Tobacco Processing Method
ESSEN; Tomas ; et al.
U.S. patent application number 17/436846 was filed with the patent office on 2022-04-28 for tobacco processing method. The applicant listed for this patent is NICOVENTURES TRADING LIMITED. Invention is credited to David BISHOP, Tomas ESSEN, Dominic Conrad WOODCOCK.
| Application Number | 20220125097 17/436846 |
| Document ID | / |
| Family ID | |
| Filed Date | 2022-04-28 |




| United States Patent Application | 20220125097 |
| Kind Code | A1 |
| ESSEN; Tomas ; et al. | April 28, 2022 |
TOBACCO PROCESSING METHOD
Abstract
A method of processing pH-treated tobacco material comprising contacting at least one pH-treated tobacco material with an effective amount of one or more gases, wherein the one or more gases reduce the pH of the pH-treated tobacco material, and wherein the reduction in pH of the pH-treated tobacco material is reversible. Also provided are methods of storing pH-treated tobacco and the use of a gas to improve the shelf-life or bulk storage time of pH-treated tobacco material.
| Inventors: | ESSEN; Tomas; (London, GB) ; BISHOP; David; (London, GB) ; WOODCOCK; Dominic Conrad; (London, GB) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Appl. No.: | 17/436846 | ||||||||||
| Filed: | February 14, 2020 | ||||||||||
| PCT Filed: | February 14, 2020 | ||||||||||
| PCT NO: | PCT/GB2020/050355 | ||||||||||
| 371 Date: | September 7, 2021 |
| International Class: | A24B 15/28 20060101 A24B015/28; A24B 3/18 20060101 A24B003/18 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Mar 7, 2019 | GB | 1903083.2 |
Claims
1. A method of processing pH-treated tobacco material comprising: contacting at least one pH-treated tobacco material with an effective amount of one or more gases, wherein the one or more gases comprises carbon dioxide, wherein the one or more gases reduce the pH of the pH-treated tobacco material from an initial pH to a lower pH, and wherein the reduction in pH of the pH-treated tobacco material is reversible, such that the pH of the tobacco material is at least partially restored to the initial pH on removal of the one or more gases.
2. The method of claim 1, wherein at least 20% by volume of the one or more gases is carbon dioxide.
3. The method of claim 1, wherein from about 25% to about 100% by volume of the one or more gases is carbon dioxide.
4. The method of claim 1, wherein from about 50% to about 100% by volume of the one or more gases is carbon dioxide.
5. The method of claim 1, wherein the one or more gases consists essentially of carbon dioxide.
6. The method of claim 1, wherein the initial pH of the at least one pH-treated tobacco material is greater than 7.
7. The method of claim 1, wherein the initial pH of the at least one pH-treated tobacco material is from about 8 to about 10.
8. The method of claim 1, wherein the lower pH of the at least one pH-treated tobacco material is less than about 7.
9. A tobacco material obtained by the method of claim 1.
10. A method of storing pH-treated tobacco material, the method comprising: i. placing pH-treated tobacco material in a storage container; and either ii. sealing the storage container; and iii. processing the pH-treated tobacco material according to the method of claim 1; or ii. processing the pH-treated tobacco material according to the method of claim 1; and iii. sealing the storage container.
11. A method of storing pH-treated tobacco material, the method comprising: i. processing pH-treated tobacco material according to the method of claim 1; ii. placing the tobacco material obtained from processing step (i) in a storage container; and iii. sealing the storage container.
12. The method of claim 10, wherein the storage container is a sealed container suitable for bulk storage of the pH-treated tobacco material, or a sealed packaging element suitable for individual storage of the pH-treated tobacco material.
13. A storage container comprising tobacco material obtained by the method of claim 10, wherein the pH of the tobacco material increases upon exposure to normal atmospheric conditions.
14. Use of carbon dioxide to improve the shelf-life or bulk storage time of pH-treated tobacco material, wherein the pH-treated tobacco material is stored in contact with carbon dioxide.
15. The method of claim 6, wherein the lower pH of the at least one pH-treated tobacco material is less than about 7.
16. The method of claim 7, wherein the lower pH of the at least one pH-treated tobacco material is less than about 7.
17. The method of claim 11, wherein the storage container is a sealed container suitable for bulk storage of the pH-treated tobacco material, or a sealed packaging element suitable for individual storage of the pH-treated tobacco material.
18. A storage container comprising tobacco material obtained by the method of claim 11, wherein the pH of the tobacco material increases upon exposure to normal atmospheric conditions.
19. A storage container comprising tobacco material obtained by the method of claim 12, wherein the pH of the tobacco material increases upon exposure to normal atmospheric conditions.
20. A method of storing pH-treated tobacco material, the method comprising: contacting at least one pH-treated tobacco material with an effective amount of one or more gases to provide a pH-treated tobacco material having a reduced pH, wherein the one or more gases comprises carbon dioxide, wherein the one or more gases reduce the pH of the pH-treated tobacco material from an initial pH above about 7 to the reduced pH below about 7; and storing the pH-treated tobacco material having the reduced pH in a sealed storage container; wherein the reduction in pH of the pH-treated tobacco material is reversible, such that the pH of the tobacco material is at least partially restored to a restored pH above about 7 upon removal from the sealed storage container.
Description
[0001] The present application is a National Phase entry of PCT Application No. PCT/GB2020/050355, filed Feb. 14, 2020 which claims priority from GB Patent Application No. 1903083.2 filed Mar. 7, 2019, each of which is hereby fully incorporated herein by reference.
TECHNICAL FIELD
[0002] The present disclosure relates to a method of processing pH-treated tobacco material, for example pH-treated tobacco granules or particles used in tobacco industry products. In addition the present disclosure relates to a pH-treated tobacco material obtained from said method, as well as a method of storing pH-treated tobacco material, and a storage container obtained from said method.
BACKGROUND
[0003] Tobacco industry products include smoking articles such as cigarettes, cigars and the like, which burn tobacco during use, as well as alternatives that release compounds from smokeable material without combusting. Examples of such products are tobacco heating devices or products which release compounds by heating, instead of burning. Tobacco industry products also include electronic aerosol provision systems which generally contain a cartomizer with a reservoir of a liquid formulation, and/or a substrate material such as a tobacco-based product, from which aerosol or vapor is generated for inhalation by a user, for example through heat vaporization. Generally nicotine and often flavorants or flavor agents are present in the liquid of the reservoir and/or the substrate material. However in some countries, local regulations or other reasons such as sensory or otherwise mean that nicotine can only be present in the substrate material.
