Prediction Of Fuel Oil Properties By Differential Scanning Calorimetry
Golisz; Suzanne R. ; et al.
U.S. patent application number 16/716986 was filed with the patent office on 2021-01-28 for prediction of fuel oil properties by differential scanning calorimetry. The applicant listed for this patent is ExxonMobil Research and Engineering Company. Invention is credited to Erin R. Fruchey, Suzanne R. Golisz, Kenneth C. H. Kar, Sheryl B. Rubin-Pitel, Madhavi Vadlamudi.
| Application Number | 20210025841 16/716986 |
| Document ID | / |
| Family ID | 1000004722740 |
| Filed Date | 2021-01-28 |




| United States Patent Application | 20210025841 |
| Kind Code | A1 |
| Golisz; Suzanne R. ; et al. | January 28, 2021 |
PREDICTION OF FUEL OIL PROPERTIES BY DIFFERENTIAL SCANNING CALORIMETRY
Abstract
Systems and methods are provided for using differential scanning calorimetry (DSC) to predict properties of fuel compositions, such as marine fuel oils. It has been discovered that various features of the data plots generated by DSC can be correlated with properties of interest for marine fuel oil compositions. The fuel composition properties that can be predicted based on DSC include, but are not limited to, density; micro carbon residue; pour point; and estimated cetane number (ECN). This can include prediction of ECN for resid-containing fuel compositions. Using DSC to predict ECN can reduce or minimize the number of resid-containing fuel oil samples that require testing using the limited availability equipment required for the IP 541 method.
| Inventors: | Golisz; Suzanne R.; (Annandale, NJ) ; Fruchey; Erin R.; (Philadelphia, PA) ; Kar; Kenneth C. H.; (Philadelphia, PA) ; Rubin-Pitel; Sheryl B.; (Newtown, PA) ; Vadlamudi; Madhavi; (Clinton, NJ) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004722740 | ||||||||||
| Appl. No.: | 16/716986 | ||||||||||
| Filed: | December 17, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62877129 | Jul 22, 2019 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | G01N 25/4833 20130101; G01N 33/2829 20130101; G01N 33/2811 20130101 |
| International Class: | G01N 25/48 20060101 G01N025/48; G01N 33/28 20060101 G01N033/28 |
Claims
1. A method for characterizing a fuel composition, the method comprising: performing differential scanning calorimetry on a fuel composition to generate a heating curve; determining a temperature corresponding to an end point temperature of a wax melting phase transition based on the heating curve; and predicting an estimated cetane number (ECN) based on the determined temperature.
2. The method of claim 1, wherein the fuel composition comprises a resid-containing fuel composition.
3. The method of claim 2, wherein the resid-containing fuel composition comprises a marine fuel oil that satisfies the standards under at least one of ISO 8217 Table 1 and ISO 8217 Table 2.
4. The method of claim 1, wherein the fuel composition comprises 1.0 wt % or more of 370.degree. C.+ components.
5. The method of claim 1, wherein the fuel composition is not clear and bright.
6. The method of claim 1, wherein the predicted ECN is 20 or more.
7. The method of claim 1, further comprising: determining that the predicted ECN is below a threshold value; and measuring a cetane value according to the method in IP 541.
8. The method of claim 1, wherein the fuel composition comprises a calculated carbon aromaticity index of 870 or less and a density at 15.degree. C. of 900 kg/m.sup.3 to 1,010 kg/m.sup.3.
9. The method of claim 1, wherein the fuel composition comprises a kinematic viscosity at 50.degree. C. of 100 centistokes to 700 centistokes.
10. The method of claim 1, wherein the fuel composition comprises a sulfur content of 0.50% or less by weight of the fuel composition.
11. The method of claim 1, wherein the fuel composition comprises a calculated aromaticity index is about 800 to about 870, a density at 15.degree. C. of 950 kg/km.sup.3 to 1,000 kg/m.sup.3, and a kinematic viscosity at 50.degree. C. of 100 cSt to 380 cSt.
12. A method for characterizing a fuel composition, the method comprising: performing differential scanning calorimetry on a fuel composition to generate at least one of a heating curve and a cooling curve; determining one or more parameters related to a wax content, a wax phase transition, or a combination thereof based on the at least one of a heating curve and a cooling curve; and predicting one or more of a density, a pour point, an estimated cetane number (ECN), and a micro carbon residue based on the determined parameters.
13. The method of claim 12, wherein the determined one or more parameters comprise an amount of wax that melts above a characterization temperature.
14. The method of claim 13, wherein the characterization temperature is at least one of 0.degree. C., 10.degree. C., and 20.degree. C.
15. The method of claim 12, wherein the determined one or more parameters comprise a temperature that corresponds to at least one of a starting temperature, an end temperature, and a peak temperature for a wax phase transition.
16. The method of claim 12, wherein the fuel composition comprises a resid-containing fuel composition.
17. The method of claim 16, wherein the resid-containing fuel composition comprises a marine fuel oil that satisfies the standards under at least one of ISO 8217 Table 1 and ISO 8217 Table 2.
