Characteristics Of Tunable Adsorbents For Rate Selective Separation Of Nitrogen From Methane
Stuckert; Nicholas R. ; et al.
U.S. patent application number 17/034748 was filed with the patent office on 2021-01-21 for characteristics of tunable adsorbents for rate selective separation of nitrogen from methane. The applicant listed for this patent is Philip A. Barrett, Steven J. Pontonio, Neil A. Stephenson, Nicholas R. Stuckert. Invention is credited to Philip A. Barrett, Steven J. Pontonio, Neil A. Stephenson, Nicholas R. Stuckert.
| Application Number | 20210016218 17/034748 |
| Document ID | / |
| Family ID | 1000005167461 |
| Filed Date | 2021-01-21 |



| United States Patent Application | 20210016218 |
| Kind Code | A1 |
| Stuckert; Nicholas R. ; et al. | January 21, 2021 |
CHARACTERISTICS OF TUNABLE ADSORBENTS FOR RATE SELECTIVE SEPARATION OF NITROGEN FROM METHANE
Abstract
The present invention generally relates to a process that utilizes tunable zeolite adsorbents in order to reduce the bed size for nitrogen removal from a methane (or a larger molecule) containing stream. The adsorbents are characterized by the rate of adsorption of nitrogen and methane and the result is a bed size that is up to an order of magnitude smaller with these characteristics (in which the rate selectivity is generally 30) than the corresponding bed size for the original tunable zeolite adsorbent that has a rate selectivity of >100x.
| Inventors: | Stuckert; Nicholas R.; (Grand Island, NY) ; Pontonio; Steven J.; (Eden, NY) ; Stephenson; Neil A.; (E. Amherst, NY) ; Barrett; Philip A.; (Tonawanda, NY) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005167461 | ||||||||||
| Appl. No.: | 17/034748 | ||||||||||
| Filed: | September 28, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| PCT/US2019/024581 | Mar 28, 2019 | |||
| 17034748 | ||||
| 62649798 | Mar 29, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | B01D 53/047 20130101; B01J 20/186 20130101; B01D 2253/108 20130101; B01D 2257/102 20130101; B01D 2256/245 20130101 |
| International Class: | B01D 53/047 20060101 B01D053/047; B01J 20/18 20060101 B01J020/18 |
Claims
1. A pressure swing adsorption process for kinetic separation of N.sub.2 from a feed stream comprising at least methane and N.sub.2, said process comprising feeding the feed stream to an adsorbent bed comprising adsorbent having: a rate of adsorption of at least 0.036 mmol/g/min for N.sub.2 as determined by the Hiden method and a rate of adsorption of methane that is 1/6.sup.th to 1/10000.sup.th the adsorbent's adsorption rate for N.sub.2 as determined by the Hiden method and recovering a product stream containing said at least methane gas with a reduced level of N.sub.2.
2. The process of claim 1 wherein the adsorbent has an adsorption rate of at least 0.143 mmol/g/min for N.sub.2 impurity as determined by the Hiden method and an adsorption rate for methane that is 1/10.sup.th to 1/1000.sup.th of the adsorption rate for N.sub.2 as determined by the Hiden method.
3. The process of claim 1 wherein said adsorbent comprises zeolite A, X, Y, chabazite, mordenite, faujasite, clinoptilolite, ZSM-5, L, Beta, or combinations thereof.
4. The process of claim 3 wherein said adsorbent is a zeolite exchanged with at least one cation selected from Li, Na, K, Mg, Ca, Sr, Ba, Cu, Ag, Zn, NH4+ and combinations or mixtures thereof
5. The process of claim 1 wherein said adsorbent is zeolite A.
6. The process according to claim 2 where the feed stream that may contain additional gas species such as ethane, propane, butane and hydrocarbons with more than 4 carbon atoms and may include adsorbents to remove said hydrocarbons.
7. A process according to claim 2 where the feed stream that may contain additional gas species such as water, carbon dioxide or sulfur species and may include adsorbents to remove said species.
8. An adsorbent for the kinetic separation of N2 impurity from a feed stream comprising at least methane and nitrogen gas, said process comprising feeding the feed stream to an adsorbent bed comprising an adsorbent having: a rate of adsorption of at least 0.036 mmol/g/min for N.sub.2 as determined by the Hiden method, and a rate of adsorption for methane that is 1/6.sup.th or less than the adsorbent's adsorption rate for N.sub.2 as determined by the Hiden method.
9. The adsorbent of claim 8 wherein the adsorbent has an adsorption rate of at least 0.143 mmol/g/min for said N.sub.2 as determined by the TGA method and an adsorption rate for the methane that is 1/10.sup.th or less of the adsorbent's adsorption rate for N.sub.2 as determined by the Hiden method.
10. The adsorbent of claim 8 which comprises zeolite A, X, Y, chabazite, mordenite, faujasite, clinoptilolite, ZSM-5, L, Beta, or combinations thereof
11. The adsorbent of claim 10 wherein said adsorbent is a zeolite is exchanged with at least one cation selected from Li, Na, K, Mg, Ca, Sr, Ba, Cu, Ag, Zn, NH4+ and combinations or mixtures thereof.
12. The adsorbent of claim 10 wherein said adsorbent is zeolite A.
Description
RELATED APPLICATIONS
[0001] This is a continuation-in-part application of and claims benefit of International Application No. PCT/US2019/024581, filed on Mar. 28, 2019, which claimed the benefit of U.S. Provisional Application Ser. No. 62/649,798, filed on Mar. 29, 2018, both of which are incorporated herein by reference.
FIELD OF THE INVENTION
[0002] The present invention generally relates to adsorbent characteristics used in a process to separate nitrogen from methane.
BACKGROUND OF THE INVENTION
[0003] Since nitrogen adsorption from methane is a relatively unexplored area, it is important to draw the background from similar adsorption processes such as pressure swing adsorption (PSA), vacuum swing adsorption (VSA) and vacuum pressure swing (VPSA) which have been commercially utilized for bulk air separation, as well as trace air contaminant removal, for many decades. In PSA and VPSA processes, compressed air is pumped through a fixed bed of an adsorbent exhibiting an adsorptive preference for one of the main constituents, typically Na in bulk air separation, CO.sub.2 and H.sub.2O in air prepurification, or CO and CO.sub.2 in H.sub.2 purification, etc., whereby an effluent product stream enriched in the lesser-adsorbed constituent is obtained. Improvements in these processes remain important goals, one principal means of which is the discovery and development of better process cycles. Significant improvements have been achieved in not only recovery of gas but also reductions in overall system size. These improvements also continue to provide important benefits even while the adsorbent being used in conjunction with the system is constantly improved and replaced with better alternatives.
[0004] A large majority of processes operate through the equilibrium adsorption of the gas mixture and kinetic separations have lately attracted considerable attention with the development of functional microporous adsorbents and efficient modeling tools. Still, relatively few steric separation processes have been commercialized. Kinetically based separation involves differences in the diffusion rates of different components of the gas mixture and allows different molecular species to be separated regardless of similar equilibrium adsorption parameters. Kinetic separations utilize adsorbents like carbon molecular sieves since they exhibit a distribution of pore sizes which allow the different gaseous species to diffuse into the adsorbent at different rates while avoiding exclusion of any component of the mixture. Kinetic separations can be used for the separation of industrial gases, for example, for the separation of nitrogen from air and argon from other gases. In the case of the nitrogen/oxygen separation (for example, oxygen and nitrogen differ in size by only 0.02 nm), the separation is efficient since the rate of transport of oxygen into the carbon sieve pore structure is markedly higher than that of nitrogen. Hence, the kinetic separation works, even though the equilibrium loading levels of oxygen and nitrogen are virtually identical.
