Methods And Compositions For Genetic Modulation Of Tumor Microenvironments
BISSONNETTE; Reid P. ; et al.
U.S. patent application number 17/410459 was filed with the patent office on 2022-04-14 for methods and compositions for genetic modulation of tumor microenvironments. The applicant listed for this patent is HUYABIO International, LLC. Invention is credited to Reid P. BISSONNETTE, Rosemary M. CESARIO, Mireille GILLINGS, Robert GOODENOW, Farbod SHOJAEI.
| Application Number | 20220110924 17/410459 |
| Document ID | / |
| Family ID | |
| Filed Date | 2022-04-14 |












View All Diagrams
| United States Patent Application | 20220110924 |
| Kind Code | A1 |
| BISSONNETTE; Reid P. ; et al. | April 14, 2022 |
METHODS AND COMPOSITIONS FOR GENETIC MODULATION OF TUMOR MICROENVIRONMENTS
Abstract
Provided herein is a therapy comprising an HDAC inhibitor (HDACi), and/or a PD-L1 and/or a PD-1 inhibitor, and/or a CTLA-4 inhibitor. The combination therapy provided herein can be a kit or the composition or a pharmaceutical composition. Also, provided herein is a method of treating cancer using the combination therapy.
| Inventors: | BISSONNETTE; Reid P.; (Carlsbad, CA) ; CESARIO; Rosemary M.; (San Diego, CA) ; GOODENOW; Robert; (San Diego, CA) ; SHOJAEI; Farbod; (San Diego, CA) ; GILLINGS; Mireille; (San Diego, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Appl. No.: | 17/410459 | ||||||||||
| Filed: | August 24, 2021 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 63070173 | Aug 25, 2020 | |||
| International Class: | A61K 31/4406 20060101 A61K031/4406; A61K 39/395 20060101 A61K039/395; C07K 16/28 20060101 C07K016/28; A61P 35/00 20060101 A61P035/00 |
Claims
1. A method of configuring a tumor microenvironment in a patient in need thereof to respond to an immune checkpoint inhibitor therapy, the method comprising: administering to the patient a tumor microenvironment configuring amount of a composition comprising a compound of formula I, or a pharmaceutically acceptable salt thereof: ##STR00009## wherein, A is phenyl or a heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkythio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, C1-C4 alkoxycarbonyl, phenyl, and a heterocyclic group; B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkylthio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, C1-C4 alkoxycarbonyl, and phenyl; Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively; Z is a bond or C1-C4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--; R.sup.1 and R.sup.2 are independently hydrogen or C1-C4 alkyl; R.sup.3 is hydrogen or C1-C4 alkyl; R.sup.4 is hydrogen or --NH.sub.2; one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkylthio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, or C1-C4 alkoxycarbonyl optionally substituted with halogen or C1-C4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen, provided, however, that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group or an alkylamino group.
2. The method of claim 1, wherein the compound of formula I is a class I, class II, or both, selective histone deacetylase inhibitor.
3. The method of claim 1, wherein the compound of formula I comprises: ##STR00010## or a pharmaceutically acceptable salt thereof.
4. The method of claim 1, wherein the administration of the compound of formula I increases an expression of one or more immune checkpoints in the tumor microenvironment.
5. The method of claim 1, wherein the method further comprises administering to the patient an immune checkpoint inhibitor.
6. The method of claim 5, wherein the immune checkpoint inhibitor comprises an inhibitor of VISTA, PD-L1, CTLA-4, PD-L2, B7-1 (CD80), B7-2 (CD86), B7-H3 (CD276), B7-H2, B7-H4 (VTCN1), HVEM (CD270, TNFRSF14), Galectin 9, Galectin3, CEACAM1 (CD66a), OX-2 (CD200), PVR (CD155), PVRL2 (Nectin-2, CD112), FGL-1, PECAM-1, TSG-6, CD47, Stabilin-1 (Clever-1), Neuropilin 1, Neuropilin 2, CD158 (family), IGSF2 (CD101), CD155, GITRL, CD137L, OX40L, LIGHT, CD70, PD-1, RGMB, CTLA-4 (CD152), BTLA, CD160, Tim-3, CD200R, TIGIT, CD112R (PVRIG), LAG-3 (CD223), PECAM-1, CD44, SIRP alpha (CD172a), or a combination thereof.
7. The method of claim 6, wherein the inhibitor comprises a small molecule compound, a nucleic acid, a peptide, a protein, a monoclonal antibody, a human antibody, a mouse antibody, a chimeric antibody, a humanized antibody, or a chimeric humanized antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a fragment or variant thereof.
8. The method of claim 4, wherein the one or more immune checkpoints comprise PD-1, PD-L1, CTLA-4, CD86, CD276/B7-H3, CD244, lymphocyte activation gene-3 (LAG-3), T cell immunoreceptor with Ig and ITIM domains (TIGIT), ecto-5'-nucleotidase (NT5E/CD73), signal regulatory protein .alpha. (SIRP.alpha.), nuclear factor of activated T cells 4 (NFATC4), poliovirus receptor (CD155), or any combination thereof.
9. The method of claim 8, wherein the one or more immune checkpoints comprise CD276/B7-H3, CD244, NT5E/CD73, or any combination thereof.
10. The method of claim 1, wherein the administration of the compound of formula I increases an expression of one or more adaptive immunity genes.
11. The method of claim 10, wherein the adaptive immunity genes comprise 4-1BB/CD137, tumor necrosis factor .alpha. (TNF.alpha.), interleukin 2 receptor alpha (IL2R.alpha.)/CD25, GZMB (granzyme B), IRF4, and chemokine (C-X3-C motif) receptor 1 (CXC3R1), chemokine (CXC motif) receptor 6 (CXCR6), CXCR3, or any combination thereof.
12. The method of claim 10, wherein the expression of the adaptive immunity genes is increased no later than seven days following an initial administration of the compound of formula I to the patient.
13. The method of claim 1, wherein the administration of the compound of formula I increases an expression of one or more natural killer (NK) cell function genes.
14. The method of claim 13, wherein the NK cell function genes comprise GZMB, killer cell lectin like receptor D1 (KLRD1/CD94), killer cell lectin like receptor C2 (NKG2c/KLRC2), natural killer cell granule protein 7 (NKG7), killer cell lectin like receptor K1 (KLRK1), or any combination thereof.
15. The method of claim 1, wherein the administration of the compound of formula I increases an expression of one or more MHC class I genes.
16. The method of claim 15, wherein the MHC class I genes comprise H2-D1, H2-K1, or both.
17. The method of claim 1, wherein the administration of the compound of formula I increases an expression of one or more MHC class II genes.
18. The method of claim 17, wherein the MHC class II genes comprise H2-Aa, H2-Eb1, or both.
19. The method of claim 1, wherein configuring the tumor microenvironment comprises increasing expression of one or more immune checkpoints in the patient and the tumor microenvironment configuring amount of the compound of formula I is an amount of the compound of formula I sufficient to increase expression of one or more immune checkpoints in the patient.
20. The method of claim 19, wherein increasing the expression of one or more immune checkpoints comprises increasing the expression of one or more of: PD-1, PD-L1, CTLA-4, CD86, CD276/B7-H3, CD244, lymphocyte activation gene-3 (LAG-3), T cell immunoreceptor with Ig and ITIM domains (TIGIT), ecto-5'-nucleotidase (NT5E/CD73), signal regulatory protein .alpha. (SIRP.alpha.), nuclear factor of activated T cells 4 (NFATC4), poliovirus receptor (CD155), or any combination thereof.
21. The method of claim 1, wherein configuring the tumor microenvironment comprises increasing an expression of one or more adaptive immunity genes in the tumor microenvironment of the patient and the tumor microenvironment configuring amount of the compound of formula I is an amount of the compound of formula I sufficient to increase expression of one or more adaptive immunity genes in the tumor microenvironment in the patient.
22. The method of claim 21, wherein increasing expression of one or more adaptive immunity genes comprises increasing expression of one or more of: 4-1BB/CD137, tumor necrosis factor .alpha. (TNF.alpha.), interleukin 2 receptor alpha (IL2R.alpha.)/CD25, GZMB (granzyme B), IRF4, and chemokine (C-X3-C motif) receptor 1 (CXC3R1), chemokine (CXC motif) receptor 6 (CXCR6), CXCR3, or any combination thereof.
23. A method of increasing an expression of one or more natural killer (NK) cell function genes in a tumor microenvironment of a patient, the method comprising administering to said patient a natural killer (NK) cell function gene expression increasing amount of a composition comprising a compound of formula I, or a pharmaceutically acceptable salt thereof.
24. The method of claim 23, wherein the NK cell function genes comprise GZMB, killer cell lectin like receptor D1 (KLRD1/CD94), killer cell lectin like receptor C2 (NKG2c/KLRC2), natural killer cell granule protein 7 (NKG7), killer cell lectin like receptor K1 (KLRK1), or any combination thereof.
25. The method of claim 1, wherein the method of configuring the tumor microenvironment comprises increasing an expression of one or more MHC class I genes in the tumor microenvironment of the patient and the tumor microenvironment configuring amount of the compound of formula I is an amount of the compound of formula I sufficient to increase expression of one or more MHC Class I genes in the tumor microenvironment of the patient.
26. The method of claim 25, wherein the MHC class I genes comprise H2-D1, H2-K1, or both.
27. The method of claim 1, wherein the method of configuring the tumor microenvironment comprises increasing an expression of one or more MHC class II genes in the tumor microenvironment of the patient and the tumor microenvironment configuring amount of the compound of formula I is an amount of the compound of formula I sufficient to increase expression of one or more MHC class II genes in the tumor microenvironment of the patient.
28. The method of claim 27 wherein increasing the expression of one or more MHC class II genes in the tumor microenvironment comprises increasing the expression of H2-Aa, H2-Eb1, or both.
29. The method of claim 1, wherein the histone deacetylase inhibitor is HBI-8000, vorinostat, romidepsin, panobinostat, belinostat, entinostat, mocetinostat, givinostat, practinostat, quisinostat, abexinostat, chr-3996, or AR-42.
30. The method claim 1, wherein the method further comprises administering to the patient an immune checkpoint inhibitor.
31. The method of claim 30, wherein the immune checkpoint inhibitor comprises an inhibitor of VISTA, PD-L1, CTLA-4, PD-L2, B7-1 (CD80), B7-2 (CD86), B7-H3 (CD276), B7-H2, B7-H4 (VTCN1), HVEM (CD270, TNFRSF14), Galectin 9, Galectin3, CEACAM1 (CD66a), OX-2 (CD200), PVR (CD155), PVRL2 (Nectin-2, CD112), FGL-1, PECAM-1, TSG-6, CD47, Stabilin-1 (Clever-1), Neuropilin 1, Neuropilin 2, CD158 (family), IGSF2 (CD101), CD155, GITRL, CD137L, OX40L, LIGHT, CD70, PD-1, RGMB, CTLA-4 (CD152), BTLA, CD160, Tim-3, CD200R, TIGIT, CD112R (PVRIG), LAG-3 (CD223), PECAM-1, CD44, SIRP alpha (CD172a), or a combination thereof.
32. The method of claim 1, wherein the tumor microenvironment configuring amount of the compound of formula I is an amount greater than about 5 mg per administration.
33. The method of claim 1, wherein the tumor microenvironment configuring amount of the compound of formula I is an amount of about 5 mg to about 50 mg per administration.
34. The method claim 1, further comprising administering an immune checkpoint inhibitor in amount of about 0.1 mg/kg to about 30 mg/kg per administration.
35. The claim 1, wherein the immune checkpoint inhibitor is present at an amount of about 0.5 mg/kg to about 15 mg/kg.
36. The method of claim 1, wherein the tumor microenvironment configuring amount of the compound of formula I is an amount sufficient to increase of the expression of one or more immune checkpoints, adaptive immunity genes, NK cell function genes, WIC class I genes, MHC class II genes, or any combination thereof, and the degree of inhibition of the one or more immune checkpoints, adaptive immunity genes, NK cell function genes, MHC class I genes, MHC class II genes is at least about 10%, about 20%, about 30%, about 40%, about 50%, about 100%, about 150%, about 2 times, about 3 times, about 4 times, about 5 times, about 10 times, about 15 times, about 20 times, or about 25 times, relative to a tumor microenvironment in a patient not administered the compound of formula I.
37. A combination comprising a therapeutically effective amount of a PD-1, PD-L1, or CTLA-4 inhibitor and a therapeutically effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof: ##STR00011## wherein, A is phenyl or a heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkythio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, C1-C4 alkoxycarbonyl, phenyl, and a heterocyclic group; B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkylthio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, C1-C4 alkoxycarbonyl, and phenyl; Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively; Z is a bond or C1-C4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--; R.sup.1 and R.sup.2 are independently hydrogen or C1-C4 alkyl; R.sup.3 is hydrogen or C1-C4 alkyl; R.sup.4 is hydrogen or --NH.sub.2; one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkylthio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, or C1-C4 alkoxycarbonyl optionally substituted with halogen or C1-C4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen, provided, however, that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group or an alkylamino group
38. The combination of claim 37, wherein the compound of formula I is: ##STR00012## or a pharmaceutically acceptable salt thereof.
39. The combination of claim 37, wherein the compound of formula I is N-(2-amino-4-fluorophenyl)-4-[[[(2E)-1-oxo-3-(3-pyridinyl)-2-propen-1-yl]- amino]methyl]benzamide.
40. The combination of claim 37, wherein the compound of formula I is administered at an amount of greater than about 5 mg per administration.
41. The combination of claim 37, wherein the compound of formula I is administered at an amount of about 5 mg to about 50 mg per administration.
42. The combination of claim 37, wherein the PD-1, PD-L1, or CTLA-4 inhibitor is a small molecule compound, a nucleic acid, a peptide, a protein, a monoclonal antibody, a human antibody, a mouse antibody, a chimeric antibody, a humanized antibody, or a chimeric humanized antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a fragment or variant thereof.
43. The combination of claim 37, wherein the PD-1, PD-L1, or CTLA-4 inhibitor is a humanized antibody comprising durvalumab, avelumab, atezolizumab, or BMS-936559.
44. The combination of claim 37, wherein the PD-1, PD-L1, or CTLA-4 inhibitor is a humanized antibody administered at an amount of about 0.1 mg/kg to about 30 mg/kg per administration.
45. The combination of claim 37, wherein the PD-1, PD-L1, or CTLA-4 inhibitor is a humanized antibody administered at an amount of about 0.5 mg/kg to about 15 mg/kg per administration.
46. The combination of claim 37, wherein the PD-1, PD-L1, or CTLA-4 inhibitor is a humanized antibody administered at an amount of about: 0.1 mg/kg, 0.3 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 5 mg/kg, 10 mg/kg, or 20 mg/kg per administration.
47. The combination claim 37, wherein the combination is suitable for administration to a cancer patient.
48. A pharmaceutical composition, comprising a combination of claim 37 and a pharmaceutically acceptable excipient.
49. A kit comprising the combination of claim 37 and optionally a pharmaceutically acceptable excipient.
50. A method for treating cancer, the method comprising administering to a cancer patient in need thereof a therapeutically effective amount of a combination of claim 37.
51. The method of claim 50, wherein the cancer is a solid tumor cancer selected from the group consisting of squamous cell carcinoma, nonsquamous cell carcinoma, non-small cell lung cancer (NSCLC), small cell lung cancer, melanoma, hepatocellular carcinoma, renal cell carcinoma, ovarian cancer, head and neck cancer, urothelial cancer, breast cancer, prostate cancer, glioblastoma, colorectal cancer, pancreatic cancer, lymphoma, leiomyosarcoma, liposarcoma, synovial sarcoma, or malignant peripheral sheath tumor (MPNST).
52. The method of claim 50, wherein the cancer is non-small cell lung cancer (NSCLC), hepatocellular carcinoma, melanoma, ovarian cancer, breast cancer, pancreatic cancer, renal cell carcinoma, or colorectal cancer.
53. The method of claim 50, wherein the cancer is lymphoma, Non-Hodgkin's lymphoma (NHL), Hodgkin's Lymphoma, Reed-Sternberg disease, multiple myeloma (MM), acute myelogenous leukemia (AML), chronic myelogenous leukemia (CML), acute lymphocytic leukemia, (ALL), or chronic lymphocytic leukemia (CLL).
54. The method of claim 50, wherein the cancer patient is treatment naive.
55. The method of claim 54, wherein the cancer patient is treatment naive for non-small cell lung cancer (NSCLC), hepatocellular carcinoma, melanoma, ovarian cancer, breast cancer, pancreatic cancer, renal cell carcinoma, or colorectal cancer.
56. The method of claim 50, wherein the compound of formula I is administered to the cancer patient as a first line therapy.
57. The method of claim 50, wherein the compound of formula I is administered to the cancer patient as a second, third, fourth, fifth, or sixth line of treatment.
58. The method of claim 50, wherein the compound of formula I is administered to the cancer patient following treatment with at least one previous anti-cancer therapy.
59. The method of claim 58, wherein the at least one previous anti-cancer therapy comprises chemotherapy, radiotherapy, surgery, targeted therapy, immunotherapy, or a combination thereof.
60. The method of claim 50, wherein the cancer is resistant to at least one anti-cancer agent.
61. The method of claim 50, wherein the compound of formula I and a PD-1 inhibitor, a PD-L1 inhibitor, or a CTLA-4 inhibitor are administered simultaneously or sequentially to the patient.
62. The method of claim 50, wherein the compound of formula I is administered 2 to 3 times per week.
63. The method of claim 50, wherein the compound of formula I is administered daily.
64. The method of claim 50, wherein a combination of a compound of formula I and one or more of a PD-1 inhibitor, a PD-L1 inhibitor, or a CTLA-4 inhibitor are administered on day 1 of an administration regimen.
65. The method of claim 50, wherein the PD-1 inhibitor, the PD-L1 inhibitor, or the CTLA-4 inhibitor is a small molecule compound, a nucleic acid, a peptide, a protein, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a variant thereof.
66. The method claim 49, wherein the PD-1 inhibitor, the PD-L1 inhibitor, or the CTLA-4 inhibitor is a PD-1, PD-L1, or CTLA-4 inhibitor antibody.
67. The method of claim 66, wherein the PD-1, PD-L1, or CTLA-4 inhibitor antibody comprises one or more of durvalumab, avelumab, atezolizumab, BMS-936559, STI-A1010, STI-A1011, STI-A1012, STI-A1013, STI-A1014, or STI-A1015 (Sorrento Therapeutics).
68. The method of claim 66, wherein the PD-1, PD-L1, or CTLA-4 inhibitor antibody comprises one or more of durvalumab, avelumab, atezolizumab, or BMS-936559.
69. The method of claim 50, wherein the combination is administered to the patient as a regimen.
70. The method of claim 69, wherein the regimen is repeated until disease progression or unacceptable toxicity.
71. The method of claim 69, wherein the regimen comprises a rest period of at least 1 day between consecutive administration periods.
72. The method of claim 69, wherein the compound of formula I of the combination is administered 2 to 3 times per week in the regimen and the PD-1, PD-L1, or CTLA-4 inhibitor antibody is administered every 2 to 3 weeks.
73. The method of claim 69, wherein the compound of formula I of the combination is administered QD for 21 days in the regimen and the PD-1, PD-L1, or CTLA-4 inhibitor antibody is administered every 2 to 3 weeks.
74. The method of claim 50, wherein the method of treating cancer inhibits metastasis of the cancer in the patient, reduces tumor or tumor burden in the patient, inhibits pre-existing metastasis of the cancer in the patient, prolongs the time to disease progression of the cancer in the patient, prolongs the survival of the patient, or increases progression-free survival of the patient.
75. A method for reducing a level of myeloid-derived suppressor cells (MDSC) or regulatory T-cells (Treg cells) in a patient in need thereof, enhancing the activity of a natural killer (NK) or cytotoxic T-cell activity in-vivo, or enhancing antibody-dependent cell-mediated cytotoxicity in a cancer patient, the method comprising administering a therapeutically effective amount of a combination of claim 37 to a patient in need thereof and determining the level of MDSCs after the administration.
76. A method for treating cancer, comprising administering a therapeutically effective amount of a combination of a histone deacetylase inhibitor (HDACi) and a PD-1, PD-L1, or CTLA-4 inhibitor to a cancer patient in need of treatment and whose cancer was previously treated with a prior therapy comprising administration of one or more of a PD-1, PD-L1, and/or CTLA-4 inhibitor in the absence of the HDACi.
77. The method of claim 76, wherein the cancer, after treatment with the prior therapy, exhibited partial response, but later developed resistance to the prior therapy, with progression of disease.
78. The method of claim 76, wherein the cancer, after treatment with the prior therapy exhibited stable disease, but later developed resistance to the prior therapy, with progression of disease.
79. The method of claim 76, wherein the cancer, after treatment with the prior therapy exhibited a complete response, but later developed resistance to the prior therapy.
80. The method of claim 76, wherein the cancer, after treatment with the prior therapy, exhibited no response to the prior therapy.
81. The method of claim 76, wherein the PD-1, PD-L1, or CTLA-4 inhibitor is a small molecule compound, a nucleic acid, a peptide, a protein, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a fragment or variant thereof.
82. The method of claim 76, wherein the PD-1, PD-L1, or CTLA-4 inhibitor comprises an antibody.
83. The method of claim 82, wherein the antibody comprises one or more of durvalumab, avelumab, atezolizumab, BMS-936559, STI-A1010, STI-A1011, STI-A1012, STI-A1013, STI-A1014, or STI-A1015.
84. The method of claim 76, wherein the HDAC inhibitor comprises a compound of formula I, or a pharmaceutically acceptable salt thereof: ##STR00013## wherein, A is phenyl or a heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkythio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, C1-C4 alkoxycarbonyl, phenyl, and a heterocyclic group; B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkylthio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, C1-C4 alkoxycarbonyl, and phenyl; Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively; Z is a bond or C1-C4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--; R.sup.1 and R.sup.2 are independently hydrogen or C1-C4 alkyl; R.sup.3 is hydrogen or C1-C4 alkyl; R.sup.4 is hydrogen or --NH.sub.2; one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 aminoalkyl, C1-C4 alkylamino, C2-C4 acyl, C2-C4 acylamino, C1-C4 alkylthio, C1-C4 perfluoroalkyl, C1-C4 perfluoroalkyloxy, or C1-C4 alkoxycarbonyl optionally substituted with halogen or C1-C4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen, provided, however, that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group or an alkylamino group.
85. The method of claim 76, wherein the HDAC inhibitor is N-(2-amino-4-fluorophenyl)-4-[[[(2E)-1-oxo-3-(3-pyridinyl)-2-propen-1-yl]- amino]methyl]benzamide.
86. The method of claim 76, wherein the HDAC inhibitor has the following formula: ##STR00014## or a pharmaceutically acceptable salt thereof.
87. The method of claim 76, wherein the HDAC inhibitor is selected from one or more of the group consisting of vorinostat, romidepsin, panobinostat, belinostat, entinostat, mocetinostat, givinostat, practinostat, quisinostat, abexinostat, chr-3996, and AR-42.
88. The method of claim 76, wherein the cancer treated is one or more of prostate, skin, ovarian cancer; cancers of non-lymphoid parenchymal organs including the heart, placenta, skeletal muscle and lung; breast cancer; cancers of the head and neck including various lymphomas, such as mantle cell lymphoma, non-Hodgkins B cell lymphoma, PTCL, adenoma, squamous cell carcinoma, laryngeal carcinoma, salivary carcinoma, thymomas and thymic carcinoma; leukemia; cancers of the retina; cancers of the esophagus; multiple myeloma; melanoma; colorectal cancer; lung cancer; cervical cancer; endometrium carcinoma; gallbladder cancer; liver cancer; thyroid follicular cancer; gastric cancer; non-small cell lung carcinoma; glioma; urotheial cancer; bladder cancer; prostate cancer; renal cell cancer; infiltrating ductal carcinoma; and glioblastoma multiform.
Description
[0001] This application claims priority under 35 U.S.C. .sctn. 119 (e) from U.S. provisional patent application No. 63/070,173, filed Aug. 25, 2020, the contents of which are fully incorporated herein by reference.
FIELD
[0002] The present invention relates to combinations of HDAC inhibitors, PD-1 inhibitors, PD-L1 inhibitors, and CTLA-4 inhibitors, among other checkpoint inhibitors, and the use of such combinations in the treatment of cancer.
BACKGROUND OF THE INVENTION
[0003] Cancer is a significant cause of morbidity and mortality worldwide. While the standards of care for many different cancer types have greatly improved over the years, current standards of care still fail to meet the need for effective therapies to improve treatment of cancer. The clinical use of immuno-oncology agents targeting cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and the programmed cell death receptor-1 (PD-1) and its ligand PD-L1, have resulted in improvements over the standard of care in the treatment of many cancer types. While these checkpoint inhibitors have produced improved clinical responses in such certain cancers, durable clinical responses only occur in approximately 10-45% of patients. Moreover, a significant number of tumors are either resistant or become refractory. Epigenetic modifiers such as histone deacetylase inhibitors (HDACi) have been successful in the treatment of some hematologic malignancies, but despite preclinical data demonstrating activity against solid tumors, this result has not translated to the clinic as a monotherapy. Accordingly, there is a need in the art for new therapies, including, for example, combination therapies for the treatment of cancers. Provided herein are solutions to these and other problems in the art.
SUMMARY OF THE INVENTION
[0004] Provided herein, inter alia, are combinations that include an HDAC inhibitor (HDACi) and a PD-L1 and/or PD-1 inhibitor, further in combination with a CTLA-4 inhibitor. The combinations include a compound of formula I and a PD-L1 and/or PD-1 inhibitor, further in combination with a CTLA-4 inhibitor. In certain instances, the PD-L1 inhibitor, PD-1 inhibitor, and/or CTLA-4 inhibitor are antibodies. In some embodiments, the combination is an HDAC inhibitor (HDACi) a PD-L1 inhibitor, and a CTLA-4 inhibitor. In some embodiments, the combination is an HDAC inhibitor (HDACi) a PD-1 inhibitor, and a CTLA-4 inhibitor.
[0005] In a first aspect of the disclosure provided herein is a combination comprising a therapeutically effective amount of a PD-L1 inhibitor, a PD-1 inhibitor, a CTLA-4 inhibitor, a CD276 inhibitor, a therapeutically effective amount of a compound of formula I, or any combination thereof, wherein formula I is:
##STR00001##
wherein, A is phenyl or a heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkythio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, phenyl, and a heterocyclic group; B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, and phenyl; Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively; Z is a bond or C.sub.1-C.sub.4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--; R.sup.1 and R.sup.2 are independently hydrogen or C.sub.1-C.sub.4 alkyl; R.sup.3 is hydrogen or C.sub.1-C.sub.4 alkyl; R.sup.4 is hydrogen or --NH.sub.2, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, or C.sub.1-C.sub.4 alkoxycarbonyl optionally substituted with halogen or C.sub.1-C.sub.4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen, provided, however, that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group or an alkylamino group. In some embodiments, said compound of formula I has the structure of formula Ia:
##STR00002##
In some embodiments, said compound of formula I is N-(2-amino-4-fluorophenyl)-4-[[[(2E)-1-oxo-3-(3-pyridinyl)-2-propen-1-yl]- amino]methyl]benzamide. In some embodiments, said PD-L1 inhibitor, PD-1 inhibitor, CTLA-4 inhibitor, and/or CD276 inhibitor is a small molecule compound, a nucleic acid, a peptide, a protein, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a fragment or variant thereof. In some embodiments, at least one of said PD-L1 inhibitor, PD-1 inhibitor, CTLA-4 inhibitor, and/or CD276 inhibitor is an antibody. In some embodiments, said inhibitor antibody is a monoclonal antibody. In some embodiments, said inhibitor antibody comprises a human antibody, a mouse antibody, a chimeric antibody, a humanized antibody, or a chimeric humanized antibody. In some embodiments, said inhibitor antibody is a human antibody or a humanized antibody. In some embodiments, said inhibitor antibody is present at an amount of about 0.1 mg/kg to about 30 mg/kg. In some embodiments, said inhibitor antibody is present at an amount of about 0.5 mg/kg to about 15 mg/kg. In some embodiments, said inhibitor antibody is present at an amount of about: 0.1 mg/kg, 0.3 mg/kg, 1 mg/kg, 2 mg/kg, 2.5 mg/kg, 3 mg/kg, 5 mg/kg, 10 mg/kg, or 20 mg/kg. In some embodiments, said combination is suitable for parenteral administration to a cancer patient. In some embodiments, said parenteral administration comprises intravenous (IV) administration.
