Method Of Treating Cancer Using Selective Estrogen Receptor Modulators
Wardell; Suzanne E. ; et al.
U.S. patent application number 17/558731 was filed with the patent office on 2022-04-14 for method of treating cancer using selective estrogen receptor modulators. The applicant listed for this patent is Duke University. Invention is credited to Donald P McDonnell, Erik R. Nelson, Suzanne E. Wardell.
| Application Number | 20220110893 17/558731 |
| Document ID | / |
| Family ID | |
| Filed Date | 2022-04-14 |











View All Diagrams
| United States Patent Application | 20220110893 |
| Kind Code | A1 |
| Wardell; Suzanne E. ; et al. | April 14, 2022 |
METHOD OF TREATING CANCER USING SELECTIVE ESTROGEN RECEPTOR MODULATORS
Abstract
Disclosed herein are methods of treating subjects suffering from estrogen receptor positive cancer of the brain by administering a selective estrogen receptor degrader (SERM). Also disclosed are methods of treating a cancer that is resistant to an estrogen receptor modulator by administering a SERM.
| Inventors: | Wardell; Suzanne E.; (Durham, NC) ; Nelson; Erik R.; (Champaign, IL) ; McDonnell; Donald P; (Chapel Hill, NC) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Appl. No.: | 17/558731 | ||||||||||
| Filed: | December 22, 2021 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16549828 | Aug 23, 2019 | |||
| 17558731 | ||||
| 15129197 | Sep 26, 2016 | 10420734 | ||
| PCT/US2015/023216 | Mar 28, 2015 | |||
| 16549828 | ||||
| 14512061 | Oct 10, 2014 | 9421264 | ||
| 15129197 | ||||
| 62129379 | Mar 6, 2015 | |||
| 61971627 | Mar 28, 2014 | |||
| 61971627 | Mar 28, 2014 | |||
| International Class: | A61K 31/137 20060101 A61K031/137; A61K 9/00 20060101 A61K009/00; A61K 31/136 20060101 A61K031/136; A61K 31/138 20060101 A61K031/138; A61K 31/40 20060101 A61K031/40; A61K 31/4196 20060101 A61K031/4196; A61K 31/4535 20060101 A61K031/4535; A61K 31/565 20060101 A61K031/565; A61K 31/5685 20060101 A61K031/5685; A61K 45/06 20060101 A61K045/06; C07C 217/78 20060101 C07C217/78; C07C 217/84 20060101 C07C217/84 |
Goverment Interests
STATEMENT OF GOVERNMENT INTEREST
[0002] This invention was made with government support under R37DK048807 awarded by the National Institutes of Health. The government has certain rights in the invention.
Claims
1. A method of treating an estrogen receptor positive breast cancer in a subject, wherein the estrogen receptor positive breast cancer is resistant to an estrogen receptor modulator, the method comprising administering a composition comprising a compound represented by the following formula I: ##STR00022## wherein TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C.sub.1-C.sub.6 alkyl group; A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; ring G represents the following formula: ##STR00023## R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; a partial structure in formula (I) represented by the following formula: ##STR00024## R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and L represents a single bond.
2. The method of claim 1, wherein the estrogen receptor positive breast cancer is de novo resistant to the estrogen receptor modulator.
3. The method of claim 1, wherein the resistance to the estrogen receptor modulator is acquired.
4. The method of claim 1, wherein the estrogen receptor modulator is a selective estrogen receptor modulator (SERM).
5. The method of 4, wherein the SERM is tamoxifen, idoxifene, raloxifene or ICI 182,780.
6. The method of claim 1, wherein R.sup.a and R.sup.b independently represent a hydrogen atom, a methyl group, an ethyl group, a n-propyl group, an iso-propyl group, a n-butyl group, an iso-butyl group, or a tert-butyl group.
7. The method of claim 1, wherein -T-Z-- represents --CH.sub.2CH.sub.2-- or --C(CH.sub.3).sub.2CH.sub.2O--.
8. The method of claim 1, wherein Y represents --CH.sub.2--N(CH.sub.2CH.sub.3)-- or --CH.sub.2--N(CH.sub.2CH.sub.2OH)--.
9. The method of claim 1, wherein each of R' independently represents a hydrogen atom or a methoxy group.
10. The method of claim 1, wherein R'' represents a hydroxyl group.
11. The method of claim 1, wherein A represents a phenylene group.
12. The method of claim 1, wherein an effective amount of the compound is administered.
13. The method of claim 12, wherein the effective amount comprises a high dosage.
14. The method of claim 1, wherein the compound is administered by oral administration, intravenous administration, intradermal injection, intramuscular injection, or subcutaneous injection.
15. The method of claim 1, further comprising administering an effective amount of at least one compound selected from the group consisting of a cyclin-dependent kinase 4 and 6 inhibitor (CDK4/6 inhibitor), an antiestrogen, a ligand of retinoic acid or retinoxic X receptor, an antiprogestin, an antiandrogen, vitamin D or metabolite thereof, a farnesyl transferase inhibitor, a PPAR.alpha. or gamma agonist and a MAP kinase inhibitor.
16. The method of claim 13, wherein the high dosage is more than about 20 mg/kg.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This patent application is a continuation U.S. patent application Ser. No. 16/549,828, filed Aug. 23, 2019, which is a continuation of U.S. patent application Ser. No. 15/129,197, filed Sep. 26, 2016, issued Sep. 24, 2019 as U.S. Pat. No. 10,420,734, which is the national stage filing under 35 U.S.C. .sctn. 371 of International Application No. PCT/US2015/023216, filed on Mar. 28, 2015, which application claims priority to U.S. Provisional Application No. 62/129,379, filed Mar. 6, 2015, and is a continuation-in-part of U.S. patent application Ser. No. 14/512,061, filed Oct. 10, 2014, issued as U.S. Pat. No. 9,421,264, on Aug. 23, 2016, which application claims priority to U.S. Provisional Application No. 61/971,627, filed Mar. 28, 2014, the entire contents of each of which are hereby incorporated by reference.
TECHNICAL FIELD
[0003] The present invention relates to methods of treating subjects suffering from estrogen receptor positive cancer of the brain by administering a selective estrogen receptor modulator (SERM) to the subject. The present invention also relates to methods of treating subjects suffering from a cancer that is resistant to an estrogen receptor modulator by administering a SERM to the subject.
BACKGROUND
[0004] The estrogen receptor (ER) is a ligand dependent transcription factor whose expression confers upon target cells the ability to respond to estrogens. In the absence of an activating ligand, ER resides in the cell in an inactive form within a large inhibitory protein complex. Upon binding ligand, however, the receptor undergoes an activating conformational change resulting in its release from the inhibitory protein complex, spontaneous dimerization and subsequent interaction with enhancers located within target genes. Depending on the promoter context of the bound receptor, and the co factors that are recruited to the receptor in a particular cell, it can either positively or negatively regulate target gene transcription. Thus, the same ER-ligand complex can have very different activities in different cells, an observation that explains how estrogens, generally considered to be reproductive hormones, exhibit activities in bone, the cardiovascular system and in brain that are unrelated to reproductive function.
[0005] Whereas the molecular determinants of ER action differ considerably between target cells, it has been anticipated that the exploitation of this complexity will yield pharmaceuticals with process or tissue selective activities. The first evidence in support of this hypothesis came from studies that probed the pharmacological activities of the `antiestrogen` tamoxifen. Identified as a high affinity antagonist of ER and developed as a treatment for ER-positive breast cancer, it soon became apparent that whereas tamoxifen could oppose estrogen action in the breast it exhibited agonist activity in the bone, uterus and in the cardiovascular system. Reflecting this spectrum of activities, tamoxifen was reclassified as a Selective Estrogen Receptor Modulator (SERM).
[0006] Breast cancer remains the most commonly diagnosed cancer among women and a leading cause of cancer mortality. While targeted therapies such as the SERM tamoxifen and aromatase inhibitors are initially effective in the treatment of estrogen receptor alpha (ESR1) positive tumors, de novo and acquired resistance remain an impediment to durable clinical responses, particularly in the setting of advanced disease. ESR1 is a therapeutic target in breast cancers that are resistant to both first and second line endocrine interventions, a finding that has prompted the development of (a) SERMs with a mechanism distinct from tamoxifen and (b) selective estrogen receptor degraders (SERDs), competitive antagonists whose interaction with ESR1 induce its proteasome dependent degradation. Fulvestrant, a SERD, has been effective as both a first- and second-line therapy in advanced breast cancer; however, the pharmaceutical properties of this drug may prove dose-limiting in relapsed/resistant breast tumors bearing ESR1 mutations known to decrease SERD potency. The clinical efficacy of fulvestrant, a Selective Estrogen Receptor Degrader (SERD) that triggers receptor degradation, has confirmed that ESR1 often remains engaged in endocrine therapy resistant cancers.
[0007] The increasing incidence of breast cancer brain metastases (BCBM) is an emerging challenge in the treatment of advanced breast cancer patients. The growing success of improved treatments of systemic disease has allowed the manifestation of BCBM that previously would not have impacted the morbidity and mortality associated with breast cancer. The privileged environment of the brain, maintained by the relatively non-porous blood brain barrier, presents a significant impediment to the successful targeting of BCBM, leading to the use of gamma knife surgery and/or whole brain radiation in an attempt to shrink or ablate brain lesions. The benefit of these treatments must be carefully balanced with neurological deficit as a result of treatment.
[0008] Although considerable advances have been made in targeting the estrogen signaling axis for the treatment of breast cancer and osteoporosis, similar progress has unfortunately not yet been accomplished in the development of safe and effective treatments for the climacteric conditions or vasomotor disturbances that are associated with estrogen deprivation. There is considerable interest in developing novel SERMs that can be used to treat vasomotor symptoms but which do not exhibit mitogenic activities in the breast or the uterus.
[0009] While tamoxifen and aromatase inhibitors have proven effective in the treatment of estrogen receptor positive (ER+) breast cancer, the incidence of resistance remains significant, particularly in the advanced/metastatic breast cancer setting. An additional class of estrogen receptor targeting therapy, selective estrogen receptor degraders (SERDs), has recently come to prominence. These agents have proven effective in pre-clinical models of breast cancers that are resistant to tamoxifen or aromatase inhibitors, leading to their evaluation in clinical trials. However, these agents also do not readily pass the blood brain barrier, suggesting that they will be ineffective in targeting BCBM. It would be beneficial to have other treatment options that can penetrate the blood brain barrier and/or selectively target tissue specific activities responsive to ER activation.
SUMMARY
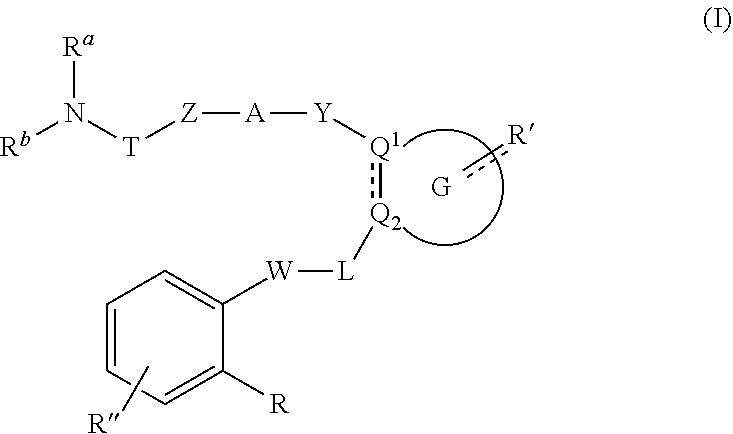
[0010] The present invention is directed to a method of treating estrogen receptor positive cancers of the brain in a subject. The method comprises administering a compound represented by the following formula I:
##STR00001##
wherein [0011] TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C1-C6 alkyl group; [0012] A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; [0013] Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; [0014] ring G represents the following formula:
[0014] ##STR00002## [0015] R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; [0016] a partial structure in formula (I) represented by the following formula:
[0016] ##STR00003## [0017] R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; and [0018] R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and [0019] L represents a single bond, or a salt thereof.
