Method For Producing Bioethanol From Dates
ALEID; SALAH MOHAMMED ; et al.
U.S. patent application number 17/106052 was filed with the patent office on 2021-05-27 for method for producing bioethanol from dates. The applicant listed for this patent is KING FAISAL UNIVERSITY. Invention is credited to SALAH MOHAMMED ALEID, Siddig Hussein Hamad.
| Application Number | 20210155960 17/106052 |
| Document ID | / |
| Family ID | 1000005373782 |
| Filed Date | 2021-05-27 |









View All Diagrams
| United States Patent Application | 20210155960 |
| Kind Code | A1 |
| ALEID; SALAH MOHAMMED ; et al. | May 27, 2021 |
METHOD FOR PRODUCING BIOETHANOL FROM DATES
Abstract
The method for producing bioethanol from dates includes manufacturing a suitable substrate for bioethanol from dates and fermenting the date substrate to produce bioethanol. The date substrate may be produced by de-pitting date fruits, heating the flesh with water to produce a mixture, filter pressing the mixture to produce a juice, and concentrated by vacuum drying to produce a date substrate. The date substrate may then be fermented in either a batch or a fed-batch culture. The fermentation may be performed with a thermophilic yeast, such K. marxianus. In an alternative embodiment the date substrate may be a date fruit extract.
| Inventors: | ALEID; SALAH MOHAMMED; (Alahsa, SA) ; Hamad; Siddig Hussein; (Alahsa, SA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005373782 | ||||||||||
| Appl. No.: | 17/106052 | ||||||||||
| Filed: | November 27, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16391497 | Apr 23, 2019 | 10870868 | ||
| 17106052 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C12M 41/36 20130101; C12M 45/02 20130101; C12P 7/08 20130101; C12M 21/12 20130101; C12N 1/16 20130101; C12M 23/08 20130101 |
| International Class: | C12P 7/08 20060101 C12P007/08; C12N 1/16 20060101 C12N001/16; C12M 1/00 20060101 C12M001/00; C12M 1/33 20060101 C12M001/33 |
Claims
1-19. (canceled)
20. A bioethanol produced from dates, comprising: e a fermented date substrate, wherein the substrate is obtained by: de-pitting date fruits to produce date fruit flesh; heating the date fruit flesh with water to produce a mixture; filter pressing the mixture to produce a juice; concentrating the juice to produce a date substrate, wherein the date substrate used for the fermentation has a starting sugar concentration ranging from 20% to 25%; and fermenting the date substrate with yeast at between 30.degree. C.-40.degree. C. for 32 hours, wherein the yeast is selected from the group consisting of S. cerevisiae and S. cerevisiae NCYC 431, to produce a 11.2-11.8% w/v bioethanol concentration.
Description
INCORPORATION BY REFERENCE OF MATERIAL SUBMITTED IN COMPUTER READABLE FORM
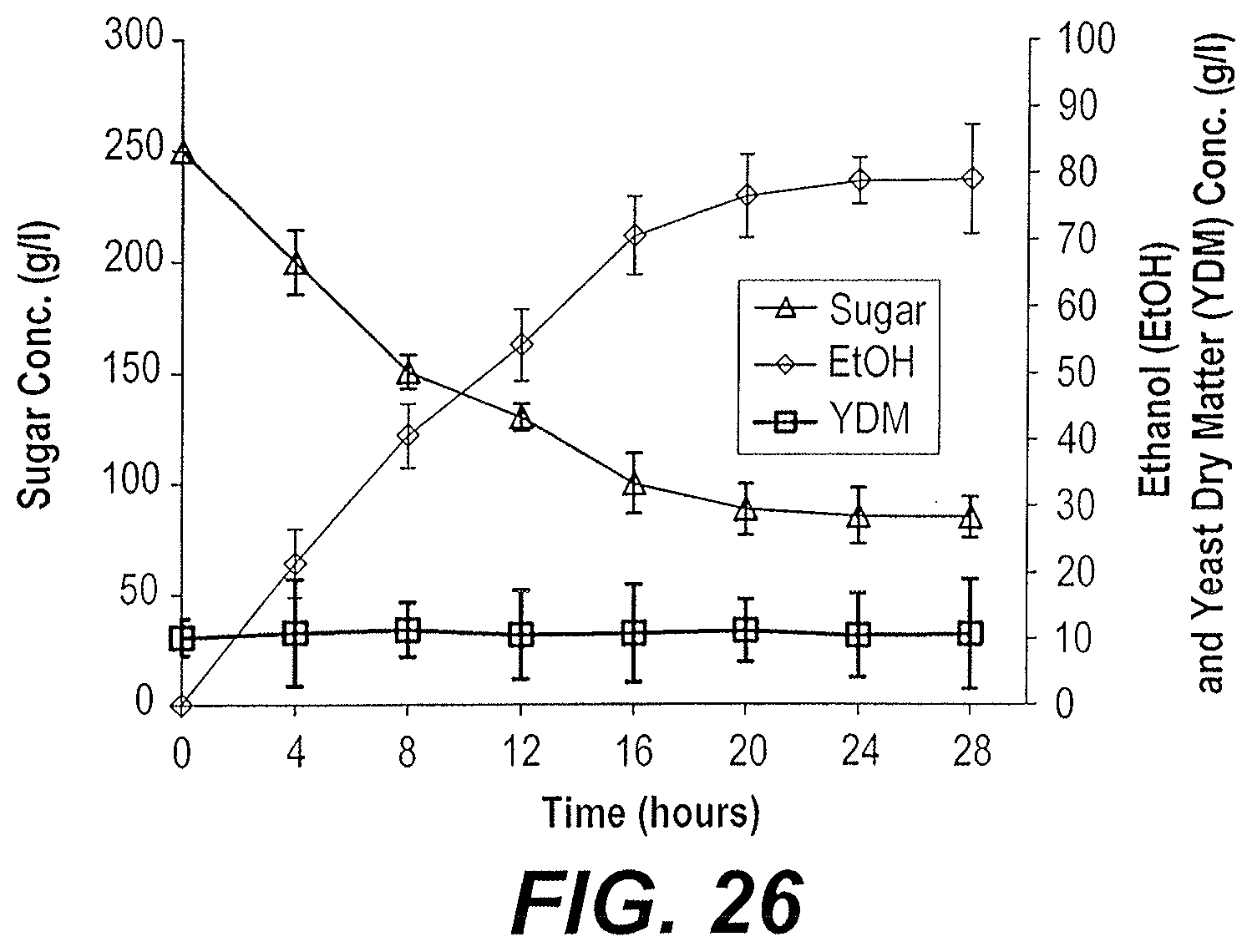
[0001] The Applicant hereby incorporates by reference the sequence listing contained in the ASCII text file titled 32087_09_sequence_listing_ST25.txt, created Mar. 14, 2019, and having 17 KB of data.
BACKGROUND
1. Field
[0002] The disclosure of the present patent application relates to biofuels, and particularly to a method for producing bioethanol from dates.
2. Description of the Related Art
[0003] Bioethanol is ethyl alcohol produced from biological sources. Bioethanol can be used as fuel, or as a fuel additive for gasoline. Bioethanol is commonly made from biomass such as corn or sugarcane. Other common sources for bioethanol include potato, cassava, barley, wheat, and hemp.
[0004] Bioethanol is manufactured from grains by milling the grains into meal, mixing the meal with water and alpha-amylase and liquefying the resulting mash. This step is followed by saccharification with a secondary enzyme to convert the liquefied starch to fermentable sugars, fermentation with yeast, distillation of the fermented mash to concentrate the alcohol, and dehydration to remove any remaining water, resulting in anhydrous ethanol.
[0005] The traditional approach to manufacturing bioethanol suffers from a number of drawbacks. The use of grains to produce bioethanol could contribute to rising food prices. The process of producing ethanol from grain is highly inefficient, requiring many wasteful steps, including hydrolysis of starch by addition of enzymes and acids.
[0006] Thus, a method for producing bioethanol from dates solving the aforementioned problems is desired.
SUMMARY
[0007] The method for producing bioethanol from dates includes manufacturing a suitable substrate for bioethanol from dates and fermenting the date substrate to produce bioethanol. In one embodiment, the date substrate can be produced by de-pitting date fruits, heating the flesh with water to produce a mixture, filter pressing the mixture to produce a juice, and concentrating the juice by vacuum drying to produce a date substrate. The date substrate may then be fermented in either a batch or a fed-batch culture. In the batch process, the entire volume of substrate may be added to a bioreactor along with a yeast inoculum and fermentation may be conducted at either 30 or 40.degree. C., depending upon the strain of yeast used as the inoculum. In the fed-batch process, the substrate may be fed gradually into the bioreactor containing the yeast inoculum at a constant feeding rate and fermentation may be conducted at either 30.degree. C. or 40.degree. C., depending upon the strain of yeast used as the inoculum.
[0008] In an alternative embodiment, the date substrate may be date extract.
[0009] These and other features of the present disclosure will become readily apparent upon further review of the following specification and drawings.
BRIEF DESCRIPTION OF THE DRAWINGS
[0010] FIG. 1: depicts a graph of ethanol production from date extract in batch using a commercial strain of S. cerevisiae.
[0011] FIG. 2: depicts a graph of specific and volumetric productivities of ethanol from date extract in batch using a commercial strain of S. cerevisiae.
[0012] FIG. 3: depicts a graph of ethanol production from molasses in batch using a commercial strain of S. cerevisiae.
[0013] FIG. 4: depicts a graph of specific and volumetric productivities of ethanol from molasses in batch using a commercial strain of S. cerevisiae.
[0014] FIG. 5: depicts a. graph of ethanol production from date extract and molasses (1:1) in batch using a commercial strain of S. cerevisiae.
[0015] FIG. 6: depicts a graph of specific and volumetric productivities of ethanol from date extract and molasses (1:1) in batch using a commercial strain of S. cerevisiae.
[0016] FIG. 7: depicts a graph of ethanol production from date extract in batch using K. marxianus NCYC 179.
[0017] FIG. 8: depicts a graph of specific and volumetric productivities of ethanol from date extract in batch using the yeast K. marxianus NCYC 179.
[0018] FIG. 9: depicts a graph of ethanol production from date extract in batch using the yeast S. cerevisiae NCYC 431.
[0019] FIG. 10: depicts a graph of the specific and volumetric productivities of ethanol from date extract in batch using the yeast S. cerevisiae NCYC 431.
