Dyes For Analysis Of Protein Aggregation
Patton; Wayne Forrest ; et al.
U.S. patent application number 16/986311 was filed with the patent office on 2021-01-14 for dyes for analysis of protein aggregation. This patent application is currently assigned to Enzo Life Sciences, Inc.. The applicant listed for this patent is Enzo Life Sciences, Inc.. Invention is credited to Anatoliy Balanda, Jack Coleman, Lijun Dai, Vladyslava Kovalska, Mykhaylo Losytskyy, Anthony Ludlam, Praveen Pande, Wayne Forrest Patton, Dee Shen, Kateryna Volkova, Sergiy M. Yarmoluk.
| Application Number | 20210009809 16/986311 |
| Document ID | / |
| Family ID | 1000005109589 |
| Filed Date | 2021-01-14 |


View All Diagrams
| United States Patent Application | 20210009809 |
| Kind Code | A1 |
| Patton; Wayne Forrest ; et al. | January 14, 2021 |
DYES FOR ANALYSIS OF PROTEIN AGGREGATION
Abstract
Provided are dyes and compositions which are useful in a number of applications, such as the detection and monitoring protein aggregation, kinetic studies of protein aggregation, neurofibrillary plaques analysis, evaluation of protein formulation stability, and analysis of molecular chaperone activity.
| Inventors: | Patton; Wayne Forrest; (Dix Hills, NY) ; Yarmoluk; Sergiy M.; (Kyiv, UA) ; Pande; Praveen; (Holbrook, NY) ; Kovalska; Vladyslava; (Kyiv, UA) ; Dai; Lijun; (Farmingville, NY) ; Volkova; Kateryna; (Kyiv, UA) ; Coleman; Jack; (East Northport, NY) ; Losytskyy; Mykhaylo; (Kyiv, UA) ; Ludlam; Anthony; (Ypsilanti, MI) ; Balanda; Anatoliy; (Kyiv, UA) ; Shen; Dee; (Glen Head, NY) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Enzo Life Sciences, Inc. Farmingdale NY |
||||||||||
| Family ID: | 1000005109589 | ||||||||||
| Appl. No.: | 16/986311 | ||||||||||
| Filed: | August 6, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15963441 | Apr 26, 2018 | |||
| 16986311 | ||||
| 15156565 | May 17, 2016 | |||
| 15963441 | ||||
| 13510976 | Feb 12, 2013 | |||
| PCT/US10/03061 | Nov 30, 2010 | |||
| 15156565 | ||||
| 12592639 | Nov 30, 2009 | 9133343 | ||
| 13510976 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C09B 23/06 20130101; C09B 23/102 20130101; G01N 2800/2835 20130101; C09B 23/0008 20130101; G01N 33/582 20130101; C09B 23/0066 20130101; G01N 2800/2821 20130101; C09B 23/0058 20130101; G01N 33/6845 20130101; C09B 23/145 20130101; C09B 23/0025 20130101; C09B 23/141 20130101; C09B 23/04 20130101; C07D 401/12 20130101; G01N 2458/30 20130101 |
| International Class: | C09B 23/14 20060101 C09B023/14; C09B 23/01 20060101 C09B023/01; C09B 23/04 20060101 C09B023/04; C09B 23/06 20060101 C09B023/06; C09B 23/10 20060101 C09B023/10; G01N 33/58 20060101 G01N033/58; G01N 33/68 20060101 G01N033/68; C07D 401/12 20060101 C07D401/12 |
Claims
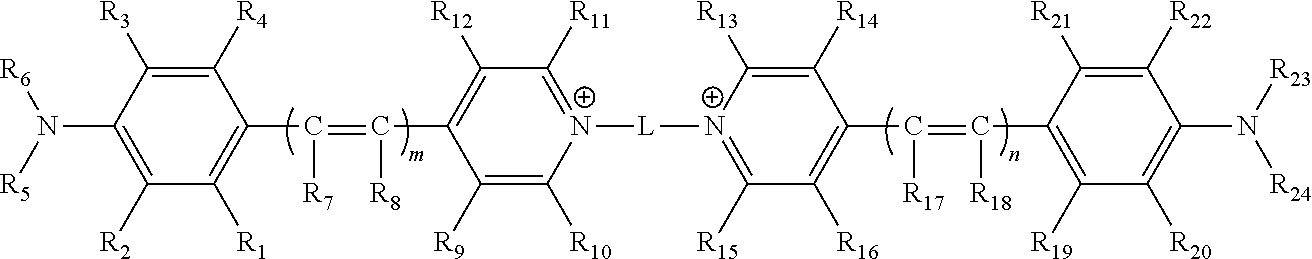
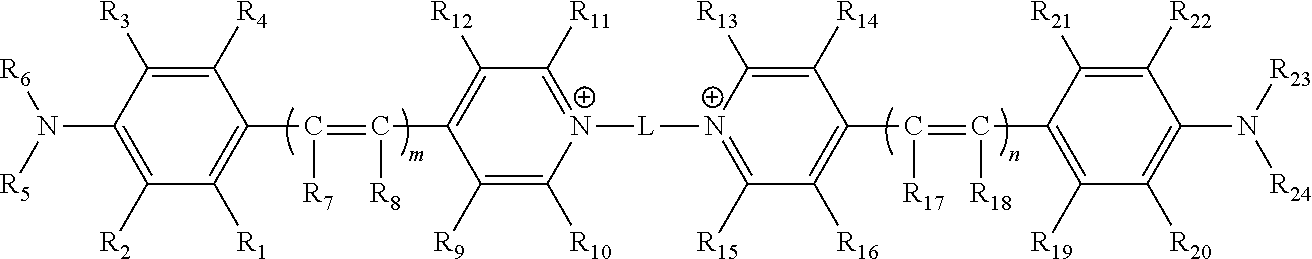
1. A compound comprising the structure ##STR00155## wherein m and n are independently 1, 2 or 3; wherein L is a linker arm comprising carbon, sulfur, oxygen, nitrogen, or any combination thereof; wherein R.sub.1, R.sub.2, R.sub.3, R.sub.4, R.sub.9, R.sub.10, R.sub.11, R.sub.12, R.sub.13, R.sub.14, R.sub.15, R.sub.16, R.sub.19, R.sub.20, R.sub.21 and R.sub.22 are independently hydrogen, halogen, amino, ammonium, nitro, sulfo, sulfonamide, carboxy, ester, cyano, phenyl, benzyl, an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, an alkoxy group wherein the alkoxy group is saturated or unsaturated, branched or linear, substituted or unsubstituted, or when taken in combination R.sub.1 and R.sub.2, or R.sub.3 and R.sub.4, or R.sub.9 and R.sub.10, or R.sub.11 and R.sub.12, or R.sub.13 and R.sub.14, or R.sub.15 and R.sub.16, or R.sub.19 and R.sub.20, or R.sub.21 and R.sub.22 form a five or six membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted, and wherein R.sub.9 and R.sub.10, or R.sub.11 and R.sub.12, or R.sub.13 and R.sub.14, or R.sub.15 and R.sub.16 can comprise alkyl chains that are joined together, wherein a quinoline moiety can be formed; wherein R.sub.7, R.sub.8, R.sub.17 and R.sub.18 are independently hydrogen, Z, an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, an alkoxy group wherein the alkoxy group is saturated or unsaturated, branched or linear, substituted or unsubstituted, or when taken together, R.sub.7 and R.sub.8 and R.sub.17 and R.sub.18, may form a 5 or 6 membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted; wherein Z comprises a carboxyl group (CO.sub.2.sup.-), a carbonate ester (COER.sub.25), a sulfonate (SO.sub.3.sup.-), a sulfonate ester (SO.sub.2ER.sub.25), a sulfoxide (SOR.sub.25), a sulfone (SO.sub.2CR.sub.25R.sub.26R.sub.27), a sulfonamide (SO2NR.sub.25R.sub.26), a phosphate (PO.sub.4.sup.=), a phosphate monoester (PO.sub.3.sup.-ER.sub.25), a phosphate diester (PO.sub.2ER.sub.25ER.sub.26), a phosphonate (PO.sub.3.sup.=) a phosphonate monoester (PO.sub.2.sup.-ER.sub.25) a phosphonate diester (POER.sub.25ER.sub.26), a thiophosphate (PSO.sub.3.sup.=), a thiophosphate monoester (PSO.sub.2.sup.-ER.sub.25) a thiophosphate diester (PSOER.sub.25ER.sub.26), a thiophosphonate (PSO.sub.2.sup.=), a thiophosphonate monoester (PSO.sup.-ER.sub.25) a thiophosphonate diester (PSER.sub.25ER.sub.26), a phosphonamide (PONR.sub.25R.sub.26NR.sub.28R.sub.29), its thioanalogue (PSNR.sub.25R.sub.26NR.sub.28R.sub.29), a phosphoramide (PONR.sub.25R.sub.26NR.sub.27NR.sub.28R.sub.29), its thioanalogue (PSNR.sub.25R.sub.26NR.sub.27NR.sub.28R.sub.29), a phosphoramidite (PO.sub.2R.sub.25NR.sub.28R.sub.29) or its thioanalogue (POSR.sub.25NR.sub.28R.sub.29) wherein E is independently O or S; wherein R.sub.25, R.sub.26, R.sub.27, R.sub.28, and R.sub.29 are independently a hydrogen, an unsubstituted straight-chain, branched or cyclic alkyl, alkenyl or alkynyl group, a substituted straight-chain, branched or cyclic alkyl, alkenyl or alkynyl group wherein one or more C, CH or CH.sub.2 groups are substituted with an O atom, N atom, S atom, or NH group, or an unsubstituted or substituted aromatic group; wherein Z is attached directly, or indirectly through a second linker arm comprising carbon, sulfur, oxygen, nitrogen, and any combinations thereof and wherein the second linker arm may be saturated or unsaturated, linear or branched, substituted or unsubstituted or any combinations thereof; wherein R.sub.5, R.sub.6, R.sub.23 and R.sub.24 can independently be hydrogen or an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, or when taken in combination R.sub.5 and R.sub.6 or R.sub.2 and R.sub.5 or R.sub.3 and R.sub.6 or R.sub.23 and R.sub.24 or R.sub.22 and R.sub.23 or R.sub.20 and R.sub.24 form a five or six membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted; and wherein said compound is modified to comprise a reactive group which is an isocyanate, isothiocyanate, monochlorotriazine, dichlorotriazine, 4,6,-dichloro-1,3,5-triazine, mono- or di-halogen substituted pyridine, mono- or di-halogen substituted diazine, maleimide, haloacetamide, aziridine, sulfonyl halide, acid halide, hydroxysuccinimide ester, hydroxysulfosuccinimide ester, imido ester, hydrazine, azidonitrophenol, azide, 3-(2-pyridyl dithio)-propionamide, glyoxal or aldehyde group.
2. The compound of claim 1, wherein the compound exhibits increased fluorescence in the presence of an aggregated form of a protein when compared to the fluorescence exhibited when the compound is in the presence of the unaggregated form of the protein.
3. The compound of claim 1, comprising the structure ##STR00156## modified to comprise the reactive group, or ##STR00157## modified to comprise the reactive group.
4. The compound of claim 4, wherein each of R.sub.5, R.sub.6, R.sub.23 and R.sub.24 are a methyl or an ethyl moiety.
5. The compound of claim 1, wherein the compound is S25, S43, TOL3, YAT2134, YAT2148, YAT2149, S13, YAT2135, YAT2324 or YAT2150, modified to comprise the reactive group.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a divisional of U.S. patent application Ser. No. 15/963,441 filed Apr. 26, 2018 which is a divisional of U.S. patent application Ser. No. 15/156,565, filed May 17, 2016, now abandoned, which is a divisional of U.S. patent application Ser. No. 13/510,976, filed Feb. 12, 2013, now abandoned, which is the U.S. national stage application of PCT/US2010/03061, filed Nov. 30, 2010, which claims priority to U.S. patent application Ser. No. 12/592,639, filed Nov. 30, 2009 (now U.S. Pat. No. 9,133,343), all of which are hereby incorporated by reference in its entirety.
SEQUENCE LISTING STATEMENT
[0002] This application contains a Sequence Listing which has been submitted in ASCII format via EFS-Web and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Aug. 5, 2020, is named ENZ-89-CIP-D1-D1-D1-SL.txt and is 955 bytes in size.
BACKGROUND OF THE INVENTION
(1) Field of the Invention
[0003] The present application generally relates to dyes and compositions comprising dyes. More particularly, provided are dyes and compositions for identifying and quantifying protein aggregation.
(2) Description of the Related Art
[0004] The deposition of insoluble protein aggregates, known as amyloid fibrils, in various tissues and organs is associated with a number of neurodegenerative diseases, including Alzheimer's, Huntington's and Parkinson's diseases, senile systemic amyloidosis and spongiform encephalopathies (Volkova et al., 2007; Stefani & Dobson, 2003). Fibrillar deposits with characteristics of amyloid are also formed by several other proteins unrelated to disease, including the whey protein beta-lactoglobulin (BLG). All amyloid fibers, independent of the protein from which they were formed, have very similar morphology: long and unbranched, a few nanometers in diameter, and they all exhibit a cross-beta X-ray diffraction pattern. The ability to form amyloid fibrils of structurally and functionally diverse proteins, some of which are not associated with amyloid-deposition diseases, suggests that this property is common to all polypeptides Such amyloid structures are also known to possess a binding affinity for certain dyes, notably, thioflavin T and congo red dyes.
[0005] Many proteins are known to be only marginally stable in solution, undergoing conformational changes due to various stresses during purification, processing and storage (Arakawa et al., 2007). Such stresses may include elevated temperature, agitation and exposure to extremes of pH, ionic strength, or various interfaces (e.g., an air-liquid interface) and high protein concentration (as observed for some monoclonal antibody formulations). A wide variety of aggregates are encountered in biopharmaceutical samples, which range in size and physiochemical characteristics (e.g., solubility, reversibility). Protein aggregates span a broad size range, from small oligomers that are only a couple nanometers in length to insoluble micron-sized aggregates that extend to millions of monomeric units. Structurally altered proteins have an especially strong tendency to aggregate, often leading to their eventual precipitation. Irreversible aggregation is a major problem for the long-term storage and stability of therapeutic proteins and for their shipment and handling.
Mechanisms of Protein Aggregation
[0006] Aggregation is a major degradation pathway that needs to be characterized and controlled during the development of protein pharmaceuticals. In the bioprocessing arena, the mechanisms of protein aggregation are still not fully understood, despite the fact that aggregation is a major problem in therapeutic protein development (Arakawa et al., 2006). One plausible mechanism is that aggregation is driven or catalyzed by the presence of a small amount of a contaminant which serves as a nucleation site. That contaminant could be a damaged form of the protein product itself, host cell proteins, or even nonprotein materials, such as leachates from the container or resin particles associated with purification of the protein.
[0007] If the contaminant is the damaged protein itself, then its aggregation may lead to soluble oligomers, which become larger aggregates, visible particulates, or insoluble precipitates. Such soluble oligomers, host-cell contaminants, or nonprotein materials may serve as a nucleus onto which native proteins assemble and are incorporated into larger aggregates. Damaged forms of a protein product can also arise from chemical modification (such as oxidation or deamidation) and from conformationally damaged forms arising from thermal stress, shear, or surface-induced denaturation. Minimizing protein aggregation thus requires ensuring both chemical and physical homogeneity; that is, chemically modified or conformationally altered proteins must be removed from the final product.
[0008] A second mechanism that often leads to protein aggregation is initiated by the partial unfolding of the native protein during its storage. Protein conformation is not rigid--the structure fluctuates around the time-averaged native structure to different extents depending upon environmental conditions. Some partially or fully unfolded protein molecules are always present at equilibrium in all protein solutions, but most such molecules simply refold to their native structure. These unfolded proteins may in some instances, however, aggregate with other such molecules or may be incorporated into an existing aggregate nucleus, eventually forming larger aggregates, as described above. Factors such as elevated temperature, shaking (shear and air-liquid interface stress), surface adsorption, and other physical or chemical stresses may facilitate partial unfolding of proteins, leading to the cascade of events that cause aggregation.
[0009] A third aggregation mechanism is reversible self-association of the native protein to form oligomers. According to the law of mass action, the content of such reversible aggregates will change with total protein concentration. The tendency of different proteins to associate reversibly with one another is highly variable, and the strength of that association typically varies significantly with solvent conditions, such as pH and ionic strength. In principle, these reversible oligomers will dissociate completely as the protein becomes highly diluted, for example, after delivery of a therapeutic protein in vivo. Consequently, this class of aggregates is generally less of a concern than irreversible aggregates. Such reversible oligomers can eventually become irreversible aggregates, however. Preventing accumulation of irreversible aggregates may thus require minimizing the reversible association as well. Further, reversible self-association of proteins can significantly alter overall pharmaceutical properties of product solutions, such as solution viscosity.
[0010] Detection of reversible aggregates can be an especially challenging task. As such, aggregates can dissociate after their dilution during attempts to measure them. Additionally, the results of any analysis method incorporating a separation process in the workflow may depend very much upon the kinetic rates of the reversible association-dissociation reactions as well as the equilibrium constants.
[0011] One consequence of the complexities of monitoring aggregate formation processes is the difficulty of linking the effect (presence of aggregates) to its underlying cause, particularly because the key damage may occur at a time or place quite separated from the observed consequence. One example arises during the large-scale production of therapeutic monoclonal antibodies (MAbs). Acid stability plays a major role in the aggregation of MAbs because the process for their purification usually involves both low-pH elution from protein-A affinity columns and acid-treatment for viral inactivation.
[0012] The exposure of MAbs to a low-pH environment can result in small but significant conformational changes that can additionally depend upon factors such as temperature, and solvent composition. While such partially unfolded MAbs may not aggregate at low pH, they may aggregate during subsequent manufacturing steps involving changes in pH or ionic strength. A larger conformational change at low pH generally leads to more aggregates upon increasing the pH. Typically, protein aggregate formation from the low-pH structure is not a fast process, but it does occur slowly from the association of damaged monomers that have not returned to their fully native structure. This and other types of protein aggregation phenomena may not manifest themselves until months after manufacturing a particular lot of protein or until later stages of the product development process. Regardless of the mechanism of aggregation, preventing aggregation problems requires sensitive and reliable technologies for quantitative determination of aggregate content and aggregate characteristics.
[0013] Since the earliest clinical applications of protein pharmaceuticals in medicine, aggregation problems have been implicated in adverse reactions in humans and other safety issues. In order to minimize such risks from therapeutic proteins in the clinic, formulations must be optimized to minimize aggregation during storage, handling, and shipping.
Analysis of Protein Aggregation
[0014] The analysis of protein aggregation can be formally classified into four experimental types (Arakawa et al., 2006, 2007; Krishnamurthy et al., 2008). The first type of protein aggregation analysis is the most conventional approach, wherein a small volume of sample is applied to a separation medium and forms a band or zone. As the band migrates through the medium, the proteins separate according to differences in size, electrophoretic charge, or mass. Gel electrophoresis, size exclusion chromatography (SEC), field flow fractionation (FFF), and the occasionally used band sedimentation technique belong to this class of methods. The movement of the band or zone in these methods is often monitored using absorbance or refractive index detection.
[0015] In the second type of analysis, the sample initially and uniformly fills a measurement cell. When an electrical or centrifugal driving force is then applied, the protein moves along the applied field, leaving a protein-depleted solvent, which creates a boundary between protein-free and protein-containing solution phases. The movement of this boundary over time is measured. This mode of separation is used in analytical ultracentrifugation-sedimentation velocity (AUC-SV) and moving-boundary electrophoresis.
[0016] The third type of analysis is a measurement of particle size with no physical separation. An example of this method is referred to as correlation spectroscopy and it measures the fluctuation of particles in solution due to Brownian motion (i.e., measures protein diffusion coefficients). Fluctuations of scattered light and of fluorescence intensity have been employed in this type of measurement. One of the most widely employed methods in this category is referred to as dynamic light scattering (DLS).
[0017] SEC is the most commonly implemented control method and has become an industry benchmark for quantification of protein aggregates. SEC is seen as a versatile technique for separation and quantification of protein aggregates because of its high precision, high throughput, ease of use, compatibility with a quality control (QC) environment, and in most cases ability to accurately quantify protein aggregates. In spite of these strengths, several concerns exist with the technique including: a potential loss of aggregates (especially multimers), interaction of samples with a column matrix, the required change of a sample buffer matrix to an SEC mobile phase, and the inherent requirement for dilution of samples. Additionally, perturbation of the distribution of protein aggregates under standard SEC methodological conditions is possible.
[0018] AUC-SV relies on hydrodynamic separation of various species in a heterogeneous protein mixture under strong centrifugal force. AUC-SV complements SEC in resolving and quantifying low levels of protein aggregates. The main advantages of AUC-SV are seen in its ability to detect and measure higher order aggregates (which may elute in the void volume of an SEC column) and to conduct these measurements without exposing samples to a column resin or SEC mobile phase. AUC-SV is considered an accurate method because it does not require standards or dissociate aggregates; thus it can be used as an orthogonal method to verify the accuracy of SEC results. AUC-SV suffers from lower precision than SEC, however. The practical aspects of AUC-SV that impact precision and accuracy are beginning to be understood better, and several recent studies have demonstrated the utility of AUC-SV to detect and quantify aggregates present at relatively low (.about.1%) levels. Despite its advantages, AUC-SV is not yet readily amenable for use as a routine release test in the biotechnology industry because of issues related to low throughput, the need for specialized equipment, performance problems at high protein concentrations, the need for skilled practitioners of the method, and difficulty in validating data analysis software.
[0019] DLS uses the time-dependent fluctuations of a scattered-light signal to calculate the hydrodynamic diameter of protein aggregates and their relative proportions. This method is highly sensitive to large aggregates because the intensity of scattered light increases proportionally with molecular weight. As a result, very large aggregates (e.g., a 1,000-mer) present at trace levels (.ltoreq.0.1%) can be detected with high sensitivity. If present, such aggregates would elute in the void volume of an SEC column or they may be filtered out. Although this method is ideal for detecting very low mass fractions of large aggregates, it cannot resolve species that are similar in size. At least a three- to five-fold difference in hydrodynamic diameter is required for resolving different species. DLS is also not amenable to use as a control method because it is semi-quantitative and very sensitive to dust or other extraneous particles. Results also depend on the algorithm used for data analysis, which is often proprietary to the manufacturer of a particular instrument.
[0020] As an orthogonal technique to SEC and AUC-SV, analytical field-flow fractionation (aFFF) has gained popularity in recent years for its ability to fractionate protein aggregates without a column. aFFF most commonly uses two fluid flows ("fields") in a channel to achieve particle separation based upon molecular weight and hydrodynamic size (diffusion coefficient). Injected macromolecular species are held in place by a cross flow on a semi-permeable membrane while a perpendicular channel flow carries molecules forward based on their diffusion coefficient, thereby providing size-based fractionation. Because aFFF involves no column interactions, it is considered a gentler separation technique than SEC. Concerns regarding the interaction of aggregates with the membrane have yet to be completely addressed, however. aFFF can be coupled with different detectors including light scattering, refractive index, and ultraviolet (UV) detectors. When compared with SEC, the precision and limit of detection of aFFF is inferior in the high-molecular-weight range, because of increased baseline noise. Experimental conditions (e.g., cross-flow rate) for reasonable separations in one size range are also not generally applicable to other size ranges, making the technique cumbersome, especially when analyzing a broad range of masses. Along with other limitations, such as the need for specialized equipment and a skilled operator, and the difficulty in validating the method prevents the use of aFFF in applications for release and stability monitoring.
[0021] Resolution and the size range that can be evaluated in one particular analysis vary widely among the above mentioned techniques. SEC cannot handle a large range of sizes because the pore size or degree of polymerization of the resin must be adjusted to the size of the protein species. If a protein sample contains widely different sizes, many techniques are unsuitable for analyzing all sizes simultaneously. FFF and DLS can cover a very large range of sizes, but in the case of DLS, resolution is generally fairly poor, and FFF entails some trade-off between resolution and dynamic range. SV-AUC is intermediate in capability relative to FFF and DLS. The dynamic range of SV-AUC is fairly good, generally a factor of 100 or more in molecular weight at any particular rotor speed. The resolution of SV-AUC is generally not ideal for separating monomer from dimer, compared with the best SEC columns (especially for lower molecular weight proteins). SV-AUC is often much better, however, than SEC for resolving moderate size oligomers, (tetramers to decamers).
[0022] The cited analytical techniques also differ significantly with respect to their overall sensitivity, in other words, their ability to detect and quantify small percentages of irreversible aggregates. SEC, FFF, and SV-AUC are all capable of detecting aggregates at levels as low as .about.0.1% when they are well separated from other species. The quantification of species that elute from SEC or FFF is quite good, but aggregates can easily be lost during the separation process. Thus, SEC and FFF may provide good precision but poor accuracy. For SV-AUC, loss of protein aggregates to surfaces is usually not a problem, but accurate quantification of small oligomers (dimer-tetramer) at total levels of .about.2% or less is quite difficult.
[0023] The sensitivity of DLS increases linearly with the stoichiometry of the protein aggregate. DLS is for all practical purposes useless for detecting oligomers smaller than an octamer, because the technique cannot resolve such oligomers from monomeric species, and for those protein aggregate species that are resolved, the accuracy of the weight fractions is quite poor, typically plus or minus factors of two to ten. DLS exhibits excellent sensitivity, however, for very large aggregate species, which can often be detected at levels far below 0.01% by weight.
[0024] Overall, no single analytical technique is ideal for every protein or is optimal for analyzing the wide range of aggregation problems that can arise with protein pharmaceutical formulation. One important industry trend are recent requests from regulatory agencies that the protein aggregation analytical method used for lot release and/or formulation development. Typically, this means SEC which is cross-checked through one or more orthogonal approaches to ensure detection of all relevant protein aggregate species. Comparison of protein aggregate content using various technologies is thus an emerging topic of interest in biotechnology research.
Fluorescent Dyes and Protein Aggregation
[0025] In a fourth method of aggregate analysis, fluorescent dyes have been used to stain amyloidogenic material in histology, while insights into the prerequisites and kinetics of amyloid formation have been obtained by the in vitro analysis of this process using similar dyes (Volkova et al., 2007, 2008; 2009; Demeule et al., 2007). The fluorescent probes, thioflavin T and Congo red, have been the most frequently used dyes to detect the presence of amyloid deposits. Both the benzothiazole dye thioflavin T and the symmetrical sulfonated azo dye congo red have been adapted to study the formation of amyloid fibrils in solution using the fluorescence properties of these molecules. The amyloid aggregates cause large enhancements in fluorescence of the dye thioflavin T, exhibit green-gold birefringence upon binding the dye congo red, and cause a red-shift in the absorbance spectrum of congo red. Amyloid fibril detection assays have suffered from several drawbacks, however, when using thioflavin T, Congo red and their derivatives. For instance, congo red can bind to native .alpha.-proteins such as citrate synthase and interleukin-2 (Khurana et al., 2001). As a consequence of its poor optical properties, the congo red derivative chrysamine-G only weakly stains neuritic plaques and cerebrovascular amyloid in postmortem tissue (Klunk et al., 1998). Furthermore, the binding of dyes can influence the stability of amyloid aggregates, and the interplay with other components (for example, during testing of potential amyloid inhibitors) is unpredictable (Murakami et al., 2003). Importantly, there exists a great variability among the different amyloid fibrils in their ability to bind congo red and thioflavin T. Fluorescence intensity using thioflavin T can vary depending upon the structure and morphology of the amyloid fibrils (Murakami et al., 2003). Despite the widespread use of thioflavin T, its application to amyloid quantification often generates inconsistent and inaccurate results. Variations in spectral properties caused by buffer conditions and protein-dye ratios result in poor reproducibility, complicating the use of thioflavin T for quantitative assessment of fibril formation. In the absence of other more reliable assays, investigators have relied heavily upon thioflavin T as a reporter probe for amyloid protein aggregation. A reliable method for amyloid quantification likely would be useful not only for detecting mature amyloid fibrils, but also for monitoring the kinetics of fibrillogenesis, which is essential for better understanding of the underlying biophysics and mechanism of the protein aggregation process. Furthermore, such an assay would be a tool for discovery and development of therapeutic compounds capable of blocking protein aggregation.
[0026] Thus the design of new dyes which can selectively interact with fibrillar amyloidogenic proteins is of substantial importance for basic research, and has a crucial practical significance for biotechnology and medicine. Dialkylamino-substituted monomethine cyanine T-284 and meso-ethyl-substituted trimethine cyanine SH-516 have demonstrated higher emission intensity and selectivity to aggregated .alpha.-synuclein (ASN) than the classic amyloid stain thioflavin T; while the trimethinecyanines T-49 and SH-516 exhibit specifically increased fluorescence in the presence of fibrillar .beta.-lactoglobulin (BLG) (Volkova et al., 2007). These dyes demonstrated the same or higher emission intensity and selectivity to aggregated BLG as thioflavin T. Recently, nile red dye has been used to detect antibody A aggregate, but it did not stain all types of protein aggregates, underscoring the need to several analytical methods in order to assess protein aggregation (Demeule et al., 2007).
Optimization of Protein Formulations
[0027] Another potential application of a fluorescence based protein aggregate detection technique relates to pharmaceutical protein formulations (U.S. Pat. Nos. 6,737,401; 5,192,737; 6,685,940; US Patent Application Publication 2008/0125361 A1). The physical stability of pharmaceutical protein formulations is of great importance because there is always a time delay between production, protein formulation and its subsequent delivery to a patient. The physical stability of a protein formulation becomes even more critical when using drug delivery devices to dispense the protein formulation, such as infusion pumps and the like. When the delivery device is worn close to the body or implanted within the body, a patient's own body heat and body motion, plus turbulence generated in the delivery tubing and pump, impart a high level of thermo-mechanical stress to a protein formulation. In addition, infusion delivery devices expose the protein to hydrophobic interfaces in the delivery syringes and catheters. These interfacial interactions tend to destabilize the protein formulation by inducing denaturation of the native structure of the protein at these hydrophobic interfaces.
[0028] In an optimized protein formulation, the protein should remain stable for several years, maintaining the active conformation, even under unfavorable conditions that may occur during transport or storage. Protein formulation screening needs to be performed before the assessment of safety, toxicity, ADME (absorption distribution metabolism excretion), pharmacology and the testing of biological activity in animals. Currently, protein formulation in the pharmaceutical industry is generally a slow process and would benefit from fast formulation screening approaches that do not require overly complicated instrumentation techniques.
[0029] The formulation of protein drugs is a difficult and time-consuming process, mainly due to the structural complexity of proteins and the very specific physical and chemical properties they possess. Most protein formulations contain excipients which are added to stabilize protein structure, such as a particular buffer system, isotonic substances, metal ions, preservatives and one or more surfactants, with various concentration ranges to be tested. The conventional analytical methods usually require a long period of time to perform, typically twenty or more days, as well as manual intervention during this period. The development of new formulations is costly in terms of time and resources. Moreover, even for a known protein formulation, batch to batch quality control analysis is often less than optimal using the current state of the art methods. Therefore, a versatile, reliable, rapid and resource-efficient analytical method is desired for both developing novel protein formulations and identifying protein stability in quality control procedures. The ideal analytical method would be sensitive, accurate, and linear over a broad range, resistant to sample-matrix interference, capable of measuring all possible structural variants of a protein, and compatible with high throughput screening.
[0030] A high throughput screening (HTS) platform for optimization of protein formulation has been proposed based upon the use of multi-well microplates (Capelle Martinus et al., 2009). Basically, such an HTS platform was envisioned to consist of two components: (i) sample preparation and (ii) sample analysis. Sample preparation involves automated systems for dispensing the drug and the formulation ingredients in both liquid and powder form. The sample analysis involves specific methods developed for each protein to investigate physical and chemical properties of the formulations in the microplates.
[0031] The techniques that could be coupled with such an HTS platform include UV-Visible absorbance/turbidity, light scatter, fluorescence intensity, resonance energy transfer, fluorescence anisotropy, Raman spectroscopy, circular dichroism, Fourier transform infrared spectroscopy (FTIR), surface plasmon resonance and fluorescence lifetime. Ideally, however, the analysis technique should be specific, quantitative, robust, cost-effective, easily accessed, easy to use and informative. Capelle Martinus et al. (2009) utilized several assays coupled with HTS to optimize a salmon calcitonin formulation: turbidity (absorbance at 350 nm), intrinsic tyrosine fluorescence, 1-anilino-naphthalene-8-sulfonate (ANS) fluorescence and Nile red fluorescence. Addition of the dyes (Nile red and ANS) were employed to examine protein conformational changes. Their findings were in accordance with the salmon calcitonin formulations that were patented and used commercially, lending credence to the concept that fluorescent probe-based approaches can be employed in protein formulation optimization activities. The use of several complementary analytical methods permits the selection of formulations using carefully designed assay criteria. The investigators found that in some cases, an increase in turbidity was observed without an increase in ANS or Nile red fluorescence. In other formulations, an increase in fluorescence was detected without an increase in turbidity. This suggests that these dyes are not necessarily measuring the exact same biophysical phenomenon as the turbidity measurements. Measuring the fluorescence of at least two dyes in combination with turbidity and intrinsic fluorescence was, therefore, recommended.
[0032] Among these techniques, fluorescence detection from externally added dyes, which enhances fluorescence intensity upon interacting with misfolded or aggregated protein, is most attractive, because this technique requires minimum protein concentration due to its high sensitivity and simple implementation on a microplate reader.
[0033] Real time stability testing of a particular formulation may demonstrate no immediately apparent effect on physical or chemical stability. Accelerated stability testing can help, therefore, in facilitating the determination of the most suitable excipients and concentrations. Storage at different target temperatures (0-50.degree. C.), illumination of samples, mechanical stress (i.e., agitation that simulates handling and transportation), multiple freeze-thaw cycles (mimicking frozen storage, freeze drying), oxygen purging, increased humidity and seeding are different ways to accelerate protein degradation.
[0034] High throughput spectroscopy is a fast and versatile method for initial screening of the physical stability of protein formulations. The microplate well-based platform could be enhanced with accelerated stress testing and methods to determine chemical stability, e.g., electrophoresis, HPLC, mass spectrometry. For instance, thioflavin T has been used to select and optimize FDA-approved surfactant(s) in insulin formulations using magnetically stirring to accelerate insulin aggregation (U.S. Pat. No. 6,737,401).
Thermal Shift Assay
[0035] Fluorescent dyes have been used to monitor protein stability by systematically varying the temperature of test samples, also known as the Thermofluor.RTM. technique (U.S. Pat. No. 6,020,141; Matulis et al., 2005; Mezzasalma et al., 2007; Volkova et al., 2008; Ericsson et al., 2006; Todd et al., 2005). Protein stability can be altered by various additives including but not limited to excipients, salts, buffers, co-solvents, metal ions, preservatives, surfactants, and ligands. Protein stability can be shifted by various stresses, including elevated temperature, referred to as thermal shift, or chemical denaturants, such as urea, guanidine isocyanate or similar agents. A protein stability shift assay offers a wide spectrum of applications in the investigation of protein refolding conditions, optimization of recombinant protein expression/purification conditions, protein crystallization conditions, selection of ligand/drug/vaccine/diagnostic reagents and protein formulations.
[0036] The classic thermal shift technology utilizes the dye SYPRO.RTM. Orange and involves the use of a melting point device to raise the temperature stepwise (Raibekas, 2008). Thermal shift technology is coupled with aggregation detection technologies, such as light scattering technology or internal fluorescence from protein (such as tyrosine or tryptophan) to monitor protein aggregation and unfolding respectively. This type of technology usually requires a high protein concentration, therefore, it is not cost-effective. In addition, thermal shift technology cannot work effectively on formulations with low protein concentrations or finalize protein formulations which require a very low detection limit (typically .about.1-5% protein aggregates).
Fluorometric Screening Assay for Protein Disulfide Isomerase (PDI)
[0037] Protein disulfide isomerase (PDI, EC5.3.4.1) is a 57-kDa enzyme expressed at high levels in the endoplasmic reticulum (ER) of eukaryotic cells (Ferrari and Soling, 1999). PDI was the first enzyme known to possess the disulfide isomerase activity and has been well characterized over the past three decades. In ER, PDI catalyzes both the oxidation and isomerization of disulfides of nascent polypeptides. Under the reducing condition of the cytoplasm, endosomes and cell surface, PDI catalyzes the reduction of protein disulfide bonds.
[0038] Folding catalysts such as PDI and peptidylprolyl isomerase accelerate slow chemical steps that accompany folding. Disulfide bond formation can occur quite rapidly, even before the completion of synthesis, but for some proteins disulfide bond formation is delayed and occurs post-translationally. PDI catalyzes disulfide formation and rearrangement by thiol/disulfide exchange during protein folding in the ER. As a member of the thioredoxin superfamily, which also includes homologs such as ERp57, PDIp, ERp72, PDIr and ERp5, PDI has two independent but non-equivalent active sites, with one positioned close to the C-terminus and another close to the N-terminus. Each site possesses two cysteine residues (CGHC) that cycle between the dithiol and disulfide oxidation states. The disulfide bond at the active site of PDI is a good oxidant that directly introduces a disulfide bond into protein substrates. The dithiol redox state is essential for catalyzing disulfide rearrangements. The necessity of having oxidized and reduced active sites for catalysis of different steps results in a redox optimum. Besides its major role in the processing and maturation of secretory proteins in ER, PDI and its homologs have been implicated in other important cellular processes. For example, cellular insulin degradation occurs in a sequential fashion with several identified steps. The initial degradative step occurs in endosomes with two or more cleavages in the B chain occurring. This is followed by reduction of disulfide bonds by PDI, or a related enzyme, generating an intact A chain and fragments of B chain. The insulin fragments are further cleaved by multiple proteolytic systems, such as the lysosomal degradation pathway.
[0039] PDI and its homologs also play roles in the processing and maturation of various secretory and cell surface proteins in the ER following their synthesis. Several in vitro studies have also suggested a chaperone function of PDI, to assist in protein folding or refolding. During ER stress, as for example during hypoxia in endothelial cells and astrocytes in the cerebral cortex, PDI is up-regulated. This indicates that PDI is involved in protecting cells under pathological or stressful conditions.
[0040] Besides ER, PDI also exists on many cell surfaces, such as endothelial cells, platelets, lymphocytes, hepatocytes, pancreatic cells and fibroblasts. For the reductive activity of plasma membrane, PDI is required for endocytosis of certain exogenous macromolecules. The cytotoxicity of diphtheria toxin is blocked by PDI inhibitors, which block the cleavage of the inter-chain disulfide bonds in the toxin. PDI also mediates reduction of disulfide bonds in human immunodeficiency virus envelope glycoprotein 120, which is essential for infectivity. PDI inhibitors can thus prevent virus entry into cells. Such functional activities make PDI and its homologs attractive drug targets.
[0041] Biochemical assays related to measuring PDI activity have been described:
[0042] (1) ScRNase assay: PDI converts scrambled (inactive) RNase into native (active) RNase that further acts on its substrate. The reported sensitivity of the assay is in the micromolar range (Lyles & Gilbert, 1991).
[0043] (2) The Insulin Turbidity Assay: PDI breaks the two disulfide bonds between the two insulin chains (A and B) that results in precipitation of the B chain. This precipitation can be monitored by measuring turbidity (absorbance at 620 nm), which in turn indicates PDI activity. Sensitivity of this assay is in the micromolar range (Lundstrom & Holmgren, 1990). Recently an end-point, high throughput screening assay of PDI isomerase activity based on enzyme-catalyzed reduction of insulin in the presence of dithiothreitol using hydrogen peroxide as a stop reagent has been developed (Smith et al., 2004; U.S. Pat. No. 6,977,142).
[0044] (3) The Di-E-GSSG assay: This is the fluorometric assay that can detect picomolar quantities of PDI and is, therefore, considered the most sensitive assay to date for detecting PDI activity. Di-E-GSSG has two eosin molecules attached to oxidized glutathione (GSSG). The proximity of eosin molecules leads to the quenching of its fluorescence. Upon breakage of the disulfide bond by PDI, however, fluorescence increases 70 fold (Raturi & Mutus 2007). Certain common excipients can cause signal generation as well, such as 2-mercaptoethanol and dithiothreitol.
[0045] In view of the important functional activities of PDI and homologous enzymes, sensitive, real-time, high throughput methods that are time and cost-effective are highly desirable.
Chaperone/Anti-Chaperone Activity
[0046] A chaperone is a protein that can assist unfolded or incorrectly folded proteins to attain their native state by providing a microenvironment in which losses due to competing folding and aggregation reactions are reduced (Puig & Gilbert, 1994). Chaperones also mediate the reversibility of pathways leading to incorrectly folded structures. One of the major complications encountered in both in vitro and in vivo protein folding is aggregation resulting from the commonly encountered low solubility of the unfolded protein or different folding intermediates. The efficiency of folding depends upon how the unfolded protein partitions between pathways leading to aggregation and pathways leading to the native structure. In vivo, the partitioning between productive and non-productive folding pathways may be influenced by "foldases" and molecular chaperones. Foldases accelerate folding by catalyzing the slow chemical steps, such as disulfide bond formation and proline isomerization that may retard folding. Molecular chaperones do not appreciably accelerate folding but bind to nonnative proteins in a way that is thought to inhibit non-productive aggregation and misfolding. In order to prevent these improper interactions, chaperones must be present at concentrations that are stoichiometric with the newly synthesized proteins. Consequently, chaperones are often found at very high concentrations in the cell.
[0047] PDI is a very abundant protein within cells. Although primarily classified as a foldase, PDI has also been shown to possess chaperone or anti-chaperone activity (Puig & Gilbert, 1994). PDI accelerates lysozyme folding, and at high concentration, it displays a chaperone-like activity that prevents lysozyme misfolding and aggregation. In addition, PDI also exhibits an unusual "anti-chaperone" activity. Under conditions that favor lysozyme aggregation, low concentrations of PDI greatly reduce the yield of native lysozyme and facilitate the formation of aggregates that are extensively cross-linked by intermolecular disulfide bonds. Similarly, PDI breaks the two disulfide bonds between two insulin chains (A and B) that results in precipitation of The B chain, thus serving as an "anti-chaperone in this case." (Lundstrom & Holmgren. 1990.
[0048] Alpha-crystallin, a major protein component of the mammalian lens of the eye, belongs to the heat shock protein (Hsp) family and acts as a molecular chaperone by preventing aggregation of target proteins (e.g. beta and gama-crystallins) under stress conditions through the formation of stable, soluble high-molecular mass complexes with them. Aggregation of BLG (beta-lactoglobulin) occurs mainly via intermolecular disulfide bond exchange. Upon heating, BLG aggregates, which can be accelerated by subjecting the protein to either an elevated pH or through the additional of DTT. .alpha.-crystallin prevents heat-induced BLG aggregation, acting as a chaperone in the absence of DTT; in the presence of DTT, however, this chaperone activity is less efficient due to faster aggregation of heated and reduced beta-lactoglobulin. Another Hsp protein, Hsp 27, protects myosin 51 from heat-induced aggregation, but not from thermal denaturation and ATPase inactivation.
[0049] Highly sensitive fluorescent probes useful to monitoring various protein functions relating to aggregation should assist in formulation optimization. Preferably, these probes should be applicable to a broad ranges of proteins and concentrations even in the presence of excipients, salts and buffers, providing sensitive limits of detection and excellent linear dynamic ranges.
BRIEF SUMMARY OF THE INVENTION
[0050] The present invention provides dyes, reagents and methods useful for detection of protein aggregates.
[0051] In some embodiments, a compound is provided. The compound comprises the structure
##STR00001##
[0052] wherein m and n are independently 1, 2 or 3;
[0053] wherein L is a linker arm comprising carbon, sulfur, oxygen, nitrogen, or any combination thereof;
[0054] wherein R.sub.1, R.sub.2, R.sub.3, R.sub.4, R.sub.9, R.sub.10, R.sub.11, R.sub.12, R.sub.13, R.sub.14, R.sub.15, R.sub.16, R.sub.19, R.sub.20, R.sub.21 and R.sub.22 are independently hydrogen, halogen, amino, ammonium, nitro, sulfo, sulfonamide, carboxy, ester, cyano, phenyl, benzyl, an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, an alkoxy group wherein the alkoxy group is saturated or unsaturated, branched or linear, substituted or unsubstituted, or when taken in combination R.sub.1 and R.sub.2, or R.sub.3 and R.sub.4, or R.sub.9 and R.sub.10, or R.sub.11 and R.sub.12, or R.sub.13 and R.sub.14, or R.sub.15 and R.sub.16, or R.sub.19 and R.sub.20, or R.sub.21 and R.sub.22 form a five or six membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted, and wherein R.sub.9 and R.sub.10, or R.sub.11 and R.sub.12, or R.sub.13 and R.sub.14, or R.sub.15 and R.sub.16 can comprise alkyl chains that are joined together, wherein a quinoline moiety can be formed;
[0055] wherein R.sub.7, R.sub.8, R.sub.17 and R.sub.18 are independently hydrogen, Z, an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, an alkoxy group wherein the alkoxy group is saturated or unsaturated, branched or linear, substituted or unsubstituted, or when taken together, R.sub.7 and R.sub.8 and R.sub.17 and R.sub.18, may form a 5 or 6 membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted; [0056] wherein Z comprises a carboxyl group (CO.sub.2.sup.-), a carbonate ester (COER.sub.25), a sulfonate (SO.sub.3.sup.-), a sulfonate ester (SO.sub.2ER.sub.25), a sulfoxide (SOR.sub.25), a sulfone (SO.sub.2CR.sub.25R.sub.26R.sub.27), a sulfonamide (SO2NR.sub.25R.sub.26), a phosphate (PO.sub.4.sup.=), a phosphate monoester (PO.sub.3.sup.-ER.sub.25), a phosphate diester (PO.sub.2ER.sub.25ER.sub.26), a phosphonate (PO.sub.3.sup.=) a phosphonate monoester (PO.sub.2.sup.-ER.sub.25) a phosphonate diester (POER.sub.25ER.sub.26), a thiophosphate (PSO.sub.3.sup.=), a thiophosphate monoester (PSO.sub.2.sup.-ER.sub.25) a thiophosphate diester (PSOER.sub.25ER.sub.26), a thiophosphonate (PSO.sub.2.sup.=), a thiophosphonate monoester (PSO.sup.-ER.sub.25) a thiophosphonate diester (PSER.sub.25ER.sub.26), a phosphonamide (PONR.sub.25R.sub.26NR.sub.28R.sub.29), its thioanalogue (PSNR.sub.25R.sub.26NR.sub.28R.sub.29), a phosphoramide (PONR.sub.25R.sub.26NR.sub.27NR.sub.28R.sub.29), its thioanalogue (PSNR.sub.25R.sub.26NR.sub.27NR.sub.28R.sub.29), a phosphoramidite (PO.sub.2R.sub.25NR.sub.28R.sub.29) or its thioanalogue (POSR.sub.25NR.sub.28R.sub.29) where E can be independently O or S; [0057] wherein R.sub.25, R.sub.26, R.sub.27, R.sub.28, and R.sub.29 are independently a hydrogen, an unsubstituted straight-chain, branched or cyclic alkyl, alkenyl or alkynyl group, a substituted straight-chain, branched or cyclic alkyl, alkenyl or alkynyl group wherein one or more C, CH or CH.sub.2 groups are substituted with an O atom, N atom, S atom, or NH group, or an unsubstituted or substituted aromatic group; [0058] wherein Z is attached directly, or indirectly through a second linker arm comprising carbon, sulfur, oxygen, nitrogen, and any combinations thereof and wherein the second linker arm may be saturated or unsaturated, linear or branched, substituted or unsubstituted or any combinations thereof; and
[0059] wherein R.sub.5, R.sub.6, R.sub.23 and R.sub.24 can independently be hydrogen or an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, or when taken in combination R.sub.5 and R.sub.6 or R.sub.2 and R.sub.5 or R.sub.3 and R.sub.6 or R.sub.23 and R.sub.24 or R.sub.22 and R.sub.23 or R.sub.20 and R.sub.24 form a five or six membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted.
[0060] In other embodiments, a compound is provided that exhibits at least three times increased fluorescence in the presence of an aggregated form of a protein when compared to the fluorescence exhibited when the compound is in the presence of the unaggregated form of the protein. In some embodiments, the compound is D95, D97, L-30, L-33, Lu-1, Lu-2, S-8, S13. S22, S25, S33, S39, S42, S43, S48, S49, SL2131, SL2592, Tio-1, TOL-2, TOL-3, TOL-5, TOL-6, TOL-7, TOL-11, YA-1, YA-3, YAT2134, YAT2135, YAT2148, YAT2149, YAT2150, YAT2213, YAT2214 or YAT2324.
[0061] A multi-dye composition comprising at least three dyes is also provided. In this composition, each of the at least three dyes exhibits increased fluorescence in the presence of an aggregated form of a protein when compared to the fluorescence exhibited when the compound is in the presence of the unaggregated form of the protein.
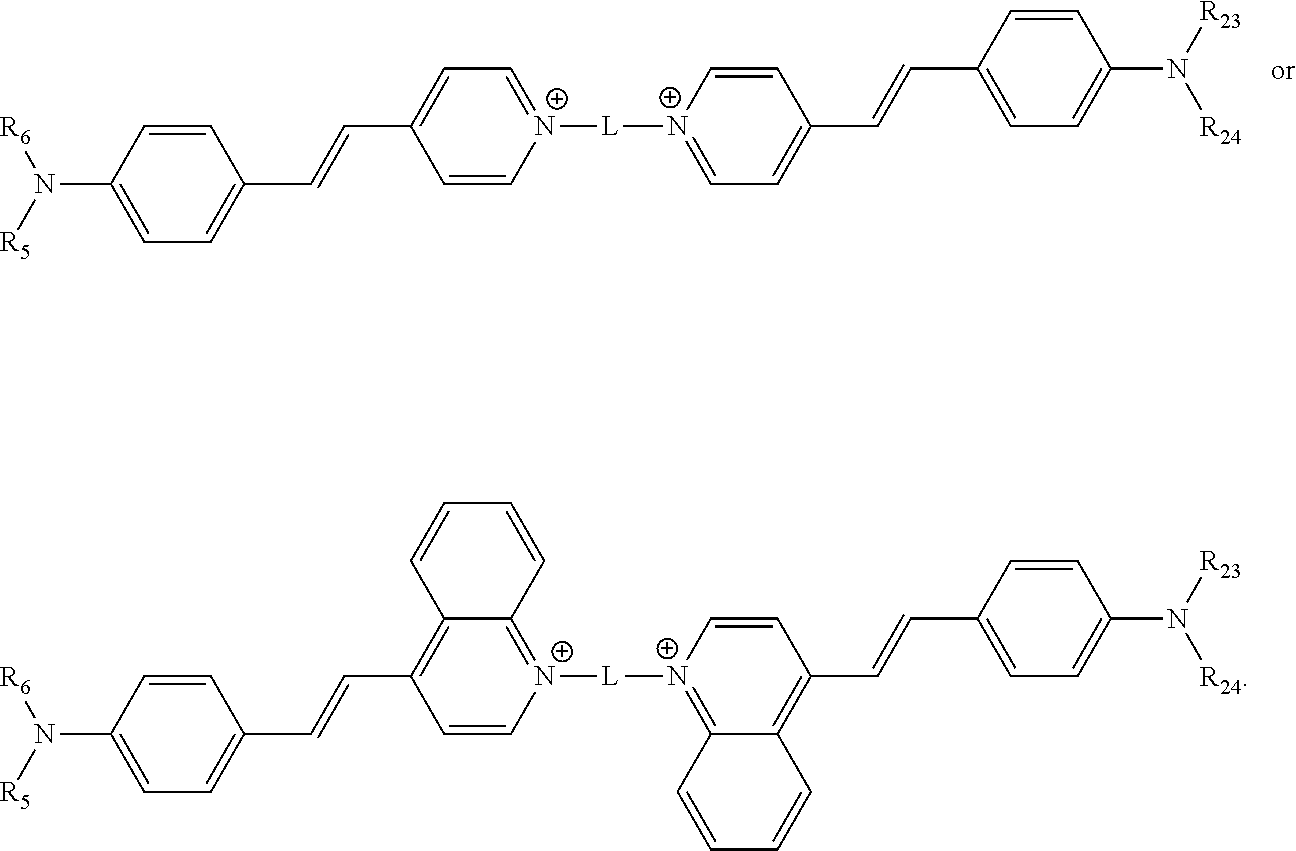
[0062] Further provided is a multi-dye composition comprising two or more dyes. In this composition, at least one of the two or more dyes comprises Dye F, Dye Fm(b), D95, D97, L-30, L-33, Lu-1, Lu-2, S-8, S13. S22, S25, S33, S39, S42, S43, S48, S49, SL2131, SL2592, Tio-1, TOL-2, TOL-3, TOL-5, TOL-6, TOL-7, TOL-11, YA-1, YA-3, YAT2134, YAT2135, YAT2148, YAT2149, YAT2150, YAT2213, YAT2214 or YAT2324.
[0063] A reactive compound comprising at least one compound from Table 1B or Table 2B is additionally provided. In these embodiments, the compound is modified by the addition of a reactive group.
[0064] Additionally, a labeled target molecule is provided. The labeled target molecule comprises a target molecule attached to the above-described reactive compound through the reactive group.
[0065] A solid support attached to the above-described reactive compound through the reactive group is also provided.
[0066] A kit for assaying aggregation of a protein is also provided. The kit comprises in packaged combination: (a) the above-described compound, and (b) instructions for using the compound for assaying aggregation of a protein.
[0067] Another kit for assaying aggregation of a protein is additionally provided. The kit comprises in packaged combination: (a) two or more compounds, wherein each of compound exhibits increased fluorescence in the presence of an aggregated form of a protein when compared to the fluorescence produced when the compound is in the presence of the unaggregated form of the protein, and (b) instructions therefor.
[0068] Additionally provided is a method for detecting an aggregate of a protein in a sample. The method comprises (a) combining the sample with the above-described compound or multidye composition; (b) measuring the amount of fluorescence in the mixture;
[0069] (c) comparing the amount of fluorescence determined in (b) with the amount of fluorescence in [0070] (i) a mixture of the compound or multidye composition with a control sample without aggregated protein, or [0071] (ii) a mixture of the compound or multidye composition with a known standard quantity of aggregated protein; and
[0072] (d) determining the aggregation of the protein in the sample based on the comparison in (c).
[0073] A method for separating aggregates of a protein from monomeric forms of the protein in a sample is also provided. The method comprises (a) combining the sample to the above-described solid support under conditions where aggregates of the protein preferentially bind to the compound; and (b) separating sample protein bound to the solid support from unbound protein. In this method, protein bound to the solid support are substantially aggregates and unbound protein is substantially monomers.
BRIEF DESCRIPTION OF THE DRAWINGS
[0074] FIG. 1, Panels A-D, are micrographs demonstrating IgG stability in two different buffer formulations.
[0075] FIG. 2 is a graph showing the fluorescence of various dye concentrations with 20 .mu.M of aggregated lysozyme.
[0076] FIGS. 3A-B are graphs showing the effect of pH on fluorescent detection sensitivity and linearity for different probes of the invention.
[0077] FIG. 4 is a graph showing the linear dynamic range of lysozyme aggregate detection using a two dye combination ST (S25 and Tol3) compared with thioflavin T.
[0078] FIG. 5 is a graph showing the effective linear dynamic range of antibody aggregate detection using a two dye combination ST (S25 and Tol3) compared with thioflavin T.
[0079] FIGS. 6A-D are graphs showing protein aggregate detection as a function of various protein species with the dyes S25, Tol3 and thioflavin T.
[0080] FIG. 7 is a graph showing the kinetics of lysozyme aggregation monitored with dyes S25, Tol3, Thioflavin T and the two dye combination ST (S25 and Tol3).
[0081] FIG. 8 is a graph showing the kinetics of IgG aggregation as a function of temperature.
[0082] FIG. 9 is a graph showing IgG aggregation induced by temperature (50.degree. C.) as a function of pH.
[0083] FIGS. 10A-C are graphs of a high-throughput protein formulation optimization workflow using IgG and the two dye combination ST (S25 and Tol3).
[0084] FIG. 11 is a graph showing measurement of the inhibition of Lysozyme aggregation by Chitotriose.
[0085] FIG. 12 is a graph showing a thermal shift assay of BLG aggregation using a dye of the present invention.
[0086] FIGS. 13A-B are graphs showing a thermal shift assay of carbonic anhydrase II aggregation at two different pH values using a dye of the present invention.
[0087] FIGS. 14A-B are graphs comparing the fluorescence response between unfolded and aggregated forms of IgG.
[0088] FIGS. 15A-C are graphs showing PDI activity monitored by turbidity and by a fluorometric assay using a dye of the present invention.
[0089] FIG. 16 is a graph showing activity assay of Hsp 27 (heat shock protein) as a chaperone preventing .beta.-lactoglobulin (BLG) aggregation induced by heat.
[0090] FIG. 17 is a graph showing fluorescence of IgG aggregates induced by stirring using a dye combination of the present invention.
[0091] FIG. 18, Panels A-B, are fluorescence micrographs of control cells (A) and cells treated with dye YAT2150 (B).
[0092] FIG. 19, Panels A-D are fluorescence micrographs of control cells (A) and cells treated with proteasome inhibitors and dye YAT2150.
[0093] FIGS. 20A-C are fluorescence micrographs of cells treated with various dyes to show that dye YAT2150 co-localizes with ubiquitin.
[0094] FIG. 21, Panels A-D, are fluorescence micrographs of control cells (A) and cells treated with amyloid beta peptide 1-42 (B, C, D) with (C, D) or without (B) treatment with SMER28, an inducer of autophagy.
[0095] FIGS. 22A (Panels A and B) and 22B (Panels A and B) are fluorescence micrographs of control cells of normal or Alzheimer's disease brain tissue after staining with thioflavin T (A) or YAT2150.
[0096] FIGS. 23A (Panels A and B) and 23B (Panels A and B) are fluorescence micrographs showing that dye YAT2150 co-localized with the Tau-13 protein in post-mortem brain tissue of Alzheimer's disease patients.
[0097] FIGS. 24A-B are graphs comparing YAT2150 (ProteoStat.RTM.) with fluorescein-p62 antibody for identifying aggresomes by flow cytometry.
DETAILED DESCRIPTION OF THE INVENTION
[0098] As used herein, the singular forms "a", "an" and "the" are intended to include the plural forms as well, unless the context clearly indicates otherwise. Additionally, the use of "or" is intended to include "and/or", unless the context clearly indicates otherwise.
[0099] The present invention provides dyes, reagents and methods that are useful for detecting protein aggregates. In some embodiments, the invention provides a family of dimeric styryl dyes containing either a picoline or lepidine ring and a dialkyl amino or alkyloxy substituent. The dyes of the invention are useful for generating fluorescence signals that depend upon the presence of an aggregated form of a protein, while conveying minimal levels of signals when only the native form of the protein is present. A number of novel dimeric styryl dyes having these properties are also disclosed. Other dyes have been described previously in the context of binding to nucleic acids, but it has been discovered that many of these dyes demonstrate a useful property where an enhanced level of fluorescence is produced after binding to aggregated forms of proteins compared to the level that is emitted in the presence of the native forms. Some of these dyes also exhibit large Stokes shifts between their absorption and emission wavelength optima thereby increasing the ease of detection.
[0100] Thus, in some embodiments, a compound is provided. The compound comprises the structure
##STR00002##
[0101] wherein m and n are independently 1, 2 or 3;
[0102] wherein L is a linker arm comprising carbon, sulfur, oxygen, nitrogen, or any combination thereof;
[0103] wherein R.sub.1, R.sub.2, R.sub.3, R.sub.4, R.sub.9, R.sub.10, R.sub.11, R.sub.12, R.sub.13, R.sub.14, R.sub.15, R.sub.16, R.sub.19, R.sub.20, R.sub.21 and R.sub.22 are independently hydrogen, halogen, amino, ammonium, nitro, sulfo, sulfonamide, carboxy, ester, cyano, phenyl, benzyl, an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, an alkoxy group wherein the alkoxy group is saturated or unsaturated, branched or linear, substituted or unsubstituted, or when taken in combination R.sub.1 and R.sub.2, or R.sub.3 and R.sub.4, or R.sub.9 and R.sub.10, or R.sub.11 and R.sub.12, or R.sub.13 and R.sub.14, or R.sub.15 and R.sub.16, or R.sub.19 and R.sub.20, or R.sub.21 and R.sub.22 form a five or six membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted, and wherein R.sub.9 and R.sub.10, or R.sub.11 and R.sub.12, or R.sub.13 and R.sub.14, or R.sub.15 and R.sub.16 can comprise alkyl chains that are joined together, wherein a quinoline moiety can be formed;
[0104] wherein R.sub.7, R.sub.8, R.sub.17 and R.sub.18 are independently hydrogen, Z, an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, an alkoxy group wherein the alkoxy group is saturated or unsaturated, branched or linear, substituted or unsubstituted, or when taken together, R.sub.7 and R.sub.8 and R.sub.17 and R.sub.18, may form a 5 or 6 membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted; [0105] wherein Z comprises a carboxyl group (CO.sub.2.sup.-), a carbonate ester (COER.sub.25), a sulfonate (SO.sub.3.sup.-), a sulfonate ester (SO.sub.2ER.sub.25), a sulfoxide (SOR.sub.25), a sulfone (SO.sub.2CR.sub.25R.sub.26R.sub.27), a sulfonamide (SO2NR.sub.25R.sub.26), a phosphate (PO.sub.4.sup.=), a phosphate monoester (PO.sub.3.sup.-ER.sub.25), a phosphate diester (PO.sub.2ER.sub.25ER.sub.26), a phosphonate (PO.sub.3.sup.=) a phosphonate monoester (PO.sub.2.sup.-ER.sub.25) a phosphonate diester (POER.sub.25ER.sub.26), a thiophosphate (PSO.sub.3.sup.=), a thiophosphate monoester (PSO.sub.2.sup.-ER.sub.25) a thiophosphate diester (PSOER.sub.25ER.sub.26), a thiophosphonate (PSO.sub.2.sup.=), a thiophosphonate monoester (PSO.sup.-ER.sub.25) a thiophosphonate diester (PSER.sub.25ER.sub.26), a phosphonamide (PONR.sub.25R.sub.26NR.sub.28R.sub.29), its thioanalogue (PSNR.sub.25R.sub.26NR.sub.28R.sub.29), a phosphoramide (PONR.sub.25R.sub.26NR.sub.27NR.sub.28R.sub.29), its thioanalogue (PSNR.sub.25R.sub.26NR.sub.27NR.sub.28R.sub.29), a phosphoramidite (PO.sub.2R.sub.25NR.sub.28R.sub.29) or its thioanalogue (POSR.sub.25NR.sub.28R.sub.29) where E can be independently O or S; [0106] wherein R.sub.25, R.sub.26, R.sub.27, R.sub.28, and R.sub.29 are independently a hydrogen, an unsubstituted straight-chain, branched or cyclic alkyl, alkenyl or alkynyl group, a substituted straight-chain, branched or cyclic alkyl, alkenyl or alkynyl group wherein one or more C, CH or CH.sub.2 groups are substituted with an O atom, N atom, S atom, or NH group, or an unsubstituted or substituted aromatic group; [0107] wherein Z is attached directly, or indirectly through a second linker arm comprising carbon, sulfur, oxygen, nitrogen, and any combinations thereof and wherein the second linker arm may be saturated or unsaturated, linear or branched, substituted or unsubstituted or any combinations thereof; and
[0108] wherein R.sub.5, R.sub.6, R.sub.23 and R.sub.24 can independently be hydrogen or an alkyl group wherein the alkyl group is saturated or unsaturated, linear or branched, substituted or unsubstituted, or when taken in combination R.sub.5 and R.sub.6 or R.sub.2 and R.sub.5 or R.sub.3 and R.sub.6 or R.sub.23 and R.sub.24 or R.sub.22 and R.sub.23 or R.sub.20 and R.sub.24 form a five or six membered ring wherein the ring is saturated or unsaturated, substituted or unsubstituted.
[0109] In many of these embodiments, the compound exhibits increased fluorescence in the presence of an aggregated form of a protein when compared to the fluorescence exhibited when the compound is in the presence of the unaggregated form of the protein.
[0110] These compounds can be modified by the addition of charged groups, as exemplified by sulfonates, phosphates, phosphonates and their derivatives and/or polar groups as exemplified by sulfoxide, sulfone and sulfonamide moieties.
[0111] It is also understood that when a dye comprises an anionic group, there will also be a cationic counterion present. Any cation may serve this purpose as long as it does not interfere with the use of the dye. Examples of cations that may serve as counterions can include but are not limited to hydrogen, sodium, potassium, lithium, calcium, cesium, ammonium, alkyl ammonium, alkoxy ammonium and pyridinium. It is also understood that when a dye comprises a cationic group, there will also be an anionic counterion present. Any anion may serve this purpose as long as it doesn't interfere with the use of the dye. Examples of anions that may serve as counterions can include but not be limited to perchlorate (ClO.sub.4.sup.-), sulfate (SO.sub.4.sup.=), sulfonate, alkane sulfonate, aryl sulfonate, phosphate, tosylate, mesylate and tetrafluoroborate moieties and halides such as a bromide, chloride, fluoride and iodide. In some cases the counterion or counterions are provided by the dye being a salt where they exist as separate ionic species. In other cases, the counterion or counterions may be present as part of the compound (sometimes called inner salts). It is understood that there may also be a combination of ions that are provided by the compound and salts. With regard to acid moieties that are shown in forms such as COOH it is also understood that these compounds may be found in ionized forms such as COO.sup.-.
[0112] It should also be appreciated by those skilled in the art that the stoichiometric number of counterion or counterions which balance the charge or charges on the compound can be the same or they can be different provided that the counterions balance the charge(s) on the compound. The combination of counterions can be selected from any of the above mentioned anions. This applies for the combination of cations also.
[0113] It should be further appreciated by those skilled in the art that the foregoing descriptions of the anions and their stoichiometric number and/or combination are applicable to the compounds and dyes of the present invention, and to methods which use these compounds and dyes.
[0114] Alkyl or alkoxy R groups in the above compounds may be substituted or unsubstituted. Examples of substitutions can include but are not limited to one or more fluorine, chlorine, bromine, iodine, hydroxy, carboxy, carbonyl, amino, cyano, nitro or azido groups as well as other alkyl or alkoxy groups. The length of the alkoxy groups may be as desired. For instance, they may independently comprise from 1 to 18 carbons in length. They may be shorter as well, for instance they may be only 1 to 6 carbons in length in a dye molecule of the present invention.
[0115] The polar groups, charged groups and other substituents may be connected to the dye directly or they may be connected by a linker arm comprising carbon, nitrogen, sulfur, oxygen or any combination thereof. The linker arm may be saturated or unsaturated, linear or branched, substituted or unsubstituted as well as any combination of the foregoing.
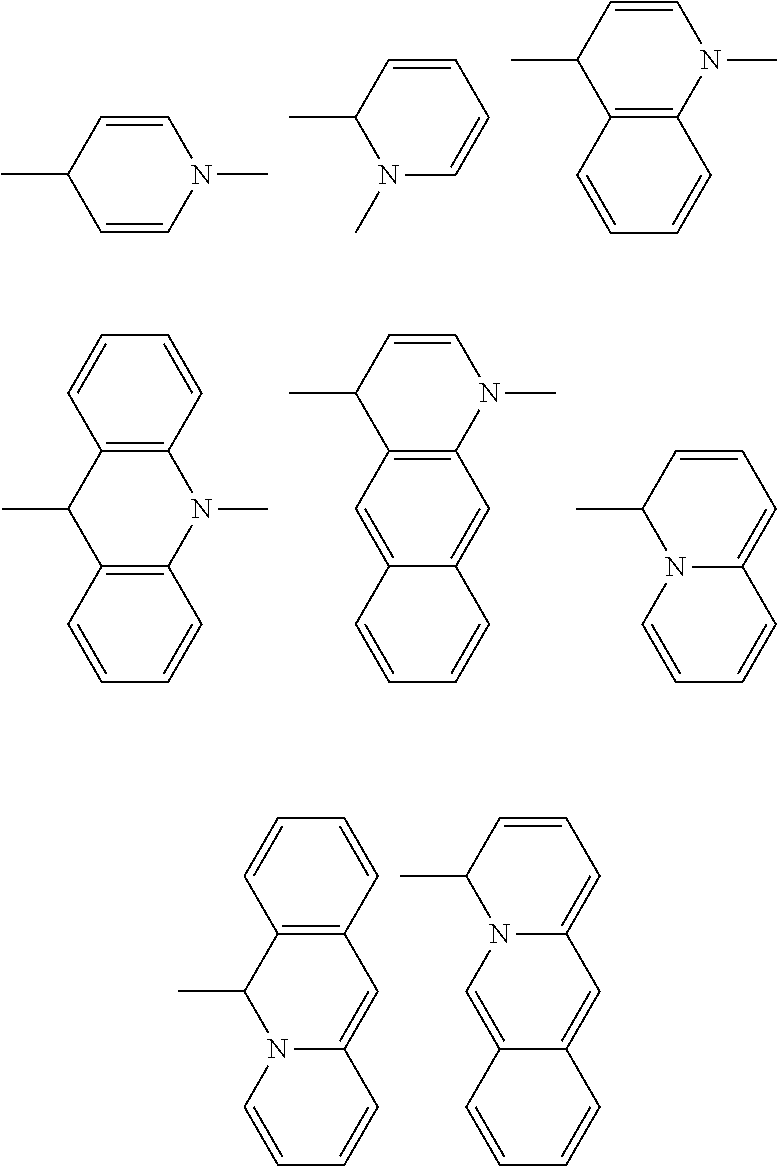
[0116] As described above some of the R groups may be joined together to form one or more fused 5 or 6 membered ring structures. It is understood that the complex rings that are formed by closure of R groups may be further substituted with any of the R groups described previously. Examples of complex rings that may be formed for the picoline or lepidine portion of the cyanine dyes of the invention can comprise but not be limited to:
##STR00003##
[0117] Examples of rings and complex rings that may be part of the styryl portion of the dye can comprise but not be limited to:
##STR00004##
[0118] In various embodiments, the compound comprises the structure
##STR00005##
[0119] In some of these embodiments, each of R.sub.5, R.sub.6, R.sub.23 and R.sub.24 are a methyl or an ethyl moiety.
[0120] As described in Example 1, numerous compounds having the above structure, as well as other compounds, were tested for the ability to exhibit increased fluorescence in the presence of an aggregated form of a protein (human .alpha.-synuclein) when compared to the fluorescence exhibited when the compound is in the presence of the unaggregated form of the protein. The excitation and emission wavelength in the presence and absence of the protein aggregate was also determined. Results of these tests, and the structures of the tested compounds, are provided in Tables 1 and 2. Table 1 gives results where the compounds exhibited a ratio of 3 or more for fluorescence from binding to protein aggregates compared to being in the presence of monomeric protein; Table 2 gives results with other compounds.
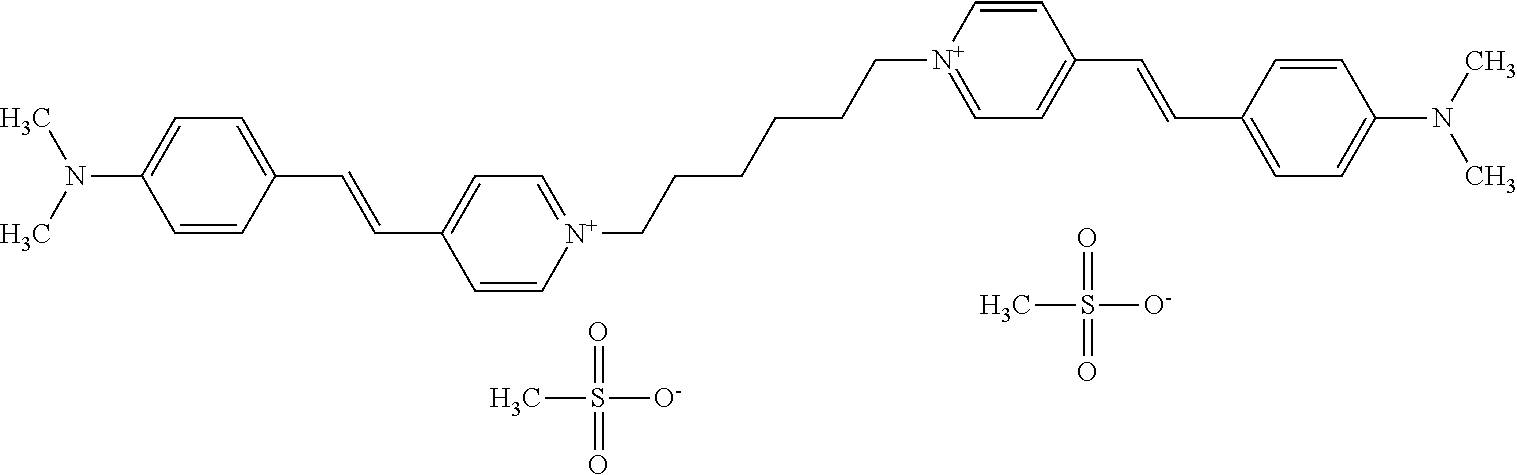
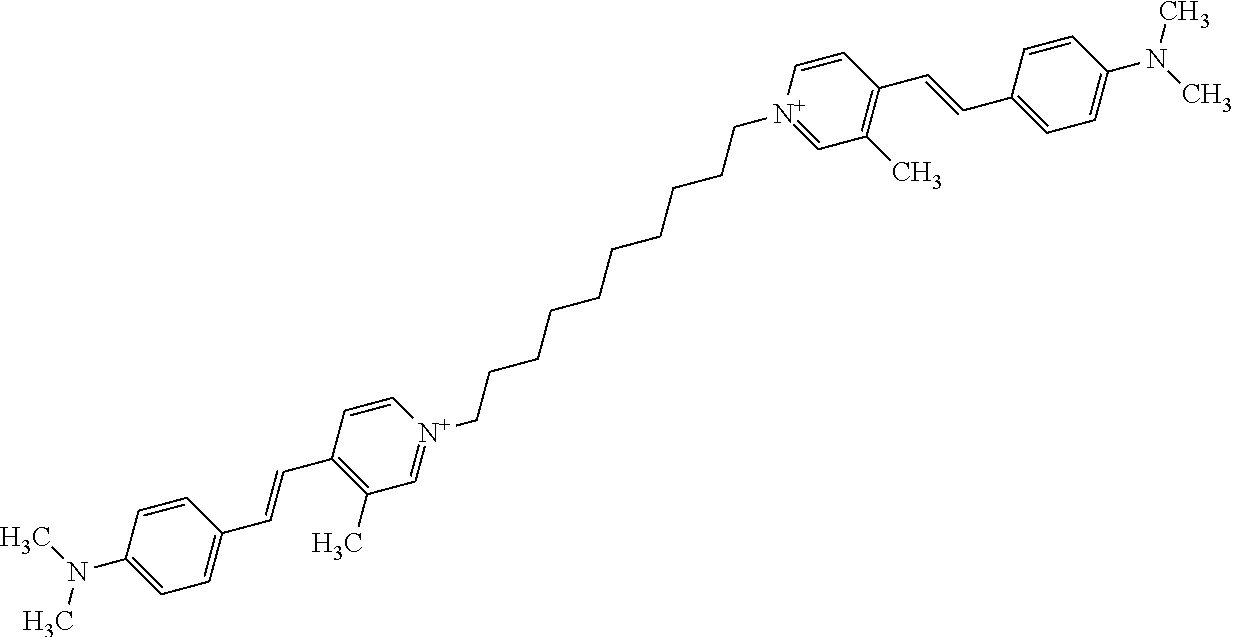
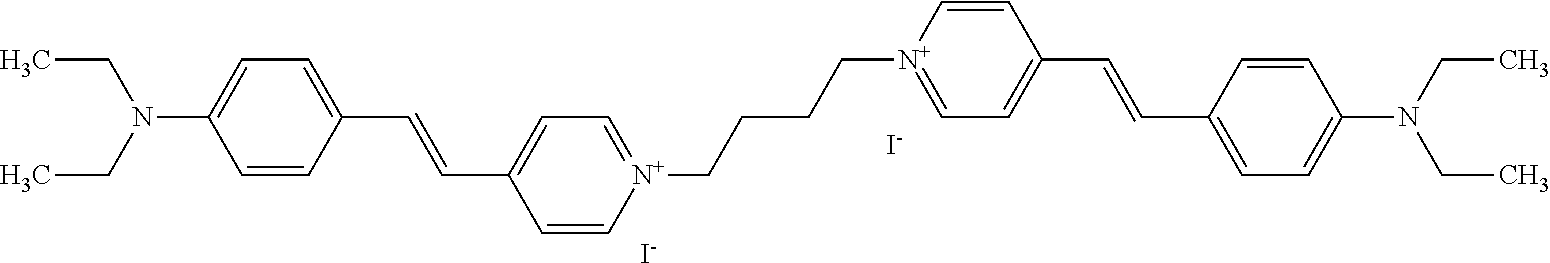
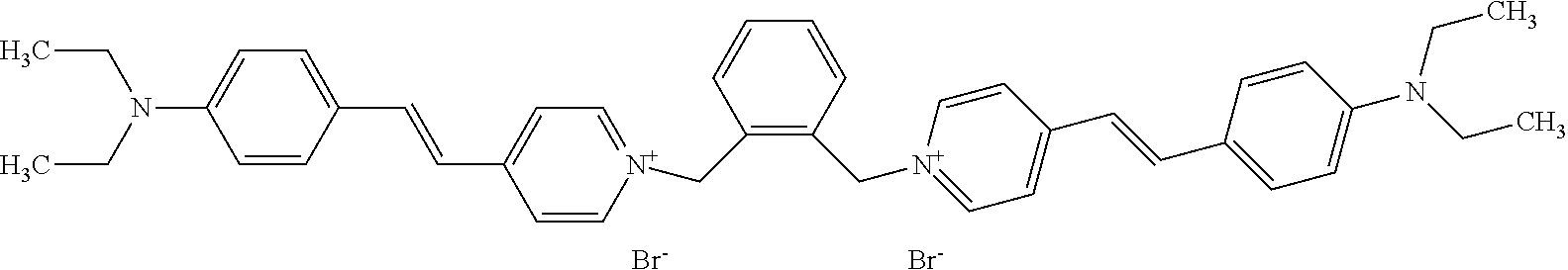
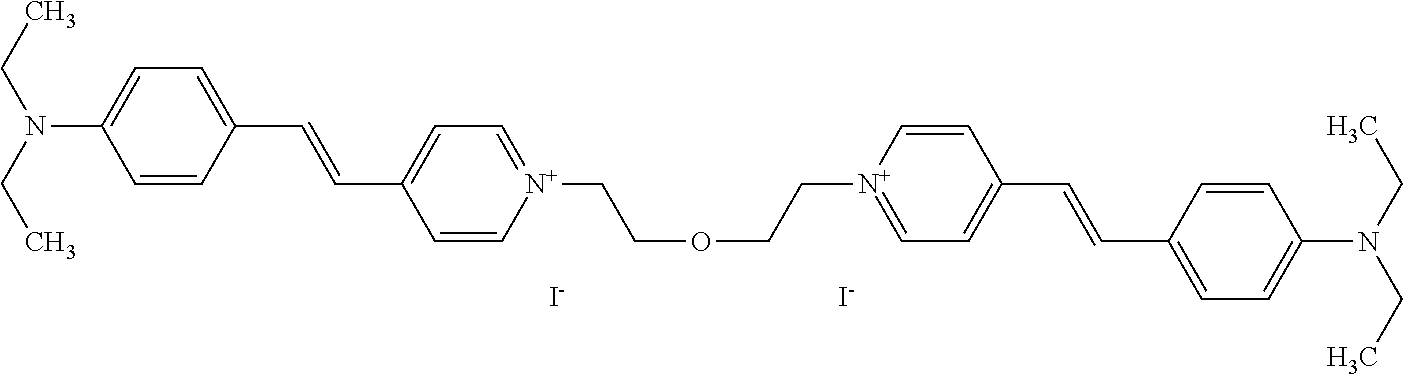
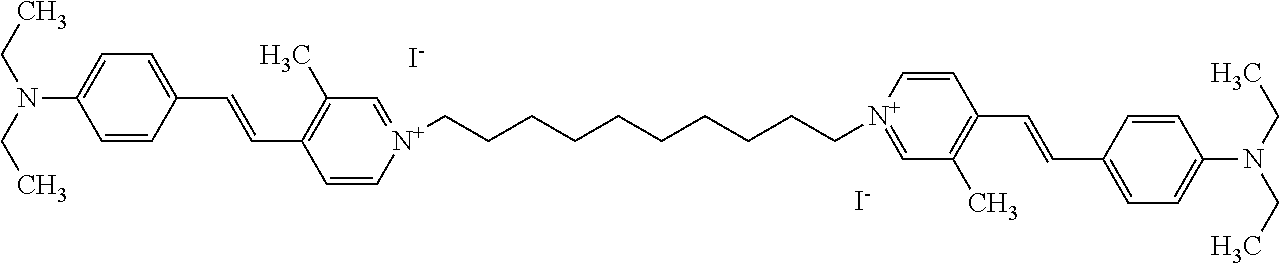
TABLE-US-00001 TABLE 1 Compounds tested that exhibit a ratio of 3 or more for fluorescence from binding to protein aggregates compared to being in the presence of monomeric protein. A. Properties of compounds. Dye Dye Dye with Dye with Alone: Alone: Aggregate: Aggregate: Fluore- Fluore- Fluore- .lamda..sub.Ex .lamda..sub.Em .lamda..sub.Ex .lamda..sub.Em cence cence cence I.sub.Agg/ Dye (nm) (nm) (nm) (nm) I.sub.dye I.sub.Mono I.sub.Agg I.sub.Mono S25 485 613 516 607 3.4 4.6 87.3 19.0 S43 527 637 550 623 0.35 0.58 27.3 47 TOL3 471 611 511 603 2.7 2.7 40.2 14.9 Yat 500 620 535 613 4.2 4.9 63.2 12.9 2134 Yat 520 632 553 625 1.2 3.4 53 15.6 2148 Yat 502 614 534 617 0.6 0.7 29.5 42 2149 Yat 485 612 515 610 6.7 9.7 42.3 4.4 2150 F 460 610 518 607 3.6 3.4 57.4 16.9 L-33 465 527 462 504 7.7 7.6 53 7.0 S49 501 584 524 576 2.2 2.2 20.1 9.1 S33 479 616 513 611 5.5 5.7 19.4 3.4 TOL- 389 539 554 603 10 6.5 22.5 3.5 11 SL- 491 578 516 578 7.4 7.5 30.3 4.1 2131 SL- 401 608 400 608 7.5 8 25.4 7.5 2592 Tio-1 494 578 526 578 2.8 2.8 19 6.8 S-13 568 662 580 670 0.2 0.2 3.1 15.5 L-30 457 515 478 512 1.5 1.8 8 4.4 YA-1 446 491 461 498 7.7 11 45 4.1 YA-3 460 514 456 537 6.4 7.9 24.2 3.1 (Diph40) TOL-2 527 595 566 600 0.5 0.5 3.5 7 [T-33] TOL-5 428 581 460 535 3.8 4.5 22.2 4.9 Dil-10 548 595 564 599 3.7 3.6 15.3 4.3 [TOL-7] S-39 540 599 577 605 3.1 3.3 11.3 3.4 Fm [b] 461 610 504 597 5.5 5.5 21 3.8 S-42 547 600 559 603 1.1 1.1 5 4.5 S-48 491 581 527 588 2.3 2.3 15.8 6.9 TOL-6 501 559 512 559 4.6 4.6 17.5 3.8 Lu-1 453 583 452 526 7.3 7.6 24 3.2 Lu-2 473 506 485 503 3.8 3 14 4.7 Yat2135 500 618 540 620 0.9 0.7 12.5 17.9 Yat2214 507 626 549 625 1.5 1.4 6.4 4.6 Yat2213 483 622 540 622 0.6 1.2 5.5 4.6 D-95 450 585 555 598 0.6 1 8.4 8.4 D-97 516 650 587 650 3.6 3.7 13.4 3.6 S-8 547 671 566 667 0.8 0.8 2.8 3.5 Yat2324 500 619 551 619 0.8 0.7 7.1 10.1 S-22 543 598 562 602 0.5 0.8 2.4 3.0 B. Structures of compounds. Dye Structure S25 ##STR00006## S43 ##STR00007## TOL3 ##STR00008## Yat 2134 ##STR00009## Yat 2148 ##STR00010## Yat 2149 ##STR00011## Yat 2150 ##STR00012## F ##STR00013## L-33 ##STR00014## S49 ##STR00015## S-33 ##STR00016## TOL-11 ##STR00017## S-22 ##STR00018## SL-2131 ##STR00019## SL-2592 ##STR00020## Tio-1 ##STR00021## S-13 ##STR00022## L-30 ##STR00023## YA-1 ##STR00024## YA-3 (Diph40) ##STR00025## TOL-2 [T-33] ##STR00026## TOL-5 ##STR00027## Dil-10 [TOL-7] ##STR00028## S-39 ##STR00029## Fm [b] ##STR00030## S-42 ##STR00031## S-48 ##STR00032## TOL-6 ##STR00033## Lu-1 ##STR00034## Lu-2 ##STR00035## Yat-2135 ##STR00036## Yat-2214 ##STR00037## Yat-2213 ##STR00038## D-95 ##STR00039## D-97 ##STR00040## S-8 ##STR00041## Yat2324 ##STR00042##
TABLE-US-00002 TABLE 2 Compounds tested that exhibit a ratio of less than 3 or more for fluorescence from binding to protein aggregates compared to being in the presence of monomeric protein. A. Properties of compounds Dye with Dye with Dye alone: Dye alone: aggregate: aggregate: Fluorescence Excitation Emission Excitation Emission enhancement: wavelength wavelength wavelength wavelength Aggregate/ Dye (nm) (nm) (nm) (nm) monomer S-11 531 594 560 600 2.6 S-12 539 597 553 599 2.2 SH-330 393 278 398 483 1.6 SH-654 370 443 359 434 0.91 SH-675 445 472 449 475 1.8 SH-975 471 631 471 630 1.8 SH-1036 478 611 464 605 2.7 Sl-2599 468 564 468 569 1.1 Sl-2600 518 535 518 536 1.9 S-7 460 612 465 609 1.1 L-28 460 654 572 577 1.4 L-31 450 527 462 534 2.7 TOL-4 488 665 458 654 1.4 TOL-10 394 544 397 539 1.4 S-26 532 593 562 602 2.7 S-29 543 597 554 600 1.6 S-44 498 586 525 582 2.8 S-45 534 596 558 600 2.3 Dbt-5 539 597 545 598 2.2 [TOL-9] S-30 530 598 570 600 1.9 Sip-7 397 576 397 576 1.2 [TOL-12] S-28 384 608 384 608 1 S-23 464 546 471 553 1.2 SH-1070 408 500 408 480 1.2 Yat2212 485 623 530 620 2.6 D-91 395 520 396 517 1.03 D-78 426 621 426 621 0.94 D-68 553 696 558 694 1.1 D-69 483 638 483 637 1.07 D-160 481 631 493 617 1.2 D-155 500 625 516 619 1.6 D-72 380 477 375 469 1.1 D-163 493 588 490 589 1.06 D-159 487 603 494 593 1.5 D-80 489 669 486 668 0.31 D-84 494 623 507 602 2.8 D-90 475 662 479 655 0.36 D-162 472 706 565 692 9.9 D-70 506 615 506 614 0.92 D-86 388 544 387 544 1 D-87 430 534 428 534 1.08 D-85 450 515 534 608 2.3 Tol-24 530 594 540 598 2.4 Yat2325 503 624 538 622 2.5 S-5 527 597 535 598 1.1 S-38 535 599 555 602 2.1 S-37 545 600 551 602 1.9 S-3 562 595 565 596 1.2 S-27 522 607 540 608 1.2 SIP-2 397 576 397 575 1.3 D-74 517 601 527 601 1 Sbt 520 592 551 594 2.8 D-75 494 554 494 555 1.5 D-71 482 585 498 587 1.4 Dbo-10 505 559 515 597 2.1 SI-1999 582 595 582 595 1 SL-42 555 567 555 567 0.9 Dimer- 431 577 440 580 1.12 NN SIP-3 398 579 408 582 0.98 SIP-10 404 582 440 590 0.79 Dst-NN-6 397 572 402 572 1 SIP-8 446 582 442 584 0.28 Dst-NN- 396 572 398 572 1.24 10 Dst-NN- 398 584 413 600 0.76 11 Dst-NN- 404 581 412 595 0.79 12 SI-1035 512 545 512 546 1 SI-1047 574 596 575 596 0.95 SI-1056 546 571 548 572 1 SL-1722 673 700 676 699 1 SL-2153 547 573 547 573 0.9 SI-2596 491 594 492 609 1.1 SI-2611 456 554 460 555 1 T-164 559 575 559 572 1 SH-0229 520 628 525 641 0.7 T-33 589 656 589 656 0.7 SH-0423 409 536 409 588 1 SH-0428 588 601 588 603 2.3 SH-0627 558 569 558 569 1.1 T-333 559 576 559 576 1 T-74 561 576 561 576 0.9 SH-0999 640 653 585 596 2 T-165 583 623 588 632 2.3 T-364 582 628 581 630 1.4 Dst-NN- 398 576 409 576 0.65 13 T-119 530 635 532 636 1 T-15 554 571 564 575 1.2 TOL-26 563 609 564 607 0.9 Dst-NN-8 366 474 374 472 1.26 SL-2057 589 603 591 605 1.26 SL-2059 582 608 582 607 0.97 SL-2132 532 604 558 609 1.46 B. Structures of compounds. Dye Structure S-11 ##STR00043## S-12 ##STR00044## SH-330 ##STR00045## SH-654 ##STR00046## SH-675 ##STR00047## SH-975 ##STR00048## SH-1036 ##STR00049## SI-2599 ##STR00050## SI-2600 ##STR00051## S-7 ##STR00052## L-28 ##STR00053## L-31 ##STR00054## L-28 ##STR00055## TOL-4 ##STR00056## TOL-10 ##STR00057## S-26 ##STR00058## S-29 ##STR00059## S-44 ##STR00060## S-45 ##STR00061## S-30 ##STR00062## Dbt-5 [TOL-9] ##STR00063## Sip-7 [TOL-12] ##STR00064## S-28 ##STR00065## S-23 ##STR00066## SH-1070 ##STR00067## Yat-2212 ##STR00068## D-91 ##STR00069## D-78 ##STR00070## D-68 ##STR00071## D-69 ##STR00072## D-160 ##STR00073## D-155 ##STR00074## D-72 ##STR00075## D-163 ##STR00076## D-159 ##STR00077## D-80 ##STR00078## D-84 ##STR00079## D-90 ##STR00080## D-162 ##STR00081## D-70 ##STR00082## D-86 ##STR00083## D-87 ##STR00084## D-85 ##STR00085## Tol-24 ##STR00086## Yat2325 ##STR00087## S-5 ##STR00088## S-38 ##STR00089## S-37 ##STR00090## S-3 ##STR00091## S-27 ##STR00092## SIP-2 ##STR00093## D-74 ##STR00094## Sbt ##STR00095## D-75 ##STR00096## D-71 ##STR00097## Dbo-10 ##STR00098## SI-1999 ##STR00099## SL-42 ##STR00100## Dimer-NN ##STR00101## SIP-3 ##STR00102## SIP-10 ##STR00103## Dst-NN-6 ##STR00104## SIP-8 ##STR00105## Dst-NN-10 ##STR00106## Dst-NN-11 ##STR00107## Dst-NN-12 ##STR00108## SI-1035 ##STR00109##
SI-1047 ##STR00110## SI-1056 ##STR00111## SL-1722 ##STR00112## SL-2153 ##STR00113## SI-2596 ##STR00114## SI-2611 ##STR00115## T-164 ##STR00116## SH-0229 ##STR00117## T-33 ##STR00118## SH-0423 ##STR00119## SH-0428 ##STR00120## SH-0627 ##STR00121## T-333 ##STR00122## T-74 ##STR00123## SH-0999 ##STR00124## T-165 ##STR00125## T-364 ##STR00126## Dst-NN-13 ##STR00127## T-119 ##STR00128## T-15 ##STR00129## TOL-26 ##STR00130## Dst-NN-8 ##STR00131## SL-2057 ##STR00132## SL-2059 ##STR00133## SL-2132 ##STR00134##
[0121] Although the compounds in Tables 1 and 2 are shown with a particular counterion, it should be understood that the compounds can also utilize other counterions as described above. As such, when the above compounds are identified by name herein, the named compound includes the structure identified in Table 1 or 2 with any counterion, unless the counterion is particularly specified.
[0122] Notable examples of compounds useful compounds from Table 1 include
##STR00135##
wherein X comprises an anion (compound S25).
##STR00136##
wherein X comprises an anion (TOL3).
##STR00137##
wherein X comprises an anion (S43).
##STR00138##
wherein X comprises an anion (YAT2134).
##STR00139##
wherein X comprises an anion (YAT2148).
##STR00140##
wherein X comprises an anion (YAT2149).
##STR00141##
wherein X comprises an anion (YAT2150).
[0123] Especially useful for many purposes are dyes that have fluorescence emissions in the range of 600-650 nM since such dyes can avoid interference of biological proteins for the application in tissue staining, such as green fluorescent proteins (GFPs). Excitation fluorescence for such dyes are preferred to be in the range of 500-600 nM. It can be seen that the dyes in Table 1 fulfill these requirements where the maxima of the fluorescence excitation spectra of these dyes in the presence of aggregates of .alpha.-synuclein (ASN) are between 511 and 553 nm, and fluorescence emission have their maxima between 603 and 625 nm. The values of the fluorescence quantum yield (QY) of the dyes of the invention in the presence of saturating concentrations of fibrillar protein are situated in the range between 0.01 and 0.08, which allow using relatively small amounts of dye for interaction with protein aggregates, tissues or cell staining. Stokes shift of the dyes of the invention are in the range of 73 to 95 nm and are much larger than the classic amyloid detection dyes, such as thioflavin T, which only has a 23 nm Stokes shift. The wider Stokes shift of the dyes of the present invention ensures a much lower overlap between excitation and emission, thus allowing more flexible filter set selection, such as a wide excitation and or emission filter to improve the brightness of the dye or increasing the exposure time to enhance the fluorescence intensity. With these considerations, particularly useful compounds from Table 1 include S25, S43, TOL3, YAT2134, YAT2148, YAT2149, S13, YAT2135 and YAT2324.
[0124] It is to be understood that with any particular dye, the excitation maximum, emission maximum, and/or ratio of fluorescence intensity in the presence of aggregates vs. monomers can vary to some extent with different proteins. Thus, the selection of a dye to use for detection of the aggregates of any particular protein could benefit from information of the fluorescence characteristics of the dye with the particular protein. Such information can be obtained for any protein-dye combination without undue experimentation, for example by using the methods described in Example 1. Nonlimiting examples of useful proteins whose aggregation could be detected using the above compounds include immunoglobulin, a DNA polymerase or a fragment thereof, .alpha.-synuclein, synphilin-1, TCR.alpha., P23H mutant of rhodopsin, .DELTA.F508 mutant of CFTR, amyloid-.beta., prion protein, Tau, SOD1, Ig light chains, ataxin-1, ataxin-3, ataxin-7, calcium channel, atrophin-1, androgen receptor, p62/sequestosomel (SQSTM1), Pael receptor, serum amyloid A, transthyretin, .beta.2-microglobulin, apolipoprotein A-1, gelsolin, atrial natriuretic factor, lysozyme, insulin, fibrinogen, crystallin, surfactant protein C, lactoferrin, .beta.ig-h3, PAPB2, corneodesmosin, neuroserpin, cochlin, RET, myelin, protein 22/0, SCAD, prolactin, lactadherin, p53, procalcitonin, cytokeratin, GFAP, ATP7B, prolyl hydroxylase PHD3, presenilin, or huntingtin.
[0125] A further consideration of the present invention, is that detection and/or quantification of aggregates may also be improved by a mixture of dyes where at least one of the dyes is one of the compounds illustrated in Table 1. The additional dye or (dyes) may also be from Table 1 or 2. The use of more than one dye may widen the breadth of proteins that will successfully generate signals after aggregation when these dyes become bound. The signal will derive from the net amount of fluorescence enhancement derived from each dye in the mixture. Particularly useful multi-dye compositions comprise dyes where the emission maximum of each dye is within 150 nm of the emission maximum of each of the other dyes. For some applications, multi-dye compositions may be even more useful where the compositions comprise dyes where the emission maximum of each dye is within 50 nm of the emission maximum of each of the other dyes.
[0126] Thus, in some embodiments, a multi-dye composition is provided. This multi-dye composition comprises at least three dyes, where each of the at least three dyes exhibits increased fluorescence in the presence of an aggregated form of a protein when compared to the fluorescence exhibited when the compound is in the presence of the unaggregated form of the protein. In some of these embodiments, each of the three dyes is selected from the group consisting of Dye F, Dye Fm(b), D95, D97, L-30, L-33, Lu-1, Lu-2, S-8, S13. S22, S25, S33, S39, S42, S43, S48, S49, SL2131, SL2592, Tio-1, TOL-2, TOL-3, TOL-5, TOL-6, TOL-7, TOL-11, YA-1, YA-3, YAT2134, YAT2135, YAT2148, YAT2149, YAT2150, YAT2213, YAT2214 and YAT2324. In other embodiments, at least one of the three dyes is selected from the group consisting of S25, S43, TOL3, YAT2134, YAT2148, YAT2149, S13, YAT2135, YAT2324 and YAT2150.
[0127] Another multi-dye composition is also provided herein. This multi-dye composition comprises two or more dyes, where at least one of the two or more dyes comprises Dye F, Dye Fm(b), D95, D97, L-30, L-33, Lu-1, Lu-2, S-8, S13. S22, S25, S33, S39, S42, S43, S48, S49, SL2131, SL2592, Tio-1, TOL-2, TOL-3, TOL-5, TOL-6, TOL-7, TOL-11, YA-1, YA-3, YAT2134, YAT2135, YAT2148, YAT2149, YAT2150, YAT2213, YAT2214 or YAT2324. In some of these embodiments, at least one of the two dyes is selected from the group consisting of S25, S43, TOL3, YAT2134, YAT2148, YAT2149, S13, YAT2135, YAT2324 and YAT2150. In other embodiments, both of the two dyes are selected from the group consisting of S25, S43, TOL3, YAT2134, YAT2148, YAT2149, S13, YAT2135, YAT2324 and YAT2150. In particular embodiments, the two dyes are S25 and TOL3. See, e.g., Example 26.
[0128] In another embodiment of the present invention, any of the above dyes further comprises a reactive group, thereby allowing their attachment to targets of interest. Examples of reactive groups that may find use in the present invention can include but not be limited to a nucleophilic reactive group, an electrophilic reactive group, a terminal alkene, a terminal alkyne, a platinum coordinate group or an alkylating agent.
[0129] There are a number of different electrophilic reactive groups that may find use with the present invention; examples can include but not be limited to isocyanate, isothiocyanate, monochlorotriazine, dichlorotriazine, 4,6,-dichloro-1,3,5-triazines, mono- or di-halogen substituted pyridine, mono- or di-halogen substituted diazine, maleimide, haloacetamide, aziridine, sulfonyl halide, acid halide, hydroxysuccinimide ester, hydroxysulfosuccinimide ester, imido ester, hydrazine, azidonitrophenol, azide, 3-(2-pyridyl dithio)-propionamide, glyoxal and aldehyde groups. Nucleophilic reactive groups can include but not be limited to reactive thiol, amine and hydroxyl groups. For purposes of synthesis of dyes, reactive thiol, amine or hydroxyl groups can be protected during various synthetic steps and the reactive groups generated after removal of the protective group. Use of a terminal alkene or alkyne groups for attachment of markers has been previously described in U.S. Patent Application Serial No. 2003/0225247, hereby incorporated by reference. The use of platinum coordinate groups for attachment of other dyes has been previously disclosed in U.S. Pat. No. 5,580,990 and the use of alkyl groups has been previously described in U.S. Pat. No. 6,593,465 B1, both of which patents are hereby incorporated by reference. In some cases the molecules that have been disclosed already have a suitable group that can be used as a reactive group; in other cases standard chemical manipulations can be used to modify a dye to comprise a desired reactive group.
[0130] Thus, the present invention provides a composition comprising any of the above-identified compounds, where such compound or compounds have been modified by the addition of a reactive group (Rx) for attachment of a target molecule thereto. The reactive group (Rx) comprises an electrophilic reactive group comprising isocyanate, isothiocyanate, monochlorotriazine, dichlorotriazine, 4,6,-dichloro-1,3,5-triazines, mono- or di-halogen substituted pyridine, mono- or di-halogen substituted diazine, maleimide, haloacetamide, aziridine, sulfonyl halide, acid halide, hydroxysuccinimide ester, hydroxysulfosuccinimide ester, imido ester, hydrazine, azidonitrophenol, azide, 3-(2-pyridyl dithio)-propionamide, glyoxal or aldehyde groups, and a combination of any of the foregoing. In another embodiment, the reactive group (Rx) comprises a nucleophilic reactive group comprising reactive thiol, amine or hydroxyl, and a combination of the foregoing. In other aspects, the reactive group (Rx) comprises a terminal alkene group, a terminal alkyne group, a nickel coordinate group or a platinum coordinate group for attachment. The reactive group (Rx) can be attached to the compound through a linker arm.
[0131] Another aspect of the present invention is a labeled target molecule comprising a target molecule attached to any of the above-described reactive compounds through the reactive group. The target molecule is not narrowly limited to any particular type of molecule, and can comprise any molecule that can be attached to the above-described reactive compounds. Nonlimiting examples of target molecules include a nucleoside, a nucleotide, an oligonucleotide, a polynucleotide, a peptide nucleic acid, a protein, a peptide, an enzyme, an antigen, an antibody, a hormone, a hormone receptor, a cellular receptor, a lymphokine, a cytokine, a hapten, a lectin, avidin, streptavidin, digoxigenin, a carbohydrate, an oligosaccharide, a polysaccharide, a lipid, a liposomes, a glycolipid, a viral particle, a viral component, a bacterial cell, a bacterial component, a eukaryotic cell, a eukaryotic cell component, a natural drug or synthetic drug, and any combination thereof.
[0132] Examples of useful target molecules and solid-phase supports can include but are not limited to a nucleoside, nucleotide, oligonucleotide, polynucleotide, peptide nucleic acid, protein, peptide, enzyme, antigen, antibody, hormone, hormone receptor, cellular receptor, lymphokine, cytokine, hapten, lectin, avidin, streptavidin, digoxigenin, carbohydrate, oligosaccharide, polysaccharide, lipid, liposomes, glycolipid, viral particle, viral component, bacterial cell, bacterial component, eukaryotic cell, eukaryotic cell component, natural drug, synthetic drug, glass particle, glass surface, natural polymers, synthetic polymers, plastic particle, plastic surface, silicaceous particle, silicaceous surface, organic molecule, dyes and derivatives thereof.
[0133] The nucleoside, nucleotide, oligonucleotide, or polynucleotide can comprise one or more ribonucleoside moieties, ribonucleotide moieties, deoxyribonucleoside moieties, deoxyribonucleotide moieties, modified ribonucleosides, modified ribonucleotides, modified deoxyribonucleosides, modified deoxyribonucleotides, ribonucleotide analogues, deoxyribonucleotide analogues or any combination thereof.
[0134] As indicated above, the target molecule of these embodiments may have dyes as targets thereby creating composite dyes. By joining the dyes of the present invention to another dye, unique properties may be enjoyed that are not present in either dye alone. For instance, if one of the dyes of the present invention is joined to another dye such that it creates an extended conjugation system, the spectral characteristics of the dye may be different than either dye component.
[0135] Another example of this method is where the conjugation systems do not overlap but the proximity allows an internal energy transfer to take place thereby extending the Stokes shift, a system that is commonly referred to as FRET (Fluorescent Resonance Energy Transfer) or Energy Transfer in short. For an example of this, see U.S. Pat. Nos. 5,401,847; 6,008,373; 5,800,996, all three of which are hereby incorporated by reference.
[0136] Other properties may also be enhanced by this joining; for example, it has been previously described that the joining together of two ethidium bromide molecules generates a dye that has enhanced binding to nucleic acids and novel fluorescent properties that are different from the monomeric forms (U.S. Patent Application Publication No. 2003/0225247, hereby incorporated by reference). Other composite dyes have been described that simultaneously enjoy both properties, i.e., enhanced binding and energy transfer (U.S. Pat. No. 5,646,264, hereby incorporated by reference). Furthermore, these composites dyes are not limited to binary constructs of only two dyes, but may comprise oligomeric or polymeric dyes. These composite dyes may be comprised of the same dye or different dyes may be joined together depending upon the properties desired.
[0137] Utility may also be achieved by attaching a dye of the present invention to a target specific moiety. Thus, binding between the target specific moiety and its corresponding target may be monitored by essentially determining the presence or amount of dye that is bound to the target. Well-known examples of such assays are hybridizations between complementary nucleic acids as well as binding that take place between antibodies and their corresponding antigens.
[0138] Other binding pairs that may be of interest can include but not be limited to ligand/receptor, hormone/hormone receptor, carbohydrate/lectin and enzyme/substrate. Assays may be carried out where one component is fixed to a solid-phase support and a corresponding partner is in solution. By binding to the component fixed to the support, the partner now becomes attached to the support as well. A well-known example of this method is the microarray assays where labeled analytes become bound to discrete sites on the microarray.
[0139] Homogeneous probe dependent assays are also well known in the art and may take advantage of the present invention. Examples of such methods are energy transfer between adjacent probes (U.S. Pat. No. 4,868,103), the Taqman exonuclease assay (U.S. Pat. Nos. 5,538,848 and 5,210,015), Molecular Beacons (U.S. Pat. Nos. 5,118,801 and 5,925,517) and various real time assays (US Patent Application Publication 2005/0137388), all of which are incorporated by reference.
[0140] Antibodies labeled with dyes of the present invention may be used in various formats. For example, an antibody with one of the dyes of the present invention may be used in an immunofluorescent plate assay or in situ analysis of the cellular location and quantity of various antigenic targets. Antibodies labeled with dyes may also be used free in solution in cell counting or cell sorting methods that use a flow cytometer or for in-vitro and in-vivo imaging of animal models.
[0141] The presence or absence of a signal may then be used to indicate the presence or absence of the target itself. An example of this is a test where it is sufficient to know whether a particular pathogen is present in a clinical specimen. On the other hand, quantitative assays may also be carried out where it is not so much the intention of evaluating if a target is present but rather the particular amount of target that is present. An example of this is the previously cited microarray assay where the particular rise or fall in the amount of particular mRNA species may be of interest.
[0142] In another embodiment of the present invention, dyes that have been disclosed above as well as dyes described previously in the literature may be attached to a carrier with a more general affinity. Dyes may be attached to intercalators that in themselves do not provide signal generation but by virtue of their binding may bring a dye in proximity to a nucleic acid. A further example is attachment of dyes to SDS molecules thereby allowing dyes to be brought into proximity to proteins. Thus this embodiment describes the adaptation of a dye or dyes that lack affinity to a general class of molecules may be adapted by linking them to non-dye molecules or macromolecules that can convey such properties.
[0143] Various applications may enjoy the benefits of binding the dyes of the present invention to appropriate targets. As described above, staining of macromolecules in a gel is a methodology that has a long history of use. More recent applications that also may find use are real time detection of amplification (U.S. Pat. Nos. 5,994,056, 6,174,670 and US Patent Application Publication 2005/0137388, all of which are hereby incorporated by reference), and binding of nucleic acids to microarrays. In situ assays may also find use where the binding of dyes of the present invention is used to identify the location or quantity of appropriate targets.
[0144] In other aspects, this invention provides a composition comprising a solid support to which is attached any of the above-described reactive compounds. In some embodiments, the solid support comprises glass particle, glass surface, natural polymers, synthetic polymers, plastic particle, plastic surface, silicaceous particle, silicaceous surface, glass, plastic or latex beads, controlled pore glass, metal particle, metal oxide particle, microplate or microarray, or any combination thereof. The aforementioned reactive group for attachment comprises or may have comprised an electrophilic reactive group comprising isocyanate, isothiocyanate, monochlorotriazine, dichlorotriazine, 4,6,-dichloro-1,3,5-triazines, mono- or di-halogen substituted pyridine, mono- or di-halogen substituted diazine, maleimide, haloacetamide, aziridine, sulfonyl halide, acid halide, hydroxysuccinimide ester, hydroxysulfosuccinimide ester, imido ester, hydrazine, azidonitrophenol, azide, 3-(2-pyridyl dithio)-propionamide, glyoxal or aldehyde groups, a nucleophilic reactive group comprising reactive thiol, amine or hydroxyl, a nickel coordinate group, a platinum coordinate group, a terminal alkene or a terminal alkyne, and any combination of the foregoing. As in the case of other embodiments previously described above, a linker arm can be usefully positioned between the compound and the reactive group, or between the solid support and the reactive group.
Reagent Kits
[0145] Commercial kits are valuable because they eliminate the need for individual laboratories to optimize procedures, saving both time and resources. They also allow better cross-comparison of results generated from different laboratories. The present invention thus additionally provides reagent kits, i.e., reagent combinations or means, comprising all of the essential elements required to conduct a desired assay method. The reagent system is presented in a commercially packaged form, as a composition or admixture where the compatibility of the reagents will allow, in a test kit, i.e., a packaged combination of one or more containers, devices or the like holding the necessary reagents, and usually written instructions for the performance of the assays. Reagent systems of the present invention include all configurations and compositions for performing the various labeling and staining formats described herein.
[0146] The reagent system will generally comprise (1) one or more dye of the present invention preferably in the form of concentrated stock solutions in an aprotic dipolar solvent, for example, DMSO designed to target specific protein aggregate structures; (2) a buffer, such as Tris-HCl or phosphate buffer; (3) a positive control comprising both protein aggregates and protein monomers in the state of solution or lyophilized powder; and (4) instructions for usage of the included reagents. Generic instruction, as well as specific instructions for the use of the reagents on particular instruments, such as a wide-field microscope, confocal microscope, flow cytometer, high content screening instrument, microplate-based detection platform, RT-PCR instrument or standard fluorometer may be provided. Recommendations regarding filter sets and/or illumination sources for optimal performance of the reagents for a particular application may be provided.
[0147] The dyes, compounds and compositions of the present invention are fluorescently detectable or localized. Techniques and fluorescence methods are well known in the art. A compilation of such techniques and methods are set forth below in Table 3 which was obtained from Hawe et al., 2008.
TABLE-US-00003 TABLE 3 Fluorescence methods and their application with extrinsic fluorescent dyes for protein characterization. Application with Noncovalent Method Information Extrinsic Dyes Steady-state Spectral information Detection of protein fluorescence (emission spectrum and structural changes by fluorescence intensity dye-protein interactions Time-resolved Fluorescence lifetime Detection of protein fluorescence structural changes by dye-protein interactions Anisotrophy Rotational motions Study of rotational (steady-state and dynamics Determination of time-resolved size of dye-protein complexes Fluorescence Translational Determination of size of correlation motions/diffusion dye-protein complexes spectroscopy (FCS) Fluorescence Visualization of Detection of large microscopy particles dye-protein complexes Determination of size and morphology of large aggregates, fibrils, etc.
For an expert review on such fluorescence methods, see the entire above cited publication by Hawe et al., 2008, pp. 1487-1499, the contents of which are incorporated herein by reference.
Protein Aggregation Detection and Analysis
[0148] Fluorescence microscopy allows an early detection of changes in protein solutions, while minimizing alterations to the observed sample after staining with appropriate dyes. In protein formulations, the ability to detect protein aggregates at early time points with the dyes of the present invention can accelerate stability testing and reduce number of samples in long term stability studies. Fluorescence microscopy provides the possibility of studying subtle changes in the aggregation state of the proteins, which is also of interest in medicine and biology, whenever protein characterization is needed. Also, fluorescence microscopy allows the characterization of high-concentration protein formulations without dilution and with minimal impact on the protein's local environment. Furthermore, high-content screening fluorescence-based imaging methods allow quantification of populations of protein aggregates including number of branches, mean fiber length, mean fiber width, size distribution, polydispersity, kinetics of formation and kinetics of disassembly.
[0149] The present invention includes an example of IgG aggregate detection using dyes of the invention by fluorescence microscopy (Examples 2 and 10; FIG. 1). The aggregate formation is barely visible before staining, but clearly becomes visible after staining.
[0150] The dyes of the invention are also capable of detecting a broader range of protein aggregates than the conventional amyloid detecting dyes, such as thioflavin T (Thio-T) or congo red. These styryl dyes are able to sensitively detect protein aggregates, ranging in size (nanometers to visually observable turbid solution to precipitates) and physicochemical characteristics (e.g., soluble or insoluble, covalent or non-covalent, reversible or irreversible). Structurally altered proteins have a strong tendency to aggregate, often leading to their precipitation. Irreversible aggregation is a major concern for long-term storage stability of therapeutic proteins and for their shipping and handling.
[0151] The styryl dyes of the present invention are also able to detect aggregates at different stages of formation induced by various stresses, such as elevated temperature, agitation and exposure to extremes of pH, ionic strength, or various interfaces (e.g., air-liquid interface) and high protein concentration (as in the case of some monoclonal antibody formulations), chemicals and protein-protein interactions (i.e., PDI-insulin interaction). These fluorescent probes are able to detect broad types and concentration ranges of proteins, in the presence of excipients, at different pH values (2.about.10) and in the presence of salts and buffers, exhibiting desirable detection limits and dynamic range, excellent sensitivity as well as linear response. This is exemplified by the broad categories of proteins/peptides system in the present invention, including lysozyme, insulin, and IgG molecules, as well as serum proteins, such as .beta.-lactoglobulin (BLG) and BSA. Therefore, these novel dyes are capable of providing quantitative analysis of protein aggregates in a robust, high throughput fashion.
[0152] Thus, the present invention provides a method for detecting the presence of aggregates of a protein in a sample. The method comprises
[0153] (a) combining the sample with any of the above-described compounds or multidye compositions to form a dye-sample mixture;
[0154] (b) measuring the amount of fluorescence in the dye-sample mixture;
[0155] (c) comparing the amount of fluorescence determined in (b) with the amount of fluorescence in [0156] (i) a mixture of the compound or multidye composition with a control sample without aggregated protein, or [0157] (ii) a mixture of the compound or multidye composition with a known standard quantity of aggregated protein; and
[0158] (d) determining the aggregation of the protein in the sample based on the comparison in (c).
[0159] In these methods, the standard quantity of aggregated protein recited in (c)(ii) can be prepared by any means known in the art. Examples include the provision of a previously determined quantity of aggregated protein, or the preparation of a standard curve derived from measurements of protein aggregates and protein monomers in selected proportions. When a standard curve is utilized, the protein for the standard curve can be the same or different protein as the protein in the sample.
[0160] The sample for this method is not limited to any particular composition. The sample can be from any prokaryote, archaea, or eukaryote, or from an environmental sample. In some embodiments, the sample is from a mammal, for example a bodily fluid of the mammal (e.g., blood [e.g., serum, plasma], bile, sputum, urine, or perspiration).
[0161] In other embodiments, the sample comprises a cell from the mammal. In some of these embodiments, the cell is intact. Such an intact cell, either fixed (see, e.g., Example 28) or living (e.g., Example 29), can be combined with the compound or multidye composition and the fluorescence is measured histologically. Here, the fluorescence can be measured by visual observation or by quantifying the amount of fluorescent light emitted from the cell, by known methods.
[0162] Example 29 exemplifies embodiments utilizing living cells where a compound can be tested for an effect on the aggregation of proteins. In these embodiments, the cell is treated with a protein, for example amyloid beta peptide (e.g., amyloid beta peptide 1-42), known to aggregate in the cell. Such cells treated with the compound can be compared with cells not treated with the compound (the control composition of (c)(ii) in the above-described methods) to determine the effect of the compound on the aggregation of the protein in the cell.
[0163] The sample of these methods can also comprise homogenized cells from a mammal that is part of a tissue from a mammal. In some embodiments, the mammal has a disorder characterized by altered protein aggregation, e.g., Alzheimer's disease, Huntington's disease, Parkinson's disease, senile systemic amyloidosis, or a spongiform encephalopathy.
[0164] These methods can be utilized to detect any known form of aggregated protein, including but not limited to aggresomes, aggresome-like structures, inclusion bodies, Lewy bodies, Mallory bodies, neurofibriliary tangles, or any combination thereof.
Protein Aggregation Kinetic Studies
[0165] Protein aggregation is an important phenomenon that alternatively is part of the normal functioning of nature or has negative consequences via its hypothesized central role in neurodegenerative diseases. A key in controlling protein aggregation is to understand the mechanism(s) of protein aggregation. Kinetic studies, including data curve-fitting, and analysis are, in turn, keys to performing rigorous mechanistic studies.
[0166] The many approaches in the literature striving to determine the kinetics and mechanism of protein aggregation can be broadly divided into three categories: (i) kinetic and thermodynamic, (ii) empirical, and (iii) other approaches. The large literature of protein aggregation can be distilled down to five classes of postulated mechanisms: i) the subsequent monomer addition mechanism, ii) the reversible association mechanism, iii) prion aggregation mechanisms, iv) an "Ockham's razor"/minimalistic model, and v) quantitative structure activity relationship (QSAR) models (Morris et al., 2009). Corresponding equations derived from aggregation kinetic data can enlighten which proposed mechanism is applicable to the specific protein.