[0020] The cancer may be Breast cancer brain metastases, Astrocytoma, Atypical Teratoid Rhabdoid Tumor (ATRT), Chondrosarcoma, Choroid Plexus Carcinoma, Craniopharyngioma, Ependymoma, Germ Cell Tumor, Glioblastoma, Glioma, Hemangioma, Juvenile Pilocytic Astrocytoma, Medulloblastoma, Meningioma, Neurofibroma, Neuronal and Mixed Neuronal-Glial Tumors, Oligoastrocytoma, Oligodendroglioma, Pineal Tumor, Pituitary Tumor, PNET--(primitive neuroectodermal tumor), Schwannoma, and Leptomeningeal metastases. Ra and Rb independently may represent a hydrogen atom, a methyl group, an ethyl group, a n-propyl group, an iso-propyl group, a n-butyl group, an iso-butyl group, or a tert-butyl group. -T-Z-- may represents --CH.sub.2CH.sub.2-- or --C(CH.sub.3).sub.2CH.sub.2O--. Y may represent --CH.sub.2--N(CH.sub.2CH.sub.3)-- or --CH.sub.2--N(CH.sub.2CH.sub.2OH)--. Each of R'' independently may represents a hydrogen atom or a methoxy group. R'' may represents a hydroxyl group. A may represents a phenylene group. The compound may be (R)-6-{2-{ethyl[4-(2-ethylaminoethyl)benzyl]amino}-4-methoxyphenyl}-5,6,7- ,8-tetrahydronaphthalen-2-ol. An effective amount of the compound may be administered. The effective amount may comprise a high dosage. The high dosage may be more than about 20 mg/kg. The high dosage may be about 20 mg/kg to about 100 mg/kg. The compound may be administered by oral administration, intravenous administration, intradermal injection, intramuscular injection, or subcutaneous injection. The method may further comprising administering an effective amount of at least one compound selected from the group consisting of a cyclin-dependent kinase 4 and 6 inhibitor (CDK4/6 inhibitor), an antiestrogen, a ligand of retinoic acid or retinoxic X receptor, an antiprogestin, an antiandrogen, vitamin D or metabolite thereof, a farnesyl transferase inhibitor, a PPAR.alpha. or gamma agonist and a MAP kinase inhibitor.
[0021] The present invention is directed to a method of treating breast cancer brain metastasis in a subject. The method comprises administering a compound represented by the following formula I:
##STR00004##
wherein [0022] TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C.sub.1-C.sub.6 alkyl group; [0023] A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; [0024] Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; [0025] ring G represents the following formula:
[0025] ##STR00005## [0026] R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; [0027] a partial structure in formula (I) represented by the following formula:
[0027] ##STR00006## [0028] R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; and [0029] R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and [0030] L represents a single bond, or a salt thereof.
[0031] R.sup.a and R.sup.b independently may represent a hydrogen atom, a methyl group, an ethyl group, a n-propyl group, an iso-propyl group, a n-butyl group, an iso-butyl group, or a tert-butyl group. -T-Z-- may represents --CH.sub.2CH.sub.2-- or --C(CH.sub.3).sub.2CH.sub.2O--. Y may represent --CH.sub.2--N(CH.sub.2CH.sub.3)-- or --CH.sub.2--N(CH.sub.2CH.sub.2OH)--. Each of R'' independently may represents a hydrogen atom or a methoxy group. R'' may represents a hydroxyl group. A may represents a phenylene group. The compound may be (R)-6-{2-{ethyl[4-(2-ethylaminoethyl)benzyl]amino}-4-methoxyphenyl}-5,6,7- ,8-tetrahydronaphthalen-2-ol. An effective amount of the compound may be administered. The effective amount may comprise a high dosage. The high dosage may be more than about 20 mg/kg. The high dosage may be about 20 mg/kg to about 100 mg/kg. The compound may be administered by oral administration, intravenous administration, intradermal injection, intramuscular injection, or subcutaneous injection. The method may further comprising administering an effective amount of at least one compound selected from the group consisting of a cyclin-dependent kinase 4 and 6 inhibitor (CDK4/6 inhibitor), an antiestrogen, a ligand of retinoic acid or retinoxic X receptor, an antiprogestin, an antiandrogen, vitamin D or metabolite thereof, a farnesyl transferase inhibitor, a PPAR.alpha. or gamma agonist and a MAP kinase inhibitor.
[0032] The present invention is directed to a method of treating a cancer in a subject, wherein the cancer is resistant to an estrogen receptor modulator. The method comprises administering a compound represented by the following formula I:
##STR00007##
wherein [0033] TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C.sub.1-C.sub.6 alkyl group; [0034] A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; [0035] Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; [0036] ring G represents the following formula:
[0036] ##STR00008## [0037] R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; [0038] a partial structure in formula (I) represented by the following formula:
[0038] ##STR00009## [0039] R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; and [0040] R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and [0041] L represents a single bond, or a salt thereof.
[0042] The cancer may be de novo resistant to the estrogen receptor modulator. The resistance to the estrogen receptor modulator may be acquired. The estrogen receptor modulator may be a selective estrogen receptor modulator (SERM). The SERM may be tamoxifen, idoxifene, raloxifene or ICI 182,780. The cancer may be breast, endometrial or ovarian cancer. The cancer may be breast cancer. R.sup.a and R.sup.b independently may represent a hydrogen atom, a methyl group, an ethyl group, a n-propyl group, an iso-propyl group, a n-butyl group, an iso-butyl group, or a tert-butyl group. -T-Z-- may represents --CH.sub.2CH.sub.2-- or --C(CH.sub.3).sub.2CH.sub.2O--. Y may represent --CH.sub.2--N(CH.sub.2CH.sub.3)-- or --CH.sub.2--N(CH.sub.2CH.sub.2OH)--. Each of R'' independently may represents a hydrogen atom or a methoxy group. R'' may represents a hydroxyl group. A may represents a phenylene group. The compound may be (R)-6-{2-{ethyl[4-(2-ethylaminoethyl)benzyl]amino}-4-methoxyphenyl}-5,6,7- ,8-tetrahydronaphthalen-2-ol. An effective amount of the compound may be administered. The effective amount may comprise a high dosage. The high dosage may be more than about 20 mg/kg. The high dosage may be about 20 mg/kg to about 100 mg/kg. The compound may be administered by oral administration, intravenous administration, intradermal injection, intramuscular injection, or subcutaneous injection. The method may further comprising administering an effective amount of at least one compound selected from the group consisting of a cyclin-dependent kinase 4 and 6 inhibitor (CDK4/6 inhibitor), an antiestrogen, a ligand of retinoic acid or retinoxic X receptor, an antiprogestin, an antiandrogen, vitamin D or metabolite thereof, a farnesyl transferase inhibitor, a PPAR.alpha. or gamma agonist and a MAP kinase inhibitor.
BRIEF DESCRIPTION OF THE DRAWINGS
[0043] The patent or application file contains at least one drawing executed in color. Copies of this patent or patent application publication with color drawing(s) will be provided by the Office upon request and payment of the necessary fee.
[0044] FIGS. 1A-1B show treatment with (R)-6-{2-{ethyl[4-(2-ethylaminoethyl)benzyl]amino}-4-methoxyphenyl}-5,6,7- ,8-tetrahydronaphthalen-2-ol ("RAD1901") results in a dose dependent reduction in ER expression and activity. FIG. 1A shows MCF7 breast cancer cells were treated 4 hours as indicated prior to western blot detection of ER and loading control cytokeratin (CK) 18. FIG. 1B shows ovariectomized mice bearing MCF7 xenograft tumors were treated daily with RAD1901 ("RAD") or tamoxifen ("Tam") in the context of continued estrogen treatment.
[0045] FIGS. 2A-2B show the mechanism by which RAD1901 downregulates ER expression. FIG. 2A shows ER.alpha. protein expression in whole cell extracts pre-treated with transcription or translation inhibitors before treatment with RAD1901, analyzed by immunoblot. FIG. 2B shows ER.alpha. mRNA expression in similarly treated cells.
[0046] FIG. 3 shows the interaction between ER and conformation-specific peptides in mammalian two-hybrid system.
[0047] FIGS. 4A-4B show RAD1901 exhibits dose dependent agonist/antagonist regulation of ER transactivation of target genes.
[0048] FIGS. 5A-5F show that RAD1901 inhibited ESR1 activity in vitro and in vivo.
[0049] FIGS. 6A-6D show that RAD1901 downregulated ESR1 expression through receptor degradation.
[0050] FIGS. 7A-7D show that the effects of RAD1901 on BT483 breast cancer cells were similar to those observed in MCF7 cells.
[0051] FIGS. 8A-8C show that the in vivo pharmacology of RAD1901 was influenced by SERD activity.
[0052] FIGS. 9A-9E show that RAD1901 exhibited dose dependent growth stimulation of MCF7 xenograft tumors.
[0053] FIGS. 10A-10E show that RAD1901 exerted biphasic agonist/antagonist activity on ESR1 in a dose dependent manner.
[0054] FIG. 11 shows the relative change in expression of ESR1 target genes and additional target genes responsive to agonists, primarily SERMs, or SERMs and agonists.
DETAILED DESCRIPTION
[0055] The present disclosure provides a method of treating a subject suffering from estrogen receptor positive cancer of the brain, such as BCBM, or a cancer that is resistant to an estrogen receptor modulator, such as tamoxifen resistant breast cancer. The methods involve administering to the subject a SERM, such as RAD1901. RAD1901 is a SERM/SERD hybrid that exhibits complex and unique pharmacology in breast cancer models, having dose-dependent agonist/antagonist activity displayed in a tissue-selective manner. RAD1901 exhibits desired pharmacological activities and exhibits significant brain penetrance when evaluated in postmenopausal women, in particular a unique dose response, with lower doses of the drug being more effective at relieving hot flashes. RAD1901 induces hot flashes in healthy postmenopausal women in a dose dependent manner, thus RAD1901 may effectively inhibit estrogen receptor action in the brain.
[0056] Turnover of ER.alpha. is significantly increased upon binding RAD1901, an activity that is more pronounced at higher drug concentrations (FIG. 1). This drug exhibits some of the characteristics that are generally attributed to selective estrogen receptor degraders (SERDs). Thus, at lower doses RAD1901 exhibits partial agonist activity, i.e., SERM activity, allowing for relief of hot flashes, but the SERD activity of the compound dominates when the receptor is exposed to higher concentrations. The present disclosure describes in vitro the mechanism by which RAD1901 impacts ER expression and investigates the possible result of such action in vivo.
[0057] As exemplified below, RAD1901 surprisingly has the ability to degrade the estrogen receptor. Both in vitro and in vivo studies have since shown that the antagonist activity of this ligand correlates with estrogen receptor degradation in a dose dependent manner. RAD1901 also inhibits estrogen dependent growth of breast cancer xenograft tumors and may be used to treat breast cancer, such as tamoxifen resistant breast cancer.
[0058] RAD1901 is unique among both SERMs and SERDs in that this drug accumulates in the brain, an environment in which SERM penetration has been historically regarded as quite low. Estrogen receptor activity has been found to be important in the growth of tumors resistant to aromatase inhibitors and/or tamoxifen, and treatment with SERDs has been shown to have clinical benefit. While the revelation that RAD1901 exhibits SERD activity certainly suggests potential utility in the treatment of progressing ER+ breast tumors, which was unappreciated prior to the present disclosure, the targeting of ER activity for the treatment of ER+ brain cancers represents a new frontier for the use of both SERMs and SERDs, as RAD1901 represents the first SERM/SERD that can sufficiently penetrate the brain to exhibit efficacy. Because BCBM is generally diagnosed late in the disease progression of ER+ metastatic breast cancer, patients will in general have already been treated with endocrine therapeutics (i.e. tamoxifen or aromatase inhibitors). Thus, while SERMs such as tamoxifen have low brain penetration and have exhibited efficacy in anecdotal cases of BCBM that are detailed in the literature, the SERD activity of RAD1901 becomes key to the therapeutic potential of this compound for treatment of BCBM, as SERDs have been found to be effective in breast cancers that are resistant to SERM or aromatase inhibitor therapy.
1. Definitions
[0059] The terms "comprise(s)," "include(s)," "having," "has," "can," "contain(s)," and variants thereof, as used herein, are intended to be open-ended transitional phrases, terms, or words that do not preclude the possibility of additional acts or structures. The singular forms "a," "and" and "the" include plural references unless the context clearly dictates otherwise. The present disclosure also contemplates other embodiments "comprising," "consisting of" and "consisting essentially of," the embodiments or elements presented herein, whether explicitly set forth or not.
[0060] For the recitation of numeric ranges herein, each intervening number there between with the same degree of precision is explicitly contemplated. For example, for the range of 6-9, the numbers 7 and 8 are contemplated in addition to 6 and 9, and for the range 6.0-7.0, the number 6.0, 6.1, 6.2, 6.3, 6.4, 6.5, 6.6, 6.7, 6.8, 6.9, and 7.0 are explicitly contemplated.
[0061] Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art. In case of conflict, the present document, including definitions, will control. Preferred methods and materials are described below, although methods and materials similar or equivalent to those described herein can be used in practice or testing of the present invention. All publications, patent applications, patents and other references mentioned herein are incorporated by reference in their entirety. The materials, methods, and examples disclosed herein are illustrative only and not intended to be limiting.
