High Entropy Alloy with Low Specific Gravity
Lee; Hoo Dam ; et al.
U.S. patent application number 17/313604 was filed with the patent office on 2022-04-14 for high entropy alloy with low specific gravity. The applicant listed for this patent is Hyundai Motor Company, Kia Corporation. Invention is credited to Kyung Sik Choi, Hoo Dam Lee, Tae Gyu Lee, Byung Ho Min.
| Application Number | 20220112580 17/313604 |
| Document ID | / |
| Family ID | 1000005723237 |
| Filed Date | 2022-04-14 |









View All Diagrams
| United States Patent Application | 20220112580 |
| Kind Code | A1 |
| Lee; Hoo Dam ; et al. | April 14, 2022 |
High Entropy Alloy with Low Specific Gravity
Abstract
A high-entropy alloy with a low specific gravity includes Fe: 16.7 to 25 at %, Cr: 10.5 to 20.6 at %, Al: 12.7 to 18 at %, remaining Ni, and impurities. The high-entropy alloy includes microstructures of a body-centered cubic (BCC) and a face-centered cubic (FCC) formed together, wherein a fraction of the FCC of the high-entropy alloy is 50 to 80%.
| Inventors: | Lee; Hoo Dam; (Seongnam-si, KR) ; Choi; Kyung Sik; (Seoul, KR) ; Lee; Tae Gyu; (Seoul, KR) ; Min; Byung Ho; (Suwon-si, KR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005723237 | ||||||||||
| Appl. No.: | 17/313604 | ||||||||||
| Filed: | May 6, 2021 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C22C 19/056 20130101 |
| International Class: | C22C 19/05 20060101 C22C019/05 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Oct 13, 2020 | KR | 10-2020-0132078 |
Claims
1. A high-entropy alloy with a low specific gravity, the high-entropy alloy comprising Fe: 16.7 to 25 at %, Cr: 10.5 to 20.6 at %, Al: 12.7 to 18 at %, remaining Ni, and impurities.
2. The high-entropy alloy according to claim 1, wherein the high-entropy alloy comprises Cr: 15 to 18 at %.
3. The high-entropy alloy according to claim 1, wherein the high-entropy alloy comprises Al: 16 to 17 at %.
4. The high-entropy alloy according to claim 1, wherein the high-entropy alloy further comprises Ti: 0.8 to 1 at %.
5. The high-entropy alloy according to claim 1, wherein the high-entropy alloy contains the impurities of 0.1 at % or less.
6. The high-entropy alloy according to claim 1, wherein a Ni:Fe content (at %) ratio of the high-entropy alloy is 2:1 to 3:1.
7. The high-entropy alloy according to claim 1, wherein the high-entropy alloy includes microstructures of a body-centered cubic (BCC) and a face-centered cubic (FCC) formed together.
8. The high-entropy alloy according to claim 7, wherein a fraction of the FCC of the high-entropy alloy is 50 to 80%.
9. The high-entropy alloy according to claim 8, wherein a sigma phase is not formed in the high-entropy alloy.
10. The high-entropy alloy according to claim 8, wherein an intermetallic compound is not formed in the high-entropy alloy.
11. The high-entropy alloy according to claim 1, wherein the high-entropy alloy has a specific gravity of 6.92 to 7.04 g/cm.sup.3.
12. The high-entropy alloy according to claim 1, wherein the high-entropy alloy has a yield strength at room temperature of 670 to 700 MPa, and a tensile strength of 880 to 920 MPa.
13. The high-entropy alloy according to claim 1, wherein a Rockwell hardness of the high-entropy alloy at 600.degree. C. is 20 to 43 HRC.
14. The high-entropy alloy according to claim 13, wherein the Rockwell hardness at 600.degree. C. is 38 to 43 HRC.
15. A high-entropy alloy with a low specific gravity comprising Fe, Cr, Al, and Ni, wherein microstructures of a body-centered cubic (BCC) and a face-centered cubic (FCC) are formed together, and a fraction of the FCC is 50 to 80%.
16. The high-entropy alloy according to claim 15, wherein a sigma phase is not formed in the high-entropy alloy.
17. The high-entropy alloy according to claim 15, wherein an intermetallic compound is not formed in the high-entropy alloy.
18. The high-entropy alloy according to claim 15, wherein the high-entropy alloy comprises Fe: 16.7 to 25 at %, Cr: 10.5 to 20.6 at %, Al: 12.7 to 18 at %, remaining Ni, and impurities.
19. The high-entropy alloy according to claim 18, wherein the high-entropy alloy further comprises Ti: 0.8 to 1 at %.
20. The high-entropy alloy according to claim 18, wherein the high-entropy alloy contains the impurities of 0.1 at % or less.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of Korean Patent Application No. 10-2020-0132078, filed on Oct. 13, 2020, which application is hereby incorporated herein by reference.
TECHNICAL FIELD
[0002] The present disclosure relates to a high-entropy alloy with a low specific gravity.
BACKGROUND
[0003] In general, a turbine being applied to an automotive turbocharger has been applied to diesel engines of all commercial vehicles, such as an SUV, bus, and truck, and agricultural machinery, and recently, it has also been applied to a gasoline engine to improve vehicle fuel economy.
