Assays For Detecting T Cell Immune Subsets And Methods Of Use Thereof
CHU; Felix ; et al.
U.S. patent application number 17/075520 was filed with the patent office on 2021-04-15 for assays for detecting t cell immune subsets and methods of use thereof. This patent application is currently assigned to Genentech, Inc.. The applicant listed for this patent is Genentech, Inc.. Invention is credited to Felix CHU, Oded FOREMAN, James ZIAI.
| Application Number | 20210109099 17/075520 |
| Document ID | / |
| Family ID | 1000005293311 |
| Filed Date | 2021-04-15 |







| United States Patent Application | 20210109099 |
| Kind Code | A1 |
| CHU; Felix ; et al. | April 15, 2021 |
ASSAYS FOR DETECTING T CELL IMMUNE SUBSETS AND METHODS OF USE THEREOF
Abstract
The present disclosure provides methods for measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample containing cancer cells and lymphocytes obtained from a subject by labeling lymphocytes that show CD4 expression in the sample, then labeling lymphocytes that show OX40 expression in the sample, then labeling lymphocytes that show Foxp3 expression in the sample, then measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample. Further provided are methods for determining the prognosis of a subject, predicting responsiveness of a subject having cancer to an OX40 agonist treatment, and methods for treating or delaying progression of cancer based on the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample.
| Inventors: | CHU; Felix; (San Francisco, CA) ; FOREMAN; Oded; (Davis, CA) ; ZIAI; James; (Emeryville, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Genentech, Inc. South San Francisco CA |
||||||||||
| Family ID: | 1000005293311 | ||||||||||
| Appl. No.: | 17/075520 | ||||||||||
| Filed: | October 20, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15849805 | Dec 21, 2017 | 10845364 | ||
| 17075520 | ||||
| 14930603 | Nov 2, 2015 | |||
| 15849805 | ||||
| 62074594 | Nov 3, 2014 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 2317/24 20130101; G01N 33/582 20130101; C12Q 1/06 20130101; C07K 16/2875 20130101; G01N 2800/7028 20130101; G01N 2333/70514 20130101; G01N 33/56972 20130101; G01N 2800/52 20130101; G01N 33/57419 20130101; A61K 38/177 20130101; C07K 2317/75 20130101 |
| International Class: | G01N 33/569 20060101 G01N033/569; G01N 33/574 20060101 G01N033/574; A61K 38/17 20060101 A61K038/17; C07K 16/28 20060101 C07K016/28; C12Q 1/06 20060101 C12Q001/06; G01N 33/58 20060101 G01N033/58 |
Claims
1-24. (canceled)
25. A method for measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from a subject, comprising the steps of: (a) labeling lymphocytes that show CD4 expression in the sample; (b) labeling lymphocytes that show OX40 expression in the sample after step (a); (c) labeling lymphocytes that show Foxp3 expression in the sample after step (b); and (d) measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample after step (c).
26-62. (canceled)
63. The method of claim 25, further comprising treating the sample with a solution comprising EDTA or citrate buffer between steps (a) and (b) and/or between steps (b) and (c).
64. The method of claim 25, wherein lymphocytes showing CD4 expression, OX40 expression, and Foxp3 expression are labeled by immunofluorescence staining.
65. The method of claim 64, wherein lymphocytes showing OX40 expression are labeled using an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7, 26 or 27.
66. The method of claim 64, wherein quantifying the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample comprises imaging said immunofluorescence staining.
67. The method of claim 25, wherein, in the sample comprising cancer cells and lymphocytes, said cancer cells are from a colorectal cancer.
68. The method of claim 25, wherein, in the sample comprising cancer cells and lymphocytes, said cancer cells are from a cancer selected from the group consisting of non-small cell lung cancer, renal cell carcinoma, bladder cancer, ovarian cancer, glioblastoma, neuroblastoma, melanoma, breast carcinoma, gastric cancer, and hepatocellular carcinoma.
69. The method of claim 68, wherein the breast carcinoma is triple-negative breast carcinoma.
70. The method of claim 25, wherein, in the sample comprising cancer cells and lymphocytes, said cancer cells are from a primary tumor.
71. The method of claim 25, wherein, in the sample comprising cancer cells and lymphocytes, said cancer cells are from a metastasis.
72. The method of claim 25, wherein the CD4+ OX40+ Foxp3+ lymphocytes are tumor-infiltrating lymphocytes.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the priority benefit of U.S. Provisional Application Ser. No. 62/074,594, filed on Nov. 3, 2014, which is incorporated herein by reference in its entirety.
SUBMISSION OF SEQUENCE LISTING ON ASCII TEXT FILE
[0002] The content of the following submission on ASCII text file is incorporated herein by reference in its entirety: a computer readable form (CRF) of the Sequence Listing (file name: 146392029000SEQLIST.TXT, date recorded: Nov. 2, 2015 size: 185 KB).
Field
[0003] The present disclosure relates to assays for detecting T cell immune subsets, as well as methods for determining prognosis, predicting responsiveness to treatment, and methods of treatment related thereto. cl Background
[0004] OX40 (also known as CD134, TNFRSF4 and ACT35) is a member of the tumor necrosis factor receptor superfamily. OX40 is not constitutively expressed on naive T cells, but is induced after engagement of the T cell receptor (TCR). The ligand for OX40, OX40L, is predominantly expressed on antigen presenting cells. OX40 is highly expressed by activated CD4+ T cells, activated CD8+ T cells, memory T cells, and regulatory T cells. OX40 signaling can provide costimulatory signals to CD4 and CD8 T cells, leading to enhanced cell proliferation, survival, effector function and migration. OX40 signaling also enhances memory T cell development and function.
[0005] Regulatory T cells (Treg) cells are highly enriched in tumors and tumor draining lymph nodes derived from multiple cancer indications, including melanoma, NSCLC, renal, ovarian, colon, pancreatic, hepatocellular, and breast cancer. In a subset of these indications, increased intratumoral T reg cell densities are associated with poor patient prognosis, suggesting that these cells play an important role in suppressing antitumor immunity. OX40 positive tumor infiltrating lymphocytes have been described.
[0006] It is clear that there continues to be a need for diagnostic, prognostic, and predictive methods to identify patients that are more likely to benefit from anti-tumor treatments that modulate OX40 activity. The invention described herein meets this need and provides other benefits.
[0007] All references cited herein, including patent applications, patent publications, and UniProtKB/Swiss-Prot Accession numbers are herein incorporated by reference in their entirety, as if each individual reference were specifically and individually indicated to be incorporated by reference.
BRIEF SUMMARY
[0008] The present disclosure describes assays for detecting T cell immune subsets, as well as methods of using these assays to determine prognosis, predict responsiveness, and/or treating or delay progress of cancer.
[0009] In certain aspects, the present disclosure provides a method for determining prognosis of a subject having cancer, comprising: (a) measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from the subject; and (b) determining the prognosis of the subject based on the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample, as compared with a reference, wherein an increased number of CD4+ OX40+ Foxp3+ lymphocytes in the sample indicates that the subject may have an improved prognosis. In some embodiments, the improved prognosis comprises increased overall survival. In some embodiments, the improved prognosis comprises increased progression-free survival.
[0010] In certain aspects, the present disclosure provides a method for treating or delaying progression of cancer in a subject, comprising: (a) measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from the subject; (b) determining the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample, as compared with a reference; and (c) if the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample is higher than the reference, administering to the subject an effective amount of an OX40 agonist.
[0011] In certain aspects, the present disclosure provides a method for treating or delaying progression of cancer in a subject, comprising administering to the subject an effective amount of an OX40 agonist, wherein a sample comprising cancer cells and lymphocytes obtained from the subject has an increased number of CD4+ OX40+ Foxp3+ lymphocytes, as compared with a reference. In some embodiments, the subject has been tested to determine if a sample comprising cancer cells and lymphocytes obtained from the subject has an increased number of CD4+ OX40+ Foxp3+ lymphocytes as compared with a reference before treatment or is selected for treatment based on a sample comprising cancer cells and lymphocytes obtained from the subject has an increased number of CD4+ OX40+ Foxp3+ lymphocytes as compared with a reference.
[0012] In certain aspects, the present disclosure provides a method for predicting responsiveness of a subject having cancer to an OX40 agonist treatment, comprising: (a) measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from the subject; and (b) classifying the subject as a responsive or non-responsive subject based on the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample, as compared with a reference, wherein an increased number of CD4+ OX40+ Foxp3+ lymphocytes in the sample indicates the subject may be responsive to the OX40 agonist treatment.
[0013] In some embodiments, the number of CD4+ OX40+ Foxp3+ lymphocytes in one or more regions of interest in the sample of a subject is measured. In some embodiments, the region of interest includes tumor cells and stroma. In some embodiments, the number of CD4+ OX40+ Foxp3+ lymphocytes is a median, mean or average number of CD4+ OX40+ Foxp3+ lymphocytes in different regions of interest of the sample from the subject. In some embodiments, the number of CD4+ OX40+ Foxp3+ lymphocytes is normalized to total cells in the region of interest of the sample. In some embodiments, the reference is based on the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from a cancer having the same type and/or stage as the cancer of the subject. In some embodiments, the reference is a median, mean, or average number of CD4+ OX40+ Foxp3+ lymphocytes in samples obtained from cancers having the same type and/or stage as the cancer of the subject. In some embodiments, the OX40 agonist is an agonist anti-human OX40 antibody. In some embodiments, the antibody is a monoclonal antibody. In some embodiments, the antibody is a humanized or human antibody. In some embodiments, the antibody comprises an IgG1 Fc region. In some embodiments, the antibody comprises an IgG4 Fc region. In some embodiments, the antibody comprises an Fc region comprising a mutation that decreases binding to an Fc receptor. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, 8 or 9; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, 10, 11, 12, 13, or 14; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4, 15 or 19; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7, 22, 23, 24, 25, 26, 27 or 28. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:26. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:27. In some embodiments, the antibody is MEDI6469 or MEDI0562. In some embodiments, the OX40 agonist comprises one or more extracellular domains of OX40L. In some embodiments, the OX40 agonist is MEDI6383. In some embodiments, measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample comprises: (a) labeling lymphocytes that show CD4 expression in the sample; (b) labeling lymphocytes that show OX40 expression in the sample after step (a); (c) labeling lymphocytes that show Foxp3 expression in the sample after step (b); and (d) measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample after step (c).
[0014] In certain aspects, the present disclosure provides a method for measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from a subject, comprising the steps of: (a) labeling lymphocytes that show CD4 expression in the sample; (b) labeling lymphocytes that show OX40 expression in the sample after step (a); (c) labeling lymphocytes that show Foxp3 expression in the sample after step (b); and (d) measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample after step (c). In some embodiments, the sample is treated with a solution comprising EDTA or citrate buffer between steps (a) and (b) and/or between steps (b) and (c). In some embodiments, lymphocytes showing CD4 expression, OX40 expression, and Foxp3 expression are labeled by immunofluorescence staining In some embodiments, lymphocytes showing OX40 expression are labeled using an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7, 26 or 27. In some embodiments, quantifying the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample comprises imaging said immunofluorescence staining.
[0015] In some embodiments, the cancer is colorectal cancer. In some embodiments, the cancer is selected from the group consisting of non-small cell lung cancer, renal cell carcinoma, bladder cancer, ovarian cancer, glioblastoma, neuroblastoma, melanoma, breast carcinoma, gastric cancer, and hepatocellular carcinoma. In some embodiments, the breast carcinoma is triple-negative breast carcinoma. In some embodiments, the sample comprising cancer cells and lymphocytes, wherein said cancer cells are from a primary tumor. In some embodiments, the sample comprising cancer cells and lymphocytes, wherein said cancer cells are from a metastasis. In some embodiments, the CD4+ OX40+ Foxp3+ lymphocytes are tumor-infiltrating lymphocytes.
[0016] In certain aspects, the present disclosure provides a method for determining prognosis of a subject having cancer, comprising: (a) measuring the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample comprising metastatic cancer cells and lymphocytes obtained from the subject; and (b) determining the prognosis of the subject based on the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample, as compared with a reference, wherein an increased number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample indicates that the subject may have an improved prognosis. In some embodiments, the improved prognosis comprises increased overall survival. In some embodiments, the improved prognosis comprises increased progression-free survival.
[0017] In certain aspects, the present disclosure provides a method for treating or delaying progression of cancer in a subject, comprising: (a) measuring the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample comprising metastatic cancer cells and lymphocytes obtained from the subject; (b) determining the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample, as compared with a reference; and (c) if the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample is higher than the reference, administering to the subject an effective amount of an OX40 agonist.
[0018] In certain aspects, the present disclosure provides a method for treating or delaying progression of cancer in a subject, comprising administering to the subject an effective amount of an OX40 agonist, wherein a sample comprising metastatic cancer cells and lymphocytes obtained from the subject has an increased number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes, as compared with a reference.
[0019] In certain aspects, the present disclosure provides a method for predicting responsiveness of a subject having cancer to an OX40 agonist treatment, comprising: (a) measuring the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample comprising metastatic cancer cells and lymphocytes obtained from the subject; and (b) classifying the subject as a responsive or non-responsive subject based on the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample, as compared with a reference, wherein an increased number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample indicates the subject may be responsive to the OX40 agonist treatment.
[0020] In some embodiments, the number of OX4+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in one or more regions of interest in the sample of a subject is measured. In some embodiments, the region of interest includes metastatic cancer cells and stroma. In some embodiments, the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes is a median, mean or average number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in different regions of interest in the sample from the subject. In some embodiments, the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes is normalized to total cells in the region of interest in the sample. In some embodiments, the reference is based on the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample comprising metastatic cancer cells and lymphocytes obtained from a cancer having the same type and/or stage as the cancer of the subject. In some embodiments, the reference is a median, mean, or average number of CD4+ OX40+ Foxp3+ lymphocytes in samples obtained from cancers having the same type and/or stage as the cancer of the subject. In some embodiments, the OX40 agonist is an agonist anti-human OX40 antibody. In some embodiments, the antibody is a monoclonal antibody. In some embodiments, the antibody is a humanized or human antibody. In some embodiments, the antibody comprises an IgG1 Fc region. In some embodiments, the antibody comprises an IgG4 Fc region. In some embodiments, the antibody comprises an Fc region comprising a mutation that decreases binding to an Fc receptor. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, 8 or 9; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, 10, 11, 12, 13, or 14; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4, 15 or 19; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7, 22, 23, 24, 25, 26, 27 or 28. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:26. In some embodiments, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:27. In some embodiments, the antibody is MEDI6469 or MEDI0562. In some embodiments, the OX40 agonist comprises one or more extracellular domains of OX40L. In some embodiments, the OX40 agonist is MEDI6383. In some embodiments, the cancer is colorectal cancer. In some embodiments, the cancer is selected from the group consisting of non-small cell lung cancer, renal cell carcinoma, bladder cancer, ovarian cancer, glioblastoma, neuroblastoma, melanoma, breast carcinoma, gastric cancer, and hepatocellular carcinoma. In some embodiments, the breast carcinoma is triple-negative breast carcinoma. In some embodiments, the OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes are tumor-infiltrating lymphocytes.
[0021] It is to be understood that one, some, or all of the properties of the various embodiments described above and herein may be combined to form other embodiments of the present invention. These and other aspects of the invention will become apparent to one of skill in the art. These and other embodiments of the invention are further described by the detailed description that follows.
BRIEF DESCRIPTION OF THE DRAWINGS
[0022] FIGS. 1A-1C show representative images of triple immunofluorescence staining for expression of CD4 (green), OX40 (white), and Foxp3 (red) on stage I colorectal cancer samples. Cell nuclei are counter-stained using DAPI (blue). Exemplary Treg and Teff cells are labeled.
[0023] FIGS. 2A-2C show representative images of triple immunofluorescence staining for expression of CD4 (green), OX40 (white), and Foxp3 (red) on stage II colorectal cancer samples. Cell nuclei are counter-stained using DAPI (blue).
[0024] FIGS. 3A-3C show representative images of triple immunofluorescence staining for expression of CD4 (green), OX40 (white), and Foxp3 (red) on stage III colorectal cancer samples. Cell nuclei are counter-stained using DAPI (blue).
[0025] FIGS. 4A-4C show representative images of triple immunofluorescence staining for expression of CD4 (green), OX40 (white), and Foxp3 (red) on stage IV colorectal cancer samples. Cell nuclei are counter-stained using DAPI (blue).
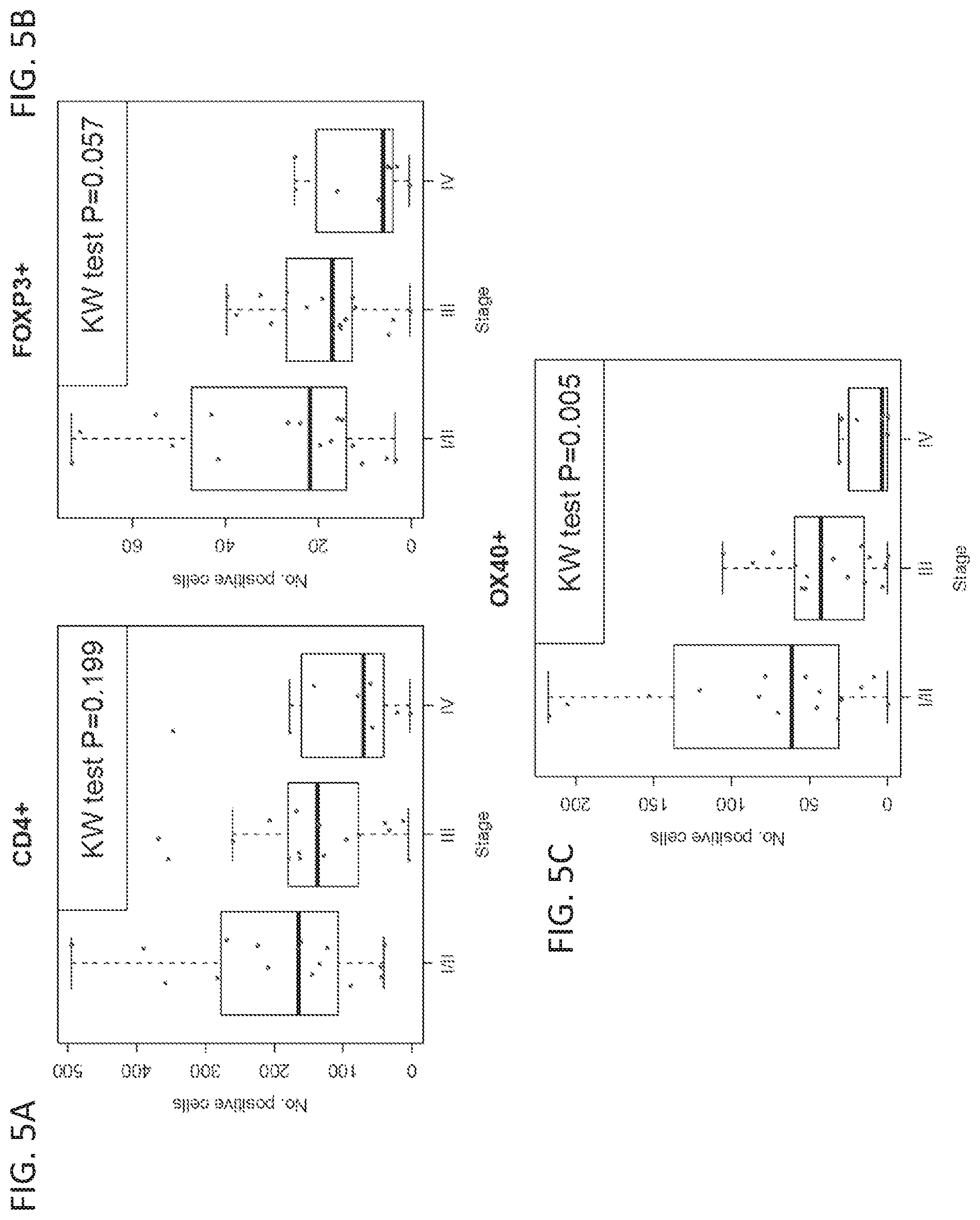
[0026] FIGS. 5A-5C show the correlation between the number of cells in a sample showing expression of specific marker(s) and cancer stage at diagnosis. Mean counts of cells that are positive for expression of specific markers are provided as follows: CD4+ (FIG. 5A), Foxp3+ (FIG. 5B), and OX40+ (FIG. 5C).
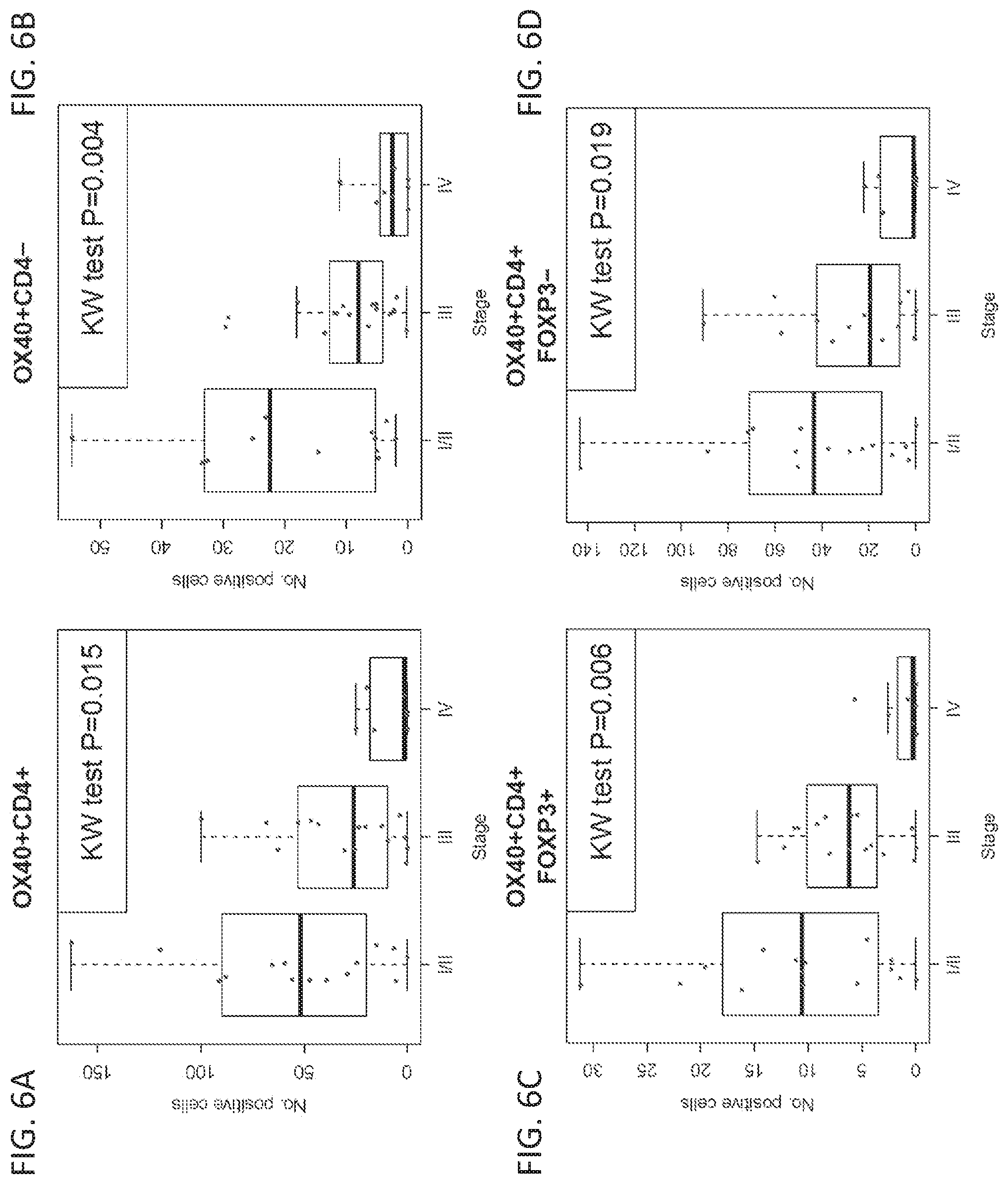
[0027] FIGS. 6A-6D show the correlation between the number of cells in a sample showing expression of specific marker(s) and cancer stage at diagnosis. Mean counts of cells that are positive for specific markers are provided as follows: OX40+ CD4+ (FIG. 6A), OX40+ CD4- (FIG. 6B), OX40+ CD4+ Foxp3+ (FIG. 6C), and OX40+ CD4+ Foxp3- (FIG. 6D).
[0028] FIGS. 7A-7C show the correlation between overall survival and the number of cells in a sample showing expression of specific marker(s). Overall survival is shown for patients whose samples contained greater than or less than the median count (as labeled) of the following cell types: CD4+ (FIG. 7A), Foxp3+ (FIG. 7B), and OX40+ (FIG. 7C). P values reflecting the difference between overall survival of patients whose samples showed cell counts above and below the median are indicated for each cell type.
[0029] FIGS. 8A-8D show the correlation between overall survival and the number of cells in a sample showing expression of specific marker(s). Overall survival is shown for patients whose samples contained greater than or less than the median count (as labeled) of the following cell types: OX40+ CD4+ (FIG. 8A), OX40+ CD4- (FIG. 8B), OX40+ CD4+ Foxp3+ (FIG. 8C), and OX40+ CD4+ Foxp3- (FIG. 8D). P values reflecting the difference between overall survival of patients whose samples showed cell counts above and below the median are indicated for each cell type.
[0030] FIGS. 9A-9C show the correlation between the number of cells showing expression of specific marker(s) in paired primary and metastatic samples (n=19). Mean counts of cells that are positive for expression of specific markers, normalized to total number of cells, are provided as follows: CD4+ (FIG. 9A), Foxp3+ (FIG. 9B), and OX40+ (FIG. 9C). Tissue types are as labeled: Brain (BN), Bone (BO-Rib), Lymph node (LN), Lung (LU), Liver (LV), Omentum (OM), Ovary (OV), and Soft tissue/retroperitoneum (SOT-Ret).
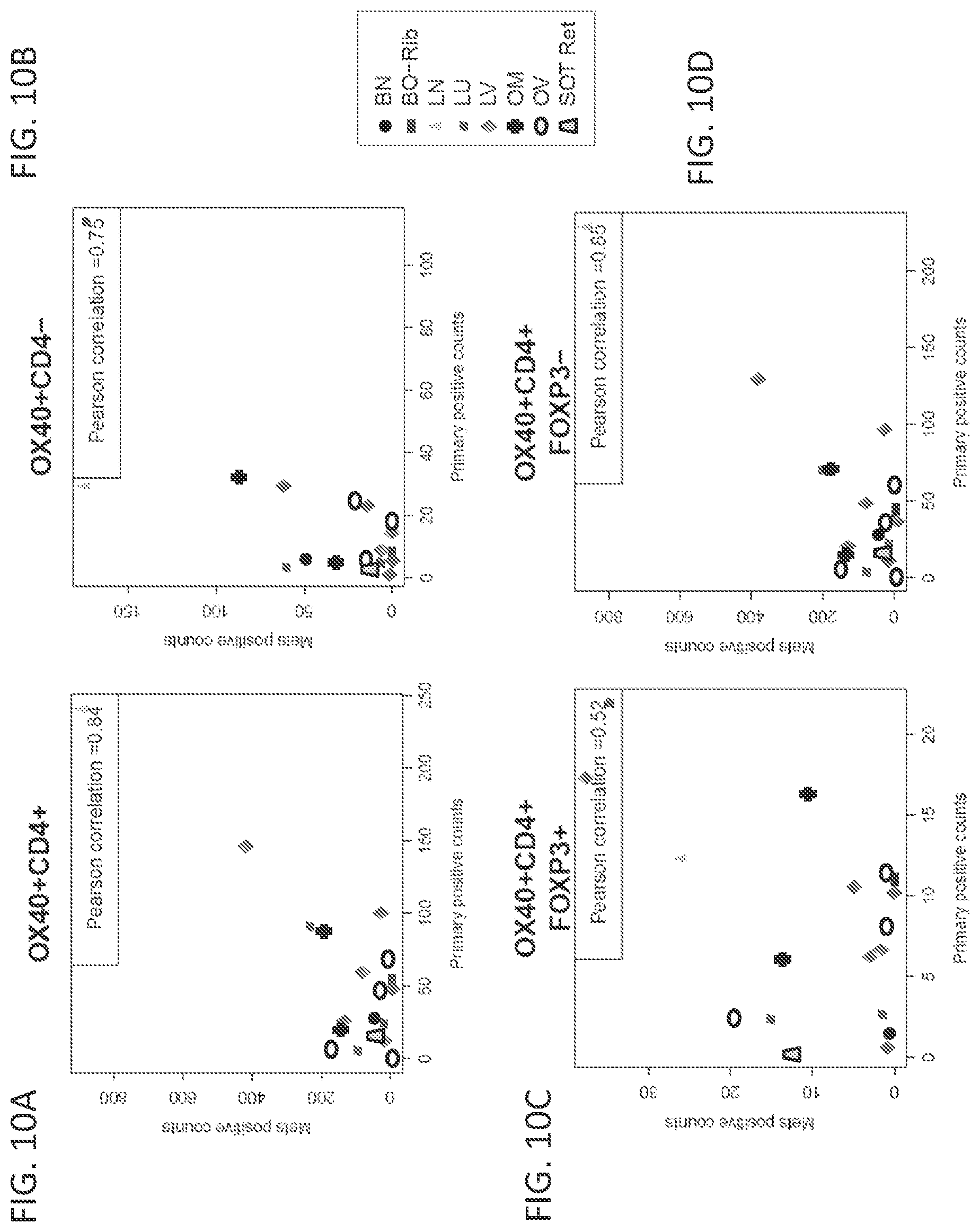
[0031] FIGS. 10A-10D show the correlation between the number of cells showing expression of specific marker(s) in paired primary and metastatic samples (n=19). Mean counts of cells that are positive for expression of specific markers, normalized to total number of cells, are provided as follows: OX40+ CD4+ (FIG. 10A), OX40+ CD4- (FIG. 10B), OX40+ CD4+ Foxp3+ (FIG. 10C), and OX40+ CD4+ Foxp3- (FIG. 10D). Tissue types are as labeled: Brain (BN), Bone (BO-Rib), Lymph node (LN), Lung (LU), Liver (LV), Omentum (OM), Ovary (OV), and Soft tissue/retroperitoneum (SOT-Ret).
[0032] FIGS. 11A-11 provide the results of multivariate Cox model analyses of CD4 expression (FIG. 11A), OX40 expression (FIG. 11B), Foxp3 expression (FIG. 11C), and the ratio of OX40+ Teff cells to OX40+ Treg cells (OX40 Teff/Treg) (FIG. 11D) adjusted for age, gender and stage (n=32/39).
DETAILED DESCRIPTION
I. General Techniques
[0033] The techniques and procedures described or referenced herein are generally well understood and commonly employed using conventional methodology by those skilled in the art, such as, for example, the widely utilized methodologies described in Sambrook et al., Molecular Cloning: A Laboratory Manual 3d edition (2001) Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.; Current Protocols in Molecular Biology (F. M. Ausubel, et al. eds., (2003)); the series Methods in Enzymology (Academic Press, Inc.): PCR 2: A Practical Approach (M. J. MacPherson, B.bD. Hames and G. R. Taylor eds. (1995)), Harlow and Lane, eds. (1988) Antibodies, A Laboratory Manual, and Animal Cell Culture (R. I. Freshney, ed. (1987)); Oligonucleotide Synthesis (M. J. Gait, ed., 1984); Methods in Molecular Biology, Humana Press; Cell Biology: A Laboratory Notebook (J. E. Cellis, ed., 1998) Academic Press; Animal Cell Culture (R. I. Freshney), ed., 1987); Introduction to Cell and Tissue Culture (J. P. Mather and P. E. Roberts, 1998) Plenum Press; Cell and Tissue Culture: Laboratory Procedures (A. Doyle, J. B. Griffiths, and D. G. Newell, eds., 1993-8) J. Wiley and Sons; Handbook of Experimental Immunology (D. M. Weir and C. C. Blackwell, eds.); Gene Transfer Vectors for Mammalian Cells (J. M. Miller and M. P. Calos, eds., 1987); PCR: The Polymerase Chain Reaction, (Mullis et al., eds., 1994); Current Protocols in Immunology (J. E. Coligan et al., eds., 1991); Short Protocols in Molecular Biology (Wiley and Sons, 1999); Immunobiology (C. A. Janeway and P. Travers, 1997); Antibodies (P. Finch, 1997); Antibodies: A Practical Approach (D. Catty., ed., IRL Press, 1988-1989); Monoclonal Antibodies: A Practical Approach (P. Shepherd and C. Dean, eds., Oxford University Press, 2000); Using Antibodies: A Laboratory Manual (E. Harlow and D. Lane (Cold Spring Harbor Laboratory Press, 1999); The Antibodies (M. Zanetti and J. D. Capra, eds., Harwood Academic Publishers, 1995); and Cancer: Principles and Practice of Oncology (V. T. DeVita et al., eds., J. B. Lippincott Company, 1993).
II. Definitions
[0034] Before describing the invention in detail, it is to be understood that this invention is not limited to particular compositions or biological systems, which can, of course, vary. It is also to be understood that the terminology used herein is for the purpose of describing particular embodiments only, and is not intended to be limiting.
[0035] As used herein and in the appended claims, the singular forms "a," "or," and "the" include plural referents unless the context clearly dictates otherwise.
[0036] Reference to "about" a value or parameter herein includes (and describes) variations that are directed to that value or parameter per se. For example, description referring to "about X" includes description of "X".
[0037] It is understood that aspects and variations of the invention described herein include "consisting" and/or "consisting essentially of" aspects and variations.
[0038] The term "dysfunction" in the context of immune dysfunction, refers to a state of reduced immune responsiveness to antigenic stimulation.
[0039] The term "dysfunctional", as used herein, also includes refractory or unresponsive to antigen recognition, specifically, impaired capacity to translate antigen recognition into downstream T-cell effector functions, such as proliferation, cytokine production (e.g., gamma interferon) and/or target cell killing.
[0040] "Enhancing T cell function" means to induce, cause or stimulate an effector or memory T cell to have a renewed, sustained or amplified biological function. Examples of enhancing T-cell function include: increased secretion of y-interferon from CD8+ effector T cells, increased secretion of y-interferon from CD4+ memory and/or effector T-cells, increased proliferation of CD4+ effector and/or memory T cells, increased proliferation of CD8+ effector T-cells, increased antigen responsiveness (e.g., clearance), relative to such levels before the intervention. In one embodiment, the level of enhancement is at least 50%, alternatively 60%, 70%, 80%, 90%, 100%, 120%, 150%, 200%. The manner of measuring this enhancement is known to one of ordinary skill in the art.
[0041] "Tumor immunity" refers to the process in which tumors evade immune recognition and clearance. Thus, as a therapeutic concept, tumor immunity is "treated" when such evasion is attenuated, and the tumors are recognized and attacked by the immune system. Examples of tumor recognition include tumor binding, tumor shrinkage and tumor clearance.
[0042] "Immunogenicity" refers to the ability of a particular substance to provoke an immune response. Tumors are immunogenic and enhancing tumor immunogenicity aids in the clearance of the tumor cells by the immune response.
[0043] An "acceptor human framework" for the purposes herein is a framework comprising the amino acid sequence of a light chain variable domain (VL) framework or a heavy chain variable domain (VH) framework derived from a human immunoglobulin framework or a human consensus framework, as defined below. An acceptor human framework "derived from" a human immunoglobulin framework or a human consensus framework may comprise the same amino acid sequence thereof, or it may contain amino acid sequence changes. In some embodiments, the number of amino acid changes are 10 or less, 9 or less, 8 or less, 7 or less, 6 or less, 5 or less, 4 or less, 3 or less, or 2 or less. In some embodiments, the VL acceptor human framework is identical in sequence to the VL human immunoglobulin framework sequence or human consensus framework sequence.
[0044] "Affinity" refers to the strength of the sum total of noncovalent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen). Unless indicated otherwise, as used herein, "binding affinity" refers to intrinsic binding affinity which reflects a 1:1 interaction between members of a binding pair (e.g., antibody and antigen). The affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (Kd). Affinity can be measured by common methods known in the art, including those described herein. Specific illustrative and exemplary embodiments for measuring binding affinity are described in the following.
[0045] An "agonist antibody," as used herein, is an antibody which activates a biological activity of the antigen it binds.
[0046] An "anti-angiogenic agent" refers to a compound which blocks, or interferes with to some degree, the development of blood vessels. An anti-angiogenic agent may, for instance, be a small molecule or antibody that binds to a growth factor or growth factor receptor involved in promoting angiogenesis. In one embodiment, an anti-angiogenic agent is an antibody that binds to vascular endothelial growth factor (VEGF), such as bevacizumab (AVASTIN).
[0047] "Antibody-dependent cell-mediated cytotoxicity" or "ADCC" refers to a form of cytotoxicity in which secreted immunoglobulin bound onto Fc receptors (FcRs) present on certain cytotoxic cells (e.g. NK cells, neutrophils, and macrophages) enable these cytotoxic effector cells to bind specifically to an antigen-bearing target cell and subsequently kill the target cell with cytotoxins. The primary cells for mediating ADCC, NK cells, express Fc.gamma.RIII only, whereas monocytes express Fc.gamma.RI, Fc.gamma.RII, and Fc.gamma.RIII. FcR expression on hematopoietic cells is summarized in Table 3 on page 464 of Ravetch and Kinet, Annu. Rev. Immunol 9:457-92 (1991). To assess ADCC activity of a molecule of interest, an in vitro ADCC assay, such as that described in U.S. Pat. Nos. 5,500,362 or 5,821,337 or 6,737,056 (Presta), may be performed. Useful effector cells for such assays include PBMC and NK cells. Alternatively, or additionally, ADCC activity of the molecule of interest may be assessed in vivo, e.g., in an animal model such as that disclosed in Clynes et al. PNAS (USA) 95:652-656 (1998). An exemplary assay for assessing ADCC activity is provided in the examples herein.
[0048] The terms "anti-OX40 antibody" and "an antibody that binds to OX40" refer to an antibody that is capable of binding OX40 with sufficient affinity such that the antibody is useful as a diagnostic and/or therapeutic agent in targeting OX40. In one embodiment, the extent of binding of an anti-OX40 antibody to an unrelated, non-OX40 protein is less than about 10% of the binding of the antibody to OX40 as measured, e.g., by a radioimmunoassay (RIA). In certain embodiments, an antibody that binds to OX40 has a dissociation constant (Kd) of .ltoreq.1 .mu.M, .ltoreq.100 nM, .ltoreq.10 nM, .ltoreq.1 nM, .ltoreq.0.1 nM, .ltoreq.0.01 nM, or .ltoreq.0.001 nM (e.g. 10.sup.-8M or less, e.g. from 10.sup.-8M to 10.sup.-13M, e.g., from 10.sup.-9 M to 10.sup.-13 M). In certain embodiments, an anti-OX40 antibody binds to an epitope of OX40 that is conserved among OX40 from different species.
[0049] As use herein, the term "binds", "specifically binds to" or is "specific for" refers to measurable and reproducible interactions such as binding between a target and an antibody, which is determinative of the presence of the target in the presence of a heterogeneous population of molecules including biological molecules. For example, an antibody that binds to or specifically binds to a target (which can be an epitope) is an antibody that binds this target with greater affinity, avidity, more readily, and/or with greater duration than it binds to other targets. In one embodiment, the extent of binding of an antibody to an unrelated target is less than about 10% of the binding of the antibody to the target as measured, e.g., by a radioimmunoassay (RIA). In certain embodiments, an antibody that specifically binds to a target has a dissociation constant (Kd) of .ltoreq.1.mu.M, .ltoreq.100 nM, .ltoreq.10 nM, .ltoreq.1 nM, or .ltoreq.0.1 nM. In certain embodiments, an antibody specifically binds to an epitope on a protein that is conserved among the protein from different species. In another embodiment, specific binding can include, but does not require exclusive binding.
[0050] The term "antibody" herein is used in the broadest sense and encompasses various antibody structures, including but not limited to monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g., bispecific antibodies), and antibody fragments so long as they exhibit the desired antigen-binding activity.
[0051] An "antibody fragment" refers to a molecule other than an intact antibody that comprises a portion of an intact antibody that binds the antigen to which the intact antibody binds. Examples of antibody fragments include but are not limited to Fv, Fab, Fab', Fab'-SH, F(ab').sub.2; diabodies; linear antibodies; single-chain antibody molecules (e.g. scFv); and multispecific antibodies formed from antibody fragments.
[0052] An "antibody that binds to the same epitope" as a reference antibody refers to an antibody that blocks binding of the reference antibody to its antigen in a competition assay by 50% or more, and conversely, the reference antibody blocks binding of the antibody to its antigen in a competition assay by 50% or more. An exemplary competition assay is provided herein.
[0053] The term "binding domain" refers to the region of a polypeptide that binds to another molecule. In the case of an FcR, the binding domain can comprise a portion of a polypeptide chain thereof (e.g. the alpha chain thereof) which is responsible for binding an Fc region. One useful binding domain is the extracellular domain of an FcR alpha chain.
[0054] A polypeptide with a variant IgG Fc with "altered" FcR, ADCC or phagocytosis activity is one which has either enhanced or diminished FcR binding activity (e.g, Fc.gamma.R) and/or ADCC activity and/or phagocytosis activity compared to a parent polypeptide or to a polypeptide comprising a native sequence Fc region.
[0055] The term "OX40," as used herein, refers to any native OX40 from any vertebrate source, including mammals such as primates (e.g. humans) and rodents (e.g., mice and rats), unless otherwise indicated. The term encompasses "full-length," unprocessed OX40 as well as any form of OX40 that results from processing in the cell. The term also encompasses naturally occurring variants of OX40, e.g., splice variants or allelic variants. The amino acid sequence of an exemplary human OX40 is shown in SEQ ID NO:1.
[0056] "OX40 activation" refers to activation, of the OX40 receptor. Generally, OX40 activation results in signal transduction.
[0057] The terms "cancer" and "cancerous" refer to or describe the physiological condition in mammals that is typically characterized by unregulated cell growth. Examples of cancer include but are not limited to, carcinoma, lymphoma, blastoma, sarcoma, and leukemia or lymphoid malignancies. More particular examples of such cancers include, but not limited to, squamous cell cancer (e.g., epithelial squamous cell cancer), lung cancer including small-cell lung cancer, non-small cell lung cancer, adenocarcinoma of the lung and squamous carcinoma of the lung, cancer of the peritoneum, hepatocellular cancer, gastric or stomach cancer including gastrointestinal cancer and gastrointestinal stromal cancer, pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, cancer of the urinary tract, hepatoma, breast cancer, colon cancer, rectal cancer, colorectal cancer, endometrial or uterine carcinoma, salivary gland carcinoma, kidney or renal cancer, prostate cancer, vulval cancer, thyroid cancer, hepatic carcinoma, anal carcinoma, penile carcinoma, melanoma, superficial spreading melanoma, lentigo maligna melanoma, acral lentiginous melanomas, nodular melanomas, multiple myeloma and B-cell lymphoma; chronic lymphocytic leukemia (CLL); acute lymphoblastic leukemia (ALL); hairy cell leukemia; chronic myeloblastic leukemia; and post-transplant lymphoproliferative disorder (PTLD), as well as abnormal vascular proliferation associated with phakomatoses, edema (such as that associated with brain tumors), Meigs' syndrome, brain, as well as head and neck cancer, and associated metastases. In certain embodiments, cancers that are amenable to treatment by the antibodies of the invention include breast cancer, colorectal cancer, rectal cancer, non-small cell lung cancer, glioblastoma, non-Hodgkins lymphoma (NHL), renal cell cancer, prostate cancer, liver cancer, pancreatic cancer, soft-tissue sarcoma, kaposi's sarcoma, carcinoid carcinoma, head and neck cancer, ovarian cancer, mesothelioma, and multiple myeloma. In some embodiments, the cancer is selected from: non-small cell lung cancer, glioblastoma, neuroblastoma, melanoma, breast carcinoma (e.g. triple-negative breast cancer), gastric cancer, colorectal cancer (CRC), and hepatocellular carcinoma. Yet, in some embodiments, the cancer is selected from: non-small cell lung cancer, colorectal cancer, glioblastoma and breast carcinoma (e.g. triple-negative breast cancer), including metastatic forms of those cancers.
[0058] The term "tumor" refers to all neoplastic cell growth and proliferation, whether malignant or benign, and all pre-cancerous and cancerous cells and tissues. The terms "cancer," "cancerous," "cell proliferative disorder," "proliferative disorder" and "tumor" are not mutually exclusive as referred to herein.
[0059] The terms "cell proliferative disorder" and "proliferative disorder" refer to disorders that are associated with some degree of abnormal cell proliferation. In one embodiment, the cell proliferative disorder is cancer.
[0060] The term "chimeric" antibody refers to an antibody in which a portion of the heavy and/or light chain is derived from a particular source or species, while the remainder of the heavy and/or light chain is derived from a different source or species.
[0061] The "class" of an antibody refers to the type of constant domain or constant region possessed by its heavy chain. There are five major classes of antibodies: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into subclasses (isotypes), e.g., IgG.sub.1, IgG.sub.2, IgG.sub.3, IgG.sub.4, IgA.sub.1, and IgA.sub.2. The heavy chain constant domains that correspond to the different classes of immunoglobulins are called .alpha., .delta., .epsilon., .gamma., and .mu., respectively.
