Cleaning Of Foundry Molds
Urban; Tobias ; et al.
U.S. patent application number 17/042240 was filed with the patent office on 2021-04-22 for cleaning of foundry molds. The applicant listed for this patent is BASF SE. Invention is credited to Michael Koch, Thomas Paasche, Tobias Urban, Henning Urch.
| Application Number | 20210114090 17/042240 |
| Document ID | / |
| Family ID | 1000005326199 |
| Filed Date | 2021-04-22 |

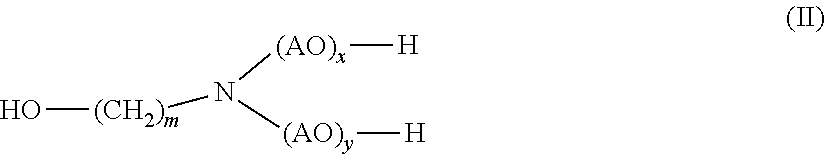




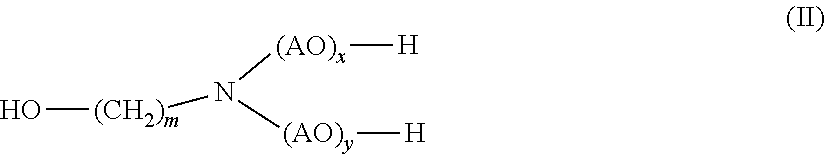
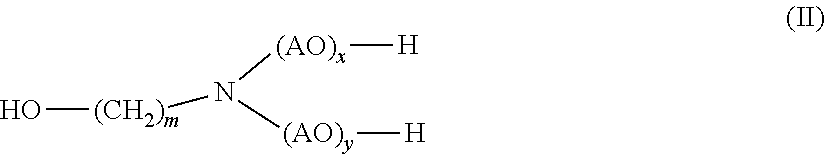



View All Diagrams
| United States Patent Application | 20210114090 |
| Kind Code | A1 |
| Urban; Tobias ; et al. | April 22, 2021 |
CLEANING OF FOUNDRY MOLDS
Abstract
The present invention relates to a method of disintegrating a foundry mold comprising sand and a polyurethane binder. The present invention further relates to a method of disintegrating a foundry mold by contacting it with a composition comprising at least one carboxylic acid amide and at least one tyalkanolamine.
| Inventors: | Urban; Tobias; (Ludwigshafen, DE) ; Urch; Henning; (Limburgerhof, DE) ; Koch; Michael; (Ludwigshafen, DE) ; Paasche; Thomas; (Ludwigshafen, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005326199 | ||||||||||
| Appl. No.: | 17/042240 | ||||||||||
| Filed: | March 21, 2019 | ||||||||||
| PCT Filed: | March 21, 2019 | ||||||||||
| PCT NO: | PCT/EP2019/057042 | ||||||||||
| 371 Date: | September 28, 2020 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | B22C 1/2273 20130101; B22D 29/002 20130101; B22D 31/002 20130101; B22C 5/08 20130101 |
| International Class: | B22D 29/00 20060101 B22D029/00; B22C 1/22 20060101 B22C001/22; B22D 31/00 20060101 B22D031/00; B22C 5/08 20060101 B22C005/08 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Mar 30, 2018 | EP | 18165295.9 |
Claims
1. A method for disintegrating a foundry mold comprising sand and a polyurethane based binder comprising (i) contacting the foundry mold with a composition comprising (a) at least one carboxylic acid amide of general formula (I) ##STR00028## wherein R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl; and (b) at least one alkanolamine of general formula (II) ##STR00029## wherein AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O, m is 1, 2, 3, 4, 5, 6 or 7, and x, y independently of one another are 0 or 1; to obtain a mixture of the sand and the polyurethane binder.
2. The method according to claim 1, wherein the sand is selected from the group consisting of natural silica sand, clay, green sand, quartz sand, chromite sand, olivine sand, special sands, and zirconium sand.
3. The method according to claim 1, wherein the polyurethane based binder comprises polyurethane, water glass, and at least one resin that is different from polyurethane.
4. The method according to claim 1, wherein R.sub.1 is selected from the group comprising of ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, isooctyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isononyl, isodecyl, 1-hydroxyethyl, 1-hydroxypropyl, 2-hydroxypropyl, 1-hydroxybutyl, 2-hydroxybutyl, 1-hydroxypentyl, 2-hydroxypentyl, 3-hydroxypentyl, 4-hydroxypentyl, 1-hydroxyhexyl, 2-hydroxyhexyl, 3-hydroxyhexyl, 4-hydroxyhexyl, and 5-hydroxyhexyl.
5. The method according to claim 1, wherein R.sub.2 and R.sub.3 are methyl.
6. The method according to claim 1, wherein m is 1, 2 or 3.
7. The method according to claim 1, wherein x and y are 0.
8. The method according to claim 1, wherein the at least one carboxylic acid amide of general formula (I) and the at least one alkanolamine of general formula (II) are present in a molar ratio of 5:1 to 1:5.
9. The method according to claim 1, further comprising (ii) separating the sand from the mixture obtained in step (i).
10. The method according to claim 1, further comprising the step of (iii) drying the sand obtained in step (ii).
11. The method according to claim 1, wherein the composition comprises at least one surfactant.
12. The method according to claim 11, wherein the at least one surfactant is a non-ionic surfactant.
13. The method according to claim 12, wherein the non-ionic surfactant is selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil, and alkoxylated alkylamine.
14. A composition comprising (a) 2-hydroxypropanamide; and (b) monoethanolamine.
15. The composition according to claim 14 for use in disintegrating a foundry mold.
16. A method for disintegrating a foundry mold comprising sand and a polyurethane based binder comprising contacting the foundry mold with a composition of claim 14 to obtain a mixture of the sand and the polyurethane binder.
Description
FIELD OF THE INVENTION
[0001] The present invention relates to a method of disintegrating a foundry mold comprising sand and a polyurethane binder. The present invention further relates to a method of disintegrating a foundry mold by contacting it with a composition comprising at least one carboxylic acid amide and at least one alkanolamine.
BACKGROUND OF THE INVENTION
[0002] All foundries produce castings by pouring molten metal into molds. The most common type of casting process is known as sand casting. Sand is used in two different ways in metal casting: as a molding material, which forms the external shape of the cast part, and as cores, which form internal void spaces in products such as engine blocks. Since sand grains do not naturally adhere to each other, binders must be introduced to cause the sand to stick together and hold its shape during the introduction of the molten metal into the mold and the cooling of the casting.
[0003] Sand casting involves manufacturing a pattern of the component to be cast, and packing sand around the pattern to produce a hollow mold. Molds are typically made in two halves to facilitate removal of the pattern, and then the molds are assembled to form a "hollow" that matches the pattern's shape. Cores made of packed sand with special binders may be inserted into a mold, prior to assembly, to form interior surfaces for complex shapes. Molten metal is poured into the mold cavity and allowed to solidify and cool. The casting is shaken out of the sand mold using vibratory machines, mechanically cleaned of extraneous metal by cutting or grinding, and blast cleaned to remove casting sand and other surface contaminants. The foundry industry generates a number of by-products, of which the largest volume is a "spent sand" that consists of sand with residuals of binders. In addition, sand casting generates residuals from metal melting and pouring, and molding processes. It is standard foundry practice to reuse molding and core making sands. Residual sand is routinely screened and returned to the system for reuse.
[0004] Various methods are employed for cleaning metal cast in sand molds which entail both mechanical and chemical actions. In the former category are included abrasive cleaning methods such as sand blasting. Chemical methods of cleaning which are available to the industry, however, produce cleaner castings than the foregoing mechanical procedures. This is effected because a great deal of trouble is occasioned by the retention of the granular sand on the casting by reason of the adhesive properties of the decomposition products of the organic binders. In such instances the adhering sand particles are immune to removal by abrasive cleaning methods and they can only be removed by attacking and dissolving the residual siliceous particles with a chemical reagent. Further, in case of the sand molds being held by the binders, sand molds can be disintegrated by treating them with the compositions or chemicals which dissolve the synthetic binders, and the sand can then be processed and reused.
[0005] U.S. Pat. No. 2,666,001 discloses the removal of residual sand from the cast by immersing in a liquid bath comprising a strongly acidic solution of fluorophosphoric acid followed by neutralization. Fluorophosphoric acid being highly toxic and corrosive, it is difficult to implement the process on a large scale.
[0006] U.S. Pat. No. 2,766,496 discloses a process of cleaning used foundry sand which comprises the steps of screening the sand in presence of water to remove oversized adventitious material, fluidizing the sand with a jet of water and passing it to relatively quiescent settling zones wherein loose carbonaceous impurities are floated off. The disadvantage of the process is that huge volumes of water waste are generated.
