Hot-dip Al-plated Steel Sheet Production Method, And Hot-dip Al-plated Steel Sheet
FURUKAWA; Shinya ; et al.
U.S. patent application number 16/982786 was filed with the patent office on 2021-01-07 for hot-dip al-plated steel sheet production method, and hot-dip al-plated steel sheet. The applicant listed for this patent is Nippon Steel Corporation. Invention is credited to Shinya FURUKAWA, Yasunori HATTORI, Koutarou ISHII.
| Application Number | 20210002752 16/982786 |
| Document ID | / |
| Family ID | |
| Filed Date | 2021-01-07 |
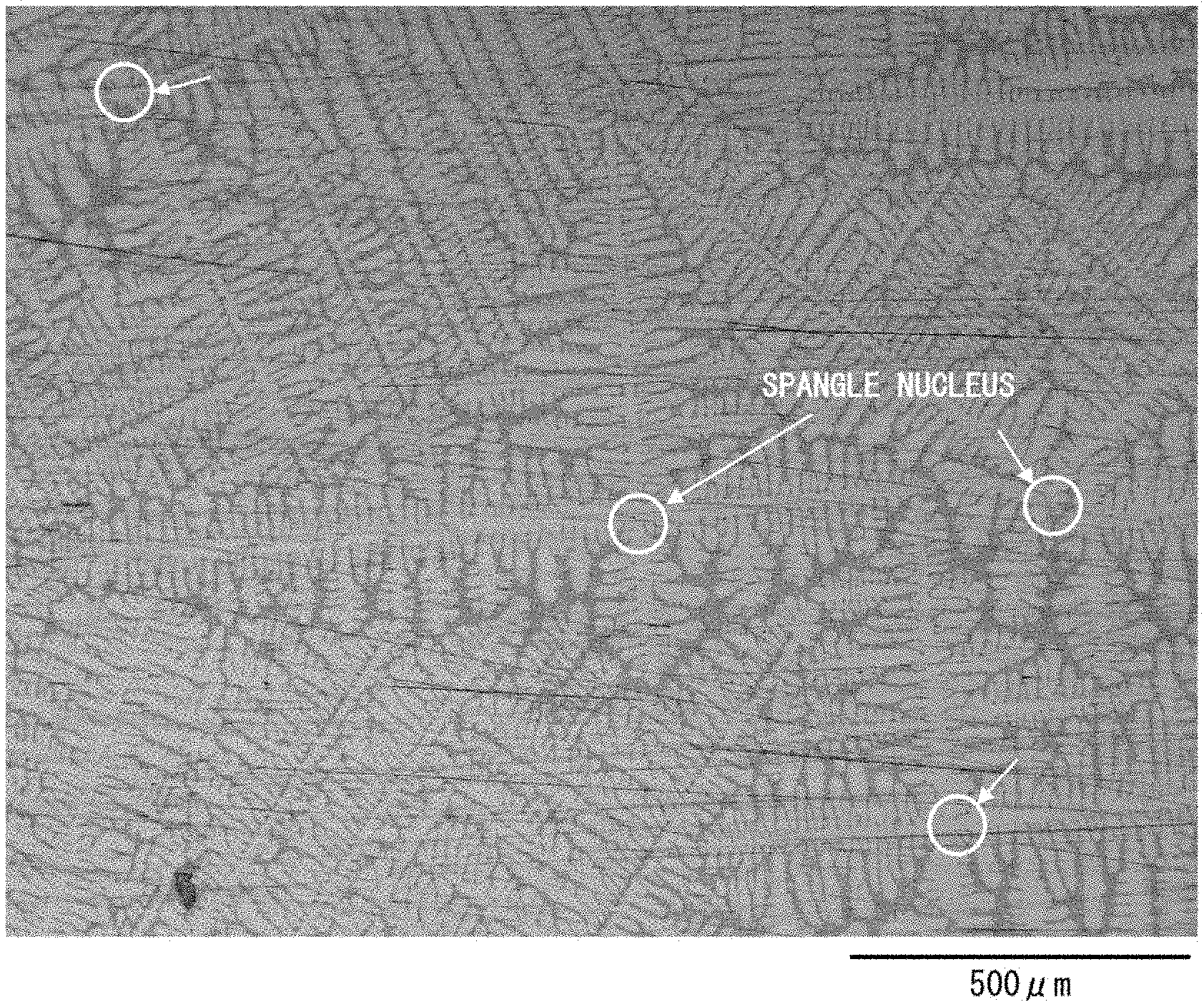



| United States Patent Application | 20210002752 |
| Kind Code | A1 |
| FURUKAWA; Shinya ; et al. | January 7, 2021 |
HOT-DIP AL-PLATED STEEL SHEET PRODUCTION METHOD, AND HOT-DIP AL-PLATED STEEL SHEET
Abstract
Provided is a method for producing a hot-dip Al-based metal-plated steel sheet including a plated layer having a surface on which fine spangle are stably formed. A method for producing a hot-dip Al-based metal-plated steel sheet, includes a composition adjusting step of adding a B-containing master alloy so as to adjust a composition of an Al-based hot-dip plating bath (3) containing aluminum as a main component, the composition being adjusted so that the Al-based hot-dip plating bath has a B concentration of not less than 0.005 mass % and a K concentration of more than 0 mass % and less than 0.0005 mass %, the K concentration being reduced in the Al-based hot-dip plating bath by supplying gas into the Al-based hot-dip plating bath so as to remove a suspended matter on a surface of the Al-based hot-dip plating bath.
| Inventors: | FURUKAWA; Shinya; (Tokyo, JP) ; ISHII; Koutarou; (Tokyo, JP) ; HATTORI; Yasunori; (Tokyo, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Appl. No.: | 16/982786 | ||||||||||
| Filed: | March 26, 2018 | ||||||||||
| PCT Filed: | March 26, 2018 | ||||||||||
| PCT NO: | PCT/JP2018/012124 | ||||||||||
| 371 Date: | September 21, 2020 |
| Current U.S. Class: | 1/1 |
| International Class: | C23C 2/12 20060101 C23C002/12; C23C 2/40 20060101 C23C002/40 |
Claims
1. A method for producing a hot-dip Al-based metal-plated steel sheet, comprising: a composition adjusting step of adding a B-containing master alloy so as to adjust a composition of an Al-based hot-dip plating bath containing aluminum as a main component, the composition being adjusted so that the Al-based hot-dip plating bath has a B concentration of not less than 0.005 mass % and a K concentration of more than 0 mass % and less than 0.0005 mass %; and a plating step of dipping a substrate steel sheet in the Al-based hot-dip plating bath, which has the composition thus adjusted, so as to cause the substrate steel sheet to pass through the Al-based hot-dip plating bath, in the composition adjusting step, the K concentration being reduced in the Al-based hot-dip plating bath by supplying gas into the Al-based hot-dip plating bath so as to remove a suspended matter on a surface of the Al-based hot-dip plating bath.
2. The method as set forth in claim 1, wherein in the composition adjusting step, the gas is supplied into the Al-based hot-dip plating bath by blowing the gas into the Al-based hot-dip plating bath.
3. The method as set forth in claim 1, wherein in the composition adjusting step, the gas is supplied into the Al-based hot-dip plating bath by using a stirring mechanism to stir the Al-based hot-dip plating bath on or near the surface of the Al-based hot-dip plating bath.
4. The method as set forth in claim 3, wherein the stirring mechanism is a stirring machine including a stirring blade.
5. The method as set forth in claim 3, wherein the stirring mechanism is a drive roll including a rotation part that is rotatable on an axis by external power, and the gas is supplied into the Al-based hot-dip plating bath by dipping a part of the rotation part in the Al-based hot-dip plating bath so as to rotate the rotation part.
6. A hot-dip Al-based metal-plated steel sheet produced by a method recited in claim 1, comprising: a substrate steel sheet; and a hot-dip Al-based metal-plated layer provided on a surface of the substrate steel sheet and having an average B concentration of not less than 0.005 mass % and an average K concentration of more than 0 mass % and less than 0.0005 mass %.
7. The hot-dip Al-based metal-plated steel sheet as set forth in claim 6, wherein the hot-dip Al-based metal-plated layer has a surface on which not less than 100 spangle crystal nuclei are present per square centimeter surface area of the hot-dip Al-based metal-plated layer.
Description
TECHNICAL FIELD
[0001] The present invention relates to a method for producing a hot-dip Al-based metal-plated steel sheet that has fine spangles.
BACKGROUND ART
[0002] A hot-dip aluminum-based metal-plated steel sheet (hereinafter referred to as a "hot-dip Al-based metal-plated steel sheet") is a steel sheet whose surface is plated with metal, containing aluminum as a main component, by a hot-dip method so that the steel sheet can have higher corrosion resistance and higher heat resistance. Such a hot-dip Al-based metal-plated steel sheet has been widely used mainly for members that are required to have heat resistance, such as exhaust gas members of automobiles and members of combustion devices.
[0003] Note that a hot-dip Al-based metal-plated steel sheet includes a plated layer having a surface on which a spangle pattern appears, the spangle pattern being formed due to dendrites, which are structures obtained by solidification of aluminum. The spangle pattern is a characteristic geometric pattern or a characteristic flower pattern, and each region (i.e., spangle) of the spangle pattern is constituted by the dendrites.
[0004] A spangle grows during solidification of aluminum after plating. Growth of the spangle progresses as below. First, a nucleus of the spangle (hereinafter referred to as a "spangle nucleus") is formed. Then, a primary dendrite arm grows from the spangle nucleus. Subsequently, a secondary dendrite arm develops from the primary dendrite arm. Growth of such dendrite arms stops due to a collision between adjacent spangles. It follows that presence of more spangle nuclei in a plated layer causes an increase in number of spangles. This causes each spangle to have a minute size.