[0062] "Complement dependent cytotoxicity" or "CDC" refers to the lysis of a target cell in the presence of complement. Activation of the classical complement pathway is initiated by the binding of the first component of the complement system (C1q) to antibodies (of the appropriate subclass), which are bound to their cognate antigen. To assess complement activation, a CDC assay, e.g., as described in Gazzano-Santoro et al., J. Immunol. Methods 202:163 (1996), may be performed. Polypeptide variants with altered Fc region amino acid sequences (polypeptides with a variant Fc region) and increased or decreased C1q binding capability are described, e.g., in U.S. Pat. No. 6,194,551 B1 and WO 1999/51642. See also, e.g., Idusogie et al. J. Immunol. 164: 4178-4184 (2000).
[0063] The term "cytostatic agent" refers to a compound or composition which arrests growth of a cell either in vitro or in vivo. Thus, a cytostatic agent may be one which significantly reduces the percentage of cells in S phase. Further examples of cytostatic agents include agents that block cell cycle progression by inducing GO/G1 arrest or M-phase arrest. The humanized anti-Her2 antibody trastuzumab (HERCEPTIN.RTM.) is an example of a cytostatic agent that induces G0/G1 arrest. Classical M-phase blockers include the vincas (vincristine and vinblastine), taxanes, and topoisomerase II inhibitors such as doxorubicin, epirubicin, daunorubicin, etoposide, and bleomycin. Certain agents that arrest G1 also spill over into S-phase arrest, for example, DNA alkylating agents such as tamoxifen, prednisone, dacarbazine, mechlorethamine, cisplatin, methotrexate, 5-fluorouracil, and ara-C. Further information can be found in Mendelsohn and Israel, eds., The Molecular Basis of Cancer, Chapter 1, entitled "Cell cycle regulation, oncogenes, and antineoplastic drugs" by Murakami et al. (W. B. Saunders, Philadelphia, 1995), e.g., p. 13. The taxanes (paclitaxel and docetaxel) are anticancer drugs both derived from the yew tree. Docetaxel (TAXOTERE.RTM., Rhone-Poulenc Rorer), derived from the European yew, is a semisynthetic analogue of paclitaxel (TAXOL.RTM., Bristol-Myers Squibb). Paclitaxel and docetaxel promote the assembly of microtubules from tubulin dimers and stabilize microtubules by preventing depolymerization, which results in the inhibition of mitosis in cells.
[0064] The term "cytotoxic agent" as used herein refers to a substance that inhibits or prevents a cellular function and/or causes cell death or destruction. Cytotoxic agents include, but are not limited to, radioactive isotopes (e.g., At.sup.211, I.sup.131I.sup.125, Y.sup.90, Re.sup.186, Re.sup.188, Sm.sup.153, Bi.sup.212, P.sup.32, Pb.sup.212 and radioactive isotopes of Lu); chemotherapeutic agents or drugs (e.g., methotrexate, adriamicin, vinca alkaloids (vincristine, vinblastine, etoposide), doxorubicin, melphalan, mitomycin C, chlorambucil, daunorubicin or other intercalating agents); growth inhibitory agents; enzymes and fragments thereof such as nucleolytic enzymes; antibiotics; toxins such as small molecule toxins or enzymatically active toxins of bacterial, fungal, plant or animal origin, including fragments and/or variants thereof; and the various antitumor or anticancer agents disclosed below.
[0065] A "depleting anti-OX40 antibody," is an anti-OX40 antibody that kills or depletes OX40-expressing cells. Depletion of OX40 expressing cells can be achieved by various mechanisms, such as antibody-dependent cell-mediated cytotoxicity and/or phagocytosis. Depletion of OX40-expressing cells may be assayed in vitro, and exemplary methods for in vitro ADCC and phagocytosis assays are provided herein. In some embodiments, the OX40-expressing cell is a human CD4+ effector T cell. In some embodiments, the OX40-expressing cell is a transgenic BT474 cell that expresses human OX40.
[0066] "Effector functions" refer to those biological activities attributable to the Fc region of an antibody, which vary with the antibody isotype. Examples of antibody effector functions include: C1q binding and complement dependent cytotoxicity (CDC); Fc receptor binding; antibody-dependent cell-mediated cytotoxicity (ADCC); phagocytosis; down regulation of cell surface receptors (e.g. B cell receptor); and B cell activation.
[0067] An "effective amount" of an agent, e.g., a pharmaceutical formulation, refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
[0068] "Fc receptor" or "FcR" describes a receptor that binds to the Fc region of an antibody. In some embodiments, an FcR is a native human FcR. In some embodiments, an FcR is one which binds an IgG antibody (a gamma receptor) and includes receptors of the Fc.gamma.RI, Fc.gamma.RII, and Fc.gamma.RIII subclasses, including allelic variants and alternatively spliced forms of those receptors. Fc.gamma.RII receptors include Fc.gamma.RIIA (an "activating receptor") and Fc.gamma.RIIB (an "inhibiting receptor"), which have similar amino acid sequences that differ primarily in the cytoplasmic domains thereof. Activating receptor Fc.gamma.RIIA contains an immunoreceptor tyrosine-based activation motif (ITAM) in its cytoplasmic domain. Inhibiting receptor Fc.gamma.RIIB contains an immunoreceptor tyrosine-based inhibition motif (ITIM) in its cytoplasmic domain. (see, e.g., Daeron, Annu. Rev. Immunol. 15:203-234 (1997)). FcRs are reviewed, for example, in Ravetch and Kinet, Annu. Rev. Immunol 9:457-92 (1991); Capel et al., Immunomethods 4:25-34 (1994); and de Haas et al., J. Lab. Clin. Med. 126:330-41 (1995). Other FcRs, including those to be identified in the future, are encompassed by the term "FcR" herein. The term "Fc receptor" or "FcR" also includes the neonatal receptor, FcRn, which is responsible for the transfer of maternal IgGs to the fetus (Guyer et al., J. Immunol. 117:587 (1976) and Kim et al., J. Immunol. 24:249 (1994)) and regulation of homeostasis of immunoglobulins. Methods of measuring binding to FcRn are known (see, e.g., Ghetie and Ward., Immunol. Today 18(12):592-598 (1997); Ghetie et al., Nature Biotechnology, 15(7):637-640 (1997); Hinton et al., J. Biol. Chem. 279(8):6213-6216 (2004); WO 2004/92219 (Hinton et al.). Binding to human FcRn in vivo and serum half life of human FcRn high affinity binding polypeptides can be assayed, e.g., in transgenic mice or transfected human cell lines expressing human FcRn, or in primates to which the polypeptides with a variant Fc region are administered. WO 2000/42072 (Presta) describes antibody variants with improved or diminished binding to FcRs. See also, e.g., Shields et al. J. Biol. Chem. 9(2):6591-6604 (2001).
[0069] The term "Fc region" herein is used to define a C-terminal region of an immunoglobulin heavy chain that contains at least a portion of the constant region. The term includes native sequence Fc regions and variant Fc regions. In one embodiment, a human IgG heavy chain Fc region extends from Cys226, or from Pro230, to the carboxyl-terminus of the heavy chain. However, the C-terminal lysine (Lys447) of the Fc region may or may not be present. Unless otherwise specified herein, numbering of amino acid residues in the Fc region or constant region is according to the EU numbering system, also called the EU index, as described in Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md., 1991.
[0070] A "functional Fc region" possesses an "effector function" of a native sequence Fc region. Exemplary "effector functions" include C1q binding; CDC; Fc receptor binding; ADCC; phagocytosis; down regulation of cell surface receptors (e.g. B cell receptor; BCR), etc. Such effector functions generally require the Fc region to be combined with a binding domain (e.g., an antibody variable domain) and can be assessed using various assays as disclosed, for example, in definitions herein.
[0071] "Human effector cells" refer to leukocytes that express one or more FcRs and perform effector functions. In certain embodiments, the cells express at least Fc.gamma.RIII and perform ADCC effector function(s). Examples of human leukocytes which mediate ADCC include peripheral blood mononuclear cells (PBMC), natural killer (NK) cells, monocytes, cytotoxic T cells, and neutrophils. The effector cells may be isolated from a native source, e.g., from blood.
[0072] "Framework" or "FR" refers to variable domain residues other than hypervariable region (HVR) residues. The FR of a variable domain generally consists of four FR domains: FR1, FR2, FR3, and FR4. Accordingly, the HVR and FR sequences generally appear in the following sequence in VH (or VL): FR1-H1(L1)-FR2-H2(L2)-FR3-H3(L3)-FR4.
[0073] The terms "full length antibody," "intact antibody," and "whole antibody" are used herein interchangeably to refer to an antibody having a structure substantially similar to a native antibody structure or having heavy chains that contain an Fc region as defined herein.
[0074] The terms "host cell," "host cell line," and "host cell culture" are used interchangeably and refer to cells into which exogenous nucleic acid has been introduced, including the progeny of such cells. Host cells include "transformants" and "transformed cells," which include the primary transformed cell and progeny derived therefrom without regard to the number of passages. Progeny may not be completely identical in nucleic acid content to a parent cell, but may contain mutations. Mutant progeny that have the same function or biological activity as screened or selected for in the originally transformed cell are included herein.
[0075] A "human antibody" is one which possesses an amino acid sequence which corresponds to that of an antibody produced by a human or a human cell or derived from a non-human source that utilizes human antibody repertoires or other human antibody-encoding sequences. This definition of a human antibody specifically excludes a humanized antibody comprising non-human antigen-binding residues.
[0076] A "human consensus framework" is a framework which represents the most commonly occurring amino acid residues in a selection of human immunoglobulin VL or VH framework sequences. Generally, the selection of human immunoglobulin VL or VH sequences is from a subgroup of variable domain sequences. Generally, the subgroup of sequences is a subgroup as in Kabat et al., Sequences of Proteins of Immunological Interest, Fifth Edition, NIH Publication 91-3242, Bethesda Md. (1991), vols. 1-3. In one embodiment, for the VL, the subgroup is subgroup kappa I as in Kabat et al., supra. In one embodiment, for the VH, the subgroup is subgroup III as in Kabat et al., supra.
[0077] A "humanized" antibody refers to a chimeric antibody comprising amino acid residues from non-human HVRs and amino acid residues from human FRs. In certain embodiments, a humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the HVRs (e.g., CDRs) correspond to those of a non-human antibody, and all or substantially all of the FRs correspond to those of a human antibody. A humanized antibody optionally may comprise at least a portion of an antibody constant region derived from a human antibody. A "humanized form" of an antibody, e.g., a non-human antibody, refers to an antibody that has undergone humanization.
[0078] The term "hypervariable region" or "HVR" as used herein refers to each of the regions of an antibody variable domain which are hypervariable in sequence ("complementarity determining regions" or "CDRs") and/or form structurally defined loops ("hypervariable loops") and/or contain the antigen-contacting residues ("antigen contacts"). Generally, antibodies comprise six HVRs: three in the VH (H1, H2, H3), and three in the VL (L1, L2, L3). Exemplary HVRs herein include:
[0079] (a) hypervariable loops occurring at amino acid residues 26-32 (L1), 50-52 (L2), 91-96 (L3), 26-32 (H1), 53-55 (H2), and 96-101 (H3) (Chothia and Lesk, J. Mol. Biol. 196:901-917 (1987));
[0080] (b) CDRs occurring at amino acid residues 24-34 (L1), 50-56 (L2), 89-97 (L3), 31-35b (H1), 50-65 (H2), and 95-102 (H3) (Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991));
[0081] (c) antigen contacts occurring at amino acid residues 27c-36 (L1), 46-55 (L2), 89-96 (L3), 30-35b (H1), 47-58 (H2), and 93-101 (H3) (MacCallum et al. J. Mol. Biol. 262: 732-745 (1996)); and
[0082] (d) combinations of (a), (b), and/or (c), including HVR amino acid residues 46-56 (L2), 47-56 (L2), 48-56 (L2), 49-56 (L2), 26-35 (H1), 26-35b (H1), 49-65 (H2), 93-102 (H3), and 94-102 (H3).
[0083] Unless otherwise indicated, HVR residues and other residues in the variable domain (e.g., FR residues) are numbered herein according to Kabat et al., supra.
[0084] An "immunoconjugate" is an antibody conjugated to one or more heterologous molecule(s), including but not limited to a cytotoxic agent.
[0085] An "individual" or "subject" is a mammal. Mammals include, but are not limited to, domesticated animals (e.g., cows, sheep, cats, dogs, and horses), primates (e.g., humans and non-human primates such as monkeys), rabbits, and rodents (e.g., mice and rats). In certain embodiments, the individual or subject is a human.
[0086] "Promoting cell growth or proliferation" means increasing a cell's growth or proliferation by at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%.
[0087] An "isolated" antibody is one which has been separated from a component of its natural environment. In some embodiments, an antibody is purified to greater than 95% or 99% purity as determined by, for example, electrophoretic (e.g., SDS-PAGE, isoelectric focusing (IEF), capillary electrophoresis) or chromatographic (e.g., ion exchange or reverse phase HPLC). For review of methods for assessment of antibody purity, see, e.g., Flatman et al., J. Chromatogr. B 848:79-87 (2007).
[0088] An "isolated" nucleic acid refers to a nucleic acid molecule that has been separated from a component of its natural environment. An isolated nucleic acid includes a nucleic acid molecule contained in cells that ordinarily contain the nucleic acid molecule, but the nucleic acid molecule is present extrachromosomally or at a chromosomal location that is different from its natural chromosomal location.
[0089] "Isolated nucleic acid encoding an anti-OX40 antibody" refers to one or more nucleic acid molecules encoding antibody heavy and light chains (or fragments thereof), including such nucleic acid molecule(s) in a single vector or separate vectors, and such nucleic acid molecule(s) present at one or more locations in a host cell.
[0090] The term "monoclonal antibody" as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical and/or bind the same epitope, except for possible variant antibodies, e.g., containing naturally occurring mutations or arising during production of a monoclonal antibody preparation, such variants generally being present in minor amounts. In contrast to polyclonal antibody preparations, which typically include different antibodies directed against different determinants (epitopes), each monoclonal antibody of a monoclonal antibody preparation is directed against a single determinant on an antigen. Thus, the modifier "monoclonal" indicates the character of the antibody as being obtained from a substantially homogeneous population of antibodies, and is not to be construed as requiring production of the antibody by any particular method. For example, the monoclonal antibodies to be used in accordance with the present invention may be made by a variety of techniques, including but not limited to the hybridoma method, recombinant DNA methods, phage-display methods, and methods utilizing transgenic animals containing all or part of the human immunoglobulin loci, such methods and other exemplary methods for making monoclonal antibodies being described herein.
[0091] A "naked antibody" refers to an antibody that is not conjugated to a heterologous moiety (e.g., a cytotoxic moiety) or radiolabel. The naked antibody may be present in a pharmaceutical formulation.
[0092] "Native antibodies" refer to naturally occurring immunoglobulin molecules with varying structures. For example, native IgG antibodies are heterotetrameric glycoproteins of about 150,000 daltons, composed of two identical light chains and two identical heavy chains that are disulfide-bonded. From N- to C-terminus, each heavy chain has a variable region (VH), also called a variable heavy domain or a heavy chain variable domain, followed by three constant domains (CH1, CH2, and CH3). Similarly, from N- to C-terminus, each light chain has a variable region (VL), also called a variable light domain or a light chain variable domain, followed by a constant light (CL) domain. The light chain of an antibody may be assigned to one of two types, called kappa (.kappa.) and lambda (.lamda.), based on the amino acid sequence of its constant domain. A "native sequence Fc region" comprises an amino acid sequence identical to the amino acid sequence of an Fc region found in nature. Native sequence human Fc regions include a native sequence human IgG1 Fc region (non-A and A allotypes); native sequence human IgG2 Fc region; native sequence human IgG3 Fc region; and native sequence human IgG4 Fc region as well as naturally occurring variants thereof.
[0093] The term "package insert" is used to refer to instructions customarily included in commercial packages of therapeutic products, that contain information about the indications, usage, dosage, administration, combination therapy, contraindications and/or warnings concerning the use of such therapeutic products.
[0094] "Percent (%) amino acid sequence identity" with respect to a reference polypeptide sequence is defined as the percentage of amino acid residues in a candidate sequence that are identical with the amino acid residues in the reference polypeptide sequence, after aligning the sequences and introducing gaps, if necessary, to achieve the maximum percent sequence identity, and not considering any conservative substitutions as part of the sequence identity. Alignment for purposes of determining percent amino acid sequence identity can be achieved in various ways that are within the skill in the art, for instance, using publicly available computer software such as BLAST, BLAST-2, ALIGN or Megalign (DNASTAR) software. Those skilled in the art can determine appropriate parameters for aligning sequences, including any algorithms needed to achieve maximal alignment over the full length of the sequences being compared. For purposes herein, however, % amino acid sequence identity values are generated using the sequence comparison computer program ALIGN-2. The ALIGN-2 sequence comparison computer program was authored by Genentech, Inc., and the source code has been filed with user documentation in the U.S. Copyright Office, Washington D.C., 20559, where it is registered under U.S. Copyright Registration No. TXU510087. The ALIGN-2 program is publicly available from Genentech, Inc., South San Francisco, Calif., or may be compiled from the source code. The ALIGN-2 program should be compiled for use on a UNIX operating system, including digital UNIX V4.0D. All sequence comparison parameters are set by the ALIGN-2 program and do not vary.
[0095] In situations where ALIGN-2 is employed for amino acid sequence comparisons, the % amino acid sequence identity of a given amino acid sequence A to, with, or against a given amino acid sequence B (which can alternatively be phrased as a given amino acid sequence A that has or comprises a certain % amino acid sequence identity to, with, or against a given amino acid sequence B) is calculated as follows:
100 times the fraction X/Y
where X is the number of amino acid residues scored as identical matches by the sequence alignment program ALIGN-2 in that program's alignment of A and B, and where Y is the total number of amino acid residues in B. It will be appreciated that where the length of amino acid sequence A is not equal to the length of amino acid sequence B, the % amino acid sequence identity of A to B will not equal the % amino acid sequence identity of B to A. Unless specifically stated otherwise, all % amino acid sequence identity values used herein are obtained as described in the immediately preceding paragraph using the ALIGN-2 computer program.
[0096] The term "pharmaceutical formulation" refers to a preparation which is in such form as to permit the biological activity of an active ingredient contained therein to be effective, and which contains no additional components which are unacceptably toxic to a subject to which the formulation would be administered.
[0097] A "pharmaceutically acceptable carrier" refers to an ingredient in a pharmaceutical formulation, other than an active ingredient, which is nontoxic to a subject. A pharmaceutically acceptable carrier includes, but is not limited to, a buffer, excipient, stabilizer, or preservative.
[0098] As used herein, "treatment" (and grammatical variations thereof such as "treat" or "treating") refers to clinical intervention in an attempt to alter the natural course of the individual being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis. In some embodiments, antibodies of the invention are used to delay development of a disease or to slow the progression of a disease.
[0099] The term "tumor" refers to all neoplastic cell growth and proliferation, whether malignant or benign, and all pre-cancerous and cancerous cells and tissues. The terms "cancer," "cancerous," "cell proliferative disorder," "proliferative disorder" and "tumor" are not mutually exclusive as referred to herein.
[0100] The term "variable region" or "variable domain" refers to the domain of an antibody heavy or light chain that is involved in binding the antibody to antigen. The variable domains of the heavy chain and light chain (VH and VL, respectively) of a native antibody generally have similar structures, with each domain comprising four conserved framework regions (FRs) and three hypervariable regions (HVRs). (See, e.g., Kindt et al. Kuby Immunology, 6.sup.th ed., W.H. Freeman and Co., page 91 (2007).) A single VH or VL domain may be sufficient to confer antigen-binding specificity. Furthermore, antibodies that bind a particular antigen may be isolated using a VH or VL domain from an antibody that binds the antigen to screen a library of complementary VL or VH domains, respectively. See, e.g., Portolano et al., J. Immunol. 150:880-887 (1993); Clarkson et al., Nature 352:624-628 (1991).
[0101] A "variant Fc region" comprises an amino acid sequence which differs from that of a native sequence Fc region by virtue of at least one amino acid modification, preferably one or more amino acid substitution(s). Preferably, the variant Fc region has at least one amino acid substitution compared to a native sequence Fc region or to the Fc region of a parent polypeptide, e.g. from about one to about ten amino acid substitutions, and preferably from about one to about five amino acid substitutions in a native sequence Fc region or in the Fc region of the parent polypeptide. The variant Fc region herein will preferably possess at least about 80% homology with a native sequence Fc region and/or with an Fc region of a parent polypeptide, and most preferably at least about 90% homology therewith, more preferably at least about 95% homology therewith.
[0102] The term "vector," as used herein, refers to a nucleic acid molecule capable of propagating another nucleic acid to which it is linked. The term includes the vector as a self-replicating nucleic acid structure as well as the vector incorporated into the genome of a host cell into which it has been introduced. Certain vectors are capable of directing the expression of nucleic acids to which they are operatively linked. Such vectors are referred to herein as "expression vectors."
[0103] A "VH subgroup III consensus framework" comprises the consensus sequence obtained from the amino acid sequences in variable heavy subgroup III of Kabat et al. In one embodiment, the VH subgroup III consensus framework amino acid sequence comprises at least a portion or all of each of the following sequences:
TABLE-US-00001 (SEQ ID NO: 214) EVQLVESGGGLVQPGGSLRLSCAAS (SEQ ID NO: 215) H1-WVRQAPGKGLEWV (SEQ ID NO: 216) H2-RFTISRDNSKNTLYLQMNSLRAEDTAVYYC (SEQ ID NO: 217) H3-WGQGTLVTVSS.
[0104] A "VL subgroup I consensus framework" comprises the consensus sequence obtained from the amino acid sequences in variable light kappa subgroup I of Kabat et al. In one embodiment, the VH subgroup I consensus framework amino acid sequence comprises at least a portion or all of each of the following sequences:
TABLE-US-00002 (SEQ ID NO: 218) DIQMTQSPSSLSASVGDRVTITC (SEQ ID NO: 219) L1-WYQQKPGKAPKLLIY (SEQ ID NO: 220) L2-GVPSRFSGSGSGTDFTLTISSLQPEDFATYYC (SEQ ID NO: 221) L3-FGQGTKVEIK.
[0105] The term "cytotoxic agent" as used herein refers to a substance that inhibits or prevents a cellular function and/or causes cell death or destruction. Cytotoxic agents include, but are not limited to, radioactive isotopes (e.g., At211, I131, I125, Y90, Re186, Re188, Sm153, Bi212, P32, Pb212 and radioactive isotopes of Lu); chemotherapeutic agents; growth inhibitory agents; enzymes and fragments thereof such as nucleolytic enzymes; and toxins such as small molecule toxins or enzymatically active toxins of bacterial, fungal, plant or animal origin, including fragments and/or variants thereof. Exemplary cytotoxic agents can be selected from anti-microtubule agents, platinum coordination complexes, alkylating agents, antibiotic agents, topoisomerase II inhibitors, antimetabolites, topoisomerase I inhibitors, hormones and hormonal analogues, signal transduction pathway inhibitors, non-receptor tyrosine kinase angiogenesis inhibitors, immunotherapeutic agents, proapoptotic agents, inhibitors of LDH-A; inhibitors of fatty acid biosynthesis; cell cycle signalling inhibitors; HDAC inhibitors, proteasome inhibitors; and inhibitors of cancer metabolism.
[0106] In one embodiment the cytotoxic agent is selected from anti-microtubule agents, platinum coordination complexes, alkylating agents, antibiotic agents, topoisomerase II inhibitors, antimetabolites, topoisomerase I inhibitors, hormones and hormonal analogues, signal transduction pathway inhibitors, non-receptor tyrosine kinase angiogenesis inhibitors, immunotherapeutic agents, proapoptotic agents, inhibitors of LDH-A, inhibitors of fatty acid biosynthesis, cell cycle signalling inhibitors, HDAC inhibitors, proteasome inhibitors, and inhibitors of cancer metabolism. In one embodiment the cytotoxic agent is a taxane. In one embodiment the taxane is paclitaxel or docetaxel. In one embodiment the cytotoxic agent is a platinum agent. In one embodiment the cytotoxic agent is an antagonist of EGFR. In one embodiment the antagonist of EGFR is N-(3-ethynylphenyl)-6,7-bis(2-methoxyethoxy)quinazolin-4-amine (e.g., erlotinib). In one embodiment the cytotoxic agent is a RAF inhibitor. In one embodiment, the RAF inhibitor is a BRAF and/or CRAF inhibitor. In one embodiment the RAF inhibitor is vemurafenib. In one embodiment the cytotoxic agent is a PI3K inhibitor.
[0107] "Chemotherapeutic agent" includes chemical compounds useful in the treatment of cancer. Examples of chemotherapeutic agents include erlotinib (TARCEVA.RTM., Genentech/OSI Pharm.), bortezomib (VELCADE.RTM., Millennium Pharm.), disulfiram, epigallocatechin gallate, salinosporamide A, carfilzomib, 17-AAG (geldanamycin), radicicol, lactate dehydrogenase A (LDH-A), fulvestrant (FASLODEX.RTM., Astra7eneca), sunitib (SUTENT.RTM., Pfizer/Sugen), letrozole (FEMARA.RTM., Novartis), imatinib mesylate (GLEEVEC.RTM., Novartis), finasunate (VATALANIB.RTM., Novartis), oxaliplatin (ELOXATIN.RTM., Sanofi), 5-FU (5-fluorouracil), leucovorin, Rapamycin (Sirolimus, RAPAMUNE.RTM., Wyeth), Lapatinib (TYKERB.RTM., GSK572016, Glaxo Smith Kline), Lonafamib (SCH 66336), sorafenib (NEXAVAR.RTM., Bayer Labs), gefitinib (IRESSA.RTM., AstraZeneca), AG1478, alkylating agents such as thiotepa and CYTOXAN.RTM. cyclosphosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; ethylenimines and methylamelamines including altretamine, triethylenemelamine, triethylenephosphoramide, triethylenethiophosphoramide and trimethylomelamine; acetogenins (especially bullatacin and bullatacinone); a camptothecin (including topotecan and irinotecan); bryostatin; callystatin; CC-1065 (including its adozelesin, carzelesin and bizelesin synthetic analogs); cryptophycins (particularly cryptophycin 1 and cryptophycin 8); adrenocorticosteroids (including prednisone and prednisolone); cyproterone acetate; 5.alpha.-reductases including finasteride and dutasteride); vorinostat, romidepsin, panobinostat, valproic acid, mocetinostat dolastatin; aldesleukin, talc duocarmycin (including the synthetic analogs, KW-2189 and CB1-TM1); eleutherobin; pancratistatin; a sarcodictyin; spongistatin; nitrogen mustards such as chlorambucil, chlomaphazine, chlorophosphamide, estramustine, ifosfamide, mechlorethamine, mechlorethamine oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, trofosfamide, uracil mustard; nitrosoureas such as carmustine, chlorozotocin, fotemustine, lomustine, nimustine, and ranimnustine; antibiotics such as the enediyne antibiotics (e.g., calicheamicin, especially calicheamicin .gamma.1I and calicheamicin .omega.1I (Angew Chem. Intl. Ed. Engl. 1994 33:183-186); dynemicin, including dynemicin A; bisphosphonates, such as clodronate; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antibiotic chromophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, caminomycin, carzinophilin, chromomycinis, dactinomycin, daunorubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, ADRIAMYCIN.RTM. (doxorubicin), morpholino-doxorubicin, cyanomorpholino-doxorubicin, 2-pyrrolino-doxorubicin and deoxydoxorubicin), epirubicin, esorubicin, idarubicin, marcellomycin, mitomycins such as mitomycin C, mycophenolic acid, nogalamycin, olivomycins, peplomycin, porfiromycin, puromycin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zorubicin; anti-metabolites such as methotrexate and 5-fluorouracil (5-FU); folic acid analogs such as denopterin, methotrexate, pteropterin, trimetrexate; purine analogs such as fludarabine, 6-mercaptopurine, thiamiprine, thioguanine; pyrimidine analogs such as ancitabine, azacitidine, 6-azauridine, carmofur, cytarabine, dideoxyuridine, doxifluridine, enocitabine, floxuridine; androgens such as calusterone, dromostanolone propionate, epitiostanol, mepitiostane, testolactone; anti-adrenals such as aminoglutethimide, mitotane, trilostane; folic acid replenisher such as frolinic acid; aceglatone; aldophosphamide glycoside; aminolevulinic acid; eniluracil; amsacrine; bestrabucil; bisantrene; edatraxate; defofamine; demecolcine; diaziquone; elfomithine; elliptinium acetate; an epothilone; etoglucid; gallium nitrate; hydroxyurea; lentinan; lonidainine; maytansinoids such as maytansine and ansamitocins; mitoguazone; mitoxantrone; mopidamnol; nitraerine; pentostatin; phenamet; pirarubicin; losoxantrone; podophyllinic acid; 2-ethylhydrazide; procarbazine; PSK.RTM. polysaccharide complex (JHS Natural Products, Eugene, Oreg.); razoxane; rhizoxin; sizofuran; spirogermanium; tenuazonic acid; triaziquone; 2,2',2''-trichlorotriethylamine; trichothecenes (especially T-2 toxin, verracurin A, roridin A and anguidine); urethan; vindesine; dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside ("Ara-C"); cyclophosphamide; thiotepa; taxoids, e.g., TAXOL (paclitaxel; Bristol-Myers Squibb Oncology, Princeton, N.J.), ABRAXANE.RTM. (Cremophor-free), albumin-engineered nanoparticle formulations of paclitaxel (American Pharmaceutical Partners, Schaumberg, Ill.), and TAXOTERE.RTM. (docetaxel, doxetaxel; Sanofi-Aventis); chloranmbucil; GEMZAR.RTM. (gemcitabine); 6-thioguanine; mercaptopurine; methotrexate; platinum analogs such as cisplatin and carboplatin; vinblastine; etoposide (VP-16); ifosfamide; mitoxantrone; vincristine; NAVELBINE.RTM. (vinorelbine); novantrone; teniposide; edatrexate; daunomycin; aminopterin; capecitabine (XELODA.RTM.); ibandronate; CPT-11; topoisomerase inhibitor RFS 2000; difluoromethylornithine (DMFO); retinoids such as retinoic acid; and pharmaceutically acceptable salts, acids and derivatives of any of the above.
[0108] Chemotherapeutic agent also includes (i) anti-hormonal agents that act to regulate or inhibit hormone action on tumors such as anti-estrogens and selective estrogen receptor modulators (SERMs), including, for example, tamoxifen (including NOLVADEX.RTM.; tamoxifen citrate), raloxifene, droloxifene, iodoxyfene, 4-hydroxytamoxifen, trioxifene, keoxifene, LY117018, onapristone, and FARESTON.RTM. (toremifine citrate); (ii) aromatase inhibitors that inhibit the enzyme aromatase, which regulates estrogen production in the adrenal glands, such as, for example, 4(5)-imidazoles, aminoglutethimide, MEGASE.RTM. (megestrol acetate), AROMASIN.RTM. (exemestane; Pfizer), formestanie, fadrozole, RIVISOR.RTM. (vorozole), FEMARA.RTM. (letrozole; Novartis), and ARIMIDEX.RTM. (anastrozole; AstraZeneca); (iii) anti-androgens such as flutamide, nilutamide, bicalutamide, leuprolide and goserelin; buserelin, tripterelin, medroxyprogesterone acetate, diethylstilbestrol, premarin, fluoxymesterone, all transretionic acid, fenretinide, as well as troxacitabine (a 1,3-dioxolane nucleoside cytosine analog); (iv) protein kinase inhibitors; (v) lipid kinase inhibitors; (vi) antisense oligonucleotides, particularly those which inhibit expression of genes in signaling pathways implicated in aberrant cell proliferation, such as, for example, PKC-alpha, Ralf and H-Ras; (vii) ribozymes such as VEGF expression inhibitors (e.g., ANGIOZYME.RTM.) and HER2 expression inhibitors; (viii) vaccines such as gene therapy vaccines, for example, ALLOVECTIN.RTM., LEUVECTIN.RTM., and VAXID.RTM.; PROLEUKIN.RTM., rIL-2; a topoisomerase 1 inhibitor such as LURTOTECAN.RTM.; ABARELIX.RTM. rmRH; and (ix) pharmaceutically acceptable salts, acids and derivatives of any of the above.
[0109] Chemotherapeutic agent also includes antibodies such as alemtuzumab (Campath), bevacizumab (AVAS TIN.RTM., Genentech); cetuximab (ERBITUX.RTM., Imclone); panitumumab (VECTIBIX.RTM., Amgen), rituximab (RITUXAN.RTM., Genentech/Biogen Idec), pertuzumab (OMNITARG.RTM., 2C4, Genentech), trastuzumab (HERCEPTIN.RTM., Genentech), tositumomab (Bexxar, Corixia), and the antibody drug conjugate, gemtuzumab ozogamicin (MYLOTARG.RTM., Wyeth). Additional humanized monoclonal antibodies with therapeutic potential as agents in combination with the compounds of the invention include: apolizumab, aselizumab, atlizumab, bapineuzumab, bivatuzumab mertansine, cantuzumab mertansine, cedelizumab, certolizumab pegol, cidfusituzumab, cidtuzumab, daclizumab, eculizumab, efalizumab, epratuzumab, erlizumab, felvizumab, fontolizumab, gemtuzumab ozogamicin, inotuzumab ozogamicin, ipilimumab, labetuzumab, lintuzumab, matuzumab, mepolizumab, motavizumab, motovizumab, natalizumab, nimotuzumab, nolovizumab, numavizumab, ocrelizumab, omalizumab, palivizumab, pascolizumab, pecfusituzumab, pectuzumab, pexelizumab, ralivizumab, ranibizumab, reslivizumab, reslizumab, resyvizumab, rovelizumab, ruplizumab, sibrotuzumab, siplizumab, sontuzumab, tacatuzumab tetraxetan, tadocizumab, talizumab, tefibazumab, tocilizumab, toralizumab, tucotuzumab celmoleukin, tucusituzumab, umavizumab, urtoxazumab, ustekinumab, visilizumab, and the anti-interleukin-12 (ABT-874/J695, Wyeth Research and Abbott Laboratories) which is a recombinant exclusively human-sequence, full-length IgG1 2 antibody genetically modified to recognize interleukin-12 p40 protein.
[0110] Chemotherapeutic agent also includes "EGFR inhibitors," which refers to compounds that bind to or otherwise interact directly with EGFR and prevent or reduce its signaling activity, and is alternatively referred to as an "EGFR antagonist." Examples of such agents include antibodies and small molecules that bind to EGFR. Examples of antibodies which bind to EGFR include MAb 579 (ATCC CRL HB 8506), MAb 455 (ATCC CRL HB8507), MAb 225 (ATCC CRL 8508), MAb 528 (ATCC CRL 8509) (see, U.S. Pat. No. 4,943, 533, Mendelsohn et al.) and variants thereof, such as chimerized 225 (C225 or Cetuximab; ERBUTIX.RTM.) and reshaped human 225 (H225) (see, WO 96/40210, Imclone Systems Inc.); IMC-11F8, a fully human, EGFR-targeted antibody (Imclone); antibodies that bind type II mutant EGFR (U.S. Pat. No. 5,212,290); humanized and chimeric antibodies that bind EGFR as described in U.S. Pat. No. 5,891,996; and human antibodies that bind EGFR, such as ABX-EGF or Panitumumab (see WO98/50433, Abgenix/Amgen); EMD 55900 (Stragliotto et al. Eur. J. Cancer 32A:636-640 (1996)); EMD7200 (matuzumab) a humanized EGFR antibody directed against EGFR that competes with both EGF and TGF-alpha for EGFR binding (EMD/Merck); human EGFR antibody, HuMax-EGFR (GenMab); fully human antibodies known as E1.1, E2.4, E2.5, E6.2, E6.4, E2.11, E6. 3 and E7.6. 3 and described in U.S. Pat. No. 6,235,883; MDX-447 (Medarex Inc); and mAb 806 or humanized mAb 806 (Johns et al., J. Biol. Chem. 279(29):30375-30384 (2004)). The anti-EGFR antibody may be conjugated with a cytotoxic agent, thus generating an immunoconjugate (see, e.g., EP659,439A2, Merck Patent GmbH). EGFR antagonists include small molecules such as compounds described in U.S. Pat. Nos: 5,616,582, 5,457,105, 5,475,001, 5,654,307, 5,679,683, 6,084,095, 6,265,410, 6,455,534, 6,521,620, 6,596,726, 6,713,484, 5,770,599, 6,140,332, 5,866,572, 6,399,602, 6,344,459, 6,602,863, 6,391,874, 6,344,455, 5,760,041, 6,002,008, and 5,747,498, as well as the following PCT publications: WO98/14451, WO98/50038, WO99/09016, and WO99/24037. Particular small molecule EGFR antagonists include OSI-774 (CP-358774, erlotinib, TARCEVA.RTM. Genentech/OSI Pharmaceuticals); PD 183805 (CI 1033, 2-propenamide, N-[4-[(3-chloro-4-fluorophenyl)amino]-7-[3-(4-morpholinyl)propoxy]-6-quin- azolinyl]-, dihydrochloride, Pfizer Inc.); ZD1839, gefitinib (IRESSA.RTM.) 4-(3'-Chloro-4'-fluoroanilino)-7-methoxy-6-(3-morpholinopropoxy)quinazoli- ne, Astra7eneca); ZM 105180 ((6-aino-4-(3-methylphenyl-amino)-quinazoline, Zeneca); BIBX-1382 (N8-(3-chloro-4-fluoro-phenyl)-N2-(1-methyl-piperidin-4-yl)-pyrimido[5,4-- d]pyrimidine-2,8-diamine, Boehringer Ingelheim); PKI-166 ((R)-4-[4-[(1-phenylethyl)amino]-1H-pyrrolo[2,3-d]pyrimidin-6-yl]-phenol)- ; (R)-6-(4-hydroxyphenyl)-4-[(1-phenylethyl)amino]-7H-pyrrolo[2,3-d]pyrimi- dine); CL-387785 (N-[4-[(3-bromophenyl)amino]-6-quinazolinyl]-2-butynamide); EKB-569 (N-[4-[(3-chloro-4-fluorophenyl)amino]-3-cyano-7-ethoxy-6-quinolinyl]-4-(- dimethylamino)-2-butenamide) (Wyeth); AG1478 (Pfizer); AG1571 (SU 5271; Pfizer); dual EGFR/HER2 tyrosine kinase inhibitors such as lapatinib (TYKERB.RTM., GSK572016 or N-[3-chloro-4-[(3 fluorophenyl)methoxy]phenyl]-6[5[[[2methylsulfonyl)ethyl]amino]methyl]-2-- furanyl]-4-quinazolinamine).
[0111] Chemotherapeutic agents also include "tyrosine kinase inhibitors" including the EGFR-targeted drugs noted in the preceding paragraph; small molecule HER2 tyrosine kinase inhibitor such as TAK165 available from Takeda; CP-724,714, an oral selective inhibitor of the ErbB2 receptor tyrosine kinase (Pfizer and OSI); dual-HER inhibitors such as EKB-569 (available from Wyeth) which preferentially binds EGFR but inhibits both HER2 and EGFR-overexpressing cells; lapatinib (GSK572016; available from Glaxo-SmithKline), an oral HER2 and EGFR tyrosine kinase inhibitor; PKI-166 (available from Novartis); pan-HER inhibitors such as canertinib (CI-1033; Pharmacia); Raf-1 inhibitors such as antisense agent ISIS-5132 available from ISIS Pharmaceuticals which inhibit Raf-1 signaling; non-HER targeted TK inhibitors such as imatinib mesylate (GLEEVEC.RTM., available from Glaxo SmithKline); multi-targeted tyrosine kinase inhibitors such as sunitinib (SUTENT.RTM., available from Pfizer); VEGF receptor tyrosine kinase inhibitors such as vatalanib (PTK787/ZK222584, available from Novartis/Schering AG); MAPK extracellular regulated kinase I inhibitor CI-1040 (available from Pharmacia); quinazolines, such as PD 153035,4-(3-chloroanilino) quinazoline; pyridopyrimidines; pyrimidopyrimidines; pyrrolopyrimidines, such as CGP 59326, CGP 60261 and CGP 62706; pyrazolopyrimidines, 4-(phenylamine)-7H-pyrrolo[2,3-d] pyrimidines; curcumin (diferuloyl methane, 4,5-bis (4-fluoroanilino)phthalimide); tyrphostines containing nitrothiophene moieties; PD-0183805 (Warner-Lamber); antisense molecules (e.g. those that bind to HER-encoding nucleic acid); quinoxalines (U.S. Pat. No. 5,804,396); tryphostins (U.S. Pat. No. 5,804,396); ZD6474 (Astra Zeneca); PTK-787 (Novartis/Schering AG); pan-HER inhibitors such as CI-1033 (Pfizer); Affinitac (ISIS 3521; Isis/Lilly); imatinib mesylate (GLEEVEC.RTM.); PKI 166 (Novartis); GW2016 (Glaxo SmithKline); CI-1033 (Pfizer); EKB-569 (Wyeth); Semaxinib (Pfizer); ZD6474 (AstraZeneca); PTK-787 (Novartis/Schering AG); INC-1C11 (Imclone), rapamycin (sirolimus, RAPAMUNE.RTM.); or as described in any of the following patent publications: U.S. Pat. No. 5,804,396; WO 1999/09016 (American Cyanamid); WO 1998/43960 (American Cyanamid); WO 1997/38983 (Warner Lambert); WO 1999/06378 (Warner Lambert); WO 1999/06396 (Warner Lambert); WO 1996/30347 (Pfizer, Inc); WO 1996/33978 (Zeneca); WO 1996/3397 (Zeneca) and WO 1996/33980 (Zeneca).
[0112] Chemotherapeutic agents also include dexamethasone, interferons, colchicine, metoprine, cyclosporine, amphotericin, metronidazole, alemtuzumab, alitretinoin, allopurinol, amifostine, arsenic trioxide, asparaginase, BCG live, bevacuzimab, bexarotene, cladribine, clofarabine, darbepoetin alfa, denileukin, dexrazoxane, epoetin alfa, elotinib, filgrastim, histrelin acetate, ibritumomab, interferon alfa-2a, interferon alfa-2b, lenalidomide, levamisole, mesna, methoxsalen, nandrolone, nelarabine, nofetumomab, oprelvekin, palifermin, pamidronate, pegademase, pegaspargase, pegfilgrastim, pemetrexed disodium, plicamycin, porfimer sodium, quinacrine, rasburicase, sargramostim, temozolomide, VM-26, 6-TG, toremifene, tretinoin, ATRA, valrubicin, zoledronate, and zoledronic acid, and pharmaceutically acceptable salts thereof.
[0113] Chemotherapeutic agents also include hydrocortisone, hydrocortisone acetate, cortisone acetate, tixocortol pivalate, triamcinolone acetonide, triamcinolone alcohol, mometasone, amcinonide, budesonide, desonide, fluocinonide, fluocinolone acetonide, betamethasone, betamethasone sodium phosphate, dexamethasone, dexamethasone sodium phosphate, fluocortolone, hydrocortisone-17-butyrate, hydrocortisone-17-valerate, aclometasone dipropionate, betamethasone valerate, betamethasone dipropionate, prednicarbate, clobetasone-17-butyrate, clobetasol-17-propionate, fluocortolone caproate, fluocortolone pivalate and fluprednidene acetate; immune selective anti-inflammatory peptides (ImSAIDs) such as phenylalanine-glutamine-glycine (FEG) and its D-isomeric form (feG) (IMULAN BioTherapeutics, LLC); anti-rheumatic drugs such as azathioprine, ciclosporin (cyclosporine A), D-penicillamine, gold salts, hydroxychloroquine, leflunomideminocycline, sulfasalazine, tumor necrosis factor alpha (TNFa) blockers such as etanercept (Enbrel), infliximab (Remicade), adalimumab (Humira), certolizumab pegol (Cimzia), golimumab (Simponi), Interleukin 1 (IL-1) blockers such as anakinra (Kineret), T cell costimulation blockers such as abatacept (Orencia), Interleukin 6 (IL-6) blockers such as tocilizumab (ACTEMERA.RTM.); Interleukin 13 (IL-13) blockers such as lebrikizumab; Interferon alpha (IFN) blockers such as Rontalizumab; Beta 7 integrin blockers such as rhuMAb Beta7; IgE pathway blockers such as Anti-M1 prime; Secreted homotrimeric LTa3 and membrane bound heterotrimer LTa1/.beta.2 blockers such as Anti-lymphotoxin alpha (LTa); radioactive isotopes (e.g., At211, I131, I125, Y90, Re186, Re188, Sm153, Bi212, P32, Pb212 and radioactive isotopes of Lu); miscellaneous investigational agents such as thioplatin, PS-341, phenylbutyrate, ET-18-OCH3, or farnesyl transferase inhibitors (L-739749, L-744832); polyphenols such as quercetin, resveratrol, piceatannol, epigallocatechine gallate, theaflavins, flavanols, procyanidins, betulinic acid and derivatives thereof; autophagy inhibitors such as chloroquine; delta-9-tetrahydrocannabinol (dronabinol, MARINOL.RTM.); beta-lapachone; lapachol; colchicines; betulinic acid; acetylcamptothecin, scopolectin, and 9-aminocamptothecin); podophyllotoxin; tegafur (UFTORAL.RTM.); bexarotene (TARGRETIN.RTM.); bisphosphonates such as clodronate (for example, BONEFOS.RTM. or OSTAC.RTM.), etidronate (DIDROCAL.RTM.), NE-58095, zoledronic acid/zoledronate (ZOMETA.RTM.), alendronate (FOSAMAX.RTM.), pamidronate (AREDIA.RTM.), tiludronate (SKELID.RTM.), or risedronate (ACTONEL.RTM.); and epidermal growth factor receptor (EGF-R); vaccines such as THERATOPE.RTM. vaccine; perifosine, COX-2 inhibitor (e.g. celecoxib or etoricoxib), proteosome inhibitor (e.g. PS341); CCI-779; tipifarnib (R11577); orafenib, ABT510; Bcl-2 inhibitor such as oblimersen sodium (GENASENSE.RTM.); pixantrone; farnesyltransferase inhibitors such as lonafarnib (SCH 6636, SARASAR.TM.); and pharmaceutically acceptable salts, acids or derivatives of any of the above; as well as combinations of two or more of the above such as CHOP, an abbreviation for a combined therapy of cyclophosphamide, doxorubicin, vincristine, and prednisolone; and FOLFOX, an abbreviation for a treatment regimen with oxaliplatin (ELOXATIN.TM.) combined with 5-FU and leucovorin.