[0167] Detection of aggregates at their nascent stages, such as intermediates consisting of a couple of monomers, is key in determining critical nucleus size and aggregate growth mechanism. In addition, kinetic studies are also very helpful in screening excipients or inhibitors that can stop or suppress protein aggregation and in assessing enzyme activity in various clinical and research settings. Hence, a sensitive kinetic assay in a robust, high-throughput manner is highly desirable in mechanism determination studies and in drug discovery. Most of the current aggregate analysis technologies, unfortunately, are neither sensitive nor accurate enough to quantify nascent aggregates. Various factors affecting aggregation can be studied by these means; a number of these are described by Bondos and Bicknell (2003) and in addition, Table 4 below is reproduced from this article (Table 1 therein) showing components (including recommended concentrations) that might be used for decreasing aggregation:
TABLE-US-00004 TABLE 4 Agents that may promote protein solubility Recommended concentration Additive range Kosmotropes MgSO.sub.4 0-0.4M (NH.sub.4).sub.2SO.sub.4 0-0.3M Na.sub.2SO.sub.4 0-0.2M Ca.sub.2SO.sub.4 0-0.2M Weak kosmotropes NaCl 0-1M KCl 0-1M Chaotropes CaCl.sub.2 0-0.2M MgCl.sub.2 0-0.2M LiCl 0-0.8M RbCl 0-0.8M NaSCN 0-0.2M NaI 0-0.4M NaClO.sub.4 0-0.4M NaBr 0-0.4M Urea 0-1.5M Amino acids Glycine 0.5-2% L-arginine 0-5M Sugars and Sucrose 0-1M polyhydric Glucose 0-2M alcohols Lactose 0.1-0.5M Ethylene glycol 0-60% v/v Xylitol 0-30% w/v Mannitol 0-15% w/v Inositol 0-10% w/v Sorbitol 0-40% w/v Glycerol 5-40% v/v Detergents Tween 80 0-0.2% w/v Tween 20 0-120 .mu.M Nonidet P-40 0-1%
[0168] The method described above can be adapted to measure the kinetics of protein aggregation, e.g., by measuring fluorescence of the protein-dye mixture at various time points while aggregation is occurring. Thus, in some embodiments, the amount of fluorescence of the above-described method is measured at preselected time intervals to detect formation of protein aggregates, wherein increasing fluorescence over time indicates formation of protein aggregates.
[0169] These embodiments encompass two methods of applying the above-described dyes into a kinetics study of protein aggregation, such as lysozyme and IgG aggregation, induced by various types of stress, including pH, shaking and temperature shift and in the presence or absence of excipient(s). The first method comprises the following steps: (1) apply a stress to a protein formulation for a certain period of time; (2) release stress by switching off the stress, such as heat or harsh pH to freeze or trap the aggregate formation; (3) fluorescence reading of these formulations by addition of selected dyes of the invention; (4) plot the relative fluorescence unit (RFU) vs. time curve and further process the kinetic curve to extract more desired information. This method is beneficial for some proteins whose aggregation can be significantly interfered with by probing dye binding (especially for nascent or intermediate aggregates, characterized by a much smaller surface area than those more matured aggregates) at stressed condition, which is minimized after the release of the stress.
[0170] The second method is more convenient compared to the first method. First, mix the dye with the protein formulation prior to the application of the stress; second, apply the stress and start recording the fluorescence response at various points of time; finally, plot a relative fluorescence unit (RFU) vs. time curve and possibly perform further processing of the curve to extract more desired information. This method, though labor saving, much more robust and accurate in time, may not be applicable for some proteins if the dye blocks, promotes or interferes with the addition of monomers to the aggregate intermediates or polymerization of aggregate intermediates. However, notwithstanding the mentioned caveats, the second method is generally preferred, since it allows for a simpler high throughput assay.
[0171] The measurement of fluorescence in these methods can be conducted using any appropriate time interval between measurements, determined by a determination of the time expected for the aggregation to occur in the particular system being investigated. In some embodiments, the preselected time intervals are less than 2 minutes. In other embodiments, the preselected time intervals are less than 10 minutes. In still other embodiments, the preselected time intervals are less than 1 hour. In additional embodiments, the preselected time intervals are more than 1 hour.
Methods of Evaluating Protein Formulation Stability Using Accelerated Stability Testing
[0172] Embodiments of the present invention are directed to reliable, time and cost-efficient methods for evaluating the relative chemical and physical stability of a particular protein formulation. Thus, embodiments of the invention are useful analytical tools for developing new protein formulations with increased stability, as well as for use in evaluating the stability of newly prepared batches of known protein formulations in quality control procedures, or the like.
[0173] Embodiments of the present invention encompass a fully automated assay of protein stability that generally requires less than one week for evaluating protein formulations. The present invention method comprises preparing two series of formulations, one formed before stress test (pre-stress formulations), another formed after stress test (post-stress formulations), followed by an adding aggregate detection reagent that include one or more dyes of the present invention. The dye or dyes of the present invention may be used alone for this purpose oror they may be used in conjunction with other commercial dyes, such as Nile red, thioflavin-T, ANS or Congo red. This is followed by comparing the fluorescence response of different formulations to rank the amount of aggregates existing within individual formulations.
[0174] In one exemplification of this method, the following 6 steps may be carried out:
[0175] Step (1). A selected group of components, including, but not limited to excipients, salts, buffers, co-solvents, metal ions, preservatives, surfactants, and ligands are collected and their stock solutions are prepared.
[0176] Step (2). Preliminary formulations comprising one or more components following a standard design of experiment procedure aimed at generating relevant information are designed and the protein formulations, preferably containing the same concentration of protein are prepared.
[0177] Step (3). A stress such as heat, agitation, rotation, harsh pH, ultrasound, shearing or the like, is simultaneously applied externally to multiple protein formulations under evaluation, which are held in individual containers, preferably in separate wells of one microplate (s), which is preferably sealed, each with zero, one or more components of interests; meanwhile, the formulation with zero component of interests, but the same protein concentration as the formulations with component (s) of interests can be prepared in a separately sealed container in bulk quantity.
[0178] Step (4). After stress is released, the bulk protein formulation that has zero components of interests is split and mixed with one or more components of interests to make up similar formulations as those subjected to the stress test, preferably in wells of another microplate. Note that the later added components of interest solutions dilute the resulted non-stressed formulations, making them less concentrated as their stressed counterpart; this can be adjusted later in the step where the probing dyes are added. These control formulations which have not experienced the stress test allow accurate evaluation of the functions of the components of interests during the stress test since components of interests themselves can affect the fluorescence response of protein aggregates to some extent.
[0179] Step (5). A solution of the dye or dyes of the present invention (and the buffer in which the dyes are dissolved) are added into the protein formulations such that post-stress formulations are more concentrated than that added to the stressed formulations to result in the same concentration of protein, components of interests and dye(s) for both pre-stress formulation and post-stress counterpart. After an incubation period, the microplates are read in a conventional plate reader by, for example, fluorescence intensity or fluorescence polarization measurement.
[0180] Step (6). The formulations can be first evaluated within the group (i.e. either pre-stress or post-stress formulations), which are preferably tested in one microplate, by comparing formulations containing one or more components of interests with that containing no components of interests. This method can eliminate the errors produced during the preparation of different plates (the sample formulation plate(s) and the control formulation plate(s), which can take 10.about.60 minutes. Then fluorescence ratio of each stress tested formulation to its corresponding control without stress application can be further calculated. The function of components of interests during stress is evaluated by using the fluorescence ratio of components of interests added before application of stress vs. after application of stress using zero components of interests as a reference. Therefore, the present invention is further directed to a method to evaluate components of interests that can stabilize or destabilize protein in order to optimize protein formulations.
[0181] The properties of the dyes of the invention allow their wide application in the protein/peptide formulation field, especially on a high-throughput technology platform. Compared with other fluorescent probes, such as intrinsic tyrosine or externally added probes, such as 1-anilino-naphthalene-8-sulfonate (ANS), Nile red or thioflavin-T, the dyes of the present invention are better capable of providing quantitative analysis of protein aggregates in a robust, high throughput fashion and are applicable to more categories of proteins under various conditions. In some instances two or more dyes of the present invention are applied to a sample. This facilitates detection of the broadest range of protein aggregates since these means provide that if one dye does not bind a particular aggregate, another can compensate for this deficiency.
Protein Stability Shift Assay Based on Fluorescent Detection of Protein Aggregation Using Exogenously Added Fluorophores
[0182] Protein stability can be altered by various components discussed in protein formulation embodiments, including, but not limited to excipients, salts, buffers, co-solvents, metal ions, preservatives, surfactants, and ligands. Protein stability can be shifted by various stresses, including elevated temperature, which is often referred to as a thermal shift or by addition of chemical denaturants, such as urea, guanidinium isocyanate or the like. A protein stability shift assay has a wide spectrum of applications in, but not limited to investigation of protein refolding conditions, optimization of recombinant protein expression/purification conditions, protein crystallization conditions, selection of ligand/drug/vaccine/diagnostic reagents and protein formulations.
[0183] The classic thermal shift technologies based on protein aggregate detection utilize a melting point device to raise the temperature stepwise, coupled with aggregation detection technologies, such as light scattering technology (an example includes but is not limited to differential static light scattering (DSLS)) to monitor protein aggregation. This type of technology usually requires a high protein concentration, therefore, it is not cost effective. In addition, it cannot work effectively on formulations with low protein concentrations or finalize protein formulations which require a very low detection limit for aggregates (typically .about.1-5%), which is usually beyond the detection limit of these classic technologies.
[0184] Thermofluor.RTM. (J&J, 3-Dimensional Pharmaceuticals, Inc, Exton, Pa., U.S. Pat. No. 6,020,141 ["the '141 patent"]) is a biophysical technique used to study (relative) protein stabilities. The solution of protein is heated up stepwise from room temperature to .about.95.degree. C. and the fluorescence is monitored at each step. The rising temperature causes protein unfolding and the fluorophore (SYPRO Orange.RTM. [Invitrogen] or ANS) partitions itself into the melted protein and hence the overall effect is an increase in fluorescence with increasing temperature. If a drug or ligand is included which binds to the protein, the mid-point of the curve can shift, arising from stabilizing or destabilizing effects (e.g., ligand binding). Thermofluor.RTM. can rank binding affinity in a rapid, HTS manner and help setup structure-activity relationship. However, this particular methodology is related to both denaturation of proteins as well as subsequent aggregations of the denatured proteins and the patent clearly indicates that the focus is on the unfolding and denaturation of proteins and as described in column 16, lines 25-56, the fluorescent probes chosen for application of this method are drawn from compounds that are "capable of binding to an unfolded or denatured receptor". However, some of the compounds that are listed (ANS, bis-ANS and JCVJ) are known to bind to aggregates (Lindgren et al., 2005) and as such no particular emphasis is laid upon distinguishing between denaturation and aggregation events. In contrast, the present invention is specifically directed towards aggregation detection.
[0185] As such, one of the embodiments of the present invention encompasses a novel thermal shift assay in which protein is heated up stepwise from room temperature to .about.95.degree. C. using a device, including, but not limited to, a microplate reader, a thermocycler, a melting device or similar equipment, preferably with a heating stage that can raise temperature stepwise and record fluorescence change simultaneously, and the fluorescence of externally added dyes of the present invention is monitored at each heating step. Since the dyes that are used in the present invention selectively interact with protein aggregates and not hydrophobic domains exposed by protein unfolding, the increase in fluorescence with increasing temperature is not due to protein unfolding as seen in the technique described in the '141 patent, but rather is due to protein aggregation. Therefore, this particular embodiment of the present invention can be applied to directly targeting at ranking components, including, but not limited to, excipients, salts, buffers, co-solvents, metal ions, preservatives, surfactants, and ligands in protein stabilization by preventing protein aggregation to improve formulations, or to screening drugs (inhibitors) preventing protein aggregates found in some diseases, including, but not limited to, organic synthetic compounds, peptides and proteins (recombinant or natural source). For most proteins, unfolding directly precedes their aggregation. Hence, similar to the unfolding-based Thermofluor.RTM. technique, the aggregation-based thermal shift assay technology embodied in this present invention also has the potential to being applied to ranking the effect of additives on protein stability. Its application can thus be expanded to more broad fields, including, but not limited to, investigation protein refolding conditions, optimization of recombinant protein expression/purification conditions, protein crystallization conditions, and selection of ligands, drug, vaccine and diagnostic reagents.
[0186] Thus, fluorescence can be measured at one or more different temperatures after forming the first mixture and the second mixture. Such different temperatures can be selected from temperatures ranging from about 4.degree. C. to about 100.degree. C. Further, fluorescence measurements can be carried out as a series of discrete temperatures, where the measuring steps are carried out after incubation at each of the different discrete temperatures, or while changing temperatures.
[0187] Another useful method of the present invention is a method for determining whether a test compound decreases aggregation of a protein. The above-described method can be utilized, where a test compound is added to a portion of the dye-sample mixture of (a) and the fluorescence of the portion with the test compound is compared to the fluorescence of the portion without the test compound to determine whether the test compound decreases aggregation of the protein, wherein decreased fluorescence in the portion with the test compound indicates that the test compound decreases aggregation of the protein.
[0188] The test compound is not limited to any particular class of compound. Nonlimiting examples include a kosmotrope, a chaotrope, an amino acid, a peptide, a reducing agent, a carbohydrate, a detergent, a surfactant, a zwitterion or a polyhydric alcohol, or any combination thereof. Any of these test compounds can have a range of concentrations from about 0 molar to about 2 molar, a range of pH values from about 4 to about 10. The test compound can also comprise a storage buffer for said protein. Such storage buffer can comprise a set of buffer formulations with a range of concentrations from about 0 molar to about 2 molar, a range of pH values from about 4 to about 10, and any combinations thereof.
Chaperone/Anti-Chaperone Activity Assays
[0189] Chaperone and anti-chaperone function oppositely in the sense that one helps prevent aggregates and the other helps induce aggregate formation. To assay activity of the opposite functions, one needs to quantitatively analyze the substrate aggregate change with time. The present invention uses methods described above in the PDI/thioredoxin activity assay to analyze chaperone/anti-chaperone activity, which has similar advantages over methods based on other aggregate detection technologies, particularly turbidity and back-scatter methods. The present invention also encompasses a kit or kits comprising similar components as the PDI isomerase activity kit (s) included in the present invention. Assays can be devised to monitor assembly or disassembly of protein aggregates or both.
[0190] Thus, in some embodiments of the above-described method,
[0191] (A) the protein is a substrate for a chaperone;
[0192] (B) the dye-sample mixture of step (a) is subjected to a stress for a time and under conditions sufficient to induce aggregation of the protein; and
[0193] (C) the amount of fluorescence determined in (b) is compared to the amount of fluorescence from the protein with the compound or multidye composition subjected to the same stress without the sample. In these embodiments, a decrease in fluorescence of the stressed dye-sample mixture with the sample when compared to the fluorescence from the protein with the compound or multidye composition but without the sample indicates that the sample comprises the chaperone.
[0194] This method can utilize any chaperone now known or later discovered, including chaperones that are small heat-shock proteins (sHSPs), as they are known in the art. Examples of chaperones include HSP33, HSP60, HSP70, HSP90 or HSP100, GRP94, GRP170, calnexin, calreticulin, HSP 40, HSP47 and ERp29, GroEL, GroES, HSP60, Cpn10, DnaK, DnaJ, Hsp70, Hsp71, Hsp72, Grp78 (BiP), PDI, Erp72, Hsx70, Hdj1, Hdj2, Mortalin, Hsc70, Hsp70-A1, fHtpG, C62.5, Hsp90 alpha, Hsp90 beta, Grp94, ClpB, ClpA, ClpX, Hsp100, Hsp104, Hsp110, TRiC, alpha crystallin, HspB1, Hsp 25, Hsp27, clusterin, GrpE, Trigger Factor, and Survival of Motor Neuron (SMN1, SMN2), or any combination thereof. The substrate can comprise any chaperone substrate now known or later discovered, including but not limited to .beta.-lactoglobulin, citrate synthase, lysozyme, immunoglobulin, CRYBB2, HSPB8, CRYAA, TGFB1I1, HNRPD or CRYAB, or any combination thereof. The reaction mixture can be incubated for a period of time from about 15 to about 60 minutes. The stress can be an elevated temperature, preferably, from about 37.degree. C. to about 95.degree. C. Alternatively, the stress can be a chaotropic agent, such as guanidine-HCl or urea, or both. The concentration of the chaotropic agent can be from about 4 to 8 M. Moreover, a plurality of these methods can be performed in parallel.
[0195] Analogously, the invention methods can be utilized to identify anti-chaperone activity. Here, the methods described above are utilized, where
[0196] (A) the protein is a substrate for an anti-chaperone; and
[0197] (B) the amount of fluorescence determined in (b) is compared to the amount of fluorescence from the protein with the compound or multidye composition without the sample. In these methods, an increase in fluorescence of the dye-sample mixture when compared to the fluorescence from the protein with the compound or multidye composition but without the sample indicates that the sample comprises the anti-chaperone.
High-Throughput Fluorometric Assay for Measuring Aggregates Formed by Members of the Thioredoxin Superfamily
[0198] Thioredoxins and related proteins act as antioxidants by facilitating the reduction of other proteins by cysteine thiol-disulfide exchange. Such exchanges can lead to intermolecular bridges being formed, thereby forming covalently linked aggregates. Thioredoxins are characterized at the level of their amino acid sequence by the presence of two vicinal cysteines in a CXXC motif. These two cysteines are the key to the ability of thioredoxin to reduce other proteins. A number of different families (thioredoxins, protein disulfide isomerases [PDI's] and glutaredoxins) form what can be considered the thioredoxin superfamily. With regard to the glutaredoxins, they share many of the functions of thioredoxins, but are reduced by glutathione rather than a specific reductase and may be assayed by the described methods of the present invention.
[0199] Thus, methods are disclosed to measure the activity of thioredoxin-like enzymes by detecting the induction of aggregates formation by utilizing any of the dyes described above. The above-described method for detecting a protein aggregate can be utilized, where
[0200] (A) the protein is a substrate for a member of the thioredoxin superfamily;
[0201] (B) a reducing agent is included in the dye-sample mixture of (a); and
[0202] (C) the dye-sample mixture of step (a) is incubated for a period of time sufficient to reduce disulfide bonds in the protein. In these methods, an increase in fluorescence of the dye-sample mixture when compared to the fluorescence from the protein with the compound or multidye composition without the sample indicates that the sample comprises the member of the thioredoxin superfamily.
[0203] Substrates here include, but are not limited to, insulin, hypoxia-inducible factor, prolyl 4-hydroxylase, HIV gp120, TXNIP, ASK1, collagen type I, alpha 1 and glucocorticoid receptor. In some embodiments, insulin is used as a substrate at a concentration of less than 0.2 mM. This method can be used to measure the amount of activity in a sample, identify the suitability of proteins as substrates for such activity, and to screen for inhibitors of these enzymes. This method may also be used to test the ability of a particular protein to be used as a substrate by a member of the thioredoxin superfamily to form aggregates. This method also allows an accurate assay of multiple samples, such as samples from patients, or therapeutic agents for drug discovery. The method can be used in a high throughput manner using a microplate, as reflected in the insulin aggregate detection example included in the present invention.
[0204] The reducing reagent concentration should be optimized to reduce the substrate disulfide bonds without minimizing the competing chemical reaction. The reducing reagents may include, but are not limited to glutathione, dithiothreitol (DTT), dithioerythritol, .beta.-mercaptoethanol, thioglycolate, and cysteine, with DTT being a preferred embodiment. A preferred DTT concentration is less than 10 mM, and more preferably less than 1 mM. The assay buffer can include those buffers that stabilize thioredoxin superfamily members and their substrates, with optimized pH, salts, chelating agents (e.g. EDTA, and the like), dyes, and potentially organic solvents such as DMSO.
[0205] When testing for the presence or amount of a particular member of the thioredoxin superfamily in a sample (or for overall activity), a variety of sources may be used that include biological tissues, biological fluids and cells. Thus for instance, samples may include cells up-regulating PDI during hypoxia or cells with surface expressed PDI, including endothelial cells, platelets, lymphocytes, hepatocytes, pancreatic cells and fibroblasts. The sample may also include a thioredoxin superfamily member complexed with other proteins, such as PDI complexed with hypoxia-inducible transcription factor HIFa. Samples may also include fragments of a member of the thioredoxin superfamily as well as recombinant forms of these members, and in vitro protein synthesis reactions that are presumed to have generated such proteins.
[0206] These methods may also find utility in identifying modulators of thioredoxin superfamily activity; such modulators can comprise enzyme mimetics, interacting proteins, competitive inhibitors, small molecular inhibitors, and the like.
[0207] The method may also comprise the use of appropriate controls for the sample, including controls that do not include any thioredoxin superfamily member activity as well as controls that do not include any reducing reagents. These controls can be used as background to be subtracted from gross signal to gain net signal induced by the enzyme activity.
[0208] A preferred addition sequence of the present invention is: (1) Substrate and related buffers; (2) Dye(s) dissolved in organic solvent(s), (3) PDI or similar thioredoxin-like enzyme (s) and related buffers; (4) Reducing reagent (s). The enzyme(s) and reducing reagents are preferred to be added with a multi-channel addition device that can simultaneously add reducing agent into the multiple assay containers, such as wells of a microplate to minimize the time interval between the addition of enzyme and the reducing reagent. This may be important for kinetic assays under some circumstances since PDI and similar thioredoxin-like enzymes can induce enzymatic reaction in the absence of reducing agent, especially with a high concentration of enzyme or substrate or both. This can minimize the background levels. The multi-channel addition device can minimize the background levels derived from the foregoing effects it may also minimize timing errors among the multiple samples to be analyzed, which can minimize statistical deviation among the samples.
[0209] In addition to the methods described above, the thioredoxin superfamily aggregation assays can be formulated into kits comprising one or more thioredoxin superfamily members, appropriate substrates, buffers, reducing agents and one or more dyes of the described in FIG. 1 as well as instructions for their use. These kits may be used for any of the applications described above.
[0210] Such member of the thioredoxin superfamily (a) can comprise a protein disulfide isomerase, a thioredoxin or a glutaredoxin, and combinations thereof. The substrate (b) in this method can comprise insulin ribonuclease, choriogonadotropin, coagulation factor, glucocorticoid receptor or HIV gp 120, and combinations thereof. The reducing agent (c) can be selected from the group comprising dithiothreitol (DTT), Tris(2-carboxyethyl)phosphine hydrochloride (TCEP HCl) or dithioerythritol (DTE), and combinations thereof. The reaction mixture can be preferably incubated for a period of time from about 15 to about 60 minutes. The protein disulfide isomerase can comprise PDI, ERp57, PDIp, ERp72, P5, PDIr, ERp28/29, ERp44, ERjd5/JPDI or ERp18, and combinations thereof.
[0211] This method can further comprise the step of terminating the reaction prior to the measuring step (iii) by adding hydrogen peroxide to the incubating reaction mixture. As in the case of earlier described embodiments of this invention, a plurality of such methods can be performed in parallel.
Assaying Various Enzymatic Activities and Post-Translational Modifications by Monitoring Protein Aggregation Status
[0212] With respect to various pathological disorders, abnormal protein aggregates are often sequestered into intracellular protein deposits such as aggresomes, aggresome-like structures, inclusion bodies. Lewy bodies or Mallory bodies (Stefani, 2004; Garcia-Mata et al., 2002). These may trigger in turn the expression of inflammatory mediators, such as cyclooxygenase 2 (COX-2) (Li et al., 2004). Disruption of the ubiquitin-proteasome pathway, as for example, thru impairment of ubiquitin hydrolase activity, triggered by modulators such as .DELTA.12-PGJ2, lactacystin .beta.-lactone or MG-132 can readily be analyzed directly in cells using the disclosed methods to detect intracellular protein deposits as well as in either cell-based or biochemical assays for screening of other selective inhibitors of the ubiquitin-proteasome pathway that lead to protein aggregation.
[0213] The principle advantages of the delineated approach relative to use of conventional substrates of ubiquitin hydrolase activity, such as ubiquitin-7-amino-4-methylcoumarin (ubiquitin-AMC), include employment of a natural protein substrate in the assay as well as an inherent signal amplification, arising from the formation of polymerized amyloid fibrils as reporters. Examples of potential protein substrates useful in this regard include, but are not limited to, immunoglobulin, .alpha.-synuclein, synphilin-1, TCR.alpha., P23H mutant of rhodopsin, .DELTA.F508 mutant of CFTR, amyloid-.beta., prion protein, Tau, SOD1, Ig light chains, ataxin-1, ataxin-3, ataxin-7, calcium channel, atrophin-1, androgen receptor, p62/sequestosomel (SQSTM1), Pael receptor, serum amyloid A, transthyretin, .beta.2-microglobulin, apolipoprotein A-1, gelsolin, atrial natriuretic factor, lysozyme, insulin, fibrinogen, crystallins, surfactant protein C, lactoferrin, .beta.ig-h3, PAPB2, corneodesmosin, neuroserpin, cochlin, RET, myelin, protein 22/0, SCAD, prolactin, lactadherin, p53, procalcitonin, cytokeratins, GFAP, ATP7B, prolyl hydroxylase PHD3, presenilin, and huntingtin. Additionally, proteins specifically engineered to be unstable or highly prone to self-association into aggregates may be employed as substrates using the disclosed assay methods.
[0214] With respect to coupled enzyme reactions the product of one reaction is used as the substrate of another, more easily-detectable reaction. The cited compositions and methods are especially advantageous in the development of biochemical assays involving coupled reactions leading to the formation of protein aggregates. In this instance no meaningful physiological relationship between the activity being monitored and the generation of the aggregated protein-dye reporter is explicitly required. The protein aggregate-dye complex is simply serving as an indicator to establish the amount of product formed in a particular catalytic reaction. For example, a protein substrate may be employed that is marginally stable under the specified solution conditions employed in the assay. When this substrate is acted upon by a histone acetyltransferase, a particular lysine residue becomes acetylated, the overall protein structure is destabilized and the protein undergoes a conformational change resulting in its aggregation. The dyes described in this disclosure are then able to bind to the aggregates, generating a fluorescent signal. While illustrated with histone acetyltransferase, a wide range of activities that could potentially modify a substrate protein, leading to its structural destabilization under the assay conditions employed, could be performed by similar approaches. In addition activities that do not directly modify the substrate protein can also be considered. For instance, an enzyme activity that leads to the acidification of the assay buffer could in turn lead to destabilization of the substrate protein structure and its aggregation.
Separation of Protein Aggregates from Protein Monomers
[0215] Those skilled in the art will appreciate that the present invention is applicable to the separation or isolation of protein aggregates from other protein forms, notably protein monomers. The dyes described above are useful in subtraction of protein aggregates from protein monomers.
[0216] Thus, the present invention provides a method for separating aggregates of a protein from monomeric forms of the protein. The method comprises
[0217] (a) combining the sample to the solid support of claim 25 under conditions where aggregates of the protein preferentially bind to the compound;
[0218] (b) separating sample protein bound to the solid support from unbound protein. In these methods, protein bound to the solid support is substantially aggregates and unbound protein is substantially monomers.
[0219] In carrying out the above isolation method, the solid support can comprise glass particle, glass surface, natural polymers, synthetic polymers, plastic particle, plastic surface, silicaceous particle, silicaceous surface, glass, plastic or latex beads, controlled pore glass, metal particle, metal oxide particle, microplate or microarray, and combinations of any of the foregoing.
[0220] Preferred embodiments are described in the following examples. Other embodiments within the scope of the claims herein will be apparent to one skilled in the art from consideration of the specification or practice of the invention as disclosed herein. It is intended that the specification, together with the examples, be considered exemplary only, with the scope and spirit of the invention being indicated by the claims, which follow the examples.
Example 1. Testing Compounds for Ability to Sense Protein Aggregation
[0221] Fluorescent readings were carried out in 50 mM Tris-HCl, pH 7.8 using 10 .mu.M dye. When present, 1 .mu.M recombinant human .alpha.-synuclein (ASN, Sigma-Aldrich, St. Louis, Mo.) as monomers, or aggregated as described in van Raaij et al. (2006) was included. Fluorescence excitation and emission spectra were collected on a Cary Eclipse fluorescence spectrophotometer (Varian, Australia). Fluorescence spectra were measured with excitation and emission slit widths set to 5 nm, and at a constant PMT voltage. Spectroscopic measurements were performed in standard quartz cells. All measurements were made at the respective excitation maxima of each dye. All measurements were carried out at room temperature. Results are summarized in Tables 1 and 2.
Example 2. Fluorescence Sensitivity of Different Protein Aggregate-Sensing Dyes in the Presence of Excipients
[0222] IgG aggregate was prepared by adjusting 5.83 mg/ml of purified goat-anti-mouse IgG (H&L, Pel Freez, Rogers, Ark.) to pH 2.7 using HCl and incubating at 22.degree. C. for 24 hours. The assay was performed using 2.8 .mu.M IgG, either native or aggregated, and a dye concentration of 0.625 .mu.M. The protein and dye were mixed together for 15 minutes at 22.degree. C., then further incubated in the presence of the excipients shown in Table 5. The fluorescence intensity of S-25, Tol3 and Y2150 were determined with a FLUOstar OPTIMA plate reader (BMG LABTECH) at excitation wavelength of 550 nm and emission wavelength of 610 nm; while the fluorescence intensity for thioflavin-T was determined using a SpectraMAX GeminiXS (Molecular device, with Softmax Pro 7.0) using an excitation wavelength of 435 nm and emission wavelength of 495 nm. The fluorescence enhancement (aggregate/native IgG) is shown.
TABLE-US-00005 TABLE 5 Effect of various concentrations of various excipients on fluorescence of four dyes in the presence of aggregated IgG. Excipients & Concentrations S25 TOL3 Y2150 Thio-T Sodium Chloride, 10 mM 14.0 16.0 14.4 1.6 Sodium Chloride, 100 mM 13.6 16.2 11.3 1.3 Sodium Chloride, 1000 mM 11.7 17.4 15.0 2.7 Calcium Chloride, 10 mM 9.7 14.9 12.4 3.1 Calcium Chloride, 50 mM 9.6 13.9 14.7 1.5 Calcium Chloride, 200 mM 6.7 14.8 13.9 1.7 Ammonium Sulfate, 10 mM 15.4 15.6 12.4 2.8 Ammonium Sulfate, 100 mM 14.6 13.4 12.5 2.6 Ammonium Sulfate, 300 mM 13.3 16.9 14.6 1.4 Sorbitol, 100 mM 16.4 20.0 17.3 3.0 Sorbitol, 300 mM 21.0 19.2 15.6 1.9 Sorbitol, 600 mM 25.4 29.3 18.7 3.6 Mannitol, 100 mM 16.7 17.5 11.2 3.1 Mannitol, 300 mM 15.2 25.2 13.8 3.7 Mannitol, 600 mM 20.9 27.5 17.7 1.8 Trehalose, 100 mM 17.8 18.9 14.0 2.2 Trehalose, 300 mM 32.1 20.1 19.4 0.2 Trehalose, 600 mM 30.1 30.4 18.9 4.8 Lactose, 100 mM 23.0 19.9 17.5 1.2 Lactose, 300 mM 38.9 34.6 31.0 1.4 Ascoric Acid, 1 mM 13.9 15.4 14.5 1.5 X100, 0.01% 19.2 6.2 4.1 5.3 X100, 0.2% 7.3 3.4 2.6 6.6 X100, 1% 2.9 1.9 1.7 3.4 Arginine, 200 mM 14.8 18.6 14.4 1.4 Arginine, 500 mM 13.5 17.6 14.3 2.0 Glycine, 0.5% 14.1 16.3 12.5 3.1 Glycine, 2% 15.1 15.5 19.0 3.2 Tween 20, 0.01% 70.8 8.5 5.5 4.6 Tween 20, 0.2% 26.7 3.4 2.6 2.6 DTT, 1 mM 13.2 13.8 11.3 1.6 Average 19.3 17.6 14.8 2.9
Example 3. Synthesis of S25
(a) Preparation of 6-methylsulfonyloxyhexyl methylsulfonate (Compound 1)
[0223] A solution of 1,6-hexanediol (13.15 g, 111.3 mmol) in 70 mL of anhydrous pyridine was cooled to 0.degree. C. using ice bath. To this methanesulfonyl chloride (27 g, 235.7 mmol) was slowly added under mixing such that the temperature was maintained at 5-6.degree. C. The combined mixture was stirred overnight at the temperature below 10.degree. C. and the precipitate formed was filtered off, washed with 20% HCl (3.times.), water (3.times.), 5% solution of sodium bicarbonate (3.times.), and then again with water (3.times.). Product was dried under vacuum to obtain Compound 1 as a white solid (yield 32.8%). The structure of Compound 1 is given below:
##STR00142##
(b) Preparation of Compound 2
[0224] A mixture of 4-methylpyridine (3.06 g, 32.9 mmol) and Compound 1 (4.11 g, 15 mmol) was heated at 120.degree. C. for 3 hours. The reaction mixture was cooled and then 4 mL of isopropyl alcohol was added and the combined mixture was refluxed for an hour. After cooling the precipitate was collected by centrifugation, washed with isopropyl alcohol (40 mL, 3.times.), followed by diethyl ether (40 ml, 3.times.) and dried under vacuum overnight to provide Compound 2 (yield 85%) which was then used without any further purification. The structure of Compound 2 is given below:
##STR00143##
(c) Preparation of S-25
[0225] To a suspension of Compound 2 (1.38 g) in n-butanol (15 mL), p-dimethylaminobenzaldehyde (0.9 g) was added and the combined mixture was stirred until it became homogeneous. To this mixture .about.24 drops of piperidine was added and it was refluxed for 4.5 hours. Upon cooling, the precipitate formed was collected by centrifugation, washed with isopropyl alcohol (40 ml, 3.times.), diethyl ether (40 ml, 2.times.) and dried under vacuum to provide dye S25 in a yield of about 68%. Abs=485 nm, Em=613 nm. The structure of S25 is given below:
##STR00144##
Example 4. Synthesis of Tol3
(a) Preparation of Compound 3
[0226] A mixture of 3,4-dimethylpyridine (1.18 g, 11 mmol) and 1,10-diiododecane (1.97 g, 5 mmol) was alloyed during 3 hours at 120.degree. C. To the reaction mixture 5 mL of isopropyl alcohol was added and the mixture was refluxed for an hour. Upon cooling, the solvent was decanted, and the residue thus obtained was washed with cold diethyl ether (40 ml, 2.times.), followed by centrifugation to remove residual solvents. The solid obtained was then dissolved in methanol (.about.4 mL) and dropwise added to cold diethyl ether. Precipitated product was collected by centrifugation, washed with diethyl ether (40 ml, 3.times.) and dried under vacuum to provide Compound 3 in 88% yield. This product was used without any further purification. The structure of Compound 3 is given below:
##STR00145##
(b) Preparation of Tol3
[0227] A mixture of Compound 3 (0.61 g), p-dimethylaminobenzaldehyde (0.3 g) and 6.about.8 drops of piperidine in 5 mL of n-butanol was refluxed for 4 hours. After cooling the precipitated solid was collected by centrifugation, washed first with isopropyl alcohol (40 ml, 3.times.), diethyl ether (40 ml, 2.times.) and then again isopropyl alcohol (40 ml, 1.times.) and diethyl ether (40 ml, 3.times.). The product was dried under vacuum to provide dye Tol3 in 82% yield. Abs=471 nm, Em=611 nm. The structure of Tol3 is given below:
##STR00146##
Example 5. Synthesis of S43
(a) Preparation of 1,1'-(1,2-phenylenebis(methylene))bis(4-methyl pyridinium) bromide (Compound 4)
[0228] A mixture of 4-methylpyridine (1.02 g) and 1,2-bis-bromomethyl-benzene (1.32 g) was heated during 2.5 hours at 120.degree. C. To the reaction mixture 5 mL of isopropyl alcohol was added and the mixture was refluxed for 2 hours. After cooling the product was filtered, washed with diethyl ether and dried under vacuum to provide Compound 4 in 87% yield. The structure of Compound 4 is given below:
##STR00147##
(b) Preparation of S43
[0229] A mixture of Compound 4 (0.45 g), p-dimethylaminobenzaldehyde (0.3 g) and 6 drops of piperidine in 5 mL of n-butanol were refluxed for 4 hours. After cooling the product was filtered and washed with isopropyl alcohol and diethyl ether. The residue obtained was recrystallized from the DMF-methanol mixture to provide S43 in 72% yield. Abs=527 nm, Em=637 nm. The structure of S43 is given below:
##STR00148##
Example 6. Synthesis of Yat 2134
(a) Preparation of 1,1'-(butane-1,4-diyl)bis(4-methylpyridinium) iodide (Compound 5)
[0230] A mixture of 4-methylpyridine (1.02 g) and 1,4-diiodobutane (1.55 g) in 5 mL of dioxane was refluxed for 8 hours. The obtained salt was precipitated with diethyl ether and filtered. The precipitate was washed with ether and dried under vacuum to provide Compound 5 in 91% yield. This product was used without any further purification. The structure of Compound 5 is given below:
##STR00149##
(b) Preparation of Yat 2134
[0231] This procedure was carried out as described previously in step (b) of Example 3 with Compound 5 (0.5 g), piperidine (.about.6 drops), p-diethylamino benzaldehyde (0.36 g) and n-butanol (5 mL). Purification was carried out by recrystallization from DMF-methanol mixture to provide Yat 2134 in 70% yield. Abs=500 nm, Em=620 nm. The structure of Yat 2134 is given below:
##STR00150##
Example 7. Synthesis of Yat 2148
[0232] A mixture of Compound 4 [0.45 g, obtained in step (a) of Example 3], p-diethylaminobenzaldehyde (0.36 g) and 6 drops of piperidine in 5 mL of n-butanol was refluxed for 4 hours. Upon cooling the product was filtered and washed with isopropyl alcohol and diethyl ether. The crude dye obtained was recrystallized from the DMF-methanol mixture to provide Yat 2148 in 69% yield. Abs=520 nm, Em=632 nm. The structure of Yat 2148 is given below:
##STR00151##
Example 8. Synthesis of Yat 2149
(a) Preparation of 1,1'-(2,2'-oxybis(ethane-2,1-diyl))bis(4-methylpyridinium) chloride (Compound 6)
[0233] A mixture of 4-methylpyridine (1.02 g) and 0.72 g of 1-Chloro-2-(2-chloro-ethoxy)-ethane (0.72 g) was heated at 120-130.degree. C. for 3-4 hours. To the reaction mixture 5 mL of isopropyl alcohol was added and the mixture was refluxed for an hour. Upon cooling the product was filtered and washed with diethyl ether to provide Compound 6 in 81% yield. This product was used without any further purification. The structure of Compound 6 is given below:
##STR00152##
(b) Preparation of Yat 2149
[0234] This procedure was carried out as described previously in step (b) of Example 3 with Compound 6 (0.33 g), piperidine (.about.6 drops), p-diethylamino benzaldehyde (0.36 g) and n-butanol (5 mL). After cooling the dye was precipitated with isopropyl alcohol or diethyl ether. In order to obtain the iodide salt, a saturated aqueous solution of KI (0.34 g) was added to the dye solution in methanol. After cooling, the product was filtered, washed with isopropyl alcohol, diethyl ether and dried under vacuum to provide Yat 2149 in 65% yield. Abs=502 nm, Em=614 nm. The structure of Yat 2149 is given below:
##STR00153##
Example 9. Synthesis of Yat 2150
[0235] This procedure was carried out as described previously in step (b) of Example 2 with Compound 3 (0.61 g), piperidine (.about.5 drops), p-diethylamino benzaldehyde (0.36 g) and n-butanol (5 mL). Purification was carried out by recrystallization from DMF-methanol mixture to provide Yat 2150 in 71% yield. Abs=485 nm, Em=612 nm. The structure of Yat 2150 is given below:
##STR00154##
Example 10 Monitoring Protein Stability in Two Different Buffer Formulations
[0236] Goat anti-mouse IgG from Vector Labs (1.5 mg) was resuspended in 150 .mu.l deionized water (dH.sub.2O). Phosphate was removed from the IgG using an Ambion NucAway spin column, following the manufacturer's instructions, briefly the column was resuspended in 700 .mu.l dH.sub.2O and allowed to hydrate for 60 minutes. Excess liquid was removed by centrifugation at 700.times.g for 2 minutes. The column was placed in a fresh collection tube and the sample was carefully loaded on the center of the column. The IgG was eluted by centrifugation at 700.times.g for 2 minutes. The purified IgG was diluted 10 fold in either 100 mM HCl or 12 mM phosphate pH 7.4, 150 mM sodium chloride. The samples were incubated for 18 hours at 37.degree. C. The solutions were stained with a final concentration of 100 mM MES, pH 6, 0.25 mg/ml IgG, 3 .mu.M S-25 and 3 .mu.M Tol3 (1:1 ratio) for at least 15 minutes. The stained protein was spotted on the surface of a glass microscope slide and overlaid with a cover slip, sealed with nail polish and observed using a BX51 microscope (Olympus, Tokyo, Japan). Images were acquired with a 40.times. objective lens (Olympus). Fluorescent images were acquired using a Texas Red filter set (Chroma Technoloogy Corp., Rockingham, Vt.). FIG. 1 shows that fibrils were formed in HCl solution, but not in the neutral phosphate buffer. The fibrils formed exhibited fluorescence that was bright and specific to the fibers using the S-25 and Tol3 dye mixture. There was little or no fluorescence when fibrils had not formed.
Example 11. Binding Curve of Different Fluorescent Probes to 20 .mu.M of Aggregated Lysozyme Protein
[0237] Lysozyme aggregates were formed by dissolving Lysozyme in 10 mM HCl to make a 1 mM Lysozyme solution (14.8 mg/ml). The Lysozyme solution was heated to 65.degree. C. with shaking at 750 rpm in an Eppendorf thermomixer for 90 hours. The lysozyme was diluted to 20 .mu.M in a 50 mM potassium phosphate solution containing different concentrations of a mixture of the dyes S-25 and Tol3. The aggregate was incubated for 15 minutes prior to measuring the fluorescence using a BioTek SynergyMx plate scanner, with excitation set at 515 nm and emission set to 603 nm, both with a 9 nm slit-width. Readings were taken in at least triplicate in a Greiner .mu.Clear black, clear bottom 96-well microplate. As can be seen in FIG. 2, there is little or no signal generated with up to 1 nM of each of the dyes. Above 1 nM, the signal increases until about 1 .mu.M each of the dyes, at which point no further signal increase is observed. This indicates that above 1 .mu.M S-25 and 1 .mu.M Tol3, the fluorescence of 20 .mu.M aggregated Lysozyme is dependent on aggregate concentration, and not dye concentration.
Example 12. pH Sensitivity of Fluorescence Response to Aggregated Lysozyme
[0238] Chicken egg white lysozyme (Sigma-Aldrich) was dissolved at 1 mM in 10 mM HCl. This monomer solution was stored at 4.degree. C. Lysozyme aggregate was formed by shaking the protein solution at 750 rpm in a Thermomixer (Eppendorf) at 65.degree. C. for 90 hours. The aggregation process was monitored by thioflavin T binding and after saturation of the fluorescence signal (for lysozyme after 90 hrs), the aggregate solution was also stored at 4.degree. C.
[0239] FIG. 3A shows the effect of pH on the aggregation-specific fluorescence of the dyes S-25 and Tol3, as determined by incubation of 4 .mu.M aggregated lysozyme, 4 .mu.M monomer lysozyme (or 8 .mu.M lysozyme monomer alone) with 0.5 .mu.M S-25 and 0.5 .mu.M Tol3 in buffers with a pH ranging from 3.about.10. The buffers used were: 8 mM glycine-HCl, pH 3; 8 mM sodium acetate, pH 4.4; 8 mM ammonium acetate, pH 6.0; 8 mM Tris-HCl, pH 7.4; 40 mM Tris-HCl, pH 7.8; 8 mM Tris-HCl, pH 8.5; and 8 mM sodium carbonate, pH 10. The dye-protein mixture was incubated at room temperature (22.degree. C.) for at least 15 minutes. Four replicates for either the 50% aggregate or monomer at each pH were prepared and the plate was scanned on a FLUOstar OPTIMA plate reader using an excitation wavelength of 550 nm and an emission of 610 nm.
[0240] FIG. 3B shows the effect of pH on the linearity of aggregation specific fluorescence, as determined using 1.25 .mu.M S-25 and 1.25 .mu.M Tol3 in 50 mM of the following buffers: succinic acid-HCl, pH 5.0; histidine-HCl, pH 7.0; and tris-HCl, pH 8.0. The total concentration of lysozyme was kept constant at 20 but the percent of the total that was aggregated as opposed to monomeric was varied from 0% to 100% aggregate. At least three replicates of each sample was prepared, incubated at 22.degree. in the dark for 15 minutes, then scanned on a FLUOstar OPTIMA plate reader using an excitation wavelength of 550 nm and an emission of 610 nm.
Example 13. Linear Dynamic Range of Lysozyme Aggregate Detection Using a Two Dye Combination ST (525& Tol3) Compared with Thioflavin T
[0241] Hen egg white lysozyme was solubilized in 10 mM HCl and heated to 65.degree. C. for 90 hours to form aggregates. The signal from the aggregate was determined after mixing aggregated lysozyme with monomeric lysozyme at different ratios such that the total lysozyme concentration remained at 20 .mu.M protein. The readings were taken in 50 mM potassium phosphate, pH 7, containing either ST (3 .mu.M S-25 and 3 .mu.M Tol3) or 5 .mu.M thioflavin T. Protein was incubated with dye for 15 minutes prior to determining the fluorescence using a BioTek Synergy Mx plate scanner, with excitation setting at 515 nm and emission setting at 603 nm, both with a 9 nm slit-width for S-25 and Tol3, and Thioflavin T was detected with excitation setting at 435 nm and emission setting at 495 nm, both with a 9 nm slit-width. Readings were taken in at least triplicate in a Greiner .mu.Clear black, clear bottom 96-well microplate. As seen in FIG. 4, the concentration curve is more linear with S25/Tol3 as compared to Thioflavin T.
Example 14. Effective Linear Dynamic Range of Antibody Aggregate Detection Using a Two Dye Combination ST (525& Tol3), Compared with Thioflavin T
[0242] Purified Rabbit anti-Goat IgG (4.26 mg/ml) was incubated in HCl, pH 2.7 at 80.degree. for 90 minutes to form aggregates. The signal from the aggregate was determined after mixing aggregate with monomer at different ratios such that the total IgG concentration remained at 240 .mu.g/ml protein. The readings were taken in 50 mM potassium phosphate, pH 7, containing either ST (3 .mu.M S-25 and 3 .mu.M Tol3) or 5 .mu.M thioflavin T. Protein was incubated with dye for 15 minutes prior to determining the fluorescence using a BioTek SynergyMx plate scanner, with excitation setting at 515 nm and emission setting at 603 nm, both with a 9 nm slit-width for S-25 and Tol3, and thioflavin T was detected with excitation setting at 435 nm and with emission setting at 495 nm, both with a 9 nm slit-width. Readings were taken in at least triplicate in a Greiner .mu.Clear black, clear bottom 96-well microplate. As can be seen in FIG. 5, the signal from ST is more than 10 times higher than the signal from thioflavin T under these conditions. Also the concentration curve is more linear with S-25/Tol3 as compared to thioflavin T.
Example 15. Protein Aggregate Detection as a Function of Protein Species
[0243] The linearity of aggregation induced fluorescence of S-25, Tol3 and Thioflavin T (Thio-T) for four different proteins was determined. The proteins were hen egg white lysozyme (results shown in FIG. 6A), rabbit anti-goat IgG (FIG. 6B), bovine insulin (FIG. 6C) and .beta.-lactoglobulin (FIG. 6D)).
[0244] Chicken egg white lysozyme aggregate solution and monomer solution as well as their mixtures were prepared as described in Example 12. The protein concentration was maintained at 20 .mu.M, and the dye concentration was 2.5 .mu.M in 50 mM Tris-HCl, pH 8. The ratio of aggregated protein to native protein was varied from 0 to 100% aggregate. Each sample was analyzed in at least 3 replicates. The mixtures were incubated in the dark at 22.degree. C. for 15 minutes, then the fluorescence intensity was determined with a FLUOstar OPTIMA plate reader (BMG LABTECH) with excitation setting at 550 nm and emission setting of 610 nm; while the fluorescence intensity for Thioflavin-T was determined using a SpectraMAX GeminiXS (Molecular Devices, with Softmax Pro 7.0) using an excitation wavelength of 435 nm and emission wavelength of 495 nm.
[0245] Rabbit-anti-goat IgG (H&L, Pel-Freez.RTM., formulated in the same manner as goat-anti-mouse IgG, described in Example 2) was diluted to 29.4 .mu.M with double deionized water adjusted to pH 2.7 using HCl. Then IgG aggregate was prepared by shaking the protein solutions at 750 rpm in a Thermomixer (Eppendorf) at 80.degree. C. for 2 hours. Using a final protein concentration of 3 .mu.M, the linearity of aggregation induced fluorescence was determined as described above for lysozyme.
[0246] Insulin aggregate was prepared by dissolving bovine pancreas insulin (Sigma-Aldrich) at 170 .mu.M in 100 mM HCl, which was subsequently transferred to a Thermomixer (Eppendorf), set at 750 rpm continuous shaking at 65.degree. C. for 150 min. Using a final protein concentration of 20 .mu.M, the linearity of aggregation induced fluorescence was determined as described above for lysozyme.
[0247] .beta.-Lactoglobulin (BLG, Sigma-Aldrich) was dissolved at 1 mM in double deionized water. The aggregate was prepared by shaking the protein solutions at 750 rpm in a Thermomixer (Eppendorf) at 80.degree. C., which was stopped after 24 hours. Using a final protein concentration of 50 .mu.M, the linearity of aggregation induced fluorescence was determined as described above for lysozyme.
Example 16. Kinetics of Lysozyme Aggregation
[0248] A 1 mM solution of hen egg white lysozyme in 10 mM HCl was incubated at 65.degree. C. in an Eppendorf thermomixer shaking at 750 rpm. At the indicated times, aliquots of the lysozyme were removed, diluted to 30 .mu.M in 100 mM Tris-HCl, pH 8.0, and incubated with 5 .mu.M of the indicated dye. After 15 minutes incubation, fluorescence intensity was determined with a FLUOstar OPTIMA plate reader (BMG LABTECH) at excitation wavelength of 550 nm and emission wavelength of 610 nm; while the fluorescence intensity for thioflavin-T was determined using a SpectraMAX GeminiXS (Molecular Devices, with Softmax Pro 7.0) using an excitation wavelength of 435 nm and emission wavelength of 495 nm. Every sample was evaluated in 4 replicates. As can be seen in FIG. 7, Tol3, S-25 and Thioflavin T all detect similar kinetics for protein aggregate formation.
Example 17. Protein Aggregation as a Function of Temperature
[0249] A 0.9 mg/ml solution of goat-anti-mouse IgG (Pel Freeze) was prepared in 73 mM sodium acetate, pH 4.5. This solution was incubated at 21.degree. C. or 50.degree. C. for various times. After incubation, the solution was diluted further to create a solution that was 50 mM histidine, pH 7, 0.45 mg/ml IgG, 2.5 .mu.M S-25 and 2.5 .mu.M Tol3. After 15 minutes further incubation, the fluorescence intensity was determined with a FLUOstar OPTIMA plate reader (BMG LABTECH) at an excitation wavelength of 550 nm and emission wavelength of 610 nm. As seen in FIG. 8, aggregation is much more rapid at 50.degree. C. than at 21.degree. C.
Example 18. Protein Aggregation as a Function of pH
[0250] Goat-anti-mouse IgG was diluted to 40 .mu.M at either pH 7.6 in sodium phosphate buffer, or adjusted to pH 2.46 using HCl. Both solutions were then kept at 21.degree. C. After various times, aliquots were removed and diluted to a final concentration of 2 .mu.M in 100 mM histidine buffer, pH 7 with 2.5 .mu.M S-25 and 2.5 .mu.M Tol3. After 15 minutes further incubation at 21.degree. C., the fluorescence intensity was recorded. As seen in FIG. 9, aggregation is observed to be much more rapid under acidic pH conditions.
Example 19. Illustration of High-Throughput Protein Formulation Optimization
[0251] (A). Goat anti-mouse IgG was diluted in sodium acetate, pH 4.5, then mixed with the excipients shown in FIG. 10A giving a final concentration of 400 mM sodium acetate, 18 .mu.M IgG and the excipient concentration shown in FIG. 10A. This mixture was heated to 50.degree. C. for 6 hours. After this incubation, the protein solution was diluted two-fold to give a final concentration of 50 mM histidine buffer, originally pH 7, 2.5 .mu.M S-25, 2.5 .mu.M Tol3 and 9 .mu.M IgG. After 30 minutes of incubation on the shaker, the fluorescence intensity was recorded on the plate reader (FLUOstar Optima). The fluorescence intensity from each individual excipient was then compared with that without any excipient (value set as 1.0) as shown on the top of the corresponding excipient bar in FIG. 10A.
[0252] (B). In the control plate, the IgG was added to the plate at the same volume and concentration as in A. above, in 400 mM sodium acetate. This mixture was heated to 50.degree. C. for 6 hours, as described above. After 6 hours, the excipient was added followed by S-25 and Tol3 to give all the final concentrations as in A. above. Similar to the sample plate, the fluorescence intensity from individual excipients was also compared with that from water without any excipient (values set as 1.0) to obtain the relative fluorescent intensity as shown on the top of the corresponding excipient bar in FIG. 10B.
[0253] (C). A ratio between the fluorescent intensity of the protein aggregated with the excipient versus the intensity derived from the protein aggregated without excipient is a good measure of the effect of the given excipient on aggregation. FIG. 10C shows the ratio of fluorescence intensity in the sample plate (A. above) divided by the fluorescence intensity of the control plate (B. above). Those compounds with a value of 1 (dotted line) do not significantly affect aggregation of IgG. Those compounds substantially higher than 1, such as 0.2% Triton X-100 induce aggregation of IgG. Those compounds with a value substantially lower than 1, such as 100 mM trehalose, inhibit or slow down aggregation of IgG.
Example 20. Inhibition of Lysozyme Aggregation by Chitotriose
[0254] Hen egg white lysozyme (300 .mu.M) was incubated with or without N,N',N''-triacetyl-chitotriose ("Chitotriose", 510 .mu.M) in 10 mM potassium phosphate, pH 7.3 for 16 hours. Aggregation was induced by 3.5 fold dilution into 50 mM potassium phosphate, pH 12.2. Aggregation was followed by removing an aliquot of the protein and diluting such that the final composition was 20 .mu.M protein, 50 mM potassium phosphate, pH 7, 3 .mu.M S-25 and 3 .mu.M Tol3. Protein was incubated with dye for 15 minutes prior to determining the fluorescence using a BioTek Synergy Mx plate scanner, with excitation setting at 515 nm and emission setting at 603 nm, both with a 9 nm slit-width. The zero time point was taken before dilution to pH 12.2. Readings were taken in at least triplicate in a Greiner .mu.Clear black, clear bottom 96-well microplate. Aggregation was followed for several weeks at room temperature (19.degree.-23.degree. C.). As seen in FIG. 11, S-25 and Tol3 easily demonstrate that Chitotriose inhibits lysozyme aggregation, as previously demonstrated by Kumar et al. (2009).
Example 21. Thermal Shift Assays of BLG Aggregation
[0255] A solution containing 4 or 16 mg/mL of .beta.-lactoglobulin (BLG) and 2.times.SYPRO.RTM. Orange dye (Molecular Probes, supplied as 5000.times. with unknown concentration) or 4 .mu.M TOL3 or 4 .mu.M S25 was prepared using 1.times.PBS, pH 7.4 as the dilution buffer. This solution was then loaded into LightCycler.RTM. capillaries (20 .mu.L, Roche Diagnostics GmbH). These capillaries were then mounted on the sample holder of a LightCycler.RTM. 480 Real-Time PCR System (Roche), programmed to heat from 28.degree. C. to 90.degree. C. at 3.degree. C./min, followed by cooling down to 28.degree. C. at the same rate. The thermal shift curves were achieved by plotting fluorescence intensity vs. temperature. After the heating cycle, protein aggregates were visually apparent. However, SYPRO.RTM. Orange dye, known to detect protein, failed to show a melting peak, probably because of a low binding affinity to the aggregated BLG; but both TOL3 and S25 were able to detect BLG thermal shift peaks due to the aggregation, as shown in FIG. 12. The temperature of aggregation detected by TOL3 or S25 both showed a protein concentration dependence, down-shifting from 81.about.83.degree. C. to 71.about.73.degree. C. when the BLG concentration was increased from 4 mg/mL to 16 mg/mL, a characteristic of protein aggregation, as opposed to protein unfolding. This demonstrates that both TOl3 and S25 are detecting aggregation thermal shift peaks of BLG, not transitions do to unfolding of the protein.
Example 22. Thermal Shift Assays of Carbonic Anhydrase II Aggregation
[0256] Carbonic anhydrase II (Sigma, 10 .mu.M) containing 5.times.SYPRO.RTM. Orange or 10 .mu.M TOL3 or S25 or Yat 2150 was prepared using either 50 mM sodium acetate, pH 4.5 or 25 mM PIPES, pH 7.0 buffer containing 100 mM NaCl and 0.5 mM EDTA. Sample preparation and the thermal shift assay were then performed using the same conditions as described in Example 21. As shown in FIG. 13, although SYPRO.RTM. orange and dyes of the invention all show thermal shift peaks, there is a .about.5.degree. C. up-shift for peaks from dyes of the invention, between pH 4.5 and pH 7.0. This also highlights the fundamentally different detection mechanism between SYPRO.RTM. Orange dye and the dyes described in this invention; the former detects protein unfolding, while the later detects protein aggregation.
Example 23. Comparison of Fluorescence Response Between Unfolded and Aggregated Form of IgG Using Dyes of the Present Invention
[0257] (A) Chemical shift assay based on internal tryptophan fluorescence: Rabbit-anti-goat IgG (Pel Freeze) in 1.times.PBS buffer of pH 7.4 was mixed with urea in 1.times.PBS to achieve a final IgG concentration of 0.25 mg/ml. After mixing on ice for 10 minutes, the fluorescence emission intensity at 330 nm was recorded by exciting at 280 nm using a MD-5020 fluorimeter (Phototechnology International). A chemical shift curve was plotted based on internal tryptophan fluorescence intensity at each given urea concentration. Results are shown in FIG. 14A. Urea denatures proteins but prevents them from aggregating.
[0258] (B) A solution containing aggregated IgG (formed as in Example 15) or monomeric IgG at 0.033 mg/mL, 4.55 M urea and 6.67 .mu.M Tol3 was prepared and transferred into a microplate. After incubating at 4.degree. C. degree for about 10 minutes, the fluorescence was recorded. Two control solutions without IgG but with the same concentration of TOl3 were included, one including 4.55M urea, another without urea. From the previous chemical shift curve generated (FIG. 14A), 4.55 M urea is known to unfold approximately 60% of the IgG. The results shown in FIG. 14B indicate that TOL3 is sensitive to IgG aggregates, which shows significant fluorescence enhancement relative to controls without IgG, but it is not sensitive to unfolded IgG monomer, which shows insignificant fluorescence enhancement relative to controls without IgG.
Example 24. PDI Isomerase Activity Assay by Monitoring Insulin Aggregation Kinetics
[0259] (A) Turbidity assay: Protein disulfide isomerase (PDI, Assay Designs) was diluted with 0.5M of sodium phosphate, pH 6.8. A mixture was made with insulin to give a final solution comprising 188 mM sodium phosphate, pH 6.8, 5 mM Tris-HCl, 2 mM EDTA, 1 mM DTT, 1 mg/mL insulin and PDI at the desired concentrations (0, 5, 10, 15, 20, 25 .mu.g/mL). The optical density (OD) at 630 nm was recorded immediately after the addition of DTT in a 96-well microplate reader at 2 minute-intervals, with every well containing 300 .mu.L solution. The OD from 0 .mu.g/mL of PDI at any time point was used as a background value and was subtracted from the OD value of samples with PDI at the same time point. Results are seen in FIG. 15A.
[0260] (B) Fluorometric assay: PDI and insulin solutions were prepared as in the turbidity assay described in (A) above. S25 and TOL3 were mixed with the insulin solution and placed into a black Greiner flat bottom 96-well plate. PDI solutions containing various amount of PDI were then added. Just prior to fluorescence recording, DTT was added. The final solution was 188 mM sodium phosphate pH 6.8, 5 mM Tris-HCl, 2 mM EDTA, 1 mM DTT, 0.225 mg/mL insulin and PDI at 0, 5, 10, and 20 .mu.g/ml. A FLUOstar Optima plate reader was used to record the fluorescence change after 5 seconds' shaking with excitation set at 550 nm and emission set at 610 nm. The fluorescence intensity from 0 .mu.g/mL of PDI solution at the corresponding time point was used as a background value and was subtracted from the corresponding reading in the presence of enzyme. Results are seen in FIGS. 15B and C. The turbidity assay and fluorometric assay, though of significantly different sensitivities, are orthogonal to each other, further supporting that dyes of the present invention monitor aggregation status and not unfolding status.
Example 25. Inhibition of .beta.-Lactoglobulin Aggregation by HSP 27
[0261] Aggregation of .beta.-lactoglobulin was monitored in the presence or absence of the chaperone HSP 27. Aggregation of 8 mg/ml .beta.-lactoglobulin was monitored using 1.25 .mu.M Tol3 and 1.25 .mu.M S25 in PBS, pH 7.4 with 2.5 mM EDTA and 0.05% sodium azide. When the chaperone HSP 27 was added it was added to a final concentration of 0.4 mg/ml. HSP 27 was also run in the absence of .beta.-lactoglobulin as a control. Aggregation was initiated by heating the protein solution to 68.degree. C. in a 96 well half-volume clear plate (Biomol International, Inc). The fluorescence intensity was then recorded every 2 minutes, with shaking between reads. The excitation wavelength was set to 550 nm and the emission was set to 610 nm on a BMG Fluorstar plate reader. The fluorescence intensity of the starting point was subtracted from the remaining points. The results (FIG. 16) indicate that Hsp 27 can significantly prevent the aggregation of BLG at a mass ratio as low as 1:20. Since Hsp 27 is binding with unfolded BLG intermediate, thus preventing protein aggregation, the dyes are detecting protein aggregation, as opposed to unfolding.
[0262] Other chaperone activity assays can be configured using .beta.-lactoglobulin or other substrates, such as citrate synthase (CS). Table 6 shows suggestions for chaperone-to-CS ratios that should find application for the disclosed assay methods.
TABLE-US-00006 TABLE 6 Chaperone:CS ratios. Chaperone system Members ADI catalog #s Chaperone:CS DnaK/DnaJ/GrpE DnaK SPP-630 1:1 or less DnaJ SPP-640 GrpE SPP-650 Hsp70/Hsp40 Hsp70 NSP-555, ESP-555, 1:1 or less SPP-758 Hdj1 SPP-400 Hdj2 SPP-405 Mortalin SPP-828 Hsc70 SPP-751 Hsp70-A1 SPP-502, ESP-502 Hsp90 Hsp90 alpha SPP-776 Depends on Hsp90 beta SPP-777 cochaperones Chaperonins Hsp60/Cpn10 NSP-540, ESP-540 1:1 or less (human) Cpn10 SPP-110 Chaperonins GroEL SPP-610 1:1 or less (bacterial) GroES SPP-620 Small heat shock Hsp25 SPP-510 20:01 .sup. proteins Hsp27 SPP-715, SPP-716 Crystallins SPP-225, SPP-226, SPP-235, SPP-236 ER chaperones Grp78 SPP-765 5:1 .sup. PDI SPP-891 10:1 .sup. Erp72 H00009601-Q01 20:1 .sup. (abnova) Grp94 SPP-766 Depends on (ER Hsp90) cochaperones Nascent chain NAC none 20:1 .sup. chaperones Trigger Factor none 20:1 .sup.
[0263] Chaperone: CS ratios are based upon the known biology of the individual systems. Active folders are likely to show significant signal at less than 1:1 molar ratio to substrate, as each chaperone complement will be able to inhibit aggregation while it actively folds. Aggregate inhibitors like the small heat shocks and trigger factor require substantially more, as they need to saturate the solution to prevent aggregation. Pairs of holders and folders (e.g., crystalline with low Hsp70 complex) may provide synergistic effects.
Example 26. Monitoring Protein Stability in an Agitated Solution
[0264] One method of creating aggregated proteins is by agitation of the protein solution. Goat-anti-mouse IgG (12.8 mg/mL, Pel-freeze Biologicals) was supplied in 10 mM sodium phosphate, 150 mM NaCl, 0.05% sodium azide, pH 7.2, filtered through 0.2 .mu.m filter. The stirring experiment was performed by stirring 200 .mu.L of IgG solution as supplied at 22.degree. C. in a 4 mL amber glass vial with flat bottom at 990 rpm using Variomag.RTM. Poly electronic stirrers. The control (without stirring) was also kept at 22.degree. C. The stirring bar was 1.times.0.4.times.0.2 cm.sup.3.
[0265] A BioTek plate reader with a filter set as 550 (excitation)/603 nm (emission) and 9 nm filter band on both excitation and emission was used to scan from the bottom of the plate. 5 .mu.L of the IgG solution at various time points (stirred or non-stirred) was added into 95 .mu.L of 2.5 .mu.M TOL3, 2.5 .mu.M of S25 and 50 mM potassium phosphate, pH 7.0 and incubated for 20 minutes. Every time point was replicated twice. The fluorescence of free dye was subtracted from that of the IgG/dye mixing solution for both the stirred sample and non-stirred control. The results (FIG. 17) indicate that the TOL3/S25 dye mix can detect agitation induced aggregation.
Example 27. Thermal Shift Assay to Find Thermally Stabilizing Buffers for DNA Polymerase I Klenow Fragment
[0266] In molecular biology, enzymes are often required that function at elevated temperatures. Enzymes produced by mesophilic organisms usually denature at elevated temperatures, followed by aggregate formation. A rapid fluorescence-based assay was developed for assessing a range of parameters impacting the thermal stability of an enzyme. Overall, protein stability was monitored by a fluorogenic dye that selectively detects aggregated protein. Stability can be measured in the presence of different buffers, cryoprotectants and excipients. By systematically raising the temperature of the protein in solution, the temperature at which the protein aggregates (T.sub.agg) can be determined. Using this method with Klenow DNA polymerase, it was determined that trehalose significantly increases T.sub.agg. The DNA polymerase activity of the enzyme is significantly enhanced at 50.degree. C. in the presence of trehalose under the same conditions. Low amounts of the detergents Tween20.RTM. and Triton X-100.RTM. (0.05%) decreased T.sub.agg and also compromised enzyme activity, especially at elevated temperatures. The assay facilitates screening for buffers and additives that structurally stabilize a protein of interest at elevated temperatures.
[0267] Fifty units of DNA polymerase I Klenow fragment (New England Biolabs, Ipswich, Mass.) was incubated in several different buffers and excipients in the presence of 2.5 .mu.M YAT2150 dye. The temperature was slowly raised and the fluorescence was determined using a Qiagen (Valencia, Calif.) Rotorgene real-time thermocycler with an excitation filter at 530 nm and an emission filter of 610 nm. Table 7 shows the detected aggregation temperatures in Buffer 1 (10 mM bis-Tris propane, 10 mM MgCl.sub.2, 1 mM dithiothreitol, pH 7), Buffer 2 (50 mM NaCl, 10 mM Tris-HCl, 10 mM MgCl.sub.2, 1 mM dithiothreitol, pH 7.9), Buffer 3 (100 mM NaCl, 50 mM Tris-HCl, 10 mM MgCl.sub.2, 1 mM dithiothreitol, pH 7.9), Buffer 4 (50 mM potassium acetate, 20 mM Tris-acetate, 10 mM magnesium acetate, 1 mM dithiothreitol, pH 7.9), Buffer 2 with 0.5 M trehalose, Buffer 2 with 0.05% Tween20.RTM. and Buffer 2 with 0.05% Triton X-100.RTM..
TABLE-US-00007 TABLE 7 Effect of various buffers on aggregation temperature of DNA polymerase I Klenow fragment. Buffer T.sub.agg Buffer #1 59.1.degree. +/-0.3.degree. Buffer #2 59.7.degree. +/-0.2.degree. Buffer #3 60.2.degree. +/-0.3.degree. Buffer #4 59.8.degree. +/-0.4.degree. Buffer #2 Trehalose 62.6.degree. +/-0.3.degree. Buffer #2 Tween 20 58.4.degree. +/-0.3.degree. Buffer #2 Triton X-100 57.8.degree. +/-0.2.degree.
[0268] This data demonstrates that trehalose significantly raises the aggregation temperature in Buffer 2. To show if this extends to enzyme activity, the DNA polymerase was incubated in Buffer 2 with 400 .mu.M dATP, dCTP, dGTP and fluorescein-12-dUTP at 37.degree. C., 42.degree. C. and 50.degree. C. After 15 minutes at the given temperature, the DNA template and primer were added for an additional 15 minutes at the given temperature. The template used was 5-ACTTCTTACT TCTTACTTCT TACTTCTTAC TTCTTACTTC TTACTTCTTA CTTCTTACTT CTTCATTGGT CATCTCGATC CATGACCTCA GC-3' and the primer was 5'-TTGCTGAGGT CATGGATCGA GA-3'. The amount of oligo extended full length is shown in Table 8, as measured by relative fluorescence of the incorporated fluorescein.
TABLE-US-00008 TABLE 8 Oligonucleotide extension by DNA polymerase I Klenow fragment using various buffer additives during incubation at three temperatures. Additive in Buffer 2 37.degree. 42.degree. 50.degree. None 0.507 0.303 0.189 0.5M Trehalose 0.567 0.449 0.308 0.05% Tween20 0.317 0.154 0.180 0.05% Triton X-100 0.257 0.159 0.170
This data shows a correlation with the increased aggregation temperature in the presence of 0.5 M trehalose and the enzyme activity at elevated temperatures.
Example 28. YAT2150 Dye Detecting Aggregated Proteins within Fixed and Permeabilized Cells
[0269] In mammalian cells, aggregated proteins may be concentrated by microtubule-dependent retrograde transport to perinuclear sites of aggregate deposition, referred to as aggresomes. Aggresomes are inclusion bodies that form when the ubiquitin-proteasome machinery is overwhelmed with aggregation-prone proteins. Typically, an aggresome forms in response to some cellular stress, such as hyperthermia, viral infection, or exposure to reactive oxygen species. Aggresomes appear to provide a cytoprotective function by sequestering the toxic, aggregated proteins and may also facilitate their ultimate elimination from cells by autophagy. Certain cellular inclusion bodies associated with human disease are thought to arise from an aggresomal response, including Lewy bodies associated with neurons in Parkinson's disease, Mallory bodies associated with liver cells in alcoholic liver disease, and hyaline inclusion bodies associated with astrocytes in amyotrophic lateral sclerosis.
[0270] The ability of YAT2150 to detect aggregated proteins within fixed and permeabilized cells was evaluated. Human cervical adenocarcinoma epithelial cell line HeLa was obtained from American Type Culture Collection (ATCC, Manassas, Va.). HeLa cells were routinely cultured in Eagle's Minimum Essential Medium (ATCC) with low glucose, supplemented with 10% fetal bovine serum (FBS) (ATCC) and 100 U/ml penicillin with 100 .mu.g/ml streptomycin (Sigma-Aldrich). Cells were maintained in a saturated, humidified atmosphere at 37.degree. C., 5% CO.sub.2 and 95% air. HeLa cells were grown on glass slides or polystyrene tissue culture dishes until .about.80% confluent. The cells were treated with various modulators or vehicle at various concentrations and time intervals, as detailed in Table 9. Proteasome inhibitors MG-132 (Enzo Life Sciences Inc.), lactacystin (Enzo Life Sciences Inc.), bortezomib (Velcade.RTM.) (Selleck Chemicals LLC, Houston, Tex.) and epoxomicin (Enzo Life Sciences Inc.) were employed in the studies. The histone deacetylase 6 inhibitor N-hydroxy-7-[5-(4-tertbutoxycarbonylaminophenyl)-3-isoxazolecarboxamido] heptamide (BML-281) was also obtained from Enzo Life Sciences Inc. Negative control cells were treated with a vehicle (DMSO, media or other solvent used to reconstitute or dilute the inducer or inhibitor) for an equal length of time under similar conditions. The cells were subsequently washed with PBS, and fixed in 4% formaldehyde in PBS for 30 min at room temperature, then permeabilized with 0.5% Triton X-100, 3 mM EDTA in PBS on ice, for 30 minutes. The cells were washed with PBS, and then 500 nM of YAT2150 dye was added. The samples were incubated for 30 minutes at room temperature, protected from light. The cells were washed with PBS, covered with glass coverslips and observed using a fluorescence microscope (Carl Zeiss MicroImaging GmbH, Jena, Germany) equipped with a Texas Red filter set. Images were acquired with a 63.times. objective lens (Carl Zeiss, Inc).
TABLE-US-00009 .mu.M Induction Cell Aggresome Treatment Target Effect used Time (hrs) Line Formation Starvation Inhibits mammalian Activates N/A 1~4 HeLa, No target of rapamycin autophagy Jurkat (mTOR) Rapamycin Inhibits mammalian Activates 0.2 6~18 HeLa, No target of rapamycin autophagy Jurkat (mTOR) PP242 ATP-competitive Activates 1 18 Hela No inhibitor of mTOR autophagy Lithium Inhibits IMPase and Activates 10,000 18 HeLa, No reduce inositol and autophagy Jurkat IP.sub.3 levels; mTOR- independent Trehalose Unknown, mTOR- Activates 50,000 6 HeLa, No independent autophagy Jurkat Bafilomycin A1 Inhibits Vacuolar- Inhibits 6~9 18 HeLa, Yes ATPase autophagy .times.10.sup.-3 Jurkat Chloroquine Alkalinizes Inhibits 10~50 18 HeLa, Yes Lysosomal pH autophagy Jurkat Tamoxifen Increases the Activates 4~10 6~18 HeLa, Yes intracellular level of autophagy Jurkat ceramide and abolishes the inhibitory effect of PI3K Verapamil Ca.sup.2+ channel blocker; Activates 40-100 18 HeLa, Yes reducesintracytosolic autophagy Jurkat Ca.sup.2+ levels; mTOR- independent Hydroxy- Alkalinizes Inhibits 10 18 HeLa, Yes chloroquine Lysosomal pH autophagy Jurkat Loperamide Ca.sup.2+ channel Activates 5 18 HeLa No blocker;reduces intra- autophagy cytosolic Ca.sup.2+ levels; mTOR-independent Clonidine Imidazoline-1 Activates 100 18 HeLa No receptor agonist; autophagy reduces cAMP levels; mTOR-independent MG-132 Selective proteasome Induce 2~5 18 HeLa, Yes inhibitor aggresome Jurkat Epoxomicin Selective proteasome Induce 0.5 18 HeLa Yes inhibitor aggresome Lactacystin Selective proteasome Induce 4 Yes inhibitor aggresome Velcade .RTM. Selective proteasome Induce 0.5 18 HeLa Yes inhibitor aggresome amyloid beta Induce oxidative Induce 25 18 SK-N- Yes peptide 1-42 stress aggresome SH Norclomipramine Alkalinizes Inhibits 5~20 18 HeLa Yes Lysosomal pH autophagy
[0271] MG-132, a relatively nonspecific proteasome inhibitor, has also been shown to perturb protein homeostasis, inducing both the unfolded protein response (UPR) and the heat shock response (HSR) (Mu et al., 2008; Murakawa et al., 2007). MG-132 is known to accelerate the formation of perinuclear aggresomes as well as inclusion bodies within cells (Beaudoin S et al., 2008). After treating cells with MG-132, YAT2150 dye was found to readily highlight aggregated protein cargo accumulating within vacuolar cytoplasmic structures, as observed by fluorescence microscopy (FIG. 18). Examination of the distribution of the fluorescent dye within cells treated with MG-132, revealed a punctate pattern of cytoplasmic staining, as well as staining of certain inclusion bodies within or immediately adjacent to the nucleus itself. Multiple cytoplasmic inclusion bodies were readily discerned using the fluorescent dye. It should be noted that true aggresomes are characterized by a single large protein aggregate in cells that co-localizes with the centrosome in a microtubule-dependent fashion. The formation of the multiplicity of inclusion bodies observed upon MG-132 treatment was not sensitive to the histone deacetylase 6 inhibitor, N-hydroxy-7-[5-(4-tertbutoxycarbonylaminophenyl)-3-isoxazolecarboxamido]h- eptamide (BML-281) (Kawaguchi et al., 2003) or nocodazole (data not shown). This suggests that the generated inclusion bodies do not meet the strictest definition for aggresomes. The described probe appears to detect aggregated protein cargo within a variety of inclusion bodies, regardless of whether they are co-localized with the centrosome or formed in a microtubule-dependent manner.
[0272] The ability to detect aggresomes and related inclusion bodies was further demonstrated using various potent, cell permeable, and selective proteasome inhibitors: lactacystin, epoxomicin and bortezomib (Velcade.RTM.), as shown in FIG. 19. Previous studies have shown that bortezomib-mediated proteasomal inhibition results in the accumulation of large quantities of ubiquitin-conjugated proteins organized into perinuclear structures termed "aggresomes" (Nawrocki et al., 2006). All of the tested proteasome inhibitors induced the accumulation of cytoplasmic inclusion bodies within the cells, as demonstrated with the YAT2150 dye. Efforts to stain inclusion bodies in living cells were not met with success, however. Instead of discrete punctuate staining of aggregated cargo, a weak, diffuse cytoplasmic staining was observed that differed little between control cells and cells treated with a proteasome inhibitor. This could possibly be due to poor access of the dye to the contents of membrane-bound vacuolar structures within the cells.
Example 29. Co-Localization of Aggregated Protein with Ubiquitinylation and Various Pathway Proteins Implicated in Autophagy
[0273] Antibodies were obtained from the following commercial sources: fluorescein-labeled p62 and LC3 reactive rabbit polyclonal antibodies and ubiquitin-reactive mouse monoclonal antibody (clone EX-9) were obtained from Enzo Life Sciences, Ltd. (Exeter, UK). These labeled conjugates were produced by direct labeling of antibodies raised to p62-derived, LC3-derived, and ubiquitin-derived peptides, respectively. A mouse monoclonal antibody reactive with human tau (clone tau-13) (Covance Inc, Emeryville, Calif.) is able to stain brain tissue early in Alzheimer's disease. It was used in conjunction with Alexa Fluor.RTM. 488 dye-labeled goat anti-mouse secondary antibody from Life Technologies (Carlsbad, Calif.). Alexa Fluor.RTM. 488 dye-labeled beta amyloid reactive mouse monoclonal antibody (clone 6E10), which is specifically reactive to amino acid residues 1-16 of the human .beta.-amyloid peptide, was obtained from Covance Inc.
[0274] For antibody co-localization studies: cells were treated overnight with 5 .mu.M MG-132, then fixed and permeabilized using the protocol in Example 28. The cells were then incubated in PBS containing 3% bovine serum albumin (blocking buffer). Fluorescein-labeled p62, LC3 and ubiquitin (clone EX-9) reactive antibodies were diluted to a concentration of 1 .mu.g/mL in blocking buffer and incubated for 1 h at room temperature. Cells were then washed in PBS containing 0.1% Tween-20 for 15 min. Next, the cells were stained with YAT2150 dye for 30 minutes at room temperature and washed with PBS, covered with glass cover slip, sealed with nail polish, and observed by fluorescence microscopy using a Texas Red filter set for the YAT2150 dye, and an FITC filter set for fluorescein-labeled antibodies, respectively. All images were acquired with a 63.times. objective lens (Carl Zeiss, Inc).
[0275] Co-localization of fluorescently-labeled ubiquitin antibody conjugate with YAT2150 dye is shown in FIG. 20A, highlighting interactions between aggregated protein cargo and ubiquitinylation status. The ubiquitin signal is observed to be co-localized exclusively with the aggregated protein cargo, but it should be remembered that cells were fixed and permeabilized, which likely removed any free ubiquitin and ubiquitinylated substrates present in the cytosol. FIG. 20B demonstrates that a fluorescein-conjugated antibody directed towards p62 (a ubiquitin-binding scaffold protein that co-localizes with ubiquitinylated protein aggregates) also co-localizes with the YAT2150 dye within cells treated with MG-132. Furthermore, co-localization between fluorescein-labeled antibodies reactive with LC3 (a ubiquitin-like autophagy cascade protein residing in the phagophore membrane) and aggregated protein cargo was demonstrated, as shown in FIG. 20C. The described co-localization studies demonstrate the capability of the YAT2150 dye to be used to analyze the interactions between aggregated protein cargo, protein post-translational modifications, and various autophagy pathway proteins. The long wavelength red emission of the fluorescent probe is especially suitable for studies using green fluorescent dye conjugates, such as fluorescein, Alexa Fluor.RTM. 488, Oregon Green.RTM. 488, BODIPY.RTM.-FL, HiLyte Fluor.TM. 488 and DyLight.RTM. 488.
Example 30. Cell Culture-Based Model Mimicking Elements of Alzheimer's Disease Pathology
[0276] The human SK-N-SH neuroblastoma cell line was obtained from American Type Culture Collection (ATCC, Manassas, Va.). SK-N-SH cells were routinely cultured in Eagle's Minimum Essential Medium (ATCC) with low glucose, supplemented with 10% fetal bovine serum (FBS) (ATCC) and 100 U/ml penicillin, 100 .mu.g/ml streptomycin (Sigma-Aldrich). Amyloid beta peptide 1-42 (21.sup.st Century Biochemicals, Marlboro, Mass.) was added to the culture medium and SKN-SH cells were incubated overnight to induce aggresome formation. SMER28 (Enzo Life Sciences Inc.), an inducer of autophagy, was employed to block this accumulation A cell culture-based assay mimicking the accumulation of .beta.-amyloid, as observed in Alzheimer's disease, was established.
[0277] FIG. 21B shows YAT2150 dye is able to detect amyloid fibrils within an SK-N-SH human neuroblastoma cell line induced to form inclusion bodies by overnight incubation with exogenously added amyloid beta 1-42 peptide. Furthermore, SMER28, a small molecule inducer of autophagy was evaluated with respect to its effect on .beta.-amyloid peptide accumulation within the cells. SMER28 has previously been shown to act via an mTOR-independent mechanism to increase autophagosome synthesis and enhance the clearance of model autophagy substrates, such as [A53T].alpha.-synuclein and mutant huntingtin fragments (Sarkar et al., 2007; Renna et al., 2010). It has also been demonstrated that SMER28 attenuates mutant huntingtin-fragment toxicity in Drosophila models, suggesting therapeutic potential. As shown in FIGS. 21C & D, SMER28 was able to substantially reduce accumulation of .beta.-amyloid peptide in SK-N-SH human neuroblastoma cells, suggesting this assay could potentially enable screening of aggregation inhibitors relevant to neurodegenerative disease, in an authentic cellular context.
Example 31. Detecting Protein Aggregates in Post-Mortem Brain Tissue Sections from Patients with Alzheimer's Disease
[0278] Post-mortem brain tissue (cerebellum) from patients with Alzheimer's disease and human adult normal brain tissue (cerebellum) were obtained from BioChain Institute, Inc. (Hayward, Calif.). All tissue samples were received from certified tissue vendors who guarantee that they were collected with informed consent from the donors and their relatives, all samples were excised by licensed physicians, all normal and diseased tissues were determined by the donor's clinical reports and all collections were made with the relevant requirements for ethics committee/IRB approvals. The frozen tissue sections were 5-10 .mu.m in thickness, mounted on positively charged glass slides, and fixed with cold acetone by the manufacturer. The embedded tissue sections were fixed in formalin immediately after excision, and embedded in paraffin. Tissue sections were .about.5 .mu.m in thickness, and mounted on positively charged glass slides by the manufacturer.
[0279] Paraffin-embedded tissue sections were deparaffinized prior to staining. Briefly, the microscope slide-mounted specimen was immersed in a xylene substitute bath until the paraffin was solubilized. The deparaffinized specimens were then washed with a series of alcohol solutions of decreasing alcohol concentration, to remove xylene, before a final wash with water. The tissue sections were then fixed with 4% formaldehyde in PBS for 15 min at 37.degree. C. Following washing in deionized water, tissue sections were stained with either 1 .mu.M thioflavin T in PBS or 500 nM YAT2150 dye for 3 min, rinsed in water and destained in 1% acetic acid for 20 min. Finally the tissues sections were washed thoroughly in water, dehydrated, covered with glass coverslips, mounted in anti-fade mounting medium and observed using a fluorescence microscope (Carl Zeiss, Inc.) with an FITC filter set for thioflavin T and a Texas Red filter set for YAT2150 dye, respectively. All images were acquired with a 63.times. objective lens (Carl Zeiss, Inc).
[0280] For the antibody co-localization studies, tissue sections were stained with YAT2150 dye as described above. The tissue sections were then blocked in PBS containing 3% bovine serum albumin (blocking buffer). Tau-reactive monoclonal antibody (clone tau-13) and Alexa Fluor.RTM. 488 labeled beta amyloid reactive monoclonal antibody (clone 6E10) were diluted to a concentration of 2 .mu.g/mL in blocking buffer and incubated for 1 h at room temperature. Tissues were then washed in PBS containing 0.1% Tween-20 for 15 min. For tissues incubated with Tau-13 antibody, the slides were subsequently incubated with Alexa Fluor.RTM. 488 goat anti-mouse secondary antibody for 30 min at room temperature. Finally the tissue sections were washed with PBS, covered with glass coverslips, mounted in anti-fade mounting medium and observed using a fluorescence microscope (Carl Zeiss, Inc.) with a Texas Red filter set for YAT2150 dye and FITC filter set for labeled antibodies, respectively. All images were acquired with a 63.times. objective lens (Carl Zeiss, Inc.).
[0281] Thioflavin T (ThT) is a widely employed histological probe for detecting the formation of amyloid fibrils in brain tissue (Gunilla et al., 1999). However, this dye is not an ideal predictor of the degree of fibrillization because its fluorescence varies substantially depending upon the structure and morphology of the amyloid fibrils. It was found that the dye generates fairly high background and weak fluorescent signal in brain tissue sections, as shown in FIG. 22A. Thus, optimized protocols for the detection of amyloid plaques in frozen and paraffin-embedded tissue sections of human brain were developed using the YAT2150 dye. Relative to ThT, this novel probe demonstrates significantly higher fluorescence emission intensity enhancement in the presence of amyloid protein fibrils and low non-specific background (shown in FIG. 22B). In addition, use of antibodies directed against .beta.-amyloid and tau protein, in conjunction with the YAT2150 dye, confirm the selectivity of the probe for detection of amyloid plaques in post-mortem brain tissue of patients with Alzheimer's disease (FIGS. 22A & B).
Example 32. Utilizing Flow Cytometry to Quantify the Accumulation of Protein Aggregates within Cells
[0282] Human leukemic Jurkat cells were obtained from ATCC. Jurkat cells were grown in suspension in RPMI medium supplemented with 10% (v/v) FBS, penicillin (100 U/ml), streptomycin (100 .mu.g/ml), and glutamine (200 mM). Jurkat cells were maintained in a saturated, humidified atmosphere at 37.degree. C., 5% CO.sub.2 and 95% air.
[0283] Jurkat cells were grown to log phase, and treated with 5 .mu.M MG-132 or with vehicle for 16 hours. At the end of the treatment, adherent cells were trypsinized; while Jurkat cells were simply collected by centrifugation (400.times.g for 5 min). Samples were resuspended at 1.times.10.sup.6 to 2.times.10.sup.6 cells per ml. For each group, triplicate samples were prepared. The cells were washed with PBS, fixed in 4% formaldehyde in PBS for 30 min and then permeabilized with 0.5% Triton X-100, 3 mM EDTA, pH 8 on ice, for 30 minutes. The cells were then washed, and resuspended in 500 .mu.L of 200 nM YAT2150 dye. The samples were incubated for 30 minutes at room temperature, protected from light. Experiments were performed using a FACS Calibur benchtop flow cytometer (BD Biosciences, San Jose, Calif.) equipped with a blue (488 nm) laser. YAT2150 dye fluorescence was measured in the FL3 channel. No washing was required prior to the flow cytometric analysis.
[0284] For the immunocytochemistry study, after fixing and permeabilizing the cells, the cells were blocked in PBS containing 3% bovine serum albumin for one hour. Fluorescein-labeled p62 antibody was diluted to a concentration of 2 .mu.g/mL in blocking buffer and incubated with the cells for 1 h at room temperature. Cells were then washed in PBS containing 0.1% Tween-20 for 15 min. Data was acquired by FACS Calibur benchtop flow cytometer (BD Biosciences, San Jose, Calif.) equipped with a blue (488 nm) laser, with the antibody signal measured in the FL1 channel.
[0285] All of the experiments were performed at least three times. Flow cytometry data were analyzed by comparison of mean fluorescence, through calculation of a term we refer to as the Aggregation Propensity Factor (APF), having the following definition.
APF=100.times.((MFI.sub.treated-MFI.sub.control)/MFI.sub.treated), wherein MFI.sub.treated and MFI.sub.control are the mean fluorescence intensity values from control and treated samples. This metric is based upon a similar approach that is commonly employed in the assessment of fluorescent signal between control and treated groups in multidrug resistance experiments, using a term referred to as Multidrug Resistance Activity Factor (MAF) (Hollo et al., 1994). APF is a unitless term measured as the difference between the amount of the YAT2150 dye accumulated within cells in the presence and absence of a proteasome inhibitor or other inducer of aggresome or inclusion body formation or protein aggregation. The fluorescence measurement in the presence of the proteasome inhibitor constitutes the maximal potential fluorescence for the given cell population when aggregated protein cargo has been generated. This represents a standardization method, which eliminates unknown cell type-specific variables that might influence dye accumulation, such as cell size, shape and volume, allowing the potential for intra- and inter-laboratory comparison of test results and APF values.
[0286] A flow cytometry cell-based assay was next developed using the YAT2150 dye. FIG. 24A demonstrates typical results of flow cytometry-based analysis of cell populations using the YAT2150 dye. Uninduced control and 5 .mu.M MG-132-treated Jurkat cells were employed in the investigation. After 16 hours treatment, fixed and permeabilized cells were stained with the YAT2150 dye and then analyzed without washing by flow cytometry. Results are presented using histogram overlay graphs. Control cells displayed minimal fluorescence staining with the dye. The YAT2150 dye signal increased about three-fold in the MG-132 treated cells, readily demonstrating that MG-132 induced protein aggregate formation in Jurkat cells. An APF value of approximately 72, as defined above, demonstrates that the control and treated cell populations were readily distinguishable by flow cytometry. For comparison, an MAF cut-off value of about 20-25 is routinely employed in flow cytometry assays of multi-drug resistance (Hollo et al., 1994). Protein aggregate accumulation in the Jurkat cells was confirmed by flow cytometry analysis using fluorescein-conjugated p62-reactive antibody (FIG. 24B). Thus, the described assay allows, for the first time, easy quantification of aggresome accumulation by flow cytometry. The advantage of the dye-based approach relative to the antibody one is that staining and analysis are much more rapid. Simultaneous staining with the fluorescein conjugated antibody and the red fluorescent dye was also feasible (data not shown).
REFERENCES
[0287] Arakawa T, Philo J S, Ejima D, Sato H, Tsumoto K (2007), "Aggregation analysis of therapeutic proteins, part 3". Bioprocess International 5 (10): 52-70 [0288] Arakawa T, Philo J S, Ejima D, Tsumoto K, Arisaka F (2006), "Aggregation analysis of therapeutic proteins, part 1". Bioprocess International 4 (10): 32-42. [0289] Arakawa T, Philo J S, Ejima D, Tsumoto K, Arisaka F (2007), "Aggregation analysis of therapeutic proteins, part 2". Bioprocess International 5 (4): 36-47. [0290] Beaudoin S, Goggin K, Bissonnette C, Grenier C, Roucou X (2008) Aggresomes do not represent a general cellular response to protein misfolding in mammalian cells. BMC Cell Biology 9:59. [0291] Bondos S and A Bicknell (2003), "Detection and prevention of protein aggregation before, during, and after purification". Analytical Biochemistry 316: 223-231. [0292] Capelle Martinus A H, Gurny R, Arvinte T (2009), "A high throughput protein formulation platform: Case study of salmon calcitonin". Pharmaceutical Research 26(1): 118-128. [0293] Demeule B, Gurny R, Arvinte T (2007), "Explorations of the application of cyanine dyes for quantitative .alpha.-synuclein detection". International Journal of Pharmaceutics 329: 37-45. [0294] Ericsson U B, Hallberg B M, DeTitta G T, Dekker N, Nordlund P (2006), "Thermofluor-based high-throughput stability optimization of proteins for structural studies". Analytical Chemistry 357: 289-298. [0295] Ferrari D M, Soling H D (1999), "The protein disulfide-isomerase family: unravelling a string of folds". Biochem. J. 339: 1-10). [0296] Garcia-Mata et al (2002) "Hassles with Taking Out the Garbage: Aggravating Aggresomes" Traffic 3: 388-396. [0297] Gunilla T, Westermark, Kenneth H. Johnson, Per Westermark (1999) Staining methods for identification of amyloid in tissue: Methods in Enzymology 1999. 309:3-25. [0298] Hawe et al. (2008), "Extrinsic Fluorescent Dyes as Tools for Protein Characterization," Pharmaceutical Research, 25:1488. [0299] Hollo Z, Homolya L, Davis C W, Sarkadi B (1994) Calcein accumulation as a fluorometric functional assay of the multidrug transporter. Biochim Biophys Acta 1191(2):384-388. [0300] Kawaguchi Y, Kovacs J J, McLaurin A, Vance J M, Ito A, Yao T P (2003) The deacetylase HDAC6 regulates aggresome formation and cell viability in response to misfolded protein stress. Cell 2003.115:727-738. [0301] Khurana R, Uversky V N, Nielsen L, Fink A L (2001), "Is Congo Red an Amyloid-specific Dye". J. Biol. Chem 276: 22715-22721. [0302] Klunk W E, Debnath M L, Koros A M, Pettegrew J W (1998) "Chrysamine-G, a lipophilic analogue of Congo Red, inhibits A beta-induced toxicity in PC12 cells.". Life Sci. 63: 1807-1814. [0303] Krishnamurthy R, Sukumar M, Das T K, Lacher N A (2008), "Emerging analytical technologies for biothererapeutics development". Bioprocess International 6 (5): 32-42. [0304] Kumar, S., Vijay Kumar Ravi and Rajaram Swaminanthan (2009), "Suppression of Lysozyme aggregation at alkaline pH by tri-N-acetyl-chitotriose" Biochimica et Biophysica Acta 1294: 913-920. [0305] Li et al. (2004) ".DELTA.12-Prostaglandin J2 inhibits the ubiquitin hydrolase UCH-L1 and elicits ubiquitin-protein aggregation without proteasome inhibition". Biochemical and Biophysical Research Communications 319: 1171-1180. [0306] Lindgren et al. (2005), Biophysical J 88: 4200-4212. [0307] Lundstrom J, Holmgren A (1990), "Protein disulfide-isomerase is a substrate for thioredoxin reductase and has thioredoxin-like activity". J. Biol. Chem. 265 (16): 9114-9120. [0308] Lyles M M, Gilbert H F (1991). "Catalysis of the oxidative folding of ribonuclease A by protein disulfide isomerase: dependence of the rate on the composition of the redox buffer" Biochemistry 30 (3): 613-619. [0309] Matulis D, Kranz J K, Salemme F R, Todd M J (2005), "Thermodynamic stability of carbonic anhydrase: Measurements of binding affinity and stoichiometry using thermofluor". Biochemistry 44: 5258-5266. [0310] Mezzasalma T M, Kranz J K, Chan W, Struble G T, Schalk-Hihi C, Deckman I C, Springer B A, Todd M J (2007), "Enhancing recombinant protein quality and yield by protein stability profiling". J. Biomolecular Dcreening 12 (3): 418-428. [0311] Morris A M, M A. Watzky, R G Finke (2009), Biochimica et Biophysica Acta (BBA)--Proteins & Proteomics 1794: 375-397. [0312] Mu T W, Ong D S, Wang Y J, Balch W E, Yates J R 3rd, Segatori L, Kelly J W (2008) Proteostasis regulators and pharmacologic chaperones synergise to correct protein misfolding diseases. Cell 134 (5):769-781. [0313] Murakami K, Irie K, Morimoto A, Ohigashi H, Shindo M, Nagao M, Shimizu T, Shirasawa T (2003), "Neurotoxicity and Physicochemical Properties of A.beta. Mutant Peptides from Cerebral Amyloid Angiopathy: Implication for the pathogenesis of cerebral amyloid angiopathy and Alzheimer's disease". J. Biol. Chem 278: 46179-46187. [0314] Murakawa Y, Sonoda E, Barber L J, Zeng W, Yokomori K, Kimura H, Niimi A, Lehmann A, Zhao G Y, Hochegger H, Boulton S J, Takeda S (2007) Inhibitors of the proteasome suppress homologous DNA recombination in mammalian cells, Cancer Res 2007. 67(18):8536-8543. [0315] Nawrocki S T, Carew J S, Pino M S, Highshaw R A, Andtbacka R H, Dunner K Jr, Pal A, Bornmann W G, Chiao P J, Huang P, Xiong H, Abbruzzese J L, McConkey D J (2006) Aggresome disruption: a novel strategy to enhance bortezomib-induced apoptosis in pancreatic cancer cells. Cancer Res 66:3773-3781. [0316] Puig A, Gilbert H F (1994), "Protein disulfide isomerase exhibits chaperone and anti-chaperone activity in the oxidative refoding of lysozyme". Journal of Biological Chemistry 269(10): 7764-7771. [0317] Raibekas A A (2008), "Estimation of protein aggregation propensity with a melting point apparatus". Analytical Biochemistry, 380: 331-332. [0318] Raturi A, Mutus B (2007). "Characterization of redox state and reductase activity of protein disulfide isomerase under different redox environments using a sensitive fluorescent assay". Free Radic. Biol. Med. 43 (1): 62-70. [0319] Renna M, Jimenez-Sanchez M, Sarkar S, Rubinsztein D C (2010) Chemical inducers of autophagy that enhance the clearance of mutant proteins in neurodegenerative diseases. J Biol Chem. 285(15):11061-11067. [0320] Sarkar S, Perlstein E O, Imarisio S, Pineau S, Cordenier A, Maglathlin R L, Webster J A, Lewis T A, O'Kane C J, Schreiber S L, Rubinsztein D C (2007) Small molecules enhance autophagy and reduce toxicity in Huntington's disease models. Nat Chem Biol. 3(6):331-338. [0321] Smith A M, Chan J, Oksenberg D, Urfer R, Wexler D S, O W A, Gao L, McAlorum A, Huang S (2004). "A high-throughput turbidometric assay for screening inhibitors of protein disulfide isomerase activity". J. Biomolecular Screening 9 (7): 614-620). [0322] Stefani M, Dobson C M (2003), "Protein aggregation and aggregate toxicity: new insights into protein folding, misfolding diseases and biological evolution". J. Mol. Med. 81: 678-699. [0323] Stefani (2004) "Protein misfolding and aggregation: new examples in medicine and biology of the dark side of the protein world". Biochimica et Biophysica Acta 1739: 5-25. [0324] Todd M J, Cummings M D, Nelen M I (2005), "Affinity assays for decrypting protein targets of unkown function". Drug Discovery Today: Technology 2 (3): 267-273. [0325] van Raaij, M. E., Segers-Nolten, I. M., Subramaniam, V. (2006) Biophys J. 91: L96 [0326] Volkova K D, Kovalska V B, Balanda A O, Losytskyy My, Golub A G, Vermeij R J, Subramaniam V, Tolmachev O I, Yarmoluk S M (2008), "Specific fluorescent detection of fibrillar .alpha.-synuclein using mono- and trimethine cyanine dyes". Bioorganic & Medicinal Chemistry 16: 1452-1459. [0327] Volkova K D, Kovalska V B, Balanda A O, Losytskyy My, Golub A G, Vermeij R J, Subramaniam V, Tolmachev O I, Yarmoluk S M (2008), "Specific fluorescent detection of fibrillar .alpha.-synuclein using mono- and trimethine cyanine dyes". Bioorganic & Medicinal Chemistry 16: 1452-1459. [0328] Volkova K D, Kovalska V B, Balanda A O, Vermeij R J, Subramaniam V, Slominskii Y L, Yarmoluk S M (2007), "Cyanine dye-protein interactions: looking for fluorescent probes for amyloid structures". J. Biochem. Biophys. Methods 70: 727-733. [0329] Volkova K D, Kovalska V B, Balanda A O, Vermeij R J, Subramaniam V, Slominskii Y L, Yarmoluk S M (2007), "Cyanine dye-protein interactions: looking for fluorescent probes for amyloid structures". J. Biochem. Biophys. Methods 70: 727-733). [0330] Volkova K D, Kovalska V B, Segers-Nolten G M, Veldhuis G, Subramaniam V, Slominskii Y L, Yarmoluk S M (2009), "Detection and characterization of protein aggregates by fluorescence microscopy". Biotechnic & Histochemistry 84 (2): 55-61. [0331] U.S. Pat. No. 4,868,103. [0332] U.S. Pat. No. 5,118,801. [0333] U.S. Pat. No. 5,192,737. [0334] U.S. Pat. No. 5,210,015. [0335] U.S. Pat. No. 5,538,848. [0336] U.S. Pat. No. 5,401,847. [0337] U.S. Pat. No. 5,580,990. [0338] U.S. Pat. No. 5,646,264. [0339] U.S. Pat. No. 5,800,996. [0340] U.S. Pat. No. 5,925,517. [0341] U.S. Pat. No. 6,008,373. [0342] U.S. Pat. No. 6,020,141. [0343] U.S. Pat. No. 6,593,465. [0344] U.S. Pat. No. 6,685,940. [0345] U.S. Pat. No. 6,737,401. [0346] U.S. Pat. No. 6,977,142. [0347] US Patent Application Publication 2003/0225247. [0348] US Patent Application Publication 2008/0125361. [0349] US Patent Application Publication 2005/0137388.
[0350] In view of the above, it will be seen that several objectives of the invention are achieved and other advantages attained.
[0351] As various changes could be made in the above methods and compositions without departing from the scope of the invention, it is intended that all matter contained in the above description and shown in the accompanying drawings shall be interpreted as illustrative and not in a limiting sense.
[0352] All references cited in this specification are hereby incorporated by reference. The discussion of the references herein is intended merely to summarize the assertions made by the authors and no admission is made that any reference constitutes prior art. Applicants reserve the right to challenge the accuracy and pertinence of the cited references.
Sequence CWU 1
1
2192DNAArtificial SequenceTemplate used in primer extension
experiment 1acttcttact tcttacttct tacttcttac ttcttacttc ttacttctta
cttcttactt 60cttcattggt catctcgatc catgacctca gc 92222DNAArtificial
SequencePrimer used in primer extension experiment 2ttgctgaggt
catggatcga ga 22












