Method Of Promoting Bone Growth By An Anti-actriia Antibody
Knopf; John ; et al.
U.S. patent application number 16/851975 was filed with the patent office on 2021-02-11 for method of promoting bone growth by an anti-actriia antibody. The applicant listed for this patent is Acceleron Pharma Inc.. Invention is credited to John Knopf, Jasbir Seehra.
| Application Number | 20210040192 16/851975 |
| Document ID | / |
| Family ID | 1000005168495 |
| Filed Date | 2021-02-11 |












View All Diagrams
| United States Patent Application | 20210040192 |
| Kind Code | A1 |
| Knopf; John ; et al. | February 11, 2021 |
METHOD OF PROMOTING BONE GROWTH BY AN ANTI-ACTRIIA ANTIBODY
Abstract
In certain aspects, the present invention provides compositions and methods for promoting bone growth and increasing bone density.
| Inventors: | Knopf; John; (Carlisle, MA) ; Seehra; Jasbir; (Lexington, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005168495 | ||||||||||
| Appl. No.: | 16/851975 | ||||||||||
| Filed: | April 17, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16567370 | Sep 11, 2019 | |||
| 16851975 | ||||
| 16266919 | Feb 4, 2019 | |||
| 16567370 | ||||
| 15278813 | Sep 28, 2016 | 10239940 | ||
| 16266919 | ||||
| 13939976 | Jul 11, 2013 | 9480742 | ||
| 15278813 | ||||
| 13357264 | Jan 24, 2012 | 8486403 | ||
| 13939976 | ||||
| 12387788 | May 6, 2009 | 8128933 | ||
| 13357264 | ||||
| 11603485 | Nov 22, 2006 | 7612041 | ||
| 12387788 | ||||
| 61126761 | May 6, 2008 | |||
| 60844855 | Sep 15, 2006 | |||
| 60783322 | Mar 17, 2006 | |||
| 60739462 | Nov 23, 2005 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 38/179 20130101; A61K 39/3955 20130101; C12N 15/1138 20130101; C07K 2319/30 20130101; A61K 38/00 20130101; C12N 2310/11 20130101; C07K 14/575 20130101; A61K 2039/505 20130101; C07K 16/22 20130101; C07K 14/71 20130101; C07K 2317/76 20130101; A61K 45/06 20130101; C12N 15/1136 20130101 |
| International Class: | C07K 16/22 20060101 C07K016/22; C07K 14/575 20060101 C07K014/575; C07K 14/71 20060101 C07K014/71; A61K 39/395 20060101 A61K039/395; A61K 45/06 20060101 A61K045/06; A61K 38/17 20060101 A61K038/17; C12N 15/113 20060101 C12N015/113 |
Claims
1. A method for promoting bone growth, increasing bone density, or increasing bone strength, the method comprising administering to a subject in need thereof an effective amount of an anti-ActRIIa antibody.
2. The method of claim 1, wherein the anti-ActRIIa antibody is a monoclonal antibody.
3. The method of claim 1, wherein the anti-ActRIIa antibody is a humanized or fully human antibody.
4. The method of claim 1, wherein the method is for promoting bone growth in a subject in need thereof.
5. The method of claim 1, wherein the method is for increasing bone density in a subject in need thereof.
6. The method of claim 1, wherein the method is for increasing bone strength in a subject in need thereof.
7. The method of claim 1, wherein the method further comprises administering a second bone-active agent.
8. The method of claim 7, wherein the bone-active agent is selected from: a bisphosphonate, an estrogen, a selective estrogen-receptor modulator, a parathyroid hormone, a calcitonin, a calcium supplement, and a vitamin D supplement.
9. A method for treating a bone-related disorder, the method comprising administering to a subject in need thereof an effective amount of an anti-ActRIIa antibody, wherein the bone-related disorder is associated with low bone density or decreased bone strength.
10. The method of claim 9, wherein the anti-ActRIIa antibody is a monoclonal antibody.
11. The method of claim 9, wherein the anti-ActRIIa antibody is a humanized or fully human antibody.
12. The method of claim 9, wherein the method further comprises administering a second bone-active agent.
13. The method of claim 12, wherein the bone-active agent is selected from: a bisphosphonate, an estrogen, a selective estrogen-receptor modulator, a parathyroid hormone, a calcitonin, a calcium supplement, and a vitamin D supplement.
14. The method of claim 9, wherein the bone-related disorder is primary osteoporosis.
15. The method of claim 9, wherein the bone-related disorder is post-menopausal osteoporosis.
16. The method of claim 9, wherein the bone-related disorder is secondary osteoporosis.
17. The method of claim 9, wherein the bone-related disorder is hypogonadal bone loss.
18. The method of claim 9, wherein the subject has a cancer that is associated with bone loss.
19. The method of claim 9, wherein the subject is the recipient of a cancer treatment regimen than is associated with bone loss.
20. The method of claim 9, wherein the bone-related disorder is bone metastases.
21-23. (canceled)
Description
RELATED APPLICATIONS
[0001] This application is a continuation of U.S. application Ser. No. 16/567,370, filed Sep. 11, 2019, which is a continuation of U.S. application Ser. No. 16/266,919, filed Feb. 4, 2019 (now abandoned), which is a continuation of U.S. application Ser. No. 15/278,813, filed Sep. 28, 2016 (now U.S. Pat. No. 10,239,940), which is a continuation of U.S. application Ser. No. 13/939,976, filed on Jul. 11, 2013 (now U.S. Pat. No. 9,480,742), which is a continuation of U.S. application Ser. No. 13/357,264, filed Jan. 24, 2012 (now U.S. Pat. No. 8,486,403), which is a divisional of U.S. application Ser. No. 12/387,788, filed May 6, 2009 (now U.S. Pat. No. 8,128,933), which is a continuation-in-part of and claims priority to U.S. application Ser. No. 11/603,485, filed Nov. 22, 2006 (now U.S. Pat. No. 7,612,041), which claims the benefit of U.S. Provisional Application No. 60/739,462, filed Nov. 23, 2005, 60/783,322, filed Mar. 17, 2006, and 60/844,855, filed Sep. 15, 2006. Application Ser. No. 12/387,788 also claims the benefit of U.S. Provisional Application No. 61/126,761, filed May 6, 2008. All the specifications of each of the foregoing applications are hereby incorporated by reference in their entirety.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing, which has been submitted via EFS-Web and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Apr. 14, 2020 is named 1848179-0002-035-107_Seq.txt and is 24,877 bytes in size.
BACKGROUND OF THE INVENTION
[0003] Disorders of the bone, ranging from osteoporosis to fractures, represent a set of pathological states for which there are few effective pharmaceutical agents. Treatment instead focuses on physical and behavioral interventions, including immobilization, exercise and changes in diet. It would be beneficial to have therapeutic agents that promote bone growth and increase bone density for the purpose of treating a variety of bone disorders.
[0004] Bone growth and mineralization are dependent on the activities of two cell types, osteoclasts and osteoblasts, although chondrocytes and cells of the vasculature also participate in critical aspects of these processes. Developmentally, bone formation occurs through two mechanisms, endochondral ossification and intramembranous ossification, with the former responsible for longitudinal bone formation and the later responsible for the formation of topologically flat bones, such as the bones of the skull. Endochondral ossification requires the sequential formation and degradation of cartilaginous structures in the growth plates that serve as templates for the formation of osteoblasts, osteoclasts, the vasculature and subsequent mineralization. During intramembranous ossification, bone is formed directly in the connective tissues. Both processes require the infiltration of osteoblasts and subsequent matrix deposition.
[0005] Fractures and other structural disruptions of bone are healed through a process that, at least superficially, resembles the sequence of developmental events of osteogenesis, including the formation of cartilaginous tissue and subsequent mineralization. The process of fracture healing can occur in two ways. Direct or primary bone healing occurs without callus formation. Indirect or secondary bone healing occurs with a callus precursor stage. Primary healing of fractures involves the reformation of mechanical continuity across a closely-set disruption. Under suitable conditions, bone-resorbing cells surrounding the disruption show a tunneling resorptive response and establish pathways for the penetration of blood vessels and subsequent healing. Secondary healing of bones follows a process of inflammation, soft callus formation, callus mineralisation and callus remodeling. In the inflammation stage, haematoma and haemorrhage formation results from the disruption of periosteal and endosteal blood vessels at the site of injury. Inflammatory cells invade the area. In soft callus formation stage, the cells produce new vessels, fibroblasts, intracellular material and supporting cells, forming granulation tissue in the space between the fracture fragments. Clinical union across the disruption is established by fibrous or cartilaginous tissue (soft callus). Osteoblasts are formed and mediate the mineralization of soft callus, which is then replaced by lamellar bone and subjected to the normal remodeling processes.
[0006] In addition to fractures and other physical disruptions of bone structure, loss of bone mineral content and bone mass can be caused by a wide variety of conditions and may result in significant medical problems. Changes to bone mass occur in a relatively predictable way over the life of an individual. Up to about age 30, bones of both men and women grow to maximal mass through linear growth of the endochondral growth plates and radial growth. After about age 30 (for trabecular bone, e.g., flat bones such as the vertebrae and pelvis) and age 40 (for cortical bone, e.g., long bones found in the limbs), slow bone loss occurs in both men and women. In women, a final phase of substantial bone loss also occurs, probably due to postmenopausal estrogen deficiencies. During this phase, women may lose an additional 10% of bone mass from the cortical bone and 25% from the trabecular compartment. Whether progressive bone loss results in a pathological condition such as osteoporosis depends largely on the initial bone mass of the individual and whether there are exacerbating conditions.
[0007] Bone loss is sometimes characterized as an imbalance in the normal bone remodeling process. Healthy bone is constantly subject to remodeling. Remodeling begins with resorption of bone by osteoclasts. The resorbed bone is then replaced by new bone tissue, which is characterized by collagen formation by osteoblasts, and subsequent calcification. In healthy individuals the rates of resorption and formation are balanced. Osteoporosis is a chronic, progressive condition, marked by a shift towards resorption, resulting in an overall decrease in bone mass and bone mineralization. Osteoporosis in humans is preceded by clinical osteopenia (bone mineral density that is greater than one standard deviation but less than 2.5 standard deviations below the mean value for young adult bone). Worldwide, approximately 75 million people are at risk for osteoporosis.
[0008] Thus, methods for controlling the balance between osteoclast and osteoblast activity can be useful for promoting the healing of fractures and other damage to bone as well as the treatment of disorders, such as osteoporosis, associated with loss of bone mass and bone mineralization.
[0009] With respect to osteoporosis, estrogen, calcitonin, osteocalcin with vitamin K, or high doses of dietary calcium are all used as therapeutic interventions. Other therapeutic approaches to osteoporosis include bisphosphonates, parathyroid hormone, calcimimetics, statins, anabolic steroids, lanthanum and strontium salts, and sodium fluoride. Such therapeutics, however, are often associated with undesirable side effects.
[0010] Thus, it is an object of the present disclosure to provide compositions and methods for promoting bone growth and mineralization.
SUMMARY OF THE INVENTION
[0011] In part, the disclosure demonstrates that molecules having activin or ActRIIa antagonist activity ("activin antagonists" and "ActRIIa antagonists") can be used to increase bone density, promote bone growth, and/or increase bone strength. In particular, the disclosure demonstrates that a soluble form of ActRIIa acts as an inhibitor of activin-ActRIIa signaling and promotes increased bone density, bone growth, and bone strength in vivo. While most pharmaceutical agents that promote bone growth or inhibit bone loss act as either anti-catabolic agents (also commonly referred to as "catabolic agents") (e.g., bisphosphonates) or anabolic agents (e.g., parathyroid hormone, PTH, when appropriately dosed), the soluble ActRIIa protein exhibits dual activity, having both catabolic and anabolic effects. Thus, the disclosure establishes that antagonists of the activin-ActRIIa signaling pathway may be used to increase bone density and promote bone growth. While soluble ActRIIa may affect bone through a mechanism other than activin antagonism, the disclosure nonetheless demonstrates that desirable therapeutic agents may be selected on the basis of an activin-ActRIIa antagonist activity. Therefore, in certain embodiments, the disclosure provides methods for using activin-ActRIIa antagonists, including, for example, activin-binding ActRIIa polypeptides, anti-activin antibodies, anti-ActRIIa antibodies, activin- or ActRIIa-targeted small molecules and aptamers, and nucleic acids that decrease expression of activin and ActRIIa, to treat disorders associated with low bone density or low bone strength, such as osteoporosis, or to promote bone growth in patients in need thereof, such as in patients having a bone fracture. Additionally, the soluble ActRIIa polypeptide promotes bone growth without causing a consistently measurable increase in muscle mass
[0012] In certain aspects, the disclosure provides polypeptides comprising a soluble, activin-binding ActRIIa polypeptide that binds to activin. ActRIIa polypeptides may be formulated as a pharmaceutical preparation comprising the activin-binding ActRIIa polypeptide and a pharmaceutically acceptable carrier. Preferably, the activin-binding ActRIIa polypeptide binds to activin with a K.sub.D less than 1 micromolar or less than 100, 10 or 1 nanomolar. Optionally, the activin-binding ActRIIa polypeptide selectively binds activin versus GDF11 and/or GDF8, and preferably with a K.sub.D that is at least 10-fold, 20-fold or 50-fold lower with respect to activin than with respect to GDF11 and/or GDF8. While not wishing to be bound to a particular mechanism of action, it is expected that this degree of selectivity for activin inhibition over GDF11/GDF8 inhibition accounts for the selective effect on bone without a consistently measurable effect on muscle. In many embodiments, an ActRIIa polypeptide will be selected for causing less than 15%, less than 10% or less than 5% increase in muscle at doses that achieve desirable effects on bone. Preferably the composition is at least 95% pure, with respect to other polypeptide components, as assessed by size exclusion chromatography, and more preferably, the composition is at least 98% pure. An activin-binding ActRIIa polypeptide for use in such a preparation may be any of those disclosed herein, such as a polypeptide having an amino acid sequence selected from SEQ ID NOs: 2, 3, 7 or 12, or having an amino acid sequence that is at least 80%, 85%, 90%, 95%, 97% or 99% identical to an amino acid sequence selected from SEQ ID NOs: 2, 3, 7, 12 or 13. An activin-binding ActRIIa polypeptide may include a functional fragment of a natural ActRIIa polypeptide, such as one comprising at least 10, 20 or 30 amino acids of a sequence selected from SEQ ID NOs: 1-3 or a sequence of SEQ ID NO: 2, lacking the C-terminal 10 to 15 amino acids (the "tail").
[0013] A soluble, activin-binding ActRIIa polypeptide may include one or more alterations in the amino acid sequence (e.g., in the ligand-binding domain) relative to a naturally occurring ActRIIa polypeptide. Examples of altered ActRIIa polypeptides are provided in WO 2006/012627, pp. 59-60, incorporated by reference herein. The alteration in the amino acid sequence may, for example, alter glycosylation of the polypeptide when produced in a mammalian, insect or other eukaryotic cell or alter proteolytic cleavage of the polypeptide relative to the naturally occurring ActRIIa polypeptide.
[0014] An activin-binding ActRIIa polypeptide may be a fusion protein that has, as one domain, an ActRIIa polypeptide (e.g., a ligand-binding portion of an ActRIIa) and one or more additional domains that provide a desirable property, such as improved pharmacokinetics, easier purification, targeting to particular tissues, etc. For example, a domain of a fusion protein may enhance one or more of in vivo stability, in vivo half life, uptake/administration, tissue localization or distribution, formation of protein complexes, multimerization of the fusion protein, and/or purification. An activin-binding ActRIIa fusion protein may include an immunoglobulin Fc domain (wild-type or mutant) or a serum albumin or other polypeptide portion that provides desirable properties such as improved pharmacokinetics, improved solubility or improved stability. In a preferred embodiment, an ActRIIa-Fc fusion comprises a relatively unstructured linker positioned between the Fc domain and the extracellular ActRIIa domain. This unstructured linker may correspond to the roughly 15 amino acid unstructured region at the C-terminal end of the extracellular domain of ActRIIa (the "tail"), or it may be an artificial sequence of 1, 2, 3, 4 or 5 amino acids or a length of between 5 and 15, 20, 30, 50 or more amino acids that are relatively free of secondary structure, or a mixture of both. A linker may be rich in glycine and proline residues and may, for example, contain a single sequence of threonine/serine and glycines or repeating sequences of threonine/serine and glycines (e.g., TG.sub.4 (SEQ ID NO: 15) or SG.sub.4 (SEQ ID NO: 16) singlets or repeats). A fusion protein may include a purification subsequence, such as an epitope tag, a FLAG tag, a polyhistidine sequence, and a GST fusion. Optionally, a soluble ActRIIa polypeptide includes one or more modified amino acid residues selected from: a glycosylated amino acid, a PEGylated amino acid, a farnesylated amino acid, an acetylated amino acid, a biotinylated amino acid, an amino acid conjugated to a lipid moiety, and an amino acid conjugated to an organic derivatizing agent. A pharmaceutical preparation may also include one or more additional compounds such as a compound that is used to treat a bone disorder. Preferably, a pharmaceutical preparation is substantially pyrogen free. In general, it is preferable that an ActRIIa protein be expressed in a mammalian cell line that mediates suitably natural glycosylation of the ActRIIa protein so as to diminish the likelihood of an unfavorable immune response in a patient. Human and CHO cell lines have been used successfully, and it is expected that other common mammalian expression systems will be useful.
[0015] As described herein, ActRIIa proteins designated ActRIIa-Fc (a form with a minimal linker between the ActRIIa portion and the Fc portion) have desirable properties, including selective binding to activin versus GDF8 and/or GDF11, high affinity ligand binding and serum half life greater than two weeks in animal models. In certain embodiments the invention provides ActRIIa-Fc polypeptides and pharmaceutical preparations comprising such polypeptides and a pharmaceutically acceptable excipient.
[0016] In certain aspects, the disclosure provides nucleic acids encoding a soluble activin-binding ActRIIa polypeptide. An isolated polynucleotide may comprise a coding sequence for a soluble, activin-binding ActRIIa polypeptide, such as described above. For example, an isolated nucleic acid may include a sequence coding for an extracellular domain (e.g., ligand-binding domain) of an ActRIIa and a sequence that would code for part or all of the transmembrane domain and/or the cytoplasmic domain of an ActRIIa, but for a stop codon positioned within the transmembrane domain or the cytoplasmic domain, or positioned between the extracellular domain and the transmembrane domain or cytoplasmic domain. For example, an isolated polynucleotide may comprise a full-length ActRIIa polynucleotide sequence such as SEQ ID NO: 4 or 5, or a partially truncated version, said isolated polynucleotide further comprising a transcription termination codon at least six hundred nucleotides before the 3'-terminus or otherwise positioned such that translation of the polynucleotide gives rise to an extracellular domain optionally fused to a truncated portion of a full-length ActRIIa. A preferred nucleic acid sequence is SEQ ID NO:14. Nucleic acids disclosed herein may be operably linked to a promoter for expression, and the disclosure provides cells transformed with such recombinant polynucleotides. Preferably the cell is a mammalian cell such as a CHO cell.
[0017] In certain aspects, the disclosure provides methods for making a soluble, activin-binding ActRIIa polypeptide. Such a method may include expressing any of the nucleic acids (e.g., SEQ ID NO: 4, 5 or 14) disclosed herein in a suitable cell, such as a Chinese hamster ovary (CHO) cell. Such a method may comprise: a) culturing a cell under conditions suitable for expression of the soluble ActRIIa polypeptide, wherein said cell is transformed with a soluble ActRIIa expression construct; and b) recovering the soluble ActRIIa polypeptide so expressed. Soluble ActRIIa polypeptides may be recovered as crude, partially purified or highly purified fractions. Purification may be achieved by a series of purification steps, including, for example, one, two or three or more of the following, in any order: protein A chromatography, anion exchange chromatography (e.g., Q sepharose), hydrophobic interaction chromatography (e.g., phenylsepharose), size exclusion chromatography, and cation exchange chromatography.
[0018] In certain aspects, an activin-ActRIIa antagonist disclosed herein, such as a soluble, activin-binding ActRIIa polypeptide, may be used in a method for promoting bone growth or increasing bone density in a subject. In certain embodiments, the disclosure provides methods for treating a disorder associated with low bone density, or to promote bone growth, in patients in need thereof. A method may comprise administering to a subject in need thereof an effective amount of activin-ActRIIa antagonist. In certain aspects, the disclosure provides uses of activin-ActRIIa antagonist for making a medicament for the treatment of a disorder or condition as described herein.
[0019] In certain aspects, the disclosure provides a method for identifying an agent that stimulates growth of, or increased mineralization of, bone. The method comprises: a) identifying a test agent that binds to activin or a ligand-binding domain of an ActRIIa polypeptide; and b) evaluating the effect of the agent on growth of, or mineralization of, bone.
[0020] In certain aspects, the disclosure provides methods for promoting bone growth, increasing bone density or increasing bone strength, by administering to a subject an effective amount of an anti-activin A antibody. Also provided are methods for treating or preventing a bone-related disorder by administering to a subject an effective amount of an anti-activin A antibody. Methods for promoting bone growth and inhibiting bone resorption in a patient, by administering to the patient an effective amount of an anti-activin A antibody are also disclosed.
