Functional Chromium Alloy Plating From Trivalent Chromium Electrolytes
IJERI; Vijaykumar ; et al.
U.S. patent application number 16/515145 was filed with the patent office on 2021-01-21 for functional chromium alloy plating from trivalent chromium electrolytes. The applicant listed for this patent is THE BOEING COMPANY. Invention is credited to Stephen P. GAYDOS, Vijaykumar IJERI, Subramanian MOHAN.
| Application Number | 20210017659 16/515145 |
| Document ID | / |
| Family ID | 1000005005284 |
| Filed Date | 2021-01-21 |
| United States Patent Application | 20210017659 |
| Kind Code | A1 |
| IJERI; Vijaykumar ; et al. | January 21, 2021 |
FUNCTIONAL CHROMIUM ALLOY PLATING FROM TRIVALENT CHROMIUM ELECTROLYTES
Abstract
The present disclosure provides electrolyte solutions for electrodeposition of chromium-iron alloys and methods of electrodepositing chromium-iron alloys. An electrolyte solution for electroplating can include a trivalent chromium salt, an oxalate compound, an iron salt, an aluminum sulfate, an alkali metal sulfate, and an alkali metal halide. An electrolyte solution can be formed by dissolving a trivalent chromium salt, an oxalate compound, an iron salt, an aluminum sulfate, an alkali metal sulfate, and an alkali metal halide in water or an aqueous solution. Electrodepositing chromium-iron alloys on a substrate can include introducing a cathode and an anode into an electrolyte solution comprising a trivalent chromium salt, an oxalate compound, an iron salt, an aluminum sulfate, an alkali metal sulfate, and an alkali metal halide. Electrodepositing can further include passing a current between the cathode and the anode through the electrolyte solution to deposit chromium and iron onto the cathode.
| Inventors: | IJERI; Vijaykumar; (Mumbai, IN) ; GAYDOS; Stephen P.; (St. Louis, MO) ; MOHAN; Subramanian; (Chennai, IN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005005284 | ||||||||||
| Appl. No.: | 16/515145 | ||||||||||
| Filed: | July 18, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C25D 3/06 20130101; C25D 17/10 20130101 |
| International Class: | C25D 3/06 20060101 C25D003/06; C25D 17/10 20060101 C25D017/10 |
Claims
1. An electrolyte solution for electroplating comprising: a trivalent chromium salt; an oxalate compound; an iron salt; an aluminum sulfate; an alkali metal sulfate; and an alkali metal halide.
2. The electrolyte solution of claim 1, wherein: the trivalent chromium salt is present in an amount ranging from about 0.3 moles per liter to about 0.9 moles per liter of the electrolyte solution; the oxalate compound is present in an amount ranging from about 0.2 moles per liter to about 1.2 moles per liter of the electrolyte solution; the iron salt is present in an amount ranging from about 0.005 moles per liter to about 0.2 moles per liter of the electrolyte solution; the aluminum sulfate is present in an amount ranging from about 0.05 moles per liter to about 0.5 moles per liter; the alkali metal sulfate is present in an amount ranging from about 0.1 moles per liter to about 2.0 moles per liter of the electrolyte solution; and the alkali metal halide is present in an amount ranging from about 0.1 moles per liter to about 0.5 moles per liter of the electrolyte solution.
3. The electrolyte solution of claim 1, wherein the iron salt is a divalent iron salt comprising one or more of iron (II) sulfate, iron (II) chloride, iron (II) acetate, and hydrates thereof.
4. The electrolyte solution of claim 1, wherein the iron salt is a trivalent iron salt comprising one or more of iron (III) sulfate, iron (III) chloride, iron (III) acetate, and hydrates thereof.
5. The electrolyte solution of claim 1, wherein the trivalent chromium salt is selected from a chromium (III) halide, a chromium (III) sulfate, or a combination thereof.
6. The electrolyte solution of claim 1, wherein the oxalate compound is selected from sodium oxalate, potassium oxalate, an acid of oxalate, or a combination thereof.
7. The electrolyte solution of claim 1, wherein the alkali metal sulfate is selected from sodium sulfate, potassium sulfate, or a combination thereof.
8. The electrolyte solution of claim 1, wherein the alkali metal halide is selected from sodium fluoride, potassium fluoride, or a combination thereof.
9. The electrolyte solution of claim 1, wherein a pH of the electrolyte solution is in a range from about 1 to about 4.
10. The electrolyte solution of claim 1, further comprising sodium lauryl sulfate, sodium lauryl ether sulfate, or a combination thereof.
11. A method of chrome plating on a substrate using an electrolyte solution, comprising: introducing a cathode and an anode into an electrolyte solution comprising a trivalent chromium salt, an oxalate compound, an iron salt, an aluminum sulfate, an alkali metal sulfate, and an alkali metal halide; and passing a current between the anode and the cathode through the electrolyte solution to deposit a chromium layer on the substrate.
12. The method of claim 11, wherein the cathode is a steel substrate, a copper substrate, a brass substrate, a nickel substrate, a copper-coated substrate, or a nickel-coated substrate.
13. The method of claim 11, wherein the anode is a platinum material, platinized titanium material, or a carbonaceous electrode material.
14. The method of claim 11, wherein the current has a current density in a range from about 10 to about 60 mA/cm.sup.2 by passing direct current between the anode and the cathode.
15. The method of claim 14, wherein the current density has a current density in a range from about 20 to about 40 mA/cm.sup.2.
16. The method of claim 14, wherein the electrolyte solution is maintained at a temperature in a range from about 20 degrees Celsius and about 60 degrees Celsius.
17. The method of claim 11, further comprising adjusting a pH of the electrolyte solution to a pH in a range from about 1.5 to about 4.
18. A substrate comprising: a chromium-iron alloy coating having a chromium content in a range of about 40 wt. % to about 90 wt. %, an iron content in a range of about 8 wt. % to about 18 wt. %, and a carbon content in a range of about 5 wt. % to about 50 wt. %.
19. The substrate of claim 18, wherein the substrate comprises one or more of steel, copper, brass, or nickel.
20. The substrate of claim 19, wherein the chromium-iron alloy coating has a thickness of from about 1 micron and about 100 microns.
Description
FIELD
[0001] The present disclosure provides electrolyte solutions for electrodeposition of chromium alloys, methods of forming electrolyte solutions, and methods of electrodepositing chromium alloys.
BACKGROUND
[0002] Chrome plating is an electroplating process that provides a chrome coating on a substrate. Hard chrome plating provides a chrome coating having a thickness typically about 10 microns or greater, thereby providing hardness and wear resistance to the coated substrate. The other type of chrome plating is decorative chrome plating, which provides a chrome coating having a thickness typically ranging from about 0.1 to about 0.5 microns. Chrome plating is often performed using baths containing chromic acid and catalysts based on fluorides, sulfates or organic acids. Chromic acid has chromium in its hexavalent form, chromium (VI), which is not environmentally preferred and expensive to dispose of.
[0003] Trivalent chromium, which has benign properties and lower waste treatment costs, is one alternative to hexavalent chromium. There has been some success implementing trivalent chromium for thin, decorative plating, however, there is still difficulty in achieving thicker or functional coatings. In addition, trivalent chromium-based baths used in decorative plating typically contain boric acid as a buffering agent.
[0004] Thus, there is a need for improved chrome plating methods and formulations of solutions used in chrome plating.
SUMMARY
[0005] The present disclosure provides electrolyte solutions for electrodeposition of chromium alloys, methods of forming electrolyte solutions, and methods of electrodepositing chromium alloys.
[0006] At least one electrolyte solution for electroplating includes a trivalent chromium salt, an oxalate compound, an iron salt, an aluminum sulfate, an alkali metal sulfate, and an alkali metal halide.
[0007] At least one electrolyte solution for electroplating includes a trivalent chromium salt in an amount ranging from about 0.3 moles per liter to about 0.9 moles per liter of the electrolyte solution. The electrolyte solution further includes an oxalate compound in an amount ranging from about 0.2 moles per liter to about 1.2 moles per liter of the electrolyte solution. The electrolyte solution further includes an iron salt in an amount ranging from about 0.005 moles per liter to about 0.2 moles per liter of the electrolyte solution. The electrolyte solution further includes an aluminum sulfate in an amount ranging from about 0.05 moles per liter to about 0.5 moles per liter. The electrolyte solution further includes an alkali metal sulfate in an amount ranging from about 0.1 moles per liter to about 2.0 moles per liter of the electrolyte solution. The electrolyte solution further includes an alkali metal halide in an amount ranging from about 0.1 moles per liter to about 0.5 moles per liter of the electrolyte solution.
[0008] At least one method of chrome plating on a substrate using an electrolyte solution is provided. The method includes dissolving in an aqueous medium a trivalent chromium salt in an amount ranging from about 0.3 moles per liter to about 0.9 moles per liter of the electrolyte solution. The method further includes dissolving an oxalate compound in an amount ranging from about 0.2 moles per liter to about 1.2 moles per liter of the electrolyte solution. The method further includes dissolving iron salt in an amount ranging from about 0.005 moles per liter to about 0.2 moles per liter of the electrolyte solution. The method further includes dissolving aluminum sulfate in an amount ranging from about 0.05 moles per liter to about 0.5 moles per liter. The method further includes dissolving an alkali metal sulfate in an amount ranging from about 0.1 moles per liter to about 2.0 moles per liter of the electrolyte solution. The method further includes dissolving an alkali metal halide in an amount ranging from about 0.1 moles per liter to about 0.5 moles per liter of the electrolyte solution. The method further comprises passing a current between a cathode and an anode through the electrolyte solution to deposit a chromium-iron alloy on a substrate.
[0009] At least one method of chrome plating on a substrate using an electrolyte solution is provided. The method includes introducing a cathode and an anode into an electrolyte solution comprising a trivalent chromium salt, an oxalate compound, an iron salt, an aluminum sulfate, an alkali metal sulfate, and an alkali metal halide. The method further includes passing a current between the anode and the cathode through the electrolyte solution to deposit a chromium-iron alloy layer on the substrate.
[0010] At least one substrate including a chromium-iron alloy coating is provided. The chromium-iron alloy coating has a chromium content in a range of about 40 wt. % to about 90 wt. %, an iron content in a range of about 8 wt. % to about 18 wt. %, and a carbon content in the range of about 5 wt. % to about 50 wt. %.
[0011] The features, functions, and advantages that have been discussed can be achieved independently in various aspects or can be combined in yet other aspects, further details of which can be seen with reference to the following description and drawings.
BRIEF DESCRIPTION OF DRAWINGS
[0012] So that the manner in which the above-recited features of the present disclosure can be understood in detail, a more particular description of the disclosure briefly summarized above can be had by reference to aspects, some of which are illustrated in the appended drawings. It is to be noted, however, that the appended drawings illustrate only typical aspects of this disclosure and are therefore not to be considered limiting of its scope, for the disclosure can admit to other equally effective aspects.