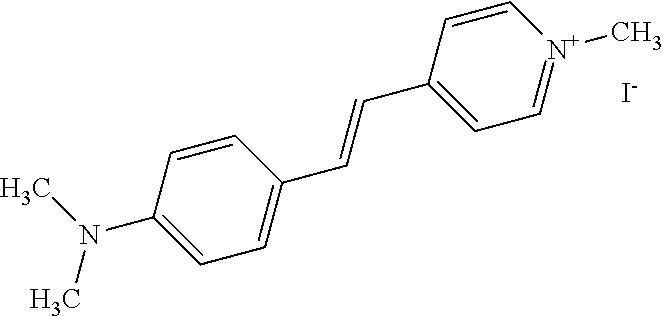
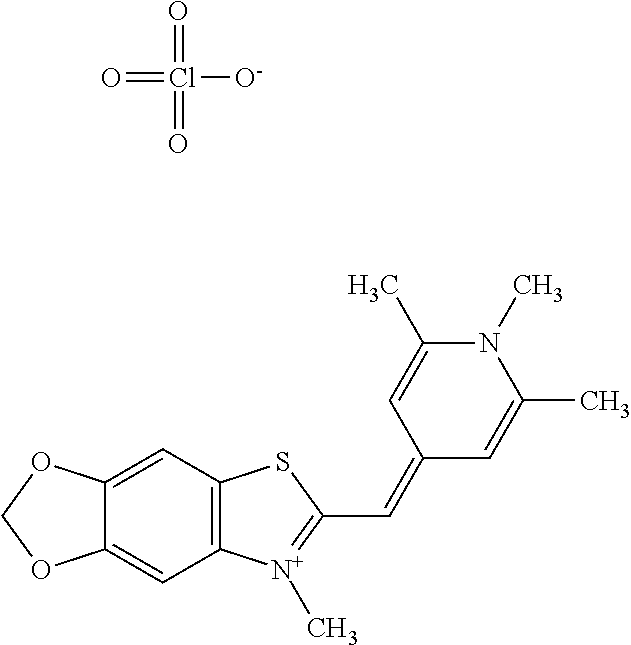
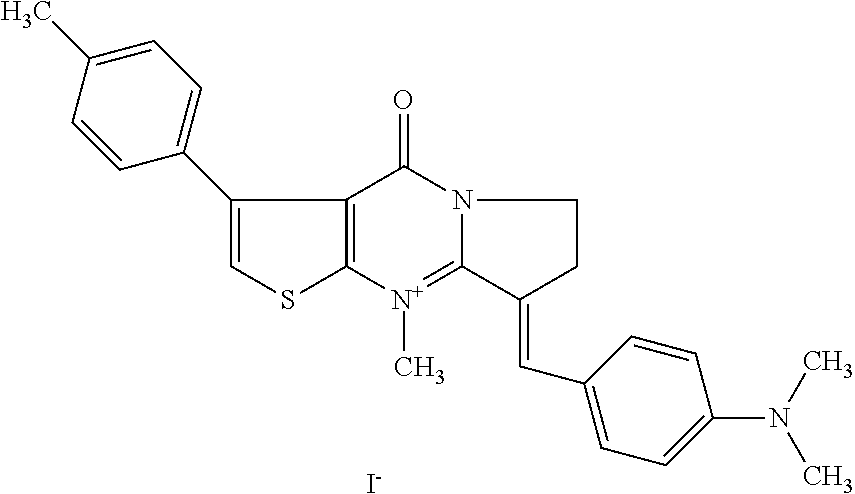
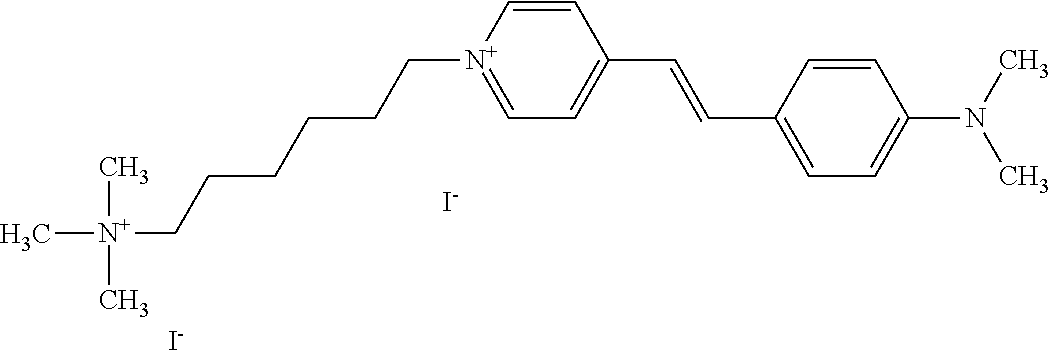
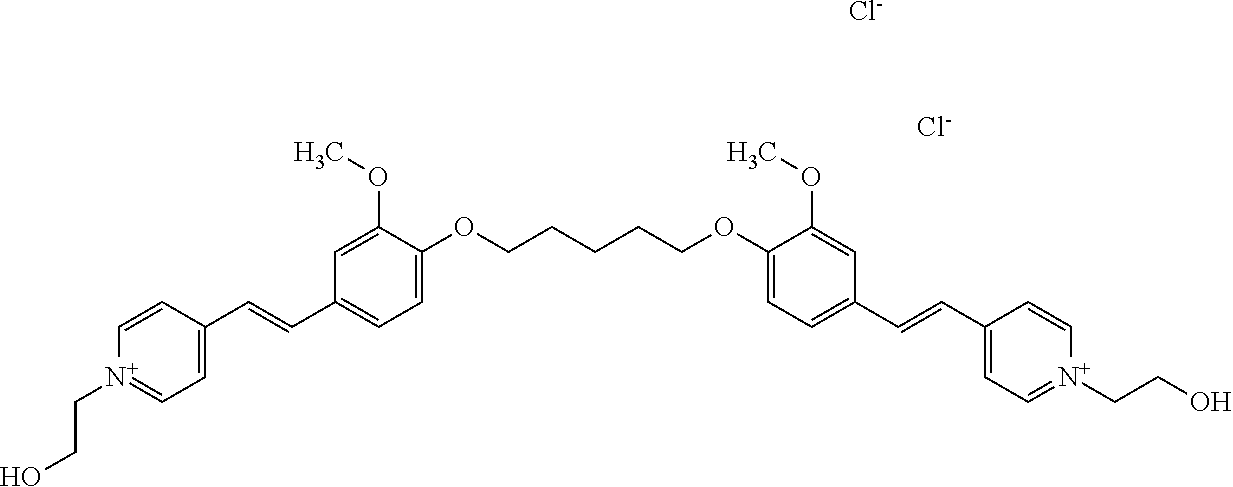
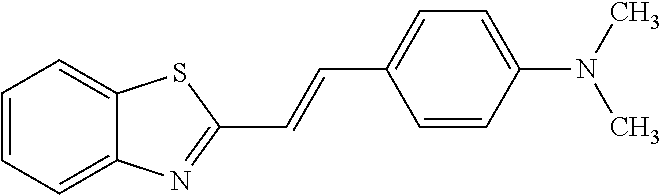
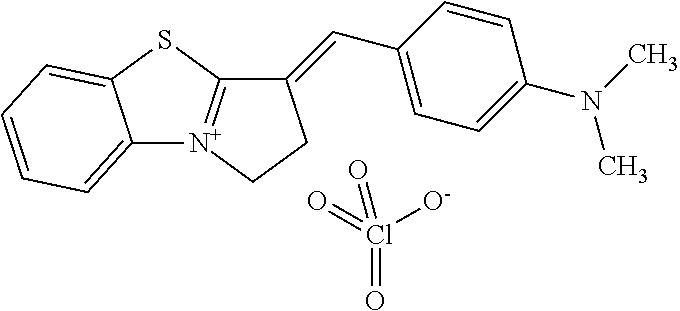
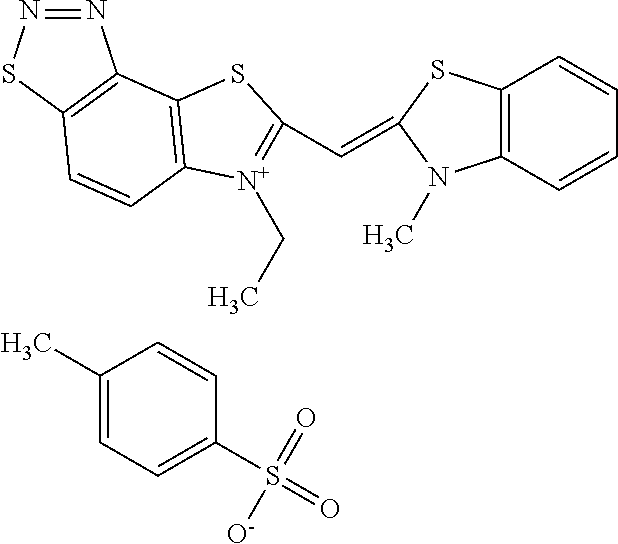
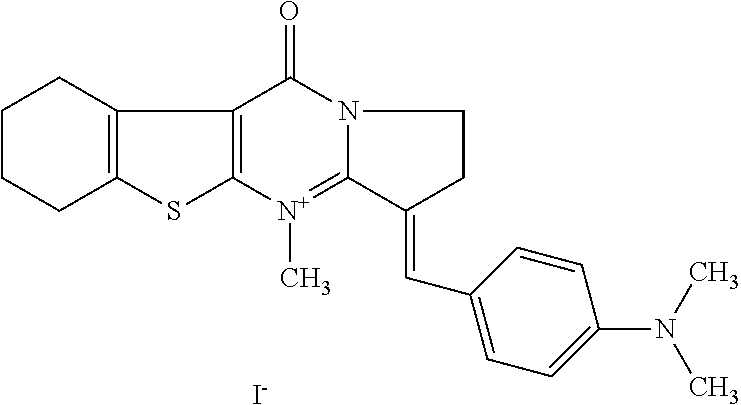
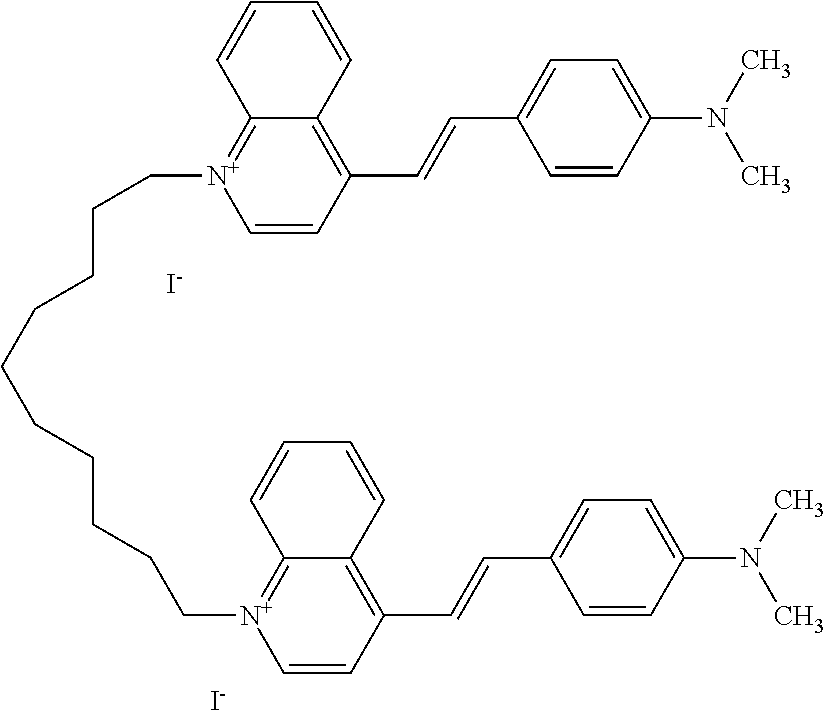
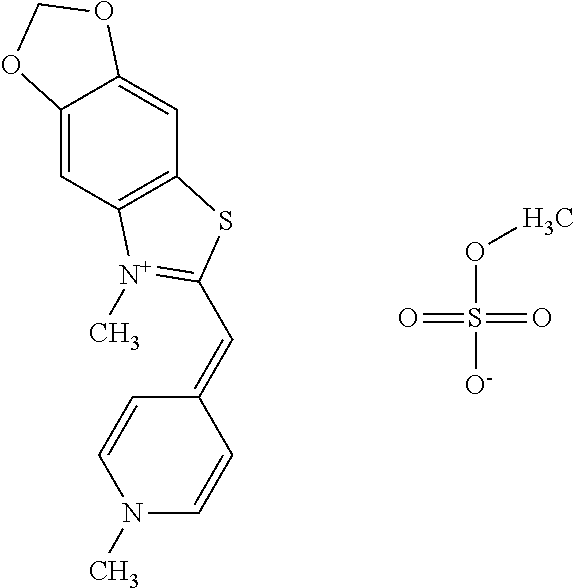
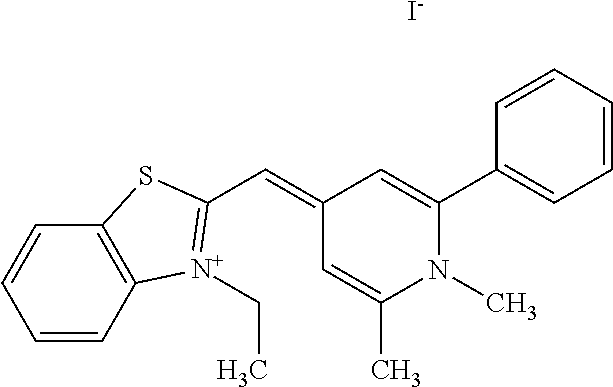
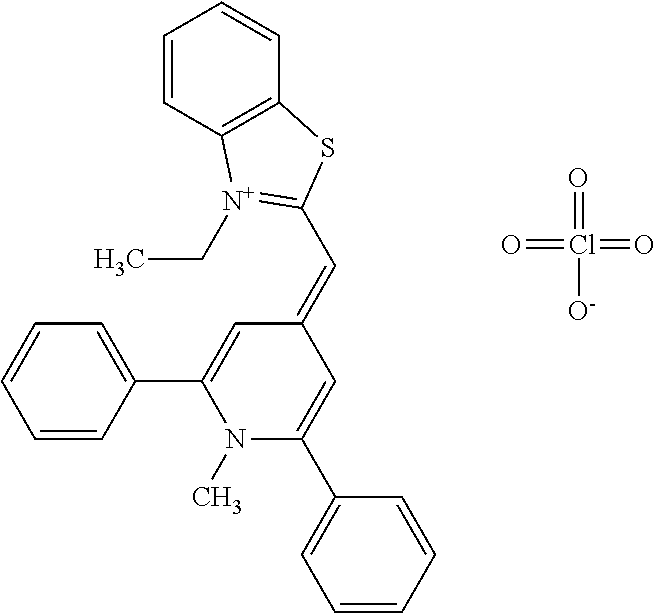
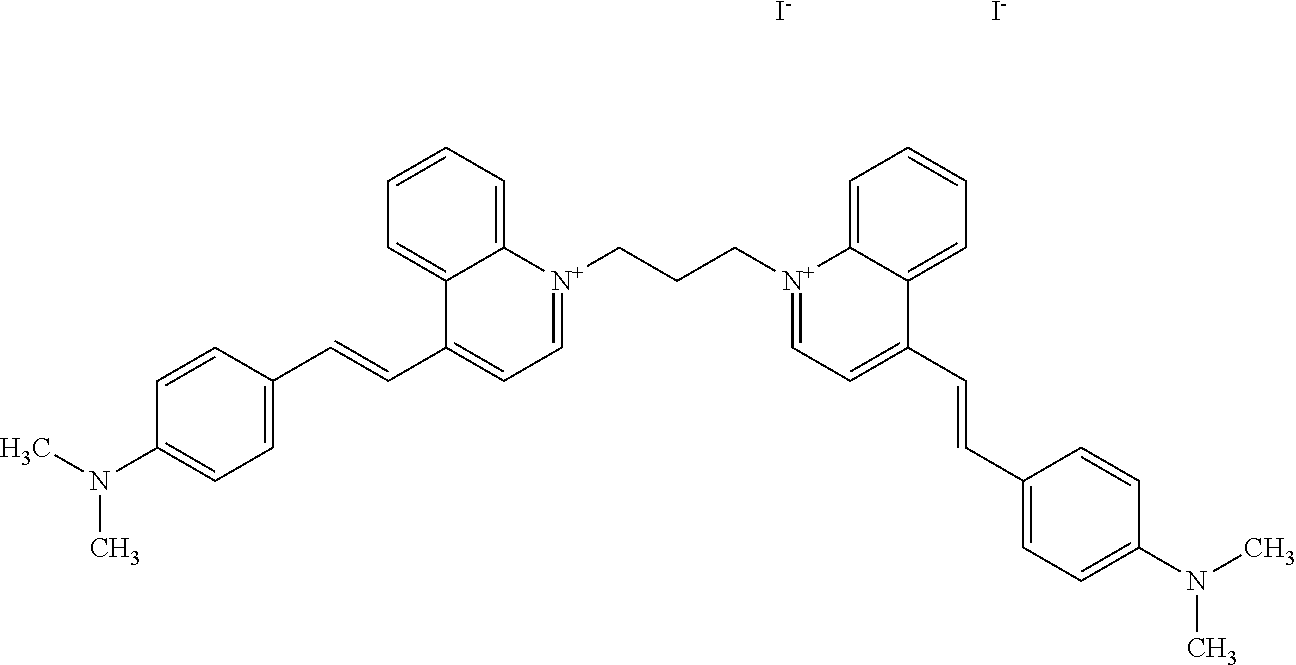
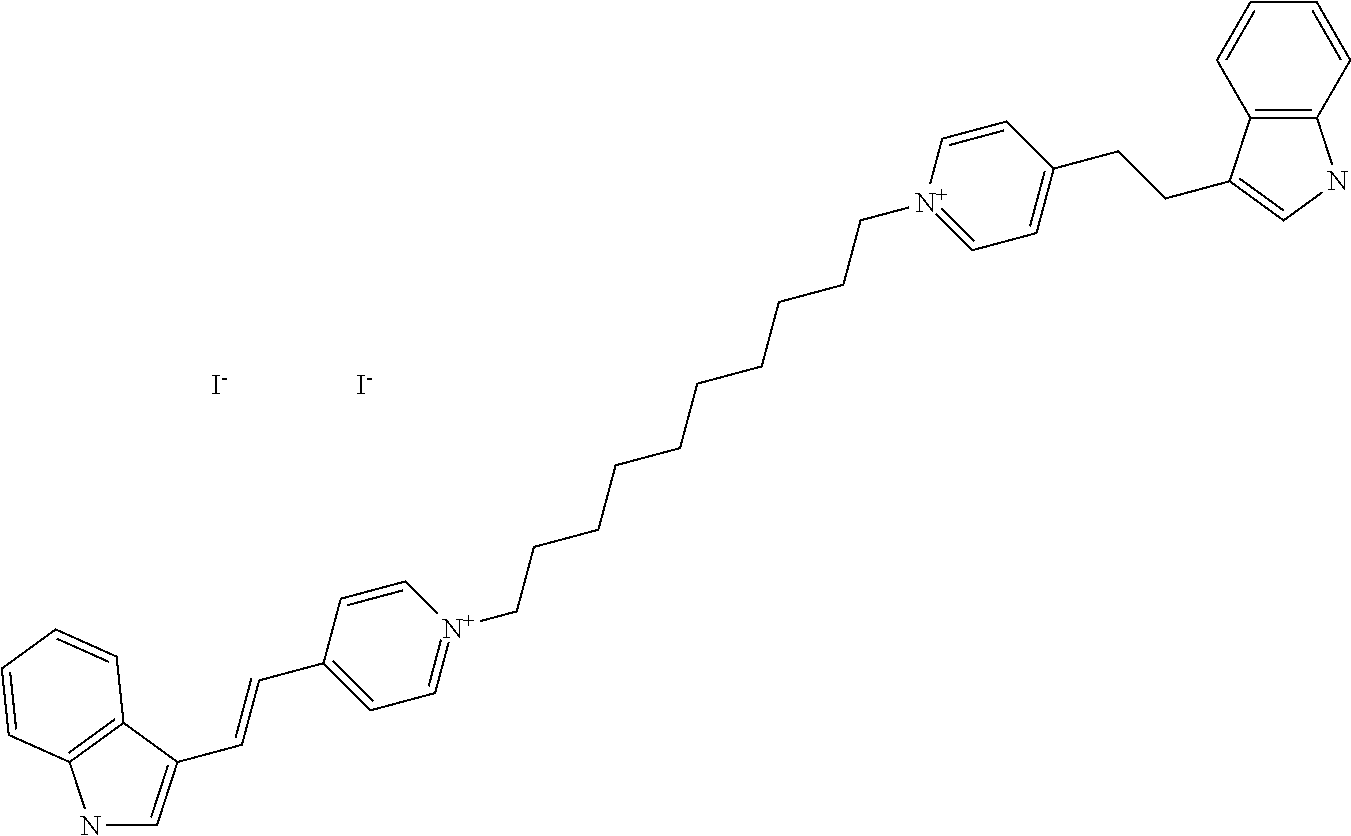
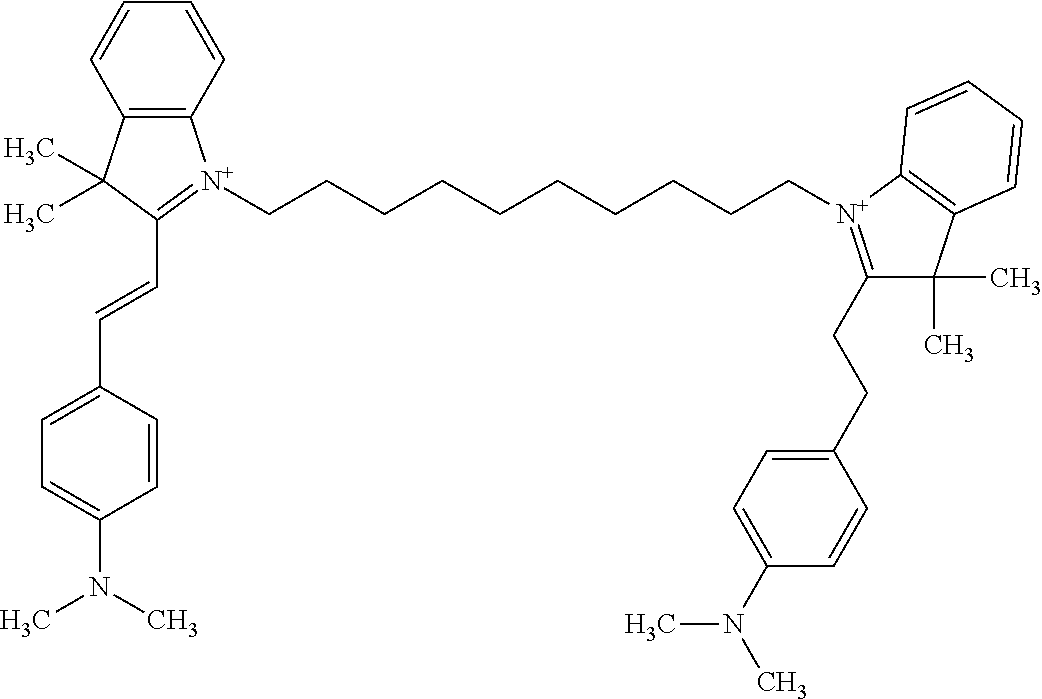
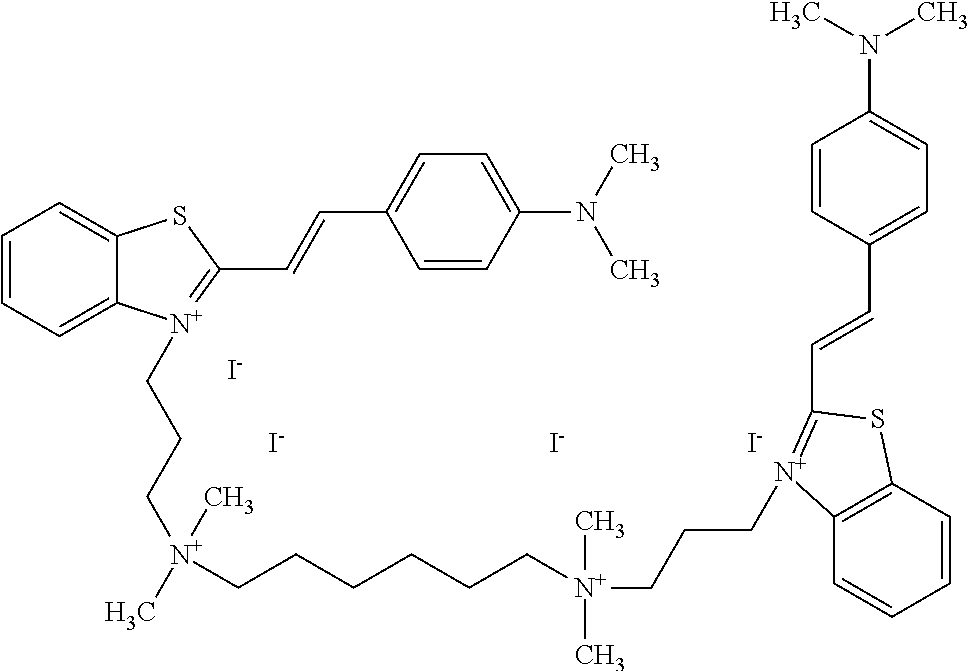
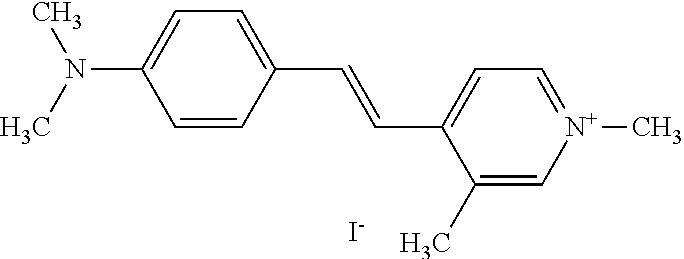
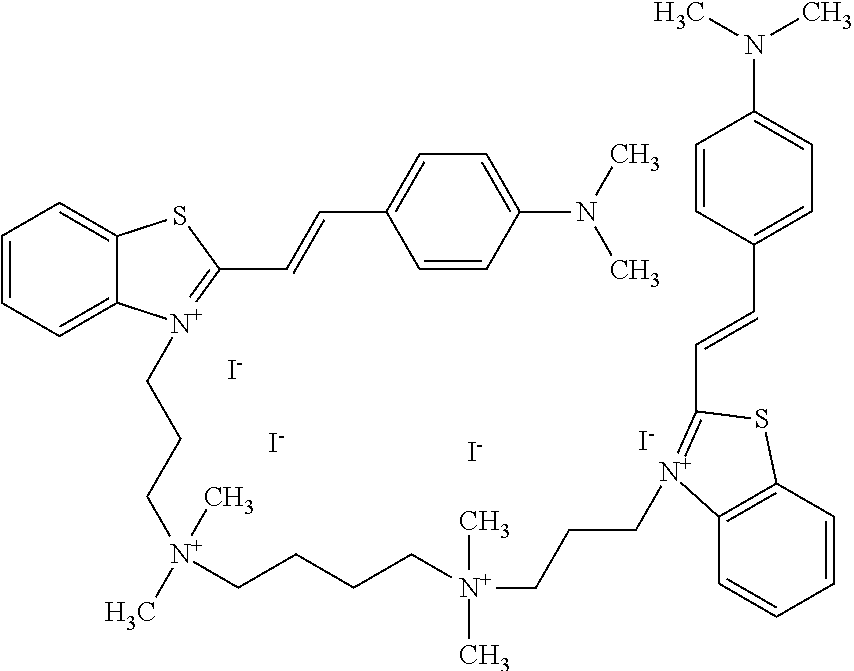
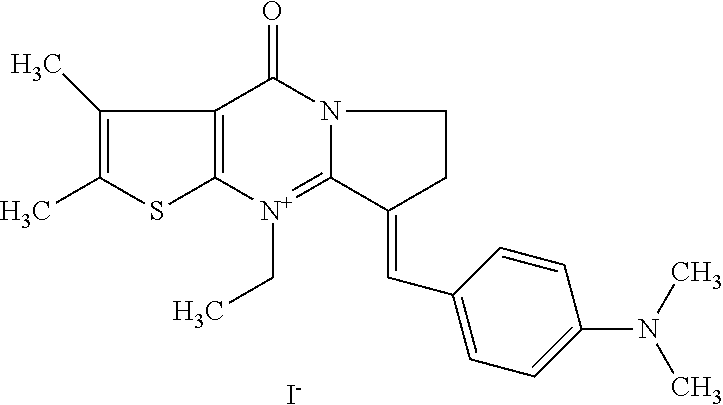
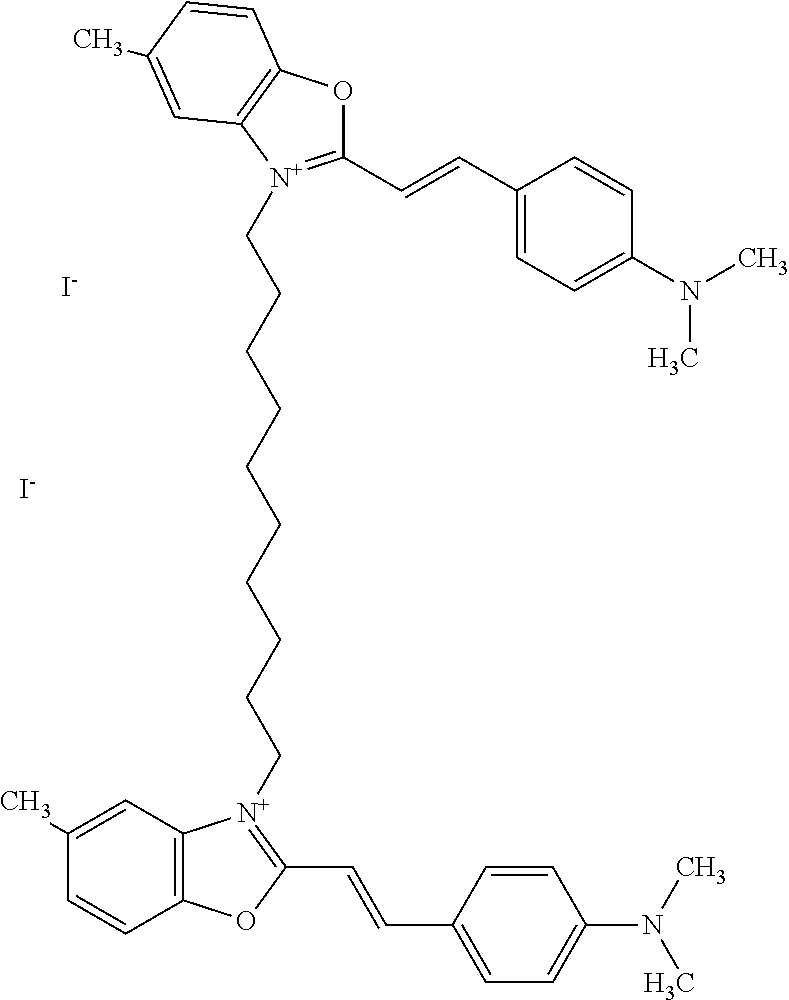
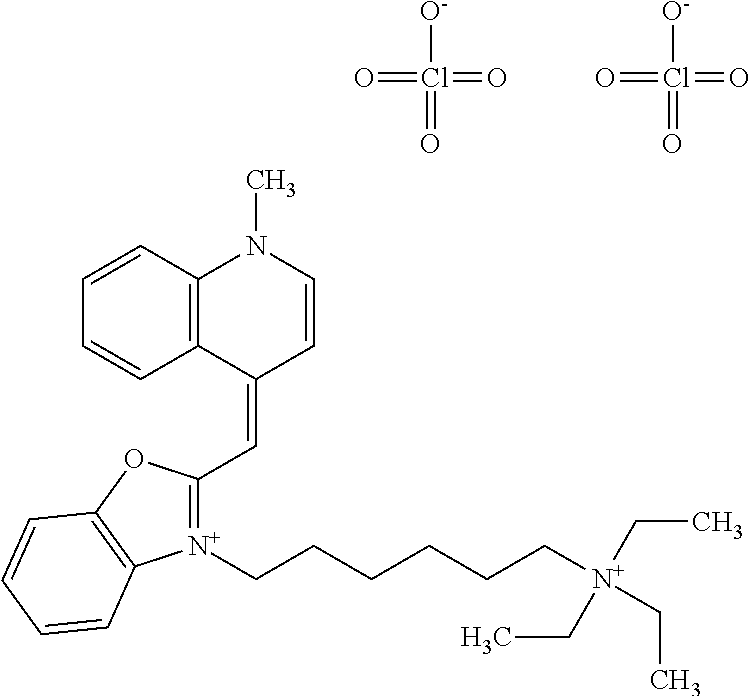
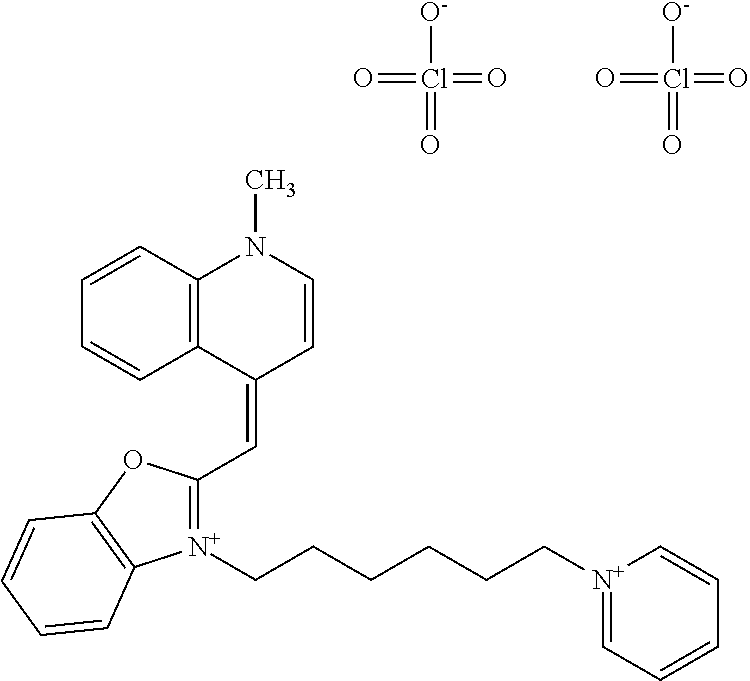
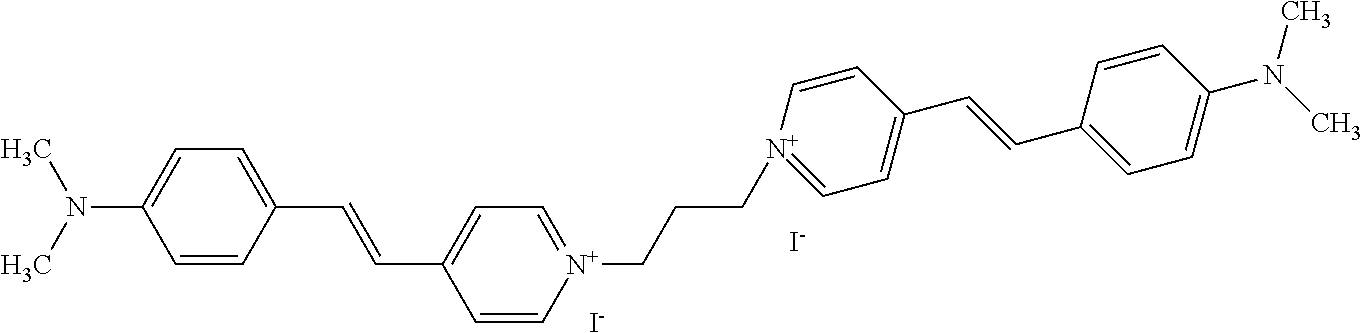
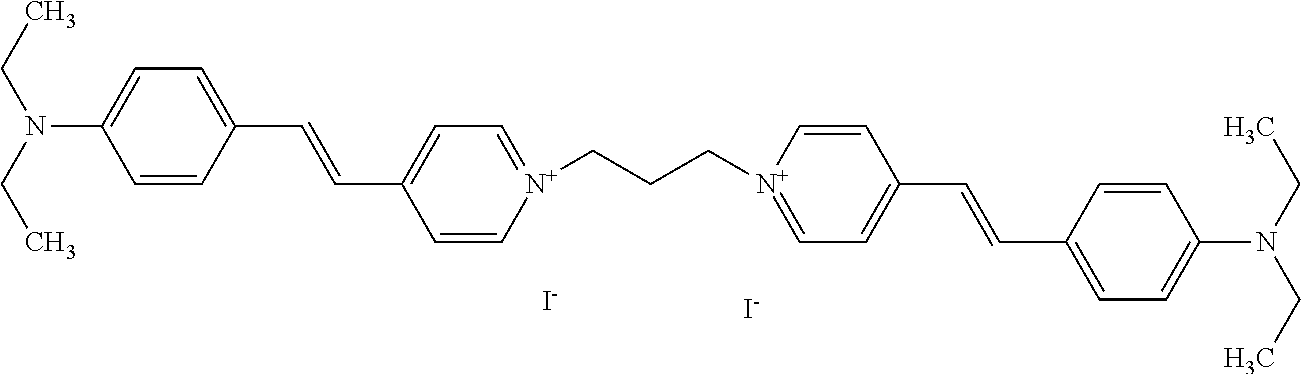
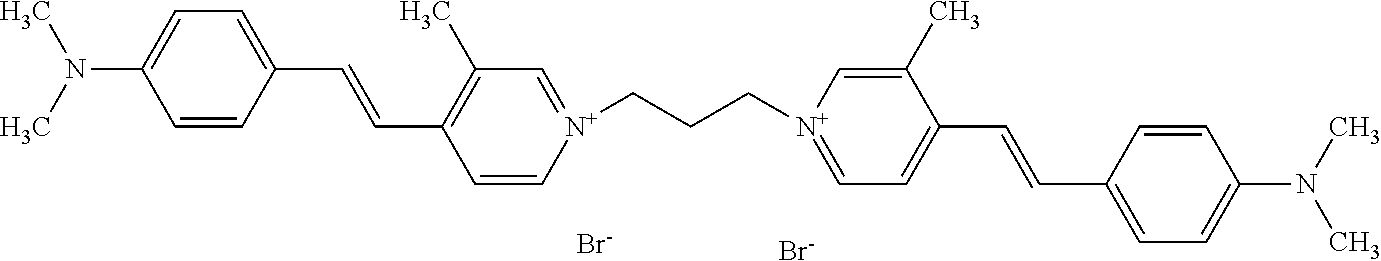
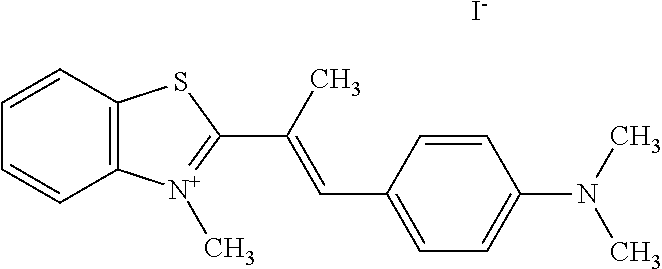
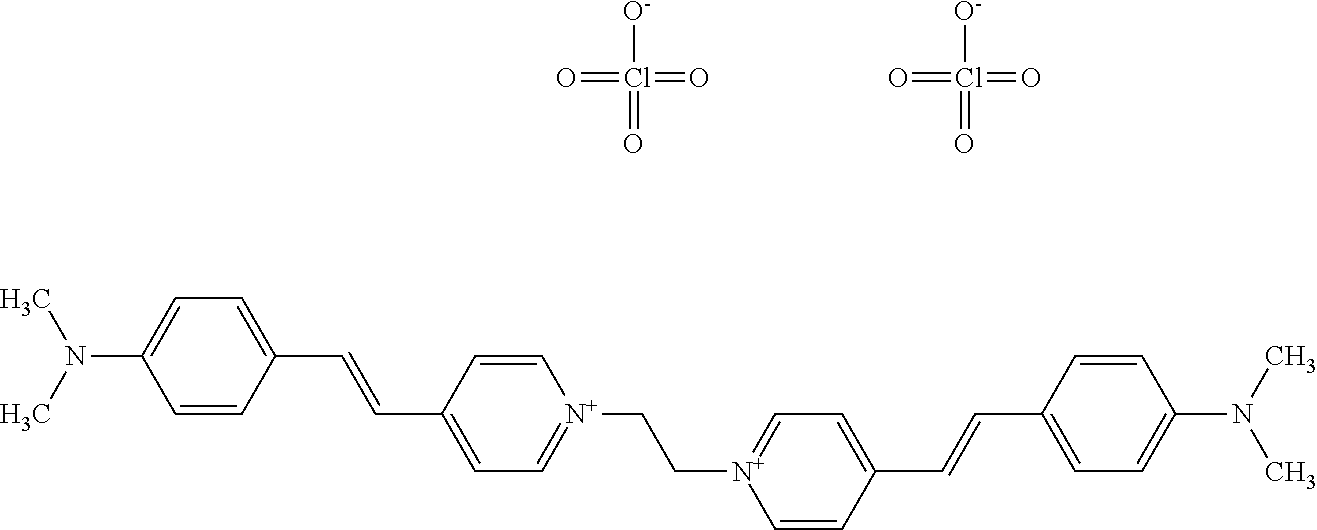
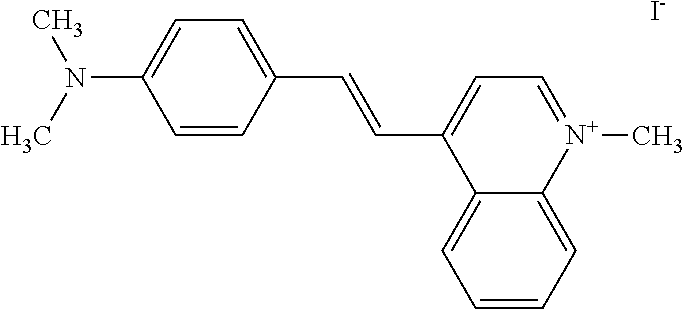
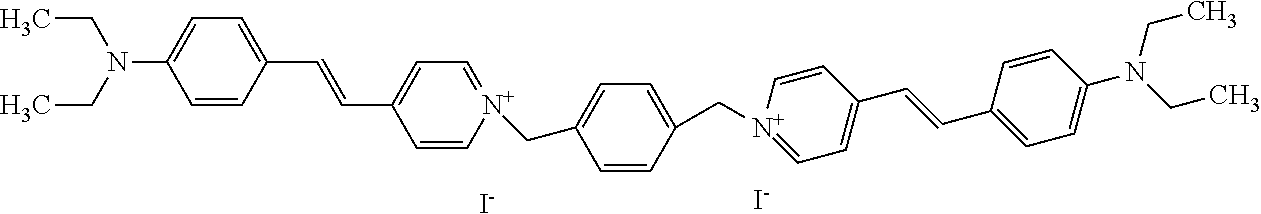
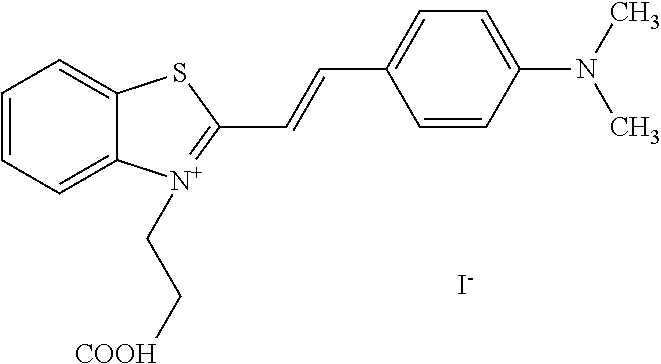
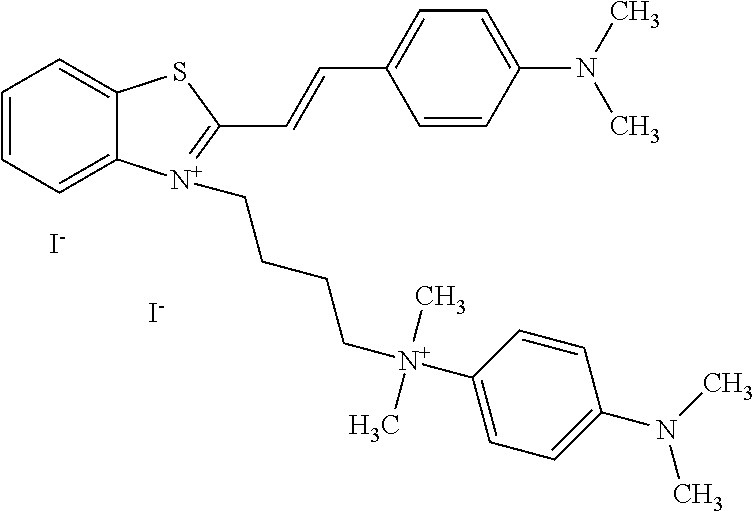
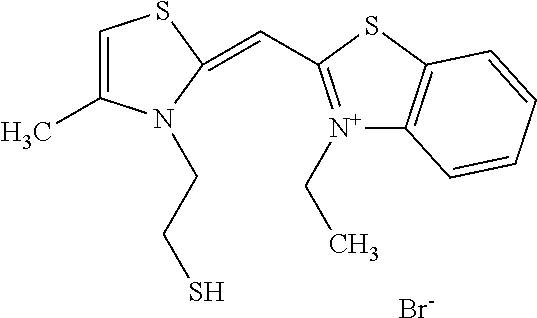
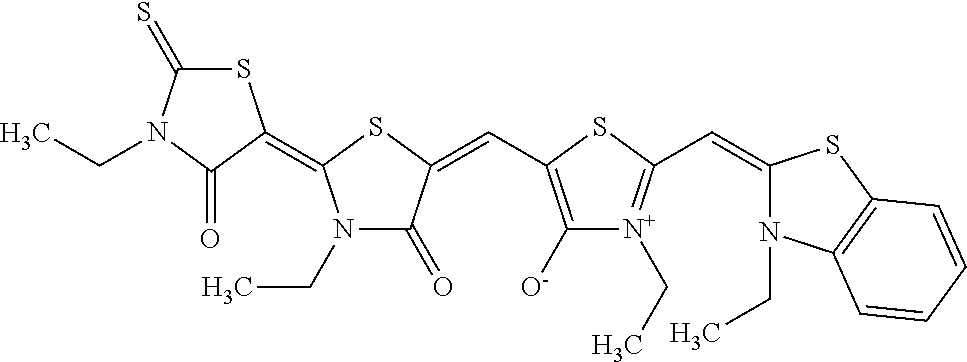
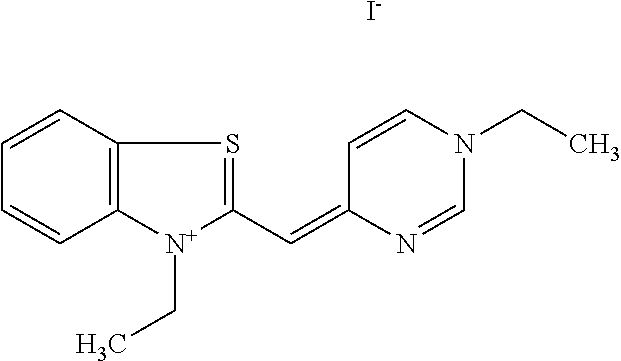
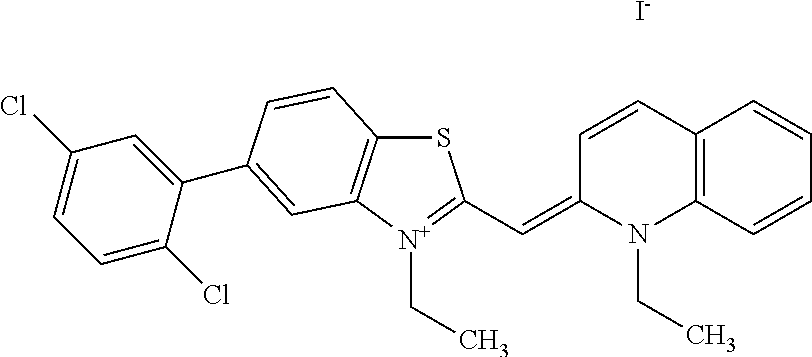
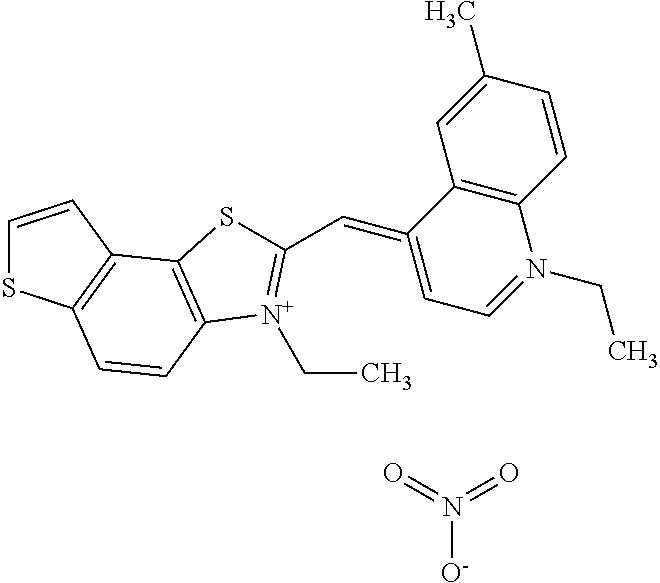
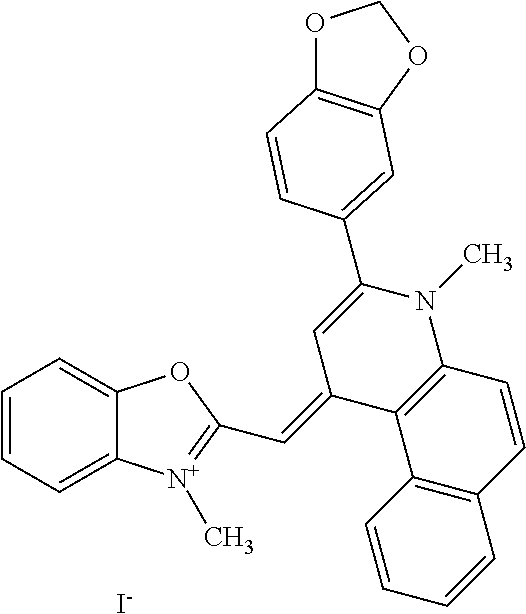
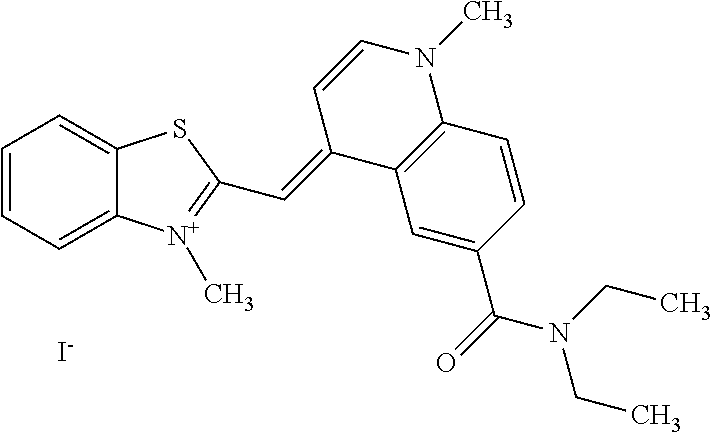
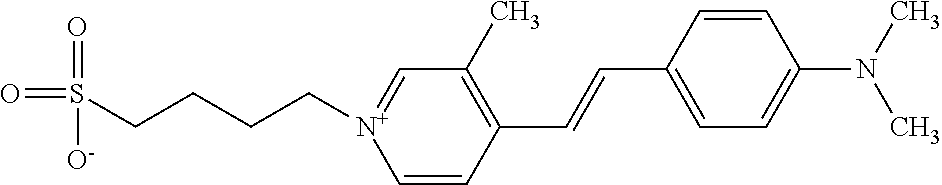
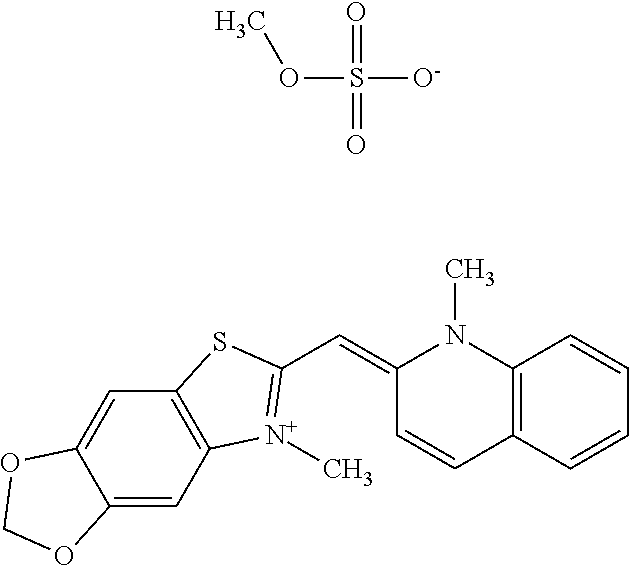
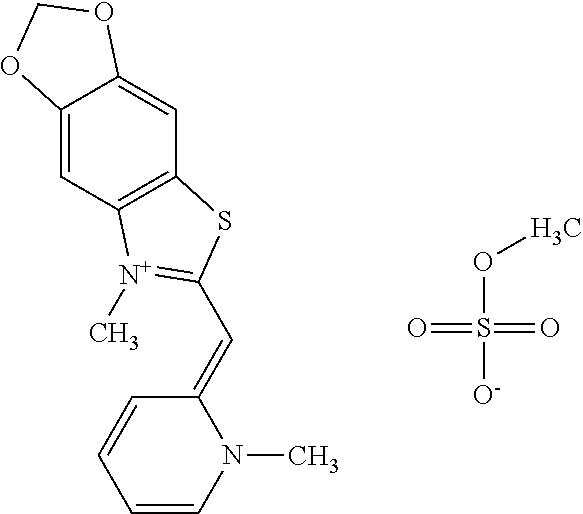
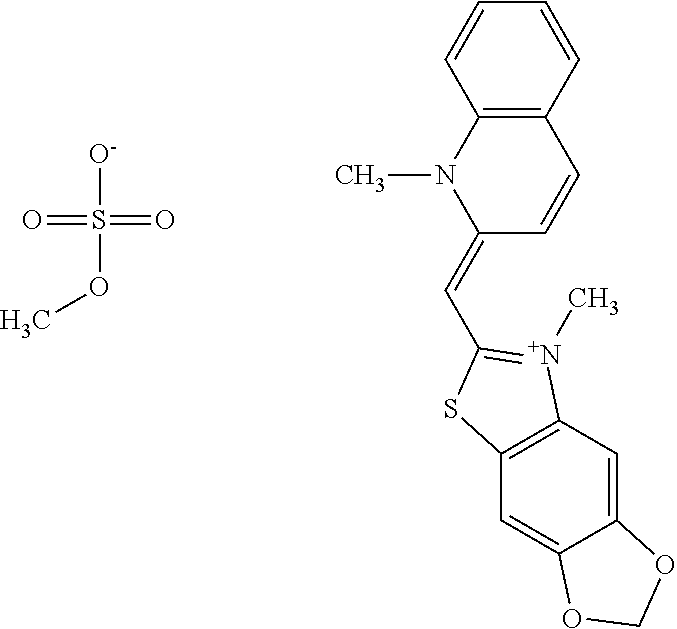
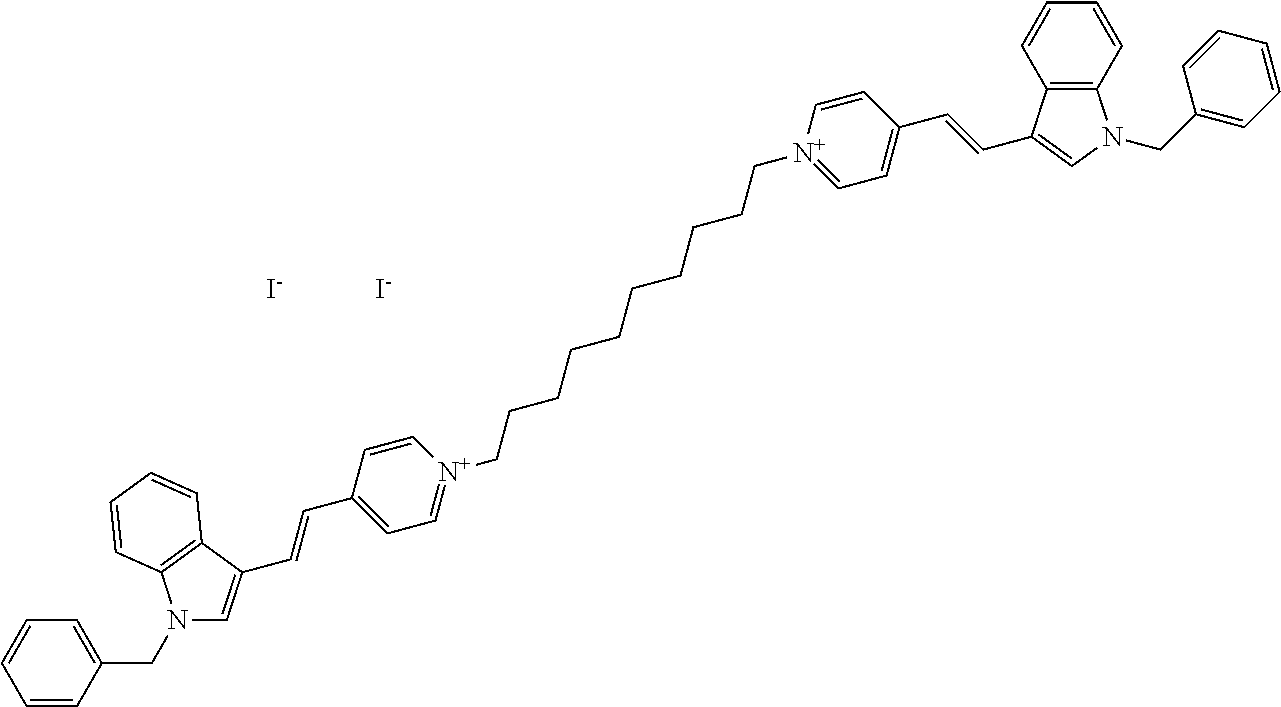
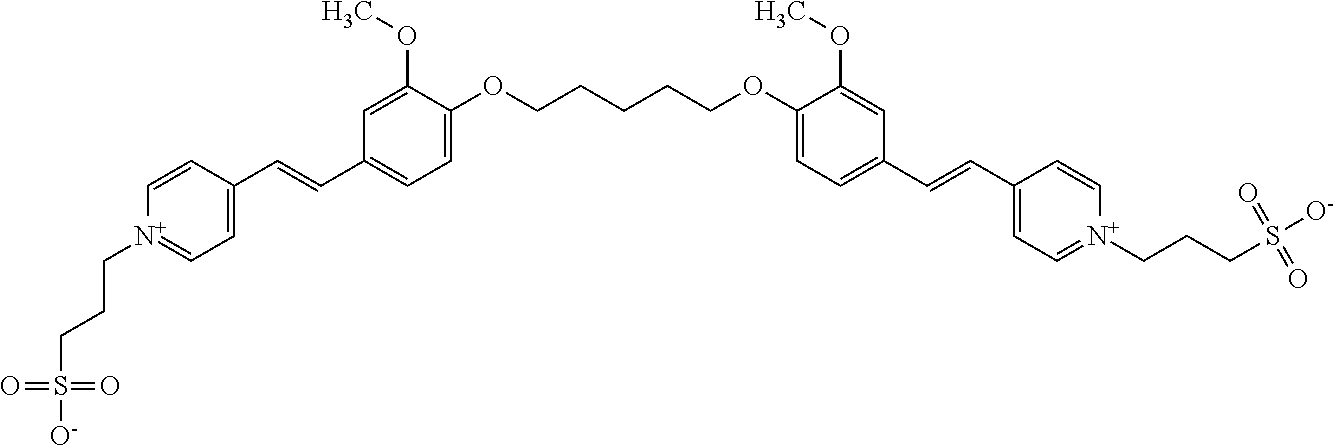
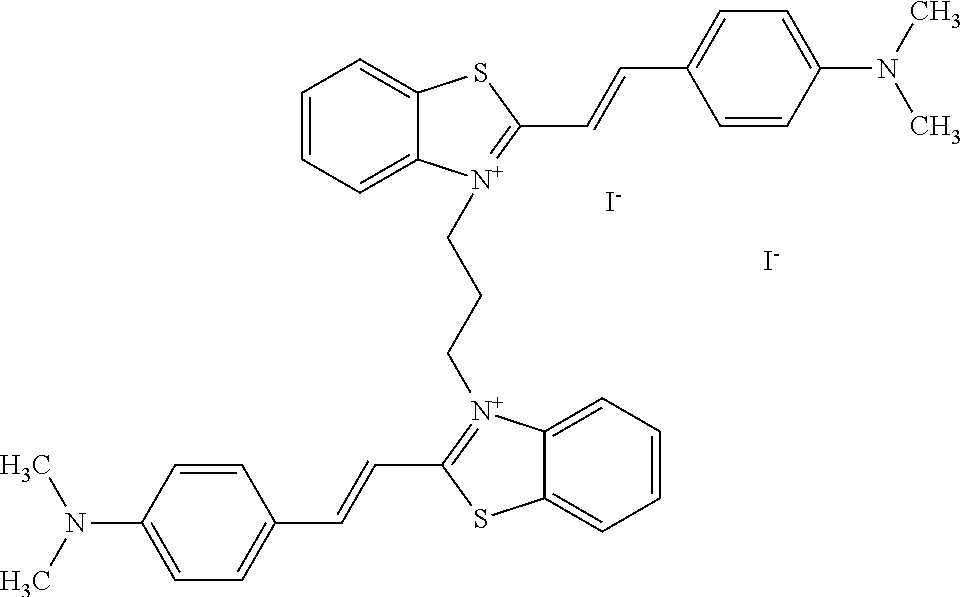
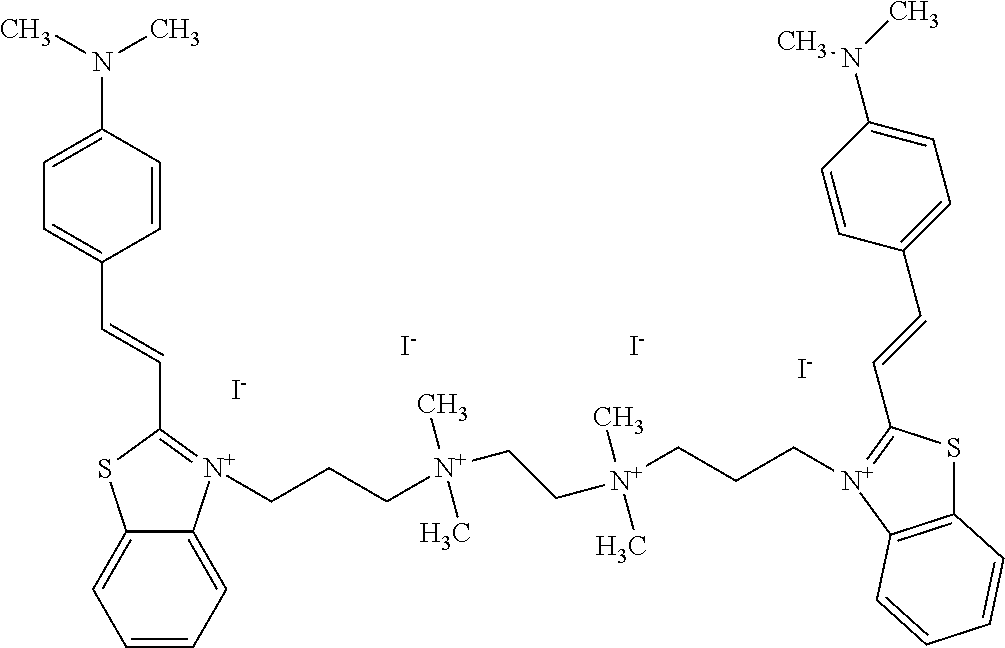

