-H3 as depicted in SEQ ID NO: 108, CDR-L1 as depicted in SEQ ID NO: 274, CDR-L2 as depicted in SEQ ID NO: 275 and CDR-L3 as depicted in SEQ ID NO: 276, CDR-H1 as depicted in SEQ ID NO: 112, CDR-H2 as depicted in SEQ ID NO: 113, CDR-H3 as depicted in SEQ ID NO: 114, CDR-L1 as depicted in SEQ ID NO: 280, CDR-L2 as depicted in SEQ ID NO: 281 and CDR-L3 as depicted in SEQ ID NO: 282, and CDR-H1 as depicted in SEQ ID NO: 106, CDR-H2 as depicted in SEQ ID NO: 107, CDR-H3 as depicted in SEQ ID NO: 108, CDR-L1 as depicted in SEQ ID NO: 274, CDR-L2 as depicted in SEQ ID NO: 275 and CDR-L3 as depicted in SEQ ID NO: 276.
[0019] In a further embodiment of the human antibody or antigen binding fragment thereof of the invention the human binding domain or antigen binding fragment thereof comprises a VH region selected from the group consisting of VH regions [0020] (a) as depicted in SEQ ID NO: 362, SEQ ID NO: 364, SEQ ID NO: 485, SEQ ID NO: 486, SEQ ID NO: 487, SEQ ID NO: 492, SEQ ID NO: 493, SEQ ID NO: 494, and SEQ ID NO: 495; [0021] (b) as depicted in SEQ ID NO: 342, SEQ ID NO: 366, SEQ ID NO: 370, SEQ ID NO: 344, SEQ ID NO: 372, SEQ ID NO: 368, SEQ ID NO: 496, SEQ ID NO: 497, SEQ ID NO: 498, SEQ ID NO: 499, SEQ ID NO: 500, SEQ ID NO: 508, SEQ ID NO: 509, SEQ ID NO: 510, SEQ ID NO: 511, SEQ ID NO: 512, SEQ ID NO: 519, SEQ ID NO: 520, SEQ ID NO: 521, SEQ ID NO: 522, SEQ ID NO: 523, SEQ ID NO: 524, SEQ ID NO: 525, SEQ ID NO: 526, SEQ ID NO: 527, SEQ ID NO: 528, SEQ ID NO: 529, SEQ ID NO: 530, SEQ ID NO: 531, SEQ ID NO: 532, SEQ ID NO: 533, SEQ ID NO: 534, SEQ ID NO: 535, SEQ ID NO: 536, SEQ ID NO: 537, and SEQ ID NO: 538; [0022] (c) as depicted in SEQ ID NO: 338, SEQ ID NO: 354, SEQ ID NO: 378, SEQ ID NO: 356, SEQ ID NO: 476, SEQ ID NO: 477, SEQ ID NO: 478, SEQ ID NO: 479, SEQ ID NO: 480, SEQ ID NO: 481, SEQ ID NO: 482, SEQ ID NO: 483, SEQ ID NO: 484, SEQ ID NO: 501, SEQ ID NO: 502, SEQ ID NO: 503, SEQ ID NO: 504, SEQ ID NO: 505, SEQ ID NO: 506, SEQ ID NO: 517, and SEQ ID NO: 518; [0023] (d) as depicted in SEQ ID NO: 352, SEQ ID NO: 360, SEQ ID NO: 388, SEQ ID NO: 386, SEQ ID NO: 340, SEQ ID NO: 346, SEQ ID NO: 374, SEQ ID NO: 348, SEQ ID NO: 390, SEQ ID NO: 463, SEQ ID NO: 464, SEQ ID NO: 465, SEQ ID NO: 466, SEQ ID NO: 467, SEQ ID NO: 468, SEQ ID NO: 469, SEQ ID NO: 470, SEQ ID NO: 471, SEQ ID NO: 472, SEQ ID NO: 473, SEQ ID NO: 474, SEQ ID NO: 475, SEQ ID NO: 488, SEQ ID NO: 489, SEQ ID NO: 490, SEQ ID NO: 491, SEQ ID NO: 513, SEQ ID NO: 514, SEQ ID NO: 515, SEQ ID NO: 516, SEQ ID NO: 540, SEQ ID NO: 541, SEQ ID NO: 542, and SEQ ID NO: 543; and [0024] (e) as depicted in SEQ ID NO: 376, SEQ ID NO: 392, SEQ ID NO: 358, SEQ ID NO: 350, and SEQ ID NO: 507.
[0025] In another embodiment the human antibody or antigen binding fragment thereof of the invention comprises the human binding domain or antigen binding fragment thereof comprising a VL region selected from the group consisting of VL regions [0026] (a) as depicted in SEQ ID NO: 418, SEQ ID NO: 420, SEQ ID NO: 580, SEQ ID NO: 581, SEQ ID NO: 582, SEQ ID NO: 587, SEQ ID NO: 588, SEQ ID NO: 589, and SEQ ID NO: 590; [0027] (b) as depicted in SEQ ID NO: 398, SEQ ID NO: 422, SEQ ID NO: 426, SEQ ID NO: 400, SEQ ID NO: 428, SEQ ID NO: 424, SEQ ID NO: 591, SEQ ID NO: 592, SEQ ID NO: 593, SEQ ID NO: 594, SEQ ID NO: 595, SEQ ID NO: 603, SEQ ID NO: 604, SEQ ID NO: 605, SEQ ID NO: 606, SEQ ID NO: 607, SEQ ID NO: 614, SEQ ID NO: 615, SEQ ID NO: 616, SEQ ID NO: 617, SEQ ID NO: 618, SEQ ID NO: 619, SEQ ID NO: 620, SEQ ID NO: 621, SEQ ID NO: 622, SEQ ID NO: 623, SEQ ID NO: 624, SEQ ID NO: 625, SEQ ID NO: 626, SEQ ID NO: 627, SEQ ID NO: 628, SEQ ID NO: 629, SEQ ID NO: 630, SEQ ID NO: 631, SEQ ID NO: 632, and SEQ ID NO: 633; [0028] (c) as depicted in SEQ ID NO: 394, SEQ ID NO: 410, SEQ ID NO: 434, SEQ ID NO: 412, SEQ ID NO: 571, SEQ ID NO: 572, SEQ ID NO: 573, SEQ ID NO: 574, SEQ ID NO: 575, SEQ ID NO: 576, SEQ ID NO: 577, SEQ ID NO: 578, SEQ ID NO: 579, SEQ ID NO: 596, SEQ ID NO: 597, SEQ ID NO: 598, SEQ ID NO: 599, SEQ ID NO: 600, SEQ ID NO: 601, SEQ ID NO: 612, and SEQ ID NO: 613; [0029] (d) as depicted in SEQ ID NO: 408, SEQ ID NO: 416, SEQ ID NO: 444, SEQ ID NO: 442, SEQ ID NO: 396, SEQ ID NO: 402, SEQ ID NO: 430, SEQ ID NO: 404, SEQ ID NO: 446, SEQ ID NO: 558, SEQ ID NO: 559, SEQ ID NO: 560, SEQ ID NO: 561, SEQ ID NO: 562, SEQ ID NO: 563, SEQ ID NO: 564, SEQ ID NO: 565, SEQ ID NO: 566, SEQ ID NO: 567, SEQ ID NO: 568, SEQ ID NO: 569, SEQ ID NO: 570, SEQ ID NO: 583, SEQ ID NO: 584, SEQ ID NO: 585, SEQ ID NO: 586, SEQ ID NO: 608, SEQ ID NO: 609, SEQ ID NO: 610, SEQ ID NO: 611, SEQ ID NO: 635, SEQ ID NO: 636, SEQ ID NO: 637, and SEQ ID NO: 638; and [0030] (e) as depicted in SEQ ID NO: 432, SEQ ID NO: 448, SEQ ID NO: 414, SEQ ID NO: 406, and SEQ ID NO: 602.
[0031] The invention further provides an embodiment of the human antibody or antigen binding fragment thereof of the invention, wherein the human binding domain or antigen binding fragment thereof comprises a VH region and a VL region selected from the group consisting of: [0032] (1) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 362+418, SEQ ID NOs: 364+420, SEQ ID NOs: 485+580, SEQ ID NOs: 486+581, SEQ ID NOs: 487+582, SEQ ID NOs: 492+587, SEQ ID NOs: 493+588, SEQ ID NOs: 494+589, and SEQ ID NOs: 495+590; [0033] (2) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 342+398, SEQ ID NOs: 366+422, SEQ ID NOs: 370+426, SEQ ID NOs: 344+400, SEQ ID NOs: 372+428, SEQ ID NOs: 368+424, SEQ ID NOs: 496+591, SEQ ID NOs: 497+592, SEQ ID NOs: 498+593, SEQ ID NOs: 499+594, SEQ ID NOs: 500+595, SEQ ID NOs: 508+603, SEQ ID NOs: 509+604, SEQ ID NOs: 510+605, SEQ ID NOs: 511+606, SEQ ID NOs: 512+607, SEQ ID NOs: 519+614, SEQ ID NOs: 520+615, SEQ ID NOs: 521+616, SEQ ID NOs: 522+617, SEQ ID NOs: 523+618, SEQ ID NOs: 524+619, SEQ ID NOs: 525+620, SEQ ID NOs: 526+621, SEQ ID NOs: 527+622, SEQ ID NOs: 528+623, SEQ ID NOs: 529+624, SEQ ID NOs: 530+625, SEQ ID NOs: 531+626, SEQ ID NOs: 532+627, SEQ ID NOs: 533+628, SEQ ID NOs: 534+629, SEQ ID NOs: 535+630, SEQ ID NOs: 536+631, SEQ ID NOs: 537+632, and SEQ ID NOs: 538+633; [0034] (3) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 338+394, SEQ ID NOs: 354+410, SEQ ID NOs: 378+434, SEQ ID NOs: 356+412, SEQ ID NOs: 476+571, SEQ ID NOs: 477+572, SEQ ID NOs: 478+573, SEQ ID NOs: 479+574, SEQ ID NOs: 480+575, SEQ ID NOs: 481+576, SEQ ID NOs: 482+577, SEQ ID NOs: 483+578, SEQ ID NOs: 484+579, SEQ ID NOs: 501+596, SEQ ID NOs: 502+597, SEQ ID NOs: 503+598, SEQ ID NOs: 504+599, SEQ ID NOs: 505+600, SEQ ID NOs: 506+601, SEQ ID NOs: 517+612, and SEQ ID NOs: 518+613; [0035] (4) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 352+408, SEQ ID NOs: 360+416, SEQ ID NOs: 388+444, SEQ ID NOs: 386+442, SEQ ID NOs: 340+396, SEQ ID NOs: 346+402, SEQ ID NOs: 374+430, SEQ ID NOs: 348+404, SEQ ID NOs: 390+446, SEQ ID NOs: 463+558, SEQ ID NOs: 464+559, SEQ ID NOs: 465+560, SEQ ID NOs: 466+561, SEQ ID NOs: 467+562, SEQ ID NOs: 468+563, SEQ ID NOs: 469+564, SEQ ID NOs: 470+565, SEQ ID NOs: 471+566, SEQ ID NOs: 472+567, SEQ ID NOs: 473+568, SEQ ID NOs: 474+569, SEQ ID NOs: 475+570, SEQ ID NOs: 488+583, SEQ ID NOs: 489+584, SEQ ID NOs: 490+585, SEQ ID NOs: 491+586, SEQ ID NOs: 513+608, SEQ ID NOs: 514+609, SEQ ID NOs: 515+610, SEQ ID NOs: 516+611, SEQ ID NOs: 540+635, SEQ ID NOs: 541+636, SEQ ID NOs: 542+637, and SEQ ID NOs: 543+638; and [0036] (5) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 376+432, SEQ ID NOs: 392+448, SEQ ID NOs: 358+414, SEQ ID NOs: 350+406, and SEQ ID NOs: 507+602.
[0037] In a further embodiment the human binding domain or antigen binding fragment thereof comprises the groups of heavy and light chains having an amino acid sequence selected from the group consisting of [0038] (1) a heavy and light chain as depicted in SEQ ID NOs: 644+680, SEQ ID NOs: 650+686, SEQ ID NOs: 747+842, SEQ ID NOs: 748+843, SEQ ID NOs: 749+844, SEQ ID NOs: 754+849, SEQ ID NOs: 755+850, SEQ ID NOs: 756+851, and SEQ ID NOs: 757+852; [0039] (2) a heavy and light chain as depicted in SEQ ID NOs: 660+696, SEQ ID NOs: 662+698, SEQ ID NOs: 668+704, SEQ ID NOs: 674+710, SEQ ID NOs: 672+708, SEQ ID NOs: 658+694, SEQ ID NOs: 758+853, SEQ ID NOs: 759+854, SEQ ID NOs: 760+855, SEQ ID NOs: 761+856, SEQ ID NOs: 762+857, SEQ ID NOs: 770+865, SEQ ID NOs: 771+866, SEQ ID NOs: 772+867, SEQ ID NOs: 773+868, SEQ ID NOs: 774+869, SEQ ID NOs: 781+876, SEQ ID NOs: 782+877, SEQ ID NOs: 783+878, SEQ ID NOs: 784+879, SEQ ID NOs: 785+880, SEQ ID NOs: 786+881, SEQ ID NOs: 787+882, SEQ ID NOs: 788+883, SEQ ID NOs: 789+884, SEQ ID NOs: 790+885, SEQ ID NOs: 791+886, SEQ ID NOs: 792+887, SEQ ID NOs: 793+888, SEQ ID NOs: 794+889, SEQ ID NOs: 795+890, SEQ ID NOs: 796+891, SEQ ID NOs: 797+892, SEQ ID NOs: 798+893, SEQ ID NOs: 799+894, and SEQ ID NOs: 800+895; [0040] (3) a heavy and light chain as depicted in SEQ ID NOs: 656+692, SEQ ID NOs: 654+690, SEQ ID NOs: 664+700, SEQ ID NOs: 670+706, SEQ ID NOs: 738+833, SEQ ID NOs: 739+834, SEQ ID NOs: 740+835, SEQ ID NOs: 741+836, SEQ ID NOs: 742+837, SEQ ID NOs: 743+838, SEQ ID NOs: 744+839, SEQ ID NOs: 745+840, SEQ ID NOs: 746+841, SEQ ID NOs: 763+858, SEQ ID NOs: 764+859, SEQ ID NOs: 765+860, SEQ ID NOs: 766+861, SEQ ID NOs: 767+862, SEQ ID NOs: 768+863, SEQ ID NOs: 779+874, and SEQ ID NOs: 780+875; [0041] (4) a heavy and light chain as depicted in SEQ ID NOs: 640+676, SEQ ID NOs: 642+678, SEQ ID NOs: 646+682, SEQ ID NOs: 648+684, SEQ ID NOs: 666+702, SEQ ID NOs: 725+820, SEQ ID NOs: 726+821, SEQ ID NOs: 727+822, SEQ ID NOs: 728+823, SEQ ID NOs: 729+824, SEQ ID NOs: 730+825, SEQ ID NOs: 731+826, SEQ ID NOs: 732+827, SEQ ID NOs: 733+828, SEQ ID NOs: 734+829, SEQ ID NOs: 735+830, SEQ ID NOs: 736+831, SEQ ID NOs: 737+832, SEQ ID NOs: 750+845, SEQ ID NOs: 751+846, SEQ ID NOs: 752+847, SEQ ID NOs: 753+848, SEQ ID NOs: 775+870, SEQ ID NOs: 776+871, SEQ ID NOs: 777+872, SEQ ID NOs: 778+873, SEQ ID NOs: 802+897, SEQ ID NOs: 803+898, SEQ ID NOs: 804+899, and SEQ ID NOs: 805+900; and [0042] (5) a heavy and light chain as depicted in SEQ ID NOs: 652+688, and SEQ ID NOs: 769+864.
[0043] In another embodiment the invention is directed to an antibody construct comprising the human antibody or antigen binding fragment thereof capable of binding to human CDH19 on the surface of a target cell as described above that is conjugated to a chemotherapeutic agent.
[0044] In one embodiment of the antibody construct of the invention a linker conjugates the chemotherapeutic agent to the human antibody or antigen binding fragment thereof.
[0045] In a preferred embodiment of the antibody construct of the invention the linker is a non-cleavable linker.
[0046] It is also preferred that the linker in the antibody construct of the invention comprises MCC.
[0047] In a further embodiment of the antibody construct of the invention the chemotherapeutic agent is conjugated to one or more lysines contained in the human antibody or antigen binding fragment thereof.
[0048] In one embodiment of the antibody construct of the invention the chemotherapeutic agent is DM1.
[0049] In a preferred embodiment of the antibody construct of the invention the average number of DM1 molecules per antibody construct is between 1 and 10.
[0050] It is also preferred for the antibody construct of the invention that the average number of DM1 molecules per antibody construct is between 3 and 7.
[0051] Moreover, it is preferred for the antibody construct of the invention that the average number of DM1 molecules per antibody construct is between 4 and 6.
[0052] In a further alternative embodiment of the antibody construct of the invention the average number of DM1 molecules per antibody construct is about 4.0, about 4.1, about 4.2, about 4.3, about 4.4, about 4.5, about 4.6, about 4.7, about 4.8, about 4.9, about 5.0, about 5.1, about 5.2, about 5.3, about 5.4, about 5.5, about 5.6, about 5.7, about 5.8, about 5.9, or about 6.0.
[0053] The invention further provides an isolated nucleic acid molecule or sequence encoding a human antibody or antigen binding fragment thereof of the invention.
[0054] Furthermore, the invention provides a vector comprising a nucleic acid sequence of the invention. Moreover, the invention provides a host cell transformed or transfected with the nucleic acid sequence of the invention or with a vector comprising the nucleic acid molecule.
[0055] In a further embodiment the invention provides a process for the production of a human antibody or an antigen binding fragment thereof of the invention, said process comprising culturing a host cell of the invention under conditions allowing the expression of the human antibody or antigen binding fragment thereof of the invention and recovering the produced antibody or antigen binding fragment thereof from the culture.
[0056] In a further embodiment the invention provides a process for the production of an antibody construct comprising a human antibody or an antigen binding fragment thereof of the invention, said process comprising culturing a host cell of the invention under conditions allowing the expression of the human antibody or antigen binding fragment thereof of the invention and recovering the produced antibody or antigen binding fragment thereof from the culture, and conjugating a chemotherapeutic agent to the recovered antibody or antigen binding fragment thereof to produce the antibody conjugate.
[0057] Moreover, the invention provides a pharmaceutical composition comprising a human antibody or antigen binding fragment thereof of the invention or an antibody construct of the invention or produced according to the process of the invention in admixture with a pharmaceutically acceptable carrier thereof.
[0058] In one embodiment the invention provides the human antibody or antigen binding fragment thereof of the invention, the antibody construct of the invention, or produced according to the process of the invention for use in the prevention, treatment or amelioration of a melanoma disease or metastatic melanoma disease. Preferably, the melanoma disease or metastatic melanoma disease is selected from the group consisting of superficial spreading melanoma, lentigo maligna, lentigo maligna melanoma, acral lentiginous melanoma and nodular melanoma.
[0059] The invention also provides a method for the treatment or amelioration of a melanoma disease or metastatic melanoma disease, comprising the step of administering to a subject in need thereof the antibody or antigen binding fragment thereof of the invention, the antibody construct of the invention, an antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention produced according to the process of the invention or a pharmaceutical composition of the invention.
[0060] In a preferred embodiment method the invention the melanoma disease or metastatic melanoma disease is selected from the group consisting of superficial spreading melanoma, lentigo maligna, lentigo maligna melanoma, acral lentiginous melanoma and nodular melanoma.
[0061] In a further embodiment, the invention provides a kit comprising an antibody or antigen binding fragment thereof of the invention, an antibody construct of the invention, an antibody or antigen binding fragment thereof of the invention or the antibody construct produced according to the process of the invention, a vector of the invention, and/or a host cell of the invention.
BRIEF DESCRIPTION OF THE DRAWINGS
[0062] FIG. 1 depicts cell viability data of Colo-699 cells that have been treated with fully human anti-CDH19 antibodies and a high concentration of a goat anti-human Fc monovalent Fab conjugated with DM1 (DM1-Fab) at a drug-antibody ratio (DAR) (.about.1.3).
[0063] FIG. 2 depicts the average cell viability data from a CHL-1 assay plotted against the average cell viability data from the Colo-699 assay.
[0064] FIG. 3 shows the relative expression of CDH19 mRNA in metastatic and primary melanoma samples.
[0065] FIG. 4 shows the expression of CDH19 protein in human tumor samples by IHC.
[0066] FIG. 5 shows the results of the analysis of tumor cell lines by flow cytometry and IHC to identify model systems with CDH19 expression similar to human tumors based on the number of CDH19 receptors present on the cell surface.
[0067] FIG. 6 shows in vitro activity of a CDH19 ADC against the model tumor cell lines.
[0068] FIG. 7 shows in vitro activity of a CDH19 ADC in model tumor cell lines at varying DAR ratios.
[0069] FIG. 8 shows in vivo activity of CDH19 ADCs in a xenograft mouse model as compared to naked CDH19 antibodies.
[0070] FIG. 9 shows in vivo activity of CDH19 ADCs in a xenograft mouse model. 4B10-DM1 Moderately Inhibited Tumor Growth at 182 .mu.g/kg DM1 in CHL-1 Xenografts
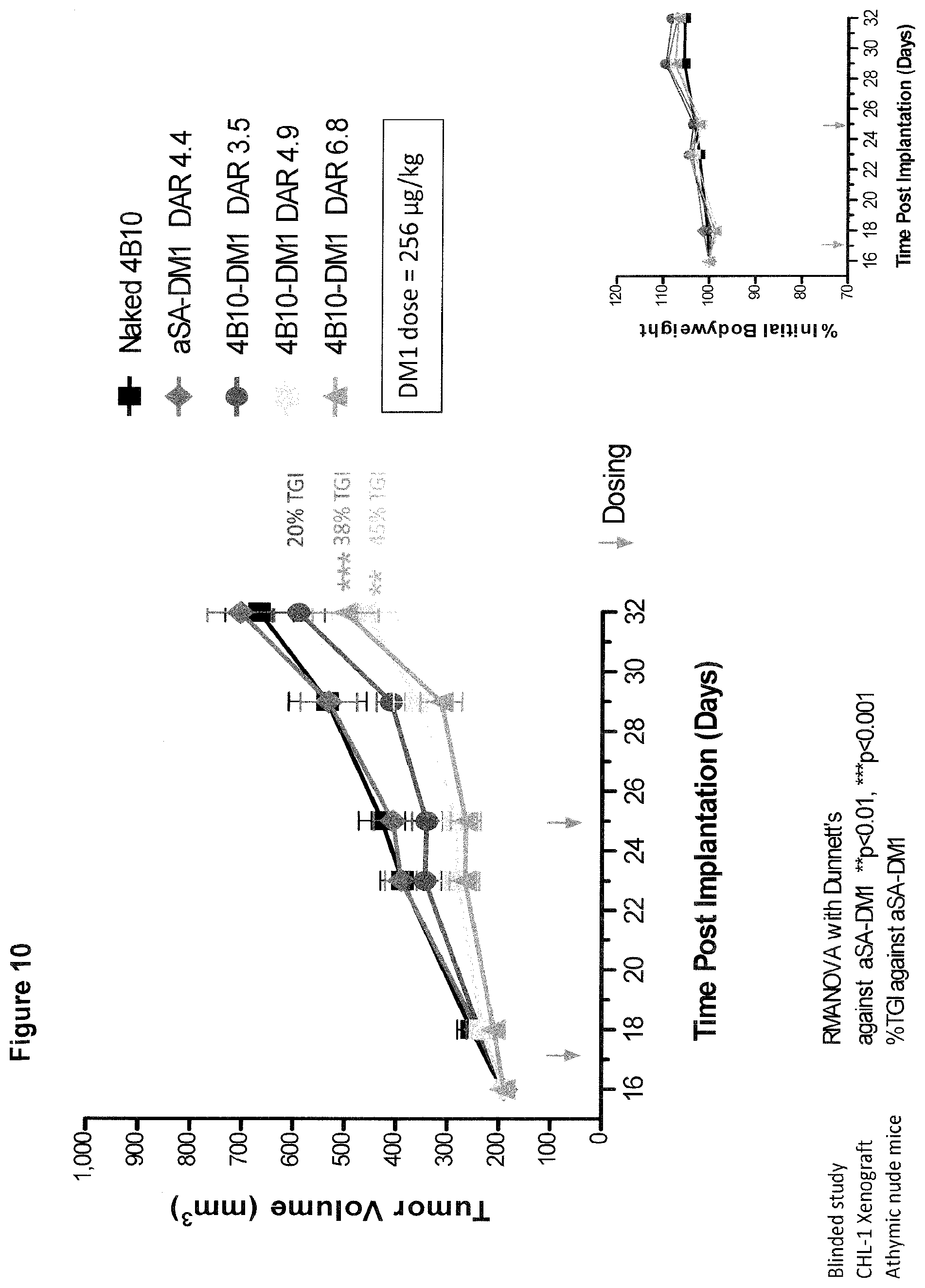
[0071] FIG. 10 shows in vivo activity of CDH19 ADCs in a xenograft mouse model. Increasing the DAR Did Not Increase Tumor Growth Inhibition in CHL-1 Xenografts
[0072] FIG. 11 shows in vivo activity of CDH19 ADCs in a xenograft mouse model. Anti-CDH19 ADCs Moderately Inhibited Tumor Growth in COLO699 Xenografts
DETAILED DESCRIPTION OF THE INVENTION
Definitions
[0073] It must be noted that as used herein, the singular forms "a", "an", and "the", include plural references unless the context clearly indicates otherwise. Thus, for example, reference to "a reagent" includes one or more of such different reagents and reference to "the method" includes reference to equivalent steps and methods known to those of ordinary skill in the art that could be modified or substituted for the methods described herein.
[0074] Unless otherwise indicated, the term "at least" preceding a series of elements is to be understood to refer to every element in the series. Those skilled in the art will recognize, or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments of the invention described herein. Such equivalents are intended to be encompassed by the present invention.
[0075] The term "and/or" wherever used herein includes the meaning of "and", "or" and "all or any other combination of the elements connected by said term".
[0076] The term "about" or "approximately" as used herein means within .+-.20%, preferably within .+-.15%, more preferably within .+-.10%, and most preferably within .+-.5% of a given value or range.
[0077] Throughout this specification and the claims which follow, unless the context requires otherwise, the word "comprise", and variations such as "comprises" and "comprising", will be understood to imply the inclusion of a stated integer or step or group of integers or steps but not the exclusion of any other integer or step or group of integer or step. When used herein the term "comprising" can be substituted with the term "containing" or "including" or sometimes when used herein with the term "having".
[0078] When used herein "consisting of" excludes any element, step, or ingredient not specified in the claim element. When used herein, "consisting essentially of" does not exclude materials or steps that do not materially affect the basic and novel characteristics of the claim.
[0079] In each instance herein any of the terms "comprising", "consisting essentially of" and "consisting of" may be replaced with either of the other two terms.
[0080] The definition of the term "antibody" includes embodiments such as monoclonal, chimeric, single chain, humanized and human antibodies, as well as antibody fragments, like, inter alia, Fab fragments. Antibody fragments or derivatives further comprise F(ab').sub.2, Fv, scFv fragments or single domain antibodies such as domain antibodies or nanobodies, single variable domain antibodies or immunoglobulin single variable domain comprising merely one variable domain, which might be VHH, VH or VL, that specifically bind an antigen or epitope independently of other V regions or domains; see, for example, Harlow and Lane (1988) and (1999), loc. cit.; Kontermann and Dubel, Antibody Engineering, Springer, 2nd ed. 2010 and Little, Recombinant Antibodies for Immunotherapy, Cambridge University Press 2009. Such immunoglobulin single variable domain encompasses not only an isolated antibody single variable domain polypeptide, but also larger polypeptides that comprise one or more monomers of an antibody single variable domain polypeptide sequence.
[0081] In line with this definition all above described embodiments of the term antibody can be subsumed under the term "antibody construct". Said term also includes diabodies or Dual-Affinity Re-Targeting (DART) antibodies. Further envisaged are (bispecific) single chain diabodies, tandem diabodies (Tandab's), "minibodies" exemplified by a structure which is as follows: (VH-VL-CH3).sub.2, (scFv-CH3).sub.2 or (scFv-CH3-scFv).sub.2, "Fc DART" antibodies and "IgG DART" antibodies, and multibodies such as triabodies. Immunoglobulin single variable domains encompass not only an isolated antibody single variable domain polypeptide, but also larger polypeptides that comprise one or more monomers of an antibody single variable domain polypeptide sequence.
[0082] Various procedures are known in the art and may be used for the production of such antibody constructs (antibodies and/or fragments). Thus, (antibody) derivatives can be produced by peptidomimetics. Further, techniques described for the production of single chain antibodies (see, inter alia, U.S. Pat. No. 4,946,778, Kontermann and Dubel (2010), loc. cit. and Little (2009), loc. cit.) can be adapted to produce single chain antibodies specific for elected polypeptide(s). Also, transgenic animals may be used to express humanized antibodies specific for polypeptides and fusion proteins of this invention. For the preparation of monoclonal antibodies, any technique, providing antibodies produced by continuous cell line cultures can be used. Examples for such techniques include the hybridoma technique (Kohler and Milstein Nature 256 (1975), 495-497), the trioma technique, the human B-cell hybridoma technique (Kozbor, Immunology Today 4 (1983), 72) and the EBV-hybridoma technique to produce human monoclonal antibodies (Cole et al., Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, Inc. (1985), 77-96). Surface plasmon resonance as employed in the BIAcore system can be used to increase the efficiency of phage antibodies which bind to an epitope of a target polypeptide, such as CDH19 (Schier, Human Antibodies Hybridomas 7 (1996), 97-105; Malmborg, J. Immunol. Methods 183 (1995), 7-13). It is also envisaged in the context of this invention that the term "antibody" comprises antibody constructs, which may be expressed in a host as described herein below, e.g. antibody constructs which may be transfected and/or transduced via, inter alia, viruses or plasmid vectors.
[0083] Furthermore, the term "antibody" as employed in the invention also relates to derivatives or variants of the antibodies described herein which display the same specificity as the described antibodies. Accordingly, the term "antibody" also subsumes antibody constructs such as different types of fragments of antibodies, which still are characterized by the feature of specific binding for CDH19.
[0084] The terms "antigen-binding domain", "antigen-binding fragment" and "antibody binding region" when used herein refer to a part of an antibody molecule that comprises amino acids responsible for the specific binding between antibody and antigen. The part of the antigen that is specifically recognized and bound by the antibody is referred to as the "epitope" as described herein above. As mentioned above, an antigen-binding domain may typically comprise an antibody light chain variable region (VL) and an antibody heavy chain variable region (VH); however, it does not have to comprise both. Fd fragments, for example, have two VH regions and often retain some antigen-binding function of the intact antigen-binding domain. Examples of antigen-binding fragments of an antibody include (1) a Fab fragment, a monovalent fragment having the VL, VH, CL and CH1 domains; (2) a F(ab')2 fragment, a bivalent fragment having two Fab fragments linked by a disulfide bridge at the hinge region; (3) a Fd fragment having the two VH and CH1 domains; (4) a Fv fragment having the VL and VH domains of a single arm of an antibody, (5) a dAb fragment (Ward et al., (1989) Nature 341:544-546), which has a VH domain; (6) an isolated complementarity determining region (CDR), and (7) a single chain Fv (scFv). Although the two domains of the Fv fragment, VL and VH are coded for by separate genes, they can be joined, using recombinant methods, by a synthetic linker that enables them to be made as a single protein chain in which the VL and VH regions pair to form monovalent molecules (known as single chain Fv (scFv); see e.g., Huston et al. (1988) Proc. Natl. Acad. Sci USA 85:5879-5883). These antibody fragments are obtained using conventional techniques known to those with skill in the art, and the fragments are evaluated for function in the same manner as are intact antibodies.
[0085] The term "monoclonal antibody" as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical except for possible naturally occurring mutations and/or post-translation modifications (e.g., isomerizations, amidations) that may be present in minor amounts. Monoclonal antibodies are highly specific, being directed against a single antigenic site. Furthermore, in contrast to conventional (polyclonal) antibody preparations which typically include different antibodies directed against different determinants (epitopes), each monoclonal antibody is directed against a single determinant on the antigen. In addition to their specificity, the monoclonal antibodies are advantageous in that they are synthesized by the hybridoma culture, uncontaminated by other immunoglobulins. The modifier "monoclonal" indicates the character of the antibody as being obtained from a substantially homogeneous population of antibodies, and is not to be construed as requiring production of the antibody by any particular method. For example, the monoclonal antibodies to be used in accordance with the present invention may be made by the hybridoma method first described by Kohler et al., Nature, 256: 495 (1975), or may be made by recombinant DNA methods (see, e.g., U.S. Pat. No. 4,816,567). The "monoclonal antibodies" may also be isolated from phage antibody libraries using the techniques described in Clackson et al., Nature, 352: 624-628 (1991) and Marks et al., J. Mol. Biol., 222: 581-597 (1991), for example.
[0086] The term "human antibody" includes antibodies having variable and constant regions corresponding substantially to human germline immunoglobulin sequences known in the art, including, for example, those described by Kabat et al. (See Kabat et al. (1991) loc. cit.). The human antibodies of the invention may include amino acid residues not encoded by human germline immunoglobulin sequences (e.g., mutations introduced by random or site-specific mutagenesis in vitro or by somatic mutation in vivo), for example in the CDRs, and in particular, CDR3. The human antibody can have at least one, two, three, four, five, or more positions replaced with an amino acid residue that is not encoded by the human germline immunoglobulin sequence. It is emphasized that the definition of human antibodies as used herein also contemplates fully human antibodies, which include only non-artificially and/or genetically altered human sequences of antibodies as those can be derived by technologies using systems such as the Xenomice.
[0087] Examples of "antibody variants" include humanized variants of non-human antibodies, "affinity matured" antibodies (see, e.g. Hawkins et al. J. Mol. Biol. 254, 889-896 (1992) and Lowman et al., Biochemistry 30, 10832-10837 (1991)) and antibody mutants with altered effector function (s) (see, e.g., U.S. Pat. No. 5,648,260, Kontermann and Dubel (2010), loc. cit. and Little (2009), loc. cit.).
[0088] As used herein, "in vitro generated antibody" refers to an antibody where all or part of the variable region (e.g., at least one CDR) is generated in a non-immune cell selection (e.g., an in vitro phage display, protein chip or any other method in which candidate sequences can be tested for their ability to bind to an antigen). This term thus preferably excludes sequences generated by genomic rearrangement in an immune cell.
[0089] The pairing of a VH and VL together forms a single antigen-binding site. The CH domain most proximal to VH is designated as CH1. Each L chain is linked to an H chain by one covalent disulfide bond, while the two H chains are linked to each other by one or more disulfide bonds depending on the H chain isotype. The VH and VL domains consist of four regions of relatively conserved sequences called framework regions (FR1, FR2, FR3, and FR4), which form a scaffold for three regions of hypervariable sequences (complementarity determining regions, CDRs). The CDRs contain most of the residues responsible for specific interactions of the antibody with the antigen. CDRs are referred to as CDR 1, CDR2, and CDR3. Accordingly, CDR constituents on the heavy chain are referred to as H1, H2, and H3, while CDR constituents on the light chain are referred to as L1, L2, and L3.
[0090] The term "variable" refers to the portions of the immunoglobulin domains that exhibit variability in their sequence and that are involved in determining the specificity and binding affinity of a particular antibody (i.e., the "variable domain(s)"). Variability is not evenly distributed throughout the variable domains of antibodies; it is concentrated in sub-domains of each of the heavy and light chain variable regions. These sub-domains are called "hypervariable" regions or "complementarity determining regions" (CDRs). The more conserved (i.e., non-hypervariable) portions of the variable domains are called the "framework" regions (FRM). The variable domains of naturally occurring heavy and light chains each comprise four FRM regions, largely adopting a .beta.-sheet configuration, connected by three hypervariable regions, which form loops connecting, and in some cases forming part of, the .beta.-sheet structure. The hypervariable regions in each chain are held together in close proximity by the FRM and, with the hypervariable regions from the other chain, contribute to the formation of the antigen-binding site (see Kabat et al., loc. cit.). The constant domains are not directly involved in antigen binding, but exhibit various effector functions, such as, for example, antibody-dependent, cell-mediated cytotoxicity and complement activation.
[0091] The terms "CDR", and its plural "CDRs", refer to a complementarity determining region (CDR) of which three make up the binding character of a light chain variable region (CDRL1, CDRL2 and CDRL3) and three make up the binding character of a heavy chain variable region (CDRH1, CDRH2 and CDRH3). CDRs contribute to the functional activity of an antibody molecule and are separated by amino acid sequences that comprise scaffolding or framework regions. The exact definitional CDR boundaries and lengths are subject to different classification and numbering systems. CDRs may therefore be referred to by Kabat, Chothia, contact or any other boundary definitions, including the numbering system described herein. Despite differing boundaries, each of these systems has some degree of overlap in what constitutes the so called "hypervariable regions" within the variable sequences. CDR definitions according to these systems may therefore differ in length and boundary areas with respect to the adjacent framework region. See for example Kabat, Chothia, and/or MacCallum (Kabat et al., loc. cit.; Chothia et al., J. Mol. Biol, 1987, 196: 901; and MacCallum et al., J. Mol. Biol, 1996, 262: 732). However, the numbering in accordance with the so-called Kabat system is preferred. The CDR3 of the light chain and, particularly, CDR3 of the heavy chain may constitute the most important determinants in antigen binding within the light and heavy chain variable regions. In some antibodies, the heavy chain CDR3 appears to constitute the major area of contact between the antigen and the antibody. In vitro selection schemes in which CDR3 alone is varied can be used to vary the binding properties of an antibody or determine which residues contribute to the binding of an antigen.
[0092] In one embodiment, the antibody of the invention may comprise from one to six of the exemplary CDRs described herein. The antibodies of the invention may be of any type including IgM, IgG (including IgG1, IgG2, IgG3, IgG4), IgD, IgA, or IgE antibody. In a specific embodiment the antigen binding protein is an IgG type antibody, e.g., a IgG1 antibody.
[0093] In one embodiment, the antibody of the invention may be a mutlispecific antibody, and notably a bispecfic antibody, also sometimes referred to as "diabodies." These are antibodies that bind to two or more different antigens or different epitopes on a single antigen. In certain embodiments, a bispecific antibody binds CDH19 and an antigen on a human effector cell (e.g., T cell). Such antibodies are useful in targeting an effector cell response against a CDH19 expressing cells, such as a tumor cell. In preferred embodiments, the human effector cell antigen is CD3 (see corresponding formats e.g. in WO 2008/119567. Methods of making bispecific antibodies are known in the art. One such method involves engineering the Fc portion of the heavy chains such as to create "knobs" and "holes" which facilitate heterodimer formation of the heavy chains when co-expressed in a cell. U.S. Pat. No. 7,695,963. Another method also involves engineering the Fc portion of the heavy chain but uses electrostatic steering to encourage heterodimer formation while discouraging homodimer formation of the heavy chains when co-expressed in a cell. WO 2009/089004, which is incorporated herein by reference in its entirety.
[0094] In one embodiment, antibody of the invention is a minibody. Minibodies are minimized antibody-like proteins comprising a scFv joined to a CH3 domain. Hu et al., 1996, Cancer Res. 56:3055-3061.
[0095] In one embodiment, the antibody of the invention is a domain antibody; see, for example U.S. Pat. No. 6,248,516. Domain antibodies (dAbs) are functional binding domains of antibodies, corresponding to the variable regions of either the heavy (VH) or light (VL) chains of human antibodies. dABs have a molecular weight of approximately 13 kDa, or less than one-tenth the size of a full antibody. dABs are well expressed in a variety of hosts including bacterial, yeast, and mammalian cell systems. In addition, dAbs are highly stable and retain activity even after being subjected to harsh conditions, such as freeze-drying or heat denaturation. See, for example, U.S. Pat. Nos. 6,291,158; 6,582,915; 6,593,081; 6,172,197; US Serial No. 2004/0110941; European Patent 0368684; U.S. Pat. No. 6,696,245, WO04/058821, WO04/003019 and WO03/002609.
[0096] In one embodiment, the antibody of the invention is an antibody fragment, that is a fragment of any of the antibodies outlined herein that retain binding specificity to CDH19. In various embodiments, the antibody binding proteins comprise, but are not limited to, a F(ab), F(ab'), F(ab')2, Fv, or a single chain Fv fragments. At a minimum, an antibody, as meant herein, comprises a polypeptide that can bind specifically to CDH19 comprising all or part of a light or heavy chain variable region, such as one or more CDRs.
[0097] Naturally occurring antibodies typically include a signal sequence, which directs the antibody into the cellular pathway for protein secretion and which is typically not present in the mature antibody. A polynucleotide encoding an antibody of the invention may encode a naturally occurring a signal sequence or a heterologous signal sequence as described below.
[0098] "Consisting essentially of" means that the amino acid sequence can vary by about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15% relative to the recited SEQ ID NO: sequence and still retain biological activity, as described herein.
[0099] In some embodiments, the antibodies of the invention are isolated proteins or substantially pure proteins. An "isolated" protein is unaccompanied by at least some of the material with which it is normally associated in its natural state, for example constituting at least about 5%, or at least about 50% by weight of the total protein in a given sample. It is understood that the isolated protein may constitute from 5 to 99.9% by weight of the total protein content depending on the circumstances. For example, the protein may be made at a significantly higher concentration through the use of an inducible promoter or high expression promoter, such that the protein is made at increased concentration levels. The definition includes the production of an antigen binding protein in a wide variety of organisms and/or host cells that are known in the art.
[0100] For amino acid sequences, sequence identity and/or similarity is determined by using standard techniques known in the art, including, but not limited to, the local sequence identity algorithm of Smith and Waterman, 1981, Adv. Appl. Math. 2:482, the sequence identity alignment algorithm of Needleman and Wunsch, 1970, J. Mol. Biol. 48:443, the search for similarity method of Pearson and Lipman, 1988, Proc. Nat. Acad. Sci. U.S.A. 85:2444, computerized implementations of these algorithms (GAP, BESTFIT, FASTA, and TFASTA in the Wisconsin Genetics Software Package, Genetics Computer Group, 575 Science Drive, Madison, Wis.), the Best Fit sequence program described by Devereux et al., 1984, Nucl. Acid Res. 12:387-395, preferably using the default settings, or by inspection. Preferably, percent identity is calculated by FastDB based upon the following parameters: mismatch penalty of 1; gap penalty of 1; gap size penalty of 0.33; and joining penalty of 30, "Current Methods in Sequence Comparison and Analysis," Macromolecule Sequencing and Synthesis, Selected Methods and Applications, pp 127-149 (1988), Alan R. Liss, Inc.
[0101] An example of a useful algorithm is PILEUP. PILEUP creates a multiple sequence alignment from a group of related sequences using progressive, pairwise alignments. It can also plot a tree showing the clustering relationships used to create the alignment. PILEUP uses a simplification of the progressive alignment method of Feng & Doolittle, 1987, J. Mol. Evol. 35:351-360; the method is similar to that described by Higgins and Sharp, 1989, CABIOS 5:151-153. Useful PILEUP parameters including a default gap weight of 3.00, a default gap length weight of 0.10, and weighted end gaps.
[0102] Another example of a useful algorithm is the BLAST algorithm, described in: Altschul et al., 1990, J. Mol. Biol. 215:403-410; Altschul et al., 1997, Nucleic Acids Res. 25:3389-3402; and Karin et al., 1993, Proc. Natl. Acad. Sci. U.S.A. 90:5873-5787. A particularly useful BLAST program is the WU-BLAST-2 program which was obtained from Altschul et al., 1996, Methods in Enzymology 266:460-480. WU-BLAST-2 uses several search parameters, most of which are set to the default values. The adjustable parameters are set with the following values: overlap span=1, overlap fraction=0.125, word threshold (T)=II. The HSP S and HSP S2 parameters are dynamic values and are established by the program itself depending upon the composition of the particular sequence and composition of the particular database against which the sequence of interest is being searched; however, the values may be adjusted to increase sensitivity.
[0103] An additional useful algorithm is gapped BLAST as reported by Altschul et al., 1993, Nucl. Acids Res. 25:3389-3402. Gapped BLAST uses BLOSUM-62 substitution scores; threshold T parameter set to 9; the two-hit method to trigger ungapped extensions, charges gap lengths of k a cost of 10+k; Xu set to 16, and Xg set to 40 for database search stage and to 67 for the output stage of the algorithms. Gapped alignments are triggered by a score corresponding to about 22 bits.
[0104] Generally, the amino acid homology, similarity, or identity between individual variant CDRs are at least 80% to the sequences depicted herein, and more typically with preferably increasing homologies or identities of at least 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, and almost 100%. In a similar manner, "percent (%) nucleic acid sequence identity" with respect to the nucleic acid sequence of the binding proteins identified herein is defined as the percentage of nucleotide residues in a candidate sequence that are identical with the nucleotide residues in the coding sequence of the antigen binding protein. A specific method utilizes the BLASTN module of WU-BLAST-2 set to the default parameters, with overlap span and overlap fraction set to 1 and 0.125, respectively.
[0105] Generally, the nucleic acid sequence homology, similarity, or identity between the nucleotide sequences encoding individual variant CDRs and the nucleotide sequences depicted herein are at least 80%, and more typically with preferably increasing homologies or identities of at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99%, and almost 100%.
[0106] Thus, a "variant CDR" is one with the specified homology, similarity, or identity to the parent CDR of the invention, and shares biological function, including, but not limited to, at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% of the specificity and/or activity of the parent CDR.
[0107] While the site or region for introducing an amino acid sequence variation is predetermined, the mutation per se need not be predetermined. For example, in order to optimize the performance of a mutation at a given site, random mutagenesis may be conducted at the target codon or region and the expressed antigen binding protein CDR variants screened for the optimal combination of desired activity. Techniques for making substitution mutations at predetermined sites in DNA having a known sequence are well known, for example, M13 primer mutagenesis and PCR mutagenesis. Screening of the mutants is done using assays of antigen binding protein activities, such as CDH19 binding.
[0108] The term "amino acid" or "amino acid residue" typically refers to an amino acid having its art recognized definition such as an amino acid selected from the group consisting of: alanine (Ala or A); arginine (Arg or R); asparagine (Asn or N); aspartic acid (Asp or D); cysteine (Cys or C); glutamine (Gln or Q); glutamic acid (Glu or E); glycine (Gly or G); histidine (His or H); isoleucine (He or I): leucine (Leu or L); lysine (Lys or K); methionine (Met or M); phenylalanine (Phe or F); pro line (Pro or P); serine (Ser or S); threonine (Thr or T); tryptophan (Trp or W); tyrosine (Tyr or Y); and valine (Val or V), although modified, synthetic, or rare amino acids may be used as desired. Generally, amino acids can be grouped as having a nonpolar side chain (e.g., Ala, Cys, He, Leu, Met, Phe, Pro, Val); a negatively charged side chain (e.g., Asp, Glu); a positively charged sidechain (e.g., Arg, His, Lys); or an uncharged polar side chain (e.g., Asn, Cys, Gln, Gly, His, Met, Phe, Ser, Thr, Trp, and Tyr).
[0109] The term "hypervariable region" (also known as "complementarity determining regions" or CDRs) when used herein refers to the amino acid residues of an antibody which are (usually three or four short regions of extreme sequence variability) within the V-region domain of an immunoglobulin which form the antigen-binding site and are the main determinants of antigen specificity. There are at least two methods for identifying the CDR residues: (1) An approach based on cross-species sequence variability (i. e., Kabat et al., loc. cit.); and (2) An approach based on crystallographic studies of antigen-antibody complexes (Chothia, C. et al., J. Mol. Biol. 196: 901-917 (1987)). However, to the extent that two residue identification techniques define regions of overlapping, but not identical regions, they can be combined to define a hybrid CDR. However, in general, the CDR residues are preferably identified in accordance with the so-called Kabat (numbering) system.
[0110] The term "framework region" refers to the art-recognized portions of an antibody variable region that exist between the more divergent (i.e., hypervariable) CDRs. Such framework regions are typically referred to as frameworks 1 through 4 (FR1, FR2, FR3, and FR4) and provide a scaffold for the presentation of the six CDRs (three from the heavy chain and three from the light chain) in three dimensional space, to form an antigen-binding surface.
[0111] Typically, CDRs form a loop structure that can be classified as a canonical structure. The term "canonical structure" refers to the main chain conformation that is adopted by the antigen binding (CDR) loops. From comparative structural studies, it has been found that five of the six antigen binding loops have only a limited repertoire of available conformations. Each canonical structure can be characterized by the torsion angles of the polypeptide backbone. Correspondent loops between antibodies may, therefore, have very similar three dimensional structures, despite high amino acid sequence variability in most parts of the loops (Chothia and Lesk, J. Mol. Biol., 1987, 196: 901; Chothia et al., Nature, 1989, 342: 877; Martin and Thornton, J. Mol. Biol, 1996, 263: 800, each of which is incorporated by reference in its entirety). Furthermore, there is a relationship between the adopted loop structure and the amino acid sequences surrounding it. The conformation of a particular canonical class is determined by the length of the loop and the amino acid residues residing at key positions within the loop, as well as within the conserved framework (i.e., outside of the loop). Assignment to a particular canonical class can therefore be made based on the presence of these key amino acid residues. The term "canonical structure" may also include considerations as to the linear sequence of the antibody, for example, as catalogued by Kabat (Kabat et al., loc. cit.). The Kabat numbering scheme (system) is a widely adopted standard for numbering the amino acid residues of an antibody variable domain in a consistent manner and is the preferred scheme applied in the present invention as also mentioned elsewhere herein. Additional structural considerations can also be used to determine the canonical structure of an antibody. For example, those differences not fully reflected by Kabat numbering can be described by the numbering system of Chothia et al and/or revealed by other techniques, for example, crystallography and two or three-dimensional computational modeling. Accordingly, a given antibody sequence may be placed into a canonical class which allows for, among other things, identifying appropriate chassis sequences (e.g., based on a desire to include a variety of canonical structures in a library). Kabat numbering of antibody amino acid sequences and structural considerations as described by Chothia et al., loc. cit. and their implications for construing canonical aspects of antibody structure, are described in the literature.
[0112] CDR3 is typically the greatest source of molecular diversity within the antibody-binding site. H3, for example, can be as short as two amino acid residues or greater than 26 amino acids. The subunit structures and three-dimensional configurations of different classes of immunoglobulins are well known in the art. For a review of the antibody structure, see Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory, eds. Harlow et al., 1988. One of skill in the art will recognize that each subunit structure, e.g., a CH, VH, CL, VL, CDR, FR structure, comprises active fragments, e.g., the portion of the VH, VL, or CDR subunit the binds to the antigen, i.e., the antigen-binding fragment, or, e.g., the portion of the CH subunit that binds to and/or activates, e.g., an Fc receptor and/or complement. The CDRs typically refer to the Kabat CDRs, as described in Sequences of Proteins of immunological Interest, US Department of Health and Human Services (1991), eds. Kabat et al. Another standard for characterizing the antigen binding site is to refer to the hypervariable loops as described by Chothia. See, e.g., Chothia, et al. (1987; J. Mol. Biol. 227:799-817); and Tomlinson et al. (1995) EMBO J. 14: 4628-4638. Still another standard is the AbM definition used by Oxford Molecular's AbM antibody modeling software. See, generally, e.g., Protein Sequence and Structure Analysis of Antibody Variable Domains. In: Antibody Engineering Lab Manual (Ed.: Duebel, S. and Kontermann, R., Springer-Verlag, Heidelberg). Embodiments described with respect to Kabat CDRs can alternatively be implemented using similar described relationships with respect to Chothia hypervariable loops or to the AbM-defined loops.
[0113] The sequence of antibody genes after assembly and somatic mutation is highly varied, and these varied genes are estimated to encode 10.sup.10 different antibody molecules (Immunoglobulin Genes, 2.sup.nd ed., eds. Jonio et al., Academic Press, San Diego, Calif., 1995). Accordingly, the immune system provides a repertoire of immunoglobulins. The term "repertoire" refers to at least one nucleotide sequence derived wholly or partially from at least one sequence encoding at least one immunoglobulin. The sequence(s) may be generated by rearrangement in vivo of the V, D, and J segments of heavy chains, and the V and J segments of light chains. Alternatively, the sequence(s) can be generated from a cell in response to which rearrangement occurs, e.g., in vitro stimulation. Alternatively, part or all of the sequence(s) may be obtained by DNA splicing, nucleotide synthesis, mutagenesis, and other methods, see, e.g., U.S. Pat. No. 5,565,332. A repertoire may include only one sequence or may include a plurality of sequences, including ones in a genetically diverse collection.
[0114] The term "binding molecule" or "antibody construct" in the sense of the present disclosure indicates any molecule capable of (specifically) binding to, interacting with or recognizing the target molecule CDH19. Such molecules or constructs may include proteinaceous parts and non-proteinaceous parts (e.g. chemical linkers or chemical cross-linking agents such as glutaraldehyde).
[0115] The term "multispecific" as used herein refers to a binding molecule which is an antibody construct and comprises at least a first and a second binding domain, wherein the first binding domain is capable of binding to one antigen or target, and the second binding domain is capable of binding to another antigen or target. Accordingly, antibody constructs according to the invention comprise at least a specificity for CDH19. The "antibody construct" of the invention also comprises multispecific binding molecules such as e.g. trispecific binding molecules, the latter ones including three binding domains.
[0116] It is also envisaged that the antibody construct of the invention has, in addition to its function to bind to the target molecules CDH19 and CD3, a further function. In this format, the antibody construct is a bi-, tri- or multifunctional antibody construct by targeting plasma cells through binding to CDH19, mediating cytotoxic T cell activity through CD3 binding and providing a further function such as a fully functional Fc constant domain mediating antibody-dependent cellular cytotoxicity through recruitment of effector cells like NK cells, a label (fluorescent etc.), a therapeutic agent such as, e.g. a toxin or radionuclide, and/or means to enhance serum half-life, etc.
[0117] The term "binding domain" characterizes in connection with the present invention a domain which is capable of specifically binding to/interacting with a given target epitope or a given target site on the target molecule CDH19.
[0118] Binding domains can be derived from a binding domain donor such as for example an antibody. It is envisaged that a binding domain of the present invention comprises at least said part of any of the aforementioned binding domains that is required for binding to/interacting with a given target epitope or a given target site on the target molecule CDH19.
[0119] It is envisaged that the binding domain of the aforementioned binding domain donors is characterized by that part of these donors that is responsible for binding the respective target, i.e. when that part is removed from the binding domain donor, said donor loses its binding capability. "Loses" means a reduction of at least 50% of the binding capability when compared with the binding donor. Methods to map these binding sites are well known in the art--it is therefore within the standard knowledge of the skilled person to locate/map the binding site of a binding domain donor and, thereby, to "derive" said binding domain from the respective binding domain donors.
[0120] The term "epitope" refers to a site on an antigen to which a binding domain, such as an antibody or immunoglobulin or derivative or fragment of an antibody or of an immunoglobulin, specifically binds. An "epitope" is antigenic and thus the term epitope is sometimes also referred to herein as "antigenic structure" or "antigenic determinant". Thus, the binding domain is an "antigen-interaction-site". Said binding/interaction is also understood to define a "specific recognition". In one example, said binding domain which (specifically) binds to/interacts with a given target epitope or a given target site on the target molecule CDH19 is an antibody or immunoglobulin, and said binding domain is a VH and/or VL region of an antibody or of an immunoglobulin.
[0121] "Epitopes" can be formed both by contiguous amino acids or non-contiguous amino acids juxtaposed by tertiary folding of a protein. A "linear epitope" is an epitope where an amino acid primary sequence comprises the recognized epitope. A linear epitope typically includes at least 3 or at least 4, and more usually, at least 5 or at least 6 or at least 7, for example, about 8 to about 10 amino acids in a unique sequence.
[0122] A "conformational epitope", in contrast to a linear epitope, is an epitope wherein the primary sequence of the amino acids comprising the epitope is not the sole defining component of the epitope recognized (e.g., an epitope wherein the primary sequence of amino acids is not necessarily recognized by the binding domain). Typically a conformational epitope comprises an increased number of amino acids relative to a linear epitope. With regard to recognition of conformational epitopes, the binding domain recognizes a three-dimensional structure of the antigen, preferably a peptide or protein or fragment thereof (in the context of the present invention, the antigen for one of the binding domains is comprised within the CDH19 protein). For example, when a protein molecule folds to form a three-dimensional structure, certain amino acids and/or the polypeptide backbone forming the conformational epitope become juxtaposed enabling the antibody to recognize the epitope. Methods of determining the conformation of epitopes include, but are not limited to, x-ray crystallography, two-dimensional nuclear magnetic resonance (2D-NMR) spectroscopy and site-directed spin labelling and electron paramagnetic resonance (EPR) spectroscopy. Moreover, the provided examples describe a further method to characterize a given binding domain by way of binning, which includes a test whether the given binding domain binds to one or more epitope cluster(s) of a given protein, in particular CDH19.
[0123] As used herein, the term "epitope cluster" denotes the entirety of epitopes lying in a defined contiguous stretch of an antigen. An epitope cluster can comprise one, two or more epitopes. The concept of epitope cluster is also used in the characterization of the features of the antibody or antigen binding fragment thereof of the invention.
[0124] The terms "(capable of) binding to", "specifically recognizing", "directed to" and "reacting with" mean in accordance with this invention that a binding domain is capable of specifically interacting with one or more, preferably at least two, more preferably at least three and most preferably at least four amino acids of an epitope.
[0125] As used herein, the terms "specifically interacting", "specifically binding" or "specifically bind(s)" mean that a binding domain exhibits appreciable affinity for a particular protein or antigen and, generally, does not exhibit significant reactivity with proteins or antigens other than CDH19. "Appreciable affinity" includes binding with an affinity of about 10.sup.-6M (KD) or stronger. Preferably, binding is considered specific when binding affinity is about 10.sup.-12 to 10.sup.-8 M, 10.sup.-12 to 10.sup.-9 M, 10.sup.-12 to 10.sup.-10 M, 10.sup.-11 to 10.sup.-8 M, preferably of about 10.sup.-11 to 10.sup.-9 M. Whether a binding domain specifically reacts with or binds to a target can be tested readily by, inter alia, comparing the reaction of said binding domain with a target protein or antigen with the reaction of said binding domain with proteins or antigens other than CDH19. Preferably, a binding domain of the invention does not essentially bind or is not capable of binding to proteins or antigens other than CDH19.
[0126] The term "does not essentially bind", or "is not capable of binding" means that a binding domain of the present invention does not bind another protein or antigen other than CDH19, i.e., does not show reactivity of more than 30%, preferably not more than 20%, more preferably not more than 10%, particularly preferably not more than 9%, 8%, 7%, 6% or 5% with proteins or antigens other than CDH19, whereby binding to CDH19, respectively, is set to be 100%.
[0127] Specific binding is believed to be effected by specific motifs in the amino acid sequence of the binding domain and the antigen. Thus, binding is achieved as a result of their primary, secondary and/or tertiary structure as well as the result of secondary modifications of said structures. The specific interaction of the antigen-interaction-site with its specific antigen may result in a simple binding of said site to the antigen. Moreover, the specific interaction of the antigen-interaction-site with its specific antigen may alternatively or additionally result in the initiation of a signal, e.g. due to the induction of a change of the conformation of the antigen, an oligomerization of the antigen, etc.
[0128] Proteins (including fragments thereof, preferably biologically active fragments, and peptides, usually having less than 30 amino acids) comprise one or more amino acids coupled to each other via a covalent peptide bond (resulting in a chain of amino acids). The term "polypeptide" as used herein describes a group of molecules, which consist of more than 30 amino acids. Polypeptides may further form multimers such as dimers, trimers and higher oligomers, i.e. consisting of more than one polypeptide molecule. Polypeptide molecules forming such dimers, trimers etc. may be identical or non-identical. The corresponding higher order structures of such multimers are, consequently, termed homo- or heterodimers, homo- or heterotrimers etc. An example for a hereteromultimer is an antibody molecule, which, in its naturally occurring form, consists of two identical light polypeptide chains and two identical heavy polypeptide chains. The terms "polypeptide" and "protein" also refer to naturally modified polypeptides/proteins wherein the modification is effected e.g. by post-translational modifications like glycosylation, acetylation, phosphorylation and the like. A "polypeptide" when referred to herein may also be chemically modified such as pegylated. Such modifications are well known in the art.
[0129] "Isolated" when used to describe the antibody or antigen binding fragment thereof or antibody construct disclosed herein, refers to the antibody or antigen binding fragment thereof or antibody construct disclosed herein that has been identified, separated and/or recovered from a component of its production environment. Preferably, the isolated the antibody or antigen binding fragment thereof or antibody construct disclosed herein is free of association with all other components from its production environment. Contaminant components of its production environment, such as that resulting from recombinant transfected cells, are materials that would typically interfere with diagnostic or therapeutic uses for the polypeptide, and may include enzymes, hormones, and other proteinaceous or non-proteinaceous solutes. In preferred embodiments, the the antibody or antigen binding fragment thereof or antibody construct will be purified (1) to a degree sufficient to obtain at least 15 residues of N-terminal or internal amino acid sequence by use of a spinning cup sequenator, or (2) to homogeneity by SDS-PAGE under non-reducing or reducing conditions using Coomassie blue or, preferably, silver stain. Ordinarily, however, an isolated antibody will be prepared by at least one purification step.
[0130] Amino acid sequence modifications of the the antibody or antigen binding fragment thereof or antibody construct described herein are contemplated. For example, it may be desirable to improve the binding affinity and/or other biological properties of the antibody. Amino acid sequence variants of the the antibody or antigen binding fragment thereof or antibody construct disclosed herein are prepared by introducing appropriate nucleotide changes into the the antibody or antigen binding fragment thereof or antibody construct nucleic acid, or by peptide synthesis.
[0131] Such modifications include, for example, deletions from, and/or insertions into, and/or substitutions of, residues within the amino acid sequences of the the antibody or antigen binding fragment thereof or antibody construct disclosed herein. Any combination of deletion, insertion, and substitution is made to arrive at the final construct, provided that the final construct possesses the desired characteristics. The amino acid changes also may alter post-translational processes of the the antibody or antigen binding fragment thereof or antibody construct disclosed herein, such as changing the number or position of glycosylation sites. Preferably, 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids may be substituted in a CDR, while 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or 25 amino acids may be substituted in the framework regions (FRs). The substitutions are preferably conservative substitutions as described herein. Additionally or alternatively, 1, 2, 3, 4, 5, or 6 amino acids may be inserted or deleted in each of the CDRs (of course, dependent on their length), while 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or 25 amino acids may be inserted or deleted in each of the FRs.
[0132] A useful method for identification of certain residues or regions of the the antibody or antigen binding fragment thereof or antibody construct disclosed herein that are preferred locations for mutagenesis is called "alanine scanning mutagenesis" as described by Cunningham and Wells in Science, 244: 1081-1085 (1989). Here, a residue or group of target residues within the the antibody or antigen binding fragment thereof or antibody construct disclosed herein is/are identified (e.g. charged residues such as arg, asp, his, lys, and glu) and replaced by a neutral or negatively charged amino acid (most preferably alanine or polyalanine) to affect the interaction of the amino acids with the epitope.
[0133] Those amino acid locations demonstrating functional sensitivity to the substitutions then are refined by introducing further or other variants at, or for, the sites of substitution. Thus, while the site for introducing an amino acid sequence variation is predetermined, the nature of the mutation per se needs not to be predetermined. For example, to analyze the performance of a mutation at a given site, ala scanning or random mutagenesis is conducted at a target codon or region and the expressed the antibody or antigen binding fragment thereof or antibody construct disclosed herein variants are screened for the desired activity.
[0134] Preferably, amino acid sequence insertions include amino- and/or carboxyl-terminal fusions ranging in length from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 residues to polypeptides containing a hundred or more residues, as well as intrasequence insertions of single or multiple amino acid residues. An insertional variant of the the antibody or antigen binding fragment thereof or antibody construct disclosed herein includes the fusion to the N- or C-terminus of the antibody to an enzyme or a fusion to a polypeptide which increases the serum half-life of the antibody.
[0135] Another type of variant is an amino acid substitution variant. These variants have preferably at least 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid residues in the the antibody or antigen binding fragment thereof or antibody construct disclosed herein replaced by a different residue. The sites of greatest interest for substitutional mutagenesis include the CDRs of the heavy and/or light chain, in particular the hypervariable regions, but FR alterations in the heavy and/or light chain are also contemplated.
[0136] For example, if a CDR sequence encompasses 6 amino acids, it is envisaged that one, two or three of these amino acids are substituted. Similarly, if a CDR sequence encompasses 15 amino acids it is envisaged that one, two, three, four, five or six of these amino acids are substituted.