[0114] Chemotherapeutic agents also include non-steroidal anti-inflammatory drugswith analgesic, antipyretic and anti-inflammatory effects. NSAIDs include non-selective inhibitors of the enzyme cyclooxygenase. Specific examples of NSAIDs include aspirin, propionic acid derivatives such as ibuprofen, fenoprofen, ketoprofen, flurbiprofen, oxaprozin and naproxen, acetic acid derivatives such as indomethacin, sulindac, etodolac, diclofenac, enolic acid derivatives such as piroxicam, meloxicam, tenoxicam, droxicam, lornoxicam and isoxicam, fenamic acid derivatives such as mefenamic acid, meclofenamic acid, flufenamic acid, tolfenamic acid, and COX-2 inhibitors such as celecoxib, etoricoxib, lumiracoxib, parecoxib, rofecoxib, rofecoxib, and valdecoxib. NSAIDs can be indicated for the symptomatic relief of conditions such as rheumatoid arthritis, osteoarthritis, inflammatory arthropathies, ankylosing spondylitis, psoriatic arthritis, Reiter's syndrome, acute gout, dysmenorrhoea, metastatic bone pain, headache and migraine, postoperative pain, mild-to-moderate pain due to inflammation and tissue injury, pyrexia, ileus, and renal colic.
[0115] The term "cytokine" is a generic term for proteins released by one cell population that act on another cell as intercellular mediators. Examples of such cytokines are lymphokines, monokines; interleukins (ILs) such as IL-1, IL-1a, IL-2, IL-3, IL-4, IL-5, IL-6, IL-7, IL-8, IL-9, IL-11, IL-12, IL-15; a tumor necrosis factor such as TNF-.alpha. or TNF-.beta.; and other polypeptide factors including LIF and kit ligand (KL) and gamma interferon. As used herein, the term cytokine includes proteins from natural sources or from recombinant cell culture and biologically active equivalents of the native-sequence cytokines, including synthetically produced small-molecule entities and pharmaceutically acceptable derivatives and salts thereof.
[0116] The term "PD-1 axis binding antagonist" is a molecule that inhibits the interaction of a PD-1 axis binding partner with either one or more of its binding partner, so as to remove T-cell dysfunction resulting from signaling on the PD-1 signaling axis--with a result being to restore or enhance T-cell function (e.g., proliferation, cytokine production, target cell killing). As used herein, a PD-1 axis binding antagonist includes a PD-1 binding antagonist, a PD-L1 binding antagonist and a PD-L2 binding antagonist.
[0117] The term "PD-1 binding antagonists" is a molecule that decreases, blocks, inhibits, abrogates or interferes with signal transduction resulting from the interaction of PD-1 with one or more of its binding partners, such as PD-L1, PD-L2. In some embodiments, the PD-1 binding antagonist is a molecule that inhibits the binding of PD-1 to its binding partners. In a specific aspect, the PD-1 binding antagonist inhibits the binding of PD-1 to PD-L1 and/or PD-L2. For example, PD-1 binding antagonists include anti-PD-1 antibodies, antigen binding fragments thereof, immunoadhesins, fusion proteins, oligopeptides and other molecules that decrease, block, inhibit, abrogate or interfere with signal transduction resulting from the interaction of PD-1 with PD-L1 and/or PD-L2. In one embodiment, a PD-1 binding antagonist reduces the negative co-stimulatory signal mediated by or through cell surface proteins expressed on T lymphocytes mediated signaling through PD-1 so as render a dysfunctional T-cell less dysfunctional (e.g., enhancing effector responses to antigen recognition). In some embodiments, the PD-1 binding antagonist is an anti-PD-1 antibody. In a specific aspect, a PD-1 binding antagonist is MDX-1 106 described herein. In another specific aspect, a PD-1 binding antagonist is Merck 3745 described herein. In another specific aspect, a PD-1 binding antagonist is CT-011 described herein.
[0118] The term "PD-L1 binding antagonists" is a molecule that decreases, blocks, inhibits, abrogates or interferes with signal transduction resulting from the interaction of PD-L1 with either one or more of its binding partners, such as PD-1, B7-1. In some embodiments, a PD-1 binding antagonist is a molecule that inhibits the binding of PD-L1 to its binding partners. In a specific aspect, the PD-L1 binding antagonist inhibits binding of PD-L1 to PD-1 and/or B7-1. In some embodiments, the PD-L1 binding antagonists include anti-PD-L1 antibodies, antigen binding fragments thereof, immunoadhesins, fusion proteins, oligopeptides and other molecules that decrease, block, inhibit, abrogate or interfere with signal transduction resulting from the interaction of PD-L1 with one or more of its binding partners, such as PD-1, B7-1. In one embodiment, a PD-L1 binding antagonist reduces the negative co-stimulatory signal mediated by or through cell surface proteins expressed on T lymphocytes mediated signaling through PD-L1 so as to render a dysfunctional T-cell less dysfunctional (e.g., enhancing effector responses to antigen recognition). In some embodiments, a PD-L1 binding antagonist is an anti-PD-L1 antibody. In a specific aspect, an anti-PD-L1 antibody is YW243.55.S70 described herein. In another specific aspect, an anti-PD-L1 antibody is MDX-1 105 described herein. In still another specific aspect, an anti-PD-L1 antibody is MPDL3280A described herein.
[0119] The term "PD-L2 binding antagonists" is a molecule that decreases, blocks, inhibits, abrogates or interferes with signal transduction resulting from the interaction of PD-L2 with either one or more of its binding partners, such as PD-1. In some embodiments, a PD-L2 binding antagonist is a molecule that inhibits the binding of PD-L2 to its binding partners. In a specific aspect, the PD-L2 binding antagonist inhibits binding of PD-L2 to PD-1. In some embodiments, the PD-L2 antagonists include anti-PD-L2 antibodies, antigen binding fragments thereof, immunoadhesins, fusion proteins, oligopeptides and other molecules that decrease, block, inhibit, abrogate or interfere with signal transduction resulting from the interaction of PD-L2 with either one or more of its binding partners, such as PD-1. In one embodiment, a PD-L2 binding antagonist reduces the negative co-stimulatory signal mediated by or through cell surface proteins expressed on T lymphocytes mediated signaling through PD-L2 so as render a dysfunctional T-cell less dysfunctional (e.g., enhancing effector responses to antigen recognition). In some embodiments, a PD-L2 binding antagonist is an immunoadhesin.
[0120] The term "phagocytosis" means the internalization of cells or particulate matter by cells. In some embodiments, the phagocytic cells or phagocytes are macrophages or neutrophils. In some embodiments, the cells are cells that express human OX40. Methods for assaying phagocytosis are known in the art and include use of microscopy to detect the presence of cells internalized within another cells. In other embodiments, phagocytosis is detected using FACS, e.g., by detecting presence of a detectably labeled cell within another cell (which may be detectably labeled, e.g., with a different label than the first cell).
[0121] The phrase "does not possess substantial activity" or "substantially no activity" with respect to an antibody, as used herein, means the antibody does not exhibit an activity that is above background level (in some embodiments, that is above background level that is statistically significant). The phrase "little to no activity" with respect to an antibody, as used herein, means the antibody does not display a biologically meaningful amount of a function. The function can be measured or detected according to any assay or technique known in the art, including, e.g., those described herein. In some embodiments, antibody function is stimulation of effector T cell proliferation and/or cytokine secretion.
[0122] The term "biomarker" or "marker" as used herein refers generally to a molecule, including a gene, mRNA, protein, carbohydrate structure, or glycolipid, the expression of which in or on a tissue or cell or secreted can be detected by known methods (or methods disclosed herein) and is predictive or can be used to predict (or aid prediction) for a cell, tissue, or patient's responsiveness to treatment regimes. In some embodiments, a biomarker may refer to a gene or protein, e.g., the level of expression of the gene or protein detected in one or more cells. In some embodiments, a biomarker may refer to a cell type of interest, e.g., the number of a cell type of interest detected in one or more samples.
[0123] By "patient sample" is meant a collection of cells or fluids obtained from a cancer patient. The source of the tissue or cell sample may be solid tissue as from a fresh, frozen and/or preserved organ or tissue sample or biopsy or aspirate; blood or any blood constituents; bodily fluids such as cerebrospinal fluid, amniotic fluid, peritoneal fluid, or interstitial fluid; cells from any time in gestation or development of the subject. The tissue sample may contain compounds which are not naturally intermixed with the tissue in nature such as preservatives, anticoagulants, buffers, fixatives, nutrients, antibiotics, or the like. Examples of tumor samples herein include, but are not limited to, tumor biopsy, fine needle aspirate, bronchiolar lavage, pleural fluid, sputum, urine, a surgical specimen, circulating tumor cells, serum, plasma, circulating plasma proteins, ascitic fluid, primary cell cultures or cell lines derived from tumors or exhibiting tumor-like properties, as well as preserved tumor samples, such as formalin-fixed, paraffin-embedded tumor samples or frozen tumor samples.
[0124] The phrase "based on expression of" when used herein means that information about expression level or presence or absence of expression (e.g., presence or absence or prevalence of (e.g., percentage of cells displaying) of the one or more biomarkers herein (e.g., presence or absence of or amount or prevalence of FcR-expressing cells, or e.g., presence or absence or amount or prevalence of human effector cells) is used to inform a treatment decision, information provided on a package insert, or marketing/promotional guidance etc.
[0125] A cancer or biological sample which "has human effector cells" is one which, in a diagnostic test, has human effector cells present in the sample (e.g., infiltrating human effector cells).
[0126] A cancer or biological sample which "has FcR-expressing cells" is one which, in a diagnostic test, has FcR-expressing present in the sample (e.g., infiltrating FcR-expressing cells). In some embodiments, FcR is Fc.gamma.R. In some embodiments, FcR is an activating Fc.gamma.R.
[0127] The phrase "recommending a treatment" as used herein refers to using the information or data generated relating to the level or presence of c-met in a sample of a patient to identify the patient as suitably treated or not suitably treated with a therapy. In some embodiments the therapy may comprise c-met antibody (e.g., onartuzumab). In some embodiments, the therapy may comprise VEGF antagonist (e.g., bevacizumab). In some embodiments, the therapy may comprise anti-human OX40 agonist antibody. The information or data may be in any form, written, oral or electronic. In some embodiments, using the information or data generated includes communicating, presenting, reporting, storing, sending, transferring, supplying, transmitting, delivering, dispensing, or combinations thereof. In some embodiments, communicating, presenting, reporting, storing, sending, transferring, supplying, transmitting, delivering, dispensing, or combinations thereof are performed by a computing device, analyzer unit or combination thereof. In some further embodiments, communicating, presenting, reporting, storing, sending, transferring, supplying, transmitting, dispensing, or combinations thereof are performed by an individual (e.g., a laboratory or medical professional). In some embodiments, the information or data includes a comparison of the amount or prevalence of FcR expressing cells to a reference level. In some embodiments, the information or data includes a comparison of the amount or prevalence of human effector cells to a reference level. In some embodiments, the information or data includes an indication that human effector cells or FcR-expressing cells are present or absent in the sample. In some embodiments, the information or data includes an indication that FcR-expressing cells and/or human effector cells are present in a particular percentage of cells (e.g., high prevalence). In some embodiments, the information or data includes an indication that the patient is suitably treated or not suitably treated with a therapy comprising anti-human OX40 agonist antibody.
[0128] The term "detection" includes any means of detecting, including direct and indirect detection.
[0129] The "amount" or "level" of a biomarker associated with an increased clinical benefit to an individual is a detectable level in a biological sample. These can be measured by methods known to one skilled in the art and also disclosed herein. The expression level or amount of biomarker assessed can be used to determine the response to the treatment.
[0130] "Elevated expression," "elevated expression levels," or "elevated levels" refers to an increased expression or increased levels of a biomarker in an individual relative to a control, such as an individual or individuals who are not suffering from the disease or disorder (e.g., cancer), a tumor with a known responsiveness to a treatment (e.g., with an OX40 agonist), an internal control (e.g., housekeeping biomarker), or a reference number (e.g., a set threshold amount, such as a threshold based on clinical outcome data).
[0131] "Reduced expression," "reduced expression levels," or "reduced levels" refers to a decrease expression or decreased levels of a biomarker in an individual relative to a control, such as an individual or individuals who are not suffering from the disease or disorder (e.g., cancer), a tumor with a known responsiveness to a treatment (e.g., with an OX40 agonist), an internal control (e.g., housekeeping biomarker), or a reference number (e.g., a set threshold amount, such as a threshold based on clinical outcome data). In some embodiments, reduced expression is little or no expression.
[0132] The term "diagnosis" is used herein to refer to the identification or classification of a molecular or pathological state, disease or condition (e.g., cancer). For example, "diagnosis" may refer to identification of a particular type of cancer. "Diagnosis" may also refer to the classification of a particular subtype of cancer, e.g., by histopathological criteria, or by molecular features (e.g., a subtype characterized by expression of one or a combination of biomarkers (e.g., particular genes or proteins encoded by said genes)).
[0133] The term "aiding diagnosis" is used herein to refer to methods that assist in making a clinical determination regarding the presence, or nature, of a particular type of symptom or condition of a disease or disorder (e.g., cancer). For example, a method of aiding diagnosis of a disease or condition (e.g., cancer) can comprise measuring certain biomarkers in a biological sample from an individual.
[0134] The term "sample," as used herein, refers to a composition that is obtained or derived from a subject and/or individual of interest that contains a cellular and/or other molecular entity that is to be characterized and/or identified, for example based on physical, biochemical, chemical and/or physiological characteristics. For example, the phrase "disease sample" and variations thereof refers to any sample obtained from a subject of interest that would be expected or is known to contain the cellular and/or molecular entity that is to be characterized. Samples include, but are not limited to, primary or cultured cells or cell lines, cell supernatants, cell lysates, platelets, serum, plasma, vitreous fluid, lymph fluid, synovial fluid, follicular fluid, seminal fluid, amniotic fluid, milk, whole blood, blood-derived cells, urine, cerebrospinal fluid, saliva, sputum, tears, perspiration, mucus, tumor lysates, and tissue culture medium, tissue extracts such as homogenized tissue, tumor tissue, cellular extracts, and combinations thereof.
[0135] By "tissue sample" or "cell sample" is meant a collection of similar cells obtained from a tissue of a subject or individual. The source of the tissue or cell sample may be solid tissue as from a fresh, frozen and/or preserved organ, tissue sample, biopsy, and/or aspirate; blood or any blood constituents such as plasma; bodily fluids such as cerebral spinal fluid, amniotic fluid, peritoneal fluid, or interstitial fluid; cells from any time in gestation or development of the subject. The tissue sample may also be primary or cultured cells or cell lines. Optionally, the tissue or cell sample is obtained from a disease tissue/organ. The tissue sample may contain compounds which are not naturally intermixed with the tissue in nature such as preservatives, anticoagulants, buffers, fixatives, nutrients, antibiotics, or the like.
[0136] As used herein, a "section" of a tissue sample is meant a single part or piece of a tissue sample, e.g. a thin slice of tissue or cells cut from a tissue sample. It is understood that multiple sections of tissue samples may be taken and subjected to analysis according to the present invention, provided that it is understood that the present invention comprises a method whereby the same section of tissue sample is analyzed at both morphological and molecular levels, or is analyzed with respect to protein or nucleic acid.
[0137] By "correlate" or "correlating" is meant comparing, in any way, the performance and/or results of a first analysis or protocol with the performance and/or results of a second analysis or protocol. For example, one may use the results of a first analysis or protocol in carrying out a second protocols and/or one may use the results of a first analysis or protocol to determine whether a second analysis or protocol should be performed. With respect to the embodiment of polypeptide analysis or protocol, one may use the results of the polypeptide expression analysis or protocol to determine whether a specific therapeutic regimen should be performed. With respect to the embodiment of polynucleotide analysis or protocol, one may use the results of the polynucleotide expression analysis or protocol to determine whether a specific therapeutic regimen should be performed.
[0138] "Individual response" or "response" can be assessed using any endpoint indicating a benefit to the individual, including, without limitation, (1) inhibition, to some extent, of disease progression (e.g., cancer progression), including slowing down and complete arrest; (2) a reduction in tumor size; (3) inhibition (i.e., reduction, slowing down or complete stopping) of cancer cell infiltration into adjacent peripheral organs and/or tissues; (4) inhibition (i.e. reduction, slowing down or complete stopping) of metastasis; (5) relief, to some extent, of one or more symptoms associated with the disease or disorder (e.g., cancer); (6) increase or extend in the length of survival, including overall survival and progression free survival; and/or (9) decreased mortality at a given point of time following treatment.
[0139] An "effective response" of a patient or a patient's "responsiveness" to treatment with a medicament and similar wording refers to the clinical or therapeutic benefit imparted to a patient at risk for, or suffering from, a disease or disorder, such as cancer. In one embodiment, such benefit includes any one or more of: extending survival (including overall survival and progression free survival); resulting in an objective response (including a complete response or a partial response); or improving signs or symptoms of cancer.
[0140] By "extending survival" is meant increasing overall or progression free survival in a treated patient relative to an untreated patient (i.e. relative to a patient not treated with the medicament), or relative to a patient who does not express a biomarker at the designated level, and/or relative to a patient treated with an approved anti-tumor agent. An objective response refers to a measurable response, including complete response (CR) or partial response (PR).
III. OX40 Agonists
[0141] Provided herein are methods for predicting responsiveness of a subject having cancer to an OX40 agonist treatment. These methods are based in part on the discovery described herein that the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample containing cancer cells and lymphocytes obtained from a subject having cancer may be used to classify the subject as responsive or non-responsive to an OX40 agonist treatment. It is a further discovery of the present disclosure that the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample containing cancer cells and lymphocytes obtained from a subject having cancer may also be used to determine prognosis of the subject and/or select a subject for treatment with an OX40 agonist of the present disclosure. It is a further discovery of the present disclosure that the number of CD4+, OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample containing metastatic cancer cells and lymphocytes obtained from a subject having cancer may also be used to determine prognosis of the subject and/or select a subject for treatment with an OX40 agonist of the present disclosure. Additionally, as described herein, OX40 agonist antibodies may further find use in methods for quantifying OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample comprising cancer cells (including primary and metastatic cancer cells) and lymphocytes obtained from a subject.
[0142] Antibodies suitable for use in the methods of the invention include antibodies that bind to human OX40. In some embodiments, the anti-OX40 antibody is an agonist antibody. Descriptions of anti-OX40 antibodies (e.g., anti-human OX40 agonist antibodies) may be found in US PG Pub. No. US2015/0307617 and International Publication No. WO/2015/153513, which are each incorporated by reference herein in their entirety.
[0143] In some embodiments, the anti-human OX40 agonist antibody binds human OX40 with an affinity of less than or equal to about 0.45 nM. In some embodiments, the anti-human OX40 antibody binds human OX40 with an affinity of less than or equal to about 0.4 nM. In some embodiments, the anti-human OX40 antibody binds human OX40 with an affinity of less than or equal to about 0.5nM. In some embodiments, the binding affinity is determined using radioimmunoassay.
[0144] In some embodiments, the anti-human OX40 agonist antibody binds human OX40 and cynomolgus OX40. In some embodiments, binding is determined using a FACS assay. In some embodiments, binding to human OX40 has an EC50 of about 0.2 ug/ml. In some embodiments, binding to human OX40 has an EC50 of about 0.3 ug/ml or lower. In some embodiments, binding to cynomolgus OX40 has an EC50 of about 1.5 ug/ml. In some embodiments, binding to cynomolgus OX40 has an EC50 of about 1.4 ug/ml.
[0145] In some embodiments, the anti-human OX40 agonist antibody does not bind to rat OX40 or mouse OX40.
[0146] In some embodiments, the anti-human OX40 agonist antibody is a depleting anti-human OX40 antibody (e.g., depletes cells that express human OX40). In some embodiments, the human OX40 expressing cells are CD4+ effector T cells. In some embodiments, the human OX40 expressing cells are Treg cells. In some embodiments, depleting is by ADCC and/or phagocytosis. In some embodiments, the antibody mediates ADCC by binding Fc.gamma.R expressed by a human effector cell and activating the human effector cell function. In some embodiments, the antibody mediates phagocytosis by binding Fc.gamma.R expressed by a human effector cell and activating the human effector cell function. Exemplary human effector cells include, e.g., macrophage, natural killer (NK) cells, monocytes, neutrophils. In some embodiments, the human effector cell is macrophage. In some embodiments, the human effector cell is NK cells. In some embodiments, depletion is not by apoptosis.
[0147] In some embodiments, the anti-human OX40 agonist antibody has a functional Fc region. In some embodiments, effector function of a functional Fc region is ADCC. In some embodiments, effector function of a functional Fc region is phagocytosis. In some embodiments, effector function of a functional Fc region is ADCC and phagocytosis. In some embodiments, the Fc region is human IgG1. In some embodiments, the Fc region is human IgG4.
[0148] In some embodiments, the anti-human OX40 agonist antibody does not induce apoptosis in OX40-expressing cells (e.g., Treg). In some embodiments, apoptosis is assayed using an antibody concentration of 30 ug/ml, e.g., by determining whether apoptosis has occurred using annexin V and proprodium iodide stained Treg.
[0149] In some embodiments, the anti-human OX40 agonist antibody enhances CD4+ effector T cell function, for example, by increasing CD4+ effector T cell proliferation and/or increasing gamma interferon production by the CD4+ effector T cell (for example, as compared to proliferation and/or cytokine production prior to treatment with anti-human OX40 agonist antibody). In some embodiments, the cytokine is gamma interferon. In some embodiments, the anti-human OX40 agonist antibody increases number of intratumoral (infiltrating) CD4+ effector T cells (e.g., total number of CD4+ effector T cells, or e.g., percentage of CD4+ cells in CD45+ cells), e.g., as compared to number of intratumoral (infiltrating) CD4+ T cells prior to treatment with anti-human OX40 agonist antibody. In some embodiments, the anti-human OX40 agonist antibody increases number of intratumoral (infiltrating) CD4+ effector T cells that express gamma interferon (e.g., total gamma interferon expressing CD4+ cells, or e.g., percentage of gamma interferon expressing CD4+ cells in total CD4+ cells), e.g., as compared to number of intratumoral (infiltrating) CD4+ T cells that express gamma interferon prior to treatment with anti-human OX40 agonist antibody.
[0150] In some embodiments, the anti-human OX40 agonist antibody increases number of intratumoral (infiltrating) CD8+ effector T cells (e.g., total number of CD8+ effector T cells, or e.g., percentage of CD8+ in CD45+ cells), e.g., as compared to number of intratumoral (infiltrating) CD8+ T effector cells prior to treatment with anti-human OX40 agonist antibody. In some embodiments, the anti-human OX40 agonist antibody increases number of intratumoral (infiltrating) CD8+ effector T cells that express gamma interferon (e.g., percentage of CD8+ cells that express gamma interferon in total CD8+ cells), e.g., compared to number of intratumoral (infiltrating) CD8+ T cells that express gamma interferon prior to treatment with anti-human OX40 agonist antibody.
[0151] In some embodiments, the anti-human OX40 agonist antibody enhances memory T cell function, for example by increasing memory T cell proliferation and/or increasing cytokine production by the memory cell. In some embodiments, the cytokine is gamma interferon.
[0152] In some embodiments, the anti-human OX40 agonist antibody inhibits Treg function, for example, by decreasing Treg suppression of effector T cell function (e.g., effector T cell proliferation and/or effector T cell cytokine secretion). In some embodiments, the effector T cell is a CD4+ effector T cell. In some embodiments, the anti-human OX40 agonist antibody reduces the number of intratumoral (infiltrating) Treg (e.g., total number of Treg or e.g., percentage of Fox3p+ cells in CD4+ cells).
[0153] In some embodiments, the anti-human OX40 agonist antibody is engineered to increase effector function (e.g., compared to effector function in a wild-type IgG1). In some embodiments, the antibody has increased binding to a Fc.gamma.receptor. In some embodiments, the antibody lacks fucose attached (directly or indirectly) to the Fc region. For example, the amount of fucose in such antibody may be from 1% to 80%, from 1% to 65%, from 5% to 65% or from 20% to 40%. In some embodiments, the Fc region comprises bisected oligosaccharides, e.g., in which a biantennary oligosaccharide attached to the Fc region of the antibody is bisected by GlcNAc. In some embodiments, the antibody comprises an Fc region with one or more amino acid substitutions which improve ADCC, e.g., substitutions at positions 298, 333, and/or 334 of the Fc region (EU numbering of residues).
[0154] In some embodiments, the anti-human OX40 agonist antibody increases OX40 signal transduction in a target cell that expresses OX40. In some embodiments, OX40 signal transduction is detected by monitoring NFkB downstream signaling.
[0155] In some embodiments, the anti-human OX40 agonist antibody is stable after treatment at 40 C for two weeks.
[0156] In some embodiments, the anti-human OX40 agonist antibody binds human effector cells, e.g., binds Fc.gamma.R (e.g., an activating Fc.gamma.R) expressed by human effector cells. In some embodiments, the human effector cell performs (is capable of performing) ADCC effector function. In some embodiments, the human effector cell performs (is capable of performing) phagocytosis effector function.
[0157] In some embodiments, the anti-human OX40 agonist antibody comprising a variant IgG1 Fc polypeptide comprising a mutation that eliminates binding to human effector cells (e.g., a DANA mutation) has diminished activity (e.g., CD4+ effector T cell function, e.g., proliferation), relative to anti-human OX40 agonist antibody comprising native sequence IgG1 Fc portion. In some embodiment, the anti-human OX40 agonist antibody comprising a variant IgG1 Fc polypeptide comprising a mutation that eliminates binding to human effector cells (e.g., a DANA mutation) does not possess substantial activity (e.g., CD4+ effector T cell function, e.g., proliferation).
[0158] In some embodiments, antibody cross-linking is required for anti-human OX40 agonist antibody function. In some embodiments, function is stimulation of CD4+ effector T cell proliferation. In some embodiments, antibody cross-linking is determined by providing anti-human OX40 agonist antibody adhered on a solid surface (e.g., a cell culture plate). In some embodiments, antibody cross-linking is determined by introducing a mutation in the antibody's IgG1 Fc portion (e.g., a DANA mutation) and testing function of the mutant antibody.
[0159] In some embodiments, the anti-human OX40 agonist antibody competes for binding to human OX40 with OX40L. In some embodiments, addition of OX40L does not enhance anti-human OX40 antibody function in an in vitro assay.
[0160] According to another embodiment, the anti-human OX40 agonist antibodies include any one, any combination, or all of the following properties: (1) binds human OX40 with an affinity of less than or equal to about 0.45 nM, in some embodiments, binds human OX40 with an affinity of less than or equal to about 0.4 nM, in some embodiments, binds human OX40 with an affinity of less than or equal to about 0.5nM, in some embodiments, the binding affinity is determined using radioimmunoassay; (2) binds human OX40 and cynomolgus OX40, in some embodiments, binding is determined using a FACS assay, (3) binds human OX40 with an EC50 of about 0.2 ug/ml, in some embodiments, binds to human OX40 has an EC50 of about 0.3 ug/ml or lower, in some embodiments, binds to cynomolgus OX40 with an EC50 of about 1.5 ug/ml, in some embodiments, binds to cynomolgus OX40 has an EC50 of about 1.4 ug/ml, (4) does not substantially bind to rat OX40 or mouse OX40, (6) is a depleting anti-human OX40 antibody (e.g., depletes cells that express human OX40), in some embodiments, the cells are CD4+ effector T cells and/or Treg cells, (7) enhances CD4+ effector T cell function, for example, by increasing CD4+ effector T cell proliferation and/or increasing gamma interferon production by the CD4+ effector T cell (for example, as compared to proliferation and/or cytokine production prior to treatment with anti-human OX40 agonist antibody), (8) enhances memory T cell function, for example by increasing memory T cell proliferation and/or increasing cytokine production by the memory cell, (9) inhibits Treg function, for example, by decreasing Treg suppression of effector T cell function (e.g., effector T cell proliferation and/or effector T cell cytokine secretion). In some embodiments, the effector T cell is a CD4+ effector T cell, (10) increases OX40 signal transduction in a target cell that expresses OX40 (in some embodiments, OX40 signal transduction is detected by monitoring NFkB downstream signaling), (11) is stable after treatment at 40C for two weeks, (12) binds human effector cells, e.g., binds Fc.gamma.R expressed by human effector cells, (13) anti-human OX40 agonist antibody comprising a variant IgG1 Fc polypeptide comprising a mutation that eliminates binding to human effector cells (e.g., N297G) has diminished activity (e.g., CD4+ effector T cell function, e.g., proliferation), relative to anti-human OX40 agonist antibody comprising native sequence IgG1 Fc portion, in some embodiment, the anti-human OX40 agonist antibody comprising a variant IgG1 Fc polypeptide comprising a mutation that eliminates binding to human effector cells (e.g., N297G) does not possess substantial activity (e.g., CD4+ effector T cell function, e.g., proliferation), (14) antibody cross-linking (e.g., by Fc receptor binding) is required for anti-human OX40 agonist antibody function.
[0161] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7.
[0162] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, at least two, or all three VH HVR sequences selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4. In one embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4. In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4 and HVR-L3 comprising the amino acid sequence of SEQ ID NO:7. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4, HVR-L3 comprising the amino acid sequence of SEQ ID NO:7, and HVR-H2 comprising the amino acid sequence of SEQ ID NO:3. In a further embodiment, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4.
[0163] In another aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, at least two, or all three VL HVR sequences selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7. In one embodiment, the antibody comprises (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7.
[0164] In another aspect, an anti-human OX40 agonist antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:4; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7.
[0165] In another aspect, the invention provides an anti-human OX40 agonist antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7.
[0166] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:26.
[0167] In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4 and HVR-L3 comprising the amino acid sequence of SEQ ID NO:26. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4, HVR-L3 comprising the amino acid sequence of SEQ ID NO:26, and HVR-H2 comprising the amino acid sequence of SEQ ID NO:3.
[0168] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:4; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:26.
[0169] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:26.
[0170] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:27.
[0171] In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4 and HVR-L3 comprising the amino acid sequence of SEQ ID NO:27. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4, HVR-L3 comprising the amino acid sequence of SEQ ID NO:27, and HVR-H2 comprising the amino acid sequence of SEQ ID NO:3.
[0172] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:4; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:27.
[0173] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:27.
[0174] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, 8 or 9; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, 10, 11, 12, 13 or 14; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4, 15, or 19; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7, 22, 23, 24, 25, 26, 27, or 28.
[0175] In one aspect, the invention provides an antibody comprising at least one, at least two, or all three VH HVR sequences selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO: 2, 8 or 9; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 3, 10, 11, 12, 13 or 14; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO: 4, 15, or 19. In one embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO: 4, 15, or 19. In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:4, 15, or 19 and HVR-L3 comprising the amino acid sequence of SEQ ID NO: 7, 22, 23, 24, 25, 26, 27, or 28. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO: 4, 15, or 19, HVR-L3 comprising the amino acid sequence of SEQ ID NO: 7, 22, 23, 24, 25, 26, 27, or 28, and HVR-H2 comprising the amino acid sequence of SEQ ID NO: 3, 10, 11, 12, 13 or 14. In a further embodiment, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO: 2, 8 or 9; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 3, 10, 11, 12, 13 or 14; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO: 4, 15, or 19.
[0176] In another aspect, the invention provides an antibody comprising at least one, at least two, or all three VL HVR sequences selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO: 5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 7, 22, 23, 24, 25, 26, 27, or 28. In one embodiment, the antibody comprises (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 7, 22, 23, 24, 25, 26, 27, or 28.
[0177] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO: 2, 8 or 9, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 3, 10, 11, 12, 13 or 14, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO: 4, 15, or 19; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 7, 22, 23, 24, 25, 26, 27, or 28.
[0178] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO: 2, 8 or 9; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 3, 10, 11, 12, 13 or 14; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO: 4, 15, or 19; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO: 7, 22, 23, 24, 25, 26, 27, or 28.
[0179] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:172; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:173; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:174; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:175. In some embodiment, HVR-H2 is not DMYPDAAAASYNQKFRE (SEQ ID NO:222),In some embodiments, HVR-H3 is not APRWAAAA (SEQ ID NO:223). In some embodiments, HVR-L3 is not QAAAAAAAT (SEQ ID NO:224).
[0180] In one aspect, the invention provides an antibody comprising at least one, at least two, or all three VH HVR sequences selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:172; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:173; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:174. In one embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:174. In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:174 and HVR-L3 comprising the amino acid sequence of SEQ ID NO:175. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:174, HVR-L3 comprising the amino acid sequence of SEQ ID NO:175, and HVR-H2 comprising the amino acid sequence of SEQ ID NO:173. In a further embodiment, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:172; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:173; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:174. In some embodiment, HVR-H2 is not DMYPDAAAASYNQKFRE (SEQ ID NO:222). In some embodiments, HVR-H3 is not APRWAAAA (SEQ ID NO:223). In some embodiments, HVR-L3 is not QAAAAAAAT (SEQ ID NO:224).
[0181] In another aspect, the invention provides an antibody comprising (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:175. In some embodiments, HVR-L3 is not QAAAAAAAT (SEQ ID NO:224).
[0182] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:172, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:173, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:174; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:175.
[0183] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:172; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:173; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:174; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:175. In some embodiment, HVR-H2 is not DMYPDAAAASYNQKFRE (SEQ ID NO:222),In some embodiments, HVR-H3 is not APRWAAAA (SEQ ID NO:223). In some embodiments, HVR-L3 is not QAAAAAAAT (SEQ ID NO:224).
[0184] All possible combinations of the above substitutions are encompassed by the consensus sequences of SEQ ID NO:172, 173, 174 and 175.
[0185] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0186] In one aspect, the invention provides an antibody comprising at least one, at least two, or all three VH HVR sequences selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33. In one embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:33. In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:33 and HVR-L3 comprising the amino acid sequence of SEQ ID NO:42. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:33, HVR-L3 comprising the amino acid sequence of SEQ ID NO:42, and HVR-H2 comprising the amino acid sequence of SEQ ID NO:30. In a further embodiment, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33.
[0187] In another aspect, the invention provides an antibody comprising at least one, at least two, or all three VL HVR sequences selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42. In one embodiment, the antibody comprises (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0188] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:33; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0189] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:42.
[0190] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:40; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0191] In another aspect, the invention provides an antibody comprising at least one, at least two, or all three VL HVR sequences selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:40; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42. In one embodiment, the antibody comprises (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:40; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0192] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:33; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:40, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0193] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:40; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:42.
[0194] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30, 31, or 32; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39, 40 or 41; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42, 43, or 44.
[0195] In one aspect, the invention provides an antibody comprising at least one, at least two, or all three VH HVR sequences selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 30, 31, or 32; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33. In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:33 and HVR-L3 comprising the amino acid sequence of SEQ ID NO: 42, 43, or 44. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:33, HVR-L3 comprising the amino acid sequence of SEQ ID NO: 42, 43, or 44, and HVR-H2 comprising the amino acid sequence of SEQ ID NO: 39, 40 or 41. In a further embodiment, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30, 31, or 32; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33.
[0196] In another aspect, the invention provides an antibody comprising at least one, at least two, or all three VL HVR sequences selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO: 39, 40 or 41; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 42, 43, or 44. In one embodiment, the antibody comprises (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO: 39, 40 or 41; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 42, 43, or 44.
[0197] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 30, 31, or 32, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:33; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO: 39, 40 or 41, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 42, 43, or 44.
[0198] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 30, 31, or 32; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO: 39, 40 or 41; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO: 42, 43, or 44.
[0199] In one aspect, the invention provides an anti-human OX40 agonist antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:175; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:177; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:178.
[0200] In one aspect, the invention provides an antibody comprising at least one, at least two, or all three VH HVR sequences selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:175; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33. In another embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:33 and HVR-L3 comprising the amino acid sequence of SEQ ID NO:177. In a further embodiment, the antibody comprises HVR-H3 comprising the amino acid sequence of SEQ ID NO:33, HVR-L3 comprising the amino acid sequence of SEQ ID NO:178, and HVR-H2 comprising the amino acid sequence of SEQ ID NO:176. In a further embodiment, the antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:176; and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33.
[0201] In another aspect, the invention provides an antibody comprising at least one, at least two, or all three VL HVR sequences selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:177; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:177. In one embodiment, the antibody comprises (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:177; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:178.
[0202] In another aspect, an antibody of the invention comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:176, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:33; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:177, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:178.
[0203] In another aspect, the invention provides an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:176; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:177; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:178.
[0204] In any of the above embodiments, an anti-OX40 agonist antibody is humanized. In one embodiment, an anti-OX40 antibody comprises HVRs as in any of the above embodiments or for any of the embodiments in FIG. 11, and further comprises an acceptor human framework, e.g. a human immunoglobulin framework or a human consensus framework. In another embodiment, an anti-OX40 antibody comprises HVRs as in any of the above embodiments, and further comprises a VH and/or VL comprising an FR sequence shown in FIG. 11.
[0205] In another aspect, an anti-human OX40 agonist antibody comprises a heavy chain variable domain (VH) sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:56, 58, 60, 62, 64, 66, 68, 70, 72, 74, 76, 78, 80, 82, 84, 86, 88, 90, 92, 94, 96, 98, 100, 108, 114 or 116. In certain embodiments, a VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:56, 58, 60, 62, 64, 66, 68, 70, 72, 74, 76, 78, 80, 82, 84, 86, 88, 90, 92, 94, 96, 98, 100, 108, 114 or 116. In certain embodiments, substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VH sequence in SEQ ID NO: SEQ ID NO:56, 58, 60, 62, 64, 66, 68, 70, 72, 74, 76, 78, 80, 82, 84, 86, 88, 90, 92, 94, 96, 98, 100, 108, 114 or 116, including post-translational modifications of that sequence. In a particular embodiment, the VH comprises one, two or three HVRs selected from: (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4.
[0206] In another aspect, an anti-human OX40 agonist antibody is provided, wherein the antibody comprises a light chain variable domain (VL) having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:57, 59, 61, 63, 65, 67, 69, 71, 73, 75, 77, 79, 81, 83, 85, 87, 89, 91, 93, 95, 97, 99, 101, 109, 115 or 117. In certain embodiments, a VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO: 57, 59, 61, 63, 65, 67, 69, 71, 73, 75, 77, 79, 81, 83, 85, 87, 89, 91, 93, 95, 97, 99, 101, 109, 115 or 117. In certain embodiments, the substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VL sequence in SEQ ID NO: 57, 59, 61, 63, 65, 67, 69, 71, 73, 75, 77, 79, 81, 83, 85, 87, 89, 91, 93, 95, 97, 99, 101, 109, 115 or 117, including post-translational modifications of that sequence. In a particular embodiment, the VL comprises one, two or three HVRs selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7.
[0207] In another aspect, an anti-human OX40 agonist antibody comprises a heavy chain variable domain (VH) sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:56. In certain embodiments, a VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:56. In certain embodiments, substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VH sequence in SEQ ID NO:56, including post-translational modifications of that sequence. In a particular embodiment, the VH comprises one, two or three HVRs selected from: (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4.
[0208] In another aspect, an anti-human OX40 agonist antibody is provided, wherein the antibody comprises a light chain variable domain (VL) having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:57. In certain embodiments, a VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO: 57. In certain embodiments, the substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VL sequence in SEQ ID NO: 57, including post-translational modifications of that sequence. In a particular embodiment, the VL comprises one, two or three HVRs selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7.
[0209] In another aspect, an anti-human OX40 agonist antibody comprises a heavy chain variable domain (VH) sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:94. In certain embodiments, a VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:94. In certain embodiments, substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VH sequence in SEQ ID NO:94, including post-translational modifications of that sequence. In a particular embodiment, the VH comprises one, two or three HVRs selected from: (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4.
[0210] In another aspect, an anti-human OX40 agonist antibody is provided, wherein the antibody comprises a light chain variable domain (VL) having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:95. In certain embodiments, a VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:95. In certain embodiments, the substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VL sequence in SEQ ID NO:95, including post-translational modifications of that sequence. In a particular embodiment, the VL comprises one, two or three HVRs selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:26.
[0211] In another aspect, an anti-human OX40 agonist antibody comprises a heavy chain variable domain (VH) sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:96. In certain embodiments, a VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:96. In certain embodiments, substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VH sequence in SEQ ID NO:96, including post-translational modifications of that sequence. In a particular embodiment, the VH comprises one, two or three HVRs selected from: (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4.
[0212] In another aspect, an anti-human OX40 agonist antibody is provided, wherein the antibody comprises a light chain variable domain (VL) having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:97. In certain embodiments, a VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:97. In certain embodiments, the substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VL sequence in SEQ ID NO:97, including post-translational modifications of that sequence. In a particular embodiment, the VL comprises one, two or three HVRs selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:27.
[0213] In another aspect, an anti-human OX40 agonist antibody comprises a heavy chain variable domain (VH) sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO: 118, 120, 122, 124, 126, 128, 130, 132, 134, 136, 138, 140, 142, 144, 146, 148. In certain embodiments, a VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO: 118, 120, 122, 124, 126, 128, 130, 132, 134, 136, 138, 140, 142, 144, 146, 148. In certain embodiments, substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VH sequence in SEQ ID NO: SEQ ID NO: 118, 120, 122, 124, 126, 128, 130, 132, 134, 136, 138, 140, 142, 144, 146, 148, including post-translational modifications of that sequence. In a particular embodiment, the VH comprises one, two or three HVRs selected from: (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO: 29, (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30, and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:33.
[0214] In another aspect, an anti-human OX40 agonist antibody is provided, wherein the antibody comprises a light chain variable domain (VL) having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO: 119, 121, 123, 125, 127, 129, 131, 133, 135, 137, 139, 141, 143, 145, 147, 149. In certain embodiments, a VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO: 119, 121, 123, 125, 127, 129, 131, 133, 135, 137, 139, 141, 143, 145, 147, 149. In certain embodiments, the substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VL sequence in SEQ ID NO: 119, 121, 123, 125, 127, 129, 131, 133, 135, 137, 139, 141, 143, 145, 147, 149, including post-translational modifications of that sequence. In a particular embodiment, the VL comprises one, two or three HVRs selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0215] In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:56 and SEQ ID NO:57, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:58 and SEQ ID NO:59, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:60 and SEQ ID NO:61, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:62 and SEQ ID NO:63, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:64 and SEQ ID NO:65, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:66 and SEQ ID NO:67, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:68 and SEQ ID NO:69, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:70 and SEQ ID NO:71, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:72 and SEQ ID NO:73, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:74 and SEQ ID NO:75, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:76 and SEQ ID NO:77, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:78 and SEQ ID NO:79, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:80 and SEQ ID NO:81, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:82 and SEQ ID NO:83, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:84 and SEQ ID NO:85, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:86 and SEQ ID NO:87, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:88 and SEQ ID NO:89, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:90 and SEQ ID NO:91, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:92 and SEQ ID NO:93, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:94 and SEQ ID NO:95, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:96 and SEQ ID NO:97, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:98 and SEQ ID NO:99, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:100 and SEQ ID NO:101, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:108 and SEQ ID NO:109, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:114 and SEQ ID NO:115, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:116 and SEQ ID NO:117, respectively, including post-translational modifications of those sequences.
[0216] In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:118 and SEQ ID NO:119, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:120 and SEQ ID NO:121, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:122 and SEQ ID NO:123, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:124 and SEQ ID NO:125, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:126 and SEQ ID NO:127, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:128 and SEQ ID NO:129, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:130 and SEQ ID NO:131, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:132 and SEQ ID NO:133, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:134 and SEQ ID NO:135, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:136 and SEQ ID NO:137, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:138 and SEQ ID NO:139, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:140 and SEQ ID NO:141, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:142 and SEQ ID NO:143, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:144 and SEQ ID NO:145, respectively, including post-translational modifications of those sequences. In one embodiment, the antibody comprises the VH and VL sequences in SEQ ID NO:146 and SEQ ID NO:147, respectively, including post-translational modifications of those sequences.
[0217] In another aspect, an anti-human OX40 agonist antibody is provided, wherein the antibody comprises a VH as in any of the embodiments provided above, and a VL as in any of the embodiments provided above.
[0218] In some embodiments, the OX40 agonist antibody is MEDI6469. In some embodiments, the OX40 agonist antibody is MEDI0562.
[0219] In a further aspect, the invention provides an antibody that binds to the same epitope as an anti-human OX40 antibody provided herein. In some embodiments, the antibody is an anti-human OX40 agonist antibody.