[0005] Kinetically based separation processes may be operated, as noted in U.S. Patent Application Publication No. 2008/0282884, as pressure swing adsorption (PSA), temperature swing adsorption (TSA), partial pressure swing or displacement purge adsorption (PPSA) or as hybrid processes comprised of components of several of these processes. These swing adsorption processes can be conducted with rapid cycles, in which case they are referred to as rapid cycle thermal swing adsorption (RCTSA), rapid cycle pressure swing adsorption (RCPSA), and rapid cycle partial pressure swing or displacement purge adsorption (RCPPSA) technologies, with the term "swing adsorption" taken to include all of these processes and combinations of them.
[0006] The faster the beds perform the steps required to complete a cycle, the smaller the beds can be when used to process a given hourly feed gas flow. Several other approaches to reducing cycle time in PSA processes have emerged which use rotary valve technologies (U.S. Pat. Nos. 4,801,308; 4,816,121; 4,968,329; 5,082,473; 5,256,172; 6,051,050; 6,063,161; 6,406,523; 6,629,525; 6,651,658; and 6,691,702). A parallel channel (or parallel passage) contactor with a structured adsorbent may be used to allow for efficient mass transfer in these rapid cycle pressure swing adsorption processes. Approaches to constructing parallel passage contactors with structured adsorbents are known (U.S. Patent Application Publication No. 2008/0282892). These demonstrate the benefit of having high rates of adsorption of the contaminant in equilibrium processes and provide the basis for why increasing rates of adsorption helps to intensify the process.
[0007] In the case of kinetic-controlled PSA processes, the adsorption and desorption are more typically caused by cyclic pressure variation, whereas in the case of TSA, PPSA and hybrid processes, adsorption and desorption may be caused by cyclic variations in temperature, partial pressure, or combinations of pressure, temperature and partial pressure, respectively. In the exemplary case of PSA, kinetic- controlled selectivity may be determined primarily by micropore mass transfer resistance (e.g., diffusion within adsorbent particles or crystals) and/or by surface resistance (e.g., narrowed micropore entrances). For successful operation of the process, a relatively and usefully large working uptake (e.g., the amount adsorbed and desorbed during each cycle) of the first component and a relatively small working uptake of the second component may preferably be achieved. Hence, the kinetic- controlled PSA process requires operation at a suitable cyclic frequency, balancing the avoidance of excessively high cycle frequency where the first component cannot achieve a useful working uptake with excessively low frequency where both components approach equilibrium adsorption values.
[0008] Some established kinetic-controlled PSA processes use carbon molecular sieve adsorbents, e.g., for air separation with oxygen comprising the first more- adsorbed component and nitrogen the second less adsorbed component. Another example of kinetic-controlled PSA is the separation of nitrogen as the first component from methane as the second component. Those may be performed over carbon molecular sieve adsorbents or more recently employing a hybrid kinetic/equilibrium PSA separation (principally kinetically based but requiring thermal regeneration periodically due to partial equilibrium adsorption of methane on the adsorbent material) over titanosilicate based adsorbents such as ETS-4 (U.S. Pat. Nos. 6,197,092 and 6,315,817). Thermal regeneration is described as the method of passing heated gas across the adsorbent bed in order to cause desorption of the methane. In order to minimize the time required for thermal regeneration, slow rates of methane uptake are chosen, which also correspond to the primary benefit of the ETS-4 which is disclosed as high rate selectivity, exceeding 100.times. the nitrogen uptake rate over methane as the primary benefit of these adsorbents. The relatively slow rate of uptake for nitrogen compared to an equilibrium process is seen as unavoidable for rate selective processes, in order to maintain high recovery. As a result, the bed sizes to process the gas are relatively large compared to equilibrium processes.
[0009] Another patent utilizing molecular sieves for the removal of nitrogen from natural gas is U.S. Pat. No. 4,964,889 which discloses the use of a clinoptilolites zeolite containing magnesium cations for the removal of nitrogen. The authors again teach the primary benefit of the zeolites is high rate selectivity, exceeding 100.times. the nitrogen uptake rate over methane as the primary benefit of these adsorbents. Again, the slow rate of uptake of nitrogen is seen as necessary and unavoidable, in order to have high recovery of methane and also to prevent methane poisoning. Again, as a result the bed sizes to process the gas are relatively large compared to equilibrium processes.
SUMMARY OF THE INVENTION
[0010] The present invention generally relates to adsorbent characteristics used in a process to separate nitrogen from methane. More specifically, the present invention relates to a process that utilizes tunable zeolite adsorbents in order to reduce the bed size for nitrogen removal from a methane (or a larger molecule) containing stream. The adsorbents are characterized by the rate of adsorption of nitrogen and methane and the result is a bed size that is up to an order of magnitude smaller with these characteristics (in which the rate selectivity is generally 30) than the corresponding bed size for the original tunable zeolite adsorbent that has a rate selectivity of >100.times..
DETASILED DESCRIPTION OF THE FIGURES
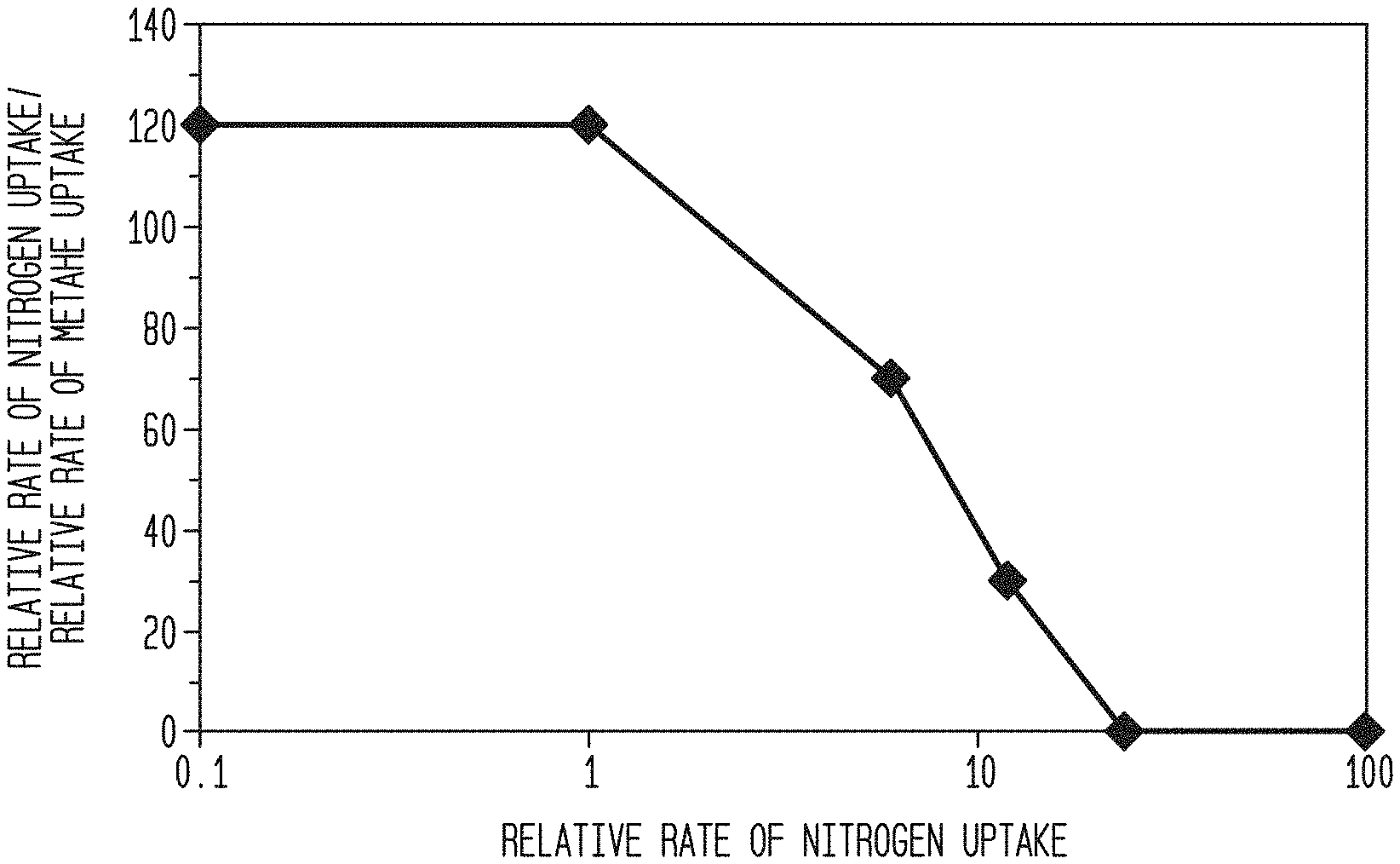
[0011] FIG. 1. Shows the rate selectivity dependence of modified 4A on changing uptake rate of nitrogen.
