Implantable Devices
ROEDER; Ryan K. ; et al.
U.S. patent application number 16/134453 was filed with the patent office on 2021-01-07 for implantable devices. The applicant listed for this patent is Gabriel L. CONVERSE, Ryan K. ROEDER, Stephen M. SMITH. Invention is credited to Gabriel L. CONVERSE, Ryan K. ROEDER, Stephen M. SMITH.
| Application Number | 20210000611 16/134453 |
| Document ID | / |
| Family ID | |
| Filed Date | 2021-01-07 |





| United States Patent Application | 20210000611 |
| Kind Code | A9 |
| ROEDER; Ryan K. ; et al. | January 7, 2021 |
IMPLANTABLE DEVICES
Abstract
Implantable devices for orthopedic, including spine and other uses are formed of porous reinforced polymer scaffolds. Scaffolds include a thermoplastic polymer forming a porous matrix that has continuously interconnected pores. The porosity and the size of the pores within the scaffold are selectively formed during synthesis of the composite material, and the composite material includes a plurality of reinforcement particles integrally formed within and embedded in the matrix and exposed on the pore surfaces. The reinforcement particles provide one or more of reinforcement, bioactivity, or bioresorption.
| Inventors: | ROEDER; Ryan K.; (Granger, IN) ; CONVERSE; Gabriel L.; (Lafayette, IN) ; SMITH; Stephen M.; (South Bend, IN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Prior Publication: |
|
||||||||||
| Appl. No.: | 16/134453 | ||||||||||
| Filed: | September 18, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14078614 | Nov 13, 2013 | |||
| 16134453 | ||||
| 12039666 | Feb 28, 2008 | |||
| 14078614 | ||||
| 60939256 | May 21, 2007 | |||
| 60904098 | Feb 28, 2007 | |||
| Current U.S. Class: | 1/1 |
| International Class: | A61F 2/44 20060101 A61F002/44; A61L 27/56 20060101 A61L027/56; B29C 67/20 20060101 B29C067/20; A61F 2/28 20060101 A61F002/28; A61L 27/54 20060101 A61L027/54; A61L 27/26 20060101 A61L027/26; A61L 27/18 20060101 A61L027/18; A61L 27/12 20060101 A61L027/12; C08J 9/36 20060101 C08J009/36; C08J 9/28 20060101 C08J009/28; C08J 9/00 20060101 C08J009/00; A61L 27/46 20060101 A61L027/46 |
Claims
1. An implantable device comprising: at least two regions comprising, (a) a central region, and (b) an outer region, at least one of the two regions comprising a porous reinforced composite scaffold material that comprises a thermoplastic polymer matrix, and a plurality of reinforcement particles distributed throughout the thermoplastic polymer matrix, and a substantially continuously interconnected plurality of pores that are distributed throughout the thermoplastic polymer matrix, each of the plurality of pores defined by voids interconnected by struts, and the other of the at least two regions comprising one of (i) a porous reinforced composite scaffold material that comprises a thermoplastic polymer matrix, and a plurality of reinforcement particles distributed throughout the thermoplastic polymer matrix, and a substantially continuously interconnected plurality of pores that are distributed throughout the thermoplastic polymer matrix, each of the plurality of pores defined by voids interconnected by struts, and (ii) a non-porous reinforced composite material that comprises a thermoplastic polymer matrix, and a plurality of reinforcement particles distributed throughout the thermoplastic polymer matrix wherein the porosity of the central region is different from the porosity of the outer region.
2. An implantable device according to claim 1, wherein porosity of the two regions varies radially from the central region to the outer region.
3. An implantable device according to claim 2, the central region comprising a porous reinforced composite scaffold material that comprises a thermoplastic polymer matrix, and a plurality of reinforcement particles distributed throughout the thermoplastic polymer matrix, and a substantially continuously interconnected plurality of pores that are distributed throughout the thermoplastic polymer matrix, each of the plurality of pores defined by voids interconnected by struts.
4. An implantable device according to claim 3, wherein the outer region is dense and essentially non-porous, and wherein the central region comprises a central void.
5. An implantable device according to claim 1, wherein the device comprises an interbody spinal fusion cage, and wherein the device comprises one or more of a radiographic marker, a hole, a notch, a pin, roughened surface on all or a portion of the device, a gripping feature for positioning of the implantable device by surgical tools, a central void of any size or shape, and a carrier material containing a growth factor agent incorporated into a void space.
6. An implantable device according to claim 1, wherein one or more of reinforcement particle volume fraction, reinforcement aspect ratio, reinforcement size, reinforcement orientation, polymer, porosity volume fraction, pore size, and pore shape varies within a region or varies from the central region to the outer region.
7. An implantable device according to claim 1, wherein reinforcement particles are both embedded within the thermoplastic polymer matrix and exposed on the struts within the pore voids.
8. An implantable device according to claim 1, wherein the reinforcement particles are present from about 20 to about 40 percent by volume, based on the volume of the thermoplastic polymer matrix, and wherein the reinforcement particles are selected from isometric, and anisometric having a mean aspect ratio (length along c-axis/length along a-axis) of greater than 1 and less than 100, and wherein the size of the reinforcement particles ranges from about 20 nm-100 .mu.m.
9. An implantable device according to claim 1, wherein the reinforcement particles comprise one or more of hydroxyapatite, calcium-deficient hydroxyapatite, carbonated calcium hydroxyapatite, beta-tricalcium phosphate (beta-TCP), alpha-tricalcium phosphate (alpha-TCP), amorphous calcium phosphate (ACP), octacalcium phosphate (OCP), tetracalcium phosphate, biphasic calcium phosphate (BCP), anhydrous dicalcium phosphate (DCPA), dicalcium phosphate dihydrate (DCPD), anhydrous monocalcium phosphate (MCPA), monocalcium phosphate monohydrate (MCPM), and combinations thereof.
10. An implantable device according to claim 1, wherein the pores within at least one of the regions have a size within the range from about 10 to about 500 .mu.m.
11. An implantable device according to claim 1, wherein the pores within at least one of the regions have a size within the range from and including about 250 to about 500 .mu.m.
12. An implantable device according to claim 1, wherein the porosity of the thermoplastic polymer matrix in at least one of the regions ranges from about 40 to about 90 percent by volume, based on the volume of the porous reinforced composite scaffold material.
13. An implantable device according to claim 1, wherein the porosity of the thermoplastic polymer matrix in at least one of the regions ranges from about 60 to about 80 percent by volume, based on the volume of the porous reinforced composite scaffold material.
14. An implantable device according to claim 1, wherein the thermoplastic polymer comprises one or a combination a biodegradable polymer, and a non-degradable polymer that comprises polyaryletherketone, polyetheretherketone, polyetherketonekteone, polyetherketone, polyethylene, high density polyethylene, ultra high molecular weight polyethylene, low density polyethylene, polyethylene oxide, polyurethane, polypropylene, polypropylene oxide, polysulfone, polymethylmethacrylate, and other polyacrylics from monomers such as bisphenol a hydroxypropylmethacrylate (bis-GMA) and/or tri(ethylene glycol) dimethacrylate, polypropylene, poly(DL-lactide), poly(L-lactide), poly(glycolide), poly(c-caprolactone), poly(dioxanone), poly(glyconate), poly(hydroxybutyrate), poly(hydroxyvalerate, poly(orthoesters), poly(carboxylates), poly(propylene fumarate), poly(phosphates), poly(carbonates), poly(anhydrides), poly(iminocarbonates), poly(phosphazenes), copolymers thereof, or blends thereof
15. A method of forming an implantable device, the method comprising: forming a porous reinforced composite scaffold material that comprises a thermoplastic polymer matrix according to the steps including: i. providing at least one thermoplastic polymer powder; reinforcement particles; and at least one porogen material; ii. preparing at least one mixture comprising thermoplastic polymer powder, reinforcement particles and porogen material; iii. consolidating the mixture; iv. densifying the mixture to form a reinforced composite scaffold preform; v. molding the reinforced composite scaffold preform to form a reinforced composite scaffold material; and vi. exposing the reinforced composite scaffold material to a leaching process to remove the porogen to form the porous reinforced composite scaffold material.
