Powderous lithium cobalt-based oxide compound for rechargeable lithium ion batteries and a method for making thereof
SONG; KyeongSe ; et al.
U.S. patent application number 16/931513 was filed with the patent office on 2021-01-28 for powderous lithium cobalt-based oxide compound for rechargeable lithium ion batteries and a method for making thereof. The applicant listed for this patent is Umicore, Umicore Korea Ltd.. Invention is credited to Maxime BLANGERO, KyeongSe SONG.
| Application Number | 20210028455 16/931513 |
| Document ID | / |
| Family ID | 1000004977073 |
| Filed Date | 2021-01-28 |


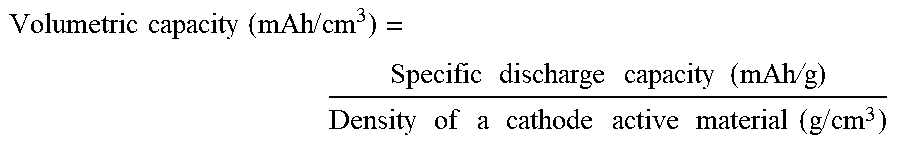

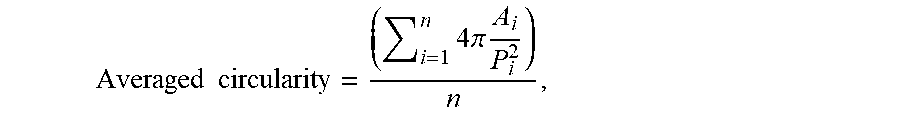
| United States Patent Application | 20210028455 |
| Kind Code | A1 |
| SONG; KyeongSe ; et al. | January 28, 2021 |
Powderous lithium cobalt-based oxide compound for rechargeable lithium ion batteries and a method for making thereof
Abstract
A lithium cobalt-based oxide cathode active material powder comprising particles having a median particle size D50 of superior or equal to 20 .mu.m, preferably 25 .mu.m, and inferior or equal to 45 .mu.m, said particles having an averaged circularity of superior or equal to 0.85 and inferior or equal to 1.00, said particles having a general formula Li.sub.1+aCo.sub.1-x-y-zAl.sub.xM'.sub.yMe.sub.zO.sub.2, wherein M' and Me comprise at least one element of the group consisting of: Ni, Mn, Nb, Ti, W, Zr, and Mg, with -0.01.ltoreq.a.ltoreq.0.01, 0.002.ltoreq.x.ltoreq.0.050, 0.ltoreq.y.ltoreq.0.020 and 0.ltoreq.z.ltoreq.0.050, the cathode active material powder having a specific floating capacity of at most 80 mAh/g.
| Inventors: | SONG; KyeongSe; (Chungcheongnam-do, KR) ; BLANGERO; Maxime; (Chungcheongnam-do, KR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004977073 | ||||||||||
| Appl. No.: | 16/931513 | ||||||||||
| Filed: | July 17, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62877364 | Jul 23, 2019 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | H01M 4/364 20130101; H01M 4/0471 20130101; H01M 10/0525 20130101; H01M 4/525 20130101; H01M 2004/021 20130101; H01M 4/131 20130101; H01M 4/505 20130101 |
| International Class: | H01M 4/525 20060101 H01M004/525; H01M 4/505 20060101 H01M004/505; H01M 4/36 20060101 H01M004/36; H01M 4/04 20060101 H01M004/04; H01M 4/131 20060101 H01M004/131; H01M 10/0525 20060101 H01M010/0525 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 23, 2019 | IB | PCT/IB2019/056282 |
| Jul 23, 2019 | IB | PCT/IB2019/056284 |
Claims
1. A lithium cobalt-based oxide cathode active material powder, which comprises particles having a median particle size D50 of superior or equal to 20.00 .mu.m, preferably 25.00 .mu.m, and inferior or equal to 45.00 .mu.m, said particles having an averaged circularity of superior or equal to 0.85 and inferior or equal to 1.00, said particles having a general formula Li.sub.1+aCo.sub.1-x-y-zAl.sub.xM'.sub.yMe.sub.zO.sub.2, wherein M' and Me comprise at least one element of the group consisting of: Ni, Mn, Nb, Ti, W, Zr, and Mg, with -0.01.ltoreq.a.ltoreq.0.01, 0.002.ltoreq.x.ltoreq.0.050, 0.ltoreq.y.ltoreq.0.020 and 0.ltoreq.z.ltoreq.0.050, said lithium cobalt-based oxide active material powder being obtained by a process comprising the steps of: a. preparing a first mixture comprising: a Li source, a first Co-bearing precursor, optionally a M' source, and an Al source, said first mixture having a Li to (Co+Al+M') molar ratio superior or equal to 1.03 and inferior or equal to 1.10, b. sintering said first mixture at a temperature of superior or equal to 950.degree. C. and inferior or equal to 1100.degree. C. in an oxygen containing atmosphere such as air, so as to obtain a first sintered agglomerated powder, and c. milling and screening the first sintered agglomerated powder so as to obtain an intermediate powder having a general formula Li.sub.1+a'Co.sub.1-x'-y'Al.sub.x'M'.sub.y'O.sub.2, M' being at least one element of the group consisting of: Ni, Mn, Nb, Ti, W, Zr, and Mg, with 0.03.ltoreq.a'.ltoreq.0.10, 0.002.ltoreq.x'.ltoreq.0.050, and 0.ltoreq.y'.ltoreq.0.02, and comprising particles having a D50 of superior or equal to 20.00 .mu.m, preferably 25.00 .mu.m, and inferior or equal to 45.00 .mu.m and an averaged circularity of superior or equal to 0.85 and inferior or equal to 1.00, d. mixing the intermediate powder with a second Co-bearing precursor and optionally, with a source of Me, to prepare a second mixture, wherein the Li to (Co+Al+M') or the Li to (Co+Al+M'+Me) molar ratio in said second mixture is superior or equal to 0.99 and inferior or equal to 1.01, e. sintering said second mixture at a temperature of superior or equal to 800.degree. C. and inferior or equal to 1100.degree. C. in an oxygen containing atmosphere, such as air, so as to obtain a second sintered agglomerated powder, and f. milling and screening said second sintered agglomerated powder so as to obtain the cathode active material powder according to the invention, the cathode active material powder having a specific floating capacity of at most 80 mAh/g obtained by the steps a. to f.
