Fluid Handling System
Schenck; Alan ; et al.
U.S. patent application number 17/107500 was filed with the patent office on 2021-03-18 for fluid handling system. The applicant listed for this patent is TC1 LLC. Invention is credited to Peter W. Bristol, Keif M. Fitzgerald, Michael L. Green, Richard L. Keenan, Paul C. Leonard, Jeffrey Paul Mills, Paul F. Muller, Alan Schenck, Keith Schubert, Joseph P. Sullivan.
| Application Number | 20210077690 17/107500 |
| Document ID | / |
| Family ID | 1000005240246 |
| Filed Date | 2021-03-18 |








| United States Patent Application | 20210077690 |
| Kind Code | A1 |
| Schenck; Alan ; et al. | March 18, 2021 |
FLUID HANDLING SYSTEM
Abstract
A system for coupling a motor assembly to a control console is disclosed. The motor assembly is configured to drive a percutaneous heart pump. The system includes a motor coupling comprising a first end having an electrical connection and a fluid connection coupled to a respective electrical conduit and fluid conduit of the motor assembly, and a second end spaced apart from the first end including an interface coupling coupled to the electrical conduit and fluid conduit. An interface member is configured to be removably coupled to the control console, the interface member comprising a corresponding connector coupleable to the interface coupling of the second end of the motor coupling, wherein the interface member is configured to provide a sterile attachment of the fluid conduit to the control console.
| Inventors: | Schenck; Alan; (Sunnyvale, CA) ; Green; Michael L.; (Pleasanton, CA) ; Fitzgerald; Keif M.; (San Jose, CA) ; Muller; Paul F.; (San Carlos, CA) ; Sullivan; Joseph P.; (Issaquah, WA) ; Schubert; Keith; (Redmond, WA) ; Bristol; Peter W.; (Seattle, WA) ; Mills; Jeffrey Paul; (Barrington, IL) ; Leonard; Paul C.; (Woodinville, WA) ; Keenan; Richard L.; (Livermore, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005240246 | ||||||||||
| Appl. No.: | 17/107500 | ||||||||||
| Filed: | November 30, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16849700 | Apr 15, 2020 | |||
| 17107500 | ||||
| 15198342 | Jun 30, 2016 | 10632241 | ||
| 16849700 | ||||
| 14203978 | Mar 11, 2014 | 9381288 | ||
| 15198342 | ||||
| 61780656 | Mar 13, 2013 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61M 60/857 20210101; A61M 1/3659 20140204; A61M 60/871 20210101; A61M 60/135 20210101; A61M 60/414 20210101; A61M 5/142 20130101; A61M 60/148 20210101; A61M 5/172 20130101; A61M 60/50 20210101; A61M 60/205 20210101 |
| International Class: | A61M 1/12 20060101 A61M001/12; A61M 1/10 20060101 A61M001/10; A61M 1/36 20060101 A61M001/36; A61M 5/142 20060101 A61M005/142; A61M 5/172 20060101 A61M005/172 |
Claims
1. A system for coupling a motor assembly to a control console, the motor assembly configured to drive a percutaneous heart pump, the system comprising: a motor coupling comprising a first end having an electrical connection and a fluid connection coupled to a respective electrical conduit and fluid conduit of the motor assembly, and a second end spaced apart from the first end including a interface coupling coupled to the electrical conduit and fluid conduit; an interface member configured to be removably coupled to the control console, the interface member comprising a corresponding connector coupleable to the interface coupling of the second end of the motor coupling, wherein the interface member is configured to provide a sterile attachment of the fluid conduit to the control console.
2. The system according to claim 1, wherein the interface member is configured to provide a simultaneous connection of the fluid conduit and the electrical conduit to the control console.
3. The system according to claim 1, wherein the interface member further comprises a locking mechanism configured to lock the interface member to the control console such that the fluid conduit and electrical conduit of the motor assembly are releasably secured to the control console.
4. The system according to claim 1, wherein the interface member is configured to place the fluid conduit in communication with a roller pump of the control console when coupled to the control console.
5. The system according to claim 1, wherein the motor assembly includes a driving assembly and a driven assembly, and wherein the electrical conduit is coupled to the driving assembly and the fluid conduit is coupled to the driven assembly.
6. The system according to claim 1, wherein the interface member comprises a plurality of electrical contacts configured to engage a respective plurality of electrical console contacts, at least one of the electrical contacts of the interface member configured to receive electrical power to drive the motor assembly.
7. The system according to claim 1, wherein the electrical conduit includes at least one conductor for carrying a signal between the interface member and a temperature sensor.
8. The system according to claim 1, wherein the motor assembly includes a driving assembly and a driven assembly, and wherein the driven assembly is configured to power an impeller of a percutaneous heart pump.
9. The system according to claim 1, wherein the interface member further comprises a locking mechanism configured to lock the fluid conduit and electrical conduit of the motor assembly to the control console, and wherein the locking mechanism includes a manually actuatable release to unlock the fluid conduit and electrical conduit from the control console.
10. The system according to claim 1, wherein the electrical conduit includes at least one conductor for providing instructions to the motor assembly.
11. The system according to claim 10, wherein the electrical conduit further comprises at least one conductor for transmitting a signal between a fluid sensor that measures at least one parameter of a fluid within the fluid conduit and the control console.
12. The system according to claim 1, wherein the interface member provides a fluid tight connection to the interface coupling.
13. A method of coupling a motor assembly to a control console of a percutaneous heart pump, the method comprising: coupling a motor coupling to an interface member, the motor coupling comprising a first end having an electrical connection and a fluid connection coupled to a respective electrical conduit and fluid conduit of the motor assembly, and a second end spaced apart from the first end including an interface coupling coupled to the electrical conduit and fluid conduit; coupling the interface member to the control console, wherein the the interface member is configured to provide a sterile attachment of the fluid conduit to the control console, and coupling the interface member to the control console provides electrical power to the motor assembly via the electrical conduit.
14. The method according to claim 13, wherein coupling the interface member to the control console further comprises simultaneously connecting the fluid conduit and the electrical conduit to the control console.
15. The method according to claim 13, wherein coupling the interface member to the control console further comprises placing the fluid conduit in communication with a roller pump of the control console.
16. The method according to claim 13, wherein coupling the interface member to the control console further comprises connecting a sensor conductor of the electrical conduit in electronic communication with the control console, the sensor carrying a signal between the interface member and a temperature sensor.
17. The method according to claim 13, wherein coupling the interface member to the control console forms a fluid tight seal.
18. The method according to claim 13, wherein coupling the interface member to the control console further comprises coupling at least one conductor of the electrical conduit in electronic communication with the control console, the at least one conductor for providing instructions to the motor assembly from the control console.
19. The method according to claim 13, wherein coupling the interface member to the control console further comprises placing at least one conductor of the electrical conduit in electronic communication with the control console, the at least one conductor for transmitting a signal between a fluid sensor that measures at least one parameter of a fluid within the fluid conduit and the control console.
20. The method according to claim 13, wherein coupling the interface member to the control console further comprises using a locking mechanism to lock the interface member to the control console such that the electrical conduit and the fluid conduit remain connected to the control console until a manually actuable release is activate to release the interface member from the control console.
Description
INCORPORATION BY REFERENCE TO ANY PRIORITY APPLICATIONS
[0001] This application is a Continuation of U.S. patent application Ser. No. 16/849,700 filed on Apr. 15, 2020, which is a Continuation of U.S. patent application Ser. No. 15/198,342 filed on Jun. 30, 2016, now issued U.S. Pat. No. 10,632,241, which is a Divisional of U.S. patent application Ser. No. 14/203,978, filed Mar. 11, 2014, now issued U.S. Pat. No. 9,381,288, which claims the benefit of priority to U.S. Provisional Patent Application No. 61/780,656, filed Mar. 13, 2013, each of which is hereby incorporated herein by reference in its entirety.
BACKGROUND OF THE INVENTION
Field of the Invention
[0002] This application is directed to pumps for mechanical circulatory support of a heart. In particular, this application is directed to a console and controller for a catheter pump and a fluid handling system configured to convey and remove fluids to and from the catheter pump.
Description of the Related Art
[0003] Heart disease is a major health problem that has high mortality rate. Physicians increasingly use mechanical circulatory support systems for treating heart failure. The treatment of acute heart failure requires a device that can provide support to the patient quickly. Physicians desire treatment options that can be deployed quickly and minimally-invasively.
[0004] Intra-aortic balloon pumps (IABP) are currently the most common type of circulatory support devices for treating acute heart failure. IABPs are commonly used to treat heart failure, such as to stabilize a patient after cardiogenic shock, during treatment of acute myocardial infarction (MI) or decompensated heart failure, or to support a patient during high risk percutaneous coronary intervention (PCI). Circulatory support systems may be used alone or with pharmacological treatment.
