Systems And Methods For Biological Metrics Measurement
Ferber; Elad ; et al.
U.S. patent application number 16/989740 was filed with the patent office on 2021-05-20 for systems and methods for biological metrics measurement. The applicant listed for this patent is Spry Health, Inc.. Invention is credited to Andrew DeKelaita, Elad Ferber, Pierre-Jean Julien Ghislain Cobut, Ramkrishnan Narayanan.
| Application Number | 20210145334 16/989740 |
| Document ID | / |
| Family ID | 1000005374063 |
| Filed Date | 2021-05-20 |












View All Diagrams
| United States Patent Application | 20210145334 |
| Kind Code | A1 |
| Ferber; Elad ; et al. | May 20, 2021 |
SYSTEMS AND METHODS FOR BIOLOGICAL METRICS MEASUREMENT
Abstract
A wearable member may include a plurality of energy transmitters that are arranged on a surface of the wearable member, each of the energy transmitters being configured to project energy into tissue of a user. A wearable member may include a plurality of energy receivers each of which is configured to generate a signal based on a received portion of the energy that is projected by one or more of the energy transmitters and reflected by the tissue of the user, wherein at least one of the energy transmitters and the energy receivers are multi-dimensionally arranged on the wearable member such that energy reflected by the tissue of the user at locations that are multi-dimensionally different is incident on the plurality of energy receivers. The processor may be configured to calculate a biological metric based on signals generated by at least part of the plurality of energy receivers.
| Inventors: | Ferber; Elad; (Woodside, CA) ; DeKelaita; Andrew; (Foster City, CA) ; Narayanan; Ramkrishnan; (San Jose, CA) ; Ghislain Cobut; Pierre-Jean Julien; (Menlo Park, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005374063 | ||||||||||
| Appl. No.: | 16/989740 | ||||||||||
| Filed: | August 10, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15717737 | Sep 27, 2017 | 10736552 | ||
| 16989740 | ||||
| 62400456 | Sep 27, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61B 5/0205 20130101; A61B 5/02438 20130101; A61B 2562/043 20130101; A61B 2562/0238 20130101; A61B 5/021 20130101; A61B 5/02433 20130101; A61B 5/0075 20130101; A61B 5/14552 20130101; A61B 5/0816 20130101; A61B 5/14546 20130101; A61B 5/7278 20130101; A61B 5/14535 20130101; A61B 5/14532 20130101 |
| International Class: | A61B 5/1455 20060101 A61B005/1455; A61B 5/00 20060101 A61B005/00; A61B 5/0205 20060101 A61B005/0205; A61B 5/024 20060101 A61B005/024; A61B 5/08 20060101 A61B005/08 |
Claims
1. A system comprising: a wearable member including: a plurality of energy transmitters that are arranged on a surface of the wearable member, each of the energy transmitters being configured to project energy into tissue of a user; and a plurality of energy receivers each of which is configured to generate a signal based on a received portion of the energy that is projected by one or more of the energy transmitters and reflected by the tissue of the user, wherein at least one of the energy transmitters and the energy receivers are multi-dimensionally arranged on the wearable member such that energy reflected by the tissue of the user at locations that are multi-dimensionally different is incident on the plurality of energy receivers; and a processor configured to calculate a biological metric based on signals generated by at least part of the plurality of energy receivers.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] The present application is a continuation of U.S. Nonprovisional patent application Ser. No. 15/717,737, filed Sep. 27, 2017, entitled "SYSTEMS AND METHODS FOR BIOLOGICAL METRICS MEASUREMENT," which claims the benefit of U.S. Provisional Patent Application No. 62/400,456, filed Sep. 27, 2016, entitled "WIDE AREA SWITCHING PHOTOPLETHYSMOGRAM," the contents of which are all incorporated herein by reference.
BACKGROUND
Technical Field
[0002] Embodiments of the present inventions relate generally to blood metrics measurement. More specifically, embodiments of the present inventions relate to non-invasive blood pressure measurement.
Description of Related Art
[0003] Wearable activity monitoring devices are growing in popularity. These devices aim to facilitate achieving a user's goal such as to lose weight, to increase physical activity, or simply to improve overall health. Many such devices may interface with computer software to allow visualization of the recorded data. Nevertheless, most devices are evolved cousins of pedometers, which measure the number of steps a user takes. Even though additional functions such as tallying the distance a user travels or calculating calorie consumptions may be added, these devices lack the ability to measure blood metrics.
[0004] Blood pressure is an important factor in both heart health and overall health. For example, elevated blood pressure may result in coronary artery disease, heart failure and hypertrophy. Accordingly, blood pressure monitoring has become an important component of patient health. Typically, blood pressure is monitored using a blood pressure gauge with an inflatable cuff. However, such devices are often uncomfortable and unable to provide continuous blood pressure measurement.
SUMMARY
[0005] A system comprises a wearable member. The wearable member may include a plurality of energy transmitters that are arranged on a surface of the wearable member, each of the energy transmitters being configured to project energy into tissue of a user. The plurality of energy receivers may each be configured to generate a signal based on a received portion of the energy that is projected by one or more of the energy transmitters and reflected by the tissue of the user, wherein at least one of the energy transmitters and the energy receivers are multi-dimensionally arranged on the wearable member such that energy reflected by the tissue of the user at locations that are multi-dimensionally different is incident on the plurality of energy receivers. The wearable member may comprise a processor configured to calculate a biological metric based on signals generated by at least part of the plurality of energy receivers.
[0006] In some embodiments, the processor is configured to separate one or more source biological signals based on the signals generated by at least part of the plurality of energy receivers, identify at least one artery signal from the one or more source biological signals, and calculate the biological metric based on the at least one artery signal. The processor may be configured to separate two or more source biological signals based on the signals generated by at least part of the plurality of energy receivers, identify two or more artery signals at two different locations of the user from the two or more source biological signals, and calculate a blood pressure of the user as the biological metric based on the two or more artery signals.
[0007] In various embodiments, the number of the energy transmitters is greater than the number of the energy receivers. In some embodiments, the number of the energy transmitters may be the same as the number of the energy receivers. The plurality of energy receivers may include two energy receivers arranged along a first line, and the plurality of energy transmitters may include two energy transmitters arranged along the first line on a first side of the two energy receivers, two energy transmitters arranged along the first line on a second side of the two energy receivers, and two or more energy transmitters arranged along a second line extending along the first line. The plurality of energy receivers may include two groups of two energy receivers arranged along a first line, each group of two energy receivers being disposed between two energy transmitters arranged along the first line, and two groups of two energy receives arranged along a second line extending along the first line, each group of two energy receivers along the second line being disposed between two energy transmitters arranged along the second line.
[0008] In some embodiments, the wearable member is wearable around a wrist of the user, and a multi-dimensional region defined by the plurality of energy transmitters ranges at least 20 mm in a direction of the wearable member corresponding to a circumferential direction of the wrist. The wearable member may be wearable around a wrist of the user, and a multi-dimensional region defined by the plurality of energy transmitters ranges at least 15 mm in a direction of the wearable member corresponding to an extending direction of an arm of the user. Each of the energy transmitters may be configured to project energy at a first wavelength and energy at a second wavelength into the tissue of the user, and each of the energy receivers is configured to generate a first signal based on a first received portion of the energy at the first wavelength and a second signal based on a second received portion of the energy at the second wavelength. The biological metric may be, for example, at least one of a systolic blood pressure, diastolic blood pressure, respiratory rate, blood oxygen, or heart rate.
[0009] An example method may comprise projecting, at a plurality of energy transmitters that are arranged on a surface of a wearable member that is attached to a user, energy into tissue of the user, generating, at a plurality of energy receivers that are arranged on the surface of the wearable member attached to the user, a signal based on a received portion of the energy that is projected by one or more of the energy transmitters and reflected by the tissue of the user, wherein at least one of the energy transmitters and the energy receivers are multi-dimensionally arranged on the wearable member such that energy reflected by the tissue of the user at locations that are multi-dimensionally different is incident on the plurality of energy receivers, and calculating a biological metric based on signals generated by at least part of the plurality of energy receivers.
[0010] An exemplary system comprises an energy transmitter, an energy receiver, and an analyzer. The energy transmitter may project energy at a first wavelength and a second wavelength into tissue of a user, the first wavelength and the second wavelength being associated with at least one nutrient of a set of nutrients in blood of the user. The energy receiver may generate a composite signal based on a fraction of the energy at the first wavelength and the second wavelength, the fraction of the energy being received through the tissue of the user. The analyzer may separate the composite signal into a first signal corresponding to the first wavelength and a second signal corresponding to the second wavelength, and detect, in the blood of the user, a concentration of the at least one nutrient of the set of nutrients based on the first signal and the second signal.
[0011] The fraction of the energy may be received by the energy receiver after the fraction of the energy is reflected by the tissue of the user. The system may comprise a wearable member. The energy transmitter and the energy receiver may be secured to the wearable member such that the energy transmitter and the energy receiver are in contact or in proximity with the tissue. The analyzer may be further configured to determine a set of blood metrics based on the first signal and the second signal, the concentration of at least one nutrient of the set of nutrients being determined based on the determined set of blood metrics. The system may further comprise a user interface configured to display at least some of the set of blood metrics. The analyzer may be further configured to compare a blood metric of the set of blood metric to a threshold and to generate an alert if the blood metric exceeds the threshold. The set of blood metrics may comprise a blood glucose concentration.
[0012] The analyzer may be further configured to determine a first AC component and a first DC component of the first signal, to determine a second AC component and a second DC component of the second signal, wherein the concentration of a nutrient of the set of nutrients is detected based on the first AC component, the first DC component, the second AC component, and the second DC component. The system may further comprise a motion detector configured to measure a level of motion, and the analyzer is configured to compare the level of motion to a threshold and to discount a measurement of the composite signal when the level of motion exceeds the threshold. A nutrient of the set of nutrients may comprise glucose.
[0013] An exemplary method may comprise projecting energy at a first wavelength and a second wavelength into tissue of a user, the first wavelength and the second wavelength being associated with at least one nutrient of a set of nutrients in blood of the user, generating a composite signal based on a fraction of the energy at the first wavelength and the second wavelength, the fraction of the energy being received through the tissue of the user, separating the composite signal into a first signal corresponding to the first wavelength and a second signal corresponding to the second wavelength, and detecting, in the blood of the user, a concentration of the at least one nutrient of the set of nutrients based on the first signal and the second signal.
[0014] Another exemplary system may comprise an energy transmitter, an energy receiver, and an analyzer. The energy transmitter may be configured to project energy at a first wavelength and a second wavelength into tissue of a user, the first wavelength and the second wavelength being associated with, in blood of the user, at least one component. The at least one component being at least one of one of glucose, hemoglobin, triglycerides, cholesterol, bilirubin, protein, albumin, blood pH, Hematocrit, cortisol, and/or electrolytes. The energy receiver may be configured to generate a composite signal based on a fraction of the energy at the first wavelength and the second wavelength, the fraction of the energy being received through the tissue of the user. The analyzer may be configured to separate the composite signal into a first signal corresponding to the first wavelength and a second signal corresponding to the second wavelength, and to detect, in the blood of the user, a concentration of the at least one component based on the first signal and the second signal.
[0015] Other features and aspects of various embodiments will become apparent from the following detailed description, taken in conjunction with the accompanying drawings, which illustrate, by way of example, the features of such embodiments.
[0016] Typically, blood pressure is measured non-invasively with a sphygmomanometer. However, such devices are often uncomfortable and do not permit continuous blood pressure measurement. Some embodiments described herein include systems and methods for non-invasive continuous blood pressure measurement. For example, a blood metrics measurement apparatus may generate multi-channel signals (e.g., PPG signals) which may be provided to a blood pressure calculation system to calculate arterial blood pressure values (e.g., systolic blood value pressure and/or diastolic blood pressure value). More specifically, the blood pressure calculation system (or the blood pressure measurement apparatus) may filter the multi-channel signals (e.g., to remove noise from the signals), select (or, "extract") subsets of "high quality" waves from the multi-channel signals, select (or, "extract") sets of features from each of the high quality waves, and generate sets of feature vectors based on the selected sets of features. In some embodiments, an empirical blood pressure model is used to calculate arterial blood pressure values based on the sets of feature vectors.
[0017] In various embodiments, a system comprises a wearable member and a blood pressure calculation system. The wearable member may include an energy transmitter configured to project energy at a first wavelength and energy at a second wavelength into tissue of a user, and an energy receiver configured to generate a first signal based on a first received portion of the energy at the first wavelength and a second signal based on a second received portion of the energy at the second wavelength, the first received portion of energy and the second received portion of energy each being received through the tissue of the user. The blood pressure calculation system may include a pre-processing module configured to filter noise (e.g., motion related noise) from the first signal and the second signal, and a wave selection module configured to identify a first subset of waves from a first set of waves of the first signal and a second subset of waves from a second set of waves of the second signal, each of the first subset of waves representing a separate approximation of an average of the first set of waves over a predetermined amount of time and each of the second subset of waves representing a separate approximation of an average of the second set of waves over the predetermined amount of time. The blood pressure calculation system may further include a feature extraction module configured to generate a first set of feature vectors and a second set of feature vectors, the first set of feature vectors generated from the first subset of waves, the second set of feature vectors generated from the second subset of waves, wherein each of the feature vectors of the first set of feature vectors and the second set of feature vectors include measurement values and metric values, the measurement values corresponding to amplitude or location points of a particular wave, the metric values generated from metric functions that use at least one of the measurement values. The blood pressure calculation system may additionally include a blood pressure processing module configured to calculate an arterial blood pressure value based on the first set of feature vectors, the second set of feature vectors, and an empirical blood pressure calculation model, the empirical blood pressure calculation model configured to receive the first set of feature vectors and the second set of feature vectors as input values. The blood pressure calculation system may further include a communication module configured to provide a message including or being based on the arterial blood pressure value.
[0018] In some embodiments, the pre-processing module is configured to filter noise from the first signal and second signal using an adaptive filter configured to remove motion noise from the first and second signals.
[0019] In some embodiments, the energy transmitter includes a first light source and a second light source, the first light source configured to project the energy at the first wavelength, the second light source configured to project the energy at the second wavelength.
[0020] In some embodiments, the first light source and the second light source are spaced at a predetermined distance from each other, and each of the first and second light sources are associated with a different corresponding photodiode energy receiver. In related embodiments, the measurement values comprise a transit time determined based on a time for blood to transit the predetermined distance between the first and second light sources.
[0021] In some embodiments, the measurement values include any of wave peak locations or amplitudes, or wave valley locations or amplitudes.
[0022] In some embodiments, the measurement values include any of an associated wave's first or higher order derivative peak locations or amplitudes, the associated wave's first or higher order derivative valley locations or amplitudes, or first or higher order moments of the associated wave.
[0023] In some embodiments, the metric functions include one or more particular metric functions that calculate a distance between two measurement values.
[0024] In some embodiments, the energy projected by the first light source and the energy projected by second light source each have the same wavelength. In related embodiments, the feature extraction module is further configured to determine a phase shift between the first signal and the second signal; calculate, based on the phase shift, any of a pulse wave velocity or a pulse transit time based on the predetermined distance; and the blood pressure calculation module is further configured to calculate the arterial blood pressure value based on first set of feature vectors, the second set of feature vectors, any of the pulse wave velocity or the pulse transit time, the empirical blood pressure calculation model, the empirical blood pressure calculation model further configured to receive the first set of feature vectors, the second set of feature vectors, and any of the pulse wave velocity or the pulse transit time as input.
[0025] In some embodiments, the first signal and the second signal each comprise a photoplethysmogram (PPG) signal
[0026] In various embodiments, a method comprises projecting, at an energy transmitter, energy at a first wavelength and energy at a second wavelength into tissue of a user; generating, at the energy transmitter, a first signal based on a first received portion of the energy at the first wavelength and a second signal based on a second received portion of the energy at the second wavelength, the first received portion of energy and the second received portion of energy each being received through the tissue of the user; filtering, at a blood pressure calculation system, noise from the first signal and second signal; identifying, at the blood pressure calculation system, a first subset of waves from a first set of waves of the first signal and a second subset of waves from a second set of waves of the second signal, each of the first subset of waves representing a separate approximation of an average of the first set of waves over a predetermined amount of time and each of the second subset of waves representing a separate approximation of an average of the second set of waves over the predetermined amount of time; generating, at the blood pressure calculation system, a first set of feature vectors and a second set of feature vectors, the first set of feature vectors generated from the first subset of waves, the second set of feature vectors generated from the second subset of waves, wherein each of the feature vectors of the first set of feature vectors and the second set of feature vectors include measurement values and metric values, the measurement values corresponding to amplitude or location points of a particular wave, the metric values generated from metric functions that use at least one of the measurement values; calculating, at the blood pressure calculation system, an arterial blood pressure value based on the first set of feature vectors, the second set of feature vectors, and an empirical blood pressure calculation model, the empirical blood pressure calculation model configured to receive the first set of feature vectors and the second set of feature vectors as input values; and providing, from the blood pressure calculation system, a message including or being based on the arterial blood pressure value.
[0027] In some embodiments, the filtering noise from the first signal and second signal comprises filtering noise from the first signal and second signal using an adaptive filter configured to remove motion noise from the first signal and the second signal.
[0028] In some embodiments, the energy transmitter includes a first light source and a second light source, the first light source configured to project the energy at the first wavelength, the second light source configured to project the energy at the second wavelength. In related embodiments, the first light source and the second light source are spaced at a predetermined distance from each other, and each of the first and second light sources are associated with a different corresponding photodiode energy receiver.
[0029] In some embodiments, the measurement values comprise a transit time determined based on a time for blood to transit the predetermined distance between the first and second light sources.
[0030] In some embodiments, the measurement values include any of wave peak locations or amplitudes, or wave valley locations or amplitudes.
[0031] In some embodiments, the measurement values include any of an associated wave's first or higher order derivative peak locations or amplitudes, the associated wave's first or higher order derivative valley locations or amplitudes, or first or higher order moments of the associated wave.
[0032] In some embodiments, the metric functions include one or more particular metric functions that calculate a distance between two measurement values.
[0033] In some embodiments, the energy projected by the first light source and the energy projected by second light source each have the same wavelength. In related embodiments, the feature extraction module is further configured to determine a phase shift between the first signal and the second signal; calculate, based on the phase shift, any of a pulse wave velocity or a pulse transit time based on the predetermined distance; and the blood pressure calculation module is further configured to calculate the arterial blood pressure value based on first set of feature vectors, the second set of feature vectors, any of the pulse wave velocity or the pulse transit time, the empirical blood pressure calculation model, the empirical blood pressure calculation model further configured to receive the first set of feature vectors, the second set of feature vectors, and any of the pulse wave velocity or the pulse transit time as input.
[0034] In various embodiments, a system comprises a communication interface configured to receive a first signal and a second signal, the first signal being based on a first received portion of energy having been previously projected at a first wavelength into tissue of a user, the second signal being based on a second received portion of energy having been previously projected at a second wavelength into the tissue of the user; a pre-processing module configured to remove noise from the first signal and the second signal; a wave selection module configured to identify a first subset of waves from the first set of waves of a first signal and a second subset of waves from a second set of waves of the second signal, each of the first subset of waves representing a separate approximation of an average of the first set of waves over a predetermined amount of time and each of the second subset of waves representing a separate approximation of an average of the second set of waves over the predetermined amount of time; a feature extraction module configured to generate a first set of feature vectors and a second set of feature vectors, the first set of feature vectors generated from the first subset of waves, the second set of feature vectors generated from the second subset of waves, wherein each of the feature vectors of the first set of feature vectors and the second set of feature vectors include measurement values and metric values, the measurement values corresponding to amplitude or location points of a particular wave, the metric values generated from metric functions that use at least one measurement value; a blood pressure processing module configured to calculate an arterial blood pressure value based on the first set of feature vectors, the second set of feature vectors, and an empirical blood pressure calculation model, the empirical blood pressure calculation model configured to receive the first set of feature vectors and the second set of feature vectors as input values; and a communication module configured to provide a message including or being based on the arterial blood pressure value.
[0035] In various embodiments, a system comprises a processor; and memory storing instructions that, when executed by the processor, cause the processor to: receive a first signal and a second signal, the first signal being based on a first received portion of energy having been previously projected at a first wavelength into tissue of a user, the second signal being based on a second received portion of energy having been previously projected at a second wavelength into the tissue of the user; filter noise (e.g., motion related noise) from the first signal and the second signal; identify a first subset of waves from a first set of waves of the first signal and a second subset of waves from a second set of waves of the second signal, each of the first subset of waves representing a separate approximation of an average of the first set of waves over a predetermined amount of time and each of the second subset of waves representing a separate approximation of an average of the second set of waves over the predetermined amount of time; generate a first set of feature vectors and a second set of feature vectors, the first set of feature vectors generated from the first subset of waves, the second set of feature vectors generated from the second subset of waves, wherein each of the feature vectors of the first set of feature vectors and the second set of feature vectors include measurement values and metric values, the measurement values corresponding to amplitude or location points of a particular wave, the metric values generated from metric functions that use at least one of the measurement values; calculate an arterial blood pressure value based on the first set of feature vectors, the second set of feature vectors, and an empirical blood pressure calculation model, the empirical blood pressure calculation model configured to receive the first set of feature vectors and the second set of feature vectors as input values; and provide a message including or being based on the arterial blood pressure value.
BRIEF DESCRIPTION OF THE DRAWINGS
[0036] FIG. 1 is a block diagram illustrating an example environment utilizing a multispectral blood metrics measurement apparatus in accordance with various embodiments.
[0037] FIG. 2 is a block diagram illustrating an exemplary multispectral blood metrics measurement apparatus, such as the multispectral blood metrics measurement apparatus illustrated in FIG. 1.
[0038] FIG. 3 illustrates an exemplary flow diagram of a method of measuring blood metrics in accordance with an embodiment of the present application.
[0039] FIG. 4 illustrates an exemplary apparatus for measuring various blood metrics in accordance with an embodiment of the present application.
[0040] FIG. 5 illustrates a display of an assessment of a current health index derived from data collected from or with a multispectral blood metrics measurement apparatus in various embodiments.
[0041] FIG. 6 illustrates a display of an assessment of an overall health index, derived from data collected from or with a multispectral blood metrics measurement apparatus in various embodiments.
[0042] FIG. 7 illustrates a display of an assessment of an overall health index, derived from data collected from or with a multispectral blood metrics measurement apparatus in various embodiments.
[0043] FIG. 8 is a block diagram illustrating an exemplary digital device that can be utilized in the implementation of various embodiments.
[0044] FIG. 9 depicts a block diagram of a system and environment for non-invasive blood pressure measurement according to some embodiments.
[0045] FIG. 10A depicts a block diagram of a blood metrics measurement apparatus according to some embodiments.
[0046] FIG. 10B depicts a block diagram of a sensor system according to some embodiments.
[0047] FIG. 11 depicts a flowchart of an example method of operation of a blood metrics measurement apparatus according to some embodiments.
[0048] FIG. 12 depicts a block diagram of a user device according to some embodiments.
[0049] FIG. 13 depicts a flowchart of an example method of operation of a user device according to some embodiments.
[0050] FIG. 14 depicts a block diagram of a blood pressure calculation system according to some embodiments.
[0051] FIG. 15A depicts a flowchart of an example method of operation of a blood pressure calculation system according to some embodiments.
[0052] FIG. 15B depicts a flowchart of an example method of pre-processing (or, "filtering") a signal according to some embodiments.
[0053] FIG. 15C depicts a blocks diagram of an example pre-processing system for filtering signals according to some embodiments.
[0054] FIG. 16A depicts a flowchart of an example method for extracting high quality waves according to some embodiments.
[0055] FIG. 16B depicts a flowchart of an example method for extracting high quality waves using a Gaussian mixture model according to some embodiments.
[0056] FIG. 16C depicts a flowchart of an example method of method for extracting high quality waves using a group similarity model according to some embodiments.
[0057] FIG. 17 depicts a block diagram of a blood metrics server according to some embodiments.
[0058] FIGS. 18-20 depict flowcharts of example methods of operation of a blood metrics server according to some embodiments.
[0059] FIG. 21 depicts an example noisy PPG signal and an example filtered PPG signal according to some embodiments.
[0060] FIG. 22 depicts an example set of waves of a PPG signal and an example high quality wave selected from the set of waves according to some embodiments.
[0061] FIG. 23 depicts example feature points of a wave according to some embodiments.
[0062] FIG. 24 depicts an example feature vector according to some embodiments.
[0063] FIGS. 25A-C show an example selected high quality wave, the first derivative of the selected high quality wave, and the second derivative of the selected high quality wave according to some embodiments.
[0064] FIG. 26 depicts example tree structures of an example empirical blood pressure calculation model according to some embodiments.
[0065] FIG. 27 depicts an example bi-Gaussian mixture model for a PPG signal according to some embodiments.
[0066] FIG. 28 depicts an example PPG signal including multiple reflections according to some embodiments.
[0067] FIG. 29A-C depict block diagram of example sensor systems according to some embodiments.
[0068] FIG. 30A-E depict arrangement of energy transmitters and energy receivers in a sensor system according to some embodiments.
