Compositions Comprising Bacterial Strains
MULDER; Imke Elisabeth ; et al.
U.S. patent application number 17/406339 was filed with the patent office on 2022-03-31 for compositions comprising bacterial strains. The applicant listed for this patent is 4D Pharma Research Limited. Invention is credited to Suaad AHMED, Anna ETTORRE, Parthena FOTIADOU, Imke Elisabeth MULDER, Helene SAVIGNAC, Samantha YUILLE.
| Application Number | 20220096565 17/406339 |
| Document ID | / |
| Family ID | 1000006016840 |
| Filed Date | 2022-03-31 |






| United States Patent Application | 20220096565 |
| Kind Code | A1 |
| MULDER; Imke Elisabeth ; et al. | March 31, 2022 |
COMPOSITIONS COMPRISING BACTERIAL STRAINS
Abstract
The invention provides compositions comprising bacterial strains for treating and preventing a neurodegenerative disorder.
| Inventors: | MULDER; Imke Elisabeth; (Aberdeen, GB) ; ETTORRE; Anna; (Aberdeen, GB) ; AHMED; Suaad; (Aberdeen, GB) ; FOTIADOU; Parthena; (Aberdeen, GB) ; YUILLE; Samantha; (Aberdeen, GB) ; SAVIGNAC; Helene; (Aberdeen, GB) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000006016840 | ||||||||||
| Appl. No.: | 17/406339 | ||||||||||
| Filed: | August 19, 2021 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16713969 | Dec 13, 2019 | 11123379 | ||
| 17406339 | ||||
| PCT/EP2018/065808 | Jun 14, 2018 | |||
| 16713969 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 9/19 20130101; A61K 9/0053 20130101; A61K 35/74 20130101 |
| International Class: | A61K 35/74 20060101 A61K035/74; A61K 9/00 20060101 A61K009/00; A61K 9/19 20060101 A61K009/19 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jun 14, 2017 | GB | 1709466.5 |
| Jun 15, 2017 | GB | 1709533.2 |
Claims
1.-34. (canceled)
35. A pharmaceutical composition comprising a bacterial strain of the genus Roseburia in a therapeutically effective amount for reducing a level of a proinflammatory cytokine in a subject when administered to the subject, and a pharmaceutically acceptable excipient, diluent, or carrier, wherein: (a) the bacterial strain is lyophilized, (b) the pharmaceutical composition does not comprise a therapeutically effective amount of any other strain, and (c) the pharmaceutical composition is formulated in an enteric capsule for delivery to an intestine of the subject.
36. The pharmaceutical composition of claim 35, wherein the proinflammatory cytokine is interleukin 6 (IL-6).
37. The pharmaceutical composition of claim 35, wherein the therapeutically effective amount comprises from about 1.times.10.sup.3 to about 1.times.10.sup.11 colony forming units (CFU).
38. The pharmaceutical composition of claim 35, wherein the pharmaceutical composition comprises from about 1.times.10.sup.6 to about 1.times.10.sup.11 colony forming units per gram (CFU/g), with respect to the weight of the pharmaceutical composition.
39. The pharmaceutical composition of claim 35, wherein the bacterial strain is of the species Roseburia hominis, Roseburia intestinalis, or Roseburia faecis.
40. The pharmaceutical composition of claim 35, wherein the bacterial strain comprises a 16S rRNA gene sequence that has at least 95% sequence identity to the polynucleotide sequence of SEQ ID NO:3, SEQ ID NO: 5, or SEQ ID NO:6.
41. The pharmaceutical composition of claim 35, wherein the bacterial strain comprises a 16S rRNA gene sequence that has at least 98% sequence identity to the polynucleotide sequence of SEQ ID NO:3, SEQ ID NO: 5, or SEQ ID NO:6.
42. The pharmaceutical composition of claim 35, wherein the bacterial strain comprises a 16S rRNA gene sequence of SEQ ID NO:3, SEQ ID NO: 5, or SEQ ID NO:6.
43. The pharmaceutical composition of claim 35, wherein the bacterial strain is the strain deposited under accession number NCIMB 42383, the strain deposited under accession number NCIMB 43043, or a biotype thereof.
44. The pharmaceutical composition of claim 43, wherein the biotype is a bacterial strain that has the same carbohydrate fermentation pattern as the strain deposited under accession number NCIMB 42383 or the strain deposited under accession number NCIMB 43043.
45. The pharmaceutical composition of claim 35, wherein the bacterial strain is capable of at least partially colonizing an intestine of the subject.
46. The pharmaceutical composition of claim 35, wherein the pharmaceutical composition is formulated for oral administration.
47. The pharmaceutical composition of claim 35, wherein the bacterial strain is live.
48. A pharmaceutical composition comprising a bacterial strain of the genus Roseburia in a therapeutically effective amount for reducing a level of the NF.kappa.B promoter activation in a subject and a pharmaceutically acceptable excipient, diluent, or carrier, wherein: (a) the bacterial strain is lyophilized, (b) the pharmaceutical composition does not comprise a therapeutically effective amount of any other strain, and (c) the pharmaceutical composition is formulated in an enteric capsule for delivery to an intestine of the subject.
49. A method of treating a neurodegenerative disorder in a subject in need thereof comprising administering to the subject a composition comprising a therapeutically effective amount of a bacterial strain of the genus Roseburia, wherein the bacterial strain comprises a 16S rRNA gene sequence that has at least 95% sequence identity to the polynucleotide sequence of SEQ ID NO:3, SEQ ID NO: 5, or SEQ ID NO:6, and wherein the administering is effective to treat the neurodegenerative disorder.
50. The method of claim 49, wherein the administering is effective to reduce a level of a proinflammatory cytokine in the subject.
51. The method of claim 49, wherein the proinflammatory cytokine is interleukin 6 (TL-6).
52. The method of claim 49, wherein the therapeutically effective amount comprises from about 1.times.10.sup.3 to about 1.times.10.sup.11 colony forming units (CFU).
53. The method of claim 49, wherein the composition comprises no more than de minimis amounts of other bacterial strains.
54. The method of claim 49, wherein the composition further comprises a pharmaceutically acceptable excipient, diluent, or carrier.
Description
CROSS-REFERENCE
[0001] This application is a continuation of U.S. application Ser. No. 16/713,969, filed Dec. 13, 2019, which is a continuation of International Application No. PCT/EP2018/065808, filed Jun. 14, 2018, which claims the benefit of Great Britain Application No. 1709466.5, filed Jun. 14, 2017, and Great Britain Application No. 1709533.2, filed Jun. 15, 2017 all of which are hereby incorporated by reference in their entirety.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ANSI format and is hereby incorporated by reference in its entirety. Said ANSI copy, created on Aug. 7, 2021, is named 56708-728_302_SL.txt and is 4,680,763 bytes in size.
TECHNICAL FIELD
[0003] This invention is in the field of compositions comprising bacterial strains isolated from the mammalian digestive tract and the use of such compositions in the treatment of disease.
BACKGROUND TO THE INVENTION
[0004] The human intestine is thought to be sterile in utero, but it is exposed to a large variety of maternal and environmental microbes immediately after birth. Thereafter, a dynamic period of microbial colonization and succession occurs, which is influenced by factors such as delivery mode, environment, diet and host genotype, all of which impact upon the composition of the gut microbiota, particularly during early life. Subsequently, the microbiota stabilizes and becomes adult-like [1]. The human gut microbiota contains more than 500-1000 different phylotypes belonging essentially to two major bacterial divisions, the Bacteroidetes and the Firmicutes [2]. The successful symbiotic relationships arising from bacterial colonization of the human gut have yielded a wide variety of metabolic, structural, protective and other beneficial functions. The enhanced metabolic activities of the colonized gut ensure that otherwise indigestible dietary components are degraded with release of by-products providing an important nutrient source for the host. Similarly, the immunological importance of the gut microbiota is well-recognized and is exemplified in germfree animals which have an impaired immune system that is functionally reconstituted following the introduction of commensal bacteria [3-5].
[0005] Dramatic changes in microbiota composition have been documented in gastrointestinal disorders such as inflammatory bowel disease (IBD). For example, the levels of Clostridium cluster XIVa bacteria are reduced in IBD patients whilst numbers of E. coli are increased, suggesting a shift in the balance of symbionts and pathobionts within the gut [6-9].
[0006] In recognition of the potential positive effect that certain bacterial strains may have on the animal gut, various strains have been proposed for use in the treatment of various diseases (see, for example, [10-13]). Also, certain strains, including mostly Lactobacillus and Bifidobacterium strains, have been proposed for use in treating various inflammatory and autoimmune diseases that are not directly linked to the intestines (see [14] and [15] for reviews). The ability of Roseburia hominis to regulate the immune system has been suggested in [16]. However, the relationship between different diseases and different bacterial strains, and the precise effects of particular bacterial strains on the gut and at a systemic level and on any particular types of diseases are poorly characterised, particularly for neurodegenerative disorders.
[0007] Recently, there has been increased interest in the art regarding alterations in the gut microbiome that may play a pathophysiological role in human brain diseases [17]. Preclinical and clinical evidence are strongly suggesting a link between brain development and microbiota [18]. A growing body of preclinical literature has demonstrated bidirectional signalling between the brain and the gut microbiome, involving multiple neurocrine and endocrine signalling systems. Indeed, increased levels of Clostridium species in the microbiome have been linked to brain disorders [19], and an imbalance of the Bacteroidetes and Firmicutes phyla has also been implicated in brain development disorders [20]. Suggestions that altered levels of gut commensals, including those of Bifidobacterium, Lactobacillus, Sutterella, Prevotella and Ruminococcus genera and of the Alcaligenaceae family are involved in immune-mediated central nervous system (CNS) disorders, are questioned by studies suggesting a lack of alteration in the microbiota between patients and healthy subjects [10]. Roseburia hominis has been proposed for treating a variety of disorders including asthma, rheumatoid arthritis and multiple sclerosis [21]
[0008] Like asthma and rheumatoid arthritis, multiple sclerosis is primarily mediated by the immune system. The immune system attacks myelinated axons in the central nervous system, destroying the myelin called plaques or lesions. Demyelination occurs in particular in the optic nerves, subpial spinal cord, brainstem, cerebellum, and juxtacortical and periventricular white matter regions.
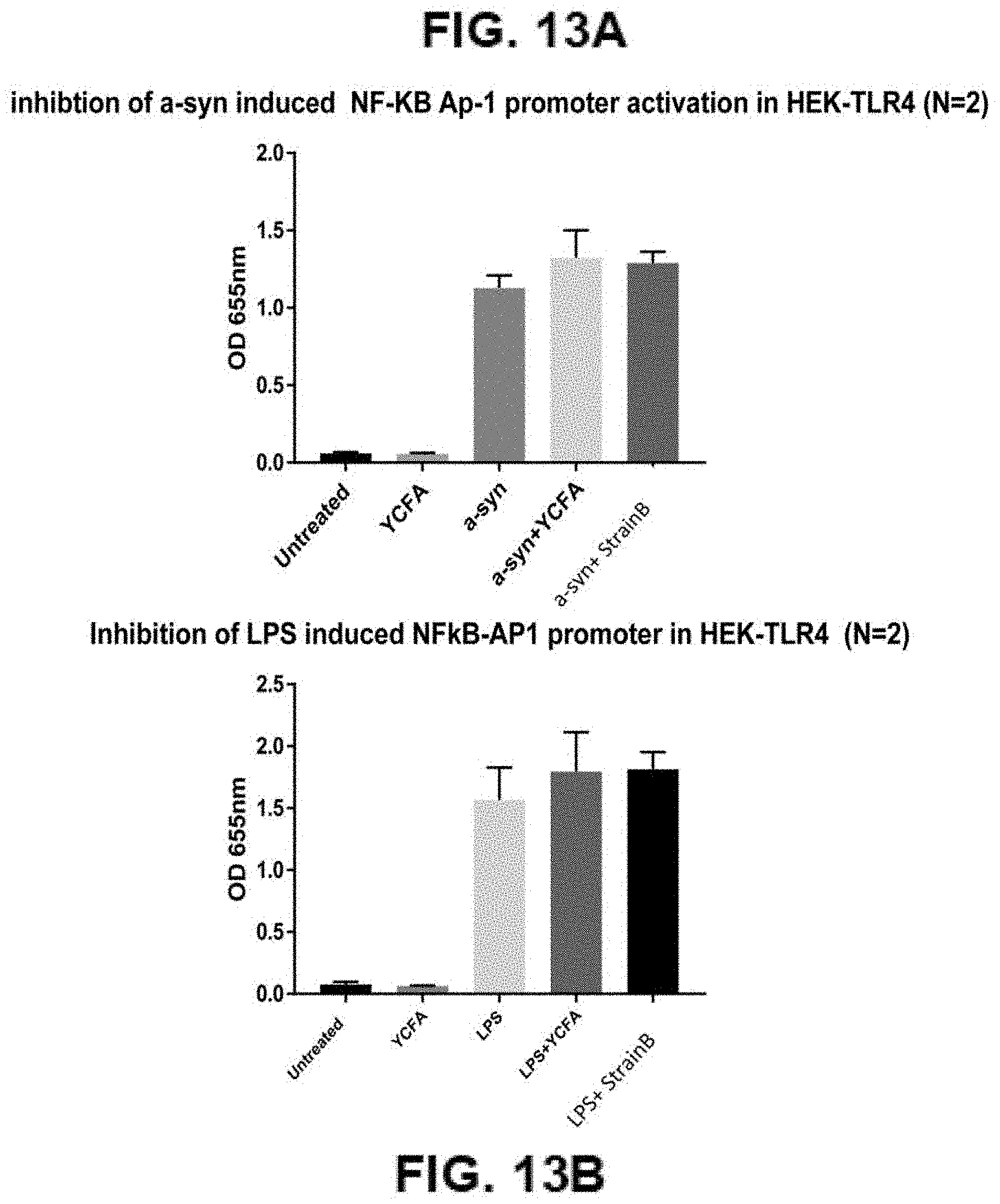
[0009] As such, multiple sclerosis has a different pathology to other neurodegenerative diseases, such as Parkinson's disease, Alzheimer's disease or dementia. For example, multiple sclerosis is commonly diagnosed in patients in their 20s and 30s, while many other neurodegenerative diseases, such as Parkinson's disease, Alzheimer's and dementia, are diagnosed predominantly in patients aged over 65 years old.
[0010] Parkinson's disease, like many neurodegenerative diseases, is primarily mediated by the accumulation of misfolded protein. Parkinson's disease is a synucleinopathology that involves the accumulation of .alpha.-synuclein, which aggregate as insoluble fibrils in Lewy bodies within the cytoplasm of the neuronal body. The accumulation of .alpha.-synuclein is toxic and impairs the functions of mitochondria, lysosomes, and endoplasmic reticulum, and interferes with microtubule transport.
[0011] Nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen, have been tested for their efficacy in treating a variety of neurological diseases, but the clinical impact of NSAIDs on neurodegenerative diseases like Parkinson's disease remains unclear. While some studies showed that chronic NSAID use is protective against Parkinson's disease, other studies could not confirm the existence of a significant relationship. A recent meta-analysis indicated that the use of non-aspirin NSAID, particularly ibuprofen, reduces the risk of PD by 15% while the use of aspirin did not show any effect [22].
[0012] This indicates that, at present, the practical effect of the link between the microbiome and human brain diseases is poorly characterised. Accordingly, more direct analytical studies are required to identify the therapeutic impact of altering the microbiome on neurodegenerative disorders. There is a requirement in the art for new methods of treating neurodegenerative disorders. There is also a requirement for the potential effects of gut bacteria to be characterised so that new therapies using gut bacteria can be developed.
SUMMARY OF THE INVENTION
[0013] The inventors have developed new therapies for treating and preventing neurodegenerative disorders. The inventors have identified that bacterial strains from the genus Roseburia may be effective for treating neurodegenerative diseases. As described in the examples, administration of compositions comprising Roseburia hominis can protect against reactive oxygen species and prevent inflammation, thus acting as a neuroprotectant. The inventors have also identified that treatment with Roseburia hominis can reduce the activation of proinflammatory molecules, such as NF.kappa.B and IL-6, by LPS and mutant .alpha.-synuclein A53T. The inventors have identified that treatment with Roseburia hominis can reduce histone deacetylation activity and lipid peroxidation in vitro, which can help to reduce cell death and apoptosis. The inventors have also identified that Roseburia hominis can produce indole that can attenuate inflammation and oxidative stress. Furthermore, the inventors have demonstrated that treatment with Roseburia hominis can increase kynurenine levels.
[0014] In a first embodiment, the invention provides a composition comprising a bacterial strain of the genus Roseburia, for use in a method of treating or preventing a neurodegenerative disorder.
[0015] In particular embodiments, the invention provides a composition comprising a bacterial strain of the genus Roseburia, for use in a method of treating or preventing a disease or condition selected from the group consisting of: Parkinson's disease, including progressive supranuclear palsy, progressive supranuclear palsy, Steele-Richardson-Olszewski syndrome, normal pressure hydrocephalus, vascular or arteriosclerotic parkinsonism and drug-induced parkinsonism; Alzheimer's disease, including Benson's syndrome; multiple sclerosis; Huntington's disease; amyotrophic lateral sclerosis; Lou Gehrig's disease; motor neurone disease; prion disease; spinocerebellar ataxia; spinal muscular atrophy; dementia, including Lewy body, vascular and frontotemporal dementia; primary progressive aphasia; mild cognitive impairment; HIV-related cognitive impairment and corticobasal degeneration.
[0016] In preferred embodiments, the invention provides a composition comprising a bacterial strain of the genus Roseburia, for use in a method of treating or preventing Parkinson's disease, such as environmental, familial or Parkinson's associated with general inflammatory status. The inventors have identified that treatment with Roseburia strains can reduce the activation of proinflammatory molecules, such as NF.kappa.B and IL-6, by LPS and mutant .alpha.-synuclein A53T in in vitro models of environmental and familial Parkinson's. In preferred embodiments, the invention provides a composition comprising a bacterial strain of the species Roseburia hominis, for use in the treatment of Parkinson's disease. Compositions using Roseburia hominis may be particularly effective for treating Parkinson's.
[0017] In preferred embodiments of the invention, the bacterial strain in the composition is of Roseburia hominis. Closely related strains may also be used, such as bacterial strains that have a 16SrRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to the 16S rRNA sequence of a bacterial strain of Roseburia hominis. Preferably, the bacterial strain has a 16S rRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to SEQ ID NO:1, 2 or 3. Preferably, the sequence identity is to SEQ ID NO:3. Preferably, the bacterial strain for use in the invention has the 16S rRNA sequence represented by SEQ ID NO:3.
[0018] In certain embodiments, the composition of the invention is for oral administration. Oral administration of the strains of the invention can be effective for neurodegenerative disorders. Also, oral administration is convenient for patients and practitioners and allows delivery to and/or partial or total colonisation of the intestine.
[0019] In certain embodiments, the composition of the invention comprises one or more pharmaceutically acceptable excipients or carriers.
[0020] In certain embodiments, the composition of the invention comprises a bacterial strain that has been lyophilised. Lyophilisation is an effective and convenient technique for preparing stable compositions that allow delivery of bacteria.
