Low Profile Non-symmetrical Stent
Brocker; David ; et al.
U.S. patent application number 17/063092 was filed with the patent office on 2021-01-21 for low profile non-symmetrical stent. This patent application is currently assigned to COOK MEDICAL TECHNOLOGIES LLC. The applicant listed for this patent is COOK MEDICAL TECHNOLOGIES LLC. Invention is credited to David Brocker, William K. Dierking, Jarin A. Kratzberg, Alan R. Leewood, Erik E. Rasmussen, Blayne A. Roeder.
| Application Number | 20210015643 17/063092 |
| Document ID | / |
| Family ID | 1000005131949 |
| Filed Date | 2021-01-21 |









View All Diagrams
| United States Patent Application | 20210015643 |
| Kind Code | A1 |
| Brocker; David ; et al. | January 21, 2021 |
LOW PROFILE NON-SYMMETRICAL STENT
Abstract
Various stents and stent-graft systems for treatment of medical conditions are disclosed. In one embodiment, an exemplary stent-graft system may be used for endovascular treatment of a thoracic aortic aneurysm. The stent-graft system may comprise proximal and distal components, each comprising a graft having proximal and distal ends, where upon deployment the proximal and distal components at least partially overlap with one another to provide a fluid passageway therebetween. The proximal component may comprise a proximal stent having a plurality of proximal and distal apices connected by a plurality of generally straight portions, where a radius of curvature of at least one of the proximal apices may be greater than the radius of curvature of at least one of the distal apices. The distal component may comprise a proximal z-stent coupled to the graft, where the proximal end of the graft comprises at least scallop formed therein that generally follows the shape of the proximal z-stent. Further, the distal component may comprise at least one z-stent stent coupled to the distal end of the graft and extending distally therefrom that reduces proximal migration of the distal component.
| Inventors: | Brocker; David; (Carmel, IN) ; Dierking; William K.; (Louisville, KY) ; Leewood; Alan R.; (Lafayette, IN) ; Roeder; Blayne A.; (Bloomington, IN) ; Kratzberg; Jarin A.; (West Lafayette, IN) ; Rasmussen; Erik E.; (Slagelse, DK) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | COOK MEDICAL TECHNOLOGIES
LLC BLOOMINGTON IN |
||||||||||
| Family ID: | 1000005131949 | ||||||||||
| Appl. No.: | 17/063092 | ||||||||||
| Filed: | October 5, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15982523 | May 17, 2018 | 10828183 | ||
| 17063092 | ||||
| 14952498 | Nov 25, 2015 | 9980834 | ||
| 15982523 | ||||
| 12904452 | Oct 14, 2010 | 9226813 | ||
| 14952498 | ||||
| 12332904 | Dec 11, 2008 | 9180030 | ||
| 12904452 | ||||
| 61016753 | Dec 26, 2007 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61F 2230/005 20130101; A61F 2/848 20130101; A61F 2002/8483 20130101; A61F 2/915 20130101; A61F 2210/0014 20130101; A61F 2002/91516 20130101; A61F 2002/8486 20130101; A61F 2002/91541 20130101; A61F 2002/075 20130101; A61F 2220/0075 20130101; A61F 2002/061 20130101; A61F 2002/826 20130101; A61F 2/852 20130101; A61F 2230/0067 20130101; A61F 2002/91533 20130101; A61F 2220/0016 20130101; A61F 2/07 20130101; A61F 2/856 20130101; A61F 2002/91508 20130101; A61F 2002/065 20130101; A61F 2250/006 20130101; A61F 2230/0054 20130101; A61F 2/89 20130101 |
| International Class: | A61F 2/89 20060101 A61F002/89; A61F 2/915 20060101 A61F002/915; A61F 2/07 20060101 A61F002/07 |
Claims
1-20. (canceled)
21. A stent-graft system for treatment of a medical condition, the stent-graft system comprising: a proximal component comprising a graft having proximal and distal ends, and further comprising a stent having a plurality of proximal and distal apices connected by a plurality of generally straight portions, where at least one of the distal apices of the stent is attached to the graft using one or more sutures, where each proximal apex comprises a first curved portion and each distal apex comprises a second curved portion, where the first curved portion and the second curved portion each comprises at least one radius of curvature, and the radius of curvature of at least one of the proximal apices is greater than the radius of curvature of at least one of the distal apices; and a distal component comprising a graft having proximal and distal ends, where, upon deployment, the proximal and distal components at least partially overlap with one another to permit continuous fluid flow therebetween, where a first radius of curvature of one of the proximal apices is greater than 1.5 mm.
22. The stent-graft system of claim 21 where the first radius of curvature of one of the proximal apices is at least two times greater than the radius of curvature of at least one of the distal apices.
23. The stent-graft system of claim 21, where the distal component further comprises a proximal z-stent coupled to the graft, and where the proximal end of the graft comprises at least one scallop formed therein that generally follows the shape of the proximal z-stent.
24. The stent-graft system of claim 21, where the distal component further comprises at least one z-stent stent coupled to the distal end of the graft and extending distally therefrom that reduces proximal migration of the distal end of the distal component.
25. The stent-graft system of claim 24, wherein the at least one z-stent stent coupled to the distal end of the graft comprises at least one barb oriented in a proximal direction.
26. The stent-graft system of claim 21 where each of the proximal apices of the stent are circumferentially offset from the distal apices.
27. The stent-graft system of claim 21 where the stent is a proximal stent, and where the proximal component comprises at least five additional z-stents coupled to the graft at locations distal to the proximal stent.
28. The stent-graft system of claim 27 where, of the at least five additional z-stents, proximal and distal z-stents are coupled to an inner surface of the graft and at least three intermediate z-stents are coupled to an outer surface of the graft.
