Sustained Drug Delivery Implant
Shiah; Jane-Guo ; et al.
U.S. patent application number 16/984971 was filed with the patent office on 2021-04-22 for sustained drug delivery implant. The applicant listed for this patent is Allergan, Inc.. Invention is credited to Chetan Pujara, Jane-Guo Shiah.
| Application Number | 20210113458 16/984971 |
| Document ID | / |
| Family ID | 1000005312724 |
| Filed Date | 2021-04-22 |




| United States Patent Application | 20210113458 |
| Kind Code | A1 |
| Shiah; Jane-Guo ; et al. | April 22, 2021 |
SUSTAINED DRUG DELIVERY IMPLANT
Abstract
Biocompatible intraocular implants may include a brimonidine free base and a biodegradable polymer associated with the brimonidine free base to facilitate the release of the brimonidine free base into an eye with the polymer matrix lasts a period of time of not more than twice the drug release duration, but more than the drug release duration.
| Inventors: | Shiah; Jane-Guo; (Irvine, CA) ; Pujara; Chetan; (Irvine, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005312724 | ||||||||||
| Appl. No.: | 16/984971 | ||||||||||
| Filed: | August 4, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16354692 | Mar 15, 2019 | |||
| 16984971 | ||||
| 15350577 | Nov 14, 2016 | 10231926 | ||
| 16354692 | ||||
| 14218324 | Mar 18, 2014 | 9610246 | ||
| 15350577 | ||||
| 14181250 | Feb 14, 2014 | |||
| 14218324 | ||||
| 61765554 | Feb 15, 2013 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61F 9/0017 20130101; Y10S 514/953 20130101; A61K 47/32 20130101; Y10S 514/956 20130101; A61F 2210/0004 20130101; Y10S 514/954 20130101; A61K 9/0051 20130101; Y10S 514/912 20130101; Y10S 514/913 20130101; A61K 31/498 20130101 |
| International Class: | A61K 9/00 20060101 A61K009/00; A61F 9/00 20060101 A61F009/00; A61K 31/498 20060101 A61K031/498; A61K 47/32 20060101 A61K047/32 |
Claims
1. A solid intraocular implant for the treatment of a posterior ocular condition in a human patient, the solid intraocular implant comprising: a brimonidine free base in an amount of about 40% by weight to about 60% by weight of the implant, based on the total weight of the implant; and a biodegradable polymer matrix comprising an acid end-capped poly (D, L-lactide) polymer and a 75:25 poly (D,L-lactide-co-glycolide) polymer; wherein the weight ratio of the acid end-capped poly (D, L-lactide) polymer to the 75:25 poly (D,L-lactide-co-glycolide) polymer is 1:1; and wherein the implant has a polymer matrix degradation time in the range of about three months to about six months when placed in the eye of a human.
2. The implant of claim 1, wherein the brimonidine free base is present in an amount of about 50% by weight of the implant, based on the total weight of the implant.
3. The implant of claim 2, wherein the implant has a brimonidine free base delivery duration of two months to four months when placed in the eye of a human.
4. The implant of claim 1, wherein the acid end-capped poly (D, L-lactide) polymer is present in an about of about 25% by weight.
5. The implant of claim 1, wherein the 75:25 poly (D,L-lactide-co-glycolide) polymer is present in an about of about 25% by weight.
6. The implant of claim 5, wherein the total weight of the implant is about 800 .mu.g.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 16/354,692, filed Mar. 15, 2019, which is a continuation of U.S. patent application Ser. No. 15/350,577 filed Nov. 14, 2016, now U.S. Pat. No. 10,231,926, issued Mar. 19, 2019, which is a continuation of U.S. patent application Ser. No. 14/218,324 filed Mar. 18, 2014, now issued as U.S. Pat. No. 9,610,246, which is a continuation of U.S. patent application Ser. No. 14/181,250 filed Feb. 14, 2014, now abandoned, which claims the benefit of U.S. Provisional Application No. 61/765,554 filed Feb. 15, 2013, the entire content of each application is incorporated herein by reference.
BACKGROUND
Field
[0002] The disclosure of the present application generally relates to drug delivery implants, and more specifically, drug delivery implants used to treat ocular conditions.
Description of the Related Art
[0003] Diabetic retinopathy is the leading cause of blindness among adults aged 20 to 74 years. It is estimated that 75,000 new cases of macular edema, 65,000 cases of proliferative retinopathy, and 12,000 to 24,000 new cases of blindness arise each year. Retinitis pigmentosa (RP) is a heterogeneous group of inherited neurodegenerative retinal diseases that cause the death of photoreceptor cells (rods and cones) that eventually leads to blindness. Glaucoma is a multifactorial optic neuropathy resulting from loss of retinal ganglion cells, corresponding atrophy of the optic nerve, and loss of visual function, which is manifested predominantly by visual field loss and decreased visual acuity and color vision. Geographic atrophy ("GA") is one of 2 forms of the advanced stage of Age-Related Macular Degeneration ("AMD"). The advanced stage of AMD refers to that stage in which visual acuity loss can occur from AMD. Retinal detachments are a significant cause of ocular morbidity. There are 3 types of retinal detachment: rhegmatogenous, tractional, and exudative.
[0004] Brimonidine (5-bromo-6-(2-imidazolidinylideneamino) quinoxaline) is an alpha-2-selective adrenergic receptor agonist effective for treating open-angle glaucoma by decreasing aqueous humor production and increasing uveoscleral outflow. Brimonidine tartrate ophthalmic solution 0.2% (marketed as ALPHAGAN.RTM.) was approved by the US Food and Drug Administration (FDA) in September 1996 and in Europe in March 1997 (United Kingdom). Brimonidine tartrate ophthalmic solution with Purite.RTM. 0.15% and 0.1% (marketed as ALPHAGAN.RTM. P) was approved by the FDA in March 2001 and August 2005, respectively. These formulations are currently indicated for lowering IOP in patients with open-angle glaucoma (OAG) and ocular hypertension (OHT).
[0005] A neuroprotective effect of brimonidine tartrate has been shown in animal models of optic nerve crush, moderate ocular hypertension, pressure-induced ischemia, and vascular ischemia. The neuroprotective effect of topical applications of brimonidine tartrate has also been explored clinically in patients with glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, and acute non-arteritic anterior ischemic optic neuropathy. However, certain limitations exist with the use of brimonidine tartrate in intraocular implants. For example, because of the size of the brimonidine tartrate molecule, the amount of drug that can be loaded into an implant may be limited. Also, the hydrophilic nature of brimonidine tartrate may limit the ability of the drug's use in sustained release formulations.
SUMMARY
[0006] Accordingly, an embodiment provides an intraocular implant for the treatment of a posterior ocular condition in a human patient including a biodegradable polymer matrix including at least one biodegradable polymer and a brimonidine free base agent, wherein the implant can be configured to deliver the brimonidine free base agent to the vitreous of an eye of a patient suffering from a posterior ocular condition for a brimonidine free base agent delivery duration of up to six months and wherein the biodegradable polymer matrix is configured to completely or almost completely degrade, once placed into the vitreous of the eye, within a period of time of about two times the brimonidine free base agent delivery duration or less. In some embodiments, the brimonidine free base agent is present in the implant in an amount of about 50% by weight of the implant, based on the total weight of the implant. In some embodiments, the implant can have a rod shape, and the rod shape can have a rod diameter of about 350 .mu.m and a rod length of about 6 mm. According to other embodiments, the brimonidine free base agent is dispersed within the biodegradable polymer matrix. In some embodiments, the at least one biodegradable polymer includes poly(D,L-lactide-co-glycolide) and poly(D,L-lactide). In some embodiments, the biodegradable polymer matrix includes at least one polymer selected from the group consisting of acid-end capped poly(D,L-lactide-co-glycolide) and acid-end capped poly(D,L-lactide). In some embodiments, the brimonidine free base agent delivery duration is in the range of about 1 month to about 6 months.