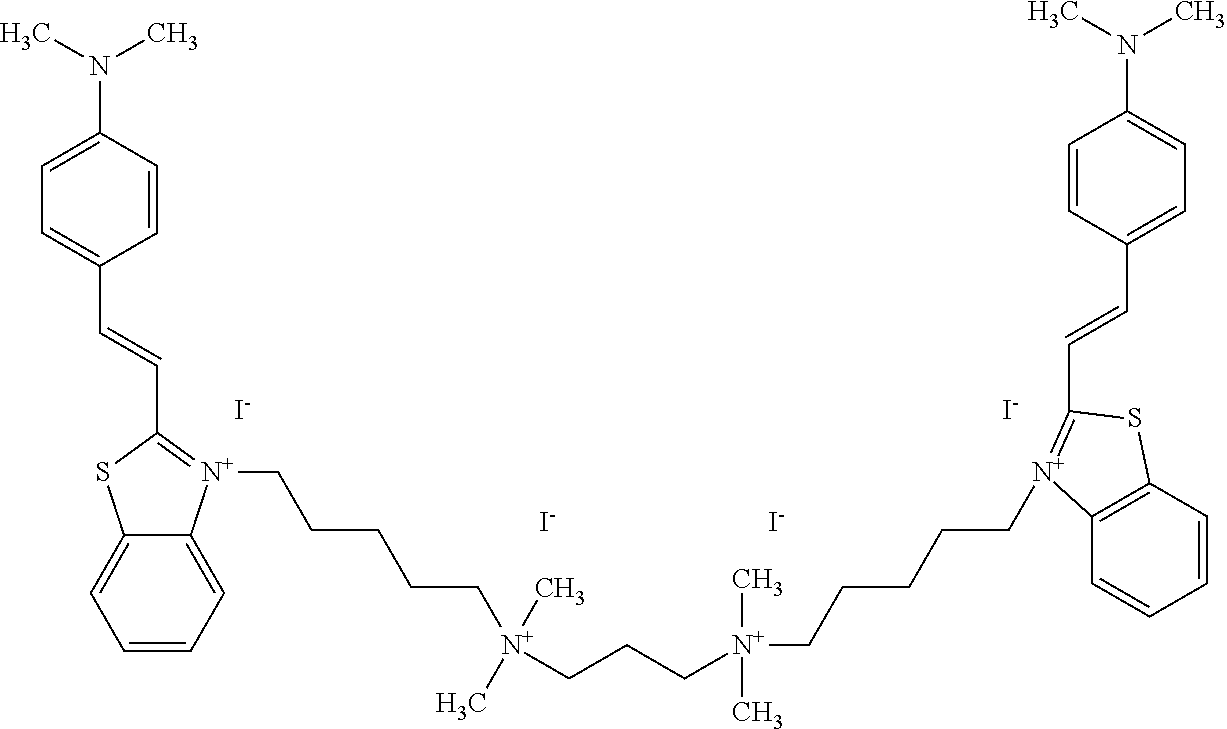
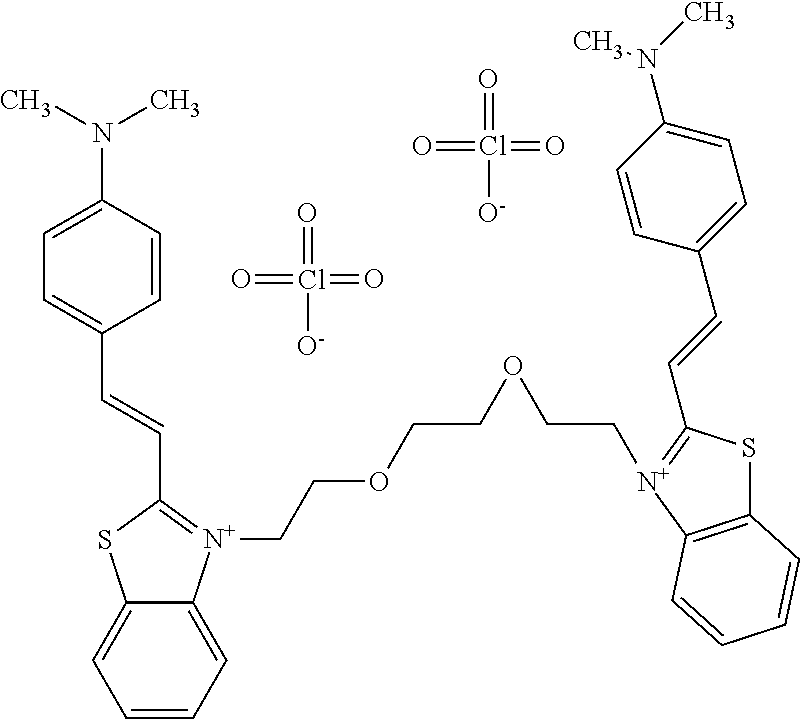
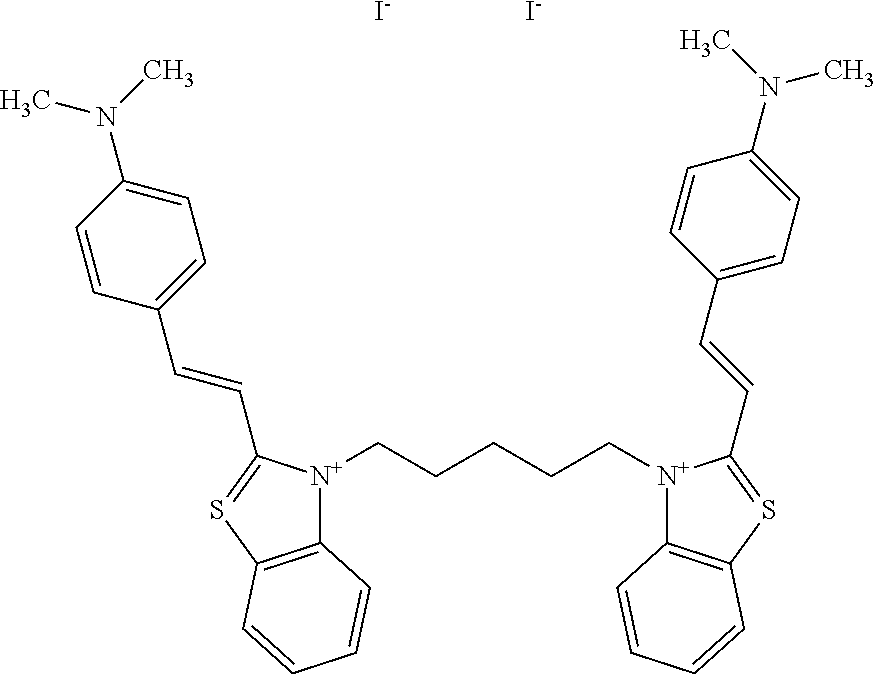
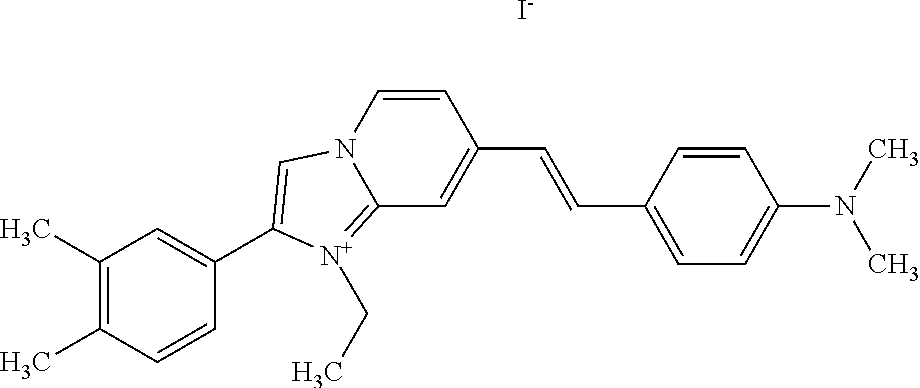
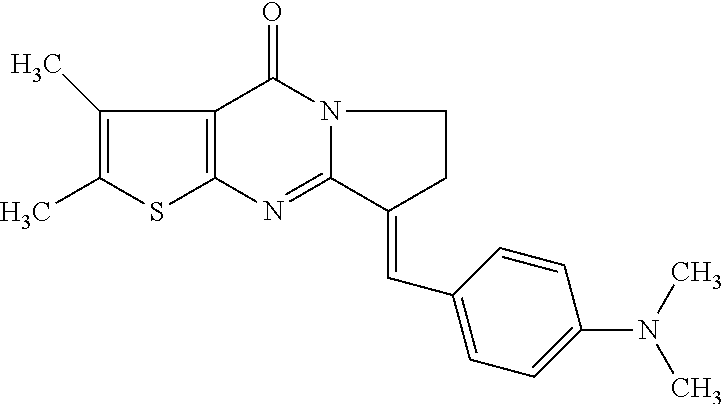
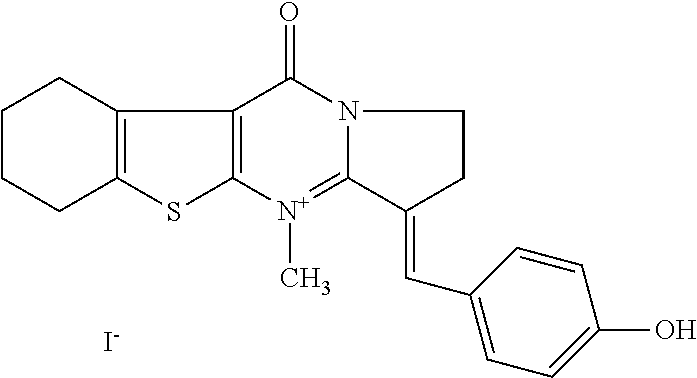
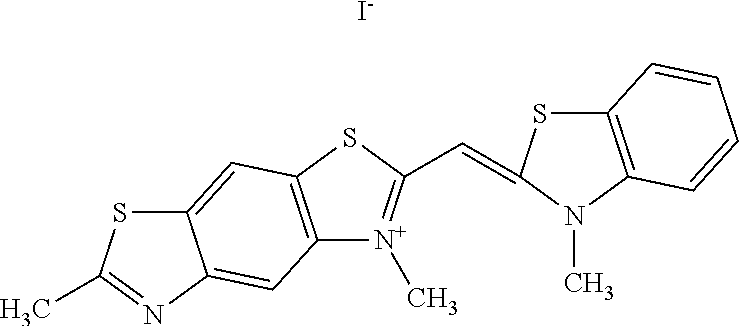
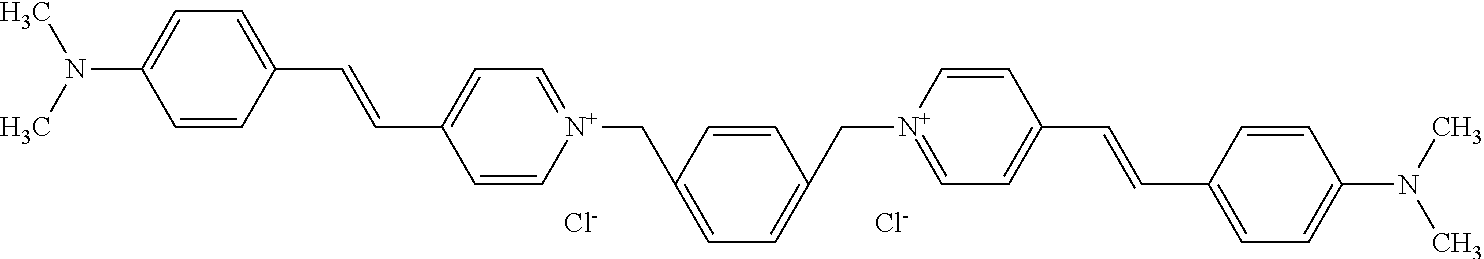
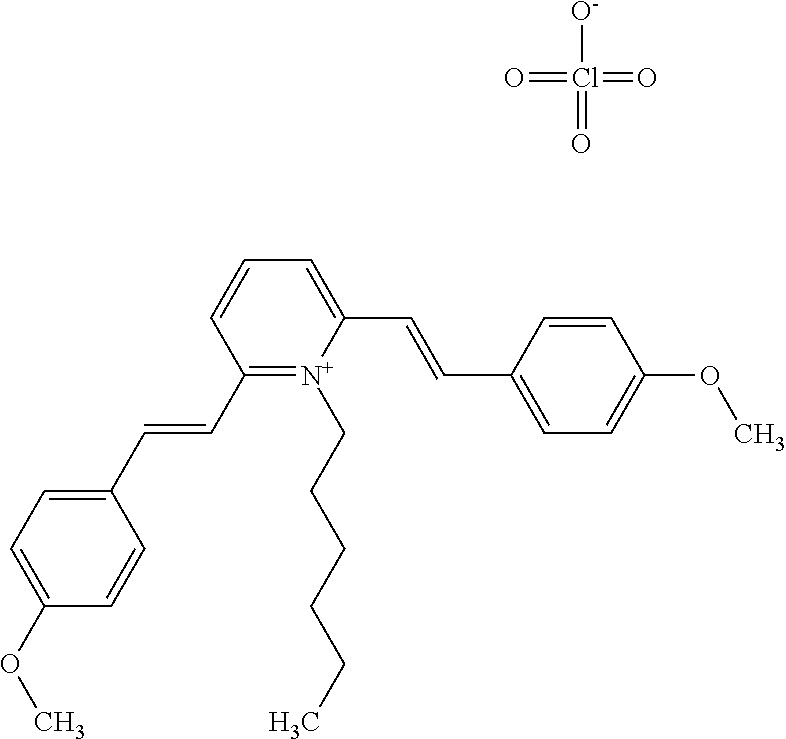