29. A stent-graft system for treatment of a medical condition, the stent-graft system comprising: a proximal component comprising a graft having proximal and distal ends, and further comprising a first stent having a plurality of proximal and distal apices connected by a plurality of generally straight portions, where at least one of the distal apices of the first stent is attached to the graft using one or more sutures, where each proximal apex comprises a first curved portion and each distal apex comprises a second curved portion, where the first curved portion and the second curved portion each comprises at least one radius of curvature, and the radius of curvature of at least one of the proximal apices is greater than the radius of curvature of at least one of the distal apices; and a distal component comprising a graft having proximal and distal ends, where, upon deployment, the proximal and distal components at least partially overlap with one another to provide a fluid passageway therebetween, where one of the proximal apices comprises a first radius of curvature, where a second radius of curvature of one of the distal apices is from about 0.6 mm to about 1.5 mm, and where a ratio of the second radius of curvature to the first radius of curvature is about 1:2.6 to about 1:18.
30. The stent-graft system of claim 29 where the distal component further comprises a proximal z-stent coupled to the graft, and where the proximal end of the graft comprises at least one scallop formed therein that generally follows the shape of the proximal z-stent.
31. The stent-graft system of claim 29 where each of the proximal apices of the first stent are circumferentially offset from the distal apices.
32. The stent-graft system of claim 29 where the proximal component comprises at least five additional z-stents coupled to the graft at locations distal to the first stent.
33. The stent-graft system of claim 29 where the first radius of curvature is about 1 mm, and the second radius of curvature is about 6 mm.
34. A stent-graft system for treatment of a medical condition, the stent-graft system comprising: a proximal component comprising a graft having proximal and distal ends, and further comprising a stent having a plurality of proximal and distal apices connected by a plurality of generally straight portions, where at least one of the distal apices of the stent is attached to the graft using one or more sutures, where each proximal apex comprises a first curved portion and each distal apex comprises a second curved portion, where the first curved portion and the second curved portion each comprises at least one radius of curvature, and the radius of curvature of at least one of the proximal apices is greater than the radius of curvature of at least one of the distal apices; and a distal component comprising a graft having proximal and distal ends, where, upon deployment, the proximal and distal components at least partially overlap with one another to provide a fluid passageway therebetween, where a first radius of curvature of one of the distal apices of the stent is at least two times less than a second radius of curvature of one of the proximal apices.
35. The stent-graft system of claim 34 where a ratio of the first radius of curvature to the second radius of curvature is about 1:2.6 to about 1:18.
36. The stent-graft system of claim 34 where each of the proximal apices of the stent are circumferentially offset from the distal apices.
37. The stent-graft system of claim 34 where the distal component further comprises a proximal z-stent coupled to the graft, and where the proximal end of the graft comprises at least one scallop formed therein that generally follows the shape of the proximal z-stent.
38. The stent-graft system of claim 34 where the at least one z-stent stent coupled to the distal end of the graft comprises at least one barb oriented in a proximal direction.
39. The stent-graft system of claim 34 where the first radius of curvature is about 1 mm, and the second radius of curvature is about 6 mm.
40. The stent-graft system of claim 34 where the stent is a proximal stent, and where the proximal component comprises at least five additional z-stents coupled to the graft at locations distal to the proximal stent.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation-in-part of U.S. Utility patent application Ser. No. 12/841,807, filed Jul. 22, 2010 and entitled "Apparatus and Methods for Deployment of a Modular Stent-Graft System," which is a continuation-in-part of U.S. Utility patent application Ser. No. 12/622,351, filed Nov. 19, 2009 and entitled "Low Profile Non-Symmetrical Stent," which claims priority to U.S. Provisional Application Ser. No. 61/016,753, filed Dec. 26, 2007, co-pending U.S. patent application Ser. No. 12/332,904, filed Dec. 11, 2008 and Ser. No. 12/472,082, filed May 26, 2009, and co-pending Great Britain Patent Application Nos. GB0920235.9, filed Nov. 18, 2009 and GB0920327.4, filed Nov. 19, 2009, each of which is incorporated herein by reference in its entirety.
BACKGROUND
[0002] The present invention relates generally to stents for use in body vessels to treat medical conditions. In particular, this invention relates to an asymmetric stent having opposing sets of curved apices, where the curved section of one set of apices has a radius of curvature that is greater than the curved section of the other set of apices, and may present a lower profile, better compliance with irregular vascular geometry, and higher sealing forces than conventional stents.
[0003] Stents may be inserted into an anatomical vessel or duct for various purposes. Stents may maintain or restore patency in a formerly blocked or constricted passageway, for example, following a balloon angioplasty procedure. Other stents may be used for different procedures, for example, stents placed in or about a graft have been used to hold the graft in an open configuration to treat an aneurysm. Additionally, stents coupled to one or both ends of a graft may extend proximally or distally away from the graft to engage a healthy portion of a vessel wall away from a diseased portion of an aneurysm to provide endovascular graft fixation.
[0004] Stents may be either self-expanding or balloon-expandable, or they can have characteristics of both types of stents. Various existing self-expanding and balloon-expandable stent designs and configurations comprise generally symmetrical end regions including one or more apices formed of nitinol or another alloy wire formed into a ring. The apices commonly comprise relatively acute bends or present somewhat pointed surfaces, which may facilitate compression of the stent to a relatively small delivery profile due to the tight bend of the apices. Although having this advantage, in some situations, such relatively acute or pointed apices may be undesirable, in particular in vessel anatomies that are curved or tortuous such as, for example, the thoracic aorta.
