Drug Releasing Coatings For Balloon Catheters
Wang; Lixiao
U.S. patent application number 17/140718 was filed with the patent office on 2021-05-20 for drug releasing coatings for balloon catheters. This patent application is currently assigned to Lutonix, Inc.. The applicant listed for this patent is Lutonix, Inc.. Invention is credited to Lixiao Wang.
| Application Number | 20210146100 17/140718 |
| Document ID | / |
| Family ID | 1000005370619 |
| Filed Date | 2021-05-20 |


| United States Patent Application | 20210146100 |
| Kind Code | A1 |
| Wang; Lixiao | May 20, 2021 |
DRUG RELEASING COATINGS FOR BALLOON CATHETERS
Abstract
Balloon catheters, methods for preparing balloon catheters, and uses of balloon catheters are disclosed. The balloon catheter includes an elongate member, an expandable balloon, and a coating layer overlying an exterior surface of the expandable balloon. The coating layer includes a total drug load of a hydrophobic therapeutic agent and a combination of additives including a first additive and a second additive. The hydrophobic therapeutic agent is paclitaxel, rapamycin, or paclitaxel and rapamycin. The first additive is a surfactant. The second additive is a chemical compound having one or more hydroxyl, amino, carbonyl, carboxyl, acid, amide, or ester groups.
| Inventors: | Wang; Lixiao; (Medina, MN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Lutonix, Inc. Franklin Lakes NJ |
||||||||||
| Family ID: | 1000005370619 | ||||||||||
| Appl. No.: | 17/140718 | ||||||||||
| Filed: | January 4, 2021 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16656268 | Oct 17, 2019 | 10912931 | ||
| 17140718 | ||||
| 15617786 | Jun 8, 2017 | 10485958 | ||
| 16656268 | ||||
| 15154662 | May 13, 2016 | 9700704 | ||
| 15617786 | ||||
| 15067739 | Mar 11, 2016 | 9757544 | ||
| 15154662 | ||||
| 14683612 | Apr 10, 2015 | 9289539 | ||
| 15067739 | ||||
| 13846143 | Mar 18, 2013 | 9005161 | ||
| 14683612 | ||||
| 12731835 | Mar 25, 2010 | 8414910 | ||
| 13846143 | ||||
| 12121986 | May 16, 2008 | 8414525 | ||
| 12731835 | ||||
| 11942452 | Nov 19, 2007 | 8414909 | ||
| 12121986 | ||||
| 14932033 | Nov 4, 2015 | 9770576 | ||
| 15154662 | ||||
| 13846358 | Mar 18, 2013 | 9180485 | ||
| 14932033 | ||||
| 12549180 | Aug 27, 2009 | 8430055 | ||
| 13846358 | ||||
| 60860084 | Nov 20, 2006 | |||
| 60880742 | Jan 17, 2007 | |||
| 60903529 | Feb 26, 2007 | |||
| 60904473 | Mar 2, 2007 | |||
| 60926850 | Apr 30, 2007 | |||
| 60981380 | Oct 19, 2007 | |||
| 60981384 | Oct 19, 2007 | |||
| 61092872 | Aug 29, 2008 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61L 2300/802 20130101; A61L 29/16 20130101; A61M 25/10 20130101; A61M 2025/1075 20130101; A61K 31/436 20130101; A61M 2025/105 20130101; A61K 31/337 20130101; A61L 31/08 20130101; A61L 2300/608 20130101; A61L 29/08 20130101; A61L 2300/416 20130101; A61L 2300/428 20130101; A61L 29/085 20130101; A61L 31/16 20130101; A61L 2300/606 20130101 |
| International Class: | A61M 25/10 20060101 A61M025/10; A61L 29/16 20060101 A61L029/16; A61L 29/08 20060101 A61L029/08; A61K 31/337 20060101 A61K031/337; A61K 31/436 20060101 A61K031/436; A61L 31/08 20060101 A61L031/08; A61L 31/16 20060101 A61L031/16 |
Claims
1. A balloon catheter for delivering a therapeutic agent to a target site of a lumen within a human, the balloon catheter comprising: an elongate member having a lumen and a distal end, an expandable balloon attached to the distal end of the elongate member and in fluidic communication with the lumen; and a coating layer overlying an exterior surface of the expandable balloon, wherein: the coating layer comprises a total drug load of a hydrophobic therapeutic agent and a combination of additives comprising a first additive and a second additive; the hydrophobic therapeutic agent is chosen from paclitaxel, rapamycin, everolimus, tacrolimus, temsirolimus, daunorubicin, doxorubicin, beta-lapachone, biologically active vitamin D, antioxidants, or combinations thereof; the first additive is one of PEG sorbitan monolaurates, PEG sorbitan monooleates, PEG sorbitan monopalmitates, PEG sorbitan monostearates, or combinations thereof; and the second additive is one or more of sorbitol, sorbitan, xylitol, gluconolactone, lactobionic acid, or an amino acid selected from the group consisting of alanine, asparagine, aspartic acid, cysteine, cystine, glycine, histidine, proline, isoleucine, leucine, methionine, phenylalanine, serine, threonine, tryptophan, tyrosine, valine, and combinations thereof.
2. The balloon catheter of claim 1, wherein the hydrophobic therapeutic agent is paclitaxel.
3. The balloon catheter of claim 1, wherein the hydrophobic therapeutic agent is rapamycin.
4. The balloon catheter of claim 1, wherein the hydrophobic therapeutic agent is a combination of antioxidants and paclitaxel or rapamycin.
5. The balloon catheter of claim 1, wherein the total drug load of the hydrophobic therapeutic agent is from 1 .mu.g to 20 .mu.g per square millimeter of the expandable balloon.
6. The balloon catheter of claim 1, wherein the total drug load of the hydrophobic therapeutic agent is from 2.5 .mu.g to 6 .mu.g per square millimeter of the expandable balloon.
7. The balloon catheter of claim 1, wherein the first additive is a PEG sorbitan monolaurate.
8. The balloon catheter of claim 7, wherein the PEG sorbitan monolaurate is PEG-20 sorbitan monolaurate.
9. The balloon catheter of claim 1, wherein the first additive is a PEG sorbitan monooleate.
10. The balloon catheter of claim 9, wherein the PEG sorbitan monooleate is PEG-20 sorbitan monooleate.
11. The balloon catheter of claim 1, wherein the first additive is one of PEG-20 sorbitan monolaurate, PEG-20 sorbitan monooleate, or combinations thereof.
12. The balloon catheter of claim 1, wherein the second additive is one of sorbitol, gluconolactone, or combinations thereof.
13. The balloon catheter of claim 1, wherein the second additive is sorbitol.
14. The balloon catheter of claim 1, wherein the second additive is gluconolactone.
15. The balloon catheter of claim 1, wherein the ratio by weight of the combination of additives in the coating layer to the hydrophobic therapeutic agent in the coating layer is about 10 to 0.5.
16. The balloon catheter of claim 1, wherein: the coating layer consists essentially of the hydrophobic therapeutic agent, the first additive, and the second additive; the hydrophobic therapeutic agent is paclitaxel; the first additive is one of PEG-20 sorbitan monolaurate, PEG-20 sorbitan monooleate, or combinations thereof; and the second additive is one of sorbitol, gluconolactone, or combinations thereof.
17. The balloon catheter of claim 1, wherein: the coating layer consists essentially of the hydrophobic therapeutic agent, the first additive, and the second additive; the hydrophobic therapeutic agent is paclitaxel; the first additive is PEG-20 sorbitan monolaurate; and the second additive is sorbitol.
18. The balloon catheter of claim 1, wherein: the coating layer consists essentially of the hydrophobic therapeutic agent, the first additive, and the second additive; the hydrophobic therapeutic agent is paclitaxel; the second additive is gluconolactone; and the total drug load of the hydrophobic therapeutic agent is from 2 .mu.g to 6 .mu.g per square millimeter of the expandable balloon.
19. The balloon catheter of claim 1, wherein the coating layer has a thickness of from about 0.1 .mu.m to 15 .mu.m.
20. A balloon catheter for delivering a therapeutic agent to a target site of a lumen within a human, the balloon catheter comprising: an elongate member having a lumen and a distal end, an expandable balloon attached to the distal end of the elongate member and in fluid communication with the lumen; and a coating layer overlying an exterior surface of the expandable balloon, wherein: the coating layer comprises a total drug load of a hydrophobic therapeutic agent and a combination of additives comprising a first additive and a second additive; the total drug load of the hydrophobic therapeutic agent is from 2.5 .mu.g to 6 .mu.g per square millimeter of the expandable balloon; the hydrophobic therapeutic agent is chosen from paclitaxel, rapamycin, everolimus, tacrolimus, temsirolimus, daunorubicin, doxorubicin, beta-lapachone, biologically active vitamin D, antioxidants, or combinations thereof; the first additive is one of PEG-20 sorbitan monolaurate, PEG-20 sorbitan monooleate, PEG-20 sorbitan monopalmitate, PEG-20 sorbitan monostearate, or combinations thereof; the second additive is one of or more of sorbitol, gluconolactone, or an amino acid selected from the group consisting of alanine, asparagine, aspartic acid, cysteine, cystine, glycine, histidine, proline, isoleucine, leucine, methionine, phenylalanine, serine, threonine, tryptophan, tyrosine, valine, and combinations thereof; and the ratio by weight of the combination of additives in the coating layer to the hydrophobic therapeutic agent in the coating layer is about 10 to 0.5.
Description
CROSS-REFERENCES TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. application Ser. No. 16/656,268, filed Oct. 17, 2019, which is a continuation of U.S. application Ser. No. 15/617,786 filed Jun. 8, 2017, which is a continuation of U.S. application Ser. No. 15/154,662 filed May 13, 2016, which is a continuation-in-part of U.S. application Ser. No. 15/067,739, filed Mar. 11, 2016, which is a continuation of U.S. application Ser. No. 14/683,612 filed Apr. 10, 2015, now U.S. Pat. No. 9,289,539, issued Mar. 22, 2016, which is a continuation of U.S. application Ser. No. 13/846,143, filed Mar. 18, 2013, now U.S. Pat. No. 9,005,161, issued Apr. 14, 2015, which is a continuation of U.S. application Ser. No. 12/731,835, filed Mar. 25, 2010, now U.S. Pat. No. 8,414,910, issued Apr. 9, 2013, which is a continuation-in-part of U.S. application Ser. No. 12/121,986, filed May 16, 2008, now U.S. Pat. No. 8,414,525, issued Apr. 9, 2013, which is a continuation-in-part of U.S. application Ser. No. 11/942,452, filed Nov. 19, 2007, now U.S. Pat. No. 8,414,909, issued Apr. 9, 2013, which claims the benefit of priority of U.S. Provisional Application No. 60/860,084, filed Nov. 20, 2006, of U.S. Provisional Application No. 60/880,742, filed Jan. 17, 2007, of U.S. Provisional Application No. 60/897,427, filed Jan. 25, 2007, of U.S. Provisional Application No. 60/903,529, filed Feb. 26, 2007, of U.S. Provisional Application No. 60/904,473, filed Mar. 2, 2007, of U.S. Provisional Application No. 60/926,850, filed Apr. 30, 2007, of U.S. Provisional Application No. 60/981,380, filed Oct. 19, 2007, and of U.S. Provisional Application No. 60/981,384, filed Oct. 19, 2007; U.S. application Ser. No. 15/154,662 being also a continuation-in-part of U.S. application Ser. No. 14/932,033, filed Nov. 4, 2015, which is a continuation of U.S. application Ser. No. 13/846,358, filed Mar. 18, 2013, now U.S. Pat. No. 9,180,485, issued Nov. 10, 2015, which is a continuation of U.S. application Ser. No. 12/549,180, filed Aug. 27, 2009, now U.S. Pat. No. 8,430,055, issued Apr. 30, 2013, which claims the benefit of priority of U.S. Provisional Application No. 61/092,872, filed Aug. 29, 2008. The full disclosures of all of the foregoing applications are incorporated by reference herein.
