Fecal Fungome And Therapeutic Efficacy Of Fecal Microbiota Transplantation
NG; Siew Chien ; et al.
U.S. patent application number 16/966853 was filed with the patent office on 2021-02-11 for fecal fungome and therapeutic efficacy of fecal microbiota transplantation. The applicant listed for this patent is The Chinese University of Hong Kong. Invention is credited to Ka Leung Francis CHAN, Siew Chien NG, Tao ZUO.
| Application Number | 20210038663 16/966853 |
| Document ID | / |
| Family ID | 1000005223560 |
| Filed Date | 2021-02-11 |











View All Diagrams
| United States Patent Application | 20210038663 |
| Kind Code | A1 |
| NG; Siew Chien ; et al. | February 11, 2021 |
FECAL FUNGOME AND THERAPEUTIC EFFICACY OF FECAL MICROBIOTA TRANSPLANTATION
Abstract
Methods are provided for identifying subjects as suitable donor or recipients for FMT, for assessing the likelihood of FMT treatment success, and for enhancing FMT treatment efficacy. Also provided are kits and compositions for FMT with enhanced efficacy.
| Inventors: | NG; Siew Chien; (Hong Kong, CN) ; ZUO; Tao; (Qingzhou, CN) ; CHAN; Ka Leung Francis; (Hong Kong, CN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005223560 | ||||||||||
| Appl. No.: | 16/966853 | ||||||||||
| Filed: | February 1, 2019 | ||||||||||
| PCT Filed: | February 1, 2019 | ||||||||||
| PCT NO: | PCT/CN2019/074353 | ||||||||||
| 371 Date: | July 31, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62625705 | Feb 2, 2018 | |||
| 62679417 | Jun 1, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C12R 1/725 20130101; A61P 1/12 20180101; C12N 1/20 20130101; A61K 36/064 20130101 |
| International Class: | A61K 36/064 20060101 A61K036/064; A61P 1/12 20060101 A61P001/12; C12N 1/20 20060101 C12N001/20 |
Claims
1. A method for assessing likelihood of effective fecal microbiota transplantation (FMT), comprising determining C. albicans level in a stool sample obtained from a potential recipient prior to FMT.
2. The method of claim 1, wherein the C. albicans level is a percentage relative abundance.
3. The method of claim 2, wherein the C. albicans level is greater than 10% and FMT is assessed as unlikely to be effective for the potential recipient.
4. The method of claim 3, wherein the recipient is administered an effective amount of an antifungal agent that suppresses C. albicans growth before FMT.
5. The method of claim 4, further comprising determining C. albicans level in a stool sample obtained from the recipient after FMT.
6. The method of claim 2, wherein the C. albicans level is no greater than 10% and FMT is assessed as likely to be effective for the potential recipient.
7. The method of claim 1, further comprising determining total fungal load in the stool sample.
8. The method of claim 7, further comprising performing FMT on the potential recipient.
9. The method of claim 1, wherein C. albicans level is determined in a first stool sample obtained from a first potential recipient prior to FMT and in a second stool sample obtained from a second potential recipient prior to FMT.
10. The method of claim 9, wherein the first potential recipient has a lower C. albicans level than the second potential recipient and is assessed to have a higher likelihood of effective FMT than the potential recipient.
11. The method of claim 9, wherein the second potential recipient is administered an effective amount of an antifungal agent that suppresses C. albicans growth before FMT.
12. The method of claim 1, wherein the potential recipient has inflammatory bowel disease (IBD) with concurrent Clostridium difficile infection (CDI).
13. The method of claim 12, wherein the C. albicans level is determined in stool samples taken from the recipient before and after FMT.
14. A method for identifying a suitable donor for FMT, comprising the step of determining C. albicans level in a stool sample obtained from a candidate.
15. The method of claim 14, wherein the C. albicans level is a percentage relative abundance.
16. The method of claim 15, wherein the C. albicans level is no greater than 0.1% and the candidate is identified as a suitable donor for FMT.
17. The method of claim 15, wherein the C. albicans level is greater than 10% and the candidate is identified as an unsuitable donor for FMT.
18. The method of claim 14, further comprising determining Saccharomyces level and Aspergillus level in the stool sample.
19. The method of claim 14, further comprising determining Escherichia level and Proteus level in the stool sample.
20. The method of claim 14, further comprising determining total fungal load in the stool sample.
21. A method for improving FMT efficacy, comprising administering to an FMT recipient prior to FMT an effective amount of an antifungal agent that suppresses C. albicans growth.
22. The method of claim 21, wherein C. albicans level is determined in a stool sample from the FMT recipient prior to administration of the antifungal agent.
23. The method of claim 21, wherein C. albicans level is determined in a stool sample from the FMT recipient after administration of the antifungal agent.
24. The method of claim 21, further comprising administering to the recipient prior to FMT an effective amount of an agent that reduces total fungal load in a stool sample taken from the recipient prior to FMT.
25. The method of claim 21, wherein the recipient has inflammatory bowel disease (IBD) with concurrent Clostridium difficile infection (CDI).
26. A kit comprising (1) a first composition comprising donor stool; and (2) a second composition comprising an effective amount of an antifungal agent that suppresses C. albicans growth.
27. The kit of claim 22, wherein the first composition comprises donor stool that has been dried, frozen, and placed in a capsule for oral ingestion.
28. The kit of claim 22, further comprising in the second composition or in a third composition an effective amount of an agent that reduces total fungal load.
29. (canceled)
Description
BACKGROUND OF THE INVENTION
[0001] This application claims priority to U.S. Provisional Patent Application No. 62/625,705, filed Feb. 2, 2018, and U.S. Provisional Patent Application No. 62/679,417, filed Jun. 1, 2018, the contents of both are hereby incorporated by reference in the entirety for all purposes.
BACKGROUND OF THE INVENTION
[0002] Clostridium difficile infection (CDI) is a symptomatic infection due to the spore-forming bacterium, Clostridium difficile. C. difficile infection is spread by bacterial spores found within feces. Risk factors for infection include antibiotic or proton pump inhibitors use, hospitalization, other health problems, and older age. Its symptoms including watery diarrhea, fever, nausea, and abdominal pain, CDI makes up about 20% of cases of antibiotic-associated diarrhea. About 453,000 cases C. difficile infection occurred in the United States in 2011, resulting in 29,000 deaths. Each year, C. difficile infections accounts for health care cost of approximately $1.5 billion. Globally, rates of C. difficile infection have increased between 2001 and 2016, typically with more women than men affected by the infections.
[0003] Fecal microbiota transplantation (FMT) is highly effective for the treatment of CDI, especially among patients suffering from recurrent CDI. Also known as stool transplant, FMT involves a process of transplanting fecal matter containing microorganism from a healthy individual into the gastrointestinal tract of a recipient. The goal of FMT is restoration of the gut microflora disrupted due to CDI by introducing (or re-introducing) healthy bacterial flora via various means of infusion of a healthy individual's stool, e.g., by colonoscopy, enema, orogastric tube, or by mouth in the form of a capsule containing freeze-dried material obtained from a healthy donor. Aside from CDI, FMT is increasingly being used to treat other intestinal and extra-intestinal diseases, including other gastrointestinal diseases, such as inflammatory bowel disease (IBD), antibiotic-resistant bacterial infection, diarrhea, constipation, irritable bowel syndrome, autism, depression, obesity, diabetes, alopecia, and the like. In addition, FMT has been used for treating certain neurological conditions, such as multiple sclerosis and Parkinson's Disease. Considering the prevalence of CDI and other conditions treatable by FMT in the human population and their significant economic implications, there exists an urgent need for developing new and improved methods for treating CDI and other disorders by FMT with enhanced efficacy. The present invention fulfills this and other related needs.
BRIEF SUMMARY OF THE INVENTION
[0004] The invention relates to novel methods and compositions useful for more effectively treating Clostridium difficile infection (CDI) and other diseases suitable by fecal microbiota transplantation (FMT) treatment. In particular, the present inventor discovered that, when elevated level of the yeast species Candida albicans is present in the gastrointestinal tract of an FMT recipient or in the stool of an FMT donor, therapeutic efficacy of FMT is negatively impacted. This finding allows the inventors to devise methods and compositions that can improve FMT effectiveness. Thus, in the first aspect, the present invention provides a novel method for assessing the likelihood of effective FMT. The method includes a step of determining C. albicans level in a stool sample obtained from a potential recipient prior to FMT is performed, i.e., before the recipient is to receive transplantation of a donor fecal material.
[0005] In some embodiments, the C. albicans level is a percentage relative abundance, or is expressed as a percentage over the total level of all fungal species in the sample. In some embodiments, when the C. albicans level is determined as greater than 10% of total fungi in the sample of the recipient, FMT is assessed as unlikely to be effective for the potential recipient. Under such a determination, the recipient in some cases will not receive FMT treatment but will receive another different therapy; in other cases, the recipient is administered an effective amount of an antifungal agent that suppresses C. albicans growth before FMT is performed. Optionally, after the administration of the antifungal agent, the C. albicans level in the recipient (e.g., in a recipient's stool sample) is again measured and determined to have been lowered before the recipient is transplanted with a composition containing donor fecal material. In some cases, C. albicans level is again determined in a stool sample obtained from the recipient after FMT.
[0006] In some embodiments, when the C. albicans level is no greater than 10% of total fungi in the sample, FMT is assessed as likely to be effective for the potential recipient. In some cases, the potential recipient is then immediately given FMT, without any further treatment or preparation such as administration of an antifungal agent in the effective amount. In some embodiments, the method further involves a step of determining total fungal load in the stool sample. A potential recipient whose total fungal load in his stool sample is found to be relatively lower than that of a second potential recipient is expected to have a higher likelihood of having a successful FMT than the second recipient. In some embodiments, multiple potential recipients of FMT are tested prior to FMT using this method for assessing their relative likelihood of success upon receiving FMT treatment. For instance, C. albicans level is determined in a first stool sample obtained from a first potential recipient prior to FMT, and in the meantime C. albicans level is determined in a second stool sample obtained from a second potential recipient prior to FMT. In some embodiments, when the first potential recipient has a lower C. albicans level than the second potential recipient and is therefore assessed to have a higher likelihood of effective FMT than the second potential recipient. In some embodiments, the second potential recipient is then administered an effective amount of an antifungal agent that suppresses C. albicans growth before FMT, whereas the first potential recipient may or may need antifungal agent treatment prior to FMT. In some embodiments, C. albicans level is determined by quantitative polymerase chain reaction (PCR). In some embodiments, the levels of all fungal species present in the sample is determined by the Internal transcribed spacer 2 (ITS2) sequencing. In some embodiments, the recipient or recipients suffer from inflammatory bowel disease (IBD) with concurrent Clostridium difficile infection (CDI). The C. albicans level in the stool may be determined before and after their FMT process. An elevated C. albicans level before or after FMT is indicative of a higher likelihood of poor outcome or ineffective FMT.
[0007] In a second aspect, the present invention provides a novel, improved method for identifying a suitable donor who would provide stool or fecal material for FMT. The method includes the step of determining C. albicans level in a stool sample obtained from a candidate who is being considered as a potential donor for FMT.
[0008] In some embodiments, the C. albicans level determined in this method is a percentage relative abundance. In some cases, when the C. albicans level is no greater than 0.1% of all fungi present in the sample, the candidate is identified as a suitable donor for FMT. Optionally, fecal matter such as stool is immediately collected from the candidate for use in FMT. In some cases, when the C. albicans level is greater than 0.1%, the candidate is deemed unsuitable as a donor for FMT. As a result, either no fecal matter is collected from the candidate at all; or fecal matter is collected for processing to be used in FMT after the candidate is given an effective amount of an antifungal agent that suppresses C. albicans growth and again tested to find a satisfactorily reduced C. albicans level in the stool sample (e.g., no greater than 0.1% of total fungi in the sample). In some embodiments, the stool sample of a candidate is tested for Saccharomyces level and Aspergillus level in addition to C. albicans level. In some embodiments, C. albicans level is determined by quantitative PCR. In some embodiments, the levels of all fungi present in the sample is determined by ITS2 sequencing. In some embodiments, the method includes the additional step of determining Escherichia level and Proteus level in the stool sample of a potential donor. Among multiple potential donors, one with a relatively high Escherichia level and a relatively low Proteus level is deemed a more suitable donor than one with a relatively low Escherichia level and/or a relatively high Proteus level. In some embodiments, the method further includes a step of determining the total fungal load in the stool sample taken from the potential donor. A potential donor whose total fungal load in his stool sample is found to be relatively lower than that of a second potential donor is expected to be a more desirable donor, i.e., provide a higher likelihood of a successful FMT, than the second donor.
