Unit Dose Foam Applicator
Jacob; Christophe ; et al.
U.S. patent application number 16/457943 was filed with the patent office on 2020-12-31 for unit dose foam applicator. The applicant listed for this patent is ELC Management LLC. Invention is credited to Herve F. Bouix, Christophe Jacob.
| Application Number | 20200405038 16/457943 |
| Document ID | / |
| Family ID | 1000004184591 |
| Filed Date | 2020-12-31 |
| United States Patent Application | 20200405038 |
| Kind Code | A1 |
| Jacob; Christophe ; et al. | December 31, 2020 |
Unit Dose Foam Applicator
Abstract
A unit dose foam applicator according to the present invention comprises an anhydrous, water soluble personal care product for topical use. The product is contained in a pod that is comprised of water soluble film, and the pod is housed in an opened-cell foam pouch. The foam applicator is activated by water. Prior to use, it is preferable if the unit dose foam applicator is disposed in a water-impermeable packette for retail distribution and storage.
| Inventors: | Jacob; Christophe; (PRADONS, FR) ; Bouix; Herve F.; (New York, NY) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004184591 | ||||||||||
| Appl. No.: | 16/457943 | ||||||||||
| Filed: | June 29, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A45D 37/00 20130101; A45D 2040/0093 20130101; A45D 33/34 20130101; A45D 40/26 20130101; A45D 2200/1018 20130101; A45D 2200/1045 20130101; A45D 34/04 20130101 |
| International Class: | A45D 37/00 20060101 A45D037/00; A45D 33/34 20060101 A45D033/34; A45D 34/04 20060101 A45D034/04; A45D 40/26 20060101 A45D040/26 |
Claims
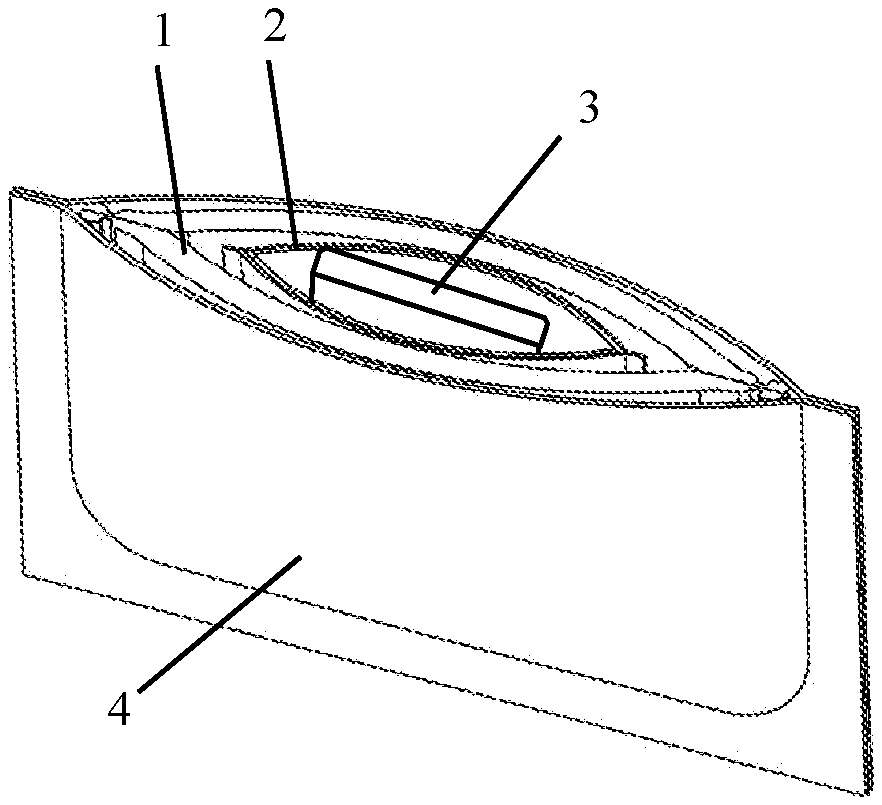
1. A unit dose foam applicator (100) comprising: an open-cell foam pouch (1), a water soluble pod (2) disposed in the foam pouch, and a water soluble anhydrous product (3) disposed in the water soluble pod.
2. A unit dose foam applicator (100) according to claim 1, wherein the foam pouch (1) comprises two sheets of foam that are bonded together along their perimeter to form a reservoir (1a) inside the foam pouch.
3. A unit dose foam applicator (100) according to claim 1, wherein the water soluble pod is made of water soluble gelatin or polyvinyl alcohol.