[0005] Presence of such a spangle does not adversely affect a quality (e.g., corrosion resistance) of a hot-dip Al-based metal-plated steel sheet. Note, however, that a hot-dip Al-based metal-plated steel sheet that is preferred in the market is a hot-dip Al-based metal-plated steel sheet that has spangles having a minute size and thus has a surface skin having an inconspicuous spangle pattern.
[0006] Under the circumstances, examples of a proposed method for producing a hot-dip aluminum-zinc alloy-plated steel sheet including a plated layer that is made of an aluminum-zinc alloy include a method in which fine spangles are formed by adding Ti, Zr, Nb, B, a boride such as aluminum boride (AlB.sub.2 or AlB.sub.12), titanium carbide (TiC), titanium boride (TiB.sub.2), or titanium aluminide (TiAl.sub.3) to a plating bath so that more substances each acting as a spangle nucleus are obtained. Such a method is disclosed in, for example, each of Patent Literatures 1 to 3.
CITATION LIST
Patent Literatures
[0007] [Patent Literature 1]
[0008] Japanese Patent Application Publication Tokukai No. 2004-115908 (Publication date: Apr. 15, 2004)
[0009] [Patent Literature 2]
[0010] Japanese Patent Application Publication Tokukai No. 2006-22409 (Publication date: Jan. 26, 2006)
[0011] [Patent Literature 3]
[0012] Japanese Patent No. 3751879 (Publication date: Dec. 16, 2005)
[0013] [Patent Literature 4]
[0014] Japanese Patent No. 5591414 (Publication date: Sep. 17, 2014)
SUMMARY OF INVENTION
Technical Problem
[0015] Note, however, that use of the above method to produce a hot-dip Al-based metal-plated steel sheet has the following problems.
[0016] Specifically, since aluminum (having a specific gravity of 2.7) is one of the lightweight metals, hot-dip aluminum is slightly lower in specific gravity than an aluminum-zinc alloy, which is an alloy of aluminum and zinc (having a specific gravity of 7.1). Thus, substances, such as Ti, titanium carbide (TiC), titanium boride (TiB.sub.2), and titanium aluminide (TiAl.sub.3), each of which is higher in specific gravity than an Al-based hot-dip plating bath, easily precipitate into a bath bottom, so that it is difficult for such substances to be uniformly dispersed in the Al-based hot-dip plating bath. This causes a problem of difficulty in stable formation of fine spangles on surfaces of hot-dip Al-based metal-plated steel sheets that are continuously produced as in industrial continuous operation.
[0017] B and aluminum boride (AlB.sub.2 or AlB.sub.12) are less different in specific gravity from an aluminum bath and thus are less likely to precipitate into a bath bottom. Note, however, that, as compared with, for example, TiB.sub.2, B and aluminum boride (AlB.sub.2 or AlB.sub.12) unfortunately bring about a less good effect of providing finer spangles.
[0018] Patent Literature 4 discloses, as a B-containing hot-dip Al-based metal-plated steel sheet, a hot-dip Al-based metal-plated steel sheet that contains B at a concentration of 0.002 mass % to 0.080 mass %. Note, however, that according to the technique disclosed in Patent Literature 4, B that is unevenly distributed over a surface of a plated layer of a hot-dip Al-based metal-plated steel sheet allows the plated layer to be more slidable against a mold, and consequently allows the plated layer to be more resistant to galling.
[0019] The present invention has been made in view of the problems, and an object of the present invention is to provide a method for producing a hot-dip Al-based metal-plated steel sheet including a plated layer having a surface on which fine spangles are stably formed.
Solution to Problem
[0020] The inventors of the present invention carried out diligent study, and finally accomplished the present invention by finding that a hot-dip Al-based metal-plated steel sheet obtained with use of an Al-based hot-dip plating bath containing both B and K in proper amounts exhibits a remarkable effect of providing finer spangles.
[0021] That is, a method for producing a hot-dip Al-based metal-plated steel sheet in accordance with an aspect of the present invention includes: a composition adjusting step of adding a B-containing master alloy so as to adjust a composition of an Al-based hot-dip plating bath containing aluminum as a main component, the composition being adjusted so that the Al-based hot-dip plating bath has a B concentration of not less than 0.005 mass % and a K concentration of more than 0 mass % and less than 0.0005 mass %; and a plating step of dipping a substrate steel sheet in the Al-based hot-dip plating bath, which has the composition thus adjusted, so as to cause the substrate steel sheet to pass through the Al-based hot-dip plating bath, in the composition adjusting step, the K concentration being reduced in the Al-based hot-dip plating bath by supplying gas into the Al-based hot-dip plating bath so as to remove a suspended matter on a surface of the Al-based hot-dip plating bath.
[0022] The method for producing a hot-dip Al-based metal-plated steel sheet in accordance with an aspect of the present invention can be configured such that in the composition adjusting step, the gas is supplied into the Al-based hot-dip plating bath by blowing the gas into the Al-based hot-dip plating bath.
[0023] The method for producing a hot-dip Al-based metal-plated steel sheet in accordance with an aspect of the present invention can be configured such that in the composition adjusting step, the gas is supplied into the Al-based hot-dip plating bath by using a stirring mechanism to stir the Al-based hot-dip plating bath on or near the surface of the Al-based hot-dip plating bath.
[0024] The method for producing a hot-dip Al-based metal-plated steel sheet in accordance with an aspect of the present invention can be configured such that the stirring mechanism is a stirring machine including a stirring blade.
[0025] The method for producing a hot-dip Al-based metal-plated steel sheet in accordance with an aspect of the present invention can be configured such that the stirring mechanism is a drive roll including a rotation part that is rotatable on an axis by external power, and the gas is supplied into the Al-based hot-dip plating bath by dipping a part of the rotation part in the Al-based hot-dip plating bath so as to rotate the rotation part.
[0026] A hot-dip Al-based metal-plated steel sheet in accordance with an aspect of the present invention is a hot-dip Al-based metal-plated steel sheet produced by a method recited above, including: a substrate steel sheet; and a hot-dip Al-based metal-plated layer provided on a surface of the substrate steel sheet and having an average B concentration of not less than 0.005 mass % and an average K concentration of more than 0 mass % and less than 0.0005 mass %.
[0027] The hot-dip Al-based metal-plated steel sheet in accordance with an aspect of the present invention is configured such that the hot-dip Al-based metal-plated layer has a surface on which not less than 100 spangle crystal nuclei are present per square centimeter surface area of the hot-dip Al-based metal-plated layer.
Advantageous Effects of Invention
[0028] An aspect of the present invention makes it possible to bring about an effect of providing a method for producing a hot-dip Al-based metal-plated steel sheet including a plated layer having a surface on which fine spangles are stably formed.
BRIEF DESCRIPTION OF DRAWINGS
[0029] FIG. 1 is an optical photomicrograph of a state in which an outermost surface of a hot-dip Al-based metal-plated steel sheet in accordance with an embodiment of the present invention has been polished so that a dendrite structure is made observable.
[0030] FIG. 2 has cross-sectional views schematically illustrating an aluminum pot and a pre-melt pot each provided in plating equipment for continuous production of a hot-dip Al-based metal-plated steel sheet. (a) of FIG. 2 is a cross-sectional view illustrating a state in which an Al-based hot-dip plating bath in a pre-melt pot is being stirred with use of a propeller stirrer. (b) of FIG. 2 is a cross-sectional view illustrating a state in which an Al-based hot-dip plating bath in a pre-melt pot is being stirred by blowing gas.
[0031] FIG. 3 is a view schematically illustrating a testing device for carrying out a black foreign substance roll retention test with respect to a plating bath for use in production of a hot-dip Al-based metal-plated steel sheet.
[0032] FIG. 4 is a perspective view illustrating a surface state of a drive roll to which a black foreign substance has adhered.
[0033] FIG. 5 is an optical photomicrograph of a state in which an outermost surface of a hot-dip Al-based metal-plated steel sheet in accordance with a comparative example of the present invention has been polished so that a dendrite structure is made observable.
DESCRIPTION OF EMBODIMENTS
Comparative Example
[0034] First of all, before describing an embodiment of the present invention, the following description will refer to FIG. 5 and discuss, as a comparative example, respective configurations of (i) a hot-dip Al-based metal-plated steel sheet and (ii) a method for producing the hot-dip Al-based metal-plated steel sheet. The hot-dip Al-based metal-plated steel sheet and the method have been found by the inventors of the present invention, and the present invention is premised thereon.
[0035] <Hot-Dip Al-Based Metal-Plated Steel Sheet in Accordance with Comparative Example>
[0036] The following description will discuss, with reference to FIG. 5, a hot-dip Al-based metal-plated steel sheet in accordance with a comparative example of the present invention. FIG. 5 is an optical photomicrograph of a state in which an outermost surface of the hot-dip Al-based metal-plated steel sheet in accordance with a comparative example of the present invention has been polished so that a dendrite structure is made observable.
[0037] The hot-dip Al-based metal-plated steel sheet is schematically produced by forming a hot-dip Al-based metal-plated layer on a surface of a substrate steel sheet by dipping the substrate steel sheet in an Al-based hot-dip plating bath, containing aluminum as a main component, so as to cause the substrate steel sheet to pass through the Al-based hot-dip plating bath. Al and Fe interdiffuse during production of the hot-dip Al-based metal-plated layer, so that an Al--Fe alloy layer is also formed between (on an interface between) (i) a steel base material of the substrate steel sheet and (ii) the hot-dip Al-based metal-plated layer. As illustrated in FIG. 5, dendrites having grown from spangle crystal nuclei are present on a surface of the hot-dip Al-based metal-plated layer. A density of the spangle crystal nuclei that are present on the surface of the hot-dip Al-based metal-plated layer will be described later.