[0007] These and other embodiments are described in greater detail below.
BRIEF DESCRIPTION OF THE FIGURES
[0008] These and other features will now be described with reference to the drawings summarized below. These drawings and the associated description are provided to illustrate one or more embodiments and not to limit the scope of the invention.
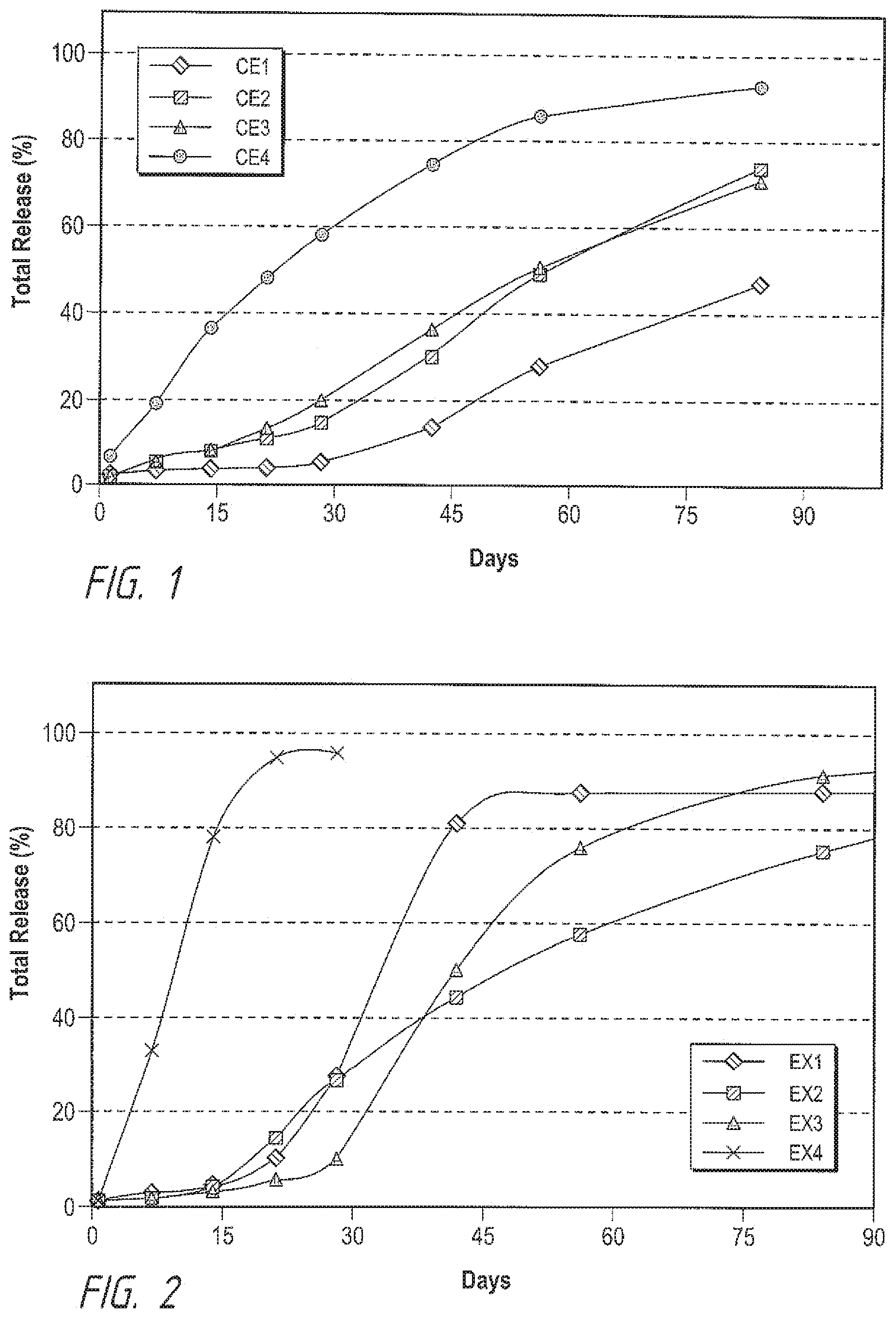
[0009] FIG. 1 illustrates brimonidine tartrate implant formulation drug release profiles in 0.01 M PBS with a pH of 7.4 at 37.degree. C., according to comparative example formulations.
[0010] FIG. 2 shows brimonidine free base implant formulation drug release profiles in 0.01 M PBS with a pH of 7.4 at 37.degree. C., according to example formulations.
[0011] FIG. 3 shows brimonidine tartrate implant formulation drug release profiles in Albino rabbits, according to comparative example formulations.
[0012] FIG. 4 shows brimonidine tartrate implant formulation drug release profiles in Cyno monkeys, according to comparative example formulations.