[0137] Generally, if amino acids are substituted in one or more or all of the CDRs of the heavy and/or light chain, it is preferred that the then-obtained "substituted" sequence is at least 60%, more preferably 65%, even more preferably 70%, particularly preferably 75%, more particularly preferably 80% identical to the "original" CDR sequence. This means that it is dependent of the length of the CDR to which degree it is identical to the "substituted" sequence. For example, a CDR having 5 amino acids is preferably 80% identical to its substituted sequence in order to have at least one amino acid substituted. Accordingly, the CDRs of the the antibody or antigen binding fragment thereof or antibody construct disclosed herein may have different degrees of identity to their substituted sequences, e.g., CDRL1 may have 80%, while CDRL3 may have 90%.
[0138] Preferred substitutions (or replacements) are conservative substitutions. However, any substitution (including non-conservative substitution or one or more from the "exemplary substitutions" listed in Table 1, below) is envisaged as long as the the antibody or antigen binding fragment thereof or antibody construct retains its capability to bind to CDH19 v and/or its CDRs have an identity to the then substituted sequence (at least 60%, more preferably 65%, even more preferably 70%, particularly preferably 75%, more particularly preferably 80% identical to the "original" CDR sequence).
[0139] Conservative substitutions are shown in Table 1 under the heading of "preferred substitutions". If such substitutions result in a change in biological activity, then more substantial changes, denominated "exemplary substitutions" in Table 1, or as further described below in reference to amino acid classes, may be introduced and the products screened for a desired characteristic.
TABLE-US-00001 TABLE 1 Amino Acid Substitutions Preferred Original Exemplary Substitutions Substitutions Ala (A) val, leu, ile val Arg (R) lys, gin, asn lys Asn (N) gin, his, asp, lys, arg gin Asp (D) glu, asn glu Cys (C) ser, ala ser Gln (Q) asn, glu asn Glu (E) asp, gin Asp Gly (G) ala Ala His (H) asn, gin, lys, arg Arg Ile (I) leu, val, met, ala, phe Leu Leu (L) norleucine, ile, val, met, ala Ile Lys (K) arg, gin, asn Arg Met (M) leu, phe, ile Leu Phe (F) leu, val, ile, ala, tyr Tyr Pro (P) ala Ala Ser (S) thr Thr Thr (T) ser Ser Trp (W) tyr, phe Tyr Tyr (Y) trp, phe, thr, ser Phe Val (V) ile, leu, met, phe, ala Leu
[0140] Substantial modifications in the biological properties of the the antibody or antigen binding fragment thereof or antibody construct of the present invention are accomplished by selecting substitutions that differ significantly in their effect on maintaining (a) the structure of the polypeptide backbone in the area of the substitution, for example, as a sheet or helical conformation, (b) the charge or hydrophobicity of the molecule at the target site, or (c) the bulk of the side chain. Naturally occurring residues are divided into groups based on common side-chain properties: (1) hydrophobic: norleucine, met, ala, val, leu, ile; (2) neutral hydrophilic: cys, ser, thr; (3) acidic: asp, glu; (4) basic: asn, gin, his, lys, arg; (5) residues that influence chain orientation: gly, pro; and (6) aromatic: trp, tyr, phe.
[0141] Non-conservative substitutions will entail exchanging a member of one of these classes for another class. Any cysteine residue not involved in maintaining the proper conformation of the the antibody or antigen binding fragment thereof or antibody construct may be substituted, generally with serine, to improve the oxidative stability of the molecule and prevent aberrant crosslinking. Conversely, cysteine bond(s) may be added to the antibody to improve its stability (particularly where the antibody is an antibody fragment such as an Fv fragment).
[0142] A particularly preferred type of substitutional variant involves substituting one or more hypervariable region residues of a parent antibody (e. g. a humanized or human antibody). Generally, the resulting variant(s) selected for further development will have improved biological properties relative to the parent antibody from which they are generated. A convenient way for generating such substitutional variants involves affinity maturation using phage display. Briefly, several hypervariable region sites (e. g. 6-7 sites) are mutated to generate all possible amino acid substitutions at each site. The antibody variants thus generated are displayed in a monovalent fashion from filamentous phage particles as fusions to the gene III product of M13 packaged within each particle. The phage-displayed variants are then screened for their biological activity (e. g. binding affinity) as herein disclosed. In order to identify candidate hypervariable region sites for modification, alanine scanning mutagenesis can be performed to identify hypervariable region residues contributing significantly to antigen binding. Alternatively, or additionally, it may be beneficial to analyze a crystal structure of the antigen-antibody complex to identify contact points between the binding domain and, e.g., human CDH19. Such contact residues and neighbouring residues are candidates for substitution according to the techniques elaborated herein. Once such variants are generated, the panel of variants is subjected to screening as described herein and antibodies with superior properties in one or more relevant assays may be selected for further development.
[0143] Other modifications of the the antibody or antigen binding fragment thereof or antibody construct are contemplated herein. For example, the the antibody or antigen binding fragment thereof or antibody construct may be linked to one of a variety of non-proteinaceous polymers, e.g., polyethylene glycol, polypropylene glycol, polyoxyalkylenes, or copolymers of polyethylene glycol and polypropylene glycol. The the antibody or antigen binding fragment thereof or antibody construct may also be entrapped in microcapsules prepared, for example, by coacervation techniques or by interfacial polymerization (for example, hydroxymethylcellulose or gelatine-microcapsules and poly (methylmethacylate) microcapsules, respectively), in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nanoparticles and nanocapsules), or in macroemulsions. Such techniques are disclosed in Remington's Pharmaceutical Sciences, 16th edition, Oslo, A., Ed., (1980).
[0144] The the antibody or antigen binding fragment thereof or antibody construct disclosed herein may also be formulated as immuno-liposomes. A "liposome" is a small vesicle composed of various types of lipids, phospholipids and/or surfactant which is useful for delivery of a drug to a mammal. The components of the liposome are commonly arranged in a bilayer formation, similar to the lipid arrangement of biological membranes. Liposomes containing the antibody are prepared by methods known in the art, such as described in Epstein et al., Proc. Natl. Acad. Sci. USA, 82: 3688 (1985); Hwang et al., Proc. Natl Acad. Sci. USA, 77: 4030 (1980); U.S. Pat. Nos. 4,485,045 and 4,544,545; and WO 97/38731 published Oct. 23, 1997. Liposomes with enhanced circulation time are disclosed in U.S. Pat. No. 5,013,556. Particularly useful liposomes can be generated by the reverse phase evaporation method with a lipid composition comprising phosphatidylcholine, cholesterol and PEG-derivatized phosphatidylethanolamine (PEG-PE). Liposomes are extruded through filters of defined pore size to yield liposomes with the desired diameter. Fab' fragments of the antibody of the present invention can be conjugated to the liposomes as described in Martin et al. J. Biol. Chem. 257: 286-288 (1982) via a disulfide interchange reaction. A chemotherapeutic agent is optionally contained within the liposome. See Gabizon et al. J. National Cancer Inst. 81 (19) 1484 (1989).
[0145] When using recombinant techniques, the antibody, antigen binding fragment thereof or antibody construct can be produced intracellularly, in the periplasmic space, or directly secreted into the medium. If the antibody, antigen binding fragment thereof or antibody construct is produced intracellularly, as a first step, the particulate debris, either host cells or lysed fragments, are removed, for example, by centrifugation or ultrafiltration. Carter et al., Bio/Technology 10: 163-167 (1992) describe a procedure for isolating antibodies which are secreted to the periplasmic space of E. coli.
[0146] The antibody, antigen binding fragment thereof or antibody construct composition prepared from the cells can be purified using, for example, hydroxylapatite chromatography, gel electrophoresis, dialysis, and affinity chromatography, with affinity chromatography being the preferred purification technique.
[0147] The term "agent" is used herein to denote a chemical compound, a mixture of chemical compounds, a biological macromolecule, or an extract made from biological materials.
[0148] The term "nucleic acid" is well known to the skilled person and encompasses DNA (such as cDNA) and RNA (such as mRNA). The nucleic acid can be double stranded and single stranded, linear and circular. Said nucleic acid molecule is preferably comprised in a vector which is preferably comprised in a host cell. Said host cell is, e.g. after transformation or transfection with the nucleic acid sequence of the invention, capable of expressing the the antibody or antigen binding fragment thereof or antibody construct disclosed herein. For that purpose the nucleic acid molecule is operatively linked with control sequences.
[0149] A vector is a nucleic acid molecule used as a vehicle to transfer (foreign) genetic material into a cell. The term "vector" encompasses--but is not restricted to--plasmids, viruses, cosmids and artificial chromosomes. In general, engineered vectors comprise an origin of replication, a multicloning site and a selectable marker. The vector itself is generally a nucleotide sequence, commonly a DNA sequence, that comprises an insert (transgene) and a larger sequence that serves as the "backbone" of the vector. Modern vectors may encompass additional features besides the transgene insert and a backbone: promoter, genetic marker, antibiotic resistance, reporter gene, targeting sequence, protein purification tag. Vectors called expression vectors (expression constructs) specifically are for the expression of the transgene in the target cell, and generally have control sequences such as a promoter sequence that drives expression of the transgene. Insertion of a vector into the target cell is usually called "transformation" for bacteria, "transfection" for eukaryotic cells, although insertion of a viral vector is also called "transduction".
[0150] As used herein, the term "host cell" is intended to refer to a cell into which a nucleic acid encoding the the antibody or antigen binding fragment thereof or antibody construct of the invention is introduced by way of transformation, transfection and the like. It should be understood that such terms refer not only to the particular subject cell but to the progeny or potential progeny of such a cell. Because certain modifications may occur in succeeding generations due to either mutation or environmental influences, such progeny may not, in fact, be identical to the parent cell, but are still included within the scope of the term as used herein.
[0151] As used herein, the term "expression" includes any step involved in the production of a the antibody or antigen binding fragment thereof or antibody construct of the invention including, but not limited to, transcription, post-transcriptional modification, translation, post-translational modification, and secretion.
[0152] The term "control sequences" refers to DNA sequences necessary for the expression of an operably linked coding sequence in a particular host organism. The control sequences that are suitable for prokaryotes, for example, include a promoter, optionally an operator sequence, and a ribosome binding site. Eukaryotic cells are known to utilize promoters, polyadenylation signals, and enhancers.
[0153] A nucleic acid is "operably linked" when it is placed into a functional relationship with another nucleic acid sequence. For example, DNA for a presequence or secretory leader is operably linked to DNA for a polypeptide if it is expressed as a preprotein that participates in the secretion of the polypeptide; a promoter or enhancer is operably linked to a coding sequence if it affects the transcription of the sequence; or a ribosome binding site is operably linked to a coding sequence if it is positioned so as to facilitate translation. Generally, "operably linked" means that the DNA sequences being linked are contiguous, and, in the case of a secretory leader, contiguous and in reading phase. However, enhancers do not have to be contiguous. Linking is accomplished by ligation at convenient restriction sites. If such sites do not exist, the synthetic oligonucleotide adaptors or linkers are used in accordance with conventional practice.
[0154] The terms "host cell," "target cell" or "recipient cell" are intended to include any individual cell or cell culture that can be or has/have been recipients for vectors or the incorporation of exogenous nucleic acid molecules, polynucleotides and/or proteins. It also is intended to include progeny of a single cell, and the progeny may not necessarily be completely identical (in morphology or in genomic or total DNA complement) to the original parent cell due to natural, accidental, or deliberate mutation. The cells may be prokaryotic or eukaryotic, and include but are not limited to bacteria, yeast cells, animal cells, and mammalian cells, e.g., murine, rat, macaque or human.
[0155] Suitable host cells include prokaryotes and eukaryotic host cells including yeasts, fungi, insect cells and mammalian cells.
[0156] The the antibody or antigen binding fragment thereof or antibody construct of the invention can be produced in bacteria. After expression, the the antibody or antigen binding fragment thereof or antibody construct of the invention, preferably the the antibody or antigen binding fragment thereof or antibody construct is isolated from the E. coli cell paste in a soluble fraction and can be purified through, e.g., affinity chromatography and/or size exclusion. Final purification can be carried out similar to the process for purifying antibody expressed e. g, in CHO cells.
[0157] In addition to prokaryotes, eukaryotic microbes such as filamentous fungi or yeast are suitable cloning or expression hosts for the the antibody or antigen binding fragment thereof or antibody construct of the invention. Saccharomyces cerevisiae, or common baker's yeast, is the most commonly used among lower eukaryotic host microorganisms. However, a number of other genera, species, and strains are commonly available and useful herein, such as Schizosaccharomyces pombe, Kluyveromyces hosts such as, e.g., K. lactis, K. fragilis (ATCC 12424), K. bulgaricus (ATCC 16045), K. wickeramii (ATCC 24178), K. waltii (ATCC 56500), K. drosophilarum (ATCC 36906), K. thermotolerans, and K. marxianus; yarrowia (EP 402 226); Pichia pastoris (EP 183 070); Candida; Trichoderma reesia (EP 244 234); Neurospora crassa; Schwanniomyces such as Schwanniomyces occidentalis; and filamentous fungi such as, e.g., Neurospora, Penicillium, Tolypocladium, and Aspergillus hosts such as A. nidulans and A. niger.
[0158] Suitable host cells for the expression of glycosylated the antibody or antigen binding fragment thereof or antibody construct of the invention, preferably antibody derived antibody constructs are derived from multicellular organisms. Examples of invertebrate cells include plant and insect cells. Numerous baculoviral strains and variants and corresponding permissive insect host cells from hosts such as Spodoptera frugiperda (caterpillar), Aedes aegypti (mosquito), Aedes albopictus (mosquito), Drosophila melanogaster (fruit fly), and Bombyx mori have been identified. A variety of viral strains for transfection are publicly available, e. g., the L-1 variant of Autographa californica NPV and the Bm-5 strain of Bombyx mori NPV, and such viruses may be used as the virus herein according to the present invention, particularly for transfection of Spodoptera frugiperda cells.
[0159] Plant cell cultures of cotton, corn, potato, soybean, petunia, tomato, Arabidopsis and tobacco can also be utilized as hosts. Cloning and expression vectors useful in the production of proteins in plant cell culture are known to those of skill in the art. See e.g. Hiatt et al., Nature (1989) 342: 76-78, Owen et al. (1992) Bio/Technology 10: 790-794, Artsaenko et al. (1995) The Plant J 8: 745-750, and Fecker et al. (1996) Plant Mol Biol 32: 979-986.
[0160] However, interest has been greatest in vertebrate cells, and propagation of vertebrate cells in culture (tissue culture) has become a routine procedure. Examples of useful mammalian host cell lines are monkey kidney CV1 line transformed by SV40 (COS-7, ATCC CRL 1651); human embryonic kidney line (293 or 293 cells subcloned for growth in suspension culture, Graham et al., J. Gen Virol. 36: 59 (1977)); baby hamster kidney cells (BHK, ATCC CCL 10); Chinese hamster ovary cells/-DHFR (CHO, Urlaub et al., Proc. Natl. Acad. Sci. USA 77: 4216 (1980)); mouse sertoli cells (TM4, Mather, Biol. Reprod. 23: 243-251 (1980)); monkey kidney cells (CVI ATCC CCL 70); African green monkey kidney cells (VERO-76, ATCC CRL1587); human cervical carcinoma cells (HELA, ATCC CCL 2); canine kidney cells (MDCK, ATCC CCL 34); buffalo rat liver cells (BRL 3A, ATCC CRL 1442); human lung cells (W138, ATCC CCL 75); human liver cells (Hep G2,1413 8065); mouse mammary tumor (MMT 060562, ATCC CCLS 1); TRI cells (Mather et al., Annals N. Y Acad. Sci. 383: 44-68 (1982)); MRC 5 cells; FS4 cells; and a human hepatoma line (Hep G2).
[0161] When using recombinant techniques, the antibody or antigen binding fragment thereof or antibody construct of the invention can be produced intracellularly, in the periplasmic space, or directly secreted into the medium. If the the antibody or antigen binding fragment thereof or antibody construct is produced intracellularly, as a first step, the particulate debris, either host cells or lysed fragments, are removed, for example, by centrifugation or ultrafiltration. Carter et al., Bio/Technology 10: 163-167 (1992) describe a procedure for isolating antibodies which are secreted to the periplasmic space of E. coli. Briefly, cell paste is thawed in the presence of sodium acetate (pH 3.5), EDTA, and phenylmethylsulfonylfluoride (PMSF) over about 30 min. Cell debris can be removed by centrifugation. Where the antibody is secreted into the medium, supernatants from such expression systems are generally first concentrated using a commercially available protein concentration filter, for example, an Amicon or Millipore Pellicon ultrafiltration unit. A protease inhibitor such as PMSF may be included in any of the foregoing steps to inhibit proteolysis and antibiotics may be included to prevent the growth of adventitious contaminants.
[0162] The the antibody or antigen binding fragment thereof or antibody construct of the invention prepared from the host cells can be purified using, for example, hydroxylapatite chromatography, gel electrophoresis, dialysis, and affinity chromatography, with affinity chromatography being the preferred purification technique.
[0163] The matrix to which the affinity ligand is attached is most often agarose, but other matrices are available. Mechanically stable matrices such as controlled pore glass or poly (styrenedivinyl) benzene allow for faster flow rates and shorter processing times than can be achieved with agarose. Where the antibody or antigen binding fragment thereof or antibody construct of the invention comprises a CH3 domain, the Bakerbond ABXMresin (J. T. Baker, Phillipsburg, N.J.) is useful for purification. Other techniques for protein purification such as fractionation on an ion-exchange column, ethanol precipitation, Reverse Phase HPLC, chromatography on silica, chromatography on heparin SEPHAROSE.TM. chromatography on an anion or cation exchange resin (such as a polyaspartic acid column), chromato-focusing, SDS-PAGE, and ammonium sulfate precipitation are also available depending on the antibody to be recovered.
[0164] The term "culturing" refers to the in vitro maintenance, differentiation, growth, proliferation and/or propagation of cells under suitable conditions in a medium.
[0165] As used herein, the term "pharmaceutical composition" relates to a composition for administration to a patient, preferably a human patient. The particular preferred pharmaceutical composition of this invention comprises the antibody or antigen binding fragment thereof or antibody construct of the invention. Preferably, the pharmaceutical composition comprises suitable formulations of carriers, stabilizers and/or excipients. In a preferred embodiment, the pharmaceutical composition comprises a composition for parenteral, transdermal, intraluminal, intraarterial, intrathecal and/or intranasal administration or by direct injection into tissue. It is in particular envisaged that said composition is administered to a patient via infusion or injection. Administration of the suitable compositions may be effected by different ways, e.g., by intravenous, intraperitoneal, subcutaneous, intramuscular, topical or intradermal administration. In particular, the present invention provides for an uninterrupted administration of the suitable composition. As a non-limiting example, uninterrupted, i.e. continuous administration may be realized by a small pump system worn by the patient for metering the influx of therapeutic agent into the body of the patient. The pharmaceutical composition comprising the antibody or antigen binding fragment thereof or antibody construct of the invention can be administered by using said pump systems. Such pump systems are generally known in the art, and commonly rely on periodic exchange of cartridges containing the therapeutic agent to be infused. When exchanging the cartridge in such a pump system, a temporary interruption of the otherwise uninterrupted flow of therapeutic agent into the body of the patient may ensue. In such a case, the phase of administration prior to cartridge replacement and the phase of administration following cartridge replacement would still be considered within the meaning of the pharmaceutical means and methods of the invention together make up one "uninterrupted administration" of such therapeutic agent.
[0166] The continuous or uninterrupted administration of these antibody or antigen binding fragment thereof or antibody constructs of the invention may be intravenous or subcutaneous by way of a fluid delivery device or small pump system including a fluid driving mechanism for driving fluid out of a reservoir and an actuating mechanism for actuating the driving mechanism. Pump systems for subcutaneous administration may include a needle or a cannula for penetrating the skin of a patient and delivering the suitable composition into the patient's body. Said pump systems may be directly fixed or attached to the skin of the patient independently of a vein, artery or blood vessel, thereby allowing a direct contact between the pump system and the skin of the patient. The pump system can be attached to the skin of the patient for 24 hours up to several days. The pump system may be of small size with a reservoir for small volumes. As a non-limiting example, the volume of the reservoir for the suitable pharmaceutical composition to be administered can be between 0.1 and 50 ml.
[0167] The continuous administration may be transdermal by way of a patch worn on the skin and replaced at intervals. One of skill in the art is aware of patch systems for drug delivery suitable for this purpose. It is of note that transdermal administration is especially amenable to uninterrupted administration, as exchange of a first exhausted patch can advantageously be accomplished simultaneously with the placement of a new, second patch, for example on the surface of the skin immediately adjacent to the first exhausted patch and immediately prior to removal of the first exhausted patch. Issues of flow interruption or power cell failure do not arise.
[0168] The inventive compositions may further comprise a pharmaceutically acceptable carrier. Examples of suitable pharmaceutical carriers are well known in the art and include solutions, e.g. phosphate buffered saline solutions, water, emulsions, such as oil/water emulsions, various types of wetting agents, sterile solutions, liposomes, etc. Compositions comprising such carriers can be formulated by well known conventional methods. Formulations can comprise carbohydrates, buffer solutions, amino acids and/or surfactants. Carbohydrates may be non-reducing sugars, preferably trehalose, sucrose, octasulfate, sorbitol or xylitol. In general, as used herein, "pharmaceutically acceptable carrier" means any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, compatible with pharmaceutical administration. The use of such media and agents for pharmaceutically active substances is well known in the art. Acceptable carriers, excipients, or stabilizers are nontoxic to recipients at the dosages and concentrations employed and include: additional buffering agents; preservatives; co-solvents; antioxidants, including ascorbic acid and methionine; chelating agents such as EDTA; metal complexes (e.g., Zn-protein complexes); biodegradable polymers, such as polyesters; salt-forming counter-ions, such as sodium, polyhydric sugar alcohols; amino acids, such as alanine, glycine, asparagine, 2-phenylalanine, and threonine; sugars or sugar alcohols, such as trehalose, sucrose, octasulfate, sorbitol or xylitol stachyose, mannose, sorbose, xylose, ribose, myoinisitose, galactose, lactitol, ribitol, myoinisitol, galactitol, glycerol, cyclitols (e.g., inositol), polyethylene glycol; sulfur containing reducing agents, such as glutathione, thioctic acid, sodium thioglycolate, thioglycerol, [alpha]-monothioglycerol, and sodium thio sulfate; low molecular weight proteins, such as human serum albumin, bovine serum albumin, gelatin, or other immunoglobulins; and hydrophilic polymers, such as polyvinylpyrrolidone. Such formulations may be used for continuous administrations which may be intravenous or subcutaneous with and/or without pump systems. Amino acids may be charged amino acids, preferably lysine, lysine acetate, arginine, glutamate and/or histidine. Surfactants may be detergents, preferably with a molecular weight of >1.2 KD and/or a polyether, preferably with a molecular weight of >3 KD. Non-limiting examples for preferred detergents are Tween 20, Tween 40, Tween 60, Tween 80 or Tween 85. Non-limiting examples for preferred polyethers are PEG 3000, PEG 3350, PEG 4000 or PEG 5000. Buffer systems used in the present invention can have a preferred pH of 5-9 and may comprise citrate, succinate, phosphate, histidine and acetate.
[0169] The compositions of the present invention can be administered to the subject at a suitable dose which can be determined e.g. by dose escalating studies by administration of increasing doses of the polypeptide of the invention exhibiting cross-species specificity described herein to non-chimpanzee primates, for instance macaques. As set forth above, the antibody or antigen binding fragment thereof or antibody construct of the invention exhibiting cross-species specificity described herein can be advantageously used in identical form in preclinical testing in non-chimpanzee primates and as drug in humans. These compositions can also be administered in combination with other proteinaceous and non-proteinaceous drugs. These drugs may be administered simultaneously with the composition comprising the polypeptide of the invention as defined herein or separately before or after administration of said polypeptide in timely defined intervals and doses. The dosage regimen will be determined by the attending physician and clinical factors. As is well known in the medical arts, dosages for any one patient depend upon many factors, including the patient's size, body surface area, age, the particular compound to be administered, sex, time and route of administration, general health, and other drugs being administered concurrently.
[0170] Preparations for parenteral administration include sterile aqueous or non-aqueous solutions, suspensions, and emulsions. Examples of non-aqueous solvents are propylene glycol, polyethylene glycol, vegetable oils such as olive oil, and injectable organic esters such as ethyl oleate. Aqueous carriers include water, alcoholic/aqueous solutions, emulsions or suspensions, including saline and buffered media. Parenteral vehicles include sodium chloride solution, Ringer's dextrose, dextrose and sodium chloride, lactated Ringer's, or fixed oils. Intravenous vehicles include fluid and nutrient replenishers, electrolyte replenishers (such as those based on Ringer's dextrose), and the like. Preservatives and other additives may also be present such as, for example, antimicrobials, anti-oxidants, chelating agents, inert gases and the like. In addition, the composition of the present invention might comprise proteinaceous carriers, like, e.g., serum albumin or immunoglobulin, preferably of human origin. It is envisaged that the composition of the invention might comprise, in addition to the polypeptide of the invention defined herein, further biologically active agents, depending on the intended use of the composition. Such agents might be drugs acting on the gastro-intestinal system, drugs acting as cytostatica, drugs preventing hyperurikemia, drugs inhibiting immunoreactions (e.g. corticosteroids), drugs modulating the inflammatory response, drugs acting on the circulatory system and/or agents such as cytokines known in the art. It is also envisaged that the antibody or antigen binding fragment thereof or antibody construct of the present invention is applied in a co-therapy, i.e., in combination with another anti-cancer medicament.
[0171] The biological activity of the pharmaceutical composition defined herein can be determined for instance by cytotoxicity assays, as described in the following examples, in WO 99/54440 or by Schlereth et al. (Cancer Immunol. Immunother. 20 (2005), 1-12). "Efficacy" or "in vivo efficacy" as used herein refers to the response to therapy by the pharmaceutical composition of the invention, using e.g. standardized NCI response criteria. The success or in vivo efficacy of the therapy using a pharmaceutical composition of the invention refers to the effectiveness of the composition for its intended purpose, i.e. the ability of the composition to cause its desired effect, i.e. depletion of pathologic cells, e.g. tumor cells. The in vivo efficacy may be monitored by established standard methods for the respective disease entities including, but not limited to white blood cell counts, differentials, Fluorescence Activated Cell Sorting, bone marrow aspiration. In addition, various disease specific clinical chemistry parameters and other established standard methods may be used. Furthermore, computer-aided tomography, X-ray, nuclear magnetic resonance tomography (e.g. for National Cancer Institute-criteria based response assessment [Cheson B D, Horning S J, Coiffier B, Shipp M A, Fisher R I, Connors J M, Lister T A, Vose J, Grillo-Lopez A, Hagenbeek A, Cabanillas F, Klippensten D, Hiddemann W, Castellino R, Harris N L, Armitage J O, Carter W, Hoppe R, Canellos G P. Report of an international workshop to standardize response criteria for non-Hodgkin's lymphomas. NCI Sponsored International Working Group. J Clin Oncol. 1999 April; 17(4):1244]), positron-emission tomography scanning, white blood cell counts, differentials, Fluorescence Activated Cell Sorting, bone marrow aspiration, lymph node biopsies/histologies, and various lymphoma specific clinical chemistry parameters (e.g. lactate dehydrogenase) and other established standard methods may be used.
[0172] Another major challenge in the development of drugs such as the pharmaceutical composition of the invention is the predictable modulation of pharmacokinetic properties. To this end, a pharmacokinetic profile of the drug candidate, i.e. a profile of the pharmacokinetic parameters that affect the ability of a particular drug to treat a given condition, can be established. Pharmacokinetic parameters of the drug influencing the ability of a drug for treating a certain disease entity include, but are not limited to: half-life, volume of distribution, hepatic first-pass metabolism and the degree of blood serum binding. The efficacy of a given drug agent can be influenced by each of the parameters mentioned above.
[0173] "Half-life" means the time where 50% of an administered drug are eliminated through biological processes, e.g. metabolism, excretion, etc.
[0174] By "hepatic first-pass metabolism" is meant the propensity of a drug to be metabolized upon first contact with the liver, i.e. during its first pass through the liver.
[0175] "Volume of distribution" means the degree of retention of a drug throughout the various compartments of the body, like e.g. intracellular and extracellular spaces, tissues and organs, etc. and the distribution of the drug within these compartments.
[0176] "Degree of blood serum binding" means the propensity of a drug to interact with and bind to blood serum proteins, such as albumin, leading to a reduction or loss of biological activity of the drug.
[0177] Pharmacokinetic parameters also include bioavailability, lag time (Tlag), Tmax, absorption rates, more onset and/or Cmax for a given amount of drug administered. "Bioavailability" means the amount of a drug in the blood compartment. "Lag time" means the time delay between the administration of the drug and its detection and measurability in blood or plasma.
[0178] "Tmax" is the time after which maximal blood concentration of the drug is reached, and "Cmax" is the blood concentration maximally obtained with a given drug. The time to reach a blood or tissue concentration of the drug which is required for its biological effect is influenced by all parameters. Pharmacokinetic parameters of bispecific single chain antibodies exhibiting cross-species specificity, which may be determined in preclinical animal testing in non-chimpanzee primates as outlined above, are also set forth e.g. in the publication by Schlereth et al. (Cancer Immunol. Immunother. 20 (2005), 1-12).
[0179] The term "toxicity" as used herein refers to the toxic effects of a drug manifested in adverse events or severe adverse events. These side events might refer to a lack of tolerability of the drug in general and/or a lack of local tolerance after administration. Toxicity could also include teratogenic or carcinogenic effects caused by the drug.
[0180] The term "safety", "in vivo safety" or "tolerability" as used herein defines the administration of a drug without inducing severe adverse events directly after administration (local tolerance) and during a longer period of application of the drug. "Safety", "in vivo safety" or "tolerability" can be evaluated e.g. at regular intervals during the treatment and follow-up period. Measurements include clinical evaluation, e.g. organ manifestations, and screening of laboratory abnormalities. Clinical evaluation may be carried out and deviations to normal findings recorded/coded according to NCI-CTC and/or MedDRA standards. Organ manifestations may include criteria such as allergy/immunology, blood/bone marrow, cardiac arrhythmia, coagulation and the like, as set forth e.g. in the Common Terminology Criteria for adverse events v3.0 (CTCAE). Laboratory parameters which may be tested include for instance hematology, clinical chemistry, coagulation profile and urine analysis and examination of other body fluids such as serum, plasma, lymphoid or spinal fluid, liquor and the like. Safety can thus be assessed e.g. by physical examination, imaging techniques (i.e. ultrasound, x-ray, CT scans, Magnetic Resonance Imaging (MRI), other measures with technical devices (i.e. electrocardiogram), vital signs, by measuring laboratory parameters and recording adverse events. For example, adverse events in non-chimpanzee primates in the uses and methods according to the invention may be examined by histopathological and/or histochemical methods.
[0181] The term "effective dose" or "effective dosage" is defined as an amount sufficient to achieve or at least partially achieve the desired effect. The term "therapeutically effective dose" is defined as an amount sufficient to cure or at least partially arrest the disease and its complications in a patient already suffering from the disease. Amounts effective for this use will depend upon the severity of the infection and the general state of the subject's own immune system. The term "patient" includes human and other mammalian subjects that receive either prophylactic or therapeutic treatment.
[0182] The term "effective and non-toxic dose" as used herein refers to a tolerable dose of an inventive antibody or antigen binding fragment thereof or antibody construct which is high enough to cause depletion of pathologic cells, tumor elimination, tumor shrinkage or stabilization of disease without or essentially without major toxic effects. Such effective and non-toxic doses may be determined e.g. by dose escalation studies described in the art and should be below the dose inducing severe adverse side events (dose limiting toxicity, DLT).
[0183] The above terms are also referred to e.g. in the Preclinical safety evaluation of biotechnology-derived pharmaceuticals S6; ICH Harmonised Tripartite Guideline; ICH Steering Committee meeting on Jul. 16, 1997.
[0184] The appropriate dosage, or therapeutically effective amount, of the antibody or antigen binding fragment thereof or antibody construct of the invention will depend on the condition to be treated, the severity of the condition, prior therapy, and the patient's clinical history and response to the therapeutic agent. The proper dose can be adjusted according to the judgment of the attending physician such that it can be administered to the patient one time or over a series of administrations. The pharmaceutical composition can be administered as a sole therapeutic or in combination with additional therapies such as anti-cancer therapies as needed.
[0185] The pharmaceutical compositions of this invention are particularly useful for parenteral administration, i.e., subcutaneously, intramuscularly, intravenously, intra-articular and/or intra-synovial. Parenteral administration can be by bolus injection or continuous infusion.
[0186] If the pharmaceutical composition has been lyophilized, the lyophilized material is first reconstituted in an appropriate liquid prior to administration. The lyophilized material may be reconstituted in, e.g., bacteriostatic water for injection (BWFI), physiological saline, phosphate buffered saline (PBS), or the same formulation the protein had been in prior to lyophilization.
[0187] In an internal analysis of proprietary mRNA expression data it has been surprisingly found that CDH19 expression is elevated in both primary and metastatic melanoma tumors compared to normal, untransformed tissues. Internal analysis also confirmed that expression of CDH19 in normal tissues is limited to neural crest derived peripheral nerve ganglia and nerve fibers. The differential CDH19 expression in normal and tumor tissues makes this protein attractive for cell-surface targeting therapeutics. Although CDH 19 was discussed as one marker as part of long lists of markers associated with some cancer types (see e.g. WO2009/055937) or Parkinson's disease (see e.g. WO2005/067391) CDH19 was never discussed as a prognostic marker or a drug target in connection with melanoma tumors.
[0188] As stated above, the present invention provides an isolated human antibody or antigen binding fragment thereof capable of binding to human CDH19 on the surface of a target cell. In a preferred embodiment the antibody or antigen binding fragment thereof comprises a monoclonal antibody or a fragment thereof.
[0189] The "CDH19 extracellular domain" or "CDH19 ECD" refers to a form of CDH19 which is essentially free of transmembrane and cytoplasmic domains of CDH19. It will be understood by the skilled artisan that the transmembrane domain identified for the CDH19 polypeptide of the present invention is identified pursuant to criteria routinely employed in the art for identifying that type of hydrophobic domain. The exact boundaries of a transmembrane domain may vary but most likely by no more than about 5 amino acids at either end of the domain specifically mentioned herein. A preferred human CDH19 ECD is shown in SEQ ID NO: 948 (aa residues 44-596). In this context it is understood that the CDH19 ECD represents the part of CDH19 on the surface of a target cell.
[0190] The affinity of the antibody or fragment thereof for human CDH19 is preferably .ltoreq.15 nM, more preferably .ltoreq.10 nM, even more preferably .ltoreq.5 nM, even more preferably .ltoreq.1 nM, even more preferably .ltoreq.0.5 nM, even more preferably .ltoreq.0.1 nM, and most preferably .ltoreq.0.05 nM. The affinity of the first binding domain for macaque CDH19 is preferably .ltoreq.15 nM, more preferably .ltoreq.10 nM, even more preferably .ltoreq.5 nM, even more preferably .ltoreq.1 nM, even more preferably .ltoreq.0.5 nM, even more preferably .ltoreq.0.1 nM, and most preferably .ltoreq.0.05 nM or even .ltoreq.0.01 nM. The affinity can be measured for example in a Biacore assay or in a Scatchard assay, e.g. as described in the Examples. The affinity gap for binding to macaque CDH19 versus human CDH19 is preferably [1:10-1:5] or [5:1-10:1], more preferably [1:5-5:1], and most preferably [1:2-3:1] or even [1:1-3:1]. Other methods of determining the affinity are well-known to the skilled person.
[0191] Human antibodies avoid some of the problems associated with antibodies that possess murine or rat variable and/or constant regions. The presence of such murine or rat derived proteins can lead to the rapid clearance of the antibodies or can lead to the generation of an immune response against the antibody by a patient. In order to avoid the utilization of murine or rat derived antibodies, human or fully human antibodies can be generated through the introduction of human antibody function into a rodent so that the rodent produces fully human antibodies.
[0192] The ability to clone and reconstruct megabase-sized human loci in YACs and to introduce them into the mouse germline provides a powerful approach to elucidating the functional components of very large or crudely mapped loci as well as generating useful models of human disease. Furthermore, the utilization of such technology for substitution of mouse loci with their human equivalents could provide unique insights into the expression and regulation of human gene products during development, their communication with other systems, and their involvement in disease induction and progression.
[0193] An important practical application of such a strategy is the "humanization" of the mouse humoral immune system. Introduction of human immunoglobulin (Ig) loci into mice in which the endogenous Ig genes have been inactivated offers the opportunity to study the mechanisms underlying programmed expression and assembly of antibodies as well as their role in B-cell development. Furthermore, such a strategy could provide an ideal source for production of fully human monoclonal antibodies (mAbs)--an important milestone towards fulfilling the promise of antibody therapy in human disease. Fully human antibodies are expected to minimize the immunogenic and allergic responses intrinsic to mouse or mouse-derivatized mAbs and thus to increase the efficacy and safety of the administered antibodies. The use of fully human antibodies can be expected to provide a substantial advantage in the treatment of chronic and recurring human diseases, such as inflammation, autoimmunity, and cancer, which require repeated antibody administrations.
[0194] One approach towards this goal was to engineer mouse strains deficient in mouse antibody production with large fragments of the human Ig loci in anticipation that such mice would produce a large repertoire of human antibodies in the absence of mouse antibodies. Large human Ig fragments would preserve the large variable gene diversity as well as the proper regulation of antibody production and expression. By exploiting the mouse machinery for antibody diversification and selection and the lack of immunological tolerance to human proteins, the reproduced human antibody repertoire in these mouse strains should yield high affinity antibodies against any antigen of interest, including human antigens. Using the hybridoma technology, antigen-specific human mAbs with the desired specificity could be readily produced and selected. This general strategy was demonstrated in connection with our generation of the first XenoMouse mouse strains, as published in 1994. (See Green et al. Nature Genetics 7:13-21 (1994)) The XenoMouse strains were engineered with yeast artificial chromosomes (YACs) containing 245 kb and 190 kb-sized germline configuration fragments of the human heavy chain locus and kappa light chain locus, respectively, which contained core variable and constant region sequences. Id. The human Ig containing YACs proved to be compatible with the mouse system for both rearrangement and expression of antibodies and were capable of substituting for the inactivated mouse Ig genes. This was demonstrated by their ability to induce B-cell development, to produce an adult-like human repertoire of fully human antibodies, and to generate antigen-specific human mAbs. These results also suggested that introduction of larger portions of the human Ig loci containing greater numbers of V genes, additional regulatory elements, and human Ig constant regions might recapitulate substantially the full repertoire that is characteristic of the human humoral response to infection and immunization. The work of Green et al. was recently extended to the introduction of greater than approximately 80% of the human antibody repertoire through introduction of megabase sized, germline configuration YAC fragments of the human heavy chain loci and kappa light chain loci, respectively. See Mendez et al. Nature Genetics 15:146-156 (1997) and U.S. patent application Ser. No. 08/759,620, filed Dec. 3, 1996, the disclosures of which are hereby incorporated by reference.
[0195] The production of the XenoMouse mice is further discussed and delineated in U.S. patent application Ser. No. 07/466,008, filed Jan. 12, 1990, Ser. No. 07/610,515, filed Nov. 8, 1990, Ser. No. 07/919,297, filed Jul. 24, 1992, Ser. No. 07/922,649, filed Jul. 30, 1992, filed Ser. No. 08/031,801, filed Mar. 15, 1993, Ser. No. 08/112,848, filed Aug. 27, 1993, Ser. No. 08/234,145, filed Apr. 28, 1994, Ser. No. 08/376,279, filed Jan. 20, 1995, Ser. No. 08/430,938, Apr. 27, 1995, Ser. No. 08/464,584, filed Jun. 5, 1995, Ser. No. 08/464,582, filed Jun. 5, 1995, Ser. No. 08/463,191, filed Jun. 5, 1995, Ser. No. 08/462,837, filed Jun. 5, 1995, Ser. No. 08/486,853, filed Jun. 5, 1995, Ser. No. 08/486,857, filed Jun. 5, 1995, Ser. No. 08/486,859, filed Jun. 5, 1995, Ser. No. 08/462,513, filed Jun. 5, 1995, Ser. No. 08/724,752, filed Oct. 2, 1996, and Ser. No. 08/759,620, filed Dec. 3, 1996 and U.S. Pat. Nos. 6,162,963, 6,150,584, 6,114,598, 6,075,181, and 5,939,598 and Japanese Patent Nos. 3 068 180 B2, 3 068 506 B2, and 3 068 507 B2. See also Mendez et al. Nature Genetics 15:146-156 (1997) and Green and Jakobovits J. Exp. Med. 188:483-495 (1998). See also European Patent No., EP 0 463151 B1, grant published Jun. 12, 1996, International Patent Application No., WO 94/02602, published Feb. 3, 1994, International Patent Application No., WO 96/34096, published Oct. 31, 1996, WO 98/24893, published Jun. 11, 1998, WO 00/76310, published Dec. 21, 2000, WO 03/47336. The disclosures of each of the above-cited patents, applications, and references are hereby incorporated by reference in their entirety.
[0196] In an alternative approach, others, including GenPharm International, Inc., have utilized a "minilocus" approach. In the minilocus approach, an exogenous Ig locus is mimicked through the inclusion of pieces (individual genes) from the Ig locus. Thus, one or more V.sub.H genes, one or more D.sub.H genes, one or more J.sub.H genes, a mu constant region, and a second constant region (preferably a gamma constant region) are formed into a construct for insertion into an animal. This approach is described in U.S. Pat. No. 5,545,807 to Surani et al. and U.S. Pat. Nos. 5,545,806, 5,625,825, 5,625,126, 5,633,425, 5,661,016, 5,770,429, 5,789,650, 5,814,318, 5,877,397, 5,874,299, and 6,255,458 each to Lonberg and Kay, U.S. Pat. Nos. 5,591,669 and 6,023.010 to Krimpenfort and Berns, U.S. Pat. Nos. 5,612,205, 5,721,367, and 5,789,215 to Berns et al., and U.S. Pat. No. 5,643,763 to Choi and Dunn, and GenPharm International U.S. patent application Ser. No. 07/574,748, filed Aug. 29, 1990, Ser. No. 07/575,962, filed Aug. 31, 1990, Ser. No. 07/810,279, filed Dec. 17, 1991, Ser. No. 07/853,408, filed Mar. 18, 1992, Ser. No. 07/904,068, filed Jun. 23, 1992, Ser. No. 07/990,860, filed Dec. 16, 1992, Ser. No. 08/053,131, filed Apr. 26, 1993, Ser. No. 08/096,762, filed Jul. 22, 1993, Ser. No. 08/155,301, filed Nov. 18, 1993, Ser. No. 08/161,739, filed Dec. 3, 1993, Ser. No. 08/165,699, filed Dec. 10, 1993, Ser. No. 08/209,741, filed Mar. 9, 1994, the disclosures of which are hereby incorporated by reference. See also European Patent No. 0 546 073 B 1, International Patent Application Nos. WO 92/03918, WO 92/22645, WO 92/22647, WO 92/22670, WO 93/12227, WO 94/00569, WO 94/25585, WO 96/14436, WO 97/13852, and WO 98/24884 and U.S. Pat. No. 5,981,175, the disclosures of which are hereby incorporated by reference in their entirety. See further Taylor et al., 1992, Chen et al., 1993, Tuaillon et al., 1993, Choi et al., 1993, Lonberg et al., (1994), Taylor et al., (1994), and Tuaillon et al., (1995), Fishwild et al., (1996), the disclosures of which are hereby incorporated by reference in their entirety.
[0197] Kirin has also demonstrated the generation of human antibodies from mice in which, through microcell fusion, large pieces of chromosomes, or entire chromosomes, have been introduced. See European Patent Application Nos. 773 288 and 843 961, the disclosures of which are hereby incorporated by reference. Xenerex Biosciences is developing a technology for the potential generation of human antibodies. In this technology, SCID mice are reconstituted with human lymphatic cells, e.g., B and/or T cells. Mice are then immunized with an antigen and can generate an immune response against the antigen. See U.S. Pat. Nos. 5,476,996, 5,698,767, and 5,958,765.
[0198] Human anti-mouse antibody (HAMA) responses have led the industry to prepare chimeric or otherwise humanized antibodies. While chimeric antibodies have a human constant region and a murine variable region, it is expected that certain human anti-chimeric antibody (HACA) responses will be observed, particularly in chronic or multi-dose utilizations of the antibody. Thus, it would be desirable to provide fully human antibodies against EGFRvIII in order to vitiate concerns and/or effects of HAMA or HACA response.
[0199] According to one embodiment the antibody of the present invention is a dimer comprising two fusion proteins created by fusing a CDH19 binding fragment of a CDH19 antibody to the Fc region of an antibody. The dimer can be made by, for example, inserting a gene fusion encoding the fusion protein into an appropriate expression vector, expressing the gene fusion in host cells transformed with the recombinant expression vector, and allowing the expressed fusion protein to assemble much like antibody molecules, whereupon interchain disulfide bonds form between the Fc moieties to yield the dimer.
[0200] The term "Fc polypeptide" as used herein includes native and mutein forms of polypeptides derived from the Fc region of an antibody. Truncated forms of such polypeptides containing the hinge region that promotes dimerization also are included. Fusion proteins comprising Fc moieties (and oligomers formed therefrom) offer the advantage of facile purification by affinity chromatography over Protein A or Protein G columns.
[0201] One suitable Fc polypeptide, described in PCT application WO 93/10151 (hereby incorporated by reference), is a single chain polypeptide extending from the N-terminal hinge region to the native C-terminus of the Fc region of a human IgG antibody. Another useful Fc polypeptide is the Fc mutein described in U.S. Pat. No. 5,457,035 and in Baum et al., 1994, EMBO J. 13:3992-4001. The amino acid sequence of this mutein is identical to that of the native Fc sequence presented in WO 93/10151, except that amino acid 19 has been changed from Leu to Ala, amino acid 20 has been changed from Leu to Glu, and amino acid 22 has been changed from Gly to Ala. The mutein exhibits reduced affinity for Fc receptors.
[0202] Alternatively, the antibody of the invention is a fusion protein comprising multiple CDH19 antibody polypeptides, with or without peptide linkers (spacer peptides). Among the suitable peptide linkers are those described in U.S. Pat. Nos. 4,751,180 and 4,935,233 or WO 88/09344.
[0203] Another method for preparing oligomeric CDH19 antibody derivatives involves use of a leucine zipper. Leucine zipper domains are peptides that promote oligomerization of the proteins in which they are found. Leucine zippers were originally identified in several DNA-binding proteins (Landschulz et al., 1988, Science 240:1759), and have since been found in a variety of different proteins. Among the known leucine zippers are naturally occurring peptides and derivatives thereof that dimerize or trimerize. Examples of leucine zipper domains suitable for producing soluble oligomeric proteins are described in PCT application WO 94/10308, and the leucine zipper derived from lung surfactant protein D (SPD) described in Hoppe et al., 1994, FEBS Letters 344:191, hereby incorporated by reference. The use of a modified leucine zipper that allows for stable trimerization of a heterologous protein fused thereto is described in Fanslow et al., 1994, Semin. Immunol. 6:267-78. In one approach, recombinant fusion proteins comprising CDH19 antibody fragment or derivative fused to a leucine zipper peptide are expressed in suitable host cells, and the soluble oligomeric CDH19 antibody fragments or derivatives that form are recovered from the culture supernatant.
[0204] Covalent modifications of antigen binding proteins are included within the scope of this invention, and are generally, but not always, done post-translationally. For example, several types of covalent modifications of the antigen binding protein are introduced into the molecule by reacting specific amino acid residues of the antigen binding protein with an organic derivatizing agent that is capable of reacting with selected side chains or the N- or C-terminal residues.
[0205] Cysteinyl residues most commonly are reacted with .alpha.-haloacetates (and corresponding amines), such as chloroacetic acid or chloroacetamide, to give carboxymethyl or carboxyamidomethyl derivatives. Cysteinyl residues also are derivatized by reaction with bromotrifluoroacetone, .alpha.-bromo-.beta.-(5-imidozoyl)propionic acid, chloroacetyl phosphate, N-alkylmaleimides, 3-nitro-2-pyridyl disulfide, methyl 2-pyridyl disulfide, p-chloromercuribenzoate, 2-chloromercuri-4-nitrophenol, or chloro-7-nitrobenzo-2-oxa-1,3-diazole.
[0206] Histidyl residues are derivatized by reaction with diethylpyrocarbonate at pH 5.5-7.0 because this agent is relatively specific for the histidyl side chain. Para-bromophenacyl bromide also is useful; the reaction is preferably performed in 0.1 M sodium cacodylate at pH 6.0.
[0207] Lysinyl and amino terminal residues are reacted with succinic or other carboxylic acid anhydrides. Derivatization with these agents has the effect of reversing the charge of the lysinyl residues. Other suitable reagents for derivatizing alpha-amino-containing residues include imidoesters such as methyl picolinimidate; pyridoxal phosphate; pyridoxal; chloroborohydride; trinitrobenzenesulfonic acid; O-methylisourea; 2,4-pentanedione; and transaminase-catalyzed reaction with glyoxylate.
[0208] Arginyl residues are modified by reaction with one or several conventional reagents, among them phenylglyoxal, 2,3-butanedione, 1,2-cyclohexanedione, and ninhydrin. Derivatization of arginine residues requires that the reaction be performed in alkaline conditions because of the high pKa of the guanidine functional group. Furthermore, these reagents may react with the groups of lysine as well as the arginine epsilon-amino group.
[0209] The specific modification of tyrosyl residues may be made, with particular interest in introducing spectral labels into tyrosyl residues by reaction with aromatic diazonium compounds or tetranitromethane. Most commonly, N-acetylimidizole and tetranitromethane are used to form O-acetyl tyrosyl species and 3-nitro derivatives, respectively. Tyrosyl residues are iodinated using .sup.125I or .sup.131I to prepare labeled proteins for use in radioimmunoassay, the chloramine T method described above being suitable.
[0210] Carboxyl side groups (aspartyl or glutamyl) are selectively modified by reaction with carbodiimides (R'--N.dbd.C.dbd.N--R'), where R and R' are optionally different alkyl groups, such as 1-cyclohexyl-3-(2-morpholinyl-4-ethyl) carbodiimide or 1-ethyl-3-(4-azonia-4,4-dimethylpentyl) carbodiimide. Furthermore, aspartyl and glutamyl residues are converted to asparaginyl and glutaminyl residues by reaction with ammonium ions.
[0211] Derivatization with bifunctional agents is useful for crosslinking antigen binding proteins to a water-insoluble support matrix or surface for use in a variety of methods. Commonly used crosslinking agents include, e.g., 1,1-bis(diazoacetyl)-2-phenylethane, glutaraldehyde, N-hydroxysuccinimide esters, for example, esters with 4-azidosalicylic acid, homobifunctional imidoesters, including disuccinimidyl esters such as 3,3'-dithiobis(succinimidylpropionate), and bifunctional maleimides such as bis-N-maleimido-1,8-octane. Derivatizing agents such as methyl-3-[(p-azidophenyl)dithio]propioimidate yield photoactivatable intermediates that are capable of forming crosslinks in the presence of light. Alternatively, reactive water-insoluble matrices such as cyanogen bromide-activated carbohydrates and the reactive substrates described in U.S. Pat. Nos. 3,969,287; 3,691,016; 4,195,128; 4,247,642; 4,229,537; and 4,330,440 are employed for protein immobilization.
[0212] Glutaminyl and asparaginyl residues are frequently deamidated to the corresponding glutamyl and aspartyl residues, respectively. Alternatively, these residues are deamidated under mildly acidic conditions. Either form of these residues falls within the scope of this invention.
[0213] Other modifications include hydroxylation of proline and lysine, phosphorylation of hydroxyl groups of seryl or threonyl residues, methylation of the .alpha.-amino groups of lysine, arginine, and histidine side chains (T. E. Creighton, Proteins: Structure and Molecular Properties, W. H. Freeman & Co., San Francisco, 1983, pp. 79-86), acetylation of the N-terminal amine, and amidation of any C-terminal carboxyl group.
[0214] Another type of covalent modification of the antigen binding protein included within the scope of this invention comprises altering the glycosylation pattern of the protein. As is known in the art, glycosylation patterns can depend on both the sequence of the protein (e.g., the presence or absence of particular glycosylation amino acid residues, discussed below), or the host cell or organism in which the protein is produced. Particular expression systems are discussed below.
[0215] Glycosylation of polypeptides is typically either N-linked or O-linked. N-linked refers to the attachment of the carbohydrate moiety to the side chain of an asparagine residue. The tri-peptide sequences asparagine-X-serine and asparagine-X-threonine, where X is any amino acid except proline, are the recognition sequences for enzymatic attachment of the carbohydrate moiety to the asparagine side chain. Thus, the presence of either of these tri-peptide sequences in a polypeptide creates a potential glycosylation site. O-linked glycosylation refers to the attachment of one of the sugars N-acetylgalactosamine, galactose, or xylose, to a hydroxyamino acid, most commonly serine or threonine, although 5-hydroxyproline or 5-hydroxylysine may also be used.
[0216] Addition of glycosylation sites to the antigen binding protein is conveniently accomplished by altering the amino acid sequence such that it contains one or more of the above-described tri-peptide sequences (for N-linked glycosylation sites). The alteration may also be made by the addition of, or substitution by, one or more serine or threonine residues to the starting sequence (for O-linked glycosylation sites). For ease, the antigen binding protein amino acid sequence is preferably altered through changes at the DNA level, particularly by mutating the DNA encoding the target polypeptide at preselected bases such that codons are generated that will translate into the desired amino acids.
[0217] Another means of increasing the number of carbohydrate moieties on the antigen binding protein is by chemical or enzymatic coupling of glycosides to the protein. These procedures are advantageous in that they do not require production of the protein in a host cell that has glycosylation capabilities for N- and O-linked glycosylation. Depending on the coupling mode used, the sugar(s) may be attached to (a) arginine and histidine, (b) free carboxyl groups, (c) free sulfhydryl groups such as those of cysteine, (d) free hydroxyl groups such as those of serine, threonine, or hydroxyproline, (e) aromatic residues such as those of phenylalanine, tyrosine, or tryptophan, or (f) the amide group of glutamine. These methods are described in WO 87/05330 published Sep. 11, 1987, and in Aplin and Wriston, 1981, CRC Crit. Rev. Biochem., pp. 259-306.
[0218] Removal of carbohydrate moieties present on the starting antigen binding protein may be accomplished chemically or enzymatically. Chemical deglycosylation requires exposure of the protein to the compound trifluoromethanesulfonic acid, or an equivalent compound. This treatment results in the cleavage of most or all sugars except the linking sugar (N-acetylglucosamine or N-acetylgalactosamine), while leaving the polypeptide intact. Chemical deglycosylation is described by Hakimuddin et al., 1987, Arch. Biochem. Biophys. 259:52 and by Edge et al., 1981, Anal. Biochem. 118:131. Enzymatic cleavage of carbohydrate moieties on polypeptides can be achieved by the use of a variety of endo- and exo-glycosidases as described by Thotakura et al., 1987, Meth. Enzymol. 138:350. Glycosylation at potential glycosylation sites may be prevented by the use of the compound tunicamycin as described by Duskin et al., 1982, J. Biol. Chem. 257:3105. Tunicamycin blocks the formation of protein-N-glycoside linkages.
[0219] Another type of covalent modification of the antigen binding protein comprises linking the antigen binding protein to various non-proteinaceous polymers, including, but not limited to, various polyols such as polyethylene glycol, polypropylene glycol or polyoxyalkylenes, in the manner set forth in U.S. Pat. Nos. 4,640,835; 4,496,689; 4,301,144; 4,670,417; 4,791,192 or 4,179,337. In addition, as is known in the art, amino acid substitutions may be made in various positions within the antigen binding protein to facilitate the addition of polymers such as PEG.
[0220] In some embodiments, the covalent modification of the antigen binding proteins of the invention comprises the addition of one or more labels.
[0221] The term "labelling group" means any detectable label. Examples of suitable labelling groups include, but are not limited to, the following: radioisotopes or radionuclides (e.g., .sup.3H, .sup.14C, .sup.15N, .sup.35S, .sup.90Y, .sup.99Tc, .sup.111In, .sup.125I, .sup.131I), fluorescent groups (e.g., FITC, rhodamine, lanthanide phosphors), enzymatic groups (e.g., horseradish peroxidase, .beta.-galactosidase, luciferase, alkaline phosphatase), chemiluminescent groups, biotinyl groups, or predetermined polypeptide epitopes recognized by a secondary reporter (e.g., leucine zipper pair sequences, binding sites for secondary antibodies, metal binding domains, epitope tags). In some embodiments, the labelling group is coupled to the antigen binding protein via spacer arms of various lengths to reduce potential steric hindrance. Various methods for labelling proteins are known in the art and may be used in performing the present invention.
[0222] In general, labels fall into a variety of classes, depending on the assay in which they are to be detected: a) isotopic labels, which may be radioactive or heavy isotopes; b) magnetic labels (e.g., magnetic particles); c) redox active moieties; d) optical dyes; enzymatic groups (e.g. horseradish peroxidase, .beta.-galactosidase, luciferase, alkaline phosphatase); e) biotinylated groups; and f) predetermined polypeptide epitopes recognized by a secondary reporter (e.g., leucine zipper pair sequences, binding sites for secondary antibodies, metal binding domains, epitope tags, etc.). In some embodiments, the labelling group is coupled to the antigen binding protein via spacer arms of various lengths to reduce potential steric hindrance. Various methods for labelling proteins are known in the art and may be used in performing the present invention.
[0223] Specific labels include optical dyes, including, but not limited to, chromophores, phosphors and fluorophores, with the latter being specific in many instances. Fluorophores can be either "small molecule" fluores, or proteinaceous fluores.
[0224] By "fluorescent label" is meant any molecule that may be detected via its inherent fluorescent properties. Suitable fluorescent labels include, but are not limited to, fluorescein, rhodamine, tetramethylrhodamine, eosin, erythrosin, coumarin, methyl-coumarins, pyrene, Malacite green, stilbene, Lucifer Yellow, Cascade BlueJ, Texas Red, IAEDANS, EDANS, BODIPY FL, LC Red 640, Cy 5, Cy 5.5, LC Red 705, Oregon green, the Alexa-Fluor dyes (Alexa Fluor 350, Alexa Fluor 430, Alexa Fluor 488, Alexa Fluor 546, Alexa Fluor 568, Alexa Fluor 594, Alexa Fluor 633, Alexa Fluor 660, Alexa Fluor 680), Cascade Blue, Cascade Yellow and R-phycoerythrin (PE) (Molecular Probes, Eugene, Oreg.), FITC, Rhodamine, and Texas Red (Pierce, Rockford, Ill.), Cy5, Cy5.5, Cy7 (Amersham Life Science, Pittsburgh, Pa.). Suitable optical dyes, including fluorophores, are described in Molecular Probes Handbook by Richard P. Haugland, hereby expressly incorporated by reference.
[0225] Suitable proteinaceous fluorescent labels also include, but are not limited to, green fluorescent protein, including a Renilla, Ptilosarcus, or Aequorea species of GFP (Chalfie et al., 1994, Science 263:802-805), EGFP (Clontech Laboratories, Inc., Genbank Accession Number U55762), blue fluorescent protein (BFP, Quantum Biotechnologies, Inc. 1801 de Maisonneuve Blvd. West, 8th Floor, Montreal, Quebec, Canada H3H 1J9; Stauber, 1998, Biotechniques 24:462-471; Heim et al., 1996, Curr. Biol. 6:178-182), enhanced yellow fluorescent protein (EYFP, Clontech Laboratories, Inc.), luciferase (Ichiki et al., 1993, J. Immunol. 150:5408-5417), .beta. galactosidase (Nolan et al., 1988, Proc. Natl. Acad. Sci. U.S.A. 85:2603-2607) and Renilla (WO92/15673, WO95/07463, WO98/14605, WO98/26277, WO99/49019, U.S. Pat. Nos. 5,292,658, 5,418,155, 5,683,888, 5,741,668, 5,777,079, 5,804,387, 5,874,304, 5,876,995, 5,925,558). All of the above-cited references are expressly incorporated herein by reference.