[0038] (Substrate Steel Sheet)
[0039] The substrate steel sheet can be selected from various steel types, including a steel type that is conventionally employed as a plating original sheet of the hot-dip Al-based metal-plated steel sheet, in accordance with a purpose for which the substrate steel sheet is used.
[0040] The substrate steel sheet can be a stainless steel sheet in a case where corrosion resistance is regarded as important. The substrate steel sheet can have a thickness of, for example, 0.4 mm to 3.2 mm. The substrate steel sheet as used herein encompasses a substrate steel strip.
[0041] (Al--Fe Alloy Layer)
[0042] The Al--Fe alloy layer is made mainly of an Al--Fe-based intermetallic compound. Note here that (i) the Al-based hot-dip plating bath preferably contains Si and (ii) the Al--Fe-based alloy layer that is formed with use of an Si-containing Al-based plating bath contains a large amount of Si. Both an Si-free Al--Fe-based alloy layer and a so-called Al--Fe--Si-based alloy layer containing Si are herein collectively referred to as an Al--Fe-based alloy layer. An increase in thickness of the Al--Fe-based alloy layer, which is made of a brittle intermetallic compound, makes a plated layer less adhesive. This hinders press formability. From the viewpoint of press formability, the Al--Fe-based alloy layer preferably has a thickness that is as small as possible. Note, however, that an excessive reduction in thickness of the Al--Fe-based alloy layer increases a process load and is therefore uneconomical. The Al--Fe-based alloy layer can ordinarily have an average thickness of not less than 0.5 .mu.m.
[0043] (Composition of Hot-Dip Al-Based Metal-Plated Layer)
[0044] The hot-dip Al-based metal-plated layer has a chemical composition that is substantially identical to a composition of a plating bath. It follows that a composition of a plated layer can be controlled by adjusting the composition of the plating bath.
[0045] Note that the hot-dip Al-based metal-plated layer refers to a plated layer formed on the surface of the substrate steel sheet and also encompasses the Al--Fe-based alloy layer. An aluminum oxide layer formed on a topmost surface of the hot-dip Al-based metal-plated steel sheet causes no particular problem because the aluminum oxide layer is very thin. Thus, the aluminum oxide layer is assumed to be encompassed in the hot-dip Al-based metal-plated layer. In a case where, for example, a coating layer such as an organic coating is further formed on a surface of the hot-dip Al-based metal-plated steel sheet by post-treatment, it is a matter of course that such a coating layer is not encompassed in the hot-dip Al-based metal-plated layer.
[0046] As such, the term "average concentration" in the hot-dip Al-based metal-plated layer herein means a concentration obtained by averaging concentrations obtained at respective points in the hot-dip Al-based metal-plated layer in a depth direction extending from a surface of the substrate steel sheet of the hot-dip Al-based metal-plated steel sheet to an outer surface of the hot-dip Al-based metal-plated layer of the hot-dip Al-based metal-plated steel sheet. Specifically, as described later, the average concentration is measured by carrying out a concentration analysis with respect to a measurement solution in which all the hot-dip Al-based metal-plated layer has been melted. Specifically, as for an element, such as B, which is concentrated on the surface of the hot-dip Al-based metal-plated layer, an average B concentration means a B concentration in the hot-dip Al-based metal-plated layer, the B concentration being obtained by averaging concentrations of B assuming that no B is concentrated on the surface of the hot-dip Al-based metal-plated layer. Furthermore, a B concentration in the Al-based hot-dip plating bath is reflected in the average B concentration in the hot-dip Al-based metal-plated layer that has been subjected to plating.
[0047] The hot-dip Al-based metal-plated layer at least contains B and K while containing Al as a main component. Note, however, that the hot-dip Al-based metal-plated layer can contain other element(s).
[0048] Si is an additional element that is necessary for prevention or reduction of growth of the Al--Fe alloy layer during hot-dip plating. An Al-based plating bath to which Si is added has a lower melting point. This is effective in reducing a plating temperature. In a case where a plating bath contains Si at a concentration of less than 1.0 mass %, the Al--Fe-based alloy layer is formed thick during hot-dip plating due to interdiffusion of Al and Fe. This causes plating peeling during processing such as press forming. In contrast, in a case where the plating bath contains Si at a concentration of more than 12.0 mass %, a plated layer is hardened. This makes it impossible to prevent or reduce cracking in a bent part of the plated layer and consequently causes the bent part to have lower corrosion resistance. The plating bath therefore preferably contains Si at a concentration of 1.0 mass % to 12.0 mass %. In particular, the plating bath that contains Si at a concentration of less than 3.0 mass % (i) allows an Si phase to be formed in a smaller amount during solidification of the plated layer and (ii) allows a primary crystal Al phase to be softened. Such a plating bath is more effective in a case where the plating bath is used for a purpose where bendability is regarded as important.
[0049] Fe that comes from, for example, the substrate steel sheet and/or a constituent member(s) of a hot-dip plating tank is mixed in the Al-based hot-dip plating bath. The hot-dip Al-based metal-plated layer ordinarily contains Fe at a concentration of not less than 0.05 mass %. Fe is permitted to be contained in the hot-dip Al-based metal-plated layer at a concentration of up to 3.0 mass %, and more preferably not more than 2.5 mass %.
[0050] Besides the above elements, an element(s) such as Sr, Na, Ca, Sb, P, Mg, Cr, Mn, Ti, Zr, and/or V may be intentionally added to the Al-based hot-dip plating bath as necessary. Alternatively, such an element(s) that come(s) from, for example, a raw material may be mixed in the Al-based hot-dip plating bath. A hot-dip Al-plated steel sheet in accordance with a comparative example of the present invention can also contain such an element(s) that has/have been conventionally commonly permitted. Specifically, for example, the hot-dip Al-plated steel sheet can contain Sr at a concentration in a range of 0 mass % to 0.2 mass %, Na at a concentration in a range of 0 mass % to 0.1 mass %, Ca at a concentration in a range of 0 mass % to 0.1 mass %, Sb at a concentration in a range of 0 mass % to 0.6 mass %, P at a concentration in a range of 0 mass % to 0.2 mass %, Mg at a concentration in a range of 0 mass % to 5.0 mass %, Cr at a concentration in a range of 0 mass % to 1.0 mass %, Mn at a concentration in a range of 0 mass % to 2.0 mass %, Ti at a concentration in a range of 0 mass % to 0.5 mass %, Zr at a concentration in a range of 0 mass % to 0.5 mass %, and/or V at a concentration in a range of 0 mass % to 0.5 mass %.
[0051] The balance of components of the hot-dip Al-based metal-plated layer which balance is different from the above elements can be constituted by Al and unavoidable impurities.
[0052] A hot-dip Al-based metal-plated steel sheet in accordance with a comparative example of the present invention includes: a substrate steel sheet; and a hot-dip Al-based metal-plated layer that is provided on a surface of the substrate steel sheet and has an average B concentration of not less than 0.005 mass % and an average K concentration of not less than 0.0004 mass %.
[0053] This makes it possible to produce a hot-dip Al-based metal-plated steel sheet including a plated layer having a surface that is beautiful in appearance because fine spangles are sufficiently formed thereon. Such a hot-dip Al-based metal-plated steel sheet can be obtained by (i) adjusting a B concentration and a K concentration in a plating bath and (ii) causing a substrate steel sheet to pass through the plating bath. This makes it possible to achieve the hot-dip Al-based metal-plated steel sheet in which fine spangles are stably formed.
[0054] By referring to FIG. 5 again, the following description will discuss a density of spangle crystal nuclei. As illustrated in FIG. 5, spangles are non-uniform and irregular in size. Note, however, that spangle crystal nuclei that are observed through, for example, an optical microscope can be distinguished from each other.
[0055] Thus, the number of spangle crystal nuclei per visual field area can be understood by counting the number of spangle crystal nuclei present in that visual field area. The number of spangle crystal nuclei per visual field area thus understood can be converted into the approximate number of spangle crystal nuclei per square centimeter area of the hot-dip Al-based metal-plated layer. Note, however, that such a counting method as described above is merely an example, and the number of spangle crystal nuclei can be counted by any other method.
[0056] The hot-dip Al-based metal-plated layer of the hot-dip Al-based metal-plated steel sheet does not necessarily need to be provided on each of both sides of the substrate steel sheet, and alternatively can be provided on at least one of the both sides of the substrate steel sheet.
[0057] <Method for Producing Hot-Dip Al-Based Metal-Plated Steel Sheet in Accordance with Comparative Example>
[0058] A hot-dip Al-based metal-plated steel sheet in accordance with a comparative example of the present invention can be produced by a hot-dip method with use of a plating bath in which a B concentration and a K concentration have been adjusted.
[0059] A method for producing a hot-dip Al-based metal-plated steel sheet in accordance with a comparative example of the present invention includes a plating step of dipping a substrate steel sheet in an aluminum-based hot-dip plating bath, containing aluminum as a main component, so as to pass the substrate steel sheet through the aluminum-based hot-dip plating bath, the aluminum-based hot-dip plating bath having a B concentration of not less than 0.005 mass % and a K concentration of not less than 0.0004 mass %.
[0060] An average concentration of each component of the hot-dip Al-based metal-plated layer which average concentration is obtained after the plating step is substantially identical to a composition of the Al-based hot-dip plating bath. Thus, the configuration makes it possible to produce a hot-dip Al-based metal-plated steel sheet including a hot-dip Al-based metal-plated layer that has an average B concentration of not less than 0.005 mass % and an average K concentration of not less than 0.0004 mass %.
[0061] At least prior to the plating step, a composition adjusting step of adjusting the composition of the Al-based hot-dip plating bath is carried out by adjusting respective concentrations of elements contained in the Al-based hot-dip plating bath. According to the method for producing the hot-dip Al-based metal-plated steel sheet in accordance with a comparative example of the present invention, the composition of the Al-based hot-dip plating bath can be adjusted in the composition adjusting step as below.