2. The lithium cobalt-based oxide cathode active material powder according to claim 1, having a press density superior or equal to 3.95 g/cm.sup.3 and inferior or equal to 4.40 g/cm.sup.3.
3. The lithium cobalt-based oxide cathode active material powder according to claim 1, having a volumetric capacity of at least 570 mAh/cm.sup.3, preferably of at most 700 mAh/cm.sup.3 .
4. The lithium cobalt-based oxide cathode active material powder according to claim 1, wherein said particles have a R-3m crystal structure.
5. The lithium cobalt-based oxide cathode active material powder according to claim 1, comprising particles having an averaged circularity of superior or equal to 0.90 and inferior or equal to 1.00, preferably of superior or equal to 0.85 and of inferior or equal to 0.95.
6. The lithium cobalt-based oxide cathode active material powder according to claim 1, wherein y and z=0.
7. A lithium-ion secondary battery suitable for electronic devices comprising the lithium cobalt-based oxide cathode active material powder according to claim 1.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to U.S. Provisional Application No. 62/877,364, filed Jul. 23, 2019; International Application No. PCT/IB2019/056282, filed Jul. 23, 2019; and International Application No. PCT/IB2019/056284, filed Jul. 23, 2019. The entire contents of each are incorporated by reference herein.
TECHNICAL FIELD AND BACKGROUND
[0002] This invention relates to a lithium cobalt-based oxide (LCO) cathode active material powder for lithium-ion secondary batteries (LIBs) suitable for portable electronic device applications.
[0003] As the functionalities and performances of portable electronic devices are constantly improving, LIBs having a higher volumetric energy density are required.
[0004] The volumetric energy density of a cathode active material powder is obtained according to a following equation:
Volumetric energy density (mAh/cm.sup.3)=volumetric capacity (mAh/cm.sup.3).times.Charge cutoff voltage (V),
, wherein:
Volumetric capacity ( mAh / cm 3 ) = Specific discharge capacity ( mAh / g ) Density of a cathode active material ( g / cm 3 ) ##EQU00001##
[0005] A higher charge cutoff voltage (such as superior or equal to 4.5V vs. Li.sup.+/Li reference potential) leads to a significant increase of the volumetric energy density of a cathode material powder.
[0006] It is therefore an object of the present invention to provide a lithium cobalt-based oxide cathode active material powder for lithium-ion secondary batteries, having an improved volumetric capacity of at least 570 mAh/cm.sup.3 obtained by the analytical methods of the present invention.
[0007] In addition to the improved volumetric capacity, the LCO cathode active material compound according to the present invention must have a sufficient structural stability at a voltage superior or equal to 4.5V so far. Such a sufficient stability is indicated by a specific floating capacity of at most 80 mAh/g (obtained by the analytical methods of the present invention) during the use of the cathode active material powder in a LIB.
SUMMARY OF THE INVENTION
[0008] This objective is achieved by providing a lithium cobalt-based oxide cathode active material powder according to claim 1.
[0009] It is indeed observed that an improved volumetric capacity of higher than 570 mAh/cm.sup.3 and a specific floating capacity of lower than 80 mAh/g, as illustrated in the results provided in Table 2, are achieved with a battery using a LCO cathode material powder according to EX1, having the following features: [0010] a median particle size D50 of 38.00 .mu.m, [0011] an averaged circularity of 0.87, and [0012] resulting from a double-sintering process wherein the Li/(Co+Al)=1.04 when S1 (at a T.degree. C.=1000.degree. C.) is applied and wherein the Li/(Co+Al)=1.00 when S2 (at a T.degree. C.=980.degree. C.) is applied.