TECHNICAL FIELD
[0002] The present disclosure relates to balloon catheters, methods of preparing balloon catheters, and uses of balloon catheters. More specifically, the present disclosure relates to balloon catheters for delivering a therapeutic agent to a blood vessel, wherein the balloon catheters have a coating layer having a hydrophobic therapeutic agent and a combination of additives.
BACKGROUND
[0003] It has become increasingly common to treat a variety of medical conditions by introducing a medical device into the vascular system or other lumen within a human. While such treatment initially appears successful, the initial success is often compromised by the recurrence of disease, such as stenosis (i.e., restenosis) after such treatment. Restenosis involves a physiological response to vascular injury caused by angioplasty. Over time, de-endothelization and injury to smooth muscle cells results in thrombus deposition, leukocyte and macrophage infiltration, smooth muscle cell proliferation/migration, fibrosis, and extracellular matrix deposition. In response to the incidence of restenosis, numerous local drug delivery systems have been developed for the treatment restenosis after balloon angioplasty.
[0004] Balloon catheters are one such local delivery system that has been found to be effective in the treatment and prevention of restenosis. Generally, balloons coated with an active agent are pressed against the wall of a blood vessel when the blood vessel is dilated to deliver the active agent. Accordingly, it may be advantageous for the active agent in the coating to be rapidly released and absorbed by blood vessel tissues. Any component in the coating which inhibits rapid release of the active agent may be disadvantageous.
[0005] The iodine contrast agent iopromide has been used with paclitaxel to coat balloon catheters and has met some success in the treatment of restenosis. While it has been reported that such contrast agent improves adhesion of paclitaxel to the balloon surface, iodinated contrast agents suffer from a variety of disadvantages. For example, when used for diagnostic procedures, iodinated contrast agents have complication rates of 5-30%. Additionally, iodinated contrast agents are associated with the risk of bradycardia, ventricular arrthymia, and fibrillation, and may also induce renal failure. Further, the Food and Drug Administration issued a second public health advisory in 2006 concerning a serious late adverse reaction to contrast agents known as Nephrogenic Systemic Fibrosis or Mephrogenic Fibrosing Dermopathy.
[0006] Moreover, iodinated X-ray contrast agents are unable to cross membrane lipid bilayers in order to enter cells of the vasculature. As a result, they are not optimally effective at carrying hydrophobic therapeutic agents such as paclitaxel into cells. The percentage of paclitaxel reported to be taken up by vascular tissue after deployment of these devices is only 5-20%. Additionally, the compatibility and/or miscibility of paclitaxel and iopromide is not optimal, and the integrity and uniformity of coating is poor. Such deficiencies adversely affect the amount and uniformity of hydrophobic therapeutic agent delivered to target tissue. Accordingly, additional embodiments of coating layers for balloon catheters are desired.
SUMMARY
[0007] In one embodiment, a balloon catheter for delivering a therapeutic agent to a blood vessel is disclosed. The balloon catheter delivers a therapeutic agent to the blood vessel while the balloon catheter is deployed at a target site of the blood vessel. The balloon catheter includes an elongate member, an expandable balloon, and a coating layer. The elongate member has a lumen and a distal end. The expandable balloon is attached to the distal end of the elongate member and is in fluid communication with the lumen of the elongate member. The coating layer overlies an exterior surface of the expandable balloon. The coating layer includes a total drug load of a hydrophobic therapeutic agent and a combination of additives including a first additive and a second additive. The hydrophobic therapeutic agent is one of paclitaxel, rapamycin, or combinations thereof. The first additive is one of PEG sorbitan monolaurates, PEG sorbitan monooleates, or combinations thereof. The second additive is one of sorbitol, sorbitan, xylitol, gluconolactone, lactobionic acid, or combinations thereof.
[0008] In another embodiment, a balloon catheter for delivering a therapeutic agent to a blood vessel is disclosed. The balloon catheter delivers a therapeutic agent to the blood vessel while the balloon catheter is deployed at a target site of the blood vessel. In this embodiment, the balloon catheter includes an elongate member, an expandable balloon, and a coating layer. The elongate member has a lumen and a distal end. The expandable balloon is attached to the distal end of the elongate member and is in fluid communication with the lumen of the elongate member. The coating layer overlies an exterior surface of the expandable balloon. In this embodiment, the coating layer includes a total drug load of a hydrophobic therapeutic agent and a combination of additives including a first additive and a second additive. The total drug load of the hydrophobic therapeutic agent is from 2.5 .mu.g to 6 .mu.g per square millimeter of the expandable balloon. The hydrophobic therapeutic agent is one of paclitaxel, rapamycin, or combinations thereof. The first additive is one of PEG-20 sorbitan monolaurate, PEG-20 sorbitan monooleate, or combinations thereof. The second additive is one of sorbitol, gluconolactone, or combinations thereof. The ratio by weight of the combination of additives in the coating layer to the hydrophobic therapeutic agent in the coating layer is about 10 to 0.5.
[0009] It is understood that both the foregoing general description and the following detailed description are exemplary and explanatory only and are not restrictive of the present disclosure as claimed.
BRIEF DESCRIPTION OF THE DRAWINGS
[0010] FIG. 1 is a perspective view of an exemplary embodiment of a balloon catheter according to the present disclosure; and
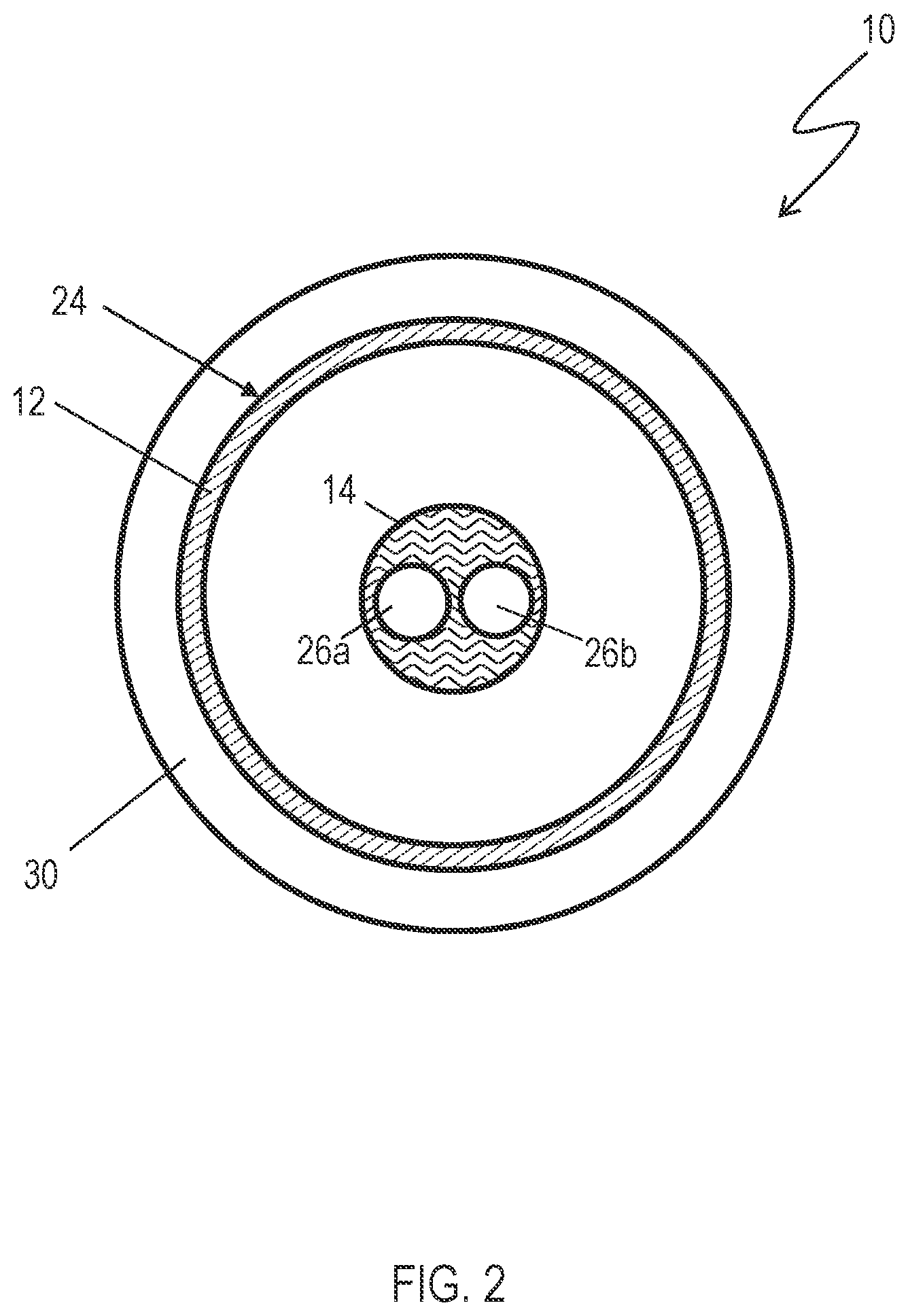
[0011] FIG. 2 is a cross-sectional view of an embodiment of the distal portion of the balloon catheter of FIG. 1, taken along line A-A, showing an exemplary coating layer.
[0012] Skilled artisans should appreciate that elements in the figures are illustrated for simplicity and clarity and are not necessarily drawn to scale. For example, the dimensions of some of the elements in the figures may be exaggerated relative to other elements, as well as conventional parts removed, to help to improve understanding of the various embodiments described herein.
DETAILED DESCRIPTION
[0013] Embodiments of the present disclosure relate to balloon catheters for delivering a therapeutic agent to a blood vessel. In one embodiment, the balloon catheter includes an elongate member, an expandable balloon, and a coating layer. Reference will now be made in detail to balloon catheters. More particularly, embodiments of the balloon catheter and, more specifically, the elongate member and the expandable balloon of the balloon catheter will now be described in detail with reference to FIG. 1. Thereafter, embodiments of the coating layer will be described in detail with reference to FIG. 2.
I. Balloon Catheters with Coating Layer
[0014] Referring to FIG. 1, a balloon catheter 10 is disclosed. The balloon catheter 10 has a proximal end 18 and a distal end 20. The balloon catheter 10 may be any suitable catheter for desired use, including conventional balloon catheters known to one of ordinary skill in the art. For example, the balloon catheter 10 may be a rapid exchange or over-the-wire catheter. The balloon catheter 10 may be made of any suitable biocompatible material.
[0015] As shown in FIGS. 1 and 2, in one embodiment, the balloon catheter 10 includes an expandable balloon 12 and an elongate member 14. The elongate member 14 extends between the proximal end 18 and the distal end 20 of the balloon catheter 10. The elongate member 14 has at least one lumen 26a, 26b and a distal end 22. The elongate member 14 may be a flexible member which is a tube made of suitable biocompatible material. The elongate member 14 may have one lumen or, as shown in FIGS. 1 and 2, more than one lumen 26a, 26b therein. For example, the elongate member 14 may include a guide-wire lumen 26b that extends to the distal end 20 of the balloon catheter 10 from a guide-wire port 15 at the proximal end 18 of the balloon catheter 10. The elongate member 14 may also include an inflation lumen 26a that extends from an inflation port 17 of the balloon catheter 10 to the inside of the expandable balloon 12 to enable inflation of the expandable balloon 12. From the embodiment of FIGS. 1 and 2, even though the inflation lumen 26a and the guide-wire lumen 26b are shown as side-by-side lumens, it should be understood that the one or more lumens present in the elongate member 14 may be configured in any manner suited to the intended purposes of the lumens including, for example, introducing inflation media and/or introducing a guide-wire. Many such configurations are well known in the art.