[0069] FIG. 31A-B depict experimentations carried out to obtain an optimum location of a sensor system, when the sensor system is used for measuring states of a radial artery on a left wrist of a subject.
[0070] FIG. 32 depicts a flowchart of an example method of method for separating signals from different sources according to some embodiments.
[0071] FIG. 33 depicts a flowchart of an example method of calculating blood pressure according to some embodiments.
DETAILED DESCRIPTION
[0072] Biometrics including blood metrics may be measured by minimally invasive procedures to address medical conditions such as diabetes or in the diagnosis and discovery of diseases. Minimal-invasive procedure based devices may have the advantages of reducing costs and decreasing the need for invasive methods, thereby increasing the comfort and well-being of users and patients. Even though these devices have revolutionized patient care, they have only been described in, and approved for, medical purposes. Minimal-invasive procedure based devices are usually out of reach for the general public because they are designed for medical uses rather than non-medical purposes such as fitness, well-being, and quality of life.
[0073] Personal devices such as sphygmomanometers or pulse oximeters measure blood pressure or oxygen levels, respectively, on a per-request basis. They usually cannot measure blood metrics real time or periodically. Real-time blood metrics data (e.g., high resolution measurements, or measurements over long periods of time) may allow these devices to facilitate users monitoring and controlling their energy levels and/or metabolism. Nutritionists, people suffering from obesity, people desiring to eat healthier, fitness enthusiasts, semi-professional athletes, people likely to have hypoglycemia, or the vast majority of the general population can benefit from these devices.
[0074] In various embodiments, a multispectral blood metric measurement apparatus monitors blood metrics, fitness, and/or metabolism levels of various users in a non-invasive manner. The multispectral blood metric measurement apparatus may be, for example, wearable technology. The multispectral blood metric measurement apparatus may measure any number of blood metrics. Blood metrics may include, for example, various nutrient blood concentrations. Blood metrics may be, for example, monitored, stored, tracked, and/or analyzed.
[0075] FIG. 1 is a block diagram illustrating an example environment 100 utilizing a multispectral blood metrics measurement apparatus 102 in accordance with various embodiments. As shown in FIG. 1, the example environment 100 may comprise a multispectral blood metrics measurement apparatus 102, one or more user systems 104, an optional analysis system 108, and a computer network 106 communicatively coupling together each of the multispectral blood metrics measurement apparatus 102, one or more user devices 110, 112, and 114 (depicted as user system 104), and/or the analysis system 108. As shown, a user system 104 may include a smartphone 110 (e.g., iPhone.RTM.), a computer 112 (e.g., a personal computer), and/or a tablet 114 (e.g., iPad.RTM.), through the computer network 106 (e.g., a Bluetooth.RTM. 4.0 personal area network), can either interact directly or indirectly with the blood metrics measurement apparatus 102.
[0076] The multispectral blood metrics measurement apparatus 102 may measure health or metabolism predictors non-invasively. The multispectral blood metrics measurement apparatus 102 may measure blood metrics such as concentrations of various nutrients over time, deliver energy into tissues of various body parts of a user, track a user's behavior pattern, detect motion, communicate various blood metric measurements, and/or receive a user's instructions. For instance, through the computer network 106, the multispectral blood metrics measurement apparatus 102 may transmit one or more blood metric measurements to, or receive instructions from, the user system 104 or the multispectral blood measurement system 108 such as which health or metabolism predictor to measure.
[0077] In some embodiments, the multispectral blood metric 102 measurement apparatus may project energy into tissue of a user and detect energy reflected from and/or transmitted through tissue of the user (e.g., the wearer of the multispectral blood metric measurement apparatus 102). The projected energy may be at multiple wavelengths that are associated with the blood metrics of interest to a user. The detected energy may be a fraction of the energy that is projected into the tissue. Energy at different wavelengths may be absorbed at a different rate that is related to a user's body state. The user's body state (e.g., heart rate, blood pressure, nutrient level, or the like) determines the amount of absorbed energy. Accordingly, energy at different wavelengths may be absorbed at different levels by a user's body. The fraction of energy received (e.g., that is reflected by the tissue or transmitted through the tissue) may be used to generate signals (e.g., composite signals) at different levels. These signals may provide information of the user's body state. This information may be obtained by analyzing waveforms of the signal in the time domain and/or the frequency domain.
[0078] In various embodiments, the multispectral blood metric measurement apparatus 102 may measure many metrics, including, but not limited to, skin conductivity, pulse, oxygen blood levels, blood pressure, blood glucose level, glycemic index, insulin index, Vvo2max, fat body composition, protein body composition, blood nutrient level (e.g., iron), body temperature, blood sodium levels, and/or naturally-produced chemical compound level (e.g., lactic acid). Nutrients may be determined based on the blood metrics to be measured. Nutrients may be measured may include, but are not limited to, glucose, hemoglobin, triglycerides, cholesterol, bilirubin, protein, albumin (i.e., egg white), and/or electrolytes (e.g., sodium, potassium, chloride, bicarbonate, etc.)
[0079] It will be appreciated that the user's body state may change dynamically and energy at a wavelength may be absorbed differently by a user over the time. By monitoring and tracking detected energy from the user's body, a user's health or condition may be more tracked. Systems and methods described herein may monitor and store blood metrics including concentrations of various nutrients. A user's history health records may be generated by using blood metrics measured at different times. In some embodiments, blood metrics measured a given time point may be compared to the history health records to detect any abnormal health conditions. The multispectral blood metric measurement apparatus may comprise a user interface where a user may input blood metrics of interest, be presented with various health reports, and/or be alerted with abnormal health conditions.
[0080] A user may comfortably wear a multispectral blood metric measurement apparatus 102 over time. The multispectral blood metric measurement apparatus 102 may comprise lightweight components. The multispectral blood metric measurement apparatus 102 may be made of hypoallergenic materials. The multispectral blood metric measurement apparatus 102 may be flexibly built so that it could fit various body parts (e.g., wrist, earlobe, ankle, or chest) of a user.
[0081] In accordance with some embodiments, the computer network 106 may be implemented or facilitated using one or more local or wide-area communications networks, such as the Internet, WiFi networks, WiMax networks, private networks, public networks, personal area networks ("PAN"), and the like. In some embodiments, the computer network 106 may be a wired network, such as a twisted pair wire system, a coaxial cable system, a fiber optic cable system, an Ethernet cable system, a wired PAN constructed with USB and/or FireWire connections, or other similar communication network. Alternatively, the computer network 106 may be a wireless network, such as a wireless personal area network, a wireless local area network, a cellular network, or other similar communication network. Depending on the embodiment, some or all of the communication connections with the computer network 106 may utilize encryption (e.g., Secure Sockets Layer [SSL]) to secure information being transferred between the various entities shown in the example environment 100.
[0082] Although FIG. 1 depicts a computer network 106 supporting communication between different digital devices, it will be appreciated that the multispectral blood metrics measurement apparatus may be directly coupled (e.g., over a cable) with any or all of the user devices 110, 112, and 114.
[0083] The user devices 110-114 may include any digital device capable of executing an application related to measuring blood metrics, presenting an application user interface through a display and/or communicating with various entities in the example environment 100 through the computer network 106. For instance, through the computer network 106, the user device 110 may receive one or more blood metric measurements from the multispectral blood metrics measurement apparatus 102, track and store the blood metric measurements, analyze the blood metric measurements, and/or provide recommendations based on the blood metric measurements. An application user interface may facilitate interaction between a user of the user system 104 and an application running on the user system 104.
[0084] In various embodiments, any of user devices 110-114 may perform analysis of the measurements from the multispectral blood metrics measurement apparatus 102, display results, provide reports, display progress, display historic readings, track measurements, track analysis, provide alerts, and/or the like.
[0085] The analysis system 108 may be any form of digital device capable of executing an analysis application for analyzing and/or measuring blood metrics. In some embodiments, the analysis system 108 may generate reports or generate alerts based on analysis or measurement of blood metrics. For instance, through the computer network 106, the analysis system 108 may receive one or more blood metric measurements from the multispectral blood metrics measurement apparatus 102, track and store blood metric measurements, analyze blood metric measurements, and/or provide recommendations based on the analysis. An application programming interface may facilitate interaction between a user, the user devices 110-114, and/or the multispectral blood metrics measurement apparatus 110 with the analysis system 108.
[0086] Computing devices (e.g., digital devices) may include a mobile phone, a tablet computing device, a laptop, a desktop computer, personal digital assistant, a portable gaming unit, a wired gaming unit, a thin client, a set-top box, a portable multi-media player, or any other type of network accessible user device known to those of skill in the art. Further, the analysis system 108 may comprise of one or more servers, which may be operating on or implemented using one or more cloud-based services (e.g., System-as-a-Service [SaaS], Platform-as-a-Service [PaaS], or Infrastructure-as-a-Service [IaaS]).
[0087] It will be understood that for some embodiments, the components or the arrangement of components may differ from what is depicted in FIG. 1.
[0088] Each of the multispectral blood metrics measurement apparatus 102, one or more user devices 110, 112, and 114, and the analysis system 108 may be implemented using one or more digital devices. An exemplary digital device is described regarding FIG. 8.
[0089] FIG. 2 is a block diagram illustrating an exemplary multispectral blood metrics measurement apparatus 200, such as the multispectral blood metrics measurement apparatus 102 illustrated in FIG. 1. The multispectral blood metrics measurement apparatus 200 comprises an analyzer 202, an energy transmitter 204, and an energy receiver 206. Various embodiments may comprise a wearable member. The wearable member may include, for example, a bracelet, glasses, necklace, ring, anklet, belt, broach, jewelry, clothing, or any other member of combination of members that allow the multispectral blood metrics measurement apparatus 200 to be close to or touch a body of the wearer.
[0090] The energy transmitter 204 and the energy receiver 206 may be secured to the wearable member such that the energy transmitter and the energy receiver may make contact or be in proximity with tissues (e.g., skin) of a user. The analyzer 202 may be coupled to the energy transmitter 204 and the energy receiver 206. In further embodiments, the multispectral blood metrics measurement apparatus 200 may comprise a communication module (not shown). The communication module may be coupled to the analyzer 202. The blood metrics measurement apparatus 200 may further comprise a driver (not shown) and a power source (not shown). The driver may be coupled to the energy transmitter 204 and the analyzer 202. The analyzer 202 may be coupled to the energy transmitter 204 via the driver. The power source may be coupled to the energy transmitter 204 via the driver. The blood metrics measurement apparatus 200 may further comprise an Analog-to-Digital Converter ("ADC") (not shown). The ADC may be coupled to the energy receiver 206 and the analyzer 202. In some embodiments, the blood metrics measurement apparatus 200 may comprise a motion sensor (e.g., an accelerometer, gyroscope, global positioning system, or the like) (not shown). The motion sensor may be coupled to the analyzer 202.
[0091] In various embodiments, the energy transmitter 204 emits energy including, but not limited to, light, into the body of the user. The energy produced by the energy transmitter may be in the direction of entering tissues. For example, the energy produced by the energy transmitter 204 is in a direction 251 entering the tissue 210. In some embodiments, the energy transmitter 204 emits energy or light at different wavelengths. The energy transmitter 204 may comprise any number of light emission diodes ("LEDs"). In some embodiments, the energy transmitter 204 comprises at least two LEDs. Each LED may be configured to emit energy at one or more wavelengths. In another example, each LED may emit light with a peak wavelength centered around a wavelength. In one example, the energy transmitter 204 may emit light with a peak wavelength centered around 500 nm to 1800 nm.
[0092] Each wavelength may correspond to one or more blood metrics of interest and/or one or more nutrients. It will be appreciated that different components of the blood and/or different nutrients may absorb energy at different wavelengths. In various embodiments, a controller, driver, analyzer 202, or the like may receive a blood metric or nutrient of interest (e.g., from a user of the multispectral blood metrics measurement apparatus 200 and/or a user device not shown). The controller, driver, analyzer 202 or the like may associate the blood metric and/or nutrient of interest with one or more wavelengths and configure one or more of the LEDs to emit energy of at least one of the one or more wavelengths. For example, the analyzer 202 may command the driver to deliver electric power to one LED that is configured to emit light at the desired wavelength.
[0093] The energy receiver 206 may detect energy associated with the energy provided by the LEDs from tissues (e.g., skin) of the user. In this example, received and/or detected energy is in the direction 252 that leaves from the tissue 210. In various embodiments, the energy receiver 206 may detect energy from the body of the user that is a fraction of the energy produced by the energy transmitter 204.
[0094] The energy transmitter 204 and the energy receiver 206 may be configured such that the energy receiver 206 detects reflected energy from tissues of the user of the multispectral blood metrics measurement apparatus 200. For example, the energy transmitter 204 and the energy receiver 206 may be configured to be disposed on one surface or side of a user's tissue. The energy transmitter 204 and the energy receiver 206 may be configured such that the energy receiver 206 detects energy from the energy transmitter 204 that passes through or reflects from the user's tissues. In some embodiments, the energy transmitter 204 and the energy receiver 206 may be configured to be disposed on different (e.g., opposite) surfaces or sides of a users' tissue.
[0095] Energy detected from tissues of a user may be detected by the energy receiver 206. The energy receiver 206 may be configured to generate a signal in response to the detected energy. In some embodiments, the energy receiver 206 may be triggered by the energy received to generate an output which may be dependent or partially dependent upon the amount of energy received. The energy receiver 206 may be configured to generate a signal (e.g., an electric current, or an electric voltage) in response to the energy received from the tissues.
[0096] The signal generated by the energy receiver 206 may be associated with one or more blood metrics and/or nutrients of interest. Energy at different wavelengths may be absorbed at a different rate that is related to a user's body state. The user's body state (e.g., heart rate, blood pressure, nutrient level, or the like) may determine the amount of energy absorbed by the body. Accordingly, energy from the user's body at different wavelengths may be detected at different levels thereby causing different responses of the energy receiver 206. The energy receiver 206 may, for example, output signals based on the level of the energy received.
[0097] The energy receiver 206 may provide information associated with the user's body state. Blood metric information may be determined (e.g., by the analyzer 202) from the output signal of the energy receiver 206.
[0098] The energy receiver 206 may comprise a set of photodetectors (e.g., a photo diode, or a photo transistor) which are configured to output a signal dependent upon photons or the like from the energy transmitter 204 that passed through tissues of the user. As discussed herein, in some embodiments, the multispectral blood metrics measurement apparatus 200 may also include a pressure sensor. The pressure sensor may be configured to generate, detect, and/or measure non-optical signals. For example, the pressure sensor may non-invasively and continuously generate, detect, and/or measure pressure pulse signals. In some embodiments, the pressure sensor measures pressure pulse waveforms associated with arterial pressure of one or more arteries of a user. For example, multispectral blood metrics measurement apparatus 200 may include the energy transmitter 204 to generate optical signals and the energy receiver 206 to receive optical signals but not the pressure sensor. Alternately, the multispectral blood metrics measurement apparatus 200 may include the pressure sensor that may produce pressure on the user's body and/or receive measurements based on that pressure but not the energy transmitter 204 or the energy receiver 206 to receive optical signals. In this example, the pressure sensor may be considered to be an energy receiver and the energy transmitter is the body of the wearer.
[0099] In various embodiments, the output signal of the energy receiver 206 is a composite of multiple signals. Each signal of the composite may be associated with energy at a wavelength which may be a portion (or fraction) of the total energy emitted by the energy transmitter 204.
[0100] The energy transmitter 204 may be configured to generate energy at a set of wavelengths. In some embodiments, the energy transmitter 204 is configured to generate energy such that energy at different wavelengths is generated sequentially and/or periodically. The energy transmitter 204 may be configured to generate energy at each particular wavelength until energy at all wavelengths of the set is generated. The period of time for the energy transmitter 204 to generate energy at all wavelengths is a generation period. Subsequent to completion of the generation period, the energy transmitter 204 may start a new generation period thereby allowing multiple measurements.
[0101] In some embodiments, the blood metrics measurement apparatus 200 may be or include the blood metrics measurement apparatus 102 described with regard to FIG. 1.
[0102] FIG. 3 illustrates an exemplary flow diagram of a method 300 of measuring blood metrics in accordance with an embodiment of the present application. At step 302, energy transmitter 204 generates and delivers energy at different wavelengths into tissues (e.g., skin) of a user. Different wavelengths may be associated with any number of nutrients, which may be associated with the blood metrics to be measured.
[0103] In some embodiments, a user may define various blood metrics and/or nutrients to be measured. Referring back to FIG. 1, a list of blood metrics and/or nutrients may be selected from a user interface (e.g., displayed on an interface of the multispectral blood metrics measurement apparatus 102, on a user device 110-114, or through the analysis system 108). The user may select one or more blood metrics and/or nutrients to be measured.
[0104] In some embodiments, a user may define a set of blood metrics to be measured on the user system 104; the multispectral blood metrics measurement apparatus 102 may provide the blood metrics to be measured to the user system 104. For example, on any device of the user system 104, a user may define one or more blood metrics by selecting one or more blood metrics from a list of blood metrics provided, for example, via the user interface.
[0105] As discussed herein, the multispectral blood metrics measurement apparatus 200 may measure, but is not limited to, skin conductivity, pulse, oxygen blood levels, blood pressure, blood glucose level, glycemic index, insulin index, Vvo2max, fat body composition, protein body composition, blood nutrient level (e.g., iron), body temperature, blood sodium levels, or naturally-produced chemical compound level (e.g., lactic acid). Nutrients may be determined based on the blood metrics to be measured. The multispectral blood metrics measurement apparatus 200 may measure nutrients, but is not limited to, glucose, hemoglobin, triglycerides, cholesterol, bilirubin, protein, albumin (i.e., egg white), or electrolytes (e.g., sodium, potassium, chloride, bicarbonate, or the like). The multispectral blood metrics measurement apparatus 200 may also measure oxygen, cortisol, and Hematocrit, for example (e.g., blood components).
[0106] In various embodiments, one or more wavelengths may be associated with a nutrient or a combination of blood components or molecules. In some embodiments, a number of wavelengths generated by the energy transmitter 204 are the number of blood components or molecules to be measured plus one. For example, when a total number of five (5) blood components and/or molecules are to be measured, a total number of six (6) wavelengths may be determined based on the blood components and/or molecules to be measured. Similarly, it will be appreciated that one or more wavelengths may be associated with a nutrient or a combination of nutrients. In some embodiments, a number of wavelengths generated by the energy transmitter 204 are the number of nutrients to be measured plus one. For example, when a total number of three (3) nutrients are to be measured, a total number of four (4) wavelengths may be determined based on the nutrients to be measured.
[0107] In some embodiments, the multispectral blood metrics measurement apparatus 200, user devices 110-114, and/or analysis system 108 may comprise a reference table of blood components, molecules, and/or nutrients and wavelengths corresponding to the blood components, molecules, and/or nutrients. A wavelength may be unique to or more generally associated with a nutrient. A reference wavelength may be unique to or more generally associated with a combination of nutrients to be measured. As such, wavelength(s) may be determined by looking up each blood components, molecules, and/or nutrients that is to be measured. Energy at the determined wavelengths may be transmitted by the energy transmitter 204 into the body.
[0108] In various embodiments, in a predetermined time duration, energy at all desired wavelengths may be generated. For each wavelength, the corresponding energy may be generated for a time period equal to a predetermined time duration divided by the number of wavelengths. For example, four (4) wavelengths may be determined and the predetermined time duration is two (2) seconds. Accordingly, energy for each wavelength may be generated for a duration of half (0.5) second.
[0109] At step 304, the energy receiver 206 detects a fraction of the energy transmitted into the user's tissue by the energy transmitter 204. The energy receiver 206 may generate a signal based on the fraction of energy detected (e.g., based on the amount of the energy detected). In one example, energy detected at step 304 may be a fraction of the energy generated at step 302 reflected by the tissue. Energy detected at step 302 may be a fraction of the energy generated at step 302 that passes through the tissue (e.g., other undetected energy may be absorbed by tissue and/or otherwise blocked). The output signal of the energy receiver 206 may be an electric current or an electric voltage, of which the amplitude may be related to the amount of the energy detected. In various embodiments, steps 302 and 304 are performed simultaneously. That is, energy generation and detection may be performed approximately simultaneously.
[0110] In various embodiments, the output signal generated by the energy receiver 206 is a composite signal of multiple signals, each of which corresponds to one or more wavelengths. The output signal produced at step 306 may be divided into individual signals, each of which is may be associated with one or more wavelengths.
[0111] In various embodiments, analysis of the signals from the energy receiver 206 may identify abnormal measurements. For example, each of the measurement may be compared to a predetermined value. If the difference between the measurement and the predetermined value is above (or below) a threshold, then the measurement may be determined to be abnormal. An abnormal value may trigger additional analysis or an alert. In some embodiments, an abnormal value is ignored (e.g., as possibly effected by noise caused by movement of the energy transmitter 204 and/or the energy receiver 206). In various embodiments, the abnormal value may be discounted (e.g., the weight of the value reduced). The degree of discount may be based, for example, on information from an accelerometer (e.g., a large acceleration may indicate that the abnormal value should be significantly discounted) and/or based on historical values. It will be appreciated that the degree of discount may be based on any number of factors.
[0112] In some embodiments, measurements may be averaged over a period of time. A Kalman filer (e.g., a nonlinear, unscented Kalman filter) may be applied to any number of measurements or averaged measurements. A motion measurement (e.g., a measurement by an accelerometer) may be considered. Upon determining a measurement is abnormal, the motion measurement for that time point may be inspected. A large measurement may indicate large vibrations or accelerations that corroborate that the measurement may be abnormal. Measurements collected in such situations are likely to have significant electrical noises.
[0113] At step 308, the analyzer 202 may analyze signals from the energy receiver 206 analyzed in the frequency domain to determine blood metrics. Concentration of a nutrient in the blood may subsequently be determined. In some embodiments, signals may be provided to a bandpass filter that separates AC components from DC components. An AC component may represent signal variation at the cardiac frequency and a DC component may represent the average overall transmitted light intensity. In some embodiments, a heart rate and/or oxygen saturation, SpO.sub.2 may be determined. The heart rate may be determined, for example, by averaging the maximum frequency to determine the rate of cardiac beats in a predetermined amount of time. The oxygen saturation SpO.sub.2 may be determined according to Equation (1):
S.sub.pO.sub.2=110-25.times.R (1),
where R is the ratio of a red and infrared normalized transmitted light intensity. R may be determined according to Equation (2):
R = A C R / D C R A C IR / D C IR , ( 2 ) ##EQU00001##
where the AC.sub.R is the AC component of the detected energy corresponding to a wavelength (e.g., red light), DC.sub.R is the DC component of the detected energy corresponding to the wavelength (e.g., red light), AC.sub.IR is the AC component of the detected energy corresponding to a different wavelength (e.g., infrared light), and DC.sub.IR is the DC component of the detected energy corresponding to the different wavelength (e.g., infrared light). In some embodiments, the AC component may be selected as the highest spectral line in the cardiac frequency band. Waveform analysis may be performed to determine the R-R interval defined by two successive AC components, an elapsed interval and the peturbation, if there is any. It will be appreciated that analysis may be performed by the analyzer 202 and/or any other digital device (e.g., any of users devices 110-114 or analysis system 108).
[0114] At step 308, state space estimation and progression may be performed to determine blood metrics. A system may be modeled according to Equation (3):
x(n+1)=f[x(n)]+u(n)
y(n)=h[x(n)]+v(n) (3),
where x(n) represents the state of the system, u(n) is process noise, y(n) is the vector of the observed signals, and v(n) is the measurement noise.
[0115] Table 1 lists one or more parameters for x(n) as well as their initial value in some embodiments:
TABLE-US-00001 TABLE 1 Parameter Symbol Initial Value Cardiac frequency f.sub.HR 1 Hz Cardiac phase .theta..sub.HR 0 Cardiac harmonic I.sub.Harmonic.sup.HR 0 amplitude Cardiac Pulse P.sub.HR 1 Pressure Point Blood Pressure B.sub.Point 1 Respiratory f.sub.Resp 0.3 Hz frequency Respiratory phase .theta..sub.Resp 0 Wavelength i = 1 . . . N I.sub..lamda..sub.i.sup.AC 0.5 max_value AC peak amplitude Wavelength i = 1 . . . N pos.sub..lamda..sub.i.sup.AC Corresponding FFT AC peak location bin to 1 Hz Wavelength i = 1 . . . N I.sub..lamda..sub.i.sup.DC 0.5 max_value DC Wavelength i = 1 . . . N I.sub..lamda..sub.i.sup.p2p 1 ADC read p2p amplitude Wavelength i = 1 . . . N .tau..sub..lamda..sub.i.sup.rise 0.1 sec rise time Wavelength i = 1 . . . N C.sub..lamda..sub.i 1 Significance coefficient Wavelength i = 1 . . . N T.sub..lamda..sub.i.sup.HRV 1 sec HRV Best Ratio pH BR.sub.pH 2 Best Ratio pCO2 BR.sub.pCO2 3 Best Ratio pHCO3-- BR.sub.pHCO3.sub.- 4 Acceleration I.sub.move 0 magnitude GPS velocity |v|.sub.GPS 0 GPS altitude |alt|.sub.GPS 0 GPS acceleration |a|.sub.GPS 0 GPS incline |incline|.sub.GPS 0 Restfulness Rest 0 Hydration Hyd 0 Systolic Blood SBP 120 mmHg Pressure Diastolic Blood DBP 80 mmHg Pressure End tidal CO2 ETCO2 40 mmHg Blood Carbon SpCO 0% Monoxide
[0116] Table 2 lists one or more parameters for y(n) as well as their initial value in some embodiments:
TABLE-US-00002 TABLE 2 Parameter Symbol Initial Blood pH pH 7.35 Blood PCO2 pCO.sub.2 24 mmol Blood PO2 pO.sub.2 24 mmol Blood PHCO3-- pHCO.sub.3.sup.- 24 mmol Blood Glucose pC.sub.6H.sub.12O.sub.6 3 mmol Cardiac Frequency f.sub.HR 1 Point Blood Pressure P.sub.Point 1 Respiratory f.sub.Resp 0.3 Hz Frequency GPS velocity |v|.sub.GPS 0 GPS altitude |alt|.sub.GPS 0 GPS acceleration |a|.sub.GPS 0 GPS incline |incline|.sub.GPS 0
[0117] Table 3 lists the state space model F(X(n)) between the parameters listed in Table 1 and Table 2 in some embodiments, where the energy wavelengths comprise 880 nm, 631 nm, 1450 nm, and 1550 nm:
TABLE-US-00003 TABLE 3 Name Symbol Equation Cardiac frequency f.sub.HR bin_to _freq ( c .lamda. i pos .lamda. i AC c .lamda. i ) ##EQU00002## Cardiac .theta..sub.HR .theta..sub.HR(n - 1) + f.sub.s.sup.-1 * .omega..sup.*, where .omega..sup.* .di-elect cons. [.omega._min, .omega._max] phase Cardiac harmonic amplitude I.sub.Harmonic.sup.HR c .lamda. i I .lamda. i p 2 p c .lamda. i ##EQU00003## Cardiac Pulse Pressure P.sub.HR ( c .lamda. i .tau. .lamda. i rise c .lamda. i ) ^ - 1 ##EQU00004## Point Blood Pressure P.sub.Point .tau..sub..lamda..sub.1.sup.rise.sup.-1 Respiratory f.sub.Resp 3) Respiratory and Heart Rate State Models: The fluctuations frequency in the respiratory rate .omega..sub.r(n) and fluctuations in the heart rate .omega..sub.ca(n) that are not due to RSA are both modeled as a first-order autoregressive process with a mean and mild non- linearity that limit the frequencies to know physiologic ranges .omega..sub.r(n + 1) = .omega..sub.r + .alpha..sub.r{s.sub.r[.omega..sub.r(n)] - .omega..sub.r} + u.sub..omega..sub.r(n) (15) .omega..sub.ca(n + 1) = .omega..sub.c + .alpha..sub.c{s.sub.c[.omega..sub.ca(n)] - .omega..sub.c} + u.sub..omega..sub.ca(n) (16) where .omega..sub.r and .omega..sub.c are the a priori estimates of the expected res- piratory and cardiac frequencies, respectively; .alpha..sub.r and .alpha..sub.c con- trol the bandwidth of the frequency fluctuations; and u.sub..omega..sub.r(n) and u.sub..omega..sub.ca(n) are white noise processes that model the random variation in the respiratory and cardiac frequencies, respectively. The instantaneous respiratory and heart rates in units of Hz are then f r ( n ) = 1 2 .pi. T s s T [ .omega. r ( n ) ] ( 17 ) ##EQU00005## f c ( n ) = 1 2 .pi. T s s c [ .omega. c ( n ) ] . ( 18 ) ##EQU00006## Respiratory .theta..sub.Resp .theta..sub.Resp(n - 1) + f.sub.s.sup.-1 * .omega..sup.*, where .omega..sup.* .di-elect cons. [.omega._min, .omega._max] phase .lamda. = 880 nm I.sub..lamda..sub.i.sup.AC From FFT AC peak .lamda. = 880 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 880 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 880 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 880 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 880 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 880 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV .lamda. = 631 nm I.sub..lamda..sub.i.sup.AC From Fast Fourier Transformation ("FFT") AC peak .lamda. = 631 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 631 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 631 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 631 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 631 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 631 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV .lamda. = 1450 nm I.sub..lamda..sub.i.sup.AC From FFT AC peak .lamda. = 1450 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 1450 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 1450 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 1450 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 1450 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 1450 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV .lamda. = 1550 nm I.sub..lamda..sub.i.sup.AC From FFT AC peak .lamda. = 1550 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 1550 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 1550 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 1550 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 1550 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 1550 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV Best Ratio BR.sub.pH Device Calibration pH Best Ratio BR.sub.pCO2 Device Calibration pCO2 Best Ratio BR.sub.pHCO3- Device Calibration pHCO3- Acceleration I.sub.move From Accelerometer magnitude GPS velocity |v|.sub.GPS From GPS GPS altitude |alt|.sub.GPS From GPS GPS |a|.sub.GPS From GPS acceleration GPS incline |incline|.sub.GPS From GPS
