Formulations Of Rifaximin And Uses Thereof
Selbo; Jon ; et al.
U.S. patent application number 16/916421 was filed with the patent office on 2020-12-24 for formulations of rifaximin and uses thereof. The applicant listed for this patent is Salix Pharmaceuticals, Inc.. Invention is credited to Pam Golden, Mohammed A. Kabir, Jon Selbo, Jing Teng.
| Application Number | 20200397904 16/916421 |
| Document ID | / |
| Family ID | 1000005064713 |
| Filed Date | 2020-12-24 |




View All Diagrams
| United States Patent Application | 20200397904 |
| Kind Code | A1 |
| Selbo; Jon ; et al. | December 24, 2020 |
FORMULATIONS OF RIFAXIMIN AND USES THEREOF
Abstract
The present invention relates to new rifaximin forms comprising solid dispersions of rifaximin, methods of making same and to their use in medicinal preparations and therapeutic methods.
| Inventors: | Selbo; Jon; (West Lafayette, IN) ; Teng; Jing; (West Lafayette, IN) ; Kabir; Mohammed A.; (Cary, NC) ; Golden; Pam; (Durham, NC) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005064713 | ||||||||||
| Appl. No.: | 16/916421 | ||||||||||
| Filed: | June 30, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15615121 | Jun 6, 2017 | |||
| 16916421 | ||||
| 14250293 | Apr 10, 2014 | 9737610 | ||
| 15615121 | ||||
| 13181481 | Jul 12, 2011 | |||
| 14250293 | ||||
| 61419056 | Dec 2, 2010 | |||
| 61363609 | Jul 12, 2010 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 9/2027 20130101; A61K 9/4858 20130101; A61K 9/4866 20130101; A61K 9/146 20130101; A61K 9/10 20130101; A61K 31/437 20130101; A61K 9/1641 20130101; A61K 47/32 20130101; A61K 47/38 20130101; A61K 9/1652 20130101; C07D 498/22 20130101; A61K 9/2054 20130101 |
| International Class: | A61K 47/38 20060101 A61K047/38; C07D 498/22 20060101 C07D498/22; A61K 9/14 20060101 A61K009/14; A61K 9/16 20060101 A61K009/16; A61K 9/10 20060101 A61K009/10; A61K 9/48 20060101 A61K009/48; A61K 9/20 20060101 A61K009/20; A61K 31/437 20060101 A61K031/437; A61K 47/32 20060101 A61K047/32 |
Claims
1-12. (canceled)
13. A method of treating a bowel related disorder in a subject in need thereof, comprising administering to the subject a solid dispersion comprising rifaximin and a polymer, wherein the polymer comprises from about 10% to about 60% by weight of the solid dispersion and is selected from the group consisting of polyvinylpyrrolidone (PVP) grade K-90, hydroxypropylmethyl cellulose phthalate (HPMC-P) grade 55, hydroxypropyl methylcellulose acetate succinate (HPMC-AS) grades HG and MG, and polymethacrylate, and wherein said solid dispersion comprises a non-crystalline fully miscible dispersion comprising said rifaximin and said polymer.
14. The method of claim 13, wherein the bowel related disorder is irritable bowel syndrome.
15. The method of claim 13, wherein the bowel related disorder is hepatic encephalopathy.
16. The solid dispersion of claim 13, wherein said rifaximin and said polymer are present in equal amounts, and each of said rifaximin and said polymer comprises from about 10% to about 50% by weight of said solid dispersion.
17. The solid dispersion of claim 14, wherein said rifaximin and said polymer are present in equal amounts, and each of said rifaximin and said polymer comprises from about 10% to about 50% by weight of said solid dispersion.
18. The solid dispersion of claim 15, wherein said rifaximin and said polymer are present in equal amounts, and each of said rifaximin and said polymer comprises from about 10% to about 50% by weight of said solid dispersion.
19. The solid dispersion of claim 13, wherein said polymer comprises HPMC-AS grade MG or HPMC-AS grade HG.
20. The solid dispersion of claim 14, wherein said polymer comprises HPMC-AS grade MG or HPMC-AS grade HG.
21. The solid dispersion of claim 15, wherein said polymer comprises HPMC-AS grade MG or HPMC-AS grade HG.
22. The solid dispersion of claim 16, wherein said polymer comprises HPMC-AS grade MG or HPMC-AS grade HG.
23. The solid dispersion of claim 17, wherein said polymer comprises HPMC-AS grade MG or HPMC-AS grade HG.
24. The solid dispersion of claim 18, wherein said polymer comprises HPMC-AS grade MG or HPMC-AS grade HG.
25. The solid dispersion of claim 13, wherein said non-crystalline fully miscible dispersion further comprises a non-ionic surfactant.
26. The solid dispersion of claim 14, wherein said non-crystalline fully miscible dispersion further comprises a non-ionic surfactant.
27. The solid dispersion of claim 15, wherein said non-crystalline fully miscible dispersion further comprises a non-ionic surfactant.
28. The solid dispersion of claim 16, wherein said non-crystalline fully miscible dispersion further comprises a non-ionic surfactant.
29. The solid dispersion of claim 19, wherein said non-crystalline fully miscible dispersion further comprises a non-ionic surfactant.
Description
RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 15/615,121, filed Jun. 6, 2017 which is a continuation of U.S. patent application Ser. No. 14/250,293, filed Apr. 10, 2014 which is a continuation of U.S. patent application Ser. No. 13/181,481 filed 12 Jul. 2011 which claims the benefit of U.S. Provisional Application No. 61/363,609 filed 12 Jul. 2010, and U.S. Provisional Application No. 61/419,056, filed 2 Dec. 2010, the entire contents of each of which are hereby incorporated herein by reference.
BACKGROUND
[0002] Rifaximin (INN; see The Merck Index, XIII Ed., 8304) is an antibiotic belonging to the rifamycin class of antibiotics, e.g., a pyrido-imidazo rifamycin. Rifaximin exerts its broad antibacterial activity, for example, in the gastrointestinal tract against localized gastrointestinal bacteria that cause infectious diarrhea, irritable bowel syndrome, small intestinal bacterial overgrowth, Crohn's disease, and pancreatic insufficiency among other diseases. It has been reported that rifaximin is characterized by a negligible systemic absorption, due to its chemical and physical characteristics (Descombe J. J. et al. Pharmacokinetic study of rifaximin after oral administration in healthy volunteers. Int J Clin Pharmacol Res, 14 (2), 51-56, (1994)).
[0003] Rifaximin is described in Italian Patent IT 1154655 and EP 0161534, both of which are incorporated herein by reference in their entirety for all purposes. EP 0161534 discloses a process for rifaximin production using rifamycin O as the starting material (The Merck Index, XIII Ed., 8301). U.S. Pat. No. 7,045,620 B1 and PCT Publication WO 2006/094662 A1 disclose polymorphic forms of rifaximin. There is a need in the art for formulations of rifaximin to better treat gastrointestinal and other diseases.
SUMMARY
[0004] Provided herein are solid dispersion forms of rifaximin with a variety of polymers and polymer concentrations.
[0005] In one aspect, provided herein are forms solid dispersion of rifaximin.
[0006] In one embodiment, the form solid dispersion of rifaximin is characterized by an XRPD substantially similar to one or more of the XRPDs of FIGS. 2, 7, 12, 17, 22, 31, and 36.
[0007] In one embodiment, the form solid dispersion of rifaximin is characterized by a Thermogram substantially similar to FIGS. 3-6, 8-11, 13-16, 18-21, 23-26, 27-30, and 32.
[0008] In one embodiment, the form has the appearance of a single glass transition temperature (Tg).
[0009] In one embodiment, a Tg of a form increases with an increased rifaximin concentration. In one embodiment, a form stressed at 70.degree. C./75% RH for 1 week, solids are still x-ray amorphous according to XRPD.
[0010] In one embodiment, a form stressed at 70.degree. C./75% RH for 3 weeks, solids are still x-ray amorphous according to XRPD.
[0011] In one embodiment, a form stressed at 70.degree. C./75% RH for 6 weeks, solids are still x-ray amorphous according to XRPD.
[0012] In one embodiment, a form stressed at 70.degree. C./75% RH for 12 weeks, solids are still x-ray amorphous according to XRPD.
[0013] In one aspect, provided herein are microgranules comprising one or more of the solid dispersion forms of rifaximin described herein.
[0014] In one embodiment, the microgranules further comprise a polymer.
[0015] In one embodiment, the polymer comprises one or more of polyvinylpyrrolidone (PVP) grade K-90, hydroxypropyl methylcellulose phthalate (HPMC-P) grade 55, hydroxypropyl methylcellulose acetate succinate (HPMC-AS) grades HG and MG, or a polymethacrylate (Eudragit.RTM. L100-55).
[0016] In specific embodiments, the microgranules comprises 25-75% polymer, 40-60% polymer, or 40-50% polymer. In an exemplary embodiment, the microgranules comprises 42-44% polymer.
[0017] In one embodiment, the microgranules comprise equal amounts of rifaximin and polymer.
[0018] In another embodiment, the microgranules further comprising an intragranular release controlling agent. In exemplary embodiments, the intragranular release controlling agent comprises a pharmaceutically acceptable excepient, disintegrant, crosprovidone, sodium starch glycolate, corn starch, microcrystalline cellulose, cellulosic derivatives, sodium bicarbonate, and sodium alginate.
[0019] In one embodiment, the intragranular release controlling agent comprises between about 2 wt % to about 40 wt % of the microgranule, about 5 wt % to about 20 wt % of the microgranule, or about 10 wt % of the microgranule.
[0020] In another embodiment, the intragranular release controlling agent comprises a pharmaceutically acceptable disintegrant, e.g., one selected from the group consisting of crosprovidone, sodium starch glycolate, corn starch, microcrystalline cellulose, cellulosic derivatives, sodium bicarbonate, and sodium alginate.
[0021] In another embodiment, the microgranules further comprise a wetting agent or surfactant, e.g., a non-ionic surfactant.
[0022] In one embodiment, the non-ionic surfactant comprises between about 2 wt % to about 10 wt % of the microgranule, between about 4 wt % to about 8 wt % of the microgranule, or about 5.0 wt % of the microgranule.
[0023] In one embodiment, the non-ionic surfactant comprises a poloxamer, e.g., poloxamer 407 also known as Pluronic F-127.
[0024] In another embodiment, the microgranules further comprise an antioxidant.
[0025] In exemplary embodiments, the antioxidant is butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT) or propyl gallate (PG).
[0026] In another embodiment, the antioxidant comprises between about 0.1 wt % to about 3 wt % of the microgranule or between about 0.5 wt % to about 1 wt % of the microgranule.
[0027] In another aspect, provided herein are pharmaceutical compositions comprising the microgranules described herein.
[0028] In one embodiment, the pharmaceutical compositions further comprise one or more pharmaceutically acceptable excepients.
[0029] In one embodiment, the pharmaceutical compositions are tablets or capsules.
[0030] In one embodiment, the pharmaceutical compositions comprises a disintegrant.
[0031] In one embodiment, the polymer comprises one or more of polyvinylpyrrolidone (PVP) grade K-90, hydroxypropyl methylcellulose phthalate (HPMC-P) grade 55, hydroxypropyl methylcellulose acetate succinate (HPMC-AS) grades HG and MG, or a polymethacrylate (Eudragit.RTM. L100-55).
[0032] In one aspect, provided herein are pharmaceutical solid dispersion formulations comprising: rifaximin, HPMC-AS, at a rifaximin to polymer ratio of 50:50, a non-ionic, surfactant polyol and a intragranular release controlling agent.
[0033] In one embodiment, the intragranular release controlling agent comprises about 10 wt % of the formulation.
[0034] In one aspect, provided herein are processes for producing a solid dispersion of rifaximin comprising: making a slurry of methanol, rifaximin, a polymer and a surfactant; spray drying the slurry; and blending the spray dried slurry with a intragranular release controlling agent.
[0035] In one aspect, provided herein are processes for producing a solid dispersion of rifaximin comprising: making a slurry of methanol, rifaximin, HPMC-AS MG and Pluronic F-127; spray drying the slurry; and blending the spray dried slurry with a intragranular release controlling agent.
[0036] In one embodiment, the intragranular release controlling agent comprises croscarmellose sodium.
[0037] A process for producing form solid dispersion of rifaximin comprising one or more of the methods listed in Tables 1-5.
[0038] In one embodiment, pharmaceutical compositions comprising SD rifaximin, a polymer, a surfactant, and a release controlling agent are provided. In one embodiment, provided are pharmaceutical compositions comprising SD rifaximin, HPMC-AS, pluronic F127, and croscarmellose Na (CS). In one embodiment, the pharmaceutical compositions are tablets or pills.
[0039] In additional embodiments, the pharmaceutical compositions further comprise fillers, glidants or lubricants.
[0040] In specific embodiments, the pharmaceutical compositions comprise the ratios of components set forth in Table 37.
[0041] Other embodiment and aspects are disclosed infra.
BRIEF DESCRIPTION OF THE DRAWINGS
[0042] FIG. 1. Chemical structure of Rifaximin.