[0062] The term "administration" or "administering," as used herein refers to providing, contacting, and/or delivery of the SERM by any appropriate route to achieve the desired effect. These agents may be administered to a subject in numerous ways including, but not limited to, orally, ocularly, nasally, intravenously, topically, as aerosols, suppository, etc. and may be used in combination.
[0063] "Aromatase inhibitor" as used herein refers to a compound that targets aromatase, which is an enzyme involved in the biosynthesis of estrogen. Aromatase inhibitors may block the production of estrogen or block the action of estrogen on receptors.
[0064] "Blood brain barrier" or "BBB" as used herein refers to a highly selective permeability barrier that separates the circulating blood from the brain extracellular fluid in the central nervous system. The blood brain barrier may prevent the certain drugs from entering brain tissue and is a limiting factor in the delivery of many peripherally-administered agents to the central nervous system.
[0065] "Breast cancer" as used herein refers to a type of cancer that originates from and develops in the breast. "Metastatic breast cancer" refers to breast cancer that spreads outside the breast to the lymph nodes, bones, or other areas.
[0066] "Breast Cancer Brain Metastases" and "BCBM" as used interchangeably herein refer to breast cancer that has metastasized to the brain. BCBM may occur in up to 10-15% of breast-cancer patients. BCBM may progress rapidly and can produce life-threatening complications such as increased intracranial pressure, herniation of the brain and seizures. Radiotherapy is a treatment of BCBM as it halts tumor progression quickly and can induce a response in the majority of patients.
[0067] "Cancer" as used herein refers to the uncontrolled and unregulated growth of abnormal cells in the body. Cancerous cells are also called malignant cells. Cancer may invade nearby parts of the body and may also spread to more distant parts of the body through the lymphatic system or bloodstream. Cancers include Adrenocortical Carcinoma, Anal Cancer, Bladder Cancer, Brain Tumor, Breast Cancer, Carcinoid Tumor, Gastrointestinal, Carcinoma of Unknown Primary, Cervical Cancer, Colon Cancer, Endometrial Cancer, Esophageal Cancer, Extrahepatic Bile Duct Cancer, Ewings Family of Tumors (PNET), Extracranial Germ Cell Tumor, Intraocular Melanoma Eye Cancer, Gallbladder Cancer, Gastric Cancer (Stomach), Extragonadal Germ Cell Tumor, Gestational Trophoblastic Tumor, Head and Neck Cancer, Hypopharyngeal Cancer, Islet Cell Carcinoma, Kidney Cancer (renal cell cancer), Laryngeal Cancer, Acute Lymphoblastic Leukemia, Leukemia, Acute Myeloid, Chronic Lymphocytic Leukemia, Chronic Myelogenous Leukemia, Hairy Cell Leukemia, Lip and Oral Cavity Cancer, Liver Cancer, Non-Small Cell Lung Cancer, Small Cell Lung Cancer, AIDS-Related Lymphoma, Central Nervous System (Primary) Lymphoma, Cutaneous T-Cell Lymphoma, Hodgkin's Disease Lymphoma, Non-Hodgkin's Disease Lymphoma, Malignant Mesothelioma, Melanoma, Merkel Cell Carcinoma, Metasatic Squamous Neck Cancer with Occult Primary, Multiple Myeloma and Other Plasma Cell Neoplasms, Mycosis Fungoides, Myelodysplastic Syndrome, Myeloproliferative Disorders, Nasopharyngeal Cancer, euroblastoma, Oral Cancer, Oropharyngeal Cancer, Osteosarcoma, Ovarian Epithelial Cancer, Ovarian Germ Cell Tumor, Pancreatic Cancer, Exocrine, Pancreatic Cancer, Islet Cell Carcinoma, Paranasal Sinus and Nasal Cavity Cancer, Parathyroid Cancer, Penile Cancer, Pituitary Cancer, Plasma Cell Neoplasm, Prostate Cancer, Rhabdomyosarcoma, Rectal Cancer, Renal Cell Cancer (cancer of the kidney), Transitional Cell Renal Pelvis and Ureter, Salivary Gland Cancer, Sezary Syndrome, Skin Cancer, Small Intestine Cancer, Soft Tissue Sarcoma, Testicular Cancer, Malignant Thymoma, Thyroid Cancer, Urethral Cancer, Uterine Cancer, Unusual Cancer of Childhood, Vaginal Cancer, Vulvar Cancer, and Wilms' Tumor.
[0068] The term "effective dosage" as used herein means a dosage of a drug effective for periods of time necessary, to achieve the desired therapeutic result. An effective dosage may be determined by a person skilled in the art and may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the drug to elicit a desired response in the individual. This term as used herein may also refer to an amount effective at bringing about a desired in vivo effect in an animal, mammal, or human, such as reducing and/or inhibiting the function of the estrogen receptor. A therapeutically effective amount may be administered in one or more administrations (e.g., the agent may be given as a preventative treatment or therapeutically at any stage of disease progression, before or after symptoms, and the like), applications or dosages and is not intended to be limited to a particular formulation, combination or administration route. It is within the scope of the present disclosure that the SERM may be administered at various times during the course of treatment of the subject. The times of administration and dosages used will depend on several factors, such as the goal of treatment (e.g., treating v. preventing), condition of the subject, etc. and can be readily determined by one skilled in the art.
[0069] "Estrogen dependent cancer" or "estrogen receptor positive cancer" as used interchangeably herein refers to a tumor that contains estrogen receptor (ER) positive cells, i.e., cells that have estrogen receptors, and respond to the presence of estrogen with increased proliferation. Estrogen dependent cancers may include breast cancer, ovarian cancer, or endometrial cancer. "Estrogen receptor positive breast cancer" is a type of breast cancer that is sensitive to estrogen.
[0070] "Estrogen receptor" or "ER" as used interchangeably herein refers to a receptor that is activated by the hormone estrogen and is a member of the nuclear hormone family of intracellular receptors. There are two different isoforms of estrogen receptor, referred to as .alpha. (also referred to as "ERa") and .beta. (also referred to as "ERb"). ERa and ERb genes are encoded by ESR1 and ESR2 gene, respectively. Hormone-activated estrogen receptors form dimers and may form homodimers or heterodimers. Both ERs are widely expressed in different tissue types.
[0071] "Estrogen-receptor downregulators" as used herein refers to a drug or compound which binds and down-regulates the expression of an estrogen-receptor.
[0072] "Estrogen receptor negative breast cancer" or "Estrogen independent breast cancer" as used interchangeably herein refers to a tumor that does not contain estrogen receptor positive cells, i.e., cells that lack estrogen receptors, and does not depend on the presence of estrogen for ongoing proliferation.
[0073] "HER2 intervention drug" or "HER2 inhibitor" as used interchangeably herein refers to a compound that targets human Epidermal Growth Factor Receptor 2 (HER2). HER2 is a member of the epidermal growth factor receptor family and is involved in the development and progression of certain aggressive types of breast cancer, such as estrogen dependent breast cancer. A HER2 inhibitor may be a tyrosine kinase or a monoclonal antibody.
[0074] "Metastatic cancer" as used herein refers to a cancer that has spread from the part of the body where it started (the primary site) to other parts of the body.
[0075] "Progesterone receptor positive cancer" as used herein refers to a tumor that contains progesterone receptor positive (PR+) cells, i.e., cells that have progesterone receptors, which respond to the presence of progesterone with increased proliferation.
[0076] "Selective estrogen receptor degraders" or "SERDs" as used interchangeably herein refers to a compound that interacts with an ER and induce a conformational change that results in the degradation of the receptor.
[0077] "Selective estrogen receptor modulators" or "SERMs" as used interchangeably herein refers to a compound that interacts with an ER and whose relative agonist/antagonist activities are manifest in a cell selective manner. The prevention of estrogen binding to the estrogen receptor may lead to decreased proliferation of estrogen dependent cancer cells.
[0078] The term "subject", "patient" or "subject in the method" as used herein interchangeably, means any vertebrate, including, but not limited to, a mammal (e.g., cow, pig, camel, llama, horse, goat, rabbit, sheep, hamsters, guinea pig, cat, dog, rat, and mouse, a non-human primate (for example, a monkey, such as a cynomolgus or rhesus monkey, chimpanzee, etc.) and a human).
[0079] In some embodiments, the subject or subject may be a human or a non-human. In some embodiments, the subject may be a human subject at risk for developing or already suffering from cancer.
[0080] "Tamoxifen resistant breast cancer" as used herein refers to a breast cancer that does respond to treatment with tamoxifen.
[0081] "Treat," "treating," or "treatment" are each used interchangeably herein to describe reversing, alleviating, or inhibiting the progress of a disease, or one or more symptoms of such disease, to which such term applies. Depending on the condition of the subject, the term also refers to preventing a disease, and includes preventing the onset of a disease, or preventing the symptoms associated with a disease. A treatment may be either performed in an acute or chronic way. The term also refers to reducing the severity of a disease or symptoms associated with such disease prior to affliction with the disease. Such prevention or reduction of the severity of a disease prior to affliction refers to administration of the SERM to a subject that is not at the time of administration afflicted with the disease. "Preventing" also refers to preventing the recurrence of a disease or of one or more symptoms associated with such disease. "Treatment" and "therapeutically," refer to the act of treating, as "treating" is defined above.
2. Methods of Treating Cancer with a SERM
[0082] The present invention is directed to methods of treating a subject suffering from cancer. The methods include administering a compound, i.e., SERM, having formula I:
##STR00010##
wherein [0083] TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C.sub.1-C.sub.6 alkyl group; [0084] A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; [0085] Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; [0086] ring G represents the following formula:
[0086] ##STR00011## [0087] R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; [0088] a partial structure in formula (I) represented by the following formula:
[0088] ##STR00012## [0089] R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; and [0090] R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and [0091] L represents a single bond, or a salt thereof.
[0092] In certain embodiments, R.sup.a and R.sup.b independently may represent a hydrogen atom, a methyl group, an ethyl group, a n-propyl group, an iso-propyl group, a n-butyl group, an iso-butyl group, or a tert-butyl group. -T-Z-- may represent --CH.sub.2CH.sub.2-- or --C(CH.sub.3).sub.2CH.sub.2O--. Y may represent --CH.sub.2--N(CH.sub.2CH.sub.3)-- or --CH.sub.2--N(CH.sub.2CH.sub.2OH)--. Each of R'' independently may represents a hydrogen atom or a methoxy group. R'' may represents a hydroxyl group. In certain embodiments, the compound may be (R)-6-{2-{ethyl[4-(2-ethylaminoethyl)benzyl]amino}-4-methoxyphenyl}-5,6,7- ,8-tetrahydronaphthalen-2-ol. Examples of other SERMS are described in U.S. Pat. No. 7,612,114, U.S. U.S. Pat. Nos. 7,960,412, 8,399,520, U.S. Patent Publication No. US 2009-0325930, and U.S. Patent Publication No. US 2006-0116364, the contents of which are incorporated by reference in their entirety. An effective amount of the compound may be administered.
(a) Dosages
[0093] In general, the dosage of administered SERM will vary depending upon such factors as the patient's age, weight, height, sex, general medical condition, and previous medical history. Typically, it is desirable to provide the recipient with a dosage of SERM, which is in the range of from about 1 pg/kg to 10 mg/kg (amount of agent/body weight of patient), although a lower or higher dosage also may be administered as circumstances dictate. Dosage regimens may be adjusted to provide the optimum desired response (e.g., a therapeutic or prophylactic response). For example, a single bolus may be administered, several divided doses may be administered over time or the dose may be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation. It is especially advantageous to formulate parenteral compositions in dosage unit form for ease of administration and uniformity of dosage. Dosage unit form as used herein refers to physically discrete units suited as unitary dosages for the mammalian subjects to be tested; each unit containing a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier. The specification for the dosage unit forms of the present invention are dictated by and directly dependent on (a) the unique characteristics of the active compound and the particular therapeutic or prophylactic effect to be achieved and (b) the limitations inherent in the art of compounding such an active compound for the treatment of sensitivity in individuals.
[0094] An exemplary, non-limiting range for a therapeutically or prophylactically effective amount of the SERM is a dose of between 0.1 and 200 mg/kg, for example between 0.1 and 10 mg/kg, or about 20 mg/kg to about 100 mg/kg. The therapeutically or prophylactically effective amount of the SERM may be between 1 and 200 mg/kg, 10 and 200 mg/kg, 20 and 200 mg/kg, 50 and 200 mg/kg, 75 and 200 mg/kg, 100 and 200 mg/kg, 150 and 200 mg/kg, 50 and 100 mg/kg, 5 and 10 mg/kg, or 1 and 10 mg/kg. It is to be noted that dosage values may vary with the type and severity of the condition to be alleviated.