[0118] Table 4 lists Y(n)=H(x(n)):
TABLE-US-00004 TABLE 4 Name Symbol Equation Blood pH pH 6.1 + log ( pHCO 3 - 0.03 pCO 2 ) ##EQU00007## Blood PCO2 pCO.sub.2 Hb CO 2 - Hb Hb * I .lamda. CO 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. CO 2 DC ) Hb CO 2 - CO 2 CO 2 + ( CO 2 Hb - Hb Hb ) * I .lamda. CO 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. CO 2 DC ) ##EQU00008## Blood PO2 pO.sub.2 Hb O 2 - Hb Hb * I .lamda. O 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. O 2 DC ) Hb O 2 - O 2 O 2 + ( O 2 Hb - Hb Hb ) * I .lamda. O 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. O 2 DC ) ##EQU00009## Blood PHCO3- pHCO.sub.3.sup.- Hb HCO 3 - - Hb Hb * I .lamda. HCO 3 - AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. HCO 3 - DC ) Hb HCO 3 - - HCO 3 - HCO 3 - + ( HCO 3 - Hb - Hb Hb ) * I .lamda. HCO 3 - AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. HCO 3 - DC ) ##EQU00010## Blood Glucose pC.sub.6H.sub.12O.sub.6 As above Cardiac f.sub.HR As in f(x(n)) Frequency Point Blood P.sub.Point As in f(x(n)) Pressure Respiratory f.sub.Resp As in f(x(n)) Frequency GPS velocity |v|.sub.GPS As in f(x(n)) GPS altitude |alt|.sub.GPS As in f(x(n)) GPS |a|.sub.GPS As in f(x(n)) acceleration GPS incline |incline|.sub.GPS As in f(x(n))
[0119] As illustrated in Tables 3 and 4, by generating energy at different wavelengths, one or more blood metrics may be determined from the detected energy. For example, cardiac frequency, cardiac phase, cardiac harmonic amplitude, cardiac pulse pressure, point blood pressure, respiratory frequency, respiratory phase, blood pH, blood pCO.sub.2, blood pHCO.sub.3-, or blood glucose, may be determined.
[0120] FIG. 4 illustrates an exemplary apparatus 400 for measuring various blood metrics in accordance with an embodiment of the present application. The apparatus 400 comprises a central unit 402, a sensor array 404, and a coupling means 408. The central unit 402 may be a wearable member made of elastic and/or flexible hypoallergenic wearable material.
[0121] In the illustrated example, the sensor array 404 is coupled to the central unit 402. The sensor array 404 may comprise any number of energy transmitters and/or energy receivers. The sensor array 404 may be detached from the central unit 402. In some embodiments, the sensor array 404 may be mechanically and electrically coupled to the central unit 402. The sensor array 404 comprises various illumination (e.g., near infra-red, infra-red, or short infra-red) and sensing array. The sensor array 404 may further comprise conductivity and/or capacity sensors. Different sensor array 404 may be provided to measure different blood metrics.
[0122] The central unit 402 may comprise an analyzer. In some embodiments, the central unit comprises an analyzer, one or more energy transmitter(s), and one or more energy receiver(s). The central unit 402 may further comprise a communication module and/or a battery compartment. The coupling means 408 are mounting screw holes in FIG. 4, however, it will be appreciated that coupling means may be optional. Further, coupling means 408 may include any kind of means including a clip, hook, switch, expanding fabric, adhesive, or the like. One of ordinary skill in the art would understand that other mounting means may be used.
[0123] The apparatus 400 further comprises a micro-USB port 406 to allow for communication with a digital device and a screen 410. Various user interfaces (e.g., lights, a display, touchscreen, or the like) may be displayed on the screen 410.
[0124] In some embodiments, the apparatus 400 may be or include the blood metrics measurement apparatus 200 described with regard to FIG. 2, or the blood metrics measurement apparatus 102 described with regard to FIG. 1.
[0125] FIGS. 5-7 are screenshots illustrating an example of presenting health analysis over a user interface in accordance with various embodiments. Various embodiments may store blood metrics and/or nutrient measurements. FIG. 5 illustrates a display 500 of an assessment of a current health index derived from data collected from or with a multispectral blood metrics measurement apparatus in various embodiments. The display may appear on the user's smartphone, for example. In various embodiments, the analyzer 202 or any digital device may analyze measurements collected over time to generate a health score that can be compared to a health threshold to provide qualitative and/or quantitative scoring. Similarly, the analyzer 202 or any digital device may analyze measurements recently collected to generate a current score that can be compared to a current health threshold to provide qualitative and/or quantitative scoring.
[0126] In some embodiments, a user interface may display a health score 502, an option for details regarding the health score 504, a current score 506, an option for details regarding the current score 508, a recommendation 510, a settings option 512, and a history of measurements 514. Options for details 504 and 506 may describe the metrics as well as the values of the metrics that went into the health score 504 and the current score 506, respectively.
[0127] In some embodiments, there is a recommendation engine configured to retrieve recommendations 510 based on the health score 504 and/or the current score 506. The settings option 512 may allow the user to configure metrics to be tracked and set alerts. In some embodiments, the user may utilize the settings options 512 to secure the information (e.g., encrypt the information and/or set passwords). The history of measurements option 514 may provide logged metrics and analysis information over time (e.g., as a chart).
[0128] It will be appreciated that the multispectral blood metrics measurement apparatus 200 and/or any digital device may generate reports based on the analysis, the metrics (e.g., blood metrics or metrics based on nutrients), historic measurements, historic analysis, or any other information. Further, alerts may be set by the multispectral blood metrics measurement apparatus 200 and/or any digital device.
[0129] It will also be appreciated that the multispectral blood metrics measurement apparatus 200 may be taking many measurements over time (e.g., many measurements every minute) and may track health and changes in metrics over time and/or in the short term. In some embodiments, if a condition is of sufficient seriousness (e.g., heart rate shows erratic beats), the multispectral blood metrics measurement apparatus 200 or any digital device may provide an alert and request assistance (e.g., from emergency personnel via the communication network).
[0130] Various health and wellness predictors such as, but not limited to, energy level, blood iron level, blood oxygen level, and blood glucose level are displayed. FIG. 6 illustrates a display 600 of an assessment of an overall health index, derived from data collected from or with a multispectral blood metrics measurement apparatus in various embodiments.
[0131] In some embodiments, a user interface may display a current score 602, energy balance information 606, sleep quality information 608, blood metrics information 610, and body composition information 612 as well as other information accessible by slider 604. Additional details may be available through buttons 614. It will be appreciated that any amount of information may be provided. In some embodiments, the display 600 summarizes information while more detailed information recommendations, measurement data, analysis information, and the like may be available through the details buttons 614 or in other screens.
[0132] Recommendations to the user based on the current and previous measurements are provided. FIG. 7 illustrates a display 700 of an assessment of an overall health index, derived from data collected from or with a multispectral blood metrics measurement apparatus in various embodiments. In some embodiments, a user interface may display a current score 702, energy level information 706, blood iron level information 708, blood oxygen level information 710, and blood glucose level 712 as well as other information accessible by slider 704. Additional details may be available through buttons 714. It will be appreciated that any amount of information may be provided. In some embodiments, the display 700 summarizes information while more detailed information recommendations, measurement data, analysis information, and the like may be available through the details buttons 714 or in other screens.
[0133] Various embodiments track and analyze blood metrics. Health recommendations may be based on instantaneous blood metrics measurements and history blood metrics measurement. In addition, blood metrics and health condition of a user may be compared to health data of the general public. For example, a user's health condition may be compared to health condition of other similar users such as users of the same gender and age group, users of the same profession, friends of a user, etc.
[0134] FIG. 8 is a block diagram of an exemplary digital device 800. The digital device 800 comprises a processor 802, a memory system 804, a storage system 806, a communication network interface 808, an I/O interface 810, and a display interface 812 communicatively coupled to a bus 814. The processor 802 is configured to execute executable instructions (e.g., programs). In some embodiments, the processor 802 comprises circuitry or any processor capable of processing the executable instructions.
[0135] The memory system 804 is any memory configured to store data. Some examples of the memory system 804 are storage devices, such as RAM or ROM. The memory system 804 can comprise the RAM cache. In various embodiments, data is stored within the memory system 804. The data within the memory system 804 may be cleared or ultimately transferred to the storage system 806.
[0136] The storage system 806 is any storage configured to retrieve and store data. Some examples of the storage system 806 are flash drives, hard drives, optical drives, and/or magnetic tape. In some embodiments, the digital device 800 includes a memory system 804 in the form of RAM and a storage system 806 in the form of flash data. Both the memory system 804 and the storage system 806 comprise computer readable media which may store instructions or programs that are executable by a computer processor including the processor 802.
[0137] The communications network interface (com. network interface) 808 can be coupled to a network (e.g., the computer network 104) via the link 816. The communication network interface 808 may support communication over an Ethernet connection, a serial connection, a parallel connection, or an ATA connection, for example. The communication network interface 808 may also support wireless communication (e.g., 802.11 a/b/g/n, WiMax). It will be appreciated that the communication network interface 808 can support many wired and wireless standards.
[0138] The optional input/output (I/O) interface 810 is any device that receives input from the user and output data. The optional display interface 812 is any device that is configured to output graphics and data to a display. In one example, the display interface 812 is a graphics adapter.
[0139] It will be appreciated that the hardware elements of the digital device 800 are not limited to those depicted in FIG. 8. A digital device 800 may comprise more or less hardware elements than those depicted. Further, hardware elements may share functionality and still be within various embodiments described herein. In one example, encoding and/or decoding may be performed by the processor 802 and/or a co-processor located on a GPU (i.e., Nvidia.RTM.).
[0140] Some embodiments described herein include systems and methods for non-invasive continuous blood pressure measurement. For example, a blood metrics measurement apparatus may generate multi-channel signals (e.g., PPG signals or pressure signals) which may be provided to a blood pressure calculation system to calculate arterial blood pressure values (e.g., systolic blood pressure values and/or diastolic blood pressure values). In some embodiments, the blood metrics measurement apparatus includes a pressure sensor for measuring pressure signals. In some embodiments, the blood pressure calculation system may pre-process (or, "filter") the multi-channel signals (e.g., to remove noise from the signals), select (or, "extract") subsets of "high quality" waves from the multi-channel signals, select (or, "extract") sets of features from the high quality waves, and generate sets of feature vectors based on the selected sets of features. In some embodiments, an empirical blood pressure model is used to calculate arterial blood pressure values based on the sets of feature vectors.
[0141] FIG. 9 depicts a block diagram of a system and environment 900 for non-invasive blood pressure measurement according to some embodiments. In some embodiments, the system and environment 900 includes a blood metrics measurement apparatus 902, a user device 904, a blood metrics server 906, a communication network 908, and a communication link 910.
[0142] The blood metrics measurement apparatus 902 may be configured to facilitate non-invasive measurement of a user's blood pressure. In some embodiments, more particularly, the blood metrics measurement apparatus 902 facilitates non-invasive continuous measurement of a user's blood pressure. It will be appreciated that non-invasive continuous measurement may include measuring arterial blood pressure in real-time without interruption (e.g., without having to inflate and deflate a cuff) and without inserting a device (e.g., a tube or catheter) into to the user's tissue or body.
[0143] In some embodiments, the blood metrics measurement apparatus 902 may project energy into tissue of a user (e.g., the wearer of the apparatus 902) and detect (or, "receive") energy reflected from and/or transmitted through tissue of the user. In some embodiments, the blood metrics measurement apparatus 902 may project energy at one or more wavelengths (e.g., 523 nm, 590 nm, 623 nm, 660 nm, 740 nm, 850 nm, 940 nm, etc.) from multiple light sources (e.g., light-emitting diodes). The detected energy may be a fraction (or, "portion") of the energy that is projected into the tissue. Energy at different wavelengths may be absorbed at a different rate that is related to a user's body state. The user's body state (e.g., heart rate, blood pressure, or the like) may determine the amount of absorbed energy. Accordingly, energy at different wavelengths may be absorbed at different levels by a user's body. The fraction of energy received (e.g., that is reflected by the tissue or transmitted through the tissue) may be used to generate signals, such as photoplethysmogram (or, "PPG") signals, at different levels. These signals may provide information of the user's body state. This information may be obtained by analyzing waveforms of the signal in a time domain and/or a frequency domain.
[0144] In some embodiments, the blood metrics measurement apparatus 902 may measure non-optical signals. For example, the blood metrics measurement apparatus 902 may be configured to non-invasively detect arterial pressure of one or more arteries of the user (e.g., radial artery or ulnar artery) based on pressure signals. Similar to optical signals, the pressure signals may be measured to provide information of the user's body state, and this information may be obtained by analyzing waveforms of the signal in a time domain and/or a frequency domain.
[0145] Functionality of the systems and modules described herein may be performed similarly with respect to both optical signals (e.g., PPG signals) and non-optical signals (e.g., pressure signals). Accordingly, it will be appreciated that signals, as used herein, may include optical signals, non-optical signals, or both.
[0146] In some embodiments, a user may comfortably wear the blood metrics measurement apparatus 902 over time. For example, the blood metrics measurement apparatus 902 may be worn without interrupting typical user activity (e.g., moving, walking, running, sleeping, etc.). The blood metrics measurement apparatus 902 may comprise lightweight components. The blood metrics measurement apparatus 902 may be made of hypoallergenic materials. The blood metrics measurement apparatus 902 may be flexibly built so that it may fit various body parts (e.g., wrist, earlobe, ankle, or chest) of a user. In some embodiments, the blood metrics measurement apparatus 902 may include some or all of the functionality of the user device 904.
[0147] In some embodiments, the blood metrics measurement apparatus 902 may be or include the apparatus 400 described with regard to FIG. 4, the blood metrics measurement apparatus 200 depicted with regard to FIG. 2, or the blood metrics measurement apparatus 102 described with regard to FIG. 1.
[0148] The user device 904 may include any digital device (e.g., mobile device) capable of executing an application related to measuring blood metrics, such as blood pressure calculation, presenting a user interface through a display and/or communicating with various entities in the example system and environment 900 through the communication network 908 and/or a communication link 910 (discussed further below). For example, through the communication link 910, the user device 902 may receive one or more blood metric measurements (e.g., one or more signals) from the blood metrics measurement apparatus 902, track and store the blood metric measurements, analyze the blood metric measurements, and/or provide recommendations and/or messages based on the blood metric measurements. An application user interface may facilitate interaction between a user of the user device 904 and an application running on the user device 904. The user device 904 may be, include, or be a part of the user system 104 described with regard to FIG. 1.
[0149] In various embodiments, the user device 904 may perform analysis of the measurements received from the blood metrics measurement apparatus 902 (e.g., calculate blood pressure values), display results, provide reports, display progress, display historic readings, track measurements, track analysis, provide alerts (or, messages), and/or the like.
[0150] The blood metrics server 906 may be configured to generate and/or store empirical blood pressure models. For example, the blood metrics server 906 may comprise one or more server computers, desktop computers, mobile devices, and/or other digital device(s). In some embodiments, the blood metrics server 906 receives and process user registration requests (e.g., user account registration requests, blood metrics measurement apparatus registration requests, etc.), provides empirical blood pressure model(s) to the user device 902 via the communication network 908, and/or the like.
[0151] As used in this paper, computing devices (e.g., digital devices) may include a mobile phone, a tablet computing device, a laptop, a desktop computer, personal digital assistant, a portable gaming unit, a wired gaming unit, a thin client, a set-top box, a portable multi-media player, or any other type of network accessible user device known to those of skill in the art. Further, the blood metrics server 908 may comprise of one or more servers, which may be operating on or implemented using one or more cloud-based services (e.g., System-as-a-Service [SaaS], Platform-as-a-Service [PaaS], or Infrastructure-as-a-Service [IaaS]).
[0152] Each of the blood metrics measurement apparatus 902, the user device 904, and the blood metrics server 906 may be implemented using one or more digital devices. An example digital device is described in FIG. 8.
[0153] In some embodiments, the communication network 908 represents one or more communication network(s). The communication network 908 may provide communication between the blood metrics measurement apparatus 902, the user device 904, and/or the blood metrics server 906. In some examples, the communication network 908 comprises digital devices, routers, cables, and/or other network topology. In other examples, the communication network 908 may be wireless and/or wireless. In some embodiments, the communication network 908 may be another type of network, such as the Internet, that may be public, private, IP-based, non-IP based, and so forth.
[0154] In some embodiments, the communication link 910 represents one or more communication network connections. The communication link 910 may provide communication between the blood metrics measurement apparatus 902 and the user device 904. In some examples, the communication link 910 comprises a network connection of the communication network 908, and/or a separate communication network. In some embodiments, the communication link 910 comprises a wireless communication link, such as a Bluetooth communication link, Wi-Fi communication link, and/or the like.
[0155] FIG. 10A depicts a block diagram 1000 of a blood metrics measurement apparatus 902 according to some embodiments. The blood metrics measurement apparatus 902 comprises an analyzer 1002, an energy transmitter 1004, an energy receiver 1006, a pressure sensor 1008, a motion sensor 1012, and a communication module 1014. Various embodiments may comprise a wearable member. The wearable member may include, for example, a bracelet, glasses, necklace, ring, anklet, belt, broach, jewelry, clothing, or any other member of combination of members that allow the blood metrics measurement apparatus 902 to be close to or touch a body of the wearer. In some embodiments, the blood metrics measurement apparatus 902 may further comprise a driver (not shown) and a power source (not shown). The power source may be coupled to the energy transmitter 1004 via the driver. The blood metrics measurement apparatus 902 may further comprise an Analog-to-Digital Converter ("ADC") (not shown). The ADC may be coupled to the energy receiver 1006 and the analyzer 202.
[0156] The analyzer 1002 may be coupled to the energy transmitter 1004, the energy receiver 1006, the pressure sensor 1008, the motion sensor 1012, and the communication module 1014. The energy transmitter 1004 and the energy receiver 1006 may be secured to the wearable member such that the energy transmitter 1004 and the energy receiver 1006 may make contact or be in proximity with tissues (e.g., skin) of a user. In various embodiments, the energy transmitter 1004 emits energy including, but not limited to, light, into the body of the user. The energy produced by the energy transmitter may be in the direction of entering tissues. For example, the energy produced by the energy transmitter 1004 is in a direction 1051 entering the tissue 1010. In some embodiments, the energy transmitter 1004 emits energy or light at different wavelengths. The energy transmitter 1004 may comprise any number of light emission diodes ("LEDs"). In some embodiments, the energy transmitter 1004 comprises at least two LEDs. Each LED may be configured to emit energy at one or more wavelengths. In another example, each LED may emit light with a peak wavelength centered around a wavelength. In one example, the energy transmitter 1004 may emit light with a peak wavelength centered around 500 nm to 1800 nm, although the wavelength may include a variety of spectrums (e.g., IR, near-IR, and the like).
[0157] Each wavelength may correspond to one or more blood metrics of interest and/or one or more nutrients. It will be appreciated that different components of the blood and/or different nutrients may absorb energy at different wavelengths. In various embodiments, a controller, driver, analyzer 1002, or the like may receive a blood metric or nutrient of interest (e.g., from a user of the blood metrics measurement apparatus 902 and/or a user device not shown). The controller, driver, analyzer 1002 or the like may associate the blood metric and/or nutrient of interest with one or more wavelengths and configure one or more of the LEDs to emit energy of at least one of the one or more wavelengths. For example, the analyzer 1002 may command the driver to deliver electric power to one LED that is configured to emit light at the desired wavelength.
[0158] The energy receiver 1006 may detect energy associated with the energy provided by the LEDs from tissues (e.g., skin) of the user. In this example, received and/or detected energy is in the direction 1052 that leaves from the tissue 1010. In various embodiments, the energy receiver 1006 may detect energy from the body of the user that is a fraction of the energy produced by the energy transmitter 1004.
[0159] The energy transmitter 1004 and the energy receiver 1006 may be configured such that the energy receiver 1006 detects reflected energy from tissues of the user of the multispectral blood metrics measurement apparatus 902. For example, the energy transmitter 1004 and the energy receiver 1006 may be configured to be disposed on one surface or side of a user's tissue. The energy transmitter 1004 and the energy receiver 1006 may be configured such that the energy receiver 1006 detects energy from the energy transmitter 1004 that passes through or reflects from the user's tissues. In some embodiments, the energy transmitter 1004 and the energy receiver 1006 may be configured to be disposed on different (e.g., opposite) surfaces or sides of a users' tissue.
[0160] The energy transmitter 1004 may be configured to generate energy at a set of wavelengths. In some embodiments, the energy transmitter 1004 is configured to generate energy such that energy at different wavelengths is generated sequentially and/or periodically. The energy transmitter 1004 may be configured to generate energy at each particular wavelength until energy at all wavelengths of a set is generated. The period of time for the energy transmitter 1004 to generate energy at all wavelengths is a generation period. Subsequent to completion of the generation period, the energy transmitter 1004 may start a new generation period thereby allowing multiple measurements.
[0161] Energy detected from tissues of a user may be detected by the energy receiver 1006. The energy receiver 1006 may be configured to generate a signal in response to the detected energy. In some embodiments, the energy receiver 1006 may be triggered by the energy received to generate an output which may be dependent or partially dependent upon the amount of energy received. The energy receiver 1006 may be configured to generate a signal (e.g., an electric current, or an electric voltage) in response to the energy received from the tissues.
[0162] The signal generated by the energy receiver 1006 may be associated with one or more blood metrics and/or nutrients of interest. Energy at different wavelengths may be absorbed at a different rate that is related to a user's body state. The user's body state (e.g., heart rate, blood pressure, nutrient level, or the like) may determine the amount of energy absorbed by the body. Accordingly, energy from the user's body at different wavelengths may be detected at different levels thereby causing different responses of the energy receiver 1006. The energy receiver 1006 may, for example, output signals based on the level of the energy received.
[0163] The energy receiver 1006 may provide information associated with the user's body state. Blood metric information may be determined (e.g., by the analyzer 1002) from the output signal of the energy receiver 1006. In some embodiments, the energy receiver 1006 may comprise a set of photodetectors (e.g., a photo diode, or a photo transistor) which are configured to output a signal dependent upon photons or the like from the energy transmitter 1004 that passed through tissues of the user.
[0164] In various embodiments, the output signal of the energy receiver 1006 is a composite of multiple signals. Each signal of the composite may be associated with energy at a wavelength which may be a portion (or fraction) of the total energy emitted by the energy transmitter 1004.
[0165] The pressure sensor 1008 may be configured to generate, detect, and/or measure non-optical signals. For example, the pressure sensor 1008 may non-invasively and continuously generate, detect and/or measure pressure pulse signals. In some embodiments, the pressure sensor 1008 measures pressure pulse waveforms associated with arterial pressure of one or more arteries of a user. In various embodiments, the blood metrics measurement apparatus 902 may include the energy transmitter 1004 to generate optical signals and the energy receiver 1006 to receive optical signals but not the pressure sensor 1008. Alternately, the blood metrics measurement apparatus 902 may include the pressure sensor 1008 that may produce pressure on the user's body and/or receive measurements based on that pressure but not the energy transmitter 1004 or the energy receiver 1006 to receive optical signals.
[0166] In some embodiments, the motion sensor 1012 may be configured to detect position and orientation of the blood metrics measurement apparatus 902, and detect motion of the blood metrics measurement apparatus 902. For example, the blood metrics measurement apparatus 902 may detect position, orientation, and motion along an x, y, or z-axis, and measured values may include velocity, acceleration, distance, and the like. In some embodiments, the motion sensor 1012 may include one or more accelerometers, gyroscope, global positioning systems, or the like. The motion sensor may be coupled to the analyzer 1002.
[0167] The communication module 1014 may be configured to send requests to and receive data from one or a plurality of systems. The communication module 1014 may send requests to and receive data from a systems through a network or a portion of a network. Depending upon implementation-specific or other considerations, the communication module 1014 may send requests and receive data through a connection (e.g., the communication link 910), all or a portion of which may be a wireless connection. The communication module 1014 may request and receive messages, and/or other communications from associated systems.
[0168] FIG. 10B depicts a block diagram of a sensor system 1060 according to some embodiments. In some embodiments, the sensor system 1060 may be disposed in the blood metrics measurement apparatus 902. For example, the energy transmitter 1004 and the energy receiver 1006 may comprise the sensor system 1060. As shown, the sensor system 1060 comprises two sets of multi-LED energy transmitters (i.e., LED set 1062 and 1066 each referred to as energy transmitters herein), and two sets of corresponding photodiode energy receivers 1064 and 1068. It will be appreciated that each pair of corresponding energy transmitters and energy receivers (e.g., energy transmitter 1062 and energy receiver 1064 as well as energy transmitter 1066 and energy receiver 1068) may be referred to as an LED-PD system, and the sensor system 1060 may include any number of such LED-PD systems, although only two are shown here.
[0169] In some embodiments, the energy transmitters 1062 and 1066 each comprise a set of LEDs (e.g., between 2-4 LEDs) configured to transmit a variety of wavelengths into tissue of a user. The corresponding energy receivers 1064 and 1068 may each comprise one or more photodiodes which receive returning light from the corresponding energy transmitters 1062 and 1066 after passage through tissue. In some embodiments, the energy transmitter 1062 and the energy receiver 1064 may be spaced at a predetermined distance (e.g., 15 mm) from the energy transmitter 1066 and the energy receiver 1068. Similarly, each energy transmitter 1062 and 1066 may be spaced at a predetermined distance (e.g., 10 mm) from a corresponding energy receiver 1064 and 1068.
[0170] In some embodiments, the sensor system 1060 may be mounted on a user's wrist along the radial artery, or other area of the user's body along arterial pathways (e.g., ulnar artery). The sensor system 1060 may include an accelerometer and gyroscope for detecting position and orientation information.
[0171] In some embodiments, the sensor system 1060 may include a pressure sensor. For example, at least one LED within each LED-PD system may be configured to sample data from tissue at a very high frequency (upwards of 1 KHz), and the other LEDs may sample at a lower rate (around 50-100 Hz). The channels sampling data at high frequency may be referred to as the fast LED channels, and the channels sampling data at lower frequency may be referred to as slow LED channels.