[0005] The thoracic aorta presents a challenging anatomy for stent grafts used to treat thoracic aneurysms or dissections. The thoracic aorta comprises a curve known as the aortic arch, which extends between the ascending thoracic aorta (closet to the heart) and the descending thoracic aorta (which extends toward the abdominal aorta). Thoracic stent grafts are used to exclude thoracic aortic aneurysms. A stent graft's ability to conform to the tortuous anatomy of the aortic arch is a major concern. Current designs sometimes lack the desired sealing ability at the proximal end of the stent graft (closest to the heart). Also, current thoracic devices present a relatively large profile which, with some patients' anatomies may be problematic. Finally, many current stents have relatively acute points that may prevent them from being used in the aortic arch for fear of undesirable interaction with the artery wall after an extended amount of time in the patient.
[0006] Therefore, a generally nonsymmetrical stent having at least one relatively rounded apex that is less invasive in an expanded state than stents with more acute apices may alleviate the above problems, while providing an improved compliance to the aortic arch and increased radial force if used as a sealing and/or alignment stent, as well as a desirable ability to be crimped to a readily introducible diameter.
[0007] As one particular example, type-A thoracic aortic dissection (TAD-A) is a condition in which the intimal layer of the ascending thoracic aorta develops a tear, allowing blood to flow into the layers of the aortic wall, causing the development of a medial or subintimal hematoma. TAD-A is associated with a strikingly high mortality rate (about one-fourth to one-half of victims die within the first 24-48 hours). The only current treatment for TAD-A is open surgery, where the chest is opened, the aorta is clamped, and a vascular prosthesis is sewn in place. Operative mortality rate for this procedure may be around 10%. Endovascular treatment of TAD-B (which affects the descending thoracic aorta) has been effective in reducing short-term and longer term mortality. Therefore, it is desirable to provide an endovascular device configured to address the anatomic challenges of the thoracic aorta.
SUMMARY
[0008] Various stents and stent-graft systems for treatment of medical conditions are disclosed. In one embodiment, an exemplary stent-graft system may be used for endovascular treatment of a thoracic aortic aneurysm.
[0009] The stent-graft system may comprise proximal and distal components, each comprising a graft having proximal and distal ends, where upon deployment the proximal and distal components at least partially overlap with one another to provide a fluid passageway therebetween. The proximal component may comprise a proximal stent having a plurality of proximal and distal apices connected by a plurality of generally straight portions, where a radius of curvature of at least one of the proximal apices may be greater than the radius of curvature of at least one of the distal apices.
[0010] The distal component may comprise a proximal z-stent coupled to the graft, where the proximal end of the graft comprises at least scallop formed therein that generally follows the shape of the proximal z-stent. Further, the distal component may comprise at least one z-stent coupled to the distal end of the graft and extending distally therefrom that reduces proximal migration of the distal component.
[0011] Advantageously, when the stent graft system is deployed, the proximal stent of the proximal component will maximize the efficacy of the proximal seal while reducing atraumatic contact with an artery wall, and further preventing distal migration of the proximal end of the proximal component. Further, the at least one scallop may advantageously reduce the potential for graft infolding, thereby reducing or eliminating the likelihood of impeded blood flow and/or endoleaks around the distal component.
[0012] Other systems, methods, features and advantages of the invention will be, or will become, apparent to one with skill in the art upon examination of the following figures and detailed description. It is intended that all such additional systems, methods, features and advantages be within the scope of the invention, and be encompassed by the following claims.
BRIEF DESCRIPTION OF THE DRAWINGS
[0013] The invention can be better understood with reference to the following drawings and description. The components in the figures are not necessarily to scale, emphasis instead being placed upon illustrating the principles of the invention. Moreover, in the figures, like referenced numerals designate corresponding parts throughout the different views.
[0014] FIGS. 1-3 show different views of a symmetrical stent;
[0015] FIG. 4 depicts an example of an asymmetric stent;
[0016] FIG. 5 diagrammatically illustrates the asymmetrical radii of curvature of the stent of FIG. 4;
[0017] FIG. 6 shows the stent of FIG. 4 in a simulated artery;
[0018] FIG. 7 depicts another example of an asymmetric stent;
[0019] FIG. 8 diagrammatically illustrates the asymmetrical radii of curvature of yet another example of a stent;
[0020] FIG. 9 shows the stent of FIG. 8 in a simulated artery;
[0021] FIG. 10 shows an end view of still another example of an asymmetric stent;
[0022] FIG. 11 shows a side view of the stent of FIG. 10;
[0023] FIG. 12 is a top perspective view of the stent of FIG. 10;
[0024] FIG. 13 shows the stent of FIG. 10 in a simulated artery;
[0025] FIG. 14 is a partial perspective of a stent-graft incorporating the stent of FIG. 10;
[0026] FIG. 15 illustrates a side view of the stent-graft of FIG. 14;
[0027] FIGS. 16-18 show a stent-graft with side branches; and
[0028] FIG. 19 is a side view of a stent-graft device configured for endovascular treatment of a thoracic aorta dissection.
[0029] FIG. 20 is a side view of a stent-graft configured for endovascular treatment of a thoracic aortic aneurysm.
DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENTS
[0030] The present invention relates generally to stents for use in body vessels to treat medical conditions. In particular, this invention relates to a novel asymmetric stent having opposing sets of curved apices, where the curved section of one set of apices has a radius of curvature that is greater than the curved section of the other set of apices, and may present a lower profile than conventional stents. The lower profile may present advantages for use in patients with particularly tortuous or small-diameter vessels.
[0031] In the present application, the term "proximal" refers to a direction that is generally closest to the heart during a medical procedure, while the term "distal" refers to a direction that is furthest from the heart during a medical procedure. Reference throughout is made to proximal and distal apices, but those of skill in the art will appreciate that the proximal-distal orientation of stents of the present invention may be reversed without exceeding the scope of the present invention.
[0032] As shown in FIGS. 4-15, this novel stent is not symmetrical like many commercially available stents, in that the radius of curvature of the opposing proximal and distal apices is different between the top and bottom of the stent. The stents may be attached to either end of a stent graft to provide sealing and may be used internally or externally to the graft material to provide support to the graft.