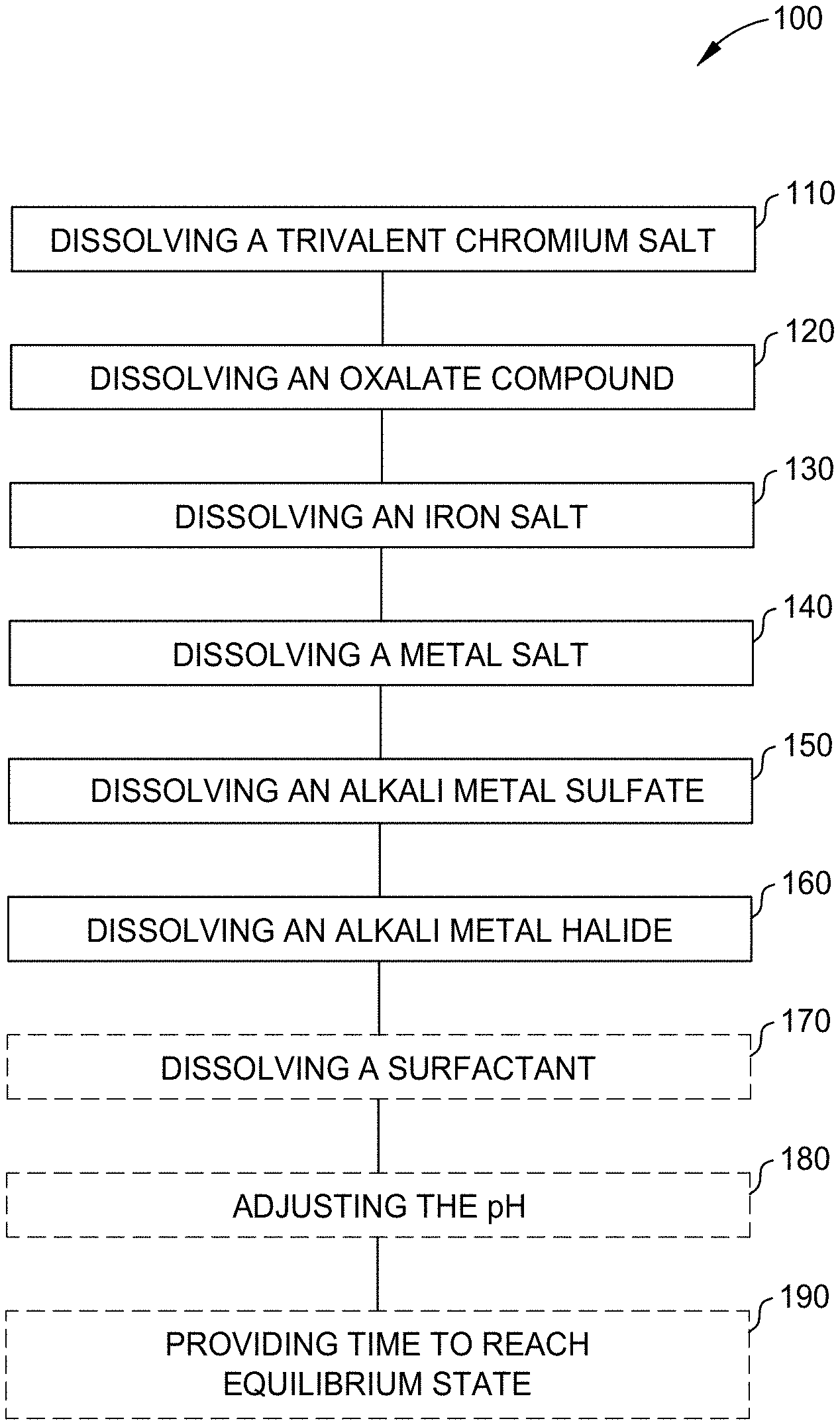
[0013] FIG. 1 is a flow diagram illustrating a method for forming an electrolyte solution according to one or more aspects of the present disclosure;
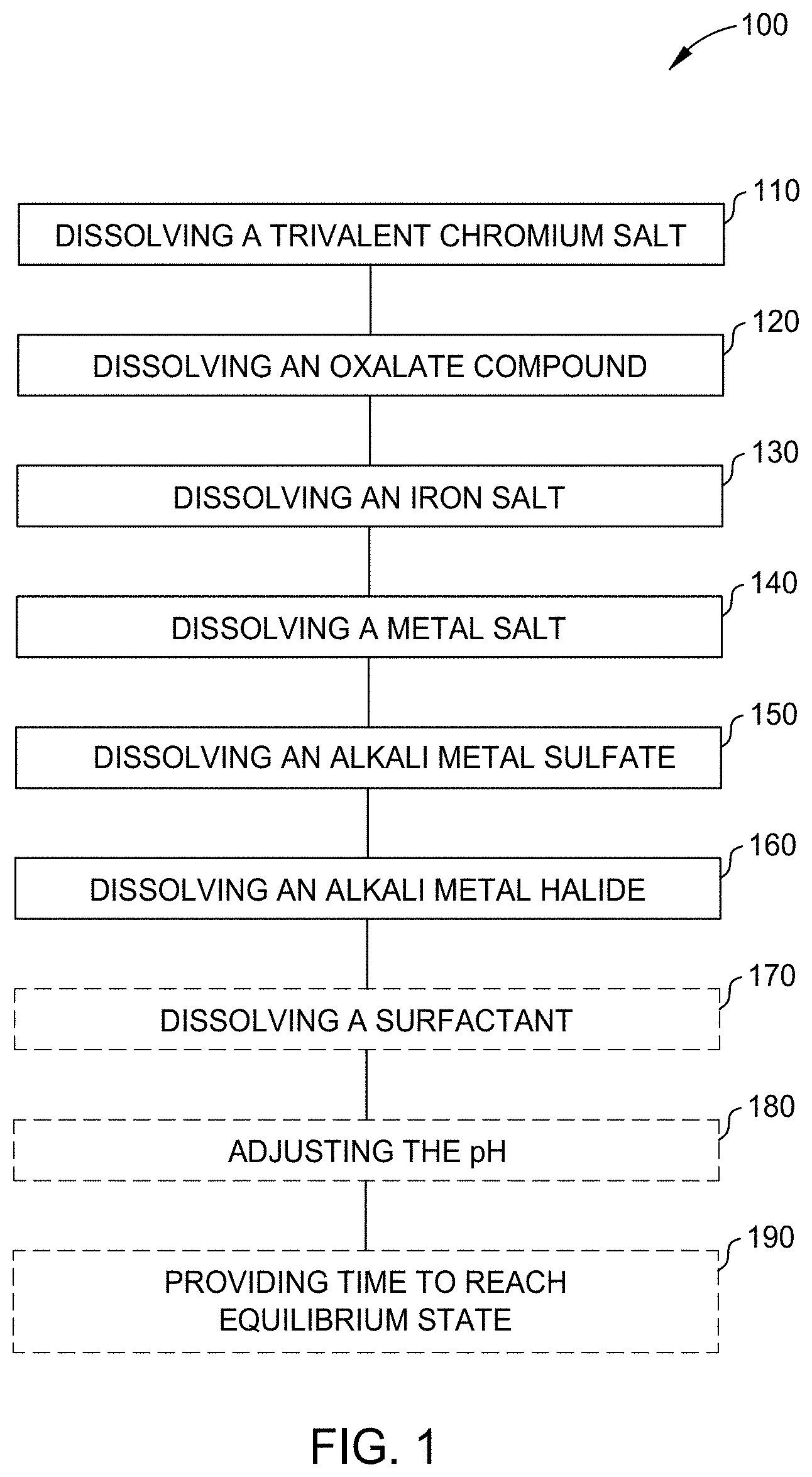
[0014] FIG. 2 is a flow diagram illustrating a method for forming a chromium alloy coating on a substrate by electrodeposition according to one or more aspects of the present disclosure;
[0015] FIG. 3 is an image of chromium-iron alloy plated substrates formed by the process of FIG. 2, each plated at a different current density;
[0016] FIG. 4 is an image of chromium-iron alloy plated substrates formed by the process of FIG. 2, each plated at a different pH;
[0017] FIG. 5 is an image of chromium-iron alloy plated substrates formed by the process of FIG. 2, each plated at a different current density;
[0018] FIG. 6 is an image of chromium-iron alloy plated substrates formed by the process of FIG. 2, each plated at a different pH;
[0019] FIG. 7 is an image of chromium-iron alloy plated substrates formed by the process of FIG. 2, each plated at a different current density;
[0020] FIG. 8 is an image of chromium-iron alloy plated substrates formed by the process of FIG. 2, each plated at a different pH; and
[0021] FIG. 9 is an image of chromium-iron alloy plated substrates formed by the process of FIG. 2, each plated at a different temperature.
[0022] To facilitate understanding, identical reference numerals have been used, wherever possible, to designate identical elements that are common to the figures. Additionally, elements of one aspect can be advantageously adapted for utilization in other aspects described herein.
DETAILED DESCRIPTION
[0023] In accordance with aspects of the present disclosure, various methods and formulations are provided for chrome plating a substrate using direct current and a trivalent chromium solution that does not include boric acid, while still resulting in a chromium layer (e.g., a chromium coating) formed on the substrate that can be structurally robust and reliable, yet cost-effective. Thus, the methods and formulations described herein can advantageously be used for hard chrome plating to form hard chromium layers (e.g., a robust, functional chromium layer of greater than 10 microns). However, the present disclosure is not limited to hard chrome plating and the methods and formulations described herein can also be advantageously used to effectively and efficiently perform decorative chrome plating, which forms decorative chromium layers (e.g., a chromium layer ranging from 0.25 micron to 1.0 micron).
[0024] The present disclosure further provides electrolyte solutions for electrodeposition of chromium-iron alloys and methods of forming chromium-iron alloys. In at least one aspect, electrolyte solutions of the present disclosure are aqueous. In at least one aspect, an electrolyte solution comprises an iron salt, such as, for example, ferrous sulfate or ferric chloride. It has been discovered that one or more of these iron salts present in an electrolyte solution provides deposition of thick chromium-iron alloy layers on a substrate, such as a steel substrate without the use of hexavalent chromium and pulsed plating. Electrolyte solutions of the present disclosure further comprise at least one complexing agent such as an oxalate compound, for example, sodium oxalate, which forms complexes with trivalent chromium ions as well as ferrous ions.
[0025] Electrolyte solutions of the present disclosure provide controllable chromium-iron alloy deposition on a substrate. In at least one aspect, a substrate is a steel substrate, copper substrate, brass substrate, copper-coated substrate, nickel-coated substrate, or other metal or metal alloy-containing substrate. In at least one aspect, the electrolyte solutions of the present disclosure when used at temperatures in a range of about 30.degree. C. to about 60.degree. C. in at a pH in a range of about 2 to about 4 provide satisfactorily hard chromium-iron alloy deposits that are considered comparable to conventional hexavalent chromium based coatings. In at least one aspect, iron content of a chromium-iron alloy of the present disclosure is from about 1 wt. % iron to about 20 wt. % iron based on the total weight of chromium and iron in the alloy.
[0026] Chromium-iron alloy coatings of the present disclosure provide similar hardness and wear resistance for substrates, such as steel substrates, to that of conventional hexavalent hard chrome plated substrates. The chromium-iron alloys of the present disclosure can be disposed on components of aircraft, spacecraft, watercraft, engine and blown-flap, exhaust-washed structures, warm-structure components for high-performance supersonic, hypersonic, and space re-entry vehicle structures, automobile parts, architectural structures such as steel bridges and propulsion structures such as power-generation turbines, vehicle engines, alternative-energy applications, and related technologies. As one specific example, alloys of the present disclosure can be disposed on steel-based landing gears and/or a bottom surface of an aircraft.
[0027] In at least one aspect, chromium-iron coatings of the present disclosure are formed using a single bath technique. The deposition vessel is a glass beaker at a lab scale or large polypropylene tanks for plating on a commercial scale. In at least one aspect, the deposition vessel contains an electrolyte solution that is prepared by mixing all ingredients of the electrolyte solution concurrently or in a stepwise manner starting with chromium salts and complexing agents followed by iron salts. The anode (e.g., graphite) is introduced into the beaker containing electrolyte solution, as described in more detail below. The deposition process is modulated by applying direct current, which creates the chromium-iron alloy coating. The thickness of the chromium-iron alloy coating can be controlled by the duration of the direct current applied to the electrolyte solution electrodes. In at least one aspect, the total thickness of a chromium-iron alloy coating is from about 1 micron to about 100 microns, such as from about 10 microns to about 50 microns, such as from about 20 microns to about 40 microns, for example about 30 microns.
[0028] Varying the thickness and composition of a chromium-iron alloy coating can be controlled by current density and time scale of a deposition process of the present disclosure.
[0029] Electrolyte solutions of the present disclosure comprise a metal salt. As used herein, metal salt can include anhydrous and /or hydrate forms of a metal salt. In at least one aspect, metal salts include one or more of a trivalent chromium salt and an iron salt. Electrolyte solutions of the present disclosure further comprise at least one complexing agent such as an alkali metal oxalate compound, for example, sodium oxalate or potassium oxalate. Complexing agents, such as alkali metal oxalate compounds, coordinate to iron ions in an electrolyte solution and promote controllable iron deposition on a substrate upon application of a current density to the electrolyte solution.
[0030] Electrolyte solutions of the present disclosure further comprise at least one buffering agent such as an aluminum salt, for example, aluminum sulfate or an aluminum halide. Buffering agents of the present disclosure maintain a desirable pH of the electrolyte solution and do not substantially interfere with chromium and iron deposition on a substrate, as described in more detail below.
[0031] Electrolyte solutions of the present disclosure further comprise at least one ionic conductivity control agent such as an alkali metal salt, for example, sodium sulfate or potassium sulfate. Ionic conductivity control agents of the present disclosure maintain a desirable conductivity of the electrolyte solution and do not substantially interfere with chromium and iron deposition on a substrate.
[0032] Electrolyte solutions of the present disclosure further comprise at least one alkali metal halide, for example, sodium fluoride or potassium fluoride. Alkali metal halides of the present disclosure provide the electrolyte solution with wetting and etching properties, and can help chromium adhesion during chrome plating.
[0033] Optionally, electrolyte solutions of the present disclosure further comprise at least one surfactant, for example, sodium lauryl sulfate, sodium lauryl ether sulfate, or potassium lauryl sulfate. Surfactants of the present disclosure reduce pitting and gas generation during chrome plating.
[0034] In at least one aspect, the pH of electrolyte solutions of the present disclosure is between about 1 and about 6, such as between about 1.5 and about 4, for example the pH is 2 or 4. In at least one aspect, the pH of electrolyte solutions of the present disclosure are controlled by addition of one or more bases, such as a sodium hydroxide (NaOH) solution, to increase the pH of the solution or addition of one or more acids, such as a sulfuric acid (H.sub.2SO.sub.4) solution, to decrease the pH of the solution. Chromium salts, iron salts, complexing agents, buffering agents, acids, and bases can be obtained from any suitable commercial source, such as MERCK-India or Sigma-Aldrich Co. LLC of St. Louis, Mo.