[0021] In certain embodiments, the invention provides a food product comprising the composition as described above.
[0022] In certain embodiments, the invention provides a vaccine composition comprising the composition as described above.
[0023] Additionally, the invention provides a method of treating or preventing neurodegenerative disorders, comprising administering a composition comprising a bacterial strain of the genus Roseburia.
[0024] In certain embodiments of the invention, the composition is for use in treating brain injury. The neuroprotective activity of the compositions of the invention and their ability to reduce levels of histone deacetylase activity (HDAC) may make them useful for treating brain injury. In preferred embodiments, the compositions of the invention are for use in treating stroke, such as treating brain injury resulting from a stroke.
[0025] In developing the above invention, the inventors have identified and characterised a bacterial strain that is particularly useful for therapy. The Roseburia intestinalis strain of the invention is shown to be effective for treating cancer. The invention also provides compositions comprising such cells, or biologically pure cultures of such cells. The invention also provides a cell of the Roseburia intestinalis strain deposited under accession number NCIMB 43043, or a derivative thereof, for use in therapy, in particular for cancer.
BRIEF DESCRIPTION OF DRAWINGS
[0026] FIG. 1: Downregulation of IL-6 secretion
[0027] FIG. 2: Inhibition of .alpha.-synuclein induced NF.kappa.B promoter activation
[0028] FIG. 3: Inhibition of LPS induced NF.kappa.B promoter activation
[0029] FIG. 4: Change in antioxidant capacity
[0030] FIG. 5: Change in total anti-oxidant capacity (lipid oxidation)
[0031] FIG. 6: Change in histone deacetylatase (HDAC) activity
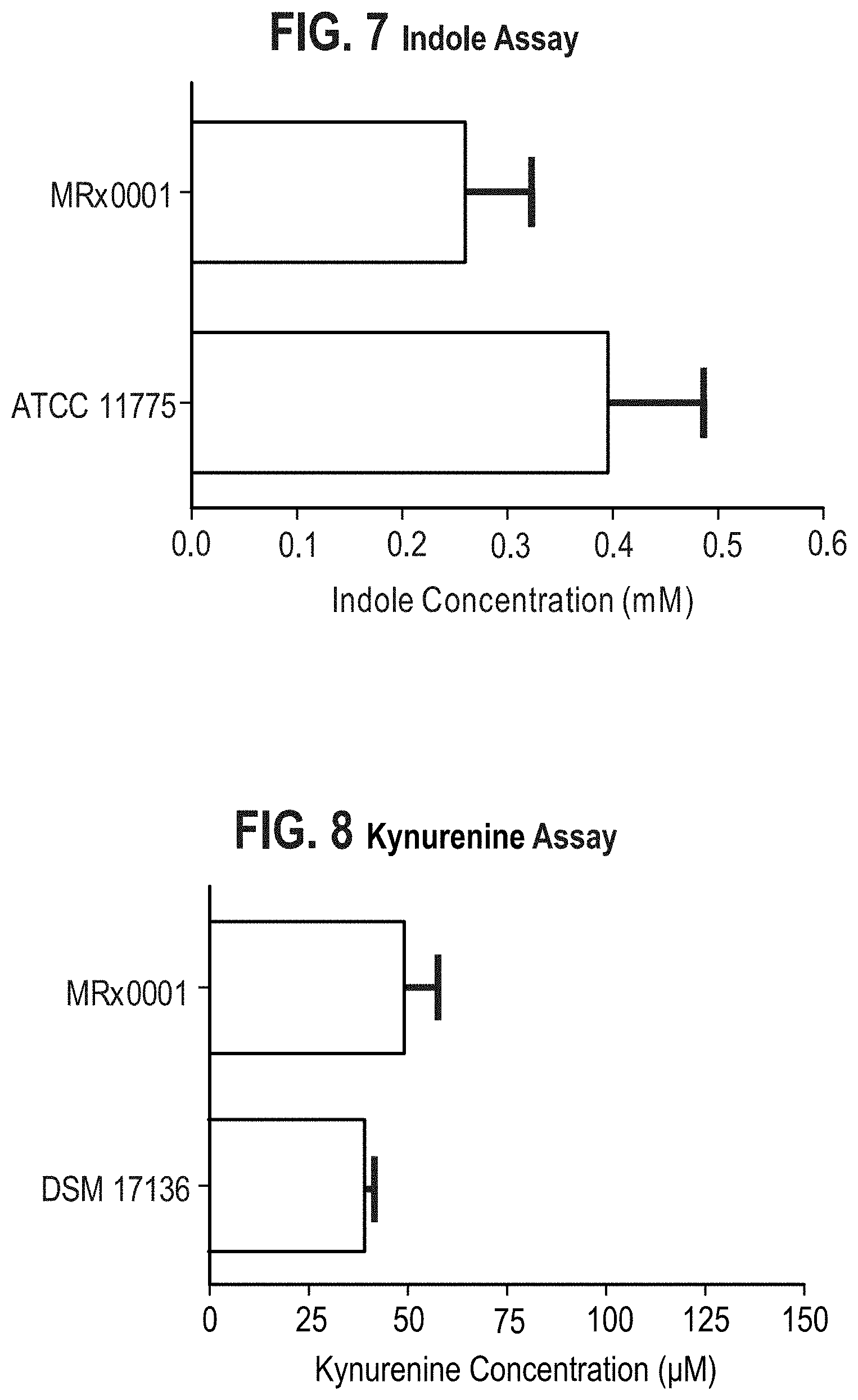
[0032] FIG. 7: Level of Indole production
[0033] FIG. 8: Level of Kyrunenine production
[0034] FIG. 9: Downregulation of IL-6 secretion by MIRX001
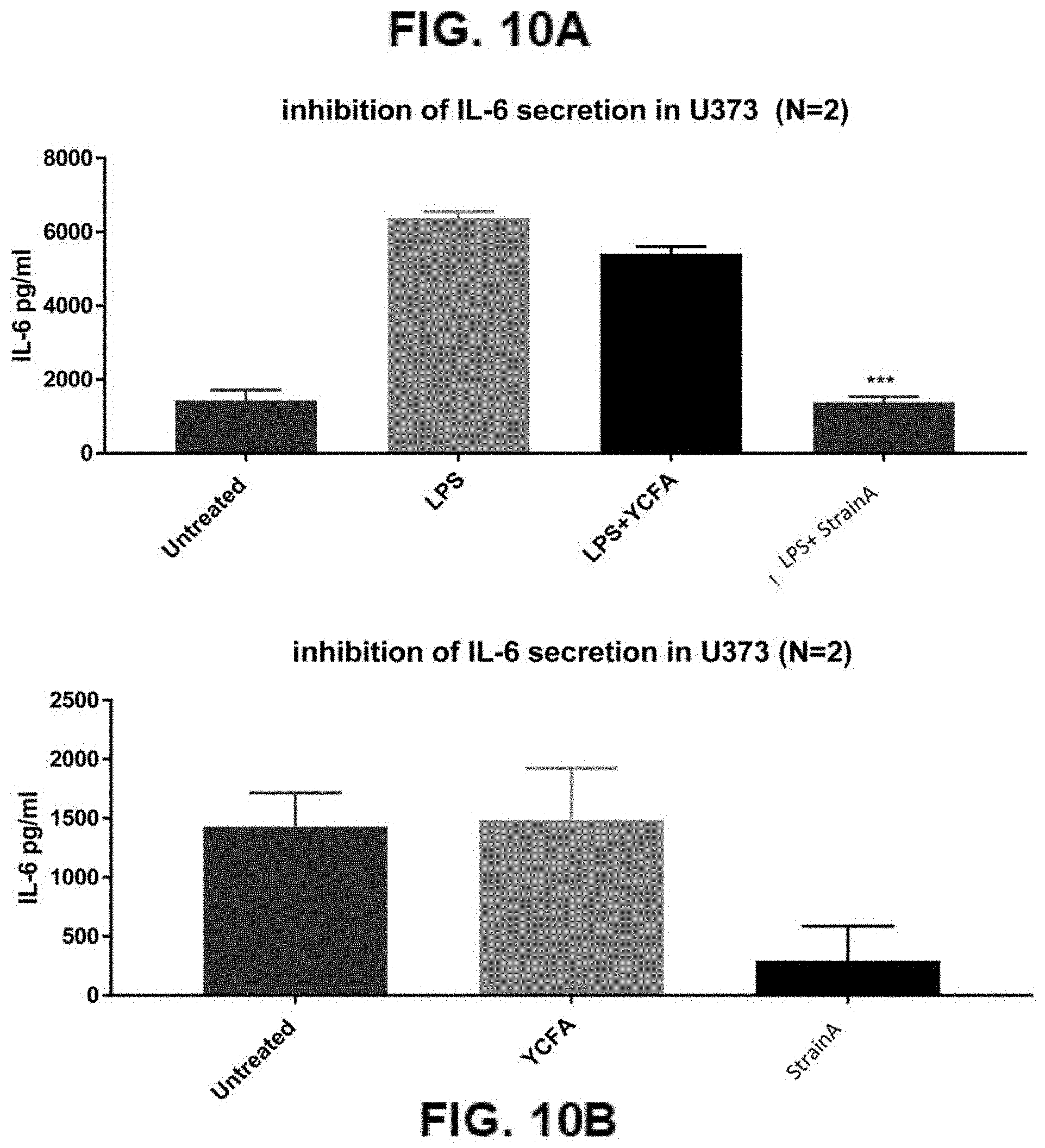
[0035] FIGS. 10A-10B: Downregulation of IL-6 secretion by strain A: Inhibition of IL-6 secretion in U373 (FIG. 10A); Inhibition of IL-6 secretion in U373 (FIG. 10B)
[0036] FIGS. 11A-11B: Inhibition of .alpha.-synuclein induced NF.kappa.B promoter activation (FIG. 11A) and Inhibition of LPS induced NF.kappa.B promoter activation by strain A (FIG. 11B)
[0037] FIGS. 12A-12B: Downregulation of IL-6 secretion by strain B: Inhibition of IL-6 secretion in U373 (FIG. 12A); Inhibition of IL-6 secretion in U373 (FIG. 12B)
[0038] FIGS. 13A-13B: Inhibition of .alpha.-synuclein induced NF.kappa.B promoter activation (FIG. 13A) and Inhibition of LPS induced NF.kappa.B promoter activation by strain B (FIG. 13B)
DISCLOSURE OF THE INVENTION
[0039] Bacterial Strains
[0040] The compositions of the invention comprise a bacterial strain of the genus Roseburia. The examples demonstrate that bacteria of this genus are useful for treating or preventing neurodegenerative disorders. The preferred bacterial strains are of the species Roseburia hominis, Roseburia faecis and Roseburia intestinalis.
[0041] Examples of Roseburia species for use in the invention include Roseburia hominis, Roseburia cecicola, Roseburia faecis, Roseburia intestinalis, and Roseburia inulinivorans. Roseburia bacteria are slightly curved rod-shaped cells that are strictly anaerobic and indigenous to the mammalian intestine. They are of the phylogenetic cluster XIVa within the Firmicutes phylum. The bacteria are butyrate-producing and are actively motile through multiple flagella present along the concave side and in a cluster at one end [23]. Roseburia hominis and Roseburia intestinalis are recently described examples.
[0042] An example of Roseburia hominis is the strain deposited under the terms of the Budapest Treaty at National Collections of Industrial, Food and Marine Bacteria (NCIMB) at NCIMB Ltd, Ferguson Building, Craibstone Estate, Bucksburn, Aberdeen, UK, AB21 9YA, on 21 Oct. 2004 by the Rowett Research Institute under the accession number NCIMB 14029.sup.T Roseburia hominis A2-183.sup.T(DSM=16839.sup.T). Other exemplary Roseburia hominis strains are described in [24]. GenBank/EMBL/DDBJ accession numbers for the 16S rRNA gene sequence of strains of Roseburia hominis are AY804148 and AJ270482 (disclosed herein as SEQ ID NO:1 and SEQ ID NO:2).
[0043] An example of Roseburia intestinalis is the strain deposited under the accession number NCIMB 13810 Roseburia intestinalis L1-82.sup.T(DSM=14610.sup.T). Another example is the Roseburia intestinalis strain as described in [24]. Reference [24] also describes exemplary Roseburia faecis and Roseburia inulinivorans strains.
[0044] The Roseburia hominis bacterium deposited under accession number NCIMB 42383 was tested in the Examples and is also referred to herein as strain 433. A 16S rRNA sequence for the 433 strain that was tested is provided in SEQ ID NO:3. Strain 433 was deposited with the international depositary authority NCIMB, Ltd. (Ferguson Building, Aberdeen, AB21 9YA, Scotland) by GT Biologics Ltd. (Life Sciences Innovation Building, Aberdeen, AB25 2ZS, Scotland) on 12 Mar. 2015 as "Roseburia hominis 433" and was assigned accession number NCIMB 42383. GT Biologics Ltd. subsequently changed its name to 4D Pharma Research Limited.
[0045] WO 2016/203221 describes administration of strain 433 to mice and shows that it can affect disease processes outside of the gut (such as asthma and arthritis). Strain 433 also affects disease processes outside of the gut in the treatment of neurodegenerative disorders described herein.
[0046] A genome sequence for strain 433 is provided in SEQ ID NO:4. This sequence was generated using the PacBio RS II platform.
[0047] The Roseburia intestinalis bacterium deposited under accession number NCIMB 43043 was tested in the Examples and is also referred to herein as strain A. A 16S rRNA sequence for strain A that was tested is provided in SEQ ID NO:5. Strain A was deposited with the international depositary authority NCIMB, Ltd. (Ferguson Building, Aberdeen, AB21 9YA, Scotland) by 4D Pharma Research Limited (Life Sciences Innovation Building, Aberdeen, AB25 2ZS, Scotland) on 3 May 2018 as "Roseburia intestinalis" and was assigned accession number NCIMB 43043.
[0048] The examples also describe a Roseburia faecis bacterium that was tested and referred to as strain B. A 16S rRNA sequence for strain B that was tested is provided in SEQ ID NO:6.
[0049] In certain embodiments, the bacterial strain for use in the invention is Roseburia hominis. In certain embodiments, the bacterial strain for use in the invention is Roseburia intestinalis. In certain embodiments, the bacterial strain for use in the invention is Roseburia faecis.
[0050] Bacterial strains closely related to the strain tested in the examples are also expected to be effective for treating or preventing neurodegenerative disease diseases and conditions mediated by IL-17 or the Th17 pathway. In certain embodiments, the bacterial strain for use in the invention has a 16S rRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to the 16S rRNA sequence of a bacterial strain of Roseburia hominis. Preferably, the bacterial strain for use in the invention has a 16S rRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to SEQ ID NO: 1, 2 or 3. Preferably, the sequence identity is to SEQ ID NO:3. Preferably, the bacterial strain for use in the invention has the 16S rRNA sequence represented by SEQ ID NO:3.
[0051] In other embodiments, the bacterial strain for use in the invention has a 16S rRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to the 16S rRNA sequence of a bacterial strain of Roseburia intestinalis. Preferably, the bacterial strain for use in the invention has a 16S rRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to SEQ ID NO:5. Preferably, the bacterial strain for use in the invention has the 16S rRNA sequence represented by SEQ ID NO:5.
[0052] In other embodiments, the bacterial strain for use in the invention has a 16S rRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to the 16S rRNA sequence of a bacterial strain of Roseburia faecis. Preferably, the bacterial strain for use in the invention has a 16S rRNA sequence that is at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% identical to SEQ ID NO:6. Preferably, the bacterial strain for use in the invention has the 16S rRNA sequence represented by SEQ ID NO:6.
[0053] Bacterial strains that are biotypes of the bacterium deposited under accession number 42383 or NCIMB 43043 are also expected to be effective for treating or preventing neurodegenerative disorders. A biotype is a closely related strain that has the same or very similar physiological and biochemical characteristics.
[0054] Strains that are biotypes of the bacterium deposited under accession number NCIIB 42383 or NCIMB 43043 and that are suitable for use in the invention may be identified by sequencing other nucleotide sequences for the bacterium deposited under accession number NCIIB 42383 or NCIMB 43043. For example, substantially the whole genome may be sequenced and a biotype strain for use in the invention may have at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% sequence identity across at least 80% of its whole genome (e.g. across at least 85%, 90%, 95% or 99%, or across its whole genome). Other suitable sequences for use in identifying biotype strains may include hsp60 or repetitive sequences such as BOX, ERIC, (GTG).sub.5 (SEQ ID NO: 7), or REP or [25]. Biotype strains may have sequences with at least 95%, 96%, 97%, 98%, 99%, 99.5% or 99.9% sequence identity to the corresponding sequence of the bacterium deposited under accession number NCIIB 42383 or NCIMB 43043.
[0055] In certain embodiments, the bacterial strain for use in the invention has a genome with sequence identity to SEQ ID NO:4. In preferred embodiments, the bacterial strain for use in the invention has a genome with at least 90% sequence identity (e.g. at least 92%, 94%, 95%, 96%, 97%, 98%, 99% or 100% sequence identity) to SEQ ID NO:4 across at least 60% (e.g. at least 65%, 70%, 75%, 80%, 85%, 95%, 96%, 97%, 98%, 99% or 100%) of SEQ ID NO:4. For example, the bacterial strain for use in the invention may have a genome with at least 90% sequence identity to SEQ ID NO:4 across 70% of SEQ ID NO:4, or at least 90% sequence identity to SEQ ID NO:4 across 80% of SEQ ID NO:4, or at least 90% sequence identity to SEQ ID NO:4 across 90% of SEQ ID NO:4, or at least 90% sequence identity to SEQ ID NO:4 across 100% of SEQ ID NO:4, or at least 95% sequence identity to SEQ ID NO:4 across 70% of SEQ ID NO:4, or at least 95% sequence identity to SEQ ID NO:4 across 80% of SEQ ID NO:4, or at least 95% sequence identity to SEQ ID NO:4 across 90% of SEQ ID NO:4, or at least 95% sequence identity to SEQ ID NO:4 across 100% of SEQ ID NO:4, or at least 98% sequence identity to SEQ ID NO:4 across 70% of SEQ ID NO:4, or at least 98% sequence identity to SEQ ID NO:4 across 80% of SEQ ID NO:4, or at least 98% sequence identity to SEQ ID NO:4 across 90% of SEQ ID NO:4, or at least 98% sequence identity to SEQ ID NO:4 across 100% of SEQ ID NO:4.
[0056] Alternatively, strains that are biotypes of the bacterium deposited under accession number NCIMB 42383 or NCIMB 43043 and that are suitable for use in the invention may be identified by using the accession number NCIMB 42383 or NCIMB 43043 deposit and restriction fragment analysis and/or PCR analysis, for example by using fluorescent amplified fragment length polymorphism (FAFLP) and repetitive DNA element (rep)-PCR fingerprinting, or protein profiling, or partial 16S or 23S rDNA sequencing. In preferred embodiments, such techniques may be used to identify other Roseburia hominis, Roseburia faecis and Roseburia intestinalis strains.