16. A method of forming an implantable device according to claim 15, wherein the at least one mixture includes one or more of: a plurality of thermoplastic polymer powders, a plurality of different reinforcement particles that vary by one or more of size, shape, and composition, a plurality of different porogen materials that vary by one or more of size, shape, and composition, and combinations of these.
17. A method of forming an implantable device according to claim 15, wherein step ii includes preparing more than one mixture comprising thermoplastic polymer powder, reinforcement particles and porogen material, wherein each of the more than one mixture differs from the other as including one or more of: different thermoplastic polymer powders, different reinforcement particles that vary by one or more of size, shape, and composition, different porogen materials that vary by one or more of size, shape, and composition, and combinations of these.
18. A method of forming an implantable device according to claim 15, wherein the step of mixing the polymer powder and the particles includes mixing with a fluid, and wherein the step of consolidating the mixture includes drying.
19. A method of forming an implantable device according to claim 15, wherein the step of densifying the mixture includes uniaxial compression.
20. A method of forming an implantable device according to claim 16, wherein the step of molding the preform includes one or more of injection molding, reaction injection molding, compression molding, transfer molding, extrusion, blow molding, pultrusion, casting, potting, and solvent casting.
21. A method of forming an implantable device according to claim 20, wherein the step of molding the preform includes one or more of compression molding and sintering the preform at a temperature within the range from about 360.degree. C. to about 380.degree. C.
22. A method of forming an implantable device according to claim 15, wherein steps iii-v are achieved by compression molding the mixture.
23. A method of forming an implantable device according to claim 15, wherein the steps ii-v are achieved by extruding the mixture.
24. A method of forming an implantable device according to claim 15, comprising selectively varying mechanical properties of the implant device by varying one or more of: the reinforcement particle volume fraction, reinforcement aspect ratio, reinforcement size, reinforcement orientation, the thermoplastic polymer powder, the porogen material, porogen size, porogen volume fraction, and porogen shape.
25. A method of forming an implantable device according to claim 15, wherein the formed porous composite scaffold material is characterized in having reinforcement particles that are both embedded within the thermoplastic polymer matrix and exposed on the struts within pore voids.
26. An implantable spinal fusion device comprising: (a) a central region having a central void and comprising a porous reinforced composite scaffold material that comprises a thermoplastic polymer matrix comprising one of polyaryletherketone, polyetheretherketone, polyetherketonekteone, polyetherketone, and a plurality of anisometric reinforcement particles comprising calcium phosphate distributed throughout the thermoplastic polymer matrix, and a substantially continuously interconnected plurality of pores that are distributed throughout the thermoplastic polymer matrix, each of the plurality of pores defined by voids interconnected by struts, wherein reinforcement particles are both embedded within the thermoplastic polymer matrix and exposed on the struts within the pore voids, wherein the porosity of the thermoplastic polymer matrix ranges from about 60 to about 80 percent by volume, based on the volume of the porous reinforced composite scaffold material, and (b) an outer region comprising one of (i) a porous reinforced composite scaffold material that comprises a thermoplastic polymer matrix, and a plurality of anisometric reinforcement particles comprising calcium phosphate distributed throughout the thermoplastic polymer matrix comprising one of polyaryletherketone, polyetheretherketone, polyetherketonekteone, polyetherketone, and a substantially continuously interconnected plurality of pores that are distributed throughout the thermoplastic polymer matrix, each of the plurality of pores defined by voids interconnected by struts, wherein reinforcement particles are both embedded within the thermoplastic polymer matrix and exposed on the struts within the pore voids, wherein the porosity of the thermoplastic polymer matrix in at least one of the regions ranges from about 60 to about 80 percent by volume, based on the volume of the porous reinforced composite scaffold material, and (ii) a non-porous reinforced composite material that comprises a thermoplastic polymer matrix, and a plurality of anisometric reinforcement particles comprising calcium phosphate distributed throughout the thermoplastic polymer matrix wherein the porosity of the two regions varies radially from the central region to the outer region, and wherein the outer region is relatively less porous and more dense than the central region.
Description
RELATED APPLICATIONS
[0001] This application is a continuation in part of and claims priority to U.S. patent application Ser. No. 14/078,614 which is a continuation of and claims priority to U.S. patent application Ser. No. 12/039,666 which claims the benefit of U.S. Provisional Patent Application Ser. Nos. 60/904,098, filed on Feb. 28, 2007 and 60/939,256, filed on May 21, 2007 all of which are incorporated herein in their entireties.
FIELD OF THE DISCLOSURE
[0002] The present disclosure relates generally to composite biomaterials and more particularly to porous composite biomaterials and related methods.
BACKGROUND
[0003] Natural bone grafts such as autogenous bone grafts (autografts) are commonly used in procedures for repairing or replacing bone defects because they provide good structural support and osteoinductivity. Natural bone grafts involve removing or harvesting tissue from another part of a host's body (e.g., typically from the iliac crest, hip, ribs, etc.) and implanting the harvested tissue in the defect site. Not only do these grafts require an added surgical procedure needed to harvest the bone tissue, these grafts have limitations, including for example, transplant site morbidity. One alternative to autografts is allografts, which involve removing and transplanting tissue from another human (e.g., from bone banks that harvest bone tissue from cadavers) to the defect site. However, allografts are known to induce infection and immunotoxicity, suffer from limited supply and variability, and have a lessened effectiveness because the cells and proteins that promote bone growth are lost during the harvesting process (e.g., during cleansing and disinfecting process). Demineralized bone matrix (DBM) is typically used to induce bone growth at defect sites, but DBM lacks the mechanical properties (e.g., stiffness, strength, toughness, etc.) necessary to be considered a viable option for load-bearing applications.
[0004] Synthetic bone substitute materials have been researched in the treatment of diseased bone (e.g., osteoporosis), injured bone (e.g., fractures), or other bone defects in lieu of natural bone grafts. Synthetic bone substitutes are viable alternatives to the more traditional methods described above. However, synthetic substitute materials used to repair diseased bones and joints should function or perform biologically and mechanically (i.e., as the structural support role of the bone itself) by, for example, mimicking the density and overall physical structure of natural bone to provide a framework for ingrowth of new tissue. One type or application of a synthetic bone substitute is a scaffold, which provides support for bone-producing cells. Scaffolds may be biodegradable, which degrade in vivo, or they may be non-biodegradable to provide permanent implant fixation (e.g., spinal fusion cages). In addition, scaffolds are typically biocompatible, and some may be bioactive, bioresorbable, osteoconductive, and/or osteoinductive. The shapability, deliverability, cost, and ability to match the mechanical properties of the surrounding host tissue are other factors that vary among different types of scaffolds and other bone substitutes.
[0005] Problems may arise when there is a mechanical mismatch between the bone substitute and the surrounding tissue. For example, metallic implants and dense ceramics have mechanical properties that are typically an order of magnitude greater than the bone tissue. As a result, a stiff metal bone substitute implant acts to "shield" the adjacent bone tissue from mechanical stresses, resulting in a weakened bone at the bone-implant interface. Furthermore, efforts to utilize porous ceramics or polymer bone cement in place of stiffer materials have been limited. For example, ceramics possess low fracture toughness, thereby making the orthopedic implant brittle (i.e., susceptible to fracture). Polymers are limited by higher compliance and lower strength, thereby limiting their ability to support physiological load levels. Additionally, conventional orthopedic implant biomaterials are not osteoconductive and bioactive, resulting in a lack of bonding between the implant and the peri-implant tissue.