[0062] The B concentration in the Al-based hot-dip plating bath is preferably adjusted by adding an aluminum master alloy containing B. The configuration allows B to be suitably dispersed in the Al-based hot-dip plating bath. The B concentration in the Al-based hot-dip plating bath can alternatively be adjusted by, for example, adding B alone or adding a boride such as aluminum boride (AlB.sub.2 or AlB.sub.12), and a method for adjusting the B concentration is not limited to any particular method. The Al-based hot-dip plating bath that contains such a raw material needs to be subjected to a treatment for uniformly dispersing B in the Al-based hot-dip plating bath.
[0063] Similarly, the K concentration in the Al-based hot-dip plating bath is preferably adjusted by adding an aluminum master alloy containing K. The configuration allows K to be suitably dispersed in the Al-based hot-dip plating bath. The K concentration in the Al-based hot-dip plating bath can alternatively be adjusted by, for example, adding K alone or adding a compound such as KF, KBF.sub.4, or K.sub.2AlF.sub.6AlB.sub.2, and a method for adjusting the K concentration is not limited to any particular method. The Al-based hot-dip plating bath that contains such a raw material needs to be subjected to a treatment for uniformly dispersing K in the Al-based hot-dip plating bath.
[0064] The B concentration and the K concentration in the Al-based hot-dip plating bath are more preferably adjusted by adding an aluminum master alloy containing B and K. The configuration allows B and K to be easily and suitably dispersed in the Al-based hot-dip plating bath by adding such an aluminum master alloy.
Embodiment 1
[0065] The following description will discuss an embodiment of the present invention. Note that (i) a hot-dip Al-based metal-plated steel sheet in accordance with an embodiment of the present invention and (ii) a method for producing the hot-dip Al-based metal-plated steel sheet have configurations similar to the configurations of the comparative example (described earlier) unless otherwise stated. Note that, unless otherwise specified, the present invention is not limited to the following description, which is provided so that subject matters of the present invention are better understood. Note also that a numerical expression such as "A to B" as used herein means "not less than A and not more than B".
[0066] The following description will schematically discuss knowledge of the present invention before discussing (i) a hot-dip Al-based metal-plated steel sheet in accordance with an embodiment of the present invention and (ii) a method for producing the hot-dip Al-based metal-plated steel sheet.
[0067] (Schematic Description of Knowledge of Present Invention)
[0068] As described earlier, a spangle pattern formed due to dendrites commonly appears on a surface of a hot-dip Al-based metal-plated layer. Various approaches have been taken so that a hot-dip Al-based metal-plated steel sheet that has spangles having a minute size and thus has a surface skin having an inconspicuous spangle pattern is produced. Note, however, that a conventional method unfortunately causes an increase in production cost because the conventional method requires, for example, a major apparatus. Alternatively, a conventional method unfortunately makes it difficult to stably form fine spangles.
[0069] The inventors of the present invention carried out diligent study and finally found new knowledge, as described as the comparative example, that a hot-dip Al-based metal-plated steel sheet obtained with use of an Al-based hot-dip plating bath containing both boron (B) and potassium (K) in proper amounts exhibits a remarkable effect of providing finer spangles.
[0070] It is still unclear specifically by what mechanism a plating bath containing both B and K enhances an effect of providing finer spangles. Note, however, that, as compared with a plating bath containing B or aluminum boride alone, a plating bath containing both B and K even in very small amounts clearly brings about a better effect of providing finer spangles. It has been known that B is concentrated on (unevenly distributed over) a surface of a plated layer. Note, however, that a plating bath containing B alone is insufficient to bring about an effect of providing finer spangles. In view of this, examples of the mechanism by which a plating bath containing both B and K enhances the effect of providing finer spangles include a mechanism in which B and K form clusters and the clusters are unevenly distributed over a surface of a plated layer so as to each serve as a spangle nucleus.
[0071] In accordance with the knowledge, the inventors of the present invention tried to continuously produce (continuously operate) the hot-dip Al-based metal-plated steel sheet in accordance with the comparative example so that the hot-dip Al-based metal-plated steel sheet would be practically used in a plating line in a factory. A result of this shows that a phenomenon in which a black foreign substance adheres to (is wound onto) a sink roll, provided in a plating bath, after a certain period of time (e.g., not less than 6 hours, though depending on a production condition) has passed since continuous production of the hot-dip Al-based metal-plated steel sheet in accordance with the comparative example was started. Then, the black foreign substance also adhered to (was also transcribed into) the hot-dip Al-based metal-plated steel sheet in the plating bath. This caused a plating defect.
[0072] That is, the above result shows the following. Specifically, relatively short time production of the hot-dip Al-based metal-plated steel sheet in accordance with the comparative example does not cause any problem. In contrast, long time continuous production of the hot-dip Al-based metal-plated steel sheet causes the problem described earlier. This makes it difficult to stably produce the hot-dip Al-based metal-plated steel sheet in accordance with the comparative example in continuous operation.
[0073] The following description will briefly discuss the sink roll with reference to FIG. 2. FIG. 2 has cross-sectional views schematically illustrating an aluminum pot 4 and a pre-melt pot 6 each provided in plating equipment for continuous production of a hot-dip Al-based metal-plated steel sheet.
[0074] As illustrated in the drawing on the right of (a) of FIG. 2, a substrate steel sheet 1, which comes from annealing equipment (not illustrated), passes through a snout 2 in the form of a tube and then is dipped into an Al-based hot-dip plating bath 3 while being isolated from the outside air. In the Al-based hot-dip plating bath, a plurality of sink rolls 5 are provided. The plurality of sink rolls 5 guide the substrate steel sheet 1 so that the substrate steel sheet 1 passes through the Al-based hot-dip plating bath 3. The number of the plurality of sink rolls 5 is not limited to any particular number. It is a problem that a black foreign substance adheres to such a sink roll 5.
[0075] The present invention has been made in view of the problems, and a further object of the present invention is to provide a method for producing a hot-dip Al-based metal-plated steel sheet, the method making it possible to continuously produce a hot-dip Al-based metal-plated steel sheet including a plated layer having a surface on which fine spangles are stably formed.
[0076] In order to solve the problems, the inventors of the present invention carried out a detailed investigation (e.g., a review of various production processes (production conditions) and an analysis of a product) concerning a cause of production of the black foreign substance during long time continuous production of the hot-dip Al-based metal-plated steel sheet. Then, the inventors of the present invention found (i) the cause of production of the black foreign substance and (ii) a method for removing a contaminant serving as that cause. Finally, the inventors of the present invention accomplished the present invention. This will be more specifically described below.
[0077] First, it was confirmed that (i) the black foreign substance that adheres to the sink roll contains, as a main component, impurities (e.g., alumina, aluminum carbide, and graphite carbon) that are unavoidably present in a plating bath, and (ii) a K concentration in the black foreign substance is relatively higher than in the plating bath.
[0078] During production of the hot-dip Al-based metal-plated steel sheet of the comparative example, K was added to a plating bath with use of an aluminum master alloy containing B and K. The aluminum master alloy will be described below.
[0079] A commercially-available B-containing aluminum master alloy (e.g., an Al-4 mass % B master alloy) is commonly produced with use of KBF.sub.4 so that Al contains B in a form of AlB.sub.2 or AlB.sub.12. Specifically, in a case where molten Al, to which KBF.sub.4 is added, continues to be stirred at a predetermined temperature, B in the form of AlB.sub.2 or AlB.sub.12 is incorporated into the molten Al. In this case, a compound that consists mainly of KAlF.sub.4 or K.sub.3AlF.sub.6 is produced as a by-product. Such a compound thus produced has a lower specific gravity than molten Al and thus easily floats up to a bath surface of the molten Al. For convenience, KBF.sub.4, KAlF.sub.4, and K.sub.3AlF.sub.6 as used herein are hereinafter collectively referred to as flux.
[0080] After the molten Al is caused to contain B, a suspended matter (can also be referred to as a "floating matter") present on a surface of the molten Al is removed. Thus, for example, an Al-4 mass % B master alloy containing B at a concentration of 4 mass % is produced. Note here that the flux, most of which can be removed by separating a bath surface suspended matter from the molten Al, is difficult to completely remove. This causes the flux to be mixed in the Al--B master alloy. The flux that is mixed in the Al--B master alloy is flux in which a B concentration is approximately 0.2 weight % in terms of a K concentration per 4 mass %.
[0081] The inventors of the present invention confirmed that a black foreign substance is also produced in a case where not the Al--B master alloy but KBF.sub.4, KAlF.sub.4, and K.sub.3AlF.sub.6 each serving as the flux are experimentally added to the plating bath. It is unclear by what mechanism the black foreign substance is produced during long time continuous production of the hot-dip Al-based metal-plated steel sheet. However, since the black foreign substance contains K in a large amount, it is possible to say the following.
[0082] Specifically, it is considered that the flux causes the impurities (e.g., alumina, aluminum carbide, and graphite carbon) that are unavoidably present in the plating bath to (i) agglomerate so as to be made bulky and (ii) adhere to the sink roll in a form of a black foreign substance. Thus, it was suggested that production of the black foreign substance during long time continuous production of the hot-dip Al-based metal-plated steel sheet can be prevented or reduced by reducing a K concentration in the plating bath, i.e., removing, in advance, not only the black foreign substance but also the flux that is mixed in the plating bath.
[0083] In view of the above, the inventors of the present invention studied a method for removing the flux that is mixed in the plating bath. As a result, the inventors of the present invention found that agglomerates that have been made bulky, such as graphite carbon, an aluminum oxide, and an Al--B--C-based compound can be floated up to a surface of the plating bath by stirring the plating bath so that gas is drawn in into the plating bath. The agglomerates are considered to float up to the surface of the plating bath due to oxidization of the impurities or adhesion of the impurities to air bubbles.
