Zinc Wrought Alloy With Improved Coatability
MELZER; Armin ; et al.
U.S. patent application number 16/623019 was filed with the patent office on 2021-05-20 for zinc wrought alloy with improved coatability. The applicant listed for this patent is Grillo-Werke AG. Invention is credited to Kathrin HESNAOUI, Armin MELZER, Frank PRENGER, Didier ROLLEZ, Markus VAN WESEL, Joanna VON KRIES, Jurgen WISNIEWSKI.
| Application Number | 20210147962 16/623019 |
| Document ID | / |
| Family ID | 1000005400244 |
| Filed Date | 2021-05-20 |



| United States Patent Application | 20210147962 |
| Kind Code | A1 |
| MELZER; Armin ; et al. | May 20, 2021 |
ZINC WROUGHT ALLOY WITH IMPROVED COATABILITY
Abstract
The present invention relates to a zinc wrought alloy with improved coating properties as compared to known wrought alloys, and to the use thereof for preparing semifinished products, forgings, turned parts, locks, screw connections, locking cylinders, sleeves, fittings, pressed parts, pneumatic parts, hydraulic parts, mountings, valves and ball valves.
| Inventors: | MELZER; Armin; (Duisburg, DE) ; PRENGER; Frank; (Duisburg, DE) ; ROLLEZ; Didier; (Duisburg, DE) ; VAN WESEL; Markus; (Duisburg, DE) ; VON KRIES; Joanna; (Hittbergen, DE) ; WISNIEWSKI; Jurgen; (Duisburg, DE) ; HESNAOUI; Kathrin; (Duisburg, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005400244 | ||||||||||
| Appl. No.: | 16/623019 | ||||||||||
| Filed: | July 2, 2018 | ||||||||||
| PCT Filed: | July 2, 2018 | ||||||||||
| PCT NO: | PCT/EP2018/067824 | ||||||||||
| 371 Date: | December 16, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C22C 18/04 20130101 |
| International Class: | C22C 18/04 20060101 C22C018/04 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 4, 2017 | EP | 17179645.1 |
Claims
1. A method of making an article, the method comprising including in the article a zinc wrought alloy having an Al content of from 5% by weight to 12% by weight, a Cu content of from 0.1% by weight to 4% by weight, an Mg content of from 0.001% by weight to 0.05% by weight, wherein Zn is the balance to 100%, the article being one or more of preparing forgings, turned parts, locks, screw connections, locking cylinders, sleeves, fittings, pressed parts, pneumatic parts, hydraulic parts, mountings, valves and/or ball valves.
2. The method according to claim 1, characterized in that lead is not included in the alloy.
3. The method according to claim 1, characterized in that the content of Al is from 5% to 12% by weight.
4. The method according to claim 1, characterized in that the content of copper in the alloy is from 0.1% to 2.5% by weight.
5. The method according to claim 1, characterized in that the content of magnesium in the alloy is from 0.003% by weight to 0.05% by weight.
6. The method according to claim 1, characterized in that the alloy further contains silicon.
7. The method according to claim 1, wherein said alloy has the following composition: Al content of from 5% to 8% by weight, Cu content of from 0.5% to 1.5% by weight, Mg content of from 0.003% to 0.05% by weight, with zinc as the balance to reach 100% by weight.
8. The method according to claim 1, wherein said alloy has the following composition: Al content of from 9% to 12% by weight, a Cu content of from 0.5% to 1.5% by weight, an Mg content of from 0.003% to 0.05% by weight, with zinc as the balance to reach 100% by weight.
9. The method according to claim 1, wherein said alloy has a Ti content of up to 1% by weight.
10. A manufactured object that comprises a zinc wrought alloy as described in claim 1, the object being a semifinished product or an article, wherein the object passes a salt spray test according to DIN EN ISO 9227:2012, wherein the test requirements are considered to be met if no corrosion in the form of white rust occurs on the surface after 96 hours.
11. A manufactured object that comprises a Use of a zinc wrought alloy as described in claim 1, the object being a semifinished product or article, wherein the articles pass a condensation water test according to DIN EN ISO 6270-2:2016, wherein the test requirements are considered to be met if no blistering or corrosion occurs on a surface of the object after 96 hours in a condensation water chamber.