[0007] U.S. Pat. No. 4,411,709, discloses removal of resin bonded sand molds and sand cores and the reconditioning of the sand for re-use, by heating the resin bonded molding sand and core sand at a sufficient temperature to be able to pyrolyze the resin binders in the sand. However, the process leads to generation of carbonaceous residues which are difficult to remove.
[0008] Therefore, it is an object of the present invention, while avoiding the abovementioned and further disadvantages, to provide a method which is acceptable in both technical and economic respects for reconditioning used (foundry) sand. It is also an object of the present invention to provide a method for disintegrating the sand present in the form of a sand mold and/or sand core from a metal casting in order to reclaim the sand for further use.
SUMMARY OF THE INVENTION
[0009] Surprisingly, it has been found that the objectives could be achieved by treating the foundry mold with a composition comprising a carboxylic acid amide and an alkanolamine. The sand which is obtained by this method can be reused with minimum steps of further processing.
[0010] Thus, in one aspect, the presently claimed invention is directed to a method for disintegrating a foundry mold comprising sand and a polyurethane binder comprising the step of
[0011] (i) contacting the foundry mold with a composition comprising
[0012] (a) at least one carboxylic acid amide of general formula (I)
##STR00001##
[0013] wherein
[0014] R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl
[0015] R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1--C.sub.12 alkyl; and
[0016] (b) at least one alkanolamine of general formula (II)
##STR00002##
[0017] wherein
[0018] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--,
[0019] m is 1, 2, 3, 4, 5, 6 or 7, and
[0020] x, y independently of one another are 0 or 1
[0021] to obtain a mixture of the sand and the polyurethane binder.
[0022] In another aspect, the presently claimed invention is directed to a composition comprising
[0023] (a) 2-hydroxypropanamide; and
[0024] (b) monoethanolamine
[0025] In another aspect, the presently claimed invention is directed to a composition comprising
[0026] (a) at least one carboxylic acid amide of general formula (I)
##STR00003##
[0027] wherein
[0028] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl
[0029] R.sub.2 and R.sub.3 denote methyl; and
[0030] (b) monoethanolamine.
[0031] In yet another aspect, the presently claimed invention is directed to the use of the composition comprising
[0032] (a) 2-hydroxypropanamide; and
[0033] (b) monoethanolamine
[0034] for disintegrating the foundry mold.
[0035] In yet another aspect, the presently claimed invention is directed to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0036] (i) contacting the foundry mold with a composition comprising [0037] (a) 2-hydroxypropanamide; and [0038] (b) monoethanolamine;
[0039] to obtain a mixture of the sand and the polyurethane binder.
DETAILED DESCRIPTION OF THE INVENTION
[0040] Before the present compositions and formulations of the invention are described, it is to be understood that this invention is not limited to particular compositions and formulations described, since such compositions and formulation may, of course, vary. It is also to be understood that the terminology used herein is not intended to be limiting, since the scope of the present invention will be limited only by the appended claims.
[0041] If hereinafter a group is defined to comprise at least a certain number of embodiments, this is meant to also encompass a group which preferably consists of these embodiments only. Furthermore, the terms "first", "second", "third" or "(a)", "(b)", "(c)", "(d)" etc. and the like in the description and in the claims, are used for distinguishing between similar elements and not necessarily for describing a sequential or chronological order. It is to be understood that the terms so used are interchangeable under appropriate circumstances and that the embodiments of the invention described herein are capable of operation in other sequences than described or illustrated herein. In case the terms "first", "second", "third" or "(A)", "(B)" and " (C)" or "(a)", "(b)", "(c)", "(d)", "i", "ii" etc. relate to steps of a method or use or assay there is no time or time interval coherence between the steps, that is, the steps may be carried out simultaneously or there may be time intervals of seconds, minutes, hours, days, weeks, months or even years between such steps, unless otherwise indicated in the application as set forth herein above or below.
[0042] In the following passages, different aspects of the invention are defined in more detail. Each aspect so defined may be combined with any other aspect or aspects unless clearly indicated to the contrary. In particular, any feature indicated as being preferred or advantageous may be combined with any other feature or features indicated as being preferred or advantageous.
[0043] Reference throughout this specification to "one embodiment" or "an embodiment" means that a particular feature, structure or characteristic described in connection with the embodiment is included in at least one embodiment of the present invention. Thus, appearances of the phrases "in one embodiment" or "in an embodiment" or "in another embodiment" in various places throughout this specification are not necessarily all referring to the same embodiment, but may. Furthermore, the particular features, structures or characteristics may be combined in any suitable manner, as would be apparent to a person skilled in the art from this disclosure, in one or more embodiments. Furthermore, while some embodiments described herein include some but not other features included in other embodiments, combinations of features of different embodiments are meant to be within the scope of the invention, and form different embodiments, as would be understood by those in the art.
[0044] In one aspect, the presently claimed invention relates to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0045] (i) contacting the foundry mold with a composition comprising
[0046] (a) at least one carboxylic acid amide of general formula (I)
##STR00004##
[0047] wherein
[0048] R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl
[0049] R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl; and
[0050] (b) at least one alkanolamine of general formula (II)
##STR00005## [0051] wherein [0052] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2-CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O [0053] m is 1, 2, 3, 4, 5, 6 or 7, and [0054] x, y independently of one another are 0 or 1
[0055] to obtain a mixture of the sand and the polyurethane binder.
[0056] For the purposes of the presently claimed invention, the term `disintegrating` includes cleaning of foundry molds of residual sand and binder which are left on it after the molding process. Additionally, it also covers the `complete breaking of the foundry mold` wherein the sand and binder is separated and the sand can be recycled. Accordingly, a mixture of sand and binder refers to a mixture in which the sand and the binder are present in form of discrete materials.
[0057] `Foundry mold` herein refers to molds made of sand and binder.
[0058] `Contacting` denotes immersing the foundry mold in the composition. It may also include adding the composition on to a metal cast. When the foundry mold is immersed in the composition, it disintegrates into sand and binder. In case when the composition is added on to a metal cast, it is done to clean the metal cast of the residual sand and binder mixture, after the process of casting.
[0059] For the purposes of the presently claimed invention, the term "C.sub.1-C.sub.12-alkyl" covers acyclic saturated hydrocarbon residues, which may be linear or branched and unsubstituted or at least mono-substituted with, as in the case of C.sub.1-C.sub.12-alkyl, 1 to 12 (i.e. 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12) carbon atoms. Similarly, the term "C.sub.2-C.sub.22-alkyl" covers acyclic saturated hydrocarbon residues, which may be linear or branched and unsubstituted or at least mono-substituted with, as in the case of C.sub.2-C.sub.32-alkyl, 2 to 22 (i.e. 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 or 22) carbon atoms. If one or more of the substituents denote an alkyl residue which is mono- or polysubstituted, this may preferably be substituted with optionally 1, 2, 3, 4 or 5, particularly preferably with 1, 2 or 3, substituents mutually independently selected from the group consisting of F, Cl, Br, I, --NO.sub.2, --CN, --OH, --SH, --NH.sub.2, --N(C.sub.1-5-alkyl).sub.2, --N(C.sub.15-alkyl-OH).sub.2, --N(C.sub.1-5-alkyl)(phenyl), --N(C.sub.15-alkyl)(CH.sub.2-phenyl), --N(C.sub.1-5-alkyl)(CH.sub.2-CH.sub.2-phenyl), --C(.dbd.O)--H, --C(.dbd.O)--C.sub.1-5-alkyl, --C(.dbd.O)-phenyl, --C(.dbd.S)--C.sub.1-5-alkyl, C(.dbd.S)-phenyl, --C(.dbd.O)--OH, --C(.dbd.O)--O--C.sub.1-5-alkyl, --C(.dbd.O)--O-phenyl, --C(.dbd.O)--NH.sub.2, --C(.dbd.O)--NH--C.sub.1-5-alkyl, --C(.dbd.O)--N(C.sub.1-5-alkyl).sub.2, --S(.dbd.O)--C.sub.1-5-alkyl, --S(.dbd.O)-phenyl, --S(.dbd.O).sub.2-C.sub.1-5-alkyl, --S(.dbd.O).sub.2-phenyl, --S(.dbd.O).sub.2--NH.sub.2 and --SO.sub.3H, wherein the above-stated-C.sub.1-5 alkyl residues may in each case be linear or branched and the above-stated phenyl residues may preferably be substituted with 1, 2, 3, 4 or 5 substituents mutually independently selected from the group consisting of F, CI, Br, I, --CN, --CF.sub.3, --OH, --NH.sub.2, --O--CF.sub.3, --SH, --O--CH.sub.3, --O--C.sub.2H.sub.5, --O--C.sub.3H.sub.7, methyl, ethyl, n-propyl, isopropyl, n-butyl, 2-butyl, isobutyl and tert.-butyl. Particularly preferred substituents may be selected mutually independently from the group consisting of F, Cl, Br, I, --NO.sub.2, --CN, --OH, --SH, --NH.sub.2, --N(CH.sub.3).sub.2, --N(C.sub.2H.sub.5).sub.2and --N(CH.sub.3)(C.sub.2H.sub.5). As used herein, "branched" denotes a chain of atoms with one or more side chains attached to it. Branching occurs by the replacement of a substituent, e.g., a hydrogen atom, with a covalently bonded alkyl radical.