[0220] In a further aspect of the invention, an anti-OX40 antibody according to any of the above embodiments is a monoclonal antibody, including a chimeric, humanized or human antibody. In one embodiment, an anti-OX40 antibody is an antibody fragment, e.g., a Fv, Fab, Fab', scFv, diabody, or F(ab')2 fragment. In another embodiment, the antibody is a full length antibody, e.g., an intact IgG1 antibody or other antibody class or isotype as defined herein. In some embodiments, the antibody is a full length intact IgG4 antibody.
[0221] Exemplary amino acid sequences corresponding to OX40 polypeptides and OX40 antibodies are provided below.
TABLE-US-00003 TABLE 2 Amino acid sequences SEQ ID Name SEQUENCE NO: Human OX40 LHCVGDTYPSNDRCCHECRPGNGMVSRCSRSQNTVCRPCGPGFY 1 (lacking the NDVVSSKPCKPCTWCNLRSGSERKQLCTATQDTVCRCRAGTQPL signal peptide) DSYKPGVDCAPCPPGHFSPGDNQACKPWTNCTLAGKHTLQPASN SSDAICEDRDPPATQPQETQGPPARPITVQPTEAWPRTSQGPSTRP VEVPGGRAVAAILGLGLVLGLLGPLAILLALYLLRRDQRLPPDAH KPPGGGSFRTPIQEEQADAHSTLAKI HVR-H1- DSYMS 2 1A7.gr.1 1A7.gr.2 1A7.gr.3 1A7.gr.4 1A7.gr.5 1A7.gr.6 1A7.gr.7 1A7.gr.NADS 1A7.gr.NADA 1A7.gr.NGDA 1A7.gr.SGDS 1A7.gr.NGSS 1A7.Ala.1 1A7.Ala.2 1A7.Ala.3 1A7.Ala.4 1A7.Ala.5 1A7.Ala.6 1A7.Ala.7 1A7.Ala.8 1A7.Ala.9 1A7.Ala.10 1A7.Ala.11 1A7.Ala.12 1A7.Ala.13 1A7.Ala.14 1A7.Ala.15 1A7.Ala.16 HVR-H2- DMYPDNGDSSYNQKFRE 3 1A7.gr.1 1A7.gr.2 1A7.gr.3 1A7.gr.4 1A7.gr.5 1A7.gr.6 1A7.gr.7 1A7.gr.DA 1A7.gr.ES 1A7.Ala.1 1A7.Ala.2 1A7.Ala.3 1A7.Ala.4 1A7.Ala.5 1A7.Ala.6 1A7.Ala.7 1A7.Ala.8 1A7.Ala.9 1A7.Ala.10 1A7.Ala.11 1A7.Ala.12 1A7.Ala.13 1A7.Ala.14 1A7.Ala.15 1A7.Ala.16 HVR-H3- APRWYFSV 4 1A7.gr.1 1A7.gr.2 1A7.gr.3 1A7.gr.4 1A7.gr.5 1A7.gr.6 1A7.gr.7 1A7.gr.DA 1A7.gr.ES 1A7.gr.NADS 1A7.gr.NADA 1A7.gr.NGDA 1A7.gr.SGDS 1A7.gr.NGSS 1A7.gr.DANADA 1A7.Ala.1 1A7.Ala.2 1A7.Ala.3 1A7.Ala.4 1A7.Ala.5 1A7.Ala.6 1A7.Ala.7 1A7-Ala.15 1A7.Ala.16 HVR-L1- RASQDISNYLN 5 1A7.gr.1 1A7.gr.2 1A7.gr.3 1A7.gr.4 1A7.gr.5 1A7.gr.6 1A7.gr.7 1A7.gr.DA 1A7.gr.ES 1A7.gr.NADS 1A7.gr.NADA 1A7.gr.NGDA 1A7.gr.SGDS 1A7.gr.NGSS 1A7.gr.DANADA 1A7.Ala.1 1A7.Ala.2 1A7.Ala.3 1A7.Ala.4 1A7.Ala.5 1A7.Ala.6 1A7.Ala.7 1A7.Ala.8 1A7.Ala.9 1A7.Ala.10 1A7.Ala.11 1A7.Ala.12 1A7.Ala.13 1A7.Ala.14 1A7.Ala.15 1A7.Ala.16 HVR-L2- YTSRLRS 6 1A7.gr.1 1A7.gr.2 1A7.gr.3 1A7.gr.4 1A7.gr.5 1A7.gr.6 1A7.gr.7 1A7.gr.DA 1A7.gr.ES 1A7.gr.NADS 1A7.gr.NADA 1A7.gr.NGDA 1A7.gr.SGDS 1A7.gr.NGSS 1A7.gr.DANADA 1A7.Ala.1 1A7.Ala.2 1A7.Ala.3 1A7.Ala.4 1A7.Ala.5 1A7.Ala.6 1A7.Ala.7 1A7.Ala.8 1A7.Ala.9 1A7.Ala.10 1A7.Ala.11 1A7.Ala.12 1A7.Ala.13 1A7.Ala.14 1A7.Ala.15 1A7.Ala.16 HVR-L3- QQGHTLPPT 7 1A7.gr.1 1A7.gr.2 1A7.gr.3 1A7.gr.4 1A7.gr.5 1A7.gr.6 1A7.gr.7 1A7.gr.DA 1A7.gr.ES 1A7.gr.NADS 1A7.gr.NADA 1A7.gr.NGDA 1A7.gr.SGDS 1A7.gr.NGSS 1A7.gr.DANADA 1A7.Ala.8 1A7.Ala.9 1A7.Ala.10 1A7.Ala.11 1A7.Ala.12 1A7.Ala.13 1A7.Ala.14 1A7.Ala.15 1A7.Ala.16 HVR-H1- DAYMS 8 1A7.gr.DA HVR-H1- ESYMS 9 1A7.gr.ES 1A7.gr.DANADA HVR-H2- DMYPDNADSSYNQKFRE 10 1A7.gr.NADS HVR-H2- DMYPDNADASYNQKFRE 11 1A7.gr.NADA 1A7.gr.DANADA HVR-H2- DMYPDNGDASYNQKFRE 12 1A7.gr.NGDA HVR-H2- DMYPDSGDSSYNQKFRE 13 1A7.gr.SGDS HVR-H2- DMYPDNGSSSYNQKFRE 14 1A7.gr.NGSS HVR-H3- APRWYFSA 15 1A7.Ala.8 HVR-H3- APRWYASV 16 1A7.Ala.9 HVR-H3- APRWAFSV 17 1A7.Ala.10 HVR-H3- APAWYFSV 18 1A7.Ala.11 HVR-H3- APRWYFAV 19 1A7.Ala.12 HVR-H3- APRAYFSV 20 1A7.Ala.13 HVR-H3- AARWYFSV 21 1A7.Ala.14 HVR-L3- QQGHTLPAT 22 1A7.Ala.1 HVR-L3- QQGHTAPPT 23 1A7.Ala.2 HVR-L3- QQGATLPPT 24 1A7.Ala.3 HVR-L3- QQGHALPPT 25 1A7.Ala.4 HVR-L3- QQAHTLPPT 26 1A7.Ala.5 HVR-L3- QQGHTLAPT 27 1A7.Ala.6
HVR-L3- QAGHTLPPT 28 1A7.Ala.7 HVR-H1- NYLIE 29 3C8.gr.1 3C8.gr.2 3C8.gr.3 3C8.gr.4 3C8.gr.5 3C8.gr.5.SG 3C8.gr.5.EG 3C8.gr.5.QG 3C9.gr.5.DQ 3C8.gr.5.DA 3C8.gr.6 3C8.gr.7 3C8.gr.8 3C8.gr.9 3C8.gr.10 3C8.gr.11 3C8.A.1 3C8.A.2 3C8.A.3 3C8.A.4 3C8.A.5 3C8.A.6 3C8.A.7 3C8.A.8 3C8.A.9 3C8.A.10 HVR-H2- VINPGSGDTYYSEKFKG 30 3C8.gr.1 3C8.gr.2 3C8.gr.3 3C8.gr.4 3C8.gr.5 3C8.gr.5.SG 3C8.gr.5.EG 3C8.gr.5.QG 3C8.gr.6 3C8.gr.7 3C8.gr.8 3C8.gr.9 3C8.gr.10 3C8.gr.11 3C8.A.1 3C8.A.2 3C8.A.3 3C8.A.4 3C8.A.5 3C8.A.6 3C8.A.7 3C8.A.8 3C8.A.9 3C8.A.10 HVR-H2- VINPGSGDAYYSEKFKG 31 3C8.gr.5.DA HVR-H2- VINPGSGDQYYSEKFKG 32 3C8.gr.5.DQ HVR-H3- DRLDY 33 3C8.gr.1 3C8.gr.2 3C8.gr.3 3C8.gr.4 3C8.gr.5 3C8.gr.5.SG 3C8.gr.5.EG 3C8.gr.5.QG 3C8.gr.5.DA 3C8.gr.5.DQ 3C8.gr.6 3C8.gr.7 3C8.gr.8 3C8.gr.9 3C8.gr.10 3C8.gr.11 3C8.A.1 3C8.A.2 3C8.A.3 3C8.A.4 3C8.A.5 3C8.A.6 3C8.A.7 HVR-H3- ARLDY 34 3C8.A.8 HVR-H3- DALDY 35 3C8.A.9 HVR-H3- DRADY 36 3C8.A.10 HVR-L1- HASQDISSYIV 37 3C8.gr.1 3C8.gr.2 3C8.gr.3 3C8.gr.4 3C8.gr.5 3C8.gr.5.SG 3C8.gr.5.EG 3C8.gr.5.QG 3C8.gr.5.DA 3C8.gr.5.DQ 3C8.gr.6 3C8.gr.7 3C8.gr.8 3C8.gr.9 3C8.gr.10 3C8.gr.11 3C8.A.1 3C8.A.2 3C8.A.3 3C8.A.4 3C8.A.5 3C8.A.6 3C8.A.7 3C8.A.8 3C8.A.9 3C8.A.10 HVR-L2- HGTNLED 38 3C8.gr.1 3C8.gr.2 3C8.gr.3 3C8.gr.4 3C8.gr.5 3C8.gr.5.DA 3C8.gr.5.DQ 3C8.gr.6 3C8.gr.7 3C8.gr.8 3C8.gr.9 3C8.gr.10 3C8.gr.11 3C8.A.1 3C8.A.2 3C8.A.3 3C8.A.4 3C8.A.5 3C8.A.6 3C8.A.7 3C8.A.8 3C8.A.9 3C8.A.10 HVR-L2- HGTNLES 39 3C8.gr5.SG HVR-L2- HGTNLEE 40 3C8.gr.5.EG HVR-L2- HGTNLEQ 41 3C8.gr.5.QG HVR-L3 VHYAQFPYT 42 3C8.gr.1 3C8.gr.2 3C8.gr.3 3C8.gr.4 3C8.gr.5 3C8.gr.5.SG 3C8.gr.5.EG 3C8.gr.5.QG 3C8.gr.5.DA 3C8.gr.5.DQ 3C8.gr.6 3C8.gr.7 3C8.gr.8 3C8.gr.9 3C8.gr.10 3C8.gr.11 3C8.A.8 3C8.A.9 3C8.A.10 HVR-L3- AHYAQFPYT 43 3C8.A.1 HVR-L3- VAYAQFPYT 44 3C8.A.2 HVR-L3- VHAAQFPYT 45 3C8.A.3 HVR-L3- VHYAAFPYT 46 3C8.A.4 HVR-L3- VHYAQAPYT 47 3C8.A.5 HVR-L3- VHYAQFAYT 48 3C8.A.6 HVR-L3- VHYAQFPAT 49 3C8.A.7 HVR-H1- DYGV.sub.L 50 1D2.gr.1 1D2.gr.2 1D2.gr.3 HVR-H2- MIWSGGTTDYNAAFIS 51 1D2.gr.1 1D2.gr.2 1D2.gr.3 HVR-H3- EEMDY 52 1D2.gr.1 1D2.gr.2 1D2.gr.3 HVR-L1- RASQDISNFLN 53 1D2.gr.1 1D2.gr.2 1D2.gr.3 HVR-L2- YTSRLHS 54 1D2.gr.1 1D2.gr.2 1D2.gr.3 HVR-L3- QQGNTLPWT 55 1D2.gr.1 1D2.gr.2 1D2.gr.3 1A7.gr.1 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 56 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.1 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 57 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.2 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 58 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITVDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.2 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 59 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.3 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 60 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTLTVDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS
1A7.gr.3 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 61 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.4 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 62 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITVDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.4 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKTVK 63 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.5 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 64 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITVDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.5 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKTVK 65 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.6 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 66 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITVDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.6 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKTVK 67 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGKDYTLTISSLQPEDFATYFCQQG HTLPPTFGQGTKVEIK 1A7.gr.7 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 68 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITVDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.7 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKTVK 69 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGKDYTLTISSLQPEDFATYFCQQG HTLPPTFGQGTKVEIK 1A7.gr.DA EVQLVQSGAEVKKPGASVKVSCKASGYTFTDAYMSWVRQAPGQ 70 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.DA DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 71 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.ES EVQLVQSGAEVKKPGASVKVSCKASGYTFTESYMSWVRQAPGQ 72 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.ES DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 73 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.NADS EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 74 V.sub.H GLEWIGDMYPDNADSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.NADS DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 75 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.NADA EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 76 V.sub.H GLEWIGDMYPDNADASYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.NADA DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 77 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.NGDA EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 78 V.sub.H GLEWIGDMYPDNGDASYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.NGDA DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 79 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.SGDS EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 80 V.sub.H GLEWIGDMYPDSGDSSYNQKFRERVTITRDTSTSTAYLELSSLRSE DTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.SGDS DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 81 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.NGSS EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 82 V.sub.H GLEWIGDMYPDNGSSSYNQKFRERVTITRDTSTSTAYLELSSLRSE DTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.NGSS DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 83 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.gr.DANADA EVQLVQSGAEVKKPGASVKVSCKASGYTFTDAYMSWVRQAPGQ 84 V.sub.H GLEWIGDMYPDNADASYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.DANADA DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 85 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.1 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 86 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.Ala.1 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 87 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPATFGQGTKVEIK 1A7.Ala.2 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 88 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.Ala.2 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 89 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTAPPTFGQGTKVEIK 1A7.Ala.3 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 90 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.Ala.3 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 91 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG ATLPPTFGQGTKVEIK 1A7.Ala.4 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 92 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.Ala.4 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 93 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HALPPTFGQGTKVEIK 1A7.Ala.5 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 94 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.Ala.5 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 95 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQA HTLPPTFGQGTKVEIK 1A7.Ala.6 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 96 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.Ala.6 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 97 VLLLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLAPTFGQGTKVEIK 1A7.Ala.7 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 98 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSVWGQGTLVTVSS 1A7.Ala.7 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 99 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQAG HTLPPTFGQGTKVEIK 1A7.Ala.8 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 100 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFSAWGQGTLVTVSS 1A7.Ala.8 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 101 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.9 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 102 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYASVWGQGTLVTVSS 1A7.Ala.9 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 103 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.10 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 104 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWAFSVWGQGTLVTVSS 1A7.Ala.10 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 105 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.11 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 106 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPAWYFSVWGQGTLVTVSS 1A7.Ala.11 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 107 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.12 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 108 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRWYFAVWGQGTLVTVSS 1A7.Ala.12 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 109 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.13 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 110 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAPRAYFSVWGQGTLVTVSS 1A7.Ala.13 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 111 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.14 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 112 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVLAARWYFSVWGQGTLVTVSS 1A7.Ala.14 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 113 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.15 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 114 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCALAPRWYFSVWGQGTLVTVSS 1A7.Ala.15 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 115 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 1A7.Ala.16 EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQ 116 V.sub.H GLEWIGDMYPDNGDSSYNQKFRERVTITRDTSTSTAYLELSSLRS EDTAVYYCVAAPRWYFSVWGQGTLVTVSS 1A7.Ala.16 DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPK 117 V.sub.L LLIYYTSRLRSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG HTLPPTFGQGTKVEIK 3C8.gr.1 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 118 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTITRDTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.1 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKAPK 119 V.sub.L LLIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHY AQFPYTFGQGTKVEIK 3C8.gr.2 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 120 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTITADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.2 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKAPK 121 V.sub.L LLIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHY AQFPYTFGQGTKVEIK 3C8.gr.3 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 122 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.3 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKAPK 123 V.sub.L LLIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHY
AQFPYTFGQGTKVEIK 3C8.gr.4 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 124 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTITADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.4 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 125 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.gr.5 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 126 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.5 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 127 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.gr.5.SG EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 128 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.5.SG DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 129 V.sub.L LIYHGTNLESGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.gr.5.EG EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 130 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.5.EG DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 131 V.sub.L LIYHGTNLEEGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.gr.5.QG EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 132 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.5.QG DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 133 V.sub.L LIYHGTNLEQGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.gr.6 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 134 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTITADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.6 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 135 V.sub.L LIYHGTNLEDGVPSRFSGSGSGADYTLTISSLQPEDFATYYCVHY AQFPYTFGQGTKVEIK 3C8.gr.7 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 136 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.7 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 137 V.sub.L LIYHGTNLEDGVPSRFSGSGSGADYTLTISSLQPEDFATYYCVHY AQFPYTFGQGTKVEIK 3C8.gr.8 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 138 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTRDTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.8 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 139 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.gr.9 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 140 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTRDTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.9 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSPKL 141 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.gr.10 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 142 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTRDTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.10 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKAFK 143 V.sub.L LLIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHY AQFPYTFGQGTKVEIK 3C8.gr.11 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 144 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTRDTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.gr.11 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKAPK 145 V.sub.L GLIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHY AQFPYTFGQGTKVEIK 3C8.A.1 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 146 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.A.1 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 147 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCAHYA QFPYTFGQGTKVEIK 3C8.A.2 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 148 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.A.2 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 149 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVAYA QFPYTFGQGTKVEIK 3C8.A.3 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 150 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.A.3 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 151 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHAA QFPYTFGQGTKVEIK 3C8.A.4 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 152 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.A.4 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 153 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA AFPYTFGQGTKVEIK 3C8.A.5 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 154 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.A.5 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 155 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QAPYTFGQGTKVEIK 3C8.A.6 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 156 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.A.6 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 157 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFAYTFGQGTKVEIK 3C8.A.7 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 158 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRLDYWGQGTLVTVSS 3C8.A.7 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 159 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPATFGQGTKVEIK 3C8.A.8 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 160 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARARLDYWGQGTLVTVSS 3C8.A.8 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 161 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.A.9 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 162 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDALDYWGQGTLVTVSS 3C8.A.9 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 163 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 3C8.A.10 EVQLVQSGAEVKKPGASVKVSCKASGYAFTNYLIEWVRQAPGQ 164 V.sub.H GLEWIGVINPGSGDTYYSEKFKGRVTLTADTSTSTAYLELSSLRSE DTAVYYCARDRADYWGQGTLVTVSS 3C8.A.10 DIQMTQSPSSLSASVGDRVTITCHASQDISSYIVWYQQKPGKSFKG 165 V.sub.L LIYHGTNLEDGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCVHYA QFPYTFGQGTKVEIK 1D2.gr.1 EVQLVESGPGLVKPSETLSLTCTVSGFSLTDYGVLWIRQPPGKGL 166 V.sub.H EWIGMIWSGGTTDYNAAFISRVTISVDTSKNQFSLKLSSVTAADT AVYYCVREEMDYWGQGTLVTVSS 1D2.gr.1 DIQMTQSPSSLSASVGDRVTITCRASQDISNFLNWYQQKPGKAPK 167 V.sub.L LLIYYTSRLHSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG NTLPWTFGQGTKVEIK 1D2.gr.2 EVQLVESGPGLVKPSETLSLTCTVSGFSLTDYGVLWIRQPPGKGL 168 V.sub.H EWIGMIWSGGTTDYNAAFISRVTISKDTSKNQVSLKLSSVTAADT AVYYCVREEMDYWGQGTLVTVSS 1D2.gr.2 DIQMTQSPSSLSASVGDRVTITCRASQDISNFLNWYQQKPGKAPK 169 V.sub.L LLIYYTSRLHSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG NTLPWTFGQGTKVEIK 1D2.gr.3 EVQLVESGPGLVKPSETLSLTCTVSGFSLTDYGVLWVRQPPGKGL 170 V.sub.H EWLGMIWSGGTTDYNAAFISRLTISKDTSKNQVSLKLSSVTAADT AVYYCVREEMDYWGQGTLVTVSS 1D2.gr.3 DIQMTQSPSSLSASVGDRVTITCRASQDISNFLNWYQQKPGKAPK 171 V.sub.L LLIYYTSRLHSGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQQG NTLPWTFGQGTKVEIK CON1 (1A7) X.sub.1X.sub.2YMS, wherein X.sub.1 is D or E, and X.sub.2 is S or A 172 HVR-H1 CON1 (1A7) DMYPDX.sub.1X.sub.2X.sub.3X.sub.4SYNQKFRE, wherein X.sub.1 is N or S, X.sub.1 is A or G, 173 HVR-H2 X.sub.3 is D or S, and X.sub.4 is A or S CON1 (1A7) APRWX.sub.1X.sub.2X.sub.3X.sub.4, wherein X.sub.1 is Y or A, X.sub.2 is A or F, X.sub.3 is 174 HVR-H3 S or A, and X.sub.4 is A or V. CON1 (1A7) QX.sub.1X.sub.2X.sub.3X.sub.4X.sub.5X.sub.6X.sub.7T, wherein X.sub.1 is A or Q, X.sub.2 is A or G, X.sub.3 is 175 HVR-L3 A or H, X.sub.4 is A or T, X.sub.5 is A or L, X.sub.6 is A or P, and X.sub.7 is A or P. CON2 (3C8) VINPGSGDX.sub.1YYSEKFKG, wherein X.sub.1 is T, A or Q. 176 HVR-H2 CON2 (3C8) HGTNLEX.sub.1, wherein X.sub.1 is S, E, or Q. 177 HVR-L2 CON2 (3C8) X.sub.1X.sub.2YAQFPYX.sub.3, wherein X.sub.1 is V or A, X.sub.2 is H or A, and X.sub.3 178 HVR-L3 is Y or A. 1A7 V.sub.L DIQMTQTTSSLSASLGDRVTISCRASQDISNYLNWYQQKPDGTVKLL 179 IYYTSRLRSGVPSRFSGSGSGKDYFLTISNLEQEDVAAYFCQQGHTLP PTFGGGTKLEIK 1A7 V.sub.H EVQLQQSGPELVKPGASVKISCKASGYTFTDSYMSWVKQSHGKTLE 180 WIGDMYPDNGDSSYNQKFREKVTLTVDKSSTTAYMEFRSLTSEDSA VYYCVLAPRWYFSVWGTGTTVTVSS 3C8 V.sub.L DILMTQSPSSMSVSLGDTVSITCHASQDISSYIVWLQQKPGKSFRGLI 181 YHGTNLEDGIPSRFSGSGSGADYSLTISSLESEDFADYYCVHYAQFPY TFGGGTKLEIK 3C8 V.sub.H QVQLQQSGAELVRPGTSVKVSCKASGYAFTNYLIEWVKQRPGQGLE 182 WIGVINPGSGDTYYSEKFKGKVTLTADKSSSTAYMQLSSLTSEDSAV YFCARDRLDYWGQGTTLTVSS 1A7.gr.5' EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQGL 225 V.sub.H EWIGDMYPDNGDSSYNQKFRERVTLTVDTSTSTAYLELSSLRSEDTA VYYCVLAPRWYFSVWGQGTLVTVSS 1A7.gr.7' EVQLVQSGAEVKKPGASVKVSCKASGYTFTDSYMSWVRQAPGQGL 226 V.sub.H EWIGDMYPDNGDSSYNQKFRERVTLTVDTSTSTAYLELSSLRSEDTA VYYCVLAPRWYFSVWGQGTLVTVSS
[0222] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in U.S. Pat. No. 7,550,140. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain comprising the sequence of
TABLE-US-00004 (SEQ ID NO: 183) EVQLVESGGGLVQPGGSLRLSCAASGFTFSNYTMNWVRQAPGKGLEWVSAI SGSGGSTYYADSVKGRFTISRDNSKNTLYLQMNSLRAEDTAVYYCAKDRYS QVHYALDYWGQGTLVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDY FPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYIC NVNHKPSNTKVDKRVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTL MISRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRV VSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPREPQVYTLPP SREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSF FLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
and/or a light chain comprising the sequence of DIVMTQSPDS LPVTPGEPASISCRSSQSLLHSNGYNYLDWYLQKAGQSPQLLIYLGSNRASGV PDRFSGSGSGTDFTLKISRVEAEDVGVYYCQQYYNHPTTFGQGTKLEIKRTVAAPSVFIFPPS DEQLKSTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDS KDSTYS LS STLTLS KADYEKHKVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO:184). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody 008 as described in U.S. Pat. No. 7,550,140. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody 008 as described in U.S. Pat. No. 7,550,140.
[0223] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in U.S. Pat. No. 7,550,140. In some embodiments, the agonist anti-human OX40 antibody comprises the sequence of MAEVQLVESGGGLVQPGGSLRLSCAASGFTFSNYTMNWVRQAPGKGLEWVSAISGSGGST YYADSVKGRFTISRDNSKNTLYLQMNSLRAEDTAVYYCAKDRYSQVHYALDYWGQGT LVTVLEGTGGSGGTGSGTGTSELDIQMTQSPDSLPVTPGEPASISCRSSQSLLHSNGYNYL DWYLQKAGQSPQLLIYLGSNRASGVPDRFSGSGSGTDFTLKISRVEAEDVGVYYCQQY YNHPTTFGQGTKLEIKRAA (SEQ ID NO:185). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody SCO2008 as described in U.S. Pat. No. 5,755,0140. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody SC02008 as described in U.S. Pat. No. 7,550,140.
[0224] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in U.S. Pat. NO. 7,550,140. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain comprising the sequence of EVQLVESGGGLVHPGGSLRLSCAGSGFTFSSYAMHWVRQAPGKGLEWVSAIGTGGGTYYA DSVMGRFTISRDNSKNTLYLQMNSLRAEDTAVYYCARYDNVMGLYWFDYWGQGTLVTVS SASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSG LYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCDKTHTCPPCPAPELLGGPSVF LFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRV VSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTIS KAKGQPREPQVYTLPPSREEMTKNQV SLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSC SVMHEALHNHYTQKSLSLSPGK (SEQ ID NO:186) and/or a light chain comprising the sequence of EIVLTQSPALSLSPGERATLSCRASQSVSSYLAWYQQKPGQAPRLLIYDASNRATGIPARFS GSGSGTDFTLTISSLEPEDFAVYYCQQRSNWPPAFGGGTKVEIKRTVAAPS VFIFPPSDEQLKS GTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEK HKVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO:187). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody 023 as described in U.S. Pat. No. 7,550,140. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody 023 as described in U.S. Pat. No. 7,550,140.
[0225] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in U.S. Pat. No. 7,960,515. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of EVQLVESGGGLVQPGGSLRLSCAASGFTFSSYSMNWVRQAPGKGLEWVSYISSSSSTIDYAD SVKGRFTISRDNAKNSLYLQMNSLRDEDTAVYYCARESGWYLFDYWGQGTLVTVSS (SEQ ID NO:188) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCRASQGISSWLAWYQQKPEKAPKSLIYAASSLQSGVPSRFS GSGSGTDFTLTISSLQPEDFATYYCQQYNSYPPTFGGGTKVEIK (SEQ ID NO:189). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody 11D4 as described in U.S. Pat. No. 7,960,515. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody 11D4 as described in U.S. Pat. No. 7,960,515.
[0226] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in U.S. Pat. No. 7,960,515. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain comprising the sequence of EVQLVESGGGLVQPGRSLRLSCAASGFTFDDYAMHWVRQAPGKGLEWVSGISWNSGSIGY ADSVKGRFTISRDNAKNSLYLQMNSLRAEDTALYYCAKDQSTADYYFYYGMDVWGQGTT VTVSSASTKGPSVFPLAPCSRSTSESTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQ SSGLYSLSSVVTVPSSNFGTQTYTCNVDHKPSNTKVDKTVERKCCVECPPCPAPPVAGPSVF LFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVQFNWYVDGVEVHNAKTKPREEQFNSTFRV VSVLTVVHQDWLNGKEYKCKVSNKGLPAPIEKTISKTKGQPREPQVYTLPPSREEMTKNQV SLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPMLDSDGSFFLYSKLTVDKSRWQQGNVFS CSVMHEALHNHYTQKSLSLSPGK (SEQ ID NO:190) and/or a light chain comprising the sequence of EIVVTQSPATLSLSPGERATLSCRASQSVS SYLAWYQQKPGQAPRLLIYDASNRATGIPARFS GSGSGTDFTLTISSLEPEDFAVYYCQQRSNWPTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSG TASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKH KVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO:191). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody 18D8 as described in U.S. Pat. No. 7,960,515. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody 18D8 as described in U.S. Pat. No. 7,960,515.
[0227] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2012/027328. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGSELKKPGASVKVSCKASGYTFTDYSMHWVRQAPGQGLKWMGWINTETGEPT YADDFKGRFVFSLDTSVSTAYLQISSLKAEDTAVYYCANPYYDYVSYYAMDYVVGQGTTVT VSS (SEQ ID NO:192) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCKASQDVSTAVAWYQQKPGKAPKWYSASYLYTGVPSRF SGSGSGTDFTFTISSLQPEDIATYYCQQHYSTPRTFGQGTKLEIK (SEQ ID NO:193). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody hu106-222 as described in WO2012/027328. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody hu106-222 as described in WO2012/027328.
[0228] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2012/027328. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of EVQLVESGGGLVQPGGSLRLSCAASEYEFPSHDMSWVRQAPGKGLELVAAINSDGGSTYYP DTMERRFTISRDNAKNSLYLQMNSLRAEDTAVYYCARHYDDYYAWFAYWGQGTMVTVSS (SEQ ID NO:194) and/or a light chain variable region comprising the sequence of EIVLTQSPATLSLSPGERATLSCRASKSVSTSGYSYMHWYQQKPGQAPRLLIYLASNLESGVP ARFSGSGSGTDFTLTISSLEPEDFAVYYCQHSRELPLTFGGGTKVEIK (SEQ ID NO:195). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody Hu119-122 as described in WO2012/027328. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody Hu119-122 as described in WO2012/027328.
[0229] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2013/028231. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain comprising the sequence of MYLGLNYVFIVFLLNGVQSEVKLEESGGGLVQPGGSMKLSCAASGFTFSDAWMDWVRQSP EKGLEWVaEIRSKANNHATYYAESVNGRFTISRDDSKSSVYLQMNSLRAEDTGIYYCTWGE VFYFDYWGQGTTLTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNS GA LTSGVHTFPAVLQS SGLYSLSSVVTVPSS SLGTQTYITCNVNHKPSNTKVDKKVEPKSCDKT HTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVH NAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPREP QVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYS KLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK (SEQ ID NO:196) and/or a light chain comprising the sequence of MRPSIQFLGLLLFWLHGAQCDIQMTQSPSSLSASLGGKVTITCKS SQDINKYIAWYQHKPGK GPRLLIHYTSTLQPGIPSRFSGSGSGRDYSFSISNLEPEDIATYYCLQYDNLLTFGAGTKLELK RTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNS QESVTEQDS KDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO:197). In some embodiments, the anti-human OX40 agonist antibody comprises a heavy chain variable region comprising the sequence of MYLGLNYVFIVFLLNGVQSEVKLEESGGGLVQPGGSMKLSCAASGFTFSDAWMDWVRQSPEKG LEWVAEIRSKANNHATYYAESVNGRFTISRDDSKSSVYLQMNSLRAEDTGIYYCTWGEVFYFDY WGQGTTLTVSS (SEQ ID NO:198) and/or a light chain variable region comprising the sequence of MRPSIQFLGLLLFWLHGAQCDIQMTQSPSSLSASLGGKVTITCKSSQDINKYIAWYQHKPGKGPR LLIHYTSTLQPGIPSRFSGSGSGRDYSFSISNLEPEDIATYYCLQYDNLLTFGAGTKLELK (SEQ ID NO:199). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody Mab CH 119-43-1 as described in WO2013/028231. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody Mab CH 119-43-1 as described in WO2013/028231.
[0230] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2013038191. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of EVQLQQSGPELVKPGASVKMSCKASGYTFTS YVMHWVKQKPGQGLEWIGYINPYNDGTKY NEKFKGKATLTSDKSSSTAYMELSSLTSEDSAVYYCANYYGSSLSMDYWGQGTSVTVSS (SEQ ID NO:200) and/or a light chain variable region comprising the sequence of DIQMTQTTSSLSASLGDRVTISCRASQDISNYLNWYQQKPDGTVKLLIYYTSRLHSGVPSRFS GSGSGTDYSLTISNLEQEDIATYFCQQGNTLPWTFGGGTKLEIKR (SEQ ID NO:201). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 20E5 as described in WO2013038191. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 20E5 as described in WO2013038191.
[0231] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2013038191. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of EVQLQQSGPELVKPGASVKISCKTSGYTFKDYTMHWVKQSHGKSLEWIGGIYPNNGGSTYN QNFKDKATLTVDKSSSTAYMEFRSLTSEDSAVYYCARMGYHGPHLDFDVWGAGTTVTVSP (SEQ ID NO:202) and/or a light chain variable region comprising the sequence of DIVMTQSHKFMSTSLGDRVSITCKASQDVGAAVaWYQQKPGQSPKLLIYWASTRHTGVPDR FTGGGSGTDFTLTISNVQSEDLTDYFCQQYINYPLTFGGGTKLEIKR (SEQ ID NO:203). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 12H3 as described in WO2013038191. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 12H3 as described in WO2013038191.
[0232] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYVMHWVRQAPGQRLEWMGYINPYNDGTK YNEKFKGRVTITSDTSASTAYMELSSLRSEDTAVYYCANYYGSSLSMDYWGQGTLVTVSS (SEQ ID NO:204) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPKLLIYYTSRLHSGVPSRFS GSGSGTDYTLTISSLQPEDFATYYCQQGNTLPWTFGQGTKVEIKR (SEQ ID NO:205). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 20E5 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 20E5 as described in WO2014148895A1.
[0233] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYVMHWVRQAPGQRLEWMGYINPYNDGTK YNEKFKGRVTITSDTSASTAYMELSSLRSEDTAVYYCANYYGSSLSMDYWGQGTLVTVSS (SEQ ID NO:204) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAVKLLIYYTSRLHSGVPSRFS GSGSGTDYTLTISSLQPEDFATYFCQQGNTLPWTFGQGTKVEIKR (SEQ ID NO:206). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 20E5 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 20E5 as described in WO2014148895A1.
[0234] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYVMHWVRQAPGQRLEWIGYINPYNDGTK YNEKFKGRATITSDTSASTAYMELSSLRSEDTAVYYCANYYGSSLSMDYWGQGTLVTVSS (SEQ ID NO:207) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPKLLIYYTSRLHSGVPSRFS GSGSGTDYTLTISSLQPEDFATYYCQQGNTLPWTFGQGTKVEIKR (SEQ ID NO:205). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 20E5 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 20E5 as described in WO2014148895A1.
[0235] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYVMHWVRQAPGQRLEWIGYINPYNDGTK YNEKFKGRATITSDTSASTAYMELSSLRSEDTAVYYCANYYGSSLSMDYWGQGTLVTVSS (SEQ ID NO:207) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAVKLLIYYTSRLHSGVPSRFS GSGSGTDYTLTISSLQPEDFATYFCQQGNTLPWTFGQGTKVEIKR (SEQ ID NO:206). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 20E5 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 20E5 as described in WO2014148895A1.
[0236] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYVMHWVRQAPGQRLEWIGYINPYNDGTK YNEKFKGRATLTSDKSASTAYMELSSLRSEDTAVYYCANYYGSSLSMDYWGQGTLVTVSS (SEQ ID NO:208) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAPKLLIYYTSRLHSGVPSRFS GSGSGTDYTLTISSLQPEDFATYYCQQGNTLPWTFGQGTKVEIKR (SEQ ID NO:205). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 20E5 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 20E5 as described in WO2014148895A1.
[0237] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGASVKVSCKASGYTFTSYVMHWVRQAPGQRLEWIGYINPYNDGTK YNEKFKGRATLTSDKSASTAYMELSSLRSEDTAVYYCANYYGSSLSMDYWGQGTLVTVSS (SEQ ID NO:208) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCRASQDISNYLNWYQQKPGKAVKLLIYYTSRLHSGVPSRFS GSGSGTDYTLTISSLQPEDFATYFCQQGNTLPWTFGQGTKVEIKR (SEQ ID NO:206). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 20E5 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 20E5 as described in WO2014148895A1.
[0238] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGSSVKVSCKASGYTFKDYTMHWVRQAPGQGLEWMGGIYPNNGGST YNQNFKDRVTITADKSTSTAYMELSSLRSEDTAVYYCARMGYHGPHLDFDVWGQGTTVTV SS (SEQ ID NO:209) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCKASQDVGAAVaWYQQKPGKAPKLLIYWASTRHTGVPSRF SGSGSGTDFTLTISSLQPEDFATYYCQQYINYPLTFGGGTKVEIKR (SEQ ID NO:210). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone clone 12H3 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 12H3 as described in WO2014148895A1.
[0239] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGSSVKVSCKASGYTFKDYTMHWVRQAPGQGLEWMGGIYPNNGGST YNQNFKDRVTITADKSTSTAYMELSSLRSEDTAVYYCARMGYHGPHLDFDVWGQGTTVTV SS (SEQ ID NO:209) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCKASQDVGAAVaWYQQKPGKAPKWYWASTRHTGVPDR FSGGGSGTDFTLTISSLQPEDFATYYCQQYINYPLTFGGGTKVEIKR (SEQ ID NO:211). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 12H3 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 12H3 as described in WO2014148895A1.
[0240] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGSSVKVSCKASGYTFKDYTMHWVRQAPGQGLEWIGGIYPNNGGSTY NQNFKDRVTLTADKSTSTAYMELSSLRSEDTAVYYCARMGYHGPHLDFDVWGQGTTVTVS S (SEQ ID NO:212) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCKASQDVGAAVaWYQQKPGKAPKWYWASTRHTGVPSRF SGSGSGTDFTLTISSLQPEDFATYYCQQYINYPLTFGGGTKVEIKR (SEQ ID NO:210). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 12H3 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 12H3 as described in WO2014148895A1.
[0241] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGSSVKVSCKASGYTFKDYTMHWVRQAPGQGLEWIGGIYPNNGGSTY NQNFKDRVTLTADKSTSTAYMELSSLRSEDTAVYYCARMGYHGPHLDFDVWGQGTTVTVS S (SEQ ID NO:212) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCKASQDVGAAVaWYQQKPGKAPKWYWASTRHTGVPDR FSGGGSGTDFTLTISSLQPEDFATYYCQQYINYPLTFGGGTKVEIKR (SEQ ID NO:211). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 12H3 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 12H3 as described in WO2014148895A1.
[0242] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGSSVKVSCKASGYTFKDYTMHWVRQAPGQGLEWIGGIYPNNGGSTY NQNFKDRATLTVDKSTSTAYMELSSLRSEDTAVYYCARMGYHGPHLDFDVWGQGTTVTVS S (SEQ ID NO:213) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCKASQDVGAAVaWYQQKPGKAPKWYWASTRHTGVPSRF SGSGSGTDFTLTISSLQPEDFATYYCQQYINYPLTFGGGTKVEIKR (SEQ ID NO:210). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 12H3 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 12H3 as described in WO2014148895A1.
[0243] In some embodiments, the agonist anti-human OX40 antibody is an anti-OX40 antibody described in WO2014148895A1. In some embodiments, the agonist anti-human OX40 antibody comprises a heavy chain variable region comprising the sequence of QVQLVQSGAEVKKPGSSVKVSCKASGYTFKDYTMHWVRQAPGQGLEWIGGIYPNNGGSTY NQNFKDRATLTVDKSTSTAYMELSSLRSEDTAVYYCARMGYHGPHLDFDVWGQGTTVTVS S (SEQ ID NO:213) and/or a light chain variable region comprising the sequence of DIQMTQSPSSLSASVGDRVTITCKASQDVGAAVaWYQQKPGKAPKWYWASTRHTGVPDR FSGGGSGTDFTLTISSLQPEDFATYYCQQYINYPLTFGGGTKVEIKR (SEQ ID NO:211). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody clone 12H3 as described in WO2014148895A1. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody clone 12H3 as described in WO2014148895A1.
[0244] In some embodiments, the agonist anti-human OX40 antibody is L106 BD (Pharmingen Product #340420). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody L106 (BD Pharmingen Pduct #340420). In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody L106 (BD Pharmingen Product #340420).
[0245] In some embodiments, the agonist anti-human OX40 antibody is ACT35 (Santa Cruz Biotechnology, Catalog #20073). In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody ACT35 (Santa Cruz Biotechnology, Catalog #20073). In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody ACT35 (Santa Cruz Biotechnology, Catalog #20073).
[0246] In some embodiments, the agonist anti-human OX40 antibody is MEDI6469. In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody MEDI6469. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody MEDI6469.
[0247] In some embodiments, the agonist anti-human OX40 antibody is MEDI0562. In some embodiments, the antibody comprises at least one, two, three, four, five or six hypervariable region (HVR) sequences of antibody MEDI0562. In some embodiments, the antibody comprises a heavy chain variable region sequence and/or a light chain variable region sequence of antibody MEDI0562.
[0248] Other OX40 Agonists
[0249] OX40 agonists useful for the methods described herein are in no way intended to be limited to antibodies. Non-antibody OX40 agonists are contemplated and well known in the art.
[0250] As described above, OX40L (also known as CD134L) serves as a ligand for OX40. As such, agonists that present part or all of OX40L may serve as OX40 agonists. In some embodiments, an OX40 agonist may include one or more extracellular domains of OX40L. Examples of extracellular domains of OX40L may include OX40-binding domains. In some embodiments, an OX40 agonist may be a soluble form of OX40L that includes one or more extracellular domains of OX40L but lacks other, insoluble domains of the protein, e.g., transmembrane domains. In some embodiments, an OX40 agonist is a soluble protein that includes one or more extracellular domains of OX40L able to bind OX40L. In some embodiments, an OX40 agonist may be linked to another protein domain, e.g., to increase its effectiveness, half-life, or other desired characteristics. In some embodiments, an OX40 agonist may include one or more extracellular domains of OX40L linked to an immunoglobulin Fc domain.
[0251] In some embodiments, an OX40 agonist may be any one of the OX40 agonists described in U.S. Pat. No. 7,696,175.
[0252] In some embodiments, an OX40 agonist may be an oligomeric or multimeric molecule. For example, an OX40 agonist may contain one or more domains (e.g., a leucine zipper domain) that allows proteins to oligomerize. In some embodiments, an OX40 agonist may include one or more extracellular domains of OX40L linked to one or more leucine zipper domains.
[0253] In some embodiments, an OX40 agonist may be any one of the OX40 agonists described in European Patent No. EP0672141 B1.
[0254] In some embodiments, an OX40 agonist may be a trimeric OX40L fusion protein. For example, an OX40 agonist may include one or more extracellular domains of OX40L linked to an immunoglobulin Fc domain and a trimerization domain (including without limitation an isoleucine zipper domain).
[0255] In some embodiments, an OX40 agonist may be any one of the OX40 agonists described in International Publication No. WO2006/121810. In some embodiments, the OX40 agonist is MEDI6383.
[0256] In a further aspect, an anti-OX40 agonist and/or antibody according to any of the above embodiments may incorporate any of the features, singly or in combination, as described below.
[0257] 1. Antibody Affinity
[0258] In certain embodiments, an antibody provided herein has a dissociation constant (Kd) of .ltoreq.1.mu.M, .ltoreq.100 nM, .ltoreq.10 nM, .ltoreq.1 nM, .ltoreq.0.1 nM, .ltoreq.0.01 nM, or .ltoreq.0.001 nM (e.g. 10-8 M or less, e.g. from 10-8 M to 10-13 M, e.g., from 10-9 M to 10-13 M).
[0259] In one embodiment, Kd is measured by a radiolabeled antigen binding assay (RIA). In one embodiment, an RIA is performed with the Fab version of an antibody of interest and its antigen. For example, solution binding affinity of Fabs for antigen is measured by equilibrating Fab with a minimal concentration of (125I)-labeled antigen in the presence of a titration series of unlabeled antigen, then capturing bound antigen with an anti-Fab antibody-coated plate (see, e.g., Chen et al., J. Mol. Biol. 293:865-881(1999)). To establish conditions for the assay, MICROTITER.RTM. multi-well plates (Thermo Scientific) are coated overnight with 5 .mu.g/ml of a capturing anti-Fab antibody (Cappel Labs) in 50 mM sodium carbonate (pH 9.6), and subsequently blocked with 2% (w/v) bovine serum albumin in PBS for two to five hours at room temperature (approximately 23.degree. C.). In a non-adsorbent plate (Nunc #269620), 100 pM or 26 pM [125I]-antigen are mixed with serial dilutions of a Fab of interest (e.g., consistent with assessment of the anti-VEGF antibody, Fab-12, in Presta et al., Cancer Res. 57:4593-4599 (1997)). The Fab of interest is then incubated overnight; however, the incubation may continue for a longer period (e.g., about 65 hours) to ensure that equilibrium is reached. Thereafter, the mixtures are transferred to the capture plate for incubation at room temperature (e.g., for one hour). The solution is then removed and the plate washed eight times with 0.1% polysorbate 20 (TWEEN-20.RTM.) in PBS. When the plates have dried, 150 .mu.l/well of scintillant (MICROSCINT-20 .TM.; Packard) is added, and the plates are counted on a TOPCOUNT.TM. gamma counter (Packard) for ten minutes. Concentrations of each Fab that give less than or equal to 20% of maximal binding are chosen for use in competitive binding assays.