[0016] The expandable balloon 12 is attached to the distal end 22 of the elongate member 14. The expandable balloon 12 has an exterior surface 24 and is inflatable. The expandable balloon 12 is in fluidic communication with a lumen of the elongate member 14, (for example, with the inflation lumen 26a). At least one lumen of the elongate member 14 is configured to receive inflation media and to pass such media to the expandable balloon 12 for its expansion. Examples of inflation media include air, saline, and contrast media.
[0017] Still referring to FIG. 1, in one embodiment, the balloon catheter 10 includes a handle assembly such as a hub 16. The hub 16 may be attached to the balloon catheter 10 at the proximal end 18 of the balloon catheter 10. The hub 16 may connect to and/or receive one or more suitable medical devices, such as a source of inflation media or a guide wire. For example, a source of inflation media (not shown) may connect to the inflation port 17 of the hub 16 (for example, through the inflation lumen 26a), and a guide wire (not shown) may be introduced to the guide-wire port 15 of the hub 16, (for example through the guide-wire lumen 26b).
[0018] Referring now to FIG. 2, a balloon catheter 10 having a coating layer 30 is disclosed. The coating layer 30 overlies an exterior surface 24 of the expandable balloon 12. The coating layer 30 includes a hydrophobic therapeutic agent and a combination of additives. In one particular embodiment, the coating layer 30 consists essentially of the hydrophobic therapeutic agent and the combination of additives. Stated another way, in this particular embodiment, the coating layer 30 includes only the therapeutic agent and the combination of additives, without any other materially significant components. In another particular embodiment, the coating layer 30 is from about 0.1 .mu.m to 15 .mu.m thick.
[0019] A. Hydrophobic Therapeutic Agent
[0020] In one embodiment, the coating layer 30 includes a hydrophobic therapeutic agent. However, the coating layer 30 may also include two or more hydrophobic therapeutic agents. The hydrophobic therapeutic agent may be a lipophilic substantially water insoluble therapeutic agent. For example, the hydrophobic therapeutic agent may be paclitaxel, rapamycin, daunorubicin, doxorubicin, lapachone, vitamin D2 and analogues and derivative thereof, and vitamin D3 and analogues and derivatives thereof. Additional suitable hydrophobic therapeutic agents generally include glucocorticoids (e.g., dexamethasone, betamethasone), hirudin, angiopeptin, aspirin, growth factors, antisense agents, polynucleotides, RNAi, siRNA, anti-cancer agents, anti-proliferative agents, oligonucleotides, and, more generally, anti-platelet agents, anti-coagulant agents, anti-mitotic agents, anti-metabolite agents, antioxidants, antiobiotic agents, anti-chemotactic, anti-inflammatory agents, photosensitizing agents, and collagen synthesis inhibitors.
[0021] With regard to antisense agents, polynucleotides, RNAi, and siRNA, suitable hydrophobic therapeutic agents include agents that inhibit inflammation and/or smooth muscle cell or fibroblast proliferation. With regard to anti-proliferative agents, suitable hydrophobic therapeutic agents include amlodipine and doxazocin. With regard to anti-platelet agents, suitable hydrophobic therapeutic agents include aspirin and dipyridamole. Aspirin may also be classified as an anti-platelet and an anti-inflammatory agent. Additionally, dipyridamole also exhibits anti-platelet characteristics. With regard to anti-coagulant agents, suitable hydrophobic therapeutic agents include heparin, protamine, hirudin, and tick anticoagulant protein.
[0022] With regard to anti-mitotic agents and anti-metabolite agents, suitable hydrophobic therapeutic agents include methotrexate, azathioprine, vincristine, vinblastine, 5-fluorouracil, adriamycin, and mutamycin. With regard to antioxidants, suitable hydrophobic therapeutic agents include probucol. With regard to antibiotic agents, suitable hydrophobic therapeutic agents include penicillin, cefoxitin, oxacillin, tobramycin, and gentamicin. With regard to photosensitizing agents, suitable hydrophobic therapeutic agents include agents for photodynamic or radiation therapy, including various porphyrin compounds such as porfimer. With regard to collagen synthesis inhibitors, suitable hydrophobic therapeutic agents include tranilast.
[0023] In one embodiment, the hydrophobic therapeutic agent includes at least one of paclitaxel or analogs thereof, rapamycin or analogs thereof, beta-lapachone or analogues thereof, biological vitamin D or analogs thereof, and combinations thereof. In one particular embodiment, the hydrophobic therapeutic agent is paclitaxel, rapamycin, or combinations thereof. In a further embodiment, the hydrophobic therapeutic agent is paclitaxel. Alternatively, in another further embodiment, the hydrophobic therapeutic agent is rapamycin.
[0024] As used herein, "derivative" refers to a chemically or biologically modified version of a chemical compound that is structurally similar to a parent compound and (actually or theoretically) derivable from that parent compound. A derivative may or may not have different chemical or physical properties of the parent compound. For example, the derivative may be more hydrophilic or it may have altered reactivity as compared to the parent compound. Derivatization (i.e., modification) may involve substitution of one or more moieties within the molecule (e.g., a change in functional group). For example, a hydrogen may be substituted with a halogen, such as fluorine or chlorine, or a hydroxyl group (--OH) may be replaced with a carboxylic acid moiety (--COOH). The term "derivative" also includes conjugates, and prodrugs of a parent compound (i.e., chemically modified derivatives which can be converted into the original compound under physiological conditions). For example, the prodrug may be an inactive form of an active agent. Under physiological conditions, the prodrug may be converted into the active form of the compound. Prodrugs may be formed, for example, by replacing one or two hydrogen atoms on nitrogen atoms by an acyl group (acyl prodrugs) or a carbamate group (carbamate prodrugs). More detailed information relating to prodrugs is found, for example, in Fleisher et al., Advanced Drug Delivery Reviews 19 (1996) 115; Design of Prodrugs, H. Bundgaard (ed.), Elsevier, 1985; or H. Bundgaard, Drugs of the Future 16 (1991) 443. The term "derivative" is also used to describe all solvates, for example hydrates or adducts (e.g., adducts with alcohols), active metabolites, and salts of the parent compound. The type of salt that may be prepared depends on the nature of the moieties within the compound. For example, acidic groups, for example carboxylic acid groups, can form alkali metal salts or alkaline earth metal salts (e.g., sodium salts, potassium salts, magnesium salts and calcium salts, as well as salts with physiologically tolerable quaternary ammonium ions and acid addition salts with ammonia and physiologically tolerable organic amines such as triethylamine, ethanolamine or tris-(2-hydroxyethyl)amine). Basic groups can form acid addition salts, for example with inorganic acids such as hydrochloric acid, sulfuric acid or phosphoric acid, or with organic carboxylic acids and sulfonic acids such as acetic acid, citric acid, benzoic acid, maleic acid, fumaric acid, tartaric acid, methanesulfonic acid or p-toluenesulfonic acid. Compounds which simultaneously contain a basic group and an acidic group, for example a carboxyl group in addition to basic nitrogen atoms, can be present as zwitterions. Salts can be obtained by customary methods known to those skilled in the art, for example by combining a compound with an inorganic or organic acid or base in a solvent or diluent, or from other salts by cation exchange or anion exchange.
[0025] As used herein, "analog" or "analogue" refers to a chemical compound that is structurally similar to another but differs slightly in composition (as in the replacement of one atom by an atom of a different element or in the presence of a particular functional group), but may or may not be derivable from the parent compound. A "derivative" differs from an "analog" or "analogue" in that a parent compound may be the starting material to generate a "derivative," whereas the parent compound may not necessarily be used as the starting material to generate an "analog."
[0026] Numerous paclitaxel analogs are known in the art. Examples of paclitaxel include docetaxol (TAXOTERE, Merck Index entry 3458), and 3'-desphenyl-3'-(4-ntirophenyl)-N-debenzoyl-N-(t-butoxycarbonyl)-10-deace- tyltaxol. Further representative examples of paclitaxel analogs that can be used as therapeutic agents include 7-deoxy-docetaxol, 7,8-cyclopropataxanes, N-substituted 2-azetidones, 6,7-epoxy paclitaxels, 6,7-modified paclitaxels, 10-desacetoxytaxol, 10-deacetyltaxol (from 10-deacetylbaccatin III), phosphonooxy and carbonate derivatives of taxol, taxol 2',7-di(sodium 1,2-benzenedicarboxylate, 10-desacetoxy-11,12-dihydrotaxol-10,12(18)-diene derivatives, 10-desacetoxytaxol, Protaxol (2'- and/or 7-O-ester derivatives), (2'- and/or 7-O-carbonate derivatives), asymmetric synthesis of taxol side chain, fluoro taxols, 9-deoxotaxane, (13-acetyl-9-deoxobaccatine III, 9-deoxotaxol, 7-deoxy-9-deoxotaxol, 10-desacetoxy-7-deoxy-9-deoxotaxol), derivatives containing hydrogen or acetyl group and a hydroxy and tert-butoxycarbonylamino, sulfonated 2'-acryloyltaxol and sulfonated 2'-O-acyl acid taxol derivatives, succinyltaxol, 2'-.gamma.-aminobutyryltaxol formate, 2'-acetyl taxol, 7-acetyl taxol, 7-glycine carbamate taxol, 2'-OH-7-PEG(5000) carbamate taxol, 2'-benzoyl and 2',7-dibenzoyl taxol derivatives, other prodrugs (2'-acetyltaxol; 2',7-diacetyltaxol; 2'succinyltaxol; 2'-(beta-alanyl)-taxol); 2'gamma-aminobutyryltaxol formate; ethylene glycol derivatives of 2'-succinyltaxol; 2'-glutaryltaxol; 2'-(N,N-dimethylglycyl)taxol; 2'-(2-(N,N-dimethylamino)propionyl)taxol; 2'orthocarboxybenzoyl taxol; 2'aliphatic carboxylic acid derivatives of taxol, Prodrugs {2'(N,N-diethylaminopropionyl)taxol, 2'(N,N-dimethylglycyl)taxol, 7(N,N-dimethylglycyl)taxol, 2',7-di-(N,N-dimethylglycyl)taxol, 7(N,N-diethylaminopropionyl)taxol, 2',7-di(N,N-diethylaminopropionyl)taxol, 2'-(L-glycyl)taxol, 7-(L-glycyl)taxol, 2',7-di(L-glycyl)taxol, 2'-(L-alanyl)taxol, 7-(L-alanyl)taxol, 2',7-di(L-alanyl)taxol, 2'-(L-leucyl)taxol, 7-(L-leucyl)taxol, 2',7-di(L-leucyl)taxol, 2'-(L-isoleucyl)taxol, 7-(L-isoleucyl)taxol, 2',7-di(L-isoleucyl)taxol, 2'-(L-valyl)taxol, 7-(L-valyl)taxol, 2'7-di(L-valyl)taxol, 2'-(L-phenylalanyl)taxol, 7-(L-phenylalanyl)taxol, 2',7-di(L-phenylalanyl)taxol, 2'-(L-prolyl)taxol, 7-(L-prolyl)taxol, 2',7-di(L-prolyl)taxol, 2'-(L-lysyl)taxol, 7-(L-lysyl)taxol, 2',7-di(L-lysyl)taxol, 2'-(L-glutamyl)taxol, 7-(L-glutamyl)taxol, 2',7-di(L-glutamyl)taxol, 2'-(L-arginyl)taxol, 7-(L-arginyl)taxol, 2',7-di(L-arginyl)taxol, taxol analogues with modified phenylisoserine side chains, TAXOTERE, (N-debenzoyl-N-tert-(butoxycaronyl)-10-deacetyltaxol, and taxanes (e.g., baccatin III, cephalomannine, 10-deacetylbaccatin III, brevifoliol, yunantaxusin and taxusin); and other taxane analogues and derivatives, including 14-beta-hydroxy-10 deacetybaccatin III, debenzoyl-2-acyl paclitaxel derivatives, benzoate paclitaxel derivatives, phosphonooxy and carbonate paclitaxel derivatives, sulfonated 2'-acryloyltaxol; sulfonated 2'-O-acyl acid paclitaxel derivatives, 18-site-substituted paclitaxel derivatives, chlorinated paclitaxel analogues, C4 methoxy ether paclitaxel derivatives, sulfenamide taxane derivatives, brominated paclitaxel analogues, Girard taxane derivatives, nitrophenyl paclitaxel, 10-deacetylated substituted paclitaxel derivatives, 14-beta-hydroxy-10 deacetylbaccatin III taxane derivatives, C7 taxane derivatives, C10 taxane derivatives, 2-debenzoyl-2-acyl taxane derivatives, 2-debenzoyl and -2-acyl paclitaxel derivatives, taxane and baccatin III analogues bearing new C2 and C4 functional groups, n-acyl paclitaxel analogues, 10-deacetylbaccatin III and 7-protected-10-deacetylbaccatin III derivatives from 10-deacetyl taxol A, 10-deacetyl taxol B, and 10-deacetyl taxol, benzoate derivatives of taxol, 2-aroyl-4-acyl paclitaxel analogues, ortho-ester paclitaxel analogues, 2-aroyl-4-acyl paclitaxel analogues and 1-deoxy paclitaxel and 1-deoxy paclitaxel analogues.