[0005] In a conventional approach, an IABP is positioned in the aorta and actuated in a counterpulsation fashion to provide partial support to the circulatory system. More recently, minimally-invasive rotary blood pumps have been developed in an attempt to increase the level of potential support (i.e., higher flow). A rotary blood pump is typically inserted into the body and connected to the cardiovascular system, for example, to the left ventricle and the ascending aorta to assist the pumping function of the heart. Other known applications pumping venous blood from the right ventricle to the pulmonary artery for support of the right side of the heart. An aim of acute circulatory support devices is to reduce the load on the heart muscle for a period of time, to stabilize the patient prior to heart transplant or for continuing support.
[0006] There is a need for improved mechanical circulatory support devices for treating acute heart failure. Fixed cross-section ventricular assist devices designed to provide near full heart flow rate are either too large to be advanced percutaneously (e.g., through the femoral artery without a cutdown) or provide insufficient flow.
[0007] There is a need for a pump with improved performance and clinical outcomes. There is a need for a pump that can provide elevated flow rates with reduced risk of hemolysis and thrombosis. There is a need for a pump that can be inserted minimally-invasively and provide sufficient flow rates for various indications while reducing the risk of major adverse events. In one aspect, there is a need for a heart pump that can be placed minimally-invasively, for example, through a 15FR or 12FR incision. In one aspect, there is a need for a heart pump that can provide an average flow rate of 4 Lpm or more during operation, for example, at 62 mmHg of head pressure. While the flow rate of a rotary pump can be increased by rotating the impeller faster, higher rotational speeds are known to increase the risk of hemolysis, which can lead to adverse outcomes and in some cases death. Accordingly, in one aspect, there is a need for a pump that can provide sufficient flow at significantly reduced rotational speeds. These and other problems are overcome by the inventions described herein.
[0008] Furthermore, in various catheter pump systems, it can be important to provide fluids to an operative device of a catheter assembly (e.g., for lubrication of moving parts and/or treatment fluids to be delivered to the patient), and to remove waste fluids from the patient's body. A controller may be provided to control the flow into and out of the catheter assembly. It can be advantageous to provide improved mechanisms for engaging the catheter assembly with the controller, which may be housed in a console.
[0009] Additionally, there is a need to reduce the time to implantation and treatment. In the case of therapy for acute heart failure in particular, the time it takes to start therapy can be critical to survival and good outcomes. For example, a difference of several minutes can be the difference between recovery and permanent brain damage for patients suffering myocardial infarction or cardiogenic shock. Accordingly, a continuing need exists to provide pump systems that can be set up, primed, and inserted faster, easier, and more effectively.
[0010] These and other problems are overcome by the inventions described herein.
SUMMARY
[0011] There is an urgent need for a pumping device that can be inserted percutaneously and also provide full cardiac rate flows of the left, right, or both the left and right sides of the heart when called for.
[0012] In one embodiment, a system for priming a catheter assembly is disclosed. The system can include a catheter assembly including an elongate body having a distal portion and an operative device coupled at the distal portion. The system can also include a priming vessel configured to receive insertion of the operative device therein. The priming vessel can include a proximal portion secured to the distal portion of the elongate body such that the elongate body is in fluid communication with the priming vessel. The priming vessel can also include a distal end through which air is expelled when a fluid is channeled through the elongate body and into the priming vessel to expel air from within the catheter assembly.
[0013] In another embodiment, an infusate system for priming a catheter assembly is disclosed. The system includes a catheter assembly including an elongate body and an operative device, a fluid reservoir configured to store an infusate fluid, and a luer coupled in flow communication with the fluid reservoir. The infusate system also includes a priming vessel configured to receive insertion of the operative device therein. The priming vessel is coupled in flow communication with the elongate body and the luer such that the luer is configured to deliver the infusate fluid to the priming apparatus to expel air from the catheter assembly.
BRIEF DESCRIPTION OF THE DRAWINGS
[0014] A more complete appreciation of the subject matter of this application and the various advantages thereof can be realized by reference to the following detailed description, in which reference is made to the accompanying drawings in which:
[0015] FIG. 1 is a schematic view of an operative device of a catheter assembly in position within the anatomy for assisting the left ventricle.
[0016] FIG. 2 is a three-dimensional perspective view of a catheter assembly, according to some embodiments.
[0017] FIG. 3A is a three-dimensional perspective view of a fluid handling system that includes a console and catheter assembly.
[0018] FIG. 3B is a three-dimensional perspective view of an interface region of the console shown in FIG. 3A.
[0019] FIG. 4 is a three-dimensional perspective view of an interface member, according to one embodiment.
[0020] FIG. 5A is a three-dimensional perspective view of a cap.
[0021] FIG. 5B is a three-dimensional perspective view of an interface member in an unlocked configuration.
[0022] FIG. 5C is a three-dimensional perspective view of an interface member in a locked configuration.
[0023] FIG. 6A is a three-dimensional perspective view of a first side of an electrical component, according to one embodiment.
[0024] FIG. 6B is a three-dimensional perspective view of a second, opposite side of the electrical component of FIG. 6A.
[0025] FIG. 7 is a schematic diagram of an infusate system, according to one embodiment.
[0026] FIG. 8 is an enlarged view of a priming apparatus shown in FIG. 2.
[0027] More detailed descriptions of various embodiments of components for heart pumps useful to treat patients experiencing cardiac stress, including acute heart failure, are set forth below.
DETAILED DESCRIPTION
[0028] This application is directed to fluid handling systems that are configured to control and/or manage fluid and electrical pathways in a catheter assembly, such as a catheter assembly of a percutaneous heart pump system. In particular, the disclosed percutaneous heart pump systems may include a catheter assembly and a console that includes a controller configured to control the fluid and electrical pathways that pass through the catheter assembly. Some of the disclosed embodiments generally relate to various configurations for coupling and engaging the catheter assembly with the console. For example, the console may be configured to control the flow rate of the pump and to monitor various physiological parameters and pump performance through the various electrical and fluid pathways of the catheter assembly. In some arrangements, the catheter assembly may be disposable, such that the catheter assembly can be discarded after use, while the console and controller are reusable. In embodiments with a reusable console and a disposable catheter assembly (or, indeed, in any embodiments where consoles and catheter assemblies may be coupled), it can be desirable to provide an effective interface between the catheter assembly and the console that completes the various fluid and electrical connections between the catheter assembly and the console.
[0029] In particular, it can be advantageous to provide an interface member at a proximal portion of the catheter assembly that is removably engageable with the console. Furthermore, to enhance usability and to minimize mistakes in making the connections, it can be important to make the interface easy to use so that users can easily connect the catheter assembly to the console before use and easily remove the catheter assembly from the console after use. Moreover, it can be important that the interface provides a secure connection between the interface member of the catheter assembly and an interface region of the console to ensure that the catheter assembly remains connected to the console uninterrupted during treatment.
[0030] As explained herein, one example of a catheter assembly is used in a percutaneous heart pump system having an operative device (e.g., an impeller assembly) that is configured to assist the patient's heart in pumping blood. The heart pump system may be configured to at least temporarily support the workload of the left ventricle in some embodiments. The exemplary heart pump can be designed for percutaneous entry through the femoral artery to a patient's heart. In particular, the exemplary impeller assembly can include a collapsible impeller and cannula, which can be inserted into the patient's vasculature at a catheter size of less than 13 FR, for example, about 12.5 FR in some arrangements. During insertion through the patient's vascular system to the heart, a sheath may maintain the impeller and cannula assembly in a stored configuration. When the impeller assembly is positioned in the left ventricle (or another chamber of a patient's heart), the impeller and cannula can expand to a larger diameter, for example to a catheter size of about 24 FR when the sheath is removed from the impeller assembly. The expanded diameter of the impeller and cannula may allow for the generation of higher flow rates, according to some embodiments.