Description
[0001] The present invention relates to a zinc wrought alloy with improved coating properties as compared to known wrought alloys, and to the use thereof for preparing semifinished products, forgings, turned parts, locks, screw connections, locking cylinders, sleeves, fittings, pressed parts, pneumatic parts, hydraulic parts, mountings, valves and ball valves.
[0002] A wide variety of zinc alloys are known in the prior art.
[0003] As an example, EP 2675971 A--"Accessory consisting of a lock accessory" may be mentioned. It discloses a zinc alloy with an Al content of from 13 to 25%, a Cu content of from 0.2 to 3.5%, and an Mg content of less than 0.1%, which is employed for lock accessories.
[0004] EP 2 385 148 A--"Zinc alloy with high creep resistance" relates to a zinc-aluminum alloy with an Al content of 10 to <25%, a Cu content of 0.05 to 3%, an Mg content of from 0.001 to 0.1%, an Mn content of 0.05% to 1.0% and an Si content of from 0.05 to 1%. The disclosed alloy has a high creeping resistance and is suitable for the furnace brazing and normal brazing of heat exchangers.
[0005] U.S. Pat. No. 3,734,785--"Zinc forging alloy" claims a zinc-based alloy with an Al content of 9 to 22%, a Cu content of 0.5 to 1.5%, and an Mg content of 0.01 to 0.03%, which is particularly suitable for hot formability.
[0006] U.S. Pat. No. 3,880,679--"Method of forming zinc-aluminum alloys with good machinability" describes zinc-aluminum alloys with an Al content of 22 to 27%, a Cu content of 0 to 10%, an Mg content of 0.01 to 1%, and a Bi content of 0.01 to 3%.
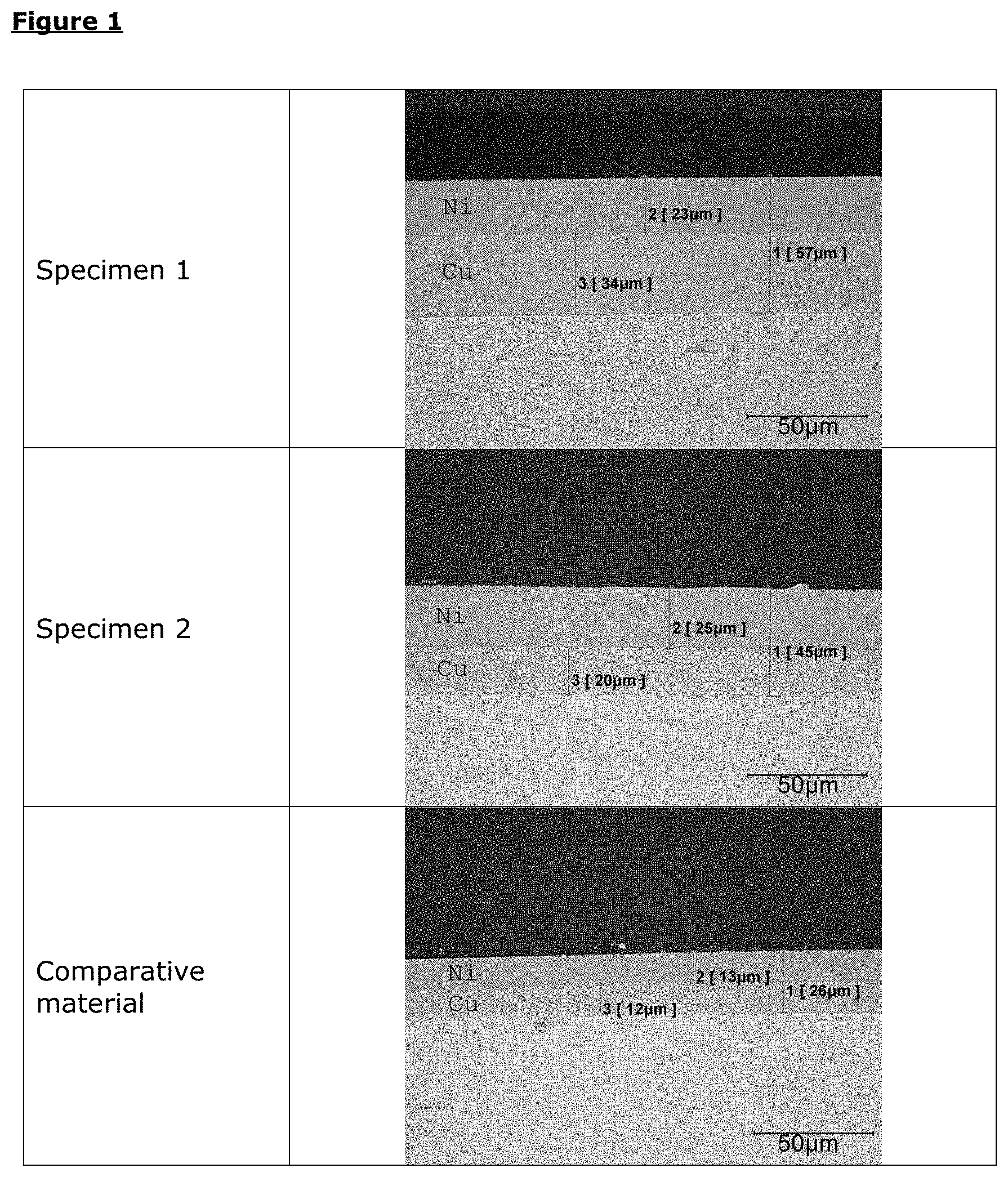
[0007] EP 0 679 198 A--"Method for producing Zn--Al--Cu alloy articles by centrifugal or die casting" describes a zinc alloy with an Al content of 6.0 to 8.0%, a Cu content of 3.2 to 4.3%, for preparing articles by centrifugal casting in a rubber mold, or pressure die-casting in a metal mold.
[0008] Further known are also zinc pressure die-casting alloys, also referred to as ZAMAK.RTM.. These consist of zinc-aluminum-copper-magnesium alloys, which cannot have the corresponding strength properties, however.
[0009] In addition to processability in terms of production, shaping or reshaping, the coatability of zinc alloys is also relevant. Semifinished products, forgings, turned parts, locks, screw connections, locking cylinders, sleeves, fittings, pressed parts, pneumatic parts, hydraulic parts, mountings, valves and ball valves consisting of corresponding alloys are often provided with different coatings for improving the appearance and/or for improving the corrosion resistance. In particular, a copper-copper-nickel coating serves for both corrosion resistance and appearance. Further, the coating with zinc and passivation mainly serves for a very high corrosion resistance, being an inexpensive alternative for a copper-copper-nickel coating.