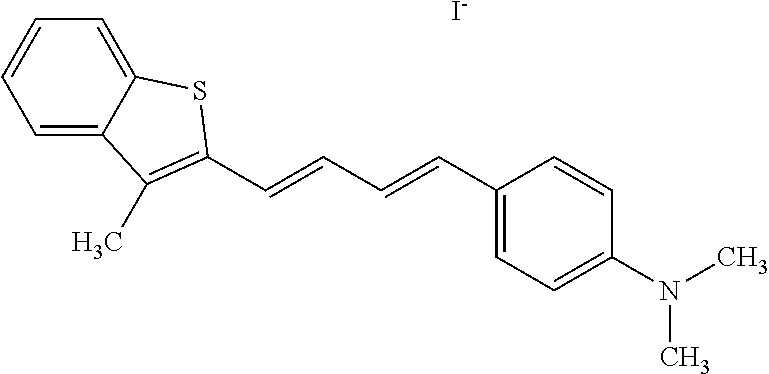
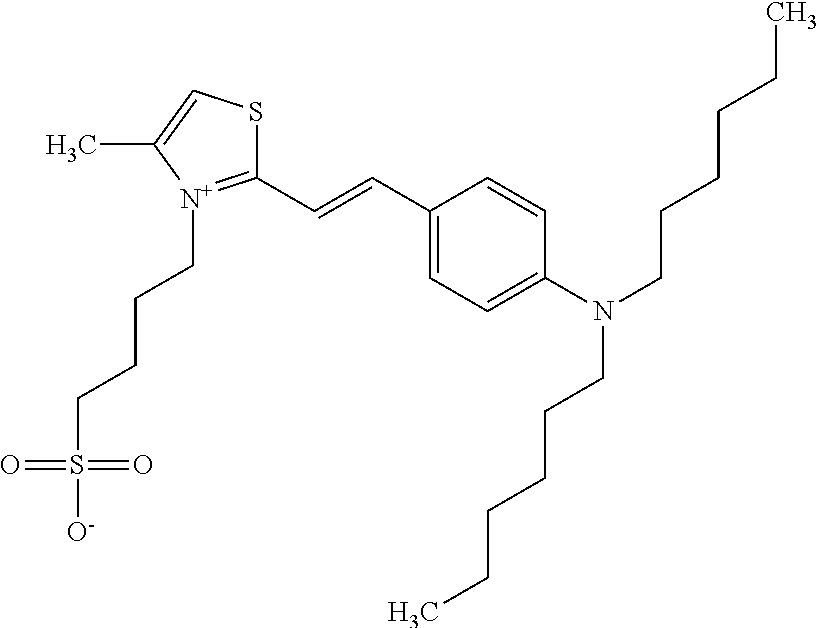
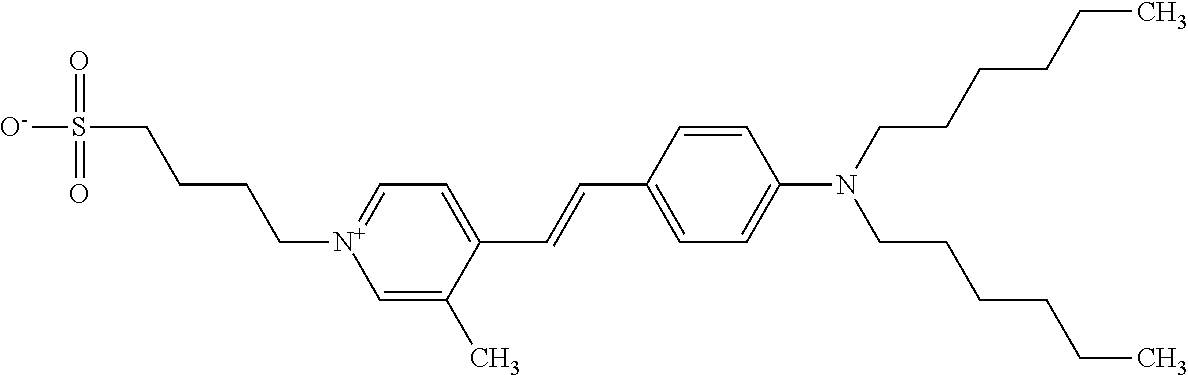
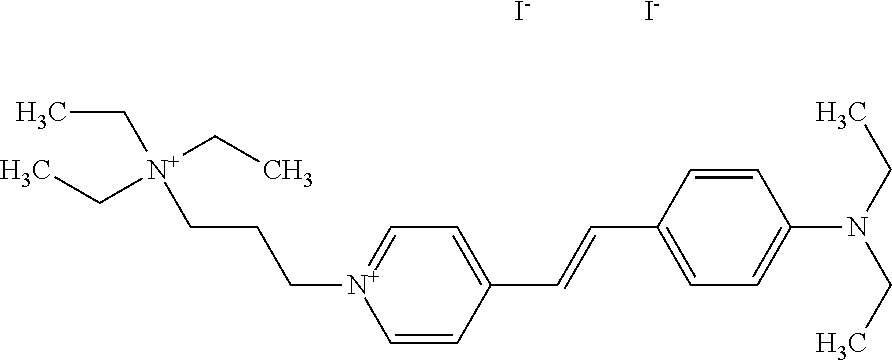
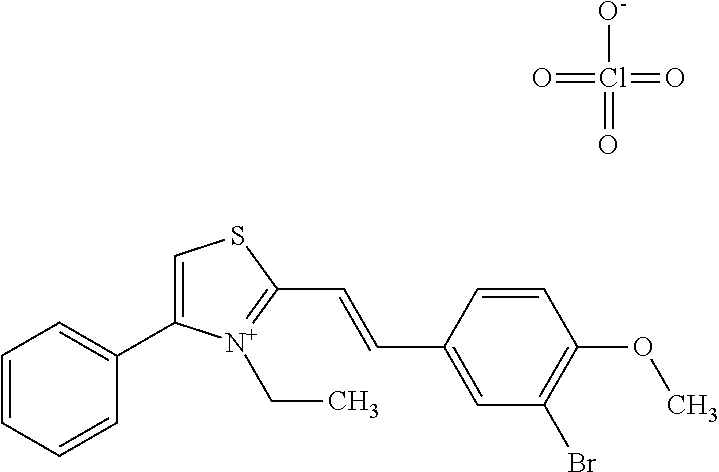
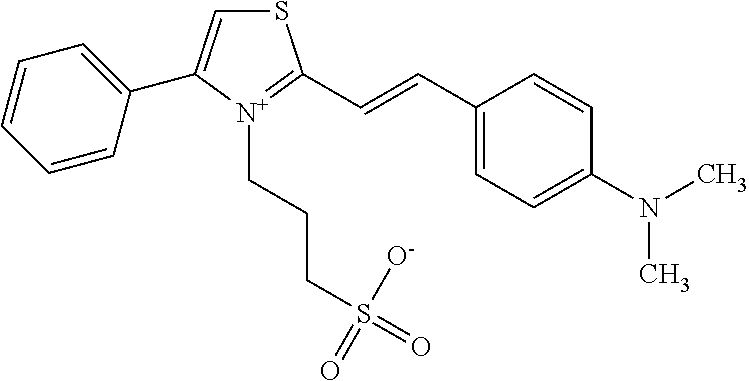
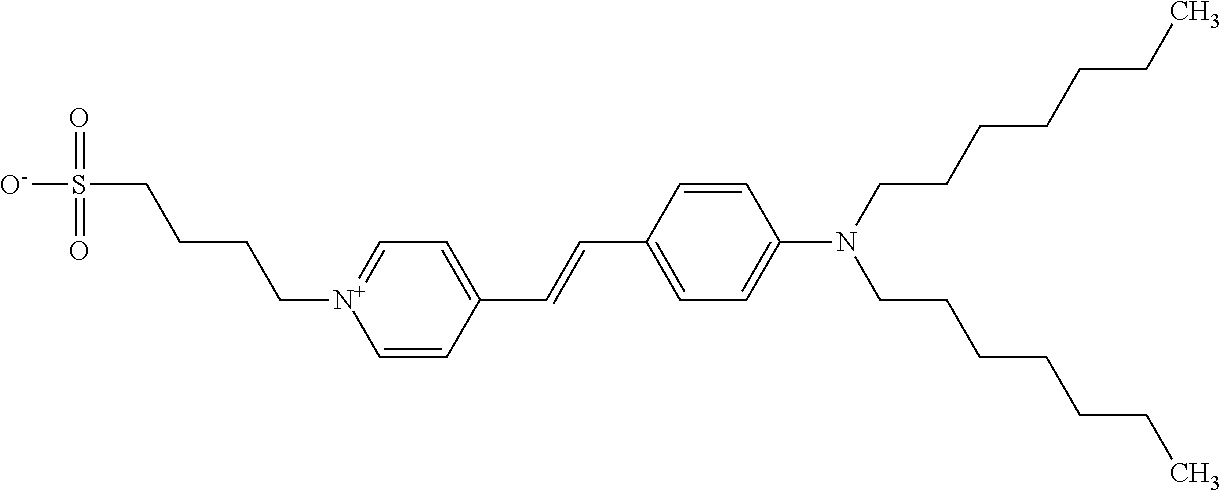
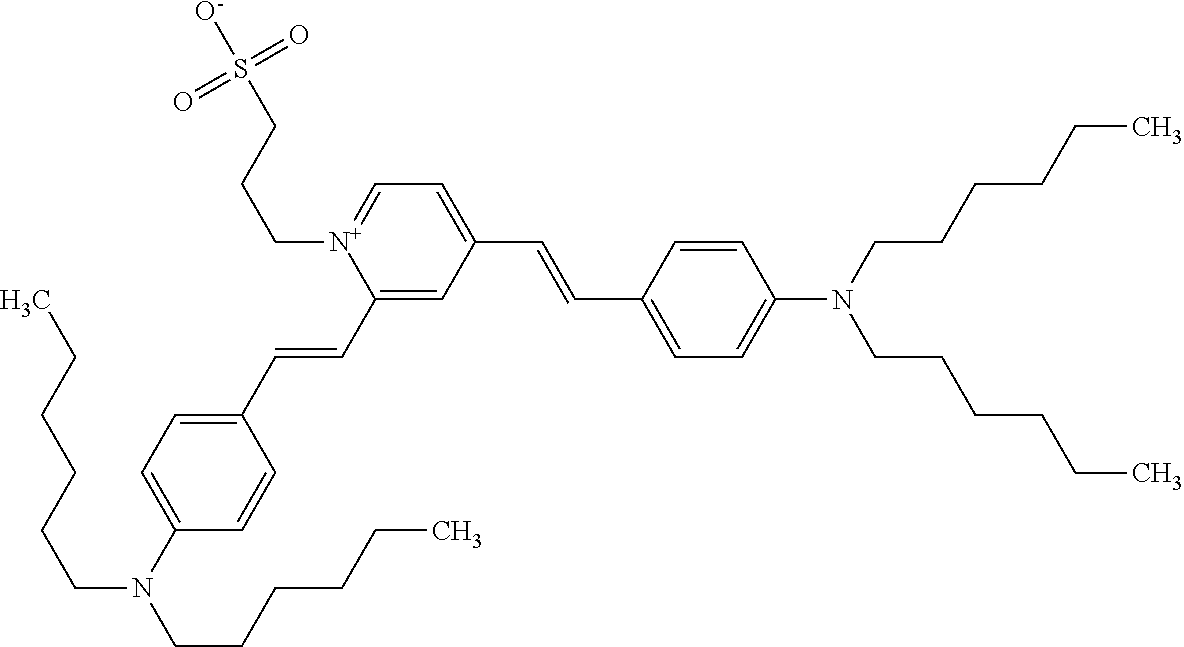
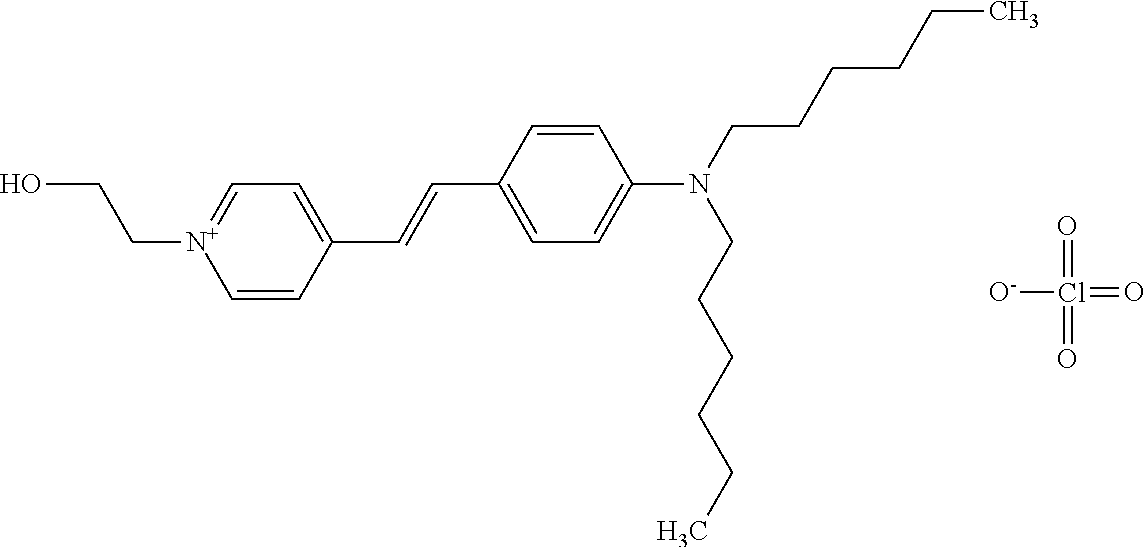
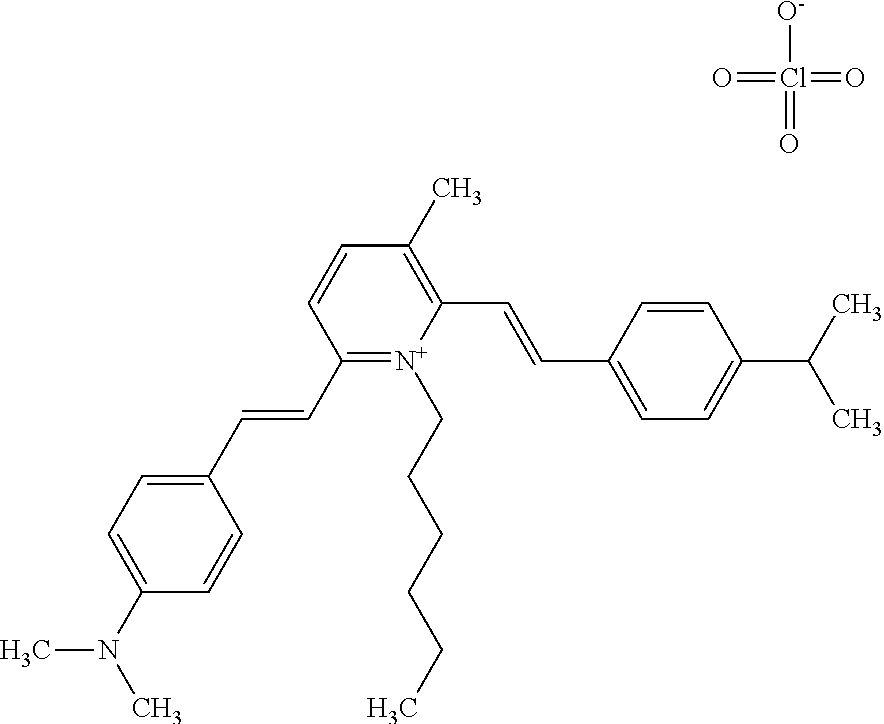
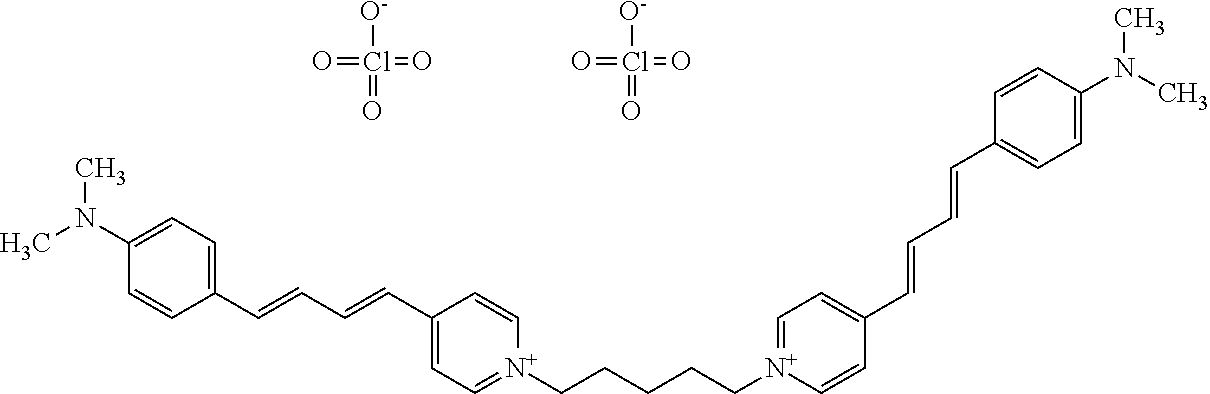
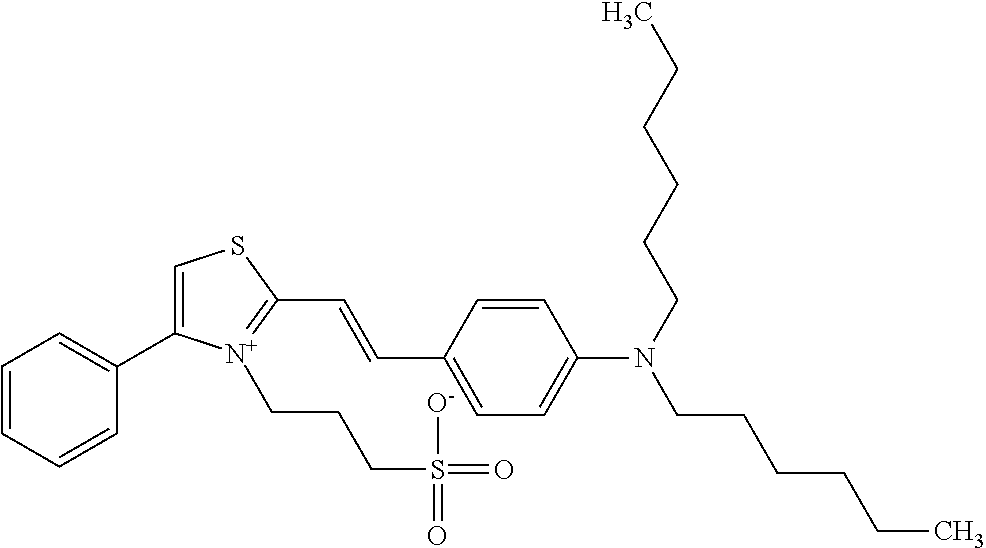
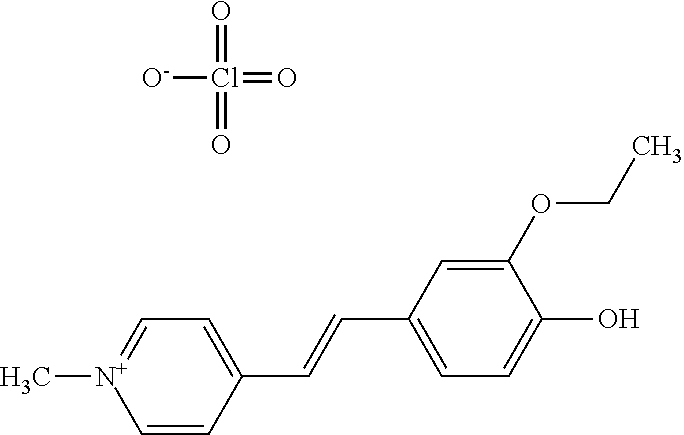
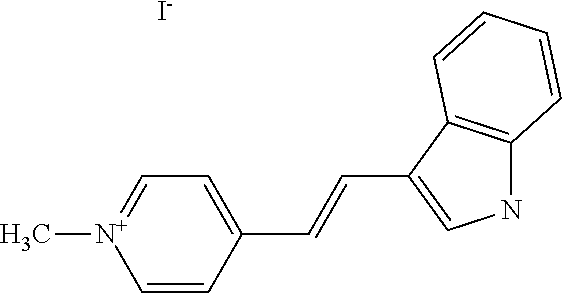
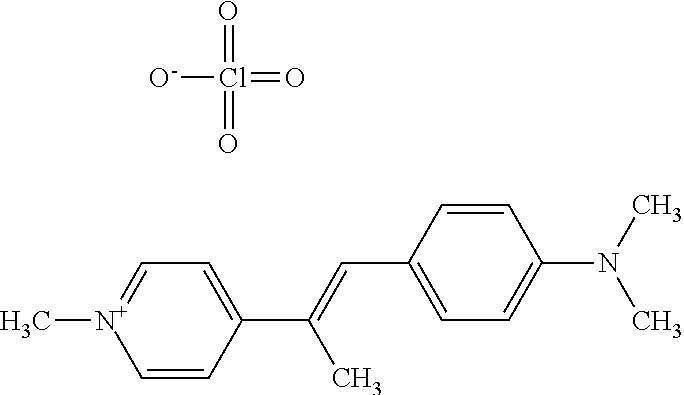
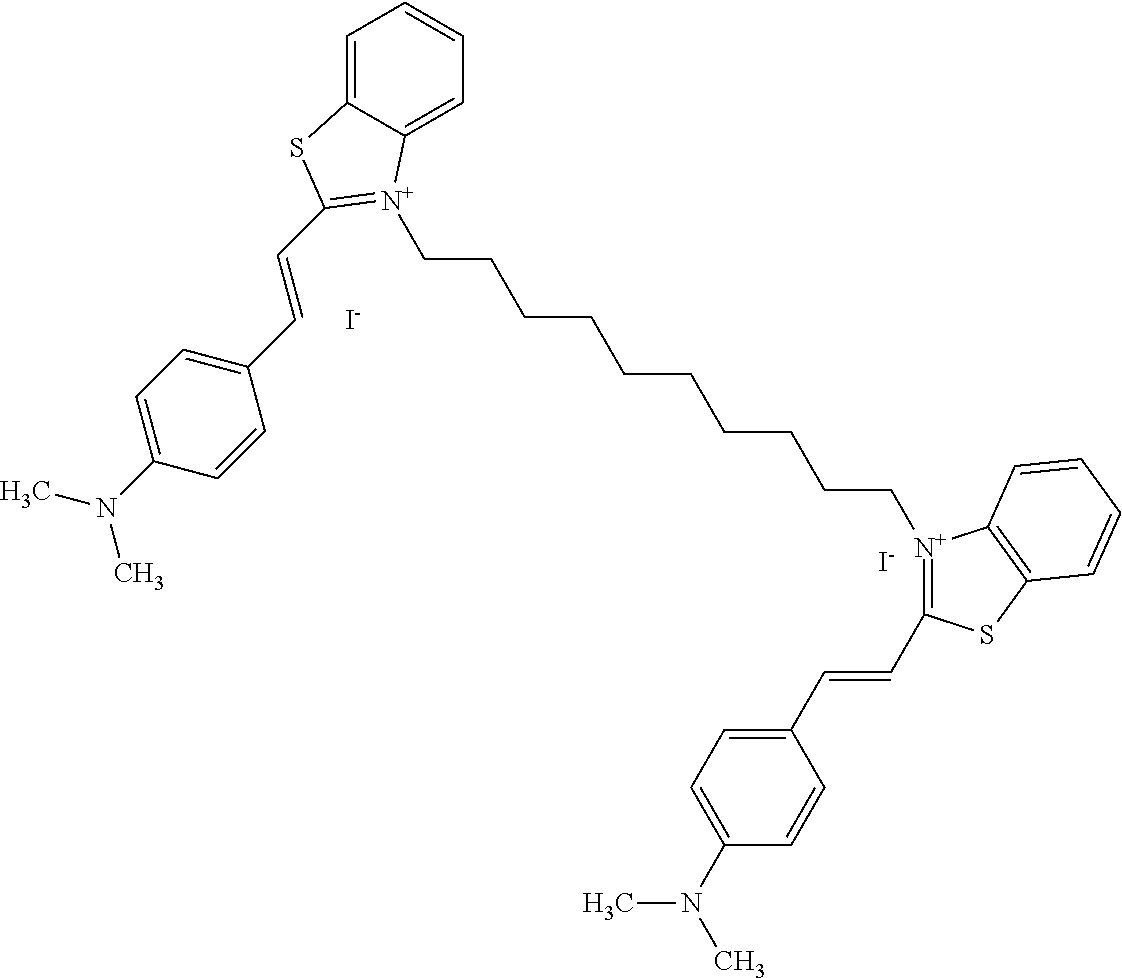
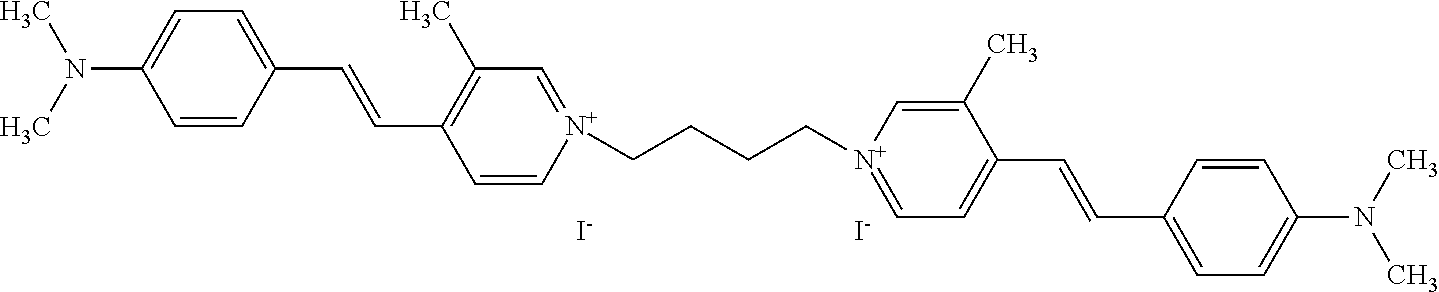
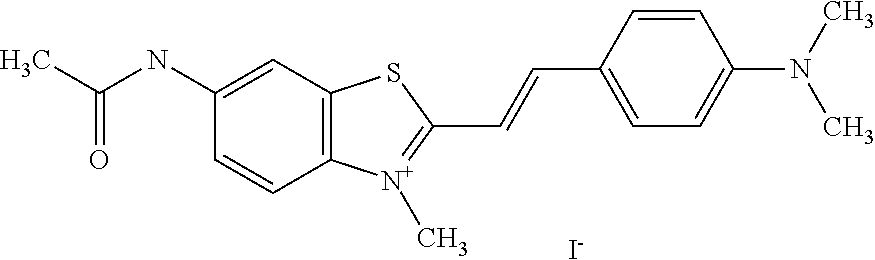
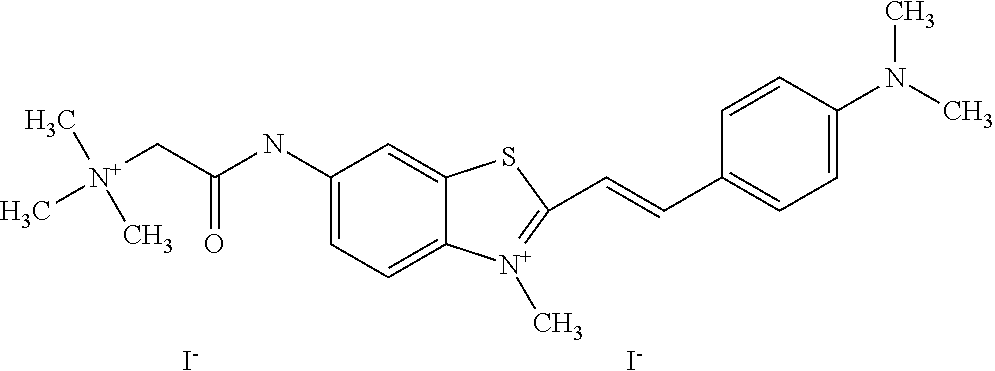
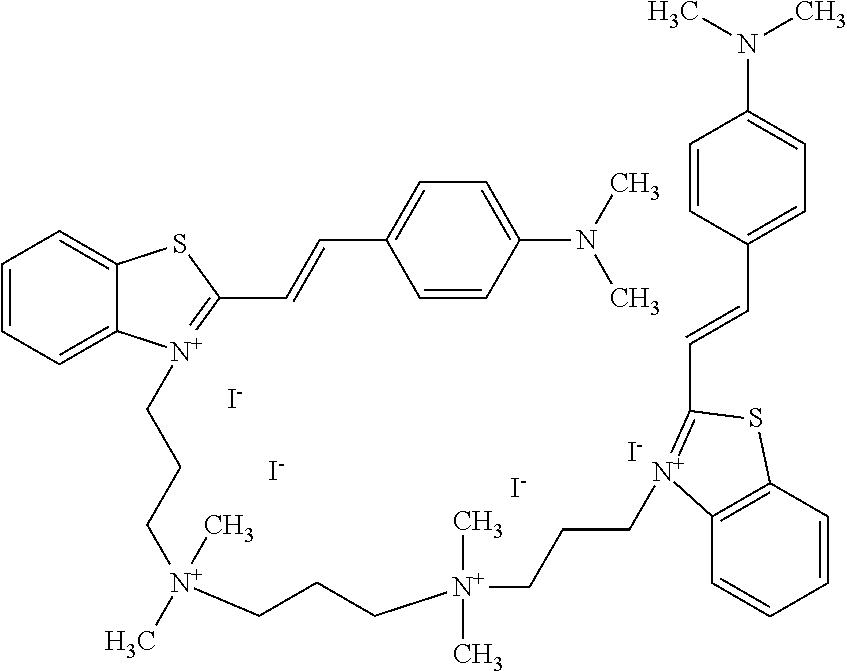
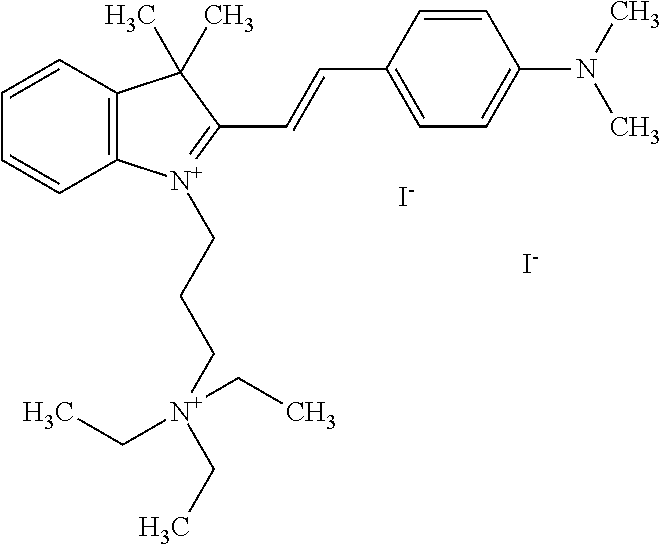
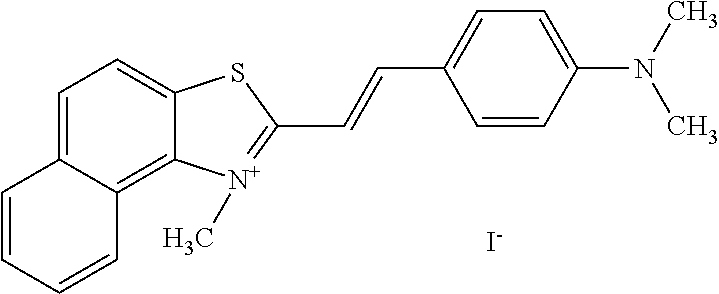
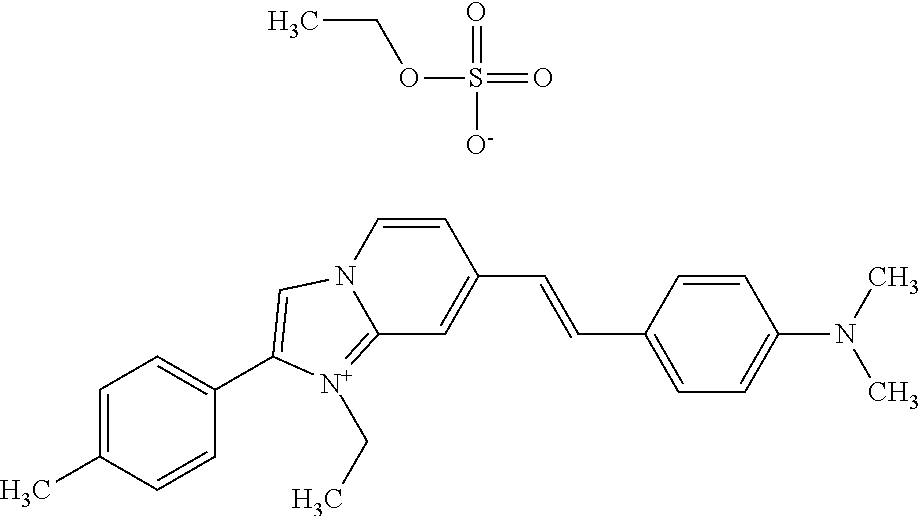
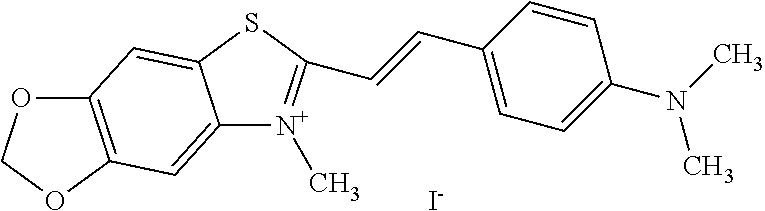
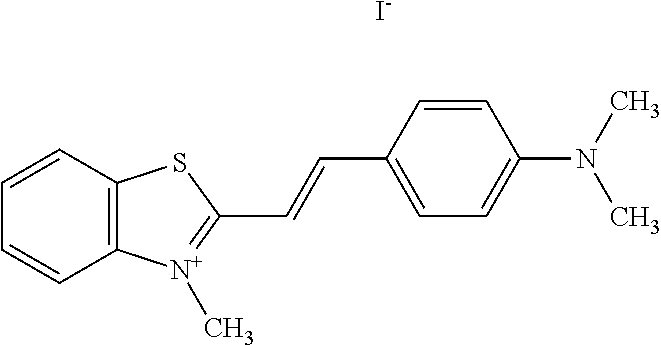
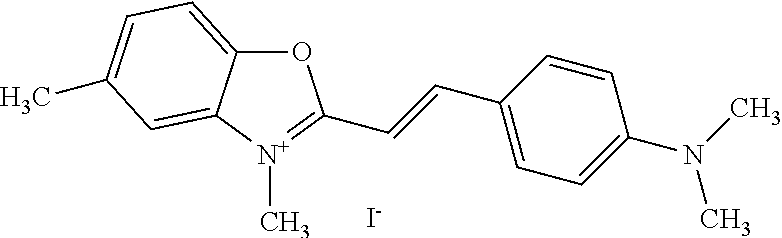
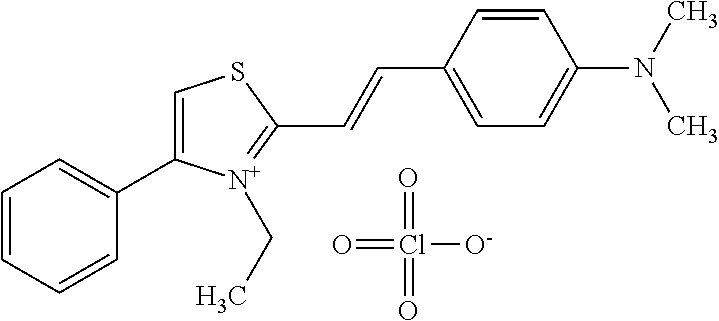
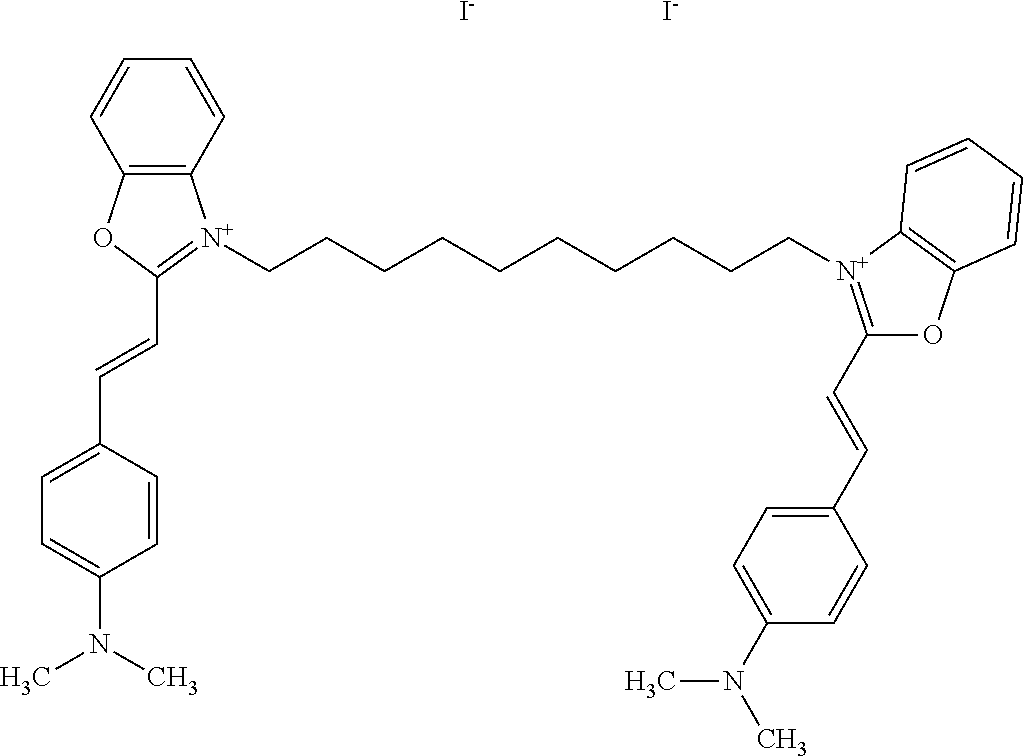
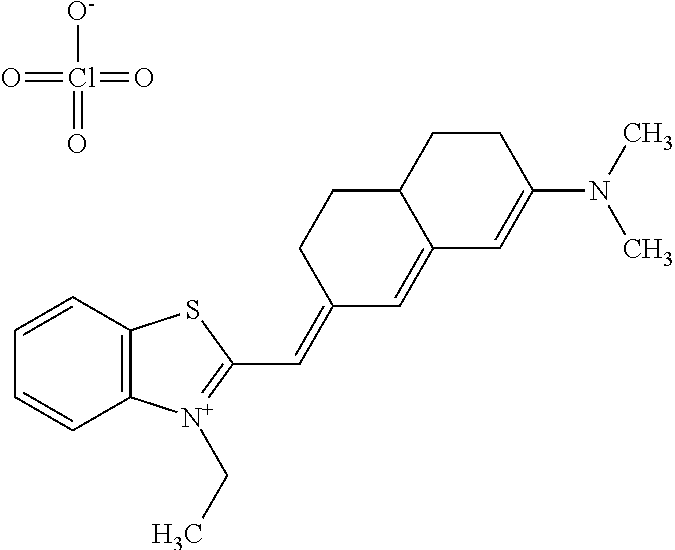
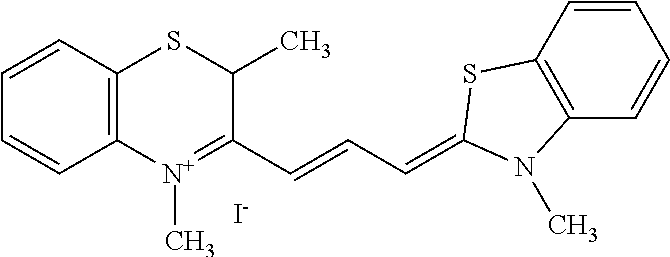
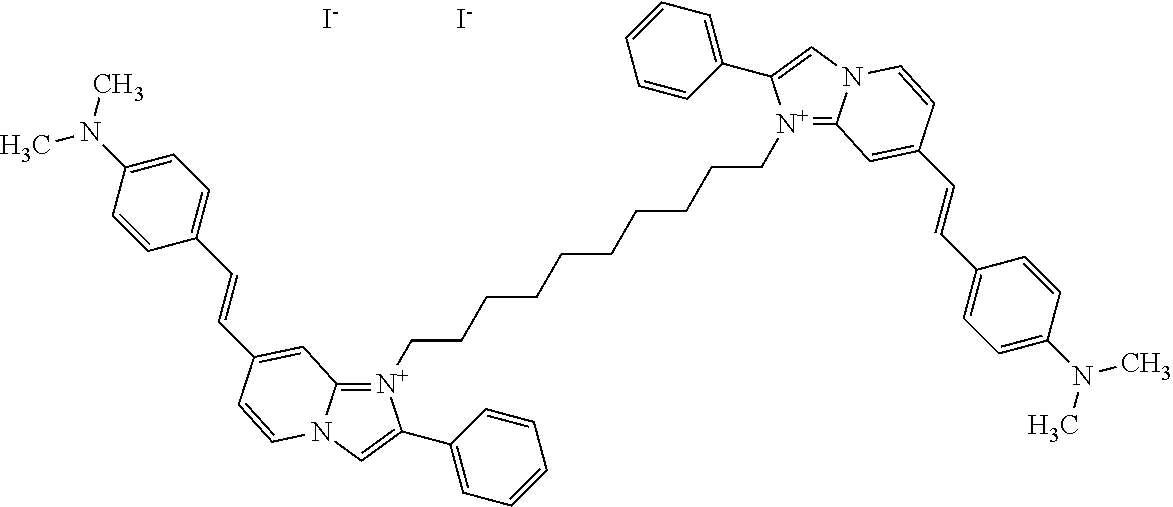
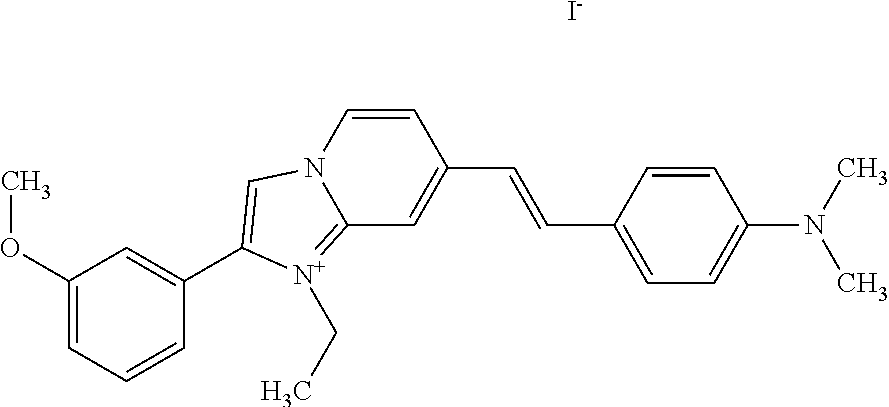
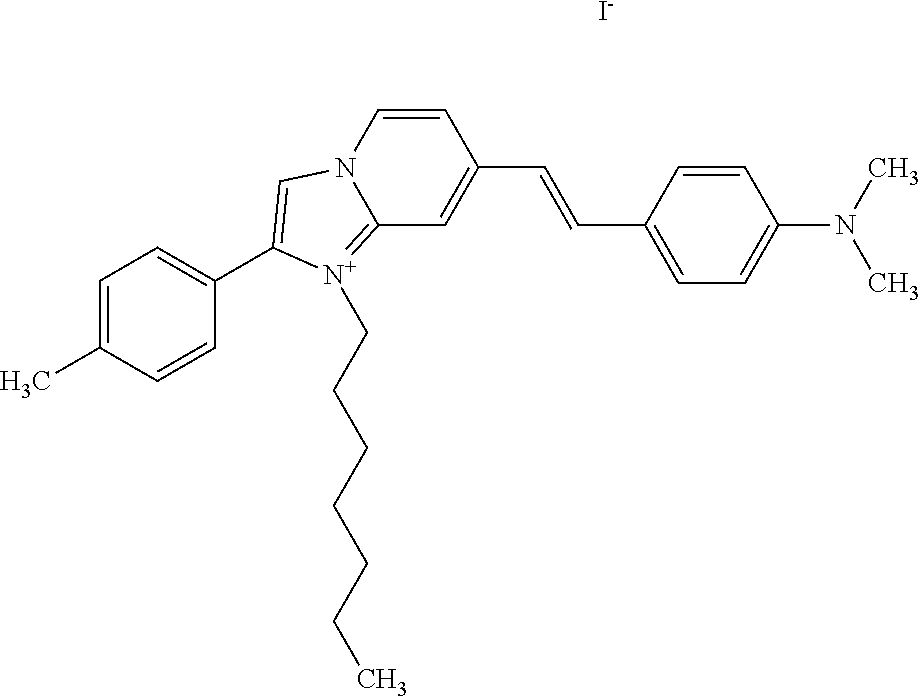
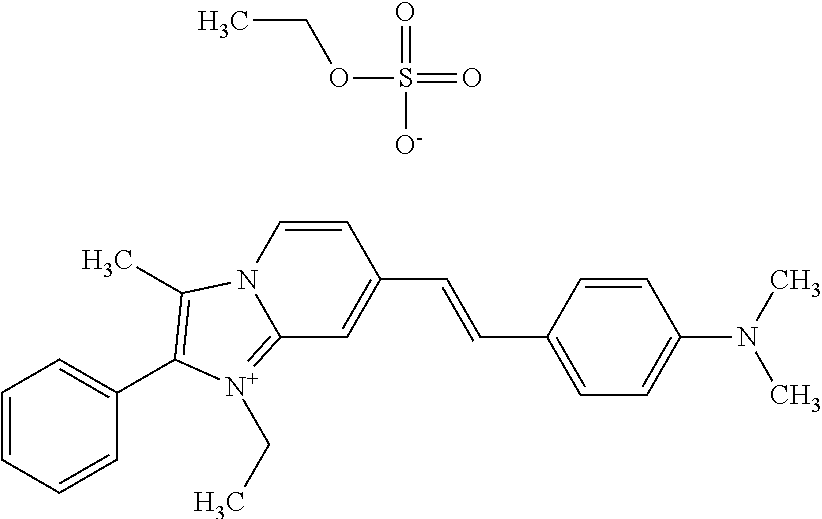
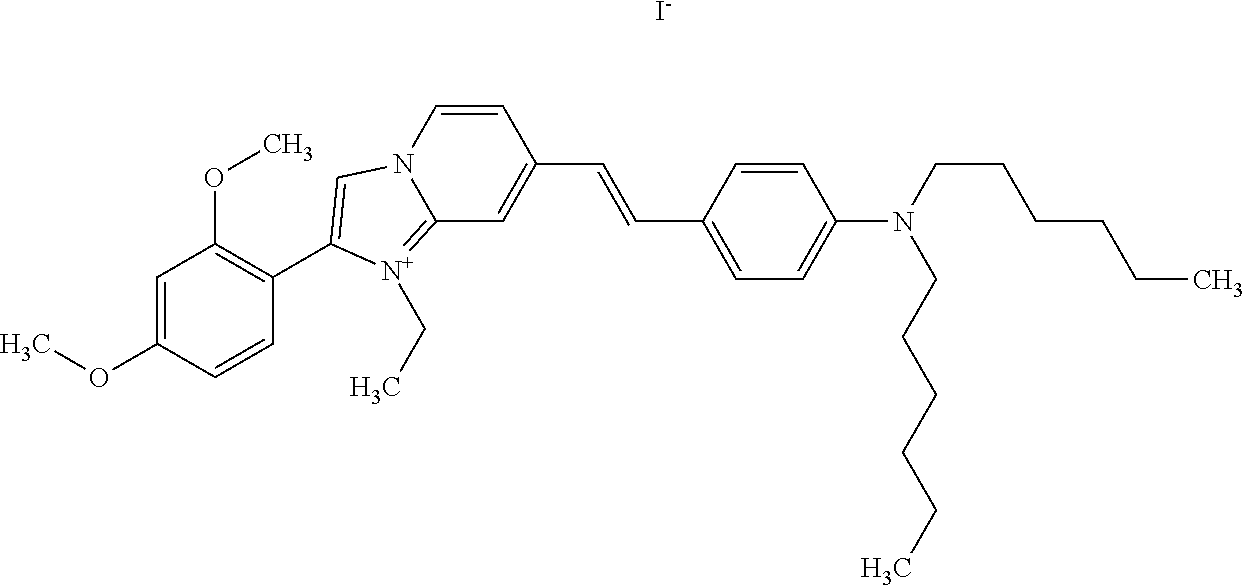