18. The method of claim 12, wherein the fuel composition comprises 1.0 wt % or more of 370.degree. C.+ components, or wherein the fuel composition is not clear and bright, or a combination thereof.
19. The method of claim 12, wherein the predicted ECN is 20 or more.
20. The method of claim 12, further comprising: determining that the predicted ECN is below a threshold value; and measuring a cetane value according to the method in IP 541.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to U.S. Provisional Application Ser. No. 62/877,129 filed Jul. 22, 2019, which is herein incorporated by reference in its entirety.
STATEMENT OF RELATED APPLICATIONS
[0002] This application is related to the U.S. Provisional Patent Application No. 62/877,129 entitled "Low Sulfur Fuel With Adequate Combustion Quality," having common inventors and assignee and filed on an even date herewith, the disclosure of which is incorporated by reference herein in its entirety.
FIELD
[0003] This application relates to prediction of properties of fuel compositions using differential scanning calorimetry, such as prediction of estimated cetane number.
BACKGROUND
[0004] Internal combustion engines are a type of engine where combustion of a fuel composition occurs in a combustion chamber to transfer chemical energy into mechanical energy. One type of internal combustion engine is a compression ignition engine in which ignition of the fuel composition is caused by elevated temperature of the air by mechanical compression. Fuel compositions used in compression ignition engines can include, but are not limited to, fuel oils, such as diesel fuels, distillate fuel oils, and residual fuel oils.
[0005] The combustion quality of a fuel for use in a compression ignition engine can be characterized based on a cetane rating or cetane number. For fuels that include primarily distillate boiling range components, one or more readily available methods are available for characterizing cetane, either as cetane rating or cetane number. Unfortunately, these readily available methods cannot be used for fuels that contain more than a de minimis amount of resid boiling range components (e.g., more than 0.1 wt % of 370.degree. C.+ components).
[0006] Ignition and combustion properties of residual fuel oils can be determined by the method specified in IP 541: Determination of Ignition and Combustion Characteristics of Residual Fuels. In this test method, multiple injections of the residual fuel oil are made into a heated and pressurized combustion chamber of constant volume. The combustion chamber pressure is monitored versus time to determine the various characteristics, including the main combustion delay (MCD). The MCD can be used to calculate an estimated cetane number (ECN). The ECN is generally accepted as an indicator of combustion quality for residual fuel oils. In order to determine, whether a fuel composition can burn in an engine, a minimum cetane number is required. For residual fuel oils, however it can be difficult to measure the MCD, because access to instruments for testing can be limited. Since MCD is used in calculation of ECN, this makes measurement of ECN difficult. Due to this limited availability, it would be desirable to have a method for predicting ECN, so that the number of samples requiring characterization according to IP 541 can be reduced or minimized.
[0007] U.S. Pat. No. 10,106,751 describes methods for using differential scanning calorimetry (DSC) to determine the viscosity index potential of a feedstock for production of lubricant base stocks.
SUMMARY
[0008] In various aspects, a method for characterizing a fuel composition, such as a marine fuel composition, is provided. The method can include performing differential scanning calorimetry on a fuel composition to generate at least one of a heating curve and a cooling curve. The method can further include determining one or more parameters related to a wax content, a wax phase transition, or a combination thereof based on the at least one of a heating curve and a cooling curve. Additionally, the method can include predicting one or more of a density, a pour point, an estimated cetane number (ECN), and a micro carbon residue based on the determined parameters.
[0009] In some aspects, the method can be used to predict an ECN, such as an ECN for a resid-containing fuel composition. In such aspects, the method can include performing differential scanning calorimetry on a fuel composition to generate a heating curve. The method can further include determining a temperature corresponding to an end point temperature of a wax melting phase transition based on the heating curve. Additionally, the method can include predicting an (ECN) based on the determined temperature.
BRIEF DESCRIPTION OF THE DRAWINGS
[0010] FIG. 1 shows an example of a correlation between a temperature at the end of a wax melting phase transition in a DSC curve and the estimated cetane number for a fuel oil.
[0011] FIG. 2 shows an example of a correlation between a temperature at the onset of a wax crystallization phase transition in a DSC curve and the density for a fuel oil.
[0012] FIG. 3 shows an example of a correlation between a temperature at a peak in a wax crystallization phase transition in a DSC curve and the estimated cetane number for a fuel oil.