[0084] In a case where such bath surface suspended matters are removed, an aluminum plating bath in which an amount of flux is reduced can be obtained. It was found that use of such an aluminum plating bath makes it possible to (i) reduce production of a black foreign substance during long time continuous production of the hot-dip Al-based metal-plated steel sheet, (ii) prevent adhesion of a foreign substance to a sink roll that is dipped in the aluminum plating bath, and (iii) prevent or reduce a plating defect caused by adhesion of a foreign substance to a plated steel sheet surface.
[0085] The above description has schematically discussed knowledge of the present invention. The following description will discuss a method for producing a hot-dip Al-based metal-plated steel sheet in accordance with an embodiment of the present invention.
[0086] <Method for Producing Hot-Dip Al-Based Metal-Plated Steel Sheet>
[0087] A method for producing a hot-dip Al-based metal-plated steel sheet in accordance with an embodiment of the present invention includes: a composition adjusting step of adding a B-containing master alloy so as to adjust a composition of an Al-based hot-dip plating bath containing aluminum as a main component, the composition being adjusted so that the Al-based hot-dip plating bath has a B concentration of not less than 0.005 mass % and a K concentration of more than 0 mass % and less than 0.0005 mass %; and a plating step of dipping a substrate steel sheet in the Al-based hot-dip plating bath, which has the composition thus adjusted, so as to cause the substrate steel sheet to pass through the Al-based hot-dip plating bath.
[0088] (Composition Adjusting Step)
[0089] Before the substrate steel sheet is dipped in the Al-based hot-dip plating bath so as to pass through the Al-based hot-dip plating bath, the composition adjusting step of adjusting a composition of the Al-based hot-dip plating bath by adjusting respective concentrations of elements contained in the Al-based hot-dip plating bath.
[0090] In a case where a B concentration in the Al-based hot-dip plating bath is adjusted by adding a B-containing master alloy, the Al-based hot-dip plating bath can have a B concentration of not less than 0.005 mass % (a boron concentration adjusting step). Examples of the B-containing master alloy include an Al--B alloy, an alloy of titanium and B (Ti--B alloy), and an alloy of nickel and B (Ni--B alloy). In a case where KBF.sub.4 is used in a process for producing the B-containing master alloy, flux containing K is mixed in the B-containing master alloy.
[0091] Thus, in a case where the B concentration in the Al-based hot-dip plating bath is adjusted by adding a master alloy containing B and flux, the K concentration in the Al-based hot-dip plating bath inevitably increases. In a case where such an Al-based hot-dip plating bath is used to continuously produce the hot-dip Al-based metal-plated steel sheet for a long time, a black foreign substance may be produced on a sink roll.
[0092] In view of the above, in the composition adjusting step of Embodiment 1, agglomerates that have been made bulky, such as graphite carbon, an aluminum oxide, and an Al--B--C-based compound are floated up to a surface of the Al-based hot-dip plating bath by supplying gas into the Al-based hot-dip plating bath by stirring the Al-based hot-dip plating bath so that gas is drawn in into the Al-based hot-dip plating bath. Such bath surface suspended matters include a flux component that is mixed in the Al-based hot-dip plating bath.
[0093] By removing such bath surface suspended matters, it is possible to reduce an amount of flux contained in the Al-based hot-dip plating bath. That is, it is possible to (i) remove K contained in the Al-based hot-dip plating bath and (ii) reduce the K concentration in the Al-based hot-dip plating bath to more than 0 mass % and less than 0.0005 mass % (a potassium concentration adjusting step). A treatment for producing and removing a bath surface suspended matter may be hereinafter referred to as a "flux removal treatment".
[0094] A method for stirring the Al-based hot-dip plating bath so that gas is drawn in into the Al-based hot-dip plating bath is not limited to any particular method. Examples of the method include (i) a method in which gas such as atmosphere is blown (bubbled) into the Al-based hot-dip plating bath and (ii) a method in which a stirring mechanism is used to stir the Al-based hot-dip plating bath on or near a surface of the Al-based hot-dip plating bath.
[0095] A specific mode of the stirring mechanism is not particularly limited. For example, the stirring mechanism can be a stirring machine including a stirring blade, such as a propeller stirrer, or can be a drive roll including a rotation part that is rotatable on an axis by external power. In a case where the drive roll is used, gas can be supplied into the Al-based hot-dip plating bath by dipping a part of the rotation part in the Al-based hot-dip plating bath (causing a part of the drive roll to be exposed from a surface of the Al-based hot-dip plating bath) so as to rotate the rotation part. Note that the Al-based hot-dip plating bath can be stirred with use of a combination of a plurality of stirring mechanisms.
[0096] A time for which to stir the Al-based hot-dip plating bath (flux removal treatment time) can be set to, for example, 0.5 h to 5 h. Note, however, that the flux removal treatment time is not particularly limited provided that a bath surface suspended matter can be produced and removed during the flux removal treatment time. Note also that the flux removal treatment can be separately carried out a plurality of times. In this case, the flux removal treatment time for each of the plurality of times may be shorter.
[0097] The following description will discuss, with reference to FIG. 2, a case where the above flux removal treatment of the composition adjusting step of Embodiment 1 is carried out in plating equipment provided in, for example, a factory. FIG. 2 has cross-sectional views schematically illustrating the aluminum pot 4 and the pre-melt pot 6 each provided in the plating equipment for continuous production of the hot-dip Al-based metal-plated steel sheet.
[0098] (a) of FIG. 2 illustrates a state in which an Al-based hot-dip plating bath 3a in the pre-melt pot 6 is being stirred with use of a propeller stirrer 7. (b) of FIG. 2 illustrates a state in which the Al-based hot-dip plating bath 3a in the pre-melt pot 6 is being stirred by blowing gas. Note that a specific mode of the plating equipment except a mode to be discussed in the following description is not particularly limited and is therefore neither illustrated nor described.
[0099] In the plating equipment, the pre-melt pot 6 ordinarily may be provided near the aluminum pot 4. An aluminum ingot and a master alloy are melted in the pre-melt pot 6, so that the Al-based hot-dip plating bath 3 to be supplied to the aluminum pot 4 is produced.
[0100] According to the flux removal treatment of Embodiment 1, a pretreatment Al-based hot-dip plating bath 3a is prepared first. The pretreatment Al-based hot-dip plating bath 3a (i) contains flux and (ii) has a composition that has been adjusted in the pre-melt pot 6 with use of, for example, an Al-4 mass % B master alloy. Then, the pretreatment Al-based hot-dip plating bath 3a is stirred in the pre-melt pot 6 so that gas (e.g., atmosphere) is drawn in into the pretreatment Al-based hot-dip plating bath 3a.
[0101] Specifically, as illustrated in (a) of FIG. 2, the propeller stirrer 7 is used to stir the pretreatment Al-based hot-dip plating bath 3a on or near a surface of the pretreatment Al-based hot-dip plating bath 3a. Alternatively, as illustrated in (b) of FIG. 2, a pipe 8 that is dipped in the pretreatment Al-based hot-dip plating bath 3a is used to blow gas into the pretreatment Al-based hot-dip plating bath 3a so as to stir the pretreatment Al-based hot-dip plating bath 3a.
[0102] Note that, though not illustrated here, the pretreatment Al-based hot-dip plating bath 3a can be alternatively configured to be stirred with use of a drive roll 22 (described later, see FIG. 3).
[0103] Then, a suspended matter on the surface of the pretreatment Al-based hot-dip plating bath 3a is removed, so that the Al-based hot-dip plating bath 3 that has been subjected to the flux removal treatment and has a lower K concentration is obtained. The Al-based hot-dip plating bath 3 is supplied to the aluminum pot 4.
[0104] The Al-based hot-dip plating bath 3 that has been subjected to the flux removal treatment carried out in the pre-melt pot 6 can be temporarily cooled and solidified so as be a solid substance (ingot), and the solid substance can be melted in the pre-melt pot 6 so as to be used, or can be fed into the aluminum pot 4.
[0105] The flux removal treatment does not necessarily need to be carried out in the pre-melt pot 6 that is provided near the aluminum pot 4. Alternatively, the Al-based hot-dip plating bath 3 can be produced, at a place distant from the aluminum pot 4, by carrying out the flux removal treatment in advance, the Al-based hot-dip plating bath 3 can be cooled and solidified so as be an ingot, and the ingot can be used.
[0106] The flux removal treatment can be alternatively carried out in the aluminum pot 4. In this case, the aluminum pot 4 can (i) include a mechanism for suppling gas into a plating bath and (ii) be configured to allow a bath surface suspended matter to be removed. It is possible to carry out the flux removal treatment while or before causing the substrate steel sheet 1 to pass through the Al-based hot-dip plating bath 3 in the aluminum pot 4.
[0107] FIG. 1 is an optical photomicrograph of a state in which an outermost surface of a hot-dip Al-based metal-plated steel sheet in accordance with Embodiment 1 has been polished so that a dendrite structure is made observable. As illustrated in FIG. 1, dendrites having grown from spangle crystal nuclei are present on a surface of a hot-dip Al-based metal-plated layer. In a case where a B concentration and a K concentration in an Al-based hot-dip plating bath fall within the respective specified ranges (described earlier), a hot-dip Al-based metal-plated steel sheet produced with use of the Al-based hot-dip plating bath can have not less than 100 spangle crystal nuclei that are present per square centimeter surface area of a plated layer.
[0108] Furthermore, use of the Al-based hot-dip plating bath allows long time continuous production of a hot-dip Al-based metal-plated steel sheet. This makes it possible to prevent or reduce occurrence of a phenomenon in which a black foreign substance adheres to a sink roll.