BRIEF DESCRIPTION OF THE DRAWINGS
[0021] The patent or application file contains at least one drawing executed in color. Copies of this patent or patent application publication with color drawing(s) will be provided by the Office upon request and payment of the necessary fee.
[0022] FIG. 1 shows the purification of ActRIIa-hFc expressed in CHO cells. The protein purifies as a single, well-defined peak.
[0023] FIG. 2 shows the binding of ActRIIa-hFc to activin and GDF-11, as measured by BiaCore.TM. assay.
[0024] FIG. 3 shows a schematic for the A-204 Reporter Gene Assay. The figure shows the Reporter vector: pGL3(CAGA)12 (described in Dennler et al, 1998, EMBO 17: 3091-3100.) The CAGA12 motif is present in TGF-Beta responsive genes (PAI-1 gene), so this vector is of general use for factors signaling through Smad 2 and 3. (AGCCAGACA) 12 Repeats shown in figure are SEQ ID NO: 18.
[0025] FIG. 4 shows the effects of ActRIIa-hFc (diamonds) and ActRIIa-mFc (squares) on GDF-8 signaling in the A-204 Reporter Gene Assay. Both proteins exhibited substantial inhibition of GDF-8 mediated signaling at picomolar concentrations.
[0026] FIG. 5 shows the effects of three different preparations of ActRIIa-hFc on GDF-11 signaling in the A-204 Reporter Gene Assay.
[0027] FIG. 6 shows examples of DEXA images of control- and ActRIIa-mFc-treated BALB/c mice, before (top panels) and after (bottom panels) the 12-week treatment period. Paler shading indicates increased bone density.
[0028] FIG. 7 shows a quantification of the effects of ActRIIa-mFc on bone mineral density in BALB/c mice over the 12-week period. Treatments were control (diamonds), 2 mg/kg dosing of ActRIIa-mFc (squares), 6 mg/kg dosing of ActRIIa-mFc (triangles) and 10 mg/kg dosing of ActRIIa-mFc (circles).
[0029] FIG. 8 shows a quantification of the effects of ActRIIa-mFc on bone mineral content in BALB/c mice over the 12-week period. Treatments were control (diamonds), 2 mg/kg dosing of ActRIIa-mFc (squares), 6 mg/kg dosing of ActRIIa-mFc (triangles) and 10 mg/kg dosing of ActRIIa-mFc (circles).
[0030] FIG. 9 shows a quantification of the effects of ActRIIa-mFc on bone mineral density of the trabecular bone in ovariectomized (OVX) or sham operated (SHAM) C57BL6 mice over after a 6-week period. Treatments were control (PBS) or 10 mg/kg dosing of ActRIIa-mFc (ActRIIa).
[0031] FIG. 10 shows a quantification of the effects of ActRIIa-mFc on the trabecular bone in ovariectomized (OVX) C57BL6 mice over a 12-week period. Treatments were control (PBS; pale bars) or 10 mg/kg dosing of ActRIIa-mFc (ActRIIa; dark bars).
[0032] FIG. 11 shows a quantification of the effects of ActRIIa-mFc on the trabecular bone in sham operated C57BL6 mice after 6 or 12 weeks of treatment period. Treatments were control (PBS; pale bars) or 10 mg/kg dosing of ActRIIa-mFc (ActRIIa; dark bars).
[0033] FIG. 12 shows the results of pQCT analysis of bone density in ovariectomized mice over 12 weeks of treatment. Treatments were control (PBS; pale bars) or ActRIIa-mFc (dark bars). y-axis: mg/ccm
[0034] FIG. 13 depicts the results of pQCT analysis of bone density in sham operated mice over 12 weeks of treatment. Treatments were control (PBS; pale bars) or ActRIIa-mFc (dark bars). y-axis; mg/ccm
[0035] FIGS. 14A and 14B show whole body DEXA analysis after 12 weeks of treatment (A) and ex vivo analysis of femurs (B). Light areas depict areas of high bone density.
[0036] FIG. 15 shows ex vivo pQCT analysis of the femoral midshaft after twelve weeks of treatment. Treatments were vehicle control (PBS, dark bars) and ActRIIa-mFc (pale bars). The four bars to the left show total bone density while the four bars to the right show cortical bone density. The first pair of bars in each set of four bars represent data from ovariectomized mice while the second pair of bars represent data from sham operated mice.
[0037] FIG. 16 shows ex vivo pQCT analysis and diaphyseal bone content of the femoral midshaft after twelve weeks of treatment. Treatments were vehicle control (PBS, dark bars) or ActRIIa-mFc (pale bars). The four bars to the left show total bone content while the four bars to the right show cortical bone content. The first pair of bars in each set of four bars represent data from ovariectomized mice while the second pair of bars represent data from sham operated mice.
[0038] FIG. 17 shows ex vivo pQCT analysis of the femoral midshaft and femoral cortical thickness. Treatments were control (PBS, dark bars) and ActRIIa-mFc (pale bars). The four bars to the left show endosteal circumference while the four bars to the right show periosteal circumference. The first pair of bars in each set of four bars represent data from ovariectomized mice while the second pair of bars represent data from sham operated mice.
[0039] FIG. 18 depicts the results of mechanical testing of femurs after twelve weeks of treatment. Treatments were control (PBS, dark bars) and ActRIIa-mFc (pale bars). The two bars to the left represent data from ovariectomized mice while the last two bars represent data from sham operated mice.
[0040] FIG. 19 shows the effects of ActrIIa-mFc on trabecular bone volume.
[0041] FIG. 20 shows the effects of ActrIIa-mFc on trabecular architecture in the distal femur.
[0042] FIG. 21 shows the effects of ActrIIa-mFc on cortical bone.
[0043] FIG. 22 shows the effects of ActrIIa-mFc on the mechanical strength of bone.
[0044] FIG. 23 shows the effects of different doses of ActRIIa-mFc on bone characteristics at three different dosages.
[0045] FIG. 24 shows bone histomorphometry indicating that ActRIIa-mFc has dual anabolic and anti-resorptive activity.
[0046] FIG. 25 shows that specific blocking of activin A signaling with an anti-Activin A antibody promotes osteoblast differentiation.
[0047] FIG. 26 shows that specific blocking of activin A signaling with an anti-Activin A antibody inhibits osteoclast differentiation.
[0048] FIG. 27 shows that blockade of activin B signaling with ActRIIa-mFc promotes osteoblast differentiation. Data are means.+-.SD. **, P<0.01.
[0049] FIG. 28 shows that blockade of activin B signaling with ActRIIa-mFc inhibits osteoclast differentiation, whereas anti-Activin A antibody is unable to block the effect of activin B. Data are means.+-.SD. **, P<0.01; NS, not significant.
DETAILED DESCRIPTION OF THE INVENTION
1. Overview
[0050] The transforming growth factor-beta (TGF-beta) superfamily contains a variety of growth factors that share common sequence elements and structural motifs. These proteins are known to exert biological effects on a large variety of cell types in both vertebrates and invertebrates. Members of the superfamily perform important functions during embryonic development in pattern formation and tissue specification and can influence a variety of differentiation processes, including adipogenesis, myogenesis, chondrogenesis, cardiogenesis, hematopoiesis, neurogenesis, and epithelial cell differentiation. The family is divided into two general branches: the BMP/GDF and the TGF-beta/Activin/BMP10 branches, whose members have diverse, often complementary effects. By manipulating the activity of a member of the TGF-beta family, it is often possible to cause significant physiological changes in an organism. For example, the Piedmontese and Belgian Blue cattle breeds carry a loss-of-function mutation in the GDF8 (also called myostatin) gene that causes a marked increase in muscle mass. Grobet et al., Nat Genet. 1997, 17(1):71-4. Furthermore, in humans, inactive alleles of GDF8 are associated with increased muscle mass and, reportedly, exceptional strength. Schuelke et al., N Engl J Med 2004, 350:2682-8.
[0051] Activins are dimeric polypeptide growth factors that belong to the TGF-beta superfamily. There are three principle activin forms (A, B, and AB) that are homo/heterodimers of two closely related .beta. subunits (.beta..sub.A.beta..sub.A, .beta..sub.B.beta..sub.B, and .beta..sub.A.beta..sub.B). The human genome also encodes an activin C and an activin E, which are primarily expressed in the liver. In the TGF-beta superfamily, activins are unique and multifunctional factors that can stimulate hormone production in ovarian and placental cells, support neuronal cell survival, influence cell-cycle progress positively or negatively depending on cell type, and induce mesodermal differentiation at least in amphibian embryos (DePaolo et al., 1991, Proc Soc Ep Biol Med. 198:500-512; Dyson et al., 1997, Curr Biol. 7:81-84; Woodruff, 1998, Biochem Pharmacol. 55:953-963). Moreover, erythroid differentiation factor (EDF) isolated from the stimulated human monocytic leukemic cells was found to be identical to activin A (Murata et al., 1988, PNAS, 85:2434). It has been suggested that activin A acts as a natural, positive regulator of erythropoiesis in the bone marrow. In several tissues, activin signaling is antagonized by its related heterodimer, inhibin. For example, during the release of follicle-stimulating hormone (FSH) from the pituitary, activin promotes FSH secretion and synthesis, while inhibin prevents FSH secretion and synthesis. Other proteins that may regulate activin bioactivity and/or bind to activin include follistatin (FS), follistatin-related protein (FSRP), .alpha..sub.2-macroglobulin, Cerberus, and endoglin.
[0052] TGF-.beta. signals are mediated by heteromeric complexes of type I and type II serine/threonine kinase receptors, which phosphorylate and activate downstream Smad proteins upon ligand stimulation (Massague, 2000, Nat. Rev. Mol. Cell Biol. 1:169-178). These type I and type II receptors are transmembrane proteins, composed of a ligand-binding extracellular domain with cysteine-rich region, a transmembrane domain, and a cytoplasmic domain with predicted serine/threonine specificity. Type I receptors are essential for signaling; and type II receptors are required for binding ligands and for expression of type I receptors. Type I and II activin receptors form a stable complex after ligand binding, resulting in phosphorylation of type I receptors by type II receptors.
[0053] Two related type II receptors, ActRIIa and ActRIIb, have been identified as the type II receptors for activins (Mathews and Vale, 1991, Cell 65:973-982; Attisano et al., 1992, Cell 68: 97-108). Besides activins, ActRIIa and ActRIIb can biochemically interact with several other TGF-.beta. family proteins, including BMP7, Nodal, GDF8, and GDF11 (Yamashita et al., 1995, J. Cell Biol. 130:217-226; Lee and McPherron, 2001, Proc. Natl. Acad. Sci. 98:9306-9311; Yeo and Whitman, 2001, Mol. Cell 7: 949-957; Oh et al., 2002, Genes Dev. 16:2749-54). ALK4 is the primary type I receptor for activins, particularly for activin A, and ALK-7 may serve as a receptor for activins as well, particularly for activin B.
[0054] As demonstrated herein, a soluble ActRIIa polypeptide (sActRIIa), which shows substantial preference in binding to activin A as opposed to other TGF-beta family members, such as GDF8 or GDF11, is effective to promote bone growth and increase bone density in vivo. While not wishing to be bound to any particular mechanism, it is expected that the effect of sActRIIa is caused primarily by an activin antagonist effect, given the very strong activin binding (picomolar dissociation constant) exhibited by the particular sActRIIa construct used in these studies. Regardless of mechanism, it is apparent from the data presented herein that ActRIIa-activin antagonists do increase bone density in normal mice and in mouse models for osteoporosis. It should be noted that bone is a dynamic tissue, with growth or shrinkage and increased or decreased density depending on a balance of factors that produce bone and stimulate mineralization (primarily osteoblasts) and factors that destroy and demineralize bone (primarily osteoclasts). Bone growth and mineralization may be increased by increasing the productive factors, by decreasing the destructive factors, or both. The terms "promote bone growth" and "increase bone mineralization" refer to the observable physical changes in bone and are intended to be neutral as to the mechanism by which changes in bone occur.
[0055] The mouse models for osteoporosis and bone growth/density that were used in the studies described herein are considered to be highly predictive of efficacy in humans, and therefore, this disclosure provides methods for using ActRIIa polypeptides and other activin-ActRIIa antagonists to promote bone growth and increase bone density in humans. Activin-ActRIIa antagonists include, for example, activin-binding soluble ActRIIa polypeptides, antibodies that bind to activin (particularly the activin A or B subunits, also referred to as .beta.A or .beta.B) and disrupt ActRIIa binding, antibodies that bind to ActRIIa and disrupt activin binding, non-antibody proteins selected for activin or ActRIIa binding (see e.g., WO/2002/088171, WO/2006/055689, WO/2002/032925, WO/2005/037989, US 2003/0133939, and US 2005/0238646 for examples of such proteins and methods for design and selection of same), randomized peptides selected for activin or ActRIIa binding, often affixed to an Fc domain. Two different proteins (or other moieties) with activin or ActRIIa binding activity, especially activin binders that block the type I (e.g., a soluble type I activin receptor) and type II (e.g., a soluble type II activin receptor) binding sites, respectively, may be linked together to create a bifunctional binding molecule. Nucleic acid aptamers, small molecules and other agents that inhibit the activin-ActRIIa signaling axis may also be used. Various proteins have activin-ActRIIa antagonist activity, including inhibin (i.e., inhibin alpha subunit), although inhibin does not universally antagonize activin in all tissues, follistatin (e.g., follistatin-288 and follistatin-315), Cerberus, FSRP, endoglin, activin C, alpha(2)-macroglobulin, and an M108A (methionine to alanine change at position 108) mutant activin A. Generally, alternative forms of activin, particularly those with alterations in the type I receptor binding domain can bind to type II receptors and fail to form an active ternary complex, thus acting as antagonists. Additionally, nucleic acids, such as antisense molecules, siRNAs or ribozymes that inhibit activin A, B, C or E, or, particularly, ActRIIa expression, can be used as activin-ActRIIa antagonists. Preferably, the activin-ActRIIa antagonist to be used will exhibit selectivity for inhibiting activin-mediated signaling versus other members of the TGF-beta family, and particularly with respect to GDF8 and GDF11. Soluble ActRIIb proteins do bind to activin, however, the wild type protein does not exhibit significant selectivity in binding to activin versus GDF8/11, and preliminary experiments suggest that this protein does not provide the desired effects on bone, while also causing substantial muscle growth. However, altered forms of ActRIIb with different binding properties have been identified (see, e.g., WO 2006/012627, pp. 55-59, incorporated herein by reference) and these proteins may achieve the desired effects on bone. Native or altered ActRIIb may be given added specificity for activin by coupling with a second, activin-selective binding agent.
[0056] The terms used in this specification generally have their ordinary meanings in the art, within the context of this invention and in the specific context where each term is used. Certain terms are discussed below or elsewhere in the specification, to provide additional guidance to the practitioner in describing the compositions and methods of the invention and how to make and use them. The scope or meaning of any use of a term will be apparent from the specific context in which the term is used.
[0057] "About" and "approximately" shall generally mean an acceptable degree of error for the quantity measured given the nature or precision of the measurements. Typically, exemplary degrees of error are within 20 percent (%), preferably within 10%, and more preferably within 5% of a given value or range of values.
[0058] Alternatively, and particularly in biological systems, the terms "about" and "approximately" may mean values that are within an order of magnitude, preferably within 5-fold and more preferably within 2-fold of a given value. Numerical quantities given herein are approximate unless stated otherwise, meaning that the term "about" or "approximately" can be inferred when not expressly stated.
[0059] The methods of the invention may include steps of comparing sequences to each other, including wild-type sequence to one or more mutants (sequence variants). Such comparisons typically comprise alignments of polymer sequences, e.g., using sequence alignment programs and/or algorithms that are well known in the art (for example, BLAST, FASTA and MEGALIGN, to name a few). The skilled artisan can readily appreciate that, in such alignments, where a mutation contains a residue insertion or deletion, the sequence alignment will introduce a "gap" (typically represented by a dash, or "A") in the polymer sequence not containing the inserted or deleted residue.
[0060] "Homologous," in all its grammatical forms and spelling variations, refers to the relationship between two proteins that possess a "common evolutionary origin," including proteins from superfamilies in the same species of organism, as well as homologous proteins from different species of organism. Such proteins (and their encoding nucleic acids) have sequence homology, as reflected by their sequence similarity, whether in terms of percent identity or by the presence of specific residues or motifs and conserved positions.
[0061] The term "sequence similarity," in all its grammatical forms, refers to the degree of identity or correspondence between nucleic acid or amino acid sequences that may or may not share a common evolutionary origin.
[0062] However, in common usage and in the instant application, the term "homologous," when modified with an adverb such as "highly," may refer to sequence similarity and may or may not relate to a common evolutionary origin.
2. ActRIIa Polypeptides
[0063] In certain aspects, the present invention relates to ActRIIa polypeptides. As used herein, the term "ActRIIa" refers to a family of activin receptor type IIa (ActRIIa) proteins from any species and variants derived from such ActRIIa proteins by mutagenesis or other modification. Reference to ActRIIa herein is understood to be a reference to any one of the currently identified forms. Members of the ActRIIa family are generally transmembrane proteins, composed of a ligand-binding extracellular domain with a cysteine-rich region, a transmembrane domain, and a cytoplasmic domain with predicted serine/threonine kinase activity.
[0064] The term "ActRIIa polypeptide" includes polypeptides comprising any naturally occurring polypeptide of an ActRIIa family member as well as any variants thereof (including mutants, fragments, fusions, and peptidomimetic forms) that retain a useful activity. For example, ActRIIa polypeptides include polypeptides derived from the sequence of any known ActRIIa having a sequence at least about 80% identical to the sequence of an ActRIIa polypeptide, and preferably at least 85%, 90%, 95%, 97%, 99% or greater identity. For example, an ActRIIa polypeptide of the invention may bind to and inhibit the function of an ActRIIa protein and/or activin. Preferably, an ActRIIa polypeptide promotes bone growth and bone mineralization. Examples of ActRIIa polypeptides include human ActRIIa precursor polypeptide (SEQ ID NO: 1) and soluble human ActRIIa polypeptides (e.g., SEQ ID NOs: 2, 3, 7 and 12).
[0065] The human ActRIIa precursor protein sequence is as follows:
TABLE-US-00001 (SEQ ID NO: 1) MGAAAKLAFAVFLISCSSGAILGRSETQECLFFNANWEKDRTNQTGVEPC YGDKDKRRHCFATWKNISGSIEIVKQGCWLDDINCYDRTDCVEKKDSPEV YFCCCEGNMCNEKFSYFPEMEVTQPTSNPVTPKPPYYNILLYSLVPLMLI AGIVICAFWVYRHHKMAYPPVLVPTQDPGPPPPSPLLGLKPLQLLEVKAR GRFGCVWKAQLLNEYVAVKIFPIQDKQSWQNEYEVYSLPGMKHENILQFI GAEKRGTSVDVDLWLITAFHEKGSLSDFLKANVVSWNELCHIAETMARGL AYLHEDIPGLKDGHKPAISHRDIKSKNVLLKNNLTACIADFGLALKFEAG KSAGDTHGQVGTRRYMAPEVLEGAINFQRDAFLRIDMYAMGLVLWELASR CTAADGPVDEYMLPFEEEIGQHPSLEDMQEVVVHKKKRPVLRDYWQKHAG MAMLCETIEECWDHDAEARLSAGCVGERITQMQRLTNIITTEDIVTVVTM VTNVDFPPKESSL
[0066] The signal peptide is single underlined; the extracellular domain is in bold and the potential N-linked glycosylation sites are double underlined.