[0035] FIG. 1 is a flow diagram illustrating a method 100 for forming an electrolyte solution according to one or more aspects of the present disclosure. As shown in FIG. 1, at operation 110, method 100 includes dissolving a trivalent chromium salt in a medium such as water or an aqueous solution to form a first electrolyte solution. The trivalent chromium salt is a trivalent chromium source. In at least one aspect, the trivalent chromium salt includes a chromium (III) halide, chromium (III) sulfate (e.g., Cr.sub.2(SO.sub.4).sub.3, Cr.sub.2(SO.sub.4).sub.3.12H.sub.2O, and/or other chromium (III) sulfates), and/or other chromium (III) salts. The chromium (III) halide can include, for example, chromium (III) chloride (e.g., CrCl.sub.3, CrCl.sub.3.5H.sub.2O, CrCl.sub.3.6H.sub.2O, and/or other chromium (III) chlorides).
[0036] In at least one aspect, the concentration of the trivalent chromium salt in the electrolyte of the present disclosure ranges from about 0.1 moles per liter (mol/L) to about 1 mol/L, such as in a range from about 0.3 mol/L to about 0.9 mol/L, such as in a range from about 0.2 mol/L to about 0.7 mol/L, such as in a range from about 0.4 mol/L to about 0.7 mol/L of the electrolyte solution, for example, in a range from about 0.5 mol/L to about 0.6 mol/L. In at least one aspect, the amount of the trivalent chromium salt that is dissolved is about 0.1 mol/L, 0.2 mol/L, 0.3 mol/L, 0.4 mol/L, 0.5 mol/L, 0.6 mol/L, 0.7 mol/L, 0.8 mol/L, 0.9 mol/L, or 1 mol/L of the electrolyte solution, where any value can form an upper end point or a lower end point, as appropriate. At concentrations above 1 mol/L, it can become difficult to dissolve the trivalent chromium salt in the electrolyte leading to solubility issues.
[0037] In at least one aspect, the trivalent chromium salt is dissolved by stirring at ambient temperature, at room temperature, at about 25.degree. C., or at a temperature ranging from about 10.degree. C. to about 40.degree. C., for example, from about 20.degree. C. to about 30.degree. C. In at least one aspect, the temperature at which operation 110. is performed can be about 10.degree. C., 15.degree. C., 20.degree. C., 25.degree. C., 30.degree. C., 35.degree. C., or 40.degree. C., where any value can form an upper end point or a lower end point, as appropriate. In at least one aspect, the stirring can be performed for about 5 minutes, 10 minutes, 15 minutes, 20 minutes, 25 minutes, or 30 minutes, where any value can form an upper end point or a lower end point, as appropriate, or until all the trivalent chromium salt has been dissolved.
[0038] At operation 120, the method 100 further includes dissolving an oxalate compound for example, in water or an aqueous solution (such as the first solution) to form an electrolyte solution, such as a second electrolyte solution. The oxalate compound includes oxalate, which can function as a complexing agent. In at least one aspect, the oxalate compound includes an alkali metal oxalate (e.g., sodium oxalate (Na.sub.2C.sub.2O.sub.4), a potassium oxalate (K.sub.2C.sub.2O.sub.4), and/or other alkali metal oxalates) and/or an acid of oxalate (e.g., oxalic acid (H.sub.2C.sub.2O.sub.4) and/or other acids of oxalate).
[0039] In at least one aspect, the concentration of the oxalate compound in the electrolyte of the present disclosure is in a range from about 0.1 mol/L to about 2.0 mol/L, such as in a range from about 0.2 mol/L to about 1.2 mol/L such as in a range from about 0.1 mol/L to about 0.9 mol/L, such as in a range from about 0.2 mol/L to about 0.7 mol/L, such as in a range from about 0.4 mol/L to about 0.7 mol/L of the electrolyte solution, for example, in a range from about 0.5 mol/L to about 0.6 mol/L. In at least one aspect, the amount of the oxalate compound that is dissolved is about 0.1 mol/L, 0.2 mol/L, 0.4 mol/L, 0.6 mol/L, 0.8 mol/L, 1.0 mol/L, 1.2 mol/L, 1.4 mol/L, 1.6 mol/L, 1.8 mol/L, or 2.0 mol/L of the electrolyte solution, where any value can form an upper end point or a lower end point, as appropriate.
[0040] In at least one aspect, to dissolve the oxalate compound and form a complex of oxalate and trivalent chromium, the oxalate compound is put in solution (e.g., the solution resulting from operation 110. or another operation performed prior to operation 120), the solution can be heated to a higher temperature ranging from about 70.degree. C. to about 80.degree. C., and the solution can be stirred for about 1 hour to about 3 hours. In at least one aspect, the solution is cooled (e.g., to ambient temperature, to room temperature, to about 25.degree. C., or to a temperature ranging from about 20.degree. C. to about 30.degree. C.). Alternatively, the oxalate compound can be dissolved without heating, in which case a complex of oxalate and trivalent chromium is formed in 3 to 4 days. Advantageously, heating the solution to a temperature ranging from about 70.degree. C. to about 80.degree. C. at operation 120 allows the electrolyte solution to be prepared more quickly. In at least one aspect, the stirring is performed for about 1 hour, 1 hour and 15 minutes, 1 hour and 30 minutes, 1 hour and 45 minutes, 2 hours, 2 hours and 15 minutes, 2 hours and 30 minutes, 2 hours and 45 minutes, or 3 hours, where any value can form an upper end point or a lower end point, as appropriate. Further, in at least one aspect, the temperature at which operation 120 is performed is about 50.degree. C., 55.degree. C., 60.degree. C., 65.degree. C., 70.degree. C., 75.degree. C., or 80.degree. C., where any value can form an upper end point or a lower end point, as appropriate.
[0041] At operation 130, the method 100 further includes dissolving an iron salt, for example, in water or an aqueous solution (such as the second electrolyte solution) to form an electrolyte solution (e.g., a third electrolyte solution). In at least one aspect, the iron salt is a divalent iron salt, a trivalent iron salt, or a combination thereof. In at least one aspect, the iron salt is ferrous sulfate heptahydrate, ferric chloride, or a combination thereof. In at least one aspect, the iron salt is a divalent iron salt. In at least one aspect, the divalent iron salt includes iron (II) sulfate, iron (II) chloride, iron (II) acetate, and/or other divalent iron salts. Each of these divalent iron salts can include its respective hydrated forms. For example, iron (II) sulfate has the formula FeSO.sub.4.xH.sub.2O, where x is a whole number (e.g., 0, 1, 2, 4, 5, 6, or 7). Accordingly, in at least one aspect, iron (II) sulfate is anhydrous iron (II) sulfate, iron (II) sulfate monohydrate, iron (II) sulfate dihydrate, iron (II) sulfate tetrahydrate, iron (II) sulfate pentahydrate, iron (II) sulfate hexahydrate, iron (II) sulfate heptahydrate, or iron (II) sulfate with another hydration state. In at least one aspect, the iron salt is a trivalent iron salt. In at least one aspect, the trivalent iron salt includes iron (III) sulfate, iron (III) chloride, iron (III) acetate, and/or other trivalent iron salts. Each of these trivalent iron salts can include its respective hydrated forms. For example, iron (III) sulfate has the formula Fe.sub.2(SO.sub.4).sub.3.xH.sub.2O, where x is a whole number (e.g., 0, 1, 2, 4, 5, 6, or 7). Accordingly, in at least one aspect, iron (III) sulfate is anhydrous iron (III) sulfate, iron (III) sulfate monohydrate, iron (III) sulfate dihydrate, iron (III) sulfate tetrahydrate, iron (III) sulfate pentahydrate, iron (III) sulfate hexahydrate, iron (III) sulfate heptahydrate, or iron (III) sulfate with another hydration state. In a further example, iron (III) chloride has the formula FeCl.sub.3.xH.sub.2O, where x is a whole number (e.g., 0, 1, 2, 4, 5, 6, or 7). Accordingly, in at least one aspect, iron (III) chloride is anhydrous iron (III) chloride, iron (III) chloride monohydrate, iron (III) chloride dihydrate, iron (III) chloride tetrahydrate, iron (III) chloride pentahydrate, iron (III) chloride hexahydrate, iron (III) chloride heptahydrate, or iron (III) chloride with another hydration state.
[0042] In at least one aspect, the concentration of the iron salt in the electrolyte of the present disclosure ranges from about 0.005 mol/L to about 0.2 mol/L, such as in a range from about 0.01 mol/L to about 0.2 mol/L, such as in a range from about 0.02 mol/L to about 0.2 mol/L of the electrolyte solution, for example, in a range from about 0.1 mol/L to about 0.2 mol/L. In at least one aspect, the amount of the iron salt that is dissolved is about 0.005 mol/L, 0.01 mol/L, 0.02 mol/L, 0.03 mol/L, 0.04 mol/L, 0.05 mol/L, 0.06 mol/L, 0.07 mol/L, 0.08 mol/L, 0.09 mol/L, 0.1 mol/L, or 0.2 mol/L of the electrolyte solution, where any value can form an upper end point or a lower end point, as appropriate. It has been found by the inventors that at concentrations above 0.2 mol/L, the deposited chromium-iron alloy coating becomes soft and can also be more susceptible to corrosion.
[0043] In at least one aspect, the iron salt is dissolved by stirring at ambient temperature, at room temperature, at about 25.degree. C., or at a temperature ranging from about 20.degree. C. to about 30.degree. C. In at least one aspect, the stirring is performed for about 5 minutes, 10 minutes, 15 minutes, 20 minutes, 25 minutes, or 30 minutes, where any value can form an upper end point or a lower end point, as appropriate, or until all the iron salt has been dissolved. In at least one aspect, the temperature at which operation 130 is performed is about 10.degree. C., 15.degree. C., 20.degree. C., 25.degree. C., 30.degree. C., 35.degree. C., or 40.degree. C., where any value can form an upper end point or a lower end point, as appropriate.
[0044] At operation 140, the method 100 further includes dissolving a metal salt, for example, in water or an aqueous solution (such as the third electrolyte solution) to form an electrolyte solution (e.g., a fourth electrolyte solution). The metal salt is a metal ion source that dissolves to provide metal ions such as aluminum ions, which can function as a buffer and can provide ionic strength due to the high valence of the metal ion in solution (e.g., Al.sup.3+). In at least one aspect, the metal salt includes a group 13 metal salt such as an aluminum salt (e.g., aluminum sulfate (Al.sub.2(SO.sub.4).sub.3), an aluminum halide such as aluminum chloride (Al Cl.sub.3), and/or other aluminum salts) and/or other metal salts.
[0045] In at least one aspect, the concentration of the metal salt in the electrolyte of the present disclosure ranges from about 0.01 mol/L to about 1.0 mol/L, such as in a range from about 0.05 mol/L to about 0.8 mol/L, such as in a range from about 0.1 mol/L to about 0.7 mol/L, such as in a range from about 0.2 mol/L to about 0.5 mol/L of the electrolyte solution, for example, in a range from about 0.2 mol/L to about 0.3 mol/L. In at least one aspect, the amount of the metal salt that is dissolved is about 0.01 mol/L, 0.05 mol/L, 0.1 mol/L, 0.2 mol/L, 0.3 mol/L, 0.4 mol/L, 0.5 mol/L, 0.6 mol/L, 0.8 mol/L, or 1.0 mol/L of the electrolyte solution, where any value can form an upper end point or a lower end point, as appropriate. At concentrations above 1.0 mol/L, it can become difficult to dissolve the metal salt in the electrolyte leading to solubility issues.
[0046] In at least one aspect, the metal salt is dissolved by stirring at ambient temperature, at room temperature, at about 25.degree. C., or at a temperature ranging from about 20.degree. C. to about 30.degree. C. In at least one aspect, the stirring is performed for about 5 minutes, 10 minutes, 15 minutes, 20 minutes, 25 minutes, or 30 minutes, where any value can form an upper end point or a lower end point, as appropriate, or until all the metal salt has been dissolved. In at least one aspect, the temperature at which operation 140 is performed is about 10.degree. C., 15.degree. C., 20.degree. C., 25.degree. C., 30.degree. C., 35.degree. C., or 40.degree. C., where any value can form an upper end point or a lower end point, as appropriate.
[0047] At operation 150, the method 100 further includes dissolving an alkali metal salt, for example, in water or an aqueous solution (such as the fourth electrolyte solution) to form an electrolyte solution (e.g., a fifth electrolyte solution). The alkali metal salt can increase the conductivity of the electrolyte solution. In at least one aspect, the alkali metal salt includes an alkali metal sulfate (e.g., sodium sulfate (Na.sub.2SO.sub.4), potassium sulfate (K.sub.2SO.sub.4), and/or other alkali metal sulfates).