[0060] Carboxylic acid amide of general formula (I)
[0061] The carboxylic acid amide of general formula (I) has the following structure
##STR00006##
[0062] wherein
[0063] R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl
[0064] R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl.
[0065] Preferably, R.sub.1 is selected from the group consisting of ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, n-tridecyl, n-tetradecyl, n-pentadecyl, n-hexadecyl, n-heptadecyl, n-octadecyl, n-nonadecyl, n-eicosyl, n-heneicosyl, n-docosyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isooctyl, isononyl, isodecyl, isoundecyl, isododecyl, isotridecyl, isotetradecyl, isopentadecyl, isohexadecyl, isoheptadecyl, isooctadecyl, isononadecyl, isoeicosyl, isoheneicosyl, isodocosyl, t-butyl, 1-hydroxyethyl, 1-hydroxypropyl, 2-hydroxypropyl, 1-hydroxybutyl, 2-hydroxybutyl, 2-hydroxybutyl, 1-hydroxypentyl, 2-hydroxypentyl, 3-hydroxypentyl, 4-hydroxypentyl, 1-hydroxyhexyl, 2-hydroxyhexyl, 3-hydroxyhexyl, 4-hydroxyhexyl and 5-hydroxyhexyl.
[0066] More preferably, R.sub.1 is selected from the group consisting of ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, isooctyl, isononyl, isodecyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isodecyl, 1-hydroxyethyl, 1-hydroxypropyl, 2-hydroxypropyl, 1-hydroxybutyl, 2-hydroxybutyl, 2-hydroxybutyl, 1-hydroxypentyl, 2-hydroxypentyl, 3-hydroxypentyl, 4-hydroxypentyl, 1-hydroxyhexyl, 2-hydroxyhexyl, 3-hydroxyhexyl, 4-hydroxyhexyl and 5-hydroxyhexyl. Most preferably, R.sub.1 is selected from the group consisting of n-octyl, n-nonyl, n-decyl, isooctyl, isononyl, isodecyl and 1-hydroxyethyl. Particularly preferably, R.sub.1 is 1-hydroxyethyl.
[0067] In an embodiment, R.sub.2 and R.sub.3 independently of each other, are selected from the group consisting of H, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isooctyl, isononyl, isodecyl, isoundecyl, isododecyl and t-butyl.
[0068] In a preferred embodiment, R.sub.2 and R.sub.3 independently of each other, are selected from the group consisting of H, methyl, ethyl, n-propyl, n-butyl, isopropyl, isobutyl and t-butyl.
[0069] Most preferably, R.sub.2 and R.sub.3 independently of each other, are selected from the group consisting of methyl, ethyl and n-propyl.
[0070] In a particularly preferred embodiment, R.sub.2 and R.sub.3 are methyl.
[0071] Accordingly, in an embodiment the compound of general formula (I) is 2-hydroxypropanamide.
[0072] Suitable carboxylic acid amides are known and commercially available under the tradenames Agnique.RTM. AMD 3L, Agnique.RTM. AMD10 and Agnique.RTM. AMD 810 from BASF SE.
[0073] In a preferred embodiment, the presently claimed invention relates to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0074] (i) contacting the foundry mold with a composition comprising [0075] (a) 2-hydroxypropanamide; and [0076] (b) at least one alkanolamine of general formula (II)
##STR00007##
[0077] wherein
[0078] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O,
[0079] m is 1, 2, 3, 4, 5, 6 or 7, and
[0080] x, y independently of one another are 0 or 1;
[0081] to obtain a mixture of the sand and the polyurethane binder.
[0082] Alkanolamine of general formula (II)
[0083] The alkanolamine of general formula (II) has the following structure
##STR00008##
[0084] wherein
[0085] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O,
[0086] m is 1, 2, 3, 4, 5, 6 or 7, and
[0087] x, y independently of one another are 0 or 1.
[0088] Preferably m is 1, 2 or 3, more preferably m is 2.
[0089] Preferably, x and y are both 0.
[0090] Accordingly, in an embodiment the compound of general formula (II) is monoethanolamine.
[0091] In a preferred embodiment, the presently claimed invention relates to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0092] (i) contacting the foundry mold with a composition comprising [0093] (a) at least one carboxylic acid amide of general formula (I)
[0093] ##STR00009## [0094] wherein [0095] R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl [0096] R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl; and [0097] (b) monoethanolamine; to obtain a mixture of the sand and the polyurethane binder.
[0098] Sand
[0099] The sand is selected from the group consisting of natural silica sand, clay, green sand, quartz sand, chromite sand, olivine sand, special sands and zirconium sand.
[0100] Silica sand in form of granular quarts is the main constituent of molding sand having enough refractoriness which can impart strength, stability and permeability to molding and core sand. But along with silica small amounts of iron oxide, alumina, lime stone (CaCO.sub.3), magnesia, soda and potash are present as impurities. The chemical composition of silica sand gives an idea of the impurities like lime, magnesia, alkalis etc. present. The presence of excessive amounts of iron oxide, alkali oxides and lime can lower the fusion point to a considerable extent which is undesirable. The silica sand can be specified according to the sand grain size and the shape (angular, sub-angular and rounded) of the sand.
[0101] Clay sand is the mixture of natural silica sand, clay, additives and water. The clay used to make wet clay sand is bentonite clay. The compressive strength is generally 0.05 to 0.1 MPa. The water content is 3.5 to 5%.
[0102] Green Sand is a mixture of silica sand, chromite sand, or zircon sand, bentonite (clay), inert sludge, anthracite, sometimes with a proportion of olivine, staurolite or graphite.
[0103] Quartz sand contains mainly crystalline silica SiO.sub.2 and additional mineral contents such as feldspars, clay minerals, mica minerals as well as carbonate and carbonaceous components.
[0104] The suitability of quartz grains as molding material is mainly determined by the SiO.sub.2 content. The higher the content, i.e. the fewer additions of low-melting compounds, the more favorable the sand will behave towards high thermal loads.
[0105] Chromite sand is a naturally occurring spinel consisting primarily of the oxides of chrome and iron.
[0106] Olivine sand is composed mainly of silica, magnesium oxide, iron oxide and aluminium oxide. It has a low thermal expansion rate compared to silica sand.
[0107] Special sands include natural mineral sands, sintering and melting products produced in granular form or turned into granular form by breaking, grinding and grading processes, or inorganic mineral sands produced by other physical-chemical methods. Special sands are different from quartz sand, particularly due to their significantly lower thermal expansion behavior within the temperature range of 20 to 600.degree. C., their heat conductivity, refractoriness, and other physical characteristics.
[0108] Preferably the sand is silica sand or special sand.
[0109] Polyurethane Based Binders
[0110] The polyurethane based binders are polyurethane based binders which may also include, in addition to polyurethane, components such as water glass (sodium silicate) and resins like phenol formaldehyde, urea formaldehyde and furan resins.
[0111] Polyurethanes are formed from polyhydroxy compounds and polyisocyanates. The polyhydroxy compounds in turn are commonly prepared from phenols and formaldehyde. The polyhydroxy component may be any organic hydroxy compound having a functionality of two or more that is soluble in the solvents employed. Such polyhydroxy compounds can include simple aliphatic polyols, amine polyols, polyether polyols, phenolic resins and mixtures of these.
[0112] The amine polyols are normally produced as the reaction product of an alkylene oxide and an amine compound. In general, any polyol containing at least one or more tertiary amine groups is considered to be within the scope of the definition of "amine polyol". The amine compounds which react with alkylene oxides to yield the amine polyols include ammonia and mono- and polyamine compounds with primary or secondary amino nitrogen. Specific examples include aliphatic amines such as primary alkyl amines, ethylene diamine, diethylene triamine and triethylene tetraamine, cycloaliphatic amines, aromatic amines, such as ortho-, meta-, and para-phenylene diamines and aniline formaldehyde resins.