[0004] Since the turbocharger technology has an effect of reducing an exhaust gas while functioning to exert a great power through an increase of a car torque, the mounting rate thereof has been increased at high speed.
[0005] The turbocharger is a kind of energy recycling device, and is a technology to rotate a turbine using a flow energy being thrown away as an engine exhaust gas and to supercharge an intake air through rotation of a compressor connected to the turbine on the same shaft.
[0006] Although the turbocharger enables a small engine to produce a high output through supercharging of the intake air, it has been pointed out that the turbocharger has the great disadvantage of the transient driving response performance degradation called turbo-lag.
[0007] Recently, in order to solve the problem of the turbo-lag as described above, a high-entropy alloy capable of improving several physical properties of the alloy has been used.
[0008] A general high-entropy alloy (HEA) is defined as a multi-element alloy that is obtained by alloying various kinds of constituent elements in similar proportions without primary elements constituting an alloy, such as steel being a general alloy, aluminum alloy, or titanium alloy. The high-entropy alloy as described above is a metal material having a high mixed entropy in the alloy, and having a single phase organization, such as face-centered cubic (FCC) or body-centered cubic (BCC), without forming an intermetallic compound or a middle phase.
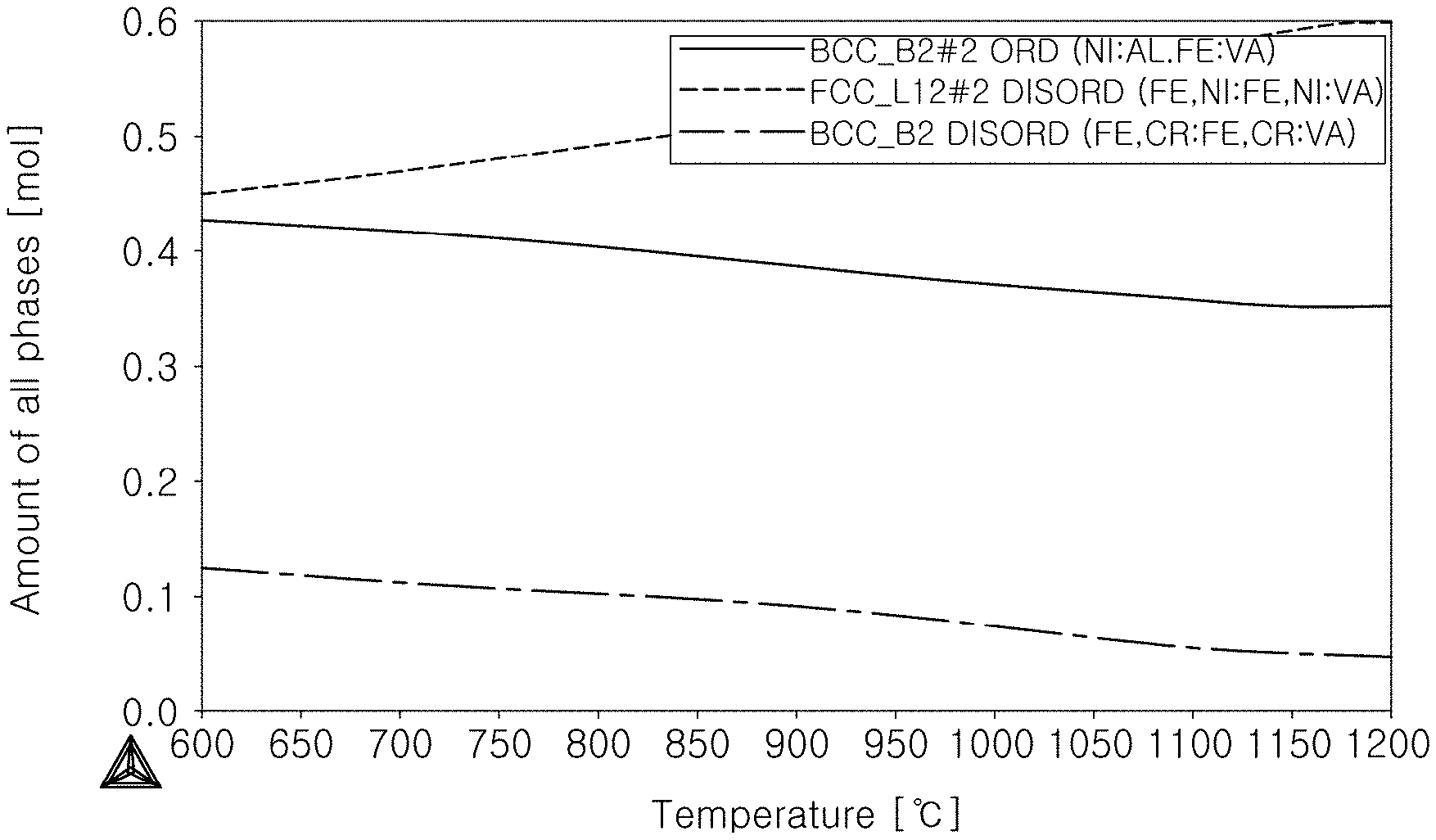
[0009] For example, a CoCrFeMnNi alloy composed of the face-centered cubic (FCC) has a high fracture toughness, and an AlCrFeNi alloy composed of the body-centered cubic (BCC) has a high strength.