[0057] In certain embodiments, strains that are biotypes of the bacterium deposited under accession number NCIMB 42383 or NCIMB 43043 and that are suitable for use in the invention are strains that provide the same pattern as the bacterium deposited under accession number NCIMB 42383 or NCIMB 43043 when analysed by amplified ribosomal DNA restriction analysis (ARDRA), for example when using Sau3AI restriction enzyme (for exemplary methods and guidance see, for example, [25]).
[0058] Alternatively, biotype strains are identified as strains that have the same carbohydrate fermentation patterns as the bacterium deposited under accession number NCIMB 42383 or NCIMB 43043. Other Roseburia strains that are useful in the compositions and methods of the invention, such as biotypes of the bacteria deposited under accession number NCIMB 42383 or NCIMB 43043, may be identified using any appropriate method or strategy, including the assays described in the examples. For instance, strains for use in the invention may be identified by culturing with neuroblastoma cells and then assessing cytokine levels and levels of neuroprotection or neuroproliferation. In particular, bacterial strains that have similar growth patterns, metabolic type and/or surface antigens to the bacterium deposited under accession number NCIMB 42383 or NCIMB 43043 may be useful in the invention. A useful strain will have comparable immune modulatory activity to the NCIMB 42383 or NCIMB 43043 strain. In particular, a biotype strain will elicit comparable effects on the neurodegenerative disease models and comparable effects on cytokine levels to the effects shown in the Examples, which may be identified by using the culturing and administration protocols described in the Examples.
[0059] A particularly preferred strain of the invention is the Roseburia hominis strain deposited under accession number NCIMB 42383. This is the exemplary 433 strain tested in the examples and shown to be effective for treating disease. Therefore, the invention provides a cell, such as an isolated cell, of the Roseburia hominis strain deposited under accession number NCIMB 42383, or a derivative thereof. The invention also provides a composition comprising a cell of the Roseburia hominis strain deposited under accession number NCIMB 42383, or a derivative thereof. The invention also provides a biologically pure culture of the Roseburia hominis strain deposited under accession number NCIMB 42383. The invention also provides a cell of the Roseburia hominis strain deposited under accession number NCIMB 42383, or a derivative thereof, for use in therapy, in particular for the diseases described herein.
[0060] A particularly preferred strain of the invention is the Roseburia intestinalis strain deposited under accession number NCIMB 43043. This is the exemplary strain A tested in the examples and shown to be effective for treating disease. Therefore, the invention provides a cell, such as an isolated cell, of the Roseburia intestinalis strain deposited under accession number NCIMB 43043, or a derivative thereof. The invention also provides a composition comprising a cell of the Roseburia intestinalis strain deposited under accession number NCIMB 43043, or a derivative thereof. The invention also provides a biologically pure culture of the Roseburia intestinalis strain deposited under accession number NCIMB 43043. The invention also provides a cell of the Roseburia intestinalis strain deposited under accession number NCIMB 43043, or a derivative thereof, for use in therapy, in particular for the diseases described herein.
[0061] A derivative of the strain deposited under accession number NCIMB 42383 or NCIMB 43043 may be a daughter strain (progeny) or a strain cultured (subcloned) from the original. A derivative of a strain of the invention may be modified, for example at the genetic level, without ablating the biological activity. In particular, a derivative strain of the invention is therapeutically active. A derivative strain will have comparable immune modulatory activity to the original NCIMB 42383 or NCIMB 43043 strain. In particular, a derivative strain will elicit comparable effects on the neurodegenerative disease models and comparable effects on cytokine levels to the effects shown in the Examples, which may be identified by using the culturing and administration protocols described in the Examples. A derivative of the NCIMB 42383 or NCIMB 43043 strain will generally be a biotype of the NCIMB 42383 or NCIMB 43043 strain.
[0062] References to cells of the Roseburia hominis strain deposited under accession number NCIMB 42383 encompass any cells that have the same safety and therapeutic efficacy characteristics as the strains deposited under accession number NCIMB 42383, and such cells are encompassed by the invention.
[0063] References to cells of the Roseburia intestinalis strain deposited under accession number NCIMB 43043 encompass any cells that have the same safety and therapeutic efficacy characteristics as the strains deposited under accession number NCIMB 43043, and such cells are encompassed by the invention.
[0064] In preferred embodiments, the bacterial strains in the compositions of the invention are viable and capable of partially or totally colonising the intestine.
[0065] The invention further provides a cell, such as an isolated cell, of the Enterococcus gallinarum strain deposited under accession number NCIMB 42761, or a derivative thereof. The invention also provides a composition comprising a cell of the Roseburia intestinalis strain deposited under accession number NCIMB 42761, or a derivative thereof. The invention also provides a biologically pure culture of the Roseburia intestinalis strain deposited under accession number NCIMB 43043. The invention also provides a cell of the Roseburia intestinalis strain deposited under accession number NCIMB 43043, or a derivative thereof, for use in therapy, in particular for the diseases described herein. A derivative of the strain deposited under accession number NCIMB 43043 may be a daughter strain (progeny) or a strain cultured (subcloned) from the original.
[0066] A derivative of a strain of the invention may be modified, for example at the genetic level, without ablating the biological activity. In particular, a derivative strain of the invention is therapeutically active. A derivative strain will have comparable immune modulatory activity to the original NCIMB 42761 strain. In particular, a derivative strain will elicit comparable effects on the neurodegenerative disease models to the effects shown in the Examples, which may be identified by using the culturing and administration protocols described in the Examples. A derivative of the NCIMB 43043 strain will generally be a biotype of the NCIMB 43043 strain.
[0067] References to cells of the Roseburia intestinalis strain deposited under accession number NCIMB 43043 encompass any cells that have the same safety and therapeutic efficacy characteristics as the strain deposited under accession number NCIMB 43043, and such cells are encompassed by the invention. Thus, in some embodiments, reference to cells of the Roseburia intestinalis strain deposited under accession number NCIMB 43043 refers only to the strain A deposited under NCIMB 43043 and does not refer to a bacterial strain that was not deposited under NCIMB 43043.
[0068] In preferred embodiments, the bacterial strain in the compositions of the invention is viable and capable of partially or totally colonising the intestine.
[0069] Therapeutic Uses
[0070] As demonstrated in the examples, the bacterial compositions of the invention are effective for treating neurodegenerative disorders. In particular, treatment with compositions of the invention increase neuroproliferation and act as a neuroprotectant against agents that destroy dopaminergic neurons. Therefore, the compositions of the invention may be useful for treating or preventing neurodegenerative disorders that are the result of neuron death.
[0071] Compositions of the invention can decrease the activation of the NF.kappa.B promoter, which activates cytokine production, for example IL-1.beta., IL-1.alpha., IL-18, TNF.alpha. and IL-6. Treating cells with mutant .alpha.-synuclein is a model for familial Parkinson's. A point mutation at position 53 from adenine to threonine leads to .alpha.-synuclein mis-folding. The incorrectly folded .alpha.-synuclein subsequently aggregates into insoluble fibrils which form Lewy bodies. Therefore, the compositions of the invention may be useful for treating or preventing neurodegenerative disorders that are the result of neuroinflammation, protein misfolding and/or environmental exposure. Compositions of the invention can be used for treatment of familial Parkinson's. Activation of the NF.kappa.B promoter is mediated through the TLR4 ligand. TL4 is known to mediate cell death in the mouse model MPTP, which simulates Parkinson's disease. Compositions of the invention can be used to inhibit the ability of TLR4 signalling to activate the NF.kappa.B promoter. Of particular relevance for PD, both TLR2 and TLR4 were found to be upregulated in brains of PD patients [26]. Moreover .alpha.-syn has been described as a ligand for TLR2 [27] and we have demonstrated that .alpha.-syn is also a ligand for TLR4 using HEK-TLR4 cells.
[0072] Compositions of the invention decrease the secretion of pro-inflammatory cytokines such as IL-6, which can be induced by lipopolysaccharide (LPS). Treatment of cells with LPS simulates Parkinson's caused by environmental factors. Compositions of the invention can be used to decrease IL-6 secretion. Compositions of the invention can be used for treatment of environmental Parkinson's.
[0073] Examples of neurodegenerative diseases to be treated by compositions of the invention include: Parkinson's disease, including progressive supranuclear palsy, progressive supranuclear palsy, Steele-Richardson-Olszewski syndrome, normal pressure hydrocephalus, vascular or arteriosclerotic parkinsonism and drug-induced parkinsonism; Alzheimer's disease, including Benson's syndrome; multiple sclerosis; Huntington's disease; amyotrophic lateral sclerosis; Lou Gehrig's disease; motor neurone disease; prion disease; spinocerebellar ataxia; spinal muscular atrophy; dementia, including Lewy body, vascular and frontotemporal dementia; primary progressive aphasia; mild cognitive impairment; HIV-related cognitive impairment and corticobasal degeneration. A further neurodegenerative diseases to be treated by compositions of the invention is progressive inflammatory neuropathy.
[0074] In certain embodiments, the compositions of the invention can be effective for treating neurodegenerative disorders that occur in elderly patients. The examples show that compositions of the invention can treat Parkinson's disease which is predominantly diagnosed in patients aged over 65 years old. In preferred embodiments, the compositions of the invention are for treating patients 65 years or older. In other certain embodiments, the patients are between 40 to 65 years old. In other embodiments, the patients are older than 40 years. In certain embodiments, the compositions of the invention are for use in treating a disease associated with old age, for example, a disease diagnosed after 50 years of age.
[0075] In certain embodiments, the compositions of the invention are for use in treating a neurodegenerative disorder mediated or characterised by the accumulation of protein, in particular mis-folded protein.
[0076] In certain embodiments, the compositions of the invention are for use in treating a neurodegenerative disorder associated with grey matter neuronal loss. In certain embodiments, the compositions of the invention are for treating a neurodegenerative disorder that is not associated with white matter lesions.
[0077] In certain embodiments, the compositions of the invention are for use in treating a neurodegenerative disorder associated with permanent symptoms.
[0078] In certain embodiments, the compositions of the invention are for use in treating a neurodegenerative disorder that is not an auto-immune disorder. In certain embodiments, the compositions of the invention are for use in treating a neurodegenerative disorder that is not multiple sclerosis.
[0079] In certain embodiments, the compositions of the invention are for use in reducing neuron death, in particular, in the treatment of neurodegenerative disorders. In certain embodiments, the compositions of the invention are for use in protecting neurons, in particular in the treatment of neurodegenerative disorders.
[0080] The neuroprotective properties of the compositions of the invention, as shown in the examples, mean that the compositions may be particularly effective for preventing or delaying onset or progression of neurodegenerative disorders. In certain embodiments, the compositions of the invention are for use in delaying onset or progression of a neurodegenerative disorders.
[0081] Modulation of the Microbiota-Gut-Brain Axis
[0082] Communication between the gut and the brain (the microbiota-gut-brain axis) occurs via a bidirectional neurohumoral communication system. Recent evidence shows that the microbiota that resides in the gut can modulate brain development and produce behavioural phenotypes via the microbiota-gut-brain axis. Indeed, a number of reviews suggest a role of the microbiota-gut-brain axis in maintaining central nervous system functionality and implicate dysfunction of the microbiota-gut-brain axis in the development of central nervous system disorders and conditions [17], [20], [28].
[0083] The bidirectional communication between the brain and the gut (i.e. the-gut-brain axis) includes the central nervous system, neuroendocrine and neuroimmune systems, including the hypothalamus-pituitary-adrenal (HPA) axis, sympathetic and parasympathetic arms of the autonomic nervous system (ANS), including the enteric nervous system (ENS) and the vagus nerve, and the gut microbiota.
[0084] As demonstrated in the examples, the compositions of the present invention can modulate the microbiota-gut-brain axis and reduce cell death associated with neurodegenerative disorders. Accordingly, the compositions of the invention may be useful for treating or preventing neurodegenerative disorders, in particular those disorders and conditions associated with dysfunction of the microbiota-gut-brain axis.
[0085] In particular embodiments, the compositions of the invention may be useful for treating or preventing a disease or condition selected from the group consisting of: Parkinson's disease, including progressive supranuclear palsy, progressive supranuclear palsy, Steele-Richardson-Olszewski syndrome, normal pressure hydrocephalus, vascular or arteriosclerotic parkinsonism and drug-induced parkinsonism; Alzheimer's disease, including Benson's syndrome; multiple sclerosis; Huntington's disease; amyotrophic lateral sclerosis; Lou Gehrig's disease; motor neurone disease; prion disease; spinocerebellar ataxia; spinal muscular atrophy; dementia; including Lewy body; vascular and frontotemporal dementia; primary progressive aphasia; mild cognitive impairment; HIV-related cognitive impairment and corticobasal degeneration.
[0086] The compositions of the invention may be particularly useful for treating or preventing chronic disease, treating or preventing disease in patients that have not responded to other therapies (such as treatment with Levodopa, dopamine agonists, MAO-B inhibitors, COMT inhibitors, Glutamate antagonists, and/or anticholinergics), and/or treating or preventing the tissue damage and symptoms associated with dysfunction of the microbiota-gut-brain axis.
[0087] In certain embodiments, the compositions of the invention modulate the CNS. In some embodiments, the compositions of the invention modulate the autonomic nervous system (ANS). In some embodiments, the compositions of the invention modulate the enteric nervous system (ENS). In some embodiments, the compositions of the invention modulate the hypothalamic, pituitary, adrenal (HPA) axis. In some embodiments, the compositions of the invention modulate the neuroendocrine pathway.
[0088] In some embodiments, the compositions of the invention modulate the neuroimmune pathway. In some embodiments, the compositions of the invention modulate the CNS, the ANS, the ENS, the HPA axis and/or the neuroendocrine and neuroimmune pathways. In certain embodiments, the compositions of the invention module the levels of commensal metabolites and/or the gastrointestinal permeability of a subject. In certain embodiments, the compositions of the invention modulate the dopaminergic system.
[0089] The signalling of the microbiota-gut-brain axis is modulated by neural systems. Accordingly, in some embodiments, the compositions of the invention modulate signalling in neural systems. In certain embodiments, the compositions of the invention modulate the signalling of the central nervous system. In some embodiments, the compositions of the invention modulate signalling in sensory neurons. In other embodiments, the compositions of the invention modulate signalling in motor neurons. In some embodiments, the compositions of the invention modulate the signalling in the ANS. In some embodiments, the ANS is the parasympathetic nervous system. In preferred embodiments, the compositions of the invention modulate the signalling of the vagus nerve. In other embodiments, the ANS is the sympathetic nervous system. In other embodiments, the compositions of the invention modulate the signalling in the enteric nervous system. In certain embodiments, the signalling of ANS and ENS neurons responds directly to luminal contents of the gastrointestinal tract. In other embodiments, the signalling of ANS and ENS neurons responds indirectly to neurochemicals produced by luminal bacteria. In other embodiments, the signalling of ANS and ENS neurons responds to neurochemicals produced by luminal bacteria or enteroendocrine cells. In certain preferred embodiments, the neurons of the ENS activate vagal afferents that influence the functions of the CNS. In some embodiments, the compositions of the invention regulate the activity of enterochromaffin cells.
[0090] Neurodegenerative Diseases
[0091] Tauopathies are neurodegenerative diseases associated with the pathological aggregation of tau protein in neurofibrillary or gliofibrillary tangles in the human brain. Alzheimer's disease is an example of a tauopathology. Synucleinopathies (also called .alpha.-Synucleinopathies) are neurodegenerative diseases characterised by the abnormal accumulation of aggregates of .alpha.-synuclein in neurons, nerve fibres or glial cells. Parkinson's disease is an example of a synucleinopathology.
[0092] There is clinical and pathological overlap between these two pathologies. Parkinson's disease patients frequently have dementia and Alzheimer's disease patients often manifest parkinsonism [29]. For example, progressive supranuclear palsy (also known as Steele-Richardson-Olszewski syndrome) has a tauopathology, but also leads to prominent parkinsonism [30]. Mutations in LRRK2 known to cause parkinsonism are associated with the accumulation of synuclein, tau, neither, or both proteins [31].
[0093] Lewy body disease (LBD) is a neurodegenerative disease that is one of the most common causes of dementia in the elderly. LBD exemplifies the existence of a continuum between tau- and synuclein-pathologies. LBD shares clinical and pathological features with Parkinson disease, Parkinson disease dementia and Alzheimer disease [29].
[0094] In particular embodiments, the compositions of the invention may be useful for treating or preventing tauopathies and/or synucleinopathies. In particular embodiments, the compositions of the invention may be useful for treating or preventing tauopathies. In particular embodiments, the compositions of the invention may be useful for treating or preventing synucleinopathies. In certain embodiments, the compositions of the invention may be useful for treating or preventing a disease or condition selected from the group consisting of: Parkinson's disease, including progressive supranuclear palsy, progressive supranuclear palsy, Steele-Richardson-Olszewski syndrome, normal pressure hydrocephalus, vascular or arteriosclerotic parkinsonism and drug-induced parkinsonism; Alzheimer's disease, including Benson's syndrome; and dementia; including Lewy body; vascular and frontotemporal dementia.
[0095] In preferred embodiments, the compositions of the invention may be useful for treating or preventing Parkinson's disease, including progressive supranuclear palsy, progressive supranuclear palsy, Steele-Richardson-Olszewski syndrome, normal pressure hydrocephalus, vascular or arteriosclerotic parkinsonism and drug-induced parkinsonism. In preferred embodiments, the compositions of the invention may be useful for treating or preventing Alzheimer's disease, including Benson's syndrome. In further preferred embodiments, the compositions of the invention may be useful for treating or preventing dementia; including Lewy body; vascular and frontotemporal dementia.
[0096] Parkinson's Disease
[0097] Parkinson's disease is a common neurodegenerative disease neuropathologically characterised by degeneration of heterogeneous populations of neural cells (dopamine-producing cells). The clinical diagnosis of Parkinson's disease requires bradykinesia and at least one of the following core symptoms: resting tremor; muscle rigidity and postural reflex impairment. Other signs and symptoms that may be present or develop during the progression of the disease are autonomic disturbances (sialorrhoea, seborrhoea, constipation, micturition disturbances, sexual functioning, orthostatic hypotension, hyperhydrosis), sleep disturbances and disturbances in the sense of smell or sense of temperature. Parkinson's disease is a neurodegenerative diseases that may develop or persist due to dysfunction of the microbiota-gut-brain axis. Therefore, in preferred embodiments, the compositions of the invention are for use in treating or preventing Parkinson's disease in a subject.
[0098] In further preferred embodiments, the invention provides a composition comprising a bacterial strain of the genus Roseburia, for use in a method of treating or preventing Parkinson's disease. Compositions comprising a bacterial strain of the genus Roseburia may improve motor and cognitive functions in models of Parkinson's disease. Treatment with Roseburia strains may modulate signalling in the central, autonomic and enteric nervous systems; may modulate the activity of the HPA axis pathway; may modulate neuroendocrine and/or neuroimmune pathways; and may modulate the levels of commensal metabolites, inflammatory markers and/or gastrointestinal permeability of a subject, all of which are implicated in the neuropathology of Parkinson's disease.