[0172] In some embodiments, the sensor system 1060 may be included as a part of the blood metrics measurement apparatus 902 described with regard to FIGS. 9 and 10A, the apparatus 400 described with regard to FIG. 4, the blood metrics measurement apparatus 200 described with regard to FIG. 2, or the blood metrics measurement apparatus 102 described with regard to FIG. 1. Similarly, the analyzer 202 may be or include a part of analyzer 1002 described with regard to FIG. 2 or the central unit 402 described with regard to FIG. 4. The energy transmitter 1004 may be or include a part of energy transmitter 204 described with regard to FIG. 2 or the central unit 402 described with regard to FIG. 4. The energy receiver 1006 may be or include energy receiver 206 described with regard to FIG. 2 or the central unit 402 described with regard to FIG. 4.
[0173] FIG. 11 depicts a flowchart 1100 of an example method of operation of a blood metrics measurement apparatus (e.g., blood metrics measurement apparatus 902) according to some embodiments. In this and other flowcharts described herein, the flowchart illustrates by way of example a sequence of steps. It should be understood the steps may be reorganized for parallel execution, or reordered, as applicable. Moreover, some steps that could have been included may have been removed to avoid providing too much information for the sake of clarity and some steps that were included could be removed, but may have been included for the sake of illustrative clarity.
[0174] In step 1102, a blood metrics measurement apparatus projects energy into tissue of a user (e.g., the user wearing blood metrics measurement apparatus). The energy may be projected from a transmitter (e.g., energy transmitter 1004) comprising a plurality of light sources (e.g., LEDs). In some embodiments, a first light source (e.g., one or more LEDs) may project light energy at a plurality of different wavelengths, such as 523 nm, 590 nm, 623 nm, 660 nm, 740 nm, 850 nm, and 940 nm, and a second light source (e.g., one or more LEDs) may project energy at the same, or substantially similar, wavelength as one of the wavelengths projected by the first light source (e.g., 523 nm, 590 nm, 623 nm, 660 nm, 740 nm, 850 nm, or 940 nm). It will be appreciated that other configuration may be used (e.g., a greater number of light sources) a greater or lesser number of wavelengths projected from the lights sources, and so forth.
[0175] In step 1104, the blood metrics measurement apparatus receives (or, "detects) portions of energy through the tissue of the user. In some embodiments, an energy receiver (e.g., energy transmitter 1006) detects a portion of the energy transmitted into the user's tissue by the energy transmitter. The energy receiver may generate a signal based on the portion of energy detected (e.g., based on the amount of the energy detected). For example, energy detected may be a portion of the energy projected at step 1102 reflected by the tissue. By way of further example, energy detected may be a portion of the energy projected at step 1102 that passes through the tissue (e.g., other undetected energy may be absorbed by tissue and/or otherwise blocked). In various embodiments, steps 1102 and 1104 are performed simultaneously or substantially simultaneously. That is, energy projection and detection may be performed approximately simultaneously.
[0176] In step 1106, the blood metrics measurement apparatus generates one or more signals based on the received portions of energy. In some embodiments, the energy receiver may generate a multi-channel PPG signal (e.g., as mentioned above). The output (or, "generated") signal of the energy receiver may be an electric current or an electric voltage, of which the amplitude may be related to the amount of the energy detected.
[0177] In various embodiments, analysis of the signals from the energy receiver may identify abnormal measurements. For example, each of the measurements may be compared to a predetermined value. If the difference between the measurement and the predetermined value is above (or below) a threshold, then the measurement may be determined to be abnormal. An abnormal value may trigger additional analysis or an alert. In some embodiments, an abnormal value is ignored (e.g., as possibly effected by noise caused by movement of the energy transmitter and/or the energy receiver). In various embodiments, the abnormal value may be discounted (e.g., the weight of the value reduced). The degree of discount may be based, for example, on information from an accelerometer (e.g., a large acceleration may indicate that the abnormal value should be significantly discounted) and/or based on historical values. It will be appreciated that the degree of discount may be based on any number of factors.
[0178] In some embodiments, measurements may be averaged over a period of time. A Kalman filer (e.g., a nonlinear, unscented Kalman filter) may be applied to any number of measurements or averaged measurements. A motion measurement (e.g., a measurement by an accelerometer) may be considered. Upon determining a measurement is abnormal, the motion measurement for that time point may be inspected. A large measurement may indicate large vibrations or accelerations that corroborate that the measurement may be abnormal. Measurements collected in such situations are likely to have significant electrical noises.
[0179] At step 1108, the blood metrics measurement apparatus analyzes signals from the energy receiver analyzed in the frequency domain to determine blood metrics. Concentration of a nutrient in the blood may subsequently be determined. In some embodiments, signals may be provided to a bandpass filter that separates AC components from DC components. An AC component may represent signal variation at the cardiac frequency and a DC component may represent the average overall transmitted light intensity. In some embodiments, a heart rate and/or oxygen saturation, SpO.sub.2 may be determined. The heart rate may be determined, for example, by averaging the maximum frequency to determine the rate of cardiac beats in a predetermined amount of time. The oxygen saturation SpO.sub.2 may be determined according to Equation (1):
S.sub.pO.sub.2=110-25.times.R (1),
[0180] where R is the ration of a red and infrared normalized transmitted light intensity. R may be determined according to Equation (2):
R = A C R / D C R A C IR / D C IR , ( 2 ) , ##EQU00011##
where the AC.sub.R is the AC component of the detected energy corresponding to a wavelength (e.g., red light), DC.sub.R is the DC component of the detected energy corresponding to the wavelength (e.g., red light), AC.sub.IR is the AC component of the detected energy corresponding to a different wavelength (e.g., infrared light), and DC.sub.IR is the DC component of the detected energy corresponding to the different wavelength (e.g., infrared light). In some embodiments, the AC component may be selected as the highest spectral line in the cardiac frequency band. Waveform analysis may be performed to determine the R-R interval defined by two successive AC components, an elapsed interval and the probation, if there is any.
[0181] It will be appreciated that analysis may be performed by the analyzer and/or any other digital device (e.g., user device or a blood metrics server such as blood metrics server 906).
[0182] State space estimation and progression may be performed to determine blood metrics. A system may be modeled according to Equation (3):
x(n+1)=f[x(n)]+u(n)
y(n)=h[x(n)]+v(n) (3),
where x(n) represents the state of the system, u(n) is process noise, y(n) is the vector of the observed signals, and v(n) is the measurement noise.
[0183] Table 1 lists one or more parameters for x(n) as well as their initial value in some embodiments:
TABLE-US-00005 TABLE 1 Parameter Symbol Initial Value Cardiac frequency f.sub.HR 1 Hz Cardiac phase .theta..sub.hr 0 Cardiac harmonic I.sub.Harmonic.sup.HR 0 amplitude Cardiac Pulse P.sub.HR 1 Pressure Point Blood Pressure P.sub.Point 1 Respiratory f.sub.Resp 0.3 Hz frequency Respiratory phase .theta..sub.Resp 0 Wavelength i = 1 . . . N I.sub..lamda..sub.i.sup.AC 0.5 max_value AC peak amplitude Wavelength i = 1 . . . N pos.sub..lamda..sub.i.sup.AC Corresponding FFT AC peak location bin to 1 Hz Wavelength i = 1 . . . N I.sub..lamda..sub.i.sup.DC 0.5 max_value DC Wavelength i = 1 . . . N I.sub..lamda..sub.i.sup.p2p 1 ADC read p2p amplitude Wavelength i = 1 . . . N .tau..sub..lamda..sub.i.sup.rise 0.1 sec rise time Wavelength i = 1 . . . N C.sub..lamda..sub.i 1 Significance coefficient Wavelength i = 1 . . . N T.sub..lamda..sub.i.sup.HRV 1 sec HRV Best Ratio pH BR.sub.pH 2 Best Ratio pCO2 BR.sub.pCO2 3 Best Ratio pHCO3-- BR.sub.pHCO3.sub.- 4 Acceleration I.sub.move 0 magnitude GPS velocity |v|.sub.GPS 0 GPS altitude |alt|.sub.GPS 0 GPS acceleration |a|.sub.GPS 0 GPS incline |incline|.sub.GPS 0 Restfulness Rest 0 Hydration Hyd 0 Systolic Blood SBP 120 mmHg Pressure Diastolic Blood DBP 80 mmHg Pressure End tidal CO2 ETCO2 40 mmHg Blood Carbon SpCO 0% Monoxide
[0184] Table 2 lists one or more parameters for y(n) as well as their initial value in some embodiments:
TABLE-US-00006 TABLE 2 Parameter Symbol Initial Blood pH pH 7.35 Blood PCO2 pCO.sub.2 24 mmol Blood PO2 pO.sub.2 24 mmol Blood PHCO3-- pHCO.sub.3.sup.- 24 mmol Blood Glucose pC.sub.6H.sub.12O.sub.6 3 mmol Cardiac Frequency f.sub.HR 1 Point Blood Pressure P.sub.Point 1 Respiratory f.sub.Resp 0.3 Frequency GPS velocity |v|.sub.GPS 0 GPS altitude |alt|.sub.GPS 0 GPS acceleration |a|.sub.GPS 0 GPS incline |incline|.sub.GPS 0
[0185] Table 3 lists the state space model F(X(n)) between the parameters listed in Table 1 and Table 2 in some embodiments, where the energy wavelengths comprise 880 nm, 631 nm, 1450 nm, and 1550 nm:
TABLE-US-00007 TABLE 3 Name Symbol Equation Cardiac frequency f.sub.HR bin_to _freq ( c .lamda. i pos .lamda. i AC c .lamda. i ) ##EQU00012## Cardiac .theta..sub.HR .theta..sub.HR(n - 1) + f.sub.s.sup.-1 * .omega..sup.*, where .omega..sup.* .di-elect cons. [.omega._min, .omega._max] phase Cardiac harmonic amplitude I.sub.Harmonic.sup.HR c .lamda. i I .lamda. i p 2 p c .lamda. i ##EQU00013## Cardiac Pulse Pressure P.sub.HR ( c .lamda. i .tau. .lamda. i rise c .lamda. i ) ^ - 1 ##EQU00014## Point Blood Pressure P.sub.Point .tau..sub..lamda..sub.1.sup.rise.sup.-1 Respiratory f.sub.Resp 3) Respiratory and Heart Rate State Models: The fluctuations frequency in the respiratory rate .omega..sub.r(n) and fluctuations in the heart rate .omega..sub.ca(n) that are not due to RSA are both modeled as a first-order autoregressive process with a mean and mild non- linearity that limit the frequencies to know physiologic ranges .omega..sub.r(n + 1) = .omega..sub.r + .alpha..sub.r{s.sub.r[.omega..sub.r(n)] - .omega..sub.r} + u.sub..omega..sub.r(n) (15) .omega..sub.ca(n + 1) = .omega..sub.c + .alpha..sub.c{s.sub.c[.omega..sub.ca(n)] - .omega..sub.c} + u.sub..omega..sub.ca(n) (16) where .omega..sub.r and .omega..sub.c are the a priori estimates of the expected res- piratory and cardiac frequencies, respectively; .alpha..sub.r and .alpha..sub.c con- trol the bandwidth of the frequency fluctuations; and u.sub..omega..sub.r(n) and u.sub..omega..sub.ca(n) are white noise processes that model the random variation in the respiratory and cardiac frequencies, respectively. The instantaneous respiratory and heart rates in units of Hz are then f r ( n ) = 1 2 .pi. T s s T [ .omega. r ( n ) ] ( 17 ) ##EQU00015## f c ( n ) = 1 2 .pi. T s s c [ .omega. c ( n ) ] . ( 18 ) ##EQU00016## Respiratory .theta..sub.Resp .theta..sub.Resp(n - 1) + f.sub.s.sup.-1 * .omega..sup.*, where .omega..sup.* .di-elect cons. [.omega._min, .omega._max] phase .lamda. = 880 nm I.sub..lamda..sub.i.sup.AC From FFT AC peak .lamda. = 880 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 880 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 880 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 880 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 880 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 880 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV .lamda. = 631 nm I.sub..lamda..sub.i.sup.AC From Fast Fourier Transformation ("FFT") AC peak .lamda. = 631 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 631 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 631 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 631 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 631 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 631 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV .lamda. = 1450 nm I.sub..lamda..sub.i.sup.AC From FFT AC peak .lamda. = 1450 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 1450 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 1450 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 1450 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 1450 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 1450 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV .lamda. = 1550 nm I.sub..lamda..sub.i.sup.AC From FFT AC peak .lamda. = 1550 nm pos.sub..lamda..sub.i.sup.AC From FFT DC .lamda. = 1550 nm I.sub..lamda..sub.i.sup.DC From Waveform analysis p2p amplitude .lamda. = 1550 nm I.sub..lamda..sub.i.sup.p2p From Waveform analysis rise time .lamda. = 1550 nm .tau..sub..lamda..sub.i.sup.rise From Waveform analysis signal trend .lamda. = 1550 nm c.sub..lamda..sub.i From Waveform analysis Significance coefficient .lamda. = 1550 nm T.sub..lamda..sub.i.sup.HRV From Waveform analysis HRV Best Ratio BR.sub.pH Device Calibration pH Best Ratio BR.sub.pCO2 Device Calibration pCO2 Best Ratio BR.sub.pHCO3- Device Calibration pHCO3- Acceleration I.sub.move From Accelerometer magnitude GPS velocity |v|.sub.GPS From GPS GPS altitude |alt|.sub.GPS From GPS GPS |a|.sub.GPS From GPS acceleration GPS incline |incline|.sub.GPS From GPS
TABLE-US-00008 TABLE 4 Table 4 lists Y(n) = H(x(n)): Name Symbol Equation Blood pH pH 6.1 + log ( pHCO 3 - 0.03 pCO 2 ) ##EQU00017## Blood PCO2 pCO.sub.2 Hb CO 2 - Hb Hb * I .lamda. CO 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. CO 2 DC ) Hb CO 2 - CO 2 CO 2 + ( CO 2 Hb - Hb Hb ) * I .lamda. CO 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. CO 2 DC ) ##EQU00018## Blood PO2 pO.sub.2 Hb O 2 - Hb Hb * I .lamda. O 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. O 2 DC ) Hb O 2 - O 2 O 2 + ( O 2 Hb - Hb Hb ) * I .lamda. O 2 AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. O 2 DC ) ##EQU00019## Blood PHCO3- pHCO.sub.3.sup.- Hb HCO 3 - - Hb Hb * I .lamda. HCO 3 - AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. HCO 3 - DC ) Hb HCO 3 - - HCO 3 - HCO 3 - + ( HCO 3 - Hb - Hb Hb ) * I .lamda. HCO 3 - AC * I .lamda. 1 DC / ( I .lamda. 1 AC * I .lamda. HCO 3 - DC ) ##EQU00020## Blood Glucose pC.sub.6H.sub.12O.sub.6 As above Cardiac f.sub.HR As in f(x(n)) Frequency Point Blood P.sub.Point As in f(x(n)) Pressure Respiratory f.sub.Resp As in f(x(n)) Frequency GPS velocity |v|.sub.GPS As in f(x(n)) GPS altitude |alt|.sub.GPS As in f(x(n)) GPS |a|.sub.GPS As in f(x(n)) acceleration GPS incline |incline|.sub.GPS As in f(x(n))
[0186] As illustrated in Tables 3 and 4, by generating energy at different wavelengths, one or more blood metrics may be determined from the detected energy. For example, cardiac frequency, cardiac phase, cardiac harmonic amplitude, cardiac pulse pressure, point blood pressure, respiratory frequency, respiratory phase, blood pH, blood pCO.sub.2, blood pHCO.sub.3-, or blood glucose, may be determined.
[0187] In step 1110, the blood metrics measurement apparatus provides the generated one or more signals for blood pressure calculation. In some embodiments, a communication module (e.g., communication module 1008) provides the one or more signals (e.g., to a blood pressure calculation system and/or user device).
[0188] FIG. 12 depicts a block diagram 1200 of a user device 904 according to some embodiments. Generally, the user device 904 may be configured to display, or otherwise present blood pressure values, messages, alerts, and/or the like. The user device 904 may also provide registration features allowing a user to register a blood metrics measurement device 902, create and/or update a user account, and communicate with other systems of the system and environment 900. In some embodiments, the user device 904 includes a user interface module 1202, a registration module 1204, a blood pressure calculation system 1206, and a communication module 1208. In some embodiments, the blood metrics measurement device 902 may include all or part of the blood pressure calculation system 1206.
[0189] The user interface module 1202 may be configured to present images and/or audio corresponding to health data, such as blood pressure values, messages, alerts, and the like. For example, the user interface module 1202 may display one or more graphical user interfaces (GUIs) to present a calculated blood pressure to a user. Example user interfaces are described further with reference to FIG. 5-7, discussed above.
[0190] The registration module 1204 may be configured to generate registration requests to create, read, update, delete, or otherwise access, registration records associated with user accounts (e.g., a user account associated with a user of the user device 904 and/or the blood metrics measurement apparatus 902) and registration records associated with the blood metrics measurement apparatus 902. In some embodiments, a user inputs user account registration information and blood metrics measurement apparatus registration information via the user interface module 1202. For example, user account registration information may include geographic attributes, demographic attributes, psychographic attributes, and/or behavioristic attributes. Accordingly, user account registration information may include some or all of the following attributes: [0191] User Account Identifier: Identifier that identifies a user account. [0192] Password: Password, or other personal identifier, used to authenticate the user account. For example, it may an alphanumerical password, biometric data (e.g., fingerprint, etc.). In some embodiments, readings or measurements from the blood metrics measurement apparatus 902 may be used to authenticate the user account. [0193] Device Identifier(s): Identifier(s) that identify one or more blood metric measurement apparatus' associated with the user account. [0194] Name: A name of the user. [0195] DOB: A date of birth of the user. [0196] Age: An age of the user. [0197] Gender: Gender of the user (e.g., female, male, transgender, or the like). [0198] Weight: A weight of the user. [0199] Height: A height of the user. [0200] Skin color: A skin color of the user. [0201] Activity Level: An activity level of the user (e.g., sedentary, lightly active, active, very active, and so forth). [0202] Geographic location: A location of the user (e.g., as determined by a location service and/or specified by the user). [0203] Blood Pressure Profile: Hypertensive, Hypotensive, Normal or unknown. [0204] Blood Glucose Profile (e.g., Diabetes information) [0205] Wrist circumference: Circumference of the user's wrist.
[0206] In some embodiments, the blood metrics measurement apparatus registration information includes some or all of the following attributes: [0207] Apparatus Identifier: Identifier that identifies a blood metrics measurement apparatus. [0208] User Account Identifier: Identifier that identifies a user account associated with the blood metrics measurement apparatus. [0209] Geographic location: A current location of the blood metrics measurement apparatus (e.g., as determined by a location service and/or specified by the user). [0210] Settings: One or more settings of the blood measurement metrics apparatus. For example, some or all of the settings may be automatically determined based on one or more user account attributes (e.g., height, weight, or the like) and/or by the user.
[0211] The blood pressure calculation system 1206 may be configured to calculate blood pressure values (e.g., systolic, diastolic), and generate messages or alerts based on those values. An example of the blood pressure calculation system 1206 is discussed further below with reference to FIG. 14.
[0212] The communication module 1208 may be configured to send requests to and receive data from one or a plurality of systems. The communication module 1208 may send requests to and receive data from a systems through a network or a portion of a network. Depending upon implementation-specific or other considerations, the communication module 1208 may send requests and receive data through a connection (e.g., the communication network 908, and/or the communication link 910), all or a portion of which may be a wireless connection. The communication module 1208 may request and receive messages, and/or other communications from associated systems.
[0213] FIG. 13 depicts a flowchart 1300 of an example method of operation of a user device (e.g. user device 904) according to some embodiments.
[0214] In step 1302, a user device registers a blood metrics measurement apparatus (e.g., blood metrics measurement apparatus 902). In some embodiments, input from a user is received by a user interface module (e.g., user interface module 1202) that triggers a registration module (e.g., registration module 1204) to generate a registration request to associate the blood metrics measurement apparatus with a user and/or the user device. The registration request may include, for example, one or more blood metrics measurement apparatus attributes. In some embodiments, a communication module (e.g., communication module 1208) provides the registration request to a blood metrics server (e.g., blood metrics server 906) for processing.
[0215] In step 1304, the user device receives one or more signals from the registered blood metrics measurement apparatus. In some embodiments, the one or more signals comprise optical signals (e.g., multi-channel PPG signals) and/or one or more non-optical signals (e.g., multi-channel pressure pulse signals). In some embodiments, the one or more signals may be received by the communication module.
[0216] In step 1306, the user device calculates one or more arterial blood pressure values (e.g., systolic values and/or diastolic values) based on the received one or more signals. In some embodiments, a blood pressure calculation system (e.g., blood pressure calculation system 1206) calculates the one or more arterial blood pressure values. Although this example depicts the user device calculating the one or more arterial blood pressure values, it will be appreciated that one or more other systems having the functionality of a blood pressure calculation system may perform the calculation. For example, in some embodiments, the blood pressure measurement apparatus and/or the blood metrics server may include such functionality and perform the calculation.
[0217] In step 1308, the user device presents a blood pressure message to the user based on at least one of the one or more calculated arterial blood pressure values. For example, the message may include some or all of the arterial blood pressure values, alerts (e.g., high BP, low BP, good BP, poor BP, etc.) based on one or more of the calculated values, and so forth. In some embodiments, the user device presents (e.g., via images, audio, vibrations, etc.) the blood pressure message or alert to the user via the user interface module, or other feature of the user device.
[0218] FIG. 14 depicts a block diagram 1400 of a blood pressure calculation system 1206 according to some embodiments. Generally, the blood pressure calculation system 1206 may be configured to calculate arterial blood pressure values of a user. The blood pressure calculation system 1206 may also store calculated arterial blood pressure values (e.g., for health tracking, etc.), and communicate with other systems of the system and environment 900. In some embodiments, the blood pressure calculation system 1206 includes a management module 1402, a signal database 1404, a wave database 1406, a wave feature database 1408, a feature vector database 1410, a blood pressure model database 1412, a blood pressure results database 1414, a rules database 1416, a pre-processing module 1418, a wave selection module 1420, a feature extraction module 1422, a blood pressure processing module 1424, and a communication module 1426.
[0219] The management module 1402 may be configured to manage (e.g., create, read, update, delete, or access) signal records 1428 stored in the signal database 1404, wave records 1430 stored in the wave database 1406, wave feature records 1432 stored in the wave feature database 1408, feature vector records 1434 stored in the feature vector database 1410, empirical blood pressure model records 1436 stored in the blood pressure model database 1412, blood pressure result records 1438 stored in the blood pressure results database 1414, and/or rules 1440-1448 stored in rules database 1416. The management module 1402 may perform these operations manually (e.g., by an administrator interacting with a GUI) and/or automatically (e.g., by one or more of the modules 1418-1424). In some embodiments, the management module 1402 comprises a library of executable instructions which are executable by a processor for performing any of the aforementioned management operations. The databases 1404-1416 may be any structure and/or structures suitable for storing the records 1428-1438 and/or the rules 1440-1448 (e.g., an active database, a relational database, a table, a matrix, an array, a flat file, and the like).
[0220] The signal records 1428 may include a variety of signals, along with associated metadata. For example, the signals may comprise optical signals (e.g., single-channel and/or multi-channel PPG signals) and/or non-optical signals (e.g., pressure pulse signals). In some embodiments, the metadata may include information obtained from the signals, such as heart rate(s) of an associated user. For example, the signal records 1428 may store some or all of the following information: [0221] Signal(s) Identifier: Identifier that identifies the stored signal(s). [0222] Signal(s): one or more signals. The signals may be raw signals (e.g., as detected by the associated blood pressure measurement apparatus), filtered or pre-processed signals (e.g., to remove noise from the signals), and/or normalized signal values (e.g., between 0-1). An example "noisy" (i.e., unfiltered) PPG signal and an example filtered PPG signal are shown in FIG. 21. It will be appreciated that as used in this paper, a "signal," such as a PPG signal or pressure pulse signal, generally refers to a filtered signal, although in some embodiments, it may also refer to an unfiltered signal instead of, or in addition to, the filtered signal. [0223] Set(s) of Waves: one or more sets of waves of a predetermined time series (e.g., 8 seconds) of the signal(s). The set of waves may include raw waves, filtered waves, and/or normalized wave values (e.g., between 0-1). An example set of waves is shown in FIG. 22. [0224] Apparatus Identifier: Identifier that identifies the blood metrics measurement apparatus that generated the signals. [0225] User Account Identifier: Identifier that identifies a user account associated with the blood metrics measurement apparatus that generated the signals. [0226] Metadata: Metadata obtained from the signals, such as heart rate or other biometric data. The metadata may also include other information of the user, such as gender, age, height, weight, skin color (e.g., obtained from the user's account information). Such metadata values may be used by the blood pressure calculation module 1424 (discussed below) to facilitate calculation of arterial blood pressure values. In some embodiments, metadata values may be provided to an empirical blood pressure model (discussed below) via sets of feature vectors (discussed below) and/or be provided separately to the model.
[0227] The wave records 1430 may include sets of waves of a signal (e.g., a signal stored in the signal database 1404), along with subsets of those waves. The subsets of waves may comprise "high quality" waves obtained from the waves of the signal. These subsets of waves may provide, for example, a more accurate blood pressure calculation that just using the signal or waves of the signal. In some embodiments, the wave records 1430 may store some or all of the following information: [0228] Signal Identifier: Identifier that identifies an associated signal. [0229] Wave Identifier(s): Identifiers for subsets of waves of the associated signal. [0230] Subset(s) of Waves: one or more subsets of waves of the associated signal. The subsets of waves may include raw waves, filtered waves, and/or normalized wave values (e.g., between 0-1). The subsets of waves may be referred to as "high quality" waves. An example wave of a subset of waves is shown in FIG. 22. [0231] Apparatus Identifier: Identifier that identifies the blood metrics measurement apparatus that generated the signals. [0232] User Account Identifier: Identifier that identifies a user account associated with the blood metrics measurement apparatus that generated the signals.
[0233] The wave feature records 1432 may include wave features of associated subsets of waves of a signal. For example, wave features may include wave peaks, wave valleys, wave edges, and/or the like. In some embodiments, the wave feature records 1432 may store some or all of the following information: [0234] Signal Identifier: Identifier that identifies an associated signal. [0235] Wave Identifier(s): Identifiers for the subsets of waves of the associated signal. [0236] Wave Features: one or more features obtained from the waves within the associated subsets of waves. The wave features may include points of the waves, such as wave peaks, wave valleys, wave edges, and/or the like. The wave features may be stored as normalized wave values (e.g., between 0-1). Example wave features are shown in FIG. 23, and discussed elsewhere herein. [0237] Apparatus Identifier: Identifier that identifies the blood metrics measurement apparatus that generated the signals. [0238] User Account Identifier: Identifier that identifies a user account associated with the blood metrics measurement apparatus that generated the signals.
[0239] The feature vector records 1434 may include sets of features generated based on the wave features of associated subsets of waves of a signal. In some embodiments, the feature vector records 1434 may store some or all of the following information: [0240] Signal Identifier: Identifier that identifies an associated signal. [0241] Wave Identifier(s): Identifiers for the subsets of waves of the associated signal. [0242] Set(s) of Feature Vectors: one or more sets of feature vectors, each feature vector comprising features extracted from a wave of an associated subset of waves. The values of a feature vector may include measurement values and metric values. For example, the measurement values may correspond to amplitude or location points of a particular wave, and the metric values may be generated from metric functions that use at least one of the measurement values. The values of a feature vector may comprise normalized values (e.g., between 0-1). An example feature vector 2400 is shown in FIG. 24. [0243] Apparatus Identifier: Identifier that identifies the blood metrics measurement apparatus that generated the signals. [0244] User Account Identifier: Identifier that identifies a user account associated with the blood metrics measurement apparatus that generated the signals.
[0245] The blood pressure model records 1436 may include one or more empirical blood pressure models (e.g., retrieved from the blood metrics server 906). The models may include various types of empirical blood pressure models. For example, a first type may be a "non-specific" model which does not require calibration in order to be used to calculate arterial blood pressure values. A second type may be a "specific" model which requires calibration in order to be used to calculate arterial blood pressure values. For example, models of the second type may require information about the user, such age, weight, height, gender, skin color, and/or the like. In some embodiments, the blood pressure records 1436 may store some or all of the following information: [0246] Model Identifier: Identifies an empirical blood pressure model. [0247] Model Type: Identifies a type of model (e.g., non-specific or specific). [0248] Model Parameters: Various model parameters (e.g., decision node parameters) and tree structures used to calculate the arterial blood pressure values based on the sets of feature vectors and/or other related information (e.g., gender, age, weight, height, skin color, etc.). Example tree structures are shown in FIG. 26. [0249] Apparatus Identifier(s): Identifier(s) that identify one or more blood metrics measurement apparatus' using the empirical blood pressure model. [0250] User Account Identifier(s): Identifier(s) that identify one or more user account(s) associated with the blood metrics measurement apparatus that use the empirical blood pressure model.
[0251] The blood pressure results records 1438 may include one or more calculated arterial blood pressure values. In some embodiments, the blood pressure results records 1438 may store some or all of the following information: [0252] Blood Pressure Result Identifier: Identifies a set of one or more calculated arterial blood pressure values. [0253] Blood Pressure Values: one or more calculated arterial blood pressure values. [0254] Date: A date and/or time the arterial blood pressure was calculated. [0255] Messages: Message identifier and/or messages generated based on the calculated blood pressure values. [0256] Signal Identifier: Identifier that identifies an associated signal. [0257] Wave Identifier(s): Identifiers for the subsets of waves of the associated signal. [0258] Feature Vectors Identifier(s): Identifiers for the one or more sets of feature vectors used to calculate the arterial blood pressure values. [0259] Apparatus Identifier: Identifier that identifies the blood metrics measurement apparatus that generated the signals. [0260] User Account Identifier: Identifier that identifies a user account associated with the blood metrics measurement apparatus that generated the signals.
[0261] Pre-Processing Rules 1440