[0010] The foregoing is intended merely to aid in the understanding of the background of the present disclosure, and is not intended to mean that the present disclosure falls within the purview of the related art that is already known to those of ordinary skill in the art.
SUMMARY
[0011] The present disclosure relates to a high-entropy alloy with a low specific gravity. Particular embodiments relate to a high-entropy alloy with a low specific gravity, which has an excellent physical property at a high temperature and a low specific gravity by controlling a fraction for generating a microstructure of a face-centered cubic (FCC).
[0012] Embodiments of the present disclosure solve problems, and provide a high-entropy alloy with a low specific gravity, which has an excellent physical property at a high temperature and a low specific gravity by controlling a ratio of a face-centered cubic (FCC) to a body-centered cubic (BCC).
[0013] According to an embodiment of the present disclosure, a high-entropy alloy with a low specific gravity includes Fe: 16.7 to 25 at %, Cr: 10.5 to 20.6 at %, Al: 12.7 to 18 at %, remaining Ni, and inevitable impurities.
[0014] It is preferable that the high-entropy alloy includes Cr: 15 to 18 at %.
[0015] It is preferable that the high-entropy alloy includes Al: 16 to 17 at %.
[0016] The high-entropy alloy further includes Ti: 0.8 to 1 at %.
[0017] It is preferable that the high-entropy alloy contains impurities of 0.1 at % or less.
[0018] It is preferable that a Ni:Fe content (at %) ratio of the high-entropy alloy is 2:1 to 3:1.
[0019] The high-entropy alloy is characterized in that microstructures of a body-centered cubic (BCC) and a face-centered cubic (FCC) are formed together.
[0020] The high-entropy alloy is characterized in that a fraction of the face-centered cubic (FCC) is 50 to 80%.
[0021] The high-entropy alloy is characterized in that a sigma phase is not formed.
[0022] The high-entropy alloy is characterized in that an intermetallic compound is not formed.
[0023] The high-entropy alloy is characterized in that a specific gravity is 6.92 to 7.04 g/cm.sup.3.
[0024] The high-entropy alloy is characterized in that a yield strength at a room temperature is 670 to 700 MPa, and a tensile strength is 880 to 920 MPa.
[0025] The high-entropy alloy is characterized in that a Rockwell hardness at 600.degree. C. is 20 to 43 HRC.
[0026] The high-entropy alloy is characterized in that the Rockwell hardness at 600.degree. C. is 38 to 43 HRC.
[0027] Meanwhile, according to an embodiment of the present disclosure, a high-entropy alloy with a low specific gravity is a high-entropy alloy including Fe, Cr, Al, and Ni, wherein microstructures of a body-centered cubic (BCC) and a face-centered cubic (FCC) are formed together, and a fraction of the face-centered cubic (FCC) is 50 to 80%.
[0028] The high-entropy alloy is characterized in that a sigma phase is not formed.
[0029] The high-entropy alloy is characterized in that an intermetallic compound is not formed.
[0030] The high-entropy alloy includes Fe: 16.7 to 25 at %, Cr: 10.5 to 20.6 at %, Al: 12.7 to 18 at %, remaining Ni, and inevitable impurities.
[0031] The high-entropy alloy further includes Ti: 0.8 to 1 at %.
[0032] It is preferable that the high-entropy alloy contains impurities of 0.1 at % or less.
[0033] According to an embodiment of the present disclosure, it is possible to implement the high-entropy alloy which can similarly maintain the high-temperature characteristics with the low specific gravity against Inconel 713 being a commercial good used for high-temperature parts by adjusting the fraction of the face-centered cubic (FCC) while forming the microstructures of the body-centered cubic (BCC) and the face-centered cubic (FCC) together through adjustment of the content of each element in the high-entropy alloy including Fe, Cr, Al, and Ni.
[0034] In particular, since the high-entropy alloy having excellent characteristics at the high temperature is implemented without using expensive alloy elements, effects of saving the manufacturing cost in case of manufacturing the parts for improving the vehicle fuel economy can be expected, and effects of applying the alloy to various parts can be expected since the existing precise alcoholic method is possible.