[0027] Other examples of paclitaxel analogs suitable for use herein include those listed in U.S. Pat. App. Pub. No. 2007/0212394, and U.S. Pat. No. 5,440,056, each of which is incorporated herein by reference.
[0028] Many rapamycin analogs are known in the art. Non-limiting examples of analogs of rapamycin include, but are not limited to, everolimus, tacrolimus, CCI-779, ABT-578, AP-23675, AP-23573, AP-23841, 7-epi-rapamycin, 7-thiomethyl-rapamycin, 7-epi-trimethoxyphenyl-rapamycin, 7-epi-thiomethyl-rapamycin, 7-demethoxy-rapamycin, 32-demethoxy-rapamycin, 2-desmethyl-rapamycin, prerapamycin, temsirolimus, and 42-O-(2-hydroxy)ethyl rapamycin.
[0029] Other analogs of rapamycin include: rapamycin oximes (U.S. Pat. No. 5,446,048); rapamycin aminoesters (U.S. Pat. No. 5,130,307); rapamycin dialdehydes (U.S. Pat. No. 6,680,330); rapamycin 29-enols (U.S. Pat. No. 6,677,357); O-alkylated rapamycin derivatives (U.S. Pat. No. 6,440,990); water soluble rapamycin esters (U.S. Pat. No. 5,955,457); alkylated rapamycin derivatives (U.S. Pat. No. 5,922,730); rapamycin amidino carbamates (U.S. Pat. No. 5,637,590); biotin esters of rapamycin (U.S. Pat. No. 5,504,091); carbamates of rapamycin (U.S. Pat. No. 5,567,709); rapamycin hydroxyesters (U.S. Pat. No. 5,362,718); rapamycin 42-sulfonates and 42-(N-carbalkoxy)sulfamates (U.S. Pat. No. 5,346,893); rapamycin oxepane isomers (U.S. Pat. No. 5,344,833); imidazolidyl rapamycin derivatives (U.S. Pat. No. 5,310,903); rapamycin alkoxyesters (U.S. Pat. No. 5,233,036); rapamycin pyrazoles (U.S. Pat. No. 5,164,399); acyl derivatives of rapamycin (U.S. Pat. No. 4,316,885); reduction products of rapamycin (U.S. Pat. Nos. 5,102,876 and 5,138,051); rapamycin amide esters (U.S. Pat. No. 5,118,677); rapamycin fluorinated esters (U.S. Pat. No. 5,100,883); rapamycin acetals (U.S. Pat. No. 5,151,413); oxorapamycins (U.S. Pat. No. 6,399,625); and rapamycin silyl ethers (U.S. Pat. No. 5,120,842), each of which is specifically incorporated by reference.
[0030] Other analogs of rapamycin include those described in U.S. Pat. Nos. 7,560,457; 7,538,119; 7,476,678; 7,470,682; 7,455,853; 7,446,111; 7,445,916; 7,282,505; 7,279,562; 7,273,874; 7,268,144; 7,241,771; 7,220,755; 7,160,867; 6,329,386; RE37,421; 6,200,985; 6,015,809; 6,004,973; 5,985,890; 5,955,457; 5,922,730; 5,912,253; 5,780,462; 5,665,772; 5,637,590; 5,567,709; 5,563,145; 5,559,122; 5,559,120; 5,559,119; 5,559,112; 5,550,133; 5,541,192; 5,541,191; 5,532,355; 5,530,121; 5,530,007; 5,525,610; 5,521,194; 5,519,031; 5,516,780; 5,508,399; 5,508,290; 5,508,286; 5,508,285; 5,504,291; 5,504,204; 5,491,231; 5,489,680; 5,489,595; 5,488,054; 5,486,524; 5,486,523; 5,486,522; 5,484,791; 5,484,790; 5,480,989; 5,480,988; 5,463,048; 5,446,048; 5,434,260; 5,411,967; 5,391,730; 5,389,639; 5,385,910; 5,385,909; 5,385,908; 5,378,836; 5,378,696; 5,373,014; 5,362,718; 5,358,944; 5,346,893; 5,344,833; 5,302,584; 5,262,424; 5,262,423; 5,260,300; 5,260,299; 5,233,036; 5,221,740; 5,221,670; 5,202,332; 5,194,447; 5,177,203; 5,169,851; 5,164,399; 5,162,333; 5,151,413; 5,138,051; 5,130,307; 5,120,842; 5,120,727; 5,120,726; 5,120,725; 5,118,678; 5,118,677; 5,100,883; 5,023,264; 5,023,263; 5,023,262; all of which are incorporated herein by reference. Additional rapamycin analogs and derivatives can be found in the following U.S. Patent Application Pub. Nos., all of which are herein specifically incorporated by reference: 20080249123, 20080188511; 20080182867; 20080091008; 20080085880; 20080069797; 20070280992; 20070225313; 20070203172; 20070203171; 20070203170; 20070203169; 20070203168; 20070142423; 20060264453; and 20040010002.
[0031] In another embodiment, the hydrophobic therapeutic agent is provided as a total drug load in the coating layer 20. The total drug load of the hydrophobic therapeutic agent in the coating layer 30 may be from 1 .mu.g to 20 .mu.g, or alternatively from 2 .mu.g to 10 .mu.g, or alternatively from 2 .mu.g to 6 .mu.g, or alternatively from 2.5 .mu.g to 6 .mu.g per square millimeter of the expandable balloon 12. The hydrophobic therapeutic agent may also be uniformly distributed in the coating layer. Additionally, the hydrophobic therapeutic agent may be provided in a variety of physical states. For example, the hydrophobic therapeutic agent may be a molecular distribution, crystal form, or cluster form.
[0032] B. Combination of Additives
[0033] The coating layer 30 also includes a combination of additives. The combination of additives includes a first additive and a second additive. In one embodiment, the first additive is a surfactant and the second additive is a chemical compound having one or more hydroxyl, amino, carbonyl, carboxyl, acid, amide, or ester groups.
1. First Additive
[0034] In one embodiment, the first additive is a surfactant. However, the first additive may also include mixtures of surfactants. Such surfactants may be anionic, cationic, zwitterionic, or non-ionic. Examples of suitable non-ionic surfactants include polyethylene glycol fatty acids, polyethylene glycol fatty acid mono and diesters, polyethylene glycol glycerol fatty acid esters, alcohol-oil transesterification products, polyglyceryl fatty acids, propylene glycol fatty acid esters, sterol and sterol derivatives, polyethylene glycol sorbitan fatty acid esters, polyethylene glycol alkyl ethers, sugar and derivatives thereof, polyethylene glycol alkyl phenols, polyoxyethylene-polyoxypropylene block copolymers, and sorbitan fatty acid esters. Examples of suitable ionic surfactants include quarternary ammonium salts, fatty acid salts, and bile salts.
[0035] With regard to polyethylene glycol (hereinafter "PEG") fatty acids, suitable hydrophilic surfactants include monoesters, esters of lauric acid, oleic acid, and stearic acid. Examples of such surfactants include PEG-8 laurate, PEG-8 oleate, PEG-8 stearate, PEG-9 oleate, PEG-10 laurate, PEG-10 oleate, PEG-12 laurate, PEG-12 oleate, PEG-15 oleate, PEG-20 laurate, and PEG-20 oleate. With regard to PEG fatty acid diesters, suitable hydrophilic surfactants include PEG-20 dilaurate, PEG-20 dioleate, PEG-20 distearate, PEG-32 dilaurate, and PEG-32 dioleate. With regard to PEG fatty acids, suitable hydrophilic surfactants include PEG-20 glyceryl laurate, PEG-30 glyceryl laurate, PEG-40 glyceryl laurate, PEG-20 glyceryl oleate, and PEG-30 glyceryl oleate.
[0036] With regard to alcohol-oil transesterification products, suitable surfactants can be prepared by reaction of alcohol or polyalcohol with a variety of natural and/or hydrogenated oils. Examples of such alcohols include glycerol, propylene glycol, ethylene glycol, PEG, sorbitol, and pentaerythritol. Examples of such oils include castor oil, hydrogenated castor oil, corn oil, olive oil, peanut oil, palm kernel oil, apricot kernel oil, or almond oil. Examples of hydrophilic surfactants prepared by such reaction include PEG-35 castor oil, PEG-40 hydrogenated castor oil, PEG-25 trioleate, PEG-60 corn glycerides, PEG-60 almond oil, PEG-40 palm kernel oil, PEG-50 castor oil, PEG-50 hydrogenated castor oil, PEG-8 caprylic/capric glycerides, and PEG-6 caprylic/capric glycerides. Examples of hydrophobic surfactants prepared by such reaction include PEG-5 hydrogenated castor oil, PEG-7 hydrogenated castor oil, PEG-9 hydrogenated castor oil, PEG-6 corn oil, PEG-6 almond oil, PEG-6 apricot kernel oil, PEG-6 olive oil, PEG-6 peanut oil, PEG-6 hydrogenated palm kernel oil, PEG-6 palm kernel oil, PEG-6 triolein, PEG-8 corn oil, PEG-20 corn glycerides, and PEG-20 almond glycerides.
