Sphingosine Pathway Modulating Compounds For The Treatment Of Cancers
Rabbani; Elazar ; et al.
U.S. patent application number 17/027761 was filed with the patent office on 2021-01-07 for sphingosine pathway modulating compounds for the treatment of cancers. This patent application is currently assigned to Enzo Biochem, Inc.. The applicant listed for this patent is Enzo Biochem, Inc.. Invention is credited to James J. Donegan, Elazar Rabbani.
| Application Number | 20210000766 17/027761 |
| Document ID | / |
| Family ID | |
| Filed Date | 2021-01-07 |



View All Diagrams
| United States Patent Application | 20210000766 |
| Kind Code | A1 |
| Rabbani; Elazar ; et al. | January 7, 2021 |
SPHINGOSINE PATHWAY MODULATING COMPOUNDS FOR THE TREATMENT OF CANCERS
Abstract
The invention provides methods and compositions for treating cancers and myeloproliferative disorders using sphingosine kinase-1 inhibitors, such as SK1-I, and selective sphingosine-1-phosphate receptor agonists, such as ozanimod.
| Inventors: | Rabbani; Elazar; (New York, NY) ; Donegan; James J.; (Amesbury, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Enzo Biochem, Inc. New York NY |
||||||||||
| Appl. No.: | 17/027761 | ||||||||||
| Filed: | September 22, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16830357 | Mar 26, 2020 | |||
| 17027761 | ||||
| 16287201 | Feb 27, 2019 | 10646459 | ||
| 16830357 | ||||
| 16017345 | Jun 25, 2018 | 10278960 | ||
| 16287201 | ||||
| 62524221 | Jun 23, 2017 | |||
| Current U.S. Class: | 1/1 |
| International Class: | A61K 31/137 20060101 A61K031/137; A61K 31/4245 20060101 A61K031/4245; A61K 45/06 20060101 A61K045/06; A61K 31/135 20060101 A61K031/135; A61P 35/00 20060101 A61P035/00; A61P 35/02 20060101 A61P035/02 |
Claims
1. A method for treating prostate cancer in a mammalian subject, comprising the step of: administering to a mammalian subject in need of treatment for prostate cancer an effective amount of ozanimod or a pharmaceutically acceptable salt thereof.
2. The method of claim 1, wherein the administering step comprises administering the hydrochloride salt of ozanimod to the mammalian subject.
3. The method of claim 1, wherein the mammalian subject is a human.
4. The method of claim 3, wherein the administering step comprises administering the hydrochloride salt of ozanimod to the mammalian subject.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 16/830,357 filed Mar. 26, 2020, which is a continuation of U.S. patent application Ser. No. 16/287,201 filed Feb. 27, 2019 (now U.S. patent no. 10,646,459), which is a divisional of U.S. patent application Ser. No. 16/017,345 filed June 25, 2018 (now U.S. Pat. No. 10,278,960), which claims the benefit of U.S. provisional patent application Ser. No. 62/524,221 filed Jun. 23, 2017, each of which is hereby incorporated by reference in its entirety.
FIELD OF THE INVENTION
[0002] The invention relates to the field of pharmaceutical treatment of cancers.
BACKGROUND
[0003] Sphingosine-1-phosphate (S1P) was discovered to be a bioactive signaling molecule over 20 years ago. Studies have since identified two related kinases, sphingosine kinase 1 and 2 (a/k/a sphingosine kinase "type I" and "type II" respectively, and SphK1 and SphK2 respectively), which catalyze the phosphorylation of sphingosine to S1P. Extracellular S1P can bind to and activate each of five S1P-specific, G protein-coupled receptors (designated S1PR.sub.1-5) to regulate cellular and physiological processes in an autocrine or paracrine manner. Selective inhibitors of each of sphingosine kinase 1 and 2, as well as both non-selective and selective agonists of S1PRs, have been developed and are known in the art.
SUMMARY
[0004] One embodiment of the invention provides a method for treating liver cancer, such as hepatocellular carcinoma (HCC), in a mammalian subject, such as a human, that includes the step of:
[0005] administering to a mammalian subject in need of treatment for liver cancer, a therapeutically effective amount of a sphingosine kinase type I inhibitor, such as SK1-I or a pharmaceutically acceptable salt thereof.
[0006] A related embodiment of the invention provides a pharmaceutical composition that includes a sphingosine kinase type I inhibitor, such as SK1-I or a pharmaceutically acceptable salt thereof for the treatment of liver cancer, such as HCC, in a mammal, such as a human patient.
[0007] Another embodiment of the invention provides a method for treating a cancer or a myeloproliferative disorder (myeloproliferative neoplasm) in a mammalian subject, such as a human, that includes the step of:
[0008] administering to a mammalian subject in need of treatment for a cancer or myeloproliferative disorder, a therapeutically effective amount of a sphingosine-1-phosphate receptor agonist, such as an agonist of one or both of sphingosine-1-phosphate receptor-1 (S1P.sub.1) and sphingosine-1-phosphate receptor-5 (S1P.sub.5) such as ozanimod (RPC1063) or a pharmaceutically acceptable salt thereof.
[0009] A related embodiment of the invention provides a pharmaceutical composition for the treatment of a cancer or a myeloproliferative disorder (myeloproliferative neoplasm) in a mammalian subject, such as a human, that includes:
[0010] a therapeutically effective amount of a sphingosine-1-phosphate receptor agonist, such as an agonist of one or both of sphingosine-1-phosphate receptor-1 (S1P.sub.1) and sphingosine-1-phosphate receptor-5 (S1P.sub.5) such as ozanimod (RPC1063) or a pharmaceutically acceptable salt thereof.
[0011] Still another embodiment of the invention provides a method for treating a cancer or a myleoproliferative disorder (myeloproliferative neoplasm), such as any of those disclosed herein, in a mammalian subject, such as a human, including the step of:
[0012] co-administering to a mammalian subject in need of treatment for a cancer or myeloproliferative disorder, a therapeutically effective amount of:
[0013] (a) a sphingosine kinase type I inhibitor, such as one disclosed in U.S. Pat. No. 8,372,888 and/or 8,314,151, such as SK1-I, or a pharmaceutically acceptable salt thereof; and
[0014] (b) one or more immune checkpoint inhibitors, which may be monoclonal antibodies, such as one or more selected from the group consisting of: PD-1 inhibitors such as mAbs Pembrolizumab (Keytruda.RTM.) and Nivolumab (Opdivo.RTM.); PD-L1 inhibitors such as mAbs Atezolizumab (Tecentriq.RTM.), Avelumab (Bavencio.RTM.), and Durvalumab (Imfinzi.RTM.); and CTLA-4 inhibitors such as mAb Ipilimumab (Yervoy.RTM.); and V-domain Ig Suppressor of T Cell Activation (VISTA) inhibitors such as mAb JNJ-61610588 (ImmuNext Inc.).
[0015] Additional features, advantages, and embodiments of the invention may be set forth or apparent from consideration of the following detailed description, drawings if any, and claims. Moreover, it is to be understood that both the foregoing summary of the invention and the following detailed description are exemplary and intended to provide further explanation without limiting the scope of the invention as claimed.
BRIEF DESCRIPTION OF THE DRAWINGS
[0016] FIG. 1 shows MTT assay data (72 hours) for various concentrations of ozanimod for four human hepatocellular carcinoma cell lines.
[0017] FIG. 2 shows MTT assay data (72 hours) for various concentrations of ABC294640 for four human hepatocellular carcinoma cell lines.
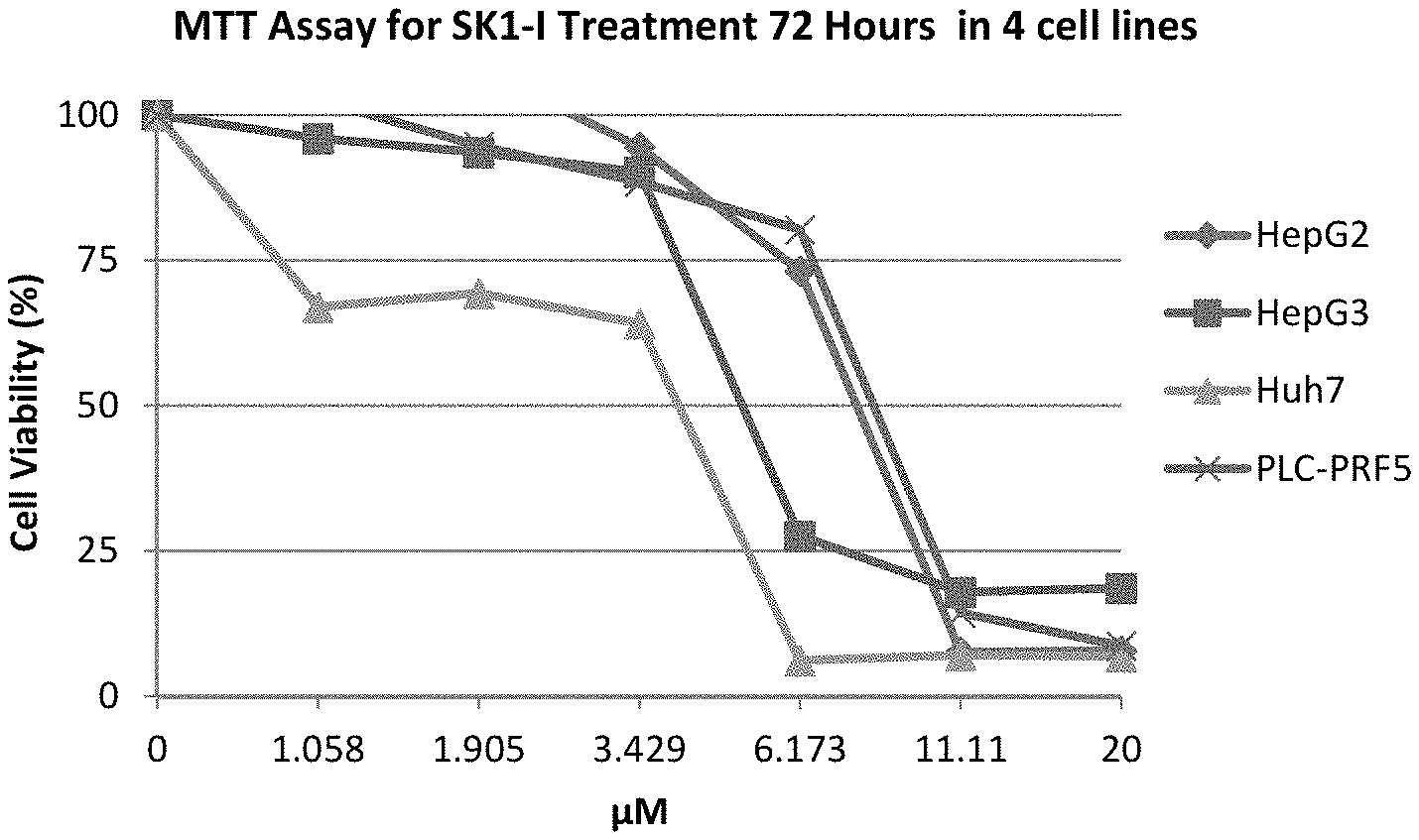
[0018] FIG. 3 shows MTT assay data (72 hours) for various concentrations of SK1-I for four human hepatocellular carcinoma cell lines.
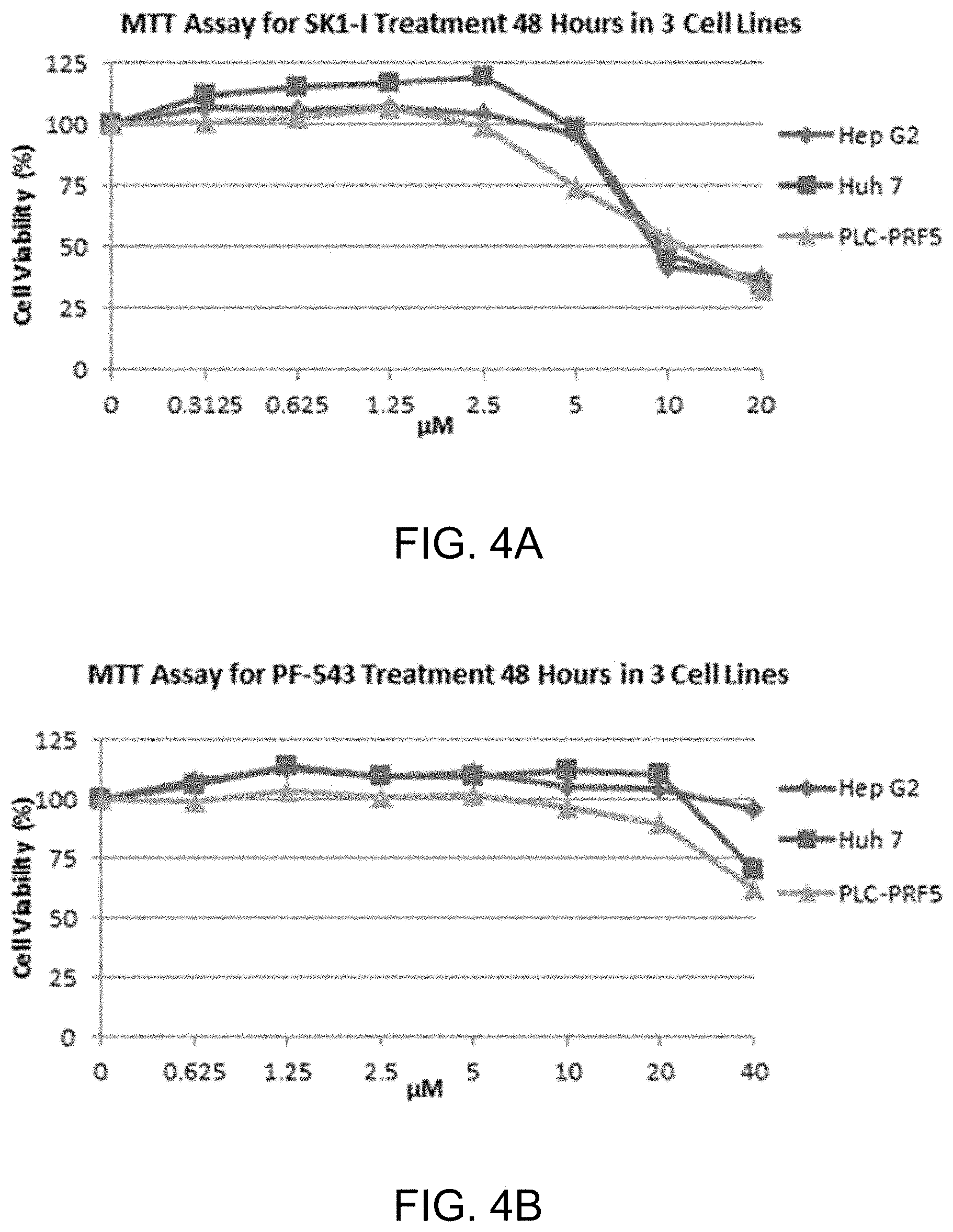
[0019] FIG. 4A shows MTT assay data (48 hours) for various concentrations of SK1-I for three human hepatocellular carcinoma cell lines. FIG. 4B shows MTT assay data (48 hours) for various concentrations of PF-543, a super potent SphK1 inhibitor, for the same three human hepatocellular carcinoma cell lines shown in FIG. 4A.