BRIEF DESCRIPTION OF THE DRAWINGS
[0006] FIG. 1A is a perspective view of an example porous composite material described herein.
[0007] FIG. 1B is a cross-sectional view of a portion of the example porous composite material of FIG. 1A.
[0008] FIG. 2A, 2B and 2C are scanning electron micrographs of a portion of the example porous composite material shown in increasing magnification.
[0009] FIGS. 3 is graphical representation of the elastic modulus of example apatite reinforced polymer composites versus the reinforcement volume fraction.
[0010] FIG. 4 shows scanning electron micrograph of a portion of the surface of an example composite material reinforced with calcium phosphate whiskers embedded within and exposed on the surface, and schematically showing the orientation of the whiskers relative to the loading direction of the material or scaffold strut.
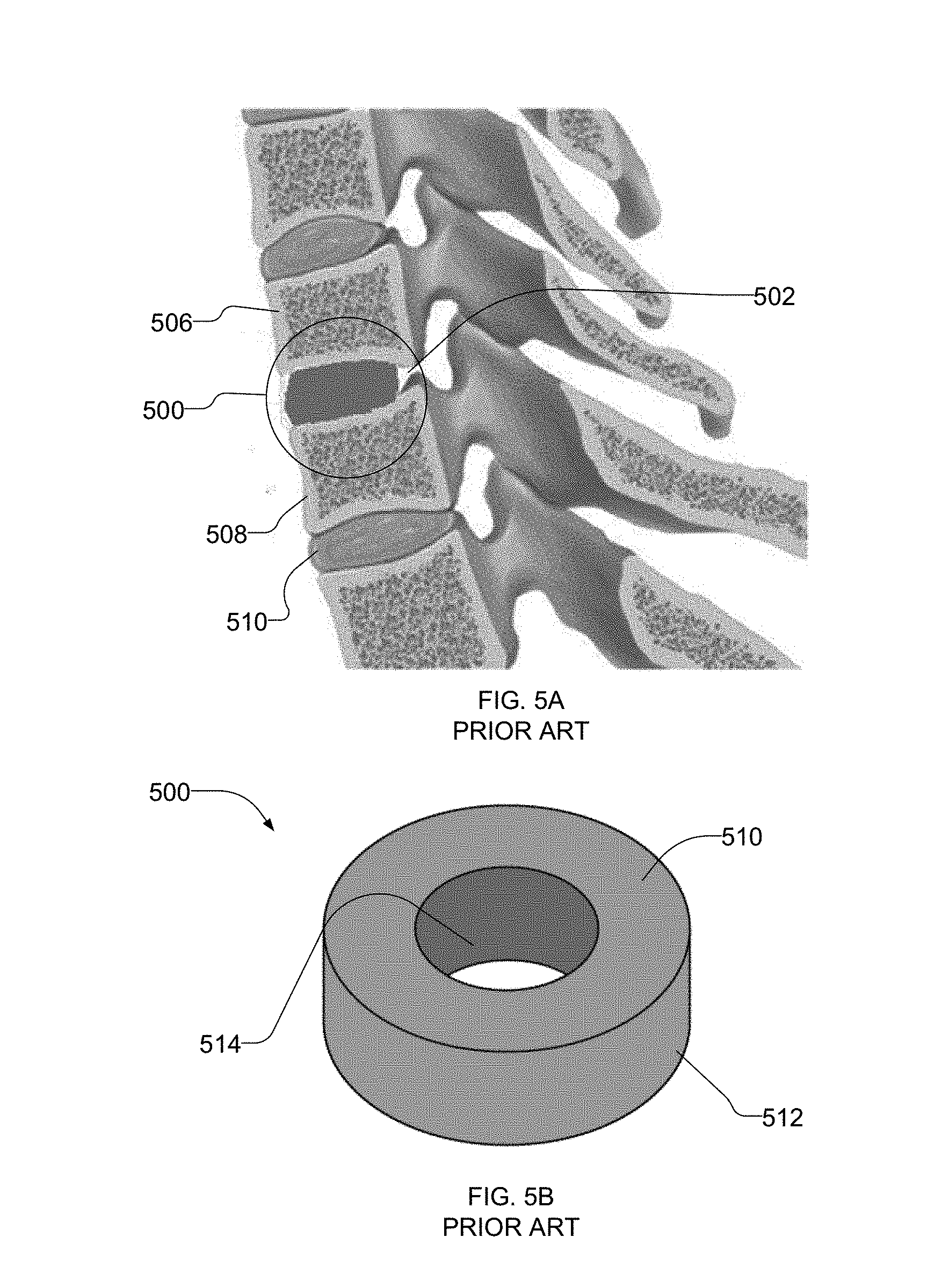
[0011] FIG. 5A is a schematic illustration of a known spinal fusion cage inserted between spinal vertebrae.
[0012] FIG. 5B illustrates the example known spinal fusion cage of FIG. 5A.
[0013] FIG. 6 illustrates another example of an implant comprising a scaffold described herein.
[0014] FIG. 7 illustrates yet another example of an implant comprising a scaffold described herein.
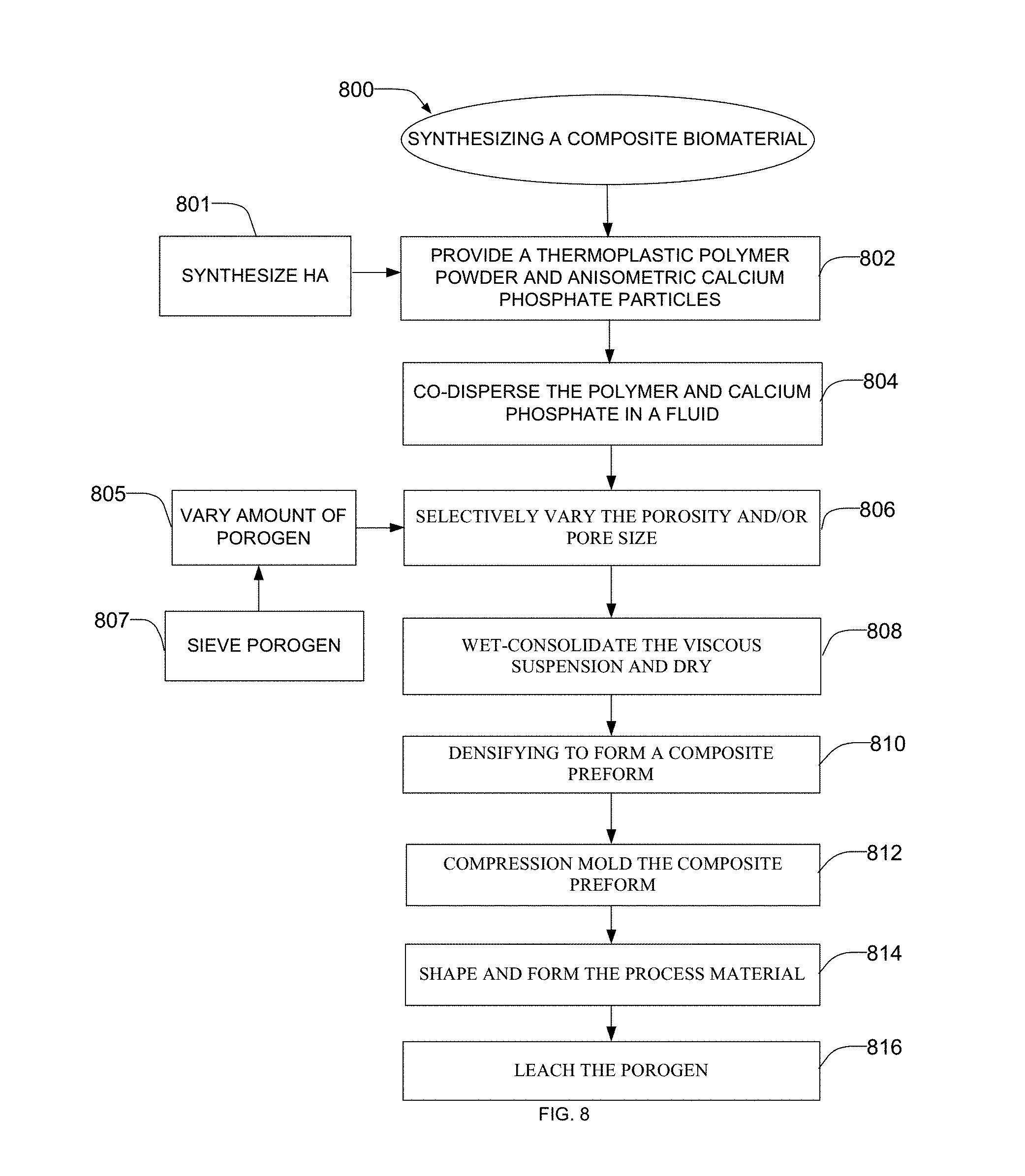
[0015] FIG. 8 is a flow diagram illustrating an example process of creating an example composite material apparatus described herein.