[0020] FIG. 11: depicts a graph of ethanol production from molasses in batch using the yeast S. cerevisiae NCYC 431.
[0021] FIG. 12: depicts a graph of specific and volumetric productivities of ethanol from molasses in batch using the yeast S. cerevisiae NCYC 431.
[0022] FIG. 13: depicts a graph of ethanol production from date extract and molasses (1:1) in batch using the yeast S. cerevisiae NCYC 431.
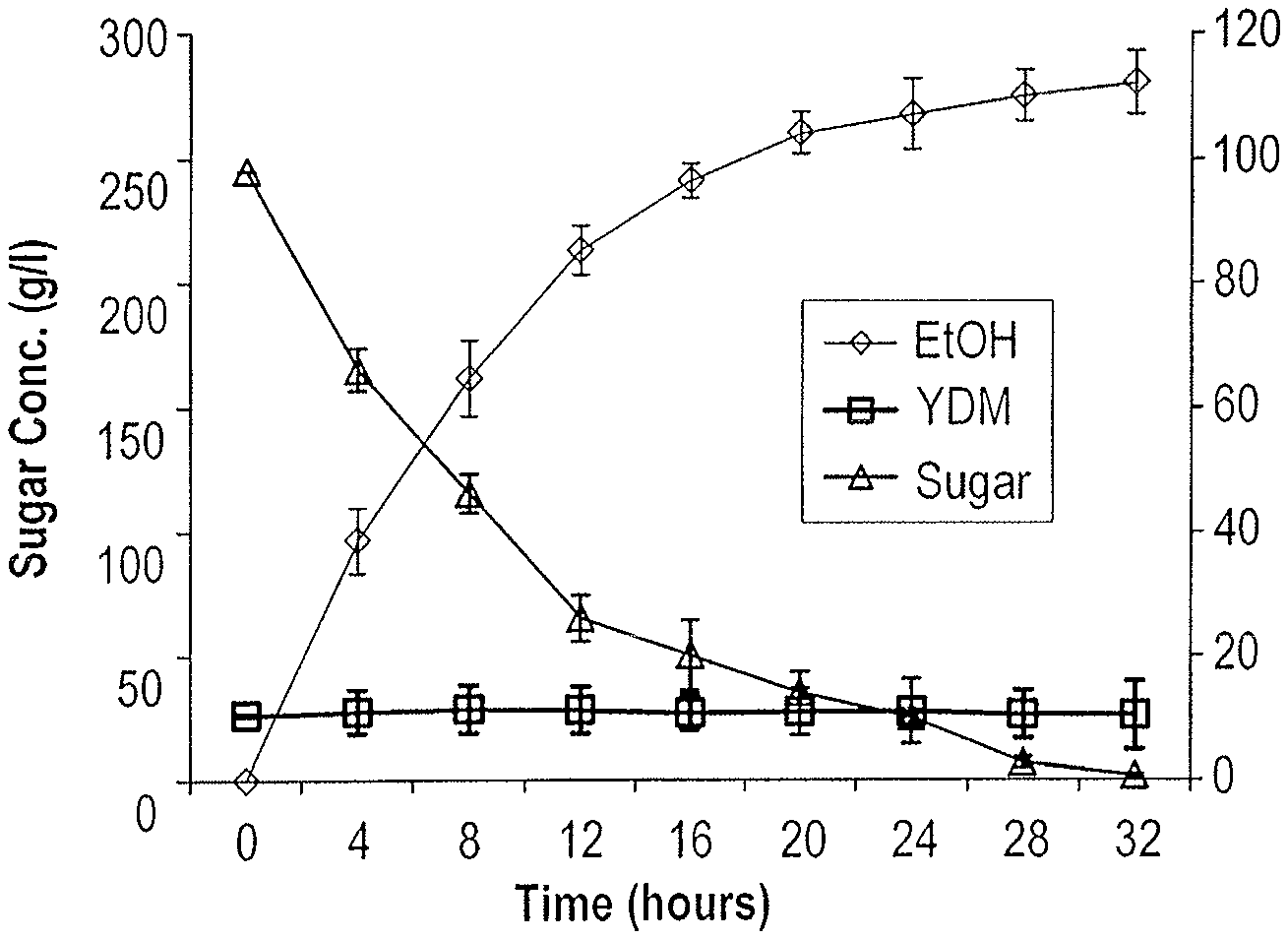
[0023] FIG. 14: depicts a graph of specific and volumetric productivities of ethanol from date extract and molasses (1:1) in batch using the yeast S. cerevisiae NCYC 431.
[0024] FIG. 15: depicts a graph of ethanol production from date extract in fed-batch using the yeast S. cerevisiae NCYC 431.
[0025] FIG. 16: depicts a graph of specific and volumetric productivities of ethanol from date extract in fed-batch using the yeast S. cerevisiae NCYC 431.
[0026] FIG. 17: depicts a graph of ethanol production from molasses in fed-batch using the yeast S. cerevisiae NCYC 431.
[0027] FIG. 18: depicts a graph of specific and volumetric productivities of ethanol from molasses in fed-batch using the yeast S. cerevisiae NCYC 431.
[0028] FIG. 19: depicts a graph of ethanol production from date extract and molasses (1:1) in fed-batch using the yeast S. cerevisiae NCYC 431.
[0029] FIG. 20: depicts a graph of specific and volumetric productivities of ethanol from date extract and molasses (1:1) in fed-batch using the yeast S. cerevisiae NCYC 431.
[0030] FIG. 21: depicts a graph of ethanol production from date extract in fed-batch using the yeast K. marxianus HH 5.
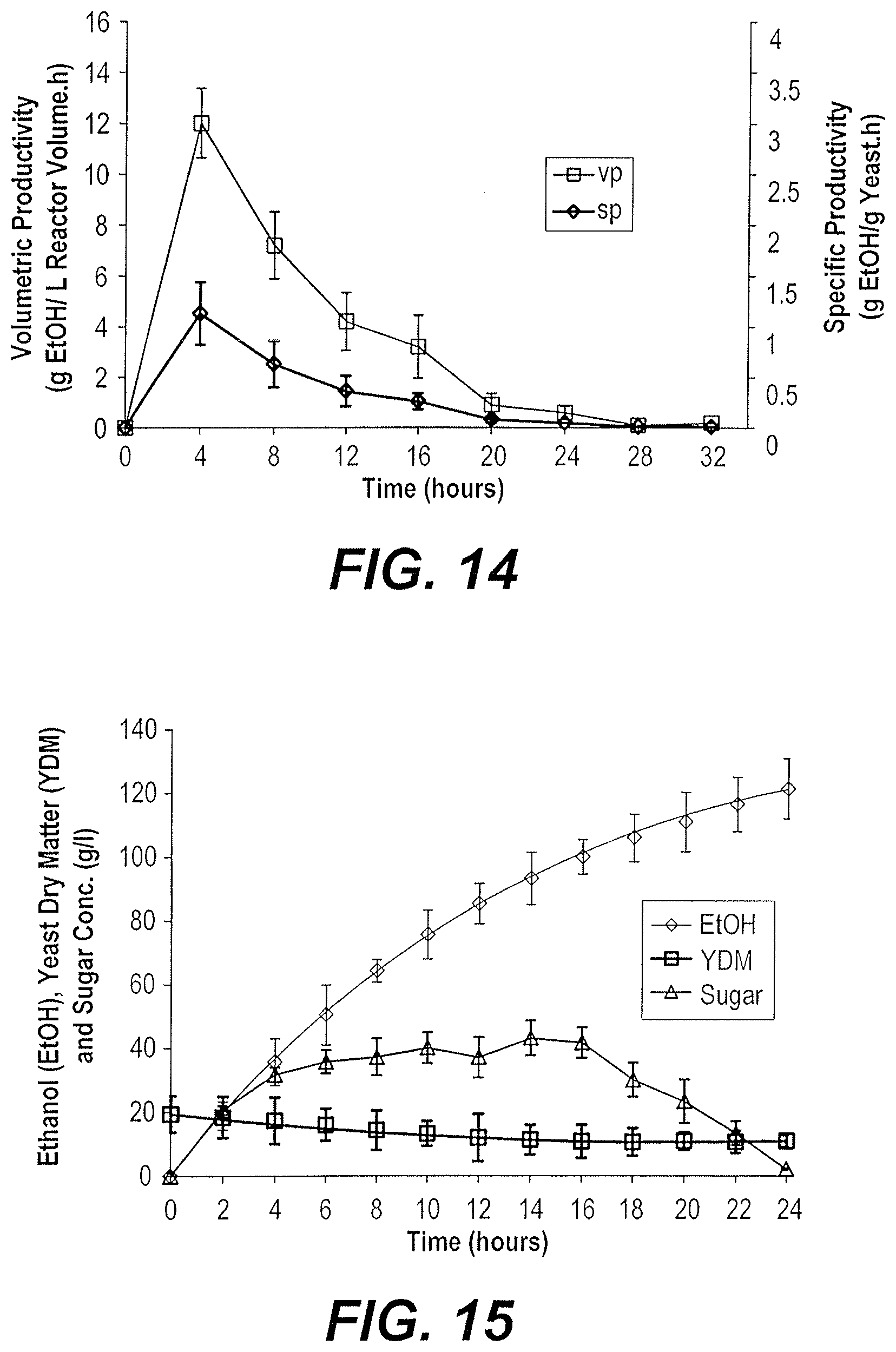
[0031] FIG. 22: depicts a graph of specific and volumetric productivities of ethanol from date extract in fed-batch using the yeast K. marxianus HH 5.
[0032] FIG. 23: depicts a graph of ethanol production from molasses in fed-batch using the yeast K. marxianus HH 5.
[0033] FIG. 24: depicts a graph of specific and volumetric productivities of ethanol from molasses in fed-batch using the yeast K. marxianus HH 5.
[0034] FIG. 25: depicts a graph of ethanol production from date extract and molasses (1:1) in fed-batch using the yeast S K. marxianus HH 5.
[0035] FIG. 26: depicts a graph of specific and volumetric productivities of ethanol from date extract and molasses (1:1) in fed-batch using the yeast K. marxianus HH 5.