[0012] FIG. 2. Outlines the TGA method sequence to measure the rates of adsorption of nitrogen and methane.
[0013] FIG. 3. Shows an example of a TGA plot that is obtained following the method outlined in FIG. 2.
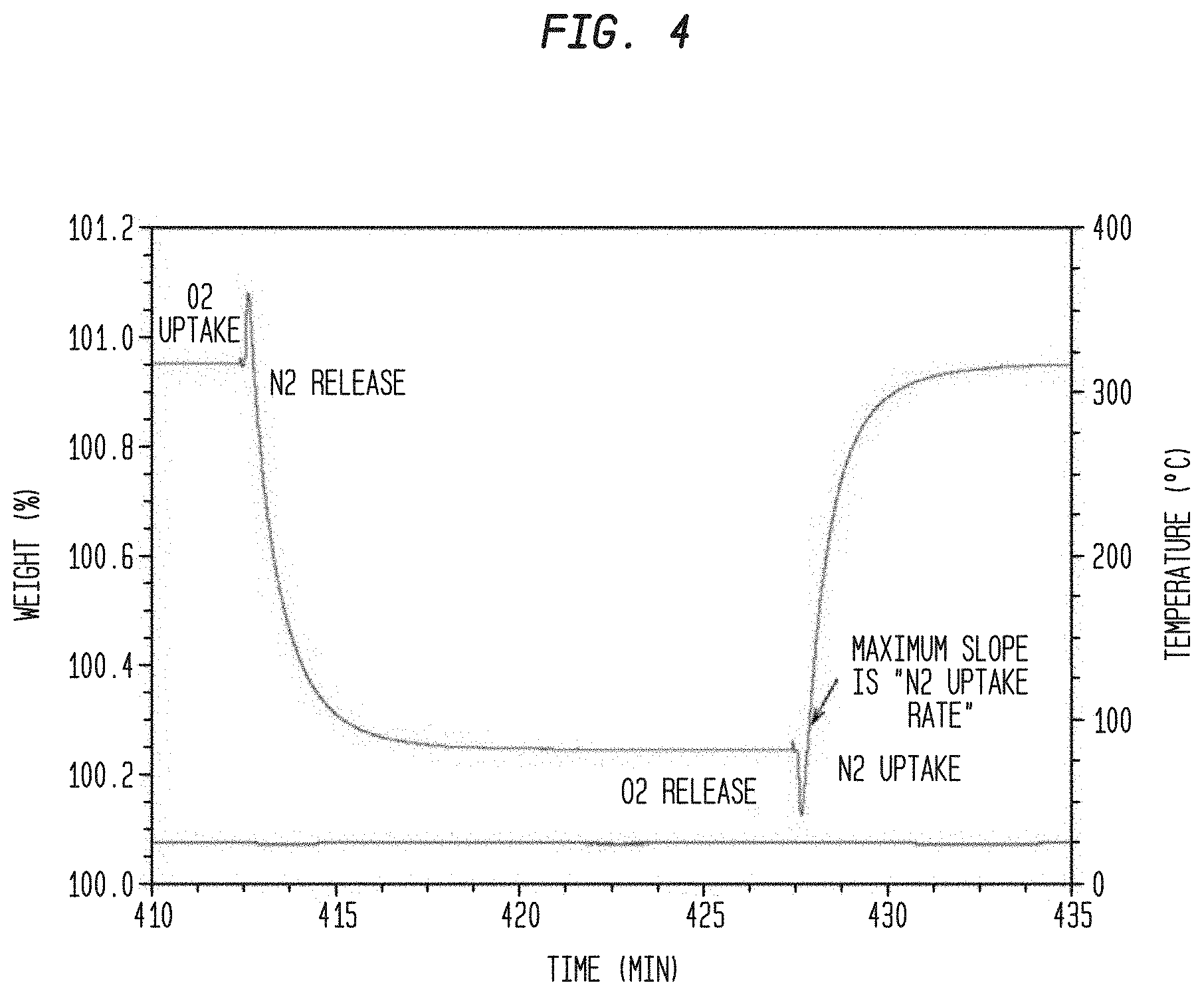
[0014] FIG. 4. Shows an expansion of the same plot in FIG. 3 to illustrate the features observed during gas switching.