[0226] As described in appended example 2 a broad number of CDH19 specific binder has been characterized with respect to identified binding characteristics and those binders were grouped into five different bins, which refers to five different subgroups of CDH19 specific binding domains. Accordingly, in one embodiment the human antibody or antigen binding fragment thereof of the invention comprises a human binding domain or antigen binding fragment thereof comprising a VH region comprising CDR-H1, CDR-H2 and CDR-H3 and a VL region comprising CDR-L1, CDR-L2 and CDR-L3 selected from the group consisting of: [0227] (a) CDR-H1 as depicted in SEQ ID NO: 52, CDR-H2 as depicted in SEQ ID NO: 53, CDR-H3 as depicted in SEQ ID NO: 54, CDR-L1 as depicted in SEQ ID NO: 220, CDR-L2 as depicted in SEQ ID NO: 221 and CDR-L3 as depicted in SEQ ID NO: 222, CDR-H1 as depicted in SEQ ID NO: 82, CDR-H2 as depicted in SEQ ID NO: 83, CDR-H3 as depicted in SEQ ID NO: 84, CDR-L1 as depicted in SEQ ID NO: 250, CDR-L2 as depicted in SEQ ID NO: 251 and CDR-L3 as depicted in SEQ ID NO: 252, CDR-H1 as depicted in SEQ ID NO: 82, CDR-H2 as depicted in SEQ ID NO: 83, CDR-H3 as depicted in SEQ ID NO: 84, CDR-L1 as depicted in SEQ ID NO: 250, CDR-L2 as depicted in SEQ ID NO: 251 and CDR-L3 as depicted in SEQ ID NO: 927, CDR-H1 as depicted in SEQ ID NO: 82, CDR-H2 as depicted in SEQ ID NO: 83, CDR-H3 as depicted in SEQ ID NO: 909, CDR-L1 as depicted in SEQ ID NO: 250, CDR-L2 as depicted in SEQ ID NO: 251 and CDR-L3 as depicted in SEQ ID NO: 927, CDR-H1 as depicted in SEQ ID NO: 52, CDR-H2 as depicted in SEQ ID NO: 53, CDR-H3 as depicted in SEQ ID NO: 54, CDR-L1 as depicted in SEQ ID NO: 220, CDR-L2 as depicted in SEQ ID NO: 221 and CDR-L3 as depicted in SEQ ID NO: 926, and CDR-H1 as depicted in SEQ ID NO: 52, CDR-H2 as depicted in SEQ ID NO: 53, CDR-H3 as depicted in SEQ ID NO: 904, CDR-L1 as depicted in SEQ ID NO: 220, CDR-L2 as depicted in SEQ ID NO: 221 and CDR-L3 as depicted in SEQ ID NO: 926; which all characterize binding domains for CDH19 grouped into bin 1; [0228] (b) CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 126, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 294, CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 132, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 300, CDR-H1 as depicted in SEQ ID NO: 136, CDR-H2 as depicted in SEQ ID NO: 137, CDR-H3 as depicted in SEQ ID NO: 138, CDR-L1 as depicted in SEQ ID NO: 304, CDR-L2 as depicted in SEQ ID NO: 305 and CDR-L3 as depicted in SEQ ID NO: 306, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 144, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 312, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 318, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 336, CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 915, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 294, CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 915, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 928, CDR-H1 as depicted in SEQ ID NO: 124, CDR-H2 as depicted in SEQ ID NO: 125, CDR-H3 as depicted in SEQ ID NO: 915, CDR-L1 as depicted in SEQ ID NO: 292, CDR-L2 as depicted in SEQ ID NO: 293 and CDR-L3 as depicted in SEQ ID NO: 929, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 336, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 942, CDR-H1 as depicted in SEQ ID NO: 166, CDR-H2 as depicted in SEQ ID NO: 167, CDR-H3 as depicted in SEQ ID NO: 168, CDR-L1 as depicted in SEQ ID NO: 334, CDR-L2 as depicted in SEQ ID NO: 335 and CDR-L3 as depicted in SEQ ID NO: 943, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 318, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 937, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 150, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 938, CDR-H1 as depicted in SEQ ID NO: 148, CDR-H2 as depicted in SEQ ID NO: 149, CDR-H3 as depicted in SEQ ID NO: 919, CDR-L1 as depicted in SEQ ID NO: 316, CDR-L2 as depicted in SEQ ID NO: 317 and CDR-L3 as depicted in SEQ ID NO: 938, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 144, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 935, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 918, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 935, CDR-H1 as depicted in SEQ ID NO: 142, CDR-H2 as depicted in SEQ ID NO: 143, CDR-H3 as depicted in SEQ ID NO: 918, CDR-L1 as depicted in SEQ ID NO: 310, CDR-L2 as depicted in SEQ ID NO: 311 and CDR-L3 as depicted in SEQ ID NO: 936, CDR-H1 as depicted in SEQ ID NO: 136, CDR-H2 as depicted in SEQ ID NO: 137, CDR-H3 as depicted in SEQ ID NO: 138, CDR-L1 as depicted in SEQ ID NO: 304, CDR-L2 as depicted in SEQ ID NO: 305 and CDR-L3 as depicted in SEQ ID NO: 933, CDR-H1 as depicted in SEQ ID NO: 136, CDR-H2 as depicted in SEQ ID NO: 137, CDR-H3 as depicted in SEQ ID NO: 917, CDR-L1 as depicted in SEQ ID NO: 304, CDR-L2 as depicted in SEQ ID NO: 305 and CDR-L3 as depicted in SEQ ID NO: 934, CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 132, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 930, CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 916, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 931, and CDR-H1 as depicted in SEQ ID NO: 130, CDR-H2 as depicted in SEQ ID NO: 131, CDR-H3 as depicted in SEQ ID NO: 916, CDR-L1 as depicted in SEQ ID NO: 298, CDR-L2 as depicted in SEQ ID NO: 299 and CDR-L3 as depicted in SEQ ID NO: 932; which all characterize binding domains for CDH19 grouped into bin 2; [0229] (c) CDR-H1 as depicted in SEQ ID NO: 94, CDR-H2 as depicted in SEQ ID NO: 95, CDR-H3 as depicted in SEQ ID NO: 96, CDR-L1 as depicted in SEQ ID NO: 262, CDR-L2 as depicted in SEQ ID NO: 263 and CDR-L3 as depicted in SEQ ID NO: 264, CDR-H1 as depicted in SEQ ID NO: 100, CDR-H2 as depicted in SEQ ID NO: 101, CDR-H3 as depicted in SEQ ID NO: 102, CDR-L1 as depicted in SEQ ID NO: 268, CDR-L2 as depicted in SEQ ID NO: 269 and CDR-L3 as depicted in SEQ ID NO: 270, CDR-H1 as depicted in SEQ ID NO: 118, CDR-H2 as depicted in SEQ ID NO: 119, CDR-H3 as depicted in SEQ ID NO: 120, CDR-L1 as depicted in SEQ ID NO: 286, CDR-L2 as depicted in SEQ ID NO: 287 and CDR-L3 as depicted in SEQ ID NO: 288, CDR-H1 as depicted in SEQ ID NO: 154, CDR-H2 as depicted in SEQ ID NO: 155, CDR-H3 as depicted in SEQ ID NO: 156, CDR-L1 as depicted in SEQ ID NO: 322, CDR-L2 as depicted in SEQ ID NO: 323 and CDR-L3 as depicted in SEQ ID NO: 324, CDR-H1 as depicted in SEQ ID NO: 100, CDR-H2 as depicted in SEQ ID NO: 101, CDR-H3 as depicted in SEQ ID NO: 912, CDR-L1 as depicted in SEQ ID NO: 268, CDR-L2 as depicted in SEQ ID NO: 269 and CDR-L3 as depicted in SEQ ID NO: 270, CDR-H1 as depicted in SEQ ID NO: 100, CDR-H2 as depicted in SEQ ID NO: 101, CDR-H3 as depicted in SEQ ID NO: 913, CDR-L1 as depicted in SEQ ID NO: 268, CDR-L2 as depicted in SEQ ID NO: 269 and CDR-L3 as depicted in SEQ ID NO: 270, CDR-H1 as depicted in SEQ ID NO: 94, CDR-H2 as depicted in SEQ ID NO: 95, CDR-H3 as depicted in SEQ ID NO: 910, CDR-L1 as depicted in SEQ ID NO: 262, CDR-L2 as depicted in SEQ ID NO: 263 and CDR-L3 as depicted in SEQ ID NO: 264, CDR-H1 as depicted in SEQ ID NO: 94, CDR-H2 as depicted in SEQ ID NO: 95, CDR-H3 as depicted in SEQ ID NO: 911, CDR-L1 as depicted in SEQ ID NO: 262, CDR-L2 as depicted in SEQ ID NO: 263 and CDR-L3 as depicted in SEQ ID NO: 264, CDR-H1 as depicted in SEQ ID NO: 118, CDR-H2 as depicted in SEQ ID NO: 119, CDR-H3 as depicted in SEQ ID NO: 120, CDR-L1 as depicted in SEQ ID NO: 286, CDR-L2 as depicted in SEQ ID NO: 287 and CDR-L3 as depicted in SEQ ID NO: 288, CDR-H1 as depicted in SEQ ID NO: 118, CDR-H2 as depicted in SEQ ID NO: 914, CDR-H3 as depicted in SEQ ID NO: 120, CDR-L1 as depicted in SEQ ID NO: 286, CDR-L2 as depicted in SEQ ID NO: 287 and CDR-L3 as depicted in SEQ ID NO: 288, and CDR-H1 as depicted in SEQ ID NO: 154, CDR-H2 as depicted in SEQ ID NO: 155, CDR-H3 as depicted in SEQ ID NO: 920, CDR-L1 as depicted in SEQ ID NO: 322, CDR-L2 as depicted in SEQ ID NO: 323 and CDR-L3 as depicted in SEQ ID NO: 324; which all characterize binding domains for CDH19 grouped into bin 3; [0230] (d) CDR-H1 as depicted in SEQ ID NO: 4, CDR-H2 as depicted in SEQ ID NO: 5, CDR-H3 as depicted in SEQ ID NO: 6, CDR-L1 as depicted in SEQ ID NO: 172, CDR-L2 as depicted in SEQ ID NO: 173 and CDR-L3 as depicted in SEQ ID NO: 174, CDR-H1 as depicted in SEQ ID NO: 10, CDR-H2 as depicted in SEQ ID NO: 11, CDR-H3 as depicted in SEQ ID NO: 12, CDR-L1 as depicted in SEQ ID NO: 178, CDR-L2 as depicted in SEQ ID NO: 179 and CDR-L3 as depicted in SEQ ID NO: 180, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 196, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 198, CDR-H1 as depicted in SEQ ID NO: 34, CDR-H2 as depicted in SEQ ID NO: 35, CDR-H3 as depicted in SEQ ID NO: 36, CDR-L1 as depicted in SEQ ID NO: 202, CDR-L2 as depicted in SEQ ID NO: 203 and CDR-L3 as depicted in SEQ ID NO: 204, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 48, CDR-L1 as depicted in SEQ ID NO: 214, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 58, CDR-H2 as depicted in SEQ ID NO: 59, CDR-H3 as depicted in SEQ ID NO: 60, CDR-L1 as depicted in SEQ ID NO: 226, CDR-L2 as depicted in SEQ ID NO: 227 and CDR-L3 as depicted in SEQ ID NO: 228, CDR-H1 as depicted in SEQ ID NO: 64, CDR-H2 as depicted in SEQ ID NO: 65, CDR-H3 as depicted in SEQ ID NO: 66, CDR-L1 as depicted in SEQ ID NO: 232, CDR-L2 as depicted in SEQ ID NO: 233 and CDR-L3 as depicted in SEQ ID NO: 234, CDR-H1 as depicted in SEQ ID NO: 70, CDR-H2 as depicted in SEQ ID NO: 71, CDR-H3 as depicted in SEQ ID NO: 72, CDR-L1 as depicted in SEQ ID NO: 238, CDR-L2 as depicted in SEQ ID NO: 239 and CDR-L3 as depicted in SEQ ID NO: 240, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 161, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 328, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 48, CDR-L1 as depicted in SEQ ID NO: 924, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 902, CDR-L1 as depicted in SEQ ID NO: 924, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 903, CDR-L1 as depicted in SEQ ID NO: 924, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 46, CDR-H2 as depicted in SEQ ID NO: 47, CDR-H3 as depicted in SEQ ID NO: 48, CDR-L1 as depicted in SEQ ID NO: 925, CDR-L2 as depicted in SEQ ID NO: 215 and CDR-L3 as depicted in SEQ ID NO: 216, CDR-H1 as depicted in SEQ ID NO: 70, CDR-H2 as depicted in SEQ ID NO: 907, CDR-H3 as depicted in SEQ ID NO: 72, CDR-L1 as depicted in SEQ ID NO: 238, CDR-L2 as depicted in SEQ ID NO: 239 and CDR-L3 as depicted in SEQ ID NO: 240, CDR-H1 as depicted in SEQ ID NO: 70, CDR-H2 as depicted in SEQ ID NO: 907, CDR-H3 as depicted in SEQ ID NO: 908, CDR-L1 as depicted in SEQ ID NO: 238, CDR-L2 as depicted in SEQ ID NO: 239 and CDR-L3 as depicted in SEQ ID NO: 240, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 901, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 922, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, CDR-H1 as depicted in SEQ ID NO: 58, CDR-H2 as depicted in SEQ ID NO: 905, CDR-H3 as depicted in SEQ ID NO: 906, CDR-L1 as depicted in SEQ ID NO: 226, CDR-L2 as depicted in SEQ ID NO: 227 and CDR-L3 as depicted in SEQ ID NO: 228, CDR-H1 as depicted in SEQ ID NO: 58, CDR-H2 as depicted in SEQ ID NO: 905, CDR-H3 as depicted in SEQ ID NO: 60, CDR-L1 as depicted in SEQ ID NO: 226, CDR-L2 as depicted in SEQ ID NO: 227 and CDR-L3 as depicted in SEQ ID NO: 228, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 161, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 939, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 921, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 939, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 940, CDR-H1 as depicted in SEQ ID NO: 160, CDR-H2 as depicted in SEQ ID NO: 161, CDR-H3 as depicted in SEQ ID NO: 162, CDR-L1 as depicted in SEQ ID NO: 941, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 196, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 922, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 901, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 922, CDR-L2 as depicted in SEQ ID NO: 197 and CDR-L3 as depicted in SEQ ID NO: 923, and
CDR-H1 as depicted in SEQ ID NO: 28, CDR-H2 as depicted in SEQ ID NO: 29, CDR-H3 as depicted in SEQ ID NO: 30, CDR-L1 as depicted in SEQ ID NO: 939, CDR-L2 as depicted in SEQ ID NO: 329 and CDR-L3 as depicted in SEQ ID NO: 330; which all characterize binding domains for CDH19 grouped into bin 4; and [0231] (e) CDR-H1 as depicted in SEQ ID NO: 76, CDR-H2 as depicted in SEQ ID NO: 77, CDR-H3 as depicted in SEQ ID NO: 78, CDR-L1 as depicted in SEQ ID NO: 244, CDR-L2 as depicted in SEQ ID NO: 245 and CDR-L3 as depicted in SEQ ID NO: 246, CDR-H1 as depicted in SEQ ID NO: 88, CDR-H2 as depicted in SEQ ID NO: 89, CDR-H3 as depicted in SEQ ID NO: 90, CDR-L1 as depicted in SEQ ID NO: 256, CDR-L2 as depicted in SEQ ID NO: 257 and CDR-L3 as depicted in SEQ ID NO: 258, CDR-H1 as depicted in SEQ ID NO: 106, CDR-H2 as depicted in SEQ ID NO: 107, CDR-H3 as depicted in SEQ ID NO: 108, CDR-L1 as depicted in SEQ ID NO: 274, CDR-L2 as depicted in SEQ ID NO: 275 and CDR-L3 as depicted in SEQ ID NO: 276, CDR-H1 as depicted in SEQ ID NO: 112, CDR-H2 as depicted in SEQ ID NO: 113, CDR-H3 as depicted in SEQ ID NO: 114, CDR-L1 as depicted in SEQ ID NO: 280, CDR-L2 as depicted in SEQ ID NO: 281 and CDR-L3 as depicted in SEQ ID NO: 282, and CDR-H1 as depicted in SEQ ID NO: 106, CDR-H2 as depicted in SEQ ID NO: 107, CDR-H3 as depicted in SEQ ID NO: 108, CDR-L1 as depicted in SEQ ID NO: 274, CDR-L2 as depicted in SEQ ID NO: 275 and CDR-L3 as depicted in SEQ ID NO: 276 which all characterize binding domains for CDH19 grouped into bin 5;
[0232] In a further embodiment of the human antibody or antigen binding fragment thereof of the invention the human binding domain or antigen binding fragment thereof comprises a VH region selected from the group consisting of VH regions [0233] (a) as depicted in SEQ ID NO: 362, SEQ ID NO: 364, SEQ ID NO: 485, SEQ ID NO: 486, SEQ ID NO: 487, SEQ ID NO: 492, SEQ ID NO: 493, SEQ ID NO: 494, and SEQ ID NO: 495; [0234] which all characterize binding domains for CDH19 grouped into bin 1; [0235] (b) as depicted in SEQ ID NO: 342, SEQ ID NO: 366, SEQ ID NO: 370, SEQ ID NO: 344, SEQ ID NO: 372, SEQ ID NO: 368, SEQ ID NO: 496, SEQ ID NO: 497, SEQ ID NO: 498, SEQ ID NO: 499, SEQ ID NO: 500, SEQ ID NO: 508, SEQ ID NO: 509, SEQ ID NO: 510, SEQ ID NO: 511, SEQ ID NO: 512, SEQ ID NO: 519, SEQ ID NO: 520, SEQ ID NO: 521, SEQ ID NO: 522, SEQ ID NO: 523, SEQ ID NO: 524, SEQ ID NO: 525, SEQ ID NO: 526, SEQ ID NO: 527, SEQ ID NO: 528, SEQ ID NO: 529, SEQ ID NO: 530, SEQ ID NO: 531, SEQ ID NO: 532, SEQ ID NO: 533, SEQ ID NO: 534, SEQ ID NO: 535, SEQ ID NO: 536, SEQ ID NO: 537, and SEQ ID NO: 538; [0236] which all characterize binding domains for CDH19 grouped into bin 2; [0237] (c) as depicted in SEQ ID NO: 338, SEQ ID NO: 354, SEQ ID NO: 378, SEQ ID NO: 356, SEQ ID NO: 476, SEQ ID NO: 477, SEQ ID NO: 478, SEQ ID NO: 479, SEQ ID NO: 480, SEQ ID NO: 481, SEQ ID NO: 482, SEQ ID NO: 483, SEQ ID NO: 484, SEQ ID NO: 501, SEQ ID NO: 502, SEQ ID NO: 503, SEQ ID NO: 504, SEQ ID NO: 505, SEQ ID NO: 506, SEQ ID NO: 517, and SEQ ID NO: 518; [0238] which all characterize binding domains for CDH19 grouped into bin 3; [0239] (d) as depicted in SEQ ID NO: 352, SEQ ID NO: 360, SEQ ID NO: 388, SEQ ID NO: 386, SEQ ID NO: 340, SEQ ID NO: 346, SEQ ID NO: 374, SEQ ID NO: 348, SEQ ID NO: 390, SEQ ID NO: 463, SEQ ID NO: 464, SEQ ID NO: 465, SEQ ID NO: 466, SEQ ID NO: 467, SEQ ID NO: 468, SEQ ID NO: 469, SEQ ID NO: 470, SEQ ID NO: 471, SEQ ID NO: 472, SEQ ID NO: 473, SEQ ID NO: 474, SEQ ID NO: 475, SEQ ID NO: 488, SEQ ID NO: 489, SEQ ID NO: 490, SEQ ID NO: 491, SEQ ID NO: 513, SEQ ID NO: 514, SEQ ID NO: 515, SEQ ID NO: 516, SEQ ID NO: 540, SEQ ID NO: 541, SEQ ID NO: 542, and SEQ ID NO: 543; [0240] which all characterize binding domains for CDH19 grouped into bin 4; and [0241] (e) as depicted in SEQ ID NO: 376, SEQ ID NO: 392, SEQ ID NO: 358, SEQ ID NO: 350, and SEQ ID NO: 507; [0242] which all characterize binding domains for CDH19 grouped into bin 5.
[0243] In another embodiment the human antibody or antigen binding fragment thereof of the invention comprises the human binding domain or antigen binding fragment thereof comprising a VL region selected from the group consisting of VL regions [0244] (a) as depicted in SEQ ID NO: 418, SEQ ID NO: 420, SEQ ID NO: 580, SEQ ID NO: 581, SEQ ID NO: 582, SEQ ID NO: 587, SEQ ID NO: 588, SEQ ID NO: 589, and SEQ ID NO: 590; [0245] which all characterize binding domains for CDH19 grouped into bin 1; [0246] (b) as depicted in SEQ ID NO: 398, SEQ ID NO: 422, SEQ ID NO: 426, SEQ ID NO: 400, SEQ ID NO: 428, SEQ ID NO: 424, SEQ ID NO: 591, SEQ ID NO: 592, SEQ ID NO: 593, SEQ ID NO: 594, SEQ ID NO: 595, SEQ ID NO: 603, SEQ ID NO: 604, SEQ ID NO: 605, SEQ ID NO: 606, SEQ ID NO: 607, SEQ ID NO: 614, SEQ ID NO: 615, SEQ ID NO: 616, SEQ ID NO: 617, SEQ ID NO: 618, SEQ ID NO: 619, SEQ ID NO: 620, SEQ ID NO: 621, SEQ ID NO: 622, SEQ ID NO: 623, SEQ ID NO: 624, SEQ ID NO: 625, SEQ ID NO: 626, SEQ ID NO: 627, SEQ ID NO: 628, SEQ ID NO: 629, SEQ ID NO: 630, SEQ ID NO: 631, SEQ ID NO: 632, and SEQ ID NO: 633; [0247] which all characterize binding domains for CDH19 grouped into bin 2; [0248] (c) as depicted in SEQ ID NO: 394, SEQ ID NO: 410, SEQ ID NO: 434, SEQ ID NO: 412, SEQ ID NO: 571, SEQ ID NO: 572, SEQ ID NO: 573, SEQ ID NO: 574, SEQ ID NO: 575, SEQ ID NO: 576, SEQ ID NO: 577, SEQ ID NO: 578, SEQ ID NO: 579, SEQ ID NO: 596, SEQ ID NO: 597, SEQ ID NO: 598, SEQ ID NO: 599, SEQ ID NO: 600, SEQ ID NO: 601, SEQ ID NO: 612, and SEQ ID NO: 613; [0249] which all characterize binding domains for CDH19 grouped into bin 3; [0250] (d) as depicted in SEQ ID NO: 408, SEQ ID NO: 416, SEQ ID NO: 444, SEQ ID NO: 442, SEQ ID NO: 396, SEQ ID NO: 402, SEQ ID NO: 430, SEQ ID NO: 404, SEQ ID NO: 446, SEQ ID NO: 558, SEQ ID NO: 559, SEQ ID NO: 560, SEQ ID NO: 561, SEQ ID NO: 562, SEQ ID NO: 563, SEQ ID NO: 564, SEQ ID NO: 565, SEQ ID NO: 566, SEQ ID NO: 567, SEQ ID NO: 568, SEQ ID NO: 569, SEQ ID NO: 570, SEQ ID NO: 583, SEQ ID NO: 584, SEQ ID NO: 585, SEQ ID NO: 586, SEQ ID NO: 608, SEQ ID NO: 609, SEQ ID NO: 610, SEQ ID NO: 611, SEQ ID NO: 635, SEQ ID NO: 636, SEQ ID NO: 637, and SEQ ID NO: 638; [0251] which all characterize binding domains for CDH19 grouped into bin 4; and [0252] (e) as depicted in SEQ ID NO: 432, SEQ ID NO: 448, SEQ ID NO: 414, SEQ ID NO: 406, and SEQ ID NO: 602; [0253] which all characterize binding domains for CDH19 grouped into bin 5.
[0254] The invention further provides an embodiment of the human antibody or antigen binding fragment thereof of the invention, wherein the human binding domain or antigen binding fragment thereof comprises a VH region and a VL region selected from the group consisting of: [0255] (1) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 362+418, SEQ ID NOs: 364+420, SEQ ID NOs: 485+580, SEQ ID NOs: 486+581, SEQ ID NOs: 487+582, SEQ ID NOs: 492+587, SEQ ID NOs: 493+588, SEQ ID NOs: 494+589, and SEQ ID NOs: 495+590; [0256] all pairs grouped into bin 1; [0257] (2) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 342+398, SEQ ID NOs: 366+422, SEQ ID NOs: 370+426, SEQ ID NOs: 344+400, SEQ ID NOs: 372+428, SEQ ID NOs: 368+424, SEQ ID NOs: 496+591, SEQ ID NOs: 497+592, SEQ ID NOs: 498+593, SEQ ID NOs: 499+594, SEQ ID NOs: 500+595, SEQ ID NOs: 508+603, SEQ ID NOs: 509+604, SEQ ID NOs: 510+605, SEQ ID NOs: 511+606, SEQ ID NOs: 512+607, SEQ ID NOs: 519+614, SEQ ID NOs: 520+615, SEQ ID NOs: 521+616, SEQ ID NOs: 522+617, SEQ ID NOs: 523+618, SEQ ID NOs: 524+619, SEQ ID NOs: 525+620, SEQ ID NOs: 526+621, SEQ ID NOs: 527+622, SEQ ID NOs: 528+623, SEQ ID NOs: 529+624, SEQ ID NOs: 530+625, SEQ ID NOs: 531+626, SEQ ID NOs: 532+627, SEQ ID NOs: 533+628, SEQ ID NOs: 534+629, SEQ ID NOs: 535+630, SEQ ID NOs: 536+631, SEQ ID NOs: 537+632, and SEQ ID NOs: 538+633; [0258] all pairs grouped into bin 2; [0259] (3) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 338+394, SEQ ID NOs: 354+410, SEQ ID NOs: 378+434, SEQ ID NOs: 356+412, SEQ ID NOs: 476+571, SEQ ID NOs: 477+572, SEQ ID NOs: 478+573, SEQ ID NOs: 479+574, SEQ ID NOs: 480+575, SEQ ID NOs: 481+576, SEQ ID NOs: 482+577, SEQ ID NOs: 483+578, SEQ ID NOs: 484+579, SEQ ID NOs: 501+596, SEQ ID NOs: 502+597, SEQ ID NOs: 503+598, SEQ ID NOs: 504+599, SEQ ID NOs: 505+600, SEQ ID NOs: 506+601, SEQ ID NOs: 517+612, and SEQ ID NOs: 518+613; [0260] all pairs grouped into bin 3; [0261] (4) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 352+408, SEQ ID NOs: 360+416, SEQ ID NOs: 388+444, SEQ ID NOs: 386+442, SEQ ID NOs: 340+396, SEQ ID NOs: 346+402, SEQ ID NOs: 374+430, SEQ ID NOs: 348+404, SEQ ID NOs: 390+446, SEQ ID NOs: 463+558, SEQ ID NOs: 464+559, SEQ ID NOs: 465+560, SEQ ID NOs: 466+561, SEQ ID NOs: 467+562, SEQ ID NOs: 468+563, SEQ ID NOs: 469+564, SEQ ID NOs: 470+565, SEQ ID NOs: 471+566, SEQ ID NOs: 472+567, SEQ ID NOs: 473+568, SEQ ID NOs: 474+569, SEQ ID NOs: 475+570, SEQ ID NOs: 488+583, SEQ ID NOs: 489+584, SEQ ID NOs: 490+585, SEQ ID NOs: 491+586, SEQ ID NOs: 513+608, SEQ ID NOs: 514+609, SEQ ID NOs: 515+610, SEQ ID NOs: 516+611, SEQ ID NOs: 540+635, SEQ ID NOs: 541+636, SEQ ID NOs: 542+637, and SEQ ID NOs: 543+638; [0262] all pairs grouped into bin 4; and [0263] (5) pairs of a VH region and a VL region as depicted in SEQ ID NOs: 376+432, SEQ ID NOs: 392+448, SEQ ID NOs: 358+414, SEQ ID NOs: 350+406, and SEQ ID NOs: 507+602; [0264] all pairs grouped into bin 5.
[0265] In a further embodiment the human binding domain or antigen binding fragment thereof comprises the groups of heavy and light chains having an amino acid sequence selected from the group consisting of [0266] (1) a heavy and light chain as depicted in SEQ ID NOs: 644+680, SEQ ID NOs: 650+686, SEQ ID NOs: 747+842, SEQ ID NOs: 748+843, SEQ ID NOs: 749+844, SEQ ID NOs: 754+849, SEQ ID NOs: 755+850, SEQ ID NOs: 756+851, and SEQ ID NOs: 757+852; [0267] all pairs grouped into bin 1; [0268] (2) a heavy and light chain as depicted in SEQ ID NOs: 660+696, SEQ ID NOs: 662+698, SEQ ID NOs: 668+704, SEQ ID NOs: 674+710, SEQ ID NOs: 672+708, SEQ ID NOs: 658+694, SEQ ID NOs: 758+853, SEQ ID NOs: 759+854, SEQ ID NOs: 760+855, SEQ ID NOs: 761+856, SEQ ID NOs: 762+857, SEQ ID NOs: 770+865, SEQ ID NOs: 771+866, SEQ ID NOs: 772+867, SEQ ID NOs: 773+868, SEQ ID NOs: 774+869, SEQ ID NOs: 781+876, SEQ ID NOs: 782+877, SEQ ID NOs: 783+878, SEQ ID NOs: 784+879, SEQ ID NOs: 785+880, SEQ ID NOs: 786+881, SEQ ID NOs: 787+882, SEQ ID NOs: 788+883, SEQ ID NOs: 789+884, SEQ ID NOs: 790+885, SEQ ID NOs: 791+886, SEQ ID NOs: 792+887, SEQ ID NOs: 793+888, SEQ ID NOs: 794+889, SEQ ID NOs: 795+890, SEQ ID NOs: 796+891, SEQ ID NOs: 797+892, SEQ ID NOs: 798+893, SEQ ID NOs: 799+894, and SEQ ID NOs: 800+895; [0269] all pairs grouped into bin 2; [0270] (3) a a heavy and light chain as depicted in SEQ ID NOs: 656+692, SEQ ID NOs: 654+690, SEQ ID NOs: 664+700, SEQ ID NOs: 670+706, SEQ ID NOs: 738+833, SEQ ID NOs: 739+834, SEQ ID NOs: 740+835, SEQ ID NOs: 741+836, SEQ ID NOs: 742+837, SEQ ID NOs: 743+838, SEQ ID NOs: 744+839, SEQ ID NOs: 745+840, SEQ ID NOs: 746+841, SEQ ID NOs: 763+858, SEQ ID NOs: 764+859, SEQ ID NOs: 765+860, SEQ ID NOs: 766+861, SEQ ID NOs: 767+862, SEQ ID NOs: 768+863, SEQ ID NOs: 779+874, and SEQ ID NOs: 780+875; [0271] all pairs grouped into bin 3; [0272] (4) a heavy and light chain as depicted in SEQ ID NOs: 640+676, SEQ ID NOs: 642+678, SEQ ID NOs: 646+682, SEQ ID NOs: 648+684, SEQ ID NOs: 666+702, SEQ ID NOs: 725+820, SEQ ID NOs: 726+821, SEQ ID NOs: 727+822, SEQ ID NOs: 728+823, SEQ ID NOs: 729+824, SEQ ID NOs: 730+825, SEQ ID NOs: 731+826, SEQ ID NOs: 732+827, SEQ ID NOs: 733+828, SEQ ID NOs: 734+829, SEQ ID NOs: 735+830, SEQ ID NOs: 736+831, SEQ ID NOs: 737+832, SEQ ID NOs: 750+845, SEQ ID NOs: 751+846, SEQ ID NOs: 752+847, SEQ ID NOs: 753+848, SEQ ID NOs: 775+870, SEQ ID NOs: 776+871, SEQ ID NOs: 777+872, SEQ ID NOs: 778+873, SEQ ID NOs: 802+897, SEQ ID NOs: 803+898, SEQ ID NOs: 804+899, and SEQ ID NOs: 805+900; [0273] all pairs grouped into bin 4; and [0274] (5) a heavy and light chain as depicted in SEQ ID NOs: 652+688, and SEQ ID NOs: 769+864 all pairs grouped into bin 5.
[0275] In another embodiment the invention is directed to an antibody construct comprising the human antibody or antigen binding fragment thereof capable of binding to human CDH19 on the surface of a target cell as described above that is conjugated to a chemotherapeutic agent.
[0276] In one embodiment of the antibody construct of the invention a linker conjugates the chemotherapeutic agent to the human antibody or antigen binding fragment thereof. Accordingly, embodiments of the antibody construct comprising of the invention include antibody drug conjugates (ADCs). Generally the antibody construct comprising of the invention comprises an antibody conjugated to a chemotherapeutic agent, e.g., a cytotoxic agent, a cytostatic agent, a toxin, or a radioactive agent. A linker molecule can be used to conjugate the drug to the antibody. A wide variety of linkers and drugs useful e.g. in ADC technology are known in the art and may be used in embodiments of the present invention. (See US20090028856; US2009/0274713; US2007/0031402; WO2005/084390; WO2009/099728; U.S. Pat. Nos. 5,208,020; 5,416,064; 5,475,092; 5,585,499; 6,436,931; 6,372,738; and 6,340,701, all incorporated herein by reference).
[0277] In certain embodiments, the antibody construct comprising of the invention comprises a linker made up of one or more linker components. Exemplary linker components include 6-maleimidocaproyl, maleimidopropanoyl, valine-citrulline, alanine-phenylalanine, p-aminobenzyloxycarbonyl, and those resulting from conjugation with linker reagents, including, but not limited to, N-succinimidyl 4-(2-pyridylthio) pentanoate ("SPP"), N-succinimidyl 4-(N-maleimidomethyl) cyclohexane-1 carboxylate ("SMCC," also referred to herein also as "MCC"), and N-succinimidyl (4-iodo-acetyl) aminobenzoate ("SIAB"). Linkers may be a "cleavable" linker or a "non-cleavable" linker (Ducry and Stump, Bioconjugate Chem. 2010, 21, 5-13; incorporated herein by reference in its entirety) Cleavable linkers are designed to release the drug when subjected to certain environment factors, e.g., when internalized into the target cell. Cleavable linkers include acid labile linkers, protease sensitive linkers, photolabile linkers, dimethyl linker or disulfide-containing linkers. Non-cleavable linkers tend to remain covalently associated with at least one amino acid of the antibody and the drug upon internalization by and degradation within the target cell. An exemplary non-cleavable linker is MCC.
[0278] In a preferred embodiment of the antibody construct of the invention the linker is a non-cleavable linker.
[0279] It is also preferred that the linker in the antibody construct of the invention comprises MCC.
[0280] In a further embodiment of the antibody construct of the invention the chemotherapeutic agent is conjugated to one or more lysines contained in the human antibody or antigen binding fragment thereof.
[0281] In certain embodiments, the antibody of the invention is conjugated to a chemotherapeutic agent. Examples of chemotherapeutic agents include alkylating agents, such as thiotepa and cyclophosphamide (CYTOXAN.TM.); alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines, such as benzodopa, carboquone, meturedopa, and uredopa; ethylenimines and methylamelamines including altretamine, triethylenemelamine, trietylenephosphoramide, triethylenethiophosphaoramide and trimethylolomelamine; acetogenins (especially bullatacin and bullatacinone); a camptothecin (including the synthetic analogue topotecan); bryostatin; callystatin; CC-1065 (including its adozelesin, carzelesin and bizelesin synthetic analogues); cryptophycins (particularly cryptophycin 1 and cryptophycin 8); dolastatin; duocarmycin (including the synthetic analogues, KW-2189 and CBI-TMI); eleutherobin; pancratistatin; a sarcodictyin; spongistatin; nitrogen mustards such as chlorambucil, chlomaphazine, cholophosphamide, estramustine, ifosfamide, mechlorethamine, mechlorethamine oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, trofosfamide, uracil mustard; nitrosureas such as carmustine, chlorozotocin, fotemustine, lomustine, nimustine, ranimustine; antibiotics, such as the enediyne antibiotics (e.g. calicheamicin, especially calicheamicin .gammal and calicheamicin theta I, see, e.g., Angew Chem. Intl. Ed. Engl. 33:183-186 (1994); dynemicin, including dynemicin A; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antiobiotic chromomophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, caminomycin, carzinophilin; chromomycins, dactinomycin, daunorubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, doxorubicin (including morpholino-doxorubicin, cyanomorpholino-doxorubicin, 2-pyrrolino-doxorubicin and deoxydoxorubicin), epirubicin, esorubicin, idarubicin, marcellomycin, nitomycins, mycophenolic acid, nogalamycin, olivomycins, peplomycin, potfiromycin, puromycin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zorubicin; anti-metabolites, such as methotrexate and 5-fluorouracil (5-FU); folic acid analogues, such as denopterin, methotrexate, pteropterin, trimetrexate; purine analogs, such as fludarabine, 6-mercaptopurine, thiamiprine, thioguanine; pyrimidine analogs such as, ancitabine, azacitidine, 6-azauridine, carmofur, cytarabine, dideoxyuridine, doxifluridine, enocitabine, floxuridine, 5-FU; androgens, such as calusterone, dromostanolone propionate, epitiostanol, mepitiostane, testolactone; anti-adrenals, such as aminoglutethimide, mitotane, trilostane; folic acid replenisher, such as frolinic acid; aceglatone; aldophosphamide glycoside; aminolevulinic acid; amsacrine; bestrabucil; bisantrene; edatraxate; defofamine; demecolcine; diaziquone; elfomithine; elliptinium acetate; an epothilone; etoglucid; gallium nitrate; hydroxyurea; lentinan; lonidamine; maytansinoids, such as maytansine and ansamitocins; mitoguazone; mitoxantrone; mopidamol; nitracrine; pentostatin; phenamet; pirarubicin; podophyllinic acid; 2-ethylhydrazide; procarbazine; PSK.RTM.; razoxane; rhizoxin; sizofuran; spirogermanium; tenuazonic acid; triaziquone; 2,2',2''-trichlorotriethylamine; trichothecenes (especially T-2 toxin, verracurin A, roridin A and anguidine); urethan; vindesine; dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside ("Ara-C"); cyclophosphamide; thiotepa; taxoids, e.g. paclitaxel (TAXOL.TM., Bristol-Myers Squibb Oncology, Princeton, N.J.) and doxetaxel (TAXOTERE.RTM., Rhone-Poulenc Rorer, Antony, France); chlorambucil; gemcitabine; 6-thioguanine; mercaptopurine; methotrexate; platinum analogs such as cisplatin and carboplatin; vinblastine; platinum; etoposide (VP-16); ifosfamide; mitomycin C; mitoxantrone; vincristine; vinorelbine; navelbine; novantrone; teniposide; 65 daunomycin; aminopterin; xeloda; ibandronate; CPT-11; topoisomerase inhibitor RFS 2000; difluoromethylomithine (DMFO); retinoic acid; capecitabine; and pharmaceutically acceptable salts, acids or derivatives of any of the above. Also included in this definition are anti-hormonal agents that act to regulate or inhibit hormone action on tumors, such as anti-estrogens including for example tamoxifen, raloxifene, aromatase inhibiting 4(5)-imidazoles, 4-hydroxytamoxifen, trioxifene, keoxifene, LY117018, onapristone, and toremifene (Fareston); and anti-androgens, such as flutamide, nilutamide, bicalutamide, leuprolide, and goserelin; siRNA and pharmaceutically acceptable salts, acids or derivatives of any of the above. Other chemotherapeutic agents that can be used with the present invention are disclosed in US Publication No. 20080171040 or US Publication No. 20080305044 and are incorporated in their entirety by reference.
[0282] It is contemplated that an antibody may be conjugated to two or more different chemotherapeutic agents or a pharmaceutical composition may comprise a mixture of antibodies wherein the antibody component is identical except for being conjugated to a different chemotherapeutic agent. Such embodiments may be useful for targeting multiple biological pathways with a target cell.
[0283] In preferred embodiments, the antibody construct comprising of the invention comprises an antibody conjugated to one or more maytansinoid molecules, which are mitotic inhibitors that act by inhibiting tubulin polymerization. Maytansinoids, including various modifications, are described in U.S. Pat. Nos. 3,896,111; 4,151,042; 4,137,230; 4,248,870; 4,256,746; 4,260,608; 4,265,814; 4,294,757; 4,307,016; 4,308,268; 4,309,428; 4,313,946; 4,315,929; 4,317,821; 4,322,348; 4331598; 4361650; 4364866; 4424219; 4450254; 4362663; 4371533; and WO 2009/099728. Maytansinoid drug moieties may be isolated from natural sources, produced using recombinant technology, or prepared synthetically. Exemplary maytansinoids include C-19-dechloro (U.S. Pat. No. 4,256,746), C-20-hydroxy (or C-20-demethyl) +/-C-19-dechloro (U.S. Pat. Nos. 4,307,016 and 4,361,650), C-20-demethoxy (or C-20-acyloxy (--OCOR), +/-dechrolo (U.S. Pat. No. 4,294,757), C-9-SH (U.S. Pat. No. 4,424,219), C-14-alkoxymethyl (demethoxy/CH2OR) (U.S. Pat. No. 4,331,598), C-14-hydroxymethyl or acyloxymethyl (CH2OH or CH2OAc) (U.S. Pat. No. 4,450,254), C-15-hydroxy/acyloxy (U.S. Pat. No. 4,364,866), C-15-methoxy (U.S. Pat. Nos. 4,313,946 and 4,315,929), C-18-N-demethyl (U.S. Pat. Nos. 4,362,663 and 4,322,348), and 4,5-deoxy (U.S. Pat. No. 4,371,533).
[0284] Various positions on maytansinoid compounds may be used as the linkage position, depending upon the type of link desired. For example, for forming an ester linkage, the C-3 position having a hydroxyl group, the C-14 position modified with hydrozymethyl, the C-15 position modified with a hydroxyl a group, and the C-20 position having a hydroxyl group are all suitable (U.S. Pat. Nos. 5,208,020, RE39,151, and 6,913,748; US Patent Appl. Pub. Nos. 20060167245 and 20070037972, and WO 2009099728).
[0285] Preferred maytansinoids include those known in the art as DM1, DM3, and DM4 (US Pat. Appl. Pub. Nos. 2009030924 and 20050276812, incorporated herein by reference). In one embodiment of the antibody construct of the invention the chemotherapeutic agent is DM1. Accordingly, in a preferred embodiment the antibody construct of the invention is an the human antibody or antigen binding fragment thereof conjugated to one or more DM1 molecules.
[0286] ADCs containing maytansinoids, methods of making such ADCs, and their therapeutic use are disclosed in U.S. Pat. Nos. 5,208,020 and 5,416,064, US Pat. Appl. Pub. No. 20050276812, and WO 2009099728 (all incorporated by reference herein). Linkers that are useful for making maytansinoid ADCs are know in the art (U.S. Pat. No. 5,208,020 and US Pat. Appl. Pub. Nos. 2005016993 and 20090274713; all incorporated herein by reference). Maytansinoid ADCs comprising an SMCC linker may be prepared as disclosed in US Pat. Publ. No. 2005/0276812.
[0287] In certain embodiments, the antibody construct comprising of the invention comprises an antibody conjugated to DM1 with an SMCC linker.
[0288] An antibody construct comprising of the invention may have 1 to 20 chemotherapeutic agents per antibody. Compositions of ADCs may be characterized by the average number of drug moieties per antibody molecule in the composition. The average number of drug moieties may be determined by conventional means such as mass spectrometry, immunoassay, and HPLC. In some instances, a homogeneous ADC population may be separated and purified by means of reverse phase HPLC or electrophoresis. Thus, pharmaceutical ADC compositions may contain a heterogeneous or homogeneous population of antibodies linked to 1, 2, 3, 4, 5, 6, 7 or more drug moieties.
[0289] Thus, in a preferred embodiment of the antibody construct of the invention the average number of DM1 molecules per antibody construct is between 1 and 10.
[0290] It is also preferred for the antibody construct of the invention that the average number of DM1 molecules per antibody construct is between 3 and 7.
[0291] Moreover, it is preferred for the antibody construct of the invention that the average number of DM1 molecules per antibody construct is between 4 and 6.
[0292] Embodiments of the invention include antibody constructs comprising an average of about 1, about 2, about 3, about 4, about 5, about 6, about 7, about 8, about 9, about 10, about 11, about 12, about 13, about 14, about 15, about 16, about 17, about 18, about 19, or about 20 DM1 molecules per antibody.
[0293] In a further alternative embodiment of the antibody construct of the invention the average number of DM1 molecules per antibody construct is about 4.0, about 4.1, about 4.2, about 4.3, about 4.4, about 4.5, about 4.6, about 4.7, about 4.8, about 4.9, about 5.0, about 5.1, about 5.2, about 5.3, about 5.4, about 5.5, about 5.6, about 5.7, about 5.8, about 5.9, or about 6.0.
[0294] In one embodiment the antibody respectively the antibody construct of the invention comprises an effector function-enhanced antibody. One of the functions of the Fc portion of an antibody is to communicate to the immune system when the antibody binds its target. This is considered "effector function". Communication leads to antibody-dependent cellular cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), and/or complement dependent cytotoxicity (CDC). ADCC and ADCP are mediated through the binding of the Fc to Fc receptors on the surface of cells of the immune system. CDC is mediated through the binding of the Fc with proteins of the complement system, e.g., C1q.
[0295] The IgG subclasses vary in their ability to mediate effector functions. For example IgG1 is much superior to IgG2 and IgG4 at mediating ADCC and CDC. Thus, in embodiments wherein a cell expressing CDH19 is targeted for destruction, an anti-CDH19 IgG1 antibody would be preferred.
[0296] The effector function of an antibody can be increased, or decreased, by introducing one or more mutations into the Fc. Embodiments of the invention include antigen binding proteins, e.g., antibodies, having an Fc engineered to increase effector function (U.S. Pat. No. 7,317,091 and Strohl, Curr. Opin. Biotech., 20:685-691, 2009; both incorporated herein by reference in its entirety). Exemplary IgG1 Fc molecules having increased effector function include (based on the Kabat numbering scheme) those have the following substitutions:
[0297] S239D/I332E
[0298] S239D/A330S/I332E
[0299] S239D/A330L/I332E
[0300] S298A/D333A/K334A
[0301] P247I/A339D
[0302] P247I/A339Q
[0303] D280H/K290S
[0304] D280H/K290S/S298D
[0305] D280H/K290S/S298V
[0306] F243L/R292P/Y300L
[0307] F243L/R292P/Y300L/P396L
[0308] F243L/R292P/Y300L/V3051/P396L
[0309] G236A/S239D/I332E
[0310] K326A/E333A
[0311] K326W/E333S
[0312] K290E/S298G/T299A
[0313] K290N/S298G/T299A
[0314] K290E/5298G/T299A/K326E
[0315] K290N/S298G/T299A/K326E
[0316] Further embodiments of the invention include antibodies, having an Fc engineered to decrease effector function. Exemplary Fc molecules having decreased effector function include (based on the Kabat numbering scheme) those have the following substitutions:
[0317] N297A (IgG1)
[0318] L234A/L235A (IgG1)
[0319] V234A/G237A (IgG2)
[0320] L235A/G237A/E318A (IgG4)
[0321] H2680/V309L/A330S/A331S (IgG2)
[0322] C220S/C226S/C229S/P238S (IgG1)
[0323] C226S/C229S/E233P/L234V/L235A (IgG1)
[0324] L234F/L235E/P331S (IgG1)
[0325] S267E/L328F (IgG1)
[0326] Another method of increasing effector function of IgG Fc-containing proteins is by reducing the fucosylation of the Fc. Removal of the core fucose from the biantennary complex-type oligosachharides attached to the Fc greatly increased ADCC effector function without altering antigen binding or CDC effector function. Several ways are known for reducing or abolishing fucosylation of Fc-containing molecules, e.g., antibodies. These include recombinant expression in certain mammalian cell lines including a FUT8 knockout cell line, variant CHO line Lec13, rat hybridoma cell line YB2/0, a cell line comprising a small interfering RNA specifically against the FUT8 gene, and a cell line coexpressing B-1,4-N-acetylglucosaminyltransferase III and Golgi .alpha.-mannosidase II. Alternatively, the Fc-containing molecule may be expressed in a non-mammalian cell such as a plant cell, yeast, or prokaryotic cell, e.g., E. coli. Thus, in certain embodiments of the invention, a composition comprises an antibody, e.g., Ab1, Ab2, Ab3, Ab4, Ab5, Ab6, Ab7, or Ab8, having reduced fucosylation or lacking fucosylation altogether.
[0327] The invention further provides an isolated nucleic acid molecule or sequence encoding a human antibody or antigen binding fragment thereof of the invention.
[0328] Furthermore, the invention provides a vector comprising a nucleic acid sequence of the invention. Moreover, the invention provides a host cell transformed or transfected with the nucleic acid sequence of the invention or with a vector comprising the nucleic acid molecule.
[0329] In a further embodiment the invention provides a process for the production of a human antibody or an antigen binding fragment thereof of the invention, said process comprising culturing a host cell of the invention under conditions allowing the expression of the human antibody or antigen binding fragment thereof of the invention and recovering the produced antibody or antigen binding fragment thereof from the culture.
[0330] In a further embodiment the invention provides a process for the production of an antibody construct comprising a human antibody or an antigen binding fragment thereof of the invention, said process comprising culturing a host cell of the invention under conditions allowing the expression of the human antibody or antigen binding fragment thereof of the invention and recovering the produced antibody or antigen binding fragment thereof from the culture, and conjugating a chemotherapeutic agent to the recovered antibody or antigen binding fragment thereof to produce the antibody conjugate.
[0331] Moreover, the invention provides a pharmaceutical composition comprising a human antibody or antigen binding fragment thereof of the invention or an antibody construct of the invention or produced according to the process of the invention in admixture with a pharmaceutically acceptable carrier thereof.
[0332] The formulations described herein are useful as pharmaceutical compositions in the treatment, amelioration and/or prevention of the pathological medical condition as described herein in a patient in need thereof. The term "treatment" refers to both therapeutic treatment and prophylactic or preventative measures. Treatment includes the application or administration of the formulation to the body, an isolated tissue, or cell from a patient who has a disease/disorder, a symptom of a disease/disorder, or a predisposition toward a disease/disorder, with the purpose to cure, heal, alleviate, relieve, alter, remedy, ameliorate, improve, or affect the disease, the symptom of the disease, or the predisposition toward the disease.
[0333] Those "in need of treatment" include those already with the disorder, as well as those in which the disorder is to be prevented. The term "disease" is any condition that would benefit from treatment with the protein formulation described herein. This includes chronic and acute disorders or diseases including those pathological conditions that predispose the mammal to the disease in question. Non-limiting examples of diseases/disorders to be treated herein include proliferative disease, a tumorous disease, or an immunological disorder.
[0334] In some embodiments, the invention provides a pharmaceutical composition comprising a therapeutically effective amount of one or a plurality of the a human antibody or antigen binding fragment thereof of the invention or an antibody construct of the invention together with a pharmaceutically effective diluents, carrier, solubilizer, emulsifier, preservative, and/or adjuvant. In certain embodiments, the antigen binding protein is an antibody, including a drug-conjugated antibody or a bispecific antibody. Pharmaceutical compositions of the invention include, but are not limited to, liquid, frozen, and lyophilized compositions.
[0335] Preferably, formulation materials are nontoxic to recipients at the dosages and concentrations employed. In specific embodiments, pharmaceutical compositions comprising a therapeutically effective amount of a human antibody or antigen binding fragment thereof of the invention or an antibody construct of the invention.
[0336] In certain embodiments, the pharmaceutical composition may contain formulation materials for modifying, maintaining or preserving, for example, the pH, osmolarity, viscosity, clarity, color, isotonicity, odor, sterility, stability, rate of dissolution or release, adsorption or penetration of the composition. In such embodiments, suitable formulation materials include, but are not limited to, amino acids (such as glycine, glutamine, asparagine, arginine, proline, or lysine); antimicrobials; antioxidants (such as ascorbic acid, sodium sulfite or sodium hydrogen-sulfite); buffers (such as borate, bicarbonate, Tris-HCl, citrates, phosphates or other organic acids); bulking agents (such as mannitol or glycine); chelating agents (such as ethylenediamine tetraacetic acid (EDTA)); complexing agents (such as caffeine, polyvinylpyrrolidone, beta-cyclodextrin or hydroxypropyl-beta-cyclodextrin); fillers; monosaccharides; disaccharides; and other carbohydrates (such as glucose, mannose or dextrins); proteins (such as serum albumin, gelatin or immunoglobulins); coloring, flavoring and diluting agents; emulsifying agents; hydrophilic polymers (such as polyvinylpyrrolidone); low molecular weight polypeptides; salt-forming counterions (such as sodium); preservatives (such as benzalkonium chloride, benzoic acid, salicylic acid, thimerosal, phenethyl alcohol, methylparaben, propylparaben, chlorhexidine, sorbic acid or hydrogen peroxide); solvents (such as glycerin, propylene glycol or polyethylene glycol); sugar alcohols (such as mannitol or sorbitol); suspending agents; surfactants or wetting agents (such as pluronics, PEG, sorbitan esters, polysorbates such as polysorbate 20, polysorbate, triton, tromethamine, lecithin, cholesterol, tyloxapal); stability enhancing agents (such as sucrose or sorbitol); tonicity enhancing agents (such as alkali metal halides, preferably sodium or potassium chloride, mannitol sorbitol); delivery vehicles; diluents; excipients and/or pharmaceutical adjuvants. See, REMINGTON'S PHARMACEUTICAL SCIENCES, 18'' Edition, (A. R. Genrmo, ed.), 1990, Mack Publishing Company.
[0337] In certain embodiments, the optimal pharmaceutical composition will be determined by one skilled in the art depending upon, for example, the intended route of administration, delivery format and desired dosage. See, for example, REMINGTON'S PHARMACEUTICAL SCIENCES, supra. In certain embodiments, such compositions may influence the physical state, stability, rate of in vivo release and rate of in vivo clearance of the antigen binding proteins of the invention. In certain embodiments, the primary vehicle or carrier in a pharmaceutical composition may be either aqueous or non-aqueous in nature. For example, a suitable vehicle or carrier may be water for injection, physiological saline solution or artificial cerebrospinal fluid, possibly supplemented with other materials common in compositions for parenteral administration. Neutral buffered saline or saline mixed with serum albumin are further exemplary vehicles. In specific embodiments, pharmaceutical compositions comprise Tris buffer of about pH 7.0-8.5, or acetate buffer of about pH 4.0-5.5, and may further include sorbitol or a suitable substitute therefore. In certain embodiments of the invention, human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention compositions may be prepared for storage by mixing the selected composition having the desired degree of purity with optional formulation agents (REMINGTON'S PHARMACEUTICAL SCIENCES, supra) in the form of a lyophilized cake or an aqueous solution. Further, in certain embodiments, the human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention may be formulated as a lyophilizate using appropriate excipients such as sucrose.
[0338] The pharmaceutical compositions of the invention can be selected for parenteral delivery. Alternatively, the compositions may be selected for inhalation or for delivery through the digestive tract, such as orally. Preparation of such pharmaceutically acceptable compositions is within the skill of the art. The formulation components are present preferably in concentrations that are acceptable to the site of administration. In certain embodiments, buffers are used to maintain the composition at physiological pH or at a slightly lower pH, typically within a pH range of from about 5 to about 8.
[0339] When parenteral administration is contemplated, the therapeutic compositions for use in this invention may be provided in the form of a pyrogen-free, parenterally acceptable aqueous solution comprising the desired human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention in a pharmaceutically acceptable vehicle. A particularly suitable vehicle for parenteral injection is sterile distilled water in which the human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention is formulated as a sterile, isotonic solution, properly preserved. In certain embodiments, the preparation can involve the formulation of the desired molecule with an agent, such as injectable microspheres, bio-erodible particles, polymeric compounds (such as polylactic acid or polyglycolic acid), beads or liposomes, that may provide controlled or sustained release of the product which can be delivered via depot injection. In certain embodiments, hyaluronic acid may also be used, having the effect of promoting sustained duration in the circulation. In certain embodiments, implantable drug delivery devices may be used to introduce the desired antigen binding protein.
[0340] Additional pharmaceutical compositions will be evident to those skilled in the art, including formulations involving human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention in sustained- or controlled-delivery formulations. Techniques for formulating a variety of other sustained- or controlled-delivery means, such as liposome carriers, bio-erodible microparticles or porous beads and depot injections, are also known to those skilled in the art. See, for example, International Patent Application No. PCT/US93/00829, which is incorporated by reference and describes controlled release of porous polymeric microparticles for delivery of pharmaceutical compositions. Sustained-release preparations may include semipermeable polymer matrices in the form of shaped articles, e.g., films, or microcapsules. Sustained release matrices may include polyesters, hydrogels, polylactides (as disclosed in U.S. Pat. No. 3,773,919 and European Patent Application Publication No. EP 058481, each of which is incorporated by reference), copolymers of L-glutamic acid and gamma ethyl-L-glutamate (Sidman et al., 1983, Biopolymers 2:547-556), poly (2-hydroxyethyl-methacrylate) (Langer et al., 1981, J. Biomed. Mater. Res. 15:167-277 and Langer, 1982, Chem. Tech. 12:98-105), ethylene vinyl acetate (Langer et al., 1981, supra) or poly-D(-)-3-hydroxybutyric acid (European Patent Application Publication No. EP 133,988). Sustained release compositions may also include liposomes that can be prepared by any of several methods known in the art. See, e.g., Eppstein et al., 1985, Proc. Natl. Acad. Sci. U.S.A. 82:3688-3692; European Patent Application Publication Nos. EP 036,676; EP 088,046 and EP 143,949, incorporated by reference.
[0341] Pharmaceutical compositions used for in vivo administration are typically provided as sterile preparations. Sterilization can be accomplished by filtration through sterile filtration membranes. When the composition is lyophilized, sterilization using this method may be conducted either prior to or following lyophilization and reconstitution. Compositions for parenteral administration can be stored in lyophilized form or in a solution. Parenteral compositions generally are placed into a container having a sterile access port, for example, an intravenous solution bag or vial having a stopper pierceable by a hypodermic injection needle.
[0342] Aspects of the invention includes self-buffering human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention formulations, which can be used as pharmaceutical compositions, as described in international patent application WO 06138181A2 (PCT/US2006/022599), which is incorporated by reference in its entirety herein.
[0343] As discussed above, certain embodiments provide human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention protein compositions, particularly pharmaceutical compositions of the invention, that comprise, in addition to the human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention, one or more excipients such as those illustratively described in this section and elsewhere herein. Excipients can be used in the invention in this regard for a wide variety of purposes, such as adjusting physical, chemical, or biological properties of formulations, such as adjustment of viscosity, and or processes of the invention to improve effectiveness and or to stabilize such formulations and processes against degradation and spoilage due to, for instance, stresses that occur during manufacturing, shipping, storage, pre-use preparation, administration, and thereafter.
[0344] A variety of expositions are available on protein stabilization and formulation materials and methods useful in this regard, such as Arakawa et al., "Solvent interactions in pharmaceutical formulations," Pharm Res. 8(3): 285-91 (1991); Kendrick et al., "Physical stabilization of proteins in aqueous solution," in: RATIONAL DESIGN OF STABLE PROTEIN FORMULATIONS: THEORY AND PRACTICE, Carpenter and Manning, eds. Pharmaceutical Biotechnology. 13: 61-84 (2002), and Randolph et al., "Surfactant-protein interactions," Pharm Biotechnol. 13: 159-75 (2002), each of which is herein incorporated by reference in its entirety, particularly in parts pertinent to excipients and processes of the same for self-buffering protein formulations in accordance with the current invention, especially as to protein pharmaceutical products and processes for veterinary and/or human medical uses.
[0345] Salts may be used in accordance with certain embodiments of the invention to, for example, adjust the ionic strength and/or the isotonicity of a formulation and/or to improve the solubility and/or physical stability of a protein or other ingredient of a composition in accordance with the invention.
[0346] As is well known, ions can stabilize the native state of proteins by binding to charged residues on the protein's surface and by shielding charged and polar groups in the protein and reducing the strength of their electrostatic interactions, attractive, and repulsive interactions. Ions also can stabilize the denatured state of a protein by binding to, in particular, the denatured peptide linkages (--CONH) of the protein. Furthermore, ionic interaction with charged and polar groups in a protein also can reduce intermolecular electrostatic interactions and, thereby, prevent or reduce protein aggregation and insolubility.
[0347] Ionic species differ significantly in their effects on proteins. A number of categorical rankings of ions and their effects on proteins have been developed that can be used in formulating pharmaceutical compositions in accordance with the invention. One example is the Hofmeister series, which ranks ionic and polar non-ionic solutes by their effect on the conformational stability of proteins in solution. Stabilizing solutes are referred to as "kosmotropic." Destabilizing solutes are referred to as "chaotropic." Kosmotropes commonly are used at high concentrations (e.g., >1 molar ammonium sulfate) to precipitate proteins from solution ("salting-out"). Chaotropes commonly are used to denture and/or to solubilize proteins ("salting-in"). The relative effectiveness of ions to "salt-in" and "salt-out" defines their position in the Hofmeister series.
[0348] Free amino acids can be used in human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention formulations in accordance with various embodiments of the invention as bulking agents, stabilizers, and antioxidants, as well as other standard uses. Lysine, proline, serine, and alanine can be used for stabilizing proteins in a formulation. Glycine is useful in lyophilization to ensure correct cake structure and properties. Arginine may be useful to inhibit protein aggregation, in both liquid and lyophilized formulations. Methionine is useful as an antioxidant.
[0349] Polyols include sugars, e.g., mannitol, sucrose, and sorbitol and polyhydric alcohols such as, for instance, glycerol and propylene glycol, and, for purposes of discussion herein, polyethylene glycol (PEG) and related substances. Polyols are kosmotropic. They are useful stabilizing agents in both liquid and lyophilized formulations to protect proteins from physical and chemical degradation processes. Polyols also are useful for adjusting the tonicity of formulations.
[0350] Among polyols useful in select embodiments of the invention is mannitol, commonly used to ensure structural stability of the cake in lyophilized formulations. It ensures structural stability to the cake. It is generally used with a lyoprotectant, e.g., sucrose. Sorbitol and sucrose are among preferred agents for adjusting tonicity and as stabilizers to protect against freeze-thaw stresses during transport or the preparation of bulks during the manufacturing process. Reducing sugars (which contain free aldehyde or ketone groups), such as glucose and lactose, can glycate surface lysine and arginine residues. Therefore, they generally are not among preferred polyols for use in accordance with the invention. In addition, sugars that form such reactive species, such as sucrose, which is hydrolyzed to fructose and glucose under acidic conditions, and consequently engenders glycation, also is not among preferred polyols of the invention in this regard. PEG is useful to stabilize proteins and as a cryoprotectant and can be used in the invention in this regard.
[0351] Embodiments of the human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention formulations further comprise surfactants. Protein molecules may be susceptible to adsorption on surfaces and to denaturation and consequent aggregation at air-liquid, solid-liquid, and liquid-liquid interfaces. These effects generally scale inversely with protein concentration. These deleterious interactions generally scale inversely with protein concentration and typically are exacerbated by physical agitation, such as that generated during the shipping and handling of a product.
[0352] Surfactants routinely are used to prevent, minimize, or reduce surface adsorption. Useful surfactants in the invention in this regard include polysorbate 20, polysorbate 80, other fatty acid esters of sorbitan polyethoxylates, and poloxamer 188.
[0353] Surfactants also are commonly used to control protein conformational stability. The use of surfactants in this regard is protein-specific since, any given surfactant typically will stabilize some proteins and destabilize others.
[0354] Polysorbates are susceptible to oxidative degradation and often, as supplied, contain sufficient quantities of peroxides to cause oxidation of protein residue side-chains, especially methionine. Consequently, polysorbates should be used carefully, and when used, should be employed at their lowest effective concentration. In this regard, polysorbates exemplify the general rule that excipients should be used in their lowest effective concentrations.
[0355] Embodiments of human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention formulations further comprise one or more antioxidants. To some extent deleterious oxidation of proteins can be prevented in pharmaceutical formulations by maintaining proper levels of ambient oxygen and temperature and by avoiding exposure to light. Antioxidant excipients can be used as well to prevent oxidative degradation of proteins. Among useful antioxidants in this regard are reducing agents, oxygen/free-radical scavengers, and chelating agents. Antioxidants for use in therapeutic protein formulations in accordance with the invention preferably are water-soluble and maintain their activity throughout the shelf life of a product. EDTA is a preferred antioxidant in accordance with the invention in this regard.
[0356] Antioxidants can damage proteins. For instance, reducing agents, such as glutathione in particular, can disrupt intramolecular disulfide linkages. Thus, antioxidants for use in the invention are selected to, among other things, eliminate or sufficiently reduce the possibility of themselves damaging proteins in the formulation.
[0357] Formulations in accordance with the invention may include metal ions that are protein co-factors and that are necessary to form protein coordination complexes, such as zinc necessary to form certain insulin suspensions. Metal ions also can inhibit some processes that degrade proteins. However, metal ions also catalyze physical and chemical processes that degrade proteins.
[0358] Magnesium ions (10-120 mM) can be used to inhibit isomerization of aspartic acid to isoaspartic acid. Ca+2 ions (up to 100 mM) can increase the stability of human deoxyribonuclease. Mg+2, Mn+2, and Zn+2, however, can destabilize rhDNase. Similarly, Ca+2 and Sr+2 can stabilize Factor VIII, it can be destabilized by Mg+2, Mn+2 and Zn+2, Cu+2 and Fe+2, and its aggregation can be increased by Al+3 ions.
[0359] Embodiments of the human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention formulations further comprise one or more preservatives. Preservatives are necessary when developing multi-dose parenteral formulations that involve more than one extraction from the same container. Their primary function is to inhibit microbial growth and ensure product sterility throughout the shelf-life or term of use of the drug product. Commonly used preservatives include benzyl alcohol, phenol and m-cresol. Although preservatives have a long history of use with small-molecule parenterals, the development of protein formulations that includes preservatives can be challenging. Preservatives almost always have a destabilizing effect (aggregation) on proteins, and this has become a major factor in limiting their use in multi-dose protein formulations. To date, most protein drugs have been formulated for single-use only. However, when multi-dose formulations are possible, they have the added advantage of enabling patient convenience, and increased marketability. A good example is that of human growth hormone (hGH) where the development of preserved formulations has led to commercialization of more convenient, multi-use injection pen presentations. At least four such pen devices containing preserved formulations of hGH are currently available on the market. Norditropin (liquid, Novo Nordisk), Nutropin AQ (liquid, Genentech) & Genotropin (lyophilized--dual chamber cartridge, Pharmacia & Upjohn) contain phenol while Somatrope (Eli Lilly) is formulated with m-cresol. Several aspects need to be considered during the formulation and development of preserved dosage forms. The effective preservative concentration in the drug product must be optimized. This requires testing a given preservative in the dosage form with concentration ranges that confer anti-microbial effectiveness without compromising protein stability.
[0360] As might be expected, development of liquid formulations containing preservatives are more challenging than lyophilized formulations. Freeze-dried products can be lyophilized without the preservative and reconstituted with a preservative containing diluent at the time of use. This shortens the time for which a preservative is in contact with the protein, significantly minimizing the associated stability risks. With liquid formulations, preservative effectiveness and stability should be maintained over the entire product shelf-life (about 18 to 24 months). An important point to note is that preservative effectiveness should be demonstrated in the final formulation containing the active drug and all excipient components.
[0361] Human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention generally will be designed for specific routes and methods of administration, for specific administration dosages and frequencies of administration, for specific treatments of specific diseases, with ranges of bio-availability and persistence, among other things. Formulations thus may be designed in accordance with the invention for delivery by any suitable route, including but not limited to orally, aurally, opthalmically, rectally, and vaginally, and by parenteral routes, including intravenous and intraarterial injection, intramuscular injection, and subcutaneous injection.
[0362] Once the pharmaceutical composition has been formulated, it may be stored in sterile vials as a solution, suspension, gel, emulsion, solid, crystal, or as a dehydrated or lyophilized powder. Such formulations may be stored either in a ready-to-use form or in a form (e.g., lyophilized) that is reconstituted prior to administration. The invention also provides kits for producing a single-dose administration unit. The kits of the invention may each contain both a first container having a dried protein and a second container having an aqueous formulation. In certain embodiments of this invention, kits containing single and multi-chambered pre-filled syringes (e.g., liquid syringes and lyosyringes) are provided. The therapeutically effective amount of a human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention protein-containing pharmaceutical composition to be employed will depend, for example, upon the therapeutic context and objectives. One skilled in the art will appreciate that the appropriate dosage levels for treatment will vary depending, in part, upon the molecule delivered, the indication for which the human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention is being used, the route of administration, and the size (body weight, body surface or organ size) and/or condition (the age and general health) of the patient. In certain embodiments, the clinician may titer the dosage and modify the route of administration to obtain the optimal therapeutic effect. A typical dosage may range from about 0.1 .mu.g/kg to up to about 30 mg/kg or more, depending on the factors mentioned above. In specific embodiments, the dosage may range from 1.0 .mu.g/kg up to about 20 mg/kg, optionally from 10 .mu.g/kg up to about 10 mg/kg or from 100 .mu.g/kg up to about 5 mg/kg.
[0363] A therapeutic effective amount of a human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention preferably results in a decrease in severity of disease symptoms, in increase in frequency or duration of disease symptom-free periods or a prevention of impairment or disability due to the disease affliction. For treating CDH19-expressing tumors, a therapeutically effective amount of human antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention, e.g. an anti-CDH19 antibody construct (ADC construct), preferably inhibits cell growth or tumor growth by at least about 20%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, or at least about 90% relative to untreated patients. The ability of a compound to inhibit tumor growth may be evaluated in an animal model predictive of efficacy in human tumors.