[0020] FIGS. 5A-D show apoptosis assay data for various concentrations of SK1-I and no-drug control for Huh7 cells. SK1-I strongly induced apoptosis in the Huh7 cells.
[0021] FIGS. 6A-D show apoptosis assay data for various concentrations of SK1-I and no-drug control for PLC-PRF5 cells. SK1-I strongly induced apoptosis in the PLC-PRF5 cells.
[0022] FIGS. 7A-D show apoptosis assay data for various concentrations of SK1-I and no-drug control for Hep 3B cells. SK1-I strongly induced apoptosis in the Hep 3B cells.
[0023] FIG. 8A-D show apoptosis assay data for various concentrations of SK1-I and no-drug control for Hep G2 cells. SK1-I strongly induced apoptosis in the Hep G2 cells.
[0024] FIGS. 9A-D show apoptosis assay data for various concentrations of ozanimod and no-drug control for Huh7 cells. Ozanimod strongly induced apoptosis in the Huh7 cells.
[0025] FIGS. 10A-D show apoptosis assay data for various concentrations of ozanimod and no-drug control for PLC-PRFS cells. Ozanimod strongly induced apoptosis in the PLC-PRFS cells.
[0026] FIGS. 11A-D show apoptosis assay data for various concentrations of ozanimod and no-drug control for Hep 3B cells. Ozanimod strongly induced apoptosis in the Hep 3B cells.
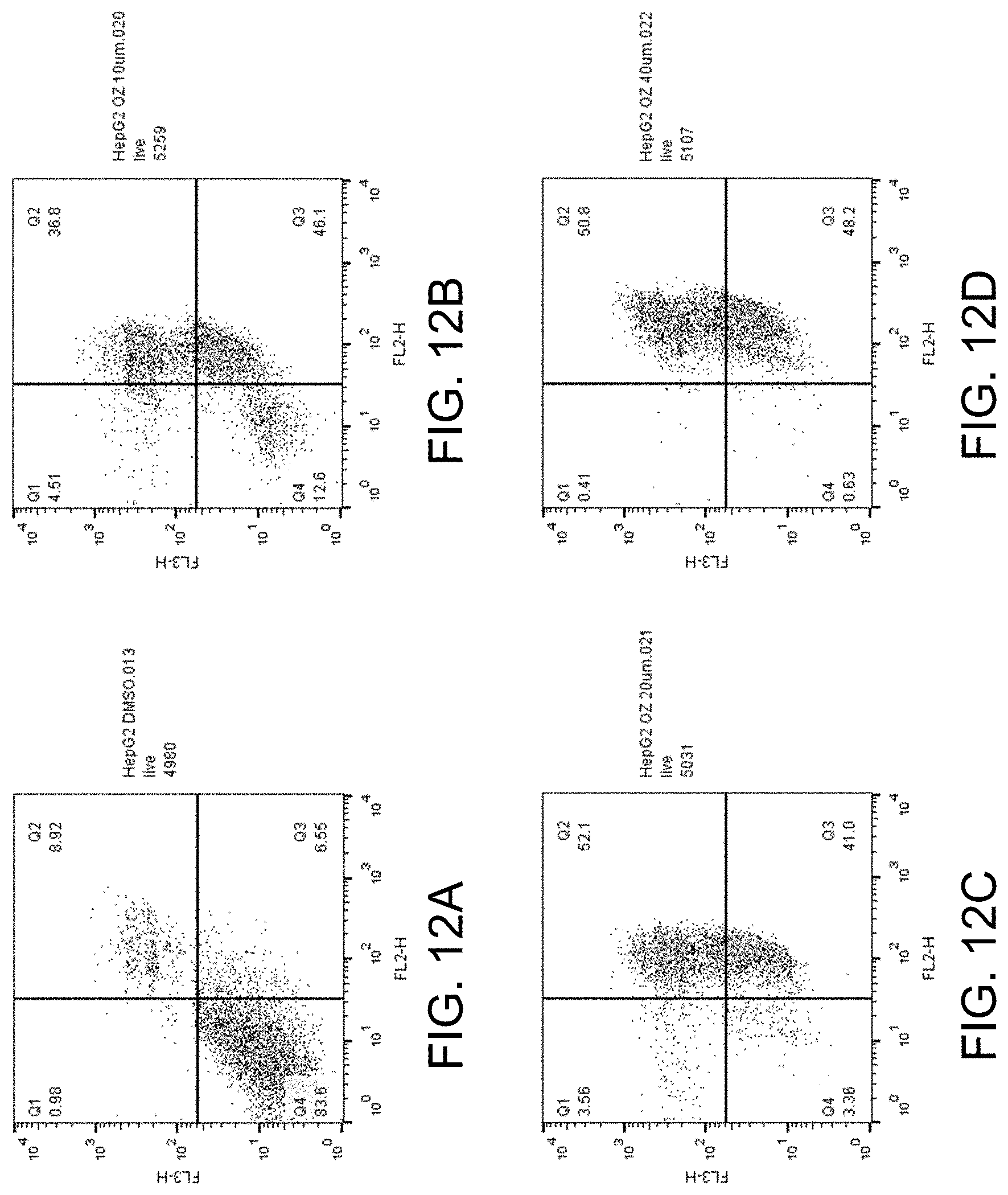
[0027] FIGS. 12A-D show apoptosis assay data for various concentrations of ozanimod and no-drug control for Hep G2 cells. Ozanimod strongly induced apoptosis in the Hep G2 cells.
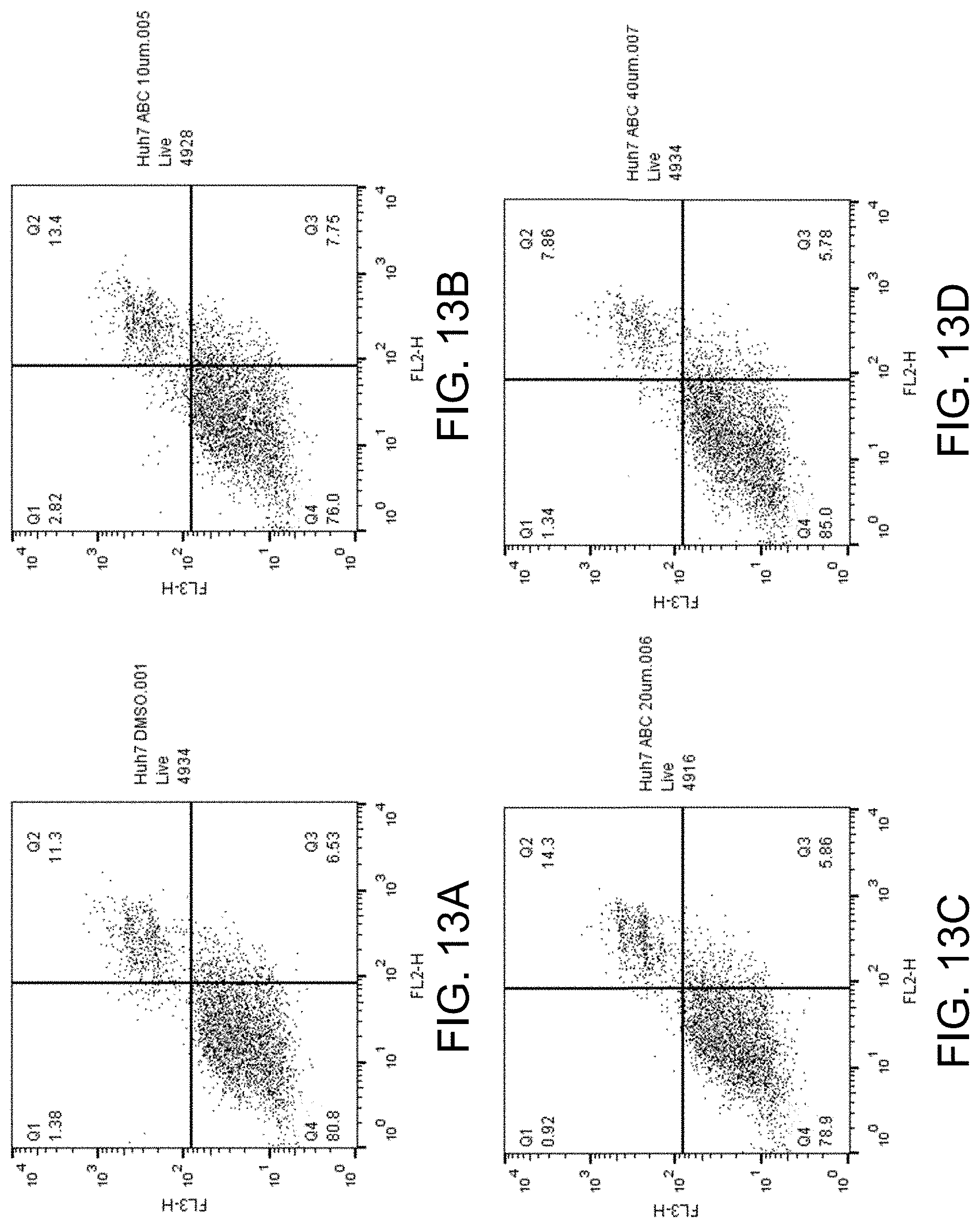
[0028] FIGS. 13A-D shows apoptosis assay data for various concentrations of ABC294640 and no-drug control for Huh7 cells. ABC294640 failed to induce apoptosis in the Huh7 cells.
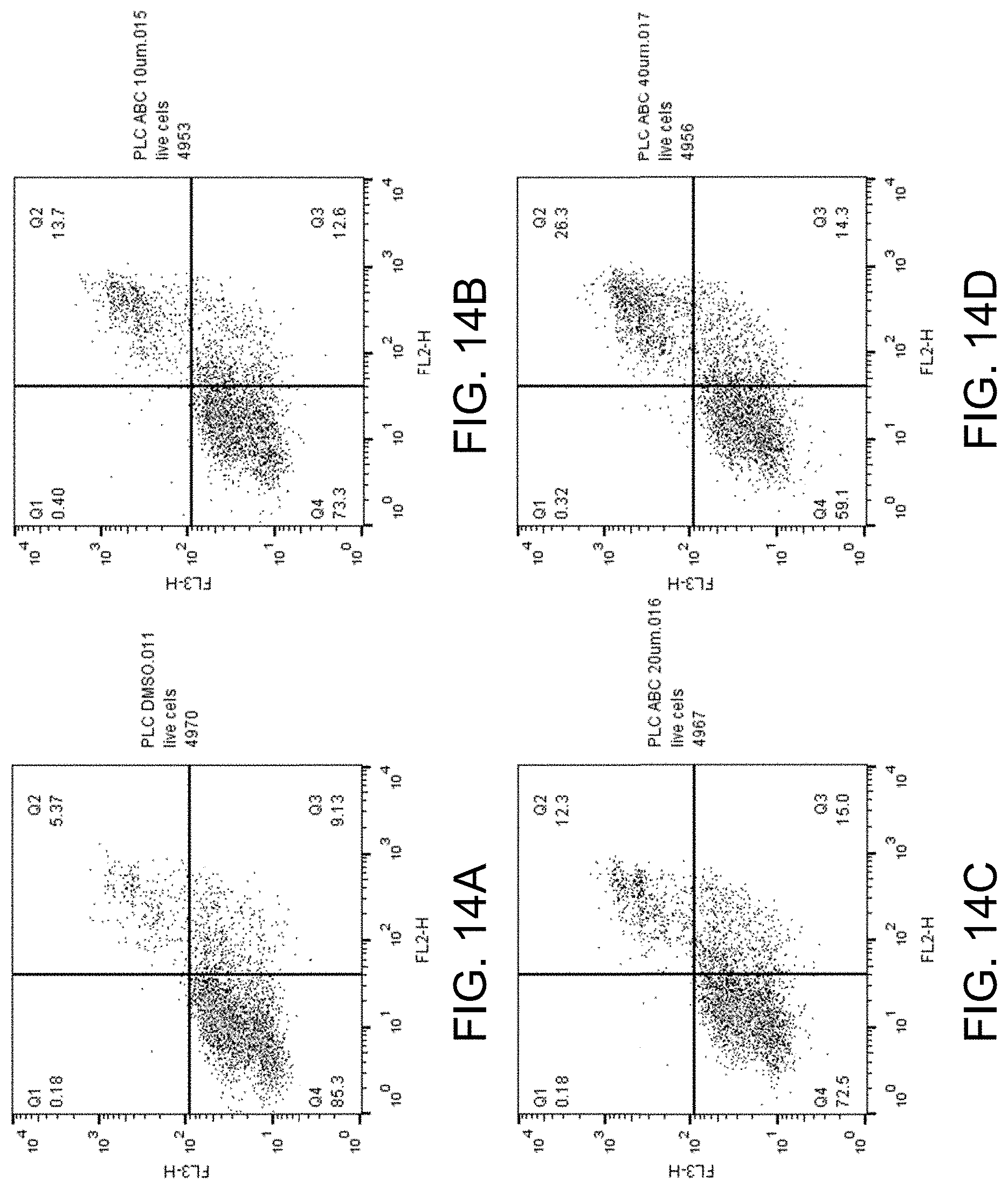
[0029] FIGS. 14A-D show apoptosis assay data for various concentrations of ABC294640 and no-drug control for PLC-PRFS cells. ABC294640 did not substantially induce apoptosis in the PLC-PRFS cells.