[0033] The asymmetric stent may be configured such that, when used with a graft, it will provide a sufficiently strong radial force at the graft's end openings to hold the graft material open against the artery wall. Also, the stent is intended to be short in length so that the graft will include flexibility sufficient to accommodate a patient's anatomy. This combination of flexibility and strong radial force provides an improved seal between the graft and artery wall. In addition, enhanced flexibility is provided as well, particularly when one or more stents are used to provide short segments and better accommodate curves.
[0034] FIG. 1 shows a conventional stent 100, which has symmetrical apices 102, 103. Specifically, the proximal apices 102 and the distal apices 103 all have generally the same radii of curvature (r.sup.1), which is illustrated in graphic form in FIG. 2. FIG. 3 is adapted from an FEA contour simulation and shows the stent 100 in a simulated artery 110, where the stent 100 is 20% oversized. The proximal and distal apices 102, 103 (circled) exert little or no pressure against the artery wall 110, while an intermediate region 107 exerts a higher pressure to provide--in one example--a total radial sealing force of 0.178 lbf. This configuration may be crimped to 18 Fr (e.g., for introduction via a catheter), with a maximum bend strain in the apices 102, 103 of about 5.8%. When using, for example, a typical NiTi wire for the stent, it is desirable not to exceed 10-12% strain to avoid increased risk of deforming the wire or adversely affecting its durability.
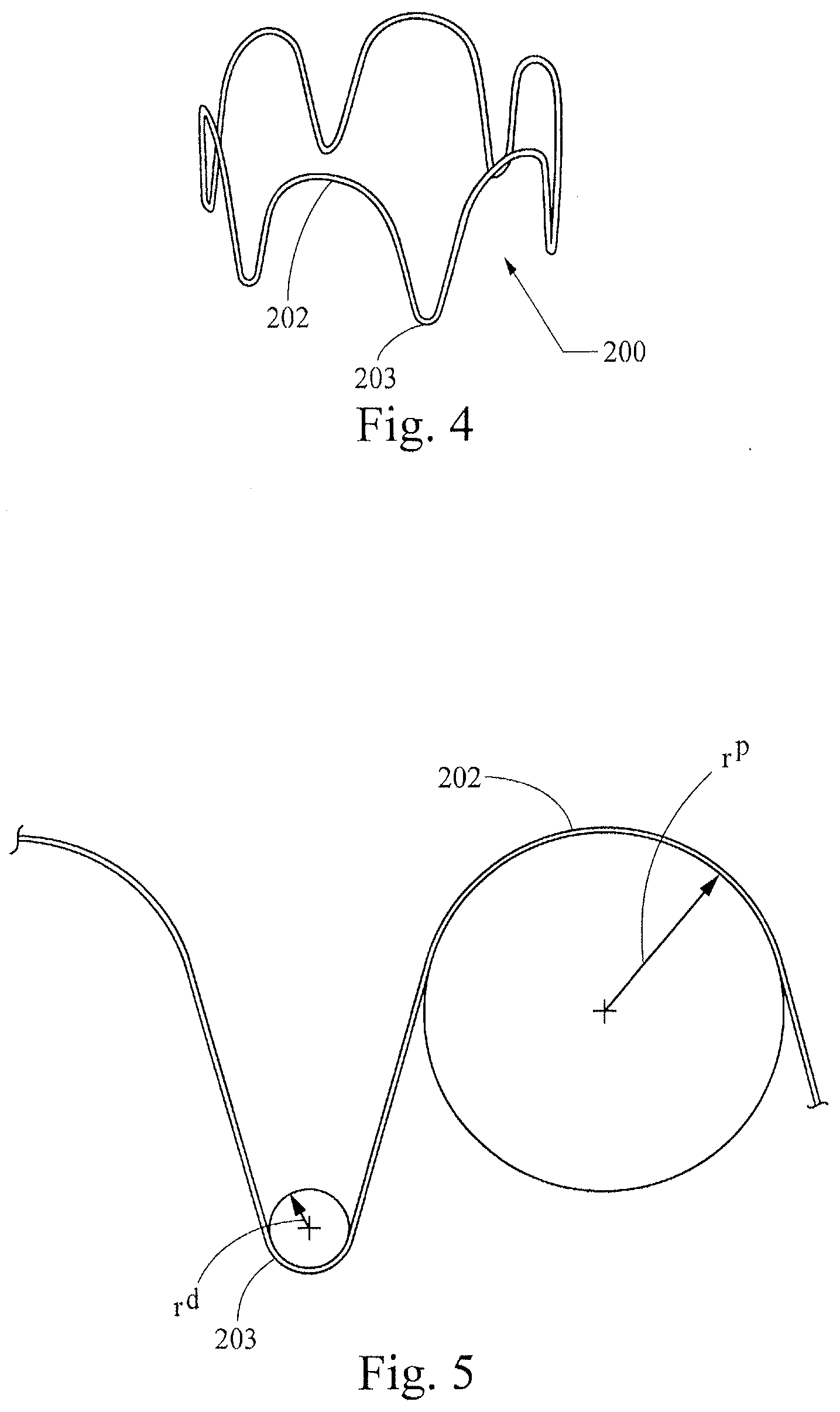
[0035] FIGS. 4-7 show a first example of a non-symmetrical stent 200, which is formed as a wire ring that has non-symmetrical proximal and distal generally curved apex portions (apices) 202, 203 separated from each other by intermediate generally straight portions. Specifically, the distal apices 203 all have generally the same radii of curvature (r.sup.d) as each other, but the distal apices' radii of curvature are different from those of the proximal apices 202 (r.sup.p). The distal apices 203 (which may be attached to and generally covered by graft material in a stent graft as described below with reference to FIGS. 14-15) are generally narrowly rounded in a manner not dissimilar from a traditional z-stent, but the proximal apices 202 are more broadly rounded. The difference in the proximal and distal apices 202, 203 is illustrated in graphic form in FIG. 5. In the illustrated example, the rounded proximal apices 202 have a radius of curvature of 6.0 mm, while the narrower distal apices 202 have a radius of curvature of 1.0 mm. In certain examples of non-symmetrical stents of the present invention, the radius of curvature of the rounded proximal apices (measured in the manner shown in FIG. 5) may be from about 4 mm to about 9 mm, and the radius of curvature of the narrower distal apices may be from about 0.5 mm to about 1.5 mm.