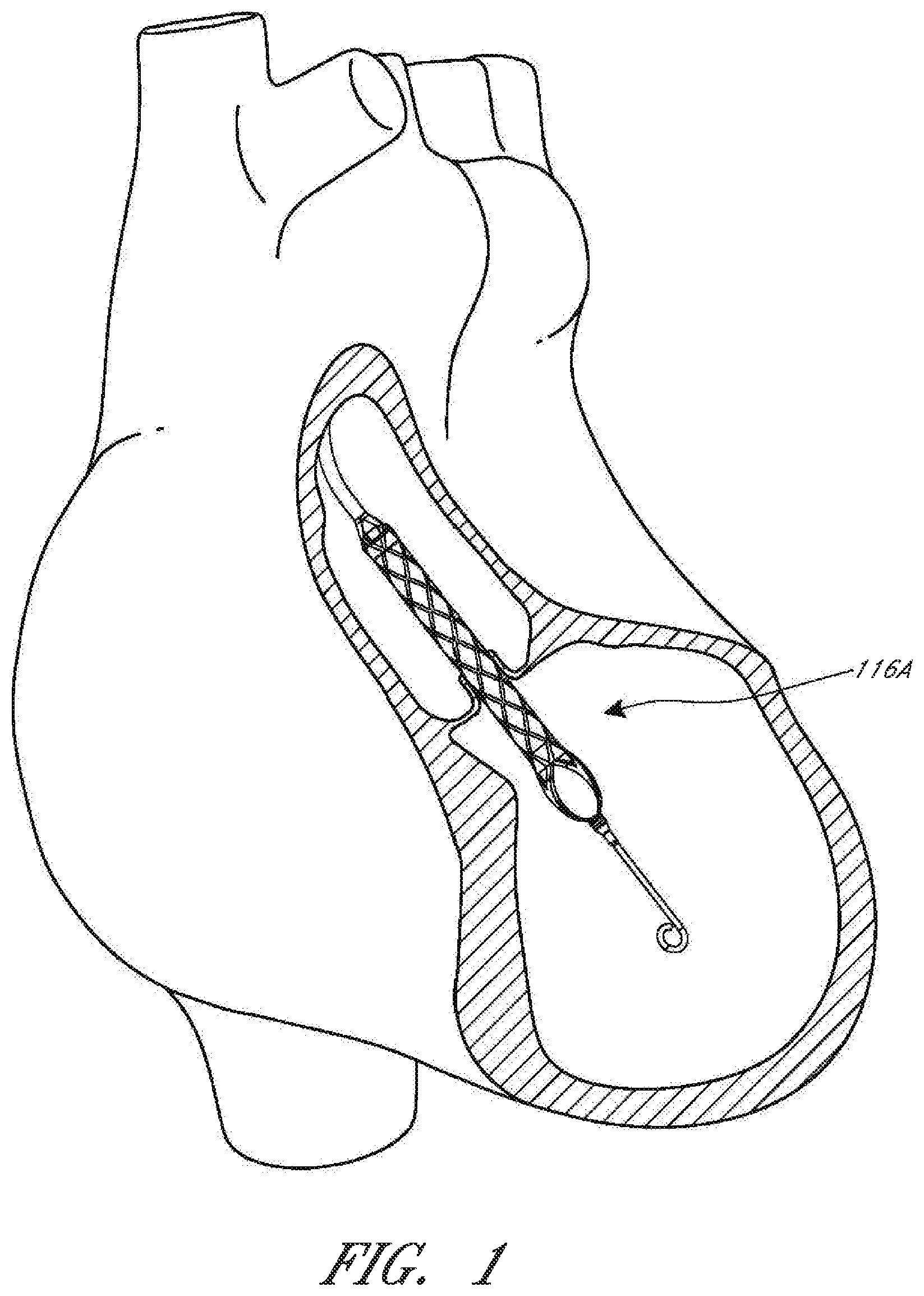
[0031] For example, FIG. 1 illustrates one use of the disclosed catheter pump system. A distal portion of the pump, which can include an impeller assembly 116A, is placed in the left ventricle (LV) of the heart to pump blood from the LV into the aorta. The pump can be used in this way to treat patients with a wide range of conditions, including cardiogenic shock, myocardial infarction, and other cardiac conditions, and also to support a patient during a procedure such as percutaneous coronary intervention. One convenient manner of placement of the distal portion of the pump in the heart is by percutaneous access and delivery using the Seldinger technique, or other methods familiar to cardiologists. These approaches enable the pump to be used in emergency medicine, a catheter lab and in other non-surgical settings. Modifications can also enable the pump 10 to support the right side of the heart. Example modifications that could be used for right side support include providing delivery features and/or shaping a distal portion that is to be placed through at least one heart valve from the venous side, such as is discussed in U.S. Pat. Nos. 6,544,216; 7,070,555; and US 2012-0203056A1, all of which are hereby incorporated by reference herein in their entirety for all purposes.
[0032] Turning to FIG. 2, a three-dimensional perspective view of a catheter assembly 100A is disclosed. The catheter assembly 100A may correspond to the disposable portion of the heart pump systems described herein. For example, the catheter assembly 100A may include the impeller assembly 116A near a distal portion of the catheter assembly 100A, an elongate body 174A extending proximally from the impeller assembly 116A, an infusion system 195 configured to supply infusate to the catheter assembly 100A, a motor assembly comprising a driven assembly 101 and a drive assembly 103, one or more conduits 302 (e.g., electrical and/or fluid conduits) extending proximally from the motor assembly, and an interface member 300 coupled at a proximal portion of the conduits 302.
[0033] Moving from the distal end of the catheter assembly 100A of FIG. 2 to the proximal end, the impeller assembly 116A may be disposed at a distal portion of the catheter assembly 100A. As explained above, the impeller assembly 116A can include an expandable cannula or housing and an impeller with one or more blades. As the impeller rotates, blood can be pumped proximally (or distally in some implementations) to function as a cardiac assist device. A priming apparatus 1400 can be disposed over the impeller assembly 116A. As explained herein with reference to FIGS. 7-8, the priming apparatus 1400 can be configured to expedite a process of expelling air from the catheter assembly 100A before insertion of the operative device of the catheter assembly into the patient.
[0034] With continued reference to FIG. 2, the elongate body 174A extends proximally from the impeller assembly 116A to an infusion system 195 configured to allow infusate to enter the catheter assembly 100A and waste fluid to leave the catheter assembly 100A. A catheter body 120A (which also passes through the elongate body 174A) can extend proximally and couple to the driven assembly 101 of the motor assembly. The catheter body 120A can pass within the elongate body 174A, such that the elongate body 174A can axially translate relative to the catheter body 120A. Axial translation of the elongate body 174A relative to the catheter body 120A can enable the expansion and collapse of the impeller assembly 116A. For example, the impeller assembly 116A, coupled to a distal portion of the catheter body 120A, may expand into an expanded state by moving the elongate body 174A proximally relative to the impeller assembly 116A. The impeller assembly 116A may self-expand into the expanded state in some embodiments. In the expanded state, the impeller assembly 116A is able to pump blood at high flow rates. After the treatment procedure, the impeller assembly 116A may be compressed into a collapsed state by advancing a distal portion 170A of the elongate body 174A distally over the impeller assembly 116A to cause the impeller assembly 116A to collapse.
[0035] As explained above, the catheter body 120A can couple to the driven assembly 101 of the motor assembly. The driven assembly 101 can be configured to receive torque applied by the drive assembly 103, which is shown as being decoupled from the driven assembly 101 and the catheter assembly 100A in FIG. 2. The drive assembly 103 can be coupled to the driven assembly 101 by engaging a proximal portion of the driven assembly 101 with the drive assembly, e.g., by inserting the proximal portion of the driven assembly 101 into an aperture 105 of the drive assembly 103.
[0036] Although not shown in FIG. 2, a drive shaft can extend from the driven assembly 101 through the catheter body 120A to couple to an impeller shaft at or proximal to the impeller assembly 116A. The drive assembly 103 can electrically communicate with a controller in a console (see, e.g., FIGS. 3A-3B), which can be configured to control the operation of the motor assembly and the infusion system 195 that supplies a flow of infusate in the catheter assembly 100A. The impeller of the impeller assembly 116A may thus be rotated remotely by the motor assembly during operation of the catheter pump in various embodiments. For example, the motor assembly can be disposed outside the patient. In some embodiments, the motor assembly is separate from the controller or console, e.g., to be placed closer to the patient. In other embodiments, the motor assembly is part of the controller. In still other embodiments, the motor assembly is miniaturized to be insertable into the patient. Such embodiments allow the drive shaft to be much shorter, e.g., shorter than the distance from the aortic valve to the aortic arch (about 5 cm or less). Some examples of miniaturized motors catheter pumps and related components and methods are discussed in U.S. Pat. Nos. 5,964,694; 6,007,478; 6,178,922; and 6,176,848, all of which are hereby incorporated by reference herein in their entirety for all purposes.
[0037] As shown in FIG. 2, the motor assembly (e.g., the drive assembly 103 and the driven assembly 101) is in electrical communication with the controller and console by way of the conduits 302, which may include electrical wires. In particular, as shown in FIG. 2, the electrical wires may extend from the motor assembly proximally to the interface member 300. To enable the controller in the console to electrically communicate with the motor assembly and/or other sensors in the catheter assembly 100A (such as pressure sensors, flow sensors, temperature sensors, bubble detectors, etc.), it can be advantageous to provide a reliable electrical connection between the interface member 300 and the console. In various embodiments disclosed herein, therefore, the removable interface member 300 may include electrical components configured to couple to one or more electrical contacts (sometimes referred to herein as interconnections) in the console. The electrical connections may be achieved in a simple, user-friendly manner. In various embodiments disclosed herein, for example, the electrical connections may be made substantially at the same time, e.g., substantially simultaneously, as fluid connections are made between the interface member 300 and console. These and other structures incorporated to reduce the complexity of operating the pump system are provided to reduce the chance of errors in set-up and delays, which for the emergency conditions in which the pump may be implemented could be life-threatening.