[0013] The cathode active material powder comprises particles having an Al to (Co+Al+M'+Me) molar ratio (x) inferior or equal to 0.050 so as to minimize a capacity loss, and superior or equal to 0.002 so as to stabilize a crystal-structure of the LCO cathode active material powder during cycling.
[0014] The cathode active material powder comprises particles having a Li to (Co+Al+M'+Me) molar ratio (1+a) superior or equal to 0.99 and inferior or equal to 1.01, preferably superior or equal to 0.995 and inferior or equal to 1.005.
[0015] If the ratio 1+a is less than 0.99 (a<-0.01), a Co dissolution at a higher voltage such as 4.50V occurs since there is no enough Li to hold the cobalt atoms in the structure of the cathode active material particles and the capacity of the cathode active material powder decreases. If the ratio 1+a is more than 1.01 (a>0.01), the cycle life of the cathode active material powder deteriorates.
[0016] In the framework of the present invention, the D50 is the volumetric median particle size and is superior or equal to 20.00 .mu.m, preferably 25.00 .mu.m, and inferior or equal to 45 .mu.m. Preferably, the cathode active material powder according to the present invention has a D50 superior or equal to 30.00 .mu.m and inferior or equal to 40.00 .mu.m.
[0017] Due a larger D50 of the LCO cathode active material powder according to the invention, in comparison with conventional D50 values (less than 20.00 .mu.m) for this type of cathode active material, the claimed LCO cathode active material powder shows packing density values which are much higher than the conventional ones. The D50 should however be less than 45.00 .mu.m, because surface scratching of the cathode during its preparation from the LCO cathode active material powder is observed for D50 values higher than this upper limit.
[0018] The present invention concerns the following embodiments:
Embodiment 1
[0019] In a first aspect, the present invention concerns a lithium cobalt-based oxide cathode active material powder, which comprises particles having a median particle size D50 of superior or equal or superior to 20.00 .mu.m+/-1.00 .mu.m, preferably 25.00 .mu.m+/-1.00 .mu.m, and inferior or equal to 45.00 .mu.m+/-1.00 .mu.m, said particles having an averaged circularity of superior or equal to 0.85 and inferior or equal to 1.00, said particles having a general formula Li.sub.1+aCo.sub.1-x-y-zAl.sub.xM'.sub.yMe.sub.zO.sub.2, wherein M' and Me comprise at least one element of the group consisting of: Ni, Mn, Nb, Ti, W, Zr, and Mg, with -0.01.ltoreq.a.ltoreq.0.01, 0.002.ltoreq.x.ltoreq.0.050, 0.ltoreq.y.ltoreq.0.020 and 0.ltoreq.z.ltoreq.0.050, said lithium cobalt-based oxide active material powder being obtained by a process comprising the steps of: [0020] a. preparing a first mixture comprising: a Li source, a first Co-bearing precursor, optionally a M' source, and an Al source, said first mixture having a Li to (Co+Al+M') molar ratio superior or equal to 1.03 and inferior or equal to 1.10, [0021] b. sintering (step S1) said first mixture at a temperature of superior or equal to 950.degree. C. and inferior or equal to 1100.degree. C. in an oxygen containing atmosphere such as air, so as to obtain a first sintered agglomerated powder, and [0022] c. milling and screening the first sintered agglomerated powder so as to obtain an intermediate powder (LCO1) having a general formula Li.sub.1+a'Co.sub.1-x'-yAl.sub.x'M'.sub.y'O.sub.2, M' being at least one element of the group consisting of: Ni, Mn, Nb, Ti, W, Zr, and Mg, with 0.03.ltoreq.a'.ltoreq.0.10, 0.002.ltoreq.x.ltoreq.0.050, and 0.ltoreq.y'.ltoreq.0.02, and comprising particles having a D50 of superior or equal to 20.00 .mu.m, preferably 25.00 .mu.m, and inferior or equal to 45.00 .mu.m, and an averaged circularity of superior or equal to 0.85 and inferior or equal to 1.00, [0023] d. mixing the intermediate powder with a second Co-bearing precursor and optionally, with a source of Me, to prepare a second mixture, wherein the Li to (Co+Al+M') or the Li to (Co+Al+M'+Me) molar ratio in said second mixture is superior or equal to 0.99 and inferior or equal to 1.01, [0024] e. sintering (step S2) said second mixture at a temperature of superior or equal to 800.degree. C. and inferior or equal to 1100.degree. C. in an oxygen containing atmosphere, such as air, so as to obtain a second sintered agglomerated powder, and [0025] f. milling and screening said first sintered agglomerated powder so as to obtain the cathode active material powder according to the invention (LCO2), said cathode active material powder having a specific floating capacity of at most 80 mAh/g obtained by the steps a. to f.
[0026] Preferably, y and z=0.