[0099] In preferred embodiments, the invention provides a composition comprising a bacterial strain of the species Roseburia hominis for use in a method of treating or preventing Parkinson's disease. Compositions using Roseburia hominis may be particularly effective for treating Parkinson's disease. In preferred embodiments, the compositions of the invention prevent, reduce or alleviate one or more of the symptoms of Parkinson's disease in a subject. In preferred embodiments, the compositions of the invention prevent, reduce or alleviate one or more core symptoms of Parkinson's disease in a subject. In certain embodiments, the compositions of the invention prevent, reduce or alleviate bradykinesia in a subject. In certain embodiments, the compositions of the invention prevent, reduce or alleviate resting tremor; muscle rigidity and/or postural reflex impairment in a subject. In certain embodiments, the compositions of the invention prevent, reduce or alleviate one or more symptoms associated with Parkinson's disease progression selected from autonomic disturbances (sialorrhoea, seborrhoea, constipation, micturition disturbances, sexual functioning, orthostatic hypotension, hyperhydrosis), sleep disturbances and disturbances in the sense of smell or sense of temperature.
[0100] In preferred embodiments, the compositions of the invention prevent, reduce or alleviate depressive symptoms comorbid with Parkinson's disease. In certain embodiments, the compositions of the invention improve verbal memory and/or executive functions. In certain embodiments, the compositions of the invention improve attention, working memory, verbal fluency and/or anxiety.
[0101] In other preferred embodiments, the compositions of the invention prevent, reduce or alleviate cognitive dysfunctions comorbid with Parkinson's disease.
[0102] In certain embodiments, the compositions of the invention prevent, reduce or alleviate Parkinson's disease progression. In certain embodiments, the compositions of the invention prevent, reduce or alleviate later motor complications. In certain embodiments, the compositions of the invention prevent, reduce or alleviate late motor fluctuations. In certain embodiments, the compositions of the invention prevent, reduce or alleviate neuronal loss. In certain embodiments, the compositions of the invention improve symptoms of Parkinson's disease dementia (PDD). In certain embodiments, the compositions of the invention prevent, reduce or alleviate impairment of executive function, attention and/or working memory. In certain embodiments, the compositions of the invention improve dopaminergic neurotransmission. In certain embodiments, the compositions of the invention prevent, reduce or alleviate impaired dopaminergic neurotransmission.
[0103] In some embodiments, the compositions of the invention improve the symptoms of Parkinson's disease according to a symptomatic or diagnostic scale. In certain embodiments, the tests for assessing symptomatic improvement of motor function in Parkinson's disease is the Unified Parkinson's Disease Rating Scale. In particular, UPDRS II considers the activity of daily life and UPDRS III considers motor-examination.
[0104] In some embodiments, the compositions of the invention improve the symptoms associated the PDD according to a symptomatic or diagnostic test and/or scale. In certain embodiments, the test or scale is selected from the Hopkins Verbal Learning Test--Revised (HVLT-R); the Delis-Kaplan Executive Function System (D-KEFS) Color-Word Interference Test; the Hamilton Depression Rating Scale (HAM-D 17; depression); the Hamilton Anxiety Rating Scale (HAM-A; anxiety) and the Unified Parkinson's Disease Rating Scale (UPDRS; PD symptom severity).
[0105] In some embodiments, the compositions of the invention improve the Clinical Global Impression--Global Improvement (CGI-I) scale for assessing psychiatric and neurological disorders. In some embodiments, the compositions of the invention display a positive effect on global social and occupational impairment of the subject with Parkinson's disease.
[0106] Alzheimer's Disease and Dementia
[0107] In DSM-5, the term dementia was replaced with the terms major neurocognitive disorder and mild neurocognitive disorder. Neurocognitive disorder is a heterogeneous class of psychiatric diseases. The most common neurocognitive disorder is Alzheimer's disease, followed by vascular dementias or mixed forms of the two. Other forms of neurodegenerative disorders (e.g. Lewy body disease, frontotemporal dementia, Parkinson's dementia, Creutzfeldt-Jakob disease, Huntington's disease, and Wernicke-Korsakoff syndrome) are accompanied by dementia.
[0108] Alzheimer's disease and dementia are also characterised by neuronal loss, so the neuroprotective and neuroproliferative effects shown in the examples for the compositions of the invention indicate that they may be useful for treating or preventing these conditions.
[0109] The symptomatic criteria for dementia under DSM-5 are evidence of significant cognitive decline from a previous level of performance in one or more cognitive domains selected from: learning and memory; language; executive function; complex attention; perceptual-motor and social cognition. The cognitive deficits must interfere with independence in everyday activities. In addition, the cognitive deficits do not occur exclusively in the context of a delirium and are not better explained by another mental disorder (for example VIDD or schizophrenia).
[0110] In addition to the primary symptom, subjects with neurodegenerative disorders display behavioural and psychiatric symptoms including agitation, aggression, depression, anxiety, apathy, psychosis and sleep-wake cycle disturbances.
[0111] Neurodegenerative disorders may develop or persist due to dysfunction of the microbiota-gut-brain axis. Therefore, in preferred embodiments, the compositions of the invention are for use in treating or preventing neurodegenerative disorders in a subject. In preferred embodiments, the neurodegenerative disorder is Alzheimer's disease. In other embodiments, the neurodegenerative disorder is selected from vascular dementias; mixed form Alzheimer's disease and vascular dementia; Lewy body disease; frontotemporal dementia; Parkinson's dementia; Creutzfeldt-Jakob disease; Huntington's disease; and Wernicke-Korsakoff syndrome.
[0112] In preferred embodiments, the compositions of the invention prevent, reduce or alleviate one or more of the symptoms of neurodegenerative disorders in a subject. In certain embodiments, the compositions of the invention prevent, reduce or alleviate the occurrence of cognitive decline in a subject. In certain embodiments, the compositions of the invention improve the level of performance of a subject with neurodegenerative disorders in one or more cognitive domains selected from: learning and memory; language; executive function; complex attention; perceptual-motor and social cognition. In some embodiments, the compositions of the invention prevent, reduce or alleviate the occurrence of one or more behavioural and psychiatric symptoms associated with neurodegenerative disorders selected from agitation, aggression, depression, anxiety, apathy, psychosis and sleep-wake cycle disturbances.
[0113] In certain embodiments, the compositions of the invention prevent, reduce or alleviate symptomatic disease by intervention in suspected pathogenic mechanisms at a preclinical stage. In certain embodiments, the compositions of the invention improve disease modification, with slowing or arrest of symptom progression. In some embodiments, the slowing or arrest of symptom progression correlates with evidence in delaying the underlying neuropathological process. In preferred embodiments, the compositions of the invention improve symptoms of neurodegenerative disorders comprising enhanced cognitive and functional improvement. In preferred embodiments, the compositions of the invention improve the behavioural and psychiatric symptoms of dementia (BPSD). In preferred embodiments, the compositions of the invention improve the ability of a subject with neurodegenerative disorder to undertake everyday activities.
[0114] In preferred embodiments, the compositions of the invention improve both cognition and functioning in a subject with Alzheimer's disease. In some embodiments, the composition of the invention improves the cognitive endpoint in a subject with Alzheimer's disease. In some embodiments, the compositions of the invention improve the functional endpoint in a subject with Alzheimer's disease. In preferred embodiments, the compositions of the invention improve the cognitive and functional endpoint in a subject with Alzheimer's disease. In yet further preferred embodiments, the compositions of the invention improve the overall clinical response (the global endpoint) in a subject with Alzheimer's disease.
[0115] In some embodiments, the compositions of the invention improve the symptoms of neurodegenerative disorders according to a symptomatic or diagnostic test. In certain embodiments, the tests for assessing symptomatic improvement of Alzheimer's disease (and other neurodegenerative disorders) are selected from objective cognitive, activities of daily living, global assessment of change, health related quality of life tests and tests assessing behavioural and psychiatric symptoms of neurodegenerative disorders.
[0116] In certain embodiments, the objective cognitive tests for assessment of symptomatic improvement use the Alzheimer's disease Assessment Scale cognitive subscale (ADAS-cog) and the classic ADAS scale. In certain embodiments, symptomatic improvement of cognition is assessed using the Neurophysiological Test Battery for Use in Alzheimer's Disease (NTB).
[0117] In some embodiments, the global assessment of change test uses the Clinical Global Impression--Global Improvement (CGI-I) scale for assessing psychiatric and neurological disorders. In some embodiments, the global scale is the Clinician's Interview Based Impression of Change plus (CIBIC-plus). In some embodiments, the global scale is the Alzheimer's Disease Cooperative Study Unit Clinician's Global Impression of Change (ADCS-CGIC).
[0118] In certain embodiments, the health related quality of life measures are the Alzheimer's Disease-Related QOL (ADRQL) and the QOL-Alzheimer's Disease (QOL-AD).
[0119] In certain embodiments, the tests assessing behavioural and psychiatric symptoms of neurodegenerative disorders are selected from the Behavioural pathology in Alzheimer's Disease Rating Scale (BEHAVE-AD); the Behavioural Rating Scale for Dementia (BRSD); the Neuropsychiatric Inventory (NPI); and the Cohen-Mansfield Agitation Inventory (CMAI).
[0120] In some embodiments, the compositions of the invention are particularly effective at preventing, reducing or alleviating neurodegenerative disorders when used in combination with another therapy for treating neurodegenerative disorders. In certain embodiments, such therapies include acetylcholinesterase inhibitors including donepezil (Aricept.RTM.), galantamine (Razadyne.RTM.) and rivastigmine (Exelon.RTM.), and memantine.
[0121] Neurochemical Factors, Neuropeptides and Neurotransmitters and the Microbiota-Gut-Brain Axis
[0122] As outlined above, the microbiota-gut-brain axis is modulated by a number of different physiological systems. The microbiota-gut-brain axis is modulated by a number of signalling molecules. Alterations in the levels of these signalling molecules results in neurodegenerative diseases. The experiments performed by the inventors indicate that administration of Roseburia species, and in particular Roseburia hominis, can modulate levels of indole and kynurenine. Dysregulation of these metabolites can lead to neurodegenerative diseases, such as Parkinson's disease.
[0123] In certain embodiments, the compositions of the invention modulate the levels of brain monoamines and metabolites thereof. In preferred embodiments the metabolite is kynurenine. In certain embodiments, the compositions of the invention modulate kynurenine, which is the main route of tryptophan metabolism, which serves as a route to nicotinamide adenine dinucleotide (NAD+) production. Kynurenine can be metabolized to neuroactive compounds such as kynurenic acid (KYNA) and 3-hydroxy-1-kynurenine (3-OH-1-KYN), and in further steps to quinolinic acid (QUIN). Dysregulation of the kynurenine pathway can lead to activation of the immune system and the accumulation of potentially neurotoxic compounds. Alterations in the kynurenine metabolism may be involved in the development of Parkinson's diseases. Kynurenine levels have been demonstrated to be decreased in the frontal cortex, putamen and substantia nigra pars compacta of patients with PD [32]. Therefore, in certain embodiments the compositions of the invention are for use in increasing the levels of kynurenine in the treatment of Parkinson's disease.
[0124] In certain embodiments of the invention the compositions of the invention can increase the levels of kynurenin. Increased levels of kynurenine have been shown to attenuated IPP+-induced neuronal cell death in vitro in a human dopaminergic neuroblastoma cell line [33]. In certain embodiments kynurenine and kynurenic acid, can activate GI aryl hydrocarbon receptor (Ahr) and GPR35 receptors. Activation of Ahr receptor induces IL-22 production, which can inhibit local inflammation. Activation of GPR35 inducing the production of inositol triphosphate and Ca2+ mobilization.
[0125] In certain embodiments, the compositions of the invention modulate the levels of indole. In preferred embodiments the metabolite is kynurenine. In certain embodiments, the compositions of the invention modulate kynurenine, which is the main route of tryptophan metabolism.
[0126] The signalling of the microbiota-gut-brain axis is modulated by levels of neurochemical factors, neuropeptides and neurotransmitters. Accordingly, in certain embodiments, the compositions of the invention modulates levels of neurochemical factors, neuropeptides and neurotransmitters. Accordingly, in certain preferred embodiments, the compositions of the invention directly alter CNS biochemistry.
[0127] The signalling of the microbiota-gut-brain axis is modulated by levels of .gamma.-aminobutyric acid (GABA). Accordingly, in preferred embodiments, the compositions of the invention modulate the levels of GABA. GABA is an inhibitory neurotransmitter that reduces neuronal excitability. In certain embodiments, the compositions of the invention increase the levels of GABA. In certain embodiments, the compositions of the invention decrease the levels of GABA. In certain embodiments, the compositions of the invention alter GABAergic neurotransmission. In certain embodiments, the compositions of the invention modulate the level of GABA transcription in different regions of the central nervous system. In certain embodiments, the commensal derived GABA crosses the blood-brain barrier and affects neurotransmission directly. In certain embodiments, the compositions of the invention lead to a reduction of GABA in the hippocampus, amygdala and/or locus coeruleus. In certain embodiments, the compositions of the invention lead to an increase of GABA in cortical regions.
[0128] Immune Response
[0129] The signalling of the microbiota-gut-brain axis is modulated by alterations in the immune response and inflammatory factors and markers. Accordingly, in certain embodiments, the compositions of the invention may modulate the immune response. In certain embodiments, the compositions of the invention modulate the systemic levels of circulating neuroimmune signalling molecules. In certain preferred embodiments, the compositions of the invention modulate pro-inflammatory cytokine production and inflammation. In certain embodiments, the compositions of the invention modulate the inflammatory state. In certain embodiments, the compositions of the invention decrease IL-6 production and secretion. In certain embodiments, the compositions of the invention decrease the activation of the NF.kappa.B promoter. In certain embodiments, the compositions of the invention are able to modulate the activation of IL-6 production by the potent pro-inflammatory endotoxin lipopolysaccharide (LPS). In certain embodiments, the compositions of the invention are able to modulate the activation of the NF.kappa.B promoter by LPS and .alpha.-synuclein mutant proteins such as A53T. Increased circulating levels of cytokines are closely associated with various neurodegenerative disorders, including Parkinson's, dementia and Alzheimer's. In certain embodiments, the compositions of the invention are for use in reducing IL-6 levels and/or NF.kappa.B levels in the treatment of a neurodegenerative disorder.
[0130] The signalling of the microbiota-gut-brain axis is modulated by levels of commensal metabolites. Accordingly, in certain embodiments, the compositions of the invention modulate the systemic levels of microbiota metabolites. In certain preferred embodiments, the compositions of the invention modulate the level of short chain fatty acids (SCFAs). In certain embodiments the level of SCFAs is increased or decreased. In some embodiments, the SCFA is butyric acid (BA) (or butyrate). In some embodiments, the SCFA is propionic acid (PPA). In some embodiments, the SCFA is acetic acid. In certain embodiments, the compositions of the invention modulate the ability of SCFAs to cross the blood-brain barrier.
[0131] Histone acetylation and deacetylation are important epigenetic regulators of gene expression. An imbalance in histone acetylation and deacetylation can result in apoptosis. Dysregulation of such histone acetyltransferases has been implicated in the pathogenesis associated with age-associated neurodegenerative diseases, such as Parkinson's disease, Huntington's disease, Alzheimer's disease, amyotrophic lateral sclerosis and cognitive decline [34]. Accordingly, in certain embodiments, the compositions of the invention can modulate histone deacetylatase activity. In certain embodiments, the compositions of the invention can reduce histone deacetylatase activity. In certain embodiments, the compositions of the invention can reduce histone acetylatase activity.
[0132] Patients with neurodegenerative diseases, including Parkinson's disease, Huntington's disease, Alzheimer's disease and amyotrophic lateral sclerosis, exhibit high levels of lipid peroxidation. Lipid are vulnerable to oxidation by reactive oxygen species, and the brain is rich in polyunsaturated fatty acids. Accordingly, in certain embodiments, the compositions of the invention can modulate lipid peroxidation. In certain embodiments, the compositions of the invention can reduce lipid peroxidation. Reducing the oxidative damage caused by reactive oxygen species can be used to target early the stages neurodegenerative diseases. Accordingly, in certain embodiments, the compositions of the invention are for use in treating early stage neurodegeneration. Also accordingly, in certain embodiments, the compositions of the invention are for use in preventing the development of a neurodegenerative disorder. In such embodiments, the compositions of the invention may be for use in a patient that has been identified as at risk of developing a neurodegenerative disorder.
[0133] The signalling of the microbiota-gut-brain axis is modulated by levels of gastrointestinal permeability. Accordingly, in some embodiments, the compositions of the invention alter the integrity of the gastrointestinal tract epithelium. In certain embodiments, the compositions of the invention modulate the permeability of the gastrointestinal tract. In certain embodiments, the compositions of the invention modulate the barrier function and integrity of the gastrointestinal tract. In certain embodiments, the compositions of the invention modulate gastrointestinal tract motility. In certain embodiments, the compositions of the invention modulate the translocation of commensal metabolites and inflammatory signalling molecules into the bloodstream from the gastrointestinal tract lumen.
[0134] The signalling of the microbiota-gut-brain axis is modulated by microbiome composition in the gastrointestinal tract. Accordingly, in certain embodiments, the compositions of the invention modulates the microbiome composition of the gastrointestinal tract. In certain embodiments, the compositions of the invention prevents microbiome dysbiosis and associated increases in toxic metabolites (e.g. LPS). In certain embodiments, the compositions of the invention modulate the levels of Clostridium in the gastrointestinal tract. In preferred embodiments, the compositions of the invention reduce the level of Clostridium in the gastrointestinal tract. In certain embodiments, the compositions of the invention reduce the levels of Campylobacter jejuni. In certain embodiments, the compositions of the invention modulate the proliferation of harmful anaerobic bacteria and the production of neurotoxins produced by these bacteria. In certain embodiments, the compositions of the invention modulate the microbiome levels of Lactobacillus and/or Bifidobacterium. In certain embodiments, the compositions of the invention modulate the microbiome levels of Sutterella, Prevotella, Ruminococcus genera and/or the Alcaligenaceae family. In certain embodiments, the compositions of the invention increase the level of Lactobacillus plantarum and/or Saccharomyces boulardii.
[0135] Brain Injury
[0136] The examples demonstrate that the compositions of the invention are neuroprotective and have HDAC inhibitory activity. HDAC2 is a crucial target for functional recovery from stroke [35] and HDAC inhibition can prevent white matter injury [36], so the compositions of the invention may be useful in the treatment of brain injury.
[0137] In certain embodiments, the compositions of the invention are for use in treating brain injury. In some embodiments, the brain injury is a traumatic brain injury. In some embodiments, the brain injury is an acquired brain injury. In some embodiments, the compositions of the invention are for use in treating brain injury resulting from trauma. In some embodiments, the compositions of the invention are for use in treating brain injury resulting from a tumour. In some embodiments, the compositions of the invention are for use in treating brain injury resulting from a stroke. In some embodiments, the compositions of the invention are for use in treating brain injury resulting from a brain haemorrhage. In some embodiments, the compositions of the invention are for use in treating brain injury resulting from encephalitis. In some embodiments, the compositions of the invention are for use in treating brain injury resulting from cerebral hypoxia. In some embodiments, the compositions of the invention are for use in treating brain injury resulting from cerebral anoxia.
