Bispecific Anti-vegf/anti-ang-2 Antibodies And Their Use In The Treatment Of Ocular Vascular Diseases
Duerr; Harald ; et al.
U.S. patent application number 16/867449 was filed with the patent office on 2021-04-01 for bispecific anti-vegf/anti-ang-2 antibodies and their use in the treatment of ocular vascular diseases. This patent application is currently assigned to Roche Glycart AG. The applicant listed for this patent is Roche Glycart AG. Invention is credited to Harald Duerr, Frank Herting, Christian Klein, Joerg Thomas Regula, Matthias Rueth, Kay-Gunnar Stubenrauch.
| Application Number | 20210095013 16/867449 |
| Document ID | / |
| Family ID | 1000005263875 |
| Filed Date | 2021-04-01 |











View All Diagrams
| United States Patent Application | 20210095013 |
| Kind Code | A1 |
| Duerr; Harald ; et al. | April 1, 2021 |
BISPECIFIC ANTI-VEGF/ANTI-ANG-2 ANTIBODIES AND THEIR USE IN THE TREATMENT OF OCULAR VASCULAR DISEASES
Abstract
The present invention relates to bispecific antibody against human vascular endothelial growth factor (VEGF/VEGF-A) and against human angiopoietin-2 (ANG-2) of human IgG1 or IgG4 subclass with mutations I253A, H310A, and H435A, methods for their production, pharmaceutical compositions containing said antibodies, and uses thereof.
| Inventors: | Duerr; Harald; (Starnberg, DE) ; Herting; Frank; (Penzberg, DE) ; Klein; Christian; (Bonstetten, CH) ; Regula; Joerg Thomas; (Muenchen, DE) ; Rueth; Matthias; (Penzberg, DE) ; Stubenrauch; Kay-Gunnar; (Penzberg, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Roche Glycart AG Schlieren CH |
||||||||||
| Family ID: | 1000005263875 | ||||||||||
| Appl. No.: | 16/867449 | ||||||||||
| Filed: | May 5, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15603259 | May 23, 2017 | 10683345 | ||
| 16867449 | ||||
| 13940091 | Jul 11, 2013 | 9695233 | ||
| 15603259 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 2039/54 20130101; C07K 2317/33 20130101; C07K 2317/21 20130101; C07K 2317/35 20130101; C07K 2317/92 20130101; A61P 27/06 20180101; C07K 2317/565 20130101; C07K 16/22 20130101; A61K 2039/505 20130101; C07K 2317/31 20130101; C07K 2317/56 20130101; C07K 2317/52 20130101; A61P 27/02 20180101 |
| International Class: | C07K 16/22 20060101 C07K016/22; A61P 27/02 20060101 A61P027/02; A61P 27/06 20060101 A61P027/06 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 13, 2012 | EP | 12176299.1 |
Claims
1. A method for the reduction of the viscosity of an antibody wherein the antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass(derived from human origin and) wherein the method comprises the modification of the antibody constant heavy chain region of human IgG1 or human IgG4 subclass with the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat).
2. The method of claim 1, wherein the antibody is a bispecific antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, wherein i) said first antigen-binding site specifically binding to VEGF comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 1, a CDR2H region of SEQ ID NO: 2, and a CDR1H region of SEQ ID NO:3, and in the light chain variable domain a CDR3L region of SEQ ID NO: 4, a CDR2L region of SEQ ID NO:5, and a CDR1L region of SEQ ID NO:6; and ii) said second antigen-binding site specifically binding to ANG-2 comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 9, a CDR2H region of, SEQ ID NO: 10, and a CDR1H region of SEQ ID NO: 11, and in the light chain variable domain a CDR3L region of SEQ ID NO: 12, a CDR2L region of SEQ ID NO: 13, and a CDR1L region of SEQ ID NO: 14, and wherein iii) the bispecific antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat).
3. The method of claim 2, wherein the bispecific antibody comprises a constant heavy chain region of human IgG1 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat) and further comprising the mutations L234A , L235A and P329G (numbering according to EU Index of Kabat).
4. An antibody obtained by the method of any one of claims 1 to 3.
5. A bispecific antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, wherein i) said first antigen-binding site specifically binding to VEGF comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 1, a CDR2H region of SEQ ID NO: 2, and a CDR1H region of SEQ ID NO:3, and in the light chain variable domain a CDR3L region of SEQ ID NO: 4, a CDR2L region of SEQ ID NO:5, and a CDR1L region of SEQ ID NO:6; and ii) said second antigen-binding site specifically binding to ANG-2 comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 9, a CDR2H region of, SEQ ID NO: 10, and a CDR1H region of SEQ ID NO: 11, and in the light chain variable domain a CDR3L region of SEQ ID NO: 12, a CDR2L region of SEQ ID NO: 13, and a CDR1L region of SEQ ID NO: 14, and wherein iii) the bispecific antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat)
6. The bispecific antibody according to claim 5, wherein i) said first antigen-binding site specifically binding to VEGF comprises as heavy chain variable domain VH an amino acid sequence of SEQ ID NO: 7, and as light chain variable domain VL an amino acid sequence of SEQ ID NO: 8, and ii) said second antigen-binding site specifically binding to ANG-2 comprises as heavy chain variable domain VH an amino acid sequence of SEQ ID NO: 15, and as light chain variable domain VL an amino acid sequence of SEQ ID NO: 16.
7. The bispecific antibody according to any one of claims 5 to 6, wherein the constant heavy chain region under iii) is of IgG1 subclass
8. The bispecific antibody according to claim 6, wherein the constant heavy chain region of IgG1 subclass further comprises the mutations L234A , L235A and P329G (numbering according to EU Index of Kabat)
9. The bispecific antibody according to any one of claims 5 to 6, wherein the constant heavy chain region under iii) is of IgG4 subclass
10. The bispecific antibody according to claim 9, wherein the constant heavy chain region of IgG4 subclass further comprises the mutations S228P and L235E (numbering according to EU Index of Kabat)
11. The bispecific antibody according to claim 9, wherein the constant heavy chain region of IgG4 subclass further comprises the mutations S228P , L235E and P329G (numbering according to EU Index of Kabat)
12. A pharmaceutical composition comprising an antibody according to any one of claims 4 and 5 to 11.
13. A nucleic acid encoding a bispecific antibody according to any one of claims 5 to 11.
14. Expression vector containing said nucleic acid according claim 13 capable of expressing said nucleic acid in a prokaryotic or eukaryotic host cell.
15. A prokaryotic or eukaryotic host cell comprising a vector according to claim 14.
16. A method for the preparation of a bispecific antibody according to any one of claims 5 to 11 comprising the steps of a) transforming a host cell with vectors comprising nucleic acid molecules encoding said antibody; b) culturing the host cell under conditions that allow synthesis of said antibody molecule; and c) recovering said antibody molecule from said culture.
17. A bispecific antibody obtained by the method of claim 16.
18. A bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 25, of SEQ ID NO: 26, of SEQ ID NO: 27, and of SEQ ID NO: 28.
19. A bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 21, of SEQ ID NO: 22., of SEQ ID NO: 23., and of SEQ ID NO: 24.
20. A bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 29, of SEQ ID NO: 30, of SEQ ID NO: 31, and of SEQ ID NO: 32.
21. A method of treatment of patient suffering from ocular vascular diseases by administering an antibody according to any one of claims 4 and 5 to 11 to a patient in the need of such treatment.
22. The method of claim 21, wherein the antibody is administered via intravitreal application.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. application Ser. No. 15/603,259, filed May 23, 2017 which is a continuation of U.S. application Ser. No. 13/940,091, filed Jul. 11, 2013, now U.S. Pat. No. 9,695,233, which claims the benefit of priority under 35 USC 119(a) to European patent application number 12176299.1, filed 13 Jul. 2012, which are incorporated herein by reference in its entirety.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted in ASCII format via EFS-Web and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Mar. 18, 2020, is named Sequence_listing.txt and is 120,540 bytes in size.
FIELD OF THE INVENTION
[0003] The present invention relates a method for the reduction of the viscosity of an antibody (including a bispecific antibody) of human IgG1 or human IgG4 subclass, to bispecific antibodies against human vascular endothelial growth factor (VEGF/VEGF-A) and against human angiopoietin-2 (ANG-2), methods for their production, pharmaceutical compositions containing said antibodies, and uses thereof.
BACKGROUND OF THE INVENTION
[0004] Angiogenesis is implicated in the pathogenesis of a variety of disorders which include solid tumors, intraocular neovascular syndromes such as proliferative retinopathies or age-related macular degeneration (AMD), rheumatoid arthritis, and psoriasis (Folkman, J., et al., J. Biol. Chem. 267 (1992) 10931-10934; Klagsbrun, M., et al., Annu. Rev. Physiol. 53 (1991) 217-239; and Garner, A., Vascular diseases, in: Pathobiology of ocular disease, A dynamic approach, Garner, A., and Klintworth, G. K. (eds.), 2nd edition, Marcel Dekker, New York (1994), pp. 1625-1710).
[0005] Ranibizumab (trade name Lucentis.RTM.) is a monoclonal antibody fragment derived from the same parent murine antibody as bevacizumab (Avastin.RTM.). However, it has been affinity matured to provide stronger binding to VEGF-A (WO 98/45331). It is known that VEGF-A blocking may be related to some systemic toxicities, therefore ranibizumab is missing an Fc part to reduce the serum half live and consequently systemic toxicities. It is an anti-angiogenic agent that has been approved to treat the "wet" type of age-related macular degeneration (ARMD), a common form of age-related vision loss.
[0006] Corneal angiogenesis assays have shown that both ANG-1 and ANG-2 had similar effects, acting synergistically with VEGF to promote growth of new blood vessels. Asahara, T., et al., Circ. Res. 83 (1998) 233-40. The possibility that there was a dose-dependent endothelial response was raised by the observation that in vitro at high concentration, ANG-2 can also be pro-angiogenic (Kim, I., et al., Oncogene 19 (2000) 4549-52). At high concentration, ANG-2 acts as an apoptosis survival factor for endothelial cells during serum deprivation apoptosis through activation of Tie2 via PI-3 Kinase and Akt pathway (Kim, I., et al., Oncogene 19 (2000) 4549-52).
[0007] WO 2010/040508 A9 and WO 2011/117329 relate to bispecific anti-VEGF/anti-ANG-2 antibodies. WO 2008/132568 relates to fusion proteins binding to growth factors. WO 2009/136352 relates to anti-angiogenic compounds. WO 2009/080253 and WO 2011/117330 relates to bispecific bivalent antibody formats. WO 2010/069532 relates to Ang2 antibodies.
[0008] Ocular vascular diseases such as age related macular degeneration (ARMD) and diabetic retinopathy (DR) are due to abnormal choroidal or retinal neovascularization respectively. They are the leading causes of visual loss in industrialized nations. Since the retina consists of well-defined layers of neuronal, glial, and vascular elements, relatively small disturbances such as those seen in vascular proliferation or edema can lead to significant loss of visual function. Inherited retinal degenerations, such as Retinitis Pigmentosa (RP), are also associated with vascular abnormalities, such as arteriolar narrowing and vascular atrophy. They affect as many as 1 in 3500 individuals and are characterized by progressive night blindness, visual field loss, optic nerve atrophy, arteriolar attenuation, and central loss of vision often progressing to complete blindness.
[0009] Ischemic retinopathies are characterized by loss or dysfunction of the retinal vasculature which results in a reduction of blood flow and hypoxia. The retina responds to hypoxia by generating signals to grow new blood vessels, but these new vessels are usually fragile and disorganized. It is the growth of these abnormal new vessels that creates most of the threat to vision since they can leak, hemorrhage or lead to scarring that may end in retinal detachment. Current treatments for ischemic retinopathies seek to halt the growth of the pathological vessels but do not address the underlying ischemia that drives their growth. Furthermore, standard treatment for diabetic retinopathy, an ischemic retinopathy that affects millions, involves destruction of a portion of the retina with a laser in an attempt to stop new vessel growth and preserve central vision. Strategies have been employed to block the function of vascular endothelial growth factor (VEGF), a major promoter of vessel growth. In the short term, anti-VEGF therapy can improve vision, but it does not address the underlying ischemia and in fact may exacerbate this condition as it inhibits all vessel growth, including beneficial collaterals. There is also the serious concern of systemic exposure of these drugs in elderly and/or diabetic patients where new vessel growth may be required in ischemic brains, hearts or limbs.
[0010] Typically for ocular diseases via intravitreal application smaller antibody fragments like Fab or Fab(2) are often used as they have a low serum half-life and the risk of systemic toxicities is lower. However this smaller fragments typically have also lower intravitreal half-lifes (e.g. due to the faster diffusion into serum) and have to be dosed typically more often.
[0011] Kim et al, Molecular Vision, 15 (2009) 2803-2812 relates to full length antibodies administered intravitreally in the eye, wherein an IgG with FcRn binding was eliminated into the blood in wild-type mice, whereas an IgY with no FcRn binding was not eliminated into the blood system. Furthermore the IgG with FcRn binding was not eliminated into the blood system in FcRn knockdown-mice.
[0012] There is a need in the art for better means for treating and preventing various ocular vascular diseases such as ischemic retinopathies.
SUMMARY OF THE INVENTION
[0013] One aspect of the invention is method for the reduction of the viscosity of an antibody wherein the antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass (derived from human origin and) wherein the method comprises the modification of the antibody constant heavy chain region of human IgG1 or human IgG4 subclass with the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat).
[0014] In one embodiment of the invention said method is characterized in that the antibody is a bispecific antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, wherein [0015] i) said first antigen-binding site specifically binding to VEGF comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 1, a CDR2H region of SEQ ID NO: 2, and a CDR1H region of SEQ ID NO:3, and in the light chain variable domain a CDR3L region of SEQ ID NO: 4, a CDR2L region of SEQ ID NO:5, and a CDR1L region of SEQ ID NO:6; and [0016] ii) said second antigen-binding site specifically binding to ANG-2 comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 9, a CDR2H region of, SEQ ID NO: 10, and a CDR1H region of SEQ ID NO: 11, and in the light chain variable domain a CDR3L region of SEQ ID NO: 12, a CDR2L region of SEQ ID NO: 13, and a CDR1L region of SEQ ID NO: 14, and wherein [0017] iii) the bispecific antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat).
[0018] In one embodiment of the invention such method is characterized in that said bispecific antibody described above comprises a constant heavy chain region of human IgG1 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat) and further comprising the mutations L234A, L235A and P329G (numbering according to EU Index of Kabat).
[0019] One embodiment of the invention is an antibody obtained by such method.
[0020] One embodiment of the invention is the use of the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat) for the reduction of the viscosity of an antibody wherein the antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass(derived from human origin).
[0021] In one embodiment of the invention said use is characterized in that the antibody is a bispecific antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2,
[0022] wherein [0023] i) said first antigen-binding site specifically binding to VEGF comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 1, a CDR2H region of SEQ ID NO: 2, and a CDR1H region of SEQ ID NO:3, and in the light chain variable domain a CDR3L region of SEQ ID NO: 4, a CDR2L region of SEQ ID NO:5, and a CDR1L region of SEQ ID NO:6; and [0024] ii) said second antigen-binding site specifically binding to ANG-2 comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 9, a CDR2H region of, SEQ ID NO: 10, and a CDR1H region of SEQ ID NO: 11, and in the light chain variable domain a CDR3L region of SEQ ID NO: 12, a CDR2L region of SEQ ID NO: 13, and a CDR1L region of SEQ ID NO: 14, and wherein [0025] iii) the bispecific antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat).
[0026] In one embodiment of the invention said specific use is characterized in that the bispecific antibody comprises a constant heavy chain region of human IgG1 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat) and further comprising the mutations L234A, L235A and P329G (numbering according to EU Index of Kabat).
[0027] The invention is further directed to a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, wherein [0028] i) said first antigen-binding site specifically binding to VEGF comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 1, a CDR2H region of SEQ ID NO: 2, and a CDR1H region of SEQ ID NO:3, and in the light chain variable domain a CDR3L region of SEQ ID NO: 4, a CDR2L region of SEQ ID NO:5, and a CDR1L region of SEQ ID NO:6; and [0029] ii) said second antigen-binding site specifically binding to ANG-2 comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 9, a CDR2H region of, SEQ ID NO: 10, and a CDR1H region of SEQ ID NO: 11, and in the light chain variable domain a CDR3L region of SEQ ID NO: 12, a CDR2L region of SEQ ID NO: 13, and a CDR1L region of SEQ ID NO: 14,
[0030] and wherein [0031] iii) the bispecific antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat).
[0032] In one embodiment said bispecific antibody is characterized in that [0033] i) said first antigen-binding site specifically binding to VEGF comprises as heavy chain variable domain VH an amino acid sequence of SEQ ID NO: 7, and as light chain variable domain VL an amino acid sequence of SEQ ID NO: 8, and [0034] ii) said second antigen-binding site specifically binding to ANG-2 comprises as heavy chain variable domain VH an amino acid sequence of SEQ ID NO: 15, and as light chain variable domain VL an amino acid sequence of SEQ ID NO: 16.
[0035] In one embodiment said bispecific antibody is characterized in that the constant heavy chain region under iii) is of human IgG1 subclass. In one embodiment said bispecific antibody of IgG1 subclass is characterized in that the constant heavy chain region of IgG1 subclass further comprises the mutations L234A, L235A and P329G (numbering according to EU Index of Kabat).
[0036] In one embodiment said bispecific antibody is characterized in that the constant heavy chain region under iii) is of human IgG4 subclass. In one embodiment said bispecific antibody of IgG4 subclass is characterized in that the constant heavy chain region of IgG4 subclass further comprises the mutations S228P and L235E (numbering according to EU Index of Kabat). In one embodiment said bispecific antibody of IgG4 subclass is characterized in that the constant heavy chain region of IgG4 subclass further comprises the mutations 228P, L235E and P329G (numbering according to EU Index of Kabat).
[0037] Still further aspects of the invention are a pharmaceutical composition comprising said bispecific antibody, said pharmaceutical composition for use in the treatment of ocular vascular diseases, the use of said bispecific antibody for the manufacture of a medicament for the treatment of ocular vascular diseases, a method of treatment of patient suffering from ocular vascular diseases by administering said bispecific antibody to a patient in the need of such treatment. In one embodiment the bispecific antibody or the pharmaceutical composition comprising said bispecific antibody is administered via intravitreal application.
[0038] A further aspect of the invention is a nucleic acid molecule encoding a heavy and/or light chain of a bispecific antibody according to the invention.
[0039] The invention further provides expression vectors containing said nucleic acid according to the invention capable of expressing said nucleic acid in a prokaryotic or eukaryotic host cell, and host cells containing such vectors for the recombinant production of a bispecific antibody according to the invention.
[0040] The invention further comprises a prokaryotic or eukaryotic host cell comprising a vector according to the invention.
[0041] The invention further comprises a method for the production of a bispecific antibody according to the invention, characterized by expressing a nucleic acid according to the invention in a prokaryotic or eukaryotic host cell and recovering said bispecific antibody from said cell or the cell culture supernatant. One embodiment is a method for the preparation of a bispecific antibody according to the invention comprising the steps of
[0042] a) transforming a host cell with vectors comprising nucleic acid molecules encoding said antibody;
[0043] b) culturing the host cell under conditions that allow synthesis of said antibody molecule; and
[0044] c) recovering said antibody molecule from said culture.
[0045] The invention further comprises the antibody obtained by such method for the production of a bispecific antibody.
[0046] Accordingly one embodiment of the invention is a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 21, of SEQ ID NO: 22, of SEQ ID NO: 23, and of SEQ ID NO: 24.
[0047] Accordingly one embodiment of the invention is a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 25, of SEQ ID NO: 26, of SEQ ID NO: 27, and of SEQ ID NO: 28.
[0048] The antibodies according to the invention have highly valuable properties due to their specific modifications in the Fc part/constant region causing a benefit for a patient suffering from ocular vascular diseases. They show high stability in the intravitreal environment and slow diffusion from the eye (compared to smaller antibody fragments without a constant heavy chain region), where the actual disease is located and treated (so treatment schedule can potentially be improved compared to non-IgG like antibodies like e.g. Fab and (Fab)2 fragments). Surprisingly compared to unmodified IgG antibodies the half-life in the eye after intravitreal application of the antibodies with the mutations I253A, H310A, and H435A in the constant region (with no more FcRn binding) was similar (only slightly reduced) (Tables 17a and 18a and FIGS. 7D and 7E), whereas the diffusion from the eye into the blood serum was similar (Table 15 and FIG. 7B). This highly valuable as it is desired for the treatment of ocular vascular diseases related to ANG2 and/or VEGF it to eliminate VEGF and Ang2 from the eye (e. via the transportation into the blood serum as anti-ANG2/ANG2 antibody complex or anti-VEGF/VEGF antibody complex). The antibodies according to the invention are cleared on the other hand quite rapidly from serum when compared to unmodified IgG antibodies (which is highly desired to reduce potential side effects arising from systemic exposure).
[0049] Surprisingly they also show lower viscosity (see FIG. 2) (compared to versions without the mutations I253A, H310A, and H435A in the constant region) and are therefore especially useful for intravitreal application through thin needles during the treatment of eye diseases (for such application typically thin needles are used and high viscosity makes an appropriate application rather difficult). The lower viscosity also allows higher concentration formulations.
[0050] Also surprisingly the antibodies according to the invention show a lower aggregation tendency (FIG. 4) during storage (compared to versions without the mutations I253A, H310A, and H435A in the Fc part) which is critical for intravitreal application in the eye (as an aggregation in the eye can lead to complications during such treatment). The bispecific antibodies according to the invention show good efficacy in inhibition of vascular diseases.
[0051] In certain embodiments, the bispecific antibodies according to the invention due to their specific modifications in the constant region (e.g. P329G LALA) show valuable properties like no binding Fcgamma receptors which reduces the risk of side effects like thrombosis and/or unwanted cell death (due to e.g. ADCC).
DESCRIPTION OF THE FIGURES
[0052] FIG. 1 Scheme of concept and advantages of <VEGF-ANG-2> IgG1 or IgG4 antibodies with AAA mutations (mutations I253A, H310A, and H435A -numbering according to EU Index of Kabat).
[0053] FIG. 2 Small-scale DLS-based viscosity measurement Extrapolated viscosity at 150 mg/mL in 200 mM Arginine/Succinate, pH 5.5 (comparison of <VEGF-ANG-2> antibodies according to the invention VEGFang2-0016 (with AAA mutations) with a reference VEGFang2-0015 (without such AAA mutations).
[0054] FIG. 3 DLS Aggregation depending on temperature (including DLS aggregation onset temperature) in 20 mM His, 140 mM NaCl, pH 6.0 5 (comparison of <VEGF-ANG-2> antibodies according to the invention VEGFang2-0016 (with AAA mutations) with a reference VEGFang2-0015 (without such AAA mutations).
[0055] FIG. 4 7 day storage at 40.degree. C. at 100 mg/ml (Decrease of Main and High Molecular Weight/HMW) increase) (comparison of <VEGF-ANG-2> antibodies according to the invention VEGFang2-0016 (with AAA mutations) which showed a lower aggregation with a reference VEGFang2-0015 (without such AAA mutations)).
[0056] FIG. 5A FcRn steady state affinity of VEGFang2-0015 (without AAA mutations): overlay of Biacore sensogramms at different concentrations shows a concentration dependent binding of VEGFang2-0015 (without AAA mutations) to FcRn.
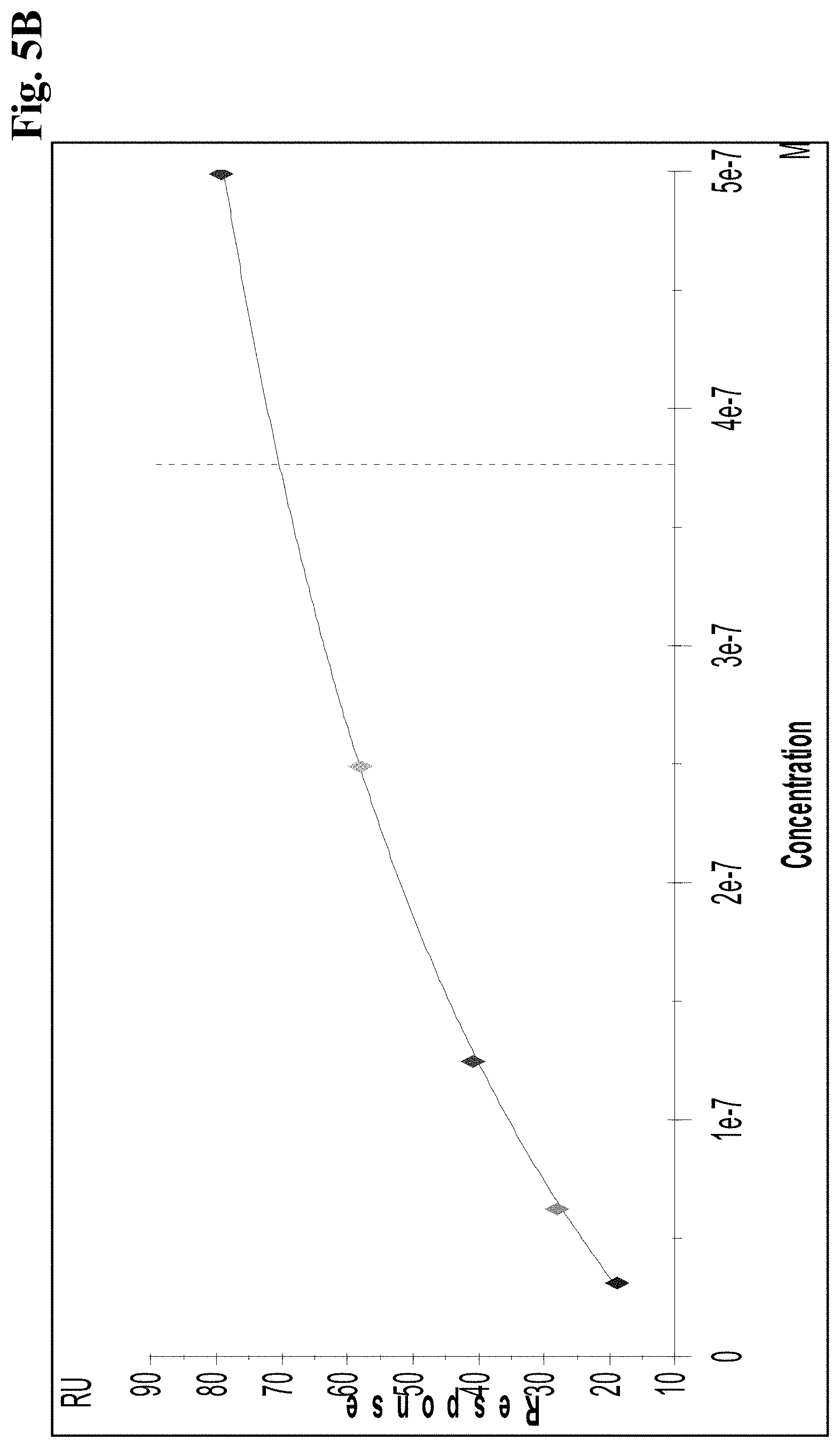
[0057] FIG. 5B FcRn steady state affinity of A: VEGFang2-0015 (without AAA mutations): the concentration dependent binding response curve of VEGFang2-0015 (without AAA mutations) shows binding to FcRn.
[0058] FIG. 5C FcRn steady state affinity of VEGFang2-0016 (with AAA mutations): overlay of Biacore sensogramms at different concentrations shows no binding to FcRn at all concentrations.
[0059] FIG. 5D FcRn steady state affinity of VEGFang2-0016 (with AAA mutations): the concentration dependent binding response curve of VEGFang2-0016 (with AAA mutations) shows no binding to FcRn.
[0060] FIG. 5E FcRn steady state affinity of VEGFang2-0016 (with AAA mutations): the concentration dependent binding response curve of VEGFang2-0016 (with AAA mutations) shows no binding to FcRn (Response range from -0.6 to 0.2 RU/concentration scale ranges from 0 to 0.35 M).
[0061] FIG. 6 FcgammaRIIIa interaction of VEGFang2-0015 without AAA mutations and VEGFang2-0016 with AAA mutations measurement (both are IgG1 subclass with P329G LALA mutations; as controls an Anti-Dig of IgG1 subclass and a IgG4 based antibody was used).
[0062] FIG. 7A Schematic Pk-ELISA Assay Principle for determination of concentrations of <VEGF/Ang2> bispecific antibodies in serum and whole eye lysates.
[0063] FIG. 7B Serum concentration after intravenous application: Comparison of compounds -VEGFang2-0015 without AAA mutations and VEGFang2-0016 with AAA mutations.
[0064] FIG. 7C Serum concentration after intravitreal application: Comparison of compounds -VEGFang2-0015 without AAA mutations and VEGFang2-0016 with AAA mutations.
[0065] FIG. 7D Eye lysates concentration of VEGFang2-0016 (with AAA mutation) in right and left eye (after intravitreal application only into the right eye in comparison to intravenous application): Significant concentrations could be detected only in the right eye after intravitreal application. After intravenous application no concentrations in eye lysates could be detected due to the low serum half-life of VEGFang2-0016 (with AAA mutation).
[0066] FIG. 7E Eye lysates concentration of VEGFang2-0015 (without AAA mutation) in right and left eye (after intravitreal application only into the right eye in comparison to intravenous application): In the right eye (and to some extent in the left eye) after intravitreal application concentrations of VEGFang2-0015 could be detected. This indicates the diffusion from the right eye into serum and from there into the left eye, which can be explained by the long half-life of VEGFang2-0015 (without AAA mutation). After intravenous application also significant concentrations in eye lysates of both eyes could be detected due to diffusion into the eyes of the serum-stable VEGFang2-0015 (without AAA mutation).
DETAILED DESCRIPTION OF THE INVENTION
[0067] In one embodiment of the invention the bispecific antibody according to the invention is bivalent.
[0068] In one aspect of the invention such bispecific, bivalent antibody according to the invention is characterized in comprising [0069] a) the heavy chain and the light chain of a first full length antibody that specifically binds to VEGF; [0070] b) the modified heavy chain and modified light chain of a second full length antibody that specifically binds to ANG-2, wherein the constant domains CL and CH1 are replaced by each other.
