Methods Of Use Of Oral Care Compositions Comprising Hops
Baig; Arif Ali ; et al.
U.S. patent application number 17/037789 was filed with the patent office on 2021-04-01 for methods of use of oral care compositions comprising hops. The applicant listed for this patent is The Procter & Gamble Company. Invention is credited to Arif Ali Baig, Tammy K. Baker, Aaron Reed Biesbrock, Samuel James St. John.
| Application Number | 20210093551 17/037789 |
| Document ID | / |
| Family ID | 1000005223814 |
| Filed Date | 2021-04-01 |








| United States Patent Application | 20210093551 |
| Kind Code | A1 |
| Baig; Arif Ali ; et al. | April 1, 2021 |
Methods Of Use Of Oral Care Compositions Comprising Hops
Abstract
Methods of use of oral care compositions comprising hops. Methods of use of anticavity oral care compositions that are free of fluoride. Methods of use of anticavity toothpaste comprising hops. Methods of use of anticavity toothpaste comprising hops that are free of fluoride. Methods of treatment, reduction, and/or prevention of caries using an oral care composition comprising hops, and optionally fluoride free.
| Inventors: | Baig; Arif Ali; (Mason, OH) ; Baker; Tammy K.; (Cincinnati, OH) ; Biesbrock; Aaron Reed; (Maineville, OH) ; St. John; Samuel James; (Cincinnati, OH) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005223814 | ||||||||||
| Appl. No.: | 17/037789 | ||||||||||
| Filed: | September 30, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62907733 | Sep 30, 2019 | |||
| 62972109 | Feb 10, 2020 | |||
| 62907735 | Sep 30, 2019 | |||
| 62972111 | Feb 10, 2020 | |||
| 62985451 | Mar 5, 2020 | |||
| 62907736 | Sep 30, 2019 | |||
| 62943940 | Dec 5, 2019 | |||
| 62994893 | Mar 26, 2020 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 8/9789 20170801; A61Q 11/00 20130101 |
| International Class: | A61K 8/9789 20060101 A61K008/9789; A61Q 11/00 20060101 A61Q011/00 |
Claims
1. A method of treating, reducing, or preventing caries comprising: (a) dispensing a toothpaste composition onto a toothbrush, (b) applying the toothpaste composition to the oral cavity for at least 1 minute, and (c) expectorating the toothpaste from the oral cavity, wherein the toothpaste composition comprises hops and abrasive.
2. The method of claim 1, wherein the toothpaste composition comprises anticavity toothpaste.
3. The method of claim 1, wherein the hops comprises hops extract.
4. The method of claim 3, wherein the hops extract comprises hops alpha acid, hops beta acid, or combinations thereof.
5. The method of claim 4, wherein the hops extract comprises less than about 1%, by weight of the extract, of hops alpha acid.
6. The method of claim 1, wherein the abrasive comprises silica abrasive, calcium abrasive, or combinations thereof.
7. The method of claim 6, wherein the calcium abrasive comprises calcium carbonate, calcium pyrophosphate, calcium phosphate, or combinations thereof.
8. The method of claim 1, wherein the toothpaste composition comprises zinc, tin, or combinations thereof.
9. The method of claim 1, wherein the toothpaste is free of fluoride.
10. A method of treating, reducing, or preventing caries comprising: (a) instructing a user to apply a toothpaste composition to a toothbrush, and (b) instructing the user to apply the toothpaste composition to an oral cavity of the user, wherein the toothpaste composition comprises hops and abrasive.
11. The method of claim 10, wherein the method further comprises: (c) instructing the user to not swallow the toothpaste composition.
12. The method of claim 10, wherein toothpaste composition is free of fluoride.
13. The method of claim 10, wherein the toothpaste composition comprises an anticavity toothpaste.
14. The method of claim 10, wherein the hops comprises hops extract.
15. The method of claim 14, wherein the hops extract comprises hops alpha acid, hops beta acid, or combinations thereof.
16. The method of claim 15, wherein the hops extract comprises less than about 1%, by weight of the extract, of hops alpha acid.
17. The method of claim 10, wherein the abrasive comprises silica abrasive, calcium abrasive, or combinations thereof.
18. The method of claim 17, wherein the calcium abrasive comprises calcium carbonate, calcium pyrophosphate, calcium phosphate, or combinations thereof.
19. The method of claim 10, wherein the oral care composition comprises zinc, tin, or combinations thereof.
20. A method of treating, reducing, or preventing caries comprising: (a) applying an oral care composition to an oral cavity, the oral care composition comprising hops, and (b) expectorating the oral care composition from the oral cavity.
Description
FIELD OF THE INVENTION
[0001] The present invention is directed to methods of use of oral care compositions comprising hops. The present invention is directed to methods of use of anticavity oral care compositions that are free of fluoride.
BACKGROUND OF THE INVENTION
[0002] Oral care compositions, such as toothpaste and/or dentifrice compositions, can be applied to the oral cavity to clean and/or maintain the aesthetics and/or health of the teeth, gums, and/or tongue. Additionally, many oral care compositions are used to deliver active ingredients directly to oral care surfaces. For example, toothpaste compositions can have a fluoride ion source, such as sodium fluoride, sodium monofluorophosphate, and/or stannous fluoride, as an anticaries drug. While the effectiveness and safety of fluoride as an anticaries drug is well established, many consumers desire other options for anticavity protection.
[0003] Unfortunately, current fluoride-free oral care compositions do not provide enough, or any, protection from caries due to the acids produced by bacteria found on the surfaces of teeth. As such, there is a need for methods of use of oral care compositions that can be fluoride-free and provide an anticavity benefit.
SUMMARY OF THE INVENTION
[0004] Disclosed herein is a method of treating, reducing, or preventing caries comprising (a) dispensing a toothpaste composition onto a toothbrush, (b) applying the toothpaste composition to the oral cavity for at least 1 minute, and (c) expectorating the toothpaste from the oral cavity, wherein the toothpaste composition comprises hops and abrasive.
[0005] Also disclosed herein is a method of treating, reducing, or preventing caries comprising: (a) instructing a user to apply a toothpaste composition to a toothbrush, and (b) instructing the user to apply the toothpaste composition to an oral cavity of the user, wherein the toothpaste composition comprises hops acid and abrasive.
[0006] Also disclosed herein is a method of treating, reducing, or preventing caries comprising: (a) applying an oral care composition to an oral cavity, the oral care composition comprising hops, and (b) expectorating the oral care composition from the oral cavity.
[0007] Also disclosed herein is hops beta acid for use as medicament, preferably wherein the medicament is an oral care composition.
[0008] Also disclosed herein is hops for use in anticavity treatment.
[0009] Also disclosed herein is the use of hops to reduce white spots in an oral cavity, preferably wherein the white spots in the oral cavity are on teeth and attributable to caries, cavities, and/or combinations thereof.
DETAILED DESCRIPTION OF THE INVENTION
[0010] The present invention is directed to method of use of compositions comprising hops in the treatment of cavities, caries, gingivitis, and/or combinations thereof. The disclosed compositions comprising hops can be used in the treatment, reduction, or prevention of caries.
[0011] Dental caries, or tooth decay, is a breakdown of the teeth due to the acids made by bacteria. Cavities are caused by the acid produced by bacteria dissolving the hard tissues of the teeth, such as enamel, dentin, and/or cementum. The acid is produced by the bacteria when the bacteria breaks down food debris or sugar on the tooth's surface.
[0012] Fluoride works by making the tooth's surface less soluble to the acid produced by the bacteria, "plaque acid." Tooth's enamel is made from hydroxyapatite (Ca.sub.5(PO.sub.4).sub.3(OH)). Hydroxyapatite can be dissolved from the enamel at a pH of under 5.5 (demineralization). If hydroxyapatite is demineralized in the presence of fluoride ions, fluorapatite (Ca.sub.5(PO.sub.4).sub.3(F)) can remineralize on the surface of a tooth's enamel. In sum, this process is a replacement of a hydroxyl (OH) ion with a fluoride (F) ion. Fluorapatite is inherently less soluble than hydroxyapatite, even under acidic conditions. Thus, fluoride works as an anticaries drug to make the tooth's surface more resistant and less soluble to plaque acid.
[0013] While not wishing to being bound by theory, it is believed that the disclosed oral care compositions have a different mechanism of action than fluoride ion therapy. In contrast to the single symptom treatment of fluoride (i.e. treating the result of plaque acid), the disclosed compositions are believed to have anticavity activity through a combination of effects that collectively lead to an anticavity effect. While not wishing to being bound by theory, it is believed that the disclosed oral care compositions have an anticavity effect by providing one or more of the following outcomes: (1) suppressing plaque acid production, (2) neutralizing plaque acids that are produced, and/or (3) reducing the solubility of the hydroxyapatite hard tissue surface.
[0014] The suppression of plaque acid production can be accomplished by providing one or more antibacterial agents to kill the source of the plaque acid the bacteria itself) and/or providing one or more biofilm modifiers to disrupt and embed antibacterial agents within the biofilm matrix.
[0015] Neutralizing the plaque acids can be accomplished by providing one or more biofilm modifiers to disrupt the biofilm matrix and/or providing one or more buffering agents to buffer the produced plaque acid and/or increasing the saturation of calcium ions on the enamel surface by providing one or more calcium ion sources.
[0016] Reducing the solubility of the hydroxyapatite hard tissue surface can be accomplished by providing one or more calcium ion sources. This can create a supersaturated environment of calcium and/or phosphate ions around the hydroxyapatite surface, which can shift the equilibrium of Formula I in favor of remineralization of hydroxyapatite according to Le Chatelier's principle.
Ca.sub.10(PO.sub.4).sub.6(OH).sub.2.revreaction.10Ca.sup.2++6PO.sub.4.su- p.3-
Formula I. Hydroxyapatite
[0017] Reducing the solubility of the hydroxyapatite hard tissue surface can also be accomplished by surface adsorbing metal ions that slow the dissolution of the enamel by forming an acid resistant surface layer. This may be accomplished, for example, by depositing stannous ions on the surface of enamel.
[0018] While each of these mechanisms might not be enough, on its own, to provide an anticavity benefit equivalent to a therapeutic dose of fluoride, in combination these mechanisms can provide an anticavity benefit. As such, the present invention is directed to compositions and methods of use of the compositions that can be fluoride-free, yet still provide an anticavity and/or anticaries benefit.
Definitions
[0019] To define more clearly the terms used herein, the following definitions are provided. Unless otherwise indicated, the following definitions are applicable to this disclosure. If a term is used in this disclosure but is not specifically defined herein, the definition from the IUPAC Compendium of Chemical Terminology, 2nd Ed (1997), can be applied, as long as that definition does not conflict with any other disclosure or definition applied herein, or render indefinite or non-enabled any claim to which that definition is applied.
[0020] The term "oral care composition", as used herein, includes a product, which in the ordinary course of usage, is not intentionally swallowed for purposes of systemic administration of particular therapeutic agents, but is rather retained in the oral cavity for a time sufficient to contact dental surfaces or oral tissues. Examples of oral care compositions include dentifrice, toothpaste, tooth gel, subgingival gel, mouth rinse, mousse, foam, mouth spray, lozenge, chewable tablet, chewing gum, tooth whitening strips, floss and floss coatings, breath freshening dissolvable strips, or denture care or adhesive product. The oral care composition may also be incorporated onto strips or films for direct application or attachment to oral surfaces.
[0021] "Active and other ingredients" useful herein may be categorized or described herein by their cosmetic and/or therapeutic benefit or their postulated mode of action or function. However, it is to be understood that the active and other ingredients useful herein can, in some instances, provide more than one cosmetic and/or therapeutic benefit or function or operate via more than one mode of action. Therefore, classifications herein are made for the sake of convenience and are not intended to limit an ingredient to the particularly stated function(s) or activities listed.
[0022] The term "orally acceptable carrier" comprises one or more compatible solid or liquid excipients or diluents which are suitable for topical oral administration. By "compatible," as used herein, is meant that the components of the composition are capable of being commingled without interaction in a manner which would substantially reduce the composition's stability and/or efficacy.
[0023] The term "substantially free" as used herein refers to the presence of no more than 0.05%, preferably no more than 0.01%, and more preferably no more than 0.001%, of an indicated material in a composition, by total weight of such composition.
[0024] The term "essentially free" as used herein means that the indicated material is not deliberately added to the composition, or preferably not present at analytically detectable levels. It is meant to include compositions whereby the indicated material is present only as an impurity of one of the other materials deliberately added.
[0025] While compositions and methods are described herein in terms of "comprising" various components or steps, the compositions and methods can also "consist essentially of" or "consist of" the various components or steps, unless stated otherwise.
[0026] As used herein, the word "or" when used as a connector of two or more elements is meant to include the elements individually and in combination; for example, X or Y, means X or Y or both.