[0048] In at least one aspect, the concentration of the alkali metal salt in the electrolyte of the present disclosure is in a range from about 0.1 mol/L to about 2.0 mol/L, such as in a range from about 0.5 mol/L to about 2.0 mol/L such as in a range from about 1.0 mol/L to about 1.5 mol/L, for example, in a range from about 1.3 mol/L to about 1.4 mol/L. In at least one aspect, the amount of the oxalate compound that is dissolved is about 0.1 mol/L, 0.2 mol/L, 0.4 mol/L, 0.6 mol/L, 0.8 mol/L, 1.0 mol/L, 1.2 mol/L, 1.4 mol/L, 1.6 mol/L, 1.8 mol/L, or 2.0 mol/L of the electrolyte solution, where any value can form an upper end point or a lower end point, as appropriate. At concentrations above 2.0 mol/L, it can become difficult to dissolve the alkali metal salt in the electrolyte leading to solubility issues.
[0049] In at least one aspect, the alkali metal salt is dissolved by stirring at ambient temperature, at room temperature, at about 25.degree. C., or at a temperature ranging from about 10.degree. C. to about 40.degree. C., for example, from about 20.degree. C. to about 30.degree. C. In at least one aspect, the temperature at which operation 150 is performed can be about 10.degree. C., 15.degree. C., 20.degree. C., 25.degree. C., 30.degree. C., 35.degree. C., or 40.degree. C., where any value can form an upper end point or a lower end point, as appropriate. In at least one aspect, the stirring can be performed for about 5 minutes, 10 minutes, 15 minutes, 20 minutes, 25 minutes, or 30 minutes, where any value can form an upper end point or a lower end point, as appropriate, or until all the alkali metal salt has been dissolved. In at least one aspect, the alkali metal salt is dissolved by stirring for 15 minutes at ambient temperature, at room temperature, at about 25.degree. C.
[0050] At operation 160, the method 100 further includes dissolving an alkali metal halide, for example, in water or an aqueous solution (such as the fifth electrolyte solution) to form an electrolyte solution (e.g., a sixth electrolyte solution). The alkali metal halide can provide the electrolyte solution with wetting and etching properties, and can help chromium adhesion during chrome plating. In at least one aspect, the alkali metal halide includes an alkali metal fluoride (e.g., sodium fluoride (NaF), potassium fluoride (KF), and/or other alkali metal fluorides) and/or other alkali metal halides.
[0051] In at least one aspect, the concentration of the alkali metal halide in the electrolyte of the present disclosure is in a range from about 0.1 mol/L to about 1.0 mol/L, such as in a range from about 0.1 mol/L to about 0.5 mol/L such as in a range from about 0.2 mol/L to about 0.5 mol/L, for example, in a range from about 0.3 mol/L to about 0.4 mol/L. In at least one aspect, the amount of the oxalate compound that is dissolved is about 0.1 mol/L, 0.2 mol/L, 0.3 mol/L, 0.4 mol/L, 0.5 mol/L, 0.6 mol/L, 0.8 mol/L, or 1.0 mol/L of the electrolyte solution, where any value can form an upper end point or a lower end point, as appropriate. At concentrations above 1.0 mol/L, it can become difficult to dissolve the alkali metal halide in the electrolyte leading to solubility issues.
[0052] In at least one aspect, the alkali metal halide is dissolved by stirring at ambient temperature, at room temperature, at about 25.degree. C., or at a temperature ranging from about 10.degree. C. to about 40.degree. C., for example, from about 20.degree. C. to about 30.degree. C. In at least one aspect, the temperature at which operation 160 is performed is about 10.degree. C., 15.degree. C., 20.degree. C., 25.degree. C., 30.degree. C., 35.degree. C., or 40.degree. C., where any value can form an upper end point or a lower end point, as appropriate. In at least one aspect, the stirring is performed for about 5 minutes, 10 minutes, 15 minutes, 20 minutes, 25 minutes, or 30 minutes, where any value can form an upper end point or a lower end point, as appropriate, or until all the alkali metal halide has been dissolved. In at least one aspect, the alkali metal halide is dissolved by stirring for 15 minutes at ambient temperature, at room temperature, at about 25.degree. C.
[0053] Optionally, at operation 170, the method 100 further includes dissolving a surfactant, for example, in water or an aqueous solution (such as the sixth electrolyte solution) to form an electrolyte solution (e.g., a seventh electrolyte solution). The surfactant can prevent or reduce pitting and reduce gas generation (e.g., chlorine gas, hydrogen gas, etc.) during chrome plating. In at least one aspect, the surfactant includes sodium lauryl sulfate (NaC.sub.12H.sub.25SO.sub.4), sodium lauryl ether sulfate (CH.sub.3(CH.sub.2).sub.11(OCH.sub.2CH.sub.2).sub.nOSO.sub.3Na), potassium lauryl sulfate (KC.sub.12H.sub.25 SO.sub.4), and/or other surfactants.
[0054] In at least one aspect, the concentration of the surfactant in the electrolyte of the present disclosure is in a range from about 0.0001 mol/L to about 0.01 mol/L. In at least one aspect, the amount of the surfactant that is dissolved can be about 0.0001 mol/L, 0.0002 mol/L, 0.0004 mol/L, 0.0006 mol/L, 0.0008 mol/L, 0.0010 mol/L, 0.0020 mol/L, 0.0040 mol/L, 0.0060 mol/L, 0.0080 mol/L, or 0.0100 mol/L of the electrolyte solution, where any value can form an upper end point or a lower end point, as appropriate. For example, in at least one aspect, the amount of sodium lauryl sulfate, sodium lauryl ether sulfate, or potassium lauryl sulfate is in a range from about 0.1 g to about 1 g per liter of the electrolyte solution. At concentrations above 0.01 mol/L, the surfactant can cause excessive foaming of the bath and nonuniform deposition.
[0055] Optionally, at operation 180 the method 100 further includes adjusting the pH of the electrolyte solution using one or more aqueous acid solutions or aqueous base solutions, such as potassium hydroxide (KOH), sodium hydroxide (NaOH), and/or sulfuric acid (H.sub.2SO.sub.4). The volume of aqueous acid solution or aqueous base solution added to the electrolyte solution is sufficiently small such that the concentration of other components (complexing agents, buffering agents, etc.) of the electrolyte solution is not substantially affected. Alternatively, solid potassium hydroxide and/or solid sodium hydroxide is added directly to the electrolyte solution and/or concentrated sulfuric acid is added directly to the electrolyte solution. In at least one aspect, the pH of the electrolyte solution is adjusted to a target pH from about 1 to about 7, such as from about 1 to about 5, such as from about 1.5 to about 4, for example 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, or 5. In at least one aspect, the pH of an electrolyte solution of the present disclosure is adjusted before passing a current through the electrolyte solution (as described in more detail below). In at least one aspect, the pH of an electrolyte solution of the present disclosure is maintained at a target pH or a target pH range during the passing of a current through the electrolyte solution.
[0056] Optionally, at operation 190, time is provided to reach equilibrium state. In at least one aspect, the solution is left to stand for a time ranging from 1 hour to 2 days to reach the equilibrium state. The time provided to reach the equilibrium state can be about 1 hour, 3 hours, 6 hours, 9 hours, 12 hours, 15 hours, 18 hours, 21 hours, 24 hours, 27 hours, 30 hours, 33 hours, 36 hours, 39 hours, 42 hours, 45 hours, or 48 hours, where any value can form an upper end point or a lower end point, as appropriate.
[0057] In at least one aspect, the method 100 is performed in the order presented. Alternatively, the method 100 is performed in a different order. In at least one aspect, some operations are performed in order while other operations are performed in a different order.
[0058] For example, operations 110 and 120 are performed in order, while operations 130, 140, 150, 160, 170, 180, and 190 are performed in a different order after operations 110 and 120. In another example, operations 110 and operations 120 are performed in a different order while operations 130, 140, 150, 160, 170, 180, and 190 are performed in order. In another example, operations 110, 120, 130, 140, 150, and 160 are performed in order, while operations 170, 180, and 190 are performed in a different order. A group of operations can be performed before another group of operations. For example, operations 110 and 120 can be performed in any order, and after operations 110 and 120 are performed, operations 130, 140, 150, and 160 are performed in any order. Other orders are contemplated as one skilled in the art will appreciate. Further, one or more of operations 170, 180, and 190 are omitted in some aspects.
[0059] FIG. 2 is a flow diagram illustrating a method 200 for forming a chromium alloy coating on a substrate by electrodeposition according to one or more aspects of the present disclosure. At operation 210, an electrolyte solution is prepared, such as by the method 100 of FIG. 1. At operation 220, the method 200 further includes adjusting and/or maintaining a pH of the electrolyte solution at a target pH or a target pH range. In at least one aspect, the target pH is a pH ranging from about 1 to about 4. The pH can be maintained at about 1, 1.2, 1.4, 1.5, 1.6, 1.8, 2, 2.2, 2.4, 2.6, 2.8, 3.0, 3.2, 3.4, 3.6, 3.8, or 4.0, where any value can form an upper end point or a lower end point, as appropriate.
[0060] At operation 230, the method 200 further includes adjusting and/or maintaining a temperature of an electrolyte solution, such as the electrolyte solution formed by operation 210. In at least one aspect, the temperature is adjusted to/maintained at a target temperature of from about 20.degree. C. to about 70.degree. C., such as from about 20.degree. C. to about 40.degree. C., such as from about 20.degree. C. to about 35.degree. C., for example 20.degree. C., 25.degree. C., or 30.degree. C., using any suitable heating or cooling apparatus. In at least one aspect, the temperature of the electrolyte solution is adjusted before passing a current through the electrolyte solution. In at least one aspect, the temperature of the electrolyte solution is maintained during the passing of a current through the electrolyte solution to maintain the appearance of a deposited layer. Maintaining the temperature within a desirable range promotes obtaining reproducible results in terms of appearance and alloy composition.
[0061] At operation 240, the method further includes introducing a cathode and an anode into the electrolyte solution, the cathode including the substrate and at operation 250, passing a current between the cathode and the anode through the electrolyte solution to deposit a chromium alloy onto the cathodic substrate. In at least one aspect, the cathodic substrate is, for example, a steel substrate, a ferrous alloy substrate, a copper substrate, a brass substrate, a nickel substrate, a copper-coated substrate (e.g., copper-coated steels or copper-coated ferrous alloys), or a nickel-coated substrate (e.g., nickel-coated steels or nickel-coated ferrous alloys).
[0062] In at least one aspect, the anode includes a carbonaceous electrode material. For example, the carbonaceous anode can be a graphite anode or other anode that includes carbon. In at least one aspect, the graphite anode is used for chloride-based electrolyte solutions (e.g., electrolyte solutions that include one or more compounds with chloride such as chromium (III) chloride), sulfate-based electrolyte solutions (e.g., electrolyte solutions that include one or more compounds with sulfate such as chromium (III) sulfate), or chloride and sulfate-based electrolyte solutions (e.g., electrolyte solutions that include one or more compounds with chloride and one or more other compounds with sulfate). Advantageously, the graphite anode or other carbonaceous anode minimizes gas evolution and formation of undesirable byproducts, as well as facilitating a desirable deposition rate (e.g., ranging from about 1 micron to about 2 microns per minute). Alternatively, a platinum anode or a platinized titanium anode can be used for sulfate-based electrolyte solutions (e.g., electrolyte solutions that include one or more compounds with sulfate such as chromium (III) sulfate). For example, the platinum anode or platinized titanium anode can be used when the electrolyte solution does not include compounds with chloride such that chlorine gas is not produced, or when the electrolyte solution has less chloride such that less chlorine gas is generated (e.g., there is no need to reduce the generation of chlorine gas using a carbonaceous anode).
[0063] In at least one aspect, passing a current between the cathode and the anode is performed using direct current. In at least one aspect, direct current having a current density of from about 50 mA/cm.sup.2 to about 600 mA/cm.sup.2, such as from about 100 mA/cm.sup.2 to about 500 mA/cm.sup.2, such as from about 100 mA/cm.sup.2 to about 400 mA/cm.sup.2, such as from about 200 mA/cm.sup.2 to about 400 mA/cm.sup.2, for example 200 mA/cm.sup.2, 250 mA/cm.sup.2, or 300 mA/cm.sup.2 is used. The value of the current density can be adjusted depending on the separation between the cathode and anode. In at least one aspect, the current density is about 50 mA/cm.sup.2, 100 mA/cm.sup.2, 150 mA/cm.sup.2, 200 mA/cm.sup.2, 250 mA/cm.sup.2, 300 mA/cm.sup.2, 350 mA/cm.sup.2, 400 mA/cm.sup.2, 450 mA/cm.sup.2, or 500 mA/cm.sup.2, where any value can form an upper end point or a lower end point, as appropriate, depending on the separation between the cathode and anode. For example, a current density ranging from about 200 mA/cm.sup.2 to about 400 mA/cm.sup.2 can be applied when the cathode and the anode is separated by about 3 cm. It has been found by the inventors that a current density in the aforementioned range minimizes the formation of undesirable hexavalent chromium byproducts while achieving a reasonable rate of deposition, for example, 0.5 to 1 micron/minute.