[0138] In preferred embodiments, the compositions of the invention are for use in treating stroke. The effects shown in the examples are particularly relevant to the treatment of stroke. Stroke occurs when blood flow to at least a part of the brain is interrupted. Without an adequate supply of blood to provide oxygen and nutrients to the brain tissue and to remove waste products from the brain tissue, brain cells rapidly begin to die. The symptoms of stroke are dependent on the region of the brain which is affected by the inadequate blood flow. Symptoms include paralysis, numbness or weakness of the muscles, loss of balance, dizziness, sudden severe headaches, speech impairment, loss of memory, loss of reasoning ability, sudden confusion, vision impairment, coma or even death. A stroke is also referred to as a brain attack or a cerebrovascular accident (CVA). The symptoms of stroke may be brief if adequate blood flow is restored within a short period of time. However, if inadequate blood flow continues for a significant period of time, the symptoms can be permanent.
[0139] In some embodiments, the stroke is cerebral ischemia. Cerebral ischemia results when there is insufficient blood flow to the tissues of the brain to meet metabolic demand. In some embodiments, the cerebral ischemia is focal cerebral ischemia, i.e. confined to a specific region of the brain. In some embodiments the cerebral ischemia is global cerebral ischemia, i.e. encompassing a wide area of the brain tissue. Focal cerebral ischemia commonly occurs when a cerebral vessel has become blocked, either partially or completely, reducing the flow of blood to a specific region of the brain. In some embodiments the focal cerebral ischemia is ischemic stroke. In some embodiments, the ischemic stroke is thrombotic, i.e. caused by a thrombus or blood clot, which develops in a cerebral vessel and restricts or blocks blood flow. In some embodiments the ischemic stroke is a thrombotic stroke. In some embodiments, the ischemic stroke is embolic, i.e. caused by an embolus, or an unattached mass that travels through the bloodstream and restricts or blocks blood flow at a site distant from its point of origin. In some embodiments the ischemic stroke is an embolic stroke. Global cerebral ischemia commonly occurs when blood flow to the brain as a whole is blocked or reduced. In some embodiments the global cerebral ischemia is caused by hypoperfusion, i.e. due to shock. In some embodiments the global cerebral ischemia is a result of a cardiac arrest.
[0140] In some embodiments the subject diagnosed with brain injury has suffered cerebral ischemia. In some embodiments, the subject diagnosed with brain injury has suffered focal cerebral ischemia. In some embodiments, the subject diagnosed with brain injury has suffered an ischemic stroke. In some embodiments, the subject diagnosed with brain injury has suffered a thrombotic stroke. In some embodiments, the subject diagnosed with brain injury has suffered an embolic stroke. In some embodiments, the subject diagnosed with brain injury has suffered global cerebral ischemia. In some embodiments, the subject diagnosed with brain injury has suffered hypoperfusion. In some embodiments, the subject diagnosed with brain injury has suffered a cardiac arrest.
[0141] In some embodiments, the compositions of the invention are for use in treating cerebral ischemia. In some embodiments, the compositions of the invention are for use in treating focal cerebral ischemia. In some embodiments, the compositions of the invention are for use treating ischemic stroke. In some embodiments, the compositions of the invention are for use in treating thrombotic stroke. In some embodiments, the compositions of the invention are for use in treating embolic stroke. In some embodiments, the compositions of the invention are for use in treating global cerebral ischemia. In some embodiments, the compositions of the invention are for use in treating hypoperfusion.
[0142] In some embodiments, the stroke is hemorrhagic stroke. Hemorrhagic stroke is caused by bleeding into or around the brain resulting in swelling, pressure and damage to the cells and tissues of the brain. Hemorrhagic stroke is commonly a result of a weakened blood vessel that ruptures and bleeds into the surrounding brain. In some embodiments, the hemorrhagic stroke is an intracerebral hemorrhage, i.e. caused by bleeding within the brain tissue itself. In some embodiments the intracerebral hemorrhage is caused by an intraparenchymal hemorrhage. In some embodiments the intracerebral hemorrhage is caused by an intraventricular hemorrhage. In some embodiments the hemorrhagic stroke is a subarachnoid hemorrhage i.e. bleeding that occurs outside of the brain tissue but still within the skull. In some embodiments, the hemorrhagic stroke is a result of cerebral amyloid angiopathy. In some embodiments, the hemorrhagic stroke is a result of a brain aneurysm. In some embodiments, the hemorrhagic stroke is a result of cerebral arteriovenous malformation (AVM).
[0143] In some embodiments the subject diagnosed with brain injury has suffered hemorrhagic stroke. In some embodiments, the subject diagnosed with brain injury has suffered an intracerebral hemorrhage. In some embodiments, the subject diagnosed with brain injury has suffered an intraparenchymal hemorrhage. In some embodiments, the subject diagnosed with brain injury has suffered an intraventricular hemorrhage. In some embodiments, the subject diagnosed with brain injury has suffered a subarachnoid hemorrhage. In some embodiments, the subject diagnosed with brain injury has suffered cerebral amyloid angiopathy. In some embodiments, the subject diagnosed with brain injury has suffered a brain aneurysm. In some embodiments, the subject diagnosed with brain injury has suffered cerebral AVM.
[0144] In some embodiments, the compositions of the invention are for use in treating hemorrhagic stroke. In some embodiments, the compositions of the invention are for use in treating an intracerebral hemorrhage. In some embodiments, the compositions of the invention are for use in treating an intraparenchymal hemorrhage. In some embodiments, the compositions of the invention are for use in treating an intraventricular hemorrhage. In some embodiments, the compositions of the invention are for use in treating a subarachnoid hemorrhage. In some embodiments, the compositions of the invention are for use in treating a cerebral amyloid angiopathy. In some embodiments, the compositions of the invention are for use in treating a brain aneurysm. In some embodiments, the compositions of the invention are for use in treating cerebral AVM.
[0145] Restoration of adequate blood flow to the brain after a period of interruption, though effective in alleviating the symptoms associated with stroke, can paradoxically result in further damage to the brain tissue. During the period of interruption, the affected tissue suffers from a lack of oxygen and nutrients, and the sudden restoration of blood flow can result in inflammation and oxidative damage through the induction of oxidative stress. This is known as reperfusion injury, and is well documented not only following stroke, but also following a heart attack or other tissue damage when blood supply returns to the tissue after a period of ischemia or lack of oxygen. In some embodiments the subject diagnosed with brain injury has suffered from reperfusion injury as a result of stroke. In some embodiments, the compositions of the invention are for use in treating reperfusion injury as a result of stroke.
[0146] A transient ischemic attack (TIA), often referred to as a mini-stroke, is a recognised warning sign for a more serious stroke. Subjects who have suffered one or more TIAs are therefore at greater risk of stroke. In some embodiments the subject diagnosed with brain injury has suffered a TIA. In some embodiments, the compositions of the invention are for use in treating a TIA. In some embodiments, the compositions of the invention are for use in treating brain injury in a subject who has suffered a TIA.
[0147] High blood pressure, high blood cholesterol, a familial history of stroke, heart disease, diabetes, brain aneurysms, arteriovenous malformations, sickle cell disease, vasculitis, bleeding disorders, use of nonsteroidal anti-inflammatory drugs (NSAIDs), smoking tobacco, drinking large amounts of alcohol, illegal drug use, obesity, lack of physical activity and an unhealthy diet are all considered to be risk factors for stroke. In particular, lowering blood pressure has been conclusively shown to prevent both ischemic and hemorrhagic strokes [37, 38]. In some embodiments, the compositions of the invention are for use in treating brain injury in a subject who has at least one risk factor for stroke. In some embodiments the subject has two risk factors for stroke. In some embodiments the subject has three risk factors for stroke. In some embodiments the subject has four risk factors for stroke. In some embodiments the subject has more than four risk factors for stroke. In some embodiments the subject has high blood pressure. In some embodiments the subject has high blood cholesterol. In some embodiments the subject has a familial history of stroke. In some embodiments the subject has heart disease. In some embodiments the subject has diabetes. In some embodiments the subject has a brain aneurysm. In some embodiments the subject has arteriovenous malformations. In some embodiments the subject has vasculitis. In some embodiments the subject has sickle cell disease. In some embodiments the subject has a bleeding disorder. In some embodiments the subject has a history of use of nonsteroidal anti-inflammatory drugs (NSAIDs). In some embodiments the subject smokes tobacco. In some embodiments the subject drinks large amounts of alcohol. In some embodiments the subject uses illegal drugs. In some embodiments the subject is obese. In some embodiments the subject is overweight. In some embodiments the subject has a lack of physical activity. In some embodiments the subject has an unhealthy diet.
[0148] The examples indicate that the compositions of the invention may be useful for treating brain injury and aiding recovery when administered before the injury event occurs. Therefore, the compositions of the invention may be particularly useful for treating brain injury when administered to subjects at risk of brain injury, such as stroke.
[0149] In certain embodiments, the compositions of the invention are for use in reducing the damage caused by a potential brain injury, preferably a stroke. The compositions may reduce the damage caused when they are administered before the potential brain injury occurs, in particular when administered to a patient identified as at risk of a brain injury.
[0150] The examples indicate that the compositions of the invention may be useful for treating brain injury and aiding recovery when administered after the injury event occurs. Therefore, the compositions of the invention may be particularly useful for treating brain injury when administered to subjects following a brain injury, such as stroke.
[0151] In some embodiments, the compositions of the invention treat brain injury by reducing motoric damage. In some embodiments, the compositions of the invention treat brain injury by improving motor function. In some embodiments, the compositions of the invention treat brain injury by improving muscle strength. In some embodiments, the compositions of the invention treat brain injury by improving memory. In some embodiments, the compositions of the invention treat brain injury by improving social recognition. In some embodiments, the compositions of the invention treat brain injury by improving neurological function.
[0152] Treatment of brain injury may refer to, for example, an alleviation of the severity of symptoms. Treatment of brain injury may also refer to reducing the neurological impairments following stroke. Compositions of the invention for use in treating stroke may be provided to the subject in advance of the onset of stroke, for example in a patient identified as being at risk of stroke. Compositions of the invention for use in treating stroke may be provided after a stroke has occurred, for example, during recovery. Compositions of the invention for use in treating stroke may be provided during the acute phase of recovery (i.e. up to one week after stroke). Compositions of the invention for use in treating stroke may be provided during the subacute phase of recovery (i.e. from one week up to three months after stroke). Compositions of the invention for use in treating stroke may be provided during the chronic phase of recovery (from three months after stroke).
[0153] In certain embodiments, the compositions of the invention are for use in combination with a secondary active agent. In certain embodiments, the compositions of the invention are for use in combination with aspirin or tissue plasminogen activator (tPA). Other secondary agents include other antiplatelets (such as clopidogrel), anticoagulants (such as heparins, warfarin, apixaban, dabigatran, edoxaban or rivaroxaban), antihypertensives (such as diuretics, ACE inhibitors, calcium channel blockers, beta-blockers or alpha-blockers) or statins. The compositions of the invention may improve the patient's response to the secondary active agent.
[0154] In certain embodiments, the compositions of the invention reduce the effect of ischemia on tissues. In certain embodiments, the compositions of the invention reduce the amount of damage to tissues caused by ischemia. In certain embodiments, the tissues damaged by ischemia are the cerebral tissues. In certain embodiments, the compositions of the invention reduce necrosis or the number of necrotic cells. In certain embodiments, the compositions of the invention reduce apoptosis or the number of apoptotic cells. In certain embodiments, the compositions of the invention reduce the number of necrotic and apoptotic cells. In certain embodiments, the compositions of the invention prevent cell death by necrosis and/or apoptosis. In certain embodiments, the compositions of the invention prevent cell death by necrosis and/or apoptosis caused by ischemia. In certain embodiments, the compositions of the invention improve the recovery of the tissue damaged by ischemia. In certain embodiments, the compositions of the invention improve the speed of clearance of necrotic cells and/or apoptotic cells. In certain embodiments, the compositions of the invention improve the efficacy of the clearance of necrotic cells and/or apoptotic cells. In certain embodiments, the compositions of the invention improve the replacement and/or regeneration of cells within tissues. In certain embodiments, the compositions of the invention improve the replacement and/or regeneration of cells within tissues damaged by ischemia. In certain embodiments, the compositions of the invention improve the overall histology of the tissue (for example upon a biopsy).
[0155] Modes of Administration
[0156] Preferably, the compositions of the invention are to be administered to the gastrointestinal tract in order to enable delivery to and/or partial or total colonisation of the intestine with the bacterial strain of the invention. Generally, the compositions of the invention are administered orally, but they may be administered rectally, intranasally, or via buccal or sublingual routes.
[0157] In certain embodiments, the compositions of the invention may be administered as a foam, as a spray or a gel.
[0158] In certain embodiments, the compositions of the invention may be administered as a suppository, such as a rectal suppository, for example in the form of a theobroma oil (cocoa butter), synthetic hard fat (e.g. suppocire, witepsol), glycero-gelatin, polyethylene glycol, or soap glycerin composition.
[0159] In certain embodiments, the composition of the invention is administered to the gastrointestinal tract via a tube, such as a nasogastric tube, orogastric tube, gastric tube, jejunostomy tube (J tube), percutaneous endoscopic gastrostomy (PEG), or a port, such as a chest wall port that provides access to the stomach, jejunum and other suitable access ports.
[0160] The compositions of the invention may be administered once, or they may be administered sequentially as part of a treatment regimen. In certain embodiments, the compositions of the invention are to be administered daily.
[0161] In certain embodiments of the invention, treatment according to the invention is accompanied by assessment of the patient's gut microbiota. Treatment may be repeated if delivery of and/or partial or total colonisation with the strain of the invention is not achieved such that efficacy is not observed, or treatment may be ceased if delivery and/or partial or total colonisation is successful and efficacy is observed.
[0162] In certain embodiments, the composition of the invention may be administered to a pregnant animal, for example a mammal such as a human in order to prevent an inflammatory or autoimmune disease developing in her child in utero and/or after it is born.
[0163] The compositions of the invention may be administered to a patient that has been diagnosed with a neurodegenerative disease, or that has been identified as being at risk of a neurodegenerative disease. The compositions may also be administered as a prophylactic measure to prevent the development of neurodegenerative disease in a healthy patient.
[0164] The compositions of the invention may be administered to a patient that has been identified as having an abnormal gut microbiota. For example, the patient may have reduced or absent colonisation by Roseburia, and in particular Roseburia hominis.
[0165] The compositions of the invention may be administered as a food product, such as a nutritional supplement.
[0166] Generally, the compositions of the invention are for the treatment of humans, although they may be used to treat animals including monogastric mammals such as poultry, pigs, cats, dogs, horses or rabbits. The compositions of the invention may be useful for enhancing the growth and performance of animals. If administered to animals, oral gavage may be used.
[0167] Compositions
[0168] Generally, the composition of the invention comprises bacteria. In preferred embodiments of the invention, the composition is formulated in freeze-dried form. For example, the composition of the invention may comprise granules or gelatin capsules, for example hard gelatin capsules, comprising a bacterial strain of the invention.
[0169] Preferably, the composition of the invention comprises lyophilised bacteria. Lyophilisation of bacteria is a well-established procedure and relevant guidance is available in, for example, references [39-41].
[0170] Alternatively, the composition of the invention may comprise a live, active bacterial culture.
[0171] In some embodiments, the bacterial strain in the composition of the invention has not been inactivated, for example, has not been heat-inactivated. In some embodiments, the bacterial strain in the composition of the invention has not been killed, for example, has not been heat-killed. In some embodiments, the bacterial strain in the composition of the invention has not been attenuated, for example, has not been heat-attenuated. For example, in some embodiments, the bacterial strain in the composition of the invention has not been killed, inactivated and/or attenuated. For example, in some embodiments, the bacterial strain in the composition of the invention is live. For example, in some embodiments, the bacterial strain in the composition of the invention is viable. For example, in some embodiments, the bacterial strain in the composition of the invention is capable of partially or totally colonising the intestine. For example, in some embodiments, the bacterial strain in the composition of the invention is viable and capable of partially or totally colonising the intestine.
[0172] In some embodiments, the composition comprises a mixture of live bacterial strains and bacterial strains that have been killed.
[0173] In preferred embodiments, the composition of the invention is encapsulated to enable delivery of the bacterial strain to the intestine. Encapsulation protects the composition from degradation until delivery at the target location through, for example, rupturing with chemical or physical stimuli such as pressure, enzymatic activity, or physical disintegration, which may be triggered by changes in pH. Any appropriate encapsulation method may be used. Exemplary encapsulation techniques include entrapment within a porous matrix, attachment or adsorption on solid carrier surfaces, self-aggregation by flocculation or with cross-linking agents, and mechanical containment behind a microporous membrane or a microcapsule. Guidance on encapsulation that may be useful for preparing compositions of the invention is available in, for example, references [42] and [43].
[0174] The composition may be administered orally and may be in the form of a tablet, capsule or powder. Encapsulated products are preferred because Roseburia are anaerobes. Other ingredients (such as vitamin C, for example), may be included as oxygen scavengers and prebiotic substrates to improve the delivery and/or partial or total colonisation and survival in vivo. Alternatively, the probiotic composition of the invention may be administered orally as a food or nutritional product, such as milk or whey based fermented dairy product, or as a pharmaceutical product.
[0175] The composition may be formulated as a probiotic.
[0176] A composition of the invention includes a therapeutically effective amount of a bacterial strain of the invention. A therapeutically effective amount of a bacterial strain is sufficient to exert a beneficial effect upon a patient. A therapeutically effective amount of a bacterial strain may be sufficient to result in delivery to and/or partial or total colonisation of the patient's intestine.
[0177] A suitable daily dose of the bacteria, for example for an adult human, may be from about 1.times.10.sup.3 to about 1.times.10.sup.11 colony forming units (CFU); for example, from about 1.times.10.sup.7 to about 1.times.10.sup.10 CFU; in another example from about 1.times.10.sup.6 to about 1.times.10.sup.10 CFU.
[0178] In certain embodiments, the composition contains the bacterial strain in an amount of from about 1.times.10.sup.6 to about 1.times.10.sup.11 CFU/g, respect to the weight of the composition; for example, from about 1.times.10.sup.8 to about 1.times.10.sup.10 CFU/g. The dose may be, for example, 1 g, 3 g, 5 g, and 10 g.
[0179] Typically, a probiotic, such as the composition of the invention, is optionally combined with at least one suitable prebiotic compound. A prebiotic compound is usually a non-digestible carbohydrate such as an oligo- or polysaccharide, or a sugar alcohol, which is not degraded or absorbed in the upper digestive tract. Known prebiotics include commercial products such as inulin and transgalacto-oligosaccharides.