[0036] In these and other examples, the ratio of the proximal apices' radius of curvature to the distal apices' radius of curvature may be about 2.6:1 to about 18:1, and desirably may be about 6:1. The outer circumference of the stent 200 preferably is generally consistent such that, in this configuration, a solid outer face around the stent 200 would form a cylinder, although the stent will most preferably provide compliance with a surface less smooth than a cylinder.
[0037] FIG. 6 is adapted from an FEA contour simulation and shows the stent 200 in a simulated artery 210, where the stent 200 is 20% oversized. The proximal and distal apices 202, 203 (circled) exert little or no pressure against the artery wall 210, while an intermediate region 204 (boxed) exerts a greater pressure to provide --in the illustrated example--a total radial sealing force of about 0.160 lbf. This configuration may be crimped to 18 Fr, with a maximum bend strain in the apices 202, 203 of about 6.5%.
[0038] FIG. 7 shows another non-symmetrical stent embodiment 250 that is very similar to the embodiment of FIGS. 4-6, but which has a shorter proximal-distal length. Each of the examples shown in FIGS. 4-7 may be manufactured in substantially the same manner as current z-stents, with a modification only of forming the proximal apices to include a greater radius of curvature than the distal apices.
[0039] FIGS. 8-9 illustrate another example of a non-symmetrical stent 300, which has a proximal "rounded roof shape" profile rather than the generally semicircular profile of the examples described above with reference to FIGS. 4-7. The profile of each proximal apex 302 includes a central fillet 302a and a pair of symmetrically opposed shoulder fillets 302b that may be generally equidistant from the central fillet 302a, or that may be disposed at varied distances therefrom. For the proximal apices of the stent 300, the central fillets 302a each have a radius of curvature of 1.0 mm, and the shoulder fillets 302b each have a fillet radius of curvature of 0.5 mm. The distal apices 304 have a radius of curvature of 1.0 mm. In another example having the rounded roof shape configuration (not shown), the central and shoulder fillets of proximal apices may each have the same radius of curvature such as, for example, 0.5 mm each, with distal apices also having a 0.5 mm radius of curvature. In other examples, the central and shoulder fillets 302a, 302b may each have a radius of curvature from about 0.5 mm to about 5 mm, and the distal apices may each have a radius of curvature of about 0.5 mm to about 1.5 mm. In another example having the rounded roof shape configuration (not shown), the ratio between the radii of curvature of the central and each shoulder fillet of the proximal apices may be about 3:1. FIG. 8 also shows three spans useful for describing desirable proportions in stent embodiments: "x" indicates the distance between the apical extremities of the shoulder fillets 302b, "y" indicates the distance between the tips of the distal apices 304, and "z" indicates the distance along a longitudinal axis between the tip of the distal apices 304 and the apical extremity of the proximal fillet 302a. Desirable embodiments may include an x:y ratio of about 1:3 to about 7:8 and a y:z ratio of about 1:1 to about 3:1. In yet another example (not shown), the filleted apices of this example may be combined with the generally semicircular apices of the example described with reference to FIGS. 4-7.
[0040] FIG. 9 is adapted from an FEA contour simulation and shows the stent 300 in a simulated artery 310, where the stent 300 is 20% oversized. The proximal and distal apices 302, 304 exert little or no pressure against the artery wall 310, while an intermediate region exerts a greater pressure to provide --in the illustrated example--a total radial sealing force of about 0.420 lbf. This configuration may be crimped to 18 Fr, with maximum bend strains in the apices that may be less than about 9% and preferably are less than about 10-12%. The greater radial sealing force of this example may provide advantages for stent placement and retention in certain circumstances as compared to existing z-stents.