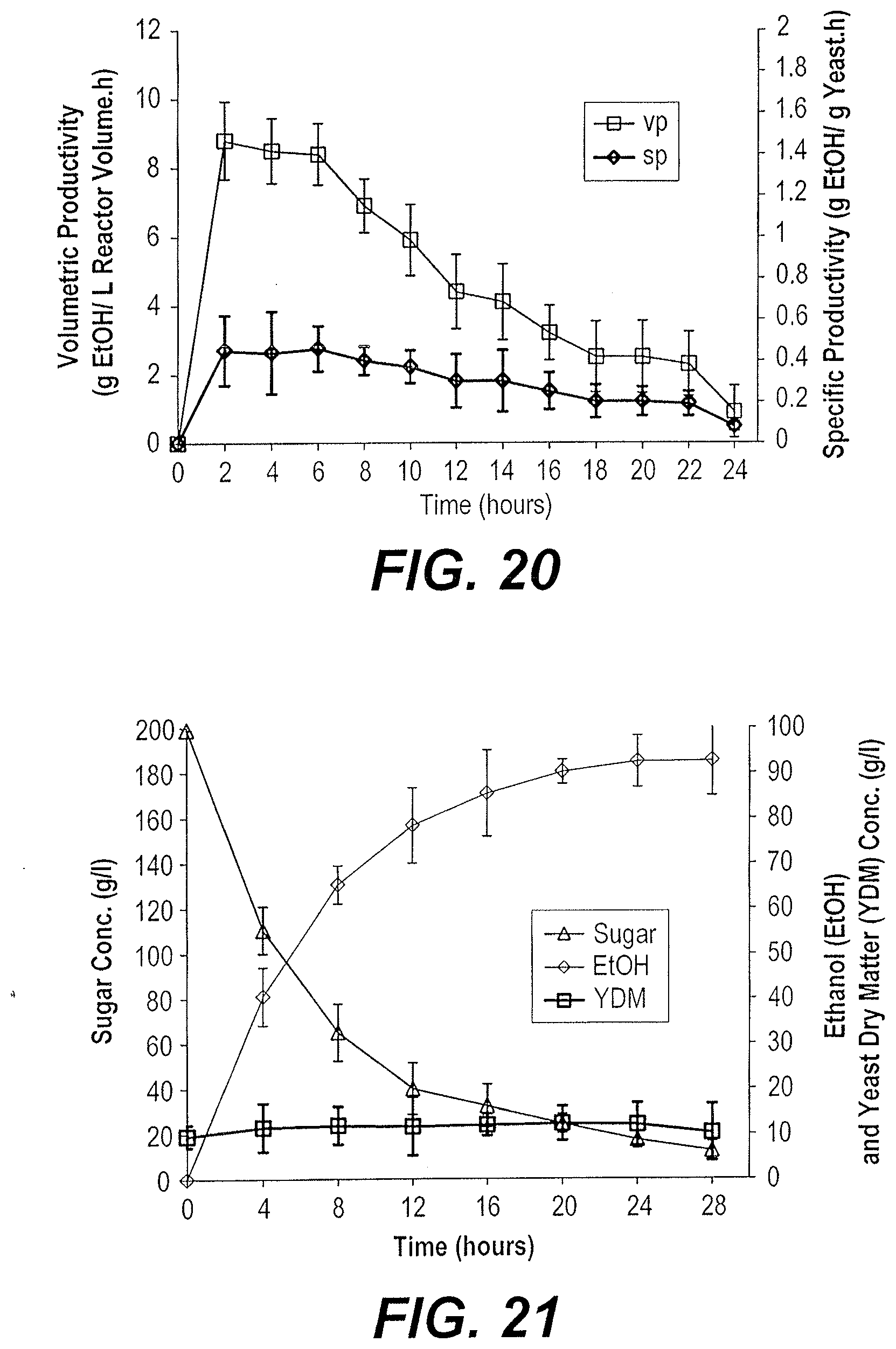
[0036] Similar reference characters denote corresponding features consistently throughout the attached drawings.
DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENTS
[0037] As used herein, the term "about," when used to modify a quantity, means within 10% of the modified quantity.
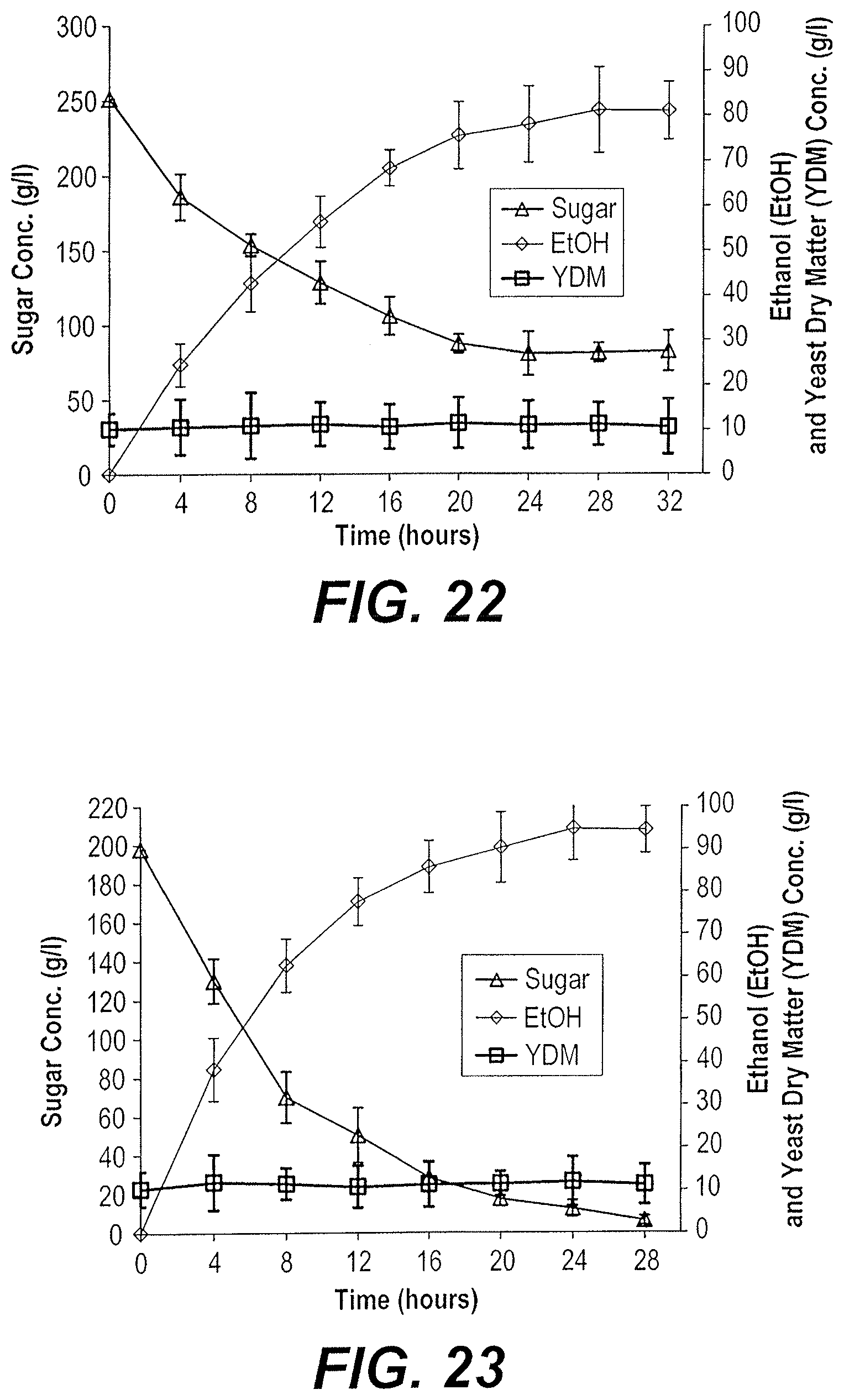
[0038] A method for producing bioethanol from dates includes manufacturing a suitable substrate from dates and fermenting the date substrate to produce bioethanol. The date substrate may be "Dips," a date substrate produced by de-pitting date fruits, heating the flesh with water to produce a mixture, filter pressing the mixture to produce a juice, and concentrating the juice by vacuum drying to produce a date substrate. The date substrate may then be fermented in either a batch or a fed-batch culture. In the batch process, the entire volume of substrate may be added to a bioreactor along with a yeast inoculum and fermentation can be conducted at about 30.degree. C. or about 40.degree. C., depending upon the strain of yeast used as the inoculum. In the fed-batch process, the substrate may be gradually fed into the bioreactor containing the yeast inoculum at a constant feeding rate and fermentation can be conducted at about 30.degree. C. or about 40.degree. C., depending upon the strain of yeast used as the inoculum.
[0039] In an embodiment, the de-pitted date fruits may be heated with an equal volume of water at about 80.degree. C. for about 30 minutes to produce the mixture.
[0040] The filter pressing step is particularly useful to remove large impurities and insoluble matter. In an embodiment, the juice may be further purified to provide a clear liquid by micro filtration using a sheet filter system prior to the concentration step. This embodiment is particularly suited to applications where it is desirable to remove smaller impurities from the juice.
[0041] In an embodiment, the vacuum drying step may involve concentrating the juice or the clear extract to about 75.degree. Brix at a low temperature, such as about 80.degree. C.
[0042] In an alternative embodiment, the date substrate may be date extract. The date extract may be produced by mixing date fruits 1:4 (w/w) with water, boiling the water for about 60-90 minutes, and filtering the mixture to produce a date extract. The date extract may then be sterilized in an autoclave at about 121.degree. C. for about 30 minutes.
[0043] In an embodiment, the date fruits may be waste date fruits, e.g., date fruits not fit for consumption. In a further embodiment the waste date fruits include one or more of the Ruzeiz, Shunaizi, and Shahal varieties of dates. In a further embodiment, the waste date fruits may be waste date fruits harvested in Saudi Arabia. In an embodiment of the present methods, one ton of waste date fruits with an average of 70% sugar content can provide an extract having about 600 kg sugars, which can provide about 300 kg to about 600 kg, e.g., 400 kg, of ethanol.