[0364] Pharmaceutical compositions may be administered using a medical device. Examples of medical devices for administering pharmaceutical compositions are described in U.S. Pat. Nos. 4,475,196; 4,439,196; 4,447,224; 4,447, 233; 4,486,194; 4,487,603; 4,596,556; 4,790,824; 4,941,880; 5,064,413; 5,312,335; 5,312,335; 5,383,851; and 5,399,163, all incorporated by reference herein.
[0365] In one embodiment the invention provides the human antibody or antigen binding fragment thereof of the invention, the antibody construct of the invention, or produced according to the process of the invention for use in the prevention, treatment or amelioration of a melanoma disease or metastatic melanoma disease. Preferably, the melanoma disease or metastatic melanoma disease is selected from the group consisting of superficial spreading melanoma, lentigo maligna, lentigo maligna melanoma, acral lentiginous melanoma and nodular melanoma.
[0366] The invention also provides a method for the treatment or amelioration of a melanoma disease or metastatic melanoma disease, comprising the step of administering to a subject in need thereof the antibody or antigen binding fragment thereof of the invention, the antibody construct of the invention, an antibody or antigen binding fragment thereof of the invention or the antibody construct of the invention produced according to the process of the invention or a pharmaceutical composition of the invention.
[0367] In a preferred embodiment method the invention the melanoma disease or metastatic melanoma disease is selected from the group consisting of superficial spreading melanoma, lentigo maligna, lentigo maligna melanoma, acral lentiginous melanoma and nodular melanoma.
[0368] In a further embodiment, the invention provides a kit comprising an antibody or antigen binding fragment thereof of the invention, an antibody construct of the invention, an antibody or antigen binding fragment thereof of the invention or the antibody construct produced according to the process of the invention, a vector of the invention, and/or a host cell of the invention.
[0369] It should be understood that the inventions herein are not limited to particular methodology, protocols, or reagents, as such can vary. The discussion and examples provided herein are presented for the purpose of describing particular embodiments only and are not intended to limit the scope of the present invention, which is defined solely by the claims.
[0370] All publications and patents cited throughout the text of this specification (including all patents, patent applications, scientific publications, manufacturer's specifications, instructions, etc.), whether supra or infra, are hereby incorporated by reference in their entirety. Nothing herein is to be construed as an admission that the invention is not entitled to antedate such disclosure by virtue of prior invention. To the extent the material incorporated by reference contradicts or is inconsistent with this specification, the specification will supersede any such material.
EXAMPLES
[0371] The following examples illustrate the invention. These examples should not be construed as to limit the scope of this invention. The examples are included for purposes of illustration, and the present invention is limited only by the claims.
Example 1--Fully Human Monoclonal Antibodies Against CDH19
[0372] 1.1 Immunization:
[0373] Fully human antibodies to Cadherin-19 (CDH19) were generated using XENOMOUSE.RTM. technology, transgenic mice engineered to express diverse repertoires of fully human IgG.kappa. and IgG.lamda. antibodies of the corresponding isotype. (U.S. Pat. Nos. 6,114,598; 6,162,963; 6,833,268; 7,049,426; 7,064,244, which are incorporated herein by reference in their entirety; Green et al., 1994, Nature Genetics 7:13-21; Mendez et al., 1997, Nature Genetics 15:146-156; Green and Jakobovitis, 1998, J. Ex. Med. 188:483-495; Kellermann and Green, Current Opinion in Biotechnology 13, 593-597, 2002).
[0374] Mice were immunized with multiple forms of Cadherin-19 immunogen, including: (1) full length human and cynomologous ("cyno") monkey cadherin-19, (2) secreted Cadherin-19 ecto-domain (amino acids 1-596), and (3) a truncated membrane bound form of human cadherin-19 (amino acids 1-624). Mice were immunized over a period of 8 to 10 weeks with a range of 16-18 boosts.
[0375] Sera were collected at approximately 5 and 9 weeks after the first injection and specific titers were determined by FACs staining of recombinant Cadherin-19 receptor transiently expressed on CHO--S cells. A total of 37 animals were identified with specific immune responses, these animals were pooled into 3 groups and advanced to antibody generation.
[0376] 1.2 Preparation of Monoclonal Antibodies
[0377] Animals exhibiting suitable titers were identified, and lymphocytes were obtained from draining lymph nodes and, if necessary, pooled for each cohort. Lymphocytes were dissociated from lymphoid tissue by grinding in a suitable medium (for example, Dulbecco's Modified Eagle Medium (DMEM); obtainable from Invitrogen, Carlsbad, Calif.) to release the cells from the tissues, and suspended in DMEM. B cells were selected and/or expanded using standard methods, and fused with suitable fusion partner using techniques that were known in the art.
[0378] After several days of culture, the hybridoma supernatants were collected and subjected to screening assays as detailed in the examples below, including confirmation of binding to human and cynomologous monkey as well as the ability to kill cell lines in secondary antibody-drug conjugate Bioassays. Hybridoma lines that were identified to have the binding and functional properties of interest were then further selected and subjected to standard cloning and subcloning techniques. Clonal lines were expanded in vitro, and the secreted human antibodies obtained for analysis and V gene sequencing was performed.
[0379] 1.3 Selection of Cadherin-19 Receptor Specific Binding Antibodies by FMAT
[0380] After 14 days of culture, hybridoma supernatants were screened for CDH19-specific monoclonal antibodies by Fluorometric Microvolume Assay Technology (FMAT) (Applied Biosystems, Foster City, Calif.). The supernatants were screened against adherent CHO cells transiently transfected with human Cadherin-19 and counter screened against CHO cells transiently transfected with the same expression plasmid that did not contain the Cadherin-19 gene.
[0381] After multiple screening campaigns, a panel of 1570 anti-Cadherin-19 binding hybridoma lines were identified and advanced to further characterization assays.
Example 2--Assessment of Fully Human Monoclonal Antibodies Against CDH19
[0382] 2.1 Additional Binding Characterization by Flow Cytometry (FACs)
[0383] FACS binding assays were performed to evaluate the binding of the anti-Cadherin-19 receptor specific antibodies to endogenous Cadherin-19 receptor expressed on the CHL-1 tumor cell lines. In addition, cross-reactive binding to murine and cynomologous monkey Cadherin-19 orthologues was also evaluated by FACs using recombinant forms of the various receptors transiently expressed on 293T cells.
[0384] FACs assays were performed by incubating hybridoma supernatants with 10,000 to 25,000 cells in PBS/2% Fetal bovine serum/2 mM Calcium Chloride at 4.degree. C. for one hour followed by two washes with PBS/2% Fetal bovine serum/2 mM Calcium Chloride. Cells were then treated with florochrome-labeled secondary antibodies at 4.degree. C. followed by one wash. The cells were resuspended in 50 .mu.l of PBS/2% FBS and antibody binding was analyzed using a FACSCalibur.TM. instrument.
[0385] 2.2 Antibody Drug Conjugate Screening of Fully Human Antibodies Derived from XenoMouse.RTM. Hybridomas
[0386] Cell killing through antibody drug conjugates requires the delivery of the conjugate into a cell through internalization and the catabolism of the drug-conjugate into a form that it is toxic to the cell. To identify antibodies with these properties, CDH19-positive cell lines (Colo-699 or CHL-1) were seeded at low cell densities and allowed to adhere overnight in a 384 well plate. XENOMOUSE.RTM. hybridoma samples containing fully human anti-CDH19 antibodies were then added to these cells in the presence of a high concentration of a goat anti-human Fc monovalent Fab conjugated with DM1 (DM1-Fab) at a relatively low drug-antibody ratio (DAR) (.about.1.3). The cells were incubated for 96 hours at 37.degree. C. and 5% CO.sub.2 in the presence of the antibody samples and the DM1-Fab. At the end of this time, the cell viability was assessed using the CellTiter-Glo.RTM. Luminescent Cell Viability reagent (Promega) according to manufacturer's recommendations.
[0387] An example of the cell viability data with the Colo-699 cells is shown in FIG. 1 and FIG. 2. The antibodies capable of delivering the DM1-Fab to the cells and inhibiting the cell growth read out with a lower luminescent signal (RLU). The top antibodies of interest from this screen are observed in the lower left corner of FIG. 1 and are denoted as open circles.
[0388] These antibodies were taken forward into a cell viability assay on CHL-1 cells. The average cell viability data from the CHL-1 assay is plotted against the average cell viability data from the Colo-699 assay (FIG. 2). The antibodies that had activity on both the Colo-699 and the CHL-1 cells are denoted as open circles on the left-hand side of the FIG. 2.
[0389] This assay was run concurrently with the FACs antibody binding assay above (2.2), and the results from these two studies were used to select the antibodies for further characterization. In total, 1570 antibodies were run through these cell based viability assays and approximately 44 antibodies were selected on the bases of in vitro cell killing and/or antibody binding for sub-cloning, V gene sequencing and expressed in recombinant form for further characterization assays as described below.
[0390] These 44 antibodies were again assayed as in Example 2 and 19 antibodies were selected that contained unique sequences. Of these 19 antibodies, 18 antibodies were analyzed and their properties characterized in Table 2 below. The data in this table was generated using FACs binding on recombinant human and cynomologous CDH-19, +/-Calcium (Ca.sup.+2) binding data on 293/CDH-19 transfectants, binding to endogenous CDH-19 on CHL-1 and Colo699 tumor cells and competition with the antibody designated as 4A9 in the table. These experiments provided the further characterizations for the grouping of these antibodies into 5 groups or bins.
TABLE-US-00002 TABLE 2 Binning of Lead panel using Antibody Binding Information LMR Bin Sequence/ Clone ID Ab ID ID Bin Characteristics 1 13589 4A9 High Endogenous binding, Calcium 13591 4F7 insensitive, sequence clustered, moderate cyno complete 4A9 competitor 2 13885 19B5 High Endogenous binding, Calcium 13880 25F8 insensitive, sequence 13882 26D1 clustered, Good cyno, 13881 26F12 = partial 4A9 competitor 27B3 13878 16H2 = 20D3 = 23E7 13879 22D1 3 13877 22G10 High Endogenous binding, moderate 13874 17H8 = 293 binding, Calcium insensitive, 23B6 = 2 sequence clusters, moderate 28D10 cyno, partial 4A9 competitor, 13883 25G10 22G10 best binder in bin. 13875 16C1 4 13590 4610 Low Endogenous and 13586 4F3 recombinant binding, 13592 4A2 Calcium sensitive, sequence 13884 23A10 diverse group, comparable 13588 2G6 cyno, No 4A9 competition 5 13876 16A4 Best endogenous binder, moderate recombinant binder, calcium insensitive, very weak cyno, No 4A9 competition.
[0391] Of these 18 antibodies. 8 antibodies were selected for further analysis of their epitope binding as described below. At least one representative antibody from each bin was selected for further analysis.
Example 3--Epitope Prediction
[0392] Epitope Prediction by 4A9 Antibody Competition and by Human/Mouse Cadherin-19 Chimeras
[0393] A 4A9 binding competition method was developed to identify antibodies that compete with 4A9 binding. In 96-well V-bottom plates (Sarstedt #82.1583.001), 50,000 transiently transfected 293T cells were incubated with 5 ug/ml of purified anti-CDH19 antibodies for 1 hr at 4.degree. C. followed by one wash with PBS/2% FBS. 25 .mu.l of 5 .mu.g/ml Alexa647-labelled 4A9 was then added to each well and the plates incubated for 1 hour at 4.degree. C. Cells were then washed two times and the amount of cell associated Alexa647-labelled 4A9 was quantitated by flow cytometry.
[0394] The experiments included negative controls consisting of PBS/2% FBS only. The average signal observed in these negative control experiments was adopted as the maximum possible signal for the assay. Antibodies were compared to this maximum signal and a percent inhibition was calculated for each well (% Inhibition=(1-(FL4 Geomean with the anti-CDH19 antibodies/Maximum FL4 Geomean signal)).
[0395] Domain binding was determined by flow cytometry as above on 293T cells transiently transfected with plasmids consisting of single or dual human CDH19 cadherin repeat domain replacements into the mouse Cadherin19 backbone cloned into the pTT5 expression vector immediately preceded by native human or murine CDH19 leader sequences and a Flag tag (SEQ ID NO: 968). The experiment included assaying the anti-CDH19 antibodies against mouse Cadherin19 to determine suitability for binning on these human/mouse chimeras.
[0396] The data from these experiments are presented in the Table below entitled as follows:
TABLE-US-00003 TABLE 3 Calcium Sensitive Binding and Epitope Prediction Summary Com- petes Hu Hu Hu Hu Mu Ca2+ with EC1- Hu EC1- Hu EC2- Hu EC4- Hu EC1- Predicted Clone Ab Sensitive 4A9 5 EC1 2 EC2 3 EC3 5 EC5 5 Epitope ID ID Bin Binding (13589) A B C D E F G H I Region 4A9 13589 1 No Yes + + + - - - - - - 44-141 14056 1 No Yes + + + - - - - - - 14057 1 No Yes + + + - - - - - - 25F8 13880 2 No Yes + + + - - - - - - 14094 2 No Yes + + + - - - - - - 14096 2 No Yes + + + - - - - - - 26D1 13882 2 No Yes + + + - - - - - - 14088 2 No Yes + + + - - - - - - 17H8 13874 3 No Yes + + + - - - - - - 14045 3 No Yes + + + - - - - - - 14048 3 No Yes + + + - - - - - - 4A2 13592 4 Yes No + - - - + + - - - 250-364 14026 4 Yes No + - - - + + - - - 4610 13590 4 Yes No + - - - + + - - - 14055 4 Yes No + - - - + + - - - 14054 4 Yes No + - - - + + - - - 2G6 13588 4 Yes No + + + + + + + + + un- 14304 4 Yes No + + + + + + + + + assignable 14039 4 Yes No + + + + + + + + + 16A4 13876 5 No No + + + - - - - - - Unassigned 14071 5 No No + + + - - - - - - complex epitope Rat anti-FLAG + + + + + + + + + Legend Table 3 Human and/or murine chimera constructs A = huCDH19(44-772) (see SEQ ID NO: 944) B = huCDH19(44-141)::muCDH19(140-770) (see SEQ ID NO: 952) C = huCDH19(44-249)::muCDH19(248-770) (see SEQ ID NO: 954) D = muCDH19(44-139)::huCDH19(142-249)::muCDH19(248-770) (see SEQ ID NO: 956) E = muCDH19(44-139)::huCDH19(142-364)::muCDH19(363-770) (see SEQ ID NO: 958) F = muCDH19(44-247)::huCDH19(250-364)::muCDH19(363-770) (see SEQ ID NO: 960) G = muCDH19(44-362)::huCDH19(365-772) (see SEQ ID NO: 962) H = muCDH19(44-461)::huCDH19(464-772) (see SEQ ID NO: 964) I = muCDH19(44-770) (see SEQ ID NO: 966)
[0397] Epitope Prediction by Human/Chicken Cadherin-19 Chimeras
[0398] Domain binding was determined by flow cytometry on 293T cells transiently transfected with plasmids consisting of single human CDH19 cadherin repeat domain replacements into the chicken Cadherin19 backbone cloned into the pTT5 expression vector immediately preceded by native human or chicken CDH19 leader sequences and a Flag tag. The experiment included assaying a subset of anti-CDH19 antibodies against chicken Cadherin19 to determine suitability for binning on these human/chicken chimeras.
[0399] The following binding assay was completed in presence of 2 mM CaCl.sub.2). In 96-well V-bottom plates (Costar 3897), 50,000 transiently transfected 293T cells were incubated with 5 ug/ml of purified anti-CDH19 antibodies for 1 hr at 4.degree. C. followed by two washes with PBS/2% FBS. 50 .mu.l of 5 .mu.g/ml Alexa647-labelled anti-human IgG secondary antibody (Jackson Immuno 109-605-098) and 2 ug/ml 7AAD (Sigma A9400) was then added to each well and the plates incubated for 15 minutes at 4.degree. C. Cells were then washed one time and the amount of cell associated Alexa647-labelled Ab was quantitated by flow cytometry. The experiments included mock transfected controls. The data from these experiments are presented in the Table below, n.d.=not determined.
TABLE-US-00004 TABLE 4 Antibody Bin C Epitope Prediction Summary Hu Ck Pre- EC1- Ed1- Hu Hu Hu Hu dicted Clone Ab. 5 5 EC1 EC2 EC3 EC5 Epitope ID ID Bin A J K L M O Region 4A9 13589 1 + - + - - - 44-141 26F12 13881 2 + - + - - - Bin A 25F8 14096 2 + - + - - - 26D1 13882 2 + - + - - - 17H8 13874 3 + - + - - - 16A4 14071 5 + - + - - - 4A2 13592 4 + - - - + - 250- 4B10 13590 4 + - - - + - 364 2G6 13588 4 + - - - + - Bin B 23A10 14077 4 + - - - + - Rat anti-FLAG + + + + + + control + Positive Binding - Negative Binding Legend Table 4 Human and/or chicken chimera constructs A = huCDH19(44-772) (see SEQ ID NO: 944) J = ckCDH19(44-776) (see SEQ ID NO: 970) K = huCDH19(44-141)::ckCDH19(142-776) (see SEQ ID NO: 971) L = ckCDH19(44-141)::huCDH19(142-249)::ckCDH19(250-776) (see SEQ ID NO: 972) M = ckCDH19(44-249)::huCDH19(250-364)::ckCDH19(365-776) (see SEQ ID NO: 973) N = ckCDH19(44-364)::huCDH19(365-463)::ckCDH19(469-776) (see SEQ ID NO: 974) O = ckCDH19(44-468)::huCDH19(464-772) (see SEQ ID NO: 975)
[0400] Epitope Prediction by Macaque/Dog or Rat/Macaque Cadherin-19 Chimeras
[0401] Domain binding was determined by flow cytometry on 293T cells transiently transfected with plasmids consisting of rhesus macaque CDH19 cadherin repeat domain 1 or segments domain 1 (designated EC1a, EC1b, EC1c) replacements into the dog Cadherin19 backbone, or rat CDH19 cadherin repeat domain 2 replacement into the rhesus Cadherin19 backbone cloned into the pTT5 expression vector immediately preceded by native rhesus or canine CDH19 leader sequences and a Flag tag. The experiment included assaying a subset of anti-CDH19 antibodies against dog, rat and macaque Cadherin19 to determine suitability for binning on these macaque/dog and rat/rhesus chimeras.
[0402] The following binding assay was completed in presence of 2 mM CaCl.sub.2). In 96-well V-bottom plates (Costar 3897), 50,000 transiently transfected 293T cells were incubated with 5 ug/ml of purified anti-CDH19 antibodies for 1 hr at 4.degree. C. followed by two washes with PBS/2% FBS. 50 .mu.l of 5 .mu.g/ml Alexa647-labelled anti-human IgG secondary antibody (Jackson Immuno 109-605-098) and 2 ug/ml 7AAD (Sigma A9400) was then added to each well and the plates incubated for 15 minutes at 4.degree. C. Cells were then washed one time and the amount of cell associated Alexa647-labelled Ab was quantitated by flow cytometry. The experiments included mock transfected controls. The data from these experiments are presented in the Table below, n.d.=not determined.
TABLE-US-00005 TABLE 5 Antibody BinA Epitope prediction Summary Rh Ca rh rh rh ra Ra Predicted EC1-5 EC1-5 EC1 EC1a EC1b EC2 EC1-5 Epitope Clone ID Ab. ID Bin P Q R S T V W Region 4A9 13589 1 + - + - - - - 44-141 + + + Bin A.1 26F12 13881 2 + - + + + - - 44-141 25F8 14096 2 + - + + + - - Bin A.2 26D1 13882 2 + - + + + - - (44- 114) 17H8 13874 3 + - + + - - - 44-141 Bin A.3 16A4 14071 5 + - + + - n.d. + (44-65) 4A2 13592 4 + - n.d. n.d. n.d. n.d. + 250- 4B10 13590 4 + + n.d. n.d. n.d. n.d. + 364 2G6 13588 4 + + n.d. n.d. n.d. n.d. + Bin B 23A10 14077 4 + + n.d. n.d. n.d. n.d. + Rat anti-FLAG + + + + + + + + Positive Binding - Negative Binding (n.d.) Not Determined Legend Table 5 Rhesus macaque, dog, and/or rat chimera constructs P = rhCDH19(44-772) (see SEQ ID NO: 976) Q = caCDH19(44-770) (see SEQ ID NO: 977) R = rhCDH19(44-141)::caCDH19(141-770) (see SEQ ID NO: 978) S = rhCDH19(44-65)::caCDH19(65-770) (see SEQ ID NO: 979) T = caCDH19(44-87)::rhCDH19(89-114)::caCDH19(115-770) (see SEQ ID NO: 980) U = caCDH19(44-120)::rhCDH19(122-137)::caCDH19(137-770) (see SEQ ID NO: 981) V = rhCDH19(44-141)::raCDH19(140-247)::rhCDH19(250-772) (see SEQ ID NO: 982) W = raCDH19(44-770) (see SEQ ID NO: 983)
[0403] The data summarized in table 5 allowed for segregating the binder of Bin A 44-141 into the following subgroups:
[0404] Bin A.1 44-141
[0405] Bin A.2 44-141 (44-114)
[0406] Bin A.3 44-141 (44-65)
[0407] Epitope Prediction by Rat/Mouse or Human/Mouse Cadherin-19 Chimeras
[0408] Domain binding was determined by flow cytometry on 293T cells transiently transfected with plasmids consisting of rat CDH19 cadherin repeat domain 3 substitutions (designated EC3a, EC3b) or human CDH19 cadherin repeat domain 3 substitution (designated EC3c) into the mouse Cadherin19 backbone cloned into the pTT5 expression vector immediately preceded by native mouse CDH19 leader sequence and a Flag tag. The experiment included assaying a subset of anti-CDH19 antibodies against human, rat and mouse Cadherin19 to determine suitability for binning on these rat/mouse and human/mouse chimeras.
[0409] The following binding assay was completed in presence of 2 mM CaCl.sub.2). In 96-well V-bottom plates (Costar 3897), 50,000 transiently transfected 293T cells were incubated with 5 ug/ml of purified anti-CDH19 antibodies for 1 hr at 4.degree. C. followed by two washes with PBS/2% FBS. 50 .mu.l of 5 .mu.g/ml Alexa647-labelled anti-human IgG secondary antibody (Jackson Immuno 109-605-098) and 2 ug/ml 7AAD (Sigma A9400) was then added to each well and the plates incubated for 15 minutes at 4.degree. C. Cells were then washed one time and the amount of cell associated Alexa647-labelled Ab was quantitated by flow cytometry. The experiments included mock transfected controls. The data from these experiments are presented in the Table below, n.d.=not determined.
TABLE-US-00006 TABLE 6 Antibody Bin B Epitope Prediction Summary Hu Mo Ra Pre- EC1- EC1- EC1- Ra Ra Hu dicted Clone Ab. 5 5 5 EC3c EC3b EC3a Epitope ID ID Bin A I W X Y Z Region 4A9 13589 1 + - - n.d. n.d. n.d. 44-141 26F12 13881 2 + - - n.d. n.d. n.d. Bin A 25F8 14096 2 + - - n.d. n.d. n.d. 26D1 13882 2 + - - n.d. n.d. n.d. 17H8 13874 3 + - - n.d. n.d. n.d. 16A4 14071 5 + - + n.d. n.d. n.d. 4A2 13592 4 + - + + - - 250- 364 4B10 13590 4 + - + + - - (324- 327) Bin B.2 2G6 13588 4 + + + + + + 250-364 23A10 14077 4 + + + n.d. n.d. n.d. Bin B.1 Rat anti- + + + + + + control FLAG + Positive Binding - Negative Binding (n.d.) Not Determined Legend Table 6 Rat/mouse or human/mouse chimera constructs A = huCDH19(44-772) (see SEQ ID NO: 944) I = muCDH19(44-770) (see SEQ ID NO: 966) W = raCDH19(44-770) (see SEQ ID NO: 983) X = muCDH19(44-323)::raCDH19(324-327)::muCDH19(328-770) (see SEQ ID NO: 984) Y = muCDH19(44-770)::raCDH19(290, 299, 308) (see SEQ ID NO: 985) Z = muCDH19(44-770)::huCDH19(271) (see SEQ ID NO: 986)
[0410] The data summarized in table 4 allowed for segregating the binder of Bin B 250-364 into the following subgroups:
[0411] Bin B.1 250-364
[0412] Bin B.2 250-364 (324-327) by rodent numeration as referenced in table 6, corresponding to residues (326-329) within human and macaque CDH19.
Example 4--Hotspot/Covariant Mutants
[0413] A total of 18 antibodies were analyzed for potential hotspots and covariance violations. The designed variants (shown below) outline amino acid substitutions capable of reducing and/or avoiding isomerization, deamidation, oxidation, covariance violations, and the like. The 80 engineered variants together with the 15 parental antibodies, thus totaling 95 sequences, were taken forward to the cloning, expression, and purification processes. Site-directed mutagenesis was performed on the engineered variants in a 96-well format. The parental antibodies and engineered variants were expressed by high throughput transient transfection in HEK 293-6E cells, purified using a modified AKTA auto-sampler and assayed for activity and biophysical characteristics. The 3 parental antibodies that had either free (unpaired) Cys or N-glycosylation site were not taken forward in this process. Those were replaced with the engineered version of the parental antibodies. The designed variants outline amino acid substitutions capable of reducing and/or avoiding isomerization, deamidation, oxidation, covariance violations, immunogenicity and the like. It will be appreciated that these variant sequences are examples of engineered antibodies within the meaning of the present application but single point and/or multiple point mutations can be combined in any combinatorial manner in order to arrive at a final desired antigen binding molecule or antibody.
Example 5--CDH19 mRNA Expression Pattern
[0414] RNA was extracted from individual patient tissues representing tumor (>70% tumor content by cell count) or normal (0% tumor content by cell count). Individual tissues were homogenized using TisssueLyzer (Qiagen, Valencia, Calif.) and total RNA extracted and purified by the mirVana total RNA extraction kit (Life Technologies, Foster City, Calif.). RNA quality and quantity checked by NanoDrop (NanoDrop, Wilmington, Del.) spectrophotometer readings and Bioanalyzer RNA profiling (Agilient Technologies, Santa Clara, Calif.). RNA was DNAse treated with DNA-free kit (Life Technologies, Foster City, Calif.) and reverse transcribed according to manufacturer's specifications using random hexamers in the High Capacity cDNA Reverse Transcription Kit (Life Technologies, Foster City, Calif.). Quantitative Real Time Polymerase Chain Reaction (qRT-PCR) was performed on cDNA using primers to CDH19, probeset Hs00253534_m1, (Life Technologies, Foster City, Calif.) or the housekeeping gene human ACTB (primers CCT GGC ACC CAG CAC AA; GCC GAT CCA CAC GGA GTA CT; probe ATC AAG ATC ATT GCT CCT CCT GAG CG). 10 .mu.L qRT-PCR reaction components; 1.0 ng/.mu.L cDNA, 2.times. Universal PCR Master Mix (Life Technologies, Foster City, Calif.), gene expression assay (ACTB; 75 nM primers, 150 nM probe. EPOR; 300 nM primers, 250 nM probe) Following the qRT-PCR amplification program: (1) activation at 50.degree. C. for 2 min; (2) denaturation at 95.degree. C. for 10 min; (3) amplification 40 cycles at 95.degree. C. for 15 s and 60.degree. C. for 1 min with fluorescence capture at each step (ABI PRISM 7900HT Sequence Detection Systems, Applied Biosystems). Threshold cycle values (C.sub.T) were determined, using Sequence Detector software version 2.3 (Applied Biosystems) and transformed to 2.sup.-.DELTA.CT for relative expression of CDH19 specific transcript to ACTB. The results are shown in FIG. 3. Of 54 unique metastatic and primary melanoma samples, the majority can be seen to overexpress CDH19 mRNA relative to the expression in samples from normal tissue.
Example 6--CDH19 Protein Expression
[0415] Expression of CDH19 protein was analyzed in human tumor samples by IHC and the results are shown in FIG. 4. Samples were fixed in 10% neutral buffered formalin for 24 hours, dehydrated and paraffin embedded. 4 .mu.m sections were cut. Sections were deparaffinized first and then heated in DIVA Decloaker solution (Biocare) for 40 minutes for antigen retrieval. Remaining IHC steps were performed at room temperature in a DAKO Autostainer. Sections were incubated for 10 minutes with Peroxidazed 1 (Biocare) to block endogenous peroxidase, followed by incubation for 10 minutes with background sniper (Biocare) to reduce nonspecific background. Section were incubated for 60 minutes with CDH19 antibody (Novo Biologicals, Catalog #H00028513-B01P) at 5 .mu.g/ml, then incubated for 30 minutes with Envision+HRP anti-mouse polymer (DAKO), followed by DAB+(DAKO) for 5 minutes. Sections were counterstained with hematoxylin (DAKO) approximately for 1 minute. CDH19 expression could be detected in 62% of tumors examined (staining intensity .gtoreq.1+ in 101 of 162 samples). 51% of the tumor samples demonstrated medium to high expression (staining intensity of 2+ to 3+ in 83 of 162 samples). CDH19 showed dense and distinct membrane staining in many samples, although in some tumors heterogeneity was noted.
Example 7--Selection of Model Cell Lines
[0416] Tumor cell lines were analyzed by flow cytometry and IHC to identify model systems with CDH19 expression similar to human tumors. Human anti-huCDH19 IgG4 antibody 4A2 was purified directly from hybridoma conditioned media. For flow cytometry, 2.times.10.sup.5 cells were incubated with 200 nM of the CDH19 4A2 antibody that was conjugated to PE at a 1:1 ratio. The incubation and subsequent wash steps were performed in the presence of 1.2 mM calcium. A tube of QuantiBRITE PE lyophilized beads with four levels of PE (BD, cat #340495) was simultaneously prepared according to the manufacturer's instructions. The beads were analyzed by flow cytometry to generate a standard curve. The PE median values obtained from the melanoma lines after FACS analysis were then calibrated against the standard curve to calculate the antibodies bound per cell (ABC), which provides an estimate of the number of receptors on each cell. IHC was performed as described in Example 6 and the results are provided in FIG. 5. The melanoma cell line CHL-1 expresses about 10,000 CDH19 molecules on the cell surface, while Colo699 cells express about 5,000 receptors. Both cell lines represent tumors with medium to high expression levels based on IHC. Expression in A2058 is very low, while LOX cells do not express any detectable CDH19 protein.
Example 8--Preparation of Antibody Drug Conjugates
[0417] DNA sequences encoding the heavy chain and light chain components of anti-CDH19 antibodies were subcloned into mammalian expression vector pTT5 and transiently co-transfected into 293-6E cells, as described in published US2005/0170450 which is incorporated in its entirety by reference. Antibodies were purified from conditioned media by protein A affinity and ion exchange chromatography. Antibodies were incubated at 3 to 5 mg/ml with 4 to 13 equivalents of SMCC-DM1 in neutral to slightly basic buffered solutions containing 50 mM sodium chloride, 2 mM EDTA, and from 5 to 15% dimethylacetamide at room temperature for up to 5 hours or at 4.degree. C. for up to 18 hours. Conjugation to DM1 and DAR determination for conjugates, is described in U.S. Pat. No. 7,368,565 and related U.S. Pat. No. 7,851,432, which are herein incorporated in their entirety by reference. Resultant antibody drug conjugates (ADCs) were purified from solutes and unconjugated drug by gel permeation or ion exchange chromatography. UV spectrophotometric measurements at 252 nm and 280 nm combined with respective molar extinction coefficients of SMCC-DM1 and antibody as defined by amino acid composition were used to algebraically determine the concentration of drug (CD) and antibody (CAb) components of ADC preparations which could be used to calculate a drug to antibody ratio (DAR) as described in U.S. Pat. No. 7,368,565. DAR determinations of ADCs were more accurately made by similar algebraic calculations based on integrated peaks measured at 252 nm and 280 nm in analytical size exclusion chromatography. Orthogonal LC/MS methods were also used to qualitatively assess random drug distribution profiles by mass. The table below describes ADCs used in the experiments for which the results are provided in FIG. 6 (lots 1,2), FIG. 7 (lots 3-10), and FIG. 8 (lots 11-14), which are representative of typical ADC preparations.
TABLE-US-00007 Exam- ADC ple lot ID hu anti-huCDH19 IgG1 antibody DAR Fig. 6 1 13590 4B10 3.6 Fig. 6 2 1462 anti-SA (anti-streptavidin control) 4.5 Fig. 7 3 13590 4B10 2.5 Fig. 7 4 13590 4B10 4.1 Fig. 7 5 13590 4B10 5.1 Fig. 7 6 13590 4B10 5.8 Fig. 7 7 13590 4B10 5 Fig. 7 8 13590 4B10 6.3 Fig. 7 9 13590 4B10 7.4 Fig. 7 10 1462 anti-SA (anti-streptavidin control) 6.5 Fig. 8 11 14096 25F8.1 (K45Q,S102A,D111E) 5.6 VL + (F90Y) VH Fig. 8 12 14045 17H8.2 (G149R) VL 4.7 Fig. 8 13 14054 4B10 (H450,A90T) VL + 5.2 (R17G) VH Fig. 8 14 1462 anti-SA (anti-streptavidin control) 5.3
Example 9--Activity of CDH19 Targeting ADCs in Model Cell Lines
[0418] The CDH19 recognizing parental antibody 4B10 (Ab ID 13590) was covalently coupled to the toxin DM1 as described in Example 8. The tumor cells were plated in 384-well microtiter plates on Day 1, and on Day 2, the ADC was titrated on the cells and incubated for additional 72 h. Cell viability was determined at the end of the experiment with CellTiterGlo reagent (Promega) according to the manufacturer's instructions. Unconjugated, free DM1 served as a positive control, and a streptavidin recognizing antibody/DM1 conjugate served as a negative control to detect non specific binding. IC50s were determined with a non-linear, 4 parameter curve fit and are shown in FIG. 6.
Example 10--Effect of Drug to Antibody Ratio (DAR) on ADC Potency
[0419] In order to assess the effect of the drug antibody ratio on the potency of the ADC molecule, the CDH19 recognizing parental antibody 4B10 (Ab ID 13590) was coupled with different amounts of DM1 as indicated in FIG. 7. The effect of DARs on ADC potency was determined in cell viability assays as described in Example 9. An increased DAR leads to increases in potency for a given DM1 concentration. This effect is more pronounced on tumor cells with lower CDH19 expression.
Example 11--Efficacy of CDH19 Targeting ADCs In Vivo
[0420] Three CDH19 recognizing engineered variant antibodies (Ab IDs 14096, 14045, 14054) were coupled to DM1 and tested in xenograft experiments. CHL-1 cells were suspended in a solution of 50% serum free medium and 50% Matrigel, and implanted subcutaneously in the flank of female athymic nude mice. Each mouse received five million cells in a volume of 200 .mu.l. When tumors reached approximately 200 mm.sup.3, mice were sorted into seven groups of 10 mice each with equivalent mean and SD tumor size per group, and dosed with test agents or controls. All treatments were administered IV in a volume of 200 .mu.l. Tumors were measured two times per week using calipers. Length, width and height measurements were taken A repeated measures ANOVA with Dunnett's post-hoc test was used to compare the difference in tumor volume between each CDH19 targeting ADC and a non-specific control ADC (anti-streptavidin coupled to DM1). The percentage of tumor growth inhibition was calculated for each CDH19 targeting ADC compared to the corresponding unconjugated antibody. All three reagents demonstrate significant inhibition of tumor growth in mice as shown in FIG. 8.
Example 12--Internalization of CDH19 Following ADC Binding
[0421] Human anti-huCDH19 IgG4 antibody 4A2 was purified directly from hybridoma conditioned media and conjugated with SMCC-DM1 as described in example 8. Because the exact sequence of parental 4A2 was unknown at the time, the DAR of this IgG4 ADC was estimated to be 4.4 using a molecular weight of 150,000 Da and an extinction coefficient of 225,000 at 280 nm. CHL-1 melanoma cells were incubated with either unconjugated or DM1 conjugated CDH19 recognizing parental antibody 4A2 in complete medium at 4.degree. C. or for 2 h at 37.degree. C. After a brief wash in PBS, cells were fixed in 3% formaldehyde/PBS for 20 min. Fixed cells were washed, blocked and permeabilized in TBST/1% BSA/5% normal donkey serum/0.3% TX-100 and incubated with rabbit anti-EEA1 (CST #3288). Following another wash step, the samples were incubated with donkey anti mouse Alexa 488 and donkey anti rabbit Alexa 554. Images were taken with a 63.times. oil lens on a Zeiss LSM 510 confocal microscope. A review of the images demonstrate that both the parental and DM1 conjugated antibody detect the membrane bound CDH19 at 4.degree. C. but get quickly internalized and co-localize with endosome markers at 37.degree. C. Thus, both the unconjugated and DM1 conjugated CDH19 antibodies are internalized by melanoma cells, and the conjugation of the drug does not appear to interfere with the internalization of the CDH19 antibody.
Example 13--Efficacy of CDH19 Targeting ADCs In Vivo
[0422] 13.1: 4B10-DM1 Moderately Inhibited Tumor Growth at 182 .mu.g/kg DM1 in CHL-1 Xenografts
[0423] A study was conducted to examine the effect of the anti-CDH19 ADC 4B10-DM1 administered once per week for two weeks in CHL-1 xenografts. CHL-1 cells were suspended in a solution of 50% serum free medium and 50% Matrigel, and implanted subcutaneously in the flank of female athymic nude mice. Each mouse received five million cells in a volume of 200 .mu.l. When tumors reached approximately 150 mm3, mice were sorted into groups of 10 mice each with equivalent mean and SD tumor size per group and dosed with test agents or controls. All treatments were administered IV in a volume of 200 .mu.l. Tumors were measured two times per week using calipers (length, width and height measurement). Body weights were recorded at each measurement. A repeated measures ANOVA with Dunnett's post-hoc test was used to compare the difference in tumor volume between mice treated with 4B10-DM1 and the ADC control. The percentage of tumor growth inhibition was calculated against the ADC control. The results are shown in FIG. 9.
[0424] 13.2: Increasing the DAR Did not Increase Tumor Growth Inhibition in CHL-1 Xenografts
[0425] A study was conducted to examine the effect of drug:antibody ratio (DAR) on efficacy of the anti-CDH19 ADC 4B10-DM1 administered once per week for two weeks in CHL-1 xenografts. CHL-1 cells were suspended in a solution of 50% serum free medium and 50% Matrigel, and implanted subcutaneously in the flank of female athymic nude mice. Each mouse received five million cells in a volume of 200 .mu.l. When tumors reached approximately 200 mm3, mice were sorted into groups of 10 mice each with equivalent mean and SD tumor size per group and dosed with test agents or controls. All treatments were administered IV in a volume of 200 .mu.l. Tumors were measured two times per week using calipers (length, width and height measurement). Body weights were recorded at each measurement. A repeated measures ANOVA with Dunnett's post-hoc test was used to compare the difference in tumor volume between mice treated with 4B10-DM1 and the ADC control. The percentage of tumor growth inhibition was calculated against the ADC control. The results are shown in FIG. 10.
[0426] 13.3: Anti-CDH19 ADCs Moderately Inhibited Tumor Growth in COLO699 Xenografts
[0427] A study was conducted to examine the effects of anti-CDH19 ADC 4B10-DM1 and an optimized variant administered once per week for two weeks on COLO699 xenografts. COLO699 cells were suspended in a solution of 50% serum free medium and 50% Matrigel, and implanted subcutaneously in the flank of female athymic nude mice. Each mouse received five million cells in a volume of 200 .mu.l. When tumors reached approximately 200 mm3, mice were sorted into groups of 10 mice each with equivalent mean and SD tumor size per group, and dosed with test agents or controls. All treatments were administered IV in a volume of 200 .mu.l. Tumors were measured two times per week using calipers (length, width and height measurement). Body weights were recorded at each measurement. A repeated measures ANOVA with Dunnett's post-hoc test was used to compare the difference in tumor volume between mice treated with 4B10-DM1 and the ADC control. The percentage of tumor growth inhibition was calculated against the ADC control. A similar study was conducted as described above (data not shown) that resulted in the same trends for tumor growth inhibition, however, that study did not reach statistical significance. The results are shown in FIG. 11.
[0428] Sequence Table:
TABLE-US-00008 TABLE Ia HEAVY CHAIN CDRs Ab Type CDR 1 CDR 2 CDR 3 1D10 NA AGCTATGGCATGCAC GTTATATGGTATGATGGAAGT AGGGCCGGTATAATAGGAAC 2C12 AATAAATACTATGCAGACTCC TACAGGCTACTACTACGGTA GTGAAGGGC TGGACGTC SEQ ID NO: 1 SEQ ID NO: 2 SEQ ID NO: 3 AA SYGMH VIWYDGSNKYYADSVKG RAGIIGTTGYYYGMDV SEQ ID NO: 4 SEQ ID NO: 5 SEQ ID NO: 6 1F10 NA AGTGGTGGTTACTACT TACATCTATTACAGTGGGAGC GATGGAAGCAGTGGCTGGTA GGAGC ACCTACTACAACCCGTCCCTC CTTCCAGCAC ACGAGT SEQ ID NO: 7 SEQ ID NO: 8 SEQ ID NO: 9 AA SGGYYWS YIYYSGSTYYNPSLTS DGSSGWYFQH SEQ ID NO: 10 SEQ ID NO: 11 SEQ ID NO: 12 2C12_LC#1 NA AGCTATGGCATGCAC GTTATATGGTATGATGGAAGT AGGGCCGGTATAATAGGAAC AATAAATACTATGCAGACTCC TACAGGCTACTACTACGGTA GTGAAGGGC TGGACGTC SEQ ID NO: 13 SEQ ID NO: 14 SEQ ID NO: 15 AA SYGMH VIWYDGSNKYYADSVKG RAGIIGTTGYYYGMDV SEQ ID NO: 16 SEQ ID NO: 17 SEQ ID NO: 18 2G6_LC#1 NA AGCTATGGCATGCAC TTTATATGGTATGATGGAAGT AGGGCCGGTATAATAGGAAC AATAAATACTATGCAGACTCC TATAGGCTACTACTACGGTA GTGAAGGAC TGGACGTC SEQ ID NO: 19 SEQ ID NO: 20 SEQ ID NO: 21 AA SYGMH FIWYDGSNKYYADSVKD RAGIIGTIGYYYGMDV SEQ ID NO: 22 SEQ ID NO: 23 SEQ ID NO: 24 2G6 NA AGCTATGGCATGCAC TTTATATGGTATGATGGAAGT AGGGCCGGTATAATAGGAAC AATAAATACTATGCAGACTCC TATAGGCTACTACTACGGTA GTGAAGGAC TGGACGTC SEQ ID NO: 25 SEQ ID NO: 26 SEQ ID NO: 27 AA SYGMH FIWYDGSNKYYADSVKD RAGIIGTIGYYYGMDV SEQ ID NO: 28 SEQ ID NO: 29 SEQ ID NO: 30 2H12 NA AGCTATGGCATGCAC GTTATATGGTATGATGGAAGT AGGGCCGGTATAATAGGAAC AATAAATACTATACAGACTCC TACAGGCTACTACTACGGTA GTGAAGGGC TGGACGTC SEQ ID NO: 31 SEQ ID NO: 32 SEQ ID NO: 33 AA SYGMH VIWYDGSNKYYTDSVKG RAGIIGTTGYYYGMDV SEQ ID NO: 34 SEQ ID NO: 35 SEQ ID NO: 36 2H12_LC#2 NA AGCTATGGCATGCAC GTTATATGGTATGATGGAAGT AGGGCCGGTATAATAGGAAC AATAAATACTATACAGACTCC TACAGGCTACTACTACGGTA GTGAAGGGC TGGACGTC SEQ ID NO: 37 SEQ ID NO: 38 SEQ ID NO: 39 AA SYGMH VIWYDGSNKYYTDSVKG RAGIIGTTGYYYGMDV SEQ ID NO: 40 SEQ ID NO: 41 SEQ ID NO: 42 4A2 NA AGTAGTGGTTACTACT TACATCTATTACACTGGGAGC GATGGAAGCAGTGGCTGGTA 5B4 GGAGC GCCTACTACAACCCGTCCCTC CTTCCAGTAT 5C5 AAGAGT SEQ ID NO: 43 SEQ ID NO: 44 SEQ ID NO: 45 AA SSGYYWS YIYYTGSAYYNPSLKS DGSSGWYFQY SEQ ID NO: 46 SEQ ID NO: 47 SEQ ID NO: 48 4A9 NA GGTTACTACTGGAGC TATTTCTCTTACAGTGGGAGC AACTGGGCCTTCCACTTTGA ACCAACTACAACCCCTCCCTC CTTC AAGAGT SEQ ID NO: 49 SEQ ID NO: 50 SEQ ID NO: 51 AA GYYWS YFSYSGSTNYNPSLKS NWAFHFDF SEQ ID NO: 52 SEQ ID NO: 53 SEQ ID NO: 54 4B10 NA AGCTATGACATGCAC GTTATATCATATGATGGAACT GAACGATATTTTGACTGGTC 4C2 AATGAATACTATGCAGACTCC TTTTGACTAC GTGAAGGGC SEQ ID NO: 55 SEQ ID NO: 56 SEQ ID NO: 57 AA SYDMH VISYDGTNEYYADSVKG ERYFDWSFDY SEQ ID NO: 58 SEQ ID NO: 59 SEQ ID NO: 60 4D2 NA AGTTATGACATGCAC GTTATATCATATGATGGAACT GAACGATATTTTGACTGGTC AATGAATACTATGCAGACTCC TTTTGACTAC GTGAAGGGC SEQ ID NO: 61 SEQ ID NO: 62 SEQ ID NO: 63 AA SYDMH VISYDGTNEYYADSVKG ERYFDWSFDY SEQ ID NO: 64 SEQ ID NO: 65 SEQ ID NO: 66 4D3 NA AGCTATGACATGGAC GTTATATGGTATGATGGAAGT GAAACTGGGGAGGgCTGGTA 4F3 AATAAAtacTATGCAGACTCC CTTCGAtctc GTGAGGGGC SEQ ID NO: 67 SEQ ID NO: 68 SEQ ID NO: 69 AA SYDMD VIWYDGSNKYYADSVRG ETGEGWYFDL SEQ ID NO: 70 SEQ ID NO: 71 SEQ ID NO: 72 4E10 NA AGCTATGACATGCAC GTTATATGGTATGATGGAAGT GAGTATAGGTACAGCTGGTA AATAAATACTATGCAGACTCC CTTTGACTAC GTGAAGGGC SEQ ID NO: 73 SEQ ID NO: 74 SEQ ID NO: 75 AA SYDMH VIWYDGSNKYYADSVKG EYRYSWYFDY SEQ ID NO: 76 SEQ ID NO: 77 SEQ ID NO: 78 4F7 NA AGTTACTCCTGGAGC TATATCTATTACAGTGGGAGC AACTGGGCCTTCCACTTTGA ACCAACTACAACCCCTCCCTC CTAC AAGAGT SEQ ID NO: 79 SEQ ID NO: 80 SEQ ID NO: 81 AA SYSWS YIYYSGSTNYNPSLKS NWAFHFDY SEQ ID NO: 82 SEQ ID NO: 83 SEQ ID NO: 84 5E3 NA AGCTATAGCATGCAC TCCATTAGTAGTAGTAGTAGT GGGGAAACTGGAACTAACTA TACATATACTACGCAGACTCA CTACTACTACGGTATGGACG GTGAAGGGC TC SEQ ID NO: 85 SEQ ID NO: 86 SEQ ID NO: 87 AA SYSMH SISSSSSYIYYADSVKG GETGTNYYYYGMDV SEQ ID NO: 88 SEQ ID NO: 89 SEQ ID NO: 90 17H8 NA AGTTACTACTGGAGC TATATCTATTACATTGGGAGC GATTCCCGGTATAGAAGTGG 23B6 ACCAACTACAACCCCTCCCTC CTGGTACGATGCTTTTGATA 28D10 AAGAGT TC SEQ ID NO: 91 SEQ ID NO: 92 SEQ ID NO: 93 AA SYYWS YIYYIGSTNYNPSLKS DSRYRSGWYDAFDI SEQ ID NO: 94 SEQ ID NO: 95 SEQ ID NO: 96 16C1 NA GGTTACTACTGGAGC TATATCTATTACATTGGGAGC GATGGGAGCAGTGGCTGGTA ACCAACTACAACCCCTCCCTC CCGGTGGTTCGACCCC AAGAGT SEQ ID NO: 97 SEQ ID NO: 98 SEQ ID NO: 99 AA GYYWS YIYYIGSTNYNPSLKS DGSSGWYRWFDP SEQ ID NO: 100 SEQ ID NO: 101 SEQ ID NO: 102 16A4 NA AGTTACTACTGGAGC TATATCTATTACAGTGGGAGC GATCAAAGGCGGATAGCAGC ACCAATTACAACCCCTCCCTC AGCTGGTACCCACTTCTACG AAGAGT GTATGGACGTC SEQ ID NO: 103 SEQ ID NO: 104 SEQ ID NO: 105 AA SYYWS YIYYSGSTNYNPSLKS DQRRIAAAGTHFYGMDV SEQ ID NO: 106 SEQ ID NO: 107 SEQ ID NO: 108 16E2 NA AGCTATGGCATGCAC GTGATATGGTATGATGGAAGT GACGGGTGGGAGCTGTCCTT 17E10 AATAAATACTATGCAGACTCC TGACTAC 20B12 GTGAAGGGC SEQ ID NO: 109 SEQ ID NO: 110 SEQ ID NO: 111 AA SYGMH VIWYDGSNKYYADSVKG DGWELSFDY SEQ ID NO: 112 SEQ ID NO: 113 SEQ ID NO: 114 22G10 NA AGTTATGCCATGAAC ACTATTAGTGGTGGTGGTGCT GGGGGAATGGGGGGATACTA AACACATACTACGCAGACTCC CTACGGTATGGACGTC GTGAAGGGC SEQ ID NO: 115 SEQ ID NO: 116 SEQ ID NO: 117 AA SYAMN TISGGGANTYYADSVKG GGMGGYYYGMDV SEQ ID NO: 118 SEQ ID NO: 119 SEQ ID NO: 120 16H2 NA AGCTACTTTATTCAC ATAATCAACCCTATTAGTGTT GGGGGGATACAGCTATGGTT 20D3 AGCACAAGCTACGCACAGAAG ACATTTTGACTAC 23E7 TTCCAGGGC SEQ ID NO: 121 SEQ ID NO: 122 SEQ ID NO: 123 AA SYFIH IINPISVSTSYAQKFQG GGIQLWLHFDY SEQ ID NO: 124 SEQ ID NO: 125 SEQ ID NO: 126 22D1 NA AGCTACTTTATTCAC ATAATCAACCCTATTAGTGTT GGGGGGATACAGCTATGGTT AGCACAAGCTACGCACAGAAG ACATTTGGACTAC TTCCAGGGC SEQ ID NO: 127 SEQ ID NO: 128 SEQ ID NO: 129 AA SYFIH IINPISVSTSYAQKFQG GGIQLWLHLDY SEQ ID NO: 130 SEQ ID NO: 131 SEQ ID NO: 132 25F8 NA AGCTACTATATTCAC ATAATCAACCCCAGTGGTGGT GGGGGAATACAGCTATGGTT AGCACAAGGTACGCACAGAAG ACATTttGACTAC TTCCAGGGC SEQ ID NO: 133 SEQ ID NO: 134 SEQ ID NO: 135 AA SYYIH IINPSGGSTRYAQKFQG GGIQLWLHFDY SEQ ID NO: 136 SEQ ID NO: 137 SEQ ID NO: 138 26F12 NA AACTACTATATGTCC ATAATCAACCCTAGTGGTGGT GGGGGGATACAACTATGGTT 27B3 GACTCAACCTACGCACAGAAG ACATTTTGACTAC TTCCAGGGC SEQ ID NO: 139 SEQ ID NO: 140 SEQ ID NO: 141 AA NYYMS IINPSGGDSTYAQKFQG GGIQLWLHFDY SEQ ID NO: 142 SEQ ID NO: 143 SEQ ID NO: 144 26D1 NA AGCTACTATATGTCC ATAATCCACCCTAGTGGTGGT GGGGGGATAAAACTATGGTT GACACAACCTACGCACAGAAG ACATTTTGACTAT TTCCAGGGC SEQ ID NO: 145 SEQ ID NO: 146 SEQ ID NO: 147 AA SYYMS IIHPSGGDTTYAQKFQG GGIKLWLHFDY SEQ ID NO: 148 SEQ ID NO: 149 SEQ ID NO: 150 25G10 NA GGTTACTACTGGAGC TATATCTATTACATTGGGAGC GATGGGAGCAGTGGCTGGTA ACCAACTACAACCCCTCCCTC CCGGTGGTTCGACCCC AAGAGT SEQ ID NO: 151 SEQ ID NO: 152 SEQ ID NO: 153 AA GYYWS YIYYIGSTNYNPSLKS DGSSGWYRWFDP SEQ ID NO: 154 SEQ ID NO: 155 SEQ ID NO: 156 23A10 NA CGCTATGGCATACAC GTTATATGGTATGATGGAAGT AGGGCCGGTATACCTGGAAC AATAAATACTATGCAGACTCC TACGGGCTACTACTATGGTA GTGAAGGGC TGGACGTC SEQ ID NO: 157 SEQ ID NO: 158 SEQ ID NO: 159 AA RYGIH VIWYDGSNKYYADSVKG RAGIPGTTGYYYGMDV SEQ ID NO: 160 SEQ ID NO: 161 SEQ ID NO: 162 19B5 NA AGCTACTTTATTCAC ATTATCAACCCTATTAGTGTT GGGGGGATACAGCTATGGTT AGCACAAGCTACGCACAGAAG ACATTTGGACTAC TTCCAGGGC SEQ ID NO: 163 SEQ ID NO: 164 SEQ ID NO: 165 AA SYFIH IINPISVSTSYAQKFQG GGIQLWLHLDY SEQ ID NO: 166 SEQ ID NO: 167 SEQ ID NO: 168
TABLE-US-00009 TABLE Ib LIGHT CHAIN CDRs Ab Type CDR 1 CDR 2 CDR 3 1D10 NA TCTGGAGATAGATTGG CAAGATACCAAGCGGCCCTCA CAGGCGTGGGACAGCAGCAC 2C12 GGGAAAAATATACTTG TGTGGTA C SEQ ID NO: 169 SEQ ID NO: 170 SEQ ID NO: 171 AA SGDRLGEKYTC QDTKRPS QAWDSSTVV SEQ ID NO: 172 SEQ ID NO: 173 SEQ ID NO: 174 1F10 NA AGGGCCAGTCGGAGTA GGTCCATCCAGCAGGGCCACT CAGCAGTATGGTAGCTCATT TTAGCAGCAGCTACTT CACT AGCC SEQ ID NO: 175 SEQ ID NO: 176 SEQ ID NO: 177 AA RASRSISSSYLA GPSSRAT QQYGSSFT SEQ ID NO: 178 SEQ ID NO: 179 SEQ ID NO: 180 2C12_LC#1 NA AGGtCTAGTCAAAGcc AAGGTTTCTAACTGGGactct ATGCAAGGTATAGTGTGGCC tcgtaTACAGTGATGG GTGCAGT AAACAcctACTTGAAT SEQ ID NO: 181 SEQ ID NO: 182 SEQ ID NO: 183 AA RSSQSLVYSDGNTYLN KVSNWDS MQGIVWPCS SEQ ID NO: 184 SEQ ID NO: 185 SEQ ID NO: 186 2G6_LC#1 NA AGGTCTAGTCAAAGCC CAGGTTTCTAACTGGGACTCT ATGCAAGATACACTGTGGCC TCGTATACAGTGATGG GTGCAGT AAACACCTACTTGAAT SEQ ID NO: 187 SEQ ID NO: 188 SEQ ID NO: 189 AA RSSQSLVYSDGNTYLN QVSNWDS MQDTLWPCS SEQ ID NO: 190 SEQ ID NO: 191 SEQ ID NO: 192 2G6 NA TCTGGAGATAGGTTGG CAAGATACCAAGCGGCCCTCA CAGGCGTGGGACAGCAGCAC GGGAAAAATATACTTG TGTGGTA C SEQ ID NO: 193 SEQ ID NO: 194 SEQ ID NO: 195 AA SGDRLGEKYTC QDTKRPS QAWDSSTVV SEQ ID NO: 196 SEQ ID NO: 197 SEQ ID NO: 198 2H12 NA TCTGGAGATAGATTGG CAAGATACCAAGCGGCCCTCA CAGGCGTGGGACAGCAGCAC GGGAAAAATATACTTG TGTGGTA C SEQ ID NO: 199 SEQ ID NO: 200 SEQ ID NO: 201 AA SGDRLGEKYTC QDTKRPS QAWDSSTVV SEQ ID NO: 202 SEQ ID NO: 203 SEQ ID NO: 204 2H12_LC#2 NA AGGTCTAGTCAAAGCC AAGGTTTCTAACTGGGACTCT ATGCAAGATACACTGTGGCC TCGTATACAGTGATGG GTGCAGT AAACACCTACTTGAAT SEQ ID NO: 205 SEQ ID NO: 206 SEQ ID NO: 207 AA RSSQSLVYSDGNTYLN KVSNWDS MQDTLWPCS SEQ ID NO: 208 SEQ ID NO: 209 SEQ ID NO: 210 4A2 NA AGGgcCAGTCGGAATA GGTCCATCCAGCAGGGccaCT CAGCAGTATGGtagctCATT 5B4 TTAGCAGCAGCTACtt CACT 5C5 aGCC SEQ ID NO: 211 SEQ ID NO: 212 SEQ ID NO: 213 AA RASRNISSSYLA GPSSRAT QQYGSSFT SEQ ID NO: 214 SEQ ID NO: 215 SEQ ID NO: 216 4A9 NA ACTGGGAGCAGCTCCA GGTAACAACAATCGGCCCTCA CAGTCCTATGACAGCagACT ACATCGGGACAGGTTA GAGTGGTTGGGTG TGCTGTACAC SEQ ID NO: 217 SEQ ID NO: 218 SEQ ID NO: 219 AA TGSSSNIGTGYAVH GNNNRPS QSYDSRLSGWV SEQ ID NO: 220 SEQ ID NO: 221 SEQ ID NO: 222 4B10 NA AGGGCCAGTCAGAGTG GGTGCATCCAGCAGGGCCACT CAGCAGTACAGTAACTCgtg 4C2 TTAGCAACACCTACTT GACG AGCC SEQ ID NO: 223 SEQ ID NO: 224 SEQ ID NO: 225 AA RASQSVSNTYLA GASSRAT QQYSNSWT SEQ ID NO: 226 SEQ ID NO: 227 SEQ ID NO: 228 4D2 NA AGGGCCAGTCAGAGTG GGTGCATCCAGCAGGGCCGCT CagcagTATAGTAacTcgtg TTAGCAACACCTACTT GACG AGCC SEQ ID NO: 229 SEQ ID NO: 230 SEQ ID NO: 231 AA RASQSVSNTYLA GASSRAA QQYSNSWT SEQ ID NO: 232 SEQ ID NO: 233 SEQ ID NO: 234 4D3 NA AGGGCCAGTCAGAGTG GGTGCATCCAGCAGGGCCACT CAGCAGTATGGTAGCTCGTG 4F3 TTAGCAGCAGCTACTT GACG AGCC SEQ ID NO: 235 SEQ ID NO: 236 SEQ ID NO: 237 AA RASQSVSSSYLA GASSRAT QQYGSSWT SEQ ID NO: 238 SEQ ID NO: 239 SEQ ID NO: 240 4E10 NA AGGGCCAGTCAGAGTG GGTGCATCCAGCAGGGTCACT CAGCAATATAGTAACTCGTG TTGGCAGCAGCTACTT GACG AGCC SEQ ID NO: 241 SEQ ID NO: 242 SEQ ID NO: 243 AA RASQSVGSSYLA GASSRVT QQYSNSWT SEQ ID NO: 244 SEQ ID NO: 245 SEQ ID NO: 246 4F7 NA ACTGGGAGCAGCTCCA GGTAACAGCAATCGGCCCTCA CAGTCCTATGACAGCAGTCT ATATCGGGACAGGTTA GAGTGGTTGGGTG TGATGTACAC SEQ ID NO: 247 SEQ ID NO: 248 SEQ ID NO: 249 AA TGSSSNIGTGYDVH GNSNRPS QSYDSSLSGWV SEQ ID NO: 250 SEQ ID NO: 251 SEQ ID NO: 252 5E3 NA TCTGGAGATAAATTGG CAAGATAGCAAGCGGCCCTCA CAGGCGTGGGACAGCAGCAC GGGATGAATATGCTTG TGTGGTA C SEQ ID NO: 253 SEQ ID NO: 254 SEQ ID NO: 255 AA SGDKLGDEYAC QDSKRPS QAWDSSTVV SEQ ID NO: 256 SEQ ID NO: 257 SEQ ID NO: 258 17H8 NA AGGGCCAGTCAGAGTG GGTGCATCCAGCAGGGCCACT CAGCAGTATGGTAAATCACC 23B6 TTGCCGGCAGCTACCT GATCACC 28D10 AGCC SEQ ID NO: 259 SEQ ID NO: 260 SEQ ID NO: 261 AA RASQSVAGSYLA GASSRAT QQYGKSPIT SEQ ID NO: 262 SEQ ID NO: 263 SEQ ID NO: 264 16C1 NA AGGGCCAGCCAGAGTG GGTGCATCCAGCAGGGCCACT CAGCAGTATGGTAACTCACC TTAGCAGCAGCTACTT GCTCACT AGCC SEQ ID NO: 265 SEQ ID NO: 266 SEQ ID NO: 267 AA RASQSVSSSYLA GASSRAT QQYGNSPLT SEQ ID NO: 268 SEQ ID NO: 269 SEQ ID NO: 270 16A4 NA AGGGCCAGTCAGAGTG GGTACATCCAGCAGGGCCACT CAGCAGTACGGTAGCTCACC TTAGCAGCAGTTATTT TTTCACT AGCC SEQ ID NO: 271 SEQ ID NO: 272 SEQ ID NO: 273 AA RASQSVSSSYLA GTSSRAT QQYGSSPFT SEQ ID NO: 274 SEQ ID NO: 275 SEQ ID NO: ***276 16E2 NA CGGGCGAGTCAGGGCA GCTGCATCCAGTTTGCAAAGT CAACACTATTTTACTTACCC 17E10 TTAGCAATTATTTAGC TCGGACG 20B12 C SEQ ID NO: 277 SEQ ID NO: 278 SEQ ID NO: 279 AA RASQGISNYLA AASSLQS QHYFTYPRT SEQ ID NO: 280 SEQ ID NO: 281 SEQ ID NO: 282 22G10 NA AGGGCCAGTCAGAGTA GGTGCATTTACCAGGGCCACT CAGCAGTATAATTACTGGCC TTAGCAGCAACTTAGC GCTCACT C SEQ ID NO: 283 SEQ ID NO: 284 SEQ ID NO: 285 AA RASQSISSNLA GAFTRAT QQYNYWPLT SEQ ID NO: 286 SEQ ID NO: 287 SEQ ID NO: 288 16H2 NA TCTGGAAGCAGCTCCA ACTAATAATCAGCGGCCCTCA GCAACATGGGATGACAGCCT 20D3 ACATCGGAAGTAATTT GAATGGTTGGGTG 23E7 TGTAAAC SEQ ID NO: 289 SEQ ID NO: 290 SEQ ID NO: 291 AA SGSSSNIGSNFVN TNNQRPS ATWDDSLNGWV SEQ ID NO: 292 SEQ ID NO: 293 SEQ ID NO: 294 22D1 NA TCTGGAAGCAGCTCCA ACTAATAATCAGCGGCCCTCA GCAACATGGGATGACAGTAT ACATCGGAAGCAATTT GAATGGTTGGGTG TGTAAAC SEQ ID NO: 295 SEQ ID NO: 296 SEQ ID NO: 297 AA SGSSSNIGSNFVN TNNQRPS ATWDDSMNGWV SEQ ID NO: 298 SEQ ID NO: 299 SEQ ID NO: 300 25F8 NA TCTGGAAGCAGCTCCA ACTAATAATCAGCGGCCCTCA GCAGCATGGGATGACAGCCT ACATCGGAAGGAATTT GAATGGTTGGGTG TGTAAAC SEQ ID NO: 301 SEQ ID NO: 302 SEQ ID NO: 303 AA SGSSSNIGRNFVN TNNQRPS AAWDDSLNGWV SEQ ID NO: 304 SEQ ID NO: 305 SEQ ID NO: 306 26F12 NA TCTGGAAGCCGCTCCA ACTAATTATCAGCGGCCCTCA GCAGTATGGGATGACAGCCT 27B3 ACATCGGAAGTAATTT GAATGGTTGGGTG TGTAAAC SEQ ID NO: 307 SEQ ID NO: 308 SEQ ID NO: 309 AA SGSRSNIGSNFVN TNYQRPS AVWDDSLNGWV SEQ ID NO: 310 SEQ ID NO: 311 SEQ ID NO: 312 26D1 NA TCTGGAAGCCGCTCCA ACTAATAATCAGCGGCCCTCA GCAGTATGGGATGACAGCCT ACATCGGAAGTAATTT GAATGGTTGGGTG TGTAAAC SEQ ID NO: 313 SEQ ID NO: 314 SEQ ID NO: 315 AA SGSRSNIGSNFVN TNNQRPS AVWDDSLNGWV SEQ ID NO: 316 SEQ ID NO: 317 SEQ ID NO: 318 25G10 NA AGGGCCAGTCAGAGTG GGTGCATCCAGCAGGGCCACT CAGCAGTATGGTAACTCACC TTAGCAGCAGCTACTT GCTCACT AGCC SEQ ID NO: 319 SEQ ID NO: 320 SEQ ID NO: 321 AA RASQSVSSSYLA GASSRAT QQYGNSPLT SEQ ID NO: 322 SEQ ID NO: 323 SEQ ID NO: 324 23A10 NA TCTGGAGATAGATTGG CAAGATAATAAGTGGCCCTCA CAGGCGTGGGACAGCAGcac GGGAGAAATATGTTTG TGTGGTA C SEQ ID NO: 325 SEQ ID NO: 326 SEQ ID NO: 327 AA SGDRLGEKYVC QDNKWPS QAWDSSTVV SEQ ID NO: 328 SEQ ID NO: 329 SEQ ID NO: 330 19B5 NA TCTGGAAGCAGGTCCA ACTAATAATCAGCGGCCCTCA GCAACATGGGATGACAGTAT ACATCGGAAGCAATTT GAATGGTTGGGTG TGTAAAC SEQ ID NO: 331 SEQ ID NO: 332 SEQ ID NO: 333 AA SGSRSNIGSNFVN TNNQRPS ATWDDSMNGWV SEQ ID NO: 334 SEQ ID NO: 335 SEQ ID NO: 336