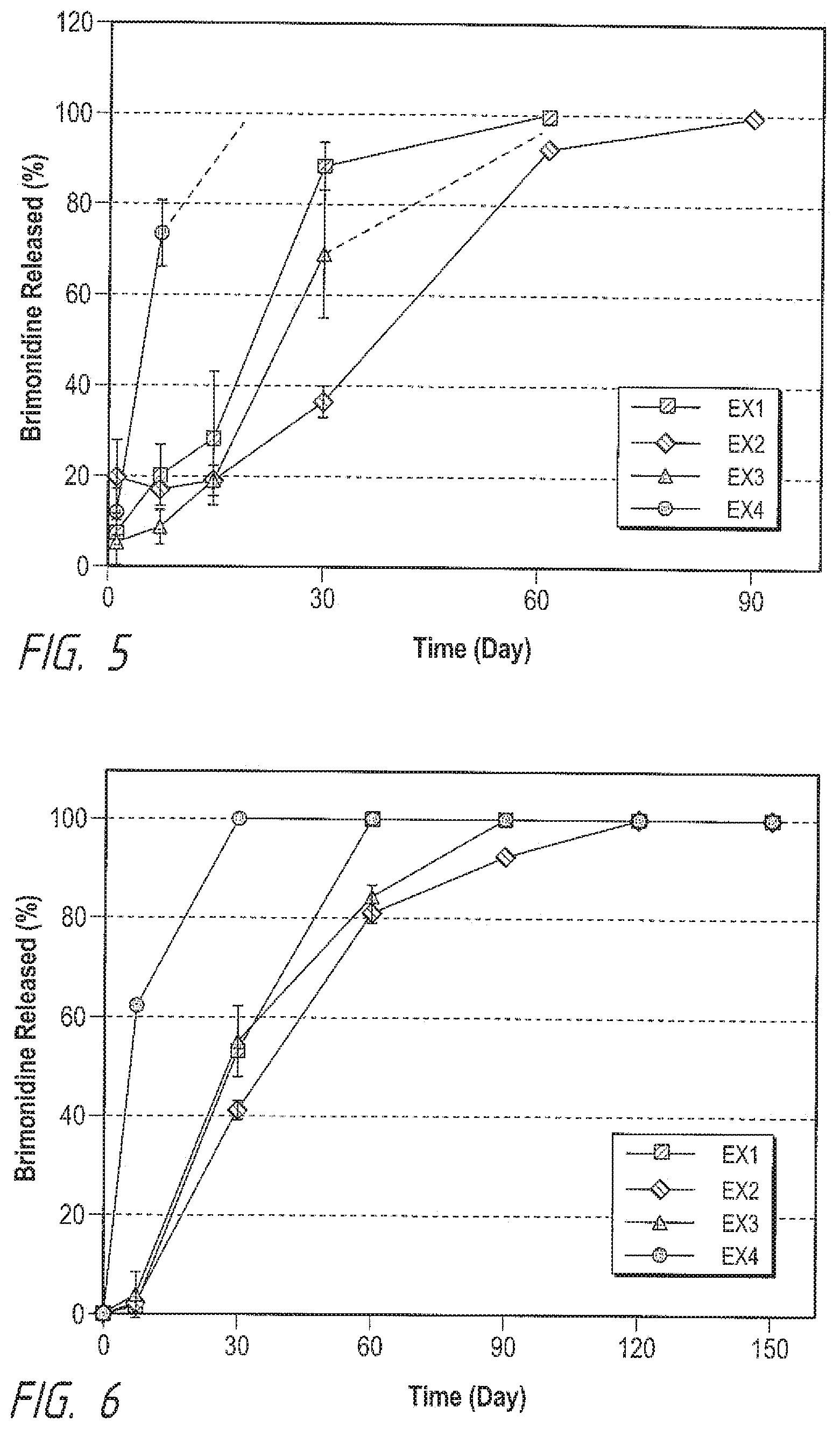
[0013] FIG. 5 illustrates brimonidine free base implant formulation drug release profiles in Albino rabbits, according to example formulations.
[0014] FIG. 6 illustrates brimonidine free base implant formulation drug release profiles in Cyno monkeys, according to example formulations.
[0015] FIG. 7 shows the drug concentration of brimonidine tartrate implant formulations in the retina (optic nerve) of Albino rabbits over time according to comparative example formulations. The dotted line indicates the human .alpha.2A EC90 concentration.
[0016] FIG. 8 shows the drug concentration of brimonidine free base implant formulations in the retina (optic nerve) of Albino rabbits over time according to example formulations. The dotted line indicates the human .alpha.2A EC90 concentration.
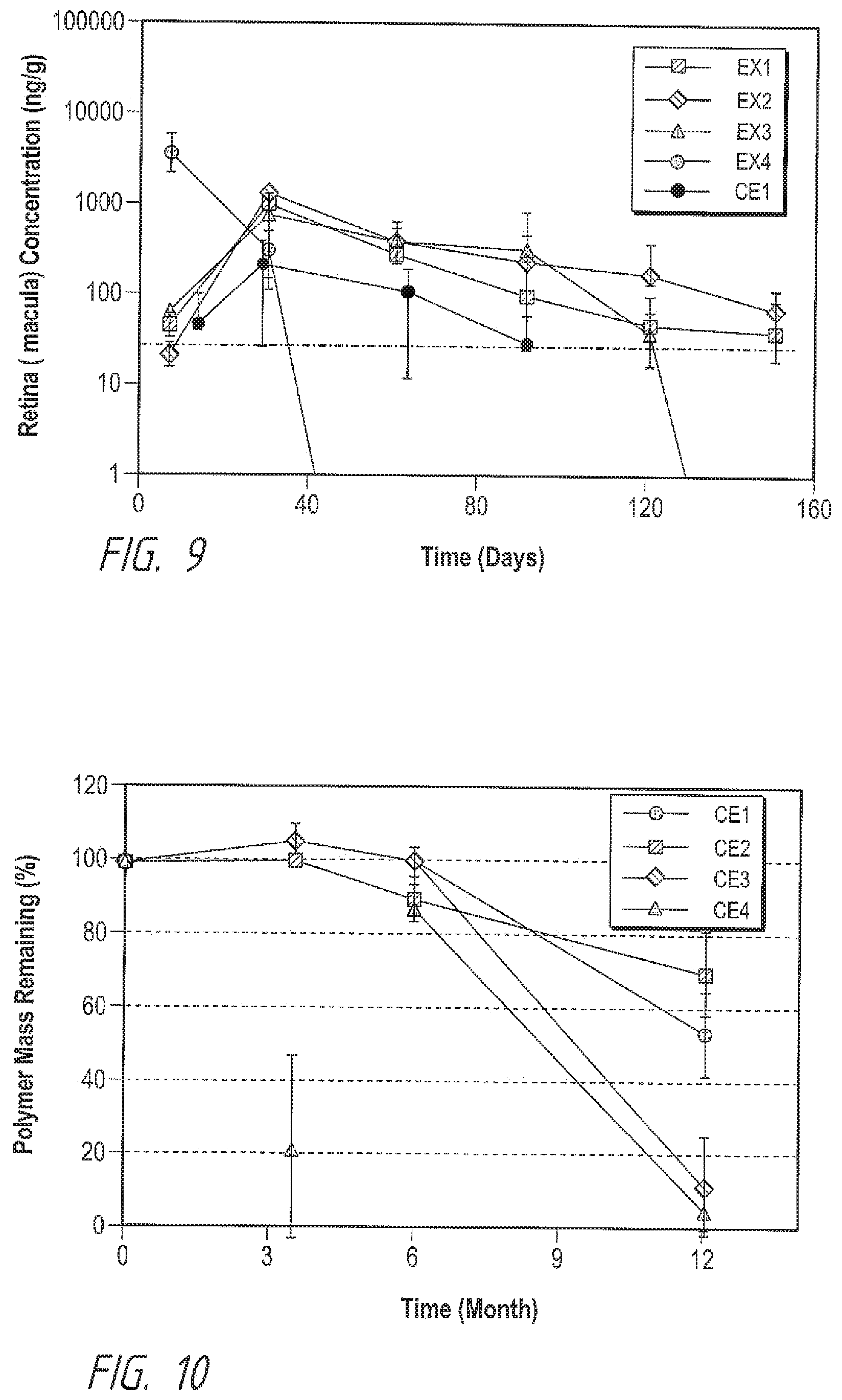
[0017] FIG. 9 illustrates the drug concentration of brimonidine free base implant formulations in the retina (macula) of Cyno monkeys over time according to example formulations. The dotted line indicates the human .alpha.2A EC90 concentration. For comparison, the CE1 brimonidine formulation is included.