[0067] The human ActRIIa soluble (extracellular), processed polypeptide sequence is as follows:
TABLE-US-00002 (SEQ ID NO: 2) ILGRSETQECLFFNANWEKDRTNQTGVEPCYGDKDKRRHCFATWKNISGS IEIVKQGCWLDDINCYDRTDCVEKKDSPEVYFCCCEGNMCNEKFSYFPEM EVTQPTSNPVTPKPP
[0068] The C-terminal "tail" of the extracellular domain is underlined. The sequence with the "tail" deleted (a .DELTA.15 sequence) is as follows:
TABLE-US-00003 (SEQ ID NO: 3) ILGRSETQECLFFNANWEKDRTNQTGVEPCYGDKDKRRHCFATWKNISGS IEIVKQGCWLDDINCYDRTDCVEKKDSPEVYFCCCEGNMCNEKFSYFPEM
[0069] The nucleic acid sequence encoding human ActRIIa precursor protein is as follows (nucleotides 164-1705 of Genbank entry NM_001616):
TABLE-US-00004 (SEQ ID NO: 4) ATGGGAGCTGCTGCAAAGTTGGCGTTTGCCGTCTTTCTTATCTCCTGTTC TTCAGGTGCTATACTTGGTAGATCAGAAACTCAGGAGTGTCTTTTCTTTA ATGCTAATTGGGAAAAAGACAGAACCAATCAAACTGGTGTTGAACCGTGT TATGGTGACAAAGATAAACGGCGGCATTGTTTTGCTACCTGGAAGAATAT TTCTGGTTCCATTGAAATAGTGAAACAAGGTTGTTGGCTGGATGATATCA ACTGCTATGACAGGACTGATTGTGTAGAAAAAAAAGACAGCCCTGAAGTA TATTTTTGTTGCTGTGAGGGCAATATGTGTAATGAAAAGTTTTCTTATTT TCCAGAGATGGAAGTCACACAGCCCACTTCAAATCCAGTTACACCTAAGC CACCCTATTACAACATCCTGCTCTATTCCTTGGTGCCACTTATGTTAATT GCGGGGATTGTCATTTGTGCATTTTGGGTGTACAGGCATCACAAGATGGC CTACCCTCCTGTACTTGTTCCAACTCAAGACCCAGGACCACCCCCACCTT CTCCATTACTAGGGTTGAAACCACTGCAGTTATTAGAAGTGAAAGCAAGG GGAAGATTTGGTTGTGTCTGGAAAGCCCAGTTGCTTAACGAATATGTGGC TGTCAAAATATTTCCAATACAGGACAAACAGTCATGGCAAAATGAATACG AAGTCTACAGTTTGCCTGGAATGAAGCATGAGAACATATTACAGTTCATT GGTGCAGAAAAACGAGGCACCAGTGTTGATGTGGATCTTTGGCTGATCAC AGCATTTCATGAAAAGGGTTCACTATCAGACTTTCTTAAGGCTAATGTGG TCTCTTGGAATGAACTGTGTCATATTGCAGAAACCATGGCTAGAGGATTG GCATATTTACATGAGGATATACCTGGCCTAAAAGATGGCCACAAACCTGC CATATCTCACAGGGACATCAAAAGTAAAAATGTGCTGTTGAAAAACAACC TGACAGCTTGCATTGCTGACTTTGGGTTGGCCTTAAAATTTGAGGCTGGC AAGTCTGCAGGCGATACCCATGGACAGGTTGGTACCCGGAGGTACATGGC TCCAGAGGTATTAGAGGGTGCTATAAACTTCCAAAGGGATGCATTTTTGA GGATAGATATGTATGCCATGGGATTAGTCCTATGGGAACTGGCTTCTCGC TGTACTGCTGCAGATGGACCTGTAGATGAATACATGTTGCCATTTGAGGA GGAAATTGGCCAGCATCCATCTCTTGAAGACATGCAGGAAGTTGTTGTGC ATAAAAAAAAGAGGCCTGTTTTAAGAGATTATTGGCAGAAACATGCTGGA ATGGCAATGCTCTGTGAAACCATTGAAGAATGTTGGGATCACGACGCAGA AGCCAGGTTATCAGCTGGATGTGTAGGTGAAAGAATTACCCAGATGCAGA GACTAACAAATATTATTACCACAGAGGACATTGTAACAGTGGTCACAATG GTGACAAATGTTGACTTTCCTCCCAAAGAATCTAGTCTATGA
[0070] The nucleic acid sequence encoding a human ActRIIa soluble (extracellular) polypeptide is as follows:
TABLE-US-00005 (SEQ ID NO: 5) ATACTTGGTAGATCAGAAACTCAGGAGTGTCTTTTCTTTAATGCTAATTG GGAAAAAGACAGAACCAATCAAACTGGTGTTGAACCGTGTTATGGTGACA AAGATAAACGGCGGCATTGTTTTGCTACCTGGAAGAATATTTCTGGTTCC ATTGAAATAGTGAAACAAGGTTGTTGGCTGGATGATATCAACTGCTATGA CAGGACTGATTGTGTAGAAAAAAAAGACAGCCCTGAAGTATATTTTTGTT GCTGTGAGGGCAATATGTGTAATGAAAAGTTTTCTTATTTTCCAGAGATG GAAGTCACACAGCCCACTTCAAATCCAGTTACACCTAAGCCACCC
[0071] In a specific embodiment, the invention relates to soluble ActRIIa polypeptides. As described herein, the term "soluble ActRIIa polypeptide" generally refers to polypeptides comprising an extracellular domain of an ActRIIa protein. The term "soluble ActRIIa polypeptide," as used herein, includes any naturally occurring extracellular domain of an ActRIIa protein as well as any variants thereof (including mutants, fragments and peptidomimetic forms). An activin-binding ActRIIa polypeptide is one that retains the ability to bind to activin, particularly activin AA, AB or BB. Preferably, an activin-binding ActRIIa polypeptide will bind to activin AA with a dissociation constant of 1 nM or less. Amino acid sequences of human ActRIIa precursor protein is provided below. The extracellular domain of an ActRIIa protein binds to activin and is generally soluble, and thus can be termed a soluble, activin-binding ActRIIa polypeptide. Examples of soluble, activin-binding ActRIIa polypeptides include the soluble polypeptide illustrated in SEQ ID NOs: 2, 3, 7, 12 and 13. SEQ ID NO:7 is referred to as ActRIIa-hFc, and is described further in the Examples. Other examples of soluble, activin-binding ActRIIa polypeptides comprise a signal sequence in addition to the extracellular domain of an ActRIIa protein, for example, the honey bee mellitin leader sequence (SEQ ID NO: 8), the tissue plaminogen activator (TPA) leader (SEQ ID NO: 9) or the native ActRIIa leader (SEQ ID NO: 10). The ActRIIa-hFc polypeptide illustrated in SEQ ID NO:13 uses a TPA leader.
[0072] Functionally active fragments of ActRIIa polypeptides can be obtained by screening polypeptides recombinantly produced from the corresponding fragment of the nucleic acid encoding an ActRIIa polypeptide. In addition, fragments can be chemically synthesized using techniques known in the art such as conventional Merrifield solid phase f-Moc or t-Boc chemistry. The fragments can be produced (recombinantly or by chemical synthesis) and tested to identify those peptidyl fragments that can function as antagonists (inhibitors) of ActRIIa protein or signaling mediated by activin.
[0073] Functionally active variants of ActRIIa polypeptides can be obtained by screening libraries of modified polypeptides recombinantly produced from the corresponding mutagenized nucleic acids encoding an ActRIIa polypeptide. The variants can be produced and tested to identify those that can function as antagonists (inhibitors) of ActRIIa protein or signaling mediated by activin. In certain embodiments, a functional variant of the ActRIIa polypeptides comprises an amino acid sequence that is at least 75% identical to an amino acid sequence selected from SEQ ID NOs: 2 or 3. In certain cases, the functional variant has an amino acid sequence at least 80%, 85%, 90%, 95%, 97%, 98%, 99% or 100% identical to an amino acid sequence selected from SEQ ID NOs: 2 or 3.
[0074] Functional variants may be generated by modifying the structure of an ActRIIa polypeptide for such purposes as enhancing therapeutic efficacy, or stability (e.g., ex vivo shelf life and resistance to proteolytic degradation in vivo). Such modified ActRIIa polypeptides when selected to retain activin binding, are considered functional equivalents of the naturally-occurring ActRIIa polypeptides. Modified ActRIIa polypeptides can also be produced, for instance, by amino acid substitution, deletion, or addition. For instance, it is reasonable to expect that an isolated replacement of a leucine with an isoleucine or valine, an aspartate with a glutamate, a threonine with a serine, or a similar replacement of an amino acid with a structurally related amino acid (e.g., conservative mutations) will not have a major effect on the biological activity of the resulting molecule. Conservative replacements are those that take place within a family of amino acids that are related in their side chains. Whether a change in the amino acid sequence of an ActRIIa polypeptide results in a functional homolog can be readily determined by assessing the ability of the variant ActRIIa polypeptide to produce a response in cells in a fashion similar to the wild-type ActRIIa polypeptide.
[0075] In certain embodiments, the present invention contemplates specific mutations of the ActRIIa polypeptides so as to alter the glycosylation of the polypeptide. Such mutations may be selected so as to introduce or eliminate one or more glycosylation sites, such as O-linked or N-linked glycosylation sites. Asparagine-linked glycosylation recognition sites generally comprise a tripeptide sequence, asparagine-X-threonine (or asparagines-X-serine) (where "X" is any amino acid) which is specifically recognized by appropriate cellular glycosylation enzymes. The alteration may also be made by the addition of, or substitution by, one or more serine or threonine residues to the sequence of the wild-type ActRIIa polypeptide (for O-linked glycosylation sites). A variety of amino acid substitutions or deletions at one or both of the first or third amino acid positions of a glycosylation recognition site (and/or amino acid deletion at the second position) results in non-glycosylation at the modified tripeptide sequence. Another means of increasing the number of carbohydrate moieties on an ActRIIa polypeptide is by chemical or enzymatic coupling of glycosides to the ActRIIa polypeptide. Depending on the coupling mode used, the sugar(s) may be attached to (a) arginine and histidine; (b) free carboxyl groups; (c) free sulfhydryl groups such as those of cysteine; (d) free hydroxyl groups such as those of serine, threonine, or hydroxyproline; (e) aromatic residues such as those of phenylalanine, tyrosine, or tryptophan; or (f) the amide group of glutamine. These methods are described in WO 87/05330 published Sep. 11, 1987, and in Aplin and Wriston (1981) CRC Crit. Rev. Biochem., pp. 259-306, incorporated by reference herein. Removal of one or more carbohydrate moieties present on an ActRIIa polypeptide may be accomplished chemically and/or enzymatically. Chemical deglycosylation may involve, for example, exposure of the ActRIIa polypeptide to the compound trifluoromethanesulfonic acid, or an equivalent compound. This treatment results in the cleavage of most or all sugars except the linking sugar (N-acetylglucosamine or N-acetylgalactosamine), while leaving the amino acid sequence intact. Chemical deglycosylation is further described by Hakimuddin et al. (1987) Arch. Biochem. Biophys. 259:52 and by Edge et al. (1981) Anal. Biochem. 118:131. Enzymatic cleavage of carbohydrate moieties on ActRIIa polypeptides can be achieved by the use of a variety of endo- and exo-glycosidases as described by Thotakura et al. (1987) Meth. Enzymol. 138:350. The sequence of an ActRIIa polypeptide may be adjusted, as appropriate, depending on the type of expression system used, as mammalian, yeast, insect and plant cells may all introduce differing glycosylation patterns that can be affected by the amino acid sequence of the peptide. In general, ActRIIa proteins for use in humans will be expressed in a mammalian cell line that provides proper glycosylation, such as HEK293 or CHO cell lines, although other mammalian expression cell lines, yeast cell lines with engineered glycosylation enzymes and insect cells are expected to be useful as well.
[0076] This disclosure further contemplates a method of generating mutants, particularly sets of combinatorial mutants of an ActRIIa polypeptide, as well as truncation mutants; pools of combinatorial mutants are especially useful for identifying functional variant sequences. The purpose of screening such combinatorial libraries may be to generate, for example, ActRIIa polypeptide variants which can act as either agonists or antagonist, or alternatively, which possess novel activities all together. A variety of screening assays are provided below, and such assays may be used to evaluate variants. For example, an ActRIIa polypeptide variant may be screened for ability to bind to an ActRIIa ligand, to prevent binding of an ActRIIa ligand to an ActRIIa polypeptide or to interfere with signaling caused by an ActRIIa ligand.
[0077] The activity of an ActRIIa polypeptide or its variants may also be tested in a cell-based or in vivo assay. For example, the effect of an ActRIIa polypeptide variant on the expression of genes involved in bone production or bone destruction may be assessed. This may, as needed, be performed in the presence of one or more recombinant ActRIIa ligand proteins (e.g., activin), and cells may be transfected so as to produce an ActRIIa polypeptide and/or variants thereof, and optionally, an ActRIIa ligand. Likewise, an ActRIIa polypeptide may be administered to a mouse or other animal, and one or more bone properties, such as density or volume may be assessed. The healing rate for bone fractures may also be evaluated. Dual-energy x-ray absorptiometry (DEXA) is a well-established, non-invasive, quantitative technique for assessing bone density in an animal. In humans central DEXA systems may be used to evaluate bone density in the spine and pelvis. These are the best predictors of overall bone density. Peripheral DEXA systems may be used to evaluate bone density in peripheral bones, including, for example, the bones of the hand, wrist, ankle and foot. Traditional x-ray imaging systems, including CAT scans, may be used to evaluate bone growth and fracture healing. The mechanical strength of bone may also be evaluated.
[0078] Combinatorially-derived variants can be generated which have a selective or generally increased potency relative to a naturally occurring ActRIIa polypeptide. Likewise, mutagenesis can give rise to variants which have intracellular half-lives dramatically different than the corresponding wild-type ActRIIa polypeptide. For example, the altered protein can be rendered either more stable or less stable to proteolytic degradation or other cellular processes which result in destruction of, or otherwise inactivation of a native ActRIIa polypeptide. Such variants, and the genes which encode them, can be utilized to alter ActRIIa polypeptide levels by modulating the half-life of the ActRIIa polypeptides. For instance, a short half-life can give rise to more transient biological effects and can allow tighter control of recombinant ActRIIa polypeptide levels within the patient. In an Fc fusion protein, mutations may be made in the linker (if any) and/or the Fc portion to alter the half-life of the protein.
[0079] A combinatorial library may be produced by way of a degenerate library of genes encoding a library of polypeptides which each include at least a portion of potential ActRIIa polypeptide sequences. For instance, a mixture of synthetic oligonucleotides can be enzymatically ligated into gene sequences such that the degenerate set of potential ActRIIa polypeptide nucleotide sequences are expressible as individual polypeptides, or alternatively, as a set of larger fusion proteins (e.g., for phage display).
[0080] There are many ways by which the library of potential homologs can be generated from a degenerate oligonucleotide sequence. Chemical synthesis of a degenerate gene sequence can be carried out in an automatic DNA synthesizer, and the synthetic genes then be ligated into an appropriate vector for expression. The synthesis of degenerate oligonucleotides is well known in the art (see for example, Narang, S A (1983) Tetrahedron 39:3; Itakura et al., (1981) Recombinant DNA, Proc. 3rd Cleveland Sympos. Macromolecules, ed. A G Walton, Amsterdam: Elsevier pp273-289; Itakura et al., (1984) Annu. Rev. Biochem. 53:323; Itakura et al., (1984) Science 198:1056; Ike et al., (1983) Nucleic Acid Res. 11:477). Such techniques have been employed in the directed evolution of other proteins (see, for example, Scott et al., (1990) Science 249:386-390; Roberts et al., (1992) PNAS USA 89:2429-2433; Devlin et al., (1990) Science 249: 404-406; Cwirla et al., (1990) PNAS USA 87: 6378-6382; as well as U.S. Pat. Nos. 5,223,409, 5,198,346, and 5,096,815).
[0081] Alternatively, other forms of mutagenesis can be utilized to generate a combinatorial library. For example, ActRIIa polypeptide variants can be generated and isolated from a library by screening using, for example, alanine scanning mutagenesis and the like (Ruf et al., (1994) Biochemistry 33:1565-1572; Wang et al., (1994) J. Biol. Chem. 269:3095-3099; Balint et al., (1993) Gene 137:109-118; Grodberg et al., (1993) Eur. J. Biochem. 218:597-601; Nagashima et al., (1993) J. Biol. Chem. 268:2888-2892; Lowman et al., (1991) Biochemistry 30:10832-10838; and Cunningham et al., (1989) Science 244:1081-1085), by linker scanning mutagenesis (Gustin et al., (1993) Virology 193:653-660; Brown et al., (1992) Mol. Cell Biol. 12:2644-2652; McKnight et al., (1982) Science 232:316); by saturation mutagenesis (Meyers et al., (1986) Science 232:613); by PCR mutagenesis (Leung et al., (1989) Method Cell Mol Biol 1:11-19); or by random mutagenesis, including chemical mutagenesis, etc. (Miller et al., (1992) A Short Course in Bacterial Genetics, CSHL Press, Cold Spring Harbor, N.Y.; and Greener et al., (1994) Strategies in Mol Biol 7:32-34). Linker scanning mutagenesis, particularly in a combinatorial setting, is an attractive method for identifying truncated (bioactive) forms of ActRIIa polypeptides.
[0082] A wide range of techniques are known in the art for screening gene products of combinatorial libraries made by point mutations and truncations, and, for that matter, for screening cDNA libraries for gene products having a certain property. Such techniques will be generally adaptable for rapid screening of the gene libraries generated by the combinatorial mutagenesis of ActRIIa polypeptides. The most widely used techniques for screening large gene libraries typically comprises cloning the gene library into replicable expression vectors, transforming appropriate cells with the resulting library of vectors, and expressing the combinatorial genes under conditions in which detection of a desired activity facilitates relatively easy isolation of the vector encoding the gene whose product was detected. Preferred assays include activin binding assays and activin-mediated cell signaling assays.
[0083] In certain embodiments, the ActRIIa polypeptides of the invention may further comprise post-translational modifications in addition to any that are naturally present in the ActRIIa polypeptides. Such modifications include, but are not limited to, acetylation, carboxylation, glycosylation, phosphorylation, lipidation, and acylation. As a result, the modified ActRIIa polypeptides may contain non-amino acid elements, such as polyethylene glycols, lipids, poly- or mono-saccharide, and phosphates. Effects of such non-amino acid elements on the functionality of a ActRIIa polypeptide may be tested as described herein for other ActRIIa polypeptide variants. When an ActRIIa polypeptide is produced in cells by cleaving a nascent form of the ActRIIa polypeptide, post-translational processing may also be important for correct folding and/or function of the protein. Different cells (such as CHO, HeLa, MDCK, 293, WI38, NIH-3T3 or HEK293) have specific cellular machinery and characteristic mechanisms for such post-translational activities and may be chosen to ensure the correct modification and processing of the ActRIIa polypeptides.
[0084] In certain aspects, functional variants or modified forms of the ActRIIa polypeptides include fusion proteins having at least a portion of the ActRIIa polypeptides and one or more fusion domains. Well known examples of such fusion domains include, but are not limited to, polyhistidine, Glu-Glu, glutathione S transferase (GST), thioredoxin, protein A, protein G, an immunoglobulin heavy chain constant region (Fc), maltose binding protein (MBP), or human serum albumin. A fusion domain may be selected so as to confer a desired property. For example, some fusion domains are particularly useful for isolation of the fusion proteins by affinity chromatography. For the purpose of affinity purification, relevant matrices for affinity chromatography, such as glutathione-, amylase-, and nickel- or cobalt-conjugated resins are used. Many of such matrices are available in "kit" form, such as the Pharmacia GST purification system and the QIAexpress.TM. system (Qiagen) useful with (HIS.sub.6 (SEQ ID NO: 17)) fusion partners. As another example, a fusion domain may be selected so as to facilitate detection of the ActRIIa polypeptides. Examples of such detection domains include the various fluorescent proteins (e.g., GFP) as well as "epitope tags," which are usually short peptide sequences for which a specific antibody is available. Well known epitope tags for which specific monoclonal antibodies are readily available include FLAG, influenza virus haemagglutinin (HA), and c-myc tags. In some cases, the fusion domains have a protease cleavage site, such as for Factor Xa or Thrombin, which allows the relevant protease to partially digest the fusion proteins and thereby liberate the recombinant proteins therefrom. The liberated proteins can then be isolated from the fusion domain by subsequent chromatographic separation. In certain preferred embodiments, an ActRIIa polypeptide is fused with a domain that stabilizes the ActRIIa polypeptide in vivo (a "stabilizer" domain). By "stabilizing" is meant anything that increases serum half life, regardless of whether this is because of decreased destruction, decreased clearance by the kidney, or other pharmacokinetic effect. Fusions with the Fc portion of an immunoglobulin are known to confer desirable pharmacokinetic properties on a wide range of proteins. Likewise, fusions to human serum albumin can confer desirable properties. Other types of fusion domains that may be selected include multimerizing (e.g., dimerizing, tetramerizing) domains and functional domains (that confer an additional biological function, such as further stimulation of bone growth or muscle growth, as desired).
[0085] As a specific example, the present invention provides a fusion protein comprising a soluble extracellular domain of ActRIIa fused to an Fc domain (e.g., SEQ ID NO: 6).
TABLE-US-00006 THTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVD(A)VSHE DPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEY KCK(A)VSNKALPVPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLT CLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGPFFLYSKLTVDKSR WQQGNVFSCSVMHEALHN(A)HYTQKSLSLSPGK*
[0086] Optionally, the Fc domain has one or more mutations at residues such as Asp-265, lysine 322, and Asn-434. In certain cases, the mutant Fc domain having one or more of these mutations (e.g., Asp-265 mutation) has reduced ability of binding to the Fc.gamma. receptor relative to a wildtype Fc domain. In other cases, the mutant Fc domain having one or more of these mutations (e.g., Asn-434 mutation) has increased ability of binding to the WIC class I-related Fc-receptor (FcRN) relative to a wildtype Fc domain.
[0087] It is understood that different elements of the fusion proteins may be arranged in any manner that is consistent with the desired functionality. For example, an ActRIIa polypeptide may be placed C-terminal to a heterologous domain, or, alternatively, a heterologous domain may be placed C-terminal to an ActRIIa polypeptide. The ActRIIa polypeptide domain and the heterologous domain need not be adjacent in a fusion protein, and additional domains or amino acid sequences may be included C- or N-terminal to either domain or between the domains.
[0088] In certain embodiments, the ActRIIa polypeptides of the present invention contain one or more modifications that are capable of stabilizing the ActRIIa polypeptides. For example, such modifications enhance the in vitro half life of the ActRIIa polypeptides, enhance circulatory half life of the ActRIIa polypeptides or reduce proteolytic degradation of the ActRIIa polypeptides. Such stabilizing modifications include, but are not limited to, fusion proteins (including, for example, fusion proteins comprising an ActRIIa polypeptide and a stabilizer domain), modifications of a glycosylation site (including, for example, addition of a glycosylation site to an ActRIIa polypeptide), and modifications of carbohydrate moiety (including, for example, removal of carbohydrate moieties from an ActRIIa polypeptide). In the case of fusion proteins, an ActRIIa polypeptide is fused to a stabilizer domain such as an IgG molecule (e.g., an Fc domain). As used herein, the term "stabilizer domain" not only refers to a fusion domain (e.g., Fc) as in the case of fusion proteins, but also includes nonproteinaceous modifications such as a carbohydrate moiety, or nonproteinaceous polymer, such as polyethylene glycol.