[0010] The prior art with previous zinc wrought alloys is problematic, especially in view of surface coating. It has been the object of the present invention to provide a zinc-based wrought alloy having improved coating properties as compared to the prior art. This is to be achieved without including lead in the alloy. In addition, the coating should have good corrosion properties, i.e., have as little tendency to corrosion as possible. Surprisingly, it has been found that this is enabled by a low aluminum content of at most 12% by weight in zinc wrought alloys.
[0011] "Alloying" basically means the preparation of an alloy by melting a metal together with at least one other metal or non-metal. If in the present application it is referred to the fact that a metal or non-metal is not alloyed to an alloy, this means that the metal or non-metal in question is not actively added.
[0012] In a first embodiment, the object of the present invention is achieved by the use of a zinc wrought alloy having an Al content of from 5% by weight to 12% by weight, a Cu content of from 0.1% by weight to 4% by weight, an Mg content of from 0.001% by weight to 0.05% by weight, wherein Zn is the balance to 100%, for preparing forgings, turned parts, locks, screw connections, locking cylinders, sleeves, fittings, pressed parts, pneumatic parts, hydraulic parts, mountings, valves and/or ball valves.
[0013] If percentages are stated with respect to components contained in the alloy in the present application, they are percent by weight unless explicitly stated otherwise. In particular, no further metals in addition to the metals mentioned are alloyed with the alloy when it is prepared. More preferably, the alloy according to the invention is free of zirconium.
[0014] Surprisingly, it has been found that the surface coating of the parts prepared can be improved by limiting the aluminum content to a maximum of 12% by weight. Other zinc wrought alloys (as disclosed, for example, in EP 2675971--"Accessory consisting of a lock accessory") have poorer coating properties. Surprisingly, an improved adhesion strength and density of the coating could be achieved by limiting the aluminum content in zinc-aluminum-copper-magnesium alloys. This results in an improved corrosion protection. All methods of surface treatment (such as electroplating, PVD, CVD, passivation, coating, cathodic dip painting, powder coating) are suitable for the present invention without limitation.