[0071] This bispecific, bivalent antibody format for the bispecific antibody specifically binding to human vascular endothelial growth factor (VEGF) and human angiopoietin-2 (ANG-2) is described in WO 2009/080253 (including Knobs-into-Holes modified CH3 domains). The antibodies based on this bispecific, bivalent antibody format are named CrossMabs.
[0072] In one embodiment such bispecific, bivalent antibody is characterized in comprising [0073] a) as heavy chain of the first full length antibody the amino acid sequence of SEQ ID NO: 25, and as light chain of the first full length antibody the amino acid sequence of SEQ ID NO: 27, and [0074] b) as modified heavy chain of the second full length antibody the amino acid sequence of SEQ ID NO: 26, and as modified light chain of the second full length antibody the amino acid sequence of SEQ ID NO: 28.
[0075] In one embodiment such bispecific, bivalent antibody is characterized in comprising [0076] a) as heavy chain of the first full length antibody the amino acid sequence of SEQ ID NO: 21, and as light chain of the first full length antibody the amino acid sequence of SEQ ID NO: 23, and [0077] b) as modified heavy chain of the second full length antibody the amino acid sequence of SEQ ID NO: 22, and as modified light chain of the second full length antibody the amino acid sequence of SEQ ID NO: 24.
[0078] In one embodiment such bispecific, bivalent antibody is characterized in comprising [0079] a) as heavy chain of the first full length antibody the amino acid sequence of SEQ ID NO: 29, and as light chain of the first full length antibody the amino acid sequence of SEQ ID NO: 31, and [0080] b) as modified heavy chain of the second full length antibody the amino acid sequence of SEQ ID NO: 30, and as modified light chain of the second full length antibody the amino acid sequence of SEQ ID NO: 32.
[0081] Accordingly one embodiment of the invention is a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 25, of SEQ ID NO: 26, of SEQ ID NO: 27, and of SEQ ID NO: 28.
[0082] Accordingly one embodiment of the invention is a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 21, of SEQ ID NO: 22, of SEQ ID NO: 23, and of SEQ ID NO: 24.
[0083] Accordingly one embodiment of the invention is a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 29, of SEQ ID NO: 30, of SEQ ID NO: 31, and of SEQ ID NO: 32.
[0084] In another aspect of the invention the bispecific antibody according to the invention is characterized in comprising [0085] a) the heavy chain and the light chain of a first full length antibody that specifically binds to VEGF; [0086] b) the heavy chain and the light chain of a second full length antibody that specifically binds to ANG-2, wherein the N-terminus of the heavy chain is connected to the C-terminus of the light chain via a peptide linker.
[0087] This bispecific, bivalent antibody format for this bispecific antibody specifically binding to human vascular endothelial growth factor (VEGF) and human angiopoietin-2 (ANG-2) is described in WO 2011/117330 including Knobs-into-Holes modified CH3 domains. The antibodies based on this bispecific, bivalent antibody format are named OAscFabs.
[0088] In one embodiment such bispecific, bivalent antibody is characterized in comprising [0089] a) as heavy chain of the first full length antibody the amino acid sequence of SEQ ID NO: 33, and as light chain of the first full length antibody the amino acid sequence of SEQ ID NO: 35, and [0090] b) as heavy chain of the second full length antibody connected to the light chain of the second full length antibody via a peptide linker the amino acid sequence of SEQ ID NO: 34.
[0091] In one embodiment such bispecific, bivalent antibody is characterized in comprising [0092] a) as heavy chain of the first full length antibody the amino acid sequence of SEQ ID NO: 36, and as light chain of the first full length antibody the amino acid sequence of SEQ ID NO: 38, and [0093] b) as heavy chain of the second full length antibody connected to the light chain of the second full length antibody via a peptide linker the amino acid sequence of SEQ ID NO: 37.
[0094] In one embodiment the antibody heavy chain variable domain (VH) and the antibody light chain variable domain (VL) of the heavy and light chain of the second full length antibody are disulfide stabilized by introduction of a disulfide bond between the following positions: heavy chain variable domain position 44 to light chain variable domain position 100 (numbering always according to EU index of Kabat (Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991)). Such further disulfide stabilization is achieved by the introduction of a disulfide bond between the variable domains VH and VL of the second full length antibody heavy and light chain. Techniques to introduce unnatural disulfide bridges for stabilization are described e.g. in WO 94/029350, Rajagopal, V., et al, Prot. Engin. 10 (1997) 1453-59; Kobayashi et al., Nuclear Medicine & Biology 25 (1998) 387-393; or Schmidt, M., et al., Oncogene 18 (1999) 1711-1721.
[0095] Accordingly one embodiment of the invention is a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 33, of SEQ ID NO: 34, and of SEQ ID NO: 35.
[0096] Accordingly one embodiment of the invention is a bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 36, of SEQ ID NO: 37, and of SEQ ID NO: 38.
[0097] In one embodiment the CH3 domains of the bispecific, bivalent antibody according to the invention is altered by the "knob-into-holes" technology which is described in detail with several examples in e.g. WO 96/027011, Ridgway J. B., et al., Protein Eng 9 (1996) 617-621; and Merchant, A. M., et al., Nat Biotechnol 16 (1998) 677-681. In this method the interaction surfaces of the two CH3 domains are altered to increase the heterodimerisation of both heavy chains containing these two CH3 domains. Each of the two CH3 domains (of the two heavy chains) can be the "knob", while the other is the "hole". The introduction of a disulfide bridge stabilizes the heterodimers (Merchant, A.M, et al., Nature Biotech 16 (1998) 677-681; Atwell, S., et al. J. Mol. Biol. 270 (1997) 26-35) and increases the yield.
[0098] In a preferred aspect of the invention all bispecific antibodies according to the invention are characterized in that
[0099] the CH3 domain of one heavy chain and the CH3 domain of the other heavy chain each meet at an interface which comprises an original interface between the antibody CH3 domains;
[0100] wherein said interface is altered to promote the formation of the bispecific antibody, wherein the alteration is characterized in that:
[0101] a) the CH3 domain of one heavy chain is altered,
[0102] so that within the original interface the CH3 domain of one heavy chain that meets the original interface of the CH3 domain of the other heavy chain within the bispecific antibody,
[0103] an amino acid residue is replaced with an amino acid residue having a larger side chain volume, thereby generating a protuberance within the interface of the CH3 domain of one heavy chain which is positionable in a cavity within the interface of the CH3 domain of the other heavy chain
[0104] and
[0105] b) the CH3 domain of the other heavy chain is altered,
[0106] so that within the original interface of the second CH3 domain that meets the original interface of the first CH3 domain within the bispecific antibody
[0107] an amino acid residue is replaced with an amino acid residue having a smaller side chain volume, thereby generating a cavity within the interface of the second CH3 domain within which a protuberance within the interface of the first CH3 domain is positionable.
[0108] Thus the antibody according to invention is preferably characterized in that [0109] the CH3 domain of the heavy chain of the full length antibody of a) and the CH3 domain of the heavy chain of the full length antibody of b) each meet at an interface which comprises an alteration in the original interface between the antibody CH3 domains; [0110] wherein i) in the CH3 domain of one heavy chain [0111] an amino acid residue is replaced with an amino acid residue having a larger side chain volume, thereby generating a protuberance within the interface of the CH3 domain of one heavy chain which is positionable in a cavity within the interface of the CH3 domain of the other heavy chain [0112] and wherein [0113] ii) in the CH3 domain of the other heavy chain [0114] an amino acid residue is replaced with an amino acid residue having a smaller side chain volume, thereby generating a cavity within the interface of the second CH3 domain within which a protuberance within the interface of the first CH3 domain is positionable.
[0115] Preferably said amino acid residue having a larger side chain volume is selected from the group consisting of arginine (R), phenylalanine (F), tyrosine (Y), tryptophan (W).
[0116] Preferably said amino acid residue having a smaller side chain volume is selected from the group consisting of alanine (A), serine (S), threonine (T), valine (V).
[0117] In one aspect of the invention both CH3 domains are further altered by the introduction of cysteine (C) as amino acid in the corresponding positions of each CH3 domain such that a disulfide bridge between both CH3 domains can be formed.
[0118] In one embodiment, the bispecific antibody comprises a T366W mutation in the CH3 domain of the "knobs chain" and T366S, L368A, Y407V mutations in the CH3 domain of the "hole chain". An additional interchain disulfide bridge between the CH3 domains can also be used (Merchant, A. M, et al., Nature Biotech 16 (1998) 677-681) e.g. by introducing a Y349C mutation into the CH3 domain of the "knobs chain" and a E356C mutation or a S354C mutation into the CH3 domain of the "hole chain".
[0119] In another embodiment, the bispecific antibody according to the invention comprises Y349C, T366W mutations in one of the two CH3 domains and E356C, T366S, L368A, Y407V mutations in the other of the two CH3 domains. In a another preferred embodiment the bispecific antibody comprises Y349C, T366W mutations in one of the two CH3 domains and S354C, T366S, L368A, Y407V mutations in the other of the two CH3 domains (the additional Y349C mutation in one CH3 domain and the additional E356C or S354C mutation in the other CH3 domain forming a interchain disulfide bridge) (numbering always according to EU index of Kabat (Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991)). But also other knobs-in-holes technologies as described by
[0120] EP 1 870 459 Al, can be used alternatively or additionally. Thus another example for the bispecific antibody are R409D; K370E mutations in the CH3 domain of the "knobs chain" and D399K; E357K mutations in the CH3 domain of the "hole chain" (numbering always according to EU index of Kabat (Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991)).
[0121] In another embodiment the bispecific antibody comprises a T366W mutation in the CH3 domain of the "knobs chain" and T366S, L368A, Y407V mutations in the CH3 domain of the "hole chain" and additionally R409D; K370E mutations in the CH3 domain of the "knobs chain" and D399K; E357K mutations in the CH3 domain of the "hole chain".
[0122] In another embodiment the bispecific antibody comprises Y349C, T366W mutations in one of the two CH3 domains and S354C, T366S, L368A, Y407V mutations in the other of the two CH3 domains or said trivalent, bispecific antibody comprises Y349C, T366W mutations in one of the two CH3 domains and S354C, T366S, L368A, Y407V mutations in the other of the two CH3 domains and additionally R409D; K370E mutations in the CH3 domain of the "knobs chain" and D399K; E357K mutations in the CH3 domain of the "hole chain".
[0123] In one embodiment of the invention the bispecific antibody according to the invention is characterized in having one or more of the following properties (determined in assays as described in Example 6 [0124] shows a lower serum concentration compared to corresponding bispecific antibody without the mutations described under iii) (96 hours after intravitreal application in mice, which are mouse FcRn deficient, but hemizygous transgenic for human FcRn); [0125] shows a similar (factor 0.8 to 1.2) concentration in whole right eye lysates compared to corresponding bispecific antibody without the mutations described under iii) (in mice, which are mouse FcRn deficient, but hemizygous transgenic for human FcRn, 96 hours after intravitreal application in the right eye).
[0126] In one embodiment the bispecific, bivalent antibody is characterized in comprising
[0127] a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in that [0128] i) said first antigen-binding site comprises as heavy chain variable domain (VH) the SEQ ID NO: 7, and as light chain variable domain (VL) the SEQ ID NO: 8; and [0129] ii) said second antigen-binding site comprises as heavy chain variable domain (VH) the SEQ ID NO: 15, and as light chain variable domain (VL) the SEQ ID NO: 16; and [0130] iii) the bispecific antibody comprises a constant heavy chain region of IgG1 or IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat) and having one or more of the following properties (determined in assays as described in Example 6 [0131] shows a lower serum concentration compared to corresponding bispecific antibody without the mutations described under iii) (96 hours after intravitreal application in mice, which are mouse FcRn deficient, but hemizygous transgenic for human FcRn); [0132] shows a similar (factor 0.8 to 1.2) concentration in whole right eye lysates compared to corresponding bispecific antibody without the mutations described under iii) (in mice, which are mouse FcRn deficient, but hemizygous transgenic for human FcRn, 96 hours after intravitreal application in the right eye).
[0133] In one embodiment the bispecific antibody is characterized in comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in that [0134] i) said first antigen-binding site comprises as heavy chain variable domain (VH) the SEQ ID NO: 7 with 1, 2 or 3 amino acid residue substitutions, and as light chain variable domain (VL) the SEQ ID NO: 8 with 1, 2 or 3 amino acid residue substitutions; and [0135] ii) said second antigen-binding site comprises as heavy chain variable domain (VH) the SEQ ID NO: 15 with 1, 2 or 3 amino acid residue substitutions, and as light chain variable domain (VL) the SEQ ID NO: with 1, 2 or 3 amino acid residue substitutions; and [0136] iii) the bispecific antibody comprises a constant heavy chain region of IgG1 or IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat)
[0137] and having one or more of the following properties (determined in assays as described in Example 6 [0138] shows a lower serum concentration compared to corresponding bispecific antibody without the mutations described under iii) (96 hours after intravitreal application in mice, which are mouse FcRn deficient, but hemizygous transgenic for human FcRn); [0139] shows a similar (factor 0.8 to 1.2) concentration in whole right eye lysates compared to corresponding bispecific antibody without the mutations described under iii) (in mice, which are mouse FcRn deficient, but hemizygous transgenic for human FcRn, 96 hours after intravitreal application in the right eye).
[0140] As used herein, "antibody" refers to a binding protein that comprises antigen-binding sites. The terms "binding site" or "antigen-binding site" as used herein denotes the region(s) of an antibody molecule to which a ligand actually binds. The term "antigen-binding site" comprises an antibody heavy chain variable domains (VH) and an antibody light chain variable domains (VL) (pair of VH/VL)).
[0141] Antibody specificity refers to selective recognition of the antibody for a particular epitope of an antigen. Natural antibodies, for example, are monospecific.
[0142] "Bispecific antibodies" according to the invention are antibodies which have two different antigen-binding specificities. Antibodies of the present invention are specific for two different antigens, VEGF as first antigen and ANG-2 as second antigen.
[0143] The term "monospecific" antibody as used herein denotes an antibody that has one or more binding sites each of which bind to the same epitope of the same antigen.
[0144] The term "valent" as used within the current application denotes the presence of a specified number of binding sites in an antibody molecule. As such, the terms "bivalent", "tetravalent", and "hexavalent" denote the presence of two binding site, four binding sites, and six binding sites, respectively, in an antibody molecule. The bispecific antibodies according to the invention are preferably "bivalent".
[0145] The term "VEGF" as used herein refers to human vascular endothelial growth factor (VEGF/VEGF-A,) the 165-amino acid human vascular endothelial cell growth factor (amino acid 27-191 of precursor sequence of human VEGF165: SEQ
[0146] ID NO: 17; amino acids 1-26 represent the signal peptide), and related 121, 189, and 206 vascular endothelial cell growth factor isoforms, as described by Leung, D. W., et al., Science 246 (1989) 1306-9; Houck et al., Mol. Endocrin. 5 (1991) 1806 -1814; Keck, P. J., et al., Science 246 (1989) 1309-12 and Connolly, D. T., et al., J. Biol. Chem. 264 (1989) 20017-24; together with the naturally occurring allelic and processed forms of those growth factors. VEGF is involved in the regulation of normal and abnormal angiogenesis and neovascularization associated with tumors and intraocular disorders (Ferrara, N., et al., Endocr. Rev. 18 (1997) 4-25; Berkman, R. A.,et al., J. Clin. Invest. 91 (1993) 153-159; Brown, L. F., et al., Human Pathol. 26 (1995) 86-91; Brown, L. F., et al., Cancer Res. 53 (1993) 4727-4735; Mattern, J., et al., Brit. J. Cancer. 73 (1996) 931-934; and Dvorak, H. F., et al., Am. J. Pathol. 146 (1995) 1029-1039). VEGF is a homodimeric glycoprotein that has been isolated from several sources and includes several isoforms. VEGF shows highly specific mitogenic activity for endothelial cells.
[0147] The term "ANG-2" as used herein refers to human angiopoietin-2 (ANG-2) (alternatively abbreviated with ANGPT2 or ANG2) (SEQ ID NO: 18) which is described e.g. in Maisonpierre, P. C., et al, Science 277 (1997) 55-60 and Cheung, A. H., et al., Genomics 48 (1998) 389-91. The angiopoietins-1 (SEQ ID NO: 19) and -2 were discovered as ligands for the Ties, a family of tyrosine kinases that is selectively expressed within the vascular endothelium (Yancopoulos, G. D., et al., Nature 407 (2000) 242-48). There are now four definitive members of the angiopoietin family. Angiopoietin-3 and -4 (Ang-3 and Ang-4) may represent widely diverged counterparts of the same gene locus in mouse and man (Kim, I., et al., FEBS Let, 443 (1999) 353-56; Kim, I., et al., J Biol Chem 274 (1999) 26523-28). ANG-1 and ANG-2 were originally identified in tissue culture experiments as agonist and antagonist, respectively (see for ANG-1: Davis, S., et al., Cell 87 (1996) 1161-69; and for ANG-2: Maisonpierre, P. C., et al., Science 277 (1997) 55-60). All of the known angiopoietins bind primarily to Tie2 (SEQ ID NO: 20), and both Ang-1 and -2 bind to Tie2 with an affinity of 3 nM (Kd) (Maisonpierre, P. C., et al., Science 277 (1997) 55-60).
[0148] An antigen-binding sites of the bispecific antibody of the invention contain six complementarity determining regions (CDRs) which contribute in varying degrees to the affinity of the binding site for antigen. There are three heavy chain variable domain CDRs (CDRH1, CDRH2 and CDRH3) and three light chain variable domain CDRs (CDRL1, CDRL2 and CDRL3). The extent of CDR and framework regions (FRs) is determined by comparison to a compiled database of amino acid sequences in which those regions have been defined according to variability among the sequences.
[0149] The antibodies of the invention comprise immunoglobulin constant regions derived from human origin of one or more immunoglobulin classes, wherein such. immunoglobulin classes include IgG, IgM, IgA, IgD, and IgE classes and, in the case of IgG and IgA, their subclasses, especially IgG1 and IgG4.
[0150] The terms "monoclonal antibody" or "monoclonal antibody composition" as used herein refer to a preparation of antibody molecules of a single amino acid composition.
[0151] The term "chimeric antibody" refers to an antibody comprising a variable region, i.e., binding region, from one source or species and at least a portion of a constant region derived from a different source or species, usually prepared by recombinant DNA techniques. Chimeric antibodies comprising a murine variable region and a human constant region are preferred. Other preferred forms of "chimeric antibodies" encompassed by the present invention are those in which the constant region has been modified or changed from that of the original antibody to generate the properties according to the invention, especially in regard to C1q binding and/or Fc receptor (FcR) binding. Such chimeric antibodies are also referred to as "class-switched antibodies.". Chimeric antibodies are the product of expressed immunoglobulin genes comprising DNA segments encoding immunoglobulin variable regions and DNA segments encoding immunoglobulin constant regions. Methods for producing chimeric antibodies involve conventional recombinant DNA and gene transfection techniques are well known in the art. See, e.g., Morrison, S. L., et al., Proc. Natl. Acad. Sci. USA 81 (1984) 6851-6855; U.S. Pat. No. 5,202,238 and U.S. Pat. No. 5,204,244.
[0152] The term "humanized antibody" refers to antibodies in which the framework or "complementarity determining regions" (CDR) have been modified to comprise the CDR of an immunoglobulin of different specificity as compared to that of the parent immunoglobulin. In a preferred embodiment, a murine CDR is grafted into the framework region of a human antibody to prepare the "humanized antibody." See, e.g., Riechmann, L., et al., Nature 332 (1988) 323-327; and Neuberger, M. S., et al., Nature 314 (1985) 268-270. Particularly preferred CDRs correspond to those representing sequences recognizing the antigens noted above for chimeric antibodies. Other forms of "humanized antibodies" encompassed by the present invention are those in which the constant region has been additionally modified or changed from that of the original antibody to generate the properties according to the invention, especially in regard to C1q binding and/or Fc receptor (FcR) binding.
[0153] The term "human antibody", as used herein, is intended to include antibodies having variable and constant regions derived from human germ line immunoglobulin sequences. Human antibodies are well-known in the state of the art (van Dijk, M. A., and van de Winkel, J. G., Curr. Opin. Chem. Biol. 5 (2001) 368-374). Human antibodies can also be produced in transgenic animals (e.g., mice) that are capable, upon immunization, of producing a full repertoire or a selection of human antibodies in the absence of endogenous immunoglobulin production. Transfer of the human germ-line immunoglobulin gene array in such germ-line mutant mice will result in the production of human antibodies upon antigen challenge (see, e.g., Jakobovits, A., et al., Proc. Natl. Acad. Sci. USA 90 (1993) 2551-2555; Jakobovits, A., et al., Nature 362 (1993) 255-258; Brueggemann, M., et al., Year Immunol. 7 (1993) 33-40). Human antibodies can also be produced in phage display libraries (Hoogenboom, H. R., and Winter, G., J. Mol. Biol. 227 (1992) 381-388; Marks, J. D., et al., J. Mol. Biol. 222 (1991) 581-597). The techniques of Cole, A., et al. and Boerner, P., et al. are also available for the preparation of human monoclonal antibodies (Cole, A., et al., Monoclonal Antibodies and Cancer Therapy, Liss, A. L., p. 77 (1985); and Boerner, P., et al., J. Immunol. 147 (1991) 86-95). As already mentioned for chimeric and humanized antibodies according to the invention the term "human antibody" as used herein also comprises such antibodies which are modified in the constant region to generate the properties according to the invention, especially in regard to C1q binding and/or FcR binding, e.g. by "class switching" i.e. change or mutation of Fc parts (e.g. from IgG1 to IgG4 and/or IgG1/IgG4 mutation).
[0154] The term "recombinant antibody", as used herein, is intended to include all human antibodies that are prepared, expressed, created or isolated by recombinant means, such as antibodies isolated from a host cell such as a NS0 or CHO cell or from an animal (e.g. a mouse) that is transgenic for human immunoglobulin genes or antibodies expressed using a recombinant expression vector transfected into a host cell. Such recombinant antibodies have variable and constant regions in a rearranged form. The recombinant antibodies according to the invention have been subjected to in vivo somatic hypermutation. Thus, the amino acid sequences of the VH and VL regions of the recombinant antibodies are sequences that, while derived from and related to human germ line VH and VL sequences, may not naturally exist within the human antibody germ line repertoire in vivo.
[0155] The "variable domain" (variable domain of a light chain (VL), variable domain of a heavy chain (VH) as used herein denotes each of the pair of light and heavy chains which is involved directly in binding the antibody to the antigen. The domains of variable human light and heavy chains have the same general structure and each domain comprises four framework (FR) regions whose sequences are widely conserved, connected by three "hypervariable regions" (or complementarity determining regions, CDRs). The framework regions adopt a .beta.-sheet conformation and the CDRs may form loops connecting the .beta.-sheet structure. The CDRs in each chain are held in their three-dimensional structure by the framework regions and form together with the CDRs from the other chain the antigen binding site. The antibody heavy and light chain CDR3 regions play a particularly important role in the binding specificity/affinity of the antibodies according to the invention and therefore provide a further object of the invention.
[0156] The terms "hypervariable region" or "antigen-binding portion of an antibody" when used herein refer to the amino acid residues of an antibody which are responsible for antigen-binding. The hypervariable region comprises amino acid residues from the "complementarity determining regions" or "CDRs". "Framework" or "FR" regions are those variable domain regions other than the hypervariable region residues as herein defined. Therefore, the light and heavy chains of an antibody comprise from N- to C-terminus the domains FR1, CDR1, FR2, CDR2, FR3, CDR3, and FR4. CDRs on each chain are separated by such framework amino acids. Especially, CDR3 of the heavy chain is the region which contributes most to antigen binding. CDR and FR regions are determined according to the standard definition of Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991).
[0157] As used herein, the term "binding" or "specifically binding" refers to the binding of the antibody to an epitope of the antigen (either human VEGF or human ANG-2) in an in vitro assay, preferably in an plasmon resonance assay (BlAcore, GE-Healthcare Uppsala, Sweden with purified wild-type antigen. The affinity of the binding is defined by the terms ka (rate constant for the association of the antibody from the antibody/antigen complex), k.sub.D (dissociation constant), and K.sub.D (k.sub.D/ka). In one embodiment binding or specifically binding means a binding affinity (K.sub.D) of 10.sup.-8 mol/l or less, in one embodiment 10.sup.-9 M to 10.sup.-13 mol/l.
[0158] The term "epitope" includes any polypeptide determinant capable of specific binding to an antibody. In certain embodiments, epitope determinant include chemically active surface groupings of molecules such as amino acids, sugar side chains, phosphoryl, or sulfonyl, and, in certain embodiments, may have specific three dimensional structural characteristics, and or specific charge characteristics. An epitope is a region of an antigen that is bound by an antibody.
[0159] In certain embodiments, an antibody is said to specifically bind an antigen when it preferentially recognizes its target antigen in a complex mixture of proteins and/or macromolecules.
[0160] The term "full length antibody" denotes an antibody consisting of two "full length antibody heavy chains" and two "full length antibody light chains". A "full length antibody heavy chain" is a polypeptide consisting in N-terminal to C-terminal direction of an antibody heavy chain variable domain (VH), an antibody constant heavy chain domain 1 (CH1), an antibody hinge region (HR), an antibody heavy chain constant domain 2 (CH2), and an antibody heavy chain constant domain 3 (CH3), abbreviated as VH-CH1-HR-CH2-CH3; and optionally an antibody heavy chain constant domain 4 (CH4) in case of an antibody of the subclass IgE. Preferably the "full length antibody heavy chain" is a polypeptide consisting in N-terminal to C-terminal direction of VH, CH1, HR, CH2 and CH3. A "full length antibody light chain" is a polypeptide consisting in N-terminal to C-terminal direction of an antibody light chain variable domain (VL), and an antibody light chain constant domain (CL), abbreviated as VL-CL. The antibody light chain constant domain (CL) can be .kappa. (kappa) or .lamda. (lambda). The two full length antibody chains are linked together via inter-polypeptide disulfide bonds between the CL domain and the CH1 domain and between the hinge regions of the full length antibody heavy chains. Examples of typical full length antibodies are natural antibodies like IgG (e.g. IgG 1 and IgG2), IgM, IgA, IgD, and IgE. The full length antibodies according to the invention can be from a single species e.g. human, or they can be chimerized or humanized antibodies. The full length antibodies according to the invention comprise two antigen binding sites each formed by a pair of VH and VL, which both specifically bind to the same antigen. The C-terminus of the heavy or light chain of said full length antibody denotes the last amino acid at the C-terminus of said heavy or light chain. The N-terminus of the heavy or light chain of said full length antibody denotes the last amino acid at the N-terminus of said heavy or light chain.
[0161] The term "peptide linker" as used within the invention denotes a peptide with amino acid sequences, which is preferably of synthetic origin. These peptides according to invention are used to connect the C-terminus of the light chain to the N-terminus of heavy chain of the second full length antibody (that specifically binds to a second antigen) via a peptide linker. The peptide linker within the second full length antibody heavy and light chain is a peptide with an amino acid sequence with a length of at least 30 amino acids, preferably with a length of 32 to 50 amino acids. In one the peptide linker is a peptide with an amino acid sequence with a length of 32 to 40 amino acids. In one embodiment said linker is (GxS)n with G=glycine, S=serine, (x=3, n=8, 9 or 10 and m=0, 1, 2 or 3) or (x=4 and n=6, 7 or 8 and m=0, 1, 2 or 3), preferably with x=4, n=6 or 7 and m=0, 1, 2 or 3, more preferably with x=4, n=7 and m=2. In one embodiment said linker is (G.sub.4S).sub.6G.sub.2.
[0162] The term "constant region" as used within the current applications denotes the sum of the domains of an antibody other than the variable region. The constant region is not involved directly in binding of an antigen, but exhibits various effector functions. Depending on the amino acid sequence of the constant region of their heavy chains, antibodies are divided in the classes: IgA, IgD, IgE, IgG and IgM, and several of these may be further divided into subclasses, such as IgG1, IgG2, IgG3, and IgG4, IgA1 and IgA2. The heavy chain constant regions that correspond to the different classes of antibodies are called .alpha., .delta., .epsilon., .gamma., and .mu., respectively. The light chain constant regions which can be found in all five antibody classes are called .kappa. (kappa) and .lamda. (lambda).
[0163] The terms "constant region derived from human origin" or " human constant region" as used in the current application denotes a constant heavy chain region of a human antibody of the subclass IgG1, IgG2, IgG3, or IgG4 and/or a constant light chain kappa or lambda region. Such constant regions are well known in the state of the art and e.g. described by Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991) (see also e.g. Johnson, G., and Wu, T. T., Nucleic Acids Res. 28 (2000) 214-218; Kabat, E. A., et al., Proc. Natl. Acad. Sci. USA 72 (1975) 2785-2788). Within the application for the numbering of positions and mutations the EU numbering system (EU Index) according to Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991) is used and referred to as "numbering according to EU Index of Kabat".
[0164] In one embodiment the bispecific antibodies according to the invention have a constant region of human IgG1 subclass (derived from human IgG1 subclass) .
[0165] In one embodiment the bispecific antibodies according to the invention have a constant region of human IgG4 subclass (derived from human IgG1 subclass).
[0166] In one embodiment the bispecific antibody according to the invention is of human IgG1 subclass with mutations L234A (Leu234Ala), L235A (Leu235Ala) and P329G (Pro329Gly). Such antibody has a reduced FcR binding (especially they show no more binding to FcRgammaI, FcRgammaII and FcRgammaIII). This especially useful to reduce potential side effects like e.g. thrombosis (Meyer, T., et al., J. Thromb. Haemost. 7 (2009) 171-81). In one embodiment the bispecific antibody according to the invention is of human IgG4 subclass with mutations S228P (Ser228Pro), L235E (Leu235Glu) and P329G (Pro329Gly). Such antibody shows reduced FcR binding as indicated above. While Pro329Ala mutation which was described already removes only two third of the FcgammaRIIIa sandwich interaction, the Pro329Gly in the antibodies according to the invention fully imparts binding of the Fc part to FcgammaRIII. This is especially useful as the binding to FcgammaRIII is involved in ADCC (antibody--dependent cellular toxicity) which leads to cell death, which may be helpful in the treatment of cancer diseases, but which can cause serious side effect in the antibody based treatment of other vascular or immunological diseases. So the antibodies according to the invention of IgG1 subclass with mutations L234A, L235A and P329G and IgG4 subclass with mutations S228P, L235E and P329G are especially useful, as they both show no more binding to FcRgammaI, FcRgammaII and FcRgammaIII.