[0027] As used herein, the articles "a" and "an" are understood to mean one or more of the material that is claimed or described, for example, "an oral care composition" or "a bleaching agent."
[0028] All measurements referred to herein are made at about 23.degree. C. (i.e. room temperature) unless otherwise specified.
[0029] Generally, groups of elements are indicated using the numbering scheme indicated in the version of the periodic table of elements published in Chemical and Engineering News, 63(5), 27, 1985. In some instances, a group of elements can be indicated using a common name assigned to the group; for example, alkali metals for Group 1 elements, alkaline earth metals for Group 2 elements, and so forth.
[0030] Several types of ranges are disclosed in the present invention. When a range of any type is disclosed or claimed, the intent is to disclose or claim individually each possible number that such a range could reasonably encompass, including end points of the range as well as any sub-ranges and combinations of sub-ranges encompassed therein.
[0031] The term "about" means that amounts, sizes, formulations, parameters, and other quantities and characteristics are not and need not be exact, but can be approximate and/or larger or smaller, as desired, reflecting tolerances, conversion factors, rounding off, measurement errors, and the like, and other factors known to those of skill in the art. In general, an amount, size, formulation, parameter or other quantity or characteristic is "about" or "approximate" whether or not expressly stated to be such. The term "about" also encompasses amounts that differ due to different equilibrium conditions for a composition resulting from a particular initial mixture. Whether or not modified by the term "about," the claims include equivalents to the quantities. The term "about" can mean within 10% of the reported numerical value, preferably within 5% of the reported numerical value.
[0032] The oral care composition can be in any suitable form, such as a solid, liquid, powder, paste, or combinations thereof. The oral care composition can be dentifrice, tooth gel, subgingival gel, mouth rinse, mousse, foam, mouth spray, lozenge, chewable tablet, chewing gum, tooth whitening strips, floss and floss coatings, breath freshening dissolvable strips, or denture care or adhesive product. The components of the dentifrice composition can be incorporated into a film, a strip, a foam, or a fiber-based dentifrice composition. The oral care composition can include a variety of active and inactive ingredients, such as, for example, but not limited to a hops extract, a tin ion source, a calcium ion source, water, a fluoride ion source, zinc ion source, one or more polyphosphates, humectants, surfactants, other ingredients, and the like, as well as any combination thereof, as described below.
[0033] Section headers are provided below for organization and convenience only. The section headers do not suggest that a compound cannot be within more than one section. In fact, compounds can fall within more than one section. For example, stannous chloride can be both a tin ion source and a biofilm modifier, stannous fluoride can be both a tin ion source and a fluoride ion source, glycine can be an amino acid, a buffering agent, and/or a biofilm modifier, among numerous other compounds that can fit amongst several categories and/or sections.
[0034] Humulus lupulus
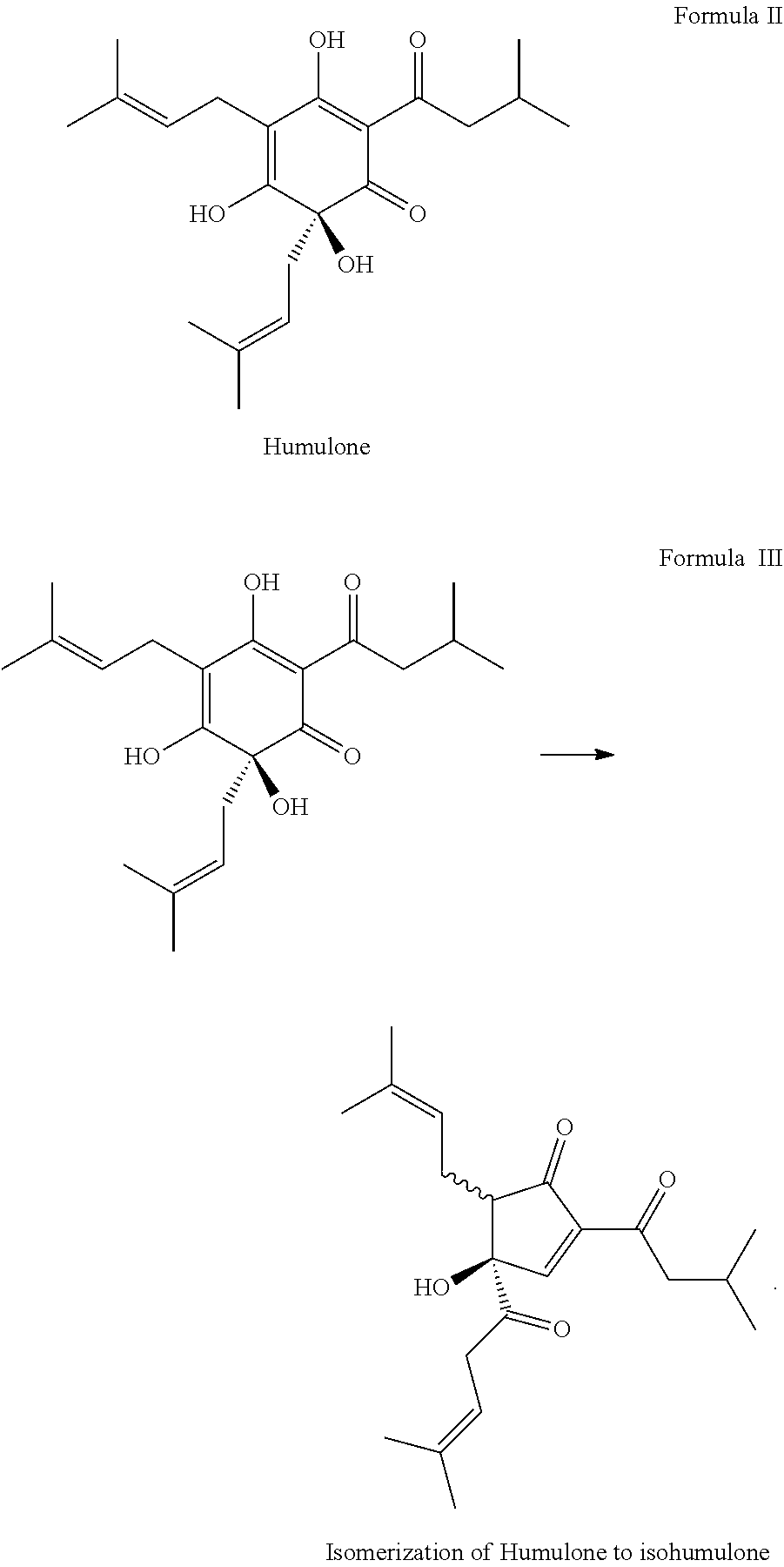
[0035] The oral care compositions of the present invention can comprise hops. The hops can comprise at least one hops compound from Formula I and/or Formula IV. The compound from Formula I and/or Formula IV can be provided by any suitable source, such as an extract from Humulus lupulus or Hops, Humulus lupulus itself, a synthetically derived compound, and/or salts, prodrugs, or other analogs thereof. The hops extract can comprise one or more hops alpha acids, one or more hops iso-alpha acids, one or more hops beta acids, one or more hops oils, one or more flavonoids, one or more solvents, and/or water. Suitable hops alpha acids (generically shown in Formula I) can include humulone (Formula II), adhumulone, cohumulone, posthumulone, prehumulone, and/or mixtures thereof. Suitable hops iso-alpha acids can include cis-isohumulone and/or trans-isohumulone. The isomerization of humulone into cis-isohumulone and trans-isohumulone can be represented by Formula III.
##STR00001##
[0036] A is the acidic hydroxyl functional group in the alpha position, B are the acidic hydroxyl functional groups in the beta position, and R is an alkyl functional group.
##STR00002##
[0037] Suitable hops beta acids can include lupulone, adlupulone, colupulone, and/or mixtures thereof. A suitable hops beta acid can include a compound a described in Formula IV, V, VI, and/or VII.
##STR00003##
[0038] B are the acidic hydroxyl functional groups in the beta position and R is an alkyl functional group.
##STR00004##
[0039] While hops alpha acids can demonstrate some antibacterial activity, hops alpha acids also have a bitter taste. The bitterness provided by hops alpha acids can be suitable for beer, but are not suitable for use in oral care compositions. In contrast, hops beta acids can be associated with a higher antibacterial and/or anticaries activity, but not as bitter a taste. Thus, a hops extract with a higher proportion of beta acids to alpha acids than normally found in nature, can be suitable for use in oral care compositions for use as an antibacterial and/or anticaries agent.
[0040] A natural hops source can comprise from about 2% to about 12%, by weight of the hops source, of hops beta acids depending on the variety of hops. Hops extracts used in other contexts, such as in the brewing of beer, can comprise from about 15% to about 35%, by weight of the extract, of hops beta acids. The hops extract desired herein can comprise at least about 35%, at least about 40%, at least about 45%, from about 35% to about 95%, from about 40% to about 90%, or from about 45% to about 99%, of hops beta acids. The hops beta acids can be in an acidic form (i.e. with attached hydrogen atom(s) to the hydroxl functional group(s)) or as a salt form.
[0041] A suitable hops extract is described in detail in U.S. Pat. No. 7,910,140, which is herein incorporated by reference in its entirety. The hops beta acids desired can be non-hydrogenated, partially hydrogenated by a non-naturally occurring chemical reaction, or hydrogenated by a non-naturally occurring chemical reaction. The hops beta acid can be essentially free of or substantially free of hydrogenated hops beta acid and/or hops acid. A non-naturally occurring chemical reaction is a chemical reaction that was conducted with the aid of chemical compound not found within Humulus lupulus, such as a chemical hydrogenation reaction conducted with high heat not normally experienced by Humulus lupulus in the wild and/or a metal catalyst.
[0042] A natural hops source can comprise from about 2% to about 12%, by weight of the hops source, of hops alpha acids. Hops extracts used in other contexts, such as in the brewing of beer, can comprise from about 15% to about 35%, by weight of the extract, of hops alpha acids. The hops extract desired herein can comprise less than about 10%, less than about 5%, less than about 1%, or less than about 0.5%, by weight of the extract, of hops alpha acids.
[0043] Hops oils can include terpene hydrocarbons, such as myrcene, humulene, caryophyllene, and/or mixtures thereof. The hops extract desired herein can comprise less than 5%, less than 2.5%, or less than 2%, by weight of the extract, of one or more hops oils.
[0044] Flavonoids present in the hops extract can include xanthohumol, 8-prenylnaringenin, isoxanthohumol, and/or mixtures thereof. The hops extract can be substantially free of, essentially free of, free of, or have less than 250 ppm, less than 150 ppm, and/or less than 100 ppm of one or more flavonoids.
[0045] As described in U.S. Pat. No. 5,370,863, hops acids have been previously added to oral care compositions. However, the oral care compositions taught by U.S. Pat. No. 5,370,863 only included up to 0.01%, by weight of the oral care composition. While not wishing to be bound by theory, it is believed that U.S. Pat. No. 5,370,863 could only incorporate a low amount of hops acids because of the bitterness of hops alpha acids. A hops extract with a low level of hops alpha acids would not have this concern.
[0046] The hops compound can be combined with or free from an extract from another plant, such as a species from genus Magnolia. The hops compounds can be combined with or free from triclosan.
[0047] The oral care composition can comprise from about 0.01% to about 10%, greater than 0.01% to about 10%, from about 0.05%, to about 10%, from about 0.1% to about 10%, from about 0.2% to about 10%, from about 0.2% to about 10%, from about 0.2% to about 5%, from about 0.25% to about 2%, from about 0.05% to about 2%, or from greater than 0.25% to about 2%, of hops, such as hops beta acid, as described herein. The hops, such as the hops beta acid, can be provided by a suitable hops extract, the hops plant itself, or a synthetically derived compound. The hops, such as hops beta acid, can be provided as neutral, acidic compounds, and/or as salts with a suitable counter ion, such as sodium, potassium, ammonia, or any other suitable counter ion.
[0048] The hops can be provided by a hops extract, such as an extract from Humulus lupulus with at least 35%, by weight of the extract, of hops beta acid and less than 1%, by weight of the hops extract, of hops alpha acid. The oral care composition can comprise 0.01% to about 10%, greater than 0.01% to about 10%, from about 0.05%, to about 10%, from about 0.1% to about 10%, from about 0.2% to about 10%, from about 0.2% to about 10%, from about 0.2% to about 5%, from about 0.25% to about 2%, from about 0.05% to about 2%, or from greater than 0.25% to about 2%, of hops extract, as described herein.
Fluoride Ion Source
[0049] The oral care composition can comprise fluoride, such as from a fluoride ion source. The fluoride ion source can comprise one or more fluoride containing compounds, such as stannous fluoride, sodium fluoride, titanium fluoride, calcium fluoride, calcium phosphate silicate fluoride, potassium fluoride, amine fluoride, sodium monofluorophosphate, zinc fluoride, and/or mixtures thereof.