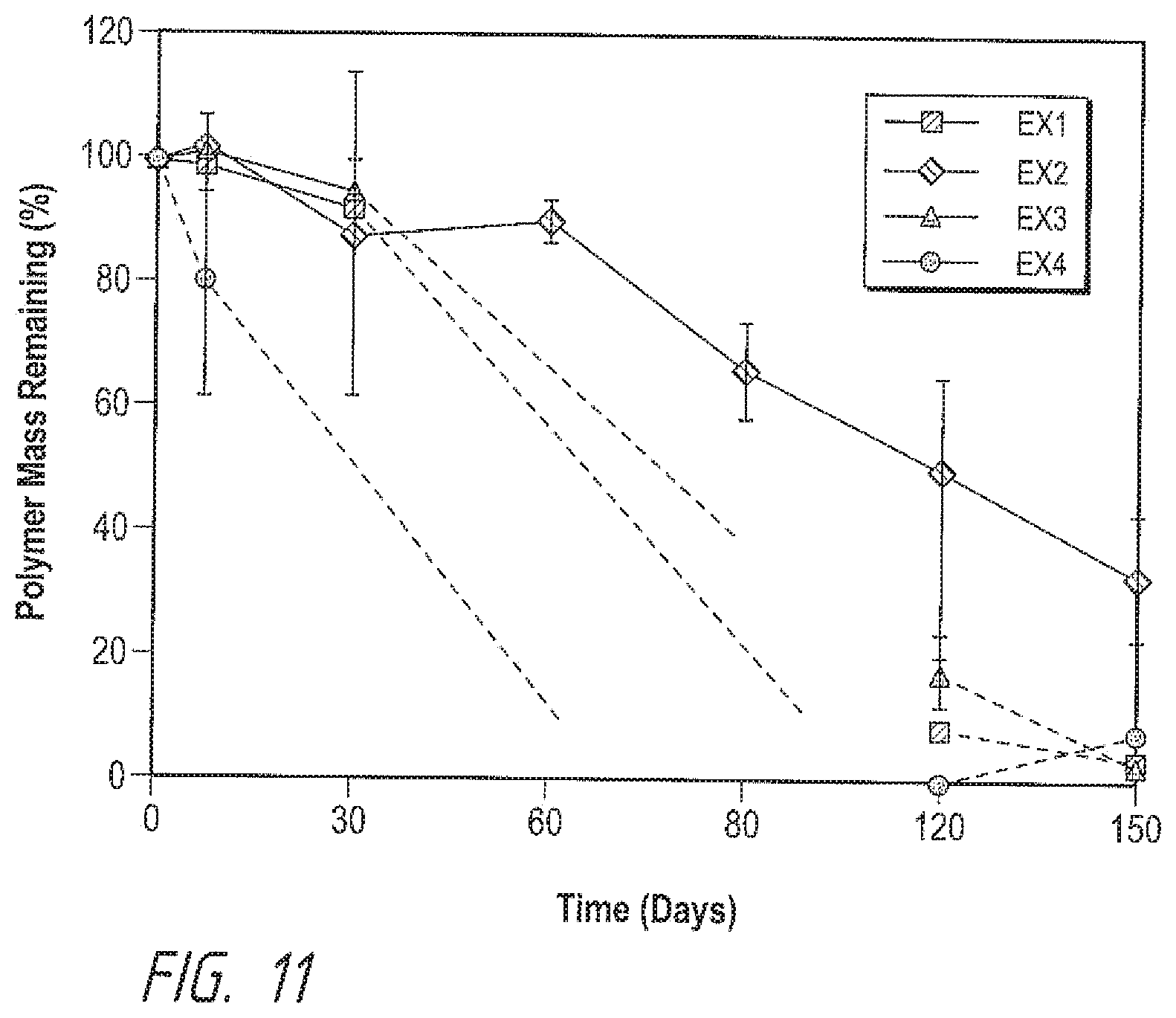
[0018] FIG. 10 illustrates the polymer matrix degradation of brimonidine tartrate implant formulations in Cyno monkeys over time, according to comparative example formulations.
[0019] FIG. 11 shows the polymer matrix degradation of brimonidine free base implant formulations in Cyno monkeys over time, according to example formulations.
[0020] FIG. 12 shows the implant image when incubating in PBS (pH 7.4, 0.01N) at 37.degree. C.
DETAILED DESCRIPTION
[0021] In general terms, an embodiment relates to brimonidine free base sustained delivery for back-of-the-eye therapeutic applications. In some embodiments, the brimonidine free base is formulated into an implant with one or more polymers in a polymer matrix, the polymers selected in order to give a target sustained delivery of the brimonidine free base and/or a target degradation of the one or more polymers. According to some embodiments, formulations of brimonidine free base and biodegradable polymer or polymers are created such that the polymer matrix will be degraded within a period of not more than twice the brimonidine free base release duration, but more than the brimonidine free base release duration. According to some embodiments, the brimonidine free base drug delivery system exhibits a target drug delivery duration of one to six months and a target matrix degradation time of two to twelve months.
[0022] Embodiments herein disclose new drug delivery systems, and methods of making and using such systems, for extended or sustained drug release into an eye, for example, to achieve one or more desired therapeutic effects. The drug delivery systems can be in the form of implants or implant elements that can be placed in an eye. The systems and methods disclosed in some embodiments herein can provide for extended release time of one or more therapeutic agent or agents. Thus, for example, a patient who has received such an implant in their eye can receive a therapeutic amount of an agent for a long or extended time period without requiring additional administrations of the agent. According to some embodiments an implant may also only remain within the eye of a patient for a targeted or limited amount of time before it degrades completely or nearly completely. By limiting the amount of time a foreign object, such as an implant is in a patient's eye or vitreous, a patient's comfort is optimized and their risk for infection or other complications is minimized. Also, complications that may arise from an implant colliding with the cornea or other part of the eye in the dynamic fluid of the vitreous can be avoided.
[0023] As used herein, an "intraocular implant" refers to a device or elements that is structured, sized, or otherwise configured to be placed in an eye. Intraocular implants are generally biocompatible with physiological conditions of an eye. Intraocular implants may be placed in an eye without disrupting vision of the eye.
[0024] As used herein, "therapeutic component" refers to a portion of an intraocular implant comprising one or more therapeutic agents or substances used to treat a medical condition of the eye. The therapeutic component may be a discrete region of an intraocular implant, or it may be homogenously distributed throughout the implant. The therapeutic agents of the therapeutic component are typically ophthalmically acceptable, and are provided in a form that does not cause adverse reactions when the implant is placed in the eye.
[0025] As used herein, an "ocular condition" is a disease ailment or condition which affects or involves the eye or one of the parts or regions of the eye. The eye can include the eyeball and the tissues and fluids that constitute the eyeball, the periocular muscles (such as the oblique and rectus muscles) and the portion of the optic nerve which is within or adjacent the eyeball.
[0026] An "anterior ocular condition" is a disease, ailment, or condition which affects or which involves an anterior (i.e. front of the eye) ocular region or site, such as a periocular muscle, an eye lid or an eye ball tissue or fluid which is located anterior to the posterior wall of the lens capsule or ciliary muscles. Thus, an anterior ocular condition can affect or involve the conjunctiva, the cornea, the anterior chamber, the iris, the posterior chamber (located behind the retina, but in front of the posterior wall of the lens capsule), the lens or the lens capsule and blood vessels and nerve which vascularize or innervate an anterior ocular region or site.
[0027] A "posterior ocular condition" is a disease, ailment or condition which primarily affects or involves a posterior ocular region or site such as choroid or sclera (in a position posterior to a plane through the posterior wall of the lens capsule), vitreous, vitreous chamber, retina, optic nerve or optic disc, and blood vessels and nerves that vascularize or innervate a posterior ocular region or site.