[0113] The phenols employed in the formation of such phenolic resins are generally any of the phenols which may be utilized in the formation of phenolic resins. Specific suitable phenols which may be used include phenol, o-cresol, m-cresol, p-cresol, 2, 5-xylenol, 3, 4-xylenol, 3, 5-xylenol, 3, 4, 5-trimethylphenol, 2-ethylphenol, 3-ethylphenol, 3, 5-diethylphenol, o-sec-butylphenol, p-butylphenol, 3, 5-dibutylphenol, p-amylphenol, p-cyclohexylphenol, o-octylphenol, o-sec-decylphenol, nonylphenol, 3, 5-dicyclohexylphenol, p-phenylphenol, p-crotylphenol, 2-methoxyphenol, 3, 5-dimethoxyphenol, 3, 4, 5-trimethoxyphenol, p-ethoxyphenol, 3-methyl-4-methoxyphenol and p-phenoxyphenol.
[0114] The furan resins that are used for foundry binders are usually prepared from reacting furfuryl alcohol with urea-formaldehyde resins, or with formaldehyde, in order to obtain low-viscosity, soluble, fusible, furan resins. In some cases, phenolic resins or other modifiers are added to the furan resins.
[0115] The aldehyde employed in the formation of the phenolic and furan resin component include any of the aldehydes heretofore employed in the formation of phenolic resins such as formaldehyde, acetaldehyde, propionaldehyde, furfuraldehyde and benzaldehyde.
[0116] The isocyanate component which can be employed in a binder, may have a functionality of 2 or more. Exemplary isocyanates are organic polyisocyanates such as tolylene-2, 4-diisocyanate, tolylene-2, 6-diisocyanate and mixtures thereof, and particularly the crude mixtures thereof that are commercially available. Other typical polyisocyanates include methylene-bis-(4-phenylisocyanate), n-hexyl diisocyanate, naphthalene-1, 5-diisocyanate, cyclopentylene-1, 3-diisocyanate, p-phenylene diisocyanate, tolylene-2, 4, 6-triisocyanate and triphenylmethane-4, 4', 4''-triisocyanate. Higher isocyanates are provided by the liquid reaction products of (1) diisocyanates and (2) polyols or polyamines and the like. In addition, isothiocyanates and mixtures of isocyanates can be employed. Also contemplated are the many impure or crude polyisocyanates that are commercially available. The isocyanate component may additionally contain a chlorosilane compound selected from the group consisting of trimethylchlorosilane, dimethyldichlorosilane, methyltrichlorosilane, tetrachlorosilane, diethychlorosilane, vinyltrichlorosilane and diphenyldichlorosilane.
[0117] Other commonly employed additives can be optionally used in the binder. The use of such materials may enhance the adhesion of the binder to the aggregate material. Examples of additives include, but are not limited to, amino silanes, epoxy silanes, mercapto silanes, hydroxy silanes and ureido silanes such as, for example, gamma-aminopropyltrimethoxysilane, gamma-hydroxypropyltrimethoxysilane, 3-ureidopropyl triethoxysilane, gamma-mercaptopropyltrimethoxysilane, gamma-glycidoxypropyltrimethoxysilane, beta-(3, 4-epoxycyclohexyl)trimethoxysilane and N-beta-(aminoethyl gamma-aminopropyltrimethoxy silane.
[0118] Surfactants
[0119] In an embodiment, the composition that is used according to the presently claimed invention further comprises at least one surfactant. The at least one surfactant is selected from the group consisting of anionic surfactant, cationic surfactant, non-ionic surfactant and amphoteric surfactant.
[0120] Suitable anionic surfactants are selected from the group consisting of salt of alkyl sulfate, alkyl ether sulfate, a-olefin sulfonate and linear alkyl benzene sulfonate.
[0121] The alkyl sulfates are compounds of the formula:
ROSO.sub.3.sup.-M.sup.+
wherein
[0122] R denotes linear or branched, unsubstituted C.sub.6-C.sub.22 alkyl,
[0123] M denotes alkali metal or ammonium cation
[0124] For the purposes of the presently claimed invention, the term "C.sub.6-C.sub.22-alkyl" covers acyclic saturated hydrocarbon residues, which may be linear or branched and unsubstituted having 6 to 22 carbon atoms.
[0125] The alkyl sulfates are obtained by sulfating the higher alcohols (C.sub.6-C.sub.22 carbon atoms) produced from the glycerides of tallow, coconut oil, suitable vegetable oil or synthetic alcohols followed by neutralization with alkali hydroxide. Thus, the alkyl sulfates also contain reaction by-products such as free salt (for example sodium chloride is the free salt by product, when neutralization agent is sodium hydroxide), free fatty alcohol, salt of fatty alcohol. Therefore, the solid content of the alkyl sulfate will be different from the active content. Active content denotes the amount of alkyl sulfate' present in the composition whereas the solid content denotes `a total of alkyl sulfate, fatty alcohol, salt of fatty alcohol and the free salt` in the composition. `Free` herein denotes that the salt is not bound to the fatty alcohol/alkyl sulfate by any kind of chemical bonding.
[0126] Alkyl ether sulfates are compounds of the formula
R'--O--(C.sub.2H.sub.4O).sub.n--SO.sub.3M
[0127] wherein
[0128] R' denotes linear or branched, unsubstituted C.sub.6C.sub.22 alkyl,
[0129] n is from 1 to 20
[0130] M denotes alkali metal or ammonium cation
[0131] The alkyl ether sulfates are produced by the ethoxylation of fatty alcohol and thus will generally be obtained in the form of mixtures comprising varying alkyl chain lengths and varying degrees of ethoxylation. Frequently such mixtures will inevitably also contain some non-ethoxylated alkyl sulfates.
[0132] .alpha.-olefin sulfonates are generally produced by sulfonating .alpha.-olefin. The .alpha.-olefins, which are sulfonated to form the surfactants used in the compositions of the present invention, may contain from about 10 to 22 carbon atoms and preferably 12 to 18 carbon atoms. They may be derived from a variety of processes such as, for example, by wax cracking, ethylene build up or dehydration of the corresponding primary alcohol. Exemplary alpha-olefins are 1-decene, 1-undecene, 1-dodccene, 1-tridecene, 1-tetradecene, 1-pentadecene, 1-hexadecene, 1-heptadecene, 1-octadecene and the like and mixtures of the aforesaid. Sulfonation of these long chain olefins is typically carried out utilizing sulfur trioxide mixed with a diluent. After the sulfonation is completed, neutralization and hydrolysis of the acid mixture is carried out so that any by-product sultones which are formed are converted to the corresponding hydroxyalkane sulfonates. Thus, as is well known in the art, the term .alpha.-olefin sulfonates as used herein includes not only the alkene sulfonate itself but also admixtures of same formed as a result of the usual sulfonation neutralization, and hydrolysis procedure with substantial proportions of the corresponding water soluble hydroxyalkane sulfonates.
[0133] Linear alkyl benzene sulfonate (LABS) is produced by sulfonation of linear alkylbenzene (LAB) and subsequent neutralization of the corresponding sulfonic acid (HLAS). Linear alkylbenzene is synthesized by the alkylation of benzene with linear olefins. Traditional processes for alkylation of aromatics compounds use Friedel-Craft type catalysts, for example, hydrofluoric acid, aluminum trichloride and the like.
[0134] Anionic surfactant may also include alkylamide sulphates of formula
R.sub.4CONHR.sub.5OSO.sub.3M
wherein
[0135] R.sub.4 denotes a C.sub.2-C.sub.22 alkyl
[0136] R.sub.5 C.sub.2-C.sub.3 alkyl radical,
[0137] M is a hydrogen atom or an alkali metal cation cation or ethoxylenated (EO) and/or propoxylenated (PO) derivatives thereof, containing on average from 0.5 to 60 EO and/or PO units;
[0138] Further anionic surfactants are, salts of C.sub.8-C.sub.24, saturated or unsaturated fatty acids, alkylglyceryl sulphonates, paraffin sulphonates, N-acyl N-alkyltaurates, alkylphosphates, isethionates, alkylsuccinamates, alkylsulphosuccinates, sulphosuccinate monoesters or diesters, N-acyl sarcosinates, alkylglycoside sulphates, polyethoxycarboxylates, the cation being an alkali metal (sodium, potassium or lithium), a substituted or unsubstituted ammonium residue (methyl-, dimethyl-, trimethyl- or tetramethylammonium, dimethylpiperidinium, etc.) or an alkanolamine derivative (monoethanolamine, diethanolamine, triethanolamine, etc.) and alkyl or alkylaryl phosphate esters.
[0139] Cationic surfactants are a well-known group of surface-active compounds which have at least one active cationic (positive ion) constituent. As the cationic surfactant, quaternary ammonium hydroxides such as octyltrimethylammonium hydroxide, dodecyltrimethylammonium hydroxide, hexadecyltrimethylammonium hydroxide, octyldimethylbenzylammonium hydroxide decyldimethylbenzylammonium hydroxide, didodecyldimethylammonium hydroxide, dioctadecyldimethylammonium hydroxide, beef tallow trimethylammonium hydroxide, and coconut oil trimethylammonium hydroxide, and their salts can be exemplified.