[0167] The term "with (the) mutations AAA" as used herein refers the mutations I253A (Ile253Ala), H310A (His310Ala), and H435A (His435Ala) in the constant heavy chain region of IgG1 or IgG4, wherein the numbering is according to the EU Index of Kabat.
[0168] The term "with (the) mutations P329G LALA" as used herein refers to the mutations L234A (Leu234Ala), L235A (Leu235Ala) and P329G (Pro329Gly) in the constant heavy chain region of IgG1 subclass, wherein the numbering is according to the EU Index of Kabat. The term "with (the) mutations SPLE" as used herein refers to the S228P (Ser228Pro) and L235E (Leu235Glu) the constant heavy chain region of IgG4 subclass, wherein the numbering is according to the EU Index of Kabat. The term "with (the) mutations SPLE and P239G" as used herein refers to the S228P (Ser228Pro), L235E (Leu235Glu) and P329G (Pro329Gly) the constant heavy chain region of IgG4 subclass, wherein the numbering is according to the EU Index of Kabat.
[0169] The antibody according to the invention is produced by recombinant means. Thus, one aspect of the current invention is a nucleic acid encoding the antibody according to the invention and a further aspect is a cell comprising said nucleic acid encoding an antibody according to the invention. Methods for recombinant production are widely known in the state of the art and comprise protein expression in prokaryotic and eukaryotic cells with subsequent isolation of the antibody and usually purification to a pharmaceutically acceptable purity. For the expression of the antibodies as aforementioned in a host cell, nucleic acids encoding the respective modified light and heavy chains are inserted into expression vectors by standard methods. Expression is performed in appropriate prokaryotic or eukaryotic host cells like CHO cells, NSO cells, SP2/0 cells, HEK293 cells, COS cells, PER.C6 cells, yeast, or E.coli cells, and the antibody is recovered from the cells (supernatant or cells after lysis). General methods for recombinant production of antibodies are well-known in the state of the art and described, for example, in the review articles of Makrides, S. C., Protein Expr. Purif 17 (1999) 183-202; Geisse, S., et al., Protein Expr. Purif. 8 (1996) 271-282; Kaufman, R. J., Mol. Biotechnol. 16 (2000) 151-160; Werner, R. G., Drug Res. 48 (1998) 870-880.
[0170] Accordingly one embodiment of the invention is a method for the preparation of a bispecific antibody according to the invention, comprising the steps of [0171] a) transforming a host cell with vectors comprising nucleic acid molecules encoding said antibody; [0172] b) culturing the host cell under conditions that allow synthesis of said antibody molecule; and [0173] c) recovering said antibody molecule from said culture.
[0174] In one embodiment the recovering step under c includes the use of a light chain constant domain specific capture reagent (which e.g. specific for the kappa or the lambda constant light chain, depending on whether a kappa or a lambda light chain in the bispecific antibody according to invention used). In one embodiment this light chain specific capture reagent is used in in a bind-and-elute-mode). Examples of such light chain constant domain specific capture reagents are e.g. KappaSelect.TM. and LambdaFabSelect.TM. from GE Healthcare/BAC, which are based on a highly rigid agarose base matrix that allows high flow rates and low back pressure at large scale. They feature a ligand that binds to the constant region of the kappa or the lambda light chain respectively (i.e. fragments lacking the constant region of the light chain will not bind; FIG. 1). Both are therefore capable of binding other target molecules containing the constant region of the light chain, for example, IgG, IgA and IgM. The ligands are attached to the matrix via a long hydrophilic spacer arm to make it easily available for binding to the target molecule. They are based on a single-chain antibody fragment that is screened for either human Ig kappa or lambda.
[0175] The bispecific antibodies are suitably separated from the culture medium by conventional immunoglobulin purification procedures such as, for example, protein A-Sepharose, hydroxylapatite chromatography, gel electrophoresis, dialysis, or affinity chromatography. DNA and RNA encoding the monoclonal antibodies is readily isolated and sequenced using conventional procedures. The hybridoma cells can serve as a source of such DNA and RNA. Once isolated, the DNA may be inserted into expression vectors, which are then transfected into host cells such as HEK 293 cells, CHO cells, or myeloma cells that do not otherwise produce immunoglobulin protein, to obtain the synthesis of recombinant monoclonal antibodies in the host cells.
[0176] Amino acid sequence variants (or mutants) of the bispecific antibody are prepared by introducing appropriate nucleotide changes into the antibody DNA, or by nucleotide synthesis. Such modifications can be performed, however, only in a very limited range. For example, the modifications do not alter the above mentioned antibody characteristics such as the IgG subclass and antigen binding, but may improve the yield of the recombinant production, protein stability or facilitate the purification.
[0177] The term "host cell" as used in the current application denotes any kind of cellular system which can be engineered to generate the antibodies according to the current invention. In one embodiment HEK293 cells and CHO cells are used as host cells. As used herein, the expressions "cell," "cell line," and "cell culture" are used interchangeably and all such designations include progeny. Thus, the words "transformants" and "transformed cells" include the primary subject cell and cultures derived therefrom without regard for the number of transfers. It is also understood that all progeny may not be precisely identical in DNA content, due to deliberate or inadvertent mutations. Variant progeny that have the same function or biological activity as screened for in the originally transformed cell are included.
[0178] Expression in NSO cells is described by, e.g., Barnes, L. M., et al., Cytotechnology 32 (2000) 109-123; Barnes, L. M., et al., Biotech. Bioeng. 73 (2001) 261-270. Transient expression is described by, e.g., Durocher, Y., et al., Nucl. Acids. Res. 30 (2002) E9. Cloning of variable domains is described by Orlandi, R., et al., Proc. Natl. Acad. Sci. USA 86 (1989) 3833-3837; Carter, P., et al., Proc. Natl. Acad. Sci. USA 89 (1992) 4285-4289; and Norderhaug, L., et al., J. Immunol. Methods 204 (1997) 77-87. A preferred transient expression system (HEK 293) is described by Schlaeger, E.-J., and Christensen, K., in Cytotechnology 30 (1999) 71-83 and by Schlaeger, E.-J., in J. Immunol. Methods 194 (1996) 191-199.
[0179] The control sequences that are suitable for prokaryotes, for example, include a promoter, optionally an operator sequence, and a ribosome binding site. Eukaryotic cells are known to utilize promoters, enhancers and polyadenylation signals.
[0180] A nucleic acid is "operably linked" when it is placed in a functional relationship with another nucleic acid sequence. For example, DNA for a pre-sequence or secretory leader is operably linked to DNA for a polypeptide if it is expressed as a pre-protein that participates in the secretion of the polypeptide; a promoter or enhancer is operably linked to a coding sequence if it affects the transcription of the sequence; or a ribosome binding site is operably linked to a coding sequence if it is positioned so as to facilitate translation. Generally, "operably linked" means that the DNA sequences being linked are contiguous, and, in the case of a secretory leader, contiguous and in reading frame. However, enhancers do not have to be contiguous. Linking is accomplished by ligation at convenient restriction sites. If such sites do not exist, the synthetic oligonucleotide adaptors or linkers are used in accordance with conventional practice.
[0181] Purification of antibodies is performed in order to eliminate cellular components or other contaminants, e.g. other cellular nucleic acids or proteins, by standard techniques, including alkaline/SDS treatment, CsCl banding, column chromatography, agarose gel electrophoresis, and others well known in the art. See Ausubel, F., et al., ed. Current Protocols in Molecular Biology, Greene Publishing and Wiley Interscience, New York (1987). Different methods are well established and widespread used for protein purification, such as affinity chromatography with microbial proteins (e.g. protein A or protein G affinity chromatography), ion exchange chromatography (e.g. cation exchange (carboxymethyl resins), anion exchange (amino ethyl resins) and mixed-mode exchange), thiophilic adsorption (e.g. with beta-mercaptoethanol and other SH ligands), hydrophobic interaction or aromatic adsorption chromatography (e.g. with phenyl-sepharose, aza-arenophilic resins, or m-aminophenylboronic acid), metal chelate affinity chromatography (e.g. with Ni(II)- and Cu(II)-affinity material), size exclusion chromatography, and electrophoretical methods (such as gel electrophoresis, capillary electrophoresis) (Vijayalakshmi, M. A., Appl. Biochem. Biotech. 75 (1998) 93-102).
[0182] The bispecific, bivalent antibodies according to the invention show benefits for human patients in need of a VEGF and ANG-2 targeting therapy.
[0183] The bivalent bispecific against human VEGF and human ANG-2 according to the current invention may have a valuable efficacy/safety profile and may provide benefits for a patient in the need of an anti-VEGF and anti-ANG-2 therapy.
[0184] One aspect of the invention is a pharmaceutical composition comprising an antibody according to the invention. Another aspect of the invention is the use of an antibody according to the invention for the manufacture of a pharmaceutical composition. A further aspect of the invention is a method for the manufacture of a pharmaceutical composition comprising an antibody according to the invention. In another aspect, the present invention provides a composition, e.g. a pharmaceutical composition, containing an antibody according to the present invention, formulated together with a pharmaceutical carrier.
[0185] As used herein, "pharmaceutical carrier" includes any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like that are physiologically compatible. Preferably, the carrier is suitable for administration administered to the subject via a local route. For example, the antibody or its composition can be administered to the subject by intraocular application e.g. by intraocular injection such as intravitreal injection. This can be performed in accordance with standard procedures known in the art. See, e.g., Ritter et al., J. Clin. Invest. 116 (2006) 3266-76; Russelakis-Carneiro et al., Neuropathol. Appl. Neurobiol. 25 (1999) 196-206; and Wray et al., Arch. Neurol. 33 (1976) 183-5.
[0186] A composition of the present invention can be administered by a variety of methods known in the art. As will be appreciated by the skilled artisan, the route and/or mode of administration will vary depending upon the desired results. To administer a compound of the invention by certain routes of administration, it may be necessary to coat the compound with, or co-administer the compound with, a material to prevent its inactivation. For example, the compound may be administered to a subject in an appropriate carrier, for example, liposomes, or a diluent. Pharmaceutically acceptable diluents include saline and aqueous buffer solutions. Pharmaceutical carriers include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion. The use of such media and agents for pharmaceutically active substances is known in the art.
[0187] Many possible modes of delivery can be used, including, but not limited to intraocular application or topical application. In one embodiment the application is intraocular and includes, but is not limited to, subconjunctival injection, intracanieral injection, injection into the anterior chamber via the termporal limbus, intrastromal injection, intracorneal injection, subretinal injection, aqueous humor injection, subtenon injection or sustained delivery device, intravitreal injection (e.g., front, mid or back vitreal injection). In one embodiment the application is topical and includes, but is not limited to eye drops to the cornea.
[0188] In one embodiment the bispecific antibody or pharmaceutical composition according to the invention is administered via intravitreal application, e.g. via intravitreal injection. This can be performed in accordance with standard procedures known in the art. See, e.g., Ritter et al., J. Clin. Invest. 116 (2006) 3266-76; Russelakis-Carneiro et al., Neuropathol. Appl. Neurobiol. 25 (1999) 196-206; and Wray et al., Arch. Neurol. 33 (1976) 183-5.
[0189] In some embodiments, therapeutic kits of the invention can contain one or more doses of a bispecific antibody present in a pharmaceutical composition described herein, a suitable device for intravitreal injection of the pharmaceutical composition, and an instruction detailing suitable subjects and protocols for carrying out the injection. In these embodiments, the compositions are typically administered to the subject in need of treatment via intravitreal injection. This can be performed in accordance with standard procedures known in the art. See, e.g., Ritter et al., J. Clin. Invest. 116 (2006) 3266-76; Russelakis-Carneiro et al., Neuropathol. Appl. Neurobiol. 25 (1999) 196-206; and Wray et al., Arch. Neurol. 33 (1976) 183-5.
[0190] The compositions may also contain adjuvants such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of presence of microorganisms may be ensured both by sterilization procedures, supra, and by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol, sorbic acid, and the like. It may also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into the compositions. In addition, prolonged absorption of the injectable pharmaceutical form may be brought about by the inclusion of agents which delay absorption such as aluminum monostearate and gelatin.
[0191] Regardless of the route of administration selected, the compounds of the present invention, which may be used in a suitable hydrated form, and/or the pharmaceutical compositions of the present invention, are formulated into pharmaceutically acceptable dosage forms by conventional methods known to those of skill in the art.
[0192] Actual dosage levels of the active ingredients in the pharmaceutical compositions of the present invention may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration, without being toxic to the patient. The selected dosage level will depend upon a variety of pharmacokinetic factors including the activity of the particular compositions of the present invention employed, the route of administration, the time of administration, the rate of excretion of the particular compound being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compositions employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
[0193] The composition must be sterile and fluid to the extent that the composition is deliverable by syringe. In addition to water, the carrier preferably is an isotonic buffered saline solution.
[0194] Proper fluidity can be maintained, for example, by use of coating such as lecithin, by maintenance of required particle size in the case of dispersion and by use of surfactants. In many cases, it is preferable to include isotonic agents, for example, sugars, polyalcohols such as mannitol or sorbitol, and sodium chloride in the composition.
[0195] The composition can comprise an ophthalmic depot formulation comprising an active agent for subconjunctival administration. The ophthalmic depot formulation comprises microparticles of essentially pure active agent, e.g., the bispecific antibody according to the invention. The microparticles comprising the bispecific antibody according to the invention can be embedded in a biocompatible pharmaceutically acceptable polymer or a lipid encapsulating agent. The depot formulations may be adapted to release all of substantially all the active material over an extended period of time. The polymer or lipid matrix, if present, may be adapted to degrade sufficiently to be transported from the site of administration after release of all or substantially all the active agent. The depot formulation can be liquid formulation, comprising a pharmaceutical acceptable polymer and a dissolved or dispersed active agent. Upon injection, the polymer forms a depot at the injections site, e.g. by gelifying or precipitating.
[0196] Another aspect of the invention is the bispecific antibody according to the invention for use in the treatment of ocular vascular diseases.
[0197] One embodiment of the invention is the bispecific antibody according to the invention for use in the treatment of ocular vascular diseases.
[0198] Another aspect of the invention is said pharmaceutical composition for use in the treatment of ocular vascular diseases.
[0199] Another aspect of the invention is the use of an antibody according to the invention for the manufacture of a medicament for the treatment of ocular vascular disease.
[0200] Another aspect of the invention is method of treatment of patient suffering from ocular vascular diseases by administering an antibody according to the invention to a patient in the need of such treatment.
[0201] The terms "ocular vascular disease" and "vascular eye disease" are use inter changeable herein and include, but are not limited to intraocular neovascular syndromes such as diabetic retinopathy, diabetic macular edema--retinopathy of prematurity, neovascular glaucoma, retinal vein occlusions, central retinal vein occlusions, macular degeneration, age-related macular degeneration, retinitis pigmentosa, retinal angiomatous proliferation, macular telangectasia, ischemic retinopathy, iris neovascularization, intraocular neovascularization, corneal neovascularization, retinal neovascularization, choroidal neovascularization, and retinal degeneration. (Garner, A., Vascular diseases, In: Pathobiology of ocular disease, A dynamic approach, Garner, A., and Klintworth, G. K., (eds.), 2nd edition, Marcel Dekker, New York (1994), pp. 1625-1710). As used herein, ocular vascular disorder refers to any pathological conditions characterized by altered or unregulated proliferation and invasion of new blood vessels into the structures of ocular tissues such as the retina or cornea. In one embodiment the ocular vascular disease is selected from the group consisting of: wet age-related macular degeneration (wet AMD), dry age-related macular degeneration (dry AMD), diabetic macular edema (DME), cystoid macular edema (CME), non-proliferative diabetic retinopathy (NPDR), proliferative diabetic retinopathy (PDR), cystoid macular edema, vasculitis (e.g. central retinal vein occlusion), papilloedema, retinitis, conjunctivitis, uveitis, choroiditis, multifocal choroiditis, ocular histoplasmosis, blepharitis, dry eye (Sjogren's disease) and other ophthalmic diseases wherein the eye disease or disorder is associated with ocular neovascularization, vascular leakage, and/or retinal edema. So the bispecific antibodies according to the invention are useful in the prevention and treatment of wet AMD, dry AMD, CME, DME, NPDR, PDR, blepharitis, dry eye and uveitis, also preferably wet AMD, dry AMD, blepharitis, and dry eye, also preferably CME, DME, NPDR and PDR, also preferably blepharitis, and dry eye, in particular wet AMD and dry AMD, and also particularly wet AMD. In some embodiments, the ocular disease is selected from the group consisting of wet age-related macular degeneration (wet AMD), macular edema, retinal vein occlusions, retinopathy of prematurity, and diabetic retinopathy.
[0202] Other diseases associated with corneal neovascularization include, but are not limited to, epidemic keratoconjunctivitis, Vitamin A deficiency, contact lens overwear, atopic keratitis, superior limbic keratitis, pterygium keratitis sicca, sjogrens, acne rosacea, phylectenulosis, syphilis, Mycobacteria infections, lipid degeneration, chemical burns, bacterial ulcers, fungal ulcers, Herpes simplex infections, Herpes zoster infections, protozoan infections, Kaposi sarcoma, Mooren ulcer, Terrien's marginal degeneration, mariginal keratolysis, rheumatoid arthritis, systemic lupus, polyarteritis, trauma, Wegeners sarcoidosis, Scleritis, Steven's Johnson disease, periphigoid radial keratotomy, and corneal graph rejection.
[0203] Diseases associated with retinal/choroidal neovascularization include, but are not limited to, diabetic retinopathy, macular degeneration, sickle cell anemia, sarcoid, syphilis, pseudoxanthoma elasticum, Pagets disease, vein occlusion, artery occlusion, carotid obstructive disease, chronic uveitis/vitritis, mycobacterial infections, Lyme's disease, systemic lupus erythematosis, retinopathy of prematurity, retinitis pigmentosa, retina edema (including macular edema), Eales disease, Bechets disease, infections causing a retinitis or choroiditis, presumed ocular histoplasmosis, Bests disease, myopia, optic pits, Stargarts disease, pars planitis, chronic retinal detachment, hyperviscosity syndromes, toxoplasmosis, trauma and post-laser complications. Other diseases include, but are not limited to, diseases associated with rubeosis (neovascularization of the angle) and diseases caused by the abnormal proliferation of fibrovascular or fibrous tissue including all forms of proliferative vitreoretinopathy.
[0204] Retinopathy of prematurity (ROP) is a disease of the eye that affects prematurely born babies. It is thought to be caused by disorganized growth of retinal blood vessels which may result in scarring and retinal detachment. ROP can be mild and may resolve spontaneously, but may lead to blindness in serious cases. As such, all preterm babies are at risk for ROP, and very low birth weight is an additional risk factor. Both oxygen toxicity and relative hypoxia can contribute to the development of ROP.
[0205] Macular degeneration is a medical condition predominantly found in elderly adults in which the center of the inner lining of the eye, known as the macula area of the retina, suffers thinning, atrophy, and in some cases, bleeding. This can result in loss of central vision, which entails inability to see fine details, to read, or to recognize faces. According to the American Academy of Ophthalmology, it is the leading cause of central vision loss (blindness) in the United States today for those over the age of fifty years. Although some macular dystrophies that affect younger individuals are sometimes referred to as macular degeneration, the term generally refers to age-related macular degeneration (AMD or ARMD).
[0206] Age-related macular degeneration begins with characteristic yellow deposits in the macula (central area of the retina which provides detailed central vision, called fovea) called drusen between the retinal pigment epithelium and the underlying choroid. Most people with these early changes (referred to as age-related maculopathy) have good vision. People with drusen can go on to develop advanced AMD. The risk is considerably higher when the drusen are large and numerous and associated with disturbance in the pigmented cell layer under the macula. Large and soft drusen are related to elevated cholesterol deposits and may respond to cholesterol lowering agents or the Rheo Procedure.
[0207] Advanced AMD, which is responsible for profound vision loss, has two forms: dry and wet. Central geographic atrophy, the dry form of advanced AMD, results from atrophy to the retinal pigment epithelial layer below the retina, which causes vision loss through loss of photoreceptors (rods and cones) in the central part of the eye. While no treatment is available for this condition, vitamin supplements with high doses of antioxidants, lutein and zeaxanthin, have been demonstrated by the National Eye Institute and others to slow the progression of dry macular degeneration and in some patients, improve visual acuity.
[0208] Retinitis pigmentosa (RP) is a group of genetic eye conditions. In the progression of symptoms for RP, night blindness generally precedes tunnel vision by years or even decades. Many people with RP do not become legally blind until their 40s or 50s and retain some sight all their life. Others go completely blind from RP, in some cases as early as childhood. Progression of RP is different in each case. RP is a type of hereditary retinal dystrophy, a group of inherited disorders in which abnormalities of the photoreceptors (rods and cones) or the retinal pigment epithelium (RPE) of the retina lead to progressive visual loss. Affected individuals first experience defective dark adaptation or nyctalopia (night blindness), followed by reduction of the peripheral visual field (known as tunnel vision) and, sometimes, loss of central vision late in the course of the disease.
[0209] Macular edema occurs when fluid and protein deposits collect on or under the macula of the eye, a yellow central area of the retina, causing it to thicken and swell. The swelling may distort a person's central vision, as the macula is near the center of the retina at the back of the eyeball. This area holds tightly packed cones that provide sharp, clear central vision to enable a person to see form, color, and detail that is directly in the line of sight. Cystoid macular edema is a type of macular edema that includes cyst formation.
[0210] Combination Therapies: In certain embodiments the bispecific antibody or pharmaceutical composition according to the invention is administered alone (without an additional therapeutic agent) for the treatment of one or more ocular vascular diseases described herein.
[0211] In other embodiments the bispecific antibody or pharmaceutical composition according to the invention is administered in combination with one or more additional therapeutic agents or methods for the treatment of one or more ocular vascular diseases described herein.
[0212] In other embodiments, the bispecific antibody or pharmaceutical composition according to the invention is formulated in combination with one or more additional therapeutic agents and administered for the treatment of one or more ocular vascular diseases described herein.
[0213] In certain embodiments, the combination treatments provided herein include administration the bispecific antibody or pharmaceutical composition according to the invention is administered sequentially with one or more additional therapeutic agents for the treatment of one or more ocular vascular diseases described herein.
[0214] The additional therapeutic agents include, but are not limited to, Tryptophanyl-tRNA synthetase (TrpRS), EyeOOl (Anti-VEGF Pegylated Aptamer), squalamine, RETAANE.RTM. (anecortave acetate for depot suspension; Alcon, Inc.), Combretastatin A4 Prodrug (CA4P), MACUGEN.RTM., MIFEPREX.RTM. (mifepristone-ru486), subtenon triamcinolone acetonide, intravitreal crystalline triamcinolone acetonide, Prinomastat (AG3340--synthetic matrix metalloproteinase inhibitor, Pfizer), fluocinolone acetonide (including fluocinolone intraocular implant, Bausch & Lomb/Control Delivery Systems), VEGFR inhibitors (Sugen), VEGF-Trap (Regeneron/Aventis), VEGF receptor tyrosine kinase inhibitors such as 4-(4-bromo-2-fluoroanilino)-6-methoxy-7-(l-methylpiperidin-4-ylme- thoxy)quinazoline (ZD6474), 4-(4-fluoro-2-methylindol-5-yloxy)-6-methoxy-7-(3-pyrrolidin-1-ylpropoxy)- quinazoline (AZD2171), vatalanib (PTK787) and SU1 1248 (sunitinib), linomide, and inhibitors of integrin v.beta.3 function and angiostatin.
[0215] Other pharmaceutical therapies that can be sued used in combination the bispecific antibody or pharmaceutical composition according to the invention is administered, include, but are not limited to, VISUDYNE.RTM. with use of a non-thermal laser, PKC 412, Endovion (NeuroSearch A/S), neurotrophic factors, including by way of example Glial Derived Neurotrophic Factor and Ciliary Neurotrophic Factor, diatazem, dorzolamide, Phototrop, 9-cis-retinal, eye medication (including Echo Therapy) including phospholine iodide or echothiophate or carbonic anhydrase inhibitors, AE-941 (AEterna Laboratories, Inc.), Sirna-027 (Sima Therapeutics, Inc.), pegaptanib (NeXstar Pharmaceuticals/Gilead Sciences), neurotrophins (including, by way of example only, NT-4/5, Genentech), Candy (Acuity Pharmaceuticals), INS-37217 (Inspire Pharmaceuticals), integrin antagonists (including those from Jerini AG and Abbott Laboratories), EG-3306 (Ark Therapeutics Ltd.), BDM-E (BioDiem Ltd.), thalidomide (as used, for example, by EntreMed, Inc.), cardiotrophin-1 (Genentech), 2-methoxyestradiol (Allergan/Oculex), DL-8234 (Toray Industries), NTC-200 (Neurotech), tetrathiomolybdate (University of Michigan), LYN-002 (Lynkeus Biotech), microalgal compound (Aquasearch/Albany, Mera Pharmaceuticals), D-9120 (Celltech Group pic), ATX-S10 (Hamamatsu Photonics), TGF-beta 2 (Genzyme/Celtrix), tyrosine kinase inhibitors (Allergan, SUGEN, Pfizer), NX-278-L (NeXstar Pharmaceuticals/Gilead Sciences), Opt-24 (OPTIS France SA), retinal cell ganglion neuroprotectants (Cogent Neurosciences), N-nitropyrazole derivatives (Texas A&M University System), KP-102 (Krenitsky Pharmaceuticals), cyclosporin A, Timited retinal translocation", photodynamic therapy, (including, by way of example only, receptor-targeted PDT, Bristol-Myers Squibb, Co.; porfimer sodium for injection with PDT; verteporfin, QLT Inc.; rostaporfin with PDT, Miravent Medical Technologies; talaporfin sodium with PDT, Nippon Petroleum; motexafin lutetium, Pharmacyclics, Inc.), antisense oligonucleotides (including, by way of example, products tested by Novagali Pharma SA and ISIS-13650, Isis Pharmaceuticals), laser photocoagulation, drusen lasering, macular hole surgery, macular translocation surgery, implantable miniature telescopes, Phi-Motion Angiography (also known as Micro-Laser Therapy and Feeder Vessel Treatment), Proton Beam Therapy, microstimulation therapy, Retinal Detachment and Vitreous Surgery, Scleral Buckle, Submacular Surgery, Transpupillary Thermotherapy, Photosystem I therapy, use of RNA interference (RNAi), extracorporeal rheopheresis (also known as membrane differential filtration and Rheotherapy), microchip implantation, stem cell therapy, gene replacement therapy, ribozyme gene therapy (including gene therapy for hypoxia response element, Oxford Biomedica; Lentipak, Genetix; PDEF gene therapy, GenVec), photoreceptor/retinal cells transplantation (including transplantable retinal epithelial cells, Diacrin, Inc.; retinal cell transplant, Cell Genesys, Inc.), and acupuncture.
[0216] Any anti-angiogenic agent can be used in combination with the bispecific antibody or pharmaceutical composition according to the invention, including, bu not limited to, those listed by Carmeliet and Jain, 2000, Nature 407:249-257. In certain embodiments, the anti-angiogenic agent is another VEGF antagonist or a VEGF receptor antagonist such as VEGF variants, soluble VEGF receptor fragments, aptamers capable of blocking VEGF or VEGFR, neutralizing anti-VEGFR antibodies, low molecule weight inhibitors of VEGFR tyrosine kinases and any combinations thereof and these include anti-VEGF aptamers (e.g. Pegaptanib), soluble recombinant decoy receptors (e.g. VEGF Trap). In certain embodiments, the anti-angiogenic agent is include corticosteroids, angiostatic steroids, anecortave acetate, angiostatin, endostatin, small interfering RNA's decreasing expression of VEGFR or VEGF ligand, post-VEGFR blockade with tyrosine kinase inhibitors, MMP inhibitors, IGFBP3, SDF-1 blockers, PEDF, gamma-secretase, Delta-like ligand 4, integrin antagonists, HIF-1 alpha blockade, protein kinase CK2 blockade, and inhibition of stem cell (i.e. endothelial progenitor cell) homing to the site of neovascularization using vascular endothelial cadherin (CD-144) and stromal derived factor (SDF)-I antibodies. Small molecule RTK inhibitors targeting VEGF receptors including PTK787 can also be used. Agents that have activity against neovascularization that are not necessarily anti-VEGF compounds can also be used and include anti-inflammatory drugs, m-Tor inhibitors, rapamycin, everolismus, temsirolismus, cyclospohne, anti-TNF agents, anti-complement agents, and nonsteroidal antiinflammatory agents. Agents that are neuroprotective and can potentially reduce the progression of dry macular degeneration can also be used, such as the class of drugs called the `neurosteroids.` These include drugs such as dehydroepiandrosterone(DHEA)(Brand names: Prastera.TM. and Fidelin.RTM.), dehydroepiandrosterone sulfate, and pregnenolone sulfate. Any AMD (age-related macular degeneration) therapeutic agent can be used in combination with the bispecific antibody or pharmaceutical composition according to the invention, including but not limited to verteporfin in combination with PDT, pegaptanib sodium, zinc, or an antioxidant(s), alone or in any combination.
[0217] The terms "subject" and "patient" are used interchangeably and refer to mammals such as human patients and non-human primates, as well as experimental animals such as rabbits, rats, and mice, and other animals. Animals include all vertebrates, e.g., mammals and non-mammals, such as dogs, cats, sheeps, cows, pigs, rabbits, chickens, and etc. Preferred subjects for practicing the therapeutic methods of the present invention are human. Subjects in need of treatment include patients already suffering from an ocular vascular disease or disorder as well as those prone to developing the disorder.
[0218] As used herein, the expressions "cell," "cell line," and "cell culture" are used interchangeably and all such designations include progeny. Thus, the words "transformants" and "transformed cells" include the primary subject cell and cultures derived therefrom without regard for the number of transfers. It is also understood that all progeny may not be precisely identical in DNA content, due to deliberate or inadvertent mutations. Variant progeny that have the same function or biological activity as screened for in the originally transformed cell are included. Where distinct designations are intended, it will be clear from the context.