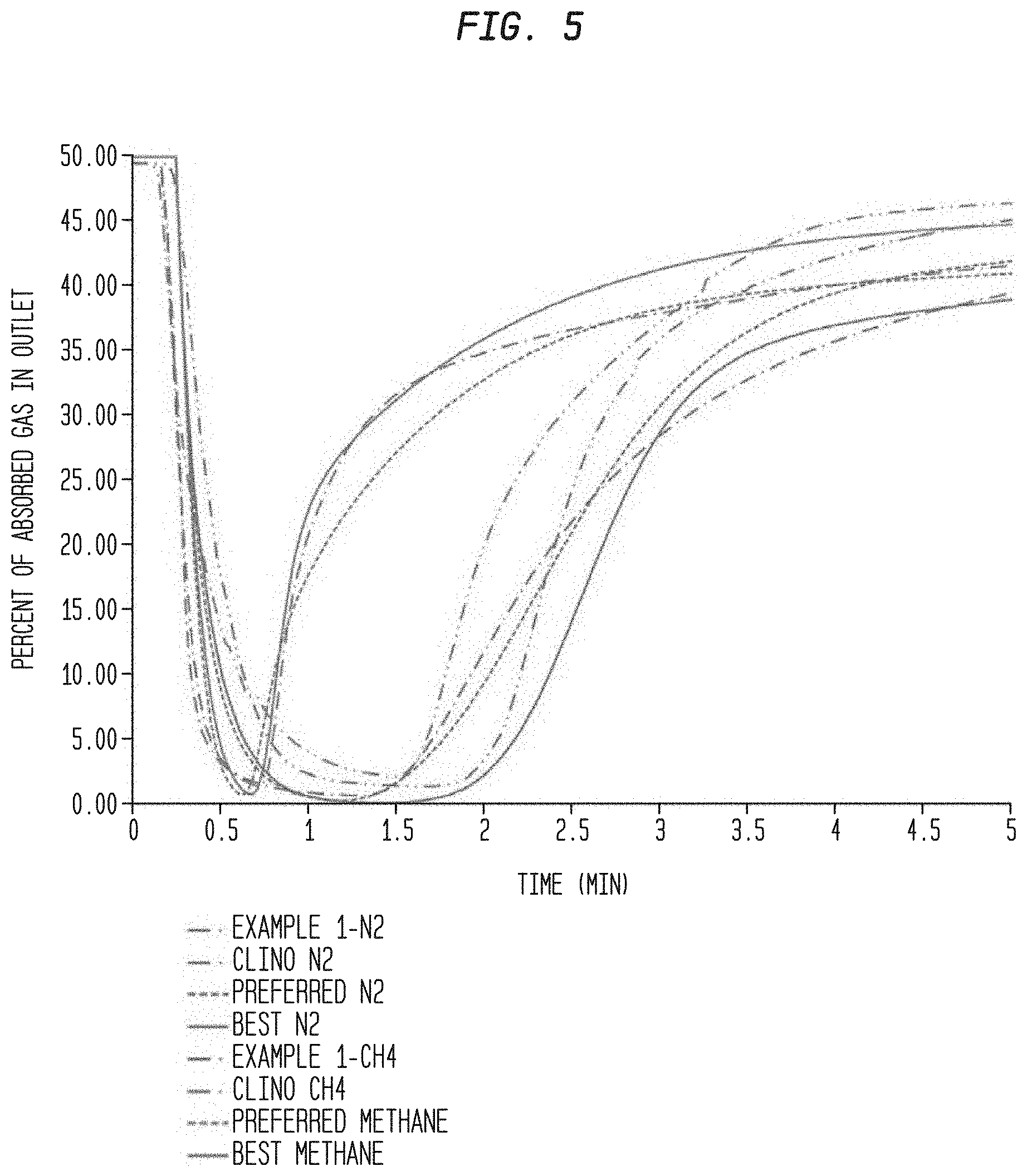
[0015] FIG. 5. A breakthrough experiment diagram showing the characterization of tunable zeolite 4A and clinoptilolite TSM-140 as compared to the minimum characteristics described herein and the ideal characteristics for this invention.
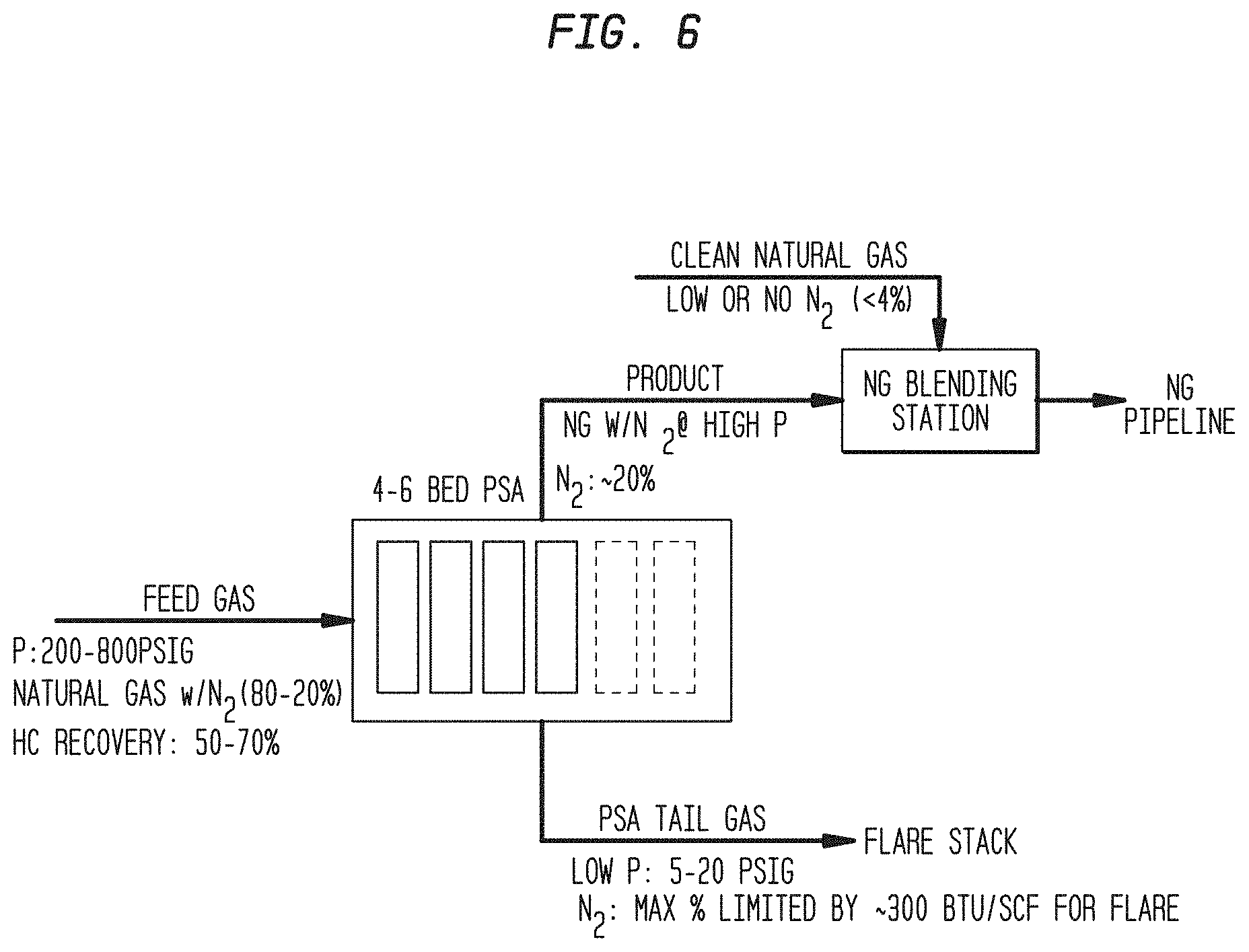
[0016] FIG. 6. A diagram showing a typical application of this system to a natural gas well head feed stream, post hydraulic fracturing.
DETAILED DESCRIPTION OF THE INVENTION
[0017] Separating nitrogen and methane has historically presented a challenge. While carbon-based adsorbents are readily available to adsorb methane from nitrogen, this leaves the methane at ambient pressure while the nitrogen is produced near the feed pressure. Typically, the methane is required at the feed pressure and the nitrogen at ambient pressure. It is then preferred to adsorb the nitrogen. The present invention generally relates to adsorbent characteristics used in a process to separate nitrogen from methane.
[0018] The adsorbents of the invention are characterized by the rate of adsorption of nitrogen and methane. These material characteristics are used in a pressure swing adsorption (PSA) process, in order to adsorb the nitrogen and allow the methane to pass through the adsorption bed at or around the feed pressure. Demonstration of effect and benefit is shown in the examples of modeling, bench characterization and pilot testing.