[0429] Anti-CDH19 Variable Region Amino Acid Sequences and Polynucleotide Sequences
TABLE-US-00010 TABLE IIa Heavy Chain Variable Region Polynucleotide and Amino acid Sequences SEQ ID NO. DESIGNATION SOURCE TYPE SEQUENCE 337 17H8 artificial nt CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTT 23B6 CGGAGACCCTGTCCCTCACGTGCACTGTCTCTGGTGGCTCCAT 28D10 CAATAGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGGAAG GGACTGGAGTGGATTGGGTATATCTATTACATTGGGAGCACCA ACTACAACCCCTCCCTCAAGAGTCGCGTCACCATATCAGTAGA CACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACC GCTGCGGACACGGCCCTGTATTACTGTGCGAGAGATTCCCGGT ATAGAAGTGGCTGGTACGATGCTTTTGATATCTGGGGCCAAGG GACAATGGTCACCGTCTCTTCA 338 17H8 artificial aa QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGK 23B6 GLEWIGYIYYIGSTNYNPSLKSRVTISVDTSKNQFSLKLSSVT 28D10 AADTALYYCARDSRYRSGWYDAFDIWGQGTMVTVSS 339 4A2 artificial nt CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTT 5B4 CACAGACCCTGTCCCTCACCTGCACTGTCTCTGGTGGCTCCAT 5C5 CAGCAGTAGTGGTTACTACTGGAGCTGGATCCGCCAGCACCCA GGGAAGGGCCTGGAGTGGATTGGGTACATCTATTACACTGGGA GCGCCTACTACAACCCGTCCCTCAAGAGTCGAGTTACCATATC AGTAGACACGTCTAAGAACCAGTTCTCCCTGAAGCTGAGCTCT GTGACTGCCGCGGACACGGCCGTGTATTACTGTGCGAGAGATG GAAGCAGTGGCTGGTACTTCCAGTATTGGGGCCAGGGCACCCT GGTCACCGTCTCCTCA 340 4A2 artificial aa QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHP 5B4 GKGLEWIGYIYYTGSAYYNPSLKSRVTISVDTSKNQFSLKLSS 5C5 VTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSS 341 16H2 artificial nt CAGGTGCAGCTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTG 20D3 GGGCCTCAGTGAAGGTTTCCTGCAAGGTTTCTGGATACACCTT 23E7 CACCAGCTACTTTATTCACTGGGTGCGCCAGGCCCCTGGACAA GGGCTTGAGTGGATGGGAATAATCAACCCTATTAGTGTTAGCA CAAGCTACGCACAGAAGTTCCAGGGCAGAGTCACCATGACCAG GGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCAGCCTG AGATCTGAGGACACGGCCGTGTATTACTGTGCGCGAGGGGGGA TACAGCTATGGTTACATTTTGACTACTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 342 16H2 artificial aa QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQ 20D3 GLEWMGIINPISVSTSYAQKFQGRVTMTRDTSTSTVFMELSSL 23E7 RSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS 343 26F12 artificial nt CAGGTGCAGTTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTG 27B3 GGGCCTCAGTGAAGGTTTCCTGCAAGGCATCTAGATACACCTT CACCAACTACTATATGTCCTGGGTGCGACAGGCCCCTGGACAA GGGCTTGAGTGGATGGGAATAATCAACCCTAGTGGTGGTGACT CAACCTACGCACAGAAGTTCCAGGGCAGACTCACCATGACCGG GGACACGTCCACGAGCACAGTCTACATGGAGCTGAGCAGCCTG AGATCTGAGGACACGGCCGTGTATTACTGTGCGAGAGGGGGGA TACAACTATGGTTACATTTTGACTACTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 344 26F12 artificial aa QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQ 27B3 GLEWMGIINPSGGDSTYAQKFQGRLTMTGDTSTSTVYMELSSL RSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS 345 4B10 artificial nt CAGGTGCAGTTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG 4C2 GGAGGTCCCTGAGACTCTCCTGTGCAGCCTCTGGATTCACCTT CAGTAGCTATGACATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTTATATCATATGATGGAACTAATG AATACTATGCAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACACTTCCAAGAACACGCTGTATTTGCAAATGAACAGCCTG AGAGCTGAGGACACGGCTGTATATTACTGTGCGAGAGAACGAT ATTTTGACTGGTCTTTTGACTACTGGGGCCAGGGAACCCTGGT CAGTGTCTCCTCA 346 4B10 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGK 4C2 GLEWVAVISYDGTNEYYADSVKGRFTISRDTSKNTLYLQMNSL RAEDTAVYYCARERYFDWSFDYWGQGTLVSVSS 347 4D3 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG 4F3 GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCTCCTT CAGTAGCTATGACATGGACTGGGTCCGCCAGACTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTTATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAGGGGCCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTTTCTGCAAATGAACAGCCTG AGAGTCGAGGACACGGCTGTGTATTACTGTGCGAGAGAAACTG GGGAGGGCTGGTACTTCGATCTCTGGGGCCGTGGCACCCTGGT CACTGTCTCCTCA 348 4D3 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFSFSSYDMDWVRQTPGK 4F3 GLEWVAVIWYDGSNKYYADSVRGRFTISRDNSKNTLFLQMNSL RVEDTAVYYCARETGEGWYFDLWGRGTLVTVSS 349 16E2 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG 17E10 GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCATCTT 20B12 CAGTAGCTATGGCATGCACTGGGTCCGCCAGACTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTGATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACATTTCCAAGAACACGCTGTATCTGCAAATGAACAGCCTG AGAGTCGAGGACACGGCTGTGTATTACTGTGCGAGAGACGGGT GGGAGCTGTCCTTTGACTACTGGGGCCAGGGAACCCTGGTCAC CGTCTCCTCA 350 16E2 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFIFSSYGMHWVRQTPGK 17E10 GLEWVAVIWYDGSNKYYADSVKGRFTISRDISKNTLYLQMNSL 20B12 RVEDTAVYYCARDGWELSFDYWGQGTLVTVSS 351 1D10 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG 2C12 GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTAGCTATGGCATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGTCAGTTATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTATCTGCAAATGAATAGCCTG AGAGCTGAGGACACGGCTGTGTATTACTGCGCGAGAAGGGCCG GTATAATAGGAACTACAGGCTACTACTACGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 352 1D10 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGK 2C12 GLEWVSVIWYDGSNKYYADSVKGRFTISRDNSKNTLYLQMNSL RAEDTAVYYCARRAGIIGTTGYYYGMDVWGQGTTVTVSS 353 16C1 artificial nt CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTT CGGAGACCCTGTCCCTCACTTGTACTGTCTCTGGTGGCTCCAT CAGTGGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGGAAG GGACTGGAGTGGATTGGGTATATCTATTACATTGGGAGCACCA ACTACAACCCCTCCCTCAAGAGTCGAGTCACCATGTCAATAGA CACGTCCAAGAACCAGTTCTCCCTGACGCTGAGCTCTTTGACC GCTGCGGACACGGCCGTGTATTTCTGTGCGAGAGATGGGAGCA GTGGCTGGTACCGGTGGTTCGACCCCTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 354 16C1 artificial aa QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGK GLEWIGYIYYIGSTNYNPSLKSRVTMSIDTSKNQFSLTLSSLT AADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSS 355 25G10 artificial nt CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTT CGGAGACCCTGTCCCTCACCTGCACTGTCTCTGGTGGCTCCAT CAGTGGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGGAAG GGACTGGAGTGGATTGGGTATATCTATTACATTGGGAGCACCA ACTACAACCCCTCCCTCAAGAGTCGAGTCACCATGTCAGTAGA CACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACC GCTGCGGACACGGCCGTGTATTACTGTGCGAGAGATGGGAGCA GTGGCTGGTACCGGTGGTTCGACCCCTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 356 25G10 artificial aa QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGK GLEWIGYIYYIGSTNYNPSLKSRVTMSVDTSKNQFSLKLSSVT AADTAVYYCARDGSSGWYRWFDPWGQGTLVTVSS 357 16A4 artificial nt CAGGTGCAGCTGCAGGAGTCgGGCCCAGGACTGGCGAAgcctt cGGAGACcctgtccctcacctgCACTGTCTCTGGTGACTCCAT CACTAGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGGAAG GGACTGGAGTGGATTGGGTATATCTATTACAGTGGGAGCACCA ATTACAACCCCTCCCTCAAGAGTCGAGTCACCATATCAGTAGA CACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGTTCTGTGACC GCTGCGGACACGGCCGTGTATTACTGTGCGAGAGATCAAAGGC GGATAGCAGCAGCTGGTACCCACTTCTACGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 358 16A4 artificial aa QVQLQESGPGLAKPSETLSLTCTVSGDSITSYYWSWIRQPPGK GLEWIGYIYYSGSTNYNPSLKSRVTISVDTSKNQFSLKLSSVT AADTAVYYCARDQRRIAAAGTHFYGMDVWGQGTTVTVSS 359 1F10 artificial nt CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTT CACAGACCCTGTCCCTCACCTGCACTGTCTCTGGTGGCTCCAT CAGCAGTGGTGGTTACTACTGGAGCTGGATCCGCCAGCACCCA GGGAAGGGCCTGGAGTGGATTGGGTACATCTATTACAGTGGGA GCACCTACTACAACCCGTCCCTCACGAGTCGAGTTACCATATC AGTAGACACGTCTAAGAACCAGTTCTCCCTGAAGCTGAGCTCT GTGACTGCCGCGGACACGGCCGTGTATTACTGTGCGAGAGATG GAAGCAGTGGCTGGTACTTCCAGCACTGGGGCCAGGGCACCCT GGTCACCGTCTCCTCA 360 1F10 artificial aa QVQLQESGPGLVKPSQTLSLTCTVSGGSISSGGYYWSWIRQHP GKGLEWIGYIYYSGSTYYNPSLTSRVTISVDTSKNQFSLKLSS VTAADTAVYYCARDGSSGWYFQHWGQGTLVTVSS 361 4A9 artificial nt CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTT CGGAGACCCTGTCCCTCACCTGCACTGTCTCTGGTGGCTCCAT CAGTGGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGAAAG GGACTGGAGTGGTTTGCATATTTCTCTTACAGTGGGAGCACCA ACTACAACCCCTCCCTCAAGAGTCGAGTCACCTTATCAGTAGA CACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACC GCTGCGGACACGGCCGTGTATTACTGTGCGAGGAACTGGGCCT TCCACTTTGACTTCTGGGGCCAGGGAACCCTGGTCACCGTCTC CCA 362 4A9 artificial aa QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGK GLEWFAYFSYSGSTNYNPSLKSRVTLSVDTSKNQFSLKLSSVT AADTAVYYCARNWAFHFDFWGQGTLVTVSS 363 4F7 artificial nt CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTT CGGAGACCCTGTCCCTCACCTGCACTGTCTCTGGTGGCTCCAT CAGTAGTTACTCCTGGAGCTGGATCCGGCAGCCCCCAGGGAAG GGACTGGAGTGGATTGGGTATATCTATTACAGTGGGAGCACCA ACTACAACCCCTCCCTCAAGAGTCGAGTCACCATATCATTAGA CACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACC GCTGCGGACACGGCCGTGTATTACTGTGCGAGGAACTGGGCCT TCCACTTTGACTACTGGGGCCAGGGAACCCTGGTCACCGTCTC CTCA 364 4F7 artificial aa QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGK GLEWIGYIYYSGSTNYNPSLKSRVTISLDTSKNQFSLKLSSVT AADTAVYYCARNWAFHFDYWGQGTLVTVSS 365 22D1 artificial nt CAGGTGCAGCTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTG GGGCCTCAGTGAGGGTTTCCTGCAAGGTTTCTGGATACACCTT CACCAGCTACTTTATTCACTGGGTACGCCAGGCCCCTGGACAA GGGCTTGAGTGGATGGGAATAATCAACCCTATTAGTGTTAGCA CAAGCTACGCACAGAAGTTCCAGGGCAGAGTCACCATGACCAG GGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCAGCCTG AGATCTGAGGACACGGCCGTGTATTACTGTGCGCGAGGGGGGA TACAGCTATGGTTACATTTGGACTACTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 366 22D1 artificial aa QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQ GLEWMGIINPISVSTSYAQKFQGRVTMTRDTSTSTVFMELSSL RSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS 367 19B5 artificial nt CAGGTGCAGTTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTG GGGCCTCAGTGAAGGTTTCCTGCAAGGTTTCTGGATACACCTT CACCAGCTACTTTATTCACTGGGTGCGCCAGGCCCCTGGACAA GGGCTTGAATGGATGGGAATTATCAACCCTATTAGTGTTAGCA CAAGCTACGCACAGAAGTTCCAGGGCAGAGTCACCATGACCAG GGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCAGcCTG AGATCTGAGGACACGGCCGTGTATTACTGTGCGCGAGGGGGGA TACAGCTATGGTTACATTTGGACTACTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 368 19B5 artificial aa QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQ GLEWMGIINPISVSTSYAQKFQGRVTMTRDTSTSTVFMELSSL RSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS 369 25F8 artificial nt CAGGTGCAGCTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTG GGGCCTCAGTGAAGGTTTCCTGCAAGGCATCTGGATACACCTT CACCAGCTACTATATTCACTGGGTGCGCCAGGCCCCTGGACAA GGACTTGAGTGGATGGGAATAATCAACCCCAGTGGTGGTAGCA CAAGGTACGCACAGAAGTTCCAGGGCAGAGTCACCATGACCAG GGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCagcctG AGATCTGAGGACACGGCCGTGTATTACTGTGCGCGAGGGGGAA TACAGCTATGGTTACATTttGACTACTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 370 25F8 artificial aa QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQ GLEWMGIINPSGGSTRYAQKFQGRVTMTRDTSTSTVFMELSSL RSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS 371 26D1 artificial nt CAGGTGCAGTTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTG GGGCCTCAGTGAAGGTTTCCTGTAAGGCATCTAGATACACCTT CACCAGCTACTATATGTCCTGGGTGCGACAGGCCCCTGGACAA GGGCTTGAGTGGATGGGAATAATCCACCCTAGTGGTGGTGACA CAACCTACGCACAGAAGTTCCAGGGCAGAGTCACCATGACCGG
GGACACGTCCACGAGCACAGTCTACATGGAGCTGAGCAGCCTG AGATCTGAGGACACGGCCGTGTATTACTGTGCGAGAGGGGGGA TAAAACTATGGTTACATTTTGACTATTGGGGCCAGGGAACCCT GGTCACCGTCTCCTCA 372 26D1 artificial aa QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQ GLEWMGIIHPSGGDTTYAQKFQGRVTMTGDTSTSTVYMELSSL RSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSS 373 4D2 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCCTCTGGATTCACCTT CAGTAGTTATGACATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTTATATCATATGATGGAACTAATG AATACTATGCAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACACTTCCAAGAACACGCTGTATTTGCAAATGAACAGCCTG AGAGCTGAGGACACGGCTGTATATTACTGTGCGAGAGAACGAT ATTTTGACTGGTCTTTTGACTACTGGGGCCAGGGAACCCTGGT CAGTGTCTCCTCA 374 4D2 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGK GLEWVAVISYDGTNEYYADSVKGRFTISRDTSKNTLYLQMNSL RAEDTAVYYCARERYFDWSFDYWGQGTLVSVSS 375 4E10 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTAGCTATGACATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTTATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACAATTCCACGAACACGCTGCATCTGCAAATGAACAGCCCG AGAGCCGAGGACACGGCTGTGTACTACTGTGCGAGAGAGTATA GGTACAGCTGGTACTTTGACTACTGGGGCCAGGGAACCCTGGT CACCGTCTCCTCA 376 4E10 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGK GLEWVAVIWYDGSNKYYADSVKGRFTISRDNSTNTLHLQMNSP RAEDTAVYYCAREYRYSWYFDYWGQGTLVTVSS 377 22G10 artificial nt GAGGTGCAACTGTTGGAGTCTGGGGGAGGCTTGGTACAGCCTG GGGGGTCCCTGAGACTCTCCTGTGCAGCCTCTGGATTCACCTT TAGCAGTTATGCCATGAACTGGGTCCGCCAGGCTCCAGGGAAG GGGCTGGAGTGGGTCTCAACTATTAGTGGTGGTGGTGCTAACA CATACTACGCAGACTCCGTGAAGGGCCGGTTCACCATCTCCAG TGACAATTCCAAGAGCACGCTGTATCTGCAAATGAACAGCCTG AGAGCCGCGGACACGGCCGTATATCACTGTGCGAAAGGGGGAA TGGGGGGATACTACTACGGTATGGACGTCTGGGGCCAAGGGAC CACGGTCACCGTCTCCTCA 378 22G10 artificial aa EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGK GLEWVSTISGGGANTYYADSVKGRFTISSDNSKSTLYLQMNSL RAADTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSS 379 2C12_LC#1 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTAGCTATGGCATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGTCAGTTATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTATCTGCAAATGAATAGCCTG AGAGCTGAGGACACGGCTGTGTATTACTGCGCGAGAAGGGCCG GTATAATAGGAACTACAGGCTACTACTACGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 380 2C12_LC#1 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGK GLEWVSVIWYDGSNKYYADSVKGRFTISRDNSKNTLYLQMNSL RAEDTAVYYCARRAGIIGTTGYYYGMDVWGQGTTVTVSS 381 2H12_LC#2 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTAGCTATGGCATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTTATATGGTATGATGGAAGTAATA AATACTATACAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTATCTGCAAATGAATAGCCTG AGAGCTGAGGACACGGCTGTGTATTACTGTGCGAGAAGGGCCG GTATAATAGGAACTACAGGCTACTACTACGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 382 2H12_LC#2 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGK GLEWVAVIWYDGSNKYYTDSVKGRFTISRDNSKNTLYLQMNSL RAEDTAVYYCARRAGIIGTTGYYYGMDVWGQGTTVTVSS 383 2G6_LC#1 artificial nt CAGGTGCAGTTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTAGCTATGGCATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCATTTATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAAGGACCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTATCTGCAAATGAAAAGCCTG AGAGCTGAGGACACGGCTGTGTATTACTGTGCGAGAAGGGCCG GTATAATAGGAACTATAGGCTACTACTACGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 384 2G6_LC#1 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGK GLEWVAFIWYDGSNKYYADSVKDRFTISRDNSKNTLYLQMKSL RAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSS 385 2H12 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTAGCTATGGCATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTTATATGGTATGATGGAAGTAATA AATACTATACAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTATCTGCAAATGAATAGCCTG AGAGCTGAGGACACGGCTGTGTATTACTGTGCGAGAAGGGCCG GTATAATAGGAACTACAGGCTACTACTACGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 386 2H12 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGK GLEWVAVIWYDGSNKYYTDSVKGRFTISRDNSKNTLYLQMNSL RAEDTAVYYCARRAGIIGTTGYYYGMDVWGQGTTVTVSS 387 2G6 artificial nt CAGGTGCAGTTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTAGCTATGGCATGCACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCATTTATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAAGGACCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTATCTGCAAATGAAAAGCCTG AGAGCTGAGGACACGGCTGTGTATTACTGTGCGAGAAGGGCCG GTATAATAGGAACTATAGGCTACTACTACGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 388 2G6 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGK GLEWVAFIWYDGSNKYYADSVKDRFTISRDNSKNTLYLQMKSL RAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSS 389 23A10 artificial nt CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTG GGAGGTCCCTGAGACTCTCCTGTGCAGCGTCTGGATTCACCTT CAGTCGCTATGGCATACACTGGGTCCGCCAGGCTCCAGGCAAG GGGCTGGAGTGGGTGGCAGTTATATGGTATGATGGAAGTAATA AATACTATGCAGACTCCGTGAAGGGCCGATTCACCATCTCCAG AGACAATTCCAAGAACACGCTGTATCTGCTAATGAACAGCCTG AGAGCCGAGGACTCGGCTGTGTATTACTGTGCGAGAAGGGCCG GTATACCTGGAACTACGGGCTACTACTATGGTATGGACGTCTG GGGCCAAGGGACCACGGTCACCGTCTCCTCA 390 23A10 artificial aa QVQLVESGGGVVQPGRSLRLSCAASGFTFSRYGIHWVRQAPGK GLEWVAVIWYDGSNKYYADSVKGRFTISRDNSKNTLYLLMNSL RAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSS 391 5E3 artificial nt GAGGTGCAGTTGGTGGAGTCTGGGGGAGGCCTGGTCAAGCCTG GGGGGTCCCTGAGACTCTCCTGTGCAGCCTCTGGATTCACCTT CAGTAGCTATAGCATGCACTGGGTCCGCCAGGCTCCAGGGAAG GGGCTGGAGTGGGTCTCATCCATTAGTAGTAGTAGTAGTTACA TATACTACGCAGACTCAGTGAAGGGCCGATTCACCATCTCCAG AGACAACGCCAAGAACTCACTGTATCTGCAAATGAACAGCCTG AGAGCCGAGGACACGGCTGTGTATTACTGTGCGAGAGGGGAAA CTGGAACTAACTACTACTACTACGGTATGGACGTCTGGGGCCA AGGGACCACGGTCACCGTCTCCTCA 392 5E3 artificial aa EVQLVESGGGLVKPGGSLRLSCAASGFTFSSYSMHWVRQAPGK GLEWVSSISSSSSYIYYADSVKGRFTISRDNAKNSLYLQMNSL RAEDTAVYYCARGETGTNYYYYGMDVWGQGTTVTVSS
TABLE-US-00011 TABLE IIB Light Chain Variable Region Polynucleotide and Amino acid Sequences SEQ ID NO. DESIGNATION SOURCE TYPE SEQUENCE 393 17H8 artificial nt GACATTGTATTGACGCAGtctCCAGGCACCCTGTCTTTGTCTC 23B6 CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCAGAGTGT 28D10 TGCCGGCAGCTACCTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTCTGGTGCATCCAGCAGGGCCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGACTT CACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTG TATTACTGTCAGCAGTATGGTAAATCACCGATCACCTTCGGCC AAGGGACACGACTGGAGATGAAAGGA 394 17H8 artificial aa DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLAWYQQKPGQ 23B6 APRLLISGASSRATGIPDRFSGSGSGTDFTLTISRLEPEDFAV 28D10 YYCQQYGKSPITFGQGTRLEMKG 395 4A2 artificial nt GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC 5B4 CAGGGGAAAGAGCCACCCTCTCTTGCAGGGCCAGTCGGAATAT 5C5 TAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTATGGTCCATCCAGCAGGGCCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGACTT CACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTACAGTG TATTACTGTCAGCAGTATGGTAGCTCATTCACTTTCGGCCCTG GGACCAAAGTGGATATCAAACGA 396 4A2 artificial aa EIVLTQSPGTLSLSPGERATLSCRASRNISSSYLAWYQQKPGQ 5B4 APRLLIYGPSSRATGIPDRFSGSGSGTDFTLTISRLEPEDFTV 5C5 YYCQQYGSSFTFGPGTKVDIKR 397 16H2 artificial nt CAGTCTGCGCTGACTCAGCCACCCTCAGCGACTGGGACCCCCG 20D3 GGCAGAGGGTCACCATCTCTTGTTCTGGAAGCAGCTCCAACAT 23E7 CGGAAGTAATTTTGTAAACTGGTACAAACAACTCCCAGGAACG GCCCCCAAAGTCCTCATCTATACTAATAATCAGCGGCCCTCAG GGGTCCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTCAGC CTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGAT TATTACTGTGCAACATGGGATGACAGCCTGAATGGTTGGGTGT TCGGCGGAGGGACCAAGCTGACCGTCCTAGGT 398 16H2 artificial aa QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGT 20D3 APKVLIYTNNQRPSGVPDRFSGSKSGTSASLAISGLQSEDESD 23E7 YYCATWDDSLNGWVFGGGTKLTVLG 399 26F12 artificial nt CAGTCTGTGCTGACTCAGTCACCCTCAGCGTCTGGGACCCCCG 27B3 GGCAGAAGGTCACCATCTCTTGTTCTGGAAGCCGCTCCAACAT CGGAAGTAATTTTGTAAACTGGTACCAGCAGCTCCCAGGAACG GCCCCCAAACTCCTCATCTATACTAATTATCAGCGGCCCTCAG GGGTCCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTCAGC CTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGGCTGAT TATTACTGTGCAGTATGGGATGACAGCCTGAATGGTTGGGTGT TCGGCGGAGGGACCAAGCTGACCGTCCTAGGT 400 26F12 artificial aa QSVLTQSPSASGTPGQKVTISCSGSRSNIGSNFVNWYQQLPGT 27B3 APKLLIYTNYQRPSGVPDRFSGSKSGTSASLAISGLQSEDEAD YYCAVWDDSLNGWVFGGGTKLTVLG 401 4B10 artificial nt GAAATTGTATTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC 4C2 CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCAGAGTGT TAGCAACACCTACTTAGCCTGGTACCATCAGAGACCTGGCCAG GCTCCCAGGCTCCTCATCTATGGTGCATCCAGCAGGGCCACTG GCATCCCAGACAGATTCAGTGGCAGTGGGTCTGGGACAGACTT CGCTCTCACCATCAGCAGTCTGGAGCCTGAAGATTTTGCAGTG TATTACTGTCAGCAGTACAGTAACTCgtgGACGTTCGGCCAAG GGACCAAGGTGGAAATCAaacGA 402 4B10 artificial aa EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLAWYHQRPGQ 4C2 APRLLIYGASSRATGIPDRFSGSGSGTDFALTISSLEPEDFAV YYCQQYSNSWTFGQGTKVEIKR 403 4D3 artificial nt GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC 4F3 CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCAGAGTGT TAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTATGGTGCATCCAGCAGGGCCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGACTT CACTCTCACCATCAGCAGACTGGAACCTGAGGATTTTGCAGTG TATTACTGTCAGCAGTATGGTAGCTCGTGGACGTTCGGCCAAG GGACCAAGGTGGAAATCAAACGA 404 4D3 artificial aa EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQ 4F3 APRLLIYGASSRATGIPDRFSGSGSGTDFTLTISRLEPEDFAV YYCQQYGSSWTFGQGTKVEIKR 405 16E2 artificial nt GACATCCAGATGACCCAGTCTCCATCCTCACTGTCTGCATCTG 17E10 TAGGAGACAGAGTCACCATCACTTGTCGGGCGAGTCAGGGCAT 20B12 TAGCAATTATTTAGCCTGGTTACAGCAGAAACCAGGGAAAGCC CCTAAGTCCCTGATCTATGCTGCATCCAGTTTGCAAAGTGGGG TCCCATCAAAGTTCAGCGGCAGTGGATCTGGGACAGATTTCAC TCTCACCATCAGCAGCCTGCAGCCTGAAGATTTTGCAACTTAT TACTGCCAACACTATTTTACTTACCCTCGGACGTTCGGCCAAG GGACCAAGGTGGAAATCAAACGA 406 16E2 artificial aa DIQMTQSPSSLSASVGDRVTITCRASQGISNYLAWLQQKPGKA 17E10 PKSLIYAASSLQSGVPSKFSGSGSGTDFTLTISSLQPEDFATY 20B12 YCQHYFTYPRTFGQGTKVEIKR 407 1D10 artificial nt TCCTATGCGCTGACTCAGCCACCCTCAGTGTCCGTGTCCCCAG 2C12 GACAGACAGCCAGCCTCACCTGCTCTGGAGATAGATTGGGGGA AAAATATACTTGCTGGTATCAGCAGAGGCCAGGCCAGTCCCCT TTGCTGGTCATCTATCAAGATACCAAGCGGCCCTCAGGGATCC CTGAGCGATTCTCTGGCTCCACCTCTGGTAACACAGCCACTCT GACCATCAGCGGGACCCAGGCTATGGATGAGGCTGACTATTAC TGTCAGGCGTGGGACAGCAGCACTGTGGTATTCGGCGGAGGGA CCAAGCTGACCGTCCTAGGT 408 1D10 artificial aa SYALTQPPSVSVSPGQTASLTCSGDRLGEKYTCWYQQRPGQSP 2C12 LLVIYQDTKRPSGIPERESGSTSGNTATLTISGTQAMDEADYY CQAWDSSTVVFGGGTKLTVLG 409 16C1 artificial nt GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGCCAGAGTGT TAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTTTGGTGCATCCAGCAGGGCCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGACTT CACTCTCACCATCAGCGGACTGGAGCCTGAAGATTTTGCAGTG TATCACTGTCAGCAGTATGGTAACTCACCGCTCACTTTCGGCG GAGGGACCAAGGTGGAGATCAAACGA 410 16C1 artificial aa EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQ APRLLIFGASSRATGIPDRFSGSGSGTDFTLTISGLEPEDFAV YHCQQYGNSPLTFGGGTKVEIKR 411 25G10 artificial nt GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCAGAGTGT TAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTTTGGTGCATCCAGCAGGGCCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGactT CACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTG TATCACTGTCAGCAGTATGGTAACTCACCGCTCACTTTCGGCG GAGGGACCAAGGTGGAGATCAAACGA 412 25G10 artificial aa EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQ APRLLIFGASSRATGIPDRFSGSGSGTDFTLTISRLEPEDFAV YHCQQYGNSPLTFGGGTKVEIKR 413 16A4 artificial nt GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC CAGGGGAAAGAGCCACCCtCTCCTGCAGGGCCAGTCAGAGTGT TAGCAGCAGTTATTTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTATGGTACATCCAGCAGGGCCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGACTT CACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTG TATTATTGTCAGCAGTACGGTAGCTCACCTTTCACTTTCGGCG GAGGGACCAAGGTGGAGATCAAACGA 414 16A4 artificial aa EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQ APRLLIYGTSSRATGIPDRFSGSGSGTDFTLTISRLEPEDFAV YYCQQYGSSPFTFGGGTKVEIKR 415 1F10 artificial nt GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCGGAGTAT TAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTATGGTCCATCCAGCAGGGCCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGACTT CACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTG TATTACTGTCAGCAGTATGGTAGCTCATTCACTTTCGGCCCTG GGACCAAAGTGGATATCAAACGA 416 1F10 artificial aa EIVLTQSPGTLSLSPGERATLSCRASRSISSSYLAWYQQKPGQ APRLLIYGPSSRATGIPDRFSGSGSGTDFTLTISRLEPEDFAV YYCQQYGSSETFGPGTKVDIKR 417 4A9 artificial nt CAGTCTGTGCTGACGCAGCCGCCCTCAGTGTCTGGGGCCCCAG GACAGAGGGTCACCATCTCCTGCACTGGGAGCAGCTCCAACAT CGGGACAGGTTATGCTGTACACTGGTACCAGCAGTTTCCAGGA ACAGCCCCCAAACTCCTCATCTATGGTAACAACAATCGGCCCT CAGGGGTTCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTC AGCCTCCCTGGCCATCACTGGGCTCCAGGCTGAGGATGAGGCT GATTATTACTGCCAGTCCTATGACAGCAGACTGAGTGGTTGGG TGTTCGGCGGAGGGACCAAGCTGACCGTCCTAGGT 418 4A9 artificial aa QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAVHWYQQFPG TAPKLLIYGNNNRPSGVPDRFSGSKSGTSASLAITGLQAEDEA DYYCQSYDSRLSGWVFGGGTKLTVLG 419 4F7 artificial nt CAGTCTGTgcTGACGCAGCCGCCCTCAGTGTCTGGGGCCCCAG GGCAGAGGGTCACCATCTCCTGCACTGGGAGCAGCTCCAATAT CGGGACAGGTTATGATGTACACTGGTATCAGCAGcttcCAGGA ACAGCCCCCAAACTCCTCATCCATGGTAACAGCAATCGGCCCT CAGGGGTCCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTC AGCCTCCCTGGCCATCACTGGGCTCCAGGCTGAGGATGAGGCT GATTATTACTGCCAGTCCTATGACAGCAGTCTGAGTGGTTGGG TGTTCGGCGGAGGGACCAGGTTGACCGTCCTAGGT 420 4F7 artificial aa QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDVHWYQQLPG TAPKLLIHGNSNRPSGVPDRFSGSKSGTSASLAITGLQAEDEA DYYCQSYDSSLSGWVFGGGTRLTVLG 421 22D1 artificial nt CAGTCTGCGCTGACTCAGCCACCCTCAGCGACTGGGACCCCCG GGCAGAGGGTCACCATCTCTTGTTCTGGAAGCAGCTCCAACAT CGGAAGCAATTTTGTAAACTGGTACAAGCAGCTCCCAGGAACG GCCCCCAAAGTCCTCATCTATACTAATAATCAGCGGCCCTCAG GGGTCCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTCAGC CTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGAT TATTACTGTGCAACATGGGATGACAGTATGAATGGTTGGGTGT TCGGCGGAGGGACCAAGCTGACCGTCCTAGGT 422 22D1 artificial aa QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGT APKVLIYTNNQRPSGVPDRFSGSKSGTSASLAISGLQSEDESD YYCATWDDSMNGWVFGGGTKLTVLG 423 19B5 artificial nt CAGTCTGCGCTGACTCAGCCACCCTCAACGACTGGGACCCCCG GGCAGAGGGTCACCATCTCTTGTTCTGGAAGCAGGTCCAACAT CGGAAGCAATTTTGTAAACTGGTACAAGCAGCTCCCAGGAACG GCCCCCAAAGTCCTCATCTATACTAATAATCAGCGGCCCTCAG GGGTCCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTCAGC CTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGAT TATTACTGCGCAACATGGGATGACAGTATGAATGGTTGGGTGT TCGGCGGAGGGACCAAACTGACCGTCCTAGGT 424 19B5 artificial aa QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNFVNWYKQLPGT APKVLIYTNNQRPSGVPDRFSGSKSGTSASLAISGLQSEDESD YYCATWDDSMNGWVFGGGTKLTVLG 425 25F8 artificial nt CAGTCTGCGCTGactCAGCCACCCTCAGCGACTGGGACCCCCG GGCAGAGGGTCACCATCTCTTGTTCTGGAAGCAGCTCCAACAT CGGAAGGAATTTTGTAAACTGGTATAAGCAGCTCCCAGGAACG GCCCCCAAAGTCCTCATTTATACTAATAATCAGCGGCCCTCAG GGGTCCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTCAGC CTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGAT TATTACTGTGCAGCATGGGATGACAGCCTGAATGGTTGGGTGT TCGGCGGAGGGACCAAGCTGACCGTCCTAGGT 426 25F8 artificial aa QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYKQLPGT APKVLIYTNNQRPSGVPDRFSGSKSGTSASLAISGLQSEDESD YYCAAWDDSLNGWVFGGGTKLTVLG 427 26D1 artificial nt CACTCTGTGCTGACTCAGTCACCCTCAGCGTCTGGGACCCCCG GACAGAGGGTCACCATCTCTTGTTCTGGAAGCCGCTCCAACAT CGGAAGTAATTTTGTAAACTGGTACCAGCAGCTCCCAGGAACG GCCCCCAAACTCCTCATCTATACTAATAATCAGCGGCCCTCAG GGGTCCCTGACCGATTCTCTGGCTCCAAGTCTGGCACCTCAGC CTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGGCTGAT TATTACTGTGCAGTATGGGATGACAGCCTGAATGGTTGGGTGT TCGGCGGAGGGACCAAGCTGACCGTCCTAGGT 428 26D1 artificial aa HSVLTQSPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGT APKLLIYTNNQRPSGVPDRFSGSKSGTSASLAISGLQSEDEAD YYCAVWDDSLNGWVFGGGTKLTVLG 429 4D2 artificial nt GAAATTGTATTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCAGAGTGT TAGCAACACCTACTTAGCCTGGTACCATCAGAGACCTGGCCAG GCTCCCAGGCTCCTCATCTATGGTGCATCCAGCAGGGCCGCTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGACTT CACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTG TATTACTGTCAGCAGTATAGTAACTCGTGGACGTTCGGCCAAG GGACCAAGGTGGAAATCAAACGA
430 4D2 artificial aa EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLAWYHQRPGQ APRLLIYGASSRAAGIPDRFSGSGSGTDFTLTISRLEPEDFAV YYCQQYSNSWTFGQGTKVEIKR 431 4E10 artificial nt GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTC CAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCAGAGTGT TGGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAG GCTCCCAGGCTCCTCATCTATGGTGCATCCAGCAGGGTCACTG GCATCCCAGACAGGTTCAGTGGCAGTGGGTCTGGGACAGATTT CACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTG TATTACTGTCAGCAATATAGTAACTCGTGGACGTTCGGCCAAG GGACCAAGGGGAAATCAAACGA 432 4E10 artificial aa EIVLTQSPGTLSLSPGERATLSCRASQSVGSSYLAWYQQKPGQ APRLLIYGASSRVTGIPDRFSGSGSGTDFTLTISRLEPEDFAV YYCQQYSNSWTFGQGTKVEIKR 433 22G10 artificial nt GAAATAGTGATGACGCAGTCTCCAGTCACCCTGTCTCTGTCTC TAGGGGAAAGAGCCACCCTCTCCTGCAGGGCCAGTCAGAGTAT TAGCAGCAACTTAGCCTGGTTCCAGCAGAAACCTGGCCAGGCT CCCAGACTCCTCATCTATGGTGCATTTACCAGGGCCACTGGTA TCCCAGCCAGGGTCAGTGGCAGTGGGTCTGGGACAGAGTTCAC TCTCACCATCAGCAGCCTGCAGTCTGAAGATTTTGCAGTTTAT TACTGTCAGCAGTATAATTACTGGCCGCTCACTTTCGGCGGAG GGACCAAGGTGGAGATCAAGCGA 434 22G10 artificial aa EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQA PRLLIYGAFTRATGIPARVSGSGSGTEFTLTISSLQSEDFAVY YCQQYNYWPLTFGGGTKVEIKR 435 2C12_LC#1 artificial nt GATGTTGTGATGactCAGtCTccActctccctgcCCGTCACCC TTGGACAGCCGGcctCCAtctcctgCAGGtCTAGTCAAAGcct cgtaTACAGTGATGGAAACAcctACTTGAATTGGTTTCAGCAG AGGCCAGGCCAATCTCCAAGGcgcctaATTTATAAGGTTTCTA ACTGGGactctGGGGtCCCAGACAGATTCAGCgGCAGTGGGTC AGGCActGATTTCACactGAAAAtCAGCAGGGTGGaggctgaG GATGTTGGGGTTTATTactgCATGCAAGGTATAGTGTGGCCGT GCAGTTTTGGCCAGGGGACCAAGCTGGAGATCAAaCgA 436 2C12_LC#1 artificial aa DVVMTQSPLSLPVTLGQPASISCRSSQSLVYSDGNTYLNWFQQ RPGQSPRRLIYKVSNWDSGVPDRFSGSGSGTDFTLKISRVEAE DVGVYYCMQGIVWPCSFGQGTKLEIKR 437 2H12_LC#2 artificial nt GATGTTGTGATGACTCAGTCTCCACTCTCCCTGCCCGTCACCC TTGGACAGCCGGCCTCCATCTCCTGCAGGTCTAGTCAAAGCCT CGTATACAGTGATGGAAACACCTACTTGAATTGGTTTCAGCAG AGGCCAGGCCAATCTCCAAGGCGCCTAATTTATAAGGTTTCTA ACTGGGACTCTGGGGTCCCAGACAGAATCAGCGGCAGTGGGTC AGGCACCGATTTCACACTGAAAATCAGCAGGGTGGAGGCTGAG GATGTTGGGGTTTATTACTGCATGCAAGATACACTGTGGCCGT GCAGTTTTGGCCAGGGGACCAAGCTGGAGATCAAACGA 438 2H12_LC#2 artificial aa DVVMTQSPLSLPVTLGQPASISCRSSQSLVYSDGNTYLNWFQQ RPGQSPRRLIYKVSNWDSGVPDRISGSGSGTDFTLKISRVEAE DVGVYYCMQDTLWPCSFGQGTKLEIKR 439 2G6_LC#1 artificial nt GaTGTTGTGATGACTCagtctccACTCTCCCTGCCCGTCACCC ttggacaGCCGGCCTccaTCTCCTGCAGGTCTAGTCAAAGCCT CGTATACAGTGATGGAAACACCTACTTGAATTGGTTTCAGCAG AGGCCAGGCCAATCTCCACGGCGCCTAATTTATCAGGTTTCTA ACTGGGACTCTGGGGTCCCAGACAGATTCAGCGGCAGTGGGTC AGGCACTGATTTCACACTGAAAATCAGCAGGGTGGAGGCTGAG GATGTTGGGATTTATTACTGCATGCAAGATACACTGTGGCCGT GCAGTTTTGGCCAGGGGACCAAGCTGGAGATCAAACGA 440 2G6_LC#1 artificial aa DVVMTQSPLSLPVTLGQPASISCRSSQSLVYSDGNTYLNWFQQ RPGQSPRRLIYQVSNWDSGVPDRFSGSGSGTDFTLKISRVEAE DVGIYYCMQDTLWPCSFGQGTKLEIKR 441 2H12 artificial nt TCCTATGAGCTGACTCAGCCACCCTCAGTGTCCGTGTCCCCAG GACAGACAGCCAGCATCACCTGCTCTGGAGATAGATTGGGGGA AAAATATACTTGCTGGTATCAGCAGAGGCCAGGCCAGTCCCCT TTGCTGGTCATCTATCAAGATACCAAGCGGCCCTCAGGGATCC CTGAGCGATTCTCTGGCTCCAACTCTGGTAACACAGCCACTCT GACCATCAGCGGGACCCAGCCTATGGATGAGGCTGACTATTAC TGTCAGGCGTGGGACAGCAGCACTGTGGTATTCGGCGGAGGGA CCAAGCTGACCGTCCtAGGT 442 2H12 artificial aa SYELTQPPSVSVSPGQTASITCSGDRLGEKYTCWYQQRPGQSP LLVIYQDTKRPSGIPERFSGSNSGNTATLTISGTQPMDEADYY CQAWDSSTVVFGGGTKLTVLG 443 2G6 artificial nt TCCTATGAACTGACTCAGCCACCCTCAGTGTCCGTGTCCCCAG GACAGACAGCCAGCATCACCTGCTCTGGAGATAGGTTGGGGGA AAAATATACTTGCTGGTATCAGCAGAGGCCAGGCCAGTCCCCT TTGCTGGTCATCTATCAAGATACCAAGCGGCCCTCAGGGATCC CTGAGCGATTCTCTGGCTCCAACTCTGGTAACACAGCCACTCT GACCATCAGCGGGACCCAGGCTATGGATGAGGCTGACTATTAC TGTCAGGCGTGGGACAGCAGCACTGTGGTATTCGGCGGAGGGA CCAAGCTGACCGTCCTAGGT 444 2G6 artificial aa SYELTQPPSVSVSPGQTASITCSGDRLGEKYTCWYQQRPGQSP LLVIYQDTKRPSGIPERFSGSNSGNTATLTISGTQAMDEADYY CQAWDSSTVVFGGGTKLTVLG 445 23A10 artificial nt TCCTATGAGCTGACTCAGCCACCCTCAGTGTCCGTGTCCCCAG GACAGACAGCCAGCATCACCTGCTCTGGAGATAGATTGGGGGA GAAATATGTTTGCTGGTATCAGCAGAAGCCAGGCCAGTCCCCT ATACTGGTCATCTATCAAGATAATAAGTGGCCCTCAGGGATCC CTGAGCGATTCTCTGGCTCCAACTCTGGGAACACAGCCACTCT GACCATCAGCGGGACCCAGGCTATGGATGAGGCTGACTATTAC TGTCAGGCGTGGGACAGCAGCACTGTGGTATTCGGCGGGGGGA CCAAGCTGACCGTCCTAGGT 446 23A10 artificial aa SYELTQPPSVSVSPGQTASITCSGDRLGEKYVCWYQQKPGQSP ILVIYQDNKWPSGIPERFSGSNSGNTATLTISGTQAMDEADYY CQAWDSSTVVFGGGTKLTVLG 447 5E3 artificial nt TCCTATGAGCTGACTCAGCCACCCTCAGTGTCCGTGTCCCCAG GACAGACAGCCAGCATCACCTGCTCTGGAGATAAATTGGGGGA TGAATATGCTTGCTGGTATCAGCAGAAGCCAGGCCAGTCCCCT GTGCTGGTCATCTATCAAGATAGCAAGCGGCCCTCAGGGATCC CTGAGCGATTCTCTGGCTCCAACTCTGGGAACACAGCCACTCT GACCATCAGCGGGACCCAGGCTATGGATGAGGCTGACTATTAC TGTCAGGCGTGGGACAGCAGCACTGTGGTATTCGGCGGAGGGA CCAAGCTGACCGTCCTAGGT 448 5E3 artificial aa SYELTQPPSVSVSPGQTASITCSGDKLGDEYACWYQQKPGQSP VIVIYQDSKRPSGIPERFSGSNSGNTATLTISGTQAMDEADYY CQAWDSSTVVFGGGTKLTVLG
TABLE-US-00012 TABLE IIc Heavy Chain Variable Region Polynucleotide and Amino acid Sequences 13586 HC [hu anti-<huCDH19> 4F3 VH] QVQLVESGGGVVQPGRSLRLSCAASGFSFSSYDMDWVRQTPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSICNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGRGTLVTVSS SEQ ID NO: 449 13589 HC [hu anti-<huCDH19> 4A9 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWFAYFSYSGSTNYNPSLKSRVTLS VDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSS SEQ ID NO: 450 13590 HC [hu anti-<huCDH19> 4B10 VH] QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYDGTNEYYADSVKGR FTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSS SEQ ID NO: 451 13874 HC [hu anti-<huCDH19> 17118.2 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTISV DTSKNQFSLKLSSVTAADTALYYCARDSRYRSGWYDAFDIWGQGTMVTVSS SEQ ID NO: 452 13875 HC [hu anti-<huCDH19> 16C1.1 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS IDTSKNQFSLTLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSS SEQ ID NO: 453 13876 HC [hu anti-<huCDH19> 16A4.1 VH] QVQLQESGPGLAKPSETLSLTCTVSGDSITSYYWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSRVTISV DTSKNQFSLKLSSVTAADTAVYYCARDQRRIAAAGTHFYGMDVWGQGTTVTVSS SEQ ID NO: 454 13877 HC [hu anti-<huCDH19> 22G10.1 VH] EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKGR FTISSDNSKSTLYLQMNSLRAADTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSS SEQ ID NO: 455 13878 HC [hu anti-<huCDH19> 20D3.1 VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 456 13879 HC [hu anti-<huCDH19> 22D1.1 VH] QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 457 13880 HC [hu anti-<huCDH19> 25F8.1 VH] QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQGR VTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 458 13881 HC [hu anti-<huCDH19> 26F12.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 459 13882 HC [hu anti-<huCDH19> 26D1.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQGR VTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSS SEQ ID NO: 460 13883 HC [hu anti-<huCDH19> 25G10.1 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS VDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYRWFDPWGQGTLVTVSS SEQ ID NO: 461 13885 HC [hu anti-<huCDH19> 19B5.1 VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 462 14022 HC [hu anti-<huCDH19> 4A2 VH] QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHPGKGLEWICIYIYYTGSAYYNPSLKSRV TISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSS SEQ ID NO: 463 14024 HC [hu anti-<huCDH19> 4A2 (1-472)(Q7E, H47P) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLKSRVT ISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSS SEQ ID NO: 464 14025 HC [hu anti-<huCDH19> 4A2 VH] QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHPGKGLEWIGYIYYTGSAYYNPSLKSRV TISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSS SEQ ID NO: 465 14026 HC [hu anti-<huCDH19> 4A2 (1-472)(Q17E, H47P) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLKSRVT ISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSS SEQ ID NO: 466 14027 HC [hu anti-<huCDH19> 4A2 (1-472)(Q17E, H47P, D111E) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLKSRVT ISVDTSKNQFSLKLSSVTAADTAVYYCAREGSSGWYFQYWGQGTLVTVSS SEQ ID NO: 467 14028 HC [hu anti-<huCDH19> 4A2 (1-472)(Q17E, H47P, D111E, W134Y) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLKSRVT ISVDTSKNQFSLKLSSVTAADTAVYYCAREGSSGYYFQYWGQGTLVTVSS SEQ ID NO: 468 14029 HC [hu anti-<huCDH19> 4A2 VH] QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHPGKGLEWIGYIYYTGSAYYNPSLKSRV TISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSS SEQ ID NO: 469 14030 HC [hu anti-<huCDH19> 4F3 (1-471)(R17G) VH] QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQTPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGRGTLVTVSS SEQ ID NO: 470 14031 HC [hu anti-<huCDH19> 4F3 (1-471)(R17G, T47A) VH] QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSICNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGRGTLVTVSS SEQ ID NO: 471 14032 HC [hu anti-<huCDH19> 4F3 (1-471)(R17G, T47A, R141Q) VH] QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGQGTLVTVSS SEQ ID NO: 472 14033 HC [hu Anti-<huCDH19> 4F3 (1-471)(R17G, T47A, D61E, D72E, R141Q) VH] QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYEGSNKYYAESVRG RFTISRDNSICNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGQGTLVTVSS SEQ ID NO: 473 14034 HC [hu anti-<huCDH19> 4F3 (1-471)(R17G, T47A, D61E, D72E, W134Y, R141Q) VH] QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYEGSNKYYAESVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGYYFDLWGQGTLVTVSS SEQ ID NO: 474 14039 HC [hu anti-<huCDH19> 2G6 (1-477)(R17G, D61E, D72E, K94N) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYEGSNKYYAESVKD RFTISRDNSKNTLYLQMNSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSS SEQ ID NO: 475 14040 HC [hu anti-<huCDH19> 16C1.1 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS IDTSKNQFSLTLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSS SEQ ID NO: 476 14041 HC [hu anti-<huCDH19> 16C1.1 (1-469)(T92K) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISCiYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS IDTSKNQFSLKLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSS SEQ ID NO: 477 14042 HC [hu anti-<huCDH19> 16C1.1 (1-469)(T92K, D109E) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISCiYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS IDTSKNQFSLKLSSLTAADTAVYFCAREGSSGWYRWFDPWGQGTLVTVSS SEQ ID NO: 478 14043 HC [hu anti-<huCDH19> 16C1.1 (1-469)(T92K, W132Y, W135Y) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISCiYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS IDTSKNQFSLKLSSLTAADTAVYFCARDGSSGYYRYFDPWGQGTLVTVSS SEQ ID NO: 479 14044 HC [hu anti-<huCDH19> 16C1.1 (1-469)(T92K) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISCiYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS IDTSKNQFSLKLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSS SEQ ID NO: 480 14045 HC [hu anti-<huCDH19> 17118.2 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTISV DTSKNQFSLKLSSVTAADTALYYCARDSRYRSGWYDAFDIWGQGTMVTVSS SEQ ID NO: 481 14046 HC [hu anti-<huCDH19> 17118.2 (1-471)(D109E) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTISV DTSKNQFSLKLSSVTAADTALYYCARESRYRSGWYDAFDIWGQGTMVTVSS SEQ ID NO: 482 14047 HC [hu anti-<huCDH19> 17118.2 (1-471)(D109E, W132Y) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTISV DTSKNQFSLKLSSVTAADTALYYCARESRYRSGYYDAFDIWGQGTMVTVSS SEQ ID NO: 483 14048 HC [hu anti-<huCDH19> 17118.2 (1-471)(D109E) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTISV DTSKNQFSLKLSSVTAADTALYYCARESRYRSGWYDAFDIWGQGTMVTVSS SEQ ID NO: 484 14049 HC [hu anti-<huCDH19> 4F7 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSRVTISL DTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDYWGQGTLVTVSS SEQ ID NO: 485 14050 HC [hu anti-<huCDH19> 4F7 VVH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSRVTISL DTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDYWGQGTLVTVSS SEQ ID NO: 486 14051 HC [hu anti-<huCDH19> 4F7 (1-468)(W113Y) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSRVTISL DTSKNQFSLKLSSVTAADTAVYYCARNYAFHFDYWGQGTLVTVSS SEQ ID NO: 487 14052 HC [hu anti-<huCDH19> 4B10 (1-471)(R17G, D61E, D72E, W134Y) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYEGTNEYYAESVKGR FTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDYSFDYWCiQGTLVSVSS SEQ ID NO: 488 14053 HC [hu anti-<huCDH19> 4B10 VH] QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYDGTNEYYADSVKGR FTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSS SEQ ID NO: 489 14054 HC [hu anti-<huCDH19> 4B10 (1-471)(R17G) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYDGTNEYYADSVKG RFTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSS SEQ ID NO: 490 14055 HC [hu anti-<huCDH19> 4B10 (1-471)(R17G, D61E, D72E) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYEGTNEYYAESVKGR FTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSS SEQ ID NO: 491 14056 HC [hu anti-<huCDH19> 4A9 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWFAYFSYSGSTNYNPSLKSRVTLS VDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSS SEQ ID NO: 492 14057 HC [hu anti-<huCDH19> 4A9 (1-468)(F55I, A56G) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYFSYSGSTNYNPSLKSRVTLS VDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSS SEQ ID NO: 493 14058 HC [hu anti-<huCDH19> 4A9 (1-468)(F55I, A56G) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYFSYSGSTNYNPSLKSRVTLS VDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSS SEQ ID NO: 494 14059 HC [hu anti-<huCDH19> 4A9 (1-468)(F55I, A56G, W113Y) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYFSYSGSTNYNPSLKSRVTLS VDTSKNQFSLKLSSVTAADTAVYYCARNYAFHFDFWGQGTLVTVSS SEQ ID NO: 495 14060 HC [hu anti-<huCDH19> 20D3.1 VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 496
14061 HC [hu anti-<huCDH19> 20D3.1 VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 497 14062 HC [hu anti-<huCDH19> 20D3.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSS SEQ ID NO: 498 14063 HC [hu anti-<huCDH19> 20D3.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSS SEQ ID NO: 499 14064 HC [hu anti-<huCDH19> 20D3.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSS SEQ ID NO: 500 14065 HC [hu anti-<huCDH19> 22G10.1 (1-470)(S82R, A99E) VH] EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKGR FTISRDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSS SEQ ID NO: 501 14066 HC [hu anti-<huCDH19> 22G10.1 (1-470)(A99E, H105Y) VH] EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKGR FTISSDNSKSTLYLQMNSLRAEDTAVYYCAKGGMGGYYYCiMDVWGQGTTVTVSS SEQ ID NO: 502 14067 HC [hu anti-<huCDH19> 22G10.1 (1-470)(A99E) VH] EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKGR FTISSDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSS SEQ ID NO: 503 14068 HC [hu anti-<huCDH19> 22G10.1 (1-470)(A99E) VH] EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKGR FTISSDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSS SEQ ID NO: 504 14069 HC [hu anti-<huCDH19> 22G10.1 (1-470)(D72E, A99E) VH] EVQLLESGGGLVQPGGSLRLSCAASGFTFSYAMNWVRQAPGKGLEWVSTISGGGANTYYAESVKGRF TISSDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSS SEQ ID NO: 505 14070 HC [hu anti-<huCDH19> 22G10.1 (1-470)(H105Y) VH] EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKGR FTISSDNSKSTLYLQMNSLRAADTAVYYCAKGGMGGYYYGMDVWGQGTTVTVSS SEQ ID NO: 506 14071 HC [hu anti-<huCDH19> 16A4.1 (1-474)(T144L) VH] QVQLQESGPGLAKPSETLSLTCTVSGDSITSYYWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSRVTISV DTSKNQFSLKLSSVTAADTAVYYCARDQRRIAAAGTHFYGMDVWGQGTLVTVSS SEQ ID NO: 507 14072 HC [hu anti-<huCDH19> 19B5.1 VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 508 14073 HC [hu anti-<huCDH19> 19B5.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSS SEQ ID NO: 509 14074 HC [hu anti-<huCDH19> 19B5.1 VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 510 14075 HC [hu anti-<huCDH19> 19B5.1 VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELS SLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 511 14076 HC [hu anti-<huCDH19> 19B5.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELS SLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSS SEQ ID NO: 512 14077 HC [hu anti-<huCDH19> 23A10.3 (1-474)(L92Q) VH] QVQLVESGGGVVQPGRSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYDGSNKYYADSVKGR FTISRDNSKNTLYLQMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSS SEQ ID NO: 513 14078 HC [hu anti-<huCDH19> 23A10.3 (1-474)(R17G, L92Q) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYDGSNKYYADSVKG RFTISRDNSKNTLYLQMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSS SEQ ID NO: 514 14079 HC [hu anti-<huCDH19> 23A10.3 (1-474)(R17G, D61E, D72E, L92Q) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYEGSNKYYAESVKGR FTISRDNSKNTLYLQMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSS SEQ ID NO: 515 14080 HC [hu anti-<huCDH19> 23A10.3 VH] QVQLVESGGGVVQPGRSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYDGSNKYYADSVKGR FTISRDNSKNTLYLLMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSS SEQ ID NO: 516 14081 HC [hu anti-<huCDH19> 25G10.1 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS VDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYRWFDPWGQGTLVTVSS SEQ ID NO: 517 14082 HC [hu anti-<huCDH19> 25G10.1 (1-469)(D109E, W132Y, W135Y) VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSRVTMS VDTSKNQFSLKLSSVTAADTAVYYCAREGSSGYYRYFDPWGQGTLVTVSS SEQ ID NO: 518 14083 HC [hu anti-<huCDH19> 26D1.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQGR VTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSS SEQ ID NO: 519 14084 HC [hu anti-<huCDH19> 26D1.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQGR VTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSS SEQ ID NO: 520 14085 HC [hu anti-<huCDH19> 26D1.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQGR VTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSS SEQ ID NO: 521 14086 HC [hu anti-<huCDH19> 26D1.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQGR VTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSS SEQ ID NO: 522 14087 HC [hu anti-<huCDH19> 26D1.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQGR VTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLYLHFDYWGQGTLVTVSS SEQ ID NO: 523 14088 HC [hu anti-<huCDH19> 26D1.1 (1-469)(R27G, G82R) VH] QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQGR VTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSS SEQ ID NO: 524 14089 HC [hu anti-<huCDH19> 26F12.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 525 14090 HC [hu anti-<huCDH19> 26F12.1 VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 526 14091 HC [hu anti-<huCDH19> 26F12.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSS SEQ ID NO: 527 14092 HC [hu anti-<huCDH19> 26F12.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSS SEQ ID NO: 528 14093 HC [hu anti-<huCDH19> 25F8.1 VH] QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQGR VTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 529 14094 HC [hu anti-<huCDH19> 25F8.1 VH] QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQGR VTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 530 14095 HC [hu anti-<huCDH19> 25F8.1 (1-469)(F90Y) VH] QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQGR VTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 531 14096 HC [hu anti-<huCDH19> 25F8.1 (1-469)(F90Y) VH] QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQGR VTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSS SEQ ID NO: 532 14097 HC [hu anti-<huCDH19> 25F8.1 (1-469)(F90Y, W133Y) VH] QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQGR VTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSS SEQ ID NO: 533 14098 HC [hu anti-<huCDH19> 22D1.1 VH] QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 534 14099 HC [hu anti-<huCDH19> 22D1.1 VH] QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 535 14100 HC [hu anti-<huCDH19> 22D1.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSS SEQ ID NO: 536 14101 HC [hu anti-<huCDH19> 22D1.1 (1-469)(W133Y) VH] QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSS SEQ ID NO: 537 14102 HC [hu anti-<huCDH19> 22D1.1 (1-469)(F90Y) VH] QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQGRV TMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSS SEQ ID NO: 538 13591 HC [hu anti-<huCDH19> 4F7 VH] QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKOLEWIGYIYYSGSTNYNPSLKSRVTISL DTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDYWGQGTLVTVSS SEQ ID NO: 539 14301 HC [hu anti-<huCDH19> 2G6 VH] QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYDGSNKYYADSVKD RFTISRDNSKNTLYLQMKSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSS SEQ ID NO: 540 14302 HC [hu anti-<huCDH19> 2G6 (1-477)(R17G, K94N) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYDGSNKYYADSVKD RFTISRDNSKNTLYLQMNSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSS SEQ ID NO: 541 14303 HC [hu anti-<huCDH19> 2G6 (1-477)(D61E, D72E) VH] QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYEGSNKYYAESVKD RFTISRDNSKNTLYLQMKSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSS SEQ ID NO: 542 14304 HC [hu anti-<huCDH19> 2G6 (1-477)(R17G) VH] QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYDGSNKYYADSVKD RFTISRDNSKNTLYLQMKSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSS SEQ ID NO: 543