[0219] The term "transformation" as used herein refers to process of transfer of a vectors/nucleic acid into a host cell. If cells without formidable cell wall barriers are used as host cells, transfection is carried out e.g. by the calcium phosphate precipitation method as described by Graham, F. L., van der Eb, A. J., Virology 52 (1973) 546-467. However, other methods for introducing DNA into cells such as by nuclear injection or by protoplast fusion may also be used. If prokaryotic cells or cells which contain substantial cell wall constructions are used, e.g. one method of transfection is calcium treatment using calcium chloride as described by Cohen, S. N., et al., PNAS. 69 (1972) 2110-2114.
[0220] As used herein, "expression" refers to the process by which a nucleic acid is transcribed into mRNA and/or to the process by which the transcribed mRNA (also referred to as transcript) is subsequently being translated into peptides, polypeptides, or proteins. The transcripts and the encoded polypeptides are collectively referred to as gene product. If the polynucleotide is derived from genomic DNA, expression in a eukaryotic cell may include splicing of the mRNA.
[0221] A "vector" is a nucleic acid molecule, in particular self-replicating, which transfers an inserted nucleic acid molecule into and/or between host cells. The term includes vectors that function primarily for insertion of DNA or RNA into a cell (e.g., chromosomal integration), replication of vectors that function primarily for the replication of DNA or RNA, and expression vectors that function for transcription and/or translation of the DNA or RNA. Also included are vectors that provide more than one of the functions as described.
[0222] An "expression vector" is a polynucleotide which, when introduced into an appropriate host cell, can be transcribed and translated into a polypeptide. An "expression system" usually refers to a suitable host cell comprised of an expression vector that can function to yield a desired expression product.
[0223] The following examples, sequence listing and figures are provided to aid the understanding of the present invention, the true scope of which is set forth in the appended claims. It is understood that modifications can be made in the procedures set forth without departing from the spirit of the invention.
TABLE-US-00001 Description of the Sequence Listing (Amino acid sequences) SEQ ID NO: 1 heavy chain CDR3H, <VEGF> ranibizumab SEQ ID NO: 2 heavy chain CDR2H, <VEGF> ranibizumab SEQ ID NO: 3 heavy chain CDR1H, <VEGF> ranibizumab SEQ ID NO: 4 light chain CDR3L, <VEGF> ranibizumab SEQ ID NO: 5 light chain CDR2L, <VEGF> ranibizumab SEQ ID NO: 6 light chain CDR1L, <VEGF> ranibizumab SEQ ID NO: 7 heavy chain variable domain VH, <VEGF> ranibizumab SEQ ID NO: 8 light chain variable domain VL, <VEGF> ranibizumab SEQ ID NO: 9 heavy chain CDR3H, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 10 heavy chain CDR2H, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 11 heavy chain CDR1H, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 12 light chain CDR3L, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 13 light chain CDR2L, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 14 light chain CDR1L, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 15 heavy chain variable domain VH, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 16 light chain variable domain VL, <ANG-2> Ang2i_LC10 variant SEQ ID NO: 17 Human vascular endothelial growth factor (VEGF); precursor sequence of human VEGF165 SEQ ID NO: 18 Human angiopoietin-2 (ANG-2) SEQ ID NO: 19 Human angiopoietin-1 (ANG-1) SEQ ID NO: 20 Human Tie-2 receptor SEQ ID NO 21 Heavy chain 1 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations (VEGFang2-0012) SEQ ID NO 22 Heavy chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations (VEGFang2-0012) SEQ ID NO 23 Light chain 1 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations (VEGFang2-0012) SEQ ID NO 24 Light chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations (VEGF-Ang2-0012) SEQ ID NO: 25 Heavy chain 1 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations and P329G LALA mutations (VEGFang2-0016) SEQ ID NO: 26 Heavy chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations and P329G LALA mutations (VEGFang2-0016) SEQ ID NO: 27 Light chain 1 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations and P329G LALA mutations (VEGFang2-0016) SEQ ID NO: 28 Light chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations and P329G LALA mutations (VEGFang2-0016) SEQ ID NO: 29 Heavy chain 1 of <VEGF-ANG-2> CrossMAb IgG4 with AAA mutations and with SPLE mutations SEQ ID NO: 30 Heavy chain 2 of <VEGF-ANG-2> CrossMAb IgG4 with AAA mutations and with SPLE mutations SEQ ID NO: 31 Light chain 1 of <VEGF-ANG-2> CrossMAb IgG4 with AAA mutations and with SPLE mutations SEQ ID NO: 32 Light chain 2 of <VEGF-ANG-2> CrossMAb IgG4 with AAA mutations and with SPLE mutations SEQ ID NO: 33 Heavy chain 1 of <VEGF-ANG-2> OAscFab IgG1 with AAA mutations SEQ ID NO: 34 Heavy chain 2 of <VEGF-ANG-2> OAscFab IgG1 with AAA mutations SEQ ID NO: 35 Light chain 1 of <VEGF-ANG-2> OAscFab IgG1 with AAA mutations SEQ ID NO: 36 Heavy chain 1 of <VEGF-ANG-2> OAscFab IgG4 with AAA mutations and with SPLE mutations SEQ ID NO: 37 Heavy chain 2 of <VEGF-ANG-2> OAscFab IgG4 with AAA mutations and with SPLE mutations SEQ ID NO: 38 Light chain 1 of <VEGF-ANG-2> OAscFab IgG4 with AAA mutations and with SPLE mutations SEQ ID NO: 39 Heavy chain 1 of <VEGF-ANG-2> CrossMAb IgG1 wild type (without AAA mutations) (VEGFang2-0201) SEQ ID NO: 40 Heavy chain 2 of <VEGF-ANG-2> CrossMAb IgG1 wild type (without AAA mutations) (VEGFang2-0201) SEQ ID NO: 41 Light chain 1 of <VEGF-ANG-2> CrossMAb IgG1 wild type (without AAA mutations) (VEGFang2-0201) SEQ ID NO: 42 Light chain 2 of <VEGF-ANG-2> CrossMAb IgG1 wild type (without AAA mutations) (VEGFang2-0201) SEQ ID NO: 43 Heavy chain 1 of <VEGF-ANG-2> CrossMAb IgG1 with P329G LALA mutations only (without AAA mutations) (VEGFang2-0015) SEQ ID NO: 44 Heavy chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with P329G LALA mutations only (without AAA mutations) (VEGFang2-0015) SEQ ID NO: 45 Light chain 1 of <VEGF-ANG-2> CrossMAb IgG1 with P329G LALA mutations only (without AAA mutations) (VEGFang2-0015) SEQ ID NO: 46 Light chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with P329G LALA mutations only (without AAA mutations) (VEGFang2-0015) SEQ ID NO: 47 kappa light chain constant region SEQ ID NO: 48 lambda light chain constant region SEQ ID NO: 49 heavy chain constant region derived from human IgG1 SEQ ID NO: 50 heavy chain constant region derived from human IgG4
[0224] In the following, embodiments of the invention are listed: [0225] 1. A bispecific antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, wherein [0226] i) said first antigen-binding site specifically binding to VEGF comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 1, a CDR2H region of SEQ ID NO: 2, and a CDR1H region of SEQ ID NO:3, and in the light chain variable domain a CDR3L region of SEQ ID NO: 4, a CDR2L region of SEQ ID NO:5, and a CDR1L region of SEQ ID NO:6; and [0227] ii) said second antigen-binding site specifically binding to ANG-2 comprises in the heavy chain variable domain a CDR3H region of SEQ ID NO: 9, a CDR2H region of, SEQ ID NO: 10, and a CDR1H region of SEQ ID NO: 11, and in the light chain variable domain a CDR3L region of SEQ ID NO: 12, a CDR2L region of SEQ ID NO: 13, and a CDR1L region of SEQ ID NO: 14, and wherein [0228] iii) the bispecific antibody comprises a constant heavy chain region of human IgG1 or human IgG4 subclass (derived from human origin and) comprising the mutations I253A, H310A, and H435A (numbering according to EU Index of Kabat). [0229] 2. The bispecific antibody according to embodiment 1, wherein [0230] i) said first antigen-binding site specifically binding to VEGF comprises as heavy chain variable domain VH an amino acid sequence of SEQ ID NO: 7, and as light chain variable domain VL an amino acid sequence of SEQ ID NO: 8, and [0231] ii) said second antigen-binding site specifically binding to ANG-2 comprises as heavy chain variable domain VH an amino acid sequence of SEQ ID NO: 15, and as light chain variable domain VL an amino acid sequence of SEQ ID NO: 16. [0232] 3. The bispecific antibody according to any one of embodiments 1 to 2, wherein the constant heavy chain region under iii) is of IgG1 subclass. [0233] 4. The bispecific antibody according to embodiment 3, wherein the constant heavy chain region of IgG1 subclass further comprises the mutations L234A, L235A and P329G (numbering according to EU Index of Kabat). [0234] 5. The bispecific antibody according to any one of embodiments 1 to 2, wherein the constant heavy chain region under iii) is of IgG4 subclass. [0235] 6. The bispecific antibody according to embodiment 5, wherein the constant heavy chain region of IgG4 subclass further comprises the mutations S228P and L235E (numbering according to EU Index of Kabat). [0236] 7. The bispecific antibody according to embodiment 5, wherein the constant heavy chain region of IgG4 subclass further comprises the mutations S228P, L235E and P329G (numbering according to EU Index of Kabat). [0237] 8. A pharmaceutical composition comprising an antibody according to any one of embodiments 1 to 7. [0238] 9. The bispecific antibody according to any one of embodiments 1 to 7 for use in the treatment of ocular vascular diseases. [0239] 10. Use of the bispecific antibody according to any one of embodiments 1 to 7 for the manufacture of a medicament for the treatment of ocular vascular diseases. [0240] 11. The bispecific antibody according to any one of embodiments 9 or 10, wherein the antibody is administered via intravitreal application. [0241] 12. A method of treatment of patient suffering from ocular vascular diseases by administering an antibody according to any one of embodiments 1 to 7 to a patient in the need of such treatment. [0242] 13. A nucleic acid encoding a bispecific antibody according to any one of embodiments 1 to 7. [0243] 14. Expression vector containing said nucleic acid according embodiment 13 capable of expressing said nucleic acid in a prokaryotic or eukaryotic host cell. [0244] 15. A prokaryotic or eukaryotic host cell comprising a vector according to embodiment 14. [0245] 16. A method for the preparation of a bispecific antibody according to embodiments 1 to 7 [0246] comprising the steps of [0247] a) transforming a host cell with vectors comprising nucleic acid molecules encoding said antibody; [0248] b) culturing the host cell under conditions that allow synthesis of said antibody molecule; and [0249] c) recovering said antibody molecule from said culture. [0250] 17. A bispecific antibody obtained by the method of embodiment 16. [0251] 18. A bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 25, of SEQ ID NO: 26, of SEQ ID NO: 27, and of SEQ ID NO: 28. [0252] 19. A bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 21, of SEQ ID NO: 22., of SEQ ID NO: 23., and of SEQ ID NO: 24. [0253] 20. A bispecific, bivalent antibody comprising a first antigen-binding site that specifically binds to human VEGF and a second antigen-binding site that specifically binds to human ANG-2, characterized in comprising the amino acid sequences of SEQ ID NO: 29, of SEQ ID NO: 30, of SEQ ID NO: 31, and of SEQ ID NO: 32.
[0254] Experimental Procedures
TABLE-US-00002 TABLE 1 Bispecific antibodies and their respective sequences Description Short Name Sequences <VEGF-ANG-2> VEGFang2-0012 SEQ ID NO: 21, CrossMAb IgG1 with SEQ ID NO: 22, AAA mutations SEQ ID NO: 23, SEQ ID NO: 24 <VEGF-ANG-2> VEGFang2-0201- SEQ ID NO: 39, CrossMAb IgG1 wild type SEQ ID NO: 40, (without AAA mutations) SEQ ID NO: 41, SEQ ID NO: 42 <VEGF-ANG-2> VEGFang2-0016 SEQ ID NO: 25, CrossMAb IgG1 with SEQ ID NO: 26, AAA mutations and SEQ ID NO: 27, P329G LALA mutations SEQ ID NO: 28 <VEGF-ANG-2> VEGFang2-0015 SEQ ID NO: 43, CrossMAb IgG1 with SEQ ID NO: 44, P329G LALA mutations SEQ ID NO: 45, only (without AAA SEQ ID NO: 46 mutations) <VEGF-ANG-2> -- SEQ ID NO: 29, CrossMAb IgG4 with SEQ ID NO: 30, AAA mutations and with SEQ ID NO: 31, SPLE mutations SEQ ID NO: 32 <VEGF-ANG-2> -- SEQ ID NO: 33, OAscFab IgG1 with AAA SEQ ID NO: 34, mutations SEQ ID NO: 35 <VEGF-ANG-2> -- SEQ ID NO: 36, OAscFab IgG4 with AAA SEQ ID NO: 37, mutations and with SPLE SEQ ID NO: 38 mutations
[0255] Please note that the term "with (the) mutations AAA" as used herein refers the mutations I253A (Ile253Ala), H310A (His310Ala), and H435A (His435Ala) in the constant heavy chain region of IgG1 or IgG4 (numbering according to EU Index of Kabat), the term "with (the) mutations P329G LALA" as used herein refers to the mutations L234A (Leu235Ala) , L235A (Leu234Ala) and P329G (Pro329Gly) in the constant heavy chain region of IgG1 subclass (numbering according to EU Index of Kabat), and the term "with (the) mutations SPLE" as used herein refers to the S228P (Ser228Pro) and L235E (Leu235Glu) the constant heavy chain region of IgG4 subclass (numbering according to EU Index of Kabat).
EXAMPLES
[0256] Materials & General Methods
[0257] General information regarding the nucleotide sequences of human immunoglobulin light and heavy chains is given in: Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991). Amino acids of antibody chains are numbered and referred to according to EU numbering (Edelman, G. M., et al., Proc. Natl. Acad. Sci. USA 63 (1969) 78-85; Kabat, E. A., et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, Md. (1991)).
[0258] Recombinant DNA Techniques
[0259] Standard methods were used to manipulate DNA as described in Sambrook, J. et al., Molecular Cloning: A laboratory manual; Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (1989). The molecular biological reagents were used according to the manufacturer's instructions.
[0260] Gene Synthesis
[0261] Desired gene segments were ordered according to given specifications at Geneart (Regensburg, Germany).
[0262] DNA Sequence Determination
[0263] DNA sequences were determined by double strand sequencing performed at MediGenomix GmbH (Martinsried, Germany) or Sequiserve GmbH (Vaterstetten, Germany).
[0264] DNA and Protein Sequence Analysis and Sequence Data Management
[0265] The GCG's (Genetics Computer Group, Madison, Wisconsin) software package version 10.2 and Infomax's Vector NT1 Advance suite version 8.0 was used for sequence creation, mapping, analysis, annotation and illustration.
[0266] Expression Vectors
[0267] For the expression of the described antibodies, variants of expression plasmids for transient expression (e.g. in HEK293-F) cells based either on a cDNA organization with or without a CMV-Intron A promoter or on a genomic organization with a CMV promoter were applied.
[0268] Beside the antibody expression cassette the vectors contained: [0269] an origin of replication which allows replication of this plasmid in E. coli, [0270] a .beta.-lactamase gene which confers ampicillin resistance in E. coli., and [0271] the dihydrofolate reductase gene from Mus musculus as a selectable marker in eukaryotic cells [0272] The transcription unit of the antibody gene was composed of the following elements: [0273] unique restriction site(s) at the 5' end [0274] the immediate early enhancer and promoter from the human cytomegalovirus, [0275] followed by the Intron A sequence in the case of the cDNA organization, [0276] a 5'-untranslated region of a human antibody gene, [0277] an immunoglobulin heavy chain signal sequence, [0278] the human antibody chain (wildtype or with domain exchange) either as cDNA or as genomic organization with the immunoglobulin exon-intron organization [0279] a 3' untranslated region with a polyadenylation signal sequence, and [0280] unique restriction site(s) at the 3' end.
[0281] The fusion genes comprising the antibody chains as described below were generated by PCR and/or gene synthesis and assembled by known recombinant methods and techniques by connection of the according nucleic acid segments e.g. using unique restriction sites in the respective vectors. The subcloned nucleic acid sequences were verified by DNA sequencing. For transient transfections larger quantities of the plasmids were prepared by plasmid preparation from transformed E. coli cultures (Nucleobond AX, Macherey-Nagel).
[0282] Cell Culture Techniques
[0283] Standard cell culture techniques were used as described in Current Protocols in Cell Biology (2000), Bonifacino, J. S., Dasso, M., Harford, J. B., Lippincott-Schwartz, J. and Yamada, K. M. (eds.), John Wiley & Sons, Inc.
[0284] The bispecific antibodies were expressed by transient co-transfection of the respective expression plasmids in in HEK29-F cells growing in suspension as described below.
Example 1
[0285] Expression and Purification
[0286] Transient Transfections in HEK293-F System
[0287] The bispecific antibodies were generated by transient transfection with the respective plasmids (e.g. encoding the heavy and modified heavy chain, as well as the corresponding light and modified light chain) using the HEK293-F system (Invitrogen) according to the manufacturer's instruction. Briefly, HEK293-F cells (Invitrogen) growing in suspension either in a shake flask or in a stirred fermenter in serum-free FreeStyle.TM. 293 expression medium (Invitrogen) were transfected with a mix of the four expression plasmids and 293fectin.TM. or fectin (Invitrogen).
[0288] For 2 L shake flask (Corning) HEK293-F cells were seeded at a density of 1.0E*6 cells/mL in 600 mL and incubated at 120 rpm, 8% CO2. The day after the cells were transfected at a cell density of ca. 1.5E*6 cells/mL with ca. 42 mL mix of A) 20 mL Opti-MEM.TM. (Invitrogen) with 600 .mu.g total plasmid DNA (1 .mu.g/mL) encoding the heavy or modified heavy chain, respectively and the corresponding light chain in an equimolar ratio and B) 20 ml Opti-MEM.TM.+1.2 mL 293 fectin or fectin (2 .mu.l/mL). According to the glucose consumption glucose solution was added during the course of the fermentation. The supernatant containing the secreted antibody was harvested after 5-10 days and antibodies were either directly purified from the supernatant or the supernatant was frozen and stored.
[0289] Purification
[0290] Bispecific antibodies were purified from cell culture supernatants by affinity chromatography using MabSelectSure-Sepharose.RTM. (for non_AAA mutants) (GE Healthcare, Sweden) or kappaSelect-Agarose (for AAA mutants) (GE Healthcare, Sweden), hydrophobic interaction chromatography using butyl-Sepharose.RTM. (GE Healthcare, Sweden) and Superdex 200 size exclusion (GE Healthcare, Sweden) chromatography.
[0291] Briefly, sterile filtered cell culture supernatants were captured on a MabSelect SuRe.TM. resin equilibrated with PBS buffer (10 mM Na.sub.2HPO.sub.4, 1 mM KH.sub.2PO.sub.4, 137 mM NaCl and 2.7 mM KC1, pH 7.4), washed with equilibration buffer and eluted with 25 mM sodium citrate at pH 3.0. The AAA mutants were captured on a kappaSelect resin equilibrated with 25 mM Tris, 50 mM NaCl, pH 7.2, washed with equilibration buffer and eluted with 25 mM sodium citrate pH 2.9. The eluted protein fractions were pooled and neutralized with 2M Tris, pH 9.0. The antibody pools were prepared for hydrophobic interaction chromatography by adding 1.6 M ammonium sulfate solution to a final concentration of 0.8 M ammonium sulfate and the pH adjusted to pH 5.0 using acetic acid. After equilibration of the butyl-Sepharose.RTM. resin with 35 mM sodium acetate, 0.8 M ammonium sulfate, pH 5.0, the antibodies were applied to the resin, washed with equilibration buffer and eluted with a linear gradient to 35 mM sodium acetate pH 5.0. The bispecific antibody containing fractions were pooled and further purified by size exclusion chromatography using a Superdex 200 26/60 GL (GE Healthcare, Sweden) column equilibrated with 20 mM histidine, 140 mM NaCl, pH 6.0. The bispecific antibody containing fractions were pooled, concentrated to the required concentration using Vivaspin ultrafiltration devices (Sartorius Stedim Biotech S.A., France) and stored at -80.degree. C.
TABLE-US-00003 TABLE 2 Yields of bispecific <YEGF-ANG-2> antibodies VEGFang2-0015 VEGFang2-0016 (with (without AAA mutation) AAA mutation) Titer supernatant 64 .mu.g/ml, n.a. (2 L = 128 mg) (2 L scale) Protein A 118 mg n.a. (MabSelectSure) (~70% monomer) Kappa Select n.a. 117 mg (~83% monomer) Butyl Sepharose 60 mg 57 mg SEC 35 mg 38 mg (>95% monomer) (>95% monomer)
[0292] Purity and antibody integrity were analyzed after each purification step by CE-SDS using microfluidic LabChip.RTM. technology (Caliper Life Science, USA). 5 .mu.l of protein solution was prepared for CE-SDS analysis using the HT Protein Express
[0293] Reagent Kit according manufacturer's instructions and analysed on LabChip.RTM. GXII system using a HT Protein Express Chip. Data were analyzed using LabChip.RTM. GX Software.
TABLE-US-00004 TABLE 3 Removal of typical side products by different sequential purification steps determined by CE-SDS. Purification Step VEGFang2-0015 VEGFang2-0016 % peak area* * analysis: CE-SDS (Caliper Labchip GXII) 3/4 1/2 3/4 1/2 (LC) mab ab (HC)2 ab (LC)2 LC mab ab (HC)2 ab 2 LC Mab 55.7 19 10.6 9.8 3.5 0.9 -- Select Sure Kappa -- 63 13.4 3.5 6.1 5.8 7.4 Select Butyl- 81.4 1.9 2.3 8.2 3.6 1.8 76.2 1.3 0.7 8.3 7.7 5.8 Sepha- rose Super- 92.4 1.8 2.6 1.4 0.5 0.5 99 1.1 n.d. n.d. n.d. n.d. dex 200_ SEC
[0294] The aggregate content of antibody samples was analyzed by high-performance SEC using a Superdex 200 analytical size-exclusion column (GE Healthcare, Sweden) in 2xPBS (20 mM Na.sub.2HPO.sub.4, 2 mM KH.sub.2PO.sub.4, 274 mM NaCl and 5.4 mM KCl, pH 7.4) running buffer at 25.degree. C. 25 .mu.g protein were injected on the column at a flow rate of 0.75 ml/min and eluted isocratic over 50 minutes.
[0295] Analogously the <VEGF-ANG-2> bispecific antibodies VEGFang2-0012 and VEGFang2-0201 were prepared and purified with the following yields:
TABLE-US-00005 VEGFang2-0012 VEGFang2-0201 (with AAA mutation) (without AAA mutation) Titer//amount -- 36 .mu.g/ml// 72 mg Scale 2.1 L 2 L Protein A -- 66 mg (MabSelectSure) (~95% monomer) kappaSelect 43 mg -- (~65% monomer) Butyl Sepharose -- 45 mg SEC 14 mg 21 mg (>98% monomer) Yield 8.5 mg hydoxylapatite (>98% monomer) Totatl yield 8.5 mg 21 mg (recovery) (20%) (30%)
[0296] Also the <VEGF-ANG-2> bispecific antibodies <VEGF-ANG-2> CrossMAb IgG4 with AAA mutations and with SPLE mutations(SEQ ID NO: 29, SEQ ID NO: 30, SEQ ID NO: 31, SEQ ID NO: 32), <VEGF-ANG-2> OAscFab IgG1 with AAA mutations(SEQ ID NO: 33, SEQ ID NO: 34, SEQ ID NO: 35)and <VEGF-ANG-2> OAscFab IgG4 with AAA mutations and with SPLE mutations(SEQ ID NO: 36, SEQ ID NO: 37, SEQ ID NO: 38) can be prepared and purified analogously.
Example 2
[0297] Analytics & Developability
[0298] Small-Scale DLS-Based Viscosity Measurement.
[0299] Viscosity measurement was essentially performed as described in (He, F. et al.,
[0300] Analytical Biochemistry 399 (2009) 141-3). Briefly, samples are concentrated to various protein concentrations in 200 mM arginine succinate, pH 5.5, before polystyrene latex beads (300 nm diameter) and Polysorbate 20 (0.02% v/v) are added. Samples are transferred into an optical 384-well plate by centrifugation through a 0.4 .mu.m filter plate and covered with paraffine oil. The apparent diameter of the latex beads is determined by dynamic light scattering at 25.degree. C. The viscosity of the solution can be calculated as .eta.=.eta.0(rh/rh,0) (.eta.: viscosity; .eta.0: viscosity of water; rh: apparent hydrodynamic radius of the latex beads; rh,0: hydrodynamic radius of the latex beads in water.
[0301] To allow comparison of various samples at the same concentration, viscosity-concentration data were fitted with the Mooney equation (Equation 1) (Mooney, Colloid Sci, 1951; Monkos, Biochem. Biophys. Acta 1997) and data interpolated accordingly.
.eta. = .eta. 0 exp ( S .PHI. 1 - K .PHI. ) Equation 1 ##EQU00001##
[0302] (S: hydrodynamic interaction parameter of the protein; K: self-crowding factor; .PHI.: volume fraction of the dissolved protein)
[0303] Results are shown in FIG. 2: VEGFang2-0016 with AAA mutations in the Fc part shows a lower viscosity at all measured temperatures compared to VEGFang2-0015 without the AAA mutations in the Fc part.
[0304] DLS Aggregation Onset Temperature
[0305] Samples are prepared at a concentration of 1 mg/mL in 20 mM Histidine/Histidine chloride, 140 mM NaCl, pH 6.0, transferred into an optical 384-well plate by centrifugation through a 0.4 .mu.m filter plate and covered with paraffine oil. The hydrodynamic radius is measured repeatedly by dynamic light scattering while the samples are heated with a rate of 0.05.degree. C./min from 25.degree. C. to 80.degree. C. The aggregation onset temperature is defined as the temperature at which the hydrodynamic radius starts to increase. Results are shown in FIG. 3. In FIG. 3 the aggregation of VEGFang2-0015 without the AAA mutations versus VEGFang2-0016 with AAA mutations in the Fc part is shown. VEGFang2-0016 showed a aggregation onset temperature of 61.degree. C. whereas VEGFang2-0015 without the AAA mutations showed a onset temperature of 60.degree. C.
[0306] DLS Timecourse
[0307] Samples are prepared at a concentration of 1 mg/mL in 20 mM Histidine/Histidine chloride, 140 mM NaCl, pH 6.0, transferred into an optical 384-well plate by centrifugation through a 0.4 .mu.m filter plate and covered with paraffine oil. The hydrodynamic radius is measured repeatedly by dynamic light scattering while the samples are kept at a constant temperature of 50.degree. C. for up to 145 hours. In this experiment, aggregation tendencies of the native, unfolded protein at elevated temperature would lead to an increase of the average particle diameter over time. This DLS-based method is very sensitive for aggregates because these contribute over-proportionally to the scattered light intensity. Even after 145 hours at 50.degree. C. (a temperature close to the aggregation-onset temperature, see above), an average particle diameter increase of only less than 0.5 nm was found for both VEGFang2-0015 and VEGFang2-0016
[0308] 7 day Storage at 40.degree. C. at 100 mg/ml (HMW Increase)
[0309] Samples are concentrated to a final concentration of 100 mg/mL in 200 mM arginine succinate, pH 5.5, sterile filtered and quiescently stored at 40.degree. C. for 7 days. Before and after storage, the content of high and low molecular weight species (HMWs and LMWs, respectively) is determined by size-exclusion chromatography. The difference in HMW and LMW content between the stored sample and a sample measured immediately after preparation is reported as "HMW increase" and "LMW increase", respectively. Results are shown in Table 4 and FIG. 4, which show that VEGFang2-0015 (without AAA mutation) shows a higher reduction of the main peak and a higher HMW increase compared to VEGF Ang2-0016 (with AAA mutation). Surprisingly VEGF Ang2-0016 (with AAA mutation) showed a lower aggregation tendency compared toVEGFang2-0015 (without AAA mutation).
TABLE-US-00006 TABLE 4 Delta Main-, HMW and LMW peaks after 7 d at 40.degree. C. delta_area % (40.degree. C.-(-80.degree. C.)) Main Peak HMW LMW VEGFang2-0015 -3.56 2.89 0.67 (-AAA mutations) VEGFang2-0016 -1.74 1.49 0.25 (+AAA mutations)
[0310] The functional analysis of anti-VEGF and anti-Ang2 bispecific antibodies was assessed by Surface Plasmon Resonance (SPR) using a BlAcore.RTM. T100 or T200 instrument (GE Healthcare) at 25.degree. C. The BIAcore.RTM. system is well established for the study of molecule interactions. SPR-technology is based on the measurement of the refractive index close to the surface of a gold coated biosensor chip. Changes in the refractive index indicate mass changes on the surface caused by the interaction of immobilized ligand with analyte injected in solution. The mass increases if molecules bind immobilized ligands on the surface, and vice versa, the mass decreases in case of dissociation of the analyte from the immobilized ligand (reflecting complex dissociation). SPR allows a continuous real-time monitoring of ligand/analyte binding and thus the determination of the association rate constant (ka), the dissociation rate constant (kd), and of the equilibrium constant (KD).
Example 3
[0311] Binding to VEGF, Ang2, FcgammaR and FcRn
[0312] VEGF Isoforms Kinetic Affinity Including Assessment of Species-Crossreactivity
[0313] Around 12000 resonance units (RU) of the capturing system (10 .mu.g/ml goat anti human F(ab)'.sub.2; Order Code: 28958325; GE Healthcare Bio-Sciences AB, Sweden) were coupled on a CMS chip (GE Healthcare BR-1005-30) at pH 5.0 by using an amine coupling kit supplied by the GE Healthcare. The sample and system buffer was PBS-T (10 mM phosphate buffered saline including 0.05% Tween20) pH 7.4. The flow cell was set to 25.degree. C.--and the sample block set to 12.degree. C.--and primed with running buffer twice. The bispecific antibody was captured by injecting a 50 nM solution for 30 sec at a flow of 5 .mu.l/min. Association was measured by injection of human hVEGF121, mouse mVEGF120 or rat rVEGF164 in various concentrations in solution for 300 sec at a flow of 30 .mu.l/min starting with 300 nM in 1:3 dilutions. The dissociation phase was monitored for up to 1200 sec and triggered by switching from the sample solution to running buffer. The surface was regenerated by 60 sec washing with a Glycine pH 2.1 solution at a flow rate of 30 .mu.l/min. Bulk refractive index differences were corrected by subtracting the response obtained from a goat anti human F(ab').sub.2 surface. Blank injections are also subtracted (=double referencing). For calculation of apparent K.sub.D and other kinetic parameters the Langmuir 1:1 model was used. Results are shown in Table 5.
