Method For Improving Engine Performance With Renewable Lubricant Compositions
VAN DAM; Willem ; et al.
U.S. patent application number 16/993438 was filed with the patent office on 2021-02-18 for method for improving engine performance with renewable lubricant compositions. The applicant listed for this patent is Chevron U.S.A. Inc.. Invention is credited to David S. LEE, Mihir K. PATEL, Willem VAN DAM.
| Application Number | 20210047577 16/993438 |
| Document ID | / |
| Family ID | 1000005060737 |
| Filed Date | 2021-02-18 |

| United States Patent Application | 20210047577 |
| Kind Code | A1 |
| VAN DAM; Willem ; et al. | February 18, 2021 |
METHOD FOR IMPROVING ENGINE PERFORMANCE WITH RENEWABLE LUBRICANT COMPOSITIONS
Abstract
Provided herein are lubricant compositions comprising renewable base oils as embodied by hydrocarbon mixtures with controlled structure characteristics in combination with lubricant additives that address performance requirements and stricter environmental and fuel economy regulations. The lubricant composition provides performance in the cold crank simulated viscosity (CCS) vs Noack volatility relationship, which allows for the formulation of lower viscosity engine oils with improved fuel economy, improved fuel economy retention, and retained LSPI prevention additionally conferring improved characteristics to other devices or apparatus requiring lubrication.
| Inventors: | VAN DAM; Willem; (Novato, CA) ; PATEL; Mihir K.; (San Ramon, CA) ; LEE; David S.; (San Ramon, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005060737 | ||||||||||
| Appl. No.: | 16/993438 | ||||||||||
| Filed: | August 14, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62886407 | Aug 14, 2019 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10M 2203/022 20130101; C10N 2030/02 20130101; C10N 2020/071 20200501; C10N 2040/25 20130101; C10N 2030/54 20200501; C10M 105/04 20130101 |
| International Class: | C10M 105/04 20060101 C10M105/04 |
Claims
1. A method of improving fuel economy comprising lubricating an internal combustion engine with a lubricant that comprises: a. (a) a mixture of base oil having at least 25 wt. % renewable base oil comprising a hydrocarbon mixture in which: i. the percentage of molecules with even carbon number is .gtoreq.80% according to FIMS; ii. the BP/BI.gtoreq.-0.6037 (Internal alkyl branching per molecule)+2.0; iii. on average there are 0.3 to 1.5 5+ methyl per molecule; b. at least one of the following additives i. dispersants ii. detergents iii. Inhibitors iv. Friction Modifiers v. Pour Point Depressants vi. Viscosity Modifiers
2. The method of claim 1, wherein the lubricating oil composition comprises from about 5 wt. % to about 30 wt. % total additive concentration and from about 70 to about 95 wt. % of the base oil mixture.
3. The method of claim 1, wherein the composition has a lubricating high temperature high shear viscosity less than 2.3 cP, based on ASTM D5481.
4. The method of claim 1, wherein the composition has a lubricating kinematic viscosity at 100.degree. C. less than or equal to 7.1 cSt, based on ASTM D445.
5. The method of claim 1, wherein the composition has a lubricating low temperature cold cranking viscosity at -35.degree. C. less than or equal to 6200 mPas, based on ASTM D5293.
6. The method of claim 1, wherein the lubricating composition has a lubricating SAE viscosity grade less than or equal to 0W-12, based on SAE J300.
7. The method of claim 1 wherein the fuel economy is improved by at least 0.3% and preferably more than 0.5%.
8. A method of improving fuel economy retention comprising lubricating an internal combustion engine with a lubricant that comprises: a. a mixture of base oil having at least 25 wt. % renewable base oil comprising a hydrocarbon mixture in which: i. the percentage of molecules with even carbon number is .gtoreq.80% according to FIMS; ii. the BP/BI.gtoreq.-0.6037 (Internal alkyl branching per molecule)+2.0; iii. on average there are 0.3 to 1.5 5+ methyl per molecule; b. at least one of the following additives i. dispersants ii. detergents iii. Inhibitors iv. Friction Modifiers v. Pour Point Depressants vi. Viscosity Modifiers wherein the lubricating oil composition comprises from about 5 wt. % to about 30 wt. % total additive concentration and from about 70 to about 95 wt. % of the base oil mixture.
9. The method of claim 7, wherein the composition has a lubricating high temperature high shear viscosity less than 2.3 cP, based on ASTM D5481.
10. The method of claim 7, wherein the composition has a lubricating kinematic viscosity at 100.degree. C. less than or equal to 7.1 cSt, based on ASTM D445.
11. The method of claim 7, wherein the composition has a lubricating low temperature cold cranking viscosity at -35.degree. C. less than or equal to 6200 mPas, based on ASTM D5293.
12. The method of claim 7, wherein the composition has a lubricating oil SAE viscosity grade less than or equal to W-12, based on SAE J300.
13. The method of claim 7, wherein typical drain interval fuel economy loss is less than 0.5%, preferably less than 0%.
14. The method of claim 7, wherein extended drain interval fuel economy loss is less than 0.4%, preferably less than 0%.
15. The method of claim 7, wherein the average fuel economy loss is less than 0.5%, preferably less than 0% throughout the life of a lubricant.
16. The method of claim 7, wherein there is an improvement in fuel economy retention and a retention in low speed pre ignition prevention in an internal combustion engine operated over an extended period of exposure of the lubricant to elevated temperature conditions.
Description
FIELD OF THE INVENTION
[0001] Methods for improving engine performance with lubricant compositions containing a renewable base oil comprising hydrocarbon mixtures and a lubricant additive have been developed which possess unique compositional characteristics and which demonstrate superior fuel economy improvement capability, fuel economy retention properties over the life of the lubricant when used to lubricate various types of internal combustion engines.
BACKGROUND OF THE INVENTION
[0002] Industry trends in engine oil formulation are moving to lower viscosity regimes (0W-X) to enhance fuel economy benefits, while also expecting to maintain current or better levels of performance. However, as the industry moves to lower viscosity, maintaining low oil consumption, and fuel economy durability becomes challenging or even unattainable with available technology. As the viscosity of the oil is reduced, the volatility increases, leading to increased evaporation of the engine oil and increased oil viscosity. Additionally, fuel economy can be enhanced by lowering high temperature high shear (HTHS) viscosity of the oil. HTHS is largely affected by viscosity modifiers which also regulate cold and high temperature viscosity, thus a high viscosity index (VI) oil is desirable. Some ultra low viscosity oils are on the market, however they are unable to meet the most stringent volatility requirements.
[0003] Base stocks are commonly used to produce various lubricants, including lubricating oils for internal combustion engines, turbines, compressors, hydraulic systems, etc. They are also used as process oils, white oils, and heat transfer fluids. Finished lubricants generally consist of two components, base oils and additives. Base oil, which could be one or a mixture of base stocks, is the major constituent in these finished lubricants and contributes significantly to their performances, such as viscosity and viscosity index, volatility, stability, and low temperature performance. In general, a few base stocks are used to manufacture a wide variety of finished lubricants by varying the mixtures of individual base stocks and individual additives.
[0004] A method of improving engine fuel efficiency with a lubricant composition containing fatty acid esters is set forth in U.S. Pat. No. 9,885,004.
[0005] The American Petroleum Institute (API) categorizes base stocks in five groups based on their saturated hydrocarbon content, sulfur level, and viscosity index (Table 1 below). Group I, II, and III base stocks are mostly derived from crude oil via extensive processing, such as solvent refining for Group I, and hydroprocessing for Group II and Group III. Certain Group III base stocks can also be produced from synthetic hydrocarbon liquids via a Gas-to-Liquids process (GTL), and are obtained from natural gas, coal or other fossil resources. Group IV base stocks, the polyalphaolefins (PAO), are produced by oligomerization of alpha olefins, such as 1-decene. Group V base stocks include everything that does not belong to Groups I-IV, such as naphthenic base stocks, polyalkylene glycols (PAG), and esters. Most of the feedstocks for large-scale base stock manufacturing are non-renewable.
TABLE-US-00001 TABLE 1 API Base Oil Classification (API 1509 Appendix E) Viscosity Saturates Index by API (ASTM ASTM Sulphur, Group D2270) D2007 % Description I 80-120 <90% >.03% Conventional (solvent refining) II 80-120 .gtoreq.90% .ltoreq..03% Hydroprocessing III .gtoreq.120 .gtoreq.90% .ltoreq..03% Severe Hydroprocessing IV PolyAlphaOlefins (PAO) V All other base stocks not included above e.g. esters
[0006] Automotive engine oils are by far the largest market for base stocks. The automotive industry has been placing more stringent performance specifications on engine oils due to requirements for lower emissions, long drain intervals, and better fuel economy. Specifically, automotive OEMs (original equipment manufacturer) have been pushing for the adoption of lower viscosity engine oils such as 0W-20 down to W-8 and even 0W-4, to lower friction losses and achieve fuel economy improvement. Base Oils with a lower Noack Volatility in an engine oil allows the formulation to retain the designed viscosity for longer operation time allowing for increased fuel economy retention and longer drain intervals discussed in U.S. Pat. No. 6,300,291. Group I and Group II's usage in engine oils with viscosity grade below 0W-20 engine oils is highly limited because formulations blended with them cannot meet the performance specifications for lower than 0W-20 engine oils, leading to increased demands for Group III and Group IV base stocks.
[0007] Group III base stocks are mostly manufactured from vacuum gas oils (VGOs) through hydrocracking and catalytic dewaxing (e.g. hydroisomerization). Group III base stocks can also be manufactured by catalytic dewaxing of slack waxes originating from solvent refining, or by catalytic dewaxing of waxes originating from Fischer-Tropsch synthesis from natural gas or coal based raw materials also known as Gas to Liquids base oils (GTL).
[0008] Manufacturing processes of Group III base stocks from VGOs is discussed in U.S. Pat. Nos. 5,993,644 and 6,974,535. Their boiling point distributions are typically higher when compared to PAOs of the same viscosity, causing them to have higher volatility than PAOs. Additionally, Group III base stocks typically have higher cold crank viscosity (i.e., dynamic viscosity according to ASTM D5293, CCS) than Group IV base stocks at equivalent viscosities.
[0009] GTL base stock processing is described in U.S. Pat. Nos. 6,420,618 and 7,282,134, as well as U.S. Patent Application Publication 2008/0156697. For example, the latter publication describes a process for preparing base stocks from a Fischer-Tropsch synthesis product, the fractions of which with proper boiling ranges are subjected to hydroisomerization to produce GTL base stocks.
[0010] Such structures and properties of GTL base stocks are described, for example, in U.S. Pat. Nos. 6,090,989 and 7,083,713, as well as U.S. Patent Application Publication 2005/0077208. In U.S. Patent Application Publication 2005/0077208, lubricant base stocks with optimized branching are described, which have alkyl branches concentrated toward the center of the molecules to improve the base stocks' cold flow properties. Nevertheless, pour points for GTL base stocks are typically worse than PAO or other synthetic hydrocarbon base stocks.
[0011] A further concern with GTL base stocks is the severely limited commercial supply, a result of the prohibitively large capital requirements for a new GTL manufacturing facility. Access to low cost natural gas is also required to profitably produce GTL base stocks. Furthermore, as GTL base stocks are typically distilled from an isomerized oil with a wide boiling point distribution, the process results in a relatively low yield to the base stock with a desired viscosity when compared to that of a typical PAO process. Due to these monetary and yield constraints there is currently only a single manufacturing plant of group III+GTL base stocks, exposing formulations that use GTL to supply chain and price fluctuation risks.
[0012] Polyalphaolefins (PAOs), or Group IV base oils, are produced by the polymerization of alphaolefins in the presence of a Friedel Crafts catalyst such as AlCl3, BF3, or BF3 complexes. For example, 1-octene, 1-decene, and 1-dodecene have been used to manufacture PAOs that have a wide range of viscosities, varying from low molecular weight and low viscosity of about 2 cSt at 100.degree. C., to high molecular weight, viscous materials with viscosities exceeding 100 cSt at 100.degree. C. The polymerization reaction is typically conducted in the absence of hydrogen; the lubricant range products are thereafter polished or hydrogenated to reduce the residual unsaturation. Processes to produce PAO based lubricants are disclosed, for example, in U.S. Pat. Nos. 3,382,291; 4,172,855; 3,742,082; 3,780,128; 3,149178; 4,956,122; 5,082,986; 7,456,329; 7,544,850; and U.S. Patent Application Publication 2014/0323665. Prior efforts to prepare various PAOs that can meet the increasingly stringent performance requirements of modern lubricants and automotive engine oil particularly have favored low viscosity polyalphaolefin base stocks derived from 1-decene, alone or in some blend with other mineral oils. However, the polyalphaolefins derived from 1-decene can be prohibitively expensive due to its limited supply. Attempts to overcome the availability constraint of 1-decene have led to the production of PAOs from C8 through C12 mixed alpha-olefin feeds, lowering the amount of 1-decene that is needed to impart the properties. However, they still do not completely remove the requirement for providing 1-decene as the predominate olefin feedstock due to performance concerns.
[0013] Similarly, previous efforts to use linear alphaolefins in the C14-C20 range made polyalphaolefins with unacceptably high pour points, which are unsuitable for use in a variety of lubricants, including 0W engine oils.
[0014] Therefore, there remains a need for a lubricant composition having properties within commercially acceptable ranges, for example, for use in automotive and other applications, with such properties including one or more of viscosity, Noack volatility, and low temperature cold-cranking viscosity. Furthermore, there remains a need for lubricant compositions having improved properties and methods of manufacture thereof, where the base stock compositions have reduced amounts of 1-decene incorporated therein, and may even preferably eliminate the use of 1-decene in the manufacture thereof.