[0009] In a third aspect, the present invention provides a method for improving FMT efficacy. The method includes the step of administering to an FMT recipient prior to FMT being performed an effective amount of an antifungal agent that suppresses C. albicans growth. In some embodiments of this method, C. albicans level is first determined in a stool sample from the FMT recipient prior to administration of the antifungal agent. In some embodiments, C. albicans level is determined in a stool sample from the FMT recipient after administration of the antifungal agent. In some embodiments, C. albicans level is determined by quantitative PCR. In some embodiments, the levels of all fungi present in the sample is determined by ITS2 sequencing. In some embodiments, the method further includes a step of administering to the recipient prior to FMT an effective amount of an agent (e.g., an anti-fungal agent, such as a broad-spectrum fungicide), which reduces total fungal load in a stool sample taken from the recipient prior to FMT. In some embodiments, the recipient is a patient suffering from inflammatory bowel disease (IBD) with concurrent Clostridium difficile infection (CDI).
[0010] In a fourth aspect, the present invention provides kits and compositions useful for enhanced FMT treatment with improved efficacy. In some embodiments, a kit for improving FMT efficacy includes a first composition comprising a donor stool material and a second composition comprising an effective amount of an antifungal agent capable of suppressing the growth of C. albicans. Typically, the first and second compositions are kept in two separate containers. In some embodiments, the first composition contains processed donor fetal matter and is formulated for FMT by direct transfer to the GI tract (e.g., via colonoscopy or via nasal intubation) or by oral ingestion. In some embodiments, the first composition comprises donor fecal matter further fortified with an additional and effective amount of one or more fungal species belonging to the genus Saccharomyces and/or the genus Aspergillus. In some embodiments, the second composition is formulated for administration of the antifungal agent (such as fluconazole) to the recipient by injection, oral ingestion, or rectal deposit. In some embodiments, the kit may further comprise, either in the second composition or in a third composition, an effective amount of an agent that reduces total fungal load. In the alternative, the kit may comprise a third composition, which comprises an effective amount of an agent that reduces total fungal load, but not the second composition. In some cases, the kit may further include printed user instructions.
[0011] Related compositions useful in FMT with improved efficacy may comprise (1) a donor stool material containing live fecal microorganisms and (2) an antifungal agent that specifically suppresses the growth or proliferation of C. albicans but exhibits no such suppressive or inhibitory effect against other fungal species. Instead of a broad-spectrum fungicide, such specific anti-C. albicans agent may be short polynucleotide in nature of (e.g., a small inhibitory RNA, microRNA, miniRNA, lncRNA, or an antisense oligonucleotide that is capable of disrupting the expression of a key gene in the life cycle of C. albicans) that is capable of specifically targeting the species only but not other closely related fungal species.
BRIEF DESCRIPTION OF THE DRAWINGS
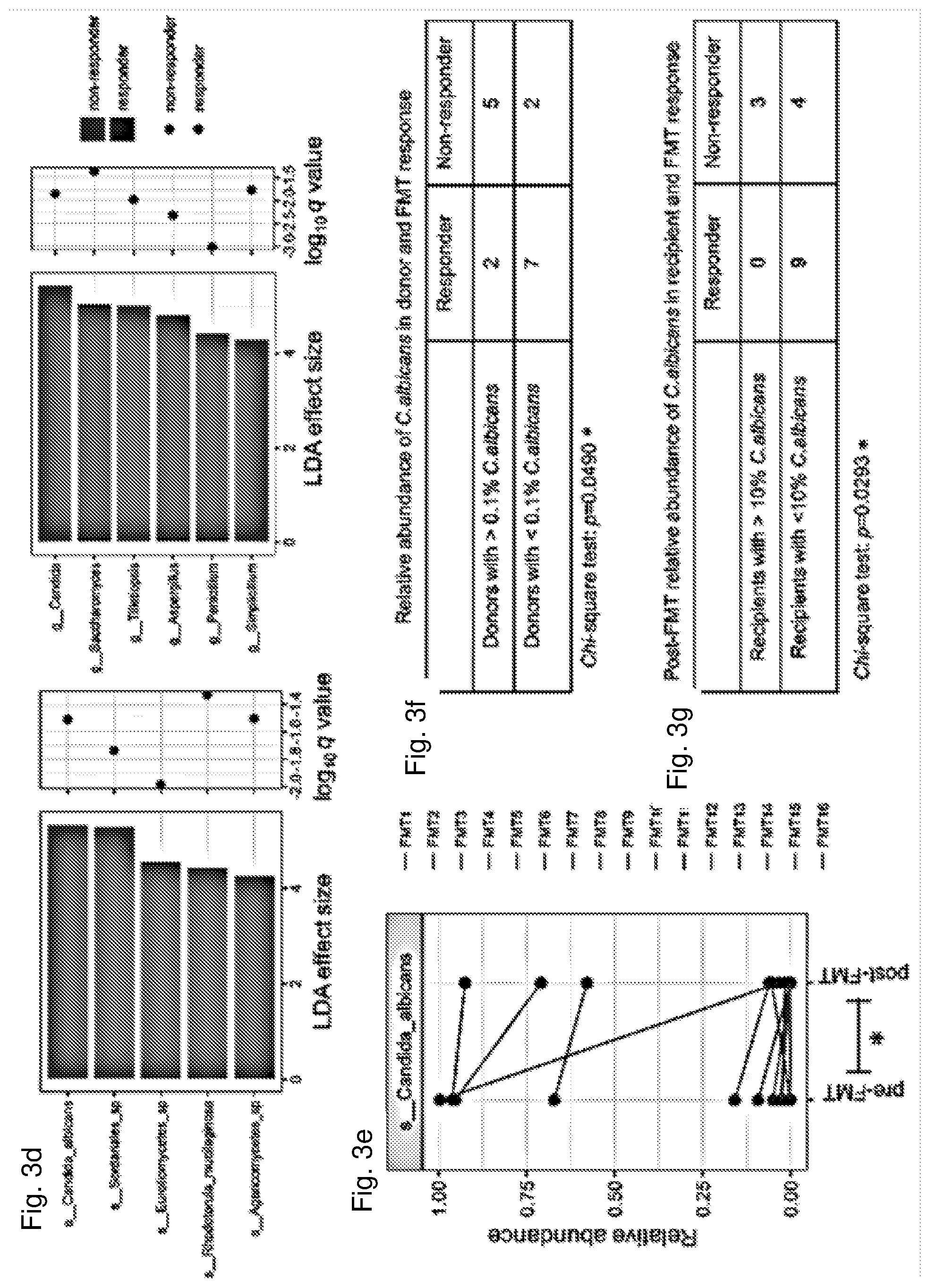
[0012] FIG. 1 Fungal alterations in CDI. (A) Comparison of the fecal mycobiota based on Shanon diversity, evenness, Chao1 richness in controls and CDI subjects. The bars are shown in median and interquartile range. The dots indicate individual values of the studied subjects. Statistical significance was determined by Mann-Whitney test, *P<0.05, **P<0.01. (B) Fungal community structure difference between controls and CDI by NMDS (Non-metric multidimensional scaling) plot based upon Bray-Curtis distance among all samples. (C) Comparison of the fecal mycobiota composition between controls and CDI subjects at the phylum level. (D) Differentially enriched fungal species between controls and CDI. Statistical significance was determined by LefSe analysis with FDR correction (only those species with q values<0.05 and LDA effect size>2 are shown). Heatmap of the presence of these differential fungal species is shown in relative abundance intensity. LDA effect size, q value (FDR-adjusted P value) and species annotation are shown. Green bars and dots indicate species enriched in controls, while red bar and dot indicate species enriched in CDI. (E) Comparison of the relative abundance of fecal C. albicans in controls and CDI subjects. The bars are shown in median and interquartile range. Statistical significance was determined by Mann-Whitney test, *P<0.05.
[0013] FIG. 2 Colonization of donor-derived fungal and bacterial taxa in FMT recipients in association with treatment response. (A) Presence of fungal operational taxonomic units (OTUs) in FMT recipients at the last follow-up. The color of the bar indicates the origin of the bacterial OTUs in the recipient. Purple indicates donor-derived OTUs colonized in the recipient, orange indicates OTUs exclusively present in recipient at baseline but not in donor at baseline, while green indicates OTUs present both in donor and in recipient before FMT. Comparison of the frequency of donor derived bacterial OTUs in FMT responders and in non-responders is shown. Statistical significance was determined by Mann-Whitney test, **P<0.01. (B) Presence of bacterial OTUs in FMT recipients at the last follow-up. Comparison of the frequency of donor-derived bacterial OTUs in FMT responders and in non-responders is shown. Statistical significance was determined by Mann-Whitney test, *P<0.05. (C) Heatmap of the abundance of differentially presented fungal genera in donor, pre-FMT and post-FMT last follow-up samples. Fungal genera with disparate presence between FMT responders and non-responders, as determined by LefSe analysis, are labeled with asterisk (genera with LDA effect size>2 and q value<0.01).
[0014] FIG. 3 Post-FMT alterations in the enteric mycobiota of CDI recipients in association with FMT response. Fecal fungal richness (A) and diversity (B) alterations in FMT recipients over the course of longitudinal follow-up and in their corresponding donors at baseline. Comparison of the fungal richness and diversity of pre-FMT samples and post-FMT samples collected at the last follow-up are shown in FMT responders and FMT non-responders respectively. Statistical significance was determined by paired Wilcox signed rank test, *P<0.05. "F" indicates FMT treated subject. "W" indicates weeks post treatment. (C) Alterations in the fecal fungal composition at the genus level in CDI recipients after FMT at different time points up to the last follow-up. (D) Differentially enriched fungal taxa across post-FMT fecal samples of FMT responders versus non-responders at the genus and species level respectively. Statistical significance level was determined by LefSe analysis with FDR correction (only those taxa with q values<0.05 and LDA effect size>2 are shown). Green bars and dots indicate taxa enriched in controls, while red bar and dot indicate taxa enriched in CDI. (E) Alterations of the relative abundance of fecal C. albicans after FMT at the last follow-up in FMT recipients. Statistical significance was determined by paired Wilcox signed rank test, *P<0.05. (F) Relative abundance of C. albicans in donor stool corresponding to FMT responders and non-responders. Statistical significance was determined by Chi-square test. (G) Relative abundance of C. albicans in stool of recipients before FMT in association with FMT response. Statistical significance was determined by Chi-square test.
[0015] FIG. 4 C. albicans compromises FMT efficacy in eradicating C. difficile infection in mice. A) Schematic diagram of C. albicans administration and stool infusion (FMT) in a murine C. difficile infection (CDI) model. Antibiotic treatment was ceased before gavage of C. albicans (CA) and C. difficile. B) Diarrhoea in mice on day 1 after stool infusion. C) Representative H&E-stained colonic sections on day 2 after stool infusion (shaded star denotes inflammatory cells infiltration, hallowed star denote ulceration, asterisk denotes oedema, arrow denotes goblet cell loss and triangle denotes herniated crypts). Scale bar, 150 .mu.m. n=5 mice per group. D) Enumeration of C. difficile in feces of mice on day 0 before FMT and day 1 post FMT (n=9 mice per group). Statistical significance represents comparisons between FMT-treated mice with C. difficile infection versus other groups by unpaired Mann-Whitney test. *P<0.05, **P<0.01. E) Enumeration of C. albicans in feces of mice both on day 0 before FMT and day 1 post FMT (n=9 mice per group). Statistical significance represents comparison between C. albicans load on day 0 before FMT and day 1 post FMT, by paired Mann-Whitney test. *P<0.05. Dot graphs show means.+-.s.e.m, performed at least two times independently.
[0016] FIG. 5 Quantification of fecal C. albicans levels in CDI subjects and healthy controls by qPCR. a, qPCR detection of C. albicans on CDI subjects and healthy controls from the discovery cohort. Comparison of the fecal C. albicans level between control and CDI was determined by Mann-Whitney test, ****P<0.0001. b, qPCR detection of C. albicans on an additional set of subjects (17 healthy individuals, 12 CDI subjects with and 12 without antibiotic use at inclusion). Statistical significance was tested by unpaired Mann-Whitney test. *P<0.05, **P<0.01. Graphs are shown in mean.+-.s.e.m. ND denotes no detectable C. albicans in the feces as determined by quantitative PCR.
[0017] FIG. 6 Longitudinal timeline of stool sample collection (expressed in weeks). "F" indicates FMT treated subject. "Donor" indicates FMT donor. "S" indicates subject treated with standard therapy (STD, vancomycin). "W" indicates weeks post treatment. Red dots indicate donor samples, green dots indicate FMT recipient samples sampled at different time points.
[0018] FIG. 7 Heatmap of the abundance of differentially presented bacterial genera in donor, pre-FMT and post-FMT last follow-up samples.
[0019] FIG. 8 Post-FMT alterations in the enteric bacterial microbiota of CDI recipients in association with FMT response. (A) Comparison of the fecal bacterial shanon diversity, evenness, chao1 richness in healthy controls and in CDI subjects. The bars are shown in median and interquartile range. The dots indicate individual values of the studied subjects. Statistical significance was determined by Mann-Whitney test, **P<0.01. Fecal bacterial richness (B) and diversity (C) alterations in FMT recipients over the course of longitudinal follow-up and in their corresponding donors at baseline. Comparison of the fungal richness and diversity of pre-FMT samples and post-FMT samples collected at the last follow-up are shown in FMT responders and FMT non-responders respectively. Statistical significance was determined by paired Wilcox signed rank test, *P<0.05. "F" indicates FMT treated subject. "W" indicates weeks post treatment.
[0020] FIG. 9 Post-antibiotic alterations in the enteric mycobiota of CDI subjects treated with vancomycin in association with treatment response. Fecal fungal richness (A) and diversity (B) alterations over the course of longitudinal follow-up in CDI subjects who received vancomycin treatment. "S" indicates CDI subject received vancomycin treatment (standerd therapy, STD). "W" indicates weeks post vancomycine treatment. (C) Frequencies of CDI individuals increased or decreased in fungal diversity and richness post treatment with respect to FMT and STD treatment. Statistical significance was determined by Chi-square test, *P<0.05. (D) Comparison of post-FMT fold change (FC) of the fecal fungal diversity relative to the pre-FMT sample in FMT responders and STD responders. Statistical significance was determined by Chi-square test, *P<0.05. (E) Comparison of post-FMT fold change (FC) of the fecal fungal richness relative to the pre-FMT sample in FMT responders and STD responders. Statistical significance was determined by Man-whitney test, *P<0.05. (G) Alterations in the fecal fungal composition at the genus level in CDI subjects on vancomycin regime at different time points up to the last follow-up.
[0021] FIG. 10 Fecal bacterial microbiota richness (A) and diversity (B) alterations in STD subjects over the course of longitudinal follow-up. "S" indicates vancomycin treated subject (STD treatment). "W" indicates weeks post treatment.
[0022] FIG. 11 Differentially enriched fungal taxa across post-treatment samples of FMT responders versus STD responders at the family, genus and species levels. Statistical significance level was determined by lefSe analysis with FDR correction (only those taxa with q values<0.05 and LDA effect size>2 are shown).
[0023] FIG. 12 Post-STD alterations of the relative abundance of fecal C. albicans at the last follow-up and at baseline in CDI subjects on vancomycin treatment. Statistical significance was determined by paired Wilcox signed rank test.
[0024] FIG. 13 Spearman correlation between fungal diversity, evenness, richness and bacterial diversity, evenness, richness, with respect to FMT and STD treatment and treatment response. Statistical significance was determined for all pairwise comparisons; significant correlations (P value<0.05) are displayed with asterisk. Blue circles and positive values indicate positive correlations, red circles and negative values indicate inverse correlations. The size and shading indicate the magnitude of the correlation where darker shades are more intensively correlated than lighter ones.
[0025] FIG. 14 Trans-kingdom interactions between bacteria and fungi. Spearman correlation plots of the relative abundance of fungal genera and bacterial genera identified to be significantly associated with CDI and controls at baseline, with respect to FMT and STD treatment and treatment response. Spearman correlation coefficients were calculated for all pairwise comparisons; Blue circles and positive values indicate positive correlations, red circles and negative values indicate inverse correlations. The size and shading indicate the magnitude of the correlation where darker shades are more intensively correlated than lighter ones. Statistical significance was determined for all pairwise comparisons; only correlations tested significant (P value<0.05) are displayed.
[0026] FIG. 15 Pre-FMT eradication of C. albicans in recipient mice restored FMT efficacy in clearing C. difficile infection. A) Schematic diagram of antifungal treatment and stool infusion (FMT) in a murine model with di-colonisation of C. albicans and C. difficile. Antifungal (fluconazole) treatment was ceased at day 4 after administration of C. albicans when C. albicans was determined negative by cultivation. "CCfF", mouse group with di-colonisation of C. albicans and C. difficile and treatments of fluconazole and FMT; "CCF", mouse group with di-colonisation of C. albicans and C. difficile and treatment of FMT; "CC", mouse group with di-colonisation of C. albicans and C. difficile. B) Enumeration of C. difficile in feces of mice on day 0 before FMT and day 1 post FMT (n=10 mice per group). Statistical significance was determined by unpaired Mann-Whitney test. *P<0.05, **P<0.01. C) Enumeration of C. albicans in feces of mice both on day 0 before FMT and day 1 post FMT (n=10 mice per group). Statistical significance represents comparison between C. albicans load on day 0 before FMT and day 1 post FMT, by paired Mann-Whitney test. **P<0.01. Dot graphs show means.+-.s.e.m, performed at least two times independently.
[0027] FIG. 16 The presence of C. albicans is linked to FMT outcomes in CDI. The absolute abundance of fecal C. albicans before and after FMT at the last follow-up in FMT recipients, assessed by quantitative PCR. Comparison of the fecal C. albicans levels between pre-FMT samples and post-FMT samples was performed by paired Wilcoxon signed rank test, *P<0.05. Comparison of the fecal C. albicans levels between the post-FMT samples of FMT responders and FMT non-responders was performed by Mann-Whitney test, .sup.$$P<0.01. ND denotes no detectable C. albicans in the feces.