[0028] Thus a posterior ocular condition can include a disease, ailment or condition such as, but not limited to, acute macular neuroretinopathy; Behcet's disease; geographic atrophy; choroidal neovascularization; diabetic uveitis; histoplasmosis; infections, such as fungal, bacterial, or viral-caused infections; macular degeneration, such as acute macular degeneration, non-exudative age related macular degeneration and exudative age related macular degeneration; edema, such as macular edema, cystoids macular edema and diabetic macular edema; multifocal choroiditis; ocular trauma which affects a posterior ocular site or location; ocular tumors; retinal disorders, such as central retinal vein occlusion, diabetic retinopathy (including proliferative diabetic retinopathy), proliferative vitreoretinopathy (PVR), retinal arterial occlusive disease, retinal detachment, uveitic retinal disease; sympathetic ophthalmia; Vogt Koyanagi-Harada (VKH) syndrome; uveal diffusion; a posterior ocular condition caused by or influenced by an ocular laser treatment; or posterior ocular conditions caused by or influenced by a photodynamic therapy, photocoagulation, radiation retinotherapy, epiretinal membrane disorders, branch retinal vein occlusion, anterior ischemic optic neuropathy, non-retinopathy diabetic retinal dysfunction, retinitis pigmentosa, and glaucoma. Glaucoma can be considered a posterior ocular condition because the therapeutic goal is to prevent the loss of or reduce the occurrence of loss of vision due to damage to or loss of retinal cells or optic nerve cells (e.g. neuroprotection).
[0029] The terms "biodegradable polymer" or "bioerodible polymer" refer to a polymer or polymers which degrade in vivo, and wherein erosion of the polymer or polymers over time occurs concurrent with and/or subsequent to the release of a therapeutic agent. A biodegradable polymer may be a homopolymer, a copolymer, or a polymer comprising more than two polymeric units. In some embodiments, a "biodegradable polymer" may include a mixture of two or more homopolymers or copolymers.
[0030] The terms "treat", "treating", or "treatment" as used herein, refer to reduction or resolution or prevention of an ocular condition, ocular injury or damage, or to promote healing of injured or damaged ocular tissue.
[0031] The term "therapeutically effective amount" as used herein, refers to the level or amount of therapeutic agent needed to treat an ocular condition, or reduce or prevent ocular injury or damage.
[0032] Those skilled in the art will appreciate the meaning of various terms of degree used herein. For example, as used herein in the context of referring to an amount (e.g., "about 6%"), the term "about" represents an amount close to and including the stated amount that still performs a desired function or achieves a desired result, e.g. "about 6%" can include 6% and amounts close to 6% that still perform a desired function or achieve a desired result. For example, the term "about" can refer to an amount that is within less than 10% of, within less than 5% of, within less than 0.1% of, or within less than 0.01% of the stated amount.
[0033] Intraocular implants can include a therapeutic component and a drug release control component or components. The therapeutic agent can comprise, or consist essentially of an alpha-2 adrenergic receptor agonist. The alpha-2 adrenergic receptor agonist may be an agonist or agent that selectively activates alpha-2 adrenergic receptors, for example by binding to an alpha-2 adrenergic receptor, relative to other types of adrenergic receptors, such as alpha-1 adrenergic receptors. The selective activation can be achieved under different conditions, such as conditions associated with the eye of a human patient.
[0034] The alpha-2 adrenergic receptor agonist of the implant is typically an agent that selectively activates alpha-2 adrenergic receptors relative to alpha-2 adrenergic receptors. In certain implants, the alpha-2 adrenergic receptor agonist selectively activates a subtype of the alpha-2 adrenergic receptors. For example, the agonist may selectively activate one or more of the alpha-2a, the alpha-2b, or the alpha-2c receptors, under certain conditions, such as physiological conditions. Under other conditions, the agonist of the implant may not be selective for alpha-2 adrenergic receptor subtypes. The agonist may activate the receptors by binding to the receptors, or by any other mechanism.
[0035] According to some embodiments, the alpha-2 receptor antagonist used is brimonidine. Brimonidine is a quinoxaline derivative having the structure:
##STR00001##
[0036] Brimonidine, an organic base, is publicly available as brimonidine free base. Brimonidine free base is generally hydrophobic.
[0037] In some embodiments, the alpha-2 adrenergic receptor antagonist may be a pharmaceutically acceptable acid addition salt of brimonidine. One such salt can be brimonidine tartrate (AGN 190342-F, 5-bromo-6-(2-imidazolidinylideneamino) quinoxaline tartrate). Both brimonidine free base and brimonidine tartrate are chemically stable and have melting points higher than 200.degree. C.
[0038] Thus, an intraocular implant can comprise, consist of, or consist essentially of a therapeutic agent such as an alpha-2 adrenergic receptor agonist such as a brimonidine salt alone (such as brimonidine tartrate), a brimonidine free base alone, or mixtures thereof.
[0039] The use of brimonidine free base in solid implant formulations has several advantages over brimonidine tartrate, such as the lower solubility of brimonidine free base lowers potential drug burst effect, and the free base drug equivalent dose per implant can be higher under the same weight. Thus, according to some embodiments, no brimonidine tartrate is included in an intraocular implant. According to some embodiment, the only therapeutic agent used in an intraocular implant is brimonidine free base.
[0040] The alpha-2 adrenergic receptor agonist may be in a particulate or powder form and entrapped by the biodegradable polymer matrix. According to an embodiment, the alpha-2 adrenergic receptor agonist is a brimonidine free base having a D90 particle size of less than about 20 .mu.m. According to another embodiment, the alpha-2 adrenergic receptor agonist is a brimonidine free base having a D90 particle size of less than about 10 .mu.m. According to another embodiment, the alpha-2 adrenergic receptor agonist is a brimonidine free base having a D90 particle size in the range of about 10 .mu.m to about 20 .mu.m.
[0041] According to some embodiments, implants can be formulated with particles of the brimonidine free base agent dispersed within the bioerodible polymer matrix. According to some embodiments, the implants can be monolithic, having the therapeutic agent homogenously distributed through the biodegradable polymer matrix, or encapsulated, where a reservoir of active agent is encapsulated by the polymeric matrix. In some embodiments, the therapeutic agent may be distributed in a non-homogeneous pattern in the biodegradable polymer matrix. For example, in an embodiment, an implant may include a first portion that has a greater concentration of the therapeutic agent (such as brimonidine free base) relative to a second portion of the implant.
[0042] The alpha-2 adrenergic receptor agonist can be present in an implant in an amount in the range of about 20% to about 70% by weight of the implant, based on the total weight of the implant. In some embodiments, the alpha-2 adrenergic receptor agonist can be present in an implant in an amount in the range of about 40% to about 60% by weight of the implant, based on the total weight of the implant. In an embodiment, the alpha-2 adrenergic receptor agonist can be present in an implant in an amount of about 40% by weight of the implant, based on the total weight of the implant. In another embodiment, the alpha-2 adrenergic receptor agonist can be present in an implant in an amount of about 50% by weight of the implant, based on the total weight of the implant. In an example embodiment, brimonidine free base can be present in an implant in an amount of about 50% by weight of the implant, about 55% by weight of the implant, about 60% by weight of the implant, or about 70% by weight of the implant, based on the total weight of the implant.