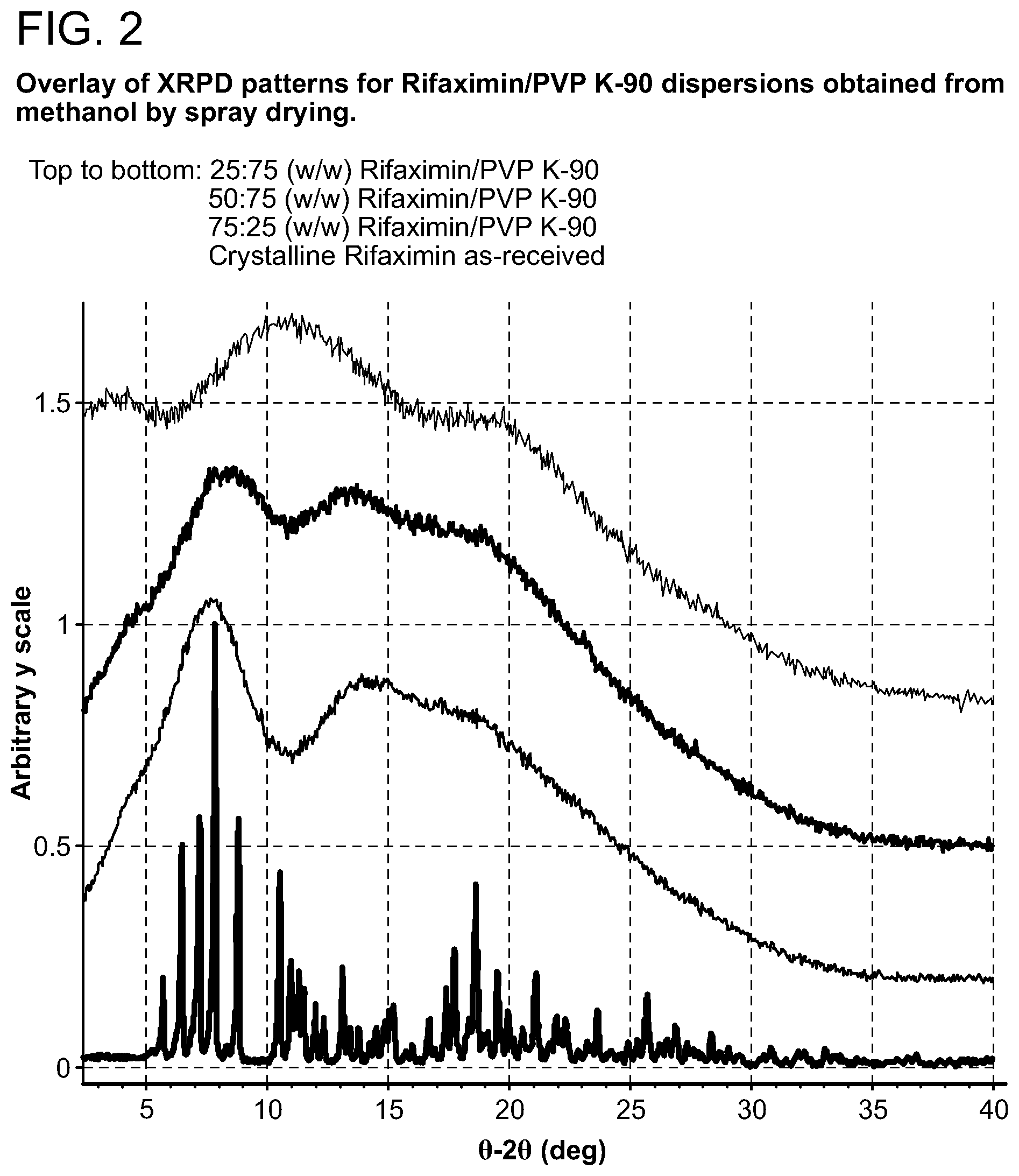
[0043] FIG. 2. Overlay of XRPD patterns for Rifaximin/PVP K-90 dispersions obtained from methanol by spray drying.
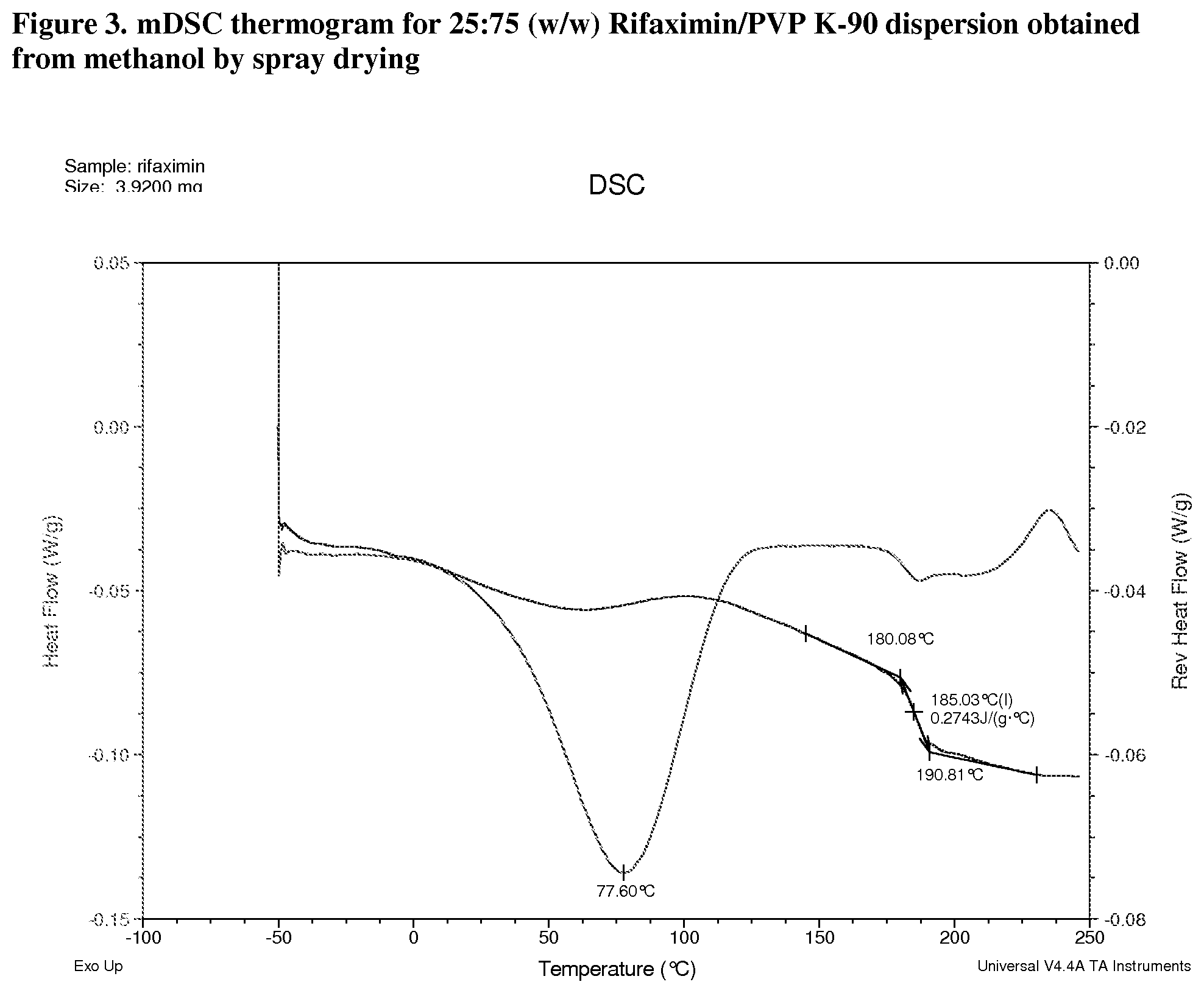
[0044] FIG. 3. mDSC thermogram for 25:75 (w/w) Rifaximin/PVP K-90 dispersion obtained from methanol by spray drying.
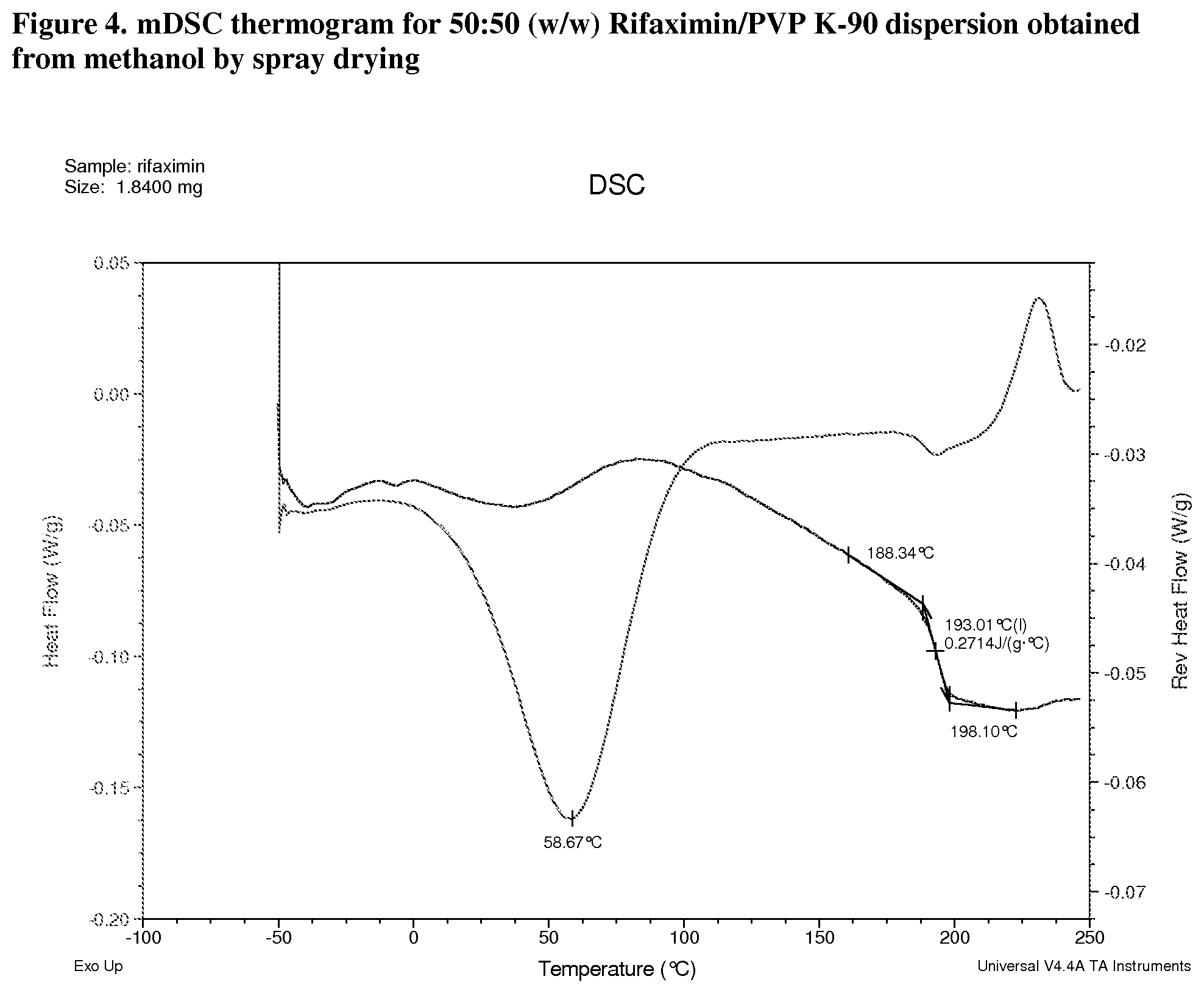
[0045] FIG. 4. mDSC thermogram for 50:50 (w/w) Rifaximin/PVP K-90 dispersion obtained from methanol by spray drying.