[0260] According to another embodiment, Kd is measured using a BIACORE.RTM. surface plasmon resonance assay. For example, an assay using a BIACORE.RTM.-2000 or a BIACORE .RTM.-3000 (BIAcore, Inc., Piscataway, N.J.) is performed at 25.degree. C. with immobilized antigen CM5 chips at .about.10 response units (RU). In one embodiment, carboxymethylated dextran biosensor chips (CM5, BIACORE, Inc.) are activated with N-ethyl-N'-(3-dimethylaminopropyl)-carbodiimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS) according to the supplier's instructions. Antigen is diluted with 10 mM sodium acetate, pH 4.8, to 5 .mu.g/ml (.about.0.2 .mu.M) before injection at a flow rate of 5 .mu.l/minute to achieve approximately 10 response units (RU) of coupled protein. Following the injection of antigen, 1 M ethanolamine is injected to block unreacted groups. For kinetics measurements, two-fold serial dilutions of Fab (0.78 nM to 500 nM) are injected in PBS with 0.05% polysorbate 20 (TWEEN-20.TM.) surfactant (PBST) at 25.degree. C. at a flow rate of approximately 25 .mu.l/min. Association rates (kon) and dissociation rates (koff) are calculated using a simple one-to-one Langmuir binding model (BIACORE.RTM. Evaluation Software version 3.2) by simultaneously fitting the association and dissociation sensorgrams. The equilibrium dissociation constant (Kd) is calculated as the ratio koff/kon. See, e.g., Chen et al., J. Mol. Biol. 293:865-881 (1999). If the on-rate exceeds 106 M-1 s-1 by the surface plasmon resonance assay above, then the on-rate can be determined by using a fluorescent quenching technique that measures the increase or decrease in fluorescence emission intensity (excitation=295 nm; emission=340 nm, 16 nm band-pass) at 25oC of a 20 nM anti-antigen antibody (Fab form) in PBS, pH 7.2, in the presence of increasing concentrations of antigen as measured in a spectrometer, such as a stop-flow equipped spectrophometer (Aviv Instruments) or a 8000-series SLM-AMINCO.TM. spectrophotometer (ThermoSpectronic) with a stirred cuvette.
[0261] 2. Antibody Fragments
[0262] In certain embodiments, an antibody provided herein is an antibody fragment. Antibody fragments include, but are not limited to, Fab, Fab', Fab'-SH, F(ab')2, Fv, and scFv fragments, and other fragments described below. For a review of certain antibody fragments, see Hudson et al. Nat. Med. 9:129-134 (2003). For a review of scFv fragments, see, e.g., Pluckthun, in The Pharmacology of Monoclonal Antibodies, vol. 113, Rosenburg and Moore eds., (Springer-Verlag, New York), pp. 269-315 (1994); see also WO 93/16185; and U.S. Pat. Nos. 5,571,894 and 5,587,458. For discussion of Fab and F(ab')2 fragments comprising salvage receptor binding epitope residues and having increased in vivo half-life, see U.S. Pat. No. 5,869,046.
[0263] Diabodies are antibody fragments with two antigen-binding sites that may be bivalent or bispecific. See, for example, EP 404,097; WO 1993/01161; Hudson et al., Nat. Med. 9:129-134 (2003); and Hollinger et al., Proc. Natl. Acad. Sci. USA 90: 6444-6448 (1993). Triabodies and tetrabodies are also described in Hudson et al., Nat. Med. 9:129-134 (2003).
[0264] Single-domain antibodies are antibody fragments comprising all or a portion of the heavy chain variable domain or all or a portion of the light chain variable domain of an antibody. In certain embodiments, a single-domain antibody is a human single-domain antibody (Domantis, Inc., Waltham, Mass.; see, e.g., U.S. Pat. No. 6,248,516 B1).
[0265] Antibody fragments can be made by various techniques, including but not limited to proteolytic digestion of an intact antibody as well as production by recombinant host cells (e.g. E. coli or phage), as described herein.
[0266] 3. Chimeric and Humanized Antibodies
[0267] In certain embodiments, an antibody provided herein is a chimeric antibody. Certain chimeric antibodies are described, e.g., in U.S. Pat. No. 4,816,567; and Morrison et al., Proc. Natl. Acad. Sci. USA, 81:6851-6855 (1984)). In one example, a chimeric antibody comprises a non-human variable region (e.g., a variable region derived from a mouse, rat, hamster, rabbit, or non-human primate, such as a monkey) and a human constant region. In a further example, a chimeric antibody is a "class switched" antibody in which the class or subclass has been changed from that of the parent antibody. Chimeric antibodies include antigen-binding fragments thereof.
[0268] In certain embodiments, a chimeric antibody is a humanized antibody. Typically, a non-human antibody is humanized to reduce immunogenicity to humans, while retaining the specificity and affinity of the parental non-human antibody. Generally, a humanized antibody comprises one or more variable domains in which HVRs, e.g., CDRs, (or portions thereof) are derived from a non-human antibody, and FRs (or portions thereof) are derived from human antibody sequences. A humanized antibody optionally will also comprise at least a portion of a human constant region. In some embodiments, some FR residues in a humanized antibody are substituted with corresponding residues from a non-human antibody (e.g., the antibody from which the HVR residues are derived), e.g., to restore or improve antibody specificity or affinity.
[0269] Humanized antibodies and methods of making them are reviewed, e.g., in Almagro and Fransson, Front. Biosci. 13:1619-1633 (2008), and are further described, e.g., in Riechmann et al., Nature 332:323-329 (1988); Queen et al., Proc. Nat'l Acad. Sci. USA 86:10029-10033 (1989); U.S. Pat. Nos. 5, 821,337, 7,527,791, 6,982,321, and 7,087,409; Kashmiri et al., Methods 36:25-34 (2005) (describing specificity determining region (SDR) grafting); Padlan, Mol. Immunol. 28:489-498 (1991) (describing "resurfacing"); Dall'Acqua et al., Methods 36:43-60 (2005) (describing "FR shuffling"); and Osbourn et al., Methods 36:61-68 (2005) and Klimka et al., Br. J. Cancer, 83:252-260 (2000) (describing the "guided selection" approach to FR shuffling).
[0270] Human framework regions that may be used for humanization include but are not limited to: framework regions selected using the "best-fit" method (see, e.g., Sims et al. J. Immunol. 151:2296 (1993)); framework regions derived from the consensus sequence of human antibodies of a particular subgroup of light or heavy chain variable regions (see, e.g., Carter et al. Proc. Natl. Acad. Sci. USA, 89:4285 (1992); and Presta et al. J. Immunol., 151:2623 (1993)); human mature (somatically mutated) framework regions or human germline framework regions (see, e.g., Almagro and Fransson, Front. Biosci. 13:1619-1633 (2008)); and framework regions derived from screening FR libraries (see, e.g., Baca et al., J. Biol. Chem. 272:10678-10684 (1997) and Rosok et al., J. Biol. Chem. 271:22611-22618 (1996)).
[0271] 4. Human Antibodies
[0272] In certain embodiments, an antibody provided herein is a human antibody. Human antibodies can be produced using various techniques known in the art. Human antibodies are described generally in van Dijk and van de Winkel, Curr. Opin. Pharmacol. 5: 368-74 (2001) and Lonberg, Curr. Opin. Immunol. 20:450-459 (2008).
[0273] Human antibodies may be prepared by administering an immunogen to a transgenic animal that has been modified to produce intact human antibodies or intact antibodies with human variable regions in response to antigenic challenge. Such animals typically contain all or a portion of the human immunoglobulin loci, which replace the endogenous immunoglobulin loci, or which are present extrachromosomally or integrated randomly into the animal's chromosomes. In such transgenic mice, the endogenous immunoglobulin loci have generally been inactivated. For review of methods for obtaining human antibodies from transgenic animals, see Lonberg, Nat. Biotech. 23:1117-1125 (2005). See also, e.g., U.S. Pat. Nos. 6,075,181 and 6,150,584 describing XENOMOUSE.TM. technology; U.S. Pat. No. 5,770,429 describing HuMab.RTM. technology; U.S. Pat. No. 7,041,870 describing K-M MOUSE.RTM. technology, and U.S. Patent Application Publication No. US 2007/0061900, describing VelociMouse.RTM. technology). Human variable regions from intact antibodies generated by such animals may be further modified, e.g., by combining with a different human constant region.
[0274] Human antibodies can also be made by hybridoma-based methods. Human myeloma and mouse-human heteromyeloma cell lines for the production of human monoclonal antibodies have been described. (See, e.g., Kozbor J. Immunol., 133: 3001 (1984); Brodeur et al., Monoclonal Antibody Production Techniques and Applications, pp. 51-63 (Marcel Dekker, Inc., New York, 1987); and Boerner et al., J. Immunol., 147: 86 (1991).) Human antibodies generated via human B-cell hybridoma technology are also described in Li et al., Proc. Natl. Acad. Sci. USA, 103:3557-3562 (2006). Additional methods include those described, for example, in U.S. Pat. No. 7,189,826 (describing production of monoclonal human IgM antibodies from hybridoma cell lines) and Ni, Xiandai Mianyixue, 26(4):265-268 (2006) (describing human-human hybridomas). Human hybridoma technology (Trioma technology) is also described in Vollmers and Brandlein, Histology and Histopathology, 20(3):927-937 (2005) and Vollmers and Brandlein, Methods and Findings in Experimental and Clinical Pharmacology, 27(3):185-91 (2005).
[0275] Human antibodies may also be generated by isolating Fv clone variable domain sequences selected from human-derived phage display libraries. Such variable domain sequences may then be combined with a desired human constant domain. Techniques for selecting human antibodies from antibody libraries are described below.
[0276] 5. Library-Derived Antibodies
[0277] Antibodies of the invention may be isolated by screening combinatorial libraries for antibodies with the desired activity or activities. For example, a variety of methods are known in the art for generating phage display libraries and screening such libraries for antibodies possessing the desired binding characteristics. Such methods are reviewed, e.g., in Hoogenboom et al. in Methods in Molecular Biology 178:1-37 (O'Brien et al., ed., Human Press, Totowa, N.J., 2001) and further described, e.g., in the McCafferty et al., Nature 348:552-554; Clackson et al., Nature 352: 624-628 (1991); Marks et al., J. Mol. Biol. 222: 581-597 (1992); Marks and Bradbury, in Methods in Molecular Biology 248:161-175 (Lo, ed., Human Press, Totowa, N.J., 2003); Sidhu et al., J. Mol. Biol. 338(2): 299-310 (2004); Lee et al., J. Mol. Biol. 340(5): 1073-1093 (2004); Fellouse, Proc. Natl. Acad. Sci. USA 101(34): 12467-12472 (2004); and Lee et al., J. Immunol. Methods 284(1-2): 119-132(2004).
[0278] In certain phage display methods, repertoires of VH and VL genes are separately cloned by polymerase chain reaction (PCR) and recombined randomly in phage libraries, which can then be screened for antigen-binding phage as described in Winter et al., Ann. Rev. Immunol., 12: 433-455 (1994). Phage typically display antibody fragments, either as single-chain Fv (scFv) fragments or as Fab fragments. Libraries from immunized sources provide high-affinity antibodies to the immunogen without the requirement of constructing hybridomas. Alternatively, the naive repertoire can be cloned (e.g., from human) to provide a single source of antibodies to a wide range of non-self and also self antigens without any immunization as described by Griffiths et al., EMBO J, 12: 725-734 (1993). Finally, naive libraries can also be made synthetically by cloning unrearranged V-gene segments from stem cells, and using PCR primers containing random sequence to encode the highly variable CDR3 regions and to accomplish rearrangement in vitro, as described by Hoogenboom and Winter, J. Mol. Biol., 227: 381-388 (1992). Patent publications describing human antibody phage libraries include, for example: U.S. Pat. No. 5,750,373, and US Patent Publication Nos. 2005/0079574, 2005/0119455, 2005/0266000, 2007/0117126, 2007/0160598, 2007/0237764, 2007/0292936, and 2009/0002360.
[0279] Antibodies or antibody fragments isolated from human antibody libraries are considered human antibodies or human antibody fragments herein.
[0280] 6. Multispecific Antibodies
[0281] In certain embodiments, an antibody provided herein is a multispecific antibody, e.g. a bispecific antibody. Multispecific antibodies are monoclonal antibodies that have binding specificities for at least two different sites. In certain embodiments, one of the binding specificities is for OX40 and the other is for any other antigen. In certain embodiments, bispecific antibodies may bind to two different epitopes of OX40. Bispecific antibodies may also be used to localize cytotoxic agents to cells which express OX40. Bispecific antibodies can be prepared as full length antibodies or antibody fragments.
[0282] Techniques for making multispecific antibodies include, but are not limited to, recombinant co-expression of two immunoglobulin heavy chain-light chain pairs having different specificities (see Milstein and Cuello, Nature 305: 537 (1983)), WO 93/08829, and Traunecker et al., EMBO J. 10: 3655 (1991)), and "knob-in-hole" engineering (see, e.g., U.S. Pat. No. 5,731,168). Multi-specific antibodies may also be made by engineering electrostatic steering effects for making antibody Fc-heterodimeric molecules (WO 2009/089004A1); cross-linking two or more antibodies or fragments (see, e.g., U.S. Pat. No. 4,676,980, and Brennan et al., Science, 229: 81 (1985)); using leucine zippers to produce bi-specific antibodies (see, e.g., Kostelny et al., J. Immunol., 148(5):1547-1553 (1992)); using "diabody" technology for making bispecific antibody fragments (see, e.g., Hollinger et al., Proc. Natl. Acad. Sci. USA, 90:6444-6448 (1993)); and using single-chain Fv (sFv) dimers (see,e.g. Gruber et al., J. Immunol., 152:5368 (1994)); and preparing trispecific antibodies as described, e.g., in Tutt et al. J. Immunol. 147: 60 (1991).
[0283] Engineered antibodies with three or more functional antigen binding sites, including "Octopus antibodies," are also included herein (see, e.g. US 2006/0025576A1).
[0284] The antibody or fragment herein also includes a "Dual Acting FAb" or "DAF" comprising an antigen binding site that binds to OX40 as well as another, different antigen (see, US 2008/0069820, for example).
[0285] 7. Antibody Variants
[0286] In certain embodiments, amino acid sequence variants of the antibodies provided herein are contemplated. For example, it may be desirable to improve the binding affinity and/or other biological properties of the antibody. Amino acid sequence variants of an antibody may be prepared by introducing appropriate modifications into the nucleotide sequence encoding the antibody, or by peptide synthesis. Such modifications include, for example, deletions from, and/or insertions into and/or substitutions of residues within the amino acid sequences of the antibody. Any combination of deletion, insertion, and substitution can be made to arrive at the final construct, provided that the final construct possesses the desired characteristics, e.g., antigen-binding.
[0287] a) Substitution, Insertion, and Deletion Variants
[0288] In certain embodiments, antibody variants having one or more amino acid substitutions are provided. Sites of interest for substitutional mutagenesis include the HVRs and FRs. Conservative substitutions are shown in Table A under the heading of "preferred substitutions." More substantial changes are provided in Table A under the heading of "exemplary substitutions," and as further described below in reference to amino acid side chain classes. Amino acid substitutions may be introduced into an antibody of interest and the products screened for a desired activity, e.g., retained/improved antigen binding, decreased immunogenicity, or improved ADCC or CDC.
TABLE-US-00005 TABLE A Original Exemplary Preferred Residue Substitutions Substitutions Ala (A) Val; Leu; Ile Val Arg (R) Lys; Gln; Asn Lys Asn (N) Gln; His; Asp, Lys; Arg Gln Asp (D) Glu; Asn Glu Cys (C) Ser; Ala Ser Gln (Q) Asn; Glu Asn Glu (E) Asp; Gln Asp Gly (G) Ala Ala His (H) Asn; Gln; Lys; Arg Arg Ile (I) Leu; Val; Met; Ala; Phe; Norleucine Leu Leu (L) Norleucine; Ile; Val; Met; Ala; Phe Ile Lys (K) Arg; Gln; Asn Arg Met (M) Leu; Phe; Ile Leu Phe (F) Trp; Leu; Val; Ile; Ala; Tyr Tyr Pro (P) Ala Ala Ser (S) Thr Thr Thr (T) Val; Ser Ser Trp (W) Tyr; Phe Tyr Tyr (Y) Trp; Phe; Thr; Ser Phe Val (V) Ile; Leu; Met; Phe; Ala; Norleucine Leu
[0289] Amino acids may be grouped according to common side-chain properties:
[0290] (1) hydrophobic: Norleucine, Met, Ala, Val, Leu, Ile;
[0291] (2) neutral hydrophilic: Cys, Ser, Thr, Asn, Gln;
[0292] (3) acidic: Asp, Glu;
[0293] (4) basic: His, Lys, Arg;
[0294] (5) residues that influence chain orientation: Gly, Pro;
[0295] (6) aromatic: Trp, Tyr, Phe.
[0296] Non-conservative substitutions will entail exchanging a member of one of these classes for another class.
[0297] One type of substitutional variant involves substituting one or more hypervariable region residues of a parent antibody (e.g. a humanized or human antibody). Generally, the resulting variant(s) selected for further study will have modifications (e.g., improvements) in certain biological properties (e.g., increased affinity, reduced immunogenicity) relative to the parent antibody and/or will have substantially retained certain biological properties of the parent antibody. An exemplary substitutional variant is an affinity matured antibody, which may be conveniently generated, e.g., using phage display-based affinity maturation techniques such as those described herein. Briefly, one or more HVR residues are mutated and the variant antibodies displayed on phage and screened for a particular biological activity (e.g. binding affinity).
[0298] Alterations (e.g., substitutions) may be made in HVRs, e.g., to improve antibody affinity. Such alterations may be made in HVR "hotspots," i.e., residues encoded by codons that undergo mutation at high frequency during the somatic maturation process (see, e.g., Chowdhury, Methods Mol. Biol. 207:179-196 (2008)), and/or residues that contact antigen, with the resulting variant VH or VL being tested for binding affinity. Affinity maturation by constructing and reselecting from secondary libraries has been described, e.g., in Hoogenboom et al. in Methods in Molecular Biology 178:1-37 (O'Brien et al., ed., Human Press, Totowa, N.J., (2001).) In some embodiments of affinity maturation, diversity is introduced into the variable genes chosen for maturation by any of a variety of methods (e.g., error-prone PCR, chain shuffling, or oligonucleotide-directed mutagenesis). A secondary library is then created. The library is then screened to identify any antibody variants with the desired affinity. Another method to introduce diversity involves HVR-directed approaches, in which several HVR residues (e.g., 4-6 residues at a time) are randomized. HVR residues involved in antigen binding may be specifically identified, e.g., using alanine scanning mutagenesis or modeling. CDR-H3 and CDR-L3 in particular are often targeted.
[0299] In certain embodiments, substitutions, insertions, or deletions may occur within one or more HVRs so long as such alterations do not substantially reduce the ability of the antibody to bind antigen. For example, conservative alterations (e.g., conservative substitutions as provided herein) that do not substantially reduce binding affinity may be made in HVRs. Such alterations may, for example, be outside of antigen contacting residues in the HVRs. In certain embodiments of the variant VH and VL sequences provided above, each HVR either is unaltered, or contains no more than one, two or three amino acid substitutions.
[0300] A useful method for identification of residues or regions of an antibody that may be targeted for mutagenesis is called "alanine scanning mutagenesis" as described by Cunningham and Wells (1989) Science, 244:1081-1085. In this method, a residue or group of target residues (e.g., charged residues such as arg, asp, his, lys, and glu) are identified and replaced by a neutral or negatively charged amino acid (e.g., alanine or polyalanine) to determine whether the interaction of the antibody with antigen is affected. Further substitutions may be introduced at the amino acid locations demonstrating functional sensitivity to the initial substitutions. Alternatively, or additionally, a crystal structure of an antigen-antibody complex to identify contact points between the antibody and antigen. Such contact residues and neighboring residues may be targeted or eliminated as candidates for substitution. Variants may be screened to determine whether they contain the desired properties.
[0301] Amino acid sequence insertions include amino- and/or carboxyl-terminal fusions ranging in length from one residue to polypeptides containing a hundred or more residues, as well as intrasequence insertions of single or multiple amino acid residues. Examples of terminal insertions include an antibody with an N-terminal methionyl residue. Other insertional variants of the antibody molecule include the fusion to the N- or C-terminus of the antibody to an enzyme (e.g. for ADEPT) or a polypeptide which increases the serum half-life of the antibody.
[0302] b) Glycosylation Variants
[0303] In certain embodiments, an antibody provided herein is altered to increase or decrease the extent to which the antibody is glycosylated. Addition or deletion of glycosylation sites to an antibody may be conveniently accomplished by altering the amino acid sequence such that one or more glycosylation sites is created or removed.
[0304] Where the antibody comprises an Fc region, the carbohydrate attached thereto may be altered. Native antibodies produced by mammalian cells typically comprise a branched, biantennary oligosaccharide that is generally attached by an N-linkage to Asn297 of the CH2 domain of the Fc region. See, e.g., Wright et al. TIBTECH 15:26-32 (1997). The oligosaccharide may include various carbohydrates, e.g., mannose, N-acetyl glucosamine (GlcNAc), galactose, and sialic acid, as well as a fucose attached to a GlcNAc in the "stem" of the biantennary oligosaccharide structure. In some embodiments, modifications of the oligosaccharide in an antibody of the invention may be made in order to create antibody variants with certain improved properties.
[0305] In one embodiment, antibody variants are provided having a carbohydrate structure that lacks fucose attached (directly or indirectly) to an Fc region. For example, the amount of fucose in such antibody may be from 1% to 80%, from 1% to 65%, from 5% to 65% or from 20% to 40%. The amount of fucose is determined by calculating the average amount of fucose within the sugar chain at Asn297, relative to the sum of all glycostructures attached to Asn 297 (e. g. complex, hybrid and high mannose structures) as measured by MALDI-TOF mass spectrometry, as described in WO 2008/077546, for example. Asn297 refers to the asparagine residue located at about position 297 in the Fc region (Eu numbering of Fc region residues); however, Asn297 may also be located about .+-.3 amino acids upstream or downstream of position 297, i.e., between positions 294 and 300, due to minor sequence variations in antibodies. Such fucosylation variants may have improved ADCC function. See, e.g., US Patent Publication Nos. US 2003/0157108 (Presta, L.); US 2004/0093621 (Kyowa Hakko Kogyo Co., Ltd). Examples of publications related to "defucosylated" or "fucose-deficient" antibody variants include: US 2003/0157108; WO 2000/61739; WO 2001/29246; US 2003/0115614; US 2002/0164328; US 2004/0093621; US 2004/0132140; US 2004/0110704; US 2004/0110282; US 2004/0109865; WO 2003/085119; WO 2003/084570; WO 2005/035586; WO 2005/035778; WO2005/053742; WO2002/031140; Okazaki et al. J. Mol. Biol. 336:1239-1249 (2004); Yamane-Ohnuki et al. Biotech. Bioeng. 87: 614 (2004). Examples of cell lines capable of producing defucosylated antibodies include Lec13 CHO cells deficient in protein fucosylation (Ripka et al. Arch. Biochem. Biophys. 249:533-545 (1986); US Pat Appl No US 2003/0157108 Al, Presta, L; and WO 2004/056312 Al, Adams et al., especially at Example 11), and knockout cell lines, such as alpha-1,6-fucosyltransferase gene, FUT8, knockout CHO cells (see, e.g., Yamane-Ohnuki et al. Biotech. Bioeng. 87: 614 (2004); Kanda, Y. et al., Biotechnol. Bioeng., 94(4):680-688 (2006); and WO2003/085107).
[0306] Antibodies variants are further provided with bisected oligosaccharides, e.g., in which a biantennary oligosaccharide attached to the Fc region of the antibody is bisected by GlcNAc. Such antibody variants may have reduced fucosylation and/or improved ADCC function. Examples of such antibody variants are described, e.g., in WO 2003/011878 (Jean-Mairet et al.); U.S. Pat. No. 6,602,684 (Umana et al.); and US 2005/0123546 (Umana et al.). Antibody variants with at least one galactose residue in the oligosaccharide attached to the Fc region are also provided. Such antibody variants may have improved CDC function. Such antibody variants are described, e.g., in WO 1997/30087 (Patel et al.); WO 1998/58964 (Raju, S.); and WO 1999/22764 (Raju, S.).
[0307] c) Fc Region Variants
[0308] In certain embodiments, one or more amino acid modifications may be introduced into the Fc region of an antibody provided herein, thereby generating an Fc region variant. The Fc region variant may comprise a human Fc region sequence (e.g., a human IgG1, IgG2, IgG3 or IgG4 Fc region) comprising an amino acid modification (e.g. a substitution) at one or more amino acid positions.
[0309] In certain embodiments, the invention contemplates an antibody variant that possesses some but not all effector functions, which make it a desirable candidate for applications in which the half life of the antibody in vivo is important yet certain effector functions (such as complement and ADCC) are unnecessary or deleterious. In vitro and/or in vivo cytotoxicity assays can be conducted to confirm the reduction/depletion of CDC and/or ADCC activities. For example, Fc receptor (FcR) binding assays can be conducted to ensure that the antibody lacks Fc.gamma.R binding (hence likely lacking ADCC activity), but retains FcRn binding ability. The primary cells for mediating ADCC, NK cells, express Fc(RIII only, whereas monocytes express Fc(RI, Fc(RII and Fc(RIII. FcR expression on hematopoietic cells is summarized in Table 3 on page 464 of Ravetch and Kinet, Annu. Rev. Immunol. 9:457-492 (1991). Non-limiting examples of in vitro assays to assess ADCC activity of a molecule of interest is described in U.S. Pat. No. 5,500,362 (see, e.g. Hellstrom, I. et al. Proc. Nat'l Acad. Sci. USA 83:7059-7063 (1986)) and Hellstrom, 1 et al., Proc. Nat'l Acad. Sci. USA 82:1499-1502 (1985); 5,821,337 (see Bruggemann, M. et al., J. Exp. Med. 166:1351-1361 (1987)). Alternatively, non-radioactive assays methods may be employed (see, for example, ACTI.TM. non-radioactive cytotoxicity assay for flow cytometry (CellTechnology, Inc. Mountain View, CA; and CytoTox 96.RTM. non-radioactive cytotoxicity assay (Promega, Madison, Wis. ). Useful effector cells for such assays include peripheral blood mononuclear cells (PBMC) and Natural Killer (NK) cells. Alternatively, or additionally, ADCC activity of the molecule of interest may be assessed in vivo, e.g., in a animal model such as that disclosed in Clynes et al. Proc. Nat'l Acad. Sci. USA 95:652-656 (1998). C1q binding assays may also be carried out to confirm that the antibody is unable to bind C1q and hence lacks CDC activity. See, e.g., C1q and C3c binding ELISA in WO 2006/029879 and WO 2005/100402. To assess complement activation, a CDC assay may be performed (see, for example, Gazzano-Santoro et al., J. Immunol. Methods 202:163 (1996); Cragg, M. S. et al., Blood 101:1045-1052 (2003); and Cragg, M.S. and M.J. Glennie, Blood 103:2738-2743 (2004)). FcRn binding and in vivo clearance/half life determinations can also be performed using methods known in the art (see, e.g., Petkova, S. B. et al., Int'l. Immunol. 18(12):1759-1769 (2006)).
[0310] In some embodiments, an antibody includes an Fc region with a mutation that decreases binding to an Fc receptor. Antibodies with reduced effector function include without limitation those with substitution of one or more of Fc region residues 238, 265, 269, 270, 297, 327 and 329 (U.S. Pat. No. 6,737,056). Such Fc mutants include Fc mutants with substitutions at two or more of amino acid positions 265, 269, 270, 297 and 327, including the so-called "DANA" Fc mutant with substitution of residues 265 and 297 to alanine (U.S. Pat. No. 7,332,581).
[0311] Certain antibody variants with improved or diminished binding to FcRs are described. (See, e.g., U.S. Pat. No. 6,737,056; WO 2004/056312, and Shields et al., J. Biol. Chem. 9(2): 6591-6604 (2001).)
[0312] In certain embodiments, an antibody variant comprises an Fc region with one or more amino acid substitutions which improve ADCC, e.g., substitutions at positions 298, 333, and/or 334 of the Fc region (EU numbering of residues).
[0313] In some embodiments, alterations are made in the Fc region that result in altered (i.e., either improved or diminished) C1q binding and/or Complement Dependent Cytotoxicity (CDC), e.g., as described in U.S. Pat. No. 6,194,551, WO 99/51642, and Idusogie et al. J. Immunol. 164: 4178-4184 (2000).
[0314] Antibodies with increased half lives and improved binding to the neonatal Fc receptor (FcRn), which is responsible for the transfer of maternal IgGs to the fetus (Guyer et al., J. Immunol. 117:587 (1976) and Kim et al., J. Immunol. 24:249 (1994)), are described in US2005/0014934A1 (Hinton et al.). Those antibodies comprise an Fc region with one or more substitutions therein which improve binding of the Fc region to FcRn. Such Fc variants include those with substitutions at one or more of Fc region residues: 238, 256, 265, 272, 286, 303, 305, 307, 311, 312, 317, 340, 356, 360, 362, 376, 378, 380, 382, 413, 424 or 434, e.g., substitution of Fc region residue 434 (U.S. Pat. No. 7,371,826).
[0315] See also Duncan & Winter, Nature 322:738-40 (1988); U.S. Pat. No. 5,648,260; U.S. Pat. No. 5,624,821; and WO 94/29351 concerning other examples of Fc region variants.
[0316] d) Cysteine Engineered Antibody Variants
[0317] In certain embodiments, it may be desirable to create cysteine engineered antibodies, e.g., "thioMAbs," in which one or more residues of an antibody are substituted with cysteine residues. In particular embodiments, the substituted residues occur at accessible sites of the antibody. By substituting those residues with cysteine, reactive thiol groups are thereby positioned at accessible sites of the antibody and may be used to conjugate the antibody to other moieties, such as drug moieties or linker-drug moieties, to create an immunoconjugate, as described further herein. In certain embodiments, any one or more of the following residues may be substituted with cysteine: V205 (Kabat numbering) of the light chain; A118 (EU numbering) of the heavy chain; and 5400 (EU numbering) of the heavy chain Fc region. Cysteine engineered antibodies may be generated as described, e.g., in U.S. Pat. No. 7,521,541.
[0318] e) Antibody Derivatives
[0319] In certain embodiments, an antibody provided herein may be further modified to contain additional nonproteinaceous moieties that are known in the art and readily available. The moieties suitable for derivatization of the antibody include but are not limited to water soluble polymers. Non-limiting examples of water soluble polymers include, but are not limited to, polyethylene glycol (PEG), copolymers of ethylene glycol/propylene glycol, carboxymethylcellulose, dextran, polyvinyl alcohol, polyvinyl pyrrolidone, poly-1,3-dioxolane, poly-1,3,6-trioxane, ethylene/maleic anhydride copolymer, polyaminoacids (either homopolymers or random copolymers), and dextran or poly(n-vinyl pyrrolidone)polyethylene glycol, propropylene glycol homopolymers, prolypropylene oxide/ethylene oxide co-polymers, polyoxyethylated polyols (e.g., glycerol), polyvinyl alcohol, and mixtures thereof. Polyethylene glycol propionaldehyde may have advantages in manufacturing due to its stability in water. The polymer may be of any molecular weight, and may be branched or unbranched. The number of polymers attached to the antibody may vary, and if more than one polymer are attached, they can be the same or different molecules. In general, the number and/or type of polymers used for derivatization can be determined based on considerations including, but not limited to, the particular properties or functions of the antibody to be improved, whether the antibody derivative will be used in a therapy under defined conditions, etc.
[0320] In another embodiment, conjugates of an antibody and nonproteinaceous moiety that may be selectively heated by exposure to radiation are provided. In one embodiment, the nonproteinaceous moiety is a carbon nanotube (Kam et al., Proc. Natl. Acad. Sci. USA 102: 11600-11605 (2005)). The radiation may be of any wavelength, and includes, but is not limited to, wavelengths that do not harm ordinary cells, but which heat the nonproteinaceous moiety to a temperature at which cells proximal to the antibody-nonproteinaceous moiety are killed.
Recombinant Methods and Compositions
[0321] Antibodies may be produced using recombinant methods and compositions, e.g., as described in U.S. Pat. No. 4,816,567. In one embodiment, isolated nucleic acid encoding an anti-OX40 antibody described herein is provided. Such nucleic acid may encode an amino acid sequence comprising the VL and/or an amino acid sequence comprising the VH of the antibody (e.g., the light and/or heavy chains of the antibody). In a further embodiment, one or more vectors (e.g., expression vectors) comprising such nucleic acid are provided. In a further embodiment, a host cell comprising such nucleic acid is provided. In one such embodiment, a host cell comprises (e.g., has been transformed with): (1) a vector comprising a nucleic acid that encodes an amino acid sequence comprising the VL of the antibody and an amino acid sequence comprising the VH of the antibody, or (2) a first vector comprising a nucleic acid that encodes an amino acid sequence comprising the VL of the antibody and a second vector comprising a nucleic acid that encodes an amino acid sequence comprising the VH of the antibody. In one embodiment, the host cell is eukaryotic, e.g. a Chinese Hamster Ovary (CHO) cell or lymphoid cell (e.g., Y0, NS0, Sp20 cell). In one embodiment, a method of making an anti-OX40 antibody is provided, wherein the method comprises culturing a host cell comprising a nucleic acid encoding the antibody, as provided above, under conditions suitable for expression of the antibody, and optionally recovering the antibody from the host cell (or host cell culture medium).
[0322] For recombinant production of an anti-OX40 antibody, nucleic acid encoding an antibody, e.g., as described above, is isolated and inserted into one or more vectors for further cloning and/or expression in a host cell. Such nucleic acid may be readily isolated and sequenced using conventional procedures (e.g., by using oligonucleotide probes that are capable of binding specifically to genes encoding the heavy and light chains of the antibody).
[0323] Suitable host cells for cloning or expression of antibody-encoding vectors include prokaryotic or eukaryotic cells described herein. For example, antibodies may be produced in bacteria, in particular when glycosylation and Fc effector function are not needed. For expression of antibody fragments and polypeptides in bacteria, see, e.g., U.S. Pat. Nos. 5,648,237, 5,789,199, and 5,840,523. (See also Charlton, Methods in Molecular Biology, Vol. 248 (B.K.C. Lo, ed., Humana Press, Totowa, N.J., 2003), pp. 245-254, describing expression of antibody fragments in E. coli.) After expression, the antibody may be isolated from the bacterial cell paste in a soluble fraction and can be further purified.
[0324] In addition to prokaryotes, eukaryotic microbes such as filamentous fungi or yeast are suitable cloning or expression hosts for antibody-encoding vectors, including fungi and yeast strains whose glycosylation pathways have been "humanized," resulting in the production of an antibody with a partially or fully human glycosylation pattern. See Gerngross, Nat. Biotech. 22:1409-1414(2004), and Li et al., Nat. Biotech. 24:210-215 (2006).
[0325] Suitable host cells for the expression of glycosylated antibody are also derived from multicellular organisms (invertebrates and vertebrates). Examples of invertebrate cells include plant and insect cells. Numerous baculoviral strains have been identified which may be used in conjunction with insect cells, particularly for transfection of Spodoptera frugiperda cells.
[0326] Plant cell cultures can also be utilized as hosts. See, e.g., U.S. Pat. Nos. 5,959,177, 6,040,498, 6,420,548, 7,125,978, and 6,417,429 (describing PLANTIBODIES.TM. technology for producing antibodies in transgenic plants).
[0327] Vertebrate cells may also be used as hosts. For example, mammalian cell lines that are adapted to grow in suspension may be useful. Other examples of useful mammalian host cell lines are monkey kidney CV1 line transformed by SV40 (COS-7); human embryonic kidney line (293 or 293 cells as described, e.g., in Graham et al., J. Gen Virol. 36:59 (1977)); baby hamster kidney cells (BHK); mouse sertoli cells (TM4 cells as described, e.g., in Mather, Biol. Reprod. 23:243-251 (1980)); monkey kidney cells (CV1); African green monkey kidney cells (VERO-76); human cervical carcinoma cells (HELA); canine kidney cells (MDCK; buffalo rat liver cells (BRL 3A); human lung cells (W138); human liver cells (Hep G2); mouse mammary tumor (MMT 060562); TRI cells, as described, e.g., in Mather et al., Annals N.Y. Acad. Sci. 383:44-68 (1982); MRC 5 cells; and FS4 cells. Other useful mammalian host cell lines include Chinese hamster ovary (CHO) cells, including DHFR-CHO cells (Urlaub et al., Proc. Natl. Acad. Sci. USA 77:4216 (1980)); and myeloma cell lines such as Y0, NS0 and Sp2/0. For a review of certain mammalian host cell lines suitable for antibody production, see, e.g., Yazaki and Wu, Methods in Molecular Biology, Vol. 248 (B.K.C. Lo, ed., Humana Press, Totowa, N.J.), pp. 255-268 (2003).
Assays
[0328] Anti-OX40 antibodies provided herein may be identified, screened for, or characterized for their physical/chemical properties and/or biological activities by various assays known in the art.
[0329] 1. Binding Assays and Other Assays
[0330] In one aspect, an antibody of the invention is tested for its antigen binding activity, e.g., by known methods such as ELISA, Western blot, etc. OX40 binding may be determined using methods known in the art and exemplary methods are disclosed herein. In one embodiment, binding is measured using radioimmunoassay. An exemplary radioimmunassay is exemplified in the Examples. OX40 antibody is iodinated, and competition reaction mixtures are prepared containing a fixed concentration of iodinated antibody and decreasing concentrations of serially diluted, unlabeled OZ X40 antibody. Cells expressing OX40 (e.g., BT474 cells stably transfected with human OX40) are added to the reaction mixture. Following an incubation, cells are washed to separate the free iodinated OX40 antibody from the OX40 antibody bound to the cells. Level of bound iodinated OX40 antibody is determined, e.g., by counting radioactivity associated with cells, and binding affinity determined using standard methods. In another embodiment, ability of OX40 antibody to bind to surface-expressed OX40 (e.g., on T cell subsets) is assessed using flow cytometry. Peripheral white blood cells are obtained (e.g., from human, cynomolgus monkey, rat or mouse) and cells are blocked with serum. Labeled OX40 antibody is added in serial dilutions, and T cells are also stained to identify T cell subsets (using methods known in the art). Following incubation of the samples and washing, the cells are sorted using flow cytometer, and data analyzed using methods well known in the art. In another embodiment, OX40 binding may be analyzed using surface plasmon resonance. An exemplary surface plasmon resonance method is exemplified in the Examples.
[0331] In another aspect, competition assays may be used to identify an antibody that competes with any of the anti-OX40 antibodies disclosed herein for binding to OX40. In certain embodiments, such a competing antibody binds to the same epitope (e.g., a linear or a conformational epitope) that is bound by any of the anti-OX40 antibodies disclosed herein. Detailed exemplary methods for mapping an epitope to which an antibody binds are provided in Morris (1996) "Epitope Mapping Protocols," in Methods in Molecular Biology vol. 66 (Humana Press, Totowa, N.J.). A competition assay is exemplified in the Examples.
[0332] In an exemplary competition assay, immobilized OX40 is incubated in a solution comprising a first labeled antibody that binds to OX40 (e.g., mab 1A7.gr.1, mab 3C8.gr5) and a second unlabeled antibody that is being tested for its ability to compete with the first antibody for binding to OX40. The second antibody may be present in a hybridoma supernatant. As a control, immobilized OX40 is incubated in a solution comprising the first labeled antibody but not the second unlabeled antibody. After incubation under conditions permissive for binding of the first antibody to OX40, excess unbound antibody is removed, and the amount of label associated with immobilized OX40 is measured. If the amount of label associated with immobilized OX40 is substantially reduced in the test sample relative to the control sample, then that indicates that the second antibody is competing with the first antibody for binding to OX40. See Harlow and Lane (1988) Antibodies: A Laboratory Manual ch.14 (Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.).
[0333] 2. Activity Assays
[0334] In one aspect, assays are provided for identifying anti-OX40 antibodies thereof having biological activity. Biological activity may include, e.g., binding OX40 (e.g., binding human and/or cynomolgus OX40), increasing OX40-mediated signal transduction (e.g., increasing NFkB-mediated transcription), depleting cells that express human OX40 (e.g., T cells), depleting cells that express human OX40 by ADCC and/or phagocytosis, enhancing T effector cell function (e.g., CD4+ effector T cell), e.g., by increasing effector T cell proliferation and/or increasing cytokine production (e.g., gamma interferon) by effector T cells, enhancing memory T cell function (e.g., CD4+ memory T cell), e.g., by increasing memory T cell proliferation and/or increasing cytokine production by memory T cells (e.g., gamma interferon), inhibiting regulatory T cell function (e.g., by decreasing Treg suppression of effector T cell function (e.g., CD4+ effector T cell function), binding human effector cells. Antibodies having such biological activity in vivo and/or in vitro are also provided.
[0335] In certain embodiments, an antibody of the invention is tested for such biological activity.
[0336] T cell costimulation may be assayed using methods known in the art and exemplary methods are disclosed herein. For example, T cells (e.g., memory or effector T cells) may be obtained from peripheral white blood cells (e.g., isolated from human whole blood using Ficoll gradient centrifugation). Memory T cells (e.g., CD4+ memory T cells) or effector T cells (e.g. CD4+ Teff cells) may be isolated from PBMC using methods known in the art. For example, the Miltenyi CD4+ memory T cell isolation kit or Miltenyi naive CD4+ T cell isolation kit may be used. Isolated T cells are cultured in the presence of antigen presenting cells (e.g., irradiated L cells that express CD32 and CD80), and activated by addition of anti-CD3 antibody in the presence or absence of OX40 agonist antibody. Effect of agonist OX40 antibody of T cell proliferation may be measured using methods well known in the art. For example, the CellTiter Glo kit (Promega) may be used, and results read on a Multilabel Reader (Perkin Elmer). Effect of agonist OX40 antibody on T cell function may also be determined by analysis of cytokines produced by the T cell. In one embodiment, production of interferon gamma by CD4+ T cells is determined, e.g., by measurement of interferon gamma in cell culture supernatant. Methods for measuring interferon gamma are well-known in the art.
[0337] Treg cell function may be assayed using methods known in the art and exemplary methods are disclosed herein. In one example, the ability of Treg to suppress effector T cell proliferation is assayed. T cells are isolated from human whole blood using methods known in the art (e.g., isolating memory T cells or naive T cells). Purified CD4+ naive T cells are labeled (e.g., with CFSE) and purified Treg cells are labeled with a different reagent. Irradiated antigen presenting cells (e.g., L cells expressing CD32 and CD80) are co-cultured with the labeled purified naive CD4+ T cells and purified Tregs. The co-cultures are activated using anti-CD3 antibody and tested in the presence or absence of agonist OX40 antibody. Following a suitable time (e.g., 6 days of coculture), level of CD4+ naive T cell proliferation is tracked by dye dilution in reduced label staining (e.g., reduced CFSE label staining) using FACS analysis.
[0338] OX40 signaling may be assayed using methods well known in the art and exemplary methods are disclosed herein. In one embodiment, transgenic cells are generated that express human OX40 and a reporter gene comprising the NFkB promoter fused to a reporter gene (e.g., beta luciferase). Addition of OX40 agonist antibody to the cells results in increased NFkB transcription, which is detected using an assay for the reporter gene.
[0339] Phagocytosis may be assayed, e.g., by using monocyte-derived macrophages, or U937 cells (a human histiocytic lymphoma cells line with the morphology and characteristics of mature macrophages). OX40 expressing cells are added to the monocyte-derived macrophages or U937 cells in the presence or absence of anti-OX40 agonist antibody. Following culturing of the cells for a suitable period of time, the percentage of phagocytosis is determined by examining percentage of cells that double stain for markers of 1) the macrophage or U937 cell and 2) the OX40 expressing cell, and dividing this by the total number of cells that show markers of the OX40 expressing cell (e.g., GFP). Analysis may be done by flow cytometry. In another embodiment, analysis may be done by fluorescent microscopy analysis.
[0340] ADCC may be assayed, e.g., using methods well known in the art. Exemplary methods are described in the definition section and an exemplary assay is disclosed in the Examples. In some embodiments, level of OX40 is characterized on an OX40 expressing cell that is used for testing in an ADCC assay. The cell may be stained with a detectably labeled anti-OX40 antibody (e.g., PE labeled), then level of fluorescence determined using flow cytometry, and results presented as median fluorescence intensity (MFI). In another embodiment, ADCC may be analyzed by CellTiter Glo assay kit and cell viability/cytotoxicity may be determined by chemioluminescence.