[0043] Suitable polymeric materials or compositions for use in the implant can include those materials which are compatible with the eye so as to cause no substantial interference with the functioning or physiology of the eye. Such materials can be at least partially or fully biodegradable.
[0044] Examples of suitable polymeric materials for the polymer matrix include polyesters. For example, polymers of D-lactic acid, L-lactic acid, racemic lactic acid, glycolic acid, polycaprolactone, and combinations thereof may be used for the polymer matrix. In some embodiments, a polyester, if used, may be a homopolymer, a copolymer, or a mixture thereof.
[0045] In some implants, copolymers of glycolic acid and lactic acid are used, where the rate of biodegradation can be controlled, in part, by the ratio of glycolic acid to lactic acid. The mol percentage (% mol) of polylactic acid in the polylactic acid polyglycolic acid (PLGA) copolymer can be between 15 mol % and about 85 mol %. In some embodiments, the mol percentage of polylactic acid in the (PLGA) copolymer is between about 35 mol % and about 65 mol %. In some embodiments, a PLGA copolymer with 50 mol % polylactic acid and 50 mol % polyglycolic acid can be used in the polymer matrix.
[0046] The polymers making up the polymer matrix may also be selected based on their molecular weight. Different molecular weights of the same or different polymeric compositions may be included in the implant to modulate the release profile. In some embodiments, the release profile of the therapeutic agent and the degradation of the polymer may be affected by the molecular weight of one or more polymers in the polymer matrix. In some embodiments, the molecular weight of one or more poly (D,L-lactide) components may be advantageously selected to control the release of the therapeutic agent and the degradation of the polymer. According to some embodiments, the average molecular weight of a polymer, such as poly (D,L-lactide), may be "low." According to some embodiments, the average molecular weight of a polymer, such as poly (D,L-lactide), may be "medium." According to some embodiments, only low molecular weight poly(D,L-lactide) is included in a polymer matrix in an intraocular implant. According to some embodiments, high molecular weight (Mw) poly(D,L-lactide)s are not present in the biodegradable polymer matrix or they are only present in a negligible amount (about 0.1% by weight of an implant, based on the total weight of the implant). By limiting the amount of high molecular weight poly(D,L-lactide) present in an implant, the matrix degradation duration may be shortened.
[0047] Some example polymers that may be used alone or in combination to form the polymer matrix include those listed in TABLE A below, the data sheets of the commercially available polymers are incorporated by reference, in their entirety:
TABLE-US-00001 TABLE A Trade Name of Molecular Commercially Weight Available Intrinsic (low, Polymer (From Viscosity medium, EVONIK) Polymer (dL/g) high) RG502S 50:50 poly (D, L- 0.16-0.24 low lactide-co-glycolide) RG502H 50:50 poly (D, L- 0.16-0.24 low lactide-co-glycolide), acid end capped RG504 50:50 poly (D, L- 0.45-0.60 medium lactide-co-glycolide) RG505 50:50 poly (D, L- 0.61-0.74 medium lactide-co-glycolide) RG752S 75:25 poly (D, L- 0.16-0.24 low lactide-co-glycolide) RG755 75:25 poly (D, L- 0.50-0.70 medium lactide-co-glycolide) RG858S 85:15 poly (D, L- 1.3-1.7 medium lactide-co-glycolide) R202H poly (D, L-lactide), 0.16-0.24 low acid end capped R203S poly (D, L-lactide) 0.25-0.35 medium R208 poly (D, L-lactide) 1.8-2.2 high
[0048] The biodegradable polymer matrix of the intraocular implant can comprise a mixture of two or more biodegradable polymers. In some embodiments, only one biodegradable polymer listed above is used in the biodegradable polymer matrix. In some embodiments, any one of the biodegradable polymers listed in the above chart can be used in an amount in the range of 12.5% w/w to 70% w/w each in a drug delivery system or implant. In some embodiments, any one of the biodegradable polymers listed in the above chart can be used in an amount in the range of 25% w/w to 50% w/w each in a drug delivery system or implant. In some embodiments, any one of the biodegradable polymers listed in the above chart can be used in an amount in the range of 20% w/w to 40% w/w each in a drug delivery system or implant. In some embodiments, any one of the biodegradable polymers listed in the above chart can be used in an amount of about 15% w/w, about 25% w/w, about 12.5% w/w, about 37.5% w/w, about 40% w/w, about 50% w/w, or about 60% w/w each in a drug delivery system or implant. For example, the implant may comprise a mixture of a first biodegradable polymer and a different second biodegradable polymer. One or more of the biodegradable polymers may have terminal acid groups.
[0049] In some embodiments, release of a therapeutic agent from a biodegradable polymer matrix in an intraocular implant can be the consequence of various mechanisms and considerations. Release of the agent can be achieved by erosion of the biodegradable polymer matrix followed by exposure of previously embedded drug particles to the vitreous of an eye receiving the implant, and subsequent dissolution and release of the therapeutic agent. The release kinetics by this form of drug release are different than that through formulations which release agent by polymer swelling alone, such as with hydrogel or methylcellulose. The parameters which may determine the release kinetics include the size of the drug particles, the water solubility of the drug, the ratio of drug to polymer, and the erosion rate of the polymers.
[0050] According to some embodiments, compositions and methods extend the brimonidine free base delivery in the vitreous with concomitantly moderate matrix degradation duration. The sustained ocular drug delivery can be achieved by formulating brimonidine free base with properly selected blend of bioerodible poly(D,L-lactide) and/or poly(D,L-lactide-co-glycolide).
[0051] According to some example embodiments, a drug delivery system or implant can contain a polymer matrix with an acid-capped poly (D,L-lactide) in an amount in the range of 25% w/w to about 50% w/w. According to some example embodiments, a drug delivery system or implant can contain a polymer matrix with an acid-capped 50:50 poly (D,L-lactide-co-glycolide) in an amount in the range of about 25% w/w to about 50% w/w or about 37.5% to about 50% w/w of the implant. According to some example embodiments, a drug delivery system or implant can contain a polymer matrix with an acid-capped 75:25 poly (D,L-lactide-co-glycolide) in an amount in the range of about 25% w/w to about 50% w/w or about 15% w/w to about 50% w/w of the implant. According to some example embodiments, a drug delivery system or implant can contain a polymer matrix with an acid-capped 85:15 poly (D,L-lactide-co-glycolide) in an amount in the range of about 25% w/w to about 50% w/w or about 30% to about 60% w/w of the implant.