[0089] In certain embodiments, the present invention makes available isolated and/or purified forms of the ActRIIa polypeptides, which are isolated from, or otherwise substantially free of, other proteins. ActRIIa polypeptides will generally be produced by expression from recombinant nucleic acids.
3. Nucleic Acids Encoding ActRIIa Polypeptides
[0090] In certain aspects, the invention provides isolated and/or recombinant nucleic acids encoding any of the ActRIIa polypeptides (e.g., soluble ActRIIa polypeptides), including fragments, functional variants and fusion proteins disclosed herein. For example, SEQ ID NO: 4 encodes the naturally occurring human ActRIIa precursor polypeptide, while SEQ ID NO: 5 encodes the processed extracellular domain of ActRIIa. The subject nucleic acids may be single-stranded or double stranded. Such nucleic acids may be DNA or RNA molecules. These nucleic acids may be used, for example, in methods for making ActRIIa polypeptides or as direct therapeutic agents (e.g., in a gene therapy approach).
[0091] In certain aspects, the subject nucleic acids encoding ActRIIa polypeptides are further understood to include nucleic acids that are variants of SEQ ID NO: 4 or 5. Variant nucleotide sequences include sequences that differ by one or more nucleotide substitutions, additions or deletions, such as allelic variants.
[0092] In certain embodiments, the invention provides isolated or recombinant nucleic acid sequences that are at least 80%, 85%, 90%, 95%, 97%, 98%, 99% or 100% identical to SEQ ID NO: 4 or 5. One of ordinary skill in the art will appreciate that nucleic acid sequences complementary to SEQ ID NO: 4 or 5, and variants of SEQ ID NO: 4 or 5 are also within the scope of this invention. In further embodiments, the nucleic acid sequences of the invention can be isolated, recombinant, and/or fused with a heterologous nucleotide sequence, or in a DNA library.
[0093] In other embodiments, nucleic acids of the invention also include nucleotide sequences that hybridize under highly stringent conditions to the nucleotide sequence designated in SEQ ID NO: 4 or 5, complement sequence of SEQ ID NO: 4 or 5, or fragments thereof. As discussed above, one of ordinary skill in the art will understand readily that appropriate stringency conditions which promote DNA hybridization can be varied. One of ordinary skill in the art will understand readily that appropriate stringency conditions which promote DNA hybridization can be varied. For example, one could perform the hybridization at 6.0.times.sodium chloride/sodium citrate (SSC) at about 45.degree. C., followed by a wash of 2.0.times.SSC at 50.degree. C. For example, the salt concentration in the wash step can be selected from a low stringency of about 2.0.times.SSC at 50.degree. C. to a high stringency of about 0.2.times.SSC at 50.degree. C. In addition, the temperature in the wash step can be increased from low stringency conditions at room temperature, about 22.degree. C., to high stringency conditions at about 65.degree. C. Both temperature and salt may be varied, or temperature or salt concentration may be held constant while the other variable is changed. In one embodiment, the invention provides nucleic acids which hybridize under low stringency conditions of 6.times.SSC at room temperature followed by a wash at 2.times.SSC at room temperature.
[0094] Isolated nucleic acids which differ from the nucleic acids as set forth in SEQ ID NOs: 4 or 5 due to degeneracy in the genetic code are also within the scope of the invention. For example, a number of amino acids are designated by more than one triplet. Codons that specify the same amino acid, or synonyms (for example, CAU and CAC are synonyms for histidine) may result in "silent" mutations which do not affect the amino acid sequence of the protein. However, it is expected that DNA sequence polymorphisms that do lead to changes in the amino acid sequences of the subject proteins will exist among mammalian cells. One skilled in the art will appreciate that these variations in one or more nucleotides (up to about 3-5% of the nucleotides) of the nucleic acids encoding a particular protein may exist among individuals of a given species due to natural allelic variation. Any and all such nucleotide variations and resulting amino acid polymorphisms are within the scope of this invention.
[0095] In certain embodiments, the recombinant nucleic acids of the invention may be operably linked to one or more regulatory nucleotide sequences in an expression construct. Regulatory nucleotide sequences will generally be appropriate to the host cell used for expression. Numerous types of appropriate expression vectors and suitable regulatory sequences are known in the art for a variety of host cells. Typically, said one or more regulatory nucleotide sequences may include, but are not limited to, promoter sequences, leader or signal sequences, ribosomal binding sites, transcriptional start and termination sequences, translational start and termination sequences, and enhancer or activator sequences. Constitutive or inducible promoters as known in the art are contemplated by the invention. The promoters may be either naturally occurring promoters, or hybrid promoters that combine elements of more than one promoter. An expression construct may be present in a cell on an episome, such as a plasmid, or the expression construct may be inserted in a chromosome. In a preferred embodiment, the expression vector contains a selectable marker gene to allow the selection of transformed host cells. Selectable marker genes are well known in the art and will vary with the host cell used.
[0096] In certain aspects of the invention, the subject nucleic acid is provided in an expression vector comprising a nucleotide sequence encoding an ActRIIa polypeptide and operably linked to at least one regulatory sequence. Regulatory sequences are art-recognized and are selected to direct expression of the ActRIIa polypeptide. Accordingly, the term regulatory sequence includes promoters, enhancers, and other expression control elements. Exemplary regulatory sequences are described in Goeddel; Gene Expression Technology: Methods in Enzymology, Academic Press, San Diego, Calif. (1990). For instance, any of a wide variety of expression control sequences that control the expression of a DNA sequence when operatively linked to it may be used in these vectors to express DNA sequences encoding an ActRIIa polypeptide. Such useful expression control sequences, include, for example, the early and late promoters of SV40, tet promoter, adenovirus or cytomegalovirus immediate early promoter, RSV promoters, the lac system, the trp system, the TAC or TRC system, T7 promoter whose expression is directed by T7 RNA polymerase, the major operator and promoter regions of phage lambda, the control regions for fd coat protein, the promoter for 3-phosphoglycerate kinase or other glycolytic enzymes, the promoters of acid phosphatase, e.g., Pho5, the promoters of the yeast .alpha.-mating factors, the polyhedron promoter of the baculovirus system and other sequences known to control the expression of genes of prokaryotic or eukaryotic cells or their viruses, and various combinations thereof. It should be understood that the design of the expression vector may depend on such factors as the choice of the host cell to be transformed and/or the type of protein desired to be expressed. Moreover, the vector's copy number, the ability to control that copy number and the expression of any other protein encoded by the vector, such as antibiotic markers, should also be considered.
[0097] A recombinant nucleic acid of the invention can be produced by ligating the cloned gene, or a portion thereof, into a vector suitable for expression in either prokaryotic cells, eukaryotic cells (yeast, avian, insect or mammalian), or both. Expression vehicles for production of a recombinant ActRIIa polypeptide include plasmids and other vectors. For instance, suitable vectors include plasmids of the types: pBR322-derived plasmids, pEMBL-derived plasmids, pEX-derived plasmids, pBTac-derived plasmids and pUC-derived plasmids for expression in prokaryotic cells, such as E. coli.
[0098] Some mammalian expression vectors contain both prokaryotic sequences to facilitate the propagation of the vector in bacteria, and one or more eukaryotic transcription units that are expressed in eukaryotic cells. The pcDNAI/amp, pcDNAI/neo, pRc/CMV, pSV2gpt, pSV2neo, pSV2-dhfr, pTk2, pRSVneo, pMSG, pSVT7, pko-neo and pHyg derived vectors are examples of mammalian expression vectors suitable for transfection of eukaryotic cells. Some of these vectors are modified with sequences from bacterial plasmids, such as pBR322, to facilitate replication and drug resistance selection in both prokaryotic and eukaryotic cells. Alternatively, derivatives of viruses such as the bovine papilloma virus (BPV-1), or Epstein-Barr virus (pHEBo, pREP-derived and p205) can be used for transient expression of proteins in eukaryotic cells. Examples of other viral (including retroviral) expression systems can be found below in the description of gene therapy delivery systems. The various methods employed in the preparation of the plasmids and in transformation of host organisms are well known in the art. For other suitable expression systems for both prokaryotic and eukaryotic cells, as well as general recombinant procedures, see Molecular Cloning A Laboratory Manual, 3rd Ed., ed. by Sambrook, Fritsch and Maniatis (Cold Spring Harbor Laboratory Press, 2001). In some instances, it may be desirable to express the recombinant polypeptides by the use of a baculovirus expression system. Examples of such baculovirus expression systems include pVL-derived vectors (such as pVL1392, pVL1393 and pVL941), pAcUW-derived vectors (such as pAcUW1), and pBlueBac-derived vectors (such as the B-gal containing pBlueBac III).
[0099] In a preferred embodiment, a vector will be designed for production of the subject ActRIIa polypeptides in CHO cells, such as a Pcmv-Script vector (Stratagene, La Jolla, Calif.), pcDNA4 vectors (Invitrogen, Carlsbad, Calif.) and pCI-neo vectors (Promega, Madison, Wis.). As will be apparent, the subject gene constructs can be used to cause expression of the subject ActRIIa polypeptides in cells propagated in culture, e.g., to produce proteins, including fusion proteins or variant proteins, for purification.
[0100] This disclosure also pertains to a host cell transfected with a recombinant gene including a coding sequence (e.g., SEQ ID NO: 4 or 5) for one or more of the subject ActRIIa polypeptides. The host cell may be any prokaryotic or eukaryotic cell. For example, an ActRIIa polypeptide of the invention may be expressed in bacterial cells such as E. coli, insect cells (e.g., using a baculovirus expression system), yeast, or mammalian cells. Other suitable host cells are known to those skilled in the art.
[0101] Accordingly, the present invention further pertains to methods of producing the subject ActRIIa polypeptides. For example, a host cell transfected with an expression vector encoding an ActRIIa polypeptide can be cultured under appropriate conditions to allow expression of the ActRIIa polypeptide to occur. The ActRIIa polypeptide may be secreted and isolated from a mixture of cells and medium containing the ActRIIa polypeptide. Alternatively, the ActRIIa polypeptide may be retained cytoplasmically or in a membrane fraction and the cells harvested, lysed and the protein isolated. A cell culture includes host cells, media and other byproducts. Suitable media for cell culture are well known in the art. The subject ActRIIa polypeptides can be isolated from cell culture medium, host cells, or both, using techniques known in the art for purifying proteins, including ion-exchange chromatography, gel filtration chromatography, ultrafiltration, electrophoresis, immunoaffinity purification with antibodies specific for particular epitopes of the ActRIIa polypeptides and affinity purification with an agent that binds to a domain fused to the ActRIIa polypeptide (e.g., a protein A column may be used to purify an ActRIIa-Fc fusion). In a preferred embodiment, the ActRIIa polypeptide is a fusion protein containing a domain which facilitates its purification. In a preferred embodiment, purification is achieved by a series of column chromatography steps, including, for example, three or more of the following, in any order: protein A chromatography, Q sepharose chromatography, phenylsepharose chromatography, size exclusion chromatography, and cation exchange chromatography. The purification could be completed with viral filtration and buffer exchange. As demonstrated herein, ActRIIa-hFc protein was purified to a purity of >98% as determined by size exclusion chromatography and >95% as determined by SDS PAGE. This level of purity was sufficient to achieve desirable effects on bone in mice and an acceptable safety profile in mice, rats and non-human primates.
[0102] In another embodiment, a fusion gene coding for a purification leader sequence, such as a poly-(His)/enterokinase cleavage site sequence at the N-terminus of the desired portion of the recombinant ActRIIa polypeptide, can allow purification of the expressed fusion protein by affinity chromatography using a Ni.sup.2+ metal resin. The purification leader sequence can then be subsequently removed by treatment with enterokinase to provide the purified ActRIIa polypeptide (e.g., see Hochuli et al., (1987) J. Chromatography 411:177; and Janknecht et al., PNAS USA 88:8972).
[0103] Techniques for making fusion genes are well known. Essentially, the joining of various DNA fragments coding for different polypeptide sequences is performed in accordance with conventional techniques, employing blunt-ended or stagger-ended termini for ligation, restriction enzyme digestion to provide for appropriate termini, filling-in of cohesive ends as appropriate, alkaline phosphatase treatment to avoid undesirable joining, and enzymatic ligation. In another embodiment, the fusion gene can be synthesized by conventional techniques including automated DNA synthesizers. Alternatively, PCR amplification of gene fragments can be carried out using anchor primers which give rise to complementary overhangs between two consecutive gene fragments which can subsequently be annealed to generate a chimeric gene sequence (see, for example, Current Protocols in Molecular Biology, eds. Ausubel et al., John Wiley & Sons: 1992).
4. Alternative Activin and ActRIIa Antagonists
[0104] The data presented herein demonstrates that antagonists of activin-ActRIIa signaling can be used to promote bone growth and bone mineralization. Although soluble ActRIIa polypeptides, and particularly ActrIIa-Fc, are preferred antagonists, and although such antagonists may affect bone through a mechanism other than activin antagonism (e.g., activin inhibition may be an indicator of the tendency of an agent to inhibit the activities of a spectrum of molecules, including, perhaps, other members of the TGF-beta superfamily, and such collective inhibition may lead to the desired effect on bone), other types of activin-ActRIIa antagonists are expected to be useful, including anti-activin (e.g., A, B, C or E) antibodies, anti-ActRIIa antibodies, antisense, RNAi or ribozyme nucleic acids that inhibit the production of ActRIIa and other inhibitors of activin or ActRIIa, particularly those that disrupt activin-ActRIIa binding.
[0105] An antibody that is specifically reactive with an ActRIIa polypeptide (e.g., a soluble ActRIIa polypeptide) and which either binds competitively to ligand with the ActRIIa polypeptide or otherwise inhibits ActRIIa-mediated signaling may be used as an antagonist of ActRIIa polypeptide activities.
[0106] Antibodies that are specifically reactive with an activin A polypeptide may also be used as an antagonist. An anti-activin A antibody herein may be an antibody that binds to the activin A monomer or to the dimer; it is expected that the former may, for example, bind to Activin AB heterodimers, while the latter may not. An anti-activin A antibody herein may therefore be selective for activin A versus activin AB, or may bind to both. In some instances, an activin A antibody may also show binding to other activins, such as activin B, C or E, as well as heterodimers of any of the foregoing. In each case, a preferred antibody will inhibit the effects of activin A on osteoclasts and/or osteoblasts. The anti-activin A antibodies may disrupt ActRIIa binding. Anti-activin A antibodies may be purchased commercially or prepared using standard techniques as described herein. Exemplary anti-activin A antibodies are disclosed in PCT Publication No. WO 2008/031061.
[0107] Antibodies that are specifically reactive with an activin B polypeptide may also be used as an antagonist. An anti-activin B antibody herein may be an antibody that binds to the activin B monomer or to the dimer; it is expected that the former may, for example, bind to Activin AB heterodimers, while the latter may not. An anti-activin B antibody herein may therefore be selective for activin B versus activin AB, or may bind to both. In some instances, an activin B antibody may also show binding to other activins, such as activin A, C or E, as well as heterodimers of any of the foregoing. In each case, a preferred antibody will inhibit the effects of activin B on osteoclasts and/or osteoblasts. The anti-activin B antibodies may disrupt ActRIIa binding. Anti-activin B antibodies may be purchased commercially or prepared using standard techniques as described herein. Exemplary anti-activin B antibody is available for purchase from R&D Systems (catalog number MAB659), Minneapolis, Minn.
[0108] By using immunogens derived from an ActRIIa polypeptide or an activin polypeptide, anti-protein/anti-peptide antisera or monoclonal antibodies can be made by standard protocols (see, for example, Antibodies: A Laboratory Manual ed. by Harlow and Lane (Cold Spring Harbor Press: 1988)). A mammal, such as a mouse, a hamster or rabbit can be immunized with an immunogenic form of the ActRIIa polypeptide or activin A, antigenic fragments thereof which are capable of eliciting an antibody response, or fusion proteins of any of the foregoing. Techniques for conferring immunogenicity on a protein or peptide include conjugation to carriers or other techniques well known in the art. An immunogenic portion of an ActRIIa or activin polypeptide can be administered in the presence of adjuvant. The progress of immunization can be monitored by detection of antibody titers in plasma or serum. Standard ELISA or other immunoassays can be used with the immunogen as antigen to assess the levels of antibodies.
[0109] Following immunization of an animal with an antigenic preparation of an ActRIIa polypeptide or Activin A polypeptide, antisera can be obtained and, if desired, polyclonal antibodies can be isolated from the serum. To produce monoclonal antibodies, antibody-producing cells (lymphocytes) can be harvested from an immunized animal and fused by standard somatic cell fusion procedures with immortalizing cells such as myeloma cells to yield hybridoma cells. Such techniques are well known in the art, and include, for example, the hybridoma technique (originally developed by Kohler and Milstein, (1975) Nature, 256: 495-497), the human B cell hybridoma technique (Kozbar et al., (1983) Immunology Today, 4: 72), and the EBV-hybridoma technique to produce human monoclonal antibodies (Cole et al., (1985) Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, Inc. pp. 77-96). Hybridoma cells can be screened immunochemically for production of antibodies specifically reactive with an ActRIIa polypeptide or Activin A and monoclonal antibodies isolated from a culture comprising such hybridoma cells.
[0110] The term "antibody" as used herein is intended to include fragments thereof which are also specifically reactive with a subject polypeptide. Antibodies can be fragmented using conventional techniques and the fragments screened for utility in the same manner as described above for whole antibodies. For example, F(ab).sub.2 fragments can be generated by treating antibody with pepsin. The resulting F(ab).sub.2 fragment can be treated to reduce disulfide bridges to produce Fab fragments. The antibody of the present invention is further intended to include bispecific, single-chain, chimeric, humanized and fully human molecules having affinity for an ActRIIa or activin polypeptide conferred by at least one CDR region of the antibody. An antibody may further comprise a label attached thereto and able to be detected (e.g., the label can be a radioisotope, fluorescent compound, enzyme or enzyme co-factor).
[0111] In certain embodiments, the antibody is a recombinant antibody, which term encompasses any antibody generated in part by techniques of molecular biology, including CDR-grafted or chimeric antibodies, human or other antibodies assembled from library-selected antibody domains, single chain antibodies and single domain antibodies (e.g., human V.sub.H proteins or camelid V.sub.HH proteins). In certain embodiments, an antibody of the invention is a monoclonal antibody, and in certain embodiments, the invention makes available methods for generating novel antibodies. For example, a method for generating a monoclonal antibody that binds specifically to an ActRIIa polypeptide or activin polypeptide may comprise administering to a mouse an amount of an immunogenic composition comprising the antigen polypeptide effective to stimulate a detectable immune response, obtaining antibody-producing cells (e.g., cells from the spleen) from the mouse and fusing the antibody-producing cells with myeloma cells to obtain antibody-producing hybridomas, and testing the antibody-producing hybridomas to identify a hybridoma that produces a monocolonal antibody that binds specifically to the antigen. Once obtained, a hybridoma can be propagated in a cell culture, optionally in culture conditions where the hybridoma-derived cells produce the monoclonal antibody that binds specifically to the antigen. The monoclonal antibody may be purified from the cell culture.
[0112] The adjective "specifically reactive with" as used in reference to an antibody is intended to mean, as is generally understood in the art, that the antibody is sufficiently selective between the antigen of interest (e.g., an ActRIIa polypeptide or Activin A) and other antigens that are not of interest that the antibody is useful for, at minimum, detecting the presence of the antigen of interest in a particular type of biological sample. In certain methods employing the antibody, such as therapeutic applications, a higher degree of specificity in binding may be desirable. Monoclonal antibodies generally have a greater tendency (as compared to polyclonal antibodies) to discriminate effectively between the desired antigens and cross-reacting polypeptides. One characteristic that influences the specificity of an antibody:antigen interaction is the affinity of the antibody for the antigen. Although the desired specificity may be reached with a range of different affinities, generally preferred antibodies will have an affinity (a dissociation constant) of about 10.sup.-6, 10.sup.-7, 10.sup.-8, 10.sup.-9 or less. Given the extraordinarily tight binding between activin and
[0113] ActRIIa, it is expected that a neutralizing anti-activin or anti-ActRIIa antibody would generally have a dissociation constant of 10.sup.-10 or less.
[0114] In addition, the techniques used to screen antibodies in order to identify a desirable antibody may influence the properties of the antibody obtained. For example, if an antibody is to be used for binding an antigen in solution, it may be desirable to test solution binding. A variety of different techniques are available for testing interaction between antibodies and antigens to identify particularly desirable antibodies. Such techniques include ELISAs, surface plasmon resonance binding assays (e.g., the Biacore.TM. binding assay, Biacore AB, Uppsala, Sweden), sandwich assays (e.g., the paramagnetic bead system of IGEN International, Inc., Gaithersburg, Md.), western blots, immunoprecipitation assays, and immunohistochemistry.