[0037] With regard to polyglyceryl fatty acids, suitable hydrophobic surfactants include polyglyceryl oleate, polyglyceryl-2 dioleate, polyglyceryl-10 trioleate, polyglyceryl oleate, polyglyceryl-2 diolate, polyglyceryl-10 trioleate, polyglyceryl stearate, polyglyceryl laurate, polyglyceryl myristate, polyglyceryl palmitate, and polyglyceryl linoleate. Suitable hydrophilic surfactants include polyglyceryl-10 laurate, polyglyceryl-10 oleate, and polyglyceryl-10 mono, diolate, polyglyceryl-10 stearate, polyglyceryl-10 linoleate, polyglyceryl-6 stearate, polyglyceryl-6 palmitate, and polyglyceryl-6 linoleate. Polyglyceryl polyricinoleates (i.e. polymuls) are also suitable surfactants.
[0038] With regard to propylene glycol fatty acid esters, suitable hydrophobic surfactants include propylene glycol monolaurate, propylene glycol ricinoleate, propylene glycol monooleate, propylene glycol dicaprylate/dicaprate, and propylene glycol dioctanoate. With regard to sterol and sterol derivatives, suitable surfactants include PEG derivatives such as PEG-24 cholesterol ether.
[0039] With regard to PEG sorbitan fatty acid esters, suitable surfactants include PEG sorbitan monolaurates, PEG sorbitan monopalmitates, PEG sorbitan monooleates, and PEG sorbitan monostearates. Examples of such surfactants include PEG-20 sorbitan monolaurate, PEG-20 sorbitan monopalmitate, PEG-20 sorbitan monooleate, and PEG-20 sorbitan monostearate. With regard to PEG alkyl ethers, suitable surfactants include PEG-3 oleyl ether and PEG-4 lauryl-ether.
[0040] With regard to sugars and derivatives thereof, suitable surfactants include sucrose monopalmitate, sucrose monolaurate, decanoyl-N-methylglucamide, n-decyl-.beta.-D-glucopyranoside, n-decyl-.beta.-D-maltopyranoside, n-dodecyl-.beta.-D-glucopyranoside, n-dodecyl-.beta.-D-maltoside, heptanoyl-N-methylglucamide, n-heptyl-.beta.-D-glucopyranoside, n-heptyl-.beta.-D-thioglucoside, n-hexyl-.beta.-D-glucopyranoside, nonanoyl-N-methylglucamide, n-nonyl-.beta.-D-glucopyranoside, octanoyl-N-methylglucamide, n-octyl-.beta.-D-glucopyranoside, and octyl-.beta.-D-thioglucopyranoside. With regard to PEG alkyl phenols, suitable surfactants include PEG-10-100 nonyl phenol, PEG-15-100 octyl phenol ether, tyloxapol, octoxynol, and nonoxynol.
[0041] With regard to polyoxyethylene-polyoxypropylene (hereinafter "POE-POP") block copolymers, such block copolymers include hydrophilic polyoxyethylene and hydrophobic polyoxypropylene moieties having the formula HO(C.sub.2H.sub.4O).sub.a(C.sub.3H.sub.6O).sub.b(C.sub.2H.sub.4O)- .sub.aH where "a" and "b" denote the number of polyoxyethylene and polyoxypropylene units, respectively. Such polymers are referred to as poloxamers. Suitable hydrophilic poloxamers include Poloxamers 108, 188, 217, 238, 288, 338, and 407. Suitable hydrophobic poloxamers include Poloxamers 124, 182, 183, 212, 313, and 335. With regard to sorbitan fatty acid esters, suitable hydrophobic surfactants include sorbitan monolaurate, sorbitan monopalmitate, sorbitan monooleate, and sorbitan monostearate.
[0042] With regard to ionic surfactants, suitable ionic surfactants include benzalkonium chloride, benzethonium chloride, cetylpyridinium chloride, docecyl trimethyl ammonium bromide, sodium docecylsulfates, dialkyl methylbenzyl ammonium chloride, edrophonium chloride, domiphen bromide, dialkylesters of sodium sulfonsuccinic acid, sodium dioctyl sulfosuccinate, sodium cholate, and sodium taurocholate.
[0043] Such surfactants are stable under heating and are capable of surviving an ethylene oxide sterilization process (which may be employed in preparing the balloon catheters). Moreover, such surfactants do not react with paclitaxel or rapamycin under the sterilization process. Additionally, such surfactants may protect the hydrophobic therapeutic agents from premature release during the device delivery process while facilitating rapid release and elution of the hydrophobic therapeutic agent.
[0044] In one embodiment, the surfactant is a PEG sorbitan monolaurate, a PEG sorbitan monooleate, or a combination thereof. In one particular embodiment, the surfactant is a PEG sorbitan monolaurate, and in a further embodiment, the surfactant is PEG-20 sorbitan monolaurate (i.e. Tween-20). In another particular embodiment, the surfactant is a PEG sorbitan monooleate, and in a further embodiment, the surfactant is PEG-20 sorbitan monooleate (i.e. Tween-80).
2. Second Additive
[0045] In one embodiment, the second additive is a chemical compound having one or more hydroxyl, amino, carbonyl, carboxyl, acid, amide, or ester groups. However, the second additive may also include mixtures of chemical compounds having one or more hydroxyl, amino, carbonyl, carboxyl, acid, amide or ester groups. The chemical compound has at least one hydroxyl group. The hydroxyl group is suitable as the hydrophilic moiety because it is unlikely to react with the hydrophobic therapeutic agent. In one particular embodiment, the chemical compound has a molecular weight less than 5,000-10,000, or alternatively less than 750-1,000, or alternatively less than 750. In a further embodiment, the chemical compound has a molecular weight of greater than 80. In an exemplary embodiment, the chemical compound has a molecular weight less than that of the hydrophobic therapeutic agent.
[0046] In one embodiment, the chemical compound has four or more hydroxyl groups. In one particular embodiment, the chemical compound having more than four hydroxyl groups has a melting point of 120.degree. C. or less. In another particular embodiment, the chemical compound has three adjacent hydroxyl groups that in stereo configuration are all on one side of the molecule. Without being bound by the theory, it is believed that the stereo configuration of the three adjacent hydroxyl groups may enhance drug binding.
[0047] Large molecules diffuse slowly. If the molecular weight of the additive or the chemical compound is high, for example above 800, above 1000, above 1200, above 1500, or above 2000, the chemical compound may elute off of the exterior surface 24 of the expandable balloon 12 too slowly. For example, the hydrophobic therapeutic agent may be released in greater than two minutes. However, if the chemical compound has more than four hydroxyl groups it will have increased hydrophilic properties such that the hydrophobic therapeutic agent may elute off of the exterior surface 24 of the expandable balloon 12 quickly. For example, the hydrophobic therapeutic agent may be released at the target site in less than two minutes. Without being bound by the theory, it is believed that the increased hydrophilicity may play a role in: (1) aiding in the elution of the coating layer 30 off of the exterior surface 24 of the expandable balloon 12; (2) accelerating the release of the hydrophobic therapeutic agent; and (3) improving or facilitating the movement of the hydrophobic therapeutic agent through the water barriers and the polar head groups of lipid bilayers in cells membranes to penetrate tissues.
[0048] Examples of chemical compounds having one or more hydroxyl, amino, carbonyl, carboxyl, acid, amide, or ester moieties include amino alcohols, hydroxyl carboxylic acids, esters, and anhydrides, hydroxyl ketones, hydroxyl lactones, hydroxyl esters, sugar phosphates, sugar sulfate ethyl oxides, ethyl glycols, amino acids and salts thereof, peptides, proteins, sorbitan, glycerol, polyalcohols, phosphates, sulfates, organic acids, esters, salts, vitamins, and combinations of amino alcohols and organic acids.
[0049] With regard to amino acids and salts thereof, suitable chemical compounds include alanine, arginine, asparagines, aspartic acid, cysteine, cystine, glutamic acid, glutamine, glycine, histidine, proline, isoleucine, leucine, lysine, methionine, phenylalanine, serine, threonine, tryptophan, tyrosine, valine, and derivatives thereof. Additionally, amino acid dimers, sugar conjugates, and derivatives thereof may also be suitable chemical compounds. For example, catecholamines such as dopamine, levodopa, carbidoga, and DOPA may also be suitable chemical compounds.
[0050] With regard to peptides, oligopeptides and peptides are suitable as chemical compounds because hydrophobic and hydrophilic amino acids may be coupled to create various sequences of amino acids, facilitating permeation of tissue by the hydrophobic therapeutic agent. With regard to proteins, suitable chemical compounds include albumins, immunoglobulins, caseins, hemoglobins, lysozymes, immunoglobins, a-2-macroglobulin, fibronectins, vitronectins, fibrinogens, and lipases. In an exemplary embodiment, the chemical compound is serum albumin. Such protein is suitable because it is water soluble and contains significant hydrophobic portions to bind the hydrophobic therapeutic agent.
[0051] With regard to vitamins, suitable chemical compounds include fat-soluble vitamins and salts thereof and water-soluble vitamins and amphiphilic derivatives thereof. Suitable examples of fat-soluble vitamins and salts thereof include alpha-tocopherol, beta-tocopherol, gamma-tocopherol, delta-tocopherol, tocopherol acetate, ergosterol, 1-alpha-hydroxycholecal-ciferol, vitamin D2, vitamin D3, alpha-carotene, beta-carotene, gamma-carotene, vitamin A, fursultiamine, methylolriboflavin, octotiamine, prosultiamine, riboflavine, vintiamol, dihydrovitamin K1, menadiol diacetate, menadiol dibutyrate, menadiol disulfate, menadiol, vitamin K1, vitamin K1 oxide, vitamins K2, vitamin K-S(II), and folic acid in free acid form. Suitable examples of water-soluble vitamins and amphiphilic derivatives thereof include acetamine, benfotiamine, pantothenic acid, cetotiamine, cyclothiamine, dexpanthenol, niacinamide, nicotinic acid, pyridoxal 5-phosphate, nicotinamide ascorbate, riboflavin, riboflavin phosphate, thiamine, folic acid, menadiol diphosphate, menadione sodium bisulfite, menadoxime, vitamin B12, vitamin K5, vitamin K6, vitamin K6, and vitamin U.
[0052] With regard to organic acids, esters, and anhydrides, suitable chemical compounds include acetic acid and anhydride, benzoic acid and anhydride, diethylenetriaminepentaacetic acid dianhydride, ethylenediaminetetraacetic dianhydride, maleic acid and anhydride, succinic acid and anhydride, diglycolic anhydride, glutaric anhydride, ascorbic acid, citric acid, tartaric acid, lactic acid, oxalic acid, aspartic acid, nicotinic acid, 2-pyrrolidone-5-carboxylic acid, and 2-pyrrolidone.
[0053] With regard to amino alcohols, alcohols, amines, acids, amides, and hydroxyl acids, suitable chemical compounds include L-ascorbic acid and salts thereof, D-glucoascorbic acid and salts thereof, tromethamine, triethanolamine, diethanolamine, meglumine, glucamine, amine alcohols, glucoheptonic acid, gluconic acid, hydroxyl ketone, hydroxyl lactone, gluconolactone, glucoheptonolactone, glucooctanoic lactone, gluconic acid lactone, mannoic lactone, ribonic acid lactone, lactobionic acid, glucosamine, glutamic acid, benzyl alcohol, benzoic acid, hydroxybenzoic acid, propyl 4-hydroxybenzoate, lysine acetate salt, gentisic acid, lactitol, sorbitol, glucitol, sugar phosphates, glucopyranose phosphate, sugar sulphates, sinapic acid, vanillic acid, vanillin, methyl paraben, propyl paraben, xylitol, 2-ethoxyethanol, sugars, galactose, glucose, ribose, mannose, xylose, sucrose, lactose, maltose, arabinose, lyxose, fructose, cyclodextrin, (2-hydroxypropyl)-cyclodextrin, acetaminophen, ibuprofen, retinoic acid, lysine acetate, gentisic acid, catechin, catechin gallate, tiletamine, ketamine, propofol, lactic acids, acetic acid, salts of any organic acid and amine described above, polyglycidol, glycerols, multiglycerols, galactitol, di(ethylene glycol), tri(ethylene glycol), tetra(ethylene glycol), penta(ethylene glycol), poly(ethylene glycol) oligomers, di(propylene glycol), tri(propylene glycol), tetra(propylene glycol, and penta(propylene glycol), poly(propylene glycol) oligomers, a block copolymer of PEG and polypropylene glycol, derivatives thereof, and combinations thereof.