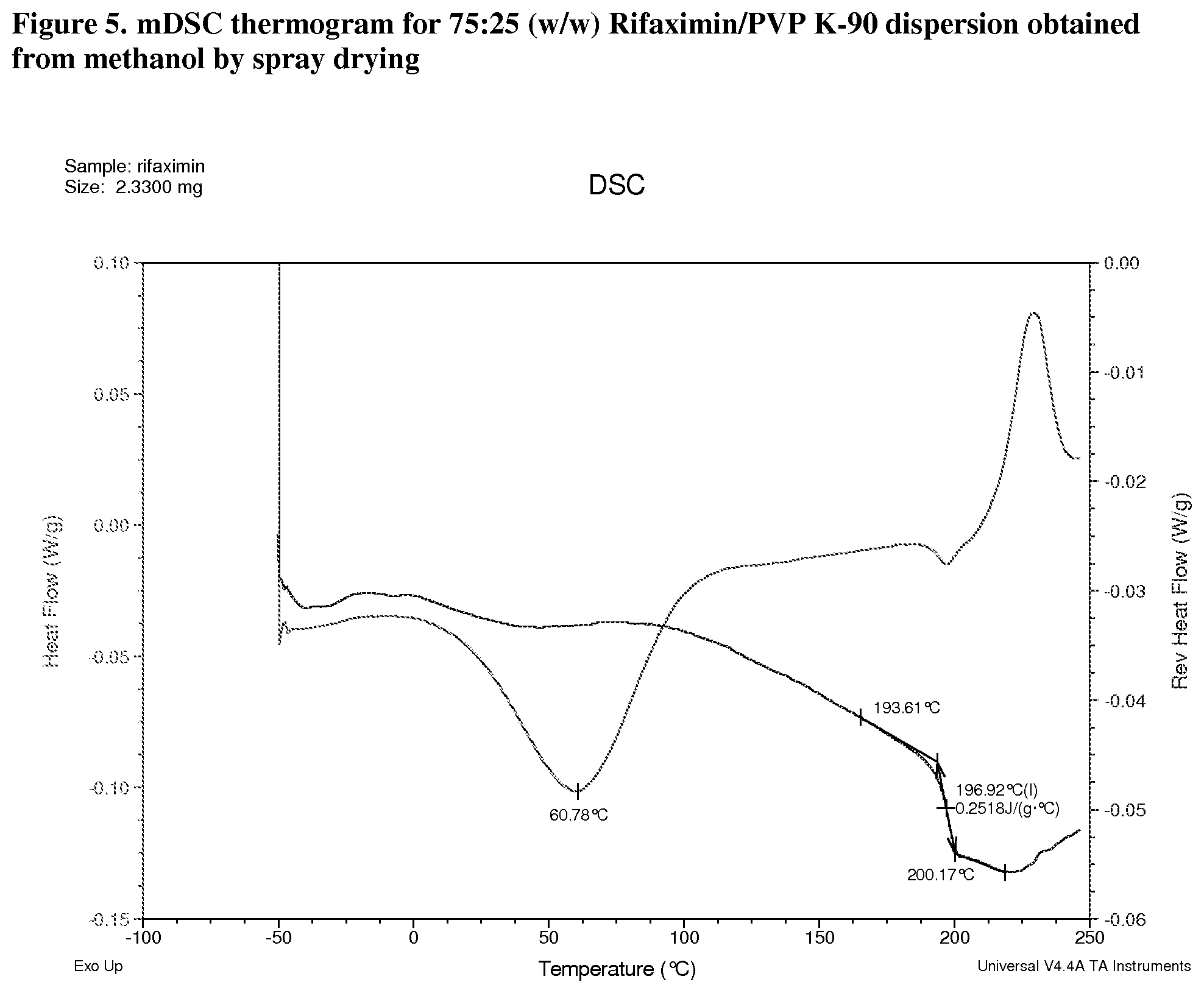
[0046] FIG. 5. mDSC thermogram for 75:25 (w/w) Rifaximin/PVP K-90 dispersion obtained from methanol by spray drying.
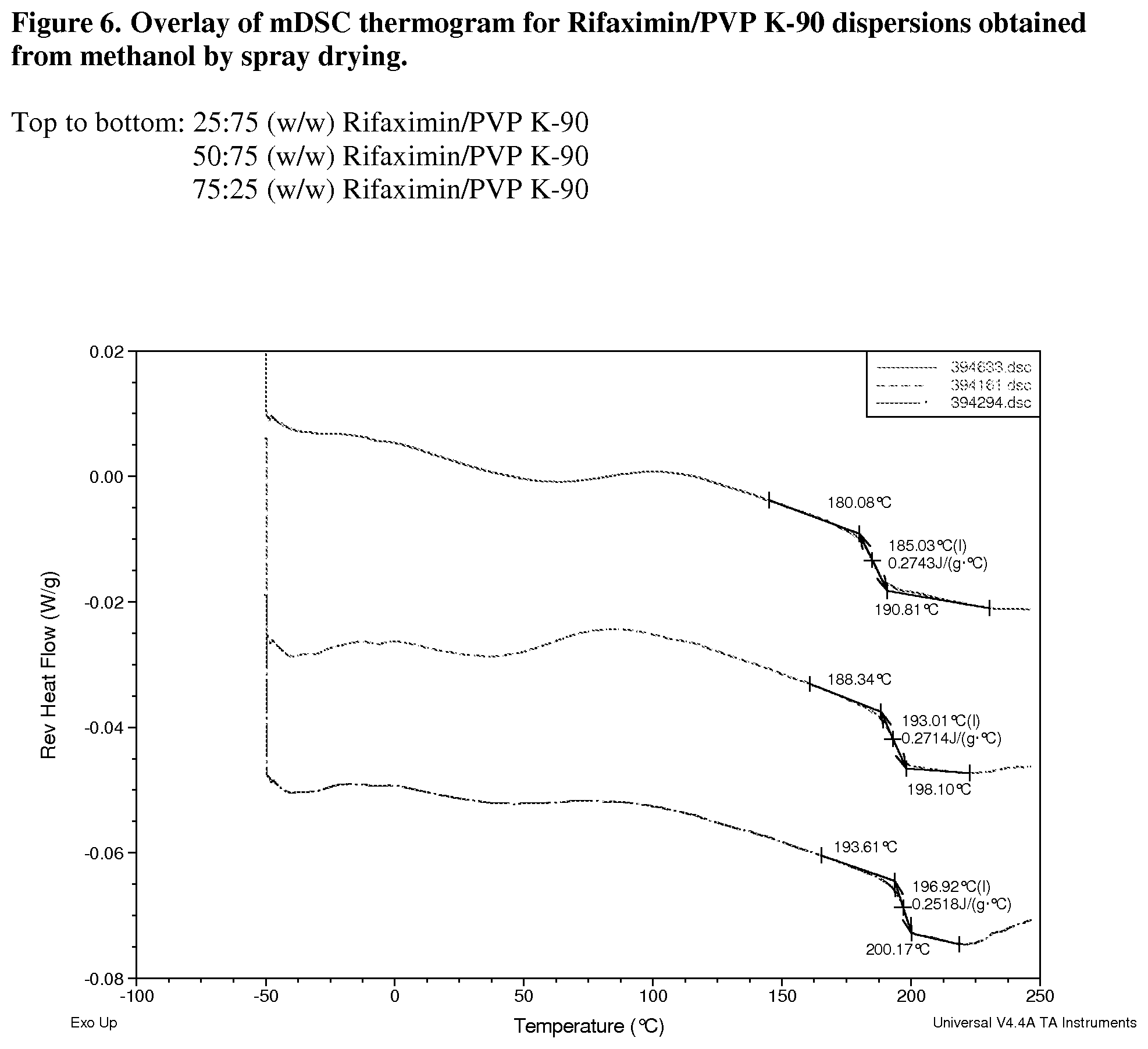
[0047] FIG. 6. Overlay of mDSC thermogram for Rifaximin/PVP K-90 dispersions obtained from methanol by spray drying.
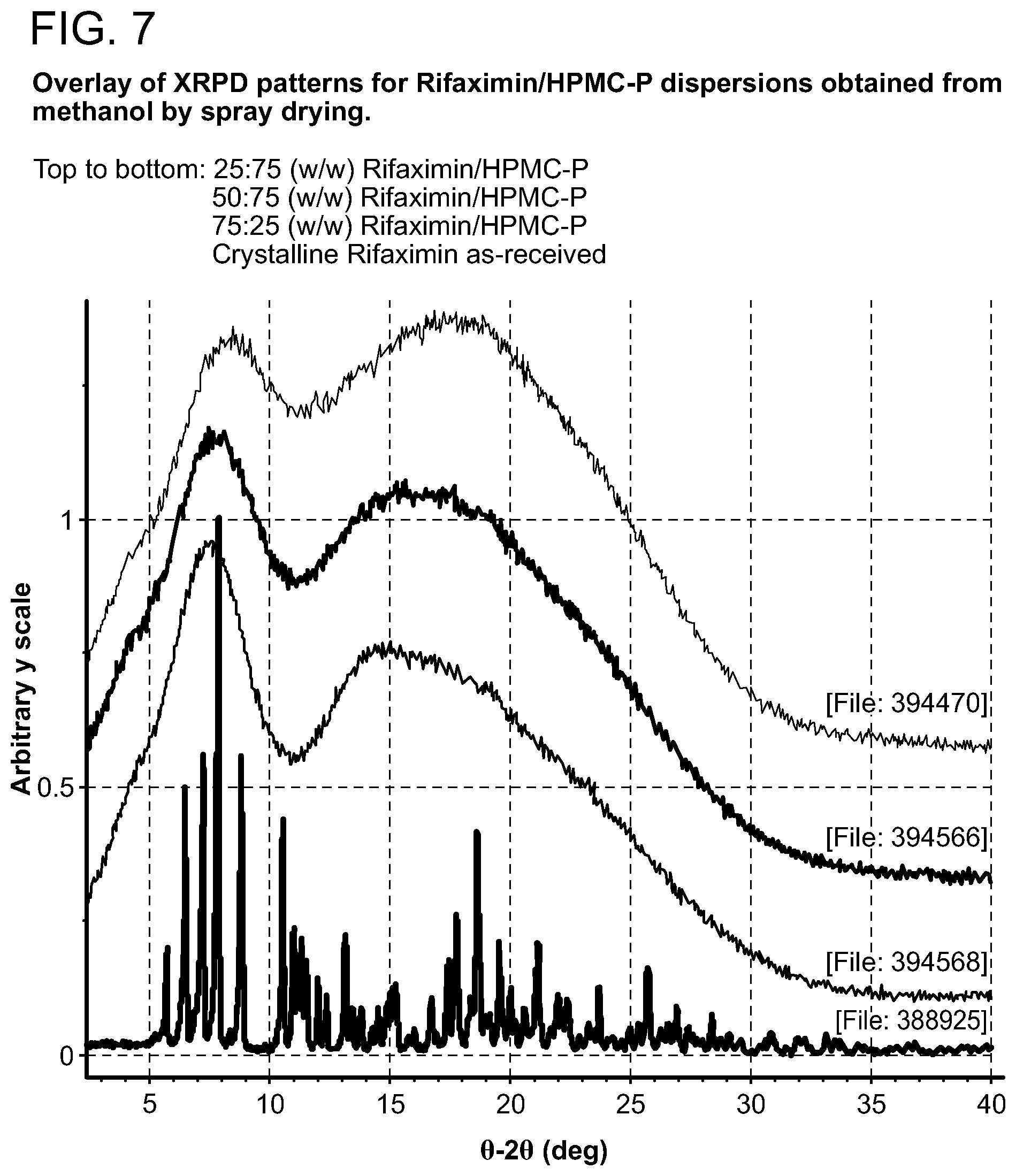
[0048] FIG. 7. Overlay of XRPD patterns for Rifaximin/HPMC-P dispersions obtained from methanol by spray drying.
[0049] FIG. 8. mDSC thermogram for 25:75 (w/w) Rifaximin/HPMC-P dispersion obtained from methanol by spray drying.
[0050] FIG. 9. mDSC thermogram for 50:50 (w/w) Rifaximin/HPMC-P dispersion obtained from methanol by spray drying.
[0051] FIG. 10. mDSC thermogram for 75:25 (w/w) Rifaximin/HPMC-P dispersion obtained from methanol by spray drying.
[0052] FIG. 11. Overlay of mDSC thermogram for Rifaximin/HPMC-P dispersions obtained from methanol by spray drying.
[0053] FIG. 12. Overlay of XRPD patterns for Rifaximin/HPMC-AS HG dispersions obtained from methanol by spray drying.
[0054] FIG. 13. mDSC thermogram for 25:75 (w/w) Rifaximin/HPMC-AS HG dispersion obtained from methanol by spray drying.
[0055] FIG. 14. mDSC thermogram for 50:50 (w/w) Rifaximin/HPMC-AS HG dispersion obtained from methanol by spray drying.
[0056] FIG. 15. mDSC thermogram for 75:25 (w/w) Rifaximin/HPMC-AS HG dispersion obtained from methanol by spray drying.
[0057] FIG. 16. Overlay of mDSC thermogram for Rifaximin/HPMC-AS HG dispersions obtained from methanol by spray drying.
[0058] FIG. 17. Overlay of XRPD patterns for Rifaximin/HPMC-AS MG dispersions obtained from methanol by spray drying.
[0059] FIG. 18. mDSC thermogram for 25:75 (w/w) Rifaximin/HPMC-AS MG dispersion obtained from methanol by spray drying.
[0060] FIG. 19. mDSC thermogram for 50:50 (w/w) Rifaximin/HPMC-AS MG dispersion obtained from methanol by spray drying.
[0061] FIG. 20. mDSC thermogram for 75:25 (w/w) Rifaximin/HPMC-AS MG dispersion obtained from methanol by spray drying.
[0062] FIG. 21. Overlay of mDSC thermogram for Rifaximin/HPMC-AS MG dispersions obtained from methanol by spray drying.
[0063] FIG. 22. Overlay of XRPD patterns for Rifaximin/Eudragit L100-55 dispersions obtained from methanol by spray drying.