[0004] Thus in these countries, an aerosol provision system will typically comprise a cartomizer with an aerosol or vapor generating element (a vaporizer), e.g. a heater, arranged to aerosolize a portion of precursor material to generate aerosol in an aerosol generation chamber, and a substrate material containing nicotine. As a user inhales on the system or device and electrical power is supplied to the heater, air is drawn into the device through inlet holes and into the aerosol generation chamber where the air mixes with the aerosolized precursor material. There is then typically a flow path connecting the aerosol generation chamber and an opening in the mouthpiece of the device via the substrate material containing nicotine, so that incoming air drawn through the aerosol generation chamber continues along the flow path via the substrate material, so as to carry aerosol out through the mouthpiece opening for inhalation by the user. The substrate material is often heated by the aerosol or vapor generating element and/or by the aerosol itself, so as to extract nicotine into the aerosol inhaled by the user.
SUMMARY
[0005] According to some embodiments described herein, there is provided a method of processing pH-treated tobacco material comprising contacting at least one pH-treated tobacco material with an effective amount of one or more gases, wherein the one or more gases reduce the pH of the pH-treated tobacco material, and wherein the reduction in pH of the pH-treated tobacco material is reversible.
[0006] The one or more gases may comprise carbon dioxide. For example, at least about 20% by volume of the one or more gases may be carbon dioxide. In one embodiment from about 25% to about 100% by volume of the one or more gases may be carbon dioxide. In another embodiment from about 50% to about 100% by volume of the one or more gases may be carbon dioxide. In another embodiment the one or more gases consist essentially of carbon dioxide, where the expression "consist essentially of" has its normal meaning in the art of including further components which do not materially affect the essential characteristics of the gases.
[0007] The initial pH of the at least one pH-treated tobacco material may be greater than 7, for example from about 8 to about 10. The pH of the pH-treated tobacco material is reduced by the one or more gases, this reduction may be to a pH value of less than 7, for example from about 5 to about 6.5. The skilled person will appreciate, however, that any reduction in pH will be advantageous and the present disclosure is not limited to a specific reduction from the initial pH.
[0008] Also provided is a tobacco material obtained by the processing method described herein.
[0009] In addition, there is provided a method of storing pH-treated tobacco material. The method can comprise (i) placing pH-treated tobacco material in a storage container; and either (ii) sealing the storage container and (iii) processing the pH-treated tobacco material according to the processing method described herein, or (ii) processing the pH-treated tobacco material according to the method described herein, and (iii) sealing the storage container.
[0010] Alternatively, the method can comprise (i) processing pH-treated tobacco material according to the method described herein, (ii) placing the tobacco material obtained from processing step (i) in a storage container, and (iii) sealing the storage container.
[0011] The storage container may be a sealed container suitable for bulk storage of the pH-treated tobacco material, or a sealed packaging element suitable for individual storage of the pH-treated tobacco material. The terms "bulk storage" and "individual storage" are discussed further herein.
[0012] Also provided is a storage container comprising tobacco material obtained by the storage method described herein, wherein the pH of the tobacco material increases upon exposure to normal atmospheric conditions.
[0013] Finally, there is provided the use of carbon dioxide to improve the shelf-life or bulk storage time of pH-treated tobacco material.
[0014] These embodiments are set out in the appended independent and dependent claims. It will be appreciated that features of the dependent claims may be combined with each other and with features of the independent claims in combinations other than those explicitly set out in the claims. Furthermore, the approaches described herein are not restricted to specific embodiments such as those set out below, but include and contemplate any appropriate combinations of features presented herein. For example, a processing method, tobacco material obtained therefrom, a storage method and/or a storage container obtained therefrom may be provided in accordance with approaches described herein which includes any one or more of the various features described below as appropriate.
BRIEF DESCRIPTION OF THE DRAWINGS
[0015] Embodiments of the disclosure will now be described, by way of example only, with reference to the accompanying drawings, in which:
[0016] FIG. 1 is a highly schematic cross-section of a storage container 1 in accordance with certain embodiments of the disclosure. As is discussed in more detail below, FIG. 1 shows a sealed storage container 1 comprising pH-treated tobacco material 2 and one or more gases 3 which consist essentially of carbon dioxide. It will be appreciated by the person skilled in the art that FIG. 1 is not drawn to scale.
[0017] FIGS. 2 to 4 are flow-charts showing the key steps of the storage method according to certain embodiments of the disclosure.
DETAILED DESCRIPTION
[0018] Aspects and features of certain examples and embodiments are discussed and described herein. Some aspects and features of certain examples and embodiments may be implemented conventionally and these are not discussed or described in detail in the interests of brevity. It will thus be appreciated that aspects and features of apparatus and methods discussed herein which are not described in detail may be implemented in accordance with any conventional techniques for implementing such aspects and features.
[0019] The present disclosure provides a method of processing pH-treated tobacco material, a tobacco material obtained by said processing method, a method of storing pH-treated tobacco material, a storage container comprising tobacco material obtained by said storage method, and the use of carbon dioxide. These aspects of the present disclosure are surprisingly able to improve the shelf-life or bulk storage time of pH-treated tobacco material, for example, pH-treated tobacco granules or particles used in tobacco industry products.
[0020] As used herein, the term "tobacco industry product" is intended to include:
[0021] combustible smoking articles, such as cigarettes, cigarillos, cigars, and tobacco for pipes or for roll-your-own or for make-your-own cigarettes (whether based on tobacco, tobacco derivatives, expanded tobacco, reconstituted tobacco, tobacco substitutes or other smokeable material);
[0022] non-combustible aerosol provision systems, such as heating devices that release compounds from aerosolizable materials without burning the aerosolizable materials, such as electronic cigarettes, tobacco heating products, and hybrid systems to generate aerosol using a combination of aerosolizable materials;
[0023] articles comprising aerosolizable material and configured to be used within one of these non-combustible aerosol provision systems; and
[0024] aerosol-free stimulant delivery articles, such as lozenges, gums, patches, articles comprising inhalable powders, and smokeless tobacco products such as snus and snuff, wherein the stimulant may or may not comprise nicotine.