[0006] Another aspect of the present disclosure comprises a pharmaceutical composition comprising a combination of any one of the embodiments described herein, and a pharmaceutically acceptable excipient.
[0007] Another aspect of the present disclosure comprises a kit comprising the combination of any of one of the embodiments described herein or a pharmaceutical composition of the embodiments described herein. In some embodiments, the kit further comprises at least one administration device. In some embodiments, components in the kit are sterilized.
[0008] Another aspect of the present disclosure comprises a method for treating cancer, said method comprising administering a therapeutically effective amount of a combination of any one of the embodiments described herein or a pharmaceutical composition of the embodiments described herein to a subject in need thereof. In some embodiments, said subject has a mutated BRAF gene. In some embodiments, said cancer is a solid tumor cancer selected from the group consisting of squamous cell carcinoma, nonsquamous cell carcinoma, non-small cell lung cancer (NSCLC), small cell lung cancer, melanoma, hepatocellular carcinoma, renal cell carcinoma, ovarian cancer, head and neck cancer, urothelial cancer, breast cancer, prostate cancer, glioblastoma, colorectal cancer, pancreatic cancer, lymphoma, leiomyosarcoma, liposarcoma, synovial sarcoma, or malignant peripheral sheath tumor (MPNST). In some embodiments, said cancer is non-small cell lung cancer (NSCLC), hepatocellular carcinoma, melanoma, ovarian cancer, breast cancer, pancreatic cancer, renal cell carcinoma, or colorectal cancer. In some embodiments, said cancer is lymphoma, Non-Hodgkin's lymphoma (NHL), Hodgkin's Lymphoma, Reed-Sternberg disease, multiple myeloma (MM), acute myelogenous leukemia (AML), chronic myelogenous leukemia (CML), acute lymphocytic leukemia, (ALL), or chronic lymphocytic leukemia (CLL). In some embodiments, said cancer patient is treatment naive. In some embodiments, said cancer patient is treatment naive for non-small cell lung cancer (NSCLC), hepatocellular carcinoma, melanoma, ovarian cancer, breast cancer, pancreatic cancer, renal cell carcinoma, or colorectal cancer. In some embodiments, said combination is administered to said cancer patient as a first line therapy. In some embodiments, said combination is administered to said cancer patient as a second, third, fourth, fifth, or sixth line of treatment. In some embodiments, said combination is administered to said cancer patient following treatment with at least one anti-cancer therapy. In some embodiments, said anti-cancer therapy comprises chemotherapy, radiotherapy, surgery, targeted therapy, immunotherapy, or a combination thereof. In some embodiments, said cancer is resistant to at least one anti-cancer agent. In some embodiments, said compound of formula I and said inhibitor of said combination are administered simultaneously or sequentially. In some embodiments, said compound of formula I is administered 2 to 3 times per week. In some embodiments, said compound of formula I is administered daily. In some embodiments, said PD-L1 inhibitor, PD-1 inhibitor, CTLA-4 inhibitor, and/or CD276 inhibitor and said compound of formula I are concomitantly administered on day 1 of an administration regimen. In some embodiments, said combination is administered to said patient as a regimen. In some embodiments, said regimen is repeated until disease progression or unacceptable toxicity. In some embodiments, said regimen comprises a rest period of at least 1 day between consecutive administration periods. In some embodiments, said compound of formula I of said combination is administered 2 to 3 times per week in said regimen and said PD-L1 inhibitor, PD-1 inhibitor, CTLA-4 inhibitor, and/or CD276 inhibitor is administered every 2 to 3 weeks. In some embodiments, said compound of formula I of said combination is administered once a day ("QD") for 21 days in said regimen and said inhibitor antibody is administered every 2 to 3 weeks. In some embodiments, said method of treating cancer inhibits metastasis of said cancer in said patient. In some embodiments, said method of treating cancer reduces tumor or tumor burden in said patient. In some embodiments, said method of treating cancer inhibits pre-existing metastasis of said cancer in said patient. In some embodiments, said method of treating cancer prolongs the time to disease progression of said cancer in said patient. In some embodiments, said method of treating cancer prolongs the survival of said patient. In some embodiments, said method of treating cancer increases progression-free survival of said patient.
[0009] Another aspect of the present disclosure comprises a method for treating cancer comprising administering a therapeutically effective amount of a combination of a histone deacetylase inhibitor (HDAC inhibitor) and a PD-L1 inhibitor and/or a PD-1 inhibitor, plus a CTLA-4 inhibitor, to a subject in need of treatment and whose cancer has been previously treated with a checkpoint inhibitor. A method for treating cancer comprising administering a therapeutically effective amount of a PD-L1 inhibitor, a PD-1 inhibitor, a CTLA-4 inhibitor, a CD276 inhibitor, a histone deacetylase inhibitor (HDAC inhibitor), or any combination thereof, to a subject in need of treatment and whose cancer has been previously treated with a checkpoint inhibitor.
[0010] Another aspect of the present disclosure comprises a method for treating cancer comprising administering a therapeutically effective amount of a PD-L1 inhibitor, a PD-1 inhibitor, a CTLA-4 inhibitor, a CD276 inhibitor, a histone deacetylase inhibitor (HDAC inhibitor), or any combination thereof, to a subject in need of treatment wherein said subject comprises a mutated BRAF gene. A method for treating cancer comprising administering a therapeutically effective amount of: a compound of formula I, wherein formula I is:
##STR00003##
wherein, A is phenyl or a heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkythio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, phenyl, and a heterocyclic group; B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, and phenyl; Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively; Z is a bond or C.sub.1-C.sub.4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--; R.sup.1 and R.sup.2 are independently hydrogen or C.sub.1-C.sub.4 alkyl; R.sup.3 is hydrogen or C.sub.1-C.sub.4 alkyl; R.sup.4 is hydrogen or --NH.sub.2, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, or C.sub.1-C.sub.4 alkoxycarbonyl optionally substituted with halogen or C.sub.1-C.sub.4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen, provided, however, that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group or an alkylamino group; and one or more inhibitor antibodies, wherein said one or more inhibitor antibodies comprise a PD-L1 inhibitor, a PD-1 inhibitor, a CTLA-4 inhibitor, a CD276 inhibitor, or any combination thereof, and wherein said one or more inhibitor antibodies are present at an amount of about 0.1 mg/kg to about 30 mg/kg; to a subject in need of treatment. In some embodiments, said compound of formula I has the structure of formula Ia:
##STR00004##
In some embodiments, said compound of formula I is N-(2-amino-4-fluorophenyl)-4-[[[(2E)-1-oxo-3-(3-pyridinyl)-2-propen-1-yl]- amino]methyl]benzamide. In some embodiments, said one or more inhibitor antibodies are monoclonal antibodies. In some embodiments, said one or more inhibitor antibodies comprise a human antibody, a mouse antibody, a chimeric antibody, a humanized antibody, or a chimeric humanized antibody. In some embodiments, said inhibitor antibody is a human antibody or a humanized antibody. In some embodiments, said cancer is a solid tumor cancer selected from the group consisting of squamous cell carcinoma, nonsquamous cell carcinoma, non-small cell lung cancer (NSCLC), small cell lung cancer, melanoma, hepatocellular carcinoma, renal cell carcinoma, ovarian cancer, head and neck cancer, urothelial cancer, breast cancer, prostate cancer, glioblastoma, colorectal cancer, pancreatic cancer, lymphoma, leiomyosarcoma, liposarcoma, synovial sarcoma, or malignant peripheral sheath tumor (MPNST). In some embodiments, said cancer patient is treatment naive. In some embodiments, said cancer patient is treatment naive for non-small cell lung cancer (NSCLC), hepatocellular carcinoma, melanoma, ovarian cancer, breast cancer, pancreatic cancer, renal cell carcinoma, or colorectal cancer. In some embodiments, said combination is administered to said cancer patient as a first line therapy. In some embodiments, said combination is administered to said cancer patient as a second, third, fourth, fifth, or sixth line of treatment. In some embodiments, said combination is administered to said cancer patient following treatment with at least one anti-cancer therapy. In some embodiments, said anti-cancer therapy comprises chemotherapy, radiotherapy, surgery, targeted therapy, immunotherapy, or a combination thereof. In some embodiments, said cancer is resistant to at least one anti-cancer agent. In some embodiments, said compound of formula I and said inhibitor of said combination are administered simultaneously or sequentially. In some embodiments, said compound of formula I is administered 2 to 3 times per week. In some embodiments, said compound of formula I is administered daily. In some embodiments, said PD-L1 inhibitor, PD-1 inhibitor, CTLA-4 inhibitor, and/or CD276 inhibitor and said compound of formula I are concomitantly administered on day 1 of an administration regimen. In some embodiments, said PD-L1 inhibitor, PD-1 inhibitor, CTLA-4 inhibitor, and/or CD276 inhibitor and said compound of formula I are administered to said patient as a regimen. In some embodiments, said regimen is repeated until disease progression or unacceptable toxicity. In some embodiments, said regimen comprises a rest period of at least 1 day between consecutive administration periods. In some embodiments, said compound of formula I is administered 2 to 3 times per week and said PD-L1 inhibitor, PD-1 inhibitor, CTLA-4 inhibitor, and/or CD276 inhibitor is administered every 2 to 3 weeks. In some embodiments, said compound of formula I of said combination is administered once a day ("QD") for 21 days in said regimen and said inhibitor antibody is administered every 2 to 3 weeks.
[0011] Another aspect of the present disclosure provides for a combination that includes a therapeutically effective amount of 1) a PD-L1 inhibitor and/or PD-1 inhibitor, 2) a therapeutically effective amount of a CTLA-4 inhibitor, and 3) a therapeutically effective amount of a compound of formula I:
##STR00005##
[0012] wherein A is phenyl or a heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkythio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, phenyl, and a heterocyclic group,
[0013] wherein B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, and phenyl,
[0014] wherein Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively,
[0015] wherein Z is a bond or C.sub.1-C.sub.4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--,
[0016] wherein R.sup.1 and R.sup.2 are independently hydrogen or C.sub.1-C.sub.4 alkyl,
[0017] wherein R.sup.3 is hydrogen or C.sub.1-C.sub.4 alkyl, and
[0018] wherein R.sup.4 is hydrogen or --NH.sub.2; and
[0019] wherein one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, or C.sub.1-C.sub.4 alkoxycarbonyl optionally substituted with halogen or C.sub.1-C.sub.4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen; provided, however, that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group, or an alkylamino group.
[0020] In one embodiment, the compound of formula I is N-(2-amino-4-fluorophenyl)-4-[[[(2E)-1-oxo-3-(3-pyridinyl)-2-propen-1-yl]- amino]methyl]benzamide, referred to herein as HBI-8000, or chidamide.
[0021] In another embodiment, the PD-L1 inhibitor is a small molecule compound, a nucleic acid, a peptide, a protein, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a fragment or variant thereof
[0022] In still another embodiment, the PD-L1 inhibitor is an antibody.
[0023] In yet another embodiment, the PD-L1 inhibitor antibody is selected from durvalumab, avelumab, atezolizumab, BMS-936559, STI-A1010, STI-A1011, STI-A1012, STI-A1013, STI-A1014, or STI-A1015 (Sorrento Therapeutics).
[0024] In another embodiment, the PD-1 inhibitor is a small molecule compound, a nucleic acid, a peptide, a protein, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a fragment or variant thereof
[0025] In still another embodiment, the PD-1 inhibitor is an antibody.
[0026] In yet another embodiment, the PD-1 antibody is selected from nivolumab, pembrolizumab, pidilizumab, REGN2810 (also known as SAR-439684), PDR001, SHR-1210 or MEDI0680.
[0027] In another embodiment, the CTLA-4 inhibitor is a small molecule compound, a nucleic acid, a peptide, a protein, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a fragment or variant thereof.
[0028] In still another embodiment, the CTLA-4 inhibitor is an antibody.
[0029] In yet another embodiment, the CTLA-4 antibody is ipilimumab.
[0030] In another aspect is a pharmaceutical composition that includes a combination described herein and a pharmaceutically acceptable excipient.
[0031] In still another aspect is a kit that includes a combination or a pharmaceutical composition as described herein.
[0032] In still another aspect is a method for treating cancer by administering a therapeutically effective amount of a combination or a pharmaceutical composition described herein to a patient in need thereof.
BRIEF DESCRIPTION OF THE DRAWINGS
[0033] The novel features of the invention are set forth with particularity in the appended claims. A better understanding of the features and advantages of the present invention will be obtained by reference to the following detailed description that sets forth illustrative embodiments, in which the principles of the invention are utilized, and the accompanying drawings of which:
[0034] FIG. 1 shows median tumor volume amongst treatment groups including a combination of compounds of formula I, a CTLA-4 inhibitory antibody, and a PD-1 inhibitory antibody. The dosing of each treatment is indicated by the arrows below the graph.
[0035] FIG. 2 shows a Kaplan-Meier survival graph for the same experimental groups from FIG. 1.
[0036] FIG. 3A shows the probability of progression free survival ("PFS") in terms of months resulting from a combination therapy comprising compounds of formula I and Nivolumab in melanoma.
[0037] FIG. 3B shows the PFS for patients treated with Nivolumab monotherapy, ipilimumab monotherapy, or a Nivolumab plus ipilimumab combination therapy.
[0038] FIG. 4 shows checkpoint inhibitor ("CPI")-naive subjects dosed with compounds of formula in combination with nivolumab.
[0039] FIG. 5 shows total time on treatment regime, termination reason, and best ORR for melanoma subjects treated with compounds of formula I and a PD-1 inhibitory antibody.
[0040] FIG. 6A shows immune gene activation in response to administration of the compounds of formula I, a PD-1 inhibitory antibody, and a combination of the compounds of formula I and a PD-1 inhibitory antibody.
[0041] FIG. 6B shows improvement on survival amongst the experimental group treated with the combination therapy compared to the compounds of formula I alone or the PD-1 inhibitory antibody alone.
[0042] FIG. 7A shows an estimated PFS for relapsed or refractory peripheral T-cell lymphoma ("RR/PTCL") patients given the compounds of formula I were used as a monotherapy.
[0043] FIG. 7B shows an estimated survival graph for relapsed or refractory peripheral T-cell lymphoma ("RR/PTCL") patients given the compounds of formula I were used as a monotherapy.
[0044] FIGS. 8A-8K shows Tumor growth inhibition (TGI) in mice treated with ICI, HBI-8000, or their combination. Syngeneic MC38 (A-D), RENCA (E&F), CT26 (G&H), and A20 (I & J) tumors were implanted in C57BL/6 or BALB/c mice, and allowed to grow until the mean tumor volume was .about.100 mm3. Animals were then randomized into groups with equivalent mean tumor volumes and treated with the indicated therapeutic agents. Data shown in FIG. 8K represent the median tumor volume for each treatment group at the indicated day post-initiation of therapy (FIGS. 8A, 8C, 8E, 8G, and 8I), as well as the individual tumor volumes per animal (FIGS. 8B, 8D, 8F, 8H, and 8J).
[0045] FIGS. 9A-9B shows Immune cell-types and pathways modulated by PD-1 Ab, HBI-8000, or their combination. Syngeneic MC38 tumors were implanted in C57BL/6 mice and allowed to grow until the mean tumor volume was .about.100 mm3. The mice were then randomized into groups of 20 mice with equivalent mean tumor volumes and treated with the indicated therapeutic agents. At days 7, 14, and 17, groups of 20 mice were killed, and the tumors were excised, fixed in formalin, and embedded in paraffin. Tumor sections were then processed for nCounter gene expression analysis as described in the Methods. FIG. 9A. Plots of the immune cell types in the TME modulated by PD-1 Ab, HBI-8000, or their combination at days 7, 14, and 17 for each treatment group. FIG. 9B. Immune checkpoints (PD1, PD-L1, CTLA4, CD86, CD276, and CD244) modulated by PD-1 Ab, HBI-8000, or their combination. The data depict the mRNA expression levels for each gene at days 7, 14, and 17. Statistical significance is as indicated in the graphs. Individual mice were tagged according to the antitumor response. Red circles (.cndot.) represent TGI>75%, inverted green triangles () TGI from 25% through 75%, and blue squares (.quadrature.) were assigned to mice with TGI<25%.
[0046] FIG. 10 shows Expression analyses of TNF.alpha., KLRD1, CCR5, CCL2, CD137, and IRF4.
[0047] FIG. 11 shows TME immune response-relevant markers modulated by PD-1 Ab, HBI-8000, or their combination. Expression of IL-2R.alpha., CD8.alpha., CCR1, ENTPD1, GZMB, and PRF1 in tumors isolated from mice in the Vehicle, HBI-8000, PD-1 Ab, and the combination of HBI-8000 and PD-1 Ab groups.
[0048] FIG. 12 shows expression of cytokine/chemokine receptors, MHC class I and class II are modulated by PD-1 Ab, HBI-8000, or their combination. nCounter data analyses identified significant differences in the expression of IL-7R, CXCR6, CX3CR1, CXCR3, H2-Aa, H2-Eb1, H2-D1, and H2-K1 in tumors treated with PD-1 Ab, HBI-8000, or their combination compared to the Vehicle-treated group.
[0049] FIG. 13 shows ICI (PD-L1 Ab) plus HBI-8000 reverses resistance to PD-1 Ab therapy and rescues mice with MC38 tumors progressing on PD-1 Ab therapy. Mice implanted with MC38 tumors were treated with PD-1 Ab as a first-line therapy for 18-21 days, at which point mice displaying stable or slow tumor growth were randomized into 1 of 6 second-line treatment groups, including Vehicle, HBI-8000, PD-1 Ab, PD-1 Ab plus HBI-8000, PD-L1 Ab, and PD-L1 Ab plus HBI-8000. Data shown represent individual tumor volumes per animal in each treatment cohort.
[0050] FIGS. 14A-14B. FIG. 14A. shows a heatmap showing the raw abundance of different immune cell types in the tumor microenvironment (TME) modulated by PD-1 Ab, HBI-8000, or their combination at day 17 for each tumor sample. Orange indicates high abundance and blue indicates low abundance. FIG. 14B. shows a heatmap of the directed global significance scores for immune pathway types in the TME modulated by PD-1 Ab, HBI-8000, or their combination at day 17 for each treatment group compared with the control, as well as the directed global (all groups regardless of treatment) significance scores for immune pathway types modulated in nonresponders (TGI<25%) vs. responders (TGI>75%), and partial responders (TGI<75%, >25%) vs. responders. Directed global significance statistics measure the extent to which a gene set's genes are upregulated or downregulated vs. the control. Red denotes gene sets whose genes exhibit extensive overexpression with the covariate, and blue denotes gene sets with extensive underexpression. Left Y-axis depicts the various immune pathway types.
[0051] FIG. 15 shows analysis of expression of LAG-3, TIGIT, NT5E, SIRP.alpha., NFATC4, and CD155 in MC38 tumors treated with vehicle, HBI-8000, PD-1 Ab, and the combination of HBI-8000 and PD-1 Ab using the NanoString nCounter PanCancer Immune Profiling Panel, as described in the FIG. 9 legend and in the Methods section.
[0052] FIG. 16 shows expression of CD40L, CD40, ICOS, NKG7, KLRC2, and KLRK1 in MC38 tumors harvested from mice treated with vehicle, HBI-8000, PD-1 Ab, and the combination of HBI-8000 and PD-1 Ab.
DETAILED DESCRIPTION
Definitions
[0053] All patents, applications, published applications and other publications cited herein are incorporated by reference in their entirety. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by those of ordinary skill in the art to which the invention belongs. The chemical structures and formulae set forth herein are constructed according to the standard rules of chemical valency known in the chemical arts. Should a discrepancy exist between a depicted structure and a name given for that structure, the depicted structure is to be accorded more weight. Where the stereochemistry of a structure or a portion of a structure is not indicated in a depicted structure or a portion of the depicted structure, the depicted structure is to be interpreted as encompassing all of its possible stereoisomers.
[0054] Any methods, devices and materials similar or equivalent to those described herein can be used in the practice of this invention. The following definitions are provided to facilitate understanding of certain terms used frequently herein and are not meant to limit the scope of the present disclosure. In the event that there is a plurality of definitions for a term herein, those in this section prevail unless stated otherwise. Headings used herein are for organizational purposes only and in no way limit the invention described herein.
[0055] The term "PD-L1 inhibitor" refers to a moiety (e.g., compound, nucleic acid, polypeptide, antibody) that decreases, inhibits, blocks, abrogates or interferes with the activity, binding of PD-L1 to its receptor, PD-1, or expression of PD-L1 (e.g., Programmed Cell Death 1 Ligand; PD-L1 (CD274); GI: 30088843), including variants, isoforms, species homologs of human PD-L1 (e.g., mouse) and analogs that have at least one common epitope with PD-L1. A PD-L1 inhibitor includes molecules and macromolecules such as, for example, compounds (small molecule compounds), nucleic acids, polypeptides, antibodies, peptibodies, diabodies, minibodies, single-chain variable fragments (ScFv), and fragments or variants thereof. Thus, a PD-L1 inhibitor as used herein refers to any moiety that antagonizes PD-L1 activity, its binding to PD-1, or its expression. PD-L1 inhibitor efficacy can be measured, for example, by its inhibitor concentration at 50% (half-maximal inhibitor concentration or IC.sub.50). PD-L1 inhibitors include exemplary compounds and compositions described herein. A PD-L1 inhibitor antibody refers to a PD-L1 inhibitor which is a monoclonal or polyclonal antibody as described herein.
[0056] The terms "durvalumab," "avelumab," "atezolizumab," "BMS-936559," "STI-A1010," "STI-A1011," "STI-A1012," "STI-A1013," "STI-A1014," and "STI-A1015" are used in accordance with their plain and ordinary meaning as understood in the art.
[0057] The term "PD-1 inhibitor" refers to a moiety (e.g., compound, nucleic acid, polypeptide, antibody) that decreases, inhibits, blocks, abrogates or interferes with the activity or expression of PD-1 (e.g., Programmed Cell Death Protein 1; PD-1 (CD279); GI: 145559515), including variants, isoforms, species homologs of human PD-1 (e.g., mouse) and analogs that have at least one common epitope with PD-1. A PD-1 inhibitor includes molecules and macromolecules such as, for example, compounds, nucleic acids, polypeptides, antibodies, peptibodies, diabodies, minibodies, single-chain variable fragments (ScFv), and fragments or variants thereof. Thus, a PD-1 inhibitor as used herein refers to any moiety that antagonizes PD-1 activity or expression. PD-1 inhibitor efficacy can be measured, for example, by its inhibitor concentration at 50% (half-maximal inhibitor concentration or IC.sub.50). PD-1 inhibitors include exemplary compounds and compositions described herein. A PD-1 antibody refers to a PD-1 inhibitor which is a monoclonal or polyclonal antibody as described herein.
[0058] The terms "nivolumab," "pembrolizumab," "pidilizumab," "AMP-224," "REGN2810," "PDR 001,", "SHR-1210", "SAR-439684" and "MEDI0680" are used in accordance with their plain and ordinary meaning as understood in the art.
[0059] The term "CTLA-4 inhibitor" refers to a moiety (e.g., compound, nucleic acid, polypeptide, antibody) that decreases, inhibits, blocks, abrogates or interferes with the activity or expression of CTLA-4, including variants, isoforms, species homologs of human CTLA-4 (e.g., mouse) and analogs that have at least one common epitope with CTLA-4. A CTLA-4 inhibitor includes molecules and macromolecules such as, for example, compounds, nucleic acids, polypeptides, antibodies, peptibodies, diabodies, minibodies, single-chain variable fragments (ScFv), and fragments or variants thereof. Thus, a CTLA-4 inhibitor as used herein refers to any moiety that antagonizes CTLA-4 activity or expression. CTLA-4 inhibitor efficacy can be measured, for example, by its inhibitor concentration at 50% (half-maximal inhibitor concentration or IC.sub.50). CTLA-4 inhibitors include exemplary compounds and compositions described herein. A CTLA-4 antibody refers to a CTLA-4 inhibitor which is a monoclonal or polyclonal antibody as described herein.
[0060] The term "ipilimumab" is used in accordance with their plain and ordinary meaning as understood in the art.
[0061] The term "CD276 inhibitor" refers to a moiety (e.g., compound, nucleic acid, polypeptide, antibody) that decreases, inhibits, blocks, abrogates or interferes with the activity or expression of CD276 (also referred to as B7-H3), including variants, isoforms, species homologs of human CD276 (e.g., mouse) and analogs that have at least one common epitope with CD276. A CD276 inhibitor includes molecules and macromolecules such as, for example, compounds, nucleic acids, polypeptides, antibodies, peptibodies, diabodies, minibodies, single-chain variable fragments (ScFv), and fragments or variants thereof. Thus, a CD276 inhibitor as used herein refers to any moiety that antagonizes CD276 activity or expression. CD276 inhibitor efficacy can be measured, for example, by its inhibitor concentration at 50% (half-maximal inhibitor concentration or IC.sub.50). CD276 inhibitors include exemplary compounds and compositions described herein. A CD276 antibody refers to a CD276 inhibitor which is a monoclonal or polyclonal antibody as described herein.
[0062] The terms "polypeptide" and "protein" are used interchangeably herein and refer to any molecule that includes at least 2 or more amino acids.
[0063] The term "Inhibitor Antibody" refers to a monoclonal or polyclonal antibody that binds to its substrate or target with sufficient strength to inhibit activity of the substrate or target. As used herein, an Inhibitor Antibody comprises a PD-L1 inhibitor antibody, PD-1 inhibitor antibody, CTLA-4 inhibitor antibody, and/or CD276 inhibitor antibody.
[0064] The term "effective amount" refers to the amount of a therapy (e.g., a combination provided herein or another active agent such as an anti-cancer agent described herein) which is sufficient to accomplish a stated purpose or otherwise achieve the effect for which it is administered. An effective amount can be sufficient to reduce and/or ameliorate the progression, development, recurrence, severity and/or duration of a given disease, disorder or condition and/or a symptom related thereto, or can be sufficient to reduce the level of activity or binding of a polypeptide (e.g., PD-L1, PD-1, CTLA-4). An effective amount can be a "therapeutically effective amount" which refers to an amount sufficient to provide a therapeutic benefit such as, for example, the reduction or amelioration of the advancement or progression of a given disease, disorder or condition, reduction or amelioration of the recurrence, development or onset of a given disease, disorder or condition, and/or to improve or enhance the prophylactic or therapeutic effect(s) of another therapy. A therapeutically effective amount of a composition described herein can enhance the therapeutic efficacy of another therapeutic agent.