[0030] FIGS. 15A-D show apoptosis assay data for various concentrations of ABC294640 and no-drug control for Hep 3B cells. ABC294640 did not substantially induce apoptosis in the Hep 3B cells.
[0031] FIGS. 16A-D show apoptosis assay data for various concentrations of ABC294640 and no-drug control for Hep G2 cells. ABC294640 failed to induce apoptosis in the Hep G2 cells.
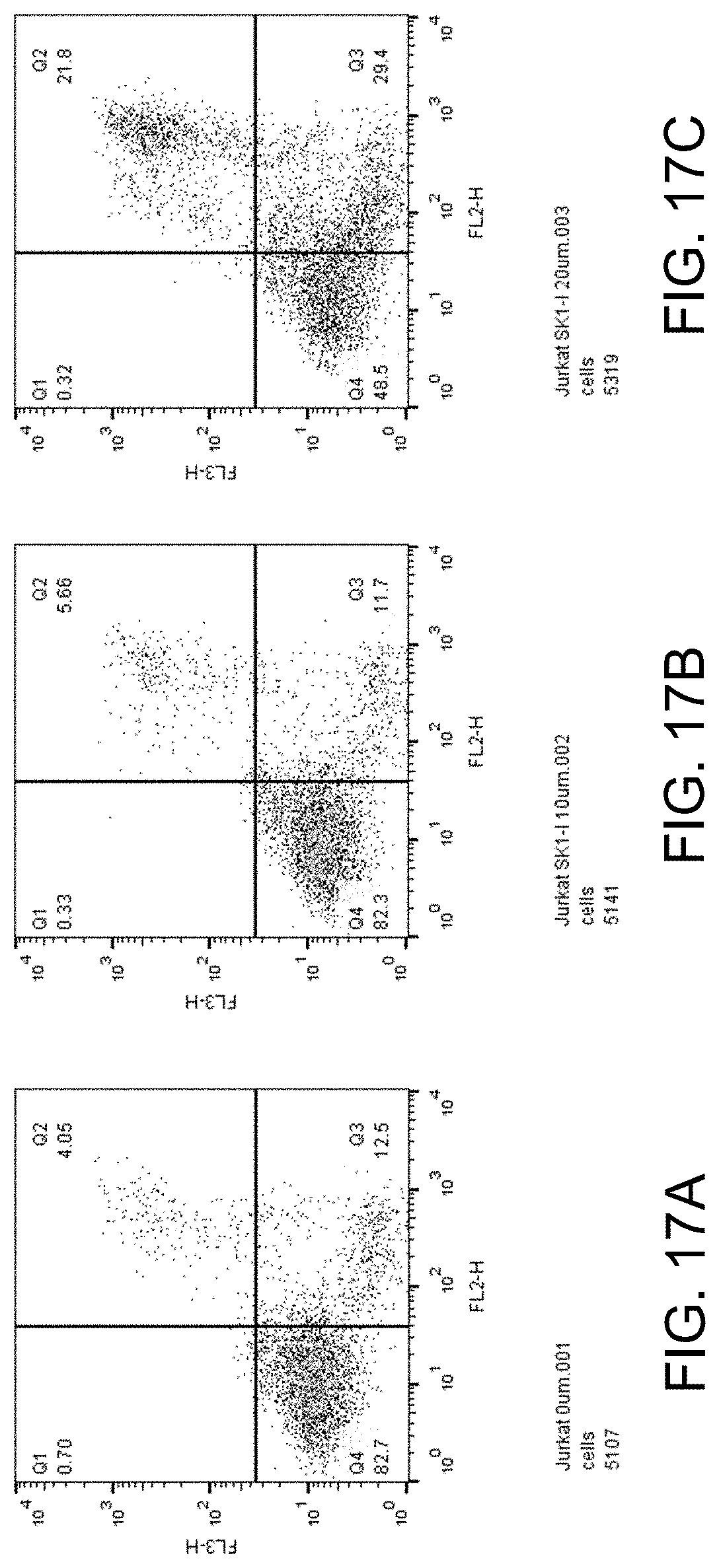
[0032] FIGS. 17A-C show apoptosis assay data for various concentrations of SK1-I and no-drug control for Jurkat cells (human T-cell leukemia cell line). SK1-I strongly induced apoptosis in the Jurkat cells.
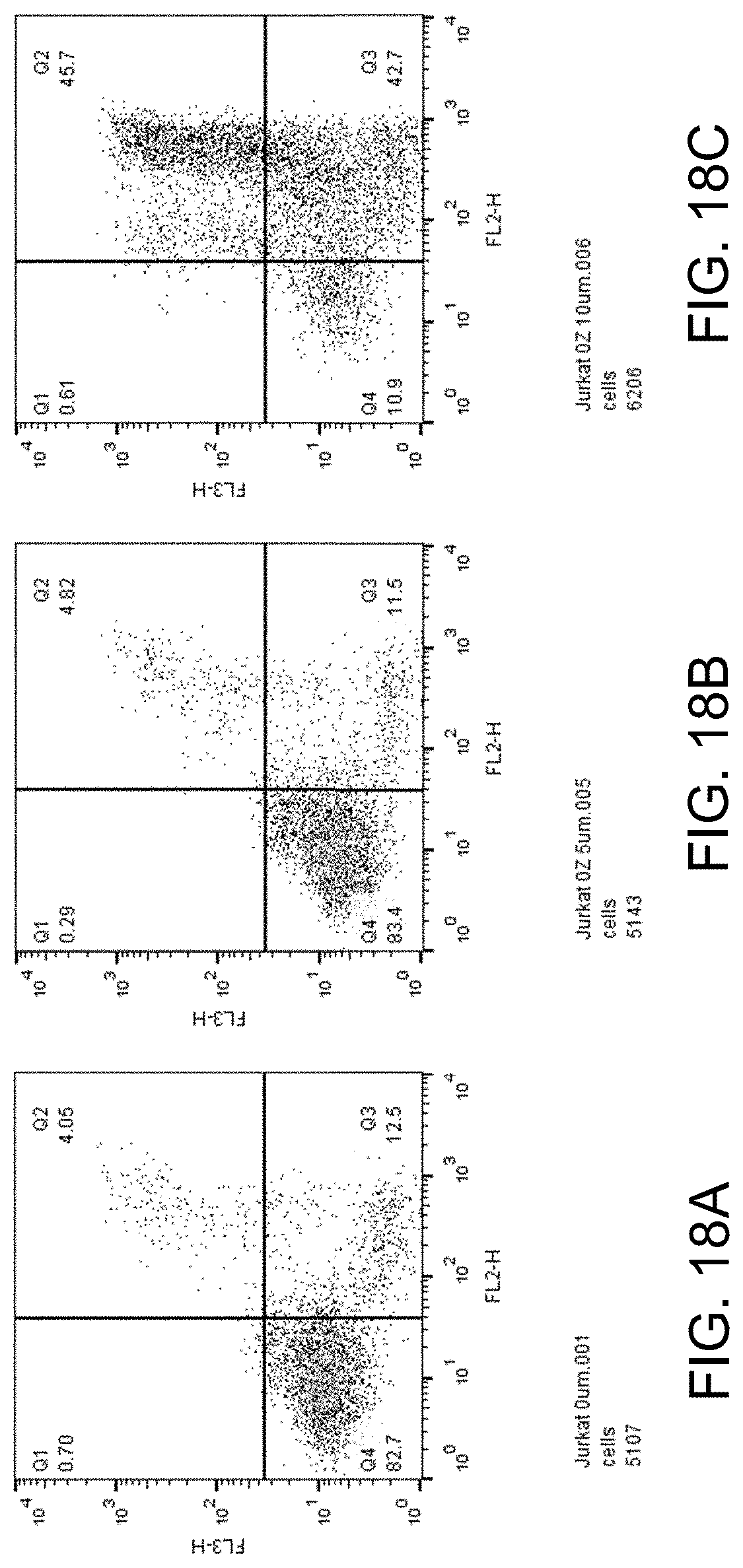
[0033] FIGS. 18A-C show apoptosis assay data for various concentrations of ozanimod and no-drug control for Jurkat cells. Ozanimod strongly induced apoptosis in the Jurkat cells.