[0027] In the Embodiment 1 according to the invention, the D50 value and the averaged circularity value of the particles of the intermediate powder (or the first sintered agglomerated powder after milling and screening--step c.) are similar to the D50 value and the averaged circularity values of the particles of the lithium cobalt-based oxide cathode active material powder according to the invention.
[0028] The D50 is a volumetric-based value (see section 1.1 below) expressed in .mu.m+/-0.01 .mu.m.
[0029] The averaged circularity is a number-based value (see section 1.7 below).
Embodiment 2
[0030] Preferably, the cathode active material powder of the Embodiment 1 has a press density superior or equal to 3.95 g/cm.sup.3 and inferior or equal to 4.40 g/cm.sup.3.
Embodiment 3
[0031] More preferably, the cathode active material powder according to the Embodiment 1 or 2 has a volumetric capacity of at least 570 mAh/cm.sup.3, preferably of at most 700 mAh/cm.sup.3.
Embodiment 4
[0032] In a fourth Embodiment, the cathode active material powder according to any of the preceding Embodiments, wherein said particles have an averaged circularity of superior or equal to 0.90 and inferior or equal to 1.00, preferably of superior or equal to 0.95 and inferior or equal to 1.00, more preferably of superior or equal to 0.85 and inferior or equal to 0.95, most preferably of superior or equal to 0.90 and inferior or equal to 0.95.
[0033] In said fourth Embodiment, the intermediate powder has particles having an averaged circularity of superior or equal to 0.90 and inferior or equal to 1.00, preferably of superior or equal to 0.95 and inferior or equal to 1.00, more preferably of superior or equal to 0.85 and inferior or equal to 0.95, most preferably of superior or equal to 0.90 and inferior or equal to 0.95.
Embodiment 5
[0034] Preferably, in a fifth Embodiment according to any of the preceding Embodiments, the first Co-bearing precursor has a D50 superior or equal to 20.00 .mu.m+/-1.00 .mu.m, preferably 25.00 .mu.m+/-1.00 .mu.m, and inferior or equal to 45 .mu.m+/-1.00 .mu.m.
[0035] More preferably, the first Co-bearing precursor has a D50 superior or equal to 35.00 .mu.m+/-1.00 .mu.m, and inferior or equal to 45.00 .mu.m+/-1.00 .mu.m, so that the lithium cobalt-based oxide cathode active material powder comprises particles having a median particle size D50 of superior or equal to 35.00 .mu.m+/-1.00 .mu.m, and inferior or equal to 45.00 .mu.m+/-1.00 .mu.m.
[0036] If the D50 of the first Co-bearing precursor is inferior to 20.00 .mu.m+/-1.00 .mu.m, it is required to increase the Li to (Co+Al+M') molar ratio of the second mixture or to sinter said second mixture at a temperature superior to 1100.degree. C.
[0037] Optionally, the first Co-bearing precursor contains Al and M'.
[0038] Preferably, the second Co-bearing precursor has a D50 inferior to 10.00 .mu.m, more preferably inferior to 5.00 .mu.m to maximize the volumetric density of the second cathode active material according to the Embodiment 1.
[0039] Li sources can be either one or more of Li.sub.2O, LiOH, LiOH.H.sub.2O, Li.sub.2CO.sub.3, and LiNO.sub.3.
[0040] Co-bearing precursors can be either one or more of CoO.sub.z, CoCO.sub.3, CoO(OH), and Co(OH).sub.2.
Embodiment 6
[0041] Preferably, in a sixth Embodiment according to any of the preceding Embodiments, the first sintering step is performed during a period of at least 3 hours and at most 20 hours.
Embodiment 7
[0042] Preferably, in a seventh Embodiment according to any of the preceding
[0043] Embodiments, the second sintering step is performed during a period of at least 1 hour and at most 20 hours.
BRIEF DESCRIPTION OF THE FIGURES
[0044] FIG. 1: Morphology of EX1.
DETAILED DESCRIPTION
[0045] The invention is further illustrated in the following examples:
1. DESCRIPTION OF ANALYSIS METHODS
[0046] 1.1. Particle Size distribution
[0047] The D50 is an indicator of a powder particle size distribution (hereafter referred to as psd) and is obtained by a laser psd measurement method. In this invention, the laser psd is measured by using a Malvern Mastersizer 2000 with Hydro 2000MU wet dispersion accessory, e.g. after having dispersed the powder in an aqueous medium. In order to improve the dispersion of the powder in the aqueous medium, sufficient ultrasonic irradiation and stirring are applied and an appropriate surfactant is introduced in the aqueous medium.
[0048] If the powder according to the invention has a multimodal psd profile, then said multimodal profile is deconvoluted, then if one or several deconvoluted modes having a D50 comprised in the 20.00 .mu.m, preferably 25.00 .mu.m, and 45.00 .mu.m range are identified, said powder has a D50 according to claim1.
[0049] If the powder according to the present invention has a monomodal psd profile with a single mode having a D50 comprised in the in the 20.00 .mu.m, preferably 25.00 .mu.m, and 45.00 .mu.m range, said powder has therefore a D50 according to claim1.