TABLE-US-00013 TABLE IId Light Chain Variable Region Amino acid Sequences 13586 LC [hu anti-<huCDH19> 4F3 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKR SEQ ID NO: 544 13589 LC [hu anti-<huCDH19> 4A9 VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAVHWYQQFPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSRLSGWVFGGGTKLTVLG SEQ ID NO: 545 13590 LC [hu anti-<huCDH19> 4B10 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLAWYHQRPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FALTISSLEPEDFAVYYCQQYSNSWTFGQGTKVEIKR SEQ ID NO: 546 13874 LC [hu anti-<huCDH19> 17H8.2 VL] DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLAWYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRLEMKG SEQ ID NO: 547 13875 LC [hu anti-<huCDH19> 16C1.1 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISGLEPEDFAVYHCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 548 13876 LC [hu anti-<huCDH19> 16A4.1 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGTSSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSPFTFGGGTKVEIKR SEQ ID NO: 549 13877 LC [hu anti-<huCDH19> 22G10.1 VL] EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTEF TLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVEIKR SEQ ID NO: 552 13878 LC [hu anti-<huCDH19> 20D3.1 VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDESDYYCATWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 554 13879 LC [hu anti-<huCDH19> 22D1.1 VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDESDYYCATWDDSMNGWVFGGGTKLTVLG SEQ ID NO: 555 13880 LC [hu anti-<huCDH19> 25F8.1 VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCAAWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 556 13881 LC [hu anti-<huCDH19> 26F12.1 VL] QSVLTQSPSASGTPGQKVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 557 13882 LC [hu anti-<huCDH19> 26D1.1 VL] HSVLTQSPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 555 13883 LC [hu anti-<huCDH19> 25G10.1 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYHCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 556 13885 LC [hu anti-<huCDH19> 19B5.1 VL] QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDESDYYCATWDDSMNGWVFGGGTKLTVLG SEQ ID NO: 557 14022 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q) VL] EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDF TLTISRLEPEDFTVYYCQQYGSSFTFGPGTKVDIKR SEQ ID NO: 558 14024 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q,T102A,P141Q) VL] EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDF TLTISRLEPEDFAVYYCQQYGSSFTFGQGTKVDIKR SEQ ID NO: 559 14025 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A) VL] EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDF TLTISRLEPEDFAVYYCQQYGSSFTFGPGTKVDIKR SEQ ID NO: 560 14026 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A) VL] EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDF TLTISRLEPEDFAVYYCQQYGSSFTFGPGTKVDIKR SEQ ID NO: 561 14027 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A, P141Q) VL] EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDF TLTISRLEPEDFAVYYCQQYGSSFTFGQGTKVDIKR SEQ ID NO: 562 14028 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A, P141Q) VL] EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDF TLTISRLEPEDFAVYYCQQYGSSFTFGQGTKVDIKR SEQ ID NO: 563 14029 LC [hu anti-<huCDH19> 4A2 (1-236)(R29Q, N30S) VL] EIVLTQSPGTLSLSPGERATLSCRASQSISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDF TLTISRLEPEDFTVYYCQQYGSSFTFGPGTKVDIKR SEQ ID NO: 564 14030 LC [hu anti-<huCDH19> 4F3 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKR SEQ ID NO: 565 14031 LC [hu anti-<huCDH19> 4F3 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKR SEQ ID NO: 566 14032 LC [hu anti-<huCDH19> 4F3 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKR SEQ ID NO: 567 14033 LC [hu anti-<huCDH19> 4F3 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKR SEQ ID NO: 568 14034 LC [hu anti-<huCDH19> 4F3 VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKR SEQ ID NO: 569 14039 LC [hu anti-<huCDH19> 2G6 (1-234)(C42S, D110E) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYTSWYQQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTAT LTISGTQAMDEADYYCQAWESSTVVFGGGTKLTVLG SEQ ID NO: 570 14040 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 571 14041 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 572 14042 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 573 14043 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 574 14044 LC [hu anti-<huCDH19> 16C1.1 (1-235)(G95R, H105Y, G141Q) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGNSPLTFGQGTKVEIKR SEQ ID NO: 575 14045 LC [hu anti-<huCDH19> 17H8.2 (1-235)(G149R) VL] DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLAWYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRLEMKR SEQ ID NO: 576 14046 LC [hu anti-<huCDH19> 17H8.2 (1-235)(G149R) VL] DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLAWYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRLEMKR SEQ ID NO: 577 14047 LC [hu anti-<huCDH19> 17H8.2 (1-235)(G149R) VL] DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLAWYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRLEMKR SEQ ID NO: 578 14048 LC [hu anti-<huCDH19> 17H8.2 (1-235)(S57Y, G149R) VL] DIVLTQSPOTLSLSPGERATLSCRASQSVAGSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRLEMKR SEQ ID NO: 579 14049 LC [hu anti-<huCDH19> 4F7 (1-239)(H57Y) VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDVHWYQQLPGTAPKLLIYGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSSLSGWVFGGGTRLTVLG SEQ ID NO: 580 14050 LC [hu anti-<huCDH19> 4F7 (1-239)(H57Y, D110E) VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDVHWYQQLPGTAPKLLIYGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYESSLSGWVFGGGTRLTVLG SEQ ID NO: 581 14051 LC [hu anti-<huCDH19> 4F7 (1-239)(D110E) VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDVHWYQQLPGTAPKLLIHGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYESSLSGWVFGGGTRLTVLG SEQ ID NO: 582 14052 LC [hu anti-<huCDH19> 4B10 (1-236)(H45Q, A90T) VL] EIVLTQSPOTLSLSPGERATLSCRASQSVSNTYLAWYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVEIKR SEQ ID NO: 583 14053 LC [hu anti-<huCDH19> 4B10 (1-236)(H45Q, A90T) VL] EIVLTQSPOTLSLSPGERATLSCRASQSVSNTYLAWYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVEIKR SEQ ID NO: 584 14054 LC [hu anti-<huCDH19> 4B10 (1-236)(H45Q, A90T) VL] EIVLTQSPOTLSLSPGERATLSCRASQSVSNTYLAWYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVEIKR SEQ ID NO: 585 14055 LC [hu anti-<huCDH19> 4B10 (1-236)(H45Q, A90T) VL] EIVLTQSPOTLSLSPGERATLSCRASQSVSNTYLAWYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGTD FTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVEIKR SEQ ID NO: 586 14056 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L) VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAVHWYQQLPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSRLSGWVFGGGTKLTVLG SEQ ID NO: 587 14057 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L) VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAVHWYQQLPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSRLSGWVFGGGTKLTVLG SEQ ID NO: 588 14058 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L, D110E) VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAVHWYQQLPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYESRLSGWVFGGGTKLTVLG SEQ ID NO: 589 14059 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L, D110E) VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAVHWYQQLPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYESRLSGWVFGGGTKLTVLG SEQ ID NO: 590 14060 LC [hu anti-<huCDH19> 20D3.1 (1-235)(S102A) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 591 14061 LC [hu anti-<huCDH19> 20D3.1 (1-235)(K45Q, S102A) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 592
14062 LC [hu anti-<huCDH19> 20D3.1 (1-235)(K45Q, S102A) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 593 14063 LC [hu anti-<huCDH19> 20D3.1 (1-235)(K45Q, S102A, D111E, N135Q) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDESLQGWVFGGGTKLTVLG SEQ ID NO: 594 14064 LC [hu anti-<huCDH19> 20D3.1 (1-235)(W109Y) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDESDYYCATYDDSLNGWVFGGGTKLTVLG SEQ ID NO: 595 14065 LC [hu anti-<huCDH19> 22G10.1 VL] EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTEF TLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVEIKR SEQ ID NO: 596 14066 LC [hu anti-<huCDH19> 22G10.1 VL] EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTEF TLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVEIKR SEQ ID NO: 597 14067 LC [hu anti-<huCDH19> 22G10.1 (1-234)(Q97E, S98P) VL] EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTEF TLTISSLEPEDFAVYYCQQYNYWPLTFGGGTKVEIKR SEQ ID NO: 598 14068 LC [hu anti-<huCDH19> 22G10.1 (1-234)(V78F, Q97E, S98P) VL] EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARFSOSGSGTEF TLTISSLEPEDFAVYYCQQYNYWPLTFGGGTKVEIKR SEQ ID NO: 599 14069 LC [hu anti-<huCDH19> 22G10.1 (1-234)(V78F, Q97E, S98P) VL] EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARFSGSGSGTEF TLTISSLEPEDFAVYYCQQYNYWPLTFGGGTKVEIKR SEQ ID NO: 600 14070 LC [hu anti-<huCDH19> 22G10.1 VL] EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTEF TLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVEIKR SEQ ID NO: 601 14071 LC [hu anti-<huCDH19> 16A4.1 (1-235)(G141Q) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGTSSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSPFTFGQGTKVEIKR SEQ ID NO: 602 14072 LC [hu anti-<huCDH19> 19B5.1 (1-235)(K45Q, S102A) VL] QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDDSMNGWVFGGGTKLTVLG SEQ ID NO: 603 14073 LC [hu anti-<huCDH19> 19B5.1 (1-235)(K45Q, S102A) VL] QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDDSMNGWVFGGGTKLTVLG SEQ ID NO: 604 14074 LC [hu anti-<huCDH19> 19B5.1 (1-235)(T11V, K45Q, S102A) VL] QSALTQPPSVTGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSMNGWVFGGGTKLTVLG SEQ ID NO: 605 14075 LC [hu anti-<huCDH19> 19B5.1 (1-235)(T11V, K45Q, S102A, D111E, N135Q) VL] QSALTQPPSVTGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDESMQGWVFGGGTKLTVLG SEQ ID NO: 606 14076 LC [hu anti-<huCDH19> 19B5.1 (1-235)(T11V, K45Q, S102A, W109Y, D111E, N135Q) VL] QSALTQPPSVTGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATYDESMQGWVFGGGTKLTVLG SEQ ID NO: 607 14077 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWYQQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTVLG SEQ ID NO: 608 14078 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWYQQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTVLG SEQ ID NO: 609 14079 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S, D110E) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWYQQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWESSTVVFGGGTKLTVLG SEQ ID NO: 610 14080 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42Y) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYVYWYQQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTVLG SEQ ID NO: 611 14081 LC [hu anti-<huCDH19> 25G10.1 (1-235)(H105Y) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 612 14082 LC [hu anti-<huCDH19> 25G10.1 (1-235)(H105Y) VL] EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGNSPLTFGGGTKVEIKR SEQ ID NO: 613 14083 LC [hu anti-<huCDH19> 26D1.1 (1-235)(S7P) VL] HSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 614 14084 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P) VL] QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 615 14085 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P, W109Y) VL] QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVYDDSLNGWVFGGGTKLTVLG SEQ ID NO: 616 14086 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P, W109Y, D111E, N135Q) VL] QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVYDESLQGWVFGGGTKLTVLG SEQ ID NO: 617 14087 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P, W109Y, D111E, N135Q) VL] QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVYDESLQGWVFGGGTKLTVLG SEQ ID NO: 618 14088 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P) VL] QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 619 14089 LC [hu anti-<huCDH19> 26F12.1 (1-235)(S7P) VL] QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 620 14090 LC [hu anti-<huCDH19> 26F12.1 (1-235)(S7P, D111E) VL] QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDESLNGWVFGGGTKLTVLG SEQ ID NO: 621 14091 LC [hu anti-<huCDH19> 26F12.1 (1-235)(S7P, D111E) VL] QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVWDESLNGWVFGGGTKLTVLG SEQ ID NO: 622 14092 LC [hu anti-<huCDH19> 26F12.1 (1-235)(S7P, W109Y, D111E, N135Q) VL] QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCAVYDESLQGWVFGGGTKLTVLG SEQ ID NO: 623 14093 LC [hu anti-<huCDH19> 25F8.1 (1-235)(K45Q) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCAAWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 624 14094 LC [hu anti-<huCDH19> 25F8.1 (1-235)(K45Q, S102A) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 625 14095 LC [hu anti-<huCDH19> 25F8.1 (1-235)(K45Q, S102A) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDDSLNGWVFGGGTKLTVLG SEQ ID NO: 626 14096 LC [hu anti-<huCDH19> 25F8.1 (1-235)(K45Q, S102A, D111E) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDESLNGWVFGGGTKLTVLG SEQ ID NO: 627 14097 LC [hu anti-<huCDH19> 25F8.1 (1-235)(K45Q, S102A, D111E, N135Q) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDESLQGWVFGGGTKLTVLG SEQ ID NO: 628 14098 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDDSMNGWVFGGGTKLTVLG SEQ ID NO: 629 14099 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A, D111E, N135Q) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDESMQGWVFGGGTKLTVLG SEQ ID NO: 630 14100 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A, W109Y, D111E, N135Q) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATYDESMQGWVFGGGTKLTVLG SEQ ID NO: 631 14101 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A, W109Y) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATYDDSMNGWVFGGGTKLTVLG SEQ ID NO: 632 14102 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A) VL] QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGTS ASLAISGLQSEDEADYYCATWDDSMNGWVFGGGTKLTVLG SEQ ID NO: 633 13591 LC [hu anti-<huCDH19> 4F7 VL] QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDVHWYQQLPGTAPKLLIHGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSSLSGWVFGGGTRLTVLG SEQ ID NO: 634 14301 LC [hu anti-<huCDH19> 2G6 (1-234)(D110E) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYTCWYQQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTAT LTISGTQAMDEADYYCQAWESSTVVFGGGTKLTVLG SEQ ID NO: 635 14302 LC [hu anti-<huCDH19> 2G6 (1-234)(C42S, D110E) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYTSWYQQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTAT LTISGTQAMDEADYYCQAWESSTVVFGGGTKLTVLG SEQ ID NO: 636 14303 LC [hu anti-<huCDH19> 2G6 (1-234)(C42S, D110E) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYTSWYQQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTAT LTISGTQAMDEADYYCQAWESSTVVFGGGTKLTVLG SEQ ID NO: 637 14304 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S) VL] SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWYQQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTVLG SEQ ID NO: 638
[0430] Anti-CDH19 Variable and Constant Region Polynucleotide and Amino Acid Sequences
TABLE-US-00014 TABLE IIIa Heavy Chain Variable and Contant Region Polynucleotide and Amino acid Sequences 2G6 SEQ ID NO: 639 CAGGTGCAGTTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTGGGAGGTCCCTGAGACTCTCCTGT GCAGCGTCTGGATTCACCTTCAGTAGCTATGGCATGCACTGGGTCCGCCAGGCTCCAGGCAAGGGG CTGGAGTGGGTGGCATTTATATGGTATGATGGAAGTAATAAATACTATGCAGACTCCGTGAAGGAC CGATTCACCATCTCCAGAGACAATTCCAAGAACACGCTGTATCTGCAAATGAAAAGCCTGAGAGCT GAGGACACGGCTGTGTATTACTGTGCGAGAAGGGCCGGTATAATAGGAACTATAGGCTACTACTAC GGTATGGACGTCTGGGGCCAAGGGACCACGGTCACCGTCTCTAGTGCCTCCACCAAGGGCCCATCG GTCTTCCCCCTGGCACCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTC AAGGACTACTTCCCCGAACCGGTGACGGTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTGCAC ACCTTCCCGGCTGTCCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCC AGCAGCTTGGGCACCCAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGAC AAGAAAGTTGAGCCCAAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTC CTGGGGGGACCGTCAGTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACC CCTGAGGTCACATGCGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTAC GTGGACGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTAC CGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAG GTCTCCAACAAAGCCCTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGA GAACCACAGGTGTACACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACC TGCCTGGTCAAAGGCTTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAG AACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTC ACCGTGGACAAGAGCAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTG CACAACCACTACACGCAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 640 QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYDGSNKYYADSVKD RFTISRDNSKNTLYLQMKSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 4A2 SEQ ID NO: 641 CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTTCACAGACCCTGTCCCTCACCTGC ACTGTCTCTGGTGGCTCCATCAGCAGTAGTGGTTACTACTGGAGCTGGATCCGCCAGCACCCAGGG AAGGGCCTGGAGTGGATTGGGTACATCTATTACACTGGGAGCGCCTACTACAACCCGTCCCTCAAG AGTCGAGTTACCATATCAGTAGACACGTCTAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACT CCCGCGGACACGGCCGTGTATTACTCTGCGAGAGATGGAAGCAGTGGCTGGTACTTCCAGTATTGG GGCCAGGGCACCCTGGTCACCGTCTCTAGTGCCTCCACCAAGGGCCCATCGGTCTTCCCCCTGGCA CCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCC GAACCGGTGACGGTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTC CTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACC CAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCC AAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCA GTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGC GTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAG GTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTC CTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCC CTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTAC ACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGC TTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACC ACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGC AGGTGGCAGCAGGGGAACGTCTTCTCATCCTCCGTGATCCATGAGGCTCTCCACAACCACTACACG CAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 642 QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 4A9 SEQ ID NO: 643 CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTTCGGAGACCCTGTCCCTCACCTGC ACTGTCTCTGGTGGCTCCATCAGTGGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGAAAGGGA CTGGAGTGGTTTGCATATTTCTCTTACAGTGGGAGCACCAACTACAACCCCTCCCTCAAGAGTCGA GTCACCTTATCAGTAGACACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACCGCTGCG GACACGGCCGTGTATTACTGTGCGAGGAACTGGGCCTTCCACTTTGACTTCTGGGGCCAGGGAACC CTGGTCACCGTCTCTAGTGCCTCCACCAAGGGCCCATCGGTCTTCCCCCTGGCACCCTCCTCCAAG AGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCCGAACCGGTGACG GTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTCCTACAGTCCTCA GGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACCCAGACCTACATC TGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCCAAATCTTGTGAC AAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCAGTCTTCCTCTTC CCCCCAAAACCCAAGGACACCCTCATTATCTCCCGGACCCCTGAGGTCACATGCGTGGTGGTCGAC GTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAGGTGCATAATGCC AAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTCCTCACCGTCCTG CACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCCCTCCCAGCCCCC ATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTACACCCTGCCCCCA TCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTATCCCAGC GACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACCACGCCTCCCGTG CTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGCAGGTGGCAGCAG GGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACGCAGAAGAGCCTC TCCCTGTCTCCGGGTAAATGA SEQ ID NO: 644 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWFAYFSYSGSTNYNPSLKSR VTLSVDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 4B10 SEQ ID NO: 645 CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTGGGAGGTCCCTGAGACTCTCCTGT GCAGCCTCTGGATTCACCTTCAGTAGCTATGACATGCACTGGGTCCGCCAGGCTCCAGGCAAGGGG CTGGAGTGGGTGGCAGTTATATCATATGATGGAACTAATGAATACTATGCAGACTCCGTGAAGGGC CGATTCACCATCTCCAGAGACACTTCCAAGAACACGCTGTATTTGCAAATGAACAGCCTGAGAGCT GAGGACACGGCTGTATATTACTGTGCGAGAGAACGATATTTTGACTGGTCTTTTGACTACTGGGGC CAGGGAACCCTGGTCAGCGTCTCTAGTGCCTCCACCAAGGGCCCATCGGTCTTCCCCCTGGCACCC TCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCCGAA CCGGTGACGGTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTCCTA CAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACCCAG ACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCCAAA TCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCAGTC TTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGCGTG GTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAGGTG CATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTCCTC ACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCCCTC CCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTACACC CTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTC TATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACCACG CCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGCAGG TGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACGCAG AAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 646 QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYDGTNEYYADSVKG RFTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 4F3 SEQ ID NO: 647 CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTGGGAGGTCCCTGAGACTCTCCTGT GCAGCGTCTGGATTCTCCTTCAGTAGCTATGACATGTACTCGGTCCGCCAGACTCCAGGCAAGGGG CTGGAGTGGGTGGCAGTTATATGGTATGATGGAAGTAATAAATACTATGCAGACTCCGTGAGGGGC CGATTCACCATCTCCAGAGACAATTCCAAGAACACGCTGTTTCTGCAAATGAACAGCCTGAGAGTC GAGGACACGGCTGTGTATTACTGTGCGAGAGAAACTGGGGAGGGCTGGTACTTCGATCTCTGGGGC CGTGGCACCCTGGTCACCGTCTCTAGTGCCTCCACCAAGGGCCCATCGGTCTTCCCCCTGGCACCC TCCTCCAAGAGCACCTCTGCGGGCACAGCGGCCCTGGGCTCCCTGCTCAAGGACTACTTCCCCGAA CCGGTGACGGTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTCCTA CAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACCCAG ACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCCAAA TCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCAGTC TTCCTCTTCCCCCCAAAACCCAAGTACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGCGTG GTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAGGTG CATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTCCTC ACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCCCTC CCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTACACC CTCCCCCCATCCCCGTAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTC TATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACCACG CCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGCAGG TGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACGCAG AAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 648 QVQLVESGGGVVQPGRSLRLSCAASGFSFSSYDMDWVRQTPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGRGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 4F7 SEQ ID NO: 649 CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTTCGGAGACCCTGTCCCTCACCTGC ACTGTCTCTGGTGGCTCCATCAGTAGTTACTCCTGGAGCTGGATCCGGCAGCCCCCAGGGAAGGGA CTGGAGTGGATTGGGTATATCTATTACAGTGGGAGCACCAACTACAACCCCTCCCTCAAGAGTCGA GTCACCATATCATTAGACACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACCGCTGCG GACACGGCCGTGTATTACTGTGCGAGGAACTGGGCCTTCCACTTTGACTACTGGGGCCAGGGAACC CTGGTCACCGTCTCTAGTGCCTCCACCAAGGGCCCATCGGTCTTCCCCCTGGCACCCTCCTCCAAG AGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCCGAACCGGTGACG GTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTCCTACAGTCCTCA GGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACCCAGACCTACATC TGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCCAAATCTTGTGAC AAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCAGTCTTCCTCTTC CCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGCGTGGTGGTGGAC GTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAGGTGCATAATGCC AAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTCCTCACCGTCCTG CACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCCCTCCCAGCCCCC ATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTACACCCTGCCCCCA TCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTATCCCAGC GACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACCACGCCTCCCGTG CTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGCAGGTGGCAGCAG GGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACGCAGAAGAGCCTC TCCCTGTCTCCGGGTAAATGA SEQ ID NO: 650 QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISLDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDYWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 16A4 SEQ ID NO: 651 CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGCGAAGCCTTCGGAGACCCTGTCCCTCACCTGC ACTGTCTCTGGTGACTCCATCACTAGTTACTACTGGAGCTGGATCCGCCAGCCCCCAGGGAAGGGA CTGGAGTGGATTGGGTATATCTATTACAGCGGGAGCACCAATTACAACCCCTCCCTCAAGAGTCGA GTCACCATATCAGTAGACACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGTTCTGTGACCGCTGCG GACACGGCCGTGTATTACTGTGCGAGAGATCAAAGGCGGATAGCAGCAGCTGGTACCCACTTCTAC GGTATGGACGTCTGGGGCCAAGGGACCACGGTCACTGTCTCCTCAGCTTCCACCAAGGGCCCATCC GTCTTCCCCCTGGCGCCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGCCCCTGGGCTGCCTGGTC AAGGACTACTTCCCCGAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCAC ACCTTCCCGGCTGTCCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCC AGCAGCTTGGGCACCCAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGAC AAGAAAGTTGAGCCCAAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTC CTGGGGGGACCGTCAGTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACC CCTGAGGTCACATGCGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTAC GTGGACGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTAC CGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAG GTCTCCAACAAAGCCCTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGA GAACCACAGGTCTACACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACC TGCCTGGTCAAAGGCTTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAG AACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTC ACCGTGGACAAGAGCAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTG CACAACCACTACACGCAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 652 QVQLQESGPGLAKPSETLSLTCTVSGDSITSYYWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTAVYYCARDQRRIAAAGTHFYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 16C1 SEQ ID NO: 653 CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTTCGGAGACCCTGTCCCTCACTTGT ACTGTCTCTGGTGGCTCCATCAGTGGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGGAAGGGA CTGGAGTGGATTGGGTATATCTATTACATTGGGAGCACCAACTACAACCCCTCCCTCAAGAGTCGA GTCACCATGTCAATAGACACGTCCAAGAACCAGTTCTCCCTGACGCTGAGCTCTTTGACCGCTGCG GACACGGCCGTGTATTTCTGTGCGAGAGATGGGAGCAGTGGCTGGTACCGGTGGTTCGACCCCTGG GGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGCG CCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCC GAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTC CTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACC CAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCC AAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCA GTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGC GTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAG GTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTC CTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCC CTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTAC ACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGC TTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACC
ACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGC AGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACG CAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 654 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSIDTSKNQFSLTLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 17H8 SEQ ID NO: 655 CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTTCGGAGACCCTGTCCCTCACGTGC ACTGTCTCTGGTGGCTCCATCAATAGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGGAAGGGA CTGGAGTGGATTGGGTATATCTATTACATTGGGAGCACCAACTACAACCCCTCCCTCAAGAGTCGC GTCACCATATCAGTAGACACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACCGCTGCG GACACGGCCCTGTATTACTGTGCGAGAGATTCCCGGTATAGAAGTGGCTGGTACGATGCTTTTGAT ATCTGGGGCCAAGGGACAATGGTCACCGTCTCTTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCC CTGGCGCCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTAC TTCCCCGAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCG GCTGTCCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTG GGCACCCAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTT GAGCCCAAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGA CCGTCAGTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTC ACATGCGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGC GTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTC AGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAAC AAAGCCCTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAG GTGTACACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTC AAAGGCTTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTAC AAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGAC AAGAGCAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCAC TACACGCAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 656 QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTALYYCARDSRYRSGWYDAFDIWGQGTMVTVSSASTKGPSVFP LAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSL GTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEV TCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSN KALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNY KTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 19B5 SEQ ID NO: 657 CAGGTGCAGTTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTGGGGCCTCAGTGAAGGTTTCCTGC AAGGTTTCTGGATACACCTTCACCAGCTACTTTATTCACTGGGTGCGCCAGGCCCCTGGACAAGGG CTTGAATGGATGGGAATTATCAACCCTATTAGTGTTAGCACAAGCTACGCACAGAAGTTCCAGGGC AGAGTCACCATGACCAGGGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCAGCCTGAGATCT GAGGACACGGCCGTCTATTACTCTGCGCGAGGGGGGATACAGCTATGGTTACATTTGGACTACTGG GGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGCG CCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCC GAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTC CTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACC CAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCC AAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCA GTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGC GTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAG GTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTC CTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCC CTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTAC ACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGC TTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACC ACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGC AGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACG CAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 658 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 20D3 SEQ ID NO: 659 CAGGTGCAGCTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTGGGGCCTCAGTGAAGGTTTCCTGC AAGGTTTCTGGATACACCTTCACCAGCTACTTTATTCACTGGGTGCGCCAGGCCCCTGGACAAGGG CTTGAGTGGATGGGAATAATCAACCCTATTAGTGTTAGCACAAGCTACGCACAGAAGTTCCAGGGC AGAGTCACCATGACCAGGGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCAGCCTGAGATCT GAGGACACGGCCGTGTATTACTGTGCGCGAGGGGGGATACAGCTATGGTTACATTTTGACTACTGG GGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGCG CCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCC GAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTC CTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACC CAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCC AAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCA GTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGC GTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAG GTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTC CTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCC CTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTAC ACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGC TTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACC ACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGC AGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACG CAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 660 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 22D1 SEQ ID NO: 661 CAGGTGCAGCTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTGGGGCCTCAGTGAGGGTTTCCTGC AAGGTTTCTGGATACACCTTCACCAGCTACTTTATTCACTGGGTACGCCAGGCCCCTGGACAAGGG CTTGAGTGGATGGGAATAATCAACCCTATTAGTGTTAGCACAAGCTACGCACAGAAGTTCCAGGGC AGAGTCACCATGACCAGGGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCAGCCTGAGATCT GAGGACACGGCCGTCTATTACTCTGCGCGACTGGGGGATACAGCTATGGTTACATTTTGACTACTG GGGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGC GCCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCC CGAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGT CCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCAC CCAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCC CAAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTC AGTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATG CGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGA GGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGT CCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGC CCTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTA CACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGG CTTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGAC CACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAG CAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACAC GCAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 662 QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDCTVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 22G10 SEQ ID NO: 663 GAGGTGCAACTGTTGGAGTCTGGGGGAGGCTTGGTACAGCCTGGGGGGTCCCTGAGACTCTCCTGT GCAGCCTCTGGATTCACCTTTAGCAGTTATGCCATGAACTGGGTCCGCCAGGCTCCAGGGAAGGGG CTGGAGTGGGTCTCAACTATTAGTGGTGGTGGTGCTAACACATACTACGCAGACTCCGTGAAGGGC CGGTTCACCATCTCCAGTGACAATTCCAAGAGCACGCTGTATCTGCAAATGAACAGCCTGAGAGCC GCGGACACGGCCGTATATCACTGTGCGAAAGGGGGAATGGGGGGATACTACTACGGTATGGACGTC TGGGGCCAAGGGACCACGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTG GCGCCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTC CCCGAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCT GTCCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGC ACCCAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAG CCCAAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCG TCAGTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACA TGCGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTG GAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGC GTCCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAA GCCCTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTG TACACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAA GGCTTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAG ACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAG AGCAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTAC ACGCAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 664 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKG RFTISSDNSKSTLYLQMNSLRAADTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 23A10 SEQ ID NO: 665 CAGGTGCAGCTGGTGGAGTCTGGGGGAGGCGTGGTCCAGCCTGGGAGGTCCCTGAGACTCTCCTGT GCAGCGTCTGGATTCACCTTCAGTCGCTATGGCATACACTGGGTCCGCCAGGCTCCAGGCAAGGGG CTGGAGTGGGTGGCAGTTATATGGTATGATGGAAGTAATAAATACTATGCAGACTCCGTGAAGGGC CGATTCACCATCTCCAGAGACAATTCCAAGAACACGCTGTATCTGCTAATGAACAGCCTGAGAGCC GAGGACTCGGCTGTGTATTACTGTCCGAGAAGGGCCGCTATACCTCGAACTACGGGCTACTACTAT GGTATGGACGTCTGGGGCCAAGGGACCACGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCC GTCTTCCCCCTGGCGCCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTC AAGGACTACTTCCCCGAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCAC ACCTTCCCGGCTGTCCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCC AGCAGCTTGGGCACCCAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGAC AAGAAAGTTGAGCCCAAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTC CTGGGGGGACCGTCAGTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACC CCTGAGGTCACATGCGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTAC GTGGACGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTAC CGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAG GTCTCCAACAAAGCCCTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGA GAACCACAGGTGTACACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACC TGCCTGGTCAAAGGCTTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAG AACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTC ACCGTGGACAAGAGCAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTG CACAACCACTACACGCAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 666 QVQLVESGGGVVQPGRSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYDGSNKYYADSVKG RFTISRDNSKNTLYLLMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 25F8 SEQ ID NO: 667 CAGGTGCAGCTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTGGGGCCTCAGTGAAGGTTTCCTGC AAGGCATCTGGATACACCTTCACCAGCTACTATATTCACTGGGTGCGCCAGGCCCCTGGACAAGGA CTTGAGTGGATGGGAATAATCAACCCCAGTGGTGGTAGCACAAGGTACGCACAGAAGTTCCAGGGC AGAGTCACCATGACCAGGGACACGTCCACGAGCACAGTCTTCATGGAGCTGAGCAGCCTGAGATCT GAGGACACGGCCGTGTATTACTGTGCGCGAGGGGGAATACAGCTATGGTTACATTTTGACTACTGG GGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGCG CCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCC GAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTC CTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACC CAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCC AAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCA GTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGC GTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAG GTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTC CTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCC CTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTAC ACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGC TTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATCGGCAGCCCGAGAACAACTACAAGACC ACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGC AGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACG CAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 668 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 25G10 SEQ ID NO: 669 CAGGTGCAGCTGCAGGAGTCGGGCCCAGGACTGGTGAAGCCTTCGGAGACCCTGTCCCTCACCTGC ACTGTCTCTGGTGGCTCCATCAGTGGTTACTACTGGAGCTGGATCCGGCAGCCCCCAGGGAAGGGA CTGGAGTGGATTGGGTATATCTATTACATTGGGAGCACCAACTACAACCCCTCCCTCAAGAGTCGA GTCACCATGTCAGTAGACACGTCCAAGAACCAGTTCTCCCTGAAGCTGAGCTCTGTGACCGCTGCG GACACGCCCGTGTATTACTGTGCGAGAGATGGGAGCAGTGGCTGGTACCGGTGGTTCGACCCCTGG GGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGCG CCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCC GAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTC CTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACC CAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCC AAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCA GTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGC GTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAG
GTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTC CTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCC CTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTAC ACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGC TTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACC ACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGC AGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACG CAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 670 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPG 26D1 SEQ ID NO: 671 CAGGTGCAGTTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTGGGGCCTCAGTGAAGGTTTCCTGT AAGGCATCTAGATACACCTTCACCAGCTACTATATGTCCTGGGTGCGACAGGCCCCTGGACAAGGG CTTGAGTGGATGGGAATAATCCACCCTAGTGGTGGTGACACAACCTACGCACAGAAGTTCCAGGGC AGAGTCACCATGACCGGGGACACGTCCACGAGCACAGTCTACATGGAGCTGAGCAGCCTGAGATCT GAGGACACGGCCGTGTATTACTGTGCGAGAGGGGGGATAAAACTATGGTTACATTTTGACTATTGG GGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGCG CCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCCC GAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTC CTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACC CAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCCC AAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTCA GTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGC GTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGAG GTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGTC CTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCC CTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTAC ACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGC TTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACC ACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAGC AGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACG CAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 672 QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 26F12 SEQ ID NO: 673 CAGGTGCAGTTGGTGCAGTCTGGGGCTGAGGTGAAGAAGCCTGGGGCCTCAGTGAAGGTTTCCTGC AAGGCATCTAGATACACCTTCACCAACTACTATATGTCCTGGGTGCGACAGGCCCCTGGACAAGGG CTTGAGTGGATGGGAATAATCAACCCTAGTGGTGGTGACTCAACCTACGCACAGAAGTTCCAGGGC AGACTCACCATGACCGGGGACACGTCCACGAGCACAGTCTACATGGAGCTGAGCAGCCTGAGATCT GAGGACACCTGCCGTCTATTACTCTGCGAGAGGTTGGATACAACTATGGTTACATTTTGACTACTG GGGCCAGGGAACCCTGGTCACCGTCTCCTCAGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGC GCCCTCCTCCAAGAGCACCTCTGGGGGCACAGCGGCCCTGGGCTGCCTGGTCAAGGACTACTTCCC CGAACCGGTGACGGTGTCGTGGAACTCAGGGGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGT CCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCAC CCAGACCTACATCTGCAACGTGAATCACAAGCCCAGCAACACCAAGGTGGACAAGAAAGTTGAGCC CAAATCTTGTGACAAAACTCACACATGCCCACCGTGCCCAGCACCTGAACTCCTGGGGGGACCGTC AGTCTTCCTCTTCCCCCCAAAACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATG CGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGTGGACGGCGTGGA GGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTGGTCAGCGT CCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGC CCTCCCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTA CACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGG CTTCTATCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGAC CACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTGGACAAGAG CAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACAC GCAGAAGAGCCTCTCCCTGTCTCCGGGTAAATGA SEQ ID NO: 674 QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
TABLE-US-00015 TABLE IIIb Light Chain Variable and Contant Region Polynucleotide and Amino acid Sequences 2G6 SEQ ID NO: 675 TCCTATGAACTGACTCAGCCACCCTCAGTGTCCGTGTCCCCAGGACAGACAGCCAGCATCACCTGC TCTGGAGATAGGTTGGGGGAAAAATATACTTGCTGGTATCAGCAGAGGCCAGGCCAGTCCCCTTTG CTGGTCATCTATCAAGATACCAAGCGGCCCTCAGGGATCCCTGAGCGATTCTCTGGCTCCAACTCT GGTAACACAGCCACTCTGACCATCAGCGGGACCCAGGCTATGGATGAGGCTGACTATTACTGTCAG GCGTGGGACAGCAGCACTGTGGTATTCGGCGGAGGGACCAAGCTGACCGTCCTAGGTCAGCCCAAG GCCAACCCCACTGTCACTCTGTTCCCGCCCTCCTCTGAGGAGCTCCAAGCCAACAAGGCCACACTA GTGTGTCTGATCAGTGACTTCTACCCGGGAGCTGTGACAGTGGCCTGGAAGGCAGATGGCAGCCCC GTCAAGGCGGGAGTGGAGACCACCAAACCCTCCAAACAGAGCAACAACAAGTACGCGGCCAGCAGC TACCTGAGCCTGACGCCCGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAGGTCACGCATGAA GGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 676 SYELTQPPSVSVSPGQTASITCSGDRLGEKYTCWYQQRPGQSPLLVIYQDTKRPSGIPERFSGSNS GNTATLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTVLGQPKANPTVTLFPPSSEELQANKATL VCLISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQVTHE GSTVEKTVAPTECS 4A2 SEQ ID NO: 677 GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTCCAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCAGTCGGAATATTAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAGGCT CCCAGGCTCCTCATCTATGGTCCATCCAGCAGGGCCACTGGCATCCCAGACAGGTTCAGTGGCAGT GGGTCTGGGACAGACTTCACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTACAGTGTATTAC TGTCAGCAGTATGGTAGCTCATTCACTTTCGGCCCTGGGACCAAAGTGGATATCAAACGTACGGTG GCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGAACTGCCTCTGTT GTGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCCCTC CAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTCAGC AGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACCCAT CAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTGA SEQ ID NO: 678 EIVLTQSPGTLSLSPGERATLSCRASRNISSSYLAWYQQKPGQAPRLLIYGPSSRATGIPDRFSGS GSGTDFTLTISRLEPEDFTVYYCQQYGSSFTFGPGTKVDIKRTVAAPSVFIFPPSDEQLKSGTASV VCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTH QGLSSPVTKSFNRGEC 4A9 SEQ ID NO: 679 CAGTCTGTGCTGACGCAGCCGCCCTCAGTGTCTGGGGCCCCAGGACAGAGGGTCACCATCTCCTGC ACTGGGAGCAGCTCCAACATCGGGACAGGTTATGCTGTACACTGGTACCAGCAGTTTCCAGGAACA GCCCCCAAACTCCTCATCTATGGTAACAACAATCGGCCCTCAGGGGTTCCTGACCGATTCTCTGGC TCCAAGTCTGGCACCTCAGCCTCCCTGGCCATCACTGGGCTCCAGGCTGAGGATGAGGCTGATTAT TACTGCCAGTCCTATGACAGCAGACTGAGTGGTTGGGTGTTCGGCGGAGGGACCAAGCTGACCGTC CTAGGTCAGCCCAAGGCCAACCCCACTGTCACTCTGTTCCCGCCCTCCTCTGAGGAGCTCCAAGCC AACAAGGCCACACTAGTGTGTCTGATCAGTGACTTCTACCCGGGAGCTGTGACAGTGGCCTGGAAG GCAGATGGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCAAACCCTCCAAACAGAGCAACAACAAG TACGCGGCCAGCAGCTACCTGAGCCTGACGCCCGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGC CAGGTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 680 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAVHWYQQFPGTAPKLLIYGNNNRPSGVPDRFSG SKSGTSASLAITGLQAEDEADYYCQSYDSRLSGWVFGGGTKLTVLGQPKANPTVTLFPPSSEELQA NKATLVCLISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAASSYLSLTPEQWKSHRSYSC QVTHEGSTVEKTVAPTECS 4B10 SEQ ID NO: 681 GAAATTGTATTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTCCAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCAGTCAGAGTGTTAGCAACACCTACTTAGCCTGGTACCATCAGAGACCTGGCCAGGCT CCCAGGCTCCTCATCTATGGTGCATCCAGCAGGGCCACTGGCATCCCAGACAGATTCAGTGGCAGT GGGTCTGGGACAGACTTCGCTCTCACCATCAGCAGTCTGGAGCCTGAAGATTTTGCAGTGTATTAC TGTCAGCAGTACAGTAACTCGTGGACGTTCGGCCAAGGGACCAAGGTGGAAATCAAACGAACTGTG GCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGAACTGCCTCTGTT GTGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCCCTC CAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTCAGC AGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACCCAT CAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTGA SEQ ID NO: 682 EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLAWYHQRPGQAPRLLIYGASSRATGIPDRFSGS GSGTDFALTISSLEPEDFAVYYCQQYSNSWTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASV VCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTH QGLSSPVTKSFNRGEC 4F3 SEQ ID NO: 683 GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTCCAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCAGTCAGAGTGTTAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAGGCT CCCAGGCTCCTCATCTATGGTGCATCCAGCAGGGCCACTGGCATCCCAGACAGGTTCAGTGGCAGT GGGTCTGGGACAGACTTCACTCTCACCATCAGCAGACTGGAACCTGAGGATTTTGCAGTGTATTAC TGTCAGCAGTATGGTAGCTCGTGGACGTTCGGCCAAGGGACCAAGGTGGAAATCAAACGTACGGTG GCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGAACTGCCTCTGTT GTGTGCCTCCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTCGAAGGTGGATAACGCCCTC CAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTCAGC AGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACCCAT CAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTGA SEQ ID NO: 684 EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGASSRATGIPDRFSGS GSGTDFTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASV VCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTH QGLSSPVTKSFNRGEC 4F7 SEQ ID NO: 685 CAGTCTGTGCTGACGCAGCCGCCCTCAGTGTCTGGGGCCCCAGGGCAGAGGGTCACCATCTCCTGC ACTGGGAGCAGCTCCAATATCGGGACAGGTTATGATGTACACTGGTATCAGCAGCTTCCAGGAACA GCCCCCAAACTCCTCATCCATGGTAACAGCAATCGGCCCTCAGGGGTCCCTGACCGATTCTCTGGC TCCAAGTCTGGCACCTCAGCCTCCCTGGCCATCACTGGGCTCCAGGCTGAGGATGAGGCTGATTAT TACTGCCAGTCCTATGACAGCAGTCTGAGTGGTTGGGTGTTCGGCGGAGGGACCAGGTTGACCGTC CTAGGTCAGCCCAAGGCCAACCCCACTGTCACTCTGTTCCCGCCCTCCTCTGAGGAGCTCCAAGCC AACAAGGCCACACTAGTGTGTCTGATCAGTGACTTCTACCCGGGAGCTGTGACAGTGGCCTGGAAG GCAGATGGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCAAACCCTCCAAACAGAGCAACAACAAG TACGCGGCCAGCAGCTACCTGAGCCTGACGCCCGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGC CAGGTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 686 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDVHWYQQLPGTAPKLLIHGNSNRPSGVPDRFSG SKSGTSASLAITGLQAEDEADYYCQSYDSSLSGWVFGGGTRLTVLGQPKANPTVTLFPPSSEELQA NKATLVCLISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAASSYLSLTPEQWKSHRSYSC QVTHEGSTVEKTVAPTECS 16A4 SEQ ID NO: 687 GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTCCAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCAGTCAGAGTGTTAGCAGCAGTTATTTAGCCTGGTACCAGCAGAAACCTGGCCAGGCT CCCAGGCTCCTCATCTATGGTACATCCAGCAGGGCCACTGGCATCCCAGACAGGTTCAGTGGCAGT GGGTCTGGGACAGACTTCACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTGTATTAT TGTCAGCAGTACGGTAGCTCACCTTTCACTTTCGGCGGAGGGACCAAGGTGGAGATCAAACGAACT GTGGCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGTACCGCCTCT GTTGTGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCC CTCCAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTC AGCAGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACC CATCAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTGA SEQ ID NO: 688 EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIYGTSSRATGIPDRFSGS GSGTDFTLTISRLEPEDFAVYYCQQYGSSPFTFGGGTKVEIKRTVAAPSVFIFPPSDEQLKSGTAS VVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVT HQGLSSPVTKSFNRGEC 16C1 SEQ ID NO: 689 GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTCCAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCATCCAGAGTGTTAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAGGCT CCCAGGCTCCTCATCTTTGGTGCATCCAGCAGGGCCACTGGCATCCCAGACAGGTTCAGTGGCAGT GGGTCTGGGACAGACTTCACTCTCACCATCAGCGGACTGGAGCCTGAAGATTTTGCAGTGTATCAC TGTCAGCAGTATGGTAACTCACCGCTCACTTTCGGCGGAGGGACCAAGGTGGAGATCAAACGAACT GTGGCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGTACCGCCTCT GTTGTGTGCCTCCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTCGAAGGTGGATAACGCC CTCCAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTC AGCAGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACC CATCAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTCTTGA SEQ ID NO: 690 EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGS GSGTDFTLTISGLEPEDFAVYHCQQYGNSPLTFGGGTKVEIKRTVAAPSVFIFPPSDEQLKSGTAS VVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVT HQGLSSPVTKSFNRGEC 17H8 SEQ ID NO: 691 GACATTGTATTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTCCAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCAGTCAGAGTGTTGCCGGCAGCTACCTAGCCTGGTACCAGCAGAAACCTGGCCAGGCT CCCAGGCTCCTCATCTCTCGTCCATCCAGCAGGGCCACTGGCATCCCAGACAGGTTCAGTGGCAGT GGGTCTGGGACAGACTTCACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTGTATTAC TGTCAGCAGTATGGTAAATCACCGATCACCTTCGGCCAAGGGACACGACTGGAGATGAAAGGAACT GTGGCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGTACCGCCTCT GTTGTGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCC CTCCAATCGGGTAACTCCCAGGAGAGTCTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTC AGCAGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACC CATCAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTGA SEQ ID NO: 692 DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLAWYQQKPGQAPRLLISGASSRATGIPDRFSGS GSGTDFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRLEMKGTVAAPSVFIFPPSDEQLKSGTAS VVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVT HQGLSSPVTKSFNRGEC 19B5 SEQ ID NO: 693 CAGTCTGCGCTGACTCAGCCACCCTCAACGACTGGGACCCCCGGGCAGAGGGTCACCATCTCTTGT TCTGGAAGCAGGTCCAACATCGGAAGCAATTTTGTAAACTGGTACAAGCAGCTCCCAGGAACGGCC CCCAAAGTCCTCATCTATACTAATAATCAGCGGCCCTCAGGGGTCCCTGACCGATTCTCTGGCTCC AAGTCTGGCACCTCAGCCTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGATTATTAC TGCGCAACATGGGATGACAGTATGAATGGTTGGGTGTTCGGCGGAGGGACCAAACTGACCGTCCTA GGTCAGCCCAAGGCTGCCCCCTCGGTCACTCTGTTCCCACCCTCCTCTGAGGAGCTTCAAGCCAAC AAGGCCACACTGGTGTGTCTCATAAGTGACTTCTACCCGGGAGCCGTGACAGTGGCCTGGAAGGCA GATAGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCACACCCTCCAAACAAAGCAACAACAAGTAC GCGGCCAGCAGCTATCTGAGCCTGACGCCTGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAG GTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 694 QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGS KSGTSASLAISGLQSEDESDYYCATWDDSMNGWVFGGGTKLTVLGQPKAAPSVTLFPPSSEELQAN KATLVCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQ VTHEGSTVEKTVAPTECS 20D3 SEQ ID NO: 695 CAGTCTCCGCTGACTCAGCCACCCTCAGCGACTGGGACCCCCGGGCAGAGGGTCACCATCTCTTGT TCTGGAAGCAGCTCCAACATCGGAAGCAATTTTGTAAACTGGTACAAGCAGCTCCCAGGAACGGCC CCCAAAGTCCTCATCTATACTAATAATCAGCGGCCCTCAGGGGTCCCTGACCGATTCTCTGGCTCC AAGTCTGGCACCTCAGCCTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGATTATTAC TGTGCAACATGGGATGACAGCCTGAATGGTTGGGTGTTCGGCGGAGGGACCAAGCTGACCGTCCTA GGTCAGCCCAAGGCTTCCCCCTCGCTCACTCTCTTCCCACCCTCCTCTGAGGAGCTTCAATCCAAC AAGGCCACACTGGTGTGTCTCATAAGTGACTTCTACCCGGGAGCCGTGACAGTGGCCTGGAAGGCA GATAGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCACACCCTCCAAACAAAGCAACAACAAGTAC GCGGCCAGCAGCTATCTGAGCCTGACGCCTGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAG GTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 696 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGS KSGTSASLAISGLQSEDESDYYCATWDDSLNGWVFGGGTKLTVLGQPKAAPSVTLFPPSSEELQAN KATLVCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQ VTHEGSTVEKTVAPTECS 22D1 SEQ ID NO: 697 CAGTCTGCGCTGACTCAGCCACCCTCAGCGACTGGGACCCCCGGGCAGAGGGTCACCATCTCTTGT TCTGGAAGCAGCTCCAACATCGGAAGCAATTTTGTAAACTGGTACAAGCAGCTCCCAGGAACCGCC CCCAAAGTCCTCATCTATACTAATAATCAGCGGCCCTCAGGGGTCCCTGACCGATTCTCTGGCTCC AAGTCTGGCACCTCAGCCTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGATTATTAC TGTGCAACATGGGATGACAGTATGAATGGTTGGGTGTTCGGCGGAGGGACCAAGCTGACCGTCCTA GGTCAGCCCAAGGCTGCCCCCTCGGTCACTCTGTTCCCACCCTCCTCTGAGGAGCTTCAAGCCAAC AAGGCCACACTGGTGTGTCTCATAAGTGACTTCTACCCGGGAGCCGTGACAGTGGCCTGGAAGGCA GATAGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCACACCCTCCAAACAAAGCAACAACAAGTAC GCGGCCAGCAGCTATCTGAGCCTGACGCCTGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAG GTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 698 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGS KSGTSASLAISGLQSEDESDYYCATWDDSMNGWVFGGGTKLTVLGQPKAAPSVTLFPPSSEELQAN KATLVCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQ VTHEGSTVEKTVAPTECS 22G10 SEQ ID NO: 699 GAAATAGTGATGACGCAGTCTCCAGTCACCCTGTCTCTGTCTCTAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCAGTCAGAGTATTAGCAGCAACTTAGCCTGGTTCCAGCAGAAACCTGGCCAGGCTCCC AGACTCCTCATCTATGGTGCATTTACCAGGGCCACTGGTATCCCAGCCAGGGTCAGTGGCAGTGGG TCTGGGACAGAGTTCACTCTCACCATCAGCAGCCTGCAGTCTGAAGATTTTGCAGTTTATTACTGT CAGCAGTATAATTACTGGCCGCTCACTTTCGGCGGAGGGACCAAGGTGGAGATCAAGCGAACTGTG GCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGTACCGCCTCTGTT GTGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCCCTC CAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTCAGC AGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACCCAT CAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTGA SEQ ID NO: 700 EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAWFQQKPGQAPRLLIYGAFTRATGIPARVSGSG SGTEFTLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASV VCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTH QGLSSPVTKSFNRGEC 23A10 SEQ ID NO: 701 TCCTATGAGCTGACTCAGCCACCCTCAGTGTCCGTGTCCCCAGGACAGACAGCCAGCATCACCTGC TCTGGAGATAGATTGGGGGAGAAATATGTTTGCTGGTATCAGCAGAAGCCAGGCCAGTCCCCTATA CTGGTCATCTATCAAGATAATAAGTGGCCCTCAGGGATCCCTGAGCGATTCTCTGGCTCCAACTCT GGGAACACAGCCACTCTGACCATCAGCGGGACCCAGGCTATGGATGAGGCTGACTATTACTGTCAG GCGTGGGACAGCAGCACTGTGGTATTCGGCGGGGGGACCAAGCTGACCGTCCTAGGTCAGCCCAAG GCTGCCCCCTCGGTCACTCTGTTCCCACCCTCCTCTGAGGAGCTTCAAGCCAACAAGGCCACACTG GTGTGTCTCATAAGTGACTTCTACCCGGGAGCCGTGACAGTGGCCTGGAAGGCAGATAGCAGCCCC GTCAAGGCGGGAGTGGAGACCACCACACCCTCCAAACAAAGCAACAACAAGTACGCGGCCAGCAGC
TATCTGAGCCTGACGCCTGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAGGTCACGCATGAA GGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 702 SYELTQPPSVSVSPGQTASITCSGDRLGEKYVCWYQQKPGQSPILVIYQDNKWPSGIPERFSGSNS GNTATLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTVLGQPKAAPSVTLFPPSSEELQANKATL VCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQVTHE GSTVEKTVAPTECS 25F8 SEQ ID NO: 703 CAGTCTGCGCTGACTCAGCCACCCTCAGCGACTGGGACCCCCGGGCAGAGGGTCACCATCTCTTGT TCTGGAAGCAGCTCCAACATCGGAAGGAATTTTGTAAACTGGTATAAGCAGCTCCCAGGAACGGCC CCCAAAGTCCTCATTTATACTAATAATCAGCGGCCCTCAGGGGTCCCTGACCGATTCTCTGGCTCC AAGTCTGGCACCTCAGCCTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGTCTGATTATTAC TGTGCAGCATGGGATGACAGCCTGAATGGTTGGGTGTTCGGCGGAGGGACCAAGCTGACCGTCCTA GGTCAGCCCAAGGCTGCCCCCTCGGTCACTCTGTTCCCACCCTCCTCTGAGGAGCTTCAAGCCAAC AAGGCCACACTGGTGTGTCTCATAAGTGACTTCTACCCGGGAGCCGTGACAGTGGCCTGGAAGGCA GATAGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCACACCCTCCAAACAAAGCAACAACAAGTAC GCGGCCAGCAGCTATCTGAGCCTGACGCCTGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAG GTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 704 QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVNWYKQLPGTAPKVLIYTNNQRPSGVPDRFSGS KSGTSASLAISGLQSEDESDYYCAAWDDSLNGWVFGGGTKLTVLGQPKAAPSVTLFPPSSEELQAN KATLVCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQ VTHEGSTVEKTVAPTECS 25G10 SEQ ID NO: 705 GAAATTGTGTTGACGCAGTCTCCAGGCACCCTGTCTTTGTCTCCAGGGGAAAGAGCCACCCTCTCC TGCAGGGCCAGTCAGAGTGTTAGCAGCAGCTACTTAGCCTGGTACCAGCAGAAACCTGGCCAGGCT CCCAGGCTCCTCATCTTTGGTGCATCCAGCAGGGCCACTGGCATCCCAGACAGGTTCAGTGGCAGT GGGTCTGGGACAGACTTCACTCTCACCATCAGCAGACTGGAGCCTGAAGATTTTGCAGTGTATCAC TGTCAGCAGTATGGTAACTCACCGCTCACTTTCGGCGGAGGGACCAAGGTGGAGATCAAACGAACT GTGGCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGTACCGCCTCT GTTGTGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCC CTCCAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTC AGCAGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACC CATCAGGCTCCTGAGCTCGCCCGTCACAAACTAGCTTCAACAGGGCTAGAGTCTTCTA SEQ ID NO: 706 EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWYQQKPGQAPRLLIFGASSRATGIPDRFSGS GSGTDFTLTISRLEPEDFAVYHCQQYGNSPLTFGGGTKVEIKRTVAAPSVFIFPPSDEQLKSGTAS VVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVT HQGLSSPVTKSFNRGEC 26D1 SEQ ID NO: 707 CACTCTGTGCTGACTCAGTCACCCTCAGCGTCTGGGACCCCCGGACAGAGGGTCACCATCTCTTGT TCTGGAAGCCGCTCCAACATCGGAAGTAATTTTGTAAACTGGTACCAGCAGCTCCCAGGAACGGCC CCCAAACTCCTCATCTATACTAATAATCAGCGGCCCTCAGGGGTCCCTGACCGATTCTCTGGCTCC AAGTCTGGCACCTCAGCCTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGGCTGATTATTAC TGTGCAGTATGGGATGACAGCCTGAATGGTTGGGTGTTCGGCGGAGGGACCAAGCTGACCGTCCTA GGTCAGCCCAAGGCTGCCCCCTCGGTCACTCTGTTCCCACCCTCCTCTGAGGAGCTTCAAGCCAAC AAGGCCACACTGGTGTGTCTCATAAGTGACTTCTACCCGGGAGCCGTGACAGTGGCCTGGAAGGCA GATAGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCACACCCTCCAAACAAAGCAACAACAAGTAC GCGCCCAGCAGCTATCTGAGCCTGACGCCTGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAG GTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 708 HSVLTQSPSASGTPGQRVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNNQRPSGVPDRFSGS KSGTSASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLGQPKAAPSVTLFPPSSEELQAN KATLVCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQ VTHEGSTVEKTVAPTECS 26F12 SEQ ID NO: 709 CAGTCTGTGCTGACTCAGTCACCCTCAGCGTCTGGGACCCCCGGGCAGAAGGTCACCATCTCTTGT TCTGGAAGCCGCTCCAACATCGGAAGTAATTTTGTAAACTGGTACCAGCAGCTCCCAGGAACGGCC CCCAAACTCCTCATCTATACTAATTATCAGCGGCCCTCAGGGGTCCCTGACCGATTCTCTGGCTCC AAGTCTGGCACCTCAGCCTCCCTGGCCATCAGTGGGCTCCAGTCTGAGGATGAGGCTGATTATTAC TGTGCAGTATGGGATGACAGCCTGAATGGTTGGGTGTTCGGCGGAGGGACCAAGCTGACCGTCCTA GGTCAGCCCAAGGCTGCCCCCTCGGTCACTCTGTTCCCACCCTCCTCTGAGGAGCTTCAAGCCAAC AAGGCCACACTGGTGTGTCTCATAAGTGACTTCTACCCGGGAGCCGTGACAGTGGCCTGGAAGGCA GATAGCAGCCCCGTCAAGGCGGGAGTGGAGACCACCACACCCTCCAAACAAAGCAACAACAAGTAC GCGGCCAGCAGCTATCTGAGCCTGACGCCTGAGCAGTGGAAGTCCCACAGAAGCTACAGCTGCCAG GTCACGCATGAAGGGAGCACCGTGGAGAAGACAGTGGCCCCTACAGAATGTTCATGA SEQ ID NO: 710 QSVLTQSPSASGTPGQKVTISCSGSRSNIGSNFVNWYQQLPGTAPKLLIYTNYQRPSGVPDRFSGS KSGTSASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGTKLTVLGQPKAAPSVTLFPPSSEELQAN KATLVCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQ VTHEGSTVEKTVAPTECS
TABLE-US-00016 TABLE 111c Heavy Chain Variable and Contant Region Polynucleotide and Amino acid Sequences 13586_HC [hu anti-<huCDH19> 4F3 VH]::huIgG1z SEQ ID NO: 711 QVQLVESGGGVVQPGRSLRLSCAASGFSFSSYDMDWVRQTPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGRGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13589_HC [hu anti-<huCDH19> 4A9 VH]::huIgG1z SEQ ID NO: 712 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWFAYFSYSGSTNYNPSLKSR VTLSVDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13590_HC [hu anti-<huCDH19> 4B10 VH]::huIgG1z SEQ ID NO: 713 QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYDGTNEYYADSVKG RFTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13874_HC [hu anti-<huCDH19> 17H8.2 VH]::huIgG1z SEQ ID NO: 714 QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTALYYCARDSRYRSGWYDAFDIWGQGTMVTVSSASTKGPSVFP LAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSL GTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEV TCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSN KALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNY KTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13875_HC [hu anti-<huCDH19> 16C1.1 VH]::huIgG1z SEQ ID NO: 715 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSIDTSKNQFSLTLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13876_HC [hu anti-<huCDH19> 16A4.1 VH]::huIgG1z SEQ ID NO: 716 QVQLQESGPGLAKPSETLSLTCTVSGDSITSYYWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTAVYYCARDQRRIAAAGTHFYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQCINVFSCSVMHEALHNHYTQKSLSLSPGK 13877_HC [hu anti-<huCDH19> 22G10.1 VH]::huIgG1z SEQ ID NO: 717 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKG RFTISSDNSKSTLYLQMNSLRAADTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13878_HC [hu anti-<huCDH19> 20D3.1 VH]::huIgG1z SEQ ID NO: 718 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13879_HC [hu anti-<huCDH19> 22D1.1 VH]::huIgG1z SEQ ID NO: 719 QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13880_HC [hu anti-huCDH19> 25F8.1 VH]::huIgG1z SEQ ID NO: 720 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13881_HC [hu anti-<huCDH19> 26F12.1 VH]::huIgG1z SEQ ID NO: 721 QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13882_HC [hu anti-<huCDH19> 26D1.1 VH]::huIgG1z SEQ ID NO: 722 QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13883_HC [hu anti-<huCDH19> 25G10.1 VH]::huIgG1z SEQ ID NO: 723 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13885_HC [hu anti-<huCDH19> 19B5.1 VH]::huIgG1z SEQ ID NO: 724 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARCiGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKENWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14022_HC [hu anti-<huCDH19> 4A2 VH]::huIgG1z SEQ ID NO: 725 QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14024_HC [hu anti-<huCDH19> 4A2 (1-472)(Q17E,H47P) VH]::huIgG1z SEQ ID NO: 726 QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14025_HC [hu anti-<huCDH19> 4A2 VH]::huIgG1z SEQ ID NO: 727 QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14026_HC [hu anti-<huCDH19> 4A2 (1-472)(Q17E,H47P) VH]::huIgG1z SEQ ID NO: 728 QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14027_HC [hu anti-<huCDH19> 4A2 (1-472) (Q17E,H47P,D111E) VH]::huIgG1z SEQ ID NO: 729 QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCAREGSSGWYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14028_HC [hu anti-<huCDH19> 4A2 (1-472) (Q17E,H47P,D111E,W134Y) VH]::huIgG1z SEQ ID NO: 730 QVQLQESGPGLVKPSETLSLTCTVSGGSISSSGYYWSWIRQPPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCAREGSSGYYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14029_HC [hu anti-<huCDH19> 4A2 VH]::huIgG1z SEQ ID NO: 731 QVQLQESGPGLVKPSQTLSLTCTVSGGSISSSGYYWSWIRQHPGKGLEWIGYIYYTGSAYYNPSLK SRVTISVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYFQYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14030_HC [hu anti-<huCDH19> 4F3 (1-471)(R17G) VH]::huIgG1z SEQ ID NO: 732 QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQTPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGRGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14031_HC [hu anti-<huCDH19> 4F3 (1-471)(R17G,T47A) VH]::huIgG1z SEQ ID NO: 733 QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGRGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14032_HC [hu anti-<huCDH19> 4F3 (1-471) (R17G,T47A,R141Q) VH]::huIgG1z SEQ ID NO: 734 QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYDGSNKYYADSVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGQGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14033_HC [hu anti-<huCDH19> 4F3 (1-471)
(R17G,T47A,D61E,D72E,R141Q) VH]::huIgG1z SEQ ID NO: 735 QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYEGSNKYYAESVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGWYFDLWGQGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14034_HC [hu anti-<huCDH19> 4F3 (1-471) (R17G,T47A,D61E,D72E,W134Y,R141Q) VH]::huIgG1z SEQ ID NO: 736 QVQLVESGGGVVQPGGSLRLSCAASGFSFSSYDMDWVRQAPGKGLEWVAVIWYEGSNKYYAESVRG RFTISRDNSKNTLFLQMNSLRVEDTAVYYCARETGEGYYFDLWGQGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14039_HC [hu anti-<huCDH19> 2G6 (1-477) (R17G,D61E,D72E,K94N) VH]::huIgG1z SEQ ID NO: 737 QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYEGSNKYYAESVKD RFTISRDNSKNTLYLQMNSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14040_HC [hu anti-<huCDH19> 16C1.1 VH]::huIgG1z SEQ ID NO: 738 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSIDTSKNQFSLTLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14041_HC [hu anti-<huCDH19> 16C1.1 (1-469)(T92K) VH]::huIgG1z SEQ ID NO: 739 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSIDTSKNQFSLKLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14042_HC [hu anti-<huCDH19> 16C1.1 (1-469) (T92K,D109E) VH]::huIgG1z SEQ ID NO: 740 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSIDTSKNQFSLKLSSLTAADTAVYFCAREGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14043_HC [hu anti-<huCDH19> 16C1.1 (1-469) (T92K,W132Y,W135Y) VH]::huIgG1z SEQ ID NO: 741 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSIDTSKNQFSLKLSSLTAADTAVYFCARDGSSGYYRYFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14044_HC [hu anti-<huCDH19> 16C1.1 (1-469)(T92K) VH]::huIgG1z SEQ ID NO: 742 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSIDTSKNQFSLKLSSLTAADTAVYFCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14045_HC [hu anti-<huCDH19> 17H8.2 VH]::huIgG1z SEQ ID NO: 743 QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTALYYCARDSRYRSGWYDAFDIWGQGTMVTVSSASTKGPSVFP LAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSL GTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEV TCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSN KALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNY KTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14046_HC [hu anti-<huCDH19> 17H8.2 (1-471)(D109E) VH]::huIgG1z SEQ ID NO: 744 QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTALYYCARESRYRSGWYDAFDIWGQGTMVTVSSASTKGPSVFP LAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSL GTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEV TCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSN KALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNY KTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14047_HC [hu anti-<huCDH19> 17H8.2 (1-471) (D109E,W132Y) VH]::huIgG1z SEQ ID NO: 745 QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTALYYCARESRYRSGYYDAFDIWGQGTMVTVSSASTKGPSVFP LAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSL GTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEV TCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSN KALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNY KTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14048_HC [hu anti-<huCDH19> 17H8.2 (1-471)(D109E) VH]::huIgG1z SEQ ID NO: 746 QVQLQESGPGLVKPSETLSLTCTVSGGSINSYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTALYYCARESRYRSGWYDAFDIWGQGTMVTVSSASTKGPSVFP LAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSL GTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEV TCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSN KALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNY KTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14049_HC [hu anti-<huCDH19> 4F7 VH]::huIgG1z SEQ ID NO: 747 QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISLDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDYWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14050_HC [hu anti-<huCDH19> 4F7 VH]::huIgG1z SEQ ID NO: 748 QVQLQESGPGLVKPSETESLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISLDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDYWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14051_HC [hu anti-<huCDH19> 4F7 (1-468)(W113Y) VH]::huIgG1z SEQ ID NO: 749 QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISLDTSKNQFSLKLSSVTAADTAVYYCARNYAFHFDYWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14052_HC [hu anti-<huCDH19> 4B10 (1-471) (R17G,D61E,D72E,W134Y) VH]::huIgG1z SEQ ID NO: 750 QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYEGTNEYYAESVKG RFTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDYSFDYWGQGTLVSVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14053_HC [hu anti-<huCDH19> 4B10 VH]::huIgG1z SEQ ID NO: 751 QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYDGTNEYYADSVKG RFTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14054_HC [hu anti-<huCDH19> 4B10 (1-471)(R17G) VH]::huIgG1z SEQ ID NO: 752 QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYDGTNEYYADSVKG RFTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14055_HC [hu anti-<huCDH19> 4B10 (1-471) (R17G,D61E,D72E) VH]::huIgG1z SEQ ID NO: 753 QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYDMHWVRQAPGKGLEWVAVISYEGTNEYYAESVKG RFTISRDTSKNTLYLQMNSLRAEDTAVYYCARERYFDWSFDYWGQGTLVSVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14056_HC [hu anti-<huCDH19> 4A9 VH]::huIgG1z SEQ ID NO: 754 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWFAYFSYSGSTNYNPSLKSR VTLSVDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14057_HC [hu anti-<huCDH19> 4A9 (1-468)(F55I,A56G) VH]::huIgG1z SEQ ID NO: 755 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYFSYSGSTNYNPSLKSR VTLSVDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14058_HC [hu anti-<huCDH19> 4A9 (1-468)(F55I,A56G) VH]::huIgG1z SEQ ID NO: 756 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYFSYSGSTNYNPSLKSR VTLSVDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDFWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14059_HC [hu anti-<huCDH19> 4A9 (1-468) (F55I,A56G,W113Y) VH]::huIgG1z SEQ ID NO: 757 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYFSYSGSTNYNPSLKSR VTLSVDTSKNQFSLKLSSVTAADTAVYYCARNYAFHFDFWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14060_HC [hu anti-<huCDH19> 20D3.1 VH]::huIgG1z SEQ ID NO: 758 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14061_HC [hu anti-<huCDH19> 20D3.1 VH]::huIgG1z SEQ ID NO: 759 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG
RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14062_HC [hu anti-<huCDH19> 20D3.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 760 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSERSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14063_HC [hu anti-<huCDH19> 20D3.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 761 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14064_HC [hu anti-<huCDH19> 20D3.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 762 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14065_HC [hu anti-<huCDH19> 22G10.1 (1-470) (S82R,A99E) VH]::huIgG1z SEQ ID NO: 763 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKG RFTISRDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14066_HC [hu anti-<huCDH19> 22G10.1 (1-470) (A99E,H105Y) VH]::huIgG1z SEQ ID NO: 764 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKG RFTISSDNSKSTLYLQMNSLRAEDTAVYYCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14067_HC [hu anti-<huCDH19> 22G10.1 (1-470)(A99E) VH]::huIgG1z SEQ ID NO: 765 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKG RFTISSDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14068_HC [hu anti-<huCDH19> 22G10.1 (1-470)(A99E) VH]::huIgG1z SEQ ID NO: 766 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKG RFTISSDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14069_HC [hu anti-<huCDH19> 22G10.1 (1-470) (D72E,A99E) VH]::huIgG1z SEQ ID NO: 767 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYAESVKG RFTISSDNSKSTLYLQMNSLRAEDTAVYHCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14070_HC [hu anti-<huCDH19> 22G10.1 (1-470)(H105Y) VH]::huIgG1z SEQ ID NO: 768 EVQLLESGGGLVQPGGSLRLSCAASGFTFSSYAMNWVRQAPGKGLEWVSTISGGGANTYYADSVKG RFTISSDNSKSTLYLQMNSLRAADTAVYYCAKGGMGGYYYGMDVWGQGTTVTVSSASTKGPSVFPL APSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLG TQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVT CVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNK ALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYK TTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14071_HC [hu anti-<huCDH19> 16A4.1 (1-474)(T144L) VH]::huIgG1z SEQ ID NO: 769 QVQLQESGPGLAKPSETLSLTCTVSGDSITSYYWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISVDTSKNQFSLKLSSVTAADTAVYYCARDQRRIAAAGTHFYGMDVWGQGTLVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14072_HC [hu anti-<huCDH19> 19B5.1 VH]::huIgG1z SEQ ID NO: 770 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14073_HC [hu anti-<huCDH19> 19B5.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 771 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14074_HC [hu anti-<huCDH19> 19B5.1 VH]::huIgG1z SEQ ID NO: 772 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14075_HC [hu anti-<huCDH19> 19B5.1 VH]::huIgG1z SEQ ID NO: 773 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIMPISVSTSYAQKFQGR VTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLAP SSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQ TYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCV VVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTT PPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14076_HC [hu anti-<huCDH19> 19B5.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 774 QVQLVQSGAEVKKPGASVKVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14077_HC [hu anti-<huCDH19> 23A10.3 (1-474)(L92Q) VH]::huIgG1z SEQ ID NO: 775 QVQLVESGGGVVQPGRSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYDGSNKYYADSVKG RFTISRDNSKNTLYLQMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14078_HC [hu anti-<huCDH19> 23A10.3 (1-474) (R17G,L92Q) VH]::huIgG1z SEQ ID NO: 776 QVQLVESGGGVVQPGGSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYDGSNKYYADSVKG RFTISRDNSKNTLYLQMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14079_HC [hu anti-<huCDH19> 23A10.3 (1-474) (R17G,D61E,D72E,L92Q) VH]::huIgG1z SEQ ID NO: 777 QVQLVESGGGVVQPGGSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYEGSNKYYAESVKG RFTISRDNSKNTLYLQMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14080_HC [hu anti-<huCDH19> 23A10.3 VH]::huIgG1z SEQ ID NO: 778 QVQLVESGGGVVQPGRSLRLSCAASGFTFSRYGIHWVRQAPGKGLEWVAVIWYDGSNKYYADSVKG RFTISRDNSKNTLYLLMNSLRAEDSAVYYCARRAGIPGTTGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14081_HC [hu anti-<huCDH19> 25G10.1 VH]::huIgG1z SEQ ID NO: 779 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSVDTSKNQFSLKLSSVTAADTAVYYCARDGSSGWYRWFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14082_HC [hu anti-<huCDH19> 25G10.1 (1-469) (D109E,W132Y,W135Y) VH]::huIgG1z SEQ ID NO: 780 QVQLQESGPGLVKPSETLSLTCTVSGGSISGYYWSWIRQPPGKGLEWIGYIYYIGSTNYNPSLKSR VTMSVDTSKNQFSLKLSSVTAADTAVYYCAREGSSGYYRYFDPWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14083_HC [hu anti-<huCDH19> 26D1.1 VH]::huIgG1z SEQ ID NO: 781 QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14084_HC [hu anti-<huCDH19> 26D1.1 VH]::huIgG1z SEQ ID NO: 782 QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14085_HC [hu anti-<huCDH19> 26D1.1 VH]::huIgG1z SEQ ID NO: 783 QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT
TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14086_HC [hu anti-<huCDH19> 26D1.1 VH]::huIgG1z SEQ ID NO: 784 QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14087_HC [hu anti-<huCDH19> 26D1.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 785 QVQLVQSGAEVKKPGASVKVSCKASRYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIKLYLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14088_HC [hu anti-<huCDH19> 26D1.1 (1-469) (R27G,G82R) VH]::huIgG1z SEQ ID NO: 786 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYMSWVRQAPGQGLEWMGIIHPSGGDTTYAQKFQG RVTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIKLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14089_HC [hu anti-<huCDH19> 26F12.1 VH]::huIgG1z SEQ ID NO: 787 QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14090_HC [hu anti-<huCDH19> 26F12.1 VH]::huIgG1z SEQ ID NO: 788 QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14091_HC [hu anti-<huCDH19> 26F12.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 789 QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14092_HC [hu anti-<huCDH19> 26F12.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 790 QVQLVQSGAEVKKPGASVKVSCKASRYTFTNYYMSWVRQAPGQGLEWMGIINPSGGDSTYAQKFQG RLTMTGDTSTSTVYMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14093_HC [hu anti-<huCDH19> 25F8.1 VH]::huIgG1z SEQ ID NO: 791 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGI1NPSGGSTRYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14094_HC [hu anti-<huCDH19> 25F8.1 VH]::huIgG1z SEQ ID NO: 792 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14095_HC [hu anti-<huCDH19> 25F8.1 (1-469)(F90Y) VH]::huIgG1z SEQ ID NO: 793 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQG RVTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14096_HC [hu anti-<huCDH19> 25F8.1 (1-469)(F90Y) VH]::huIgG1z SEQ ID NO: 794 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQG RVTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14097_HC [hu anti-<huCDH19> 25F8.1 (1-469) (F90Y,W133Y) VH]::huIgG1z SEQ ID NO: 795 QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYYIHWVRQAPGQGLEWMGIINPSGGSTRYAQKFQG RVTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLYLHFDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14098_HC [hu anti-<huCDH19> 22D1.1 VH]::huIgG1z SEQ ID NO: 796 QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14099_HC [hu anti-<huCDH19> 22D1.1 VH]::huIgG1z SEQ ID NO: 797 QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14100_HC [hu anti-<huCDH19> 22D1.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 798 QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14101_HC [hu anti-<huCDH19> 22D1.1 (1-469)(W133Y) VH]::huIgG1z SEQ ID NO: 799 QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVFMELSSLRSEDTAVYYCARGGIQLYLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14102_HC [hu anti-<huCDH19> 22D1.1 (1-469)(F90Y) VH]::huIgG1z SEQ ID NO: 800 QVQLVQSGAEVKKPGASVRVSCKVSGYTFTSYFIHWVRQAPGQGLEWMGIINPISVSTSYAQKFQG RVTMTRDTSTSTVYMELSSLRSEDTAVYYCARGGIQLWLHLDYWGQGTLVTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGT QTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTC VVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKA LPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKT TPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 13591_HC [hu anti-<huCDH19> 4F7 VH]::huIgG1z SEQ ID NO: 801 QVQLQESGPGLVKPSETLSLTCTVSGGSISSYSWSWIRQPPGKGLEWIGYIYYSGSTNYNPSLKSR VTISLDTSKNQFSLKLSSVTAADTAVYYCARNWAFHFDYWGQGTLVTVSSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYI CNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD VSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAP IEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14301_HC [hu anti-<huCDH19> 2G6 VH]::huIgG1z SEQ ID NO: 802 QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYDGSNKYYADSVKD RFTISRDNSKNTLYLQMKSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14302_HC [hu anti-<huCDH19> 2G6 (1-477)(R17G,K94N) VH]::huIgG1z SEQ ID NO: 803 QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYDGSNKYYADSVKD RFTISRDNSKNTLYLQMNSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14303_HC [hu anti-<huCDH19> 2G6 (1-477)(D61E,D72E) VH]::huIgG1z SEQ ID NO: 804 QVQLVESGGGVVQPGRSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYEGSNKYYAESVKD RFTISRDNSKNTLYLQMKSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK 14304_HC [hu anti-<huCDH19> 2G6 (1-477)(R17G) VH]::huIgG1z SEQ ID NO: 805 QVQLVESGGGVVQPGGSLRLSCAASGFTFSSYGMHWVRQAPGKGLEWVAFIWYDGSNKYYADSVKD RFTISRDNSKNTLYLQMKSLRAEDTAVYYCARRAGIIGTIGYYYGMDVWGQGTTVTVSSASTKGPS VFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPS SSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRT PEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCK VSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
TABLE-US-00017 TABLE IIId Light Chain Variable and Contant Region Polynucleotide and Amino acid Sequences 13586 LC [hu anti-<huCDH19> 4F3 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 806 13589 LC [hu anti-<huCDH19> 4A9 VL]::huLLC-C1 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAV HWYQQFPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSRLSGWVFGGG TKLTVLGQPKANPTVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 807 13590 LC [hu anti-<huCDH19> 4B10 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLA WYHQRPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFALTISSLEPEDFAVYYCQQYSNSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 808 13874 LC [hu anti-<huCDH19> 17118.2 VL]::huKLC DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLA WYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRL EMKGTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 809 13875 LC [hu anti-<huCDH19> 16C1.1 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISGLEPEDFAVYHCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 810 13876 LC [hu anti-<huCDH19> 16A4.1 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIYGTSSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSPFTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 811 13877 LC [hu anti-<huCDH19> 22G10.1 VL]::huKLC EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAW FQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTE FTLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 812 13878 LC [hu anti-<huCDH19> 20D3.1 VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCATWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 813 13879 LC [hu anti-<huCDH19> 22D1.1 VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCATWDDSMNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 814 13880 LC [hu anti-<huCDH19> 25F8.1 VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVN WYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCAAWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 815 13881 LC [hu anti-<huCDH19> 26F12.1 VL]::huLLC-C2 QSVLTQSPSASGTPGQKVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 816 13882 LC [hu anti-<huCDH19> 26D1.1 VL]::huLLC-C2 HSVLTQSPSASGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 817 13883 LC [hu anti-<huCDH19> 25G10.1 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYHCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 818 13885 LC [hu anti-<huCDH19> 19B5.1 VL]::huLLC-C2 QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNFVN WYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCATWDDSMNGWVFGGGT ICLTVLGQPICAAPSVTLFPPSSEELQANKATLVC LISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSN NKYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEK TVAPTECS SEQ ID NO: 819 14022 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAWY QQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTDFTL TISRLEPEDFTVYYCQQYGSSFTFGPGTKVDIKRT VAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREA KVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTL TLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC SEQ ID NO: 820 14024 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A, P141Q) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLA WYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSFTFGQGTKVD IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSICADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 821 14025 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLA WYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSFTFGPGTKVD IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 822 14026 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLA WYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSFTFGPGTKVD IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 823 14027 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A, P141Q) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLA WYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSFTFGQGTKVD IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 824 14028 LC [hu anti-<huCDH19> 4A2 (1-236)(N30Q, T102A, P141Q) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASRQISSSYLAW YQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGTD FTLTISRLEPEDFAVYYCQQYGSSFTFGQGTKVDI KRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYP REAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLS STLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNR GEC SEQ ID NO: 825 14029 LC [hu anti-<huCDH19> 4A2 (1-236)(R290, N30S) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSISSSYLA WYQQKPGQAPRLLIYGPSSRATGIPDRFSGSGSGT DFTLTISRLEPEDFTVYYCQQYGSSFTFGPGTKVD IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSONSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 826 14030 LC [hu anti-<huCDH19> 4F3 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKEKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 827 14031 LC [hu anti-<huCDH19> 4F3
VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQICPGQAPRLLIYGASSRATGIPDRFSGSGSG TDFTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 828 14032 LC [hu anti-<huCDH19> 4F3 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVEI KRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYP REAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLS STLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNR GEC SEQ ID NO: 829 14033 LC [hu anti-<huCDH19> 4F3 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLAWY QQKPGQAPRLLIYGASSRATGIPDRFSGSGSGTDFTL TISRLEPEDFAVYYCQQYGSSWTFGQGTKVEIKRT VAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREA KVQWKVDNALQSONSQESVTEQDSKDSTYSLSSTL TLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC SEQ ID NO: 830 14034 LC [hu anti-<huCDH19> 4F3 VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 831 14039 LC [hu anti-<huCDH19> 2G6 (1-234)(C42S, D110E) VL]::huLLC-C1 SYELTQPPSVSVSPGQTASITCSGDRLGEKYTSWY QQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWESSTVVFGGGTKLTV LGQPKANPTVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTE CS SEQ ID NO: 832 14040 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 833 14041 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL]::hul(LC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 834 14042 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL]::hul(LC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 835 14043 LC [hu anti-<huCDH19> 16C1.1 (1-235)(H105Y) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISGLEPEDFAVYYCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 836 14044 LC [hu anti-<huCDH19> 16C1.1 (1-235)(G95R, H105Y, G141Q) VL]::hul(LC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGNSPLTFGQGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 837 14045 LC [hu anti-<huCDH19> 17H8.2 (1-235)(G149R) VL]::huKLC DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLA WYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRL EMKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 838 14046 LC [hu anti-<huCDH19> 17H8.2 (1-235)(G149R) VL]::huKLC DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLA WYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRL EMKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 839 14047 LC [hu anti-<huCDH19> 17118.2 (1-235)(G149R) VL]::huKLC DIVLTQSPGTLSLSPGERATLSCRASQSVAGSYLA WYQQKPGQAPRLLISGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRL EMKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 840 14048 LC [hu anti-<huCDH19> 17118.2 (1-235)(S57Y, G149R) VL]::huKLC DIVLTQSPOTLSLSPGERATLSCRASQSVAGSYLA WYQQKPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGKSPITFGQGTRL EMKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 841 14049 LC [hu anti-<huCDH19> 4F7 (1-239)(H57Y) VL]::huLLC-C2 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDV HWYQQLPGTAPKLLIYGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSSLSGWVFGGG TRLTVLGQPKANPTVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 842 14050 LC [hu anti-<huCDH19> 4F7 (1-239)(1157Y, D110E) VL]::huLLC-C2 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDV HWYQQLPGTAPKLLIYGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYESSLSGWVFGGG TRLTVLGQPKANPTVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 843 14051 LC [hu anti-<huCDH19> 4F7 (1-239)(D110E) VL]::huLLC-C2 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDV HWYQQLPGTAPKLLIHGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYESSLSGWVFGGG TRLTVLGQPKANPTVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 844 14052 LC [hu anti-<huCDH19> 4B10 (1-236)(H45Q, A90T) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLA WYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 845 14053 LC [hu anti-<huCDH19> 4B10 (1-236)(11450, A90T) VL]::huKLC EIVLTQSPOTLSLSPGERATLSCRASQSVSNTYLA WYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 846 14054 LC [hu anti-<huCDH19> 4B10 (1-236)(11450, A90T) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLA WYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 847 14055 LC [hu anti-<huCDH19> 4B10 (1-236)(H45Q, A90T) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSNTYLA WYQQRPGQAPRLLIYGASSRATGIPDRFSGSGSGT DFTLTISSLEPEDFAVYYCQQYSNSWTFGQGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 848 14056 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L) VL]::huLLC-C1 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAV HWYQQLPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSRLSGWVFGGG TKLTVLGQPKANPTVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 849 14057 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L) VL]::huLLC-C1 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAV HWYQQLPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSRLSGWVFGGG TKLTVLGQPKANPTVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADOSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 850
14058 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L, D110E) VL]::huLLC-C1 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAV HWYQQLPGTAPKLLIYGNNNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYESRLSGWVEGGG TKLTVLGQPKANPTVTLEPPSSEELQANKATLVCL ISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 851 14059 LC [hu anti-<huCDH19> 4A9 (1-239)(F47L, D110E) VL]::huLLC-C1 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYAV HWYQQLPGTAPKWYGNNNRPSGVPDRFSGSKSGTS ASLAITGLQAEDEADYYCQSYESRLSGWVFGGGTK LTVLGQPKANPTVTLFPPSSEELQANKATLVCLIS DFYPGAVTVAWKADGSPVKAGVETTKPSKQSNNKY AASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVA PTECS SEQ ID NO: 852 14060 LC [hu anti-<huCDH19> 20D3.1 (1-235)(S102A) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 853 14061 LC [hu anti-<huCDH19> 20D3.1 (1-235)(K45Q, S102A) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSLNGWVFGGGT KLTVLGQPICAAPSVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 854 14062 LC [hu anti-<huCDH19> 20D3.1 (1-235)(K45Q, S102A) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSLNGWVEGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 855 14063 LC [hu anti-<huCDH19> 20D3.1 (1-235)(K45Q, S102A, D111E, N135Q) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDESLQGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 856 14064 LC [hu anti-<huCDH19> 20D3.1 (1-235)(W109Y) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYKQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCATYDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 857 14065 LC [hu anti-<huCDH19> 22G10.1 VL]::huKLC EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAW FQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTE FTLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 858 14066 LC [hu anti-<huCDH19> 22G10.1 VL]::huKLC EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAW FQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTE FTLTISSLQSEDFAVYYCQQYNYWPLTFGGGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 859 14067 LC [hu anti-<huCDH19> 22G10.1 (1-234)(097E, S981P) VL]::huKLC EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAW FQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTE FTLTISSLEPEDFAVYYCQQYNYWPLTFGGGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 860 14068 LC [hu anti-<huCDH19> 22G10.1 (1-234)(V78F, 097E, S98P) VL]::huKLC EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAW FQQKPGQAPRLLIYGAFTRATGIPARFSGSGSGTE FTLTISSLEPEDFAVYYCQQYNYWPLTFGGGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNEY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 861 14069 LC [hu anti-<huCDH19> 22G10.1 (1-234)(V78F, 097E, S98P) VL]::huKLC EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAW FQQKPGQAPRLLIYGAFTRATGIPARFSGSGSGTE FTLTISSLEPEDFAVYYCQQYNYWPLTFGGGTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 862 14070 LC [hu anti-<huCDH19> 22G10.1 VL]::huKLC EIVMTQSPVTLSLSLGERATLSCRASQSISSNLAW FQQKPGQAPRLLIYGAFTRATGIPARVSGSGSGTE FTLTISSLQSEDFAVYYCQQYNYWPLTEGGOTKVE IKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNEY PREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSL SSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFN RGEC SEQ ID NO: 863 14071 LC [hu anti-<huCDH19> 16A4.1 (1-235)(G141Q) VL]::huKLC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIYGTSSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGSSPFTFGQGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 864 14072 LC [hu anti-<huCDH19> 19B5.1 (1-235)(K45Q, S102A) VL]::huLLC-C2 QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSMNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 865 14073 LC [hu anti-<huCDH19> 19B5.1 (1-235)(K450, S102A) VL]::huLLC-C2 QSALTQPPSTTGTPGQRVTISCSGSRSNIGSNEVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSMNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 866 14074 LC [hu anti-<huCDH19> 19B5.1 (1-235)(T11V, K450, S102A) VL]::huLLC-C2 QSALTQPPSVTGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSMNGWVFGGOT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 867 14075 LC [hu anti-<huCDH19> 19B5.1 (1-235)(T11V, K45Q, S102A, D111E, N135Q) VL]::huLLC-C2 QSALTQPPSVTGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDESMQGWVEGGGT KLTVLGQPKAAPSVTLEPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 868 14076 LC [hu anti-<huCDH19> 19B5.1 (1-235)(T11V, K45Q, S102A, W109Y, D111E, N135Q) VL]::huLLC-C2 QSALTQPPSVTGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATYDESMQGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 869 14077 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S) VL]::huLLC-C2 SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWY QQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTV LGQPKAAPSVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTE CS SEQ ID NO: 870 14078 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S) VL]::huLLC-C2 SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWY QQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTV LGQPKAAPSVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTE CS SEQ ID NO: 871 14079 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S, D110E) VL]::huLLC-C2 SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWY QQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWESSTVVFGGGTKLTV LGQPKAAPSVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTE CS SEQ ID NO: 872 14080 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42Y) VL]::huLLC-C2 SYELTQPPSVSVSPGQTASITCSGDRLGEKYVYWY QQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA
TLTISGTQAMDEADYYCQAWDSSTVVFOGGTKLTV LGQPKAAPSVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTE CS SEQ ID NO: 873 14081 LC [hu anti-<huCDH19> 25G10.1 (1-235)(H105Y) VL]::hul(LC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 874 14082 LC [hu anti-<huCDH19> 25G10.1 (1-235)(H105Y) VL]::hul(LC EIVLTQSPGTLSLSPGERATLSCRASQSVSSSYLA WYQQKPGQAPRLLIFGASSRATGIPDRFSGSGSGT DFTLTISRLEPEDFAVYYCQQYGNSPLTFGGGTKV EIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNF YPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSF NRGEC SEQ ID NO: 875 14083 LC [hu anti-<huCDH19> 26D1.1 (1-235)(S7P) VL]::huLLC-C2 HSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 876 14084 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P) VL]::huLLC-C2 QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 877 14085 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P, W109Y) VL]::huLLC-C2 QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVYDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 878 14086 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P, W109Y, D111E, N135Q) VL]::huLLC-C2 QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVYDESLQGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 879 14087 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P, W109Y, D111E, N1350) VL]::huLLC-C2 QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVYDESLQGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 880 14088 LC [hu anti-<huCDH19> 26D1.1 (1-235)(H1Q, S7P) VL]::huLLC-C2 QSVLTQPPSASGTPGQRVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 881 14089 LC [hu anti-<huCDH19> 26E12.1 (1-235)(S7P) VL]::huLLC-C2 QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 882 14090 LC [hu anti-<huCDH19> 26F12.1 (1-235)(S7P, D111E) VL]::huLLC-C2 QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDESLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 883 14091 LC [hu anti-<huCDH19> 26E12.1 (1-235)(S7P, D111E) VL]::huLLC-C2 QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVWDESLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 884 14092 LC [hu anti-<huCDH19> 26E12.1 (1-235)(S7P, W109Y, D111E, N1350) VL]::huLLC-C2 QSVLTQPPSASGTPGQKVTISCSGSRSNIGSNFVN WYQQLPGTAPKLLIYTNYQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAVYDESLQGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 885 14093 LC [hu anti-<huCDH19> 25E8.1 (1-235)(K45Q) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDESDYYCAAWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 886 14094 LC [hu anti-<huCDH19> 25E8.1 (1-235)(K45Q, S102A) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 887 14095 LC [hu anti-<huCDH19> 25F8.1 (1-235)(K450, S102A) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDDSLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 888 14096 LC [hu anti-<huCDH19> 25E8.1 (1-235)(K450, S102A, D111E) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDESLNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 889 14097 LC [hu anti-<huCDH19> 25E8.1 (1-235)(K45Q, S102A, D111E, N135Q) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGRNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCAAWDESLQGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 890 14098 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K450, S102A) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSMNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 891 14099 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A, D111E, N135Q) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDESMQGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 892 14100 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A, W109Y, D111E, N1350) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATYDESMQGWVFOGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 893 14101 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K450, S102A, W109Y) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATYDDSMNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 894 14102 LC [hu anti-<huCDH19> 22D1.1 (1-235)(K45Q, S102A) VL]::huLLC-C2 QSALTQPPSATGTPGQRVTISCSGSSSNIGSNFVN
WYQQLPGTAPKVLIYTNNQRPSGVPDRFSGSKSGT SASLAISGLQSEDEADYYCATWDDSMNGWVFGGGT KLTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNK YAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTV APTECS SEQ ID NO: 895 13591 LC [hu anti-<huCDH19> 4F7 VL]::huLLC-C1 QSVLTQPPSVSGAPGQRVTISCTGSSSNIGTGYDV HWYQQLPGTAPKLLIHGNSNRPSGVPDRFSGSKSG TSASLAITGLQAEDEADYYCQSYDSSLSGWVFGGG TRLTVLGQPKANPTVTLFPPSSEELQANKATLVCL ISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNN KYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKT VAPTECS SEQ ID NO: 896 14301 LC [hu anti-<huCDH19> 2G6 (1-234)(D110E) VL]::huLLC-C1 SYELTQPPSVSVSPGQTASITCSGDRLGEKYTCWY QQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWESSTVVFGGGTKLTV LGQPKANPTVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPIE CS SEQ ID NO: 897 14302 LC [hu anti-<huCDH19> 2G6 (1-234)(C42S, D110E) VL]::huLLC-C1 SYELTQPPSVSVSPGQTASITCSGDRLGEKYTSWY QQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWESSTVVFGGGTKLTV LGQPKANPTVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPIE CS SEQ ID NO: 898 14303 LC [hu anti-<huCDH19> 2G6 (1-234)(C42S, D110E) VL]::huLLC-C1 SYELTQPPSVSVSPGQTASITCSGDRLGEKYTSWY QQRPGQSPLLVIYQDTKRPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWESSTVVFGGGTKLTV LGQPKANPTVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPIE CS SEQ ID NO: 899 14304 LC [hu anti-<huCDH19> 23A10.3 (1-231)(C42S) VL]::huLLC-C2 SYELTQPPSVSVSPGQTASITCSGDRLGEKYVSWY QQKPGQSPILVIYQDNKWPSGIPERFSGSNSGNTA TLTISGTQAMDEADYYCQAWDSSTVVFGGGTKLTV LGQPKAAPSVTLFPPSSEELQANKATLVCLISDFY PGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAAS SYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTE CS SEQ ID NO: 900
TABLE-US-00018 TABLE Iva HEAVY CHAIN CDRs Ab Type CDR 1 CDR 2 CDR 3 14039 AA SYGMH FIWYE RAGIIGT 14303 SEQ ID GSNKY IGYYYGM NO: 28 YAESV DV KD SEQ ID SEQ ID NO: 30 NO: 901 14027 AA SSGYY YIYYT EGSSGW WS GSAYY YFQY SEQ ID NPSLK SEQ ID NO: 46 S NO: 902 SEQ ID NO: 47 14028 AA SSGYY YIYYT EGSSGY WS GSAYY YFQY SEQ ID NPSLK SEQ ID NO: 46 S NO: 903 SEQ ID NO: 47 14059 AA GYYWS YFSYS NYAFHF SEQ ID GSTNY DF NO: 52 NPSLK SEQ ID S NO: 904 SEQ ID NO: 53 14052 AA SYDMH VISYE ERYFD SEQ ID GTNEY YSFDY NO: 58 YAESV SEQ ID KG NO: 906 SEQ ID NO: 905 14055 AA SYDMH VISYE ERYFDW SEQ ID GTNEY SFDY NO: 58 YAESV SEQ ID KG NO: 60 SEQ ID NO: 905 14033 AA SYDMD VIWYE ETGEGW SEQ ID GSNKY YFDL NO: 70 YAESV SEQ ID RG NO: 72 SEQ ID NO: 907 14034 AA SYDMD VIWYE ETGEGY SEQ ID GSNKY YFDL NO: 70 YAESV SEQ ID RG NO: 908 SEQ ID NO: 907 14051 AA SYSWS YIYYS NYAFH SEQ ID GSTNY FDY NO: 82 NPSLK SEQ ID S NO: 909 SEQ ID NO: 83 14046 AA SYYWS YIYYI ESRYRS 14048 SEQ ID GSTNY GWYDAF NO: 94 NPSLK DI S SEQ ID SEQ ID NO: 910 NO: 95 14047 AA SYYWS YIYYI ESRYRS SEQ ID GSTNY GYYDAF NO: 94 NPSLK DI S SEQ ID SEQ ID NO: 911 NO: 95 14042 AA GYYWS YIYYI EGSSGW SEQ ID GSTNY YRWFDP NO: 100 NPSLK SEQ ID S NO: 912 SEQ ID NO: 101 14043 AA GYYWS YIYYI DGSSGY SEQ ID GSTNY YRYFDP NO: 100 NPSLK SEQ ID S NO: 913 SEQ ID NO: 101 14069 AA SYAMN TISGG GGMGGY SEQ ID GANTY YYGMDV NO: 118 YAESV SEQ ID KG NO: 120 SEQ ID NO: 914 14062 AA SYFIH IINPI GGIQLY 14063 SEQ ID SVSTS LHFDY 14064 NO: 124 YAQKF SEQ ID QG NO: 915 SEQ ID NO: 125 14100 AA SYFIH IINPI GGIQLY 14101 SEQ ID SVSTS LHLDY NO: 130 YAQKF SEQ ID QG NO: 916 SEQ ID NO: 131 14097 AA SYYIH IINPS GGIQLY SEQ ID GGSTR LHFDY NO: 136 YAQKF SEQ ID QG NO: 917 SEQ ID NO: 137 14091 AA NYYMS IINPS GGIQL 14092 SEQ ID GGDST YLHFDY NO: 142 YAQKF SEQ ID QG NO: 918 SEQ ID NO: 143 14087 AA SYYMS IIHPS GGIKLY SEQ ID GGDTT LHFDY NO: 148 YAQKF SEQ ID QG NO: 919 SEQ ID NO: 149 14082 AA GYYWS YIYYI EGSSGY SEQ ID GSTNY YRYFDP NO: 154 NPSLK S SEQ ID NO: 155 SEQ ID NO: 920 14079 AA RYGIH VIWYE RAGIPGT SEQ ID GSNKY TGYYYGM NO: 160 YAESV DV KG SEQ ID SEQ ID NO: 162 NO: 921 14073 AA SYFIH IINPI GGIQLY 14076 SEQ ID SVSTS LHLDY NO: 1 YAQKF SEQ ID QG NO: 3 SEQ ID NO: 2 AA SYGMH VIWYD RAGIIG SEQ ID GSNKY TTGYYY NO: 4 YADSV GMDV KG SEQ ID SEQ ID NO: 6 NO: 5
TABLE-US-00019 TABLE IVb LIGHT CHAIN CDRs Ab Type CDR 1 CDR 2 CDR 3 14039 AA SGDRL QDTKR QAWES 14302 GEKYT PS STW 14303 S SEQ ID SEQ ID SEQ ID NO: 197 NO: 923 NO: 922 14301 AA SGDRL QDTKR QAWES GEKYT PS STW C SEQ ID SEQ ID SEQ ID NO: 197 NO: 923 NO: 196 14022 AA RASRQ GPSSR QQYGS 14024 ISSSY AT SFT 14025 LA SEQ ID SEQ ID 14026 SEQ ID NO: 215 NO: 216 14027 NO: 924 14028 14029 AA RASQS GPSSR QQYGS ISSSY AT SFT LA SEQ ID SEQ ID SEQ ID NO: 215 NO: 216 NO: 925 14058 AA TGSSS GNNNR QSYES 14059 NIGTG PS RLSGW YAVH SEQ ID V SEQ ID NO: 221 SEQ ID NO: 220 NO: 926 14050 AA TGSSS GNSNR QSYES 14051 NIGTG PS SLSGW YDVH SEQ ID V SEQ ID NO: 251 SEQ ID NO: 250 NO: 927 14063 AA SGSSS TNNQR ATWDE NIGSN PS SLQGW FVN SEQ ID V SEQ ID NO: 293 SEQ ID NO: 292 NO: 928 14064 AA SGSSS TNNQR ATYDD NIGSN PS SLNGW FVN SEQ ID V SEQ ID NO: 293 SEQ ID NO: 292 NO: 929 14099 AA SGSSS TNNQR ATWDE NIGSN PS SMQGW FVN SEQ ID V SEQ ID NO: 299 SEQ ID NO: 298 NO: 930 14100 AA SGSSS TNNQR ATYDE NIGSN PS SMQGW FVN SEQ ID V SEQ ID NO: 299 SEQ ID NO: 298 NO: 931 14101 AA SGSSS TNNQR ATYDD NIGSN PS SMNGW FVN SEQ ID V SEQ ID NO: 299 SEQ ID NO: 298 NO: 932 14096 AA SGSSS TNNQR AAWDE NIGRN PS SLNGW FVN SEQ ID V SEQ ID NO: 305 SEQ ID NO: 304 NO: 933 14097 AA SGSSS TNNQR AAWDE NIGRN PS SLQGW FVN SEQ ID V SEQ ID NO: 305 SEQ ID NO: 304 NO: 934 14090 AA SGSRS TNYQR AVWDE 14091 NIGSN PS SLNGW FVN SEQ ID V SEQ ID NO: 311 SEQ ID NO: 310 NO: 935 14092 AA SGSRS TNYQR AVYDE NIGSN PS SLQGW FVN SEQ ID V SEQ ID NO: 311 SEQ ID NO: 310 NO: 936 14085 AA SGSRS TNNQR AVYDD NIGSN PS SLNGW FVN SEQ ID V SEQ ID NO: 317 SEQ ID NO: 316 NO: 937 14086 AA SGSRS TNNQR AVYDE 14087 NIGSN PS SLQGW FVN SEQ ID V SEQ ID NO: 317 SEQ ID NO: 316 NO: 938 14077 AA SGDRL QDNKW QAWDS 14078 GEKYV PS STW 14304 S SEQ ID SEQ ID SEQ ID NO: 329 NO: 330 NO: 939 14079 AA SGDRL QDNKW QAWES GEKYV PS STW S SEQ ID SEQ ID SEQ ID NO: 329 NO: 940 NO: 939 14080 AA SGDRL QDNKW QAWDS GEKYV PS STW Y SEQ ID SEQ ID SEQ ID NO: 329 NO: 330 NO: 941 14075 AA SGSRS TNNQR ATWDE NIGSN PS SMQGW FVN SEQ ID V SEQ ID NO: 335 SEQ ID NO: 334 NO: 942 14076 AA SGSRS TNNQR ATYDE NIGSN PS SMQGW FVN SEQ ID V SEQ ID NO: 335 SEQ ID NO: 334 NO: 943
[0431] Human and Cynomologous Monkey Cadherin-19 Sequences