[0044] In an embodiment, the fermentation may be conducted using a yeast such as S. cerevisiae, K. marxianus, or the like. In a further embodiment, the fermentation may be conducted at about 30.degree. C. using a yeast strain such as S. cerevisiae, K. marxianus, S. cerevisiae NCYC 432, K. marxianus NCYC 179, or the like. In an alternative embodiment, the fermentation may be conducted at about 40.degree. C. using a thermophilic yeast strain. The thermophilic yeast strain may be a thermophilic strain of K. marxianus, e.g., a strain isolated from local habitats in Alahsa, Saudi Arabia. The thermophilic K. marxianus strain may include at least one of the DNA sequences of SEQ ID NOs: 5-16. The thermophilic yeast strain may be K. marxianus HH 5. The K. marxianus HH 5 strain may include the DNA sequences of SEQ ID NO: 5 and/or SEQ ID NO: 6.
[0045] In an embodiment, the fermentation may be conducted using date substrate with a starting sugar concentration ranging from about 20% to about 25%.
[0046] The following examples illustrate the present teachings:
EXAMPLE 1
Chemical Analysis of Dips
[0047] As discussed above, Dips were manufactured by de-pitting date fruits and heating the flesh with an equal amount of water at 80.degree. C. for 30 minutes to produce a mixture; filter pressing the mixture to produce a juice; micro filtering the juice to a clear liquid using a sheet filter system; and concentrating the clear extract to 75 Brix at low temperature (80.degree. C.) by vacuum drying. Chemical analysis of Dips manufactured according to this process demonstrated that the Dips included the following nutrients: about 80% sugars, mainly in form of fructose (41%) and glucose (38%), and a small amount of sucrose (1%); about 2% crude protein; 1.1 g/kg phosphorus; 14.9 g/kg potassium; 0.8 g/kg magnesium; 240 ppm pantothenic acid; and 2.7 ppm biotin. (See Table 1) Therefore, Dips used as carbon and energy source in alcoholic fermentation would be expected to yield about 400 kg ethanol (about 500 liters).
EXAMPLE 2
Chemical Analysis of Date Extracts
[0048] Waste date fruits were obtained from a date processing factory in Al Hofuf City, Saudi Arabia. They were mainly a collection from the varieties Ruzeiz, Shunaizi, and Shahal. Extraction of syrup from the fruits was performed as follows: I :4 (w/w) fruit: water, boiling for about 60-90 minutes and filtration. The resulting substrate was sterilized in 5-liter bottles by autoclaving at 121.degree. C. for 30 minutes. The sterilized substrate contained about 30% sugar.
[0049] The chemical composition of the date sterilized substrate was assessed, including determining protein concentration using the Kjeldhal method, M-Inositol concentration using and biotin and pantothenic acid concentration according to the vitaFast vitamin testing method, using an ELISA Reader (Multiskan EX Model No. 355, Thermolabsystem-Finlan). M-Inositol was determined with High Performance Liquid Chromatography (HPLC) using a Shimadzu Japan Model 2003, equipped with a RID 10A refractive index detector, CLC NH2 6.times.150 column, LC 10ATP pump, and CTO 10AC VP oven. The mobile phase was 83% CH3CN:17% water (v/v), flow rate 1 ml/min., column pressure 200 KgF/cm.sup.2. Ethanol and sugar (glucose, fructose, and sucrose) concentrations were determined using the enzymatic UV method (Boehringer Mannheim). Mineral concentrations (Mg, P, K, and S) were determined using the atomic absorption method. The average chemical composition of the date sterilized substrate is shown in Table 1.
TABLE-US-00001 TABLE 1 Date Extract Nutrient Analysis Chemicals Total sugars (% w/w) Content Fruit 68-72 Thin Extract 25-30 Extracted Fruit 10.2-10.8 pH 4.3-4.5 Crude protein (% w/w) 2.0-2.5 Magnesium (g/kg) 0.5-0.9 Potassium (g/kg) 14.0-16.0 Phosphorous (g/kg) PO.sub.4 or P 1.0-1.4 Pantothenic acid (ppm) 200-240 Biotin (ppm) 2.0-3.0 m-Inositol (ppm) traces
[0050] The date substrate contained all nutrients needed for a small amount of yeast growth (See Table 1) and no addition of mineral medium was needed. The fruits contained 68-72% (w/w) sugars on wet basis and the thin extract contained 25-30% (w/w) sugars on wet basis, with 10.2-10.8% (w/w) sugar content remaining in the extracted fruit. Hence, the efficiency of our extraction system was about 85%. As a result, a ton of waste date fruits with an average of 70% sugar content can be expected to produce a date substrate containing about 600 kg sugars, which in turn would be expected to produce about 300 kg bioethanol. If the efficiency of extraction is raised, such as by using high quality equipment, the yield of bioethanol would be predicted to increase to more than 315 kg from a ton of waste date fruits. If the sugar content of the fruits is increased, this would also be predicted to increase the resulting bioethanol yield.
EXAMPLE 3
Identification of Thermotolerant Yeasts
[0051] A screening program was carried out to search for thermotolerant yeasts from the local habitat in Saudi Arabia that can be used for ethanol production. Samples of microbially spoiled fruits, especially date fruits, were collected from Hofuf markets and farms. Yeasts involved in spoilage were isolated by inoculation on PDA dishes and incubation at 35-50.degree. C. for 2-3 days. Colonies of different forms were picked, purified by successive streaking on PDA dishes, and the pure cultures were kept in the refrigerator in PDA tubes for identification.
[0052] The strains were cultivated on Malt Extract Agar for 3 days in the dark at 25.degree. C. DNA was isolated using the MoBio-UltraClean.TM. Microbial DNA Isolation Kit according to the manufacturer's instructions. Fragments including the 26S ribosomal RNA gene, Large Subunit D1 and D2 region (LSU) were amplified using the primers LROR: ACCCGCTGAACTTAAGC (SEQ ID NO: 1) and LR5: TCCTGAGGGAAACTTCG (SEQ ID NO: 2). Fragments including the Internal Transcribed Spacer 1 and 2 and the 5.8S gene (ITS) were amplified using the primers LS266: GCATTCCCAAACAACTCGACTC (SEQ ID NO: 3) and V9G TTACGTCCCTGCCCTTTGTA (SEQ ID NO: 4). The PCR fragments were sequenced with the ABI Prism.RTM. Big Dye.TM. Terminator v. 3.0 Ready Reaction Cycle sequencing Kit. Samples were analyzed on an ABI PRISM 3730 Genetic Analyzer and contigs were assembled using the forward and reverse sequences with the SeqMan program from the LaserGene package. The sequence was compared in a large yeast database of CBS-KNAW Fungal Biodiversity Centre with sequences of known strains.
[0053] Twenty two isolates that showed good growth at 45.degree. C. were obtained from the screening program. Twelve of these isolates grew at 50.degree. C., and hence were regarded as thermophilic. Preliminary identification tests were carried out on the 12 thermophilic isolates, including morphological examination of the yeast colonies grown on PDA, microscopic examination of the cell form, and ability to ferment glucose. The outcome of these tests indicated that the 12 thermophilic isolates belonged to at least to 3 different genera. Molecular identification of these 12 thermophilic isolates by DNA sequencing was carried out and the results are shown in Table 2. The isolates belonged to three genera and species, namely Kluyveromyces marxianus (6 isolates), Clavispora lusitaniae (5 isolates and Wickerhamomyces anomalus (1 isolate). C. lusitaniae is an opportunistic pathogen, hence it was not considered for ethanol production. Preliminary tests showed that W. anomalus was not a good ethanol producer. However, promising preliminary results for ethanol production were obtained for the strains of K. marxianus, and the best of these isolates was K. marxianus 05-1 or "HH5."
[0054] The isolates belonging to the same species had different genetic sequences at the tested loci, indicating that they are different strains of the species. The sequence of the 26S ribosomal RNA gene, Large Subunit D1 and D2 region (LSU) of the K. marxianus 05-1 isolate includes SEQ ID. NO. 6. The sequence of the 26S ribosomal RNA gene, Large Subunit D1 and D2 region (LSU) of the K. marxianus 12-1 isolate includes SEQ ID. NO. 8. The sequence of the 26S ribosomal RNA gene, Large Subunit D1 and D2 region (LSU) of the K. marxianus 14G-1 isolate includes SEQ ID. NO. 10. The sequence of the 26S ribosomal RNA gene, Large Subunit D1 and D2 region (LSU) of the K. marxianus 14W-1 isolate includes SEQ ID. NO. 12. The sequence of the 26S ribosomal RNA gene, Large Subunit D1 and D2 region (LSU) of the K. marxianus 23-1 isolate includes SEQ ID. NO. 14. The sequence of the 26S ribosomal RNA gene, Large Subunit D1 and D2 region (LSU) of the K. marxianus 25-1 isolate includes SEQ ID. NO. 16. The sequence of the Internal Transcribed Spacer 1 and 2 and the 5.8S gene (ITS) of the K. marxianus 05-1 isolate includes SEQ ID. NO. 5. The sequence of the Internal Transcribed Spacer 1 and 2 and the 5.8S gene (ITS) of the K. marxianus 12-1 isolate includes SEQ ID. NO. 7. The sequence of the Internal Transcribed Spacer 1 and 2 and the 5.8S gene (ITS) of the K. marxianus 14G-1 isolate includes SEQ ID. NO. 9. The sequence of the Internal Transcribed Spacer 1 and 2 and the 5.8S gene (ITS) of the K. marxianus 14W-1 isolate includes SEQ ID. NO. 11. The sequence of the Internal Transcribed Spacer 1 and 2 and the 5.8S gene (ITS) of the K. marxianus 23-1 isolate includes SEQ ID. NO. 13. The sequence of the Internal Transcribed Spacer 1 and 2 and the 5.8S gene (ITS) of the K. marxianus 25-1 isolate includes SEQ ID. NO. 15.
TABLE-US-00002 TABLE 2 Identification of Yeast Isolates Using Morphological and Molecular Characteristics Strain Name 05-1 Kluyveromyces marxianus (E. C. Hansen) Van der Walt (named K. marxianus HH5) 12-1 Kluyveromyces marxianus (E. C. Hansen) Van der Walt 14G-1.sub. Kluyveromyces marxianus (E. C. Hansen) Van der Walt 14W-1 Kluyveromyces marxianus (E. C. Hansen) Van der Walt 23-1 Kluyveromyces marxianus (E. C. Hansen) Van der Walt 25-1 Kluyveromyces marxianus (E. C. Hansen) Van der Walt 15-2 Clavispora lusitaniae Rodr. Mir. 26-2 Clavispora lusitaniae Rodr. Mir. 28-2 Clavispora lusitaniae Rodr. Mir. 45-2 Clavispora lusitaniae Rodr. Mir. 50-2 Clavispora lusitaniae Rodr. Mir. 48-2 Wickerhamomyces anomalus (E. C. Hansen) Kurtzman, Robnett & Basehoar-Powers
EXAMPLE 4
Fermentation Experimental Design
[0055] The yeast strains were first propagated in shake flasks and then in a bioreactor containing suitable medium, harvested by filtration to a cake and kept in the refrigerator to be used as inocula.