[0065] The term "regimen" refers to a protocol for dosing and timing the administration of one or more therapies (e.g., combinations described herein or another active agent such as an anti-cancer agent described herein) for treating a disease, disorder, or condition described herein. A regimen can include periods of active administration and periods of rest as known in the art. Active administration periods include administration of combinations and compositions described herein and the duration of time of efficacy of such combinations and compositions. Rest periods of regimens described herein include a period of time in which no compound is actively administered, and in certain instances, includes time periods where the efficacy of such compounds can be minimal. Combination of active administration and rest in regimens described herein can increase the efficacy and/or duration of administration of the combinations and compositions described herein.
[0066] The terms "therapies" and "therapy" refer to any protocol(s), method(s), and/or agent(s) that can be used in the prevention, treatment, management, and/or amelioration of a disease, disorder, or condition or one or more symptoms thereof. In certain instances the term refers to active agents such as an anti-cancer agent described herein. The terms "therapy" and "therapy" can refer to anti-viral therapy, anti-bacterial therapy, anti-fungal therapy, anti-cancer therapy, biological therapy, supportive therapy, and/or other therapies useful in treatment, management, prevention, or amelioration of a disease, disorder, or condition or one or more symptoms thereof known to one skilled in the art, for example, a medical professional such as a physician.
[0067] The term "patient" or "subject" refers to a mammal, such as a human, bovine, rat, mouse, dog, monkey, ape, goat, sheep, cow, or deer. Generally a patient as described herein is human.
[0068] The terms "inhibition", "inhibit", "inhibiting" refer to a reduction in the activity, binding, or expression of a polypeptide or reduction or amelioration of a disease, disorder, or condition or a symptom thereof. Inhibiting as used here can include partially or totally blocking stimulation, decreasing, preventing, or delaying activation or binding, or inactivating, desensitizing, or down-regulating protein or enzyme activity or binding.
[0069] Antibodies described herein can be polyclonal or monoclonal and include xenogeneic, allogeneic, or syngeneic forms and modified versions thereof (e.g., humanized or chimeric). An "antibody" is intended to mean a polypeptide product of B cells within the immunoglobulin class of polypeptides that is able to bind to a specific molecular antigen and is composed of two identical pairs of polypeptide chains, wherein each pair has one heavy chain (about 50-70 kDa) and one light chain (about 25 kDa) and each amino-terminal portion of each chain includes a variable region of about 100 to about 130 or more amino acids and each carboxy-terminal portion of each chain includes a constant region (See Borrebaeck (ed.) (1995) Antibody Engineering, Second Edition, Oxford University Press.; Kuby (1997) Immunology, Third Edition, W.H. Freeman and Company, New York). Specific molecular antigens that can be bound by an antibody described herein include PD-L1, PD-1, CTLA-4, and their epitopes.
[0070] The term "monoclonal antibody(ies)" refers to a population of antibody molecules that contain one species of an antigen binding site capable of immunoreacting with a particular epitope of an antigen, whereas the term "polyclonal antibody(ies)" refers to a population of antibody molecules that contain multiple species of antigen binding sites capable of interacting with a particular antigen. A monoclonal antibody, typically displays a single binding affinity for a particular antigen with which it immunoreacts. For example, the monoclonal antibodies to be used in accordance with the present invention can be made by a variety of techniques, including, for example, the hybridoma method (e.g., Kohler and Milstein, Nature, 256:495-97 (1975); Hongo et al., Hybridoma, 14 (3): 253-260 (1995), Harlow et al., Antibodies: A Laboratory Manual, (Cold Spring Harbor Laboratory Press, 2nd ed. 1988); Hammerling et al., in: Monoclonal Antibodies and T-Cell Hybridomas 563-681 (Elsevier, N.Y., 1981)), recombinant DNA methods (see, e.g., U.S. Pat. No. 4,816,567), phage-display technologies (see, e.g., Clackson et al., Nature, 352: 624-628 (1991); Marks et al., J Mol. Biol. 222: 581-597 (1992); Sidhu et al., J. Mal. Biol. 338(2): 299-310 (2004); Lee et al., J. Mal. Biol. 340(5): 1073-1093 (2004); Fellouse, Proc. Natl. Acad. Sci. USA 101(34): 12467-12472 (2004); and Lee et al., J. Immunol. Methods 284(1-2): 119-132 (2004), and technologies for producing human or human-like antibodies in animals that have parts or all of the human immunoglobulin loci or genes encoding human immunoglobulin sequences (see, e.g., WO 1998/24893; WO 1996/34096; WO 1996/33735; WO 1991/10741; Jakobovits et al., Proc. Natl. Acad. Sci. USA 90: 2551 (1993); Jakobovits et al., Nature 362: 255-258 (1993); Bruggemann et al., Year in Immunol. 7:33 (1993); U.S. Pat. Nos. 5,545,807; 5,545,806; 5,569,825; 5,625,126; 5,633,425; and U.S. Pat. No. 5,661,016; Marks et al., Bio/Technology 10: 779-783 (1992); Lon berg et al., Nature 368: 856-859 (1994); Morrison, Nature 368: 812-813 (1994); Fishwild et al., Nature Biotechnol. 14: 845-851 (1996); Neuberger, Nature Biotechnol. 14: 826 (1996); and Lonberg and Huszar, Intern. Rev. Immunol. 13: 65-93 (1995).
[0071] The monoclonal antibodies herein also include "chimeric" antibodies (immunoglobulins) in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is(are) identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (U.S. Pat. No. 4,816,567; Morrison et al., Proc. Natl. Acad. Sci. USA, pp. 6851-6855 (1984)). "Humanized antibody(ies)" can be considered as a subset of chimeric antibodies described herein.
[0072] The term "human" when used in reference to an antibody or a functional fragment thereof (e.g., "humanized antibody(ies))" refers an antibody or functional fragment thereof that has a human variable region or a portion thereof corresponding to human germline immunoglobulin sequences. Such human germline immunoglobulin sequences are described by Kabat et al. (1991) Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91-3242. A human antibody, in the context of the present invention, can include an antibody that binds to PD-L1 or variants thereof as described herein.
[0073] In certain instances a human antibody is an antibody that possesses an amino acid sequence corresponding to that of an antibody produced by a human and/or has been made using any of the techniques for making human antibodies as disclosed herein. Human antibodies can be produced using various techniques known in the art, including phage-display libraries. Hoogenboom and Winter, J. Mol. Biol., 227:381 (1991); Marks et al., J. Mal. Biol., 222:581 (1991). Also available for the preparation of human monoclonal antibodies are methods described in Cole et al., Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, p. 77 (1 985); Boemer et al., J. Immunol., 147(1):86-95 (1991). See also van Dijk and van de Winkel, Curr. Opin. Pharmacol., 2:368-74 (2001). Human antibodies can be prepared by administering the antigen to a transgenic animal that has been modified to produce such antibodies in response to antigenic challenge, but whose endogenous loci have been disabled, e.g., immunized xenomice (see, e.g., U.S. Pat. Nos. 6,075.181 and 6, 150,584 regarding XENOMOUSE technology). See also, for example, Li et al., Proc. Natl. Acad. Sci. USA, 103:3557-3562 (2006) regarding human antibodies generated via a human B-cell hybridoma technology.
[0074] A "humanized antibody" refers to antibodies made by a non-human cell having variable or variable and constant regions which have been altered to more closely resemble antibodies that would be made by a human cell. For example, by altering the non-human antibody amino acid sequence to incorporate amino acids found in human germline immunoglobulin sequences. The humanized antibodies of the invention can include amino acid residues not encoded by human germline immunoglobulin sequences (e.g., mutations introduced by random or site-specific mutagenesis in vitro or by somatic mutation in vivo), for example in the CDRs. Humanized antibodies can also include antibodies in which CDR sequences derived from the germline of another mammalian species, such as a mouse, have been grafted onto human framework sequences.
[0075] Humanized forms of non-human (e.g., murine) antibodies are antibodies that contain minimal sequence derived from non-human immunoglobulin. In one embodiment, a humanized antibody is a human immunoglobulin (recipient antibody) in which residues from a hypervariable region of the recipient are replaced by residues from an hypervariable region of a nonhuman species (donor antibody) such as mouse, rat, rabbit or non-human primate having the desired specificity, affinity, and/or capacity. In some instances, framework ("FR") residues of the human immunoglobulin are replaced by corresponding non-human residues. Furthermore, humanized antibodies can comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications can be made to further refine antibody performance, such as binding affinity. In general, a humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the hypervariable loops correspond to those of a non-human immunoglobulin sequence, and all or substantially all of the FR regions are those of a human immunoglobulin sequence, although the FR regions can include one or more individual FR residue substitutions that improve antibody performance, such as binding affinity, isomerization, immunogenicity, etc. The number of these amino acid substitutions in the FR are typically no more than 6 in the H chain, and in the L chain, no more than 3. The humanized antibody optionally can also include at least a portion of an immunoglobulin constant region (Fc), which can be a human immunoglobulin. Exemplary methods and humanized antibodies include those described by Jones et al. Nature 321:522-525 (1986); Riechmann et al. Nature 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol. 2:593-596 (1992); Vaswani and Hamilton, Ann. Allergy. Asthma & Immunol. 1:105-115 (1998); Harris, Biochem. Soc. Transactions 23:1035-1038 (1995); Burle and Gross, Curr. Op. Biotech. 5:428-433 (1994); and U.S. Pat. Nos. 6,982,321 and 7,087,409.
[0076] The term "functional fragment" when used in reference to an antibody refers to a portion of the antibody including heavy or light chain polypeptides that retains some or all of the binding activity as the antibody from which the fragment was derived. Such functional fragments can include, for example, an Fd, Fv, Fab, F(ab'), F(ab).sub.2, F(ab').sub.2, single chain Fv (ScFv), diabody, triabody, tetrabody and minibody. Other functional fragments can include, for example, heavy or light chain polypeptides, variable region polypeptides or CDR polypeptides or portions thereof so long as such functional fragments retain binding activity. Such antibody binding fragments can be found described in, for example, Harlow and Lane, Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory, New York (1989); Myers (ed.), Molec. Biology and Biotechnology: A Comprehensive Desk Reference, New York: VCH Publisher, Inc.; Huston et al., Cell Biophysics, 22:189-224 (1993); Phickthun and Skerra, Meth. Enzymol., 178:497-515 (1989) and in Day, E.D., Advanced Immunochemistry, Second Ed., Wiley-Liss, Inc., New York, N.Y. (1990). Antibody Engineering, Second Edition, Oxford University Press, 1995.
[0077] The term "heavy chain" when used in reference to an antibody refers to a polypeptide chain of about 50-70 kDa, wherein the amino-terminal portion includes a variable region of about 120 to 130 or more amino acids and a carboxy-terminal portion that includes a constant region. The constant region can be one of five distinct types, referred to as alpha (.alpha.), delta (.delta.), epsilon (.epsilon.), gamma (.gamma.) and mu (.mu.), based on the amino acid sequence of the heavy chain constant region. The distinct heavy chains differ in size: .alpha., .delta. and .gamma. contain approximately 450 amino acids, while .mu. and .epsilon. contain approximately 550 amino acids. When combined with a light chain, these distinct types of heavy chains give rise to five well known classes of antibodies, IgA, IgD, IgE, IgG and IgM, respectively, including four subclasses of IgG, namely IgG1, IgG2, IgG3 and IgG4. A heavy chain can be a human heavy chain.
[0078] The term "light chain" when used in reference to an antibody refers to a polypeptide chain of about 25 kDa, wherein the amino-terminal portion includes a variable region of about 100 to about 110 or more amino acids and a carboxy-terminal portion that includes a constant region. The approximate length of a light chain is 211 to 217 amino acids. There are two distinct types, referred to as kappa (.kappa.) of lambda (.lamda.) based on the amino acid sequence of the constant domains. Light chain amino acid sequences are well known in the art. A light chain can be a human light chain.
[0079] The term "variable domain" or "variable region" refers to a portion of the light or heavy chains of an antibody that is generally located at the amino-terminal of the light or heavy chain and has a length of about 120 to 130 amino acids in the heavy chain and about 100 to 110 amino acids in the light chain, and are used in the binding and specificity of each particular antibody for its particular antigen. The variable domains can differ extensively in sequence between different antibodies. The variability in sequence is concentrated in the CDRs while the less variable portions in the variable domain are referred to as framework regions (FR). The CDRs of the light and heavy chains are primarily responsible for the interaction of the antibody with antigen. Numbering of amino acid positions used herein is according to the EU Index, as in Kabat et al. (1991) Sequences of proteins of immunological interest. (U.S. Department of Health and Human Services, Washington, D.C.) 5.sup.th Ed. A variable region can be a human variable region.
[0080] A CDR refers to one of three hypervariable regions (H1, H2 or H3) within the non-framework region of the immunoglobulin (Ig or antibody) VH .beta.-sheet framework, or one of three hypervariable regions (L1, L2 or L3) within the non-framework region of the antibody VL .beta.-sheet framework. Accordingly, CDRs are variable region sequences interspersed within the framework region sequences. CDR regions are well known to those skilled in the art and have been defined by, for example, Kabat as the regions of most hypervariability within the antibody variable (V) domains (Kabat et al., J. Biol. Chem. 252:6609-6616 (1977); Kabat, Adv. Prot. Chem. 32:1-75 (1978)). CDR region sequences also have been defined structurally by Chothia as those residues that are not part of the conserved .beta.-sheet framework, and thus are able to adapt different conformations (Chothia and Lesk, J. Mol. Biol. 196:901-917 (1987)). Both terminologies are well recognized in the art. The positions of CDRs within a canonical antibody variable domain have been determined by comparison of numerous structures (Al-Lazikani et al., J. Mol. Biol. 273:927-948 (1997); Morea et al., Methods 20:267-279 (2000)). Because the number of residues within a hypervariable region varies in different antibodies, additional residues relative to the canonical positions are conventionally numbered with a, b, c and so forth next to the residue number in the canonical variable domain numbering scheme (Al-Lazikani et al., supra (1997)). Such nomenclature is similarly well known to those skilled in the art.
[0081] For example, CDRs defined according to either the Kabat (hypervariable), Chothia (structural), or MacCallum (J. Mol. Biol. 262:732-745 (1996)) designations, as set forth in the Table 1 below:
TABLE-US-00001 TABLE 1 CDR Definitions Kabat.sup.1 Chothia .sup.2 MacCallum .sup.3 Loop Location V.sub.H CDR1 31-35 26-32 30-35 linking B and C strands V.sub.H CDR2 50-65 53-55 47-58 linking C' and C'' strands V.sub.H CDR3 95-102 96-101 93-101 linking F and G strands V.sub.L CDR1 24-34 26-32 30-36 linking B and C strands V.sub.L CDR2 50-56 50-52 46-55 linking C' and C'' strands V.sub.L CDR3 89-97 91-96 89-96 linking F and G strands .sup.1Residue numbering follows the nomenclature of Kabat et al., supra .sup.2 Residue numbering follows the nomenclature of Chothia et al., supra
[0082] The term "cancer" refers to any physiological condition in mammals characterized by unregulated cell growth. Cancers described herein include solid tumors and hematological (blood) cancers. A "hematological cancer" refers to any blood borne cancer and includes, for example, myelomas, lymphomas and leukemias. A "solid tumor" or "tumor" refers to a lesion and neoplastic cell growth and proliferation, whether malignant or benign, and all pre-cancerous and cancerous cells and tissues resulting in abnormal tissue growth. "Neoplastic," as used herein, refers to any form of dysregulated or unregulated cell growth, whether malignant or benign, resulting in abnormal tissue growth.
[0083] The terms "treating" or "treatment" refer to any indicia of success or amelioration of the progression, severity, and/or duration of a disease, pathology or condition, including any objective or subjective parameter such as abatement; remission; diminishing of symptoms or making the injury, pathology or condition more tolerable to the patient; slowing in the rate of degeneration or decline; making the final point of degeneration less debilitating; or improving a patient's physical or mental well-being.
[0084] The term "enhance" refers to an increase or improvement in the function or activity of a protein or cell after administration or contacting with a combination described herein compared to the protein or cell prior to such administration or contact.
[0085] The term "administering" refers to the act of delivering a combination or composition described herein into a subject by such routes as oral, mucosal, topical, suppository, intravenous, parenteral, intraperitoneal, intramuscular, intralesional, intrathecal, intranasal or subcutaneous administration. Parenteral administration includes intravenous, intramuscular, intra-arteriole, intradermal, subcutaneous, intraperitoneal, intraventricular, and intracranial administration. Administration generally occurs after the onset of the disease, disorder, or condition, or its symptoms but, in certain instances, can occur before the onset of the disease, disorder, or condition, or its symptoms (e.g., administration for patients prone to such a disease, disorder, or condition).
[0086] The term "coadministration" refers to administration of two or more agents (e.g., a combination described herein and another active agent such as an anti-cancer agent described herein). The timing of coadministration depends in part of the combination and compositions administered and can include administration at the same time, just prior to, or just after the administration of one or more additional therapies, for example cancer therapies such as chemotherapy, hormonal therapy, radiotherapy, or immunotherapy. The compound of the invention can be administered alone or can be coadministered to the patient. Coadministration is meant to include simultaneous or sequential administration of the compound individually or in combination (more than one compound or agent). Thus, the preparations can also be combined, when desired, with other active substances (e.g., to reduce metabolic degradation). The compounds described herein can be used in combination with one another, with other active agents known to be useful in treating cancer.
[0087] The term "anti-cancer agent" is used in accordance with its plain ordinary meaning and refers to a composition having anti-neoplastic properties or the ability to inhibit the growth or proliferation of cells. In embodiments, an anti-cancer agent is a chemotherapeutic. In embodiments, an anti-cancer agent is an agent identified herein having utility in methods of treating cancer. In embodiments, an anti-cancer agent is an agent approved by the FDA or similar regulatory agency of a country other than the USA, for treating cancer.
[0088] The term "chemotherapeutic" or "chemotherapeutic agent" is used in accordance with its plain ordinary meaning and refers to a chemical composition or compound having anti-neoplastic properties or the ability to inhibit the growth or proliferation of cells. "Chemotherapy" refers to a therapy or regimen that includes administration of a chemotherapeutic or anti-cancer agent described herein.
[0089] The terms "halo," "halogen," and "halide" refer to --F, --Cl, --Br, and --I.
[0090] The term "alkyl" by itself or as part of another substituent refers to, unless otherwise stated, a straight (e.g., unbranched) or branched carbon chain (or carbon), or combination thereof, having no unsaturation and can include mono-, di- and multivalent radicals. An alkyl as defined herein can be designated by its number of carbon atoms (e.g., C.sub.1-C.sub.10 means one to ten carbons). Alkyls herein can include C.sub.1-C.sub.10, C.sub.1-C.sub.8, C.sub.1-C.sub.6, and C.sub.1-C.sub.4 lengths. A "perfluoroalkyl" refers to an alkyl in which all of the hydrogens in the alkyl chain are replaced with fluoro.
[0091] The term "alkoxy" refers to an alkyl group (e.g., C.sub.1-C.sub.10, C.sub.1-C.sub.8, C.sub.1-C.sub.6, and C.sub.1-C.sub.4 alkyl) attached to the remainder of the molecule via an oxygen linker (--O--). Exemplary alkoxy groups include groups having the formula --OR, where R is branched or linear alkyl. A "perfluoroalkoxyl" moiety refers to an alkoxy in which all of the hydrogens in the alkyl chain are replaced with fluoro.
[0092] The term "aminoalkyl" refers to an alkyl group (e.g., C.sub.1-C.sub.10, C.sub.1-C.sub.8, C.sub.1-C.sub.6, and C.sub.1-C.sub.4 alkyl) in which one or more hydrogen atoms are replaced with an amino group
[0093] The term "alkylamino" refers to an alkyl group (e.g., C.sub.1-C.sub.10, C.sub.1-C.sub.8, C.sub.1-C.sub.6, and C.sub.1-C.sub.4 alkyl) attached to the remainder of the molecule via a nitrogen linker (--NR--). Exemplary alkylamino groups include N-methylamino, N-ethylamino, N-isopropylamino, and the like.
[0094] The term "acyl" refers to a moiety having the formula, --C(O)R, where R is a substituted or unsubstituted alkyl, haloalkyl, or amino group. The term "acylamino" refers to an acyl moiety having an attached amino group and includes, for example, such moieties as acetylamino, propionylamino, butyrylamino, isobuytrylamino, and others.
[0095] The term "alkythio" refers to an alkyl group (e.g., C.sub.1-C.sub.10, C.sub.1-C.sub.8, C.sub.1-C.sub.6, and C.sub.1-C.sub.4 alkyl) attached to the remainder of the molecule via a sulfur linker (--S--). Exemplary alkylthio groups include methylthio, ethylthio, propylthio, and others.
[0096] The term "heterocycle" or "heterocyclyl" refers to a stable 3- to 15-membered monocyclic group that is saturated or unsaturated and contains one or more heteroatoms (e.g., N, O, or S). Exemplary heterocycles include, but are not limited to morpholinyl, piperidinyl, piperazinyl, pyranyl, pyrrolidinyl, pyrrolinyl, imidazolidinyl, imidazolinyl, oxetanyl, azetidinyl, and others.
[0097] 1. Compositions
[0098] Provided herein are combinations (e.g., combination therapies and compositions) useful for treating a variety of diseases, disorders, and symptoms thereof, including for example, cancer. The combinations described herein include an HDAC inhibitor and a PD-L1 inhibitor and/or PD-1 inhibitor, and further a CTLA-4 inhibitor. In one non-limiting example a benzamide HDAC inhibitor of formula I is provided, and examples of PD-L1 inhibitors, PD-1 inhibitors, and CTLA-4 inhibitors are described herein. In one aspect is a combination that includes a therapeutically effective amount of a PD-L1 inhibitor and/or PD-1 inhibitor, a CTLA-4 inhibitor, and a therapeutically effective amount of a compound of formula I:
##STR00006##
wherein:
[0099] A is a phenyl or heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkythio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, phenyl, and a heterocyclic group;
[0100] B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, and phenyl;
[0101] Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively;
[0102] Z is a bond or C.sub.1-C.sub.4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--;
[0103] R.sup.1 and R.sup.2 are independently hydrogen or C.sub.1-C.sub.4 alkyl;
[0104] R.sup.3 is hydrogen or C.sub.1-C.sub.4 alkyl;
[0105] R.sup.4 is hydrogen or --NH.sub.2; and
[0106] one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, or C.sub.1-C.sub.4 alkoxycarbonyl optionally substituted with halogen or C.sub.1-C.sub.4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen,
[0107] provided that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group or an alkylamino group.
[0108] In certain instances A is phenyl or phenyl optionally substituted with halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkythio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, phenyl, or a heterocyclic group. A can be a heterocyclic group (e.g., a 5 to 10-membered heterocyclic group) containing a --N--, --S--, or --O-- moiety. In certain instances A is a 5 to 10-membered N-heterocyclic moiety having 1, 2, 3, 4, or more nitrogen heteroatoms, such as for example, pyrrolidinyl, pyrrolinyl, imidazolidinyl, imdazolyl, pyrazolidinyl, pyrazolyl, oxazolidinyl, oxazolyl, thiazolidinyl, thiazolyl, piperidinyl, pyridinyl, piperizinyl, diazinyl, tetrazolyl, triazinyl, tetrazinyl, azepinyl, diazepinyl, azocanyl, or azocinyl. A can be a saturated or unsaturated 5 to 10 membered N-heterocyclic moiety. In certain instances A is a 6-membered N-heterocyclic moiety, such as for example, pyridine.
[0109] In certain embodiments, B is phenyl. B can be phenyl optionally substituted with a small moiety such as, for example, halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, or C.sub.1-C.sub.4 alkyl. In some embodiments B is phenyl substituted with halogen. In other embodiments, B is substituted with an electron donating group (EDG). In still other embodiments, B is phenyl substituted with an electron withdrawing group (EWG). In yet another embodiment, B is phenyl substituted with C.sub.1-C.sub.4 alkyl. B can be methyl-, ethyl-, or propyl-substituted phenyl. B can be methoxy-, ethoxy-, or propoxy-substituted phenyl.
[0110] In certain instances Y is --C(O)NH--CH.sub.2--. In certain embodiments, Z is a bond. Z can be a methylene, ethylene, or propylene moiety. In some embodiments, Z is --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--.
[0111] R.sup.1 and R.sup.2 are in certain instances both hydrogen. R.sup.1 and R.sup.2 can both be C.sub.1-C.sub.4 alkyl, for example, R.sup.1 and R.sup.2 can both be methyl, ethyl, or propyl. In certain instances if one of R.sup.1 or R.sup.2 is hydrogen the other is C.sub.1-C.sub.4 alkyl (e.g., methyl). R.sup.3 can be hydrogen. In other embodiments, R.sup.3 is C.sub.1-C.sub.4 alkyl (e.g., methyl or ethyl).
[0112] R.sup.4 can be --NH.sub.2. In certain instances R.sup.4 is --NH.sub.2 where one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen. When R.sup.4 is --NH.sub.2, X.sup.2 or X.sup.3 can be halogen. In one embodiment R.sup.4 is --NH.sub.2 and X.sup.2 is halogen. In such instances X.sup.2 can be --F.
[0113] In another embodiment, R.sup.1, R.sup.2, and R.sup.3 are hydrogen where Z is a bond, R.sup.4 is --NH.sub.2 and Y is --C(O)NH--CH.sub.2--. In such embodiments, A can be a heterocyclic moiety as described above and B can be phenyl. X.sup.1, X.sup.2, X.sup.3, or X.sup.4 can be halogen (e.g., --F) or --NH.sub.2.
[0114] The compound of formula I can be a compound as substantially described by U.S. Pat. Nos. 7,244,751 and 7,550,490 both of which are incorporated herein by reference in their entirety for all purposes. In one embodiment the compound of formula I is N-(2-amino-4-fluorophenyl)-4-[[[(2E)-1-oxo-3-(3-pyridinyl)-2-propen-1-yl]- amino]methyl]benzamide. In another embodiment the compound of formula I has the formula Ia as set forth below:
##STR00007##
[0115] Compounds of formula I as described herein include pharmaceutically acceptable salts, pharmaceutically acceptable stereoisomers, prodrugs, enantiomers, diastereomers, hydrates, co-crystals, and polymorphs thereof.
[0116] In certain instances, the combination includes a compound of formula I (e.g., Ia) present at an amount of greater than about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg. The combination can include a compound of formula I present at an amount greater than about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 6 mg, 7 mg, 8 mg, 9 mg, or 10 mg. In certain instances the compound of formula I is present in an amount greater than about 5 mg or about 10 mg. The combination can include a compound of formula I present at an amount greater than about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg.
[0117] The combination can include a compound present in an amount of at least about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg. The combination can include a compound of formula I present at an amount of at least about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 6 mg, 7 mg, 8 mg, 9 mg, or 10 mg. In certain instances the compound of formula I is present in an amount of at least about 5 mg or about 10 mg. The combination can include a compound of formula I present at an amount of at least about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg.
[0118] The combination can include a compound present in an amount of about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg. The combination can include a compound of formula I present at an amount of about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 6 mg, 7 mg, 8 mg, 9 mg, or 10 mg. In certain instances the compound of formula I is present in an amount of about 5 mg or about 10 mg. The combination can include a compound of formula I present at an amount of about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg.