[0095] In some embodiments, the SERM can be administered to a patient in an amount of about 10 mg/day to about 500 mg/day, about 10 mg/day to about 200 mg/day (e.g., 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, or 200 mg/day), 20 mg/day to about 100 mg/day, 100 mg/day to about 200 mg/day, or about 200 mg/day to about 500 mg/day (e.g., 190, 200, 210, 220, 230, 240, 250, 260, 270, 280, 290, 300, 310, 320, 330, 340, 350, 360, 370, 380, 390, 400, 410, 420, 430, 440, 450, 460, 470, 480, 490, 500, 510, 520, 530, 540, 550, 560, 570, 580, 590, 600, 610, 620, 630, 640, 650, 660, 670, 680, 690, or 700 mg/day), inclusive of any single or multi-dose daily administration regimen that falls within that total daily dose range. In some embodiments, the dose is from about 20 mg/day to about 100 mg/day. Additionally, one of ordinary skill in the art would also know how to adjust or modify variables such as dosage, dosage schedules, and routes of administration, as appropriate, for a given subject.
[0096] Further, the SERM dose may be determined by a person skilled in the art and may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the SERM to elicit a desired response in the individual. The dose is also one in which toxic or detrimental effects, if any, of the SERM are outweighed by the therapeutically beneficial effects. It is to be further understood that for any particular subject, specific dosage regimens should be adjusted over time according to the individual need and the professional judgment of the person administering or supervising the administration of the compositions, and that dosage ranges set forth herein are exemplary only and are not intended to limit the scope or practice of the claimed composition.
(b) Rad 1901
[0097] The SERM may be RAD 1901. Although RAD 1901 may effectively treat vasomotor symptoms, its pharmacology is complex. Treatment of ER positive breast cancer cells with RAD 1901 in MCF7 and BT483 cells resulted in a pronounced dose dependent down regulation of the receptor expression. RAD 1901 is a unique SERM in that it apparently has a relative agonist/antagonist activity in the brain that is determined by dose. At low doses RAD 1901, behaves as a SERM as it exhibits estrogenic activity. At high doses, RAD 1901 may function as a SERD, reversing the response.
[0098] While targeting of the estrogen signaling axis has proven effective in the treatment of breast cancer and osteoporosis, implementing a safe therapy that mitigates the vasomotor. While efforts are being made to address this unmet medical need using tissue specific estrogen complexes (TSECs) that combine estrogens and SERMs with the intention of inhibiting estrogen action only in some tissues (i.e., breast and uterus), preliminary clinical data suggest that RAD 1901 may accomplish the same medical goal without exposing the patient to estrogen. The apparent dose dependent down regulation of ER by RAD1901 suggest that at a therapeutic (low) dose, RAD1901 may be mediating some level of agonist activity, while a higher dose results in more extensive SERM activity and an effective blockade in estrogen signaling, thereby exacerbating vasomotor symptoms.
[0099] The binding of RAD1901 enables the presentation of protein-protein interaction surfaces within the hinge region (flexible structure linking the DNA and hormone binding domains) of ESR1 that are involved with transcriptional activity. Identification of the coregulators that interact with these surfaces on ESR1 enable a definition of their importance in RAD1901 pharmacology. As the occupancy of the receptor increases, it is targeted for degradation, which results in a quantitative inhibition of ESR1 signaling. This pharmacological profile resembles that of a classical agonist, such as 17.beta.-estradiol, where the agonist signal is terminated by proteasome-dependent receptor degradation. The pharmacological actions of RAD1901 represent an uncoupling of ESR1-dependent transcriptional activation from degradation. At appropriate doses, the SERD activity manifested by RAD1901 may result in useful clinical activity in breast cancer.
[0100] As disclosed herein, RAD1901 inhibited estrogen activation of ESR1 in vitro and in vivo, inhibited estrogen-dependent breast cancer cell proliferation and xenograft tumor growth, and mediated dose-dependent downregulation of ESR1 protein. Doses of RAD1901 that were insufficient to induce ESR1 degradation were shown to result in activation of ESR1 target genes and in stimulation of xenograft tumor growth. RAD1901 may be used as targeted therapy for the treatment of breast cancer brain metastases.
[0101] In some embodiments, a low dose of RAD 1901 may be about 0 mg/kg to about 25 mg/kg, about 0 mg/kg to about 20 mg/kg, about 0 mg/kg to about 15 mg/kg, about 0 mg/kg to about 10 mg/kg, about 0 mg/kg to about 5 mg/kg, about 1 mg/kg to about 25 mg/kg, about 1 mg/kg to about 20 mg/kg, about 1 mg/kg to about 15 mg/kg, about 1 mg/kg to about 10 mg/kg, about 1 mg/kg to about 5 mg/kg, about 2 mg/kg to about 25 mg/kg, about 2 mg/kg to about 20 mg/kg, about 2 mg/kg to about 15 mg/kg, about 2 mg/kg to about 10 mg/kg, about 2 mg/kg to about 5 mg/kg, about 3 mg/kg to about 25 mg/kg, about 3 mg/kg to about 20 mg/kg, about 3 mg/kg to about 15 mg/kg, about 3 mg/kg to about 10 mg/kg, about 3 mg/kg to about 5 mg/kg, about 4 mg/kg to about 25 mg/kg, about 4 mg/kg to about 20 mg/kg, about 4 mg/kg to about 15 mg/kg, about 4 mg/kg to about 10 mg/kg, about 4 mg/kg to about 5 mg/kg, about 5 mg/kg to about 25 mg/kg, about 5 mg/kg to about 20 mg/kg, about 5 mg/kg to about 15 mg/kg, about 5 mg/kg to about 10 mg/kg, about 5 mg/kg to about 7.5 mg/kg. In some embodiments, a low dose of RAD 1901 may be less than about 25 mg/kg, about 24 mg/kg, about 23 mg/kg, about 22 mg/kg, about 21 mg/kg, about 20 mg/kg, about 19 mg/kg, about 18 mg/kg, about 17 mg/kg, about 16 mg/kg, about 15 mg/kg, about 14 mg/kg, about 13 mg/kg, about 12 mg/kg, about 11 mg/kg, about 10 mg/kg, about 9 mg/kg, about 8 mg/kg, about 7 mg/kg, about 6 mg/kg, about 5 mg/kg, about 4 mg/kg, about 3 mg/kg, about 2 mg/kg, or about 1 mg/kg.
[0102] In some embodiments, a high dose of RAD 1901 may be about 15 mg/kg to about 500 mg/kg, about 15 mg/kg to about 250 mg/kg, about 15 mg/kg to about 200 mg/kg, about 15 mg/kg to about 150 mg/kg, about 15 mg/kg to about 100 mg/kg, about 15 mg/kg to about 75 mg/kg, about 20 mg/kg to about 500 mg/kg, about 20 mg/kg to about 250 mg/kg, about 20 mg/kg to about 200 mg/kg, about 20 mg/kg to about 150 mg/kg, about 20 mg/kg to about 100 mg/kg, about 20 mg/kg to about 75 mg/kg, about 25 mg/kg to about 500 mg/kg, about 25 mg/kg to about 250 mg/kg, about 25 mg/kg to about 200 mg/kg, about 25 mg/kg to about 150 mg/kg, about 25 mg/kg to about 100 mg/kg, or about 25 mg/kg to about 75 mg/kg. In some embodiments, a high dose of RAD 1901 may be more than about 15 mg/kg, 20 mg/kg, about 25 mg/kg, about 30 mg/kg, about 35 mg/kg, about 40 mg/kg, about 45 mg/kg, about 50 mg/kg, about 55 mg/kg, about 60 mg/kg, about 65 mg/kg, about 70 mg/kg, about 75 mg/kg, about 80 mg/kg, about 85 mg/kg, about 90 mg/kg, about 95 mg/kg, about 100 mg/kg, about 105 mg/kg, about 110 mg/kg, about 115 mg/kg, about 120 mg/kg, about 125 mg/kg, about 130 mg/kg, about 135 mg/kg, about 140 mg/kg, about 145 mg/kg, about 150 mg/kg, about 155 mg/kg, about 160 mg/kg, about 165 mg/kg, about 170 mg/kg, about 175 mg/kg, about 180 mg/kg, about 185 mg/kg, about 190 mg/kg, about 195 mg/kg, about 200 mg/kg, about 250 mg/kg, about 300 mg/kg, about 350 mg/kg, about 400 mg/kg, about 450 mg/kg or about 500 mg/kg.
[0103] In some embodiments, RAD1901 can be administered to a patient in an amount of about 10 mg/day to about 500 mg/day, about 10 mg/day to about 200 mg/day (e.g., 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, or 200 mg/day), 20 mg/day to about 100 mg/day, 100 mg/day to about 200 mg/day, or about 200 mg/day to about 500 mg/day (e.g., 190, 200, 210, 220, 230, 240, 250, 260, 270, 280, 290, 300, 310, 320, 330, 340, 350, 360, 370, 380, 390, 400, 410, 420, 430, 440, 450, 460, 470, 480, 490, 500, 510, 520, 530, 540, 550, 560, 570, 580, 590, 600, 610, 620, 630, 640, 650, 660, 670, 680, 690, or 700 mg/day), inclusive of any single or multi-dose daily administration regimen that falls within that total daily dose range. In some embodiments, the dose is from about 20 mg/day to about 100 mg/day. Additionally, one of ordinary skill in the art would also know how to adjust or modify variables such as dosage, dosage schedules, and routes of administration, as appropriate, for a given subject.
3. Methods of Treating Estrogen Receptor Positive Cancer of the Brain
[0104] The methods described above may be used to treat an estrogen receptor positive cancer of the brain. In some embodiments, the cancer may include subtypes of brain tumors that may express ER, such as Breast Cancer Brain Metastases (BCBM), Astrocytoma, Chondrosarcoma, Craniopharyngioma, Glioblastoma, Glioma, Hemangioma, Medulloblastoma, Meningioma, Neurofibroma, Neuronal and Mixed Neuronal-Glial Tumors, Oligoastrocytoma, Pituitary Tumor, PNET--(primitive neuroectodermal tumor), Schwannomak, or Leptomeningeal metastases. In some embodiments, the cancer may be other cancers such as Atypical Teratoid Rhabdoid Tumor (ATRT), Choroid Plexus Carcinoma, Ependymoma, Germ Cell Tumor, Juvenile Pilocytic Astrocytoma, Oligodendroglioma, or Pineal Tumor.
(a) Breast Cancer Brain Metastases
[0105] The methods described above may be used to treat a subject suffering from breast cancer brain metastases. 10-20% of breast cancer patients ultimately experience breast cancer metastasis to the brain, i.e., BCBM. 30-40% of BCBM express ER. ER expression is retained in 50-65% of BCBM that arise from ER+/PR+ tumors, despite treatment of the initial tumor with endocrine therapies. As many as 50% of BCBM express the estrogen receptor, and the brain is an environment rich in aromatase activity, suggesting that estrogen levels and signaling may be of importance in the establishment and maintenance of BCBM. Therefore, the availability of the SERM, such as RAD 1901 or a similar SERM with significant brain penetrance, that can induce estrogen receptor turnover in BCBM may provide therapeutic benefit in the treatment of BCBM.
[0106] Few treatments can access the brain, one of three most frequent sites of metastasis for breast cancer, leaving targeted or whole brain radiation as the standard of care for patients diagnosed by BCBM. In the majority of BCBM occurring in patients diagnosed with ESR1-positive breast tumors, ESR1 expression is retained, and the high expression of aromatase in the brain suggests that the disruption of ESR1 signaling may be beneficial in this setting. Therefore, the availability of the SERM, such as RAD1901 or a similar SERM with significant brain penetrance, that can induce estrogen receptor turnover in BCBM may provide therapeutic benefit in the treatment of BCBM.
(b) Astrocytoma
[0107] Astrocytoma is a type of cancer of the brain that originate in astrocytes, which are a particular kind of glial cells, start-shaped brains cells in the cerebrum. Low ERb expression has been shown to be associated with the progression of astrocytoma.
(c) Chondrosarcoma
[0108] Chondrosarcoma is a type of tumor that affects the bones and joints. Chondrosarcoma grow from the types of cells that make cartilage in the skull. In the head, these tumors grow inside the bones at the base of the back part of the skull and may be very close to the nerves and blood vessels around the brainstem. ER is present and active in chondrosarcoma tumors.
(d) Craniopharyngioma
[0109] Craniopharyngioma is a benign tumor that develops near the pituitary gland. ER may be present in craniopharyngioma and may be associated with improved disease prognosis.
(e) Glioblastoma multiforme
[0110] Glioblastoma multiforme, also known as "glioblastoma," is the most common and most aggressive malignant primary brain tumor in humans, involving glial cells and accounting for 52% of all functional tissue brain tumor cases and 20% of all intracranial tumors. Glioblastoma may express both ERs, which may play a role in etiology and treatment.