[0262] The pre-processing rules 1440 define attributes and/or functions for filtering signals. For example, a signal may be corrupted with noise from various sources (e.g., high frequency ambient noise, electronic noise, and the like). In some embodiments, the signal may be the raw signal data from a photodiode, output of a pressure sensor, output of an accelerometer, output of a gyroscope, and the like. The signal may be filtered to remove corrupting noise, as well as enhance a strength of the signal.
[0263] The pre-processing module 1418 may be configured to execute the pre-processing rules 1440. Thus, for example, the pre-processing module 1418, using some or all of the associated signals and/or values stored in the signal records 1428, may filter signals and store the resulting filtered signals. The wave database 1406 may be configured to store the signals filtered by the pre-processing module 1418.
[0264] Wave Selection Rules 1442
[0265] The wave selection rules 1442 define attributes and/or functions for selecting (or, "extracting") high quality waves from a set of waves of a signal. The high quality waves may form a subset of waves.
[0266] In some embodiments, signal measurements are sensitive to motion, pressure, and ambient light distortions. Noise-removal filters may not be entirely effective when there is intense noise. It may be helpful to select high quality signal waves from measured time-series data to reduce or eliminate the effects of motion, pressure, and/or ambient light distortions.
[0267] In some embodiments, the wave selection rules 1442 identifies candidate waves from a signal, and determines whether any of the candidate waves satisfy a model match criterion (e.g., a Gaussian mixture model or group similarity model). Candidate waves that pass the model match criterion may be classified as high quality waves. In some embodiments, candidate waves includes waves having valleys whose frequency match, or substantially match, the heart rate of the user or other heart rate value (e.g. a default heart rate value).
[0268] A group similarity model may be preferable. For example, while a Gaussian mixture model may perform well with "smoothed" signals (e.g., low pass filtered at 4-6 Hz), the Gaussian mixture model may fail, or perform poorly, if the signal includes more features. FIG. 22 depicts an example smoothed signal 2202, and FIG. 28 depicts an example signal including more features (e.g., low pass filtered at 10 Hz revealing multiple reflections including, for example, secondary peaks, such as iliac and renal reflections). In some embodiments, the Gaussian mixture model may be unable to match more than 2 peak results in a reflection of waves that reveal features critical in the prediction (or, "estimation) of blood pressure. In some embodiments, additional Gaussian mixture models may be incorporated to address such issues, although this may lead to added complexity in optimization (e.g., model fitting), as well as added constraints to ensure specificity to physiologically meaningful signals against noise.
[0269] In some embodiments, a group similarity model may overcome some or all of the limitations described above. Generally, high quality (or high fidelity) waves that correspond with arterial blood flow corresponding to a single cycle typically occur in groups. In cases where there is motion, it may be unlikely that an individual wave would have a high quality when waves before it were not of high quality. In the group similarity model, candidate waves may be individually matched to an existing wave buffer (e.g., of the wave database 1406). In some embodiments, the wave buffer may include some or all previous candidates waves added in succession in a first in first out (FIFO) method. The size of the wave buffer may be set to include a predetermined number of waves (e.g., 15 waves), and may be altered. For example, increasing the wave buffer to include additional candidate waves may require that candidate waves match a larger set of previously extracted candidate waves. In some embodiments, in order to measure similarity, the candidate waves may be re-parameterized to a fixed length (e.g., using a cubic spline).
[0270] In various embodiments, the group similarity model applies a similarity measure to determine if, and to what extent, a candidate wave matches some or all of the existing waves in the wave buffer. In one example, the similarity measure may be an average correlation between the candidate wave and the individual waves in the wave buffer. In other embodiments, the similarity measure may include a like sum of squared differences, joint entropy or mutual information, and the like. In this example, if the similarity measure is greater than a threshold (e.g. 0.95 for correlation), this wave is selected for further processing. In some embodiments, both the candidate waves that are selected for further processing and the candidate waves that are not selected for further processing may be added to the wave buffer (e.g., for subsequent applications of the group similarity model).
[0271] The group similarity model may process waves with multiple reflections (e.g., as depicted in FIG. 28). In some embodiments, the group similarity model may continuously update a template (e.g., the wave buffer) against which incoming candidate waves are matched, and that template may indicate the true underlying signal, which may be different from user to user, instead of relying on a predetermined template. The group similarity model may reduce false positive identification of candidate waves relative to other models (e.g., Gaussian mixture model).
[0272] The wave selection module 1420 may be configured to execute the wave selection rules 1442. Thus, for example, the wave selection module 1420, using some or all of the associated waves and/or values stored in the signal records 1428, may identify one or more subsets of "high quality" waves. The wave database 1406 may be configured to store the subsets of waves identified by the wave selection module 1420. An example result of execution of the wave selection rules 1442 is shown in FIG. 22.
[0273] Feature Extraction Rules 1444
[0274] The feature extraction rules 1444 define attributes and/or functions for identifying (or, "extracting") features from waves (e.g., high quality waves). In some embodiments, features may be concatenated to form feature vectors.
[0275] Features may include, but are not limited to, pulse transit time (PTT) features, reflection features, signal level and range features, signal metric features, optical ratio features, heart rate features, wave width and derivative features, user information features, and/or pressure features. In some embodiments, PTT features include a transit time feature, joint entropy feature, and wave type feature. Pulse transit time may be measured as the time taken for the pulse pressure wave to propagate along the length of the arterial tree. For example, this may be the difference in time between the onset of the R-wave on ECG, and the pulse wave peak on the finger. In other embodiments, the transit time may be measured as the time taken for blood to travel from one LED-PD system to another LED-PD system (e.g., as depicted in FIG. 10B), which may be separated from each other by a predetermined distance (e.g., 15 mm). Sampling at a very high rate (>2 KHz) may help resolve features in both LED-PD systems. In some embodiments, the transit time may be measured as (1) the distance between the valleys in the corresponding waves in respective LED-PD systems at the start of the systolic cycle, and/or (2) the distance between the peaks in first derivatives between the two LED-PD systems. The transit time may be expressed in any units (e.g., milliseconds).
[0276] The joint entropy feature may be a measurement of similarity between fast channels of respective LED-PD systems. High pulse pressure may correlate with low entropy or high mutual information shared between the fast LED channels. The joint entropy feature may be expressed in bytes.
[0277] Wave type features may include different types of waves. For example, different types of waves may include slow type waves and fast type waves. In some embodiments, the wave types feature quantifies the relative position of the systolic peak within the wave (0<WaveType<1) for each of the fast channels in respective LED-PD systems, where small values correspond to a slow type waves and larger values correspond to fast type waves.
[0278] A pulse decomposition may track a pulse pressure wave. As the pressure wave travels in the arterial systems, it may encounter branching points where the diameter of arteries may decrease rapidly. The pressure wave may bounce on such branching points and send reflection waves in different (e.g., opposite) directions. In some embodiments, the original pressure wave plus the reflections may form the observed pressure pulse, which in turn may be observed in a signal (e.g., PPG signal). Multiple reflections (e.g., four reflections) may be used to identify multiple features and amplitudes (e.g., four features and four amplitudes). The time of occurrence of the systolic peak from the start of the systolic cycle (i.e., systolic peak time) may also be included as a feature. In some embodiments, a ratio between the second derivatives at the bottom and peak of the systolic cycle may be used as a feature. This feature may be correlated with arterial stiffness. In some embodiments, ten features may be individually identified for each LED channel.
[0279] The signal level and range features may comprise the mean value and range of a signal (e.g., PPG signal) determined for each of the channels. In some embodiments, the signal metrics features include Hjorth parameters, perfusion, kurtosis, and energy features. Hjorth parameters may describe activity, mobility and complexity of the signal, and are commonly used tools in EEG analysis. Perfusion may be measured as the ratio between the AC and DC components of the signal (i.e. the range of the signal and the mean). Kurtosis may be measured over the signal analysis window. Energy features may include variance of the signal measured over the analysis window. In some embodiments, optical ratio features may be calculated as the ratio of the range of the LEDs (measured in pairs across wavelengths) after normalization of the respective signals, and heart rate features may be an actual user heart rate or a default heart rate.
[0280] In some embodiments, the wave width and derivative features may include time distances within a wave to its main peak at different amplitude locations. One example is shown in FIG. 25A for 0% amplitude location. In FIG. 25A, d1 and d2 represent the distance between the rising (falling) edge value of the wave and its main (systolic) peak. The secondary (diastolic) peak may be extracted from the first order derivative (FOD) of the wave. For this, FOD may be smoothed (e.g., a simple moving average filter may be used). In some embodiments, after extracting main (systolic) and secondary (diastolic) peaks, reflection (augmentation) index (the ratio of diastolic peak and systolic peak amplitudes), inflection point area ratio (the ratio of areas under the wave that are separated by the diastolic inflection point), and/or stiffness index (the ratio of patient's height to the time distance between the systolic and diastolic peaks) may be determined from the first order derivative of the wave.
[0281] In the example of FIG. 25C, the second order derivative of the wave has multiple peaks and valley points (e.g., labeled (a), (b) and (e)). In some embodiments additional peak and valleys may be present. If the sampling frequency is low or under exercise conditions (e.g., about 25 Hz in FIG. 25C), only waves (a), (b) and (e) may be identifiable. In some embodiments, for higher sampling frequencies (e.g., .gtoreq.200 Hz), additional peaks and valleys may be identifiable. The ratio of these values may be included into a feature vector.
[0282] As indicated, an example selected (or, extracted) wave, first order derivative of the selected wave, and the second derivative of the selected wave are shown in FIGS. 25A-C, respectively.
[0283] In some embodiments, if two LEDs at the same wavelength are positioned at different locations of the same artery, a phase shift may be obtained between the measured signals (e.g., due to blood flow). This may facilitate calculation of pulse wave velocity (PWV) and/or pulse transit time (PTT), both of which may be included in a feature vector, and/or otherwise provided to the empirical blood pressure model used to calculate arterial blood pressure values.
[0284] The user information features may include, for example, a user's gender, age, skin color, height and/or weight. The user information may be included in a feature vector, and/or otherwise provided to the empirical blood pressure model used to calculate arterial blood pressure values. In some embodiments, pressure features may include the mean value of the pressure signal over the analysis window (or "baseline mean") and range of the pressure signal over the analysis window (or, "AC").
[0285] The feature extraction module 1422 may be configured to execute the feature extraction rules 1444. Thus, for example, the feature extraction module 1420, using some or all of the associated subsets of waves and/or values stored in the wave records 1430, may identify one or more feature of the waves within the subsets of waves, and generate corresponding sets of feature vectors. An example of a feature vector 2400 is shown in FIG. 24. The wave feature database 1408 may be configured to store the wave features identified by the feature extraction module 1422, and the feature vector database 1410 may be configured to store the generated sets of feature vectors.
[0286] Blood Pressure Processing Rules 1446
[0287] The blood pressure processing rules 1446 define attributes and/or functions for calculating arterial blood pressure values of a user. In some embodiments, the blood pressure processing rules 1446 specify, identify, and/or define the empirical blood pressure model to use for calculating arterial blood pressure of a user. The rules 1446 may further define input values for the empirical blood pressure model. For example, input values may comprises the sets of features vectors, and/or other attributes of the user (e.g., age, gender, height, weight, skin color, etc.), assuming such attributes have not been included in the feature vectors.
[0288] The blood pressure processing module 1424 may be configured to execute the blood pressure processing rules 1446. Thus, for example, the blood pressure processing module 1424, using an empirical blood pressure model stored in the blood pressure model database 1412, along with some or all of the associated sets of feature vectors and/or values stored in the feature vector records 1438, may calculate one or more arterial blood pressure values. The blood pressure results database 1414 may be configured to store the blood pressure values calculated by the blood pressure processing module 1424.
[0289] Message Rules 1448
[0290] The message rules 1448 define attributes and/or functions for generating messages and/or alerts based on arterial blood pressure values. In some embodiments, the message rules 1448 may define rules that cause the blood pressure calculation system 1206 to provide calculate blood pressure values to a user. In some embodiments, the message rules 1448 may include threshold values and/or conditions that when exceeded and/or satisfied, trigger a message or alert. For example, a threshold value (or value range) and/or threshold condition may be associated with varying blood pressure levels (e.g., hypotension, normal blood pressure, prehypertension, stage 1 hypertension, stage 2 hypertension, etc.), and a calculated blood pressure value which satisfies a corresponding threshold condition or value may trigger a message or alert (e.g., indicating the corresponding blood pressure level).
[0291] In some embodiments, the communication module 1426 may be configured to execute the message rules 1448. Thus, for example, the communication module 1420, using some or all of the blood pressure values stored in the blood pressure results records 1438, may generate one or more messages. The communication module 1426 may be configured to provide those messages to a user.
[0292] In some embodiments, the communication module 1426 may be configured to send requests to and receive data from one or a plurality of systems. The communication module 1426 may send requests to and receive data from a systems through a network or a portion of a network. Depending upon implementation-specific or other considerations, the communication module 1426 may send requests and receive data through a connection (e.g., the communication network 908, and/or the communication link 910), all or a portion of which may be a wireless connection. The communication module 1426 may request and receive messages, and/or other communications from associated systems.
[0293] FIG. 15A depicts a flowchart 1500 of an example method of operation of a blood pressure calculation system (e.g., blood pressure calculation system 1206) according to some embodiments.
[0294] In step 1502, the blood pressure calculation system stores one or more empirical blood pressure models. For example, the one or more empirical blood pressure models may be received from a blood metrics server (e.g., blood metrics server 906), and may comprise specific or non-specific empirical blood pressure models. In some embodiments, a management module (e.g., management module 1402) stores the one or more empirical blood pressure models in a blood pressure model database (e.g., blood pressure results database 1414), and/or a communication module (e.g., communication module 1426) receives the one or more empirical blood pressure models.
[0295] In step 1504, the blood pressure calculation system receives a first signal (e.g., a single-channel or multi-channel PPG signal) and a second signal (e.g., a single-channel or multi-channel PPG signal). For example, the first signal may comprise a 7-channel PPG signal generated from light energy emitted at seven different wavelengths (e.g., 523 nm, 590 nm, 623 nm, 660 nm, 740 nm, 850 nm, 940 nm) from one or more light sources (e.g., seven different LEDs), and the second signal may comprise a PPG signal generated from light energy generated from an additional light source (e.g., LED) at the same, or substantially similar, wavelength as one the wavelengths of the first signal. Using multi-channel signals and/or different light sources may help ensure, for example, that good quality signals may be obtained in a variety of circumstances (e.g., a user moving, walking, running, sleeping, and so forth). By way of further example, the first and/or second signals may comprise a pressure pulse signal. In some embodiments, a communication module communication module (e.g., communication module 1426 and/or communication module 1208) receives the first and second signals from a blood metrics measurement apparatus (e.g., blood metrics measurement apparatus 902).
[0296] In step 1504, the blood pressure calculation system pre-processes (or, filters) the first and second signals. In some embodiments, a pre-processing module executes pre-processing rules to filter the first and second signals. An example filtering method is shown and described with reference to FIG. 15B.
[0297] In steps 1508-1510, the blood pressure calculation system identifies a first subset of waves (or, "high quality" waves) from a first set of waves of the first signal and a second subset of waves (or, "high quality" waves) from a second set of waves of the second signal. Each of the first subset of waves may represent a separate approximation of an average of the first set of waves over a first predetermined amount of time (e.g., 8 seconds). Similarly, each of the second subset of waves may represent a separate approximation of an average of the second set of waves over a second predetermined amount of time (e.g., 8 seconds). In some embodiments, the first and second predetermined amounts of time may be the same or they may be different. In some embodiments, a wave selection module (e.g., wave selection module 1420) identifies the subsets of waves.
[0298] In steps 1512-1514, the blood pressure calculation system generates a first set of feature vectors and a second set of feature vectors. The first set of feature vectors may be generated from the first subset of waves, and the second set of feature vectors may be generated from the second subset of waves. Each of the feature vectors may include measurement values and/or metric values. For example, the measurement values may correspond to amplitude (e.g., peak-to-peak amplitude, peak amplitude, semi-amplitude, root mean square amplitude, pulse amplitude, etc.) and/or location points of a particular wave (e.g., a corresponding high quality wave), and the metric values may be generated from metric functions that use at least one of the measurement values. In some embodiments, a feature extraction module (e.g., feature extraction module 1420) generates the sets of feature vectors.
[0299] In some embodiments, measurement values may include wave peak locations and/or amplitudes, wave valley locations and/or amplitudes, a wave's first or higher order derivative peak locations and/or amplitudes, a wave's first or higher order derivative valley locations and/or amplitudes, and/or or first or higher order moments of a wave. In various embodiments, the metric functions may include one or more particular metric functions that calculate a distance between a plurality of measurement values. For example, a metric function may calculate a distance between location points of a particular wave (e.g., a particular wave peak and a particular wave valley).
[0300] In step 1516, the blood pressure calculation system may select an empirical blood pressure model. For example, the blood pressure calculation system may select an empirical blood pressure model from the one or more empirical blood pressure models stored in the blood pressure model database. In some embodiments, a blood pressure processing module (e.g., blood pressure processing module 1422) selects the empirical blood pressure model.
[0301] In some embodiments, the blood pressure calculation system may operate in different modes. For example, in a first mode of operation, the blood pressure calculation system may utilize a non-specific type of empirical blood pressure model which does not require further calibration in order to be used to calculation arterial blood pressure values. Accordingly, in such a first mode of operation, selecting the empirical blood pressure model may comprise selecting the non-specific empirical blood pressure model from the empirical blood pressure models stored in the blood pressure database. To continue the example, in a second mode of operation, the blood pressure calculation system may utilize a specific type of empirical blood pressure model which requires at least some calibration prior to being used to calculate arterial blood pressure values. In such a second mode of operation, selecting an empirical blood pressure model may comprise selecting an empirical blood pressure model from the one or more blood pressure models stored in the blood pressure based on one or more attributes of the user (e.g., gender, weight, height, skin color, and/or age) and/or other parameters. In some embodiments, a blood pressure processing module (e.g., blood pressure processing module 1422) selects the empirical blood pressure model.
[0302] In step 1518, the blood pressure calculation system calculates one or more arterial blood pressure values based on the first set of feature vectors, the second set of feature vectors, and an empirical blood pressure calculation model (e.g., the model selected in step 1516). For example, the empirical blood pressure calculation model may be configured to receive the first set of feature vectors and the second set of feature vectors as input values. In some embodiments, the blood pressure processing module calculates the one or more arterial blood pressure values.
[0303] In step 1520, the blood pressure calculation system may generate and/or provide a message including or being based on the arterial blood pressure. In some embodiments, the communication module generates and/or provides the message to a user.
[0304] FIG. 15B depicts a flowchart 1530 of an example method of pre-processing (or, "filtering") a signal according to some embodiments. Although only one signal is discussed here, it will be appreciated that multiple signals may be filtered (e.g., simultaneously) using some or all of the steps described below.
[0305] In step 1532, a blood pressure calculation system 1206 receives a signal at least partially corrupted with noise (or, a "noisy" signal). For example, the signal may be an output from a photodiode, a pressure sensor, an accelerometer, a gyroscope, or the like. The noise may include one or more types of noise, such as high frequency ambient noise, electronic noise, and so forth. For example, the noise may be motion-related noise associated with venous blood movement, changes in sensor coupling with the tissue of the user, and so forth. In some embodiments, a communication module (e.g., communication module 1426) receives the signal from a blood metrics measurement apparatus.
[0306] In step 1534, the blood pressure calculation system 1206 generates and/or applies a low pass filter to the signal. For example, the blood pressure calculation system 1206 may apply a finite impulse response (FIR) low pass filter with a predetermined cut off frequency (e.g., 10 Hz) or frequency range (e.g., 6 Hz-12 Hz). In some embodiments, the low pass filter may be configured to pass signals having a frequency lower than the predetermined cut off frequency, and/or configured to attenuate signals having frequencies higher than the cutoff frequency. In some embodiments, a pre-processing module applies the low pass filter.
[0307] In step 1536, the blood pressure calculation system 1206 normalizes the sign. For example, the blood pressure calculation system may normalizes the signal by subtracting and dividing by the mean (DC) value of the signal. In some embodiments, the pre-processing module 1418 normalizes the signal.
[0308] In step 1538, the blood pressure calculation system 1206 inverts the signal. For example, the blood pressure calculation system may invert the signal such that an increasing in amplitude corresponds with an increase in volume in the arteries. In some embodiments, the pre-processing module 1418 inverts the signal.
[0309] In step 1540, the blood pressure calculation system 1206 applies a high pass filter to the signal. In some embodiments, the high pass filter may be configured to pass signals having a frequency higher than a certain cutoff frequency, and/or configured to attenuate signals having frequencies lower than the cutoff frequency. For example, the high pass filter may be an FIR filter having a predetermined cut off frequency (e.g., 0.6 Hz). In another example, the high pass filter may be a Savitzksy-Golay filter configured to remove low frequency fluctuations arising from respiration rate.
[0310] In step 1542, the blood pressure calculation system 1206 performs a blind source separation on the signal to isolate signals associated with arteries from the venous pulsations. In some embodiments, this step does not apply to non-optical signals. In various embodiments, signals may typically contain both venous and arterial components to varying degrees, and signal data collected from a channel may be dependent on the position on the tissue of a user where the data is collected, as well as the wavelength of the light source, which may have an effect on the depth of penetration of that light wavelength into tissue, as well as scattering. If a channel is composed of two independent components (e.g., arterial and venous signals), the components may be isolated by using multiple channels using independent component analysis. In some embodiments, the pre-processing module performs the blind source separation.
[0311] In step 1544, the blood pressure calculation system 1206 generates and/or applies an adaptive filter to the signal. In some embodiments, the adaptive filter removes noise from the signal using one or more other signals that may be related to the noise as a noise reference. For example, the adaptive filter may be configured to remove motion related noise from the signal using an accelerometer signal as a noise reference. In some embodiments, motion information may be captured by an accelerometer along three axis (e.g., x, y, and z-axis). Motion along each access may be captured by a component of the accelerometer. In various embodiments, the blood pressure calculation system calculates a sum of the three accelerometer components, and uses the sum as a noise reference. Alternately, the blood pressure calculation system 1206 may determine the largest varying component and uses that component as the reference. In some embodiments, the blood pressure calculation system may use each component successively to form three adaptive filters in sequence. In some embodiments, the pre-processing module generates and/or applies the adaptive filter.
[0312] In step 1546, the blood pressure calculation system 1206 outputs a filtered signal (i.e., the signal filtered by some or all of the steps 1532-1544). In some embodiments, the pre-processing module 1418 outputs the filtered signal.
[0313] FIG. 15C depicts a blocks diagram 1550 of an example filtering system 1552 for filtering signals according to some embodiments. The filtering system 1552 includes an adaptive filter 1554 that may be continuously updated. The adaptive filter 1554 may be dynamically modified such that its convolution with the noise reference 1556 creates a noise component that is maximally similar to the motion related noise in the signal 1558 corrupted with motion noise. The filtering system 1552 may be configured to optimize the adaptive filter 1552 to minimize the output signal 1560 from the adaptive filter 1552. For example, the output signal 1560 may be minimal when a filtered version 1562 of the noise reference 1556 has removed a threshold portion (e.g., greater than 80%) of the corresponding noise present in the noise-corrupted signal 1558. Various minimization methods may be used (e.g., recursive least square minimization) to produce the minimized output signal 1560.
[0314] FIG. 16A depicts a flowchart 1600 of an example method for extracting high quality waves according to some embodiments. For example, some or all of the following steps may be applied to a segment (e.g., an 8 second segment or 12 second segment) of each channel of a multi-channel signal to identify subsets of waves of the multi-channel signal.
[0315] In step 1602, the blood pressure calculation system (e.g., blood pressure calculation system 1206) identifies peak values smaller (or, less) than a mean subtracted by a standard deviation of the signal. In some embodiments, a wave selection module performs the identification.
[0316] In step 1604, the blood pressure calculation system selects peak valleys whose distances with a next peak valley are within a threshold value (e.g., 5%) of a heart rate value and whose intensities are within a threshold value (e.g., 5%) of each other. In some embodiments, the wave selection module performs the selection.
[0317] In step 1606, the blood pressure calculation system determines whether there is only one identifiable peak between the detected valleys, whose intensity value is greater than a mean plus one standard deviation. In some embodiments, the wave selection module performs the determination.
[0318] In step 1608, the blood pressure calculation system identifies one or more candidate waves. For example, the one or more candidate waves may be identified from steps 1602-1606. As used herein, it will be appreciated that candidate waves may also be referred to as "model candidate waves." In some embodiments, the wave selection module performs the identification.
[0319] In step 1610, the blood pressure calculation system determines if one or more of the candidate waves satisfy a model match criterion (e.g., a Gaussian mixture model or a group similarity model). An example method using a Gaussian mixture model is depicted in FIG. 16B, and an example method using a group similarity model is depicted in FIG. 16C. In some embodiments, the wave selection module performs the determination.
[0320] In step 1612, the blood pressure calculation system selects the waves satisfying the model match criterion. The selected waves may be grouped into subsets of "high quality" waves. In some embodiments, the selected waves may be stored in a training dataset with the associated channel number. In some embodiments, the wave selection module performs the selection and/or storage.
[0321] FIG. 16B depicts a flowchart 1620 of an example method for extracting high quality waves using a Gaussian mixture model according to some embodiments. For example, some or all of the following steps may be applied to a segment of each channel of a multi-channel signal to identify subsets of waves of the multi-channel signal.
[0322] In step 1622, the blood pressure calculation system (e.g., blood pressure calculation system 1206) identifies one or more candidate waves. For example, the one or more candidate waves may be identified using steps 1602-1606. In some embodiments, a wave selection module performs the identification.
[0323] In step 1624, the blood pressure calculation system determines whether a candidate wave after normalization (e.g., 0-1), satisfies a bi-Gaussian mixture model and the model mismatch error is within a predetermined (e.g., user-defined) threshold value (e.g., 2%). In some embodiments, the wave selection module performs the determination. FIG. 27 depicts an example bi-Gaussian mixture model 2700 for a PPG signal.
[0324] In step 1626, the blood pressure calculation system determines whether the fitted model has a reflection coefficient between minimum and maximum possible values (e.g., 0.3-0.7), where the reflection coefficient is defined as the ratio of the diastolic peak to the systolic peak values. In some embodiments, the wave selection module performs the determination.