[0028] FIG. 17 Comparison of the total fungal load in the feces of controls and CDI subjects. Statistical significance was determined by Mann-Whitney test, ***P<0.001.
[0029] FIG. 18 The total fecal fungal load and C. albicans in inflammatory bowel disease (IBD). a. Comparison of the total fungal load in the feces of controls and IBD subjects, including patients with CD and UC. Statistical significance was determined by Mann-Whitney test, ***P<0.001, *P<0.05. b. Comparison of C. albicans levels in the feces of controls and CDI subjects.
[0030] FIG. 19 The total fecal fungal load in irritable bowel syndrome (IBS). Comparison of the total fungal load in the feces of controls and IBS subjects. Statistical significance was determined by Mann-Whitney test, ***P<0.001, **P<0.05.
[0031] FIG. 20 Post-FMT alterations in the taxonomic composition of the bacterial microbiota of CDI recipients in association with FMT response. Bacterial configurations in FMT recipients over the course of longitudinal follow-up and in their corresponding donors at baseline, at the phylum (a) and family (b) levels. c, Heatmap of the relative abundance of differentially presented bacterial genera in donor, pre-FMT and post-FMT last follow-up samples. d, Differentially presented bacteria taxa across post-FMT samples of FMT responders versus non-responders at the phylum, family, and genus levels. Statistical significance level was determined by LefSe analysis with FDR correction (only those taxa with q values<0.05 and LDA effect size>2 are shown).
[0032] FIG. 21 Quantification of fecal C. albicans levels in IBD concurrent with CDI subjects before and after FMT. The fecal C. albicans level (gDNA content/fecal DNA) was determined by qPCR. PRB, peri rectal bleeding.
[0033] Definitions
[0034] The term "fecal microbiota transplantation (FMT)" or "stool transplant" refers to a medical procedure during which fecal matter containing live fecal microorganisms (bacteria, fungi, and he like) obtained from a healthy individual is transferred into the gastrointestinal tract of a recipient to restore healthy gut microflora that has been disrupted or destroyed by a variety of medical conditions. Typically, the fecal matter from a healthy donor is first processed into an appropriate form for the transplantation, which can be made through direct deposit into the lower gastrointestinal tract such as by colonoscopy, or by nasal intubation, or through oral ingestion of an encapsulated material containing dried and frozen fecal matter. Clostridium difficile infection (CDI) is the condition most commonly treated by FMT, although a number of other diseases and disorders including in the digestive system and in the nervous system have been reported to be successfully treated by FMT.
[0035] The term "inhibiting" or "inhibition," as used herein, refers to any detectable negative effect on a target biological process, such as RNA/protein expression of a target gene, the biological activity of a target protein, cellular signal transduction, cell proliferation, and the like. Typically, an inhibition is reflected in a decrease of at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90% or greater in the target process (e.g., growth or proliferation of fungal cells), or any one of the downstream parameters mentioned above, when compared to a control. "Inhibition" further includes a 100% reduction, i.e., a complete elimination, prevention, or abolition of a target biological process or signal. The other relative terms such as "suppressing," "suppression," "reducing," and "reduction" are used in a similar fashion in this disclosure to refer to decreases to different levels (e.g., at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90% or greater decrease compared to a control level) up to complete elimination of a target biological process or signal. On the other hand, terms such as "activate," "activating," "activation," "increase," "increasing," "promote," "promoting," "enhance," "enhancing," or "enhancement" are used in this disclosure to encompass positive changes at different levels (e.g., at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 200%, or greater such as 3, 5, 8, 10, 20-fold increase compared to a control level) in a target process or signal.
[0036] As used herein, "C. albicans" refers to a fungal species belonging to the Candida genus, Saccharomycetaceae family, Saccharomycetales order, Saccharomycetes class, and Ascomycota division. A common member of the human gut flora, C. albicans is a potential yeast pathogen capable of causing opportunistic infection in humans, especially in those with compromised immune system.
[0037] The term "antifungal agent" refers to any substance that is capable of inhibiting, suppressing, or preventing the growth or proliferation of fungal species, especially those of the Ascomycota division, such as C. albicans. Known agents with fungicidal activity include amphotericin B, echinocandin, fluconazole, nystatin, and clotrimazole.
[0038] "Percentage relative abundance," when used in the context of describing the presence of a particular fungal species (e.g., C. albicans) in relation to all fungal species present in the same environment, refers to the relative amount of the fungal species out of the amount of all fungal species as expressed in a percentage form. For instance, the percentage relative abundance of C. albicans can be determined by comparing the quantity of C. albicans-specific DNA (e.g., determined by quantitative polymerase chain reaction) in one given sample with the quantity of all fungal DNA (e.g., determined by quantitative PCR and sequencing based on the Internal transcribed spacer 2 or ITS2 sequence) in the same sample.
[0039] "Absolute abundance," when used in the context of describing the presence of a particular fungal species (e.g., C. albicans) in the feces, refers to the amount of DNA derived from the fungal species out of the amount of all DNA in a fecal sample. For instance, the absolute abundance of C. albicans can be determined by comparing the quantity of C. albicans-specific DNA (e.g., determined by quantitative polymerase chain reaction) in one given sample with the quantity of all fecal DNA in the same sample.
[0040] "Total fungal load" of a fecal sample, as used herein, refers to the amount of all fungal DNA out of the amount of all DNA in the fecal sample. For instance, the absolute abundance of fungi can be determined by comparing the quantity of fungal specific DNA (e.g., 18S rDNA determined by quantitative polymerase chain reaction) in one given sample with the quantity of all fecal DNA in the same sample.
[0041] The term "effective amount," as used herein, refers to an amount of a substance that produces a desired effect (e.g., an inhibitory or suppressive effect on C. albicans growth) for which the substance (e.g., an antifungal agent) is used or administered. The effects include the prevention, inhibition, or delaying of any pertinent biological process during C. albicans growth or development to any detectable extent. The exact amount will depend on the nature of the substance (the active agent), the manner of use/administration, and the purpose of the application, and will be ascertainable by one skilled in the art using known techniques as well as those described herein.
[0042] As used herein, the term "about" denotes a range of value that is +/-10% of a specified value. For instance, "about 10" denotes the value range of 10+/-10.times.10%, i.e., 9 to 11.
DETAILED DESCRIPTION OF THE INVENTION
I. Introduction
[0043] The invention provides a novel approach for assessing the likelihood of effective FMT prior to the procedure being performed as well as for improving the effectiveness of the FMT procedure. During their studies, the present inventors discovered that the presence and relative abundance of certain fungal species both in a recipient's gastrointestinal tract and in a donor's stool directly correlate with the outcome of FMT. In particular, the fungal species Candida albicans of the Saccharomycetaceae family is found to negatively impact the effectiveness of FMT. The detection of C. albicans in a potential donor's stool thus can be used to guide donor selection, whereas analysis of C. albicans level in an FMT recipient can determine whether the subject is immediately ready for FMT or should be treated with an antifungal agent that suppresses C. albicans growth prior to FMT in order to optimize the therapeutic outcome.
II. FMT Donors and Recipients
[0044] Patients suffering from CDI, especially recurring CDI, are often considered as recipients for FMT treatment. Aside from CDI, other diseases and conditions, including those of digestive system or nervous system such as colitis, irritable bowel syndrome, multiple sclerosis, Parkinson's Disease, diabetes mellitus, and obesity are also beginning to be considered for FMT treatment.
[0045] Fecal matter used in FMT is obtained from a healthy donor and then processed into appropriate forms for the intended means of delivery in the upcoming FMT procedure. Up until now, the general criterion for an FMT donor is simply that the donor is a healthy individual without any known diseases or disorders especially in the digestive tract, although some preference is often given to the members of the same household as the recipient.
[0046] The present inventors have discovered in their studies that elevated presence of one particular fungal species, C. albicans, in a recipient's gastrointestinal tract or in a donor stool (which is used in the transplantation after being processed) can significantly reduce efficacy of FMT treatment in a patient. In contrast, successful FMT has been observed as correlating with elevated presence of other fungal species, such as those belonging to the genus of Saccharomyces or Aspergillus, in a recipient or in a donor stool. This revelation enables the initial screening of individuals as appropriate FMT donors as well as the initial screening of patients as likely candidates for successful FMT treatment: if a candidate donor's stool contains an elevated level of C. albicans (e.g., greater than 0.1% of total fungi), the candidate is deemed as unsuitable as an FMT donor, and his stool should not be taken or used in FMT; if a candidate's stool sample shows no or only low level of C. albicans (e.g., no greater than 0.1% of total fungi), then the candidate is deemed an appropriate FMT donor and his fecal material can be immediately retrieved for processing and later used in FMT. On the other hand, if a patient who has been proposed to receive FMT treatment, and his stool sample shows an elevated level of C. albicans (e.g., greater than 10% of total fungi), then the patient is deemed to be unsuitable to receive FMT and is therefore not to be given FMT, as the therapy is likely to be unsuccessful; if a patient's stool sample shows no or only a low level of C. albicans (e.g., no greater than 10% of total fungi), the patient is deemed an appropriate recipient for FMT who is likely to enjoy therapeutic success from FMT, and thus can start FMT treatment immediately without other steps of preparation or pre-treatment.
[0047] Various methods have been reported in the literature for determining the levels of all fungal species in a sample, for example, amplification (e.g., by PCR) and sequencing of fungal polynucleotide sequence by using the Internal transcribed spacer 2 (ITS2) sequence. On the other hand, the level of any given fungal species may be determined by amplification and sequencing of its signature 18S rRNA sequence. A percentage abundance is often used as a parameter to indicate the relative level of a fungal species in a given environment.
III. Methods for Improving FMT Efficacy
[0048] The discovery by the present inventors revealing the direct correlation between an elevated level of C. albicans in FMT donor or recipient and reduced efficacy of FMT treatment not only allows one to devise an initial screening process to identify appropriate donors and recipients for the FMT procedure, it also enables different methods for improving FMT efficacy by reducing the level of C. albicans in a donor and in a recipient prior to the FMT treatment.
[0049] As discussed in the above section, when a candidate donor's stool is tested and found to contain an elevated level of C. albicans (e.g., greater than 0.1% of total fungi), the candidate is deemed as unsuitable as an FMT donor, and his stool should not be taken for use in FMT as it is unlikely to result in a successful FMT treatment if used. Similarly, when a patient or proposed FMT recipient whose stool is tested and found to contain an elevated level of C. albicans (e.g., greater than 10% of total fungi), the patient is deemed as an unsuitable recipient for FMT, and he should not immediately undergo FMT due to the high probability of an ineffective outcome. Yet these cases of expected unsuccessful treatment outcome can be readily improved in view of the inventors' discovery.
[0050] First, for a patient who has been considered for receiving FMT but who has also been deemed an unsuitable recipient of FMT due to an elevated level of C. albicans (e.g., above 10% of total fungi) found in his/her stool sample, which indicates a diminished chance of a successful FMT, measures can be taken to lower his/her level of C. albicans before FMT is commenced so that a much greater efficacy can be achieved for the FMT procedure. For instance, an antifungal agent capable of suppressing the growth or proliferation of C. albicans can be administered to the patient in an effective amount such that the level of C. albicans in the patient's digestive track and in the feces is significantly reduced (e.g., no more than 10% of total fungi) prior to the start of the FMT procedure. In this case, the patient's C. albicans level is to be determined twice: once at the initial screening stage, a second time after the initial level is deemed too high for an effective FMT and after an antifungal agent has been given to the patient. Once the C. albicans level is confirmed as lowered to a percentage that would allow satisfactory FMT outcome, the patient is then ready to undergo FMT treatment.
[0051] Second, for a candidate who has been deemed improper to serve as an FMT donor due to a higher level of C. albicans in his stool, the expected undesirable FMT outcome can be remedied by treating the candidate donor with an effective amount of an antifungal agent capable of suppressing the growth or proliferation of C. albicans can be administered. Since the donor's body, especially the gastrointestinal track, contains a vast collection of microorganisms many of which are important for the health of gut microflora and for the success of FMT, a useful antifungal agent for this purpose cannot be a broad-spectrum fungicide. Rather, it should be an agent that narrowly and precisely targets the species of C. albicans without significantly affecting other fungal species, including those closely related to C. albicans. Although the agent may be of any chemical compound in nature, small polynucleotides (e.g., siRNAs, miRNAs, miniRNAs, lncRNAs, or antisense DNAs/RNAs) may be the most effective in achieving the specific task of disrupting the expression of one or more key genes in the life cycle of C. albicans so as to specifically inhibit the proliferation of the target species only without significant impact on other closely related fungal species.