[0341] The binding affinities of various antibodies to Fc.gamma.RIA, Fc.gamma.RIIA, Fc.gamma.RIIB, and two allotypes of Fc.gamma.RIIIA (F158 and V158) may be measured in ELISA-based ligand-binding assays using the respective recombinant Fc.gamma.receptors. Purified human Fc.gamma.receptors are expressed as fusion proteins containing the extracellular domain of the receptor y chain linked to a Gly/6.times. His/glutathione S-transferase (GST) polypeptide tag at the C-terminus. The binding affinities of antibodies to those human Fc.gamma.receptors are assayed as follows. For the low-affinity receptors, i.e. Fc.gamma.RIIA (CD32A), Fc.gamma.RIIB (CD32B), and the two allotypes of Fc.gamma.RIIIA (CD16), F-158 and V-158, antibodies may be tested as multimers by cross-linking with a F(ab')2 fragment of goat anti-human kappa chain (ICN Biomedical; Irvine, Calif.) at an approximate molar ratio of 1:3 antibody:cross-linking F(ab')2. Plates are coated with an anti-GST antibody (Genentech) and blocked with bovine serum albumin (BSA). After washing with phosphate-buffered saline (PBS) containing 0.05% Tween-20 with an ELx405.TM. plate washer (Biotek Instruments; Winooski, Vt.), Fc.gamma.receptors are added to the plate at 25 ng/well and incubated at room temperature for 1 hour. After the plates are washed, serial dilutions of test antibodies are added as multimeric complexes and the plates were incubated at room temperature for 2 hours. Following plate washing to remove unbound antibodies, the antibodies bound to the Fc.gamma.receptor are detected with horseradish peroxidase (HRP)-conjugated F(ab')2 fragment of goat anti-human F(ab')2 (Jackson ImmunoResearch Laboratories; West Grove, Pa. ) followed by the addition of substrate, tetramethylbenzidine (TMB) (Kirkegaard & Perry Laboratories; Gaithersburg, Md.). The plates are incubated at room temperature for 5-20 minutes, depending on the Fc.gamma.receptors tested, to allow color development. The reaction is terminated with 1 M H3PO4 and absorbance at 450 nm was measured with a microplate reader (SpectraMax.RTM.190, Molecular Devices; Sunnyvale, Calif.). Dose-response binding curves are generated by plotting the mean absorbance values from the duplicates of antibody dilutions against the concentrations of the antibody. Values for the effective concentration of the antibody at which 50% of the maximum response from binding to the Fc.gamma.receptor is detected (EC50) were determined after fitting the binding curve with a four-parameter equation using SoftMax Pro (Molecular Devices).
[0342] To select for antibodies which induce cell death, loss of membrane integrity as indicated by, e.g., propidium iodide (PI), trypan blue or 7AAD uptake may be assessed relative to control. A PI uptake assay can be performed in the absence of complement and immune effector cells. OX40 expressing cells are incubated with medium alone or medium containing of the appropriate monoclonal antibody at e.g., about 10 .mu.g/ml. The cells are incubated for a time period (e.g., 1 or 3 days). Following each treatment, cells are washed and aliquoted. In some embodiments, cells are aliquoted into 35 mm strainer-capped 12.times.75 tubes (1 ml per tube, 3 tubes per treatment group) for removal of cell clumps. Tubes then receive PI (10 .mu.g/ml). Samples may be analyzed using a FACSCAN.TM. flow cytometer and FACSCONVERT.TM. CellQuest software (Becton Dickinson).
[0343] Cells for use in any of the above in vitro assays include cells or cell lines that naturally express OX40 or that have been engineered to express OX40. Such cells include activated T cells, Treg cells and activated memory T cells that naturally express OX40. Such cells also include cell lines that express OX40 and cell lines that do not normally express OX40 but have been transfected with nucleic acid encoding OX40. Exemplary cell lines provided herein for use in any of the above in vitro assays include transgenic BT474 cells (a human breast cancer cell line) that express human OX40
[0344] It is understood that any of the above assays may be carried out using an immunoconjugate of the invention in place of or in addition to an anti-OX40 antibody.
[0345] It is understood that any of the above assays may be carried out using anti-OX40 antibody and an additional therapeutic agent.
Immunoconjugates
[0346] The invention also provides immunoconjugates comprising an anti-OX40 antibody herein conjugated to one or more cytotoxic agents, such as chemotherapeutic agents or drugs, growth inhibitory agents, toxins (e.g., protein toxins, enzymatically active toxins of bacterial, fungal, plant, or animal origin, or fragments thereof), or radioactive isotopes.
[0347] In one embodiment, an immunoconjugate is an antibody-drug conjugate (ADC) in which an antibody is conjugated to one or more drugs, including but not limited to a maytansinoid (see U.S. Pat. Nos. 5,208,020, 5,416,064 and European Patent EP 0 425 235 B1); an auristatin such as monomethylauristatin drug moieties DE and DF (MMAE and MMAF) (see U.S. Pat. Nos. 5,635,483 and 5,780,588, and 7,498,298); a dolastatin; a calicheamicin or derivative thereof (see U.S. Pat. Nos. 5,712,374, 5,714,586, 5,739,116, 5,767,285, 5,770,701, 5,770,710, 5,773,001, and 5,877,296; Hinman et al., Cancer Res. 53:3336-3342 (1993); and Lode et al., Cancer Res. 58:2925-2928 (1998)); an anthracycline such as daunomycin or doxorubicin (see Kratz et al., Current Med. Chem. 13:477-523 (2006); Jeffrey et al., Bioorganic & Med. Chem. Letters 16:358-362 (2006); Torgov et al., Bioconj. Chem. 16:717-721 (2005); Nagy et al., Proc. Natl. Acad. Sci. USA 97:829-834 (2000); Dubowchik et al., Bioorg. & Med. Chem. Letters 12:1529-1532 (2002); King et al., J. Med. Chem. 45:4336-4343 (2002); and U.S. Pat. No. 6,630,579); methotrexate; vindesine; a taxane such as docetaxel, paclitaxel, larotaxel, tesetaxel, and ortataxel; a trichothecene; and CC1065.
[0348] In another embodiment, an immunoconjugate comprises an antibody as described herein conjugated to an enzymatically active toxin or fragment thereof, including but not limited to diphtheria A chain, nonbinding active fragments of diphtheria toxin, exotoxin A chain (from Pseudomonas aeruginosa), ricin A chain, abrin A chain, modeccin A chain, alpha-sarcin, Aleurites fordii proteins, dianthin proteins, Phytolaca americana proteins (PAPI, PAPII, and PAP-S), momordica charantia inhibitor, curcin, crotin, sapaonaria officinalis inhibitor, gelonin, mitogellin, restrictocin, phenomycin, enomycin, and the tricothecenes.
[0349] In another embodiment, an immunoconjugate comprises an antibody as described herein conjugated to a radioactive atom to form a radioconjugate. A variety of radioactive isotopes are available for the production of radioconjugates. Examples include At211, I131, I125, Y90, Re186, Re188, Sm153, Bi212, P32, Pb212 and radioactive isotopes of Lu. When the radioconjugate is used for detection, it may comprise a radioactive atom for scintigraphic studies, for example tc99m or I123, or a spin label for nuclear magnetic resonance (NMR) imaging (also known as magnetic resonance imaging, mri), such as iodine-123 again, iodine-131, indium-111, fluorine-19, carbon-13, nitrogen-15, oxygen-17, gadolinium, manganese or iron.
[0350] Conjugates of an antibody and cytotoxic agent may be made using a variety of bifunctional protein coupling agents such as N-succinimidyl-3-(2-pyridyldithio) propionate (SPDP), succinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC), iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adipimidate HCl), active esters (such as disuccinimidyl suberate), aldehydes (such as glutaraldehyde), bis-azido compounds (such as bis (p-azidobenzoyl) hexanediamine), bis-diazonium derivatives (such as bis-(p-diazoniumbenzoyl)-ethylenediamine), diisocyanates (such as toluene 2,6-diisocyanate), and bis-active fluorine compounds (such as 1,5-difluoro-2,4-dinitrobenzene). For example, a ricin immunotoxin can be prepared as described in Vitetta et al., Science 238:1098 (1987). Carbon-14-labeled 1-isothiocyanatobenzyl-3-methyldiethylene triaminepentaacetic acid (MX-DTPA) is an exemplary chelating agent for conjugation of radionucleotide to the antibody. See WO94/11026. The linker may be a "cleavable linker" facilitating release of a cytotoxic drug in the cell. For example, an acid-labile linker, peptidase-sensitive linker, photolabile linker, dimethyl linker or disulfide-containing linker (Chari et al., Cancer Res. 52:127-131 (1992); U.S. Pat. No. 5,208,020) may be used.
[0351] The immunuoconjugates or ADCs herein expressly contemplate, but are not limited to such conjugates prepared with cross-linker reagents including, but not limited to, BMPS, EMCS, GMBS, HBVS, LC-SMCC, MBS, MPBH, SBAP, SIA, SIAB, SMCC, SMPB, SMPH, sulfo-EMCS, sulfo-GMBS, sulfo-KMUS, sulfo-MBS, sulfo-SIAB, sulfo-SMCC, and sulfo-SMPB, and SVSB (succinimidyl-(4-vinylsulfone)benzoate) which are commercially available (e.g., from Pierce Biotechnology, Inc., Rockford, Ill., U.S.A).
IV. Predictive and Prognostic Methods
[0352] Provided herein are methods for determining the prognosis of a subject having cancer by measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample containing cancer cells and lymphocytes obtained from the subject; and determining the prognosis of the subject based on the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample, as compared with a reference, wherein an increased number of CD4+ OX40+ Foxp3+ lymphocytes in the sample indicates that the subject may have an improved prognosis. These methods are based in part on the discovery described herein that increased prevalence of CD4+ OX40+ Foxp3+ lymphocytes (i.e., OX40+ Treg cells) in a tumor sample correlates with improved patient prognosis. Further provided herein are methods for determining the prognosis of a subject having cancer by measuring the number of CD4+, OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample containing metastatic cancer cells and lymphocytes obtained from the subject; and determining the prognosis of the subject based on the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample, as compared with a reference, wherein an increased number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample indicates that the subject may have an improved prognosis. These methods are based in part on the discovery described herein that increased prevalence of CD4+ lymphocytes, total OX40+ lymphocytes, CD4+ OX40+ Foxp3+ lymphocytes (i.e., OX40+ Treg cells), or CD4+ OX40+ Foxp3- lymphocytes (i.e., OX40+ Teff cells) in a tumor sample correlates with improved patient prognosis, and that the prevalence of each of these lymphocyte subtypes is strongly correlated in matched primary tumor and metastatic samples from a subject.
[0353] Certain aspects of the present disclosure relate to measuring the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample comprising cancer cells and lymphocytes obtained from the subject. In some embodiments, a sample may include cancer cells and lymphocytes. For example, the sample may be a tumor sample. A tumor sample may include cancer cells, lymphocytes, leukocytes, stroma, blood vessels, connective tissue, basal lamina, and any other cell type in association with the tumor. A sample may be obtained from a subject by any method known in the art, including without limitation a biopsy, endoscopy, or surgical procedure. In some embodiments, a sample may be prepared by methods such as freezing, fixation (e.g., by using formalin or a similar fixative), and/or embedding in paraffin wax. In some embodiments, a sample may be sectioned.
[0354] In some embodiments, the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a tumor is measured. In some embodiments, the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a metastatic tumor is measured. In any or all of the above cases, the lymphocytes may be tumor-infiltrating lymphocytes. As used herein, any lymphocyte associated with a tumor may be a tumor-infiltrating lymphocyte. In some embodiments, the tumor-infiltrating lymphocyte may be associated with cancer cells in the tumor. In some embodiments, the tumor-infiltrating lymphocyte may be associated with the tumor stroma.
[0355] As used herein, a CD4+ OX40+ Foxp3+ cell (e.g., a lymphocyte or leukocyte) may refer to any cell that has increased expression of CD4 as compared with a reference, increased expression of OX40 as compared with a reference, and increased expression of Foxp3 as compared with a reference. As used herein, a CD4+ OX40+ Foxp3- cell (e.g., a lymphocyte or leukocyte) may refer to any cell that has increased expression of CD4 as compared with a reference, increased expression of OX40 as compared with a reference, and decreased expression of Foxp3 as compared with a reference. As used herein, an OX40+ cell (e.g., a lymphocyte or leukocyte) may refer to any cell that has increased expression of OX40 as compared with a reference. The phrases "compared with" and "compared to" may be used interchangeably herein. In some embodiments, expression may refer to protein expression. In some embodiments, expression may refer to mRNA expression.
[0356] In some embodiment, tumor may refer to a physical mass containing a plurality of cancer cells, e.g., cells showing the characteristics of any of the cancers described herein. Examples of tumors may include primary tumors of any of the above types of cancer or metastatic tumors at a second site derived from any of the above types of cancer. In some embodiments, a tumor may contain cancer cells as well as tumor stroma.
[0357] In some embodiments, within a tumor section, a region of interest may be identified. A region of interest may refer to any subset of a tumor section selected for analysis. In some embodiments, a region of interest may include a portion of a tumor section, which may include cancer cells, lymphocytes, and, optionally, tumor stroma. Lymphocytes may be associated with cancer cells and/or tumor stroma. In some embodiments, more than one region of interest from a sample may be analyzed (e.g., if a main tumor mass and satellite mass are observed).
[0358] The level of expression of CD4, OX40, and/or Foxp3 may be compared with a reference. Many methods for comparing the expression level of a gene or protein of interest to a reference are known in the art. For example, in some embodiments, a reference may be expression level of a tumor with a known responsiveness to a treatment (e.g., with an OX40 agonist) or prognosis, a control (e.g., expression level of a housekeeping biomarker), or a reference number (e.g., a set threshold level of expression, such as a threshold based on clinical outcome data). In some embodiments, a comparison with a reference may refer to a comparison with a reference number (e.g., a predetermined level of expression) or a comparison with a reference sample.
[0359] In some embodiments, the expression level of a gene or protein of interest (e.g., CD4, OX40, and/or Foxp3) in a cell may be compared with the expression level of the gene of interest in other cells in the sample. For example, the sample may be assayed for the expression level of a gene or protein of interest (e.g., CD4, OX40, and/or Foxp3), and cells that show increased expression of the gene or protein of interest relative to other cells in the sample may be considered "positive" for expression of the gene or protein of interest. In some embodiments, the sample may be assayed for the expression level of a gene or protein of interest (e.g., CD4, OX40, and/or Foxp3), and a global threshold for expression level may be applied. Cells that show expression at or above the global threshold may be considered "positive" for expression of the gene or protein of interest, whereas cells that show expression below the global threshold may be considered "negative" for expression of the gene or protein of interest. In some embodiments, the expression level of a gene or protein of interest (e.g., CD4, OX40, and/or Foxp3) in a cell may be compared with the expression level of the gene of interest in other cells in the region of interest.
[0360] Many methods for assaying the expression level of a gene or protein of interest are known in the art. For example, in some embodiments, expression level may be assayed by immunofluorescence staining, immunohistochemistry, imaging (e.g., microscopic imaging, such as fluorescence microscopy), flow cytometry, in situ hybridization, immunoprecipitation, and the like. In some embodiments, lymphocytes showing CD4 expression, OX40 expression, and Foxp3 expression are labeled by immunofluorescence staining. Exemplary methods for immunofluorescence staining are described herein.
[0361] In some embodiments, expression level may refer to a median or mean expression level detected for a cell in a sample. In some embodiments, expression level may refer to the maximum expression level detected for a cell. For example, if assayed by immunofluorescence staining and immunofluorescence imaging, expression level of a gene or protein of interest in a cell may be based on the median fluorescence intensity detected in the area corresponding to the cell (e.g., as defined by morphology, membrane staining, and/or a nuclear staining).
[0362] In some embodiments, an absolute number of cells of interest (e.g., OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- cells) may be measured and used. In some embodiments, a relative number of cells of interest (e.g., OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- cells) may be measured or used. For example, the number of CD4+ OX40+ Foxp3+ cells may be normalized to the total amount of cells detected in a sample, or in a region of interest from the sample. In some embodiments, the number of cells of interest may be an average, mean, or median of number of cells of interest in different regions of interest of a sample or different samples from the subject.
[0363] Certain aspects of the present disclosure relate to determining the prognosis of the subject based on the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample, as compared with a reference of the present disclosure. In some embodiments, an increased number (e.g., absolute or normalized number) of cells of interest (e.g., e.g., OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- cells) as compared with a reference of the present disclosure may indicate an improved prognosis. Certain aspects of the present disclosure relate to determining the prognosis of the subject based on the number of CD4+ OX40+ Foxp3- lymphocytes in the sample, as compared with a reference of the present disclosure. In some embodiments, an increased number (e.g., absolute or normalized number) of cells of interest (e.g., CD4+ OX40+ Foxp3- cells) as compared with a reference of the present disclosure may indicate an improved prognosis. Certain aspects of the present disclosure relate to determining the prognosis of the subject based on the number of OX40+ lymphocytes in the sample, as compared with a reference of the present disclosure. In some embodiments, an increased number (e.g., absolute or normalized number) of cells of interest (e.g., OX40+ cells) as compared with a reference of the present disclosure may indicate an improved prognosis.
[0364] In some embodiments, prognosis may refer to overall survival and/or progression-free survival. In some embodiments, an improved prognosis may predict increased overall survival. In some embodiments, an improved prognosis may predict increased progression-free survival. In some embodiments, an improved prognosis may predict increased overall survival and increased progression-free survival. In some embodiments, other factors may additionally be considered to determine a prognosis, including without limitation, age, the type and/or location of a cancer, stage, cancer grade, overall health, family history, and so forth.
[0365] In some embodiments, a reference of the present disclosure may refer to a number of cells of interest (e.g., OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- cells) in a sample comprising cancer cells and lymphocytes obtained from a cancer having the same type and/or stage as the cancer of the subject. For example, if the subject presents with stage I colorectal cancer, the sample from the patient may be compared with a reference sample from another patient or patients with stage I colorectal cancer. An increased number of CD4+ OX40+ Foxp3+ cells in the patient sample as compared with the reference sample may indicate an improved prognosis. It is a discovery of the present disclosure that the numbers of OX40+ Treg and OX40+ Teff are correlated with prognosis (e.g., overall survival) when the analyses are adjusted for factors such as age, gender, and cancer stage.
[0366] In some embodiments, the reference is based on the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from a cancer having the same type and/or stage as the cancer of the subject. In some embodiments, the reference is based on the number of CD4+ OX40+ Foxp3- lymphocytes in a sample comprising metastatic cancer cells and lymphocytes obtained from a cancer having the same type and/or stage as the cancer of the subject. In some embodiments, the reference is based on the number of OX40+ lymphocytes in a sample comprising metastatic cancer cells and lymphocytes obtained from a cancer having the same type and/or stage as the cancer of the subject. For example, a set of samples obtained from cancers having a shared characteristic (e.g., the same cancer type and/or stage) may be studied from a population, such as with a clinical outcome study. This set may be used to derive a reference, e.g., a reference number, to which a subject's sample may be compared. As such, the number of OX40+, CD4+ OX40+ Foxp3+, or OX40+, CD4+ OX40+ Foxp3- lymphocytes in a sample may be correlated to clinical outcome. Therefore, a reference number of OX40+, CD4+ OX40+ Foxp3+, or OX40+, CD4+ OX40+ Foxp3- lymphocytes may be used for comparison with a subject's sample to predict responsiveness of a subject to an OX40 agonist treatment and/or determine the prognosis of a subject.
[0367] In some embodiments, the reference is the mean number of OX40+, CD4+ OX40+ Foxp3+, or OX40+, CD4+ OX40+ Foxp3- lymphocytes in samples obtained from cancers having the same type and/or stage as the cancer of the subject. In some embodiments, the reference is the median number of OX40+, CD4+ OX40+ Foxp3+, or OX40+, CD4+ OX40+ Foxp3- lymphocytes in samples obtained from cancers having the same type and/or stage as the cancer of the subject. In some embodiments, the reference is the average number of OX40+, CD4+ OX40+ Foxp3+, or OX40+, CD4+ OX40+ Foxp3- lymphocytes in samples obtained from cancers having the same type and/or stage as the cancer of the subject.
[0368] Provided herein are methods for predicting responsiveness of a subject having cancer to an OX40 agonist treatment by measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample containing cancer cells and lymphocytes obtained from the subject; and classifying the subject as a responsive or non-responsive subject based on the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample, as compared with a reference, where an increased number of CD4+ OX40+ Foxp3+ lymphocytes in the sample indicates a responsive subject, as compared with a subject whose sample has a decreased number of CD4+ OX40+ Foxp3+ lymphocytes. Further provided herein are methods for predicting responsiveness of a subject having cancer to an OX40 agonist treatment by measuring the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample containing metastatic cancer cells and lymphocytes obtained from the subject; and classifying the subject as a responsive or non-responsive subject based on the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample, as compared with a reference, where an increased number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in the sample indicates a responsive subject, as compared with a subject whose sample has a decreased number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes. Without wishing to be bound to theory, it is thought that patients having tumors with increased numbers of OX40+, CD4+ OX40+ Foxp3+, and/or CD4+ OX40+ Foxp3- lymphocytes may be more likely to respond to OX40 agonist treatment, compared to patients whose tumors have fewer such cells. Predicting a patient's responsiveness to a treatment may be highly advantageous in selecting a treatment option with the greatest likelihood of producing a successful clinical outcome, either by positively selecting a treatment option that is deemed more likely to work, or by eliminating a treatment option that is deemed less likely to work. Any of the methods herein for measuring OX40+, CD4+ OX40+ Foxp3+ or CD4+ OX40+ Foxp3- lymphocytes of the present disclosure may find use in these methods.
[0369] In some embodiments, a subject is classified as responsive or non-responsive based on the number of OX40+, CD4+ OX40+ Foxp3+, or CD4+ OX40+ Foxp3- lymphocytes in a sample, as compared with a reference. Any of the references described herein may be used. For example, the number of CD4+ OX40+ Foxp3+ lymphocytes in a patient sample may be compared to a reference number (e.g., one based on clinical outcome studies), or a sample with known responsiveness to treatment.
[0370] In some embodiments, responsiveness to treatment may refer to any one or more of: extending survival (including overall survival and progression free survival); resulting in an objective response (including a complete response or a partial response); or improving signs or symptoms of cancer. In some embodiments, responsiveness may refer to improvement of one or more factors according to the published set of RECIST or Immune-Related Response Criteria guidelines for determining the status of a tumor in a cancer patient, i.e., responding, stabilizing, or progressing. For a more detailed discussion of these guidelines, see Eisenhauer et al., Eur J Cancer 2009;45: 228-47; Topalian et al., N Engl J Med 2012;366:2443-54; Wolchok et al., Clin Can Res 2009;15:7412-20; and Therasse, P., et al. J. Natl. Cancer Inst. 92:205-16 (2000). A responsive subject may refer to a subject whose tumor(s) show improvement, e.g., according to one or more factors based on RECIST or Immune-Related Response criteria. A non-responsive subject may refer to a subject whose tumor(s) do not show improvement, e.g., according to one or more factors based on RECIST or Immune-Related Response criteria. In some embodiments, responsiveness may include immune activation. In some embodiments, responsiveness may include treatment efficacy. In some embodiments, responsiveness may include immune activation and treatment efficacy.
[0371] Conventional response criteria may not be adequate to characterize the anti-tumor activity of immunotherapeutic agents, which can produce delayed responses that may be preceded by initial apparent radiological progression, including the appearance of new lesions. Therefore, modified response criteria have been developed that account for the possible appearance of new lesions and allow radiological progression to be confirmed at a subsequent assessment. Accordingly, in some embodiments, responsiveness may refer to improvement of one of more factors according to immune-related response criteria2(irRC). See, e.g., Wolchok et al., Clin Can Res 2009;15:7412 -20. In some embodiments, new lesions are added into the defined tumor burden and followed, e.g., for radiological progression at a subsequent assessment. In some embodiments, presence of non-target lesions are included in assessment of complete response and not included in assessment of radiological progression. In some embodiments, radiological progression may be determined only on the basis of measurable disease and/or may be confirmed by a consecutive assessment .gtoreq.4 weeks from the date first documented.
[0372] Certain aspects of the present disclosure relate to methods for measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample comprising cancer cells and lymphocytes obtained from a subject. In some embodiments, the methods include: (a) labeling lymphocytes that show CD4 expression in the sample; (b) labeling lymphocytes that show OX40 expression in the sample after step (a); (c) labeling lymphocytes that show Foxp3 expression in the sample after step (b); and (d) measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample after step (c). These methods may find use in any of the other methods described herein, e.g., methods for predicting responsiveness of a subject having cancer to an OX40 agonist treatment, methods for determining the prognosis of a subject having cancer by measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample containing cancer cells and lymphocytes obtained from the subject, and/or methods for treating or delaying progression of cancer in a subject.
[0373] In some embodiments, lymphocytes that show expression of CD4, OX40, and/or Foxp3 are labeled. Any suitable method known in the art and/or described herein for labeling a cell that shows expression of a marker may be used. In some embodiments, lymphocytes that show expression of CD4, OX40, and/or Foxp3 are labeled using an antibody that specifically binds to CD4, OX40, or Foxp3. For example, such antibodies may be used for assays including without limitation immunofluorescence staining, flow cytometry, immunoprecipitation, and the like. In some embodiments, lymphocytes showing CD4 expression, OX40 expression, and Foxp3 expression are labeled by immunofluorescence staining. In some embodiments, lymphocytes that show expression of CD4, OX40, and/or Foxp3 are labeled for in situ hybridization using a probe that specifically binds to CD4, OX40, or Foxp3 transcripts. In some embodiments, expression of CD4, OX40, and/or Foxp3 may be labeled using the same technique. In some embodiments, expression of CD4, OX40, and/or Foxp3 may be labeled using multiple techniques.
[0374] In some embodiments, imaging techniques are used to quantify expression of CD4, OX40, and/or Foxp3 corresponding to labeled lymphocytes. For example, fluorescence microscopy may be used to measure the amount of CD4, OX40, and/or Foxp3 expression in a sample. Image analysis techniques, such as those described above, may be used to quantify expression of each marker on a cell-by-cell basis, enabling the enumeration of cells of interest (e.g., CD4+ OX40+ Foxp3+ lymphocytes).
[0375] In some embodiments, lymphocytes that show CD4 expression in a sample are labeled first, followed by lymphocytes that show OX40 expression in the sample, followed by lymphocytes that show Foxp3 expression in the sample. In some embodiments, detection of CD4 expression may occur before detection of OX40 expression, and/or detection of OX40 expression may occur before detection of Foxp3 expression. In some embodiments, each marker may be labeled in succession, but the detection of all three markers may be performed at the same time (e.g., by multichannel fluorescence microscopy). It is to be noted that detection of three separate fluorophores may not occur simultaneously; rather, "at the same time" may encompass successive imaging of a sample using a first channel, then a second channel, then a third channel, to create a composite, three-color image of a sample showing expression levels of CD4, OX40, and Foxp3.
[0376] In some embodiments, antigen retrieval techniques are used between labeling steps. Antigen retrieval techniques are widely known and used in the art to improve specific binding of antibodies to antigens. Without wishing to be bound to theory, these techniques are thought to uncover and/or recover antigenic sites by breaking cross-linkages or otherwise altering protein conformation. Antigen retrieval techniques often include heat, chemical, and/or enzymatic treatment of samples, such as fixed and/or paraffin-embedded samples.
[0377] In some embodiments, a sample may be treated with a solution containing an effective amount of an antigen retrieval agent between labeling lymphocytes that show CD4 expression in the sample and labeling lymphocytes that show OX40 expression in the sample and/or between labeling lymphocytes that show OX40 expression in the sample and labeling lymphocytes that show Foxp3 expression in the sample. In some embodiments, a sample may be heated while being treated with an antigen retrieval agent.
[0378] In some embodiments, the antigen retrieval agent contains EDTA. In some embodiments, the antigen retrieval agent includes an EDTA buffer. Effective amounts of various antigen retrieval agents may be determined empirically, such as by assaying the effectiveness of staining with a particular antibody, and effective amounts of many antigen retrieval agents are known in the art. For example, a sample may be treated with 1mM EDTA buffer. In some embodiments, the EDTA buffer may be at pH 8.0, pH 8.5, or pH 9.0.
[0379] In some embodiments, the antigen retrieval agent contains a citrate buffer. For example, DAKO.RTM. Target Retrieval Solution or sodium citrate buffer may be used. Effective amounts of various antigen retrieval agents may be determined empirically, such as by assaying the effectiveness of staining with a particular antibody, and effective amounts of many antigen retrieval agents are known in the art. For example, a sample may be treated with 10mM sodium citrate buffer. In some embodiments, the citrate buffer may be at pH 6.0.
[0380] It is known in the art that different primary antibodies used for immunofluorescence staining may have different effective dilutions. In some embodiments, an anti-CD4 antibody in concentrated form may be diluted between 1:50-1:500 for use. In some embodiments, an anti-CD4 antibody in concentrated form may be diluted 1:50 for use. In some embodiments, the anti-CD4 antibody may be clone 4B12 (Fisher Scientific, Waltham, Mass.). In some embodiments, an anti-OX40 antibody in concentrated form may be diluted between 1:200-1:600 for use. In some embodiments, an anti-OX40 antibody in concentrated form may be diluted 1:400 for use. In some embodiments, the anti-OX40 antibody may be 1A7. In some embodiments, an anti-Foxp3 antibody in concentrated form may be diluted between 1:100-1:500 for use. In some embodiments, an anti-Foxp3 antibody in concentrated form may be diluted 1:100 for use. In some embodiments, the anti-Foxp3 antibody may be clone 236A/E7 (Abcam, Cambridge, UK).
[0381] Various antibodies known in the art that specifically bind to OX40 may be used in the detection assays described herein. In some embodiments, lymphocytes that show expression of OX40 are labeled by one of the antibodies described herein. In some embodiments, lymphocytes that show expression of OX40 are labeled by an antibody comprising (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7.
[0382] In some embodiments, lymphocytes that show expression of OX40 are labeled by antibody clone 1A7. In some embodiments, lymphocytes that show expression of OX40 are labeled by an antibody comprising a heavy chain variable region comprising the amino acid sequence of EVQLQQSGPELVKPGASVKISCKASGYTFTDSYMSWVKQSHGKTLEWIGDMYPDNGDSSY NQKFREKVTLTVDKSSTTAYMEFRSLTSEDSAVYYCVLAPRWYFSVWGTGTTVTVSS (SEQ ID NO:) and/or a light chain variable region comprising the amino acid sequence of DIQMTQTTSSLSASLGDRVTISCRASQDISNYLNWYQQKPDGTVKLLIYYTSRLRSGVPSRFS GSGSGKDYFLTISNLEQEDVAAYFCQQGHTLPPTFGGGTKLEIK (SEQ ID NO:).
[0383] In some embodiments, the anti-OX40 antibody for use in a method of diagnosis or detection is an anti-human OX40 antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7. In some embodiments, the anti-OX40 antibody comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:4; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7. In some embodiments, the OX40 antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:7. In some embodiments, the antibody comprises a heavy chain variable domain (VH) sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:179. In certain embodiments, a VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:179. In certain embodiments, substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VH sequence in SEQ ID NO:179, including post-translational modifications of that sequence. In a particular embodiment, the VH comprises one, two or three HVRs selected from: (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:2, (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:3, and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:4. In some embodiments, the antibody comprises a light chain variable domain (VL) having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:180. In certain embodiments, a VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO: 180. In certain embodiments, the substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VL sequence in SEQ ID NO: 180, including post-translational modifications of that sequence. In a particular embodiment, the VL comprises one, two or three HVRs selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:5; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:6; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:7.
[0384] In some embodiments, the anti-OX40 antibody used in the method of diagnosis or detection is an anti-human OX40 antibody comprising at least one, two, three, four, five, or six HVRs selected from (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:31; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42. In some embodiments, the anti-OX40 antibody comprises (a) a VH domain comprising at least one, at least two, or all three VH HVR sequences selected from (i) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29, (ii) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30, and (iii) HVR-H3 comprising an amino acid sequence selected from SEQ ID NO:31; and (b) a VL domain comprising at least one, at least two, or all three VL HVR sequences selected from (i) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37, (ii) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39, and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42. In some embodiments, the anti-OX40 antibody comprises (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29; (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30; (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:31; (d) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (f) HVR-L3 comprising an amino acid sequence selected from SEQ ID NO:42. In some embodiment, the anti-OX40 antibody comprises a heavy chain variable domain (VH) sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:181. In certain embodiments, a VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:181. In certain embodiments, substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VH sequence in SEQ ID NO:181, including post-translational modifications of that sequence. In a particular embodiment, the VH comprises one, two or three HVRs selected from: (a) HVR-H1 comprising the amino acid sequence of SEQ ID NO:29, (b) HVR-H2 comprising the amino acid sequence of SEQ ID NO:30, and (c) HVR-H3 comprising the amino acid sequence of SEQ ID NO:31. In some embodiments, the anti-OX40 antibody comprises a light chain variable domain (VL) having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:182. In certain embodiments, a VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human OX40 agonist antibody comprising that sequence retains the ability to bind to OX40. In certain embodiments, a total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO: 182. In certain embodiments, the substitutions, insertions, or deletions occur in regions outside the HVRs (i.e., in the FRs). Optionally, the anti-human OX40 agonist antibody comprises the VL sequence in SEQ ID NO: 182, including post-translational modifications of that sequence. In a particular embodiment, the VL comprises one, two or three HVRs selected from (a) HVR-L1 comprising the amino acid sequence of SEQ ID NO:37; (b) HVR-L2 comprising the amino acid sequence of SEQ ID NO:39; and (c) HVR-L3 comprising the amino acid sequence of SEQ ID NO:42.
[0385] In some embodiments, the anti-OX40 antibody comprises a VH sequence of SEQ ID NO: 179. In some embodiments, the anti-OX40 antibody comprises a VL sequence of SEQ ID NO: 180. In some embodiments, the anti-OX40 antibody comprises a VH sequence of SEQ ID NO:179 and a VL sequence of SEQ ID NO: 180. In some embodiments, the anti-OX40 antibody comprises a VH sequence of SEQ ID NO: 181. In some embodiments, the anti-OX40 antibody comprises a VL sequence of SEQ ID NO: 182. In some embodiments, the anti-OX40 antibody comprises a VH sequence of SEQ ID NO:181 and a VL sequence of SEQ ID NO: 182.
V. Methods of Treatment
[0386] In one aspect, provided herein are methods for treating or delaying progression of cancer in an individual comprising administering to the individual an effective amount of an OX40 agonist. The methods of this disclosure may find use, inter alia, in treating conditions where enhanced immunogenicity is desired such as increasing tumor immunogenicity for the treatment of cancer or T cell dysfunctional disorders. A variety of cancers may be treated, or their progression may be delayed, by these methods.
[0387] Certain aspects of the present disclosure relate to methods for treating or delaying progression of cancer in a subject. In some embodiments, the methods include measuring the number of CD4+ OX40+ Foxp3+ lymphocytes in a sample containing cancer cells and lymphocytes obtained from the subject; determining the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample, as compared with a reference; and if the number of CD4+ OX40+ Foxp3+ lymphocytes in the sample is higher than the reference, administering to the subject an effective amount of an OX40 agonist. In some embodiments, the methods include administering to the subject an effective amount of an OX40 agonist, where a sample comprising cancer cells and lymphocytes obtained from the subject has an increased number of CD4+ OX40+ Foxp3+ lymphocytes, as compared with a reference. In some embodiments, the methods include measuring the number of CD4+ OX40+ Foxp3- lymphocytes in a sample containing cancer cells (e.g., metastatic cancer cells) and lymphocytes obtained from the subject; determining the number of CD4+ OX40+ Foxp3- lymphocytes in the sample, as compared with a reference; and if the number of CD4+ OX40+ Foxp3- lymphocytes in the sample is higher than the reference, administering to the subject an effective amount of an OX40 agonist. In some embodiments, the methods include administering to the subject an effective amount of an OX40 agonist, where a sample comprising (e.g., metastatic cancer cells) cancer cells and lymphocytes obtained from the subject has an increased number of CD4+ OX40+ Foxp3- lymphocytes, as compared with a reference. In some embodiments, the methods include measuring the number of OX40+ lymphocytes in a sample containing metastatic cancer cells and lymphocytes obtained from the subject; determining the number of OX40+ lymphocytes in the sample, as compared with a reference; and if the number of OX40+ lymphocytes in the sample is higher than the reference, administering to the subject an effective amount of an OX40 agonist. In some embodiments, the methods include administering to the subject an effective amount of an OX40 agonist, where a sample comprising metastatic cancer cells and lymphocytes obtained from the subject has an increased number of OX40+ lymphocytes, as compared with a reference. Any of the methods described herein for measuring the number of OX40+ lymphocytes in a sample may be used.
[0388] In some embodiments, the OX40 agonist is administered to a subject wherein a sample containing cancer cells (including metastatic cancer cells) and lymphocytes from the subject have been detected for having increase number of OX40+, CD4+ OX40+ Foxp3+, and/or CD4+ OX40+ Foxp3- lymphocytes in the sample as compared to a reference. In some embodiments, the increase may be one that is considered increased to a skilled person. For example, such increase may be at least about 0.5 fold, at least about 1 fold, at least about 2 fold, or at least about 5 fold relative to the reference.
[0389] In some embodiments, a cancer to be treated by the methods of the present disclosure includes, but is not limited to, squamous cell cancer (e.g., epithelial squamous cell cancer), lung cancer including small-cell lung cancer, non-small cell lung cancer, adenocarcinoma of the lung and squamous carcinoma of the lung, cancer of the peritoneum, hepatocellular cancer, gastric or stomach cancer including gastrointestinal cancer and gastrointestinal stromal cancer, pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, cancer of the urinary tract, hepatoma, breast cancer, colon cancer, rectal cancer, colorectal cancer, endometrial or uterine carcinoma, salivary gland carcinoma, kidney or renal cancer, prostate cancer, vulval cancer, thyroid cancer, hepatic carcinoma, anal carcinoma, penile carcinoma, melanoma, superficial spreading melanoma, lentigo maligna melanoma, acral lentiginous melanomas, nodular melanomas, multiple myeloma and B-cell lymphoma; chronic lymphocytic leukemia (CLL); acute lymphoblastic leukemia (ALL); hairy cell leukemia; chronic myeloblastic leukemia; and post-transplant lymphoproliferative disorder (PTLD), as well as abnormal vascular proliferation associated with phakomatoses, edema (such as that associated with brain tumors), Meigs' syndrome, brain, as well as head and neck cancer, and associated metastases. In some embodiments, the cancer is colorectal cancer. In some embodiments, the cancer is selected from non-small cell lung cancer, glioblastoma, neuroblastoma, renal cell carcinoma, bladder cancer, ovarian cancer, melanoma, breast carcinoma, gastric cancer, and hepatocellular carcinoma. In some embodiments, the cancer is triple-negative breast carcinoma. In some embodiments, the cancer may be an early stage cancer or a late stage cancer. In some embodiments, the cancer may be a primary tumor. In some embodiments, the cancer may be a metastatic tumor at a second site derived from any of the above types of cancer.
[0390] In some embodiments, examples of cancer further include, but are not limited to, B-cell lymphoma (including low grade/follicular non-Hodgkin's lymphoma (NHL); small lymphocytic (SL) NHL; intermediate grade/follicular NHL; intermediate grade diffuse NHL; high grade immunoblastic NHL; high grade lymphoblastic NHL; high grade small non-cleaved cell NHL; bulky disease NHL; mantle cell lymphoma; AIDS-related lymphoma; and Waldenstrom's Macroglobulinemia); chronic lymphocytic leukemia (CLL); acute lymphoblastic leukemia (ALL); Hairy cell leukemia; chronic myeloblastic leukemia; and post-transplant lymphoproliferative disorder (PTLD), as well as abnormal vascular proliferation associated with phakomatoses, edema (such as that associated with brain tumors), B-cell proliferative disorders, and Meigs' syndrome. More specific examples include, but are not limited to, relapsed or refractory NHL, front line low grade NHL, Stage III/IV NHL, chemotherapy resistant NHL, precursor B lymphoblastic leukemia and/or lymphoma, small lymphocytic lymphoma, B-cell chronic lymphocytic leukemia and/or prolymphocytic leukemia and/or small lymphocytic lymphoma, B-cell prolymphocytic lymphoma, immunocytoma and/or lymphoplasmacytic lymphoma, lymphoplasmacytic lymphoma, marginal zone B-cell lymphoma, splenic marginal zone lymphoma, extranodal marginal zone--MALT lymphoma, nodal marginal zone lymphoma, hairy cell leukemia, plasmacytoma and/or plasma cell myeloma, low grade/follicular lymphoma, intermediate grade/follicular NHL, mantle cell lymphoma, follicle center lymphoma (follicular), intermediate grade diffuse NHL, diffuse large B-cell lymphoma, aggressive NHL (including aggressive front-line NHL and aggressive relapsed NHL), NHL relapsing after or refractory to autologous stem cell transplantation, primary mediastinal large B-cell lymphoma, primary effusion lymphoma, high grade immunoblastic NHL, high grade lymphoblastic NHL, high grade small non-cleaved cell NHL, bulky disease NHL, Burkitt's lymphoma, precursor (peripheral) large granular lymphocytic leukemia, mycosis fungoides and/or Sezary syndrome, skin (cutaneous) lymphomas, anaplastic large cell lymphoma, angiocentric lymphoma.
[0391] In some embodiments, examples of cancer further include, but are not limited to, B-cell proliferative disorders, which further include, but are not limited to, lymphomas (e.g., B-Cell Non-Hodgkin's lymphomas (NHL)) and lymphocytic leukemias. Such lymphomas and lymphocytic leukemias include e.g. a) follicular lymphomas, b) Small Non-Cleaved Cell Lymphomas/Burkitt's lymphoma (including endemic Burkitt's lymphoma, sporadic Burkitt's lymphoma and Non-Burkitt's lymphoma), c) marginal zone lymphomas (including extranodal marginal zone B-cell lymphoma (Mucosa-associated lymphatic tissue lymphomas, MALT), nodal marginal zone B-cell lymphoma and splenic marginal zone lymphoma), d) Mantle cell lymphoma (MCL), e) Large Cell Lymphoma (including B-cell diffuse large cell lymphoma (DLCL), Diffuse Mixed Cell Lymphoma, Immunoblastic Lymphoma, Primary Mediastinal B-Cell Lymphoma, Angiocentric Lymphoma-Pulmonary B-Cell Lymphoma), f) hairy cell leukemia, g) lymphocytic lymphoma, Waldenstrom's macroglobulinemia, h) acute lymphocytic leukemia (ALL), chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL), B cell prolymphocytic leukemia, i) plasma cell neoplasms, plasma cell myeloma, multiple myeloma, plasmacytoma, and/or j) Hodgkin's disease.
[0392] In some embodiments of any of the methods, the cancer is a B-cell proliferative disorder. In some embodiments, the B-cell proliferative disorder is lymphoma, non-Hodgkins lymphoma (NHL), aggressive NHL, relapsed aggressive NHL, relapsed indolent NHL, refractory NHL, refractory indolent NHL, chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma, leukemia, hairy cell leukemia (HCL), acute lymphocytic leukemia (ALL), or mantle cell lymphoma. In some embodiments, the B-cell proliferative disorder is NHL, such as indolent NHL and/or aggressive NHL. In some embodiments, the B-cell proliferative disorder is indolent follicular lymphoma or diffuse large B-cell lymphoma.
[0393] It is a discovery of the present disclosure that the OX40+ status of immune infiltrates found on primary tumors and metastases are strongly correlated. That is to say, matched samples representing a primary tumor and a metastatic site from the same patient strongly correlate with respect to the number of OX40+ cells present (e.g., CD4+ OX40+ Foxp3+ Treg or CD4+ OX40+ Foxp3- Teff cells). Advantageously, this allows a clinician to sample a metastatic lesion for OX40+ status and determine the likely OX40+ status of the primary tumor, or vice versa. Certain aspects of the present disclosure relate to samples comprising cancer cells and lymphocytes. Therefore, in some embodiments, the cancer cells may be from a primary tumor, while in other embodiments, the cancer cells may be from a metastasis.
[0394] In some embodiments, the individual has cancer or is at risk of developing cancer. In some embodiments, the treatment results in a sustained response in the individual after cessation of the treatment. In some embodiments, the individual has cancer that may be at early stage or late stage. In some embodiments, the individual is a human. In some embodiments, the individual is a mammal, such as domesticated animals (e.g., cows, sheep, cats, dogs, and horses), primates (e.g., humans and non-human primates such as monkeys), rabbits, and rodents (e.g., mice and rats).
[0395] In some embodiments, provided is a method for treating or delaying progression of cancer in an individual comprising administering to the individual an effective amount of an OX40 agonist, and further comprising administering an additional therapy. The additional therapy may be radiation therapy, surgery (e.g., lumpectomy and a mastectomy), chemotherapy, gene therapy, DNA therapy, viral therapy, RNA therapy, immunotherapy, bone marrow transplantation, nanotherapy, monoclonal antibody therapy, or a combination of the foregoing. The additional therapy may be in the form of adjuvant or neoadjuvant therapy. In some embodiments, the additional therapy is the administration of small molecule enzymatic inhibitor or anti-metastatic agent. In some embodiments, the additional therapy is the administration of side-effect limiting agents (e.g., agents intended to lessen the occurrence and/or severity of side effects of treatment, such as anti-nausea agents, etc.). In some embodiments, the additional therapy is radiation therapy. In some embodiments, the additional therapy is surgery. In some embodiments, the additional therapy is a combination of radiation therapy and surgery. In some embodiments, the additional therapy is gamma irradiation. In some embodiments, the additional therapy is therapy targeting PI3K/AKT/mTOR pathway, HSP90 inhibitor, tubulin inhibitor, apoptosis inhibitor, and/or chemopreventative agent. The additional therapy may be one or more of the chemotherapeutic agents described hereabove.