[0115] Examples of categories of nucleic acid compounds that are activin or ActRIIa antagonists include antisense nucleic acids, RNAi constructs and catalytic nucleic acid constructs. A nucleic acid compound may be single or double stranded. A double stranded compound may also include regions of overhang or non-complementarity, where one or the other of the strands is single stranded. A single stranded compound may include regions of self-complementarity, meaning that the compound forms a so-called "hairpin" or "stem-loop" structure, with a region of double helical structure. A nucleic acid compound may comprise a nucleotide sequence that is complementary to a region consisting of no more than 1000, no more than 500, no more than 250, no more than 100 or no more than 50, 35, 30, 25, 22, 20 or 18 nucleotides of the full-length ActRIIa nucleic acid sequence or activin .beta.A or activin .beta.B nucleic acid sequence. The region of complementarity will preferably be at least 8 nucleotides, and optionally at least 10 or at least 15 nucleotides, and optionally between 15 and 25 nucleotides. A region of complementarity may fall within an intron, a coding sequence or a noncoding sequence of the target transcript, such as the coding sequence portion. Generally, a nucleic acid compound will have a length of about 8 to about 500 nucleotides or base pairs in length, and optionally the length will be about 14 to about 50 nucleotides. A nucleic acid may be a DNA (particularly for use as an antisense), RNA or RNA:DNA hybrid. Any one strand may include a mixture of DNA and RNA, as well as modified forms that cannot readily be classified as either DNA or RNA. Likewise, a double stranded compound may be DNA:DNA, DNA:RNA or RNA:RNA, and any one strand may also include a mixture of DNA and RNA, as well as modified forms that cannot readily be classified as either DNA or RNA. A nucleic acid compound may include any of a variety of modifications, including one or modifications to the backbone (the sugar-phosphate portion in a natural nucleic acid, including internucleotide linkages) or the base portion (the purine or pyrimidine portion of a natural nucleic acid). An antisense nucleic acid compound will preferably have a length of about 15 to about 30 nucleotides and will often contain one or more modifications to improve characteristics such as stability in the serum, in a cell or in a place where the compound is likely to be delivered, such as the stomach in the case of orally delivered compounds and the lung for inhaled compounds. In the case of an RNAi construct, the strand complementary to the target transcript will generally be RNA or modifications thereof. The other strand may be RNA, DNA or any other variation. The duplex portion of double stranded or single stranded "hairpin" RNAi construct will preferably have a length of 18 to 40 nucleotides in length and optionally about 21 to 23 nucleotides in length, so long as it serves as a Dicer substrate. Catalytic or enzymatic nucleic acids may be ribozymes or DNA enzymes and may also contain modified forms. Nucleic acid compounds may inhibit expression of the target by about 50%, 75%, 90% or more when contacted with cells under physiological conditions and at a concentration where a nonsense or sense control has little or no effect. Preferred concentrations for testing the effect of nucleic acid compounds are 1, 5 and 10 micromolar. Nucleic acid compounds may also be tested for effects on, for example, bone growth and mineralization.
5. Screening Assays
[0116] In certain aspects, the present invention relates to the use of ActRIIa polypeptides (e.g., soluble ActRIIa polypeptides) and activin polypeptides to identify compounds (agents) which are agonist or antagonists of the activin-ActRIIa signaling pathway. Compounds identified through this screening can be tested to assess their ability to modulate bone growth or mineralization in vitro. Optionally, these compounds can further be tested in animal models to assess their ability to modulate tissue growth in vivo.
[0117] There are numerous approaches to screening for therapeutic agents for modulating tissue growth by targeting activin and ActRIIa polypeptides. In certain embodiments, high-throughput screening of compounds can be carried out to identify agents that perturb activin or ActRIIa-mediated effects on bone. In certain embodiments, the assay is carried out to screen and identify compounds that specifically inhibit or reduce binding of an ActRIIa polypeptide to activin. Alternatively, the assay can be used to identify compounds that enhance binding of an ActRIIa polypeptide to activin. In a further embodiment, the compounds can be identified by their ability to interact with an activin or ActRIIa polypeptide.
[0118] A variety of assay formats will suffice and, in light of the present disclosure, those not expressly described herein will nevertheless be comprehended by one of ordinary skill in the art. As described herein, the test compounds (agents) of the invention may be created by any combinatorial chemical method. Alternatively, the subject compounds may be naturally occurring biomolecules synthesized in vivo or in vitro. Compounds (agents) to be tested for their ability to act as modulators of tissue growth can be produced, for example, by bacteria, yeast, plants or other organisms (e.g., natural products), produced chemically (e.g., small molecules, including peptidomimetics), or produced recombinantly. Test compounds contemplated by the present invention include non-peptidyl organic molecules, peptides, polypeptides, peptidomimetics, sugars, hormones, and nucleic acid molecules. In a specific embodiment, the test agent is a small organic molecule having a molecular weight of less than about 2,000 daltons.
[0119] The test compounds of the invention can be provided as single, discrete entities, or provided in libraries of greater complexity, such as made by combinatorial chemistry. These libraries can comprise, for example, alcohols, alkyl halides, amines, amides, esters, aldehydes, ethers and other classes of organic compounds. Presentation of test compounds to the test system can be in either an isolated form or as mixtures of compounds, especially in initial screening steps. Optionally, the compounds may be optionally derivatized with other compounds and have derivatizing groups that facilitate isolation of the compounds. Non-limiting examples of derivatizing groups include biotin, fluorescein, digoxygenin, green fluorescent protein, isotopes, polyhistidine, magnetic beads, glutathione S transferase (GST), photoactivatible crosslinkers or any combinations thereof.
[0120] In many drug screening programs which test libraries of compounds and natural extracts, high throughput assays are desirable in order to maximize the number of compounds surveyed in a given period of time. Assays which are performed in cell-free systems, such as may be derived with purified or semi-purified proteins, are often preferred as "primary" screens in that they can be generated to permit rapid development and relatively easy detection of an alteration in a molecular target which is mediated by a test compound. Moreover, the effects of cellular toxicity or bioavailability of the test compound can be generally ignored in the in vitro system, the assay instead being focused primarily on the effect of the drug on the molecular target as may be manifest in an alteration of binding affinity between an ActRIIa polypeptide and activin.
[0121] Merely to illustrate, in an exemplary screening assay of the present invention, the compound of interest is contacted with an isolated and purified ActRIIa polypeptide which is ordinarily capable of binding to activin. To the mixture of the compound and ActRIIa polypeptide is then added a composition containing an ActRIIa ligand. Detection and quantification of ActRIIa/activin complexes provides a means for determining the compound's efficacy at inhibiting (or potentiating) complex formation between the ActRIIa polypeptide and activin. The efficacy of the compound can be assessed by generating dose response curves from data obtained using various concentrations of the test compound. Moreover, a control assay can also be performed to provide a baseline for comparison. For example, in a control assay, isolated and purified activin is added to a composition containing the ActRIIa polypeptide, and the formation of ActRIIa/activin complex is quantitated in the absence of the test compound. It will be understood that, in general, the order in which the reactants may be admixed can be varied, and can be admixed simultaneously. Moreover, in place of purified proteins, cellular extracts and lysates may be used to render a suitable cell-free assay system.
[0122] Complex formation between the ActRIIa polypeptide and activin may be detected by a variety of techniques. For instance, modulation of the formation of complexes can be quantitated using, for example, detectably labeled proteins such as radiolabeled (e.g., .sup.32P, .sup.35S, .sup.14C or .sup.3H), fluorescently labeled (e.g., FITC), or enzymatically labeled ActRIIa polypeptide or activin, by immunoassay, or by chromatographic detection.
[0123] In certain embodiments, the present invention contemplates the use of fluorescence polarization assays and fluorescence resonance energy transfer (FRET) assays in measuring, either directly or indirectly, the degree of interaction between an ActRIIa polypeptide and its binding protein. Further, other modes of detection, such as those based on optical waveguides (PCT Publication WO 96/26432 and U.S. Pat. No. 5,677,196), surface plasmon resonance (SPR), surface charge sensors, and surface force sensors, are compatible with many embodiments of the invention.
[0124] Moreover, the present invention contemplates the use of an interaction trap assay, also known as the "two hybrid assay," for identifying agents that disrupt or potentiate interaction between an ActRIIa polypeptide and its binding protein. See for example, U.S. Pat. No. 5,283,317; Zervos et al. (1993) Cell 72:223-232; Madura et al. (1993) J Biol Chem 268:12046-12054; Bartel et al. (1993) Biotechniques 14:920-924; and Iwabuchi et al. (1993) Oncogene 8:1693-1696). In a specific embodiment, the present invention contemplates the use of reverse two hybrid systems to identify compounds (e.g., small molecules or peptides) that dissociate interactions between an ActRIIa polypeptide and its binding protein. See for example, Vidal and Legrain, (1999) Nucleic Acids Res 27:919-29; Vidal and Legrain, (1999) Trends Biotechnol 17:374-81; and U.S. Pat. Nos. 5,525,490; 5,955,280; and 5,965,368.
[0125] In certain embodiments, the subject compounds are identified by their ability to interact with an ActRIIa or activin polypeptide of the invention. The interaction between the compound and the ActRIIa or activin polypeptide may be covalent or non-covalent. For example, such interaction can be identified at the protein level using in vitro biochemical methods, including photo-crosslinking, radiolabeled ligand binding, and affinity chromatography (Jakoby W B et al., 1974, Methods in Enzymology 46: 1). In certain cases, the compounds may be screened in a mechanism based assay, such as an assay to detect compounds which bind to an activin or ActRIIa polypeptide. This may include a solid phase or fluid phase binding event. Alternatively, the gene encoding an activin or ActRIIa polypeptide can be transfected with a reporter system (e.g., .beta.-galactosidase, luciferase, or green fluorescent protein) into a cell and screened against the library preferably by a high throughput screening or with individual members of the library. Other mechanism based binding assays may be used, for example, binding assays which detect changes in free energy. Binding assays can be performed with the target fixed to a well, bead or chip or captured by an immobilized antibody or resolved by capillary electrophoresis. The bound compounds may be detected usually using colorimetric or fluorescence or surface plasmon resonance.
[0126] In certain aspects, the present invention provides methods and agents for modulating (stimulating or inhibiting) bone formation and increasing bone mass. Therefore, any compound identified can be tested in whole cells or tissues, in vitro or in vivo, to confirm their ability to modulate bone growth or mineralization. Various methods known in the art can be utilized for this purpose.
[0127] For example, the effect of the ActRIIa or activin polypeptides or test compounds on bone or cartilage growth can be determined by measuring induction of Msx2 or differentiation of osteoprogenitor cells into osteoblasts in cell based assays (see, e.g., Daluiski et al., Nat Genet. 2001, 27(1):84-8; Hino et al., Front Biosci. 2004, 9:1520-9). Another example of cell-based assays includes analyzing the osteogenic activity of the subject ActRIIa or activin polypeptides and test compounds in mesenchymal progenitor and osteoblastic cells. To illustrate, recombinant adenoviruses expressing an activin or ActRIIa polypeptide can be constructed to infect pluripotent mesenchymal progenitor C3H10T1/2 cells, preosteoblastic C2C12 cells, and osteoblastic TE-85 cells. Osteogenic activity is then determined by measuring the induction of alkaline phosphatase, osteocalcin, and matrix mineralization (see, e.g., Cheng et al., J bone Joint Surg Am. 2003, 85-A(8):1544-52).
[0128] The present invention also contemplates in vivo assays to measure bone or cartilage growth. For example, Namkung-Matthai et al., Bone, 28:80-86 (2001) discloses a rat osteoporotic model in which bone repair during the early period after fracture is studied. Kubo et al., Steroid Biochemistry & Molecular Biology, 68:197-202 (1999) also discloses a rat osteoporotic model in which bone repair during the late period after fracture is studied. Andersson et al., J. Endocrinol. 170:529-537 describe a mouse osteoporosis model in which mice are ovariectomized, which causes the mice to lose substantial bone mineral content and bone mineral density, with the trabecular bone losing roughly 50% of bone mineral density. Bone density could be increased in the ovariectomized mice by administration of factors such as parathyroid hormone. In certain aspects, the present invention makes use of fracture healing assays that are known in the art. These assays include fracture technique, histological analysis, and biomechanical analysis, which are described in, for example, U.S. Pat. No. 6,521,750, which is incorporated by reference in its entirety for its disclosure of experimental protocols for causing as well as measuring the extent of fractures, and the repair process.
6. Exemplary Therapeutic Uses
[0129] In certain embodiments, activin-ActRIIa antagonists (e.g., ActRIIa polypeptides) of the present invention can be used for treating or preventing a disease or condition that is associated with bone damage, whether, e.g., through breakage, loss or demineralization. In certain embodiments, the present invention provides methods of treating or preventing bone damage in an individual in need thereof through administering to the individual a therapeutically effective amount of an activin-ActRIIa antagonist, particularly an ActRIIa polypeptide. In certain embodiments, the present invention provides methods of promoting bone growth or mineralization in an individual in need thereof through administering to the individual a therapeutically effective amount of an activin-ActRIIa antagonist, particularly an ActRIIa polypeptide. These methods are preferably aimed at therapeutic and prophylactic treatments of animals, and more preferably, humans. In certain embodiments, the disclosure provides for the use of activin-ActRIIa antagonists (particularly soluble ActRIIa polypeptides and neutralizing antibodies targeted to activin or ActRIIa) for the treatment of disorders associated with low bone density or decreased bone strength.
[0130] As used herein, a therapeutic that "prevents" a disorder or condition refers to a compound that, in a statistical sample, reduces the occurrence of the disorder or condition in the treated sample relative to an untreated control sample, or delays the onset or reduces the severity of one or more symptoms of the disorder or condition relative to the untreated control sample. The term "treating" as used herein includes prophylaxis of the named condition or amelioration or elimination of the condition once it has been established. In either case, prevention or treatment may be discerned in the diagnosis provided by a physician and the intended result of administration of the therapeutic agent.
[0131] The disclosure provides methods of inducing bone and/or cartilage formation, preventing bone loss, increasing bone mineralization or preventing the demineralization of bone. For example, the subject activin-ActRIIa antagonists have application in treating osteoporosis and the healing of bone fractures and cartilage defects in humans and other animals. ActRIIa or activin polypeptides may be useful in patients that are diagnosed with subclinical low bone density, as a protective measure against the development of osteoporosis.
[0132] In one specific embodiment, methods and compositions of the present invention may find medical utility in the healing of bone fractures and cartilage defects in humans and other animals. The subject methods and compositions may also have prophylactic use in closed as well as open fracture reduction and also in the improved fixation of artificial joints. De novo bone formation induced by an osteogenic agent contributes to the repair of congenital, trauma-induced, or oncologic resection induced craniofacial defects, and also is useful in cosmetic plastic surgery. In certain cases, the subject activin-ActRIIa antagonists may provide an environment to attract bone-forming cells, stimulate growth of bone-forming cells or induce differentiation of progenitors of bone-forming cells. Activin-ActRila antagonists of the invention may also be useful in the treatment of osteoporosis.
[0133] Methods and compositions of the invention can be applied to conditions characterized by or causing bone loss, such as osteoporosis (including secondary osteoporosis), hyperparathyroidism, Cushing's disease, Paget's disease, thyrotoxicosis, chronic diarrheal state or malabsorption, renal tubular acidosis, or anorexia nervosa.
[0134] Osteoporosis may be caused by, or associated with, various factors. Being female, particularly a post-menopausal female, having a low body weight, and leading a sedentary lifestyle are all risk factors for osteoporosis (loss of bone mineral density, leading to fracture risk). Persons having any of the following profiles may be candidates for treatment with an ActRIIa antagonist: a post-menopausal woman and not taking estrogen or other hormone replacement therapy; a person with a personal or maternal history of hip fracture or smoking; a post-menopausal woman who is tall (over 5 feet 7 inches) or thin (less than 125 pounds); a man with clinical conditions associated with bone loss; a person using medications that are known to cause bone loss, including corticosteroids such as Prednisone.TM., various anti-seizure medications such as Dilantin.TM. and certain barbiturates, or high-dose thyroid replacement drugs; a person having type 1 diabetes, liver disease, kidney disease or a family history of osteoporosis; a person having high bone turnover (e.g., excessive collagen in urine samples); a person with a thyroid condition, such as hyperthyroidism; a person who has experienced a fracture after only mild trauma; a person who has had x-ray evidence of vertebral fracture or other signs of osteoporosis.
[0135] As noted above, osteoporosis can also result as a condition associated with another disorder or from the use of certain medications. Osteoporosis resulting from drugs or another medical condition is known as secondary osteoporosis. In a condition known as Cushing's disease, the excess amount of cortisol produced by the body results in osteoporosis and fractures. The most common medications associated with secondary osteoporosis are the corticosteroids, a class of drugs that act like cortisol, a hormone produced naturally by the adrenal glands. Although adequate levels of thyroid hormones (which are produced by the thyroid gland) are needed for the development of the skeleton, excess thyroid hormone can decrease bone mass over time. Antacids that contain aluminum can lead to bone loss when taken in high doses by people with kidney problems, particularly those undergoing dialysis. Other medications that can cause secondary osteoporosis include phenytoin (Dilantin) and barbiturates that are used to prevent seizures; methotrexate (Rheumatrex, Immunex, Folex PFS), a drug for some forms of arthritis, cancer, and immune disorders; cyclosporine (Sandimmune, Neoral), a drug used to treat some autoimmune diseases and to suppress the immune system in organ transplant patients; luteinizing hormone-releasing hormone agonists (Lupron, Zoladex), used to treat prostate cancer and endometriosis; heparin (Calciparine, Liquaemin), an anticlotting medication; and cholestyramine (Questran) and colestipol (Colestid), used to treat high cholesterol. Bone loss resulting from cancer therapy is widely recognized and termed cancer therapy induced bone loss (CTIBL). Bone metastases can create cavities in the bone that may be corrected by treatment with activin-ActRIIa antagonists.
[0136] In a preferred embodiment, activin-ActRIIa antagonists, particularly a soluble ActRIIa, disclosed herein may be used in cancer patients. Patients having certain tumors (e.g. prostate, breast, multiple myeloma or any tumor causing hyperparathyroidism) are at high risk for bone loss due to tumor-induced bone loss as well as bone metastases and therapeutic agents. Such patients may be treated with activin-ActRIIa antagonists even in the absence of evidence of bone loss or bone metastases. Patients may also be monitored for evidence of bone loss or bone metastases, and may be treated with activin-ActRila antagonists in the event that indicators suggest an increased risk. Generally, DEXA scans are employed to assess changes in bone density, while indicators of bone remodeling may be used to assess the likelihood of bone metastases. Serum markers may be monitored. Bone specific alkaline phosphatase (BSAP) is an enzyme that is present in osteoblasts. Blood levels of BSAP are increased in patients with bone metastasis and other conditions that result in increased bone remodeling. Osteocalcin and procollagen peptides are also associated with bone formation and bone metastases. Increases in BSAP have been detected in patients with bone metastasis caused by prostate cancer, and to a lesser degree, in bone metastases from breast cancer. Bone Morphogenetic Protein-7 (BMP-7) levels are high in prostate cancer that has metastasized to bone, but not in bone metastases due to bladder, skin, liver, or lung cancer. Type I Carboxy-terminal telopeptide (ICTP) is a crosslink found in collagen that is formed during to the resorption of bone. Since bone is constantly being broken down and reformed, ICTP will be found throughout the body. However, at the site of bone metastasis, the level will be significantly higher than in an area of normal bone. ICTP has been found in high levels in bone metastasis due to prostate, lung, and breast cancer. Another collagen crosslink, Type I N-terminal telopeptide (NTx), is produced along with ICTP during bone turnover. The amount of NTx is increased in bone metastasis caused by many different types of cancer including lung, prostate, and breast cancer. Also, the levels of NTx increase with the progression of the bone metastasis. Therefore, this marker can be used to both detect metastasis as well as measure the extent of the disease. Other markers of resorption include pyridinoline and deoxypyridinoline. Any increase in resorption markers or markers of bone metastases indicate the need for activin-ActRIIa antagonist therapy in a patient.