[0015] In addition to the technical demands for the automotive industry, environmental awareness and regulations are driving manufacturers to use renewable feedstocks and raw materials in the production of base stocks and lubricants. It is known that esters and some Group III hydrocarbon base stocks (U.S. Pat. No. 9,862,906B2) of renewable and biological origin have been used in applications such as refrigeration compressor lubricants, hydraulic oils and metal working fluids, and more recently in automotive and industrial lubricants (US20170240832A1). Common biological sources for hydrocarbons are natural oils, which can be derived from plant sources such as canola oil, castor oil, sunflower seed oil, rapeseed oil, peanut oil, soy bean oil, and tall oil, or palm oil. Other commercial sources of hydrocarbons include engineered microorganisms such as Algae or Yeast.
[0016] Due to increasing demand for high performing lubricant base stocks there is a continuing need for improved hydrocarbon mixtures. The industry requires these hydrocarbon mixtures to have superior Noack Volatility, and low temperature viscometric properties that can meet stricter engine oil requirements, preferably from renewable sources.
[0017] The Automotive industry is trending towards downsizing gasoline engines coupled with turbocharger and gasoline direct injection to meet current and future CO2 regulations. To maximize fuel economy, these engines are commonly calibrated to operate at low speeds and high engine loads. Many of these downsized, turbocharged engines have experienced uncontrolled combustion events when engines operate under low speed-high load conditions, commonly known as Low Speed Pre-ignition (LSPI). LSPI is random in nature and gives a sudden rise in the pressure inside the cylinder which can result in the catastrophic engine failure.
[0018] Many studies have evaluated the impact of the calcium treat rate and concluded that increasing calcium content increases LSPI occurrences. [12-14] Some researchers have proposed alternative lubricant formulating strategies to reduce the occurrence of LSPI. [12-14] In general, these lubricants were formulated by reducing the amount of LSPI promoter (calcium containing detergent) and increasing the amount of LSPI inhibitors (ZDDP, MoDTC, Mg containing detergent etc.). However, reducing calcium content beyond a certain limit can increase piston deposit, increase varnish, increase acid concentration and reduction in the oil drain interval. Hence, many studies have restricted the amount of calcium containing detergent to 0.1 wt. %.
[0019] Therefore a lubricant composition which can improve fuel economy retention while reducing LSPI is desired.
SUMMARY OF THE INVENTION
[0020] The present invention relates to a method of improving engine performance by supplying an internal combustion engine with a lubricant composition, containing a saturated hydrocarbon mixture and lubricant additive with well-controlled structural characteristics that address the performance requirements driven by the stricter environmental and fuel economy regulations for automotive engine oils. The branching characteristics of the hydrocarbon molecules of the base oil portion are controlled to consistently provide a composition that has a surprising CCS viscosity at -35.degree. C. (ASTM D5329) and Noack volatility (ASTM D5800) relationship.
[0021] An embodiment of the invention is a method where supplying the lubricant composition to an internal combustion engine and operation of the engine at elevated operating temperatures results in a Fuel Economy Improvement (FEI) that is at least 0.3% better than for a conventional lubricant of equal viscosity. In a preferred embodiment the FEI is at least 0.5% better than for a conventional lubricant of equal viscosity.
[0022] A further embodiment of the invention is a method where supplying the lubricant composition to an internal combustion engine and operation of the engine over an extended period of time results in a Fuel Economy Retention (FER) benefit that is at least 0.5% better than for a conventional lubricant of equal viscosity. In a preferred embodiment the FER is at least 1.0% better than for a conventional lubricant of equal viscosity.
[0023] A further embodiment is a method where supplying the lubricant composition to an internal combustion engine and operation of the engine at elevated operating temperature over an extended period of time results in an LSPI Prevention Retention benefit by limiting the additive metal concentration increase. In a preferred embodiment the Calcium concentration increase is limited to no more than 15%.
[0024] An important aspect of the present invention relates to a lubricant composition possessing a renewable base oil with a saturated hydrocarbon mixture having greater than 80% of the molecules with an even carbon number according to FIMS, with the mixture exhibiting a branching characteristic of BP/BI.gtoreq.-0.6037 (Internal alkyl branching per molecule)+2.0, and when the hydrocarbon mixture is analyzed by carbon NMR as a whole, has on average at least 0.3 to 1.5 5+ methyl branches per molecule.
[0025] One way to synthesize the hydrocarbon mixture disclosed herein is through oligomerization of C14-C20 alpha or internal-olefins, followed by hydroisomerization of the oligomers. Using C14-C20 olefins would ease the demand for high-price 1-decene and other crude oil or synthetic gas based olefins as feedstocks, and making available alternate sources of olefin feedstocks such as those derived from C14-C20 alcohols. The hydrocarbon compositions are derived from one or more olefin co-monomers, where said olefin comonomers are oligomerized to dimers, trimers, and higher oligomers. The oligomers are then subjected to hydroisomerization. The resulting hydrocarbon mixtures have excellent pour point, volatility and viscosity characteristics and additive solubility properties.
BRIEF DESCRIPTION OF THE DRAWINGS
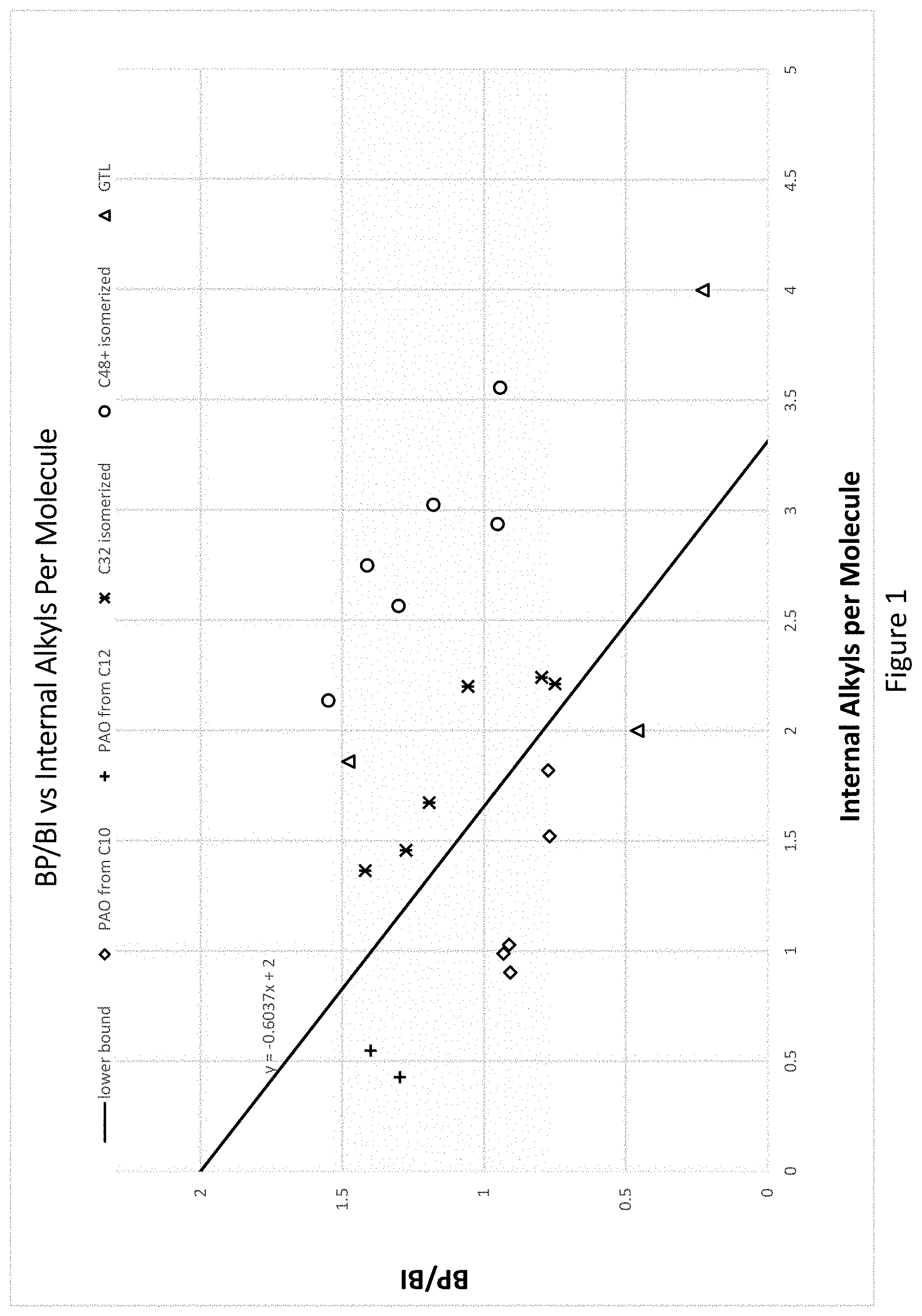
[0026] FIG. 1 illustrates the relationship between BP/BI and Internal Alkyl Branches per Molecule for various hydrocarbons, including low-viscosity PAO manufactured from 1-decene and 1-dodecene, GTL base oils, and hydroisomerized hexadecene oligomers. The straight line in the plot depicts the equation of BP/BI=-0.6037 (Internal alkyl branching per molecule)+2.0.
[0027] FIG. 2 illustrates the relationship between BP/BI and 5+ Methyl Branches per Molecule for various hydrocarbons, including low-viscosity PAO manufactured from 1-decene and 1-dodecene, GTL base oils, and hydroisomerized hexadecene oligomers. It demonstrates that the 5+ Methyl Branches per Molecules for the hydrocarbon mixtures disclosed in this patent fall in a unique range of 0.3-1.5.
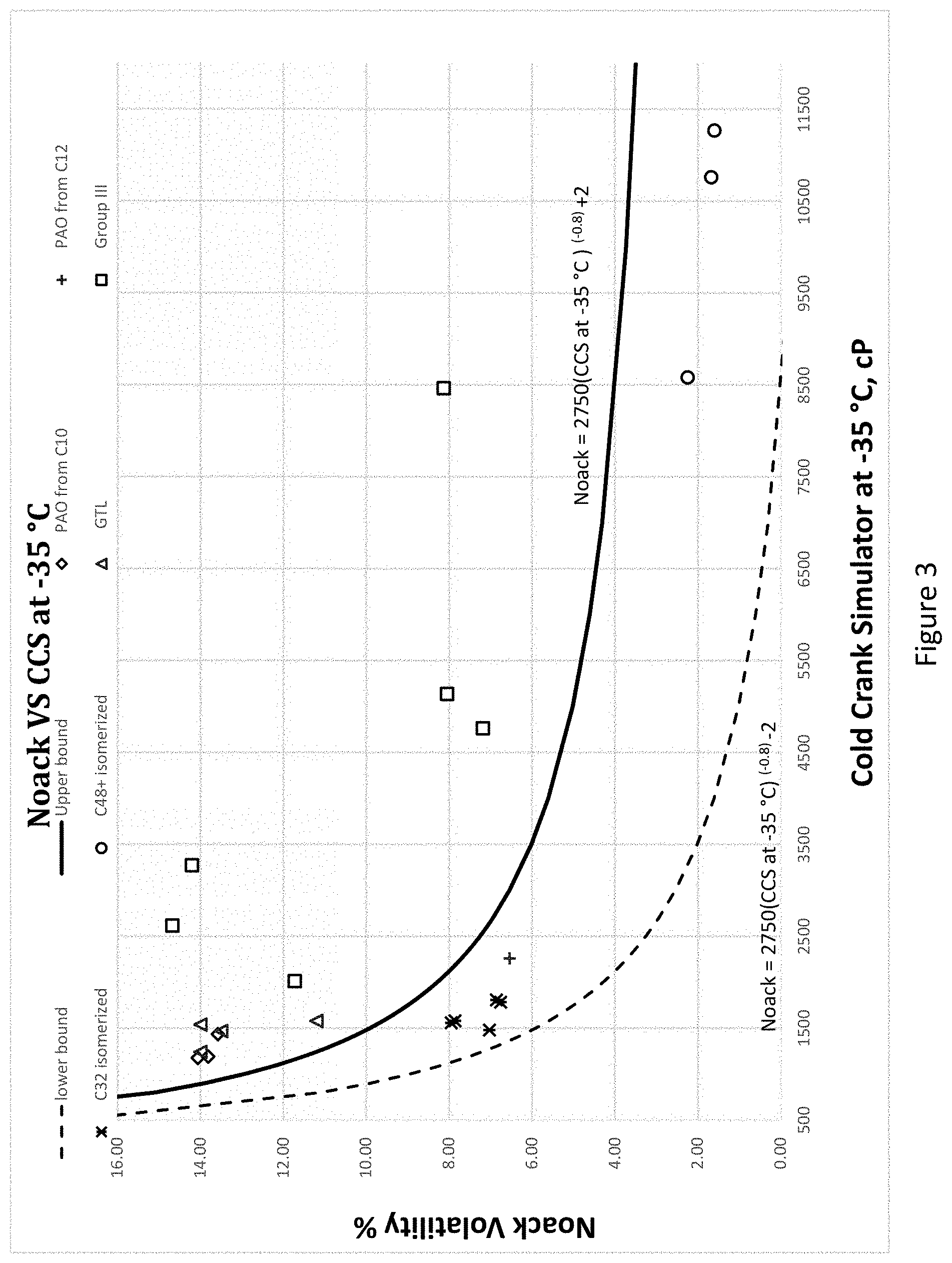
[0028] FIG. 3 illustrates the relationship between NOACK volatility and CCS at -35.degree. C. for various hydrocarbons, including low-viscosity PAO manufactured from 1-decene and 1-dodecene, GTL base oils, Group III base oils, and hydroisomerized hexadecene oligomers. The solid line and dotted line depicts the upper limit and lower limit of the Noack vs. CCS at -35.degree. C. exhibited by the present unique hydrocarbon mixture, which are NOACK=2,750 (CCS at -35.degree. C.)(-0.8)+2 and NOACK=2,750 (CCS at -35.degree. C.)(-0.8)-2, respectively.