[0015] Alloying with lead is not necessary. Preferably, lead is not alloyed. Preferred is a Pb content in the alloy according to the invention of <0.003% by weight, which is present in the alloy as an impurity of zinc, in particular, but not as an additional alloy component.
[0016] Particularly preferred is the use of a zinc wrought alloy with a content of Al of from 5% to 12% by weight, preferably from 5% to 8% by weight, or from 9% to 12% by weight. These ranges are preferred because all alloys are supereutectic therein, and there is a first beta phase in the crystal structure. This beta phase is preferred because it recrystallizes at room temperature very slowly (>10 years).
[0017] Particularly preferred is the use of a zinc wrought alloy with a content of Cu of from 0.1% by weight to 2.5% by weight, especially from 0.5% by weight to 1.5% by weight. This range is preferred to achieve the maximum mechanical strength, and to avoid the risk of forming of a brittle epsilon phase in the crystal structure.
[0018] Particularly preferred is the use of a zinc wrought alloy with a content of Mg from 0.003% to 0.05% by weight, especially from 0.003% by weight to 0.03% by weight. This range serves as a precaution to prevent intercrystalline corrosion by the residual traces of impurities.
[0019] The zinc wrought alloy used according to the invention may further contain silicon. If it contains silicon, the content of silicon in the alloy is in particular within a range of from 0.005% by weight to 0.02% by weight. The silicon content is determined by the selection of Al content, because it is an impurity in aluminum.
[0020] It has been found that an alloy having an Al content of from 5% to 8% by weight, a Cu content of from 0.5% by weight to 1.5% by weight, an Mg content of from 0.003% by weight to 0.05% by weight, with zinc as the balance to reach 100% by weight, has particularly good coating properties.
[0021] Particularly preferred according to the invention is a zinc wrought alloy with an Al content of from 9% by weight to 12% by weight, a Cu content of from 0.5% by weight to 1.5% by weight, an Mg content of from 0.003% by weight to 0.05% by weight, with zinc as the balance to reach 100% by weight. Corresponding alloys have good coating properties. Therefore, such an alloy, or the use thereof, is preferred.
[0022] In addition to the mentioned components, an alloy according to the invention may further comprise up to 1% by weight titanium (Ti). The titanium content of at most 1% in the zinc alloy is limited by the solubility of titanium. Titanium is an extremely effective alloy element, strongly affecting the microstructure and the mechanical properties of the alloy already in the ppm range because of its lattice structure. Corresponding alloys have an improved machinability.
[0023] In addition to the mentioned components, the alloy used according to the invention may also comprise impurities resulting from the fact that these components are derived from recycling. However, for the usual sources of the components, these are not critical. Common impurities are the presence of Cd, Pb, Sn and/or Fe. Preferably, these impurities are contained only in very small amounts, so that they do not adversely affect the properties of the alloy according to the invention. Therefore, preferred is a Pb content of <0.003% by weight, and/or a Cd content of <0.003% by weight, especially <0.0005% by weight, and/or an Sn content of <0.001% by weight, especially of <0.0005% by weight, and/or an Fe content of <0.05% by weight. Preferably, the content of all stated impurities is below the mentioned upper limits. Preferably, the content of all impurities is 0.07% by weight or less.
[0024] Thus, the present invention relates to the use of the zinc wrought alloy according to the invention for preparing semifinished products and articles with improved coating properties. When reference is made to the alloy according to the invention in the present application, this is supposed to mean the alloy used according to the invention.
[0025] Further included according to the invention is a semifinished product or article obtainable by processing the zinc wrought alloy according to the invention. This semifinished product may be, in particular, a billet, an extruded section, a drawn section, a wire, a strip, a powder, or a pressure die-cast alloy. In particular, the article may be a forging, turned part, lock, screw connection, locking cylinder, sleeve, fitting, pressed part, pneumatic part, hydraulic part, mounting, valve or ball valve.
[0026] The semifinished product according to the invention, especially the billet, can be prepared, for example, by casting the zinc wrought alloy according to the invention into a mold. When the alloy according to the invention has been formed into a billet shape, for example, a section can be prepared therefrom by reshaping by means of extrusion.