[0064] In response to passing a current between the cathode and the anode, chromium and iron deposit onto the cathodic substrate. Operation 250 is performed until a chromium alloy coating layer having a desired thickness is formed on the substrate. In at least one aspect, the chromium alloy coating layer is a chromium-iron alloy having from about 1 wt. % iron to about 60 wt. % iron, such as from about 1 wt. % iron to about 20 wt. % iron, such as from about 1 wt. % iron to about 5 wt. % iron or from about 10 wt. % iron to about 20 wt. % iron based on the total weight of the alloy. For example, the chromium-iron alloy can have a wt. % iron of about 1 wt. %, 2 wt. %, 10 wt. %, 11 wt. %, or 12 wt. %. Furthermore, the chromium-iron alloy has from about 80 wt. % chromium to about 99 wt. % chromium, such as from about 85 wt. % chromium to about 95 wt. % chromium, for example about 99 wt. % chromium, 98 wt. % chromium, 90 wt. % chromium, 89 wt. % chromium, or 88 wt. % chromium, based on the total weight of the alloy.
[0065] In response to performing operation 250, chromium is deposited on the substrate. In at least one aspect, chromium and carbon are co-deposited on the substrate. In at least one aspect, operation 250 is performed until a chromium layer (e.g., a chromium coating) or a chromium-carbon layer (e.g., a chromium carbide coating) having a desired thickness (e.g., a thickness greater than about 5 microns) is formed on the substrate. In at least one aspect, the chromium layer having a thickness greater than about 5 microns can have hardness greater than about 800 HV.
[0066] Aspects
[0067] Clause 1. An electrolyte solution for electroplating comprising: a trivalent chromium salt; an oxalate compound; an iron salt; an aluminum sulfate; an alkali metal sulfate; and an alkali metal halide.
[0068] Clause 2. The electrolyte solution of clause 1, wherein the trivalent chromium salt is present in an amount ranging from about 0.3 mol per liter to about 0.9 mol per liter of the electrolyte solution; the oxalate compound is present in an amount ranging from about 0.2 mol per liter to about 1.2 mol per liter of the electrolyte solution; the iron salt is present in an amount ranging from about 0.005 mol per liter to about 0.2 mol per liter of the electrolyte solution; the aluminum sulfate is present in an amount ranging from about 0.05 mol per liter to about 0.5 mol per liter; the alkali metal sulfate is present in an amount ranging from about 0.1 mol per liter to about 2.0 mol per liter of the electrolyte solution; and the alkali metal halide is present in an amount ranging from about 0.1 mol per liter to about 0.5 mol per liter of the electrolyte solution.
[0069] Clause 3. The electrolyte solution of clause 1 or 2, wherein the iron salt is a divalent iron salt comprising one or more of iron (II) sulfate, iron (II) chloride, iron (II) acetate, and hydrates thereof.
[0070] Clause 4. The electrolyte solution of any of clauses 1 to 3, wherein the iron salt is a trivalent iron salt comprising one or more of iron (III) sulfate, iron (III) chloride, iron (III) acetate, and hydrates thereof.
[0071] Clause 5. The electrolyte solution of any of clauses 1 to 4, wherein the trivalent chromium salt is selected from a chromium (III) halide, a chromium (III) sulfate, or a combination thereof.
[0072] Clause 6. The electrolyte solution of any of clauses 1 to 5, wherein the oxalate compound is selected from sodium oxalate, potassium oxalate, an acid of oxalate, or a combination thereof.
[0073] Clause 7. The electrolyte solution of any of clauses 1 to 6, wherein the alkali metal sulfate is selected from sodium sulfate, potassium sulfate, or a combination thereof.
[0074] Clause 8. The electrolyte solution of any of clauses 1 to 7, wherein the alkali metal halide is selected from sodium fluoride, potassium fluoride, or a combination thereof.
[0075] Clause 9. The electrolyte solution of any of clauses 1 to 8, wherein the pH of the electrolyte solution is in a range from about 1 to about 4.
[0076] Clause 10. The electrolyte solution of any of clauses 1 to 9, further comprising sodium lauryl sulfate, sodium lauryl ether sulfate, or a combination thereof.
[0077] Clause 11. An electrolyte solution for electroplating comprising a trivalent chromium salt in an amount ranging from about 0.3 mol per liter to about 0.9 mol per liter of the electrolyte solution; an oxalate compound in an amount ranging from about 0.2 mol per liter to about 1.2 mol per liter of the electrolyte solution; an iron salt in an amount ranging from about 0.005 mol per liter to about 0.2 mol per liter of the electrolyte solution; an aluminum sulfate in an amount ranging from about 0.05 mol per liter to about 0.5 mol per liter; an alkali metal sulfate in an amount ranging from about 0.1 mol per liter to about 2.0 mol per liter of the electrolyte solution; and an alkali metal halide in an amount ranging from about 0.1 mol per liter to about 0.5 mol per liter of the electrolyte solution.
[0078] Clause 12. The electrolyte solution of clause 11, wherein the iron salt is a divalent iron salt comprising one or more of iron (II) sulfate, iron (II) chloride, iron (II) acetate, and hydrates thereof.
[0079] Clause 13. The electrolyte solution of clause 11 or 12, wherein the iron salt is a trivalent iron salt comprising one or more of iron (III) sulfate, iron (III) chloride, iron (III) acetate, and hydrates thereof.
[0080] Clause 14. The electrolyte solution of any of clauses 11 to 13, wherein the trivalent chromium salt is selected from a chromium (III) halide, a chromium (III) sulfate, or a combination thereof.
[0081] Clause 15. The electrolyte solution of any of clauses 11 to 14, wherein the oxalate compound is selected from sodium oxalate, potassium oxalate, an acid of oxalate, or a combination thereof.
[0082] Clause 16. The electrolyte solution of any of clauses 11 to 15, wherein the alkali metal sulfate is selected from sodium sulfate, potassium sulfate, or a combination thereof.
[0083] Clause 17. The electrolyte solution of any of clauses 11 to 16, wherein the alkali metal halide is selected from sodium fluoride, potassium fluoride, or a combination thereof.
[0084] Clause 18. The electrolyte solution of any of clauses 11 to 17, wherein the pH of the electrolyte solution is in a range from about 1 to about 4.
[0085] Clause 19. The electrolyte solution of any of clauses 11 to 18, further comprising sodium lauryl sulfate, sodium lauryl ether sulfate, or a combination thereof.
[0086] Clause 20. A method of chrome plating on a substrate using an electrolyte solution, comprising dissolving in an aqueous medium a trivalent chromium salt in an amount ranging from about 0.3 mol per liter to about 0.9 mol per liter of the electrolyte solution; dissolving a oxalate compound in an amount ranging from about 0.2 mol per liter to about 1.2 mol per liter of the electrolyte solution; dissolving iron salt in an amount ranging from about 0.005 mol per liter to about 0.2 mol per liter of the electrolyte solution; dissolving aluminum sulfate in an amount ranging from about 0.05 mol per liter to about 0.5 mol per liter; dissolving an alkali metal sulfate in an amount ranging from about 0.1 mol per liter to about 2.0 mol per liter of the electrolyte solution; dissolving an alkali metal halide in an amount ranging from about 0.1 mol per liter to about 0.5 mol per liter of the electrolyte solution; and passing a current between a cathode and an anode through the electrolyte solution to deposit chromium on a substrate.
[0087] Clause 21. The method of clause 20, wherein the cathode is a steel substrate, a copper substrate, a brass substrate, a nickel substrate, a copper-coated substrate, or a nickel-coated substrate.
[0088] Clause 22. The method of clause 20 or 21, wherein the anode is a platinum material, platinized titanium material, or a carbonaceous electrode material.
[0089] Clause 23. The method of any of clauses 20 to 22, wherein the current has a current density in a range from about 150 to about 600 mA/cm.sup.2 by passing direct current between the anode and the cathode.
[0090] Clause 24. The method of any of clauses 20 to 23, wherein the current density has a current density in a range from about 200 to about 400 mA/cm.sup.2.
[0091] Clause 25. The method of any of clauses 20 to 24, wherein the electrolyte solution is maintained at a temperature in a range from about 20 degrees Celsius and about 60 degrees Celsius.
[0092] Clause 26. The method of any of clauses 20 to 25, further comprising adjusting a pH of the electrolyte solution to a pH in a range from about 1.5 to about 4.
[0093] Clause 27. The method of any of clauses 20 to 26, wherein the iron salt is a divalent iron salt comprising one or more of iron (II) sulfate, iron (II) chloride, iron (II) acetate, and hydrates thereof.
[0094] Clause 28. The method of any of clauses 20 to 27, wherein the iron salt is a trivalent iron salt comprising one or more of iron (III) sulfate, iron (III) chloride, iron (III) acetate, and hydrates thereof.
[0095] Clause 29. The method of any of clauses 20 to 28, wherein the trivalent chromium salt is selected from a chromium (III) halide, a chromium (III) sulfate, or a combination thereof.
[0096] Clause 30. The method of any of clauses 20 to 29, wherein the oxalate compound is selected from sodium oxalate, potassium oxalate, an acid of oxalate, or a combination thereof.
[0097] Clause 31. The method of any of clauses 20 to 30, wherein the alkali metal sulfate is selected from sodium sulfate, potassium sulfate, or a combination thereof.
[0098] Clause 32. The method of any of clauses 20 to 31, wherein the alkali metal halide is selected from sodium fluoride, potassium fluoride, or a combination thereof.
[0099] Clause 33. The method of any of clauses 20 to 32, further comprising dissolving sodium lauryl sulfate, sodium lauryl ether sulfate, or a combination thereof in an amount ranging from about 0.1 grams per liter to about 1 gram per liter of the electrolyte solution.
[0100] Clause 34. A method of chrome plating on a substrate using an electrolyte solution, comprising: introducing a cathode and an anode into an electrolyte solution comprising a trivalent chromium salt, an oxalate compound, an iron salt, an aluminum sulfate, an alkali metal sulfate, and an alkali metal halide; and passing a current between the anode and the cathode through the electrolyte to deposit a chromium layer on the substrate.
[0101] Clause 35. The method of clause 34, wherein the cathode is a steel substrate, a copper substrate, a brass substrate, a nickel substrate, a copper-coated substrate, or a nickel-coated substrate.
[0102] Clause 36. The method of clause 34 or 35, wherein the anode is a platinum material, platinized titanium material, or a carbonaceous electrode material.
[0103] Clause 37. The method of any of clauses 34 to 36, wherein the current has a current density in a range from about 100 to about 600 mA/cm.sup.2 by passing direct current between the anode and the cathode.
[0104] Clause 38. The method of any of clauses 34 to 37, wherein the current density has a current density in a range from about 200 to about 400 mA/cm.sup.2.
[0105] Clause 39. The method of any of clauses 34 to 38, wherein the electrolyte solution is maintained at a temperature in a range from about 20 degrees Celsius and about 60 degrees Celsius.
[0106] Clause 40. The method of any of clauses 34 to 39, further comprising adjusting a pH of the electrolyte solution to a pH in a range from about 1.5 to about 4.
[0107] Clause 41. The method of any of clauses 34 to 40, wherein the iron salt is a divalent iron salt comprising one or more of iron (II) sulfate, iron (II) chloride, iron (II) acetate, and hydrates thereof.
[0108] Clause 42. The method of any of clauses 34 to 41, wherein the iron salt is a trivalent iron salt comprising one or more of iron (III) sulfate, iron (III) chloride, iron (III) acetate, and hydrates thereof.
[0109] Clause 43. The method of any of clauses 34 to 42, wherein the trivalent chromium salt is selected from a chromium (III) halide, a chromium (III) sulfate, or a combination thereof.
[0110] Clause 44. The method of any of clauses 34 to 43, wherein the oxalate compound is selected from sodium oxalate, potassium oxalate, an acid of oxalate, or a combination thereof.
[0111] Clause 45. The method of any of clauses 34 to 44, wherein the alkali metal sulfate is selected from sodium sulfate, potassium sulfate, or a combination thereof.
[0112] Clause 46. The method of any of clauses 34 to 45, wherein the alkali metal halide is selected from sodium fluoride, potassium fluoride, or a combination thereof.
[0113] Clause 47. The method of any of clauses 34 to 46, further comprising dissolving sodium lauryl sulfate, sodium lauryl ether sulfate, or a combination thereof in an amount ranging from about 0.1 grams per liter to about 1 gram per liter of the electrolyte solution.
[0114] Clause 48. A substrate comprising: a chromium-iron alloy coating having a chromium content in a range of about 40 wt. % to about 90 wt. %, an iron content in a range of about 8 wt. % to about 18 wt. %, and a carbon content in the range of about 5 wt. % to about 50 wt. %.
[0115] Clause 49. The substrate of clause 48, wherein the substrate comprises one or more of steel, copper, brass, or nickel.
[0116] Clause 50. The substrate of clause 48 or 49, wherein the coating has a thickness of from about 1 micron and about 100 microns.
EXAMPLES
[0117] The following non-limiting examples are provided to further illustrate aspects described herein. However, the examples are not intended to be all-inclusive and are not intended to limit the scope of the aspects described herein.