[0314] Ang2 Solution Affinity Including Assessment of Species-Crossreactivity
[0315] Solution affinity measures the affinity of an interaction by determining the concentration of free interaction partners in an equilibrium mixture. The solution affinity assay involves the mixing of an <VEGF-ANG-2> bispecific antibody, kept at a constant concentration, with a ligand (=Ang2) at varying concentrations. Maximum possible resonance units (e.g. 17000 resonance units (RU)) of an antibody was immobilized on the CMS chip (GE Healthcare BR-1005-30) surface at pH 5.0 using an amine coupling kit supplied by the GE Healthcare. The sample and system buffer was HBS-P pH 7.4. Flow cell was set to 25.degree. C. and sample block to 12.degree. C. and primed with running buffer twice. To generate a calibration curve increasing concentrations of Ang2 were injected into a BlAcore flowcell containing the immobilized VEGF-ANG-2> bispecific antibody. The amount of bound Ang2 was determined as resonance units (RU) and plotted against the concentration. Solutions of each ligand (11 concentrations from 0 to 200 nM for the VEGF-ANG-2> bispecific antibody) were incubated with 10 nM Ang2 and allowed to reach equilibrium at room temperature. Free Ang2 concentrations were determined from calibration curve generated before and after measuring the response of solutions with known amounts of Ang2. A 4-parameter fit was set with XLfit4 (IDBS Software) using Model 201 using free Ang2 concentration as y-axis and used concentration of antibody for inhibition as x-axis. The affinity was calculated by determining the inflection point of this curve. The surface was regenerated by one time 30 sec washing with a 0.85% H.sub.3PO.sub.4 solution at a flow rate of 30 .mu.l/min. Bulk refractive index differences were corrected by subtracting the response obtained from a blank-coupled surface. Results are shown in Table 6.
[0316] FeRn Steady State Affinity
[0317] For FcRn measurement a steady state affinity was used to compare bispecific antibodies against each other. Human FcRn was diluted into coupling buffer (10 .mu.g/ml, Na-Acetate pH5.0) and immobilized on a C1-Chip (GE Healthcare BR-1005-35) by targeted immobilization procedure using a BlAcore wizard to a final response of 200 RU. Flow cell was set to 25.degree. C. and sample block to 12.degree. C. and primed with running buffer twice. The sample and system buffer was PBS-T (10 mM phosphate buffered saline including 0.05% Tween20) pH 6.0. To assess different IgG concentrations for each antibody, a concentration of 62.5 nM, 125 nM and 250 nM, 500 nM was prepared. Flow rate was set to 30 .mu.l/min and the different samples were injected consecutively onto the chip surface choosing 180 sec association time. The surface was regenerated by injected PBS-T pH 8 for 60 sec at a flow rate of 30 .mu.l/min. Bulk refractive index differences were corrected by subtracting the response obtained from a blank surface. Buffer injections are also subtracted (=double referencing). For calculation of steady state affinity the method from the Bia-Evaluation software was used. Briefly, the RU values (RU max) were plotted against the analysed concentrations, yielding a dose-response curve. Based on a 2-parametric fit, the upper asymptote is calculated, allowing the determination of the half-maximal RU value and hence the affinity. Results are shown in FIG. 5 and Table 7. Analogously the affinity to cyno, mouse and rabbit FcRn can be determined.
[0318] FcgammaRIIIa Measurement
[0319] For FcgammaRIIIa measurement a direct binding assay was used. Around 3000 resonance units (RU) of the capturing system (1 .mu.g/ml Penta-His; Quiagen) were coupled on a CMS chip (GE Healthcare BR-1005-30) at pH 5.0 by using an amine coupling kit supplied by the GE Healthcare. The sample and system buffer was HBS-P+pH 7.4. The flow cell was set to 25.degree. C.--and sample block to 12.degree. C.--and primed with running buffer twice. The FcgammaRIIIa -His-receptor was captured by injecting a 100 nM solution for 60 sec at a flow of 5 .mu.l/min. Binding was measured by injection of 100 nM of bispecific antibody or monospecific control antibodies (anti-Dig for IgG1 subclass and an IgG4 subclass antibody) for 180 sec at a flow of 30 .mu.l/. The surface was regenerated by 120 sec washing with Glycine pH 2.5 solution at a flow rate of 30 .mu.l/min. Because FcgammaRIIIa binding differs from the Langmuir 1:1 model, only binding/no binding was determined with this assay. In a similar manner FcgammaRIa , and FcgammaRIIa binding can be determined. Results are shown in FIG. 6, where it follows that by introduction of the mutations P329G LALA no more binding to FcgammaRIIIa could be detected.
[0320] Assessment of Independent VEGF- and Ang2-Binding to the <VEGF-ANG-2> Bispecific Antibodies
[0321] Around 3500 resonance units (RU) of the capturing system (10 .mu.g/ml goat anti human IgG; GE Healthcare Bio-Sciences AB, Sweden) were coupled on a CM4 chip (GE Healthcare BR-1005-34) at pH 5.0 by using an amine coupling kit supplied by the GE Healthcare. The sample and system buffer was PBS-T (10 mM phosphate buffered saline including 0.05% Tween20) pH 7.4. The temperature of the flow cell was set to 25.degree. C. and of the sample block to 12.degree. C. Before capturing, the flow cell was primed with running buffer twice.
[0322] The bispecific antibody was captured by injecting a 10 nM solution for 60 sec at a flow of 5 .mu.l/min. Independent binding of each ligand to the bispecific antibody was analysed by determining the active binding capacity for each ligand, either added sequentially or simultaneously (flow of 30 .mu.l/min): [0323] 1. Injection of human VEGF with a concentration of 200 nM for 180 sec (identifies the single binding of the antigen). [0324] 2. Injection of human Ang2 with a concentration of 100 nM for 180 sec (identifies single binding of the antigen). [0325] 3. Injection of human VEGF with a concentration of 200 nM for 180 sec followed by an additional injection of human Ang2 with a concentration of 100 nM for 180 sec (identifies binding of Ang2 in the presence of VEGF). [0326] 4. Injection of human Ang2 with a concentration of 100 nM for 180 sec followed by an additional injection of human VEGF with a concentration of 200 nM (identifies binding of VEGF in the presence of Ang2). [0327] 5. Co-Injection of human VEGF with a concentration of 200 nM and of human Ang2 with a concentration of 100 nM for 180 sec (identifies the binding of VEGF and of Ang2 at the same time).
[0328] The surface was regenerated by 60 sec washing with a 3 m MgCl2 solution at a flow rate of 30 .mu.l/min. Bulk refractive index differences were corrected by subtracting the response obtained from a goat anti human IgG surface.
[0329] The bispecific antibody is able to bind both antigens mutual independently if the resulting final signal of the approaches 3, 4 & 5 equals or is similar to the sum of the individual final signals of the approaches 1 and 2. Results are shown in Table 9, where both antibodies VEGFang2-0016, VEGFang2-0012 are shown to be able to bind mutual independently to VEGF and ANG2
[0330] Assessment of Simultaneous VEGF- and Ang2-binding to the <VEGF-ANG-2> Bispecific Antibodies
[0331] First, around 1600 resonance units (RU) of VEGF (20 .mu.g/ml) were coupled on a CM4 chip (GE Healthcare BR-1005-34) at pH 5.0 by using an amine coupling kit supplied by the GE Healthcare. The sample and system buffer was PBS-T (10 mM phosphate buffered saline including 0.05% Tween 20) pH 7.4. Flow cell was set to 25.degree. C. and sample block to 12.degree. C. and primed with running buffer twice. Second, 50 nM solution of the bispecific antibody was injected for 180 sec at a flow of 30 .mu.l/min. Third, hAng-2 was injected for 180 sec at a flow of 30 .mu.l/min. The binding response of hAng-2 depends from the amount of the bispecific antibody bound to VEGF and shows simultaneous binding. The surface was regenerated by 60 sec washing with a 0.85% H3PO4 solution at a flow rate of 30 .mu.l/min. Simultaneous binding is shown by an additional specific binding signal of hAng2 to the previous VEGF bound <VEGF-ANG-2> bispecific antibodies. For both bispecific antibodies VEGFang2-0015 and VEGFang2-0016 simultaneous VEGF- and Ang2-binding to the <VEGF-ANG-2> bispecific antibodies could be detected (data not shown).
TABLE-US-00007 TABLE 5 Results: Kinetic affinities to VEGF isoforms from different species VEGFang2- VEGFang2- VEGFang2- VEGFang2- 0015- 0016- 0012- 0201- apparent apparent apparent apparent affinity affinity affinity affinity Human VEGF .ltoreq.1 pM .ltoreq.1 pM .ltoreq.1 pM .ltoreq.1 pM 121 (out of (out of (out of (out of Biacore Biacore Biacore Biacore specification) specification) specification) specification) mouseVEGF no binding no binding no binding no binding 120 Rat VEGF 13 nM 14 nM 24 nM 35 nM 164
TABLE-US-00008 TABLE 6 Results: Solution affinities to Ang2 VEGFang2- VEGFang2- VEGFang2- VEGFang2-- 0015 KD 0016 KD 0012 KD 0201 KD [nM] [nM] [nM] [nM] humanAng2 8 20 20 tbd cynoAng2 5 13 10 tbd mouseAng2 8 13 8 tbd rabbitAng2 4 11 8 tbd
TABLE-US-00009 TABLE 7 Results: Affinity to FcRn of <VEGF-ANG-2> bispecific antibodies VEGFang2- VEGFang2- VEGFang2- VEGFang2-- 0015 0016 0012 0201 [affinity] [affinity] [affinity] [affinity] Human 0.8 .mu.M no binding no binding 0.8 .mu.M FcRn Cyno 0.9 .mu.M no binding no binding 1.0 .mu.M FcRn Mouse 0.2 .mu.M no binding no binding 0.2 .mu.M FcRn
TABLE-US-00010 TABLE 8 Results Binding to FcgammaRI-IIIa VEGFang2- VEGFang2- VEGFang2- VEGFang2- 0015 0016 0012 0201 FcyRIa No binding No binding Binding Binding FcyRIIa No binding No binding No binding Binding FcyRIIIa No binding No binding No binding Binding
TABLE-US-00011 TABLE 9 Results: Independent binding of VEGF-and Ang2 to <VEGF-ANG-2> bispecific antibodies 3) first 4) first 5) VEGF Ang2 Coinjection then then Ang2 + 1) Ang2 2) VEGF Ang2 VEGF VEGF [RUmax] [RUmax] [RUmax] [RUmax] [RUmax] VEGFang2- 174 50 211 211 211 0016 VEGFang2- 143 43 178 177 178 0012
Example 4
[0332] Mass Spectrometry
[0333] This section describes the characterization of <VEGF-ANG-2> bispecific antibodies with emphasis on the correct assembly. The expected primary structures were confirmed by electrospray ionization mass spectrometry (ESI-MS) of the deglycosylated, and intact or IdeS-digested (IgG-degrading enzyme of S. pyogenes) <VEGF-ANG-2> bispecific antibodies. The IdeS-digestion was performed with 100 .mu.g purified antibody incubated with 2 .mu.g IdeS protease (Roche) in 100 mmol/L NaH.sub.2PO.sub.4/Na.sub.2HPO.sub.4, pH 7.1 at 37.degree. C. for 5 h. Subsequently, the antibodies were deglycosylated with N-Glycosidase F, Neuraminidase and O-glycosidase (Roche) in 100 mmol/L NaH.sub.2PO.sub.4/Na.sub.2HPO.sub.4, pH 7.1 at 37.degree. C. for up to 16 h at a protein concentration of 1 mg/ml and subsequently desalted via HPLC on a Sephadex G25 column (GE Healthcare). The total mass was determined via ESI-MS on a maXis 4G UHR-QTOF MS system (Bruker Daltonik) equipped with a TriVersa NanoMate.RTM. source (Advion).
[0334] The masses obtained for the IdeS-digested, deglycosylated (Table 10), or intact, deglycosylated (Table 11) molecules correspond to the predicted masses deduced from the amino acid sequences for the <VEGF-ANG-2> bispecific antibodies consisting of two different light chains LC.sub.Ang2 and LC.sub.Lucentis, and two different heavy chains HC.sub.Ang2 and HC.sub.Lucentis.
TABLE-US-00012 TABLE 10 Masses of the deglycosylated and IdeS-digested bispecific <VEGF/ANG2> antibodies VEGFang2-0201 (without AAA mutation) andVEGFang2-0012 (with AAA mutation) F(ab')2 of the VEGF- Deglycosylated Fc of ANG-2> bispecific the VEGF-ANG-2> antibody bispecific antibody Predicted Observed Predicted Observed Average Average Average Average Mass Mass Mass Mass Sample [Da] [Da] [Da] [Da] VEGFang2- 99360.8 99360.7 47439.2 47430.1 0201 VEGFang2- 99360.8 99361.1 47087.7 47082.0 0012
TABLE-US-00013 TABLE 11 Masses of the deglycosylated <VEGF/ANG2> antibodies VEGFang2-0016 (with AAA mutation) and VEGFang2-0015 (without AAA mutation) Deglycosylated VEGF-ANG-2> bispecific antibody Predicted Observed Average Average Mass Mass [Da] [Da] VEGFang2- 146156.9 146161.2 0016 VEGFang2- 146505.3 146509.4 0015
Example 5
[0335] Fc-Rn Chromatography
[0336] Coupling to Streptavidin Sepharose:
[0337] One gram streptavidin Sepharose.RTM. (GE Healthcare) was added to the biotinylated and dialyzed receptor and incubated for two hours with shaking. The receptor derivatized sepharose was filled in a 1 ml XK column (GE Healthcare).
[0338] Chromatography Using the FcRn Affinity Column:
[0339] Conditions:
[0340] column dimensions: 50 mm.times.5 mm
[0341] bed height: 5 cm
[0342] loading: 50 .mu.g sample
[0343] equilibration buffer: 20 mM MES, with 150 mM NaCl, adjusted to pH 5.5
[0344] elution buffer: 20 mM Tris/HC1, with 150 mM NaCl, adjusted to pH 8.8
[0345] elution: 7.5 CV equilibration buffer, in 30 CV to 100% elution buffer, 10 CV elution buffer
[0346] Hu FcRn Affinity Column Chromatography
[0347] In the following table retention times of <VEGF-ANG-2> bispecific antibodies on affinity columns comprising human FcRn are given. Data were obtained using the conditions above. In the following Table retention times of <VEGF-ANG-2> bispecific antibodies on human FcRn are given.
TABLE-US-00014 TABLE 12 Results: retention times of <VEGF-ANG-2> bispecific antibodies retention time antibody [min] VEGFAng2-0015 78.5 (without AAA mutation) VEGFAng2-0201 78.9 (without AAA mutation) VEGFAng2-0012 2.7 (Void-peak) (with AAA mutation) VEGFAng2-0016 2.7 (Void-peak) (with AAA mutation)
Example 6
[0348] Pharmacokinetic(PK) Properties
[0349] PK Data with Fc-Rn Mice Transgenic for Human FcRn
[0350] In Life Phase
[0351] The study included female C57BL/6J mice (background); mouse FcRn deficient, but hemizygous transgenic for human FcRn (huFcRn, line 276-/tg)
[0352] Part 1
[0353] All mice were injected once intravitreally into the right eye with 2 .mu.L/animal of the appropriate solution (i.e. 21 .mu.g compound/animal (VEGFAng2-0015 (without AAA mutation) or 23.6 .mu.g compound/animal (VEGFAng2-0016 (with AAA mutation).
[0354] Mice were allocated to 2 groups with 6 animals each. Blood samples are taken from group 1 at 2, 24 and 96 hours and from group 2 at 7, 48 and 168 hours after dosing.
[0355] Injection into the vitreous of the right mouse eye was performed by using the NanoFil Microsyringe system for nanoliter injection from World Precision
[0356] Instruments, Inc., Berlin, Germany. Mice were anesthetized with 2.5% Isoflurane and for visualization of the mouse eye a Leica MZFL 3 microscope with a 40 fold magnification and a ring-light with a Leica KL 2500 LCD lightning was used. Subsequently, 2 .mu.L of the compound were injected using a 35-gauge needle.
[0357] Blood was collected via the retrobulbar venous plexus of the contralateral eye from each animal for the determination of the compound levels in serum.
[0358] Serum samples of at least 50 .mu.l were obtained from blood after 1 hour at RT by centrifugation (9300 xg) at 4.degree. C. for 3 min. Serum samples were frozen directly after centrifugation and stored frozen at -80.degree. C. until analysis. Treated eyes of the animals of group 1 were isolated 96 hours after treatment and of the animals of group 2 168 hours after treatment. Samples were stored frozen at -80.degree. C. until analysis.
[0359] Part 2
[0360] All mice were injected once intravenously via the tail vein with 200 .mu.L/animal of the appropriate solution (i.e. 21 .mu.g compound/animal (VEGFAng2-0015 (without AAA mutation) or 23.6 .mu.g compound/animal (VEGFAng2-0016 (with AAA mutation).
[0361] Mice were allocated to 2 groups with 5 animals each. Blood samples are taken from group 1 at 1, 24 and 96 hours and from group 2 at 7, 48 and 168 hours after dosing. Blood was collected via the retrobulbar venous plexus from each animal for the determination of the compound levels in serum.
[0362] Serum samples of at least 50 .mu.l were obtained from blood after 1 hour at RT by centrifugation (9300 xg) at 4.degree. C. for 3 min. Serum samples were frozen directly after centrifugation and stored frozen at -80.degree. C. until analysis.
[0363] Preparation of Whole Eye Lysates (Mice)
[0364] The eye lysates were gained by physico-chemical disintegration of the whole eye from laboratory animals. For mechanical disruption, each eye was transferred into a 1.5-mL micro vial with conical bottom. After freeze and thawing, the eyes were washed with 1 mL cell washing buffer once (Bio-Rad, Bio-Plex Cell Lysis Kit, Cat. No. 171-304011). In the following step, 5004 of freshly prepared cell lysis buffer were added and the eyes were grinded using a 1.5 mL tissue grinding pestle (Kimble Chase, 1.5 mL pestle, Art. No. 749521-1500). The mixture was then frozen and thawed five times and grinded again. To separate lysate from remaining tissue the samples were centrifuged for 4 min at 4500 g. After centrifuging the supernatant was collected and stored at -20.degree. C. until further analysis in the quantification ELISA.
[0365] Analysis
[0366] The concentrations of the <VEGF/ANG2> antibodies in mice serum and eye lysates were determined with an enzyme linked immunosorbent assay (ELISA)
[0367] For quantification of <VEGF/ANG2> antibodies in mouse serum samples and eye lysates, a standard solid-phase serial sandwich immunoassay with biotinylated and digoxigenated monoclonal antibodies used as capture and detection antibodies was performed. To verify the integrity of the bispecifity of the analyte the biotinylated capture antibody recognizes the anti-VEGF-binding site whereas the digoxigenated detection antibody will bind to the anti-Ang2 binding site of the analyte. The bound immune complex of capture antibody, analyte and detection antibody on the solid phase of the streptavidin coated micro titer plate (SA-MTP) is then detected with a horseradish-peroxidase coupled to an anti-digoxigenin antibody. After washing unbound material from the SA-MTP and addition of ABTS-substrate, the gained signal is proportional to the amount of analyte bound on the solid phase of the SA-MTP. Quantification is then done by converting the measured signals of the samples into concentrations referring to calibrators analyzed in parallel.
[0368] In a first step the SA-MTP was coated with 100 .mu.L/well of biotinylated capture antibody solution (mAb<Id<VEGF>>M-2.45.51-IgG-Bi(DDS)) with a concentration of 1 .mu.g/mL for one hour at 500 rpm on a MTP-shaker. Meanwhile calibrators, QC-samples and samples were prepared. Calibrators and QC-samples are diluted to 2% serum matrix; samples were diluted until the signals were within the linear range of the calibrators.
[0369] After coating the SA-MTP with capture antibody, the plate was washed three times with washing buffer and 300 .mu.L/well. Subsequently 100 .mu.l/well of the calibrators, QC-samples and samples were pipetted on the SA-MTP and incubated again for one hour at 500 rpm. The analyte was now bound with its anti-VEGF binding site via the capture antibody to the solid phase of the SA-MTP. After incubation and removal of unbound analyte by washing the plate 100 .mu.L/well of the first detection antibody (mAb<Id-<Ang2>>M-2.6.81-IgG-Dig(XOSu)) with a concentration of 250 ng/mL was added to the SA-MTP. Again, the plate was incubated for one hour at 500 rpm on a shaker. After washing, 100 .mu.L/well of the second detection antibody (pAb<Digoxigenin>S-Fab-POD (poly)) at a concentration of 50 mU/mL was added to the wells of the SA-MTP and the plate was incubated again for one hour at 500 rpm. After a final washing step to remove excess of detection antibody, 100 .mu.L/well substrate (ABTS) is added. The antibody-enzyme conjugate catalyzes the color reaction of the ABTS.RTM. substrate. The signal was then measured by an ELISA reader at 405 nm wavelength (reference wavelength: 490 nm ([405/490] nm)).
[0370] Pharmacokinetic Evaluation
[0371] The pharmacokinetic parameters were calculated by non-compartmental analysis, using the pharmacokinetic evaluation program WinNonlin.TM. (Pharsight), version 5.2.1.
[0372] Results: A) Serum Concentrations
[0373] Results for serum concentrations are shown in Tables 13 to 16 and FIG. 7B to 7C
TABLE-US-00015 TABLE 13 VEGFAng2-0015 (without AAA mutation): Comparison of serum concentrations after intravitreal and intravenous application Serum concentration Serum concentration after intravitreal after intravenous application application Average conc. Average conc. ID [.mu.g/mL] [.mu.g/mL] 1 h 17.7 2 h 9.8 7 h 10.4 12.1 24 h 6.4 8.3 48 h 6.5 6.9 96 h 3.4 4.1 168 h 2.9 2.7
TABLE-US-00016 TABLE 14 VEGFAng2-0016 (with AAA mutation): Comparison of serum concentrations after intravitreal and intravenous application Serum concentration Serum concentration after intravitreal after intravenous application application Average conc. Average conc. ID [.mu.g/mL] [.mu.g/mL] 1 h 18.4 2 h 7.0 7 h 8.7 10.0 24 h 2.2 3.3 48 h 1.0 1.0 96 h 0.1 0.1 168 h 0.0 0.0
TABLE-US-00017 TABLE 15 VEGFang2-0015 (without AAA mutation) and VEGFang2-0016 (with AAA mutation): Comparison of serum concentrations after intravitreal application) VEGFang2-0015 VEGFang2-0016 (without AAA mutation) (with AAA mutation) Average conc. Average conc. ID [.mu.g/mL] [.mu.g/mL] 2 h 9.8 7.0 7 h 10.4 8.7 24 h 6.4 2.2 48 h 6.5 1.0 96 h 3.4 0.1 168 h 2.9 0.0
TABLE-US-00018 TABLE 16 VEGFang2-0015 (without AAA mutation) and VEGFang2-0016 (with AAA mutation): Comparison of serum concentrations after intravenous application VEGFang2-0015 VEGFang2-0016 (without AAA mutation) (with AAA mutation) Average conc. Average conc. ID [.mu.g/mL] [.mu.g/mL] 1 h 17.7 18.4 7 h 12.1 10.0 24 h 8.3 3.3 48 h 6.9 1.0 96 h 4.1 0.1 168 h 2.7 0.0
[0374] Results: B) Concentrations in Eye-Lysates of Left and Right Eyes
[0375] Results for concentrations in eye lysates are shown in Tables 17 to 18 and FIGS. 7D to 7E
TABLE-US-00019 TABLE 17 a: Concentrations of VEGFang2-0015 (without AAA mutation) in eye lysates after intra vitreal application into right eye Mean conc. values from n = 6 mice ID mean conc. [ng/mL] 96 h Left eye 8.7 Right eye 46.1 168 h Left eye 4.3 Right eye t 12.9 b: Concentrations of VEGFang2-0015 (without AAA mutation) in eye lysates after intravenous application Mean conc. values from n = 5 mice ID mean conc. [ng/mL] 96 h Left eye 4.2 Right eye 7.5 168 h Left eye 3.4 Right eye 6.1
TABLE-US-00020 TABLE 18 a: Concentrations of VEGFang2-0016 (with AAA mutation) in eye lysates after intra vitreal application into right eye Mean conc. values from n = 5 mice ID mean conc. [ng/mL] 96 h Left eye 0.3 Right eye 34.5 168 h Left eye 0.1 Right eye 9.0 b: Concentrations of VEGFang2-0016 (with AAA mutation) in eye lysates after intravenous application Mean conc. values from n = 5 mice ID mean conc. [ng/mL] 96 h Left eye 0.0 Right eye 0.1 168 h Left eye 0.0 Right eye 0.1
[0376] Summary of Results:
[0377] After intravitreal application the bispecific <VEGF/ANG2> antibody according to the invention VEGFang2-0016 (with AAA mutation) shows similar concentrations (after 96 and 168 hours) in the eye lysates as compared to the bispecific <VEGF/ANG2> antibody without AAA mutation VEGFang2-0015.
[0378] Also after intravitreal application the bispecific <VEGF/ANG2> antibody according to the invention VEGFang2-0016 (with AAA mutation) shows in addition a faster clearance and shorter half-life in the serum as compared to the bispecific <VEGF/ANG2> antibody without AAA mutation VEGFang2-0015.
Example 7
[0379] Mouse Cornea Micropocket Angiogenesis Assay
[0380] To test the anti-angiogenic effect bispecific <VEGF/ANG2> antibody with the respective anti-VEGF VH and VL of SEQ ID NO: 7 and 8 and the anti-ANG2 VH and VL of SEQ ID NO: 15 and 16 on VEGF-induced angiogenesis in vivo, we perform the mouse corneal angiogenesis assay. In this assay a VEGF soaked Nylaflo.RTM. disc is implanted into a pocket of the avascular cornea at a fixed distance to the limbal vessels. Vessels immediately grow into the cornea towards the developing VEGF gradient. 8 to 10 weeks old female Balb/c mice were purchased from Charles River, Sulzfeld, Germany. The protocol is modified according to the method described by Rogers, M. S., et al., Nat. Protoc. 2 (2007) 2545-2550. Briefly, micropockets with a width of about 500 .mu.m are prepared under a microscope at approximately 1 mm from the limbus to the top of the cornea using a surgical blade and sharp tweezers in the anesthetized mouse. The disc (Nylaflo.RTM., Pall Corporation, Michigan) with a diameter of 0.6 mm is implanted and the surface of the implantation area was smoothened. Discs are incubated in corresponding growth factor or in vehicle for at least 30 min. After 3, 5 and 7 days (or alternatively only after 3, 5 or 7 days) , eyes are photographed and vascular response is measured. The assay is quantified by calculating the percentage of the area of new vessels per total area of the cornea.
[0381] The discs are loaded with 300 ng VEGF or with PBS as a control and implanted for 7 days. The outgrowth of vessels from the limbus to the disc is monitored over time on day 3, 5 and/or 7. One day prior to disc implantation the antibodies are administered intravenously at a dose of 10 mg/kg (due to the intravenous application the serum-stable VEGFang2-0015 (without AAA mutation) which only differs from VEGFang2-0016 by the AAA mutation and has the same anti-VEGF and anti-ANG2 VHs and VLs to mediate efficacy, is used as surrogate) for testing the anti-angiogenic effect on VEGF-induced angiogenesis in vivo. Animals in the control group receive vehicle. The application volume is 10 ml/kg.