(f) Glioma
[0111] Glioma is a type of tumor that starts in the brain or spine and arises from glial cells. Gliomas make up approximately 30% of all brain and central nervous system tumors and 80% of all malignant brain tumors. Glioma may express both ERs, which may play a role in etiology and treatment. Glioma may be responsive to tamoxifen treatment.
(g) Hemangioma
[0112] Hemangioma is a benign and usually self-involuting tumor (swelling or growth) of the endothelial cells that line blood vessels. Hemangioma is characterized by increased number of normal or abnormal vessels filled with blood. Human vascular endothelial cells express ER isoforms and are responsive to tamoxifen treatment. Hemangioma may be intracranial hemangiomas or cutaneous hemangiomas.
(h) Medulloblastoma
[0113] Medulloblastoma is a highly malignant primary brain tumor that originates in the cerebellum or posterior fossa. Medulloblastomas may originate from immature or embryonal cells at their earliest stage of development. Medulloblastoma may express ER isoforms. ER isoforms are associated with growth and migration of these cells.
(i) Meningioma
[0114] Meningiomas are a diverse set of tumors arising from the meninges, i.e., the membranous layers surrounding the central nervous system. Meningioma may express both ER isoforms. Meningioma may be responsive to tamoxifen treatment.
j) Neurofibroma
[0115] Neurofibroma is a benign nerve sheath tumor in the peripheral nervous system.
[0116] Neurofibromas arise from nonmyelinating-type Schwann cells that exhibit biallelic inactivation of the NF1 gene that codes for the protein neurofibromin. Neurofibroma may be ER positive.
(k) Neuronal & Mixed Neuronal-Glial Tumors
[0117] Neuronal & Mixed Neuronal-Glial Tumors are rare, benign tumors that come from ganglion-type cells, i.e., groups of nerve cells. ER may be present in these tumors.
(l) Oligoastrocytoma
[0118] Oligoastrocytomas are a subset of brain tumors that present with an appearance of mixed glial cell origin, astrocytoma and oligodendroglioma. Oligoastrocytoma may have a lasting response to tamoxifen treatment.
(m) Pituitary Tumor
[0119] A pituitary tumor is an abnormal growth in the pituitary gland. Both ER.alpha. and ERb may be detected in pituitary tumors.
(n) Primitive Neuroestodermal Tumor (PNET)
[0120] Primitive neuroestodermal tumor is a neural crest tumor. The majority of the cells in the PNET are derived from neuroectoderm but have not developed and differentiated in the way a normal neuron would, and so the cells appear "primitive. ERa may be present and may increase metastatic potential via extracellular signal-regulated Kinase (ERK) activation.
(o) Schwannoma
[0121] Schwannoma (also known as an "neurilemmoma," "neurinoma," "neurolemmoma," and "Schwann cell tumor") is a benign nerve sheath tumor composed of Schwann cells, which normally produce the insulating myelin sheath covering peripheral nerves. Schwannoma may express ER.alpha..
(p) Leptomeningeal Metastases
[0122] Leptomeningeal metastasis is breast cancer metastasis to the membranes (meninges) surrounding the brain and spinal cord. A durable response has been observed in patient(s) treated with aromatase inhibitors, which suggests possible responsiveness to ER targeted therapies.
4. Methods of Treating a Cancer that is Resistant to an Estrogen Receptor Modulator
[0123] The methods described above may be used to treat a cancer that is resistant to an estrogen receptor modulator. The resistance to the estrogen receptor modulator may be acquired. The estrogen receptor modulator may be a selective estrogen receptor modulator (SERM). The SERM may be tamoxifen, idoxifene, raloxifene, or ICI 182,780. The cancer may be breast, endometrial or ovarian cancer. The cancer may be tamoxifen resistant breast cancer.
[0124] Because patients diagnosed with BCBM are likely to have already progressed following endocrine therapies, RAD1901 may be used in breast tumors resistant to tamoxifen and/or aromatase inhibitors. RAD1901 may be beneficial in the BCBM setting when combined with additional therapeutic(s) that will ensure the inhibition of growth of peripheral metastases.
5. Mechanisms of Delivery
[0125] The SERM may be formulated to be compatible with its intended route of administration. Examples of routes of administration include, but are not limited to, parenteral, e.g., intravenous, intradermal, subcutaneous, oral, intranasal (e.g., inhalation), transdermal (e.g., topical), transmucosal, and rectal administration. In a specific embodiment, the SERM is formulated in accordance with routine procedures as a pharmaceutical composition adapted for intravenous, subcutaneous, intramuscular, oral, intranasal, or topical administration to human beings. Typically, compositions for intravenous administration are solutions in sterile isotonic aqueous buffer. Where necessary, the composition may also include a solubilizing agent and a local anesthetic such as lignocaine to ease pain at the site of the injection.
[0126] Various delivery systems are known and can be used to administer one or more SERMs or the combination of one or more SERMs and a prophylactic agent or therapeutic agent useful for preventing, managing, treating, or ameliorating a disorder or one or more symptoms thereof, e.g., encapsulation in liposomes, microparticles, microcapsules, receptor-mediated endocytosis (see, e.g., Wu and Wu, J. Biol. Chem. 262:4429-4432 (1987)), etc. Methods of administering a prophylactic or therapeutic agent of the SERM include, but are not limited to, parenteral administration (e.g., intradermal, intramuscular, intraperitoneal, intravenous and subcutaneous), epidural administration, intratumoral administration, and mucosal administration (e.g., intranasal and oral routes).
6. Combination Treatments
[0127] The methods described above may include a combination treatment of the compound of formula I with other drugs and/or other conventional cancer therapies, such as hormone therapy.
(a) Combination Drugs
[0128] The methods may further include administering an effective amount of at least one compound of a cyclin-dependent kinase 4 and 6 inhibitor (CDK4/6 inhibitor), an antiestrogen, a ligand of retinoic acid or retinoic X receptor, an antiprogestin, an antiandrogen, vitamin D or metabolite thereof, a farnesyl transferase inhibitor, a PPAR.alpha. or gamma agonist and a MAP kinase inhibitor.
(b) Conventional Cancer Therapies
[0129] Conventional cancer therapies may include surgery, radiation therapy, chemotherapy, hormone therapy, and targeted therapy. Examples of surgery include open craniotomy with maximal excision, which may be followed by radiation therapy. Examples of radiation therapy include whole-brain irradiation, fractionated radiotherapy, and radiosurgery, such as stereotactic radiosurgery, e.g., Gamma Knife radiosurgery. Examples of chemotherapy include anthracyclines, such as doxorubicin (Adriamycin, Doxil), epirubicin (Ellence), and daunorubicin (Cerubidine, DaunoXome), capecitabine (Xeloda), carboplatin (Paraplatin), cisplatin, cyclophosphamide (Cytoxan), eribulin (Halaven), fluorouracil (also called 5-fluorouracil or 5-FU; Adrucil), gemcitabine (Gemzar), ixabepilone (Ixempra), methotrexate (Amethopterin, Mexate, Folex), mitoxantrone (Novantrone), mutamycin (Mitomycin), taxanes, such as paclitaxel (Taxol, Abraxane), and docetaxel (Taxotere), thiotepa (Thioplex), vincristine (Oncovin, Vincasar PES, Vincrex), and vinorelbine (Navelbine). Examples of targeted therapy include trastuzumab (Herceptin), lapatinib (Tykerb), bevacizumab (Avastin), pertuzumab (Perjeta), and everolimus (Afinitor).
[0130] i. Endocrine Therapy (Hormone Therapy)
[0131] Endocrine therapy, also known as hormonal therapy, hormone therapy, and hormone treatment, is a treatment that adds, blocks, or removes hormones. For example, hormones may be given to adjust low hormone levels. Synthetic hormones or other drugs may be given to block the body's natural hormones to slow or stop the growth of certain cancers (such as prostate and breast cancer). Endocrine therapy may also include surgery to remove the gland that makes a certain hormones.
[0132] Examples of hormone therapy include selective estrogen receptor modulators (SERMs), such as tamoxifen, raloxifene, endoxifene, toremifene, lasofoxifene, pipendoxifene, bazedoxifene, and ospemifene, aromatase inhibitors, such anastrozole, letrozole, exemestane, formestane, fadrozole, aminoglutethimide, and testolactone, a HER2 intervention drug, such as a HER2 inhibitor, such as Herceptin (trastuzumab), pertuzumab, and lapatinib, and estrogen-receptor downregulators, such as fulvestrant (ICI 182,780).
7. Subject or Subject in the Method
[0133] The methods described above are directed to treating a subject with a SERM. The subject treated by the methods described above may be a subject or patient suffering from or at risk of suffering from an estrogen receptor positive cancer of the brain, such as BCBM, or a cancer that is resistant to an estrogen receptor modulator, such as tamoxifen resistant breast cancer. The subject may be diagnosed or identified as having or at risk of having cancer using known methods and assays, such as a biopsy. The subject may be treated with SERM alone or in combination with another drug and/or conventional cancer therapy, as described above. The subject may be treated with the SERM as a neoadjuvant therapy or post-surgery. The present invention has multiple aspects, illustrated by the following non-limiting examples.
EXAMPLES
[0134] The foregoing may be better understood by reference to the following examples, which are presented for purposes of illustration and are not intended to limit the scope of the invention.
Example 1
In Vitro Analysis of ER Degradation
[0135] 48 hours prior to treatment, MCF7 cells were plated in phenol red free DMEM/F12 media supplemented with 8% charcoal stripped fetal bovine serum, non-essential amino acids, and sodium pyruvate. After 20 hours of treatment with the indicated ligands, i.e., estradiol ("E2"; Sigma-Aldrich), antiestrogen ICI 182,780 ("IC"; Sigma-Aldrich), and 4-hydroxytamoxifen ("40HT"; Sigma-Aldrich), (0-1 .mu.M), cells were washed and lysed in RIPA lysis buffer (50 mM Tris, pH 8, 150 mM NaCl, 1% IGEPAL, 0.02% SDS, 0.5% sodium deoxycholate, 1 mM EDTA). 50 .mu.g of cleared lysate was resolved by SDS-PAGE and analyzed by immunoblot detection of ER.alpha. or cytokeratin 18 (loading control), as illustrated in FIG. 1A.
Example 2
In Vivo Analysis of RAD1901 in MCF7 Xenograft Tumors
[0136] A total of 90 female Nu/Nu mice were ovariectomized and implanted subcutaneous simultaneously with an estrogen pellet (Innovative Research of America) releasing 0.72 mg estradiol (E2) over 60 days. 2 days later an approximately 6 mm3 fragment of an MCF7 xenograft tumor (isolated from a recently sacrificed estrogen treated nu/nu mouse) was inserted subcutaneous into the axial mammary gland. Tumor growth (by caliper measurement) and animal body weight were monitored 3.times. weekly until tumor volume reached .about.0.2 cm.sup.3. Mice (n.about.10) were randomized to the following groups: estrogen control (corn oil vehicle), E2+ RAD1901 (20 mg/kg; Radius Health, Inc.), E2+ Tamoxifen (20 mg/kg; Sigma-Aldrich). Treatments were formulated in sterile corn oil and were administered daily by subcutaneous injection. After 3 weeks of treatments, animals were euthanized and serum and tissues saved for analysis. FIG. 1B depicts tumor volume analyzed using non-linear curve fit and exponential growth calculation (Graphpad Prism), followed by two-way ANOVA and Bonferroni analysis.
Example 3
[0137] Mechanism by which RAD1901 Downregulates ER Expression
[0138] While the reduced levels of ER following treatment with RAD1901 results in receptor degradation, whether the drug influences the transcriptional activity of the gene encoding ER was determined (FIG. 2). MCF7 cells were pre-treated with Vehicle (Veh) or translation (cyclohexamide--CHX, 10 .mu.g/ml), proteasome (MG132, 30 .mu.M) or transcription (Actinomycin D--Actin. D, 100 ng/ml) inhibitors for 2 hours prior to 6 hours of treatment with RAD1901 (0.1 or 1 .mu.M), or 0.1 .mu.M ICI 182,780 (ICI) or raloxifene (Ral). FIG. 2A shows ER.alpha. protein expression in whole cell extracts was analyzed by immunoblot, as in Example 1. FIG. 2B shows cells treated, as indicated, were washed in PBS prior to lysis. RNA isolation (BioRad) and reverse transcription (iScript; BioRad) were performed per kit manufacturer's instructions. qRT-PCR of cDNA was done using iQ SYBR Green Supermix (Bio-Rad) per kit instructions and performed using the iCycler optical system with associated software (Bio-Rad). mRNA abundance was calculated using the .DELTA..DELTA.CT method to normalize ER.alpha. mRNA expression to similarly detected housekeeping gene 36B4.