[0019] In order to intensify the process and reduce the bed sizes of the adsorption system, an adsorbent having an increased rate of adsorption of nitrogen was developed. An examination of the role of the uptake rate of nitrogen, the selectivity that was correspondingly displayed in the material compared to the uptake rate of methane, and the final product purity desired, demonstrated that the long-held wisdom of higher selectivity was incorrect. What is best is a high uptake rate of the contaminant (nitrogen in this case), and even a moderate selectivity of around 6x is sufficient to provide similar performance to state of the art materials that have a selectivity generally around 100.times..
[0020] Previous adsorbent applications describe characteristics of adsorbents that are favorable or required for a separation or to improve a separation. Previous patents have described materials that are favorable for kinetic-controlled purification of gases or processes that are favorable for kinetic-controlled separations. In U.S. Pat. No. 6,315,817 as an example, a specification for which variant of ETS-4 or the characteristics required is missing, and the majority of ETS-4 products that can be made to fit the description do not work. Specifically, barium exchanged ETS-4 is commercially available for this separation; however the moisture content is noted to be critical to performance. No characteristics exist for rate selective adsorption of nitrogen from natural gas (methane). The only characterization is that the benefit of the material is the high rate selectivity (up to and exceeding 100.times.) of the rate of uptake of nitrogen over methane. The result is very large bed sizes that have come to define the entire area of rate selective adsorbent processes.
[0021] This present invention defines tunable adsorbent characteristics and a process that allows one to reduce the bed sizes for nitrogen removal from a methane (or a larger molecule) containing stream. The result is a bed size that is up to an order of magnitude smaller with these characteristics (in which the rate selectivity is generally 30) than the corresponding bed size for the original tunable zeolite adsorbent that has a rate selectivity of >100.times..
[0022] In one embodiment adsorbents having crystalline inorganic frameworks can be utilized in accordance with the present invention. Crystalline inorganic adsorbents are defined as any microporous aluminosilicate having a regular arrangement of atoms in a space lattice. Zeolites are a preferred crystalline inorganic framework. Zeolites are porous crystalline aluminosilicates which comprise assemblies of SiO.sub.4 and AlO.sub.4 tetrahedra joined together through sharing of oxygen atoms. The general stoichiometric unit cell formula for a zeolite framework is:
M.sub.x/m(AlO.sub.2)x(SiO.sub.2)zH.sub.2O
where M is the cation with a valence of m, z is the number of water molecules in each unit cell, and x and y are integers such that y/x is greater than or equal to 1. The ratio of oxygen atoms to combined aluminum and silicon atoms is equal to 2. Therefore, each aluminum atom introduces a negative charge of one (-1) on the zeolite framework which is balanced by that of a cation. To activate the zeolite the water molecules are completely or substantially removed by raising the temperature or pulling vacuum. This results in a framework with the remaining atoms intact producing cavities connected by channels or pores. The channel size is determined by the number of atoms which form the apertures leading to the cavities as well as cation type and position. Changing the position and type of the cation allows one to change and fine tune channel size and the properties of the zeolite, including its selectivity. For instance, the sodium form of Zeolite A has a pore size of .about.4.ANG. and is called a 4A molecular sieve. If at least 40% of the sodium ions are exchanged with a larger potassium ion, the pore size is reduced to .about.3.ANG.. If these are exchanged with >70% calcium, one calcium ion replaces two sodium ions and the pore opening is increased to .about.5.ANG.. The ability to adjust pores to precisely determine uniform openings allows for molecules smaller than its pore diameter to be adsorbed while excluding larger molecules. The Si/Al ratio can also be varied to modify the framework structure and provide selectivity required for a given separation. This is why zeolites, known as molecular sieves, are very effective in separating on the basis of size.
[0023] Some non-limiting examples of zeolites that can be employed in the context of the invention include zeolite A, X, Y, chabazite, mordenite, faujasite, clinoptilolite ZSM-5, L, Beta, or combinations thereof. The above zeolites can be exchanged with cations including Li, Na, K, Mg, Ca, Sr, Ba, Cu, Ag, Zn, NH4+ and mixtures thereof. In one embodiment zeolite 4A is a preferred adsorbent.
[0024] The adsorbents are characterized by the rate of adsorption of nitrogen and methane. These material characteristics are used in a pressure swing adsorption (PSA) process, in order to adsorb the nitrogen and allow the methane to pass through the adsorption bed at or around the feed pressure. Demonstration of effect and benefit is shown in the examples of modeling, bench characterization and pilot testing. Previous disclosures on cycles do not define the rate characteristics required for the cycle to work. This is an important consideration for rate selective materials as the majority of materials fail to deliver sufficient separation under any process conditions even while providing a substantial difference in adsorption rates. When rate selectivities are disclosed for materials for this separation, they are generally 100.times. or higher.
[0025] Surprisingly, it has been found that lowering rate selectivity of the adsorbent allows one to reduce the bed size required to process a specific feed stream thereby lowering cost performance. It has also been noted that a higher rate selectivity generally corresponds to a lower uptake rate of nitrogen. When attempting to shrink the apparent pore size of an adsorbent, the decreasing rate of the larger molecule occurs much faster than the smaller molecule. However, the decreasing rate of uptake of nitrogen decreases the productivity of the adsorbent during a fixed period of time. Thus, it is important to find a balance between the two instead. One might think that increasing the rate of uptake of the larger molecule would cause the adsorbent to saturate with the larger molecule. In typical rate selective separations however, steady state adsorption is achieved relatively quickly in a matter of minutes to hours to a few days (N.sub.2PSA, or carbon molecular sieves for example). In a scenario where it is desired to decrease the rate to the point that the larger molecule did not reach steady state during operation, the rate of uptake of nitrogen would have to be slowed substantially such that the adsorbent productivity would suffer even more and would not be practically useful. Another alternative has been to employ periodic thermal regeneration of the adsorbent, however even in this case the productivity of the adsorbent suffers significantly. The essentially finding here is that when two overlapping distributions of gas sizes are present, in order to find the optimal rate, one must take into account the rate of adsorption of the contaminant, rather than seek only higher selectivity.
[0026] In accordance with the present invention it was discovered that contaminant rate selective adsorbent must have a rate of contaminant uptake at least 6.times. greater than the product, and ideally >20.times. product, as characterized by the rate measurement from a gravimetric pressure microbalance such as a Hiden IGA unit i.e., it is not required to have a rate selectivity of greater than 100.times. the product as taught by the state-of-the-art in the field.
[0027] According to the invention, the adsorbent is characterized by the following characteristics: [0028] 1. The rate of uptake of nitrogen is greater than 0.1 wt %/min (0.036 mmol/g/min), in another embodiment greater than 0.4wt % min determined by the gravimetric method going from vacuum to 1 Bar pressure of >99.9% Nitrogen (less than 0.1 ppm H.sub.2O) at 35.degree. C. for nitrogen measurements and from vacuum to 1 Bar pressure of >99.9% Methane (less than 0.1 ppm H.sub.2O) at 35.degree. C. for methane measurements, these targets may vary up to 50% for differing pressures and desired product compositions, and these numbers are based on a feed gas stream of 600 psig. [0029] 2. The rate of methane uptake, as characterized by a pressure microbalance gravimetric system such the Hiden IGA, must be less than 1/6.sup.th the uptake rate of nitrogen at 1 atm >99.9% CH4, in another embodiment less than 1/10.sup.th at 1 atm >99.9% CH.sub.4. The rate of methane uptake should obviously not be zero and should be greater than 1/10000.sup.th, in another embodiment greater than 1/1000.sup.th the uptake rate of nitrogen at 1 atm >99.9% CH.sub.4. In one embodiment the rate of methane uptake, as characterized by the Hiden unit, is less than 1/6.sup.th but greater than 1/10000.sup.th the uptake rate of nitrogen at 1 atm >99.9% CH.sub.4, in another embodiment less than 1/10.sup.th but greater than 1/1000.sup.th at 1 atm >99.9% CH.sub.4.