[0054] Such chemical compounds are stable under heating and are capable of surviving an ethylene oxide sterilization process. Moreover, such chemical compounds do not react with paclitaxel or rapamycin under the sterlization process. In particular, without being bound by the theory, it is believed that hydroxyl, ester, and amide groups are unlikely to react with hydrophobic therapeutic agents such as paclitaxel or rapamycin. However, with specific regard to L-ascorbic acid and salts thereof as well as diethanolamine, such chemical compounds do not necessarily survive such sterilization processes and do react with paclitaxel. Additionally, in some instances, amine and acid groups (e.g., benzoic acid, gentisic acid, diethanolamine, and ascorbic acid) do react with hydrophobic therapeutic agents such as paclitaxel and may not be stable under ethylene oxide sterilization, heating, and aging processes. Accordingly, different sterilization methods should be employed with regard to such specific chemical compounds. Finally, while the chemical compounds described herein rapidly elute the hydrophobic therapeutic agent off of the expandable balloon 12 during deployment at the target site, absorption of the hydrophobic therapeutic agent by tissue is unexpectedly high. Such is particularly apparent with regard to hydroxyl lactones including ribonic acid lactone and gluconolactone.
[0055] In one embodiment, the chemical compound is sorbitol, sorbitan, xylitol, gluconolactone, lactobionic acid, or a combination thereof. In one particular embodiment, the chemical compound is sorbitol, gluconolactone, or combinations thereof. In a further embodiment, the chemical compound is sorbitol. Alternatively, in a further particular embodiment, the chemical compound is gluconolactone.
[0056] Additionally, the surfactants and combinations of surfactants described herein may also be employed in combination with the chemical compounds and combinations of chemical compounds described herein. For example, in one particular embodiment, the coating layer includes a hydrophobic therapeutic agent, a first additive, and a second additive, wherein the hydrophobic therapeutic agent is paclitaxel, the first additive is PEG-20 sorbitan monolaurate, PEG-20 sorbitan monooleate, or combinations thereof, and the second additive is one of sorbitol, gluconolactone, or combinations thereof. In another particular embodiment, the coating layer includes a hydrophobic therapeutic agent, a first additive, and a second additive, wherein the hydrophobic therapeutic agent is paclitaxel, the first additive is PEG-20 sorbitan monolaurate, and the second additive is sorbitol. In yet another particular embodiment, the coating layer includes a hydrophobic therapeutic agent, a first additive, and a second additive, wherein the hydrophobic therapeutic agent is paclitaxel, and the second additive is gluconolactone.
[0057] In one embodiment, the combination of additives in the coating layer 30 is from 1 .mu.g to 20 .mu.g. In another embodiment, the combination of additives and the hydrophobic therapeutic agent are provided in specific weight ratios in the coating layer 30. For example, the ratio by weight of the combination of additives in the coating layer 30 to the hydrophobic therapeutic agent in the coating layer 30 is about 20 to 0.05, or alternatively about 10 to 0.5, or alternatively about 5 to 0.8. Alternatively, in another embodiment, the ratio by weight of the hydrophobic therapeutic agent in the coating layer 30 to the combination of additives is about 0.5 to 3. In yet another embodiment, the balloon catheter 10 is capable of releasing the hydrophobic therapeutic agent and the combination of additives to the tissue in about 0.1 to 30 minutes, or alternatively in about 0.1 to 10 minutes, or alternatively in about 0.2 to 2 minutes, or alternatively in about 0.1 to 1 minute. Such timing of release refers to the timing during which the expandable balloon 12 is inflated and pressing the coating layer 30 into contact with the target tissue.
[0058] C. Solvents
[0059] The hydrophobic therapeutic agent is water insoluble. The combination of additives is soluble in aqueous solvent and polar organic solvent. Such aqueous solvent and polar organic solvents may be employed in preparing the coating layer. Suitable aqueous solvents include water. Suitable polar organic solvents include alkanes, aromatic solvents, alcohols, ethers, esters/acetates, ketones, and mixtures thereof.
[0060] With regard to alkanes, suitable solvents include hexane, octane, cyclohexane, and heptane. With regard to aromatic solvents, suitable solvents include benzene, toluene, and xylene. With regard to alcohols, suitable solvents include methanol, ethanol, propanol, and isopropanol, diethylamide, ethylene glycol monoethyl ether, transcutol, and benzyl alcohol. With regard to ethers, suitable solvents include dioxane, dimethyl ether and tetrahydrofuran. With regard to esters/acetates, suitable solvents include ethyl acetate and isobutyl acetate. With regard to ketones, suitable solvents include acetone, acetonitrile, diethyl ketone, and methyl ethyl ketone. Suitable solvents also include mixtures of aqueous solvents and polar organic solvents. For example, suitable mixtures of aqueous solvents and polar organic solvents may include water and ethanol, water and acetone, and water and methanol, water and tetrahydrofuran.
[0061] Embodiments of the balloon catheter have been described in detail. Further embodiments directed to methods of preparing balloon catheters will now be described.
II. Methods for Preparing Balloon Catheters
[0062] Methods for preparing a balloon catheter 10 may include (1) preparing a coating solution including a solvent, a therapeutic agent, and a combination of additives, (2) loading a metering dispenser with the coating solution, (3) inflating the balloon catheter 10 to 0 to 3 atm, and rotating the balloon catheter 10 about the longitudinal axis of the catheter and/or moving the balloon catheter 10 in a linear direction along the longitudinal or transverse axis of the balloon catheter 10, (4) dispensing the coating solution from the metering dispenser onto an exterior surface 24 of the balloon catheter 10 and flowing the coating solution on the surface of the balloon catheter 10 while the balloon catheter 10 is rotating and/or linearly moving, (5) evaporating the solvent, forming a coating layer 30 on the balloon catheter 10, (6) drying, folding, and wrapping the balloon catheter 10, and (7) sterilizing the balloon catheter 10. In one embodiment, the method for preparing the balloon catheter 10 further includes (8) drying the balloon catheter 10 after sterilization.
[0063] The coating solution includes a solvent, a hydrophobic therapeutic agent, and a combination of additives. In one particular embodiment, the coating solution consists essentially of the solvent, the hydrophobic therapeutic agent, and the combination of additives. The solvent, the hydrophobic therapeutic agent, and the combination of additives are as previously discussed. The content of the hydrophobic therapeutic agent in the coating solution can be from 0.5-50% by weight based on the total weight of the solution. The content of the combination of additives in the coating solution can be from 1-45% by weight, from 1 to 40% by weight, or from 1-15% by weight based on the total weight of the solution. The amount of solvent employed is dependent upon the coating process and viscosity.
[0064] In one particular embodiment, loading the metering dispenser with the coating solution, inflating the balloon catheter 10 to 0 atm to 3 atm, dispensing the coating solution from the metering dispenser, and evaporating the solvent, (as set forth in (2)-(5) above), occur concomitantly. In another embodiment, loading the metering dispenser with the coating solution, inflating the balloon catheter 10 to 0 atm to 3 atm, dispensing the coating solution from the metering dispenser, and evaporating the solvent, (as set forth in (2)-(5) above) are repeated until a therapeutically effective amount of the hydrophobic therapeutic agent in the coating solution is deposited on the exterior surface 24 of the balloon catheter 10.
[0065] A. Preparing a Coating Solution
[0066] With regard to preparing the coating solution, suitable techniques for preparing the coating solution include dispersing, dissolving, diffusing, or otherwise mixing the solvent, the hydrophobic therapeutic agent, and the combination of additives (as previously discussed). In one embodiment, the coating solution is prepared by mixing the solvent, the hydrophobic therapeutic agent, and the combination of additives together simultaneously.
[0067] Alternatively, in another embodiment, the coating solution may be prepared by adding the hydrophobic therapeutic agent and the combination of additives sequentially to the solvent. Such technique of sequentially adding components to the solvent may be based upon solubility of such components and/or other parameters known in the art. For example, the coating solution may be prepared by first adding the hydrophobic therapeutic agent to the solvent and then adding the combination of additives. Alternatively, the combination of additives may be added first to the solvent, after which the hydrophobic therapeutic agent may be added. Adding the combination of additives first to the solvent may be beneficial wherein a hydrophobic therapeutic does not sufficiently dissolve in a solvent (when added prior to the combination of additives). Without being bound by the theory, it is believed that the combination of additives will increase the solubility of the hydrophobic therapeutic agent in the solvent. In some embodiments, preparation of the coating solution may also involve homogenization under high shear conditions and optionally under pressure. In some embodiments, the preparation of the coating solution may also involve filtering the coating solution. For example, in one particular embodiment, the coating solution is prepared by: (1) mixing a fixed amount of the hydrophobic therapeutic agent, the combination of additives, and the solvent, (2) stirring the resulting mixture at room temperature, for example, or with slight heating such as to less than 60.degree. C. until a homogenous solution is obtained, and (3) filtering the solution through a 0.45 .mu.m filter.
[0068] Alternatively, in another embodiment, combinations of two or more solvents may be used, for example, by combining two solvents prior to addition of a hydrophobic therapeutic agent and a combination of additives. As another example, combinations of two or more solvents may be used by adding a hydrophobic therapeutic agent to one solvent and a combination of additives to another solvent and then combining. As still another example, combinations of two or more solvents may be used by adding only one of the hydrophobic therapeutic agent or the combination of additives to one solvent and then adding the second solvent and the remaining hydrophobic therapeutic agent or the combination of additives.
[0069] B. Loading a Metering Dispenser with the Coating Solution
[0070] With regard to loading a metering dispenser with the coating solution, suitable metering dispensers are as disclosed in U.S. Pub. No. 2010/0055294, the disclosure of which is incorporated by reference in its entirety. In one embodiment, the metering dispenser is a portion of an apparatus for coating a balloon catheter 10. The apparatus for coating a balloon catheter 10 may be semi-manual or automated. Briefly, in one embodiment, such apparatus includes an apparatus capable of rotating the balloon catheter 10 around its longitudinal axis and moving the balloon catheter 10 back and forth in the direction of its longitudinal or transverse axis, a controller coordinating the dispenser and the apparatus, and a coating solution storage container.
[0071] In one embodiment, the metering dispenser includes a dispensing tip connected to the metering dispenser for easy coating application. The dispensing tip may include a hub and a tip. In one particular embodiment, the hub is connected to the metering dispenser. The tip may be used to apply coating on the balloon catheter 10 either by contact or non-contact. The tip opening may have different shapes including, but not limited to, circular, oval, square, and rectangular. Additionally, the tip may be straight or with an angle (135.degree., 45.degree. or 90.degree.) and may be rigid or flexible. The tip may be tapered, non-tapered, Teflon-lined, Teflon-coated, and Teflon-lined and crimped or may be a brush. In one embodiment, the dispensing tip is made of metals, metal alloys, and a metal with a polymer coating or lining. For example, in one embodiment, the dispensing tip is made of stainless steel, polyethylene, polypropylene, polyesters, polyamides, polyurethanes, PTFE, metal with a PTFE coating or lining.
