Antigen-specific Helper T-cell Receptor Genes
SUGIYAMA; Haruo ; et al.
U.S. patent application number 17/076842 was filed with the patent office on 2021-03-11 for antigen-specific helper t-cell receptor genes. The applicant listed for this patent is International Institute of Cancer Immunology, Inc.. Invention is credited to Fumihiro FUJIKI, Haruo SUGIYAMA.
| Application Number | 20210070832 17/076842 |
| Document ID | / |
| Family ID | 1000005237171 |
| Filed Date | 2021-03-11 |









View All Diagrams
| United States Patent Application | 20210070832 |
| Kind Code | A1 |
| SUGIYAMA; Haruo ; et al. | March 11, 2021 |
ANTIGEN-SPECIFIC HELPER T-CELL RECEPTOR GENES
Abstract
The present invention pertains to polynucleotides that encode CDR3 in TCR-[alpha] and TCR-[beta] chain genes of CD4.sup.+ helper T-cells that are specific to WT1 helper peptides having an amino acid sequence represented by SEQ ID NO: 123. The present invention further pertains to the peptides encoded by said polynucleotides. The present invention further pertains to CD4.sup.+ T cells into which TCR genes that contain said polynucleotides have been introduced, the induction of WT1-specific cytotoxic T-lymphocytes (CTLs) using the CD4.sup.+ T-cells, the treatment of cancer, etc.
| Inventors: | SUGIYAMA; Haruo; (Osaka, JP) ; FUJIKI; Fumihiro; (Kawabe-gun, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005237171 | ||||||||||
| Appl. No.: | 17/076842 | ||||||||||
| Filed: | October 22, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16163448 | Oct 17, 2018 | |||
| 17076842 | ||||
| 14427465 | Mar 11, 2015 | 10815288 | ||
| PCT/JP2013/074748 | Sep 12, 2013 | |||
| 16163448 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 38/00 20130101; C12Q 2600/158 20130101; G01N 33/56972 20130101; G01N 2333/4703 20130101; C07K 14/7051 20130101; C12Q 1/6886 20130101; C12Q 1/6881 20130101; C12N 5/0636 20130101; C07K 16/28 20130101; A61K 35/17 20130101; G01N 2333/70514 20130101 |
| International Class: | C07K 14/725 20060101 C07K014/725; C12N 5/0783 20060101 C12N005/0783; A61K 35/17 20060101 A61K035/17; C12Q 1/6886 20060101 C12Q001/6886; C07K 16/28 20060101 C07K016/28; C12Q 1/6881 20060101 C12Q001/6881; G01N 33/569 20060101 G01N033/569 |
Foreign Application Data
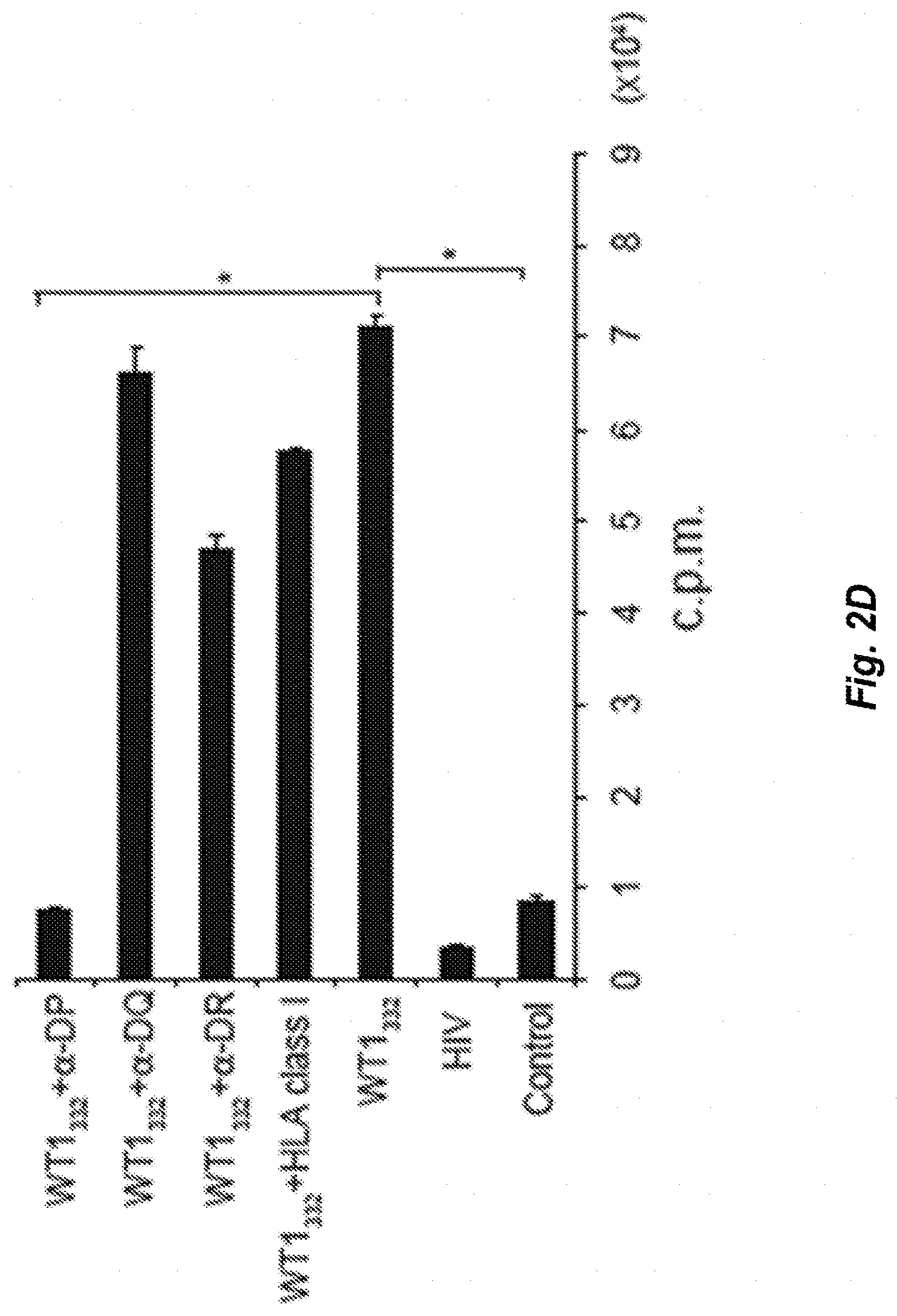
| Date | Code | Application Number |
|---|---|---|
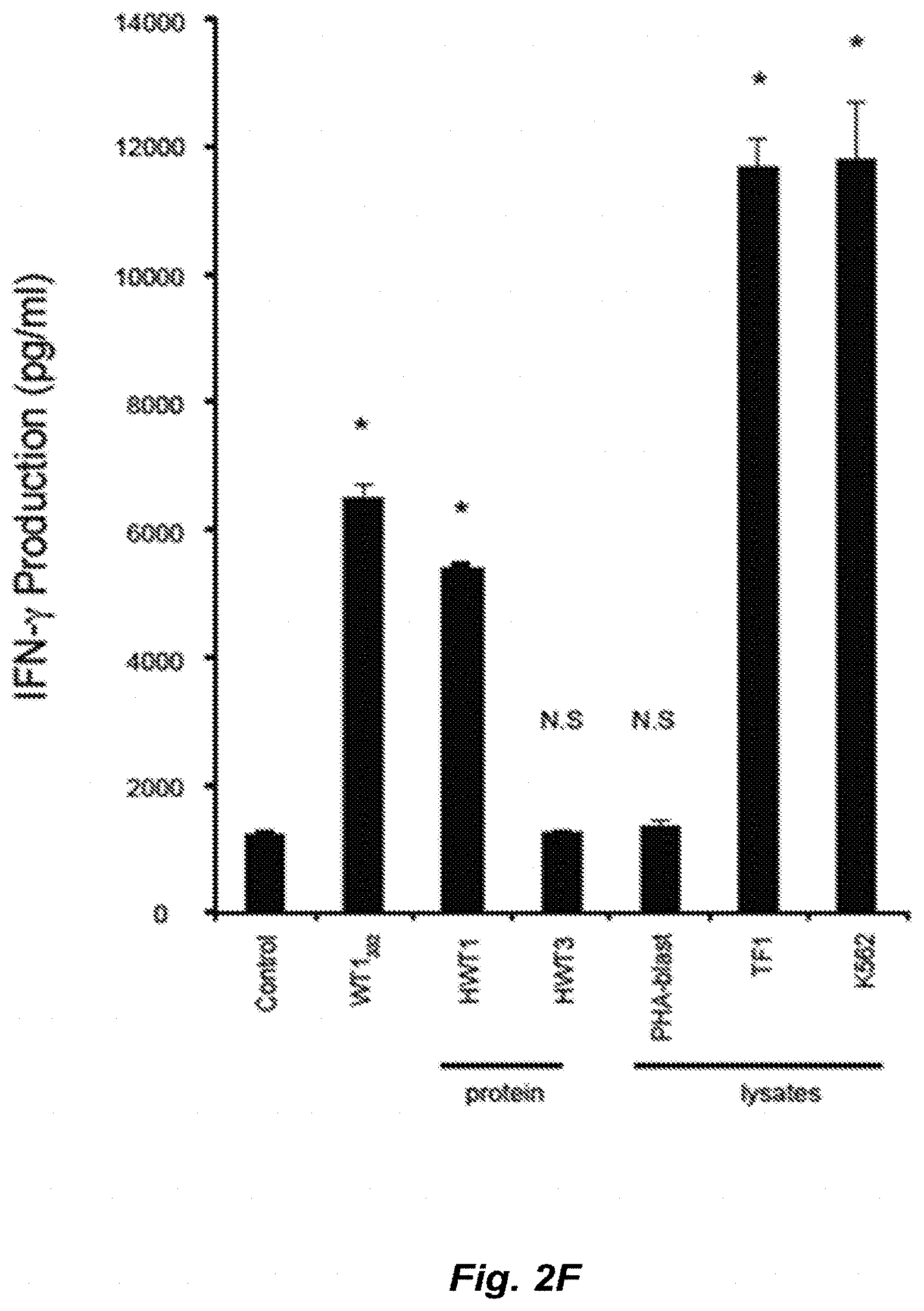
| Sep 12, 2012 | JP | 2012-200480 |
Claims
1-22. (canceled)
23. An .alpha.CDR3 peptide encoded by a polynucleotide having a nucleotide sequence selected from the group consisting of SEQ ID NOs: 1, 3, 5, 8, 10, 11, 13, 14, 16, 18, 20, 22, 23, 25, 27, 28, 30, 31, 33, 35, 37, 39, 41, 43, 45, 47, 49, 51, 53, 55, 56, and 58, wherein said polynucleotide encodes CDR3 of .alpha.-chain of TCR of a CDR.sup.+ helper T-cell specific to a WT1 helper peptide (WT1.sub.332 peptide) comprising SEQ ID NO: 123 or a sequence at least 90% identical to SEQ ID NO: 123.
24. A .beta.CDR3 peptide encoded by a polynucleotide having a nucleotide sequence selected from the group consisting of SEQ ID NOs: 2, 4, 6, 7, 9, 12, 15, 17, 19, 21, 24, 26, 29, 32, 34, 36, 38, 40, 42, 44, 46, 48, 50, 52, 54, 57, and 59, wherein said polynucleotide encodes CDR3 of .beta.-chain of TCR of a CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide.