[0025] As is common in the art, the terms "vapor" and "aerosol" and related terms such as "vaporize", "volatilize" and "aerosolize" may be used interchangeably. Aerosol provision systems or devices may therefore be referred to herein as "vapor provision systems or devices", "aerosol delivery systems or devices", "electronic vapor provision systems or devices", "electronic aerosol provision systems or devices", or "e-cigarettes or electronic cigarettes". These terms may be used interchangeably and are intended to refer to non-combustible aerosol provision systems or devices such as a heating device that releases compounds from aerosolizable material(s), e.g. tobacco material, without burning the aerosolizable material(s). The non-combustible aerosol provision system can include electronic cigarettes or e-cigarettes that create aerosol from aerosol precursor materials by heating or other techniques such as vibration; and hybrid systems that provide aerosol via a combination of aerosol precursor materials and solid substrate materials, for example hybrid systems containing liquid or gel precursor materials and a solid substrate material.
[0026] In arriving at the present disclosure, the inventors recognized that a tobacco industry product containing pH-treated tobacco material often fails to provide a satisfactory experience to a user when the pH-treated tobacco material has been stored for a significant period of time prior to use. This was observed when the pH-treated tobacco was stored in bulk or in individual compartments, but was particularly prevalent after storage of the tobacco in individual compartments, and the unsatisfactory user experience was primarily due to a reduction in the amount of nicotine available.
[0027] As is known in the art, nicotine has two basic nitrogens and can exist in a un-protonated, a mono-protonated or a di-protonated form. The protonated forms are not volatile, whilst the un-protonated form is highly volatile and is readily able to enter the gas phase with a vapor pressure at 25.degree. C. of 5.5 Pa. To promote the release of nicotine, tobacco material is therefore generally pH treated before being used in tobacco industry products. The pH treatment raises the pH of the tobacco material from an acidic pH (e.g. 5.5) to an alkaline pH (e.g. 9.5) thereby deprotonating the nicotine and converting it into its volatile, free base form as shown below:
##STR00001##
[0028] The inventors realized, however, that the pH treatment and deprotonation of nicotine was causing significant nicotine stability issues when storing the pH-treated tobacco material for a length of time. Although the availability of nicotine from the tobacco material was improved, pH treatment simultaneously reduced the shelf-life of the tobacco due to the volatility of the free-base nicotine.
[0029] Accordingly there is a desire for nicotine stability of a pH-treated tobacco material to be improved, and in particular for the shelf-life and bulk storage time of a pH-treated tobacco material to be increased. The present disclosure addresses these needs whilst improving product performance and user experience by contacting pH-treated tobacco material with an effective amount of one or more gases so as to reversibly reduce the pH of the pH-treated tobacco material. The reversible nature of the present method is particularly advantageous because it allows the methods described herein to be incorporated into existing tobacco processing and treatment technology, and also to be applied to bulk storage and individual (e.g. blister or the like) storage of a pH-treated tobacco material.
[0030] Without wishing to be bound by any one theory, the inventors believe that the reversible reduction in pH by the one or more gases is caused by the reversible formation of a weak acid from the gas(es) and water. When the gas(es) used in the present method includes carbon dioxide, the carbon dioxide is believed to react with water present in the pH-treated tobacco material and/or atmosphere so as to form carbonic acid, lowering the pH and protonating nicotine. On exposure to the normal environment or normal atmospheric conditions, i.e. release of the tobacco material from the storage container, the carbonic acid converts back to carbon dioxide, returning the pH towards its previous level, and deprotonating nicotine. The reversible formation of carbonic acid by carbon dioxide can be represented by the following equilibria:
CO.sub.2(aq)+H.sub.2OH.sub.2CO.sub.3(aq)
H.sub.2CO.sub.3(aq)H.sup.+(aq)+HCO.sub.3.sup.-(aq)
HCO.sub.3.sup.-(aq)H.sup.+(aq)+CO.sub.3.sup.2-(aq)
[0031] For ease of reference, these and further features of the present disclosure are now discussed under appropriate section headings. However, the teachings under each section are not necessarily limited to the section in which they are found.
Tobacco Material
[0032] The present disclosure concerns a pH-treated tobacco material. The terms "pH-treated tobacco material", "pH-treated tobacco substrate" or "pH-treated tobacco" are used interchangeably herein and refer to pH-treated tobacco, i.e. tobacco that has been subject to a pH treatment as is known in the art and described herein. As discussed further below, pH treatment increases the pH of the tobacco. The identity of the tobacco material is not, however, limited.
[0033] The tobacco material before pH treatment is referred to as "tobacco material", "tobacco starting material" or "starting tobacco material".