[0137] Activin-ActRIIa antagonists may be conjointly administered with other pharmaceutical agents. Conjoint administration may be accomplished by administration of a single co-formulation, by simultaneous administration or by administration at separate times. Activin-ActRIIa antagonists may be particularly advantageous if administered with other bone-active agents. A patient may benefit from conjointly receiving activin-ActRIIa antagonist and taking calcium supplements, vitamin D, appropriate exercise and/or, in some cases, other medication. Examples of other medications incude, bisphosphonates (alendronate, ibandronate and risedronate), calcitonin, estrogens, parathyroid hormone and raloxifene. The bisphosphonates (alendronate, ibandronate and risedronate), calcitonin, estrogens and raloxifene affect the bone remodeling cycle and are classified as anti-resorptive medications. Bone remodeling consists of two distinct stages: bone resorption and bone formation. Anti-resorptive medications slow or stop the bone-resorbing portion of the bone-remodeling cycle but do not slow the bone-forming portion of the cycle. As a result, new formation continues at a greater rate than bone resorption, and bone density may increase over time. Teriparatide, a form of parathyroid hormone, increases the rate of bone formation in the bone remodeling cycle. Alendronate is approved for both the prevention (5 mg per day or 35 mg once a week) and treatment (10 mg per day or 70 mg once a week) of postmenopausal osteoporosis. Alendronate reduces bone loss, increases bone density and reduces the risk of spine, wrist and hip fractures. Alendronate also is approved for treatment of glucocorticoid-induced osteoporosis in men and women as a result of long-term use of these medications (i.e., prednisone and cortisone) and for the treatment of osteoporosis in men. Alendronate plus vitamin D is approved for the treatment of osteoporosis in postmenopausal women (70 mg once a week plus vitamin D), and for treatment to improve bone mass in men with osteoporosis. Ibandronate is approved for the prevention and treatment of postmenopausal osteoporosis. Taken as a once-a-month pill (150 mg), ibandronate should be taken on the same day each month. Ibandronate reduces bone loss, increases bone density and reduces the risk of spine fractures. Risedronate is approved for the prevention and treatment of postmenopausal osteoporosis. Taken daily (5 mg dose) or weekly (35 mg dose or 35 mg dose with calcium), risedronate slows bone loss, increases bone density and reduces the risk of spine and non-spine fractures. Risedronate also is approved for use by men and women to prevent and/or treat glucocorticoid-induced osteoporosis that results from long-term use of these medications (i.e., prednisone or cortisone). Calcitonin is a naturally occurring hormone involved in calcium regulation and bone metabolism. In women who are more than 5 years beyond menopause, calcitonin slows bone loss, increases spinal bone density, and may relieve the pain associated with bone fractures. Calcitonin reduces the risk of spinal fractures. Calcitonin is available as an injection (50-100 IU daily) or nasal spray (200 IU daily). Estrogen therapy (ET)/Hormone therapy (HT) is approved for the prevention of osteoporosis. ET has been shown to reduce bone loss, increase bone density in both the spine and hip, and reduce the risk of hip and spinal fractures in postmenopausal women. ET is administered most commonly in the form of a pill or skin patch that delivers a low dose of approximately 0.3 mg daily or a standard dose of approximately 0.625 mg daily and is effective even when started after age 70. When estrogen is taken alone, it can increase a woman's risk of developing cancer of the uterine lining (endometrial cancer). To eliminate this risk, healthcare providers prescribe the hormone progestin in combination with estrogen (hormone replacement therapy or HT) for those women who have an intact uterus. ET/HT relieves menopause symptoms and has been shown to have a beneficial effect on bone health. Side effects may include vaginal bleeding, breast tenderness, mood disturbances and gallbladder disease. Raloxifene, 60 mg a day, is approved for the prevention and treatment of postmenopausal osteoporosis. It is from a class of drugs called Selective Estrogen Receptor Modulators (SERMs) that have been developed to provide the beneficial effects of estrogens without their potential disadvantages. Raloxifene increases bone mass and reduces the risk of spine fractures. Data are not yet available to demonstrate that raloxifene can reduce the risk of hip and other non-spine fractures. Teriparatide, a form of parathyroid hormone, is approved for the treatment of osteoporosis in postmenopausal women and men who are at high risk for a fracture. This medication stimulates new bone formation and significantly increases bone mineral density. In postmenopausal women, fracture reduction was noted in the spine, hip, foot, ribs and wrist. In men, fracture reduction was noted in the spine, but there were insufficient data to evaluate fracture reduction at other sites. Teriparatide is self-administered as a daily injection for up to 24 months.
7. Pharmaceutical Compositions
[0138] In certain embodiments, activin-ActRIIa antagonists (e.g., ActRIIa polypeptides) of the present invention are formulated with a pharmaceutically acceptable carrier. For example, an ActRIIa polypeptide can be administered alone or as a component of a pharmaceutical formulation (therapeutic composition). The subject compounds may be formulated for administration in any convenient way for use in human or veterinary medicine.
[0139] In certain embodiments, the therapeutic method of the invention includes administering the composition systemically, or locally as an implant or device. When administered, the therapeutic composition for use in this invention is, of course, in a pyrogen-free, physiologically acceptable form. Therapeutically useful agents other than the ActRIIa antagonists which may also optionally be included in the composition as described above, may be administered simultaneously or sequentially with the subject compounds (e.g., ActRIIa polypeptides) in the methods of the invention.
[0140] Typically, ActRIIa antagonists will be administered parentally. Pharmaceutical compositions suitable for parenteral administration may comprise one or more ActRIIa polypeptides in combination with one or more pharmaceutically acceptable sterile isotonic aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents. Examples of suitable aqueous and nonaqueous carriers which may be employed in the pharmaceutical compositions of the invention include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
[0141] Further, the composition may be encapsulated or injected in a form for delivery to a target tissue site (e.g., bone). In certain embodiments, compositions of the present invention may include a matrix capable of delivering one or more therapeutic compounds (e.g., ActRIIa polypeptides) to a target tissue site (e.g., bone), providing a structure for the developing tissue and optimally capable of being resorbed into the body. For example, the matrix may provide slow release of the ActRIIa polypeptides. Such matrices may be formed of materials presently in use for other implanted medical applications.
[0142] The choice of matrix material is based on biocompatibility, biodegradability, mechanical properties, cosmetic appearance and interface properties. The particular application of the subject compositions will define the appropriate formulation. Potential matrices for the compositions may be biodegradable and chemically defined calcium sulfate, tricalciumphosphate, hydroxyapatite, polylactic acid and polyanhydrides. Other potential materials are biodegradable and biologically well defined, such as bone or dermal collagen.
[0143] Further matrices are comprised of pure proteins or extracellular matrix components. Other potential matrices are non-biodegradable and chemically defined, such as sintered hydroxyapatite, bioglass, aluminates, or other ceramics. Matrices may be comprised of combinations of any of the above mentioned types of material, such as polylactic acid and hydroxyapatite or collagen and tricalciumphosphate. The bioceramics may be altered in composition, such as in calcium-aluminate-phosphate and processing to alter pore size, particle size, particle shape, and biodegradability.
[0144] In certain embodiments, methods of the invention can be administered for orally, e.g., in the form of capsules, cachets, pills, tablets, lozenges (using a flavored basis, usually sucrose and acacia or tragacanth), powders, granules, or as a solution or a suspension in an aqueous or non-aqueous liquid, or as an oil-in-water or water-in-oil liquid emulsion, or as an elixir or syrup, or as pastilles (using an inert base, such as gelatin and glycerin, or sucrose and acacia) and/or as mouth washes and the like, each containing a predetermined amount of an agent as an active ingredient. An agent may also be administered as a bolus, electuary or paste.
[0145] In solid dosage forms for oral administration (capsules, tablets, pills, dragees, powders, granules, and the like), one or more therapeutic compounds of the present invention may be mixed with one or more pharmaceutically acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose, and/or acacia; (3) humectants, such as glycerol; (4) disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds; (7) wetting agents, such as, for example, cetyl alcohol and glycerol monostearate; (8) absorbents, such as kaolin and bentonite clay; (9) lubricants, such a talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, and mixtures thereof; and (10) coloring agents. In the case of capsules, tablets and pills, the pharmaceutical compositions may also comprise buffering agents. Solid compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
[0146] Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, microemulsions, solutions, suspensions, syrups, and elixirs. In addition to the active ingredient, the liquid dosage forms may contain inert diluents commonly used in the art, such as water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor, and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, and mixtures thereof. Besides inert diluents, the oral compositions can also include adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, coloring, perfuming, and preservative agents.
[0147] Suspensions, in addition to the active compounds, may contain suspending agents such as ethoxylated isostearyl alcohols, polyoxyethylene sorbitol, and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
[0148] The compositions of the invention may also contain adjuvants, such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of the action of microorganisms may be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It may also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into the compositions. In addition, prolonged absorption of the injectable pharmaceutical form may be brought about by the inclusion of agents which delay absorption, such as aluminum monostearate and gelatin.
[0149] It is understood that the dosage regimen will be determined by the attending physician considering various factors which modify the action of the subject compounds of the invention (e.g., ActRIIa polypeptides). The various factors include, but are not limited to, amount of bone weight desired to be formed, the degree of bone density loss, the site of bone damage, the condition of the damaged bone, the patient's age, sex, and diet, the severity of any disease that may be contributing to bone loss, time of administration, and other clinical factors. Optionally, the dosage may vary with the type of matrix used in the reconstitution and the types of compounds in the composition. The addition of other known growth factors to the final composition, may also affect the dosage. Progress can be monitored by periodic assessment of bone growth and/or repair, for example, X-rays (including DEXA), histomorphometric determinations, and tetracycline labeling.
[0150] Experiments with mice have demonstrated that effects of ActRIIa-Fc on bone are detectable when the compound is dosed at intervals and amounts sufficient to achieve serum concentrations of 0.2 .mu.g/kg or greater, and serum levels of 1 .mu.g/kg or 2 .mu.g/kg or greater are desirable for achieving significant effects on bone density and strength. Although there is no indication that higher doses of ActRIIa-Fc are undesirable due to side effects, dosing regimens may be designed to reach serum concentrations of between 0.2 and 15 .mu.g/kg, and optionally between 1 and 5 .mu.g/kg. In humans, serum levels of 0.2 .mu.g/kg may be achieved with a single dose of 0.1 mg/kg or greater and serum levels of 1 .mu.g/kg may be achieved with a single dose of 0.3 mg/kg or greater. The observed serum half-life of the molecule is between about 20 and 30 days, substantially longer than most Fc fusion proteins, and thus a sustained effective serum level may be achieved, for example, by dosing with 0.2-0.4 mg/kg on a weekly or biweekly basis, or higher doses may be used with longer intervals between dosings. For example, doses of 1-3 mg/kg might be used on a monthly or bimonthly basis, and the effect on bone may be sufficiently durable that dosing is necessary only once every 3, 4, 5, 6, 9, 12 or more months.
[0151] In certain embodiments, the present invention also provides gene therapy for the in vivo production of ActRIIa polypeptides. Such therapy would achieve its therapeutic effect by introduction of the ActRIIa polynucleotide sequences into cells or tissues having the disorders as listed above. Delivery of ActRIIa polynucleotide sequences can be achieved using a recombinant expression vector such as a chimeric virus or a colloidal dispersion system. Preferred for therapeutic delivery of ActRIIa polynucleotide sequences is the use of targeted liposomes.
[0152] Various viral vectors which can be utilized for gene therapy as taught herein include adenovirus, herpes virus, vaccinia, or, preferably, an RNA virus such as a retrovirus. Preferably, the retroviral vector is a derivative of a murine or avian retrovirus. Examples of retroviral vectors in which a single foreign gene can be inserted include, but are not limited to: Moloney murine leukemia virus (MoMuLV), Harvey murine sarcoma virus (HaMuSV), murine mammary tumor virus (MuMTV), and Rous Sarcoma Virus (RSV). A number of additional retroviral vectors can incorporate multiple genes. All of these vectors can transfer or incorporate a gene for a selectable marker so that transduced cells can be identified and generated. Retroviral vectors can be made target-specific by attaching, for example, a sugar, a glycolipid, or a protein. Preferred targeting is accomplished by using an antibody. Those of skill in the art will recognize that specific polynucleotide sequences can be inserted into the retroviral genome or attached to a viral envelope to allow target specific delivery of the retroviral vector containing the ActRIIa polynucleotide. In a preferred embodiment, the vector is targeted to bone or cartilage.
[0153] Alternatively, tissue culture cells can be directly transfected with plasmids encoding the retroviral structural genes gag, pol and env, by conventional calcium phosphate transfection. These cells are then transfected with the vector plasmid containing the genes of interest. The resulting cells release the retroviral vector into the culture medium.
[0154] Another targeted delivery system for ActRIIa polynucleotides is a colloidal dispersion system. Colloidal dispersion systems include macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes. The preferred colloidal system of this invention is a liposome. Liposomes are artificial membrane vesicles which are useful as delivery vehicles in vitro and in vivo. RNA, DNA and intact virions can be encapsulated within the aqueous interior and be delivered to cells in a biologically active form (see e.g., Fraley, et al., Trends Biochem. Sci., 6:77, 1981). Methods for efficient gene transfer using a liposome vehicle, are known in the art, see e.g., Mannino, et al., Biotechniques, 6:682, 1988. The composition of the liposome is usually a combination of phospholipids, usually in combination with steroids, especially cholesterol. Other phospholipids or other lipids may also be used. The physical characteristics of liposomes depend on pH, ionic strength, and the presence of divalent cations.
[0155] Examples of lipids useful in liposome production include phosphatidyl compounds, such as phosphatidylglycerol, phosphatidylcholine, phosphatidylserine, phosphatidylethanolamine, sphingolipids, cerebrosides, and gangliosides. Illustrative phospholipids include egg phosphatidylcholine, dipalmitoylphosphatidylcholine, and di stearoylphosphatidylcholine. The targeting of liposomes is also possible based on, for example, organ-specificity, cell-specificity, and organelle-specificity and is known in the art.
EXEMPLIFICATION
[0156] The invention now being generally described, it will be more readily understood by reference to the following examples, which are included merely for purposes of illustration of certain embodiments and embodiments of the present invention, and are not intended to limit the invention.
Example 1
ActRIIa-Fc Fusion Proteins
[0157] Applicants constructed a soluble ActRIIa fusion protein that has the extracellular domain of human ActRIIa fused to a human or mouse Fc domain with a minimal linker in between. The constructs are referred to as ActRIIa-hFc and ActRIIa-mFc, respectively.
[0158] ActRIIa-hFc is shown below as purified from CHO cell lines (SEQ ID NO: 7):
TABLE-US-00007 ILGRSETQECLFFNANWEKDRTNQTGVEPCYGDKDKRRHCFATWKNISGS IEIVKQGCWLDDINCYDRTDCVEKKDSPEVYFCCCEGNMCNEKFSYFPEM EVTQPTSNPVTPKPPTGGGTHTCPPCPAPELLGGPSVFLEPPKPKDTLMI SRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVV SVLTVLHQDWLNGKEYKCKVSNKALPVPIEKTISKAKGQPREPQVYTLPP SREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGS FFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
[0159] The ActRIIa-hFc and ActRIIa-mFc proteins were expressed in CHO cell lines. Three different leader sequences were considered:
TABLE-US-00008 (i) Honey bee mellitin (HBML): (SEQ ID NO: 8) MKFLVNVALVFMVVYISYIYA (ii) Tissue Plasminogen Activator (TPA): (SEQ ID NO: 9) MDAMKRGLCCVLLLCGAVFVSP (iii) Native: (SEQ ID NO: 10) MGAAAKLAFAVFLISCSSGA.
[0160] The selected form employs the TPA leader and has the following unprocessed amino acid sequence:
TABLE-US-00009 (SEQ ID NO: 13) MDAMKRGLCCVLLLCGAVFVSPGAAILGRSETQECLFFNANWEKDRTNQT GVEPCYGDKDKRRHCFATWKNISGSIEIVKQGCWLDDINCYDRTDCVEKK DSPEVYFCCCEGNMCNEKFSYFPEMEVTQPTSNPVTPKPPTGGGTHTCPP CPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWY VDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKAL PVPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCLVKGFYPSDIA VEWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVM HEALHNHYTQKSLSLSPGK
[0161] This polypeptide is encoded by the following nucleic acid sequence:
TABLE-US-00010 (SEQ ID NO: 14) ATGGATGCAATGAAGAGAGGGCTCTGCTGTGTGCTGCTGCTGTGTGGAGC AGTCTTCGTTTCGCCCGGCGCCGCTATACTTGGTAGATCAGAAACTCAGG AGTGTCTTTTTTTAATGCTAATTGGGAAAAAGACAGAACCAATCAAACTG GTGTTGAACCGTGTTATGGTGACAAAGATAAACGGCGGCATTGTTTTGCT ACCTGGAAGAATATTTCTGGTTCCATTGAATAGTGAAACAAGGTTGTTGG CTGGATGATATCAACTGCTATGACAGGACTGATTGTGTAGAAAAAAAAGA CAGCCCTGAAGTATATTTCTGTTGCTGTGAGGGCAATATGTGTAATGAAA AGTTTTCTTATTTTCCGGAGATGGAAGTCACACAGCCCACTTCAAATCCA GTTACACCTAAGCCACCCACCGGTGGTGGAACTCACACATGCCCACCGTG CCCAGCACCTGAACTCCTGGGGGGACCGTCAGTCTTCCTCTTCCCCCCAA AACCCAAGGACACCCTCATGATCTCCCGGACCCCTGAGGTCACATGCGTG GTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAACTGGTACGT GGACGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGT ACAACAGCACGTACCGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGAC TGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGCCCTCCC AGTCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAAC CACAGGTGTACACCCTGCCCCCATCCCGGGAGGAGATGACCAAGAACCAG GTCAGCCTGACCTGCCTGGTCAAAGGCTTCTATCCCAGCGACATCGCCGT GGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACCACGCCTC CCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTATAGCAAGCTCACCGTG GACAAGAGCAGGTGGCAGCAGGGGAACGTCTTCTCATGCTCCGTGATGCA TGAGGCTCTGCACAACCACTACACGCAGAAGAGCCTCTCCCTGTCTCCGG GTAAATGAGAATTC
[0162] Both ActRIIa-hFc and ActRIIa-mFc were remarkably amenable to recombinant expression. As shown in FIG. 1, the protein was purified as a single, well-defined peak of protein. N-terminal sequencing revealed a single sequence of -ILGRSETQE (SEQ ID NO: 11). Purification could be achieved by a series of column chromatography steps, including, for example, three or more of the following, in any order: protein A chromatography, Q sepharose chromatography, phenylsepharose chromatography, size exclusion chromatography, and cation exchange chromatography. The purification could be completed with viral filtration and buffer exchange. The ActRIIa-hFc protein was purified to a purity of >98% as determined by size exclusion chromatography and >95% as determined by SDS PAGE.
[0163] ActRIIa-hFc and ActRIIa-mFc showed a high affinity for ligands, particularly activin A. GDF-11 or Activin A ("ActA") were immobilized on a Biacore CM5 chip using standard amine coupling procedure. ActRIIa-hFc and ActRIIa-mFc proteins were loaded onto the system, and binding was measured. ActRIIa-hFc bound to activin with a dissociation constant (K.sub.D) of 5.times.10.sup.-12, and the protein bound to GDF11 with a K.sub.D of 9.96.times.10.sup.-9. See FIG. 2. ActRIIa-mFc behaved similarly.
[0164] An A-204 Reporter Gene Assay was used to evaluate the effects of ActRIIa-hFc proteins on signaling by GDF-11 and Activin A. Cell line: Human Rhabdomyosarcoma (derived from muscle). Reporter vector: pGL3(CAGA)12 (Described in Dennler et al, 1998, EMBO 17: 3091-3100.) See FIG. 3. The CAGA12 motif is present in TGF-Beta responsive genes (PAI-1 gene) , so this vector is of general use for factors signaling through Smad2 and 3.
[0165] Day 1: Split A-204 cells into 48-well plate.
[0166] Day 2: A-204 cells transfected with 10 .mu.g pGL3(CAGA)12 or pGL3(CAGA)12 (10 .mu.g)+pRLCMV (1 .mu.g) and Fugene.
[0167] Day 3: Add factors (diluted into medium+0.1% BSA). Inhibitors need to be preincubated with Factors for 1 hr before adding to cells. 6 hrs later, cells rinsed with PBS, and lyse cells.
[0168] This is followed by a Luciferase assay. Typically in this assay, in the absence of any inhibitors, Activin A shows roughly 10 fold stimulation of reporter gene expression and an ED50.about.2 ng/ml. GDF-11: 16 fold stimulation, ED50: .about.1.5 ng/ml. GDF-8 shows an effect similar to GDF-11.
[0169] As shown in FIG. 4, ActRIIa-hFc and ActRIIa-mFc inhibit GDF-8 mediated signaling at picomolar concentrations. As shown in FIG. 5, three different preparations of ActRIIa-hFc inhibited GDF-11 signaling with an IC50 of approximately 200 pM.
[0170] The ActRIIa-hFc was very stable in pharmacokinetic studies. Rats were dosed with 1 mg/kg, 3 mg/kg or 10 mg/kg of ActRIIa-hFc protein and plasma levels of the protein were measured at 24, 48, 72, 144 and 168 hours. In a separate study, rats were dosed at 1 mg/kg, 10 mg/kg or 30 mg/kg. In rats, ActRIIa-hFc had an 11-14 day serum half life and circulating levels of the drug were quite high after two weeks (11 .mu.g/ml, 110 .mu.g/ml or 304 .mu.g/ml for initial administrations of 1 mg/kg, 10 mg/kg or 30 mg/kg, respectively.) In cynomolgus monkeys, the plasma half life was substantially greater than 14 days and circulating levels of the drug were 25 .mu.g/ml, 304 .mu.g/ml or 1440 .mu.g/ml for initial administrations of 1 mg/kg, 10 mg/kg or 30 mg/kg, respectively. Preliminary results in humans suggests that the serum half life is between about 20 and 30 days.
Example 2
ActRIIa-mFc Promotes Bone Growth In Vivo
[0171] Normal female mice (BALB/c) were dosed with ActRIIa-mFc at a level of 1 mg/kg/dose, 3 mg/kg/dose or 10 mg/kg/dose, with doses given twice weekly. Bone mineral density and bone mineral content were determined by DEXA, see FIG. 6.
[0172] In BALB/c female mice, DEXA scans showed a significant increase (>20%) in bone mineral density and content as a result of ActRIIa-mFc treatment. See FIGS. 7 and 8.