[0056] Fermentation experiments were done in batch and fed-batch cultures using shake flasks, a BioFlo 110 bioreactor (New Brunswick Scientific) with 10 liter working volume and an Applikon autoclavable Bioreactor system (Applikon Biotechnology, the Netherlands) with one liter working volume. In the batch process, the whole substrate was added to the bioreactor at the beginning of fermentation and the process continued to its end. In the fed-batch process the substrate was fed to the fermenter at constant feeding rates. Fermentations using the commercial baker's yeast, S. cerevisiae NCYC 432, and K. marxianus NCYC 179 were at 30.degree. C., while fermentation using the thermophilic yeast K. marxianus HH 5 (isolated by the present inventors) was at 40.degree. C. Samples were taken from the bioreactor every 2-4 hours and the concentrations of ethanol, sugars and yeast dry matter were determined.
[0057] A 3 X 4 X 2 (three variables) factorial experimental design was employed with three types of substrates (date extract, molasses, and a mixture of 1:1 date extract/molasses), 4 production organisms (local and imported) and two fermentation processes (batch and fed-batch) (See Table 3). A randomized block design was chosen to run the experiment. A collection of waste date fruits and 2 fermentation temperatures (30 and 40.degree. C.) were chosen based on information from the literature. All of the above parameters were used as useful means for modeling and optimizing small-scale operations. Variables measured in this study included yield of ethanol on substrate (g ethanol/g sugar), and the concentrations (g/l) of ethanol, sugars, and yeast biomass.
TABLE-US-00003 TABLE 3 Experimental Design Independent Variable Factor Substrate 100% Dips 100% 1:1 Dips/ Molasses Molasses Production Organism Local isolate Imported strains (one strain) (three strains) Production process Batch Fed-Batch
[0058] Ethanol and sugar (glucose, fructose, and sucrose) concentrations were determined using the enzymatic UV method (Boehringer Mannheim). Yeast biomass concentration was determined by centrifugation of 5 ml broth at 2000 rpm for 5 minutes in dry centrifugal tube, and overnight drying at 105.degree. C.
[0059] The yeasts used were S. cerevisiae NCYC 431, Kluyveromyces marxianus NCYC 179, a commercial baker's yeast strain of the species S. cerevisiae purchased from the local market, and K. marxianus HH 5 (isolated by the present inventors). Batch and fed-batch fermentation processes were used at different final sugar concentrations. For comparison, substrates of pure molasses and a 1:1 mixture of date extract and molasses were also used for production. The fermentation experiments were done using shake flasks, a Biollo 110 bioreactor (New Brunswick Scientific) with 10 liter working volume and an Applikon autoclavable Bioreactor system (Applikon Biotechnology, the Netherlands) with one liter working volume.
[0060] Date substrate was extracted as described in Example 1. The fruits used were waste fruits collected from a date processing factory in Hofuf City, mainly made of the varieties Ruzeiz, Shunaizi, and Shahal. Because yeast growth during ethanol production is normally minimal, sugars in the production substrate are mainly converted to ethanol and no significant amount of yeast biomass is formed.
[0061] Four yeast strains were used for production including a commercial baker's yeast strain of the species S. cerevisiae, K. marxianus NCYC 179, S. cerevisiae NCYC 431 and the strain K. marxianus HH 5 (isolated by the present inventors). Production was in batch and fed-batch cultures at fermentation temperatures of 30 and 40.degree. C. Three substrates were used, namely pure date extract, pure molasses and a mixture of date extract and molasses at 1:1 ratio, with sugar concentrations of 20 to 25%. The results, all of which were averages of three runs, are presented in FIGS. 1 to 26.
EXAMPLE 5
Fermentation Testing with Commercial S. Cerevisiae
[0062] The strain of the baker's yeast from the species S. cerevisiae was used for ethanol production in batch processes at fermentation temperatures of 30.degree. C. on substrates of pure date extract, pure molasses and a 1:1 mixture of date extract and molasses. Samples were taken every 4 hours and the concentrations of ethanol, sugar and yeast dry matter were measured. The results are shown in FIGS. 1-6.
[0063] Ethanol production from date extract continued at a relatively high rate during the first 12 hours of fermentation, the concentration increased steadily during this period to reach 85.2 g/l (FIG. 1). After that, the production rate decreased towards the end of the fermentation period of 32 hours and the final ethanol concentration reached was 112.1 g/l (11.2% w/v). The course of ethanol productivity is illustrated in FIG. 2. The specific and volumetric productivities were relatively high during the first 12 hours of fermentation, after which they decreased steadily to reach very low values towards the end of the fermentation period. During the first 4 hours of fermentation the specific productivity was 0.92 g ethanol per g yeast per hour (g/g. h) and the volumetric productivity was 9.7 g ethanol per liter reactor volume per hour (g/l. h). At the end of fermentation after 32 hours, the specific productivity decreased to 0.05 g/g. h and the volumetric productivity decreased to 0.5 g/l. h. It is apparent that increased ethanol concentration in the fermentation medium was inhibitory to production, and the maximum amount produced and tolerated by the yeast was 11.2% (w/v). The specific productivity is mainly a measure of the efficiency of the yeast in ethanol production and the volumetric productivity is mainly a measure of the efficiency of the bioreactor system. The overall yield of ethanol on sugar was 91.5% of the theoretical. There was no significant yeast growth during fermentation, as yeast biomass concentration remained almost constant during the whole period of fermentation (FIG. 1). The sugar was almost completely consumed, with only traces remaining at the end of the fermentation period (FIG. 1). Generally, the values obtained for the yield of ethanol on sugar and for the specific and volumetric productivities are comparable to values reported for different organisms and substrates.
[0064] Substrates of pure molasses and a 1:1 mixture of date extract and molasses were used for ethanol production under conditions similar to those of the pure date extract. The results of molasses fermentation are presented in FIGS. 3 and 4. These results were quite similar to those of pure date extract (FIG. 3). Most production occurred in the first 12 hours of fermentation, after which production rate started to slow down and the final ethanol concentration reached was 108.4 g/l (10.8% w/v). The specific and volumetric productivities were also comparable to those of the pure date extract (FIG. 4). The specific productivity after 4 hours of fermentation was 1.05 g/g. h and the volumetric was 10.0 g/l. h, and both decreased to 0.06 g/g. h and 0.6 g/l. h at the end of fermentation, respectively. The yield of ethanol on sugar was 90.3% of the theoretical, which was a little lower than that of pure date extract, but statistical analysis showed that the difference was not significant. No significant yeast growth occurred and the sugar provided was almost completely utilized with only traces remaining at the end of the fermentation period (FIG. 3). A similar result was obtained using the 1:1 date extract and molasses mixture (FIGS. 5 and 6). The final ethanol concentration was 114.0 g/l (11.4% w/v), the specific and volumetric productivities 0.94 g/g. h and 9.2 g/l. h after 4 hours and 0.05 g/g. h and 0.5 g/l. h at the end of fermentation, respectively. The yield of ethanol on sugar was 90.1% of the theoretical and was not significantly different from the yields obtained for the other two substrates as statistical analysis showed. Again, no significant yeast growth occurred and the sugar was largely consumed with very small amount remaining at the end of fermentation (FIG. 5). It can therefore be said that this commercial yeast strain of the species S. cerevisiae can produce ethanol from date extract in efficiency comparable to that of the reference substrate molasses. That is to say that date extract substrate is quite suitable for ethanol production.
EXAMPLE 6
Fermentation Testing with K. Marxianus NCYC 179
[0065] The strain Kluyveromyces marxianus NCYC 179 was used for ethanol production in batch processes at fermentation temperatures of 30.degree. C. on a substrate of pure date extract. Samples were taken every 4 hours and the concentrations of ethanol, sugar and yeast dry matter were measured. The results are shown in FIGS. 7 and 8.
[0066] The performance of this yeast was generally weak. The concentration of ethanol after the first 4 hours of fermentation was only 25.6 g/l, which was much lower than the concentration reached from the commercial baker's yeast reported above (FIG. 7). The maximum ethanol concentration was 71 g/l (7.1% w/v); it was reached after 28 hours of fermentation. The specific and volumetric productivities were also much lower than the values reported above for the commercial baker's yeast. They were 0.62 g/g. h for the specific productivity and 6.3 for the volumetric productivity after 4 hours of fermentation and 0.05 g/g. h and zero after 28 hours, respectively (FIG. 8). No increase in ethanol concentration occurred when fermentation was continued to 36 hours. The yield was only 54.9% of the theoretical, which was much lower than that obtained from the commercial yeast. The sugar was not fully utilized, and 95.6 g/l remained unconsumed in the fermentation broth. This suggests that this yeast cannot produce and tolerate more than 7% w/v ethanol. For this reason, further experiments were not performed with this yeast strain.
EXAMPLE 7
Fermentation Testing with S. Cerevisiae NCYC 431
[0067] The yeast strain S. cerevisiae NCYC 431 was used for ethanol production in hatch processes at fermentation temperatures of 30.degree. C. on substrates of pure date extract, pure molasses and a 1:1 mixture of date extract and molasses. Samples were taken every 4 hours and the concentrations of ethanol, sugar and yeast dry matter were determined. The results are shown in FIGS. 9-14.
[0068] Ethanol production from date extract substrate started at a relatively high rate compared to the other yeast strains discussed above. The concentration reached 45 g/l in the first 4 hours and continued to increase at a decreasing rate till it reached 117.6 g/l (11.8% w/v) at the end of fermentation of 32 hours (FIG. 9). The specific productivity started at 1.05 g/g. h in the first 4 hours and ended at 0.03 g/g. h at the end of fermentation, while the volumetric productivity was 11.3 and 0.4 g/l. h after 4 and 32 hours of fermentation, respectively (FIG. 10). The yield of ethanol on sugar was 94.6% of the theoretical, indicating that sugar was mainly converted into ethanol and no significant amounts of other metabolic products were formed. Sugar provided was consumed to traces remaining and no significant yeast growth occurred.