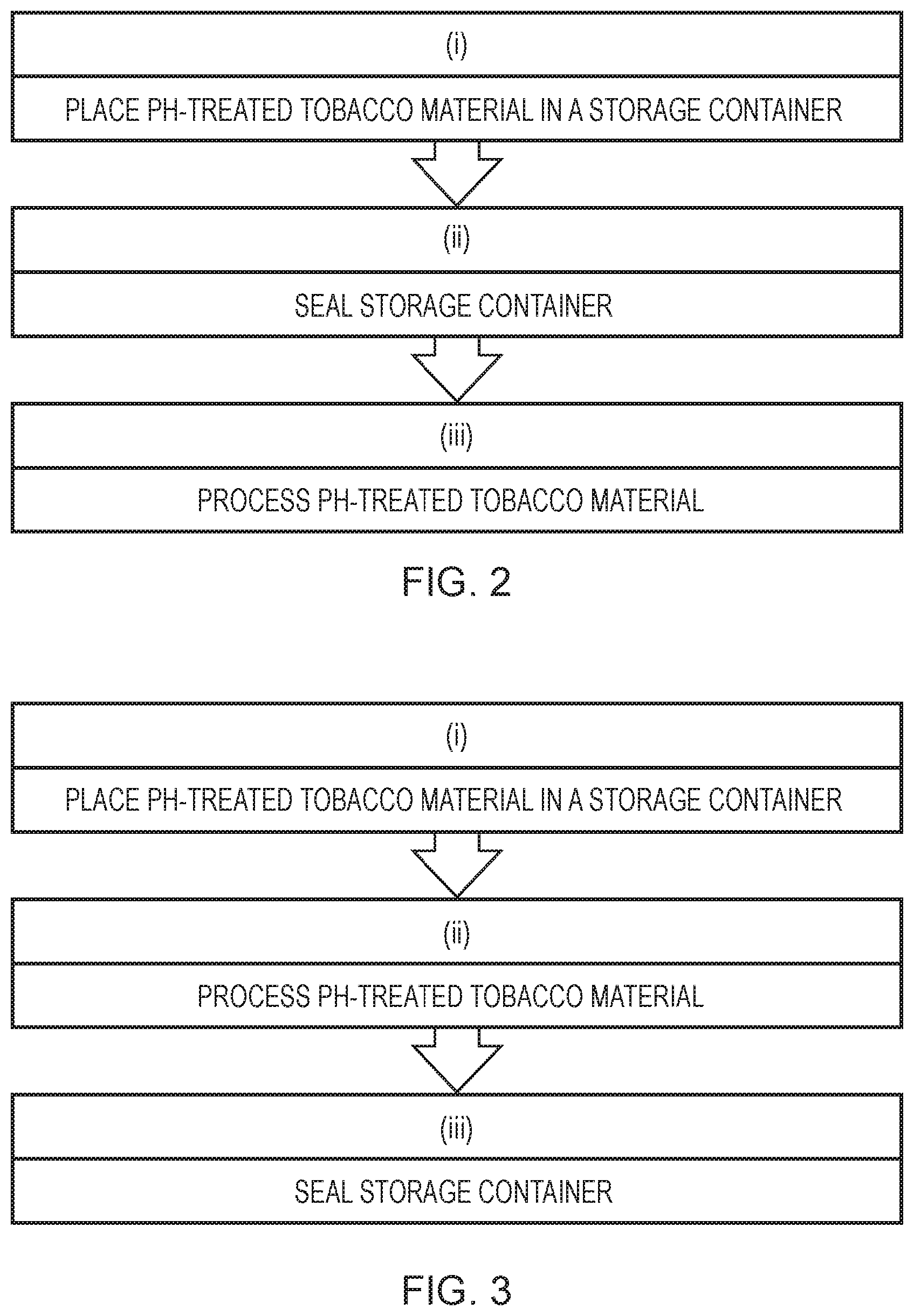
[0034] The tobacco starting material can be any type or grade of tobacco and includes any part, such as for example, the leaves or stems, of any member of the genus Nicotiana and reconstituted materials thereof. The tobacco starting material is, in one embodiment, from the species Nicotiana tabacum.
[0035] The tobacco starting material may be from one variety of tobacco or from more than one variety of tobacco. As is known in the art, the latter can be referred to as a blend. Examples of tobacco varieties which may be used include, but are not limited to, Virginia, Burley, Oriental and Rustica tobaccos. The tobacco starting material may also comprise tobacco of a certain grade or quality. For example, the tobacco starting material may comprise tobacco of high, medium and/or low grade.
[0036] The tobacco starting material may be subjected to known practices such as drying and curing. In one embodiment the tobacco starting material is cured prior to any pH-treatment.
[0037] The tobacco starting material can also be in any suitable form. The tobacco starting material can be solid, liquid or gel. In one embodiment the tobacco starting material is a solid or a gel. In one embodiment the tobacco starting material is a solid. In another embodiment the tobacco starting material is in the form of a gel. In one embodiment the tobacco starting material is a solid and selected from cut-rag, ground tobacco or baled leaf.
[0038] In one embodiment the tobacco starting material is a solid and either prior, at the same time as, or after pH treatment is processed into the form of particles, beads, granules, extrudates or the like, by any method known in the art, e.g. dry or wet granulation, chopping, shredding, cutting, crushing, milling, grinding, extruding or a combination thereof. Smaller tobacco particles, beads, granules, extrudates, or the like have a greater surface area to volume ratio and so are known to exhibit enhanced release of volatile components compared to particles of larger sizes. This is described, for example, in WO2019016535.
[0039] In one embodiment the tobacco starting material is pre-treated or pre-processed into particles, beads, granules or extrudates of the desired size, and then subjected to pH treatment. For example, the tobacco starting material may be ground into particles and then pH-treated as shown in FIG. 1 of WO2019016535.
[0040] In another embodiment the tobacco starting material is pre-processed or pre-treated into particles, beads, granules, extrudates or the like at the same time as pH treatment. The shape and/or size of the tobacco particles are not limited in the context of the present disclosure. The skilled person will be aware of suitable particle sizes and shapes.
[0041] The tobacco starting material used in the present disclosure will include nicotine. The nicotine may be inherently present in the tobacco starting material and/or it may be added thereto. If any nicotine is added to the tobacco starting material, this will typically take place after pH treatment. Nicotine may also be added before pH treatment if so desired.
[0042] The nicotine content of the tobacco starting material is not limited and depends on the desired dosage when the nicotine is aerosolized and inhaled by a user. Generally the nicotine content is between about 0.2 wt % and about 6 wt %, on a dry weight basis relative to the total weight of the tobacco starting material. All of the wt % values for the amount of nicotine are on a dry weight basis.
[0043] In one embodiment nicotine is present in an amount of no greater than about 6 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to about 6 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to about 6 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to about 6 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1.8 to about 6 wt % based on the total weight of the tobacco starting material.
[0044] In one embodiment nicotine is present in an amount of no greater than about 5 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to about 5 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to about 5 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to about 5 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1.8 to about 5 wt % based on the total weight of the tobacco starting material.
[0045] In one embodiment nicotine is present in an amount of no greater than about 4 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to about 4 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to about 4 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to about 4 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1.8 to about 4 wt % based on the total weight of the tobacco starting material.
[0046] In one embodiment nicotine is present in an amount of no greater than about 3 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to about 3 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to about 3 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to about 3 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1.8 to about 3 wt % based on the total weight of the tobacco starting material.
[0047] In one embodiment nicotine is present in an amount of no greater than about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of no greater than about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.5 to about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.5 to about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to about 1.8 wt % based on the total weight of the tobacco starting material.
[0048] In one embodiment nicotine is present in an amount of less than about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of less than about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to less than about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.4 to less than about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.5 to less than about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.5 to less than about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to less than about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 0.8 to less than about 1.8 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to less than about 1.9 wt % based on the total weight of the tobacco starting material. In one embodiment nicotine is present in an amount of from about 1 to less than about 1.8 wt % based on the total weight of the tobacco starting material.