[0173] Thus, antagonism of ActRIIa caused increased bone density and content in normal female mice. As a next step, the effect of ActRIIa-mFc on bone in a mouse model for osteoporosis was tested.
[0174] Andersson et al. (2001), established that ovariectomized mice suffered substantial bone loss (rougly 50% loss of trabecular bone six weeks post-operation), and that bone loss in these mice could be corrected with candidate therapeutic agents, such as parathyroid hormone.
[0175] Applicants used C57BL6 female mice that were ovariectomized (OVX) or sham operated at 4-5 weeks of age. Eight weeks after surgery, treatment with ActRIIa-mFc (10 mg/kg, twice weekly) or control (PBS) was initiated. Bone density was measured by CT scanner.
[0176] As shown in FIG. 9, untreated, ovariectomized mice showed substantial loss of trabecular bone density relative to the sham controls after six weeks. ActRIIa-mFc treatment restored bone density to the level of the sham operated mice. At 6 and 12 weeks of the treatment, ActRIIa-mFc caused substantial increase in trabecular bone of OVX mice. See FIG. 10. After 6 weeks of treatment, bone density increased by 24% relative to PBS controls. After 12 weeks, the increase was 27%.
[0177] In the sham operated mice, ActRIIa-mFc also caused a substantial increase in trabecular bone. See FIG. 11. After 6 and 12 weeks, the treatment produced a 35% increase relative to controls.
[0178] In an additional set of experiments, ovariectomized (OVX) or sham operated mice as described above were treated with ActRIIa-mFc (10 mg/kg, twice weekly) or control (PBS) over twelve weeks. Similar to the results described above for ActRIIa-mFc, OVX mice receiving ActRIIa-mFc exhibited an increase in trabecular bone density of 15% by as early as four weeks and 25% after 12 weeks of treatment (FIG. 12). Sham operated mice receiving ActRIIa-mFc similarly showed an increase in trabecular bone density of 22% by as early as four weeks and of 32% after 12 weeks of treatment (FIG. 13).
[0179] After twelve weeks of treatment with ActRIIa-mFc, whole body and ex vivo femur DEXA analysis showed that treatment induces an increase in bone density in both ovariectomized and sham operated mice (FIGS. 14A and 14B, respectively). These results are also supported by ex vivo pQCT analysis of the femoral midshaft which demonstrated a significant increase in both total and cortical bone density after twelve weeks of treatment with ActRIIa-mFc. Vehicle-treated control ovariectomized mice exhibited bone densities that were comparable to vehicle-treated control sham operated mice (FIG. 15). In addition to bone density, bone content increased following ActRIIa-mFC treatment. Ex vivo pQCT analysis of the femoral midshaft demonstrated a significant increase in both total and cortical bone content after twelve weeks of treatment with ActRIIa-mFc while both ovariectomized and sham operated vehicle control-treated mice exhibited comparable bone content (FIG. 16). Ex vivo pQCT analysis of the femoral midshaft also showed that ActRIIa-mFc treated mice did not show a change in periosteal circumference; however ActRIIa-mFc treatment resulted in a decrease in endosteal circumference indicating an increase in cortical thickness due to growth on the inner surface of the femur (FIG. 17).
[0180] Mechanical testing of femurs determined that ActRIIa-mFc was able to increase the extrinsic characteristics of the bone (maximal load, stiffness and energy to break) which contributed to a significant increase in the intrinsic properties (ultimate strength) of the bones. Ovariectomized mice treated with ActRIIa-mFc exhibited increased bone strength to levels beyond sham operated, vehicle treated controls, indicating a complete reversal of the osteoporotic phenotype (FIG. 18).
[0181] These data demonstrate that an activin-ActRIIa antagonist can increase bone density in normal female mice and, furthermore, correct defects in bone density, bone content, and ultimately bone strength, in a mouse model of osteoporosis.
[0182] In a further set of experiments, mice were ovariectomized or sham operated at 4 weeks, and beginning at 12 weeks received either placebo or ActRIIa-mFc (2 times/week, 10 mg/kg) (also referred to as RAP-11 in FIGS. 19-24), for a further period of 12 weeks. A variety of bone parameters were evaluated. As shown in FIG. 19, ActRIIa-mFc increased vertebral trabecular bone volume to total volume ratios (BV/TV) in both the OVX and SHAM operated mice. ActRIIa-mFc also improved the trabecular architecture (FIG. 20), increased cortical thickness (FIG. 21) and improved bone strength (FIG. 22). As shown in FIG. 23, ActRIIa-mFc produced desirable effects at a range of doses from 1 mg/kg to 10 mg/kg.
[0183] Bone histomorphometry was conducted at a 2 week time point in sham operated mice. These data, presented in FIG. 24, demonstrate that ActRIIa-mFc has a dual effect, both inhibiting bone resorption and promoting bone growth. Thus ActRIIa-mFc stimulates bone growth (anabolic effect) and inhibits bone resorption (anti-catabolic effect).
Example 4
Alternative ActRIIa-Fc Proteins
[0184] An alternative construct may have a deletion of the C-terminal tail (the final 15 amino acids of the extracellular domain of ActRIIa. The sequence for such a construct is presented below (Fc portion underlined) (SEQ ID NO: 12):
TABLE-US-00011 ILGRSETQECLFFNANWEKDRTNQTGVEPCYGDKDKRRHCFATWKNISGS IEIVKQGCWLDDINCYDRTDCVEKKDSPEVYFCCCEGNMCNEKFSYFPEM TGGGTHTCPPCPAPELLGGPSVFLEPPKPKDTLMISRTPEVTCVVVDVSH EDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKE YKCKVSNKALPVPIEKTISKAKGQPREPQVYTLPPSREEMTKNQVSLTCL VKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQ QGNVFSCSVMHEALHNHYTQKSLSLSPGK
Example 5
Anti-Activin A Antibodies Promote Osteoblast Differentiation
[0185] Osteoblast mineralization of normal human osteoblast cells. Normal human osteoblast (NHOst) cells (Lonza Walkersville) were cultured in Minimal Essential Medium Alpha (MEM-.alpha.) without phenol red (Invitrogen), 10% fetal bovine serum (FBS), 5 units penicillin-streptomycin and 0.25 .mu.g/ml amphotericin B. Mineralization experiments were performed in 12 well plates seeded at 2.5.times.10.sup.4 cells/well using cells between passages 3 and 6. Mineralization media was the same as above except for the addition of 2% heat-inactivated, charcoal-stripped FBS, 20 mM HEPES and 1.8 mM CaCl.sub.2. Media was supplemented with 100 nM dexamethasone and 10 nM .beta.-glycerol phosphate (Dex .beta.GP).+-.50 ng/ml activin A or .+-.10 ug/ml anti-activin A (Anti-ActA; Cat No. MAB3381, R&D Systems). Differentiation media was changed every 2-3 days over a 14 day period. NHOst cells were analyzed for bone mineral deposition using an osteogenesis quantitation kit (Chemicon). Cells were rinsed with PBS, fixed in formalin free fixative and stained with alizarin red.
[0186] As shown in FIG. 25, Activin A blocks mineralization by NHOst osteoblast precursor cells. This inhibition is rescued by the addition of anti-activin A antibody thus indicating that blocking activin A signaling increases/restores osteoblast matrix mineralization.
Example 6
Anti-Activin A Antibodies Inhibit Osteoclast Differentiation
[0187] Osteoclast differentiation and TRAP ELISA. The mouse macrophage cell line RAW264.7 (ATCC TIB-71) was cultured in Dulbecco's Modified Eagle Medium high glucose 1.times. (Gibco), 10% FBS, pen-strep, and amphotericin B. Osteoclast differentiation experiments were performed in 12 well plates seeded at 4.times.10.sup.4 cells/well in MEM-.alpha. without phenol red, and 10% charcoal-stripped FBS. Media was supplemented with 20 ng/ml receptor activator of nuclear factor kappa B ligand (RANKL), 30 ng/ml macrophage colony-stimulating factor (MCSF), .+-.20 ng/ml activin A, or .+-.10 ug/ml anti-activin A (Anti-ActA; Cat No. MAB3381, R&D Systems). Differentiation media was changed every 2-3 days over the 12 day assay period. Media was collected for MouseTRAP.TM. assay (Immunodiagnostics Systems) which is a solid phase immunofixed enzyme activity assay for the determination of mouse tartrate-resistant acid phosphatase form 5b (TRAP).
[0188] Bone marrow derived osteoclast differentiation and TRAP ELISA. Male C57BL/6NTac mice were euthanized and bone marrow cells were isolated from the long bones of the hindleg (tibia and fibula). 5.times.10.sup.4 cells/ml were cultured and tested as the RAW264.7 cells above. Media was supplemented with 10 ng/ml RANKL, 30 ng/ml MCSF, .+-.50 ng/ml activin A, or .+-.10 ug/ml anti-activin A (Anti-ActA; Cat No. MAB3381, R&D Systems) and collected for mouse TRAP assay.
[0189] RANKL/MCSF was used to differentiate RAW264.7 and bone marrow derived cells into osteoclast cells. As shown in FIG. 26, the addition of activin A significantly increases TRAP secretion, and by correlation, osteoclast cell number. This effect of activin A is reversed by anti-activin A antibody, thus indicating that inhibition of activin A signaling prevents osteoclast differentiation.
Example 7
ActRIIa-Fc Inhibits Activin B and Promotes Osteoblast Differentiation
[0190] Osteoblast mineralization of normal human osteoblast cells. Normal human osteoblast (NHOst) cells (Lonza Walkersville) were cultured in Minimal Essential Medium Alpha (MEM-.alpha.) without phenol red (Invitrogen), 10% fetal bovine serum (SAFC Biosciences), 5 units penicillin-streptomycin, and 0.25 .mu.g/ml amphotericin B. Mineralization experiments were performed in 12-well plates seeded at 5.times.10.sup.4 cells/well using cells between passages 3 and 6. Mineralization media was the same as above except for the addition of 10% heat-inactivated, charcoal-stripped fetal bovine serum, 20 mM HEPES, and 1.8 mM CaCl.sub.2. Media was supplemented with 100 nM dexamethasone and 10 nM .beta.-glycerol phosphate (Dex .beta.GP).+-.50 ng/ml activin B (R&D Systems) and .+-.20 .mu.g/ml ActRIIa-mFc. Differentiation media was changed every 2-3 days over a 14-day period. NHOst cells were analyzed for bone mineral deposition using an osteogenesis quantitation kit (Chemicon). Cells were rinsed with PBS, fixed in formalin-free fixative, and stained with alizarin red.
[0191] As shown in FIG. 27, activin B blocks differentiation/mineralization by NHOst osteoblast precursor cells. This inhibition is rescued by the addition of ActRIIa-mFc, thus indicating that blocking activin B signaling increases/restores osteoblast matrix mineralization.
Example 8
Anti-Activin A Antibodies Inhibit Osteoclast Differentiation
[0192] Osteoclast differentiation and TRAP ELISA. The mouse macrophage cell line RAW264.7 (ATCC TIB-71) was cultured in Dulbecco's Modified Eagle Medium high glucose 1.times. (Gibco), 10% fetal bovine serum, 5 units penicillin-streptomycin, and 0.25 .mu.g/ml amphotericin B. Osteoclast differentiation experiments were performed in 12-well plates seeded at 4.times.10.sup.4 cells/well in MEM-.alpha. without phenol red, 10% charcoal-stripped fetal bovine serum, 5 units penicillin-streptomycin, and 0.25 .mu.g/ml amphotericin B. Media was supplemented with 20 ng/ml receptor activator of nuclear factor kappa B ligand (RANKL, R&D Systems), 30 ng/ml macrophage colony-stimulating factor (MCSF, R&D Systems), .+-.50 ng/ml activin B, and .+-.10 .mu.g/ml anti-activin A antibody (Anti-ActA; Cat No. MAB3381, R&D Systems). Differentiation media was changed every 2-3 days over the 12-day assay period. Media was collected for MouseTRAP.TM. assay (Immunodiagnostics Systems) which is a solid phase immunofixed enzyme activity assay for the determination of mouse tartrate-resistant acid phosphatase form 5b (TRAP 5b).
[0193] RANKL/MCSF was used to differentiate RAW264.7 precursor cells into osteoclast cells. As shown in FIG. 28, the addition of activin B significantly increases TRAP 5b secretion, and by correlation, osteoclast cell number. This effect of activin B is reversed by ActRIIa-mFc, thus indicating that blocking activin B signaling prevents osteoclast differentiation. In contrast, anti-activin A antibody is unable to reverse the TRAP 5b effect of activin B.
INCORPORATION BY REFERENCE
[0194] All publications and patents mentioned herein are hereby incorporated by reference in their entirety as if each individual publication or patent was specifically and individually indicated to be incorporated by reference.
[0195] While specific embodiments of the subject matter have been discussed, the above specification is illustrative and not restrictive. Many variations will become apparent to those skilled in the art upon review of this specification and the claims below. The full scope of the invention should be determined by reference to the claims, along with their full scope of equivalents, and the specification, along with such variations.
Sequence CWU 1
1
181513PRTHomo sapiens 1Met Gly Ala Ala Ala Lys Leu Ala Phe Ala Val
Phe Leu Ile Ser Cys1 5 10 15Ser Ser Gly Ala Ile Leu Gly Arg Ser Glu
Thr Gln Glu Cys Leu Phe 20 25 30Phe Asn Ala Asn Trp Glu Lys Asp Arg
Thr Asn Gln Thr Gly Val Glu 35 40 45Pro Cys Tyr Gly Asp Lys Asp Lys
Arg Arg His Cys Phe Ala Thr Trp 50 55 60Lys Asn Ile Ser Gly Ser Ile
Glu Ile Val Lys Gln Gly Cys Trp Leu65 70 75 80Asp Asp Ile Asn Cys
Tyr Asp Arg Thr Asp Cys Val Glu Lys Lys Asp 85 90 95Ser Pro Glu Val
Tyr Phe Cys Cys Cys Glu Gly Asn Met Cys Asn Glu 100 105 110Lys Phe
Ser Tyr Phe Pro Glu Met Glu Val Thr Gln Pro Thr Ser Asn 115 120
125Pro Val Thr Pro Lys Pro Pro Tyr Tyr Asn Ile Leu Leu Tyr Ser Leu
130 135 140Val Pro Leu Met Leu Ile Ala Gly Ile Val Ile Cys Ala Phe
Trp Val145 150 155 160Tyr Arg His His Lys Met Ala Tyr Pro Pro Val
Leu Val Pro Thr Gln 165 170 175Asp Pro Gly Pro Pro Pro Pro Ser Pro
Leu Leu Gly Leu Lys Pro Leu 180 185 190Gln Leu Leu Glu Val Lys Ala
Arg Gly Arg Phe Gly Cys Val Trp Lys 195 200 205Ala Gln Leu Leu Asn
Glu Tyr Val Ala Val Lys Ile Phe Pro Ile Gln 210 215 220Asp Lys Gln