[0050] 1.2. Pressed Density
[0051] The pressed density (PD) is measured according to the following procedure: 3 grams of a LCO cathode active material powder is filled into a pellet die with a diameter "d" of 1.3 cm. A pressure of 207 MPa is applied for 30 seconds. After relaxing the load, the thickness "t" of the pressed LCO cathode active material powder is measured. The pressed density PD is 3 g divided by the volume of the pressed powder (.pi..times.(d/2).sup.2.times.t).
[0052] 1.3. Inductively Coupled Plasma
[0053] The inductively coupled plasma (ICP) method is used to measure the content of elements such as Li, Co, and Al by using an Agillent ICP 720-ES device.
[0054] 2 g of a powder sample is dissolved in 10 mL high purity hydrochloric acid in an Erlenmeyer flask. The flask is covered by a glass and heated on a hot plate until complete dissolution of the precursor is achieved. After being cooled to the room temperature, the solution is moved to a 100 mL volumetric flask. After having filled the flask with the solution, the volumetric flask is filled with deionized water up to the 100 mL mark. 5 mL of the resulting solution is transferred into a 50 mL volumetric flask for a 2.sup.nd dilution, where the volumetric flask is filled with 10% hydrochloric acid up to the 50 mL mark and then homogenized. Finally, this 50 mL solution is used in the ICP measurement.
[0055] 1.4. High Angular Resolution Synchrotron X-Ray Diffraction
[0056] High angular resolution synchrotron powder x-ray diffraction (SXRD) is carried out on the BL04-MSPD beamline of the ALBA synchrotron (Cerdanyola del Valles, Spain). All powders were packed in 0.5 mm diameter capillaries. The typical 20 angular range was from 0.degree. to 70.degree. with 0.006.degree. angular step and 3 minutes accumulation time. The patterns were recorded in a Debye-Scherrer geometry with a wavelength of .lamda.=0.825.ANG.+/-0.010.ANG..
[0057] 1.5. Crystallographic Characterization
[0058] Inorganic Crystal Structure Database (ICSD, provided by FIZ Karlsruhe and the U.S. Secretary of Commerce) contains information on all inorganic crystal-structures published since 1913. peak positions in the obtained diffraction pattern and the elements in a powder sample (e.g. Li, Co, O, Al) are searched in the ICSD so as to determine a crystal-structure of a power sample.
[0059] 1.6. Electrochemical Analysis: Capacity and a Floating Test Analysis
[0060] 1.6.1. Coin Cells Preparation
[0061] Coin cells that are used in a discharge capacity and floating test analysis are assembled according to the following steps:
[0062] Step 1) Preparation of a Cathode:
[0063] A slurry that contains the solids: a LCO cathode active material powder, a conductor (Super P, Timcal) and a binder (KF #9305, Kureha) in a weight ratio 90:5:5, and a solvent (NMP, Sigma-Aldrich) are mixed in a high speed homogenizer so as to obtain a homogenized slurry. The homogenized slurry is spread on one side of an aluminum foil using a doctor blade coater with a 230 .mu.m gap. the slurry-coated aluminum foil is dried in an oven at 120.degree. C., then pressed using a calendaring tool, and dried again in a vacuum oven to remove the solvent completely.
[0064] Step 2) Coin Cell Assembly:
[0065] A coin cell is assembled in a glovebox which is filled with an inert gas (argon). For the discharge capacity analysis, a separator (Celgard) is located between the cathode and a piece of lithium foil used as an anode. For the floating test, two pieces of separator are located between the cathode and an anode, which consists of graphite. 1M LiPF.sub.6 in EC:DMC (1:2 in volume) is used as electrolyte and dropped between separator and electrodes. Then, the coin cell is completely sealed to prevent leakage of electrolyte.
[0066] 1.6.2. Discharge Capacity Analysis
[0067] The first charge and discharge capacity (CQ1 and DQ1) are measured by constant current mode with 0.1C rate, where 1C is defined as 160 mAh/g and charge cutoff voltage is 4.30V and discharge cutoff voltage is 3.0V. The volumetric discharge capacity DQ1V (mAh/cm.sup.3) is obtained according to multiplying DQ1 by PD.
[0068] 1.6.3. Floating Test Analysis
[0069] The floating test analyses the crystal-stability of LCO compounds at a high voltage at an elevated temperature.
[0070] The prepared coin cell is tested according to the following charge protocol: the coin cell is first charged to 4.5V at constant current mode with C/20 rate (1C=160 mAh/g) in a 50.degree. C. chamber. The coin cell is then kept at constant voltage (4.5V) for 5 days (120 hours), which is a very severe condition.
[0071] Once side reactions or metal dissolution happen, there will be a voltage drop. The electrochemical instrument will automatically compensate the (loss of) current to keep the voltage constant. Therefore, the recorded current is a measure of the ongoing side reactions during cycling.