[0140] Examples of amphoteric surfactants comprise betaines, sulphobetaines and carboxylates and sulphonates of fatty acids and of imidazole, such as alkyldimethylbetaines, alkylamidopropyldimethylbetaines, alkyldimethylsulphobetaines or alkylamidopropyldimethylsulphobetaines, such as Mirataine CBS sold by the company Rhodia, and the products of condensation of fatty acids and of protein hydrolysates; alkylamphoacetates or alkylamphodiacetates in which the alkyl group contains from 6 to 20 carbon atoms; amphoteric alkylpolyamine derivatives such as Amphionic XL.RTM. sold by Rhodia and Ampholac 71/X.RTM. and Ampholac 7C/X.RTM. sold by Berol Nobel.
[0141] The at least one surfactant is preferably a non-ionic surfactant.
[0142] The at least one non-ionic surfactant is selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine.
[0143] The poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether is preferably poly(C.sub.2-C.sub.3)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether.
[0144] Preferred poly(C.sub.2-C.sub.3)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether are preferably compounds of the formula
R.sub.6--(O--CH.sub.2-CH.sub.2),(O--CH(CH.sub.3)--CH.sub.2).sub.r--OH
wherein
[0145] R.sub.6 is linear or branched C.sub.8-C.sub.22-alkyl;
[0146] q is a natural number from 1 to 50; and
[0147] r is 0 or is a natural number from 1 to 30, with the proviso that 2.ltoreq.q+r.ltoreq.50.
[0148] In a preferred embodiment, the poly(C.sub.2-C.sub.3)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether are preferably compounds of the formula
R.sub.6--(O--CH.sub.2-CH.sub.2).sub.q(O--CH(CH.sub.3)--CH.sub.2).sub.r--- OH
wherein
[0149] R.sub.6 is linear or branched C.sub.8-C.sub.22-alkyl;
[0150] q is a natural number from 1 to 15; and
[0151] r is 0 or is a natural number from 1 to 20, with the proviso that 2.ltoreq.q+r.ltoreq.30.
[0152] Within the context of the present invention, the term "alkyl", as used herein, refers to acyclic saturated aliphatic residues, including linear or branched alkyl residues. Furthermore, the alkyl residue is unsubstituted and includes as in the case of C.sub.8-C.sub.22 alkyl 8 to 22 carbon atoms.
[0153] Representative examples of linear and branched C.sub.8-C.sub.22 alkyl include, but are not limited to, n-octyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, n-tridecyl, n-tetradecyl, n-pentadecyl, n-hexadecyl, n-heptadecyl, n-octadecyl, n-nonadecyl, n-eicosyl, n-heneicosyl, n-docosyl, 2-ethylhexyl, 2-propyl-heptyl, 2-butyl-1-octyl, 2-pentyl-1-nonyl, isooctyl, isodecyl, isoundecyl, isododecyl, isotridecyl, isotetradecyl, isopentadecyl, isohexadecyl, isoheptadecyl, isooctadecyl, isononadecyl, isoeicosyl, isoheneicosyl, and isodocosyl.
[0154] The poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether have a molecular weight of from 300 g/mol to 2000 g/mol determined by .sup.1H-NMR spectroscopy using for instance a 400 MHz spectrometer by Bruker. The molecular weight can be determined by the integration of the signals of the poly(C.sub.2--C.sub.4)alkylene glycol backbone and the comparison of this integral with the integral of the mono-(C.sub.8--C.sub.22)-alkyl signals.
[0155] The poly(C.sub.2--C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether can be prepared by alkoxylation of fatty alcohol. When the fatty alcohol R.sup.1--OH which is used for the synthesis of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether is derived from a natural source it is common to have mixtures, e.g. of C.sub.16 and C.sub.18 alcohols or C.sub.12 and C.sub.14 alcohols. Fatty alcohol R.sup.1--OH can also be synthesized (for example by oxo process) from olefin mixtures and in this case, it is common to have mixtures e.g. of C.sub.13 and C.sub.15 alcohols.
[0156] Suitable poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether are known and commercially available, e.g. Lutensol.RTM. XL from BASF SE.
[0157] The poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester are preferably compounds of the formula
R.sub.6--CO--(O--CH.sub.2--CH.sub.2).sub.q(O--CH(CH.sub.3)--CH.sub.2).su- b.r--R.sub.7
wherein
[0158] R.sub.6 is linear or branched C.sub.8-C.sub.22-alkyl;
[0159] R.sub.7 is H or linear or branched C.sub.1-C.sub.8 alkyl;
[0160] q is a natural number from 1 to 50; and
[0161] r is 0 or is a natural number from 1 to 30, with the proviso that 2.ltoreq.q+r.ltoreq.50
[0162] Representative examples of linear or branched C.sub.8-C.sub.22 alkyl include, but are not limited to, n-octyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, n-tridecyl, n-tetradecyl, n-pentadecyl, n-hexa-decyl, n-heptadecyl, n-octadecyl, n-nonadecyl, n-eicosyl, n-heneicosyl, n-docosyl, 2-ethylhexyl, 2-propyl-heptyl, 2-butyl-1-octyl, 2-pentyl-1-nonyl, isooctyl, isodecyl, isoundecyl, isododecyl, isotridecyl, isotetradecyl, isopentadecyl, isohexadecyl, isoheptadecyl, isooctadecyl, isononadecyl, isoeicosyl, isoheneicosyl, isodocosyl and isomers thereof.
[0163] Representative examples of linear or branched C.sub.1-C.sub.8 alkyl include, but are not limited to, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isooctyl, t-butyl, 2-ethylhexyl and isomers thereof.
[0164] The poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid esters have a molecular weight of from 300 to 2000 determined by .sup.1H-NMR spectroscopy using for instance a 400 MHz spectrometer by Bruker. The molecular weight can be determined by the integration of the signals of the poly(C.sub.2-C.sub.4)alkylene glycol backbone and the comparison of this integral with the integral of the mono-(C.sub.8-C.sub.22)-alkyl signals.
[0165] The alkoxylated oil is preferably an ethoxylated oil, more preferably ethoxylated derivative of castor oil with a hydrophilic/lipophilic balance of about 14 (HLB 14), such as Emulan.RTM. EL.
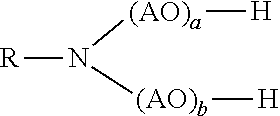
[0166] The alkoxylated alkylamine are preferably compounds of the formula
##STR00010##
wherein
[0167] R is linear or branched, substituted or unsubstituted C.sub.8-C.sub.22-alkyl or linear or branched, substituted or unsubstituted C.sub.8-C.sub.22-alkenyl;
[0168] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O,
[0169] a is a natural number from 1 to 20; and
[0170] b is a natural number from 1 to 20, with the proviso that 2.ltoreq.a+b.ltoreq.40.
[0171] For the purpose of the presently claimed invention, the term "alkenyl" covers acyclic unsaturated hydrocarbon residues, which may be linear or branched and unsubstituted or at least mono-substituted and comprise at least one double bond, preferably 1, 2 or 3 double bonds, with, as in the case of C.sub.8-C.sub.22 alkenyl, 8 to 22 (i.e. 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22) carbon atoms. If one or more of the substituents denote an alkenyl residue which is mono- or polysubstituted, this may preferably be substituted with optionally 1, 2, 3, 4 or 5, particularly preferably with 1, 2 or 3, substituents mutually independently selected from the group consisting of F, Cl, Br, I, --NO.sub.2, --CN, --OH, --SH, --NH.sub.2, --N(C.sub.1-5-alkyl).sub.2, --N(C.sub.15-alkyl-OH).sub.2, --N(C.sub.1-5-alkyl)(phenyl), --N(C.sub.15-alkyl)(CH.sub.2-phenyl), --N(C.sub.1-5-alkyl)(CH.sub.2-CH.sub.2-phenyl), --C(.dbd.O)--H, --C(.dbd.O)--C.sub.1-5-alkyl, --C(.dbd.O)-phenyl, --C(.dbd.S)--C.sub.1-5-alkyl, C(.dbd.S)-phenyl, --C(.dbd.O)--OH, --C(.dbd.O)--O--C.sub.1-5-alkyl, --C(.dbd.O)--O-phenyl, --C(.dbd.O)--NH.sub.2, --C(.dbd.O)--NH--C.sub.1-5-alkyl, --C(.dbd.O)--N(C.sub.1-5-alkyl).sub.2, --S(.dbd.O)--C.sub.1-5-alkyl, --S(.dbd.O)-phenyl, --S(.dbd.O).sub.2-C.sub.1-5-alkyl, --S(.dbd.O).sub.2-phenyl, --S(.dbd.O).sub.2--NH.sub.2 and --SO.sub.3H, wherein the above-stated-C.sub.1-5 alkyl residues may in each case be linear or branched and the above-stated phenyl residues may preferably be substituted with 1, 2, 3, 4 or 5 substituents mutually independently selected from the group consisting of F, Cl, Br, I, --CN, --CF.sub.3, --OH, --NH.sub.2, --O--CH.sub.3--SH, --O--CH.sub.3, --O--C.sub.2H.sub.5, --O--C.sub.3H.sub.7, methyl, ethyl, n-propyl, isopropyl, n-butyl, 2-butyl, isobutyl and tert-butyl. Particularly preferred substituents may be selected mutually independently from the group consisting of F, Cl, Br, I, --NO.sub.2, --CN, --OH, --SH, --NH.sub.2, --N(CH.sub.3).sub.2, --N(C.sub.2H.sub.5).sub.2and --N(CH.sub.3)(C.sub.2H.sub.5).