DETAILED DESCRIPTION
[0016] In general, the example methods, apparatus, and materials described herein provide a biocompatible, bioactive synthetic porous composite for use as synthetic bone substitute materials. The synthetic composite may provide a synthetic porous scaffold for use in an orthopedic implant and/or be injectable via percutaneous or surgical injection to cure in vivo. Because the example composite material is used to form a scaffold or matrix that is used in an implantable device, the descriptions of one or more of these structures may also describe one or more of the other structures. The synthetic porous composites are tailored to mimic biological and mechanical properties of bone tissue for implant fixation, synthetic bone graft substitutes, tissue engineering scaffolds, interbody spinal fusion, or other orthopedic applications. An example porous composite material described herein reduces subsidence and/or bone resorption resulting from mechanical mismatch problems between a synthetic scaffold of an implant device and the peri-implant tissue. Additionally, porosity and/or the pore sizes of the example synthetic composite are tailorable to specific applications to effectively promote the vascularization and growth of bone in the pores and/or void spaces of the example scaffolds, thereby improving bonding between the scaffolds and peri-implant tissue.
[0017] The example composite material or scaffolds are synthesized or made through a process that enables reinforcement particles to be integrally formed with or embedded within polymer matrices. In this manner, the polymer matrices embedded with the reinforcement material provide improved material properties (e.g., stiffness, fatigue strength, and toughness). The reinforcement particles are also exposed on a surface of the matrices, which promotes bioactivity and/or bioresorption. Additionally, the process provides flexibility to tailor the level of reinforcement particles and porosity for a desired application. For example, a porogen material may be used to vary the porosity, while the pore size is tailored by, for example, sieving the porogen to a desired size.
[0018] By varying the volume of the reinforcement particles and the porosity of the example scaffold, the mechanical properties (e.g., stiffness, strength, toughness, etc.) of the example scaffold of the implant device may be tailored to match those of the adjacent peri-implant bone tissue to reduce mechanical mismatch problems. Reducing mechanical mismatch provides a decreased risk of subsidence, stress shielding, bone resorption, and/or subsequent failure of adjacent peri-implant bone tissue. Additionally, the example scaffold of the implant device may include a significantly high porosity to promote bone ingrowth, while exhibiting significantly higher effective mechanical properties such as, for example, the mechanical properties of trabecular bone.
[0019] In particular, the example composite material includes a continuous porous biocompatible matrix having a thermoplastic polymer matrix reinforced with anisometric calcium phosphate particles. More specifically, in one example, a composite material includes a polyetheretherketone (PEEK) or a polyetherketoneketone (PEKK) matrix reinforced with various volume fractions of hydroxyapatite (HA) whiskers (e.g., 20 or 40 volume percent), wherein the matrix is approximately between and including 1% and 95%, and in some embodiments between and including 40% and 90%, and in some particular embodiments between and including 70% and 90% porous. In another example, the porous matrix includes a biocompatible, microporous polymer cage reinforced with anisometric calcium phosphate particles and bone morphogenic protein (BMP) such as, for example, rhBMP-2, which can be dispersed or accommodated by the void spaces and/or pores of the example porous scaffold and/or exposed on the surface of the example porous scaffold. Additionally, the BMP binds to the calcium phosphate further localizing the BMP to the surface of the scaffold or matrix.
[0020] The example composite materials described herein may be used for applications such as, for example, synthetic bone graft substitutes, bone ingrowth surfaces applied to existing implants, tissue engineering scaffolds, interbody spinal fusion cages, etc. In each of the applications, carrier materials (e.g., collagen, hydrogels, etc.) containing growth factors, such as BMP, may be incorporated into the pore space of the scaffold of the implant device to further enhance osteoinduction and/or osteoconduction to promote osteointegration.
[0021] Human bone tissues exhibit substantial variation in mechanical properties depending on the tissue density and microstructure. The properties are highly dependent on anatomic location and apparent density of the measured specimen. For example, a femur includes a cortical bone that has a relative porosity on the order of about 5-15%, and a trabecular bone that has a porosity on the order of about 75-95%. Due to the highly significant porosity differences, the trabecular bone exhibits significantly lower effective mechanical properties compared to the cortical bone. Therefore, depending on the application, synthetic composite materials for use as scaffolds and/or spinal fusion cages or other implant devices should possess the mechanical properties exhibited by the cortical bone or the trabecular bone, but must also have effective porosity to promote bone growth.
[0022] To avoid the mechanical mismatch problems, such as stress shielding, the example scaffold of the implant device described herein may be tailored to substantially match or mimic the mechanical properties (e.g., stiffness, strength, toughness, etc.) of the adjacent and/or substituted bone tissue. Several factors may be varied during the synthesis of the composite material and scaffold of the implant device to tailor the mechanical properties including the calcium phosphate reinforcement volume fraction, aspect ratio, size and orientation; the polymer; and the size, volume fraction, shape and directionality of the void space and/or porosity. Tailoring the mechanical properties of the scaffold reduces the likelihood of mechanical mismatch leading to a decreased risk of subsidence, stress shielding, bone resorption and/or subsequent failure of adjacent vertebrae.
[0023] FIG. 1A illustrates the example synthetic porous composite material 100 described herein. FIG. 1B is a cross-sectional view of a portion of the example porous composite material 100 of FIG. 1A. The example synthetic composite material 100 provides a synthetic porous scaffold 101 for use or in as an orthopedic implant.
[0024] The example synthetic porous composite material 100 includes a porous thermoplastic polymer (e.g., a PEEK polymer) matrix 102 having anisometric calcium phosphate reinforcement particles 104 integrally formed or embedded with the matrix 102 and/or exposed on a surface of the matrix 102. In this manner, the polymer matrix 102 embedded with the reinforcement particles 104 provides high material strength, and the reinforcement particles 104 exposed on the surface of the matrix 102 promote bioactivity and/or bioresorption. The porous polymer matrix 102 includes a substantially continuous porosity and a plurality of pores 106 to enable bone ingrowth into the porous matrix 102. In addition, the matrix 102 is substantially continuously interconnected via a plurality of struts 108. Furthermore, at least one of the plurality of struts 108 may be a load-bearing strut.