[0072] In another embodiment, the dispensing tip has an opening and a flexible tail. The flexible tail may be threaded through the tip opening of the dispensing tip or may be attached to the side of the tip. In embodiments, the flexible tail contacts the expandable balloon 12 to be coated. In some embodiments, the metering dispenser is a syringe, a syringe pump, a metering pipette, or an automatic metering system. In another embodiment, the automatic metering system includes a micro linear pump module, a dispensing controller module, a dispensing tip and other accessories from IVEK Corporation.
[0073] C. Inflating the Balloon Catheter
[0074] With regard to inflating the expandable balloon 12 of the balloon catheter 10, in one embodiment, the expandable balloon 12 is inflated from 0 atm to about 3 atm. Additionally, the balloon catheter 10 may be rotated about the longitudinal axis thereof and/or moved in a linear direction along the longitudinal or transverse axis thereof.
[0075] In one particular embodiment, the apparatus for coating the balloon catheter 10 concurrently rotates the balloon catheter 10 around its longitudinal axis at uniform rotational or tangential speed and translocates the balloon catheter 10 back and forth at uniform frequency in a longitudinal direction. More particularly, in this particular embodiment, the apparatus for coating the balloon catheter 10 moves the balloon catheter 10 linearly back and forth along a rail with uniform frequency while rotating the balloon catheter 10 at uniform rotational/tangential speed.
[0076] In an alternative embodiment, the metering dispenser moves linearly back and forth along a rail with uniform frequency while the apparatus capable of rotating the balloon catheter 10 rotates the balloon catheter 10 at uniform rotational/tangential speed.
[0077] D. Dispensing the Coating Solution from the Metering Dispenser onto an Exterior Surface 24 of the Balloon Catheter 10
[0078] With regard to dispensing the coating solution from the metering dispenser onto an exterior surface 24 of the balloon catheter 10, in one embodiment, the coating solution is dispensed from the metering dispenser while the balloon catheter 10 is rotating and/or linearly moving. During dispensing, the coating solution flows continuously to the exterior surface 24 of the balloon catheter 10 without forming droplets. In one embodiment, the drops of the coating solution move back and forth longitudinally and transversely over the exterior surface 24 of the balloon catheter 10 while the solvent evaporates, resulting in the consistent and uniform deposition of coating solution over the exterior surface 24 of the balloon catheter 10 and resulting in a uniform dry coating layer over the exterior surface 24 of the balloon catheter 10. Without being bound by the theory, it is believed that the rotational and traversal movements allow the flexible tail to break the surface tension between the coating and the expandable balloon 12, forming a uniform coating on the exterior surface 24 of the balloon catheter 10.
[0079] During dispensing, in one embodiment, the balloon catheter 10 and/or the dispensing tip of the metering dispenser move transversely and rotationally. In one particular embodiment, the rotation speed is from 0.1 to 10 revolutions per second, or alternatively from 0.5 to 5 revolutions per second, or alternatively from 0.8 to 2 revolutions per second. The linear or transverse speed is from 0.1 mm per second to 100 mm per second, or alternatively from 1 mm per second to 75 mm per second, or from 2 mm per second to 50 mm per second. The dispensing time is from 2 seconds to 300 seconds, or alternatively from 5 seconds to 120 second. The dispensing time is dependent upon the dispensing coating volume and diameters and lengths of the balloon catheter 10.
[0080] E. Evaporating the Solvent Forming a Coating Layer 30 on the Balloon Catheter 10
[0081] With regard to evaporating the solvent, in one embodiment, the solvent is evaporated while the coating solution is moving at a uniform speed. Without being bound by the theory, it is believed that such technique forms a substantially uniform dry coating layer 30 on the exterior surface 24 of the balloon catheter 10. In one particular embodiment, the apparatus for coating the balloon catheter 10 includes a fan for accelerating solvent evaporation.
[0082] F. Drying, Folding, and Wrapping the Balloon Catheter 10
[0083] With regard to drying, folding, and wrapping the balloon catheter 10, the coating solution may solidify on the exterior surface 24 of the balloon catheter 10 during the transverse and rotational motion of the balloon catheter 10. In one embodiment, the time of solidifying of the coating layer 30 on the expandable balloon 12 after dispensing of the liquid coating is from 0.1 minutes to 10 minutes, or alternatively from 0.5 minutes to 5 minutes. Following solidification, in one embodiment, the coated balloon catheter 10 is dried at room temperature for 12 hours to 24 hours.
[0084] The expandable balloon 12 may be folded after drying. Upon folding, the dried and folded balloon catheter 10 is rewrapped. Additionally, in one particular embodiment, a properly sized balloon protector is placed upon the wrapped balloon catheter 10 and the balloon catheter 10 is packaged.
[0085] G. Sterilizing the Balloon Catheter 10
[0086] With regard to sterilizing the balloon catheter 10, suitable sterilization techniques may include, but should not be limited to, sterilization with ethylene oxide and sterilization with an electron beam. In one particular embodiment, the balloon catheter 10 is sterilized with ethylene oxide.
[0087] H. Drying the Balloon Catheter 10 after Sterilization
[0088] After sterilizing the balloon catheter 10, in one embodiment, the balloon catheter 10 is dried. More particularly, in one embodiment, the balloon catheter 10 is dried under vacuum at about 0.degree. C. to 100.degree. C. for approximately 2 hours to 56 hours. In another embodiment, the balloon catheter 10 is dried under vacuum at about 5.degree. C. to about 45.degree. C. Without being bound by the theory, it is believed that the drying process improves integrity of the coating layer 30, protects loss of coating components during transit through body passages to the target treatment site, and improves drug absorption in the tissue.
[0089] I. Optional Post Treatment with DMSO
[0090] After depositing the coating layer 30 on the balloon catheter 10, in one embodiment, DMSO or other solvent is applied to the surface of the coating layer 20. The DMSO or other solvent may be applied to the surface by dipping, spraying, or any other suitable method known in the art. DMSO readily dissolves hydrophobic therapeutic agents and penetrates the membranes of target cells. DMSO may also enhance tissue absorption. More particularly, without being bound by the theory, it is believed that DMSO displaces water from the lipid head groups and protein domains of the lipid bilayer of the cell membrane of target cells, which indirectly loosens the lipid structure and accelerates absorption and penetration of the hydrophobic therapeutic agent.
[0091] Embodiments of the preparation of balloon catheters have been described in detail. Further embodiments directed to uses of balloon catheters will now be described.
III. Uses of Balloon Catheters
[0092] Uses of the balloon catheters as previously discussed are disclosed. Generally, the balloon catheter 10 is employed to deliver a hydrophobic therapeutic agent to a blood vessel while the balloon catheter 10 is deployed at a target site of the blood vessel. More particularly, the balloon catheter 10 is employed to deliver an anti-proliferative hydrophobic therapeutic agent (such as paclitaxel or rapamycin) to vascular tissue through brief, direct pressure contact at high drug concentration during balloon angioplasty. Such techniques may be employed in treating a total occlusion or a narrowing of passages. Such techniques may also be employed to inhibit hyperplasia and restenosis, as the hydrophobic therapeutic agent may be retained in the target tissue at the delivery site.
[0093] The balloon catheter 10 may also be employed in combination with other methods for treating the vasculature, such as with photodynamic therapy or atherectomy. With regard to photodynamic therapy, photodynamic therapy is a procedure where light or irradiated energy is used to kill target cells in a patient. A light-activated photosensitizing hydrophobic therapeutic agent may be delivered to specific areas of tissue by the balloon catheter 10. A targeted light or radiation source selectively activates the hydrophobic therapeutic agent to produce a cytotoxic response and mediate a therapeutic anti-proliferative effect.
[0094] With regard to atherectomy, atherectomy is a procedure employed to remove plaque from arteries. More specifically, atherectomy removes plaque from peripheral and coronary arteries. The medical device used for peripheral or coronary atherectomy may be a laser catheter, a rotational atherectomy device such as a Rotablator.RTM. (Boston Scientific, Quincy, Mass.), or a direct atherectomy device on the end of a catheter. During atherectomy, a catheter is inserted into the body and advanced through an artery to the area of narrowing. After the atherectomy has removed some of the plaque, balloon angioplasty using the coated balloon catheter 10 may be performed. In addition, stenting may also be performed thereafter, or simultaneous with expansion of the coated expandable balloon 12 as described above.
EXAMPLES
[0095] The following examples include embodiments of balloon catheters and coating layers within the scope of the present disclosure. While the following examples are considered to embody the present disclosure, the examples should not be interpreted as limitations upon the present disclosure.
Example 1
Preparation of Coating Solutions
[0096] Experimental Protocol. Coating solutions for balloon catheters were prepared. More particularly, coating solutions for balloon catheters including Formulations I-XI were prepared. Formulations I-XI are set forth in Table 1 below.
TABLE-US-00001 TABLE 1 FORMU- HYDROPHOBIC LATION THERAPEUTIC NO. AGENT ADDITIVE(S) SOLVENT I Paclitaxel Gluconolactone Acetone, Ethanol, Water II Paclitaxel Tween 20, N- Acetone, Ethanol octanoyl N- methylglucamine III Paclitaxel Tween 20, Acetone, Ethanol, Sorbitol Water IV Paclitaxel Meglumine, Acetone, Ethanol Gensitic Acid V Paclitaxel Lactobionic Acid, Acetone, Ethanol, Diethanolamine Water VI Paclitaxel N-Octanoyl-N- Acetone, Ethanol methylglucamine VII Paclitaxel Meglumine, Acetone, Ethanol, Lactic Acid Water VIII Paclitaxel Gensitic Acid, Acetone, Ethanol, Diethanolamine Water IX Paclitaxel Triton X-100, Acetone, Ethanol N-heptanoyl N- Methylglucamine X Paclitaxel Ultravist 370 Ethanol, Acetone XI Paclitaxel N/A Ethanol, Acetone
[0097] Preparation of Coating Solution Formulations I-XI in Table I is set forth below.
[0098] Formulation I. 50-100 mg (0.06-0.12 mmole) paclitaxel, 1-1.6 ml acetone, 1-1.6 ml ethanol, 0.4-1.0 ml water, and 50-200 mg gluconolactone were mixed.
[0099] Formulation II. 35-70 mg (0.042-0.084 mmole) paclitaxel, 0.5-1.0 ml acetone, 0.5-1.0 ml ethanol, 35-70 mg Tween 20, and 35-70 mg N-octanoyl N-methylglucamine were mixed.
[0100] Formulation III. 35-70 mg (0.042-0.084 mmole) paclitaxel, 0.4-1.0 ml acetone, 0.4-1.0 ml ethanol, 0.2-0.4 ml water, 35-70 mg Tween 20, and 35-70 mg sorbitol were mixed.
[0101] Formulation IV. 40-80 mg (0.048-0.096 mmole) paclitaxel, 0.5-1.0 ml acetone, 0.5-1.0 ml ethanol, 40-80 mg meglumine, and 32-64 mg gensitic acid (equal molar ratio with meglumine) were mixed.
[0102] Formulation V. 35-70 mg (0.042-0.084 mmole) paclitaxel, 0.4-0.8 ml acetone, 0.4-0.8 ml ethanol, 0.25-0.50 ml water, 35-70 mg lactobionic acid, and 10-20 mg diethanolamine (equal molar ratio with lactobionic acid) were mixed.