[0172] Preferably, R is linear, unsubstituted C.sub.8-C.sub.22-alkenyl; AO is CH.sub.2--CH.sub.2--O, a is a natural number from 1 to 10; b is a natural number from 1 to 10, with the proviso that 5.ltoreq.a+b.ltoreq.20.
[0173] More preferably, alkoxylated alkylamine is an ethoxylated alkylamine and most preferably oleyl amine ethoxylate. Suitable alkoxylated alkylamine are known and commercially available, e.g. Lutensol.RTM. FA12 from BASF SE.
[0174] Particularly preferably, the non-ionic surfactant is selected from alkyl polyethylene glycol ether, ethoxylated castor oil and oleyl amine ethoxylate.
[0175] In a preferred embodiment the presently claimed invention relates to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0176] (i) contacting the foundry mold with a composition comprising [0177] (a) at least one carboxylic acid amide of general formula (I)
[0177] ##STR00011## [0178] wherein [0179] R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2--C.sub.22 alkyl [0180] R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl; [0181] (b) at least one alkanolamine of general formula (II)
[0181] ##STR00012## [0182] wherein [0183] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O, [0184] m is 1, 2, 3, 4, 5, 6 or 7, and [0185] x, y independently of one another are 0 or 1; and [0186] (c) at least one non-ionic surfactant;
[0187] to obtain a mixture of the sand and the polyurethane binder.
[0188] In another preferred embodiment the presently claimed invention relates to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0189] (i) contacting the foundry mold with a composition comprising
[0190] (a) at least one carboxylic acid amide of general formula (I)
##STR00013##
[0191] wherein
[0192] R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl
[0193] R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl;
[0194] (b) at least one alkanolamine of general formula (II)
##STR00014##
wherein [0195] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O, [0196] m is 1, 2, 3, 4, 5, 6 or 7, and [0197] x, y independently of one another are 0 or 1; and
[0198] (c) at least one non-ionic surfactant selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine;
[0199] to obtain a mixture of the sand and the polyurethane binder.
[0200] Water
[0201] In a preferred embodiment, the composition that is used according to the present invention does not contain water. In another preferred embodiment, water is added to the composition. The amount of water, if added to the composition, is in the range of 0.1 wt. % to 12.0 wt. %, based on the total weight of the composition. The disintegration of the foundry mold takes a longer time, in the range of 100 minutes to 180 minutes when the composition comprises water, compared to the composition without water.
[0202] Additives
[0203] The foundry mold may further comprise at least one additive. Additives are the materials generally added to the sand and binder mixture to develop some special property in the sand.
[0204] Some commonly used additives for enhancing the properties of molding and core sands are coal dust, corn flour, dextrin, sea coal, pitch, wood flour, silica flour.
[0205] Coal Dust
[0206] Coal dust is added mainly for producing a reducing atmosphere during casting process. This reducing atmosphere results in any oxygen in the poles becoming chemically bound so that it cannot oxidize the metal. It is usually added in the molding sands for making molds for production of grey iron and malleable cast iron castings.
[0207] Corn Flour
[0208] Corn flour belongs to the starch family of carbohydrates and is used to increase the collapsibility of the molding and core sand. It is completely volatilized by heat in the sand mould, thereby leaving space between the sand grains. This allows free movement of sand grains, which finally gives rise to mould wall movement and decreases the mold expansion and hence defects in castings. Corn sand if added to molding sand and core sand improves significantly strength of the mold and core.
[0209] Dextrin
[0210] Dextrin also belongs to starch family of carbohydrates that behaves also in a manner similar to that of the corn flour. Dextrin increases dry strength of the molds.
[0211] Sea Coal
[0212] Sea coal is the fine powdered bituminous coal which positions its place among the pores of the silica sand grains in molding sand and core sand. When heated, sea coal changes to coke which fills the pores and is unaffected by water. Because to this, the sand grains become restricted and cannot move into a dense packing pattern. Thus, sea coal reduces the mould wall movement and the permeability in mold and core sand and hence makes the mold and core surface clean and smooth.
[0213] Pitch
[0214] Pitch is distilled form of soft coal. It can be added from 0.02% to 2% in mold and core sand. Pitch enhances hot strengths, surface finish on mold surfaces and behaves exactly in a manner similar to that of sea coal.
[0215] Wood Flour
[0216] Wood flour is a fibrous material mixed with a granular material like sand. Wood flour is relatively long thin fibers prevent the sand grains from making contact with one another. wood flour can be added in between 0.05% to 2% in mold and core sand. Wood flour volatilizes when heated, thus allowing the sand grains room to expand. Wood flour will increase mould wall movement and decrease expansion defects. Wood flour also increases collapsibility of both mold and core.
[0217] Pulverized Silica or Silica Flour
[0218] Silica flour is called as pulverized silica. Pulverized silica can be easily added up to 3% which increases the hot strength and finish on the surfaces of the molds and cores. It also reduces metal penetration in the walls of the molds and cores.
[0219] In another aspect, the presently claimed invention is directed to a composition comprising
[0220] (a) 2-hydroxypropanamide; and
[0221] (b) monoethanolamine.
[0222] In another embodiment, the presently claimed invention is directed to a composition comprising
[0223] (a) 2-hydroxypropanamide;
[0224] (b) monoethanolamine; and
[0225] (c) at least one non-ionic surfactant;
[0226] In another embodiment, the presently claimed invention is directed to a composition comprising
[0227] (a) 2-hydroxypropanamide;
[0228] (b) monoethanolamine; and
[0229] (c) at least one non-ionic surfactant selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine.
[0230] In yet another aspect, the presently claimed invention is directed to the use of the composition comprising
[0231] (a) 2-hydroxypropanamide; and
[0232] (b) monoethanolamine
[0233] for disintegrating the foundry mold.
[0234] In another embodiment, the presently claimed invention is directed to the use of the composition comprising
[0235] (a) 2-hydroxypropanamide;
[0236] (b) monoethanolamine; and
[0237] (c) at least one non-ionic surfactant;
[0238] for disintegrating the foundry mold.
[0239] In another embodiment, the presently claimed invention is directed to the use of the composition comprising
[0240] (a) 2-hydroxypropanamide;
[0241] (b) monoethanolamine; and
[0242] (c) at least one non-ionic surfactant selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene [0243] glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic [0244] acid ester, alkoxylated oil and alkoxylated alkylamine;
[0245] for disintegrating the foundry mold.
[0246] In yet another aspect, the presently claimed invention is directed to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0247] (i) contacting the foundry mold with a composition comprising [0248] (a) 2-hydroxypropanamide; and [0249] (b) monoethanolamine;
[0250] to obtain a mixture of the sand and the polyurethane binder.
[0251] In an embodiment, the presently claimed invention is directed to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0252] (i) contacting the foundry mold with a composition comprising [0253] (a) 2-hydroxypropanamide; [0254] (b) monoethanolamine; and [0255] (c) at least one non-ionic surfactant;
[0256] to obtain a mixture of the sand and the polyurethane binder.
[0257] In another embodiment, the presently claimed invention is directed to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0258] (i) contacting the foundry mold with a composition comprising [0259] (a) 2-hydroxypropanamide; [0260] (b) monoethanolamine; and [0261] (c) at least one non-ionic surfactant selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine;
[0262] to obtain a mixture of the sand and the polyurethane binder.
[0263] In another aspect, the presently claimed invention is directed to a composition comprising
[0264] (a) at least one carboxylic acid amide of general formula (I)
##STR00015##
[0265] wherein
[0266] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl;
[0267] R.sub.2 and R.sub.3 denote methyl; and
[0268] (b) monoethanolamine.