[0025] FIGS. 2A-2C are scanning electron micrographs showing increasing magnification of a portion of an example scaffold 200 with struts 202. The example scaffold 200 is a PEEK scaffold reinforced with 40% by volume HA whiskers 204. FIG. 2A illustrates the architecture or matrix 206 of the scaffold 200 and FIG. 2B illustrates an enlarged portion of the struts 202. In the illustrated example, the HA whiskers 204 are integrally formed and/or embedded within the matrix 206 of the scaffold 200 for reinforcement. The HA whiskers 204 are also exposed on a surface 208 of the matrix of the scaffold 200 for bioactivity and/or bioresorption, as noted above. As shown in FIG. 2C, the HA whiskers 204 are aligned in a sheet texture and are exposed on the surface 208 of the struts 202.
[0026] The thermoplastic polymer of the example scaffolds described herein may be a biodegradable polymer for synthetic bone graft substitute applications, or nonbiodegradable for implant fixation applications. The thermoplastic polymer includes a continuous matrix of a composite material and is biocompatible and/or bioresorbable as described above. Additionally, or alternatively, the polymer may be a radiolucent polymer, bioresorbable (i.e., a material capable of being resorbed by a patient under normal physiological conditions) and/or non-bioresorbable, as desired. Further, the thermoplastic polymer matrix may include a polymer suitable for injection via percutaneous or surgical injection so that the composite material 100 cures in vivo.
[0027] Suitable non-resorbable polymers include, without limitation, polyaryletherketone (PAEK), polyetheretherketone (PEEK), polyetherketonekteone (PEKK), polyetherketone (PEK), polyethylene, high density polyethylene (HDPE), ultra high molecular weight polyethylene (UHMWPE), low density polyethylene (LDPE), polyethylene oxide (PEO), polyurethane, polypropylene, polypropylene oxide (PPO), polysulfone, polypropylene, copolymers thereof, and blends thereof. Suitable bioresorbable polymers include, without limitation, poly(DL-lactide) (PDLA), poly(L-lactide) (PLLA), poly(glycolide) (PGA), poly(c-caprolactone) (PCL), poly(dioxanone) (PDO), poly(glyconate), poly(hydroxybutyrate) (PHB), poly(hydroxyvalerate (PHV), poly(orthoesters), poly(carboxylates), poly(propylene fumarate), poly(phosphates), poly(carbonates), poly(anhydrides), poly(iminocarbonates), poly(phosphazenes), copolymers thereof, and blends thereof. Suitable polymers that are injectable via percutaneous or surgical injection that cure in vivo include, without limitation, polymethylmethacrylate (PMMA), and other polyacrylics from monomers such as bisphenol a hydroxypropylmethacrylate (bis-GMA) and/or tri(ethylene glycol) dimethacrylate (TEG-DMA).
[0028] Although synthetic substitute composite materials made of polymers satisfy the functional criteria of an implantable device because they are, for example, formable and inexpensive, polymers alone lack biological efficacy to promote bone growth and/or may lack requisite mechanical properties to support load levels. To increase the mechanical properties of the polymers, the polymers are reinforced. The reinforcement particles 104 described above may be, for example, calcium phosphate. In other possible embodiments according to the invention, the reinforced composite material may comprise other reinforcement particles, for instance particles that are other than calcium phosphate. Further, the reinforcement particles may be other than anisometric. In yet other embodiments, the reinforcement particles may comprise a blend of different particles that vary in terms of composition, size, and shape.
[0029] The aspect ratio, size, volume fraction and degree of preferred orientation of the calcium phosphate particles 104 (e.g., HA whisker particles) may be tailored for the desired material properties and implant performance. For example, consider the information presented in FIG. 3, which is a graphical illustration of the elastic modulus of HA whisker and powder reinforced polymer composite materials versus the volume fraction percentage of the apatite calcium phosphates that is mixed with the polymer matrix. The shaded areas of FIG. 3 show approximate regions for the given mechanical property of the human cortical bone tissue. As depicted in the graph, the elastic modulus of the composite materials increases with increasing HA content. In addition, increasing the level of HA reinforcement in polymer composites increases cellular activity during osteointegration.
[0030] The calcium phosphate reinforcement particles 104 may be in the form of single crystals or dense polycrystals but are at least in some portion anisometric. "Anisometric" refers to any particle morphology (shape) that is not equiaxed (e.g., spherical), such as whiskers, plates, fibers, etc. Anisometric particles are usually characterized by an aspect ratio. For example, HA single crystals are characterized by the ratio of dimensions in the c- and a-axes of the hexagonal crystal structure. Thus, the anisometric particles in the present disclosure have an aspect ratio greater than 1. In one example, the mean aspect ratio of the reinforcement particles is from greater than 1 to about 100. In accordance with the various embodiments, the mean aspect ranges from greater than 1, to 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 49, 49, 50, 60, 70, 80, 90, and up to and including 100, including increments and ranges therein and there between.
[0031] By further example, the reinforcement particles can be provided in an amount of from about 1% by volume of the composite biomaterial to about 60% by volume, based on the volume of the thermoplastic polymer matrix, and for example, from about 20% by volume to about 50% by volume. In accordance with the various embodiments, the volume of reinforcement particles present in the thermoplastic polymer matrix can range from about 1% to about 60%, from about 5% to about 55%, from about 10 to about 50%, from about 15 to about 45%, from about 15 to about 30%, from about 20 to about 40%, from about 25 to about 35%, and any suitable combination, sub-combination, range, or sub-range thereof by volume, based on the volume of the thermoplastic polymer matrix. Thus, the reinforcement particles may be present, by volume, based on the total volume of the thermoplastic polymer matrix, from about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 49, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, to about 60 volume percent, including increments and ranges therein and there between. Due to their morphology, the calcium phosphate reinforcements particles 104 may be oriented in bulk or near the surface of the polymer matrix 102 to provide directional properties, if desired. For example, if the reinforcement particles 104 are predominately aligned within the matrix 102 the morphological alignment of the particles 104 provides anisotropy for the overall composite 100, which can be tailored to be similar to the anisotropic mechanical properties of bone tissues. For example, FIG. 4 shows a micrograph of anisometric calcium phosphate reinforcement 400 on the surface of a dense composite polymer matrix 402. Also shown in FIG. 4 is a schematic illustration of a portion of matrix 402 and illustratively showing the orientation of the reinforcement particles 400 relative to the loading direction of the material and/or a scaffold strut, which is, for example, at an angle .theta..
[0032] Furthermore, there are no limits on the size or amount of the calcium phosphate particles 104 in matrix 102, provided that the calcium phosphate particles 104 are dispersed within and/or exposed at the surface of the polymer matrix 102. For example, the reinforcement particles 104 may have a maximum dimension from about 20 nm to about 2 mm, and for example, between and including 20 nm to about 100 .mu.m. While both nano- and micro-scale calcium phosphate particles improve the mechanical properties of the example synthetic composite material 100 described herein, nano-scale calcium phosphate particles are particularly effective for enhancing bioresorbability and cell attachment, and micro-scale particles are particularly effective for obtaining a uniform dispersion within the matrix 102. Thus, the reinforcement particles may have a size from about 20 nm to about 20, 30, 40, 50, 60, 70, 80, 90, 100, 200, 300, 400, 500, 600, 700, 800, 900 nm, and to about 1 .mu.m to about 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 200, 300, 400, 500, 600, 700, 800, 900 .mu.m, and to about 1 mm and up to and including 2 mm, including increments and ranges therein and there between.
[0033] Suitable calcium phosphates may include, without limitation, calcium HA, HA whiskers, HA, carbonated calcium HA, beta-tricalcium phosphate (beta-TCP), alpha-tricalcium phosphate (alpha-TCP), amorphous calcium phosphate (ACP), octacalcium phosphate (OCP), tetracalcium phosphate, biphasic calcium phosphate (BCP), anhydrous dicalcium phosphate (DCPA), dicalcium phosphate dihydrate (DCPD), anhydrous monocalcium phosphate (MCPA), monocalcium phosphate monohydrate (MCPM), and combinations thereof. The calcium phosphate could include calcium HA, carbonated calcium HA, or beta-tricalcium phosphate (beta-TCP), etc.