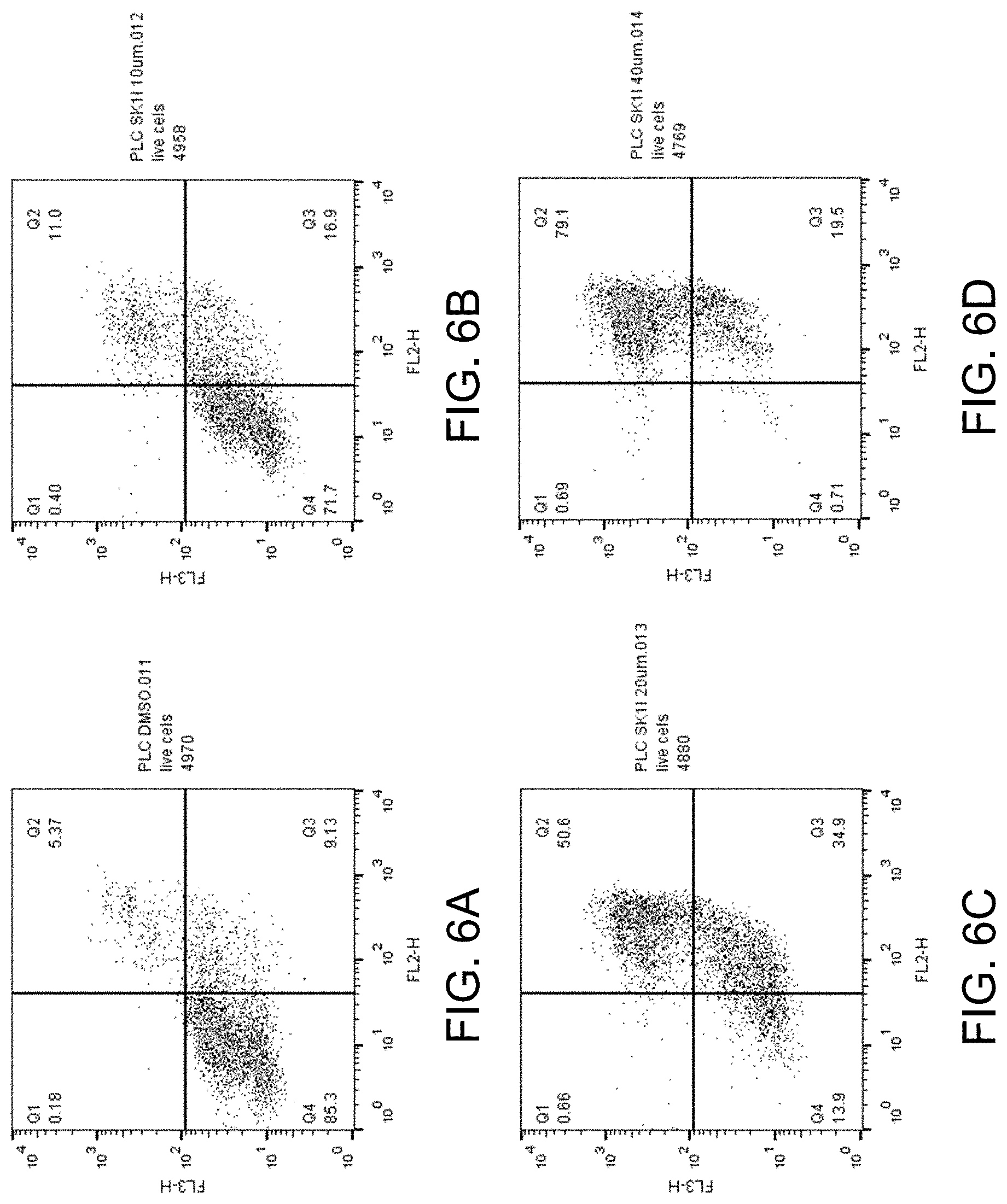
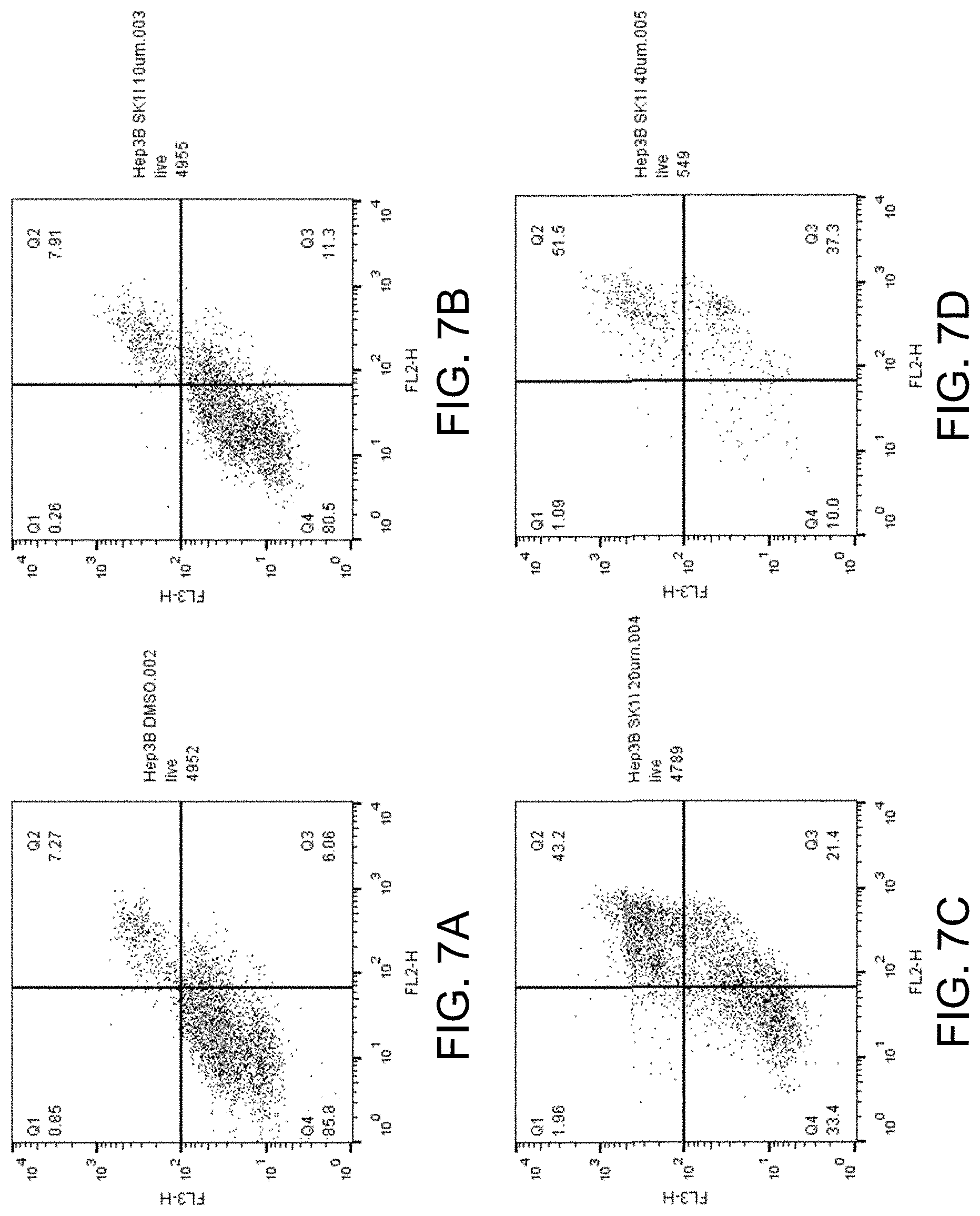
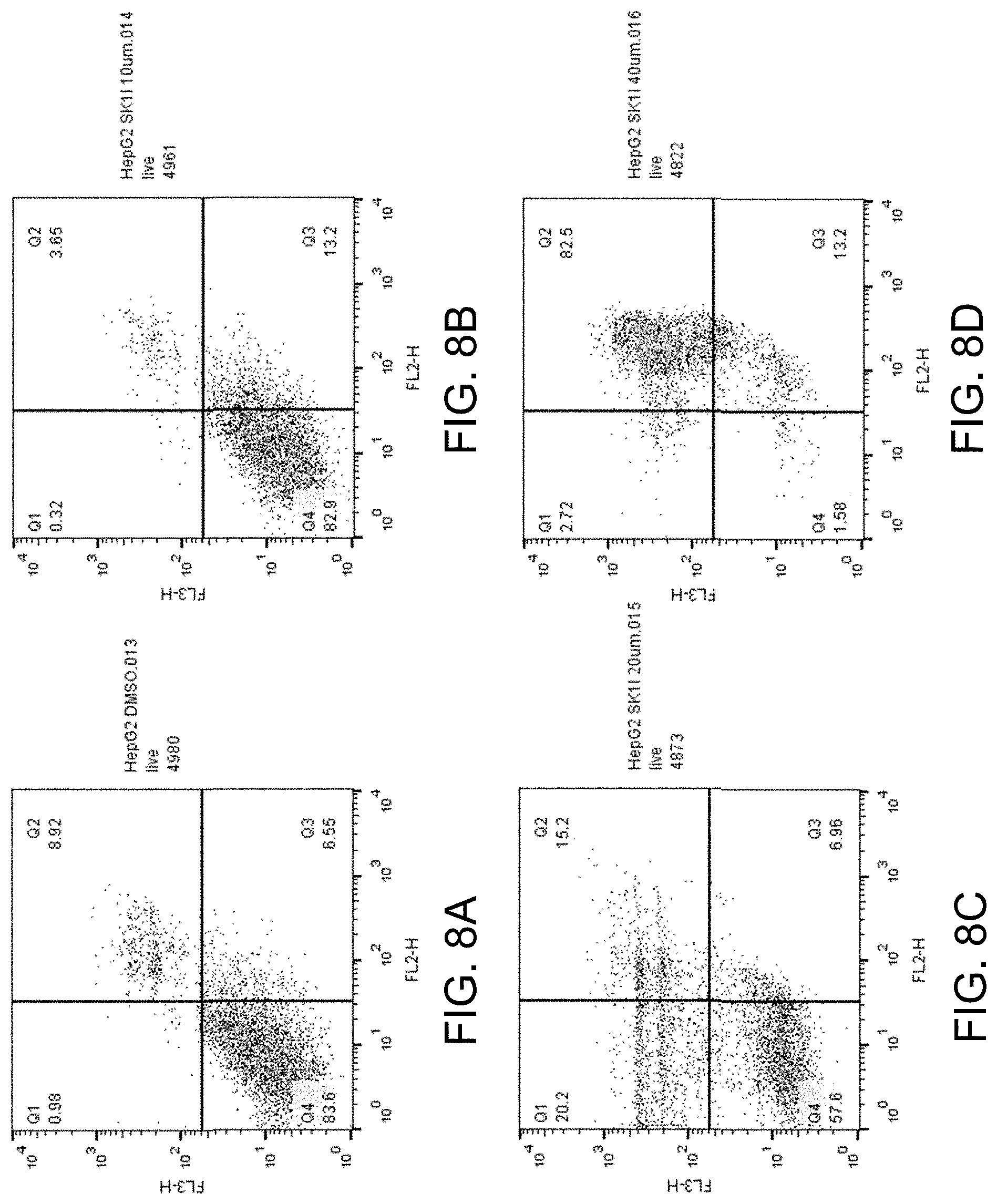
[0034] FIGS. 19A-D show apoptosis assay data for various concentrations of SK1-I and no-drug control for primary human hepatocytes. SK1-I did not induce apoptosis of the primary human hepatocytes at any concentration tested.
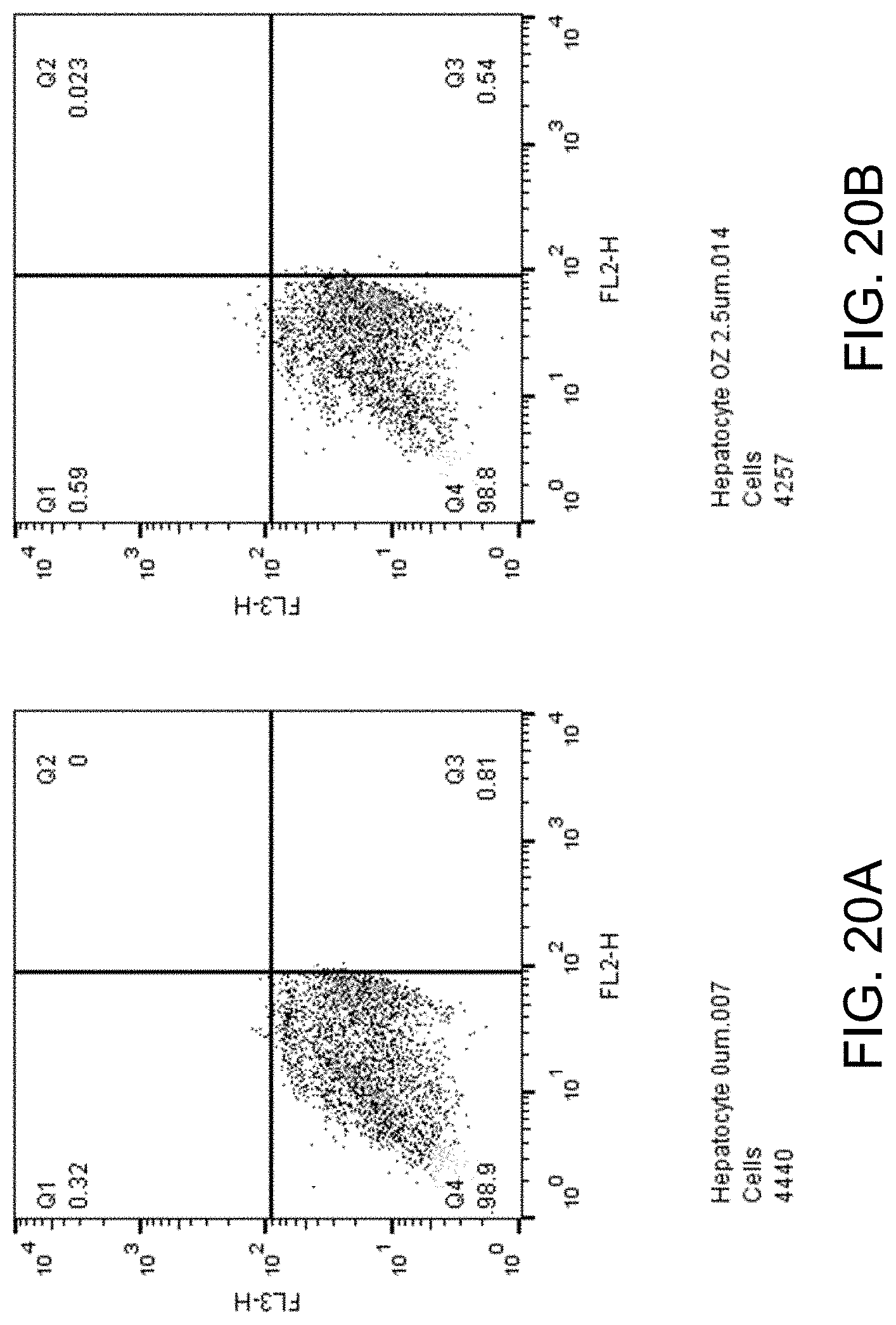
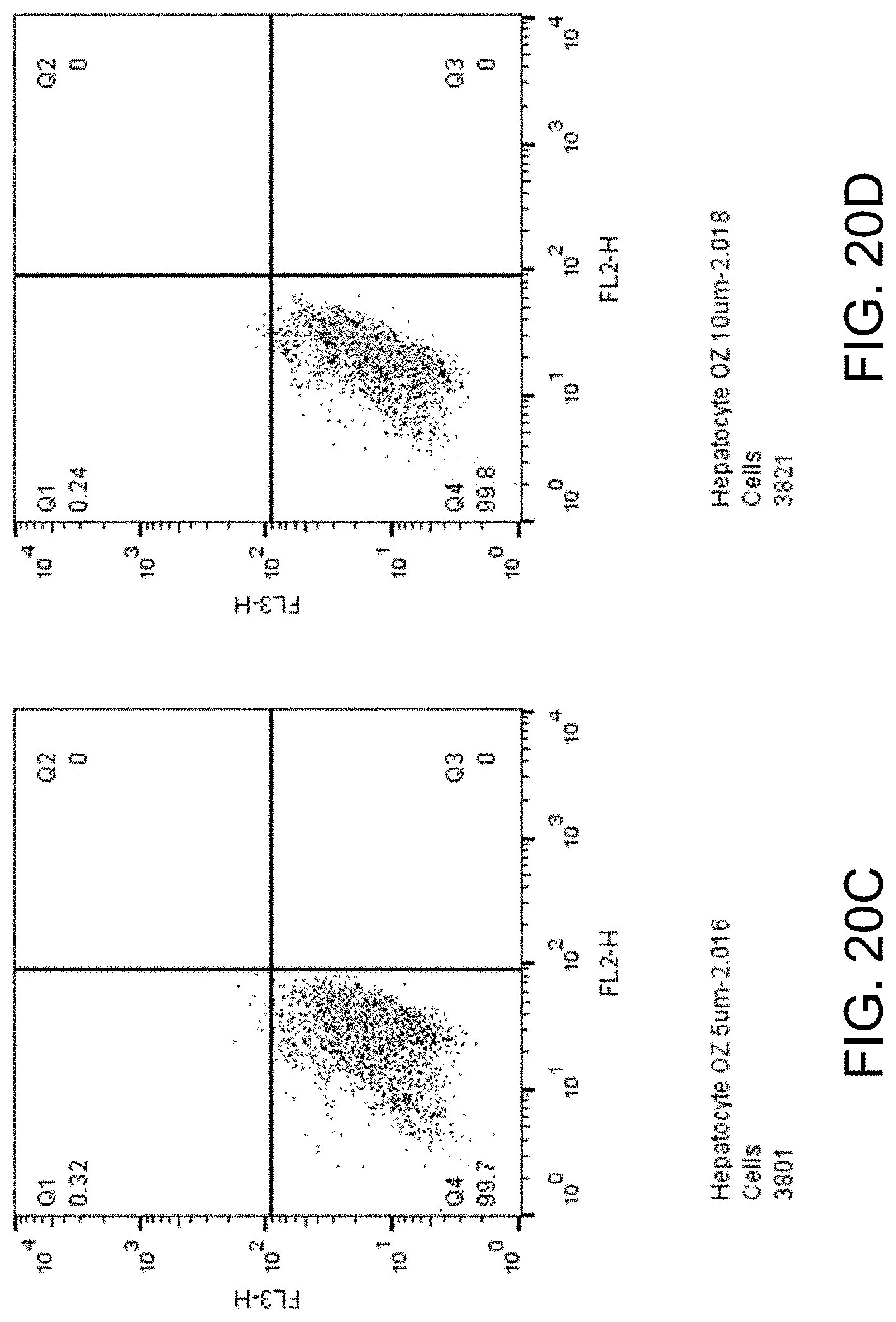
[0035] FIGS. 20A-D show apoptosis assay data for various concentrations of ozanimod and no-drug control for primary human hepatocytes. Ozanimod did not induce apoptosis of the primary human hepatocytes at any concentration tested.