[0049] Depending on the application, the tobacco starting material or pH-treated tobacco material can include one or more further components. Such further components may be conventional in the sense that they are typically included in tobacco material or pH-treated tobacco material for tobacco industry products. In one embodiment the one or more further components include an active agent other than nicotine. This active agent can be a compound which has a biological effect on a user when inhaled, and may, for example, may be selected from botanicals or mixtures thereof, which are synthetic or natural in origin.
[0050] The active agent may be a physiologically active material, which is a material intended to achieve or enhance a physiological response. The active substance may for example be selected from nutraceuticals, nootropics and psychoactives. The active substance may be naturally occurring or synthetically obtained. The active substance may comprise for example nicotine, caffeine, taurine, theine, vitamins such as B6 or B12 or C, melatonin, cannabinoids, or constituents, derivatives, or combinations thereof. The active substance may comprise one or more constituents, derivatives or extracts of tobacco, cannabis or another botanical.
[0051] In some embodiments, the active substance comprises nicotine. In some embodiments, the active substance comprises caffeine, melatonin or vitamin B12.
[0052] As noted herein, the active substance may comprise or be derived from one or more botanicals or constituents, derivatives or extracts thereof. As used herein, the term "botanical" includes any material derived from plants including, but not limited to, extracts, leaves, bark, fibres, stems, roots, seeds, flowers, fruits, pollen, husk, shells or the like. Alternatively, the material may comprise an active compound naturally existing in a botanical, obtained synthetically. The material may be in the form of liquid, gas, solid, powder, dust, crushed particles, granules, pellets, shreds, strips, sheets, or the like. Example botanicals are tobacco, eucalyptus, star anise, hemp, cocoa, cannabis, fennel, lemongrass, peppermint, spearmint, rooibos, chamomile, flax, ginger, Ginkgo biloba, hazel, hibiscus, laurel, licorice (liquorice), matcha, mate, orange skin, papaya, rose, sage, tea such as green tea or black tea, thyme, clove, cinnamon, coffee, aniseed (anise), basil, bay leaves, cardamom, coriander, cumin, nutmeg, oregano, paprika, rosemary, saffron, lavender, lemon peel, mint, juniper, elderflower, vanilla, wintergreen, beefsteak plant, curcuma, turmeric, sandalwood, cilantro, bergamot, orange blossom, myrtle, cassis, valerian, pimento, mace, damien, marjoram, olive, lemon balm, lemon basil, chive, carvi, verbena, tarragon, geranium, mulberry, ginseng, theanine, theacrine, maca, ashwagandha, damiana, guarana, chlorophyll, baobab or any combination thereof. The mint may be chosen from the following mint varieties: Mentha arventis, Mentha c.v., Mentha niliaca, Mentha piperita, Mentha piperita citrata c.v., Mentha piperita c.v., Mentha spicata crispa, Mentha cardifolia, Memtha longifolia, Mentha suaveolens variegata, Mentha pulegium, Mentha spicata c.v. and Mentha suaveolens.
[0053] In some embodiments, the active substance comprises or is derived from one or more botanicals or constituents, derivatives or extracts thereof and the botanical is tobacco.
[0054] In some embodiments, the active substance comprises or derived from one or more botanicals or constituents, derivatives or extracts thereof and the botanical is selected from eucalyptus, star anise, cocoa and hemp.
[0055] In some embodiments, the active substance comprises or derived from one or more botanicals or constituents, derivatives or extracts thereof and the botanical is selected from rooibos and fennel.
[0056] The one or more further components may also include one or more flavors or flavoring agents. Such flavors or flavoring agents are known in the art, and may be inherently present in the tobacco starting material or may be added to the tobacco starting material or pH-treated tobacco material. As used herein, the terms "flavor" and "flavorant" refer to materials which, where local regulations permit, may be used to create a desired taste, aroma or other somatosensorial sensation in a product for adult consumers.
[0057] They may include naturally occurring flavor materials, botanicals, extracts of botanicals, synthetically obtained materials, or combinations thereof. For example, such flavors or flavoring agents may be selected from the group consisting of liquorice, hydrangea, Japanese white bark magnolia leaf, chamomile, fenugreek, clove, menthol, Japanese mint, aniseed, cinnamon, herb, wintergreen, cherry, berry, peach, apple, spearmint, peppermint, lavender, cardamom, celery, cascarilla, Drambuie, bourbon, scotch, whiskey, nutmeg, sandalwood, bergamot, geranium, honey essence, rose oil, vanilla, lemon oil, orange oil, cassia, caraway, cognac, jasmine, ylang-ylang, sage, fennel, piment, ginger, anise, coriander, coffee, flavor enhancers, bitterness receptor site blockers, sensorial receptor site activators or stimulators, sugars and/or sugar substitutes (e.g. sucralose, acesulfame potassium, aspartame, saccharine, cyclamates, lactose, sucrose, glucose, fructose, sorbitol, or mannitol), and other additives such as charcoal, chlorophyll, minerals, botanicals, or breath freshening agents.
[0058] The flavor(s) or flavoring agent(s) may be imitation, synthetic or natural ingredients or blends thereof, and may be in any suitable form, for example, oil, liquid, or powder, or liquid such as an oil, solid such as a powder or gas. It will be understood by the skilled person that the flavor(s) or flavoring agent(s) may be a multi-component flavor or flavoring agent or a single component flavor or flavoring agent.
[0059] In some embodiments, the flavor comprises menthol, spearmint and/or peppermint. In some embodiments, the flavor comprises flavor components of cucumber, blueberry, citrus fruits and/or red berry. In some embodiments, the flavor comprises eugenol. In some embodiments, the flavor comprises flavor components extracted from tobacco. In some embodiments, the flavor comprises flavor components extracted from cannabis.
[0060] In some embodiments, the flavor may comprise a sensate, which is intended to achieve a somatosensorial sensation which are usually chemically induced and perceived by the stimulation of the fifth cranial nerve (trigeminal nerve), in addition to or in place of aroma or taste nerves, and these may include agents providing heating, cooling, tingling, numbing effect.
[0061] A suitable heat effect agent may be, but is not limited to, vanillyl ethyl ether and a suitable cooling agent may be, but not limited to eucalyptol, WS-3.
[0062] With respect to the flavor(s) inherently present in the tobacco starting material, their release to a user from the pH-treated tobacco material may be enhanced by further treatment. The skilled person will know of suitable treatments and appreciate that such treatment can take place before, during or after the pH treatment of the tobacco starting material.