[0118] Example 1
[0119] The components of Example 1 were mixed in a stepwise manner with the chromium chloride hexahydrate and the sodium oxalate mixed first followed by the metal salts. The pH of Example 1 was .about.2.2.
TABLE-US-00001 Chromium chloride hexahydrate 159 g/L 0.6 M (CrCl.sub.3.cndot.6H.sub.2O) Sodium oxalate 80.4 g/L 0.6 M (Na.sub.2C.sub.2O.sub.4) Ferrous sulfate hetpahydrate 2.78 g/L 0.01 M (FeSO.sub.4.cndot.7H.sub.2O) Aluminum sulfate (Al.sub.2(SO.sub.4).sub.3) 126.1 g/L 0.2 M Sodium sulfate (Na.sub.2SO.sub.4) 184.6 g/L 1.3 M Sodium fluoride (NaF) 16.8 g/L 0.4 M
[0120] Example 2
[0121] The components of Example 2 were mixed in a stepwise manner with the chromium chloride hexahydrate and the sodium oxalate mixed first followed by the metal salts. The components of Example 2 are similar to the components of Example 1, except that the amount of ferrous sulfate heptahydrate was increased 10 times compared to the components of Example 1. The pH of Example 1 was .about.2.2.
TABLE-US-00002 Chromium chloride hexahydrate 159 g/L 0.6 M (CrCl.sub.3.cndot.6H.sub.2O) Sodium oxalate 80.4 g/L 0.6 M (Na.sub.2C.sub.2O.sub.4) Ferrous sulfate hetpahydrate 27.8 g/L 0.1 M (FeSO.sub.4.cndot.7H.sub.2O) Aluminum sulfate (Al.sub.2(SO.sub.4).sub.3) 126.1 g/L 0.2 M Sodium sulfate (Na.sub.2SO.sub.4) 184.6 g/L 1.3 M Sodium fluoride (NaF) 16.8 g/L 0.4 M
[0122] Example 3
[0123] The components of Example 3 were mixed in a stepwise manner with the chromium chloride hexahydrate and the sodium oxalate mixed first followed by the metal salts. The components of Example 3 are similar to the components of Example 1, except that ferrous sulfate heptahydrate is replaced with ferric chloride in Example 3. The pH of Example 1 was approximately 2.2.
TABLE-US-00003 Chromium chloride hexahydrate 159 g/L 0.6 M (CrCl.sub.3.cndot.6H.sub.2O) Sodium oxalate 80.4 g/L 0.6 M (Na.sub.2C.sub.2O.sub.4) Ferric chloride 1.62 g/L 0.01 M (FeCl.sub.3) Aluminum sulfate (Al.sub.2(SO.sub.4).sub.3) 126.1 g/L 0.2 M Sodium sulfate (Na.sub.2SO.sub.4) 184.6 g/L 1.3 M Sodium fluoride (NaF) 16.8 g/L 0.4 M
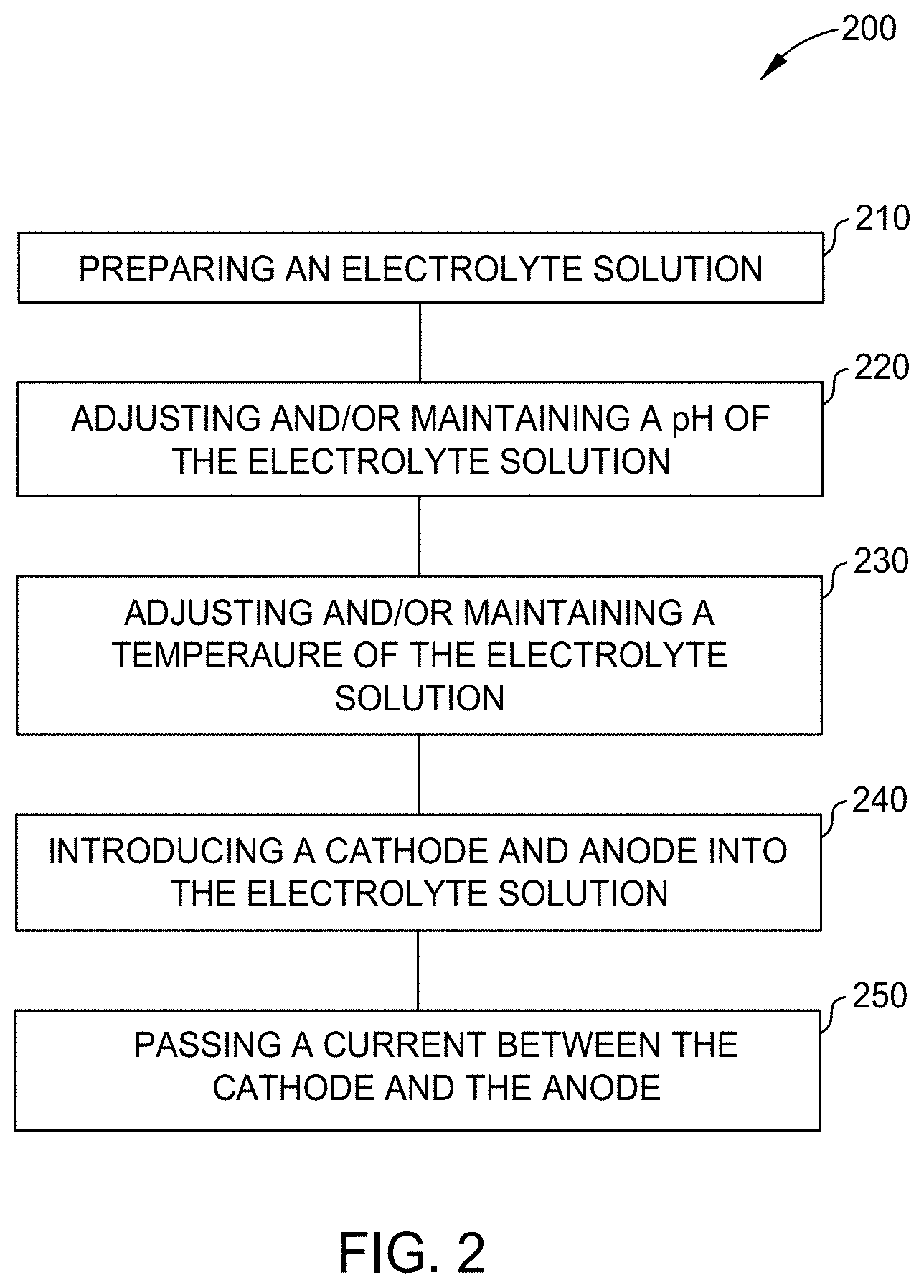
[0124] FIG. 3 is an image 300 of chromium-iron alloy plated substrates 310, 320, 330, and 340, formed by the process of FIG. 2, each plated at a different current density using the electrolyte solution of Example 1. For each chromium-iron alloy plated substrate 310, 320, 330, and 340, the plating parameters were a plating time of approximately 1 hour while maintaining the electrolyte solution at a temperature of 30.degree. C. and a pH of approximately 2.2. Chromium-iron alloy plated substrate 310 was plated at a current density of 100 mA/cm.sup.2 resulting in a chromium-iron alloy layer having a thickness of 12 .mu.m. Chromium-iron alloy plated substrate 320 was plated at a current density of 200 mA/cm.sup.2 resulting in a chromium-iron alloy layer having a thickness of 15 .mu.m. Chromium-iron alloy plated substrate 330 was plated at a current density of 250 mA/cm.sup.2 resulting in a chromium-iron alloy layer having a thickness of 28 .mu.m. Chromium-iron alloy plated substrate 340 was plated at a current density of 300 mA/cm.sup.2 resulting in a chromium-iron layer having a thickness of 32 .mu.m. X-ray fluorescence was performed on a portion of each coating to determine the chromium content and the iron content by wt. %. It is noted that X-ray fluorescence does not detect carbon, thus in at least one aspect, the chromium-iron alloys described herein also contain carbon even though carbon is not shown in the X-ray fluorescence results. The results are depicted in Table I.
[0125] As illustrated in Table I, any current density ranging from 100 mA/cm.sup.2 to about 300 mA/cm.sup.2 provides deposition of a chromium-iron alloy layer. A current density ranging from about 200 mA/cm.sup.2 to about 280 mA/cm.sup.2 provides a thick chromium-iron alloy layer with a low iron content. Further, a current density of 100 mA/cm.sup.2 provides the thinnest chromium-iron alloy with a high iron content relative to a current density ranging from about 200 mA/cm.sup.2 to about 300 mA/cm.sup.2.
TABLE-US-00004 TABLE I Current Density Chromium Iron Thickness (mA/cm.sup.2) (wt. %) (wt. %) (.mu.m) 100 10.2 89.8 12 200 82.4 17.6 15 250 92.7 7.3 28 300 94.3 5.7 32
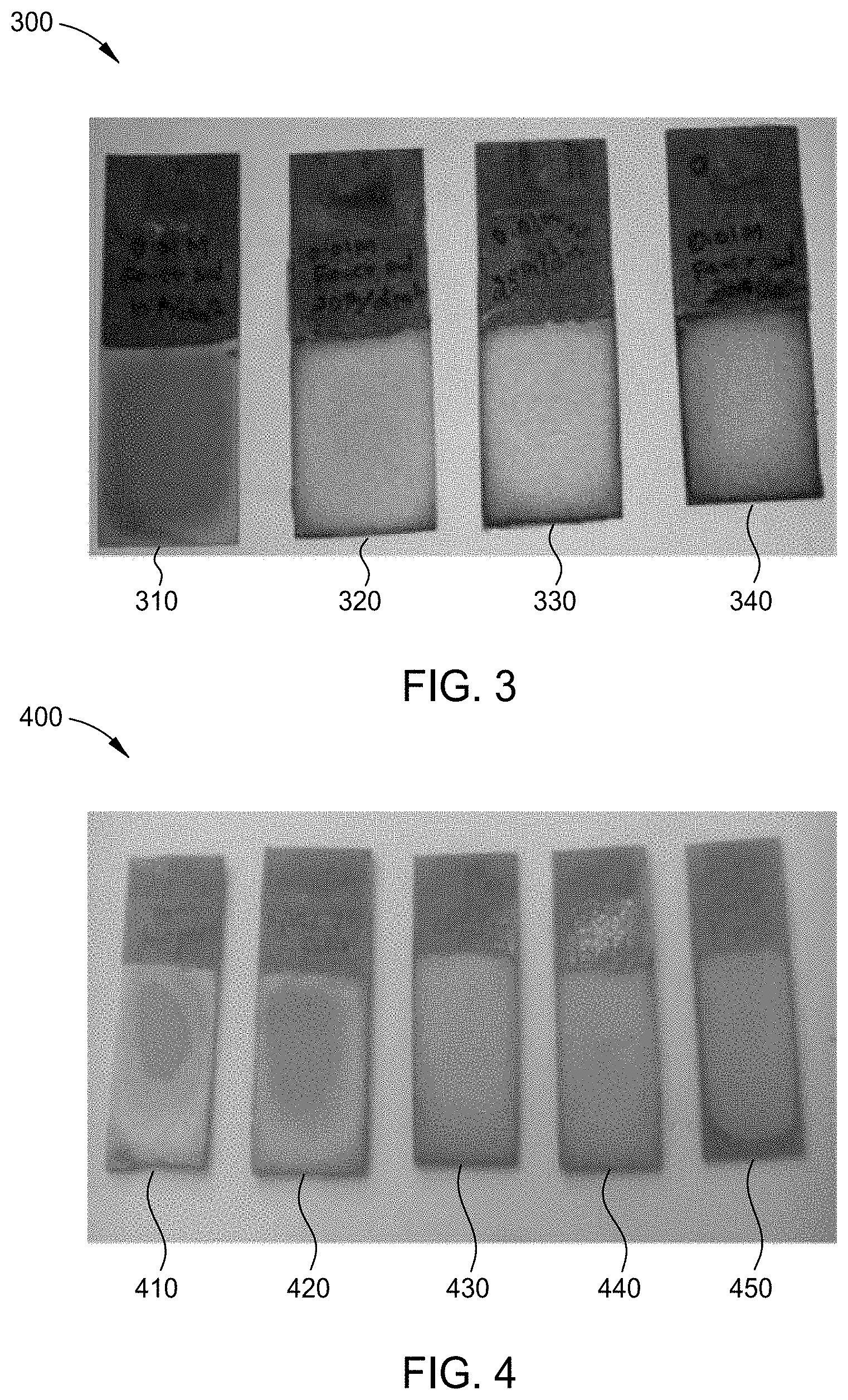
[0126] FIG. 4 is an image 400 of chromium-iron alloy plated substrates 410, 420, 430, 440, and 450 formed by the process of FIG. 2, each plated at a different pH using the electrolyte solution of Example 1. For each chromium-iron alloy plated substrate 410, 420, 430, 440, and 450, the plating parameters were a direct current at a current density of 250 mA/cm.sup.2 at a plating time of approximately 1 hour while maintaining the electrolyte solution at a temperature of 30.degree. C. Chromium-iron alloy plated substrate 410 was plated at a pH of 1.0 resulting in patchy chromium-iron alloy deposits, which were not measured but were believed to be less than 10 .mu.m. Chromium-iron alloy plated substrate 420 was plated at a pH of 2.0 resulting in patchy chromium-iron alloy deposits, which were not measured, but were believed to be less than 10 .mu.m. Chromium-iron alloy plated substrate 430 was plated at a pH of 2.5 resulting in a chromium-iron alloy layer having a thickness of 20 .mu.m. Chromium-iron alloy plated substrate 440 was plated at a pH of 3.0 resulting in a chromium-iron alloy layer having a thickness of 28 .mu.m. Chromium-iron alloy plated substrate 450 was plated at a pH of 3.5 resulting in a chromium-iron alloy layer having a thickness of 45 .mu.m. X-ray fluorescence was performed on a portion of each coating to determine the chromium content and the iron content by wt. %. The results are depicted in Table II.