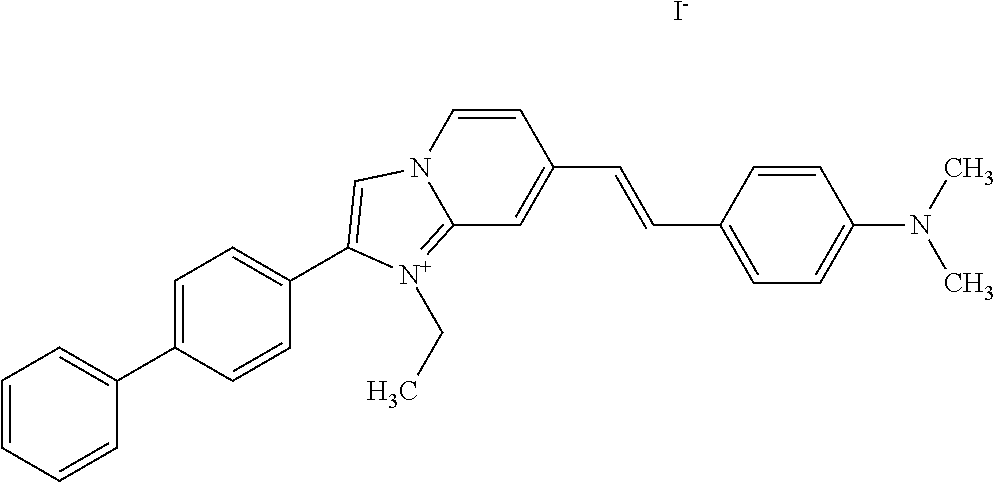
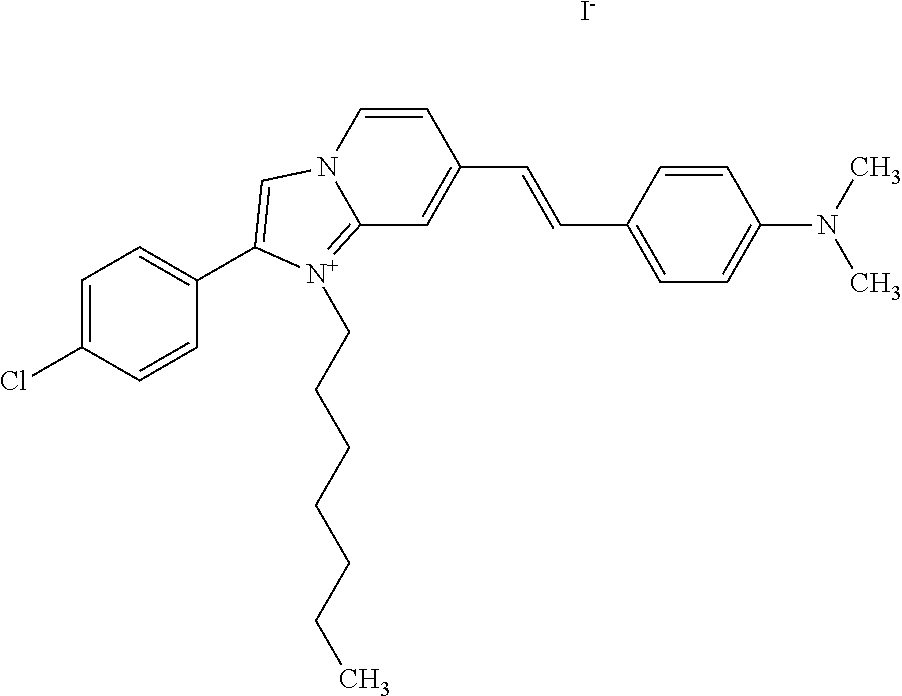
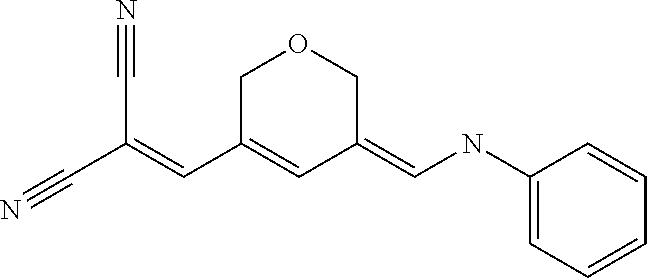
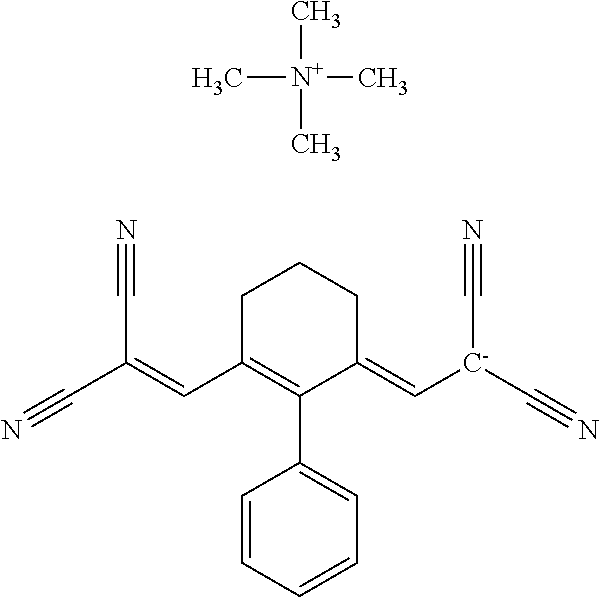
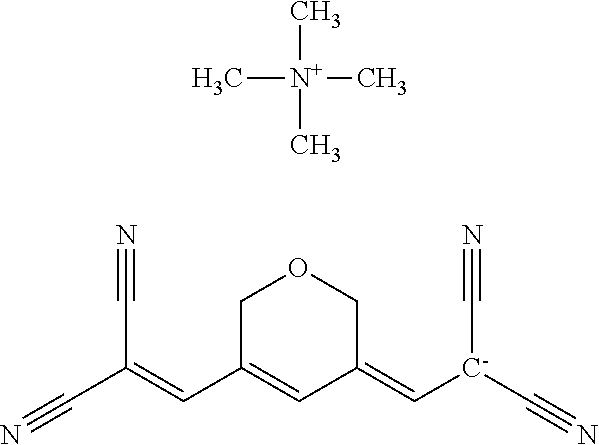
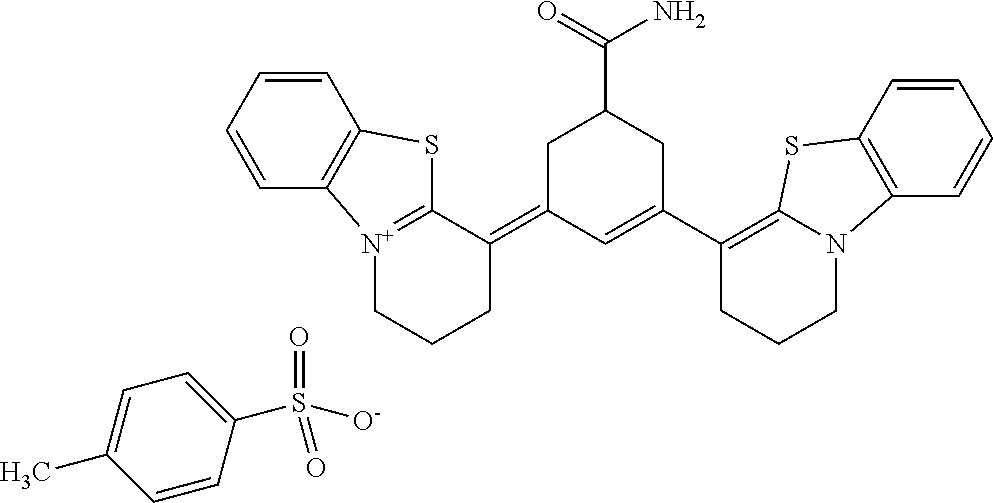
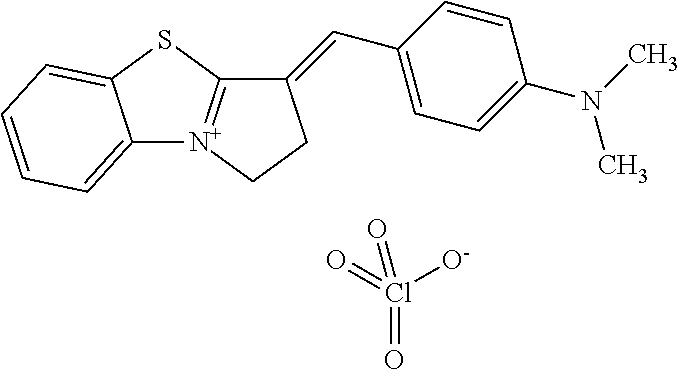
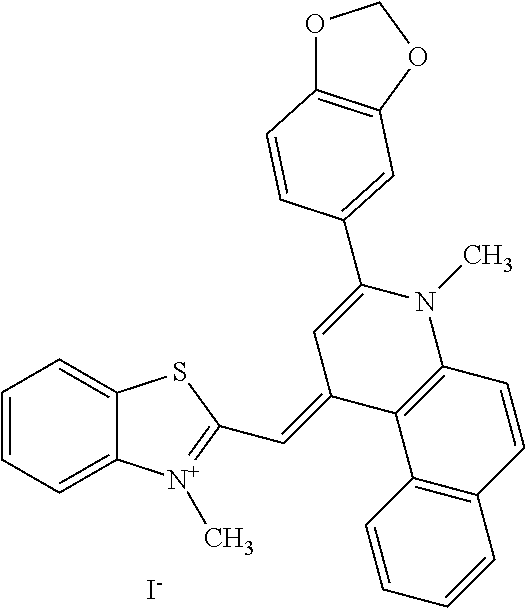
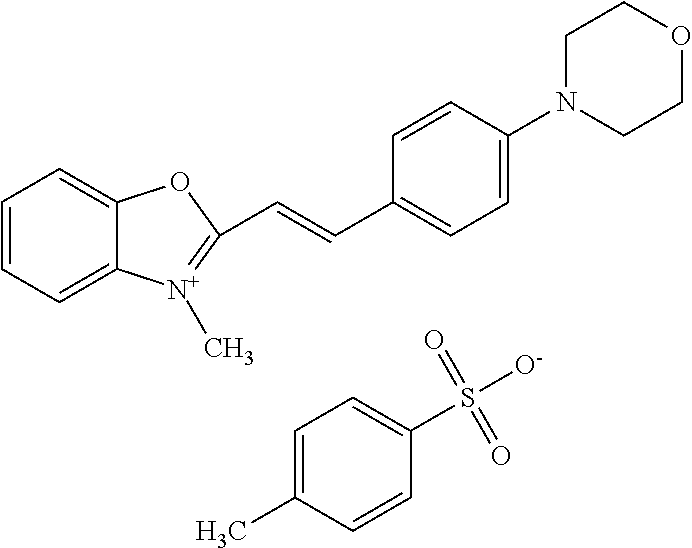
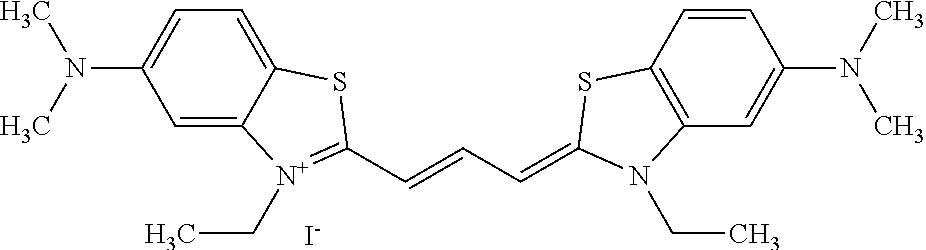
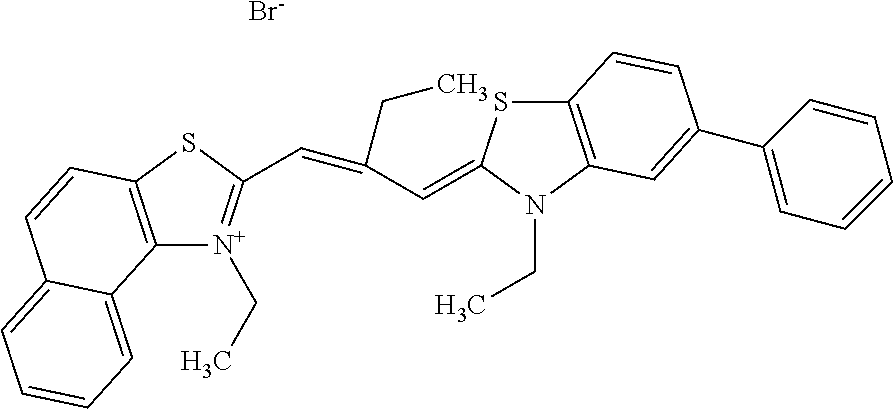
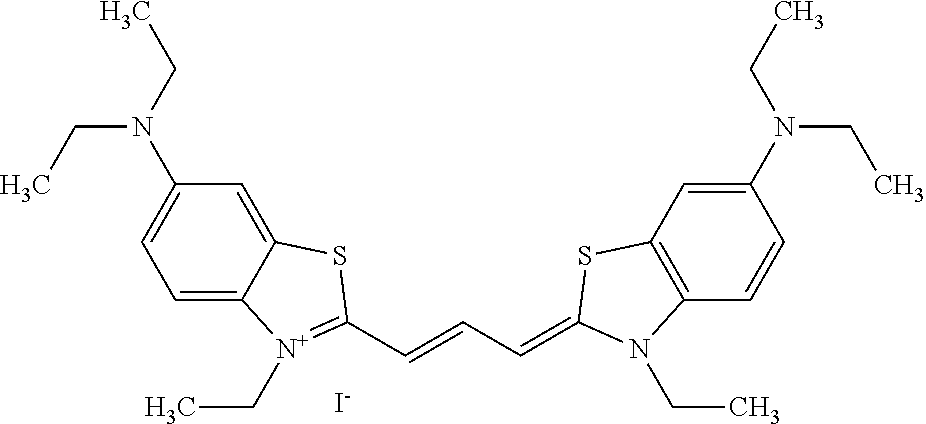
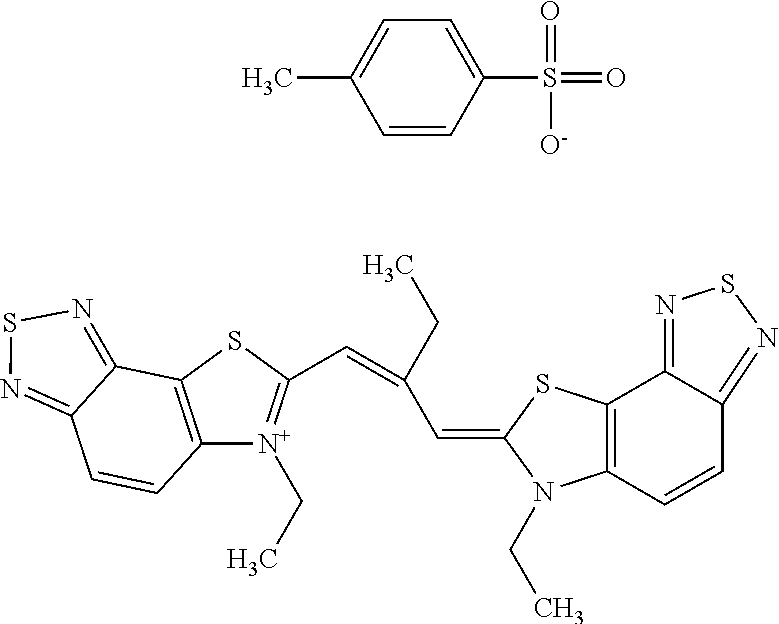
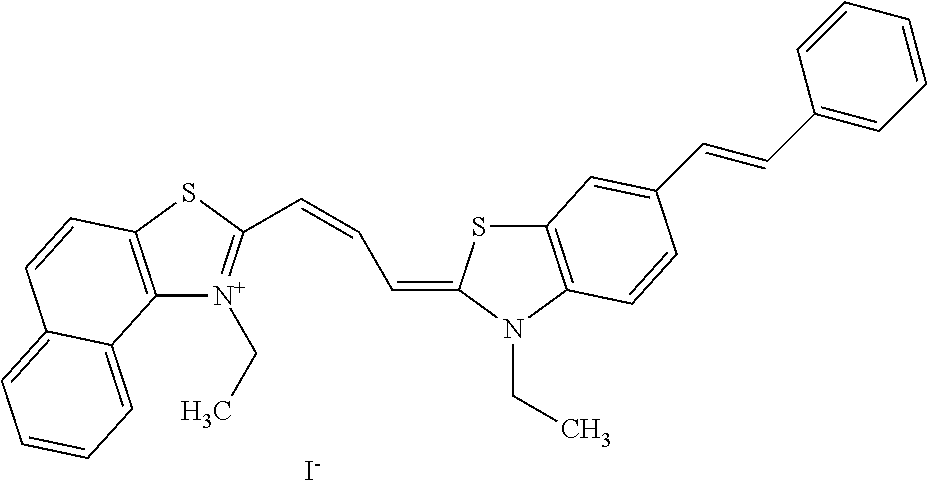
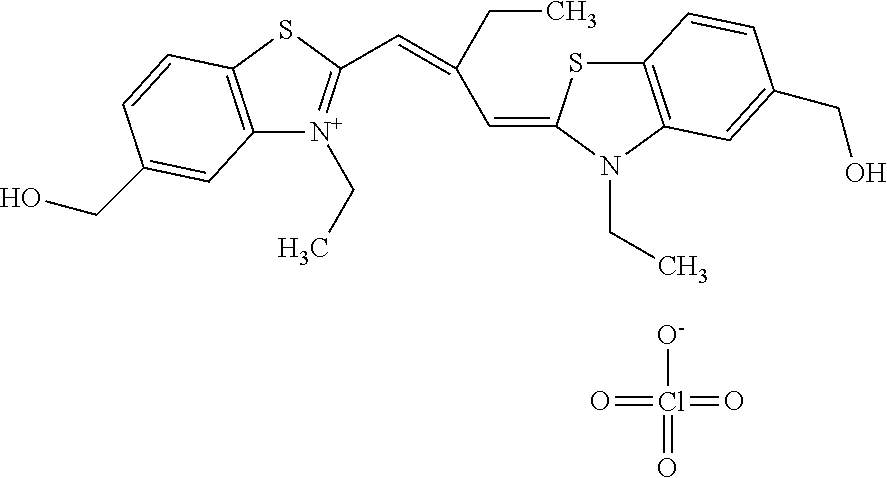
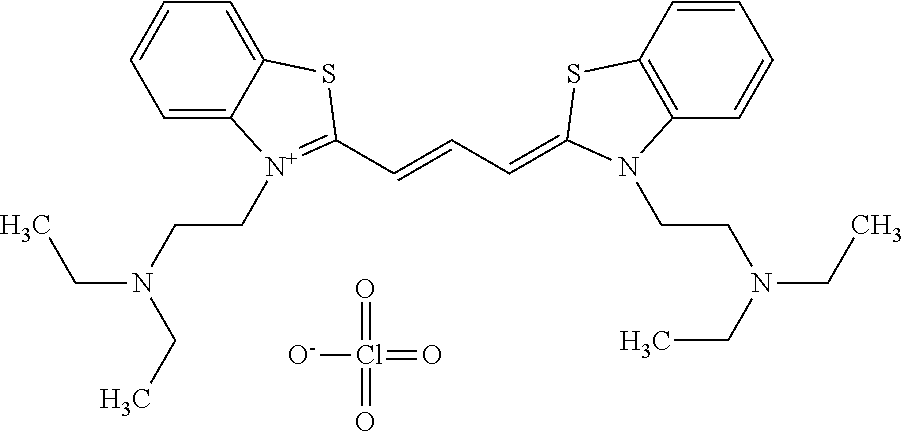