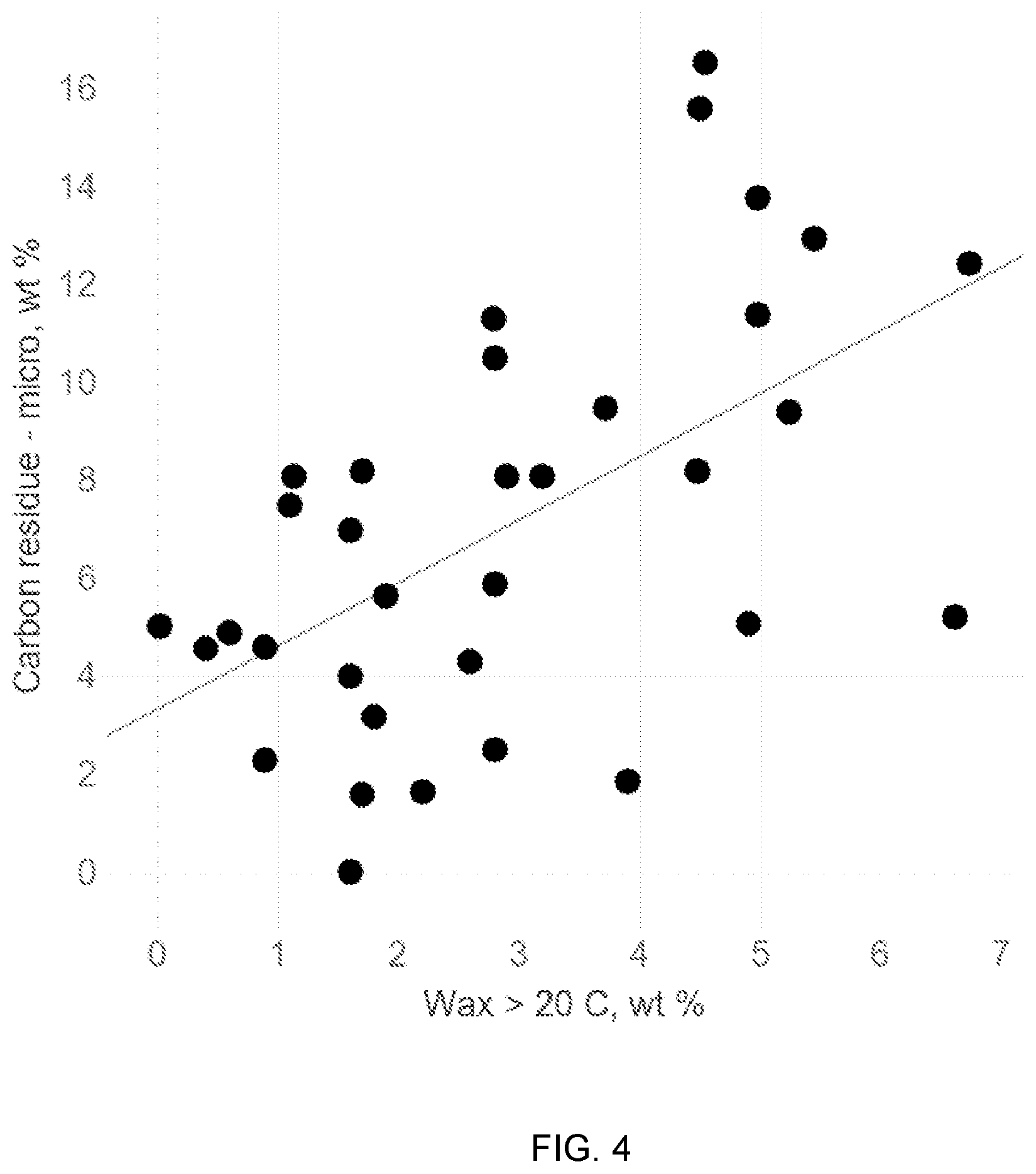
[0013] FIG. 4 shows an example of a correlation between an amount of wax that melts above 20.degree. C. based on a DSC curve and the micro carbon residue content for a fuel oil.
[0014] FIG. 5 shows an example of a correlation between a temperature at a peak in a wax crystallization phase transition in a DSC curve and the pour point for a fuel oil.