[0064] FIG. 23. mDSC thermogram for 25:75 (w/w) Rifaximin/Eudragit L100-55 dispersion obtained from methanol by spray drying.
[0065] FIG. 24. mDSC thermogram for 50:50 (w/w) Rifaximin/Eudragit L100-55 dispersion obtained from methanol by spray drying.
[0066] FIG. 25. mDSC thermogram for 75:25 (w/w) Rifaximin/Eudragit L100-55 dispersion obtained from methanol by spray drying.
[0067] FIG. 26. Overlay of mDSC thermogram for Rifaximin/Eudragit L100-55 dispersions obtained from methanol by spray drying.
[0068] FIG. 27. mDSC thergram for 25:75 (w/w) Rifaximin/HPMC-P dispersion stressed at 40.degree. C./75% RH for 7 d.
[0069] FIG. 28. mDSC thergram for 75:25 (w/w) Rifaximin/HPMC-AS HG dispersion stressed at 40.degree. C./75% RH for 7 d.
[0070] FIG. 29. mDSC thergram for 75:25 (w/w) Rifaximin/HPMC-AS MG dispersion stressed at 40.degree. C./75% RH for 7 d.
[0071] FIG. 30. mDSC thergram for 25:75 (w/w) Rifaximin/Eudragit L100-55 dispersion stressed at 40.degree. C./75% RH for 7 d.
[0072] FIG. 31. XRPD pattern for 50:50 (w/w) Rifaximin/HPMC-AS MG dispersion.
[0073] FIG. 32. Modulate DSC thermograms for 50:50 (w/w) Rifaximin/HPMC-AS MG dispersion.
[0074] FIG. 33. TG-IR analysis for 50:50 (w/w) Rifaximin/HPMC-AS MG dispersion TGA data.
[0075] FIG. 34. TG-IR analysis for 50:50 (w/w) Rifaximin/HPMC-AS MG dispersion Gram-Schmidt plot and waterfall plot.
[0076] FIG. 35. TG-IR analysis for 50:50 (w/w) Rifaximin/HPMC-AS MG dispersion.
[0077] FIG. 36. XRPD pattern for 25:75 (w/w) Rifaximin/HPMC-P dispersion.
[0078] FIG. 37. Modulate DSC thermograms for 25:75 (w/w) Rifaximin/HPMC-P dispersion.
[0079] FIG. 38. TG-IR analysis for 25:75 (w/w) Rifaximin/HPMC-P dispersion--TGA data.
[0080] FIG. 39. TG-IR analysis for 25:75 (w/w) Rifaximin/HPMC-P dispersion--Gram-Schmidt plot and waterfall plot.
[0081] FIG. 40. TG-IR analysis for 25:75 (w/w) Rifaximin/HPMC-P dispersion.
[0082] FIG. 41. Overlay of pre-processed XRPD patterns in multivariate mixture analysis.
[0083] FIG. 42. Estimated Concentrations of Rifaximin (blue) and HPMC-AS MG (red) using Unscrambler MCR analysis.
[0084] FIG. 43. Estimated XRPD patterns of Rifaximin (blue) and HPMC-AS MG (red) using Unscrambler MCR analysis.
[0085] FIG. 44. Overlay of estimated XRPD pattern of pure rifaximin using MCR and measured XRPD pattern of 100% rifaximin.
[0086] FIG. 45. Overlay of estimated XRPD pattern of pure HPMC-AS MG using MCR and measured XRPD pattern of 100% HPMC-AS MG.
[0087] FIG. 46. An exemplary XRPD pattern for combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion.
[0088] FIG. 47. A modulate DSC thermogram for combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion.
[0089] FIG. 48. A TG-IR analysis for combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion--TGA thermogram.
[0090] FIG. 49. An exemplary TG-IR analysis for combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion.
[0091] FIG. 50. An exemplary overlay of IR spectra for X-ray amorphous Rifaximin and combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion.
[0092] FIG. 51. An exemplary overlay of Ramam spectra for X-ray amorphous Rifaximin and combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion.
[0093] FIG. 52. A particle size analysis report for combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion.
[0094] FIG. 53. An exemplary dynamic vapor sorption (DVS) analysis for combined solids of Rifaximin/HPMC-AS MG/Pluronic ternary dispersion.
[0095] FIG. 54. An exemplary overlay of XRPD patterns for Rifaximin/HPMC-AS MG/Pluronic ternary dispersion post-DVS solids and solids as-prepared.
[0096] FIG. 55. An exemplary overlay of XRPD patterns for Rifaximin ternary dispersion post-stressed samples and as-prepared sample.
[0097] FIG. 56. An exemplary mDSC thermgram for Rifaximin ternary dispersion after 70.degree. C./75% RH 1 week.
[0098] FIG. 57. An exemplary mDSC thermgram for Rifaximin ternary dispersion after 70.degree. C./75% RH 3 weeks.
[0099] FIG. 58. An exemplary mDSC thermgram for Rifaximin ternary dispersion after 40.degree. C./75% RH 6 weeks.
[0100] FIG. 59. An exemplary mDSC thermgram for Rifaximin ternary dispersion after 40.degree. C./75% RH 12 weeks.
[0101] FIG. 60. Pharmacokinetic data of solid dispersion in dogs.
[0102] FIG. 61. Rifaximin SD capsules dissolution; acid phase: 0.1 N HCl with variable exposure time. Buffer phase: pH 6.8 with 0.45% SDS.
[0103] FIG. 62. Rifaximin SD capsules dissolution; acid phase: 2 hours; buffer phase: pH 6.8.
[0104] FIG. 63. Rifaximin capsule dissolution; phosphate buffer pH 6.8 with 0.45% SDS.
[0105] FIG. 64A-D. Rifaximin spray dried dispersion (SDD) capsule dissolution. FIG. 64A acid phase 2 hours, buffer phase: P. Buffer, pH. 7.4. FIG. 64B acid phase: 0.1N HCl with various exposure times, buffer phase: P. buffer, pH 7.4 with 0.45% SDS. FIG. 64C shows the general structure of hydroxypropyl methylcellulose (HMPC). FIG. 64D represents the percent released at 30 min as a function of pH.
[0106] FIG. 65A-B. Rifamixin SDD with 10% CS formulation. FIG. 65A kinetic solubility Rifamixin SD granules. 10% wt % CS sodium FaSSIF, 10% wt % CS sodium FeSSIF. FIG. 65B dissolution profiles SDD tablet 10% CS. 0.2% SLS, pH4.5; 0.2% SLS, pH5.5; 0.2% SLS, pH 7.4; FaSSIF.
[0107] FIG. 66A-B. Rifaximin SDD with 10% CS formulation. Rifaxamin SDD capsules dissolution: FIG. 66A acid phase 2 hours, buffer phase: P. Buffer, pH. 7.4. With 0.45% SDS; without SDS. FIG. 66B acid phase: 0.1N HCl with variable exposure times, buffer phase: P. buffer, pH 7.4 with 0.45% SDS.
[0108] FIG. 67A-B. Effects of media pH on dissolution. FIG. 67A Rifaxamin SDD tablet dissolution. Acid phase: 2 hours, pH 2.0, FIG. 67B Dissolution profiles 0.2% SDS at pH 4.5, SDD tablet dissolution at various levels of CS: 0%, 2.5%, 5%, and 10% CS.
[0109] FIG. 68A-B. Effects of media pH on dissolution. FIG. 68A Rifaxamin SDD tablet dissolution at various levels of CS: 0%, 2.5%, 5%, and 10% CS, 0.2% SDS at pH 5.5. FIG. 68B Dissolution profiles SDD tablet dissolution at various levels of CS: 0%, 2.5%, 5%, and 10% CS, 0.2% SDS at pH 7.4.
[0110] FIG. 69A-B. Effects of media pH on dissolution. FIG. 69A Rifaxamin SDD tablet dissolution 2.5% CS, 0.2% SLS, pH4.5, 0.2% SLS, pH 5.5, 0.2% SLS, pH 7.4. FIG. 69B Rifaxamin SDD tablet dissolution 0% CS, 0.2% SLS, pH4.5, 0.2% SLS, pH 5.5, 0.2% SLS, pH 7.4.
[0111] FIG. 70A-B. Effects of media pH on dissolution. FIG. 70A Rifaxamin SDD tablet dissolution 10% CS, 0.2% SLS, pH4.5, 0.2% SLS, pH 5.5, 0.2% SLS, pH 7.4. FIG. 70B Rifaxamin SDD tablet dissolution 5% CS, 0.2% SLS, pH4.5, 0.2% SLS, pH 5.5, 0.2% SLS, pH 7.4.
[0112] FIG. 71A-B. CS release mechanism. FIG. 71A Kinetic solubility in FaSSIF media, pH 6.5, FIG. 71B slope vs. time point.
[0113] FIG. 72 depicts an overlay of XRPD patterns of rifaximin quaternary samples spray dried from methanol. The top is a rifaximin quaternary sample containing 0.063 wt % BHA. The second is rifaximin quaternary sample containing 0.063 wt % BHT. The third: is rifaximin quaternary sample containing 0.094 wt % PG, and the bottom is a spray dried rifaximin ternary dispersion.
[0114] FIG. 73 depicts an mDSC thermogram of rifaximin quaternary sample containing 0.063 wt % BHA.
[0115] FIG. 74 depicts an mDSC thermogram of rifaximin quaternary sample containing 0.063 wt % BHT.
[0116] FIG. 75 depicts a mDSC thermogram of rifaximin quaternary sample containing 0.094 wt % PG.
[0117] FIG. 76 depicts an XRPD pattern comparison of rifaximin solid dispersion powder 42.48% w/w with roller compacted material of rifaximin blend. Top: Rifaximin Solid Dispersion Powder 42.48% w/w; Bottom: roller compacted rifaximin blend.
[0118] FIG. 77 depicts the pharmacokinetics of rifaximin following administration of varying forms and formulations following a single oral dose of 2200 mg in dogs.
[0119] FIG. 78 depicts Rifaximin SDD in dogs.
[0120] FIG. 79 depicts the quotient study design.
[0121] FIG. 80 summarizes the dose escalation/regional absorption study, part A dose escalation/dose selection.
[0122] FIG. 81 depicts representative subject data from a dose escalation study.
[0123] FIG. 82 depicts representative subject data from a dose escalation study.
[0124] FIG. 83 depicts mean dose escalation data, on a linear scale.
[0125] FIG. 84 depicts mean dose escalation data, on a log scale.
[0126] FIG. 85 depicts a summary of Rifaximin SDD dose escalation studies.
[0127] FIG. 86 is a Table of dose/dosage form comparison.
[0128] FIG. 87 is a Table of dose/dosage form comparison. This table compares SDD at increasing doses to the current crystalline formulation in terms of systemic PK.
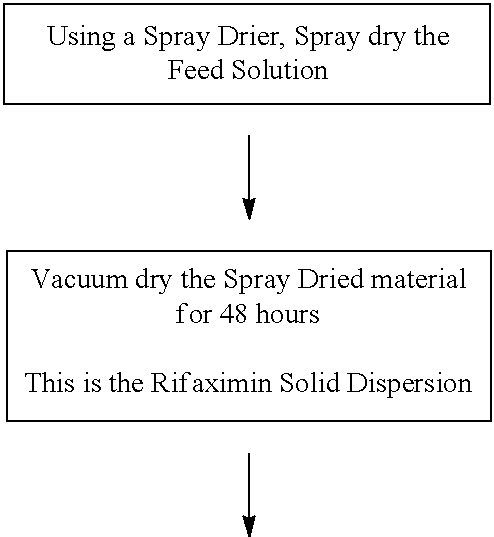
DETAILED DESCRIPTION
[0129] Embodiments described herein relate to the discovery of new solid dispersion forms of rifaximin with a variety of polymers and polymer concentrations. In one embodiment the use of one or more of new solid dispersion forms of the antibiotic known as Rifaximin (INN), in the manufacture of medicinal preparations for the oral or topical route is contemplated. For example, the solid dispersion forms of rifaximin are used to create pharmaceutical compositions, e.g., tablets or capsules, or microgranules comprising solid dispersion forms of rifaximin. Exemplary methods for producing rifaximin microgranules are set forth in the examples. Rifaximin microgranules can be formulated into pharmaceutical compositions as described herein.
[0130] Embodiments described herein also relate to administration of such medicinal preparations to a subject in need of treatment with antibiotics. Provided herein are solid dispersion forms of rifaximin with a variety of polymers and polymer concentrations.
[0131] As used herein, the term "intragranular release controlling agent" include agents that cause a pharmaceutical composition, e.g., a microgranule, to breakdown thereby releasing the active ingredient, e.g., rifaximin. Exemplary intragranular release controlling agent, include disintegrants such as crosprovidone, sodium starch glycolate, corn starch, microcrystalline cellulose, cellulosic derivatives, sodium bicarbonate, and sodium alginate.
[0132] In one embodiment, the intragranular release controlling agent comprises between about 2 wt % to about 40 wt % of the microgranule, about 5 wt % to about 20 wt % of the microgranule, about 8-15 wt % or about 10 wt % of the microgranule.