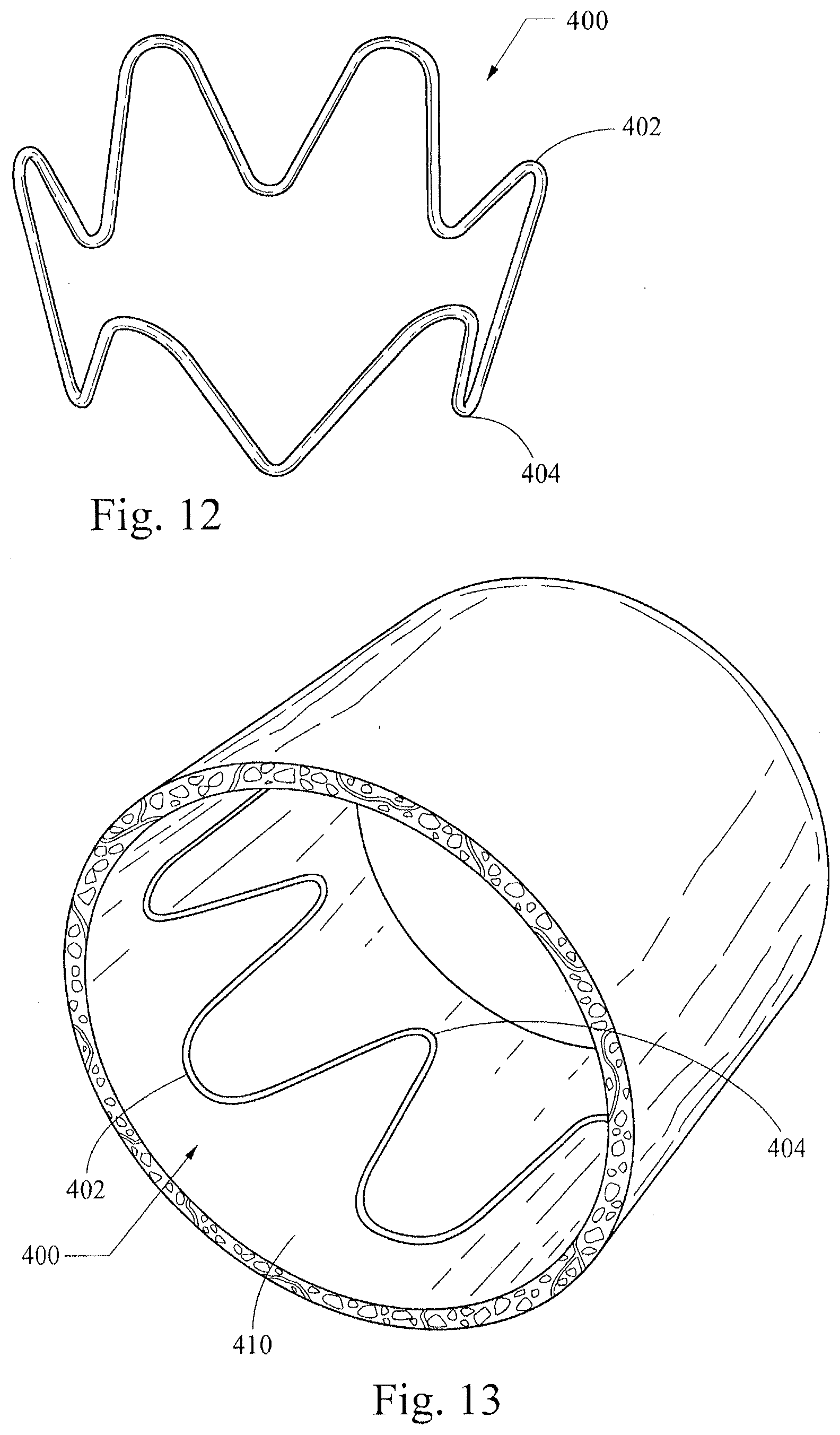
[0041] FIGS. 10-13 illustrate another example of a non-symmetrical stent 400, which has an expanded "flower configuration" as shown in FIG. 10. Specifically, when the stent 400 is in an expanded configuration, the circumference around the proximal more-rounded apices 402 is greater than the circumference around the distal less-rounded apices 404, which is shown most clearly in FIGS. 11-14. In this configuration a solid outer face around an expanded stent 400 would form a frustum of a cone. This configuration may be manufactured in the same manner as the examples described above with reference to FIGS. 4-7 (i.e., producing a stent with a generally uniform outer circumference), with an added step that may include drawing the distal apices 404 into a smaller circumference upon suturing them to a smaller diameter graft material. Alternatively, or in addition, the stent 400 may be heat-set to impose the desired shape.
[0042] FIG. 13 is adapted from an FEA contour simulation and shows the stent 400 in a simulated artery 410, where the stent 400 is 20% oversized. Surprisingly, the contour of pressure distribution along proximal and distal apices 402, 404 as well as an intermediate region is generally uniform throughout the stent circumference. The illustrated configuration provides a total radial sealing force of about 0.187 lbf. This property of generally uniform pressure distribution may provide advantages in certain applications of providing a seal and/or presenting less abrasion of a vessel wall through graft material as compared to stents with less uniform pressure distribution.
[0043] FIGS. 14-15 show two different views of a stent graft 500 using a stent example 400 of the present invention described above with reference to FIGS. 10-13. The stent graft 500 is shown in an expanded state and may be configured for use in treating a thoracic aortic aneurysm. The stent 400 is disposed at the proximal end of a generally cylindrical graft sleeve 502, to which its distal apices 404 are secured by sutures 504. The stent graft 500 also includes a series of z-stents 510a-d disposed distally from the stent 400. The first z-stent 510a is attached to the inner circumference of the graft 502, and the other z-stents 510b-510d are attached to the outer diameter of the graft 502. The proximal end of the stent 400 extends beyond the proximal end of the graft in a manner that may facilitate anchoring the graft in a vessel of a patient (e.g., a blood vessel).
[0044] The rounded points on the stent may protrude from the graft material only a small amount as is shown in FIGS. 14-15. In this example, only a small portion of the bare wire will be exposed to the artery wall. These unique (larger radii) rounded points are far less likely to perforate the artery wall than sharper points of a different stent configuration. Advantageously, this asymmetric stent design will maximize the efficacy of the seal while preserving the condition of the artery wall. Specifically, the narrower stent apices will provide for desirable radial expansion/sealing force, and the broader rounded apices will provide for a desirably atraumatic contact with an artery wall.
[0045] FIGS. 16-18 show a stent-graft embodiment 600 that includes a non-symmetrical stent 602 having more broadly rounded proximal apices 604 and more narrowly rounded distal apices 606. The stent 602 is attached by sutures to the inner surface (not shown) or outer surface of a generally columnar graft 610, which includes other stents 608. A second layer of graft material 612 is also attached to the inner circumference of the graft 610 midway down its length and extends proximally through the inner circumference of the stent 602.