[0034] As described above, a synthetic composite material 100 not only bears physiological levels of load, but also promotes oseteointegration--the direct structural and functional connection between the living bone and the surface of the load-bearing implant. The bioactive calcium phosphate particles 104 (e.g., HA whiskers) exposed on the surface of the example porous matrix 102 promote a stable bone-implant interface. Osteointegration also requires the vascularization and growth of bone into an implant via interconnected and/or continuous porosity.
[0035] Thus, the size, volume fraction, shape, and directionality of the void spaces and/or pores 106 may be tailored to optimize osteoconduction and implant mechanical properties. The pores 106 may be any size or shape, while maintaining a continuous network to promote a fusion through the formation of new bone tissue in the void spaces and/or pores 106. For example, the pores 106 may be present throughout the matrix 102 as illustrated in FIG. 1A. Also, the pores 106 may be functionally graded in any material or implant direction, for example radially as shown in FIGS. 6 and 7, from a highly porous region to a relatively dense region or may include a void space. The change in porosity from one region to another may be very distinct, for example as shown in FIGS. 6 and 7, or gradual. Furthermore, the graded change may be uniform or variant. In addition, instead of a graded change there may be a combination of materials having two or more densities of pores. The central void may be any shape or size, and may receive (e.g., be filled) a material, a structure, or the composite material 100 (i.e., the composite material graded from the porous outer surface to a dense center), thereby forming a porous outer perimeter and a dense central region. Examples are further described in connection with FIGS. 6 and 7 below. It will be appreciated by one of ordinary skill in the art that the term "void" as used in connection with a feature of an implant, for example as shown in FIG. 5B, formed with the porous scaffold refers to a cavity or hole that is other than a pore.
[0036] As discussed in greater detail below, the porosity and/or pore sizes 106 may be selectively formed by the inclusion of, for example, a porogen material during synthesis of the composite material 100. Pores sizes may range from about 100 .mu.m to about 500 .mu.m, and, for example, from about 250 .mu.m to about 500 .mu.m. The example composite biomaterial 100 may additionally contain some fraction of microporosity within scaffold struts that is less than about 10 .mu.m in size. In accordance with the various embodiments, pores present in the thermoplastic polymer matrix can each have a size that ranges from about 10 .mu.m to about 500 .mu.m, including from about 10 .mu.m to about 100 .mu.m, from about 25 to about 85 .mu.m, from about 40 .mu.m to about 65 .mu.m, and from about 100 .mu.m to about 500 .mu.m, from about 150 .mu.m to about 450 .mu.m, from about 200 .mu.m to about 400 .mu.m, from about 250 .mu.m to about 350 .mu.m, and any suitable combination, sub-combination, range, or sub-range thereof. In some thermoplastic polymer matrix includes pores having sizes that are different, wherein at least a portion of the pores has a different size than other pores, each pore having a different size within the range from about from about 10 .mu.m to about 500 .mu.m. Thus, the pores may have a size from about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 49, 49, 50, 60, 70, 80, 90, 100, 200, 300, 400 and up to and including 500 .mu.m, including increments and ranges therein and there between.
[0037] The total amount of porosity within porous regions may range up to 95%, including from about 1% to about 90% by volume, and, for example, between and including about 70% to 90% by volume. In accordance with the various embodiments, the extent of porosity in the porous reinforced composite scaffold can range from about 1% to about 95%, from about 5% to about 90%, from about 10 to about 85%, from about 15 to about 80% from about 20 to about 75%, from about 25 to about 70%, from about 30 to about 65%, from about 35 to about 60%, from about 40 to about 55%, from about 45 to about 50%, and any suitable combination, sub-combination, range, or sub-range thereof by volume, based on the volume of the porous reinforced composite scaffold. Thus, the extent of pores, by volume, based on the total volume of the porous reinforced composite scaffold, can be from about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 49, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70. 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87. 88, 89 to about 90 volume percent, including increments and ranges therein and there between. However, the porosity may also be tailored via other processes such as, for example, microsphere sintering, fiber weaving, solvent casting, electrospinning, freeze drying (lyophilization), thermally induced phase separation, gas foaming, and rapid prototyping processes such as solid freeform fabrication, robotic deposition (aka, robocasting), selective laser sintering, fused deposition modeling, three-dimensional printing, laminated object manufacturing, stereolithography, etc., or any other suitable process(es) or combination(s) thereof.
[0038] Additionally, the example composite material 100 may optionally include additives, if desired. For example, the composite material 100 may include one or more surface-active agents to enhance interfacial bonding between the reinforcement particles 104 and the polymer matrix 102. The void spaces and/or pores 106 may accommodate and deliver one or more growth factors such as, for example, BMP, to enhance osteoinductivity and/or bone regeneration. Furthermore, the void spaces and/or pores 106 may also accommodate and deliver one or more transcription factors, matrix metalloproteinases, peptides, proteins, bone cells, progenitor cells, blood plasma, bone marrow aspirate, or combinations thereof, to improve or speed bone regeneration, or resorption and replacement of the biomaterial. In some examples, the void spaces and/or pores 106 may further accommodate a carrier material that may be incorporated into the void spaces and/or pores 106. The carrier material may include, for example, a collagen sponge, membrane, or a hydrogel material to deliver the growth factor material such as, for example, the BMP. The calcium phosphate reinforcements 104 exposed on the surface of the porous matrix 102, along with the porosity, improves the retention of the BMP within the matrix 102 and at the peri-implant interface.
[0039] FIG. 5A is an illustration showing a known interbody spinal fusion cage 500. The example spinal fusion cage 500 is implanted in the inter-vertebral space 502 between two adjacent vertebrae 506 and 508. A disc 510, due to degeneration, herniation, etc., is typically removed and replaced by the spinal fusion cage 500. The spinal fusion cage 500 is used to support or restore vertebral height, and, thus, stabilize or retain adjacent vertebrae 506 and 508 in a desired position. Additionally, the spinal fusion cage 500 is to promote fusion between the vertebrae 506 and 508.
[0040] FIG. 5B is an enlarged illustration of the known spinal fusion cage 500 of FIG. 5A. A typical spinal fusion cage 500 includes a body 510 having a dense outer region 512 and a void 514 at its center. The dense outer surface 512 may be made of PEEK, titanium, or other material that can be used to support the vertebrae 506 and 508. However, the spinal fusion cage 500 made of a PEEK, titanium, etc., cannot attach to the bone. Thus, to promote bone ingrowth, the center void 514 is typically provided with a packing material (not shown) such as, a natural bone graft, a collagen sponge that retains bone growth factors, or the spinal fusion cage 500 is coated with the bone growth factors or other agents that promote osteoinduction.
[0041] The example porous scaffold 101 having the composite material 100 described herein, and with respect to FIG. 1A, can be implemented with the spinal fusion cage 500 of FIGS. 5A and 5B to replace the packing materials, such as natural bone graft. As noted above, the porous scaffold 101 promotes bone ingrowth and the pores 106 may accommodate or deliver for example, a BMP, to further improve rate of growth (fusion rate). Furthermore, the calcium phosphate (e.g., HA whisker) binds to the body 510 and localizes the BMP within the central void 514, which further promotes osteointegration.
[0042] FIG. 6 illustrates another example scaffold or matrix 600 implemented with the example composite material 100 described herein. The example scaffold 600 may be implemented as an interbody spinal fusion cage. The scaffold 600 includes a body 602 having a porous polymer matrix 604 integrally formed or embedded with anisometric calcium phosphate particles 606. Furthermore, the matrix 604 of the example scaffold 600 includes a radiolucent polymer (e.g., PEEK) integrally formed or embedded with anisometric calcium phosphate reinforcements 606 such as, for example, HA whiskers. The radiolucent polymer provides improved radiographic analysis of fusion following implantation. The example scaffold 600 may also include a BMP such as, for example, rhBMP-2. In another example, the example scaffold 600 is a biocompatible, microporous polymer scaffold or matrix supplemented with anisometric calcium phosphate reinforcements and BMP.