[0119] A compound of formula I can be present in the combinations described herein relative to the weight of the patient (e.g., mg/kg). In some instances, the compound of formula I is present in an amount equivalent to about: 0.0001 mg/kg to about 200 mg/kg, 0.001 mg/kg to about 200 mg/kg, 0.01 mg/kg to about 200 mg/kg, 0.01 mg/kg to about 150 mg/kg, 0.01 mg/kg to about 100 mg/kg, 0.01 mg/kg to about 50 mg/kg, 0.01 mg/kg to about 25 mg/kg, 0.01 mg/kg to about 10 mg/kg, or 0.01 mg/kg to about 5 mg/kg, 0.05 mg/kg to about 200 mg/kg, 0.05 mg/kg to about 150 mg/kg, 0.05 mg/kg to about 100 mg/kg, 0.05 mg/kg to about 50 mg/kg, 0.05 mg/kg to about 25 mg/kg, 0.05 mg/kg to about 10 mg/kg, or 0.05 mg/kg to about 5 mg/kg, 0.5 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 150 mg/kg, 0.5 mg/kg to about 100 mg/kg, 0.5 mg/kg to about 50 mg/kg, 0.5 mg/kg to about 25 mg/kg, 0.5 mg/kg to about 10 mg/kg, or 0.5 mg/kg to about 5 mg/kg. In other instances the compound of formula I is present in an amount equivalent to about: 1 mg/kg to about 200 mg/kg, 1 mg/kg to about 150 mg/kg, 1 mg/kg to about 100 mg/kg, 1 mg/kg to about 50 mg/kg, 1 mg/kg to about 25 mg/kg, 1 mg/kg to about 10 mg/kg, or 1 mg/kg to about 5 mg/kg.
PD-L1 Inhibitors
[0120] PD-L1 inhibitors useful in the combinations described herein include any molecule capable of inhibiting, blocking, abrogating or interfering with the binding of PD-L1 to PD-1, activity or expression of PD-L1. In particular, a PD-L1 inhibitor can be a small molecule compound, a nucleic acid, a polypeptide, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a functional fragment or variant thereof. In one instance the PD-L1 inhibitor is a small molecule compound (e.g., a compound having a molecule weight of less than about 1000 Da). In one embodiment, the PD-L1 inhibitor is CA-170 (AUPM-170; Curis, Inc.), In other instances, useful PD-L1 inhibitors in the combinations described herein include nucleic acids and polypeptides. The PD-L1 inhibitor can be a polypeptide (e.g., macrocyclic polypeptide) such as those exemplified in U.S. Patent Application Publication No.: 2014/0294898, which is incorporated herein by reference in its entirety and for all purposes. In one example, the PD-L1 inhibitor is an antibody, peptibody, diabody, minibody, ScFv, or a functional fragment thereof. In another example, the PD-L1 inhibitor is a PD-L1 inhibitor antibody. The PD-L1 inhibitor antibody can be a monoclonal or polyclonal antibody. In certain embodiments, the PD-L1 inhibitor antibody is a monoclonal antibody.
[0121] PD-L1 antibodies include all known types of antibodies and functional fragments thereof, including but not limited to, those exemplified herein such as, for example, human antibodies, mouse antibodies, chimeric antibodies, humanized antibodies, or chimeric humanized antibodies.
[0122] In one embodiment, the PD-L1 inhibitor antibody is a human antibody. In another embodiment, the PD-L1 inhibitor antibody is a mouse antibody. In still another embodiment, the PD-L1 inhibitor antibody is a chimeric antibody. In yet another embodiment, the PD-L1 inhibitor antibody is a humanized antibody. In yet another embodiment, the PD-L1 inhibitor antibody is a chimeric humanized antibody. The PD-L1 inhibitor antibody can be a human antibody or humanized antibody. The PD-L1 inhibitor antibody can be durvalumab, avelumab, atezolizumab, BMS-936559, STI-A1010, STI-A1011, STI-A1012, STI-A1013, STI-A1014, or STI-A1015. In some embodiments, two or more PD-L1 antibodies are administered in combination with a compound of formula I as described herein.
[0123] The PD-L1 inhibitor antibody can be durvalumab. Durvalumab is an Fc optimized monoclonal antibody directed against PD-L1, with potential immune checkpoint inhibitory and anti-neoplastic activities. Without being bound by any particular theory, durvalumab binds to PD-L1, thereby blocking its binding to and activation of its receptor, PD-1, which can be expressed on activated T-cells. This can reverse T-cell inactivation and activate the immune system to exert a cytotoxic T-lymphocyte (CTL) response against PD-L1-expressing tumor cells. The Fc region of durvalumab is modified in such a way that it does not induce either antibody-dependent cytotoxicity (ADCC) or complement-dependent cytotoxicity (CDC).
[0124] The PD-L1 inhibitor antibody can be avelumab. Avelumab is a human immunoglobulin G1 (IgG1) monoclonal antibody directed against PD-L1, with potential immune checkpoint inhibitory and anti-neoplastic activities. Without being bound by any particular theory, avelumab binds to PD-L1 and prevents the interaction of PD-L1 with its receptor, PD-1. This inhibits the activation of PD-1 and its downstream signaling pathways. This can restore immune function through the activation of cytotoxic T-lymphocytes (CTLs) targeted to PD-L1-overexpressing tumor cells. Avelumab appears to induce an antibody-dependent cellular cytotoxic (ADCC) response against PD-L1-expressing tumor cells.
[0125] The PD-L1 inhibitor antibody can be atezolizumab. Atezolizumab is a human, Fc optimized, monoclonal antibody directed against the protein ligand PD-L1, with potential immune checkpoint inhibitory and anti-neoplastic activities. Without being bound by any particular theory, atezolizumab binds to PD-L1, blocking its binding to and activation of its receptor, PD-1, expressed on activated T-cells, which may enhance the T-cell-mediated immune response to neoplasms and reverse T-cell inactivation. In addition, by binding to PD-L1, atezolizumab also appears to prevent binding of PD-L1 to B7.1 expressed on activated T cells, which can further enhance the T-cell-mediated immune response. The Fc region of atezolizumab is modified in such a way that it does not induce either antibody-dependent cytotoxicity (ADCC) or complement-dependent cytotoxicity (CDC).
[0126] The PD-L1 inhibitor antibody can be BMS-936559. BMS-936559 is a fully human IgG4 monoclonal antibody directed against PD-L1, with potential immune checkpoint inhibitory activity. Without being bound by any particular theory, BMS-936559 binds to PD-L1 and inhibits its binding to both PD-1 and CD80.
[0127] The PD-L1 inhibitor antibody can be STI-A1010, STI-A1011, STI-A1012, STI-A1013, STI-A1014, or STI-A1015. STI-A1010, STI-A1011, STI-A1012, STI-A1013, STI-A1014, and STI-A1015 (Sorrento Therapeutics) are fully human monoclonal antibodies that are each directed against PD-L1.
PD-1 Inhibitors
[0128] PD-1 inhibitors useful in the combinations described herein include any molecule capable of inhibiting, blocking, abrogating or interfering with the activity or expression of PD-1. In particular, a PD-1 inhibitor can be a small molecule compound, a nucleic acid, a polypeptide, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a functional fragment or variant thereof. In one instance the PD-1 inhibitor is a small molecule compound (e.g., a compound having a molecule weight of less than about 1000 Da.) In other instances, useful PD-1 inhibitors in the combinations described herein include nucleic acids and polypeptides. The PD-1 inhibitor can be a polypeptide (e.g., macrocyclic polypeptide) such as those exemplified in U.S. Patent Application Publication No.: 2014/0294898, which is incorporated herein by reference in its entirety and for all purposes. In one example, the PD-1 inhibitor is an antibody, peptibody, diabody, minibody, ScFv, or a functional fragment thereof. In one example, the PD-1 inhibitor is AMP-224 (GSK).
[0129] AMP-224 is a recombinant fusion protein comprising an extracellular domain of the PD-1 ligand programmed cell death ligand 2 (PD-L2) and an Fc region of human IgG. Certain cancers can evade and suppress the immune system, in part, and without being bound by any particular theory by interactions between PD-1 and B7-H1. AMP-224 appears to block this interaction and therefore appears to overcome immune suppression.
[0130] In another example, the PD-1 inhibitor is a PD-1 antibody. The PD-1 antibody can be a monoclonal or polyclonal antibody. In certain embodiments, the PD-1 antibody is a monoclonal antibody.
[0131] PD-1 antibodies include all known types of antibodies and functional fragments thereof, including but not limited to, those exemplified herein such as, for example, human antibodies, mouse antibodies, chimeric antibodies, humanized antibodies, or chimeric humanized antibodies.
[0132] In one embodiment, the PD-1 antibody is a human antibody. In another embodiment, the PD-1 antibody is a mouse antibody. In still another embodiment, the PD-1 antibody is a chimeric antibody. In yet another embodiment, the PD-1 antibody is a humanized antibody. In yet another embodiment, the PD-1 antibody is a chimeric humanized antibody. The PD-1 antibody can be a human antibody or humanized antibody. The PD-1 antibody can be nivolumab, pembrolizumab, pidilizumab, REGN2810, PDR 001, or MEDI0680. In some embodiments, two or more PD-1 antibodies are administered in combination with a compound of formula I as described herein.
[0133] The PD-1 antibody can be nivolumab. Nivolumab (marketed as OPDIVO) is a fully human monoclonal antibody directed against PD-1 with immunopotentiation activity. Without being bound by any particular theory, nivolumab binds to and blocks the activation of PD-1 by its cognate ligands, resulting in the activation of T-cells and cell-mediated immune responses against tumor cells or pathogens.
[0134] The PD-1 antibody can be pembrolizumab. Pembrolizumab (MK-3475, marketed as KEYTRUDA) is a humanized monoclonal IgG4 antibody directed against human cell surface receptor PD-1 with potential immunopotentiating activity. Without being bound by any particular theory, pembrolizumab binds to PD-1, an inhibitory signaling receptor expressed on the surface of activated T cells, and blocks the binding to and activation of PD-1 by its cognate ligands. The blocking of binding and activity results in the activation of T-cell-mediated immune responses against tumor cells.
[0135] The PD-1 antibody can be pidilizumab. Pidilizumab (CT-011) is a humanized monoclonal antibody directed against human PD-1 with immunomodulating and antitumor activities. Without being bound by any particular theory, pidilizumab blocks interaction between the receptor PD-1 with its ligands, resulting in the attenuation of apoptotic processes in lymphocytes, primarily effector/memory T cells, and the augmentation of the anti-tumor activities of NK cells.
[0136] The PD-1 antibody can be REGN2810. REGN2810 is a human monoclonal antibody directed against PD-1, with potential immune checkpoint inhibitory and anti-neoplastic activity. Without being bound by any particular theory REGN2810 binds to PD-1, inhibits binding to its cognate ligand, and prevents the activation of its downstream signaling pathways. This can restore immune function through the activation of cytotoxic T-cells.
[0137] The PD-1 antibody can be PDR 001. PDR 001 is a fully humanized monoclonal antibody directed against PD-1, with immune checkpoint inhibitory and anti-neoplastic activities. Without being bound by any particular theory, PDR 001 binds to PD-1 expressed on activated T-cells and blocks the interaction with its cognate ligands. The inhibition of ligand binding prevents PD-1-mediated signaling and results in both T-cell activation and the induction of T-cell-mediated immune responses against tumor cells.
[0138] The PD-1 antibody can be MEDI0680 (AMP-514) is a monoclonal antibody directed against the PD-1, with potential immunomodulating and anti-neoplastic activity. Without being bound by any particular theory, MEDI0680 appears to inhibit the activation of PD-1 and its downstream signaling pathways. This inhibition can restore immune function through the activation both of T-cells and cell-mediated immune responses against PD-1 overexpressing tumor cells.
CTLA-4 Inhibitors
[0139] CTLA-4 inhibitors useful in the combinations described herein include any molecule capable of inhibiting, blocking, abrogating or interfering with the activity or expression of CTLA-4. In particular, a CTLA-4 inhibitor can be a small molecule compound, a nucleic acid, a polypeptide, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a functional fragment or variant thereof. In one instance the CTLA-4 inhibitor is a small molecule compound (e.g., a compound having a molecule weight of less than about 1000 Da.) In other instances, useful CTLA-4 inhibitors in the combinations described herein include nucleic acids and polypeptides. The CTLA-4 inhibitor can be a polypeptide (e.g., macrocyclic polypeptide). In one example, the CTLA-4 inhibitor is an antibody, peptibody, diabody, minibody, ScFv, or a functional fragment thereof. In one example, the CTLA-4 inhibitor is ipilimumab.
[0140] In another example, the CTLA-4 inhibitor is a CTLA-4 antibody. The CTLA-4 antibody can be a monoclonal or polyclonal antibody. In certain embodiments, the CTLA-4 antibody is a monoclonal antibody.
[0141] CTLA-4 antibodies include all known types of antibodies and functional fragments thereof, including but not limited to, those exemplified herein such as, for example, human antibodies, mouse antibodies, chimeric antibodies, humanized antibodies, or chimeric humanized antibodies. In one embodiment, the CTLA-4 antibody is a human antibody. In another embodiment, the CTLA-4 antibody is a mouse antibody. In still another embodiment, the CTLA-4 antibody is a chimeric antibody. In yet another embodiment, the CTLA-4 antibody is a humanized antibody. In yet another embodiment, the CTLA-4 antibody is a chimeric humanized antibody. The CTLA-4 antibody can be a human antibody or humanized antibody. The CTLA-4 antibody can be administered in combination with a compound of formula I as described herein.
CD276 Inhibitors
[0142] CD276 (B7-H3) is a relatively newly discovered, but important member of the immune checkpoint family. CD276 is expressed on antigen-presenting cells in active/inflamed "hot" tumor micro-environments ("TMEs") and suppresses CD8.sup.+ cytotoxic T cells. CD276 expression is upregulated with administration of a compound of formula I as described herein. CD276 inhibitors useful in the combinations described herein include any molecule capable of inhibiting, blocking, abrogating or interfering with the activity or expression of CD276. In particular, a CD276 inhibitor can be a small molecule compound, a nucleic acid, a polypeptide, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or a functional fragment or variant thereof. In one instance the CD276 inhibitor is a small molecule compound (e.g., a compound having a molecule weight of less than about 1000 Da.) In other instances, useful CD276 inhibitors in the combinations described herein include nucleic acids and polypeptides. The CD276 inhibitor can be a polypeptide (e.g., macrocyclic polypeptide). In one example, the CD276 inhibitor is an antibody, peptibody, diabody, minibody, ScFv, or a functional fragment thereof.
[0143] In another example, the CD276 inhibitor is a CD276 antibody. The CD276 antibody can be a monoclonal or polyclonal antibody. In certain embodiments, the CD276 antibody is a monoclonal antibody.
[0144] CD276 antibodies include all known types of antibodies and functional fragments thereof, including but not limited to, those exemplified herein such as, for example, human antibodies, mouse antibodies, chimeric antibodies, humanized antibodies, or chimeric humanized antibodies.
[0145] In one embodiment, the CD276 antibody is a human antibody. In another embodiment, the CD276 antibody is a mouse antibody. In still another embodiment, the CD276 antibody is a chimeric antibody. In yet another embodiment, the CD276 antibody is a humanized antibody. In yet another embodiment, the CD276 antibody is a chimeric humanized antibody. The CD276 antibody can be a human antibody or humanized antibody. The CD276 antibody can be administered in combination with a compound of formula I as described herein, or with any of the other compositions described herein.
[0146] A PD-L1 inhibitor antibody, PD-1 inhibitor antibody, CTLA-4 inhibitor antibody, and/or CD276 inhibitor antibody (any one of which is referred to as "Inhibitor Antibody" herein) can be of any antibody isotype. The term isotype refers to the antibody class that is encoded by heavy chain constant region genes. The heavy chains of a given antibody or functional fragment determine the class of that antibody or functional fragment: IgM, IgG, IgA, IgD or IgE. Each class can have either .kappa. or .lamda.. light chains. The term subclass refers to the minor differences in amino acid sequences of the heavy chains that differentiate the subclasses. In humans there are two subclasses of IgA (subclasses IgA1 and IgA2) and there are four subclasses of IgG (subclasses IgG1, IgG2, IgG3 and IgG4). Such classes and subclasses are well known to those skilled in art.
[0147] Useful Inhibitor Antibodies bind to their substrates with sufficient strength to inhibit activity of the substrate (e.g., PD-L1, PD-1, CTLA-4, and/or CD276). The term bind as used herein refers to an interaction between molecules to form a complex. Interactions can be, for example, non-covalent interactions including hydrogen bonds, ionic bonds, hydrophobic interactions, and/or van der Waals interactions. A complex can also include the binding of two or more molecules held together by covalent or non-covalent bonds, interactions or forces. Binding of an antibody or functional fragment thereof can be detected using, for example, an enzyme-linked immunosorbant assay or any one of a number of methods that are well known to those skilled in the art.
[0148] The strength of the total non-covalent interactions between a single antigen-binding site on an Inhibitor Antibody or functional fragment and a single epitope of a target molecule is the affinity of the antibody or functional fragment for that epitope. The ratio of association (k.sub.1) to dissociation (k.sub.1) of an antibody or functional fragment thereof to a monovalent antigen (k.sub.1/k.sub.1) is the association constant K, which is a measure of affinity. The value of K varies for different complexes of antibody or functional fragment and antigen and depends on both k.sub.1 and k.sub.-1. The association constant K for an antibody or functional fragment of the invention can be determined using any method provided herein or any other method well known to those skilled in the art.
[0149] The affinity at one binding site does not always reflect the true strength of the interaction between an antibody or functional fragment and an antigen. When complex antigens containing multiple, repeating antigenic determinants come in contact with antibodies containing multiple binding sites, the interaction of such an antibody or functional fragment with antigen at one site will increase the probability of a reaction at a second site. The strength of such multiple interactions between a multivalent antibody and antigen is called the avidity. The avidity of an antibody or functional fragment can be a better measure of its binding capacity than is the affinity of its individual binding sites. For example, high avidity can compensate for low affinity as is sometimes found for pentameric IgM antibodies, which can have a lower affinity than IgG, but the high avidity of IgM, resulting from its multivalence, enables it to bind antigen effectively.
[0150] The specificity of an Inhibitor Antibody or functional fragment thereof refers to the ability of an individual antibody or functional fragment thereof to react with only one antigen (e.g., a single epitope of PD-L1, PD-1, and CTLA-4). An antibody or functional fragment can be considered specific when it can distinguish differences in the primary, secondary or tertiary structure of an antigen or isomeric forms of an antigen.
[0151] The Inhibitor Antibody can be present in an amount as a measure with regards to the weight of the patient in need thereof. For example, the Inhibitor Antibody can be present in an amount of about: 0.1 mg/kg to about 50 mg/kg, 0.1 mg/kg to about 40 mg/kg, 0.1 mg/kg to about 30 mg/kg, 0.1 mg/kg to about 25 mg/kg, 0.1 mg/kg to about 20 mg/kg, 0.1 mg/kg to about 15 mg/kg, 0.1 mg/kg to about 10 mg/kg, 0.1 mg/kg to about 7.5 mg/kg, 0.1 mg/kg to about 5 mg/kg, 0.1 mg/kg to about 2.5 mg/kg, or about 0.1 mg/kg to about 1 mg/kg. The Inhibitor Antibody can be present in an amount of about: 0.5 mg/kg to about 50 mg/kg, 0.5 mg/kg to about 40 mg/kg, 0.5 mg/kg to about 30 mg/kg, 0.5 mg/kg to about 25 mg/kg, 0.5 mg/kg to about 20 mg/kg, 0.5 mg/kg to about 15 mg/kg, 0.5 mg/kg to about 10 mg/kg, 0.5 mg/kg to about 7.5 mg/kg, 0.5 mg/kg to about 5 mg/kg, 0.5 mg/kg to about 2.5 mg/kg, or about 0.5 mg/kg to about 1 mg/kg. The Inhibitor Antibody can be present in an amount of about 0.5 mg/kg to about 5 mg/kg or about 0.1 mg/kg to about 10 mg/kg. The Inhibitor Antibody can be present in an amount of about 0.1 mg/kg to about 20 mg/kg or about 0.1 mg/kg to about 30 mg/kg.
[0152] In still other embodiments, the Inhibitor Antibody can be present at an amount of about: 0.1 mg/kg, 0.5 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 10 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg, 35 mg/kg, 40 mg/kg, or 50 mg/kg. The Inhibitor Antibody can be present at an amount of about: 1 mg/kg, 2 mg/kg, 3 mg/kg, 5 mg/kg, 10 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, or 30 mg/kg. The Inhibitor Antibody can be present at an amount of about: 3 mg/kg, 10 mg/kg, 20 mg/kg, or 30 mg/kg.
[0153] The Inhibitor Antibody can be present in the combination at an amount of about: 1 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 40 mg, 50 mg, 60 mg, 70 mg, 75 mg, 80 mg, 90 mg, 100 mg, 150 mg, or 200 mg. The Inhibitor Antibody can be present in the combination at an amount of about: 250 mg, 300 mg, 400 mg, 500 mg, 600 mg, 700 mg, 800 mg, 900 mg, 1000 mg, 1100 mg, 1200 mg, 1300 mg, 1400 mg, 1500 mg, 1600 mg, 1700 mg, 1800 mg, 1900 mg, or 2000 mg. The Inhibitor Antibody can be present in the combination at an amount of about 1000 mg to about 2000 mg. The Inhibitor Antibody can be present in the combination at an amount of about: 1 mg to about 10 mg, 10 mg to about 20 mg, 25 mg to about 50 mg, 30 mg to about 60 mg, 40 mg to about 50 mg, 50 mg to about 100 mg, 75 mg to about 150 mg, 100 mg to about 200 mg, 200 mg to about 500 mg, 500 mg to about 1000 mg, 1000 mg to about 1200 mg, 1000 mg to about 1500 mg, 1200 mg to about 1500 mg, or 1500 to about 2000 mg.
[0154] The Inhibitor Antibody can be present in the combination in an amount of about 0.1 mg/mL, 0.5 mg/mL, 1 mg/mL, 2 mg/mL, 3 mg/mL, 4 mg/mL, 5 mg/mL, 6 mg/mL, 7 mg/mL, 8 mg/mL, 9 mg/mL, 10 mg/mL, 15 mg/mL, 20 mg/mL, 25 mg/mL, 30 mg/mL, 40 mg/mL, 50 mg/mL, 60 mg/mL, 70 mg/mL, 80 mg/mL, 90 mg/mL, 100 mg/mL, 150 mg/mL, 200 mg/mL, 250 mg/mL, 300 mg/mL, 400 mg/mL, or 500 mg/mL. In one embodiment, the Inhibitor Antibody is present in the combination in an amount of about: 1 mg/mL to about 10 mg/mL, 5 mg/mL to about 10 mg/mL, 5 mg/mL to about 15 mg/mL, 10 mg/mL to about 25 mg/mL; 20 mg/mL to about 30 mg/mL; 25 mg/mL to about 50 mg/mL, or 50 mg/mL to about 100 mg/mL.
[0155] In certain instances the therapeutically effective amount of an Inhibitor Antibody is determined as an amount provided in a package insert provided with the Inhibitor Antibody. The term package insert refers to instructions customarily included in commercial packages of medicaments approved by the FDA or a similar regulatory agency of a country other than the USA, which contains information about, for example, the usage, dosage, administration, contraindications, and/or warnings concerning the use of such medicaments.
[0156] Compounds of formula I as described herein can be provided in amounts that are synergistic with the amount of the PD-L1 and/or PD-1 inhibitor, and a CTLA-4 inhibitor. The term synergistic refers to a combination described herein (e.g., a compound of formula I and a PD-L1 and/or PD-1 inhibitor, plus a CTLA-4 inhibitor--including coadministration with another active agent such as an anti-cancer agent described herein) or a combination of regimens such as those described herein that is more effective than the additive effects of each individual therapy or regimen.
[0157] A synergistic effect of a combination described herein can permit the use of lower dosages of one or more of the components of the combination (e.g., a compound of formula I, or a PD-L1 inhibitor, or a PD-1 inhibitor, or a CTLA-4 inhibitor). A synergistic effect can permit less frequent administration of at least one of the administered therapies (e.g., a compound of formula I, or a PD-L1 inhibitor, or a PD-1 inhibitor, or a CTLA-4 inhibitor) to a subject with a disease, disorder, or condition described herein. Such lower dosages and reduced frequency of administration can reduce the toxicity associated with the administration of at least one of the therapies (e.g., a compound of formula I, or a PD-L1 inhibitor, or a PD-1 inhibitor, or a CTLA-4 inhibitor) to a subject without reducing the efficacy of the treatment. A synergistic effect as described herein avoid or reduce adverse or unwanted side effects associated with the use of any therapy.
[0158] 2. Pharmaceutical Compositions
[0159] Combinations described herein can be provided as a pharmaceutical composition suitable for administration via any route to a patient described herein including but not limited to: oral, mucosal (e.g., nasal, inhalation, pulmonary, sublingual, vaginal, buccal, or rectal), parenteral (e.g., subcutaneous, intravenous, bolus injection, intramuscular, or intra-arterial), topical (e.g., eye drops or other ophthalmic preparations), transdermal or transcutaneous administration to a patient.
[0160] Exemplary of dosage forms include: tablets; caplets; capsules (e.g., gelatin capsules); cachets; lozenges; suppositories; powders; gels; liquid dosage forms suitable for parenteral administration to a patient; and sterile solids (e.g., crystalline or amorphous solids) that can be reconstituted to provide liquid dosage forms suitable for parenteral administration to a patient.
[0161] Pharmaceutical compositions and dosage forms described herein typically include one or more excipients. Suitable excipients are well known to those skilled in the art of pharmacy. Whether a particular excipient is suitable for incorporation into a pharmaceutical composition or dosage form depends on a variety of factors such as, for example, the intended route of administration to the patient. Pharmaceutical compositions described herein can include other agents such as stabilizers, lubricants, buffers, and disintegrants that can reduce the rate by which an active ingredient can decompose in a particular formulation.
[0162] Pharmaceutical compositions described herein can in certain instances include additional active agents other than those in the combinations described herein (e.g., an anti-cancer agent such as those described herein) in an amount provided herein.
[0163] In one embodiment, the compound of formula I is provided in an oral dosage form such as a tablet or capsule. In another embodiment, the compound of formula I is supplied as a powder (e.g., lyophilized powder) that can be resuspended in a liquid suitable for parenteral administration.
[0164] PD-L1 inhibitors, PD-1 inhibitors, and CTLA-4 inhibitors described herein can be provided in forms convenient to or facilitate their administration to a patient. For example, where the inhibitor is an Inhibitor Antibody as described herein, the inhibitor can be formulated as a ready to use solution for parenteral administration. In other examples, the inhibitor, including for example an Inhibitor Antibody, can be formulated as a powder (e.g., lyophilized powder) that can be resuspended in a liquid suitable for parenteral administration. In one embodiment, the combination includes an Inhibitor Antibody formulated for intravenous administration. In still another embodiment the combination includes a compound of formula I formulated as an oral dosage form (e.g., a tablet or capsule) and an Inhibitor Antibody formulated for intravenous administration.
[0165] Combinations described herein can be provided as controlled release pharmaceutical products, which have a goal of improving drug therapy over that achieved by their non-controlled counterparts. Controlled release formulations can extend activity of the drug, reduce dosage frequency, and increase subject compliance. In addition, controlled release formulations can be used to affect the time of onset of action or other characteristics, such as blood levels of the drug, and can thus affect the occurrence of side (e.g., adverse) effects.