[0050] The fluoride ion source and the tin ion source can be the same compound, such as for example, stannous fluoride, which can generate tin ions and fluoride ions. Additionally, the fluoride ion source and the tin ion source can be separate compounds, such as when the tin ion source is stannous chloride and the fluoride ion source is sodium monofluorophosphate or sodium fluoride.
[0051] The fluoride ion source and the zinc ion source can be the same compound, such as for example, zinc fluoride, which can generate zinc ions and fluoride ions. Additionally, the fluoride ion source and the zinc ion source can be separate compounds, such as when the zinc ion source is zinc phosphate and the fluoride ion source is stannous fluoride.
[0052] The fluoride ion source can be essentially free of or free of stannous fluoride. Thus, the oral care composition can comprise sodium fluoride, potassium fluoride, amine fluoride, sodium monofluorophosphate, zinc fluoride, and/or mixtures thereof.
[0053] The oral care composition can comprise a fluoride ion source capable of providing from about 50 ppm to about 5000 ppm, and preferably from about 500 ppm to about 3000 ppm of free fluoride ions. To deliver the desired amount of fluoride ions, the fluoride ion source may be present in the oral care composition at an amount of from about 0.0025% to about 5%, from about 0.01% to about 10%, from about 0.2% to about 1%, from about 0.5% to about 1.5%, or from about 0.3% to about 0.6%, by weight of the oral care composition. Alternatively, the oral care composition can comprise less than 0.1%, less than 0.01%, be essentially free of, substantially free of, or free of a fluoride ion source.
Tin Ion Source
[0054] The oral care composition of the present invention can comprise tin, such as from a tin ion source. The tin ion source can be any suitable compound that can provide tin ions in an oral care composition and/or deliver tin ions to the oral cavity when the dentifrice composition is applied to the oral cavity. The tin ion source can comprise one or more tin containing compounds, such as stannous fluoride, stannous chloride, stannous bromide, stannous iodide, stannous oxide, stannous oxalate, stannous sulfate, stannous sulfide, stannic fluoride, stannic chloride, stannic bromide, stannic iodide, stannic sulfide, and/or mixtures thereof. Tin ion source can comprise stannous fluoride, stannous chloride, and/or mixture thereof. The tin ion source can also be a fluoride-free tin ion source, such as stannous chloride.
[0055] The oral care composition can comprise from about 0.0025% to about 5%, from about 0.01% to about 10%, from about 0.2% to about 1%, from about 0.5% to about 1.5%, or from about 0.3% to about 0.6%, by weight of the oral care composition, of a tin ion source.
Ca Ion Source
[0056] The oral care composition of the present invention can comprise calcium, such as from a calcium ion source. The calcium ion source can be any suitable compound or molecule that can provide calcium ions in an oral care composition and/or deliver calcium ions to the oral cavity when the oral care composition is applied to the oral cavity. The calcium ion source can comprise a calcium salt, a calcium abrasive, and/or combinations thereof. In some cases, a calcium salt may also be considered a calcium abrasive or a calcium abrasive may also be considered a calcium salt.
[0057] The calcium ion source can comprise a calcium abrasive. The calcium abrasive can be any suitable abrasive compound that can provide calcium ions in an oral care composition and/or deliver calcium ions to the oral cavity when the oral care composition is applied to the oral cavity. The calcium abrasive can comprise one or more calcium abrasive compounds, such as calcium carbonate, precipitated calcium carbonate (PCC), ground calcium carbonate (GCC), chalk, dicalcium phosphate, calcium pyrophosphate, and/or mixtures thereof.
[0058] The calcium ion source can comprise a calcium salt, or a compound that can provide calcium ions in an oral care composition and/or deliver calcium ions to the oral cavity when the oral care composition is applied to the oral cavity that can not act as an abrasive. The calcium salt can comprise one or more calcium compounds, such as calcium chloride, calcium nitrate, calcium phosphate, calcium lactate, calcium oxalate, calcium oxide, calcium gluconate, calcium citrate, calcium bromide, calcium iodate, calcium iodide, hydroxyapatite, fluorapatite, calcium sulfate, calcium glycerophosphate, and/or combinations thereof.
[0059] The oral care composition can comprise from about 5% to about 70%, from about 10% to about 50%, from about 10% to about 60%, from about 20% to about 50%, from about 25% to about 40%, or from about 1% to about 50% of a calcium ion source.
Buffering Agent
[0060] The oral care composition can comprise a buffering agent. The buffering agent can be a weak acid or base that can maintain a particular pH at a selected site in the oral cavity. For example, the buffering agent can maintain a pH at a tooth's surface to mitigate the impact of plaque acids produced by bacteria. The buffering agent can comprise a conjugate acid of an ion also present in the oral care composition. For example, if the calcium ion source comprises calcium carbonate, the buffering agent can comprise a bicarbonate anion (-HCO.sub.3.sup.-). The buffering agent can comprise a conjugate acid/base pair, such as citric acid and sodium citrate.
[0061] Suitable buffering systems can include phosphate, citrate salts, carbonate/bicarbonate salts, a tris buffer, imidazole, urea, borate, and/or combinations thereof. Suitable buffering agents include bicarbonate salts, such as sodium bicarbonate, glycine, orthophosphate, arginine, urea, and or/combinations thereof.
[0062] The oral care composition can comprise from about 1% to about 30%, from about 5% to about 25% or from about 10% to about 20%, of one or more buffering agents.
Biofilm Modifier
[0063] The oral care composition can comprise one or more biofilm modifiers. A biofilm modifier can comprise a polyol, an ammonia generating compound, and/or a glucosyltransferase inhibitor.
[0064] A polyol is an organic compound with more than one hydroxyl functional groups. The polyol can be any suitable compound that can weakly associate, interact, or bond to tin ions while the oral care composition is stored prior to use. The polyol can be a sugar alcohol, which area class of polyols that can be obtained through the hydrogenation of sugar compounds with the formula (CHOH).sub.nH.sub.2. The polyol can be glycerin, erythritol, xylitol, sorbitol, mannitol, butylene glycol, lactitol, and/or combinations thereof. The oral care composition can comprise 0.01% to about 70%, from about 5% to about 70%, from about 5% to about 50%, from about 10% to about 60%, from about 10% to about 25%, or from about 20% to about 80%, by weight of the oral care composition, of a polyol.
[0065] The ammonia generating compound can be any suitable compound that can generate ammonia upon delivery to the oral cavity. Suitable ammonia generating compounds include arginine, urea, and/or combinations thereof. The oral care composition can comprise from about 0.01% to about 10%, from about 1% to about 5%, or from about 1% to about 25% of one or more ammonia generating compounds.
[0066] The glucosyltransferase inhibitor can be any suitable compound that can inhibit a glucosyltransferase. Glucosyltransferases are enzymes that can establish natural glycosidic linkages. In particular, these enzymes break down poly- or oligosaccharide moieties into simple sugars for bacteria associated with dental caries. As such, any compound that can inhibit this process can belp prevent dental caries. Suitable glucosyltransferase inhibitors include oleic acid, epicatechin, tannins, tannic acid, moenomycin, caspofungin, ethambutol, lufenuron, and/or combinations thereof. The oral care composition can comprise from about 0.001% to about 5%, from about 0.01% to about 2%, or about 1% of one or more glucosyltransferase inhibitors.
Metal Ion Source
[0067] The oral care composition can comprise metal, such as from a metal ion source comprising one or more metal ions. The metal ion source can comprise or be in addition to the tin ion source and/or the zinc ion source, as described herein. Suitable metal ion sources include compounds with metal ions, such as, but not limited to Sn, Zn, Cu, Mn, Mg, Sr, Ti, Fe, Mo, B, Ba, Ce, Al, In and/or mixtures thereof. The trace metal source can be any compound with a suitable metal and any accompanying ligands and/or anions.
[0068] Suitable ligands and/or anions that can be paired with metal ion sources include, but are not limited to acetate, ammonium sulfate, benzoate, bromide, borate, carbonate, chloride, citrate, gluconate, glycerophosphate, hydroxide, iodide, oxide, propionate, D-lactate, DL-lactate, orthophosphate, pyrophosphate, sulfate, nitrate, tartrate, and/or mixtures thereof.
[0069] The oral care composition can comprise from about 0.01% to about 10%, from about 1% to about 5%, or from about 0.5% to about 15% of a metal ion source.
Antibacterial Agents
[0070] The oral care composition can comprise one or more antibacterial agents. Suitable antibacterial agents include any molecule that provides antibacterial activity in the oral cavity. Suitable antibacterial agents include hops acids, tin ion sources, benzyl alcohol, sodium benzoate, menthylglycyl acetate, menthyl lactate, L-menthol, o-neomenthol, chlorophyllin copper complex, phenol, oxyquinoline, and/or combinations thereof.
[0071] The oral care composition can comprise from about 0.01% to about 10%, from about 1% to about 5%, or from about 0.5% to about 15% of an antibacterial agent.
Bioactive Materials
[0072] The oral care composition can also include bioactive materials suitable for the remineralization of a tooth. Suitable bioactive materials include bioactive glasses, Novamin.TM., Recaldent.TM. hydroxyapatite, one or more amino acids, such as, for example, arginine, citrulline, glycine, lysine, or histidine, or combinations thereof. Suitable examples of compositions comprising arginine are found in U.S. Pat. Nos. 4,154,813 and 5,762,911, which are herein incorporated by reference in their entirety. Other suitable bioactive materials include any calcium phosphate compound. Other suitable bioactive materials include compounds comprising a calcium source and a phosphate source.
[0073] Amino acids are organic compounds that contain an amine functional group, a carboxyl functional group, and a side chain specific to each amino acid. Suitable amino acids include, for example, amino acids with a positive or negative side chain, amino acids with an acidic or basic side chain, amino acids with polar uncharged side chains, amino acids with hydrophobic side chains, and/or combinations thereof. Suitable amino acids also include, for example, arginine, histidine, lysine, aspartic acid, glutamic acid, serine, threonine, asparagine, glutamine, cysteine, selenocysteine, glycine, proline, alanine, valine, isoleucine, leucine, methionine, phenylalanine, tyrosine, tryptophan, citrulline, ornithine, creatine, diaminobutonic acid, diaminoproprionic acid, salts thereof, and/or combinations thereof.
[0074] Bioactive glasses are comprising calcium and/or phosphate which can be present in a proportion that is similar to hydroxyapatite. These glasses can bond to the tissue and are biocompatible. Bioactive glasses can include a phosphopeptide, a calcium source, phosphate source, a silica source, a sodium source, and/or combinations thereof.
[0075] The oral care composition can comprise from about 0.01% to about 20%, from about 0.1% to about 10%, or from about 1% to about 10% of a bioactive material by weight of the oral care composition.
Abrasive
[0076] The oral care composition can comprise a calcium abrasive, as described herein, and/or a non-calcium abrasive, such as bentonite, silica gel (by itself, and of any structure), precipitated silica, amorphous precipitated silica (by itself, and of any structure as well), hydrated silica, perlite, titanium dioxide, calcium pyrophosphate, dicalcium phosphate dihydrate, alumina, hydrated alumina, calcined alumina, aluminum silicate, insoluble sodium metaphosphate, insoluble potassium metaphosphate, insoluble magnesium carbonate, zirconium silicate, particulate thermosetting resins and other suitable abrasive materials. Such materials can be introduced into the oral care compositions to tailor the polishing characteristics of the target dentifrice formulation. The oral care composition can comprise from about 5% to about 70%, from about 10% to about 50%, from about 10% to about 60%, from about 20% to about 50%, from about 25% to about 40%, or from about 1% to about 50%, by weight of the oral care composition, of the non-calcium abrasive.
[0077] Alternatively, the oral care composition can be substantially free of, essentially free of, or free of silica, alumina, or any other non-calcium abrasive. The oral care composition can comprise less than about 5%, less than about 1%, less than about 0.5%, less than about 0.1%, or 0% of a non-calcium abrasive, such as silica and/or alumina.
Water
[0078] The oral care composition of the present invention can be anhydrous, a low water formulation, or a high water formulation. In total, the oral care composition can comprise from 0% to about 99%, from about 5% to about 75%, about 20% or greater, about 30% or greater, or about 50% or greater by weight of the composition, of water. Preferably, the water is USP water.
[0079] In a high water oral care composition and/or toothpaste formulation, the oral care composition comprises from about 45% to about 75%, by weight of the composition, of water. The high water oral care composition and/or toothpaste formulation can comprise from about 45% to about 65%, from about 45% to about 55%, or from about 46% to about 54%, by weight of the composition, of water. The water may be added to the high water formulation and/or may come into the composition from the inclusion of other ingredients.
[0080] In a low water oral care composition and/or toothpaste formulation, the oral care composition comprises from about 5% to about 45%, by weight of the composition, of water. The low water oral care composition can comprise from about 5% to about 35%, from about 10% to about 25%, or from about 20% to about 25%, by weight of the composition, of water. The water may be added to the low water formulation and/or may come into the composition from the inclusion of other ingredients.