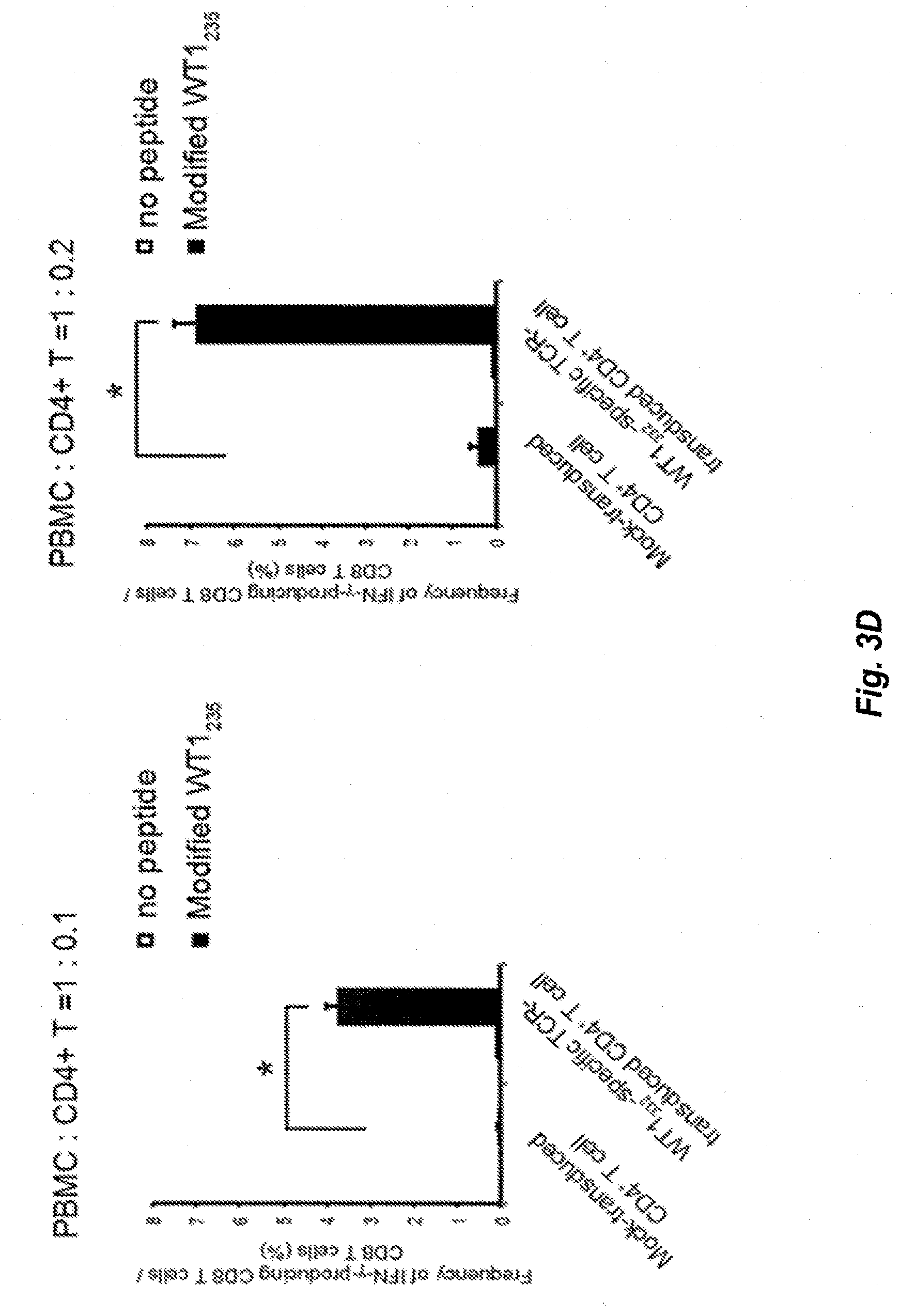
25. A pair of peptides encoded by a pair of polynucleotides chosen from: SEQ ID NO:1 and SEQ ID NO:2; SEQ ID NO:3 and SEQ ID NO:4; SEQ ID NO:5 and SEQ ID NO:6; SEQ ID NO:3 and SEQ ID NO:7; SEQ ID NO:8 and SEQ ID NO:9; SEQ ID NO:10 and SEQ ID NO:12; SEQ ID NO:11 and SEQ ID NO:12; SEQ ID NO:13 and SEQ ID NO:15; SEQ ID NO:14 and SEQ ID NO:15 SEQ ID NO:16 and SEQ ID NO:17 SEQ ID NO:18 and SEQ ID NO:19 SEQ ID NO:20 and SEQ ID NO:21 SEQ ID NO:22 and SEQ ID NO:24 SEQ ID NO:23 and SEQ ID NO:24 SEQ ID NO:25 and SEQ ID NO:26 SEQ ID NO:27 and SEQ ID NO:4 SEQ ID NO:28 and SEQ ID NO:29 SEQ ID NO:30 and SEQ ID NO:32 SEQ ID NO:31 and SEQ ID NO:32 SEQ ID NO:33 and SEQ ID NO:34 SEQ ID NO:35 and SEQ ID NO:36 SEQ ID NO:37 and SEQ ID NO:38 SEQ ID NO:39 and SEQ ID NO:40 SEQ ID NO:41 and SEQ ID NO:42 SEQ ID NO:43 and SEQ ID NO:44 SEQ ID NO:45 and SEQ ID NO:46 SEQ ID NO:47 and SEQ ID NO:48 SEQ ID NO:49 and SEQ ID NO:50 SEQ ID NO:51 and SEQ ID NO:52 SEQ ID NO:53 and SEQ ID NO:54 SEQ ID NO:55 and SEQ ID NO:57 SEQ ID NO:56 and SEQ ID NO:57; or SEQ ID NO:58 and SEQ ID NO:59 wherein each of the polynucleotide sequences may be a complementary sequence or a degenerate sequence thereof.
26. A chip comprising the peptide according to claim 23 or 24, or the pair of the peptides according to claim 25.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This is a divisional of application Ser. No. 16/163,448, filed Oct. 17, 2018, which is a divisional of application Ser. No. 14/427,465, filed Mar. 11, 2015, which is a national stage entry under 35 U.S.C. .sctn. 371(c) of International Application No. PCT/JP2013/074748, filed Sep. 12, 2013, and claims priority to Japanese Patent Application No. 2012-200480, filed Sep. 12, 2012, all of which are incorporated herein by reference.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Nov. 25, 2020, is named 05273_0155-02000_SL.txt and is 21,331 bytes in size.
TECHNICAL FIELD
[0003] The present invention relates to polynucleotides contained in T cell receptor (TCR) genes of cancer antigen-specific helper T-cells. In particular, the present invention relates to polynucleotides encoding complementarity determining region 3 (CDR3) of each .alpha.-chain and .beta.-chain of TCR of CD4.sup.+ helper T-cells specific to a WT1 helper peptide having an amino acid sequence shown in SEQ ID NO: 123. The present invention also relates to polypeptides encoded by these polynucleotides. Further, the present invention relates to CD4.sup.+ T-cells into which TCR genes containing these polynucleotides are introduced, induction and enhancement of WT1-specific cytotoxic T cells (WT1-specific CTL) using them, and treatment of cancers using them, and so on.
BACKGROUND ART
[0004] WT1 gene (Wilms' tumor 1 gene) is a gene which was identified as a gene responsible for Wilms tumor which is a renal cancer in children (Non-patent Documents 1 and 2). WT1 is a transcription factor having a zinc finger structure. Initially, WT1 gene was considered to be a tumor suppressor gene. However, subsequent studies (Non-patent Documents 3, 4, 5 and 6) showed that WT1 gene rather functions as an oncogene in hematopoietic tumors and solid cancers.
[0005] It was showed that peptide-specific cytotoxic T cells (CTLs) were induced by stimulating peripheral mononuclear cells in vitro using a WT1 peptide and that these CTLs damage tumor and cancer cells in hematopoietic tumors and solid cancers which express WT1 endogenously. Because CTL recognizes a WT1 peptide in a form of a complex in which the WT1 peptide is bound to a MHC class I molecule, such a WT1 peptide differs in accordance to MHC class I subtype (Patent Document 1, Non-Patent Document 7, Patent Documents 2, 3 and 4).
[0006] Existence of helper T cells specific to a cancer antigen is important for effective induction of CTL (Non-Patent Document 8). Helper T cells are induced and activated by recognizing a complex of a MHC class II molecule and an antigen peptide on antigen-presenting cells. Activated helper T cells produce cytokines such as IL-2, IL-4, IL-5, IL-6, or interferons, and help proliferation, differentiation and maturation of B cells. Activated helper T cells also have a function to promote proliferation, differentiation and maturation of other subset of T cells (such as Tc cells). Thus, because activated helper T cells have a function to activate immune system by promoting proliferation and activation of B cells and T cells, it is considered that enhancing the function of helper T cells via a MHC class II binding antigen peptide (helper peptide) to enhance the effect of a cancer vaccine is useful in cancer immunotherapy (Non-Patent Document 9).
[0007] Examples of helper peptides relating to WT1 which are presently recognized are a peptide binding to HLA-DRB1*04:01 molecule (Non-Patent Document 10), a peptide binding to HLA-DRB1*04:05 molecule, a peptide binding to HLA-DRB1*15:02 molecule (Patent Document 5), a peptide binding to HLA-DRB1*04:05 molecule, HLA-DRB1*15:02 molecule, HLA-DRB1*15:01 molecule, HLA-DPB1*09:01 molecule and HLA-DPB1*05:01 molecule (Patent Document 6).
[0008] However, sequences of T cell receptor (TCR) genes of antigen-specific CD4.sup.+ helper T-cells which recognize a helper peptide have not been known at all.
DOCUMENTS OF BACKGROUND ART
Patent Documents
[0009] Patent Document 1: WO2003/106682 [0010] Patent Document 2: WO2005/095598 [0011] Patent Document 3: WO2007/097358 [0012] Patent Document 4: PCT/JP2007/074146 [0013] Patent Document 5: WO2005/045027 [0014] Patent Document 6: WO2008/105462
Non-Patent Documents
[0014] [0015] Non-Patent Document 1: Daniel A. Haber et al., Cell. 1990 Jun. 29; 61(7):1257-69. [0016] Non-Patent Document 2: Call K M et al., Cell. 1990 Feb. 9; 60(3):509-20. [0017] Non-Patent Document 3: Menke A L et al., Int Rev Cytol. 1998; 181:151-212. Review. [0018] Non-Patent Document 4: Yamagami T et al., Blood. 1996 Apr. 1; 87(7):2878-84. [0019] Non-Patent Document 5: Inoue K et al., Blood. 1998 Apr. 15; 91(8):2969-76. [0020] Non-Patent Document 6: Tsuboi A et al., Leuk Res. 1999 May; 23(5):499-505. [0021] Non-Patent Document 7: Oka Y et al., Immunogenetics. 2000 February; 51(2):99-107. [0022] Non-Patent Document 8: Gao F G et al., Cancer Res. 2002 Nov. 15; 62(22):6438-41. [0023] Non-Patent Document 9: Zeng G, J Immunother. 2001 May; 24(3):195-204.
[0024] Non-Patent Document 10: Knights A J et al., Cancer Immunol Immunother. 2002 July; 51(5):271-81.
SUMMARY OF INVENTION
Problem to be Solved by the Invention
[0025] Problems to be solved by the invention were to determine sequences of TCR genes of CD4.sup.+ helper T-cells specific to a WT1 helper peptide, to obtain CD4.sup.+ T-cells into which these TCR genes have been introduced, to enhance induction of WT1 specific CTL using such cells, and to treat or prevent cancers, and so on.
Means to Solve the Problem
[0026] The inventors studied vary hard to solve the above problems, and succeeded in isolating .alpha.-chain genes and .beta.-chain genes of TCR of CD4.sup.+ helper T-cells specific to a WT1 helper peptide, and determined each CDR3 sequence. Further, the inventors introduced TCR genes containing the sequences thus determined into CD4.sup.+ T-cells, and succeeded in enhancing induction of WT1 specific CTL and damaging WT1 expressing cancer cells by use of the CD4.sup.+ T-cells. Thus, the inventors have completed the present invention.