[0030] In additional embodiments the adsorbent is characterized by the following: [0031] 3. A heat of adsorption as determined by isotherm measurements fit with the LRC method show that a heat of adsorption (i.e. -A2).gtoreq.10 kcal/mol and .ltoreq.25 kcal/mol. [0032] 4. A heat of adsorption of methane (or a larger molecule) is .ltoreq.200% that of nitrogen, in another embodiment from about 50% to about 200% of that of nitrogen, and in another embodiment from about 50% to .ltoreq.125% of that of nitrogen. [0033] 5. A total adsorption capacity as determined by the Hiden gravimetric measurements that are allowed up to one week to equilibrate, which is preferably greater than 0.4 mmol/g N.sub.2. Adsorption capacity of nitrogen of .gtoreq.0.2 wt %, in another embodiment .gtoreq.0.7 wt % for a fresh sample activated at 350 to 400.degree. C. for 8 hours under vacuum and measured in a 1 Bar of >99.9% nitrogen at 35.degree. C.
[0034] While these characteristics are primarily described for the separation of nitrogen from methane (natural gas), it should be noted that they will apply to other kinetic based separations as well. The process may also include other adsorbents to remove a range of contaminants that are present in the feed stream including hydrocarbons that contain more than 4 carbon atoms, moisture, carbon dioxide, sulfur containing species or other species that may reduce the working capacity of the adsorbent described herein. These adsorbents could comprise activated carbon, silica, alumina, zeolites, titanosilicates, iron based, amine containing adsorbents or mixtures thereof. Typically, silica and alumina adsorbents are used for initial water removal, followed by zeolites. Typically, titanosilicates, zeolites, activated carbon, amine containing, or iron-based adsorbents are used for sulfur removal. Typically, zeolites, titanosilicates, activated carbon, silica or amine containing adsorbents are used for carbon dioxide removal. Typically, silica gel or activated carbon are used for hydrocarbon removal.
[0035] In the event that one of these adsorbents fails to remove the species, thermal regeneration may be performed to remove that species from the adsorbent described and still fall within the realm of this invention which is to eliminate thermal regeneration from being used to remove the product gas of the invention. Methods to characterize the adsorbent are given below.
Pilot Description
[0036] The pilot system is a pressure swing adsorption system that operates by exploiting the difference in adsorption capacity of an adsorbent for the gas of interest over a specific pressure range. When the vessel containing the adsorbent is pressurized, the adsorbent will selectively adsorb the contaminant from the gas stream and thus remove it from the product stream that exits through the other end of the vessel. When vessel is depressurized, the contaminant will desorb and the adsorbent will be ready to process the feed stream again. This process is made into a semi-continuous batch process by having 1 vessel or more than 1 vessel available to process the gas at the majority of all times. With more than 1 vessel to process gas, additional options are available to further increase efficiency by retaining pressurized gas in dead volume spaces (piping or the heads of the vessels) and the process then has the ability to generate a continuous stream of product.
[0037] The conceptual process flow diagram is presented in FIG. 6.
[0038] The pilot system employs multiple PSA vessels to achieve the desired nitrogen rejection and hydrocarbon recovery target. The current pilot PSA design consists of 4-6 vessels with process steps consisting of 1 bed on feed and 1 bed on blowdown at a time. There are 2-3 equalization steps as well as product pressurization and purge steps. The pilot system was designed to process up to 17 kscfd and capable of using 1 to 4 inch diameter beds. During the initial construction of the pilot test system the bed size was selected to be 1 inch due to the adsorbent performance and with considerations of adsorbent manufacturing. The height was based on maximum available height in the container. The remaining components of the design were based on similar 6 bed PSA pilot plant already in operation. Full range control valves were used for all valves. The system was constructed entirely of stainless steel grade 316. Additionally, a pretreatment system of 304 stainless steel was designed and built as H2S compatible in order to remove all condensed liquids and sulfur before entering the PSA portion of the system.
LRC Description
[0039] Adsorbents were characterized using the loading ratio correlation (LRC) method as described herein and based on the article "Multicomponent Adsorption Equilibria on Molecular Sieves" by Yon and Turnock, published as part of the AIChE Symposium Series, 117, Vol. 67, in 1971 in Adsorption Technology. Isotherm measurements were performed by using an IGA balance as described below, for temperatures of 20.degree. C., 35.degree. C. and 50.degree. C.
Hiden IGA Description (Equilibrium and Rate)
[0040] Rate and equilibrium characterization of samples were performed using a Hiden IGA pressure microbalance (Model#HAS022650) which measures single component gas uptake and was used to examine the adsorption of N.sub.2 and CH.sub.4. The samples were loaded and gas adsorptions were measured as instructed in the IGA Systems User Manual #HA-085-060. Each sample was loaded and activated in situ under vacuum with a temperature ramp of 0.7 C/min to between 350 and 400.degree. C. and held for 12 hours. It was then cooled to the adsorption test temperature at a rate of 1.degree. C./min. The amount of gas adsorbed by the adsorbent is measured in micrograms at a fixed temperature controlled by a constant temperature bath. The pressures points are taken from 0.1 bar to 10 Bar allowing up to 7 days to reach equilibrium. Equilibrium and leak check verification is done by a desorption isotherm that matches the adsorption isotherm. A buoyancy correction was determined using helium and this was used to adjust the microgram weight for buoyancy effects using the molecular weight of the gas being measured. The buoyancy corrected microgram weight was used to calculate uptake using standard methods and using the activated sample weight. For rate measurements, the test gas (N.sub.2 or CH.sub.4) was introduced at 1 Bar then the sample was held at pressure recording the weight as a function of time. System dynamics require approximately 2 minutes to stabilize. Weight data after 2 minutes were corrected for buoyancy and converted to uptakes in weight % or mmol/g and the uptake versus time data were fit to a first order process to obtain rates. Each material was tested first for N.sub.2, prior to being reactivated before repeating the test using CH.sub.4.
Breakthrough Description
[0041] A breakthrough test system was created to test the adsorbent samples using a 12'' long 1'' pipe filled with adsorbent. A breakthrough test was run by first saturating the bed with a flow of 300 sccm at 400 psig of 99% methane (where methane is >99.99%) and 1% helium (where helium is >99.99%) gas for 2 hours, then a flow of 300 sccm of a 49.75/49.75/0.5 mixture of N.sub.2 (where nitrogen is >99.99%)/CH.sub.4/He was introduced as a feed gas to the adsorbent bed and the outlet gas was measured using a gas chromatography mass spectrometer. The breakthrough was recorded as a nitrogen breakthrough example. After 30 minutes this flow was switched to 300 sccm of 99% nitrogen and 1% helium and held for 2 hours. Then the flow was switched back to the 300 sccm of 49.75/49.75/0.5 mixture of N.sub.2/CH.sub.4/He and this was recorded as the methane breakthrough. These breakthrough curves were then used with gPROMS software provided by Process Systems Enterprise, Inc. (PSE) to automatically perform parameter estimation of a model that was created as a replica of the system. The libraries supplied with the adsorption aspect of Process Builder from PSE are sufficient to replicate these results. A detailed description and instructions on how to perform these simulations is provided by PSE.