[0127] As illustrated in Table II, any pH ranging from 2.5 to 3.5 provides deposition of a chromium-iron alloy layer. A pH range of 2.5 to 3.0 advantageously provides a thick chromium-iron alloy layer having a desired iron content. A pH of 3.5 provides a thicker chromium-iron alloy layer with a lower iron content relative to a pH range of 2.5 to 3.0.
TABLE-US-00005 TABLE II Chromium Iron Thickness pH (wt. %) (wt. %) (.mu.m) 1.5 2.7 97.3 <10 2.0 27.6 72.4 <10 2.5 74.1 25.9 20 3.0 85.9 14.1 28 3.5 90.3 9.7 45
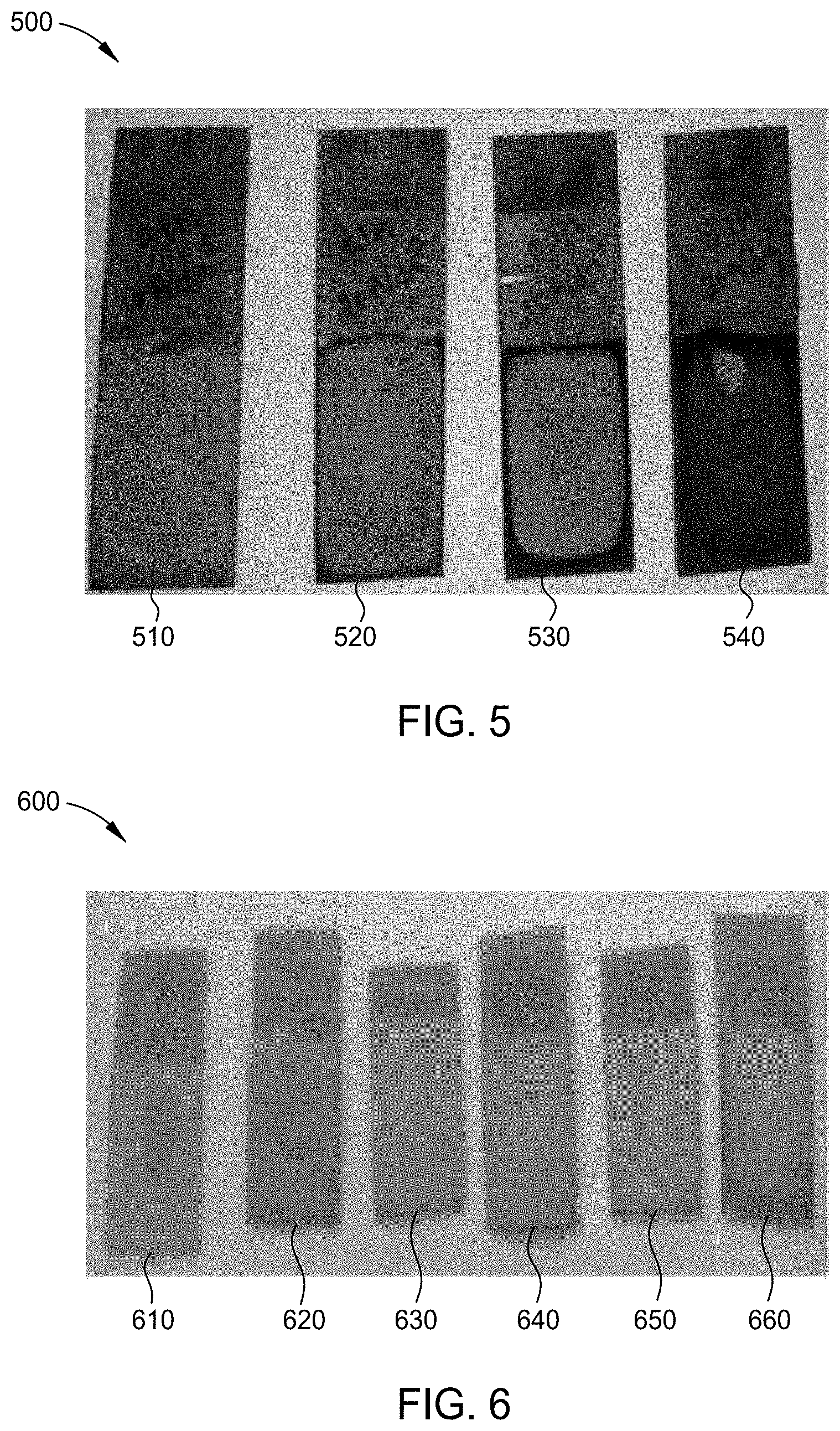
[0128] FIG. 5 is an image 500 of chromium-iron alloy plated substrates 510, 520, 530, and 540 formed by the process of FIG. 2, each plated at a different current density using the electrolyte solution of Example 2. For each chromium-iron alloy plated substrate 510, 520, 530, and 540, the plating parameters were a plating time of approximately 1 hour while maintaining the electrolyte solution at a temperature of 30.degree. C. and a pH of approximately 2.2. Chromium-iron alloy plated substrate 510 was plated at a current density of 100 mA/cm.sup.2 resulting in a chromium-iron alloy layer having a thickness of 30 .mu.m. Chromium-iron alloy plated substrate 520 was plated at a current density of 200 mA/cm.sup.2 resulting in a chromium-iron alloy layer having a thickness of 40 .mu.m. Chromium-iron alloy plated substrate 530 was plated at a current density of 250 mA/cm.sup.2 resulting in a chromium-iron alloy layer having a thickness of 70 .mu.m. Chromium-iron alloy plated substrate 540 was plated at a current density of 300 mA/cm.sup.2 resulting in a chromium-iron layer having a thickness of 50 .mu.m. X-ray fluorescence was performed on a portion of each coating to determine the chromium content and the iron content by wt. %. The results are depicted in Table III.
[0129] As illustrated in Table III, any current density ranging from 100 mA/cm.sup.2 to about 300 mA/cm.sup.2 provides deposition of a chromium-iron alloy. A current density ranging from about 100 mA/cm.sup.2 to about 250 mA/cm.sup.2 provides a thick chromium-iron alloy layer. Further, at a current density of 300 mA/cm.sup.2 the thickness of the chromium-iron alloy decreases relative to the thickness of the chromium-iron alloy deposited at a current density of 250 mA/cm.sup.2.
TABLE-US-00006 TABLE III Current Density Chromium Iron Thickness (mA/cm.sup.2) (wt. %) (wt. %) (.mu.m) 100 27.6 72.4 30 200 52.5 47.5 40 250 60.5 39.5 70 300 59.2 40.8 50
[0130] FIG. 6 is an image 600 of chromium-iron alloy plated substrates 610, 620, 630, 640, 650, and 660 formed by the process of FIG. 2, each plated at a different pH using the electrolyte solution of Example 2. For each chromium-iron alloy plated substrate 610, 620, 630, 640, 650, and 660, the plating parameters were a direct current at a current density of 250 mA/cm.sup.2 at a plating time of approximately 1 hour while maintaining the electrolyte solution at a temperature of 30.degree. C. Chromium-iron alloy plated substrate 610 was plated at a pH of 1.5 resulting in patchy chromium-iron alloy deposit. Chromium-iron alloy plated substrate 620 was plated at a pH of 2.0 resulting in patchy chromium-iron alloy deposits. Chromium-iron alloy plated substrate 630 was plated at a pH of 2.5 resulting in a chromium-iron alloy layer having a thickness of 50 .mu.m. Chromium-iron alloy plated substrate 640 was plated at a pH of 3.0 resulting in a chromium-iron alloy layer having a thickness of 70 .mu.m. Chromium-iron alloy plated substrate 650 was plated at a pH of 3.5 resulting in a chromium-iron alloy layer having a thickness of 50 .mu.m. Chromium-iron alloy plated substrate 660 was plated at a pH of 4.0. X-ray fluorescence was performed on a portion of each coating to determine the chromium content and the iron content by wt. %. The results are depicted in Table IV.
[0131] As illustrated in Table IV, any pH ranging from 1.5 to 4.0 provides deposition of a chromium-iron alloy layer. A pH range of 2.0 to 4.0 advantageously provides a thick chromium-iron alloy layer having a desired high chromium content. A pH of 3.0 provides a thicker chromium-iron alloy layer with good quality deposits. At a pH of 4, the deposits exhibited burning at the edges, while at a pH of 2 or lower, the deposits were patchy.
TABLE-US-00007 TABLE IV Chromium Iron Thickness pH (wt. %) (wt. %) (.mu.m) 1.5 20.9 79.1 Patchy deposit 2.0 34.8 65.2 Patchy deposit 2.5 38.4 61.6 50 3.0 43.8 56.8 70 3.5 49.0 51.0 50 4.0 50.6 49.4 Burning at edges
[0132] FIG. 7 is an image 700 of chromium-iron alloy plated substrates 710, 720, 730, 740, 750, and 760 formed by the process of FIG. 2, each plated at a different current density using the electrolyte solution of Example 3. For each chromium-iron alloy plated substrate 710, 720, 730, 740, 750, and 760, the plating parameters were a plating time of approximately 1 hour while maintaining the electrolyte solution at a temperature of 30.degree. C. and a pH of approximately 2.2. Chromium-iron alloy plated substrate 710 was plated at a current density of 100 mA/cm.sup.2 resulting in patchy chromium-iron alloy deposits believed to be less than 10 .mu.m thick. Chromium-iron alloy plated substrate 720 was plated at a current density of 150 mA/cm.sup.2 resulting in patchy chromium-iron alloy deposits believed to be less than 10 .mu.m thick. Chromium-iron alloy plated substrate 730 was plated at a current density of 200 mA/cm.sup.2 resulting in a chromium-iron alloy layer having a thickness of 10 .mu.m. Chromium-iron alloy plated substrate 740 was plated at a current density of 300 mA/cm.sup.2 resulting in a chromium-iron layer having a thickness of 30 .mu.m. Chromium-iron alloy plated substrate 750 was plated at a current density of 400 mA/cm.sup.2 resulting in a chromium-iron layer having a thickness of 50 .mu.m. Chromium-iron alloy plated substrate 760 was plated at a current density of 500 mA/cm.sup.2 resulting in deposits, which exhibited burning at the edges.
[0133] As illustrated in Table V, any current density ranging from 200 mA/cm.sup.2 to about 400 mA/cm.sup.2 provides deposition of a chromium-iron alloy. A current density ranging from about 300 mA/cm.sup.2 to about 400 mA/cm.sup.2 provides a thick chromium-iron alloy layer. Further at a current density of 500 mA/cm.sup.2 the thickness of the chromium-iron alloy increased relative to the thickness of the chromium-iron alloy deposited at a current density of 400 mA/cm.sup.2, but the deposits at the edges were burned (black powder).