[0027] That is, the present invention provides:
(1) A polynucleotide (referred as ".alpha.CDR3 polynucleotide") having a nucleotide sequence selected from the group consisting of SEQ ID NOs: 1, 3, 5, 8, 10, 11, 13, 14, 16, 18, 20, 22, 23, 25, 27, 28, 30, 31, 33, 35, 37, 39, 41, 43, 45, 47, 49, 51, 53, 55, 56 and 58, wherein said polynucleotide encodes CDR3 of .alpha.-chain of TCR of a CD4.sup.+ helper T-cell specific to a WT1 helper peptide (WT1.sub.332 peptide) having an amino acid sequence shown in SEQ ID NO: 123 or a variant sequence thereof. (2) A polynucleotide (referred as ".beta.CDR3 polynucleotide") having a nucleotide sequence selected from the group consisting of SEQ ID NOs: 2, 4, 6, 7, 9, 12, 15, 17, 19, 21, 24, 26, 29, 32, 34, 36, 38, 40, 42, 44, 46, 48, 50, 52, 54, 57 and 59, wherein said polynucleotide encodes CDR3 of .beta.-chain of TCR of a CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide. (3) A pair of a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide, wherein each polynucleotide has the following nucleotide sequence:
TABLE-US-00001 .alpha. CDR3 polynucleotide .beta.CDR3 polynucleotide SEQ ID NO: 1 SEQ ID NO: 2 SEQ ID NO: 3 SEQ ID NO: 4 SEQ ID NO: 5 SEQ ID NO: 6 SEQ ID NO: 3 SEQ ID NO: 7 SEQ ID NO: 8 SEQ ID NO: 9 SEQ ID NO: 10 SEQ ID NO: 12 SEQ ID NO: 11 SEQ ID NO: 12 SEQ ID NO: 13 SEQ ID NO: 15 SEQ ID NO: 14 SEQ ID NO: 15 SEQ ID NO: 16 SEQ ID NO: 17 SEQ ID NO: 18 SEQ ID NO: 19 SEQ ID NO: 20 SEQ ID NO: 21 SEQ ID NO: 22 SEQ ID NO: 24 SEQ ID NO: 23 SEQ ID NO: 24 SEQ ID NO: 25 SEQ ID NO: 26 SEQ ID NO: 27 SEQ ID NO: 4 SEQ ID NO: 28 SEQ ID NO: 29 SEQ ID NO: 30 SEQ ID NO: 32 SEQ ID NO: 31 SEQ ID NO: 32 SEQ ID NO: 33 SEQ ID NO: 34 SEQ ID NO: 35 SEQ ID NO: 36 SEQ ID NO: 37 SEQ ID NO: 38 SEQ ID NO: 39 SEQ ID NO: 40 SEQ ID NO: 41 SEQ ID NO: 42 SEQ ID NO: 43 SEQ ID NO: 44 SEQ ID NO: 45 SEQ ID NO: 46 SEQ ID NO: 47 SEQ ID NO: 48 SEQ ID NO: 49 SEQ ID NO: 50 SEQ ID NO: 51 SEQ ID NO: 52 SEQ ID NO: 53 SEQ ID NO: 54 SEQ ID NO: 55 SEQ ID NO: 57 SEQ ID NO: 56 SEQ ID NO: 57 SEQ ID NO: 58 SEQ ID NO: 59
provided that said sequence may be a complementary sequence or a degenerate sequence thereof. (4) A TCR gene comprising the .alpha.CDR3 polynucleotide and the .beta.CDR3 polynucleotide in any one of pairs of (3). (5) The TCR gene of (4) obtainable from a CD4.sup.+ T-cell specific to WT1.sub.332 peptide. (6) A method for producing a CD4.sup.+ helper cell specific to WT1.sub.332 peptide, comprising introducing the TCR gene of (4) into a CD4.sup.+ T cell. (7) A CD4.sup.+ helper T-cell obtainable by the method of (5). (8) A vector comprising a TCR gene which comprises the .alpha.CDR3 polynucleotide and the .beta.CDR3 polynucleotide in any one of pairs of (3). (9) The method of (6) wherein said introduction is performed using the vector of (8). (10) A method for enhancing the induction of a WT1 specific CTL, comprising co-culturing the CD4.sup.+ helper T-cell of (7) and a peripheral mononuclear cell. (11) A WT1-specific CTL obtainable by the method of (10). (12) A method for the treatment or prevention of a cancer in a subject, comprising introducing the CD4.sup.+ helper T-cell of (7) into the subject. (13) A pharmaceutical composition for the treatment or prevention of a cancer, comprising the CD4.sup.+ helper T-cell of (7). (14) Use of the CD4.sup.+ helper T-cell of (7) for the manufacture of a medicine for the treatment or prevention of a cancer. (15) A DNA chip comprising the .alpha.CDR3 polynucleotide of (1), the .beta.CDR3 polynucleotide of (2), or both of the .alpha.CDR3 polynucleotide of (1) and the .beta.CDR3 polynucleotide of (2). (16) A method for measuring the frequency of CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide in a sample, comprising using the DNA chip of (15). (17) An .alpha.CDR3 peptide encoded by any one of the .alpha.CDR3 polynucleotides of (1). (18) A .beta.CDR3 peptide encoded by any one of the .beta.CDR3 polynucleotides of (2). (19) A pair of peptides encoded by any one of pairs of the polynucleotides of (3). (20) A chip comprising the peptide of (17) or (18), or the pair of the peptides of (19). (21) An antibody against any one of the peptides of any one of (17)-(19). (22) A method for measuring the frequency of CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide in a sample, comprising using the antibody of (21).
Effect of the Invention
[0028] According to the present invention, a CD4.sup.+ helper T-cell is obtained into which a TCR gene having the CDR3 nucleotide sequence determined by the present invention has been introduced. A WT1-specific CTL can be induced using said CD4.sup.+ helper T-cell, and a cancer can be treated or prevented effectively. Further, a DNA chip is prepared using the TCR sequences, and frequency of WT1.sub.332-specific CD4.sup.+ helper T-cells in a sample can be measured.
BRIEF DESCRIPTION OF DRAWINGS
[0029] FIG. 1A shows nucleotide sequences of CDR3 .alpha.-chains and .beta.-chains of TCR of CD4.sup.+ helper T-cells obtained by the present invention, and amino acid sequences of CDR3 encoded thereby. Number in parentheses positioned at the right end of each sequence presents SEQ ID NOs in SEQUENCE LISTING. V-GENE, J-GENE and J-GENE describe V region, J region and D region in individual genes, respectively.
[0030] FIG. 1B shows nucleotide sequences of CDR3 .alpha.-chains and .beta.-chains of TCR of CD4.sup.+ helper T-cells obtained by the present invention, and amino acid sequences of CDR3 encoded thereby. Number in parentheses positioned at the right end of each sequence presents SEQ ID NOs in SEQUENCE LISTING. V-GENE, J-GENE and J-GENE describe V region, J region and D region in individual genes, respectively.
[0031] FIG. 2A shows interferon-.gamma. production by WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes shown in Table 3 have been introduced.
[0032] FIG. 2B shows IL-2 production by WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes shown in Table 3 have been introduced.
[0033] FIG. 2C shows TNF-.alpha. production response of WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced, to WT1.sub.332 peptide concentration.
[0034] FIG. 2D shows proliferation ability of WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced, in systems into which several kinds of substances are added. WT1.sub.332 represents the culture in the presence of WT1.sub.332 peptide. .alpha.-DP represents the culture in the presence of an anti-HLA-DP antibody. .alpha.-DQ represents the culture in the presence of an anti-HLA-DQ antibody. .alpha.-DR represents the culture in the presence of an anti-HLA-DR antibody. HLA class I represents the culture in the presence of an anti-HLA class I antibody. HIV represents the culture in the presence of HIV peptide (FRKQNPDIVIYQYMDDLYVG) (SEQ ID NO: 124).
[0035] FIG. 2E shows proliferation of WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced, in the presence of PBMC pulsed with several kinds of substances. WT1.sub.332 represents the stimulation by autologous PBMC pulsed with WT1.sub.332 peptide. HWT1 represents the stimulation by autologous PBMC pulsed with full length of WT1 protein. HWT3 represents the stimulation by autologous PBMC pulsed with truncated WT1 protein (not containing WT1.sub.332 sequence). PHA-blast represents the stimulation by PBMC pulsed with PHA-blast lysate. TF1 represents the stimulation by PBMC pulsed with a lysate of leukemic cell line TF-1 expressing WT1. K562 represents the stimulation by PBMC pulsed with a lysate of leukemic cell line K562 expressing WT1.
[0036] FIG. 2F shows IFN-.gamma. production by WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced, in the presence of PBMC pulsed with several kinds of substances. WT1.sub.332 represents the stimulation by autologous PBMC pulsed with WT1.sub.332 peptide. HWT1 represents the stimulation by autologous PBMC pulsed with full length of WT1 protein. HWT3 represents the stimulation by autologous PBMC pulsed with truncated WT1 protein (not containing WT1.sub.332 sequence). PHA-blast represents the stimulation by PBMC pulsed with PHA-blast lysate. TF1 represents the stimulation by PBMC pulsed with a lysate of leukemic cell line TF-1 expressing WT1. K562 represents the stimulation by PBMC pulsed with a lysate of leukemic cell line K562 expressing WT1.
[0037] FIG. 2G shows the average of the production of several kinds of cytokines in response to WT1.sub.332 peptide by WT1.sub.332-specific CD4.sup.+ helper T-cell lines from three healthy subjects (HLA-DPB1*05:01-positive) into which TCR genes have been introduced. Black bars represent with the stimulation by WT1.sub.332 peptide. White bars represent without the stimulation.
[0038] FIG. 3A shows the frequency of CD3.sup.+CD8.sup.+T cells in case PBMC and WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced are co-cultured at the ratio as shown in the figure.
[0039] FIG. 3B shows the frequency of the modified WT1.sub.23/HLA-A*24:02 tetramer-positive CD8.sup.+T cells in case PBMC and WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced are co-cultured at the ratio as shown in the figure.
[0040] FIG. 3C shows the cell number of WT1-specific CTL in case PBMC and WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced are co-cultured at the ratio as shown in the figure.
[0041] FIG. 3D shows the frequency of CD8.sup.+T cells expressing interferon-.gamma. in response to the stimulation by the modified WT1.sub.235 in case PBMC and WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced are co-cultured at the ratio as shown in the figure.
[0042] FIG. 4A shows the damage as the lysis (%) of HLA-DPB1*05:01-positive leukemic cell line TF-1 expressing WT1 and HLA-DPB1*05:01-negative leukemic cell line TF-1 expressing WT1 by WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced at the E:T ratio as shown in the figure.
[0043] FIG. 4B shows the damage as the lysis (%) of HLA-DPB1*05:01-positive B-LCL cells which have been enforced to express WT1 and HLA-DPB1*05:01-positive B-LCL cells which do not express WT1 by WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced at the E:T ratio as shown in the figure.
[0044] FIG. 4C shows the damage as lysis (%) of K562 cell line and HLA-DPB1*05:01-positive leukemic cell line C2F8 expressing WT1 by WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced at the E:T ratio as shown in the figure.
[0045] FIG. 4D shows the results by flow-cytometry for the expression of GranzymeB (left) and Perfolin (right) in WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced.
[0046] FIG. 4E shows the results by flow-cytometry for the frequency of CD107a producing cells and IFN-.gamma. producing cells of WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced, cultured according to the method described in Example 4.
[0047] FIG. 4F is a bar graph showing the results for the comparison of the cell damaging activity of WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced against HLA-CPB1*05:01-positive TF-1 cells pretreated with Ac-IETD-Cho, with the cell damaging activity of WT1.sub.332-specific CD4.sup.+ helper T-cells into which TCR genes have been introduced against TF-1 cells pretreated with DMSO. Height of bars represents the average value with bars of standard deviation. Asterisk represents p<0.05.
[0048] FIG. 5 shows survival curves showing anti-tumor activity in NOG (Registered Trade Mark) mice by human CD4.sup.+ T-cells into which TCR genes from WT1.sub.332-specific CD4.sup.+ T-cells have been introduced. The solid line represents the survival curve of mice into which human CD4.sup.+ T-cells have been transferred, into which HLA-DPB1*05:01 restricted WT1.sub.332-specific TCR have been introduced. The broken line represents the survival curve of mice into which human CD4.sup.+ T-cells have been transferred, into which a control vector have been introduced.
DESCRIPTION OF EMBODIMENTS
[0049] The present invention is based on the determination of the polynucleotides encoding .alpha.-chain containing CDR3 (hereinafter referred as ".alpha.CDR3 polynucleotide") and the polynucleotides encoding .beta.-chain containing CDR3 (hereinafter referred as ".beta.CDR3 polynucleotide") of TCR of CD4.sup.+ helper T-cell clones specific to a WT1 helper peptide. Thus, in one aspect, the present invention provides .alpha.CDR3 polynucleotides having nucleotide sequences shown in FIG. 1 (nucleotide sequences selected from the group consisting of SEQ ID NOs: 1, 3, 5, 8, 10, 11, 13, 14, 16, 18, 20, 22, 23, 25, 27, 28, 30, 31, 33, 35, 37, 39, 41, 43, 45, 47, 49, 51, 53, 55, 56, 58), and .beta.CDR3 polynucleotides having nucleotide sequences shown in FIG. 1 (nucleotide sequences selected from the group consisting of SEQ ID NOs: 2, 4, 6, 7, 9, 12, 15, 17, 19, 21, 24, 26, 29, 32, 34, 36, 38, 40, 42, 44, 46, 48, 50, 52, 54, 57, 59).
[0050] Preferably, from the viewpoint of expression of receptor function, an .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide contained in each clone are contained in one TCR. That is, it is preferable that an .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide corresponding to each clone form a pair as shown in FIG. 1. Therefore, in a further aspect, the present invention provides a pair of a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide, wherein each polynucleotide constituting the pair has a nucleotide sequence shown in FIG. 1. Combination of a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide differs according to each clone. The nucleotide sequence of a pair of an .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide in each clone are as shown in FIG. 1.
[0051] A polynucleotide having a nucleotide sequence complementary to a .alpha.CDR3 polynucleotide or a .beta.CDR3 polynucleotide is also included in a .alpha.CDR3 polynucleotide or a .beta.CDR3 polynucleotide. In addition, a degenerate sequence of a .alpha.CDR3 polynucleotide or a .beta.CDR3 polynucleotide is also included in a .alpha.CDR3 polynucleotide or a .beta.CDR3 polynucleotide so long as it encodes the peptide shown in FIG. 1.