[0325] In step 1628, the blood pressure calculation system selects the candidate waves satisfying the conditions of steps 1622-1626. The selected candidate waves may comprise high quality waves. The high quality waves may be stored in a wave database. In some embodiments, the wave selection module performs the selection and/or storage.
[0326] FIG. 16C depicts a flowchart 1640 of an example method of method for extracting high quality waves using a group similarity model according to some embodiments. For example, some or all of the following steps may be applied to a segment of each channel of a multi-channel signal to identify subsets of waves of the multi-channel signal.
[0327] In step 1642, the blood pressure calculation system identifies one or more candidate waves. For example, the one or more candidate waves may be identified using steps 1602-1606. In some embodiments, a wave selection module performs the identification.
[0328] In step 1644, the blood pressure calculation system applies a similarity measure to determine if at least one of the one or more candidate waves match one or more other waves (e.g., waves stored in a wave buffer). In some embodiments, the wave selection module applies the similarity measure.
[0329] In step 1646, if at least one candidate wave matches the other waves, the blood pressure calculation system selects and identified the at least one candidate wave as high quality (step 1648), and stores the at least one candidate wave with the other waves (step 1650). The candidate waves that do not match may also be stored with the other waves (step 1650). In some embodiments, the wave selection performs the selection, identification, and/or storage.
[0330] FIG. 17 depicts a block diagram 1700 of a blood metrics server 906 according to some embodiments. Generally, the blood metrics server 906 may be configured to generate and/or store empirical blood pressure models, provide empirical blood pressure models to a blood pressure calculation system, register user account and blood metrics measurement apparatus`, and communicate with other systems of the system and environment 900. In some embodiments, the blood metrics server 906 includes a management module 1702, a registration database 1704, a blood pressure database 1706, a model generation and selection module 1708, and a communication module 1710.
[0331] The management module 1702 may be configured to manage (e.g., create, read, update, delete, or access) user account records 1714 and blood metrics measurement apparatus records 1716 stored in the registration database 1704, and blood pressure model records 1718 stored in the blood pressure model database 1706. The management module 1402 may perform these operations manually (e.g., by an administrator interacting with a GUI) and/or automatically (e.g., by one or more of the modules 1708 or 1710). In some embodiments, the management module 1702 comprises a library of executable instructions which are executable by a processor for performing any of the aforementioned management operations. The databases 1704 and 1706 may be any structure and/or structures suitable for storing the records 1714-1718 (e.g., an active database, a relational database, a table, a matrix, an array, a flat file, and the like).
[0332] The user account records 1714 may include a variety of information associated with users, user accounts, associated blood metrics measurement apparatus' and/or associated user devices. For example, the user account records 1714 may store some or all of the following information: [0333] User Account Identifier: Identifier that identifies a user account. [0334] Password: Password, or other personal identifier, used to authenticate the user account. For example, it may an alphanumerical password, biometric data (e.g., fingerprint, etc.). In some embodiments, readings or measurements from the blood metrics measurement apparatus 902 may be used to authenticate the user account. [0335] Device Identifier(s): Identifier(s) that identify one or more blood metrics measurement apparatus' associated with the user account and/or user device. [0336] Name: A name of the user. [0337] DOB: A date of birth of the user. [0338] Age: An age of the user. [0339] Gender: Gender of the user (e.g., female, male, transgender, or the like). [0340] Weight: A weight of the user. [0341] Height: A height of the user. [0342] Skin color: A skin color of the user. [0343] Activity Level: An activity level of the user (e.g., sedentary, lightly active, active, very active, and so forth). [0344] Geographic location: A location of the user (e.g., as determined by a location service and/or specified by the user). [0345] Blood Pressure Profile: Hypertensive, Hypotensive, Normal or unknown. [0346] Blood Glucose Profile (e.g., Diabetes information) [0347] Wrist circumference: Circumference of the user's wrist.
[0348] The blood metrics measurement apparatus registration records 1716 may include a variety of information associated with users, user accounts, associated blood metrics measurement apparatus' and/or associated user devices. For example, the blood metrics measurement apparatus registration records 1716 may store some or all of the following information: [0349] Apparatus Identifier: Identifier that identifies a blood metrics measurement apparatus. [0350] User Account Identifier: Identifier that identifies a user account and/or user device associated with the blood metrics measurement apparatus. [0351] Geographic location: A current location of the blood metrics measurement apparatus (e.g., as determined by a location service and/or specified by the user). [0352] Settings: One or more settings of the blood measurement metrics apparatus.
[0353] For example, some or all of the settings may be automatically determined based on one or more user account attributes (e.g., height, weight, etc.) and/or by the user.
[0354] The blood pressure model database 1706 may include one or more empirical blood pressure model records 1718. The models may include various types of empirical blood pressure models. For example, a first type may be a non-specific model which does not require calibration in order to be used to calculate arterial blood pressure values. A second type may be a specific model which requires calibration in order to be used to calculate arterial blood pressure values. For example, models of the second type may require information about the user, such age, weight, height, gender, skin color, and/or the like. In some embodiments, the blood pressure records 1718 may store some or all of the following information: [0355] Model Identifier: Identifies an empirical blood pressure model. [0356] Model Type: Identifies a type of model (e.g., non-specific or specific). [0357] Model Parameters: Various model parameters (e.g., decision node parameters) and tree structures used to calculate the arterial blood pressure values based on the sets of feature vectors and/or other related information (e.g., gender, age, weight, height, skin color, etc.). Example tree structures are shown in FIG. 26. [0358] Apparatus Identifier(s): Identifier(s) that identify one or more blood metrics measurement apparatus' using the empirical blood pressure model. [0359] User Account Identifier(s): Identifier(s) that identify one or more user account(s) associated with the blood metrics measurement apparatus that use the empirical blood pressure model.
[0360] The model generation and selection module 1708 may be configured to generate empirical blood pressure models and/or identify an empirical blood pressure models stored in the records 1718 to provide to a blood pressure calculation system. For example, an empirical blood pressure model may be identified in response to a request from a user device or a blood pressure calculation system. In various embodiments, the model generation and selection module 1708 identifies a non-specific empirical blood pressure model which does not require further calibration prior to being used by a blood pressure calculation system to calculate arterial blood pressure values. In some embodiments, the model generation and selection module 1708 includes some or all of the functionality of a wave selection module, feature extraction module, blood pressure processing module, along with associated features and/or functionality (e.g., a signal database, wave database, and so forth).
[0361] In one example, an empirical blood pressure model may be identified from a set of models (e.g., stored in the records 1718) by the model generation and selection module 1708 based upon a modified Random Forests algorithm using random decision trees in a regression mode. For example, decision trees may comprise a plurality of nodes for decision making. Two examples of decision trees are shown in FIG. 26. Each internal node may correspond to one comparison point of the input values (e.g., one feature (F.sub.i, i=1, . . . ,N) a feature vector) and edges may represent the path of the outcomes. Leaves (the end points) may represent desired target points. In this example, target points are equivalent to particular blood pressure values.
[0362] In some embodiments, a specified (or, predetermined) number of decision trees are constructed. Each decision tree may result in a blood pressure target, but majority voting may give a single blood pressure estimate (or, calculation). In a model training (or, "calibration") phase, the decision node parameters (a.sub.1, . . . ,a.sub.N, b.sub.1, . . . ,b.sub.N, . . . ), combination of features, and the structure of the decision trees may be learned, for example, by randomly subsampling the training data and the features, and tracking the error in an unobserved part of the training data. In some embodiments, these parameters and tree structures may comprise the empirical blood pressure models generated and/or stored the blood metrics server 906, and/or used by the blood pressure calculation system 1206 to calculate arterial blood pressure values.
[0363] In some embodiments, grown decision trees may learn highly complex patterns. However, they may be prone to overfitting (e.g., sensitive to noise). The model generation and selection module 1708 may address this problem. For example, a tree bagging (or, bootstrap aggregating) technique, in which samples are randomly selected with replacement or without replacement from a training list. The assignment of unseen samples may be performed by taking a majority vote. Although one decision tree may be sensitive to noise, using multiple decision trees and averaging results may significantly decrease the variance as long as the trees are not correlated. In some embodiments, the model generation and selection module 1708 combines tree bagging with feature bagging where the subset of features are also selected randomly with replacement. In some embodiments, the model generation and selection module 1708 may randomly subsample the training dataset without replacement. In some embodiments, the model generation and selection module 1708 may include several parameters to be tuned for a minimum number of leaves and/or number of trees.
[0364] In some embodiments, during a model calibration phase, the datasets coming from different subjects may be randomly partitioned to training sets and test sets. In some embodiments, generally, 70-80% of the datasets may be used in training, and the datasets of the same subject may not be both training and testing sets, i.e., test sets and training sets should be disjointed. If the size of the data (number of patients or users) is limited (e.g., .ltoreq.150), a bucketwise randomization may be applied (e.g., to help sure that there is enough training data for certain ranges of blood pressure). For example, the bucket edges may be selected as 80, 100, 130, 160, 180, 230. Then, for example, random partitioning may be separately applied to data sets within a certain bucket so that the ratio of training and test samples will be the same.
[0365] In some embodiments, the parameters of the tree bagging technique are optimized using k-fold cross validation. Generally, in k-fold cross validation, the existing dataset may be randomly partitioned into training and testing k-times (e.g., k.gtoreq.1). This may be done, for example, to decrease the bias of the results to the selected test and training sets. Error metrics between the ground truth and estimated blood pressure values in test dataset are averaged k-times. Those metrics may include, and are not limited to, mean square error (MSE, Example Equation 1, shown below), root mean square error (RMSE, Example Equation 2, shown below), median absolute deviation (MAD, Example Equation 3, shown below), and/or coefficient of determination (R.sup.2, Example Equation 4, shown below). In some embodiments, the model giving optimal (or, the lowest) error values may be selected and used by the blood pressure calculation system 1206 to calculate arterial blood pressure values.
MSE ( y ^ ) = 1 N i = 1 N ( y i - y ^ i ) 2 Example Equation 1 RMSE ( y ^ ) = 1 N i = 1 N ( y i - y ^ i ) 2 Example Equation 2 MAD ( y ^ ) = median ( y - y ^ ) Example Equation 3 R 2 ( y ^ ) = 1 - M S E ( y ^ ) var ( y ) Example Equation 4 ##EQU00021##
[0366] In some embodiments, the model generation and selection module 1708 minimizes overfitting. For example, each fold (or, k-fold) may be individually trained. Feature sets may be identified for each fold through backward or floating search optimizations where features are added or removed (e.g., to improve cost). In some embodiments, in order to enable cross-validation (CV) or bootstrapping (BS) performance match an unseen test set accurately, a measure of test-train cost may be calculated. This may be performed, for example, to inform feature selection approaches for adding or removal of features. For example, Test-Train cost may be defined as
C T T = 1 - R M S E Train R M S E T e s t , ##EQU00022##
where C.sub.TT may be close to "0" if test error and train error tracked each other closely, and may be "1" if train error was smaller than test (e.g., overfitting). This may be applied, for example, when a new feature is added (e.g., not present in the current optimal feature set), or when an existing featured is removed (e.g., from the current optimal feature set). In some embodiments, the corresponding condition which must be met to continue adding or removing a feature may be calculated as current cost<(previous cost+.alpha. F C.sub.TT), where .alpha. is the weight associated with the Test-Train cost, and F is "1" for testing feature removal and "-1" for testing feature addition. While this may have the effect of reducing model complexity while increasing RMSE, model complexity is not directly minimized, which could have the desired effect of allowing model complexity to be relatively large without penalty as long as C.sub.TT is relatively small.
[0367] In some embodiments, the communication module 1710 may be configured to send requests to and receive data from one or a plurality of systems. The communication module 1710 may send requests to and receive data from a systems through a network or a portion of a network. Depending upon implementation-specific or other considerations, the communication module 1710 may send requests and receive data through a connection (e.g., the communication network 908, and/or the communication link 910), all or a portion of which may be a wireless connection. The communication module 1710 may request and receive messages, and/or other communications from associated systems.
[0368] FIG. 18 depicts a flowchart 1800 of an example method of operation of a blood metrics server (e.g., blood metrics server 906) according to some embodiments.
[0369] In step 1802, the blood metrics server receives and processes user account and/or blood pressure measurement apparatus registration requests. In some embodiments a communication module (e.g., communication module 1712) receives the requests. An example registration method is shown in FIG. 20.
[0370] In step 1804, the blood metrics server receives and processes a request for an empirical blood pressure model. In some embodiments, the communication module receives the request from a user device (e.g., user device 904) and/or blood pressure calculation system (e.g., blood pressure calculation system 1206). For example, the user device and/or blood pressure calculation system may periodically request an updated model.
[0371] In step 1806, the blood metrics server selects an empirical blood pressure model. For example, the model may be selected in response to the request, although in some embodiments, the blood metrics server may automatically select a model in order to push down an updated model to the user device. In some embodiments, a model generation and selection module (e.g., model selection 1708) performs the selection.
[0372] In step 1808, the blood metrics server provides the selected empirical blood pressure to the user device and/or blood pressure calculation system. In some embodiments, the communication module provides the selected empirical blood pressure model.
[0373] FIG. 19 depicts a flowchart 1900 of an example method of operation of a blood metrics server (e.g., blood metrics server 906) according to some embodiments.
[0374] In step 1902, the blood metrics server receives a first signal (e.g., a single-channel or multi-channel PPG signal) and a second signal (e.g., a single-channel or multi-channel PPG signal) for each of a plurality of users (e.g., training subjects). For example, the first signal may comprise a 7-channel PPG signal generated from light energy emitted at seven different wavelengths (e.g., 523 nm, 590 nm, 623 nm, 660 nm, 740 nm, 850 nm, 940 nm) from one or more light sources (e.g., seven different LEDs), and the second signal may comprise a single-channel PPG signal generated from light energy generated from an additional light source (e.g., LED) at the same, or substantially similar, wavelength as one the wavelengths of the first signal. Using multi-channel signals and/or different light sources may help ensure, for example, that good quality signals may be obtained in a variety of circumstances (a user moving, walking, running, sleeping, and so forth). In some embodiments, a communication module (e.g., communication module 1712 receives the first and second signals from a blood metrics measurement apparatus (e.g., blood metrics measurement apparatus 902) and/or other blood pressure measurement devices, such as other types of non-invasive devices (e.g., sphygmomanometer) and/or invasive devices (e.g., catheters, tubes, etc.).
[0375] In step 1904, the blood metrics server pre-processes (or filters) the first and second signals. In some embodiments, a pre-processing module filters the first and second signals.
[0376] In step 1906-1908, the blood metrics server identifies, for each of the plurality of users, a first subset of waves (or, "high quality" waves) from a first set of waves of the first signal and a second subset of waves (or, "high quality" waves) from a second set of waves of the second signal, each of the first subset of waves representing a separate approximation of an average of the first set of waves over a predetermined amount of time and each of the second subset of waves representing a separate approximation of an average of the second set of waves over the predetermined amount of time. In some embodiments, a model generation and selection module (e.g., model generation and selection module 1708) identifies the subsets of waves.
[0377] In step 19010-1912, the blood metrics server generates, for each of the plurality of users, a first set of feature vectors and a second set of feature vectors. The first set of feature vectors may be generated from the first subset of waves, and the second set of feature vectors may be generated from the second subset of waves. Each of the feature vectors may include measurement values and metric values. For example, the measurement values may correspond to amplitude (e.g., peak-to-peak amplitude, peak amplitude, semi-amplitude, root mean square amplitude, pulse amplitude, etc.) or location points of a particular wave (e.g., a corresponding high quality wave), and the metric values may be generated from metric functions that use at least one of the measurement values. In some embodiments, the model generation and selection module generates the sets of feature vectors.
[0378] In step 1914, the blood metrics server generates a plurality of empirical blood pressure models based on the feature vectors. In some embodiments, the model generation and selection module generates the models.
[0379] In step 1916, the blood metrics server identifies a particular one of the plurality of empirical blood pressure models. The identified model may be used to calculate arterial blood pressure values without requiring further calibration (e.g., a non-specific type of empirical blood pressure model). In some embodiments, the model generation and selection module performs the identification.
[0380] In step 1918, the blood metrics server stores the particular one of the plurality of empirical blood pressure models. In some embodiments, a management module (e.g., management module 1702) stores the particular model in a database (e.g., blood pressure model database 1706).
[0381] FIG. 20 depicts a flowchart 2000 of an example method of operation of a blood metrics server (e.g., blood metrics server 906) according to some embodiments.
[0382] In step 2002, the blood metrics server receives a user account registration request from a user device (e.g., user device 904) via a communication network (e.g., communication network 908). The user account registration request may include, for example, a username (e.g., "jsmith2015), password, full name (e.g., "John Smith"), birthdate, gender, physical characteristics (e.g., height, weight, etc.), and so forth. In some embodiments a communication module (e.g., communication module 1716) receives the user account registration request.
[0383] In step 2004, if the blood metrics server approves the registration request, the blood metrics server may create a new user account registration record (e.g., user account registration record 1714) in a registration database (e.g., registration database 1704) and/or an account for the user. In some embodiments, a management module (e.g., management module 1702) creates the user account registration record.
[0384] In step 2006, the blood metrics server receives a log-in request from the user device. The log-in request may include, for example, the username and password. If the log-in credentials are correct, the blood metrics server logs the user account into the user device. In some embodiments the communication module receives the log-in request.
[0385] In step 2008, the blood metrics server receives a blood metrics measurement apparatus registration request from the user device. The blood metrics measurement apparatus registration request may include, for example, an apparatus identifier that identifies the blood metrics measurement apparatus, and a user account identifier associated the user account and/or user device. The apparatus identifier may be obtained by the user device by a variety of methods such as scanning a physical feature (e.g., a tag, code, or the like) of the blood metrics measurement apparatus, entering the identifier manually via the user device, and/or the like. In some embodiments the communication module receives the blood metrics measurement apparatus registration request.
[0386] In step 2010, the blood metrics server updates the registration database with the blood metrics measurement apparatus registration request by either creating a new record (e.g., a record 1716) or, if the apparatus identifier is already stored in one of the records (e.g., records 1716), updating that particular record. In some embodiments, the management module updates the registration database.
[0387] In step 2012, the blood metrics server provides a registration success message to the user device when the registration database has been updated. In some embodiments the communication module provides the message.
[0388] FIG. 21 depicts an example noisy PPG signal 2100 and an example filtered PPG signal 2150 according to some embodiments.
[0389] FIG. 22 depicts an example set of waves 2200 of a PPG signal and an example high quality wave 2202 selected from the set of waves 2200 according to some embodiments. FIG. 22 further depicts an example bi-Gaussian fitted signal 2204 according to some embodiments.
[0390] FIG. 23 depicts example feature points 2302a-u of a wave 2300 according to some embodiments. For example, the feature points 2302a-u may comprise location points and/or amplitudes. In some embodiments, some or all of the features points 2302a-u may correspond to measurement values which may be used by one or more metric functions.
[0391] FIGS. 25A-C show an example selected high quality wave 2500, the first derivative 2502 of the selected high quality wave 2500, and the second derivative 2504 of the selected high quality wave 2500 according to some embodiments.
[0392] FIG. 26 depicts example tree structures 2602 and 2604 of an example empirical blood pressure calculation model 2600 according to some embodiments.
[0393] It will be appreciated that a "user device," "apparatus," "server," "module," "system," and/or "database" may comprise software, hardware, firmware, and/or circuitry. In one example, one or more software programs comprising instructions capable of being executable by a processor may perform one or more of the functions of the user devices, servers, modules, systems, and/or databases described herein. In another example, circuitry may perform the same or similar functions. Alternative embodiments may comprise more, less, or functionally equivalent user devices, apparatus, servers, modules, systems, and/or databases, and still be within the scope of present embodiments. For example, the blood metrics measurement apparatus 902 may include some or all of the functionality of the user device 904 (e.g., user interface module 1202, blood pressure calculation system 1206, etc.), the blood metrics server 906 may include some or all of the functionality of the blood pressure calculation system 1206, and so forth.
[0394] Various embodiments described herein eliminate or reduce sensitivity by using a combined array of photodiodes (energy receivers) and LEDs (energy transmitters). An array of photodiodes and LEDs may be utilized to choose an optimal or preferred path. Using this method, a light path area may be chosen to yield the preferred signal in a wider area.
[0395] FIG. 29A depicts a schematic diagram of an example sensor system 2900 according to some embodiments. The sensor system 2900 may be disposed in the blood metrics measurement apparatus 200 (see FIG. 2) or the blood metrics measurement apparatus 902 (see FIG. 10). For example, the sensor system 2900 may comprise the energy transmitter 204 and the energy receiver 206 shown in FIG. 2. In another example, the sensor system 2900 comprises the energy transmitter 1004 and the energy receiver 1006.
[0396] The sensor system 2900 may include an optical sensor array 2902 including one or more energy transmitters 2904 (e.g., LEDs or other lights sources) and one or more corresponding energy receivers 2906 (e.g., photodiodes or other photosensors). It will be appreciated that each pair of corresponding energy transmitters and energy receivers (e.g., energy transmitter 2904 and energy receiver 2906) may be referred to as an LED-PD system. The sensor system 2900 may include any number of such LED-PD systems.
[0397] The energy transmitters 2904 may each comprise any number of LEDs (e.g., between 2-6 LEDs) configured to transmit a variety of wavelengths into tissue of a user. The energy transmitters 2904 may be or include the energy transmitter 204 (see FIG. 2) and/or the energy transmitters 1004 (see FIG. 10A). The corresponding energy receivers 2906 may each comprise one or more photodiodes which receive returning light from the corresponding energy transmitters 2904 after passage through the user's tissue. The energy receivers 2906 may be or include the energy receiver 206 (see FIG. 2) and/or the energy receiver 1006 (see FIG. 10A).
[0398] In some embodiments, the energy transmitter 2904 and the energy receiver 2906 may be spaced from each other at a predetermined distance (e.g., 2 mm), and/or adjacent pairs of the energy transmitters 2904 and the energy receivers 2906 may be spaced at a predetermined distance (e.g., 15 mm). In various embodiments, by measuring a pulse transit time (PTT.sub.m) based on two signal patterns obtained from at least two pairs of the energy transmitters and energy receivers, it is possible to obtain a pulse wave velocity (PWV) by dividing a distance between the two measuring areas (e.g., measuring points). Measuring areas may correspond to mid-points between any energy transmitter and any energy receiver (e.g., between any energy transmitter and any energy receiver of the two pairs). The pulse wave velocity (PWV) may be used to obtain blood metrics including, but not limited to, blood pressure or other blood metric.
[0399] In various embodiments, the sensor system 2900 may be mounted on a user's wrist along the radial artery 2908, and/or another area of the user's body along arterial pathways (e.g., the ulnar artery). The sensor system 2900 may include an accelerometer and gyroscope (e.g., motion sensor 1012 of FIG. 10) for detecting position and orientation information. The sensor system 2900 may include a pressure sensor (e.g., pressure sensor 1008 of FIG. 10).
[0400] In some embodiments, at least one LED within each LED-PD system may be configured to sample data from tissue at a very high frequency (upwards of 1 KHz), and other LEDs may sample at a lower rate (around 50-100 Hz). The channels sampling data at high frequency may be referred to as the fast LED channels, and the channels sampling data at lower frequency may be referred to as slow LED channels.
[0401] It will be appreciated that the velocity of blood moving through a user's arteries may vary within a range of 5 m/s to 15 m/s. This velocity may be correlated with various factors such as, for example, age, arterial stiffness, and/or blood pressure (some of which may be interrelated). Different sensor locations in a PPG measurement device may cause a significant difference in signal characteristics. Some locations of sensors may prevent PPG sensors from receiving a high-quality signal that enables high resolution analysis in time and frequency domains. The signal shape of capillary tissue, venous tissue and of arterial tissue may be different. In some embodiments, a 5 mm translation may be the difference between an arterial signal and a signal that is practically unusable, and even simple measurements (e.g., heart rate measurement) may become difficult.
[0402] In order to obtain an arterial signal with a higher degree of flexibility in device placement, the blood metrics measurement apparatus 2902 may utilize an array of LED/photodiode pairs (or, "channels") that facilitate measurement of PPG signals at any number of locations (e.g., 12 different locations). FIG. 29A shows an example configuration of an LED-PD sensor array used in the blood metrics apparatus 2902. FIG. 29A shows a sideways profile of the blood metrics apparatus 2902 of a user. FIG. 29B shows an example configuration of sensors 2904 and 2906 mounted on the left wrist (facing the palm) mounted along the direction of the radial artery 2910 of a user.
[0403] In the example of FIGS. 29A and 29B, the LED-PD pairs 2904 and 2906 are separated by a known fixed distance (e.g., 15 mm), which allows a pulse wave velocity (or "PWV") to be derived from a modified pulse transit time. It will be appreciated that the LED-PD pairs may be separated by any number of fixed distances, including, for example, 8 mm-20 mm.
[0404] The pairs of LED-PD systems or channels may operate at a high sampling rate (e.g. 1.4 KHz) where the LEDs are activated at this sampling rate, and the photodiode measures light returned from tissue. In one example. twelve PPG time series are generated in this manner, one from each LED-photodiode pair. In one example, in order to be able to resolve a 3 mms time shift, a signal of at least about 700 Hz may be utilized. To avoid introducing error when sampling that signal, the sampling rate may be set at the nyquist frequency for that signal, which may correspond to 1.4 KHz.
[0405] FIG. 29C depict a reflection-based photoplethysmogram sensor layout in some embodiments. Energy transmitter 2912 (e.g., energy transmitter 2904) may emit light into tissue of a user. The light is reflected, gathered, and sensed by energy receiver 2916 (e.g., energy receiver 2906). The cross 2914 denotes the center point (e.g., measuring area) between these elements where the light is reflected. Blood vessels in the close vicinity of the cross 2914 (center point) may determine the majority of the signal's characteristics. The penetration depth of light may depend on the energy transmitter's directionality and direction, as well as its wavelength. Having a single point (or a single area) of measurement can be highly sensitive to positioning and motion.
[0406] FIG. 30A depicts an example arrangement of energy transmitters and energy receivers in a sensor system 370 according to some embodiments. As discussed regarding FIG. 29A, the sensor system 370 may be disposed in the blood metrics measurement apparatus 200. For example, the sensor system 370 may comprise the energy transmitters 204 and the energy receivers 206 shown in FIG. 2. The sensor system 370 may include an optical sensor array including a plurality of energy transmitters 3002a-3002h (collectively referred to as 3002) (e.g., LEDs or other lights sources) and a plurality of energy receivers 3004a-3004b (collectively referred to as 3004) (e.g., photodiodes or other photosensors). In this example, the number of the energy transmitters 3002 is different from the number of the energy receivers 3004. The horizontal direction 3007 of FIG. 30A corresponds to a circumferential direction of a wrist of a subject, when the sensor system 370 is attached on a surface of the wrist.
[0407] The layout of FIG. 30A may yield high sensitivity across a wide area. The area 3008 is an area in which a maximum or increased signal quality point can be identified and sensed.