TABLE-US-00008 TABLE V Current Density Thickness (mA/cm.sup.2) (.mu.m) 100 (<10) patchy deposit 150 (<10) patchy deposit 200 10 300 30 400 50 500 (100) burning at edges
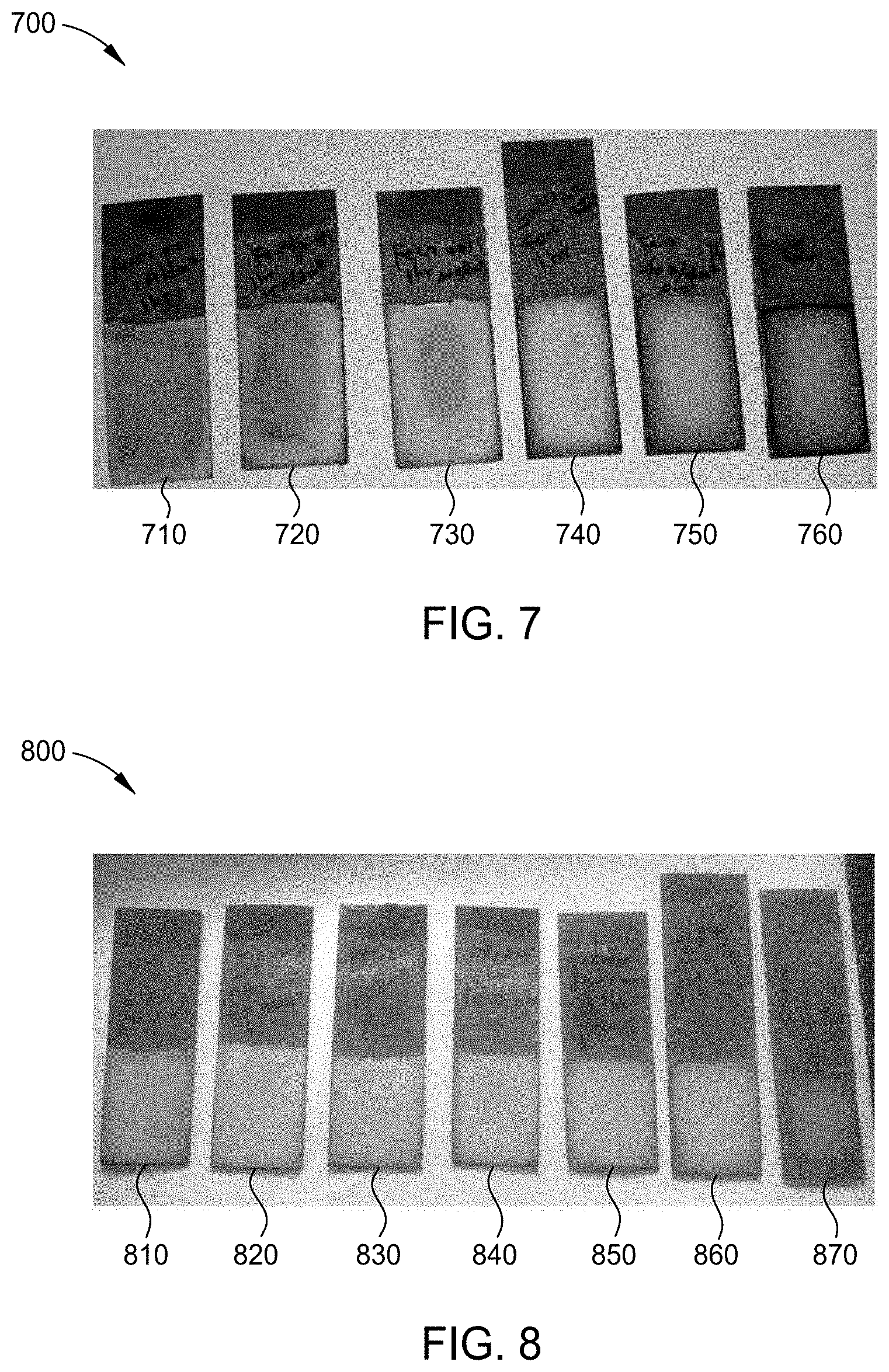
[0134] FIG. 8 is an image 800 of chromium-iron alloy plated substrates 810, 820, 830, 840, 850, 860, and 870 formed by the process of FIG. 2, each plated at a different pH using the electrolyte solution of Example 3. For each chromium-iron alloy plated substrate 810, 820, 830, 840, 850, 860, and 870, the plating parameters were a direct current at a current density of 250 mA/cm.sup.2 at a plating time of approximately 1 hour while maintaining the electrolyte solution at a temperature of approximately 30.degree. C. Chromium-iron alloy plated substrate 810 was plated at a pH of 1.0 resulting in a chromium-iron alloy layer having a thickness of 20 .mu.m. Chromium-iron alloy plated substrate 820 was plated at a pH of 1.5 resulting in a chromium-iron alloy layer having a thickness of 24 .mu.m. Chromium-iron alloy plated substrate 830 was plated at a pH of 2.0 resulting in a chromium-iron alloy layer having a thickness of 20 .mu.m. Chromium-iron alloy plated substrate 840 was plated at a pH of 3.0 resulting in a chromium-iron alloy layer having a thickness of 16 .mu.m. Chromium-iron alloy plated substrate 860 was plated at a pH of 3.5 resulting in a chromium-iron alloy layer having a thickness of 16 .mu.m. Chromium-iron alloy plated substrate 870 was plated at a pH of 4.0 resulting in patchy chromium-iron alloy deposits of low thickness. X-ray fluorescence was performed on a portion of coatings deposited at a pH of 2 and a pH of 2.5 to determine the chromium content and the iron content by wt. %. The results are depicted in Table VI.
[0135] As illustrated in Table VI, any pH ranging from 1.0 to 4.0 provides deposition of a chromium-iron alloy. A pH ranging from 1.0 to 2.5 advantageously provides a thicker chromium-iron alloy layer than a pH that is higher. Further, a pH ranging from 1.5 to 2.0 provides the thickest chromium-iron alloy layer.
TABLE-US-00009 TABLE VI Chromium Iron Thickness pH (wt. %) (wt. %) (.mu.m) 1.0 20 1.5 24 2.0 45 55 24 2.5 46 54 20 3.0 16 3.5 16 4.0 patchy deposits
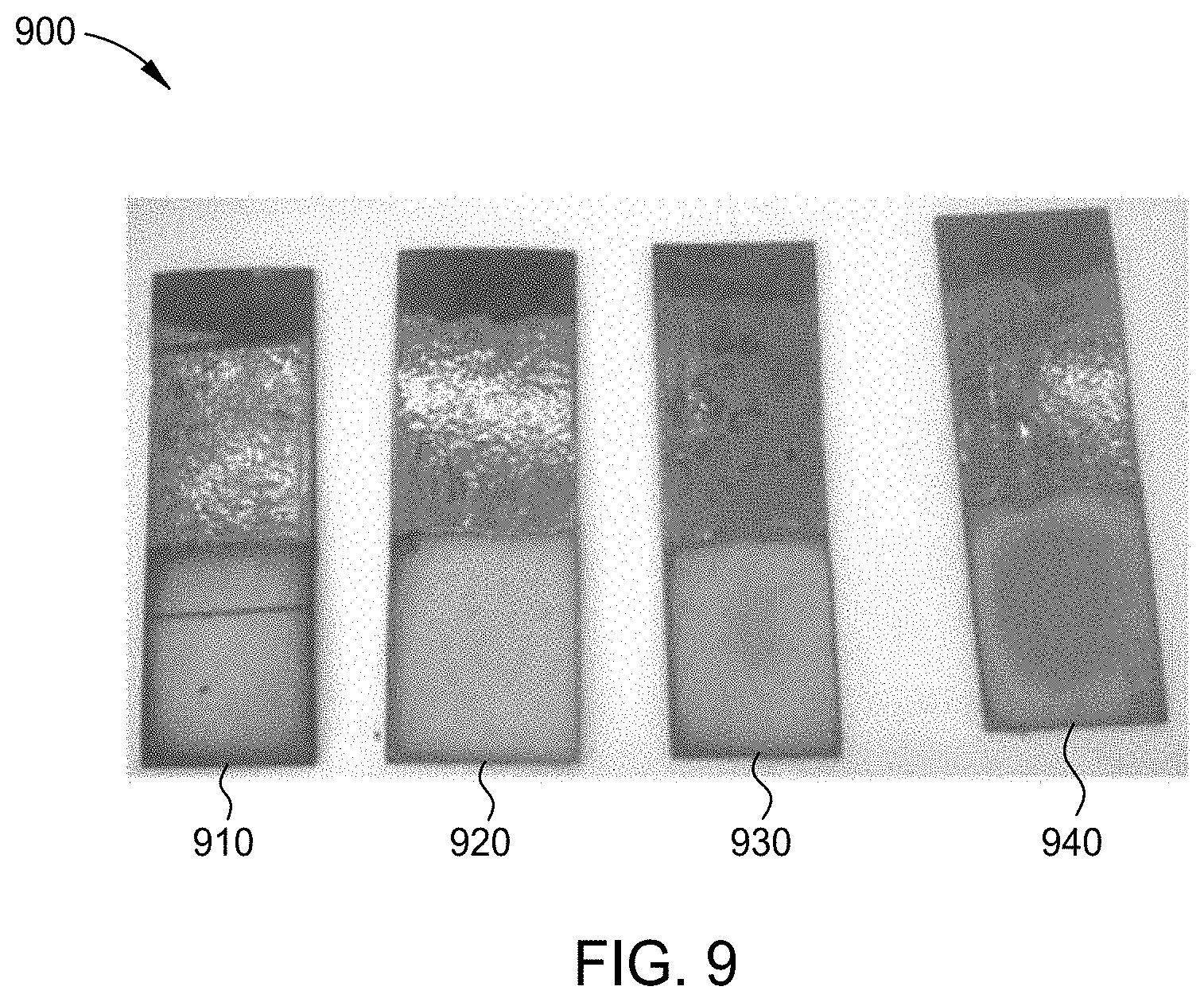
[0136] FIG. 9 is an image 900 of chromium-iron alloy plated substrates 910, 920, 930, and 940 formed by the process of FIG. 2, each plated at a different temperature using the electrolyte solution of Example 3. For each chromium-iron alloy plated substrate 910, 920, 930, and 940, the plating parameters were a direct current at a current density of 250 mA/cm.sup.2 at a plating time of approximately 1 hour while maintaining the electrolyte solution at a pH of approximately 2.1. Chromium-iron alloy plated substrate 910 was plated at a temperature of 30 .degree. C. resulting in a chromium-iron alloy layer having a thickness of 40 .mu.m. Chromium-iron alloy plated substrate 920 was plated at a temperature of 40.degree. C. resulting in a chromium-iron alloy layer having a thickness of 30 .mu.m. Chromium-iron alloy plated substrate 930 was plated at a temperature of 50.degree. C. resulting in a chromium-iron alloy layer having a thickness of 30 .mu.m. Chromium-iron alloy plated substrate 940 was plated at a temperature of 60.degree. C. resulting in a chromium-iron alloy layer having a low thickness. The results are depicted in Table VII.
[0137] As illustrated by Table VII, any temperature ranging from about 30.degree. C. to about 50.degree. C. provides deposition of a chromium-iron alloy layer. The operating temperature of 30.degree. C. provides the thickest chromium-iron alloy layer, but the deposits at 40.degree. C. were aesthetically better.
TABLE-US-00010 TABLE VII Temperature Chromium Iron Thickness (.degree. C.) (wt. %) (wt. %) (.mu.m) 30 40 60 40 40 41 59 30 50 43 57 30 60 45 55 14
[0138] Using the electrolyte solution of Example 3, chromium-iron-alloy coatings were deposited on a Taber Wear panels by a direct current at a current density of 300 mA/cm.sup.2 for a plating time of approximately 2 hours while maintaining the electrolyte solution at a temperature of approximately 40.degree. C. and a pH of approximately 2.5. The alloy composition of the coating as measured by SEM-EDS was chromium at .about.41 wt. %, iron at .about.10 wt. %, and carbon at .about.49 wt. %. Hardness values were measured on `as plated` coupons as well as after baking the coupons at approximately 190.degree. C. for 23 hours. The obtained chromium-iron-alloy coatings were evaluated as to wear resistance. Measurement of wear resistance was performed using a Taber wear test specified according to ASTM D 4060. The abrasive wheels used were CS10, with a load of 500 g per wheel, abrasion was performed for 10,000 cycles, at a rotary speed of 60 rpm. The Taber wear index was less than 10. The average of 5 hardness measurements for the "as plated" chromium-iron alloy layer was 1056 HV. The average of 5 hardness measurements for the `baked` chromium-iron alloy layer was 1187 HV.
[0139] Using the electrolyte solution of Example 1, chromium-iron-alloy coatings were deposited on steel substrates by a direct current at a current density of 250 mA/cm.sup.2 for a plating time of approximately 2 hours while maintaining the electrolyte solution at a temperature of approximately 35.degree. C. and a pH of approximately 2.5. The alloy composition of the coating as measure by SEM-EDS was chromium at .about.80 wt. %, iron at .about.12 wt. %, and carbon at .about.8 wt. %. Hardness values were measured on `as plated` coupons as well as after baking the coupons at approximately 190.degree. C. for 23 hours. The average of 5 hardness measurements for the "as plated" chromium-iron alloy layer was 1147 HV. The average of 5 hardness measurements for the `baked` chromium-iron alloy layer was 1249 HV.
[0140] Overall, the present disclosure provides improved electrolyte solutions for electrodeposition of chromium-iron alloys, methods of forming chromium-iron alloys, and methods of electrodepositing chromium-iron alloys.
[0141] The descriptions of the various aspects of the present disclosure have been presented for purposes of illustration, but are not intended to be exhaustive or limited to the aspects disclosed. Many modifications and variations will be apparent to those of ordinary skill in the art without departing from the scope and spirit of the described aspects. The terminology used herein was chosen to best explain the principles of the aspects, the practical application or technical improvement over technologies found in the marketplace, or to enable others of ordinary skill in the art to understand the aspects disclosed herein. While the foregoing is directed to aspects of the present disclosure, other and further aspects of the present disclosure can be devised without departing from the basic scope thereof.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.