[0052] Immediately upon completion of FMT procedure, the recipient may be further monitored by continuous testing of the level of C. albicans in the stool samples on a daily basis for up to 5 days post-FMT while the clinical symptoms of the condition being treated are also being monitored in order to assess FMT outcome and the corresponding C. albicans level in the recipient.
IV. Kits and Compositions for Improved FMT
[0053] The present invention also provides novel kits and compositions that can be used for improving FMT efficacy. For example, in a kit for treating a patient in need of FMT, a first composition intended for transplantation into a patient or FMT recipient and a second composition intended to be administered to the recipient for reducing the level of C. albicans in the recipient. The first composition comprises a fecal material from a donor, which has been processed, formulated, and packaged to be in an appropriate form in accordance with the delivery means in the FMT procedure, which may be by direct deposit in the recipient's lower gastrointestinal track (e.g., wet or semi-wet form) or by oral ingestion (e.g., frozen dried encapsulated). The second composition comprises an antifungal agent capable of suppressing the growth/proliferation of C. albicans, which may be a broad-spectrum fungicide or a specific inhibitor of the C. albicans species, and one or more pharmaceutically acceptable excipient. The composition is formulated for the intended delivery method of the antifungal agent, for example, by injection (intravenous, intraperitoneal, intramuscular, or subcutaneous injection) or by oral ingestion or by local deposit (e.g., suppositories). The first and second compositions are often kept separately in two different containers in the kit. Typically, the kit will further include printed material providing detailed instructions for users of the kit, such as providing information of the schedule and dosing arrangement for administering the first and second compositions to a recipient.
[0054] In another aspect of this invention, alternative compositions useful in FMT with improved efficacy may be devised to contain at least these two components: (1) a donor stool material containing live fecal microorganisms, and (2) an antifungal agent that specifically suppresses the growth or proliferation of C. albicans but exhibits no such suppressive or inhibitory effect against other fungal species. Component (2) preferably is not a broad-spectrum fungicide; rather, it should be a specific anti-C. albicans agent. For example, it may be short polynucleotide in nature of, e.g., a small inhibitory RNA, microRNA, miniRNA, lncRNA, or an antisense oligonucleotide, that is capable of disrupting the expression of at least one key gene in the life cycle of C. albicans, such that the agent is capable of specifically targeting the species only without significantly affecting other closely related fungal species. Component (2) is particularly useful in the case of a donor's stool containing a level of C. albicans too high to permit a satisfactory FMT outcome, as it is capable of locally and specifically suppressing the proliferation of C. albincans so as to ensure the success of FMT despite the less than desirable quality of the donor fecal material.
EXAMPLES
[0055] The following examples are provided by way of illustration only and not by way of limitation. Those of skill in the art will readily recognize a variety of non-critical parameters that could be changed or modified to yield essentially the same or similar results.
Example 1
Introduction
[0056] Fecal microbiota transplantation (FMT) is effective in treating recurrent Clostridium difficile infection (CDI) and is increasingly being utilized in other human diseases. Whilst bacteria colonization in recipients after FMT has been established, little is known of the role of the gut mycobiota. In this study the present inventors show gut fungal dysbiosis in CDI and identify that donor-derived fungi colonization in recipient is associated with FMT response. Mycobiota profiling in CDI reveals over-presentation of C. albicans and decreased fungal diversity, richness and evenness compared with healthy controls. Cure after FMT was observed when donor-derived fungal taxa predominated in recipients' mycobiota. FMT responders display a high prevalence of Saccharomyces and Aspergillus whilst non-responders and individuals treated with antibiotics display a dominant presence of Candida. High abundance of C. albicans in recipient before FMT and in donor stool nullifies FMT efficacy in eradicating CDI. Furthermore, C. albicans compromises FMT efficacy in a mouse model of CDI, while anti-fungal treatment reestablishes its efficacy. This study furthers the knowledge of human gut mycobiota dynamics and their contribution to FMT, and it enables an understanding of personalized donor-recipient selection in future FMT studies for various human diseases.
[0057] The past decade has witnessed an increasing use of fecal microbiota transplantation (FMT) as a promising treatment option for several diseases .sup.1-3, yet success rates are variable with a cure rate of 85-90% in recurrent Clostridium difficile infections (CDI).sup.3-5 and a response rate of 30-40% in inflammatory bowel disease.sup.6-8. Such variations may be related to disease traits, recipient factors or donor characteristics. The mechanisms underlying a successful FMT and its relationship with gut microbial profiles in donor-recipient pairs remain elusive. To date, the efficacy of FMT has been mostly ascribed to the restoration of the bacterial microbiota and a sustained co-existence of donor and recipient bacterial strains.sup.9-12. Recently, bacteriophages have been shown to be altered in CDI after FMT and these changes were associated with treatment outcome .sup.13-15. The human gastrointestinal tract is also colonized by a large population of fungi, collectively referred to as the mycobiota, which play an important role in human health.sup.16,17. Gut mycobiota contribute to normal human physiology and in some cases can recapitulate the benefit of intestinal bacteria via regulating host immunity and maintaining intestinal homeostasis.sup.16,17,18. Whether donor-derived mycobiota can colonize a recipient host, the fate of donor and recipient mycobiota after FMT and their relationship with treatment outcomes are unknown. The inventors performed internal transcribed spacer 2 (ITS2) and 16S rDNA sequencing in FMT-treated subjects with CDI to explore the effects of FMT on the gut mycobiome in association with treatment outcome. A proof-of-causality study was conducted in C. difficile-infected mice to confirm the role of gut mycobiota in FMT response.
Results
Gut Fungal Dysbiosis in CDI
[0058] The fecal mycobiomes were compared between 31 CDI subjects and 23 healthy controls. There was a significant decrease in fungal diversity, evenness and richness in CDI compared with controls (Mann-Whitney test, p=0.0120, 0.0309, and 0.0043, respectively, FIG. 1a). The fungal communities of CDI subjects were significantly separated from those of healthy controls at the OTU level (based on Bray-Curtis distance, adonis test p=0.003, FIG. 1b). At the phylum level, Ascomycota was expanded in CDI compared with controls (Mann-Whitney test, p=0.0083, FIG. 1c). At the species level, 17 fungal species were found to be differentially present between CDI and controls (LefSe analysis with FDR adjusted q<0.05, FIG. 1d). Amongst these species, only C. albicans was significantly enriched in CDI (FIG. 1d, e, Mann-Whitney test, p=0.0080), whereas 16 other species were enriched in controls. In line with the observation at the species level, more taxa were enriched in controls than in CDI, as determined by LefSe analysis (9 versus 1 at the order level, 15 versus 2 at the family level, 16 versus 1 at the genus level, Table 2). Altogether, these data indicate dysbiosis of the enteric mycobiota in patients with CDI.
[0059] Over-presentation of C. albicans in absolute abundance in CDI was confirmed through quantitative PCR (FIG. 5a). Antibiotic use has been shown to be a major contributor to the development of CDI by decreasing bacterial colonization resistance. The effect of antibiotics on C. albicans levels in CDI was further assessed. Stool samples were collected from new consecutive CDI patients, including 12 CDI patients with antibiotics exposure, 12 CDI patients with no antibiotics exposure at inclusion, and 17 healthy controls. Significantly higher levels of fecal C. albicans were found in CDI subjects exposed to antibiotics at inclusion, compared with controls (FIG. 5b, Mann-Whitney test, p=0.0131). C. albicans levels were also significantly higher in CDI subjects not exposed to antibiotics at inclusion when compared with controls (FIG. 5b, Mann-Whitney test, p=0.0469). These data indicate that both CDI and antibiotics are contributors to increased levels of C. albicans.
Donor fungi colonization in recipient is associated with FMT response
[0060] It was then explored whether FMT leads to colonization of donor-derived fungi and its association with treatment efficacy. Changes in the gut mycobiomes of recipients after FMT were monitored at multiple time points in 16 CDI subjects, using pre-FMT samples of each donor-recipient pair as a baseline for FMT (FIG. 6). Amongst 16 CDI subjects treated with FMT, nine remained symptom-free with a negative stool C. difficile toxin at the last follow-up (termed responders, FMT1-FMT9), whilst seven developed recurrence of CDI (termed non-responders, FMT10-FMT16) (Table 1). It was next investigated whether donor-derived fungi and bacteria in recipients may influence FMT outcomes. Subjects who responded to FMT demonstrated a larger proportion of fungal and bacterial OTUs that were transferred and predominated in the feces of recipients after FMT, compared to those who did not respond (Mann-Whitney test, p=0.0068 and 0.0164 respectively for comparison of donor-derived fungal and bacterial OTU ratios in recipients, FIG. 2a, b). The community structure at the genus level showed a higher abundance of the genera Aspergillus and Penicillum in FMT responders than in non-responders (FIG. 2c). In contrast, the genera Candida and Simplicillium were significantly enriched in FMT non-responders. Analogously, a similar pattern was observed at the bacterial community structure. FMT responders displayed bacterial abundance resembling that of the donor, whereas FMT non-responders showed inadequate abundance of donor-enriched bacteria at the last follow-up post FMT (FIG. 7). Of note, in recipients FMT12 and FMT16, bacterial configurations at the last follow-up after FMT were similar to that of healthy controls, but their gut mycobiota configurations differed significantly from that of healthy controls. These data indicate that restoration of the gut mycobiota is at least as important as, if not more than, restoration of the bacterial microbiota in CDI recipients. Taken together, these data indicate that the final proportion of donor-derived fungal and bacterial taxa and alterations of the fecal fungal composition in the recipient post FMT were associated with treatment outcome of FMT.
FMT Alters the Gut Mycobiota Distinct from Antibiotic Treatment
[0061] CDI subjects who responded to FMT showed a significant increase in fungal richness and diversity (Wilcoxon matched-pairs singed rank test, p=0.0273 and p=0.0474 respectively, FIG. 3a, b). Although baseline bacterial diversity, evenness and richness were significantly lower in CDI subjects compared to controls (Mann-Whitney test, all p<0.0001, FIG. 6a), after FMT there was a significant increase in bacterial richness (Wilcoxon matched-pairs singed rank test, p=0.019) and a marginally significant increase (Wilcoxon matched-pairs singed rank test, p=0.098) in bacterial diversity in FMT responders. During post-FMT follow-up, there were profound differences in the gut mycobiota configurations across different donor-recipient pairs, however a significantly higher prevalence of the genus Candida was observed across the serial post-FMT fecal samples of FMT non-responders relative to that of responders (FIG. 3c, d). In contrast, the genera Saccharomyces and Aspergillus were present in higher abundances in FMT responders than in non-responders (FIG. 3c, d). Discriminative analysis identified disparately presented taxa between post-FMT samples of FMT responders and non-responders, at the genus and species levels (FIG. 3d). C. albicans was the most prominent species enriched after FMT in non-responders.
[0062] C. albicans markedly decreased after FMT (Wilcoxon matched-pairs singed rank test, p=0.0458, FIG. 3d, e). Interestingly, both the abundance of C. albicans in donor feces and in post-FMT recipient fecal samples were associated with FMT treatment outcome. FMT recipients transplanted with a donor feces with C. albicans<0.1% in the fungal community achieved a response to FMT treatment, compared to those transplanted with a donor feces with C. albicans>0.1% (Chi-square test p=0.049, FIG. 3f). Recipients with an initial high abundance of C. albicans before FMT and continuing to have a relative abundance of C. albicans>10% after FMT all experienced a disease recurrence after FMT (Chi-square test p=0.029, FIG. 3g). These data indicate that the presence of C. albicans compromises FMT efficacy. The absolute abundance of C. albicans (in fecal input DNA) was markedly decreased after FMT in FMT responder group (Wilcoxon matched-pairs singed rank test, p=0.0391, FIG. 16). Interestingly, FMT non-responders exhibited significantly higher post-FMT fecal C. albicans levels in absolute abundance than FMT responders [Mann-Whitney test, p=0.0018, Log.sub.10 transformed effect size 3.05 (95% CI: 1.48-4.29), FIG. 16], indicating C. albicans can be a marker for disease recurrence and/or pathogenesis.
[0063] The effect of antibiotics on the gut mycobiota was also assessed across longitudinal time-points in 8 CDI subjects treated with vancomycin (STD treatment, FIG. 6, Table 1). Five of the eight subjects remained symptom-free with a negative stool C. difficile toxin at the last follow-up (termed responders, STD1-STD5), while three developed recurrence of CDI (termed non-responders, STD6-STD8). Unlike FMT, vancomycin induced inconsistent alterations in the fungal richness and diversity during longitudinal follow-up (FIG. 9a, b). There was no significant difference in the fungal richness or diversity between STD responders and non-responders after FMT, although vancomycin resulted in a significant increase in bacterial diversity in responders after FMT (FIG. 10, Wilcoxon matched-pairs singed rank test, p=0.0198).
[0064] FMT and vancomycin led to an increase in the gut fungal diversity in 81.3% (13 out of 16) and 37.5% (3 out of 8) of CDI subjects, respectively (Chi-square test p=0.032, FIG. 8c), and an increase in the gut fungal richness in 68.8% (11 out of 16) and 37.5% (3 out of 8) of CDI subjects, respectively (FIG. 8c). FMT responders showed a significantly higher fold-change post FMT in both fungal richness and fungal diversity compared to STD responders (Mann-Whitney test p=0.019 and Chi-square test p=0.05 respectively, FIG. 8d, e). Collectively, these data indicate that FMT may be more influential in orchestrating the gut mycobiota than antibiotics.