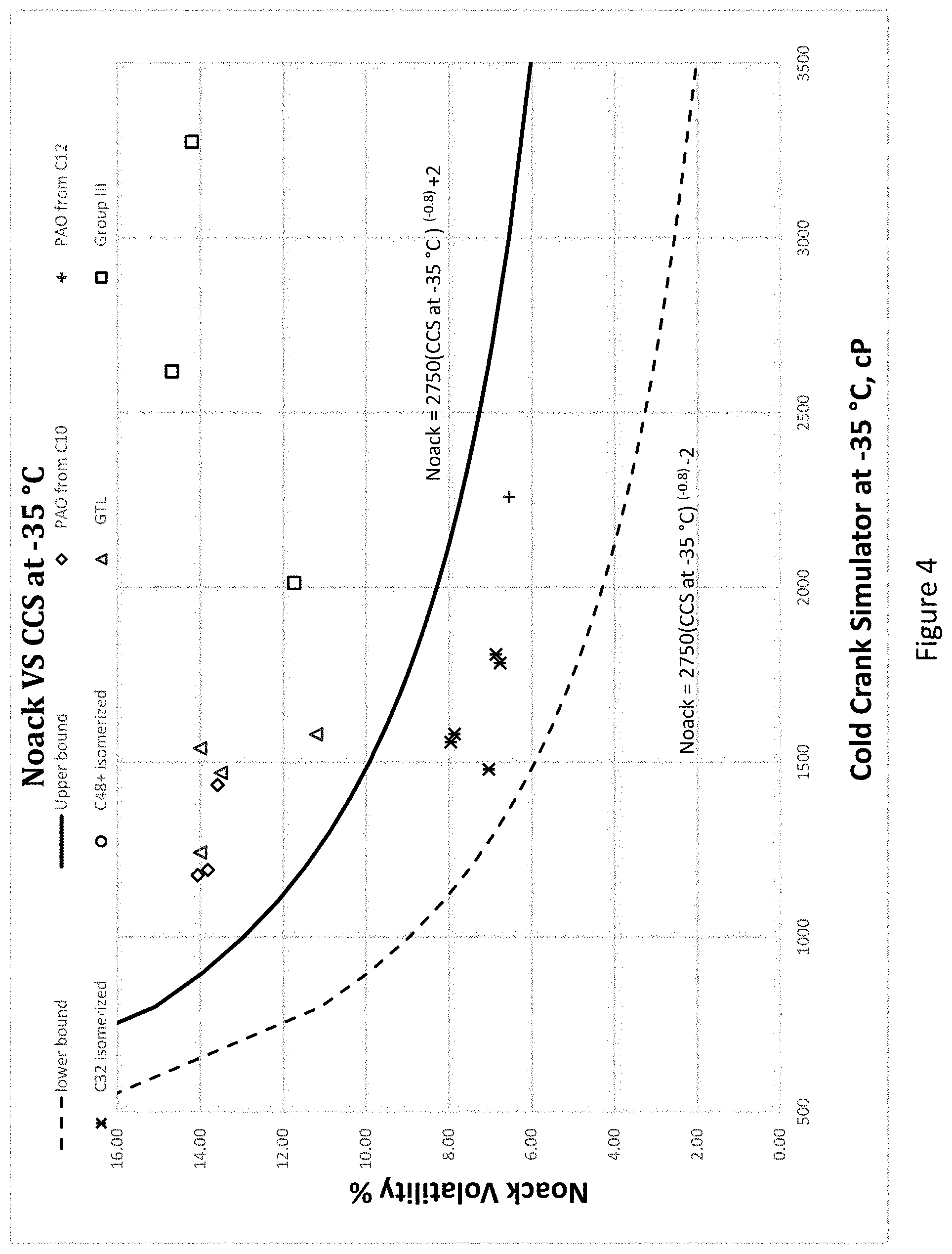
[0029] FIG. 4 is an enlarged view of FIG. 3 in the range of 800-2,800 cP of CCS at -35.degree. C.
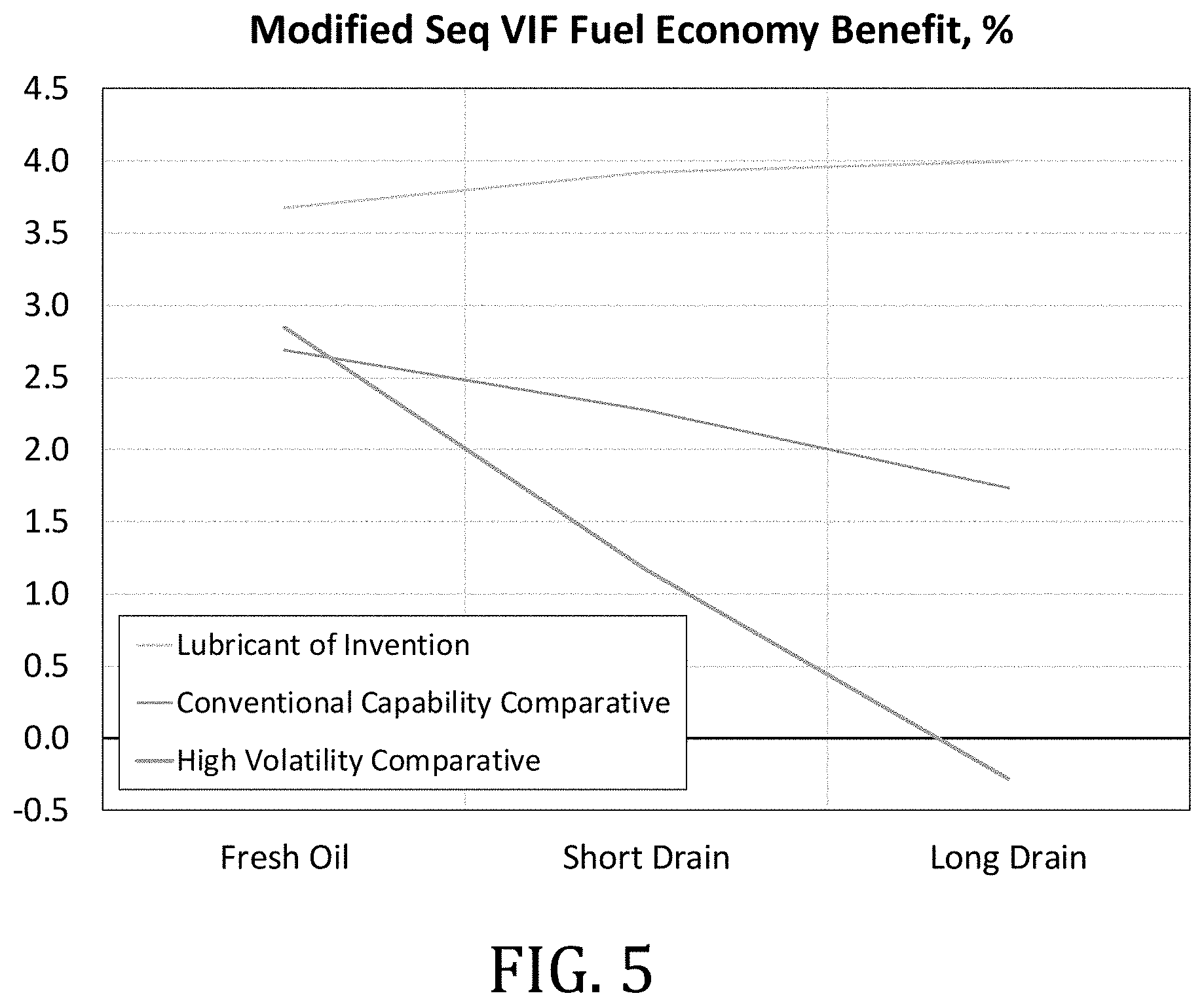
[0030] FIG. 5 is a graph of the fuel economy benefit as measured in a modified sequence VIF test using three different lubricant compositions.
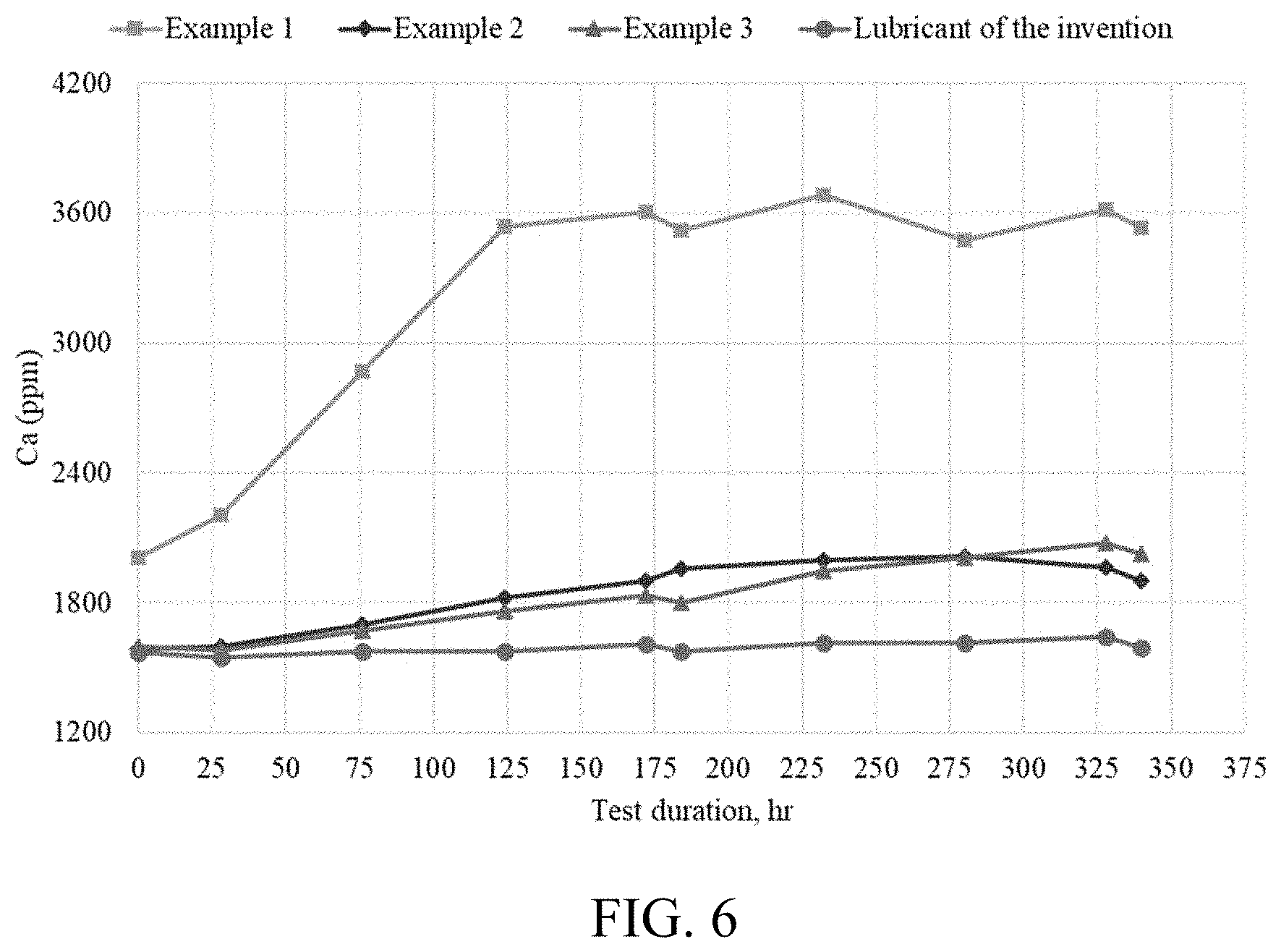
[0031] FIG. 6 is a graph of the calcium content of three different oils as measured in an extended duration test.
DETAILED DESCRIPTION OF THE INVENTION
[0032] All numerical values within the detailed description and the claims herein are modified by "about" or "approximately" the indicated value, and take into account experimental error and variations that would be expected by a person having ordinary skill in the art.
[0033] Disclosed herein is a method for improving engine performance; specifically supplying lubricant compositions imparting improved fuel economy, improved fuel economy retention, and improved LSPI Prevention Retention wherein the lubricant compositions contain a renewable base oil stock containing a saturated hydrocarbon mixture having a unique branching structure as characterized by NMR that makes it suitable to be used as a high-quality synthetic base stock and a lubricant additive. The hydrocarbon mixture has outstanding properties including extremely low volatility, good low-temperature properties, etc., which are important performance attributes of high-quality base stocks. To be specific, the mixture comprises greater than 80% of the molecules with an even carbon number according to FIMS. The branching characteristics of the hydrocarbon mixture by NMR comprises a BP/BI in the range .gtoreq.-0.6037 (Internal alkyl branching per molecule)+2.0. Moreover, on average, at least 0.3 to 1.5 of the internal methyl branches are located more than four carbons away from the end carbon. A saturated hydrocarbon with this unique branching structure exhibits a surprising cold crank simulated viscosity (CCS) vs. Noack volatility relationship that is beneficial for blending low-viscosity automotive engine oils.