[0081] In an anhydrous oral care composition and/or toothpaste formulation, the oral care composition comprises less than about 10%, by weight of the composition, of water. The anhydrous composition comprises less than about 5%, less than about 1%, or 0%, by weight of the composition, of water. The water may be added to the anhydrous formulation and/or may come into the composition from the inclusion of other ingredients.
[0082] A mouth rinse formulation comprises from about 75% to about 99%, from about 75% to about 95%, or from about 80% to about 95% of water.
[0083] The composition can also comprise other orally acceptable carrier materials, such as alcohol, humectants, polymers, surfactants, and acceptance improving agents, such as flavoring, sweetening, coloring and/or cooling agents.
pH
[0084] The pH of the disclosed composition can be from about 4 to about 10, from about 7 to about 10, greater than 7 to about 10, greater than 8 to about 10, greater than 7, greater than 7.5, greater than 8, greater than 9, or from about 8.5 to about 10.
Zinc Ion Source
[0085] The oral care composition can comprise zinc, such as from a zinc ion source. The zinc ion source can comprise one or more zinc containing compounds, such as zinc fluoride, zinc lactate, zinc oxide, zinc phosphate, zinc chloride, zinc acetate, zinc hexafluorozirconate, zinc sulfate, zinc tartrate, zinc gluconate, zinc citrate, zinc malate, zinc glycinate, zinc pyrophosphate, zinc metaphosphate, zinc oxalate, and/or zinc carbonate. The zinc ion source can be a fluoride-free zinc ion source, such as zinc phosphate, zinc oxide, and/or zinc citrate.
[0086] The zinc ion source may be present in the total oral care composition at an amount of from about 0.01% to about 10%, from about 0.2% to about 1%, from about 0.5% to about 1.5%, or from about 0.3% to about 0.6%, by weight of the dentifrice composition.
Polyphosphates
[0087] The oral care composition can comprise polyphosphate, such as from a polyphosphate source. A polyphosphate source can comprise one or more polyphosphate molecules. Polyphosphates are a class of materials obtained by the dehydration and condensation of orthophosphate to yield linear and cyclic polyphosphates of varying chain lengths. Thus, polyphosphate molecules are generally identified with an average number (n) of polyphosphate molecules, as described below. A polyphosphate is generally understood to consist of two or more phosphate molecules arranged primarily in a linear configuration, although some cyclic derivatives may be present.
[0088] Preferred polyphosphates are those having an average of two or more phosphate groups so that surface adsorption at effective concentrations produces sufficient non-bound phosphate functions, which enhance the anionic surface charge as well as hydrophilic character of the surfaces. Preferred in this invention are the linear polyphosphates having the formula: XO (XPO.sub.3).sub.nX, wherein X is sodium, potassium, ammonium, or any other alkali metal cations and n averages from about 2 to about 21. Alkali earth metal cations, such as calcium, are not preferred because they tend to form insoluble fluoride salts from aqueous solutions comprising a fluoride ions and alkali earth metal cations. Thus, the oral care compositions disclosed herein can be free of, essentially free of, or substantially free of calcium pyrophosphate.
[0089] Some examples of suitable polyphosphate molecules include, for example, pyrophosphate tripolyphosphate (n=3), tetrapolyphosphate (n=4), sodaphos polyphosphate (n=6), hexaphos polyphosphate (n=13), benephos polyphosphate (n=14), hexametaphosphate (n=21), which is also known as Glass H. Polyphosphates can include those polyphosphate compounds manufactured by FMC Corporation, ICL Performance Products, and/or Astaris.
[0090] The oral care composition can comprise from about 0.01% to about 15%, from about 0.1% to about 10%, from about 0.5% to about 5%, from about 1 to about 20%, or about 10% or less, by weight of the oral care composition, of the polyphosphate source.
Humectants
[0091] The oral care composition can comprise one or more humectants, have low levels of a humectant, be essentially free of, be substantially free of, or be free of a humectant. Humectants serve to add body or "mouth texture" to an oral care composition or dentifrice as well as preventing the dentifrice from drying out. Suitable humectants include polyethylene glycol (at a variety of different molecular weights), propylene glycol, glycerin (glycerol), erythritol, xylitol, sorbitol, mannitol, butylene glycol, lactitol, hydrogenated starch hydrolysates, and/or mixtures thereof. The oral care composition can comprise one or more humectants each at a level of from 0 to about 70%, from about 5% to about 50%, from about 10% to about 60%, or from about 20% to about 80%, by weight of the oral care composition.
Surfactants
[0092] The oral care composition can comprise one or more surfactants. The surfactants can be used to make the compositions more cosmetically acceptable. The surfactant is preferably a detersive material which imparts to the composition detersive and foaming properties. Suitable surfactants are safe and effective amounts of anionic, cationic, nonionic, zwitterionic, amphoteric and betaine surfactants.
[0093] Suitable anionic surfactants include, for example, the water soluble salts of alkyl sulfates having from 8 to 20 carbon atoms in the alkyl radical and the water-soluble salts of sulfonated monoglycerides of fatty acids having from 8 to 20 carbon atoms. Sodium lauryl sulfate (SLS) and sodium coconut monoglyceride sulfonates are examples of anionic surfactants of this type. Other suitable anionic surfactants include sarcosinates, such as sodium lauroyl sarcosinate, taurates, sodium lauryl sulfoacetate, sodium lauroyl isethionate, sodium laureth carboxylate, and sodium dodecyl benzene sulfonate. Combinations of anionic surfactants can also be employed.
[0094] Another suitable class of anionic surfactants are alkyl phosphates. The surface active organophosphate agents can have a strong affinity for enamel surface and have sufficient surface binding propensity to desorb pellicle proteins and remain affixed to enamel surfaces. Suitable examples of organophosphate compounds include mono-, di- or triesters represented by the general structure below wherein Z.sub.1, Z.sub.2, or Z.sub.3 may be identical or different with at least one being an organic moiety. Z.sub.1, Z.sub.2, or Z.sub.3 can be selected from linear or branched, alkyl or alkenyl group of from 1 to 22 carbon atoms, optionally substituted by one or more phosphate groups; alkoxylated alkyl or alkenyl, (poly)saccharide, polyol or polyether group.
##STR00005##
Some other agents include alkyl or alkenyl phosphate esters represented by the following structure:
##STR00006##
wherein R.sub.1 represents a linear or branched, alkyl or alkenyl group of from 6 to 22 carbon atoms, optionally substituted by one or more phosphate groups; n and m, are individually and separately, 2 to 4, and a and b, individually and separately, are 0 to 20; Z and Z may be identical or different, each represents hydrogen, alkali metal, ammonium, protonated alkyl amine or protonated functional alkylamine, such as analkanolamine, or a R--(OCH2)(OCH)-- group. Examples of suitable agents include alkyl and alkyl (poly)alkoxy phosphates such as lauryl phosphate; PPGS ceteareth-10 phosphate; laureth-1 phosphate; laureth-3 phosphate; laureth-9 phosphate; trilaureth-4 phosphate; C.sub.12-18 PEG 9 phosphate: and sodium dilaureth-10 phosphate. The alkyl phosphate can be polymeric. Examples of polymeric alkyl phosphates include those containing repeating alkoxy groups as the polymeric portion, in particular 3 or more ethoxy, propoxy isopropoxy or butoxy groups.
[0095] Other suitable anionic surfactants are sarcosinates, isethionates and taurates, especially their alkali metal or ammonium salts. Examples include: lauroyl sarcosinate, myristoyl sarcosinate, palmitoyl sarcosinate, stearoyl sarcosinate oleoyl sarcosinate, or combinations thereof.
[0096] Other suitable anionic surfactants include sodium or potassium alkyl sulfates, such as sodium lauryl sulfate, acyl isethionates, acyl methyl isethionates, alkyl ether carboxylates, acyl alaninates, acyl gulatames, acyl glycinates, acyl sarconsinates, sodium methyl acyl taurates, sodium laureth sulfosuccinates, alpha olefin sulfonates, alkyl benze sulfonates, sodium lauroyl lactylate, sodium laurylglucosides hydroxypropyl sulfonate, and/or combinations.
[0097] Zwitterionic or amphoteric surfactants useful herein include derivatives of aliphatic quaternary ammonium, phosphonium, and Sulfonium compounds, in which the aliphatic radicals can be straight chain or branched, and one of the aliphatic substituents contains from 8 to 18 carbon atoms and one contains an anionic water-solubilizing group, e.g., carboxy, sulfonate, sulfate, phosphate or phosphonate. Suitable betaine surfactants are disclosed in U.S. Pat. No. 5,180,577. Typical alkyl dimethyl betaines include decyl betaine or 2-(N-decyl-N,N-dimethylammonio) acetate, coco-betaine or 2-(N-coco-N,N-dimethyl ammonio)acetate, myristyl betaine, palmityl betaine, lauryl betaine, cetyl betaine, cetyl betaine, stearyl betaine, etc. The amidobetaines can be exemplified by cocoamidoethyl betaine, cocoamidopropyl betaine (CADB), and lauramidopropyl betaine. Other suitable amphoteric surfactants include betaines, sultaines, sodium laurylamphoacetates, alkylamphodiacetates, and/or combinations thereof.
[0098] Cationic surfactants useful in the present invention include, for example, derivatives of quaternary ammonium compounds having one long alkyl chain containing from 8 to 18 carbon atoms such as lauryl trimethylammonium chloride; cetyl pyridinium chloride; cetyl trimethyl-ammonium bromide; cetyl pyridinium fluoride or combinations thereof.
[0099] Nonionic surfactants that can be used in the compositions of the present invention include, for example, compounds produced by the condensation of alkylene oxide groups (hydrophilic in nature) with an organic hydrophobic compound which may be aliphatic or alkylaromatic in nature. Examples of suitable nonionic surfactants can include the Pluronics.RTM. which are poloxamers, polyethylene oxide condensates of alkyl phenols, products derived from the condensation of ethylene oxide with the reaction product of propylene oxide and ethylene diamine, ethylene oxide condensates of aliphatic alcohols, long chain tertiary amine oxides, long chain tertiary phosphine oxides, long chain dialkyl sulfoxides and combinations of such materials. Other suitable non-ionic surfactants includes alkyl glucamides, alkyl glucosides, and/or combinations thereof.
[0100] The one or more surfactants can also include one or more natural and/or naturally derived surfactants. Natural surfactants can include surfactants that are derived from natural products and/or surfactants that are minimally or not processed. Natural surfactants can include hydrogenated, non-hydrogenated, or partially hydrogenated vegetable oils, olus oil, passiflora incarnata oil, candelilla cera, coco-caprylate, caprate, dicaprylyl ether, lauryl alcohol, myristyl myristate, dicaprylyl ether, caprylic acid, caprylic ester, octyl decanoate, octyl octanoate, undecane, tridecane, decyl oleate, oleic acid decylester, cetyl palmitate, stearic acid, palmitic acid, glyceryl stearate, hydrogenated, non-hydrogenated, or partially hydrogenated vegetable glycerides, Polyglyceryl-2 dipolyhydroxystearate, cetearyl alcohol, sucrose polystearate, glycerin, octadodecanol, hydrolyzed, partially hydrolyzed, or non-hydrolyzed vegetable protein, hydrolyzed, partially hydrolyzed, or non-hydrolyzed wheat protein hydrolysate, polyglyceryl-3 diisostearate, glyceryl oleate, myristyl alcohol, cetyl alcohol, sodium cetearyl sulfate, cetearyl alcohol, glyceryl laurate, capric triglyceride, coco-glycerides, lectithin, dicaprylyl ether, xanthan gum, sodium coco-sulfate, ammonium lauryl sulfate, sodium cocoyl sulfate, sodium cocoyl glutamate, polyalkylglucosides, such as decyl glucoside, cetearyl glucoside, cetyl stearyl polyglucoside, coco-glucoside, and lauryl glucoside, and/or combinations thereof. Natural surfactants can include any of the Natrue ingredients marketed by BASF, such as, for example, CegeSoft.RTM., Cetiol.RTM., Cutina.RTM., Dehymuls.RTM., Emulgade.RTM., Emulgin.RTM., Eutanol.RTM., Gluadin.RTM., 1Lameform.RTM., LameSoft.RTM., Lanette.RTM., Monomuls.RTM., Myritol.RTM., Plantacare.RTM., Plantaquat.RTM., Platasil.RTM., Rheocare.RTM., Sulfopon.RTM., Texapon.RTM., and/or combinations thereof.