TABLE-US-00020 TABLE V SEQ ID DESIG- NO. NATION SOURCE TYPE SEQUENCE 944 Human Human aa MNCYLLLRFMLGIPLLWPCL Cadherin- GATENSQTKKVKQPVRSHLR 19 VKRGWVWNQFFVPEEMNTTS HHIGQLRSDLDNGNNSFQYK LLGAGAGSTFIIDERTGDIY AIQKLDREERSLYILRAQVI DIATGRAVEPESEFVIKVSD INDNEPKFLDEPYEAIVPEM SPEGTLVIQVTASDADDPSS GNNARLLYSLLQGQPYFSVE PTTGVIRISSKMDRELQDEY WVIIQAKDMIGQPGALSGTT SVLIKLSDVNDNKPIFKESL YRLTVSESAPTGTSIGTIMA YDNDIGENAEMDYSIEEDDS QTFDIITNHETQEGIVILKK KVDFEHQNHYGIRAKVKNHH VPEQLMKYHTEASTTFIKIQ VEDVDEPPLFLLPYYVFEVF EETPQGSFVGVVSATDPDNR KSPIRYSITRSKVFNINDNG TITTSNSLDREISAWYNLSI TATEKYNIEQISSIPLYVQV LNINDHAPEFSQYYETYVCE NAGSGQVIQTISAVDRDESI EEHHFYFNLSVEDTNNSSFT IIDNQDNTAVILTNRTGFNL QEEPVFYISILIADNGIPSL TSTNTLTIHVCDCGDSGSTQ TCQYQELVLSMGFKTEVIIA ILICIMIIFGFIFLTLGLKQ RRKQILFPEKSEDFRENIFQ YDDEGGGEEDTEAFDIAELR SSTIMRERKTRKTTSAEIRS LYRQSLQVGPDSAIFRKFIL EKLEEANTDPCAPPFDSLQT YAFEGTGSLAGSLSSLESAV SDQDESYDYLNELGPRFKRL ACMFGSAVQSNN 945 Human Human nt atgaactgttatttactgct Cadherin- gcgttttatgttgggaattc 19 ctctcctatggccttgtctt ggagcaacagaaaactctca aacaaagaaagtcaagcagc cagtgcgatctcatttgaga gtgaagcgtggctgggtgtg gaaccaattttttgtaccag aggaaatgaatacgactagt catcacatcggccagctaag atctgatttagacaatggaa acaattctttccagtacaag cttttgggagctggagctgg aagtacttttatcattgatg aaagaacaggtgacatatat gccatacagaagcttgatag agaggagcgatccctctaca tcttaagagcccaggtaata gacatcgctactggaagggc tgtggaacctgagtctgagt ttgtcatcaaagtttcggat atcaatgacaatgaaccaaa attcctagatgaaccttatg aggccattgtaccagagatg tctccagaaggaacattagt tatccaggtgacagcaagtg atgctgacgatccctcaagt ggtaataatgctcgtctcct ctacagcttacttcaaggcc agccatatttttctgttgaa ccaacaacaggagtcataag aatatcttctaaaatggata gagaactgcaagatgagtat tgggtaatcattcaagccaa ggacatgattggtcagccag gagcgttgtctggaacaaca agtgtattaattaaactttc agatgttaatgacaataagc ctatatttaaagaaagttta taccgcttgactgtctctga atctgcacccactgggactt ctacaggaacaatcatggca tatgataatgacataggaga gaatgcagaaatggattaca gcattgaagaggatgattcg caaacatttgacattattac taatcatgaaactcaagaag gaatagttatattaaaaaag aaagtggatcttgagcacca gaaccactacggtattagag caaaagttaaaaaccatcat gttcctgagcagctcatgaa gtaccacactgaggcttcca ccactttcattaagatccag gtggaagatgttgatgagcc tcctcttttcctccttccat attatgtatttgaagttttt gaagaaaccccacagggatc atttgtaggcgtggtgtctg ccacagacccagacaatagg aaatctcctatcaggtattc tattactaggagcaaagtgt tcaatatcaatgataatggt acaatcactacaagtaactc actgcatcgtgaaatcagtg cttggtacaacctaagtatt acagccacagaaaaatacaa tatagaacagatctcttcga tcccactgtatgtgcaagtt cttaacatcaatgatcatgc tcctgagttctctcaatact atgagacttatgtttgtgaa aatgcaggctccggtcaggt aattcagactatcagtgcag tggatagagatgaatccata gaagagcaccatttttactt taatctatctgtagaagaca ctaacaattcaagttttaca atcatagataaccaagataa cacagctgtcattttgacta atagaactggttttaacctt caagaagaacctgtcttcta catctccatcttaattgccg acaatggaatcccgtcactt acaagtacaaacacccttac catccatgtctgtgactgtg gtgacagtgggagcacacag acctgccagtaccaggagct tgtgctttccatgggattca agacagaagtcatcattgct attctcatttgcattatgat catatttgggtttatttttt tgactttgggtttaaaacaa cggagaaaacagattctatt tcctgagaaaag tgaagatttcagagagaata tattccaatatgatgatgaa gggggtggagaagaagatac agaggcctttgatatagcag agctgaggagtagtaccata atgcgggaacgcaagactcg gaaaaccacaagcgctgaga tcaggagcctatacaggcag tctttgcaagttggccccga cagtgccatattcaggaaat tcattctggaaaagctcgaa gaagctaatactgatccgtg tgcccctccttttgattccc tccagacctacgcttttgag ggaacagggtcattagctgg atccctgagctccttagaat cagcagtctctgatcacgat gaaagctatgattaccttaa tgagttgggacctcgcttta aaagattagcatgcatgttt ggttctgcagtgcagtcaaa taattag 946 Cyno Macaca aa MNCYLLLPFMLGIPLLWPCL Cadherin- fasci- GATENSQTKKVQQPVGSHLR 19 cularis VKRGWVWNQFFVPEEMNTTS HHVGRLRSDLDNGNNSFQYK LLGAGAGSTFIIDERTGDIY AIEKLDREERSLYILRAQVI DITTGRAVEPESEFVIKVSD INDNEPKFLDEPYEAIVPEM SPEGTLVIQVTASDADDPSS GNNARLLYSLLQGQPYFSVE PTTGVIRISSKMDRELQDEY WVIIQAKDMIGQPGALSGTT SVLIKLSDVNDNKPIFKESL YRLTVSESAPTGTSIGTIMA YDNDIGENAEMDYSIEEDDS QTFDIITNHETQEGIVILKK KVNFEHQNHYGIRAKVKNHH VDEQLMKYHTEASTTFIKIQ VEDVDEPPLFLLPYYIFEIF EETPQGSFVGVVSATDPDNR KSPIRYSITRSKVFNIDDNG TITTTNSLDREISAWYNLSI TATEKYNIEQISSIPVYVQV LNINDHAPEFSQYYESYVCE NAGSGQVIQTISAVDRDESI EEHHFYFNLSVEDTNSSSFT IIDNQDNTAVILTNRTGFNL QEEPIFYISILIADNGIPSL TSTNTLTIHVCDCDDSGSTQ TCQYQELMLSMGFKTEVIIA ILICIMVIFGFIFLTLGLKQ RRKQILFPEKSEDFRENIFR YDDEGGGEEDTEAFDVAALR SSTIMRERKTRKTTSAEIRS LYRQSLQVGPDSAIFRKFIL EKLEEADTDPCAPPFDSLQT YAFEGTGSLAGSLSSLESAV SDQDESYDYLNELGPRFKRL ACMFGSAVQSNN 947 Cyno Macaca nt ATGAATTGTTATTTACTGCT Cadherin- fasci- GCCTTTTATGTTGGGAATTC 19 cularis CTCTCCTATGGCCTTGTCTT GGAGCAACAGAAAACTCTCA AACAAAGAAAGTCCAGCAGC CAGTAGGATCTCATCTGAGA GTGAAGCGTGGCTGGGTGTG GAACCAATTTTTTGTACCAG AGGAAATGAATACGACTAGT CATCACGTTGGCCGGCTAAG ATCTGATTTAGACAATGGAA ACAATTCTTTCCAGTACAAG CTTTTGGGAGCTGGAGCTGG AAGTACTTTTATCATTGATG AAAGAACAGGTGACATATAT GCCATAGAGAAGCTTGATAG AGAGGAGCGATCCCTCTACA TCTTAAGAGCCCAGGTAATA GACATCACTACTGGAAGGGC TGTGGAACCTGAGTCTGAGT TTGTCATCAAAGTTTCGGAT ATCAATGACAATGAACCAAA ATTCCTAGATGAACCTTATG AGGCCATTGTACCAGAGATG TCTCCAGAAGGAACATTAGT CATCCAGGTGACAGCAAGTG ATGCTGATGACCCTTCAAGT GGTAATAATGCTCGTCTCCT CTACAGCTTATTACAAGGCC AGCCATATTTTTCTGTTGAA CCAACAACAGGAGTCATAAG AATATCTTCTAAAATGGATA GAGAACTGCAAGATGAGTAT TGGGTAATCATTCAAGCCAA GGACATGATTGGTCAGCCAG GAGCGTTGTCTGGAACAACG AGTGTATTAATTAAACTTTC AGATGTTAATGACAATAAGC CTATATTTAAAGAAAGTTTA TACCGCCTGACGGTCTCTGA ATCTGCACCCACTGGGACTT CTATAGGAACAATCATGGCA TATGATAATGACATAGGAGA GAATGCAGAAATGGATTACA GCATTGAAGAGGATGATTCA CAGACATTTGACATTATTAC
TAATCATGAAACTCAAGAAG GAATAGTTATATTAAAAAAG AAAGTGAATTTTGAGCACCA GAACCACTATGGTATTAGAG CAAAAGTTAAAAACCATCAT GTTGATGAGCAGCTCATGAA ATACCACACTGAAGCTTCCA CCACTTTCATTAAGATCCAG GTGGAAGATGTTGATGAGCC TCCTCTTTTCCTCCTTCCGT ATTACATATTTGAAATTTTT GAAGAAACCCCACAAGGATC ATTTGTAGGCGTGGTGTCTG CCACAGACCCAGACAATAGG AAATCTCCTATCAGGTATTC TATTACTAGGAGCAAAGTGT TCAATATCGATGATAATGGT ACAATCACTACAACTAACTC ACTGGATCGGGAAATCAGTG CTTGGTACAACCTAAGTATT ACAGCCACAGAAAAATACAA TATAGAGCAGATCTCTTCGA TCCCAGTGTATGTGCAAGTT CTTAATATCAATGATCATGC TCCTGAGTTCTCTCAATACT ATGAGAGTTATGTTTGTGAA AATGCAGGCTCTGGTCAGGT AATTCAGACTATCAGTGCAG TGGATAGAGATGAATCCATA GAAGAGCACCATTTTTACTT TAATCTATCTGTAGAAGACA CTAACTCTTCAAGTTTTACA ATCATAGACAATCAAGATAA CACAGCTGTCATTTTGACTA ATAGAACTGGTTTTAACCTT CAAGAAGAGCCCATCTTCTA CATCTCCATCTTAATTGCCG ACAATGGAATCCCGTCACTT ACAAGTACAAACACCCTTAC CATCCATGTCTGTGACTGTG ATGACAGTGGGAGCACACAG ACCTGCCAGTACCAGGAGCT TATGCTTTCCATGGGATTCA AGACAGAAGTCATCATTGCT ATTCTCATTTGCATTATGGT AATATTTGGGTTTATTTTTT TGACTTTGGGTTTAAAACAA CGGAGAAAACAGATTCTATT TCCTGAGAAAAG TGAAGATTTCAGAGAGAATA TATTCCGATATGATGACGAA GGGGGTGGAGAAGAAGATAC AGAGGCCTTTGACGTAGCAG CGCTGAGGAGTAGCACCATA ATGCGGGAACGCAAGACTCG GAAAACCACCAGCGCTGAGA TCAGGAGCCTATACAGGCAG TCTTTGCAAGTTGGCCCCGA CAGTGCCATATTCAGGAAGT TCATCCTGGAAAAGCTCGAA GAAGCTGATACTGATCCGTG TGCCCCTCCTTTTGATTCCC TCCAGACCTACGCTTTTGAG GGAACAGGGTCATTAGCTGG ATCCCTGAGCTCCTTAGAAT CAGCTGTCTCTGATCAGGAT GAAAGCTATGATTACCTTAA CGAGTTGGGACCTCGCTTTA AAAGATTAGCATGCATGTTT GGTTCTGCAGTGCAGTCAAA TAATTAG 948 secreted Human aa MNCYLLLRFMLGIPLLWPCL Cadherin- GATENSQTKKVKQPVRSHLR 19 VKRGWVWNQFFVPEEMNTTS ecto- HHIGQLRSDLDNGNNSFQYK domain LLGAGAGSTFIIDERTGDIY (amino AIQKLDREERSLYILRAQVI acids DIATGRAVEPESEFVIKVSD 1-596) INDNEPKFLDEPYEAIVPEM SPEGTLVIQVTASDADDPSS GNNARLLYSLLQGQPYFSVE PTTGVIRISSKMDRELQDEY WVIIQAKDMIGQPGALSGTT SVLIKLSDVNDNKPIFKESL YRLTVSESAPTGTSIGTIMA YDNDIGENAEMDYSIEEDDS QTFDIITNHETQEGIVILKK KVDFEHQNHYGIRAKVKNHH VPEQLMKYHTEASTTFIKIQ VEDVDEPPLFLLPYYVFEVF EETPQGSFVGVVSATDPDNR KSPIRYSITRSKVFNINDNG TITTSNSLDREISAWYNLSI TATEKYNIEQISSIPLYVQV LNINDHAPEFSQYYETYVCE NAGSGQVIQTISAVDRDESI EEHHFYFNLSVEDTNNSSFT IIDNQDNTAVILTNRTGFNL QEEPVFYISILIADNGIPSL TSTNTLTIHVCDCGDSGSTQ TCQYQELVLSMGFKTE 949 secreted Human nt atgaactgttatttactgct Cadherin- gcgttttatgttgggaattc 19 ctctcctatggccttgtctt ecto- ggagcaacagaaaactctca domain aacaaagaaagtcaagcagc (amino cagtgcgatctcatttgaga acids gtgaagcgtggctgggtgtg 1-596) gaaccaattttttgtaccag aggaaatgaatacgactagt catcacatcggccagctaag atctgatttagacaatggaa acaattctttccagtacaag cttttgggagctggagctgg aagtacttttatcattgatg aaagaacaggtgacatatat gccatacagaagcttgatag agaggagcgatccctctaca tcttaagagcccaggtaata gacatcgctactggaagggc tgtggaacctgagtctgagt ttgtcatcaaagtttcggat atcaatgacaatgaaccaaa attcctagatgaaccttatg aggccattgtaccagagatg tctccagaaggaacattagt tatccaggtgacagcaagtg atgctgacgatccctcaagt ggtaataatgctcgtctcct ctacagcttacttcaaggcc agccatatttttctcttgaa ccaacaacaggagtcataag aatatcttctaaaatggata gagaactgcaagatgagtat tgggtaatcattcaagccaa ggacatgattggtcagccag gagcgttgtctggaacaaca agtgtattaattaaactttc agatgttaatgacaataagc ctatttttaaagaaagttta taccgcttgactgtctctga atctgcacccactgggactt ctataggaacaatcatggca tatgataatgacataggaga gaatgcagaaatggattaca gcattgaagaggatgattcg caaacatttgacattattac taatcatgaaactcaagaag gaatagttatattaaaaaag aaagtggattttgagcacca gaaccactacggtattagag caaaagttaaaaaccatcat gttcctgagcagctcatgaa gtaccacactgaggcttcca ccactttcattaagatccag gcggaagatgttgatgagcc tcctcttttcctccttccat attatgtatttgaagttttt gaagaaaccccacagggatc atttgtaggcgtggtgtctg ccacagacccagacaatagg aaatctcctatcaggtattc tattactaggagcaaagtgt tcaatatcaatgacaatggt acaatcactacaagtaactc actggatcgtgaaatcagtg cttggtacaacctaagtatt acagccacagaaaaatacaa tatagaacagatctcttcga tcccactgtatgtgcaagtt cttaacatcaatgatcatgc tcctgagttctctcaatact atgagacttatgtttgtgaa aatgcaggctctggtcaggt aattcagactatcagtgcag tggatagagatgaatccata gaagagcaccatttttactt taatctatctgtagaagaca ctaacaattcaagttttaca atcatagataatcaagataa cacagctgtcattttgacta atagaactggttttaacctt caagaagaacctgtcttcta catctccatcttaattgccg acaatggaatcccgtcactt acaagtacaaacacccttac catccatgtctgtgactgtg gtgacagtgggagcacacag acctgccagtaccaggagct tgtgctttccatgggattca agacagaa 950 truncated Human aa MNCYLLLRFMLGIPLLWPCL membrane GATENSQTKKVKQPVRSHLR bound VKRGWVWNQFFVPEEMNTTS form HHIGQLRSDLDNGNNSFQYK of human LLGAGAGSTFIIDERTGDIY cadherin- AIQKLDREERSLYILRAQVI 19 DIATGRAVEPESEFVIKVSD (amino INDNEPKFLDEPYEAIVPEM acids SPEGTLVIQVTASDADDPSS 1-624) GNNARLLYSLLQGQPYFSVE PTTGVIRISSKMDRELQDEY WVIIQAKDMIGQPGALSGTT SVLIKLSDVNDNKPIFKE SLYRLTVSESAPTGTSIGTI MAYDNDIGENAEMDYSIEED DSQTFDIITNHETQEGIVIL KKKVDFEHQNHYGIRAKVKN KHVPEQLMKYHTEASTTFIK IQVEDVDEPPLFLLPYYVFE VFEETPQGSFVGVVSATDPD NRKSPIRYSITRSKVFNIND NGTITTSNSLDREISAWYNL SITATEKYNIEQISSIPLYV QVLNINDHAPEFSQYYETYV CENAGSGQVIQTISAVDRDE SIEEHHFYFNLSVEDTNNSS FTIIDNQDNTAVILTNRTGF NLQEEPVFYISILIADNGIP SLTSTNTLTIHVCDCGDSGS TQTCQYQELVLSMGFKTEVI IAILICIMIIFGFIFLTLGL KQRRKQ 951 truncated Humanl nt atgaactgttatttactgct membrane gcgttttatgttgggaattc bound ctctcctatggccttgtctt form of ggagcaacagaaaactctca human aacaaagaaagtcaagcagc cadherin- cagtgcgatctcatttgaga 19 gtgaagcgtggctgggtgtg (amino gaaccaattttttgtaccag acids aggaaatgaatacgactagt 1-624) catcacatcggccagctaag atctgatttagacaatggaa acaattctttccagtacaag cttttgggagctggagctgg aagtacttttatcattgatg aaagaacaggtgacatatat gccatacagaagcttgatag agaggagcgatccctctaca tcttaagagcccaggtaata gacatcgctactggaagggc tgtggaacctgagtctgagt ttgtcatcaaagtttcggat atcaatgacaatgaaccaaa attcctagatgaaccttatg aggccattgtaccagagatg
tctccagaaggaacattagt tatccaggtgacagcaagtg atgctgacgatccctcaagt ggtaataatgctcgtctcct ctacagcttacttcaaggcc agccatatttttctgttgaa ccaacaacaggagtcataag aatatcttctaaaatggata gagaactgcaagatgagtat tgggtaatcattcaagccaa ggacatgattggtcagccag gagcgttgtctggaacaaca agtgtattaattaaactttc agatgttaatgacaataagc ctatatttaaagaaagttta taccgcttgactgtctctga atctgcacccactgggactt ctataggaacaatcatggca tatgataatgacataggaga gaatgcagaaatggattaca gcattgaagaggatgattcg caaacatttgacattattac taatcatgaaactcaagaag gaatagttatattaaaaaag aaagtggattttgagcacca gaaccactacggtattagag caaaagttaaaaaccatcat gttcctgagcagctcatgaa gtaccacactgaggcttcca ccactttcattaagatccag gtggaagatgttgatgagcc tcctcttttcctccttccat attatgtatttgaagttttt gaagaaaccccacagggatc atttgtaggcgtggtgtctg ccacagacccagacaatagg aaatctcctatcaggtattc tattactaggagcaaagtgt tcaatatcaatgataatggt acaatcactacaagtaactc actggatcgtgaaatcagtg cttggtacaacctaagtatt acagccacagaaaaatacaa tatagaacagatctcttcga tcccactgtatgtgcaagtt cttaacatcaatgatcatgc tcctgagttctctcaatact atgagacttatgtttgtgaa aatgcaggctctggtcaggt aattcagactatcagtgcag tggatagacatgaatccaLa gaagagcaccatttttactt taatctatctgtagaagaca ctaacaattcaagttttaca atcatagataatcaagataa cacagctgtcattttgacta atagaactggttttaacctt caagaagaacctgtcttcta catctccatcttaattgccg acaatggaatcccgtcactt acaagtacaaacacccttac catccatgtctgtgactgtg gtgacagtgggagcacacag acctgccagtaccaggagct tgtgctttccatgggattca agacagaagtcatcattgct attctcatttgcattatgat catatttgggtttatttttt tgactttgggtttaaaacaa cggagaaaacag 952 C137897 artifi- aa GWVWNQFFVPEEMNTTSHHI huCDH19 cial GQLRSDLDNGNNSFQYKLLG (44-141) AGAGSTFIIDERTGDIYAIQ muCDH19 KLDREERSLYILRAQVIDIA (140-770) TGRAVEPESEFVIKVSDIND NEPRFLDEPYEAIVPEMSPE GTFVIKVTANDADDPSTGYH ARILYNLERGQPYFSVEPTT GVIRISSKMDRELQDTYCVI IQAKDMLGQPGALSGTTTVS IKLSDINDNKPIFKESFYRF TISESAPIGTSIGKIMAYDD DIGENAEMEYSIEDDDSKIF DIIIDMDTQEGIVILKKKVD FEQQSYYGIRAKVKNCHVDE ELAPAHVNASTTYIKVQVED EDEPPVFLLPYYILEIPEGK PYGTIVGTVSATDPDRRQSP MRYYLTGSKMFDINDNGTII TTNMLDREVSAWYNLTVTAT ETYNVQQISSAHVYVQVFNI NDNAPEFSQFYETYVCENAE SGEIVQIISAIDRDESIEDH HFYFNHSLEDTNNSSFMLTD NQDNTAVILSNRTGFNLKEE PVFYMIILIADNGIPSLTST NTLTIQVCDCGDSRNTETCA NKGLLFIMGFRTEAIIAIMI CVMVIFGFFFLILALKQRRK ETLFPEKTEDFRENIFCYDD EGGGEEDSEAFDIVELRQST VMRSRKPQRSKSAEIRSLYR QSLQVGPDSAIFRKFILEKL SEAMTDPCAPPFDSLQTFAY EGTGSSAGSLSSLASRDTDQ EDDFDYLNDLGPRFKRLASM FGSAVQPNN 953 C137897 artifi- nt ggctgggtgtggaaccaatt huCDH19 cial ttttgtaccagaggaaatga (44-141) atacgactagtcatcacatc muCDH19 ggccagctaagatctgattt (140-770) agacaatggaaacaattctt tccagtacaagcttttggga gctggagctggaagtacttt tatcattgatgaaagaacag gtgacatatatgccatacag aagcttgatagagaggagcg atccctctacatcttaagag cccaggtaatagacatcgct actggaagggctgtggaacc tgagtctgagtttgtcatca aagtttcggatatcaatgac aatgaacccagattcctaga tgaaccatatgaggccattg tacctgagatgtctccagaa ggaacatttgtcatcaaggt gacagccaatgacgcagatg atccttcaactggctatcat gctcgcatcctatacaactt agaacgaggtcaaccatact tttctgttgagccaacaaca ggagtcataaggatatcttc taagatggatagagagttgc aagatacatactgtgtaatt attcaagccaaggacatgct cggtcagcctggagccttgt ctggaacaacaaccgtatca attaagctgtcagatattaa tgacaacaagccaatattca aagaaagtttctaccgcttc actatatctgaatctgcacc cattggaacatcaataggga aaattatggcatatgatgat gacataggggagaatgcaga gatggagtacagcattgaag atgatgattcaaaaatattt gacataatcattgacaatga cacccaagaagggatagtta tacttaaaaagaaagttgat tttgagcagcagagctatta tggcattagagctaaggtta aaaactgccatgtggatgaa gagcttgcacctgcccatgt taacgcttccacaacctaca ttaaagttcaagtagaagat gaagatgaacctcctgtttt cctcttaccatattacatac ttgaaattcctgaaggaaaa ccatatggaacaattgtggg gacggtttctgccacagacc cagatcgaagacaatctcct atgagatattatctcactgg aagcaaaatgtttgatatca atgacaatggaacaataatc accactaacatgcttgacag agaggtcagtgcttggtaca acttgactgtcacagctact gaaacatacaatgtacaaca gatctcttcagcccatgttt atgtacaagtctttaacatt aacgacaatgctccagagtt ctctcaattctatgagactt atgtttgtgaaaatgctgaa tctggtgagatagttcagat catcagtgcaattgatagag atgagtccatagaagatcac catttttactttaatcactc tctggaagacacaaacaact caagttttatgctaacagac aatcaagataacacagctgt aattctgagtaatagaactg gtttcaatcttaaagaagag cctgtcttctacattgatca tcttgattgctgataacggga tcccatctctcacaagcaca aacactctcactatccaagt ctgtgactgtggagacagta gaaacacagaaacttgtgct aacaagggacttctctttat catgggattcagaacagagg caataattgccatcatgata tgtgttatggtaatatttgg gtttttctttttgattcttg ctctgaaacagcgaagaaag gagactctatttccagagaa gactgaagactttagggaga atatattttgctatgatgat gaaggcggcggggaagaaga ctcggaagcctttgacatcg tagagctgagacaaagtaca gtaatgagagaaagaaagcc tcagagaagcaagagtgcgg agatcaggagcttgtacagg cagtccctgcaggtgggccc agacagtgccatatttcgaa aatttatcccagagaagctt gaagaagccaacacagaccc atgtgctcccccctttgatt cactacagacgtttgcctat gagggaacagggtcaicagc tggctctctgagctccttgg catccagagacactgatcag gaggatgacttcgactacct taatgacctgggacctcgtt ttaaaagattagcaagcatg tttggctctgcagtacaacc caacaattag 954 C137896 artifi- aa GWVWNQFFVPEEMNTTSHHI huCDH19 cial GQLRSDLDNGNNSFQYKLLG (44-249) AGAGSTFIIDERTGDIYAIQ muCDH19 KLDREERSLYILRAQVIDIA (248-770) TGRAVEPESEFVIKVSDIND NEPKFLDSPYEAIVPEMSPE GTLVIQVTASDADDPSSGNN ARLLYSLLQGQPYFSVEPTT GVIRISSKMDRELQDEYWVI IQAKDMIGQPGALSGTTSVL IKLSDVNDNKPIFKESFYRF TISESAPIGTSIGKIMAYDD DIGENAEMEYSIEDDDSKIF DIIIDNDTQEGIVILKKKVD FEQQSYYGIRAKVKNCHVDE ELAPAHVNASTTYIKVQVED EDEPPVFLLPYYILEIPEGK PYGTIVGTVSATDPDRRQSP MRYYLTGSKMFDINDNGTII TTNMLDREVSAWYNLTVTAT ETYNVQQISSAHVYVQVFNI NDNAPEFSQFYETYVCENAE SGEIVQIISAIDRDESIEDH HFYFNHSLEDTNNSSFMLTD NQDNTAVILSNRTGFNLKEE PVFYMIILIADNGIPSLTST NTLTIQVCDCGDSRNTETCA NKGLLFIMGFRTEAIIAIMI CVMVIFGFFFLILALKQRRK ETLFPEKTEDFRENIFCYDD EGGGEEDSEAFDIVELRQST
VMRERKPQRSKSAEIRSLYR QSLQVGPDSAIFRKFILEKL SEANTDPCAPPFDSLQTFAY EGTGSSAGSLSSLASRDTDQ EDDFDYLNDLGPRFKRLASM FGSAVQPNH 955 C137896 artifi- nt ggctgggtgtggaaccaatt huCDH19 cial ttttgtaccagaggaaatga (44-249) atacgactagtcatcacatc muCDH19 ggccagctaagatctgattt (248-770) agacaa tggaaacaattctttccagt acaagcttttgggagctgga gctggaagtacttttatcat tgatgaaagaacaggttgac atatatgccatacagaagct tgatagagaggagcgatccc tctacatcttaagagcccag gtaatagacatcgctactgg aagggctgtggaacctgagt ctgagtttgtcatcaaagtt tcggatatcaatgacaatga accaaaattcctagatgaac cttatgaggccattgtacca gagatgtctccagaaggaac attagttatccaggtgacag caagtgatgctgacgatccc tcaagtggtaataatgctcg tctcctctacagcttacttc aaggccagccatatttttct gttgaaccaacaacaggagt cataagaatatcttctaaaa tggatagagaactgcaagat gagtattgggtaatcattca agccaaggacatgattggtc agccaggagcgttgtctgga acaacaagtgtattaattaa actttcagatgttaatgaca acaagccaatattcaaagaa agtttctaccgcttcactat atctgaatctgcacccattg gaacatcaatagggaaaatt atggcatatgatgatgacat aggggagaatgcagagatgg agtacagcattgaagatgat gattcaaaaatatttgacat aatcattgacaatgacaccc aagaagggatagttatactt aaaaagaaagttgattttga gcagcagagctattatggca ttagagctaaggttaaaaac tgccatgtggatgaagagct tgcacctgcccatgttaacg cttccacaacctacattaaa gttcaagtagaagatgaaga tgaacctcctgttttcctct taccatattacatacttgaa attcctgaaggaaaacca~a tggaacaattgtggggacgg tttctgccacagacccagat cgaagacaatctcctatgag atattatctcactggaagca aaatgtttgatatcaatgac aatggaacaataatcaccac taacatgcttgacagagagg tcagtgcttggtacaacttg actgtcacagctactgaaac atacaatgtacaacagatct cttcagcccatgtttatgta caagtctttaacattaacga caatgctccagagttctctc aattctatgagacttatgtt tgtgaaaatgctgaatctgg tgagatagttcagatcatca gtgcaattgatagagatgag tccatagaagatcaccattt ttactttaatcactctctgg aagacacaaacaactcaagt tttatgctaacagacaatca agataacacagctgtaattc tgagtaatagaactggtttc aatcttaaagaagagcctgt cttctacatgatcatcttga ttgctgataacgggatccca tctctcacaagcacaaacac tctcactatccaagtctgtg actgtggagacagtacaaac acagaaacttgtgctaacaa gcgacttctctttatcatgg gattcagaacagaggcaata attgccatcatgatatgtgt tatggtaatatttgggtttt tctttttgattcttgctctg aaacagcgaagaaaggagac tctatttccagagaagactg aagactttagggagaatata ttttgctatgatgatgaagg cggcggggaagaagactcgg aagcctttgacatcgtagag ctgagacaaagtacagtaat gagagaaagaaagcctcaga gaagcaagagtgcggagatc aggagcttgtacaggcagtc cctgcaggtgggcccagaca gtgccatatttcgaaaattt atcctagagaagcttgaaga agccaacacagacccatgtg ctcccccctttgattcacta cagacgtttgcctatgaggg aacagggtcatcagctggct ctctgagctccttggcatcc agagacactgatcaggagga tgacttcgactaccttaatg acctgggacctcgttttaaa agattagcaagcatgtttgg ctctgcagtacaacccaaca attag 956 C137913 artifi- aa AWVWRPFVVLEEMDDIQCVG muCDH19 cial KLRSDLDNGNNSFQYKLLGI (44-139) GAGSFSINERTGEICAIQKL huCDH19 DREEKSLYILRAQVIDTTIG (142-249) KAVETESEFVIRVLDINDNE muCDH19 PKFLDEPYEAIVPEMSPEGT (248-770) LVIQVTASDADDPSSGNNAR LLYSLLQGQPYFSVEPTTGV IRISSKMDRELQDEYWVIIQ AKDMIGQPGALSGTTSVLIK LSDVNDNKPIFKESFYRFTI SESAPIGTSIGKIMAYDDDI GENAEMEYSIEDDDSKIFDI IIDNDTQEGIVILKKKVDFE QQSYYGIRAKVKNCHVDEEL APAHVNASTTYIKVQVEDED EPPVFLLPYYILEIPEGKPY GTIVGTVSATDPDRRQSPMR YYLTGSKMFDINDNGTIITT NMLDREVSAWYNLTVTATET YNVQQISSAHVYVQVFNIND NAPEFSQFYETYVCENAESG EIVQIISAIDRDESIEDHHF YFNHSLEDTNNSSFMLTDNQ DNTAVILSNRTGFNLKEEPV FYMIILIADNGIPSLTSTNT LTIQVCDCGDSRNTETCANK GLLFIMGFRTEAIIAIMICV MVIFGFFFLILALKQRRKET LFPEKTEDFRENIFCYDDEG GGEEDSEAFDIVELRQSTVM RERKPQRSKSAEIRSLYRQS LQVGPDSAIFRKFILEKLEE ANTDPCAPPFDSLQTFAYEG TGSSAGSLSSLASRDTDQED DFDYLNDLGPRFKRLASMFG SAVQPNN 957 C137913 artifi- nt gcctgggtgtggagaccatt muCDH19 cial tgttgttctagaagaaatgg (44-139) atgatatacaatgtgttgga huCDH19 aagctaagatctgacttaga (142-249) caatggaaacaactctttcc muCDH19 agtacaagctactggggatt (248-770) ggcgctggaagctttagcat taatgaaagaacaggtgaaa tatgtgccatac agaagcttgatagagaggaa aaatccctctacattctgag agcccaggtaatagacacca ctattgggaaggctgtggaa actgaatccgagtttgtcat cagagttttggatatcaatg acaatgaaccaaaattccta gatgaaccttatgaggccat tgtaccagagatgtctccag aaggaacattagttatccag gtgacagcaagtgatgctga cgatccctcaagtggtaata atgctcgtctcctctacagc ttacttcaaggccagccata tttttctgttgaaccaacaa caggagtcataagaatatct tctaaaatggatagagaact gcaagatgagtattgggtaa tcattcaagccaaggacatg attggtcagccaggagcgtt gtctggaacaacaagtgtat taattaaactttcagatgtt aatgacaacaagccaatatt caaagaaagtttctaccgct tcactatatctgaatctgca cccattggaacatcaatagg gaaaattatggcatatgatg atgacataggggagaatgca gagatggagtacagcattga agatgatgattcaaaaatat ttgacataatcattgacaat gacacccaagaagggatagt tatacttaaaaagaaagttg attttgagcagcagagctat tatggcattagagctaaggt taaaaactgccatgtggatg aagagcttgcacctgcccat gttaacgcttccacaaccta cattaaagttcaagtagaag atgaagatgaacctcctgtt ttcctcttaccatattacat acttgaaattcctgaaggaa aaccatatggaacaattgtg gggacggtttctgccacaga cccagatcgaagacaatctc ctatgagatattatctcact ggaagcaaaatgtttgatat caatgacaatggaacaataa tcaccactaacatgcttgac agagaggtcagtgcttggta caacttgactgtcacagcta ctgaaacatacaatgtacaa cagatctcttcagcccatgt ttatgtacaagtctttaaca ttaacgacaatgctccagag ttctctcaa-tctatgagac ttatgtttgtgaaaatgctg aatctggtgagatagttcag atcatcagtgcaattgatag agatgagtccatagaagatc accatttttactttaatcac tctctggaagacacaaacaa ctcaagttttatgctaacag acaatcaagataacacagct gtaattctgagtaatagaac tggtttcaatcttaaagaag agcctgtcttctacatgatc atcttgattgctgataacgg gatcccatctctcacaagca caaacactctcactatccaa gtctgtgactgtggagacag tagaaacacagaaacttgtg ctaacaagggacttctcttt atcatgggattcagaacaga ggcaacaattgccatcatga tatgtgttatggtaatattt gggtttttctttttgattct tgctctgaaacagcgaagaa aggagactctacttccagag aagactgaagactttaggga gaatatattttgctatgatg atgaaggcggcggggaagaa gactcggaagcctttgacat cgtagagctgagacaaagta cagcaatgagagaaagaaag
cctcagagaagcaagagtgc ggagatcaggagcttgtaca ggcagtccctgcaggtgggc ccagacagtgccatatttcg aaaatttatcctagagaagc ttgaagaagccaacacagac ccatgtgctcccccctttga ttcactacagacgtttgcct atgagggaacagggtcatca gctggctctctgagctcctt ggcatccagagacactgatc aggaggatgacttcgactac cttaatgacctgggacctcg ttttaaaagattagcaagca tgtttggctctgcagtacaa cccaacaattag 958 C137847 artifi- aa AWVWRPFVVLEEMDDIQCVG muCDH19 cial KLRSDLDNGNNSFQYKLLGI (44-139) GAGSFSINERTGEICAIQKL huCDH19 DREEKSLYILRAQVIOTTIG (142-364) KAVETESEFVIRVLDINDNE muCDH19 PKFLDEPYEAIVPEMSPEGT (363-770) LVIQVTASDADDPSSGNNAR LLYSLLQGQPYFSVEPTTGV IRISSKMDRELQDEYWVIIQ AKDMIGQPGALSGTTSVLIK LSDVNDNKPIFKESLYRLTV SESAPTGTSIGTIMAYDNDI GENAEMDYSIEEDDSQTFDI ITNHETQEGIVILKKKVDFE HQNHYGIRAKVKNHHVPEQL MKYHTEASTTFIKIQVEDVD EPPVFLLPYYILEIPEGKPY GTIVGTVSATDPDRRQSPMR YYLTGSKMFDINDNGTIITT NMLDREVSAWYNLTVTATET YNVQQISSAHVYVQVFNIND NAPEFSQFYETYVCENAESG EIVQIISAIDRDESIEDHHF YFNHSLEDTNNSSFMLTDNQ DNTAVILSNRTGFNLKEEPV FYMIILIADNGIPSLTSTNT LTIQVCDCGDSRNTETCANK GLLFIMGFRTEAIIAIMICV MVIFGFFFLILALKQRRKET LFPEKTEDFRENIFCYDDEG GGEEDSEAFDIVELRQSTVM RERKPQRSKSAEIRSLYRQS LQVGPDSAIFRKFILEKLEE ANTDPCAPPFDSLQTFAYEG TGSSAGSLSSLASRDTDQED DFDYLNDLGPRFKRLASMFG SAVQPNN 959 C137847 artifi- nt gcctgggtgtggagaccatt muCDH19 cial tgttgttctagaagaaatgg (44-139) atgatatacaatgtgttgga huCDH19 aagctaagatctgacttaga (142-364) caatggaaacaactctttcc muCDH19 agtacaagctactggggatt (363-770) ggcgctggaagctttagcat taatgaaagaacaggtgaaa tatgtgccatacagaagctt gatagagaggaaaaatccct ctacattctgagagcccagg taatagacaccactattggg aaggctgtggaaactgaatc cgagtttgtcatcagagttt tggatatcaatgacaatgaa ccaaaattcctagatgaacc ttatgaggccattgtaccag agatgtctccagaaggaaca ttagttatccaggtgacagc aagtgatgctgacgatccct caagtggtaataatgctcgt ctcctctacagcttacttca aggccagccatatttttctg ttgaaccaacaacaggagtc ataagaatatcttctaaaat ggatagagaactgcaagatg agtattgggtaatcattcaa gccaaggacatgattggtca gccaggagcgttgtctggaa caacaagtgtattaattaaa ctttcagatgttaatgacaa taagcctatatttaaagaaa gtttataccgcttgactgtc tctgaatctgcacccactgg gacttctataggaacaatca tggcatatgataatgacata ggagagaatgcagaaatgga ttacagcattgaagaggatg attcgcaaacatttgacatt attactaatcatgaaactca agaaggaatagttatattaa aaaagaaagtggattttgag caccagaaccactacggtat tagagcaaaagttaaaaacc atcatgttcctgagcagctc atgaagtaccacactgaggc ttccaccactttcattaaga tccaggtggaagatgttgat gaacctcctgttttcctctt accatattacatacttgaaa ttcctgaaggaaaaccatat ggaacaattgtggggacggt ttctgccacagacccagatc gaagacaatctcctatgaga tattatctcactggaagcaa aatgtttgatatcaatgaca atggaacaataatcaccact aacatgcttgacagagaggt cagtgcttggtacaacttga ctgtcacagctactgaaaca tacaatgtacaacagatctc ttcagcccatgtttatgtac aagtctttaacattaacgac aatgctccagagttctctca attctatgagacttatgttt gtgaaaatgctgaatctggt gagatagttcagatcatcag tgcaattgatagagatgagt ccatagaagatcaccatttt tactttaatcactctctgga agacacaaacaactcaagtt ttatgctaacagacaatcaa gataacacagctgtaattct gagtaatagaactggtttca atcttaaagaagagcctgtc ttctacatgatcatcttgat tgctgataacgggatcccat ctctcacaagcacaaacact ctcactatccaagtctgtga ctgtggagacagtagaaaca cagaaacttgtgctaacaag ggacttctctttatcatggg attcagaacagaggcaataa ttgccatcatgatatgtgtt atggtaatatttgggttttt ctttttgattcttgctctga aacagcgaagaaaggagact ctatttccagagaagactga agactttagggagaatatat tttgctatgatgatgaaggc ggcggggaagaagactcgga agcctttgacatcgtagagc tgagacaaagtacagtaatg agagaaagaaagcctcagag aagcaagagtgcggagatca ggagcttgtacaggcagtcc ctgcaggtgggcccagacag tgccatatttcgaaaattta tcctagagaagcttgaagaa gccaacacagacccatgtgc tcccccctttgattcactac agacgtttgcctatgaggga acagggtcatcagctggctc tctgagctccttggcatcca gagacactgatcaggaggat gacttcgactaccttaatga cctgggacctcgttttaaaa gattagcaagcatgtttggc tctgcagtacaacccaacaa ttag 960 C137911 artifi- aa AWVWRPFVVLEEMDDIQCVG muCDH19 cial KLRSDLDNGNNSFQYKLLGI (44-247) GAGSFSINERTGEICAIQKL huCDH19 DREEKSLYILRAQVIDTTIG (250-364) KAVETESEFVIRVLDINDNE muCDH19 PRFLDEPYEAIVPEMSPEGT (363-770) FVIKVTANDADDPSTGYHAR ILYNLERGQPYFSVEPTTGV IRISSKMDRELQDTYCVIIQ AKDMLGQPGALSGTTTVSIK LSDINDNKPIFKESLYRLTV SESAPTGTSIGTIMAYDNDI GENAEMDYSIEEDDSQTFDI ITNHETQEGIVILKKKVDFE HQNHYGIRAKVKNHHVPEQL MKYHTEASTTFIKIQVEDVD EPPVFLLPYYILEIPEGKPY GTIVGTVSATDPDRRQSPMR YYLTGSKMFDINDNGTIITT NMLDREVSAWYNLTVTATET YNVQQISSAHVYVQVFNIND NAPEFSQFYETYVCENAESG EIVQIISAIDRDESIEDHHF YFNHSLEDTNNSSFMLTDNQ DNTAVILSNRTGFNLKEEPV FYMIILIADNGIPSLTSTNT LTIQVCDCGDSRNTETCANK GLLFIMGFRTEAIIAIMICV MVIFGFFFLILALKQRRKET LFPEKTEDFRENIFCYDDEG GGEEDSEAFDIVELRQSTVM RERKPQRSKSAEIRSLYRQS LQVGPDSAIFRKFILEKLEE ANTDPCAPPFDSLQTFAYEG TGSSAGSLSSLASRDTDQED DFDYLNDLGPRF 961 C137911 artifi- nt gcctgggtgtggagaccatt muCDH19 cial tgttgttctagaagaaatgg (44-247) atgatatacaatgtgttgga huCDH19 aagctaagatctgacttaga (250-364) caatggaaacaactctttcc muCDH19 agtacaagctactggggatt (363-770) ggcgctggaagctttagcat taatgaaagaacaggtgaaa tatgtgccatacagaagctt gatagagaggaaaaatccct ctacattctgagagcccagg taatagacaccactattggg aaggctgtggaaactgaatc cgagtttgtcatcagagttt tggatatcaatgacaatgaa cccagattcctagatgaacc atatgaggccattgtacctg agat gtctccagaaggaacatttg tcatcaaggtgacagccaat gacgcagatgatccttcaac tggctatcatgctcgcatcc tatacaacttagaacgaggt caaccatacttttctgttga gccaacaacaggagtcataa ggatatcttctaagatggat agagagttgcaagatacata ctgtgtaattattcaagcca aggacatgctcggtcagcct ggagccttgtctggaacaac aaccgtatcaattaagctgt cagatattaatgacaataag cctatatttaaagaaagttt ataccgcttgactgtctctg aatctgcacccactgggact tctataggaacaatcatggc atatgataatgacataggag agaatgcagaaatggattac agcattgaagaggatgattc gcaaacatttgacattatta ctaatcatgaaactcaagaa ggaatagttatattaaaaaa gaaagtggattttgagcacc agaaccactacggtattaga gcaaaagttaaaaaccatca tgttcctgagcagctcatga agtaccacactgaggcttcc accactttcattaagatcca
ggtggaagatgttgatgaac ctcctgttttcctcttacca tattacatacttgaaattcc tgaaggaaaaccatatggaa caattgtggggacggtttct gccacagacccagatcgaag acaatctcctatgagatatt atctcactggaagcaaaatg tttgatatcaatgacaatgg aacaataatcaccactaaca tgcttgacagagaggtcagt gcttggtacaacttgactgt cacagctactgaaacataca atgtacaacagatctcttca gcccatgtttatgtacaagt ctttaacattaacgacaatg ctccagagttctctcaattc tatgagacttatgtttgtga aaatgctgaatctggtgaga tagttcagatcatcagtgca attgatagagatgagtccat agaagatcaccatttttact ttaatctatctgtagaagac actaacaattcaagttttac aatcatagataatcaagata acacagctgtcattttgagt aatagaactggtttcaatct taaagaagagcctgtcttct acatgatcatcttgattgct gataacgggatcccatctct cacaagcacaaacactctca ctatccaagtctgtgactgt ggagacagtagaaacacaga aacttgtgctaacaagggac ttctctttatcatgggattc agaacagaggcaataattgc catcatgatatgtgttatgg taatatttgggtttttcttt ttgattcttgctctgaaaca gcgaagaaaggagactctat ttccagagaagactgaagac tttagggagaatatattttg ctatgatgatgaaggcggcg gggaagaagatacagaggcc tttgatatagcagagctgag gagtagtaccataatgcggg aacgcaagactcggaaaacc acaagcgcggagatcaggag cttgtacaggcagtccctgc aggtgggcccagacagtgcc atatttcgaaaatttatcct agagaagcttgaagaagcca acacagacccatgtgctccc ccctttgattcactacagac gtttgcctatgagggaacag ggtcatcagctggctctctg agctccttagaatcagcagt ctctgatcaggatgaaagct atgattaccttaatgagttg ggacctcgctttaaaagatt agcatgcatgtttggctctg cagtacaacccaacaattag 962 C137917 artifi- aa AWVWRPFvvLEEMDDIQCVG muCDH19 cial KLRSDLDNGNNSFQYKLLGI (44-362) GAGSFSINERTGEICAIQKL huCDH19 DREEKSLYILRAQVIDTTIG (365-772) KAVETESEFVIRVLDINDNE PRFLDEPYEAIVPEMSPEGT FVIKVTANDADDPSTGYHAR ILYNLERGQPYFSVEPTTGV IRISSKMDRELQDTYCVIIQ AKDMLGQPGALSGTTTVSIK LSDINDNKPIFKESFYRFTI SESAPIGTSIGKIMAYDDDI GENAEMEYSIEDDDSKIFDI IIDNDTQEGIVILKKKVDFE QQSYYGIRAKVKNCHVDEEL APAHVNASTTYIKVQVEDED EPPLFLLPYYVFEVFEETPQ GSFVGvvSATDPDNRKSPIR YSITRSKVFNINDNGTITTS NSLDREISAWYNLSITATEK YNIEQISSIPLYVQVLNIND HAPEFSQYYETYVCENAGSG QVIQTISAVDRDESIEEHHF YFNLSVEDTNNSSFTIIDNQ DNTAVILTNRTGFNLQEEPV FYISILIADNGIPSLTSTNT LTIHVCDCGDSGSTQTCQYQ ELVLSMGFKTEVIIAILICI MIIFGFIFLTLGLKQRRKQI LFPEKSEDFRENIFQYDDEG GGEEDTEAFDIAELRSSTIM RERKTRKTTSAEIRSLYRQS LQVGPDSAIFRKFILEKLEE ANTDPCAPPFDSLQTYAFEG TGSLAGSLSSLESAVSDQDE SYDYLNELGPRFKRLACMFG SAVQSNN 963 C137917 artifi- nt gcctgggtgtggagaccatt muCDH19 cial tgtcgttctagaagaaatgg (44-362) atgatatacaatgtgttgga huCDH19 aagctaagatctgacttaga (365-772) caatggaaacaactctttcc agtacaagctactggggatt ggcgctggaagctttagcat taatgaaagaacaggtgaaa tatgtgccatacagaagctt gatagagaggaaaaatccct ctacattctgagagcccagg taatagacaccactattggg aaggctgtggaaactgaatc cgagtttgtcatcagagttt tggatatcaatgacaatgaa cccagattcctagatgaacc atatgaggccattgtacctg agatgtctccagaaggaaca tttgtcatcaaggtgacagc caatgacgcagatgatcctt caactggctatcatgctcgc atcctataca acttagaacgaggtcaacca tacttttctgttgagccaac aacaggagtcataaggatat cttctaagatggatagagag ttgcaagatacatactgtgt aattattcaagccaaggaca tgctcggtcagcctggagcc ttgtctggaacaacaaccgt atcaattaagctgtcagata ttaatgacaacaagccaata ttcaaagaaagtttctaccg cttcactatatctgaatctg cacccattggaacatcaata gggaaaattatggcatatga tgatgacataggggagaatg cagagatggagtacagcatt gaagatgatgattcaaaaat atttgacataatcattgaca atgacacccaagaagggata gttatacttaaaaagaaagt tgattttgagcagcagagct attatggcattagagctaag gttaaaaactgccatgtgga tgaagagcttgcacctgccc atgttaacgcttccacaacc tacattaaagttcaagtaga agatgaagatgagcctcctc ttttcctccttccatattat gtatttgaagtttttgaaga aaccccacagggaLcatttg taggcgtggtgtctgccaca gacccagacaataggaaatc tcctatcaggtattctatta ctaggagcaaagtgttcaat atcaatgataatggtacaat cactacaagtaactcactgg atcgtgaaatcagtgct~gg tacaacctaagtattacagc cacagaaaaatacaatatag aacagatctcttcgatccca ctgtatgtgcaagttcttaa catcaatgatcatgctcctg agttctctcaatactatgag acttatgtttgtgaaaatgc aggctctggtcaggtaattc agactatcagtgcagtggat agagatgaatccatagaaga gcaccatttttactttaatc tatctgtagaagacactaac aattcaagttttacaatcat agataatcaagataacacag ctgtcattttgactaataga actggttttaaccttcaaga agaacctgtcttctacatct ccattcttaattgccgacaat ggaatcccgtcacttacaag tacaaacacccttaccatcc atgtctgtgactgtggtgac agtgggagcacacagacctg ccagtaccaggagcttgtgc tttccatgggattcaagaca gaagtcatcattgctattct catttgcattatgatcatat ttgggtttatttttttgact ttgggtttaaaacaacggag aaaacagattctatttcctg agaaaagtgaagatttcaga gagaatatattccaatatga tgatgaagggggtggagaag aagatacagaggcctttgat atagcagagctgaggagtag taccataatgcgggaacgca agactcggaaaaccacaagc gctgagatcaggagcctata caggcagtctttgcaagttg gccccgacagtgccatattc aggaaattcattctggaaaa gctcgaagaagctaatactg atccgtgtgcccctcctttt gattccctccagacctacgc ttttgagggaacagggtcat tagctggatccctgagctcc ttagaatcagcagtctctga tcaggatgaaagctatgatt accttaatgagttgggacct cgctttaaaagattagcatg catgtttggttctgcagtgc agtcaaataattag 964 C137915 artifi- aa AWVWRPFWLEEMDDIQCVGK muCDH19 cial LRSDLDNGNNSFQYKLLGIG (44-461) AGSFSINERTGEICAIQKLD huCDH19 REEKSLYILRAQVIDTTIGK (464-772) AVETESEFVIRVLDINDNEP RFLDSPYEAIVPEMSPEGTF VIKVTANDADDPSTGYHARI LYNLERGQPYFSVEPTTGVI RISSKMDRELQDTYCVIIQA KDMLGQPGALSGTTTVSIKL SDINDNKPIFKESFYRFTIS ESAPIGTSIGKIMAYDDDIG ENAEMEYSIEDDDSKIFDil iDNDTQEGIVILKKKVDFSQ QSYYGIRAKVKNCHVDEELA PAHVNASTTYIKVQVEDEDE PPVFLLPYYILEIPEGKPYG TIVGTVSATDPDRRQSPMRY YLTGSKMFDINDN3TIITTN MLDREVSAWYNLTVTATETY NVQQISSAHVYVQVFNINDH APEFSQYYETYVCENAGSGQ VIQTISAVDRDESIEEHHFY FNLSVEDTNNSSFTIIDNQD NTAVILTNRTGFNLQEEPVF YISILIADNGIPSLTSTNTL TIHVCDCGDSGSTQTCQYQE LVLSMGFKTEVIIAILICIM IIFGFIFLTLGLKQRRKQIL FPEKSEDFRENIFQYDDEGG GEEDTEAFDIAELRSSTIMR ERKTRKTTSAEIRSLYRQSL QVGPDSAIFRKFILEKLEEA NTDPCAPPFDSLQTYAFEGT GSLAGSLSSLESAVSDQDES YDYLNELGPRFKRLACMFGS AVQSNN 965 C137915 artifi- nt gcctgggtgtggagaccatt
muCDH19 cial tgttgttctagaagaaatgg (44-461) atgatatacaatgtgttgga huCDH19 aagctaagatctgacttaga (464-772) caatggaaacaactctttcc agtacaagctactggggatt ggcgctggaagctttagcat taatgaaagaacaggtgaaa tatgtgccatacagaagctt gatagagaggaaaaatccct ctacattctgagagcccagg taatagacaccactattggg aaggctgtggaaactgaatc cgagtttgtcatcagagttt tggatatcaatgacaatgaa cccagattcctagatgaacc atatgaggccattgtacctg agatgtctccagaaggaaca tttgtcatcaaggtgacagc caatgacgcagatgatcctt caactggctatcatgctcgc atcctatacaacttagaacg aggtcaaccatacttttctg ttgagccaacaacaggagtc ataaggatatcttctaagat ggatagagagttgcaa gatacatactgtgtaattat tcaagccaaggacatgctcg gtcagcctggagccttgtct ggaacaacaaccgtatcaat taagctgtcagatattaatg acaacaagccaatattcaaa gaaagtttctaccgcttcac tatatctgaatctgcaccca ttggaacatcaatagggaaa attatggcatatgatgatga cataggggagaatgcagaga tggactacagcattgaagat gatgattcaaaaatatttga cataatcattgacaatgaca cccaagaagggatagttata cttaaaaagaaagttgattt tgagcagcagagctattatg gcattagagctaaggttaaa aactgccatgtggatgaaga gcttgcacctgcccatgtta acgcttccacaacctacatt aaagttcaagtagaagatga agatgaacctcctgttttcc tcttaccatattacatactt gaaattcctgaaggaaaacc atatggaacaattgtgggga cggtttctgccacagaccca gatcgaagacaatctcctat gagatattatctcactggaa gcaaaatgtttgaLatcaat gacaatggaacaataatcac cactaacatgcttgacagag aggtcagtgcttggtacaac ttgactgtcacagctactga aacatacaatgtacaacaga tctcttcagcccatgtttat gtacaagtctttaacattaa tgatcatgctcctgagttct ctcaatactatgagacttat gtttgtgaaaatgcaggctc tggtcaggtaattcagacta tcagtgcagtggatagagat gaatccatagaacagcacca tttttactttaatctatctg tagaagacactaacaattca agttttacaatcatagataa tcaagataacacagctgtca ttttgactaatagaactggt tttaaccttcaagaagaacc tgtcttctacatctccatct taattgccgacaatggaatc ccgtcacttacaagtacaaa cacccttaccatccaLgtct gtgactgtggtgacagtggg agcacacagacctgccagta ccaggagctttgtgctttcca tgggattcaagacagaagtc atcattgctattctcatttg cattatgatcatatttgggt ttatttttttgactttgggt ttaaaacaacggagaaaaca gattctatttcctgagaaaa gtgaagatttcagagagaat atattccaatatgatgatga agggcgtggagaagaagata cagaggcctttgatatagca gagctgaggagtagtaccat aatgcgggaacgcaagactc ggaaaaccacaagcgctgag atcaggagcctatacaggca gtctttgcaagttggccccg acagtgccatattcaggaaa ttcattctggaaaagctcga agaagctaatactgatccgt gtgcccctccttttgattcc ctccagacctacgcttttga gggaacagggtcattagctg catccctgagctccttagaa tcagcagtctctgatcagga tgaaagctatgattacctta atgagttgggacctcgcttt aaaagattagcatgcatgtt tggttctgcagtgcagtcaa ataattag 966 C71144 artifi- aa AWVWRPFVVLEEMDDIQCVG muCDH19 cial KLRSDLDNGNNSFQYKLLGI (44-770) GAGSFSINERTGEICAIQKL DREEKSLYILRAQVIDTTIG KAVETESEFVIRVLDINDNE PRFLDEPYEAIVPEMSPEGT FVIKVTANDADDPSTGYHAR ILYNLERGQPYFSVEPTTGV IRISSKMDRELQDTYCVIIQ AKDMLGQPGALSGTTTVSIK LSDINDNKPIFKESFYRFTI SESAPIGTSIGKIMAYDDDI GENAEMEYSIEDDDSKIFDI IIDNDTQEGIVILKKKVDFE QQSYYGIRAKVKNCHVDEEL APAHVNASTTYIKVQVEDED EPPVFLLPYYILEIPEGKPY GTIVGTVSATDPDRRQSPMR YYLTGSKMFDINDNSTIITT NMLDREVSAWYNLTVTATET YNVQQISSAHVYVQVFNIND NAPEFSQFYETYVCENAESG EIVQIISAIDRDESIEDHHF YFNHSLEDTNNSSFMLTDNQ DNTAVILSNRTGFNLKEEPV FYMIILIADNGIPSLTSTNT LTIQVCDCGDSRNTETCANK GLLFIMGFRTEAIIAIMICV MVIFGFFFLILALKQRRKET LFPEKTEDFRENIFCYDDEG GGEEDSEAFDIVELRQSTVM RERKPQRSKSAEIRSLYRQS LQVGPDSAIFRKFILEKLEE ANTDPCAPPFDSLQTFAYEG TGSSAGSLSSLASRDTDQED DFDYLNDLGPRFKRLASMFG SAVQPNN 967 C71144 artifi- nt gcctgggtgtggagaccatt muCDH19 cial tgttgttctagaagaaatgg (44-770) atgatatacaatgtgttgga aagctaagatctgacttaga caatggaaacaactctttcc agtacaagctactggggatt ggcgctggaagctttagcat taatgaaagaacaggtgaaa tatgtgccatacagaagctt gatagagaggaaaaatccct ctacattctgagagcccagg taatagacaccactattggg aaggctgcggaaactgaatc cgagtttgtcatcagagttt tggatatcaatgacaatgaa cccagattcctagatgaacc atatgaggccattgtacctg agatgtctccagaaggaaca tttgtcatcaaggtgacagc caatgacgcagatgatcctt caactggctatcatgctcgc atcctatacaacttagaacg aggtcaaccatacttttctg ttgagccaacaacaggagtc ataaggatatcttctaagat ggatagagagttgcaagata catactgtgtaattattcaa gccaaggacatgctcggtca gcctggagccttgtctggaa caacaaccgtatcaattaag ctgtcagatattaatgacaa caagccaatattcaaagaaa gtttctaccgcttcactata tctgaatctgcacccattgg aacatcaatagggaaaatta tggcatatgatgatgacata ggggagaatgcagagatgga gtacagcattgaagatgatg attcaaaaatatttgacata atcattgacaatgacaccca agaagggatagttatactta aaaagaaagttgattttgag cagcagagccaitatggcat tagagctaaggttaaaaact gccatgtggatgaagagctt gcacctgcccatgttaacgc ttccacaacctacattaaag ttcaagtagaagatgaagat gaacctcctgttttcctctt accatattacatacttgaaa ttcctgaaggaaaaccatat ggaacaattgtggggacggt ttctgccacagacccagatc gaagacaatctcctatgaga tattatctcactggaagcaa aatgtttgatatcaatgaca atggaacaataatcaccact aacatgcttgacagagaggt cagtgcttggtacaacttga ctgtcacagctacLgaaaca tacaatgtacaacagatctc ttcagcccatgtttatgtac aagtctttaacattaacgac aatgctccagagttctctca attctatgagacttatgttt gtgaaaatgctgaatctggt gagatagttcagatcatcag tgcaattgatagagatgagt ccatagaagatcaccatttt tactttaatcactctctgga agacacaaacaactcaagtt ttatgctaacagacaatcaa gataacacagctgtaattct gagtaatagaactggtttca atcttaaagaagagcctgtc ttctacatgatcatcttgat tgctgataacgggatcccat ctctcacaagcacaaacact ctcactatccaagtctgtga ctgtggagacagtagaaaca cagaaacttgtgctaacaag ggacttctctttatcatggg attcagaacagaggcaataa ttgccatcatgatatgtgtt atggtaatatttgggttttt ctttttgatttctttgctctg aaacagcgaagaaaggagact ctatttccagagaagactga agactttagggagaatatat tttgctatgatgatgaaggc ggcggggaagaagactcgga agcctttgacatcgtagagc tgagacaaagtacagtaatg agagaaagaaagcctcagag aagcaagagtgcggagatca ggagcttgtacaggcagtcc ctgcaggtgggcccagacag tgccatatttcgaaaattta tcctagagaagcttgaagaa gccaacacagacccatgtgc tcccccctttgattcactac agacgtttgcctatgaggga acagggtcatcagctggctc
tctgagctccttggcatcca gagacaccgatcaggaggat gacttcgactaccttaatga cctgggacctcgttttaaaa gattagcaagcatgtttggc tctgcagtacaacccaacaa ttag 968 Flag Tag artifi- aa DYKDDDDK cial 969 Flag Tag artifi- nt gactacaaagacgatgacga cial caag 970 ckCDH19 artifi- aa MNCSTFLSLVLALVQLQLCS (1-43):: cial PTTQIFSAQKTDQSYTTIRR FLAG::ckC VKRDYKDDDDKGWVWEPLFV DH19 TEEETSTMPMYVGQLKSDLD (44-776) KEDGSLQYILTGEGADSIFF INEHGKIYVRQKLDREKKSF YILRAQVINRKTRHPIEPDS EFIIKVRDINDHEPQFLDGP YVATVPEMSPEGTSVTQVTA TDGDDPSYGNNARLLYSLIQ GQPYFSVEPKTGVIRMTSQM DRETKDQYLVVIQAKDMVGQ AGAFSATATVTINLSDVNDN PPKFQQRLYYLNVSEEAPVG TTVGRLLAEDSDIGENAAMN YFIEEDSSDVFGIITDRETQ EGIIILKKRVDYESKRKHSV RVKAVNRYIDDRFLKEGPFE DITIVQISVVDADEPPVFTL ESYVMEIAEGVVSGSLVGTV SARDLDNDDSSVRYSIVQGL HLKRLFSINEHNGTIITTEP LDREKASWHNITVTATETRN PEKISEANVYIQVLDVNDHA PEFSKYYETFVCENAVPGQL IQNISAVDKDDSAENHRFYF SLAQATNSSHFTVKDNQDNT AGIFTA3SGFSRKEQFYFFL PILILDNGSPPLTSTNTLTV TVCDCDTEVNTLYCRYGAFL YSIGLSTEALVAVLACLLIL LVFFLAIIGIRQQRKKTLFS EKVEEFRENIVRYDDEGGGE EDTEAFDISALRTRAVLRTH KPRKKITTEIHSLYRQSLQV GPDSAIFRQFISEKLEEANT DPSVPPYDSLQTYAFEGTGS LAGSLSSLGSNTSDVDQNYE YLVGWGPPFKQLAGMYTSQR STRD 971 huCDH19(1- MNCYLLLRFMLGIPLLWPCL 43)::FLAG:: GATENSQTKKVKQPVRSHLR hu(44- VKRDYKDDDDKGWVWNQFFV 141):: PEEMNTTSHHIGQLRSDLDN ckCDH1 GNNSFQYKLLGAGAGSTFII 9(142-776) DERTGDIYAIQKLDREERSL YILRAQVIDIATGRAVEPES EFVIKVSDINDHEPQFLDGP YVATVPEMSPEGTSVTQVTA TDGDDPSYGKNARLLYSLIQ GQPYFSVEPKTGVIRKTSQM DRETKDQYLVVIQAKDMVGQ AGAFSATATVTIKLSDVNDN PPKFQQRLYYLNVSEEAPVG TTVGRLLAEDSDIGENAAMN YFIEEDSSDVFGIITDRETQ EGIIILKKRVDYESKRKHSV RVKAVNRYIDDRFLKEGPFE DITIVQISVVDADEPPVFTL ESYVMEIAEGVVSGSLVGTV SARDLDNDDSSVRYSIVQGL HLKRLFSINE HNGTIITTEPLDREKASWHN ITVTATETRNPEKISEANVY IQVLDVNDHAPEFSKYYETF VCENAVPGQLIQNISAVDKD DSAENHRFYFSLAQATNSSH FTVKDNQDNTAGIFTAGSGF SRKEQFYFFLPILILDNGSP PLTSTNTLTVTVCDCDTEVN TLYCRYGAFLYSIGLSTEAL VAVLACLLILLVFFLAIIGI RQQRKKTLFSEKVEEFRENI VRYDDEGGGEEDTEAFDISA LRTRAVLRTHKPRKKITTEI HSLYRQSLQVGPDSAIFRQF ISEKLEEANTDPSVPPYDSL QTYAFEGTGSLAGSLSSLGS NTSDVDQNYEYLVGWGPPFK QLAGMYTSQRSTRD 972 ckCDH19(1- MNCSTFLSLVLALVQLQLCS 43):: PTTQIFSAQKTDQSYTTIRR FLAG::ckC VKRDYKDDDDKGWVWEPLFV DH19(44- TEEETSTMPMYVGQLKSDLD 141):: KEDGSLQYILTGEGADSIFF huCDH1 INEHGKIYVRQKLDREKKSF 9(142- YILRAQVINRKTRHPIEPDS 249):: EFIIKVRDINDNEPKFLDEP ckCDH1 YEAIVPEMSPEGTLVIQVTA 9(250-776) SDADDPSSGNNARLLYSLLQ GQPYFSVEPTTGVIRISSKM DRELQDEYWVIIQAKDMIGQ PGALSGTTSVLIKLSDVNDN PPKFQQRLYYLNVSEEAPVG TTVGRLLAEDSDIGENAAMN YFIEEDSSDVFGIITDRETQ EGIIILKKRVDYESKRKHSV RVKAVNRYIDDRFLKEGPFE DITIVQISVVDADEPPVFTL ESYVMEIAEGVVSGSLVGTV SARDLDNDDSSVRYSIVQGL HLKRLFSINEHNGTIITTEP LDREKASWHNITVTATETRN PEKISEANVYIQVLDVNDHA PEFSKYYETFVCENAVPGQL IQNISAVDKDDSAENHRFYF SLAQATNSSHFTVKDNQDNT AGIFTAGSGFSRKEQFYFFL PILILDNGSPPLTSTNTLTV TVCDCDTEVNTLYCRYGAFL YSIGLSTEALVAVLACLLIL LVFFLAIIGIRQQRKKTLFS EKVEEFRENIVRYDDEGGGE EDTEAFDISALRTRAVLRTH KPRKKITTEIHSLYRQSLQV GPDSAIFRQFISEKLEEANT DPSVPPYDSLQTYAFEGTGS LAGSLSSLGSNTSDVDQNYE YLVGWGPPFKQLAGMYTSQR STRD 973 ckCDH19(1- MNCSTFLSLVLALVQLQLCS 43):: PTTQIFSAQKTDQSYTTIRR FLAG::ckC VKRDYKDDDDKGWVWEPLFV DH19(44- TEEETSTMPMYVGQLKSDLD 249):: KEDGSLQYILTGEGADSIFF huCDH1 INEHGKIYVRQKLDREKKSF 9(250- YILRAQVINRKTRHPIEPDS 364):: EFIIKVRDINDHEPQFLDGP ckCDH1 YVATVPEMSPEGTSVTQVTA 9(365-776) TDGDDPSYGKNARLLYSLIQ GQPYFSVEPKTGVIRKTSQM DRETKDQYLWIQAKDMVGQA GAFSATATVTINLSDVNDNK PIFKESLYRLTVSESAPTGT SIGTIMAYDNDIGENAEMDY SIEEDDSQTFDIITNHETQE GIVILKKKVDFEHQNHYGIR AKVKNHHVPEQLMKYHTEAS TTFIKIQVEDVDEPPVFTLE SYVMEIAEGWSGSLVGTVSA RDLDNDDSSVRYSIVQGLHL KRLFSINEHNGTIITTEPLD REKASWHNITVTATETRNPE KISEANVYIQVLDVNDHAPE FSKYYETFVCENAVPGQLIQ NISAVDKDDSAENHRFYFSL AQATNSSHFTVKDNQDNTAG IFTAGSGFSRKEQFYFFLPI LILDNGSPPLTSTNTLTVTV CDCDTEVNTLYCRYGAFLYS IGLSTEALVAVLACLLILLV FFLAIIGIRQQRKKTLFSEK VEEFRENIVRYDDEGGGEED TEAFDISALRTRAVLRTHKP RKKITTEIHSLYRQSLQVGP DSAIFRQFISEKLEEANTDP SVPPYDSLQTYAFEGTGSLA GSLSSLGSNTSDVDQNYEYL VGWGPPFKQLAGMYTSQRST RD 974 ckCDH19(1- MNCSTFLSLVLALVQLQLCS 43):: PTTQIFSAQKTDQSYTTIRR FLAG::ckC VKRDYKDDDDKGWVWEPLFV DH19(44- TEEETSTMPMYVGQLKSDLD 364):: KEDGSLQYILTGEGADSIFF huCDH1 INEHGKIYVRQKLDREKKSF 9(365- YILRAQVINRKTRHPIEPDS 463):: EFIIKVRDINDHEPQFLDGP ckCDH1 YVATVPEMSPEGTSVTQVTA 9(469-776) TDGDDPSYGNNARLLYSLIQ GQPYFSVEPKTGVIRMTSQM DRETKDQYLVVIQAKDMVGQ AGAFSATATVTINLSDVNDN PPKFQQRLYYLNVSEEAPVG TTVGRLLAEDSDIGENAAMN YFIEEDSSDVFGIITDRETQ EGIIILKKRVDYESKRKHSV RVKAVNRYIDDRFLKEGPFE DITIVQISVVDADEPPLFLL PYYVFEVFEETPQGSFVGVV SATDPDNRKSPIRYSITRSK VFNINDNGTITTSNSLDREI SAWYNLSITATEKYNIEQIS SIPLYVQVLNINDHAPEFSK YYETFVCENAVPGQLIQNIS AVDKDDSAENHRFYFSLAQA TNSSHFTVKDNQDNTAGIFT AGSGFSRKEQFYFFLPILIL DNGSPPLTSTNTLTVTVCDC DTEVNTLYCRYGAFLYSIGL STEALVAVLACLLILLVFFL AIIGIRQQRKKTLFSEKVEE FRENIVRYDDEGGGEEDTEA FDISALRTRAVLRTHKPRKK ITTEIHSLYRQSLQVGPDSA IFRQFISEKLEEANTDPSVP PYDSLQTYAFEGTGSLAGSL SSLGSNTSDVDQNYEYLVGW GPPFKQLAGMYTSQRSTRD 975 (1- MNCSTFLSLVLALVQLQLCS 43):: PTTQIFSAQKTDQSYTTIRR FLAG::ckC VKRDYKDDDDKGWVWEPLFV DH19(44- TEEETSTMPMYVGQLKSDLD 468):: KEDGSL huCDH1 QYILTGEGADSIFFIKEHGK 9(464-772) IYVRQKLDREKKSFYILRAQ VINRKTRHPIEPDSEFIIKV RDINDHEPQFLEGPYVATVP EMSPEGTSVTQVTATDGDDP SYGNNARLLYSLIQGQPYFS VEPKTGVIRMTSQMDRETKD QYLVVIQAKDMVGQAGAFSA TATVTINLSDVNDNPPKFQQ RLYYLNVSEEAPVGTTVGRL LAEDSDIGENAAMNYFIEED SSDVFGIITDRETQEGIIIL KKRVDYESKRKHSVRVKAVN RYIDDRFLKEGPFEDITIVQ ISVVDADEPPVFTLESYVME IAEGVVSGSLVGTVSARDLD MDDSSVRYSIVQGLHLKRLF SINEHNGTIITTEPLDREKA SWHNITVTATETRNPEKISE ANVYIQVLDVNDHAPEFSQY YETYVCENAGSGQVIQTISA VDRDESIEEHHFYFNLSVED TNNSSFTIIDNQDNTAVILT NRTGFNLQEEPVFYISILIA DNGIPSLTSTNTLTIHVCDC GDSGSTQTCQYQELVLSMGF KTEVIIAILICIMIIFGFIF LTLGLKQRRKQILFPEKSED
FRENIFQYDDEGGGEEDTEA FDIAELRSSTIMRERKTRKT TSAEIRSLYRQSLQVGPDSA IFRKFILEKLEEANTDPCAP PFDSLQTYAFEGTGSLAGSL SSLESAVSDQDESYDYLNEL GPRFKRLACMFGSAVQSNN 976 rhCDH19(1- MNCYLLLPFMLGIPLLKPCL 43):: GATENSQTKKVQQPVGSHLR FLAG::rhC VKRDYKDDDDKGWVWNQFFV DH19 PEEMNTTSHHVGRLRSDIDN (44-772) GNNSFQYKLLGAGAGSTFII DERTGDIYAIEKLDREERSL YILRAQVIDITTGRAVEPES EFVIKVSDINDNEPKFLDEP YEAIVPEMSPEGTLVIQVTA SDADDPSSGNNARLLYSLLQ GQPYFSVEPTTGVIRISSKM DRELQDEYWVIIQAKDMIGQ PGALSGTTSVLIKLSDVNDN KPIFKESLYRLTVSESAPTG TSIGTIMAYDNDIGENAEMD YSIEEDDSQTFDIITNHETQ EGIVILKKKVNFEHQNHYGI RAKVKNHHVDEQLMKYHTEA STTFIKIQVEDVDEPPLFLL PYYIFEIFEETPQGSFVGVV SATDPDNRKSPIRYSITRSK VFNIDDNGTITTTNSLDREI SAWYNLSITATEKYNIEQIS SIPVYVQVLNINDHAPEFSQ YYESYVCENAGSGQVIQTIS AVDRDESIEEHHFYFNLSVE DTNSSSFTIIDNQDNTAVIL TNRTGFNLQEEPIFYISILI ADNGIPSLTSTNTLTIHVCD CDDSGSTQTCQYQELMLSMG FKTEVIIAILICIMVIFGFI FLTLGLKQRRKQILFPEKSE DFRENIFRYDDEGGGEEDTE AFDVAALRSSTIMRERKTRK TTSAEIRSLYRQSLQVGPDS AIFRKFILEKLEEADTDPCA PPFDSLQTYAFEGTGSLAGS LSSLESAVSDQDESYDYLNE LGPRFKRLACMFGSAVQSNM 977 caCDH19(1- QFFVPEEMNKTDYHIGQLRS 42):: DLDNGNNSFQYKLLGAGAGS FLAG::caC IFVIDERTGDIYAIQKLDRE DH19 ERSLYTLRAQVIDSTTGRAV (43-770) EPESEFVIRVSDINDNEPKF LDEPYEAIVPEMSPEGTLVI QVTATDADDPASGNNARLLY SLLQGQPYFSIEPTTGVIRI SSKMDRELQDEYWVIIQAKD MIGLPGALSGTTSVLIKLSD VNDNKPIFKERLYRLTVSES APTGTSIGRIMAYDNDIGEN AEMDYSIEDDSQTFDIITNN ETQEGIVILKKKVDFEHQNH YLIRANVKNRHVAEHLMEYH VEASTTFVRVQVEDEDEPPV FLLPYYLFEILEESPHGSFV GMVSATDPDQRKSPIRYSIT RSKVFSIDDNGTIITTNPLD REISAWYNLSITATEKYNVQ QISAVPVYVQVLNINDHAPE FSEYYDSYVCENAGSGQVIQ TISAVDRDESVEDHHFYFNL SVEDTKNSSFIIIDNEDNTA VILTNRTGFSLQEEPVFYIS VLIADNGIPSLTSTNTLTIH ICDCDDYGSTQTCRDKDLLL SMGFRTEVILAILISIMIIF GFIFLILGLKQRRKPTLFPE KGEDFRENIFRYDDEGGGEE DTEAFDIVQLRSSTIMRERK TRKTAAAEIRSLYRQSLQVG PDSAIFRKFILEKLEEANTD PCAPPFDSLQTYAFEGTGSL AGSLSSLGSAVSDQDENYDY LNELGPRFKRLACMFGSAMQ SNN 978 rhCDH19(1- MNCYLLLPFMLGIPLLWPCL 43): GATENSQTKKVQQPVGSHLR FLAG::rhC VKRDYKDDDDKGWVWNQFFV DH19(44- PEEMNTTSHHVGRLRSDLDN 141):: GNNSFQYKLLGAGAGSTFII caCDH1 DERTGDIYAIEKLDREERSL 9(141-770) YILRAQVIDITTGRAVEPES EFVIKVSDINDNEPKFLDEP YEAIVPEMSPEGTLVIQVTA TDADDPASGNNARLLYSLLQ GQPYFSIEPTTGVIRISSKM DRELQDEYKVIIQAKDMIGL PGALSGTTSVLIKLSDVNDN KPIFKERLYRITVSESAPTG TSIGRIMAYDNDIGENAEMD YSIEDDSQTFDIITNNETQE GIVILKKKVDFEHQNHYLIR ANVKNRHVAEHLMEYHVEAS TTFVRVQVEDEDEPPVFLLP YYLFEILEESPHGSFVGMVS ATDPDQRKSPIRYSITRSKV FSIDDNGTIITTNPLDREIS AWYNLSITATEKYNVQQISA VPVYVQVLNINDHAPEFSEY YDSYVCENAGSGQVIQTISA VDRDESVEDHHFYFNLSVED TKNSSFIIIDNEDNTAVILT NRTGFSLQEEPVFYISVLIA DNGIPSLTSTNTLTIHICDC DDYGSTQTCRDKDLLLSMGF RTEVILAILISIMIIFGFIF LILGLKQRRKPTLFPEKGED FRENIFRYDDEGGGEEDTEA FDIVQLRSSTIMRERKTRKT AAAEIRSL YRQSLQVGPDSAIFRKFILE KLEEANTDPCAPPFDSLQTY AFEGTGSLAGSLSSLGSAVS DQDENYDYLNELGPRFKRLA CMFGSAMQSNN 979 rhCDH19(1- MNCYLLLPFMLGIPLLWPCL 43):: GATENSQTKKVQQPVGSHLR FLAG::rhC VKRDYKDDDOKGWVWNQFFV DH19(44- PEEMNTTSHHVGRLRSDLDN 65):: GNNSFQYKLLGAGAGSIFVI caCDH19 DERTGDIYAIQKLDREERSL (65-770) YTLRAQVIDSTTGRAVEPES EFVIRVSDINDNEPKFLDEP YEAIVPEMSPEGTLVIQVTA TDADDPASGNNARLLYSLLQ GQPYFSIEPTTGVIRISSKM DRELQDEYWVIIQAKDMIGL PGALSGTTSVLIKLSDVNDN KPIFKERLYRLTVSESAPTG TSIGRIMAYDNDIGENAEMD YSIEDDSQTFDIITNNETQE GIVILKKKVDFEHQNHYLIR ANVKNRHVAEHLMEYHVEAS TTFVRVQVEDEDEPPVFLLP YYLFEILEESPHGSFVGMVS ATDPDQRKSPIRYSITRSKV FSIDDNGTIITTNPLDREIS AWYNLSITATEKYNVQQISA VPVYVQVLNINDHAPEFSEY YDSYVCENAGSGQVIQTISA VDRDESVEDHKFYFNLSVED TKNSSFIIIDNEDNTAVILT NRTGFSLQEEPVFYISVLIA DNGIPSLTSTNTLTIHICDC DDYGSTQTCRDKDLLLSMGF RTEVILAILISIMIIFGFIF LILGLKQRRKPTLFPEKGED FRENIFRYDDEGGGEEDTEA FDIVQLRSSTIMRERKTRKT AAAEIRSLYRQSLQVGPDSA IFRKFILEKLEEANTDPCAP PFDSLQTYAFEGTGSLAGSL SSLGSAVSDQDENYDYLNEL GPRFKRLACMFGSAMQSNN 980 caCDH19(1- MNYCFLLPLMLGIPLIWPCF 43):: TASESSKTEVKHQAGSHLRV FLAG::caC KRDYKDDDDKGWMWNQFFVP DH19(44- EEMNKTDYHIGQLRSDLDNG 87):: NNSFQYKLLGAGAGSTFIID rhCDH19(89- ERTGDIYAIEKLDREERSLY 114):: ILRAQVIDSTTGRAVEPESE caCDH1 FVIRVSDINDNEPKFLDEPY 9(115-770) EAIVPEMSPEGTLVIQVTAT DADDPASGNNARLLYSLLQG QPYFSIEPTTGVIRISSKMD RELQDEYWVIIQAKDMIGLP GALSGTTSVLIKLSDVNDNK PIFKERLYRLTVSESAPTGT SIGRIMAYDNDIGENAEMDY SIEDDSQTFDIITNNETQEG IVILKKKVDFEHQNHYLIRA KVKNRHVAEHLMEYHVEAST TFVRVQVEDEDEPPVFLLPY YLFEILEESPHGSFVGMVSA TDPDQRKSPIRYSITRSKVF SIDDNGTIITTNPLDREISA WYNLSITATEKYNVQQISAV PVYVQVLNINDHAPEFSEYY DSYVCENAGSGQVIQTISAV DRDESVEDHHFYFNLSVEDT KNSSFIIIDNEDNTAVILTN RTGFSLQEEPVFYISVLIAD NGIPSLTSTNTLTIHICDCD DYGSTQTCRDKDLLLSMGFR TEVILAILISIMIIFGFIFL ILGLKQRRKPTLFPEKGEDF RENIFRYDDEGGGEEDTEAF DIVQLRSSTIMRERKTRKTA AAEIRSLYRQSLQVGPDSAI FRKFILEKLEEANTDPCAPP FDSLQTYAFEGTGSLAGSLS SLGSAVSDQDENYDYLNELG PRFKRLACMFGSAMQSNN 981 caCDH19(1- MNYCFLLPLMLGIPLIWPCF 43):: TASESSKTEVKHQAGSHLRV FLAG::caC KRDYKDDDDKGWMWNQFFVP DH19(44- EEMNKTDYHIGQLRSDLDNG 120):: NNSFQYKLLGAGAGSIFVID rhCDH1 ERTGDIYAIQKLDREERSLY 9(122- TLRAQVIDITTGRAVEPESE 137):: FVIKVSDINDNEPKFLDEPY caCDH1 EAIVPEMSPEGTLVIQVTAT 9(137-770) DADDPASGNNARLLYSLLQG QPYFSIEPTTGVIRISSKMD RELQDEYWVIIQAKDMIGLP GALSGTTSVLIKLSDVNDNK PIFKERLYRLTVSESAPTGT SIGRIMAYDNDIGENAEMDY SIEDDSQTFDIITNNETQEG IVILKKKVDFEHQNHYLIRA NVKNRHVAEHLMEYHVEAST TFVRVQVEDEDEPPVFLLPY YLFEILEESPHGSFVGMVSA TDPDQRKSPIRYSITRSKVF SIDDNGTIITTNPLDREISA WYNLSITATEKYNVQQISAV PVYVQVLNINDHAPEFSEYY DSYVCENAGSGQVIQTISAV DRDESVEDEHFYFNLSVEDT KNSSFIIIDNEDNTAVILTN RTGFSLQEEPVFYISVLIAD NGIPSLTSTNTLTIHICDCD DYGSTQTCRDKDLLLSMGFR TEVILAILISIMIIFGFIFL ILGLKQRRKPTLFPEKGEDF RENIFRYDDEGGGEEDTEAF DIVQLRSSTIMRERKTRKTA AAEIRSLYRQSLQVGPDSAI FRKFILEKLEEANTDPCAPP FDSLQTYAFEGTGSLAGSLS SLGSAVSDQDENYDYLNELG PRFKRLACMFGSAMQSNN 982 rhCDH19(1- MNCYLLLPFMLGIPLLWPCL 43):: GATENSQTKKVQQPVGSHLR FLAG::rhC VKRDYKDDDDKGWVWNQFFV DH19(44- PEEMNTTSHHVGRLRSDLDN
141):: GNNSFQYKLLGAGAGSTFII raCDH19 DERTGDIYAIEKLDREERSL (140- YILRAQVIDITTGRAVEPES 247):: EFVIKVSDINDNEPRFLDEP rhCDH1 YEAIVPEMSPEGTFVIKVTA 9(250-772) NDADDPTSGYHARILYNLEQ GQPYFSVEPTTGVIRISSKM DRELQDTYCVIIQAKDMLGQ PGALSGTTTISIKLSDINDN KPIFKESLYRLTVSESAPTG TSIGTIMAYDNDIGENAEMD YSIEEDDSQTFDIITNHETQ EGIVILKKKVNFEHQNHYGI RAKV KNHHVDEQLMKYHTEASTTF IKIQVEDVDEPPLFLLPYYI FEIFEETPQGSFVGVVSATD PDNRKSPIRYSITRSKVFNI DDNGTITTTNSLDREISAWY NLSITATEKYNIEQISSIPV YVQVLNINDHAPEFSQYYES YVCENAGSGQVIQTISAVDR DESIEEKHFYFNLSVEDTNS SSFTIIDNQDNTAVILTNRT GFNLQEEPIFYISILIADNG IPSLTSTNTLTIHVCDCDDS GSTQTCQYQELMLSMGFKTE VIIAILICIMVIFGFIFLTL GLKQRRKQILFPEKSEDFRE NIFRYDDEGGGEEDTEAFDV AALRSSTIMRERKTRKTTSA EIRSLYRQSLQVGPDSAIFR KFILEKLEEADTDPCAPPFD SLQTYAFEGTGSLAGSLSSL ESAVSDQDESYDYLNELGPR FKRLACMFGSAVQSNN 983 raCDH19(1- MNHYFLKYWILMVPLIWPCL 43):: KVAETLKIEKAQRAVPSLGR FLAG::raC AKRDYKDDDDKGWVWKQFVV DH19 PEEMDTIQHVGRLRSDLDNG (44-770) NNSFQYKLLGTGDGSFSIDE KTGDIFAMQKLDREKQSLYI LRAQVIDTTIGKAVEPESEF VIRVSDVNDNEPRFLDEPYE AIVPEMSPEGTFVIKVTAND ADDPTSGYHARILYNLEQGQ PYFSVEPTTGVIRISSKMDR ELQDTYCVIIQAKDMLGQPG ALSGTTTISIKLSDINDNKP IFKESFYRFTISESAPSGTT IGKIMAYDDDIGENAEMDYS IEDDESQIFDIVIDNETQEG IVILKKKVDFEHQNHYGIRV KVKNCHVDEELAPAHVNAST TYIKVQVEDEDEPPTFLLPY YIFEIPEGKPYGTMVGTVSA VDPDRRQSPMRYSLIGSKMF DINGNGTIVTTNLLDREVSA WYNLSVTATETYNVQQISSA HVYVQVLNINDHAPEFSQLY ETYVCENAESGEIIQTISAI DRDESIEDHHFYFNHSVEDT NNSSFILTDNQDNTAVILSN RAGFSLKEETVFYMIILIAD NGIPPLTSTNTLTIQVCDCG DSRSTETCTSKELLFIMGFK AEAIIAIVICVMVIFGFIFL ILALKQRRKETLFPEKTEDF RENIFCYDDEGGGEEDSEAF DIIELRQSTVMRERKPRKSR SAEIRSLYRQSLQVGPDSAI FRKFILEKLEEANTDSSAPP FDSLQTFAYEGTGSSAGSLS SLGSSVTDQEDDFDYLNDLG PCFKRLANMFGSAVQPDN 984 (1- MNYCFLKHWILMIPLLWPCL 43):: KVSETLKAEKARRTVPSTWR FLAG::mu AKRDYKDDDDKAWVWRPFVV CDH 19 LEEMDDIQCVGKLRSDLDNG (44-323):: NNSFQYKLLGIGAGSFSINE raCDH19 RTGEICAIQKLDREEKSLYI (324- LRAQVIDTTIGKAVETESEF 327):: VIRVLDINDNEPRFLDEPYE muCDH1 AIVPEMSPEGTFVIKVTAND 9(328-770) ADDPSTGYHARILYNLERGQ PYFSVEPTTGVIRISSKMDR ELQDTYCVIIQAKDMLGQPG ALSGTTTVSIKLSDINDNKP IFKESFYRFTISESAPIGTS IGKIMAYDDDIGENAEMEYS IEDDDSKIFDIIIDNDTQEG IVILKKKVDFEHQNHYGIRA KVKNCHVDEELAPAHVNAST TYIKVQVEDEDEPPVFLLPY YILEIPEGKPYGTIVGTVSA TDPDRRQSPMRYYLTGSKMF DINDNGTIITTNMLDREVSA WYNLTVTATETYNVQQISSA HVYVQVFNINDNAPEFSQFY ETYVCENAESGEIVQIISAI DRDESIEDEHFYFNHSLEDT NNSSFMLTDNQDNTAVILSN RTGFNLKEEPVFYMIILIAD NGIPSLTSTNTLTIQVCDCG DSRNTETCANKGLLFIMGFR TEAIIAIMICVMVIFGFFFL ILALKQRRKETLFPEKTEDF RENIFCYDDEGGGEEDSEAF DIVELRQSTVMRERKPQRSK SAEIRSLYRQSLQVGPDSAI FRKFILEKLEEANTDPCAPP FDSLQTFAYEGTGSSAGSLS SLASRDTDQEDDFDYLNDLG PRFKRLASMFGSAVQPNN 985 muCDH19(1- MNYCFLKHWILMIPLLWPCL 43):: KVSETLKAEKARRTVPSTWR FLAG::mu AKRDYKDDDDKAWVWRPFVV CDH19(44- LEEMDDIQCVGKLRSDLDNG 770):: NNSFQYKLLGIGAGSFSINE raCDH19 RTGEICAIQKLDREEKSLYI (290, 299, LRAQYIDTTIGKAVETESEF 308) VIRVLDINDNEPRFLDEPYE AIVPEMSPEGTFVIKVTAND ADDPSTGYHARILYNLERGQ PYFSVEPTTGVIRISSKMDR ELQDTYCVIIQAKDMLGQPG ALSGTTTVSIKLSDINDNKP IFKESFYRFTISESAPIGTS IGKIMAYDDDIGENAEMEYS IEDDDSKIFDIIIDNDTQEG IVILKKKVDFEQQSYYGIRA KVKNCHVDEELAPAHVNAST TYIKVQVEDEDEPFVFLLPY YILEIPEGKPYGTIVGTVSA TDPDRRQSPMRYYLTGSKMF DINDNGTIITTMMLDREVSA WYNLTVTATETYNVQQISSA HVYVQVFNINDNAPEFSQFY ETYVCENAESGEIVQIISAI DRDESIEDKHFYFNHSLEDT NNSSFMLTDNQDNIAVILSN RTGFNLKEEPVFYMIILIAD NGIPSLTSTNTLTIQVCDCG DSRNTETCANKGLLFIMGFR TEAIIAIMICVMVIFGFFFL ILALKQRRKETLFPEKTEDF RENIFCYDDEGGGEEDSEAF DIVELRQSTVMRERKPQRSK SAEIRSLYRQSLQVGPDSAI FRKFILEKLEEANTDPCAPP FDSLQTFAYEGTGSSAGSLS SLASRDTDQEDDFDYLNDLG PRFKRLASMFGSAVQPNN 986 muCDH19(1- MNYCFLKHWILMIPLLWPCL 43):: KVSETLKAEKARRTVPSTWR FLAG::mu AKRDYKDDDDKAWVWRPFVV CDH19(44- LEEMDDIQCVGKLRSDLDNG 770):: NNSPQYKLLGIGAGSFSINE huCDH1 RTGEICAIQKLDREEKSLYI 9(271) LRAQVIDTTIGKAVETESEF VIRVLDINDNEPRFLDEPYE AIVPEMSPEGTFVIKVTAND ADDPSTGYHARILYNLERGQ PYFSVEPTTGVIRISSKMDR ELQDTYCVIIQAKDMLGQPG ALSGTTTVSIKLSDINDNKP IFKESFYRFTISESAPTGTS IGKIMAYDDDIGENAEMEYS IEDDDSKIFDIIIDNDTQEG IVILKKKVDFEQQSYYGIRA KVKNCHVDEELAPAHVNAST TYIKVQVEDEDEPPVFLLPY YILEIPEGKPYGTIVGTVSA TDPDRRQSPMRYYLTGSKMF DINDNGTIITTNMLDREVSA WYNLTVTATETYNVQQISSA HVYVQVFNINDNAPEFSQFY ETYVCENAESGEIVQIISAI DRDESIEDHHFYFNHSLEDT NNSSFMLTDNQDNTAVILSN RTGFNLKEEPVFYMIILIAD NGIPSLTSTNTLTIQVCDCG DSRNTETCANKGLLFIMGFR TEAIIAIMICVMVIFGFFFL ILALKQRRKETLFPEKTEDF RENIFCYDDEGGGEEDSEAF DIVELRQSTVMRERKPQRSK SAEIRSLYRQSLQVGPDSAI FRKFILEKLEEANTDPCAPP FDSLQTFAYEGTGSSAGSLS SLASRDTDQEDDFDYLNDLG PRFKRLASMFGSAVQPNN
Sequence CWU 0 SQTB SEQUENCE LISTING The patent application
contains a lengthy "Sequence Listing" section. A copy of the
"Sequence Listing" is available in electronic form from the USPTO
web site
(https://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20220127354A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
0 SQTB SEQUENCE LISTING The patent application contains a lengthy
"Sequence Listing" section. A copy of the "Sequence Listing" is
available in electronic form from the USPTO web site
(https://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20220127354A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
References
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.