[0396] Such combination therapies noted above encompass combined administration (where two or more therapeutic agents are included in the same or separate formulations), and separate administration, in which case, administration of the OX40 agonist of the invention can occur prior to, simultaneously, and/or following, administration of the additional therapeutic agent or agents. In one embodiment, administration of the OX40 agonist and administration of an additional therapeutic agent occur within about one month, or within about one, two or three weeks, or within about one, two, three, four, five, or six days, of each other. OX40 agonists of the invention can also be used in combination with radiation therapy.
[0397] In some embodiments, an anti-human OX40 agonist may be administered in conjunction with a chemotherapy or chemotherapeutic agent. In some embodiments, an anti-human OX40 agonist may be administered in conjunction with a radiation therapy or radiotherapeutic agent. In some embodiments, an anti-human OX40 agonist may be administered in conjunction with a targeted therapy or targeted therapeutic agent. In some embodiments, an anti-human OX40 agonist may be administered in conjunction with an immunotherapy or immunotherapeutic agent, for example a monoclonal antibody.
[0398] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with in combination with a PD-1 axis binding antagonist. A PD-1 axis binding antagonist includes but is not limited to a PD-1 binding antagonist, a PD-L1 binding antagonist and a PD-L2 binding antagonist. Alternative names for "PD-1" include CD279 and SLEB2. Alternative names for "PD-L1" include B7-H 1, B7-4, CD274, and B7-H. Alternative names for "PD-L2" include B7-DC, Btdc, and CD273. In some embodiments, PD-1, PD-L1, and PD-L2 are human PD-1, PD-L1 and PD-L2. In some embodiments, the PD-1 binding antagonist is a molecule that inhibits the binding of PD-1 to its ligand binding partners. In a specific aspect the PD-1 ligand binding partners are PD-L1 and/or PD-L2. In another embodiment, a PD-L1 binding antagonist is a molecule that inhibits the binding of PD-L1 to its binding partners. In a specific aspect, PD-L1 binding partners are PD-1 and/or B7-1. In another embodiment, the PD-L2 binding antagonist is a molecule that inhibits the binding of PD-L2 to its binding partners. In a specific aspect, a PD-L2 binding partner is PD-1. The antagonist may be an antibody, an antigen binding fragment thereof, an immunoadhesin, a fusion protein, or oligopeptide. In some embodiment, the PD-1 binding antagonist is an anti-PD-1 antibody (e.g., a human antibody, a humanized antibody, or a chimeric antibody). In some embodiments, the anti-PD-1 antibody is selected from the group consisting of MDX-1 106, Merck 3475 and CT-011. In some embodiments, the PD-1 binding antagonist is an immunoadhesin (e.g., an immunoadhesin comprising an extracellular or PD-1 binding portion of PD-L1 or PD-L2 fused to a constant region (e.g., an Fc region of an immunoglobulin sequence). In some embodiments, the PD-1 binding antagonist is AMP-224. In some embodiments, the PD-L1 binding antagonist is an anti-PD-L1 antibody. In some embodiments, the anti-PD-L1 binding antagonist is selected from the group consisting of YW243.55.S70, MPDL3280A (atezolizumab), MEDI4736 (durvalumab), MDX-1105, and MSB0010718C (avelumab). MDX-1 105, also known as BMS-936559, is an anti-PD-L1 antibody described in WO2007/005874. Antibody YW243.55.S70 (heavy and light chain variable region sequences shown in SEQ ID Nos. 20 and 21, respectively) is an anti-PD-L1 described in WO 2010/077634 A1. MDX-1 106, also known as MDX-1 106-04, ONO-4538 or BMS-936558, is an anti-PD-1 antibody described in WO2006/121168. Merck 3745, also known as MK-3475, SCH-900475, pembrolizumab, and KEYTRUDA.RTM., is an anti-PD-1 antibody described in WO2009/114335. CT-011, also known as hBAT, hBAT-1, and pidilizumab, is an anti-PD-1 antibody described in WO2009/101611. AMP-224, also known as B7-DCIg, is a PD-L2-Fc fusion soluble receptor described in WO2010/027827 and WO201 1/066342. In some embodiments, the anti-PD-1 antibody is MDX-1 106. Alternative names for "MDX-1106" include MDX-1 106-04, ONO-4538, BMS-936558, Nivolumab, or OPDIVO.RTM.. In some embodiments, the anti-PD-1 antibody is Nivolumab (CAS Registry Number: 946414-94-4). In some embodiments, the anti-PD-1 antibody is selected from the group consisting of MDX-1106 (nivolumab, OPDIVO.RTM.), Merck 3475 (MK-3475, pembrolizumab, KEYTRUDA.RTM.), CT-011 (Pidilizumab), MEDI-0680 (AMP-514), PDR001, REGN2810, BGB-108, and BGB-A317.
[0399] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antagonist directed against CTLA-4 (also known as CD152), e.g., a blocking antibody. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with ipilimumab (also known as MDX-010, MDX-101, or Yervoy.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with tremelimumab (also known as ticilimumab or CP-675,206). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antagonist directed against B7-H3 (also known as CD276), e.g., a blocking antibody. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with MGA271. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antagonist directed against a TGF beta, e.g., metelimumab (also known as CAT-192), fresolimumab (also known as GC1008), or LY2157299.
[0400] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment comprising adoptive transfer of a T cell (e.g., a cytotoxic T cell or CTL) expressing a chimeric antigen receptor (CAR). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment comprising adoptive transfer of a T cell comprising a dominant-negative TGF beta receptor, e.g, a dominant-negative TGF beta type II receptor. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment comprising a HERCREEM protocol (see, e.g., ClinicalTrials.gov Identifier NCT00889954).
[0401] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an agonist directed against CD137 (also known as TNFRSF9, 4-1BB, or ILA), e.g., an activating antibody. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with urelumab (also known as BMS-663513). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an agonist directed against CD40, e.g., an activating antibody. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with CP-870893. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an agonist directed against OX40 (also known as CD134), e.g., an activating antibody. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a different anti-OX40 antibody (e.g., AgonOX). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an agonist directed against CD27, e.g., an activating antibody. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with CDX-1127. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antagonist directed against indoleamine-2,3-dioxygenase (IDO). In some embodiments, with the IDO antagonist is 1-methyl-D-tryptophan (also known as 1-D-MT).
[0402] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antibody-drug conjugate. In some embodiments, the antibody-drug conjugate comprises mertansine or monomethyl auristatin E (MMAE). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with and anti-NaPi2b antibody-MMAE conjugate (also known as DNIB0600A or RG7599). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with trastuzumab emtansine (also known as T-DM1, ado-trastuzumab emtansine, or KADCYLA.RTM., Genentech). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with DMUC5754A. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antibody-drug conjugate targeting the endothelin B receptor (EDNBR), e.g., an antibody directed against EDNBR conjugated with MMAE.
[0403] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an angiogenesis inhibitor. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antibody directed against a VEGF, e.g., VEGF-A. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with bevacizumab (also known as AVASTIN.RTM., Genentech). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antibody directed against angiopoietin 2 (also known as Ang2). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with MEDI3617.
[0404] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antineoplastic agent. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an agent targeting CSF-1R (also known as M-CSFR or CD115). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with anti-CSF-1R (also known as IMC-CS4). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an interferon, for example interferon alpha or interferon gamma. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with Roferon-A (also known as recombinant Interferon alpha-2a). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with GM-CSF (also known as recombinant human granulocyte macrophage colony stimulating factor, rhu GM-CSF, sargramostim, or Leukine.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with IL-2 (also known as aldesleukin or Proleukin.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with IL-12. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antibody targeting CD20. In some embodiments, the antibody targeting CD20 is obinutuzumab (also known as GA101 or Gazyva.RTM.) or rituximab. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an antibody targeting GITR. In some embodiments, the antibody targeting GITR is TRX518.
[0405] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a cancer vaccine. In some embodiments, the cancer vaccine is a peptide cancer vaccine, which in some embodiments is a personalized peptide vaccine. In some embodiments the peptide cancer vaccine is a multivalent long peptide, a multi-peptide, a peptide cocktail, a hybrid peptide, or a peptide-pulsed dendritic cell vaccine (see, e.g., Yamada et al., Cancer Sci, 104:14-21, 2013). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an adjuvant. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment comprising a TLR agonist, e.g., Poly-ICLC (also known as Hiltonol.RTM.), LPS, MPL, or CpG ODN. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with tumor necrosis factor (TNF) alpha. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with IL-1. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with HMGB1. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an IL-10 antagonist. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an IL-4 antagonist. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an IL-13 antagonist. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an HVEM antagonist. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an ICOS agonist, e.g., by administration of ICOS-L, or an agonistic antibody directed against ICOS. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment targeting CX3CL1. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment targeting CXCL9. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment targeting CXCL10. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a treatment targeting CCL5. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an LFA-1 or ICAM1 agonist. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a Selectin agonist.
[0406] In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a targeted therapy. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of B-Raf. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with vemurafenib (also known as Zelboraf.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with dabrafenib (also known as Tafinlar.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with erlotinib (also known as Tarceva.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of a MEK, such as MEK1 (also known as MAP2K1) or MEK2 (also known as MAP2K2). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with cobimetinib (also known as GDC-0973 or XL-518). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with trametinib (also known as Mekinist.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of K-Ras. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of c-Met. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with onartuzumab (also known as MetMAb). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of Alk. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with AF802 (also known as CH5424802 or alectinib). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of a phosphatidylinositol 3-kinase (PI3K). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with BKM120. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with idelalisib (also known as GS-1101 or CAL-101). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with perifosine (also known as KRX-0401). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of an Akt. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with MK2206. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with GSK690693. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with GDC-0941. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with an inhibitor of mTOR. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with sirolimus (also known as rapamycin). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with temsirolimus (also known as CCI-779 or Torisel.RTM.). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with everolimus (also known as RAD001). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with ridaforolimus (also known as AP-23573, MK-8669, or deforolimus). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with OSI-027. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with AZD8055. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with INK128. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with a dual PI3K/mTOR inhibitor. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with XL765. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with GDC-0980. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with BEZ235 (also known as NVP-BEZ235). In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with BGT226. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with GSK2126458. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with PF-04691502. In some embodiments, an OX40 agonist (e.g., an anti-human OX40 agonist antibody) may be administered in conjunction with PF-05212384 (also known as PKI-587).
[0407] An OX40 agonist of the invention (and any additional therapeutic agent) can be administered by any suitable means, including parenteral, intrapulmonary, and intranasal, and, if desired for local treatment, intralesional administration. Parenteral infusions include intramuscular, intravenous, intraarterial, intraperitoneal, or subcutaneous administration. Dosing can be by any suitable route, e.g. by injections, such as intravenous or subcutaneous injections, depending in part on whether the administration is brief or chronic. Various dosing schedules including but not limited to single or multiple administrations over various time-points, bolus administration, and pulse infusion are contemplated herein.
[0408] OX40 agonists of the invention would be formulated, dosed, and administered in a fashion consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners. The antibody need not be, but is optionally formulated with one or more agents currently used to prevent or treat the disorder in question. The effective amount of such other agents depends on the amount of antibody present in the formulation, the type of disorder or treatment, and other factors discussed above. These are generally used in the same dosages and with administration routes as described herein, or about from 1 to 99% of the dosages described herein, or in any dosage and by any route that is empirically/clinically determined to be appropriate.
[0409] For the prevention or treatment of disease, the appropriate dosage of an OX40 agonist of the invention (when used alone or in combination with one or more other additional therapeutic agents) will depend on the type of disease to be treated, the type of antibody, the severity and course of the disease, whether the antibody is administered for preventive or therapeutic purposes, previous therapy, the patient's clinical history and response to the antibody, and the discretion of the attending physician. The antibody is suitably administered to the patient at one time or over a series of treatments. Depending on the type and severity of the disease, about 1 .sub.lag/kg to 40 mg/kg of antibody can be an initial candidate dosage for administration to the patient, whether, for example, by one or more separate administrations, or by continuous infusion. One typical daily dosage might range from about 1 .mu.g/kg to 100 mg/kg or more, depending on the factors mentioned above. For repeated administrations over several days or longer, depending on the condition, the treatment would generally be sustained until a desired suppression of disease symptoms occurs. Such doses may be administered intermittently, e.g. every week or every three weeks (e.g. such that the patient receives from about two to about twenty, or e.g. about six doses of the antibody). An initial higher loading dose, followed by one or more lower doses may be administered. However, other dosage regimens may be useful. The progress of this therapy is easily monitored by conventional techniques and assays.
[0410] In some embodiments of the methods of the present disclosure, the cancer has elevated levels of T cell infiltration. As used herein, T cell infiltration of a cancer may refer to the presence of T cells, such as tumor-infiltrating lymphocytes (TILs), within or otherwise associated with the cancer tissue. It is known in the art that T cell infiltration may be associated with improved clinical outcome in certain cancers (see, e.g., Zhang et al., N. Engl. J. Med. 348(3):203-213 (2003)). In some embodiments, the TILs may be OX40+. In some embodiments, the TILs may be CD4+ OX40+ Foxp3+ Treg or CD4+ OX40+ Foxp3- Teff cells.
[0411] The specification is considered to be sufficient to enable one skilled in the art to practice the invention. Various modifications of the invention in addition to those shown and described herein will become apparent to those skilled in the art from the foregoing description and fall within the scope of the appended claims.
VI. Kits and Articles of Manufacture
[0412] For use in the methods described above and herein, kits or articles of manufacture are also provided. Such kits may comprise at least one reagent specific for detecting CD4+, OX40+, CD4+OX40+Foxp3+, or CD4+OX40+Foxp3- lymphocytes in a sample comprising cancer cells and lymphocytes from a subject. In some embodiments, kits or articles of manufacture comprises an antibody that binds to human CD4 (e.g., NeoMarkers clone 4B12), an antibody that binds to human OX40 (e.g., antibody 1A7), and/or an antibody that binds to FoxP3 (e.g., Abcam clone 236A/E7). In some embodiments, the kits or articles of manufacture further comprise an OX40 agonist (e.g., an anti-OX40 agonist antibody) described herein. In some embodiments, the kits or articles of manufacture further comprise other reagents and/or buffers, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes. The reagents and antibodies may be in a container. Suitable containers include, for example, bottles, vials, syringes, IV solution bags, etc. The containers may be formed from a variety of materials such as glass or plastic. In some embodiments, the kits and article of manufactures further comprise a label or packages insert (that may be on or associated with a container) providing instructions to use the reagents and/or antibodies in methods described herein.
[0413] All publications, patents, and patent applications cited herein are hereby incorporated by reference in their entirety for all purposes.
EXAMPLES
[0414] The invention can be further understood by reference to the following examples, which are provided by way of illustration and are not meant to be limiting.
Example 1
Inverse Correlation between T-Cell Subsets and Cancer Stage
[0415] Increased numbers of OX40+ T cells in the tumor microenvironment of colorectal cancer (CRC) patients have been associated with improved outcome (Petty, J.K., et al. (2002) Am. J. Surg. 183(5):512-8). However, the OX40+ T cell population is heterogeneous and includes, among others, CD4+Foxp3+ regulatory T cells (Tregs) as well as CD4+Foxp3- effector T cells (Teff).
[0416] To study the functional significance of these T cell subsets, a multiplex immunofluorescence assay was developed to evaluate the expression of OX40 in certain CD4+ T cell subsets. This assay was utilized to determine whether OX40+ cell subsets and clinical outcome are associated in colorectal cancer (CRC) patients.
[0417] Materials and Methods
Case Selection
[0418] Formalin-fixed paraffin-embedded (FFPE) CRC specimens including primary site (n=48) and matched metastases (n=19) were included from a collection annotated with treatment histories and survival outcomes. Patient ages ranged from 26-85 yrs. and Stage I (4), Stage II (12), Stage III (17) and Stage IV (8) disease were represented. Cases were included well- or moderately-differentiated adenocarcinoma (n=36) with additional cases with either poorly-differentiated (n=4) or mucinous features (8).
Immunofluorescence Staining
[0419] A sequential approach was followed for all immunolabelings, including the double and triple round of immunofluorescence staining. CD4 (NeoMarkers clone 4B12), FoxP3 (Abcam clone 236A/E7), and OX40 (Clone 1A7) antibodies were used.
[0420] Formalin fixed, paraffin embedded human tissue sections were cut at 4 um, deparaffinized and rehydrated through a graded series of alcohols. Sections were then pre-treated for antigen retrieval by incubation in a PT Module (Thermo Scientific, Waltham, Mass.) using EDTA Retrieval (Lab Vision, Fremont, Calif.) at 65.degree. C. Sections were then heated to 99.degree. C. for 20 minutes, followed by 20 minutes of cooling. After pretreatment with EDTA Retrieval, endogenous peroxidase activity was quenched by incubating sections in 3% H.sub.2O.sub.2 at room temperature for 4 minutes.
[0421] Sections were then stained using antigen retrieval techniques. As such, sections were stained first using the anti-CD4 primary antibody (diluted 1:50 in 10% normal horse serum, NHS), then the anti-OX40 primary antibody (diluted to 10 .mu.g/mL in 10% NHS), and finally the anti-Foxp3 primary antibody (diluted to 10 .mu.g/mL in 10% NHS). Sections were treated with antigen retrieval agent between anti-CD4 and anti-OX40 incubation, as well as between anti-OX40 and anti-Foxp3 incubation, as described below.
[0422] For each primary antibody staining step, sections were blocked for avidin/biotin using an Avidin/Biotin Blocking Kit (Vector Laboratories, Burlingame, Calif.) according to manufacturer's instructions. Sections were then rinsed and subsequently blocked for non-specific binding sites with 10% Horse serum/3% BSA/PBS. Sections were incubated in primary antibody solution for 60 minutes at room temperature, followed by incubation with a biotinylated horse-anti-mouse (Vector Labs). Sections were subsequently incubated with Vector Elite ABC-HRP reagent (Vector Labs). After rinsing, sections were stained with secondary antibody solution per manufacturer's instructions. The following secondary antibodies were used: Alexa 488-tyramide conjugate (CD4), Alexa 555-tyramide conjugate (Foxp3), or Alexa 647-tyramide conjugate (OX40) (Life Technologies, Grand Island, N.Y.).
[0423] Between primary antibody staining steps, after incubation with secondary antibody solution, sections were incubated in EDTA buffer (Thermo Scientific, Waltham, Mass.) or DAKO target retrieval solution (Dako North America, Carpinteria, Calif.) per manufacturer's instructions at 99.degree. C. for 20 minutes, followed by 20 minutes cooling. After the last secondary antibody incubation, slides were counterstained with DAPI and cover-slipped with ProLong Gold Antifade Mountant (Life Technologies). For negative controls, sections were incubated with appropriate naive isotype controls for each marker in place of the primary antibody.
Digital Imaging and Analysis
[0424] Images were acquired by the Ariol SL-50 automated slide scanning platform (Leica Microsystems, Buffalo Grove, Ill.) at 100x final magnification using standard fluorescence filters. The entire tiled image was exported and analyzed in the Matlab software package (ver. R2012b, Mathworks, Natick, Mass.).
[0425] For each tumor section, at least one region of interest was manually identified and subjected to analysis. For some tumors, multiple regions of interest were selected (e.g., if both a main tumor mass and satellite mass were observed on the slide). Within each region of interest, individual cell nuclei were segmented using a radial symmetry based method applied to the DAPI channel (Veta, M., et al. (2013) PLoS ONE 8(7):e70221), and then scored by the presence of signal above a global intensity threshold in the nucleus (FoxP3), or the area immediately surrounding each nucleus (CD4 and OX40). The global threshold for each marker was determined empirically such that false positive calls of obviously negative cells, in samples with and without being exposed to the primary labeling antibody, were less than 0.5% of all cells identified.
Statistical Analysis
[0426] Overall survival was calculated from the date of diagnosis to the date of death from any cause; data for patients still alive were censored at the date the patient was last known to be alive. Association between level of tumor-associated T cell subsets (dichotomized by median) and overall survival was evaluated using log-rank tests. To assess the association between tumor-associated T cell subsets and TNM stage at diagnosis, Kruskal-Wallis tests were employed. No multiple testing correction was applied.
[0427] A multivariate Cox regression model was employed to assess the strength of association between overall survival and OX40 expression (dichotomized at median) adjusted for age (>=60 vs <60), gender (male vs female) and stage (IV vs III vs I/II).
[0428] Results
[0429] Tumors from 48 CRC patients including primary site (n=48) and matched metastases (n=19) were stained and digitally imaged.
[0430] FIGS. 1A-1C show representative immunofluorescence images of stage I CRC samples that were stained for expression of CD4, OX40, and Foxp3. These images demonstrate the presence of CD4+ OX40+ Foxp3+ Treg cells as well as CD4+ OX40+ Foxp3- Teff cells. Representative immunofluorescence images of stage II (FIGS. 2A-2C), stage III (FIGS. 3A-3C), and stage IV (FIGS. 4A-4C) CRC samples were also captured. To summarize these data, decreased numbers of total OX40+ cells, as well as CD4+ OX40+ Foxp3+ Treg cells, were observed in stage IV samples, compared to stage I-III samples.
[0431] From these images, tumor-associated T cell subsets were enumerated. The correlation between the number of cells corresponding to specific tumor-associated T cell subsets and tumor stage was analyzed as described above. FIGS. 5A-6D show the results of these correlation analyses, with the T cell subsets selected for study provided as follows: CD4+ (FIG. 5A), Foxp3+ (FIG. 5B), OX40+ (FIG. 5C), OX40+ CD4+ (FIG. 6A), OX40+ CD4- (FIG. 6B), OX40+ CD4+ Foxp3+ (FIG. 6C), and OX40+ CD4+ Foxp3- (FIG. 6D).
[0432] These results indicate statistically significant inverse correlations of OX40+ cells, OX40+ Treg cells, and OX40+ Teff cells with increased stage at diagnosis. These associations remained statistically significant when counts were normalized to total cells. Total CD4+ or Foxp3+ cells did not show significant association with stage.
Example 2
Presence of Increased OX40+ Lymphocytes is Associated with Improved Survival
[0433] Because of the observed correlations between OX40+ T cell subsets and cancer stage described above, the presence of these T cell subsets was next analyzed with respect to prognosis.
[0434] The prevalence of T cell subsets in the tumor samples described above was analyzed to determine potential association with patient prognosis (e.g., overall survival). FIGS. 7A-8D show the results of these correlation analyses, with the T cell subsets selected for study provided as follows: CD4+ (FIG. 7A), Foxp3+ (FIG. 7B), OX40+ (FIG. 7C), OX40+ CD4+ (FIG. 8A), OX40+ CD4- (FIG. 8B), OX40+ CD4+ Foxp3+ (FIG. 8C), and OX40+ CD4+ Foxp3- (FIG. 8D). Overall survival was plotted for patients whose samples showed expression of the labeled marker(s) above or below the median of all samples, as indicated in each plot.
[0435] These analyses indicated that increased prevalence of CD4+ (p=0.019), total OX40+ (p=0.046), and OX40+ Treg (p=0.022) cells correlated with improved overall survival. Higher prevalence of OX40+ Teff cells also showed a trend of improved overall survival, but did not reach statistical significance. When normalized to total number of cells, CD4+ (p=0.02) and total OX40+ (p=0.041) cells remained statistically significant. Treg number showed a closer correlation to survival than did Teff (p=0.265), though neither achieved statistical significance.
[0436] Importantly, CD4, OX40, and Foxp3 IHC staining remained significantly associated with overall survival in a multivariate Cox PH model, adjusting for age, gender and stage. FIGS. 11A-11D provide the results of these analyses of CD4 expression (FIG. 11A), OX40 expression (FIG. 11B), Foxp3 expression (FIG. 11C), and the ratio of OX40+ Teff cells to OX40+ Treg cells (FIG. 11D). These results indicate that these markers remain prognostic even after adjusting for baseline characteristics (e.g., age, gender, and stage).
[0437] These results demonstrate that higher numbers of total OX40+ cells and OX40+ Treg cells are associated with improved prognosis in CRC.
Example 3
Correlation between OX40+ Status of Immune Infiltrates of Primary Tumors and Metastases
[0438] Given the correlations observed between OX40+ cells found in tumor samples and tumor stage and patient survival, OX40+ T cell subsets were next analyzed in paired primary and metastatic tumor samples.
[0439] FIGS. 9A-10D show the results of these correlation analyses, with the T cell subsets selected for study provided as follows: CD4+ (FIG. 9A), Foxp3+ (FIG. 9B), OX40+ (FIG. 9C), OX40+ CD4+ (FIG. 10A), OX40+ CD4- (FIG. 10B), OX40+ CD4+ Foxp3+ (FIG. 10C), and OX40+ CD4+ Foxp3- (FIG. 10D).
[0440] Analysis of paired primary and metastatic samples (n=19) showed strong correlations between positive cell counts for CD4 (r=0.75), total OX40+ (r=0.84), OX40+ Treg (0.52), and OX40+Teff (r=0.85) subsets in primary and metastatic samples. These associations remained strong when counts were normalized to total cells. These results show that the OX40+ status of immune infiltrates of primary tumors and metastases are strongly correlated.
[0441] Taken together, these results demonstrate that higher numbers of total OX40+ cells and OX40+ Treg cells are associated with improved prognosis in CRC. The triple immunofluorescence assay described herein may be useful in characterizing associations between OX40+ Treg and Teff cell subsets and clinical outcome in multiple solid tumor types. Moreover, incorporating this assay into clinical trials may help identify patients and indications that are likely to respond to therapeutics targeting OX40.
[0442] All patents, patent applications, documents, and articles cited herein are herein incorporated by reference in their entireties.
Sequence CWU 1
1
2261249PRTHomo sapiens 1Leu His Cys Val Gly Asp Thr Tyr Pro Ser Asn
Asp Arg Cys Cys His1 5 10 15Glu Cys Arg Pro Gly Asn Gly Met Val Ser
Arg Cys Ser Arg Ser Gln 20 25 30Asn Thr Val Cys Arg Pro Cys Gly Pro
Gly Phe Tyr Asn Asp Val Val 35 40 45Ser Ser Lys Pro Cys Lys Pro Cys
Thr Trp Cys Asn Leu Arg Ser Gly 50 55 60Ser Glu Arg Lys Gln Leu Cys
Thr Ala Thr Gln Asp Thr Val Cys Arg65 70 75 80Cys Arg Ala Gly Thr
Gln Pro Leu Asp Ser Tyr Lys Pro Gly Val Asp 85 90 95Cys Ala Pro Cys
Pro Pro Gly His Phe Ser Pro Gly Asp Asn Gln Ala 100 105 110Cys Lys
Pro Trp Thr Asn Cys Thr Leu Ala Gly Lys His Thr Leu Gln 115 120
125Pro Ala Ser Asn Ser Ser Asp Ala Ile Cys Glu Asp Arg Asp Pro Pro
130 135 140Ala Thr Gln Pro Gln Glu Thr Gln Gly Pro Pro Ala Arg Pro
Ile Thr145 150 155 160Val Gln Pro Thr Glu Ala Trp Pro Arg Thr Ser
Gln Gly Pro Ser Thr 165 170 175Arg Pro Val Glu Val Pro Gly Gly Arg
Ala Val Ala Ala Ile Leu Gly 180 185 190Leu Gly Leu Val Leu Gly Leu
Leu Gly Pro Leu Ala Ile Leu Leu Ala 195 200 205Leu Tyr Leu Leu Arg
Arg Asp Gln Arg Leu Pro Pro Asp Ala His Lys 210 215 220Pro Pro Gly
Gly Gly Ser Phe Arg Thr Pro Ile Gln Glu Glu Gln Ala225 230 235
240Asp Ala His Ser Thr Leu Ala Lys Ile 24525PRTArtificial
SequenceSynthetic Construct 2Asp Ser Tyr Met Ser1 5317PRTArtificial
SequenceSynthetic Construct 3Asp Met Tyr Pro Asp Asn Gly Asp Ser
Ser Tyr Asn Gln Lys Phe Arg1 5 10 15Glu48PRTArtificial
SequenceSynthetic Construct 4Ala Pro Arg Trp Tyr Phe Ser Val1
5511PRTArtificial SequenceSynthetic Construct 5Arg Ala Ser Gln Asp
Ile Ser Asn Tyr Leu Asn1 5 1067PRTArtificial SequenceSynthetic
Construct 6Tyr Thr Ser Arg Leu Arg Ser1 579PRTArtificial
SequenceSynthetic Construct 7Gln Gln Gly His Thr Leu Pro Pro Thr1
585PRTArtificial SequenceSynthetic Construct 8Asp Ala Tyr Met Ser1
595PRTArtificial SequenceSynthetic Construct 9Glu Ser Tyr Met Ser1
51017PRTArtificial SequenceSynthetic Construct 10Asp Met Tyr Pro
Asp Asn Ala Asp Ser Ser Tyr Asn Gln Lys Phe Arg1 5 10
15Glu1117PRTArtificial SequenceSynthetic Construct 11Asp Met Tyr
Pro Asp Asn Ala Asp Ala Ser Tyr Asn Gln Lys Phe Arg1 5 10
15Glu1217PRTArtificial SequenceSynthetic Construct 12Asp Met Tyr
Pro Asp Asn Gly Asp Ala Ser Tyr Asn Gln Lys Phe Arg1 5 10
15Glu1317PRTArtificial SequenceSynthetic Construct 13Asp Met Tyr
Pro Asp Ser Gly Asp Ser Ser Tyr Asn Gln Lys Phe Arg1 5 10
15Glu1417PRTArtificial SequenceSynthetic Construct 14Asp Met Tyr
Pro Asp Asn Gly Ser Ser Ser Tyr Asn Gln Lys Phe Arg1 5 10
15Glu158PRTArtificial SequenceSynthetic Construct 15Ala Pro Arg Trp
Tyr Phe Ser Ala1 5168PRTArtificial SequenceSynthetic Construct
16Ala Pro Arg Trp Tyr Ala Ser Val1 5178PRTArtificial
SequenceSynthetic Construct 17Ala Pro Arg Trp Ala Phe Ser Val1
5188PRTArtificial SequenceSynthetic Construct 18Ala Pro Ala Trp Tyr
Phe Ser Val1 5198PRTArtificial SequenceSynthetic Construct 19Ala
Pro Arg Trp Tyr Phe Ala Val1 5208PRTArtificial SequenceSynthetic
Construct 20Ala Pro Arg Ala Tyr Phe Ser Val1 5218PRTArtificial
SequenceSynthetic Construct 21Ala Ala Arg Trp Tyr Phe Ser Val1
5229PRTArtificial SequenceSynthetic Construct 22Gln Gln Gly His Thr
Leu Pro Ala Thr1 5239PRTArtificial SequenceSynthetic Construct
23Gln Gln Gly His Thr Ala Pro Pro Thr1 5249PRTArtificial
SequenceSynthetic Construct 24Gln Gln Gly Ala Thr Leu Pro Pro Thr1
5259PRTArtificial SequenceSynthetic Construct 25Gln Gln Gly His Ala
Leu Pro Pro Thr1 5269PRTArtificial SequenceSynthetic Construct
26Gln Gln Ala His Thr Leu Pro Pro Thr1 5279PRTArtificial
SequenceSynthetic Construct 27Gln Gln Gly His Thr Leu Ala Pro Thr1
5289PRTArtificial SequenceSynthetic Construct 28Gln Ala Gly His Thr
Leu Pro Pro Thr1 5295PRTArtificial SequenceSynthetic Construct
29Asn Tyr Leu Ile Glu1 53017PRTArtificial SequenceSynthetic
Construct 30Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu Lys
Phe Lys1 5 10 15Gly3117PRTArtificial SequenceSynthetic Construct
31Val Ile Asn Pro Gly Ser Gly Asp Ala Tyr Tyr Ser Glu Lys Phe Lys1
5 10 15Gly3217PRTArtificial SequenceSynthetic Construct 32Val Ile
Asn Pro Gly Ser Gly Asp Gln Tyr Tyr Ser Glu Lys Phe Lys1 5 10
15Gly335PRTArtificial SequenceSynthetic Construct 33Asp Arg Leu Asp
Tyr1 5345PRTArtificial SequenceSynthetic Construct 34Ala Arg Leu
Asp Tyr1 5355PRTArtificial SequenceSynthetic Construct 35Asp Ala
Leu Asp Tyr1 5365PRTArtificial SequenceSynthetic Construct 36Asp
Arg Ala Asp Tyr1 53711PRTArtificial SequenceSynthetic Construct
37His Ala Ser Gln Asp Ile Ser Ser Tyr Ile Val1 5 10387PRTArtificial
SequenceSynthetic Construct 38His Gly Thr Asn Leu Glu Asp1
5397PRTArtificial SequenceSynthetic Construct 39His Gly Thr Asn Leu
Glu Ser1 5407PRTArtificial SequenceSynthetic Construct 40His Gly
Thr Asn Leu Glu Glu1 5417PRTArtificial SequenceSynthetic Construct
41His Gly Thr Asn Leu Glu Gln1 5429PRTArtificial SequenceSynthetic
Construct 42Val His Tyr Ala Gln Phe Pro Tyr Thr1 5439PRTArtificial
SequenceSynthetic Construct 43Ala His Tyr Ala Gln Phe Pro Tyr Thr1
5449PRTArtificial SequenceSynthetic Construct 44Val Ala Tyr Ala Gln
Phe Pro Tyr Thr1 5459PRTArtificial SequenceSynthetic Construct
45Val His Ala Ala Gln Phe Pro Tyr Thr1 5469PRTArtificial
SequenceSynthetic Construct 46Val His Tyr Ala Ala Phe Pro Tyr Thr1
5479PRTArtificial SequenceSynthetic Construct 47Val His Tyr Ala Gln
Ala Pro Tyr Thr1 5489PRTArtificial SequenceSynthetic Construct
48Val His Tyr Ala Gln Phe Ala Tyr Thr1 5499PRTArtificial
SequenceSynthetic Construct 49Val His Tyr Ala Gln Phe Pro Ala Thr1
5505PRTArtificial SequenceSynthetic Construct 50Asp Tyr Gly Val
Leu1 55116PRTArtificial SequenceSynthetic Construct 51Met Ile Trp
Ser Gly Gly Thr Thr Asp Tyr Asn Ala Ala Phe Ile Ser1 5 10
15525PRTArtificial SequenceSynthetic Construct 52Glu Glu Met Asp
Tyr1 55311PRTArtificial SequenceSynthetic Construct 53Arg Ala Ser
Gln Asp Ile Ser Asn Phe Leu Asn1 5 10547PRTArtificial
SequenceSynthetic Construct 54Tyr Thr Ser Arg Leu His Ser1
5559PRTArtificial SequenceSynthetic Construct 55Gln Gln Gly Asn Thr
Leu Pro Trp Thr1 556117PRTArtificial SequenceSynthetic Construct
56Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1
5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp
Ser 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu
Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn
Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr
Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp
Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser
Val Trp Gly Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
11557107PRTArtificial SequenceSynthetic Construct 57Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
10558117PRTArtificial SequenceSynthetic Construct 58Glu Val Gln Leu
Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met
Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly
Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55
60Arg Glu Arg Val Thr Ile Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr65
70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly
Thr Leu 100 105 110Val Thr Val Ser Ser 11559107PRTArtificial
SequenceSynthetic Construct 59Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 10560117PRTArtificial
SequenceSynthetic Construct 60Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Leu Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11561107PRTArtificial SequenceSynthetic
Construct 61Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys
Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys
Val Glu Ile Lys 100 10562117PRTArtificial SequenceSynthetic
Construct 62Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe
Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser
Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Val Asp Thr
Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr
Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
11563107PRTArtificial SequenceSynthetic Construct 63Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Gly Lys Thr Val Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
10564117PRTArtificial SequenceSynthetic Construct 64Glu Val Gln Leu
Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met
Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly
Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55
60Arg Glu Arg Val Thr Ile Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr65
70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly
Thr Leu 100 105 110Val Thr Val Ser Ser 11565107PRTArtificial
SequenceSynthetic Construct 65Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Thr Val Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 10566117PRTArtificial
SequenceSynthetic Construct 66Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Ile Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11567107PRTArtificial SequenceSynthetic
Construct 67Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Thr Val
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Lys Asp Tyr Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr
Tyr Phe Cys Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe Gly Gln
Gly Thr Lys Val Glu Ile Lys 100 10568117PRTArtificial
SequenceSynthetic Construct 68Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Ile Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11569107PRTArtificial SequenceSynthetic
Construct 69Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Thr Val
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Lys Asp Tyr Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Phe Cys
Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys
Val Glu Ile Lys 100 10570117PRTArtificial SequenceSynthetic
Construct 70Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe
Thr Asp Ala 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser
Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr
Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr
Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
11571107PRTArtificial SequenceSynthetic Construct 71Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
10572117PRTArtificial SequenceSynthetic Construct 72Glu Val Gln Leu
Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Glu Ser 20 25 30Tyr Met
Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly
Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55
60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65
70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly
Thr Leu 100 105 110Val Thr Val Ser Ser 11573107PRTArtificial
SequenceSynthetic Construct 73Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 10574117PRTArtificial
SequenceSynthetic Construct 74Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Ala Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11575107PRTArtificial SequenceSynthetic
Construct 75Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys
Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys
Val Glu Ile Lys 100 10576117PRTArtificial SequenceSynthetic
Construct 76Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe
Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Ala Asp Ala Ser
Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr
Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr
Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
11577107PRTArtificial SequenceSynthetic Construct 77Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
10578117PRTArtificial SequenceSynthetic Construct 78Glu Val Gln Leu
Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met
Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly
Asp Met Tyr Pro Asp Asn Gly Asp Ala Ser Tyr Asn Gln Lys Phe 50 55
60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65
70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly
Thr Leu 100 105 110Val Thr Val Ser Ser 11579107PRTArtificial
SequenceSynthetic Construct 79Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 10580117PRTArtificial
SequenceSynthetic Construct 80Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Ser Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11581107PRTArtificial SequenceSynthetic
Construct 81Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys
Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys
Val Glu Ile Lys 100 10582117PRTArtificial SequenceSynthetic
Construct 82Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe
Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Gly Ser Ser Ser
Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr
Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr
Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
11583107PRTArtificial SequenceSynthetic Construct 83Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
10584117PRTArtificial SequenceSynthetic Construct 84Glu Val Gln Leu
Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ala 20 25 30Tyr Met
Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly
Asp Met Tyr Pro Asp Asn Ala Asp Ala Ser Tyr Asn Gln Lys Phe 50 55
60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65
70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly
Thr Leu 100 105 110Val Thr Val Ser Ser 11585107PRTArtificial
SequenceSynthetic Construct 85Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro Pro 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 10586117PRTArtificial
SequenceSynthetic Construct 86Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11587107PRTArtificial SequenceSynthetic
Construct 87Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys
Gln Gln Gly His Thr Leu Pro Ala 85 90 95Thr Phe Gly Gln Gly Thr Lys
Val Glu Ile Lys 100 10588117PRTArtificial SequenceSynthetic
Construct 88Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe
Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser
Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr
Ser Thr Ser Thr Ala Tyr65 70 75
80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr
Leu 100 105 110Val Thr Val Ser Ser 11589107PRTArtificial
SequenceSynthetic Construct 89Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Ala Pro Pro 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 10590117PRTArtificial
SequenceSynthetic Construct 90Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11591107PRTArtificial SequenceSynthetic
Construct 91Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys
Gln Gln Gly Ala Thr Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys
Val Glu Ile Lys 100 10592117PRTArtificial SequenceSynthetic
Construct 92Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe
Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser
Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr
Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr
Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
11593107PRTArtificial SequenceSynthetic Construct 93Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Ala Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
10594117PRTArtificial SequenceSynthetic Construct 94Glu Val Gln Leu
Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met
Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly
Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55
60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65
70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly
Thr Leu 100 105 110Val Thr Val Ser Ser 11595107PRTArtificial
SequenceSynthetic Construct 95Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Ala His Thr Leu Pro Pro 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 10596117PRTArtificial
SequenceSynthetic Construct 96Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 11597107PRTArtificial SequenceSynthetic
Construct 97Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile
Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro
Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro
Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr
Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys
Gln Gln Gly His Thr Leu Ala Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys
Val Glu Ile Lys 100 10598117PRTArtificial SequenceSynthetic
Construct 98Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro
Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe
Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly
Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser
Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr
Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr
Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
11599107PRTArtificial SequenceSynthetic Construct 99Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Ala Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105100117PRTArtificial SequenceSynthetic Construct 100Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Ala Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115101107PRTArtificial SequenceSynthetic Construct 101Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105102117PRTArtificial SequenceSynthetic Construct 102Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Ala Ser Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115103107PRTArtificial SequenceSynthetic Construct 103Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105104117PRTArtificial SequenceSynthetic Construct 104Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Ala Phe Ser Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115105107PRTArtificial SequenceSynthetic Construct 105Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105106117PRTArtificial SequenceSynthetic Construct 106Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Pro Ala Trp Tyr Phe Ser Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115107107PRTArtificial SequenceSynthetic Construct 107Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105108117PRTArtificial SequenceSynthetic Construct 108Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ala Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115109107PRTArtificial SequenceSynthetic Construct 109Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55
60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65
70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105110117PRTArtificial SequenceSynthetic Construct 110Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Ala Tyr Phe Ser Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115111107PRTArtificial SequenceSynthetic Construct 111Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105112117PRTArtificial SequenceSynthetic Construct 112Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Ala Arg Trp Tyr Phe Ser Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115113107PRTArtificial SequenceSynthetic Construct 113Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105114117PRTArtificial SequenceSynthetic Construct 114Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115115107PRTArtificial SequenceSynthetic Construct 115Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105116117PRTArtificial SequenceSynthetic Construct 116Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Ala Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly
Gln Gly Thr Leu 100 105 110Val Thr Val Ser Ser
115117107PRTArtificial SequenceSynthetic Construct 117Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu
Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly His Thr
Leu Pro Pro 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105118114PRTArtificial SequenceSynthetic Construct 118Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu
Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe
50 55 60Lys Gly Arg Val Thr Ile Thr Arg Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr
Leu Val Thr Val 100 105 110Ser Ser119107PRTArtificial
SequenceSynthetic Construct 119Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105120114PRTArtificial
SequenceSynthetic Construct 120Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Ile Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser121107PRTArtificial SequenceSynthetic Construct 121Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105122114PRTArtificial SequenceSynthetic Construct 122Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser123107PRTArtificial
SequenceSynthetic Construct 123Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105124114PRTArtificial
SequenceSynthetic Construct 124Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Ile Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser125107PRTArtificial SequenceSynthetic Construct 125Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105126114PRTArtificial SequenceSynthetic Construct 126Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser127107PRTArtificial
SequenceSynthetic Construct 127Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105128114PRTArtificial
SequenceSynthetic Construct 128Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser129107PRTArtificial SequenceSynthetic Construct 129Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Ser Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105130114PRTArtificial SequenceSynthetic Construct 130Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75
80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr
Val 100 105 110Ser Ser131107PRTArtificial SequenceSynthetic
Construct 131Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala
Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp
Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser
Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Glu Gly Val
Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu
Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr
Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr
Lys Val Glu Ile Lys 100 105132114PRTArtificial SequenceSynthetic
Construct 132Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys
Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala
Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln
Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr
Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp
Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg
Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp
Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105 110Ser
Ser133107PRTArtificial SequenceSynthetic Construct 133Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile
Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40
45Tyr His Gly Thr Asn Leu Glu Gln Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln
Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105134114PRTArtificial SequenceSynthetic Construct 134Glu Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu
Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe
50 55 60Lys Gly Arg Val Thr Ile Thr Ala Asp Thr Ser Thr Ser Thr Ala
Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr
Leu Val Thr Val 100 105 110Ser Ser135107PRTArtificial
SequenceSynthetic Construct 135Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Ala
Asp Tyr Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105136114PRTArtificial
SequenceSynthetic Construct 136Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser137107PRTArtificial SequenceSynthetic Construct 137Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Ala Asp Tyr Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105138114PRTArtificial SequenceSynthetic Construct 138Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Arg Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser139107PRTArtificial
SequenceSynthetic Construct 139Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105140114PRTArtificial
SequenceSynthetic Construct 140Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser141107PRTArtificial SequenceSynthetic Construct 141Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Pro Lys Leu Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105142114PRTArtificial SequenceSynthetic Construct 142Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Arg Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser143107PRTArtificial
SequenceSynthetic Construct 143Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Phe Lys Leu Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105144114PRTArtificial
SequenceSynthetic Construct 144Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Arg Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser145107PRTArtificial SequenceSynthetic Construct 145Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105146114PRTArtificial SequenceSynthetic Construct 146Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser147107PRTArtificial
SequenceSynthetic Construct 147Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Ala His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105148114PRTArtificial
SequenceSynthetic Construct 148Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser149107PRTArtificial SequenceSynthetic Construct 149Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val Ala Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105150114PRTArtificial SequenceSynthetic Construct 150Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser151107PRTArtificial
SequenceSynthetic Construct 151Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Ala Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105152114PRTArtificial SequenceSynthetic Construct 152Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser153107PRTArtificial
SequenceSynthetic Construct 153Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Ala Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105154114PRTArtificial
SequenceSynthetic Construct 154Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser155107PRTArtificial SequenceSynthetic Construct 155Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Ala Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105156114PRTArtificial SequenceSynthetic Construct 156Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser157107PRTArtificial
SequenceSynthetic Construct 157Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Ala Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105158114PRTArtificial
SequenceSynthetic Construct 158Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser159107PRTArtificial SequenceSynthetic Construct 159Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Ala 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105160114PRTArtificial SequenceSynthetic Construct 160Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Ala Arg Leu Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser161107PRTArtificial
SequenceSynthetic Construct 161Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105162114PRTArtificial
SequenceSynthetic Construct 162Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu Ile Glu Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Val Ile Asn Pro Gly
Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Leu Thr Ala Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Asp Ala Leu Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val 100 105
110Ser Ser163107PRTArtificial SequenceSynthetic Construct 163Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr
20 25 30Ile Val Trp Tyr Gln Gln Lys Pro Gly Lys Ser Phe Lys Gly Leu
Ile 35 40 45Tyr His Gly Thr Asn Leu Glu Asp Gly Val Pro Ser Arg Phe
Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Val His Tyr
Ala Gln Phe Pro Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys 100 105164114PRTArtificial SequenceSynthetic Construct 164Glu
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr
20 25 30Leu Ile Glu Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu
Lys Phe 50 55 60Lys Gly Arg Val Thr Leu Thr Ala Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Asp Arg Ala Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val 100 105 110Ser Ser165107PRTArtificial
SequenceSynthetic Construct 165Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys His
Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile Val Trp Tyr Gln Gln Lys
Pro Gly Lys Ser Phe Lys Gly Leu Ile 35 40 45Tyr His Gly Thr Asn Leu
Glu Asp Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Val His Tyr Ala Gln Phe Pro Tyr 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105166113PRTArtificial
SequenceSynthetic Construct 166Glu Val Gln Leu Val Glu Ser Gly Pro
Gly Leu Val Lys Pro Ser Glu1 5 10 15Thr Leu Ser Leu Thr Cys Thr Val
Ser Gly Phe Ser Leu Thr Asp Tyr 20 25 30Gly Val Leu Trp Ile Arg Gln
Pro Pro Gly Lys Gly Leu Glu Trp Ile 35 40 45Gly Met Ile Trp Ser Gly
Gly Thr Thr Asp Tyr Asn Ala Ala Phe Ile 50 55 60Ser Arg Val Thr Ile
Ser Val Asp Thr Ser Lys Asn Gln Phe Ser Leu65 70 75 80Lys Leu Ser
Ser Val Thr Ala Ala Asp Thr Ala Val Tyr Tyr Cys Val 85 90 95Arg Glu
Glu Met Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val Ser 100 105
110Ser167107PRTArtificial SequenceSynthetic Construct 167Asp Ile
Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Phe 20 25
30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45Tyr Tyr Thr Ser Arg Leu His Ser Gly Val Pro Ser Arg Phe Ser
Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly Asn
Thr Leu Pro Trp 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105168113PRTArtificial SequenceSynthetic Construct 168Glu Val
Gln Leu Val Glu Ser Gly Pro Gly Leu Val Lys Pro Ser Glu1 5 10 15Thr
Leu Ser Leu Thr Cys Thr Val Ser Gly Phe Ser Leu Thr Asp Tyr 20 25
30Gly Val Leu Trp Ile Arg Gln Pro Pro Gly Lys Gly Leu Glu Trp Ile
35 40 45Gly Met Ile Trp Ser Gly Gly Thr Thr Asp Tyr Asn Ala Ala Phe
Ile 50 55 60Ser Arg Val Thr Ile Ser Lys Asp Thr Ser Lys Asn Gln Val
Ser Leu65 70 75 80Lys Leu Ser Ser Val Thr Ala Ala Asp Thr Ala Val
Tyr Tyr Cys Val 85 90 95Arg Glu Glu Met Asp Tyr Trp Gly Gln Gly Thr
Leu Val Thr Val Ser 100 105 110Ser169107PRTArtificial
SequenceSynthetic Construct 169Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Phe 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
His Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly Asn Thr Leu Pro Trp 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105170113PRTArtificial
SequenceSynthetic Construct 170Glu Val Gln Leu Val Glu Ser Gly Pro
Gly Leu Val Lys Pro Ser Glu1 5 10 15Thr Leu Ser Leu Thr Cys Thr Val
Ser Gly Phe Ser Leu Thr Asp Tyr 20 25 30Gly Val Leu Trp Val Arg Gln
Pro Pro Gly Lys Gly Leu Glu Trp Leu 35 40 45Gly Met Ile Trp Ser Gly
Gly Thr Thr Asp Tyr Asn Ala Ala Phe Ile 50 55 60Ser Arg Leu Thr Ile
Ser Lys Asp Thr Ser Lys Asn Gln Val Ser Leu65 70 75 80Lys Leu Ser
Ser Val Thr Ala Ala Asp Thr Ala Val Tyr Tyr Cys Val 85 90 95Arg Glu
Glu Met Asp Tyr Trp Gly Gln Gly Thr Leu Val Thr Val Ser 100 105
110Ser171107PRTArtificial SequenceSynthetic Construct 171Asp Ile
Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Ile Thr Cys Arg Ala Ser Gln Asp Ile Ser Asn Phe 20 25
30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45Tyr Tyr Thr Ser Arg Leu His Ser Gly Val Pro Ser Arg Phe Ser
Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Gly Asn
Thr Leu Pro Trp 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 1051725PRTArtificial SequenceSynthetic ConstructVARIANT1Xaa = D
or EVARIANT2Xaa = S or A 172Xaa Xaa Tyr Met Ser1
517317PRTArtificial SequenceSynthetic ConstructVARIANT6Xaa = N or
SVARIANT7Xaa = A or GVARIANT8Xaa = D or SVARIANT9Xaa = A or S
173Asp Met Tyr Pro Asp Xaa Xaa Xaa Xaa Ser Tyr Asn Gln Lys Phe Arg1
5 10 15Glu1748PRTArtificial SequenceSynthetic ConstructVARIANT5Xaa
= Y or AVARIANT6Xaa = A or FVARIANT7Xaa = S or AVARIANT8Xaa = A or
V 174Ala Pro Arg Trp Xaa Xaa Xaa Xaa1 51759PRTArtificial
SequenceSynthetic ConstructVARIANT2Xaa = A or QVARIANT3Xaa = A or
GVARIANT4Xaa = A or HVARIANT5Xaa = A or TVARIANT6Xaa = A or
LVARIANT7Xaa = A or PVARIANT8Xaa = A or P 175Gln Xaa Xaa Xaa Xaa
Xaa Xaa Xaa Thr1 517617PRTArtificial SequenceSynthetic
ConstructVARIANT9Xaa = T, A or Q 176Val Ile Asn Pro Gly Ser Gly Asp
Xaa Tyr Tyr Ser Glu Lys Phe Lys1
5 10 15Gly1777PRTArtificial SequenceSynthetic ConstructVARIANT7Xaa
= S, E, or Q 177His Gly Thr Asn Leu Glu Xaa1 51789PRTArtificial
SequenceSynthetic ConstructVARIANT1Xaa = V or AVARIANT2Xaa = H or
AVARIANT9Xaa = Y or A 178Xaa Xaa Tyr Ala Gln Phe Pro Tyr Xaa1
5179107PRTArtificial SequenceSynthetic Construct 179Asp Ile Gln Met
Thr Gln Thr Thr Ser Ser Leu Ser Ala Ser Leu Gly1 5 10 15Asp Arg Val
Thr Ile Ser Cys Arg Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn
Trp Tyr Gln Gln Lys Pro Asp Gly Thr Val Lys Leu Leu Ile 35 40 45Tyr
Tyr Thr Ser Arg Leu Arg Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Lys Asp Tyr Phe Leu Thr Ile Ser Asn Leu Glu Gln65
70 75 80Glu Asp Val Ala Ala Tyr Phe Cys Gln Gln Gly His Thr Leu Pro
Pro 85 90 95Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile Lys 100
105180117PRTArtificial SequenceSynthetic Construct 180Glu Val Gln
Leu Gln Gln Ser Gly Pro Glu Leu Val Lys Pro Gly Ala1 5 10 15Ser Val
Lys Ile Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr
Met Ser Trp Val Lys Gln Ser His Gly Lys Thr Leu Glu Trp Ile 35 40
45Gly Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe
50 55 60Arg Glu Lys Val Thr Leu Thr Val Asp Lys Ser Ser Thr Thr Ala
Tyr65 70 75 80Met Glu Phe Arg Ser Leu Thr Ser Glu Asp Ser Ala Val
Tyr Tyr Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly
Thr Gly Thr Thr 100 105 110Val Thr Val Ser Ser
115181107PRTArtificial SequenceSynthetic Construct 181Asp Ile Leu
Met Thr Gln Ser Pro Ser Ser Met Ser Val Ser Leu Gly1 5 10 15Asp Thr
Val Ser Ile Thr Cys His Ala Ser Gln Asp Ile Ser Ser Tyr 20 25 30Ile
Val Trp Leu Gln Gln Lys Pro Gly Lys Ser Phe Arg Gly Leu Ile 35 40
45Tyr His Gly Thr Asn Leu Glu Asp Gly Ile Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Ala Asp Tyr Ser Leu Thr Ile Ser Ser Leu Glu
Ser65 70 75 80Glu Asp Phe Ala Asp Tyr Tyr Cys Val His Tyr Ala Gln
Phe Pro Tyr 85 90 95Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile Lys 100
105182114PRTArtificial SequenceSynthetic Construct 182Gln Val Gln
Leu Gln Gln Ser Gly Ala Glu Leu Val Arg Pro Gly Thr1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Ala Phe Thr Asn Tyr 20 25 30Leu
Ile Glu Trp Val Lys Gln Arg Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Val Ile Asn Pro Gly Ser Gly Asp Thr Tyr Tyr Ser Glu Lys Phe
50 55 60Lys Gly Lys Val Thr Leu Thr Ala Asp Lys Ser Ser Ser Thr Ala
Tyr65 70 75 80Met Gln Leu Ser Ser Leu Thr Ser Glu Asp Ser Ala Val
Tyr Phe Cys 85 90 95Ala Arg Asp Arg Leu Asp Tyr Trp Gly Gln Gly Thr
Thr Leu Thr Val 100 105 110Ser Ser183451PRTArtificial
SequenceSynthetic Construct 183Glu Val Gln Leu Val Glu Ser Gly Gly
Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala
Ser Gly Phe Thr Phe Ser Asn Tyr 20 25 30Thr Met Asn Trp Val Arg Gln
Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40 45Ser Ala Ile Ser Gly Ser
Gly Gly Ser Thr Tyr Tyr Ala Asp Ser Val 50 55 60Lys Gly Arg Phe Thr
Ile Ser Arg Asp Asn Ser Lys Asn Thr Leu Tyr65 70 75 80Leu Gln Met
Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Lys
Asp Arg Tyr Ser Gln Val His Tyr Ala Leu Asp Tyr Trp Gly 100 105
110Gln Gly Thr Leu Val Thr Val Ser Ser Ala Ser Thr Lys Gly Pro Ser
115 120 125Val Phe Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly Gly
Thr Ala 130 135 140Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu
Pro Val Thr Val145 150 155 160Ser Trp Asn Ser Gly Ala Leu Thr Ser
Gly Val His Thr Phe Pro Ala 165 170 175Val Leu Gln Ser Ser Gly Leu
Tyr Ser Leu Ser Ser Val Val Thr Val 180 185 190Pro Ser Ser Ser Leu
Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His 195 200 205Lys Pro Ser
Asn Thr Lys Val Asp Lys Arg Val Glu Pro Lys Ser Cys 210 215 220Asp
Lys Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu Leu Gly225 230
235 240Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr Leu
Met 245 250 255Ile Ser Arg Thr Pro Glu Val Thr Cys Val Val Val Asp
Val Ser His 260 265 270Glu Asp Pro Glu Val Lys Phe Asn Trp Tyr Val
Asp Gly Val Glu Val 275 280 285His Asn Ala Lys Thr Lys Pro Arg Glu
Glu Gln Tyr Asn Ser Thr Tyr 290 295 300Arg Val Val Ser Val Leu Thr
Val Leu His Gln Asp Trp Leu Asn Gly305 310 315 320Lys Glu Tyr Lys
Cys Lys Val Ser Asn Lys Ala Leu Pro Ala Pro Ile 325 330 335Glu Lys
Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val 340 345
350Tyr Thr Leu Pro Pro Ser Arg Glu Glu Met Thr Lys Asn Gln Val Ser
355 360 365Leu Thr Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala
Val Glu 370 375 380Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys
Thr Thr Pro Pro385 390 395 400Val Leu Asp Ser Asp Gly Ser Phe Phe
Leu Tyr Ser Lys Leu Thr Val 405 410 415Asp Lys Ser Arg Trp Gln Gln
Gly Asn Val Phe Ser Cys Ser Val Met 420 425 430His Glu Ala Leu His
Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser 435 440 445Pro Gly Lys
450184219PRTArtificial SequenceSynthetic Construct 184Asp Ile Val
Met Thr Gln Ser Pro Asp Ser Leu Pro Val Thr Pro Gly1 5 10 15Glu Pro
Ala Ser Ile Ser Cys Arg Ser Ser Gln Ser Leu Leu His Ser 20 25 30Asn
Gly Tyr Asn Tyr Leu Asp Trp Tyr Leu Gln Lys Ala Gly Gln Ser 35 40
45Pro Gln Leu Leu Ile Tyr Leu Gly Ser Asn Arg Ala Ser Gly Val Pro
50 55 60Asp Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Lys
Ile65 70 75 80Ser Arg Val Glu Ala Glu Asp Val Gly Val Tyr Tyr Cys
Gln Gln Tyr 85 90 95Tyr Asn His Pro Thr Thr Phe Gly Gln Gly Thr Lys
Leu Glu Ile Lys 100 105 110Arg Thr Val Ala Ala Pro Ser Val Phe Ile
Phe Pro Pro Ser Asp Glu 115 120 125Gln Leu Lys Ser Gly Thr Ala Ser
Val Val Cys Leu Leu Asn Asn Phe 130 135 140Tyr Pro Arg Glu Ala Lys
Val Gln Trp Lys Val Asp Asn Ala Leu Gln145 150 155 160Ser Gly Asn
Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser 165 170 175Thr
Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu 180 185
190Lys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser
195 200 205Pro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys 210
215185219PRTArtificial SequenceSynthetic Construct 185Asp Ile Gln
Met Thr Gln Ser Pro Asp Ser Leu Pro Val Thr Pro Gly1 5 10 15Glu Pro
Ala Ser Ile Ser Cys Arg Ser Ser Gln Ser Leu Leu His Ser 20 25 30Asn
Gly Tyr Asn Tyr Leu Asp Trp Tyr Leu Gln Lys Ala Gly Gln Ser 35 40
45Pro Gln Leu Leu Ile Tyr Leu Gly Ser Asn Arg Ala Ser Gly Val Pro
50 55 60Asp Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Lys
Ile65 70 75 80Ser Arg Val Glu Ala Glu Asp Val Gly Val Tyr Tyr Cys
Gln Gln Tyr 85 90 95Tyr Asn His Pro Thr Thr Phe Gly Gln Gly Thr Lys
Leu Glu Ile Lys 100 105 110Arg Thr Val Ala Ala Pro Ser Val Phe Ile
Phe Pro Pro Ser Asp Glu 115 120 125Gln Leu Lys Ser Gly Thr Ala Ser
Val Val Cys Leu Leu Asn Asn Phe 130 135 140Tyr Pro Arg Glu Ala Lys
Val Gln Trp Lys Val Asp Asn Ala Leu Gln145 150 155 160Ser Gly Asn
Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser 165 170 175Thr
Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu 180 185
190Lys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser
195 200 205Pro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys 210
215186450PRTArtificial SequenceSynthetic Construct 186Glu Val Gln
Leu Val Glu Ser Gly Gly Gly Leu Val His Pro Gly Gly1 5 10 15Ser Leu
Arg Leu Ser Cys Ala Gly Ser Gly Phe Thr Phe Ser Ser Tyr 20 25 30Ala
Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40
45Ser Ala Ile Gly Thr Gly Gly Gly Thr Tyr Tyr Ala Asp Ser Val Met
50 55 60Gly Arg Phe Thr Ile Ser Arg Asp Asn Ser Lys Asn Thr Leu Tyr
Leu65 70 75 80Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr
Tyr Cys Ala 85 90 95Arg Tyr Asp Asn Val Met Gly Leu Tyr Trp Phe Asp
Tyr Trp Gly Gln 100 105 110Gly Thr Leu Val Thr Val Ser Ser Ala Ser
Thr Lys Gly Pro Ser Val 115 120 125Phe Pro Leu Ala Pro Ser Ser Lys
Ser Thr Ser Gly Gly Thr Ala Ala 130 135 140Leu Gly Cys Leu Val Lys
Asp Tyr Phe Pro Glu Pro Val Thr Val Ser145 150 155 160Trp Asn Ser
Gly Ala Leu Thr Ser Gly Val His Thr Phe Pro Ala Val 165 170 175Leu
Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser Val Val Thr Val Pro 180 185
190Ser Ser Ser Leu Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His Lys
195 200 205Pro Ser Asn Thr Lys Val Asp Lys Arg Val Glu Pro Lys Ser
Cys Asp 210 215 220Lys Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu
Leu Leu Gly Gly225 230 235 240Pro Ser Val Phe Leu Phe Pro Pro Lys
Pro Lys Asp Thr Leu Met Ile 245 250 255Ser Arg Thr Pro Glu Val Thr
Cys Val Val Val Asp Val Ser His Glu 260 265 270Asp Pro Glu Val Lys
Phe Asn Trp Tyr Val Asp Gly Val Glu Val His 275 280 285Asn Ala Lys
Thr Lys Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg 290 295 300Val
Val Ser Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly Lys305 310
315 320Glu Tyr Lys Cys Lys Val Ser Asn Lys Ala Leu Pro Ala Pro Ile
Glu 325 330 335Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro
Gln Val Tyr 340 345 350Thr Leu Pro Pro Ser Arg Glu Glu Met Thr Lys
Asn Gln Val Ser Leu 355 360 365Thr Cys Leu Val Lys Gly Phe Tyr Pro
Ser Asp Ile Ala Val Glu Trp 370 375 380Glu Ser Asn Gly Gln Pro Glu
Asn Asn Tyr Lys Thr Thr Pro Pro Val385 390 395 400Leu Asp Ser Asp
Gly Ser Phe Phe Leu Tyr Ser Lys Leu Thr Val Asp 405 410 415Lys Ser
Arg Trp Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met His 420 425
430Glu Ala Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro
435 440 445Gly Lys 450187214PRTArtificial SequenceSynthetic
Construct 187Glu Ile Val Leu Thr Gln Ser Pro Ala Thr Leu Ser Leu
Ser Pro Gly1 5 10 15Glu Arg Ala Thr Leu Ser Cys Arg Ala Ser Gln Ser
Val Ser Ser Tyr 20 25 30Leu Ala Trp Tyr Gln Gln Lys Pro Gly Gln Ala
Pro Arg Leu Leu Ile 35 40 45Tyr Asp Ala Ser Asn Arg Ala Thr Gly Ile
Pro Ala Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu
Thr Ile Ser Ser Leu Glu Pro65 70 75 80Glu Asp Phe Ala Val Tyr Tyr
Cys Gln Gln Arg Ser Asn Trp Pro Pro 85 90 95Ala Phe Gly Gly Gly Thr
Lys Val Glu Ile Lys Arg Thr Val Ala Ala 100 105 110Pro Ser Val Phe
Ile Phe Pro Pro Ser Asp Glu Gln Leu Lys Ser Gly 115 120 125Thr Ala
Ser Val Val Cys Leu Leu Asn Asn Phe Tyr Pro Arg Glu Ala 130 135
140Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln Ser Gly Asn Ser
Gln145 150 155 160Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr
Tyr Ser Leu Ser 165 170 175Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr
Glu Lys His Lys Val Tyr 180 185 190Ala Cys Glu Val Thr His Gln Gly
Leu Ser Ser Pro Val Thr Lys Ser 195 200 205Phe Asn Arg Gly Glu Cys
210188118PRTArtificial SequenceSynthetic Construct 188Glu Val Gln
Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu
Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser Tyr 20 25 30Ser
Met Asn Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40
45Ser Tyr Ile Ser Ser Ser Ser Ser Thr Ile Asp Tyr Ala Asp Ser Val
50 55 60Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser Leu
Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Asp Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Arg Glu Ser Gly Trp Tyr Leu Phe Asp Tyr Trp
Gly Gln Gly Thr 100 105 110Leu Val Thr Val Ser Ser
115189107PRTArtificial SequenceSynthetic Construct 189Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Arg Ala Ser Gln Gly Ile Ser Ser Trp 20 25 30Leu
Ala Trp Tyr Gln Gln Lys Pro Glu Lys Ala Pro Lys Ser Leu Ile 35 40
45Tyr Ala Ala Ser Ser Leu Gln Ser Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Asn Ser
Tyr Pro Pro 85 90 95Thr Phe Gly Gly Gly Thr Lys Val Glu Ile Lys 100
105190124PRTArtificial SequenceSynthetic Construct 190Glu Val Gln
Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Arg1 5 10 15Ser Leu
Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Asp Asp Tyr 20 25 30Ala
Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40
45Ser Gly Ile Ser Trp Asn Ser Gly Ser Ile Gly Tyr Ala Asp Ser Val
50 55 60Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser Leu
Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Leu
Tyr Tyr Cys 85 90 95Ala Lys Asp Gln Ser Thr Ala Asp Tyr Tyr Phe Tyr
Tyr Gly Met Asp 100 105 110Val Trp Gly Gln Gly Thr Thr Val Thr Val
Ser Ser 115 120191106PRTArtificial SequenceSynthetic Construct
191Glu Ile Val
Val Thr Gln Ser Pro Ala Thr Leu Ser Leu Ser Pro Gly1 5 10 15Glu Arg
Ala Thr Leu Ser Cys Arg Ala Ser Gln Ser Val Ser Ser Tyr 20 25 30Leu
Ala Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro Arg Leu Leu Ile 35 40
45Tyr Asp Ala Ser Asn Arg Ala Thr Gly Ile Pro Ala Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Glu
Pro65 70 75 80Glu Asp Phe Ala Val Tyr Tyr Cys Gln Gln Arg Ser Asn
Trp Pro Thr 85 90 95Phe Gly Gln Gly Thr Lys Val Glu Ile Lys 100
105192122PRTArtificial SequenceSynthetic Construct 192Gln Val Gln
Leu Val Gln Ser Gly Ser Glu Leu Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Tyr 20 25 30Ser
Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Lys Trp Met 35 40
45Gly Trp Ile Asn Thr Glu Thr Gly Glu Pro Thr Tyr Ala Asp Asp Phe
50 55 60Lys Gly Arg Phe Val Phe Ser Leu Asp Thr Ser Val Ser Thr Ala
Tyr65 70 75 80Leu Gln Ile Ser Ser Leu Lys Ala Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Asn Pro Tyr Tyr Asp Tyr Val Ser Tyr Tyr Ala
Met Asp Tyr Trp 100 105 110Gly Gln Gly Thr Thr Val Thr Val Ser Ser
115 120193107PRTArtificial SequenceSynthetic Construct 193Asp Ile
Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Ile Thr Cys Lys Ala Ser Gln Asp Val Ser Thr Ala 20 25
30Val Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45Tyr Ser Ala Ser Tyr Leu Tyr Thr Gly Val Pro Ser Arg Phe Ser
Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Phe Thr Ile Ser Ser Leu
Gln Pro65 70 75 80Glu Asp Ile Ala Thr Tyr Tyr Cys Gln Gln His Tyr
Ser Thr Pro Arg 85 90 95Thr Phe Gly Gln Gly Thr Lys Leu Glu Ile Lys
100 105194120PRTArtificial SequenceSynthetic Construct 194Glu Val
Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser
Leu Arg Leu Ser Cys Ala Ala Ser Glu Tyr Glu Phe Pro Ser His 20 25
30Asp Met Ser Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Leu Val
35 40 45Ala Ala Ile Asn Ser Asp Gly Gly Ser Thr Tyr Tyr Pro Asp Thr
Met 50 55 60Glu Arg Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser
Leu Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala
Val Tyr Tyr Cys 85 90 95Ala Arg His Tyr Asp Asp Tyr Tyr Ala Trp Phe
Ala Tyr Trp Gly Gln 100 105 110Gly Thr Met Val Thr Val Ser Ser 115
120195111PRTArtificial SequenceSynthetic Construct 195Glu Ile Val
Leu Thr Gln Ser Pro Ala Thr Leu Ser Leu Ser Pro Gly1 5 10 15Glu Arg
Ala Thr Leu Ser Cys Arg Ala Ser Lys Ser Val Ser Thr Ser 20 25 30Gly
Tyr Ser Tyr Met His Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro 35 40
45Arg Leu Leu Ile Tyr Leu Ala Ser Asn Leu Glu Ser Gly Val Pro Ala
50 55 60Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile
Ser65 70 75 80Ser Leu Glu Pro Glu Asp Phe Ala Val Tyr Tyr Cys Gln
His Ser Arg 85 90 95Glu Leu Pro Leu Thr Phe Gly Gly Gly Thr Lys Val
Glu Ile Lys 100 105 110196469PRTArtificial SequenceSynthetic
Construct 196Met Tyr Leu Gly Leu Asn Tyr Val Phe Ile Val Phe Leu
Leu Asn Gly1 5 10 15Val Gln Ser Glu Val Lys Leu Glu Glu Ser Gly Gly
Gly Leu Val Gln 20 25 30Pro Gly Gly Ser Met Lys Leu Ser Cys Ala Ala
Ser Gly Phe Thr Phe 35 40 45Ser Asp Ala Trp Met Asp Trp Val Arg Gln
Ser Pro Glu Lys Gly Leu 50 55 60Glu Trp Val Ala Glu Ile Arg Ser Lys
Ala Asn Asn His Ala Thr Tyr65 70 75 80Tyr Ala Glu Ser Val Asn Gly
Arg Phe Thr Ile Ser Arg Asp Asp Ser 85 90 95Lys Ser Ser Val Tyr Leu
Gln Met Asn Ser Leu Arg Ala Glu Asp Thr 100 105 110Gly Ile Tyr Tyr
Cys Thr Trp Gly Glu Val Phe Tyr Phe Asp Tyr Trp 115 120 125Gly Gln
Gly Thr Thr Leu Thr Val Ser Ser Ala Ser Thr Lys Gly Pro 130 135
140Ser Val Phe Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly Gly
Thr145 150 155 160Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe Pro
Glu Pro Val Thr 165 170 175Val Ser Trp Asn Ser Gly Ala Leu Thr Ser
Gly Val His Thr Phe Pro 180 185 190Ala Val Leu Gln Ser Ser Gly Leu
Tyr Ser Leu Ser Ser Val Val Thr 195 200 205Val Pro Ser Ser Ser Leu
Gly Thr Gln Thr Tyr Ile Thr Cys Asn Val 210 215 220Asn His Lys Pro
Ser Asn Thr Lys Val Asp Lys Lys Val Glu Pro Lys225 230 235 240Ser
Cys Asp Lys Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu 245 250
255Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr
260 265 270Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys Val Val Val
Asp Val 275 280 285Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp Tyr
Val Asp Gly Val 290 295 300Glu Val His Asn Ala Lys Thr Lys Pro Arg
Glu Glu Gln Tyr Asn Ser305 310 315 320Thr Tyr Arg Val Val Ser Val
Leu Thr Val Leu His Gln Asp Trp Leu 325 330 335Asn Gly Lys Glu Tyr
Lys Cys Lys Val Ser Asn Lys Ala Leu Pro Ala 340 345 350Pro Ile Glu
Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro 355 360 365Gln
Val Tyr Thr Leu Pro Pro Ser Arg Asp Glu Leu Thr Lys Asn Gln 370 375
380Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile
Ala385 390 395 400Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn
Tyr Lys Thr Thr 405 410 415Pro Pro Val Leu Asp Ser Asp Gly Ser Phe
Phe Leu Tyr Ser Lys Leu 420 425 430Thr Val Asp Lys Ser Arg Trp Gln
Gln Gly Asn Val Phe Ser Cys Ser 435 440 445Val Met His Glu Ala Leu
His Asn His Tyr Thr Gln Lys Ser Leu Ser 450 455 460Leu Ser Pro Gly
Lys465197233PRTArtificial SequenceSynthetic Construct 197Met Arg
Pro Ser Ile Gln Phe Leu Gly Leu Leu Leu Phe Trp Leu His1 5 10 15Gly
Ala Gln Cys Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser 20 25
30Ala Ser Leu Gly Gly Lys Val Thr Ile Thr Cys Lys Ser Ser Gln Asp
35 40 45Ile Asn Lys Tyr Ile Ala Trp Tyr Gln His Lys Pro Gly Lys Gly
Pro 50 55 60Arg Leu Leu Ile His Tyr Thr Ser Thr Leu Gln Pro Gly Ile
Pro Ser65 70 75 80Arg Phe Ser Gly Ser Gly Ser Gly Arg Asp Tyr Ser
Phe Ser Ile Ser 85 90 95Asn Leu Glu Pro Glu Asp Ile Ala Thr Tyr Tyr
Cys Leu Gln Tyr Asp 100 105 110Asn Leu Leu Thr Phe Gly Ala Gly Thr
Lys Leu Glu Leu Lys Arg Thr 115 120 125Val Ala Ala Pro Ser Val Phe
Ile Phe Pro Pro Ser Asp Glu Gln Leu 130 135 140Lys Ser Gly Thr Ala
Ser Val Val Cys Leu Leu Asn Asn Phe Tyr Pro145 150 155 160Arg Glu
Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln Ser Gly 165 170
175Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr Tyr
180 185 190Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu
Lys His 195 200 205Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu
Ser Ser Pro Val 210 215 220Thr Lys Ser Phe Asn Arg Gly Glu Cys225
230198138PRTArtificial SequenceSynthetic Construct 198Met Tyr Leu
Gly Leu Asn Tyr Val Phe Ile Val Phe Leu Leu Asn Gly1 5 10 15Val Gln
Ser Glu Val Lys Leu Glu Glu Ser Gly Gly Gly Leu Val Gln 20 25 30Pro
Gly Gly Ser Met Lys Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe 35 40
45Ser Asp Ala Trp Met Asp Trp Val Arg Gln Ser Pro Glu Lys Gly Leu
50 55 60Glu Trp Val Ala Glu Ile Arg Ser Lys Ala Asn Asn His Ala Thr
Tyr65 70 75 80Tyr Ala Glu Ser Val Asn Gly Arg Phe Thr Ile Ser Arg
Asp Asp Ser 85 90 95Lys Ser Ser Val Tyr Leu Gln Met Asn Ser Leu Arg
Ala Glu Asp Thr 100 105 110Gly Ile Tyr Tyr Cys Thr Trp Gly Glu Val
Phe Tyr Phe Asp Tyr Trp 115 120 125Gly Gln Gly Thr Thr Leu Thr Val
Ser Ser 130 135199126PRTArtificial SequenceSynthetic Construct
199Met Arg Pro Ser Ile Gln Phe Leu Gly Leu Leu Leu Phe Trp Leu His1
5 10 15Gly Ala Gln Cys Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu
Ser 20 25 30Ala Ser Leu Gly Gly Lys Val Thr Ile Thr Cys Lys Ser Ser
Gln Asp 35 40 45Ile Asn Lys Tyr Ile Ala Trp Tyr Gln His Lys Pro Gly
Lys Gly Pro 50 55 60Arg Leu Leu Ile His Tyr Thr Ser Thr Leu Gln Pro
Gly Ile Pro Ser65 70 75 80Arg Phe Ser Gly Ser Gly Ser Gly Arg Asp
Tyr Ser Phe Ser Ile Ser 85 90 95Asn Leu Glu Pro Glu Asp Ile Ala Thr
Tyr Tyr Cys Leu Gln Tyr Asp 100 105 110Asn Leu Leu Thr Phe Gly Ala
Gly Thr Lys Leu Glu Leu Lys 115 120 125200119PRTArtificial
SequenceSynthetic Construct 200Glu Val Gln Leu Gln Gln Ser Gly Pro
Glu Leu Val Lys Pro Gly Ala1 5 10 15Ser Val Lys Met Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Val Met His Trp Val Lys Gln
Lys Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Tyr Ile Asn Pro Tyr
Asn Asp Gly Thr Lys Tyr Asn Glu Lys Phe 50 55 60Lys Gly Lys Ala Thr
Leu Thr Ser Asp Lys Ser Ser Ser Thr Ala Tyr65 70 75 80Met Glu Leu
Ser Ser Leu Thr Ser Glu Asp Ser Ala Val Tyr Tyr Cys 85 90 95Ala Asn
Tyr Tyr Gly Ser Ser Leu Ser Met Asp Tyr Trp Gly Gln Gly 100 105
110Thr Ser Val Thr Val Ser Ser 115201108PRTArtificial
SequenceSynthetic Construct 201Asp Ile Gln Met Thr Gln Thr Thr Ser
Ser Leu Ser Ala Ser Leu Gly1 5 10 15Asp Arg Val Thr Ile Ser Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Asp Gly Thr Val Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
His Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Tyr Ser Leu Thr Ile Ser Asn Leu Glu Gln65 70 75 80Glu Asp Ile
Ala Thr Tyr Phe Cys Gln Gln Gly Asn Thr Leu Pro Trp 85 90 95Thr Phe
Gly Gly Gly Thr Lys Leu Glu Ile Lys Arg 100 105202121PRTArtificial
SequenceSynthetic Construct 202Glu Val Gln Leu Gln Gln Ser Gly Pro
Glu Leu Val Lys Pro Gly Ala1 5 10 15Ser Val Lys Ile Ser Cys Lys Thr
Ser Gly Tyr Thr Phe Lys Asp Tyr 20 25 30Thr Met His Trp Val Lys Gln
Ser His Gly Lys Ser Leu Glu Trp Ile 35 40 45Gly Gly Ile Tyr Pro Asn
Asn Gly Gly Ser Thr Tyr Asn Gln Asn Phe 50 55 60Lys Asp Lys Ala Thr
Leu Thr Val Asp Lys Ser Ser Ser Thr Ala Tyr65 70 75 80Met Glu Phe
Arg Ser Leu Thr Ser Glu Asp Ser Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Met Gly Tyr His Gly Pro His Leu Asp Phe Asp Val Trp Gly 100 105
110Ala Gly Thr Thr Val Thr Val Ser Pro 115 120203108PRTArtificial
SequenceSynthetic Construct 203Asp Ile Val Met Thr Gln Ser His Lys
Phe Met Ser Thr Ser Leu Gly1 5 10 15Asp Arg Val Ser Ile Thr Cys Lys
Ala Ser Gln Asp Val Gly Ala Ala 20 25 30Val Ala Trp Tyr Gln Gln Lys
Pro Gly Gln Ser Pro Lys Leu Leu Ile 35 40 45Tyr Trp Ala Ser Thr Arg
His Thr Gly Val Pro Asp Arg Phe Thr Gly 50 55 60Gly Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Asn Val Gln Ser65 70 75 80Glu Asp Leu
Thr Asp Tyr Phe Cys Gln Gln Tyr Ile Asn Tyr Pro Leu 85 90 95Thr Phe
Gly Gly Gly Thr Lys Leu Glu Ile Lys Arg 100 105204119PRTArtificial
SequenceSynthetic Construct 204Gln Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Val Met His Trp Val Arg Gln
Ala Pro Gly Gln Arg Leu Glu Trp Met 35 40 45Gly Tyr Ile Asn Pro Tyr
Asn Asp Gly Thr Lys Tyr Asn Glu Lys Phe 50 55 60Lys Gly Arg Val Thr
Ile Thr Ser Asp Thr Ser Ala Ser Thr Ala Tyr65 70 75 80Met Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Asn
Tyr Tyr Gly Ser Ser Leu Ser Met Asp Tyr Trp Gly Gln Gly 100 105
110Thr Leu Val Thr Val Ser Ser 115205108PRTArtificial
SequenceSynthetic Construct 205Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
His Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Tyr Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln Gln Gly Asn Thr Leu Pro Trp 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys Arg 100 105206108PRTArtificial
SequenceSynthetic Construct 206Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Val Lys Leu Leu Ile 35 40 45Tyr Tyr Thr Ser Arg Leu
His Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Tyr Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Phe Cys Gln Gln Gly Asn Thr Leu Pro Trp 85 90 95Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys Arg 100 105207119PRTArtificial
SequenceSynthetic Construct 207Gln Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Val Met His Trp Val Arg Gln
Ala Pro Gly Gln Arg Leu Glu Trp Ile 35 40 45Gly Tyr Ile Asn Pro Tyr
Asn Asp Gly Thr Lys Tyr Asn Glu
Lys Phe 50 55 60Lys Gly Arg Ala Thr Ile Thr Ser Asp Thr Ser Ala Ser
Thr Ala Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Asn Tyr Tyr Gly Ser Ser Leu Ser Met
Asp Tyr Trp Gly Gln Gly 100 105 110Thr Leu Val Thr Val Ser Ser
115208119PRTArtificial SequenceSynthetic Construct 208Gln Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Val
Met His Trp Val Arg Gln Ala Pro Gly Gln Arg Leu Glu Trp Ile 35 40
45Gly Tyr Ile Asn Pro Tyr Asn Asp Gly Thr Lys Tyr Asn Glu Lys Phe
50 55 60Lys Gly Arg Ala Thr Leu Thr Ser Asp Lys Ser Ala Ser Thr Ala
Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Asn Tyr Tyr Gly Ser Ser Leu Ser Met Asp Tyr
Trp Gly Gln Gly 100 105 110Thr Leu Val Thr Val Ser Ser
115209121PRTArtificial SequenceSynthetic Construct 209Gln Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ser1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Lys Asp Tyr 20 25 30Thr
Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40
45Gly Gly Ile Tyr Pro Asn Asn Gly Gly Ser Thr Tyr Asn Gln Asn Phe
50 55 60Lys Asp Arg Val Thr Ile Thr Ala Asp Lys Ser Thr Ser Thr Ala
Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Arg Met Gly Tyr His Gly Pro His Leu Asp Phe
Asp Val Trp Gly 100 105 110Gln Gly Thr Thr Val Thr Val Ser Ser 115
120210108PRTArtificial SequenceSynthetic Construct 210Asp Ile Gln
Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Lys Ala Ser Gln Asp Val Gly Ala Ala 20 25 30Val
Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40
45Tyr Trp Ala Ser Thr Arg His Thr Gly Val Pro Ser Arg Phe Ser Gly
50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln
Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Ile Asn
Tyr Pro Leu 85 90 95Thr Phe Gly Gly Gly Thr Lys Val Glu Ile Lys Arg
100 105211108PRTArtificial SequenceSynthetic Construct 211Asp Ile
Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Ile Thr Cys Lys Ala Ser Gln Asp Val Gly Ala Ala 20 25
30Val Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45Tyr Trp Ala Ser Thr Arg His Thr Gly Val Pro Asp Arg Phe Ser
Gly 50 55 60Gly Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Ile
Asn Tyr Pro Leu 85 90 95Thr Phe Gly Gly Gly Thr Lys Val Glu Ile Lys
Arg 100 105212121PRTArtificial SequenceSynthetic Construct 212Gln
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ser1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Lys Asp Tyr
20 25 30Thr Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Gly Ile Tyr Pro Asn Asn Gly Gly Ser Thr Tyr Asn Gln
Asn Phe 50 55 60Lys Asp Arg Val Thr Leu Thr Ala Asp Lys Ser Thr Ser
Thr Ala Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Met Gly Tyr His Gly Pro His Leu
Asp Phe Asp Val Trp Gly 100 105 110Gln Gly Thr Thr Val Thr Val Ser
Ser 115 120213121PRTArtificial SequenceSynthetic Construct 213Gln
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ser1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Lys Asp Tyr
20 25 30Thr Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Gly Ile Tyr Pro Asn Asn Gly Gly Ser Thr Tyr Asn Gln
Asn Phe 50 55 60Lys Asp Arg Ala Thr Leu Thr Val Asp Lys Ser Thr Ser
Thr Ala Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Met Gly Tyr His Gly Pro His Leu
Asp Phe Asp Val Trp Gly 100 105 110Gln Gly Thr Thr Val Thr Val Ser
Ser 115 12021425PRTArtificial SequenceSynthetic Construct 214Glu
Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10
15Ser Leu Arg Leu Ser Cys Ala Ala Ser 20 2521513PRTArtificial
SequenceSynthetic Construct 215Trp Val Arg Gln Ala Pro Gly Lys Gly
Leu Glu Trp Val1 5 1021630PRTArtificial SequenceSynthetic Construct
216Arg Phe Thr Ile Ser Arg Asp Asn Ser Lys Asn Thr Leu Tyr Leu Gln1
5 10 15Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 20
25 3021711PRTArtificial SequenceSynthetic Construct 217Trp Gly Gln
Gly Thr Leu Val Thr Val Ser Ser1 5 1021823PRTArtificial
SequenceSynthetic Construct 218Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys
2021915PRTArtificial SequenceSynthetic Construct 219Trp Tyr Gln Gln
Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile Tyr1 5 10
1522032PRTArtificial SequenceSynthetic Construct 220Gly Val Pro Ser
Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe Thr1 5 10 15Leu Thr Ile
Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr Cys 20 25
3022110PRTArtificial SequenceSynthetic Construct 221Phe Gly Gln Gly
Thr Lys Val Glu Ile Lys1 5 1022217PRTArtificial SequenceSynthetic
Construct 222Asp Met Tyr Pro Asp Ala Ala Ala Ala Ser Tyr Asn Gln
Lys Phe Arg1 5 10 15Glu2238PRTArtificial SequenceSynthetic
Construct 223Ala Pro Arg Trp Ala Ala Ala Ala1 52249PRTArtificial
SequenceSynthetic Construct 224Gln Ala Ala Ala Ala Ala Ala Ala Thr1
5225117PRTArtificial SequenceSynthetic Construct 225Glu Val Gln Leu
Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met
Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly
Asp Met Tyr Pro Asp Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55
60Arg Glu Arg Val Thr Leu Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr65
70 75 80Leu Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Val Leu Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly
Thr Leu 100 105 110Val Thr Val Ser Ser 115226117PRTArtificial
SequenceSynthetic Construct 226Glu Val Gln Leu Val Gln Ser Gly Ala
Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala
Ser Gly Tyr Thr Phe Thr Asp Ser 20 25 30Tyr Met Ser Trp Val Arg Gln
Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Asp Met Tyr Pro Asp
Asn Gly Asp Ser Ser Tyr Asn Gln Lys Phe 50 55 60Arg Glu Arg Val Thr
Leu Thr Val Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Leu Glu Leu
Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Leu
Ala Pro Arg Trp Tyr Phe Ser Val Trp Gly Gln Gly Thr Leu 100 105
110Val Thr Val Ser Ser 115
D00001

D00002

D00003

D00004
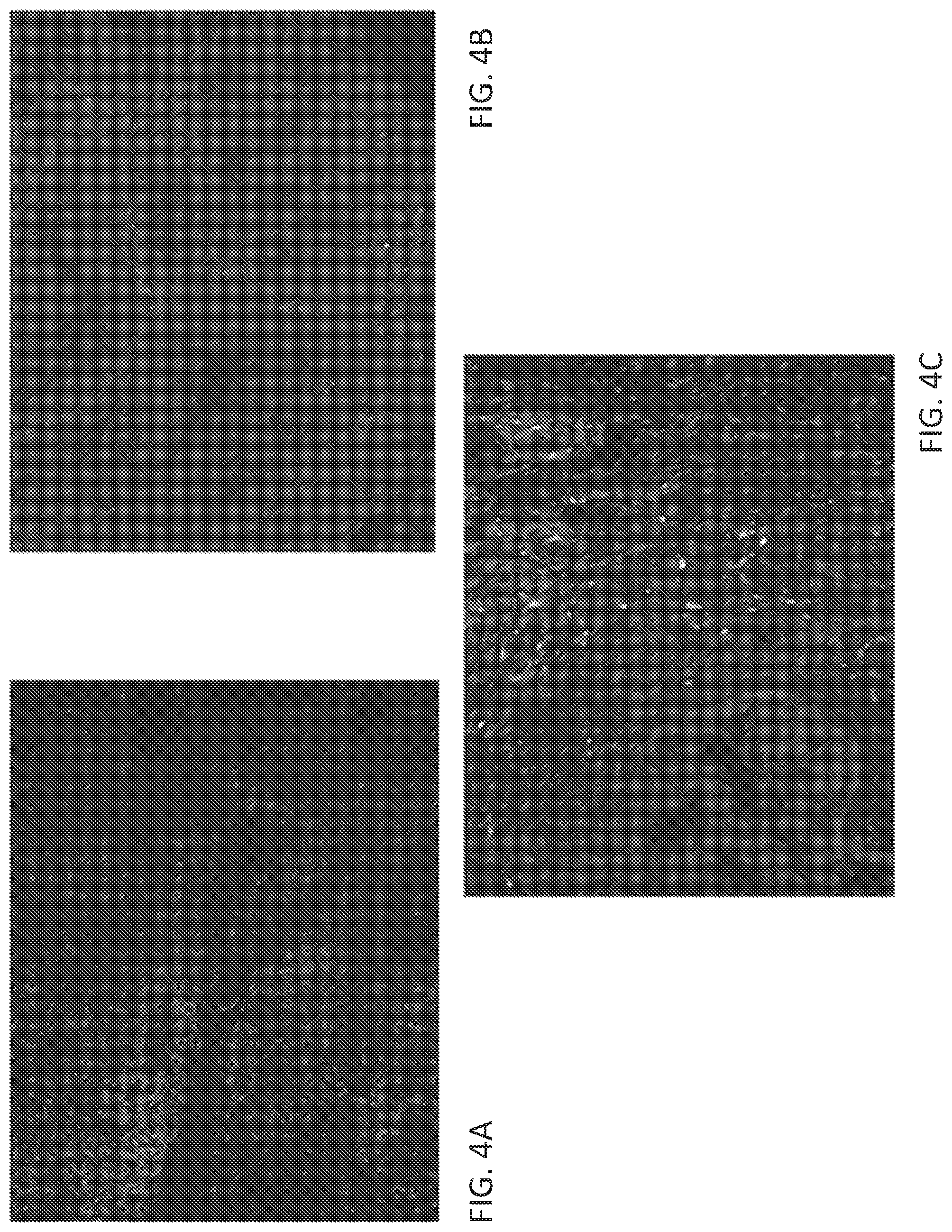
D00005

D00006

D00007

D00008

D00009

D00010

D00011

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.