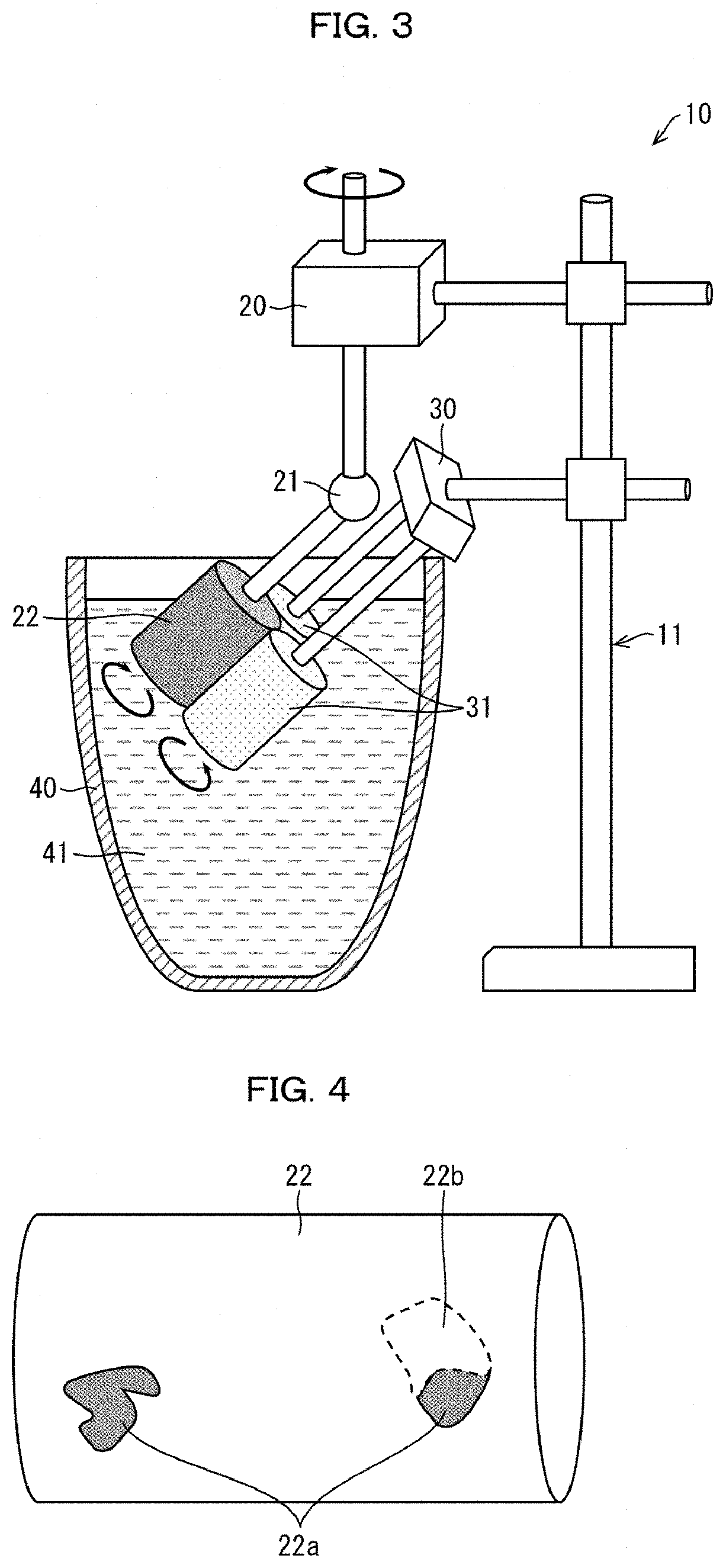
[0109] Thus, a method for producing a hot-dip Al-based metal-plated steel sheet in accordance with Embodiment 1 makes it possible to continuously produce a hot-dip Al-based metal-plated steel sheet including a plated layer having a surface on which fine spangles are stably formed.
[0110] Note here that, in a case where an Al-based hot-dip plating bath has a B concentration of less than 0.005 mass %, a hot-dip Al-based metal-plated steel sheet produced with use of the Al-based hot-dip plating bath cannot bring about a good effect of providing finer spangles. In contrast, in a case where an Al-based hot-dip plating bath has a B concentration of more than 0.50 mass %, an effect, brought about by a hot-dip Al-based metal-plated steel sheet produced with use of the Al-based hot-dip plating bath, of providing finer spangles is saturated. Thus, an increase in average B concentration in an Al-based hot-dip plating bath to more than 0.50 mass % does not achieve superiority in effect, brought about by a hot-dip Al-based metal-plated steel sheet produced with use of the Al-based hot-dip plating bath, of providing finer spangles.
[0111] In a case where a B concentration in an Al-based hot-dip plating bath increases to some extent, an effect, brought about by a hot-dip Al-based metal-plated steel sheet produced with use of the Al-based hot-dip plating bath, of providing finer spangles is saturated. It is therefore unnecessary in an aspect of the present invention to set an upper limit of the B concentration.
[0112] Note that a hot-dip Al-based metal-plated layer that has an average B concentration of more than 2.0% may cause a deterioration in corrosion resistance. Thus, an Al-based hot-dip plating bath preferably has a B concentration of 0.005 mass % to 2.0 mass % from the viewpoint of corrosion resistance of a hot-dip Al-based metal-plated steel sheet.
[0113] In a case where an Al-based hot-dip plating bath in which an amount of flux contained therein has been reduced has a K concentration of not less than 0.0005 mass % and the Al-based hot-dip plating bath is used to continuously produce a hot-dip Al-based metal-plated steel sheet for a long time, a phenomenon in which a black foreign substance adheres to a sink roll. This may cause a plating defect to be produced in the hot-dip Al-based metal-plated steel sheet thus produced.
[0114] An Al-based hot-dip plating bath preferably has a B concentration of not less than 0.02 mass % and not more than 2.0 mass % and a K concentration of more than 0 mass % and less than 0.0005 mass %. With the configuration, a hot-dip Al-based metal-plated steel sheet produced with use of the Al-based hot-dip plating bath can have not less than 300 spangle crystal nuclei that are present per square centimeter surface area of a plated layer. This makes it possible to produce a hot-dip Al-based metal-plated steel sheet having a surface that is more beautiful in appearance.
[0115] In the composition adjusting step, a composition of the Al-based hot-dip plating bath, containing Al as a main component and containing at least B and K, can be adjusted so that the Al-based hot-dip plating bath contains an element(s) different from Al, B, and K. Specifically, as in the case of the comparative example (described earlier), an element(s) such as Si, Fe, Sr, Na, Ca, Sb, P, Mg, Cr, Mn, Ti, Zr, and/or V can be intentionally added to the Al-based hot-dip plating bath as necessary.
[0116] The balance of components of the hot-dip Al-based metal-plated layer which balance is different from the above elements can be constituted by Al and unavoidable impurities.
[0117] (Plating Step)
[0118] An average concentration of each component of a hot-dip Al-plated layer which average concentration is obtained after the plating step is substantially identical to a composition of the Al-based hot-dip plating bath. Thus, the configuration makes it possible to produce a hot-dip Al-based metal-plated steel sheet including a hot-dip Al-based metal-plated layer that has an average B concentration of not less than 0.005 mass % and an average K concentration of more than 0 mass % to less than 0.00005 mass %.
[0119] The present invention is not limited to the embodiments, but can be altered by a skilled person in the art within the scope of the claims. The present invention also encompasses, in its technical scope, any embodiment derived by combining technical means disclosed in differing embodiments.
Example 1
[0120] An Al-20 mass % Si master alloy was used to adjust an Si concentration in an aluminum plating bath to 9 mass %. Then, an Al-4 mass % B master alloy was added, in a predetermined amount, to the aluminum plating bath so that a B concentration in the aluminum plating bath was adjusted to 0% to 1%. Table 1 shows a result obtained by carrying out a chemical analysis with respect to a composition of the Al-4 mass % B master alloy added. Each figure shown in Table 1 indicates mass %.
TABLE-US-00001 TABLE 1 Chemical composition (mass %) B Si Fe K 4.2 0.13 0.16 0.20
[0121] Assuming that Fe coming from, for example, a substrate steel sheet and a member of a pot would be unavoidably mixed in the aluminum plating bath during continuous production, cast iron that was made of a material of which the pot was made was melted so that an Fe concentration in the aluminum plating bath was adjusted to 2.0 mass %.
[0122] By thus adjusting the composition, Al-based hot-dip plating baths indicated by plating bath Nos. 1 to 10 shown in Table 2 were produced. Note that B and K are present in the Al-4 mass % B master alloy at a constant ratio. Thus, for each of the plating bath Nos. 2, 3, and 4, in each of which a K concentration in a plating bath is lower than a lower limit of an analytical limit, a K concentration value found by ratio calculation is shown. Note that no Al-4 mass % B master alloy was added to the plating bath No. 1.
TABLE-US-00002 TABLE 2 Plating Plating bath composition (mass %) bath No. Si B K Fe 1 9.0 0 0 2.0 2 9.0 0.004 0.00019* 2.0 3 9.0 0.005 0.00024* 2.0 4 9.0 0.010 0.00048* 2.0 5 9.0 0.015 0.00071 2.0 6 9.0 0.020 0.00095 2.0 7 9.0 0.030 0.0014 2.0 8 9.0 0.10 0.0048 2.0 9 9.0 0.50 0.024 2.0 10 9.0 1.0 0.048 2.0 *Calculated value
[0123] (Analysis of Components of Al-Based Hot-Dip Plating Bath by ICP)
[0124] Note here that the following description will discuss a method, carried out in Example 1, of a quantitative analysis of components contained in an Al-based hot-dip plating bath.