BRIEF DESCRIPTION OF THE DRAWINGS
[0035] The above and other objects, features and advantages of embodiments of the present disclosure will be more clearly understood from the following detailed description taken in conjunction with the accompanying drawings, in which:
[0036] FIG. 1A is a state diagram for comparative example 2;
[0037] FIG. 1B is a state diagram for embodiment 1;
[0038] FIG. 1C is a state diagram for embodiment 2;
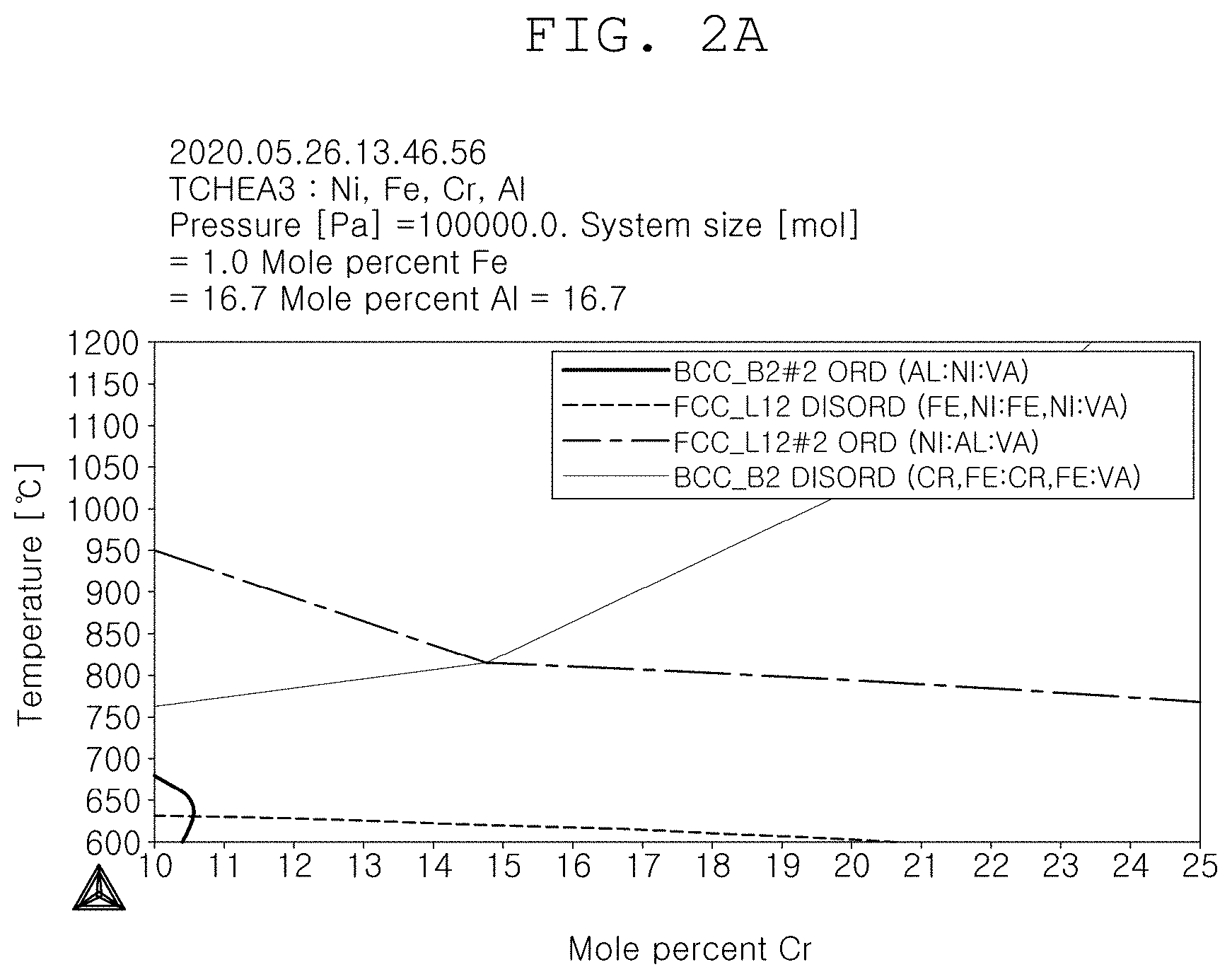
[0039] FIG. 2A is a state diagram according to the content of Cr in [Ni.sub.33.3Fe.sub.33.3Al.sub.16.7];
[0040] FIG. 2B is a state diagram according to the content of Al in [Ni.sub.33.3Fe.sub.33.3Cr.sub.16.7];
[0041] FIG. 3A is a state diagram for embodiment 3;
[0042] FIG. 3B is a state diagram for embodiment 4;
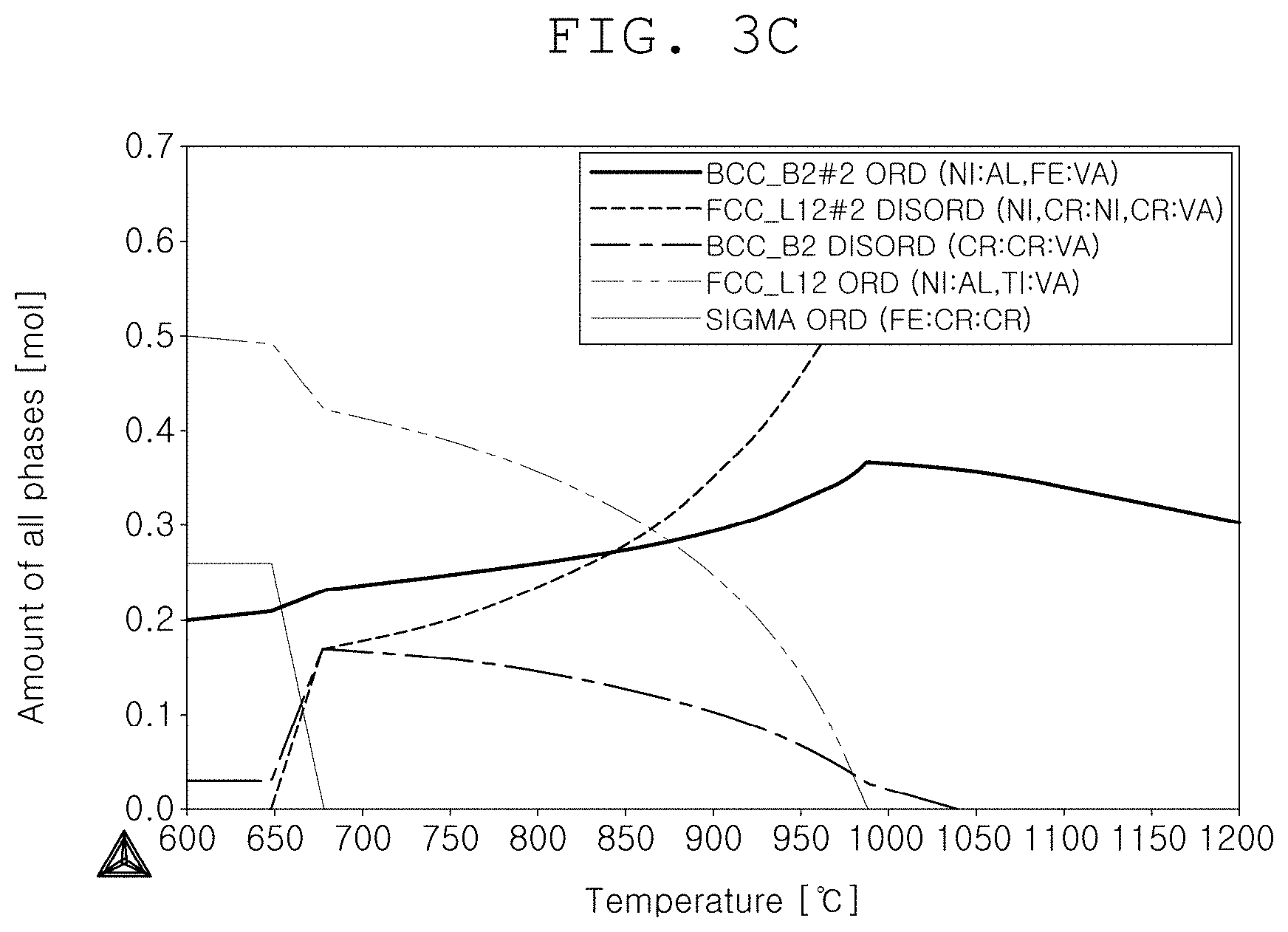
[0043] FIG. 3C is a state diagram for [Ni.sub.38.6Fe.sub.25Cr.sub.16.7Al.sub.16.7Ti.sub.3];
[0044] FIG. 4A is a state diagram according to the addition of Mn in [Ni.sub.49.1Fe.sub.16.7Cr.sub.16.7Al.sub.16.7Ti.sub.1] alloy;
[0045] FIG. 4B is a state diagram according to the addition of Co in [Ni.sub.49.1Fe.sub.16.7Cr.sub.16.7Al.sub.16.7Ti.sub.1] alloy;
[0046] FIG. 5A is a microphotograph showing the surface of a conventional material after heat treatment; and
[0047] FIG. 5B is a microphotograph showing the surface of embodiment 4 after heat treatment.
DETAILED DESCRIPTION OF ILLUSTRATIVE EMBODIMENTS
[0048] Hereinafter, embodiments of the present disclosure will be described in more detail with reference to the accompanying drawings. However, the present disclosure is not limited to embodiments as disclosed hereinafter, but may be embodied in various different forms. However, the embodiments as described hereinafter are only for specific details provided to complete the present disclosure and to assist those of ordinary skill in the art to which the present disclosure pertains in a comprehensive understanding of the disclosure.