[0043] Additionally, the example scaffold 600 may be formed so that the pores are functionally graded in any material or implant direction, for example radially as shown in FIG. 6, from a highly porous center or central region 608 to relatively dense outer region or surface 610. The change in porosity from one region to another may be very distinct or gradual from the central region 608 to the outer region 610. Further, the graded change may be uniform or variant. The dense outer region 610 provides structural integrity along with the advantages of the composite material 100 described herein. In the illustrated example, the porous structure 604 has pore sizes that range between and including about 100 .mu.m and about 500 .mu.m, and in some embodiments, between and including about 250 .mu.m and about 500 .mu.m, and a porosity that ranges approximately between and including 1% and 95%, and in some embodiments between and including 40% and 90%, and in some particular embodiments between and including 70% and 90% porous. Furthermore, the spinal fusion cage material 600 may include microporosity having pore sizes less than about 10 .mu.m.
[0044] FIG. 7 illustrates another example scaffold 700. The porous scaffold 700 includes a porous matrix 702 having the composite material 100 described herein. The porous scaffold 700 includes a center or central void 704 that may be any shape or size. Additionally, or alternatively, the central void 704 may receive a material 706, a stem, or any other substance or structure, illustratively depicted by dashed lines. For example, the central void 704 may receive a stem 706 (e.g., an implant) such as, for example, a titanium stem, a dense composite stem (e.g., a PEEK composite stem), or any other suitable material or structure.
[0045] Additionally, in other examples, the scaffold 700 is formed so that the pores are functionally graded in any material or implant direction, for example radially as shown in FIG. 7, from a from the high porous outer region or surface 702 to a relatively dense center or central region 708. The change in porosity from one region to another may be very distinct or gradual from the central region 708 to the highly porous outer region 702. Further, the graded change may be uniform or variant. In this manner, the example scaffold 700 forms a porous perimeter having a dense core, where the material is continuous from the porous perimeter to the dense core. The dense central region 708 provides structural integrity along with the advantages of the composite material 100 described herein. In the illustrated example, the porous matrix 702 and the dense central region 708 may have pore sizes that range between and including about 100 .mu.m and about 500 .mu.m, and in some embodiments, between and including about 250 .mu.m and about 500 .mu.m, and a porosity that ranges approximately between and including 1% and 95%, and in some embodiments between and including 40% and 90%, and in some particular embodiments between and including 70% and 90% porous, and including within any of the ranges as described herein above. Furthermore, the spinal fusion cage 700 may include a microporosity having pore sizes less than about 10 .mu.m, and including within any of the ranges as described herein above.
[0046] In other examples, the composite material 100 and/or the scaffolds 101, 600, 700 may also include a roughened surface such as, for example, serrated teeth, that come into direct contact with the adjacent peri-implant tissue to prevent movement relative to the peri-implant tissue after implantation. Additionally, or alternatively, although not shown, the scaffolds 101, 600, 700 may include holes, notches, pins, radiographic markers, or other features that may be gripped or otherwise used for positioning of the implants comprising the scaffolds 101, 600, 700 by minimally invasive surgical tools and procedures.
[0047] The example composite material 100 and/or the scaffolds 101, 600, 700 may be manufactured by methods common to reinforced thermoplastic and thermosetting polymers, including but not limited to injection molding, reaction injection molding, compression molding, transfer molding, extrusion, blow molding, pultrusion, casting/potting, solvent casting, microsphere sintering, fiber weaving, solvent casting, electrospinning, freeze drying (lyophilization), thermally induced phase separation, gas foaming, and rapid prototyping processes such as solid freeform fabrication, robotic deposition (aka, robocasting), selective laser sintering, fused deposition modeling, three-dimensional printing, laminated object manufacturing, stereolithography, etc., or any other suitable process(es) or combination(s) thereof.
[0048] FIG. 8 is a flowchart of an example method 800 that may be used to synthesize the example composite material 100 and/or scaffolds 101, 600, 700 described herein. While an example manner of synthesizing the example composite material 100 and/or scaffolds 101, 600, 700 has been illustrated in FIG. 8, one or more of the steps and/or processes illustrated in FIG. 8 may be combined, divided, re-arranged, omitted, eliminated and/or implemented in any other way. Further still, the example method of FIG. 8 may include one or more processes and/or steps in addition to, or instead of, those illustrated in FIG. 8, and/or may include more than one of any or all of the illustrated processes and/or steps. Further, although the example method is described with reference to the flow chart illustrated in FIG. 8, persons of ordinary skill in the art will readily appreciate that many other methods of synthesizing the example composite material 100 and/or scaffolds 101, 600, 700 may alternatively be used.
[0049] The composite material 100 and/or the scaffolds 101, 600, 700 are processed using a powder processing approach in conjunction with compression molding and particle leaching techniques and is particularly suited for achieving a high concentration (e.g., >40 vol %) of well-dispersed (and aligned, if desired) anisometric calcium phosphate reinforcements (e.g., HA whiskers) in a thermoplastic matrix (e.g., PEEK) with minimal degradation of the calcium phosphate size/shape during processing. In this manner, the calcium phosphate reinforcement volume fraction, aspect ratio, size and orientation; the polymer; and the size, volume fraction, shape and directionality of the void space and/or porosity may be tailored to vary the mechanical properties of the composite material 100 and/or scaffolds 101, 600, 700.
[0050] A polymer such as, for example, PEEK, and anisometric calcium phosphate particles, such as HA whiskers, are provide in powder form (block 802). The PEEK polymer powder may have, for example, a mean particle size of about 26 .mu.m. The HA whiskers may be synthesized (block 801) using, for example, the chelate decomposition method.
[0051] The PEEK powder and the synthesized HA whiskers are co-dispersed in a fluid (block 804) such as, for example ethanol, and mixed (block 804) using, for example, ultrasonication under constant stirring--forming a viscous suspension.
[0052] After the polymer powder and the HA whiskers are mixed, the porosity of the mixture is selectively varied and/or tailored (block 806). In one example, the porosity may be formed and tailored by the addition of a suitable porogen material such as, for example, NaCl, wax, polysaccharides (sugars), cellulose, etc. The extent of the porosity can be controlled by varying the amount of porogen used (block 805), while the pore size could be tailored by sieving the porogen (block 807) to a desired size prior to mixing the porogen with the polymer mixture. In another examples, the porosity and/or the pore size of the polymer matrix may be selectively varied using any other suitable methods and/or process(es) such as, for example, microsphere sintering, fiber weaving, solvent casting, electrospinning, freeze drying (lyophilization), thermally induced phase separation, gas foaming, and rapid prototyping processes such as solid freeform fabrication, robotic deposition (aka, robocasting), selective laser sintering, fused deposition modeling, three-dimensional printing, laminated object manufacturing, stereolithography, etc., or any other suitable process(es) or combination(s) thereof. The viscous suspension is wet-consolidated (block 808) by, for example, vacuum filtration and drying to remove any residual fluid (i.e., ethanol). The composite mixture is densified (block 810) by, for example, uniaxial compression, to form a composite preform.