4. A unit dose foam applicator (100) according to claim 1, wherein the amount of product disposed in the water soluble pod is in the range of 1 mL to 20 mL.
5. A unit dose foam applicator (100) according to claim 1, disposed in a packette (4).
Description
FILED OF THE INVENTION
[0001] The invention relates to foam pads for the application of a water-soluble product to an application surface, especially for the application of topical treatments, such as cosmetic, personal care or medicinal products to skin, hair or nails.
BACKGROUND
[0002] In the personal care industry, foam pads that are formed into a pouch are known. The pouch typically houses a water-soluble cosmetic or personal care product. When water is absorbed into and through the foam pad, the water soluble product in the pouch dissolves, and diffuses back out through the foam pad until it reaches the outer surface of the pad. From there, the product may be transferred to a surface, such as skin or hair. The transfer is effected by gliding the foam pad over the surface with some amount of compression of the pad to squeeze the product out. For example, U.S. Pat. No. 5,762,946 discloses foam pads comprised of two sheets of foam of substantially identical size and contours, which are superposed edge to edge and between which there is encapsulated a solid form of a water-soluble or water-emulsifiable product (such as a cosmetic, dermatological or pharmaceutical product), the two sheets being held together by at least one weld along their periphery. The products described in this patent are in the form of a powder, solid gel or cake. Because the foam is open cell or half-open cell, powder products have a tendency to leak out through the foam before the intended use. For the same reason, this design is even less suitable for liquid or soft gel products.
[0003] Water soluble unit dose packs, sometimes called pods, are also known. These include pouches made from gelatin or from one or more water soluble films, like polyvinyl alcohol (PVOH). These have been used to contain a single dose of concentrated detergents and cleaners for industrial and home use. For example, US2003-148908 discloses an anhydrous detergent encapsulated in a water soluble film based on polyvinyl alcohol in which it is packaged as a unit dose. The films used to form pods are typically only a few thousandths of an inch thick, while the product reservoir is large enough to hold one dose of product, for example, 2 mL, 5 mL or 10 mL of product. When dropped into water or aqueous solution, the pod dissolves and releases its contents into the solution. Such unit dose packs are now common for use in a laundry washing machine or dishwasher.
OBJECTIVES
[0004] A main objective of the invention is to provide a unit dose of a topical product in a single-use, disposable foam applicator. The applicator is activated by water, and able to distribute the product on an application surface.
SUMMARY
[0005] A unit dose foam applicator according to the present invention comprises an anhydrous, water soluble personal care product for topical use. The product is contained in a pod that is comprised of water soluble film, and the pod is housed in an opened-cell foam pouch. Prior to use, it is preferable if the foam applicator is disposed in a water-impermeable packette for retail distribution and storage.