[0049] According to a high-entropy alloy according to an embodiment of the present disclosure, it is possible to excellently maintain the physical properties, such as hardness and strength, at room temperature and at a high temperature since a ratio of a face-centered cubic (FCC) is controlled through adjustment of the content of alloy elements in Fe--Cr--Al--Ni alloy series in which microstructures of a body-centered cubic (BCC) and the face-centered cubic (FCC) are formed together.
[0050] Specifically, a high-entropy alloy according to an embodiment of the present disclosure includes Fe: 16.7 to 25 at %, Cr: 10.5 to 20.6 at %, Al: 12.7 to 18 at %, remaining Ni, and inevitable impurities. Further, the high-entropy alloy further may further include Ti: 0.8 to 1 at %, and it is preferable to adjust the impurities to 0.1 at % or less.
[0051] In the following description, unless specially mentioned, % that is described as a unit of a composition range means at %.
[0052] Fe, Cr, Al, and Ni are elements constituting a high-entropy alloy, and through the content adjustment of alloy elements, the microstructures of the body-centered cubic (BCC) and the face-centered cubic (FCC) are formed together, and the ratio of the face-centered cubic (FCC) is adjusted at the same time.
[0053] For example, it is preferable to control the ratio of the face-centered cubic (FCC) to be at the level of 50 to 80% through the content adjustment of the alloy elements.
[0054] Hereinafter, the reason to limit the ratio of a content of each alloy element and a relative content will be described.
[0055] In the following measurement, a specimen was manufactured while changing the content of respective ingredients as shown in Table 1 below. For example, a high-entropy mother alloy molten metal was prepared by dissolving and alloying the respective ingredients in an Ar atmosphere using an are melter after quantification while changing the content of the respective ingredients as shown in Table 1. Then, the specimen was manufactured by injecting the high-entropy mother alloy molten metal into a mold.