[0034] In one embodiment, the hydrocarbon mixtures described herein are the product of oligomerization of olefins and a subsequent hydroisomerization. C14 to C20 olefins are oligomerized to form an oligomer distribution consisting of unreacted monomer, dimers (C28-C40), and trimers and higher oligomers (.gtoreq.C42). The unreacted monomers are distilled off for possible re-use in a subsequent oligomerization. The remaining oligomers are then hydroisomerized to achieve the final branching structures described herein which consistently impart a surprising cold crank simulated viscosity (CCS) vs. Noack volatility relationship.
Definitions
[0035] Renewable as used herein means any biologically derived composition, including fatty alcohols, olefins, or oligomers. Such compositions may be made, for nonlimiting example, from biological organisms designed to manufacture specific oils, as discussed in WO 2012/141784, but do not include petroleum distilled or processed oils such as, for nonlimiting example, mineral oils. A suitable method to assess materials derived from renewable resources is through "Standard Test Methods for Determining the Biobased Content of Solid, Liquid, and Gaseous Samples Using Radiocarbon Analysis" (ASTM D6866-12 or ASTM D6866-11). Counts from 14C in a sample can be compared directly or through secondary standards to SRM 4990C. A measurement of 0% 14C relative to the appropriate standard indicates carbon originating entirely from fossils (e.g., petroleum based). A measurement of 100% 14C indicates carbon originating entirely from modern sources (See, e.g., WO 2012/141784, incorporated herein by reference).
[0036] Viscosity is the physical property that measures the fluidity of the base stock. Viscosity is a strong function of temperature. Two commonly used viscosity measurements are dynamic viscosity and kinematic viscosity. Dynamic viscosity measures the fluid's internal resistance to flow. Cold cranking simulator (CCS) viscosity at -35.degree. C. for engine oil is an example of dynamic viscosity measurements. The SI unit of dynamic viscosity is Pas. The traditional unit used is centipoise (cP), which is equal to 0.001 Pas (or 1 m Pas). The industry is slowly moving to SI units. Kinematic viscosity is the ratio of dynamic viscosity to density. The SI unit of kinematic viscosity is mm2/s. The other commonly used units in industry are centistokes (cSt) at 40.degree. C. (KV40) and 100.degree. C. (KV100) and Saybolt Universal Second (SUS) at 100.degree. F. and 210.degree. F. Conveniently, 1 mm2/s equals 1 cSt. ASTM D5293 and D445 are the respective methods for CCS and kinematic viscosity measurements. "Viscosity Grade" as used herein refers to the lubricant composition containing a renewable base oil formulated to meet the definition of an SAE XW-YY lubricant (where X can be 0 or 5, and YY can be 4, 8, 12, 16, or 20). The properties for the various Viscosity Grades are further defined in the SAE J300 Industry Standard
[0037] Viscosity Index (VI) is an empirical number used to measure the change in the base stock's kinematic viscosity as a function of temperature. The higher the VI, the less relative change is in viscosity with temperature. High VI base stocks are desired for most of the lubricant applications, especially in multigrade automotive engine oils and other automotive lubricants subject to large operating temperature variations. ASTM D2270 is a commonly accepted method to determine VI.
[0038] Pour point is the lowest temperature at which movement of the test specimen is observed. It is one of the most important properties for base stocks as most lubricants are designed to operate in the liquid phase. Low pour point is usually desirable, especially in cold weather lubrication. ASTM D97 is the standard manual method to measure pour point. It is being gradually replaced by automatic methods, such as ASTM D5950 and ASTM D6749. ASTM D5950 with 1.degree. C. testing interval is used for pour point measurement for the examples in this patent.
[0039] Volatility is a measurement of oil loss from evaporation at an elevated temperature. It has become a very important specification due to emission and operating life concerns, especially for lighter grade base stocks. Volatility is dependent on the oil's molecular composition, especially at the front end of the boiling point curve. Noack (ASTM D5800) is a commonly accepted method to measure volatility for automotive lubricants. The Noack test method itself simulates evaporative loss in high temperature service, such as an operating internal combustion engine.
[0040] Fuel Economy Improvement: the reduction in fuel consumption for an engine running on the candidate oil relative to that observed with the same engine running at the same test conditions on a reference oil.
[0041] Fuel Economy Retention: the capability of a lubricant to maintain its level of fuel consumption over a period of time where that lubricant is exposed to aging conditions in an internal combustion engine. Fuel Economy Retention can also be expressed as the capability of a lubricant to retain its fuel economy improvement over a period of time where the lubricant is exposed to aging conditions in an internal combustion engine. Fuel Economy Retention can also be expressed as a lack of fuel economy loss over a period of time where the lubricant is exposed to aging conditions in an internal combustion engine.
[0042] Boiling point distribution is the boiling point range that is defined by the True Boiling Points (TBP) at which 5% and 95% materials evaporates. It is measured by ASTM D2887 herein.
[0043] NMR Branching Analysis Branching parameters measured by NMR spectroscopy for the hydrocarbon characterization include:
[0044] Branching Index (BI): the percentage of methyl hydrogens appearing in the chemical shift range of 0.5 to 1.05 ppm among all hydrogens appearing in the 1H NMR chemical range 0.5 to 2.1 ppm in an isoparaffinic hydrocarbon.
[0045] Branching Proximity (BP): the percentage of recurring methylene carbons which are four or more number of carbon atoms removed from an end group or branch appearing at 13C NMR chemical shift 29.8 ppm.
[0046] Internal Alkyl Carbons: is the number of methyl, ethyl, or propyl carbons which are three or more carbons removed from end methyl carbons, that includes 3-methyl, 4-methyl, 5+ methyl, adjacent methyl, internal ethyl, n-propyl and unknown methyl appearing between 13C NMR chemical shift 0.5 ppm and 22.0 ppm, except end methyl carbons appearing at 13.8 ppm.
[0047] 5+ Methyl Carbons: is the number of methyl carbons attached to a methine carbon which is more than four carbons away from an end carbon appearing at 13C NMR chemical shift 19.6 ppm in an average isoparaffinic molecule.
[0048] The NMR spectra were acquired using Bruker AVANCE 500 spectrometer using a 5 mm BBI probe. Each sample was mixed 1:1 (wt:wt) with CDCl3. The 1H NMR was recorded at 500.11 MHz and using a 9.0 .mu.s (30.degree.) pulse applied at 4 s intervals with 64 scans co-added for each spectrum. The 13C NMR was recorded at 125.75 MHz using a 7.0 .mu.s pulse and with inverse gated decoupling, applied at 6 sec intervals with 4096 scans co-added for each spectrum. A small amount of 0.1 M Cr(acac)3 was added as a relaxation agent and TMS was used as an internal standard.
[0049] The branching properties of the lubricant base stock samples of the present invention are determined according to the following six-step process. Procedure is provided in detail in US 20050077208 A1, which is incorporated herein in its entirety. The following procedure is slightly modified to characterize the current set of samples:
[0050] 1) Identify the CH branch centers and the CH3 branch termination points using the DEPT Pulse sequence (Doddrell, D. T.; D. T. Pegg; M. R. Bendall, Journal of Magnetic Resonance 1982, 48, 323ff.).
[0051] 2) Verify the absence of carbons initiating multiple branches (quaternary carbons) using the APT pulse sequence (Patt, S. L.; J. N. Shoolery, Journal of Magnetic Resonance 1982, 46, 535ff.).
[0052] 3) Assign the various branch carbon resonances to specific branch positions and lengths using tabulated and calculated values (Lindeman, L. P., Journal of Qualitative Analytical Chemistry 43, 1971 1245ff; Netzel, D. A., et. al., Fuel, 60, 1981, 307ff.).
Branch NMR Chemical Shift (ppm)
TABLE-US-00002 [0053] TABLE 2 Describes ppm shift of alkyl branching by Carbon NMR Branch NMR Chemical Shift (ppm) 2-methyl 22.5 3-methyl 19.1 or 11.4 4-methyl 14.0 5+ methyl 19.6 Internal ethyl 10.8 n-propyl 14.4 Adjacent methyl 16.7
[0054] 4) Quantify the relative frequency of branch occurrence at different carbon positions by comparing the integrated intensity of its terminal methyl carbon to the intensity of a single carbon (total integral/number of carbons per molecule in the mixture). For example, number of 5+ methyl branches per molecule is calculated from the signal intensity at a chemical shift of 19.6 ppm relative to intensity of a single carbon.
[0055] For the unique case of the 2-methyl branch, where both the terminal and the branch methyl occur at the same resonance position, the intensity was divided by two before doing the frequency of branch occurrence calculation.
[0056] If the 4-methyl branch fraction is calculated and tabulated, its contribution to the 5+ methyls must be subtracted to avoid double counting.
[0057] Unknown methyl branches are calculated from contribution of signals that appear between 5.0 ppm and 22.5 ppm, however not including any branches reported in Table 2.
[0058] 5) Calculate the Branching Index (BI) and Branching Proximity (BP) using the calculations described in U.S. Pat. No. 6,090,989, which is incorporated by reference herein in its entirety.
[0059] 6) Calculate the total internal alkyl branches per molecule by adding up the branches found in steps 3 and 4, except the 2-methyl branches. These branches would include 3-methyl, 4-methyl, 5+ methyl, internal ethyl, n-propyl, adjacent methyl and unknown methyl.
[0060] FIMS Analysis: The hydrocarbon distribution of the current invention is determined by FIMS (field ionization mass spectroscopy). FIMS spectra were obtained on a Waters GCT-TOF mass spectrometer. The samples were introduced via a solid probe, which was heated from about 40.degree. C. to 500.degree. C. at a rate of 50.degree. C. per minute. The mass spectrometer was scanned from m/z 40 to m/z 1000 at a rate of 5 seconds per decade. The acquired mass spectra were summed to generate one averaged spectrum which provides carbon number distribution of paraffins and cycloparaffins containing up to six rings.
[0061] Hydrocarbon Structure and Properties The structure of the hydrocarbon mixtures disclosed herein are characterized by FIMS and NMR. FIMS analysis demonstrate that more than 80% of the molecules in the hydrocarbon mixtures have an even carbon number.
[0062] The unique branching structure of the hydrocarbon mixtures disclosed herein are characterized by NMR parameters, such as BP, BI, internal alkyl branching, and 5+ methyls. BP/BI of the hydrocarbon mixtures are in the range of .gtoreq.-0.6037 (Internal alkyl branching per molecule)+2.0. The 5+ methyls of the hydrocarbon mixtures average from 0.3 to 1.5 per molecule.
[0063] The hydrocarbon mixture can be classified into two carbon ranges based on the carbon number distribution, C28 to C40 carbons, and greater than or equal to C42. Generally, about or greater than 95% of the molecules present in each hydrocarbon mixture have carbon numbers within the specified range. Representative molecular structures for the C28 to C40 range can be proposed based on the NMR and FIMS analysis. Without wishing to be bound to any one particular theory, it is believed that the structures made by oligomerization and hydroisomerization of olefins has methyl, ethyl, butyl branches distributed throughout the structure and the branch index and branch proximity contribute to the surprisingly good low temperature properties of the product. Exemplary structures in the present hydrocarbon mixture are as follows:
##STR00001##
[0064] The unique branching structure and narrow carbon distribution of the hydrocarbon mixtures makes them suitable to be used as high-quality synthetic base oils, especially for low-viscosity engine oil applications. The hydrocarbon mixtures exhibit: [0065] a KV100 in the range of 3.0-10.0 cSt; [0066] a pour point in the range of -20 to -55.degree. C.; [0067] a Noack and CCS at -35.degree. C. relationship such that Noack is between 2750 (CCS at -35.degree. C.).sup.(-0.8).+-.2.
[0068] The Noack and CCS relationship for the hydrocarbon mixtures are shown in FIGS. 3 and 4. In each figure, the top line represents Noack=2750 (CCS at -35.degree. C.)(-0.8)+2 and the bottom graph line represents Noack=2750 (CCS at -35.degree. C.)(-0.8)-2. More preferably the hydrocarbon mixtures have a Noack and CCS at -35 C relationship such that the Noack is between Noack=2750 (CCS at -35.degree. C.)(-0.8)+0.5 and Noack=2750 (CCS at -35.degree. C.)(-0.8)-2. Hydrocarbon mixtures that are closer to the origin in FIGS. 3 and 4 have been found more advantageous for low viscosity engine oils due to the low volatility and decreased viscosity at -35.degree. C.
[0069] A hydrocarbon mixture in accordance with the present invention with carbon numbers in the range of C28 to C40, and in another embodiment carbon numbers in the range of from C28 to C36, or in another embodiment molecules with a carbon number of C32, will generally exhibit the following characteristics in addition to the characteristics of BP/BI, Internal alkyl branches per molecule, 5+ methyl branches per molecule, and Noack/CCS relationship described above: [0070] a KV100 in the range of 3.0-6.0 cSt; [0071] a VI in the range of 11 ln(BP/BI)+135 to 11 ln(BP/BI)+145; and [0072] a pour point in the range of 33 ln(BP/BI)-45 to 33 ln(BP/BI)-35.
[0073] In one embodiment, the KV100 for the C28-C40 hydrocarbon mixture ranges from 3.2 to 5.5 cSt; in another embodiment the KV100 ranges from 4.0 to 5.2 cSt; and from 4.1 to 4.5 cSt in another embodiment.