[0052] A polynucleotide having a nucleotide sequence identity of 70% or more, for example 75% or more, 80% or more, 85% or more, or 90% or more, for example 92% or more, 94% or more, 96% or more, or 98% or more, to that of a .alpha.CDR3 polynucleotide, is also included in a .alpha.CDR3 polynucleotide. A polynucleotide having a nucleotide sequence identity of 70% or more, for example 75% or more, 80% or more, 85% or more, or 90% or more, for example 92% or more, 94% or more, 96% or more, or 98% or more, to that of a .beta.CDR3 polynucleotide, is also included in a .beta.CDR3 polynucleotide.
[0053] A polynucleotide having a nucleotide sequence hybridizing to a nucleotide sequence of a .alpha.CDR3 polynucleotide under a stringent condition is also included in a .alpha.CDR3 polynucleotide. A polynucleotide having a nucleotide sequence hybridizing to a nucleotide sequence of a .beta.CDR3 polynucleotide under a stringent condition is also included in a .beta.CDR3 polynucleotide.
[0054] Examples of stringent hybridization conditions include a condition where hybridization is performed in a solution containing 5.times.SSC, 7% (w/v) SDS, 100 .mu.g/ml denatured salmon sperm DNA and 5.times.Denhardt' solution at 48-52.degree. C., and then washing is performed in 0.1.times.SSC, 0.5.times.SSC, 1.times.SSC or 2.times.SSC; or a condition where hybridization is performed in a solution containing 250 mM NaCl, 25 mM trisodium citrate, 1% SDS, 50% formamide and 200 .mu.g/ml denatured salmon sperm DNA at 42.degree. C., and then washing is performed in a solution containing 15 mM NaCl, 1.5 mM trisodium citrate and 0.1% SDS.
[0055] In another aspect, the present invention provides peptides encoded by .alpha.CDR3 polynucleotides and .beta.CDR3 polynucleotides (referred as ".alpha.CDR3 peptide" and ".beta.CDR3 peptide", respectively). These peptides have the amino acid sequences shown in FIG. 1. Preferably, these peptides form a pair of a .alpha.CDR3 peptide and a .beta.CDR3 peptide corresponding to each clone as shown in FIG. 1.
[0056] In the present specification, an amino acid sequence of a peptide is expressed by conventional one-letter system or three-letter system.
[0057] A peptide encoded by variants of a .alpha.CDR3 polynucleotide or a .beta.CDR3 polynucleotide is also included in .alpha.CDR3 peptide or .beta.CDR3 peptide. A peptide having an amino acid sequence identity of 70% or more, for example 75% or more, 80% or more, 85% or more, or 90% or more, for example 92% or more, or 94% or more, to that of a .alpha.CDR3 peptide, is also included in a .alpha.CDR3 peptide. A peptide having an amino acid sequence identity of 70% or more, for example 75% or more, 80% or more, 85% or more, or 90% or more, for example 92% or more, or 94% or more, to that of a .beta.CDR3 peptide, is also included in a .beta.CDR3 peptide. In addition, a peptide having an amino acid sequence of a .alpha.CDR3 peptide in which one to several (for example, one, two, three, four or five) amino acids are substituted, deleted or added is also included in a .alpha.CDR3 peptide; and a peptide having an amino acid sequence of a .beta.CDR3 peptide in which one to several (for example, one, two, three, four or five) amino acids are substituted, deleted or added is also included in a .beta.CDR3 peptide. It is noted that these variant peptides has similar properties to those of the original .alpha.CDR3 peptides or .beta.CDR3 peptides.
[0058] These polynucleotides and polypeptides can be prepared using chemical methods and/or biological methods well-known in the art.
[0059] In the present invention, the WT1 helper peptide is a peptide having an amino acid sequence shown in SEQ ID NO: 123 (Lys Arg Tyr Phe Lys Leu Ser His Leu Gln Met His Ser Arg Lys His) or a variant amino acid sequence thereof (These peptides are referred as "WT1.sub.332 peptide"). WT1.sub.332 peptide may have a partial sequence or a variant sequence of WT1 polypeptide. A peptide consisting of an amino acid sequence shown in SEQ ID NO: 123 or a variant sequence thereof is an example of such a peptide.
[0060] It is known that WT1.sub.332 peptide has an ability to bind to a HLA-DRB1*15:01 molecule, a HLA-DPB1*09:01 molecule, HLA-DPB1*05:01 molecule, HLA-DRB1*04:05 molecule or a HLA-DRB1*15:02 molecule.
[0061] A variant sequence of the amino acid sequence shown in SEQ ID NO: 123 as above mentioned refers to an amino acid sequence shown in SEQ ID NO: 123 in which one to several (for example, one, two, three, four or five) amino acids are substituted, deleted or added. Or, a variant sequence of the amino acid sequence shown in SEQ ID NO: 123 as above mentioned refers to an amino acid sequence having an identity of 70% or more, for example 75% or more, 80% or more, 85% or more, or 90% or more, to the amino acid sequence shown in SEQ ID NO: 123. Preferably, a peptide having the amino acid sequence shown in SEQ ID NO: 123 or a variant sequence thereof has a length of 25 amino acids or less. A peptide having a variant sequence of the amino acid sequence shown in SEQ ID NO: 123 has similar properties to those of the peptide having the amino acid sequence shown in SEQ ID NO: 123.
[0062] In a further aspect, the present invention relates to a TCR gene containing a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide belonging to any pair shown in FIG. 1. Such a TCR gene may be isolated from a CD4.sup.+ T-cell specific to WT1.sub.332 peptide, or may be prepared using well-known genetic engineering technology.
[0063] In a further aspect, the present invention relates to a CD4.sup.+ helper T-cell (referred as "TCR gene introduced CD4.sup.+ helper T-cell") obtained by introducing a TCR gene containing a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide belonging to any pair shown in FIG. 1 into a CD4.sup.+ T-cell. A TCR gene introduced CD4.sup.+ helper T-cell shows WT1.sub.332-specific and HLA class II-restricted proliferation and cytokine production.
[0064] A skilled person in the art can easily introduce a TCR gene containing a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide belonging to any one of pairs shown in FIG. 1 into a CD4.sup.+ T-cell. For example, the introduction of a TCR gene can be done using various kinds of vectors, electroporation, or a gene gun, etc. A TCR gene to be introduced can be modified for the purpose such as improvement of TCR expression efficiency.
[0065] Therefore, in a further aspect, the present invention provides a vector containing a TCR gene containing a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide belonging to any pair shown in FIG. 1.
[0066] Introduction of a TCR gene may be done by inserting a .alpha.-chain gene containing a .alpha.CDR3 polynucleotide and a .beta.-chain gene containing a .beta.CDR3 polynucleotide into individual vectors, and introducing these vectors into a CD4.sup.+ T-cell.
[0067] Examples of CD4.sup.+ T-cells into which a TCR gene containing a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide is introduced include CD4.sup.+ T-cells derived from a HLA-DRB1*15:01-positive subject, a HLA-DPB1*09:01-positive subject, a HLA-DPB1*05:01-positive subject, a HLA-DRB1*04:05-positive subject or a HLA-DRB1*15:02-positive subject, but not limited to them. In addition, CD4.sup.+ T-cells may be derived from a subject having a cancer, may be derived from a subject having no cancer (a healthy subject), or may be derived from a donor for bone marrow transplantation.
[0068] In further aspect, the present invention also relates to a WT1.sub.332-specific CD4.sup.+ helper T-cell comprising a TCR gene containing a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide belonging to any pair shown in FIG. 1.
[0069] Induction of a WT1-specific CTL can be enhanced using a TCR gene-introduced CD4.sup.+ helper T-cell. Particularly, induction of WT1-specific CTL can be enhanced by co-culturing a TCR gene-introduced CD4.sup.+ helper T-cell and a peripheral mononuclear cell. Therefore, in further aspect, the present invention provides a method for enhancing the induction of a WT1-specific CTL, comprising co-culturing a TCR gene-introduced CD4.sup.+ helper T-cell and a peripheral mononuclear cell. In another aspect, the present invention relates to a WT1-specific CTL obtainable by said method.
[0070] Methods and conditions for co-culturing a TCR gene-introduced CD4.sup.+ helper T-cell and a peripheral mononuclear cell are well known in the art. Such methods can be performed either in vivo or in vitro. One kind of a TCR gene-introduced CD4.sup.+ helper T-cell may be used for enhancing the induction of a WT1-specific CTL. However, preferably two or more kinds of TCR gene-introduced CD4.sup.+ helper T-cells are used.
[0071] Examples of peripheral mononuclear cells used in the method for enhancing the induction of WT1-specific CTLs of the present invention include peripheral mononuclear cells derived from a HLA-DRB1*15:01-positive subject, a HLA-DPB1*09:01-positive subject, a HLA-DPB1*05:01-positive subject, a HLA-DRB1*04:05-positive subject or a HLA-DRB1*15:02-positive subject, but not limited to them. Preferably, the peripheral mononuclear cells and the CD4.sup.+ T-cells are those which have been obtained from a subject in which a cancer should be treated or prevented.
[0072] In the co-cultivation, it is preferable that WT1.sub.332 peptide and/or other WT1 peptides co-exist. Examples of other WT1 peptides include those which have ability to bind to a HLA-DRB1*15:01 molecule, a HLA-DPB1*09:01 molecule, a HLA-DPB1*05:01 molecule, a HLA-DRB1*04:05 molecule or a HLA-DRB1*15:02 molecule, but not limited to them.
[0073] If necessary, the WT1-specific CTLs obtained by the method mentioned above may be further cultured to make the cell numbers increase, and then administrated to a subject, in order to treat or prevent a cancer in the subject. In such a treatment or prevention of a cancer, it is preferable to co-administrate WT1.sub.332 peptide and/or other WT1 peptides. By the action of the WT1-specific CTL, CTLs specific to other cancer antigens can also be induced.
[0074] A TCR gene-introduced CD4.sup.+ helper T-cell can damage cancer cells expressing WT1. Therefore, in further aspect, the present invention is a method for the treatment or prevention of a cancer in a subject, comprising introducing a TCR gene-introduced CD4.sup.+ helper T-cell into the subject.
[0075] In further aspect, the present invention provides a pharmaceutical composition comprising a TCR gene-introduced CD4.sup.+ helper T-cell for the treatment or prevention of a cancer, a use of a TCR gene-introduced CD4.sup.+ helper T-cell for the manufacture of a medicament for the treatment or prevention of a cancer, and a use of a TCR gene-introduced CD4.sup.+ helper T-cell for the treatment or prevention of a cancer.
[0076] As used herein, "treatment" of a cancer refers not only to the treatment of a cancer such as the inhibition of progress of a cancer, the reduction of a cancer and the destruction of a cancer, but also to the prevention of recurrence of a cancer.
[0077] Examples of subjects in which a cancer is treated or prevented include a HLA-DRB1*15:01-positive subject, a HLA-DPB1*09:01-positive subject, a HLA-DPB1*05:01-positive subject, a HLA-DRB1*04:05-positive subject or a HLA-DRB1*15:02-positive subject, but not limited to them. The above subject is not limited to a cancer patient, and may be a person not having a cancer (including a healthy person), or may be a donor for bone marrow transplantation.
[0078] Embodiments of the method for the treatment or prevention, the pharmaceutical composition, and the use as mentioned above are described below. However, the embodiments are not limited to those. First, CD4.sup.+ T-cells are taken from a peripheral blood of a cancer patient who needs a treatment, and a TCR gene containing a .alpha.CDR3 polynucleotide and a .beta.CDR3 polynucleotide is introduced into the CD4.sup.+ T-cells to obtain TCR gene-introduced CD4.sup.+ helper T-cells. The TCR gene-introduced CD4.sup.+ helper T-cells thus obtained are administered to the cancer patient. Before the administration, the TCR gene-introduced CD4.sup.+ helper T-cells can be cultured and proliferated under appropriate conditions to obtain a sufficient number of cells, and then they can be administered to the cancer patient.
[0079] Either one kind of TCR gene-introduced CD4.sup.+ helper T-cell or two or more kinds of TCR gene-introduced CD4.sup.+ helper T-cells may be administered. From the viewpoint of improvement of treatment or prevention effect, it is preferable that two or more kinds of TCR gene-introduced CD4.sup.+ helper T-cells are administered to a subject.
[0080] In case that TCR gene-introduced CD4.sup.+ helper T-cells are administered to a subject, a physician can appropriately decide conditions such as the number of cells to be administered, the frequency of the administration, the interval of the administration. For example, TCR gene-introduced CD4.sup.+ helper T-cells may be administered only once, or administered separately several times. Typically, in case of an adult subject, the number of the TCR gene-introduced CD4.sup.+ helper T-cells per dose is in a range between about 10.sup.9 and about 10.sup.11, but not limited these numbers.