[0166] 3. Kits
[0167] The combinations and pharmaceutical compositions described herein can be provided as part of a kit. Such kits can, for example, improve patient compliance or improve the accuracy or ease of preparation for administering the combination. The kit includes a compound of formula I where the compound is supplied in a formulation as described herein.
[0168] Kits of the invention can include the combinations described herein having the same or different formulation. Each component of a combination described herein in a kit can be supplied in a separate, individual container. Alternatively or additionally, components of the combinations described herein can be supplied in a single container. In such instances, the container can be a container that is ready for administration to a patient in need thereof, such as for example, an IV bag, ampoule, or a syringe. In one embodiment, the compound of formula I in the kit is formulated for oral administration (e.g., a tablet, capsule, or sachet).
[0169] The contents of kits described herein can be provided in sterile form. The kit and its contents can be provided in a form that is ready for administration to the subject in need. In such instances, the components of the combination of the kit are supplied as a formulation and optionally in an administration device such that administration requires little to no further action by the user. Where kits include administration devices, such devices include devices known and understood by those skilled in the art for routes of administration described herein, such as but not limited to, syringes, pumps, bags, cups, inhalers, droppers, patches, creams, or injectors.
[0170] 4. Method
[0171] The combinations, pharmaceutical compositions, and kits described herein are useful for treating diseases, disorders, or alleviating or eliminating the symptoms of diseases and disorders such as, for example, cancer. It is to be understood that the methods described herein pertain to administration of combinations and pharmaceutical compositions described herein, and such combinations and pharmaceutical compositions can be provided in the form of a kit as described herein. Provided herein are methods of treating cancer by administering a therapeutically effective amount of a combination described herein to a patient in need thereof. Also provided herein are methods of managing cancer by administering therapeutically effective amount of a combination described herein to a patient in need thereof.
[0172] In some embodiments, the combination is used to treat cancer. In some embodiments, the cancer is a cancer described herein.
[0173] In some embodiments, the combination is an HDAC inhibitor (HDACi) a PD-L1 inhibitor, and a CTLA-4 inhibitor. In some embodiments, the combination is an HDAC inhibitor (HDACi) a PD-1 inhibitor, and a CTLA-4 inhibitor.
[0174] Combinations useful in the methods described herein include a compound of formula I:
##STR00008##
[0175] where:
[0176] A is a phenyl or heterocyclic group, optionally substituted with 1 to 4 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkythio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, phenyl, and a heterocyclic group;
[0177] B is phenyl optionally substituted with 1 to 3 substituents selected from the group consisting of halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, C.sub.1-C.sub.4 alkoxycarbonyl, and phenyl;
[0178] Y is a moiety comprising --CO-- which is linear and in which the distances between the centroid of ring B (W1), the centroid of ring A (W2) and an oxygen atom as a hydrogen bond acceptor in the moiety Y (W3) are: W1-W2=about 6.0 .ANG., W1-W3=about 3.0 .ANG. to about 6.0 .ANG., and W2-W3=about 4.0 .ANG. to about 8.0 .ANG., respectively;
[0179] Z is a bond or C.sub.1-C.sub.4 alkylene, --O--, --S--, --NH--, --CO--, --CS--, --SO--, or --SO.sub.2--;
[0180] R.sup.1 and R.sup.2 are independently hydrogen or C.sub.1-C.sub.4 alkyl;
[0181] R.sup.3 is hydrogen or C.sub.1-C.sub.4 alkyl;
[0182] R.sup.4 is hydrogen or --NH.sub.2, and
[0183] one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen, --OH, --NH.sub.2, --NO.sub.2, --CN, --COOH, C.sub.1-C.sub.4 alkyl, C.sub.1-C.sub.4 alkoxy, C.sub.1-C.sub.4 aminoalkyl, C.sub.1-C.sub.4 alkylamino, C.sub.2-C.sub.4 acyl, C.sub.2-C.sub.4 acylamino, C.sub.1-C.sub.4 alkylthio, C.sub.1-C.sub.4 perfluoroalkyl, C.sub.1-C.sub.4 perfluoroalkyloxy, or C.sub.1-C.sub.4 alkoxycarbonyl optionally substituted with halogen or C.sub.1-C.sub.4 alkyl, while the others of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 are independently hydrogen,
[0184] provided that when R.sup.4 is hydrogen, one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2, an aminoalkyl group or an alkylamino group.
[0185] Compounds of formula I useful in the methods described herein include compounds as substantially described hereinabove. In certain instances, the compound of formula I used to treat cancer in the methods provided herein includes compounds where R.sup.1, R.sup.2, and R.sup.3 are hydrogen. In certain instances Y is --C(O)NH--CH.sub.2--. In certain instances, R.sup.3 can be C.sub.1-C.sub.4 alkyl as described above. A of formula I can be a 5 to 10-membered heterocyclic moiety. In particular, and as described above, useful embodiments of the compound of formula I include compounds where A is N-heterocycle, such as for example, a 5 or 6 membered heterocyclic moiety. A can be, in certain instances, a pyridinyl.
[0186] The compound of formula I useful in the methods described herein can be a compound where R.sup.4 is --NH.sub.2 amount of 0.1 mg/kg, 0.3 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 5 mg/kg, 10 mg/kg, or 20 mg/kg and at least of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is --NH.sub.2 or halogen. In certain instances, the compound of formula I for use in the methods described herein includes compounds where R.sup.4 is --NH.sub.2 and at least one of X.sup.1, X.sup.2, X.sup.3, or X.sup.4 is halogen (e.g., --F). In one embodiment, the compound of formula I is a compound having the structure of formula Ia as set forth above.
[0187] The PD-L1 inhibitors, PD-1 inhibitors, and CTLA-4 inhibitors for use in the methods described herein are those inhibitors described herein. For example, the PD-L1 inhibitors, PD-1 inhibitors, and CTLA-4 inhibitors can be a small molecule compound, a nucleic acid, a polypeptide, an antibody, a peptibody, a diabody, a minibody, a single-chain variable fragment (ScFv), or functional fragment or variant thereof. In other examples, the inhibitor can be an Inhibitor Antibody as set forth above.
Target Cancers
[0188] The cancer can be a solid tumor. The cancer can be a hematological cancer. In certain instances, the cancer is a solid tumor selected from the group consisting of squamous cell carcinoma, non-squamous cell carcinoma, non-small cell lung cancer (NSCLC), small cell lung cancer, melanoma, hepatocellular carcinoma, renal cell carcinoma, ovarian cancer, head and neck cancer, urothelial cancer, breast cancer, prostate cancer, glioblastoma, colorectal cancer, pancreatic cancer, lymphoma, leiomyosarcoma, liposarcoma, synovial sarcoma, or malignant peripheral sheath tumor (MPNST).
[0189] In particular embodiments, the cancer is a solid tumor selected from non-small cell lung cancer (NSCLC), hepatocellular carcinoma, melanoma, ovarian cancer, breast cancer, pancreatic cancer, renal cell carcinoma, or colorectal cancer. The cancer can be non-small cell lung cancer (NSCLC). The cancer can be hepatocellular carcinoma. The cancer can be melanoma. The cancer can be ovarian cancer. The cancer can be breast cancer. The cancer can be pancreatic cancer. The cancer can be renal cell carcinoma. The cancer can be colorectal cancer.
[0190] Provided herein are methods of treating NSCLC by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and an Inhibitor Antibody. In some embodiments, the NSCLC is Stage IIA or Stage IIB. The NSCLC can be a Stage IIIA or Stage IIIB cancer. The NSCLC can be a Stage IV cancer. Staging of cancers as described herein is described by the American Joint Committee on Cancer TNM classification of malignant tumors cancer staging notation as is well understood in the art. Those of skill in the art will readily understand other staging classification systems are available and applicable to the methods described herein. In certain instances, the method is a method of treating Stage IIIA or IIIB NSCLC by administering a combination described herein that includes a compound of formula I and an Inhibitor Antibody.
[0191] Still further provided herein are methods of treating melanoma by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and an Inhibitor Antibody. In some embodiments the melanoma is a Stage IIA, IIB, or IIC cancer. In another embodiment, the melanoma is a Stage IIIA, Stage IIIB, or Stage IIIC cancer. In still another embodiment, the melanoma is a Stage IV cancer. In one aspect the method is a method of treating Stage II (e.g., Stage IIA, IIB, or IIC) melanoma by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and an Inhibitor Antibody.
[0192] Also provided herein are methods of treating breast cancer by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and an Inhibitor Antibody. The breast cancer can be HER2 negative breast cancer. The breast cancer can be a HER2 positive breast cancer. The breast cancer can be triple-negative breast cancer. In some embodiments the breast cancer is a Stage IA or Stage D3 cancer. In another embodiment, the breast cancer is a Stage IIA or Stage IIB cancer. In still another embodiment, the breast cancer is a Stage IIIA, Stage IIIB, or Stage IIIC cancer. In yet another embodiment, the breast cancer is a Stage IV cancer.
[0193] In other embodiments, the cancer is a hematological cancer selected from lymphoma, Non-Hodgkin's lymphoma (NHL), Hodgkin's Lymphoma, Reed-Sternberg disease, multiple myeloma (MM), acute myelogenous leukemia (AML), chronic myelogenous leukemia (CIVIL), acute lymphocytic leukemia, (ALL), or chronic lymphocytic leukemia (CLL). In certain embodiments, the cancer is Hodgkin's Lymphoma or Reed-Sternberg disease.
[0194] The combinations described herein can be administered to a cancer patient at any time following diagnosis. For example, the cancer patient can be treatment naive (e.g., has not received a cancer therapy for the diagnosed cancer). The cancer patient can be treatment naive for one cancer but can be diagnosed with one or more other cancers resulting from, for example, metastasis or malignancy. The cancer patient can be immune checkpoint naive for one or more cancers. The cancer patient can have a cancer that is refractory. In certain instances, the combinations described herein are administered as a first line therapy (e.g., the first therapy administered to a treatment naive cancer patient) to a patient in need thereof
[0195] However, cancer morbidity and mortality is often associated with ineffective therapy or a cancer gaining resistant to or becoming refractory to one or more cancer therapies. The combinations described herein can, therefore, be administered to patients in need thereof as a second, third, fourth, fifth, sixth, or more line of treatment. The combinations described herein can be administered to a cancer patient who has been treated with at least one anti-cancer therapy or anti-cancer agent. In certain instances the patient has received at least one anti-cancer therapy including, for example, chemotherapy, radiotherapy, surgery, targeted therapy, immunotherapy, or a combination thereof. The patient can have a cancer that is resistant/refractory to treatment with at least one anti-cancer agent.
[0196] The methods of treating cancers herein include treating subjects who have been treated with a checkpoint inhibitor and have experienced no response to treatment, or a partial response, or stable disease, but then develop resistance to treatment with progression of disease or who have experienced a complete response to treatment, but then develop resistance to treatment with progression of disease (as defined by RECIST or other criteria). Resistance is defined as disease progression during treatment or a lack of response to treatment. Such Inhibitor Antibody treatment failures can be treated with an Inhibitor Antibody in combination with an HDAC inhibitor, such as, without limitation, HBI-8000 or an HDAC inhibitor that inhibits cancer-associated Class I HDAC selected from one or more of HDAC1, HDAC2, or HDAC3. In some instances the HDAC inhibitor also inhibits Class IIb HDAC1.
[0197] Response Criteria
[0198] Recist:
[0199] RECIST is a set of established criteria or standards, internationally recognized for evaluating patient response, stability and progression in clinical trials and in the clinical practice. Originally published in 2000, and revised in 2009 (Eisenhauer E A, et al.; New response criteria in solid tumors: revised RECIST guideline (version 1.1); Eur J Cancer 2009; 45:228-47), as a joint effort of the European Organization for Research and Treatment of Cancer, the National Cancer Institute of the United States and the National Cancer Institute of Canada Clinical Trials Group, RECIST has traditionally been utilized in the evaluation of response to chemotherapy.
[0200] Evaluation of Target Lesions:
[0201] Complete Response (CR): Disappearance of all target lesions; Partial Response (PR): At least a 30% decrease in the sum of the LD (longest diameter) of target lesions, taking as reference the baseline sum LD; Stable Disease (SD): Neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD, taking as reference the smallest sum LD since the treatment started; Progressive Disease (PD): At least a 20% increase in the sum of the LD of target lesions, taking as reference the smallest sum LD recorded since the treatment started or the appearance of one or more new lesions.
[0202] Evaluation of Non-Target Lesions
[0203] Complete Response (CR): Disappearance of all non-target lesions and normalization of tumor marker level; Incomplete Response/Stable Disease (SD): Persistence of one or more non-target lesion(s) or/and maintenance of tumor marker level above the normal limits; Progressive Disease (PD): Appearance of one or more new lesions and/or unequivocal progression of existing non-target lesions.
[0204] Other Response Criteria
[0205] Other response criteria include the Immune-Related Response Criteria or iRECIST, as defined by Wolchok et al., in 2009 (Wolchok J D, et al.; Guidelines for the Evaluation of Immune Therapy Activity in Solid Tumors: Immune-Related Response Criteria. Clin Cancer Res 2009; 15(23):7412-20) and the revised International Working Group Response Criteria (Cheson B D et al., Revised response criteria for malignant lymphoma. J. Clin. Oncol. 2007; 25:579-586).
[0206] The methods of treating cancer include methods for inhibiting cell growth by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein.
[0207] Also provided herein are methods of inhibiting metastasis of a cancer in a patient in need thereby by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. In some embodiments, metastasis is inhibited by at least about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%.
[0208] In another aspect is a method of reducing pre-existing tumor metastasis in a cancer patient in need thereof by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. In some embodiments, pre-existing tumor metastasis is reduced by at least about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%.
[0209] In still another aspect the methods of treating cancer also provide for methods for reducing tumor burden in an individual by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. In some embodiments, tumor burden is reduced by at least about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%.
[0210] In another aspect the methods of treating cancer also provide for methods for reducing tumor burden in an individual by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. In some embodiments, tumor burden is reduced by at least about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%.
[0211] The methods of treating cancer described herein also provide for methods for increasing or otherwise prolonging time to disease progression of certain stages (including advanced stages of cancer such as Stage III and IV cancer described herein). Time to disease progression can be prolonged in a patient by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. In some embodiments, the increase is a comparison between the time to disease progression without treatment and with treatment with a combination described herein. In some embodiments, the methods described herein prolong the time to disease progression by at least 1 week, 2 weeks, 3 weeks, 4 weeks, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 1 year, or more, including values therein.
[0212] The methods of treating cancer described herein also provide for methods for increasing or otherwise prolonging survival (including overall survival) of patients diagnosed with cancer as described herein. Patient survival can be prolonged by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. In some embodiments, the increase is a comparison between the survival without treatment and with treatment with a combination as described herein. In some embodiments, the methods described herein prolong survival by at least 1 week, 2 weeks, 3 weeks, 4 weeks, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 1 year, 2 years, or more, including values therein.
[0213] The methods of treating cancer described herein also provide for methods for increasing progression-free survival of patients diagnosed with cancer as described herein. Patient progression-free survival can be prolonged by administering a therapeutically effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. In some embodiments, the increase is a comparison between the progression-free survival without treatment and with treatment with a combination as described herein. In some embodiments, the methods described herein increase progression-free survival by at least 1 week, 2 weeks, 3 weeks, 4 weeks, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 1 year, 2 years, or more, including values therein.
[0214] Also provided herein are methods of reducing a level of myeloid-derived suppressor cells (MDSC) in a patient in need thereof by administering an effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. The reduction of MDSC can benefit the treatment of a cancer described herein. The level of MDSC in a human patient can be measured before, during, and after administration of a combination described herein. In some embodiments, it can be useful to compare pre- and post-administration amounts of MDSC in the patient. A reduction in the amount, level, or number of MDSC following administration can indicate effectiveness of the combination in, for example, treating a cancer described herein. MD SC levels can be monitored over the course of a treatment or regimen described herein with a combination described herein. In such instances, the determination of MD SC levels at various points during the course of administration can indicate the effectiveness of the regimen.
[0215] Methods of reducing the percentage or level of Treg cells in a patient in need thereof are also provided herein. Such methods include administering an effective amount of a combination described herein where the combination includes a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor described herein. The reduction of Treg cells can benefit the treatment of a cancer described herein. The level of Treg cells in a human patient can be measured before, during, and after administration of a combination described herein. In some embodiments, it can be useful to compare pre- and post-administration amounts of Treg cells in the patient. A reduction in the amount, level, or number of Treg cells following administration can indicate effectiveness of the combination in, for example, treating a cancer described herein. Treg cell levels can be monitored over the course of a treatment or regimen described herein with a combination described herein. In such instances, the determination of Treg cell levels at various points during the course of administration can indicate the effectiveness of the regimen.
[0216] The combinations described herein can be useful in methods of enhancing activity of natural killer (NK) cells. The combinations described herein can also be useful in methods of enhancing activity of cytotoxic T-cells. The methods of enhancing include contacting a NK cell or cytotoxic T-cell with a combination described herein where the combination enhances the activity of the NK cell or cytotoxic T-cell relative to its activity prior to the contact. In some embodiments, the enhanced activity of the NK cell or cytotoxic T-cell is in a cancer patient who has been administered a combination as described herein.
[0217] The combinations described herein can also enhance antibody-dependent cell-mediated cytotoxicity in a cancer patient upon administration of a combination as described herein.
[0218] The combinations described herein can include administration of each therapy (e.g., a compound of formula I and a PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor), where the administration is performed simultaneously or sequentially (in either order). In one embodiment, the compound of formula I and the PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor are administered simultaneously (e.g., within at least 1 to 5 min of each other). In another embodiment, the compound of formula I and the PD-L1 inhibitor and/or PD-1 inhibitor, plus a CTLA-4 inhibitor are administered sequentially (e.g., within at least 10 min, 15 min, 30 min, 1 h, 2 h, 5 h, 10 h, 12 h, 1 day, 2 days, 5 days, 7 days, 14 days, or 21 days of each other).
[0219] The compound of formula I can be administered, for example, once a day (QD), twice daily (BID), once a week (QW), twice weekly (BIW), three times a week (TIW), or monthly (QM) regularly on a continuous base or intermittent base such as BIW for 3 months then resume a month later. For example, the compound of formula I can be administered BID. The compound of formula I can be administered TIW. In certain instances, the compound of formula I is administered 2 to 3 times a week. In another embodiment, the compound of formula I is administered QD. The compound can be administered QD for about: 1 day to about 7 days, 1 day to about 14 days, 1 day to about 21 days, 1 day to about 28 days, or daily until disease progression or unacceptable toxicity. The administration of a compound of formula I can, in part, depend upon the tolerance of the patient where greater tolerance can allow greater or more frequent administration. Alternatively, where a patient shows poor tolerance to a compound of formula I, a less amount of the compound or a less frequent dosing can be performed. Compounds of formula I can be administered in any regimen as described herein.
[0220] For example, a compound of formula I can be administered at an amount of about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg, QD. For example, a compound of formula I can be administered at an amount of about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg, BIW. For example, a compound of formula I can be administered at an amount of about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg, TIW. For example, a compound of formula I can be administered at an amount of about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg, QW. For example, a compound of formula I can be administered at an amount of about: 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 60 mg, 70 mg, 80 mg, 85 mg, 90 mg, 100 mg, 125 mg, 150 mg, 175 mg, or 200 mg, Q2W. For example, a compound of formula I can be administered at an amount of about 5 mg or about 10 mg, QD. For example, a compound of formula I can be administered at an amount of about 5 mg or about 10 mg, BIW. For example, a compound of formula I can be administered at an amount of about 5 mg or about 10 mg, TIW. For example, a compound of formula I can be administered at an amount of about 5 mg or about 10 mg, QW. For example, a compound of formula I can be administered at an amount of about 5 mg or about 10 mg, Q2W. Administration of a compound of formula I can be continuous. Administration of a compound of formula I can be intermittent.
[0221] For example, a compound of formula I can be administered at an amount of about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg, QD. For example, a compound of formula I can be administered at an amount of about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg, BIW. For example, a compound of formula I can be administered at an amount of about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg, TIW. For example, a compound of formula I can be administered at an amount of about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg, QW. For example, a compound of formula I can be administered at an amount of about: 1 mg to about 10 mg, 1 mg to about 25 mg, 1 mg to about 50 mg, 5 mg to about 10 mg, 5 mg to about 25 mg, 5 mg to about 50 mg, 10 mg to about 25 mg, 10 mg to about 50 mg, 50 mg to about 100 mg, or 100 mg to about 200 mg, Q2W. Administration of a compound of formula I can be continuous. Administration of a compound of formula I can be intermittent.
[0222] r example, a compound of formula I can be administered at an amount of about: 0.0001 mg/kg to about 200 mg/kg, 0.001 mg/kg to about 200 mg/kg, 0.01 mg/kg to about 200 mg/kg, 0.01 mg/kg to about 150 mg/kg, 0.01 mg/kg to about 100 mg/kg, 0.01 mg/kg to about 50 mg/kg, 0.01 mg/kg to about 25 mg/kg, 0.01 mg/kg to about 10 mg/kg, or 0.01 mg/kg to about 5 mg/kg, 0.05 mg/kg to about 200 mg/kg, 0.05 mg/kg to about 150 mg/kg, 0.05 mg/kg to about 100 mg/kg, 0.05 mg/kg to about 50 mg/kg, 0.05 mg/kg to about 25 mg/kg, 0.05 mg/kg to about 10 mg/kg, or 0.05 mg/kg to about 5 mg/kg, 0.5 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 150 mg/kg, 0.5 mg/kg to about 100 mg/kg, 0.5 mg/kg to about 50 mg/kg, 0.5 mg/kg to about 25 mg/kg, 0.5 mg/kg to about 10 mg/kg, or 0.5 mg/kg to about 5 mg/kg, QD. For example, a compound of formula I can be administered at an amount of about: 0.0001 mg/kg to about 200 mg/kg, 0.001 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 150 mg/kg, 0.5 mg/kg to about 100 mg/kg, 0.5 mg/kg to about 50 mg/kg, 0.5 mg/kg to about 25 mg/kg, 0.5 mg/kg to about 10 mg/kg, or 0.5 mg/kg to about 5 mg/kg, BIW. For example, a compound of formula I can be administered at an amount of about: 0.0001 mg/kg to about 200 mg/kg, 0.001 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 150 mg/kg, 0.5 mg/kg to about 100 mg/kg, 0.5 mg/kg to about 50 mg/kg, 0.5 mg/kg to about 25 mg/kg, 0.5 mg/kg to about 10 mg/kg, or 0.5 mg/kg to about 5 mg/kg, TIW. For example, a compound of formula I can be administered at an amount of about: 0.0001 mg/kg to about 200 mg/kg, 0.001 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 150 mg/kg, 0.5 mg/kg to about 100 mg/kg, 0.5 mg/kg to about 50 mg/kg, 0.5 mg/kg to about 25 mg/kg, 0.5 mg/kg to about 10 mg/kg, or 0.5 mg/kg to about 5 mg/kg, QW. For example, a compound of formula I can be administered at an amount of about: 0.0001 mg/kg to about 200 mg/kg, 0.001 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 200 mg/kg, 0.5 mg/kg to about 150 mg/kg, 0.5 mg/kg to about 100 mg/kg, 0.5 mg/kg to about 50 mg/kg, 0.5 mg/kg to about 25 mg/kg, 0.5 mg/kg to about 10 mg/kg, or 0.5 mg/kg to about 5 mg/kg, Q2W. In one example, a compound of formula I can be administered at an amount of about 15 mg/kg to about 75 mg/kg, QD. In another example, a compound of formula I can be administered at an amount of about 20 mg/kg to about 50 mg/kg. In still another example, a compound of formula I can be administered at an amount of about 0.001 mg/kg, 0.01 mg/kg, 0.05 mg/kg, 0.1 mg/kg, 0.5 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 10 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg, 40 mg/kg, 50 mg/kg, 60 mg/kg, 70 mg/kg, 80 mg/kg, 90 mg/kg, 100 mg/kg, 125 mg/kg, 150 mg/kg, 175 mg/kg, or 200 mg/kg. Administration of a compound of formula I can be continuous. Administration of a compound of formula I can be intermittent.
[0223] For example, a compound of formula I can be administered at an amount of about: 1 mg/kg to about 200 mg/kg, 1 mg/kg to about 150 mg/kg, 1 mg/kg to about 100 mg/kg, 1 mg/kg to about 50 mg/kg, 1 mg/kg to about 25 mg/kg, 1 mg/kg to about 10 mg/kg, or 1 mg/kg to about 5 mg/kg, QD. For example, a compound of formula I can be administered at an amount of about: 1 mg/kg to about 200 mg/kg, 1 mg/kg to about 150 mg/kg, 1 mg/kg to about 100 mg/kg, 1 mg/kg to about 50 mg/kg, 1 mg/kg to about 25 mg/kg, 1 mg/kg to about 10 mg/kg, or 1 mg/kg to about 5 mg/kg, BIW. For example, a compound of formula I can be administered at an amount of about: 1 mg/kg to about 200 mg/kg, 1 mg/kg to about 150 mg/kg, 1 mg/kg to about 100 mg/kg, 1 mg/kg to about 50 mg/kg, 1 mg/kg to about 25 mg/kg, 1 mg/kg to about 10 mg/kg, or 1 mg/kg to about 5 mg/kg, TIW. For example, a compound of formula I can be administered at an amount of about: 1 mg/kg to about 200 mg/kg, 1 mg/kg to about 150 mg/kg, 1 mg/kg to about 100 mg/kg, 1 mg/kg to about 50 mg/kg, 1 mg/kg to about 25 mg/kg, 1 mg/kg to about 10 mg/kg, or 1 mg/kg to about 5 mg/kg, QW. For example, a compound of formula I can be administered at an amount of about: 1 mg/kg to about 200 mg/kg, 1 mg/kg to about 150 mg/kg, 1 mg/kg to about 100 mg/kg, 1 mg/kg to about 50 mg/kg, 1 mg/kg to about 25 mg/kg, 1 mg/kg to about 10 mg/kg, or 1 mg/kg to about 5 mg/kg, Q2W. In one example, a compound of formula I can be administered at an amount of about 15 mg/kg to about 75 mg/kg, QD. In another example, a compound of formula I can be administered at an amount of about 20 mg/kg to about 50 mg/kg. In still another example, a compound of formula I can be administered at an amount of about 0.001 mg/kg, 0.01 mg/kg, 0.05 mg/kg, 0.1 mg/kg, 0.5 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 10 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg, 40 mg/kg, 50 mg/kg, 60 mg/kg, 70 mg/kg, 80 mg/kg, 90 mg/kg, 100 mg/kg, 125 mg/kg, 150 mg/kg, 175 mg/kg, or 200 mg/kg. Administration of a compound of formula I can be continuous. Administration of a compound of formula I can be intermittent.
[0224] As used herein, the term daily is intended to mean that a therapeutic compound of a combination described herein, such as a compound of formula I, is administered once or more than once each day for a period of time. The term continuous is intended to mean that a therapeutic compound of a combination described herein, such as a compound of formula I, is administered daily for an uninterrupted period of at least 10 days to 52 weeks. The term intermittent or intermittently as used herein is intended to mean stopping and starting at either regular or irregular intervals. For example, intermittent administration of a therapeutic compound of a combination described herein, such as a compound of formula I, includes administration for one to six days per week (e.g., 2 to 3 times per week or QD), administration in cycles (e.g., daily administration for two to eight consecutive weeks, then a rest period with no administration at least one day), or, for example, administration on alternate days.