[0180] In certain embodiments, the probiotic composition of the present invention includes a prebiotic compound in an amount of from about 1 to about 30% by weight, respect to the total weight composition, (e.g. from 5 to 20% by weight). Carbohydrates may be selected from the group consisting of: fructo-oligosaccharides (or FOS), short-chain fructo-oligosaccharides, inulin, isomalt-oligosaccharides, pectins, xylo-oligosaccharides (or XOS), chitosan-oligosaccharides (or COS), beta-glucans, arable gum modified and resistant starches, polydextrose, D-tagatose, acacia fibers, carob, oats, and citrus fibers. In one aspect, the prebiotics are the short-chain fructo-oligosaccharides (for simplicity shown herein below as FOSs-c.c); said FOSs-c.c. are not digestible carbohydrates, generally obtained by the conversion of the beet sugar and including a saccharose molecule to which three glucose molecules are bonded.
[0181] In certain embodiments, the compositions of the invention are used in combination with another therapeutic compound for treating or preventing the neurodegenerative disorder. In some embodiments, the compositions of the invention are administered with nutritional supplements that modulate neuroprotection or neuroproliferation. In preferred embodiments, the nutritional supplements comprise or consist of nutritional vitamins. In certain embodiments, the vitamins are vitamin B6, magnesium, dimethylglycine (vitamin B16) and vitamin C. In certain embodiments, the compositions of the invention are administered in combination with another probiotic.
[0182] In certain embodiments, the compositions of the invention are for use in enhancing the effect of a second agent on a neurodegenerative disease. The immune modulatory effects of the compositions of the invention may make the brain more susceptible to conventional therapies such as Levodopa, dopamine agonists, MAO-B inhibitors, COMT inhibitors, Glutamate antagonists, or anticholinergics, which are exemplary secondary agents to be administered in combination (sequentially or contemporaneously) with the compositions of the invention.
[0183] The compositions of the invention may comprise pharmaceutically acceptable excipients or carriers. Examples of such suitable excipients may be found in the reference [44]. Acceptable carriers or diluents for therapeutic use are well known in the pharmaceutical art and are described, for example, in reference [45]. Examples of suitable carriers include lactose, starch, glucose, methyl cellulose, magnesium stearate, mannitol, sorbitol and the like. Examples of suitable diluents include ethanol, glycerol and water. The choice of pharmaceutical carrier, excipient or diluent can be selected with regard to the intended route of administration and standard pharmaceutical practice. The pharmaceutical compositions may comprise as, or in addition to, the carrier, excipient or diluent any suitable binder(s), lubricant(s), suspending agent(s), coating agent(s), solubilising agent(s). Examples of suitable binders include starch, gelatin, natural sugars such as glucose, anhydrous lactose, free-flow lactose, beta-lactose, corn sweeteners, natural and synthetic gums, such as acacia, tragacanth or sodium alginate, carboxymethyl cellulose and polyethylene glycol. Examples of suitable lubricants include sodium oleate, sodium stearate, magnesium stearate, sodium benzoate, sodium acetate, sodium chloride and the like. Preservatives, stabilizers, dyes and even flavouring agents may be provided in the pharmaceutical composition. Examples of preservatives include sodium benzoate, sorbic acid and esters of p-hydroxybenzoic acid. Antioxidants and suspending agents may be also used.
[0184] The compositions of the invention may be formulated as a food product. For example, a food product may provide nutritional benefit in addition to the therapeutic effect of the invention, such as in a nutritional supplement. Similarly, a food product may be formulated to enhance the taste of the composition of the invention or to make the composition more attractive to consume by being more similar to a common food item, rather than to a pharmaceutical composition. In certain embodiments, the composition of the invention is formulated as a milk-based product. The term "milk-based product" means any liquid or semi-solid milk- or whey-based product having a varying fat content. The milk-based product can be, e.g., cow's milk, goat's milk, sheep's milk, skimmed milk, whole milk, milk recombined from powdered milk and whey without any processing, or a processed product, such as yoghurt, curdled milk, curd, sour milk, sour whole milk, butter milk and other sour milk products. Another important group includes milk beverages, such as whey beverages, fermented milks, condensed milks, infant or baby milks; flavoured milks, ice cream; milk-containing food such as sweets.
[0185] In some embodiments, the compositions of the invention comprise one or more bacterial strains of the genus Roseburia and do not contain bacteria from any other genera, or which comprise only de minimis or biologically irrelevant amounts of bacteria from another genera. Thus, in some embodiments, the invention provides a composition comprising one or more bacterial strains of the genus Roseburia, which does not contain bacteria from any other genera or which comprises only de minimis or biologically irrelevant amounts of bacteria from another genera, for use in therapy. In some embodiments, the compositions of the invention comprise one or more bacterial strains of the species Roseburia hominis and do not contain bacteria from any other species, or which comprise only de minimis or biologically irrelevant amounts of bacteria from another species. Thus, in some embodiments, the invention provides a composition comprising one or more bacterial strains of the species Roseburia hominis, which does not contain bacteria from any other species or which comprises only de minimis or biologically irrelevant amounts of bacteria from another species, for use in therapy.
[0186] In some embodiments, the compositions of the invention comprise one or more bacterial strains of the species Roseburia intestinalis and do not contain bacteria from any other species, or which comprise only de minimis or biologically irrelevant amounts of bacteria from another species. Thus, in some embodiments, the invention provides a composition comprising one or more bacterial strains of the species Roseburia intestinalis, which does not contain bacteria from any other species or which comprises only de minimis or biologically irrelevant amounts of bacteria from another species, for use in therapy.
[0187] In some embodiments, the compositions of the invention comprise one or more bacterial strains of the species Roseburia faecis and do not contain bacteria from any other species, or which comprise only de minimis or biologically irrelevant amounts of bacteria from another species. Thus, in some embodiments, the invention provides a composition comprising one or more bacterial strains of the species Roseburia faecis, which does not contain bacteria from any other species or which comprises only de minimis or biologically irrelevant amounts of bacteria from another species, for use in therapy.
[0188] In some embodiments, the compositions of the invention comprise one or more bacterial strains of the species Roseburia hominis and do not contain bacteria from any other Roseburia species, or which comprise only de minimis or biologically irrelevant amounts of bacteria from another Roseburia species. Thus, in some embodiments, the invention provides a composition comprising one or more bacterial strains of the species Roseburia hominis, which does not contain bacteria from any other Roseburia species or which comprises only de minimis or biologically irrelevant amounts of bacteria from another Roseburia species, for use in therapy.
[0189] In some embodiments, the compositions of the invention comprise one or more bacterial strains of the species Roseburia intestinalis and do not contain bacteria from any other Roseburia species, or which comprise only de minimis or biologically irrelevant amounts of bacteria from another Roseburia species. Thus, in some embodiments, the invention provides a composition comprising one or more bacterial strains of the species Roseburia intestinalis, which does not contain bacteria from any other Roseburia species or which comprises only de minimis or biologically irrelevant amounts of bacteria from another Roseburia species, for use in therapy.
[0190] In some embodiments, the compositions of the invention comprise one or more bacterial strains of the species Roseburia faecis and do not contain bacteria from any other Roseburia species, or which comprise only de minimis or biologically irrelevant amounts of bacteria from another Roseburia species. Thus, in some embodiments, the invention provides a composition comprising one or more bacterial strains of the species Roseburia faecis, which does not contain bacteria from any other Roseburia species or which comprises only de minimis or biologically irrelevant amounts of bacteria from another Roseburia species, for use in therapy.
[0191] In certain embodiments, the compositions of the invention contain a single bacterial strain or species and do not contain any other bacterial strains or species. Such compositions may comprise only de minimis or biologically irrelevant amounts of other bacterial strains or species. Such compositions may be a culture that is substantially free from other species of organism.
[0192] In some embodiments, the invention provides a composition comprising a single bacterial strain of the genus Roseburia, which does not contain bacteria from any other strains or which comprises only de minimis or biologically irrelevant amounts of bacteria from another strain for use in therapy.
[0193] In some embodiments, the invention provides a composition comprising a single bacterial strain of the species Roseburia hominis, which does not contain bacteria from any other strains or which comprises only de minimis or biologically irrelevant amounts of bacteria from another strain for use in therapy.
[0194] In some embodiments, the invention provides a composition comprising a single bacterial strain of the species Roseburia intestinalis, which does not contain bacteria from any other strains or which comprises only de minimis or biologically irrelevant amounts of bacteria from another strain for use in therapy.
[0195] In some embodiments, the invention provides a composition comprising a single bacterial strain of the species Roseburia faecis, which does not contain bacteria from any other strains or which comprises only de minimis or biologically irrelevant amounts of bacteria from another strain for use in therapy.
[0196] In some embodiments, the compositions of the invention comprise more than one bacterial strain. For example, in some embodiments, the compositions of the invention comprise more than one strain from within the same species (e.g. more than 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 35, 40 or 45 strains), and, optionally, do not contain bacteria from any other species. In some embodiments, the compositions of the invention comprise less than 50 strains from within the same species (e.g. less than 45, 40, 35, 30, 25, 20, 15, 12, 10, 9, 8, 7, 6, 5, 4 or 3 strains), and, optionally, do not contain bacteria from any other species. In some embodiments, the compositions of the invention comprise 1-40, 1-30, 1-20, 1-19, 1-18, 1-15, 1-10, 1-9, 1-8, 1-7, 1-6, 1-5, 1-4, 1-3, 1-2, 2-50, 2-40, 2-30, 2-20, 2-15, 2-10, 2-5, 6-30, 6-15, 16-25, or 31-50 strains from within the same species and, optionally, do not contain bacteria from any other species. The invention comprises any combination of the foregoing.
[0197] In some embodiments, the composition comprises a microbial consortium. For example, in some embodiments, the composition comprises the Roseburia bacterial strain as part of a microbial consortium. For example, in some embodiments, the Roseburia bacterial strain is present in combination with one or more (e.g. at least 2, 3, 4, 5, 10, 15 or 20) other bacterial strains from other genera with which it can live symbiotically in vivo in the intestine. For example, in some embodiments, the composition comprises a bacterial strain of Roseburia in combination with a bacterial strain from a different genus. In some embodiments, the microbial consortium comprises two or more bacterial strains obtained from a faeces sample of a single organism, e.g. a human. In some embodiments, the microbial consortium is not found together in nature. For example, in some embodiments, the microbial consortium comprises bacterial strains obtained from faeces samples of at least two different organisms. In some embodiments, the two different organisms are from the same species, e.g. two different humans. In some embodiments, the two different organisms are an infant human and an adult human. In some embodiments, the two different organisms are a human and a non-human mammal.
[0198] In some embodiments, the composition of the invention additionally comprises a bacterial strain that has the same safety and therapeutic efficacy characteristics as strain MRx0001, but which is not MRx0001, or which is not a Roseburia hominis.
[0199] In some embodiments, the composition of the invention additionally comprises a bacterial strain that has the same safety and therapeutic efficacy characteristics as strain A, but which is not strain A, or which is not a Roseburia intestinalis.
[0200] In some embodiments in which the composition of the invention comprises more than one bacterial strain, species or genus, the individual bacterial strains, species or genera may be for separate, simultaneous or sequential administration. For example, the composition may comprise all of the more than one bacterial strain, species or genera, or the bacterial strains, species or genera may be stored separately and be administered separately, simultaneously or sequentially. In some embodiments, the more than one bacterial strains, species or genera are stored separately but are mixed together prior to use.
[0201] In some embodiments, the bacterial strain for use in the invention is obtained from human adult faeces. In some embodiments in which the composition of the invention comprises more than one bacterial strain, all of the bacterial strains are obtained from human adult faeces or if other bacterial strains are present they are present only in de minimis amounts. The bacteria may have been cultured subsequent to being obtained from the human adult faeces and being used in a composition of the invention.
[0202] In some embodiments, the bacterial strain for use in the invention is obtained from human infant faeces. In some embodiments in which the composition of the invention comprises more than one bacterial strain, all of the bacterial strains are obtained from human infant faeces or if other bacterial strains are present they are present only in de minimis amounts. The bacteria may have been cultured subsequent to being obtained from the human infant faeces and being used in a composition of the invention.
[0203] As mentioned above, in some embodiments, the one or more Roseburia bacterial strains is/are the only therapeutically active agent(s) in a composition of the invention. In some embodiments, the bacterial strain(s) in the composition is/are the only therapeutically active agent(s) in a composition of the invention.
[0204] The compositions for use in accordance with the invention may or may not require marketing approval.
[0205] In certain embodiments, the invention provides the above pharmaceutical composition, wherein said bacterial strain is lyophilised. In certain embodiments, the invention provides the above pharmaceutical composition, wherein said bacterial strain is spray dried. In certain embodiments, the invention provides the above pharmaceutical composition, wherein the bacterial strain is lyophilised or spray dried and wherein it is live. In certain embodiments, the invention provides the above pharmaceutical composition, wherein the bacterial strain is lyophilised or spray dried and wherein it is viable. In certain embodiments, the invention provides the above pharmaceutical composition, wherein the bacterial strain is lyophilised or spray dried and wherein it is capable of partially or totally colonising the intestine. In certain embodiments, the invention provides the above pharmaceutical composition, wherein the bacterial strain is lyophilised or spray dried and wherein it is viable and capable of partially or totally colonising the intestine.
[0206] In some cases, the lyophilised bacterial strain is reconstituted prior to administration. In some cases, the reconstitution is by use of a diluent described herein.
[0207] The compositions of the invention can comprise pharmaceutically acceptable excipients, diluents or carriers.
[0208] In certain embodiments, the invention provides a pharmaceutical composition comprising: a bacterial strain of the invention; and a pharmaceutically acceptable excipient, carrier or diluent; wherein the bacterial strain is in an amount sufficient to treat a neurodegenerative disorder when administered to a subject in need thereof.
[0209] In certain embodiments, the invention provides pharmaceutical composition comprising: a bacterial strain of the invention; and a pharmaceutically acceptable excipient, carrier or diluent; wherein the bacterial strain is in an amount sufficient to treat or prevent a neurodegenerative disorder.
[0210] In certain embodiments, the invention provides the above pharmaceutical composition, wherein the amount of the bacterial strain is from about 1.times.10.sup.3 to about 1.times.10.sup.11 colony forming units per gram with respect to a weight of the composition.
[0211] In certain embodiments, the invention provides the above pharmaceutical composition, wherein the composition is administered at a dose of 1 g, 3 g, 5 g or 10 g.
[0212] In certain embodiments, the invention provides the above pharmaceutical composition, wherein the composition is administered by a method selected from the group consisting of oral, rectal, subcutaneous, nasal, buccal, and sublingual.
[0213] In certain embodiments, the invention provides the above pharmaceutical composition, comprising a carrier selected from the group consisting of lactose, starch, glucose, methyl cellulose, magnesium stearate, mannitol and sorbitol.
[0214] In certain embodiments, the invention provides the above pharmaceutical composition, comprising a diluent selected from the group consisting of ethanol, glycerol and water.
[0215] In certain embodiments, the invention provides the above pharmaceutical composition, comprising an excipient selected from the group consisting of starch, gelatin, glucose, anhydrous lactose, free-flow lactose, beta-lactose, corn sweetener, acacia, tragacanth, sodium alginate, carboxymethyl cellulose, polyethylene glycol, sodium oleate, sodium stearate, magnesium stearate, sodium benzoate, sodium acetate and sodium chloride.
[0216] In certain embodiments, the invention provides the above pharmaceutical composition, further comprising at least one of a preservative, an antioxidant and a stabilizer.
[0217] In certain embodiments, the invention provides the above pharmaceutical composition, comprising a preservative selected from the group consisting of sodium benzoate, sorbic acid and esters of p-hydroxybenzoic acid.
[0218] In certain embodiments, the invention provides the above pharmaceutical composition, wherein said bacterial strain is lyophilised.
[0219] In certain embodiments, the invention provides the above pharmaceutical composition, wherein when the composition is stored in a sealed container at about 4-C or about 25-C and the container is placed in an atmosphere having 50% relative humidity, at least 80% of the bacterial strain as measured in colony forming units, remains after a period of at least about: 1 month, 3 months, 6 months, 1 year, 1.5 years, 2 years, 2.5 years or 3 years.
[0220] In some embodiments, the composition of the invention is provided in a sealed container comprising a composition as described herein. In some embodiments, the sealed container is a sachet or bottle. In some embodiments, the composition of the invention is provided in a syringe comprising a composition as described herein.
[0221] The composition of the present invention may, in some embodiments, be provided as a pharmaceutical formulation. For example, the composition may be provided as a tablet or capsule. In some embodiments, the capsule is a gelatine capsule ("gel-cap").
[0222] In some embodiments, the compositions of the invention are administered orally. Oral administration may involve swallowing, so that the compound enters the gastrointestinal tract, and/or buccal, lingual, or sublingual administration by which the compound enters the blood stream directly from the mouth.
[0223] Pharmaceutical formulations suitable for oral administration include solid plugs, solid microparticulates, semi-solid and liquid (including multiple phases or dispersed systems) such as tablets; soft or hard capsules containing multi- or nano-particulates, liquids (e.g. aqueous solutions), emulsions or powders; lozenges (including liquid-filled); chews; gels; fast dispersing dosage forms; films; ovules; sprays; and buccal/mucoadhesive patches.
[0224] In some embodiments the pharmaceutical formulation is an enteric formulation, i.e. a gastro-resistant formulation (for example, resistant to gastric pH) that is suitable for delivery of the composition of the invention to the intestine by oral administration. Enteric formulations may be particularly useful when the bacteria or another component of the composition is acid-sensitive, e.g. prone to degradation under gastric conditions.
[0225] In some embodiments, the enteric formulation comprises an enteric coating. In some embodiments, the formulation is an enteric-coated dosage form. For example, the formulation may be an enteric-coated tablet or an enteric-coated capsule, or the like. The enteric coating may be a conventional enteric coating, for example, a conventional coating for a tablet, capsule, or the like for oral delivery. The formulation may comprise a film coating, for example, a thin film layer of an enteric polymer, e.g. an acid-insoluble polymer.
[0226] In some embodiments, the enteric formulation is intrinsically enteric, for example, gastro-resistant without the need for an enteric coating. Thus, in some embodiments, the formulation is an enteric formulation that does not comprise an enteric coating. In some embodiments, the formulation is a capsule made from a thermogelling material. In some embodiments, the thermogelling material is a cellulosic material, such as methylcellulose, hydroxymethylcellulose or hydroxypropylmethylcellulose (HPMC). In some embodiments, the capsule comprises a shell that does not contain any film forming polymer. In some embodiments, the capsule comprises a shell and the shell comprises hydroxypropylmethylcellulose and does not comprise any film forming polymer (e.g. see [46]). In some embodiments, the formulation is an intrinsically enteric capsule (for example, Vcaps.RTM. from Capsugel).
[0227] In some embodiments, the formulation is a soft capsule. Soft capsules are capsules which may, owing to additions of softeners, such as, for example, glycerol, sorbitol, maltitol and polyethylene glycols, present in the capsule shell, have a certain elasticity and softness. Soft capsules can be produced, for example, on the basis of gelatine or starch. Gelatine-based soft capsules are commercially available from various suppliers. Depending on the method of administration, such as, for example, orally or rectally, soft capsules can have various shapes, they can be, for example, round, oval, oblong or torpedo-shaped. Soft capsules can be produced by conventional processes, such as, for example, by the Scherer process, the Accogel process or the droplet or blowing process.