[0074] The VI for the C28-C40 hydrocarbon mixture ranges from 125 to 155 in one embodiment and from 135 to 145 in another embodiment.
[0075] The Pour Point of the hydrocarbon mixture, in one embodiment ranges from 25 to -55.degree. C. and from 35 to -45.degree. C. in another embodiment.
[0076] The boiling point range of the C28-C40 hydrocarbon mixture in one embodiment is no greater than 125.degree. C. (TBP at 95%-TBP at 5%) as measured by ASTM D2887; no greater than 100.degree. C. in another embodiment; no greater than 75.degree. C. in one embodiment; no greater than 50.degree. C. in another embodiment; and no greater than 30.degree. C. in one embodiment. In the preferred embodiments, those with a boiling point range no greater than 50.degree. C., and even more preferably no greater than 30.degree. C., give a surprisingly low Noack Volatility (ASTM D5800) for a given KV100.
[0077] The C28-C40 hydrocarbon mixture in one embodiment has a Branching Proximity (BP) in the range of 14-30 with a Branching Index (BI) in the range of 15-25; and in another embodiment a BP in the range of 15-28 and a BI in the range of 16-24.
[0078] The Noack volatility (ASTM D5800) of the C28-C40 hydrocarbon mixture is less than 16 wt. % in one embodiment; less than 12 wt. % in one embodiment; less than 10 wt. % in one embodiment; less than 8 wt. % in one embodiment and less than 7 wt. % in one embodiment. The C28-C40 hydrocarbon mixture in one embodiment also has a CCS viscosity at -35.degree. C. of less than 2700 cP; of less than 2000 cP in another embodiment; of less than 1700 cP in one embodiment; and less than 1500 cP in one embodiment.
[0079] The hydrocarbon mixture with the carbon number range of C42 and greater will generally exhibit the following characteristics, in addition to the characteristics of BP/BI, internal alkyl branches per molecule, 5+ methyl branches per molecule, and Noack and CCS at -35.degree. C. relationship described above: [0080] a KV100 in the range of 6.0-10.0 cSt; [0081] a VI in the range of 11 ln(BP/BI)+145 to 11 ln(BP/BI)+160; and [0082] a Pour Point in the range of 33 ln(BP/BI)-40 to 33 ln(BP/BI)-25.
[0083] The hydrocarbon mixture comprising C42 carbons or greater, in one embodiment has a KV100 in the range of 8.0 to 10.0 cSt, and in another embodiment from 8.5 to 9.5 cSt.
[0084] The VI of the hydrocarbon mixture having .gtoreq.42 carbons is 140-170 in one embodiment; and, from 150-160 in another embodiment.
[0085] The pour point in one embodiment ranges from -15 to -50.degree. C.; and, from -20 to -40.degree. C. in another embodiment.
[0086] In one embodiment, the hydrocarbon mixture comprising .gtoreq.42 carbons has a BP in the range of 18-28 with a BI in the range of 17-23. In another embodiment, the hydrocarbon mixture has a BP in the range of 18-28 and a BI in the range of 17-23.
[0087] In general, both hydrocarbon mixtures disclosed above exhibit the following characteristics: [0088] at least 80% of the molecules have an even carbon number according to FIMS; [0089] a KV100 in the range of 3.0-10.0 cSt; Sa pour point in the range of -20 to -55.degree. C.; [0090] a Noack and CCS @ -35.degree. C. relationship such that Noack is between 2750 (CCS @-35.degree. C.).sup.(-0.8)-2; [0091] a BP/BI in the range of .gtoreq.-0.6037 (Internal alkyl branching)+2.0 per molecule; and, [0092] on average from 0.3 to 1.5 5+ methyl branches per molecule.
Synthesis
[0093] Provided herein are possible processes or methods to make the disclosed hydrocarbon mixtures. The novel hydrocarbon mixtures disclosed herein can be synthesized via olefin oligomerization to achieve the desired carbon chain length, followed by hydroisomerization to improve their cold-flow properties, such as pour point and CCS, etc. In one embodiment, olefins of C14 to C20 in length are oligomerized using an acid catalyst to form an oligomer mixture. The olefins can be sourced from natural occurring molecules, such as crude oil or gas based olefins, or from ethylene polymerizations. In some variations, about 100% of the carbon atoms in the olefin feedstocks described herein may originate from renewable carbon sources. For example, an alpha-olefin co-monomer may be produced by oligomerization of ethylene derived from dehydration of ethanol produced from a renewable carbon source. In some variations, an alpha-olefin co-monomer may be produced by dehydration of a primary alcohol other than ethanol that is produced from a renewable carbon source. Said renewable alcohols can be dehydrated into olefins, using gamma alumina or sulfuric acid. In some embodiments, modified or partially hydrogenated terpene feedstocks derived from renewable resources are coupled with one or more olefins that are derived from renewable resources.
[0094] In one embodiment, an olefin monomer between C14 to C20 is oligomerized in the presence of BF3 and/or BF3 promoted with a mixture of an alcohol and/or an ester, such as a linear alcohol and an alkyl acetate ester, using a continuously stirred tank reactor (CSTR) with an average residence time of 60 to 400 minutes. In another embodiment, the C14 to C20 olefin monomers are oligomerized in the presence of BF3 and/or promoted BF3 using a continuously stirred tank reactor with an average residence time of 90 to 300 minutes. In yet another embodiment, the C14 to C20 the olefin monomers are oligomerized in the presence of BF3 and/or promoted BF3 using a continuously stirred tank reactor with an average residence time of 120 to 240 minutes. The temperature of the oligomerization reaction may be in a range of from 10.degree. C. to 90.degree. C. However, in one preferred embodiment, the temperature is maintained in the range of from 15 to 75.degree. C., and most preferably 20.degree. C. to 40.degree. C., for the duration of the reaction.
[0095] Suitable Lewis acids catalysts for the oligomerization process include metalloid halides and metal halides typically used as Friedel-Crafts catalysts, e.g., AlCl.sub.3, BF.sub.3, BF.sub.3 complexes, BCl.sub.3, AlBr.sub.3, TiCl.sub.3, TiCl.sub.4, SnCl.sub.4, and SbCl.sub.5. Any of the metalloid halide or metal halide catalysts can be used with or without a co-catalyst protic promoter (e.g., water, alcohol, acid, or ester). In one embodiment, the oligomerization catalyst is selected from the group consisting of zeolites, Friedel-Crafts catalysts, Bronsted acids, Lewis acids, acidic resins, acidic solid oxides, acidic silica aluminophosphates, Group IVB metal oxides, Group VB metal oxides, Group VIB metal oxides, hydroxide or free metal forms of Group VIII metals, and any combination thereof.
[0096] Proper control of the oligomerization reaction temperature and residence time within a CSTR is needed to ensure the dimer portion (C28-C40) of the oligomerization product has branching proximity (BP) between 25 to 35, preferably between 27-35, more preferably between 27-33, and most preferably between 28-32, if the dimer portion were to be saturated without isomerization to a Br index of less than 100 mg Br.sub.2/100 g (ASTM D2710). A branching proximity which is too low prior to hydroisomerization will lead to isomerized hydrocarbon mixtures that fall under the solid line in FIG. 1 and will result in a less desirable higher CCS viscosity at -35.degree. C. value for a given Noack volatility to fit within the range shown in FIGS. 3 and 4. Conversely, a branching proximity which is too high will require greater isomerization to reach an acceptable pour point, which will increase the Noack volatility and the CCS at -35.degree. C. simultaneously. In one embodiment, the unsaturated oligomer product is distilled to remove the unreacted monomer. For example, the unreacted monomer may be separated from the oligomer product, such as via distillation, and can be recycled back into the mixture of the first and/or second feedstocks for oligomerization thereof.
[0097] The oligomer product is then hydroisomerized to provide the additional internal alkyl branches required to achieve the ideal branching characteristics. In one embodiment, the whole oligomer product, including both the dimers (C28-C40) and heavier oligomers (.gtoreq.C42), are hydroisomerized prior to separation by distillation. The hydroisomerized product is then separated into the final hydrocarbon products by distillation. In another embodiment, the dimers and heavier oligomers are fractionated and hydroisomerized separately.
[0098] Hydroisomerization catalysts useful in the present invention usually comprises a shape-selective molecular sieve, a metal or metal mixture that is catalytically active for hydrogenation, and a refractory oxide support. The presence of a hydrogenation component leads to product improvement, especially VI and stability. Typical catalytically active hydrogenation metals include chromium, molybdenum, nickel, vanadium, cobalt, tungsten, zinc, platinum, and palladium. Platinum and palladium are especially preferred, with platinum mostly preferred. If platinum and/or palladium is used, the metal content is typically in the range of 0.1 to 5 weight percent of the total catalyst, usually from 0.1 to 2 weight percent, and not to exceed 10 weight percent. Hydroisomerization catalysts are discussed, for example, in U.S. Pat. Nos. 7,390,763 and 9,616,419, as well as U.S. Patent Application Publications 2011/0192766 and 2017/0183583.
[0099] The conditions for hydroisomerization are tailored to achieve an isomerized hydrocarbon mixture with specific branching properties, as described above, and thus will depend on the characteristics of feed used. The reaction temperature is generally between about 200.degree. C. and 400.degree. C., preferably between 260.degree. C. to 370.degree. C., most preferably between 288.degree. C. to 345.degree. C., at a liquid hourly space velocity (LHSV) generally between about 0.5 hr.sup.-1 and about 20 hr.sup.-1. The pressure is typically from about 15 psig to about 2500 psig, preferably from about 50 psig to about 2000 psig, more preferably from about 100 psig to about 1500 psig. Low pressure provides enhanced isomerization selectivity, which results in more isomerization and less cracking of the feed, thus leading to an increased yield.
[0100] Hydrogen is present in the reaction zone during the hydroisomerization process, typically in a hydrogen to feed ratio from about 0.1 to 10 MSCF/bbl (thousand standard cubic feet per barrel), preferably from about 0.3 to about 5 MSCF/bbl. Hydrogen may be separated from the product and recycled to the reaction zone.
[0101] In one embodiment, an additional step of hydrogenation is added before the hydroisomerization to protect the downstream hydroisomerization catalyst. In another embodiment, an additional step of hydrogenation or hydrofinishing is added after the hydroisomerization to further improve the saturation and stability of the hydrocarbon mixture.
[0102] The hydroisomerized hydrocarbon mixtures are comprised of dimers having carbon numbers in the range of C28-C40, and a mixture of trimers+having carbon numbers of C42 and greater. Each of the hydrocarbon mixtures will exhibit a BP/BI in the range of .gtoreq.-0.6037 (internal alkyl branching) 2.0 per molecule, and, on average, from 0.3 to 1.5 methyl branches on the fifth or greater position per molecule. Importantly, at least 80% of the molecules in each composition also have an even carbon number as determined by FIMS. In another embodiment, each of the hydrocarbon compositions will also exhibit a Noack and CCS at -35.degree. C. relationship such that the Noack is between 2750 (CCS at -35.degree. C.).sup.(-0.8)-2. These characteristics allow for the formulation of low-viscosity engine oils as well as many other high-performance lubricant products.
[0103] In one embodiment, C16 olefins are used as the feed for the oligomerization reaction. When using C16 olefins as the feed, the hydroisomerized dimer product generally exhibits a KV100 of 4.3 cSt with <8% Noack loss and a CCS at -35.degree. C. of approximately 1,700 cP. The extremely low Noack volatility is due to the high starting boiling point and narrow boiling point distribution when compared other 3.9 to 4.4 cSt synthetic base stocks. This makes it ideal for use in low viscosity engine oils with strict volatility requirements. The excellent CCS and pour point characteristics are due to the branching characteristics discussed above. In one embodiment, the material has a pour point of .ltoreq.-40.degree. C. This is required to pass critical engine oil formulation requirements for 0W formulations, including Mini-Rotary Viscosity (ASTM D4684) and Scanning Brookfield Viscosity (ASTM D2983) specifications.
Finished Lubricant Formulations for Improving Engine Performance
[0104] Herein is described the improvement of fuel economy, and the retention of fuel economy and LSPI prevention of ultra-low viscosity engine oils formulated using renewable base oils compared to conventional base oils. Finished lubricant compositions containing renewable base oils as herein described, not only provide reduced friction and consequently improved fuel economy when fresh, but also unexpectedly maintain reduced friction when aged compared to conventional base oils formulations. These benefits were leveraged into finished engine oil formulations to demonstrate the fuel economy improvement and fuel economy retention benefits using a modified Sequence VIF fuel economy engine test stand. The test results indicate that the use of renewable base oils in finished engine oil formulations provides an unexpected and significant fresh oil fuel economy improvement which cannot be explained from the viscometrics of the renewable-based lubricant. In addition, the aged oil fuel economy is remarkably well retained compared to conventional base oil engine oil formulations, amounting to an unprecedented fuel economy improvement and retention over the lifetime of the lubricant in the engine.
[0105] Also described herein is the reduced amount of Calcium increase over the extended duration of exposure to elevated operating temperature in the Sequence VIF engine, which will result in an improved capability to retain the LSPI prevention characteristics of the lubricant composition.