[0408] In one example, since the energy emitted by one of the energy transmitters 3002 is reflected by tissue of a user and the reflected energy is incident on one of the two energy receivers 3004, the energy incident on the energy receiver 3004 can be used to measure a state approximately at a midpoint (e.g., a measuring area) 3006 between the energy transmitters 3002 and the energy receiver 3004. The following are examples from FIG. 30A:
(1) Energy transmitter 3002a may emit energy that is generally reflected by measuring area 3006a and subsequently received by energy receiver 3004a. (2) Energy transmitter 3002b may emit energy that is generally reflected by measuring area 3006b and subsequently received by energy receiver 3004b. (3) Energy transmitter 3002c may emit energy that is generally reflected by measuring area 3006c and subsequently received by energy receiver 3004a. (4) Energy transmitter 3002d may emit energy that is generally reflected by measuring area 3006d and subsequently received by energy receiver 3004b. (5) Energy transmitter 3002e may emit energy that is generally reflected by measuring area 3006e and subsequently received by energy receiver 3004a. (6) Energy transmitter 3002f may emit energy that is generally reflected by measuring area 3006f and subsequently received by energy receiver 3004b. (7) Energy transmitter 3002g may emit energy that is generally reflected by measuring area 3006g and subsequently received by energy receiver 3004a. (8) Energy transmitter 3002h may emit energy that is generally reflected by measuring area 3006h and subsequently received by energy receiver 3004b.
[0409] The measuring areas 3006a-h (collectively referred to as 3006) may each correspond to a midpoint between one of the energy transmitters 3002a-h and one of the energy transmitters 3004a-b, respectively. In some embodiments, energy is generated from one energy transmitter (e.g., 3002a) at a different time than when energy is generated by another energy transmitter (e.g., 3002e). An energy receiver (e.g., 3004a) operates to receive energy (e.g., from one or more of the energy transmitters) in order to measure a state at one or more (e.g., two) measuring areas. Since a state at an arbitrary point within an area defined by the measuring areas 3006a-h can be obtained from a linear combination of states at two or more measuring areas 3006a-h, the sensor system 3000 may be capable of measure a state at points within a measuring area 3008 defined by the measuring areas 3006a-h.
[0410] When using a sensor system for measurement of a state (e.g., through arterial tissue) of a user, it may be important to measure the state of tissue at a specific measuring area, because signal shape received through a specific tissue (e.g., arterial tissue) is largely different from signal shape received through tissue of other types (e.g., capillary tissue and venous tissue). It will be appreciated that measuring an inappropriate tissue may cause inaccurate measurement. Advantageously, by arranging a plurality of energy transmitters 3002 and a plurality of energy receiver 3004 in multi-dimensional directions (e.g., in a circumferential direction of a wrist and an extending direction of an arm), it is possible to obtain a multi-dimensional measuring area 3008, which allows for multi-dimensional displacement of the sensor system 370 on a measuring surface of the user. Furthermore, since all or some of the multi-dimensional measuring area 3008 may be obtained, it is possible to obtain measurement signals at two or more different measuring points along a single blood vessel (e.g., a radial artery) even if the blood vessel does not extend along a predetermined arrangement direction of the measuring points. As a result, in order to obtain some blood metrics (e.g., blood pressure), the system is enabled to utilize signals at two or more different measuring areas.
[0411] In some embodiments, signals are obtained at any number of measuring areas 3006a-h. A quality of signal obtained at each of the measuring area 3006a-h may be quantitatively assessed according to a predetermined parameter (e.g., SNR). Signals of which quality that are above a certain threshold may be selected. Thereafter, among the selected signals, two signals of which measuring areas 3006 are closest to each other may be selected. Subsequently, the two signals obtained from the closest measuring areas 3006 are averaged, and the averaged signal may be identified as the best quality signal reflecting a state in interest.
[0412] In various embodiments, to choose one or more signal points (e.g., signals generated by energy reflected by a measurement area), signal quality may be assessed. Signals may be associated with a measurement area (e.g., one of measurement areas 3006a-h) and only those signals are selected that are above a specific threshold. Of those signals, two that are reflected by measurement areas that are close together may be selected. The signals (or properties thereof) may be averaged and the point of measurement may be at the measurement area.
[0413] Although the measurement area is depicted as crosses in FIG. 30A, it will be appreciated that energy reflected at the measurement area may be somewhat dispersed and energy may be received in an area larger than a specific point.
[0414] FIG. 30B depicts an example arrangement of energy transmitters and energy receivers in a sensor system 3010 according to some embodiments. The sensor system 3010 may be disposed in the blood metrics measurement apparatus 200. For example, the sensor system 3010 may comprise the energy transmitters 204 and the energy receivers 206 shown in FIG. 2. The sensor system 3010 may include a top optical sensor array 3012a (e.g., including energy transmitters 3014a-h and energy receivers 3016a and b) and a bottom optical sensor array 3012b (e.g., including energy transmitters 3014a-d and i-l and energy receivers 3016c and d). It will be appreciated that energy transmitters 3014a-d may be utilized in both sensor arrays 3012a-b thereby allowing fewer energy transmitters when compared to two separate arrays that do not share energy transmitters.
[0415] In this example, the top optical sensor array 3012a may include a plurality of energy transmitters (e.g., LEDs or other lights sources) and a plurality of energy receives (e.g., photodiodes or other photosensors). The bottom optical sensor array 3012b may include a plurality of energy transmitters (e.g., LEDs or other lights sources) and a plurality of energy (e.g., photodiodes or other photosensors), in a symmetrical manner. The sensor system 3010 includes energy transmitters 3014a-d (collectively referred to as 3014), and the energy transmitters 3014a-d are shared by the top optical sensor array 3012a and the bottom sensor array 3012b.
[0416] Advantageously, since the energy transmitters 3014a-d are shared by the top optical sensor array 382a and the bottom sensor array 382b, the number of the energy transmitters 384a-d can be reduced but obtain measurement at two separate measuring areas 3018 and 3020, which leads to energy saving for transmission of energy from energy transmitters 3014a-d. Also, since the number of the energy transmitters can be reduced, measurement at two separate measuring areas can be achieved with a smaller device coverage on the user.
[0417] Furthermore, since all or some of the multi-dimensional measuring areas 3018 and 3020 may be obtained, it is possible to obtain measurement signals at two or more different measuring points along a single blood vessel (e.g., a radial artery) even if the blood vessel does not extend along a predetermined arrangement direction of the measuring points.
[0418] FIG. 30C depicts an example arrangement of energy transmitters and energy receivers in a sensor system 3022 disposed on a circuit board of an apparatus according to some embodiments. For example, the sensor system 3022 may comprise the circuit board, and the energy transmitters 3024a-h and energy receivers a-h. In this example, the top array comprises an energy transmitter 3024a positioned next to energy receivers 3026a-b followed by two more energy transmitters 3024b-c. The energy transmitters 3024b-c are followed by energy receivers 3026c-d which is followed in turn by energy transmitter 3024d. The bottom array comprises an energy transmitter 3024e positioned next to energy receivers 3026e-f followed by two more energy transmitters 3024f-g. The energy transmitters 3024f-g are followed by energy receivers 3026g-h which is followed in turn by energy transmitter 3024h. These arrays enable measuring areas for receiving signals. The measuring areas allow for arteries (e.g., radial artery) or veins to be traverse the arrays in many directions (e.g., not just vertically).
[0419] FIG. 30D depicts an example of a single array in some embodiments. The array includes LED 1 (e.g., energy transmitter) followed by PD A-B (e.g., photodiodes), followed by LEDs 2-3, Photodiodes C-D, and LED 4. Measuring areas are represented by circles. At the first measuring area (starting on the left of FIG. 30D), energy transmitted by LED 1 (at any number of wavelengths) is reflected by tissue of a user to photodiode B. At the second measuring area, energy transmitted by LED 2 (at any number of wavelengths) is reflected by tissue of a user to photodiode A. Similarly, at the third measuring area, energy transmitted by LED 3 is reflected by the tissue of the user and received by PD B. At the fourth measuring area, energy transmitted by LED 2 is reflected by the tissue of the user and received by PD C. At the fifth measuring area, energy transmitted by LED 3 is reflected by the tissue of the user and received by PD D and at the sixth measuring area, energy transmitted by LED 4 is reflected by the tissue of the user and received by PD C.
[0420] LEDs 1-4 may correspond to energy transmitters 3024a-d or energy transmitters 3024e-h. PDs A-D may correspond to energy receivers 3026a-d or energy receivers 3026e-h.
[0421] It will be appreciated that an energy receiver may provide energy (e.g., at any number of wavelengths) into tissue of a user in different directions and be received by different energy receivers. For example, LED2 may project first energy in a first direction that is reflected at the second measuring area (LED 2 PD A) to be received by PD A. Similarly, LED2 may project energy at a second direction that is reflected at the fourth measuring area (LED 2 PD C) to be received by PD C. In another example, LED3 may project first energy in a first direction that is reflected at the third measuring area (LED 3 PD B) to be received by PD B. Similarly, LED3 may project energy at a second direction that is reflected at the fifth measuring area (LED 3 PD D) to be received by PD D. This creases a larger measuring area to enable tracking of any number of arteries and/or any number of veins even if the arter(ies) and/or vein(s) do not necessarily travel laterally up the user's wrist.
[0422] FIG. 30E is an enlarged view of the energy transmitters and energy receivers in the sensor system 3028. The sensor system 3028 may include a sensor array including a plurality of energy transmitters 3030a-3030h (e.g., LEDs or other lights sources) and a plurality of energy receivers 3032a-3032h (e.g., photodiodes or other photosensors) which means that the number of the energy transmitters 3030 is the same as the number of the energy receivers 3032.
[0423] According to the arrangement of the energy transmitters 3030a-3030h (collectively referred to as 3030) and the energy receivers 3032a-3032h (collectively referred to as 3032), twelve measuring areas are obtained. Those twelve measuring areas include six measuring points (denoted by crosses) on a top row corresponding to: i) a pair of the energy transmitter 3030a and the energy receiver 3032b; ii) a pair of the energy transmitter 3030b and the energy receiver 3032a; iii) a pair of the energy transmitter 3030c and the energy receiver 3032b; iv) a pair of the energy transmitter 3030b and the energy receiver 3032c; v) a pair of the energy transmitter 3030c and the energy receiver 3032d; and vi) a pair of the energy transmitter 3030d and the energy receiver 3032c. Also, the twelve measuring points include six measuring points (denoted by crosses) on a bottom row corresponding to: i) a pair of the energy transmitter 3030e and the energy receiver 3032f; ii) a pair of the energy transmitter 3030f and the energy receiver 3032e; iii) a pair of the energy transmitter 3030g and the energy receiver 3032f; iv) a pair of the energy transmitter 3030f and the energy receiver 3032g; v) a pair of the energy transmitter 3030g and the energy receiver 3032h; and vi) a pair of the energy transmitter 3030h and the energy receiver 3032g. As a result, a measuring area 3040 defined by the twelve measuring points is obtained.
[0424] Accordingly, similarly to the sensor system 3000 depicted in FIG. 30A and the sensor system 3010 depicted in FIG. 30B, the sensor systems (e.g., 3022 and 3028) depicted in FIGS. 30C-E can obtain multi-dimensional measuring area by multi-dimensional arrangement of the energy transmitters 3030 and the receivers 3032, which allow for more flexible placement of the sensor system on a surface of a user.
[0425] Also, similarly to the sensor system 3000 depicted in FIG. 30A and the sensor system 3010 depicted in FIG. 30B, timing to generate energy from one energy transmitter (e.g., 3030a) may be differentiated from timing to generate energy from another energy transmitter (e.g., 3030c) when an energy receiver (e.g., 3032b) operates to receive energy, in order to measure the state of tissue at a single measuring point. In some embodiments, the energy transmitters 3030 and the energy receivers 3032 are arranged such that the measuring points are arranged at a constant distance (e.g., 5 mm) in each row, and that a constant distance (e.g., 15 mm) is formed between the rows, so as to sufficiently cover a region of tissue in interest (e.g., a radial artery).
[0426] In the embodiment shown in FIG. 30E, each of the top row and the bottom row of the arrangement of the energy transmitters 3030 and the energy receivers 3032, the sensor system 3028 is configured such that two energy receivers 3032 are consecutively arranged and the two energy receives 3032 are disposed between two the energy transmitters 3030. Such an arrangement of the energy transmitters 3030 and the energy receivers 3032 allows for more dense arrangement thereof, in comparison to regular arrangement of pairs of one energy transmitter 3030 and one energy receiver 3032 (i.e., the energy transmitters 3030 and the energy receivers 3032 are alternately arranged in each of top and bottom rows). Further, since two energy receivers 3032 are consecutively arranged and two energy transmitters 3030 are consecutively arranged in the configuration of the sensor system 3028, some circuit components may be shared by the two consecutive energy receivers 3032, some circuit components may be shared by the two consecutive energy transmitters 3030.
[0427] FIG. 31A-B depict experimentations carried out to obtain an optimum or preferred location of a sensor system (such as the sensor systems 3000, 3010, 3022, and 3028 depicted in FIG. 30A-E), when the sensor system is used for measuring states of a radial artery on a wrist (e.g., left wrist) of a subject (e.g., a user). Measurement of the state of the radial artery on a wrist is useful for measuring metrics such as pulse rate, blood pressure, and oxygen saturation, because the radial artery on a wrist is close to a body surface of the subject and thus more accessible by the sensor system employing optical signal transmission.
[0428] FIG. 31A depicts spacing of energy transmitters and energy receivers in some embodiments. Spacing may be based on a methodology of a test to obtain an optimum or preferred measuring area to measure states of a radial artery on a left wrist of subjects. The x-y coordinate shown in FIG. 31A corresponds to an area on a surface of a left wrist when a left palm faces upward. In this example, the x and y axes of FIG. 31A correspond to a direction of a crease between the left wrist and left palm and a direction in which a left arm extends, respectively. The origin of the x-y coordinate in this example corresponds to a left edge of the left wrist on the crease. Units in FIG. 31A are in millimeters (mm). Two smaller squares correspond to locations of energy transmitters (e.g., LEDs and/or other lights sources). Two larger black squares correspond to location of energy receivers (e.g., photodiodes and/or other photosensors). The circle corresponds to the center of the sensor system.
[0429] In the test, the width (W) of the left wrist was measured for each subject, and then the center of the sensor system was positioned at W/2 in +x axis, and at (.times.0+15) in -y axis (where x.sub.0 is a distance of a measuring gauge from the crease and initially 10 mm). Thereafter, while adjusting y.sub.0 (where y.sub.0 is a distance of the center from the wrist median W/2) and x.sub.0, three measuring points that obtained high quality signals (as compared to other obtained signals from transmitters and receivers in other positions) were identified. According to at least one test, it was found that the three highest quality signals were obtained in a range of 20-30 mm in the crease direction from left edges of left wrists, and in a range of 15-40 mm in an arm-extending direction from the creases between left wrists and left palms.
[0430] FIG. 31B depicts optimum or preferred measuring areas to measure states of a radial artery on a left wrist of subjects. In the tests results shown in FIG. 31B, contours of left wrists of subjects (about 30 subjects) were traced, and the traced contours of left lists were delineated and reparameterized. Then, left edges of the left wrists were aligned (e.g., rotated and/or translated to minimize distance) to a common template. The horizontal axis of FIG. 31B corresponds to a distance in a crease direction of left wrists in millimeters (mm), and vertical axis of FIG. 31B corresponds to a height of left wrists in millimeters (mm). The circles in FIG. 31B shows most optimal or preferred measuring area (where the most high-quality signal was obtained) of each subject. According to the test, it was found that the most high-quality signals were obtained in a range of 5-20 mm in the crease direction from left edges of left wrists.
[0431] According to the tests depicted in FIG. 31A-B, in an embodiment, a measuring area defined by measuring points of a sensor system (e.g., the sensor systems 3000, 3010, 3022, and 3028 depicted in FIG. 30A-E) is configured to cover at least part of 5-30 mm in a crease direction from a left edge of left wrists of subjects and at least part of 15-40 mm in an arm-extending direction from the creases between left wrists and left palms of subjects. In a more particular embodiment, a multi-dimensional region defined by the plurality of energy transmitters (e.g., any number of the energy transmitters 3030a-h in FIG. 30E) ranges at least 20 mm in a direction of a wearable member (e.g., central unit 402 in FIG. 4) corresponding to a circumferential direction of the wrist. In another more particular embodiment, a multi-dimensional region defined by the energy transmitters (e.g., any number of the energy transmitters 3030a-h in FIG. 30E) ranges at least 15 mm in a direction of the wearable member (e.g., central unit 402 in FIG. 4) corresponding to an extending direction of an arm of the user.
[0432] In another embodiment, the sensor array 404 of the apparatus 400 depicted in FIG. 4 is located to cover this optimum or preferred range, for example, when apparatus 400 is attached to left wrists of subjects such that the screen 410 faces along the back of left hands. In a more specific implementation, a center the sensor array 404 in a circumferential direction of wrist is positioned at or adjustable to a position that is offset from 180.degree. opposite position of the center of the screen 410, when the apparatus is wound around left wrists with fit such that the apparatus 400 does not freely move around wrists.
[0433] FIG. 32 depicts a flowchart 3200 of an example method of separating source signals from measured signals by a sensor system (e.g., the sensor systems 3000, 3010, 3022, and 3028 depicted in FIG. 30A-E) and identifying at least an arterial signal from the separated source signals according to some embodiments. In one example, the source separating method according to the flowchart 3200 depicted in FIG. 32 may be carried out during the blind source separation performed in step 1542 of the flow chart 1530 depicted in FIG. 15B.
[0434] In step 3202, a wearable blood metrics measurement apparatus (e.g., blood metrics measurement apparatus 200 or sensor systems 3000, 3010, 3022, or 3028 depicted in FIG. 30A-E) measures channel signals each of which corresponds to a state of tissue at a measuring area (e.g., midpoint) of one of energy transmitters and one of energy receivers of a sensor system of the apparatus for a certain period of time (e.g., 10 sec). The wearable blood metrics measurement apparatus measures the channel signals for each of different wavelengths (e.g., infrared, red, green, and blue lights, or the like) and the following steps 3204-3214 may be carried out for each wavelength. For example, in the embodiment of the sensor system shown in FIG. 31E, twelve channel signals corresponding to twelve measuring points are obtained for each of different wavelengths.
[0435] In step 3204, the wearable blood metrics measurement apparatus selects two or more channel signals that have high quality (e.g., high SNR or high signal-to-noise ratio) from the measured channel signals. In some embodiments, a processor of or associated with the wearable blood metrics measurement apparatus selects channel signals that have an SNR higher than a certain threshold from the measured channel signals. Some channel signals may include relatively large noise because of poor positioning of the sensor system, loose sensor placement, and/or interference by foreign materials (such as dusts, sweats, scabs, and tattoos). By choosing channel signals with better SNR, it is possible to obtain more accurate source signals. In some other embodiments, the wearable blood metrics measurement apparatus selects channel signals from which the high quality waves are obtainable through the method depicted in one of the flowcharts 1600, 1620, and 1640 depicted in FIG. 16A-C, respectively, as the high-quality channel signals.
[0436] In step 3206, the wearable blood metrics measurement apparatus phase-shifts the selected channel signals to co-phase the selected channel signals. Depending on the measurement points, the channel signals may be slightly offset in time due to pulse wave velocity. By co-phasing the channel signals, the aspect of the pulse wave velocity can be removed, and more accurate calculation of source signals can be carried out.
[0437] In step 3208, the wearable blood metrics measurement apparatus determines values of one or more parameters to calculate source signals. The parameters to be used may include distance between measuring points of channel signals, correlation of signal wave form between multiple channel signals, O.sub.2 saturation (SpO.sub.2) corresponding to a channel signal (if obtained), rise time corresponding to each channel signal, and respiratory power ratio of a low respiratory frequency range (e.g., 0.1-0.6 Hz) to the entire respiratory frequency range (e.g., 0-2.5 Hz). For example, a long distance between a plurality of measuring points may reduce a parameter value for association of multiple channel signals, because it is more likely that the channel signals come from different blood vessels. In another example, high correlation of wave forms among multiple channel signals may increase a parameter value for association of multiple channel signals, because it is more likely that the channel signals come from the similar signal sources, such as arterial blood flow, a venous blood flow, a capillary blood flow, or motion artifacts.
[0438] In step 3210, the wearable blood metrics measurement apparatus determines values of a mixing matrix based on the determined values of the one or more parameters. The values of the mixing matrix are determined such that one or more source signals include only signal components thereof and exclude signal components of the other source signals and noises.
[0439] In step 3212, which is an optional step, the wearable blood metrics measurement apparatus may update a previously-determined values of a mixing matrix with the values of the mixing matrix calculated in step 3210. Since it is possible to assume that a subject's physiology does not change significantly in a short period of time (e.g. few ten seconds), but may change in a longer period of time (e.g., one hour), depending on the health condition of the subject. It may be beneficial to use a previously-determined values of a mixing matrix to save power to determine the values of the mixing matrix. Thus, it may be preferable to carry out the determination of the mixing matrix values for each certain period that is sufficiently long (e.g., few minutes), especially when the wearable blood metrics measurement apparatus uses a mobile battery. However, calculating source signals based on a previously-determined values of a mixing matrix that was obtained a long time ago (e.g., one hour) may lead to inaccurate calculation of source signals. By updating the mixing matrix, it is possible to more accurately calculate the source signals.
[0440] In step 3214, the wearable blood metrics measurement apparatus calculates each of one or more source signals based on the selected channel signals and the determined values of the mixing matrix. In one embodiment, the wearable blood metrics measurement apparatus calculates each of one or more source signals by multiplying the values of the selected channel signals with the values of the mixing matrix.
[0441] In step 3216, the wearable blood metrics measurement apparatus identifies one or more source signals corresponding to an arterial signal, and optionally one or more source signals corresponding to a venous signal, one or more source signals corresponding to a capillary signal, and/or one or more source signals corresponding to a respiratory signal.
[0442] Some methods may identify the arterial signal from the other signals, in particular, the venous signal. In one example, a method utilizing O.sub.2 saturation (SpO.sub.2) obtained from source signals of different wavelengths (e.g., infrared and read lights) is employed. According to the O.sub.2 method, source signals are sorted based on the value of O.sub.2 saturation (SpO.sub.2), and then source signals with the highest O.sub.2 saturation (or higher than a certain threshold) are identified as an arterial signal and source signals with the lowest O.sub.2 saturation (or lower than a certain threshold) are identified as a venous signal.
[0443] In another example, a rise-time method utilizing a rise time (or "crest" time), which is an average time period of valley to peak in each waveform of the source signal, is employed. According to the rise-time method, source signals are sorted based on the value of the rise time, and then source signals with the shortest rise time (or shorter than a certain threshold) are identified as an arterial signal and source signals with the longest rise time (or longer than a certain threshold) are identified as a venous signal.
[0444] In another example, a respiratory rate method utilizing a ratio of a low frequency component in a respiratory frequency range (0-2.5 Hz) is employed. According to the respiratory rate method, a power ratio of a low respiratory frequency component (0.1-0.6 Hz) of each source signal with respect to the entire respiratory frequency range (0-2.5 Hz) is calculated, and then source signals with the highest power ratio (or higher than a certain threshold) are identified as a venous signal, and source signals with the lowest power ratio (or lower than a certain threshold) are identified as an arterial signal.
[0445] In step 3218, the wearable blood metrics measurement apparatus calculates one or more biological metrics such as blood metrics (e.g., blood pressure, O.sub.2 saturation, arterial stiffness, and so on) based on source signals that are identified as an arterial signal. In addition the wearable blood metrics measurement apparatus calculates one or more biological metrics such as a respiratory rate based on one or more source signals that are identified as a venous signal, an O2 consumption rate based on a source signal that is identified as an arterial signal and a source signal that is identified as a venous signal. Furthermore, when the wearable blood metrics measurement apparatus includes a motion sensor (e.g., an accelerometer, gyroscope, global positioning system, or the like), the wearable blood metrics measurement apparatus can more accurately calculates the respiratory rate, because repetitive motion due to respiration can be obtained from the motion sensor. Moreover when the wearable blood metrics measurement apparatus includes the motion sensor, the wearable blood metrics measurement apparatus includes the motion sensor can calculate step counts based on input from the motion sensor, and perform gain analysis.
[0446] FIG. 33 depicts a flowchart 3300 of an example method of calculating blood pressure according to some embodiments.
[0447] In step 3302, a wearable blood metrics measurement apparatus (e.g., blood metrics measurement apparatus 200) selects at least two signals at different locations corresponding to an arterial signal, which is obtainable through the source separation process described above, the at least two signals associated with corresponding optical sensors of the wearable blood metrics measurement apparatus. In some embodiments, a blood pressure processing module (e.g., blood pressure processing module 1424) of a blood pressure calculation system (e.g., blood pressure calculation system 1206) uses one or more signal selection rules to perform the selection.
[0448] In step 3304, the wearable blood metrics measurement apparatus obtains first signal data (e.g., PPG signal data) from a first signal of the at least two signals (e.g., a pair of signals) over a predetermined period of time. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to obtain the first signal data.
[0449] In step 3306, the wearable blood metrics measurement apparatus obtains second signal data from a second signal of the at least two signals over the predetermined period of time. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to obtain the first signal data to obtain the second signal data.
[0450] In step 3308, the wearable blood metrics measurement apparatus applies a frequency transform function (e.g., a Fourier transform) to each of the first signal data and the second signal data to a transform the first signal data and the second signal data to a frequency domain. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to apply the frequency transform function.
[0451] In step 3310, the wearable blood metrics measurement apparatus determines a first phase value for a first frequency component of the first signal data in the frequency domain. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to perform the determination.
[0452] In step 3312, the wearable blood metrics measurement apparatus determines a second phase value for a second frequency component of the second signal data in the frequency domain
[0453] In step 3314, the wearable blood metrics measurement apparatus determines a phase difference value between the first phase value and the second phase value. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to perform the determination.
[0454] In step 3316, the wearable blood metrics measurement apparatus determines a time shift value between the first signal data and the second signal data based on the phase difference value. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to perform the determination.
[0455] In step 3318, the wearable blood metrics measurement apparatus determines a modified pulse transmit time based on the time shift value between the first signal data and the second signal data, the modified pulse transit time representing a transit time for a pressure wavefront to travel from a first optical sensor of the corresponding optical sensors and a second optical sensor of the corresponding optical sensors. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to perform the determination.
[0456] In step 3320, the wearable blood metrics measurement apparatus determines a pulse wave velocity based on the modified pulse transit time and a distance between the at least two locations. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules to perform the determination.
[0457] In step 3322, the wearable blood metrics measurement apparatus calculates an arterial blood pressure value based on the pulse wave velocity. In some embodiments, the blood pressure processing module of the blood pressure calculation systems uses one or more modified pulse transit time rules and/or one or more blood pressure processing rules (e.g., blood pressure processing rules 1446) to perform the calculation.
[0458] In step 3324, the wearable blood metrics measurement apparatus provides a message including or being based on the arterial blood pressure value. In some embodiments, a user interface module of the blood metrics measurement apparatus provides the message using one or more message rules (e.g., message rules 1448), for example, by displaying the message on a screen (e.g., screen 410).
[0459] The present invention(s) are described above with reference to example embodiments. It will be appreciated that various modifications may be made and other embodiments may be used without departing from the broader scope of the present invention(s). Therefore, these and other variations upon the example embodiments are intended to be covered by the present invention(s).
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014
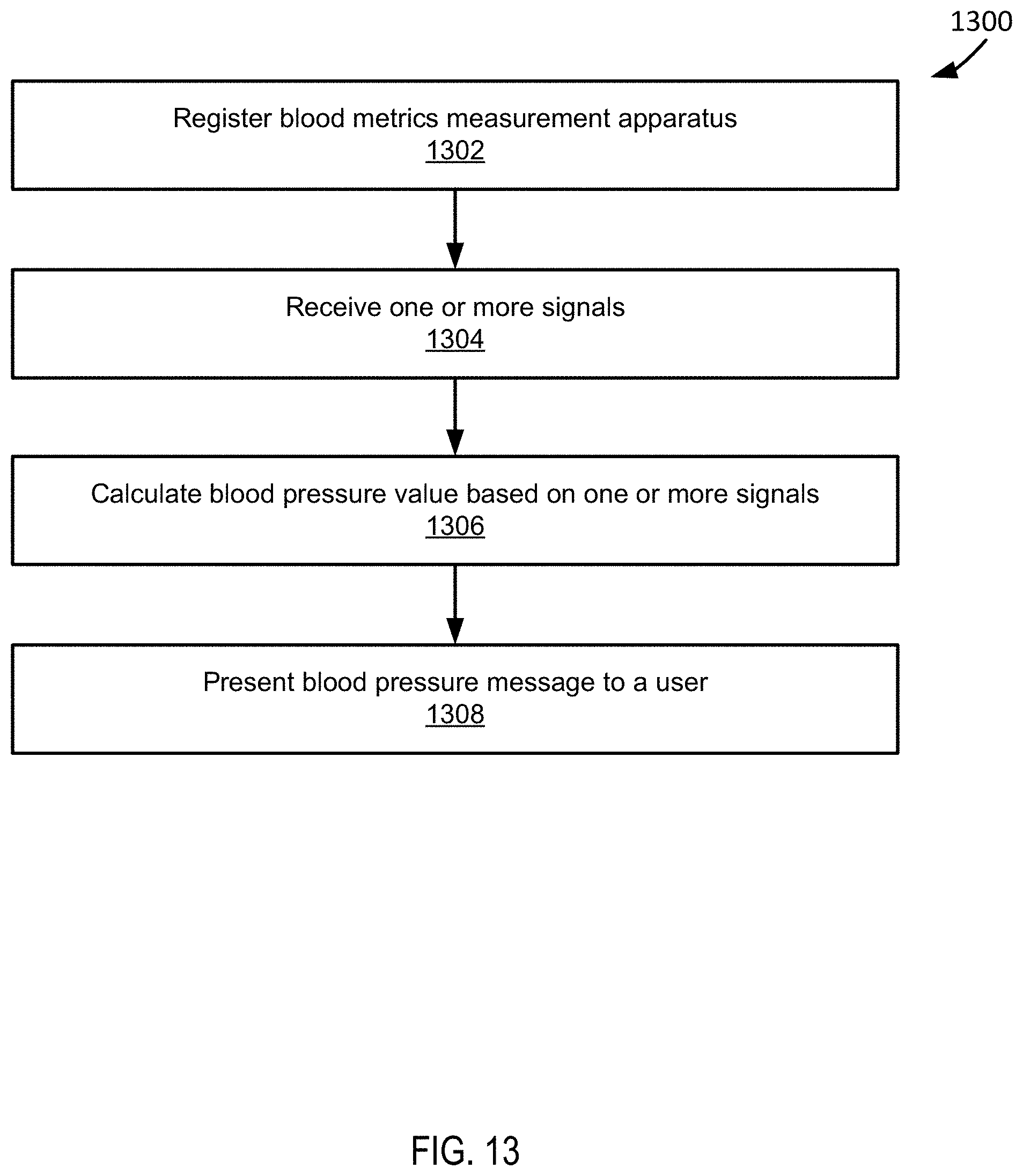
D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025