[0038] The mechanical components rotatably supporting the impeller within the impeller assembly 116A permit high rotational speeds while controlling heat and particle generation that can come with high speeds. The infusion system 195 may deliver a cooling and lubricating solution to the distal portion of the catheter assembly 100A for these purposes. As shown in FIG. 2, the infusion system 195 may be in fluid communication with the interface member 300 by way of the conduits 302, which may also include fluid conduits or tubes. Because the catheter assembly 100A may be disposable and/or removable from a console, it can be important to securely couple interface member 300 to the console. Furthermore, it can be important to provide an easy-to-use interface such that users can easily complete fluid connections that remain secure during a treatment procedure. Maintaining security of the connection is important because the fluids and signals carried by the conduits 302 enable the impeller to operate in a continuous manner. Stoppage of the pump system may require the catheter assembly 100A to be removed from the patient and replaced in certain circumstances, which may be life-threatening or extremely inconvenient at a minimum.
[0039] When activated, the catheter pump system can effectively increase the flow of blood out of the heart and through the patient's vascular system. In various embodiments disclosed herein, the pump can be configured to produce a maximum flow rate (e.g. low mm Hg) of greater than 4 Lpm, greater than 4.5 Lpm, greater than 5 Lpm, greater than 5.5 Lpm, greater than 6 Lpm, greater than 6.5 Lpm, greater than 7 Lpm, greater than 7.5 Lpm, greater than 8 Lpm, greater than 9 Lpm, or greater than 10 Lpm. In various embodiments, the pump can be configured to produce an average flow rate at 62 mmHg pressure head of greater than 2 Lpm, greater than 2.5 Lpm, greater than 3 Lpm, greater than 3.5 Lpm, greater than 4 Lpm, greater than 4.25 Lpm, greater than 4.5 Lpm, greater than 5 Lpm, greater than 5.5 Lpm, or greater than 6 Lpm.
[0040] Various aspects of the pump and associated components are similar to those disclosed in U.S. Pat. Nos. 7,393,181; 8,376,707; 7,841,976; 7,022,100; and 7,998,054, and in U.S. Pub. Nos. 2011/0004046; 2012/0178986; 2012/0172655; 2012/0178985; and 2012/0004495, the entire contents of each of which are incorporated herein for all purposes by reference. In addition, this application incorporates by reference in its entirety and for all purposes the subject matter disclosed in each of the following concurrently filed applications: application Ser. No. 13/802,556, which corresponds to attorney docket no. THOR.072A, entitled "DISTAL BEARING SUPPORT," filed on Mar. 13, 2013; application Ser. No. 13/801,833, which corresponds to attorney docket no. THOR.089A, entitled "SHEATH SYSTEM FOR CATHETER PUMP," filed on Mar. 13, 2013; application Ser. No. 13/802,570, which corresponds to attorney docket no. THOR.090A, entitled "IMPELLER FOR CATHETER PUMP," filed on Mar. 13, 2013; application Ser. No. 13/801,528, which corresponds to attorney docket no. THOR.092A, entitled "CATHETER PUMP," filed on Mar. 13, 2013; and application Ser. No. 13/802468, which corresponds to attorney docket no. THOR.093A, entitled "MOTOR ASSEMBLY FOR CATHETER PUMP," filed on Mar. 13, 2013.
Fluid Handling System
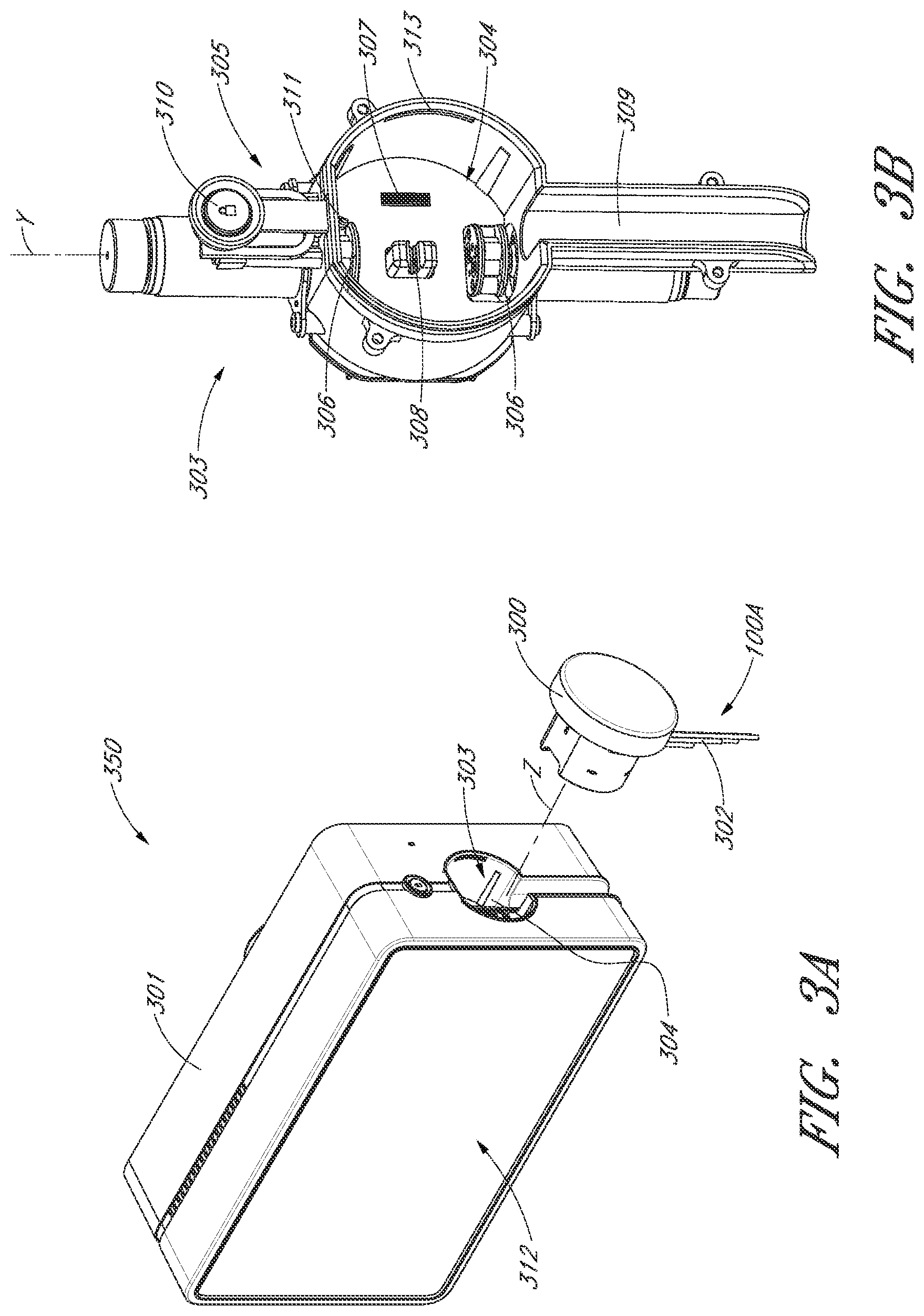
[0041] FIG. 3A is a three-dimensional perspective view of a fluid handling system 350 that includes a console 301 and the catheter assembly 100A of FIG. 2. The console 301 can provide electrical power, control signals, medical fluids (e.g., saline) for infusion, and fluid waste extraction to the catheter assembly 100A by way of its interface with the interface member 300. In this manner, a plurality of fluid connections can advantageously be made with a single interface. As illustrated in FIG. 2, for example, the removable interface member 300 may be disposed at a proximal portion of the catheter assembly 100A and may be configured to couple to the console 301 at an interface region 303.
[0042] In some embodiments, the fluid handling system 350 can be configured to deliver fluids to and/or remove fluids from the catheter assembly 100A. As discussed above and in the incorporated patent references, saline and/or other medical solutions can lubricate and/or cool component between the motor assembly and the operative device. If desired, waste fluids can be removed from the catheter assembly 100A using the fluid handling system 350. In some embodiments, the fluid handling system 350 can include a multilumen catheter body having a proximal end and a distal end. The catheter body can include one or more lumens through which medical solutions (e.g., saline), waste fluids, and/or blood can flow. To drive fluid through the catheter assembly 100A (e.g., into and/or out of the catheter assembly 100A), the console 301 may include one or more pump(s) configured to apply positive or negative pressure to the catheter assembly 100A when the catheter assembly 100A is coupled to the console 301 and engages the pump(s).
[0043] In addition, the fluid handling system 350 may also be configured to provide electrical communication between the console 301 and the catheter assembly 100A. For example, the console can include a controller (e.g., a processor) that is programmed to control and/or manage the operation of the motor assembly. The console 301 may also include electrical interfaces configured to supply power to the motor assembly and/or other components that are driven by electrical power when the interface member 300 is coupled to the console 301. Moreover, one or more electrical or electronic sensors may be provided in the catheter assembly 100A and may electrically couple to the console 301 by way of the fluid handling system 350. The embodiments disclosed herein may thereby provide fluid and electrical connections between the catheter assembly 100A and the console 301.