DETAILED DESCRIPTION
[0036] The invention provides new uses of sphingosine kinase-1 inhibitors, such as SK1-I, and selective sphingosine-1-phosphate receptor agonists, such as ozanimod, for treating cancers, such as a liver cancer, and myeloproliferative neoplasms (myeloproliferative disorders), in mammals, such as human patients. The invention also provides new uses of selective sphingosine kinase type I inhibitors, such as SK1-I, and selective sphingosine-1-phosphate receptor agonists, such as ozanimod, for inducing apoptosis and/or necrosis of mammalian, such as human, cancer cells, such as liver cancer cells, and myeloproliferative neoplasm cells.
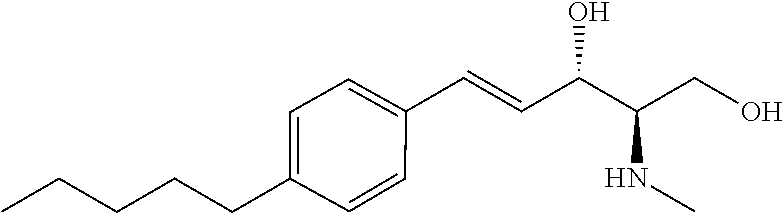
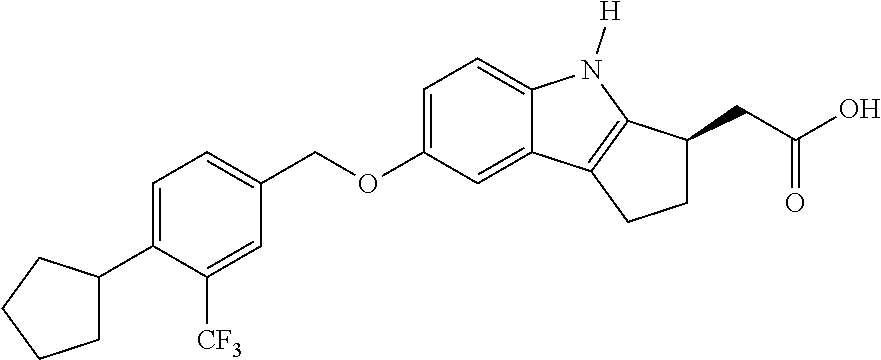
[0037] Sphingosine kinase 1 inhibitors used in various embodiments of the invention may, for example, include any of those disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, each of which is hereby incorporated by reference in its entirety herein, or pharmaceutically acceptable salts thereof. The sphingosine kinase I inhibitor may, for example, be (E,2R,3 S)-2-(methyl amino)-5 -(4-pentylphenyl)pent-4-ene-1,3 -diol (also known as SK1-I), or a pharmaceutically acceptable salt thereof such as but not limited to a hydrochloride salt. The structure of SK1-I is shown below.
##STR00001##
[0038] See also Paugh etal., Blood, 2008 112: 1382-1391.
[0039] The sphingosine kinase I inhibitor may, for example, be a compound having the structure
##STR00002##
[0040] or a pharmaceutically acceptable salt of the compound such as but not limited to a hydrochloride salt.
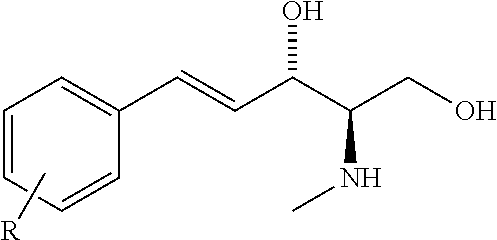
[0041] The sphingosine kinase I inhibitor may, for example, be a compound having the structure
##STR00003##
[0042] wherein R is selected from a straight carbon chain, a branched carbon chain, a straight carbon chain comprising one or more heteroatoms, a branched carbon chain comprising one or more heteroatoms, a cyclic ring, a heterocyclic ring, an aromatic ring, a hetero-aromatic ring, or any combination of the foregoing,
[0043] or a pharmaceutically acceptable salt thereof such as but not limited to a hydrochloride salt.
[0044] The sphingosine kinase I inhibitor may, for example, be a compound having the structure
##STR00004##
[0045] wherein R is 3,4-dimethoxy, 4-phenyl or 3-pentyl, or a pharmaceutically acceptable salt thereof such as but not limited to a hydrochloride salt.
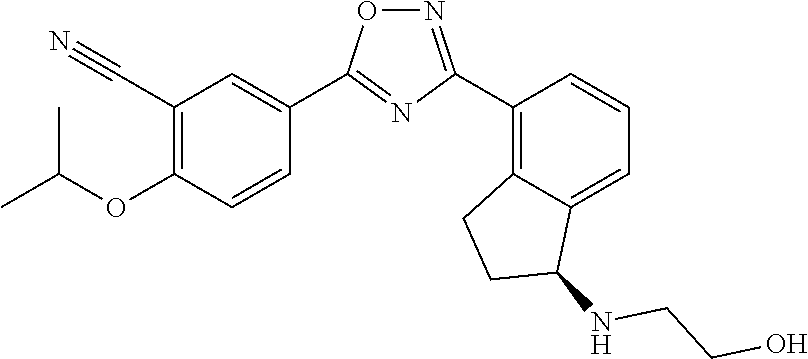
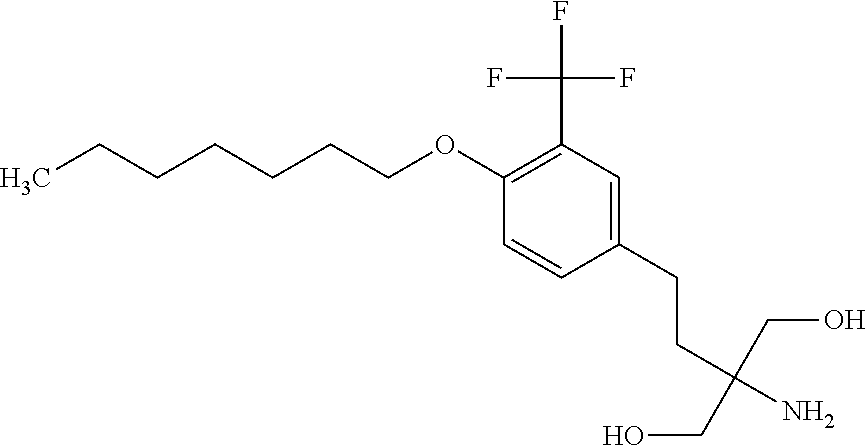
[0046] Sphingosine-1-phosphate receptor agonists used in various embodiments of the invention may, for example, be any of those disclosed in any of U.S. Pub. Nos. 20110172202, 20130231326, and 20150299149, or pharmaceutically acceptable salts thereof. The agonists may be agonists of one or both of sphingosine-1-phosphate receptor-1 (S1P.sub.1) and sphingosine-1-phosphate receptor-5 (S1P.sub.b 5) and may have little or at least no substantial agonist activity against other sphingosine-1-phosphate receptors (in a mammal such as a human). The sphingosine-1-phosphate receptor agonist used may, for example, be 5-[3-[(1 S)-1-(2-hydroxyethylamino)-2,3-dihydro-1H-inden-4-yl]-1,2,4-oxad- iazol-5-yl]-2-propan-2-yloxybenzonitrile (also known as ozanimod and RPC1063) or a pharmaceutically acceptable salt thereof such as but not limited to a hydrochloride salt. The structure of ozanimod is shown below.
##STR00005##
[0047] See also Scott et al., British Journal of Pharmacology 2016 173:1778-1792.
[0048] The sphingosine-1-phosphate receptor agonist may, for example, be etrasimod or a pharmaceutically acceptable salt thereof such as but not limited to a hydrochloride salt. The structure of etrasimod is shown below.
##STR00006##
[0049] The sphingosine-1-phosphate receptor agonist may, for example, be amiselimod or a pharmaceutically acceptable salt thereof such as but not limited to a hydrochloride salt. The structure of amiselimod is shown below.
##STR00007##
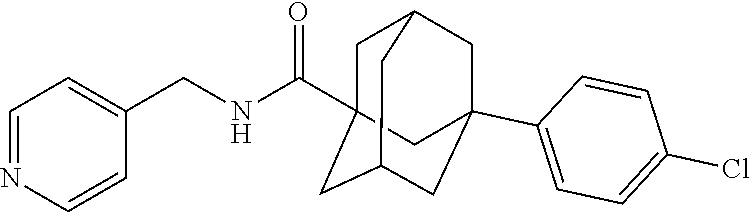
[0050] ABC294640 (also known as Yeliva.RTM.) used in the experiments presented herein is a reported sphingosine kinase-2 selective inhibitor, namely the compound (7 S)-3 -(4-chlorophenyl)-N-(pyridin-4-ylmethyl)adamantane-1 -carboxamide. The structure of ABC294640 is shown below.
##STR00008##
[0051] See also French etal., J. Pharmacol. Exp. Ther. 2010, 333, 129-139.
Experiments
[0052] MTT cell viability assays evaluating the effect of different concentrations of each of ozanimod, ABC294640 and SK1-I on four human hepatocellular carcinoma cell lines,
[0053] Hep G2, Hep 3B, Huh 7 and PLC-PRF5 were performed. These four cells line were selected for the study because they are among HCC cells lines whose gene expression profiles most closely resemble those of primary HCC tumors. See Chen et al., BMC Medical Genomics 2015, 8(Suppl 2):S5. The following concentrations of the compounds were tested. [0054] Ozanimod: 200 .mu.M, 111.1 .mu.M, 61.73 .mu.M, 34.29 .mu.M, 19.05 .mu.M, 10.58 and 0 .mu.M. [0055] ABC294640: 200 .mu.M, 111.1 .mu.M, 61.73 .mu.M, 34.29 .mu.M, 19.05 .mu.M, 10.58 and 0 .mu.M. [0056] SK1-I: 20 .mu.M, 11.11 .mu.M, 6.173 .mu.M, 3.429 .mu.M, 1.905 .mu.M, 1.058 and 0 .mu.M.
[0057] The following MTT assay protocol was followed. [0058] Prepared stock solutions: 50 mM ozanimod, 50 mM ABC294640, 10 mM SK1-I in DMSO. [0059] Plated 20000 cells in 160 .sub.11.1 medium per well in 96-well plates for each cell line and incubated at 37.degree. C. overnight. [0060] Prepared compound serial dilutions: for ozanimod and ABC294640: dilute stock 50 mM 1:50 in medium to 1000 .mu.M; for SK1-I, dilute stock 10 mM 1:50 in medium to 100 .mu.M, them make serial 1:1.8 fold serial dilution to the titration. For negative control, diluted DMSO 1:50 to medium. [0061] Added 40 .sub.11.1 negative control and serially titrated compounds to 160 .mu.l of cells for each cell line. Performed in triplicate for each cell line. [0062] Incubated at 37.degree. C. for 72 hours. [0063] Performed assay using Vybrant MTT Cell Proliferation Assay kit (V-13154) (Molecular Probes) from Thermo Fisher Scientific (Waltham, Mass. USA).
[0064] The results of these 72-hour treatment MTT assays are shown in FIGS. 1-3 as follows.
[0065] FIG. 1 shows the MTT assay data (72 hours) for ozanimod for the four human hepatocellular carcinoma cell lines.
[0066] FIG. 2 shows the MTT assay data (72 hours) for ABC294640 for the four human hepatocellular carcinoma cell lines.
[0067] FIG. 3 shows the MTT assay data (72 hours) for SK1-I for the four human hepatocellular carcinoma cell lines.