[0027] Further, the zinc wrought alloy according to the invention can be processed by different reshaping methods. Such reshaping methods include, in particular, rolling, forging and drawing. Further, the semifinished products prepared from the zinc wrought alloy according to the invention are processed further, for example, into forgings, turned parts, locks, screw connections, locking cylinders, sleeves, fittings, pressed parts, pneumatic parts, hydraulic parts, mountings, valves and ball valves using different processing methods. These articles according to the invention are excellently suitable for being subjected to coating methods.
[0028] The zinc wrought alloy according to the invention and the articles prepared therefrom exhibit improved coating properties as compared to conventional ZnAl/ZnAlCu/ZnAlCuMg alloys. These include an improved adhesion and density of the coating on the substrate material, and an improved corrosion resistance resulting therefrom, which shows in the results in a salt spray mist test, condensation water test, or thermal shock test.
[0029] The requirement for the invention is to enhance the coatability and corrosion resistance. This object was achieved by limiting the aluminum content in the alloy according to the invention to a maximum of 12% by weight. There may be mentioned, in particular, the coating properties that were significantly improved thereby, so that an improved corrosion resistance can be achieved. In the experiments, it is found that this can be achieved by reducing the aluminum content.
[0030] The prepared products comprising and, in particular, made of the zinc alloy according to the invention have an excellent processability, especially coatability, caused by the optimum composition of zinc, aluminum, copper and magnesium.
[0031] In the following Examples, the present invention is further explained in a non-limiting way, and advantages over the prior art are pointed out.
EXAMPLES
[0032] A qualification of zinc wrought alloys is usually affected by means of the following five methods delimited from one another:
Method 1 (Reshaping Method):
[0033] The billet was heated at 250.degree. C. in an oven. Thereafter, the billet was extruded into a round section. Further, the extruded round rod was drawn to a final dimension of 20 mm. The testing requirements are considered to be met if no signs of surface cracks or blisters have formed.
Method 2: (Layer Thickness Test by Polished Section Analysis):
[0034] As a second method, a microscopic examination of the coating by analogy with DIN EN ISO 1463:2004-08 is performed. This serves for testing the layer thickness and the bonding of the coating to the substrate material. For this purpose, polished transverse sections of the coated specimens are prepared. Thereupon, the sections are embedded, ground and polished. For a differentiation of the different microstructural components and the coating, the surface of the specimen is etched with aqueous sodium hydroxide (NaOH) solution. The testing requirements are considered to be met if the coating has a good bonding to the substrate material and reaches the desired layer thickness.
Method 3 (Corrosion Resistance by Salt Spray Test):
[0035] According to DIN EN ISO 9227:2012, a salt spray test with a sodium chloride solution having a neutral pH is performed. The testing requirements are considered to be met if no corrosion in the form of white rust occurs on the surface after 96 hours in the salt spray test.
Method 4 (Condensation Water Test):
[0036] According to DIN EN ISO 6270-2:2016, a test with a condensation water constant climate is performed. Thus, distilled water is filled into the base tray of a suitable sealed chamber. The chamber is heated at (40.+-.3) .degree. C., reaching a relative humidity of about 100%. The test requirements are considered to be met if no blistering or corrosion (in the form of white rust) occurs on the surface of the specimen after 96 hours in a condensation water chamber.
Method 5 (Thermal Shock Test):
[0037] In a thermal shock test by analogy with DIN EN ISO 2819:1995, the test specimen is stored at (220.+-.10) .degree. C. for 30 minutes and then immediately quenched in water with a temperature of 15.degree. C. to 25.degree. C. The test requirements are considered to be met if no chipping or blistering of the coating occurs.
[0038] Since corrosion resistance is presently in the focus, the alloys according to the invention were analyzed under this aspect and compared with prior art alloys. The compositions of the comparative alloys are shown in Table 1. Table 2 shows the compositions of the alloys according to the invention.
TABLE-US-00001 TABLE 1 Comparative materials Components Unit Zinc alloy Aluminum % by weight 13-25 Copper % by weight 0.2-3.5 Magnesium % by weight <0.1 Lead % by weight <0.004 Zinc % by weight balance
[0039] A zinc wrought alloy as described in EP 2 675 971 was used as a comparative material (information in column "zinc alloy" in Table 1).