[0044] As explained above, the fluid handling system 350 may provide a removable interface between the catheter assembly 100A and the console 301, which may include various components, including, e.g., one or more pump(s), processors (e.g., the controller), electrical interconnections, etc. For example, to activate one or more pumps in the console 301 and/or to engage one or more electrical connections between the console 301 and the interface member 300, a user may simply insert a distal portion of the interface member 300 (e.g., including a closure member) along the illustrated Z-direction into an aperture 304 of the interface region 303 until the pump(s) are engaged and the electrical connection(s) are formed. In some aspects, the insertion of the interface member along the Z-direction may engage the pump(s) and complete the electrical connection(s) substantially simultaneously.
[0045] In some embodiments, the interface member 300 may be secured to the console 301 by engaging a locking device between the interface region 303 and the interface member 300. One convenient way to engage a locking device is by rotating a portion of the interface member 300 relative to another portion of the interface member or relative to the console 301, as explained herein. For example, rotation of an outermost structure (opposite the direction Z), sometimes referred to herein as a "cap" relative to the console may engage a locking mechanism configured to mechanically secure the interface member 300 to the console 301 to prevent the interface member 300 from being accidentally disengaged during a treatment procedure.
[0046] The console 301 may also include a user interface 312, which may comprise a display device and/or a touch-screen display. The user may operate the percutaneous heart pump system by interacting with the user interface 312 to select, e.g., desired flow rates and other treatment parameters. The user may also monitor properties of the procedure on the user interface 312.
[0047] FIG. 3B is a three-dimensional perspective view of the interface region 303 of the console 301 shown in FIG. 3A. The interface region 303 can include the aperture 304 configured to receive the distal portion of the interface member 303. The aperture 304 may include a generally circular cavity shaped and sized to receive a portion of the interface member 300. A bubble detector 308 (e.g., an optical sensor in some embodiments) can be positioned at a back wall of the aperture 304. The bubble detector 308 may include a recess portion defined by two walls sized and shaped to receive a segment of tubing. When fluid flows through the tubing (see, e.g., bubble detector tube segment 326 in FIG. 4), the bubble detector 308 may monitor the fluid to determine whether or not the fluid includes unwanted matter, e.g., bubbles of air or other gas. In some embodiments, the bubble detector 308 may measure the amount (number or volume) of bubbles in the fluid passing though the tube segment. It should be appreciated that it can be important to detect bubbles in the treatment fluid to avoid inducing embolisms in the patient. The bubble detector 308 may electrically communicate with the controller in the console 301 and can indicate the amount of bubbles in the treatment fluid. The console 301, in turn, can alert the user if there are bubbles in the treatment fluid.
[0048] The interface region 303 can also include one or more pumps, e.g., peristaltic pumps in some embodiments. The peristaltic pumps can be used to pump fluid into or out of the catheter assembly 100A to deliver medical fluids and to remove waste fluids, respectively. Such pumps may employ one or more rollers 306 to control delivery of a fluid within a respective tube (see, e.g., pump tube segments 324a, 324b of FIG. 4). For example, the one or more pump rollers 306 can be housed within the console 301. As shown, two pump rollers 306 are mounted about their rotational axes (e.g., the Y-direction illustrated in FIG. 3B) at the back wall of the aperture 304. The pump rollers 306 can be rotated by a peristaltic pump motor within the console (not shown in FIGS. 3A-3B). As explained in more detail herein with respect to FIG. 4 below, the rollers 306 can engage pump tube segments 324a, 324b to pump fluid into or out of the catheter assembly 100A. The pump rollers 306 may be configured to be received within occlusion bed regions of the interface member 300 (see, e.g., occlusion beds 322a and 322b of FIG. 4) to pump fluid through the catheter assembly 100A.
[0049] An electrical interconnect 307 can also be provided in the back wall of the aperture 304. The electrical interconnect 307 can be configured to provide power to the motor assembly and/or electrical signals or instructions to control the operation of the motor assembly. The electrical interconnect 307 can also be configured to receive electrical signals indicative of sensor readings for monitoring pressure, flow rates, and/or temperature of one or more components in the catheter assembly 100A. A recessed channel 309 can extend from the bottom of the aperture 304 along the side to the lower edge of the console 301. The recessed channel 309 can be shaped and sized to receive one or more of the conduits 302 (e.g., electrical and/or fluid conduits) extending between the interface member 300 and the motor assembly. In one embodiment, all of the conduits 302 can be received within the channel 309 providing a flush side surface when the interface member 300 is disposed in the interface aperture 304.
[0050] In addition, it can be important to ensure that the interface member 300 is controllably secured within the console 301 such that it is engaged and disengaged only when the user desires to engage or disengage the interface member 300 from the console 301. For example, as explained in more detail herein relative to FIGS. 5A-5C, the interface region 303 can include a groove 313 sized and shaped to receive a locking mechanism (e.g., a tab or flange projecting in the X direction) on the interface member 300. In one embodiment, a disengaging member 305 includes a spring-loaded release mechanism 310 provided above the aperture 304 and a pin 311 that can be inserted into a hole in the interface member 300 (see, e.g., FIGS. 5A-5C and the accompanying disclosure below). As explained below with respect to FIGS. 5A-5C, the pin 311 can assist in releasing the interface member 300 relative to the console 301. The spring-loaded release mechanism 310 can be pressed to release the pin 311 and unlock the interface member 300 from the console 301. As explained herein, the spring-loaded release mechanism 310 can therefore act as a further safety mechanism to ensure that the cassette is not accidentally disengaged by the user.
Removable Interface Member
[0051] FIG. 4 is a three-dimensional perspective view of the interface member 300, according to one embodiment. The interface member 300 can comprise a body that is shaped and sized to fit into the interface region 303 of the console 301. As shown in FIG. 4, the interface member 300 can have a substantially circular profile, and is sometimes referred to as a puck. In some embodiments, the interface member 300 can include an outer body 333 operably coupled to a manual interface 320, sometimes referred to as a cap. The manual interface 320 is generally palm-sized so that a user can receive it in their hand and operate it comfortably, e.g., with finger pressure on the outer rim of the cap. One or more occlusion beds can be formed or provided at the interface between the interface member 300 and the console 301, e.g., in or on the interface member 300. For example, first and second occlusion beds 322a and 322b may be formed in the interface member 300. As shown in FIG. 4, for example, the occlusion beds 322a, 322b, can include arcuate recessed regions formed in the interface member 300.
[0052] The interface member 300 can further include first and second pump tube segments 324a, 324b positioned along the occlusion beds 322a, 322b formed in the interface member 300. When the interface member 300 is inserted into the console 301, the pump rollers 306 can engage with the interface member 300 and compress the tube segment(s) 324a, 324b against the occlusion bed(s) 322a, 322b, respectively. As the pump motor(s) in the console 301 rotate the rollers 306, fluid flows into uncompressed portions of the tube segment(s) 324a, 324b and continues flowing throughout the catheter assembly 100A. For example, by compressing the tube segments 324a, 324b, the fluid may be pumped into or out of the catheter assembly 100A by way of the conduits 302 extending from the interface member 300 to the motor assembly and distally beyond the motor assembly.
[0053] Because the tolerances for the peristaltic pump can be rather tight, the body of the interface member 300 (e.g., the outer body 333 and/or an inner body, such as inner body 339 illustrated in FIGS. 5B-5C) can be formed with precise tolerances (e.g., molded from a unitary structure in some implementations) such that when the interface member 300 is inserted into the console 301, the pump rollers 306 precisely and automatically engage with the tube segments 324a, 324b and occlusion beds 322a, 322b to reliably occlude the tube segments 324a, 324b and pump fluids through the catheter assembly 100A. Thus, when the interface member 300 is inserted sufficiently far into the interface region 303, the pump in the console 301 can automatically engage the interface member 300.
[0054] For example, the gap between the rollers 306 and the occlusion beds 322a, 322b can be less than about two wall thicknesses of the tube segments 324a, 324b in some arrangements, such that the tubes 324a, 324b can be effectively occluded. Due to the precise tolerances of the interface member 300, the pump can be engaged by simply inserting the interface member 300 into the console 301. There is no need to separately activate the pump in some embodiments. The dimensions of the interface member 300 may be selected such that the occlusion bed(s) 322a, 322b automatically engages the respective pump rollers 306 upon insertion of the interface member 300 into the console 301.
[0055] The above configuration provides several advantages. As one of skill in the art will appreciate from the description herein, the interface member 300 and interface region 303 provide an easy-to-use, quick connection of the tubing segments to one or more respective rollers 306. Moreover, the components can be manufactured easily and cost-effectively because only certain components require tight tolerances and the interface of member 300 to region 303 is essentially self-aligning. The interface also eliminates any need to engage the pump through a second mechanism or operator step, streamlining operation of the heart pump and simplifying the engagement of the catheter assembly 100A to the console 301. Also, in implementations where the console 301 is mounted on an IV pole with rollers, or another type of lightweight cart, for example, the simplified engagement mechanisms disclosed herein can be advantageous because there is only a minimal applied force against the pole, which prevents the pole from rolling or tipping when the pump is engaged.