TABLE-US-00001 TABLE 1 Classification Ni Fe Cr Al Ti conventional material Inconel 713 (commercial material) comparative example 1 25 25 25 25 -- comparative example 2 33.3 33.3 16.7 16.7 -- embodiment 1 41.6 25 16.7 16.7 -- embodiment 2 50 16.7 16.7 16.7 -- embodiment 3 40.6 25 16.7 16.7 0.8 embodiment 4 49.9 16.7 16.7 16.7 1
[0056] Meanwhile, since the high-entropy alloy having a BCC structure showed a higher strength than the strength of the high-entropy alloy having an FCC structure as the room temperature characteristics, the microstructures were observed with respect to specimens according to the conventional material, comparative example 1, and comparative example 2 of Table 1, and the Rockwell hardness (HRC) for each temperature was measured. The results of the measurement are shown in Table 2 below.
TABLE-US-00002 TABLE 2 Rockwell hardness(HRC) Classification microstructure 25.degree. C. 400.degree. C. 600.degree. C. 700.degree. C. 800.degree. C. commercial material FCC + precipitation 44 45 43 42 39 phase comparative example 1 BCC 40 37 28 10 1 or less comparative example 2 BCC + FCC 33.6 27 22 15 3.8
[0057] As can be known from Table 2, in case of comparative example 1 based on BCC being known to have excellent hardness at room temperature (25.degree. C.), it was able to be confirmed that the hardness similar to the hardness at room temperature was maintained until 600.degree. C., but the physical property was abruptly decreased at a temperature exceeding 600.degree. C., and at a high temperature (700.degree. C. and 800.degree. C.), hardness characteristics lower than those of Inconel 713 material that is the conventional material were shown.
[0058] In contrast, in case of comparative example 2 in which the BCC and the FCC were formed together, low hardness was shown as a whole in the whole temperature zone due to the forming of the FCC, but it was able to be confirmed that high hardness characteristics were shown due to the containing of the FCC as the temperature went higher.
[0059] Accordingly, it was able to be confirmed that the physical property at the high temperature could be improved by controlling the FCC generation fraction.
[0060] Next, the reason to determine the ratio of another alloy element to Ni for entropy control will be described.
[0061] First, the content ratio of Fe to Ni that is a base alloy element will be described.
[0062] In order to find out the fraction relationship between the hardness at a high temperature and FCC (ordered FCC (.gamma.')+disordered FCC (.gamma.)), the microstructure fraction, hardness, and ductility of specimens according to comparative examples 1 and 2 and embodiments 1 and 2 were measured, and the results of the measurement are shown in Table 3 below.
[0063] Further, state diagrams of comparative example 2, and embodiments 1 and 2 are shown in FIGS. 1A to 1C, respectively. FIG. 1A is a state diagram for comparative example 2, FIG. 1B is a state diagram for embodiment 1, and FIG. 1C is a state diagram for embodiment 2.
TABLE-US-00003 TABLE 3 ORD constitutive fraction (600.degree. C. reference fraction) FCC fraction elongation Ordered Disordered Ordered Disordered fraction in FCC HRC (at (at room Classification BCC BCC FCC(.gamma.') FCC(.gamma.) (%) (%) 800.degree. C.) temp) comparative example 1 28 72 -- -- -- -- 1 about 2% comparative example 2 45 13 -- 42 42% 0% 3.8 15% or more embodiment 1 31 15 22 32 54% 40% 15.2 about 2.5% embodiment 2 10 18 72 0 72% 100% 30 about 1.5%
[0064] As can be known from Table 3 and FIG. 1A, in case of comparative example 2 corresponding to an AlCrFe.sub.2Ni.sub.2 alloy, it was able to be confirmed that the Fe:Ni content ratio (at %) was 2:2. As the microstructure, an ordered BCC, disordered BCC, and disordered FCC were mixed and created.
[0065] Meanwhile, as can be known from Table 3 and FIGS. 1B and 1C, it was able to be confirmed that ordered FCC (.gamma.')+disordered FCC(.gamma.), being a new phase, could be formed through the Fe content control.
[0066] In particular, if the Fe:Ni content ratio (at %) is 2:2 as in comparative example 2, only the disordered FCC(.gamma.) was created, and the hardness improvement effect at a high temperature was incomplete.
[0067] Further, if Fe was replaced by Ni and the Fe:Ni content ratio (at %) was 1:2 as in embodiment 1, it was able to be confirmed that ordered FCC(.gamma.') was created together with the ordered BCC, disordered BCC, and disordered FCC, and the hardness at the high temperature was improved.
[0068] Further, if the Fe:Ni content ratio (at %) is 1:3 as in embodiment 2, the disordered FCC(.gamma.) fraction capable of securing the ductility was reduced as the temperature went lower, and only ordered FCC(.gamma.') remained to cause an abrupt brittleness to occur.
[0069] Meanwhile, if the disordered FCC fraction in the FCC (ordered FCC+disordered FCC) in the microstructural crystalline structure disappears, the brittleness is increased. Accordingly, it is preferable that the FCC total fraction is 50 to 80% to suppress the occurrence of the abrupt brittleness in the high-entropy alloy. More preferably, the maximum value of the FCC total fraction does not exceed 72%.