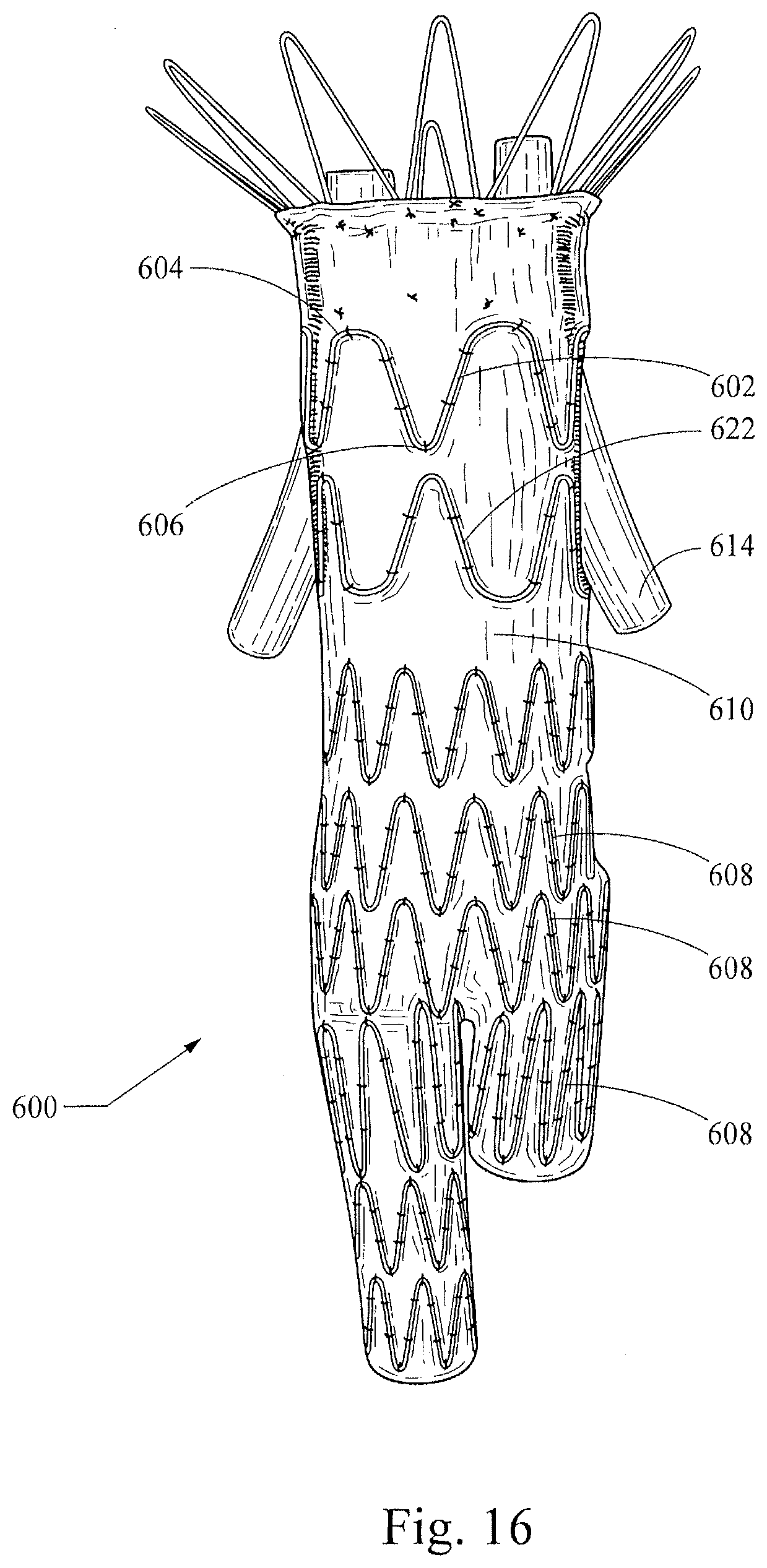
[0046] As shown in the end view of FIG. 17, this construction provides a passage for branch structures 614 (that may be embodied, for example, as tubular or non-tubular stents, stent-grafts, shown here for the sake of illustration as generic tubular structures), which pass through the passage formed between the two layers 610, 612 and through an aperture 611 in the graft 610. The tubular structures 614 will advantageously be disposed generally transversely through the inner radius of the more broadly rounded proximal apices 604 of the stent 602, which provides atraumatic columnar support for the graft 610 as well as an anchor for the tubular structures 614. The stent-graft 600 may be particularly useful for treatment of an abdominal aortic aneurysm (AAA) that is immediately adjacent to, or that goes across, the renal arteries such that it has a short neck and lacks a contact area that is sufficient to create an effective proximal seal and avoid the proximal Type I endoleaks that may occur with some currently-available AAA stent-grafts. Those of skill in the art will appreciate that the stent-graft 600 will allow general occlusion of the AAA, while providing patent passage through the descending aorta and from the aorta to the renal arteries. Specifically, a stent-graft configured in the manner of the stent-graft embodiment 600, which includes a modular design that may include branch stents and/or stent-grafts, will allow a seal to be formed above the renal arteries and below the celiac and superior mesenteric arteries. Also, as shown in FIG. 16, a second non-symmetrical stent 622 may be placed adjacent the first non-symmetrical stent 602 in an opposite orientation that will provide additional atraumatic support for the branching tubular structures 614.
[0047] FIG. 19 shows a stent-graft device 700 configured for endovascular treatment of a thoracic aorta dissection. The device 700 includes a non-symmetrical alignment stent 702 attached to a first end of a tubular graft material 704. A sealing stent 706 is attached in the central lumenal graft space proximate the alignment stent 702. The sealing stent 706 preferably is configured with a high radial force to promote efficacious sealing of the graft material 704 against a vessel wall. A body stent 708 configured here as a z-stent is disposed on the exterior of the graft material 704 and preferably is configured to provide longitudinal and circumferential stability/columnar support for the graft material of the device 700, such that it will conform to the vasculature and resist buckling when deployed in torturous anatomy such as the ascending thoracic aorta. A bare cannula stent 710 (such as, for example, a cut nitinol stent) is attached in the tubular graft material 704 at the opposite end from the alignment stent 702. This cannula stent 710 preferably is a conformable kink-resistant stent that provides distal sealing and migration-resistance. In a deployment of the device 700 to treat an aortic dissection, the alignment stent 702 preferably will be disposed proximal (nearer the heart) relative to the vessel tear, with the graft material traversing the tear in a manner generally sealing it from blood flow. And, the distal cannula stent 710 will help conform to the vasculature and retain a seal for treatment of the dissection. One or more of the sealing stent 706, body stent 708, and bare stent 710 may include one or more barbed projections configured to help anchor the device 700.