[0056] The pump tube segments 324a, 324b can be mounted on the interface body 300 near or in the respective occlusion beds 322a, 322b. As illustrated, the first and second pump tube segments 324a, 324b can be configured to engage with the pump rollers 306 in the console 301, as explained above. The first and second pump tube segments 324a, 324b can have an arcuate shape (which may be pre-formed in various arrangements) that generally conforms to the curved shape of each respective occlusion bed 322a, 322b. The pump rollers 306 within the console 301 can thereby be positioned within the occlusion beds 322a, 322b to compress the tube segments 324a, 324b against the wall of the occlusion beds 322a, 322b. In addition, a bubble detector tube segment 326 can also be mounted in or on the interface member 300 and can be configured to engage with or be positioned adjacent to the bubble detector 308 illustrated in FIG. 3B. The bubble detector tube segment 326 can be any suitable shape. As illustrated, the bubble detector tube segment can be substantially straight and can be sized and shaped to be received by the bubble detector 308 within the console 301. As explained above with respect to FIGS. 3A-3B, the bubble detector 308 (which may be an optical sensor) can be used to detect air bubbles in the treatment or lubricating fluid being supplied to the patient.
[0057] The tube segments can be fluidly connected to the remainder of the catheter assembly 100A, including, e.g., one or more lumens of the catheter body, by way of the conduits 302. In operation, therefore, the removable interface member 300 may allow fluid to be pumped into and out of the patient within a controlled system, e.g., such that the fluids within the catheter assembly 100A can be pumped while maintaining a sterile environment for the fluids. Depending on the implementation, the volume of medical solution into the catheter body can be equal to, or can exceed by a minimum amount, the volume of medical solution out of the catheter body to assure that blood does not enter a blood-free portion of the heart pump.
[0058] In addition, one or more electrical contacts 328 can be provided in the interface member 300. The electrical contacts 328 can be any suitable electrical interface configured to transmit electrical signals between the console 301 and the catheter assembly 100A (e.g., the motor assembly and/or any suitable sensors). For example, the electrical contacts 328 can be configured to electrically couple to the electrical interconnect 307 disposed in the console 301. Electrical control signals and/or power may be transmitted between the console 301 and the catheter assembly 100A by way of the electrical connection between the electrical contacts 328 and the electrical interconnect 307. Advantageously, the electrical connection between the electrical contacts 328 and the electrical interconnect 307 may be formed or completed when the interface member 300 is inserted into the interface region 303 of the console 301. For example, in some embodiments, the electrical connection between the electrical contacts 328 and the electrical interconnect 307 may be formed substantially simultaneously with the fluid connection (e.g., the engagement of the pump) when the interface member 300 is inserted into the interface region 303. In some aspects, for example, the electrical connection can be formed by inserting electrical pins from the electrical contacts 328 into corresponding holes of the electrical interconnect 307 of the console 301, or vice versa.
[0059] Further, as shown in FIG. 4, the manual interface 320 can be mechanically coupled to a proximal portion of the outer body 333 and may be configured to rotate relative to the outer body 333 in a constrained manner, as explained below relative to FIGS. 5A-5C. For example, the outer body 333 can include one or more locking apertures 331 configured to receive locking tabs 332 that are configured to lock the manual interface 320 relative to the console 301. Moreover, as explained below relative to FIGS. 5A-5C, the outer body 333 may include a pin hole 321 sized and shaped to receive the pin 311 illustrated in FIG. 3B to releasably couple the removable interface member 300 relative to the console 301.
[0060] One will appreciate from the description herein that the configuration of the pump rollers, occlusion bed, and tubing can be modified depending on the application in accordance with the present inventions. For example, the configuration may be modified to provide easier access for service and repair. In various embodiments, the pump rollers may be disposed external to the console. In various embodiments, the pump rollers and occlusion bed may be both disposed within the cassette. In various embodiments, the console includes a mechanism to actuate the pump rollers in the cassette. In various embodiments, the rollers may be fixed. In various embodiments, the rollers may be configured to rotate, translate, or both. The rollers and/or the occlusion bed may be positioned on a base that is configured to move. In some embodiments, the console-cassette interface can include a positive pressure interface to pump fluid (e.g., saline) into the patient's vasculature and a negative pressure interface to pump fluid (e.g., waste fluid) out of the patient's vasculature.
Locking Mechanism
[0061] As discussed above, the interface member 300 advantageously can be fully engaged with the console 301 by simply inserting it into a correspondingly shaped aperture 304 in the housing of the console 301. When interface member 300 is brought into adjacency with a back wall of the interface region 303 of the console, e.g., when the interface member 300 is inserted into the aperture 304, the fluid handling and electrical connections are made, and the system 350 is operational. A locking mechanism in the interface member 300 can be provided for additional security, which can be particularly useful for patient transport and other more dynamic settings. For example, it is desirable to ensure that the catheter assembly 100A is secured to the console 301 during the entire procedure to ensure that the procedure is not disrupted due to accidental disengagement of the interface member 300 from the console 301.
[0062] In one embodiment, the locking mechanism can be disposed between the console 301 and the interface member 300 and can be configured to be engaged by a minimal movement of an actuator. For example, the manual interface 320 can be provided to cause engagement of a locking device by a small rotational turn of the manual interface 320 relative to the console 301.
[0063] FIG. 5A is a three-dimensional perspective view of the manual interface 320. As shown in FIG. 5A, the manual interface 320 can include or be coupled with an internal cam 335. The cam 335 can include one or more protruding lobes, such as lobes 336a and 336b. Further, the cam 335 can include a recessed region 337 recessed inwardly relative to the lobes 336a, 336b. The cam 335 can also include a stepped region 338 which can enable the interface member 300 to be locked and unlocked relative to the console 301, as explained herein.
[0064] FIG. 5B is a three-dimensional perspective view of an interface member 300A in an unlocked configuration, and FIG. 5C is a three-dimensional perspective view of an interface member 300B in a locked configuration. It should be appreciated that the interface members 300A, 300B of FIGS. 5B and 5C are illustrated without the outer body 333, which has been hidden in FIGS. 5B and 5C for purposes of illustration. Unless otherwise noted, the components of FIGS. 5B and 5C are the same as or similar to the components illustrated with respect to FIG. 4. As shown in FIGS. 5B and 5C, the interface members 300A, 300B can include an inner body 339 that can be disposed within the outer body 333 shown in FIG. 4. The occlusion beds 322a, 322b can be formed in the inner body 339 of the interface member 300A, 300B, as shown in FIGS. 5B-5C; however, in other arrangements, the occlusion beds 322a, 322b may be formed in the outer body 333 or other portions of the interface member 300A, 300B. In addition, as shown in FIGS. 5A and 5B, an electrical component 340 can be disposed in a recess or other portion of the inner body 339. Additional details regarding the electrical component 340 are explained below with respect to FIGS. 6A-6B.
[0065] The inner body 339 of the interface member 300A, 300B can further include a protrusion 330 that includes the tab 332 at a distal portion of the protrusion 330. When the interface member 300A is in the unlocked configuration, the protrusion 330 can be disposed in or near the recess 337 of the cam 335 in the manual interface 320. The cam 335 may therefore not contact or apply a force against the protrusion 330 when the interface member 300A is in the unlocked configuration, as shown in FIG. 5B.
[0066] However, once the interface member 300 is inserted into the console 301, the interface member 300 can be locked into place by rotating the manual interface 320 relative to the inner body 339 and the console 301, e.g., rotated in the A-direction illustrated in FIG. 5B. When the manual interface 320 is rotated, the internal cam 335 is also rotated within the interface member 300A, 300B. Once the cam is rotated, the lobes 336a, 336b of the cam 335 can engage with the one or more protrusions 330 of the inner body 339 and can push the protrusions 330 outwardly relative to the inner body 339. In one embodiment, the tabs 332 may extend outwardly through the locking apertures 331 formed on the outer body 333. When the tab(s) 332 are pushed through the locking aperture(s) 331, the tabs 332 project laterally outward from the outer body 333. In this position, in some embodiments, each of the tabs 332 can lock into the groove(s) 313 in the console 301 (see FIG. 3B) to secure the interface member 300B to the console 301. Thus, in the unlocked position, the tab 332 can be substantially flush with the outer surface of the interface member 300A, and in the locked position, the tab 332 can extend through the locking aperture 331 and lock into the grooves 313 in the console 301.