[0133] In another embodiment, the microgranule comprises a surfactant, e.g., a non-ionic surfactant. In one embodiment, the non-ionic surfactant comprises between about 2 wt % to about 10 wt % of the microgranule, between about 4 wt % to about 8 wt % of the microgranule, about 6 to about 7 wt % of the microgranule, or about 5.0 wt % of the microgranule.
[0134] In another embodiment, the microgranule comprises an antioxidant. In one embodiment, the antioxidant comprises between about 0.1 wt % to about 3 wt % of the microgranule, between 0.3 wt % to about 2 wt % or between about 0.5 wt % to about 1 wt % of the microgranule.
[0135] As used herein, the term "intragranular" refers to the components that reside within the microgranule. As used herein, the term "extragranular" refers to the components of the pharmaceutical composition that are not contained within the microgranule.
[0136] As used herein, the term polymorph is occasionally used as a general term in reference to the forms of rifaximin and includes within the context, salt, hydrate, polymorph co-crystal and amorphous forms of rifaximin. This use depends on context and will be clear to one of skill in the art.
[0137] As used herein, the term "about" when used in reference to x-ray powder diffraction pattern peak positions refers to the inherent variability of the peaks depending on, for example, the calibration of the equipment used, the process used to produce the polymorph, the age of the crystallized material and the like, depending on the instrumentation used. In this case the measure variability of the instrument was about .+-.0.2 degrees 2-.theta.. A person skilled in the art, having the benefit of this disclosure, would understand the use of "about" in this context. The term "about" in reference to other defined parameters, e.g., water content, C.sub.max, t.sub.max, AUC, intrinsic dissolution rates, temperature, and time, indicates the inherent variability in, for example, measuring the parameter or achieving the parameter. A person skilled in the art, having the benefit of this disclosure, would understand the variability of a parameter as connoted by the use of the word about.
[0138] As used herein, "similar" in reference to a form exhibiting characteristics similar to, for example, an XRPD, an IR, a Raman spectrum, a DSC, TGA, NMR, SSNMR, etc, indicates that the polymorph or cocrystal is identifiable by that method and could range from similar to substantially similar, so long as the material is identified by the method with variations expected by one of skill in the art according to the experimental variations, including, for example, instruments used, time of day, humidity, season, pressure, room temperature, etc.
[0139] As used herein, "rifaximin solid dispersion," "rifaximin ternary dispersion," "solid dispersion of rifaximin," "solid dispersion", "solid dispersion forms of rifaximin", "SD", "SDD", and "form solid dispersion of rifaximin" are intended to have equivalent meanings and include rifaximin polymer dispersion composition. These compositions are XRPD amorphous, but distinguishable from XRPD of amorphous rifaximin. As shown in the Examples and Figures, the rifaximin polymer dispersion compositions are physically chemically distinguishable from amorphous rifaximin, including different Tg, different XRPD profiles and different dissolution profiles.
[0140] Polymorphism, as used herein, refers to the occurrence of different crystalline forms of a single compound in distinct hydrate status, e.g., a property of some compounds and complexes. Thus, polymorphs are distinct solids sharing the same molecular formula, yet each polymorph may have distinct physical properties. Therefore, a single compound may give rise to a variety of polymorphic forms where each form has different and distinct physical properties, such as solubility profiles, melting point temperatures, hygroscopicity, particle shape, density, flowability, compactibility and/or x-ray diffraction peaks. The solubility of each polymorph may vary, thus, identifying the existence of pharmaceutical polymorphs is essential for providing pharmaceuticals with predictable solubility profiles. It is desirable to investigate all solid state forms of a drug, including all polymorphic forms, and to determine the stability, dissolution and flow properties of each polymorphic form. Polymorphic forms of a compound can be distinguished in a laboratory by X-ray diffraction spectroscopy and by other methods such as, infrared spectrometry. For a general review of polymorphs and the pharmaceutical applications of polymorphs see G. M. Wall, Pharm Manuf. 3, 33 (1986); J. K. Haleblian and W. McCrone, J Pharm. Sci., 58, 911 (1969); and J. K. Haleblian, J. Pharm. Sci., 64, 1269 (1975), all of which are incorporated herein by reference.
[0141] As used herein, "subject" includes organisms which are capable of suffering from a bowel disorder or other disorder treatable by rifaximin or who could otherwise benefit from the administration of rifaximin solid dispersion compositions as described herein, such as human and non-human animals. The term "non-human animals" includes all vertebrates, e.g., mammals, e.g., rodents, e.g., mice, and non-mammals, such as non-human primates, e.g., sheep, dog, cow, chickens, amphibians, reptiles, etc. Susceptible to a bowel disorder is meant to include subjects at risk of developing a bowel disorder infection, e.g., subjects suffering from one or more of an immune suppression, subjects that have been exposed to other subjects with a bacterial infection, physicians, nurses, subjects traveling to remote areas known to harbor bacteria that causes travelers' diarrhea, subjects who drink amounts of alcohol that damage the liver, subjects with a history of hepatic dysfunction, etc.
[0142] The language "a prophylactically effective amount" of a composition refers to an amount of a rifaximin solid dispersion formulation or otherwise described herein which is effective, upon single or multiple dose administration to the subject, in preventing or treating a bacterial infection.
[0143] The language "therapeutically effective amount" of a composition refers to an amount of a rifaximin solid dispersion effective, upon single or multiple dose administration to the subject to provide a therapeutic benefit to the subject. In one embodiment, the therapeutic benefit is wounding or killing a bacterium, or in prolonging the survivability of a subject with such a bowel or skin disorder. In another embodiment, the therapeutic benefit is inhibiting a bacterial infection or prolonging the survival of a subject with such a bacterial infection beyond that expected in the absence of such treatment.
[0144] Rifaximin exerts a broad antibacterial activity in the gastrointestinal tract against localized gastrointestinal bacteria that cause infectious diarrhea, including anaerobic strains. It has been reported that rifaximin is characterized by a negligible systemic absorption, due to its chemical and physical characteristics (Descombe J. J. et al. Pharmacokinetic study of rifaximin after oral administration in healthy volunteers. Int J Clin Pharmacol Res, 14 (2), 51-56, (1994)).
[0145] In respect to possible adverse events coupled to the therapeutic use of rifaximin, the induction of bacterial resistance to the antibiotics is of particular relevance.
[0146] From this point of view, any differences found in the systemic absorption of the forms of rifaximin disclosed herein may be significant, because at sub-inhibitory concentration of rifaximin, such as in the range from 0.1 to 1 .mu.g/ml, selection of resistant mutants has been demonstrated to be possible (Marchese A. et al. In vitro activity of rifaximin, metronidazole and vancomycin against Clostridium difficile and the rate of selection of spontaneously resistant mutants against representative anaerobic and aerobic bacteria, including ammonia-producing species. Chemotherapy, 46(4), 253-266, (2000)).
[0147] Forms, formulations and compositions of rifaximin have been found to have differing in vivo bioavailability properties. Thus, the polymorphs disclosed herein would be useful in the preparation of pharmaceuticals with different characteristics for the treatment of infections. This would allow generation of rifaximin preparations that have significantly different levels of adsorption with C.sub.max values from about 0.0 ng/ml to 5.0 .mu.g/ml. This leads to preparation of rifaximin compositions that are from negligibly to significantly adsorbed by subjects undergoing treatment. One embodiment described herein is modulating the therapeutic action of rifaximin by selecting the proper form, formulation and/or composition, or mixture thereof, for treatment of a subject. For example, in the case of invasive bacteria, the most bioavailable form, formulation and/or composition can be selected from those disclosed herein, whereas in case of non-invasive pathogens less adsorbed forms, formulations and/or compositions of rifaximin can be selected, since they may be safer for the subject undergoing treatment. A form, formulation and/or composition of rifaximin may determine solubility, which may also determine bioavailability.
[0148] For XRPD analysis, accuracy and precision associated with third party measurements on independently prepared samples on different instruments may lead to variability which is greater than .+-.0.1.degree. 2.theta.. For d-space listings, the wavelength used to calculate d-spacings was 1.541874 .ANG., a weighted average of the Cu-K.alpha.1 and Cu-K.alpha.2 wavelengths. Variability associated with d-spacing estimates was calculated from the USP recommendation, at each d-spacing, and provided in the respective data tables and peak lists.
Methods of Treatment
[0149] Provided herein are methods of treating, preventing, or alleviating bowel related disorders comprising administering to a subject in need thereof an effective amount of one or more of the solid dispersion compositions of rifaximin. Bowel related disorders include one or more of irritable bowel syndrome, diarrhea, microbe associated diarrhea, Clostridium difficile associated diarrhea, travelers' diarrhea, small intestinal bacterial overgrowth, Crohn's disease, diverticular disease, chronic pancreatitis, pancreatic insufficiency, enteritis, colitis, hepatic encephalopathy, minimal hepatic encephalopathy or pouchitis.
[0150] The length of treatment for a particular bowel disorder will depend in part on the disorder. For example, travelers' diarrhea may only require treatment duration of 12 to about 72 hours, while Crohn's disease may require treatment durations from about 2 days to 3 months. Dosages of rifaximin will also vary depending on the diseases state. Proper dosage ranges are provided herein infra. The polymorphs and cocrystals described herein may also be used to treat or prevent apathology in a subject suspected of being exposed to a biological warfare agent.
[0151] The identification of those subjects who are in need of prophylactic treatment for bowel disorder is well within the ability and knowledge of one skilled in the art. Certain of the methods for identification of subjects which are at risk of developing a bowel disorder which can be treated by the subject method are appreciated in the medical arts, such as family history, travel history and expected travel plans, the presence of risk factors associated with the development of that disease state in the subject. A clinician skilled in the art can readily identify such candidate subjects, by the use of, for example, clinical tests, physical examination and medical/family/travel history.
[0152] Topical skin infections and vaginal infections may also be treated with the rifaximin compositions described herein. Thus, described herein are methods of using a solid dispersion composition of rifaximin (SD rifaximin compositions) to treat vaginal infections, ear infections, lung infections, periodontal conditions, rosacea, and other infections of the skin and/or other related conditions. Provided herein are vaginal pharmaceutical compositions to treat vaginal infection, particularly bacterial vaginosis, to be administered topically, including vaginal foams and creams, containing a therapeutically effective amount of SD rifaximin compositions, preferably between about 50 mg and 2500 mg. Pharmaceutical compositions known to those of skill in the art for the treatment of vaginal pathological conditions by the topical route may be advantageously used with SD rifaximin compositions. For example, vaginal foams, ointments, creams, gels, ovules, capsules, tablets and effervescent tablets may be effectively used as pharmaceutical compositions containing SD rifaximin compositions, which may be administered topically for the treatment of vaginal infections, including bacterial vaginosis. Also provided herein are method of using SD rifaximin compositions to treat gastric dyspepsia, including gastritis, gastroduodenitis, antral gastritis, antral erosions, erosive duodenitis and peptic ulcers. These conditions may be caused by the Helicobacter pylori. Pharmaceutical formulations known by those of skill in the art with the benefit of this disclosure to be used for oral administration of a drug may be used. Provided herein are methods of treating ear infections with SD rifaximin compositions. Ear infections include external ear infection, or a middle and inner ear infection. Also provided herein are methods of using SD rifaximin compositions to treat or prevent aspiration pneumonia and/or sepsis, including the prevention of aspiration pneumonia and/or sepsis in patients undergoing acid suppression or undergoing artificial enteral feedings via a Gastrostomy/Jejunostomy or naso/oro gastric tubes; prevention of aspiration pneumonia in patients with impairment of mental status, for example, for any reason, for subjects undergoing anesthesia or mechanical ventilation that are at high risk for aspiration pneumonia. Provided herein are methods to treat or to prevent periodontal conditions, including plaque, tooth decay and gingivitis. Provided herein are methods of treating rosacea, which is a chronic skin condition involving inflammation of the cheeks, nose, chin, forehead, or eyelids.
Pharmaceutical Preparations
[0153] Embodiments also provide pharmaceutical compositions, comprising an effective amount of one or more SD rifaximin compositions, or microgranules comprising SD forms of rifaximin described herein (e.g., described herein and a pharmaceutically acceptable carrier). In a further embodiment, the effective amount is effective to treat a bacterial infection, e.g., small intestinal bacterial overgrowth, Crohn's disease, hepatic encephalopathy, antibiotic associated colitis, and/or diverticular disease. Embodiments also provide pharmaceutical compositions, comprising an effective amount of rifaximin SD compositions.
[0154] For examples of the use of rifaximin to treat Travelers' diarrhea, see Infante RM, Ericsson C D, Zhi-Dong J, Ke S, Steffen R, Riopel L, Sack D A, DuPont, HL., Enteroaggregative Escherichia coli Diarrhea in Travelers: Response to Rifaximin Therapy. Clinical Gastroenterology and Hepatology. 2004; 2:135-138; and Steffen R, M. D., Sack D A, M.D., Riopel L, PhD, Zhi-Dong J, Ph.D., Sturchler M, M.D., Ericsson C D, M.D., Lowe B, M. Phil., Waiyaki P, Ph.D., White M, Ph.D., DuPont H L, M. D. Therapy of Travelers' Diarrhea With Rifaximin on Various Continents. The American Journal of Gastroenterology. May 2003, Volume 98, Number 5, all of which are incorporated herein by reference in their entirety. Examples of treating hepatic encephalopathy with rifaximin see, for example, N. Engl J Med. 2010_362_1071-1081.