[0063] In one embodiment the pH-treated tobacco material does not undergo any further treatment.
[0064] In one embodiment the tobacco starting material or pH-treated tobacco material does not contain any added acid(s). In one embodiment the tobacco starting material or pH-treated tobacco material does not contain any added organic acid selected from the group consisting of benzoic acid, levulinic acid, malic acid, maleic acid, fumaric acid, citric acid, lactic acid, acetic acid, succinic acid, and mixtures thereof.
pH-Treated Tobacco Material
[0065] As discussed above, the terms "pH-treated tobacco material", "pH-treated tobacco substrate" and "pH-treated tobacco" are used interchangeably herein to refer to a tobacco starting material which has undergone a treatment so as to increase its pH. The level of increase is not limited because it will depend on the initial pH of the tobacco starting material, and this depends on numerous factors including for example, the type of tobacco, pH treatment agent and the relative amounts of the tobacco starting material and pH treatment agent.
[0066] The pH-treated tobacco material may be in any form, e.g. solid, liquid or gel. In one embodiment the pH-treated tobacco material is a solid or a gel. In another embodiment the pH-treated tobacco material is a solid. In another embodiment the pH-treated tobacco material is a gel. The form of the tobacco material is discussed in detail above (including the formation of particles, granules, beads and extrudates by methods known in the art and discussed in WO2019016535) and the skilled person would understand that the form of the tobacco starting material dictates the form of the pH-treated tobacco material.
[0067] In one embodiment the pH treatment modifies the pH of the tobacco starting material from an acidic pH (less than 7) to an alkaline pH (greater than 7). In another embodiment the pH treatment modifies the pH so that the nicotine present in the tobacco starting material is deprotonated. The skilled person will know of suitable techniques to detect the nicotine protonation and/or deprotonation level(s), e.g. .sup.1H NMR spectroscopy, and thus be able to readily monitor and control the pH treatment.
[0068] Generally the normal, unadjusted pH of cured tobacco is slightly acidic, usually within the range of about 4.5 to about 6.5 and frequently approximately 5, for example 5.5. A pH treatment will then increase the pH to a basic pH of greater than 7. Thus in one embodiment the pH treatment modifies the pH of the tobacco starting material from a pH in the range of about 4 to less than 7, to a pH of greater than 7. In another embodiment the pH treatment modifies the pH of the tobacco starting material from a pH in the range of about 5 to less than 7, to a pH of greater than 7.
[0069] As is known in the art, the pH of tobacco is measured using the CORESTA Recommended Method No. 69, "Determination of pH of Tobacco and Tobacco Products" (2017). This Recommended Method is applicable for the determination of pH in tobacco and tobacco products that are in the range 4 to 10, and involves preparing an aqueous extract of the tobacco or tobacco product sample and its pH determined using a pH electrode. The CORESTA Recommended Method No. 69, "Determination of pH of Tobacco and Tobacco Products" (2017) is incorporated herein by reference.
[0070] Considering now the pH of the pH-treated tobacco material in more detail and noting that these pH values can be combined with the above disclosure of the pH of the tobacco starting material: in one embodiment the pH-treated tobacco material has a pH of about 7 to about 14. In another embodiment the pH-treated tobacco material has a pH of about 7 to about 13. In another embodiment the pH-treated tobacco material has a pH of about 7 to about 12. In another embodiment the pH-treated tobacco material has a pH of about 7 to about 11. In another embodiment the pH-treated tobacco material has a pH of about 7 to about 10.
[0071] In one embodiment the pH-treated tobacco material has a pH of greater than about 8. In another embodiment the pH-treated tobacco material has a pH of about 8 to about 14. In another embodiment the pH-treated tobacco material has a pH of about 8 to about 13. In another embodiment the pH-treated tobacco material has a pH of about 8 to about 12. In another embodiment the pH-treated tobacco material has a pH of about 8 to about 11. In another embodiment the pH-treated tobacco material has a pH of about 8 to about 10.
[0072] In one embodiment the pH-treated tobacco material has a pH of about 9.5.
[0073] In another embodiment the pH-treated tobacco material has a pH of about 8.5.
[0074] Any pH treatment known in the art may be used to prepare the pH-treated tobacco material for the present disclosure, and the skilled person will be aware of suitable treatment methods. The pH treatment can, for example, involve applying a basic solution to the tobacco starting material. Such a method is described in WO2015025158 and is incorporated herein by reference. The basic solution can be an aqueous sodium carbonate solution or any other suitable basic solution such as sodium hydroxide or sodium phosphate.
[0075] As discussed above, the pH treatment can also follow pre-processing or pre-treatment of the tobacco starting material into a form with reduced particle size as described in WO2019016535. After pH treatment the tobacco material may be dried to a particular moisture content. Such drying methods are known in the art along with desirable moisture contents, the latter typically depending on the application of the tobacco material.
[0076] Alternatively after pH treatment the tobacco material may be left to cool for a period of time, without external application of refrigeration or the like.
Reversible pH Reduction by One or More Gases
[0077] The present disclosure relies on contacting pH-treated tobacco material as described herein with an effective amount of one or more gases, wherein the one or more gases reduce the pH of the pH-treated tobacco material, and wherein the reduction in pH of the pH-treated tobacco material is reversible.
[0078] By the term "effective amount" is meant an amount of the one or more gases which is suitable to reduce the pH of the pH-treated tobacco material from its initial pH to a lower pH. As noted above, the level of reduction is not crucial because any reduction is advantageous. In one embodiment the effective amount of one or more gases is an amount suitable to reduce the initial pH of the pH-treated tobacco material by at least 0.1 pH. In another embodiment the reduction is at least 0.3 pH. In another embodiment the reduction is at least 0.5 pH. In another embodiment the reduction is at least 1 pH. In one embodiment the reduction is such that the pH-treated tobacco material has a pH of less than about 7, i.e. an acidic pH. Using routine techniques, the skilled person would be able to determine such an amount of the one or more gases being used.
[0079] The term "reversible" has its normal meaning in the art, i.e. the reduction in pH caused by the one or more gases can be readily reversed so as to at least partially restore the pH-treated tobacco material to its initial pH. For example, if the one or more gases reduce the pH of the pH-treated tobacco material from about 9.5 to about 5.5, this reduction can be reversed to return the pH of the pH-treated tobacco material to about 7.5 or about 8.5. In one embodiment the reversal is at least about 50%, i.e. the reduction in pH is restored by at least about 50%. In another embodiment the reversal is at least about 60% or at least about 75% or at least about 80%. In one embodiment the reversal is at least about 90% or at least about 95%. In one embodiment the reversal is about 100%.