[0103] Formulation VI. 35-70 mg (0.042-0.084 mmole) paclitaxel, 0.5-1.0 ml acetone, 0.5-1.0 ml ethanol, and 70-140 mg N-octanoyl N-methylglucamine were mixed.
[0104] Formulation VII. 35-70 mg (0.042-0.084 mmole) paclitaxel, 0.4-0.8 ml acetone, 0.4-0.8 ml ethanol, 0.2-0.4 ml water, 35-70 mg meglumine, and 18-36 mg lactic acid (equal molar ratio with meglumine) were mixed.
[0105] Formulation VIII. 50-100 mg (0.06-0.12 mmole) paclitaxel, 0.8-1.6 ml acetone, 0.8-1.6 ml ethanol, 0.4-1.0 ml water, 50-100 mg gensitic acid, and 30-60 mg diethanolamine (equal molar ratio with gensitic acid) were mixed.
[0106] Formulation IX. 35-70 mg (0.042-0.084 mmole) paclitaxel, 0.5-1.0 ml acetone, 0.5-1.0 ml ethanol, 35-70 mg Triton X-100, and 35-70 mg N-heptanoyl N-methylglucamine were mixed.
[0107] Formulation X--Comparison Solution 1. 50 mg (0.06 mmole) paclitaxel, 1 ml ethanol, 0.2 ml acetone, 0.042 ml Ultravist 370 were mixed.
[0108] Formulation XI--Comparison Solution 2. 40 mg (0.048 mmole) paclitaxel, 0.5 ml ethanol, 0.5 ml acetone were mixed.
Example 2
Coating of PTCA Balloon Catheters
[0109] Experimental Protocol. PTCA balloon catheters were coated with the coating solutions including Formulations I-XI prepared in Example 1. More specifically, 6 PTCA balloon catheters (3.5 and 3.0 mm in diameter and 20 mm in length) were inflated at 1-3 atm. The inflated balloon was loaded with one of the coating solutions including Formulations I-XI from Example 1. A sufficient amount of paclitaxel on the balloon (3 microgram per square mm) was obtained. The inflated balloons were folded, and then dried. The coated folded balloon was then rewrapped and sterilized for animal testing.
Example 3
Delivering Paclitaxel from the Coated PTCA Balloon Catheters to Target Sites
[0110] Experimental Protocol. Paclitaxel was delivered to target sites in the coronary vasculature of pigs from the coated PTCA balloon catheters prepared in Example 2. More specifically, the coated PTCA balloon catheters from Example 2 were inserted into a target site in the coronary vasculature (LAD, LCX and RCA) of a 25-45 pound pig. The balloons were inflated to about 12 atm. The overstretch ratio (the ratio of balloon diameter to vessel diameter) was about 1.15-1.20. The paclitaxel delivered into the target tissue during 30-60 seconds of inflation. The balloon catheter was then deflated and withdrawn from the animal body. The target blood vessel was harvested 0.25-24 hours after the procedure. The paclitaxel content in the target tissue and the residual paclitaxel remaining on the balloon were analyzed by tissue extraction and HPLC.
[0111] In some of these animal studies, a stent was crimped on the coated balloon catheters from Example 2 prior to deployment. In chronic animal tests, angiography was performed before and after all interventions and at 28-35 days after the procedure. Luminal diameters were measured and late lumen loss was calculated. Late lumen loss is the difference between the minimal lumen diameter measured after a period of follow-up time (usually weeks to months after an intervention, such as angioplasty and stent placement in the case of this example) and the minimal lumen diameter measured immediately after the intervention. Restenosis may be quantified by the diameter stenosis, which is the difference between the mean lumen diameters at follow-up and immediately after the procedure divided by the mean lumen diameter immediately after the procedure. The animal test results for the Formulations I-XI from Example 1 are reported below.
[0112] Experimental Results. All data is an average of five or six experimental data points.
[0113] Formulation I The paclitaxel content of the Formulation I from Example 1 on the 3.5 mm balloon catheters was 3.26 .mu.g/mm.sup.2. After the procedure, the paclitaxel on the balloon was 15.92 .mu.g, or 2.3% of the total paclitaxel loaded on the balloon. The paclitaxel content in tissue harvested 15-30 minutes after the procedure was 64.79 .mu.g, or 9.2% of the total paclitaxel content originally loaded on the balloon. When an 18 mm stent was deployed by the coated balloon, the residual paclitaxel on the balloon was 31.96 .mu.g, or 4.5% of paclitaxel load, and the paclitaxel content in tissue harvested 15-30 minutes after the procedure was 96.49 .mu.g, or 13.7% of paclitaxel load. The stretch ratio is 1.3 in the procedure. The late lumen loss after 28-35 days was 0.10 (sd 0.2) mm. The diameter stenosis is 3.3%.
[0114] Formulation II. The paclitaxel content of the Formulation II from Example 1 on the 3.5 mm balloon catheters was 3.08 .mu.g/mm.sup.2. After the procedure, the residual paclitaxel on the balloon was 80.58 .mu.g, or 11.4% of the total paclitaxel load. The paclitaxel content in tissue harvested 15-30 minutes after the procedure was 42.23 .mu.g, or 6.0% of the total paclitaxel load. After 28-35 days late lumen loss was 0.30 (sd 0.23) mm. The diameter stenosis was 5.4%.
[0115] Formulation III. The paclitaxel content of the Formulation III from Example 1 on the 3.5 mm balloon catheters was 3.61 .mu.g/mm.sup.2. After the procedure, the residual paclitaxel on the balloon was 174.24 .mu.g, or 24.7% of the total paclitaxel load. The paclitaxel content in tissue harvested 15-30 minutes after the procedure was 83.83 .mu.g, or 11.9% of the total drug load. When deployed with a pre-crimped 18 mm stent, the residual paclitaxel on the balloon is 114.53 .mu.g, or 16.1% of the total paclitaxel load, and the paclitaxel content in tissue harvested 15-30 minutes post procedure was 147.95 .mu.g, or 18.1% of the total paclitaxel load. The stretch ratio was 1.3 in the procedure. The late lumen loss after 28-35 days was 0.10 (sd 0.1) mm. The diameter stenosis was 3.4%.
[0116] Formulation IV. The paclitaxel content of the Formulation IV from Example 1 on the 3.5 mm balloon catheters was 4.71 .mu.g/mm.sup.2. After the procedure, the residual paclitaxel on the balloon was 44.39 .mu.g, or 6.3% of the total paclitaxel load. The paclitaxel content in the tissue harvested 15-30 minutes after the procedure was 77.87 .mu.g, or 11.0% of the total paclitaxel load. After 28-35 days late lumen loss was 0.23 (sd 0.44) mm. The diameter stenosis was 7.3%.
[0117] Formulation V. The paclitaxel content of the Formulation V from Example 1 on the 3.5 mm balloon catheters was 3.85 .mu.g/mm.sup.2. After the procedure, residual paclitaxel on the balloon was 24.59 .mu.g, or 3.5% of the total paclitaxel load. The paclitaxel content in tissue harvested 15-30 minutes after the procedure was 37.97 .mu.g, or 5.4% of the total paclitaxel load. After 28-35 days late lumen loss was 0.33 (sd 0.14) mm. The diameter stenosis was 6.7%.
[0118] Formulation VI. The paclitaxel content of the Formulation VI from Example 1 on the 3.5 mm balloon catheters was 3.75 .mu.g/mm.sup.2. After the procedure, residual paclitaxel on the balloon was 0.82 .mu.g, or 0.1% of the total paclitaxel load. The paclitaxel content in tissue harvested 60 minutes after the procedure was 45.23 .mu.g, or 5.5% of the total paclitaxel load. After 28-35 days late lumen loss was 0.49 (sd 0.26) mm. The diameter stenosis was 11.3%.
[0119] Formulation VII. The paclitaxel content of the Formulation VII from Example 1 on the 3.5 mm balloon catheters was 3.35 .mu.g/mm.sup.2. After the procedure, the residual paclitaxel on the balloon was 62.07 .mu.g, or 7.5% of the total paclitaxel load. The paclitaxel content in tissue harvested 60 minutes after the procedure was 40.55 .mu.g, or 4.9% of the total paclitaxel load. After 28-35 days late lumen loss was 0.47 (sd 0.33) mm. The diameter stenosis was 9.9%.
[0120] Formulation VIII. The paclitaxel content of the Formulation VIII from Example 1 on the 3.5 mm balloon catheters was 3.41 .mu.g/mm.sup.2. After the procedure, residual paclitaxel on the balloon was 50.0 .mu.g, or 6.0% of the total paclitaxel load. The paclitaxel content in tissue harvested 60 minutes post procedure was 26.72 .mu.g, or 3.2% of the total paclitaxel load. After 28-35 days late lumen loss was 0.36 (sd 0.41) mm. The diameter stenosis was 9.3%.
[0121] Formulation IX. The paclitaxel content of the Formulation IX from Example 1 on the 3.5 mm balloon catheters was 3.10 .mu.g/mm.sup.2. After the procedure, residual paclitaxel on the balloon was 1.9% of the total paclitaxel load. The paclitaxel content in tissue harvested 2 hours after the procedure was 34.17 .mu.g, or 5.0% of the total paclitaxel load. In tissue harvested 24 hours after the procedure, the paclitaxel content in tissue was 28.92 .mu.g, or 4.2% of the total paclitaxel load.
[0122] Control Formulation. The drug content of control formulation (uncoated balloon) on the 3.5 mm balloon catheters was 0.0 .mu.g/mm.sup.2. After the procedure, residual drug on the balloon was 0% of the total drug load. The drug content in tissue harvested 15 minutes after the procedure was 0 .mu.g. In tissue harvested 24 hours after the procedure, the drug content in tissue was 0 .mu.g. After 28-35 days late lumen loss was 0.67 (sd 0.27) mm. The diameter stenosis is 20.8%. In the second repeat experiment, the stretch ratio was 1.3. The late lumen loss was 1.1 (sd 0.1). The diameter stenosis was 37.5%.
[0123] Formulation X--Comparison Solution 1. The paclitaxel content of the Formulation X from Example 1 on the 3.5 mm balloon catheters was 3.21 .mu.g/mm.sup.2. After the procedure, residual paclitaxel on the balloon was 13.52 .mu.g, or 1.9% of the total paclitaxel load. The paclitaxel content in the tissue was 28.32 .mu.g, or 4.0% of the total paclitaxel load. When the balloon was deployed with a pre-crimped 18 mm stent, residual paclitaxel on the balloon was 26.45 .mu.g, or 3.7% of the total paclitaxel load. The paclitaxel content in tissue was 113.79 .mu.g, or 16.1% of drug load. After 28-35 days, late lumen loss was 0.27 (sd 0.15) mm. The diameter stenosis was 7.1%.
[0124] Formulation XI--Comparison Solution 2. The paclitaxel content of the Formulation XI (without additive) on the 3.5 mm balloon catheters was 4.22 .mu.g/mm.sup.2. After the procedure, residual paclitaxel on the balloon was 321.97 .mu.g, or 45.6% of the total paclitaxel load. The paclitaxel content in the tissue was 12.83 .mu.g, or 1.8% of the total paclitaxel load.
[0125] Surprisingly, the concentration of paclitaxel absorbed by porcine coronary artery tissue after deployment of balloons coated with the Formulations I-IX from Example 1 according to embodiments of the present disclosure was higher than that delivered by balloons coated with the Formulation X from Example 1 and higher than those coated with paclitaxel alone, the Formulation XI from Example 1. The late lumen loss after 28-35 days follow up was less than the control (uncoated balloon).
* * * * *
D00000

D00001

D00002

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.