[0225] Where the inhibitor is an Inhibitor Antibody, it can be administered according to established regimens such as those provided in a package insert. The Inhibitor Antibody can be administered in an amount described herein and can be administered QW, once every 2 weeks (Q2W), once every 3 weeks (Q3W), or once every 4 weeks (Q4W). In one embodiment, the Inhibitor Antibody is administered Q2W or Q4W. In another embodiment, the Inhibitor Antibody is administered Q2W. In yet another embodiment, the Inhibitor Antibody is administered Q3W. In still another embodiment, the Inhibitor Antibody is administered BIW for at least 3 weeks. In still another embodiment, the Inhibitor Antibody is administered Q4W.
[0226] For example, the Inhibitor Antibody can be administered at an amount of about 0.1 mg/kg to about 30 mg/kg (including for example 0.1 mg/kg, 0.3 mg/kg, 0.5 mg/kg, 0.7 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 6 mg/kg, 7 mg/kg, 8 mg/kg, 10 mg/kg, 12 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg), QW. For example, the Inhibitor Antibody can be administered at an amount of about 0.1 mg/kg to about 30 mg/kg (including for example 0.1 mg/kg, 0.3 mg/kg, 0.5 mg/kg, 0.7 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 6 mg/kg, 7 mg/kg, 8 mg/kg, 10 mg/kg, 12 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg), Q2W. For example, the Inhibitor Antibody can be administered at an amount of about 0.1 mg/kg to about 30 mg/kg (including for example 0.1 mg/kg, 0.3 mg/kg, 0.5 mg/kg, 0.7 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 6 mg/kg, 7 mg/kg, 8 mg/kg, 10 mg/kg, 12 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg), Q4W. For example, the Inhibitor Antibody can be administered at an amount of about 0.1 mg/kg to about 30 mg/kg (including for example 0.1 mg/kg, 0.3 mg/kg, 0.5 mg/kg, 0.7 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 6 mg/kg, 7 mg/kg, 8 mg/kg, 10 mg/kg, 12 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg), B4W (twice every 4 weeks). For example, the Inhibitor Antibody can be administered at an amount of about 0.1 mg/kg to about 30 mg/kg (including for example 0.1 mg/kg, 0.3 mg/kg, 0.5 mg/kg, 0.7 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, 4 mg/kg, 5 mg/kg, 6 mg/kg, 7 mg/kg, 8 mg/kg, 10 mg/kg, 12 mg/kg, 15 mg/kg, 20 mg/kg, 25 mg/kg, 30 mg/kg), Q3W. For example, the Inhibitor Antibody can be administered at an amount of about 1000 mg to about 2000 mg (including for example 1000 mg, 1100 mg, 1200 mg, 1300 mg, 1400 mg, 1500 mg, 1600 mg), Q2W. For example, the Inhibitor Antibody can be administered at an amount of about 1000 mg to about 2000 mg (including for example 1000 mg, 1100 mg, 1200 mg, 1300 mg, 1400 mg, 1500 mg, 1600 mg), Q3W. For example, the Inhibitor Antibody can be administered at an amount of about 1000 mg to about 2000 mg (including for example 1000 mg, 1100 mg, 1200 mg, 1300 mg, 1400 mg, 1500 mg, 1600 mg), Q4W. Administration of the Inhibitor Antibody can be continuous. Administration of the Inhibitor Antibody can be intermittent.
[0227] The Inhibitor Antibody can be administered as an intravenous infusion over about 10, 20, 30, 40, 50, or 60 or more minutes. the Inhibitor Antibody can be administered as an intravenous infusion over about 60 minutes once every 1, 2, 3, 4, 5 or more weeks. the Inhibitor Antibody can be administered as an intravenous infusion over about 60 minutes once every two weeks. the Inhibitor Antibody can be administered as an intravenous infusion over about 60 minutes once every three weeks. the Inhibitor Antibody can be administered as an intravenous infusion over about 60 minutes once every four weeks. the Inhibitor Antibody can be administered as an intravenous infusion according to a package insert. Administration of Inhibitor Antibody can be continuous. Administration of Inhibitor Antibody can be intermittent.
[0228] The combinations described herein can be administered in a regimen. The regimen can be structured to provide therapeutically effective amounts of a compound of formula I and an inhibitor, such as an Inhibitor Antibody, over a predetermined period of time (e.g., an administration time). The regimen can be structured to limit or prevent side-effects or undesired complications of each of the components of the combination described herein. The regimen can be structured in a manner that results in increased effect for both therapies of the combination (e.g., synergy). Regimens useful for treating cancer can include any number of days of administration which can be repeated as necessary. Administration periods can be broken by a rest period that includes no administration of at least one therapy. For example, a regimen can include administration periods that include 2, 3, 5, 7, 10, 15, 21, 28, or more days. These periods can be repeated. For example, a regimen can include a set number of days as previously described where the regimen is repeated 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, or more times.
[0229] Regimens can include a rest period of at least 1, 2, 3, 5, 7, 10, or more days, where at least one therapy is no longer administered to a patient. The rest period can be determined by, for example, monitoring the reaction of the patient to the drug or by measuring the efficacy of the treatment. A rest period can be applicable to a single therapy, such that only one therapy of a combination described herein is discontinued in the rest period but the other therapy(ies) are still administered. Rest periods can be applied to all of the therapies administered to the subject such that the subject receives no therapy for a set period of time during the rest period.
[0230] Regimens described herein for the treatment of cancer using the combinations described herein can be continued until disease progression or unacceptable toxicity.
[0231] Regimens for administration of combinations described herein include, for example administration of a compound of formula I BIW or TIW and administration of a PD-L1 and/or PD-1 inhibitor, plus CTLA-4 inhibitor. For example, a compound of formula I can be administered QD for about 21 days and an Inhibitor Antibody described herein can be administered Q2W or Q4W). For example, a compound of formula I can be administered BIW or TIW and an Inhibitor Antibody described herein can be administered Q2W. In another exemplary regimen, a compound of formula I can be administered BIW or TIW and an Inhibitor Antibody can be administered BIW for 2 or 3 weeks. In still another exemplary regimen, a compound of formula I can be administered BIW or TIW and an Inhibitor Antibody can be administered Q4W. In still another exemplary regimen, a compound of formula I can be administered BIW and an inhibitor described herein can be administered Q2W, Q3W, or Q4W. In certain instances, such regimens include administration of an Inhibitor Antibody administered Q2W, Q3W, or Q4W. In yet another exemplary regimen, a compound of formula I can be administered TIW and an inhibitor described herein can be administered Q2W, Q3W, or Q4W. In certain instances, such regimens include administration of an Inhibitor Antibody administered Q2W, Q3W, or Q4W. In certain instances, such regimens include administration of a compound of formula I administered QD. In certain instances, such regimens include administration of a compound of formula I administered QD for at least 21 days. In yet another exemplary regimen, a compound of formula I can be administered QD or QW and an inhibitor (e.g., an Inhibitor Antibody) is administered Q2W, Q3W, or Q4W.
[0232] The regimen can be a regimen for administration of an Inhibitor Antibody with a compound of formula I as described herein. In one exemplary regimen including an Inhibitor Antibody, a compound of formula I can be administered BIW or TIW and an Inhibitor Antibody is administered in accordance with the prescribing information provided in, for example, a package insert. In another exemplary regimen, an Inhibitor Antibody is administered at an amount of about 1 mg/kg to about 20 mg/kg on day 1 of the regimen, and Q2W thereafter until disease progression or unacceptable toxicity and a compound of formula I is administered BIW or TIW over the same period of time. In another exemplary regimen, an Inhibitor Antibody is administered at an amount of about 1 mg/kg to about 20 mg/kg on day 1 of a regimen, and Q3W thereafter until disease progression or unacceptable toxicity and a compound of formula I is administered BIW or TIW over the same period of time. an Inhibitor Antibody can be administered Q4W with a compound of formula I, where the compound of formula I is administered, for example, BIW or TIW during the course of such a regimen. an Inhibitor Antibody can be administered Q2W with a compound of formula I, where the compound of formula I is administered, for example, BIW or TIW during the course of such a regimen. In still another exemplary regimen, an Inhibitor Antibody can be administered Q2W or Q4W with a compound of formula I, where the compound of formula I is administered, for example, QD or QW during the course of such a regimen. Such regimens can be repeated as described above (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or more times).
[0233] In another exemplary regimen including an Inhibitor Antibody, a compound of formula I can be administered QD and an Inhibitor Antibody is administered in accordance with the prescribing information provided in, for example, a package insert. In another exemplary regimen, an Inhibitor Antibody is administered at an amount of about 1 mg/kg to about 20 mg/kg on day 1 of the regimen, and Q2W thereafter until disease progression or unacceptable toxicity and a compound of formula I is administered QD over the same period of time. In another exemplary regimen, an Inhibitor Antibody is administered at an amount of about 1 mg/kg to about 20 mg/kg on day 1 of a regimen, and Q3W thereafter until disease progression or unacceptable toxicity and a compound of formula I is administered QD over the same period of time. an Inhibitor Antibody can be administered Q4W with a compound of formula I, where the compound of formula I is administered QD during the course of such a regimen. an Inhibitor Antibody can be administered Q2W with a compound of formula I, where the compound of formula I is administered QD during the course of such a regimen. Such regimens can be repeated as described above (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or more times).
[0234] It should also be appreciated that the combinations described herein for treating cancer can be coadministered with other active agents other than those present in the combinations described herein (e.g., anti-cancer agents). Regimens for administration of a combination described herein, including the exemplary regimens set forth above, can be modified as necessary to include administration of such active agents. Administration of such active agents, e.g., anti-cancer agents, can be performed QD, QW, QM, BID, BIW, TIW, Q2W, Q3W, or Q4W, or in accordance with prescribing information for such anti-cancer agents as set forth, for example, in a package insert. Exemplary anti-cancer agents include but are not limited to: ABRAXANE; abiraterone; ace-11; aclarubicin; acivicin; acodazole hydrochloride; acronine; actinomycin; acylfulvene; adecypenol; adozelesin; adriamycin; aldesleukin; all trans-retinoic acid (ATRA); altretamine; ambamustine; ambomycin; ametantrone acetate; amidox; amifostine; aminoglutethimide; aminolevulinic acid; amrubicin; amsacrine; anagrelide; anastrozole; andrographolide; antarelix; anthramycin; aphidicolin glycinate; apurinic acid; ara-CDP-DL-PTBA; arginine deaminase; ARRY-162; ARRY-300; ARRY-142266; AS703026; asparaginase; asperlin; asulacrine; atamestane; atrimustine; axinastatin 1; axinastatin 2; axinastatin 3; azasetron; azatoxin; azatyrosine; azacitidine; AZD8330; azetepa; azotomycin; balanol; batimastat; BAY 11-7082; BAY 43-9006; BAY 869766; bendamustine; benzochlorins; benzodepa; benzoylstaurosporine; beta-alethine; betaclamycin B; betulinic acid; b-FGF inhibitor; bicalutamide; bisantrene; bisaziridinylspermine; bisnafide; bisnafide dimesylate; bistratene A; bisantrene hydrochloride; bleomycin; bleomycin sulfate; busulfan; bizelesin; breflate; bortezomib; brequinar sodium; bropirimine; budotitane; buthionine sulfoximine; bryostatin; cactinomycin; calusterone; calcipotriol; calphostin C; camptothecin derivatives; capecitabine; carboxamide-amino-triazole; carboxyamidotriazole; CaRest M3; CARN 700; caracemide; carbetimer; carboplatin; carmustine; carubicin hydrochloride; carzelesin; castanospermine; cecropin B; cedefingol; celecoxib; cetrorelix; chlorins; chloroquinoxaline sulfonamide; cicaprost; chlorambucil; Chlorofusin; cirolemycin; cisplatin; CI-1040; cis-porphyrin; cladribine; clomifene analogues; clotrimazole; collismycin A; collismycin B; combretastatin A4; combretastatin analogue; conagenin; crambescidin 816; crisnatol; crisnatol mesylate; cryptophycin 8; cryptophycin A derivatives; curacin A; cyclopentanthraquinones; cycloplatam; cypemycin; cyclophosphamide; cytarabine; cytarabine ocfosfate; cytolytic factor; cytostatin; dacarbazine; dactinomycin; daunorubicin; daunorubicin hydrochloride; decarbazine; dacliximab; dasatinib; decitabine; dehydrodidemnin B; deslorelin; dexamethasone; dexifosfamide; dexrazoxane; dexverapamil; dexormaplatin; dezaguanine; dezaguanine mesylate; diaziquone; didemnin B; didox; diethylnorspermine; dihydro 5 azacytidine; dihydrotaxol; 9-dioxamycin; diphenyl spiromustine; docosanol; dolasetron; docetaxel; doxorubicin; doxorubicin hydrochloride; doxifluridine; droloxifene; droloxifene citrate; dromostanolone propionate; dronabinol; duazomycin; duocarmycin SA; ebselen; ecomustine; edelfosine; edrecolomab; edatrexate; eflornithine hydrochloride; eflornithine; elemene; emitefur; elsamitrucin; enloplatin; enpromate; epipropidine; epirubicin; epirubicin hydrochloride; epristeride; erbulozole; eribulin; esorubicin hydrochloride; estramustine; estramustine phosphate sodium; etanidazole; etoposide; etoposide phosphate; etoprine; exemestane; fadrozole; fadrozole hydrochloride; fazarabine; fenretinide; filgrastim; finasteride; flavopiridol; flezelastine; fluasterone; floxuridine; fludarabine phosphate; fludarabine; fluorodaunorubicin hydrochloride; forfenimex; formestane; fluorouracil; floxouridine; flurocitabine; fosquidone; fostriecin sodium; fostriecin; fotemustine; gadolinium texaphyrin; gallium nitrate; galocitabine; ganirelix; gelatinase inhibitors; gemcitabine; geldanamycin; gossyphol; GDC-0973; GSK1120212/trametinib; herceptin; hydroxyurea; hepsulfam; heregulin; hexamethylene bisacetamide; hypericin; ibandronic acid; ibrutinib; idarubicin; idarubicin hydrochloride; ifosfamide; canfosfamide; ilmofo sine; iproplatin; idoxifene; idramantone; ilmofo sine; ilomastat; imidazoacridones; imatinib (e.g., GLEEVEC); imiquimod; iobenguane; iododoxorubicin; ipomeanol; irinotecan; irinotecan hydrochloride; irsogladine; isobengazole; isohomohalicondrin B; itasetron; iimofosine; interleukin Il (including recombinant interleukin IL-2; or r1L.sub.2); interferon alfa-2a; interferon alfa-2b; interferon alfa-n1; interferon alfa-n3; interferon beta-la; interferon gamma-1b; jasplakinolide; kahalalide F; lamellarin N triacetate; lanreotide; leinamycin; lenograstim; lentinan sulfate; leptolstatin; letrozole; leuprorelin; levamisole; liarozole; lissoclinamide 7; lobaplatin; lombricine; lometrexol; lonidamine; losoxantrone; lovastatin; loxoribine; lurtotecan; lutetium texaphyrin; lysofylline; lanreotide acetate; lapatinib; letrozole; leucovorin; leuprolide acetate; liarozole hydrochloride; lometrexol sodium; lomustine; lenalidomide; lenvatinib; losoxantrone hydrochloride; LY294002; pomalidomide; maitansine; mannostatin A; marimastat; masoprocol; maspin; matrilysin inhibitors; menogaril; merbarone; meterelin; methioninase; metoclopramide; MIF inhibitor; mifepristone; miltefosine; mirimostim; mitoguazone; mitolactol; mitonafide; mitoxantrone; mofarotene; molgramostim; mopidamol; mycaperoxide B; myriaporone; maytansine; mechlorethamine hydrochloride; megestrol acetate; melengestrol acetate; melphalan; mercaptopurine; methotrexate; methotrexate sodium; metoprine; meturedepa; mitindomide; mitocarcin; mitocromin; mitogillin; mitomalcin; mitomycin; mitosper; mitotane; mitoxantrone hydrochloride; mycophenolic acid; nafarelin; nagrestip; napavin; naphterpin; nartograstim; nedaplatin; nemorubicin; neridronic acid; nilutamide; nisamycin; nitric oxide modulators; nitroxide antioxidant; nitrullyn; nocodazole; nogalamycin; oblimersen (GENASENSE); octreotide; okicenone; oligonucleotides; onapristone; ondansetron; ondansetron; oracin; oral cytokine inducer; ormaplatin; oxisuran; oxaloplatin; osaterone; oxaliplatin; oxaunomycin; palauamine; palmitoylrhizoxin; pamidronic acid; panaxytriol; panomifene; parabactin; pazelliptine; pegaspargase; peldesine; pentosan polysulfate sodium; pentostatin; pentrozole; perflubron; perfosfamide; perillyl alcohol; phenazinomycin; phenylacetate; phosphatase inhibitors; picibanil; pilocarpine hydrochloride; pirarubicin; piritrexim; placetin A; placetin B; porfiromycin; prednisone; prostaglandin J2; pyrazoloacridine; paclitaxel; PD035901; PD184352; PD318026; PD98059; peliomycin; pentamustine; peplomycin sulfate; PKC412; pipobroman; piposulfan; piroxantrone hydrochloride; plicamycin; plomestane; podophyllotoxin; polyphenol E; porfimer sodium; porfiromycin; prednimustine; procarbazine; procarbazine hydrochloride; puromycin; puromycin hydrochloride; pyrazofurin; raltitrexed; ramosetron; retelliptine demethylated; rhizoxin; rituximab; RII retinamide; rogletimide; rohitukine; romurtide; roquinimex; rubiginone B 1; ruboxyl; riboprine; romidepsin; safingol; safingol hydrochloride; saintopin; sarcophytol A; sargramostim; semustine; sizofiran; sobuzoxane; sodium borocaptate; sodium phenylacetate; solverol; sonermin; sorafenib; sunitinib; sparfosic acid; spicamycin D; spiromustine; splenopentin; spongistatin 1; Spongistatin 2; Spongistatin 3; Spongistatin 4; Spongistatin 5; Spongistatin 6; Spongistatin 7; Spongistatin 8; and Spongistatin 9; squalamine; stipiamide; stromelysin inhibitors; sulfinosine; suradista; suramin; swainsonine; SB239063; selumetinib/AZD6244; simtrazene; SP600125; sparfosate sodium; sparsomycin; spirogermanium hydrochloride; spiroplatin; streptonigrin; streptozocin; sulofenur; tallimustine; tamoxifen methiodide; tauromustine; tazarotene; tecogalan sodium; tegafur; tellurapyrylium; temoporfin; temozolomide; teniposide; tetrachlorodecaoxide; tetrazomine; thaliblastine; thiocoraline; thrombopoietin; thymalfasin; thymopoietin receptor agonist; thymotrinan; tirapazamine; titanocene bichloride; topsentin; toremifene; tretinoin; triacetyluridine; triciribine; trimetrexate; triptorelin; tropisetron; turosteride; tyrphostins; talisomycin; TAK-733; taxotere; tegafur; teloxantrone hydrochloride; teroxirone; testolactone; thiamiprine; thioguanine; thiotepa; tiazofurin; tirapazamine; toremifene citrate; trastuzumab; trestolone acetate; triciribine phosphate; trimetrexate; trimetrexate glucuronate; triptorelin; tubulozole hydrochloride; tumor necrosis factor-related apoptosis-inducing ligand (TRAIL); UBC inhibitors; ubenimex; U0126; uracil mustard; uredepa; vapreotide; variolin B; velaresol; veramine; verteporfin; vinorelbine; vinxaltine; vitaxin; vinblastine; vinblastine sulfate; vincristine sulfate; vindesine; vindesine sulfate; vinepidine sulfate; vinglycinate sulfate; vinleurosine sulfate; vinorelbine tartrate; vinrosidine sulfate; vinzolidine sulfate; vorozole; wortmannin; XL518; zanoterone; zeniplatin; zilascorb; zinostatin stimalamer; zinostatin; and zorubicin hydrochloride.
[0235] Other exemplary anti-cancer agents include Erbulozole (e.g., R-55104); Dolastatin 10 (e.g., DLS-10 and NSC-376128); Mivobulin isethionate (e.g., CI-980); NSC-639829; Discodermolide (e.g., NVP-XX-A-296); ABT-751 (Abbott; e.g., E-7010); Altorhyrtin A; Altorhyrtin C); Cemadotin hydrochloride (e.g., LU-103793 and NSC-D-669356); Epothilone A; Epothilone B; Epothilone C; Epothilone D; Epothilone E; Epothilone F; Epothilone B N-oxide; Epothilone A N-oxide; 16-aza-epothilone B; 21-aminoepothilone B; 21-hydroxyepothilone D; 26-fluoroepothilone; Auristatin PE (e.g., NSC-654663); Soblidotin (e.g., TZT-1027); LS-4559-P (Pharmacia; e.g., LS-4577); LS-4578 (Pharmacia; e.g., LS-477-P); LS-4477 (Pharmacia); LS-4559 (Pharmacia); RPR-112378 (Aventis); DZ-3358 (Daiichi); FR-182877 (Fujisawa; e.g., WS-9265B); GS-164 (Takeda); GS-198 (Takeda); KAR-2 (Hungarian Academy of Sciences); B SF-223651 (BASF; e.g., ILX-651 and LU-223651); SAH-49960 (Lilly/Novartis); SDZ-268970 (Lilly/Novartis); AM-97 (Armad/Kyowa Hakko); AM-132 (Armad); AM-138 (Armad/Kyowa Hakko); IDN-5005 (Indena); Cryptophycin 52 (e.g., LY-355703); AC-7739 (Ajinomoto; e.g., AVE-8063A and CS-39.HC1); AC-7700 (Ajinomoto; e.g., AVE-8062; AVE-8062A; CS-39-L-Ser.HC1; and RPR-258062A); Vitilevuamide; Tubulysin A; Canadensol; CA-170 (Curis, Inc.); Centaureidin (e.g., NSC-106969); T-138067 (Tularik; e.g., T-67; TL-138067 and TI-138067); COBRA-1 (Parker Hughes Institute; e.g., DDE-261 and WHI-261); H10 (Kansas State University); H16 (Kansas State University); Oncocidin A1 (e.g., BTO-956 and DIME); DDE-313 (Parker Hughes Institute); Fijianolide B; Laulimalide; SPA-2 (Parker Hughes Institute); SPA-1 (Parker Hughes Institute; e.g., SPIKET-P); 3-IAABU (Cytoskeleton/Mt. Sinai School of Medicine; e.g., MF-569); Narcosine (e.g., NSC-5366); Nascapine; D-24851 (Asta medica); A-105972 (Abbott); Hemiasterlin; 3-BAABU (Cytoskeleton/Mt. Sinai School of Medicine; e.g., MF-191); TMPN (Arizona State University); Vanadocene acetylacetonate; T-138026 (Tularik); Monsatrol; lnanocine (e.g., NSC-698666); 3-IAABE (Cytoskeleton/Mt. Sinai School of Medicine); A-204197 (Abbott); T-607 (Tuiarik; e.g., T-900607); RPR-115781 (Aventis); Eleutherobins (e.g., Desmethyleleutherobin; Desaetyleleutherobin; lsoeleutherobin A; and Z-Eleutherobin); Caribaeoside; Caribaeolin; Halichondrin B; D-64131 (Asta medica); D-68144 (Asta medica); Diazonamide A; A-293620 (Abbott); NPI-2350 (Nereus); Taccalonolide A; TUB-245 (Aventis); A-259754 (Abbott); Diozostatin; (-)-Phenylahistin (e.g., NSCL-96F037); D-62638 (Asta medica); D-62636 (Asta medica); Myoseverin B; D-43411 (Zentaris; e.g., D-81862); A-289099 (Abbott); A-318315 (Abbott); HTI-286 (e.g., SPA-110; trifluoroacetate salt) (Wyeth); D-82317 (Zentaris); D-82318 (Zentaris); SC-12983 (NCI); Resverastatin phosphate sodium; BPR-OY-007 (National Health Research Institutes); and SSR-250411 (Sanofi)); goserelin; leuprolide; triptolide; homoharringtonine; topotecan; itraconazole; deoxyadenosine; sertraline; pitavastatin; clofazimine; 5-nonyloxytryptamine; vemurafenib; dabrafenib; gefitinib (IRESSA); erlotinib (TARCEVA); cetuximab (ERBITUX); lapatinib (TYKERB); panitumumab (VECTIBIX); vandetanib (CAPRELSA); afatinib/BIBW2992; CI-1033/canertinib; neratinib/HKI-272; CP-724714; TAK-285; AST-1306; ARRY334543; ARRY-380; AG-1478; dacomitinib/PF299804; OSI-420/desmethyl erlotinib; AZD8931; AEE726; pelitinib/EKB-569; CUDC-101; WZ8040; WZ4002; WZ3146; AG-490; XL647; PD153035; 5-azathioprine; 5-aza-2'-deoxycytidine; 17-N-Allylamino-17-Demethoxygeldanamycin (17-AAG); 20-epi-1,25 dihydroxyvitamin D3; 5 ethynyluracil; and BMS-599626.
[0236] In certain embodiments, the combinations described herein are coadministered with an anti-cancer agent described above, where the anti-cancer agent has known activity against a particular cancer (e.g., gemcitibine coadministered with a combination described herein for treating pancreatic cancer). The anti-cancer agents above can be approved for use in treating certain indications (e.g., certain cancers) at concentrations, amounts, and using treatment regimens known in the art.
[0237] It is understood that modifications which do not substantially affect the activity of the various embodiments of this invention are also included within the definition of the invention provided herein.
HBI-8000 as an Epigenetic Modifier
[0238] Treatment with immune checkpoint inhibitors (ICIs) targeting cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and the programmed cell death receptor/ligand-1 (PD-1)/(PD-L1) axis is effective against many cancer types. Not all patients experience a durable response to ICIs, however, due in part to unresponsiveness or acquired resistance. Epigenetic changes within the tumor microenvironment may alter the response to immunotherapy. In combination with ICIs, class I-selective HDAC inhibitors reinvigorate immune responses. We used HBI-8000 as an epigenetic immunomodulator to reprogram the tumor microenvironment from immunologically cold (nonresponsive) to hot (responsive). We tested this in preclinical syngeneic mouse tumor immunotherapy models.
[0239] Syngeneic tumors were created in 8-week-old female mice using the following cell lines: MC38 and CT26 murine colon carcinoma, RENCA renal adenocarcinoma, A20 B-cell lymphoma, and 4T1 mammary carcinoma cells (MC38 in C57BL/6; CT26, A20, and 4T1 in BALB/c). Tumors were grown to .about.100 mm.sup.3 prior to initiating treatment. Mice were treated daily with HBI-8000 (orally), alone or in combination with PD-1, PD-1L, or CTLA-4 antibodies (intraperitoneally). The NanoString nCounter PanCancer Immune Profiling Panel was used to evaluate the expression of immune response-related genes in MC38 tumors treated with HBI-8000 alone or in combination with ICIs at several time-points.
[0240] Compared with single-agent ICI therapy, HBI-8000 augmented the activity of ICI antibodies targeting either PD-1, the PD-1 ligand, or CTLA-4, and significantly increased tumor regression (p<0.05) in several preclinical models. Gene expression analysis of the treated MC38 tumors revealed significant changes in mRNA expression of immune checkpoints, with enhanced dendritic cell and antigen-presenting cell functions, improved innate and adaptive immunity scores, and modulation of several important immune response-relevant genes and major histocompatibility class I and II molecules.