Example 4
Conformational Changes Induced in ER as a Consequence of Binding of RAD1901
[0139] A series of short peptides whose ability to interact with ER.alpha. is influenced by the nature of the bound ligand were previously identified (see Chang et al. Methods Enzymol. (2003) 364: 118-42; Huang et al. Mol Endocrinol. (2002) 16(8): 1778-92; Connor et al. Cancer Res. (2001) 61(7): 2917-22; Chang et al. Mol Cell Biol. (1999) 19(12):8226-39; and Norris et al. Science (1999) 285(5428): 744-6). The interaction of these "conformational probes" can be measured in vitro or within intact cells and thus enables the definition of ligand induced conformational changes in the receptor. This is significant, since differences in receptor conformation facilitate the engagement of different coregulatory proteins resulting in different pharmacological activities. Application of this technology has led to the determination that their impact on receptor structure is a distinguishing feature of ER ligands.
[0140] FIG. 3 shows that RAD1901 induces a unique conformation of ER.alpha. as shown in the interaction between ER and conformation-specific peptides in a mammalian two-hybrid system. Triplicate wells of SKBR3 cells were transfected (Lipofectin per manufacturer's instructions) with plasmids expressing ER.alpha. fused to VP16 together with Gal4DBD alone (control) or fused to ER interacting peptides noted on the horizontal axis. Cells were then treated with RAD1901 (1 nM-1 .mu.M) or the indicated ER ligands (100 nM). Interaction of ER.alpha. with the Gal4DBD peptide constructs was detected through activation of a Gal4 responsive luciferase reporter construct and was normalized to detected .beta.-galactosidase activity generated by a co-transfected constitutive expression vector. Ligand classes recognized by each probe are indicated below the graph.
Example 5
[0141] RAD1901 Possesses Dose Dependent Agonist and/or Antagonist Potential
[0142] The complex pharmacological activities exhibited by RAD1901 suggest that it will exhibit a unique gene expression profile in target cells. A recent extensive microarray analysis has identified "sentinel" subsets of ER-responsive genes that can differentiate between ER agonists, SERMs and SERDs. For example, some of these are responsive to only the SERM tamoxifen, while others display a graded response to SERMs with varying agonist/antagonist potential, and yet others are repressed by agonists or SERMs and are induced only by SERDs. RAD1901 exhibits dose dependent agonist/antagonist regulation of ER transactivation of target genes (FIG. 4). MCF7 breast cancer cells were treated 24 hours with RAD1901 (0-1 .mu.M) in the presence or absence of 17.beta.-estradiol (1 nM). RNA isolation and the analysis of the expression of target genes Anterior gradient protein 2 (AGR2) and KCNK6 was conducted as in Example 3.
Example 6
Materials and Methods
[0143] Reagents. Purchased ESR1 ligands included 17.beta.-estradiol (Sigma), ICI 182,780 (Tocris), tamoxifen (Sigma), raloxifene (Tocris), and 4-hydroxytamoxifen (Sigma). (R)-6-(2-(N-(4-(2-(ethylamino)ethyl)benzyl)-N-ethylamino)-4-methoxyphenyl- )-5,6,7,8-tetrahydronaphthalen-2-ol dihydrochloride was provided by Radius Pharmaceuticals. Bazedoxifene and GW7604 were synthesized as previously described (Miller et al., (2001) J Med Chem 44 (11):1654-1657; U.S. Pat. No. 5,681,835). Ligands were dissolved in ethanol or DMSO.
[0144] Cell culture. MCF7 and SKBR3 cell lines were maintained in DMEM/F12 or RPMI media (Invitrogen), respectively, supplemented with 8% fetal bovine serum (FBS, Gemini), non-essential amino acids (Invitrogen), and sodium pyruvate (Invitrogen). Unless otherwise indicated, cells were plated for experiments in media lacking phenol red and supplemented with 8% charcoal stripped FBS (CFS, Gemini). LTED MCF7 cells were maintained and plated for experiments in phenol red free DMEM/F12 media supplemented with 8% CFS that had been charcoal stripped twice. 48 hours after plating, cells were treated with ESR1 ligands as indicated, and were harvested for immunoblot or real time quantitative PCR analysis 24 hours after treatment. Cell lines were authenticated by STR analysis performed by ATCC in 2013.
[0145] Immunoblot analysis. Protein expression was analyzed as described (Wittmann et al., (2007) Cancer Res 67 (19):9549-9560) using antibodies purchased from Sigma-Aldrich--A5441 (.beta.-actin) and Santa Cruz Biotechnology--sc-6259 (cytokeratin 18), sc-20680 (lamin A), sc-5546 (.alpha.-tubulin) and sc-8005 (ESR1).
[0146] RNA isolation and real time quantitative PCR. RNA isolation and analysis was performed as described (Wardell et al., (2001) Biochem Pharmacol 82 (2):122-130). mRNA abundance was calculated using the .DELTA..DELTA.CT method (Wardell et al., (2001) Biochem Pharmacol 82 (2):122-130). Primer sequences are available upon request.
[0147] Proliferation assays. Assays evaluating the effects of SERDs and SERMs on cell proliferation were performed as described (Wardell et al., (2013) Clin Cancer Res 19 (9):2420-2431).
[0148] Transfections. Mammalian 2-hybrid analysis of VP16-ESR1 with conformation-selective peptide probes was performed essentially as previously described (Wardell et al., (2012) Mol Endocrinol 26 (7):1235-1248).
[0149] In vivo studies. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the Duke University Institutional Animal Care and Use Committee.
[0150] Uterine wet weight analysis: Ovariectomized (10 days prior) female C57Bl/6 mice (Charles River) were treated daily (n=5) for 3 days with vehicle or estradiol benzoate (10 .mu.g/kg sc) as well as vehicle, raloxifene (10 mg/kg sc), or RAD1901 (0.1-100 mg/kg sc). Ligands were dissolved in corn oil (Spectrum chemicals). On day 4, mice were euthanized, and tissues were retained for analysis. Uterine wet weight was calculated as a ratio of uterus weight upon removal to body weight post-mortem.
[0151] Xenograft tumor analysis: Estrogen-stimulated MCF7 tumors were initiated in the axial mammary gland of 6-week old estrogen-treated (0.72 mg/60 days pellet sc, Innovative Research of America) ovariectomized female NU/NU mice (in-house colony) by serial transfer and were measured as described (Wardell et al., (2013) Clin Cancer Res 19 (9):2420-2431). FIG. 5: At .about.0.1 cm.sup.3 tumor volume, mice were randomized (n=9-10) to daily treatment with vehicle, RAD1901 (20 mg/kg), or tamoxifen (20 mg/kg). FIG. 9: At .about.0.1 cm.sup.3 tumor volume, the estrogen pellet was surgically removed, and mice were randomized (n=6-10) to daily treatment with vehicle or RAD1901 (0.3-10 mg/kg). Treatments were formulated as above. Animal tissues were processed and analyzed as described (Wardell et al., (2013) Clin Cancer Res 19 (9):2420-2431).
Example 7
RAD1901 Inhibits ESR1 Activity In Vitro and In Vivo
[0152] The ability of RAD1901 (FIG. 5A) to modulate the 17-.beta. estradiol (E2) dependent transcriptional activity of the human estrogen receptor alpha (ESR1) and human estrogen receptor beta (ESR2) was assessed in transiently transfected SKBR3 cells using a synthetic reporter gene. FIG. 5A shows the chemical structure of the SERM RAD1901. For FIG. 5B, SKBR3 cells were plated in phenol red free media supplemented with charcoal stripped FBS (CFS) 24 hours prior to transfection with an ERE-luciferase reporter together with ESR1 or ESR2 expression vectors. 24 hours after transfection, cells were treated with E2 (10 nM) together with RAD1901 (10.sup.-10-10.sup.-6 M) for 24 hours prior to harvest and analysis of luciferase activity normalized to co-transfected .beta.-galactosidase control. For FIG. 5C, MCF7 cells were plated in phenol red free media supplemented with CFS 48 hours prior to treatment with 10.sup.-9 M E2 together with ICI 182,780 (ICI), RAD1901 (RAD), GW7604, or 4-hydroxytamoxifen (40HT) (10.sup.-11-10.sup.-6 M) for 24 hours. mRNA levels of ESR1 target gene trefoil factor 1 (TFF1) were assessed using RT qPCR following RNA isolation. mRNA expression was normalized to the similarly detected 36B4 housekeeping gene, and expression levels are presented as fold change as compared to the vehicle-treated control. For FIG. 5D, MCF7 cells were plated in phenol red free media supplemented with CFS 24 hours prior to treatment, and were treated with 10.sup.-9 M E2 as well as with the indicated ligands (10.sup.-11-10.sup.-6 M) on days 1, 4, and 6 of an 8 day proliferation assay. DNA content as assessed by fluorescence was measured as a surrogate for cell proliferation. The relative increase in DNA fluorescence was calculated by normalizing to baseline values detected in a duplicate plate of cells that was harvested on day 1 prior to the initial treatment. Data are representative of at least 3 independent experiments. For FIGS. 5E-5F, MCF7 cell derived tumors were implanted into ovariectomized estrogen-treated nu/nu mice. When tumor volume reached .about.0.1 cm.sup.3, animals (n=9-10) were randomized to receive daily treatment with vehicle, tamoxifen (Tam, 20 mg/kg sc) or RAD1901 (RAD) (20 mg/kg sc). For FIG. 5E, mean tumor volume.+-.SEM per day of treatment was presented. Significance (2-way ANOVA of matched values followed by Bonferroni comparison) as compared to the vehicle control was indicate d (*p<0.0001). For FIG. 5F, expression of ESR1 target genes in tumors was analyzed essentially as in (C).
[0153] In this analysis it was determined that although RAD1901 effectively inhibited E2-dependent activation of an ERE-luciferase reporter by either isoform, it was a more potent inhibitor of ESR1 (100-fold) (FIG. 5B). Similarly, it was demonstrated in MCF7 breast cancer cells that RAD1901 inhibited E2-dependent (a) induction of target gene transcription and (b) stimulation of cell proliferation with an efficacy and potency similar to that of the SERMs 4-hydroxytamoxifen (40HT) and raloxifene (Ralox) and the SERDs ICI 182,780 (ICI, fulvestrant) and GW7604 (FIG. 5C-5D). To assess the activity of RAD1901 in vivo, a xenograft tumor study was conducted in estrogen-treated immunocompromised mice using the well-characterized ESR1-dependent MCF7 cell model. In this study, it was observed that RAD1901 (20 mg/kg) inhibited E2-stimulated growth of the tumors with efficacy similar to tamoxifen (20 mg/kg). RAD1901 and tamoxifen were also shown to suppress the expression of the ESR1-target genes PGR and FHL1 to the same degree in treated tumors, a result that confirms target engagement (FIGS. 5E-5F).
Example 8
RAD1901 Exhibits the Pharmacological Properties of a Selective Estrogen Receptor Degrader (SERD)
[0154] RAD1901 not only functioned as an ESR1 antagonist, but also downregulated the expression of this receptor. ESR1 expression was significantly downregulated in MCF-7 cells treated with RAD1901. For FIG. 6A, MCF7 cells were treated for 24 hours with ICI (10.sup.-13-10.sup.-7 M) or RAD1901 (RAD), (10.sup.-11-10.sup.-5 M). Expression of ESR1 and loading control cytokeratin 18 (CK18--FIG. 7A) in whole cell extracts were detected by immunoblot (top). ESR1 levels relative to CK18 were quantitated by densitometry using Adobe Photoshop (bottom). For FIG. 6B, MCF7 cells were plated as in FIG. 5B prior to 1 hour pre-treatment with vehicle or MG132 (10 .mu.g/ml), followed by 6 hours of treatment with 10.sup.-7 M vehicle, ICI, Ral or RAD1901 (RAD), (10.sup.-8-10.sup.-6). ESR1 expression was detected as in FIG. 6A. For FIGS. 6C-6D, LTED MCF7 cells were plated in phenol red free media supplemented with FBS that was stripped of growth factors twice using charcoal. For FIG. 6C, after 48 hours, cells were treated for 24 hours with E2 (10.sup.-7 M) or SERDs (10.sup.-6 M) and ESR1 was analyzed as in FIG. 6A. For FIG. 6D, LTED MCF7 cells were treated with ICI or RAD1901 (RAD), (10.sup.-11-10.sup.-6 M) on days 1, 4, and 6 of an 8 day proliferation assay and analyzed as in FIG. 5.