[0101] Other specific examples of surfactants include sodium lauryl sulfate, sodium lauryl isethionate, sodium lauroyl methyl isethionate, sodium cocoyl glutamate, sodium dodecyl benzene sulfonate, alkali metal or ammonium salts of lauroyl sarcosinate, myristoyl sarcosinate, palmitoyl sarcosinate, stearoyl sarcosinate and oleoyl sarcosinate, polyoxyethylene sorbitan monostearate, isostearate and laurate, sodium lauryl sulfoacetate, N-lauroyl sarcosine, the sodium, potassium, and ethanolamine salts of N-lauroyl, N-myristoyl, or N-palmitoyl sarcosine, polyethylene oxide condensates of alkyl phenols, cocoamidopropyl betaine, lauramidopropyl betaine, palmityl betaine, sodium cocoyl glutamate, and the like. Additional surfactants desired include fatty acid salts of glutamate, alkyl glucoside, salts of taurates, betaines, caprylates, and/or mixtures thereof. The oral care composition can also be sulfate free.
[0102] The oral care composition can comprise one or more surfactants each at a level from about 0.01% to about 15%, from about 0.3% to about 10%, or from about 0.3% to about 2.5%, by weight of the oral care composition.
Thickening Agents
[0103] The oral care composition can comprise one or more thickening agents. Thickening agents can be useful in the oral care compositions to provide a gelatinous structure that stabilizes the dentifrice and/or toothpaste against phase separation. Suitable thickening agents include polysaccharides, polymers, and/or silica thickeners.
[0104] The thickening agent can comprise one or more polysaccharides. Some non-limiting examples of polysaccharides include starch; glycerite of starch; gums such as gum karaya (sterculia gum), gum tragacanth, gum arabic, gum ghatti, gum acacia, xanthan gum, guar gum and cellulose gum; magnesium aluminum silicate (Veegum); carrageenan; sodium alginate; agar-agar; pectin; gelatin; cellulose compounds such as cellulose, microcrystalline cellulose, carboxymethyl cellulose, hydroxyethyl cellulose, hydroxypropyl cellulose, hydroxymethyl cellulose, hydroxymethyl carboxypropyl cellulose, methyl cellulose, ethyl cellulose, and sulfated cellulose; natural and synthetic clays such as hectorite clays; and mixtures thereof.
[0105] Other polysaccharides that are suitable for use herein include carageenans, gellan gum, locust bean gum, xanthan gum, carbomers, poloxamers, modified cellulose, and mixtures thereof. Carageenan is a polysaccharide derived from seaweed. There are several types of carageenan that may be distinguished by their seaweed source and/or by their degree of and position of sulfation. The thickening agent can comprise kappa carageenans, modified kappa carageenans, iota carageenans, modified iota carageenans, lambda carrageenan, and mixtures thereof. Carageenans suitable for use herein include those commercially available from the FMC Company under the series designation "Viscarin," including but not limited to Viscarin TP 329, Viscarin TP 388, and Viscarin TP 389.
[0106] The thickening agent can comprise one or more polymers. The polymer can be a polyethylene glycol (PEG), a polyvinylpyrrolidone (PVP), polyacrylic acid, a polymer derived from at least one acrylic acid monomer, a copolymer of maleic anhydride and methyl vinyl ether, a crosslinked polyacrylic acid polymer, of various weight percentages of the oral care composition as well as various ranges of average molecular ranges. Alternatively, the oral care composition can be free of, essentially free of, or substantially free of a copolymer of maleic anhydride and methyl vinyl ether.
[0107] The thickening agent can comprise one or more inorganic thickening agents. Some non-limiting examples of suitable inorganic thickening agents include colloidal magnesium aluminum silicate, silica thickeners. Useful silica thickeners include, for example, include, as a non-limiting example, an amorphous precipitated silica such as ZEODENT.RTM. 165 silica. Other non-limiting silica thickeners include ZEODENT.RTM. 153, 163, and 167, and ZEOFREE.RTM. 177 and 265 silica products, all available from Evonik Corporation, and AEROSIL.RTM. fumed silicas.
[0108] The oral care composition can comprise from 0.01% to about 15%, from 0.1% to about 10%, from about 0.2% to about 5%, or from about 0.5% to about 2% of one or more thickening agents.
Prenylated Flavonoids
[0109] The oral care composition can comprise prenylated flavonoid. Flavonoids are a group of natural substances found in a wide range of fruits, vegetables, grains, bark, roots, stems, flowers, tea, and wine. Flavonoids can have a variety of beneficial effects on health, such as antioxidative, anti-inflammatory, antimutagenic, anticarcinogenic, and antibacterial benefits. Prenylated flavonoids are flavonoids that include at least one prenyl functional group (3-methylbut-2-en-1-yl, as shown in Formula VIII), which has been previously identified to facilitate attachment to cell membranes. Thus, while not wishing to being bound by theory, it is believed that the addition of a prenyl group, i.e. prenylation, to a flavonoid can increase the activity of the original flavonoid by increasing the lipophilicity of the parent molecule and improving the penetration of the prenylated molecule into the bacterial cell membrane. Increasing the lipophilicity to increase penetration into the cell membrane can be a double-edged sword because the prenylated flavonoid will tend towards insolubility at high Log P values (high lipophilicity). Log P can be an important indicator of antibacterial efficacy.
[0110] As such, the term prenylated flavonoids can include flavonoids found naturally with one or more prenyl functional groups, flavonoids with a synthetically added prenyl functional group, and/or prenylated flavonoids with additional prenyl functional groups synthetically added.
##STR00007##
[0111] Prenyl Function Group with R Representing the other Portions of the Molecule
[0112] Other suitable functionalities of the parent molecule that improve the structure-activity relationship (e.g,. structure-MIC relationship) of the prenylated molecule include additional heterocycles containing nitrogen or oxygen, alkylamino chains, or alkyl chains substituted onto one or more of the aromatic rings of the parent flavonoid.
[0113] Flavonoids can have a 15-carbon skeleton with at least two phenyl rings and at least one heterocyclic ring. Some suitable flavonoid backbones can be shown in Formula IX (flavone backbone), Formula X (isoflavan backbone), and/or Formula XI (neoflavonoid backbone).
##STR00008##
[0114] Other suitable subgroups of flavonoids include anthocyanidins, anthoxanthins, flavanones, flavanonols, flavans, isoflavonoids, chalcones and/or combinations thereof.
[0115] Prenylated flavonoids can include naturally isolated prenylated flavonoids or naturally isolated flavonoids that are synthetically altered to add one or more prenyl functional groups through a variety of synthetic processes that would be known to a person of ordinary skill in the art of synthetic organic chemistry.
[0116] Other suitable prenylated flavonoids can include Bavachalcone, Bavachin, Bavachinin, Corylifol A, Epimedin A, Epimedin AI, Epimedin B, Epimedin C, Icariin, Icariside I, Icariside II, Icaritin, Isobavachalcone, Isoxanthohumol, Neobavaisoflavone, 6-Prenylnaringenin, 8-Prenylnaringenin, Sophoraflavanone G, (-)-Sophoranone, Xanthohumol, Quercetin, Macelignan, Kuraridin, Kurarinone, Kuwanon G, Kuwanon C, Panduratin A, 6-geranylnaringenin, Australone A, 6,8-Diprenyleriodictyol, dorsmanin C, dorsmanin F, 8-Prenylkaempferol, 7-O-Methylluteone, luteone, 6-prenylgenistein, isowighteone, lupiwighteone, and/or combinations thereof. Other suitable prenylated flavonoids include cannflavins, such as Cannflavin A, Cannflavin B, and/or Cannflavin C.
[0117] Preferably, the prenylated flavonoid has a high probability of having a MIC of less than about 25 ppm for S. aureus, a gram-positive bacterium. Suitable prenylated flavonoids include Bavachin, Bavachinin, Corylifol A, Icaritin, Isoxanthohumol, Neobavaisoflavone, 6-Prenylnaringenin, 8-Prenylnaringenin, Sophoraflavanone G, (-)-Sophoranone, Kurarinone, Kuwanon C, Panduratin A, and/or combinations thereof.
[0118] Preferably, the prenylated flavonoid has a high probability of having a MIC of less than about 25 ppm for E. coli, a gram-negative bacterium. Suitable prenylated flavonoids include Bavachinin, Isoxanthohumol, 8-Prenylnaringenin, Sophoraflavanone G, Kurarinone, Panduratin A, and/or combinations thereof.
[0119] Approximately 1000 prenylated flavonoids have been identified from plants. According to the number of prenylated flavonoids reported before, prenylated flavonones are the most common subclass and prenylated flavanols is the rarest sub-class. Even though natural prenylated flavonoids have been detected to have diversely structural characteristics, they have a narrow distribution in plants, which are different to the parent flavonoids as they are present almost in all plants. Most of prenylated flavonoids are found in the following families, including Cannabaceae, Guttiferae, Leguminosae, Moraceae, Rutaceae and Umbelliferae. Leguminosae and Moraceae, due to their consumption as fruits and vegetables, are the most frequently investigated families and many novel prenylated flavonoids have been explored. Humulus lupulus of the Cannabaceae include 8-prenylnaringenin and xanthohumol, which can play a role in the health benefits of beer.
[0120] The prenylated flavonoid can be incorporated through a hops extract, incorporated in a separately added extract, or added as a separate component of the oral care compositions disclosed herein.
[0121] Suitable prenylated flavonoids can have a particular octanol-water partitioning coefficient. The octanol-water partitioning coefficient can be used to predict the lipophilicity of a compound. Without wishing to being bound by theory, it is believed that compounds that fall within the ranges described herein will be able to enter and/or disrupt the primarily hydrophobic phospholipid bilayer that makes of the cell membrane of microorganisms. Thus, the octanol-water partitioning coefficient can be correlated to the antibacterial effect of prenylated flavonoids. Suitable prenylated flavonoids can have a log P of at least about 2, at least about 4, from about 2 to about 10, from about 4 to about 10, from about 4 to about 7, or from about 4 to about 7.
[0122] The oral care composition can comprise at least about 0.001%, from about 0.001% to about 5%, from about 0.01% to about 2%, from about 0.0001% to about 2%, or at least about 0.05% of prenylated flavonoid.
Other Ingredients
[0123] The oral care composition can comprise a variety of other ingredients, such as flavoring agents, sweeteners, colorants, preservatives, buffering agents, or other ingredients suitable for use in oral care compositions, as described below.
[0124] Flavoring agents also can be added to the oral care composition. Suitable flavoring agents include oil of wintergreen, oil of peppermint, oil of spearmint, clove bud oil, menthol, anethole, methyl salicylate, eucalyptol, cassia, 1-menthyl acetate, sage, eugenol, parsley oil, oxanone, alpha-irisone, marjoram, lemon, orange, propenyl guaethol, cinnamon, vanillin, ethyl vanillin, heliotropine, 4-cis-heptenal, diacetyl, methyl-para-tert-butyl phenyl acetate, and mixtures thereof. Coolants may also be part of the flavor system. Preferred coolants in the present compositions are the paramenthan carboxyamide agents such as N-ethyl-p-menthan-3-carboxamide (known commercially as "WS-3") or N-(Ethoxycarbonylmethyl)-3-p-menthanecarboxamide (known commercially as "WS-5"), and mixtures thereof. A flavor system is generally used in the compositions at levels of from about 0.001% to about 5%, by weight of the oral care composition. These flavoring agents generally comprise mixtures of aldehydes, ketones, esters, phenols, acids, and aliphatic, aromatic and other alcohols. Sweeteners can be added to the oral care composition to impart a pleasing taste to the product.
[0125] Suitable sweeteners include saccharin (as sodium, potassium or calcium saccharin), cyclamate (as a sodium, potassium or calcium salt), acesulfame-K, thaumatin, neohesperidin dihydrochalcone, ammoniated glycyrrhizin, dextrose, levulose, sucrose, mannose, sucralose, stevia, and glucose.
[0126] Colorants can be added to improve the aesthetic appearance of the product. Suitable colorants include without limitation those colorants approved by appropriate regulatory bodies such as the FDA and those listed in the European Food and Pharmaceutical Directives and include pigments, such as TiO.sub.2, and colors such as FD&C and D&C dyes.
[0127] Preservatives also can be added to the oral care compositions to prevent bacterial growth. Suitable preservatives approved for use in oral compositions such as methylparaben, propylparaben, benzoic acid, and sodium benzoate can be added in safe and effective amounts.
[0128] Titanium dioxide may also be added to the present composition. Titanium dioxide is a white powder which adds opacity to the compositions. Titanium dioxide generally comprises from about 0.25% to about 5%, by weight of the oral care composition.
[0129] Other ingredients can be used in the oral care composition, such as desensitizing agents, healing agents, other caries preventative agents, chelating/sequestering agents, vitamins, amino acids, proteins, other anti-plaque/anti-calculus agents, opacifiers, antibiotics, anti-enzymes, enzymes, pH control agents, oxidizing agents, antioxidants, and the like.
Methods of Use
[0130] The oral care compositions of the present invention can be used in the treatment, reduction, and/or prevention of caries, cavities, gingivitis, and/or combinations thereof.