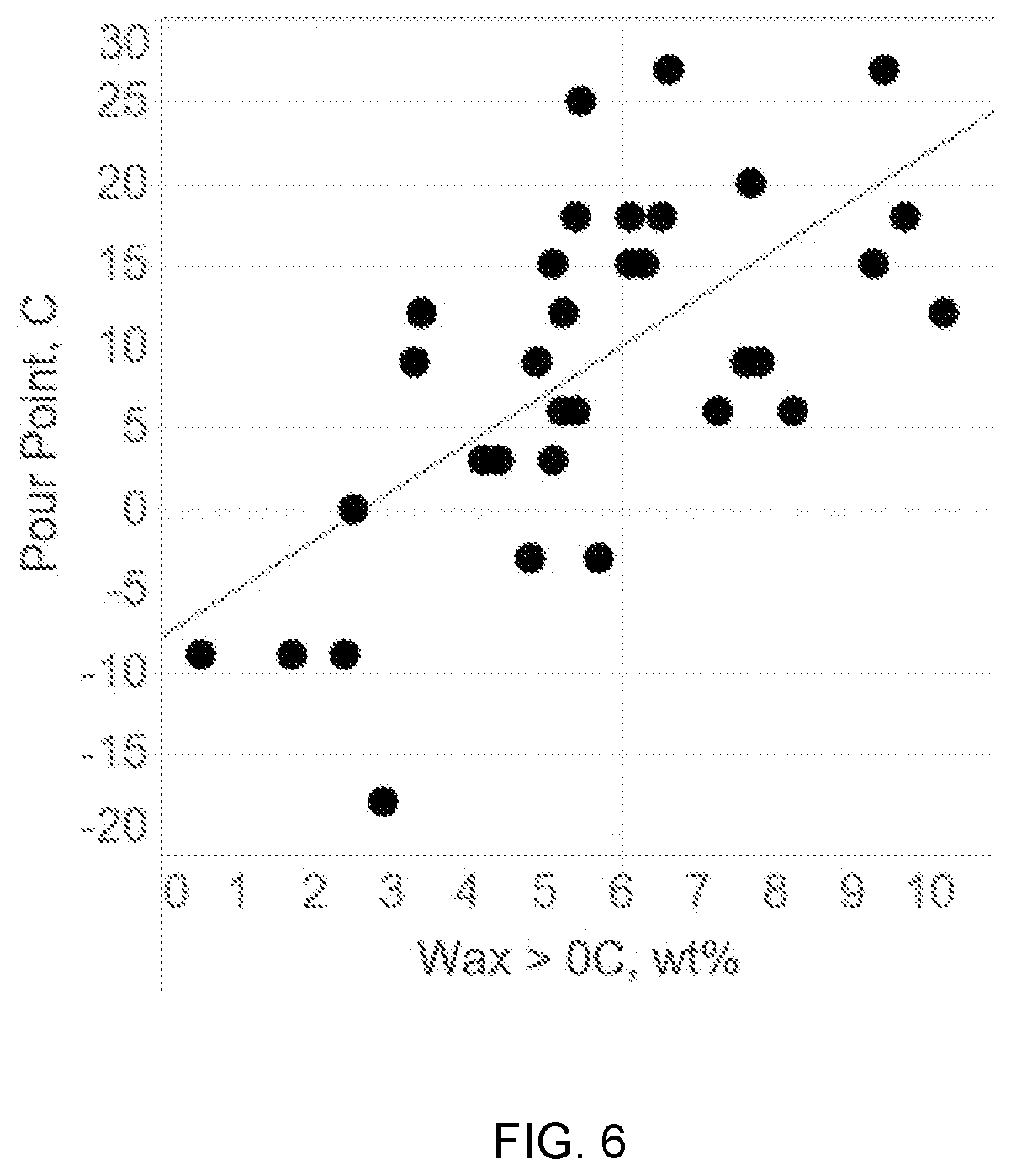
[0015] FIG. 6 shows an example of a correlation between an amount of wax that melts above 0.degree. C. based on a DSC curve and the pour point for a fuel oil.
DESCRIPTION
[0016] In various aspects, systems and methods are provided for using differential scanning calorimetry (DSC) to predict properties of fuel compositions, such as marine fuel oils. It has been discovered that various features of the data plots generated by DSC can be correlated with properties of interest for marine fuel oil compositions. The fuel oil properties that can be predicted based on DSC include, but are not limited to, density; micro carbon residue; pour point; and estimated cetane number (ECN). In some aspects, DSC can be used to predict ECN for fuel compositions including 0.1 wt % or more of 370.degree. C.+ components. Fuel compositions (such as fuel oils) containing 0.1 wt % or more of 370.degree. C.+ components are defined herein as resid-containing fuel compositions. In other aspects, DSC can be used to predict ECN for fuel oils including 1.0 wt % or more of 370.degree. C.+ components. Additionally or alternately, a resid-containing fuel composition can be identified based on the appearance of the fuel composition not being bright and clear. Using DSC to predict ECN can reduce or minimize the number of resid-containing fuel oil samples that require testing using the limited availability equipment required for the IP 541 method.
[0017] Many distillate fuels, such as automotive diesel, have limited boiling ranges that allow a variety of methods to be used to determine the combustion quality of the fuel. For example, the cetane number of a diesel fuel can be determined according to the method in ASTM D613. However, such methods of determining combustion quality for distillate fuels are typically performed in test apparatus that will foul if resid-boiling range components are introduced into the apparatus. Unlike automotive diesel fuels, many types of marine fuel oils that include distillate boiling range components can also include components corresponding to atmospheric resid, such as components with a boiling point of 343.degree. C. or more, or 370.degree. C. or more. Due to such resid-boiling range components, many conventional methods for characterization of combustion quality are not suitable for marine fuel oils. As a result, IP 541 is the only method available for characterizing combustion quality for marine fuel oils. Unfortunately, the availability of the IP 541 test is limited to a few systems world-wide that are capable of performing the required method.