Modeling Description
[0042] The results from the breakthrough test and parameters obtained from the modeling were used with the methodology described by Mehrotra, et al. in Arithmetic Approach for Complex PSA Cycle Scheduling, Adsorption, 2010, pp. 113-126, vol. 16, Springer Science+Business Media which details the basis for modeling PSA processes. These simulations were performed using Process Builder, from PSE.
TGA Rate Measurements
[0043] A TGA method was developed to assess comparative nitrogen rates that involves both an in-situ activation step followed by adsorption tests using oxygen and nitrogen at 25.degree. C. The thermogravimetric method using a TA Instruments Q500 system installed in a glove box to minimize the impact of air leaks. Nitrogen, and oxygen, gases supplied to the instrument were high purity. The balance purge gas and gas 1 was nitrogen and a gas 2 corresponds to oxygen. For all experiments, a balance purge of 5 cc/minute was used and the gas directly over the sample was set to 95 cc/minute (nitrogen or oxygen). A sampling frequency of 0.5 sec/point was used for all adsorption steps. Alumina pans were used for all studies and the sample size after activation was in the range 100 to 120 mg. The sample activation was performed by heating the sample under nitrogen purge at 2.degree. C. per minute to 150.degree. C., maintaining isothermal for 60 minutes, heating at 5.degree. C./minute to 350.degree. C., holding at 350.degree. C. for 120 minutes, then cooling to 25.degree. C. The nitrogen equilibrium capacity at atmospheric pressure and 25.degree. C. is reported as the weight gain on cooling under nitrogen relative to the minimum weight at 350.degree. C. (the activated sample weight). An assessment of relative rate for different samples and preparation is captured by switching from nitrogen to oxygen. A transient weight gain is observed followed by a drop attributable to oxygen uptake followed by nitrogen leaving. A corresponding switch from oxygen back to nitrogen results in a transient weight loss followed by a weight gain attributable to oxygen loss followed by nitrogen pickup. Values reported as "nitrogen uptake rate" correspond to the maximum slope observed in the nitrogen uptake portion and is equivalent also to the peak in the derivative weight with respect to time for the same step. Values are reported in weight %/minute. Rate measurements for selectivity determinations relied exclusively on a Hiden pressure microbalance rather than the TGA method.
EXAMPLE 1. MODELING RESULTS FOR HIGHER SELECTIVITY RATIOS
[0044] This example demonstrates that once the rate selectivity is above 30, the ratio of uptake rates (N.sub.2/CH.sub.4) as measured via Hiden microbalance, does not significantly impact performance until a ratio of uptake rates is greater than 1,000,000 which has not been achieved in an economically viable offering. At a ratio of 1, the system works against the desired separation to instead produce a purified product of nitrogen. At ratios above 5, the adsorption of methane becomes too low on a normal cycle and the product of purified methane begins to emerge. At ratios above 35 the adsorption of methane fails to negatively impact the performance of the system with proper process cycles and the system performs at peak performance for the majority of selectivity ratios studied. The exception is that above a selectivity of 1,000,000, then the adsorbent does not reasonably saturate with methane during the expected lifetime of the system (>5 years) and thereby increases the working capacity of nitrogen almost 100% vs CSS conditions. Selectivity ratios of almost 1,000,000 have never been reported in literature and are currently .about.10,000 times higher than the state of the art.
TABLE-US-00001 Ratio of rates of uptake BSF at year 5 (N.sub.2/CH.sub.4) (lbs/MMscfd Feed) 1 -- 10 3600 35 1200 100 1200 1000000 1200 10000000 600 For 35% N.sub.2 in feed to 20% N.sub.2 in product at a recovery of 80% at 35 C.
EXAMPLE 2. MODELING RESULTS DEMONSTRATIONG REDUCED SELECTIVITY, AND HIGHER UPTAKE OF NITROGEN BENEFIT
[0045] The commercial performance of the tunable zeolite 4A was modeled with a relative rate of 0.9 wt % N.sub.2/min characterized material. The recovery is the total hydrocarbons recovered from the 4-bed system. The production is the relative production of the system at different conditions. The purity is the methane concentration of the product. The N2 rate is the rate of uptake of nitrogen on the material relative to the 0.9 wt % N.sub.2/min uptake rate material. The CH.sub.4 rate is the rate of uptake of methane on the modeled material relative to the same material basis which was 0.03wt % CH.sub.4/min. The feed concentration is 35% N.sub.2, 65% Methane at 35.degree. C.
TABLE-US-00002 TABLE 1 the rate selectivity dependence of modified 4A (WO201715431164) for changing uptake rate of nitrogen. Recovery Purity Production N.sub.2 Rate CH.sub.4 Rate 66% 90% 100% 100% 100% 60% 90% 90% 100% 200% 47% 90% 71% 100% 400% 52% 90% 99% 20% 100% 21% 90% 95% 4% 100% 74% 90% 160% 200% 200% -- 55% -- 800% 800% 53% 90% 90% 20% 20%
Table 1 show that doubling the uptake rate of methane decreases the recovery of a fixed bed size system, but only by .about.10%. This demonstrates that higher selectivity only has a marginal benefit in this regime.
[0046] In the case where the rate of uptake of both nitrogen and methane are doubled the theoretical performance is significantly higher, however this assumes the rate selectivity ratio is maintained, which unfortunately it is not. This demonstrates that higher uptake rates of nitrogen are preferred, as described herein.
[0047] A 400% increase in methane uptake begins to lower the recovery substantially more, however the material is still viable for the separation. This further demonstrates that higher selectivity is not the most important consideration even at the edges of the proposed characteristics described herein.
[0048] If an 800% increase to both rates is modeled, the material is no longer able to maintain a product purity, and instead begins to remove methane from nitrogen. This is an effect of accounting for physical restrictions within the system related to valve open speeds and gas flows across the adsorbent. If a system were designed to mitigate these, the higher rates of adsorption could be tolerated, and the system size could be reduced even more. However, it is important to note here that typical adsorption systems have physical limits that render them unable to utilize such high rate materials and the practical design point for materials when considering these factors is a slower uptake rate of nitrogen and methane.
[0049] In the case of lowered rates of adsorption of nitrogen but maintained or lowered rates of adsorption of methane we see a drop in the process performance, but the performance is not affected by the rate of methane uptake in this regime. This suggests that at a certain point, slowing the uptake rate of methane no longer benefits the process as shown in example 1. At a certain point, the rate of uptake of nitrogen is sufficiently slow that the process begins to perform very poorly at a fixed bed size and can only be remedied with very costly increases to the bed size. This demonstrates the improvement discovered here of increased rates of nitrogen uptake that the expense of even larger increases to the rate of methane uptake.
EXAMPLE 3. FASTER SYSTEM RESPONSE
[0050] Since one of the benefits of higher selectivity is the increased time to reach cyclic steady state (CSS), it's important to note that CSS is reached significantly faster with these new material characteristics. A defining characteristic of state-of-the-art materials is strong competing adsorption via methane which results in a lowered working capacity/minimal working capacity after adsorbent saturation. In a normal PSA cycle with ETS-4, very large adsorbent beds must be utilized with very low recovery systems to generate a moderate purity product. To counter this, one can implement methods to desaturate the adsorbent and balance the economics of large beds with low recovery (normal) or high capital (with desaturation). One example of desaturation is thermal regeneration. U.S. Pat. No. 6,444,012 to Dolan et al describes a method to desaturate the adsorbent by heating the product stream (largely methane) in order to force methane out of the pores via a TSA process. This consumes methane, energy (for heating) and requires replacement capital of the beds not undergoing thermal regeneration in order to maintain continuous operation. Additionally, this requires that the adsorption rate of methane is very slow. A very slow adsorption rate of methane usually is associated with a slow adsorption rate of nitrogen. CSS loading capacity of methane on Tunable 4A was reached in approximately 30 minutes via modeling and pilot experiments for the highest performing adsorption rates tested. This has additional benefits such as being able to respond rapidly to changing feed conditions. Oil and gas wells typically have significant fluctuations and variations. Responding to these is an additional benefit of faster adsorption rate-based processes.