[0052] The drug delivery systems are designed to release brimonidine free base at therapeutic levels to the vitreous for a sustained period of time (the brimonidine free base delivery duration), then degrade over period of time in the range of half the brimonidine free base delivery duration to a time equivalent to the brimonidine free base delivery duration. According to other embodiments, the drug delivery system including the polymer matrix can degrade over a period of time of about one quarter the brimonidine free base delivery duration to about one half the brimonidine free base delivery duration. According to other embodiments, the drug delivery system including the polymer matrix can degrade over a period of time of about one third the brimonidine free base delivery duration to about one half the brimonidine free base delivery duration. According to other embodiments, the drug delivery system including the polymer matrix can degrade over a period of time equivalent to about the brimonidine free base delivery duration to about twice the brimonidine free base delivery duration. For example, in an embodiment, an intraocular implant may include a mixture of brimonidine free base and a biodegradable polymer matrix that releases brimonidine free base over a period of time of three months, then the polymer matrix degrades for a period of an additional 2 months until the implant is completely degraded or almost completely degraded. According to some embodiments, the brimonidine free base delivery duration is a period of time in the range of about 1 month to about 6 months, about 1 month to about 5 months, about 1 month to about 3 months, about 1 month to about 4 months, about 2 months to about 4 months, or about 3 months to about 6 months. According to some embodiments, the polymer matrix degradation time for the total drug delivery system is in the range of about 1 month to about 7 months, about 1 month to about 6 months, about 3 months to about 7 months, about 1 month to about 4 months, about 3 months to about 4 months, about 4 months to about 5 months, about 5 months to about 7 months, or about 3 months to about 6 months. According to some embodiments, the polymer matrix degradation time for the drug delivery system is fewer than 10 weeks, fewer than 8 weeks, fewer than 6 weeks, or fewer than 4 weeks.
[0053] According to one example embodiment, a biodegradable intraocular implant comprises brimonidine free base associated with a biodegradable polymer matrix, which comprises a mixture of different biodegradable polymers. The brimonidine free base is present in the implant in an amount of 50% by weight, based on the total weight of the implant. A first biodegradable polymer is an acid end capped poly (D,L-lactide) having an inherent viscosity of between 0.16 dL/g and 0.24 dL/g, and comprising 25% by weight of the implant, based on the total weight of the implant. A second biodegradable polymer is a PLGA copolymer having 75 mol % polylactic acid and 25 mol % polyglycolic acid. The PLGA copolymer has an inherent viscosity of between 0.16 dL/g and 0.24 dL/g, and the PLGA copolymer comprises 25% of weight of the implant, based on the total weight of the implant. Such a mixture is effective in releasing an effective amount of the brimonidine free base over a delivery duration of about three months, then degrading the polymer matrix over the span of one-two additional months, less than twice the brimonidine free base delivery duration.
[0054] According to another example embodiment, a biodegradable intraocular implant comprises brimonidine free base associated with a biodegradable polymer matrix, which comprises a single type of biodegradable polymer. The brimonidine free base is present in the implant in an amount of 50% by weight, based on the total weight of the implant. In this embodiment, the biodegradable polymer matrix is made of a PLGA copolymer having 85 mol % polylactic acid and 15 mol % polyglycolic acid. The PLGA copolymer has an inherent viscosity of between 1.3 dL/g and 1.7 dL/g, and the PLGA copolymer comprises 50% of weight of the implant, based on the total weight of the implant. Such a mixture is effective in releasing an effective amount of the brimonidine free base over a delivery duration of about three or four months, then degrading the polymer matrix over the span of one-two additional months, less than twice the brimonidine free base delivery duration.
Manufacture of Implants
[0055] According to some embodiments, intraocular implants can be formed through suitable polymer processing methods. In an embodiment, a mixture of a therapeutic agent (such as brimonidine free base) may be blended with PLA and/or PLGA polymers in a mixer, such as a Turbula mixer. In an embodiment, the intraocular implants are formed by extrusion. Extrusion can be performed by a suitable extruder, such as a Haake extruder. After the therapeutic agent and the polymer matrix have been blended together, they can then be force fed into an extruder and extruded into filaments. The extruded filaments may then be cut into implants with a target weight. In some embodiments, a 800 .mu.g implant may be cut to deliver about 300 .mu.g, 400 .mu.g, or 500 .mu.g of drug over the brimonidine free base delivery duration. Implants can then be loaded into an injection device, such as a 25 G applicator and sterilized. According to some embodiments, the extruded filaments are cut to a weight of less than 1000 .mu.g, less than 800 .mu.g, or less than 600 .mu.g. In some embodiments, the implants can be gamma sterilized. The implants can be gamma sterilized at doses such as 20 kGy to 60 kGy, 25 kGy to 50 kGy, 25 kGy to 40 kGy, and the like.
Methods for Treatment
[0056] According to an embodiment, a method for treating a posterior ocular condition includes administering an implant, such as the implants disclosed herein, to a posterior segment of an eye of a human or animal patient, and preferably a living human or animal. In some embodiments, a method of treating a patient may include placing the implant directly into the posterior chamber of the eye. In some embodiments, a method of treating a patient may comprise administering an implant to the patient by at least one of intravitreal injection, subconjunctival injection, subtenon injections, retrobulbar injection, and suprachoroidal injection.
[0057] In at least one embodiment, a method of treating retinitis pigmentosa, glaucoma, macular degeneration, and/or geographic atrophy in a patient comprises administering one or more implants containing brimonidine free base, as disclosed herein, to a patient by at least one of intravitreal injection, subconjunctival injection, sub-tenon injection, retrobulbar injection, and suprachoroidal injection. A syringe apparatus including an appropriately sized needle, for example, a 27 gauge needle or a 30 gauge needle, can be effectively used to inject the composition with the posterior segment of an eye of a human or animal. According to some embodiments, no more than one injection is administered to the patient to treat the condition. According to other embodiments, more than one injection is administered to the patient to treat the condition.
Examples
[0058] Example intraocular implants containing brimonidine tartrate or brimonidine free base and a biodegradable polymer matrix were created and tested for their release and degradation properties. The brimonidine tartrate or brimonidine free base was first weighed and blended with PLA and/or PLGA polymers in a Turbula mixer for 30 minutes. The resulting powder blend was then fed to the Haake extruder by a force feeder. The extruded filaments were cut to implants with a target weight, e.g., 857 .mu.g or 800 .mu.g to deliver 300 .mu.g brimonidine tartrate or 400 .mu.g brimonidine free base per implant. Implants were loaded into 25 G applicators and gamma-sterilized at 25 to 40 kGy dose. The potency per implant was confirmed by a HPLC assay.
[0059] Examples and Comparative Examples of formulation compositions using brimonidine tartrate (as Comparative Examples 1-4) and brimonidine free base (Examples 1-4) as the drug are shown in Tables B and C, and their drug release profiles are shown in FIGS. 1 and 2, respectively. In FIGS. 1 and 2, they axis is number of days and they axis is the percentage (%) of total release. For in vitro drug release testing, four implants per each formulation were randomly cut from extruded filaments, gamma sterilized, and incubated in 10 mL of 0.01M PBS pH 7.4 in a shaking water bath set at 37.degree. C. and 50 rpm. The drug release was sampled at designated time point, and the drug content was analyzed by a HPLC assay. The release medium was completely replaced with fresh medium during each sampling time point. The polymer Mw degradation rate constant k, as determined by incubating implant samples in 0.01M PBS pH 7.4 at 25.degree. C. and their Mw determined by size exclusion chromatography, is included in Tables B and C as well.