[0018] An example of the benefit of being able to predict estimated cetane number (ECN) is the ability to use DSC to assist with determining if a fuel composition (such as a marine fuel oil) has "adequate combustion quality". Adequate combustion quality can correspond to an ECN of 7 or greater. By using DSC, a prediction of ECN can be determined. A sufficiently high predicted ECN can provide a high degree of certainty that the measured ECN would be 7 or greater, thus avoiding the need to perform a test according to the method in IP 541.
[0019] In some aspects, the prediction of ECN for a marine fuel oil sample based on DSC can be achieved based on a correlation of ECN with the end point temperature for the wax melting phase transition (Tm end) from the DSC heating curve. Without being bound by any particular theory, the correlation between ECN and Tm end suggests that as the melting temperature of the wax increases, it becomes more difficult to burn the wax. The highest cetane molecules are long-chain paraffinic molecules. Although the most familiar forms of wax are also long-chain paraffinic molecules, it is believed that the higher melting waxes do not simply follow the trend of just increasing carbon number of long-chain paraffinic molecules. Instead, it is believed that the higher melting waxes that are typically derived from the residual component in the fuel have a molecular structure that is less favorable for combustion. As a result, as the melting temperature of wax increases, it is believed that the higher melting wax has a greater tendency to include components with lower cetane values, such as branched paraffins, naphthenes, and/or aromatics.
[0020] In addition to predicting ECN, other properties of a marine fuel oil can be predicted based on DSC characterization of a sample. Such additional properties can include micro carbon residue, pour point, and density. Density was found to correlate with both the temperature for onset of wax crystallization (Tc onset) and the temperature corresponding to the peak for crystallization (Tc peak) from the DSC cooling curve. Micro carbon residue was found to correlate with the percentage of wax that melts above 20.degree. C. The pour point was found to correlates with the percentage of wax at various temperatures, as well as Tc peak.
[0021] Without being bound by any particular theory, it is believed that the correlation between the density and Tc onset or Tc peak is due in part to the fact that as the crystallization temperature(s) increase, the molecular weight of the molecules that contribute to the wax is likely increasing. As the molecular weight increases, the ratio of carbon to hydrogen is increasing, thus increasing the density.
[0022] Micro carbon residue is a measure of the coke-forming tendency of a fuel. As the amount of wax that melts above 20.degree. C. increases, it has been observed that the carbon residue also increases. Without being bound by any particular theory, the higher the melting temperature of the wax, the more likely it is to form coke due in part to the increased amount of carbon-carbon bonds. Therefore, if a fuel has an increased amount of higher-melting-temperature wax, it should also have more micro carbon residue.
[0023] Pour point is a measure of the temperature at which the fuel can no longer be poured. It has been found that pour point is correlated with both Tc peak and the amount of wax that crystallizes at various characterization temperatures. Generally, crystallization of wax contributes to preventing fuel from flowing. Additionally, higher amounts of wax will cause a fuel to stop flowing at higher temperatures than fuels with lower wax content. As a result, correlation of pour point with the amount of wax in fuel that is crystalized at characterization temperatures above 0.degree. C., 10.degree. C., or 20.degree. C. is expected.
[0024] In various aspects, the equations from the above correlations, including the correlation for ECN, may be used to predict the fuel oil properties from the respective measured DSC parameters.
[0025] In this discussion, a temperature related to a wax phase transition is defined as a temperature that is characteristic of melting and/or crystallization phase transition for wax in a heating curve or cooling curve for a sample generated by differential scanning calorimetry. The temperature can correspond to a temperature at the start of a phase transition (i.e., a start of melting or a start of crystallization), a temperature at the end of a phase transition, or a temperature corresponding to a peak in the heating/cooling curve during a phase transition. A wax melting phase transition is defined as a melting phase transition in a heating curve for a sample generated by differential scanning calorimetry.
Marine Fuel Oil Properties
[0026] Differential scanning calorimetry (DSC) can be used to characterize any convenient type of fuel composition that contains wax-like components. In some aspects, additional benefits can be achieved by using DSC to characterize a resid-containing fuel oil. A fuel composition can also have a flash point of 60.degree. C. or more. As described above, in aspects where the fuel composition is a resid-containing fuel oil, the resid-containing fuel oil can include 0.1 wt % or more of 370.degree. C.+ components. More generally, a resid-containing fuel oil can include 0.1 wt % to 90 wt % of 370.degree. C.+ components, or 0.1 wt % to 80 wt %, or 0.1 wt % to 50 wt %, or 0.1 wt % to 20 wt %, or 0.1 wt % to 10 wt %, or 1.0 wt % to 90 wt %, or 1.0 wt % to 80 wt %, or 1.0 wt % to 50 wt %, or 10 wt % to 80 wt %, or 10 wt % to 50 wt %. ASTM D2887 is a suitable method for determining the distillation ranges of a fuel oil sample. If ASTM D2887 is not suitable for a particular sample, ASTM D7169 can be used instead.