[0106] Lubricant compositions containing the renewable base oil herein described can be employed in a variety of lubricant-related end uses, such as a lubricant oil or grease for a device or apparatus requiring lubrication of moving and/or interacting mechanical parts, components, or surfaces. Useful apparatuses include engines and machines. More specific equipment includes, but is not limited to, gasoline fired engines, diesel fired engines, natural gas fired engines, gear boxes, wind turbines and circulating hydraulic pumps. Lubricant compositions containing the base oil as herein described may be use in the formulation of automotive crank case lubricants, automotive gear oils, transmission oils, many industrial lubricants including circulation lubricant, industrial gear lubricants, grease, compressor oil, pump oils, refrigeration lubricants, hydraulic lubricants, metal working fluids.
[0107] These benefits were observed in a Sequence VIF engine test modified for extended time/duration and elevated temperature. A Sequence VIF engine test as described in the ASTM D8226, as known to one of skill in the art, comprises a comparative fuel economy improvement (FEI) assessment of the fuel-saving capabilities of automotive engine oils under repeatable laboratory conditions for low-viscosity oils.
[0108] The test parameters are (1) The test duration is 196 hours, (2) Fuel consumption is measured for six speed/load/temperature test conditions for an SAE 20W-30 baseline (BL) lubricant to ensure consistent engine response, (3) The candidate lubricant is introduced and aged for 16 hours at aging conditions and then fuel consumption is measured for six test conditions for FEI 1, (4) The candidate lubricant is left in the engine and aged for 109 hours at aging conditions for FEI 2, (5) Fuel consumption for each of the six test conditions for BL is repeated at the end of the test to further ensure consistent engine response over the duration of the test. (5) FEI 1, FEI 2 and FEI Sum (FEI 1 plus FEI 2) are calculated from the comparisons of the fuel economy measurements for the candidate oil with the fuel economy measurements on the baseline lubricant (BL).
[0109] Fuel consumption is measured for six speed/load/temperature test conditions for an SAE 20W-30 baseline (BL) lubricant to ensure consistent engine response. The candidate lubricant is introduced and aged for 16 hours at aging conditions and then fuel consumption is measured for six test conditions (Table 3). Fuel consumption for each of the six test conditions for BL is repeated at the end of the test to further ensure consistent engine response over the duration of the test.
TABLE-US-00003 TABLE 3 Operating targets of a Sequence VIF engine test are: Test Conditions Stage 1 Stage 2 Stage 3 Stage 4 Stage 5 Stage 6 Speed, rpm 2000 2000 1500 695 695 695 Torque, Nm 105 105 105 20 20 40 Lubricant 100 65 100 100 35 100 Temp, .degree. C. Coolant-in 94 65 94 94 35 94 temp, .degree. C.
[0110] Modifications to this Sequence VIF engine test conditions are presented.
TABLE-US-00004 TABLE 4 Modification of sequence VIF conditions Stages Sequence VIF Mod. Sequence VIF 1 SOT BL FE 20W-30 20W-30 2 Flushing CO CO 3 Aging Phase 1 120.degree. C./16 h 120.degree. C./16 h 4 CO FE (FEI1) Test conditions Test conditions (Table 3) (Table 3)* 5 Aging Phase 2 120.degree. C./109 h 140.degree. C./144 h 6 CO FE (FEI2) Test conditions Test conditions (Table 3) (Table 3)* 7 Flushing 20W-30 -- 8 EOT BL FE 20W-30 -- 9 Aging Phase 3 -- 140.degree. C./144 h 10 CO FE (FEI3) -- Test conditions (Table 3)* 11 Flushing -- 20W-30 12 EOT BL FE -- 20W-30 SOT--Start of Test: BL--Baseline Oil; CO--Candidate Oil; FE--Fuel Economy Measurement; EOT--End of the test; *FEI 1, 2, 3 based on unweighted fuel consumption data
[0111] The lubricant of this invention comprises renewable base oil used in the base oil mixture and further comprises one or more of the following additives: dispersant, inhibitors, antioxidants, detergents, friction modifiers, pour point depressants, viscosity modifiers and the like.
[0112] In one embodiment the amount of renewable base oil in the total amount of base oil is at least 20 wt. %. In another embodiment, the amount of renewable base oil in the total amount of base oil is from about 20 wt. % to about 100 wt. %. As is known in the art, the fuel economy of an internal combustion engine is in part determined by the viscosity and by the frictional characteristics of the lubricant. The frictional characteristics of a lubricant are determined by the additive chemistry in a sense that the surface-active additives such as detergents anti-wear additives and friction modifiers can form a solid, friction-altering surface layer on the metal surfaces in the lubricated contacts in the engine. Base oils are not expected, by those skilled in the art, to play a role in altering the friction in an engine as long as the viscosity of the lubricants is not changed.
[0113] An embodiment of the invention describes a method to derive a fuel economy improvement from a lubricant that comprises a renewable base oil and has equal viscosity and the same additives as a comparative lubricant that does not contain any of the renewable base oil.
[0114] In one embodiment, the fuel economy improvement, when employing a lubricating oil composition comprising 90 wt. % of the renewable base oil and 10 wt. % of an additive combination that includes dispersants, detergents, inhibitors, friction modifier, and pour point depressant, is at least 0.3%, preferably 0.5% better than for a conventional lubricant of equal viscosity that does not comprise a renewable base oil.
[0115] As is known in the art, fuel economy improvement measurements are performed on fresh lubricants or on lubricants that have been exposed to a minor amount of aging at conditions that represent moderately severe driving conditions. Also known is that in many cases the driving conditions can be made more severe by elevated ambient conditions, or by exposing the lubricant to aging conditions for an extended duration, such as when oil change intervals are extended. However, there are no standardized tools to determine the fuel economy for a lubricant exposed to more severe aging conditions, see Table 4, line 5 for severe aging conditions as described herein.
[0116] This invention describes a method to derive a fuel economy retention (FER) benefit from a lubricant that comprises a renewable base oil and has equal viscosity and the same additives as a comparative lubricant that does not contain any of the renewable base oil. The fuel economy retention (FER) is evaluated after a period of aging representative of a typical shorter oil change interval. Separately, FER is evaluated after a period of aging representative of an extended oil change interval.
[0117] In one embodiment, the fuel economy retention at conditions representative of a shorter oil change interval, when employing a lubricating oil composition comprising 90 wt. % of the renewable base oil and 10 wt. % of an additive combination that includes dispersants, detergents, inhibitors, friction modifier, and pour point depressant, is at least 1.0% better, preferably 1.5% better than for a conventional lubricant of equal viscosity that does not comprise a renewable base oil.
[0118] In one embodiment, the fuel economy retention at conditions representative of a shorter oil change interval, when employing a lubricating oil composition comprising 45 wt. % of the renewable base oil, 45% nonrenewable base oil and 10 wt. % of an additive combination that includes dispersants, detergents, inhibitors, friction modifier, and pour point depressant, is at least 0.5% better, preferably 1.0% than a conventional lubricant of equal viscosity that does not comprise a renewable base oil.
[0119] In one embodiment, the fuel economy retention (FER) at conditions representative of an extended oil change interval, when employing a lubricating oil composition comprising 90 wt. % of the renewable base oil and 10 wt. % of an additive combination that includes dispersants, detergents, inhibitors, friction modifier, and pour point depressant, is at least 1.0% better, preferably 1.5% better than for a conventional lubricant of equal viscosity that does not comprise a renewable base oil.
[0120] In one embodiment, the fuel economy retention (FER) at conditions representative of an extended oil change interval, when employing a lubricating oil composition comprising 45 wt. % of the renewable base oil, 45% non-renewable base oil and 10 wt. % of an additive combination that includes dispersants, detergents, inhibitors, friction modifier, and pour point depressant, is at least 0.5% better, preferably 1.0% better than for a conventional lubricant of equal viscosity that does not comprise a renewable base oil.
[0121] In one embodiment, the fuel economy retention over a life of a lubricant, when employing a lubricating oil composition comprising 45 wt. % of the renewable base oil, 45% nonrenewable base oil and 10 wt. % of an additive combination that includes dispersants, detergents, inhibitors, friction modifier, and pour point depressant, is at least 0.5% better, preferably 1.0% better than a conventional lubricant of equal viscosity that does not comprise a renewable base oil.
[0122] "Life of lubricant" as described herein comprises the period from when the lubricant is put into the engine to the point where the condition of the lubricant has degraded to a level where engine damage may occur if the lubricant is not replenished.
[0123] In one embodiment, the fuel economy retention over a life of a lubricant, when employing a lubricating oil composition comprising 90 wt. % of the renewable base oil and 10 wt. % of an additive combination that includes dispersants, detergents, inhibitors, friction modifier, and pour point depressant, is at least 1.0% better, preferably 1.5% better than a conventional lubricant of equal viscosity that does not comprise a renewable base oil.
[0124] A further embodiment of the invention is a method of improving FER while retaining the low speed pre ignition (LSPI) prevention capability in the operation of an internal combustion engine over an extended period of time. Lubricant formulations were aged by exposing them to the higher temperature in the GM High Feature 3.6 L LY7 V6 engine attached to a dynamometer. Lubricant formulations were aged in 2 steps. In the aging stage 1, the lubricant formulation was subjected to 120.degree. C. for 16 hours. In the subsequent aging stage, lubricant formulation was subjected to 140.degree. C. for 288 hours. During the aging stages, the lubricant was sampled at every 25 hours for used oil analysis and chemical characterization to monitor changes in the lubricant additives during the test.
[0125] As shown in the table 9 and FIG. 6, the Comparative Oils (Examples 1, 2 and 3) all demonstrated a significant increase in the calcium content as they aged in the engine. The calcium content of Comparative Oils increased in the range of 20% to 76% at the end of the test. Comparatively, the lubricant compositions of the invention, as described herein maintained a calcium concentration very close to the original value throughout the test duration.
[0126] Previous studies have concluded that higher viscosity base stocks increase LSPI and volatility of the lubricants has insignificant influence on LSPI occurrences. In the present work, all the lubricants are formulated with base stocks with similar kinematic viscosities. Based on the conclusions from previous studies, one would predict that all four oils would show the same behavior with respect to LSPI occurrences. However, previous studies have concluded that increasing the concentration of calcium leads to an increase in the LSPI rate. Comparative Oils 1, 2 and 3 have shown a significant increase in the calcium content as these lubricants were exposed to higher temperature and extended duration aging. Based on the conclusion of previous studies and results shown in the table 9, we are predicting that all three Comparative Oils will show increasing LSPI occurrences with the aging. In contrast, the lubricant compositions as described herein will retain its LSPI prevention performance throughout the test.
[0127] In certain variations, a base stock prepared according to the methods described herein is blended with one or more additional base stocks, e.g., one or more commercially available PAOs, a Gas to Liquid (GTL) base stock, one or more mineral base stocks, a vegetable oil base stock, an algae-derived base stock, a second base stock as described herein, or any other type of renewable base stock. Any effective amount of additional base stock may be added to reach a blended base oil having desired properties. For example, blended base oils can comprise a ratio of a first base stock as described herein to a second base stock (e.g., a commercially available base oil PAO, a GTL base stock, one or more mineral base stocks, a vegetable oil base stock, an algae derived base stock, a second base stock as described herein) that is about is from about 1-99%, from about 1-80%, from about 1-70%, from about 1-60%, from about 1-50%, from about 1-40%, from about 1-30%, from about 1-20%, or from about 1-10%, based on the total weight of the composition may be made.
[0128] Also disclosed herein are lubricant compositions comprising a hydrocarbon mixture described herein. In some variations, the lubricant compositions comprise a base oil comprising at least a portion of a hydrocarbon mixture produced by any of the methods described herein, and one or more additives selected from the group of antioxidants, viscosity modifiers, pour point depressants, foam inhibitors, detergents, dispersants, dyes, markers, rust inhibitors or other corrosion inhibitors, emulsifiers, de-emulsifiers, antiwear agents, friction modifiers, thermal stability improvers, multifunctional additives (e.g., an additive that functions as both an antioxidant and a dispersant) or any combination thereof. Lubricant compositions may comprise hydrocarbon mixtures described herein and any lubricant additive, combination of lubricant additives, or available additive package.
[0129] Any of the compositions described herein that are used as a base stock may be present at greater than about 1% based on the total weight of a finished lubricant composition. In certain embodiments, the amount of the base stock in the formulation is greater than about 2, 5, 15 or 20 wt. % based on the total weight of the formulation. In some embodiments, the amount of the base oil in the composition is from about 1-99%, from about 1-80%, from about 1-70%, from about 1-60%, from about 1-50%, from about 1-40%, from about 1-30%, from about 1-20%, or from about 1-10% based on the total weight of the composition. In certain embodiments, the amount of base stock in formulations provided herein is about 1%, 5%, 7%, 10%, 13%, 15%, 20%, 30%, 40%, 5 As is known in the art, types and amounts of lubricant additives are selected in combination with a base oil so that the finished lubricant composition meets certain industry standards or specifications for specific applications. In general, the concentration of each of the additives in the composition, when used, may range from about 0.001 wt. % to about 20 wt. %, from about 0.01 wt. % to about 10 wt. %, from about 0.1 wt. % to about 5 wt. % or from about 0.1 wt. % to about 2.5 wt. %, based on the total weight of the composition. Further, the total amount of the additives in the composition may range from about 0.001 wt. % to about 50 wt. %, from about 0.01 wt. % to about 40 wt. %, from about 0.01 wt. % to about 30 wt. %, from about 0.01 wt. % to about 20 wt. %), from about 0.1 wt. % to about 10 wt. %, or from about 0.1 wt. % to about 5 wt. %, based on the total weight of the composition. 0%, 60%, 70%, 80%, 90%, or 99% based on total weight of the formulation.