[0269] In another embodiment, the presently claimed invention is directed to a composition comprising
[0270] (a) at least one carboxylic acid amide of general formula (I)
##STR00016##
[0271] wherein
[0272] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl;
[0273] R.sub.2 and R.sub.3 denote methyl;
[0274] (b) monoethanolamine; and
[0275] (c) at least one non-ionic surfactant.
[0276] In another embodiment, the presently claimed invention is directed to a composition comprising
[0277] (a) at least one carboxylic acid amide of general formula (I)
##STR00017##
[0278] wherein
[0279] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl;
[0280] R.sub.2 and R.sub.3 denote methyl;
[0281] (b) monoethanolamine; and
[0282] (c) at least one non-ionic surfactant selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine.
[0283] In yet another aspect, the presently claimed invention is directed to the use of the composition comprising
[0284] (a) at least one carboxylic acid amide of general formula (I)
##STR00018##
[0285] wherein
[0286] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl;
[0287] R.sub.2 and R.sub.3 denote methyl; and
[0288] (b) monoethanolamine,
[0289] for disintegrating the foundry mold.
[0290] In another embodiment, the presently claimed invention is directed to the use of the composition comprising
[0291] (a) at least one carboxylic acid amide of general formula (I)
##STR00019##
[0292] wherein
[0293] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl;
[0294] R.sub.2 and R.sub.3 denote methyl;
[0295] (b) monoethanolamine; and
[0296] (c) at least one non-ionic surfactant;
[0297] for disintegrating the foundry mold.
[0298] In another embodiment, the presently claimed invention is directed to the use of the composition comprising
[0299] (a) at least one carboxylic acid amide of general formula (I)
##STR00020##
[0300] wherein
[0301] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl;
[0302] R.sub.2 and R.sub.3 denote methyl;
[0303] (b) monoethanolamine; and
[0304] (c) at least one non-ionic surfactant selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine;
[0305] for disintegrating the foundry mold.
[0306] In yet another aspect, the presently claimed invention is directed to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0307] (i) contacting the foundry mold with a composition comprising [0308] (a) at least one carboxylic acid amide of general formula (I)
[0308] ##STR00021## [0309] wherein [0310] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl; [0311] R.sub.2 and R.sub.3 denote methyl; and [0312] (b) monoethanolamine;
[0313] to obtain a mixture of the sand and the polyurethane binder.
[0314] In an embodiment, the presently claimed invention is directed to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0315] (i) contacting the foundry mold with a composition comprising [0316] (a) at least one carboxylic acid amide of general formula (I)
[0316] ##STR00022## [0317] wherein [0318] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl; [0319] R.sub.2 and R.sub.3 denote methyl; [0320] (b) monoethanolamine; and [0321] (c) at least one non-ionic surfactant;
[0322] to obtain a mixture of the sand and the polyurethane binder.
[0323] In another embodiment, the presently claimed invention is directed to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the step of
[0324] (i) contacting the foundry mold with a composition comprising [0325] (a) at least one carboxylic acid amide of general formula (I)
[0325] ##STR00023## [0326] wherein [0327] R.sub.1 denotes linear or branched, unsubstituted C.sub.2-C.sub.12 alkyl; [0328] R.sub.2 and R.sub.3 denote methyl; [0329] (b) monoethanolamine; and [0330] (c) at least one non-ionic surfactant selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine;
[0331] to obtain a mixture of the sand and the polyurethane binder.
[0332] In another preferred embodiment, the presently claimed invention relates to a method for disintegrating the foundry mold comprising sand and a polyurethane binder comprising the steps of
[0333] (i) contacting the foundry mold with a composition comprising
[0334] (a) at least one carboxylic acid amide of general formula (I)
##STR00024##
[0335] wherein
[0336] R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl
[0337] R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl; and
[0338] (b) at least one alkanolamine of general formula (II)
##STR00025##
wherein
[0339] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O,
[0340] is 1, 2, 3, 4, 5, 6 or 7, and
[0341] x, y independently of one another are 0 or 1,
[0342] to obtain a mixture of the sand and the polyurethane binder;
[0343] (ii) separating the sand from the mixture obtained in step (i); and
[0344] (iii) drying the sand obtained in step (ii).
[0345] The sand is separated from the mixture of sand and polyurethane binder by one or more methods known in the art, namely reconditioning, thermal methods, dry methods etc. Reconditioning merely contemplates that the used sand is passed over magnetic pulleys, through lump breakers and screens, and then aerated to remove fines. Thermal methods of treatment, include a roasting furnace, a fluid bed furnace and a rotary kiln. The thermal calcine system subjects the sand to temperatures in the range of 1200.degree.-1500.degree. in the presence of excess oxygen, thereby removing carbonaceous additives. Dry method includes mechanical methods which comprise impacting sand grains to fracture the "shells" of old bonds from individual grains. There are several modes that would fall under the category of being dry, including (a) centrifuging sand against an enclosure; (b) a pneumatically shooting sand against a target, or additionally causing two separate streams of sand to intersect for scrubbing; and (c) mulling at low kinetic energy levels to squeeze the sand grains under pressure of a wheel.
[0346] The sand is dried by processes known to a person skilled in the art. The sand is passed through a fluid bed furnace. Hot sand is then passed through a cooling drum where the sand is subsequently cleaned and cooled to room temperature by means of cooling air. The sand obtained after drying is routinely screened and returned to the system for reuse. The sand can be recycled and reused in the foundry for making foundry molds for a number of cycles.
[0347] However, as the sand is repeatedly used, the particles eventually become too fine for the molding process; and, hence can also be put to other application, if this sand cannot be used for moulding, such as: [0348] 1. Asphalt Concrete: Substitution of up to 15% spent sand for conventional asphalt concrete fine aggregate. [0349] 2. Compost Additive: Bulking agent for composted yard waste, to produce topsoil or topsoil additive. [0350] 3. Concrete: Substitution for regular sand in structural grade concrete, at low percentages. [0351] 4. Bricks and Pavers: Encapsulation in a proprietary, high pressure, pozzolanic process that can encapsulate and chemically bind various waste materials in C-grade fly ash (a fine particulate ash produced by coal-burning electrical power plants). The ambient-temperature process results in bricks that are cost effective and can be shaped to meet end-user requirements
[0352] The present invention offers one or more of following advantages: [0353] 1. The method of the present invention can be used for a variety of foundry molds with varied sand and binder composition. [0354] 2. Small amount of the composition is effective in disintegrating the foundry mold.
[0355] In the following, specific embodiments of the present invention are described:
[0356] 1. A method for disintegrating the foundry mold comprising sand and a polyurethane based binder comprising at least the step of [0357] (i) contacting the foundry mold with a composition comprising [0358] (a) at least one carboxylic acid amide of general formula (I)
[0358] ##STR00026## wherein R.sub.1 denotes linear or branched, unsubstituted or hydroxy substituted C.sub.2-C.sub.22 alkyl R.sub.2 and R.sub.3 independently of each other, denote H or linear or branched, unsubstituted or substituted C.sub.1-C.sub.12 alkyl; and
[0359] (b) at least one alkanolamine of general formula (II)
##STR00027##
wherein
[0360] AO is, identical or different, selected from the group consisting of CH.sub.2--CH.sub.2--O, CH(CH.sub.3)--CH.sub.2--O, CH.sub.2--CH(CH.sub.3)--O, CH(C.sub.2H.sub.5)--CH--O, C(CH.sub.3).sub.2--CH.sub.2--O, CHC(CH.sub.3).sub.2--O and CH.sub.2--CH(C.sub.2H.sub.5)--O,
[0361] m is 1, 2, 3, 4, 5, 6 or 7, and
[0362] x, y independently of one another are 0 or 1;
[0363] to obtain a mixture of the sand and the polyurethane binder.
[0364] 2. The method according to embodiment 1, wherein the sand is selected from the group consisting of natural silica sand, clay, green sand, quartz sand, chromite sand, olivine sand, special sands and zirconium sand.
[0365] 3. The method according to embodiment 1, wherein the polyurethane based binder comprises polyurethane, water glass and at least one resin that is different from polyurethane.
[0366] 4. The method according to embodiment 1, wherein R.sub.1 is selected from the group consisting of ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, n-tridecyl, n-tetradecyl, n-pentadecyl, n-hexadecyl, n-heptadecyl, n-octadecyl, n-nonadecyl, n-eicosyl, n-heneicosyl, n-docosyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isooctyl, isononyl, isodecyl, isoundecyl, isododecyl, isotridecyl, isotetradecyl, isopentadecyl, isohexadecyl, isoheptadecyl, isooctadecyl, isononadecyl, isoeicosyl, isoheneicosyl, isodocosyl, t-butyl, 1-hydroxyethyl, 1-hydroxypropyl, 2-hydroxypropyl, 1-hydroxybutyl, 2-hydroxybutyl, 2-hydroxybutyl, 1-hydroxypentyl, 2-hydroxypentyl, 3-hydroxypentyl, 4-hydroxypentyl, 1-hydroxyhexyl, 2-hydroxyhexyl, 3-hydroxyhexyl, 4-hydroxyhexyl and 5-hydroxyhexyl.