[0048] FIG. 20 shows a stent-graft device 800 configured for endovascular treatment of a thoracic aortic aneurysm. The device 800 includes a proximal component 802 and a distal component 822. The proximal component 802 has a tubular graft material 805 having proximal and distal ends 803a and 803b, and further may comprise a proximal stent 804 attached to the proximal end 803a of the graft material 805. The proximal stent 804 preferably is a non-symmetrical alignment stent provided in accordance with the stent 400 described above, such that the rounded points on the stent may protrude from the graft material and are less likely to perforate the artery wall than sharper points of a different stent configuration. In a deployment of the device 800 to treat a thoracic aortic aneurysm, the alignment stent 804 preferably will be disposed proximal (nearer the heart) relative to the aneurysm, with the graft material 805 traversing the aneurysm in a manner generally sealing it from blood flow.
[0049] The proximal component 802 further comprises a series of z-stents 810a-810f disposed distally from the proximal stem 804. A proximal z-stent 810a may be attached to the inner circumference of the graft material 805, other z-stents 810b-810e may be attached to the outer diameter of the graft material 805, and a distal z-stent 810f may be attached to the inner circumference of the graft material 805, as depicted in FIG. 20. However, it should be noted that some stents depicted on the inner circumference of the graft material 805 alternatively may be attached to the outer diameter of the graft material 805, and vice versa.
[0050] Moreover, in one embodiment, the proximal stent 804 extending from the graft material 805 may at least partially overlap with the most proximal z-stent 810a, as depicted in FIG. 20, The overlap may be range from about 1.0 mm to about 3.0 mm, and more preferably about 2.0 mm.
[0051] The distal component 822 has a graft material 825 having proximal and distal ends 823a and 823b, and a series of z-stents 830a-830g. In the example shown, the proximal three z-stents 830a-830c are attached to the inner circumference of the graft material 805, three other z-stents 830d-830f are attached to the outer diameter of the graft material 805, and a distal z-stent 830g is attached to the inner circumference of the graft material 805. However, like the proximal component 802, the z-stents of the distal component 822 may be attached to either the inner circumference or the outer diameter of the graft material 805.
[0052] The proximal end 823a of the graft material 825 may comprise one or more scallops 845. Preferably, a plurality of scallops 845 are provided that closely follow the shape of the proximal z-stent 830a, such that portions of the graft material 825 are cut out just proximal to the z-stent 830a, as shown in FIG. 20. In this manner, the plurality of scallops 845 may advantageously reduce the potential for graft infolding, thereby reducing or eliminating the likelihood of impeded blood flow and/or endoleaks around the distal component 822.
[0053] At the distal end 823b of the distal component 822, there is also a distally extending z-stent stent 834, which has a proximal end that may be fastened to the graft 825 using sutures, and then is exposed distal to the graft material, as shown in FIG. 20. The distally extending stent 834 has barbs 836 on some of its struts and the barbs 836 are directed proximally. The barbs 836 may be formed integrally with the stent 834, or formed externally and attached thereto, and reduce or prevent proximal migration of the distal end 823b of the distal component 822. Further, one or more radiopaque markers may be provided to facilitate correct positioning of the distal end of the distal component 822.
[0054] The proximal and distal components 802 and 822 may be introduced and deployed using separate deployment systems. In one embodiment, the proximal component 802 is introduced using a delivery system having a curved tip, while the distal component 822 is introduced using a delivery system having a straight tip.
[0055] When the stent graft device 800 of FIG. 20 is deployed to treat a thoracic aortic aneurysm, proximal and distal components 802 and 822 at least partially overlap with one another to provide a fluid passageway therebetween. The proximal end 823a of the distal component 822 may be deployed either inside the distal end 803b of the proximal component 802, although in other embodiments the proximal component 802 may be deployed inside of the distal component 822. This means that in deploying the stent graft assembly 800, either the proximal or distal components 802 and 822 may be deployed first and the other portion subsequently deployed depending upon the requirements in a particular case. In either case, it is preferable to have at least two stents overlap to facilitate sealing between the first and second portions. Also, by having an overlap of at least two stents, relative movement between the first and second portions is less likely to cause parting of the first and second portions when it is deployed and pulsating blood flow through the stent graft causes sideways movement.
[0056] Advantageously, when the stent graft device 800 of FIG. 20 is deployed, the proximal stent 804, which preferably is a non-symmetrical alignment stent, will maximize the efficacy of the proximal seal while reducing atraumatic contact with an artery wall, as noted above, and preventing distal migration of the proximal end of the stent graft. Further, as noted above, the plurality of scallops 845 may advantageously reduce the potential for graft infolding, thereby reducing or eliminating the likelihood of impeded blood flow and/or endoleaks around the distal component 822.
[0057] Stent examples of the present invention may be constructed of NiTi alloys or other materials presently known or yet to be developed, all within the scope of the present invention. The stents preferably are made from Nitinol wire and will therefore be MRI compatible. In another preferable embodiment, a stent may be made from a laser-cut Nitinol cannula, effectively rendering it a seamless or nearly-seamless wire-like construction. Nitinol's superelastic properties will facilitate the stents ability to be crimped down into a low profile delivery system.
[0058] While various embodiments of the invention have been described, the invention is not to be restricted except in light of the attached claims and their equivalents. Moreover, the advantages described herein are not necessarily the only advantages of the invention and it is not necessarily expected that every embodiment of the invention will achieve all of the advantages described.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.