[0130] In some variations, the base oils described herein are formulated in lubricant compositions for use as two cycle engine oils, as transmission oils, as hydraulic fluids, as compressor oils, as turbine oils and greases, as automotive engine oils, as gear oils, as marine lubricants, and as process oils. Process oils applications include but are not limited to: rolling mill oils, coning oils, plasticizers, spindle oils, polymeric processing, release agents, coatings, adhesives, sealants, polish and wax blends, drawing oils, and stamping oils, rubber compounding, pharmaceutical process aids, personal care products, and inks.
[0131] In yet other variations, the base oils described herein are formulated as industrial oil or grease formulations comprising at least one additive selected from antioxidants, anti-wear agents, extreme pressure agents, defoamants, detergent/dispersant, rust and corrosion inhibitors, thickeners, tackifiers, and demulsifiers. It is also contemplated that the base stocks of the invention may be formulated as dielectric heat transfer fluids composed of relatively pure blends of compounds selected from aromatic hydrocarbons, polyalphaolefins, polyol esters, and natural vegetable oils, along with additives to improve pour point, increase stability and reduce oxidation rate.
[0132] The present invention will be further illustrated by the following examples, which are not intended to be limiting.
EXAMPLES
Examples 1-6 (C28-C40 Hydrocarbon Mixtures)
Example 1
[0133] 1-Hexadecene with less than 8% branched and internal olefins was oligomerized under BF3 with a co-catalyst composition of Butanol and Butyl Acetate. The reaction was held at 20.degree. C. during semi-continuous addition of olefins and co-catalyst. The residence time was 90 minutes. The unreacted monomer was then distilled off, leaving behind less than 0.1% monomer distillation bottoms. A subsequent distillation was performed to separate the dimer from the trimer+ with less than 5% trimer remained in the dimer cut.
[0134] The dimers were then hydroisomerized with a noble-metal impregnated aluminoslicate of MRE structure type catalyst bound with alumina. The reaction was carried out in a fixed bed reactor at 500 psig and 307.degree. C. Cracked molecules were separated from the hydroisomerized C16 dimer using an online stripper.
Example 2
[0135] The oligomerization and subsequent distillation were performed identically to Example 1. The dimers were then hydroisomerized with a noble-metal impregnated aluminoslicate of MRE structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 313.degree. C. Cracked molecules were separated from the hydroisomerized C16 dimers using an online stripper.
Example 3
[0136] The oligomerization and subsequent distillation were performed identically to Example 1. The dimers were then hydroisomerized with a noble-metal impregnated aluminoslicate of MRE structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 324.degree. C. Cracked molecules were separated from the hydroisomerized C16 dimers using an online stripper.
Example 4
[0137] The oligomerization and subsequent distillation were performed identically to Example 1. The dimers were then hydroisomerized with a noble-metal impregnated aluminoslicate of MTT structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 316.degree. C. Cracked molecules were separated from the hydroisomerized C16 dimers using an online stripper.
Example 5
[0138] The oligomerization and subsequent distillation were performed identically to Example 1. The dimers were then hydroisomerized with a noble-metal impregnated aluminoslicate of MTT structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 321.degree. C. Cracked molecules were separated from the hydroisomerized C16 dimers using an online stripper.
Example 6
[0139] The oligomerization and subsequent distillation were performed identically to Example 1. The dimers were then hydroisomerized with a noble-metal impregnated aluminoslicate of MTT structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 332.degree. C. Cracked molecules were separated from the hydroisomerized C16 dimers using an online stripper.
Examples 7-12 (C42 Hydrocarbon Mixtures)
Example 7
[0140] 1-Hexadecene with less than 8% branched and internal olefins was oligomerized under BF3 with a co-catalyst composition of Butanol and Butyl Acetate. The reaction was held at 20.degree. C. during semi-continuous addition of olefins and co-catalyst. The residence time was 90 minutes. The unreacted monomer was then distilled off, leaving behind less than 0.1% monomer distillation bottoms. A subsequent distillation was performed to separate the dimer from the trimer and higher oligomers, the resulting dimer has less than 5% trimer.
[0141] The trimer and higher oligomers (trimer+) cut was then hydroisomerized with a noble-metal impregnated aluminoslicate of MRE structure type catalyst bound with alumina. The reaction was carried out in a fixed bed reactor at 500 psig and 313.degree. C. Cracked molecules were separated from the hydroisomerized C16 trimer+ using an online stripper.
Example 8
[0142] The oligomerization and subsequent distillations were performed identically to Example 7. The trimer+ cut was then hydroisomerized with a noble-metal impregnated aluminoslicate of MRE structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 318.degree. C. Cracked molecules were separated from the hydroisomerized C16 trimer+ using an online stripper.
Example 9
[0143] The oligomerization and subsequent distillations were performed identically to Example 7. The trimer+ cut was then hydroisomerized with a noble-metal impregnated aluminoslicate of MRE structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 324.degree. C. Cracked molecules were separated from the hydroisomerized C16 trimer+ using an online stripper.
Example 10
[0144] The oligomerization and subsequent distillations were performed identically to Example 7. The trimer+ cut was then hydroisomerized with a noble-metal impregnated aluminoslicate of MTT structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 321.degree. C. Cracked molecules were separated from the hydroisomerized C16 trimer+ using an online stripper.
Example 11
[0145] The oligomerization and subsequent distillations were performed identically to Example 7. The trimer+ cut was then hydroisomerized with a noble-metal impregnated aluminoslicate of MTT structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 327.degree. C. Cracked molecules were separated from the hydroisomerized C16 trimer+ using an online stripper.
Example 12
[0146] The oligomerization and subsequent distillations were performed identically to Example 7. The trimer+ cut was then hydroisomerized with a noble-metal impregnated aluminoslicate of MTT structure type catalyst bound with alumina. The reaction was carried out in a fix bed reactor at 500 psig and 332.degree. C. Cracked molecules were separated from the hydroisomerized C16 trimer+ using an online stripper.
[0147] Inspection results for the hydrocarbon mixtures obtained in examples 1-12 are summarized in Table 3 below.
TABLE-US-00005 TABLE 5 Pour CCS BP/ Internal 5+ KV40, KV100, Noack. Point at -35.degree. Example BI Alkyl Methyl cSt cSt VI wt. % (.degree. C.) C., cP No. 1 1.42 1.36 0.32 18.57 4.306 144 6.9 -30 1809 No. 2 1.19 1.67 0.50 18.67 4.297 142 NM* -36 1384 No. 3 0.80 2.24 0.95 19.01 4.290 136 7.9 -51 1581 No. 4 1.28 1.46 0.42 18.76 4.324 143 7.0 -32 1480 No. 5 1.06 2.20 0.60 18.85 4.313 141 NM* -38 1430 No. 6 0.75 2.21 0.88 18.99 4.303 138 8.0 -50 1558 No. 7 1.55 2.14 0.76 49.66 8.764 156 1.6 -19 26272 No. 8 1.30 2.57 1.17 49.99 8.744 154 1.6 -24 11278 No. 9 0.94 3.56 1.37 50.76 8.730 151 1.7 -34 10769 No. 10 1.41 2.75 1.14 48.93 8.642 156 1.9 -22 124967 No. 11 1.18 3.03 1.18 49.09 8.597 154 2.3 -28 18252 No. 12 0.95 2.94 1.29 49.44 8.533 150 2.2 -35 8589 *NM: not measure
[0148] Comparative GTL and PAO Base Stocks
[0149] Characterization results of comparable GTL and PAO samples used in FIGS. 1-4 are summarized in Table 4. GTL comparative examples shown in the following publications: GTL #1 WO2007068795, GTL #2 WO2007068795, GTL #3 US2005007720. The PAO comparative examples were measured using the techniques described above on commercially available samples.
TABLE-US-00006 TABLE 6 Pour CCS BP/ Internal 5+ KV40, KV100, Noack, Point at -35.degree. Samples BI Alkyl Methyl cSt cSt I wt. % (.degree. C.) C., cP GTL #1 0.46 2 2 GTL #2 0.23 4 2 GTL #3 1.57 1.86 1.75 23.62 5.488 82 -9 C10 PAO #1 0.91 1.03 0.00 16.60 3.831 24 13.6 <-60 1435 C10 PAO #2 0.91 0.90 0.00 16.77 3.828 21 14.1 <-60 1117 C10 PAO #3 0.77 1.52 0.14 16.61 3.809 19 14.8 <-60 1192 C10 PAO #4 0.94 0.77 0.00 16.59 3.803 21 12.6 <-60 1324 C10 PAO #5 0.77 1.82 0.18 46.33 7.746 36 3.7 <-60 C10 PAO #6 0.93 0.99 0.05 46.50 7.795 36 3.4 <-60 C12 PAO #1 1.30 0.43 0.00 23.71 4.990 41 6.5 -47 2260 C12 PAO #2 1.40 0.55 0.00 39.05 7.139 47 3.2 -42
[0150] When the foregoing data is depicted graphically, the important structural and property differences of the hydrocarbon mixtures of the present invention, as compared to prior art hydrocarbon mixtures, are clearly seen and support the surprisingly improved properties of the present hydrocarbon mixtures. FIGS. 1-4 graphically depict several of the above characterizations.
[0151] FIG. 1 illustrates the relationship between BP/BI and Internal Alkyl Branches per Molecule for the various hydrocarbon mixtures. The straight line in the plot depicts the equation of BP/BI -0.6037 (Internal alkyl branching per molecule)+2.0. All of the hydrocarbon mixtures of the present invention are above the line. While a few of the prior art hydrocarbon mixtures are also above the line, they do not meet other important characteristics of the present hydrocarbon mixtures, as shown in FIGS. 2-4.
[0152] FIG. 2 illustrates the relationship between BP/BI and 5+ Methyl Branches per Molecule for the various hydrocarbon mixtures. It demonstrates that the 5+ Methyl Branches per Molecules for the present hydrocarbon mixtures fall in a unique range of 0.3-1.5. All of the prior art mixtures fall outside the range.
[0153] FIGS. 3 and 4 illustrate the relationship between NOACK volatility and CCS at -35.degree. C. for the various hydrocarbon mixtures. Some commercially available Group III base oils, which do not meet the requirement of 80% even carbon number by FIMS, are additionally included. The solid line and dotted line depicts the upper limit and lower limit of the Noack vs. CCS at -35.degree. C. exhibited by the present unique hydrocarbon mixture, which are NOACK=2,750 (CCS at -35.degree. C.).sup.(-0.8)+2 and NOACK=2,750 (CCS at -35.degree. C.).sup.(-0.8)-2, respectively. It can be seen that all of the present hydrocarbon mixtures fall within the range, whereas essentially all of the prior art samples fall outside of the range with the exception of a PAO of a higher viscosity that does not have the desired branching as seen in FIGS. 1 and 2. FIG. 4 is an enlarged view of FIG. 3 in the range of 800-2,800 cP of CCS at -35.degree. C. In general, for an engine oil formulation, a preferable base stock will fall as close as possible to the origin of FIGS. 3 and 4, as a lower Noack volatility for a given CCS viscosity at -35.degree. C. is ideal for modern engine oil formulations such as 0W-20 through 0W-8 formulations.
[0154] The foregoing data and figures demonstrate the unique branching characteristics of the present hydrocarbon mixture, as characterized by NMR, and the resulting unique properties. The novel combination of structural characteristics has been found to lead to outstanding properties, including extremely low volatility and good low-temperature properties, which are important performance attributes of high-quality base stocks.
Example 13
[0155] TABLE 7 shows a comparison of three lubricant compositions; conventional capability, high volatility and lubricant compositions of the invention as described herein when used in a modified sequence VIF test for fuel economy improvement benefit. As shown in Table 7, the lubricant of the invention which contained 90 wt. % renewable base oil and 10 wt. % of an additive package containing dispersants, detergents, inhibitors, friction modifier, and pour point depressant, was blended to meet the viscosity grade definition of an SAE 0W-8 lubricant. The kinematic viscosity of the lubricant at 100.degree. C. was 5.47 cSt, the CCS Viscosity at -35.degree. C. was 2400 cP, and the Noack volatility was 7%. A comparative lubricant 1 was made with 90% of an API Group IV base oil and 10 wt. % of the same additive package containing dispersants, detergents, inhibitors, friction modifier, and pour point depressant. This comparative lubricant was also blended to meet the viscosity grade definition of an SAE 0W-8 lubricant. The kinematic viscosity of the comparative lubricant at 100.degree. C. was 4.82 cSt, the CCS Viscosity at -35.degree. C. was 2000 cP, and the Noack volatility was 10.8%. A third oil used for comparative purposes was purchased as it is a commercially available lubricating oil. This lubricant also met the viscosity grade definition of an SAE 0W-8 lubricant. The kinematic viscosity of the third commercially available lubricant at 100.degree. C. was 5.31 cSt, the CCS Viscosity at -35.degree. C. was 1236 cP, and the Noack volatility was 30.8%.