[0125] A plating bath piece obtained by cooling and solidifying a part of an Al-based hot-dip plating bath was melted, by heating, in a mixed acid (mixed solution of 40 ml of nitric acid and 10 ml of hydrochloric acid). Thereafter, ultrapure water was added so that a resultant solution had a fixed volume. A solution that had been obtained from the plating bath and whose volume had been fixed was regarded as a solution for use in a quantitative analysis of the components contained in the Al-based hot-dip plating bath (hereinafter referred to as a "quantitative analysis solution"). Thereafter, a composition of the components contained in the Al-based hot-dip plating bath was found by carrying out the following two types of quantitative analyses with respect to the quantitative analysis solution.
[0126] A quantitative analysis of Si, B, and Fe was carried out by inductively coupled plasma atomic emission spectroscopy (ICP-AE). A quantitative analysis of K was carried out by inductively coupled plasma mass spectrometry (ICP-MS).
[0127] Respective compositions of the Al-based hot-dip plating baths to be discussed in the following description were similarly found by carrying out such a quantitative analysis as described above.
[0128] (Flux Removal Treatment)
[0129] Each of the Al-based hot-dip plating baths shown in Table 2 was used to carry out a flux removal treatment. Specifically, gas was supplied into an Al-based hot-dip plating bath, whose amount was set to 23 kg, so that a bath surface suspended matter was produced so as to be removed. Gas was supplied into the Al-based hot-dip plating bath by carrying out the following methods (i) to (iii).
[0130] (i) Gas Bubbling [0131] Gas: Atmosphere [0132] Gas flow rate: 3 L/min [0133] Diameter of pipe dipped in plating bath: Inner diameter of 2 mm
[0134] (ii) Propeller Stirring [0135] Rotational speed of propeller: 500 rpm [0136] Shape of propeller: Including three rotary vanes and having a diameter of 50 mm
[0137] (iii) Rotation of Dipped Roll Part of which is Exposed from Bath Surface [0138] Dimension of roll: 60 mm in diameter.times.100 mm in length [0139] Rotational speed of roll: 130 rpm
[0140] Table 3 collectively shows (i) the Al-based hot-dip plating baths each of which had been subjected to the flux removal treatment and (ii) Al-based hot-dip plating baths none of which had been subjected to the flux removal treatment.
TABLE-US-00003 TABLE 3 Before flux removal treatment Plating Plating bath composition (mass %) bath No. B K Example of 3 0.005 0.00024* present 4 0.010 0.00048* invention 5 0.015 0.00071 0.015 0.00071 0.015 0.00071 6 0.020 0.00095 7 0.030 0.0014 8 0.10 0.0048 9 0.50 0.024 10 1.0 0.048 Comparative 1 0 0 Example 2 0.004 0.00019* 5 0.015 0.00071 5 0.015 0.00071 5 0.015 0.00071 6 0.020 0.00095 7 0.030 0.0014 8 0.10 0.0048 9 0.50 0.025 10 1.0 0.050 Flux removal treatment Treatment B K Plating Treatment time concentration concentration bath No. method (h) (mass %) (mass %) Example of 3 Propeller stirring 1 0.005 tr present 4 Roll rotation 1 0.010 tr invention 5 Gas bubbling 0.5 0.015 tr 1 0.015 tr 5 0.015 tr Propeller stirring 0.5 0.015 tr 1 0.015 tr 5 0.015 tr Roll rotation 0.5 0.015 tr 1 0.015 tr 5 0.015 tr 6 Gas bubbling 0.5 0.020 tr 1 0.020 tr 5 0.020 tr 7 Propeller stirring 1 0.030 tr 8 Roll rotation 1 0.10 tr 9 Gas bubbling 1 0.50 tr 10 Propeller stirring 1 1.0 tr Comparative 1 None -- 0 0 Example 2 None -- 0.004 0.00019* 5 None -- 0.015 0.00071 5 None -- 0.015 0.00071 5 None -- 0.015 0.00071 6 None -- 0.020 0.00095 7 None -- 0.030 0.0014 8 None -- 0.10 0.0048 9 None -- 0.50 0.025 10 None -- 1.0 0.050 *calculated value
[0141] The plating bath Nos. 3 to 10, each of which had been subjected to the flux removal treatment, each had a K concentration that was reduced to an amount as low as a trace amount (indicated by "tr" in Table 3). In contrast, it is revealed that none of the plating bath Nos. 3 to 10 changed in B concentration.
[0142] (Production of Hot-Dip Al-Based Metal-Plated Steel Sheet and Number of Spangle Crystal Nuclei on Plated Layer Surface)
[0143] (i) The Al-based hot-dip plating baths each of which had been subjected to the flux removal treatment and (ii) the Al-based hot-dip plating baths none of which had been subjected to the flux removal treatment were used to produce hot-dip Al-based metal-plated steel sheets as below.
[0144] A cold rolled annealed steel sheet having a thickness of 0.8 mm and a chemical composition shown in Table 4 was regarded as a substrate steel sheet.
TABLE-US-00004 TABLE 4 Chemical composition (mass %) C Si Mn P S Al O N 0.033 <0.01 0.23 <0.01 0.013 0.01 0.0027 0.0025
[0145] Plating experimental equipment was used to produce a hot-dip Al-based metal-plated steel sheet (material under test) by (i) dipping the substrate steel sheet in the Al-based hot-dip plating bath, (ii) pulling up the substrate steel sheet thus dipped, and (iii) solidifying a plated layer at a predetermined cooling rate. Table 5 shows conditions under which an aluminum-plated steel sheet was produced.
TABLE-US-00005 TABLE 5 Plating bath temperature 650.degree. C.-680.degree. C. Plating bath dipping time 2 sec Cooling rate 13.degree. C./s Amount of adhered plating per one side Approximately 20 .mu.m
[0146] A hot-dip Al-based metal-plated steel sheet obtained was subjected to the following investigation.
[0147] A surface of each material under test was buffed, and an outermost layer ranging from a surface of a plated layer to a depth of 5 .mu.m was smoothed, so that a dendrite structure was made observable. Then, an optical microscope was used to calculate the number of spangle crystal nuclei that are present per square centimeter surface area of the plated layer. A surface appearance was evaluated by the following criterion, and "Very good" and "Good" were each regarded as "passed".
[0148] Very good: Not less than 200 spangle crystal nuclei were present per square centimeter surface area of the plated layer.
[0149] Good: Not less than 100 and less than 200 spangle crystal nuclei were present per square centimeter surface area of the plated layer.
[0150] Poor: Not less than 50 and less than 100 spangle crystal nuclei were present per square centimeter surface area of the plated layer.
[0151] Very poor: Less than 50 spangle crystal nuclei were present per square centimeter surface area of the plated layer.
[0152] (Method for Determining Black Foreign Substance Long Time Roll Retention During Long Time Continuous Production)
[0153] The Al-based hot-dip plating baths were each tested on black foreign substance roll retention during long time continuous production. It is possible to easily determine, with use of the testing device (see FIG. 3) and by the following evaluation method, whether a foreign substance adheres to (is wound onto) a sink roll in a plating bath during long time continuous production of a hot-dip Al-based metal-plated steel sheet.
[0154] FIG. 3 is a view schematically illustrating a testing device 10 for carrying out a black foreign substance roll retention test with respect to a plating bath for use in production of a hot-dip Al-based metal-plated steel sheet. FIG. 3 illustrates a plating pot in a form of a cross-sectional view so as to intelligibly show a structure of the testing device.
[0155] The testing device 10 illustrated in FIG. 3 was used to determine whether an impurity (black foreign substance) is wound onto a sink roll during long time continuous production.
[0156] As illustrated in FIG. 3, the testing device 10 includes a support 11, a motor 20 supported by the support 11, and a fixture 30 supported by the support 11. The motor 20 is connected with the drive roll 22 via a universal joint 21 so as to rotate the drive roll 22.
[0157] The fixture 30 pivotally supports two non-drive rolls 31 each of which is rotatable. The two non-drive rolls 31 and the drive roll 22 are provided side by side so as to be in contact with each other. In a case where the drive roll 22 rotates, the two non-drive rolls 31 also rotate in response to rotation of the drive roll 22.
[0158] A roll bundle of the two non-drive rolls 31 and the drive roll 22 were dipped in an Al-based hot-dip plating bath 41, stored in a plating pot 40, so as to incline to a surface of the Al-based hot-dip plating bath 41.
[0159] The above roll bundle was dipped in each of Al-based hot-dip plating baths 41 having respective various compositions so that the drive roll 22 was rotated for a predetermined time under conditions shown in Table 6. Then, the drive roll 22 was pulled up.
TABLE-US-00006 TABLE 6 Bath temperature 680.degree. C. Testing time 6 h Circumferential velocity of roll 130 rpm Material of roll S55C Dimension of roll .PHI.60 mm .times. 100 mm
[0160] A surface state of the drive roll 22 having been pulled up was observed so that it was determined whether a black foreign substance had been wound onto the drive roll 22.
[0161] FIG. 4 is a plan view illustrating the surface state of the drive roll 22 to which a black foreign substance has adhered. In the drive roll 22, cases may occur where (i) a black foreign substance 22a adheres to the drive roll 22 so as to be visually recognizable and (ii) the black foreign substance 22a that is provided with a swollen part is observed below an adhered bath 22b in a case where the adhered bath 22b is broken, the adhered bath 22b having adhered to a surface of the swollen part so as to be like a skin (see FIG. 4).
[0162] The Al-based hot-dip plating baths having the respective compositions were each tested on black foreign substance roll retention with use of the testing device 10 so that an area of an adhered black foreign substance per 180 cm.sup.2 surface area was measured on a surface of the drive roll 22 pulled up. Evaluation was carried out by the following criteria, and a case where an area of an adhered black foreign substance on the surface of the drive roll 22 was less than 1 cm.sup.2 ("Good" in "Dipped roll" column in Table 6) was regarded as "passed". Note that an area of an adhered black foreign substance includes an area of the black foreign substance 22a having adhered below the adhered bath 22b.