[0072] The specific floating capacity (QF) is the total amount capacity (mAh/g) during the floating test. After the floating test, the coin cell is disassembled. The anode and the separator (localized close to the anode) are analyzed by ICP for a metal dissolution analysis. The measured cobalt content is normalized by the total amount of active material in the electrode so that a specific cobalt dissolution value (Co.sub.Dis) is obtained.
[0073] 1.7. Morphology Analysis
[0074] The morphology of a powder sample is analyzed with a Scanning Electron Microscopy (SEM) technique. The measurement is performed with a JEOL JSM-6000. An image of the powder sample is recorded with a magnification of 500 times to demonstrate the averaged circularity of the powder sample particles. In the SEM image, ten particles are selected and the circularity of these particles is calculated as follows:
Circularity = 4 .pi. A P 2 ##EQU00002##
, wherein A is an area of a particle, P is a perimeter of a particle, these parameters being obtained using an ImageJ software (reference is made to the Sections 30.2 to 30.7--"Set measurement" of the Image J User Guide version IJ 1.46r).
[0075] The averaged circularity according to the invention may be expressed as follows:
Averaged circularity = ( i = 1 n 4 .pi. A i P i 2 ) n , ##EQU00003##
wherein n is the number of particles i analyzed according to the below-provided protocol. The averaged circularity is then a number-based average value.
[0076] A sufficient number of particles is at least 10 for a SEM image recorded with a magnification of 500 times. The at least 10 particles have a size of at least 20.00 .mu.m.
[0077] As mentioned above, the calculation of the circularity implies the measurement of: [0078] i) The perimeter which is effected by: a) determining an outside boundary of the SEM image of a particle, by b) decomposing the outside boundary into individual segment-based selections, each of these selections having an individual perimeter, and by c) adding the values of the lengths of the individual perimeters so as to obtain the value of the perimeter of a particle; and [0079] ii) The area which is done by: adding a plurality of pixel areas included in a surface defined by the outside boundary.
[0080] An averaged circularity of 1.00 means that the particles representative of a sample have a spherical shape.
[0081] An averaged circularity inferior to 1.00 means that the particles representative of a sample have a non-spherical shape.
[0082] An averaged circularity superior to 0 and inferior to 1 refers to an ellipsoidal shape.
[0083] The invention is further illustrated in the following examples:
2. EXAMPLES AND COMPARATIVE EXAMPLES
Example 1
[0084] A CoCO.sub.3 powder having a D50 of 38.00 .mu.m and an Al.sub.2O.sub.3 powder are mixed so as to obtain a mixture having an Al to (Co+Al) molar ratio of 0.04 and the mixture is heated at 600.degree. C. for 3 hours under a flow of air to prepare an Al coated Co oxide "CAO1". The CAO1 powder and Li.sub.2CO.sub.3 are mixed so as to obtain a mixture having a Li to (Co+Al) molar ratio of 1.04 and the mixture is heated at 1000.degree. C. for 10 hours under a flow of air. The sintered powder is grinded and named LCO1A-EX1 having a general formula of Li.sub.1.04Co.sub.0.96Al.sub.0.04O.sub.2 and a D50 of 37.00 .mu.m.
[0085] LCO1B-EX1, which is prepared by a same procedure as LCO1A-EX1 except that the Li to (Co+Al) molar ratio is 1.06, has a general formula of Li.sub.1.06Co.sub.0.96Al.sub.0.04O.sub.2 and a D50 of 39.00 .mu.m.
[0086] A Co.sub.3O.sub.4 powder having a D50 of 3.00 .mu.m and Al.sub.2O.sub.3 powder are mixed so as to obtain a mixture having an Al to (Co+Al) molar ratio of 0.04, and the mixture is heated at 1000.degree. C. for 10 hours under a flow of air to prepare an Al coated Co oxide "CAO2".
[0087] LCO1A-EX1 and CAO2 are mixed to prepare EX1A having a general formula Li.sub.1.00Co.sub.0.96Al.sub.0.04O.sub.2. The mixture is heated at 980.degree. C. for an hour under a flow of air. The sintered powder is grinded and named EX1A.
[0088] EX1B is prepared by a same procedure as EX1A except that LCO1B-EX1 is used instead of LCO1A-EX1.
[0089] EX1A and EX1B are according to the present invention.
Comparative Example 1
[0090] CAO2 and Li.sub.2CO.sub.3 are mixed so as to obtain a mixture having an Al to (Co+Al) molar ratio of 1.00, and the mixture is heated at 1000.degree. C. for 10 hours under a flow of air. The sintered powder is grinded and named LCO1A-CEX1 which has a general formula Li.sub.1.00Co.sub.0.96Al.sub.0.04O.sub.2 and a D50 of 4 .mu.m.