[0155] Embodiments also provide pharmaceutical compositions comprising rifaximin SD compositions and a pharmaceutically acceptable carrier. Embodiments of the pharmaceutical composition further comprise excipients, for example, one or more of a diluting agent, binding agent, lubricating agent, intragranular release controlling agent, e.g., a disintegrating agent, coloring agent, flavoring agent or sweetening agent. One composition may be formulated for selected coated and uncoated tablets, hard and soft gelatin capsules, sugar-coated pills, lozenges, wafer sheets, pellets and powders in sealed packet. For example, compositions may be formulated for topical use, for example, ointments, pomades, creams, gels and lotions.
[0156] In an embodiment, the rifaximin SD composition is administered to the subject using a pharmaceutically-acceptable formulation, e.g., a pharmaceutically-acceptable formulation that provides sustained or delayed delivery of the SD rifaximin composition to a subject for at least 2, 4, 6, 8, 10, 12 hours, 24 hours, 36 hours, 48 hours, one week, two weeks, three weeks, or four weeks after the pharmaceutically-acceptable formulation is administered to the subject. The pharmaceutically-acceptable formulations may contain microgranules comprising rifaximin as described herein.
[0157] In certain embodiments, these pharmaceutical compositions are suitable for topical or oral administration to a subject. In other embodiments, as described in detail below, the pharmaceutical compositions described herein may be specially formulated for administration in solid or liquid form, including those adapted for the following: (1) oral administration, for example, drenches (aqueous or non-aqueous solutions or suspensions), tablets, boluses, powders, granules, pastes; (2) parenteral administration, for example, by subcutaneous, intramuscular or intravenous injection as, for example, a sterile solution or suspension; (3) topical application, for example, as a cream, ointment or spray applied to the skin; (4) intravaginally or intrarectally, for example, as a pessary, cream or foam; or (5) aerosol, for example, as an aqueous aerosol, liposomal preparation or solid particles containing the compound.
[0158] The phrase "pharmaceutically acceptable" refers to those SD rifaximin compositions and cocrystals presented herein, compositions containing such compounds, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
[0159] The phrase "pharmaceutically-acceptable carrier" includes pharmaceutically-acceptable material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material, involved in carrying or transporting the subject chemical from one organ, or portion of the body, to another organ, or portion of the body. Each carrier is preferably "acceptable" in the sense of being compatible with the other ingredients of the formulation and not injurious to the subject. Some examples of materials which can serve as pharmaceutically-acceptable carriers include: (1) sugars, such as lactose, glucose and sucrose; (2) starches, such as corn starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter and suppository waxes; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, corn oil and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydroxide; (15) alginic acid; (16) pyrogen-free water; (17) isotonic saline; (18) Ringer's solution; (19) ethyl alcohol; (20) phosphate buffer solutions; and (21) other non-toxic compatible substances employed in pharmaceutical formulations.
[0160] Wetting agents, emulsifiers and lubricants, such as sodium lauryl sulfate and magnesium stearate, as well as coloring agents, release agents, coating agents, sweetening, flavoring and perfuming agents, preservatives and antioxidants can also be present in the compositions.
[0161] Examples of pharmaceutically-acceptable antioxidants include: (1) water soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like; (2) oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, alpha-tocopherol, and the like; and (3) metal chelating agents, such as citric acid, ethylenediamine tetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
[0162] Methods of preparing these compositions include the step of bringing into association a SD rifaximin composition(s) or microgranules containing the SD rifaximin compositions with the carrier and, optionally, one or more accessory ingredients. In general, the formulations are prepared by uniformly and intimately bringing into association a SD rifaximin composition with liquid carriers, or finely divided solid carriers, or both, and then, if necessary, shaping the product.
[0163] Compositions suitable for oral administration may be in the form of capsules, cachets, pills, tablets, lozenges (using a flavored basis, usually sucrose and acacia or tragacanth), powders, granules, or as a solution or a suspension in an aqueous or non-aqueous liquid, or as an oil-in-water or water-in-oil liquid emulsion, or as an elixir or syrup, or as pastilles (using an inert base, such as gelatin and glycerin, or sucrose and acacia) and/or as mouth washes and the like, each containing a predetermined amount of a SD rifaximin composition(s) as an active ingredient. A compound may also be administered as a bolus, electuary or paste.
[0164] The SD compositions of rifaximin disclosed herein can be advantageously used in the production of medicinal preparations having antibiotic activity, containing rifaximin, for both oral and topical use. The medicinal preparations for oral use will contain an SD composition of rifaximin together with the usual excipients, for example diluting agents such as mannitol, lactose and sorbitol; binding agents such as starches, gelatines, sugars, cellulose derivatives, natural gums and polyvinylpyrrolidone; lubricating agents such as talc, stearates, hydrogenated vegetable oils, polyethylenglycol and colloidal silicon dioxide; disintegrating agents such as starches, celluloses, alginates, gums and reticulated polymers; coloring, flavoring, disintegrants, and sweetening agents.
[0165] Embodiments described herein include SD rifaximin composition administrable by the oral route, for instance coated and uncoated tablets, of soft and hard gelatin capsules, sugar-coated pills, lozenges, wafer sheets, pellets and powders in sealed packets or other containers.
[0166] Pharmaceutical compositions for rectal or vaginal administration may be presented as a suppository, which may be prepared by mixing one or more SD rifaximin composition(s) with one or more suitable nonirritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the rectum or vaginal cavity and release the active agent. Compositions which are suitable for vaginal administration also include pessaries, tampons, creams, gels, pastes, foams or spray formulations containing such carriers as are known in the art to be appropriate.
[0167] Dosage forms for the topical or transdermal administration of a SD rifaximin composition(s) include powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants. The active SD rifaximin composition(s) may be mixed under sterile conditions with a pharmaceutically-acceptable carrier, and with any preservatives, buffers, or propellants which may be required.
[0168] Ointments, pastes, creams and gels may contain, in addition to SD rifaximin composition(s), excipients, such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
[0169] Powders and sprays can contain, in addition to a SD rifaximin composition(s), excipients such as lactose, talc, silicic acid, aluminium hydroxide, calcium silicates and polyamide powder, or mixtures of these substances. Sprays can additionally contain customary propellants, such as chlorofluorohydrocarbons and volatile unsubstituted hydrocarbons, such as butane and propane.
[0170] The SD rifaximin composition(s) can be alternatively administered by aerosol. This is accomplished by preparing an aqueous aerosol, liposomal preparation or solid particles containing the compound. A non-aqueous (e.g., fluorocarbon propellant) suspension could be used. Sonic nebulizers are preferred because they minimize exposing the agent to shear, which can result in degradation of the compound.
[0171] An aqueous aerosol is made, for example, by formulating an aqueous solution or suspension of the agent together with conventional pharmaceutically-acceptable carriers and stabilizers. The carriers and stabilizers vary with the requirements of the particular compound, but typically include non-ionic surfactants (Tweens, Pluronics, or polyethylene glycol), innocuous proteins like serum albumin, sorbitan esters, oleic acid, lecithin, amino acids such as glycine, buffers, salts, sugars or sugar alcohols. Aerosols generally are prepared from isotonic solutions.
[0172] Transdermal patches have the added advantage of providing controlled delivery of a SD rifaximin composition(s) to the body. Such dosage forms can be made by dissolving or dispersing the agent in the proper medium. Absorption enhancers can also be used to increase the flux of the active ingredient across the skin. The rate of such flux can be controlled by either providing a rate controlling membrane or dispersing the active ingredient in a polymer matrix or gel.
[0173] Ophthalmic formulations, eye ointments, powders, solutions and the like, are also contemplated as being within the scope of the invention.
[0174] Pharmaceutical compositions suitable for parenteral administration may comprise one or more SD rifaximin composition(s) in combination with one or more pharmaceutically-acceptable sterile isotonic aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents.
[0175] Examples of suitable aqueous and non-aqueous carriers which may be employed in the pharmaceutical compositions include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
[0176] When the SD rifaximin composition(s) are administered as pharmaceuticals, to humans and animals, they can be given per se or as a pharmaceutical composition containing, for example, 0.1 to 99.5% (more preferably, 0.5 to 90%) of active ingredient in combination with a pharmaceutically-acceptable carrier.
[0177] Regardless of the route of administration selected, the SD rifaximin composition(s) are formulated into pharmaceutically-acceptable dosage forms by methods known to those of skill in the art.
[0178] Actual dosage levels and time course of administration of the active ingredients in the pharmaceutical compositions may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular subject, composition, and mode of administration, without being toxic to the subject. An exemplary dose range is from 25 to 3000 mg per day. Other doses include, for example, 600 mg/day, 1100 mg/day and 1650 mg/day. Other exemplary doses include, for example, 1000 mg/day, 1500 mg/day, from between 500 mg to about 1800 mg/day or any value in-between.
[0179] A preferred dose of the SD rifaximin composition disclosed herein is the maximum that a subject can tolerate without developing serious side effects. Preferably, the SD rifaximin composition is administered at a concentration of about 1 mg to about 200 mg per kilogram of body weight, about 10 to about 100 mg/kg or about 40 mg to about 80 mg/kg of body weight. Ranges intermediate to the above-recited values are also intended to be part. For example, doses may range from 50 mg to about 2000 mg/day.
[0180] In combination therapy treatment, the other drug agent(s) are administered to mammals (e.g., humans, male or female) by conventional methods. The agents may be administered in a single dosage form or in separate dosage forms. Effective amounts of the other therapeutic agents are well known to those skilled in the art. However, it is well within the skilled artisan's purview to determine the other therapeutic agent's optimal effective-amount range. In one embodiment in which another therapeutic agent is administered to an animal, the effective amount of the rifaximin SD composition is less than its effective amount in case the other therapeutic agent is not administered. In another embodiment, the effective amount of the conventional agent is less than its effective amount in case the rifaximin SD composition is not administered. In this way, undesired side effects associated with high doses of either agent may be minimized. Other potential advantages (including without limitation improved dosing regimens and/or reduced drug cost) will be apparent to those skilled in the art.
[0181] In various embodiments, the therapies (e.g., prophylactic or therapeutic agents) are administered less than 5 minutes apart, less than 30 minutes apart, 1 hour apart, at about 1 hour apart, at about 1 to about 2 hours apart, at about 2 hours to about 3 hours apart, at about 3 hours to about 4 hours apart, at about 4 hours to about 5 hours apart, at about 5 hours to about 6 hours apart, at about 6 hours to about 7 hours apart, at about 7 hours to about 8 hours apart, at about 8 hours to about 9 hours apart, at about 9 hours to about 10 hours apart, at about 10 hours to about 11 hours apart, at about 11 hours to about 12 hours apart, at about 12 hours to 18 hours apart, 18 hours to 24 hours apart, 24 hours to 36 hours apart, 36 hours to 48 hours apart, 48 hours to 52 hours apart, 52 hours to 60 hours apart, 60 hours to 72 hours apart, 72 hours to 84 hours apart, 84 hours to 96 hours apart, or 96 hours to 120 hours part. In preferred embodiments, two or more therapies are administered within the same subject's visit.
[0182] In certain embodiments, one or more compounds and one or more other therapies (e.g., prophylactic or therapeutic agents) are cyclically administered. Cycling therapy involves the administration of a first therapy (e.g., a first prophylactic or therapeutic agent) for a period of time, followed by the administration of a second therapy (e.g., a second prophylactic or therapeutic agent) for a period of time, optionally, followed by the administration of a third therapy (e.g., prophylactic or therapeutic agent) for a period of time and so forth, and repeating this sequential administration, i.e., the cycle in order to reduce the development of resistance to one of the therapies, to avoid or reduce the side effects of one of the therapies, and/or to improve the efficacy of the therapies.
[0183] In certain embodiments, the administration of the same compounds may be repeated and the administrations may be separated by at least 1 day, 2 days, 3 days, 5 days, 10 days, 15 days, 30 days, 45 days, 2 months, 75 days, 3 months, or at least 6 months. In other embodiments, the administration of the same therapy (e.g., prophylactic or therapeutic agent) other than a SD rifaximin composition may be repeated and the administration may be separated by at least 1 day, 2 days, 3 days, 5 days, 10 days, 15 days, 30 days, 45 days, 2 months, 75 days, 3 months, or at least 6 months.
[0184] Certain indications may require longer treatment times. For example, travelers' diarrhea treatment may only last from between about 12 hours to about 72 hours, while a treatment for Crohn's disease may be from between about 1 day to about 3 months. A treatment for hepatic encephalopathy may be, for example, for the remainder of the subject's life span. A treatment for IBS may be intermittent for weeks or months at a time or for the remainder of the subject's life.