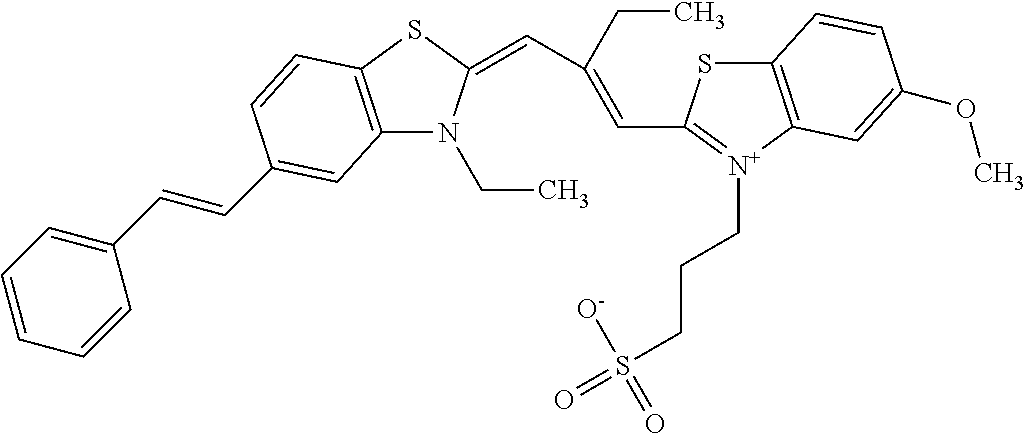
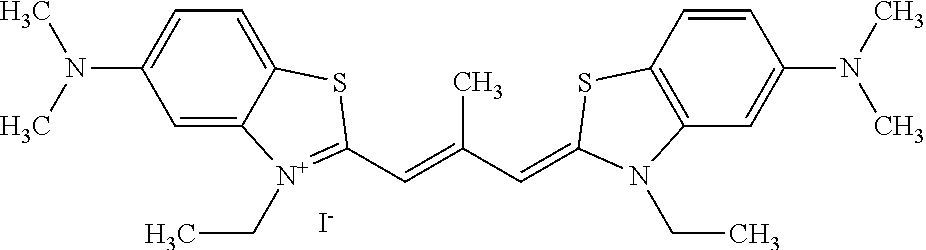
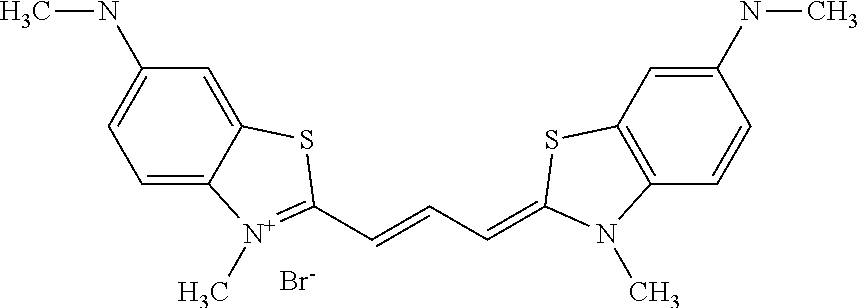
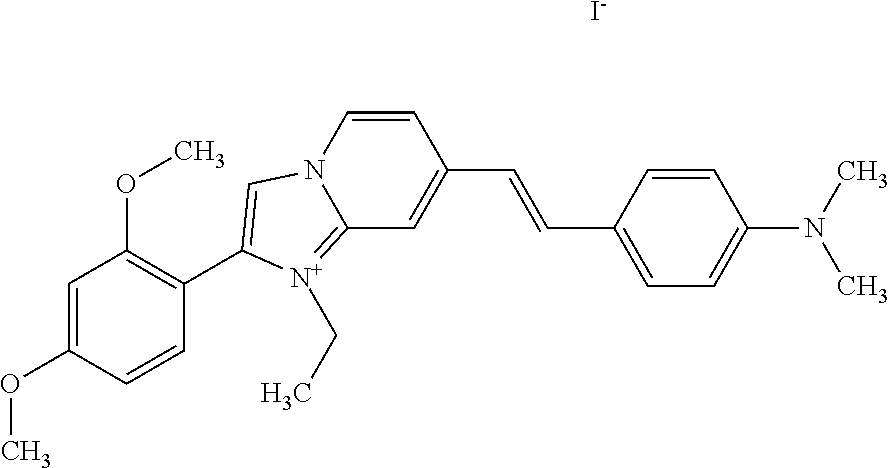
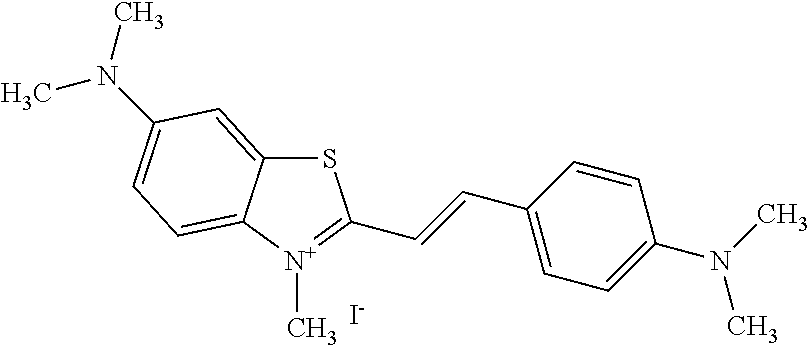
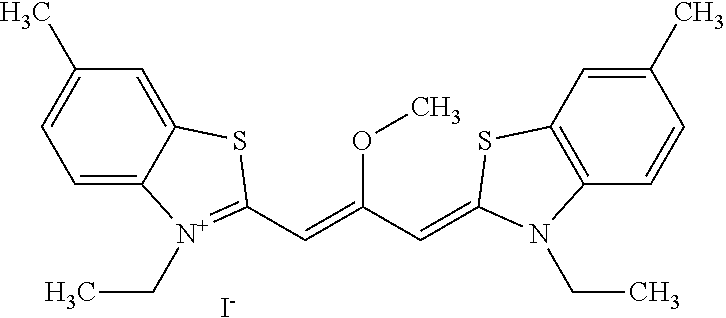
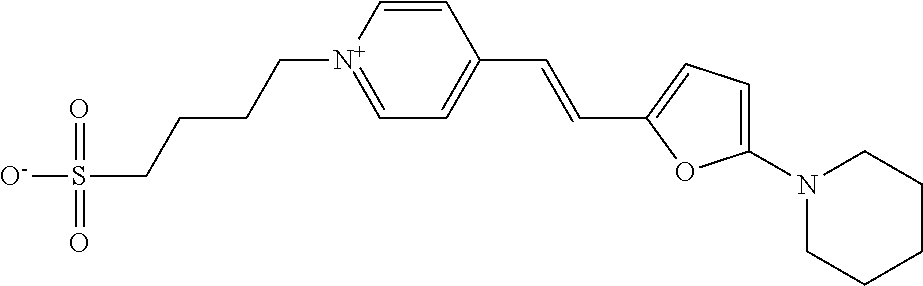
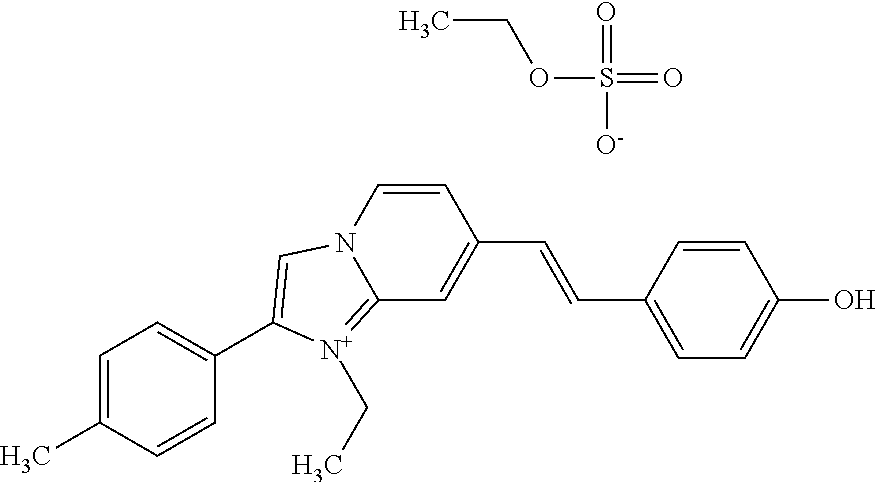
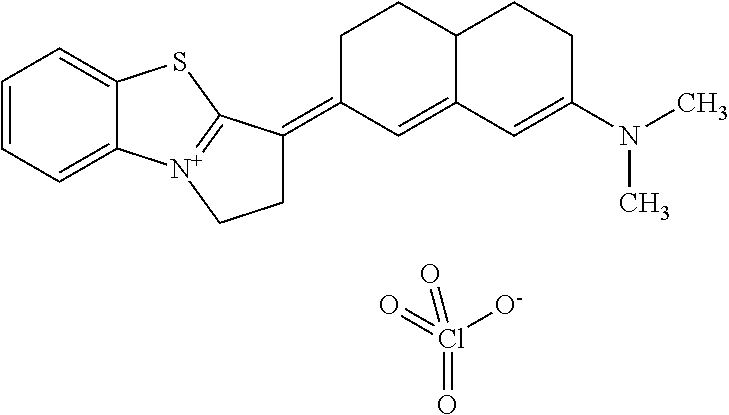
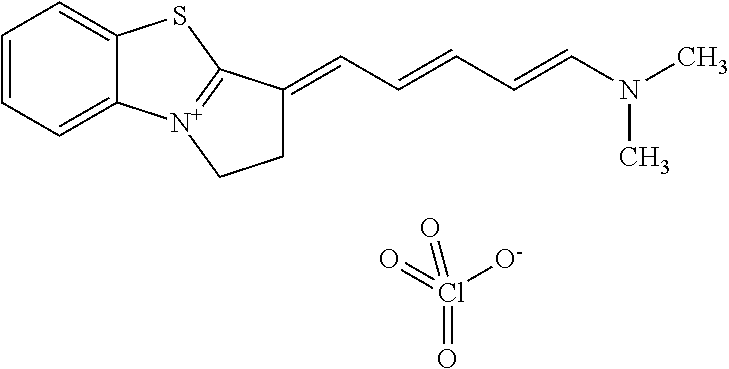
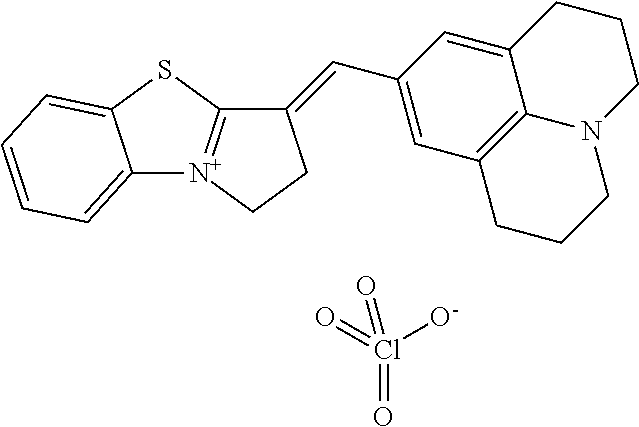
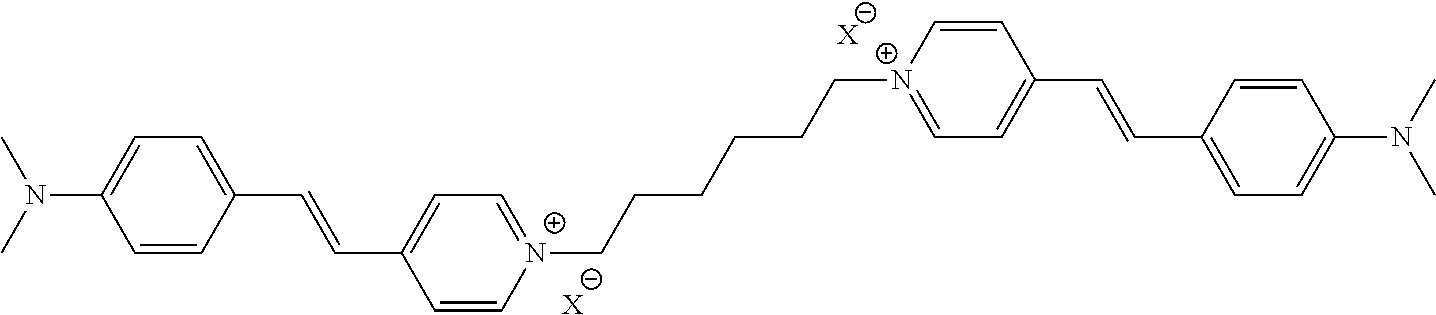
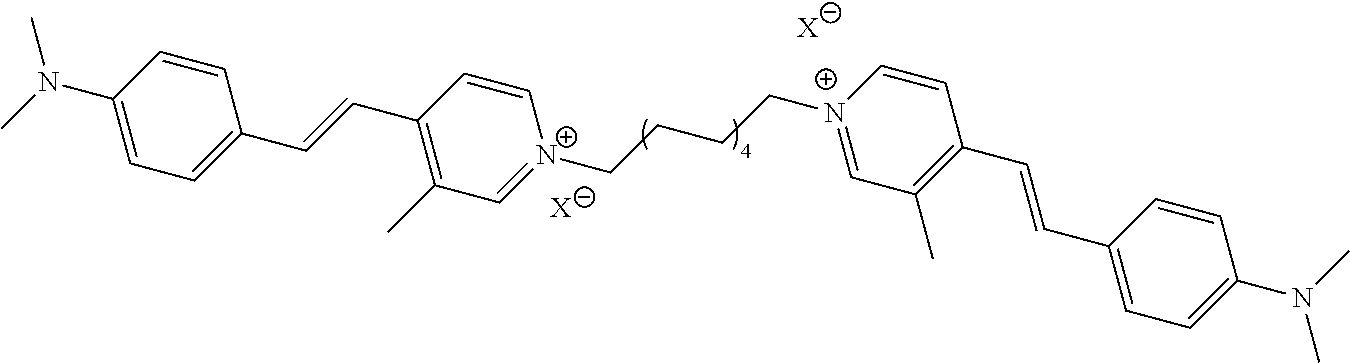
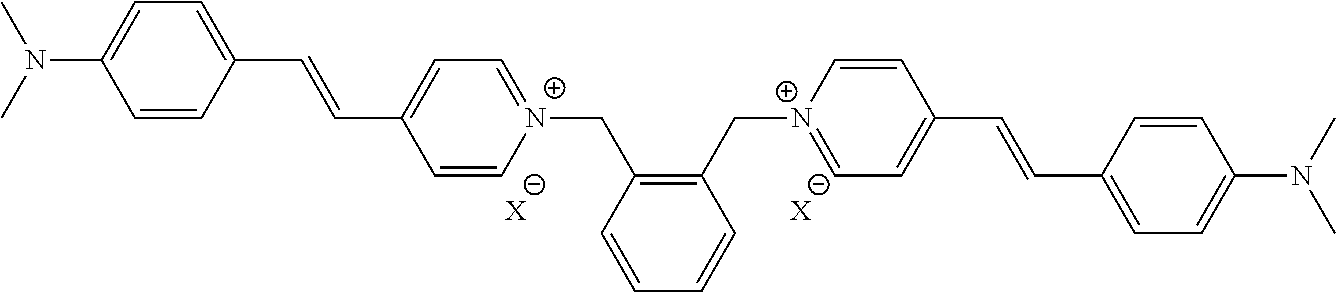
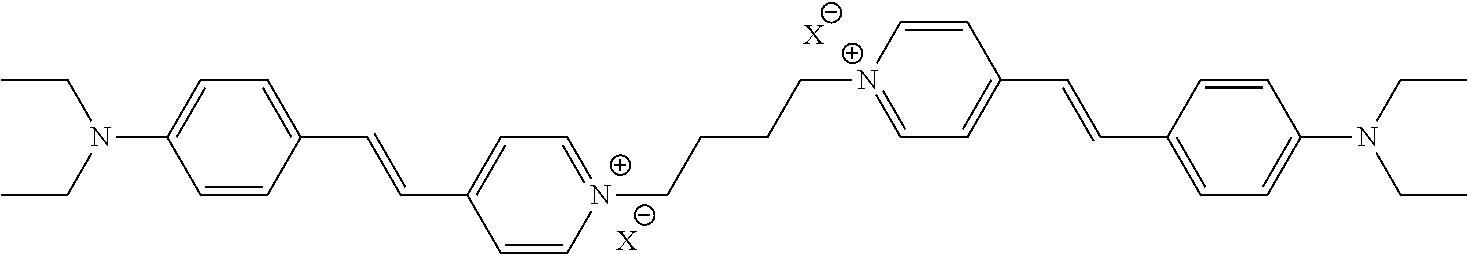
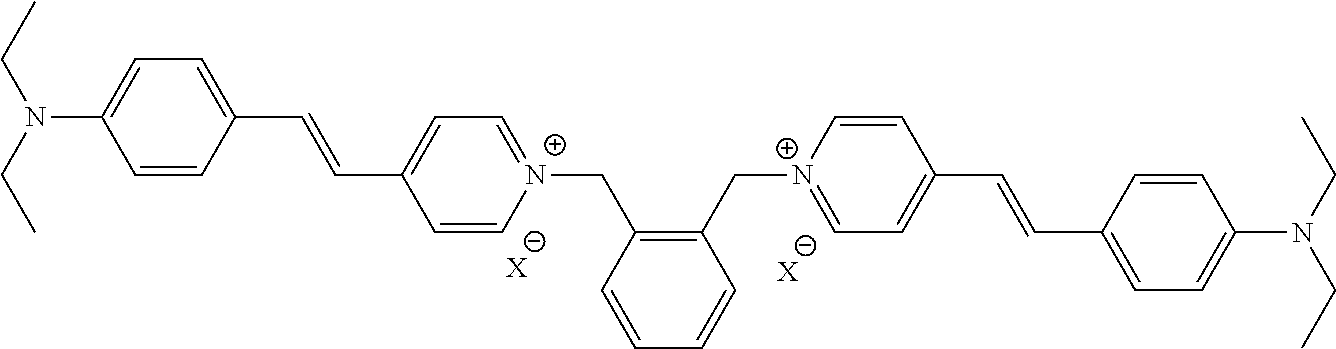
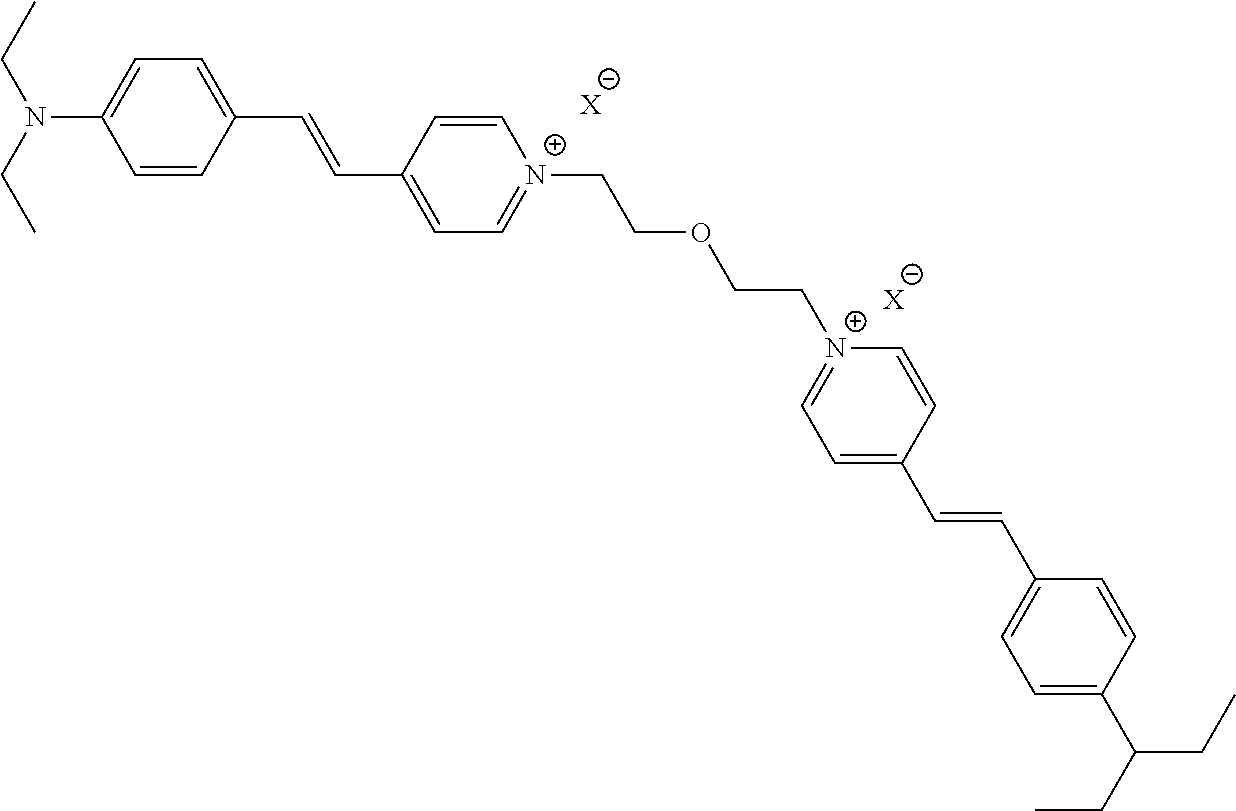
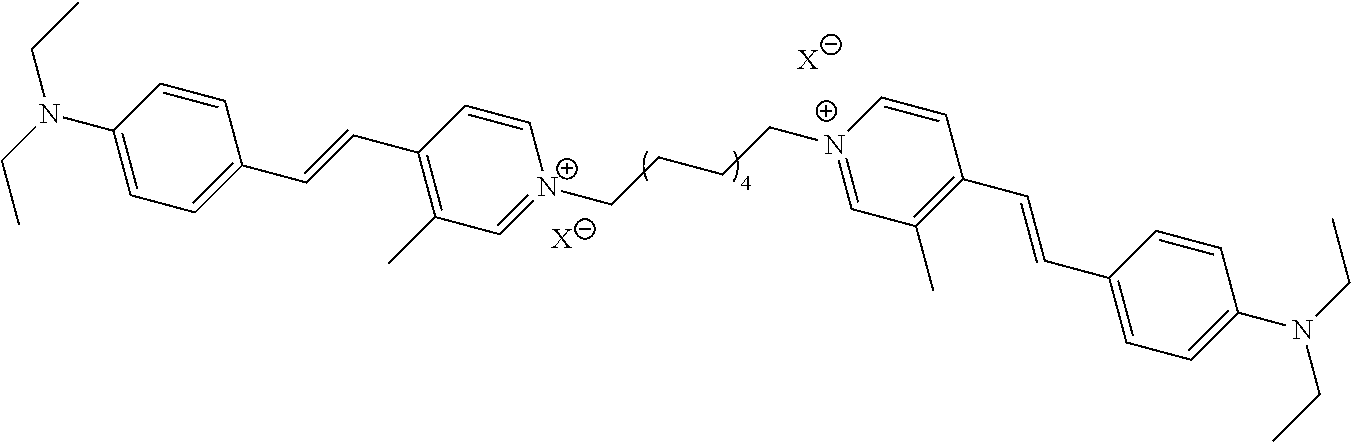
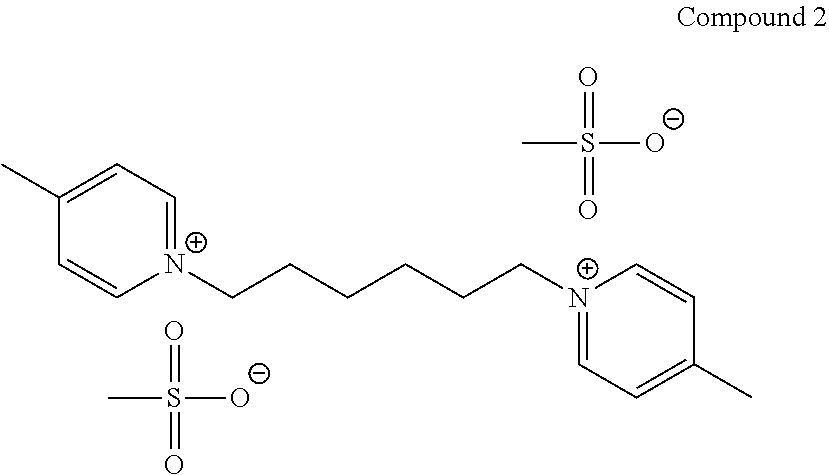
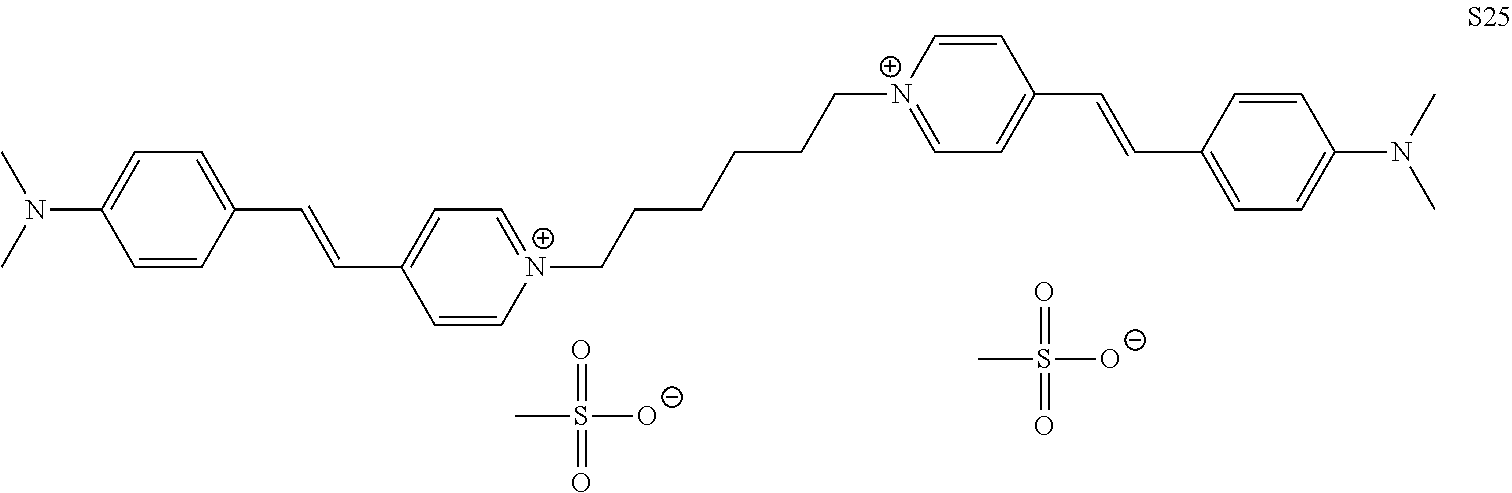
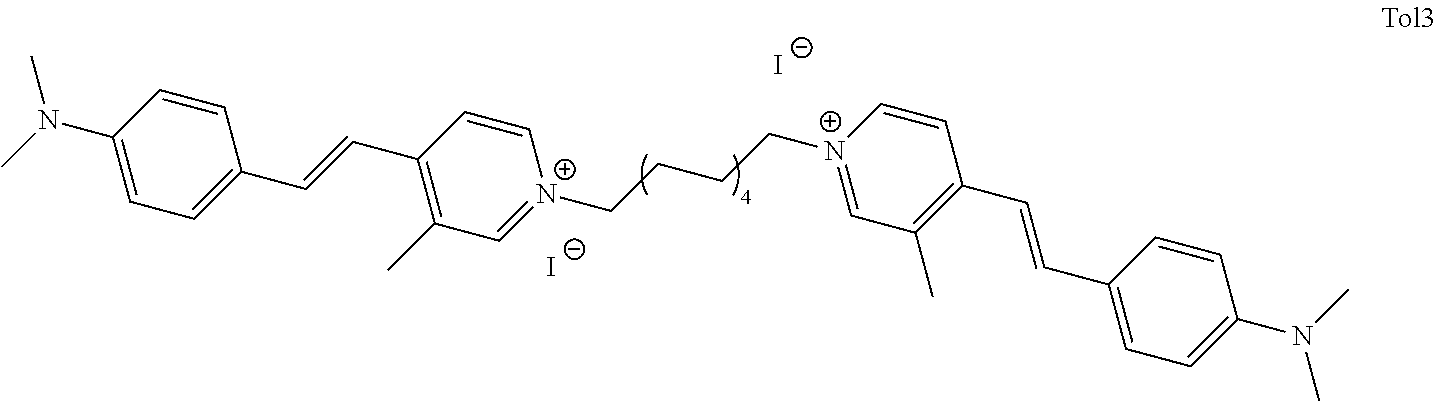

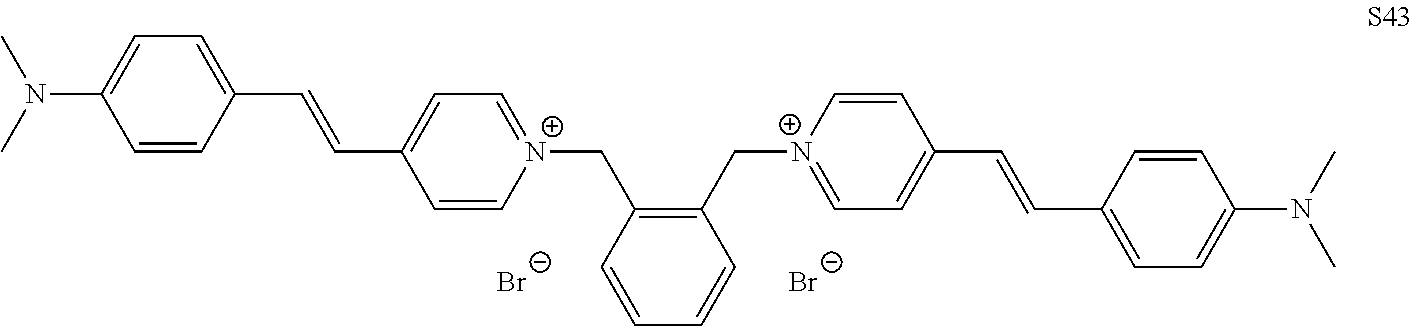
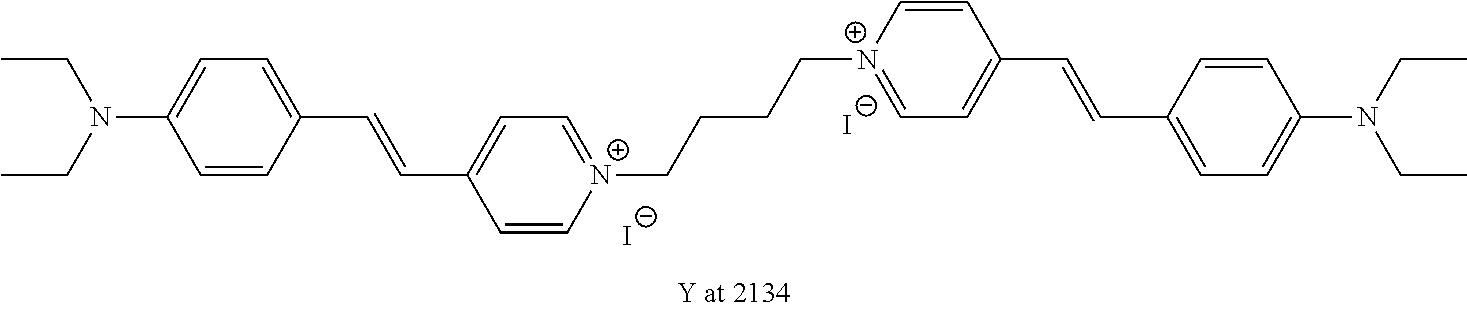
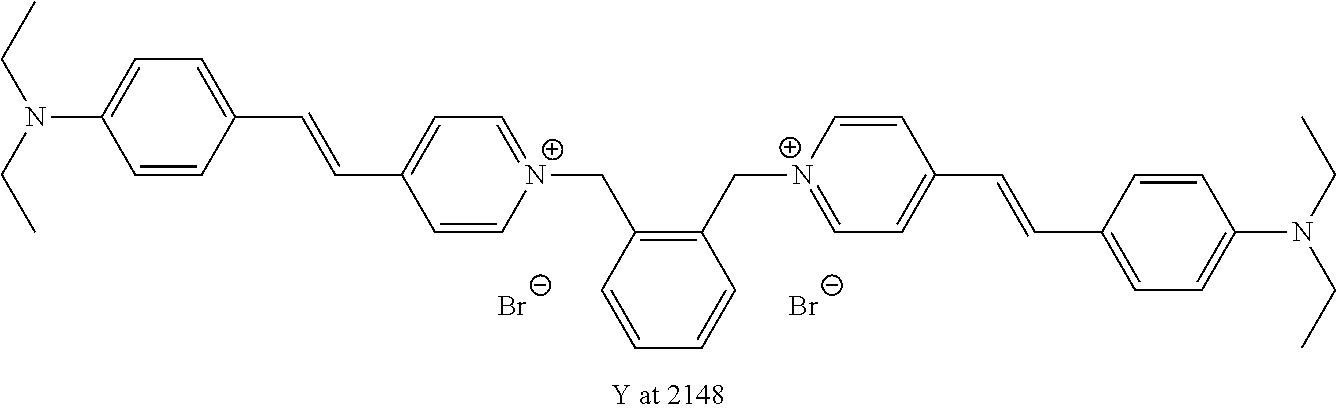
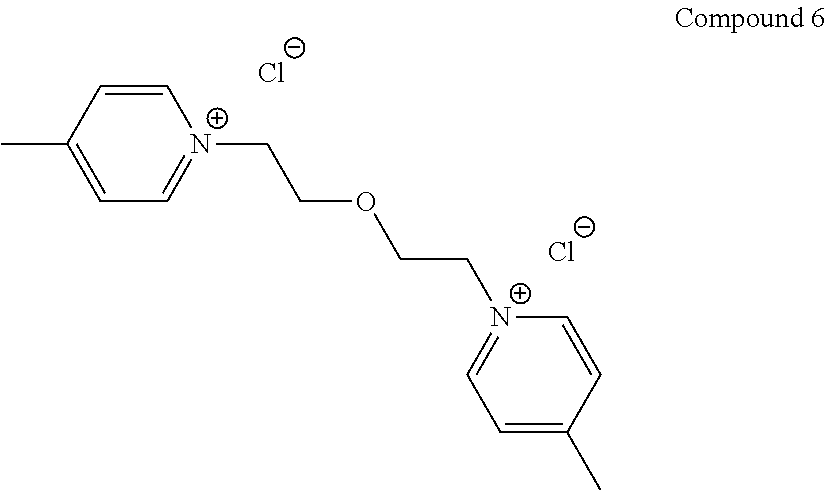

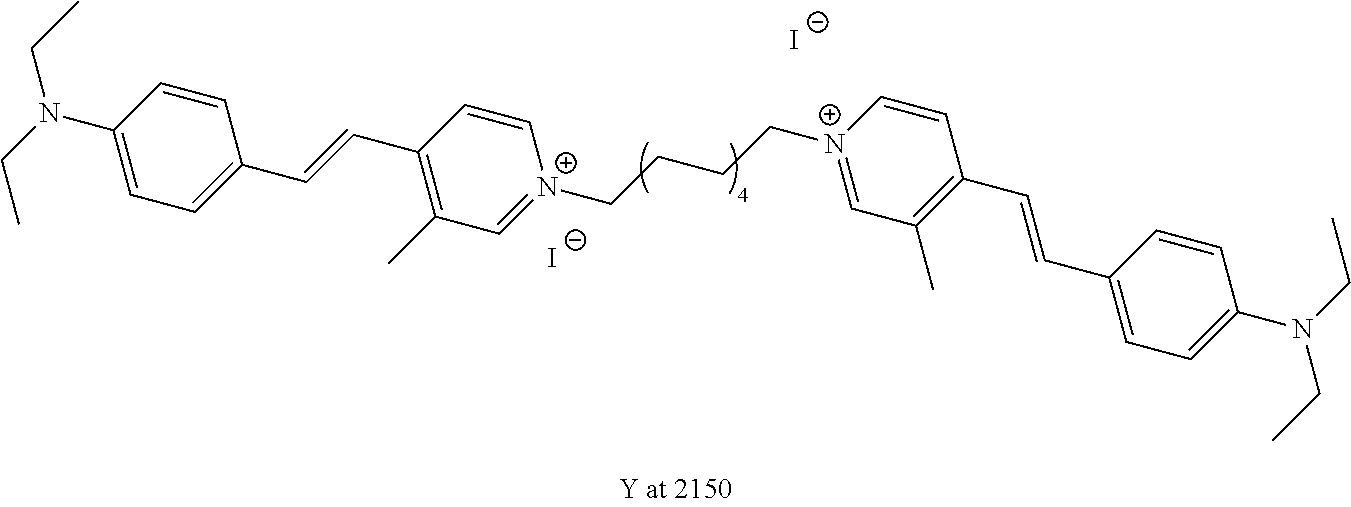
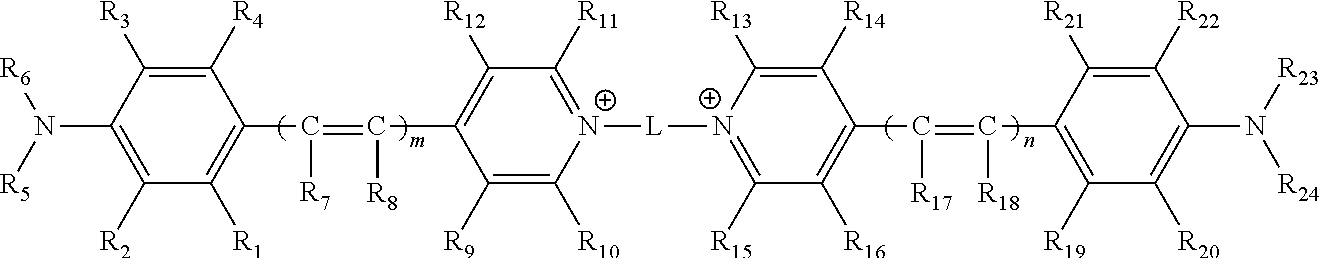
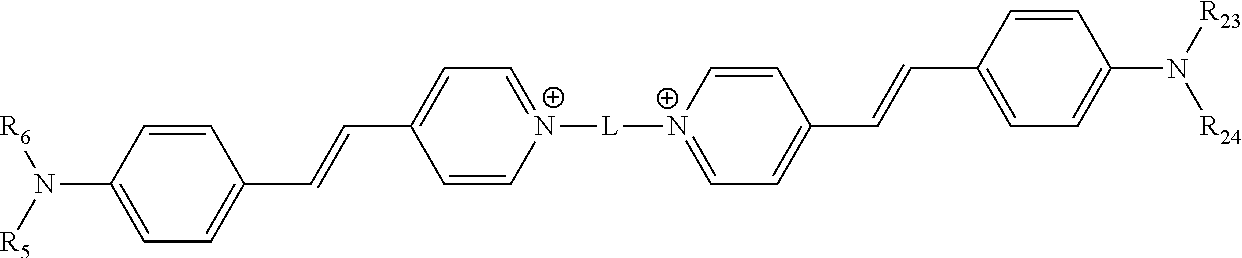
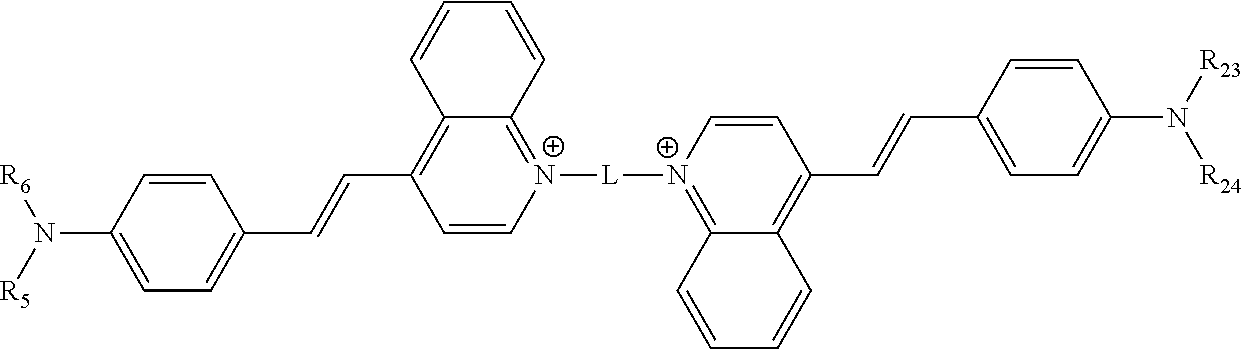
D00001

D00002
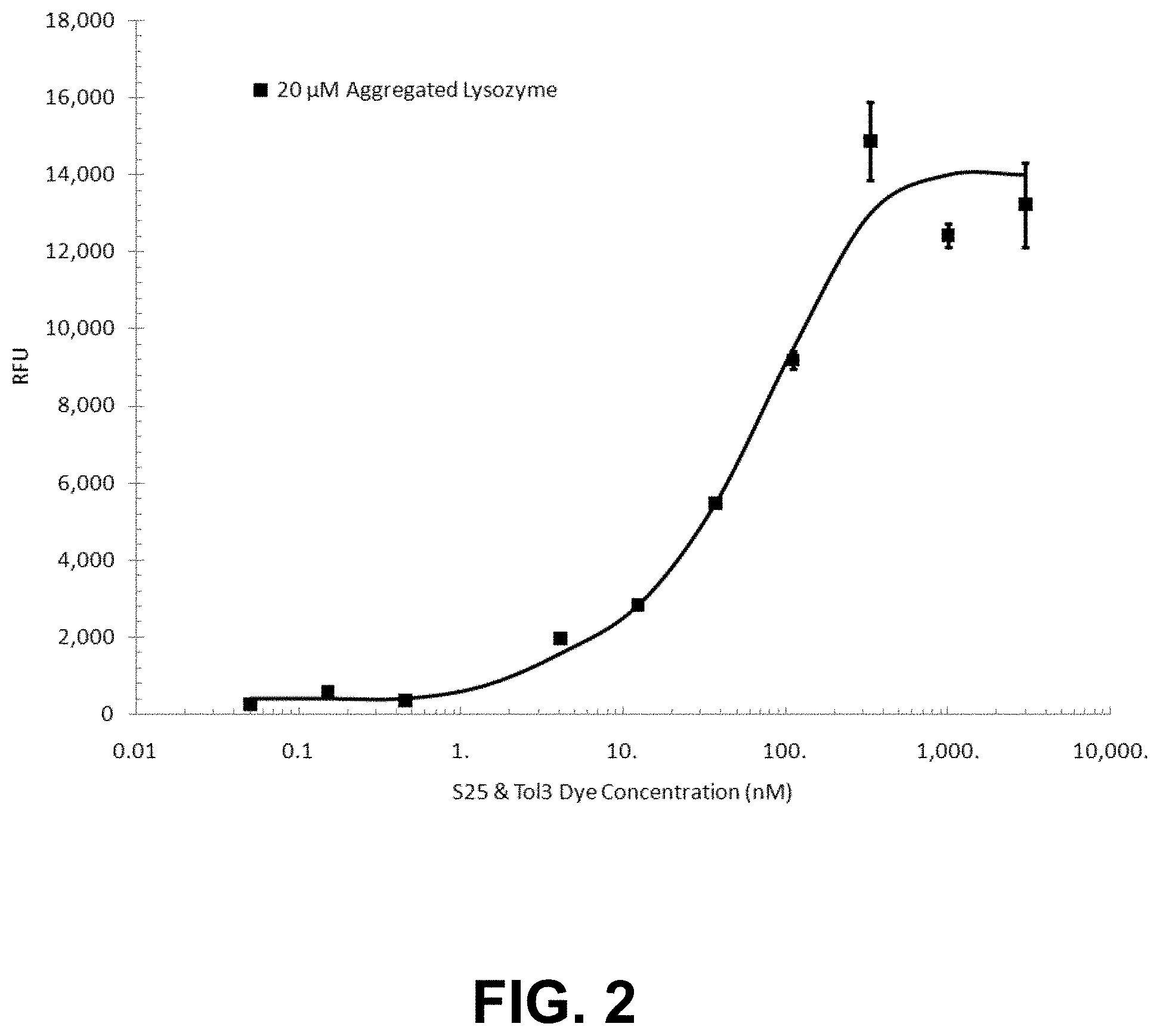
D00003
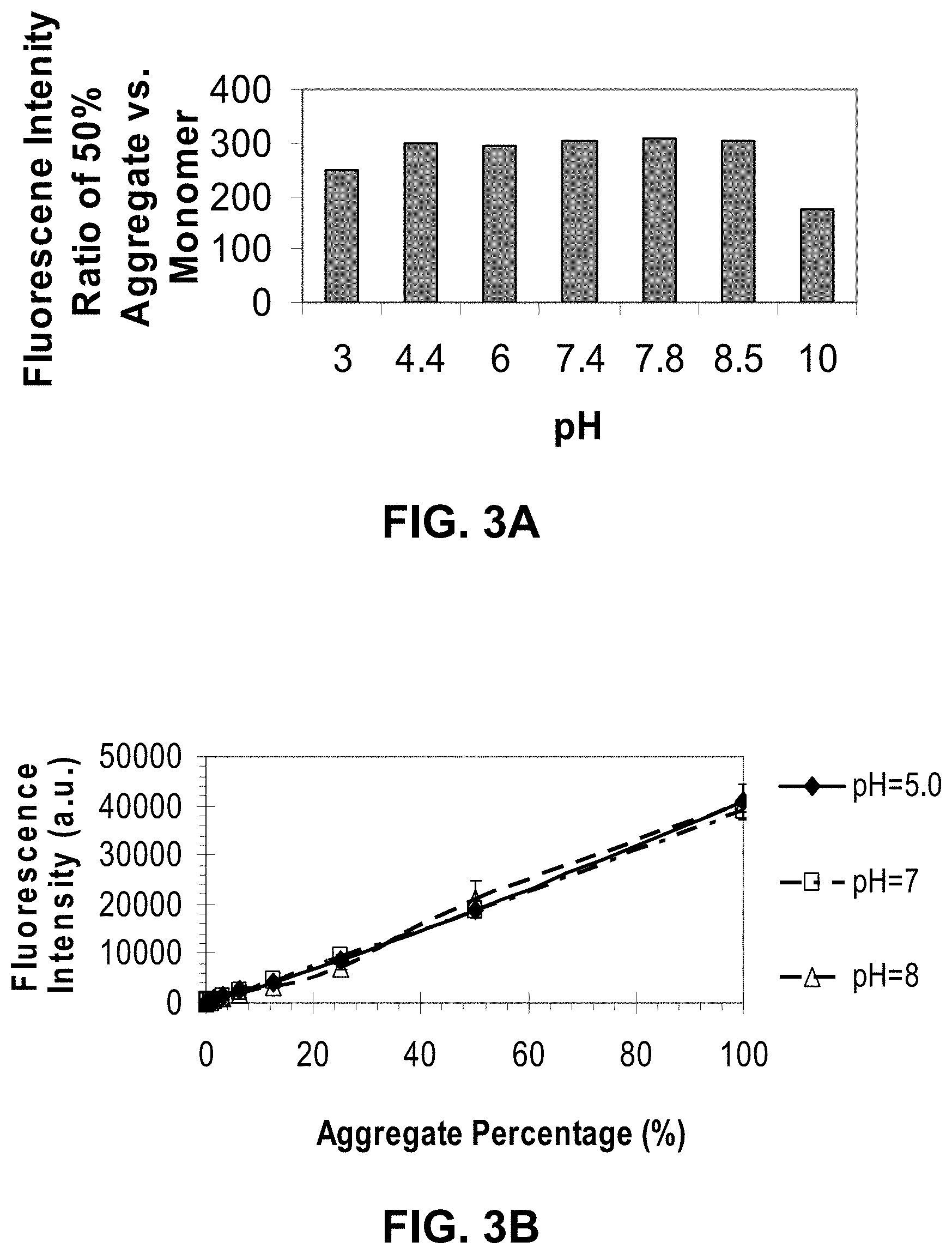
D00004
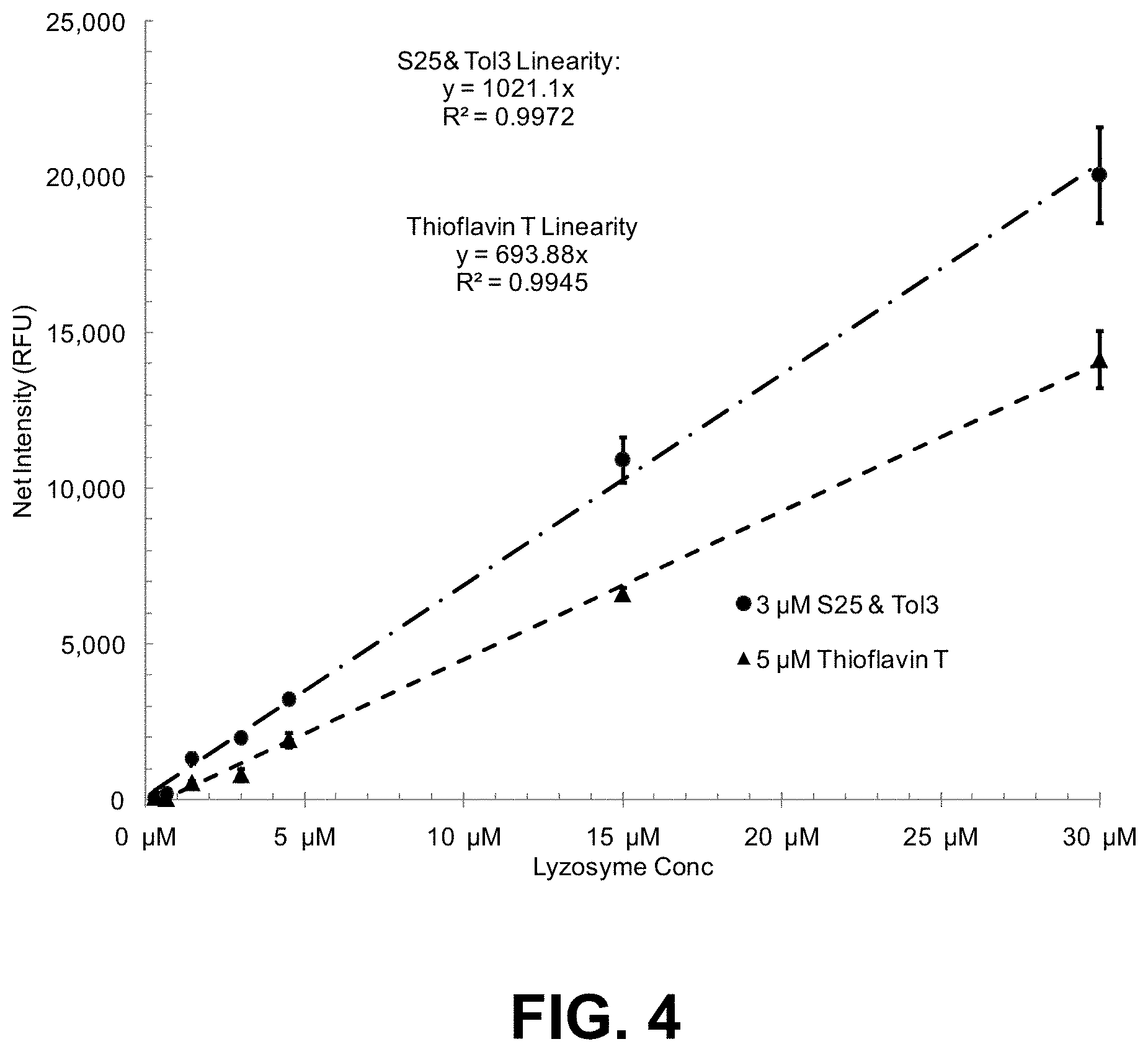
D00005
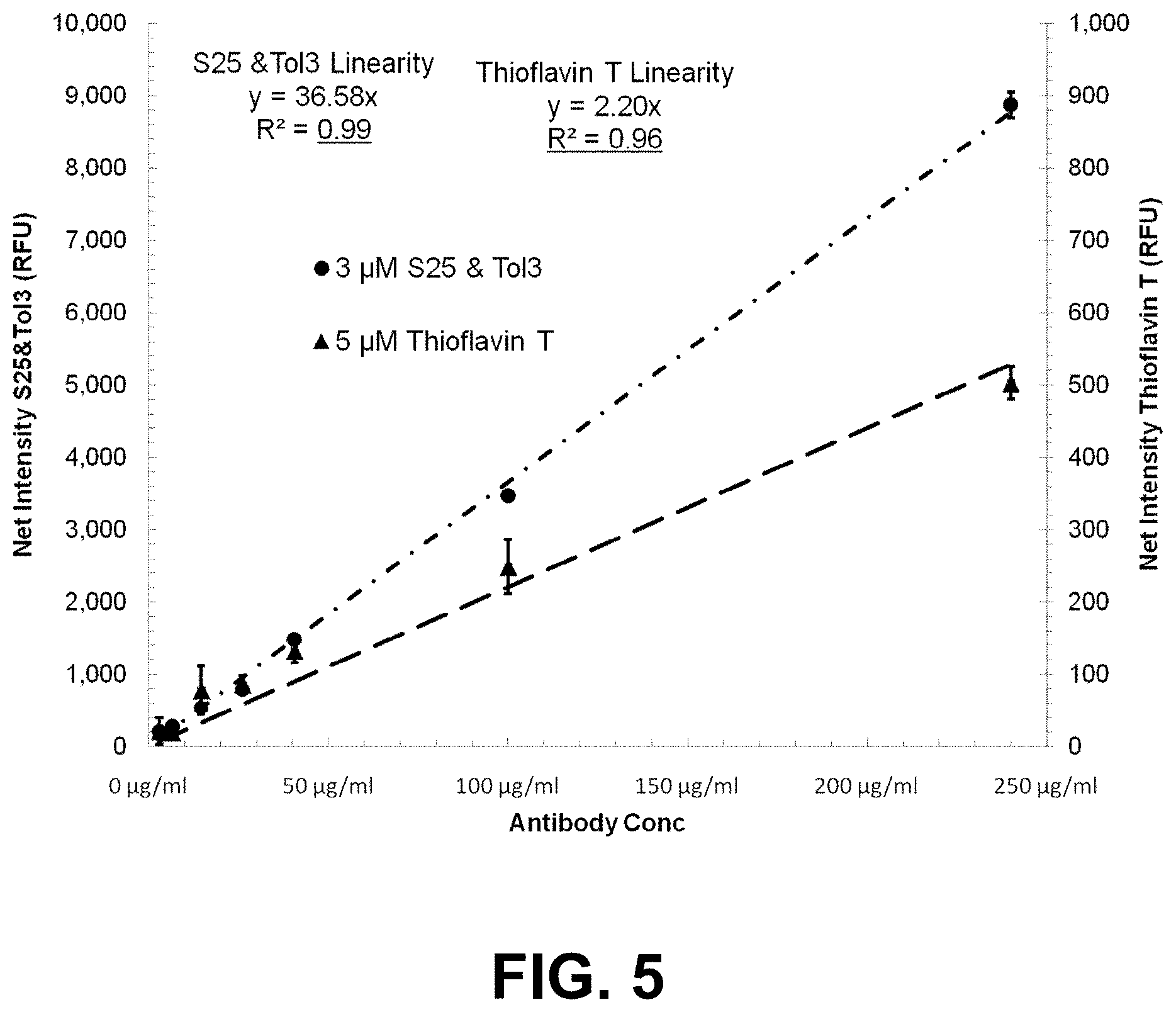
D00006