TABLE-US-00002 TABLE B Brimonidine tartrate formulation comparative example composition, dimension and degradation kinetic parameters Brimonidine Polymer Excipient, % w/w Implant Implant Implant k at 37 C. Tartrate, R R R RG RG Diameter Length Weight (1/day), in Formulation % w/w 202H 203S 208 752S 858S (.mu.m) (mm) (.mu.g) vitro CE 1 35 40 25 356 ~6 857 0.0041 CE2 35 65 356 ~6 857 0.0033 CE3 35 48 17 356 ~6 857 0.0073 CE4 35 15 40 10 356 ~6 857 0.0064
TABLE-US-00003 TABLE C Brimonidine free base example formulation composition, dimension and degradation kinetic parameter Brimonidine Polymer Excipient, % w/w Implant Implant Implant k at 37 C. free base, R RG RG RG RG Diameter Length Weight (1/day), in Formulation % w/w 202H 502H 502S 752S 858S (.mu.m) (mm) (.mu.g) vitro EX 1 50 50 356 ~6 800 0.02 EX 2 50 50 356 ~6 800 0.012 EX 3 50 25 25 356 ~6 800 0.012 EX 4 50 37.5 12.5 356 ~6 800 0.057
[0060] The polymer matrix degradation was then analyzed both in vitro and in vivo. For in vitro study, the polymer Mw degradation rate constant k as described above was used to calculate the degradation time for the polymer Mw degraded to 1000 Da t(1000) by assuming the degradation follows first order kinetics. For in vivo study, the polymer matrix degradation was determined by harvesting the implant samples that were injected to the vitreous of New Zealand rabbit. The results are summarized in Table D.
TABLE-US-00004 TABLE D Brimonidine formulation in vitro and in vivo drug release and polymer matrix degradation time In Vitro Rabbit Calc. Matrix Drug Drug Degradation Drug Matrix Substance Formulation Release t(1000) Release Degradation Brimo CE 1 6 months ~30 months >6 months >>6 months Tartrate CE 2 4 months ~28 months 5 months >>6 months CE 3 4 months ~15 months 4.5 months >>6 months CE 4 3 months ~14 months 3 months >6 month Brimo EX 1 3 months ~3 months ~2 months 2 months Free EX 2 4 months ~7 months ~3 months 4 months Base EX 3 3 months ~5 months ~3 months 3 months EX 4 1 month ~1 months ~1 month 1 month
In Vitro Testing of Intraocular Implants Containing Brimonidine and a Biodegradable Polymer Matrix
Weight Loss Study
[0061] For the implant weight loss study, each implant was first weighed, moved to a plastic micromesh cassette, and incubated in a glass jar filled with PBS (pH 7.4, 0.01M) before placed in a shaking water bath set at 37.degree. C. and 50 rpm. The implants were harvested at designated time points and dried under vacuum. The weights of the dried implants were recorded and the implant weight loss was calculated. The results are summarized in Table E and show that the brimonidine free base implants lose weight more quickly than those of brimonidine tartrate, implying and illustrating the difference in matrix degradation rate.
TABLE-US-00005 TABLE E Implant weight loss in PBS (pH 7.4, 0.01M) at 37.degree. C. Remaining Weight Time (wk) CE 1 CE 2 CE 3 CE 4 EX 1 EX 2 EX 3 EX 4 1 99.7% 99.7% 99.7% 99.5% 99.4% 99.5% 99.7% 99.3% 2 98.8% 99.4% 98.9% 91.7% 94.2% 100.7% 99.0% 0.0% 4 98.5% 95.5% 95.7% 78.7% 0.0% 95.0% 72.2% 6 97.9% 93.8% 93.0% 63.2% 81.0% 0.0% 8 98.8% 96.6% 89.3% 67.0% 0.0% 10 93.1% 85.7% 81.5% 57.3% 12 84.9% 74.3% 72.6% 61.9% 14 84.3% 40.4% 72.7% 67.0% 16 81.2% 66.9% 70.2% 51.5% 18 78.6% 71.9% 65.5% 53.9%
Implant Swelling
[0062] To investigate the implant swelling, each implant was incubated in 20 mL of PBS (pH 7.4, 0.01M) in a glass scintillation vial and placed in a shaking water bath set at 37.degree. C. and 50 rpm. The implant images were recorded and summarized in FIG. 12. The results show that brimonidine free base implants swelled and degraded much faster than those of brimonidine tartrate.
[0063] The drug releases of brimonidine tartrate formulations in rabbit and monkey eyes are shown in FIGS. 3 and 4, respectively. The drug releases of brimonidine free base formulations in rabbit and monkey eyes are shown in FIGS. 5 and 6.
[0064] The in vivo drug release profiles were determined by retrieving the implants from the vitreous humor at designated time points. The implant mass was recorded before and after in vivo implantation to determine the quantity of residual polymer matrix. The drug release rates in both animal models showed that Example 4 had the highest release rate, followed by Example 1, then Example 3, then Example 2 demonstrated the slowest drug release rate.
[0065] The drug concentration of brimonidine tartrate formulations in the retina (optic nerve) of Albino rabbit eyes are shown in FIG. 7. All formulations maintained the brimonidine concentration above the human .alpha.2A EC90 (88 nM, 25.7 ng/mL) for more than 3 months. For brimonidine free base formulations, the drug concentrations in retina (optic nerve in rabbit and macula in monkey) were determined, and the results are shown in FIGS. 8 and 9 for rabbit and monkey, respectively. The period for brimonidine concentration above the human .alpha.2A EC90 in the rabbit optic nerve was <3 months for all formulations. In a contrast, the time of brimonidine concentration above the human .alpha.2A EC90 in the monkey macula was >4 months for all formulations except Example 4 that lasted about one month.
[0066] The polymer matrix degradation of brimonidine tartrate and free base formulations in monkey eyes are shown in FIGS. 10 and 11, respectively. For brimonidine tartrate formulations, less than 50% of matrix was degraded for Comparative Example 1 and Comparative Example 2 formulations in one year, while that for Comparative Example 3 and Comparative Example 4 reached more than 90%. For brimonidine free base formulations, all formulations became small and hard to handle after one month, except Example 2, that the polymer matrix was expected to last for about six months. The in vitro matrix degradation observation matches the in vivo results.
[0067] The polymer matrix degradation of brimonidine tartrate and free base formulations in rabbit eyes were analyzed by photo images, and the matrix degradation time is longer than 6 months for brimonidine tartrate formulations and shorter than 4 months for brimonidine free base formulations.
[0068] The polymers used in the formulations include, but not limited to, poly(D,L-lactide) and poly(D,L-lactide-co-glycolide). They are summarized in Table A.
[0069] The four brimonidine free base formulations demonstrated implants with controlled drug release from one to four months and polymer matrixes lasting for less than two times the drug release duration. In contrast, the brimonidine tartrate formulations delivered the drug for a comparable duration as the brimonidine free base formulations, but the polymer matrix lasted more than two times of the drug release duration.
[0070] Although this invention has been disclosed in the context of certain preferred embodiments and examples, it will be understood by those skilled in the art that the present invention extends beyond the specifically disclosed embodiments to other alternative embodiments and/or uses of the invention and obvious modifications and equivalents thereof. In addition while the number of variations of the invention have been shown and described in detail, other modifications, which are within the scope of this invention, will be readily apparent to those of skill in the art based on this disclosure. It is also contemplated that various combinations or subcombinations of the specific features and aspects of the embodiments can be made and still fall within the scope of the invention. Accordingly, it should be understood that various features and aspects of the disclosed embodiments can be combined with, or substituted for, one another in order to perform varying modes of the disclosed invention. Thus, it is intended that the scope of the present invention herein disclosed should not be limited by the particular disclosed embodiments described above, but should be determined only by a fair reading of the claims.
* * * * *

D00001

D00002

D00003

D00004

D00005

D00006

D00007

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.