[0065] Taxonomical analysis was performed to further elaborate the effect of antibiotics on the fungal community and to discern differences between FMT and antibiotics in modulating the gut mycobiota. After vancomycin treatment, fungal compositions exhibited similar configurations during follow-up across STD subjects, with a marked expansion of the genus Candida (FIG. 8f). To define differentially enriched fungal taxa between subjects who responded to FMT and vancomycin, we implemented LefSe analysis across all follow-up samples of treatment responders. FMT treatment enriched the genera Saccharomyces and Cryptococcus in those who responded, whereas vancomycin disparately enriched a panel of fungal genera in STD responders after treatment, which included Candida, Talaromyces, Erythrobasidium, Periconia, Stemphylium, Ganoderma (FIG. 11). At the family level, FMT caused an enrichment of Saccharomycetacean and Herpotrichiellaceae, while vancomycin caused an enrichment of Intertae sedis (FIG. 11). There was no statistically significant difference in the relative abundance of C. albicans between STD responders and non-responders, however a decrease in C. albicans was seen in STD responders after vancomycin treatment (FIG. 12). Subject STD7 who had a post-STD relative abundance of C. albicans>10% developed CDI recurrence after vancomycin treatment, further substantiating the importance of alleviation of C. albicans for eradicating CDI.
Trans-kingdom Interactions Between Gut Mycobiota and Bacterial Microbiota are Associated with Treatment Outcome
[0066] To characterize the ecological network of the gut mycobiota and bacterial microbiota, the correlation of the a-diversity (diversity, evenness and richness) of the fungal community with that of the bacterial community was evaluated. Among the post-treatment samples of FMT responders, significant positive correlations were found between fungal diversity and bacterial diversity, and between fungal richness and bacterial diversity, evenness, and richness (Spearman's correlation, permutation test, P<0.05, FIG. 13). In the post-treatment samples of FMT non-responders and STD responders, the correlation between bacterial and fungal communities showed a depletion of correlations between fungal richness and other bacterial and fungal .alpha. diversity indexes. The correlations were completely abolished across the post-treatment samples of STD non-responders. The correlations of fungal genera with bacterial genera were further assessed in controls and CDI subjects in association with treatment response. Significant inverse correlations between control-enriched bacteria, including butyrate-producing Roseburia, and CDI-enriched Candida were observed in FMT responders and STD responders after treatment, paralleling a prevalence of positive correlations between control-enriched bacteria and control-enriched fungi among which correlation of Roseburia and Aspergillus was present in both FMT responders and STD responders (FIG. 14). However, those who did not respond to either FMT or STD displayed an apparent contraction in the number of fungal-bacterial correlations after treatment, compared to FMT responders and STD responders. These data suggest the importance of restoration of an intricate and homeostatic fungal-bacterial ecosystem in maintaining treatment response.
C. albicans Compromises FMT Efficacy in a Murine Model of CDI
[0067] C. albicans was the most prominent species associated with treatment failure of FMT in CDI, suggesting a possible causal relationship. This assumption is further supported by reports whereby CDI recurrence was observed after antibiotics treatment.sup.3,19, as antibiotics contribute to the expansion of Candida. To determine the causal relationship between C. albicans and response to FMT, the efficacy of FMT in eliminating C. difficile was assessed using a C. difficile induced-diarrhea murine model in three groups of mice: (i) mice infused with human stool preparation, (ii) mice colonized with C. albicans then infused with human stool preparation, and (iii) mice infused with human stool preparation supplemented with C. albicans during fecal transplantation (FIG. 4a). FMT was effective in ameliorating diarrhea, intestinal inflammation, and decreasing C. difficile burden, compared to CDI group, while no difference in C. difficile load was observed among all groups before FMT (FIG. 4b-d). However, mice that was colonized with C. albicans prior to FMT or those infused with donor stool supplemented with C. albicans suffered significant diarrhea, intestinal inflammation, and augmented C. difficile burden post FMT, when compared with mice administered with a single infusion of human stool (FIG. 4b-d). There were high levels of C. albicans in these recipient mice on day 1 post FMT, though a decrease in C. albicans load was observed after FMT in mice colonized with C. albicans prior to FMT (FIG. 4e). An anti-fungal agent, fluconazole, to eradicate C. albicans in a group of recipient mice prior to human stool infusion (FMT) (FIG. 15a). C. difficile load was then compared after human stool infusion between mice with and without anti-fungal treatment. Anti-fungal treatment in recipient mice colonized with C. albicans before human stool infusion restored the efficacy of FMT in clearing C. difficile infection (FIG. 15b). These data demonstrate that the existence of C. albicans, either in the recipient or in the donor, negates the efficacy of FMT in clearing C. difficile, while antifungal treatment reestablishes its efficacy. These results highlight that persistent fungal dysbiosis with aberrant presence of C. albicans can confer an unfavourable FMT outcome in CDI.
Total Fungal Load is Increased in CDI
[0068] The total fecal fungal load was significantly higher in CDI than in controls [Mann-Whitney test, p=0.0004, Log.sub.log transformed effect size 1.32 (95% CI: 0.62-1.97), FIG.17].
Total Fungal Load and C. albicans in Inflammatory Bowel Disease (IBD)
[0069] The total fecal fungal load was significantly higher in patients with IBD, including patients with Crohn's disease (CD) and Ulcerative colitis (UC)--two subtypes of IBD, than in controls (Mann-Whitney test, p=0.0003, p=0.0225, respectively, FIG. 18a). The fecal presence ratio of C. albicans is higher in CD than in controls, and 3 CD patients exhibiting the highest C. albicans levels had a history of recent exposure to antibiotics (FIG. 18b).
Total Fungal Load is Increased in Irritable Bowel Syndrome (IBS)
[0070] The total fecal fungal load was significantly higher in IBS than in controls [Mann-Whitney test, p=0.0237, FIG.19].
Bacterial Alterations in CDI after FMT in Association with FMT Outcome
[0071] The present inventors explored the composition of the bacterial microbiota after FMT in relation to FMT outcomes, at various taxonomic levels (FIG. 20). Actinobacteria, Bacteroidetes (phylum-level taxa), Lachnospiraceae, Clostridiaceae, and Ruminococcaceae (family-level taxa), Clostridium, Blautia, and Faecalibacterium (genus-level taxa) were significantly more enriched in FMT responders than in non-responders after FMT. However, bacteria from the phylum Proteobacteria were more abundant in FMT non-responders relative to FMT responders. FMT responders displayed bacterial abundances resembling that of the donor, whereas FMT non-responders showed inadequate relative abundances of donor-enriched bacteria at the last follow-up after FMT (FIG. 20c). LefSe analysis on the fecal bacteriomes of donors at the genus level identified Escherichia and Proteus as the differentially enriched genera in FMT responders' donor stool and in FMT non-responders' donor stool respectively (LDA effect size 2.58 and 2.35, FDR adjusted q=0.017 and 0.006, respectively).
Discussion
[0072] This is the first study to characterize the gut mycobiota in CDI and to elucidate mycobiota alterations after FMT in relation to treatment outcome. Patients with CDI showed enteric fungal dysbiosis. Importantly, disease recurrence after FMT was associated with several important findings including persistent fungal dysbiosis, low levels of donor-derived fungal colonization, high abundance of C. albicans in the recipient stool before FMT and the presence of C. albicans in the donor stool. The observations that disease cure requires both fungal and bacterial colonization from the donor provides new and important insights into the potential therapeutic importance of the gut mycobiota in treatment outcome in FMT, beyond the bacterial microbiota. These data also highlight a new concept in FMT that the abundance of fecal C. albicans both in recipient before treatment and in donor are critical components when considering implementation of FMT. Integration of more in-depth mycobiota analysis in donor-recipient pair may lead to personalized and targeted gut microbial therapy in the future.
[0073] Although studies of the gut mycobiota have lagged behind that of the gut bacterial microbiota, fungi are increasingly being considered as important players of the gut and interactions between pathogenic and commensal fungal and bacterial communities are crucial in the maintenance of human health and disease pathogenesis.sup.16. Furthermore, disruption of the gut mycobiota has deleterious effect on host immunity.sup.17. Despite high interpersonal variability of the gut mycobiota in patients with CDI, there was a significant expansion of the genus Candida and the species C. albicans. Interestingly, FMT induced an increase in the genus Saccharomyces along with a marked contraction of Candida and C. albicans after FMT in treatment responders, while recipients who demonstrated high abundance of C. albicans in the stool after FMT (10%) or those whose donor had a high abundance of (0.1%) of C. albicans were more likely to have disease recurrence after FMT. Over-presentation of C. albicans in the recipient stool after FMT may largely contribute to treatment failure.
[0074] The role of fungal commensals in educating the human immune system has gained new appreciation in intestinal disease. In the steady state, bacterial communities keep fungi in check in the gut. Fungi are major causes of infections among immunocompromised or hospitalized patients with serious underlying diseases and comorbidities. Candida species remain the most important cause of opportunistic infections worldwide, affecting predominantly elderly patients.sup.20. Candidalysin was recently unveiled as a fungal toxin from C. albicans critical for mucosal infection.sup.21. Commensal bacteria inhibit C. albicans colonization through activation of HIF-1.alpha. and LL-37.sup.22. Antibiotic treatment selectively and effectively eradicates the bacterial community but consequently leads to fungal outgrowth, particularly the Candida species.sup.23,24. Antibiotics or immunosuppressants are effective in the short term but they likely compromise the immune system in the longer term. A compromised immune system creates a more favourable environment to expansion of Candida and overgrowth of Candida can alter the recovery of the gut bacterial microbiota after cessation of antibiotic treatment .sup.25,26. In this study, the over-presence of Candida species in recipients might account for the high failure rate of FMT in CDI. In DSS-induced colitis mouse model as well as patients with inflammatory bowel disease (IBD), C. albicans and Candida were significantly enriched.sup.27-29. Antifungal treatment decreased Candida prevalence and ameliorated inflammatory responses in DSS colitis mice.sup.29. However, disruption of fungal communities by long-term use of antifungals aggravated severity of DSS colitis and allergic airway.sup.30. Collectively, these data implicate the importance of the gut fungal-bacterial homeostasis in host health. These data suggest that the establishment of a balanced gut fungal and bacterial community via FMT is important to eradicate CDI, as FMT non-responders showed abrogated fungi-bacteria correlations in a-diversity and taxa when compared with responders.
[0075] In conclusion, gut mycobiota alterations may determine treatment outcome in FMT. The persistence of fungal dysbiosis, particularly the presence of C. albicans, can incur CDI recurrence. The findings disclosed herein highlight the importance of both "optimal" donor selection and pre-FMT eradication of C. albicans in recipient during FMT practice, where future FMT therapy should incorporate detailed characterization and stratification of both donor and recipient fecal mycobiomes. These results provide a framework for future investigations into the contribution of donor/recipient mycobiota profiles and gut fungi-bacteria interactions in FMT treatment for various human diseases.
Methods
[0076] Study subjects and treatment outcome
[0077] The current study was a sub-study from a randomised controlled trial (RCT) of FMT versus vancomycin (standard therapy, STD) for patients with CDI. Consecutive CDI subjects enrolled into this randomised controlled trial were invited to participate in a substudy of assessment of fecal microbiota. Patients were included if they had three or more loose or watery stools per day for at least two consecutive days or eight or more soft or loose stools in 48 hours and a positive stool test for C. difficile based on a two-step testing algorithm in our hospital, a positive GDH (Glutamate dehydrogenase) screening test followed by a positive polymerase chain reaction (PCR) test of C. difficile. A total of 31 subjects with CDI and 24 healthy household controls were recruited and stool samples at baseline were obtained for analyses of fungal and bacterial microbiomes. Among them, 24 CDI subjects consented to have stool samples collected serially after treatment for microbiome analysis. 16 CDI subjects were treated with FMT and 8 were treated with vancomycin, and they were followed up at baseline and at weeks 2, 4, 10 and 16 after treatment (FIG. 6). Subjects in the FMT group received 5 days of vancomycin followed by donor infused stool via nasojejunal route and those who had STD received oral vancomycin 500 mg orally four times per day for 10 days. A computer-generated randomization schedule was used to assign patients to the treatment sequences. All patients kept a stool diary and were questioned about stool frequency and consistency and medication use.
[0078] Treatment response was defined as an absence of diarrhea or persistent diarrhea that could be explained by other causes with a negative stool test for C. difficile toxin, while relapse was defined as diarrhea with a positive stool test for C. difficile toxin. Treatment cure is defined as symptom resolution and a negative Clostridium difficile toxin in stool until the last follow-up (last follow-up is referred to as the last stool collection time point, as shown in FIG. 6). 9 of the 16 subjects who had FMT (FMT1-FMT9), and 5 of the 8 patients (STD1-STD5), who had vancomycin were cured of CDI (termed responders, Table 1) at a median follow-up of 16 weeks. CDI recipients FMT11 and FMT12 shared the same donor, and this donor was termed "Donor11". Clinical data of the subjects and collected stool samples are shown in Table 3. None of the patients had received antibiotics or proton pump inhibitors after FMT.
Study design Patient inclusion criteria:
[0079] 1. C. difficile infection was defined as diarrhea (.gtoreq.3 soft, loose or watery stools per day for at least 2 consecutive days or .gtoreq.8 soft or loose stools in 48 hours) and a positive stool test for C. difficile toxin; and
[0080] 2. Age.gtoreq.18; and
[0081] 3. Written informed consent obtained
Patient exclusion criteria:
[0082] 1. The presence of human immunodeficiency virus (HIV) infection with a CD4 count of less than 240
[0083] 2. Pregnancy
[0084] 3. GI Bleeding
[0085] 4. Acute coronary syndrome
Donor screening:
[0086] Donors included individuals who are spouses or partners, first-degree relatives, other relatives, friends, and individuals unknown to the patient. They were screened with a questionnaire and excluded if they had taken antibiotics within the preceding 3 months; were on major immunosuppressive agents, including chemotherapeutic agents; had known or recent exposure to HIV, hepatitis B or C; had a current communicable disease; participated in high-risk sexual behaviors; used illicit drugs; traveled within 6 months to areas with endemic diarrheal illnesses; or had history of inflammatory bowel disease, irritable bowel syndrome or chronic diarrhea, gastrointestinal malignancy or polyposis. In addition, donor was screened for HBsurface Ag, Anti-HBc, Anti-HCV, Anti-HIV, Syphilis EIA, stool microscopy, culture and sensitivity, stool cyst, ova, parasite, norovirus and C. difficile (cytotoxin and PCR assay). All subjects and collected stool samples are listed in Table 1.
[0087] The donors for the FMT group were healthy household controls and the donor stool samples analyzed were the same samples used for FMT. All subjects provided written informed consent.
[0088] Family members provided donor stool for subjects randomised to FMT arm. Cure after FMT or vancomycin therapy was defined as symptom resolution and negative Clostridium difficile toxin in stool at last follow-up by PCR assay. Relapse was defined as diarrhea with a positive stool test for C. difficile toxin.
[0089] This was a randomised but not blinded study. However for mycobiome and bacterial microbiome analyses on stool samples, assessments were initially performed by analysts who were blinded to the clinical outcome of the studied subjects. When the profiled mycobiome and bacterial microbiome data were available for each individual subject, correlation was then made to associate microbiome profiles with treatment outcomes of subjects.
Infusion of Donor Stool
[0090] In subjects who received FMT, a nasoduodenal tube was inserted with radiology guidance. Donor feces was diluted with 500 ml of sterile saline (0.9%), blended and the supernatant was strained with filter paper and poured in a sterile bottle. Within 6 hours after collection of feces by the donor, the solution was infused through a nasoduodenal tube (2 to 3 minutes per 50 ml). The tube was removed 30 minutes after the infusion, and patients were monitored for 2 hours. In subjects with received FMT, a minimum of 50g of donor stool was collected on the same day of infusion and used within 6 hours of collection.
Fecal DNA Extraction
[0091] Fecal DNA was isolated as described below. 100 mg fecal sample was pre-washed with 1 ml ddH.sub.2O and pelleted by centrifugation at 10,000.times.g for 1 minute. The fecal pellet was re-suspended in 800 .mu.l TE buffer (pH 7.5), supplemented with 1.6 .mu.l 2-Mercaptoethanol and 500 U lyticase (Sigma), and incubated at 37.degree. C. for 60 min. The sample was then centrifuged at 10,000.times.g for 2 minutes and fecal DNA was subsequently extracted from the pellet using ZR Fecal DNA miniPrep kit (Zymo Research, Orange, Calif.) according to the protocol. Briefly, fecal pellet was added to the BashingBeadLysis Tube with 750 .mu.l Lysis solution, and then processed at maximum speed for 10 minutes. The lysates were centrifugeed at .gtoreq.10,000.times.g for 1 minute. The supernatant was transferred to a Zymo-Spin.TM. IV Spin Filter in a collection tube and centrifuged at 7,000.times.g for 1 minute. About 1,200 .mu.l of fecal DNA binding buffer was added to the filtrate in the collection tube, followed by concentration and purification in a new filter tube. Finally, a total of 50 .mu.l eluted DNA with a concentration at 20-100 ng/.mu.l was prepared for each sample.
Fungal ITS2 Sequencing and Quality Control
[0092] The final fecal DNA for fungal sequencing was amplified based upon ITS2 (Internal transcribed spacer 2) region using primers as below and PrimeSTAR HS DNA Polymerase kit (TaKaRa, Japan). The primer pairs are ITS2-F: 5'-GCATCGATGAAGAACGCAGC-3', ITS2-R: 5'-TCCTCCGCTTATTGATATGC-3'. ITS2 amplicons were generated with 38 cycles of 3-step PCR: 98.degree. C. 10 s, 59.degree. C. 10 s, and 72.degree. C. 30 s. PCR samples were then sequenced on the Illumina MiSeq PE300 platform (2.times.300 bp, BGI, China), 151,524.+-.97,694 (number.+-.SD) clean sequences obtained on average (sequence statistics in Table 4).
[0093] Raw reads were filtered by SOAPnuke (v 1.5.3) (web site: soap.genomics.org.cn/) developed by BGI as follows: (i) adaptors removed, (ii) read removed if N base is more than 3% of the read, (iii) read removed if bases with quality low than 20 were more than 40% of read, (iv) all duplicates removed. Quality control and data analysis were further implemented in PIPITS (v 1.4.5).sup.31. Briefly, PIPITS_PREP prepares raw reads from Illumina MiSeq sequencers for ITS extraction; PIPITS_FUNITS extracts ITS2 from the reads; and PIPITS_PROCESS analyses the reads to produce operational taxonomic unit (OTU) abundance tables and the RDP taxonomic assignment table for downstream analysis. The quality trimmed and ITS2 extracted reads were aligned to fungi UNITE database exploiting RDP classifier 2.10 for taxonomic assignment to produce operational taxonomic unit (OTU) abundance table (based on sequence identity.gtoreq.97% identity) and phylotype abundance tables at different taxonomic levels, for downstream analysis.
[0094] The fungal OTU and phylptype abundance data were imported into R 3.2.3. Richness, diversity, and evenness calculation were performed using the estimated richness function of the phyloseq package. Spearman correlation and their significance were calculated using the cor and cor.test functions in R, respectively. For the fungal-bacterial taxa comparisons, Spearman correlations were calculated for the relative abundance of the differentially presented fungal taxa and the bacterial taxa determined to be significantly associated with disease by Lefse analysis. Correlation plots were generated using the corrplot package in R. Heat maps were generated using the pheatmap package in R.
Quantitative PCR for Detection of C. albicans in Human Fecal DNA Samples
[0095] C. albicans loads in human stools were quantified by qPCR analysis (SsoAdvanced SYBR Green Supermix, Bio-Rad) of extracted human fecal DNA using C. albicans specific primers: C. albicans-F 5'-CCTGTTTGAGCGTCGTTTCTC-3'; C. albicans-R 5'-TTTGGTTAGACCTAAGCCATTGTCA-3'. C. albicans abundance was determined using standard curves constructed with reference genomic DNA (gDNA) of C. albicans.
Quantitative PCR for Detection of Total Fungal Load in Human Fecal DNA Samples
[0096] Total fungal loads in human stools were quantified by TaqMan qPCR analysis (Premix Ex Taq.TM., TaKaRa) of extracted human fecal DNA using primers.sup.36: Fungi-quant-F 5'-GGRAAACTCACCAGGTCCAG-3'; Fungi-quant-R 5'-GSWCTATCCCCAKCACGA-3', and probe: 5'-TGGTGCATGGCCGTT-3'.
LEfSe Linear Discriminant Analysis
[0097] To compare differences in the configurations of fungal and bacterial microbiomes between CDI patients and healthy controls, between FMT responders and non-responders, between FMT treatment and vancomycin (STD) treatment, Lefse analyses were performed on the Huttenhower lab Galaxy server (web site: huttenhower.sph.harvard.edu/galaxy/) by importing the viral and bacterial relative abundance values and associated sample metadata, with FDR adjusted p value<0.05 considered significant and effect size calculated.
Calculation of Donor Transferred OTUs in Recipients
[0098] In samples after FMT, if a fungal or bacterial OTU was not present in the recipient baseline sample but present both in the corresponding donor baseline sample and in the recipient post-FMT sample, the OTU was defined as "donor derived"; if an OTU was not present in the corresponding donor baseline sample but detected both in the recipient baseline sample and in the recipient post-FMT sample, the OTU was defined as "recipient exclusive"; if an OTU was present across the recipient baseline sample, the recipient post-FMT sample and the corresponding donor baseline sample, the OTU was defined as "donor-recipient co-existed."
Bacterial 16S rRNA Sequencing and Data Analysis
[0099] The final fecal DNA samples were subject to bacterial 16S rRNA V4 region amplification and sequenced on the Illumina MiSeq PE250 platform (2.times.250 bp, BGI, China), 132,081.+-.65,429 (number.+-.SD) sequences obtained on average (sequence statistics in Table 5). Quality control and data analysis were implemented in mothur (v 1.38.0) as previously described.sup.32. Any sequences with ambiguous bases and anything longer than 275 bp were removed, and aligned against the non-redundant Greengenes database (v 13.8).sup.33 using the NAST algorithm. Any sequences that failed to align with the V3-4 region were discarded. The remaining sequences were trimmed to the same alignment coordinates over which they fully overlapped, followed by removal of homopolymers and detection for the presence of chimeras by UChime.
[0100] The resulting sequences were classified against the Greengenes database and annotated with deepest level taxa represented by pseudo-bootstrap confidence scores of at least 80% averaged over 1,000 iterations of the naive Bayesian classifier. Any sequences that were classified as either being originated from archaea, eukarya, chloroplasts, mitochondria, or unknown kingdoms, were removed. The annotated sequences were assigned to phylotypes according to their consensus taxonomy with which at least 80% of the sequences agreed. Closed reference operational taxonomic units (OTUs) sharing 97% identity were clustered as well and assigned taxonomy according to the Greengenes database. Lefse analysis was performed to define bacterial taxa associated with CDI and healthy controls. The relative abundance of these abundance-differential taxa identified by LefSe in pre-FMT baseline samples and post-FMT last follow-up samples were plotted using pheatmap R package.
[0101] Mouse Husbandry and Model of C. difficile Infection
[0102] Studies were conducted on 4- to 6-week old demale C57BL/6 that were reared in groups of 9. Individual mice were randomized after arrival. Mice were subjected to a previously described model of CDI.sup.34. Briefly, mice were given an antibiotic cocktail of kanamycin (0.4 mg/mL), gentamicin (0.035 mg/mL), colistin (850 U/mL), metronidazole (0.215 mg/mL), and vancomycin (0.045 mg/mL) (all antibiotics were purchased from Sigma-Aldrich, St. Louis, Mo.) in their drinking water for 3 days. Mice were then given 2 days of recovery before administration of 10.sup.7 spores of C. difficile in PBS via oral gavage. Animal grouping and research scheme were designed as shown in FIG. 4a. On day 1 post stool infusion, diarrhea was evaluated by stool water content, calculated as stool weight loss after air drying at 70.degree. C. for 4 hours. Colons were harvested, fixed in 4% formalin solution and embedded in paraffin. Sections were stained with hemotoxylin and eosin for histological assessment.
[0103] For antifungal experiment, animal grouping and research scheme were designed as shown in FIG. 15a. Mice was initially colonized with C. albicans (2.times.10.sup.8 cfu per mouse) after 3 days of antibiotic cocktail treatment in the drinking water, followed by 4 days of fluconazole treatment supplemented in the drinking water (0.5 mg/mL, Sigma). Then the mice were subjected to C. difficile administration (10.sup.7 spores per mice) through gavage after a consecutive 1.5-day antibiotic cocktail- and 1.5-day free water- drinking. Human stool infusion was performed 2 days later after C. difficile gavage. Both C. difficile load and C. albicans load were enumerated by cultivation on Day 0 before FMT and Day 1 after FMT.
C. albicans Administration and Donor Stool Infusion in Mice
[0104] C. albicans (10231, purchased from ATCC, USA) was administered to mice (2.times.10.sup.8 cfu per mouse) via gavage after 3-day antibiotic treatment or supplemented in donor stool slurry at the time of donor stool infusion. Human stool from a healthy volunteer (Chinese, male, age 28 years), without presence of C. albicans as measured by qPCR, was obtained with informed consent. For stool microbiota infusions, approximately 500 mg of stool samples were cut in an anaerobic chamber and suspended in 5 ml of phosphate-buffered saline. Mice were colonized by oral gavage of 150 .mu.l of fecal slurry with or without supplementation of C. albicans on day 2 after C. difficile challenge.
Quantification of C. difficile and C. albicans Burdens in Mouse Feces
[0105] Mouse stool were collected both before and after stool infusion. Fecal C. difficile and C. albicans burdens on day 0 before and day 1 after stool infusion were measured by cultivation. Samples were diluted in PBS and respectively plated on taurocholate cycloserine cefoxitin fructose agar (TCCFA) for quantification of C. difficile burden, on Sabouraud dextrose agar (SDA) for quantification of C. albicans load. Stool samples prior to C. albicans colonization from antibiotic-treated mice were plated on SDA to ensure that mice were C. albicans culture negative.
Data Availability
[0106] Sequence data and accompanying metadata have been deposited to the NCBI Sequence Read Archive under BioProject accession numbers PRJNA419097 and PRJNA419104.
Example 2
C. albicans Level Associated with Unfavorable FMT Outcome in IBD
[0107] The fecal C. albicans level was investigated in three IBD patients with concurrent CDI, and subsequently followed them up after FMT (FIG. 21). Disease symptoms were ameliorated soon after FMT. However, all three patients manifested unfavorable FMT outcomes at different time-points post FMT. In accordance with the finding for CDI patients, these IBD patients all showed increased fecal C. albicans levels after FMT. Taken into consideration the previous observation that C. albicans levels were also higher in IBD than in Controls (FIG. 18), it indicates that C. albicans may play an unfavorable role in IBD and IBD-FMT.
[0108] All patents, patent applications, and other publications, including GenBank Accession Numbers, cited in this application are incorporated by reference in the entirety for all purposes.
TABLE-US-00001 TABLE 1 Duration of Outcome Severe/ follow up (till last Subject Sex Age Smoking Moderate (wks) follow up) FMT1 M 80 Ex-smoker Moderate 16 Cured FMT2 M 52 No Severe 27 Cured FMT3 M 38 No Moderate 17 Cured FMT4 F 76 No Moderate 18 Cured FMT5 M 63 No Severe 18 Cured FMT6 M 88 No Severe 23 Cured FMT7 M 45 Cured FMT8 M 90 Cured FMT9 F 52 Cured FMT10 M 45 Ex-smoker Severe 20 Recurrence at week 19 FMT11 F 83 No Moderate 11 Recurrence at week 5 FMT12 F 38 No Severe 28 Recurrence at week 28 FMT13 M 81 Recurrence at week 2 FMT14 M 65 Recurrence at week 2 FMT15 F 90 Recurrence at week 4 FMT16 M 83 Recurrence at week 4 STD1 F 78 smoker Severe 14 Cured STD2 F 83 No Severe 17 Cured STD3 F 99 No Moderate 26 Cured STD4 F 85 Cured STD5 F 92 Cured STD6 M 88 Ex-smoker Severe 20 Recurrence at week 12 STD7 M 93 No Moderate 7 Recurrence at week 7 STD8 M 63 Recurrence at week 5