[0081] In the treatment or prevention method, the pharmaceutical composition and the use described above, it is preferable that WT1.sub.332 peptide and/or other WT1 peptides are co-administered. A physician can appropriately decide amount and frequency of administration of WT1.sub.332 peptide and/or other WT1 peptides. In addition, other anti-cancer therapies or preventions may be combined.
[0082] The method for the treatment or prevention, the pharmaceutical composition, and the use described above can be applied to various kinds of cancers, but not limited to, for example, hematologic malignancies, such as acute myelocytic leukemia, acute lymphocytic leukemia, malignant lymphoma, multiple myeloma, chronic myelocytic leukemia, myelodysplastic syndrome, and recurrence after the hematopoietic stem cell transplantation of the same type; solid cancers, such as tongue cancer, gingival cancer, mouth floor cancer, pharyngeal cancer, larynx cancer, salivary gland cancer, and thyroid cancer; thoracic cancers, such as breast cancer, lung cancer, and thymic cancer; gastrointestinal cancers, such as colon cancer, small intestine cancer, gastric cancer, pancreatic cancer, liver cancer, bile duct cancer, gastrointestinal endocrine tumor, and gastrointestinal carcinoid; cancers of urinary and genital tract, such as renal cancer, urothelial cancer, germinoma, Wilms' tumor, prostate cancer, uterine body cancer, cervical cancer, uterine sarcoma, and ovarian malignancy; musculoskeletal malignancies, such as primary malignancy of bone (e.g., osteosarcoma and Ewing's sarcoma) and soft tissue sarcoma; and other cancers, such as skin cancer, neuroblastoma, malignant glioma (glioblastoma), primary malignant lymphoma of the central nervous system, medulloblastoma, and PNET.
[0083] The CDR3 regions are the most diverse portions and are the most responsible parts for the specificity of antigen recognition. Thus, the sequences of the .alpha.CDR3 polynucleotides, the .beta.CDR3 polynucleotides, the .alpha.CDR3 peptides, and the .beta.CDR3 peptides of the present invention are considered peculiar to the CD4.sup.+ helper T-cells specific to WT1.sub.332 peptide. Therefore, in case that a polynucleotide encoding a CDR region of a .alpha.-chain and a .beta.-chain, or a peptide corresponding to the CDR region have the sequence of the polynucleotide or the peptide of the present invention, the CD4.sup.+ helper T-cell is considered to be specific to WT1.sub.332 peptide.
[0084] For example, (i) a DNA chip comprising one or more kinds of .alpha.CDR3 polynucleotides, (ii) a DNA chip comprising one or more kinds of .beta.CDR3 polynucleotides, or (iii) a DNA chip comprising both one or more kinds of .alpha.CDR3 polynucleotides and one or more kinds of .beta.CDR3 polynucleotides can be used to measure the frequency of CD4.sup.+ helper T-cells specific to WT1.sub.332 peptide in a sample. Particularly, a sample is prepared by lysing cells in a specimen obtained from a subject and extracting nucleic acids, and the sample is contacted with the DNA chip.
[0085] For example, in case that a sample is contacted with the chip (i), and hybridization is found at any position, the same sample is contacted with the chip (ii) to confirm whether hybridization is found or not. Then, in case that any hybridization in the chip (i) and any hybridization in the chip (ii) occur with any .alpha.CDR3 polynucleotide and any .beta.CDR3 polynucleotide which constitute any pair shown in FIG. 1, it can be judged that a CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide having a functional TCR exists in the sample. Using the chip (III), the above process can be done in one step.
[0086] A DNA chip may be in any form such as a microchip and a microarray. These chips can be prepared by a well-known method. For example, .alpha.CDR3 polynucleotides and .beta.CDR3 polynucleotides can be immobilized on a glass substrate by a well-known method. It is preferable that a label which can indicate presence or absence of hybridization and amount of the hybridization is attached to DNAs in a sample or DNA sequences on a chip.
[0087] Not only a DNA chip but also techniques such as southern blotting, northern blotting, colony hybridization can be used to measure frequency of CD4.sup.+ helper T-cells specific to WT1.sub.332 peptide in a sample.
[0088] In addition, a .alpha.CDR3 peptide and a .beta.CDR3 peptide can be used to obtain an antibody to a CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide. A CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide can be detected using such an antibody. A receptor of a CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide can also be stimulated using such an antibody. Such stimulation can be done either in vivo or in vitro.
[0089] A chip comprising .alpha.CDR3 peptides, a chip comprising .beta.CDR3 peptides, or a chip comprising both .alpha.CDR3 peptides and .beta.CDR3 peptides can also be used to detect an antibody to a CD4.sup.+ helper T-cell specific to WT1.sub.332 peptide.
[0090] A chip comprising these peptides can be prepared using a well-known method. It is preferable to add a label which can determine presence or absence of a specific binding to peptides in a sample or peptides on a chip.
[0091] A chip comprising antibodies to .alpha.CDR3 peptides and/or .beta.CDR3 peptides can also be used to determine kind and amount of .alpha.CDR3 peptides and/or .beta.CDR3 peptides in a sample, or to determine kind and amount of CD4.sup.+ helper T-cells specific to WT1.sub.332 peptide in a sample.
[0092] A chip to which these antibodies are immobilized can be prepared using a well-known method. It is preferable to add a label which can determine presence or absence of a specific binding to peptides in a sample or antibodies on a chip.
[0093] Description of Sequences
SEQ ID NOs: 1 to 59 are nucleotide sequences encoding CDR3 contained in TCR of CD4.sup.+ helper T-cell clones. SEQ ID NOs: 60 to 118 are amino acid sequences of CDR3 contained in TCR of CD4.sup.+ helper T-cell clones. SEQ ID NO: 119 is a reverse primer for amplifying TCR.alpha. chain. SEQ ID NO: 120 is a reverse primer for amplifying TCR.beta. chain. SEQ ID NO: 121 is a reverse primer for amplifying TCR.beta. chain. SEQ ID NO: 122 is a primer for determining CDR3 nucleotide sequences. SEQ ID NO: 123 is an amino acid sequence of WT1.sub.332 peptide. SEQ ID NO: 124 is an amino acid sequence of HIV peptide. SEQ ID NO: 125 is an amino acid sequence of a variant of a naturally occurring WT1 peptide.
[0094] The present invention is described more particularly and more concretely by showing examples below. However, it should not be construed that examples limit the scope of the present invention.
Example 1
Example 1 Establishment of WT1.sub.332-Specific CD4.sup.+ T-Cell Clones and Isolation and Sequencing of T-Cell Receptor (TCR) Genes
[0095] The experimental procedures were as follows.
[0096] (1) Method of Establishing WT1.sub.332-Specific CD4.sup.+ T-Cell Clones
[0097] (i) Peripheral blood mononuclear cells (PBMCs) derived from a healthy subject are harvested and seeded into 24-well plates at 3.times.106 cells/well. X-VIVO 15 medium supplemented with 10% AB serum and 40 IU/ml IL-2 is used as a medium.
[0098] (ii) WT1.sub.332 peptide is added to the above i at a final concentration of 20 .mu.g/ml and the cells are cultured for 7 days.
[0099] (iii) After 7 days, the cells are collected and prepared with X-VIVO 15 medium supplemented with 10% AB serum so that the cell density is 1.times.107 cells/ml, and then, seeded by 100 .mu.L each into 96 well, round bottom plates.
[0100] (iv) WT1.sub.332 peptide, BD GolgiStop.TM. (BD Bioscience) and CD28/CD49d Costimulatory Reagent (BD Bioscience) are added to X-VIVO 15 medium supplemented with 10% AB serum at final concentrations of 40 .mu.g/ml, 4 .mu.g/ml, and 4 .mu.g/ml, respectively.
[0101] (v) The above iv is added by 100 .mu.L each to the above iii.
[0102] (vi) Anti-human CD154-APC-labeled antibody (BD Bioscience) is added by 10 .mu.l each to the above v and the plates are incubated in 5% CO.sub.2 incubator for 6 hours at 37.degree. C.
[0103] (vii) After incubation, the cells are collected and stained with anti-human CD4-APC-H7-labeled antibody (BD Bioscience) and anti-human CD3-Pacific Blue-labeled antibody (BD Bioscience) as well as 7-AAD (eBioscience) for removing dead cells.
[0104] (viii) PBMCs are harvested from 3 healthy subjects, mixed, irradiated with 30 Gy of .gamma.-ray, and prepared with X-VIVO 15 medium supplemented with 10% AB serum at a final concentration of 10%, IL-2 at a final concentration of 100 IU/ml, and PHA at a final concentration of 3 .mu.g/ml so that the cell density is 1.times.106 cells/ml. These prepared cells are seeded by 100 .mu.L each into 96 well, round bottom plates.
[0105] (ix) 7-ADD-CD3.sup.+CD4.sup.+CD154.sup.+ cell fraction, i.e., a fraction containing WT1.sub.332-specific CD4.sup.+ T-cells is single-cell sorted into each well of the above viii using FACSAria cell sorter.
[0106] (x) After culture for 10-14 days, the proliferated cells in each well are used as independent CD4.sup.+ T-cell clones.
[0107] (2) Screening of WT1.sub.332-Specific CD4.sup.+ T-Cell Clones
[0108] (i) Each CD4.sup.+ T-cell clone of the above (1)-x is prepared with X-VIVO 15 medium supplemented with 1% AB serum so that the cell density is 3.times.105 cells/ml.
[0109] (ii) Autologous PBMCs pulsed with WT1.sub.332 or not pulsed with any peptides are irradiated with 30 Gy of .gamma.-ray and prepared with X-VIVO 15 medium supplemented with 1% AB serum so that the cell density is 1.times.106 cells/ml.
[0110] (iii) The above (2)-i and ii are seeded by 100 .mu.L each into 96 well, round bottom plates.
[0111] (iv) After culture for 2 days, 3H-thymidine is added to each well at 1 .mu.Ci/well.
[0112] (v) After 18 hours, the 3H-thymidine incorporated into each CD4.sup.+ T-cell clone is measured and the CD4.sup.+ T-cell clones showing WT1.sub.332-specific proliferative response are selected. These selected clones are used as WT1.sub.332-specific CD4.sup.+ T-cell clones.
[0113] (vi) The culture of the WT1.sub.332-specific CD4.sup.+ T-cell clones is performed with stimulation of the WT1.sub.332-specific CD4.sup.+ T-cell clones by co-culturing with PBMCs that were prepared by irradiating autologous PBMCs pulsed with WT1.sub.332 at a frequency of once per 1-2 weeks or so with 30 Gy of .gamma.-ray.
[0114] (3) Isolation of TCR Genes Using 5'-RACE (Rapid Amplification of cDNA End) Method
[0115] (i) WT1.sub.332-specific CD4.sup.+ T-cell clones are cultured for 10 days or more from the last stimulation. This is to prevent contamination with T-cells contained in autologous PBMCs that are used for the stimulation.
[0116] (ii) The WT1.sub.332-specific CD4.sup.+ T-cell clones are pelleted, TRIzol reagent (Invitrogen) is added thereto, and RNA is extracted according to its manual.
[0117] (iii) cDNAs are synthesized from the RNA extracted in the above (3)-ii using SMARTer.TM. RACE cDNA Amplification Kit (Clontech).
[0118] (iv) TCR .alpha.-chain and .beta.-chain genes are amplified by using the cDNAs synthesized in the above (3)-iii as templates. In regard to used primers, UPM primer included in MARTer.TM. RACE cDNA Amplification Kit was used as a forward primer and the following TCR-specific primers were used as reverse primers:
TABLE-US-00002 C.alpha.3'UTR-primer: (SEQ ID No: 119) 5'-CAC AGG CTG TCT TAC AAT CTT GCA GAT C-3' C.beta.1-3UTR-primer: (SEQ ID No: 120) 5'-CTC CAC TTC CAG GGC TGC CTT CA-3' C.beta.2-3UTR-primer: (SEQ ID No: 121) 5'-TGA CCT GGG ATG GTT TTG GAG CTA-3'.
[0119] (v) The amplification of the TCR genes was performed using KOD FX available from ToYoBo under conditions of 94.degree. C., 3 min->(98.degree. C., 10 sec->68.degree. C., 1 min).times.35 cycles.
[0120] (vi) The size of PCR products are confirmed using agarose gel electrophoresis and bands of near 1 kbp are cut from gel and purified.
[0121] (vii) After adenines are added to the PCR products purified in the above (3)-vi using Taq polymerase, the resultants are ligated into pCR 2.1 vectors.