Sequence CWU 1
1
50114PRTArtificialheavy chain CDR3H, <VEGF>ranibizumab 1Tyr
Pro Tyr Tyr Tyr Gly Thr Ser His Trp Tyr Phe Asp Val1 5
10217PRTArtificialheavy chain CDR2H, <VEGF>ranibizumab 2Trp
Ile Asn Thr Tyr Thr Gly Glu Pro Thr Tyr Ala Ala Asp Phe Lys1 5 10
15Arg35PRTArtificialheavy chain CDR1H, <VEGF>ranibizumab 3His
Tyr Gly Met Asn1 549PRTArtificiallight chain CDR3L,
<VEGF>ranibizumab 4Gln Gln Tyr Ser Thr Val Pro Trp Thr1
557PRTArtificiallight chain CDR2L, <VEGF>ranibizumab 5Phe Thr
Ser Ser Leu His Ser1 5611PRTArtificiallight chain CDR1L,
<VEGF>ranibizumab 6Ser Ala Ser Gln Asp Ile Ser Asn Tyr Leu
Asn1 5 107123PRTArtificialheavy chain variable domain VH,
<VEGF>ranibizumab 7Glu Val Gln Leu Val Glu Ser Gly Gly Gly
Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala Ser
Gly Tyr Asp Phe Thr His Tyr 20 25 30Gly Met Asn Trp Val Arg Gln Ala
Pro Gly Lys Gly Leu Glu Trp Val 35 40 45Gly Trp Ile Asn Thr Tyr Thr
Gly Glu Pro Thr Tyr Ala Ala Asp Phe 50 55 60Lys Arg Arg Phe Thr Phe
Ser Leu Asp Thr Ser Lys Ser Thr Ala Tyr65 70 75 80Leu Gln Met Asn
Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Lys Tyr
Pro Tyr Tyr Tyr Gly Thr Ser His Trp Tyr Phe Asp Val 100 105 110Trp
Gly Gln Gly Thr Leu Val Thr Val Ser Ser 115
1208107PRTArtificiallight chain variable domain VL,
<VEGF>ranibizumab 8Asp Ile Gln Leu Thr Gln Ser Pro Ser Ser
Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Ser Ala
Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro
Gly Lys Ala Pro Lys Val Leu Ile 35 40 45Tyr Phe Thr Ser Ser Leu His
Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp
Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala
Thr Tyr Tyr Cys Gln Gln Tyr Ser Thr Val Pro Trp 85 90 95Thr Phe Gly
Gln Gly Thr Lys Val Glu Ile Lys 100 105920PRTArtificialheavy chain
CDR3H, <ANG-2> Ang2i_LC10 variant 9Ser Pro Asn Pro Tyr Tyr
Tyr Asp Ser Ser Gly Tyr Tyr Tyr Pro Gly1 5 10 15Ala Phe Asp Ile
201017PRTArtificialheavy chain CDR2H, <ANG-2> Ang2i_LC10
variant 10Trp Ile Asn Pro Asn Ser Gly Gly Thr Asn Tyr Ala Gln Lys
Phe Gln1 5 10 15Gly115PRTArtificialheavy chain CDR1H, <ANG-2>
Ang2i_LC10 variant 11Gly Tyr Tyr Met His1 51211PRTArtificiallight
chain CDR3L, <ANG-2> Ang2i_LC10 variant 12Gln Val Trp Asp Ser
Ser Ser Asp His Trp Val1 5 10137PRTArtificiallight chain CDR2L,
<ANG-2> Ang2i_LC10 variant 13Asp Asp Ser Asp Arg Pro Ser1
51411PRTArtificiallight chain CDR1L, <ANG-2> Ang2i_LC10
variant 14Gly Gly Asn Asn Ile Gly Ser Lys Ser Val His1 5
1015129PRTArtificialheavy chain variable domain VH, <ANG-2>
Ang2i_LC10 variant 15Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val
Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly
Tyr Thr Phe Thr Gly Tyr 20 25 30Tyr Met His Trp Val Arg Gln Ala Pro
Gly Gln Gly Leu Glu Trp Met 35 40 45Gly Trp Ile Asn Pro Asn Ser Gly
Gly Thr Asn Tyr Ala Gln Lys Phe 50 55 60Gln Gly Arg Val Thr Met Thr
Arg Asp Thr Ser Ile Ser Thr Ala Tyr65 70 75 80Met Glu Leu Ser Arg
Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg Ser Pro
Asn Pro Tyr Tyr Tyr Asp Ser Ser Gly Tyr Tyr Tyr 100 105 110Pro Gly
Ala Phe Asp Ile Trp Gly Gln Gly Thr Met Val Thr Val Ser 115 120
125Ser16110PRTArtificiallight chain variable domain VL,
<ANG-2> Ang2i_LC10 variant 16Ser Tyr Val Leu Thr Gln Pro Pro
Ser Val Ser Val Ala Pro Gly Gln1 5 10 15Thr Ala Arg Ile Thr Cys Gly
Gly Asn Asn Ile Gly Ser Lys Ser Val 20 25 30His Trp Tyr Gln Gln Lys
Pro Gly Gln Ala Pro Val Leu Val Val Tyr 35 40 45Asp Asp Ser Asp Arg
Pro Ser Gly Ile Pro Glu Arg Phe Ser Gly Ser 50 55 60Asn Ser Gly Asn
Thr Ala Thr Leu Thr Ile Ser Arg Val Glu Ala Gly65 70 75 80Asp Glu
Ala Asp Tyr Tyr Cys Gln Val Trp Asp Ser Ser Ser Asp His 85 90 95Trp
Val Phe Gly Gly Gly Thr Lys Leu Thr Val Leu Gly Gln 100 105
11017191PRTHomo sapiens 17Met Asn Phe Leu Leu Ser Trp Val His Trp
Ser Leu Ala Leu Leu Leu1 5 10 15Tyr Leu His His Ala Lys Trp Ser Gln
Ala Ala Pro Met Ala Glu Gly 20 25 30Gly Gly Gln Asn His His Glu Val
Val Lys Phe Met Asp Val Tyr Gln 35 40 45Arg Ser Tyr Cys His Pro Ile
Glu Thr Leu Val Asp Ile Phe Gln Glu 50 55 60Tyr Pro Asp Glu Ile Glu
Tyr Ile Phe Lys Pro Ser Cys Val Pro Leu65 70 75 80Met Arg Cys Gly
Gly Cys Cys Asn Asp Glu Gly Leu Glu Cys Val Pro 85 90 95Thr Glu Glu
Ser Asn Ile Thr Met Gln Ile Met Arg Ile Lys Pro His 100 105 110Gln
Gly Gln His Ile Gly Glu Met Ser Phe Leu Gln His Asn Lys Cys 115 120
125Glu Cys Arg Pro Lys Lys Asp Arg Ala Arg Gln Glu Asn Pro Cys Gly
130 135 140Pro Cys Ser Glu Arg Arg Lys His Leu Phe Val Gln Asp Pro
Gln Thr145 150 155 160Cys Lys Cys Ser Cys Lys Asn Thr Asp Ser Arg
Cys Lys Ala Arg Gln 165 170 175Leu Glu Leu Asn Glu Arg Thr Cys Arg
Cys Asp Lys Pro Arg Arg 180 185 19018496PRTHomo sapiens 18Met Trp
Gln Ile Val Phe Phe Thr Leu Ser Cys Asp Leu Val Leu Ala1 5 10 15Ala
Ala Tyr Asn Asn Phe Arg Lys Ser Met Asp Ser Ile Gly Lys Lys 20 25
30Gln Tyr Gln Val Gln His Gly Ser Cys Ser Tyr Thr Phe Leu Leu Pro
35 40 45Glu Met Asp Asn Cys Arg Ser Ser Ser Ser Pro Tyr Val Ser Asn
Ala 50 55 60Val Gln Arg Asp Ala Pro Leu Glu Tyr Asp Asp Ser Val Gln
Arg Leu65 70 75 80Gln Val Leu Glu Asn Ile Met Glu Asn Asn Thr Gln
Trp Leu Met Lys 85 90 95Leu Glu Asn Tyr Ile Gln Asp Asn Met Lys Lys
Glu Met Val Glu Ile 100 105 110Gln Gln Asn Ala Val Gln Asn Gln Thr
Ala Val Met Ile Glu Ile Gly 115 120 125Thr Asn Leu Leu Asn Gln Thr
Ala Glu Gln Thr Arg Lys Leu Thr Asp 130 135 140Val Glu Ala Gln Val
Leu Asn Gln Thr Thr Arg Leu Glu Leu Gln Leu145 150 155 160Leu Glu
His Ser Leu Ser Thr Asn Lys Leu Glu Lys Gln Ile Leu Asp 165 170
175Gln Thr Ser Glu Ile Asn Lys Leu Gln Asp Lys Asn Ser Phe Leu Glu
180 185 190Lys Lys Val Leu Ala Met Glu Asp Lys His Ile Ile Gln Leu
Gln Ser 195 200 205Ile Lys Glu Glu Lys Asp Gln Leu Gln Val Leu Val
Ser Lys Gln Asn 210 215 220Ser Ile Ile Glu Glu Leu Glu Lys Lys Ile
Val Thr Ala Thr Val Asn225 230 235 240Asn Ser Val Leu Gln Lys Gln
Gln His Asp Leu Met Glu Thr Val Asn 245 250 255Asn Leu Leu Thr Met
Met Ser Thr Ser Asn Ser Ala Lys Asp Pro Thr 260 265 270Val Ala Lys
Glu Glu Gln Ile Ser Phe Arg Asp Cys Ala Glu Val Phe 275 280 285Lys
Ser Gly His Thr Thr Asn Gly Ile Tyr Thr Leu Thr Phe Pro Asn 290 295
300Ser Thr Glu Glu Ile Lys Ala Tyr Cys Asp Met Glu Ala Gly Gly
Gly305 310 315 320Gly Trp Thr Ile Ile Gln Arg Arg Glu Asp Gly Ser
Val Asp Phe Gln 325 330 335Arg Thr Trp Lys Glu Tyr Lys Val Gly Phe
Gly Asn Pro Ser Gly Glu 340 345 350Tyr Trp Leu Gly Asn Glu Phe Val
Ser Gln Leu Thr Asn Gln Gln Arg 355 360 365Tyr Val Leu Lys Ile His
Leu Lys Asp Trp Glu Gly Asn Glu Ala Tyr 370 375 380Ser Leu Tyr Glu
His Phe Tyr Leu Ser Ser Glu Glu Leu Asn Tyr Arg385 390 395 400Ile
His Leu Lys Gly Leu Thr Gly Thr Ala Gly Lys Ile Ser Ser Ile 405 410
415Ser Gln Pro Gly Asn Asp Phe Ser Thr Lys Asp Gly Asp Asn Asp Lys
420 425 430Cys Ile Cys Lys Cys Ser Gln Met Leu Thr Gly Gly Trp Trp
Phe Asp 435 440 445Ala Cys Gly Pro Ser Asn Leu Asn Gly Met Tyr Tyr
Pro Gln Arg Gln 450 455 460Asn Thr Asn Lys Phe Asn Gly Ile Lys Trp
Tyr Tyr Trp Lys Gly Ser465 470 475 480Gly Tyr Ser Leu Lys Ala Thr
Thr Met Met Ile Arg Pro Ala Asp Phe 485 490 49519498PRTHomo sapiens
19Met Thr Val Phe Leu Ser Phe Ala Phe Leu Ala Ala Ile Leu Thr His1
5 10 15Ile Gly Cys Ser Asn Gln Arg Arg Ser Pro Glu Asn Ser Gly Arg
Arg 20 25 30Tyr Asn Arg Ile Gln His Gly Gln Cys Ala Tyr Thr Phe Ile
Leu Pro 35 40 45Glu His Asp Gly Asn Cys Arg Glu Ser Thr Thr Asp Gln
Tyr Asn Thr 50 55 60Asn Ala Leu Gln Arg Asp Ala Pro His Val Glu Pro
Asp Phe Ser Ser65 70 75 80Gln Lys Leu Gln His Leu Glu His Val Met
Glu Asn Tyr Thr Gln Trp 85 90 95Leu Gln Lys Leu Glu Asn Tyr Ile Val
Glu Asn Met Lys Ser Glu Met 100 105 110Ala Gln Ile Gln Gln Asn Ala
Val Gln Asn His Thr Ala Thr Met Leu 115 120 125Glu Ile Gly Thr Ser
Leu Leu Ser Gln Thr Ala Glu Gln Thr Arg Lys 130 135 140Leu Thr Asp
Val Glu Thr Gln Val Leu Asn Gln Thr Ser Arg Leu Glu145 150 155
160Ile Gln Leu Leu Glu Asn Ser Leu Ser Thr Tyr Lys Leu Glu Lys Gln
165 170 175Leu Leu Gln Gln Thr Asn Glu Ile Leu Lys Ile His Glu Lys
Asn Ser 180 185 190Leu Leu Glu His Lys Ile Leu Glu Met Glu Gly Lys
His Lys Glu Glu 195 200 205Leu Asp Thr Leu Lys Glu Glu Lys Glu Asn
Leu Gln Gly Leu Val Thr 210 215 220Arg Gln Thr Tyr Ile Ile Gln Glu
Leu Glu Lys Gln Leu Asn Arg Ala225 230 235 240Thr Thr Asn Asn Ser
Val Leu Gln Lys Gln Gln Leu Glu Leu Met Asp 245 250 255Thr Val His
Asn Leu Val Asn Leu Cys Thr Lys Glu Gly Val Leu Leu 260 265 270Lys
Gly Gly Lys Arg Glu Glu Glu Lys Pro Phe Arg Asp Cys Ala Asp 275 280
285Val Tyr Gln Ala Gly Phe Asn Lys Ser Gly Ile Tyr Thr Ile Tyr Ile
290 295 300Asn Asn Met Pro Glu Pro Lys Lys Val Phe Cys Asn Met Asp
Val Asn305 310 315 320Gly Gly Gly Trp Thr Val Ile Gln His Arg Glu
Asp Gly Ser Leu Asp 325 330 335Phe Gln Arg Gly Trp Lys Glu Tyr Lys
Met Gly Phe Gly Asn Pro Ser 340 345 350Gly Glu Tyr Trp Leu Gly Asn
Glu Phe Ile Phe Ala Ile Thr Ser Gln 355 360 365Arg Gln Tyr Met Leu
Arg Ile Glu Leu Met Asp Trp Glu Gly Asn Arg 370 375 380Ala Tyr Ser
Gln Tyr Asp Arg Phe His Ile Gly Asn Glu Lys Gln Asn385 390 395
400Tyr Arg Leu Tyr Leu Lys Gly His Thr Gly Thr Ala Gly Lys Gln Ser
405 410 415Ser Leu Ile Leu His Gly Ala Asp Phe Ser Thr Lys Asp Ala
Asp Asn 420 425 430Asp Asn Cys Met Cys Lys Cys Ala Leu Met Leu Thr
Gly Gly Trp Trp 435 440 445Phe Asp Ala Cys Gly Pro Ser Asn Leu Asn
Gly Met Phe Tyr Thr Ala 450 455 460Gly Gln Asn His Gly Lys Leu Asn
Gly Ile Lys Trp His Tyr Phe Lys465 470 475 480Gly Pro Ser Tyr Ser
Leu Arg Ser Thr Thr Met Met Ile Arg Pro Leu 485 490 495Asp
Phe201124PRTHomo sapiens 20Met Asp Ser Leu Ala Ser Leu Val Leu Cys
Gly Val Ser Leu Leu Leu1 5 10 15Ser Gly Thr Val Glu Gly Ala Met Asp
Leu Ile Leu Ile Asn Ser Leu 20 25 30Pro Leu Val Ser Asp Ala Glu Thr
Ser Leu Thr Cys Ile Ala Ser Gly 35 40 45Trp Arg Pro His Glu Pro Ile
Thr Ile Gly Arg Asp Phe Glu Ala Leu 50 55 60Met Asn Gln His Gln Asp
Pro Leu Glu Val Thr Gln Asp Val Thr Arg65 70 75 80Glu Trp Ala Lys
Lys Val Val Trp Lys Arg Glu Lys Ala Ser Lys Ile 85 90 95Asn Gly Ala
Tyr Phe Cys Glu Gly Arg Val Arg Gly Glu Ala Ile Arg 100 105 110Ile
Arg Thr Met Lys Met Arg Gln Gln Ala Ser Phe Leu Pro Ala Thr 115 120
125Leu Thr Met Thr Val Asp Lys Gly Asp Asn Val Asn Ile Ser Phe Lys
130 135 140Lys Val Leu Ile Lys Glu Glu Asp Ala Val Ile Tyr Lys Asn
Gly Ser145 150 155 160Phe Ile His Ser Val Pro Arg His Glu Val Pro
Asp Ile Leu Glu Val 165 170 175His Leu Pro His Ala Gln Pro Gln Asp
Ala Gly Val Tyr Ser Ala Arg 180 185 190Tyr Ile Gly Gly Asn Leu Phe
Thr Ser Ala Phe Thr Arg Leu Ile Val 195 200 205Arg Arg Cys Glu Ala
Gln Lys Trp Gly Pro Glu Cys Asn His Leu Cys 210 215 220Thr Ala Cys
Met Asn Asn Gly Val Cys His Glu Asp Thr Gly Glu Cys225 230 235
240Ile Cys Pro Pro Gly Phe Met Gly Arg Thr Cys Glu Lys Ala Cys Glu
245 250 255Leu His Thr Phe Gly Arg Thr Cys Lys Glu Arg Cys Ser Gly
Gln Glu 260 265 270Gly Cys Lys Ser Tyr Val Phe Cys Leu Pro Asp Pro
Tyr Gly Cys Ser 275 280 285Cys Ala Thr Gly Trp Lys Gly Leu Gln Cys
Asn Glu Ala Cys His Pro 290 295 300Gly Phe Tyr Gly Pro Asp Cys Lys
Leu Arg Cys Ser Cys Asn Asn Gly305 310 315 320Glu Met Cys Asp Arg
Phe Gln Gly Cys Leu Cys Ser Pro Gly Trp Gln 325 330 335Gly Leu Gln
Cys Glu Arg Glu Gly Ile Pro Arg Met Thr Pro Lys Ile 340 345 350Val
Asp Leu Pro Asp His Ile Glu Val Asn Ser Gly Lys Phe Asn Pro 355 360
365Ile Cys Lys Ala Ser Gly Trp Pro Leu Pro Thr Asn Glu Glu Met Thr
370 375 380Leu Val Lys Pro Asp Gly Thr Val Leu His Pro Lys Asp Phe
Asn His385 390 395 400Thr Asp His Phe Ser Val Ala Ile Phe Thr Ile
His Arg Ile Leu Pro 405 410 415Pro Asp Ser Gly Val Trp Val Cys Ser
Val Asn Thr Val Ala Gly Met 420 425 430Val Glu Lys Pro Phe Asn Ile
Ser Val Lys Val Leu Pro Lys Pro Leu 435 440 445Asn Ala Pro Asn Val
Ile Asp Thr Gly His Asn Phe Ala Val Ile Asn 450 455 460Ile Ser Ser
Glu Pro Tyr Phe Gly Asp Gly Pro Ile Lys Ser Lys Lys465 470 475
480Leu Leu Tyr Lys Pro Val Asn His Tyr Glu Ala Trp Gln His Ile Gln
485 490 495Val Thr Asn Glu Ile Val Thr Leu Asn Tyr Leu Glu Pro Arg
Thr Glu 500 505 510Tyr Glu Leu Cys Val Gln Leu Val Arg Arg Gly Glu
Gly Gly
Glu Gly 515 520 525His Pro Gly Pro Val Arg Arg Phe Thr Thr Ala Ser
Ile Gly Leu Pro 530 535 540Pro Pro Arg Gly Leu Asn Leu Leu Pro Lys
Ser Gln Thr Thr Leu Asn545 550 555 560Leu Thr Trp Gln Pro Ile Phe
Pro Ser Ser Glu Asp Asp Phe Tyr Val 565 570 575Glu Val Glu Arg Arg
Ser Val Gln Lys Ser Asp Gln Gln Asn Ile Lys 580 585 590Val Pro Gly
Asn Leu Thr Ser Val Leu Leu Asn Asn Leu His Pro Arg 595 600 605Glu
Gln Tyr Val Val Arg Ala Arg Val Asn Thr Lys Ala Gln Gly Glu 610 615
620Trp Ser Glu Asp Leu Thr Ala Trp Thr Leu Ser Asp Ile Leu Pro
Pro625 630 635 640Gln Pro Glu Asn Ile Lys Ile Ser Asn Ile Thr His
Ser Ser Ala Val 645 650 655Ile Ser Trp Thr Ile Leu Asp Gly Tyr Ser
Ile Ser Ser Ile Thr Ile 660 665 670Arg Tyr Lys Val Gln Gly Lys Asn
Glu Asp Gln His Val Asp Val Lys 675 680 685Ile Lys Asn Ala Thr Ile
Thr Gln Tyr Gln Leu Lys Gly Leu Glu Pro 690 695 700Glu Thr Ala Tyr
Gln Val Asp Ile Phe Ala Glu Asn Asn Ile Gly Ser705 710 715 720Ser
Asn Pro Ala Phe Ser His Glu Leu Val Thr Leu Pro Glu Ser Gln 725 730
735Ala Pro Ala Asp Leu Gly Gly Gly Lys Met Leu Leu Ile Ala Ile Leu
740 745 750Gly Ser Ala Gly Met Thr Cys Leu Thr Val Leu Leu Ala Phe
Leu Ile 755 760 765Ile Leu Gln Leu Lys Arg Ala Asn Val Gln Arg Arg
Met Ala Gln Ala 770 775 780Phe Gln Asn Val Arg Glu Glu Pro Ala Val
Gln Phe Asn Ser Gly Thr785 790 795 800Leu Ala Leu Asn Arg Lys Val
Lys Asn Asn Pro Asp Pro Thr Ile Tyr 805 810 815Pro Val Leu Asp Trp
Asn Asp Ile Lys Phe Gln Asp Val Ile Gly Glu 820 825 830Gly Asn Phe
Gly Gln Val Leu Lys Ala Arg Ile Lys Lys Asp Gly Leu 835 840 845Arg
Met Asp Ala Ala Ile Lys Arg Met Lys Glu Tyr Ala Ser Lys Asp 850 855
860Asp His Arg Asp Phe Ala Gly Glu Leu Glu Val Leu Cys Lys Leu
Gly865 870 875 880His His Pro Asn Ile Ile Asn Leu Leu Gly Ala Cys
Glu His Arg Gly 885 890 895Tyr Leu Tyr Leu Ala Ile Glu Tyr Ala Pro
His Gly Asn Leu Leu Asp 900 905 910Phe Leu Arg Lys Ser Arg Val Leu
Glu Thr Asp Pro Ala Phe Ala Ile 915 920 925Ala Asn Ser Thr Ala Ser
Thr Leu Ser Ser Gln Gln Leu Leu His Phe 930 935 940Ala Ala Asp Val
Ala Arg Gly Met Asp Tyr Leu Ser Gln Lys Gln Phe945 950 955 960Ile
His Arg Asp Leu Ala Ala Arg Asn Ile Leu Val Gly Glu Asn Tyr 965 970
975Val Ala Lys Ile Ala Asp Phe Gly Leu Ser Arg Gly Gln Glu Val Tyr
980 985 990Val Lys Lys Thr Met Gly Arg Leu Pro Val Arg Trp Met Ala
Ile Glu 995 1000 1005Ser Leu Asn Tyr Ser Val Tyr Thr Thr Asn Ser
Asp Val Trp Ser 1010 1015 1020Tyr Gly Val Leu Leu Trp Glu Ile Val
Ser Leu Gly Gly Thr Pro 1025 1030 1035Tyr Cys Gly Met Thr Cys Ala
Glu Leu Tyr Glu Lys Leu Pro Gln 1040 1045 1050Gly Tyr Arg Leu Glu
Lys Pro Leu Asn Cys Asp Asp Glu Val Tyr 1055 1060 1065Asp Leu Met
Arg Gln Cys Trp Arg Glu Lys Pro Tyr Glu Arg Pro 1070 1075 1080Ser
Phe Ala Gln Ile Leu Val Ser Leu Asn Arg Met Leu Glu Glu 1085 1090
1095Arg Lys Thr Tyr Val Asn Thr Thr Leu Tyr Glu Lys Phe Thr Tyr
1100 1105 1110Ala Gly Ile Asp Cys Ser Ala Glu Glu Ala Ala 1115
112021453PRTArtificialHeavy chain 1 of <VEGF-ANG-2> CrossMAb
IgG1 with AAA mutations (VEGFang2-0012) 21Glu Val Gln Leu Val Glu
Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser
Cys Ala Ala Ser Gly Tyr Asp Phe Thr His Tyr 20 25 30Gly Met Asn Trp
Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40 45Gly Trp Ile
Asn Thr Tyr Thr Gly Glu Pro Thr Tyr Ala Ala Asp Phe 50 55 60Lys Arg
Arg Phe Thr Phe Ser Leu Asp Thr Ser Lys Ser Thr Ala Tyr65 70 75
80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95Ala Lys Tyr Pro Tyr Tyr Tyr Gly Thr Ser His Trp Tyr Phe Asp
Val 100 105 110Trp Gly Gln Gly Thr Leu Val Thr Val Ser Ser Ala Ser
Thr Lys Gly 115 120 125Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys
Ser Thr Ser Gly Gly 130 135 140Thr Ala Ala Leu Gly Cys Leu Val Lys
Asp Tyr Phe Pro Glu Pro Val145 150 155 160Thr Val Ser Trp Asn Ser
Gly Ala Leu Thr Ser Gly Val His Thr Phe 165 170 175Pro Ala Val Leu
Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser Val Val 180 185 190Thr Val
Pro Ser Ser Ser Leu Gly Thr Gln Thr Tyr Ile Cys Asn Val 195 200
205Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys Lys Val Glu Pro Lys
210 215 220Ser Cys Asp Lys Thr His Thr Cys Pro Pro Cys Pro Ala Pro
Glu Leu225 230 235 240Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro
Lys Pro Lys Asp Thr 245 250 255Leu Met Ala Ser Arg Thr Pro Glu Val
Thr Cys Val Val Val Asp Val 260 265 270Ser His Glu Asp Pro Glu Val
Lys Phe Asn Trp Tyr Val Asp Gly Val 275 280 285Glu Val His Asn Ala
Lys Thr Lys Pro Arg Glu Glu Gln Tyr Asn Ser 290 295 300Thr Tyr Arg
Val Val Ser Val Leu Thr Val Leu Ala Gln Asp Trp Leu305 310 315
320Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn Lys Ala Leu Pro Ala
325 330 335Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg
Glu Pro 340 345 350Gln Val Tyr Thr Leu Pro Pro Cys Arg Asp Glu Leu
Thr Lys Asn Gln 355 360 365Val Ser Leu Trp Cys Leu Val Lys Gly Phe
Tyr Pro Ser Asp Ile Ala 370 375 380Val Glu Trp Glu Ser Asn Gly Gln
Pro Glu Asn Asn Tyr Lys Thr Thr385 390 395 400Pro Pro Val Leu Asp
Ser Asp Gly Ser Phe Phe Leu Tyr Ser Lys Leu 405 410 415Thr Val Asp
Lys Ser Arg Trp Gln Gln Gly Asn Val Phe Ser Cys Ser 420 425 430Val
Met His Glu Ala Leu His Asn Ala Tyr Thr Gln Lys Ser Leu Ser 435 440
445Leu Ser Pro Gly Lys 45022463PRTArtificialHeavy chain 2 of
<VEGF-ANG-2> CrossMAb IgG1 with AAA mutations (VEGFang2-0012)
22Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1
5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Gly
Tyr 20 25 30Tyr Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu
Trp Met 35 40 45Gly Trp Ile Asn Pro Asn Ser Gly Gly Thr Asn Tyr Ala
Gln Lys Phe 50 55 60Gln Gly Arg Val Thr Met Thr Arg Asp Thr Ser Ile
Ser Thr Ala Tyr65 70 75 80Met Glu Leu Ser Arg Leu Arg Ser Asp Asp
Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg Ser Pro Asn Pro Tyr Tyr Tyr
Asp Ser Ser Gly Tyr Tyr Tyr 100 105 110Pro Gly Ala Phe Asp Ile Trp
Gly Gln Gly Thr Met Val Thr Val Ser 115 120 125Ser Ala Ser Val Ala
Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp 130 135 140Glu Gln Leu
Lys Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn145 150 155
160Phe Tyr Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu
165 170 175Gln Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser
Lys Asp 180 185 190Ser Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser
Lys Ala Asp Tyr 195 200 205Glu Lys His Lys Val Tyr Ala Cys Glu Val
Thr His Gln Gly Leu Ser 210 215 220Ser Pro Val Thr Lys Ser Phe Asn
Arg Gly Glu Cys Asp Lys Thr His225 230 235 240Thr Cys Pro Pro Cys
Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val 245 250 255Phe Leu Phe
Pro Pro Lys Pro Lys Asp Thr Leu Met Ala Ser Arg Thr 260 265 270Pro
Glu Val Thr Cys Val Val Val Asp Val Ser His Glu Asp Pro Glu 275 280
285Val Lys Phe Asn Trp Tyr Val Asp Gly Val Glu Val His Asn Ala Lys
290 295 300Thr Lys Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val
Val Ser305 310 315 320Val Leu Thr Val Leu Ala Gln Asp Trp Leu Asn
Gly Lys Glu Tyr Lys 325 330 335Cys Lys Val Ser Asn Lys Ala Leu Pro
Ala Pro Ile Glu Lys Thr Ile 340 345 350Ser Lys Ala Lys Gly Gln Pro
Arg Glu Pro Gln Val Cys Thr Leu Pro 355 360 365Pro Ser Arg Asp Glu
Leu Thr Lys Asn Gln Val Ser Leu Ser Cys Ala 370 375 380Val Lys Gly
Phe Tyr Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn385 390 395
400Gly Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser
405 410 415Asp Gly Ser Phe Phe Leu Val Ser Lys Leu Thr Val Asp Lys
Ser Arg 420 425 430Trp Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met
His Glu Ala Leu 435 440 445His Asn Ala Tyr Thr Gln Lys Ser Leu Ser
Leu Ser Pro Gly Lys 450 455 46023214PRTArtificialLight chain 1 of
<VEGF-ANG-2> CrossMAb IgG1 with AAA mutations (VEGFang2-0012)
23Asp Ile Gln Leu Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1
5 10 15Asp Arg Val Thr Ile Thr Cys Ser Ala Ser Gln Asp Ile Ser Asn
Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Val
Leu Ile 35 40 45Tyr Phe Thr Ser Ser Leu His Ser Gly Val Pro Ser Arg
Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser
Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln
Tyr Ser Thr Val Pro Trp 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu
Ile Lys Arg Thr Val Ala Ala 100 105 110Pro Ser Val Phe Ile Phe Pro
Pro Ser Asp Glu Gln Leu Lys Ser Gly 115 120 125Thr Ala Ser Val Val
Cys Leu Leu Asn Asn Phe Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln
Trp Lys Val Asp Asn Ala Leu Gln Ser Gly Asn Ser Gln145 150 155
160Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser
165 170 175Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu Lys His Lys
Val Tyr 180 185 190Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser Pro
Val Thr Lys Ser 195 200 205Phe Asn Arg Gly Glu Cys
21024213PRTArtificialLight chain 2 of <VEGF-ANG-2> CrossMAb
IgG1 with AAA mutations (VEGF-Ang2-0012) 24Ser Tyr Val Leu Thr Gln
Pro Pro Ser Val Ser Val Ala Pro Gly Gln1 5 10 15Thr Ala Arg Ile Thr
Cys Gly Gly Asn Asn Ile Gly Ser Lys Ser Val 20 25 30His Trp Tyr Gln
Gln Lys Pro Gly Gln Ala Pro Val Leu Val Val Tyr 35 40 45Asp Asp Ser
Asp Arg Pro Ser Gly Ile Pro Glu Arg Phe Ser Gly Ser 50 55 60Asn Ser
Gly Asn Thr Ala Thr Leu Thr Ile Ser Arg Val Glu Ala Gly65 70 75
80Asp Glu Ala Asp Tyr Tyr Cys Gln Val Trp Asp Ser Ser Ser Asp His
85 90 95Trp Val Phe Gly Gly Gly Thr Lys Leu Thr Val Leu Ser Ser Ala
Ser 100 105 110Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser
Lys Ser Thr 115 120 125Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val
Lys Asp Tyr Phe Pro 130 135 140Glu Pro Val Thr Val Ser Trp Asn Ser
Gly Ala Leu Thr Ser Gly Val145 150 155 160His Thr Phe Pro Ala Val
Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser 165 170 175Ser Val Val Thr
Val Pro Ser Ser Ser Leu Gly Thr Gln Thr Tyr Ile 180 185 190Cys Asn
Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys Lys Val 195 200
205Glu Pro Lys Ser Cys 21025453PRTArtificialHeavy chain 1 of
<VEGF-ANG-2> CrossMAb IgG1 with AAA mutations and P329G LALA
mutations (VEGFang2-0016) 25Glu Val Gln Leu Val Glu Ser Gly Gly Gly
Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala Ser
Gly Tyr Asp Phe Thr His Tyr 20 25 30Gly Met Asn Trp Val Arg Gln Ala
Pro Gly Lys Gly Leu Glu Trp Val 35 40 45Gly Trp Ile Asn Thr Tyr Thr
Gly Glu Pro Thr Tyr Ala Ala Asp Phe 50 55 60Lys Arg Arg Phe Thr Phe
Ser Leu Asp Thr Ser Lys Ser Thr Ala Tyr65 70 75 80Leu Gln Met Asn
Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Lys Tyr
Pro Tyr Tyr Tyr Gly Thr Ser His Trp Tyr Phe Asp Val 100 105 110Trp
Gly Gln Gly Thr Leu Val Thr Val Ser Ser Ala Ser Thr Lys Gly 115 120
125Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly Gly
130 135 140Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu
Pro Val145 150 155 160Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser
Gly Val His Thr Phe 165 170 175Pro Ala Val Leu Gln Ser Ser Gly Leu
Tyr Ser Leu Ser Ser Val Val 180 185 190Thr Val Pro Ser Ser Ser Leu
Gly Thr Gln Thr Tyr Ile Cys Asn Val 195 200 205Asn His Lys Pro Ser
Asn Thr Lys Val Asp Lys Lys Val Glu Pro Lys 210 215 220Ser Cys Asp
Lys Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu Ala225 230 235
240Ala Gly Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr
245 250 255Leu Met Ala Ser Arg Thr Pro Glu Val Thr Cys Val Val Val
Asp Val 260 265 270Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp Tyr
Val Asp Gly Val 275 280 285Glu Val His Asn Ala Lys Thr Lys Pro Arg
Glu Glu Gln Tyr Asn Ser 290 295 300Thr Tyr Arg Val Val Ser Val Leu
Thr Val Leu Ala Gln Asp Trp Leu305 310 315 320Asn Gly Lys Glu Tyr
Lys Cys Lys Val Ser Asn Lys Ala Leu Gly Ala 325 330 335Pro Ile Glu
Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro 340 345 350Gln
Val Tyr Thr Leu Pro Pro Cys Arg Asp Glu Leu Thr Lys Asn Gln 355 360
365Val Ser Leu Trp Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala
370 375 380Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys
Thr Thr385 390 395 400Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe
Leu Tyr Ser Lys Leu 405 410 415Thr Val Asp Lys Ser Arg Trp Gln Gln
Gly Asn Val Phe Ser Cys Ser 420 425 430Val Met His Glu Ala Leu His
Asn Ala Tyr Thr Gln Lys Ser Leu Ser 435 440 445Leu Ser Pro Gly Lys
45026463PRTArtificialHeavy
chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with AAA mutations and
P329G LALA mutations (VEGFang2-0016) 26Gln Val Gln Leu Val Gln Ser
Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys
Lys Ala Ser Gly Tyr Thr Phe Thr Gly Tyr 20 25 30Tyr Met His Trp Val
Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40 45Gly Trp Ile Asn
Pro Asn Ser Gly Gly Thr Asn Tyr Ala Gln Lys Phe 50 55 60Gln Gly Arg
Val Thr Met Thr Arg Asp Thr Ser Ile Ser Thr Ala Tyr65 70 75 80Met
Glu Leu Ser Arg Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr Cys 85 90
95Ala Arg Ser Pro Asn Pro Tyr Tyr Tyr Asp Ser Ser Gly Tyr Tyr Tyr
100 105 110Pro Gly Ala Phe Asp Ile Trp Gly Gln Gly Thr Met Val Thr
Val Ser 115 120 125Ser Ala Ser Val Ala Ala Pro Ser Val Phe Ile Phe
Pro Pro Ser Asp 130 135 140Glu Gln Leu Lys Ser Gly Thr Ala Ser Val
Val Cys Leu Leu Asn Asn145 150 155 160Phe Tyr Pro Arg Glu Ala Lys
Val Gln Trp Lys Val Asp Asn Ala Leu 165 170 175Gln Ser Gly Asn Ser
Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp 180 185 190Ser Thr Tyr
Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr 195 200 205Glu
Lys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser 210 215
220Ser Pro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys Asp Lys Thr
His225 230 235 240Thr Cys Pro Pro Cys Pro Ala Pro Glu Ala Ala Gly
Gly Pro Ser Val 245 250 255Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr
Leu Met Ala Ser Arg Thr 260 265 270Pro Glu Val Thr Cys Val Val Val
Asp Val Ser His Glu Asp Pro Glu 275 280 285Val Lys Phe Asn Trp Tyr
Val Asp Gly Val Glu Val His Asn Ala Lys 290 295 300Thr Lys Pro Arg
Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val Ser305 310 315 320Val
Leu Thr Val Leu Ala Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys 325 330
335Cys Lys Val Ser Asn Lys Ala Leu Gly Ala Pro Ile Glu Lys Thr Ile
340 345 350Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val Cys Thr
Leu Pro 355 360 365Pro Ser Arg Asp Glu Leu Thr Lys Asn Gln Val Ser
Leu Ser Cys Ala 370 375 380Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala
Val Glu Trp Glu Ser Asn385 390 395 400Gly Gln Pro Glu Asn Asn Tyr
Lys Thr Thr Pro Pro Val Leu Asp Ser 405 410 415Asp Gly Ser Phe Phe
Leu Val Ser Lys Leu Thr Val Asp Lys Ser Arg 420 425 430Trp Gln Gln
Gly Asn Val Phe Ser Cys Ser Val Met His Glu Ala Leu 435 440 445His
Asn Ala Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys 450 455
46027214PRTArtificialLight chain 1 of <VEGF-ANG-2> CrossMAb
IgG1 with AAA mutations and P329G LALA mutations (VEGFang2-0016)
27Asp Ile Gln Leu Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1
5 10 15Asp Arg Val Thr Ile Thr Cys Ser Ala Ser Gln Asp Ile Ser Asn
Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Val
Leu Ile 35 40 45Tyr Phe Thr Ser Ser Leu His Ser Gly Val Pro Ser Arg
Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser
Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln
Tyr Ser Thr Val Pro Trp 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu
Ile Lys Arg Thr Val Ala Ala 100 105 110Pro Ser Val Phe Ile Phe Pro
Pro Ser Asp Glu Gln Leu Lys Ser Gly 115 120 125Thr Ala Ser Val Val
Cys Leu Leu Asn Asn Phe Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln
Trp Lys Val Asp Asn Ala Leu Gln Ser Gly Asn Ser Gln145 150 155
160Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser
165 170 175Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu Lys His Lys
Val Tyr 180 185 190Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser Pro
Val Thr Lys Ser 195 200 205Phe Asn Arg Gly Glu Cys
21028213PRTArtificialLight chain 2 of <VEGF-ANG-2> CrossMAb
IgG1 with AAA mutations and P329G LALA mutations (VEGFang2-0016)
28Ser Tyr Val Leu Thr Gln Pro Pro Ser Val Ser Val Ala Pro Gly Gln1
5 10 15Thr Ala Arg Ile Thr Cys Gly Gly Asn Asn Ile Gly Ser Lys Ser
Val 20 25 30His Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro Val Leu Val
Val Tyr 35 40 45Asp Asp Ser Asp Arg Pro Ser Gly Ile Pro Glu Arg Phe
Ser Gly Ser 50 55 60Asn Ser Gly Asn Thr Ala Thr Leu Thr Ile Ser Arg
Val Glu Ala Gly65 70 75 80Asp Glu Ala Asp Tyr Tyr Cys Gln Val Trp
Asp Ser Ser Ser Asp His 85 90 95Trp Val Phe Gly Gly Gly Thr Lys Leu
Thr Val Leu Ser Ser Ala Ser 100 105 110Thr Lys Gly Pro Ser Val Phe
Pro Leu Ala Pro Ser Ser Lys Ser Thr 115 120 125Ser Gly Gly Thr Ala
Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe Pro 130 135 140Glu Pro Val
Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser Gly Val145 150 155
160His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser
165 170 175Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr
Tyr Ile 180 185 190Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val
Asp Lys Lys Val 195 200 205Glu Pro Lys Ser Cys
21029450PRTArtificialHeavy chain 1 of <VEGF-ANG-2> CrossMAb
IgG4 with AAA mutations and with SPLE mutations 29Glu Val Gln Leu
Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg
Leu Ser Cys Ala Ala Ser Gly Tyr Asp Phe Thr His Tyr 20 25 30Gly Met
Asn Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40 45Gly
Trp Ile Asn Thr Tyr Thr Gly Glu Pro Thr Tyr Ala Ala Asp Phe 50 55
60Lys Arg Arg Phe Thr Phe Ser Leu Asp Thr Ser Lys Ser Thr Ala Tyr65
70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Ala Lys Tyr Pro Tyr Tyr Tyr Gly Thr Ser His Trp Tyr Phe
Asp Val 100 105 110Trp Gly Gln Gly Thr Leu Val Thr Val Ser Ser Ala
Ser Thr Lys Gly 115 120 125Pro Ser Val Phe Pro Leu Ala Pro Cys Ser
Arg Ser Thr Ser Glu Ser 130 135 140Thr Ala Ala Leu Gly Cys Leu Val
Lys Asp Tyr Phe Pro Glu Pro Val145 150 155 160Thr Val Ser Trp Asn
Ser Gly Ala Leu Thr Ser Gly Val His Thr Phe 165 170 175Pro Ala Val
Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser Val Val 180 185 190Thr
Val Pro Ser Ser Ser Leu Gly Thr Lys Thr Tyr Thr Cys Asn Val 195 200
205Asp His Lys Pro Ser Asn Thr Lys Val Asp Lys Arg Val Glu Ser Lys
210 215 220Tyr Gly Pro Pro Cys Pro Pro Cys Pro Ala Pro Glu Phe Glu
Gly Gly225 230 235 240Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys
Asp Thr Leu Met Ala 245 250 255Ser Arg Thr Pro Glu Val Thr Cys Val
Val Val Asp Val Ser Gln Glu 260 265 270Asp Pro Glu Val Gln Phe Asn
Trp Tyr Val Asp Gly Val Glu Val His 275 280 285Asn Ala Lys Thr Lys
Pro Arg Glu Glu Gln Phe Asn Ser Thr Tyr Arg 290 295 300Val Val Ser
Val Leu Thr Val Leu Ala Gln Asp Trp Leu Asn Gly Lys305 310 315
320Glu Tyr Lys Cys Lys Val Ser Asn Lys Gly Leu Pro Ser Ser Ile Glu
325 330 335Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln
Val Cys 340 345 350Thr Leu Pro Pro Ser Gln Glu Glu Met Thr Lys Asn
Gln Val Ser Leu 355 360 365Ser Cys Ala Val Lys Gly Phe Tyr Pro Ser
Asp Ile Ala Val Glu Trp 370 375 380Glu Ser Asn Gly Gln Pro Glu Asn
Asn Tyr Lys Thr Thr Pro Pro Val385 390 395 400Leu Asp Ser Asp Gly
Ser Phe Phe Leu Val Ser Arg Leu Thr Val Asp 405 410 415Lys Ser Arg
Trp Gln Glu Gly Asn Val Phe Ser Cys Ser Val Met His 420 425 430Glu
Ala Leu His Asn Ala Tyr Thr Gln Lys Ser Leu Ser Leu Ser Leu 435 440
445Gly Lys 45030460PRTArtificialHeavy chain 2 of <VEGF-ANG-2>
CrossMAb IgG4 with AAA mutations and with SPLE mutations 30Gln Val
Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser
Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Gly Tyr 20 25
30Tyr Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met
35 40 45Gly Trp Ile Asn Pro Asn Ser Gly Gly Thr Asn Tyr Ala Gln Lys
Phe 50 55 60Gln Gly Arg Val Thr Met Thr Arg Asp Thr Ser Ile Ser Thr
Ala Tyr65 70 75 80Met Glu Leu Ser Arg Leu Arg Ser Asp Asp Thr Ala
Val Tyr Tyr Cys 85 90 95Ala Arg Ser Pro Asn Pro Tyr Tyr Tyr Asp Ser
Ser Gly Tyr Tyr Tyr 100 105 110Pro Gly Ala Phe Asp Ile Trp Gly Gln
Gly Thr Met Val Thr Val Ser 115 120 125Ser Ala Ser Val Ala Ala Pro
Ser Val Phe Ile Phe Pro Pro Ser Asp 130 135 140Glu Gln Leu Lys Ser
Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn145 150 155 160Phe Tyr
Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu 165 170
175Gln Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp
180 185 190Ser Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala
Asp Tyr 195 200 205Glu Lys His Lys Val Tyr Ala Cys Glu Val Thr His
Gln Gly Leu Ser 210 215 220Ser Pro Val Thr Lys Ser Phe Asn Arg Gly
Glu Cys Pro Pro Cys Pro225 230 235 240Pro Cys Pro Ala Pro Glu Phe
Glu Gly Gly Pro Ser Val Phe Leu Phe 245 250 255Pro Pro Lys Pro Lys
Asp Thr Leu Met Ala Ser Arg Thr Pro Glu Val 260 265 270Thr Cys Val
Val Val Asp Val Ser Gln Glu Asp Pro Glu Val Gln Phe 275 280 285Asn
Trp Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro 290 295
300Arg Glu Glu Gln Phe Asn Ser Thr Tyr Arg Val Val Ser Val Leu
Thr305 310 315 320Val Leu Ala Gln Asp Trp Leu Asn Gly Lys Glu Tyr
Lys Cys Lys Val 325 330 335Ser Asn Lys Gly Leu Pro Ser Ser Ile Glu
Lys Thr Ile Ser Lys Ala 340 345 350Lys Gly Gln Pro Arg Glu Pro Gln
Val Tyr Thr Leu Pro Pro Cys Gln 355 360 365Glu Glu Met Thr Lys Asn
Gln Val Ser Leu Trp Cys Leu Val Lys Gly 370 375 380Phe Tyr Pro Ser
Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro385 390 395 400Glu
Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser 405 410
415Phe Phe Leu Tyr Ser Arg Leu Thr Val Asp Lys Ser Arg Trp Gln Glu
420 425 430Gly Asn Val Phe Ser Cys Ser Val Met His Glu Ala Leu His
Asn Ala 435 440 445Tyr Thr Gln Lys Ser Leu Ser Leu Ser Leu Gly Lys
450 455 46031214PRTArtificialLight chain 1 of <VEGF-ANG-2>
CrossMAb IgG4 with AAA mutations and with SPLE mutations 31Asp Ile
Gln Leu Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Ile Thr Cys Ser Ala Ser Gln Asp Ile Ser Asn Tyr 20 25
30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Val Leu Ile
35 40 45Tyr Phe Thr Ser Ser Leu His Ser Gly Val Pro Ser Arg Phe Ser
Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Ser
Thr Val Pro Trp 85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
Arg Thr Val Ala Ala 100 105 110Pro Ser Val Phe Ile Phe Pro Pro Ser
Asp Glu Gln Leu Lys Ser Gly 115 120 125Thr Ala Ser Val Val Cys Leu
Leu Asn Asn Phe Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln Trp Lys
Val Asp Asn Ala Leu Gln Ser Gly Asn Ser Gln145 150 155 160Glu Ser
Val Thr Glu Gln Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser 165 170
175Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr
180 185 190Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser Pro Val Thr
Lys Ser 195 200 205Phe Asn Arg Gly Glu Cys
21032213PRTArtificialLight chain 2 of <VEGF-ANG-2> CrossMAb
IgG4 with AAA mutations and with SPLE mutations 32Ser Tyr Val Leu
Thr Gln Pro Pro Ser Val Ser Val Ala Pro Gly Gln1 5 10 15Thr Ala Arg
Ile Thr Cys Gly Gly Asn Asn Ile Gly Ser Lys Ser Val 20 25 30His Trp
Tyr Gln Gln Lys Pro Gly Gln Ala Pro Val Leu Val Val Tyr 35 40 45Asp
Asp Ser Asp Arg Pro Ser Gly Ile Pro Glu Arg Phe Ser Gly Ser 50 55
60Asn Ser Gly Asn Thr Ala Thr Leu Thr Ile Ser Arg Val Glu Ala Gly65
70 75 80Asp Glu Ala Asp Tyr Tyr Cys Gln Val Trp Asp Ser Ser Ser Asp
His 85 90 95Trp Val Phe Gly Gly Gly Thr Lys Leu Thr Val Leu Ser Ser
Ala Ser 100 105 110Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Cys
Ser Arg Ser Thr 115 120 125Ser Glu Ser Thr Ala Ala Leu Gly Cys Leu
Val Lys Asp Tyr Phe Pro 130 135 140Glu Pro Val Thr Val Ser Trp Asn
Ser Gly Ala Leu Thr Ser Gly Val145 150 155 160His Thr Phe Pro Ala
Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser 165 170 175Ser Val Val
Thr Val Pro Ser Ser Ser Leu Gly Thr Lys Thr Tyr Thr 180 185 190Cys
Asn Val Asp His Lys Pro Ser Asn Thr Lys Val Asp Lys Arg Val 195 200
205Glu Ser Lys Tyr Gly 21033453PRTArtificialHeavy chain 1 of
<VEGF-ANG-2> OAscFab IgG1 with AAA mutations 33Glu Val Gln
Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu
Arg Leu Ser Cys Ala Ala Ser Gly Tyr Asp Phe Thr His Tyr 20 25 30Gly
Met Asn Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val 35 40
45Gly Trp Ile Asn Thr Tyr Thr Gly Glu Pro Thr Tyr Ala Ala Asp Phe
50 55 60Lys Arg Arg Phe Thr Phe Ser Leu Asp Thr Ser Lys Ser Thr Ala
Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Lys Tyr Pro Tyr Tyr Tyr Gly Thr Ser His Trp
Tyr Phe Asp Val 100 105 110Trp Gly Gln Gly Thr Leu Val Thr Val Ser
Ser Ala Ser Thr Lys Gly
115 120 125Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser
Gly Gly 130 135 140Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe
Pro Glu Pro Val145 150 155 160Thr Val Ser Trp Asn Ser Gly Ala Leu
Thr Ser Gly Val His Thr Phe 165 170 175Pro Ala Val Leu Gln Ser Ser
Gly Leu Tyr Ser Leu Ser Ser Val Val 180 185 190Thr Val Pro Ser Ser
Ser Leu Gly Thr Gln Thr Tyr Ile Cys Asn Val 195 200 205Asn His Lys
Pro Ser Asn Thr Lys Val Asp Lys Lys Val Glu Pro Lys 210 215 220Ser
Cys Asp Lys Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu225 230
235 240Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp
Thr 245 250 255Leu Met Ala Ser Arg Thr Pro Glu Val Thr Cys Val Val
Val Asp Val 260 265 270Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp
Tyr Val Asp Gly Val 275 280 285Glu Val His Asn Ala Lys Thr Lys Pro
Arg Glu Glu Gln Tyr Asn Ser 290 295 300Thr Tyr Arg Val Val Ser Val
Leu Thr Val Leu Ala Gln Asp Trp Leu305 310 315 320Asn Gly Lys Glu
Tyr Lys Cys Lys Val Ser Asn Lys Ala Leu Pro Ala 325 330 335Pro Ile
Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro 340 345
350Gln Val Cys Thr Leu Pro Pro Ser Arg Asp Glu Leu Thr Lys Asn Gln
355 360 365Val Ser Leu Ser Cys Ala Val Lys Gly Phe Tyr Pro Ser Asp
Ile Ala 370 375 380Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn
Tyr Lys Thr Thr385 390 395 400Pro Pro Val Leu Asp Ser Asp Gly Ser
Phe Phe Leu Val Ser Lys Leu 405 410 415Thr Val Asp Lys Ser Arg Trp
Gln Gln Gly Asn Val Phe Ser Cys Ser 420 425 430Val Met His Glu Ala
Leu His Asn Ala Tyr Thr Gln Lys Ser Leu Ser 435 440 445Leu Ser Pro
Gly Lys 45034705PRTArtificialHeavy chain 2 of <VEGF-ANG-2>
OAscFab IgG1 with AAA mutations 34Ser Tyr Val Leu Thr Gln Pro Pro
Ser Val Ser Val Ala Pro Gly Gln1 5 10 15Thr Ala Arg Ile Thr Cys Gly
Gly Asn Asn Ile Gly Ser Lys Ser Val 20 25 30His Trp Tyr Gln Gln Lys
Pro Gly Gln Ala Pro Val Leu Val Val Tyr 35 40 45Asp Asp Ser Asp Arg
Pro Ser Gly Ile Pro Glu Arg Phe Ser Gly Ser 50 55 60Asn Ser Gly Asn
Thr Ala Thr Leu Thr Ile Ser Arg Val Glu Ala Gly65 70 75 80Asp Glu
Ala Asp Tyr Tyr Cys Gln Val Trp Asp Ser Ser Ser Asp His 85 90 95Trp
Val Phe Gly Gly Gly Thr Lys Leu Thr Val Leu Gly Gln Pro Lys 100 105
110Ala Ala Pro Ser Val Thr Leu Phe Pro Pro Ser Ser Glu Glu Leu Gln
115 120 125Ala Asn Lys Ala Thr Leu Val Cys Leu Ile Ser Asp Phe Tyr
Pro Gly 130 135 140Ala Val Thr Val Ala Trp Lys Ala Asp Ser Ser Pro
Val Lys Ala Gly145 150 155 160Val Glu Thr Thr Thr Pro Ser Lys Gln
Ser Asn Asn Lys Tyr Ala Ala 165 170 175Ser Ser Tyr Leu Ser Leu Thr
Pro Glu Gln Trp Lys Ser His Arg Ser 180 185 190Tyr Ser Cys Gln Val
Thr His Glu Gly Ser Thr Val Glu Lys Thr Val 195 200 205Ala Pro Thr
Glu Cys Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser 210 215 220Gly
Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly225 230
235 240Gly Gly Gly Ser Gly Gly Gln Val Gln Leu Val Glu Ser Gly Ala
Glu 245 250 255Val Lys Lys Pro Gly Ala Ser Val Lys Val Ser Cys Lys
Ala Ser Gly 260 265 270Tyr Thr Phe Thr Gly Tyr Tyr Met His Trp Val
Arg Gln Ala Pro Gly 275 280 285Gln Gly Leu Glu Trp Met Gly Trp Ile
Asn Pro Asn Ser Gly Gly Thr 290 295 300Asn Tyr Ala Gln Lys Phe Gln
Gly Arg Val Thr Met Thr Arg Asp Thr305 310 315 320Ser Ile Ser Thr
Ala Tyr Met Glu Leu Ser Arg Leu Arg Ser Asp Asp 325 330 335Thr Ala
Val Tyr Tyr Cys Ala Arg Ser Pro Asn Pro Tyr Tyr Tyr Asp 340 345
350Ser Ser Gly Tyr Tyr Tyr Pro Gly Ala Phe Asp Ile Trp Gly Gln Gly
355 360 365Thr Met Val Thr Val Ser Ser Ala Ser Thr Lys Gly Pro Ser
Val Phe 370 375 380Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly Gly
Thr Ala Ala Leu385 390 395 400Gly Cys Leu Val Lys Asp Tyr Phe Pro
Glu Pro Val Thr Val Ser Trp 405 410 415Asn Ser Gly Ala Leu Thr Ser
Gly Val His Thr Phe Pro Ala Val Leu 420 425 430Gln Ser Ser Gly Leu
Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser 435 440 445Ser Ser Leu
Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His Lys Pro 450 455 460Ser
Asn Thr Lys Val Asp Lys Lys Val Glu Pro Lys Ser Cys Asp Lys465 470
475 480Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu Leu Gly Gly
Pro 485 490 495Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr Leu
Met Ala Ser 500 505 510Arg Thr Pro Glu Val Thr Cys Val Val Val Asp
Val Ser His Glu Asp 515 520 525Pro Glu Val Lys Phe Asn Trp Tyr Val
Asp Gly Val Glu Val His Asn 530 535 540Ala Lys Thr Lys Pro Arg Glu
Glu Gln Tyr Asn Ser Thr Tyr Arg Val545 550 555 560Val Ser Val Leu
Thr Val Leu Ala Gln Asp Trp Leu Asn Gly Lys Glu 565 570 575Tyr Lys
Cys Lys Val Ser Asn Lys Ala Leu Pro Ala Pro Ile Glu Lys 580 585
590Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val Tyr Thr
595 600 605Leu Pro Pro Cys Arg Asp Glu Leu Thr Lys Asn Gln Val Ser
Leu Trp 610 615 620Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala
Val Glu Trp Glu625 630 635 640Ser Asn Gly Gln Pro Glu Asn Asn Tyr
Lys Thr Thr Pro Pro Val Leu 645 650 655Asp Ser Asp Gly Ser Phe Phe
Leu Tyr Ser Lys Leu Thr Val Asp Lys 660 665 670Ser Arg Trp Gln Gln
Gly Asn Val Phe Ser Cys Ser Val Met His Glu 675 680 685Ala Leu His
Asn Ala Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly 690 695
700Lys70535214PRTArtificialLight chain 1 of <VEGF-ANG-2>
OAscFab IgG1 with AAA mutations 35Asp Ile Gln Leu Thr Gln Ser Pro
Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys
Ser Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln
Lys Pro Gly Lys Ala Pro Lys Val Leu Ile 35 40 45Tyr Phe Thr Ser Ser
Leu His Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly
Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp
Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Ser Thr Val Pro Trp 85 90 95Thr
Phe Gly Gln Gly Thr Lys Val Glu Ile Lys Arg Thr Val Ala Ala 100 105
110Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu Lys Ser Gly
115 120 125Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe Tyr Pro Arg
Glu Ala 130 135 140Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln Ser
Gly Asn Ser Gln145 150 155 160Glu Ser Val Thr Glu Gln Asp Ser Lys
Asp Ser Thr Tyr Ser Leu Ser 165 170 175Ser Thr Leu Thr Leu Ser Lys
Ala Asp Tyr Glu Lys His Lys Val Tyr 180 185 190Ala Cys Glu Val Thr
His Gln Gly Leu Ser Ser Pro Val Thr Lys Ser 195 200 205Phe Asn Arg
Gly Glu Cys 21036450PRTArtificialHeavy chain 1 of
<VEGF-ANG-2> OAscFab IgG4 with AAA mutations and with SPLE
mutations 36Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro
Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Tyr Asp Phe
Thr His Tyr 20 25 30Gly Met Asn Trp Val Arg Gln Ala Pro Gly Lys Gly
Leu Glu Trp Val 35 40 45Gly Trp Ile Asn Thr Tyr Thr Gly Glu Pro Thr
Tyr Ala Ala Asp Phe 50 55 60Lys Arg Arg Phe Thr Phe Ser Leu Asp Thr
Ser Lys Ser Thr Ala Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Lys Tyr Pro Tyr Tyr Tyr
Gly Thr Ser His Trp Tyr Phe Asp Val 100 105 110Trp Gly Gln Gly Thr
Leu Val Thr Val Ser Ser Ala Ser Thr Lys Gly 115 120 125Pro Ser Val
Phe Pro Leu Ala Pro Cys Ser Arg Ser Thr Ser Glu Ser 130 135 140Thr
Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu Pro Val145 150
155 160Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser Gly Val His Thr
Phe 165 170 175Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser
Ser Val Val 180 185 190Thr Val Pro Ser Ser Ser Leu Gly Thr Lys Thr
Tyr Thr Cys Asn Val 195 200 205Asp His Lys Pro Ser Asn Thr Lys Val
Asp Lys Arg Val Glu Ser Lys 210 215 220Tyr Gly Pro Pro Cys Pro Pro
Cys Pro Ala Pro Glu Phe Glu Gly Gly225 230 235 240Pro Ser Val Phe
Leu Phe Pro Pro Lys Pro Lys Asp Thr Leu Met Ala 245 250 255Ser Arg
Thr Pro Glu Val Thr Cys Val Val Val Asp Val Ser Gln Glu 260 265
270Asp Pro Glu Val Gln Phe Asn Trp Tyr Val Asp Gly Val Glu Val His
275 280 285Asn Ala Lys Thr Lys Pro Arg Glu Glu Gln Phe Asn Ser Thr
Tyr Arg 290 295 300Val Val Ser Val Leu Thr Val Leu Ala Gln Asp Trp
Leu Asn Gly Lys305 310 315 320Glu Tyr Lys Cys Lys Val Ser Asn Lys
Gly Leu Pro Ser Ser Ile Glu 325 330 335Lys Thr Ile Ser Lys Ala Lys
Gly Gln Pro Arg Glu Pro Gln Val Cys 340 345 350Thr Leu Pro Pro Ser
Gln Glu Glu Met Thr Lys Asn Gln Val Ser Leu 355 360 365Ser Cys Ala
Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala Val Glu Trp 370 375 380Glu
Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro Pro Val385 390
395 400Leu Asp Ser Asp Gly Ser Phe Phe Leu Val Ser Arg Leu Thr Val
Asp 405 410 415Lys Ser Arg Trp Gln Glu Gly Asn Val Phe Ser Cys Ser
Val Met His 420 425 430Glu Ala Leu His Asn Ala Tyr Thr Gln Lys Ser
Leu Ser Leu Ser Leu 435 440 445Gly Lys 45037702PRTArtificialHeavy
chain 2 of <VEGF-ANG-2> OAscFab IgG4 with AAA mutations and
with SPLE mutations 37Ser Tyr Val Leu Thr Gln Pro Pro Ser Val Ser
Val Ala Pro Gly Gln1 5 10 15Thr Ala Arg Ile Thr Cys Gly Gly Asn Asn
Ile Gly Ser Lys Ser Val 20 25 30His Trp Tyr Gln Gln Lys Pro Gly Gln
Ala Pro Val Leu Val Val Tyr 35 40 45Asp Asp Ser Asp Arg Pro Ser Gly
Ile Pro Glu Arg Phe Ser Gly Ser 50 55 60Asn Ser Gly Asn Thr Ala Thr
Leu Thr Ile Ser Arg Val Glu Ala Gly65 70 75 80Asp Glu Ala Asp Tyr
Tyr Cys Gln Val Trp Asp Ser Ser Ser Asp His 85 90 95Trp Val Phe Gly
Gly Gly Thr Lys Leu Thr Val Leu Gly Gln Pro Lys 100 105 110Ala Ala
Pro Ser Val Thr Leu Phe Pro Pro Ser Ser Glu Glu Leu Gln 115 120