TABLE-US-00002 TABLE 2 Order level lefSe analysis enriched in LDA effect order group size q value o_Saccharomycetales CDI 5.26234241 0.00141993 o_Incertae_sedis Control 4.22542515 0.01743251 o_Ustilaginales Control 4.64245919 0.01113039 o_Wallemiales Control 3.86277202 0.02933565 o_Eurotiales Control 5.02946522 3.45E-05 o_Trechisporales Control 4.62024264 0.00835961 o_Agaricostilbales Control 4.77590173 0.04495598 o_Mucorales Control 3.94917134 0.00275496 o_Chaetothyriales Control 3.96218634 0.03957291 o_unidentified Control 4.43843789 0.0011891 Family level lefSe analysis enriched in LDA effect family group size q value f_Incertae_sedis CDI 5.361603211 0.00032083 f_Diatrypaceae CDI 3.647432029 0.02396599 f_Lichtheimiaceae Control 3.776192836 0.03565053 f_Marasmiaceae Control 4.460718116 0.04495598 f_Cordycipitaceae Control 3.018060628 0.01080346 f_Trichocomaceae Control 4.998286724 3.71E-05 f_Monascaceae Control 4.442880786 0.00806352 f_Ustilaginaceae Control 3.308596392 0.01113039 f_Agaricostilbaceae Control 3.637118408 0.04495598 f_unidentified Control 4.382578512 0.00989125 f_Pichiaceae Control 4.234924293 0.00153938 f_Rhizopodaceae Control 3.759990223 0.03298649 f_Pleosporaceae Control 4.266635541 0.04792052 f_Mucoraceae Control 3.546579211 0.00818474 f_Eremotheciaceae Control 3.514966042 0.01499872 f_Wallemiaceae Control 3.255471141 0.02933565 f_Hydnodontaceae Control 3.172134768 0.01944714 Genus level lefSe analysis enriched in LDA effect genus group size q value g_Candida CDI 5.382628239 0.00018873 g_Wallemia Control 3.198526789 0.02933565 g_Trechispora Control 3.55075498 0.01944714 g_Lentinula Control 3.687563833 0.04495598 g_Alternaria Control 4.061524753 0.00543258 g_Talaromyces Control 3.815612518 0.04495598 g_Aspergillus Control 4.856125262 8.75E-05 g_Pichia Control 3.729764446 0.00177011 g_Thermomyces Control 3.268072407 0.0138274 g_Rhizopus Control 3.612096939 0.03298649 g_unidentified Control 4.780192159 0.00077864 g_Simplicillium Control 2.905283827 0.01080346 g_Monascus Control 4.416347845 0.00806352 g_Mucor Control 3.444062001 0.00818474 g_Penicillium Control 4.397256252 7.64E-05 g_Sterigmatomyces Control 3.736578962 0.04495598 g_Eremothecium Control 3.41388614 0.01499872 Species level lefSe analysis enriched in species group LDA effect size q value s_Candida_albicans CDI 4.883094137 0.0128582 s_Hanseniaspora_sp CDI 2.476489287 0.02396599 s_Penicillium_sp CDI 3.159815075 0.02226132 s_Aspergillus_austroafricanus Control 2.820485891 0.02586708 s_Penicillium_dierckxii Control 3.112142685 0.04495598 s_Eurotiomycetes_sp Control 4.236581004 0.00632072 s_Pseudozyma_churashimaensis Control 2.766834362 0.04495598 s_Thermomyces_lanuginosus Control 3.158801272 0.0138274 s_Ustilaginaceae_sp Control 2.551290082 0.04335394 s_Monascus_purpureus Control 4.436483036 0.00806352 s_Penicillium_brocae Control 3.202903826 0.00668459 s_Wallemia_mellicola Control 3.029805635 0.02933565 s_Sterigmatomyces_halophilus Control 3.158900806 0.04495598 s_Eremothecium_sinecaudum Control 3.158887769 0.04495598 s_Pseudozyma_sp Control 2.865506029 0.04495598 s_Rhodotorula_dairenensis Control 3.652021075 0.04495598 s_Aspergillus_penicillioides Control 3.45521592 0.03459752 s_Ophiostomataceae_sp Control 3.730559216 0.04495598 s_Mucor_racemosus Control 2.732298798 0.00354366 s_Lichtheimiaceae_sp Control 3.462009204 0.04495598 s_Penicillium_steckii Control 2.538239261 0.03894247
TABLE-US-00003 TABLE 3 FMT/STD Randomization sample_name number baseline_comparasion Sample_collection sample_number# arm F1W0 FMT1 CDI longitudinal 3 FMT F1W2 FMT1 NA longitudinal 4 FMT F1W6 FMT1 NA longitudinal 5 FMT Control1 Donor1 Control cross-sectional 6 Donor F2W0 FMT2 CDI longitudinal 9 FMT F2W2 FMT2 NA longitudinal 10 FMT F2W4 FMT2 NA longitudinal 11 FMT Control2 Donor2 Control cross-sectional 13 Donor F3W0 FMT3 CDI longitudinal 18 FMT F3W2 FMT3 NA longitudinal 19 FMT F3W4 FMT3 NA longitudinal 20 FMT F3W10 FMT3 NA longitudinal 21 FMT F3W17 FMT3 NA longitudinal 22 FMT Control3 Donor3 Control cross-sectional 23 Donor F4W0 FMT4 CDI longitudinal 26 FMT F4W2 FMT4 NA longitudinal 27 FMT F4W4 FMT4 NA longitudinal 28 FMT F4W5 FMT4 NA longitudinal 29 FMT F4W18 FMT4 NA longitudinal 31 FMT Control4 Donor4 Control cross-sectional 32 Donor F5W0 FMT5 CDI longitudinal 38 FMT F5W2 FMT5 NA longitudinal 39 FMT F5W10 FMT5 NA longitudinal 40 FMT F5W18 FMT5 NA longitudinal 41 FMT Control5 Donor5 Control cross-sectional 42 Donor F6W0 FMT6 CDI longitudinal 43 FMT F6W2 FMT6 NA longitudinal 44 FMT F6W11 FMT6 NA longitudinal 46 FMT Control6 Donor6 Control cross-sectional 47 Donor F7W0 FMT7 CDI longitudinal 89 FMT F7W4 FMT7 NA longitudinal 90 FMT F7W12 FMT7 NA longitudinal 91 FMT Control7 Donor7 Control cross-sectional 78 Donor F8W0 FMT8 CDI longitudinal 83 FMT F8W20 FMT8 NA longitudinal 106 FMT Control8 Donor8 Control cross-sectional 79 Donor F9W0 FMT9 CDI longitudinal 127 FMT F9W2 FMT9 NA longitudinal 128 FMT F9W4 FMT9 NA longitudinal 129 FMT Control9 Donor9 Control cross-sectional 130 Donor F10W0 FMT10 CDI longitudinal 50 FMT F10W2 FMT10 NA longitudinal 51 FMT F10W6 FMT10 NA longitudinal 52 FMT F10W10 FMT10 NA longitudinal 53 FMT Control10 Donor10 Control cross-sectional 54 Donor F11W0 FMT11 CDI longitudinal 62 FMT F11W2 FMT11 NA longitudinal 63 FMT F11W4 FMT11 NA longitudinal 64 FMT Control11 Donor11 Control cross-sectional 65 Donor F12W0 FMT12 CDI longitudinal 66 FMT F12W4 FMT12 NA longitudinal 68 FMT F12W10 FMT12 NA longitudinal 69 FMT F13W0 FMT13 CDI longitudinal 82 FMT F13W2 FMT13 NA longitudinal 102 FMT F13W13 FMT13 NA longitudinal 103 FMT Contal13 Donor13 NA longitudinal 80 Donor F14W0 FMT14 CDI longitudinal 113 FMT F14W4 FMT14 NA longitudinal 115 FMT Contal14 Donor14 Control cross-sectional 119 FMT F15W0 FMT15 CDI longitudinal 131 FMT F15W2 FMT15 NA longitudinal 132 FMT F15W4 FMT15 NA longitudinal 133 FMT Contal15 Donor15 Control cross-sectional 134 Donor F16W0 FMT16 CDI longitudinal 7 FMT F16W4 FMT16 NA longitudinal 86 FMT Contal16 Donor16 Control cross-sectional 8 Donor S1W0 STD1 CDI longitudinal 33 Std therapy S1W2 STD1 NA longitudinal 34 Std therapy S1W4 STD1 NA longitudinal 35 Std therapy S2W0 STD2 CDI longitudinal 58 Std therapy S2W2 STD2 NA longitudinal 59 Std therapy S2W4 STD2 NA longitudinal 60 Std therapy S2W10 STD2 NA longitudinal 61 Std therapy S3W0 STD3 CDI longitudinal 71 Std therapy S3W2 STD3 NA longitudinal 72 Std therapy S3W4 STD3 NA longitudinal 73 Std therapy S4W0 STD4 CDI longitudinal 81 Std therapy S4W2 STD4 NA longitudinal 98 Std therapy S4W10 STD4 NA longitudinal 100 Std therapy S5W0 STD5 CDI longitudinal 107 Std therapy S5W2 STD5 NA longitudinal 108 Std therapy S5W4 STD5 NA longitudinal 109 Std therapy S5W10 STD5 NA longitudinal 110 Std therapy S5W13 STD5 NA longitudinal 111 Std therapy S5W23 STD5 NA longitudinal 112 Std therapy S6W0 STD6 CDI longitudinal 14 Std therapy S6W2 STD6 NA longitudinal 15 Std therapy S6W10 STD6 NA longitudinal 17 Std therapy S7W0 STD7 CDI longitudinal 24 Std therapy S7W2 STD7 NA longitudinal 25 Std therapy S8W0 STD8 CDI longitudinal 120 Std therapy S8W4 STD8 NA longitudinal 121 Std therapy CDI25 NA CDI cross-sectional 1 NA Control12 NA NA cross-sectional 2 NA CDI26 NA CDI cross-sectional 88 NA Control17 NA Control cross-sectional 37 NA CDI27 NA CDI cross-sectional 48 NA Control18 NA Control cross-sectional 49 NA CDI28 NA CDI cross-sectional 55 NA Control19 NA Control cross-sectional 57 NA CDI29 NA CDI cross-sectional 70 NA CDI30 NA CDI cross-sectional 74 NA Control20 NA Control cross-sectional 75 NA DI31 NA CDI cross-sectional 122 NA CDI32 NA CDI cross-sectional 124 NA Control21 NA Control cross-sectional ANS2357 NA Control22 NA Control cross-sectional ANS2331 NA Control23 NA Control cross-sectional ANS2237 NA Control24 NA Control cross-sectional ANS2467 NA time_point_post_treatment donor House Collect (FMT/STD, relationship hold ID sample_name Date week) Age Sex to patient (family ID) F1W0 27 Oct. 2015 0 80 M A F1W2 16 Nov. 2015 2 A F1W6 14 Dec. 2015 6 A Control1 27 Oct. 2015 35 F Daughter A F2W0 13 Feb. 2015 0 52 M B F2W2 6 Mar. 2015 2 B F2W4 20 Mar. 2015 4 B Control2 12 Feb. 2015 51 F Wife B F3W0 20 Mar. 2015 0 38 M C F3W2 14 Apr. 2015 2 C F3W4 28 Apr. 2015 4 C F3W10 2 Jun. 2015 10 C F3W17 28 Jul. 2015 17 C Control3 20 Mar. 2015 73 M Father C F4W0 3 Jun. 2015 0 76 F D F4W2 20 Jun. 2015 2 D F4W4 7 Jul. 2015 4 D F4W5 13 Jul. 2015 5 D F4W18 16 Oct. 2015 18 D Control4 1 Jun. 2015 53 F Daughter D F5W0 30 Jul. 2015 0 63 M E F5W2 18 Aug. 2015 2 E F5W10 19 Oct. 2015 10 E F5W18 14 Dec. 2015 18 E Control5 31 Jul. 2015 36 F E F6W0 21 Aug. 2015 0 88 M F F6W2 17 Sep. 2015 2 F F6W11 20 Nov. 2015 11 F Control6 24 Aug. 2015 41 M Son F F7W0 1 Feb. 2016 0 45 M G F7W4 7 Mar. 2016 4 G F7W12 9 May 2016 12 G Control7 8 Jan. 2016 35 M No G relationship F8W0 21 Jan. 2016 0 90 M H F8W20 20 Jun. 2016 20 H Control8 22 Jan. 2016 36 F granddaughter H F9W0 15 Sep. 2016 0 52 F I F9W2 30 Sep. 2016 2 I F9W4 14 Oct. 2016 4 I Control9 9 Sep. 2016 28 F No I relationship F10W0 26 Aug. 2015 0 45 M J F10W2 22 Sep. 2015 2 J F10W6 22 Oct. 2015 6 J F10W10 18 Nov. 2015 10 J Control10 2 Sep. 2015 21 M Son J F11W0 30 Sep. 2015 0 83 F K F11W2 18 Oct. 2015 2 K F11W4 4 Nov. 2015 4 K Control11 25 Sep. 2015 57 M Son K F12W0 24 Sep. 2015 0 38 F L F12W4 5 Nov. 2015 4 L F12W10 28 Dec. 2015 10 L F13W0 15 Jan. 2016 0 81 M M F13W2 16 Feb. 2016 2 M F13W13 28 Apr. 2016 13 M Contal13 27 Jan. 2016 43 F Daughter M F14W0 21 Mar. 2016 0 65 M N F14W4 29 Apr. 2016 4 N Contal14 22 Mar. 2016 33 M Son N F15W0 14 Sep. 2016 0 90 F O F15W2 5 Oct. 2016 2 O F15W4 18 Oct. 2016 4 O Contal15 13 Sep. 2016 52 M Son O F16W0 26 Nov. 2015 0 83 M P F16W4 8 Jan. 2016 4 P Contal16 11 Dec. 2015 27 F Maid P S1W0 14 Jul. 2015 0 78 F S1W2 24 Jul. 2015 2 S1W4 10 Aug. 2015 4 S2W0 24 Sep. 2015 0 83 F S2W2 5 Oct. 2015 2 S2W4 19 Oct. 2015 4 S2W10 30 Nov. 2015 10 S3W0 20 Oct. 2015 0 99 F S3W2 2 Nov. 2015 2 S3W4 16 Nov. 2015 4 S4W0 7 Jan. 2016 0 85 F S4W2 25 Jan. 2016 2 S4W10 21 Mar. 2016 10 S5W0 16 Mar. 2016 0 92 F S5W2 31 Mar. 2016 2 S5W4 19 Apr. 2016 4 S5W10 3 Jun. 2016 10 S5W13 17 Jun. 2016 13 S5W23 26 Aug. 2016 23 S6W0 6 Mar 2015 0 88 M S6W2 18 Mar. 2015 2 S6W10 5 May 2015 10 S7W0 7 May 2015 0 93 M S7W2 22 May 2015 2 S8W0 19 Jul. 2016 0 63 M S8W4 19 Aug. 2016 4 CDI25 0 86 F Q Control12 4 Mar. 2015 Q CDI26 30 Dec. 2015 0 88 M Control17 21 Jul. 2015 55 F CDI27 31 Aug. 2015 0 66 F R Control18 26 Aug. 2015 41 M R CDI28 7 Sep. 2015 0 84 M S Control19 8 Sep. 2015 42 M Son S CDI29 8 Oct. 2015 0 76 M CDI30 11 Dec. 2015 0 25 F T
Control20 24 Dec. 2015 33 M Brother T DI31 28 Jul. 2016 80 F CDI32 29 Aug. 2016 0 52 M Control21 Control22 Control23 Control24
TABLE-US-00004 TABLE 4 Clean. sample_num- clean_data. Dupli- data. sample_name Aer Sequencing_platform strategy length Nreads raw_reads clean_reads raw_data cation Mbp. F1W0 A3 Illumina_Miseq PE300 294 0.16 179802 81730 45.46 0 24.27 F1W2 A4 Illumina_Miseq PE300 300 0 616376 139656 22.66 0 41.9 F1W6 A5 Illumina_Miseq PE300 300 0.228 1031864 316410 30.66 0 94.92 Control1 A6 Illumina_Miseq PE300 300 0.139 433810 181802 41.91 0 54.54 F2W0 A9 Illumina_Miseq PE300 299 0.14 202246 158372 78.31 0 47.43 F2W2 A10 Illumina_Miseq PE300 300 0.13 238288 185840 77.99 0 55.75 F2W4 A11 Illumina_Miseq PE300 298 0.14 274630 230666 83.99 0 68.97 Control2 A13 Illumina_Miseq PE300 300 0.165 339154 46712 13.77 0 14.01 F3W0 A18 Illumina_Miseq PE300 300 0 231822 93316 40.25 0 27.99 F3W2 A19 Illumina_Miseq PE300 300 0.105 618984 262320 42.38 0 78.7 F3W4 A20 Illumina_Miseq PE300 300 0.13 801066 294282 36.74 0 88.28 F3W10 A21 Illumina_Miseq PE300 300 0.131 762700 277712 36.41 0 83.31 F3W17 A22 Illumina_Miseq PE300 300 0.139 696480 71662 10.29 0 21.5 Control3 A23 Illumina_Miseq PE300 300 0 207068 84600 40.86 0 25.38 F4W0 A26 Illumina_Miseq PE300 300 0 209102 67856 32.45 0 20.36 F4W2 A27 Illumina_Miseq PE300 300 0 216998 88906 40.97 0 26.67 F4W4 A28 Illumina_Miseq PE300 300 0.112 240322 93194 38.78 0 27.96 F4W5 A29 Illumina_Miseq PE300 300 0.001 132580 50098 37.79 0 15.03 F4W18 A31 Illumina_Miseq PE300 300 0 173430 49710 28.66 0 14.91 Control4 A32 Illumina_Miseq PE300 300 0.001 1708684 752020 44.01 0 225.61 F5W0 A38 Illumina_Miseq PE300 293 0.14 248084 109264 44.04 0 32.34 F5W2 A39 Illumina_Miseq PE300 297 0.14 181924 81388 44.74 0 24.25 F5W10 A40 Illumina_Miseq PE300 300 0.075 140624 54390 38.68 0 16.32 F5W18 A41 Illumina_Miseq PE300 296 0.13 192510 94050 48.85 0 27.89 Control5 A42 Illumina_Miseq PE300 300 0.208 215010 91448 42.53 0 27.43 F6W0 A43 Illumina_Miseq PE300 298 0.15 282544 219516 77.69 0 65.64 F6W2 A44 Illumina_Miseq PE300 300 0.035 298672 118338 39.62 0 35.5 F6W11 A46 Illumina_Miseq PE300 300 0.045 275570 122034 44.28 0 36.61 Control6 A47 Illumina_Miseq PE300 297 0.12 344408 244256 70.92 0 72.54 F7W0 A89 Illumina_Miseq PE300 300 0.083 257738 78176 30.33 0 23.45 F7W4 A90 Illumina_Miseq PE300 293 0.18 202056 105384 52.16 0 31.25 F7W12 A91 Illumina_Miseq PE300 300 0.068 211482 17624 8.33 0 5.29 Control7 A78 Illumina_Miseq PE300 300 0.074 321992 36754 11.41 0 11.03 F8W0 A83 Illumina_Miseq PE300 300 0 400454 105068 26.24 0 31.52 F8W20 A106 Illumina_Miseq PE300 300 0.018 329840 55682 16.88 0 16.7 Control8 A79 Illumina_Miseq PE300 300 0.037 271614 136442 50.23 0 40.93 F9W0 A127 Illumina_Miseq PE300 297 0.15 222020 181952 81.95 0 54.22 F9W2 A128 Illumina_Miseq PE300 296 0.15 303888 236462 77.81 0 69.64 F9W4 A129 Illumina_Miseq PE300 294 0.14 378688 327536 86.49 0 96.13 Control9 A130 Illumina_Miseq PE300 300 0.055 308178 107542 34.9 0 32.26 F10W0 A50 Illumina_Miseq PE300 299 0.15 365022 166948 45.74 0 49.5 F10W2 A51 Illumina_Miseq PE300 298 0.13 342394 166724 48.69 0 49.6 F10W6 A52 Illumina_Miseq PE300 294 0.14 444320 194454 43.76 0 57.07 F10W10 A53 Illumina_Miseq PE300 300 0.019 652970 251992 38.59 0 75.6 Control10 A54 Illumina_Miseq PE300 300 0.017 888528 237676 26.75 0 71.3 F11W0 A62 Illumina_Miseq PE300 293 0.15 220522 108276 49.1 0 31.89 F11W2 A63 Illumina_Miseq PE300 300 0.117 347680 112438 32.34 0 33.73 F11W4 A64 Illumina_Miseq PE300 300 0.14 265458 123800 46.64 0 36.89 Control11 A65 Illumina_Miseq PE300 300 0.076 364878 147312 40.37 0 44.19 F12W0 A66 Illumina_Miseq PE300 299 0.13 385064 168008 43.63 0 49.98 F12W4 A68 Illumina_Miseq PE300 293 0.13 239996 94998 39.58 0 28.12 F12W10 A69 Illumina_Miseq PE300 294 0.13 367404 180104 49.02 0 52.86 F13W0 A82 Illumina_Miseq PE300 300 0.015 505218 166652 32.99 0 50 F13W2 A102 Illumina_Miseq PE300 300 0.073 233864 84538 36.15 0 25.36 F13W13 A103 Illumina_Miseq PE300 297 0.14 196280 83390 42.49 0 24.64 Control13 A80 Illumina_Miseq PE300 300 0.058 306382 124604 40.67 0 37.38 F14W0 A113 Illumina_Miseq PE300 298 0.15 183402 137498 74.97 0 40.84 F14W4 A115 Illumina_Miseq PE300 297 0.12 298982 138884 46.45 0 41.11 Control14 A119 Illumina_Miseq PE300 300 0.13 205292 159234 77.56 0 47.37 F15W0 A131 Illumina_Miseq PE300 300 0.14 629728 298766 47.44 0 88.58 F15W2 A132 Illumina_Miseq PE300 299 0.15 357030 171648 48.08 0 50.81 F15W4 A133 Illumina_Miseq PE300 300 0.15 309054 151058 48.88 0 45.09 Control15 A134 Illumina_Miseq PE300 300 0 471798 185850 39.39 0 55.76 F16W0 A7 Illumina_Miseq PE300 300 0.185 346110 73132 21.13 0 21.94 F16W4 A86 Illumina_Miseq PE300 299 0.12 327154 132150 40.39 0 39.58 Control16 A8 Illumina_Miseq PE300 300 0.147 292252 39628 13.56 0 11.89 S1W0 A33 Illumina_Miseq PE300 300 0.001 770414 328156 42.59 0 98.45 S1W2 A34 Illumina_Miseq PE300 299 0.14 175118 75890 43.34 0 22.65 S1W4 A35 Illumina_Miseq PE300 298 0.15 234894 110164 46.9 0 32.83 S2W0 A58 Illumina_Miseq PE300 297 0.14 335058 249376 74.43 0 73.94 S2W2 A59 Illumina_Miseq PE300 294 0.14 405606 276022 68.05 0 81.98 S2W4 A60 Illumina_Miseq PE300 296 0.12 176070 132478 75.24 0 39.21 S2W10 A61 Illumina_Miseq PE300 300 0.068 346262 38884 11.23 0 11.67 S3W0 A71 Illumina_Miseq PE300 300 0.108 199368 44866 22.5 0 13.46 S3W2 A72 Illumina_Miseq PE300 296 0.15 269004 119998 44.61 0 35.52 S3W4 A73 Illumina_Miseq PE300 293 0.07 291644 58196 19.95 0 17.05 S4W0 A81 Illumina_Miseq PE300 300 0.015 712372 170362 23.91 0 51.11 S4W2 A98 Illumina_Miseq PE300 299 0.15 410054 186270 45.43 0 55.32 S4W10 A100 Illumina_Miseq PE300 300 0.066 223792 94782 42.35 0 28.43 S5W0 A107 Illumina_Miseq PE300 300 0 431392 221564 51.36 0 66.47 S5W2 A108 Illumina_Miseq PE300 296 0.13 209076 72748 34.8 0 21.53 S5W4 A109 Illumina_Miseq PE300 294 0.14 245218 101786 41.51 0 30.03 S5W10 A110 Illumina_Miseq PE300 293 0.14 267352 112302 42.01 0 33.07 S5W13 A111 Illumina_Miseq PE300 293 0.14 309046 123178 39.86 0 36.21 S5W23 A112 Illumina_Miseq PE300 300 0.086 407234 89072 21.87 0 26.72 S6W0 A14 Illumina_Miseq PE300 296 0.13 300734 121426 40.38 0 36.12 S6W2 A15 Illumina_Miseq PE300 294 0.14 381812 175862 46.06 0 52.14 S6W10 A17 Illumina_Miseq PE300 293 0.09 373944 109182 29.2 0 32.26 S7W0 A24 Illumina_Miseq PE300 300 0 385622 128024 33.2 0 38.41 S7W2 A25 Illumina_Miseq PE300 297 0.12 280482 115920 41.33 0 34.54 S8W0 A120 Illumina_Miseq PE300 298 0.13 211678 100168 47.32 0 29.95 S8W4 A121 Illumina_Miseq PE300 299 0.14 219782 94604 43.04 0 28.33 CDI25 A1 Illumina_Miseq PE300 297 0.13 266926 117804 44.13 0 35.16 Control12 A2 Illumina_Miseq PE300 296 0.13 380378 163882 43.08 0 48.84 CDI26 A88 Illumina_Miseq PE300 300 0.029 539830 199604 36.98 0 59.88 Control17 A37 Illumina_Miseq PE300 300 0.256 343310 112180 32.68 0 33.65 CDI27 A48 Illumina_Miseq PE300 300 0.12 386578 128450 33.23 0 38.53 Control18 A49 Illumina_Miseq PE300 297 0.13 353998 247752 69.99 0 73.09 CDI28 A55 Illumina_Miseq PE300 296 0.14 288796 124028 42.95 0 36.96 Control19 A57 Illumina_Miseq PE300 300 0.071 866640 91022 10.5 0 27.31 CDI29 A70 Illumina_Miseq PE300 298 0.12 331604 159998 48.25 0 47.52 CDI30 A74 Illumina_Miseq PE300 294 0.15 222838 163664 73.45 0 48.2 CDI31 A122 Illumina_Miseq PE300 300 0.071 218720 46710 21.36 0 14.01 CDI32 A124 Illumina_Miseq PE300 298 0.15 541002 285192 52.72 0 84.42 Control20 A136 Illumina_Miseq PE300 300 0.056 425346 154348 36.29 0 46.3 Control21 A137 Illumina_Miseq PE300 300 0.125 817664 347726 42.53 0 104.32 Control22 A138 Illumina_Miseq PE300 300 0.147 1584946 635954 40.12 0 190.79 Control23 A139 Illumina_Miseq PE300 300 0.155 540728 250244 46.28 0 75.07