[0027] In some aspects, a suitable virgin fraction of a whole or partial crude oil can be used as a fuel oil. In other aspects, a fuel composition can be formed from any convenient number of source components, such as various types of distillate and/or resid boiling range fractions. Such components can include fractions corresponding to a combination of a distillate and resid boiling range components. Examples of distillate components can include, but are not limited to, hydrotreated straight run distillates, hydrocracker distillates, hydrotreated gas oils, heavy vacuum gas oils, light vacuum gas oils, heavy atmospheric gas oils, light cycle oils, light coker gas oils, heavy cycle oils, heavy coker gas oils, and steam cracked gas oils. Examples of suitable residual components may include, but are not limited to, vacuum residuals from fractionating (total/partial) crude oils, atmospheric residuals from fractionating (total/partial) crude oils, visbreaker residuals, FCC bottoms, hydrotreated residual, and deasphalted residuals A distillate fraction is defined herein as a fraction having a T10 distillation point of 150.degree. C. or more and a T95 distillation point of 343.degree. C. or less. A resid fraction is defined herein as a fraction having a T10 distillation point of 343.degree. C. or more. It is noted that according to these definitions, a resid-containing fuel oil (including 0.1 wt % or more of 370.degree. C.+ components) could potentially be formed using only distillate fractions, if the distillate fractions contained sufficient 370.degree. C.+ components so that the resulting fuel oil contained a sufficient amount of 370.degree. C.+ components.
[0028] In addition to and/or as an alternative to specifying a boiling range for a fuel oil, one or more other properties of a fuel oil can be specified. For example, a fuel oil can have a sulfur content of 10 wppm to 100,000 wppm, or 1000 wppm to 100,000 wppm, or 10 wppm to 20,000 wppm, or 100 wppm to 20,000 wppm, or 1000 wppm to 20,000 wppm, or 10 wppm to 10,000 wppm, or 100 wppm to 10,000 wppm. In some aspects, the fuel oil can correspond to a fuel oil containing a reduced amount of sulfur. In such aspects, the fuel oil can have a sulfur content of 10 wppm to 4000 wppm, or 10 wppm to 1000 wppm, or 100 wppm to 4000 wppm, or 100 wppm or 1000 wppm.
[0029] In some aspects, a fuel composition characterized by DSC can correspond to a fuel composition that has "adequate combustion quality". As used herein, a fuel composition is defined as having "adequate combustion quality" where the fuel composition has an estimated cetane number (ECN) of about 7 or greater. The technique for determining ECN is described in IP 541/06: Determination of Ignition and Combustion Characteristics of Residual Fuels. In some aspects, a predicted ECN determined by performing DSC can be used to identify fuel compositions that are predicted to have an ECN greater than 7, as compared with fuel compositions that require testing according to IP 541 to verify the ECN.
[0030] Other properties of a marine fuel composition that can be characterized include, but are not limited to, flash point (according to ISO 2719 A), pour point (ISO 3016), kinematic viscosity (ISO 3104), calculated carbon aromaticity index (CCAI), density (ISO 3675), and boiling range (D2287, or D7169 if D2287 is not appropriate for a sample). For example, the flash point of a marine fuel oil can be 60.degree. C. or more, or 80.degree. C. or more, or 100.degree. C. or more, or 120.degree. C. or more, such as up to 200.degree. C. or possibly still higher. Additionally or alternately, the pour point can be 20.degree. C. or less, or 10.degree. C. or less, or 5.degree. C. or less, or 0.degree. C. or less, such as down to -20.degree. C. or possibly still lower. Additionally or alternately, the kinematic viscosity at 50.degree. C. can be 5 cSt to 1000 cSt, or 5 cSt to 300 cSt, or 5 cSt to 150 cSt, or 15 cSt to 1000 cSt, or 15 cSt to 300 cSt, or 15 cSt to 150 cSt, or 25 cSt to 1000 cSt, or 25 cSt to 300 cSt, or 25 cSt to 150 cSt. For example, the kinematic viscosity at 50.degree. C. can be at least 5 cSt, or at least 15 cSt. It is noted that fuel oils with a kinematic viscosity at 50.degree. C. of 15 cSt or higher can be beneficial, as such fuel oils typically do not require any cooling prior to use in order to be compatible with a marine engine. Additionally or alternately, the micro carbon residue of the marine fuel oil can be 5.0 wt % or less, or 4.0 wt % or less, such as down to 0.5 wt % or possibly still lower, as determined according to ISO 10370. Further additionally or alternately, the fuel composition can have a CCAI value of 870 or less, or 850 or less. For example, the CCAI value can be 750 to 870, or 800 to 870. Still further additionally or alternately, the fuel composition can have a density at 15.degree. C. of 860 kg/m.sup.3 to 1010 kg/m.sup.3.
[0031] In addition to the above, in some aspects a marine fuel composition can correspond to a resid fuel composition that meets the requirements of ISO 8217, Fuel Standard Sixth Edition 2017, Table 2. In other aspects, a marine fuel composition can correspond to a distillate marine fuel that meets the requirements of ISO 8217, Fuel Standard Sixth Edition 2017, Table 1.