BRIEF DESCRIPTION OF THE FIGURES
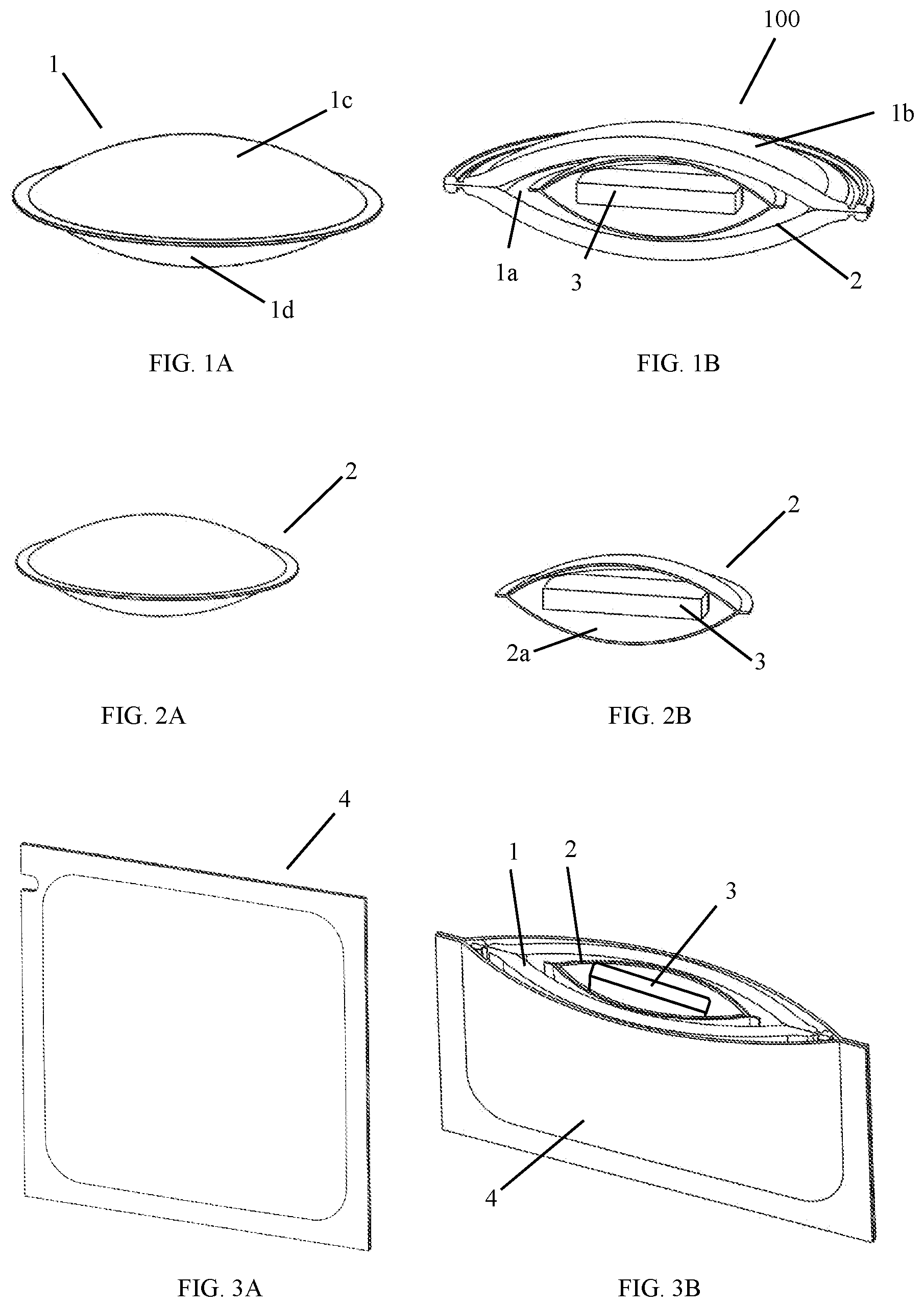
[0006] FIG. 1a depicts a unit dose foam applicator according to the invention.
[0007] FIG. 1b is a cut-away of the applicator of FIG. 1a, in which a water soluble pod can be seen.
[0008] FIG. 2a depicts a water soluble pod that houses a single dose of a water soluble anhydrous product.
[0009] FIG. 2b is a cut-away of the water soluble pod of FIG. 2a.
[0010] FIG. 3a depicts a packette in which a unit dose foam applicator according to the invention may be disposed for retail distribution and storage.
[0011] FIG. 3b is a cut-away of the packette of FIG. 3a, in which is disposed a unit dose foam applicator according to the invention.
DETAILED DESCRIPTION
[0012] The present invention is described in terms of a personal care or cosmetic product, but other types of products are also contemplated. These include topical medical treatments, such as wound care products. The invention is described in terms of application to a surface of the human body (i.e., skin, hair or nails), but other types of surfaces are also contemplated. These include any surfaces in need of cleansing and/or sterilization.
[0013] The term "comprises" means that a recitation of one or more elements may not be limited to those elements that are explicitly recited.
[0014] A unit dose foam applicator (100) according to the present invention comprises an opened-cell foam pouch (1), a water soluble pod (2) disposed in the foam pouch, and a water soluble anhydrous product (3) disposed in the water soluble pod. The unit dose foam applicator preferably comprises a water-impermeable packette (4).
[0015] The Foam Pouch
[0016] Referring to FIGS. 1A and 1B, a foam pouch (1) for use in the present invention may comprise two sheets of foam (1c, 1d) that are substantially the same size and shape. When laid one sheet on top of the other, the perimeter of the sheets may be bonded together by any suitable means, such as high frequency welding, mechanical compression, adhesive or stitching. This creates a reservoir (1a) inside the pouch, into which one or more water soluble elements may be placed prior to completing the perimeter seal. Suitable foams for this purpose include open cell foams (i.e. reticulated foams). By "open cell" foam, we mean a foam wherein more than half of the cells are open. The high concentration of open cells allows fluids to travel through the foam pouch, in either direction, which is required by the present invention. Common open cell foam materials include polyurethane, polyvinyl chloride, neoprene, polyether, polyester and open cell rubber. Closed cell foams are specifically excluded, because a liquid cannot travel through the foam. The size of the cells in the open cell foam may typically range from 0.5 mm to 5.0 mm, but the invention can be practiced with even smaller cell sizes. The two sheets of foam can be made of different materials, different densities, and have different surface features, such as flocking and color. As long as the pouch remains dry, the water soluble elements remain encapsulated within the pouch. However, when an exterior surface (1b) of the pouch is wetted, water is able to flow through the opened cell structure until it reaches the reservoir (1a). The water soluble elements within the reservoir become wet and begin to dissolve.