[0068] 48-hour treatment MTT assays were also performed as follows. FIG. 4A shows MTT assay data (48 hours) for various concentrations of SK1-I for three human hepatocellular carcinoma cell lines, Hep G2, Huh 7, and PLC-PRF5. FIG. 4B shows MTT assay data (48 hours) for various concentrations of PF-543, a super potent SphK1 inhibitor (see Schnute et al., Biochem. J. (2012) 444, 79-88), for the same three human hepatocellular carcinoma cell lines shown in FIG. 4A. This data shows that SK1-I is more effective at killing hepatocellular carcinoma cells than PF-543 despite the latter drug's much greater potency in inhibiting SphK1.
[0069] Apoptosis/necrosis assays evaluating the effect of different concentrations of each of ozanimod, ABC294640 and SK1-I on the four human hepatocellular carcinoma cell lines, Hep G2, Hep 3B, Huh 7 and PLC-PRF5 were also performed.
[0070] The protocol used for the apoptosis assays was: [0071] Plated cells in 6-well plates and incubated at 37.degree. C. overnight. [0072] Prepared concentrations of test compound and control compound DMSO in media. [0073] Ozanimod: 5 .mu.M, 10 and 20 .mu.M. [0074] ABC294640: 10 .mu.M, 20 and 40 .mu.M. [0075] SK1-I: 10 .mu.M, 20 and 40 .mu.M. [0076] Control: 0.2% DMSO. [0077] Aspirated the medium from the 6-well plates and added test compound concentrations in media or 0.2% DMSO control in media. Incubated at 37.degree. C. for 24 hours. [0078] Collected and processed the cells following the flow cytometry protocol of the GFP Certified.RTM. Apoptosis/Necrosis detection kit from Enzo Life Sciences, Inc. (product no. ENZ-51002; Farmingdale, N.Y., USA).
[0079] FIGS. 5-16 present graphs plotting the data from these 24-hour apoptosis assays for the different concentrations of compounds and no-drug control for the various cell lines. Channel FL2 picks up the apoptosis signal and channel FL3 picks up the necrosis signal. Data points in Quadrant 3 (Q3) in the graphs corresponds to cells undergoing apoptosis (cells positive for the apoptosis detection reagent of the assay). Data points in Quadrant 2 (Q2) in the graphs corresponds to cells that are positive for the apoptosis detection reagent and positive for the necrosis detection reagent of the assay (indicative of late-stage apoptosis).
[0080] FIGS. 5A-D show the apoptosis assay data for various concentrations of SK1-I and no-drug control for Huh7 cells. FIG. 5A shows the results for no-drug control. FIG. 5B shows the results for treatment with 10 .mu.M SK1-I. FIG. 5C shows the results for treatment with 20 .mu.M SK1-I. FIG. 5D shows the results for treatment with 40 .mu.M SK1-I. SK1-I strongly induced apoptosis in the Huh7 cells.
[0081] FIGS. 6A-D show the apoptosis assay data for various concentrations of SK1-I and no-drug control for PLC-PRFS cells. FIG. 6A shows the results for no-drug control. FIG. 6B shows the results for treatment with 10 .mu.M SK1-I. FIG. 6C shows the results for treatment with 20 .mu.M SK1-I. FIG. 6D shows the results for treatment with 40 .mu.M SK1-I. SK1-I strongly induced apoptosis in the PLC-PRFS cells.
[0082] FIGS. 7A-D show the apoptosis assay data for various concentrations of SK1-I and no-drug control for Hep 3B cells. FIG. 7A shows the results for no-drug control. FIG. 7B shows the results for treatment with 10 .mu.M SK1-I. FIG. 7C shows the results for treatment with 20 .mu.M SK1-I. FIG. 7D shows the results for treatment with 40 .mu.M SK1-I. SK1-I strongly induced apoptosis in the Hep 3B cells.
[0083] FIGS. 8A-D show the apoptosis assay data for various concentrations of SK1-I and no-drug control for Hep G2 cells. FIG. 8A shows the results for no-drug control. FIG. 8B shows the results for treatment with 10 .mu.M SK1-I. FIG. 8C shows the results for treatment with 20 .mu.M SK1-I. FIG. 8D shows the results for treatment with 40 .mu.M SK1-I. SK1-I strongly induced apoptosis in the Hep G2 cells.
[0084] FIGS. 9A-D show the apoptosis assay data for various concentrations of ozanimod and no-drug control for Huh7 cells. FIG. 9A shows the results for no-drug control. FIG. 9B shows the results for treatment with 5 .mu.M ozanimod. FIG. 9C shows the results for treatment with 10 .mu.M ozanimod. FIG. 9D shows the results for treatment with 20 .mu.M ozanimod. Ozanimod strongly induced apoptosis in the Huh7 cells.
[0085] FIGS. 10A-D show the apoptosis assay data for various concentrations of ozanimod and no-drug control for PLC-PRF5 cells. FIG. 10A shows the results for no-drug control. FIG. 10B shows the results for treatment with 5 .mu.M ozanimod. FIG. 10C shows the results for treatment with 10 .mu.M ozanimod. FIG. 10D shows the results for treatment with 20 .mu.M ozanimod. Ozanimod strongly induced apoptosis in the PLC-PRFS cells.
[0086] FIGS. 11A-D show the apoptosis assay data for various concentrations of ozanimod and no-drug control for Hep 3B cells. FIG. 11A shows the results for no-drug control. FIG. 11B shows the results for treatment with 10 .mu.M ozanimod. FIG. 11C shows the results for treatment with 20 .mu.M ozanimod. FIG. 11D shows the results for treatment with 40 .mu.M ozanimod. Ozanimod strongly induced apoptosis in the Hep 3B cells.
[0087] FIGS. 12A-D show the apoptosis assay data for various concentrations of ozanimod and no-drug control for Hep G2 cells. FIG. 12A shows the results for no-drug control. FIG. 12B shows the results for treatment with 10 .mu.M ozanimod. FIG. 12C shows the results for treatment with 20 .mu.M ozanimod. FIG. 12D shows the results for treatment with 40 .mu.M ozanimod. Ozanimod strongly induced apoptosis in the Hep G2 cells.
[0088] FIGS. 13A-D show the apoptosis assay data for various concentrations of ABC294640 and no-drug control for Huh7 cells. FIG. 13A shows the results for no-drug control. FIG. 13B shows the results for treatment with 10 .mu.M ABC294640. FIG. 13C shows the results for treatment with 20 .mu.M ABC294640. FIG. 13D shows the results for treatment with 40 .mu.M ABC294640. ABC294640 failed to induce apoptosis in the Huh7 cells.
[0089] FIGS. 14A-D show the apoptosis assay data for various concentrations of ABC294640 and no-drug control for PLC-PRF5 cells. FIG. 14A shows the results for no-drug control. FIG. 14B shows the results for treatment with 10 .mu.M ABC294640. FIG. 14C shows the results for treatment with 20 .mu.M ABC294640. FIG. 14D shows the results for treatment with 40 .mu.M ABC294640. ABC294640 did not substantially induce apoptosis in the PLC-PRF5 cells.
[0090] FIGS. 15A-D show the apoptosis assay data for various concentrations of ABC294640 and no-drug control for Hep 3B cells. FIG. 15A shows the results for no-drug control. FIG. 15B shows the results for treatment with 10 .mu.M ABC294640. FIG. 15C shows the results for treatment with 20 .mu.M ABC294640. FIG. 15D shows the results for treatment with 40 .mu.M ABC294640. ABC294640 did not substantially induce apoptosis in the Hep 3B cells.
[0091] FIGS. 16A-D show the apoptosis assay data for various concentrations of ABC294640 and no-drug control for Hep G2 cells. FIG. 16A shows the results for no-drug control. FIG. 16B shows the results for treatment with 10 .mu.M ABC294640. FIG. 16C shows the results for treatment with 20 .mu.M ABC294640. FIG. 16D shows the results for treatment with 40 .mu.M ABC294640. ABC294640 failed to induce apoptosis in the Hep G2 cells.
[0092] FIGS. 17A-C show apoptosis assay data for various concentrations of SK1-I and no-drug control for Jurkat cells (human T-cell leukemia cell line). FIG. 17A shows the results for no-drug control. FIG. 17B shows the results for treatment with 10 .mu.M SK1-I. FIG. 17C shows the results for treatment with 20 .mu.M SK1-I. SK1-I strongly induced apoptosis in the Jurkat cells.
[0093] FIGS. 18A-C show apoptosis assay data for various concentrations of ozanimod and no-drug control for Jurkat cells. FIG. 18A shows the results for no-drug control. FIG. 18B shows the results for treatment with 5.mu.M ozanimod. FIG. 18C shows the results for treatment with 10 .mu.M ozanimod. Ozanimod strongly induced apoptosis in the Jurkat cells.
[0094] In still further experiments, the effects of SK1-I and ozanimod on normal (non-cancerous) primary human hepatocytes were investigated. Fresh human hepatocytes in a 12-well plate (HUF12) were obtained from Triangle Research Labs (Durham, N.C., USA; part of Lonza Group) and handled according to the supplier's protocol. The shipping medium was aspirated from each well and replaced with 1 ml per well of warm Hepatocyte Maintenance Medium. The plate was then placed in a 5% CO.sub.2 incubator at 37.degree. C. and the hepatocytes were allowed to acclimate overnight. The Hepatocyte Maintenance Medium was replaced before treatment with drug or no-drug control, and the hepatocytes were treated with 0 .mu.M (no-drug control), 10 .mu.M, 20 .mu.M, or 40 .mu.M SK1-I or 0 .mu.M, 2.5 .mu.M, 5 .mu.M, or 10 .mu.M ozanimod for 24 hours under incubation. The cells were then harvested and analyzed using the GFP Certified.RTM. Apoptosis/Necrosis detection kit. The results are shown in FIGS. 19A-D for SK1-I and FIGS. 20A-D for ozanimod as follows.
[0095] FIGS. 19A-D show apoptosis assay data for various concentrations of SK1-I and no-drug control for primary human hepatocytes. FIG. 19A shows the results for no-drug control. FIG. 19B shows the results for treatment with 10 .mu.M SK1-I. FIG. 19C shows the results for treatment with 20 .mu.M SK1-I. FIG. 19D shows the results for treatment with 40 .mu.M SK1-I. SK1-I did not induce apoptosis of the primary human hepatocytes at any concentration tested.
[0096] FIGS. 20A-D show apoptosis assay data for various concentrations of ozanimod and no-drug control for primary human hepatocytes. FIG. 20A shows the results for no-drug control. FIG. 20B shows the results for treatment with 2.5 .mu.M ozanimod. FIG. 20C shows the results for treatment with 5.mu.M ozanimod. FIG. 20D shows the results for treatment with 10 .mu.M ozanimod. Ozanimod did not induce apoptosis of the primary human hepatocytes at any concentration tested.
[0097] Without limitation, the following embodiments are also provided.
Embodiments Involving Sphingosine Kinase 1 (SphK1) Inhibitors
[0098] Embodiment 1. A method for treating liver cancer in a mammalian subject, such as a human, including the step of:
[0099] administering to a mammalian subject in need of treatment for liver cancer, an effective amount of a sphingosine kinase type I inhibitor.
[0100] Embodiment 2. The method of embodiment 1, wherein the liver cancer is hepatic cell carcinoma (HCC).
[0101] Embodiment 3. The pharmaceutical composition of embodiment 1, wherein the liver cancer is selected from the group consisting of fibrolamellar HCC, cholangiocarcinoma (bile duct cancer) and angiosarcoma.
[0102] Embodiment 4. The method of any one of the preceding embodiments, wherein the sphingosine kinase type I inhibitor at least substantially does not inhibit sphingosine kinase type II.
[0103] Embodiment 5. The method of any one of the preceding embodiments, wherein the sphingosine kinase type I inhibitor includes a sphingosine kinase type I inhibitor disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, or a pharmaceutically acceptable salt thereof.
[0104] Embodiment 6. The method of any one of embodiments 1-5, wherein the sphingosine kinase type I inhibitor includes SK1-I or a pharmaceutically acceptable salt thereof.
[0105] Embodiment 7. The method of any one of the preceding embodiments, wherein said administration includes parenteral administration.
[0106] Embodiment 8. The method of embodiment 7, wherein said administration is via injection, such as intravenous injection, intramuscular injection, or subcutaneous injection.
[0107] Embodiment 9. The method of any one of embodiments 1-6, wherein said administration includes non-parenteral administration.
[0108] Embodiment 10. The method of any one of embodiments 1-6, wherein said administration includes oral administration by ingestion.
[0109] Embodiment 11. The method of embodiment 10, wherein said oral administration includes administering a dosage form including the sphingosine kinase type I inhibitor and at least one pharmaceutically acceptable excipient.
[0110] Embodiment 12. The method of embodiment 11, wherein the dosage form is selected from the group consisting of a tablet, a capsule, and a gel cap.
[0111] Embodiment 13. The method of any one of embodiments 1-6, wherein said administration includes administration via the alimentary canal.
[0112] Embodiment 14. The method of any one of the preceding embodiments, further including the step of:
[0113] co-administering to the subject an effective amount of ozanimod or a pharmaceutically acceptable salt thereof.
[0114] Embodiment 15. A pharmaceutical composition for the treatment of a liver cancer in a mammalian subject, such as a human, including:
[0115] a therapeutically effective amount of a sphingosine kinase type I inhibitor.
[0116] Embodiment 16. The pharmaceutical composition of embodiment 15, wherein the liver cancer is hepatic cell carcinoma (HCC).