[0122] (viii) HST02 competent cells are transformed with the above (3)-vii, plasmids are purified from single colonies, and then, sequenced.
[0123] (ix) The sequence analysis is performed using the International Immunogenetics Information System (http://www.imgt.org/IMGT_vquest/vquest?livret=0&Option=humanTcR) and each TCR gene is identified.
[0124] With regard to the above (3) "isolation of TCR genes using 5'-RACE (Rapid Amplification of cDNA End) method", the detailed experimental procedure is shown below.
[0125] (3-1) RNA Extraction
[0126] RNA extraction from T-cell clones was performed using TRIzol Reagent (Invitrogen). As for T-cell clones used, the clones cultured without antigen-stimulation in the presence of IL-2 over 3 weeks were prepared for the purpose of preventing contamination with feeder cells.
[0127] (3-2) Cloning of Full-Length TCR (T-Cell Receptor) cDNA Using 5'-RACE (Rapid Amplification of cDNA Ends) Method
[0128] For cloning of TCR .alpha./.beta., SMARTer.TM. RACE cDNA Amplification Kit (Clontech) was used. Firstly, 5'-RACE reaction was performed according to its manual, and thereby 1st strand cDNA was synthesized. Then, in order to obtain full-length TCR .alpha.-chain and .beta.-chain cDNAs, PCR reaction was performed by using reverse primers specific to each of 3'UTRs (Untranslated Regions) and universal primer (UPM) which is included in the kit and using the synthesized 1st strand cDNA as a template. The used primers are as follows.
TABLE-US-00003 C.alpha. 3'UTR-RACE-primer: (SEQ ID No: 119) CACAGGCTGTCTTACAATCTTGCAGATC C.beta.1 3'UTR-RACE-primer: (SEQ ID No: 120) CTCCACTTCCAGGGCTGCCTTCA c.beta.2 3'UTR-RACE-primer: (SEQ ID No: 121) TGACCTGGGATGGTTTTGGAGCTA
[0129] Further, PCR reaction was performed in the following reaction solution composition using KOD FX (TOYOBO).
TABLE-US-00004 TABLE 1 2x PCR buffer for KOD FX 12.5 .mu.l 2 mM ciNTPs 5.0 .mu.l 10x UPM 2.5 .mu.l 10 .mu.M reverse primer 1.0 .mu.l Template DNA 1.0 .mu.l KOD FX (1.0 U/.mu.l) 0.5 .mu.l distilled water up to 25 .mu.l Volume of the reaction solution 25 .mu.l PCR cycle is as follows: 94.degree. C., 2 min -> (98.degree. C., 10 sec -> 68.degree. C., 1 min) .times. 35 cycles ->15.degree. C., hold
[0130] After the PCR reaction, 1.0% agarose gel electrophoresis was performed, single bands of near 900-1000 bp were cut, and the PCR products were purified with 50 .mu.l of distilled water using QIAquick Gel Extraction Kit (QIAGEN). It is necessary to add adenine to both ends of the PCR products for TA-cloning. The addition of adenine was performed using Platinum Taq DNA polymerase (invitrogen) as follows.
[0131] (1) 2.times. reaction solution shown in the table below is prepared.
TABLE-US-00005 TABLE 2 10x PCR buffer 10 .mu.l 2 mM dNTPs 10 .mu.l 25 mM MgCl2 8.0 .mu.l Platinum Taq polymerase 1.0 .mu.l 21 .mu.l of distilled water is added so that a total volume is 50 .mu.l.
[0132] (2) 2.times. reaction solution is incubated at 95.degree. C. for 5 minutes.
[0133] (3) The purified PCR products are added thereto.
[0134] (4) The resultants are incubated at 72.degree. C. for 10 minutes.
[0135] PCR products added with adenines were purified and concentrated by ethanol precipitation, and then, inserted into pCR 2.1 vectors (invitrogen) using DNA Ligation Kit, Mighty Mix> (TaKaRa). pCR 2.1 vectors comprising the PCR products were introduced into HST02 competent cells by transformation and cloned.
[0136] (3-3) Purification of Plasmids Comprising Full-Length TCR .alpha.-Chain and .beta.-Chain cDNAs
[0137] The transformed HST02 competent cells were plated on ampicillin/LB plates and incubated at 37.degree. C. Then, single colonies were picked into ampicillin/LB liquid medium, and incubated 37.degree. C. while being stirred at 200 rpm. Then, plasmids were purified from the Escherichia coli solution using AUTOMATIC DNA ISOLATION SYSTEM PI-50 (KURABO).
[0138] (3-4) Determination of CDR3 Sequence of TCR by Sequencing
[0139] For sequencing of the purified plasmids, BigDye.RTM. Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems) was used. In addition, M13 reverse primer: caggaaacagctatgac (SEQ ID No: 122) was used. For analysis of TCR and CD3, IMGT/V-QUEST (http://www.imgt.org/IMGT_vquest/share/textes/) was utilized.
[0140] The determined nucleotide and amino acid sequences of CDR3 are shown in FIG. 1. In some of the clones, there were 2 kinds of .alpha.-chain and 2 kinds of CDR 3 sequences.
Example 2
Example 2 Introduction of T-Cell Receptor (TCR) Genes Derived from a WT1.sub.332-Specific CD4.sup.+ T-Cell to Human CD4.sup.+ T-Cells
[0141] It was confirmed that human CD4.sup.+ T-cell transduced with T-cell receptor (TCR) genes derived from a WT1.sub.332-specific CD4.sup.+ T-cell showed proliferative response and production of cytokines in a WT1.sub.332-specific and HLA class II-restricted manner.
[0142] TCR genes shown in Table 3 were isolated from clone 9 which is the CD4.sup.+ T-cell clone which specifically recognizes WT1.sub.332 in an HLA-DPB1*05:01-restricted manner. These TCR genes were transduced into CD4.sup.+ T-cells derived from peripheral blood of healthy subjects by using lentivirus vectors, and the response to WT1.sub.332 was examined by using the productions of cytokines (interferon-.gamma. and IL-2) as indicators (FIGS. 2A and B). In addition, CD4.sup.+ T-cells transduced with lentivirus vectors not carrying TCR genes (indicated as mock) were used as a control. CD4.sup.+ T-cells transduced with WT1.sub.332-specific TCR genes (referred as "WT1.sub.332-TCR-transduced CD4.sup.+ T-cells" in the section of Examples) produced INF-.gamma. and IL-2 in response only to WT1.sub.332, i.e., in a WT1.sub.332-specific manner. On the other hand, the mock-transduced CD4.sup.+ T-cells did not show WT1.sub.332-specific production of cytokines.
[0143] The effect of the concentration of WT1.sub.332 peptide on the expression of cytokine by WT1.sub.332-TCR-transduced CD4.sup.+ T-cells was examined. WT1.sub.332-TCR-transduced CD4.sup.+ T-cells were stimulated with various concentrations of WT1.sub.332 peptide for 4 hours, and intracellular cytokine staining assay was performed to examine the ratio of TNF-.alpha.-producing CD4.sup.+ T-cells to CD4.sup.+ T-cells. The results are shown in FIG. 2C. The production of the cytokine was WT1.sub.332 peptide concentration-dependent and ED50 was 4.85 .mu.M.
[0144] When a proliferation potency of the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells was examined, a WT1.sub.332-specific, strong proliferation potency was found and the proliferative response was markedly inhibited by anti-HLA-DP antibody (FIG. 2D).
[0145] Next, the proliferative response and IFN-.gamma. production of the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells to autologous PBMCs pulsed with WT1.sub.332 peptide, autologous PBMCs pulsed with full-length WT1 protein, autologous PBMCs pulsed with truncated WT1 protein (not comprising WT1.sub.332 sequence), PBMCs pulsed with the lysate of PHA-blast, PBMCs pulsed with the lysate of leukemia cell line TF-1 expressing WT1, and PBMCs pulsed with the lysate of leukemia cell line K562 expressing WT1 were examined. The cell proliferation was measured by [3H]-thymidine incorporation, and IFN-.gamma. was measured by ELISA. The results are shown in FIG. 2E and FIG. 2F, respectively. It was found that the proliferation and IFN-.gamma. production of the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells were markedly stimulated by PBMCs pulsed with the lysate of leukemia cell lines (TF-1 and K562) expressing WT1 and also stimulated by autologous PBMCs pulsed with WT1.sub.332 peptide and autologous PBMCs pulsed with full-length WT1 protein.
[0146] Further, the production of various cytokines that responded to WT1.sub.332 peptides of WT1.sub.332-TCR-transduced CD4.sup.+ T-cell lines prepared similarly to those of Example 2, that were derived from 3 healthy (HLA-DPB1*05:01 positive) donors (i.e., three kinds of cell lines), was also examined. The mean values of the cytokine-producing abilities of the three kinds of cell lines are shown in FIG. 2G. Th1-type cytokines such as IL-2, IFN-.gamma., TNF-.alpha. and GM-CSF were produced in large amount.
TABLE-US-00006 TABLE 3 TCR genes isolated form clone 9 V J D gene segment gene CDR3 sequence Va TRAV13- TRAJ - CAENSGGSNYKLTF 8.2 2*01 53*01 (SEQ ID No:73) Vb TRAB6- TRBJ1- TRBD0 CASTAGASDQPQHF 13.3 1*01 5*01 1*01 (SEQ ID No:74)
Example 3
Example 3 Enhanced Induction of WT1-Specific CTLs by Human CD4.sup.+ T-Cells Transduced with TCR Genes Derived from WT1.sub.332-Specific CD4.sup.+ T-Cells
[0147] Generally, it is known that CD4.sup.+ T-cell serves as helper T-cell and is important for introduction and maintenance of CD8.sup.+ T-cells (CTLs) that are the primary effector cells that attack cancer cells. Thus, it was examined whether WT1.sub.332-TCR-transduced CD4.sup.+ T-cells enhanced the induction of WT1-specific CTLs.
[0148] PBMCs of HLA-A*24:02 and HLA-DPB1*05:01-positive healthy subjects were mixed with WT1.sub.332-TCR-transduced CD4.sup.+ T-cells prepared from the same healthy subjects at the ratio of 10:1 and 5:1 (indicated as 1:0.1 and 1:0.2 in FIG. 3) and incubated for 1 week in the presence of a modified WT1.sub.235 peptide (wherein M, the second amino acid of natural WT1 peptide binding to HLA-A*24:02 molecule, was modified into Y (CYTWNQMNL) (SEQ ID No:125)) that is an HLA-A*24:02-restricted CTL epitope derived from WT1 and WT1.sub.332. Then, the resultant was stimulated again with the modified WT1.sub.235 peptide (wherein the binding ability to HLA-A*24:02 molecule were enhanced) and further incubated for 1 week. No IL-2 was added in a series of cultures in order to correctly evaluate the help activity of CD4.sup.+ T-cells. After 2 weeks cultures in total, it was examined whether the induction of WT1-specific CTLs was enhanced by the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells by using frequencies of CD8.sup.+ T-cells, modified WT1.sub.235/HLA-A*24:02 tetramer-positive CD8.sup.+ T-cells, and modified WT1.sub.235-specific interferon-.gamma. (INF-.gamma.)-expressing CD8.sup.+ T-cells as indicators. As a result, the frequency of CD8.sup.+ T-cells was significantly higher when co-cultured with the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells as compared when cultured with the mock-transduced CD4.sup.+ T-cells as a control (FIG. 3A). In addition, in regard to the modified WT1.sub.235/HLA-A*24:02 tetramer positive CD8.sup.+ T-cells that are WT1-specific CTLs, a clearly positive population was found when co-cultured with the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells, however, it was not found in the control (FIG. 3B). Calculating the cell number of the WT1-specific CTLs present in 100,000 lymphocytes from these results, the cell number was about 28 times higher when co-cultured with the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells compared with the control (FIG. 3C). Likewise, the frequency of the CD8.sup.+ T-cells expressing INF-.gamma. by the stimulation with the modified WT1.sub.235 was also significantly high when co-cultured with the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells (FIG. 3D). From the above, it was revealed that the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells enhanced the induction of WT1-specific CTLs.
Example 4
Example 4 HLA-DPB1*05:01-Restricted Damage of WT1-Expressing Leukemia Cells by Human CD4.sup.+ T-Cells Transduced with TCR Genes Derived from WT1.sub.332-Specific CD4.sup.+ T-Cells
[0149] Next, the cytotoxic activity, i.e., killing activity, of WT1.sub.332-TCR-transduced CD4.sup.+ T-cells was evaluated.