[0091] LCO1B-CEX1, LCO1C-CEX1, and LCO1D-CEX1 are prepared by a same procedure as LCO1A-CEX1 except that the Li to (Co+Al) molar ratios in the mixture are 1.02, 1.04, and 1.06, respectively. The general formulas of LCO1B-CEX1, LCO1C-CEX1, and LCO1D-CEX1 are Li.sub.1.02Co.sub.0.96Al.sub.0.04O.sub.2, Li.sub.1.04Co.sub.0.96Al.sub.0.04O.sub.2, and Li.sub.1.06Co.sub.0.96Al.sub.0.04O.sub.2, respectively. The D50 of LCO1B-CEX1, LCO1C-CEX1, and LCO1D-CEX1 are 8 .mu.m, 15 .mu.m, 20 .mu.m, respectively.
[0092] LCO1A-CEX1 is heated at 980.degree. C. for an hour under a flow of air. The sintered powder is grinded and named CEX1A which has a general formula Li.sub.1.00Co.sub.0.96Al.sub.0.04O.sub.2.
[0093] LCO1B-CEX1 and CAO2 are mixed so as to obtain a mixture having Li to (Co+Al) molar ratio of 1.00. The mixture is heated at 980.degree. C. for an hour under a flow of air. The sintered powder is grinded and named CEX1B having a general formula Li.sub.1.00Co.sub.0.96Al.sub.0.04O.sub.2.
[0094] CEX1C and CEX1D are prepared by a same procedure as CEX1B except that LCO1C-CEX1 and LCO1D-CEX1 are used instead of LCO1B-CEX1.
[0095] CEX1A, CEX1B, CEX1C, and CEX1D are not according to the present invention.
3. DISCUSSION
[0096] Table 1 shows the key preparation conditions of the LCO cathode active material powders according to Example 1 and Comparative example 1. EX1A and EX1B are prepared by the two sintering steps according to the method claimed in the present invention. The methods to prepare CEX1A and CEX1B are not according to the present invention because neither the D50 of Co precursor of LCO1 is superior to 20 .mu.m nor the ratio 1+a' is superior or equal to 1.03. The methods to prepare CEX1C and CEX1D are also not according to the present invention because the D50 of LCO1 is not superior to 20 .mu.m.
[0097] Table 2 shows analytical results, obtained according to the analysis method described in the section 1.2. Pellet density, 1.6.2. discharge capacity analysis, 1.6.3. floating test analysis, and 1.4. cross-SEM Al EDX mapping analysis, of LCO compounds in Example 1, Comparative example 1, and Comparative example 2.
[0098] DQ1V corresponds the volumetric capacity of batteries. The parameters QF and Co.sub.Dis are obtained by the floating test (cfr. section 1.6.3) and are indicators of the crystal-structural stability at a high voltage such as 4.50V or higher. QF and Co.sub.Dis should be as low as possible.
[0099] EX1A and EX1B have lower QF and Co.sub.Dis as well as higher DQ1V.
TABLE-US-00001 TABLE 1 Formula of LCO1 and D50 of Co precursor or LCO1 D50 of Co precursor of Crystal Averaged Example ID LCO1 ID Formula of LCO1 LCO1 (.mu.m) structure* circularity EX1A LCO1A-EX1 Li.sub.1.04Co.sub.0.96Al.sub.0.04O.sub.2 38 R-3 m 0.87 EX1B LCO1B-EX1 Li.sub.1.06Co.sub.0.96Al.sub.0.04O.sub.2 38 R-3 m 0.91 CEX1A LCO1A-CEX1 Li.sub.1.00Co.sub.0.96Al.sub.0.04O.sub.2 3 R-3 m 0.68 CEX1B LCO1B-CEX1 Li.sub.1.02Co.sub.0.96Al.sub.0.04O.sub.2 3 R-3 m 0.77 CEX1C LCO1C-CEX1 Li.sub.1.04Co.sub.0.96Al.sub.0.04O.sub.2 3 R-3 m 0.83 CEX1D LCO1D-CEX1 Li.sub.1.06Co.sub.0.96Al.sub.0.04O.sub.2 3 R-3 m 0.81 *acquired by SXRD (cfr. sections 1.4 and 1.5)
TABLE-US-00002 TABLE 2 Analytical results Related Psd Li/(Co + Sintering temperature Electrochemical property Example to the D50 Al) (.degree. C.) PD DQ1V QF CO.sub.Dis IDs invention (.mu.m) S1 S2 S1 S2 (g/cm.sup.3) (mAh/cm.sup.3) (mAh/g) (mg/g) EX1A Yes 38 1.04 1.00 1000 980 4.0 584.0 76 9 EX1B Yes 40 1.06 1.00 1000 980 4.1 583.2 73 10 CEX1A No 5 1.00 1.00 1000 980 3.3 508.2 169 28 CEX1B No 9 1.02 1.00 1000 980 3.4 518.8 99 14 CEX1C No 16 1.04 1.00 1000 980 3.6 542.7 70 9 CEX1D No 21 1.06 1.00 1000 980 3.7 552.0 69 8
[0100] The present invention is covered by the following clauses:
[0101] 1. A lithium cobalt-based oxide cathode active material powder, which comprises particles having a median particle size D50 of superior or equal to 20.00 .mu.m+/-1.00 .mu.m, preferably 25.00 .mu.m+/-1.00 .mu.m, and inferior or equal to 45.00 .mu.m+/-1.00 .mu.m, said particles having an averaged circularity of superior or equal to 0.85 and inferior or equal to 1.00, said particles having a general formula Li.sub.1+aCo.sub.1-x-y-zAl.sub.xM'.sub.yMe.sub.zO.sub.2, wherein M' and Me comprise at least one element of the group consisting of: Ni, Mn, Nb, Ti, W, Zr, and Mg, with -0.01.ltoreq.a.ltoreq.0.01, 0.002.ltoreq.x.ltoreq.0.050, 0.ltoreq.y.ltoreq.0.020 and 0.ltoreq.z.ltoreq.0.050, said lithium cobalt-based oxide active material powder being obtained by a process comprising the steps of: [0102] a. preparing a first mixture comprising: a Li source, a first Co-bearing precursor, optionally a M' source, and an Al source, said first mixture having a Li to (Co+Al+M') molar ratio superior or equal to 1.03 and inferior or equal to 1.10, [0103] b. sintering said first mixture at a temperature of superior or equal to 950.degree. C. and inferior or equal to 1100.degree. C. in an oxygen containing atmosphere such as air, so as to obtain a first sintered agglomerated powder, and [0104] c. milling and screening the first sintered agglomerated powder so as to obtain an intermediate powder having a general formula Li.sub.1+aCo.sub.1-x'-y'Al.sub.x'M'.sub.y'O.sub.2, M' being at least one element of the group consisting of: Ni, Mn, Nb, Ti, W, Zr, and Mg, with 0.03.ltoreq.a'.ltoreq.0.10, 0.002.ltoreq.x'.ltoreq.0.050, and 0.ltoreq.y'.ltoreq.0.02, and comprising particles having a D50 of superior or equal to 20.00 .mu.m, preferably superior or equal to 25.00 .mu.m, more preferably superior or equal to 35.00 .mu.m, and inferior or equal to 45.00 .mu.m, preferably inferior or equal to 40.00 .mu.m, and an averaged circularity of superior or equal to 0.85 and inferior or equal to 1.00, preferably inferior or equal to 0.95, [0105] d. mixing the intermediate powder with a second Co-bearing precursor and optionally, with a source of Me, to prepare a second mixture, wherein the Li to (Co+Al+M') or the Li to (Co+Al+M'+Me) molar ratio in said second mixture is superior or equal to 0.99 and inferior or equal to 1.01, [0106] e. sintering said second mixture at a temperature of superior or equal to 800.degree. C. and inferior or equal to 1100.degree. C. in an oxygen containing atmosphere, such as air, so as to obtain a second sintered agglomerated powder, and [0107] f. milling and screening said first sintered agglomerated powder so as to obtain the cathode active material powder according to the invention, the cathode active material powder having a specific floating capacity of at most 80 mAh/g obtained by the steps a. to f.
[0108] 2. The lithium cobalt-based oxide cathode active material powder according to clause 1, having a press density superior or equal to 3.95 g/cm.sup.3 and inferior or equal to 4.40 g/cm.sup.3.
[0109] 3. The lithium cobalt-based oxide cathode active material powder according to clause 1 or 2, having a volumetric capacity of at least 570 mAh/cm.sup.3, preferably of at most 700 mAh/cm.sup.3.
[0110] 4. The lithium cobalt-based oxide cathode active material powder according to according to any of the preceding clauses, wherein said particles have a R-3m crystal structure.
[0111] 5. The lithium cobalt-based oxide cathode active material powder according to any of the preceding clauses, comprising particles having an averaged circularity of superior or equal to 0.90 and inferior or equal to 1.00, preferably inferior or equal to 0.95.
[0112] 6. The lithium cobalt-based oxide cathode active material powder according to any of the preceding clauses, wherein y and z=0.
[0113] 7. A lithium-ion secondary batteries comprising the lithium cobalt-based oxide cathode active material powder according to any of the preceding clauses.
* * * * *
D00000

D00001

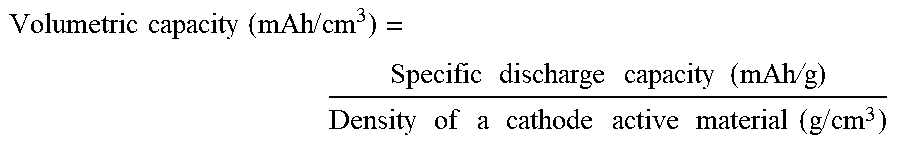

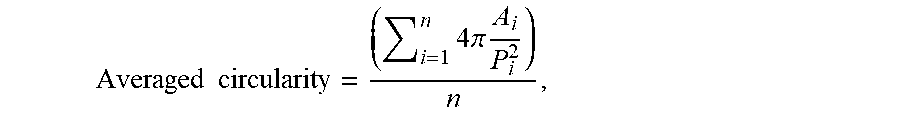
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.