[0117] Embodiment 17. The pharmaceutical composition of embodiment 15, wherein the liver cancer is selected from the group consisting of Fibrolamellar HCC, Cholangiocarcinoma (bile duct cancer) and Angiosarcoma.
[0118] Embodiment 18. The pharmaceutical composition of any one of the preceding embodiments, wherein the sphingosine kinase type I inhibitor at least substantially does not inhibit sphingosine kinase type II.
[0119] Embodiment 19. The pharmaceutical composition of any one of the preceding embodiments, wherein the sphingosine kinase type I inhibitor includes a sphingosine kinase type I inhibitor disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, or a pharmaceutically acceptable salt thereof.
[0120] Embodiment 20. The pharmaceutical composition of any one of embodiment 15-18, wherein the sphingosine kinase type I inhibitor includes SK1-I or a pharmaceutically acceptable salt thereof.
[0121] Embodiment 21. The pharmaceutical composition of any one of the preceding embodiments, wherein said composition is for parenteral administration.
[0122] Embodiment 22. The pharmaceutical composition of embodiment 21, wherein said composition is for administration via injection, such as intravenous injection, intramuscular injection, or subcutaneous injection.
[0123] Embodiment 23. The pharmaceutical composition of any one of embodiments 15-20, wherein said composition is for non-parenteral administration.
[0124] Embodiment 24. The pharmaceutical composition of any one of embodiments 15-20, wherein said composition is for oral administration by ingestion.
[0125] Embodiment 25. The pharmaceutical composition of any one of embodiment 15-24, further including at least one pharmaceutically acceptable excipient.
[0126] Embodiment 26. The pharmaceutical composition of any one of embodiments 15-25, wherein said composition is a solid dosage form.
[0127] Embodiment 27. The pharmaceutical composition of embodiment 24, provided in a dosage form selected from the group consisting of a liquid, a tablet, a capsule, and a gel cap.
[0128] Embodiment 28. The pharmaceutical composition of any one of embodiments 15-20, wherein said composition is for administration via the alimentary canal.
[0129] Embodiment 29. The pharmaceutical composition of any one of embodiments 15-28, further including a therapeutically effective amount of ozanimod or a pharmaceutically acceptable salt thereof.
[0130] Embodiment 30. A method for inducing apoptosis of mammalian liver cancer cells, such as hepatocellular carcinoma (HCC) cells, including the step of:
[0131] contacting the mammalian liver cancer cells with an effective amount of a selective sphingosine kinase type I inhibitor, such as any of those disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, such as SK1-I, or a pharmaceutically acceptable salt thereof.
[0132] Embodiment 31. Use of a selective sphingosine kinase type I inhibitor, such as any of those disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, such as SK1-I, or a pharmaceutically acceptable salt thereof, for inducing apoptosis of mammalian liver cancer cells, such as hepatocellular carcinoma (HCC) cells.
[0133] Embodiments involving sphingosine-1-phosphate receptor agonists
[0134] Embodiment 32. A method for treating a cancer or a myleoproilferative disorder (myeloproliferative neoplasm) in a mammalian subject, such as a human, including the step of:
[0135] administering to a mammalian subject in need of treatment for a cancer or myeloproliferative disorder, a therapeutically effective amount of a sphingosine-1-phosphate receptor agonist, such as an agonist of one or both of sphingosine-1-phosphate receptor-1 (S1P.sub.1) and sphingosine-1-phosphate receptor-5 (S1P.sub.5) such as ozanimod (RPC1063) or a pharmaceutically acceptable salt thereof.
[0136] Embodiment 33. The method of embodiment 32, wherein the myeloproliferative disorders is selected from the group consisting of: Chronic myelogenous leukemia (e.g, BCR-ABL1-positive); Chronic neutrophilic leukemia; Polycythemia vera; Primary myelofibrosis; Essential thrombocythemia; Chronic eosinophilic leukemia (not otherwise specified); and Mastocytosis.
[0137] Embodiment 34. The method of embodiment 32, wherein said cancer is a hematological malignancy.
[0138] Embodiment 35. The method of embodiment 34, wherein said hematological malignancy is selected from the group consisting of: leukemias, lymphomas and myelomas.
[0139] Embodiment 36. The method of embodiment 35, wherein said hematological malignancy is selected from the group consisting of: Acute lymphoblastic leukemia (ALL); Acute myelogenous leukemia (AML); Chronic lymphocytic leukemia (CLL); Chronic myelogenous leukemia (CML); Acute monocytic leukemia (AMoL); Hodgkin's lymphomas (e.g., any of main four subtypes); and Non-Hodgkin's lymphomas (any subtype).
[0140] Embodiment 37. The method of embodiment of embodiment 32, wherein said cancer is a solid organ cancer.
[0141] Embodiment 38. The method of embodiment 32, wherein the solid organ cancer is selected from the group consisting of: Adipose tissue cancers such as Liposarcoma,
[0142] Myxoid liposarcoma adipose; Bladder cancer; Bone cancers such as Chondroblastoma, Chordoma, Ewings sarcoma, Osteosarcoma, Spindle cell tumor; Brain tumors such as Ganglioneuroblastoma, Ganglioneuroma, Glioblastoma, Malignant peripheral nerve sheath tumor, Neuroblastoma, Neurofibroma, Schwannoma brain; Connective tissue cancers such as Chondromyxoid fibroma, Chondrosarcoma, Dedifferentiated chondrosarcoma, Fibromatosis, Monophasic synovial sarcoma; Esophageal adenocarcinoma; Oral squamous cell carcinoma; Kidney cancers such as Kidney carcinoma, Renal cell carcinoma; Liver cancers such as Hepatocellular carcinoma (HCC), Fibrolamellar HCC, Cholangiocarcinoma (bile duct cancer) and Angiosarcoma; Lung cancer such as NSCLC, SCLC; Uterine tumors; Head and Neck cancers such as head and neck squamous cell carcinoma; Ovarian tumors; Prostate cancer; Muscle tissue cancers such as Acute quadriplegic myopathy; Skin cancers such as Melanoma, Sarcoma, Kaposi sarcoma; Alveolar rhabdomyo sarcoma, Embryonal rhabdomyo sarcoma, Leiomyosarcoma; Germ cell tumors such as of the testes, testicular cancer; and Thyroid cancer such as Thyroid adenocarcinoma.
[0143] Embodiment 39. The method of embodiment 33, wherein the solid organ cancer is liver cancer.
[0144] Embodiment 40. The method of embodiment 39, wherein the liver cancer is hepatic cell carcinoma (HCC).
[0145] Embodiment 41. The method of any one of embodiments 32-40, wherein the sphingosine-1-phosphate receptor agonist at least substantially does not agonize sphingosine-1-phosphate receptors other than types -1 and -5.
[0146] Embodiment 42. The method of any one of embodiments 32-41, wherein the sphingosine-1-phosphate receptor agonist includes a sphingosine-1-phosphate receptor agonist disclosed in any of U.S. Pub. Nos. 20110172202, 20130231326, and 20150299149 or a pharmaceutically acceptable salt thereof.
[0147] Embodiment 43. The method of any one of embodiments 32-42, wherein the sphingosine-1-phosphate receptor agonist includes ozanimod or a pharmaceutically acceptable salt thereof.
[0148] Embodiment 44. The method of any one of embodiments 32-43, wherein said administration includes parenteral administration.
[0149] Embodiment 45. The method of embodiment 44, wherein said administration is via injection, such as intravenous injection, intramuscular injection, or subcutaneous injection.
[0150] Embodiment 46. The method of any one of embodiments 32-43, wherein said administration includes non-parenteral administration.
[0151] Embodiment 47. The method of any one of embodiments 32-43, wherein said administration includes oral administration by ingestion.
[0152] Embodiment 48. The method of embodiment 47, wherein said oral administration includes administering a dosage form including the sphingosine-1-phosphate receptor agonist and at least one pharmaceutically acceptable excipient.
[0153] Embodiment 49. The method of embodiment 48, wherein the dosage form is selected from the group consisting of a liquid, a tablet, a capsule, and a gel cap.
[0154] Embodiment 50. The method of any one of embodiments 32-43, wherein said administration includes administration via the alimentary canal.
[0155] Embodiment 51. The method of any one of embodiments 32-50, further including the step of:
[0156] co-administering to the subject an effective amount of a sphingosine kinase type I inhibitor, such as SK1-I, or a pharmaceutically acceptable salt thereof.
[0157] Embodiment 52. The method of any one of embodiments 32-51, further including the step of:
[0158] co-administering to the subject a therapeutically effective amount of a cellular ceramide generation promoter, such as 6-[(2 S,4 R,6 E)-4-Methyl-2-(methylamino)-3-oxo-6-octenoic acid]cyclosporin D (Valspodor; PSC833) or a pharmaceutically acceptable salt thereof.
[0159] Embodiment 53. The method of any one of embodiments 32-52, further including the step of:
[0160] coadministering to the subject a therapeutically effective amount of ceramide.
[0161] Embodiment 54. A pharmaceutical composition for the treatment of a cancer or a myeloproliferative disorder (myeloproliferative neoplasm) in a mammalian subject, such as a human, including:
[0162] a therapeutically effective amount of a sphingosine-1-phosphate receptor agonist, such as an agonist of one or both of sphingosine-1-phosphate receptor-1 (S1P.sub.1) and sphingosine-1-phosphate receptor-5 (S1P.sub.5) such as ozanimod (RPC1063) or a pharmaceutically acceptable salt thereof.
[0163] Embodiment 55. The pharmaceutical composition of embodiment 54, wherein the myeloproliferative disorders is selected from the group consisting of: Chronic myelogenous leukemia (BCR-ABL1-positive); Chronic neutrophilic leukemia; Polycythemia vera; Primary myelofibrosis; Essential thrombocythemia; Chronic eosinophilic leukemia (not otherwise specified); and Mastocytosis.
[0164] Embodiment 56. The pharmaceutical composition of embodiment 54, wherein said cancer is a hematological malignancy.
[0165] Embodiment 57. The pharmaceutical composition of embodiment 56, wherein said hematological malignancy is selected from the group consisting of: leukemias, lymphomas and myelomas.
[0166] Embodiment 58. The pharmaceutical composition of embodiment 57, wherein said hematological malignancy is selected from the group consisting of: Acute lymphoblastic leukemia (ALL); Acute myelogenous leukemia (AML); Chronic lymphocytic leukemia (CLL); Chronic myelogenous leukemia (CML); Acute monocytic leukemia (AMoL); Hodgkin's lymphomas (e.g., any of main four subtypes); and Non-Hodgkin's lymphomas (any subtype).
[0167] Embodiment 59. The pharmaceutical composition of embodiment of embodiment 54, wherein said cancer is a solid organ cancer.
[0168] Embodiment 60. The pharmaceutical composition of embodiment 59, wherein the solid organ cancer is selected from the group consisting of: Adipose tissue cancers such as Liposarcoma, Myxoid liposarcoma adipose; Bladder cancer; Bone cancers such as Chondroblastoma, Chordoma, Ewings sarcoma, Osteosarcoma, Spindle cell tumor; Brain tumors such as Ganglioneuroblastoma, Ganglioneuroma, Malignant peripheral nerve sheath tumor, Neuroblastoma, Neurofibroma, Schwannoma brain; Connective tissue cancers such as Chondromyxoid fibroma, Chondrosarcoma, Dedifferentiated chondrosarcoma, Fibromatosis, Monophasic synovial sarcoma; Esophageal adenocarcinoma; Oral squamous cell carcinoma; Kidney cancers such as Kidney carcinoma, Renal cell carcinoma; Liver cancers such as Hepatocellular carcinoma (HCC), Fibrolamellar HCC, Cholangiocarcinoma (bile duct cancer) and Angiosarcoma; Lung cancer such as NSCLC, SCLC; Uterine tumors; Head and Neck cancers such as head and neck squamous cell carcinoma; Ovarian tumors; Prostate cancer; Muscle tissue cancers such as Acute quadriplegic myopathy; Skin cancers such as Melanoma, Sarcoma, Kaposi sarcoma; Alveolar rhabdomyo sarcoma, Embryonal rhabdomyo sarcoma, Leiomyosarcoma; Germ cell tumors such as of the testes, testicular cancer; and Thyroid cancer such as Thyroid adenocarcinoma.
[0169] Embodiment 61. The pharmaceutical composition of embodiment 59, wherein the solid organ cancer is a liver cancer.
[0170] Embodiment 62. The pharmaceutical composition of embodiment 61, wherein the liver cancer is selected from the group consisting of hepatic cell carcinoma (HCC), fibrolamellar HCC, cholangiocarcinoma (bile duct cancer) and angiosarcoma.
[0171] Embodiment 63. The pharmaceutical composition of any one of embodiments 54-62, wherein the sphingosine-1-phosphate receptor agonist at least substantially does not agonize sphingosine-1-phosphate receptors other than types -1 and -5.
[0172] Embodiment 65. The pharmaceutical composition of any one of embodiments 54-64, wherein the sphingosine-1-phosphate receptor agonist includes a sphingosine-1-phosphate receptor agonist disclosed in any of U.S. Pub Nos. 20110172202, 20130231326, and 20150299149.
[0173] Embodiment 66. The pharmaceutical composition of any one of embodiments 54-64, wherein the sphingosine-1-phosphate receptor agonist includes ozanimod or a pharmaceutically acceptable salt thereof.
[0174] Embodiment 67. The pharmaceutical composition of any one of embodiments 54-66, wherein said composition is for parenteral administration.