Compositions and Formulations
[0185] Rifaximin solid dispersions, pharmaceutical compositions comprising SD rifaximin or microgranules comprising rifaxmin solid dispersions, can be made from, for example, polymers including polyvinylpyrrolidone (PVP) grade K-90, hydroxypropyl methylcellulose phthalate (HPMC-P) grade 55, hydroxypropyl methylcellulose acetate succinate (HPMC-AS) grades HG and MG, and a polymethacrylate (Eudragit.RTM. L100-55). Rifaximin solid dispersion compositions are comprised of, for example, 10:90, 15:85, 20:80, 25:75, 30:70, 40:60, 50:50 60:40, 70:30, 75:25, 80:20, 85:15, and 90:10 (Rifaximin/polymer, by weight). Preferred solid dispersions are comprised of 25:75, 50:50 and 75:25 (Rifaximin/polymer, by weight). In addition to rifaximin and polymer, solid dispersions may also comprise surfactants, for example, non-ionic, surfactant polyols.
[0186] An example of a formulation comprises about 50:50 (w/w) Rifaximin:HPMC-AS MG with from between about 2 wt % to about 10 wt % of a non-ionic, surfactant polyol, for example, Pluronic F-127.
[0187] One example of a formulation comprises 50:50 (w/w) Rifaximin:HPMC-AS MG with about 5.9 wt %) of a non-ionic, surfactant polyol, for example, Pluronic F-127. Spray dried rifaximin ternary dispersion (50:50 (w/w) rifaximin:HPMC-AS MG with 5.9 wt % Pluronic F-127) was blended with 10 wt % croscarmellose sodium and then filled into gelatin capsules. Each capsule contains 275 mg of rifaximin and the blend formulation is 85:5:10 of 50:50 (w/w) Rifaximin:HPMC-AS MG: Pluronic: croscarmellose sodium (calculated in total solids). Other examples of microgranules and pharmaceutical compositions comprising SD rifaximin are described in the examples.
[0188] To form the rifaximin solid dispersion, the components, e.g., rifaximin, polymer and methanol are mixed and then spray dried. Exemplary conditions are summarized in Table 9 and the procedure outlined below and in Examples 3 and 4.
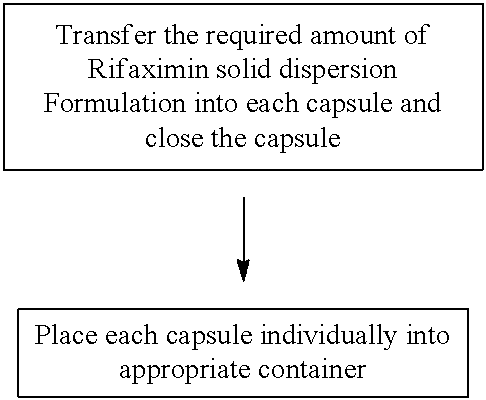
[0189] Exemplary Spray Drying Process Parameters, include for example: [0190] Spray Dryer--e.g., PSD 1; [0191] Single or multi-fluid nozzle: e.g., a two Fluid Niro Nozzle; [0192] Nozzle orifice--0.1-10 mm; [0193] Inlet gas temperature--75-150.+-.5 deg C.; [0194] Process gas flow (mmH2O)--20-70, preferred 44; [0195] Atomizing gas pressure--0.7-1 bar; [0196] Feed rate--2-7 kg/Hr; [0197] Outlet temperature--30-70.+-.3 deg C.; [0198] Solution temperature--20-50 deg C.; and [0199] Post spray drying vacuum dry at 20-60 deg C., for between about 2 and 72 hrs.
Article of Manufacture
[0200] Another embodiment includes articles of manufacture that comprise, for example, a container holding a rifaximin SD pharmaceutical composition suitable for oral or topical administration of rifaximin in combination with printed labeling instructions providing a discussion of when a particular dosage form should be administered with food and when it should be taken on an empty stomach. Exemplary dosage forms and administration protocols are described infra. The composition will be contained in any suitable container capable of holding and dispensing the dosage form and which will not significantly interact with the composition and will further be in physical relation with the appropriate labeling. The labeling instructions will be consistent with the methods of treatment as described hereinbefore. The labeling may be associated with the container by any means that maintain a physical proximity of the two, by way of non-limiting example, they may both be contained in a packaging material such as a box or plastic shrink wrap or may be associated with the instructions being bonded to the container such as with glue that does not obscure the labeling instructions or other bonding or holding means.
[0201] Another aspect is an article of manufacture that comprises a container containing a pharmaceutical composition comprising SD rifaximin composition or formulation wherein the container holds preferably rifaximin composition in unit dosage form and is associated with printed labeling instructions advising of the differing absorption when the pharmaceutical composition is taken with and without food.
[0202] Packaged compositions are also provided, and may comprise a therapeutically effective amount of rifaximin. Rifaximin SD composition and a pharmaceutically acceptable carrier or diluent, wherein the composition is formulated for treating a subject suffering from or susceptible to a bowel disorder, and packaged with instructions to treat a subject suffering from or susceptible to a bowel disorder.
[0203] Kits are also provided herein, for example, kits for treating a bowel disorder in a subject. The kits may contain, for example, one or more of the solid dispersion forms of rifaximin and instructions for use. The instructions for use may contain proscribing information, dosage information, storage information, and the like.
[0204] Packaged compositions are also provided, and may comprise a therapeutically effective amount of an SD rifaximin composition and a pharmaceutically acceptable carrier or diluent, wherein the composition is formulated for treating a subject suffering from or susceptible to a bowel disorder, and packaged with instructions to treat a subject suffering from or susceptible to a bowel disorder.
[0205] The present invention is further illustrated by the following examples, which should not be construed as further limiting. The contents of all figures and all references, patents and published patent applications cited throughout this application, as well as the Figures, are expressly incorporated herein by reference in their entirety.
EXAMPLES
[0206] The chemical structure of Rifaximin is shown below in FIG. 1.
Example 1. Solid Dispersions of Rifaximin
[0207] Various polymers were formulated with rifaximin into solids prepared by methanol and spray drying at small scale (.about.1 g). Polymers, including polyvinylpyrrolidone (PVP) grade K-90, hydroxypropyl methylcellulose phthalate (HPMC-P) grade 55, hydroxypropyl methylcellulose acetate succinate (HPMC-AS) grades HG and MG, and a polymethacrylate (Eudragit.RTM. L100-55) were used. Solids have compositions of 25:75, 50:50 and 75:25 (Rifaximin/polymer, by weight).
[0208] Samples generated were observed under polarized light microscope after preparation and were characterized by XRPD. The results are included in Table 1 through Table 5. Birefringence with extinction (B/E) was not observed for any of the samples, indicating solids without crystalline order were obtained. No sharp peaks were evident by visual inspection of XRPD patterns of these samples, consistent with non-crystalline materials, as shown in FIG. 2 (with PVP K-90), FIG. 7 (with HPMC-P), FIG. 12 (with HMPC-AS HG), FIG. 12 (with HMPC-AS MG), and FIG. 17 (with Eudragit L100-55).
[0209] Materials were characterized by mDSC where the appearance of a single glass transition temperature (Tg), provides support for a non-crystalline fully miscible dispersion. All the dispersions prepared with PVP K-90 display a single apparent Tg at approximately 185.degree. C. (FIG. 3, 25:75 w/w), 193.degree. C. (FIG. 4, 50:50 w/w), and 197.degree. C. (FIG. 5, 75:25) respectively. The change in heat capacity (.DELTA.Cp) at Tg is approximately 0.3 J/g.degree. C. for each dispersion. A non-reversible endotherm, which is likely due to the residual solvent in the materials, was observed in each of Rifaximin/PVP K-90 dispersions centered at approximately 78.degree. C., 59.degree. C. and 61.degree. C.
[0210] From FIG. 6, Tg of Rifaximin/PVP K-90 dispersions increases with the increased Rifaximin concentration, which is due to the higher Tg of Rifaximin (199.degree. C.) than PVP K-90 (174.degree. C.). Evidence of a single Tg may suggest that the components of the dispersion are intimately mixed, or miscible.
[0211] Dispersions prepared with other polymers also display a single apparent Tg, as a step change in the reversing heat flow signal by mDSC. Dispersions prepared with HPMC-P exhibit Tg at 153.degree. C. (FIG. 8, 25:75 w/w), 161.degree. C. (FIG. 9, 50:50 w/w) and 174.degree. C. (FIG. 10, 75:25 w/w) respectively, with .DELTA.Cp at Tg approximately 0.4 J/g.degree. C.
[0212] With HPMC-AS HG, dispersions display Tg at 137.degree. C. (FIG. 13, 25:75 w/w), 154.degree. C. (FIG. 14, 50:50 w/w) and 177.degree. C. (FIG. 15, 75:25 w/w) respectively; .DELTA.Cp at Tg is approximately 0.4 or 0.3 J/g.degree. C.
[0213] With HPMC-AS MG, dispersions display Tg at 140.degree. C. (FIG. 18, 25:75 w/w), 159.degree. C. (FIG. 19, 50:50 w/w) and 177.degree. C. (FIG. 10, 75:25 w/w) respectively; .DELTA.Cp at Tg is approximately 0.4 or 0.3 J/g.degree. C.
[0214] Dispersions prepared with Eudragit L100-55 exhibit Tg at 141.degree. C. with .DELTA.Cp approximately 0.5 J/g.degree. C. (FIG. 23, 25:75 w/w), 159.degree. C. with .DELTA.Cp approximately 0.3 J/g.degree. C. (FIG. 24, 50:50 w/w), and 176.degree. C. with .DELTA.Cp at Tg approximately 0.2 J/g.degree. C. (FIG. 25, 75:25 w/w) respectively.
[0215] Similarly, as shown in FIG. 11 (with HPMC-P), FIG. 16 (with HPMC-AS HG), FIG. 21 (with HPMC-AS MG, and FIG. 26 (with Eudragit L100-55), Tg of material in each set of Rifaximin/polymer dispersions increases with the increased Rifaximin concentration due to the higher Tg of Rifaximin.
Physical Stability Assessment
[0216] An assessment of physical stability for rifaximin/polymer dispersions was conducted under stress conditions of aqueous solutions at different biologically relevant conditions, including 0.1N HCl solution at 37.degree. C. and pH 6.5 FASSIF buffer at 37.degree. C., elevated temperature/relative humidity (40.degree. C./75% RH), and elevated temperature/dry (60.degree. C.). The x-ray amorphous rifaximin--only sample prepared from methanol by spray drying was also stressed under the same conditions for comparison.
Stress in 0.1N HCl Solution at 37.degree. C.
[0217] For the assessment of physical stability for samples in a 0.1N HCl solution maintained at 37.degree. C., observations were made and microscopy images were acquired using polarized light at different time points including 0, 6 and 24 hrs, as summarized in Table 6. Based on the absence of birefringent particles when samples were observed by PLM, dispersions prepared with HPMC-AS HG and HPMC-AS MG display the highest physical stability under this particular stress condition. The results of this study for each of samples are discussed below.
[0218] X-ray amorphous Rifaximin stressed in 0.1N HCl solution at 37.degree. C. at 0, 6, and 24 hrs showed evidence of birefringence/extinctions was observed at 6 hrs, indicating the occurrence of devitrification of the material.
[0219] Samples at compositions of 25:75 and 50:50 (w/w) crystallized at 6 hrs; sample at 75:25 (w/w) composition crystallized within 24 hrs while no evidence of crystallization was observed at 6 hrs or earlier. The decreased stability of Rifaximin/PVP K-90 dispersions in 0.1N HCl solution with increased PVP K-90 concentration may due to the high solubility of PVP K-90 in the solution.
[0220] Irregular aggregates without birefringence/extinctions were observed for dispersion prepared with HPMC-P at t=0 hr, the initial time point when 0.1N HCl solution was just added into solids. After 24 hrs, samples at compositions of 25:75 and 50:50 (w/w) remained as non-birefringent aggregates, indicating no occurrence of devitrification under the conditions examined. Evidence of crystallization was observed for sample of 75:25 (w/w) composition at 6 hrs. No birefringence/extinctions were observed for all of dispersions prepared with HPMC-AS HG and HPMC-AS MG after 24 hrs, suggesting these samples are resistant to devitrification upon exposure to 0.1N HCl solution for 24 hrs.
[0221] For dispersions prepared with Eudragit L100-55, upon exposure to 0.1N HCl solution for 24 hrs, birefringent particles with extinctions were observed only in the sample at 50:50 (w/w) composition. Considered that no evidence of crystallization was observed for dispersions of compositions at 25:75 and 75:25 (w/w), it is unknown whether such birefringence was caused by some foreign materials or by crystalline solids indicating the occurrence of devitrification.
Stress in pH 6.5 FASSIF Buffer at 37.degree. C.