[0228] Culturing Methods
[0229] The bacterial strains for use in the present invention can be cultured using standard microbiology techniques as detailed in, for example, references [47-49].
[0230] The solid or liquid medium used for culture may be YCFA agar or YCFA medium. YCFA medium may include (per 100 ml, approximate values): Casitone (1.0 g), yeast extract (0.25 g), NaHCO.sub.3 (0.4 g), cysteine (0.1 g), K.sub.2HPO.sub.4 (0.045 g), KH.sub.2PO.sub.4 (0.045 g), NaCl (0.09 g), (NH.sub.4).sub.2SO.sub.4 (0.09 g), MgSO.sub.4.7H.sub.2O (0.009 g), CaCl.sub.2 (0.009 g), resazurin (0.1 mg), hemin (1 mg), biotin (1 .mu.g), cobalamin (1 .mu.g), p-aminobenzoic acid (3 .mu.g), folic acid (5 .mu.g), and pyridoxamine (15 .mu.g).
[0231] Bacterial Strains for Use in Vaccine Compositions
[0232] The inventors have identified that the bacterial strains of the invention are useful for treating or preventing neurodegenerative disorders. This is likely to be a result of the effect that the bacterial strains of the invention have on the host immune system. Therefore, the compositions of the invention may also be useful for preventing neurodegenerative disorders, when administered as vaccine compositions. In certain such embodiments, the bacterial strains of the invention may be killed, inactivated or attenuated. In certain such embodiments, the compositions may comprise a vaccine adjuvant. In certain embodiments, the compositions are for administration via injection, such as via subcutaneous injection.
[0233] General
[0234] The practice of the present invention will employ, unless otherwise indicated, conventional methods of chemistry, biochemistry, molecular biology, immunology and pharmacology, within the skill of the art. Such techniques are explained fully in the literature. See, e.g., references [50] and [51-57], etc.
[0235] The term "comprising" encompasses "including" as well as "consisting" e.g. a composition "comprising" X may consist exclusively of X or may include something additional e.g. X+Y.
[0236] The term "about" in relation to a numerical value x is optional and means, for example, x.+-.10%.
[0237] The word "substantially" does not exclude "completely" e.g. a composition which is "substantially free" from Y may be completely free from Y. Where necessary, the word "substantially" may be omitted from the definition of the invention.
[0238] References to a percentage sequence identity between two nucleotide sequences means that, when aligned, that percentage of nucleotides are the same in comparing the two sequences. This alignment and the percent homology or sequence identity can be determined using software programs known in the art, for example those described in section 7.7.18 of ref. [58]. A preferred alignment is determined by the Smith-Waterman homology search algorithm using an affine gap search with a gap open penalty of 12 and a gap extension penalty of 2, BLOSUM matrix of 62. The Smith-Waterman homology search algorithm is disclosed in ref [59].
[0239] Unless specifically stated, a process or method comprising numerous steps may comprise additional steps at the beginning or end of the method, or may comprise additional intervening steps. Also, steps may be combined, omitted or performed in an alternative order, if appropriate. Various embodiments of the invention are described herein. It will be appreciated that the features specified in each embodiment may be combined with other specified features, to provide further embodiments. In particular, embodiments highlighted herein as being suitable, typical or preferred may be combined with each other (except when they are mutually exclusive).
MODES FOR CARRYING OUT THE INVENTION
Example 1--Efficacy of Bacterial Inocula to Reduce IL-6 Secretion
[0240] Summary
[0241] Activation of proinflammatory cytokines has been associated with neuron damage in neurodegenerative disease. Lipopolysaccharide (LPS) is a known stimulator of the proinflammatory cytokine IL-6. Human glioblastoma astrocytoma cells were treated with compositions comprising bacterial strains according to the invention in combination with LPS to observe their ability to modulate the levels of IL-6.
[0242] Material and Methods
[0243] Bacterial Strain
[0244] Roseburia hominis MRx0001
[0245] Cell Line
[0246] MG U373 is a human glioblastoma astrocytoma derived from a malignant tumour and were purchased from Sigma-Aldrich (cat n. 08061901-1VL). MG U373 human glioblastoma astrocytoma cells were grown in MEM (Sigma Aldrich, cat n. M-2279) supplemented with 10% FBS, 1% Pen Strep, 4 mM L-Glut, 1.times. MEM Non essential Amino Acid solution and 1.times. Sodium Piruvate.
[0247] Method
[0248] Once grown the MG U373 cells were plated on 24-well plate at 100,000 cells/well. The cells were treated with LPS (1 ug/mL) alone or with 10% of bacteria supernatant from MRx0001 for 24 h. A control was also performed where the cells were incubated in untreated media. The cells were treated with 10% of bacteria supernatant from MRx0001 or YCFA alone for 24 h. A control was also performed where the cells were incubated in untreated media. Afterwards the cell free supernatants were collected, centrifuged at 10,000 g for 3 min at 4.degree. C. IL-6 was measured using the Human IL-6 ELISA Kit from Peprotech (cat n. #900-K16) according to manufacturer instruction.
[0249] Results
[0250] The results of these experiments are shown in FIG. 1 and FIG. 9. Treatment of neuroblastoma cells with LPS and the bacteria strain led to a decrease in the level of IL-6 secreted to the same level as the control cells that were untreated with LPS (FIG. 1). Treatment of neuroblastoma cells with Roseburia hominis led to a decrease in the level of IL-6 secreted below the levels observed with the cells treated with the control (FIG. 9).
Example 2--Efficacy of Bacterial Inocula to Reduce NF.kappa.B Activation
[0251] Summary
[0252] Activation of the NF.kappa.B promoter leads to the production of proinflammatory cytokines including IL-10, IL-la, IL-18, TNF.alpha. and IL-6. The NF.kappa.B promoter can be activated by .alpha.-synuclein and LPS by stimulating the TLR4 ligand. Mutations in .alpha.-synuclein, such as .alpha.-synuclein A53T, are implicated in familial Parkinson's. Treatment of neuronal cells with LPS simulates Parkinson's caused by environmental factors. The ability of compositions comprising bacterial strains according to the invention to inhibit the activation of the NF.kappa.B promoter was investigated.
[0253] Material and Methods
[0254] Bacterial Strain
[0255] Roseburia hominis MRx0001
[0256] Cell Line
[0257] Human Hek blue TLR4 were purchased from InvivoGen (cat n. hkb-htlr4). Human Hek blue TLR4 were grown in DMEM high glucose (Sigma Aldrich, cat n. D-6171) supplemented with 10% FBS, 1% Pen Strep, 4 mM L-Glut, Normocin and 1.times. HEK Blue selection solution.
[0258] Method
[0259] Once grown the Human Hek blue cells were plated in 96 well plate at 25,000 cells/well in 4 replicates. One set of cells were treated with .alpha.-synuclein A53T (1 ug/mL) alone or with 10% of bacteria supernatant from MRx0001 for 22 h. The second set of cells were treated with LPS (10 ng/mL, from Salmonella enterica serotype Typhimurium, Sigma Aldrich, cat n. L6143) alone or with 10% of bacteria supernatant from MRx0001 for 22 h. The cells were subsequently spun down and 20 ul of the supernatant was mixed with 200 ul of Quanti Blue reagent (InvivoGen, cat n. rep-qb2), incubated for 2 h and absorbance read at 655 nm.
[0260] Results
[0261] The results of these experiments are shown in FIGS. 2 and 3. FIG. 2 shows that the activation of the NF.kappa.B promoter by .alpha.-synuclein is inhibited by MRx0001. FIG. 3 shows that the activation of the NF.kappa.B promoter by LPS is inhibited by MRx0001.
Example 3--Efficacy of Bacterial Inocula to Alter Antioxidant Capacity
[0262] Summary
[0263] The ability of compositions comprising bacterial strains according to the invention to alter the antioxidant capacity. The antioxidant capacity of the bacterial strain was established using the well-known ABTS (2,2'-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid)) assay.
[0264] Bacterial Strain
[0265] Roseburia hominis MRx0001
[0266] Method
[0267] Bacterial cells (10.sup.6 or greater) were collected and centrifuged. They were resuspended in assay buffer (using three times the pellet volume). The suspension was sonicated on ice for 5 minutes and then spun down at 12,000.times.g for 10 minutes. The supernatant was removed and measured using the ABTS assay kit produced by Sigma Aldrich (code CS0790), in accordance with manufacturer's instructions.
[0268] Results
[0269] The results of these experiments are shown in FIG. 4. FIG. 4 shows that MRx0001 has an antioxidant capacity of approximately 1 mM compared to Trolox.
Example 4--Efficacy of Bacterial Inocula to Alter Lipid Peroxidation Levels
[0270] Summary
[0271] The ability of compositions comprising bacterial strains according to the invention to alter lipid peroxidation levels was investigated. The thiobarbituric reactive substances assay (TBARs) was used to measure the by-products of lipid peroxidation.
[0272] Material and Methods
[0273] Bacterial Strain
[0274] Roseburia hominis MRx0001
[0275] Method
[0276] Bacterial cells (10.sup.6 or greater) were collected and centrifuged, a wash step was performed with isotonic saline before the pellet was re-suspensed in potassium chloride assay buffer. The suspension was sonicated on ice for 10 minutes and then spun down at 10,000.times.g for 10 minutes. The supernatant was removed and the level of lipid peroxidation evaluated using the thiobarbituric reactive substances assay.
[0277] Results
[0278] The results of the experiments are shown in FIG. 5. FIG. 5 shows that MRx0001 is able to inhibit lipid peroxidation by approximately 18%, which is a higher antioxidant capacity than the positive control, butylated hydroxytoluene (1% w/v).
Example 5--Efficacy of Bacterial Inocula on Histone Deacetylatase Activity
[0279] Summary
[0280] The ability of compositions comprising bacterial strains according to the invention to alter histone deacetylatase activity was investigated. Dysregulation of histone deacetylatase has been implicated in the pathogenesis associated with age-associated neurodegenerative diseases.
[0281] Material and Methods
[0282] Bacterial Strain
[0283] Roseburia hominis MRx0001
[0284] Cell Line
[0285] The cell line HT-29 was used because histone deacetylase is present.
[0286] Method
[0287] Cell free supernatants of stationary phase bacterial cultures were isolated by centrifugation and filtering in a 0.22 uM filter. HT-29 cells were used 3 days' post confluence and stepped down in 1 mL DTS 24 hours prior to commencement of the experiment. The HT-29 cells were challenged with 10% cell free supernatant diluted in DTS and this was left to incubate for 48 hours. Nuclease proteins were then extracted using the Sigma Aldrich Nuclease extraction kit and samples were snap frozen prior to HDAC activity measurement. HDAC activity was assessed fluorometrically using the Sigma Aldrich (UK) kit.
[0288] Results
[0289] The results of the experiments are shown in FIG. 6. FIG. 6 shows that MRx0001 is able reduce the levels of histone deacetylase activity.
Example 6--Level of Indole Production in Bacteria
[0290] Summary
[0291] The ability of the bacteria of the invention to produce indole was investigated. Indole has been implicated in attenuating inflammation and oxidative stress.
[0292] Material and Methods
[0293] Bacterial Strain
[0294] Roseburia hominis MRx0001
[0295] ATCC 11775 is a bacterial reference strain that is known to produce indole.
[0296] Method
[0297] Intact bacterial cells in stationary phase were incubated with 6 mM Tryptophan for 48 hours. Bacterial species which possess the enzyme tryptophanase will utilise tryptophan as a substrate to produce indole. Following the 48 hour incubation period, the supernatant was removed and added to Kovac's reagent for quantification of indole. Standards, stock solutions and reagents were prepared using standardised methods validated in-house.
[0298] Results
[0299] The results of the experiments are shown in FIG. 7. FIG. 7 shows that MRx0001 has the capacity to produce indole from tryptophan, at concentrations of approximately 0.25 mM.
Example 7--Level of Kynurenine Production in Bacteria
[0300] Summary
[0301] The ability of the bacteria of the invention to produce kynurenine was investigated. Dysregulation of the kynurenine pathway can lead to activation of the immune system and the accumulation of potentially neurotoxic compounds. Alterations in the kynurenine metabolism may be involved in the development of Parkinson's diseases.
[0302] Bacterial Strain
[0303] Roseburia hominis MRx0001
[0304] DSM 17136 is a strain of Bacteroides copricola that is known to produce kynurenine.
[0305] Method
[0306] Cell free supernatants of stationary phase bacterial cultures were isolated by centrifugation and filtering in a 0.22 uM filter and frozen until use. Kynurenine standards, stock solutions and reagents were prepared using standardised methods validated in-house. Sample were treated with trichloroacetic acid and centrifuged at 10,000.times.g for 10 minutes at 4.degree. C. The supernatant was collected and dispensed into a 96 well plate. Ehrlich's reagent was used for kynurenine detection and added at a ratio of 1:1.
[0307] Results
[0308] The results of the experiments are shown in FIG. 8. FIG. 8 shows that MRx0001 has the capacity to produce kynurenine at a concentration of approximately 50 .mu.M.
Example 8--Stability Testing
[0309] A composition described herein containing at least one bacterial strain described herein is stored in a sealed container at 25.degree. C. or 4.degree. C. and the container is placed in an atmosphere having 30%, 40%, 50%, 60%, 70%, 75%, 80%, 90% or 95% relative humidity. After 1 month, 2 months, 3 months, 6 months, 1 year, 1.5 years, 2 years, 2.5 years or 3 years, at least 50%, 60%, 70%, 80% or 90% of the bacterial strain shall remain as measured in colony forming units determined by standard protocols.
Example 9--Efficacy of Bacterial Inocula to Reduce IL-6 Secretion
[0310] Summary
[0311] Activation of proinflammatory cytokines has been associated with neuron damage in neurodegenerative disease. Lipopolysaccharide (LPS) is a known stimulator of the proinflammatory cytokine IL-6. Human glioblastoma astrocytoma cells were treated with compositions comprising bacterial strains according to the invention in combination with LPS to observe their ability to modulate the levels of IL-6.
[0312] Material and Methods
[0313] Bacterial Strain
[0314] Roseburia intestinalis Strain A
[0315] Roseburia faecis Strain B
[0316] Cell Line
[0317] MG U373 is a human glioblastoma astrocytoma derived from a malignant tumour and were purchased from Sigma-Aldrich (cat n. 08061901-1VL). MG U373 human glioblastoma astrocytoma cells were grown in MEM (Sigma Aldrich, cat n. M-2279) supplemented with 10% FBS, 1% Pen Strep, 4 mM L-Glut, 1.times. MEM Non essential Amino Acid solution and 1.times. Sodium Piruvate. Method
[0318] Once grown the MG U373 cells were plated on 24-well plate at 100,000 cells/well. The cells were treated with the following conditions for 24 h: [0319] LPS (1 ug/mL) [0320] LPS with 10% of bacteria supernatant from Strain A [0321] LPS with 10% of bacteria supernatant from Strain B [0322] LPS with YCFA media [0323] YCFA
[0324] A control was also performed where the cells were incubated in untreated media.
[0325] Afterwards the cell free supernatants were collected, centrifuged at 10,000 g for 3 min at 4.degree. C. IL-6 was measured using the Human IL-6 ELISA Kit from Peprotech (cat n. #900-K16) according to manufacturer instruction.
[0326] Results
[0327] The results of these experiments are shown in FIGS. 10 and 12. Treatment of neuroblastoma cells with LPS and the bacteria strain A and B led to a decrease in the level of IL-6 secreted to the same level as the control cells that were untreated with LPS (FIGS. 10A and 12A).
[0328] Treatment of neuroblastoma cells with Roseburia intestinalis and Roseburia faecis led to a decrease in the level of IL-6 secreted below the levels observed with the cells treated with the control (FIGS. 10B and 12B).
Example 10--Efficacy of Bacterial Inocula to Reduce NF.kappa.B Activation
[0329] Summary
[0330] Activation of the NF.kappa.B promoter leads to the production of proinflammatory cytokines including IL-1.beta., IL-1.alpha., IL-18, TNF.alpha. and IL-6. The NF.kappa.B promoter can be activated by .alpha.-synuclein and LPS by stimulating the TLR4 ligand. Mutations in .alpha.-synuclein, such as .alpha.-synuclein A53T, are implicated in familial Parkinson's. Treatment of neuronal cells with LPS simulates Parkinson's caused by environmental factors. The ability of compositions comprising bacterial strains according to the invention to inhibit the activation of the NF.kappa.B promoter was investigated.
[0331] Material and Methods
[0332] Bacterial Strain
[0333] Roseburia intestinalis Strain A
[0334] Roseburia faecis Strain B
[0335] Cell Line
[0336] Human Hek blue TLR4 were purchased from InvivoGen (cat n. hkb-htlr4). Human Hek blue TLR4 were grown in DMEM high glucose (Sigma Aldrich, cat n. D-6171) supplemented with 10% FBS, 1% Pen Strep, 4 mM L-Glut, Normocin and 1.times. HEK Blue selection solution.
[0337] Method
[0338] Once grown the Human Hek blue cells were plated in 96 well plate at 25,000 cells/well in 4 replicates. One set of cells were treated with .alpha.-synuclein A53T (1 ug/mL) alone or with 10% of bacteria supernatant from Strain A or Strain B for 22 h. The second set of cells were treated with LPS (10 ng/mL, from Salmonella enterica serotype Typhimurium, Sigma Aldrich, cat n. L6143) alone or with 10% of bacteria supernatant from Strain A or Strain B for 22 h. The cells were subsequently spun down and 20 ul of the supernatant was mixed with 200 ul of Quanti Blue reagent (InvivoGen, cat n. rep-qb2), incubated for 2 h and absorbance read at 655 nm.
[0339] Results
[0340] The results of these experiments are shown in FIGS. 11 and 13.