[0070] For this, it was able to be confirmed that it was preferable to maintain the Fe content equal to or higher than 16.7 at % and equal to or lower than 25 at %.
[0071] Next, the reason to limit the content of Cr and Al will be described.
[0072] FIG. 2A is a state diagram according to the content of Cr in [Ni.sub.33.3Fe.sub.33.3Al.sub.16.7], and FIG. 2B is a state diagram according to the content of Al in [Ni.sub.33.3Fe.sub.33.3Cr.sub.16.7].
[0073] As can be known from FIG. 2A, if the content of Cr becomes less than 16.7 at %, the FCC content is increased, and in addition, the ordered FCC (.gamma.') is increased. Further, the specific gravity is increased. In particular, if the Cr content is less than 10.5 at %, the ordered BCC is newly created in the neighborhood of 650.degree. C., and thus the lowest value of the content is limited to 10.5 at %.
[0074] Further, if the Cr content is increased, the FCC content is reduced, and if the FCC content exceeds 20.6 at %, the disordered FCC disappears, and thus the phase control is unable to be performed.
[0075] Accordingly, by limiting the Cr content to 10.5 to 20.6 at %, it is possible to control the FCC/BCC phase fraction, and preferably, by controlling the same to be 15 to 18 at %, a stable physical property can be secured within the range in which the characteristic change is not big.
[0076] As can be known from FIG. 2B, if the content of Al becomes lower than 16.7 at %, the FCC content is increased, and the specific gravity is increased. In accordance with the increase of the specific gravity, the effect is halved when being applied to the parts. In particular, if the content of Al is lower than 12.7 at %, the BCC phase disappears to make the phase control impossible.
[0077] Further, if the content of Al is higher than 18 at %, the BCC phase disappears, and thus it is preferable to limit the content of Al to 12.7 to 18 at %, and more preferably, in case of the control to 16 to 17 at %, a stable physical property can be secured within the range in which the characteristic change is not big.
[0078] Meanwhile, in order to maintain the characteristics of the high-entropy alloy, it is preferable to maintain the content of Cr and Al to Cr: 10.5 to 20.6 at % and Al: 12.7 to 18 at %. Preferably, it is good to maintain the content of Cr and Al to Cr: 15 to 18 at % and Al: 16 to 17 at %. Most preferably, it is good to maintain the content of Cr and Al to 16.7 at %, respectively.
[0079] If the content of Cr and Al deviates from a suggested range, the set ratio of ordered FCC (.gamma.')+disordered FCC(.gamma.) is changed, and thus the phase control becomes impossible.
[0080] In order to find out the reason to limit the content of Cr and Al as described above, content-adjusted specimens were prepared as shown in Table 4, and the microstructural fraction and hardness of the prepared specimens were measured. The results of the measurement are shown in Table 5.
TABLE-US-00004 TABLE 4 Classification Ni Fe Cr Al embodiment 1 41.6 25 16.7 16.7 embodiment 1-1 43.3 25 15 16.7 embodiment 1-2 40.3 25 18 16.7 embodiment 1-3 42.3 25 16.7 16 embodiment 1-4 41.3 25 16.7 17 embodiment 2 50 16.7 16.7 16.7 embodiment 2-1 51.6 16.7 15 16.7 embodiment 2-2 48.6 16.7 18 16.7 embodiment 2-3 50.6 16.7 16.7 16 embodiment 2-4 49.6 16.7 16.7 17
TABLE-US-00005 TABLE 5 FCC fraction (%) (ORD FCC/ constitutive fraction (600.degree. C. reference fraction) ORD FCC + FCC Ordered Disordered Ordered Disordered DISORD FCC fraction HRC (at HRC at Classification BCC BCC FCC(.gamma.') FCC(.gamma.) fraction) change room temp) 800.degree. C. embodiment 1 31 15 22 32 55 (59) -- 25.4 15.2 embodiment 1-1 28 12 31 29 60 (48) 9.1% 24 16 embodiment 1-2 32 15 24 29 53 (54) -3.6% 26 14 embodiment 1-3 27 14 29 30 59 (50) 7.3% 24.5 15.5 embodiment 1-4 34 16 19 31 50 (62) -9.1% 27 13 embodiment 2 10 18 72 0 72 (100) -- 30.5 30.1 embodiment 2-1 15 7 76 0 76 (100) 5.6% 29 31 embodiment 2-2 19 12 68 0 68 (100) -5.6% 31 29 embodiment 2-3 6 18 76 0 76 (100) 5.6% 29.5 31 embodiment 2-4 12 18 70 0 70 (100) -2.8% 30.5 30
[0081] As can be known from Table 4 and Table 5, in order to maintain the change of the set FCC fraction within 10%, as suggested, it is preferable to maintain Cr: 15 to 18 at % and Al: 16 to 17 at %, and in this case, it was able to be confirmed that the hardness change at room temperature and at a high temperature was incomplete.
[0082] Further, as can be confirmed from Table 5, it was able to be confirmed that the FCC fraction could be maintained 50 to 80%, and preferably, 50 to 76%, in case of maintaining Cr: 15 to 18 at % and Al: 16 to 17 at %.
[0083] Next, the influence on alloy elements additionally contained in the high-entropy alloy according to embodiments of the present disclosure, which is a quaternary high-entropy alloy, will be described.