[0175] Embodiment 68. The pharmaceutical composition of embodiment 67, wherein said composition is for administration via injection, such as intravenous injection, intramuscular injection, or subcutaneous injection.
[0176] Embodiment 69. The pharmaceutical composition of any one of embodiments 54-66, wherein said composition is for non-parenteral administration.
[0177] Embodiment 70. The pharmaceutical composition of any one of embodiments 54-66, wherein said composition is for oral administration by ingestion.
[0178] Embodiment 71. The pharmaceutical composition of any one of embodiments 54-70, further including at least one pharmaceutically acceptable excipient.
[0179] Embodiment 72. The pharmaceutical composition of any one of embodiments 54-71, wherein said composition is a solid dosage form.
[0180] Embodiment 73. The pharmaceutical composition of embodiment 72, provided in a dosage form selected from the group consisting of a tablet, a capsule, and a gel cap.
[0181] Embodiment 74. The pharmaceutical composition of any one of embodiments 54-66, wherein said composition is for administration via the alimentary canal.
[0182] Embodiment 75. The pharmaceutical composition of any one of embodiments 54-74, further including a therapeutically effective amount of a sphingosine kinase type I inhibitor, such as one disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, such as SK1-I, or a pharmaceutically acceptable salt thereof.
[0183] Embodiment 76. The pharmaceutical composition of any one of embodiments 54-74, for use in combination with a therapeutically effective amount of a sphingosine kinase type I inhibitor, such as one disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, such as SK1-I, or a pharmaceutically acceptable salt thereof.
[0184] Embodiment 77. The pharmaceutical composition of any one of embodiments 54-76, further including a therapeutically effective amount of a cellular ceramide generation promoter, such as 6-[(2 S,4 R,6 E)-4-Methyl-2-(methylamino)-3-oxo-6-octenoic acid]cyclosporin D (Valspodor; PSC833) or a pharmaceutically acceptable salt thereof.
[0185] Embodiment 78. The pharmaceutical composition of any one of embodiments 54-76, for use in combination with a therapeutically effective amount of a cellular ceramide generation promoter, 6-[(2 S,4 R,6 E)-4-Methyl-2-(methylamino)-3-oxo-6-octenoic acid]cyclosporin D (Valspodor; PSC833) or a pharmaceutically acceptable salt thereof.
[0186] Embodiment 79. The pharmaceutical composition of any one of embodiments 54-78 for use in combination with a therapeutically effective amount of ceramide.
[0187] Embodiment 80. A method for inducing apoptosis of mammalian cancer cells, such as liver cancer cells, such as hepatocellular carcinoma (HCC) cells, including the step of:
[0188] contacting the mammalian cancer cells with an effective amount of a sphingosine-1-phosphate receptor agonist, such as an agonist of one or both of sphingosine-1-phosphate receptor-1 (S1P.sub.1) and sphingosine-1-phosphate receptor-5 (S1P.sub.5) such as any of those disclosed in U.S. Pub Nos. 20110172202, 20130231326, and 20150299149, such as ozanimod (RPC1063), or a pharmaceutically acceptable salt thereof.
[0189] Embodiment 81. Use of a sphingosine-1-phosphate receptor agonist, such as an agonist of one or both of sphingosine-1-phosphate receptor-1 (S1P.sub.1) and sphingosine-1-phosphate receptor-5 (S1P.sub.5) such as any of those disclosed in U.S. Pub Nos. 20110172202, and 20130231326, and 20150299149, such as ozanimod (RPC1063), or a pharmaceutically acceptable salt thereof, for inducing apoptosis of mammalian cancer cells, such as liver cancer cells, such as, hepatocellular carcinoma (HCC) cells.
[0190] For embodiments involving ceramide, the ceramide may, for example, be formulated/co-formulated in an acid stable lipid vesicle/particle composition as disclosed in U.S. Pub. No. 20140271824, which is hereby incorporated by reference in its entirety, and administered/co-administered, for example by injection, such as intravenous injection, or orally.
[0191] Any of the method of treatment/use embodiments set forth herein may further include the step of: co-administering one or more immune checkpoint inhibitors, which may be monoclonal antibodies (mABs). The immune checkpoint inhibitor may be selected from the group consisting of the following: PD-1 inhibitors such as mAbs Pembrolizumab (Keytruda.RTM.) and Nivolumab (Opdivo.RTM.); PD-Ll inhibitors such as mAbs Atezolizumab (Tecentriq.RTM.), Avelumab (Bavencio.RTM.), and Durvalumab (Imfinzi.RTM.); and CTLA-4 inhibitors such as mAb Ipilimumab (Yervoy.RTM.); and V-domain Ig Suppressor of T Cell Activation (VISTA) inhibitors such as mAb JNJ-61610588 (ImmuNext Inc.). Similarly, any of the pharmaceutical composition embodiments of the invention may be for use in combination with one or more immune checkpoint inhibitors, such as those disclosed herein.
[0192] Still another embodiment of the invention provides a method for treating a cancer or a myleoproliferative disorder (myeloproliferative neoplasm), such as any of those disclosed herein, for example, a liver cancer, in a mammalian subject, such as a human, including the step of:
[0193] co-administering to a mammalian subject in need of treatment for a cancer or myeloproliferative disorder, a therapeutically effective amount of:
[0194] (a) a sphingosine kinase type I inhibitor, such as one disclosed in U.S. Pat. Nos. 8,372,888 and/or 8,314,151, such as SK1-I, or a pharmaceutically acceptable salt thereof; and
[0195] (b) one or more immune checkpoint inhibitors, which may be monoclonal antibodies, such as one or more selected from the group consisting of: PD-1 inhibitors such as mAbs Pembrolizumab (Keytruda.RTM.) and Nivolumab (Opdivo.RTM.); PD-Ll inhibitors such as mAbs Atezolizumab (Tecentriq.RTM.), Avelumab (Bavencio.RTM.), and Durvalumab (Imfinzi.RTM.); and CTLA-4 inhibitors such as mAb Ipilimumab (Yervoy.RTM.); and V-domain Ig Suppressor of T Cell Activation (VISTA) inhibitors such as mAb JNJ-61610588 (ImmuNext Inc.).
[0196] Immune checkpoint inhibitors may, for example, be administered by injection in the dosages described herein and/or at the currently approved dosages for said inhibitors.
[0197] The amount of compound that is effective for the treatment or prevention of a condition, alone or in combination with other compounds, may be determined by standard techniques. In addition, in vitro and/or in vivo assays may optionally be employed to help identify optimal dosage ranges. The precise dose to be employed will also depend on, e.g., the route of administration and the seriousness of the condition, and can be decided according to the judgment of a practitioner and/or each patient's circumstances. In other examples thereof, variations will necessarily occur depending upon the weight and physical condition (e.g., hepatic and renal function) of the patient being treated, the affliction to be treated, the severity of the symptoms, the frequency of the dosage interval, the presence of any deleterious side-effects, and the particular compound utilized, among other things.
[0198] Administration may be as a single dose or as a divided dose. In one embodiment, an effective dosage is administered once per month until the condition is abated. In another embodiment, the effective dosage is administered once per week, or twice per week or three times per week until the condition is abated. An effective dosage may, for example, be administered at least once daily or at least or at least once every two-days, or at least once every three days, four days, five days, six days or seven days. In another embodiment, an effective dosage amount is administered about every 24 h until the condition is abated. In another embodiment, an effective dosage amount is administered about every 12 h until the condition is abated. In another embodiment, an effective dosage amount is administered about every 8 h until the condition is abated. In another embodiment, an effective dosage amount is administered about every 6 h until the condition is abated. In another embodiment, an effective dosage amount is administered about every 4 h until the condition is abated.
[0199] The therapeutically effective dose may be expressed in terms of the amount of the compound(s) or pharmaceutically acceptable salts thereof administered per unit body weight of the subject per day of treatment, or the total amount administered per day of treatment. A daily dose may, for example, be at least 0.005 mg/kg of body weight, at least 0.01 mg/kg of body weight, at least 0.025 mg/kg of body weight, at least 0.05 mg/kg of body weight, at least 0.1 mg/kg of body weight, at least 0.2 mg/kg of body weight, at least 0.3 mg/kg of body weight, at least 0.4 mg/kg of body weight, at least 0.5 mg/kg of body weight, at least 0.6 mg/kg of body weight, at least 0.7 mg/kg of body weight, at least 0.8 mg/kg of body weight, at least 0.9 mg/kg of body weight, at least 1 mg/kg of body weight, at least 1.5 mg/kg of body weight, at least 2 mg/kg of body weight, at least 2.5 mg/kg of body weight, at least 3 mg/kg of body weight, at least 3.5 mg/kg of body weight, at least 4 mg/kg of body weight, at least 4.5 mg/kg of body weight, at least 5 mg/kg of body weight, or at one of said doses. A total daily dose may, for example, be in the range of 0.005 mg/kg to 5 mg/kg or any subrange or value therein, such as 0.025 to 5 mg/kg body weight, such as 0.05 to 5 mg/kg body weight. A total daily dose may, for example be in the range of 0.1 mg to 1,000 mg total or any subrange or value therein, such as 0.1 mg to 1,000 mg, such as 0.1 mg to 100 mg, such as 0.1 mg to 50 mg, such as 0.5 mg to 50 mg, such as 1.0 mg to 50 mg, such as 5 mg to 50 mg, or 0.1 mg to 10 mg, such as 0.5 mg to 10 mg. For SK1-I and related SphK1 inhibitors disclosed U.S. Pat. Nos. 8,372,888 and 8,314,151, and pharmaceutically acceptable salts thereof, a daily dose for human subjects may, for example, also be in the range of 0.5 mg/kg to 5 mg/kg or any subrange or value therein, such as 1 mg/kg to 4 mg/kg, such as 1 mg/kg to 3 mg/kg, or, for example, a total daily dose of 5 mg to 50 mg or any subrange or value therein, such as 10 mg to 40 mg, such as 20 mg to 40 mg. For ozanimod and related sphingosine-1-phosphate receptor agonists disclosed in U.S. Pub Nos. 20110172202, 20130231326, and 20150299149, and pharmaceutically acceptable salts thereof, a daily dose for human subjects may, for example, also be in the range of 0.1 mg to 10 mg or any subrange or value therein, such as 0.1 mg to 5 mg, such as 0.5 to 5 mg, such as 0.5 mg to 2.5 mg, such as 0.5 mg to 1.5 mg. A pharmaceutical composition according to the invention may, for example, include a daily dose amount of the compound as set forth herein.
[0200] The duration of treatment by administration of a therapeutic compound or combination according to the invention may continue for a plurality of days, such as for at least one week, at least two weeks, at least three weeks, at least four weeks, at least two months, at least three months, at least four months, at least five months, at least six months, at least seven months, at least eight months, at least nine months, at least 10 months, at least 11 months, at least 12 months, at least 11/2 years, at least 2 years, at least three years, at least four years, or may continue indefinitely.
[0201] The terms co-administration and co-administering mean that each of the things being co-administered is administered to a subject in such temporal proximity that each (or its active metabolite(s)) is present in active form in the subject for an at least partially overlapping period of time. Accordingly, co-administration may include, simultaneous administration, such as when the things being administered are part of the same pharmaceutical composition, or sequential administration of the things being co-administered, for example, within the same day of each other, within 12 hours of each other, within 6 hours of each other, within 3 hours of each other, within 1 hours of each other, or within 15 minutes of each other. The things being administered may be administered by the same route, such as by oral ingestion or injection, or by different routes.
[0202] Pharmaceutically acceptable salts and the selection and preparation thereof are well known in the art. Such salts include but are not limited to hydrochloride, citrate, glycolate, fumarate, malate, tartrate, mesylate, esylate, cinnamate, isethionate, sulfate, phosphate, diphosphate, nitrate, hydrobromide, hydroiodide, succinate, formate, acetate, dichloroacetate, lactate, p-toluenesulfonate, pamitate, pidolate, pamoate, salicylate, 4-aminosalicylate, benzoate, 4-acetamido benzoate, glutamate, aspartate, glycolate, adipate, alginate, ascorbate, besylate, camphorate, camphorsulfonate, camsylate, caprate, caproate, cyclamate, laurylsulfate, edisylate, gentisate, galactarate, gluceptate, gluconate, glucuronate, oxoglutarate, hippurate, lactobionate, malonate, maleate, mandalate, napsylate, napadisylate, oxalate, oleate, sebacate, stearate, succinate, thiocyanate, undecylenate, and xinafoate.
[0203] It should be noted that the indefinite articles "a" and "an" and the definite article "the" are used in the present application to mean one or more unless the context clearly dictates otherwise. Further, the term "or" is used in the present application to mean the disjunctive "or" or the conjunctive "and." It should also be understood that wherever in the present application the term comprising or including (or a term of similar scope) is recited in connection with the description of any embodiment or part thereof, a corresponding embodiment or part thereof reciting instead the term consisting essentially of or the term consisting of (or a term of similar scope) is also disclosed.
[0204] All publications, patents, patent applications and other documents cited in this application are hereby incorporated by reference in their entireties for all purposes.
[0205] While various specific embodiments have been illustrated and described, it will be appreciated that various changes can be made without departing from the spirit and scope of the invention(s). Moreover, features described in connection with one embodiment of the invention may be used in conjunction with other embodiments, even if not explicitly exemplified in combination within.
* * * * *








D00000

D00001

D00002

D00003

D00004

D00005
D00006
D00007
D00008

D00009

D00010

D00011
D00012
D00013
D00014

D00015

D00016
D00017
D00018

D00019

D00020
D00021
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.