TABLE-US-00003 wt %/min N2 CSS Time Pilot CSS Time model uptake (min) (min) 1.2 20 22.5 0.9 30 30 0.6 120 45 0.1 -- 270
EXAMPLE 4. LOW RATE SELECTIVITY, HIGH UPTAKE RATE ADSORBENTS
[0051] A material was made to demonstrate the proposed benefit of higher uptake rates of nitrogen even at reduced overall rate selectivity, demonstrating the benefit illustrated by the model in example 1.
[0052] 23.00 lbs. of zeolite 4A powder supplied by Jianlong (as 4A-D) on a dry weight basis (29.50 lbs. wet weight) was placed in a WAM MLH50 plow mixer. With the mixer agitating, 2.16 lbs of MR-2404 (a solventless silicone containing silicone resin from Dow Corning) was pumped in at rate of 0.07 lb/min. After the MR-2404 addition was completed, 9.2 lbs of water was added at a rate of 0.3 lb/min under constant stirring in the plow mixer. At the end of the water addition, plow mixing was continued for an additional 5 minutes. The plow mixed powder product labeled hereinafter "the formulation" was transferred to a tilted rotating drum mixer having internal working volume of .about.75 L and agitated therein at a speed of 24 rpm. Mixing of the formulation was continued while beads were gradually formed which had a porosity, as measured using a Micromeritics Autopore IV Hg porosimeter on the calcined product, in the 30-35% range. The beads were subjected to a screening operation to determine the yield and harvest those particles in the 8.times.16 U.S. mesh size range. The product beads were air dried overnight prior to calcination using a shallow tray method at temperatures up to 595.degree. C. The shallow tray calcination method used a General Signal Company Blue-M electric oven equipped with a dry air purge. .about.500 g. dry wt. of the 8.times.16 U.S. mesh adsorbent was spread out in a stainless steel mesh tray to provide a thin layer. A purge of 200 SCFH of dry air was fed to the oven during calcination. The temperature was set to 90.degree. C., followed by a 6 hour dwell time. The temperature was then increased to 200.degree. C. gradually over the course of a 6 hour period, and further increased to 300.degree. C. over a 2 hour period and finally increased to 595.degree. C. over a 3 hour period and held there for 1 hour before cooling to 450.degree. C. after which the adsorbent was removed, immediately bottled in a sealed bottle and placed in a dry nitrogen purged drybox. The calcined beads were rescreened to harvest those particles in the 8.times.16 U.S. mesh range.
[0053] Characterization of the tunable 4A samples calcined at 595.degree. C. was performed using a thermogravimetric screening method as described earlier in "TGA description". The nitrogen uptake rate as performed in the test was determined to be .about.0.2 weight %/minute as measured using the TGA method disclosed herein. When the product beads in Example 1 were calcined up to 575.degree. C., the nitrogen uptake rate as performed in the test was determined to be .about.0.7 weight %/minute as measured using the TGA method disclosed herein. Subsequently, when the product beads in Example 1 were calcined up to 555.degree. C., the nitrogen uptake rate as performed in the test was determined to be .about.1.2 weight %/minute as measured using the TGA method disclosed herein.
Breakthrough Data from Model and Lab Experiment
[0054] The breakthrough data demonstrates the achievement of the required rate characteristics, and is shown in the FIG. 5 by the minimum required rates of adsorption for nitrogen, and maximum rates of adsorption of methane, and the ideal rates of adsorption for both of these. It is clear in the FIG. 5 that the actual adsorbent had rates of adsorption of both components in between these two extremes. These two extremes also determine the characteristics alternative materials need to meet in order to have high performance in this process. Also shown in the FIG. 5 is clinopotilolite (clino) TSM-140 which is commercialy available. This state of the art material does not have the uptake rate of nitrogen to meet the characteristics described here.
Pilot Data
[0055] The relative rate of uptake correlates to the TGA measurement. The selectivity is the rate of uptake of nitrogen divided by the rate of uptake of methane as determined by the breakthrough test and model fitting. The pilot recovery is the recovery observed in the pilot system for a feed concentration of 35% nitrogen, a flow rate of 120 scfh using four 1'' beds filled 5.5' tall. The product impurity was held at 20% nitrogen. The results show that with increasing selectivity, the pilot recovery falls significantly, due to the inability of the material to process enough gas to overcome the losses from void spaces. This is an unfortunate reality of high selectivity is that is typically corresponds to reduced uptake rate of nitrogen. When this is considered, the recovery rises substantially, up to a point that the process is unable to take advantage of the higher rate of uptake to due to low response time of the system and the fast uptake of methane which ultimately both work to lower recovery. One way that others have overcome this low recovery is to increase the bed size, thereby increasing the amount of gas the material is able to process and offsetting the void space losses. Since this modification is no longer necessary, this recovery gain is equivalent on a commercial scale to lower bed sizes.
TABLE-US-00004 TABLE 2 showing the impact of relative uptake rate on selectivity and on final recovery in the pilot system. N.sub.2 rate of uptake Rate Selectivity Pilot wt %/min (N.sub.2/CH.sub.4) Recovery 0.1 120 2% 0.6 70 6% 0.9 44 24% 1.2 30 20%
EXAMPLE 5. MODELING SENSITIVITY TO PRODUCT PURITY AND PRESSURE
[0056] Another study was conducted to determine the optimal adsorbent for a product purity of 5% nitrogen compared to 20% nitrogen and for a feed stream of 200 psig compared to 600 psig. The results show that some variation in the preferred adsorption uptake rate of nitrogen and the subsequent ratio of uptakes compared to methane exists, but that this variation is typically limited to +/-50% between applications. Additionally, while the optimal target can vary up to 50%, the performance for a 50% variation in uptake rates does not generally cause a larger variation in process performance.
TABLE-US-00005 TABLE 3 Model N.sub.2 rate of Rate Projected uptake Selectivity Pilot Feed Product wt %/min (N2/CH4) Recovery impurity impurity 0.1 120 4% 35% 20% 0.6 70 8% 35% 20% 0.9 44 28% 35% 20% 1.2 30 22% 35% 20% 0.1 120 8% 10% 5% 0.6 70 34% 10% 5% 0.9 44 32% 10% 5% 1.2 30 28% 10% 5%
Table 3 shows that for different processing feed impurities and desired product impurity levels, the optimal adsorption rate of nitrogen can vary accordingly. In particular, as the impurity level is reduced, the impact of selectivity becomes more important and the importance of the uptake rate of nitrogen begins to fall. This should be considered when selecting the optimal characteristics for the process. It also leads to the prospective of multiple layers of varying uptake rate tunable zeolite 4A adsorbents in order to best accomplish a separation process when there is a potential large variation in impurity levels through the process, as a method to best reduce the overall bed size of the system.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.