TABLE-US-00001 Sequences (Roseburia hominis strain A2-181 16S ribosomal RNA gene, partial sequence - AY804148) SEQ ID NO: 1 1 taaaggttga tcctggctca ggatgaacgc tggaggcgtg cttaacacat gcaagtcgaa 61 cgaagcactt taattgattt cttcggaatg aagtttttgt gactgagtgg cggacgggtg 121 agtaacgcgt gggtaacctc gctcatacag ggggataaca gttggaaacg actgctaata 181 ccgcataagc gcacaggatt gcatgatcca gtgtgaaaaa ctccggtggt atgagatgga 241 cccgcgtctg attagccagt tggcggggta acggcccacc aaagcgacga tcagtagccg 301 acctgagagg gtgaccggcc acattgggac tgagacacgg cccaaactcc tacgggaggc 361 agcagtgggt aatattgcac aatgggggaa accctgatgc agcgacgccg agtgagcgaa 421 gaagtatttc ggtatgtaaa gctctatcag caggaagaag aatgacggta cctgactaaa 481 aagcaccggc taaatacgtg ccagcagccg cggtaatacg tatggtgcaa gcgttatccg 541 gatttactgg gtgtaaaggg agcgcaggcg gtacggcaag tctgatgtga aatcccgggg 601 ctcaaccccg gtactgcatt ggaaactgtc ggactagggt gtctgagggg taagtggaat 661 tcctagtgta gcggtgaaat gcgtagatat taggaggaac accagtggcg aaggcggctt 721 actggacgat tactgacgct gaggctcgaa agcgtgggga gcaaacagga ttagataccc 781 tggtagtcca cgccgtaaac gatgaatact aggtgtcggg gagcattgct cttcggtgcc 841 gcagcaaacg caataagtat tccacctggg gagtacgttc gcaagaatga aactcaaagg 901 aattgacggg gacccgcaca agcggtggag catgtggttt aattcgaagc aacgcgaaga 961 accttaccaa gtcttgacat cccactgaca aagtatgtaa tgtactttct cttcggagca 1021 gtggtgacag gtggtgcatg gttgtcgtca gctcgtgtcg tgagatgttg ggttaagtcc 1081 cgcaacgagc gcaaccccta ttcttagtag ccagcggttt ggccgggcac tctagggaga 1141 ctgccaggga taacctggag gaaggtgggg atgacgtcaa atcatcatgc cccttatgac 1201 ttgggctaca cacgtgctac aatggcgtaa acaaagggaa gcaatcccgc gagggggagc 1261 aaatctcaaa aataacgtct cagttcggac tgtagtctgc aactcgacta cacgaagctg 1321 gaatcgctag taatcgcgaa tcagaatgtc gcggtgaata cgttcccggg tcttgtacac 1381 accgcccgtc acaccatggg agttggtaat gcccgaagtc agtgacccaa ccgcaaggag 1441 ggagctgccg aagcaggact gataactggg gtgaagtcgt aacaagt (Roseburia hominis A2-183 16S rRNA gene, type strain A2-183T - AJ270482) SEQ ID NO: 2 1 gatcctggct caggatgaac gctggcggcg tgcttaacac atgcaagtcg aacgaagcac 61 tttaattgat ttcttcggaa tgaagttttt gtgactgagt ggcggacggg tgagtaacgc 121 gtgggtaacc tgcctcatac agggggataa cagttggaaa cgactgctaa taccgcataa 181 gcgcacagga ttgcatgatc cagtgtgaaa aactccggtg gtatgagatg gacccgcgtc 241 tgattagcca gttggcgggg taacggccca ccaaagcgac gatcagtagc cgacctgaga 301 gggtgaccgg ccacattggg actgagacac ggcccaaact cctacgggag gcagcagtgg 361 ggaatattgc acaatggggg aaaccctgat gcagcgacgc cgcgtgagcg aagaagtatt 421 tcggtatgta aagctctatc agcagggaag aagaatgcgg tacctgacta agaagcaccg 481 gctaaatacg tgccagcagc cgcggtaata cgtatggtgc aagcgttatc cggatttact 541 gggtgtaaag ggagcgcagg cggtacggca agtctgatgt gaaatcccgg ggctcaaccc 601 cggtactgca ttggaaactg tcggactaga gtgtcggagg ggtaagtgga attcctagtg 661 tagcggtgaa atgcgtagat attaggagga acaccagtgg cgaaggcggc ttactggacg 721 attactgacg ctgaggctcg aaagcgtggg gagcaaacag gattagatac cctggtagtc 781 cacgccgtaa acgatgaata ctaggtgtcg gggagcattg ctcttcggtg ccgcagcaaa 841 cgcaataagt attccacctg gggagtacgt tcgcaagaat gaaactcaaa ggaattgacg 901 gggacccgca caagcggtgg agcatgtggt ttaattcgaa gcaacgcgaa gaaccttacc 961 aagtcttgac atcccactga cagagtatgt aatgtacttt ctcttcggag cagtggtgac 1021 aggtggtgca tggttgtcgt cagctcgtgt cgtgagatgt tgggttaagt cccgcaacga 1081 gcgcaacccc tattcttagt agccagcggt tcggccgggc actctaggga gactgccagg 1141 gataacctgg aggaaggtgg ggatgacgtc aaatcatcat gccccttatg acttgggcta 1201 cacacgtgct acaatggcgt aaacaaaggg aagcaatccc gcgaggggga gcaaatctca 1261 aaaataacgt ctcagttcgg actgtagtct gcaactcgac tacacgaagc tggaatcgct 1321 agtaatcgcg aatcagaatg tcgcggtgaa tacgttcccg ggtcttgtac acaccgcccg 1381 tcacaccatg ggagttggta atgcccgaag tcagtgaccc aaccgcaagg agggagctgc 1441 cgaaggcagg actgataact ggggtgaagt cgtaacaagg gtacg (consensus 16S rRNA sequence for Roseburia hominis strain 433) SEQ ID NO: 3 AAGAGTTTGGGHCAGGCTCAGGATGAACGCTGGCGGCGTGCTTAACACATGCAAGTCGAACGAAGCACTTTAAT- TGA TTTCTTCGGAATGAAGTTTTTGTGACTGAGTGGCGGACGGGTGAGTAACGCGTGGGTAACCTGCCTCATACAGG- GGG ATAACAGTTGGAAACGACTGCTAATACCGCATAAGCGCACAGGATTGCATGATCCAGTGTGAAAAACTCCGGTG- GTA TGAGATGGACCCGCGTCTGATTAGCCAGTTGGCGGGGTAACGGCCCACCAAAGCGACGATCAGTAGCCGACCTG- AGA GGGTGACCGGCCACATTGGGACTGAGACACGGCCCAAACTCCTACGGGAGGCAGCAGTGGGGAATATTGCACAA- TGG GGGAAACCCTGATGCAGCGACGCCGCGTGAGCGAAGAAGTATTTCGGTATGTAAAGCTCTATCAGCAGGGAAGA- AGA ATGACGGTACCTGACTAAGAAGCACCGGCTAAATACGTGCCAGCAGCCGCGGTAATACGTATGGTGCAAGCGTT- ATC CGGATTTACTGGGTGTAAAGGGAGCGCAGGCGGTACGGCAAGTCTGATGTGAAATCCCGGGGCTCAACCCCGGT- ACT GCATTGGAAACTGTCGGACTAGAGTGTCGGAGGGGTAAGTGGAATTCCTAGTGTAGCGGTGAAATGCGTAGATA- TTA GGAGGAACACCAGTGGCGAAGGCGGCTTACTGGACGATTACTGACGCTGAGGCTCGAAAGCGTGGGGAGCAAAC- AGG ATTAGATACCCTGGTAGTCCACGCCGTAAACGATGAATACTAGGTGTCGGGGAGCATTGCTCTTCGGTGCCGCA- GCA AACGCAATAAGTATNCCACCTGGGGAGTACGTTCGCAAGAATGAAACTCAAAGGAATTGACGGGGACCCGCACA- AGC GGTGGAGCNTGTGGTTTAATTCGAAGCAACGCGAAGAACCTTACCAAGTCTTGACATCCCACTGACAGAGTATG- TAA TGTACTTTCTCTTCGGAGCAGTGGTGACAGGTGGTGCATGGTTGTCGTCAGCTCGTGTCGTGAGATGTTGGGTT- AAG TCCCGCAACGAGCGCAACCCCTATTCTTAGTAGCCAGCGGTTTGGCCGGGCACTCTAGGGAGACTGCCAGGGAT- AAC CTGGAGGAAGGTGGGGATGACGTCAAATCATCATGCCCCTTATGACTTGGGCTACACACGTGCTACAATGGCGT- AAA CAAAGGGAAGCAATCCCGCGAGGGGGAGCAAATCTCAAAAATAACGTCTCAGTTCGGACTGTAGTCTGCAACTC- GAC TACACGAAGCTGGAATCGCTAGTAATCGCGAATCAGAATGTCGCGGTGAATACGTTCCCGGGTCTTGTACACAC- CGC CCGTCACACCATGGGAGTTGGTAATGCCCGAAGTCAGTGACCCAACCGCAAGGAGGGAGCTGCCGAAGGCAGGA- CTG ATAACTGGGGTGAAGTCTACRSAGGGTAGCCGTRMMC. (Consensus 16S rRNA sequence, Strain A Roseburia intestinalis) SEQ ID NO: 5 gctccctcct tgcggttggg tcactgactt cgggcattac caactcccat ggtgtgacgg 60 gcggtgtgta caagacccgg gaacgtattc accgcgacat tctgattcgc gattactagc 120 gattccagct tcgtgcagtc gagttgcaga ctgcagtccg aactgagacg ttatttttga 180 gatttgctcc ccctcgcagg ctcgcttccc tttgtttacg ccattgtagc acgtgtgtag 240 cccaagtcat aaggggcatg atgatttgac gtcatcccca ccttcctcca ggttatccct 300 ggcagtctcc ctagagtgcc cggcttaccc gctggctact aagaataggg gttgcgctcg 360 ttgcgggact taacccaaca tctcacgaca cgagctgacg acaaccatgc accacctgtc 420 accgatgctc cgaagagaaa acacattaca tgttctgtca tcgggatgtc aagacttggt 480 aaggttcttc gcgttgcttc gaattaaacc acatgctcca ccgcttgtgc gggtccccgt 540 caattccttt gagtttcatt cttgcgaacg tactccccag gtggaatact tattgcgttt 600 gctgcggcac cgaagagcaa tgctccccga cacctagtat tcatcgttta cggcgtggac 660 taccagggta tctaatcctg tttgctcccc acgctttcga gcctcagcgt cagtaatcgt 720 ccagtaagcc gccttcgcca ctggtgttcc tcctaatatc tacgcatttc accgctacac 780 taggaattcc acttacccct ccgacactct agtccgacag tttccaatgc agtaccgggg 840 ttgagccccg ggctttcaca tcagacttgc cgtaccgcct gcgctccctt tacacccagt 900 aaatccggat aacgcttgca ccatacgtat taccgcggct gctggcacgt atttagccgg 960 tgcttcttag tcaggtaccg tcatttcttc ttccctgnct gatagagctt tacataccga 1020 aatacttctt cgctcacgcg gcgtcgctgc atcagggttt cccccattgt gcaatattcc 1080 ccactgctgc ctcccgtagg agtttgggcc gtgtctcagt cccaatgtgg ccggtcaccc 1140 tctcaggtcg gctactgatc gtcgctttgg taggccgtta ccccaccaac tggctaatca 1200 gacgcgggtc catctcatac caccggagtt tttcacacca ggtcatgcga ccctgtgcgc 1260 ttatgcggta ttagcagtcg tttccaactg ttatccccct gtatgaggca ggttacccac 1320 gcgttactca cccgtccgcc actcagtcac aaaatcttca ttccgaagaa atcaaataaa 1380 gtgcttcgtt cgactgca 1398 (Consensus 16S rRNA sequence, Strain B Roseburia faecis) SEQ ID NO: 6 agctccctcc ttgcggttgg gtcactgact tcggacattt ccaactccca tggtgtgacg 60 ggcggtgtgt acaagacccg ggaacgtatt caccgcagca ttctgatctg cgattactag 120 cgattccagc ttcgtgtagt cgggttgcag actacagtcc gaactgagac gttatttttg 180 agatttgctc ggcctcacgg ctttgcttcc ctttgtttac gccattgtag cacgtgtgta 240 gcccaagtca taaggggcat gatgatttga cgtcatcccc gccttcctcc aggttatccc 300 tggcagtctc cctagagtgc ccggccgaac cgctggctac taaggacagg ggttgcgctc 360 gttgcgggac ttaacccaac atctcacgac acgagctgac gacaaccatg caccacctgt 420 caccgatgct ccgaagagaa agtacattac atactctgtc atcgggatgt caagacttgg 480 taaggttctt cgcgttgctt cgaattaaac cacatgctcc accgcttgtg cgggtccccg 540 tcaattcctt tgagtttcat tcttgcgaac gtactcccca ggtggaatac ttattgcgtt 600 tgctgcggca ccgaagagca atgctccccg acacctagta ttcatcgttt acggcgtgga 660 ctaccagggt atctaatcct gtttgctccc cacgctttcg agcctcagcg tcagttatcg 720 tccagtaagc cgccttcgcc actggtgttc ctcctaatat ctacgcattt caccgctaca 780 ctaggaattc cacttacccc tccgacactc tagtacgaca gtttccaatg cagtaccggg 840 gttgagcccc gggctttcac atcagacttg ccgcaccgcc tgcgctccct ttacacccag 900 taaatccgga taacgcttgc accatacgta ttaccgcggc tgctggcacg tatttagccg 960
gtgcttctta gtcaggtacc gtcattcttc ttccctgctg atagagcttt acataccgaa 1020 atacttcttc gctcacgcgg cgtcgctgca tcagggtttc ccccattgtg caatattccc 1080 cactgctgcc tcccgtagga gtttgggccg tgtctcagtc ccaatgtggc cggtcaccct 1140 ctcaggtcgg ctactgatcg tcgctttggt aggccgttac cctgccaact ggctaatcag 1200 acgcgggtcc atctcatacc accggagttt ttcacaccgg atcatgcgat cctgtgcgct 1260 tatgcggtat tagcagtcgt ttccaactgt tatccccctg tatgaggcag gttacccacg 1320 cgttactcac ccgtccgcca ctcagtcaca aaatcttcat ttccgaagaa aatcaaatag 1380 agtgcttcgt ccgactgcag 1400
REFERENCES
[0341] [1] Spor et al. (2011) Nat Rev Microbiol. 9(4):279-90. [0342] [2] Eckburg et al. (2005) Science. 10; 308(5728):1635-8. [0343] [3] Macpherson et al. (2001) Microbes Infect. 3(12):1021-35 [0344] [4] Macpherson et al. (2002) Cell Mol Life Sci. 59(12):2088-96. [0345] [5] Mazmanian et al. (2005) Cell 15; 122(1):107-18. [0346] [6] Frank et al. (2007) PNAS 104(34):13780-5. [0347] [7] Scanlan et al. (2006) J Clin Microbiol. 44(11):3980-8. [0348] [8] Kang et al. (2010) Inflamm Bowel Dis. 16(12):2034-42. [0349] [9] Machiels et al. (2013) Gut. 63(8):1275-83. [0350] [10] WO 2013/050792 [0351] [11] WO 03/046580 [0352] [12] WO 2013/008039 [0353] [13] WO 2014/167338 [0354] [14] Goldin and Gorbach (2008) Clin Infect Dis. 46 Suppl 2:S96-100. [0355] [15] Azad et al. (2013) BMJ. 347:f6471. [0356] [16] WO2013/050792 [0357] [17] Mayer et al (2014) The Journal of Neuroscience 34(46):15490-15496 [0358] [18] Cryan and Dinan (2015) Neuropsychopharmacology, 40: 241-2. [0359] [19] Zhou and Foster (2015) Neuropsychiatric Disease and Treatment 11: 715-723. [0360] [20] Wang and Kasper (2014) Brain Behav Immun. 38: 1-12. [0361] [21] WO2016/203221 [0362] [22] Chapter 38--Nonsteroidal anti-inflammatory drugs exposure and the central nervous system (2014) Handbook of Clinical Neurology 119, 577-584 [0363] [23] Stanton and Savage (1983) Appl Environ Microbiol. 45(5):1677-84 [0364] [24] Duncan et al. (2006) Int. J Syst. Evol. Microbiol. 56: 2437-2441 [0365] [25] Srntkovi et al. (2011) J. Microbiol. Methods, 87(1):10-6. [0366] [26] Pal R et al, Neurol Res 2016, 38(12):1111-1122 [0367] [27] Daniele S G et al, Sci Signal 2015, 8(376):ra45 [0368] [28] Wang et al. (2016) J Neurogastroenterol Motil 22: 589-605. [0369] [29] Foguem & Manckoundia (2018) Current Neurology and Neuroscience Reports, 18: 24 [0370] [30] Ludolph et al. (2009) Eur J Neurol. 16(3): 297-309. [0371] [31] Galpern & Lang (2006) Neurological Progress 59 (3) 449-458 [0372] [32] Zadori et al (2012) Journal of Neural Transmission, 119, 2, 275-283 [0373] [33] Lee et al (2008) European J. Cell Biology 87:389-397 [0374] [34] Pirooznia and Elefant (2013) Front Cell Neurosci. 7: 30. [0375] [35] Tang, et al. (2017) J Am Heart Assoc, 6(10). [0376] [36] Wang et al. (2015) PNAS 112(9):2583-2858 [0377] [37] Psaty et al. (2003) JAMA, 289(19):2534-44 [0378] [38] Lancet. (1995) 346(8991-8992):1647-53 [0379] [39] Miyamoto-Shinohara et al. (2008) J. Gen. Appl. Microbiol., 54, 9-24. [0380] [40] Cryopreservation and Freeze-Drying Protocols, ed. by Day and McLellan, Humana Press. [0381] [41] Leslie et al. (1995) Appl. Environ. Microbiol. 61, 3592-3597. [0382] [42] Mitropoulou et al. (2013) J Nutr Metab. (2013) 716861. [0383] [43] Kailasapathy et al. (2002) CurrIssues Intest Microbiol. 3(2):39-48. [0384] [44] Handbook of Pharmaceutical Excipients, 2nd Edition, (1994), Edited by A Wade and PJ Weller [0385] [45] Remington's Pharmaceutical Sciences, Mack Publishing Co. (A. R. Gennaro edit. 1985) [0386] [46] US 2016/0067188 [0387] [47] Handbook of Microbiological Media, Fourth Edition (2010) Ronald Atlas, CRC Press. [0388] [48] Maintaining Cultures for Biotechnology and Industry (1996) Jennie C. Hunter-Cevera, Academic Press [0389] [49] Strobel (2009) Methods Mol Biol. 581:247-61. [0390] [50] Gennaro (2000) Remington: The Science and Practice of Pharmacy. 20th edition, ISBN: 0683306472. [0391] [51] Molecular Biology Techniques: An Intensive Laboratory Course, (Ream et al., eds., 1998, Academic Press). [0392] [52] Methods In Enzymology (S. Colowick and N. Kaplan, eds., Academic Press, Inc.) [0393] [53] Handbook of Experimental Immunology, Vols. I-IV (D. M. Weir and C. C. Blackwell, eds, 1986, Blackwell Scientific Publications) [0394] [54] Sambrook et al. (2001) Molecular Cloning: A Laboratory Manual, 3rd edition (Cold Spring Harbor Laboratory Press). [0395] [55] Handbook of Surface and Colloidal Chemistry (Birdi, K. S. ed., CRC Press, 1997) [0396] [56] Ausubel et al. (eds) (2002) Short protocols in molecular biology, 5th edition (Current Protocols). [0397] [57] PCR (Introduction to Biotechniques Series), 2nd ed. (Newton & Graham eds., 1997, Springer Verlag) [0398] [58] Current Protocols in Molecular Biology (F. M. Ausubel et al., eds., 1987) Supplement 30 [0399] [59] Smith & Waterman (1981) Adv. Appl. Math. 2: 482-489.
Sequence CWU 0 SQTB SEQUENCE LISTING The patent application
contains a lengthy "Sequence Listing" section. A copy of the
"Sequence Listing" is available in electronic form from the USPTO
web site
(https://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20220096565A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
0 SQTB SEQUENCE LISTING The patent application contains a lengthy
"Sequence Listing" section. A copy of the "Sequence Listing" is
available in electronic form from the USPTO web site
(https://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20220096565A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
References
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.