Differential Scanning Calorimetry
[0032] In various aspects, differential scanning calorimetry (DSC) can be used to generate DSC heating and cooling curves. While the heating and cooling curves are directly related to the behavior of wax within a fuel oil sample, it has been discovered that other properties of a fuel oil, including estimated cetane number, can also be predicted.
[0033] The DSC cooling and heating curves for a feedstock correspond to heat flow as a function of temperature. The DSC curves are determined by first heating the feedstock sample to a temperature sufficient to melt all the residual wax contained in the feedstock. The measurement is typically preferably started at 80.degree. C. but can vary based on the feed, such as starting at a temperature of 100.degree. C. or 120.degree. C. The feedstock sample is then cooled at a cooling rate of 0.5.degree. C. to 20.degree. C. per minute and preferably 1.degree. C. to 10.degree. C. per minute. The feedstock is cooled to a temperature sufficient to completely solidify the feedstock sample. For most feedstock samples, this will be between -10.degree. C. to -80.degree. C.
[0034] The DSC heating curve is then created by heating at a rate of approximately 5.degree. C. to 20.degree. C. per minute, such as 10.degree. C. per minute. Preferably, the cooling and heating rates should be kept consistent to keep the correlation accurate. For the examples of DSC heating and cooling curves in this application, the curves were obtained with a commercially available DSC unit but any equivalent machine could be used.
[0035] Both the heating and cooling curves were inspected to determine the following temperatures; onset of crystallization (Tc onset) from the cooling curve, the peak of crystallization (Tc peak) from the cooling curve, and the end of melt (Tm end) from the heating curve. These values can be readily identified in the respective cooling and heating curves.
[0036] The amount wax in the samples was also determined based on integration of the melting peak in the heating curve. A first-principles translation of the heating curve to the physical amount of wax corresponding to these phase changes would require detailed knowledge of the sample composition and the corresponding heats of fusion for each molecular species. Since such detailed information may be unavailable, an empirical correlation relating the wax content of representative hydrocarbons to DSC heat input as a function of temperature can be determined. This correlation can then be used to calculate the wax distribution versus temperature of test samples from their experimentally measured heating curves. A person skilled in the art with the benefit of this description could create a new fundamentally-based model for this application. The operative equation is:
W=.DELTA.H/A(T) (1)
[0037] In the above equation, W is the wax content of a sample. .DELTA.H represents the amount of heat absorbed by the droplet of sample in the DSC when a melting/freezing phase transition occurs, as indicated by the presence of a peak in the heating (or cooling) trace. A(T) is a scaling factor that can optionally be dependent on the temperature at which the freezing transition occurs. Without being bound by any particular theory, it is believed that the above relationship is suitable for determining a wax content base in part on the nature of the melting/freezing transition during heating and/or cooling. A DSC performs measurements on a droplet of a wax-containing sample. It is believed that the heat of fusion for the wax within the droplet is the dominant contribution to the heat of fusion for the entire droplet. As a result, the heat of fusion for the entire droplet can be related by a scaling factor of some type to the amount of wax in the droplet.
[0038] In Equation (1), the scaling factor A(T) is shown as a function of temperature. Without being bound by any particular theory, it is believed that a scaling factor having the form A(T)=a.sub.1+a.sub.2T is one suitable option for a scaling factor. Other functional forms may also be suitable.
[0039] The above type of correlation was used to determine the weight percentage of wax in a sample. The weight percent of wax at different temperatures was also used as a parameter in this invention. Three different temperature cut points were used. The percentage of wax that melted above a characterization temperature of 0.degree. C., 10.degree., and 20.degree. C. was used as a parameter.
Examples
[0040] Over 40 samples of low sulfur marine fuel were evaluated by DSC. The resulting heating and cooling curves were used to determine the parameters of Tc onset, Tc max, Tm end, wax content, and percentage of wax that melted above 0.degree. C., 10.degree. C., and 20.degree. C. These parameters derived from the heating and cooling curves were then analyzed relative to measured property values (or property values calculated from measured values) for the 40 marine fuel samples to determine whether a predictive correlation could be developed between one or more of the parameters and a desired property.
[0041] The measured properties considered for correlation were density by ISO 3675, kinematic viscosity at 50.degree. C. (KV50) by ISO 3104, pour point by ISO 3016, carbon residue (micro method) by ISO 10370, and ECN by IP 541. The fuel oil properties that were calculated from measured properties were the Bureau of Mines Correlation Index (BMCI) and the calculated carbon aromaticity index (CCAI). These properties were plotted against the different parameters from the DSC to show correlations. The threshold for a correlation was defined as a p-value of less than 0.001.
[0042] Based on the p-value threshold, no correlations were found between the DSC measured parameters and the property values for KV50, BMCI, or CCAI. Correlations were identified, however, for density, pour point, micro carbon residue, and ECN.