TABLE-US-00007 TABLE 7 Lubricating oil compositions and fuel economy improvements Lubricant of Comparative Examples the invention Example 1 Example 2 Group IV base oil -- 89.91 -- Renewable base oil 89.91 -- -- Additive Package 9.59 9.59 -- Pour Pt. Dep. 0.5 0.5 -- Group IV base oil (Diluent oil) -- -- -- Commercial Oil -- -- 100 Total 100 100 100 Kinematic Viscosity @ 100.degree. C. 5.5 4.8 5.3 CCS @ -35.degree. C. 2400 2000 1236 HTHS @ 150.degree. C. 1.94 1.71 1.96 SAE Viscosity Grade 0W-8 0W-8 0W-8 NOACK @ 250.degree. C. 6.5 10.8 30.9 % FE Change relative to ref. oil 3.7 2.52 2.84
[0156] A 5900 ml sample of the lubricant of the invention and the two comparative lubricants were added to a GM 3.6 L V-6 engine following a triple flushing procedure to eliminate all the remnants of the reference lubricant that is evaluated before and after each test on a candidate lubricant. Fuel Economy Improvements (FEI) are expressed as a percentage increase or reduction in the amount of fuel consumed relative to the fuel consumed on the reference lubricant which is an SAE 20W-30 Viscosity Grade oil. Table 7 points out that comparative lubricants of an SAE 0W-8 Viscosity Grade exhibit a fuel economy benefit based on their low viscosity, in the range of 2.5 to 3.0% Fuel Economy Improvement (FEI). The 3.7% FEI that was found on the lubricant of the invention is a highly surprising result as it improves the FE by 1.2% relative to the Conventional Capability Comparative Example 1, which represents a best-in-class result using high quality Group IV base oil. The result is especially surprising considering that the viscosity of these lubricants is nearly identical and the additive system, which is responsible for the frictional characteristics, was completely identical.
Example 14
TABLE-US-00008 [0157] TABLE 8 Lubricant of Comparative the invention Exam- Exam- Exam- Exam- ple 1 ple 2 ple 1 ple 2 Commercial Oil 100 -- -- -- Non-renewable -- 89.91 44.26 -- synthetic base oil Renewable base oil -- -- 46.65 89.91 Additive Package -- 9.59 9.59 9.59 Pour Pt. Dep. -- 0.5 0.5 0.5 Total 100 100 100 100 KV @ 100.degree. C. 5.3 4.8 4.4 5.5 CCS @ -35.degree. C. 1236 2000 1394 2400 HTHS @ 150.degree. C. 1.96 1.71 1.65 1.94 SAE Viscosity Grade 0W-8 0W-8 0W-8 0W-8 NOACK @ 250.degree. C. 30.9 10.8 14.2 6.5 % FE change relative to 1.17 1.88 3.07 3.92 ref. oil after typical drain interval (172 h) % FE change relative to -0.28 2.04 3.37 4 ref. oil after extended drain interval (328 h) Average % FE change 0.445 1.96 3.219 3.958 over life of a lubricant
[0158] TABLE 8 shows a comparison of four lubricant compositions, two showing conventional capability and two lubricant compositions of the invention as described herein when used in a modified Sequence VIF test for fuel economy retention benefit. As shown in the Table 8 lubricating oil composition of the invention 1 which contained 45 wt. % renewable base oil, 45% non-renewable base oil and 10 wt. % of an additive package containing dispersants, detergents, inhibitors, friction modifier, and pour point depressant, was blended to meet the viscosity grade definition of an SAE 0W-8 lubricant. The kinematic viscosity of the lubricant at 100.degree. C. was 4.4 cSt, the CCS Viscosity at -35.degree. C. was 1394 cP, and the Noack volatility was 14%. A lubricating oil composition of the invention 2 which contained 90 wt. % renewable base oil and 10 wt. % of an additive package containing dispersants, detergents, inhibitors, friction modifier, and pour point depressant, was blended to meet the viscosity grade definition of an SAE 0W-8 lubricant. The kinematic viscosity of the lubricant at 100.degree. C. was 5.5 cSt, the CCS Viscosity at -35.degree. C. was 2400 cP, and the Noack volatility was 7%. A comparative oil 1 used for comparative purposes was purchased as it is a commercially available lubricating oil. This lubricant also met the viscosity grade definition of an SAE 0W-8 lubricant. The kinematic viscosity of the third commercially available lubricant at 100.degree. C. was 5.31 cSt, the CCS Viscosity at -35.degree. C. was 1236 cP, and the Noack volatility was 30.8%. A comparative lubricant 2 was made with 90% of an API Group IV base oil and 10 wt. % of the same additive package containing dispersants, detergents, inhibitors, friction modifier, and pour point depressant. This comparative lubricant was also blended to meet the viscosity grade definition of an SAE 0W-8 lubricant. The kinematic viscosity of the comparative lubricant at 100.degree. C. was 4.82 cSt, the CCS Viscosity at -35.degree. C. was 2000 cP, and the Noack volatility was 10.8%.
[0159] 5900 ml samples of the lubricants of the invention and the two comparative lubricants were added to a GM 3.6 L V-6 engine following a triple flushing procedure to eliminate all the remnants of the reference lubricant that is evaluated before and after each test on a candidate lubricant. Fuel Economy Improvements (FEI) are expressed as a percentage increase or reduction in the amount of fuel consumed relative to the fuel consumed on the reference lubricant which is an SAE 20W-30 Viscosity Grade oil.
[0160] The GM 3.6 L engine was operated at conditions that are defined in more detail in the ASTM Sequence VIF engine test, with the exception of the lubricant temperature which was increased from 120.degree. C. to 140.degree. C. during the extended aging period. Detailed description of modified test conditions is summarized in table 4. Each candidate test was started with a fuel economy measurement on the lubricant in its fresh state, followed by additional fuel economy measurements after 172 h of lubricant aging and again after 328 h of lubricant aging. After 172 hours of aging, the lubricant would experience degradation similar to typical drain interval conditions. Hence fuel economy measurement conducted after 172 hours test duration represents degradation in the fuel economy at typical drain interval conditions. After 328 hours of aging, the lubricant would experience degradation similar to extended drain interval conditions. Hence fuel economy measurement conducted after 328 hours test duration represents degradation in the fuel economy at extended drain interval conditions. The aging conditions are defined in more detail in the ASTM Sequence VIF engine test. The fuel economy evaluations were based on the total fuel consumption measurements of the 6 stages that are defined in more detail in the table 3.
[0161] Table 8 also summarizes fuel economy change (%) relative to the reference oil after lubricants were aged similar to typical and extended drain interval conditions. Furthermore, table 8 summarizes average change in the fuel economy (%) over the life of a lubricants. Average change in the fuel economy values were calculated by averaging the values of fuel economy change (%) at typical and extended drain intervals.
[0162] As shown in the table 8, comparative lubricants after aging in the engine at elevated temperature, show a quickly increasing disadvantage relative to the lubricant of the invention, which does not suffer at all from a loss of fuel economy benefit, in other words, it has superior fuel economy retention over the life of the lubricant. For example, when comparative example 1 and 2 were aged similar to typical drain interval conditions, they exhibit fuel economy improvement in the range of 1% to 2%. While lubricants of the invention retain fuel economy improvement benefits in the range of 3% to 4%.
[0163] Furthermore, when comparative examples 1 was aged similar to extended drain interval conditions, it lost all its fuel economy improvement benefits and showed higher fuel consumption than the reference oil. Comparative example 2 maintained its fuel economy benefits around 2% when it was aged similar to extended drain interval conditions. While lubricants of the invention retained fuel economy improvement benefits in the range of 3% to 4% when they were aged in the extended drain interval conditions.
[0164] In typical and extended drain interval conditions, lubricants of the invention 1 and 2 shows fuel economy retention benefits by about 1% to 2% better than comparative examples, which represents best-in-class results using high quality group IV base oil. These results are highly surprising. The improvement in the fuel economy retention benefits cannot be explained by differences in the NOACK volatility. For example, in the extended drain interval conditions, high volatility comparative examples 1 and best-in-class comparative 2 shows 20% NOACK volatility difference and about 2% fuel economy benefits. Based on this, one skilled in the art would expect the lubricant of the invention 2 would bring 0.4% fuel economy benefits based on 4% NOACK volatility difference between comparative example 2 and lubricant of the invention 2. The result however points out that there is >2% fuel economy benefits between comparative example 2 and lubricant of the invention 2. Furthermore, the lubricant of the invention 1 exhibited a 1.3% fuel economy improvement relative to best in class comparative example 2 even though its NOACK volatility is about 4% higher.
TABLE-US-00009 TABLE 9 Lubricant Lubricant Exam- Exam- of the of the ple 1 ple 2 invention 1 invention 2 Commercial Oil 100 -- -- -- Non-renewable -- 89.91 44.26 -- synthetic base oil Renewable base oil -- -- 46.65 89.91 Additive Package -- 9.59 9.59 9.59 Pour Pt. Dep. -- 0.5 0.5 0.5 Total 100 100 100 100 KV @ 100.degree. C. 5.3 4.8 4.4 5.5 CCS @ -35.degree. C. 1236 2000 1394 2400 HTHS @ 150.degree. C. 1.96 1.71 1.65 1.94 SAE Viscosity Grade 0W-8 0W-8 0W-8 0W-8 NOACK @ 250.degree. C. 30.9 10.8 14.2 6.5 Ca at start of the test 2006 1585 1597 1573 Ca at end of the test 3534 1902 2024 1595 (340 hr) % increase in Ca 76.17 20% 26.73 1.3%
[0165] TABLE 9 shows a comparison of four lubricant compositions, two showing conventional capability and two lubricant compositions of the invention as described herein when used in a modified Sequence VIF test, showing the reduced level of Calcium concentration increase after exposure to elevated temperature in the engine over the duration of an extended drain interval.
REFERENCES
[0166] 1. Amann, M., Alger, T., and Mehta, D., "The Effect of EGR on Low-Speed Pre-Ignition in Boosted SI Engines," SAE Int. J Engines 4(1):235-245, 2011, doi:10.4271/2011-01-0339. [0167] 2. Amann, M., Alger, T., Westmoreland, B., and Rothmaier, A., "The Effects of Piston Crevices and Injection Strategy on Low-Speed Pre-Ignition in Boosted SI Engines," SAE Int. J. Engines 5(3):1216-1228, 2012, doi:10.4271/2012-01-1148. [0168] 3. Ritchie, A., Boese, D., and Young, A., "Controlling Low-Speed Pre-Ignition in Modern Automotive Equipment Part 3: Identification of Key Additive Component Types and Other Lubricant Composition Effects on Low-Speed Pre-Ignition," SAE Int. J. Engines 9(2):832-840, 2016, doi:10.4271/2016-01-0717. [0169] 4. Takeuchi, K., Fujimoto, K., Hirano, S., and Yamashita, M., "Investigation of Engine Oil Effect on Abnormal Combustion in Turbocharged Direct Injection--Spark Ignition Engines," SAE Int. J. Fuels Lubr. 5(3):1017-1024, 2012, doi:10.4271/2012-01-1615. [0170] 5. Takeuchi, K., Fujimoto, K., Hirano, S., and Yamashita, M., "Investigation of Engine Oil Effect on Abnormal Combustion in Turbocharged Direct Injection--Spark Ignition Engines," SAE Int. J. Fuels Lubr. 5(3):1017-1024, 2012, doi:10.4271/2012-01-1615. [0171] 6. Boese D. a. R. A., "Controlling low-speed pre-ignition in modern automotive equipment: Defining approaches to and methods for analyzing data in new studies of lubricant- and fuel-related effects," in [0172] 7. Presentation at 20th International Colloquium Tribology, Germany, Jan. 12-14, 2016. [0173] 8. Onodera, K., Kato, T., Ogano, S., Fujimoto, K. et al., "Engine Oil Formulation Technology to Prevent Pre-ignition in Turbocharged Direct Injection Spark Ignition Engines," SAE Technical Paper 2015-01-2027, 2015, doi:10.4271/2015-01-2027. [0174] 9. Boese, D., Ritchie, A., and Young, A., "Controlling Low-Speed Pre-Ignition in Modern Automotive Equipment: Defining Approaches to and Methods for Analyzing Data in New Studies of Lubricant and Fuel-Related Effects (Part 2)," SAE Technical Paper 2016-01-0716, 2016, doi:10.4271/2016-01-0716. [0175] 10. Dahnz, C., Han, K., Spicher, U. et al., "Investigations on Pre-Ignition in Highly Supercharged SI Engines," SAE Int. J. Engines 3(1):214-224, 2010, doi:10.4271/2010-01-0355. [0176] 11. Takeuchi, K., Fujimoto, K., Hirano, S., and Yamashita, M., "Investigation of Engine Oil Effect on Abnormal Combustion in Turbocharged Direct Injection--Spark Ignition Engines," SAE Int. J. Fuels Lubr. 5(3):1017-1024, 2012, doi:10.4271/2012-01-1615. [0177] 12. Kocsis, M., Briggs, T., and Anderson, G., "The Impact of Lubricant Volatility, Viscosity and Detergent Chemistry on Low Speed Pre-Ignition Behavior," SAE Int. J. Engines 10(3):2017, doi:10.4271/2017-01-0685. [0178] 13. Michlberger, A., Sutton, M., and Dohner, B., "Low Speed Pre-Ignition (LSPI) Durability--A Study of LSPI in Fresh and Aged Engine Oils," SAE Technical Paper 2018-01-0934, 2018, doi:10.4271/2018-01-0934. [0179] 14. Hirano, S., Yamashita, M., Fujimoto, K., and Kato, K., "Investigation of Engine Oil Effect on Abnormal Combustion in Turbocharged Direct Injection--Spark Ignition Engines (Part 2)," SAE Technical Paper 2013-01-2569, 2013, doi:10.4271/2013-01-2569. [0180] 15. Andrews, A., Burns, R., Dougherty, R., Deckman, D. et al., "Investigation of Engine Oil Base Stock Effects on Low Speed Pre-Ignition in a Turbocharged Direct Injection SI Engine," SAE Int. J. Fuels Lubr. 9(2):2016, doi:10.4271/2016-01-9071.
* * * * *

D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.