[0053] Following the initial densification, the preform is compression molded (block 812) and/or sintered at elevated temperatures (e.g., approximately 20.degree. C. to 400.degree. C.) sufficient to fuse the polymer particles with minimal damage to the calcium phosphate reinforcements. The process or composite material may be heated to a desired processing temperature and the implant may be shaped or formed (block 814). Densifying and molding the composite material includes aligning the calcium phosphate reinforcement particles (e.g., HA whiskers) morphologically and/or crystallographically within the scaffold struts. Thus, in accordance with the various embodiments, the temperature for molding is in the range (.degree. C.) from and including 20 to about 30, 40, 50, 60, 70, 80, 90, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, 200, 210, 220, 230, 240, 250, 260, 270, 280, 290, 300, 310, 320, 330, 340, 350, 360, 370, 380, 390 to 400.degree. C. including increments and ranges therein and there between. In some embodiments, as shown in the examples,
[0054] The scaffold may have any shape and/or size (e.g., any polygonal shape) and can be formed by methods common to reinforced thermoplastic and thermosetting polymers, including but not limited to injection molding, reaction injection molding, compression molding, transfer molding, extrusion, blow molding, pultrusion, casting/potting, solvent casting, and rapid prototyping processes such as solid freeform fabrication, robotic deposition (aka, robocasting), selective laser sintering, fused deposition modeling, three-dimensional printing, laminated object manufacturing, stereolithography, etc., or any other suitable process(es). The composite material 100 and/or the scaffolds 101, 600, 700, are formed by the mold walls and/or machining after molding.
[0055] The composite material undergoes a leaching process (block 816) to remove, for example, the porogen used during synthesis of the composite material. The leaching may occur, for example, via a dissolution method, heating method, and/or any other suitable methods and/or process(es). More specifically, dissolution may include immersing the scaffold in a fluid, such as, for example, deionized water.
[0056] Furthermore, viscous flow of the polymer/reinforcement mixture during molding can be designed to tailor the preferred orientation of the anisometric reinforcements in the implant. Additionally, surface-active agents may be added during the mixing process and/or to the surface of the composite material to enhance interfacial bonding between reinforcement particles and the matrix.
EXAMPLES
[0057] The following example is provided to further illustrate the example apparatus and methods described herein and, of course, should not be construed as in any way limiting in scope. It is to be understood by one of ordinary skill in the art that the following examples are neither comprehensive nor exhaustive of the many types of methods and apparatus which may be prepared in accordance with the present disclosure.
[0058] In the example, commercially available PEKK and sodium chloride (NaCl) powders with mean particle sizes of 70 and 250 .mu.m, respectively, were used as-received. HA whiskers were synthesized using the chelate decomposition method. The as-synthesized HA whiskers were measured by optical microscopy to have a mean length of 21.6 .mu.m, width of 2.8 .mu.m and aspect ratio of 7.6.
[0059] In the example, composite scaffolds with 75, 82.5 and 90% porosity were processed with 0-40 vol % HA whisker reinforcement. Appropriate amounts of polymer powder and HA whiskers were co-dispersed in ethanol via a sonic dismembrator and mechanical stirring at 1200 rpm. Following dispersion, the appropriate amount of the NaCl (i.e., porogen) was added to the suspension and mixed by hand using a Teflon coated spatula. The total scaffold volume consisted of the material volume plus the pore volume. Thus, the reinforcement level was calculated based the desired material volume, while the porosity level was calculated based on the total scaffold volume. After mixing, the viscous suspension was wet-consolidated using vacuum filtration. The powder mixture was dried overnight in a forced convection oven at 90.degree. C. and densified at 125 MPa in a cylindrical pellet die using a hydraulic platen press. The die and densified powder mixture was heated in a vacuum oven to the desired processing temperature and transferred to a hydraulic platen press for compression molding. Scaffolds with 82.5 and 90% porosity were molded at 350.degree. C., while scaffolds with 75% porosity were molded at 350, 365 and 375.degree. C. A pressure of 250 MPa was applied to the die as the polymer solidified. After cooling to room temperature, the sintered composite pellet was ejected from the die and placed approximately 300 mL deionized water for at least 72 h to dissolve the NaCl crystals. The deionized water was changed daily. The as-molded composite scaffolds had a diameter of 1 cm and were machined to a height of 1 cm.
[0060] In the example, un-confined compression tests were performed to investigate the mechanical properties of the composite scaffolds. Specimens were tested on an electromagnetic test instrument in phosphate buffered saline (PBS) at 37.degree. C. using a crosshead speed of 1 mm/min. Force-displacement data was used to calculate the elastic modulus, compressive yield stress (CYS), and failure strain of the composite scaffolds. One-way analysis of variance (ANOVA) was used to compare mechanical properties between experimental groups. The compressive properties of HA whisker reinforced PEKK scaffolds were tabulated in table 1.
[0061] The table below provides mechanical properties of the example PEKK scaffold reinforced with HA whiskers that was processed using a compression molding/particle leaching method such as, for example, the method 800 of FIG. 8 as implemented in the description above. The mechanical properties of HA whisker reinforced PEKK were evaluated in uniaxial compression. Tensile properties of the HA whisker reinforced PEKK scaffolds were evaluated prior to scaffold fabrication. As shown in the table, for a given reinforcement level, the compressive modulus decreased with increased porosity, and the yield strength decreased with increased porosity. Scaffolds with 0% vol % HA whisker reinforcement and 75% and 90% porosity exhibited moduli of 69.5 and 0.75 MPa, while scaffolds with 40 vol % HA whisker reinforcement and 75%, 82% and 90% porosity exhibited moduli of 54.0, 15.7 and 0.23 MPa, respectively. Scaffolds with 0 vol % HA whisker reinforcement and 75% and 90% porosity exhibited yield strengths of 1.25 MPa and 0.15 MPa, respectively. Scaffolds with 40 vol % HA whisker reinforcement and 75%, 82% and 90% porosity exhibited yield strengths of 0.52 MPa, 0.13 MPa and 0.04 MPa, respectively. The HA content also affected the modulus and failure strain of the scaffolds. A scaffold having 75% porosity and 20 vol % reinforcement HA whisker exhibited modulus of 106.3 MPa, compared to a modulus of 69.5 MPa for scaffolds with 0 vol % HA whisker reinforcement.
TABLE-US-00001 TABLE 1 HA Molding Elastic Failure Porosity Content Temperature Modulus CYS Strain (%) (Vol %) (.degree. C.) (MPa) (MPa) (%) 75 0 350 69.5 (12.2) 1.25 (0.16) 5.7 (0.4) 75 0 365 100.7 (10.7) 2.04 (0.26) 4.4 (1.2) 75 0 375 95.6 (10.5) 2.55 (0.34) 4.4 (1.1) 75 20 350 106.3 (15.0) 1.28 (0.13) 3.7 (1.1) 75 20 365 115.4 (13.4) 1.77 (0.30) 2.9 (0.6) 75 20 375 141.1 (39.2) 2.28 (0.35) 2.9 (0.7) 75 40 350 54.0 (18.3) 0.52 (0.17) 2.2 (0.3) 75 40 365 84.3 (41.5) 1.15 (0.38) 2.7 (0.5) 75 40 375 120.2 (29.8) 1.65 (0.34) 2.3 (1.9) 82 40 350 15.7 (6.7) 0.13 (0.03) 2.6 (1.9) 90 0 350 0.75 (0.18) 0.15 (0.04) 30 (0.0) 90 40 350 0.23 (0.13) 0.04 (0.01) 30 (0.0)
[0062] The example methods and apparatus described herein offer synthetic porous composite material that may be used for synthetic bone substitutes for implant fixation, fraction fixation, synthetic bone graft substitutes, interbody spinal fusion, tissue engineering scaffolds, or other applications. Many aspects of the of the porous composite material may be tailored to provide specific mechanical, biological, and surgical functions, such as, the polymer composition and molecular orientation, porosity and pore size of the porous matrix, or the HA reinforcement content, morphology, preferred orientation, and size.
[0063] Although the teachings of the present disclosure have been illustrated in connection with certain examples, there is no intent to limit the present disclosure to such examples. On the contrary, the intention of this application is to cover all modifications and examples fairly falling within the scope of the appended claims either literally or under the doctrine of equivalents.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.