[0241] These findings show that HBI-8000 mediates epigenetic modifications in the tumor microenvironment, leading to improved efficacy of ICIs.
[0242] The MC38 and CT26 syngeneic murine colon carcinomas, RENCA renal adenocarcinoma, and 4T1 mammary carcinoma cells were obtained from ATCC (Manassas, Va.), and the A20 cells were obtained from Covance (Princeton, N.J.). Cells were passaged and maintained using the protocols provided by the vendors. HBI-8000 was supplied by HUYA Bioscience International. HBI-8000 (HUYA Bioscience International) was formulated in 10% hydroxypropyl-.beta.-cyclodextrin and 10% propylene glycol in deionized water, pH 2.5. Dosing solutions were prepared fresh weekly and stored at 4.degree. C. Animals were dosed orally daily with 50 mg/kg HBI-8000 for 21 days.
[0243] Monoclonal antibodies (mAbs) to mouse PD-1 (clone RPM-14), PDL-1 (clone (10F.9G2), and CTLA-4 (clone 9H10) were purchased from Bio-X-Cell (West Lebanon, N.H.). Antibody dosing solutions were prepared in sterile phosphate-buffered saline on each dosing day, and stored at 4.degree. C. Mice were intraperitoneally injected with the PD-1 antibody (Ab) or PD-L1 Ab (10 mg/kg) twice weekly for 3 weeks. CTLA-4 Ab (2.5 mg/kg) was administered intraperitoneally on days 1, 4, and 7.
[0244] All animal research studies were approved and overseen by the Institutional Animal Care and Use Committees of Charles River (MC38, CT26, 4T1) and Champions Oncology (RENCA). All mice obtained from Charles River (Morrisville, N.C.) were female and 8 weeks old when the tumors were implanted. For MC38 tumors, C57BL/6 mice were implanted subcutaneously in the right flank with 1.times.106 MC38 cells (0.1-mL cell suspension). For CT26 tumors, BALB/c mice were injected subcutaneously in the right flank with 3.times.105 CT26 tumor cells (0.1-mL cell suspension). For A20, BALB/c mice were implanted subcutaneously in the right flank with 1.times.106 A20 cells (0.1-mL cell suspension). For 4T1, BALB/c mice were implanted orthotopically in the mammary fat pad with 1.times.106 4T1 cells (0.1-mL cell suspension). Tumor growth was monitored until reaching an average volume of 100 mm3, at which time the mice were randomized into the various treatment groups (day 0). Treatments were initiated on day 1. Tumor volume was calculated using caliper measurements according to the following formula: Tumor volume (mm{circumflex over ( )}3)=(w{circumflex over ( )}2.times.1)/2, where w=width and l=length (in mm) of the tumor.
[0245] To establish a model of PD-1 antibody failure or stable disease, 150 mice were initially treated biweekly for 3 weeks with first-line anti-PD-1 Ab (5 mg/kg, intraperitoneal administration). Mice bearing tumors that exhibited either slow progression or stable disease (slow progression was defined as; stable disease was defined as 3 consecutive measurements with no significant change in tumor volume) were subsequently re-enrolled into second-line therapy groups (n=10/group) including Vehicle, HBI-8000, PD-1 Ab, PD-1 Ab plus HBI-8000, PD-L1 Ab, and PD-L1 Ab plus HBI-8000.
[0246] NanoString nCounter PanCancer Immune Profiling Panel Gene Expression Studies
[0247] Gene expression studies were carried out using excised MC38 tumors (n=20 animals/treatment) isolated from syngeneic C57BL/6 mice treated for 7, 14, or 17 days with HBI-8000 (50 mg/kg, daily), anti-PD-1 (10 mg/kg, biweekly), or the combination of HBI-8000+anti-PD-1 (50 mg/kg, daily, 10 mg/kg, biweekly). At study termination, tumor samples from the treated mice were collected and fixed in formalin for 24 h and transferred to EtOH, followed by the preparation of formalin-fixed paraffin embedded blocks. Tumor sections (5-10 .mu.m) were prepared from the formalin-fixed paraffin-embedded blocks, and total RNA was isolated from tissue scraped from 4 to 6 slides using the protocol recommended by NanoString Technologies (Seattle, Wash.). The nCounter PanCancer Immune Profiling panel developed and provided by NanoString Technologies was initially selected for expression analyses with an additional 20 genes added as a Panel Plus Codeset. The additional genes were predicted to be regulated by HBI-8000+/-ICI treatment. The nCounter assays were performed according to the manufacturer's instructions using the nCounter FLEX system.
[0248] Gene expression data were analyzed using nSolver software provided by NanoString Technologies, Inc. Raw data were normalized to the geometric mean values of the internal synthetic positive controls and geometric means of the housekeeping genes, as recommended by the manufacturer. The NanoString Technologies' nSolver Analysis Software 4.0 generated cell type scores, pathway scores, heatmaps, and individual gene normalized data from the nCounter PanCancer Immune Profiling Panel Plus dataset. The cell type score quantifies cell populations using marker genes for given cell types; by centering the mean at 0 for each cell type, immune cell type abundance can be compared on the same scale. The same method was used to generate immune-relevant pathway scores; summarizing the data from multiple genes in a pathway into a single score allowed for comparison between treatments for pathway analysis.
[0249] Normalized gene expression data for individual genes was exported from nSolver, annotated with percent of tumor growth inhibition (% TGI), and then imported into GraphPad Prism 7.04. The % TGI was used to group animals into 3 categories, as follows: nonresponders (TGI<25%), partial responders (TGI 25-75%), and responders (TGI>75%). Gene expression data for each mouse was color-coded (TGI<25%, TGI 25-75%, TGI>75%) to track gene expression with tumor response and used to determine if changes in gene expression correlated with the tumor response.
[0250] Differences in tumor size among groups were assessed using 2-tailed statistical analyses. The statistical tests were conducted using Prism ver. 7.04 (GraphPad, San Diego, Calif.). The results are reported as nonsignificant (ns) at P>0.05, significant (*) at 0.01.ltoreq.P<0.05, very significant (**) at 0.001.ltoreq.P<0.01, and extremely significant (***) at P<0.001.
[0251] Combining HBI-8000 with antagonist mAbs to mouse PD-1, PD-L1, and CTLA-4 enhances the antitumor responses and leads to tumor regression.
[0252] To test whether HBI-8000 augments the antitumor effects of inhibiting the PD-(L)1 immune checkpoint axis, we treated mice bearing MC38 syngeneic tumors with HBI-8000, a mouse PD-1 Ab, or HBI-8000 plus PD-1 Ab (FIG. 8A, B). FIG. 8B shows tumor growth in individual mice. In addition, we treated MC38 tumor-bearing mice with HBI-8000, a mouse PD-L1 Ab, or HBI-8000 plus PD-L1 Ab (FIG. 8C, D). Treatment with a single agent (HBI-8000, PD-1 Ab, or PD-L1 Ab) did not significantly affect tumor growth or survival (data not shown). Tumor regression (i.e., absence of detectable tumor) was not seen in any of the single agent cohorts, and all tumors continued to grow throughout the study. In contrast, combining either the PD-1 Ab or the PD-L1 Ab with HBI-8000 produced a statistically significant and reproducibly synergistic decrease or delay in tumor growth and progression (FIG. 8A, C). To corroborate these results, we extended our investigations to 3 other syngeneic tumor models. Mice bearing RENCA or A20 tumors were treated with the same modalities and similar results were generated. Single-agent HBI-8000, PD-1 Ab (RENCA, FIG. 8E, F), or PD-L1 Ab (A20, FIG. 8I, J) did not significantly affect either median tumor growth or survival. As seen in the MC38 model, however, the combination of either a PD-1 Ab or PD-L1 Ab with HBI-8000 produced a significant and synergistic decrease or delay in tumor growth and progression, and importantly, an increase in the number of mice with tumor regression. Finally, we tested HBI-8000, a mouse CTLA-4 Ab, or HBI-8000 plus CTLA-4 Ab in the CT26 model (FIG. 8G, H). Similar to ICIs targeting PD-1 and PD-L1, the CTLA-4 Ab alone did not significantly affect tumor growth. Combining HBI-8000 with CTLA-4 Ab produced a highly significant delay in tumor progression, with 20% of tumor-bearing mice experiencing complete regression.
[0253] In summary, irrespective of the mouse tumor model or ICI Ab, single-agent therapy did not inhibit/regress tumor growth in any of the models tested. In all treatments combined with HBI-8000, we observed tumor regression after treatment, with subsets of tumors showing a significant delay in progression or outright regression. The data indicate that combining HDACi HBI-8000 with an ICI Ab was very efficacious in multiple animal models.
[0254] HBI-8000 epigenetically reprograms the TME and increases the expression of genes indicating enhanced antigen presentation, dendritic cell function, and effector cell antitumor cytotoxicity.
[0255] To investigate the mechanism of action of HBI-8000 in combination therapy with ICIs, larger groups of mice (n=20/group) were implanted with MC38 tumors to sufficiently power the statistical analysis. A baseline no-treatment tumor-bearing group was killed 1 day before initiating treatment. Mice in each treatment arm (n=20) were killed at days 7, 14, and 17 post treatment initiation. The NanoString nCounter PanCancer Immune Profiling Panel analysis allows for clustering of immune response-related genes into "gene sets" comprising a collection of genes selected as being representative of an element of the immune response (i.e., cell type, pathway), and provides a high level view of the antitumor response, which is depicted as scatterplots in FIG. 9A. While all the scores were elevated in the PD-1 Ab plus HBI-8000 combination agent cohorts, it is noteworthy that a subset of cell type scores was augmented by either PD-1 Ab or HBI-8000 alone as early as day 7. Scores for exhausted CD8 T cells and neutrophils were predominantly augmented by the PD-1 Ab. In contrast, HBI-8000 augmented the scores corresponding to dendritic cells, macrophages, NK cells, cytotoxic cells, and CD45 cells, demonstrating that HBI-8000 alone had a profound conditioning or priming effect on immune-relevant gene expression within the TME, and suggesting that it reprograms the TME such that ICI therapy is more effective.
[0256] The de novo generation of new tumor-selective T cell clones might be a key factor in the response to the PD-1/PD-L1 checkpoint blockade. Because data from a preliminary study suggested that HBI-8000, alone or in combination with PD-1 Ab, has profound effects on the early or priming phase of the immune response, we investigated changes in the expression of genes associated with dendritic cell functions, antigen processing, and MHC class II antigen presentation. Consistently, gene expression analysis in single agent HBI-8000-treated tumors showed at least partial co-clustering with the response to the HBI-8000 plus ICI Ab combination therapy within these gene sets (FIG. 14A). HBI-8000 also co-clustered with combination therapy at the level of MHC class I antigen expression and presentation, which is important for effector T cell recognition and killing of tumor cells (FIG. 14B). Unsupervised hierarchical clustering of the indicated immune cell type scores vs. treatment and tumor response (FIG. 14A) showed that gene expression changes representative of these scores were most notable in the PD-1 Ab plus HBI-8000 combination cohort, and in responders vs. nonresponders (FIG. 14B). The analysis also demonstrated segregated clustering of the adaptive vs. innate response cells types. Not surprisingly, PD-1 Ab plus HBI-8000 combination therapy co-clustered with gene expression sets representing high response rates (TGI>75%), which was observed for both adaptive and innate immune cell types. HBI-8000 also co-clustered with the HBI-8000/PD-1 Ab combination in modulating the expression of gene sets associated with cytokines, chemokines and their receptors, and with adaptive immunity-related genes. The data suggest that the class I/II selective HDACi can epigenetically modulate gene expression patterns within the TME, which contributes to multiple facets of the antitumor immune response, leading to the priming of effector T and B cells, the recognition of tumor cells by T cells with a consequent shift in the expression of relevant cytokines and corresponding receptors, and the augmentation of both innate and adaptive immune responses. Also see heat map data Table 2 below.
TABLE-US-00002 TABLE 2 Heat map data for FIG. 14B Partial APD-1 + Response APD-1 HBI- Response HBI - vs. No vs. 8000 vs. vs. No 8000 vs. Response Control Control Response Control Gene Cluster 0.7 1.5 1.8 2 2.7 Cytokines and Receptors 0.5 1.5 1.8 2 2.7 Innate 0.5 1.5 1.5 2 2.7 TLR 0.5 1.5 2 2 2.7 Adhesion 0.7 1.5 1.8 1.5 2.7 Cancer progression 1 1.8 1.8 2 3 Humoral 1 1.5 1.8 1.8 2.8 Macrophage function -0.5 1.7 2 1.8 2.7 Adaptive -1.5 1.5 1.8 1.8 2.7 Inflammation -1.5 1.5 1.8 1.8 2.8 Transporter Function -1.5 1.5 1.8 1.8 2.8 Leukocyte Function 0.5 1.5 1.8 1.8 2 Apoptosis 0.5 1.3 1.5 1.2 2 Cell cycle -2 1.5 1.3 1.6 2.2 Pathogen Receptors -1.5 1.5 1.3 1.3 2 Senescence 0.5 1 1.5 2 2.5 Interleukin 0.5 1 1.5 2 2.5 Chemokine and Receptors 0.5 1 1.5 2 2.5 TNF superfamily 0.5 1 1.5 2 2.5 B cell Functions 0.5 1 1.7 2.2 2.7 CD molecules 0.7 1.2 1.5 2.2 2.5 T-cell Functions 0.5 1.5 1.5 1.8 1.8 Class I MHC mediated antigen process 0.5 1.5 1.5 2 1.8 Interferon 1.5 1.5 1.5 2.5 2 NK cell Function 0.5 1.5 0 2 2.8 Complement pathway -2 1 1.5 0 2 Basic Cell Function 1.5 1.8 1.8 3 2.8 MHC 1.5 1.8 1.8 2.5 2.8 Antigen processing 0.5 1.8 1.8 2 2.8 Dendritic cell Functions 1.5 1.5 1.8 4 2.8 MHC class II antigen presentation 1.5 1.8 1 4 2.8 Costimulation by the CD28 family 1 1.5 3 2 3 Microglial Functions
[0257] HBI-8000 alone or in combination with PD-1 Ab induces changes in several immune checkpoints within the TME.
[0258] The changes observed in immune checkpoints in the MC38 TME are shown in FIG. 9B. The data plots are color-coded to represent the tumor growth inhibition response seen for each individual animal, set arbitrarily for the purpose of illustration as tumor growth inhibition >75%, 25% through 75%, or less than 25% to represent responders, stable disease, and progressers, respectively. We observed increased expression of the immune checkpoints PD-1, PD-L1, CTLA-4, and CD86 (CD28L), the expression levels of which correlated with antitumor efficacy and tumor regression (FIG. 9B). We also observed statistically significant changes in the expression of immune checkpoints CD276/B7-H3 and CD244 (FIG. 9B), as well as lymphocyte activation gene-3 (LAG-3), T cell immunoreceptor with Ig and ITIM domains (TIGIT), ecto-5'-nucleotidase (NT5E/CD73), signal regulatory protein .alpha. (SIRP.alpha.), nuclear factor of activated T cells 4 (NFATC4), and poliovirus receptor (CD155; FIG. 15). Modulation in the expression of the above genes indicates a shift from a noninflamed (cold) TME to an inflamed (hot) TME, and correlated with the antitumor response in MC38 tumor-bearing mice.
[0259] HBI-8000 alone or in combination with PD-1 Ab induces changes in immune markers in the TME, including co-stimulators, markers of cytotoxicity, cytokines and associated receptors, and MHC.
[0260] In addition to analyzing the effects of HBI-8000, PD-1 Ab, or their combination on various immune pathways, cell type functional scores, and immune checkpoint markers, we examined the effect of HBI-8000, PD-1 Ab, and their combination on a number of individual genes relevant to either innate or adaptive immunity (FIGS. 10-12, and Supplemental FIGS. 15, 16). Genes modulated predominantly by the PD-1 Ab included CD8a (FIG. 11), inducible T cell costimulator (ICOS/CD278), and CD40/CD40L (FIG. 16). PD-1 Ab was also the driver for changes in the expression of genes involved in T cell recruitment, memory, and the CD8 T cell response, including CXCR6 (FIG. 12), ICOS, CD40, and its ligand CD40L/CD154 (FIG. 16). Our analysis of the nCounter data (FIG. 11) showed increased T-effector and interferon-y gene scores, which, along with increases in granzyme B (GZMB) and perforin-1 (PRF1), are collectively consistent with an enhanced T-effector and interferon-.gamma. gene, reflecting enhanced existing immune competency.
[0261] The modulation of many genes occurred, indicating that the TME inflammation status was dependent exclusively on the combination of HBI-8000 plus PD-1 Ab. Examples included the co-stimulator CD86 (FIG. 9B), chemoattractant receptors C--C chemokine receptor (CCR) 5 (FIG. 10), and CCR1 (FIG. 11), which are important for initial events in effector T-cell differentiation, markers of increased tumor reactive effector cells, e.g., ectonucleoside triphosphate diphosphohydrolase-1 (ENTPD1/CD39; FIG. 11), PRF1 (FIG. 11), and effector T cell memory precursors (interleukin 7 receptor [IL7R] and interferon regulatory factor 4 [IRF4], FIGS. 12, 10, respectively). Because HBI-8000 enhances both CD8 T cell and NK cell activity and functions, relevant genes modulated predominantly by HBI-8000 are of great interest. HBI-8000 alone, and not PD-1 Ab alone, drove changes in the expression of 4-1BB/CD137 (FIG. 10) and tumor necrosis factor .alpha. (TNF.alpha.; FIG. 10), interleukin 2 receptor alpha (IL2R.alpha.)/CD25 and GZMB (FIG. 11), IRF4 (FIG. 10), and chemokine (C-X3-C motif) receptor 1 (CXC3R1), chemokine (CXC motif) receptor (CXCR)6, and CXCR3 (FIG. 12), genes that are relevant to an initial cytokine or CD8 effector response, tumor infiltrating lymphocyte (TIL) recruitment, effector cell differentiation, and effector memory.
[0262] Importantly, many genes were modulated by HBI-8000 alone relatively early (day 7) in the antitumor response (e.g., 4-1BB/CD137, CD86, TNF.alpha., CCR5, chemokine (C--C motif) ligand 2 (CCL2), IL2R.alpha./CD25 [FIGS. 10, 9b, 10, 10, 10, 11, respectively], and CCR1 and GZMB [FIG. 11]). Consistent with reports of HBI-8000 having a positive effect on NK cell functions and innate immunity, we observed that HBI-8000 alone or combined with PD-1 Ab modulated the expression of GZMB (FIG. 11), killer cell lectin like receptor D1 (KLRD1/CD94; FIG. 10), and killer cell lectin like receptor C2 (NKG2c/KLRC2), natural killer cell granule protein 7 (NKG7), and killer cell lectin like receptor K1 (KLRK1; FIG. 16). Finally, and consistent with the upward shifts seen in all scores relevant for antigen presentation machinery and supportive of antigen presentation or tumor cell recognition, we observed increases in the expression of several MHC class I (H2-D1, H2-K1) and II genes (H2-Aa, H2-Eb1) (FIG. 12), predominantly in response to HBI-8000 alone (H2-D1, H2-K1) or the combination of HBI-8000 and PD-1 Ab (H2-Aa, H2-Eb1). This is an important observation and relevant to the reversal of known mechanisms of resistance to ICIs, namely the loss of MHC class I and class II molecules, which impede tumor cell recognition by effector CD8 T cells, as well the presentation of tumor antigens, including neoantigens, to naive de novo antitumor immune cells.
[0263] HBI-8000 combined with ICI rescues mice progressing on single-agent ICI therapy in a model of stable disease leading to acquired resistance and progression.
[0264] Human cancer patients receiving ICI therapy often experience a transient response or stable disease, but eventually develop resistance and progress, a challenge to which major efforts are directed. Because gene expression data showed that HBI-8000 alone induced positive changes in a significant number of immune-related pathway scores and genes, we examined the ability of HBI-8000 to halt or even reverse progression in mice first treated with single-agent ICI therapy, alone or in combination with an ICI. To explore the effect of HBI-8000 plus ICI on acquired resistance, we developed a model based on the repeated observations that tumor-bearing mice treated initially (first-line) with single agent PD-1 Ab or PD-L1 Ab display 4 patterns of growth: approximately 20% experience rapid progression, approximately 20% experience complete regression, and approximately 60% experience stable tumor growth (defined as 3 consecutive tumor volume measurements with no significant change) or slow progression (relative to rapid growth and progression), which somewhat approximates the clinical situation. Using the above model, we treated a large cohort of tumor-bearing mice with PD-1 Ab alone. Once they reached the criteria for stable disease or slow progression, they were randomized into 6 treatment arms as indicated in FIG. 13. We compared the effect of halting treatment (Vehicle), continuing to treat with PD-1 Ab, or continuing PD-1 Ab in combination with HBI-8000. We also compared the effect of mAbs directed against the reciprocal target, PD-L1, treating mice with PD-L1 Ab, alone or in combination with HBI-8000. The results of one representative experiment are shown in FIG. 13. In mice failing PD-1 Ab therapy, HBI-8000 was modestly active as a second-line therapy, with a complete response in 2 mice and a partial response in 3 mice at the end of the study. The second course of PD-1 Ab, however, failed to significantly affect tumor growth. The modest delay seen in overall tumor growth provided by treatment with PD-L1 Ab alone was not significant, but there was 1 complete responder (tumor regression) and 4 partial responders. A second course of anti-PD-1 therapy combined with HBI-8000 produced no delay in tumor growth compared with anti-PD-1 alone. In contrast, combination therapy with HBI-8000 and anti-PD-L1 significantly (p<0.05) inhibited tumor growth, indicating that mice progressing on one ICI therapy would see benefit from an alternative ICI in combination with HBI-8000.
[0265] Class I-selective HDAC inhibitors reinvigorate immune response when combined with ICIs. On the basis of recent reports, HBI-8000 will function as an epigenetic immunomodulator to reprogram the TME, converting immunologically cold or nonresponsive tumors to hot or responsive tumors, and tested this hypothesis in preclinical syngeneic mouse models of tumor immunotherapy. The ability of HBI-8000 as an HDACi to modulate several immune pathways important to antitumor immunity indicated that these changes in the TME epigenome may significantly improve overall responses to ICIs. This is consistent with accumulating evidence that benzamide class I-selective HDACi can reprogram the TME epigenome to improve the antitumor efficacy of ICIs (7, 35-38, 50, 51). Indeed, HBI-8000 combined with any of the 3 ICIs tested (PD-1 Ab, PD-L1 Ab, and CTLA-4 Ab) displayed enhanced tumor growth inhibition. The nCounter data suggest that the activity of HBI-8000 extended to both adaptive and innate immune functionalities. This is consistent with changes we observed in the expression of several immune checkpoint molecules associated with an immune T cell-inflamed TME. Interestingly, the gene expression responses observed followed 3 patterns (Table 1): i) those that were predominantly driven by PD-1 Ab and the combination, ii) those that were predominantly driven by HBI-8000, and iii) those were modulated primarily or exclusively by the combination, clearly indicating cooperativity between HBI-8000 and anti-PD-1 in the induction of expression of these genes. Notably, CD276/B7-H3 and CD244/2B4 (FIG. 9B) as well as CD73/NT5E (FIG. 15) were modulated primarily by HBI-8000, with little or no contribution from the addition of PD-1 Ab, again suggestive of an epigenetic reprograming or "priming" effect on the TME by the HDACi.
[0266] HBI-8000, either alone or in combination with PD-1 Ab, altered the expression of several immune checkpoints, many of which offer potential targets for immunotherapy combinations with HBI-8000. Interestingly, this appeared to be a cooperative effect of HBI-8000 and PD-1 Ab in most cases, as neither agent alone was sufficient. In some cases, however, such as CD276/B7-H3 and CD244/2B4, increased expression was mediated by HBI-8000 alone. CD276 is expressed on antigen-presenting cells and plays an important role in the inhibition of T cell activation and function. The increase in CD276/B7-H3 expression by HBI-8000 may correlate with the observed augmentation of dendritic cells and associated antigen presenting machinery by HBI-8000. It may also affect the innate immune response and protect tumor cells from NK-mediated cytotoxicity. CD244 is an immunoregulatory receptor in the signaling lymphocyte activation molecule (SLAM) family with both activating and inhibitory properties that seems to function primarily to mediate inhibitory signaling and T cell exhaustion, and offers another potential target for immunotherapy.
[0267] Tumor-infiltrating lymphocytes are associated with a survival benefit in several cancer types and with the response to immunotherapy. The requirements for maintaining a CD8 T cell TIL response against human cancer cells may depend on the presence of stem-like T cells, a distinct subpopulation of CD8 T cells within tumors. Stem-like T cells are delineated by the expression of TCF1, IL7R, and IL2R.alpha./CD25 (changes observed in our nCounter data) as well as the co-stimulatory molecules CD28, CD226, and CD2. Stem-like T cells terminally differentiate into effector CD8 T cells, which express higher levels of granzymes, perforin, and checkpoint molecules. These stem-like T cells reside in dense antigen-presenting cell niches within the tumor, and tumors that fail to form these structures are not extensively infiltrated by T cells. Moreover, patients with progressive disease lack these immune niches. The increased dendritic cell, MHC class I and II antigen presentation machinery scores together with an increase in both MHC class I and II gene expression driven by HBI-8000 may contribute to the formation and maintenance of these antigen-presenting cell niches, leading to a CD8 T cell TIL response in the TME. Indeed, HBI-8000 in combination with PD-1 Ab or PD-L1 Ab induced an increase in the expression of CD8 in TILs (FIG. 11), along with higher levels of interferon-.quadrature., granzymes, perforin, and checkpoint molecules in treated tumors. It remains unclear if the increase in immune checkpoint activity in the combined regimen with HBI-8000 is a consequence of the epigenetic changes induced directly on tumor or immune cells or the result of a shift in TME cytokine/chemokine profiles. The current data, however, suggest that HBI-8000 alters the TME epigenome, which is necessary for expanding and maintaining both stem-like and effector CD8 cell populations, resulting in more numerous and activated CD8 effector cells as reflected by the increase in the cytotoxic cell, NK CD56dim, CD8 and CD8 vs. exhausted CD8 scores.
[0268] An important and under-appreciated mechanism of adaptive tumor resistance is the epigenetic or mutational silencing of the apoptosis machinery. Immunogenic tumor cell death can drive the priming and clonal expansion of tumor-selective effector T cells, but it is ultimately the ability of cytolytic cells to kill tumor cells. HBI-8000 can directly induce cell cycle arrest and apoptosis in a large number of tumor cells and tumor cell lines [, NCI-60 Panel (data not shown)], but has also been shown to potentiate the cytotoxic activity of a number of anticancer agents by skewing the balance of expression toward pro-apoptotic proteins, and thus triggering the apoptotic response. Based on the current data, as well as recent reports describing immunomodulatory activities of other class I selective HDACi, there are at least 2 mechanisms at play: i) induction of immunomodulatory activities, including boosting antigen presentation and tumor cell recognition by immune effector cells and ii) immunogenic cell death, leading to the release of neoantigens and a potential increase in T cell priming and de novo generation of new tumor-selective effector T cell clones. Evidence is accumulating that a robust and durable antitumor immune response depends on the generation of novel tumor selective T cell clones and not necessarily the reinvigoration or reprogramming of exhausted T cells. The observed shift in the CD8 effector T cell to exhausted T cell ratio may reflect an influx of new tumor-selective T cells.