TABLE-US-00005 TABLE 5 Sequencing_Strategy. Raw- Read_length. sample_name sample_NUMBER Sequencing_platform PE.SE. Reads bp. F1W0 B3 Illumina_Miseq PE250 139020 250 F1W2 B4 Illumina_Miseq PE250 453216 250 F1W6 B5 Illumina_Miseq PE250 210766 250 Control1 B6 Illumina_Miseq PE250 197162 250 F2W0 B9 Illumina_Miseq PE250 145174 250 F2W2 B10 Illumina_Miseq PE250 165058 250 F2W4 B11 Illumina_Miseq PE250 157286 250 Control2 B13 Illumina_Miseq PE250 178348 250 F3W0 B18 Illumina_Miseq PE250 392454 250 F3W2 B19 Illumina_Miseq PE250 230982 250 F3W4 B20 Illumina_Miseq PE250 112378 250 F3W10 B21 Illumina_Miseq PE250 230772 250 F3W17 B22 Illumina_Miseq PE250 311224 250 Control3 B23 Illumina_Miseq PE250 523854 250 F4W0 B26 Illumina_Miseq PE250 174036 250 F4W2 B27 Illumina_Miseq PE250 220990 250 F4W4 B28 Illumina_Miseq PE250 241624 250 F4W5 B29 Illumina_Miseq PE250 180292 250 F4W18 B31 Illumina_Miseq PE250 201408 250 Control4 B32 Illumina_Miseq PE250 421468 250 F5W0 B38 Illumina_Miseq PE250 141266 250 F5W2 B39 Illumina_Miseq PE250 144132 250 F5W10 B40 Illumina_Miseq PE250 218376 250 F5W18 B41 Illumina_Miseq PE250 238492 250 Control5 B42 Illumina_Miseq PE250 350844 250 F6W0 B43 Illumina_Miseq PE250 266526 250 F6W2 B44 Illumina_Miseq PE250 236454 250 F6W11 B46 Illumina_Miseq PE250 233562 250 Control6 B47 Illumina_Miseq PE250 305446 250 F7W0 B89 Illumina_Miseq PE250 237086 250 F7W4 B90 Illumina_Miseq PE250 235976 250 F7W12 B91 Illumina_Miseq PE250 250973 250 Control7 B78 Illumina_Miseq PE250 203468 250 F8W0 B83 Illumina_Miseq PE250 131878 250 F8W20 B106 Illumina_Miseq PE250 167805 250 Control8 B79 Illumina_Miseq PE250 242486 250 F9W0 B127 Illumina_Miseq PE250 196849 250 F9W2 B128 Illumina_Miseq PE250 204701 250 F9W4 B129 Illumina_Miseq PE250 174369 250 Control9 B130 Illumina_Miseq PE250 158498 250 F10W0 B50 Illumina_Miseq PE250 283910 250 F10W2 B51 Illumina_Miseq PE250 200238 250 F10W6 B52 Illumina_Miseq PE250 170482 250 F10W10 B53 Illumina_Miseq PE250 178994 250 Control10 B54 Illumina_Miseq PE250 824186 250 F11W0 B62 Illumina_Miseq PE250 215272 250 F11W2 B63 Illumina_Miseq PE250 195352 250 F11W4 B64 Illumina_Miseq PE250 193674 250 Control11 B65 Illumina_Miseq PE250 183434 250 F12W0 B66 Illumina_Miseq PE250 130132 250 F12W4 B68 Illumina_Miseq PE250 442748 250 F12W10 B69 Illumina_Miseq PE250 376794 250 F13W0 B82 Illumina_Miseq PE250 295522 250 F13W2 B102 Illumina_Miseq PE250 163065 250 F13W13 B103 Illumina_Miseq PE250 207191 250 Control13 B80 Illumina_Miseq PE250 462154 250 F14W0 B113 Illumina_Miseq PE250 223935 250 F14W4 B115 Illumina_Miseq PE250 259086 250 Control14 B119 Illumina_Miseq PE250 247736 250 F15W0 B131 Illumina_Miseq PE250 154491 250 F15W2 B132 Illumina_Miseq PE250 148992 250 F15W4 B133 Illumina_Miseq PE250 112623 250 Control15 B134 Illumina_Miseq PE250 253469 250 F16W0 B7 Illumina_Miseq PE250 115544 250 F16W4 B86 Illumina_Miseq PE250 281377 250 Control16 B8 Illumina_Miseq PE250 192726 250 S1W0 B33 Illumina_Miseq PE250 127724 250 S1W2 B34 Illumina_Miseq PE250 156738 250 S1W4 B35 Illumina_Miseq PE250 173534 250 S2W0 B58 Illumina_Miseq PE250 223636 250 S2W2 B59 Illumina_Miseq PE250 185150 250 S2W4 B60 Illumina_Miseq PE250 159388 250 S2W10 B61 Illumina_Miseq PE250 344816 250 S3W0 B71 Illumina_Miseq PE250 241924 250 S3W2 B72 Illumina_Miseq PE250 273290 250 S3W4 B73 Illumina_Miseq PE250 332982 250 S4W0 B81 Illumina_Miseq PE250 192778 250 S4W2 B98 Illumina_Miseq PE250 198910 250 S4W10 B100 Illumina_Miseq PE250 157771 250 S5W0 B107 Illumina_Miseq PE250 239494 250 S5W2 B108 Illumina_Miseq PE250 230334 250 S5W4 B109 Illumina_Miseq PE250 264477 250 S5W10 B110 Illumina_Miseq PE250 283660 250 S5W13 B111 Illumina_Miseq PE250 281066 250 S5W23 B112 Illumina_Miseq PE250 269234 250 S6W0 B14 Illumina_Miseq PE250 144110 250 S6W2 B15 Illumina_Miseq PE250 122768 250 S6W10 B17 Illumina_Miseq PE250 177230 250 S7W0 B24 Illumina_Miseq PE250 141758 250 S7W2 B25 Illumina_Miseq PE250 135760 250 S8W0 B120 Illumina_Miseq PE250 225001 250 S8W4 B121 Illumina_Miseq PE250 231234 250 CDI25 B1 Illumina_Miseq PE250 93298 250 Control12 B2 Illumina_Miseq PE250 127534 250 CDI26 B88 Illumina_Miseq PE250 205071 250 Control17 B37 Illumina_Miseq PE250 253674 250 CDI27 B48 Illumina_Miseq PE250 168938 250 Control18 B49 Illumina_Miseq PE250 356614 250 CDI28 B55 Illumina_Miseq PE250 144894 250 Control19 B57 Illumina_Miseq PE250 474850 250 CDI29 B70 Illumina_Miseq PE250 225218 250 CDI30 B74 Illumina_Miseq PE250 232140 250 Control20 B75 Illumina_Miseq PE250 279832 250 DI31 B122 Illumina_Miseq PE250 241976 250 CDI32 B124 Illumina_Miseq PE250 166333 250 Clean_Data. Raw_Data Read_GC Adapter_Rate Duplication_Rate N_rate sample_name . . . Clean_Reads . . . . . . . . . . . . F1W0 36.41 50614 54.77 0 -- 0 F1W2 49.63 224930 55.19 0 -- 0 F1W6 54.7 115298 54.43 0 -- 0 Control1 56.09 110582 53.81 0 -- 0 F2W0 36.12 52432 56.19 0 -- 0 F2W2 39.87 65806 53.31 0 -- 0 F2W4 51.06 80306 55.57 0 -- 0 Control2 53.95 96226 53.59 0 -- 0 F3W0 37.82 148440 51.66 0 -- 0 F3W2 44.39 102532 54.01 0 -- 0 F3W4 47.17 53006 53.36 0 -- 0 F3W10 44.67 103090 52.81 0 -- 0 F3W17 44.88 139674 54.17 0 -- 0 Control3 48.24 252724 53.82 0 -- 0 F4W0 41.06 71452 54.33 0 -- 0 F4W2 43.23 95524 55.18 0 -- 0 F4W4 36.16 87376 55.01 0 -- 0 F4W5 47.99 86526 58.15 0 -- 0 F4W18 43.77 88154 54.42 0 -- 0 Control4 49.82 209980 53.83 0 -- 0 F5W0 37.14 52462 53.82 0 -- 0 F5W2 32.13 46310 53.24 0 -- 0 F5W10 38.59 84272 54.1 0 -- 0 F5W18 36.26 86488 53.76 0 -- 0 Control5 46.31 162472 54.36 0 -- 0 F6W0 37.13 98972 54.74 0 -- 0 F6W2 44.09 104262 53.8 0 -- 0 F6W11 45.15 105462 54.06 0 -- 0 Control6 55.31 168944 54.78 0 -- 0 F7W0 79.42 188300 54.91 0 -- 0 F7W4 83.49 197021 53.01 0 -- 0 F7W12 79.33 199092 53.2 0 -- 0 Control7 50.32 102382 54.61 0 -- 0 F8W0 26.67 35168 54.94 0 -- 0 F8W20 83.11 139457 53.09 0 -- 0 Control8 58.72 142396 55.31 0 -- 0 F9W0 80.26 157992 52.39 0 -- 0 F9W2 77.69 159032 51.85 0 -- 0 F9W4 79.82 139188 53.08 0 -- 0 Control9 81.99 129950 52.62 0 -- 0 F10W0 33.77 95884 55.08 0 -- 0 F10W2 30.26 60588 55.17 0 -- 0 F10W6 38.49 65614 54.36 0 -- 0 F10W10 36.65 65606 54.59 0 -- 0 Control10 40.69 335336 53.36 0 -- 0 F11W0 42.74 92012 53.28 0 -- 0 F11W2 44.86 87634 53.31 0 -- 0 F11W4 29.48 57096 55.18 0 -- 0 Control11 47.41 86968 55.82 0 -- 0 F12W0 28.16 36646 51.56 0 -- 0 F12W4 38.11 168732 52.41 0 -- 0 F12W10 44.61 168084 54.27 0 -- 0 F13W0 35.97 106314 54.32 0 -- 0 F13W2 82.16 133971 54.28 0 -- 0 F13W13 80.73 167256 53.56 0 -- 0 Control13 48.56 224408 53.21 0 -- 0 F14W0 81.37 182212 52.37 0 -- 0 F14W4 76.58 198409 53.36 0 -- 0 Control14 78.41 194254 51.97 0 -- 0 F15W0 79.7 123134 56.75 0 -- 0 F15W2 77.43 115366 51.66 0 -- 0 F15W4 81.68 91988 55.07 0 -- 0 Control15 74.17 187995 52.93 0 -- 0 F16W0 47.42 54786 55.67 0 -- 0 F16W4 73.93 208021 51.76 0 -- 0 Control16 49.58 95562 55.47 0 -- 0 S1W0 48.49 61934 53.3 0 -- 0 S1W2 40.52 63506 53.78 0 -- 0 S1W4 38.11 66142 54.08 0 -- 0 S2W0 31.94 71434 55.8 0 -- 0 S2W2 32.01 59258 55.44 0 -- 0 S2W4 28.23 44998 55.05 0 -- 0 S2W10 39.19 135144 52.08 0 -- 0 S3W0 32.86 79506 55.38 0 -- 0 S3W2 38.59 105472 55.84 0 -- 0 S3W4 37.47 124756 53.04 0 -- 0 S4W0 45.56 87836 53.87 0 -- 0 S4W2 81.35 161811 53.93 0 -- 0 S4W10 76.87 121286 51.88 0 -- 0 S5W0 84.11 201431 55.84 0 -- 0 S5W2 82.31 189592 55.29 0 -- 0 S5W4 78.33 207172 52.18 0 -- 0 S5W10 81.24 230451 52.84 0 -- 0 S5W13 78.64 221027 55.94 0 -- 0 S5W23 76.53 206040 52.41 0 -- 0 S6W0 50.6 72918 54.66 0 -- 0 S6W2 35.01 42986 55.06 0 -- 0 S6W10 52.6 93230 53.52 0 -- 0 S7W0 41.43 58734 54.25 0 -- 0 S7W2 33.35 45270 55.72 0 -- 0 S8W0 77.52 174431 52.82 0 -- 0 S8W4 78.69 181960 52.87 0 -- 0 CDI25 29.23 27272 54.54 0 -- 0 Control12 52.3 66694 55.41 0 -- 0 CDI26 61.56 126248 53.34 0 -- 0 Control17 46.68 118412 51.69 0 -- 0 CDI27 32.75 55330 54.64 0 -- 0 Control18 48.47 172844 54.23 0 -- 0 CDI28 27.61 39998 54.42 0 -- 0 Control19 50.34 239062 55.47 0 -- 0 CDI29 28.02 63114 55.92 0 -- 0 CDI30 36.89 85644 56.9 0 -- 0 Control20 57.13 159864 54.04 0 -- 0 DI31 82.29 199112 52.56 0 -- 0 CDI32 81.76 135989 54.71 0 -- 0
REFERENCES
[0109] 1. Smits, L. P., Bouter, K. E. C., de Vos, W. M., Borody, T. J. & Nieuwdorp, M. Therapeutic Potential of Fecal Microbiota Transplantation. Gastroenterology 145, 946-953 (2013). [0110] 2. Vrieze, A., et al. Transfer of Intestinal Microbiota From Lean Donors Increases Insulin Sensitivity in Individuals With Metabolic Syndrome. Gastroenterology 143, 913-+(2012). [0111] 3. van Nood, E., et al. Duodenal Infusion of Donor Feces for Recurrent Clostridium difficile. New Engl J Med 368, 407-415 (2013). [0112] 4. Lee, C. H., et al. Frozen vs Fresh Fecal Microbiota Transplantation and Clinical Resolution of Diarrhea in Patients With Recurrent Clostridium difficile Infection A Randomized Clinical Trial. Jama-J Am Med Assoc 315, 142-149 (2016). [0113] 5. Drekonja, D., et al. Fecal Microbiota Transplantation for Clostridium difficile
[0114] Infection A Systematic Review. Ann Intern Med 162, 630-U230 (2015). [0115] 6. Colman, R. J. & Rubin, D. T. Fecal microbiota transplantation as therapy for inflammatory bowel disease: A systematic review and meta-analysis. Journal of Crohns & Colitis 8, 1569-1581 (2014). [0116] 7. De Leon, L. M., Watson, J. B. & Kelly, C. R. Transient Flare of Ulcerative Colitis After Fecal Microbiota Transplantation for Recurrent Clostridium difficile Infection. Clin Gastroenterol H 11, 1036-1038 (2013). [0117] 8. Kelly, C. R., Kahn, S. A. & Kashyap, P. Update on Fecal Microbiota Transplantation 2015: Indications, Methodologies, Mechanisms, and Outlook (vol 149, pg 223, 2015). Gastroenterology 149, 1644-1644 (2015). [0118] 9. Khoruts, A. & Sadowsky, M. J. Therapeutic transplantation of the distal gut microbiota. Mucosal Immunol 4, 4-7 (2011). [0119] 10. Manichanh, C., et al. Reshaping the gut microbiome with bacterial transplantation and antibiotic intake. Genome Res 20, 1411-1419 (2010). [0120] 11. Rea, M. C., et al. Effect of broad- and narrow-spectrum antimicrobials on Clostridium difficile and microbial diversity in a model of the distal colon. Proceedings of the National Academy of Sciences of the United States of America 108, 4639-4644 (2011). [0121] 12. Li, S. S., et al. Durable coexistence of donor and recipient strains after fecal microbiota transplantation. Science 352, 586-589 (2016). [0122] 13. Zuo, T., et al. Bacteriophage transfer during faecal microbiota transplantation in Clostridium difficile infection is associated with treatment outcome. Gut, gutjnl-2017-313952 (2017). [0123] 14. Chehoud, C., et al. Transfer of Viral Communities between Human Individuals during Fecal Microbiota Transplantation. mBio 7(2016). [0124] 15. Broecker, F., et al. Long-term changes of bacterial and viral compositions in the intestine of a recovered Clostridium difficile patient after fecal microbiota transplantation. Molecular Case Studies 2, a000448 (2016). [0125] 16. Limon, J. J., Skalski, J. H. & Underhill, D. M. Commensal Fungi in Health and Disease. Cell host & microbe 22, 156-165 (2017). [0126] 17. Iliev, I. D. & Leonardi, I. Fungal dysbiosis: immunity and interactions at mucosal barriers. Nat Rev Immunol 17, 635-646 (2017). [0127] 18. Jiang, T. T., et al. Commensal Fungi Recapitulate the Protective Benefits of Intestinal Bacteria. Cell host & microbe 22, 809-816 e804 (2017). [0128] 19. Leffler, D. A. & Lamont, J. T. Clostridium difficile Infection. New Engl J Med 373, 287-288 (2015). [0129] 20. Flevari, A., Theodorakopoulou, M., Velegraki, A., Armaganidis, A. & Dimopoulos, G Treatment of invasive candidiasis in the elderly: a review. Clin Intery Aging 8, 1199-1208 (2013). [0130] 21. Moyes, D. L., et al. Candidalysin is a fungal peptide toxin critical for mucosal infection. Nature 532, 64-+(2016). [0131] 22. Fan, D., et al. Activation of HIF-1 alpha and LL-37 by commensal bacteria inhibits Candida albicans colonization. Nat Med 21, 808-+(2015). [0132] 23. Mason, K. L., et al. Interplay between the Gastric Bacterial Microbiota and Candida albicans during Postantibiotic Recolonization and Gastritis. Infection and immunity 80, 150-158 (2012). [0133] 24. Dollive, S., et al. Fungi of the Murine Gut: Episodic Variation and Proliferation during Antibiotic Treatment. PloS one 8(2013). [0134] 25. Downward, J. R. E., Falkowski, N. R., Mason, K. L., Muraglia, R. & Huffnagle, G B. Modulation of Post-Antibiotic Bacterial Community Reassembly and Host Response by Candida albicans. Sci Rep-Uk 3(2013). [0135] 26. Mason, K. L., et al. Candida albicans and Bacterial Microbiota Interactions in the
[0136] Cecum during Recolonization following Broad-Spectrum Antibiotic Therapy.
[0137] Infection and immunity 80, 3371-3380 (2012). [0138] 27. Sokol, H., et al. Fungal microbiota dysbiosis in IBD. Gut 66, 1039-1048 (2017). [0139] 28. Sartor, R. B. & Wu, G D. Roles for Intestinal Bacteria, Viruses, and Fungi in
[0140] Pathogenesis of Inflammatory Bowel Diseases and Therapeutic Approaches. Gastroenterology 152, 327-+(2017). [0141] 29. Iliev, I. D., et al. Interactions Between Commensal Fungi and the C-Type Lectin Receptor Dectin-1 Influence Colitis. Science 336, 1314-1317 (2012). [0142] 30. Wheeler, M. L., et al. Immunological Consequences of Intestinal Fungal Dysbiosis. Cell host & microbe 19, 865-873 (2016). [0143] 31. Gweon, H. S., et al. PIPITS: an automated pipeline for analyses of fungal internal transcribed spacer sequences from the Illumina sequencing platform. Methods Ecol Evol 6, 973-980 (2015). [0144] 32. Schloss, P. D., et al. Introducing mothur: Open-Source, Platform-Independent,
[0145] Community-Supported Software for Describing and Comparing Microbial Communities. Appl Environ Microb 75, 7537-7541 (2009). [0146] 33. McDonald, D., et al. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. Isme J 6, 610-618 (2012). [0147] 34. Chen, X. H., et al. A Mouse Model of Clostridium difficile-Associated Disease. Gastroenterology 135, 1984-1992 (2008). [0148] 35. Liu, C. M., et al. FungiQuant: a broad-coverage fungal quantitative real-time PCR assay. Bmc Microbiol 12, 255 (2012).
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025

D00026

D00027

D00028

D00029

D00030

D00031

D00032

D00033

D00034

D00035

D00036

D00037

D00038

D00039

D00040

D00041

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.