[0150] Firstly, HLA-DPB1*05:01 gene was isolated and transfected into leukemia cell line TF-1 expressing WT1 to prepare HLA-DPB1*05:01-positive TF-1 cells. As shown in FIG. 4A, the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells strongly damaged HLA-DPB1*05:01-positive TF-1 cells, however, they did not exhibit cytotoxic activity on HLA-DPB1*05:01-negative TF-1 cells. Then, in order to confirm whether this cytotoxic activity is WT1-specific, B-LCL(+) was prepared by overexpressing WT1 gene in HLA-DPB1*05:01-positive B-LCL cells not expressing WT1 (indicated as B-LCL(-)), and these cells were used as target cells to evaluate the cytotoxic activity of WT1.sub.332-TCR-transduced CD4.sup.+ T-cells. As shown in FIG. 4B, B-LCL(+) was strongly damaged by the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells, however, B-LCL(-) was not damaged. From these results, it was revealed that the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells had the HLA-DPB1*05:01-restricted and WT1-specific cytotoxic activity. Further, the cytotoxic activity of the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells was confirmed by using leukemia cell line C2F8 which was HLA-DPB1*05:01-positive and expressed WT1 (FIG. 4C).
[0151] Next, it was examined whether WT1.sub.332-TCR-transduced CD4.sup.+ T-cells exerted the cytotoxic activity via the granzyme B and perforin pathway. High expressions of granzyme B and perforin were found in the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells (FIG. 4D).
[0152] WT1.sub.332-TCR-transduced CD4.sup.+ T-cells and CD4.sup.+ T-cells similarly treated with mock-vector (mock-transduced CD4.sup.+ T-cells) were cultured with HLA-DPB1*05:01-positive TF-1 cells pulsed with WT1.sub.332 peptide or HLA-DPB1*05:01-positive TF-1 cells not pulsed with WT1.sub.332 peptide for 5 hours in the presence of anti-CD107a-APC-monoclonal antibody. Then, IFN-.gamma.-staining was performed and the resultants were subjected to Flow cytometry. The co-expression of IFN-.gamma. and CD107a was found in the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells only when the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells were incubated with HLA-DPB1*05:01-positive TF-1 cells pulsed with WT1.sub.332 peptide (FIG. 4E). This shows that the degranulation occurs in the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells.
[0153] To confirm whether the cytotoxic activity of the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells was dependent on the granzyme B/perforin pathway, HLA-DPB1*05:01-positive TF-1 cells pretreated with 100 .mu.M of a granzyme-inhibitor, Ac-IETD-Cho, were used as target cells. The HLA-DPB1*05:01-positive TF-1 cells were pretreated with 100 .mu.M of Ac-IETD-Cho or DMSO (control) for 2 hours, then labeled with .sup.51Cr, and incubated with WT1.sub.332-TCR-transduced CD4.sup.+ T-cells, and .sup.51Cr releasing assay was performed. The cytotoxic activity of the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells on HLA-DPB1*05:01-positive TF-1 cells pretreated with Ac-IETD-Cho was markedly lower compared with the cytotoxic activity on TF-1 cells pretreated with DMSO (FIG. 4F).
[0154] Considering these results together, it was confirmed that the WT1.sub.332-TCR-transduced CD4.sup.+ T-cells obtained by the present invention directly recognized HLA-DPB1*05:01-positive leukemia cells expressing WT1 and damaged them via granzyme B/perforin pathway.
Example 5
Example 5 Anti-Tumor Effect in NOG.RTM. Mouse by Human CD4.sup.+ T-Cells Transduced with TCR Genes Derived from WT1.sub.332-Specific CD4.sup.+ T-Cells
[0155] WT1-expressing HLA-DPB1*05:01-positive human leukemia cells C2F8 (5.times.104 cells) were transferred to NOG.RTM. mice (7 mice) via tail vein. Next day, as an experimental group, human CD4.sup.+ T-cells transduced with HLA-DPB1*05:01-restricted WT1.sub.332-specific TCR genes (SEQ ID Nos: 14 and 15) (5.times.106 cells) and T-cell-depleted human peripheral blood mononuclear cells (2.times.106 cells) from the same subject as antigen-presenting cells were transferred to the above NOG.RTM. mice (3 mice). As a control, human CD4.sup.+ T-cells transduced with the control vector (5.times.106 cells) and T-cell-depleted human peripheral blood mononuclear cells (2.times.106 cells) from the same subject as antigen-presenting cells were transferred to the above NOG.RTM. mice (4 mice).
[0156] After 1 and 2 weeks, human CD4.sup.+ T-cells transduced with HLA-DPB1*05:01-restricted WT1.sub.332-specific TCR genes (5.times.106 cells) were transferred to the mice of the experimental group via tail vein. Human CD4.sup.+ T-cells transduced with control vector (5.times.106 cells) were transferred to the control mice via tail vein. Then, the survival of mice was examined.
[0157] The results are shown in FIG. 5. Since the survival rate of the mice of the experimental group exceeded the survival rate of the mice of control, it was shown that HLA-DPB1*05:01-restricted WT1.sub.332-specific TCR-transduced human CD4.sup.+ T-cells had an anti-tumor effect in vivo.
INDUSTRIAL APPLICABILITY
[0158] The present invention can be used in the fields of pharmaceuticals for treating or preventing cancer, of reagents for cancer research, and of cancer test kits or reagents, and the like.
Sequence CWU 1
1
125142DNAHomo sapiens 1tgtgctgtgg gtagctctag caacacaggc aaactaatct
tt 42251DNAHomo sapiens 2tgcgccagca gccaagacgc tatagggaca
ggggttttga aactgttttt t 51336DNAHomo sapiens 3tgtgctgctt cgtttggaaa
tgagaaatta accttt 36445DNAHomo sapiens 4tgtgccagca gcgcctcgga
cagggagacg tatggctaca ccttc 45542DNAHomo sapiens 5tgtgcggaag
ctggtggtac tagctatgga aagctgacat tt 42642DNAHomo sapiens
6tgtgccagct gtcggactag ctcctacaat gagcagttct tc 42739DNAHomo
sapiens 7tgtgccagca gcttcactag cacagatacg cagtatttt 39839DNAHomo
sapiens 8tgtgctgccc cctctggcaa cacaggcaaa ctaatcttt 39942DNAHomo
sapiens 9tgtgccagca caatagcggg gggggaggat acgcagtatt tt
421030DNAHomo sapiens 10tgtgccgtga ccggtaacca gttctatttt
301142DNAHomo sapiens 11tgtgcagcaa gtgtgggatc agatggccag aagctgctct
tt 421245DNAHomo sapiens 12tgtgccagca gtttcttccg cagggacggg
gagacccagt acttc 451354DNAHomo sapiens 13tgtgttgtga gtgacaatcg
tgctggtggt actagctatg gaaagctgac attt 541442DNAHomo sapiens
14tgtgcagaga atagtggagg tagcaactat aaactgacat tt 421542DNAHomo
sapiens 15tgtgccagca cggcaggggc gagcgatcag ccccagcatt tt
421639DNAHomo sapiens 16tgtgcctccc ggaataatgc aggcaacatg ctcaccttt
391748DNAHomo sapiens 17tgtgccagca gttactcgaa ccgcgggtat aattcacccc
tccacttt 481836DNAHomo sapiens 18tgtgctacgg acgagggaaa tgagaaatta
accttt 361942DNAHomo sapiens 19tgtgccagca gcttagtcgg cgtctcctac
gagcagtact tc 422039DNAHomo sapiens 20tgtgcagagg ctgtctatgg
gaacaacaga ctcgctttt 392142DNAHomo sapiens 21tgtgccagtg acaggacagg
tagcgacact gaagctttct tt 422236DNAHomo sapiens 22tgtgctgtgg
ataactatgg tcagaatttt gtcttt 362345DNAHomo sapiens 23tgcatcgtca
agggtggggg atacaacttc aacaaatttt acttt 452442DNAHomo sapiens
24tgtgccagta gtatgggggt cggggacact gaagctttct tt 422539DNAHomo
sapiens 25tgtgctacgg acgggggggg agcccagaag ctggtattt 392642DNAHomo
sapiens 26tgtgccagca ccggacaggg gatcggaaac accatatatt tt
422742DNAHomo sapiens 27tgtgctctat atagtggagg tagcaactat aaactgacat
tt 422836DNAHomo sapiens 28tgtgcaatga gtcttaacga ctacaagctc agcttt
362936DNAHomo sapiens 29tgcagcgcaa acgacgggac agatacgcag tatttt
363036DNAHomo sapiens 30tgtgcagccc cagggggaga tgacaagatc atcttt
363142DNAHomo sapiens 31tgtgctctag cccaaactga cagctggggg aaattccagt
tt 423242DNAHomo sapiens 32tgtgccagca gcttaccagg acaggcctac
gagcagtact tc 423351DNAHomo sapiens 33tgtgctgtga gtgatcaaaa
actcacggga ggaggaaaca aactcacctt t 513451DNAHomo sapiens
34tgcagtgcta gagatcccaa cagggggtgg aacaccgggg agctgttttt t