[0067] In some embodiments, the protrusion 330 can be a cantilevered protrusion from the inner body 339. As mentioned above, it can be important to maintain tight tolerances between the occlusion beds 322a, 322b, which is also formed in the interface member, and the pump rollers 306 when the interface member 300 engages with the console 301. Because the occlusion beds 322a, 322b may be formed in the same body as the cantilevered protrusions 330, conventional manufacturing processes, such as molding processes, can be used to manufacture the interface member 300 (e.g., the outer body 333 and/or the inner body 339) according to precise dimensions. Thus, the protrusion(s) 330, tab(s) 332 and the occlusion bed(s) 322a, 322b can be made within tight dimensional tolerances, and the tab(s) 332 and/or protrusion(s) 330 can be positioned relative to the occlusion bed(s) 322a, 322b with very high precision such that when the interface member 300 is engaged with the console 301, the tube segments 324a, 324b are optimally occluded. Moreover, because the interface member 300 can be locked by rotating the manual interface 320 on the interface member 300, only minimal forces are applied to the console 301. This enhances the advantages of minimizing disruption of a mobile cart or IV pole to which the system may be coupled.
Disengagement Mechanism
[0068] It can also be important to provide a disengagement mechanism configured to decouple the interface member 300 from the console 301. With reference to FIGS. 3B, 4, 5B, and 5C, the disengaging member 305 of the console 301 can be configured to disengage and unlock the interface member 300 from the console 301. For example, the pin 311 may be spring-loaded such that when the interface member 300A is in the unlocked configuration, the pin 311 extends through the pin hole 321 of the outer body 333 but only contacts a side surface of one of the lobes 336b of the cam 335. Thus, in the unlocked configuration of the interface member 300A, the pin 311 may simply slide along the cam surface, permitting rotation of the manual interface 320 relative to the pin 311 and the console 301.
[0069] As shown in FIGS. 3B and 5C, however, when the interface member 300B is rotated into a locked configuration, the pin 311 can engage with the stepped region 338 of the internal cam 335, e.g., the spring-biased pin 311 can extend into the stepped region 338 or shoulder of the cam 335. By engaging the stepped region 338, the pin 311 prevents the cam 335 from rotating from the locked configuration to the unlocked configuration. A user can disengage the cassette by pressing the spring-loaded release mechanism 310 to release the spring and remove the pin 311 from the stepped region 338. The pin 311 can thereby be disengaged from the stepped region 338, and the internal cam 335 can rotate back into the unlocked position. When the cam 335 is moved back into the unlocked position, the tab 332 can be withdrawn from the groove 313 in the console 301 to unlock the interface member 300.
Electrical Interconnections, Components, and Cables
[0070] FIG. 6A is a three-dimensional perspective view of a first side of the electrical component 340 illustrated in FIG. 4. FIG. 6B is a three-dimensional perspective view of a second, opposite side of the electrical component 340 of FIG. 6A. As shown in FIGS. 5B-5C, the electrical component 340 may be disposed in a recess of the interface member 300. The electrical component 340 can be any suitable electrical or electronic component, including, e.g., a printed circuit board (PCB) configured to provide an electrical interface between various components in the catheter assembly 100A and the console 301. As explained herein, the electrical component 340 can form an electrical interface between the interface member 300 and the console 301 to provide electrical communication between the console 301 and the catheter assembly 100A (such as the motor assembly and/or various sensors).
[0071] For example, the electrical component 340 of the interface member 300 can include the one or more electrical contacts 328 configured to mate with the corresponding electrical interconnect 307 in the console 301. The electrical contacts 328 and/or the electrical interconnect 307 can be, for example, nine-pin electrical interconnects, although any suitable interconnect can be used. The motor assembly that drives the operative device (e.g., impeller) of the catheter pump can be electrically connected to the interface member 300 by way of one or more electrical cables, e.g., the conduits 302. In turn, the console 301 can be coupled to a power source, which can drive the catheter pump motor assembly by way of the interface member's contacts 328 and the electrical conduits 302 connecting the interface member 300 to the motor assembly. The electrical component 340 can also include communications interconnects configured to relay electrical signals between the console 301 and the catheter pump motor assembly or other portions of the catheter assembly 100A. For example, a controller within the console 301 (or interface member) can send instructions to the catheter pump motor assembly via the electrical component 340 between the console 301 and the interface member 300. In some embodiments, the electrical component 340 can include interconnects for sensors (such as pressure or temperature sensors) within the catheter assembly 100A, including sensors at the operative device. The sensors may be used to measure a characteristic of the fluid in one or more of the tubes (e.g., saline pressure). The sensors may be used to measure an operational parameter of the system (e.g., ventricular or aortic pressure). The sensors may be provided as part of an adjunctive therapy.
[0072] The electrical component 340 within the interface member 300 can be used to electrically couple the cable (and the motor assembly, sensors, etc.) with the corresponding interconnects 307 in the console 301. For example, one or more internal connectors 346 and 348 on the second side of the electrical component 340 may provide electrical communication between the contacts 328 (configured to couple to the interconnects 307 of the console 301) and the catheter assembly 100. For example, electrical cables (e.g., the conduits 302) can couple to a first internal connector 346 and a second internal connector 348. The internal connectors 346, 348 may electrically communicate with the contacts 328 on the first side of the electrical component 340, which in turn communicate with the interconnects 307 of the console 301.
[0073] In various embodiments, the electrical component 340 is fluidly sealed to prevent the internal electronics from getting wet. This may be advantageous in wet and/or sterile environments. This may also advantageously protect the electronics in the event one of the fluid tubes leaks or bursts, which is a potential risk in high pressure applications.
[0074] In addition, the electrical component 340 (e.g., PCB) can include various electrical or electronic components mounted thereon. As shown in FIG. 6B, for example, two pressure sensors 344a, 344b can be mounted on the electrical component 340 to detect the pressure in the pump tube segments 324a, 324b. The pressure sensors 344a, 344b may be used to monitor the flow of fluids in the tube segments 324a, 324b to confirm proper operation of the heart pump, for example, confirming a proper balance of medical solution into the catheter body and waste out of the catheter body. Various other components, such as a processor, memory, or an Application-Specific Integrated Circuit (ASIC), can be provided on the circuit board. For example, respective pressure sensor ASICs 345a, 345b can be coupled to the pressure sensors 344a, 344b to process the signals detected by the pressure sensors 344a, 344b. The processed signals may be transmitted from the ASICs 345a, 345b to the console 301 by way of internal traces (not shown) in the PCB and the contacts 328.
Priming and Infusate System and Apparatus
[0075] One embodiment of an infusate system 1300 is illustrated in FIG. 7. Various components described herein can be understood in more detail by referencing the patent applications incorporated by reference herein. The infusate system 1300 can be configured to supply treatment and/or lubricating fluids to the operative device of the catheter assembly (e.g., an impeller assembly 116), and to remove waste fluid from the assembly. Furthermore, as explained herein, an elongate body 174 can be slidably disposed over a catheter body 120, such that there may be gaps or channels between the outer surface of the catheter body 120 and the inner surface of the elongate body 174. Such gaps or channels can contain air pockets harmful to the patient during a medical procedure. In addition, the lumen or lumens extending within the catheter body 120 also can contain air pockets harmful to the patient. Thus, it is desirable to expel air from both the lumens within catheter body 120 and the gaps or channels disposed between the elongate body 174 and the catheter body 120 before conducting a treatment procedure.
[0076] The system 1300 of FIG. 7 may be configured to supply fluid to the catheter assembly during treatment, to remove waste fluid during treatment, and/or to expel air from the elongate body 174, e.g., between the inner surface of the elongate body 174 and the outer surface of the catheter body 120 before treatment. In this embodiment, an interface member 1313 (similar to or the same as the interface member 300 described herein, in some aspects) may be provided to connect various components of the catheter assembly, as discussed herein. An outer sheath tubing 1303a can extend from a fluid reservoir 1305 to a luer 102 configured to be coupled to an infusate device. As shown in FIG. 7, the outer sheath tubing 1303a can be configured to deliver fluid to the outer sheath, e.g., the space between the elongate body 174 and the catheter body 120. The fluid reservoir 1305 may optionally include a pressure cuff to urge fluid through the outer sheath tubing 1303a. Pressure cuffs may be particularly useful in fluid delivery embodiments using gravity-induced fluid flow. The luer 102 can be configured to deliver infusate or other priming fluid to the elongate body 174 to expel air from the elongate body 174 as described herein in order to "prime" the system 1300. In addition, a pressure sensor 1309a, which may be disposed on a motor housing 1314, can be coupled to the outer sheath tubing 1303a to measure the pressure of the infusate or priming fluid flowing through the outer sheath tubing 1303a and into the luer 102. The motor housing 1314 illustrated in FIG. 7 may be the same as or similar to the motor assembly described above with reference to FIG. 2, for example, when the drive assembly 103 is coupled to the driven assembly 101.