[0163] A black foreign substance may adhere to each of the drive roll 22 and the non-drive rolls 31. In view of this, a measurement of an area of an adhered black foreign substance per 180 cm.sup.2 surface area can be alternatively carried out with respect to the non-drive rolls 31.
[0164] Table 7 collectively shows results of the test described earlier. Note that base plating bath Nos. shown in Table 7 correspond to the respective plating bath Nos. shown in Table 3.
TABLE-US-00007 TABLE 7 Base Before flux removal treatment plating Plating bath composition (mass %) No. bath No. B K Example of 1 3 0.005 0.00024* present 2 4 0.010 0.00048* invention 3 5 0.015 0.00071 4 5 6 0.015 0.00071 7 8 9 0.015 0.00071 10 11 12 6 0.020 0.00095 13 14 15 7 0.030 0.0014 16 8 0.10 0.0048 17 9 0.50 0.024 18 10 1.0 0.048 Comparative 19 1 0 0 Example 20 2 0.004 0.00019 21 5 0.015 0.00071 22 5 0.015 0.00071 23 5 0.015 0.00071 24 6 0.020 0.00095 25 7 0.030 0.0014 26 8 0.10 0.0048 27 9 0.50 0.04 28 10 1.0 48 Flux removal treatment Base Treatment B K plating Treatment time concentration concentration No. bath No. method (h) (mass %) (mass %) Example of 1 3 Propeller stirring 1 0.005 tr present 2 4 Roll rotation 1 0.010 tr invention 3 5 Gas bubbling 0.5 0.015 tr 4 1 0.015 tr 5 5 0.015 tr 6 Propeller stirring 0.5 0.015 tr 7 1 0.015 tr 8 5 0.015 tr 9 Roll rotation 0.5 0.015 tr 10 1 0.015 tr 11 5 0.015 tr 12 6 Gas bubbling 0.5 0.020 tr 13 1 0.020 tr 14 5 0.020 tr 15 7 Propeller stirring 1 0.030 tr 16 8 Roll rotation 1 0.10 tr 17 9 Gas bubbling 1 0.50 tr 18 10 Propeller stirring 1 1.0 tr Comparative 19 1 None -- 0 0 Example 20 2 None -- 0.004 0.00019* 21 5 None -- 0.015 0.00071 22 5 None -- 0.015 0.00071 23 5 None -- 0.015 0.00071 24 6 None -- 0.020 0.00095 25 7 None -- 0.030 0.0014 26 8 None -- 0.10 0.0048 27 9 None -- 0.50 0.025 28 10 None -- 1.0 0.050 Base Number of Grade of plating spangle crystal surface No. bath No. Dipped roll nuclei (per cm.sup.2) appearance Example of 1 3 Good 120 Good present 2 4 Good 150 Good invention 3 5 Good 240 Very good 4 Good 230 Very good 5 Good 220 Very good 6 Good 230 Very good 7 Good 220 Very good 8 Good 220 Very good 9 Good 250 Very good 10 Good 230 Very good 11 Good 210 Very good 12 6 Good 320 Very good 13 Good 320 Very good 14 Good 350 Very good 15 7 Good Not less than 400 Very good 16 8 Good Not less than 400 Very good 17 9 Good Not less than 400 Very good 18 10 Good Not less than 400 Very good Comparative 19 1 Good 2 Very poor Example 20 2 Good 10 Very poor 21 5 Poor 200 Very good 22 5 Poor 200 Very good 23 5 Poor 200 Very good 24 6 Poor 300 Very good 25 7 Poor Not less than 400 Very good 26 8 Poor Not less than 400 Very good 27 9 Poor Not less than 400 Very good 28 10 Poor Not less than 400 Very good *calculated value
[0165] As shown in Example Nos. 1 to 18 of the present invention, in Example in which a B concentration in the Al-based hot-dip plating bath falls within the scope of the present invention, not less than 100 spangle crystal nuclei were present per square centimeter surface area of an aluminum plated layer of a hot-dip Al-based metal-plated steel sheet produced, and a good effect of providing finer spangles was exhibited.
[0166] Furthermore, in each of Example Nos. 1 to 18 of the present invention, a K concentration of less than 0.0005% was obtained due to the flux removal treatment, and no black foreign substance was wound onto the drive roll 22. This shows that it is possible in long time continuous production to (i) reduce production of a black foreign substance and (ii) prevent adhesion of a foreign substance to a sink roll.
[0167] That is, a hot-dip Al-based metal-plated steel sheet including a plated layer having a surface on which fine spangles are stably formed can be continuously produced by using any of the Al-based hot-dip plating baths indicated by Example Nos. 1 to 18 of the present invention and each subjected to the flux removal treatment.
[0168] In contrast, in each of (i) Comparative Example No. 19, in which no Al-4 mass % B master alloy was added, and (ii) Comparative Example No. 20, in which an Al-4 mass % B master alloy was added in a small amount, a K concentration in the plating bath falls within the scope of the present invention, whereas a B concentration in the plating bath falls outside the scope of the present invention. Thus, no black foreign substance was wound onto the drive roll 22, but fewer (e.g., not more than 10) spangle crystal nuclei were present per square centimeter surface area of an aluminum plated layer of a hot-dip Al-based metal-plated steel sheet produced, so that a less good effect of providing finer spangles was exhibited.
[0169] Furthermore, in each of Comparative Example Nos. 21 to 28, a B concentration in the plating bath falls within the scope of the present invention, whereas a K concentration in the plating bath falls outside the scope of the present invention. Thus, not less than 200 spangle crystal nuclei were present per square centimeter surface area of an aluminum plated layer of a hot-dip Al-based metal-plated steel sheet produced, and a good effect of providing finer spangles was exhibited, but a black foreign substance was wound onto the drive roll 22.
REFERENCE SIGNS LIST
[0170] 1 Substrate steel sheet [0171] 3 Al-based hot-dip plating bath
* * * * *
D00000
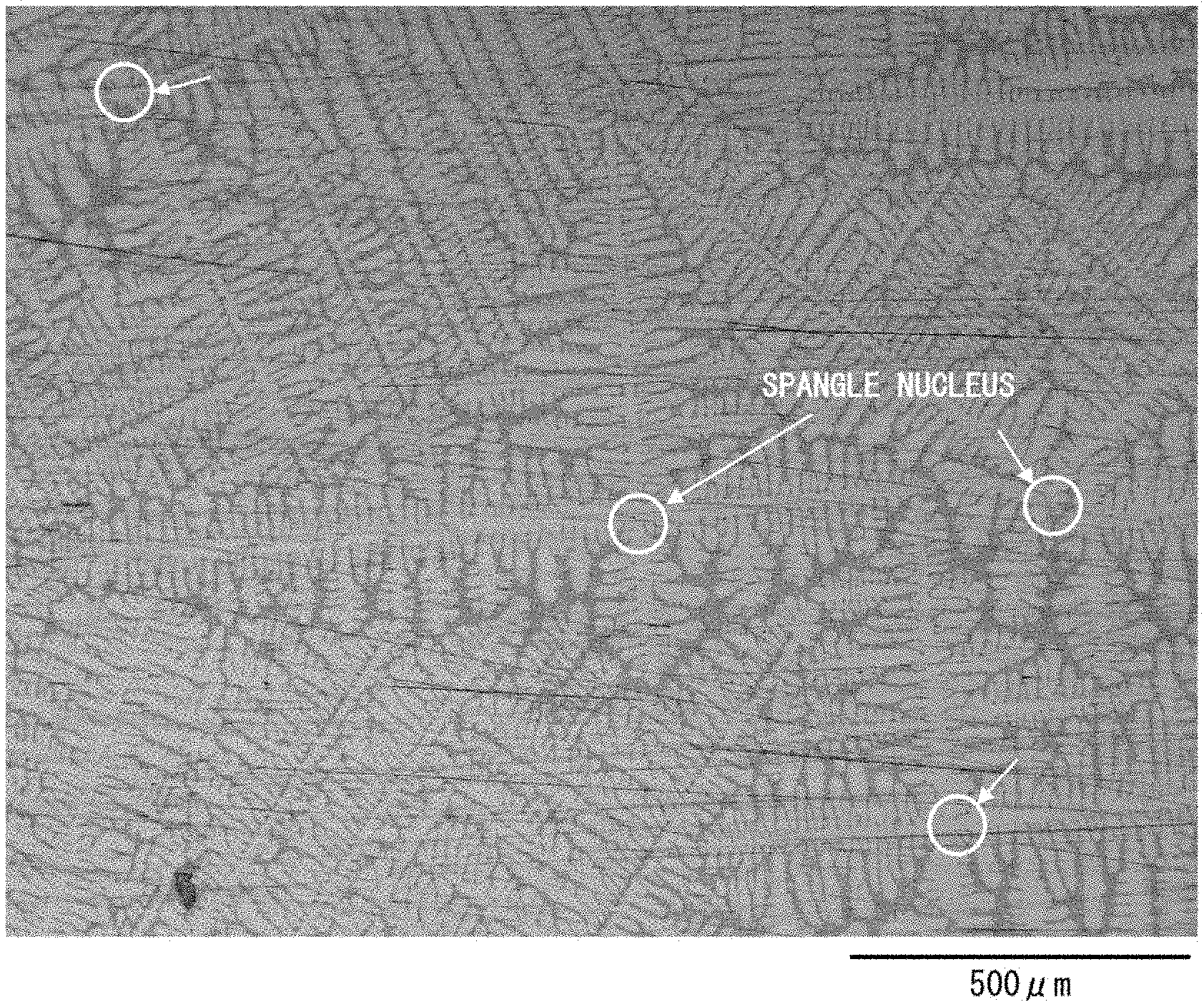
D00001

D00002

D00003

D00004

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.