[0040] From the following alloys according to the invention, billets having a diameter of 135 mm were prepared, which served as a starting point for the qualification:
TABLE-US-00002 TABLE 2 Alloys according to the invention Components Unit Specimen 1 Specimen 2 Aluminum % by weight 5-8 9-12 Copper % by weight 0.5-1.5 0.5-1.5 Magnesium % by weight 0.003-0.05 0.003-0.05
[0041] The above described tests were performed for testing the corrosion resistance.
Results:
[0042] Before the coating process, the specimens were pretreated in accordance with their alloy composition. Thereafter, the specimens were coated with different systems and tested according to the above-mentioned testing requirements. In the coating process, the same process times were used for all specimens for direct comparability.
[0043] For a clear evaluation of the specimens, a system with the following legend is used: [0044] ++: requirements of the corrosion test procedure are met particularly well [0045] +: requirements of the corrosion test procedure are met [0046] 0: requirements of the corrosion test procedure are met in part [0047] -: requirements of the corrosion test procedure are not met
[0048] By way of example, the coating with a cyanidic Cu--Cu--Ni system is shown in some detail.
Results of Method 2 (Layer Thickness Test by Polished Section Analysis):
TABLE-US-00003 [0049] TABLE 3 Layer thickness test by polished section analysis Copper [.mu.m] Nickel [.mu.m] Total [.mu.m] Specimen 1 34 23 57 Specimen 2 20 25 45 Comparative material 12 13 26
TABLE-US-00004 TABLE 4 Evaluation of the specimens by means of layer thickness results Specimen 1 ++ Specimen 2 ++ Comparative material 0
[0050] Microscopic examinations of the specimens including information of layer thickness are shown in FIG. 1. Please note that the different copper layers cannot be distinguished in the micrograph of the polished section.
[0051] Surprisingly, the results showed that specimen 1 and specimen 2 exhibit a higher layer thickness as compared to the comparative material. This shows that the present invention achieves a higher deposition rate. This accelerates the process of coating, which is an advantage. Further, a good bonding to the substrate material can be seen in the polished section micrographs of specimen 1 and specimen 2, while the comparative material in part shows defects between the copper layer and the substrate material.
Results of Method 3 (Corrosion Resistance by Salt Spray Test):
TABLE-US-00005 [0052] TABLE 5 Evaluation of specimens by means of salt spray test Specimen 1 + Specimen 2 + Comparative material 0
[0053] The salt spray test showed that no corrosion (in the form of white rust) has occurred on the surface of the specimens for specimen 1 and specimen 2. Thus, the substrate material under the coating was not attacked despite a highly stressing environment.
Results of Method 4 (Condensation Water Test):
TABLE-US-00006 [0054] TABLE 6 Evaluation of specimens by means of condensation water test Specimen 1 + Specimen 2 ++ Comparative material 0
[0055] Micrographs of the specimens are shown in FIG. 2. With specimen 1, a rough surface was formed, but on which no defects or blisters could be identified. The surface of specimen 2 remained surprisingly unchanged and showed neither roughening nor blisters. The comparative material formed large blisters by the test. The present invention met the testing requirements.
Results of Method 5 (Thermal Shock Test):
TABLE-US-00007 [0056] TABLE 7 Evaluation of specimens by means of thermal shock test Specimen 1 + Specimen 2 + Comparative material 0
[0057] Micrographs of the specimens are shown in FIG. 3. The results show that specimen 1 and specimen 2 showed no chipping or blistering. Thus, the testing requirements are considered to be met for the present invention. The comparative material in turn showed blisters that were in part pronounced.
[0058] In the following, the other coating systems are evaluated in summary
TABLE-US-00008 TABLE 8 General evaluation of the specimens Comparative Coating Specimen 1 Specimen 2 material Cu--Cu--Ni (cyanidic) + ++ 0 Cu--Cu--Ni (cyanide-free) + ++ 0
[0059] Surprisingly, the present invention had improved coating properties as compared to the prior art. This was shown in the different testing methods. The testing requirements were met in all methods. The bonding of the coating to the substrate material was good according to the requirements, and neither corrosion (in the form of white rust) nor chipping nor blistering occurred on the surface of the specimens. Further, the present invention surprisingly showed an enhanced deposition rate and thus achieved shorter process times. The particularly good test results of specimen 2 are to be mentioned.
[0060] In addition, a more environment-friendly kind of coating is surprisingly enabled by the present invention by means of cyanide-free copper plating.
* * * * *
D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.