[0069] Ethanol production from molasses substrate using the yeast strain S. cerevisiae NCYC 431 is presented in FIGS. 11 and 12. Ethanol concentration after 4 hours was 50.5 g/l and reached 118.2 g/l (11.8% w/v) at the end of the fermentation period of 32 hours (FIG. 11). The specific and volumetric productivities were high, starting at 1.15 g/g. h and 12.6 g/l. h in the first 4 hours, respectively and ending at 0.05 g/g. h and 0.6 WE h after 32 hours, respectively (FIG. 12). The yield of ethanol on sugar was also high, reaching a value of 96.6% of the theoretical, but statistical analysis showed that this result was not significantly different from the 94.6% obtained using the date extract substrate. As expected, sugar provided was almost completely consumed and no significant yeast growth occurred.
[0070] Ethanol production from the mixed substrate using strain S. cerevisiae NCYC 431 (1:1 date extract and molasses) followed a quite similar trend to the other substrates presented above (FIGS. 13 and 14). Ethanol concentration in the first 4 hours was 47.9 g/l and reached 113.8 g/l (11.4% w/v) after 32 hours of fermentation (FIG. 13). The specific and volumetric productivities were 1.13 g/g. h and 12 g/l. h after 4 hours, respectively and ended at 0.01 g/g. h and 0.2 g/l. h after 32 hours, respectively (FIG. 14). The yield of ethanol on sugar was 95.4% of the theoretical, it was the second highest, but was not significantly different from the yields of pure date extract and pure molasses.
[0071] Statistical analysis comparing the yields of ethanol on sugar obtained from the baker's yeast strain S. cerevisiae and from the yeast strain S. cerevisiae NCYC 431 showed that the yields of the latter were significantly higher. It can therefore be concluded that the yeast strain S. cerevisiae NCYC 431 was the best producer of ethanol from date extract substrate. Production by this yeast stain was therefore further tested in fed-batch experiments.
[0072] The strain S. cerevisiae NCYC 431 was used for ethanol production in a fed-batch process of fermentation to attempt to further improve its productivity. Production was at a temperature of 30.degree. C. on substrates of pure date extract, pure molasses and a 1:1 mixture of date extract and molasses. The substrates were fed at constant rates of 200 ml/h and feeding time was 15 hours to deliver 3 liters of substrate containing about 50% sugars. Samples were taken every 2 hours and the concentrations of ethanol, sugar and yeast dry matter were measured. The results are shown in FIGS. 15-20.
[0073] The results of the experiments using date extract as substrate are shown in FIGS. 15 and 16. Ethanol concentration reached 18.8 g/l in the first 2 hours of fermentation (FIG. 15). The concentration increased steadily and reached its maximum of 121.5 g/l (12.2% w/v) in 24 hours. The total amount of ethanol produced was not much higher than that obtained in the batch culture discussed above. The main difference was in the fermentation time, which was 24 hours in the fed-batch process compared to 32 hours in the batch process. The specific productivity decreased much slower compared to that of the batch process (FIG. 16). In the batch process the yeast was affected by the combined inhibitory effect of high concentrations of sugar and ethanol, whereas in the fed-batch process there was no inhibitory effect of sugar because its concentration remained relatively low throughout the fermentation time. Sugar concentration in the fermentation medium increased at a low rate till the end of substrate feeding after 15 hours, then decreased quickly to reach traces at the end of fermentation. No significant yeast growth occurred. The volumetric productivity in turn remained also relatively high during the whole time of fermentation indicating the absence of the combined inhibitory effect of sugar and ethanol (FIG. 16). Ethanol yield on sugar was 97.2% of the theoretical, indicating a high efficiency in converting sugar into ethanol without formation of significant amounts of other metabolic products.
[0074] Molasses substrate gave quite similar results to date extract substrate. As can be seen in FIGS. 17 and 18, production proceeded in the same way as those of the date extract substrate. The concentration of ethanol reached 20.3 g/l in the first two hours of fermentation and continued to increase steadily to reach its Maximum of 119.9 g/l (12% w/v) after 24 hours of fermentation (FIG. 17). The specific productivity remained fairly high over the whole period of fermentation (FIG. 18). Sugar concentration increased slowly to reach 46.3 g/l towards the end of substrate feeding, and decreased quickly after that to reach very low levels (FIG. 18). No significant amount of yeast growth was noticed. The yield of ethanol on sugar was 96.4% of the theoretical, which was a little lower than that obtained from the date extract but the difference was not significant as statistical analysis revealed.
[0075] The substrate obtained from mixing date extract with molasses at a 1:1 ratio was utilized by the yeast in a similar efficiency as the former substrates discussed above (FIGS. 19 and 20). Ethanol concentration was 17.6 g/l after 2 hours of fermentation and reached 116.3 g/l (11.6% w/v) at the end of fermentation after 24 hours (FIG. 19). The specific productivity remained relatively high during the whole fermentation period at values of 0.45 and 0.2 g/g. h (FIG. 20). Sugar concentration increased slowly during substrate feeding, and then decreased quickly to reach traces at the end of fermentation (FIG. 19). No significant yeast growth occurred. The yield of ethanol on sugar was 95.1% of theoretical and was not significantly different from the yields of the other two substrates discussed above.
[0076] Date extract gave ethanol yields comparable to those obtained from the reference substrate molasses.
EXAMPLE 8
Fermentation Testing with K. Marxianus HH5
[0077] The strain Kluyveromyces marxianus HH5, isolated by the research team from local habitats in Saudi Arabia, was used for ethanol production in batch processes at fermentation temperatures of 40.degree. C. on substrates of pure date extract, pure molasses and a 1:1 mixture of date extract and molasses. The results are shown in FIGS. 21 to 26.
[0078] Production in a substrate of date extract containing about 20% sugar started at a relatively high rate. Ethanol concentration reached 40.5 g/l in the first 4 hours, which is comparable to values obtained from the other yeasts described above (FIG. 21). Production continued at a reduced rate, and the maximum ethanol concentration was 92.7 g/l, reached in 24 hours of fermentation, which then remained almost constant till the end of fermentation after 28 hours. About 13 g/l sugar remained unconsumed in the fermentation broth and no significant increase in yeast concentration was observed. The yield of ethanol on sugar was 92.7% of the theoretical, which was quite good for a wild yeast strain. The yeast seemed to have converted most consumed sugars to ethanol without significant amounts of other metabolic products.
[0079] When a substrate of date extract containing about 25% sugar concentration was used, the rate of production decreased drastically. Only 24.5 g/l ethanol concentration was reached in the first 4 hours of fermentation compared to 40.5 g/l when 20% sugar concentration was used (FIG. 22). Production continued after that at a low rate and the highest ethanol concentration was 81.2 g/l, reached after 28 hours of fermentation, and no further increase in ethanol concentration was observed when fermentation time was extended to 32 hours. This suggests that this yeast is sensitive to high sugar concentration. The yield of ethanol on sugar was 64.7% of the theoretical, which was very low compared to the yield of 92.7% obtained when a 20% sugar concentration was used. Therefore, it can be concluded that this yeast can produce and tolerate up to about 9% w/v or 11% v/v ethanol concentration. This high productivity is particularly of interest given that it can be achieved at a fermentation temperature of 40.degree. C. The combination of high productivity and higher fermentation temperature suggests that this method could reduce the costs of cooling during fermentation and heating during distillation.
[0080] The yeast used molasses as substrate for ethanol production in a quite similar way as its use of date extract, confirming suitability of date extract as substrate for ethanol production. As can be seen in FIG. 23, ethanol concentration was 38.4 g/l after 4 hours of fermentation and reached 94.8 g/l in 24 hours then remained almost constant till 28 hours of fermentation. This result confirms that the yeast K. marxianus HH 5 can produce and tolerate ethanol concentrations in range of about 9-10% w/v (11-12% v/v). About 6 g/l sugar remained unconsumed and no significant yeast growth occurred. The yield of ethanol on sugar was 95.8% of the theoretical, which was higher than that obtained from date extract but the difference was not significant as statistical analysis showed. It is apparent that most of the sugar consumed was used for ethanol production and no significant amounts of other metabolic products were formed. When sugar concentration in the molasses substrate was raised to 25%, the performance of the yeast became very weak. The highest ethanol concentration reached was about 7.1% w/v (about 9% v/v) in 28 hours of fermentation, which was the lowest concentration obtained (FIG. 24). The yield of ethanol on sugar was only 56.9% of the theoretical, and about 98 g/l sugar remained unconsumed. This is a very low yield compared to the 95.8% obtained from molasses substrate containing 20% sugar, and confirms the result achieved when date extract substrate containing 25% sugar was used. This indicates again that the yeast K. marxianus HH 5 is sensitive to high sugar and ethanol concentrations.
[0081] Substrates of mixed date extract and molasses that contained 20% sugar gave a little lower substrate containing 20% sugar, and confirmed the result achieved when date extract substrate containing 25% sugar was used. This indicates again that the yeast K. marxianus HH 5 is sensitive to high sugar and ethanol concentrations.
[0082] Substrates of mixed date extract and molasses that contained 20% sugar gave a little lower ethanol yields compared to substrates of pure date extract and pure molasses containing the same sugar concentrations. The maximum ethanol concentration reached was 8.7% w/v (11% v/v) in 28 hours of fermentation and the yield of ethanol on sugar was 85.7% of the theoretical (FIG. 25). No considerable yeast growth occurred and about 17 g/l sugar remained in the fermentation broth unconsumed. In substrates containing sugar concentrations of about 25%, the yield of ethanol on sugar dropped to 63.3% of the theoretical compared to the 85.7% obtained from the substrates containing 20% sugar. The maximum ethanol concentration reached was 7.9 w/v (10% v/v) in 28 hours of fermentation (FIG. 26) with no significant yeast growth and about 85 g/l sugar remaining unconsumed.
[0083] It is to be understood that the method for producing bioethanol from dates is not limited to the specific embodiments described above, but encompasses any and all embodiments within the scope of the generic language of the following claims enabled by the embodiments described herein, or otherwise shown in the drawings or described above in terms sufficient to enable one of ordinary skill in the art to make and use the claimed subject matter.