[0080] The reversible nature of the pH reduction in the present disclosure is particularly advantageous because it means that the nicotine is only converted to its volatile, free base form when the pH-treated tobacco material is exposed to normal atmospheric conditions (e.g. air). It also reduces the loss of available nicotine because the processing method does not have a permanent impact or effect on the nicotine in the pH-treated material. The present disclosure can also be readily applied to bulk storage and individual (e.g. blister, capsule etc.) storage of the pH-treated tobacco material.
[0081] In one embodiment, the one or more gases comprise carbon dioxide. In an alternative embodiment, the one or more gases consist essentially of carbon dioxide. In a further alternative embodiment, the one or more gases consist of carbon dioxide; i.e. the gas is carbon dioxide.
[0082] In one embodiment, the one or more gases comprise at least 20% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 25% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 30% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 35% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 40% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 45% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 50% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 55% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 60% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 65% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 70% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 75% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 80% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 85% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 90% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 95% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 97% by volume carbon dioxide. In one embodiment, the one or more gases comprise at least 99% by volume carbon dioxide.
[0083] In one embodiment, the one or more gases comprise from about 20% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 25% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 30% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 35% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 40% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 45% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 50% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 55% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 60 to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 65% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 70% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 75% to about 100% by volume carbon dioxide. In one embodiment, the one or more gases comprise from about 80% to about 100% by volume carbon dioxide.
[0084] In one embodiment, the pH-treated tobacco material has a pH of greater than 7 and the one or more gases comprise from about 25% to about 100% by volume carbon dioxide.
[0085] In another embodiment, the pH-treated tobacco material has a pH of greater than 7 and the one or more gases consist essentially of carbon dioxide.
[0086] In another embodiment, the pH-treated tobacco material has a pH of greater than 7 and the one or more gases comprise from about 50% to about 100% by volume carbon dioxide.
[0087] In the same manner as the level of increase caused by the pH treatment, the reduction in pH by the one or more gases is not specifically limited. It will depend on the pH of the pH-treated tobacco material and on the one or more gases being used, amongst other factors. Given the effect of the reduction in pH--namely the protonation of nicotine--the pH of the pH-treated tobacco material is, in one embodiment, reduced to an acidic pH, i.e. a pH of less than 7.
[0088] In one embodiment the reduction in pH by the one or more gases is about 0.1 pH. In another embodiment the reduction in pH is about 0.2 pH. In another embodiment the reduction in pH is about 0.3 pH. In another embodiment the reduction in pH is about 0.4 pH. In another embodiment the reduction in pH is about 0.5 pH. In another embodiment the reduction in pH is about 1.0 pH. In another embodiment the reduction in pH is about 1.5 pH. In another embodiment the reduction in pH is about 2.0 pH. In another embodiment the reduction in pH is about 2.5 pH. In another embodiment the reduction in pH is about 3.0 pH.
[0089] In one embodiment the one or more gases reduce the pH of the pH-treated tobacco material to less than 7. In one embodiment the one or more gases reduce the pH of the pH-treated tobacco material to less than about 6.5. In one embodiment the one or more gases reduce the pH of the pH-treated tobacco material to less than about 6. In one embodiment the one or more gases reduce the pH of the pH-treated tobacco material to less than about 5. In one embodiment the one or more gases reduce the pH of the pH-treated tobacco material to less than about 4. In one embodiment the pH of the pH-treated tobacco material is reduced by the one or more gases to about 5.5.
[0090] The present disclosure also provides a tobacco material obtained by the method described herein. The tobacco material obtained by the processing method differs from those of the art at least by the interaction of the one or more gases (e.g. carbon dioxide) with the material and the reversible modification of the pH.
[0091] Without wishing to be bound by theory, the inventors believe that there is an interaction between the one or more gases and water present in the pH-treated tobacco material and/or atmosphere which results in the formation of a weak acid, lowering the pH and protonating nicotine. This pH modification is, however, reversible in that on exposure of the tobacco to normal atmospheric conditions, it returns towards its initial value. The tobacco material obtained by the processing method is therefore unique at least due to its "dynamic" pH. At the molecular level the tobacco material will also differ from known tobacco because of the interaction of the one or more gas(es) with the tobacco surface.
Storage Method
[0092] Along with a method for processing a pH-treated tobacco material, the present disclosure provides a method of storing pH-treated tobacco material. The method has at least three possible configurations and the key steps of these configurations are set out as flow-charts in FIGS. 2, 3 and 4.
[0093] The method may comprise (i) placing the pH-treated tobacco material in a storage container, (ii) sealing the storage container, and (iii) processing the pH-treated tobacco material according to the method described herein. This configuration is shown in FIG. 2.
[0094] Alternatively the method may comprise (i) placing the pH-treated tobacco material in a storage container, (ii) processing the pH-treated tobacco material according to the method described herein, and (iii) sealing the storage container. This configuration is shown in FIG. 3.
[0095] In a further alternative, the method may comprise (i) processing the pH-treated tobacco material according to the method described herein, (ii) placing the tobacco material obtained from processing step (i) in a storage container and (iii) sealing the storage container. This configuration is shown in FIG. 4.
[0096] FIG. 1 is a schematic cross-sectional view of an example storage container. It includes a storage container 1 comprising pH-treated tobacco material 2 and one or more gases 3 which consist essentially of carbon dioxide. As would be understood by the skilled person, the one or more gases may be as defined above. The example storage container of FIG. 1 may be prepared by placing pH-treated tobacco material in the storage container, sealing the storage and then contacting the pH-treated tobacco material with an effective amount of one or more gases, wherein the one or more gases reduce the pH of the pH-treated tobacco material, and wherein the reduction in pH of the pH-treated tobacco material is reversible, i.e. processing the pH-treated tobacco material according to the method described herein (see FIG. 2). Alternatively the example storage container of FIG. 1 may be prepared by placing pH-treated tobacco material in the storage container, contacting the pH-treated tobacco material with an effective amount of one or more gases, wherein the one or more gases reduce the pH of the pH-treated tobacco material, and wherein the reduction in pH of the pH-treated tobacco material is reversible (i.e. processing the pH-treated tobacco material according to the method described herein), and then sealing the storage container (FIG. 3). As a further alternative, the example storage container of FIG. 1 may be prepared by contacting the pH-treated tobacco material with an effective amount of one or more gases, wherein the one or more gases reduce the pH of the pH-treated tobacco material, and wherein the reduction in pH of the pH-treated tobacco material is reversible (i.e. processing the pH-treated tobacco material according to the method described herein), placing the pH-treated tobacco material in the storage container and sealing the storage container (FIG. 4).