D00026

D00027
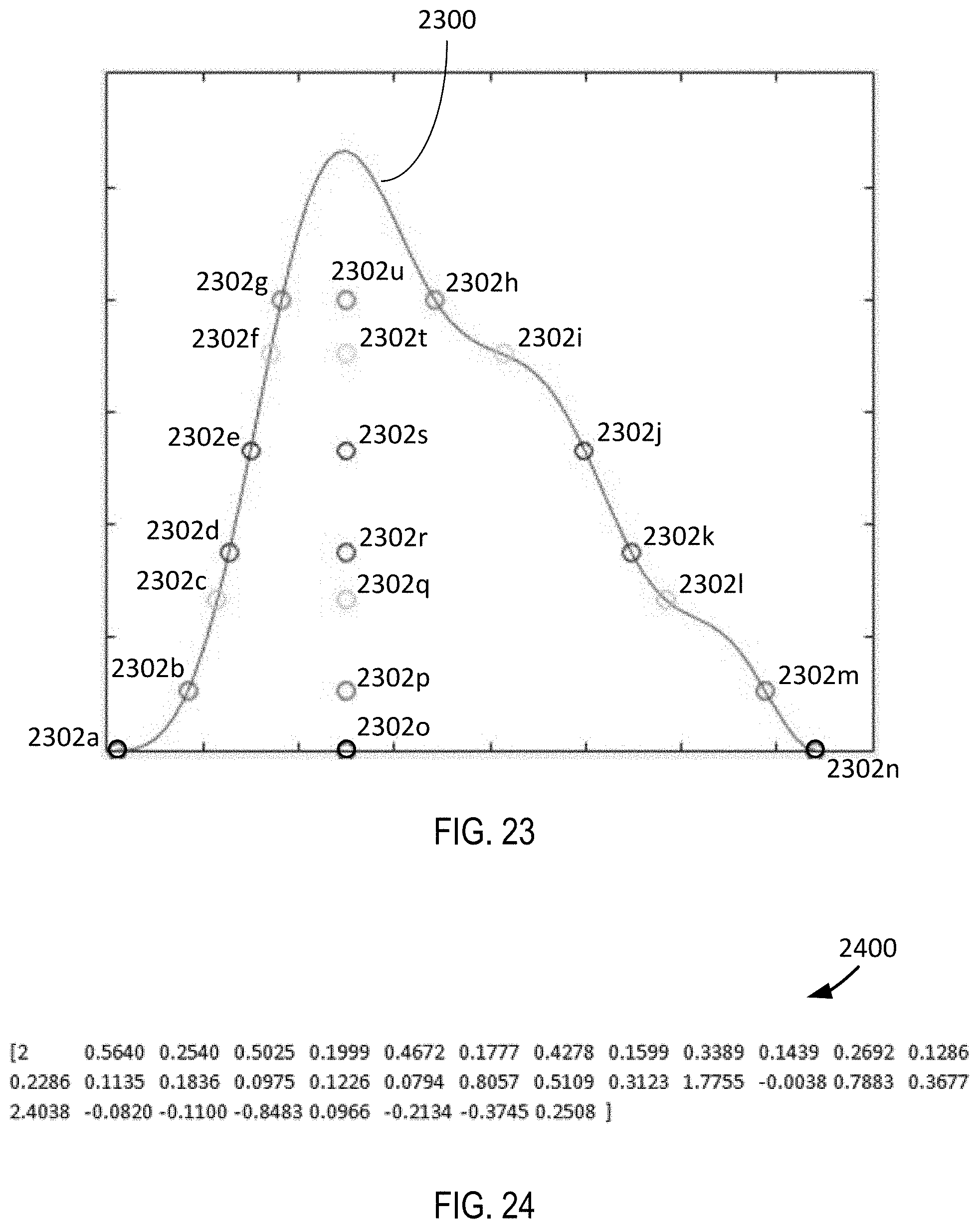
D00028

D00029

D00030

D00031

D00032

D00033

D00034

D00035

D00036

D00037

D00038

D00039



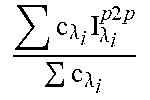












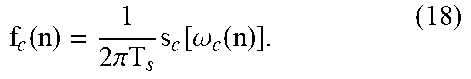


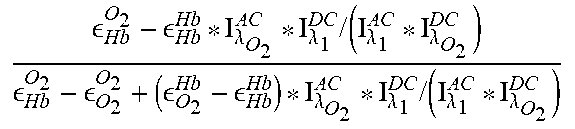



XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.