[0131] The oral care compositions useful for the methods include hops, as described above, such as hops provided from a hops extract and/or an extract of Humulus lupulus. As described herein hops beta acid can be useful as an anticavity agent. Thus, the addition of hops to any oral care composition can provide anticavity protection.
[0132] The oral care composition can include primary packaging, such as a tube, bottle, and/or tub, The primary package can be placed within secondary package, such as a carton, shrink wrap, or the like. Instructions for use of the oral care composition can be printed on the primary package and/or the secondary package. The scope of the method is intended to include instructions provided by a manufacturer, distributor, and/or producer of the oral care composition.
[0133] If the oral care composition is a toothpaste, the user can be instructed to dispense the toothpaste from the toothpaste tube.
[0134] The user can be instructed. to apply a portion of the toothpaste onto a toothbrush. The portion of the toothpaste can be of any suitable shape, such as strip, a pea-sized amount, or various other shapes that would fit onto any mechanical and/or manual brush head. The user can be instructed to apply a strip of the toothpaste that is at least about 1 inch, at least about 0.5 inch, at least 1 inch, and/or at least 0.5 inch long to the bristles of a toothbrush, such as soft-bristled toothbrush.
[0135] The user can be instructed to apply pea-sized or grain of rice-sized portion of the toothpaste to the bristles of a toothbrush, such as in the case of use by children of less than 6 years old and/or less than 2 years old.
[0136] The user can be instructed to brush their teeth for at least about 30 seconds, at least about 1 minute, at least about 90 seconds, at least about 2 minutes, at least 30 seconds, at least 1 minute, at least 90 seconds, and/or at least 2 minutes.
[0137] The user can be instructed to brush their teeth thoroughly and/or as directed by a physician and/or dentist.
[0138] The user can be instructed to brush their teeth after each meal. The user can be instructed to brush their teeth at least once per day, at least twice per day, and/or at least three times per day. The user can be instructed to brush their teeth no more than three times a day, such as to prevent Sn staining. The user can be instructed to brush their teeth in the morning and/or in the evening prior to sleeping.
[0139] The user can be instructed to not swallow the toothpaste composition due to the inclusion of ingredients that are not suitable for ingestion, such as fluoride. However, in the case of an oral care composition comprising hops, but free of fluoride, the user may not need to be instructed to not swallow the toothpaste. The user may be instructed to expectorate (or spit out) the toothpaste composition after the cessation of the brushing cycle.
[0140] If the oral care composition is a mouth rinse, the user can be instructed to dispense the mouth rinse from a bottle containing the mouth rinse.
[0141] The user can be instructed to use the mouth rinse at least once a day, at least twice a day, and/or at least three times a day.
[0142] The user can be instructed to use the mouth rinse composition after the use of toothpaste and/or floss.
[0143] The user can be instructed to swish a portion of rinse in the oral cavity, such as between ti re teeth, for a period of time. The user can be instructed to vigorously swish a portion of the rinse.
[0144] The user can be instructed to use be from about 5 mL to about 50 mL, from about 10 mL to about 40 mL, 10 mL, 20 mL, 25 mL, 30 mL, 40 mL, 2 teaspoonfuls, and/or 4 teaspoonfuls of mouth rinse.
[0145] The user can be instructed to swish the mouth rinse for at least about 30 seconds, at least about 1 minute, at least about 90 seconds, at least about 2 minutes, at least 30 seconds, at least 1 minute, at least 90 seconds, and/or at least 2 minutes.
[0146] The user can be instructed to not swallow the mouth rinse composition due to the inclusion of ingredients that are not suitable for ingestion, such as fluoride. However, in the case of an oral care composition comprising hops, but free of fluoride, the user may not need to be instructed to not swallow the mouth rinse. The user may be instructed to expectorate (or spit out) the mouth rinse composition after the cessation of the rinse cycle.
[0147] The usage instructions for the oral care composition, such as for a toothpaste composition and/or a mouth rinse composition, can vary based on age. For example, adults and children that are at least 6 or at least 2 can have one usage instruction while children under 6 or under 2 can have a second usage instruction.
[0148] The oral care composition comprising hops, as described herein, can be useful as medicament, such as in an anticavity treatment, as described herein. Suitable medicaments include oral care compositions, toothpaste compositions, mouth rinse compositions, floss coatings, chewing gums, and/or other suitable compositions to be applied in the oral cavity.
[0149] Additionally, the oral care composition, as described herein, can be used to reduce the number and/or intensity of white spots on teeth, which can be attributable to caries presence with the oral cavity.
[0150] The methods described above can be useful for any oral care composition comprising hops, as described herein. The oral care composition comprising hops can also include other ingredients described above, such as metal, tin, zinc, fluoride, calcium, biofilm modifier, polyphosphate, abrasive, humectant, flavor, sweetener, prenylated flavonoid, thickening agent, buffering agent, and/or water. Additionally, the oral care compositions useful in the disclosed methods can be substantially free of, essentially free of, or free of fluoride.
COMBINATIONS
[0151] A. Hops beta acid for use as medicament, preferably wherein the medicament is an oral care composition, more preferred is a toothpaste, a mouth rinse, a floss coating, or combinations thereof. [0152] B. Hops beta acid as disclosed in A, wherein the hops beta acid comprises less than about 1%, by weight of the hops beta acid, of hops alpha acid, preferably wherein the hops beta acid is essentially free of, substantially free of, or more preferred free of hops alpha acid. [0153] C. Hops beta acid as disclosed in A or B, wherein the medicament comprises metal, preferably wherein the metal comprises tin, zinc, calcium, or combinations thereof, more preferably wherein the tin comprises stannous fluoride, stannous chloride, or combinations thereof. [0154] D. Hops beta acid as disclosed in any of A to C, wherein the medicament comprises abrasive, preferably wherein the abrasive comprises silica abrasive, calcium abrasive, or combinations thereof, more preferably wherein the calcium abrasive comprise calcium carbonate, calcium pyrophosphate, calcium phosphate, or combinations thereof. [0155] E. Hops beta acid as disclosed in any of A to D, wherein the medicament is substantially free of, essentially free of, or preferably free of fluoride. [0156] F. Hops beta acid as disclosed in any of A to E, for use in prevention, reduction and treatment of gingivitis, white spots in an oral cavity, oral cavities, caries, or a combination thereof, preferably wherein the white spots in the oral cavity are on teeth and attributable to caries, cavities, and/or combinations thereof. [0157] G. Hops for use in prevention, reduction and treatment of gingivitis, white spots in an oral cavity, oral cavities, caries, or a combination thereof, preferably wherein the white spots in the oral cavity are on teeth and attributable to caries, cavities, and/or combinations thereof. [0158] H. Hops as disclosed in G, wherein the hops comprises hops extract, preferably wherein the hops extract comprises hops beta acid, hops alpha acid, or combinations thereof, more preferably wherein the hops beta acid comprises less than about 1%, by weight of the extract, of hops alpha acid, more preferred wherein the hops is essentially free of, substantially free of, or free of hops alpha acid. [0159] I. Hops as disclosed in G or H, wherein the hops is provided in an oral care composition, preferably wherein the oral care composition is a toothpaste, mouth rinse, floss coating, or combinations thereof. [0160] J. Hops as disclosed in I, wherein the oral care composition further comprises an abrasive, preferably wherein the abrasive comprises silica abrasive, calcium abrasive, or combinations thereof, more preferably wherein the calcium abrasive comprise calcium carbonate, calcium pyrophosphate, calcium phosphate, or combinations thereof. [0161] K. Hops as disclosed in I or J, wherein the oral care composition further comprises a metal, preferably wherein the metal comprises tin, zinc, calcium, or combinations thereof, more preferably wherein the tin comprises stannous fluoride, stannous chloride, or combinations thereof. [0162] L. Hops as disclosed in any of I to K, wherein the oral care composition is substantially free of, essentially free of, or preferably free of fluoride. [0163] M. Hops as disclosed in any of Ito L; wherein: the user is instructed to use the oral care composition, preferably a toothpaste; wherein the instruction to use comprise: [0164] (a) Dispensing the oral care composition, preferably the toothpaste onto a toothbrush; [0165] (b) Applying the oral care composition, preferably the toothpaste, to the oral cavity for at least 1 minute; and [0166] (c) Expectorating the oral care composition, preferably the toothpaste, from the oral cavity. [0167] N. Hops as disclosed in M; wherein the instruction further comprises to not swallow the oral composition.
EXAMPLES
[0168] The invention is further illustrated by the following examples, which are not to be construed in any way as imposing limitations to the scope of this invention. Various other aspects, modifications, and equivalents thereof which, after reading the description herein, may suggest themselves to one of ordinary skill in the art without departing from the spirit of the present invention or the scope of the appended claims.
Toothpaste Composition Instruction Example 1
[0169] The oral care compositions described herein can include the following instructions:
[0170] Adults and children 12 years of age and older: Brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician.
[0171] Children under 12 years of age: ask a dentist
[0172] This instruction is placed on the carton containing the toothpaste composition including hops and/or on the toothpaste tube comprising the toothpaste composition including hops. The instruction can also include "Do not swallow."
[0173] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the toothpaste composition would instruct a user to perform these steps by placing the instructions on the carton, tube, and/or combinations thereof.
Toothpaste Composition Instruction Example 2
[0174] The oral care compositions described herein can include the following instructions:
[0175] Adults and children 12 years of age and older: [0176] Apply at least a 1-inch strip of the product onto a soft bristle toothbrush [0177] Brush teeth thoroughly for at least 1 minute twice a day (morning and evening), and not more than 3 times a day or as recommended by a dentist or doctor. Make sure to brush all areas of the teeth. Minimize swallowing. Spit out after brushing.
[0178] Children under 12 years of age: consult a dentist or doctor.
[0179] This instruction is placed on the carton containing the toothpaste composition including hops and/or on the toothpaste tube comprising the toothpaste composition including hops.
[0180] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the toothpaste composition would instruct a user to perform these steps by placing the instructions on the carton, tube, and/or combinations thereof.
Toothpaste Composition Instruction Example 3
[0181] The oral care compositions described herein can include the following instructions:
[0182] Adults and children 2 years of age and older: [0183] Brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician.
[0184] Children 2 to 6 years: [0185] Use only a pea sized amount and supervise child's brushing and rinsing (to minimize swallowing)
[0186] Children under 2 years: [0187] Ask a dentist or physician
[0188] This instruction is placed on the carton containing the toothpaste composition including hops and/or on the toothpaste tube comprising the toothpaste composition including hops. The instruction can also include "Do not swallow."
[0189] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the toothpaste composition would instruct a user to perform these steps by placing the instructions on the carton, tube, and/or combinations thereof.
Toothpaste Composition Instruction Example 4
[0190] The oral care compositions described herein can include the following instructions:
[0191] Adults and children 2 years of age and older: [0192] Brush teeth thoroughly after each meal or at least twice a day or use as directed by a dentist [0193] To minimize swallowing use a pea-sized amount in children under 6 [0194] Supervise children's brushing until good habits are established [0195] Use only a pea sized amount and supervise child's brushing and rinsing
[0196] Children under 2 years: [0197] Ask a dentist or physician
[0198] This instruction is placed on the carton containing the toothpaste composition including hops and/or on the toothpaste tube comprising the toothpaste composition including hops. The instruction can also include "Do not swallow."
[0199] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the toothpaste composition would instruct a user to perform these steps by placing the instructions on the carton, tube, and/or combinations thereof.
Toothpaste Composition Instruction Example 5
[0200] The oral care compositions described herein can include the following instructions:
[0201] Adults and children 12 years of age and older: [0202] Apply at least a 1-inch strip of the product onto a soft-bristled toothbrush. Brush teeth thoroughly for at least 1 minute twice a day (morning and evening) or as recommended by a dentist. [0203] Children under 12 years old: ask a dentist
[0204] This instruction is placed on the carton containing the toothpaste composition including hops and/or on the toothpaste tube comprising the toothpaste composition including hops. The instruction can also include "Do not swallow."
[0205] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the toothpaste composition would instruct a user to perform these steps by placing the instructions on the carton, tube, and/or combinations thereof.
Mouth Rinse Composition Instruction Example 1
[0206] The oral care compositions described herein can include the following instructions:
[0207] Adults and children 12 years of age and older: [0208] Use twice daily after brushing your teeth with a toothpaste [0209] Vigorously swish 10 mL (2 teaspoonfuls) of rinse between your teeth for 1 minute and then spit out. [0210] Do not eat or drink for 30 minutes after rinsing. [0211] Supervise children as necessary until capable of using without supervision.
[0212] This instruction is placed on the carton containing the mouth rinse composition including hops and/or on the mouth rinse bottle comprising the mouth rinse composition including hops. The instruction can also include "Do not swallow."
[0213] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the mouth rinse composition would instruct a user to perform these steps by placing the instructions on the carton, bottle, and/or combinations thereof.