[0077] As illustrated in the embodiment of FIG. 7, inner catheter tubing 1303b can extend between the motor housing 1314 and the fluid reservoir 1305, by way of a T-junction 1320. The inner catheter tubing 1303b can be configured to deliver fluid to the lumen or lumens within catheter body 120 during treatment and/or to expel air from the catheter 120 and prime the system 1300. A pumping mechanism 1306a, such as a roller pump for example, can be provided along inner catheter tubing 1303b to assist in pumping the infusate or priming fluid through the system 1300. As explained herein, the roller pump can be a peristaltic pump in some arrangements. In addition, an air detector 1308 can be coupled to the inner catheter tubing 1303b and can be configured to detect any air or bubbles introduced into the system 1300. In some embodiments, a pressure sensor 1309b can couple to inner catheter tubing 1303b to detect the pressure of the fluid within the tubing. Additionally, a filter 1311 can be employed to remove debris and other undesirable particles from the infusate or priming fluid before the catheter body 120 is infused or primed with liquid. In some embodiments, the air detector 1308, the pressure sensor 1309b, and the pumping mechanism 1306a can be coupled to the interface member 1313 described above (such as the interface member 300). One or more electrical lines 1315 can connect the motor housing 1314 with the cassette 1313. The electrical lines 1315 can provide electrical signals for energizing a motor or for powering the sensor 1309a or for other components. To expel air from the catheter body 120, infusate or priming fluid can be introduced at the proximal end of the catheter assembly. The fluid can be driven distally to drive air out of the catheter body 120 to prime the system.
[0078] In some aspects, a waste fluid line 1304 can fluidly connect the catheter body 120 with a waste reservoir 1310. A pressure sensor 1309c, which may be disposed on or coupled to the interface member 1313, can measure the pressure of the fluid within the waste fluid line 1304. A pumping mechanism 1306b, such as a roller pump, for example, can be coupled to the interface member 1313 and can pump the waste fluid through the waste fluid line 1304 to the waste reservoir 1310.
[0079] FIG. 8 is an enlarged view of the priming apparatus 1400 shown in FIG. 2. As explained above, the priming apparatus 1400 may be disposed over the impeller assembly 116A near the distal end 170A of the elongate body 174A. The priming apparatus 1400 can be used in connection with a procedure to expel air from the impeller assembly 116A, e.g., any air that is trapped within the housing or that remains within the elongate body 174A near the distal end 170A. For example, the priming procedure may be performed before the pump is inserted into the patient's vascular system, so that air bubbles are not allowed to enter and/or injure the patient. The priming apparatus 1400 can include a primer housing 1401 configured to be disposed around both the elongate body 174A and the impeller assembly 116A. A sealing cap 1406 can be applied to the proximal end 1402 of the primer housing 1401 to substantially seal the priming apparatus 1400 for priming, i.e., so that air does not proximally enter the elongate body 174A and also so that priming fluid does not flow out of the proximal end of the housing 1401. The sealing cap 1406 can couple to the primer housing 1401 in any way known to a skilled artisan. However, in some embodiments, the sealing cap 1406 is threaded onto the primer housing by way of a threaded connector 1405 located at the proximal end 1402 of the primer housing 1401. The sealing cap 1406 can include a sealing recess disposed at the distal end of the sealing cap 1406. The sealing recess can be configured to allow the elongate body 174A to pass through the sealing cap 1406.
[0080] The priming operation can proceed by introducing fluid into the sealed priming apparatus 1400 to expel air from the impeller assembly 116A and the elongate body 174A. Fluid can be introduced into the priming apparatus 1400 in a variety of ways. For example, fluid can be introduced distally through the elongate body 174A into the priming apparatus 1400. In other embodiments, an inlet, such as a luer, can optionally be formed on a side of the primer housing 1401 to allow for introduction of fluid into the priming apparatus 1400.
[0081] A gas permeable membrane can be disposed on a distal end 1404 of the primer housing 1401. The gas permeable membrane can permit air to escape from the primer housing 1401 during priming.
[0082] The priming apparatus 1400 also can advantageously be configured to collapse an expandable portion of the catheter assembly 100A. The primer housing 1401 can include a funnel 1415 where the inner diameter of the housing decreases from distal to proximal. The funnel may be gently curved such that relative proximal movement of the impeller housing causes the impeller housing to be collapsed by the funnel 1415. During or after the impeller housing has been fully collapsed, the distal end 170A of the elongate body 174A can be moved distally relative to the collapsed housing. After the impeller housing is fully collapsed and retracted into the elongate body 174A of the sheath assembly, the catheter assembly 100A can be removed from the priming housing 1400 before a percutaneous heart procedure is performed, e.g., before the pump is activated to pump blood. The embodiments disclosed herein may be implemented such that the total time for infusing the system is minimized or reduced. For example, in some implementations, the time to fully infuse the system can be about six minutes or less. In other implementations, the infusate time can be less than 5 minutes, less than 4 minutes, or less than 3 minutes. In yet other implementations, the total time to infuse the system can be about 45 seconds or less. It should be appreciated that lower infusate times can be advantageous for use with cardiovascular patients.
Preparing a Percutaneous Heart Pump for Insertion into the Vasculature
[0083] As discussed herein and in the incorporated patent applications, in various embodiments the heart pump is inserted in a less invasive manner, e.g., using techniques that can be employed in a catheter lab.
[0084] Prior to insertion of the catheter assembly 100A of the heart pump, various techniques can be used to prepare the system for insertion. For example, as discussed in connection with FIG. 8, the catheter assembly 100A can be primed to remove gas that could be contained therein prior to any method being performed on the patient. This priming technique can be performed by placing a distal portion of the catheter assembly 100A in a priming vessel, such as the apparatus 1400. Thereafter, a media is delivered into the catheter assembly 100A under pressure to displace any potentially harmful matter, e.g., air or other gas, out of the catheter assembly 100A. In one technique, the apparatus 1400 is filled with a biocompatible liquid such as saline. Thereafter, a biocompatible liquid such as saline is caused to flow distally through the catheter assembly 100 to displace air in any of the cavities formed therein, as discussed above. A pressure or flow rate for priming can be provided that is suitable for priming, e.g., a pressure or flow rate that is lower than the operational pressure or flow rate.
[0085] In one technique, the biocompatible liquid is pushed under positive pressure from the proximal end through the catheter assembly 100A until all gas is removed from voids therein. One technique for confirming that all gas has been removed is to observe the back-pressure or the current draw of the pump. As discussed above, the priming apparatus 1400 can be configured to permit gas to escape while preventing saline or other biocompatible liquid from escaping. As such, the back-pressure or current draw to maintain a pre-selected flow will change dramatically once all gas has been evacuated.
[0086] In another technique, the priming apparatus 1400 can include a source of negative pressure for drawing a biocompatible liquid into the proximal end of the catheter assembly 100A. Applying a negative pressure to the priming apparatus 1400 can have the advantage of permitting the catheter assembly 100A to be primed separate from the pumps that are used during operation of the heart pump. As a result, the priming can be done in parallel with other medical procedures on the patient by an operator that is not directly working on the patient.
[0087] In another approach, a positive pressure pump separate from the pump that operates the heart pump can be used to prime under positive pressure applied to the proximal end. Various priming methods may also be expedited by providing a separate inlet for faster filling of the enclosed volume of the priming apparatus 1400.
Collapsing an Expandable Housing of a Fully Primed Catheter Assembly
[0088] A further aspect of certain methods of preparing the catheter assembly 100A for insertion into a patient can involve collapsing the impeller housing 116A. The collapsed state of the impeller housing 116A reduces the size, e.g., the crossing profile, of the distal end of the system. This enables a patient to have right, left or right and left side support through a small vessel that is close to the surface of the skin, e.g., using catheter lab-type procedures. As discussed above, in one technique the priming apparatus 1400 has a funnel configuration that has a large diameter at a distal end and a smaller diameter at a proximal end. The funnel gently transitions from the large to the small diameter. The small diameter is close to the collapsed size of the impeller housing 116A and the large diameter is close to or larger than the expanded size of the impeller housing 116A. In one method, after the catheter assembly 100A has been primed, the impeller housing 116A can be collapsed by providing relative movement between the priming apparatus 1400 and the impeller housing 116A. For example, the priming housing 1400 can be held in a fixed position, e.g., by hand, and the catheter assembly 100A can be withdrawn until at least a portion of the impeller assembly 116A is disposed in the small diameter segment of the priming apparatus 1400. Thereafter, the elongate body 174A of the sheath assembly can be advanced over the collapsed impeller assembly 116A.
[0089] In another technique, the catheter assembly 100A is held still and the priming apparatus 1400 is slid distally over the impeller assembly 116A to cause the impeller assembly 116A to collapse. Thereafter, relative movement between the elongate body 174A and the impeller assembly 116A can position the distal end 170A of the elongate body 174A over the impeller assembly 116A after the catheter assembly 100A has been fully primed.
[0090] Although the inventions herein have been described with reference to particular embodiments, it is to be understood that these embodiments are merely illustrative of the principles and applications of the present inventions. It is therefore to be understood that numerous modifications can be made to the illustrative embodiments and that other arrangements can be devised without departing from the spirit and scope of the present inventions as defined by the appended claims. Thus, it is intended that the present application cover the modifications and variations of these embodiments and their equivalents.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.