125Ala Asn Lys Ala Thr Leu Val Cys Leu Ile Ser Asp Phe Tyr Pro Gly
130 135 140Ala Val Thr Val Ala Trp Lys Ala Asp Ser Ser Pro Val Lys
Ala Gly145 150 155 160Val Glu Thr Thr Thr Pro Ser Lys Gln Ser Asn
Asn Lys Tyr Ala Ala 165 170 175Ser Ser Tyr Leu Ser Leu Thr Pro Glu
Gln Trp Lys Ser His Arg Ser 180 185 190Tyr Ser Cys Gln Val Thr His
Glu Gly Ser Thr Val Glu Lys Thr Val 195 200 205Ala Pro Thr Glu Cys
Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser 210 215 220Gly Gly Gly
Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly225 230 235
240Gly Gly Gly Ser Gly Gly Gln Val Gln Leu Val Glu Ser Gly Ala Glu
245 250 255Val Lys Lys Pro Gly Ala Ser Val Lys Val Ser Cys Lys Ala
Ser Gly 260 265 270Tyr Thr Phe Thr Gly Tyr Tyr Met His Trp Val Arg
Gln Ala Pro Gly 275 280 285Gln Gly Leu Glu Trp Met Gly Trp Ile Asn
Pro Asn Ser Gly Gly Thr 290 295 300Asn Tyr Ala Gln Lys Phe Gln Gly
Arg Val Thr Met Thr Arg Asp Thr305 310 315 320Ser Ile Ser Thr Ala
Tyr Met Glu Leu Ser Arg Leu Arg Ser Asp Asp 325 330 335Thr Ala Val
Tyr Tyr Cys Ala Arg Ser Pro Asn Pro Tyr Tyr Tyr Asp 340 345 350Ser
Ser Gly Tyr Tyr Tyr Pro Gly Ala Phe Asp Ile Trp Gly Gln Gly 355 360
365Thr Met Val Thr Val Ser Ser Ala Ser Thr Lys Gly Pro Ser Val Phe
370 375 380Pro Leu Ala Pro Cys Ser Arg Ser Thr Ser Glu Ser Thr Ala
Ala Leu385 390 395 400Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu Pro
Val Thr Val Ser Trp 405 410 415Asn Ser Gly Ala Leu Thr Ser Gly Val
His Thr Phe Pro Ala Val Leu 420 425 430Gln Ser Ser Gly Leu Tyr Ser
Leu Ser Ser Val Val Thr Val Pro Ser 435 440 445Ser Ser Leu Gly Thr
Lys Thr Tyr Thr Cys Asn Val Asp His Lys Pro 450 455 460Ser Asn Thr
Lys Val Asp Lys Arg Val Glu Ser Lys Tyr Gly Pro Pro465 470 475
480Cys Pro Pro Cys Pro Ala Pro Glu Phe Glu Gly Gly Pro Ser Val Phe
485 490 495Leu Phe Pro Pro Lys Pro Lys Asp Thr Leu Met Ala Ser Arg
Thr Pro 500 505 510Glu Val Thr Cys Val Val Val Asp Val Ser Gln Glu
Asp Pro Glu Val 515 520 525Gln Phe Asn Trp Tyr Val Asp Gly Val Glu
Val His Asn Ala Lys Thr 530 535 540Lys Pro Arg Glu Glu Gln Phe Asn
Ser Thr Tyr Arg Val Val Ser Val545 550 555 560Leu Thr Val Leu Ala
Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys 565 570 575Lys Val Ser
Asn Lys Gly Leu Pro Ser Ser Ile Glu Lys Thr Ile Ser 580 585 590Lys
Ala Lys Gly Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro 595 600
605Cys Gln Glu Glu Met Thr Lys Asn Gln Val Ser Leu Trp Cys Leu Val
610 615 620Lys Gly Phe Tyr Pro Ser Asp Ile Ala Val Glu Trp Glu Ser
Asn Gly625 630 635 640Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro Pro
Val Leu Asp Ser Asp 645 650 655Gly Ser Phe Phe Leu Tyr Ser Arg Leu
Thr Val Asp Lys Ser Arg Trp 660 665 670Gln Glu Gly Asn Val Phe Ser
Cys Ser Val Met His Glu Ala Leu His 675 680 685Asn Ala Tyr Thr Gln
Lys Ser Leu Ser Leu Ser Leu Gly Lys 690 695
70038214PRTArtificialLight chain 1 of <VEGF-ANG-2> OAscFab
IgG4 with AAA mutations and with SPLE mutations 38Asp Ile Gln Leu
Thr Gln
Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile
Thr Cys Ser Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr
Gln Gln Lys Pro Gly Lys Ala Pro Lys Val Leu Ile 35 40 45Tyr Phe Thr
Ser Ser Leu His Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly
Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75
80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Ser Thr Val Pro Trp
85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys Arg Thr Val Ala
Ala 100 105 110Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu
Lys Ser Gly 115 120 125Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe
Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln Trp Lys Val Asp Asn Ala
Leu Gln Ser Gly Asn Ser Gln145 150 155 160Glu Ser Val Thr Glu Gln
Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser 165 170 175Ser Thr Leu Thr
Leu Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr 180 185 190Ala Cys
Glu Val Thr His Gln Gly Leu Ser Ser Pro Val Thr Lys Ser 195 200
205Phe Asn Arg Gly Glu Cys 21039453PRTArtificialHeavy chain 1 of
<VEGF-ANG-2> CrossMAb IgG1 wild type (without AAA mutations)
(VEGFang2-0201) 39Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val
Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Tyr
Asp Phe Thr His Tyr 20 25 30Gly Met Asn Trp Val Arg Gln Ala Pro Gly
Lys Gly Leu Glu Trp Val 35 40 45Gly Trp Ile Asn Thr Tyr Thr Gly Glu
Pro Thr Tyr Ala Ala Asp Phe 50 55 60Lys Arg Arg Phe Thr Phe Ser Leu
Asp Thr Ser Lys Ser Thr Ala Tyr65 70 75 80Leu Gln Met Asn Ser Leu
Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Lys Tyr Pro Tyr
Tyr Tyr Gly Thr Ser His Trp Tyr Phe Asp Val 100 105 110Trp Gly Gln
Gly Thr Leu Val Thr Val Ser Ser Ala Ser Thr Lys Gly 115 120 125Pro
Ser Val Phe Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly Gly 130 135
140Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr Phe Pro Glu Pro
Val145 150 155 160Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser Gly
Val His Thr Phe 165 170 175Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr
Ser Leu Ser Ser Val Val 180 185 190Thr Val Pro Ser Ser Ser Leu Gly
Thr Gln Thr Tyr Ile Cys Asn Val 195 200 205Asn His Lys Pro Ser Asn
Thr Lys Val Asp Lys Lys Val Glu Pro Lys 210 215 220Ser Cys Asp Lys
Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu225 230 235 240Leu
Gly Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr 245 250
255Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys Val Val Val Asp Val
260 265 270Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp Tyr Val Asp
Gly Val 275 280 285Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu Glu
Gln Tyr Asn Ser 290 295 300Thr Tyr Arg Val Val Ser Val Leu Thr Val
Leu His Gln Asp Trp Leu305 310 315 320Asn Gly Lys Glu Tyr Lys Cys
Lys Val Ser Asn Lys Ala Leu Pro Ala 325 330 335Pro Ile Glu Lys Thr
Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro 340 345 350Gln Val Tyr
Thr Leu Pro Pro Cys Arg Asp Glu Leu Thr Lys Asn Gln 355 360 365Val
Ser Leu Trp Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala 370 375
380Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys Thr
Thr385 390 395 400Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe Leu
Tyr Ser Lys Leu 405 410 415Thr Val Asp Lys Ser Arg Trp Gln Gln Gly
Asn Val Phe Ser Cys Ser 420 425 430Val Met His Glu Ala Leu His Asn
His Tyr Thr Gln Lys Ser Leu Ser 435 440 445Leu Ser Pro Gly Lys
45040463PRTArtificialHeavy chain 2 of <VEGF-ANG-2> CrossMAb
IgG1 wild type (without AAA mutations) (VEGFang2-0201) 40Gln Val
Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser
Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Gly Tyr 20 25
30Tyr Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met
35 40 45Gly Trp Ile Asn Pro Asn Ser Gly Gly Thr Asn Tyr Ala Gln Lys
Phe 50 55 60Gln Gly Arg Val Thr Met Thr Arg Asp Thr Ser Ile Ser Thr
Ala Tyr65 70 75 80Met Glu Leu Ser Arg Leu Arg Ser Asp Asp Thr Ala
Val Tyr Tyr Cys 85 90 95Ala Arg Ser Pro Asn Pro Tyr Tyr Tyr Asp Ser
Ser Gly Tyr Tyr Tyr 100 105 110Pro Gly Ala Phe Asp Ile Trp Gly Gln
Gly Thr Met Val Thr Val Ser 115 120 125Ser Ala Ser Val Ala Ala Pro
Ser Val Phe Ile Phe Pro Pro Ser Asp 130 135 140Glu Gln Leu Lys Ser
Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn145 150 155 160Phe Tyr
Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu 165 170
175Gln Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp
180 185 190Ser Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala
Asp Tyr 195 200 205Glu Lys His Lys Val Tyr Ala Cys Glu Val Thr His
Gln Gly Leu Ser 210 215 220Ser Pro Val Thr Lys Ser Phe Asn Arg Gly
Glu Cys Asp Lys Thr His225 230 235 240Thr Cys Pro Pro Cys Pro Ala
Pro Glu Leu Leu Gly Gly Pro Ser Val 245 250 255Phe Leu Phe Pro Pro
Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr 260 265 270Pro Glu Val
Thr Cys Val Val Val Asp Val Ser His Glu Asp Pro Glu 275 280 285Val
Lys Phe Asn Trp Tyr Val Asp Gly Val Glu Val His Asn Ala Lys 290 295
300Thr Lys Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val
Ser305 310 315 320Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly
Lys Glu Tyr Lys 325 330 335Cys Lys Val Ser Asn Lys Ala Leu Pro Ala
Pro Ile Glu Lys Thr Ile 340 345 350Ser Lys Ala Lys Gly Gln Pro Arg
Glu Pro Gln Val Cys Thr Leu Pro 355 360 365Pro Ser Arg Asp Glu Leu
Thr Lys Asn Gln Val Ser Leu Ser Cys Ala 370 375 380Val Lys Gly Phe
Tyr Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn385 390 395 400Gly
Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser 405 410
415Asp Gly Ser Phe Phe Leu Val Ser Lys Leu Thr Val Asp Lys Ser Arg
420 425 430Trp Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met His Glu
Ala Leu 435 440 445His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser
Pro Gly Lys 450 455 46041214PRTArtificialLight chain 1 of
<VEGF-ANG-2> CrossMAb IgG1 wild type ( without AAA mutations)
(VEGFang2-0201) 41Asp Ile Gln Leu Thr Gln Ser Pro Ser Ser Leu Ser
Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Ser Ala Ser Gln
Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro Gly Lys
Ala Pro Lys Val Leu Ile 35 40 45Tyr Phe Thr Ser Ser Leu His Ser Gly
Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp Phe Thr
Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala Thr Tyr
Tyr Cys Gln Gln Tyr Ser Thr Val Pro Trp 85 90 95Thr Phe Gly Gln Gly
Thr Lys Val Glu Ile Lys Arg Thr Val Ala Ala 100 105 110Pro Ser Val
Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu Lys Ser Gly 115 120 125Thr
Ala Ser Val Val Cys Leu Leu Asn Asn Phe Tyr Pro Arg Glu Ala 130 135
140Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln Ser Gly Asn Ser
Gln145 150 155 160Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr
Tyr Ser Leu Ser 165 170 175Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr
Glu Lys His Lys Val Tyr 180 185 190Ala Cys Glu Val Thr His Gln Gly
Leu Ser Ser Pro Val Thr Lys Ser 195 200 205Phe Asn Arg Gly Glu Cys
21042213PRTArtificialLight chain 2 of <VEGF-ANG-2> CrossMAb
IgG1 wild type (without AAA mutations) (VEGFang2-0201) 42Ser Tyr
Val Leu Thr Gln Pro Pro Ser Val Ser Val Ala Pro Gly Gln1 5 10 15Thr
Ala Arg Ile Thr Cys Gly Gly Asn Asn Ile Gly Ser Lys Ser Val 20 25
30His Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro Val Leu Val Val Tyr
35 40 45Asp Asp Ser Asp Arg Pro Ser Gly Ile Pro Glu Arg Phe Ser Gly
Ser 50 55 60Asn Ser Gly Asn Thr Ala Thr Leu Thr Ile Ser Arg Val Glu
Ala Gly65 70 75 80Asp Glu Ala Asp Tyr Tyr Cys Gln Val Trp Asp Ser
Ser Ser Asp His 85 90 95Trp Val Phe Gly Gly Gly Thr Lys Leu Thr Val
Leu Ser Ser Ala Ser 100 105 110Thr Lys Gly Pro Ser Val Phe Pro Leu
Ala Pro Ser Ser Lys Ser Thr 115 120 125Ser Gly Gly Thr Ala Ala Leu
Gly Cys Leu Val Lys Asp Tyr Phe Pro 130 135 140Glu Pro Val Thr Val
Ser Trp Asn Ser Gly Ala Leu Thr Ser Gly Val145 150 155 160His Thr
Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser 165 170
175Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr Tyr Ile
180 185 190Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys
Lys Val 195 200 205Glu Pro Lys Ser Cys 21043453PRTArtificialHeavy
chain 1 of <VEGF-ANG-2> CrossMAb IgG1 with P329G LALA
mutations only (without AAA mutations) (VEGFang2-0015) 43Glu Val
Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser
Leu Arg Leu Ser Cys Ala Ala Ser Gly Tyr Asp Phe Thr His Tyr 20 25
30Gly Met Asn Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val
35 40 45Gly Trp Ile Asn Thr Tyr Thr Gly Glu Pro Thr Tyr Ala Ala Asp
Phe 50 55 60Lys Arg Arg Phe Thr Phe Ser Leu Asp Thr Ser Lys Ser Thr
Ala Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala
Val Tyr Tyr Cys 85 90 95Ala Lys Tyr Pro Tyr Tyr Tyr Gly Thr Ser His
Trp Tyr Phe Asp Val 100 105 110Trp Gly Gln Gly Thr Leu Val Thr Val
Ser Ser Ala Ser Thr Lys Gly 115 120 125Pro Ser Val Phe Pro Leu Ala
Pro Ser Ser Lys Ser Thr Ser Gly Gly 130 135 140Thr Ala Ala Leu Gly
Cys Leu Val Lys Asp Tyr Phe Pro Glu Pro Val145 150 155 160Thr Val
Ser Trp Asn Ser Gly Ala Leu Thr Ser Gly Val His Thr Phe 165 170
175Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser Val Val
180 185 190Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr Tyr Ile Cys
Asn Val 195 200 205Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys Lys
Val Glu Pro Lys 210 215 220Ser Cys Asp Lys Thr His Thr Cys Pro Pro
Cys Pro Ala Pro Glu Ala225 230 235 240Ala Gly Gly Pro Ser Val Phe
Leu Phe Pro Pro Lys Pro Lys Asp Thr 245 250 255Leu Met Ile Ser Arg
Thr Pro Glu Val Thr Cys Val Val Val Asp Val 260 265 270Ser His Glu
Asp Pro Glu Val Lys Phe Asn Trp Tyr Val Asp Gly Val 275 280 285Glu
Val His Asn Ala Lys Thr Lys Pro Arg Glu Glu Gln Tyr Asn Ser 290 295
300Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu His Gln Asp Trp
Leu305 310 315 320Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn Lys
Ala Leu Gly Ala 325 330 335Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys
Gly Gln Pro Arg Glu Pro 340 345 350Gln Val Tyr Thr Leu Pro Pro Cys
Arg Asp Glu Leu Thr Lys Asn Gln 355 360 365Val Ser Leu Trp Cys Leu
Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala 370 375 380Val Glu Trp Glu
Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys Thr Thr385 390 395 400Pro
Pro Val Leu Asp Ser Asp Gly Ser Phe Phe Leu Tyr Ser Lys Leu 405 410
415Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn Val Phe Ser Cys Ser
420 425 430Val Met His Glu Ala Leu His Asn His Tyr Thr Gln Lys Ser
Leu Ser 435 440 445Leu Ser Pro Gly Lys 45044463PRTArtificialHeavy
chain 2 of <VEGF-ANG-2> CrossMAb IgG1 with P329G LALA
mutations only (without AAA mutations) (VEGFang2-0015) 44Gln Val
Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser
Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Gly Tyr 20 25
30Tyr Met His Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met
35 40 45Gly Trp Ile Asn Pro Asn Ser Gly Gly Thr Asn Tyr Ala Gln Lys
Phe 50 55 60Gln Gly Arg Val Thr Met Thr Arg Asp Thr Ser Ile Ser Thr
Ala Tyr65 70 75 80Met Glu Leu Ser Arg Leu Arg Ser Asp Asp Thr Ala
Val Tyr Tyr Cys 85 90 95Ala Arg Ser Pro Asn Pro Tyr Tyr Tyr Asp Ser
Ser Gly Tyr Tyr Tyr 100 105 110Pro Gly Ala Phe Asp Ile Trp Gly Gln
Gly Thr Met Val Thr Val Ser 115 120 125Ser Ala Ser Val Ala Ala Pro
Ser Val Phe Ile Phe Pro Pro Ser Asp 130 135 140Glu Gln Leu Lys Ser
Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn145 150 155 160Phe Tyr
Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu 165 170
175Gln Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp
180 185 190Ser Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala
Asp Tyr 195 200 205Glu Lys His Lys Val Tyr Ala Cys Glu Val Thr His
Gln Gly Leu Ser 210 215 220Ser Pro Val Thr Lys Ser Phe Asn Arg Gly
Glu Cys Asp Lys Thr His225 230 235 240Thr Cys Pro Pro Cys Pro Ala
Pro Glu Ala Ala Gly Gly Pro Ser Val 245 250 255Phe Leu Phe Pro Pro
Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr 260 265 270Pro Glu Val
Thr Cys Val Val Val Asp Val Ser His Glu Asp Pro Glu 275 280 285Val
Lys Phe Asn Trp Tyr Val Asp Gly Val Glu Val His Asn Ala Lys 290 295
300Thr Lys Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val
Ser305 310 315 320Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly
Lys Glu Tyr Lys 325 330 335Cys Lys Val Ser Asn Lys Ala Leu Gly Ala
Pro Ile Glu Lys Thr Ile 340 345 350Ser Lys Ala Lys Gly Gln Pro Arg
Glu Pro Gln Val Cys Thr Leu Pro 355 360 365Pro Ser Arg Asp Glu Leu
Thr Lys Asn
Gln Val Ser Leu Ser Cys Ala 370 375 380Val Lys Gly Phe Tyr Pro Ser
Asp Ile Ala Val Glu Trp Glu Ser Asn385 390 395 400Gly Gln Pro Glu
Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser 405 410 415Asp Gly
Ser Phe Phe Leu Val Ser Lys Leu Thr Val Asp Lys Ser Arg 420 425
430Trp Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met His Glu Ala Leu
435 440 445His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly
Lys 450 455 46045214PRTArtificialLight chain 1 of
<VEGF-ANG-2> CrossMAb IgG1 with P329G LALA mutations only
(without AAA mutations) (VEGFang2-0015) 45Asp Ile Gln Leu Thr Gln
Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile
Thr Cys Ser Ala Ser Gln Asp Ile Ser Asn Tyr 20 25 30Leu Asn Trp Tyr
Gln Gln Lys Pro Gly Lys Ala Pro Lys Val Leu Ile 35 40 45Tyr Phe Thr
Ser Ser Leu His Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly
Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75
80Glu Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Ser Thr Val Pro Trp
85 90 95Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys Arg Thr Val Ala
Ala 100 105 110Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu
Lys Ser Gly 115 120 125Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe
Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln Trp Lys Val Asp Asn Ala
Leu Gln Ser Gly Asn Ser Gln145 150 155 160Glu Ser Val Thr Glu Gln
Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser 165 170 175Ser Thr Leu Thr
Leu Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr 180 185 190Ala Cys
Glu Val Thr His Gln Gly Leu Ser Ser Pro Val Thr Lys Ser 195 200
205Phe Asn Arg Gly Glu Cys 21046213PRTArtificialLight chain 2 of
<VEGF-ANG-2> CrossMAb IgG1 with P329G LALA mutations only
(without AAA mutations) (VEGFang2-0015) 46Ser Tyr Val Leu Thr Gln
Pro Pro Ser Val Ser Val Ala Pro Gly Gln1 5 10 15Thr Ala Arg Ile Thr
Cys Gly Gly Asn Asn Ile Gly Ser Lys Ser Val 20 25 30His Trp Tyr Gln
Gln Lys Pro Gly Gln Ala Pro Val Leu Val Val Tyr 35 40 45Asp Asp Ser
Asp Arg Pro Ser Gly Ile Pro Glu Arg Phe Ser Gly Ser 50 55 60Asn Ser
Gly Asn Thr Ala Thr Leu Thr Ile Ser Arg Val Glu Ala Gly65 70 75
80Asp Glu Ala Asp Tyr Tyr Cys Gln Val Trp Asp Ser Ser Ser Asp His
85 90 95Trp Val Phe Gly Gly Gly Thr Lys Leu Thr Val Leu Ser Ser Ala
Ser 100 105 110Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser
Lys Ser Thr 115 120 125Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val
Lys Asp Tyr Phe Pro 130 135 140Glu Pro Val Thr Val Ser Trp Asn Ser
Gly Ala Leu Thr Ser Gly Val145 150 155 160His Thr Phe Pro Ala Val
Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser 165 170 175Ser Val Val Thr
Val Pro Ser Ser Ser Leu Gly Thr Gln Thr Tyr Ile 180 185 190Cys Asn
Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys Lys Val 195 200
205Glu Pro Lys Ser Cys 21047107PRTHomo sapiens 47Arg Thr Val Ala
Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu1 5 10 15Gln Leu Lys
Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe 20 25 30Tyr Pro
Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln 35 40 45Ser
Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser 50 55
60Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu65
70 75 80Lys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser
Ser 85 90 95Pro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys 100
10548105PRThomo sapiens 48Gln Pro Lys Ala Ala Pro Ser Val Thr Leu
Phe Pro Pro Ser Ser Glu1 5 10 15Glu Leu Gln Ala Asn Lys Ala Thr Leu
Val Cys Leu Ile Ser Asp Phe 20 25 30Tyr Pro Gly Ala Val Thr Val Ala
Trp Lys Ala Asp Ser Ser Pro Val 35 40 45Lys Ala Gly Val Glu Thr Thr
Thr Pro Ser Lys Gln Ser Asn Asn Lys 50 55 60Tyr Ala Ala Ser Ser Tyr
Leu Ser Leu Thr Pro Glu Gln Trp Lys Ser65 70 75 80His Arg Ser Tyr
Ser Cys Gln Val Thr His Glu Gly Ser Thr Val Glu 85 90 95Lys Thr Val
Ala Pro Thr Glu Cys Ser 100 10549330PRTHomo sapiens 49Ala Ser Thr
Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser Thr
Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr 20 25 30Phe
Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser 35 40
45Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser
50 55 60Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln
Thr65 70 75 80Tyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys
Val Asp Lys 85 90 95Lys Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr
Cys Pro Pro Cys 100 105 110Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser
Val Phe Leu Phe Pro Pro 115 120 125Lys Pro Lys Asp Thr Leu Met Ile
Ser Arg Thr Pro Glu Val Thr Cys 130 135 140Val Val Val Asp Val Ser
His Glu Asp Pro Glu Val Lys Phe Asn Trp145 150 155 160Tyr Val Asp
Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu 165 170 175Glu
Gln Tyr Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu 180 185
190His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn
195 200 205Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala
Lys Gly 210 215 220Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro
Ser Arg Asp Glu225 230 235 240Leu Thr Lys Asn Gln Val Ser Leu Thr
Cys Leu Val Lys Gly Phe Tyr 245 250 255Pro Ser Asp Ile Ala Val Glu
Trp Glu Ser Asn Gly Gln Pro Glu Asn 260 265 270Asn Tyr Lys Thr Thr
Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe 275 280 285Leu Tyr Ser
Lys Leu Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn 290 295 300Val
Phe Ser Cys Ser Val Met His Glu Ala Leu His Asn His Tyr Thr305 310
315 320Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys 325 33050327PRTHomo
sapiens 50Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Cys
Ser Arg1 5 10 15Ser Thr Ser Glu Ser Thr Ala Ala Leu Gly Cys Leu Val
Lys Asp Tyr 20 25 30Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly
Ala Leu Thr Ser 35 40 45Gly Val His Thr Phe Pro Ala Val Leu Gln Ser
Ser Gly Leu Tyr Ser 50 55 60Leu Ser Ser Val Val Thr Val Pro Ser Ser
Ser Leu Gly Thr Lys Thr65 70 75 80Tyr Thr Cys Asn Val Asp His Lys
Pro Ser Asn Thr Lys Val Asp Lys 85 90 95Arg Val Glu Ser Lys Tyr Gly
Pro Pro Cys Pro Ser Cys Pro Ala Pro 100 105 110Glu Phe Leu Gly Gly
Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys 115 120 125Asp Thr Leu
Met Ile Ser Arg Thr Pro Glu Val Thr Cys Val Val Val 130 135 140Asp
Val Ser Gln Glu Asp Pro Glu Val Gln Phe Asn Trp Tyr Val Asp145 150
155 160Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu Glu Gln
Phe 165 170 175Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu
His Gln Asp 180 185 190Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val
Ser Asn Lys Gly Leu 195 200 205Pro Ser Ser Ile Glu Lys Thr Ile Ser
Lys Ala Lys Gly Gln Pro Arg 210 215 220Glu Pro Gln Val Tyr Thr Leu
Pro Pro Ser Gln Glu Glu Met Thr Lys225 230 235 240Asn Gln Val Ser
Leu Thr Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp 245 250 255Ile Ala
Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys 260 265
270Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe Leu Tyr Ser
275 280 285Arg Leu Thr Val Asp Lys Ser Arg Trp Gln Glu Gly Asn Val
Phe Ser 290 295 300Cys Ser Val Met His Glu Ala Leu His Asn His Tyr
Thr Gln Lys Ser305 310 315 320Leu Ser Leu Ser Leu Gly Lys 325
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012
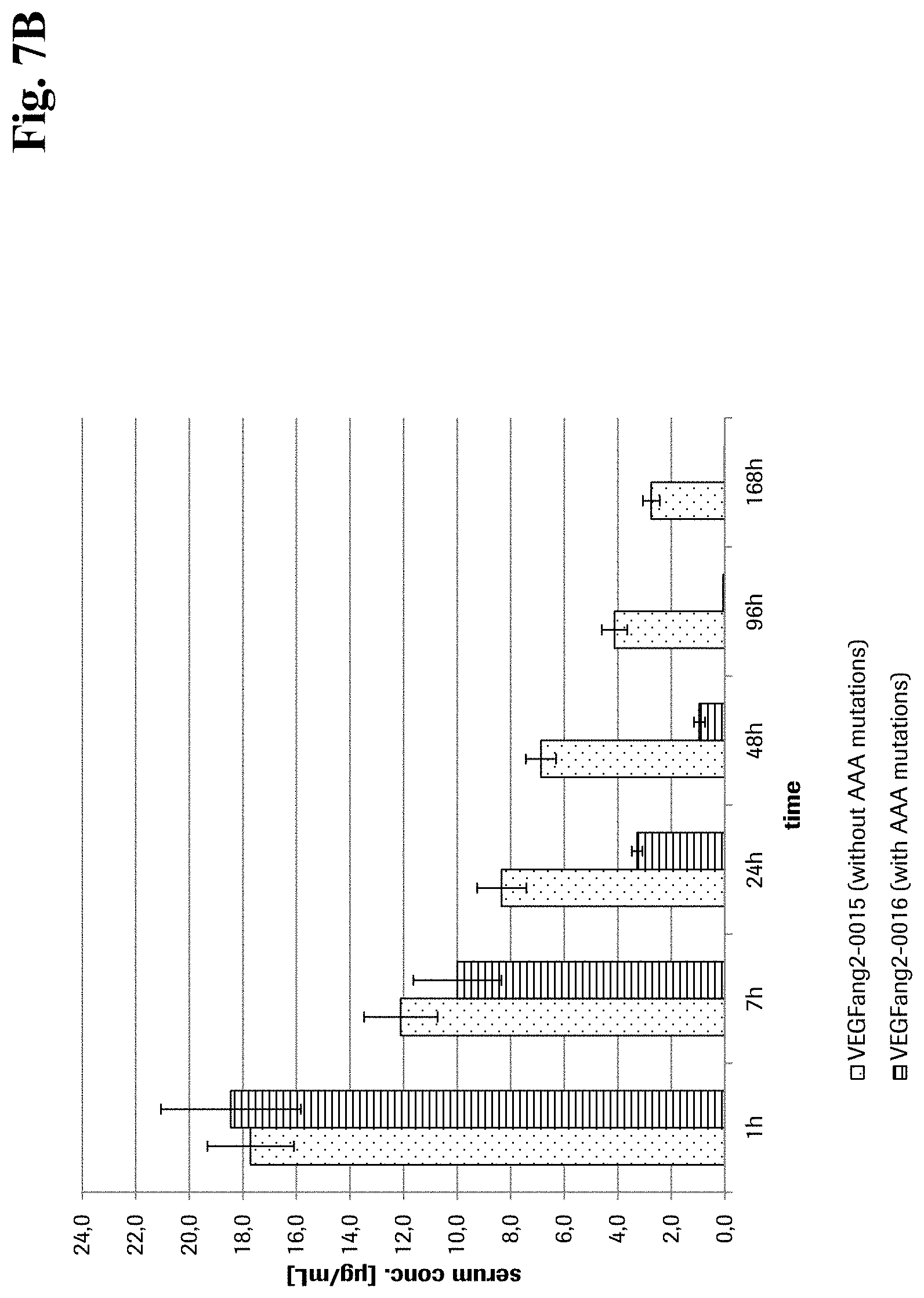
D00013

D00014

D00015


S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.