[0097] The present disclosure further provides a storage container obtained by said storage method. An example storage container is shown in FIG. 1; although the skilled person will appreciate that the present disclosure is not limited to the specific arrangement shown in FIG. 1. This figure is just provided by way of example.
[0098] The storage container can be an apparatus or a container used for bulk storage, such as a sealed bag, box or the like. The storage container can also be an apparatus or container used for storage of pH-treated tobacco material on a smaller scale, this type of storage is referred to herein as "individual storage" because it is intended to refer to storage of consumer portions or individual portions of tobacco rather than larger quantities stored during shipment etc. In one embodiment the individual storage is a blister pack where each blister contains a single dosage or portion of the tobacco material. In another embodiment the individual storage is a tobacco pod.
[0099] In one embodiment the storage container is a sealed container. The sealing of the container is not limited and can be achieved by any method known in the art which results in an air-tight seal. In one embodiment the storage container is a hermetically sealed container. In another embodiment the store is a sealed blister in a blister pack or a sealed tobacco pod. Again the sealing can be achieved by any method known in the art which results in an air-tight seal.
[0100] Advantageously, the storage container obtained by the above method can be used in the bulk shipment and storage of pH treated tobacco material to increase bulk storage time, or in blister packs, tobacco pods or the like to increase shelf-life of pH-treated tobacco material. The terms "bulk storage time" and "shelf-life" are discussed further below.
Shelf-life and Bulk Storage Time
[0101] The present disclosure further provides the use of carbon dioxide to increase the shelf-life or bulk storage time of pH-treated tobacco material. By the term "bulk storage time" is meant the time when the pH-treated tobacco material is subject to bulk storage or stored in bulk, e.g. during shipment. By the term "shelf-life" is meant the time when the pH-treated tobacco material is available to a user, e.g. the time when the product is available for purchase and use. Advantageously the present disclosure provides an improvement in both scenarios.
[0102] Consequently, the present disclosure also provides an improvement in supply chain time, i.e. the time from sealing the blister, capsule, pod or the like at source to the time when the product is available for purchase.
[0103] An increase or improvement in shelf-life or bulk storage time can be shown by measuring the blend nicotine and/or aerosol nicotine over time. Measurement of aerosol nicotine is by methods known in the art such as the CORESTA Recommended Method Nos. 81 (June 2015) and 84 (June 2017). Measurement of blend nicotine is also by methods known in the art such as the CORESTA Recommended Method No. 82 (February 2005).
[0104] CORESTA Recommended Method No. 81 (June 2015) is entitled "Routine Analytical Machine for E-Cigarette Aerosol Generation and Collection--Definitions and Standard Conditions", and includes the requirements needed for generation and collection of e-cigarette aerosol for analytical testing purposes. CORESTA Recommended Method No. 84 (June 2017) is entitled "Determination of Glycerin, Propylene Glycol, Water, and Nicotine in the Aerosol of E-Cigarettes by Gas Chromatographic Analysis" and refers to the CORESTA Recommended Method No. 81 (June 2015). CORESTA Recommended Method No. 84 details how aerosol is generated and collected from electronic cigarettes by a standard procedure. The collected matter is then dissolved in a solvent and the glycerin, propylene glycol, water and nicotine content of this solution are determined by gas chromatography. Aerosol collected mass may be determined gravimetrically, and results are expressed as the weight of analyte collected per device, per weight of collected aerosol, per puff, or per puff set as warranted. The equipment and reagents for the analysis, including the gas chromatography analysis, are detailed in the CORESTA Method.
[0105] CORESTA Recommended Method No. 62 is entitled "Determination of nicotine in tobacco and tobacco products gas chromatographic analysis", and describes how the nicotine content of a sample of tobacco or a tobacco product (referred to herein as "blend nicotine") is determined by liquid/liquid extraction into an organic extraction solvent containing an internal standard, followed by gas chromatographic analysis with flame ionization detection. Either MTBE or n-hexane may be used as the organic extraction solvent since no significant difference is seen between the results obtained by the two different methods. The equipment and reagents for the analysis are detailed in the CORESTA Method.
[0106] The blend and/or aerosol nicotine is measured in the present disclosure according to the above-mentioned CORESTA methods, namely CORESTA Recommended Method Nos. 81, 84 and 62. These CORESTA methods are therefore incorporated herein by reference.
[0107] In one embodiment an increase in shelf-life or bulk storage time can be determined by measuring the blend nicotine or aerosol nicotine of a pH-treated tobacco material stored under carbon dioxide, and comparing it with the blend or aerosol nicotine of a pH-treated tobacco material not stored under carbon dioxide, e.g. stored in air. To determine whether the storage time or shelf-life is improved by the use of carbon dioxide according to the present disclosure, all variables should be kept constant except for the storage environment. For example, two samples of a pH-treated tobacco material could be stored under ambient temperature and pressure for 3 months, one could be stored under air and one could be stored under carbon dioxide. The blend nicotine and/or aerosol nicotine could then be measured according to the above-described CORESTA methods for both samples and their comparison would demonstrate whether the shelf-life and storage time had been improved (i.e. increased) by the present disclosure.
[0108] The various embodiments described herein are presented only to assist in understanding and teaching the claimed features. These embodiments are provided as a representative sample of embodiments only, and are not exhaustive and/or exclusive. It is to be understood that advantages, embodiments, examples, functions, features, structures, and/or other aspects described herein are not to be considered limitations on the scope of the disclosure as defined by the claims or limitations on equivalents to the claims, and that other embodiments may be utilized and modifications may be made without departing from the scope of the claimed disclosure. Various embodiments of the present disclosure may suitably comprise, consist of, or consist essentially of, appropriate combinations of the disclosed elements, components, features, parts, steps, means etc. other than those specifically described herein. In addition, this disclosure may include other disclosures not presently claimed, but which may be claimed in future.
* * * * *

D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.