Sequence CWU 1
1
16117DNAArtificial Sequenceprimer 1acccgctgaa cttaagc
17217DNAArtificial SequencePrimer 2tcctgaggga aacttcg
17322DNAArtificial Sequenceprimer 3gcattcccaa acaactcgac tc
22420DNAArtificial Sequenceprimer 4ttacgtccct gccctttgta
2051000DNAKluyveromyces marxianus 5ttagtgaggc ctcaggattt gcttagagaa
gggggcaact ccatctcaga gcgaaaaatc 60tggtcaaact tggtcattta gaggaactaa
aagtcgtaac aaggtttccg taggtgaacc 120tgcggaagga tcattaaaga
ttatgaatga atagattact gggggaatcg tctgaacaag 180gcctgcgctt
aattgcgcgg ccagttcttg attctctgct atcagttttc tatttctcat
240cctaaacaca atggagtttt ttctctatga actacttccc tggagagctc
gtctctccag 300tggacataaa cacaaacaat attttgtatt atgaaaaact
attatactat aaaatttaat 360attcaaaact ttcaacaacg gatctcttgg
ttctcgcatc gatgaagaac gcagcgaatt 420gcgatatgta ttgtgaattg
cagattttcg tgaatcatca aatctttgaa cgcacattgc 480gccctctggt
attccagggg gcatgcctgt ttgagcgtca tttctctctc aaacctttgg
540gtttggtagt gagtgatact cgtctcgggt taacttgaaa gtggctagcc
gttgccatct 600gcgtgagcag ggctgcgtgt caagtctatg gactcgactc
ttgcacatct acgtcttagg 660tttgcgccaa ttcgtggtaa gcttgggtca
tagagactca taggtgttat aaagactcgc 720tggtgtttgt ctccttgagg
catacggctt taaccaaaac tctcaaagtt tgacctcaaa 780tcaggtagga
gtacccgctg aacttaagca tatcaataag cggaggaaaa gaaaccaacc
840gggattgcct tagtaacggc gagtgaagcg gcaaaagctc aaatttgaaa
tctggcgtct 900tcgacgtccg agttgtaatt tgaagaaggc gactttgtag
ctggtccttg tctatgttcc 960ttggaacagg acgtcataga gggtgagaat
cccgtgtggc 10006800DNAKluyveromyces marxianus 6ttagtacggc
gaagtgaagc ggcaaaagct caaatttgaa atctggcgtc ttcgacgtcc 60gagttgtaat
ttgaagaagg cgactttgta gctggtcctt gtctatgttc cttggaacag
120gacgtcatag agggtgagaa tcccgtgtgg cgaggatccc agttatttgt
aaagtgcttt 180cgacgagtcg agttgtttgg gaatgcagct ctaagtgggt
ggtaaattcc atctaaagct 240aaatattggc gagagaccga tagcgaacaa
gtacagtgat ggaaagatga aaagaacttt 300gaaaagagag tgaaaaagta
cgtgaaattg ttgaaaggga agggcatttg atcagacatg 360gcgtttgctt
cggctttcgc tgggccagca tcagttttag cggttggata aatcctcggg
420aatgtggctc tgcttcggta gagtgttata gcccgtggga atacagccag
ctgggactga 480ggattgcgac ttttgtcaag gatgctggcg taatggttaa
atgccgcccg tcttgaaaca 540cggaccaagg agtctaacgt ctatgcgagt
gtttgggtgt aaaacccgta cgcgtaatga 600aagtgaacgt aggtgagggc
ccgcaagggt gcatcatcga ccgatcctga tgtcttcgga 660tggatttgag
taagagcata gctgttggga cccgaaagat ggtgaactat gcctgaatag
720ggtgaagcca gaggaaactc tggtggaggc tcgtagcggt tctgacgtgc
aaatcgatcg 780tcgaatttgg gtatagggcg 80071000DNAKluyveromyces
marxianus 7ttagtgaggc ctcaggattt gcttagagaa gggggcaact ccatctcaga
gcgaaaaatc 60tggtcaaact tggtcattta gaggaactaa aagtcgtaac aaggtttccg
taggtgaacc 120tgcggaagga tcattaaaga ttatgaatga atagattact
gggggaatcg tctgaacaag 180gcctgcgctt aattgcgcgg ccagttcttg
attctctgct atcagttttc tatttctcat 240cctaaacaca atggagtttt
ttctctatga actacttccc tggagagctc gtctctccag 300tggacataaa
cacaaacaat attttgtatt atgaaaaact attatactat aaaatttaat
360attcaaaact ttcaacaacg gatctcttgg ttctcgcatc gatgaagaac
gcagcgaatt 420gcgatatgta ttgtgaattg cagattttcg tgaatcatca
aatctttgaa cgcacattgc 480gccctctggt attccagggg gcatgcctgt
ttgagcgtca tttctctctc aaacctttgg 540gtttggtagt gagtgatact
cgtctcgggt taacttgaaa gtggctagcc gttgccatct 600gcgtgagcag
ggctgcgtgt caagtctatg gactcgactc ttgcacatct acgtcttagg
660tttgcgccaa ttcgtggtaa gcttgggtca tagagactca taggtgttat
aaagactcgc 720tggtgtttgt ctccttgagg catacggctt taaccaaaac
tctcaaagtt tgacctcaaa 780tcaggtagga gtacccgctg aacttaagca
tatcaataag cggaggaaaa gaaaccaacc 840gggattgcct tagtaacggc
gagtgaagcg gcaaaagctc aaatttgaaa tctggcgtct 900tcgacgtccg
agttgtaatt tgaagaaggc gactttgtag ctggtccttg tctatgttcc
960ttggaacagg acgtcataga gggtgagaat cccgtgtggc
10008800DNAKluyveromyces marxianus 8cttagtacgg cgagtgaagc
ggcaaaagct caaatttgaa atctggcgtc ttcgacgtcc 60gagttgtaat ttgaagaagg
cgactttgta gctggtcctt gtctatgttc cttggaacag 120gacgtcatag
agggtgagaa tcccgtgtgg cgaggatccc agttatttgt aaagtgcttt
180cgacgagtcg agttgtttgg gaatgcagct ctaagtgggt ggtaaattcc
atctaaagct 240aaatattggc gagagaccga tagcgaacaa gtacagtgat
ggaaagatga aaagaacttt 300gaaaagagag tgaaaaagta cgtgaaattg
ttgaaaggga agggcatttg atcagacatg 360gcgtttgctt cggctttcgc
tgggccagca tcagttttag cggttggata aatcctcggg 420aatgtggctc
tgcttcggta gagtgttata gcccgtggga atacagccag ctgggactga
480ggattgcgac ttttgtcaag gatgctggcg taatggttaa atgccgcccg
tcttgaaaca 540cggaccaagg agtctaacgt ctatgcgagt gtttgggtgt
aaaacccgta cgcgtaatga 600aagtgaacgt aggtgagggc ccgcaagggt
gcatcatcga ccgatcctga tgtcttcgga 660tggatttgag taagagcata
gctgttggga cccgaaagat ggtgaactat gcctgaatag 720ggtgaagcca
gaggaaactc tggtggaggc tcgtagcggt tctgacgtgc aaatcgatcg
780tcgaatttgg gtatagggcg 80091000DNAKluyveromyces marxianus
9tagtgaggcc tcaggatttg ctttagagaa gggggcaact tccatctcag tgcgaaaaat
60ctggtcaaac ttggtcattt agaggaacta aaagtcgtaa caaggtttcc gtaggtgaac
120ctgcggaagg atcattaaag attatgaatg aatagattac tgggggaatc
gtctgaacaa 180ggcctgcgct taattgcgcg gccagttctt gattctctgc
tatcagtttt ctatttctca 240tcctaaacac aatggagttt tttctctatg
aactacttcc ctggagagct cgtctctcca 300gtggacataa acacaaacaa
tattttgtat tatgaaaaac tattatacta taaaatttaa 360tattcaaaac
tttcaacaac ggatctcttg gttctcgcat cgatgaagaa cgcagcgaat
420tgcgatatgt attgtgaatt gcagattttc gtgaatcatc aaatctttga
acgcacattg 480cgccctctgg tattccaggg ggcatgcctg tttgagcgtc
atttctctct caaacctttg 540ggtttggtag tgagtgatac tcgtctcggg
ttaacttgaa agtggctagc cgttgccatc 600tgcgtgagca gggctgcgtg
tcaagtctat ggactcgact cttgcacatc tacgtcttag 660gtttgcgcca
attcgtggta agcttgggtc atagagactc ataggtgtta taaagactcg
720ctggtgtttg tctccttgag gcatacggct ttaaccaaaa ctctcaaagt
ttgacctcaa 780atcaggtagg agtacccgct gaacttaagc atatcaataa
gcggaggaaa agaaaccaac 840cgggattgcc ttagtaacgg cgagtgaagc
ggcaaaagct caaatttgaa atctggcgtc 900ttcgacgtcc gagttgtaat
ttgaagaagg cgactttgta gctggtcctt gtctatgttc 960cttggaacag
gacgtcatag agggtgagaa tcccgtgtgg 100010800DNAKluyveromyces
marxianus 10ttagtacggc gtagtgaagc ggcaaaagct caaatttgaa atctggcgtc
ttcgacgtcc 60gagttgtaat ttgaagaagg cgactttgta gctggtcctt gtctatgttc
cttggaacag 120gacgtcatag agggtgagaa tcccgtgtgg cgaggatccc
agttatttgt aaagtgcttt 180cgacgagtcg agttgtttgg gaatgcagct
ctaagtgggt ggtaaattcc atctaaagct 240aaatattggc gagagaccga
tagcgaacaa gtacagtgat ggaaagatga aaagaacttt 300gaaaagagag
tgaaaaagta cgtgaaattg ttgaaaggga agggcatttg atcagacatg
360gcgtttgctt cggctttcgc tgggccagca tcagttttag cggttggata
aatcctcggg 420aatgtggctc tgcttcggta gagtgttata gcccgtggga
atacagccag ctgggactga 480ggattgcgac ttttgtcaag gatgctggcg
taatggttaa atgccgcccg tcttgaaaca 540cggaccaagg agtctaacgt
ctatgcgagt gtttgggtgt aaaacccgta cgcgtaatga 600aagtgaacgt
aggtgagggc ccgcaagggt gcatcatcga ccgatcctga tgtcttcgga
660tggatttgag taagagcata gctgttggga cccgaaagat ggtgaactat
gcctgaatag 720ggtgaagcca gaggaaactc tggtggaggc tcgtagcggt