[0043] FIG. 1 shows the measured ECN for the various fuel oil samples relative to the value of Tm end in the corresponding DSC heating curve. As shown in FIG. 1, a reasonable linear correlation was found between the measured ECN value and Tm end (p-value 0.0002003). Based on the difficulty of performing the IP 541 method and/or limited availability of the equipment for performing the IP 541 method, the correlation shown in FIG. 1 can potentially be used in several ways. One option can be to fit an equation to the data points to develop a predictive equation over at least a portion of the range of ECN values. FIG. 1 shows an example of such a fitted equation. The equation shown in FIG. 1 corresponds to
ECN=-0.751034*<Tm end,.degree. C.>+89.5941 (2)
[0044] In some aspects, an ECN value predicted from a relationship such as Equation 2 could be used to in place of a measured ECN value.
[0045] Alternatively, the correlation between Tm end and ECN can be used as a screening tool to reduce or minimize the number of ECN measurements that are performed according to IP 541. When used as a screening tool, an equation such as Equation 2 can be used to determine a predicted ECN value. When the predicted ECN value is above a threshold quantity, it can be determined that the corresponding measured ECN value would be above a target level. For example, for some fuel oils, it can be desirable for the fuel oil to have an ECN value greater than 7.0, so that the fuel oil has adequate combustion quality. Based on FIG. 1, all fuel oils with a predicted ECN value of 20 or more have a corresponding measured ECN value of 7.0 or more. In this type of example, if a fuel oil has a predicted ECN value of 20 or more, it would not be necessary to perform an ECN measurement according to IP 541. For a predicted ECN value of less than 20, an ECN measurement would be performed according to IP 541 to verify that the fuel oil has an ECN of greater than 7.0.
[0046] In addition to the correlation for ECN, several other properties were found to correlate with one or more DSC parameters. Density at 15.degree. C. was found to correlate with both Tc onset and Tc peak. Micro carbon residue was found to correlate with the percentage of wax that melts above 20.degree. C. Pour point was found to correlate with the percentage of wax at all three temperatures, as well as with Tc peak. The equations from these correlations may be used to predict the fuel oil properties from the measured DSC parameter.
[0047] FIG. 2 shows the measured density for the fuel oil samples relative to the corresponding DSC values for Tc onset for the samples. FIG. 3 similarly shows the measured density for the fuel oil samples relative to the corresponding DSC Tc peak values. As shown in FIG. 2 and FIG. 3, a reasonable linear correlation was found relative to density for both Tc onset (p-value 0.0007886) and Tc peak (p-value 0.0001630).
[0048] FIG. 4 shows the measured micro carbon residue content for the fuel oil samples relative to the corresponding DSC value for the amount of wax that melts above 20.degree. C. As shown in FIG. 4, a reasonable linear correlation was found relative to micro carbon residue content for the amount of wax that melts above 20.degree. C. (p-value 0.0006526).
[0049] FIG. 5 shows the measured pour point for the fuel oil samples relative to the corresponding DSC value for Tc peak. FIG. 6 shows the measured pour point for the fuel oil samples relative to the amount of wax that melts above a characterization temperature of 0.degree. C. It is noted that a substantially similar plot could be made for the measured pour point relative to the amount of wax that melts above a characterization temperature of 10.degree. C. or 20.degree. C. As shown in FIG. 5 and FIG. 6, a reasonable linear correlation was found relative to pour point for both Tc peak (p-value 0.0001682) and the amount of wax that melts above 0.degree. C., 10.degree. C., or 20.degree. C. (p-values<0.0001).
[0050] While the disclosure has been described with respect to a number of embodiments and examples, those skilled in the art, having benefit of this disclosure, will appreciate that other embodiments can be devised which do not depart from the scope and spirit of the disclosure as disclosed herein. Although individual embodiments are discussed, the present disclosure covers all combinations of all those embodiments.
[0051] While compositions, methods, and processes are described herein in terms of "comprising," "containing," "having," or "including" various components or steps, the compositions and methods can also "consist essentially of" or "consist of" the various components and steps. The phrases, unless otherwise specified, "consists essentially of" and "consisting essentially of" do not exclude the presence of other steps, elements, or materials, whether or not, specifically mentioned in this specification, so long as such steps, elements, or materials, do not affect the basic and novel characteristics of the disclosure, additionally, they do not exclude impurities and variances normally associated with the elements and materials used.
[0052] All numerical values within the detailed description and the claims herein modified by "about" or "approximately" with respect to the indicated value are intended to take into account experimental error and variations that would be expected by a person having ordinary skill in the art.
[0053] For the sake of brevity, only certain ranges are explicitly disclosed herein. However, ranges from any lower limit may be combined with any upper limit to recite a range not explicitly recited, as well as, ranges from any lower limit may be combined with any other lower limit to recite a range not explicitly recited, in the same way, ranges from any upper limit may be combined with any other upper limit to recite a range not explicitly recited.
* * * * *
D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.