D00007
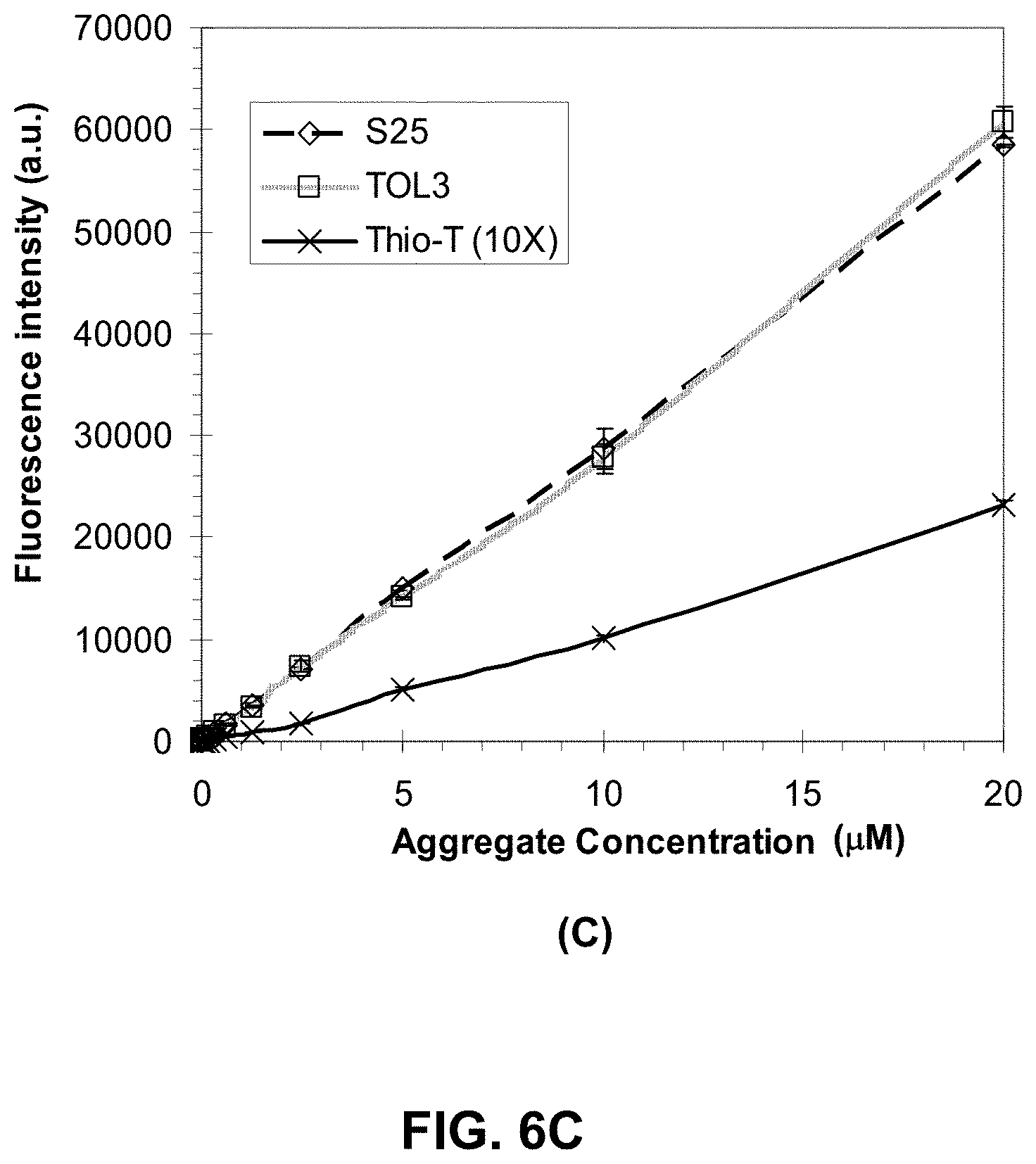
D00008

D00009
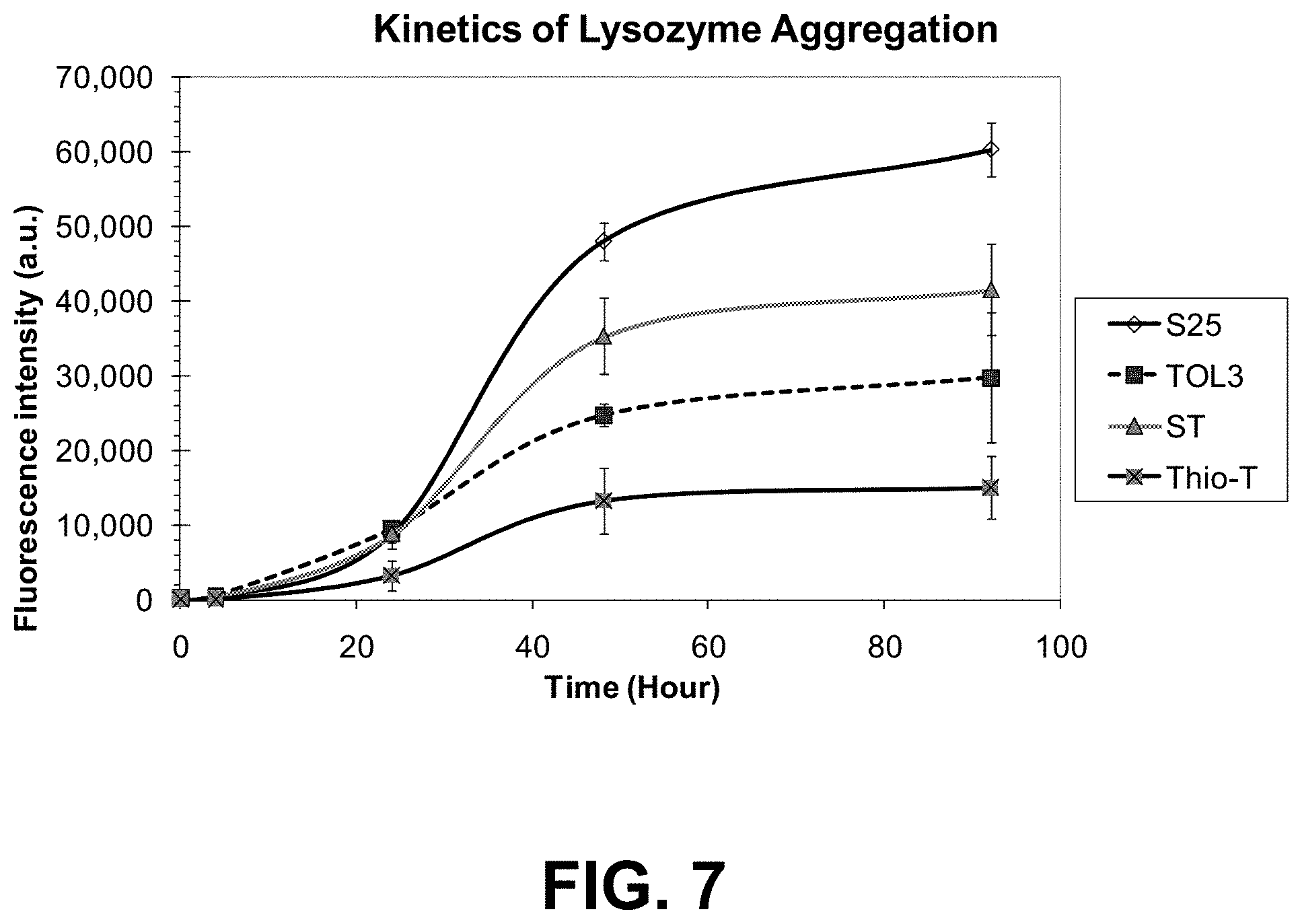
D00010

D00011
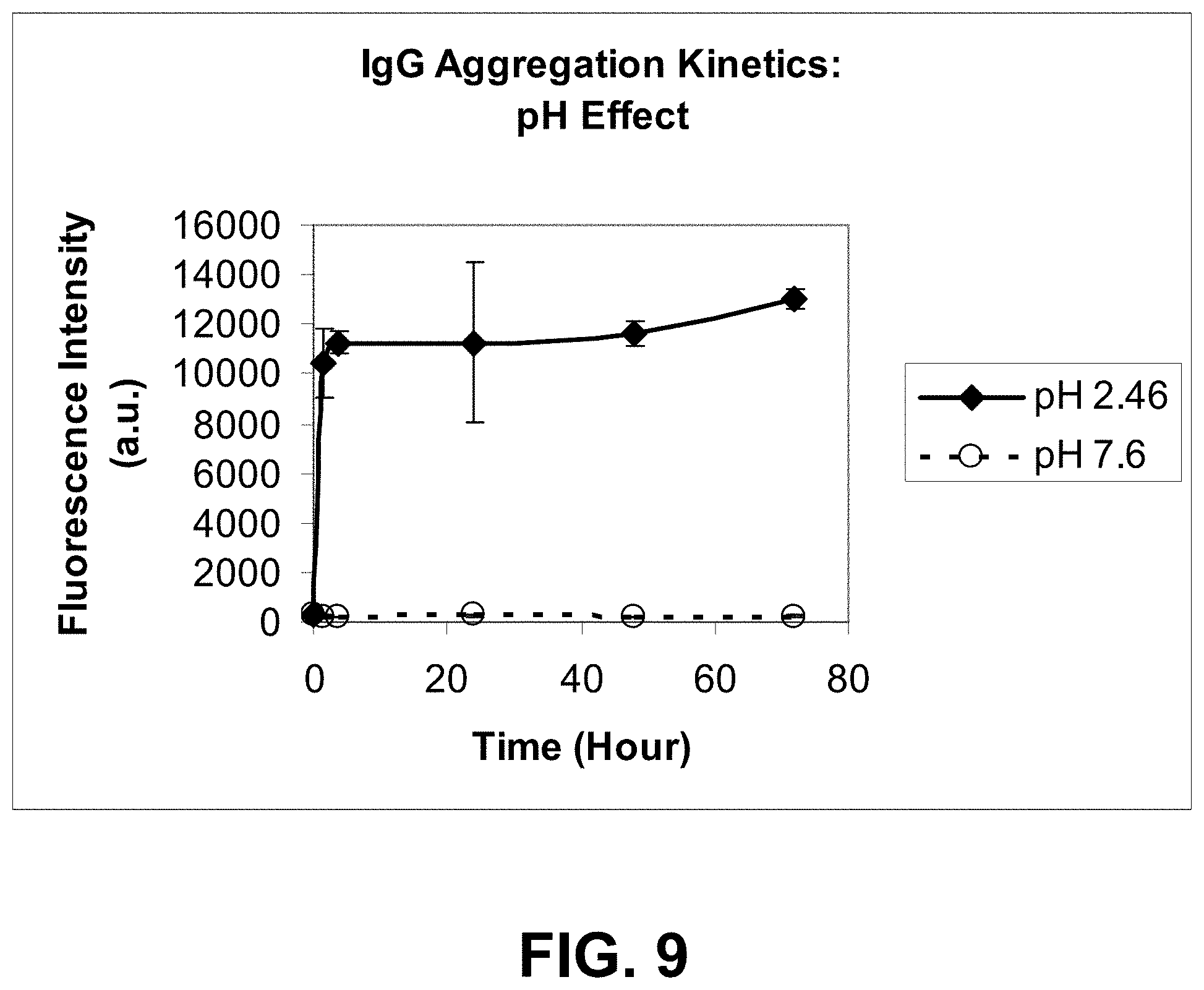
D00012

D00013

D00014

D00015
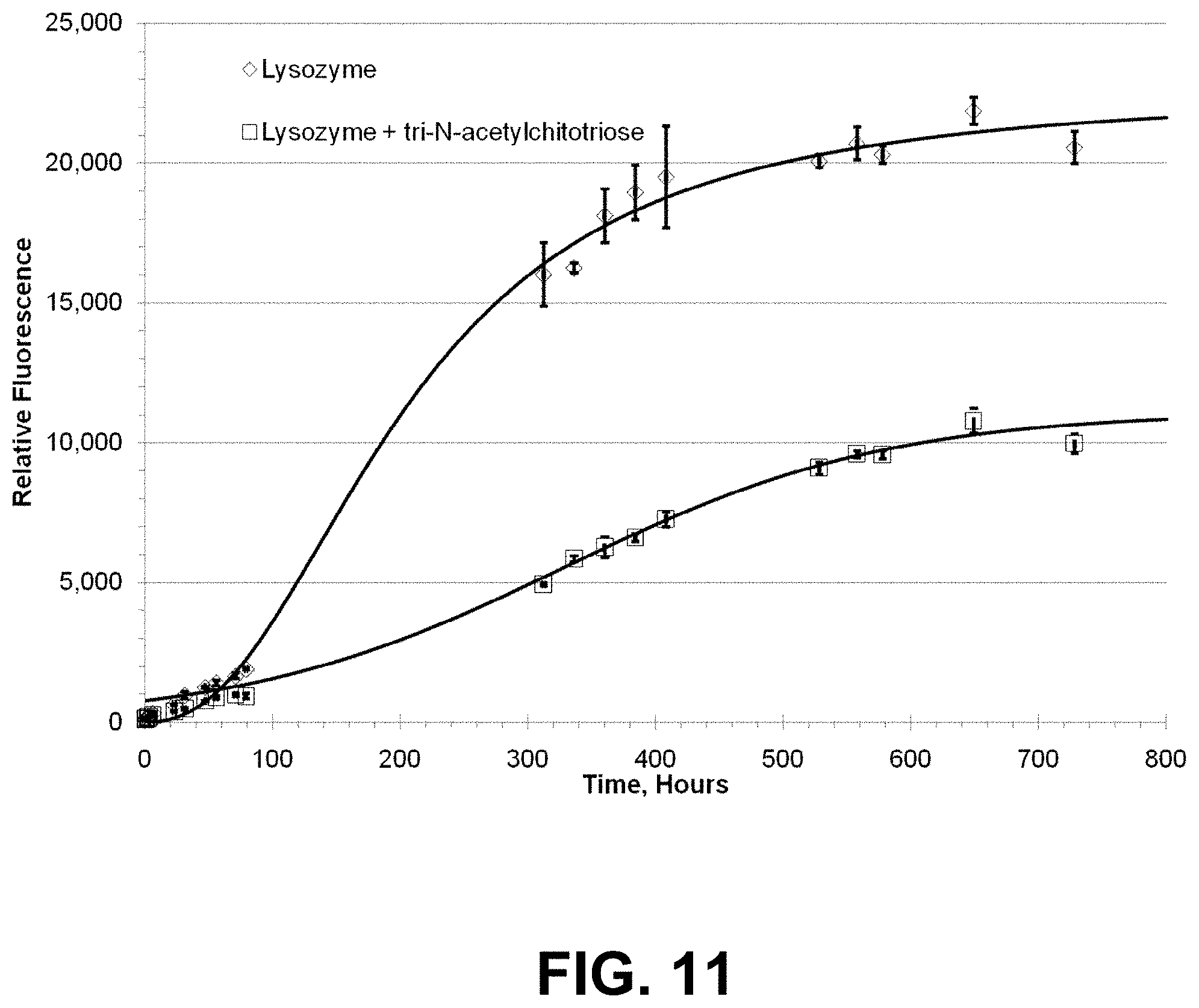
D00016
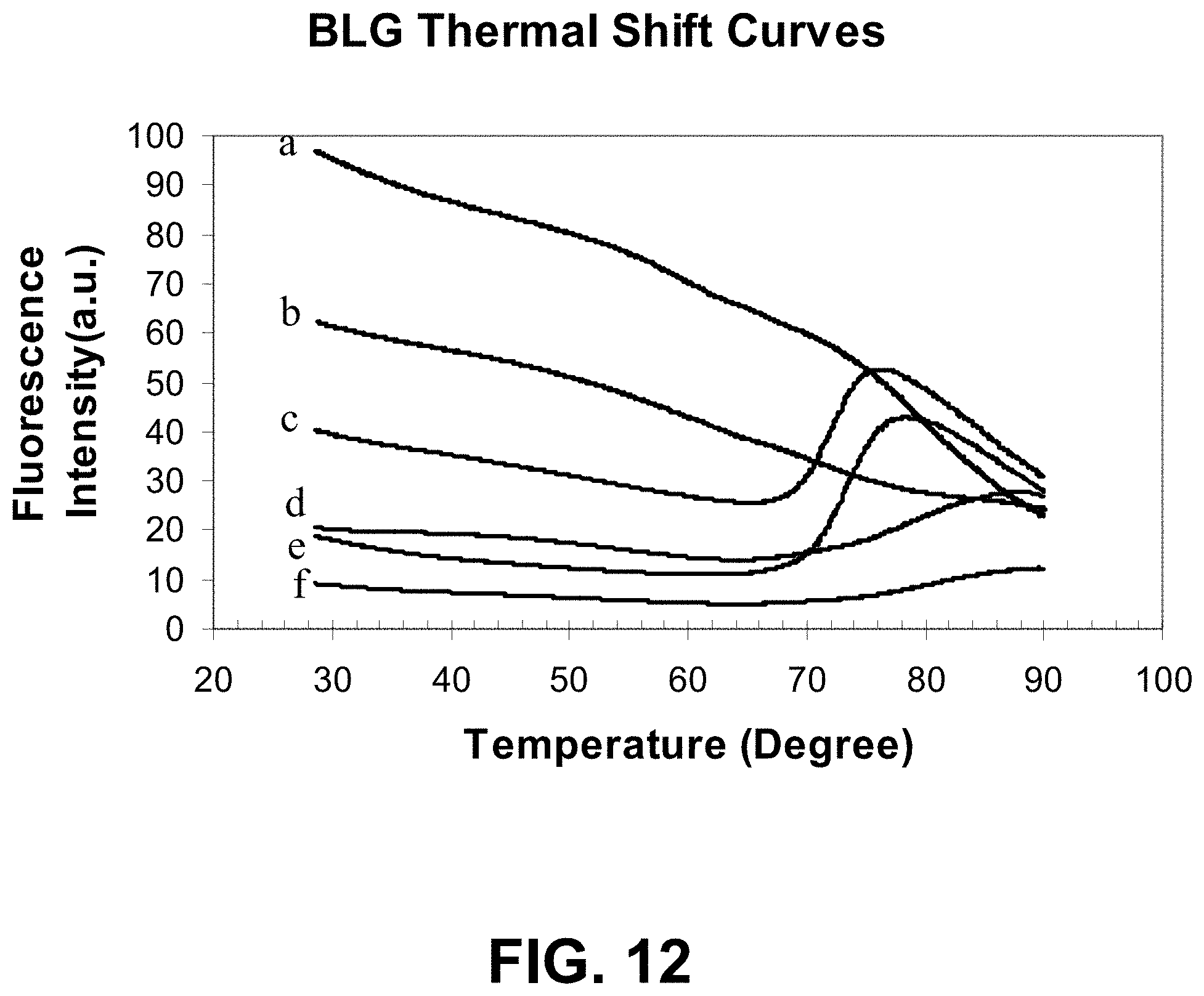
D00017
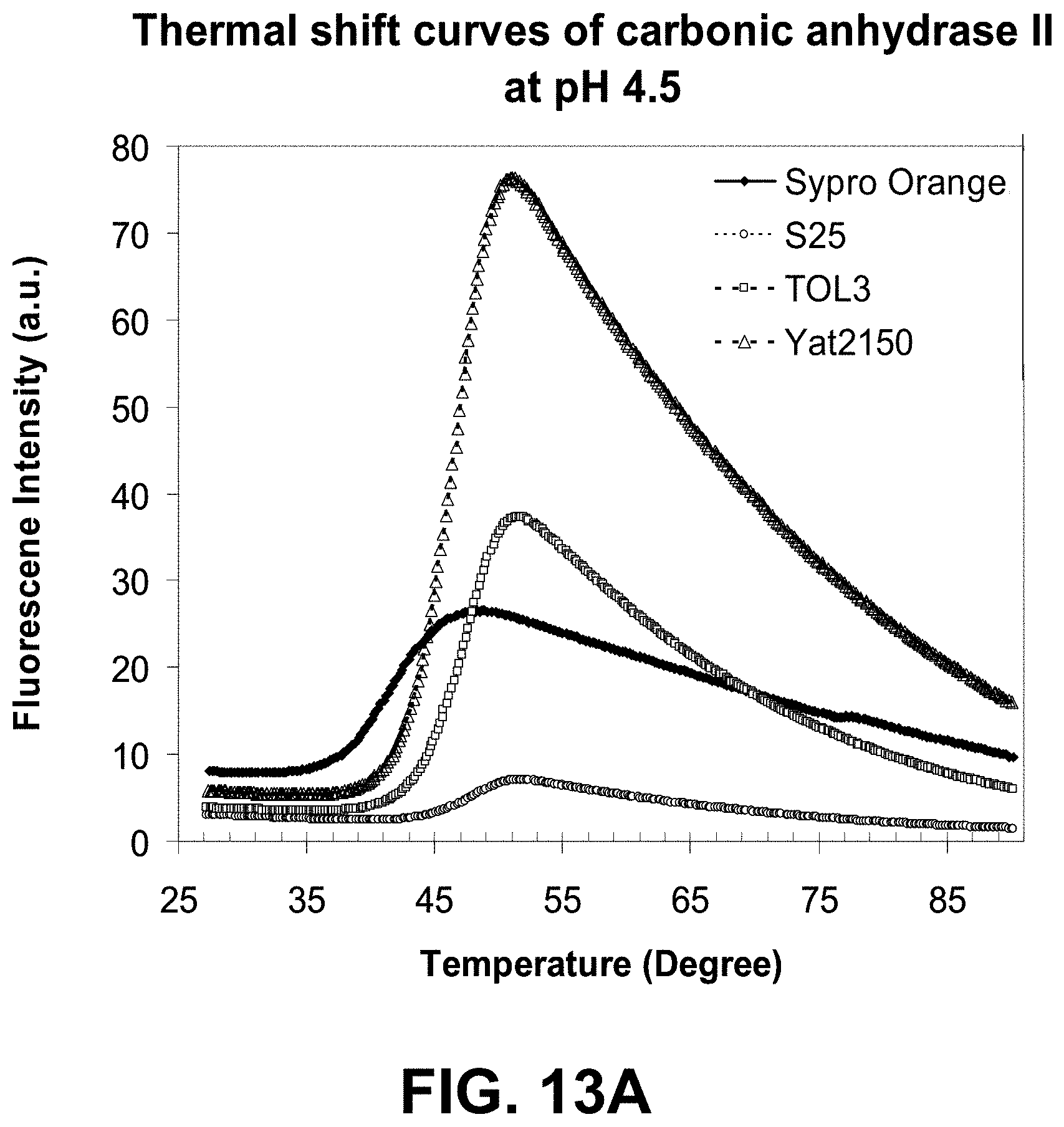
D00018
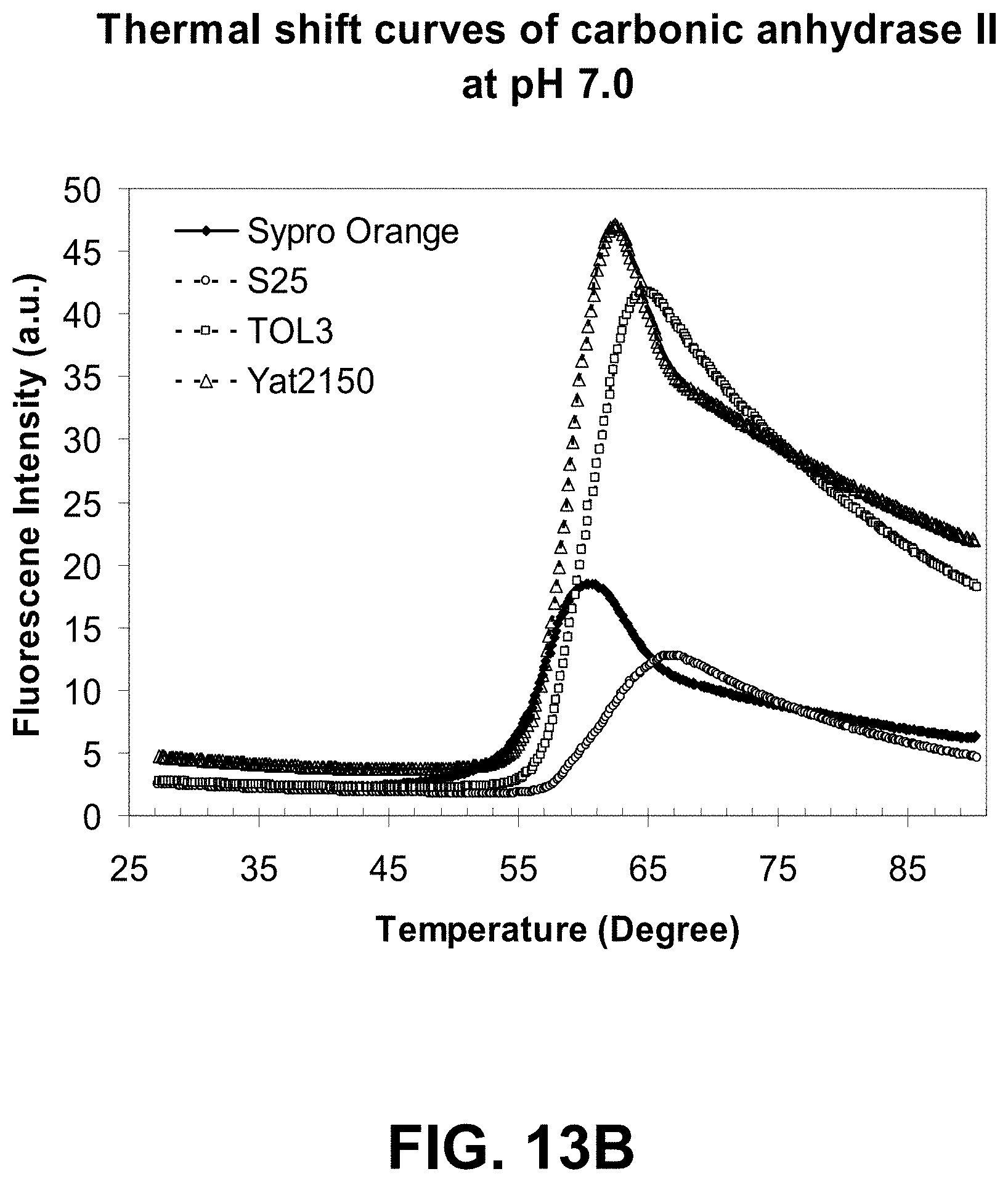
D00019
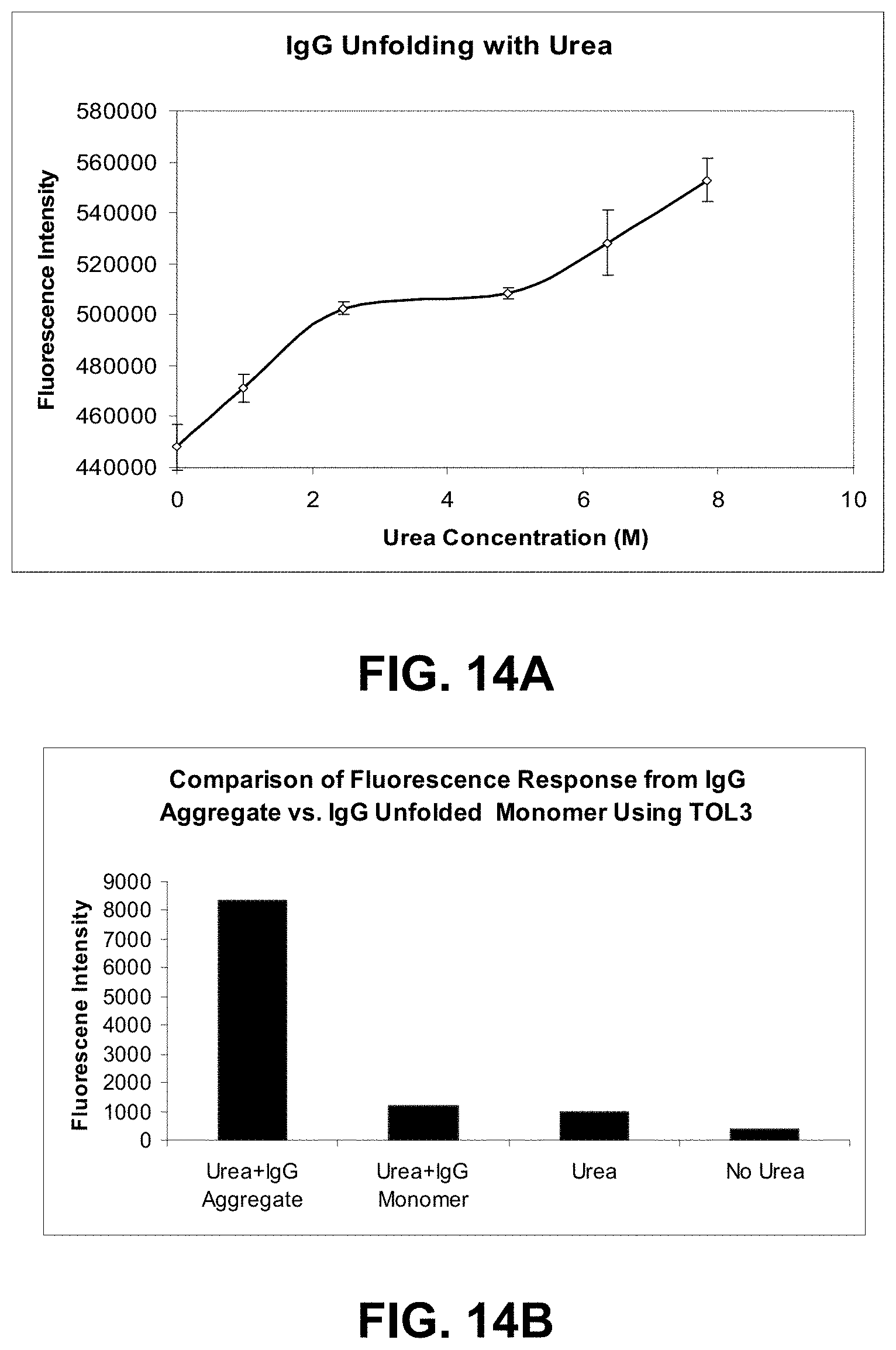
D00020

D00021
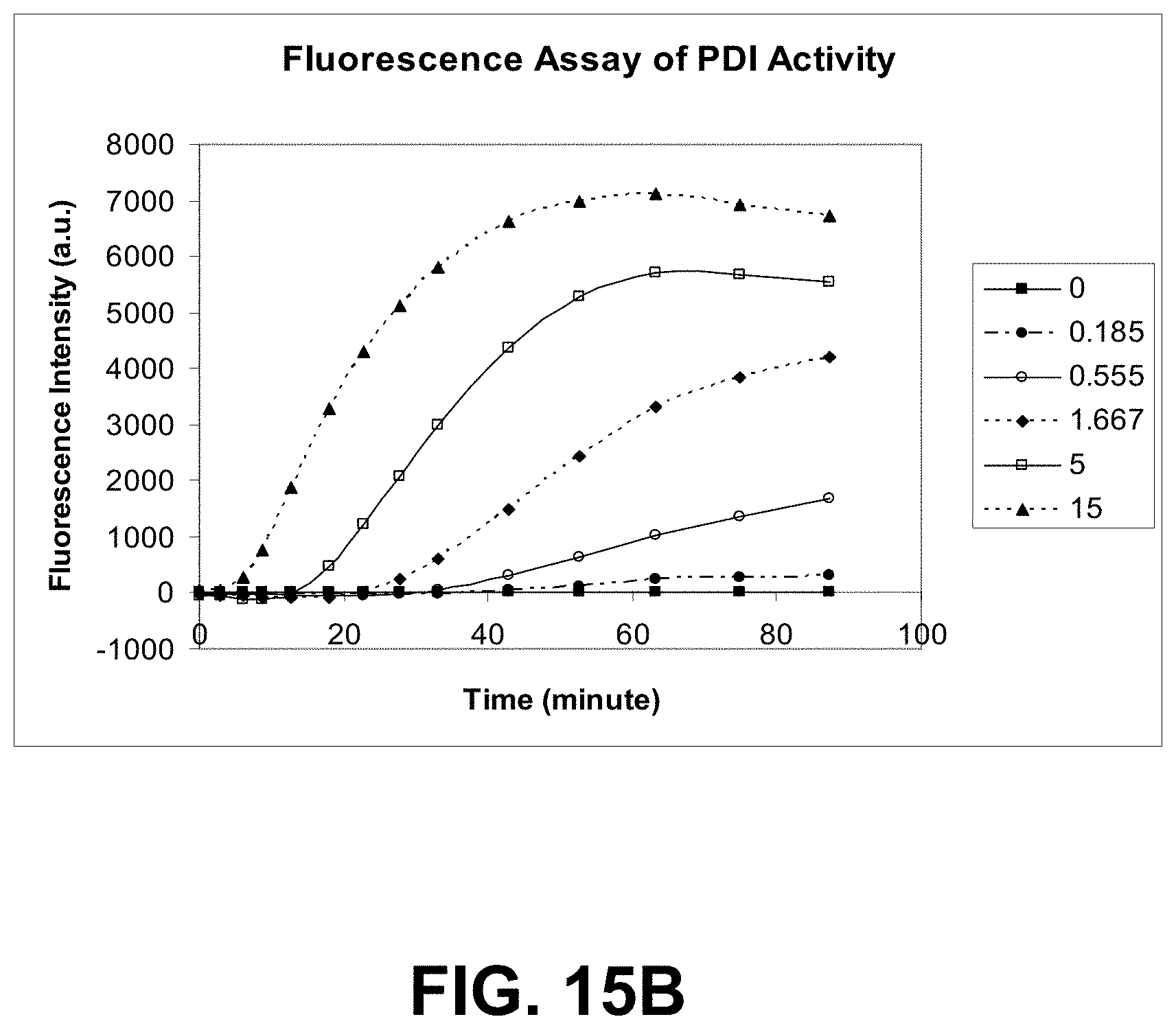
D00022
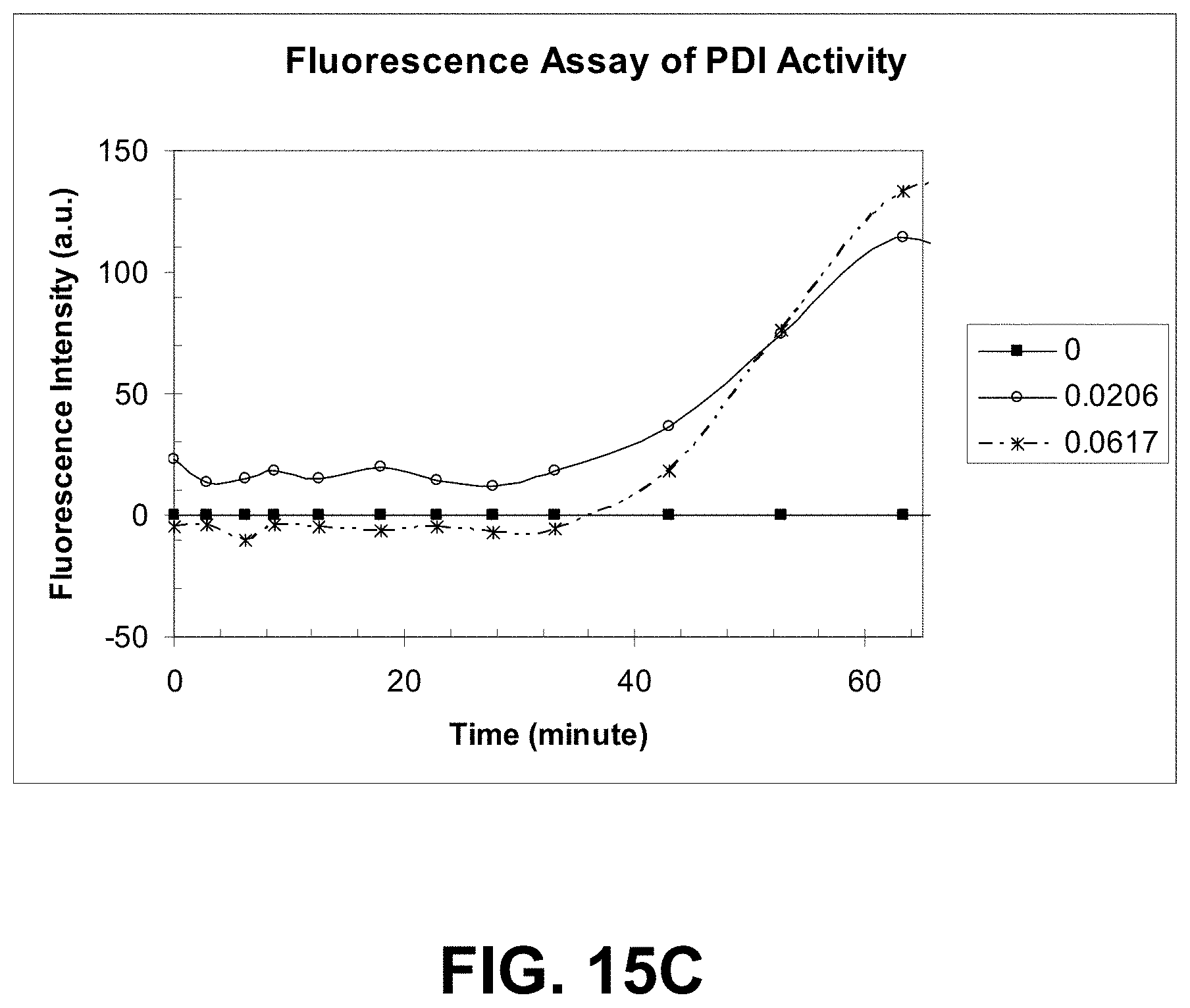
D00023
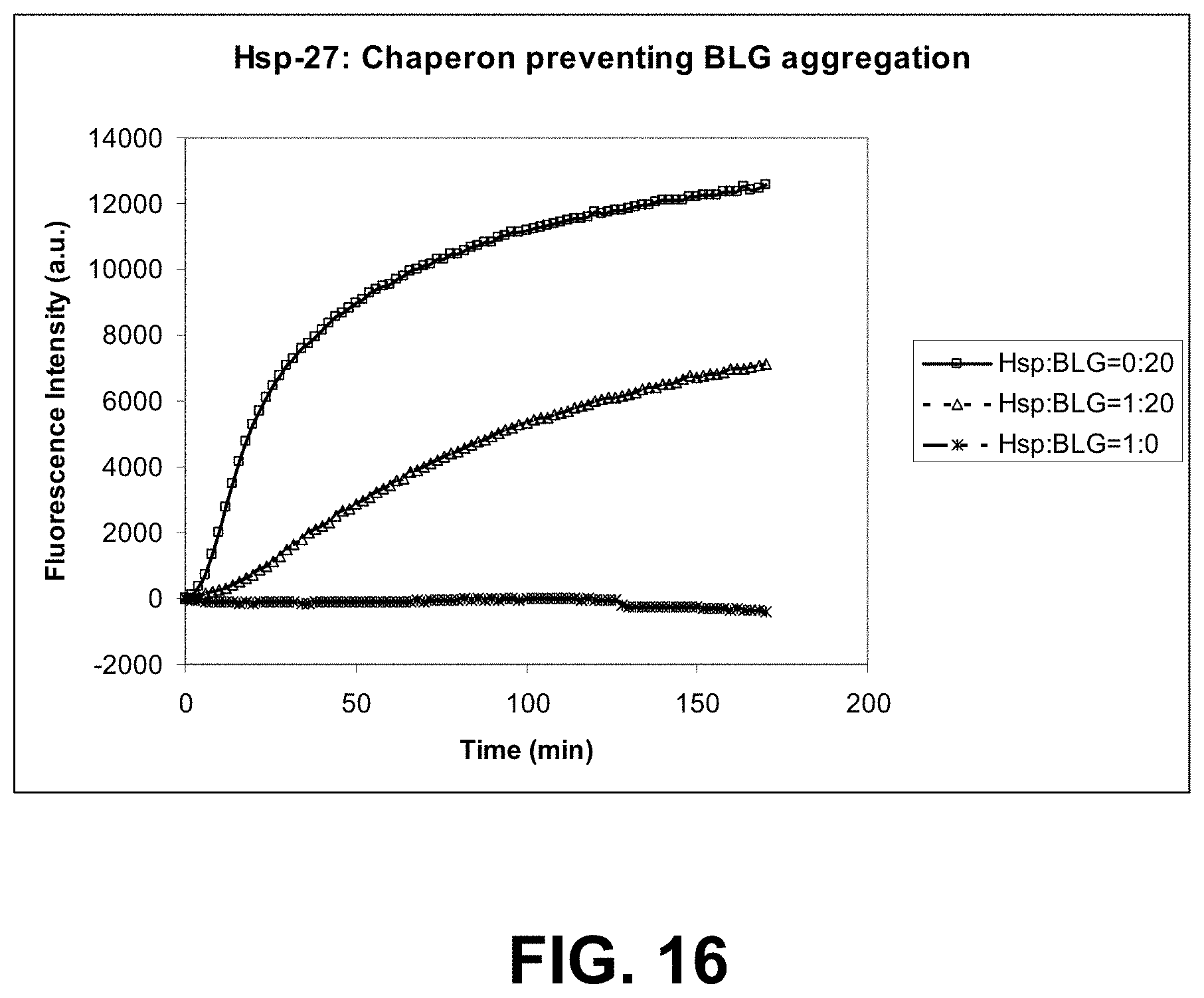
D00024
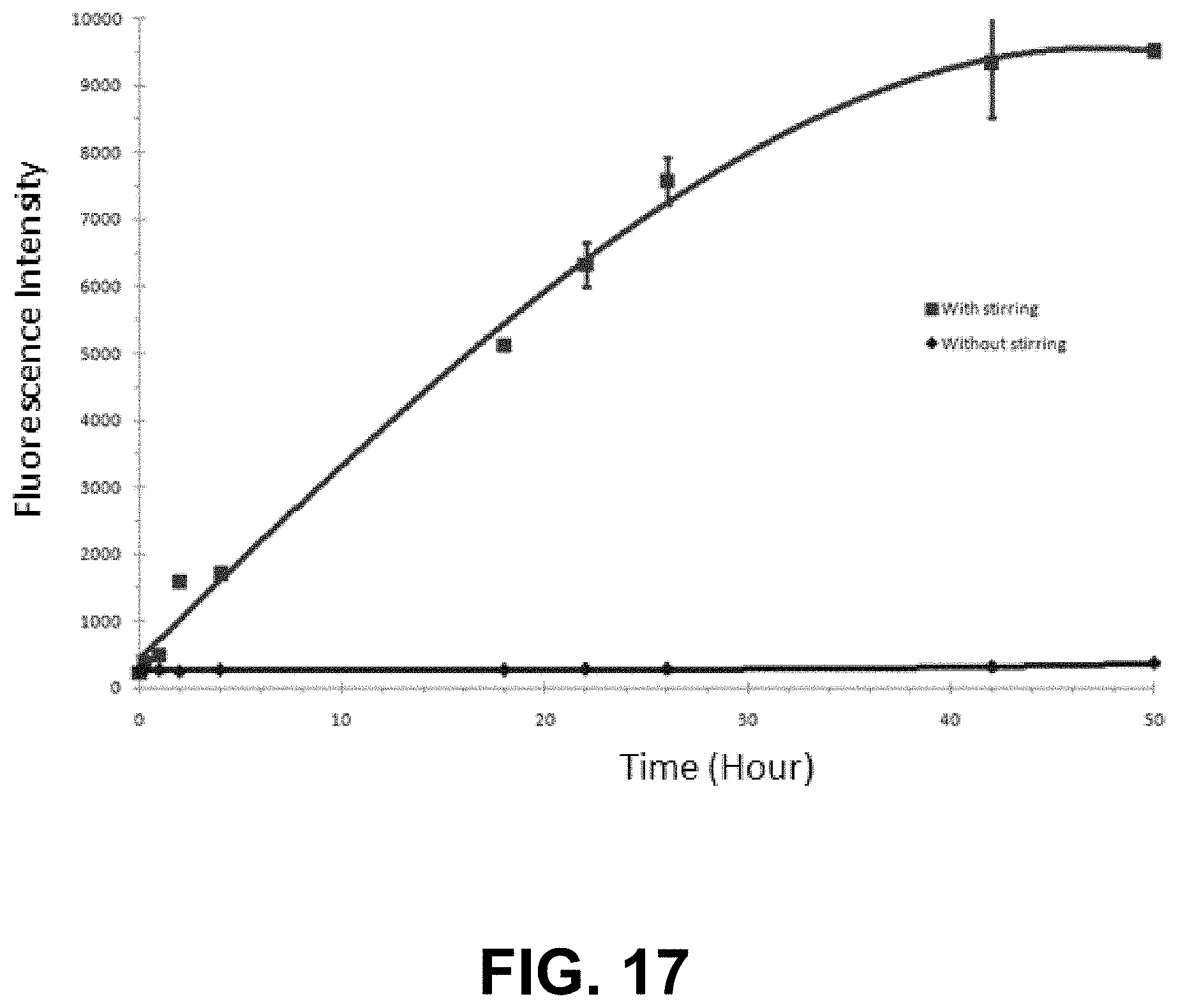
D00025
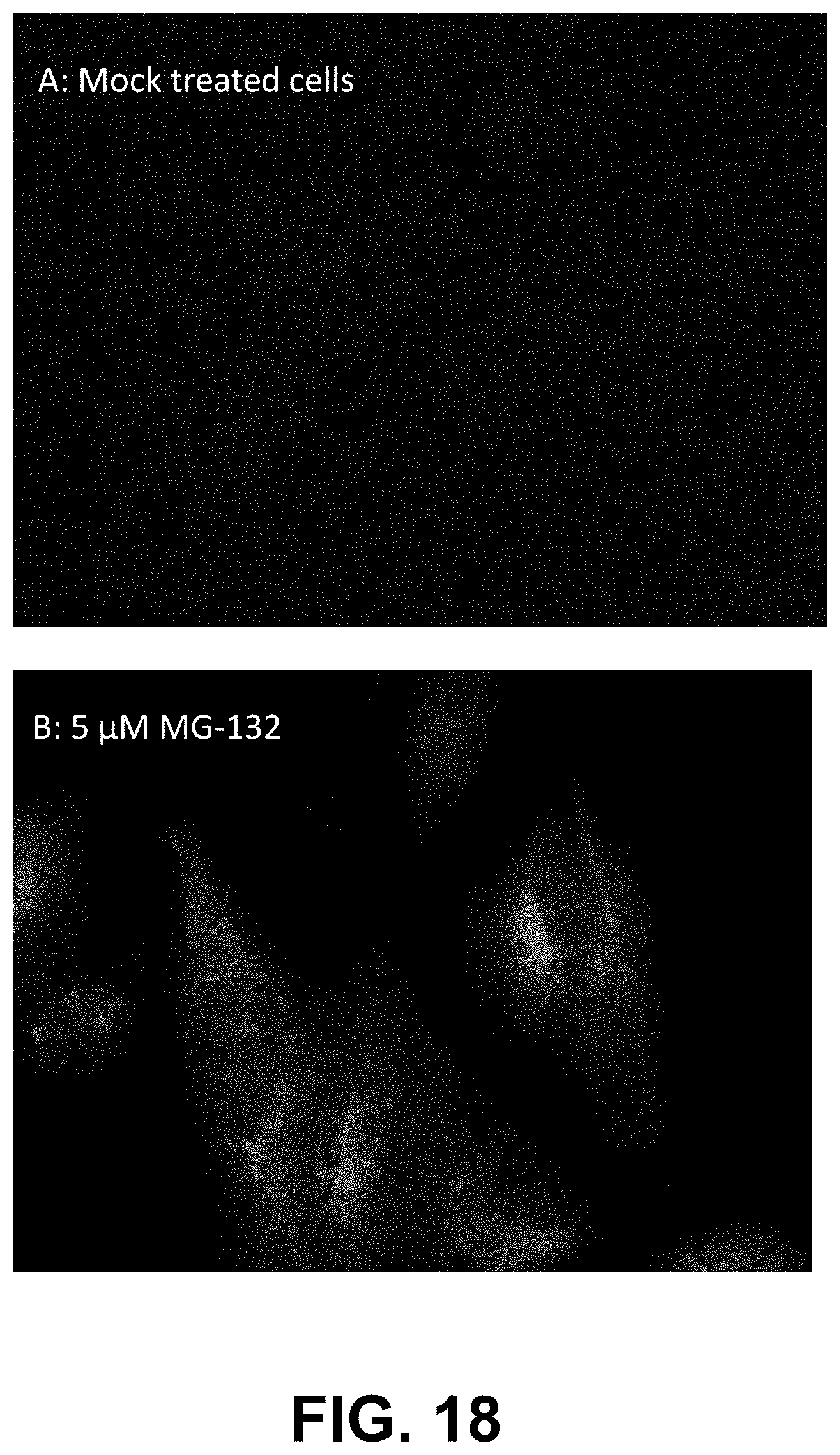
D00026

D00027

D00028

D00029

D00030

D00031
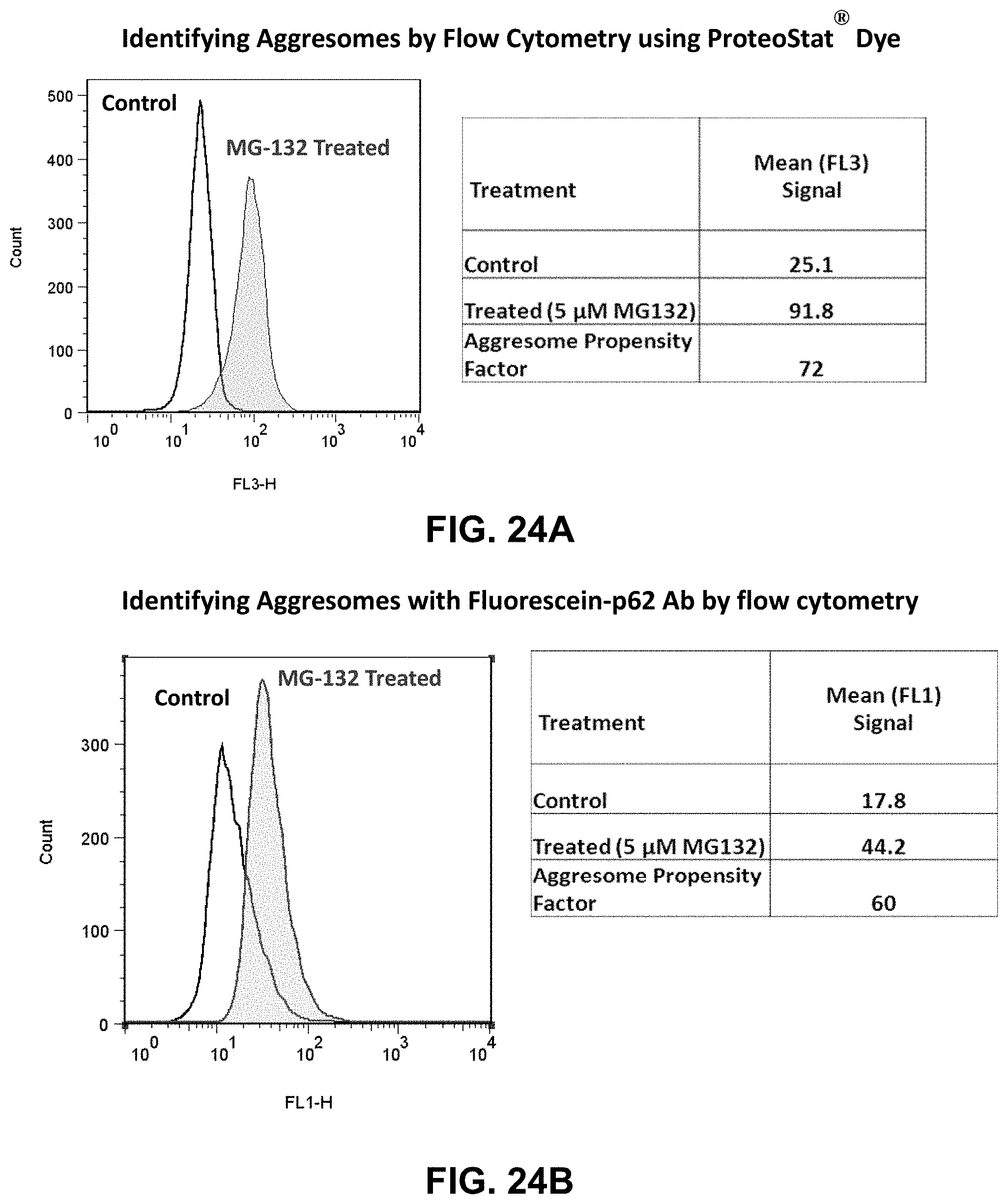
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.