[0155] For FIG. 7A, cytokeratin 18 (CK18) loading control was detected for samples illustrated in FIG. 6A. For FIG. 7B, BT483 cells were plated in phenol red free media supplemented with CFS 48 hours prior to treatment with 10.sup.-9 M E2 together with ICI 182,780 (ICI), RAD1901 (RAD), GW7604, or 4-hydroxytamoxifen (40HT) (10.sup.-11-10.sup.-6 M) for 24 hours. mRNA levels of ESR1 target gene trefoil factor 1 (TFF1) were assessed using RT qPCR following RNA isolation. mRNA expression was normalized to the similarly detected 36B4 housekeeping gene, and expression levels are presented as fold change as compared to the vehicle-treated control. For FIG. 7C, BT483 cells were treated for 24 hours with ICI (10.sup.-13-10.sup.-7 M) or RAD1901 (RAD), (10.sup.-11-10.sup.-5 M). Expression of ESR1 and loading control cytokeratin 18 in whole cell extracts were detected by immunoblot (right). ESR1 levels relative to CK18 were quantitated by densitometry using Adobe Photoshop (left). For FIG. 7D, BT483 cells were plated in phenol red free media supplemented with CFS 24 hours prior to treatment, and were treated with 10.sup.-9 M E2 as well as with the indicated ligands (10.sup.-11-10.sup.-6 M) on days 1, 4, and 6 of an 8 day proliferation assay. DNA content as assessed by fluorescence was measured as a surrogate for cell proliferation. The relative increase in DNA fluorescence was calculated by normalizing to baseline values detected in a duplicate plate of cells that was harvested on day 1 prior to the initial treatment. Data are representative of at least 3 independent experiments.
[0156] The downregulation of the ESR1 by RAD1901 was a significant contributor to its antagonist efficacy (FIG. 6A). RAD1901 had no effect on ESR1 mRNA expression (not shown). However, as observed in ICI-treated cells, the downregulation of ESR1 by RAD1901 was completely blocked by pre-treatment of cells with the proteasome inhibitor MG132 (FIG. 6B). Similar results were obtained in the BT483 breast cancer cell line (FIGS. 7B-7D). For comparative purposes, ESR1 expression levels were evaluated in cells treated with tamoxifen, which stabilizes ESR1 expression, and SERDs bazedoxifene (BZA), GW7604 and ICI. These studies revealed that the degree of downregulation of ESR1 by RAD1901 was similar to that achieved by BZA under the same conditions (FIG. 6C).
[0157] To assess the potential significance of this SERD activity, the activity of RAD1901 was evaluated in LTED MCF7 cells, an accepted model of aromatase resistance in which compounds with ER-antagonist activity alone, like tamoxifen, were minimally effective (not shown). In this assay, it was demonstrated that, like ICI and BZA, RAD1901 downregulated ESR1 expression and inhibited cell proliferation (FIG. 6D). Thus, considering its antagonist activity in several models of breast cancer and its ability to downregulate ESR1, it was appropriate to classify RAD1901 as a SERD.
Example 9
RAD1901 Exhibits Dose Dependent SERD Activity In Vivo
[0158] The observation that RAD1901 exhibited the pharmacological properties of a SERD was unexpected given that it was (a) identified in screens for compounds that manifest ESR1 agonist activity in the CNS and (b) evaluated in clinical trials as a potential treatment for the vasomotor symptoms (hot flashes) associated with menopause, an indication for which only estrogens have proven effective. However, the results of the clinical trials for hot flashes revealed that RAD1901 exhibited a complex pharmacology. At the lowest dose tested it appeared to effectively suppress hot flashes but was ineffective at the higher doses tested. This inverted U-shaped pharmacology suggested that at low doses this compound may have favorable agonist activity, but at higher doses the ability of RAD1901 to induce ESR1 turnover dominates. A series of studies was performed to explore the functional consequences of the complex pharmacological activities of RAD1901 in vivo.
[0159] As a first step, the impact of RAD1901 on uterine wet weight in mice was evaluated. For this study, increasing doses of RAD1901 was administered daily to ovariectomized C57Bl/6 mice receiving vehicle alone or E2 (10 .mu.g/kg; a physiological replacement dose). A group of mice receiving 10 mg/kg of raloxifene was included for comparative purposes and for reference. Ovariectomized C57Bl/6 mice (n=5) were treated daily for 3 days with (FIG. 8A) vehicle or (FIG. 8B) estradiol (10 .mu.g/kg) together with vehicle, Ralox (10 mg/kg) or RAD1901 (RAD), (0.3-100 mg/kg). At euthanasia, body weight as well as uterine wet weight was measured prior to cryopreservation of the uterus. When administered as a single agent, a statistically significant increase in uterine weight was observed in those animals receiving the lowest dose of RAD1901 (0.3 mg/kg). For FIG. 8C, ESR1 and .beta.-actin expression in extracts made from pulverized uterine tissues were analyzed by immunoblot as in FIG. 5 (left). ESR1 expression relative to .beta.-actin was quantitated as in FIG. 6 (right). Significant downregulation (*p<0.05) of ESR1 was determined by ANOVA followed by Bonferroni comparison.
[0160] At doses of 1 mg/kg and above, the uterine wet weights of treated animals were indistinguishable from vehicle treated animals (FIG. 8A). Further, RAD1901 administration was shown to inhibit E2-dependent increases in uterine wet weight (FIG. 8B), an activity that tracked with the dose dependent downregulation of ESR1 expression (FIG. 8C). Notably, no decrease in ESR1 expression was observed in animals treated with 0.3 mg/kg, the dose where the agonist activity of RAD1901 in the uterine wet weight assay was observed (FIG. 8C). As observed in clinical trial for hot flashes, RAD1901 exhibited a complex biphasic pharmacology that manifests as antagonist activity at the higher doses.
Example 10
[0161] RAD1901 Exhibits Biphasic Activity with Respect to ESR1-Dependent Tumor Growth
[0162] As shown above, high dose RAD1901 (20 mg/kg) inhibited the growth of ESR1-dependent MCF7-cell derived tumors in mice. However, the observation that uterine wet weight was increased in mice treated with doses of RAD1901 that were lower than that required to effect ESR1 turnover highlighted the need to examine whether a similar biphasic pharmacology was manifest in breast tumors. To further evaluate the pharmacology of RAD1901, a second xenograft tumor study was conducted in which MCF7 tumors were established under estrogen stimulation. For FIG. 9A, MCF7 xenograft tumors were initiated in ovariectomized female nu/nu mice as in FIG. 5. Estrogen pellets were surgically removed when tumors reached .about.0.1 cm.sup.3 volume, and animals (n=6-10) then received daily treatment with vehicle or RAD1901 (RAD), (0.3-10 mg/kg sc). Mean tumor volume.+-.SEM per day of treatment was presented. Significance as compared to the vehicle (2-way ANOVA of matched values followed by Bonferroni comparison) was indicated (*p<0.05, **p<0.0005). For FIG. 9B, uterine wet weight at sacrifice (measured as in FIG. 8) and % change in tumor volume (as compared to size at randomization) calculated using the final measurement recorded for mice in (FIG. 9A) are graphically presented. For FIGS. 9C-9D, expression of ESR1 target genes in tumors was analyzed essentially as in FIG. 5. Estrogen only samples from FIG. 5D were included for comparison. For FIG. 9E, ESR1 levels in tumor tissues were analyzed as in FIG. 8 and were normalized to similarly detected Lamin-A. Significant downregulation (*p<0.05) was determined by ANOVA followed by Bonferroni comparison.
[0163] When tumors reached .about.0.1 cm.sup.3 volume, estrogen was discontinued and animals were randomized to treatment with vehicle or RAD1901 (0.3, 1, 3, or 10 mg/kg). As observed in the uterine weight assay, RAD1901 exhibited a biphasic response, in that significant stimulation of tumor growth was observed in animals treated with 1 or 3 mg/kg RAD1901 that was not apparent at the higher dose (FIG. 9A). Although certainly less than that which was observed following E2 stimulation (FIG. 5E), the increased tumor volume in the 1 and 3 mg/kg groups was significant and of similar magnitude to that for partial ESR1 agonists. Interestingly, an evaluation of the final tumor size and uterine wet weight of these animals at sacrifice revealed that the pharmacology of RAD1901 was affected by both dose and tissue. Specifically, as reported above in a different strain of mice, stimulation of uterine weight was apparent in mice treated with only the 0.3 mg/kg dose of RAD1901, while tumor size was significantly increased in 1 or 3 mg/kg RAD1901 groups (FIG. 9B). Whereas the expression of classical ESR1 target genes such as PGR was not observed in the tumors in the 1 or 3 mg/kg treatment groups (FIG. 9C), the expression of AGR2 (FIG. 9D) and others SERM-regulated genes associated with tamoxifen resistance (not shown) was observed. Immunoblot analysis of tumor extracts revealed a dose-dependent downregulation of ESR1 by RAD1901 that reflected its actions as an inhibitor of tumor growth (FIG. 9E). Together these data highlight the complex pharmacology of RAD1901 and stress the importance of developing biomarkers that read on the partial agonist activity of the drug and which can be used for dose optimization in clinical studies.
Example 11
[0164] Elucidation of the Mechanisms that Distinguish RAD1901 from Other SERDs
[0165] Whether the dose-dependent agonist/antagonist activity of RAD1901 could be observed at the level of target gene regulation was determined. Specific sets of ESR1 target genes whose expression was differentially regulated by estrogens and SERMs and which could be used to distinguish between different SERMs and SERDs have been identified. A subset of these genes, those regulated by (a) agonists (with no or minimal response to SERMs), (b) SERMs (with no or little response to E2), or (c) either agonists or SERMs, were selected to profile RAD1901 activity.
[0166] MCF7 cells were treated for 24 hours with 10.sup.-7 M vehicle (Veh), ICI, 40HT, raloxifene (Ralox), E2 (10.sup.-9 M) or RAD1901 (10.sup.-9-10.sup.-6 M). The expression of ESR1 target genes responsive to (FIG. 10A) agonists, (FIG. 10B) primarily SERMs, or (FIG. 10C) SERMs and agonists was analyzed as in FIG. 5. Relative changes of these and additional target genes designed to evaluate dose dependent response to RAD1901 are presented in FIG. 11. Significant target gene regulation (*p<0.05) as compared to the vehicle control was detected by 2-way ANOVA followed by Fisher's LSD (Graphpad Prism 6). FIG. 10D shows the interaction between ESR1 and conformation-specific peptides in a mammalian two-hybrid system. Triplicate wells of SKBR3 cells were transfected with plasmids expressing ESR1 fused to VP16 together with Gal4DBD alone (control) or Gal4DBD fused to ESR1 interacting peptides noted on the horizontal axis. Cells were then treated with the indicated ESR1 ligands (10.sup.-7 M unless otherwise indicated). Interaction of ESR1 with the Gal4DBD peptide constructs was detected through activation of a Gal4-responsive luciferase reporter construct and was normalized to detected .beta.-galactosidase activity expressed in a constitutive manner using a second vector. Normalized response was expressed as fold increase over the detected level of interaction between Gal4DBD alone and ESR1-VP16 in the absence of ligand (Veh). For FIG. 10E, the effect of SERMs and SERDs (10.sup.-6 M) on the proliferation of MCF7 cells in response to E2 (10.sup.-9 M) or insulin (2.times.10.sup.-9 M) was evaluated as in FIG. 5. Statistically similar treatments (2-way ANOVA followed by Bonferroni comparison) are indicated by letters. For FIG. 11, MCF7 cells were treated for 24 hours with 10.sup.-7 M vehicle (Veh), ICI, 40HT, raloxifene (Ralox), E2 (10.sup.-9 M) or RAD1901 (10.sup.-9-10.sup.-6 M). The expression of ESR1 target genes responsive to (A) agonists, (B) primarily SERMs, or (C) SERMs and agonists was analyzed using RT qPCR following RNA isolation. mRNA expression was normalized (.DELTA..DELTA.CT analysis) to the similarly detected 36B4 housekeeping. The profiles presented in dendogram format were analyzed with the Ward hierarchical clustering algorithm (JMP 11) using standardized data.
[0167] Reflecting the pharmacology observed in vivo, the expression pattern of these target genes exhibited a biphasic response to RAD1901 with agonist activity been observed at lower doses and more complete antagonist activity apparent at higher doses (FIGS. 10A-10C and FIG. 11). These data confirmed the unique pharmacology of RAD1901 and suggested that this drug may function by a mechanism that was distinct from other ESR1 downregulators.