[0269] Using a model of resistance to ICI and tumor progression, we found that second-line HBI-8000 in combination with an ICI rescued a percentage of mice failing ICI therapy (FIG. 13). The ability of HBI-8000 to enable the immune system to target resistant cancer cells may be due in part to its putative effect on antigen presentation and clonal repopulation of the immune response, or its ability to enhance the reinvigoration of exhausted T cells, or both. Ultimately, HBI-8000 and other class I-selective HDACi may epigenetically alter regulatory mechanisms that contribute to achieving a threshold of immunogenic (proinflammatory) signaling that is required to elicit an anti-tumor or autoimmune response.
[0270] In addition to targeting class I HDACs, HBI-8000 inhibits the activity of class II HDAC10, which is involved in adaptive resistance to the antitumor immune response. In a recent study, knockdown of HDAC10 recapitulated the effects of HDAC inhibitors on immunotherapy biomarkers. Therefore, targeting HDAC10 in addition to inhibiting HDACs 1, 2, and 3 may provide further support for the role of HBI-8000 as an epigenetic modulator and primer of the TME.
[0271] In summary, our data provides a deeper understanding of the effect of class I HDAC inhibitors on the TME. Consistent with the preclinical data presented here, clinical data for HBI-8000 in combination with nivolumab suggest enhancement of activity of nivolumab by HBI-8000 in patients with melanoma, renal cell carcinoma, and non-small cell lung cancer (https://clinicaltrials.gov/ct2/show/NCT02718066), where the durability and sustainability of response appears elevated even after treatment cessation. This contrasts with other attempts to use HDACi with checkpoint inhibitors to generate clinical responses in patients who have failed prior treatment with ICI (https://www.ascopost.com/News/59894). The current preclinical data may further explain the efficacy and durability of HBI-8000 in combination with nivolumab in the clinical setting. Future studies will be aimed at better understanding the durability of the responses elicited by HBI-8000 by interrogating patient samples through cellular and molecular analysis.
ABBREVIATIONS
[0272] Ab antibody
[0273] CTLA-4 cytotoxic T-lymphocyte-associated protein 4
[0274] CCL2 chemokine (C-C motif) ligand 2
[0275] CCR chemokine (C-C motif) receptor
[0276] CXCR chemokine (CXC motif) receptor
[0277] CXC3R1 chemokine (C-X3-C motif) receptor 1
[0278] ENTPD1 ectonucleoside triphosphate diphosphohydrolase-1
[0279] GZMB granzyme B
[0280] HDAC histone deacetylase
[0281] HDACi histone deacetylase inhibitor
[0282] ICIs immune checkpoint inhibitors
[0283] ICOS inducible T cell costimulator
[0284] IL2Ra interleukin 2 receptor alpha
[0285] IL7R interleukin 7 receptor
[0286] IRF4 interferon regulatory factor 4
[0287] KLRC2 killer cell lectin like receptor C.sub.2
[0288] KLRD1 killer cell lectin like receptor D1
[0289] KLRK1 killer cell lectin like receptor K1
[0290] LAG-3 lymphocyte activation gene-3
[0291] mAb monoclonal antibody
[0292] MHC major histocompatibility complex
[0293] NK natural killer
[0294] NKG7 natural killer cell granule protein 7
[0295] NFATC4 nuclear factor of activated T cells 4
[0296] NT5E ecto-5'-nucleotidase
[0297] PD-1 programmed cell death receptor-1
[0298] PD-L1 programmed cell death receptor-1 ligand 1
[0299] PRF1 perforin-1
[0300] SIRP.alpha. signal regulatory protein .alpha.
[0301] TGI tumor growth inhibition
[0302] TIGIT T cell immunoreceptor with Ig and ITIM domains
[0303] TIL tumor infiltrating lymphocyte
[0304] TME tumor microenvironment
[0305] TNF.alpha. tumor necrosis factor alpha
TABLE-US-00003 Immune System Treatment HBI-8000 Anti-PD1 Combo Function Genes D 7 D 14 D 17 D 7 D 14 D 17 D 7 D 14 D 17 Innate Immunity CCL2 + + - - - - + + + Antigen H2-Aa + + - + + - + + + H2-EB1 + + - + + - + + + Presentation H2-D1 + + + + + + + + + H2-K1 + + + + + + + - + CCR5 + + - - + - + + + Checkpoint PD-1 - - - + + + + + + Molecules PD-L1 + - - + + + + + + CTLA4 + - - + + + + + + CD86 - - - - - - - - - Co-Stimulatory 4-1BB + + + + - - + + - Effector Response TNF.alpha. + + + + + - + + + KLRD1 + - - + - - + + + IL-2R.alpha. + + - + - - + + + CD8.alpha. - - - + - - + + + CCR1 + + + + + - + + + ENTPD1 + - - + - - + + + GZMb + + + + + + + + + PRF1 + + - + + + + + + Memory T Cells IL-7R + - - - - - + + + CXCR6 + - - + - + + + + CX3CR1 + + + + + - + + + CXCR3 - - - + - - + - +
[0306] Although the invention has been described with reference to the disclosed embodiments, those skilled in the art will readily appreciate that the specific examples and studies detailed above are only illustrative of the invention. It should be understood that various modifications can be made without departing from the spirit of the invention. Accordingly, the invention is limited only by the following claims.
EXAMPLES
[0307] The following examples are included for illustrative purposes only and are not intended to limit the scope of the invention.
Example 1
[0308] The rationale for pursuing cancer immunotherapy as a therapeutic option has been driven by a long history of evidence that tumors can be recognized as non-self (similar to immune detection of pathogens like virus-infected cells), rather than as self (normal tissue). The immune system sees tumors as non-self mostly through early detection of molecules displayed on tumor cells that can be recognized as foreign by the immune system, which ideally becomes activated and effectively attacks and eliminates the tumor cells. A number of steps must precede an activated immune response, during which antitumor immune cells can enter (infiltrate) and engage malignant cells within the now immunologically inflamed or "hot" tumor. This is commonly referred to as the Cancer-Immunity Cycle (Chen et al.). Tumor cells however can adapt over time and evade or become resistant to an antitumor immune response. A number of such resistance mechanisms are now known, and the currently approved immunotherapies have been developed to block some of these resistance mechanisms. For example, the antibodies directed against the CTLA-4 ligand (Yervoy.RTM., ipilumumab), the PD-1 receptor (Opdivo.RTM., nivolumab; Keytruda.RTM., pembrolizumab, and others) or its ligand PD-L1 (Tecentriq.RTM., atezolizumab; and others) target these immune checkpoints and, at least in a portion of patients, relieve antitumor resistance mediated through the CTLA-4/B7.1 and B7.2 immune checkpoint inhibitory axis or PD-1/PD-L1 checkpoint inhibitory axis. Although the use of these immune checkpoint targeting antibodies has resulted in significantly improved patient benefit and produced remarkable clinical responses in various cancers, a significant number of patients have tumors that are either inherently resistant or develop resistance, their tumors become non-inflamed, lack immune cell infiltrates (TILs) and are referred to as immunologically "cold" and the disease eventually progresses. For this reason, rational drug combinations with these checkpoint inhibitors and other immunomodulating agents are imperative to improve the rate of durable responses and patient survival. (See West A C and Johnstone R W. (2014) New and emerging HDAC inhibitors for cancer treatment. J. Clin. Invest. 124 (1): 30-39).
[0309] HBI-8000 is a histone deacetylase inhibitor (HDACi), which as an epigenetic regulator that can change the expression of genes, up or down, without changing the DNA sequence, and therefore has the ability to alter the expression of genes which are aberrant, silenced or overexpressed in cancer cells [West and Johnstone, 2014]. Recently there have been several reports describing the ability of some HDACi (e.g., HBI-8000) to enhance antitumor immunity through positive effects on a number of the tumor resistance mechanisms. HDACi's can restore an inflamed or "hot" immune tumor environment with immune cell infiltrates that become active and drive antitumor immunity.
[0310] The immunomodulatory effects of the HDACi HBI-8000 on the tumor micro-environment ("TME") have been well documented. HBI-8000 administration increases the influx of CD8.sup.+ T cells and NK cells, and improves their function. HBI-8000 administration also reduces the number and activity of regulatory T cells (TREGs) and myeloid-derived suppressor cells (MDSCs), and promotes conversion of M2 (suppressive) to M1 (antitumor) macrophages. HBI-8000 administration also increases PD-1 and PD-L1 expression in "cold" tumors, along with several other important immune signatures indicative of cold to hot conversion--this process starts early and increases over time, as does the number of responders. HBI-8000 drives positive changes in dendritic cell scores and signatures in the TME, positive changes in antigen presentation, processing, and display pathways, e.g. MHC Class I and Class II expression (mechanisms of tumor evasion). HBI-8000 increases the ratio of active CD8.sup.+ effector T cells to "exhausted" CD8 T cells and the cytotoxic score and signature, implying re-activation of inactive tumor selective T cells. HBI-8000 drives changes in the tumor cells themselves, priming and sensitizing them to the antitumor immune response--increasing apoptosis scores and signatures, indicating re-expression of the apoptotic machinery needed for killing the tumor cells. HBI-8000 driven TME changes also results in increased presence and activity of NK cells and M1 macrophages (innate immune system), both of which contribute to the overall antitumor immune response.
[0311] To determine the efficacy of HBI-8000 in combination with anti-CTLA-4 and anti-PD-1 checkpoint inhibitor antibodies, a study was conducted using the syngeneic MC38 colon adenocarcinoma in female C57BL/6 mice. The present study consisted of eleven groups (n=9 or 8 of female C57BL/6 mice bearing subcutaneous MC38 tumors (mean volume: 106 mm.sup.3-111 mm.sup.3) on Day 1 of the study, when dosing was initiated. Vehicle (10% Hydroxypropyl-(3-Cyclodextrin, 10% Propylene glycol in DI water, pH 2.5) and HBI-8000 (50 mg/kg) were administered orally (p.o.), once daily for twenty one days (qd.times.21). Anti-CTLA-4 was administered intraperitoneally (i.p.) at a dose of 2.5 mg/kg on days 1, 4, and 7. Anti-PD-1 was administered i.p. at 5 mg/kg, twice a week for two weeks (biw.times.21.
TABLE-US-00004 TABLE 2 Group Number of No. animals Treatment 1 Treatment 2 Treatment 3 1 8 Vehicle 2 9 HBI-8000 50 mg/kg; qdx21 3 8 CTLA-4 mAb 2.5 mg/kg; d 1, d 4, d 7 4 8 PD-1 mAb 5 mg/kg; biw x 2 5 8 CTLA-4 mAb HBI-8000 2.5 mg/kg; 50 mg/kg; d 1, d 4, d 7 qdx21 6 9 CTLA-4 mAb PD-1 mAb 2.5 mg/kg; 5 mg/kg; d 1, d 4, d 7 biw x 2 7 8 CTLA-4 mAb PD-1 mAb HBI-8000 2.5 mg/kg; 5 mg/kg; 50 mg/kg; d 1, d 4, d 7 biw x 2 qdx21 CTLA-4 mAb = hybridoma clone 9H10 PD-1 mAb = hybridoma clone RMPI-14
[0312] Animals were euthanized when tumor volumes reached 3000 mm.sup.3 or on the last day of the study (Day 43), whichever came first, and the time to endpoint (TTE) was calculated. Treatment outcome was determined from percent tumor growth delay (% TGD), defined as the percent increase in median TTE for treated versus control mice, with differences between the treatment groups deemed statistically significant at P<0.05 using logrank survival analysis. Mice were also monitored for complete regression (CR) and partial regression (PR) responses. Treatment tolerability was assessed by body weight (BW) measurements and frequent observation for clinical signs of treatment-related (TR) side effects.
[0313] The results of these experiments are found in FIG. 1. The triplet combination therapy comprising a compound of formula I, a CTLA-4 inhibitor as described herein, and a PD-1 inhibitor as described herein resulted in statistically significant tumor volume reduction. The subjects in the triplet combination group also showed a 40-50% survival rate at the conclusion of the experiments. (See FIG. 2). Collectively, these results show that the compounds of formula I enhance the activity of the triplet combination therapy.
Example 2
[0314] FIG. 3A depicts the probability of progression free survival ("PFS") in terms of months resulting from a combination therapy comprising compounds of formula I and Nivolumab in melanoma. This probability was generated from the results published in the New England Journal of Medicine showing the PFS for patients treated with Nivolumab monotherapy, ipilimumab monotherapy, or a Nivolumab plus ipilimumab combination therapy. (See FIG. 3B). The "tailing" of the plot in FIG. 3B from the combination therapy suggests a synergistic effect between the Nivolumab and ipilimumab.
[0315] To test this hypothesis, compounds of formula I were administered in combination with Nivolumab to 20 patients suffering from melanoma ("MEL") (15 of these patients represented 1.sup.st line of treatment), 11 patients suffering from renal cell carcinoma ("RCC"), and 13 patients suffering from non-small cell lung cancer ("NSCLC"). Prior to testing, the safety profiles of various dosages of the compounds of formula I were tested by administering escalating doses of the compounds of formula I in combination with the standard dose of nivolumab. A 30 mg BIW was established. The profile of the 20 MEL patients is depicted in Table 3.
TABLE-US-00005 TABLE 3 Checkpoint-Naive Melanoma Patients (N = 20) Characteristics Male/Female, n (%) 13/7 (65/35) Median age, years (range) 64.5 (28-79) <65, n (%) 10 (50) 66-75 6 (30) >75 4 (20) ECOG Score, n (%) 0 13 (65) 1 7 (35) Stage at study entry, n (%) M1a 5 (25) M1b 12 (60) M1c 3 (15) Elevated LDH, n (%) 2 (10) Median tumor burden (target 38 (10-167) lesions), mm (range) Median time since diagnosis, 13.8 (0.7-66.1) months (range) BRAF status, n (%) Mutated 4 (20) Wildtype 1 (5) Unknown 15 (75) Prior surgery, n (%) 17 (85) Prior radiation, n (%) 6 (30) Prior systemic therapy, n (%).sup.a Chemotherapy 1 (5) Immune therapy (excluding 3.sup.b (15) PD-(L)1 inhibitor) Other 2.sup.c (10) Footnotes: .sup.a2 subjects received multiple therapies .sup.bipilimumab (2); cellular immunotherapy (1) .sup.cvemurafenib (1); 1 subject received MEK inhibitor & BRAF inhibitor
[0316] Imaging studies were performed every 8 weeks to assess tumor response according to RECIST v1.1. Tumors were observed for Objective Response Rate (ORR), Disease Control Rate (DCR), and Standard Disease (SD). The results of the imaging studies are summarized in Table 4. These results are further detailed in FIG. 4. The waterfall plot of FIG. 4 shows CPI-naive subjects dosed with compounds of formula I in combination with nivolumab. Each bar represents a single patient's best response as defined by the sum of target lesion diameters, measured in terms of change in percent (baseline is 0% change). Bars falling within +20% increase in tumor size and -30% decrease in tumor size are considered stable disease. Further characteristics of the subjects from the study are summarized in Table 5. And the PFS characterized by metastasis distribution is summarized in Table 6 while the distribution of PFS characterized by metastatic sites is summarized in Table 7.
[0317] The group of MEL subjects were analyzed for total time on treatment regime, termination reason, and best ORR. The status of the subject's BRAF gene is also noted. The results are summarized in the swimmer plot of FIG. 5. All treatments were first-line unless noted with "2L."
TABLE-US-00006 TABLE 4 Phase 1b/2 MEL RCC NSCLC Total Enrolled 20 11 13 44 Evaluable 18 9 8 35 ORR 67% 33% 38% 51% SD 28% 44% 38% 34% DCR 94% 78% 75% 86%
TABLE-US-00007 TABLE 5 Characteristics of Subjects Days Baseline Characteristics Days of Tx Days to Treatment BRAF Prior Since last M Mets HBI- Best PD or D/C Subject Mut PD-L1 Tx Syst Tx LDH Category Sites 8000 Nivo Response Death Reason 090201 Radiation N/A Normal M1b Lung 669 687 PR 724+ 090408 Radiation N/A Normal M1b Lung, 81 57 PR 525+ Node 010716 Wt Neg lpi 121 Normal M1a Nodes 487+ 487+ PR 487+ 090304 None N/A Normal M1b Lung 459 419 SD 459+ 090724 Wt None N/A Normal M1c Pancreas 427+ 427+ PR 427+ 090723 Pos Excision N/A Normal M1b Lung, 40 253 PR 383+ Node 090731 Wt None N/A Normal M1a Nodes 361+ 361+ PR 361+ 090727 Wt None N/A Normal M1b Lung 109 336 CR 355+ 090721 None N/A High M1c Liver, 169 141 PR 343+ Lung 090733 V600E None N/A Normal M1b Lung 341+ 341+ CR 341+ 090732 None N/A Normal M1c Lung, 187 295 PR 292+ PI Adrenal, decision Node 010719 unknown None N/A Normal M1c Liver, 97 169 SD 197 Clin PD Lung, Nodes 090406 Radiation N/A Normal M1a Muscle 109 46 CR 178 PI discretin, PD date = death 090722 None N/A High M1b Lung 31 169 SD 168+ not included in ORR due to <8 weeks of HBI-8000 090711 Surg N/A Normal M1c Liver 110 183 SD 168 Resection 090730 Pos None N/A Normal M1a Soft 163 169 SD 163 D/C due to tissue clinical progression 090728 Pos None N/A Normal M1b Lung, 137 141 SD 137 D/C due to Soft clinical tissue progression 090302 vemuraf/ 30 Normal M1b Lung 109 127 PR 110 TIL 090717 Pos TKI 334 Normal M1b Lung 81 85 PD 57
TABLE-US-00008 TABLE 6 PFS by Metastasis Distribution PFS (days) N = 19 M1a M1b M1c >400 n = 5 1 3 1 200-400 n = 6 1 3 2 100-200 n = 7 2 3 2 <100 n = 1 1
TABLE-US-00009 TABLE 7 Distribution of PFS by Metastatic Sites PFS Days M Stage N >400 200-400 100-200 <100 M1a 4 1 1 2 0 M1b 10 3 3 3 1 M1c 5 1 1 2 0
Example 3
[0318] Additional studies were conducted to test the effect of a combination therapy comprising compounds of formula I and nivolumab in melanoma patients with prior immune checkpoint treatment. In these studies, 8 patients were evaluable. 2 of the 8 patients showed PR. 4 of the 8 patients showed SD. Further, the ORR was 25% in this group, while the DCR was 75%. A patient from this study had a tumor that was NRAS positive. Further, the patient had high LDH and an unknown level of PD-L1 expression. This patient had extensive prior treatment including surgery, radiation, ipilimumab+nivolumab, nivolumab maintenance, T-vec and pembrolizumab, TIL+high dose IL-2. This patient achieved a PR in 54 days and was on treatment for over 249 days. This PR is suggestive of epigenetic effects on the tumor. The characteristics and outcome summaries for the patients in this study are summarized in Table 8. Based on these results, combined with the data collected from melanoma-naive patients (FIG. 5), it is recommended that the combination treatment of the compounds of formula I and nivolumab is used as a second-line treatment for patients having failed BRAF/MEK inhibitors (in patients with a BRAF mutant).
TABLE-US-00010 TABLE 8 Patients Characteristics and Outcome Summary-Checkpoint Inhibitor Treated Prior Systemic Days Baseline Characteristics Days of Tx Days to Treatment BRAF PD- Tx (best Since Last M Mets HBI- Best PD or D/C Subject Mut L1 response) Syst Tx LDH Category Sites 8000 Nivo Response Death Reason 010717 Wt Ipi x 4 (PR) N/A Normal M1a Node, 98 112 SD 112 Clinical Pembro X soft progression. 29 (SD) tissue Died of CHF in hospice 090720 V600R Ipi (PD) N/A Normal M1c Liver, 55 57 PD 55 Pembro (PD) Node Dabra/Tram (PD) Fludarabine/ cyclophospamide/ IL-2 (PD) Vemur/cobi (toxicity) 090729 V600E Adj 8 months Normal M1a Soft 444+ 444+ PR 444+ Interferon tissue TIL Vemurafenib Neoadj Pembro 090737 Wt Adj 4 months High M1a Renal 199 209 PR 358+ Interferon hilar Nivo/ipi + mass nivo Pelvic T-Vec mass Pembro Sub-cu TIL + high mass dose node IL-2 090738 V600K Dabra/Tram 14 months Normal M1c Nodes, 201 225 SD 243 Death not Pembro 2 liver related to years Ipi/ study pembro X4 treatment Pembro 090739 unknown Pembro (SD) N/A Normal M1a Soft 103 113 SD 133+ Pt withdrew Pembro + tissue from study ipi (SD) nodules to pursue other treatment
Example 4
[0319] The tolerability of the compounds of formula I, nivolumab, and a combination of the compounds of formula I and nivolumab were tested. The Phase 2 clinical dosage of 30 mg BIW of the compounds of formula I was administered. Among 63 subjects with adverse event ("AE") data available, Treatment Emergent Adverse Event ("TEAE"), 52% were considered related to treatment ("TRAE"). Among TRAEs, 47% were associated with the combination of the compounds of formula I and nivolumab ("NIVO"), 39% compounds of formula I alone, and 14% NIVO alone. Less frequent TRAEs associated with NIVO alone were lipase increase (n=6), rash (n=10), TSH increase (n=5), amylase increase (n=4). Further, among Grade 3 AEs, only fatigue, headache, diarrhea, nausea, vomiting were symptomatic--others were asymptomatic. Grade 3 fatigue in one subject responded to a low dose oral steroid. The fatigue in another subject was resolved by withholding drug only--no intervention necessary. Grade 3 diarrhea was resolved with an over counter drug. Grade 3 headache responded to over counter drug and did not recur on subsequent dosings. In addition, nausea and vomiting responded to oral drug. No AE was difficult to manage in oncology practice, and none caused medical concerns by investigators. Other asymptomatic AEs were abnormalities of blood test, most did not require treatments. When counting AEs with symptoms, only 4 out of 20 subjects experienced clinically significant AEs on nivo+compounds of formula I. The findings of this study are summarized in Table 9 below.
TABLE-US-00011 TABLE 9 Tolerability Profile HBI-8000 + NIVO TRAE .gtoreq. 5% frequency and number of subject n (% of N) Association Compounds of Compounds of Formula I + NIVO Formula I NIVO AE Term G1-2 G3 G4 G1-2 G3 G4 G1-2 G3 G4 Fatigue 24 (38) 4 (6) 0 0 0 0 0 0 0 Diarrhea 19 (30) 3 (5) 0 0 0 0 4 (6) 1 (2) 0 Lymphocyte decrease 6 (10) 1 (2) 0 0 0 0 0 0 0 Neutrophil decrease 4 (6) 2 (3) 0 8 (13) 5 (8) 1 (2) 0 0 0 WBC decrease 6 (10) 1 (2) 0 11 (17) 1 (2) 0 0 0 0 Platelet decrease 10 (16) 0 0 23 (37) 3 (5) 0 0 0 0 Anemia 0 0 0 14 (22) 1 (2) 0 0 0 0 Hypophospatemia 0 0 0 6 (10) 4 (6) 0 0 0 0
Example 5
[0320] Immune gene activation in response to administration of the compounds of formula I, a PD-1 inhibitory antibody, and a combination of the compounds of formula I and a PD-1 inhibitory antibody was examined using an MC38 tumor model. The results are summarized in FIG. 6A. These results show that the combination of the compounds of formula I and a PD-1 inhibitory antibody synergistically activated immune gene expression in the tumor microenvironment. FIG. 6B summarizes the improvement on survival amongst the experimental group treated with the combination therapy compared to the compounds of formula I alone or the PD-1 inhibitory antibody alone.
Example 6
[0321] The compounds of formula I were used as a monotherapy for relapsed or refractory peripheral T-cell lymphoma ("RR/PTCL"). A dosage of 40 mg biw was approved for this study. A summary of the results is found in Table 10. These results were compared to efficacy/history benchmarks for several known treatments for RR/PTCL (see Table 11). The time after prior treatment was 96 days (vs. 222 days for romidepsin P2 trial). The estimated PFS and OS of the experiment are found in FIGS. 7A-B. For these results, the PD date was due to investigator judgement. The median PFS (months) was 7.6.
TABLE-US-00012 TABLE 10 Example 6 Results Summary Patients Numbers Phase 2 Enrolled Total 55 Evaluable Total 43 Complete Responses 4 Partial Responses 16 Stable Disease 12 ORR 46% DCR 74% Pharmaceuticals and Medical 30% Devices Agency ("PMDA") Target ORR
TABLE-US-00013 TABLE 11 Efficacy vs. Historical Benchmarks for RR/PTCL Treatments RR/PTCL Treatment ORR Epidaza (China) (PMS) 28% (35%) Forodesine 22% Pralatrexate 45% Romidepsin 43%
Example 7
[0322] The compounds of formula I were used as a monotherapy for relapsed or refractory aggressive adult T-cell lymphoma ("RR/ATL"). A dosage of 40 mg biw was approved for this study. A summary of the results is found in Table 12. These results were compared to efficacy/history benchmarks for several known treatments for RR/ATL (see Table 13). The time after prior treatment was 88 days (vs. 234 days for lenalidomide P2 trial). All patients had received mogamulizumab ("moga") (lenalidomide ORR in moga patients was 18%).
TABLE-US-00014 TABLE 12 Example 7 Results Summary Patients Numbers Phase 2 Enrolled Total 23 Evaluable Total 20 Partial Responses 8 Stable Disease 4 ORR 40% DCR 60% Pharmaceuticals and 30% Medical Devices Agency ("PMDA") Target ORR
TABLE-US-00015 TABLE 13 Efficacy vs. Historical Benchmarks for RR/ATL Treatments RR/PTCL Treatment ORR Compounds of Formula I Phase I 75% Mogamulizumab 50% Lenalidomide 42%
[0323] While preferred embodiments of the present invention have been shown and described herein, it will be obvious to those skilled in the art that such embodiments are provided by way of example only. Numerous variations, changes, and substitutions will now occur to those skilled in the art without departing from the invention. It should be understood that various alternatives to the embodiments of the invention described herein may be employed in practicing the invention. It is intended that the following claims define the scope of the invention and that methods and structures within the scope of these claims and their equivalents be covered thereby.
* * * * *
References













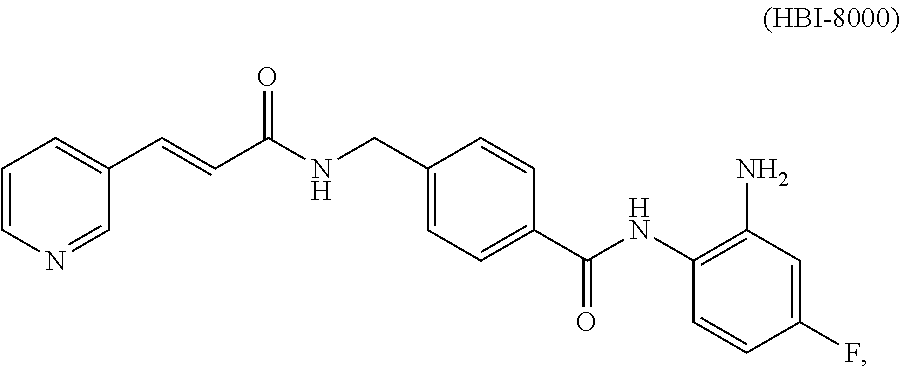
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.