[0017] The foam applicator can be virtually any shape, but in terms of a circular applicator, and for the applications envisioned, a bath product applicator may range from about 50 mm to about 150 mm in diameter, and hold a quantity of the product between about 5 ml and about 15 ml. For a cleanser or makeup remover for the face, the diameter of the applicator may be from about 20 mm to about 50 mm, and the quantity of product may be from about 2 ml to about 10 ml.
The Water Soluble Pod
[0018] Water that reaches the reservoir (1a) of the pouch (1), comes into contact with the water soluble elements within the pouch. The first water soluble element that comes into contact with water is a water soluble pack or pod (2). A typical water soluble pod is made from a water soluble gelatin or a polyvinyl alcohol (PVOH) film that is only a few thousandths of an inch thick. The water soluble pod may be any shape, but is preferably the same overall shape as the foam pouch. The interior (2a) of the pod is at least large enough to hold a single dose of a water soluble anhydrous product (3). When water in the reservoir (1a) contacts the pod, the pod begins to dissolve. This exposes the water soluble product to the water in the reservoir. Preferably, in the course of a single use of the foam applicator (100), the water soluble pod completely dissolves.
The Anhydrous Product
[0019] The second water soluble element that comes into contact with water in the reservoir (1a) is the water soluble anhydrous product (3). The anhydrous product is sealed in the water soluble pod (2). Once the pod begins to dissolve, the anhydrous product comes into contact with water in the reservoir, and also begins to dissolve. Before it dissolves, the anhydrous product may be solid or liquid; particulate or gel. The only limitations on the type of product are that the product be anhydrous to a degree that does not degrade the water soluble pod before the intended time of use by a consumer. Of particular interest are substantially anhydrous color cosmetic products, such as foundations and concealers; sun care products such as SPF lotions and after-sun treatments; and skin care products that impart some benefit to the skin. The amount of product sealed in the water soluble pod may range from 1 mL to 20 mL, for example, 1 mL to 10 mL; 1 mL to 5 mL; 2 mL to 15 mL; 2 mL to 10 mL; 2 mL to 5 mL, and 5 mL to 15 mL. As the water soluble anhydrous product (3) dissolves, an aqueous solution accumulates in the reservoir (1a) of the pouch (1). Preferably, in the course of a single use of the foam applicator (100), all of the water soluble anhydrous product dissolves.
The Packette
[0020] Optionally, but preferably, each single unit dose foam applicator, as described herein, is disposed in its own packette (4) for retail distribution and storage. Preferably, the packette is substantially impermeable to water and air. A substantially water-impermeable packette will ensure that the water soluble pod (2) and water soluble anhydrous product (3) do not dissolve before the time of intended use. Thereafter any number of packettes may be packaged for retail sale. For example, the final product can be a set box comprising 10 to 30 packettes.
[0021] Alternatively, a number of single unit dose foam applicators (100) can be packaged into the same water-tight, air-tight bag for retail distribution. After the bag is opened by a consumer, the length of time that the pads are exposed to the air may be too short to adversely affect the product water soluble pod (2) or the water soluble anhydrous product (3).
Method of Use
[0022] When a user wants to use the single dose foam applicator according to the invention, she may tear open a packette, and remove the foam applicator (100). She then wets the foam pouch (1), and compresses the foam pouch with her hands to drive the water into the reservoir (1a) of the pouch. As the water soluble anhydrous product (3) dissolves, an aqueous solution accumulates in the reservoir (1a) of the pouch (1). As the foam applicator is further handled and compressed, the aqueous solution of product diffuses out of the reservoir, through the opened cell structure of the foam, until it reaches an exterior surface (1b) of the pouch. From there, the solution of product may be transferred to an application surface, such as skin, hair or nails. The transfer is effected by gliding the foam pouch over the surface with some amount of compression of the pouch to prevent the aqueous solution from flowing back into the reservoir. The process may continue until substantially all of the aqueous solution has been released from the pouch, and transferred to an application surface. The foam applicator may be used to evenly spread the product on the application surface, and to touch up or freshen a previously applied personal care product.
* * * * *
D00000

D00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.