tactgacgtg caaatcgatc 780gtcgaatttg ggtatagggc
800111000DNAKluyveromyces marxianus 11gtgaggcctc aggatttgct
tagagaaggg ggcaactcca tctcagagcg aaaaatctgg 60tcaaacttgg tcatttagag
gaactaaaag tcgtaacaag gtttccgtag gtgaacctgc 120ggaaggatca
ttaaagatta tgaatgaata gattactggg ggaatcgtct gaacaaggcc
180tgcgcttaat tgcgcggcca gttcttgatt ctctgctatc agttttctat
ttctcatcct 240aaacacaatg gagttttttc tctatgaact acttccctgg
agagctcgtc tctccagtgg 300acataaacac aaacaatatt ttgtattatg
aaaaactatt atactataaa atttaatatt 360caaaactttc aacaacggat
ctcttggttc tcgcatcgat gaagaacgca gcgaattgcg 420atatgtattg
tgaattgcag attttcgtga atcatcaaat ctttgaacgc acattgcgcc
480ctctggtatt ccagggggca tgcctgtttg agcgtcattt ctctctcaaa
cctttgggtt 540tggtagtgag tgatactcgt ctcgggttaa cttgaaagtg
gctagccgtt gccatctgcg 600tgagcagggc tgcgtgtcaa gtctatggac
tcgactcttg cacatctacg tcttaggttt 660gcgccaattc gtggtaagct
tgggtcatag agactcatag gtgttataaa gactcgctgg 720tgtttgtctc
cttgaggcat acggctttaa ccaaaactct caaagtttga cctcaaatca
780ggtaggagta cccgctgaac ttaagcatat caataagcgg aggaaaagaa
accaaccggg 840attgccttag taacggcgag tgaagcggca aaagctcaaa
tttgaaatct ggcgtcttcg 900acgtccgagt tgtaatttga agaaggcgac
tttgtagctg gtcctttgtc tatgttcctt 960ggaacaagga cgtcataaga
agggtgagaa tcccgtgtgg 100012800DNAKluyveromyces marxianus
12cttagtacgg cgtagtgaag cggcaaaagc tcaaatttga aatctggcgt cttcgacgtc
60cgagttgtaa tttgaagaag gcgactttgt agctggtcct tgtctatgtt ccttggaaca
120ggacgtcata gagggtgaga atcccgtgtg gcgaggatcc cagttatttg
taaagtgctt 180tcgacgagtc gagttgtttg ggaatgcagc tctaagtggg
tggtaaattc catctaaagc 240taaatattgg cgagagaccg atagcgaaca
agtacagtga tggaaagatg aaaagaactt 300tgaaaagaga gtgaaaaagt
acgtgaaatt gttgaaaggg aagggcattt gatcagacat 360ggcgtttgct
tcggctttcg ctgggccagc atcagtttta gcggttggat aaatcctcgg
420gaatgtggct ctgcttcggt agagtgttat agcccgtggg aatacagcca
gctgggactg 480aggattgcga cttttgtcaa ggatgctggc gtaatggtta
aatgccgccc gtcttgaaac 540acggaccaag gagtctaacg tctatgcgag
tgtttgggtg taaaacccgt acgcgtaatg 600aaagtgaacg taggtgaggg
cccgcaaggg tgcatcatcg accgatcctg atgtcttcgg 660atggatttga
gtaagagcat agctgttggg acccgaaaga tggtgaacta tgcctgaata
720gggtgaagcc agaggaaact ctggtggagg ctcgtagcgg ttctgacgtg
caaatcgatc 780gtcgaatttg ggtatagggc 800131000DNAKluyveromyces
marxianus 13ttagtgaggc ctcaggattt gcttagagaa gggggcaact ccatctcaga
gcgaaaaatc 60tggtcaaact tggtcattta gaggaactaa aagtcgtaac aaggtttccg
taggtgaacc 120tgcggaagga tcattaaaga ttatgaatga atagattact
gggggaatcg tctgaacaag 180gcctgcgctt aattgcgcgg ccagttcttg
attctctgct atcagttttc tatttctcat 240cctaaacaca atggagtttt
ttctctatga actacttccc tggagagctc gtctctccag 300tggacataaa
cacaaacaat attttgtatt atgaaaaact attatactat aaaatttaat
360attcaaaact ttcaacaacg gatctcttgg ttctcgcatc gatgaagaac
gcagcgaatt 420gcgatatgta ttgtgaattg cagattttcg tgaatcatca
aatctttgaa cgcacattgc 480gccctctggt attccagggg gcatgcctgt
ttgagcgtca tttctctctc aaacctttgg 540gtttggtagt gagtgatact
cgtctcgggt taacttgaaa gtggctagcc gttgccatct 600gcgtgagcag
ggctgcgtgt caagtctatg gactcgactc ttgcacatct acgtcttagg
660tttgcgccaa ttcgtggtaa gcttgggtca tagagactca taggtgttat
aaagactcgc 720tggtgtttgt ctccttgagg catacggctt taaccaaaac
tctcaaagtt tgacctcaaa 780tcaggtagga gtacccgctg aacttaagca
tatcaataag cggaggaaaa gaaaccaacc 840gggattgcct tagtaacggc
gagtgaagcg gcaaaagctc aaatttgaaa tctggcgtct 900tcgacgtccg
agttgtaatt tgaagaaggc gactttgtag ctggtccttg tctatgttcc
960ttggaacagg acgtcataga gggtgagaat cccgtgtggc
100014820DNAKluyveromyces marxianus 14gacaccggga tgccttagta
acggcgaagt gaagcggcaa aagctcaaat ttgaaatctg 60gcgtcttcga cgtccgagtt
gtaatttgaa gaaggcgact ttgtagctgg tccttgtcta 120tgttccttgg
aacaggacgt catagagggt gagaatcccg tgtggcgagg atcccagtta
180tttgtaaagt gctttcgacg agtcgagttg tttgggaatg cagctctaag
tgggtggtaa 240attccatcta aagctaaata ttggcgagag accgatagcg
aacaagtaca gtgatggaaa 300gatgaaaaga actttgaaaa gagagtgaaa
aagtacgtga aattgttgaa agggaagggc 360atttgatcag acatggcgtt
tgcttcggct ttcgctgggc cagcatcagt tttagcggtt 420ggataaatcc
tcgggaatgt ggctctgctt cggtagagtg ttatagcccg tgggaataca
480gccagctggg actgaggatt gcgacttttg tcaaggatgc tggcgtaatg
gttaaatgcc 540gcccgtcttg aaacacggac caaggagtct aacgtctatg
cgagtgtttg ggtgtaaaac 600ccgtacgcgt aatgaaagtg aacgtaggtg
agggcccgca agggtgcatc atcgaccgat 660cctgatgtct tcggatggat
ttgagtaaga gcatagctgt tgggacccga aagatggtga 720actatgcctg
aatagggtga agccagagga aactctggtg gaggctcgta gcggttctga
780cgtgcaaatc gatcgtcgaa tttgggtata gggcgaaaga
820151000DNAKluyveromyces marxianus 15tagtgaggcc tcaggatttg
cttagagaag ggggcaactc catctcagag cgaaaaatct 60ggtcaaactt ggtcatttag
aggaactaaa agtcgtaaca aggtttccgt aggtgaacct 120gcggaaggat
cattaaagat tatgaatgaa tagattactg ggggaatcgt ctgaacaagg
180cctgcgctta attgcgcggc cagttcttga ttctctgcta tcagttttct
atttctcatc 240ctaaacacaa tggagttttt tctctatgaa ctacttccct
ggagagctcg tctctccagt 300ggacataaac acaaacaata ttttgtatta
tgaaaaacta ttatactata aaatttaata 360ttcaaaactt tcaacaacgg
atctcttggt tctcgcatcg atgaagaacg cagcgaattg 420cgatatgtat
tgtgaattgc agattttcgt gaatcatcaa atctttgaac gcacattgcg
480ccctctggta ttccaggggg catgcctgtt tgagcgtcat ttctctctca
aacctttggg 540tttggtagtg agtgatactc gtctcgggtt aacttgaaag
tggctagccg ttgccatctg 600cgtgagcagg gctgcgtgtc aagtctatgg
actcgactct tgcacatcta cgtcttaggt 660ttgcgccaat tcgtggtaag
cttgggtcat agagactcat aggtgttata aagactcgct 720ggtgtttgtc
tccttgaggc atacggcttt aaccaaaact ctcaaagttt gacctcaaat
780caggtaggag tacccgctga acttaagcat atcaataagc ggaggaaaag
aaaccaaccg 840ggattgcctt agtaacggcg agtgaagcgg caaaagctca
aatttgaaat ctggcgtctt 900cgacgtccga gttgtaattt gaagaaggcg
actttgtagc tggtccttgt ctatgttcct 960tggaacagga cgtcatagag
ggtgagaatc ccgtgtggcg 100016800DNAKluyveromyces marxianus
16ttagtacggc gagtgaagcg gcaaaagctc aaatttgaaa tctggcgtct tcgacgtccg
60agttgtaatt tgaagaaggc gactttgtag ctggtccttg tctatgttcc ttggaacagg
120acgtcataga gggtgagaat cccgtgtggc gaggatccca gttatttgta
aagtgctttc 180gacgagtcga gttgtttggg aatgcagctc taagtgggtg
gtaaattcca tctaaagcta 240aatattggcg agagaccgat agcgaacaag
tacagtgatg gaaagatgaa aagaactttg 300aaaagagagt gaaaaagtac
gtgaaattgt tgaaagggaa gggcatttga tcagacatgg 360cgtttgcttc
ggctttcgct gggccagcat cagttttagc ggttggataa atcctcggga
420atgtggctct gcttcggtag agtgttatag cccgtgggaa tacagccagc
tgggactgag 480gattgcgact tttgtcaagg atgctggcgt aatggttaaa
tgccgcccgt cttgaaacac 540ggaccaagga gtctaacgtc tatgcgagtg
tttgggtgta aaacccgtac gcgtaatgaa 600agtgaacgta ggtgagggcc
cgcaagggtg catcatcgac cgatcctgat gtcttcggat 660ggatttgagt
aagagcatag ctgttgggac ccgaaagatg gtgaactatg cctgaatagg
720gtgaagccag aggaaactct ggtggaggct cgtagcggtt actgacgtgc
aaatcgatcg 780tcgaatttgg gtatagggcg 800
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012
D00013

D00014
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.