[0168] The pharmacology of ESR1 ligands reflects their influence on the overall structure of the receptor and on the impact which this has on coregulator recruitment. Given the distinct pharmacology exhibited by RAD1901, it may enable ESR1 to adopt a unique conformation. A conformational profiling tool was used to interrogate the ESR1-RAD1901 complex. In this assay a modified two-hybrid assay was used to assess the binding of a series of short peptides that survey the protein-protein interaction surfaces on ESR1 that were presented when occupied by different ligands. As shown in FIG. 10D, the interaction profile observed in the presence of RAD1901 was completely distinct from any other known ligand. Notable was the ability of RAD1901 to disengage the classical coregulator-binding surface (AF2) as indicated by the decreased interaction with the peptides that report on "agonist activity" (ASC1 and PGC1). No interaction was observed with peptides that report on the ESR1 structures adopted upon binding tamoxifen (.alpha..beta.V) or ICI (.beta.I2). There was significant interaction of all in the presence of RAD1901, a peptide that reports on a cryptic protein-protein interaction surface in the hinge region of the receptor. RAD1901 enabled ESR1 to assume a unique conformation that was distinct from that apparent upon binding agonists, the SERM tamoxifen, and other known SERDs (FIG. 10D).
[0169] In addition to classical agonists, ESR1 transcriptional activity can also be induced by treating cells with growth factors such as EGF, IGF1 or insulin. Whereas the mechanisms underlying this "ligand-independent" activity were likely to be complex, this alternate pathway of activation contributes to resistance to endocrine therapy in breast cancer. The comparative pharmacology of RAD1901 was evaluated for its ability to suppress growth factor dependent activation of ESR1. In this assay, it was determined that although all of the SERMs and SERDs tested inhibited E2-stimulated proliferation, only BZA and ICI efficiently inhibited insulin-stimulated proliferation, while 40HT, Ralox, and RAD1901 were without effect (FIG. 10E). These results confirm the unique mechanism of action of RAD1901. RAD1901 may be delivered in combination with another drug that inhibits ligand-independent activation of ESR1 to achieve maximal clinical response.
[0170] It is understood that the foregoing detailed description and accompanying examples are merely illustrative and are not to be taken as limitations upon the scope of the invention, which is defined solely by the appended claims and their equivalents.
[0171] Various changes and modifications to the disclosed embodiments will be apparent to those skilled in the art. Such changes and modifications, including without limitation those relating to the chemical structures, substituents, derivatives, intermediates, syntheses, compositions, formulations, or methods of use of the invention, may be made without departing from the spirit and scope thereof.
[0172] For reasons of completeness, various aspects of the invention are set out in the following numbered clauses:
[0173] Clause 1. A method of treating estrogen receptor positive cancers of the brain in a subject, the method comprising administering a compound represented by the following formula I:
##STR00013##
wherein [0174] TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C.sub.1-C.sub.6 alkyl group; [0175] A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; [0176] Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; [0177] ring G represents the following formula:
[0177] ##STR00014## [0178] R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; [0179] a partial structure in formula (I) represented by the following formula:
[0179] ##STR00015## [0180] R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; and [0181] R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and [0182] L represents a single bond, or a salt thereof.
[0183] Clause 2. The method of clause 1, wherein the cancer is Breast cancer brain metastases, Astrocytoma, Atypical Teratoid Rhabdoid Tumor (ATRT), Chondrosarcoma, Choroid Plexus Carcinoma, Craniopharyngioma, Ependymoma, Germ Cell Tumor, Glioblastoma, Glioma, Hemangioma, Juvenile Pilocytic Astrocytoma, Medulloblastoma, Meningioma, Neurofibroma, Neuronal and Mixed Neuronal-Glial Tumors, Oligoastrocytoma, Oligodendroglioma, Pineal Tumor, Pituitary Tumor, PNET--(primitive neuroectodermal tumor), Schwannoma, and Leptomeningeal metastases.
[0184] Clause 3. A method of treating breast cancer brain metastasis in a subject, the method comprising administering a compound represented by the following formula I:
##STR00016##
wherein [0185] TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C.sub.1-C.sub.6 alkyl group; [0186] A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; [0187] Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; [0188] ring G represents the following formula:
[0188] ##STR00017## [0189] R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; [0190] a partial structure in formula (I) represented by the following formula:
[0190] ##STR00018## [0191] R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; and [0192] R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and [0193] L represents a single bond, or a salt thereof.
[0194] Clause 4. A method of treating a cancer in a subject, wherein the cancer is resistant to an estrogen receptor modulator, the method comprising administering a compound represented by the following formula I:
##STR00019##
wherein [0195] TZ represents a C.sub.1-C.sub.4 alkylene group or --CR.sup.f'R.sup.g'--CH.sub.2--O-- wherein R.sup.f' and R.sup.g' independently represent hydrogen or a C.sub.1-C.sub.6 alkyl group; [0196] A represents a 5- to 14-membered heteroarylene group which may have a substituent or a C.sub.6-C.sub.14 arylene group which may have a substituent; [0197] Y represents --CH.sub.2--NR.sup.c-- wherein R.sup.c represents hydrogen or a C.sub.1-C.sub.6 alkyl group which may have a substituent; [0198] ring G represents the following formula:
[0198] ##STR00020## [0199] R' represents 1 to 4 substituents independently selected from a hydrogen atom, a C.sub.1-C.sub.6 alkoxy group, and a hydroxyl group; [0200] a partial structure in formula (I) represented by the following formula:
[0200] ##STR00021## [0201] R'' represents hydrogen, a hydroxyl group that may be further protected by a protecting group or a C.sub.1-C.sub.6 alkoxy group which may have a substituent; and [0202] R.sup.a and R.sup.b are the same as or different from each other and each represents a hydrogen atom, a C.sub.1-C.sub.6 alkyl group which may have a substituent, or a C.sub.3-C.sub.8 cycloalkyl group which may have a substituent, or when R.sup.a and R.sup.b are bonded together, they may form, together with the nitrogen atom that is adjacent to R.sup.a and R.sup.b, a 4- to 10-membered single ring which may have a substituent; and [0203] L represents a single bond, or a salt thereof.
[0204] Clause 5. The method of clause 4, wherein the cancer is de novo resistant to the estrogen receptor modulator.
[0205] Clause 6. The method of clause 4, wherein the resistance to the estrogen receptor modulator is acquired.
[0206] Clause 7. The method of clause 4, wherein the estrogen receptor modulator is a selective estrogen receptor modulator (SERM).
[0207] Clause 8. The method of clause 7, wherein the SERM is tamoxifen, idoxifene, raloxifene or ICI 182,780.
[0208] Clause 9. The method of any one of clauses 4-8, wherein the cancer is breast, endometrial or ovarian cancer.
[0209] Clause 10. The method of any one of clauses 4-9, wherein the cancer is breast cancer.
[0210] Clause 11. The method of any one of clauses 1-10, wherein R.sup.a and R.sup.b independently represent a hydrogen atom, a methyl group, an ethyl group, a n-propyl group, an iso-propyl group, a n-butyl group, an iso-butyl group, or a tert-butyl group.
[0211] Clause 12. The method of any one of clauses 1-10, wherein -T-Z-- represents --CH.sub.2CH.sub.2-- or --C(CH.sub.3).sub.2CH.sub.2O--.
[0212] Clause 13. The method of any one of clauses 1-10, wherein Y represents --CH.sub.2--N(CH.sub.2CH.sub.3)-- or --CH.sub.2--N(CH.sub.2CH.sub.2OH)--.
[0213] Clause 14. The method of any one of clauses 1-10, wherein each of R'' independently represents a hydrogen atom or a methoxy group.
[0214] Clause 15. The method of any one of clauses 1-10, wherein R'' represents a hydroxyl group.
[0215] Clause 16. The method of any one of clauses 1-10, wherein A represents a phenylene group.
[0216] Clause 17. The method of any one of clauses 1-10, wherein the compound is (R)-6-{2-{ethyl[4-(2-ethylaminoethyl)benzyl]amino}-4-methoxyphenyl}-5,6,7- ,8-tetrahydronaphthalen-2-ol.
[0217] Clause 18. The method of any one of clauses 1-10, wherein an effective amount of the compound is administered.
[0218] Clause 19. The method of any one of clauses 1-10, wherein the effective amount comprises a high dosage.
[0219] Clause 20. The method of clause 19, wherein the high dosage is more than about 20 mg/kg.
[0220] Clause 21. The method of clause 19 or 20, wherein the high dosage is about 20 mg/kg to about 100 mg/kg.
[0221] Clause 22. The method of any one of clauses 1-10, wherein the compound is administered by oral administration, intravenous administration, intradermal injection, intramuscular injection, or subcutaneous injection.
[0222] Clause 23. The method of any one of clauses 1-10, further comprising administering an effective amount of at least one compound selected from the group consisting of a cyclin-dependent kinase 4 and 6 inhibitor (CDK4/6 inhibitor), an antiestrogen, a ligand of retinoic acid or retinoxic X receptor, an antiprogestin, an antiandrogen, vitamin D or metabolite thereof, a farnesyl transferase inhibitor, a PPAR.alpha. or gamma agonist and a MAP kinase inhibitor.
* * * * *























D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011
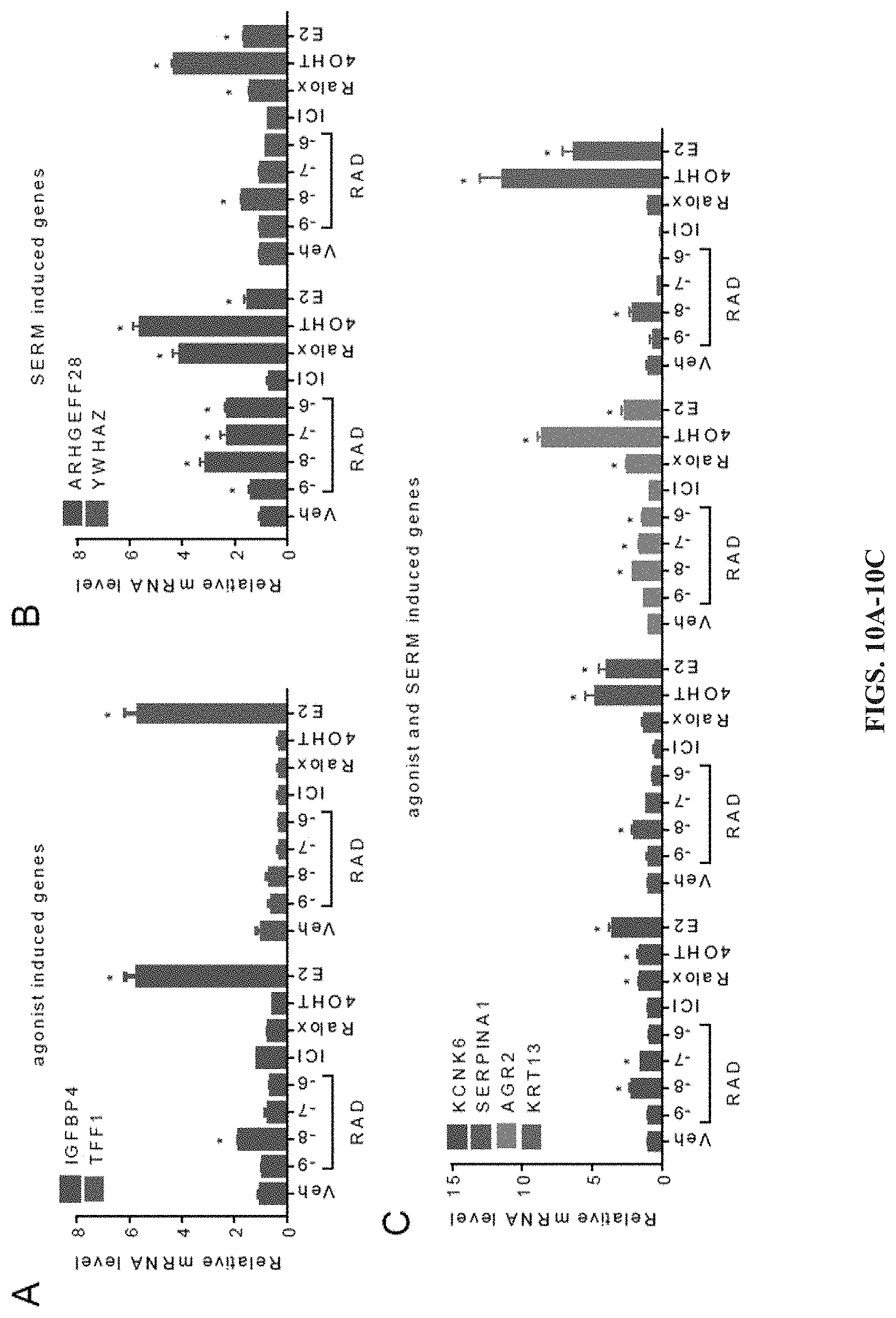
D00012

D00013

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.