[0084] FIG. 3A is a state diagram for embodiment 3, FIG. 3B is a state diagram for embodiment 4, and FIG. 3C is a state diagram for [Ni.sub.38.6Fe.sub.25Cr.sub.16.7Al.sub.16.7Ti.sub.3].
[0085] As can be known from FIGS. 3A to 3C, it was able to be confirmed that the ordered FCC(.gamma.') precipitation temperature was changed in accordance with the Ti addition, and in case of improving the ordered FCC(.gamma.') precipitation temperature, it can be analogized that the hardness will be improved at a higher temperature.
[0086] However, as can be known from FIG. 3C, if Ti was added over a predetermined range, it was able to be confirmed that a sigma phase was created and thus the precipitation temperature of the ordered FCC(.gamma.') was abruptly increased.
[0087] However, the sigma phase corresponds to needle-shaped particles, and since the creation thereof may cause the alloy brittleness, it is required to control the Ti content.
[0088] Accordingly, it is preferable that the content of Ti is added with 0.8 to 1 at %, and in accordance with the addition of Ti, the precipitation temperature of the ordered FCC(.gamma.') can be increased to about 150 to 200.degree. C.
[0089] However, if the content of Ti exceeds the suggested range, there occurs a problem in that the needle-shaped signal phase is formed and thus the brittleness is abruptly increased.
[0090] Next, the influence exerted on the microstructure by impurities contained in the high-entropy alloy will be described.
[0091] FIG. 4A is a state diagram according to addition of Mn in [Ni.sub.49.1Fe.sub.16.7Cr.sub.16.7Al.sub.16.7Ti.sub.1] alloy, and FIG. 4B is a state diagram according to addition of Co in [Ni.sub.49.1Fe.sub.16.7Cr.sub.16.7Al.sub.16.7Ti.sub.1] alloy.
[0092] It is preferable that the content of the remaining impurities except the primary alloy ingredients of the high-entropy alloy according to an embodiment of the present disclosure is limited to 0.1 at % or less.
[0093] As can be known from FIGS. 4A and 4B, the high-entropy alloy is configured as a single phase by an interaction of elements of the same element group (similar atomic radius and weight required) in the periodic table, but if another element is added, there is a high probability that an intermetallic compound rather than the BCC or FCC is created, and since the addition of the same group elements, such as Mn and Co, may not only hurt the phase fraction characteristics but also make a phase having high brittleness, such as the sigma phase, it is preferable to limit the content to 0.1 at % or less.
[0094] Next, the hardness characteristics according to the temperature of the high-entropy alloy according to embodiments of the present disclosure and the physical properties of the yield strength and the tensile strength at room temperature were evaluated, and the specific gravity was measured. The results of the measurement are shown in Table 6 and Table 7.
TABLE-US-00006 TABLE 6 Rockwell hardness(HRC) Classification 25.degree. C. 400.degree. C. 600.degree. C. 700.degree. C. 800.degree. C. embodiment 1 25.4 26 20 17 15.2 embodiment 2 30.5 33 32 31 30.1 embodiment 3 36 37 38 38 35.1 embodiment 4 40 42 43 43 39.7 commercial 44 45 43 42 39 material
TABLE-US-00007 TABLE 7 specific gravity yield tensile specific ratio against strength strength gravity conventional Classification (MPa) (MPa) (g/cm.sup.3) material embodiment 1 670 900 6.96 12.1% embodiment 2 685 880 7.04 11.1% embodiment 3 680 920 6.92 12.6% embodiment 4 700 890 6.99 11.7% commercial 650 900 7.91 -- material
[0095] As can be known from Table 6 and Table 7, according to embodiments of the present disclosure, it was able to be confirmed that the high hardness could be secured at 600.degree. C. or more, and the yield strength and the tensile strength having a level similar to the conventional material or an improved level at room temperature could be secured.
[0096] Further, it was able to be confirmed that the embodiments had a lower specific gravity than that of Inconel 713 being a commercial good by about 10% or more, and thus the light weight could be achieved.
[0097] Next, in order to find out the oxidation resistance characteristics of the high-entropy alloy according to embodiments of the present disclosure, heat treatment was performed for 100 hr. at 900.degree. C. with respect to specimens of the conventional material and embodiment 4, and thereafter, the depth of an oxide layer formed on the surface of the specimen was measured.
[0098] FIG. 5A is a microphotograph showing the surface of a conventional material after heat treatment, and FIG. 5B is a microphotograph showing the surface of embodiment 4 after heat treatment.
[0099] As can be known from FIGS. 5A and 5B, it was able to be confirmed that an oxide layer was formed with a depth of about 10 .mu.m on the surfaces of the specimens of both the conventional material and embodiment 4.
[0100] Through the above-described results, it was able to be confirmed that the high-temperature oxidation amount according to the embodiment of the present disclosure was similar to that of Inconel 713 being the conventional material.
[0101] Although the present disclosure has been described with reference to the accompanying drawings and the preferred embodiments as described above, the present disclosure is not limited thereto, but also encompasses claims to be described later. Accordingly, those of ordinary skill in the art to which the present disclosure pertains will appreciate that various modifications, additions and substitutions are possible, without departing from the scope and spirit of the disclosure as disclosed in the appended claims.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.