Mouth Rinse Composition Instruction Example 2
[0214] The oral care compositions described herein can include the following instructions:
[0215] Adults and children 6 years of age and older: Use twice a day after brushing your teeth with a toothpaste. Vigorously swish 1.0 ml (2 teaspoonfuls) of rinse between your teeth for 1 minute and then spit out. Do not eat or drink for 30 minutes after rinsing. Instruct children under 12 years of age in good rinsing habits (to minimize swallowing). Supervise children as necessary until capable of using without supervision.
[0216] Children under 6 years of age: Consult a dentist or doctor.
[0217] This instruction is placed on the carton containing the mouth rinse composition including hops and/or on the mouth rinse bottle comprising the mouth rinse composition including hops. The instruction can also include "Do not swallow."
[0218] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the mouth rinse composition would instruct a user to perform these steps by placing the instructions on the carton, bottle, and/or combinations thereof.
Mouth Rinse Composition Instruction Example 3
[0219] The oral care compositions described herein can include the following instructions:
[0220] Use after your normal brushing and flossing routine; rinse toothpaste from mouth prior to use. Adults and children 6 yrs. & older: Rinse for 30 seconds with 20 mL (4 teaspoonfuls) twice a day. Children 6 years to under 12 years of age: supervise use. Children under 6 years of age: do not use.
[0221] This instruction is placed on the carton containing the mouth rinse composition including hops and/or on the mouth rinse bottle comprising the mouth rinse composition including hops. The instruction can also include "Do not swallow."
[0222] Thus, in total, a user would perform these steps while a manufacturer, distributor, and/or producer of the mouth rinse composition would instruct a user to perform these steps by placing the instructions on the carton, bottle, and/or combinations thereof.
Experimental Methods
Acid Production and Acid Inhibition [%]
[0223] Acid production and acid inhibition were determined with the in vitro plaque glycolysis model (iPGRM). The purpose of this technique is to provide a simple and quick method for determining if compounds have an influence on the metabolic pathways that plaque microorganisms utilize to produce toxins that adversely affect gingival health.
[0224] The in vitro plaque glycolysis model (iPGRM) is a technique in which plaque is grown from human saliva and treated with various agents to determine anti-glycolytic activity of treatments. When bacteria convert sugar into energy with the help of enzymes, acids are formed. These acids demineralize and damage the dental enamel. The purpose of this technique is to provide a simple and quick method for determining if treatment compounds have an inhibitory effect on the metabolic pathways that plaque microorganisms utilize for the production of acids or toxins and/or inhibit their growth. For the purposes of the work here, if the test therapeutic compositions contain Sn, the Sn placebo should be tested. Additionally, the antibacterial composition should be tested with respect to its placebo to determine the iPGRM value for the antibacterial composition only. This is important if buffers, e.g., bicarbonate, orthophosphate, calcium carbonate, are present in the composition in addition to the antibacterial composition.
[0225] A plaque biofilm was grown on glass rods from fresh pooled human saliva and Trypticase Soy Broth (TSB) at 37.degree. C. over 2 days by dipping glass rods into and out of media in a reciprocating motion. Treatments were 2 minutes of dentifrice slurry in water (1:5) or diluted treatment in water (1:5). After treatments, biofilms were incubated with TSB and sucrose until pH indicator showed a color change (.about.6 hrs). The pH of the media solutions was then measured to determine the amount of glycolysis inhibition relative to a negative control.
[0226] On Day 1, new glass rods (5 mm.times.90 mm) were polished approximately 25 mm from the un-tapered end on a lathe with silicon carbide paper of 240, 320, 400, and 600 grit used sequentially. After the initial polishing, the rods should be polished with 600 grit paper before each test. After polishing, rods were stored until ready to run test. Enough rods should be polished for a full rack of treatments. A rack can treat 12 compositions with 4 replicates of each composition such that the rack has 48 rods.
[0227] On Day 2, saliva was collected daily during the test from a panel of 5-10 people by paraffin stimulation and was refrigerated at 4.degree. C. until it was needed throughout the day. Pool saliva carefully (do not pour in wax/mucus) and mix thoroughly before use. The rods were removed from storage, rinsed with deionized water to remove any sanding residue, disinfected in 70% ethanol/water solution, and were allowed to dry on a sterile surface. Subsequently, the rods were loaded into a hanging rack of holders that were used to dip the rods continuously into media vials containing growth media. The rod heights were adjusted and each rod was secured in place using a rubber o-ring. In the early afternoon, 7 mL of growth media (160 g of a solution of 3% TSB with 3% sucrose was mixed with 240 g pooled human saliva. This TSB/sucrose solution should be sterilized by autoclave before combining with the pooled human saliva.) into media vials. The media vials were arranged under the hanging rods on a rack in an incubation oven. The incubator has been previously modified such that a dipping motor can dip the rods into the media vials submerging 1.5 cm of the rod into the growth media at a frequency of 1 dip per minute without the rods touching the walls of the media vial. The rods were dipped overnight this way.
[0228] On Day 3, an enriched growth media was prepared (500 g of a solution of 3% TSB and 10% sucrose was mixed with 33 g pooled human saliva. This TSB/sucrose solution should be sterilized by autoclave before combining with the pooled human saliva.). This enriched growth media was pipetted into a new set of media vials (7 mL per vial) and was swapped for the overnight growth media from Day 1. The rods were dipped throughout the day in this enriched growth media for 5 hours at 37.degree. C. in the incubation oven. At the end of the day, a new overnight growth media was prepared (40 g of a solution of 3% TSB was mixed with 360 g pooled human saliva and 0.5 g sucrose), pipetted into a new set of media vials, and swapped for the enriched growth media. The rods were dipped overnight in the same fashion as on the first day.
[0229] On Day 4, a glycolysis media was prepared by combining 0.15 g TSB, 25 g sucrose, and 500 mL deionized water resulting in a solution of 0.03% TSB and 0.5% sucrose in water. This solution was mixed then sterilized in an autoclave. The pH was then adjusted to 6.5 using 0.1 M HCl and pipetted into new media vials (7 mL). Two extra vials were filled than were needed for the rack of rods as pH blanks. Two drops of chlorophenol red solution were added to each of the 4 tubes that contained the negative control (Crest Cavity Protection slurry). Three drops of bromocresol purple solution were added to 2 tubes that contained the positive control (1% Chlorhexidine solution). Set the rack aside until treatments are complete. Vials were prepared containing 12 mL of deionized water to rinse off the treatments. Vials were prepared containing the treatment slurries/solutions (7 mL) of homogenized treatment and water. The rods were dipped into the treatment vials for 2 minutes, rinsed for 10 dips in a first set rinse vials, rinsed for 10 dips in a second set of rinse vials, rinsed for 10 dips in a third set of rinse vials, and returned to the incubator rack. The entire biofilm was treated and rinsed. Once all treatments were complete, the biofilms on the rods were fully submerged in the glycolysis media inside the incubation oven with no dipping for 2 hours. After two hours, the dipping apparatus was activated. The total incubation time was between 3 to 7 hours. Incubation is terminated when the pH value in the glycolysis media of the negative controls is between 4.8-5.6, more ideally 4.9-5.2, and when the pH value in the glycolysis media of the positive controls is above the negative control. If the indicator dye in the positive control turns yellow, i.e., the pH has dropped beneath 5.2, the incubation has gone on too long and the test will need to be repeated.
[0230] After incubation termination on Day 4, the rods were removed from the glycolysis media and allowed to dry in the oven. The glycolysis media was removed from the incubation oven, allowed to return to room temperature, and the pH was measured in each vial and the blank vials to determine the average pH change of the media following treatment. The change in pH is determined with respect to the blank vials. If the final pH of the blank is less than 6.6, the test needs to be repeated. If the difference between the positive and negative control is not significant in a student's t-test, the test needs to be repeated. If the change in pH of the negative control with respect to the blank is less than 1, the test needs to be repeated.
[0231] After the pH values of all the vials were measured, the .DELTA.pH per vial was determined by subtracting its pH from the average pH of the blanks. The glycolysis inhibition efficacy is determined from the following formula. The average .DELTA.pH of a treatment was determined by averaging the results from the four replicate vials per treatment.
Acid Inhibition ( % ) = 1 0 0 - ( Avg .DELTA. pH sample Avg .DELTA. pH neg ctrl ) .times. 1 0 0 Formula 2 ##EQU00001##
[0232] If the efficacy of the positive control (1% Chlorhexidine solution) is not between about 65% to about 85% with respect to the negative control (Crest Cavity Protection, Procter & Gamble, Cincinnati, Ohio), the test was repeated.
Anticaries Activity
[0233] The test design used here is similar to those found in the FDA Method #37 of the Fluoride Anti-Caries OTC Monograph. The major variations are the diet used (MIT 200 rather than #469), the caries score method (Keyes method rather than HMA), and treatment frequency. Experimental procedures were conducted according to the FDA regulations Part 58.
[0234] Using litters as a covariate, the use of between 50 and 58 (depending on the type of fluoride) animals per treatment group satisfies the most stringent power requirements of the ADA's Council on Dental Therapeutics 20% clinical difference between treatments at 80% power. However, we have been routinely using 40 animals per treatment group and both the ADA's CDT and the FDA have consistently accepted these tests. This requires initiating the study with 40 animals per group. Twenty-three (23) litters provided these animals. When studies are sized as such, treatment differences of approximately 16% have been found to be significant on occasion, thus is generally considered the cusp of clinical significance.
[0235] All protocols are reviewed and approved by the Institutional Animal Care and Use Committee prior to the receipt of animals.
[0236] The animals were weanling mixed-sex Sprague Dawley rats; weighing 29-53 grams. Due to the shipping schedule of the supplier, the dams were received with their entire birth litter. The litters were received when the pups were 6 days of age and litter size was reduced at 8 days of age to ten (10) pups per litter. Twenty-five (25) litters were purchased. The five extra litters were to allow for any mortality prior to stratification. Any unused animals were euthanized after the study stratification.
[0237] The litters were maintained in large solid-bottom (box-type) cages with dams until the pups were weaned at 18 days of age. Starting at 9 days of pup age, the dams were rotated daily among the litters until the pups were weaned at 18 days of age. The pups were maintained in the box cages until 21 days of age. At that time, the pups were stratified and housed in pairs in suspended wire-bottomed cages that had been cleaned and sanitized prior to usage. The change in caging was required to prevent artificially increasing the caries rate due to direct contact bedding. The cages were arranged so that all animals of the individual groups were together and the cages were labeled with group designation and treatment (treatment code) that the animals received.
[0238] When the pups were 21 days of age they were given unique numbers by ear-punch with records kept of littermates. Animals were assigned to groups in such a manner that groups were balanced for litter, weight and sex. There were 40 animals per group.
[0239] Upon receipt, dams and litters were provided rodent lab diet until the pups were 8 days of age. On day 8 (pup age) dams and litters were provided Diet MIT 305. Pups were provided Diet MIT 200 ad libitum at day 18 (pup age) and throughout the test period. All animals were provided with deionized water ad libitum.
[0240] Box caging was changed at day 13 and again at day 18 of pup age. Following administration of the inoculum, box cages and the bedding were decontaminated by autoclaving prior to sanitizing. Cage boards were changed three times a week at the time when fresh food and water were given (Monday, Wednesday and Friday). Clean and sanitized water bottles and food jars were provided weekly. Suspended caging and banks were sanitized bi-weekly. The animals were observed daily by a staff member and weekly by the attending veterinarian for any signs of health problems. The animals were housed in an AAALAC-accredited facility. Room temperature was maintained at 72.degree. F. (.+-.6.degree. F.) with 10-15 air changes per hour and a 12-hour light cycle.
[0241] On day 15 (pup age), the animals received an oral inoculation of streptomycin-resistant S. sobrinus 6715 (ATCC strain #27352) culture. This involved flooding the mouth with 0.2 ml of culture/animal. On day 18 (pup age) the animals were inoculated with S. sobrinus for three consecutive days (age 18, 19 and 20 days). This involved placing 0.1 ml of the S. sobrinus culture on the occlusal surfaces of each of the mandibular molar quadrants, putting 10 cc of this concentration-adjusted culture into each sanitized and filled water bottle, and lightly spraying the bedding with no more than 10 cc of the remaining culture. All water bottles were removed and sanitized 24 hours after inoculum has been added. The inoculums were administered to the animals with a 200 micropipette.
[0242] The treatment phase began at day 22 of pup age. Each treatment had a labeled plastic beaker that was designated for that treatment only. Fresh materials (i.e., obtained from the stock supply) were used for each treatment. The dentifrices were mixed in a 1:1 ratio (by weight) with deionized water. Specifically, 10 grams of dentifrice was weighed into a 30 ml beaker; 10 grams of deionized water was then added to the dentifrice. The mixture was then stirred by hand (30 seconds) with a clean micro spatula for the purpose of creating a smooth mixture. The beaker containing the slurry and a small magnetic stirring bar was placed on a magnetic stirrer, which was set at the lowest speed and allowed to stir for four (4) minutes prior to treatm4ent. The slurry was prepared immediately prior to each treatment.