Ser Trp Gln Asn Glu Tyr Glu Val Tyr Ser Leu Pro Gly225 230 235
240Met Lys His Glu Asn Ile Leu Gln Phe Ile Gly Ala Glu Lys Arg Gly
245 250 255Thr Ser Val Asp Val Asp Leu Trp Leu Ile Thr Ala Phe His
Glu Lys 260 265 270Gly Ser Leu Ser Asp Phe Leu Lys Ala Asn Val Val
Ser Trp Asn Glu 275 280 285Leu Cys His Ile Ala Glu Thr Met Ala Arg
Gly Leu Ala Tyr Leu His 290 295 300Glu Asp Ile Pro Gly Leu Lys Asp
Gly His Lys Pro Ala Ile Ser His305 310 315 320Arg Asp Ile Lys Ser
Lys Asn Val Leu Leu Lys Asn Asn Leu Thr Ala 325 330 335Cys Ile Ala
Asp Phe Gly Leu Ala Leu Lys Phe Glu Ala Gly Lys Ser 340 345 350Ala
Gly Asp Thr His Gly Gln Val Gly Thr Arg Arg Tyr Met Ala Pro 355 360
365Glu Val Leu Glu Gly Ala Ile Asn Phe Gln Arg Asp Ala Phe Leu Arg
370 375 380Ile Asp Met Tyr Ala Met Gly Leu Val Leu Trp Glu Leu Ala
Ser Arg385 390 395 400Cys Thr Ala Ala Asp Gly Pro Val Asp Glu Tyr
Met Leu Pro Phe Glu 405 410 415Glu Glu Ile Gly Gln His Pro Ser Leu
Glu Asp Met Gln Glu Val Val 420 425 430Val His Lys Lys Lys Arg Pro
Val Leu Arg Asp Tyr Trp Gln Lys His 435 440 445Ala Gly Met Ala Met
Leu Cys Glu Thr Ile Glu Glu Cys Trp Asp His 450 455 460Asp Ala Glu
Ala Arg Leu Ser Ala Gly Cys Val Gly Glu Arg Ile Thr465 470 475
480Gln Met Gln Arg Leu Thr Asn Ile Ile Thr Thr Glu Asp Ile Val Thr
485 490 495Val Val Thr Met Val Thr Asn Val Asp Phe Pro Pro Lys Glu
Ser Ser 500 505 510Leu2115PRTHomo sapiens 2Ile Leu Gly Arg Ser Glu
Thr Gln Glu Cys Leu Phe Phe Asn Ala Asn1 5 10 15Trp Glu Lys Asp Arg
Thr Asn Gln Thr Gly Val Glu Pro Cys Tyr Gly 20 25 30Asp Lys Asp Lys
Arg Arg His Cys Phe Ala Thr Trp Lys Asn Ile Ser 35 40 45Gly Ser Ile
Glu Ile Val Lys Gln Gly Cys Trp Leu Asp Asp Ile Asn 50 55 60Cys Tyr
Asp Arg Thr Asp Cys Val Glu Lys Lys Asp Ser Pro Glu Val65 70 75
80Tyr Phe Cys Cys Cys Glu Gly Asn Met Cys Asn Glu Lys Phe Ser Tyr
85 90 95Phe Pro Glu Met Glu Val Thr Gln Pro Thr Ser Asn Pro Val Thr
Pro 100 105 110Lys Pro Pro 1153100PRTHomo sapiens 3Ile Leu Gly Arg
Ser Glu Thr Gln Glu Cys Leu Phe Phe Asn Ala Asn1 5 10 15Trp Glu Lys
Asp Arg Thr Asn Gln Thr Gly Val Glu Pro Cys Tyr Gly 20 25 30Asp Lys
Asp Lys Arg Arg His Cys Phe Ala Thr Trp Lys Asn Ile Ser 35 40 45Gly
Ser Ile Glu Ile Val Lys Gln Gly Cys Trp Leu Asp Asp Ile Asn 50 55
60Cys Tyr Asp Arg Thr Asp Cys Val Glu Lys Lys Asp Ser Pro Glu Val65
70 75 80Tyr Phe Cys Cys Cys Glu Gly Asn Met Cys Asn Glu Lys Phe Ser
Tyr 85 90 95Phe Pro Glu Met 10041542DNAHomo sapiens 4atgggagctg
ctgcaaagtt ggcgtttgcc gtctttctta tctcctgttc ttcaggtgct 60atacttggta
gatcagaaac tcaggagtgt cttttcttta atgctaattg ggaaaaagac
120agaaccaatc aaactggtgt tgaaccgtgt tatggtgaca aagataaacg
gcggcattgt 180tttgctacct ggaagaatat ttctggttcc attgaaatag
tgaaacaagg ttgttggctg 240gatgatatca actgctatga caggactgat
tgtgtagaaa aaaaagacag ccctgaagta 300tatttttgtt gctgtgaggg
caatatgtgt aatgaaaagt tttcttattt tccagagatg 360gaagtcacac
agcccacttc aaatccagtt acacctaagc caccctatta caacatcctg
420ctctattcct tggtgccact tatgttaatt gcggggattg tcatttgtgc
attttgggtg 480tacaggcatc acaagatggc ctaccctcct gtacttgttc
caactcaaga cccaggacca 540cccccacctt ctccattact agggttgaaa
ccactgcagt tattagaagt gaaagcaagg 600ggaagatttg gttgtgtctg
gaaagcccag ttgcttaacg aatatgtggc tgtcaaaata 660tttccaatac
aggacaaaca gtcatggcaa aatgaatacg aagtctacag tttgcctgga
720atgaagcatg agaacatatt acagttcatt ggtgcagaaa aacgaggcac
cagtgttgat 780gtggatcttt ggctgatcac agcatttcat gaaaagggtt
cactatcaga ctttcttaag 840gctaatgtgg tctcttggaa tgaactgtgt
catattgcag aaaccatggc tagaggattg 900gcatatttac atgaggatat
acctggccta aaagatggcc acaaacctgc catatctcac 960agggacatca
aaagtaaaaa tgtgctgttg aaaaacaacc tgacagcttg cattgctgac
1020tttgggttgg ccttaaaatt tgaggctggc aagtctgcag gcgataccca
tggacaggtt 1080ggtacccgga ggtacatggc tccagaggta ttagagggtg
ctataaactt ccaaagggat 1140gcatttttga ggatagatat gtatgccatg
ggattagtcc tatgggaact ggcttctcgc 1200tgtactgctg cagatggacc
tgtagatgaa tacatgttgc catttgagga ggaaattggc 1260cagcatccat
ctcttgaaga catgcaggaa gttgttgtgc ataaaaaaaa gaggcctgtt
1320ttaagagatt attggcagaa acatgctgga atggcaatgc tctgtgaaac
cattgaagaa 1380tgttgggatc acgacgcaga agccaggtta tcagctggat
gtgtaggtga aagaattacc 1440cagatgcaga gactaacaaa tattattacc
acagaggaca ttgtaacagt ggtcacaatg 1500gtgacaaatg ttgactttcc
tcccaaagaa tctagtctat ga 15425345DNAHomo sapiens 5atacttggta
gatcagaaac tcaggagtgt cttttcttta atgctaattg ggaaaaagac 60agaaccaatc
aaactggtgt tgaaccgtgt tatggtgaca aagataaacg gcggcattgt
120tttgctacct ggaagaatat ttctggttcc attgaaatag tgaaacaagg
ttgttggctg 180gatgatatca actgctatga caggactgat tgtgtagaaa
aaaaagacag ccctgaagta 240tatttttgtt gctgtgaggg caatatgtgt
aatgaaaagt tttcttattt tccagagatg 300gaagtcacac agcccacttc
aaatccagtt acacctaagc caccc 3456225PRTArtificial
SequenceDescription of Artificial Sequence Synthetic
polypeptideMOD_RES(43)..(43)Asp or AlaMOD_RES(100)..(100)Lys or
AlaMOD_RES(212)..(212)Asn or Ala 6Thr His Thr Cys Pro Pro Cys Pro
Ala Pro Glu Leu Leu Gly Gly Pro1 5 10 15Ser Val Phe Leu Phe Pro Pro
Lys Pro Lys Asp Thr Leu Met Ile Ser 20 25 30Arg Thr Pro Glu Val Thr
Cys Val Val Val Xaa Val Ser His Glu Asp 35 40 45Pro Glu Val Lys Phe
Asn Trp Tyr Val Asp Gly Val Glu Val His Asn 50 55 60Ala Lys Thr Lys
Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val65 70 75 80Val Ser
Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly Lys Glu 85 90 95Tyr
Lys Cys Xaa Val Ser Asn Lys Ala Leu Pro Val Pro Ile Glu Lys 100 105
110Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val Tyr Thr
115 120 125Leu Pro Pro Ser Arg Glu Glu Met Thr Lys Asn Gln Val Ser
Leu Thr 130 135 140Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala
Val Glu Trp Glu145 150 155 160Ser Asn Gly Gln Pro Glu Asn Asn Tyr
Lys Thr Thr Pro Pro Val Leu 165 170 175Asp Ser Asp Gly Pro Phe Phe
Leu Tyr Ser Lys Leu Thr Val Asp Lys 180 185 190Ser Arg Trp Gln Gln
Gly Asn Val Phe Ser Cys Ser Val Met His Glu 195 200 205Ala Leu His
Xaa His Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly 210 215
220Lys2257344PRTArtificial SequenceDescription of Artificial
Sequence Synthetic polypeptide 7Ile Leu Gly Arg Ser Glu Thr Gln Glu
Cys Leu Phe Phe Asn Ala Asn1 5 10 15Trp Glu Lys Asp Arg Thr Asn Gln
Thr Gly Val Glu Pro Cys Tyr Gly 20 25 30Asp Lys Asp Lys Arg Arg His
Cys Phe Ala Thr Trp Lys Asn Ile Ser 35 40 45Gly Ser Ile Glu Ile Val
Lys Gln Gly Cys Trp Leu Asp Asp Ile Asn 50 55 60Cys Tyr Asp Arg Thr
Asp Cys Val Glu Lys Lys Asp Ser Pro Glu Val65 70 75 80Tyr Phe Cys
Cys Cys Glu Gly Asn Met Cys Asn Glu Lys Phe Ser Tyr 85 90 95Phe Pro
Glu Met Glu Val Thr Gln Pro Thr Ser Asn Pro Val Thr Pro 100 105
110Lys Pro Pro Thr Gly Gly Gly Thr His Thr Cys Pro Pro Cys Pro Ala
115 120 125Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro
Lys Pro 130 135 140Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val
Thr Cys Val Val145 150 155 160Val Asp Val Ser His Glu Asp Pro Glu
Val Lys Phe Asn Trp Tyr Val 165 170 175Asp Gly Val Glu Val His Asn
Ala Lys Thr Lys Pro Arg Glu Glu Gln 180 185 190Tyr Asn Ser Thr Tyr
Arg Val Val Ser Val Leu Thr Val Leu His Gln 195 200 205Asp Trp Leu
Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn Lys Ala 210 215 220Leu
Pro Val Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro225 230
235 240Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Glu Glu Met
Thr 245 250 255Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe
Tyr Pro Ser 260 265 270Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln
Pro Glu Asn Asn Tyr 275 280 285Lys Thr Thr Pro Pro Val Leu Asp Ser
Asp Gly Ser Phe Phe Leu Tyr 290 295 300Ser Lys Leu Thr Val Asp Lys
Ser Arg Trp Gln Gln Gly Asn Val Phe305 310 315 320Ser Cys Ser Val
Met His Glu Ala Leu His Asn His Tyr Thr Gln Lys 325 330 335Ser Leu
Ser Leu Ser Pro Gly Lys 340821PRTApis mellifera 8Met Lys Phe Leu
Val Asn Val Ala Leu Val Phe Met Val Val Tyr Ile1 5 10 15Ser Tyr Ile
Tyr Ala 20922PRTUnknownDescription of Unknown Tissue plasminogen
activator peptide 9Met Asp Ala Met Lys Arg Gly Leu Cys Cys Val Leu
Leu Leu Cys Gly1 5 10 15Ala Val Phe Val Ser Pro
201020PRTUnknownDescription of Unknown Native peptide 10Met Gly Ala
Ala Ala Lys Leu Ala Phe Ala Val Phe Leu Ile Ser Cys1 5 10 15Ser Ser
Gly Ala 20119PRTArtificial SequenceDescription of Artificial
Sequence Synthetic peptide 11Ile Leu Gly Arg Ser Glu Thr Gln Glu1
512329PRTArtificial SequenceDescription of Artificial Sequence
Synthetic polypeptide 12Ile Leu Gly Arg Ser Glu Thr Gln Glu Cys Leu
Phe Phe Asn Ala Asn1 5 10 15Trp Glu Lys Asp Arg Thr Asn Gln Thr Gly
Val Glu Pro Cys Tyr Gly 20 25 30Asp Lys Asp Lys Arg Arg His Cys Phe
Ala Thr Trp Lys Asn Ile Ser 35 40 45Gly Ser Ile Glu Ile Val Lys Gln
Gly Cys Trp Leu Asp Asp Ile Asn 50 55 60Cys Tyr Asp Arg Thr Asp Cys
Val Glu Lys Lys Asp Ser Pro Glu Val65 70 75 80Tyr Phe Cys Cys Cys
Glu Gly Asn Met Cys Asn Glu Lys Phe Ser Tyr 85 90 95Phe Pro Glu Met
Thr Gly Gly Gly Thr His Thr Cys Pro Pro Cys Pro 100 105 110Ala Pro
Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro Lys 115 120
125Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys Val
130 135 140Val Val Asp Val Ser His Glu Asp Pro Glu Val Lys Phe Asn
Trp Tyr145 150 155 160Val Asp Gly Val Glu Val His Asn Ala Lys Thr
Lys Pro Arg Glu Glu 165 170 175Gln Tyr Asn Ser Thr Tyr Arg Val Val
Ser Val Leu Thr Val Leu His 180 185 190Gln Asp Trp Leu Asn Gly Lys
Glu Tyr Lys Cys Lys Val Ser Asn Lys 195 200 205Ala Leu Pro Val Pro
Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln 210 215 220Pro Arg Glu
Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Glu Glu Met225 230 235
240Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr Pro
245 250 255Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu
Asn Asn 260 265 270Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly
Ser Phe Phe Leu 275 280 285Tyr Ser Lys Leu Thr Val Asp Lys Ser Arg
Trp Gln Gln Gly Asn Val 290 295 300Phe Ser Cys Ser Val Met His Glu
Ala Leu His Asn His Tyr Thr Gln305 310 315 320Lys Ser Leu Ser Leu
Ser Pro Gly Lys 32513369PRTArtificial SequenceDescription of
Artificial Sequence Synthetic polypeptide 13Met Asp Ala Met Lys Arg
Gly Leu Cys Cys Val Leu Leu Leu Cys Gly1 5 10 15Ala Val Phe Val Ser
Pro Gly Ala Ala Ile Leu Gly Arg Ser Glu Thr 20 25 30Gln Glu Cys Leu
Phe Phe Asn Ala Asn Trp Glu Lys Asp Arg Thr Asn 35 40 45Gln Thr Gly
Val Glu Pro Cys Tyr Gly Asp Lys Asp Lys Arg Arg His 50 55 60Cys Phe
Ala Thr Trp Lys Asn Ile Ser Gly Ser Ile Glu Ile Val Lys65 70 75
80Gln Gly Cys Trp Leu Asp Asp Ile Asn Cys Tyr Asp Arg Thr Asp Cys
85 90 95Val Glu Lys Lys Asp Ser Pro Glu Val Tyr Phe Cys Cys Cys Glu
Gly 100 105 110Asn Met Cys Asn Glu Lys Phe Ser Tyr Phe Pro Glu Met
Glu Val Thr 115 120 125Gln Pro Thr Ser Asn Pro Val Thr Pro Lys Pro
Pro Thr Gly Gly Gly 130 135 140Thr His Thr Cys Pro Pro Cys Pro Ala
Pro Glu Leu Leu Gly Gly Pro145 150 155 160Ser Val Phe Leu Phe Pro
Pro Lys Pro Lys Asp Thr Leu Met Ile Ser 165 170 175Arg Thr Pro Glu
Val Thr Cys Val Val Val Asp Val Ser His Glu Asp 180 185 190Pro Glu
Val Lys Phe Asn Trp Tyr Val Asp Gly Val Glu Val His Asn 195 200
205Ala Lys Thr Lys Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val
210 215 220Val Ser Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly
Lys Glu225 230 235 240Tyr Lys Cys Lys Val Ser Asn Lys Ala Leu Pro
Val Pro Ile Glu Lys 245 250 255Thr Ile Ser Lys Ala Lys Gly Gln Pro
Arg Glu Pro Gln Val Tyr Thr 260 265 270Leu Pro Pro Ser Arg Glu Glu
Met Thr Lys Asn Gln Val Ser Leu Thr 275 280 285Cys Leu Val Lys Gly
Phe Tyr Pro Ser Asp Ile Ala Val Glu Trp Glu 290 295 300Ser Asn Gly
Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu305 310 315
320Asp Ser Asp Gly Ser Phe Phe Leu Tyr Ser Lys Leu Thr Val Asp Lys
325 330 335Ser Arg Trp Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met
His Glu 340 345 350Ala Leu His Asn His Tyr Thr Gln Lys Ser Leu
Ser Leu Ser Pro Gly 355 360 365Lys141114DNAArtificial
SequenceDescription of Artificial Sequence Synthetic polynucleotide
14atggatgcaa tgaagagagg gctctgctgt gtgctgctgc tgtgtggagc agtcttcgtt
60tcgcccggcg ccgctatact tggtagatca gaaactcagg agtgtctttt tttaatgcta
120attgggaaaa agacagaacc aatcaaactg gtgttgaacc gtgttatggt
gacaaagata 180aacggcggca ttgttttgct acctggaaga atatttctgg
ttccattgaa tagtgaaaca 240aggttgttgg ctggatgata tcaactgcta
tgacaggact gattgtgtag aaaaaaaaga 300cagccctgaa gtatatttct
gttgctgtga gggcaatatg tgtaatgaaa agttttctta 360ttttccggag
atggaagtca cacagcccac ttcaaatcca gttacaccta agccacccac
420cggtggtgga actcacacat gcccaccgtg cccagcacct gaactcctgg
ggggaccgtc 480agtcttcctc ttccccccaa aacccaagga caccctcatg
atctcccgga cccctgaggt 540cacatgcgtg gtggtggacg tgagccacga
agaccctgag gtcaagttca actggtacgt 600ggacggcgtg gaggtgcata
atgccaagac aaagccgcgg gaggagcagt acaacagcac 660gtaccgtgtg
gtcagcgtcc tcaccgtcct gcaccaggac tggctgaatg gcaaggagta
720caagtgcaag gtctccaaca aagccctccc agtccccatc gagaaaacca
tctccaaagc 780caaagggcag ccccgagaac cacaggtgta caccctgccc
ccatcccggg aggagatgac 840caagaaccag gtcagcctga cctgcctggt
caaaggcttc tatcccagcg acatcgccgt 900ggagtgggag agcaatgggc
agccggagaa caactacaag accacgcctc ccgtgctgga 960ctccgacggc
tccttcttcc tctatagcaa gctcaccgtg gacaagagca ggtggcagca
1020ggggaacgtc ttctcatgct ccgtgatgca tgaggctctg cacaaccact
acacgcagaa 1080gagcctctcc ctgtctccgg gtaaatgaga attc
1114155PRTArtificial SequenceDescription of Artificial Sequence
Synthetic peptide 15Thr Gly Gly Gly Gly1 5165PRTArtificial
SequenceDescription of Artificial Sequence Synthetic peptide 16Ser
Gly Gly Gly Gly1 5176PRTArtificial SequenceDescription of
Artificial Sequence Synthetic 6xHis tag 17His His His His His His1
518108DNAArtificial SequenceDescription of Artificial Sequence
Synthetic polynucleotide 18agccagacaa gccagacaag ccagacaagc
cagacaagcc agacaagcca gacaagccag 60acaagccaga caagccagac aagccagaca
agccagacaa gccagaca 108
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016
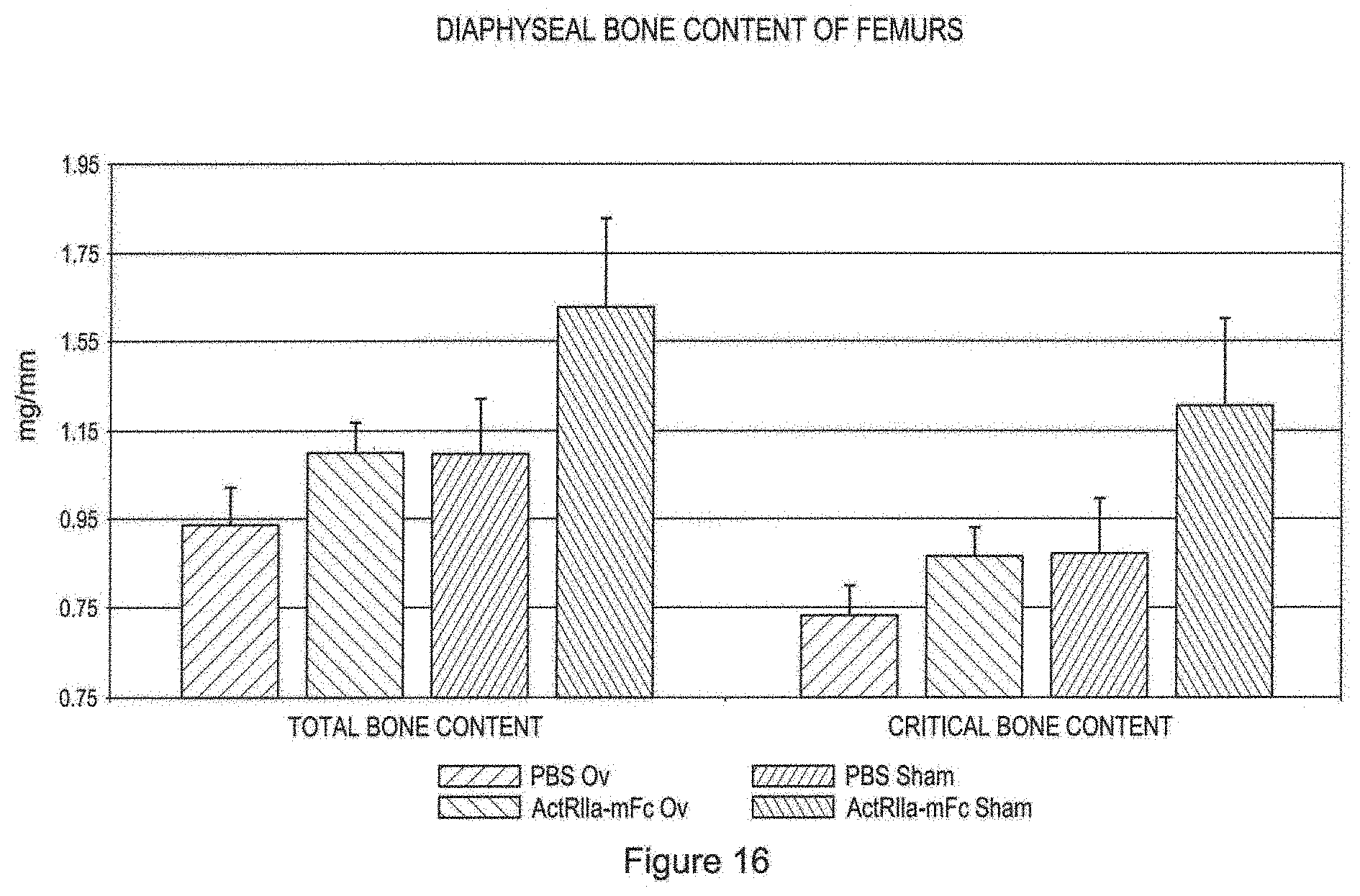
D00017

D00018

D00019

D00020
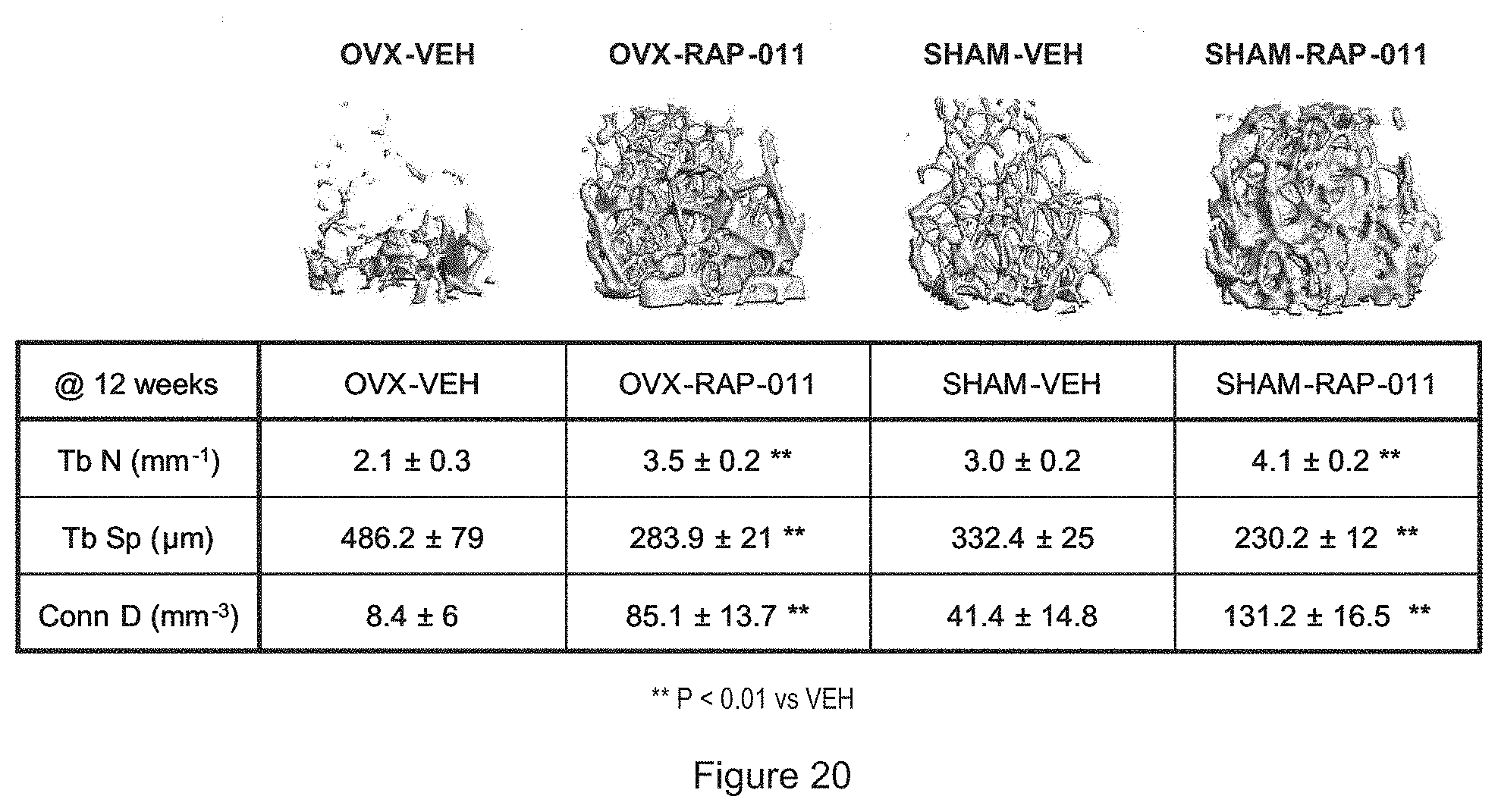
D00021

D00022

D00023

D00024

D00025

D00026

D00027

D00028

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.