513536DNAHomo sapiens 35tgtgctacgg actctggcac cgacaagctc atcttt
363648DNAHomo sapiens 36tgtgccagca gcttggatcg ggtggggaca gggaccgagc
agtacttc 483745DNAHomo sapiens 37tgtgcagaga gtaaacggaa aacctcctac
gacaaggtga tattt 453842DNAHomo sapiens 38tgtgccagca gcccctcaac
ggggcaagag acccagtact tc 423948DNAHomo sapiens 39tgtgctgtgc
aggccgttag tggaggtagc aactataaac tgacattt 484039DNAHomo sapiens
40tgtgcctgga gtccccaaac aacagggtta gagtacttc 394145DNAHomo sapiens
41tgtgctgtga gtgatgggga taactatggt cagaattttg tcttt 454233DNAHomo
sapiens 42tgtgccagca gcttacagat agctcagtac ttc 334342DNAHomo
sapiens 43tgtgctggcc tggagggccc aaacgactac aagctcagct tt
424442DNAHomo sapiens 44tgtgccaccg cctcacaggg gccaaaagag acccagtact
tc 424533DNAHomo sapiens 45tgtgcagcaa acagagatga caagatcatc ttt
334639DNAHomo sapiens 46tgtgccagcg gaatacaggg tacctacgag cagtacttc
394736DNAHomo sapiens 47tgtgctctaa ggcagggagc ccagaagctg gtattt
364851DNAHomo sapiens 48tgcagcgttg ataggtcttc tagcgggtcg aacaccgggg
agctgttttt t 514930DNAHomo sapiens 49tgtgccgcta acaatgccag
actcatgttt 305042DNAHomo sapiens 50tgtgccagca gcctcggggg gtcggtagag
acccagtact tc 425139DNAHomo sapiens 51tgtgctgtgg gtacaacagg
aacctacaaa tacatcttt 395239DNAHomo sapiens 52tgtgccagca gttctagggg
ggcacagccc cagcatttt 395336DNAHomo sapiens 53tgtgctctaa gtacaggctt
tcagaaactt gtattt 365448DNAHomo sapiens 54tgtgccagca gcccactagg
cgcgagctcc tacaatgagc agttcttc 485539DNAHomo sapiens 55tgtgctgtga
gagccgctgg caccgacaag ctcatcttt 395645DNAHomo sapiens 56tgtgcaatga
gcgcgaggtc tgggggttac cagaaagtta ccttt 455742DNAHomo sapiens
57tgtgccagca gcccgggcag ggaaagcggg gagctgtttt tt 425839DNAHomo
sapiens 58tgtgctctaa gggcttataa caccgacaag ctcatcttt 395945DNAHomo
sapiens 59tgtgccagca gcttagcgac cgggacagca tacgagcagt acttc
456014PRTHomo sapiens 60Cys Ala Val Gly Ser Ser Ser Asn Thr Gly Lys
Leu Ile Phe1 5 106117PRTHomo sapiens 61Cys Ala Ser Ser Gln Asp Ala
Ile Gly Thr Gly Val Leu Lys Leu Phe1 5 10 15Phe6212PRTHomo sapiens
62Cys Ala Ala Ser Phe Gly Asn Glu Lys Leu Thr Phe1 5 106315PRTHomo
sapiens 63Cys Ala Ser Ser Ala Ser Asp Arg Glu Thr Tyr Gly Tyr Thr
Phe1 5 10 156414PRTHomo sapiens 64Cys Ala Glu Ala Gly Gly Thr Ser
Tyr Gly Lys Leu Thr Phe1 5 106514PRTHomo sapiens 65Cys Ala Ser Cys
Arg Thr Ser Ser Tyr Asn Glu Gln Phe Phe1 5 106613PRTHomo sapiens
66Cys Ala Ser Ser Phe Thr Ser Thr Asp Thr Gln Tyr Phe1 5
106713PRTHomo sapiens 67Cys Ala Ala Pro Ser Gly Asn Thr Gly Lys Leu
Ile Phe1 5 106814PRTHomo sapiens 68Cys Ala Ser Thr Ile Ala Gly Gly
Glu Asp Thr Gln Tyr Phe1 5 106910PRTHomo sapiens 69Cys Ala Val Thr
Gly Asn Gln Phe Tyr Phe1 5 107014PRTHomo sapiens 70Cys Ala Ala Ser
Val Gly Ser Asp Gly Gln Lys Leu Leu Phe1 5 107115PRTHomo sapiens
71Cys Ala Ser Ser Phe Phe Arg Arg Asp Gly Glu Thr Gln Tyr Phe1 5 10
157218PRTHomo sapiens 72Cys Val Val Ser Asp Asn Arg Ala Gly Gly Thr
Ser Tyr Gly Lys Leu1 5 10 15Thr Phe7314PRTHomo sapiens 73Cys Ala
Glu Asn Ser Gly Gly Ser Asn Tyr Lys Leu Thr Phe1 5 107414PRTHomo
sapiens 74Cys Ala Ser Thr Ala Gly Ala Ser Asp Gln Pro Gln His Phe1
5 107513PRTHomo sapiens 75Cys Ala Ser Arg Asn Asn Ala Gly Asn Met
Leu Thr Phe1 5 107616PRTHomo sapiens 76Cys Ala Ser Ser Tyr Ser Asn
Arg Gly Tyr Asn Ser Pro Leu His Phe1 5 10 157712PRTHomo sapiens
77Cys Ala Thr Asp Glu Gly Asn Glu Lys Leu Thr Phe1 5 107814PRTHomo
sapiens 78Cys Ala Ser Ser Leu Val Gly Val Ser Tyr Glu Gln Tyr Phe1
5 107913PRTHomo sapiens 79Cys Ala Glu Ala Val Tyr Gly Asn Asn Arg
Leu Ala Phe1 5 108014PRTHomo sapiens 80Cys Ala Ser Asp Arg Thr Gly
Ser Asp Thr Glu Ala Phe Phe1 5 108112PRTHomo sapiens 81Cys Ala Val
Asp Asn Tyr Gly Gln Asn Phe Val Phe1 5 108215PRTHomo sapiens 82Cys
Ile Val Lys Gly Gly Gly Tyr Asn Phe Asn Lys Phe Tyr Phe1 5 10
158314PRTHomo sapiens 83Cys Ala Ser Ser Met Gly Val Gly Asp Thr Glu
Ala Phe Phe1 5 108413PRTHomo sapiens 84Cys Ala Thr Asp Gly Gly Gly
Ala Gln Lys Leu Val Phe1 5 108514PRTHomo sapiens 85Cys Ala Ser Thr
Gly Gln Gly Ile Gly Asn Thr Ile Tyr Phe1 5 108614PRTHomo sapiens
86Cys Ala Leu Tyr Ser Gly Gly Ser Asn Tyr Lys Leu Thr Phe1 5
108712PRTHomo sapiens 87Cys Ala Met Ser Leu Asn Asp Tyr Lys Leu Ser
Phe1 5 108812PRTHomo sapiens 88Cys Ser Ala Asn Asp Gly Thr Asp Thr
Gln Tyr Phe1 5 108912PRTHomo sapiens 89Cys Ala Ala Pro Gly Gly Asp
Asp Lys Ile Ile Phe1 5 109014PRTHomo sapiens 90Cys Ala Leu Ala Gln
Thr Asp Ser Trp Gly Lys Phe Gln Phe1 5 109114PRTHomo sapiens 91Cys
Ala Ser Ser Leu Pro Gly Gln Ala Tyr Glu Gln Tyr Phe1 5
109217PRTHomo sapiens 92Cys Ala Val Ser Asp Gln Lys Leu Thr Gly Gly
Gly Asn Lys Leu Thr1 5 10 15Phe9317PRTHomo sapiens 93Cys Ser Ala
Arg Asp Pro Asn Arg Gly Trp Asn Thr Gly Glu Leu Phe1 5 10
15Phe9412PRTHomo sapiens 94Cys Ala Thr Asp Ser Gly Thr Asp Lys Leu
Ile Phe1 5 109516PRTHomo sapiens 95Cys Ala Ser Ser Leu Asp Arg Val
Gly Thr Gly Thr Glu Gln Tyr Phe1 5 10 159615PRTHomo sapiens 96Cys
Ala Glu Ser Lys Arg Lys Thr Ser Tyr Asp Lys Val Ile Phe1 5 10
159714PRTHomo sapiens 97Cys Ala Ser Ser Pro Ser Thr Gly Gln Glu Thr
Gln Tyr Phe1 5 109816PRTHomo sapiens 98Cys Ala Val Gln Ala Val Ser
Gly Gly Ser Asn Tyr Lys Leu Thr Phe1 5 10 159913PRTHomo sapiens
99Cys Ala Trp Ser Pro Gln Thr Thr Gly Leu Glu Tyr Phe1 5
1010015PRTHomo sapiens 100Cys Ala Val Ser Asp Gly Asp Asn Tyr Gly
Gln Asn Phe Val Phe1 5 10 1510111PRTHomo sapiens 101Cys Ala Ser Ser
Leu Gln Ile Ala Gln Tyr Phe1 5 1010214PRTHomo sapiens 102Cys Ala
Gly Leu Glu Gly Pro Asn Asp Tyr Lys Leu Ser Phe1 5 1010314PRTHomo
sapiens 103Cys Ala Thr Ala Ser Gln Gly Pro Lys Glu Thr Gln Tyr Phe1
5 1010411PRTHomo sapiens 104Cys Ala Ala Asn Arg Asp Asp Lys Ile Ile
Phe1 5 1010513PRTHomo sapiens 105Cys Ala Ser Gly Ile Gln Gly Thr
Tyr Glu Gln Tyr Phe1 5 1010612PRTHomo sapiens 106Cys Ala Leu Arg
Gln Gly Ala Gln Lys Leu Val Phe1 5 1010717PRTHomo sapiens 107Cys
Ser Val Asp Arg Ser Ser Ser Gly Ser Asn Thr Gly Glu Leu Phe1 5 10
15Phe10810PRTHomo sapiens 108Cys Ala Ala Asn Asn Ala Arg Leu Met
Phe1 5 1010914PRTHomo sapiens 109Cys Ala Ser Ser Leu Gly Gly Ser
Val Glu Thr Gln Tyr Phe1 5 1011013PRTHomo sapiens 110Cys Ala Val
Gly Thr Thr Gly Thr Tyr Lys Tyr Ile Phe1 5 1011113PRTHomo sapiens
111Cys Ala Ser Ser Ser Arg Gly Ala Gln Pro Gln His Phe1 5
1011212PRTHomo sapiens 112Cys Ala Leu Ser Thr Gly Phe Gln Lys Leu
Val Phe1 5 1011316PRTHomo sapiens 113Cys Ala Ser Ser Pro Leu Gly
Ala Ser Ser Tyr Asn Glu Gln Phe Phe1 5 10 1511413PRTHomo sapiens
114Cys Ala Val Arg Ala Ala Gly Thr Asp Lys Leu Ile Phe1 5
1011515PRTHomo sapiens 115Cys Ala Met Ser Ala Arg Ser Gly Gly Tyr
Gln Lys Val Thr Phe1 5 10 1511614PRTHomo sapiens 116Cys Ala Ser Ser
Pro Gly Arg Glu Ser Gly Glu Leu Phe Phe1 5 1011713PRTHomo sapiens
117Cys Ala Leu Arg Ala Tyr Asn Thr Asp Lys Leu Ile Phe1 5
1011815PRTHomo sapiens 118Cys Ala Ser Ser Leu Ala Thr Gly Thr Ala
Tyr Glu Gln Tyr Phe1 5 10 1511928DNAArtificial SequenceDescription
of Artificial Sequence Synthetic reverse primer for the
amplification of TCR alpha-chain 119cacaggctgt cttacaatct tgcagatc
2812023DNAArtificial SequenceDescription of Artificial Sequence
Synthetic reverse primer for the amplification of TCR beta-chain
120ctccacttcc agggctgcct tca 2312124DNAArtificial
SequenceDescription of Artificial Sequence Synthetic reverse primer
for the amplification of TCR beta-chain 121tgacctggga tggttttgga
gcta 2412217DNAArtificial SequenceDescription of Artificial
Sequence Synthetic primer for determination of a CDR3 nucleotide
sequence 122caggaaacag ctatgac 1712316PRTHomo sapiens 123Lys Arg
Tyr Phe Lys Leu Ser His Leu Gln Met His Ser Arg Lys His1 5 10
1512420PRTHomo sapiens 124Phe Arg Lys Gln Asn Pro Asp Ile Val Ile
Tyr Gln Tyr Met Asp Asp1 5 10 15Leu Tyr Val Gly 201259PRTHomo
sapiens 125Cys Tyr Thr Trp Asn Gln Met Asn Leu1 5
References
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018
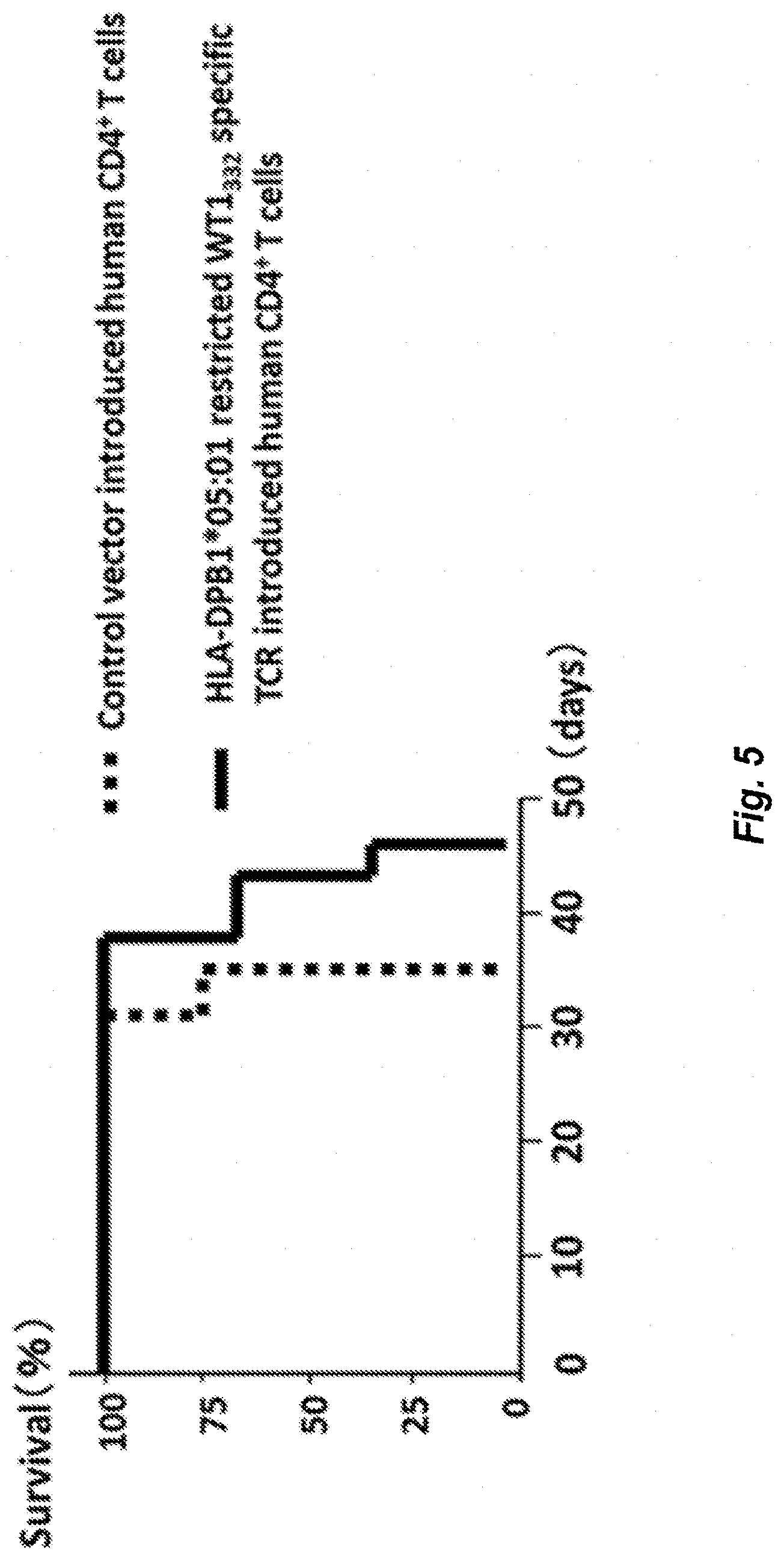
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.