[0243] A cotton-tipped applicator was dipped into the slurry (for 2 seconds) and was applied to one-half of the rat's mouth in such a way that the sides of the applicator came into contact with both the mandibular and maxillary molars on one side of the mouth. The treatment was accomplished by using a rolling motion of the sides of the applicator over the mandibular and maxillary molar teeth for 15 seconds. The applicator was dipped into the slurry for the second time (again, for 2 seconds) and the other side of the rat's mouth similarly treated for 15 seconds. A new applicator was used for each animal.
[0244] Treatments were administered twice daily for five days with a single daily treatment on weekends. The first treatment each day began at approximately the same time every day, and the second treatment did begin no earlier than six hours after the first treatment. Singular treatments were given at a 24-hour interval on weekends. Treatment materials were stored at room temperature. All treatment products were returned to sponsor at study completion.
[0245] One week after the initiation of the inoculation regimen and at study termination, an oral swabbing was taken from each rat using a sterile cotton swab (six-inch, single-tipped applicator). The microorganisms on the mandibular and maxillary molar teeth were sampled, using a rolling motion of the swab for 15 seconds on one side of the mouth, rolled over the tongue, and rolled over the molar teeth on the other side of the mouth for an additional 15 seconds. Immediately after the applicator was removed from the animal's mouth, it was streaked across half of a 100 mm petri plate containing Mitis Salivarius agar to which 200 units/ml of streptomycin sulfate was added. The plates were incubated for 48 hours at 37.degree. C. with 10% CO.sub.2. The colony count taken after the 48 hours of incubation was recorded in the logbook.
[0246] The experimental duration of the rat caries studies was three weeks. Immediately prior to termination, all animals were observed for any visual signs of ill health or pathology, individually weighed and an oral swabbing taken to confirm S. sobrinus implantation. The animals were euthanized by carbon dioxide inhalation. Code numbers were assigned to each animal and the heads were then removed, placed in individual jars along with the code number, and cooked under pressure (10 PSI for 12 minutes). The hemijaws were then removed and freed of all soft tissue.
[0247] The cleaned hemijaws (four quadrants) were put into plastic vials with the code numbers taped to the vial. A murexide solution (0.3 g murexide; 300 ml DI H2O and 700 ml of ethanol) was added to each vial and the jaws were allowed to stain overnight. The jaws were then rinsed and allowed to air dry.
[0248] The hemijaws were microscopically examined for smooth surface caries, sectioned, and then microscopically examined for sulcal and interproximal caries using the Keyes Method. The scoring method is detailed in Navia, J N, Animal Models in Dental Research, pp 287-290, 1977; and Keyes, PH, J. Dent. Res. 37:1088-1099, 1958. All analyses were performed using SAS statistical software, version 9.4. The groups were compared using analysis of variance (ANOVA), with a fixed effect for group and a random effect for litter. The litter effect was included in the models to reduce a known factor affecting the variability of the measurements. Pair-wise comparisons between groups were made using Tukey's multiple comparisons procedure to control the overall significance level (.alpha.=0.05) of the comparisons.
[0249] The specific types of data, which were tabulated, and statistically analyzed may include:
[0250] 1) Mortality Data Experimental Phase [0251] a. Initial number of animals [0252] b. Final number of animals [0253] c. Percent mortality
[0254] 2) Growth Data Experimental Phase [0255] a. Initial body weight (mean.+-.S.E.M.)
[0256] b. Final body weight (mean.+-.S.E.M.)
[0257] 3) Caries Experience [0258] a. Enamel and dentinal involvement of smooth surface (buccal, lingual) lesions (mean.+-.S.E.M.) [0259] b. Enamel and dentinal involvement of interproximal lesions (mean.+-.S.E.M.) [0260] c. Enamel and dentinal involvement of total smooth surface (buccal, lingual & interproximal) lesions (mean.+-.S.E.M.) [0261] d. Enamel and dentinal involvement of sulcal lesions (mean.+-.S.E.M.) [0262] e. Total caries involvement combining the scores from the Keyes method of scoring smooth surface, interproximal, and sulcal caries (mean.+-.S.E.M.)
Preparation of Oral Care Compositions
[0263] The oral care compositions of TABLE 1A were prepared by combining one or more humectants, water, sweetener(s), tin ion source, sodium gluconate, and/or flavor(s) to create a liquid mixture. The liquid mixture was homogenized at 25.degree. C. for 2 minutes. Next, sodium hydroxide (50% solution) was added to the liquid mixture and the liquid mixture was homogenized at 25.degree. C. for 2 minutes. A separate powder mixture was prepared by combining a portion of the calcium ion source and any thickening agents, such as xanthan gum and/or sodium carboxymethylcellulose. The powder mixture was then combined with the liquid mixture. Next, the surfactant, such as sodium lauryl sulfate, was added to the mixture. The contents were homogenized at 25.degree. C. for 2 minutes. The hops extract was then combined with the mixture and homogenized at 25.degree. C. for 2 minutes. Finally, the remaining portion of the calcium ion source and the buffering agent were combined with the mixture and homogenized at 25.degree. C. for 2 minutes.
TABLE-US-00001 TABLE 1A Oral Care Compositions Formula A Formula B Formula C Glycerin 49.10 47.80 -- Sorbitol -- -- 37.98 Water -- -- 13.00 Sodium Monofluorophosphate -- 1.15 -- Sodium Gluconate 1.00 1.00 1.00 SnCl.sub.2 1.10 1.10 1.10 CaCO.sub.3 32.00 32.00 32.00 Xanthan Gum 0.50 0.50 0.30 Carboxymethylcellulose -- -- 1.00 Carbomer 1.00 1.00 -- Sodium Lauryl Sulfate 1.40 1.40 1.29 Flavor 1.00 1.00 1.00 Sodium Saccharin 0.40 0.40 0.50 Stevia Glycosides 0.30 0.30 -- Sodium Hydroxide 1.20 1.35 0.33 Hops Beta Acid Extract* 0.50 0.50 0.50 Sodium Bicarbonate 10.00 10.00 10.00 Titanium Dioxide 0.50 0.50 -- *Hops Beta Acid Extract supplied by Hopsteiner .RTM., with 45% hops beta acids and less than 1% hops alpha acids
[0264] Formula A, B, and C, as shown in TABLE 1A, were prepared in accordance with the Experimental Methods, described above. The Hops Beta Acids were supplied by Hopsteiner.RTM. as an extract from Humulus lupulus. The Hopsteiner.RTM. extract was approximately 45%, by weight of the extract, of hops beta acids and less than 1%, by weight of the extract, of hops alpha acids. Formula A and B have hops beta acids with no independently added water. Formula A and B differ only in that Formula B includes a fluoride ion source (sodium monofluorophosphate, NaMFP), which was used in the total enamel caries test shown in TABLE 4. Formula C include hops beta acids without fluoride in a high-water chassis.
TABLE-US-00002 TABLE 1B Hops Beta Acids Extract Specification Ingredient Amount (wt %) Hops Beta Acids 45 .+-. 2 Hops Alpha Acids 0.4 .+-. 0.3 Hops oils 1.5 .+-. 0.5 Propylene Glycol 20 .+-. 15 Water <8% pH 11 .+-. 0.5
[0265] TABLE 1B describes the hops beta acid extract provided by Hopsteiner.RTM.. Since the hops beta acids are provided as an extract, there can be some variability in the amounts of certain ingredients. However, the extract comprises approximately 45%, by weight of the extract, of the hops beta acids and approximately 0.4%, by weight of the extract, of hops alpha acids. This is dramatically different to previous hops extracts which typically have more hops alpha acids than hops beta acids. Other minor ingredients may be present in the Hops Beta Acid extract.
TABLE-US-00003 TABLE 2 Acid Production and Glycolysis Inhibition [%] after treatment of 3-day biofilm Acid Production Mean Acid (.DELTA.pH) Inhibition (%) Treatment Mean SE Mean SE Crest .RTM. Cavity Protection (NaF) 1.55 0.027 3.43 1.68 Crest .RTM. Pro-Health .TM. Advanced (SnF.sub.2) 0.91 0.038 43.14 2.39 Crest .RTM. Gum Care (SnF.sub.2 + SnCl.sub.2) 0.51 0.025 68.49 1.55 1% Chlorhexidine 0.4 0.018 75.12 1.11 Formula A (Hops Beta Acids + SnCl.sub.2) -0.05 0.022 102.96 1.34 Formula C (Hops Beta Acids + SnCl.sub.2) 0 0.013 100.08 0.79
[0266] TABLE 2 displays the change in acid production in a 3-day biofilm using the iPGRM test described herein. After treatment with a commercial Crest.RTM. toothpaste containing sodium fluoride, the acid product mean is 1.55 with a mean inhibition of 3.43%. Treatment of the 3-day biofilm with a Crest.RTM. toothpaste containing stannous fluoride resulted in decreases in acid production and a net mean acid inhibition of 43.14% (Crest.RTM. Pro-Health.TM.) and 68.49% (Crest.RTM. Gum Care). This was an expected result as the stannous ion is known to act as an antibacterial, which can lower the number of bacteria producing acid in the biofilm. Crest.RTM. Gum Care performed slightly better than Crest.RTM. Pro-Health.TM. 0due to additional amounts of stannous ions (i.e. SnCl.sub.2). Chlorhexidine is an antibacterial agent that can be prescribed to treat gingivitis. Treatment with chlorhexidine resulted in a mean acid inhibition of 75.12%, which was not unexpected because chlorhexidine is known as an extremely effective antibacterial agent.
[0267] TABLE 2 also shows that Formula A and C displayed essentially zero acid production with a mean acid inhibition of approximately 100%. It was unexpected that Formula A and C (without a fluoride ion source) would be essentially 100% effective at preventing acid production without having fluoride and/or chlorhexidine, two of the most prevalent and effective antibacterial agents.
TABLE-US-00004 TABLE 3 Total Enamel Caries Total Enamel % Positive % Negative Treatment Caries Control Control Silica Placebo.sup.a 38.70 .+-. 2.06 47 0 Silica + NaMFP.sup.b 31.55 .+-. 2.08 75 19 Crest .RTM. Cavity 25.28 .+-. 1.99 100 35 Protection.sup.c Formula A 26.23 .+-. 2.01 96 32 .sup.aSilica placebo is the negative control without any fluoride ion source .sup.bNaMFP at 1000 ppm .sup.cNaF 1100 ppm + Silica is the positive control with silica and a fluoride ion source
[0268] TABLE 3 displays the results from the rat caries test, FDA No. 37. The rat caries test a biological test method required by the U.S. FDA monograph to show efficacy of an anticaries drug, currently including only fluoride ion sources. As shown in TABLE 3, the negative control, Silica Placebo, is a toothpaste with silica abrasive, but not any fluoride ion source. Silica+NaMFP is a USP NaMFP toothpaste with sodium monofluorophosphate and a silica abrasive. Crest.RTM. Cavity Protection is a toothpaste with sodium fluoride and a silica abrasive. In contrast, Formula A has no fluoride ion source or silica abrasive, but has hops beta acids and stannous chloride. Unexpectedly, Formula A (26.23), without any fluoride ion source, performed similar to the Crest.RTM. Cavity Protection (25.28), a dentifrice with 1100 ppm sodium fluoride. Additionally, Formula A performed better than a dentifrice with sodium monofluorophosphate with silica abrasive (26.23 for Formula A vs. 31.55 for NaMFP). The only anticaries drugs listed in the U.S. FDA monograph are stannous fluoride, sodium fluoride, and sodium monofluorophosphate. In other words, Formula A is a fluoride-free anticavity toothpaste.
[0269] The dimensions and values disclosed herein are not to be understood as being strictly limited to the exact numerical values recited. Instead, unless otherwise specified, each such dimension is intended to mean both the recited value and a functionally equivalent range surrounding that value. For example, a dimension disclosed as "40 mm" is intended to mean "about 40 mm."
[0270] Every document cited herein, including any cross referenced or related patent or application and any patent application or patent to which this application claims priority or benefit thereof, is hereby incorporated herein by reference in its entirety unless expressly excluded or otherwise limited. The citation of any document is not an admission that it is prior art with respect to any invention disclosed or claimed herein or that it alone, or in any combination with any other reference or references, teaches, suggests or discloses any such invention. Further, to the extent that any meaning or definition of a term in this document conflicts with any meaning or definition of the same term in a document incorporated by reference, the meaning or definition assigned to that term in this document shall govern.
[0271] While particular embodiments of the present invention have been illustrated and described, it would be obvious to those skilled in the art that various other changes and modifications can be made without departing from the spirit and scope of the invention. It is therefore intended to cover in the appended claims all such changes and modifications that are within the scope of this invention.
* * * * *









XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.