[0367] 5. The method according to embodiment 1, wherein R.sub.1 is selected from the group comprising of ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, isooctyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isononyl, isodecyl, 1-hydroxyethyl, 1-hydroxypropyl, 2-hydroxypropyl, 1-hydroxybutyl, 2-hydroxybutyl, 1-hydroxypentyl, 2-hydroxypentyl, 3-hydroxypentyl, 4-hydroxypentyl, 1-hydroxyhexyl, 2-hydroxyhexyl, 3-hydroxyhexyl, 4-hydroxyhexyl and 5-hydroxyhexyl.
[0368] 6. The method according to embodiment 1, wherein R.sub.2 and R.sub.3 independently of each other, are selected from the group consisting of H, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, isopropyl, isobutyl, isopentyl, isohexyl, isoheptyl, isooctyl, isononyl, isodecyl, isoundecyl, isododecyl and t-butyl.
[0369] 7. The method according to one or more of embodiment 1 to 6, wherein R.sub.2 and R.sub.3 independently of each other, are selected from the group consisting of H, methyl, ethyl, n-propyl, n-butyl, isopropyl, isobutyl and t-butyl.
[0370] 8. The method according to one or more of embodiments 1 to 7, wherein R.sub.2 and R.sub.3 are methyl.
[0371] 9. The method according to embodiment 1, wherein m is 1, 2 or 3.
[0372] 10. The method according to embodiment 1, wherein x and y are 0.
[0373] 11. The method according to one or more of embodiment 1 to 10, wherein the at least one carboxylic acid amide of general formula (I) and the at least one alkanolamine of general formula (II) are present in a molar ratio of 5:1 to 1:5.
[0374] 12. The method according to embodiment 1, wherein contacting in step (i) is achieved by immersing the foundry mold at a temperature in the range of 10 to 200.degree. C.
[0375] 13. The method according to embodiment 1, wherein the step (i) is carried out for a time period of .gtoreq.10 min. to .ltoreq.60 min.
[0376] 14. The method according to one or more of embodiments 1 to 13, further comprising the step of [0377] (ii) separating the sand from the mixture obtained in step (i).
[0378] 15. The method according to one or more of embodiments 1 to 14, further comprising the step of [0379] (iii) drying the sand obtained in step (ii).
[0380] 16. The method according to embodiment 1, wherein the composition comprises at least one surfactant.
[0381] 17. The method according to embodiment 16, wherein the at least one surfactant is a non-ionic surfactant.
[0382] 18. The method according to embodiment 17, wherein the non-ionic surfactant is selected from the group consisting of poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-alkyl ether, poly(C.sub.2-C.sub.4)alkylene glycol mono-(C.sub.8-C.sub.22)-carboxylic acid ester, alkoxylated oil and alkoxylated alkylamine.
[0383] 19. The method according to embodiments 17 or 18, wherein the non-ionic surfactant is selected from alkyl polyethylene glycol ether, ethoxylated castor oil and oleyl amine ethoxylate.
[0384] 20. The method according to one or more of embodiments 1 to 19, wherein the composition comprises water.
[0385] 21. A composition comprising [0386] (a) 2-hydroxypropanamide; and [0387] (b) monoethanolamine.
[0388] 22. Use of the composition according to embodiment 21 for disintegrating foundry mold.
[0389] 23. A method for disintegrating the foundry mold comprising sand and binder comprising the step of [0390] (i) contacting the foundry mold with the composition according to embodiment 21;
[0391] to obtain a mixture of the sand and the binder.
EXAMPLES
[0392] Compounds
[0393] Monoethanol Amine
[0394] Agnique.RTM. AMD 3L (N,N-Dimethyl lactamide)
[0395] Lutensol.RTM. FA12 (Oleylamine ethoxylate)
[0396] Lutensol.RTM. XL60 (C.sub.10-Guerbet alcohol ethoxylates)
[0397] Emulan.RTM. EL (Castor oil ethoxylate)
[0398] Agnique.RTM. AMD 10 (C.sub.10 fatty acid N,N-Dimethylamide)
[0399] Agnique.RTM. AMD 810 (C.sub.8/C.sub.10 fatty acid N,N-Dimethylamide) are available from BASF SE, Ludwigshafen, Germany.
[0400] Foundry mold sample of size 9 cm.times.2.2 cm.times.2.2 cm, made of PU-sand composite was pre-pared according to state of the art.
[0401] General Procedure
[0402] The foundry mold sample was immersed (one-third) in the composition as disclosed in Examples 1 to 6, at room temperature. The time of collapse of the sample was determined. The time of collapse is the time taken for the disintegration of the foundry mold sample completely resulting into the mixture of sand and binder.
TABLE-US-00001 Amount of carboxylic Amount Amount acid of of Time of Carboxylic amide alkanolamine surfactant collapse Example acid amide (wt. %) Alkanolamine (wt. %) Surfactant (wt. %) (minutes) 1 Agnique .RTM. 75 monoethanol 25 none -- 60 AMD 3L amine 2 Agnique .RTM. 25 monoethanol 25 Lutensol .RTM. 50 60 AMD 3L amine XL60 3 Agnique .RTM. 50 monoethanol 25 Lutensol .RTM. 25 60 AMD 3L amine FA12 4 Agnique .RTM. 50 monoethanol 25 Emulan .RTM. 25 60 AMD 3L amine EL 5 Agnique .RTM. 25 monoethanol 25 Lutensol .RTM. 50 35 AMD 10 amine XL60 6 Agnique .RTM. 25 monoethanol 25 Lutensol .RTM. 50 45 AMD 810 amine XL60
* * * * *

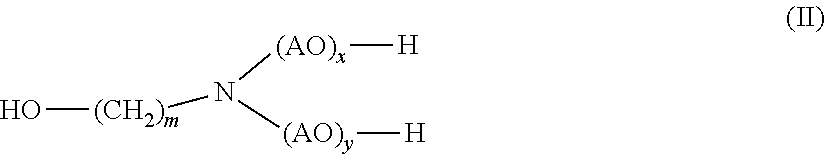




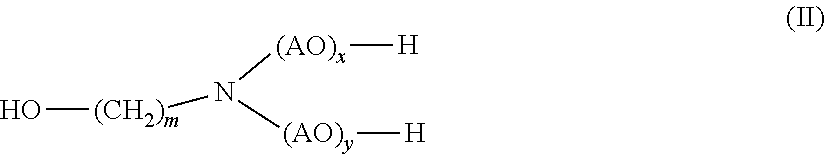
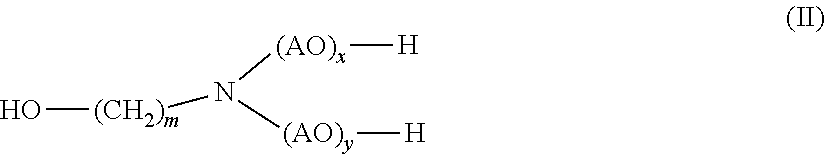





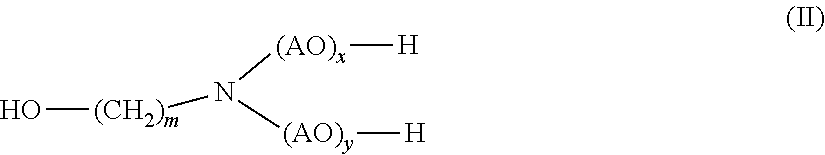










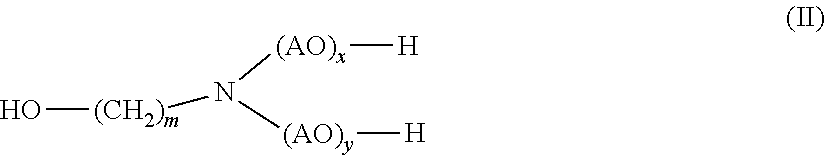



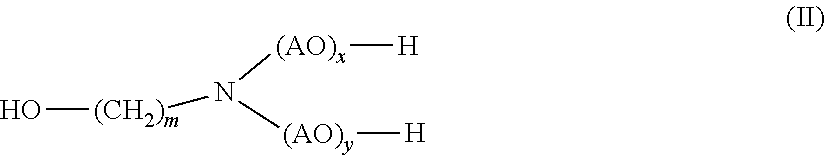
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.