[0222] An assessment of physical stability of dispersions prepared was also performed in pH 6.5 FASSIF buffer maintained at 37.degree. C. X-ray amorphous Rifaximin material was also stressed under same condition for comparison. PLM observations indicated that dispersions prepared from HPMC-AS HG and HPMC-AS MG display the highest physical stability under this stress condition. X-ray amorphous rifaximin-only material crystallized within 6 hrs, so did all rifaximin/PVP K-90 dispersions. For dispersions prepared with HPMC-P, birefringent particles with extinctions were observed in samples at 50:50 and 75:25 (w/w) compositions within 6 hrs, indicating the occurrence of devitrification in materials. No evidence of any birefringence/extinctions was observed in 25:75 (w/w) rifaximin/HPMC-P dispersion material after 24 hrs. No birefringence/extinctions were observed for all of dispersions prepared with HPMC-AS HG and HPMC-AS MG after 24 hrs, suggesting these samples are resistant to devitrification upon exposure to pH 6.5 FASSIF buffer for 24 hrs. Rifaximin/Eudragit L100-55 dispersions at 50:50 and 75:25 (w/w) compositions crystallized with 6 hrs while no evidence of crystallization was observed in the sample at 25:75 (w/w) composition after 24 hrs.
Stress at 40.degree. C./75% RH Condition
[0223] The samples including all the dispersions and x-ray amorphous rifaximin-only material were assessed for evidence of crystallization based on observations by microscopy using polarized light. Each of the samples remained as irregular aggregates without birefringence/extinctions after stressed at 40.degree. C./75% RH condition for 7 days.
[0224] Modulated DSC analyses were carried out on selected samples including 25:75 (w/w) rifaximin/HPMC-P, 75:25 (w/w) rifaximin/HPMC-AS HG, 75:25 (w/w) rifaximin/HPMC-AS MG, and 25:75 (w/w) Rifaximin/Eudragit L100-55 to inspect for evidence of phase separation after exposure to 40.degree. C./75% RH for 7 days. All of samples display a single apparent Tg at approximately 148.degree. C. (FIG. 27, 25:75 (w/w) HPMC-P), 177.degree. C. (FIG. 28, 75:25 (w/w) HPMC-AS HG) 152.degree. C. (FIG. 29, 75:25 (w/w) HPMC-AS MG) and 140.degree. C. (FIG. 30, 25:75 (w/w) Eudragit L100-55) respectively, indicating the components of each dispersion remained intimately miscible after stress. Although crimped with manual pin-hole DSC pan was used, the release of moisture from sample upon heating can still be observed from non-reversible heat flow signals.
Stress at 60.degree. C./Dry Condition
[0225] All the dispersions and x-ray amorphous rifaximin-only material were also stressed at 60.degree. C./dry condition for 7 days and were assessed for evidence of crystallization based on observations by microscopy using polarized light. Each of the samples remained as irregular aggregates without birefringence/extinctions after stressed at this condition for 7 days.
Rifaximin Solid Dispersions by Spray Drying
[0226] Based on the experimental results from screen, HPMC-AS MG and HPMC-P were used to prepare additional quantities of solid dispersions at gram-scale by spray drying. The operating parameters used for processing are presented in Table 9. Based on visual inspection, both dispersions were x-ray amorphous by XRPD (FIG. 31 and FIG. 36).
Characterization of 50:50 (w/w) Rifaximin/HPMC-AS MG Dispersion
[0227] Characterization and results for the 50% API loading HPMC-AS MG are summarized in Table 10. The sample was x-ray amorphous based on high resolution XRPD. A single Tg at approximately 154.degree. C. was observed from the apparent step change in the reversing heat flow signal in mDSC with the change of heat capacity 0.4 J/g .degree. C. A non-reversible endotherm was observed at approximately 39.degree. C. which is likely due to the residual solvent in the materials (FIG. 32). TG-IR analysis was carried out in order to determine volatile content on heating. TGA data for this material is shown in FIG. 34. There was a 0.5% weight loss up to .about.100.degree. C. A Gram-Schmidt plot corresponding to the overall IR intensity associated with volatiles released by solids upon heating at 20.degree. C./min is shown in FIG. 33. There was a dramatic increase of intensity of released volatiles after .about.8 minutes, with a maximum at .about.11.5 minutes. The waterfall plot (FIG. 34) and the linked IR spectrum (FIG. 35) are indicative of the loss of water loss up to -8 minutes then methanol and some unknown volatiles thereafter. This is consistent with the dramatic change in the slope in the TGA and may indicate decomposition of material.
Characterization of 25:75 (w/w) Rifaximin/HPMC-P Dispersion
[0228] Characterization and results for the 25% API loading dispersion of HPMC-P are summarized in Table 11. Solids were x-ray amorphous based on high resolution XRPD (FIG. 36). By mDSC, there is a single Tg at approximately 152.degree. C. from the apparent step change in the reversing heat flow signal. The change of heat capacity is 0.4 J/g .degree. C. (FIG. 37). A non-reversible endotherm, which is likely due to the residual solvent in the materials, was observed at approximately 46.degree. C. Volatiles generated on heating were analyzed by TG-IR. The total weight loss of sample was approximately 1.5 wt % to 100.degree. C. and the dramatic change in the slope occurs at approximately 178.degree. C. (FIG. 38). The Gram-Schmidt plot (FIG. 39) shows a small increase of intensity upon heating after .about.2 minutes, followed by negligible change of intensity until .about.9 minutes. Then dramatic change of intensity can be observed with a maximum at .about.11 minutes, followed by a final increase of intensity above .about.12 minutes. As seen in the waterfall plot (FIG. 39), some volatiles were released during entire heating period (data is shown in FIG. 40 using the linked IR spectrum at different time points as an example). The sample released water during entire heating period and methanol after .about.9 minutes.
Dispersions Miscibility Study by Multivariate Mixture Analysis
[0229] For Rifaximin/HPMC-AS MG dispersions prepared by spray drying, a multivariate mixture analysis was performed using the XRPD data to examine the physical state of the components and inspect for evidence of miscibility. The analysis was done with MATLAB (v7.6.0) and Unscrambler (v 9.8) and it was not performed under cGMP guidelines. XRPD patterns of all the samples were truncated with their baseline corrected, and unit area normalized before analysis. The pre-possessed XRPD patterns are shown in FIG. 41.
[0230] In the analysis, Rifaximin and HPMC-AS MG were assumed to be separated phases (no miscibility) and the compositions of Rifaximin and HPMC-AS MG in each sample were estimated based on this assumption. As shown in FIG. 42, the estimated ratios of Rifaximin to HPMC-AS MG based on pure separated phases did not agree with samples actual compositions, especially for the samples with high compositions of HPMC-AS MG (low Rifaximin loading). Also, the calculated XRPD patterns for Rifaximin and HMPC-AS MG based on the assumption of separated phases (FIG. 43) compared to actual experimental XRPD patterns for Rifaximin (FIG. 44) and HPMC-AS MG (FIG. 45) were generated. Although the calculated Rifaximin pattern is similar to its experimental pattern, the calculated HMPC-AS MG pattern is quite different from its experimental pattern. Both results suggest that Rifaximin and HPMC-AS MG are not separated phases but miscible in the dispersions. The differences in the estimated and actual compositions are likely due to the interaction between Rifaximin and HPMC-AS MG.
TABLE-US-00001 TABLE 1 Solid Dispersion Attempts for Rifaximin/PVP K-90 by Spray Drying Description (a, b) Habit/Description Analysis Result (c) (25:75) solids orange; XRPD x-ray amorph. PVP K-90 aggregates, irregular, mDSC 185.degree. C. (T.sub.g, no B/E midpoint); 0.3 J/g .degree. C. (.DELTA.C.sub.p) (50:50) solids orange; XRPD x-ray amorph. PVP K-90 aggregates, irregular, mDSC 193.degree. C. (T.sub.g, no B/E midpoint); 0.3 J/g .degree. C. (.DELTA.C.sub.p) (75:25) solids orange; XRPD x-ray amorph. PVP K-90 aggregates, irregular, mDSC 197.degree. C. (T.sub.g, no B/E midpoint); 0.3 J/g .degree. C. (.DELTA.C.sub.p) (a): approximate ratio of Rifaximin to polymer, by weight; (b): samples stored in freezer over desiccant after prepared.
TABLE-US-00002 TABLE 2 Solid Dispersion Attempts for Rifaximin/HPMC-P by Spray Drying Description (a, b) Habit/Description Analysis Result (c) (25:75) solids light orange; XRPD x-ray amorph. HPMC-P aggregates, irregular, mDSC 153.degree. C. (T.sub.g, midpoint); no B/E 0.4 J/g .degree. C. (.DELTA.C.sub.p) (50:50) solids orange; XRPD x-ray amorph. HPMC-P aggregates, irregular, mDSC 161.degree. C. (T.sub.g, midpoint); no B/E 0.4 J/g .degree. C. (.DELTA.C.sub.p) (75:25) solids orange; XRPD x-ray amorph. HPMC-P aggregates, irregular, mDSC 174.degree. C. (T.sub.g, midpoint); no B/E 0.4 J/g .degree. C. (.DELTA.C.sub.p) (a): approximate ratio of Rifaximin to polymer, by weight; (b): samples stored in freezer over desiccant after prepared.
TABLE-US-00003 TABLE 3 Solid Dispersion Attempts for Rifaximin/HPMC-AS HG by Spray Drying Description (a, b) Habit/Description Analysis Result (c) (25:75) solids light orange; XRPD x-ray amorph. HPMC-AS HG aggregates, irregular, mDSC 137.degree. C. (T.sub.g, no B/E midpoint); 0.4 J/g .degree. C. (.DELTA.C.sub.p) (50:50) solids orange; XRPD x-ray amorph. HPMC-AS HG aggregates, irregular, mDSC 154.degree. C. (T.sub.g, no B/E midpoint); 0.4 J/g .degree. C. (.DELTA.C.sub.p) (75:25) solids orange; XRPD x-ray amorph. HPMC-AS HG aggregates, irregular, mDSC 177.degree. C. (T.sub.g, no B/E midpoint); 0.3 J/g .degree. C. (.DELTA.C.sub.p) (a): approximate ratio of Rifaximin to polymer, by weight; (b): samples stored in freezer over desiccant after prepared.
TABLE-US-00004 TABLE 4 Solid Dispersion Attempts for Rifaximin/HPMC-AS MG by Spray Drying Description (a, b) Habit/Description Analysis Result (c) (25:75) solids light orange; XRPD x-ray amorph. HPMC-AS MG aggregates, mDSC 140.degree. C. (T.sub.g, midpoint); irregular, no B/E 0.4 J/g .degree. C. (.DELTA.C.sub.p) (50:50) solids orange; XRPD x-ray amorph. HPMC-AS MG aggregates, mDSC 159.degree. C. (T.sub.g, midpoint); irregular, no B/E 0.4 J/g .degree. C. (.DELTA.C.sub.p) (75:25) solids orange; XRPD x-ray amorph. HPMC-AS MG aggregates, mDSC 177.degree. C. (T.sub.g, midpoint); irregular, no B/E 0.3 J/g .degree. C. (.DELTA.C.sub.p) (a): approximate ratio of Rifaximin to polymer, by weight; (b): samples stored in freezer over desiccant after prepared.
TABLE-US-00005 TABLE 5 Solid Dispersion Attempts for Rifaximin/Eudragit L100-55 by Spray Drying Description (a, b) Habit/Description Analysis Result (c) (25:75) solids light orange, XRPD x-ray amorph. Eudragit L100-55 aggregates, mDSC 141.degree. C. (T.sub.g, irregular, no B/E midpoint); 0.5 J/g .degree. C. (.DELTA.C.sub.p) (50:50) solids orange; XRPD x-ray amorph. Eudragit L100-55 aggregates, mDSC 159.degree. C. (T.sub.g, irregular, no B/E midpoint); 0.3 J/g .degree. C. (.DELTA.C.sub.p) (75:25) solids orange; XRPD x-ray amorph. Eudragit L100-55 aggregates, mDSC 176.degree. C. (T.sub.g, irregular, no B/E midpoint); 0.2 J/g .degree. C. (.DELTA.C.sub.p) (a): approximate ratio of Rifaximin to polymer, by weight; (b): samples stored in freezer over desiccant after prepared.
TABLE-US-00006 TABLE 6 Physical Stability Assessment in 0.1N HCl at 37.degree. C. for Rifaximin and Rifaximin Dispersions Prepared in Methanol by Spray Drying Description Time (a) (b) Habit/Description Analysis Results (100:0) 0 -- PLM agg., irr., no B/E Rifaximin- agg., irr., no B/E only 6 hrs orange solids, no PLM agg., no B/E + a liquid left few B/E particles clear view of B/E particles 24 hrs orange solids, PLM agg., no B/E + a solution cloudy few B/E particles (25:75) 0 -- PLM agg., irr., no B/E PVP K-90 agg., irr., no B/E 6 hrs orange solids, PLM agg., no B/E + solution slightly B/E particles yellow clear view of B/E particles 24 hrs orange solids, PLM agg., no B/E + solution slightly B/E particles yellow (50:50) 0 -- PLM agg., irr., no B/E PVP K-90 agg., irr., no B/E 6 hrs orange solids, PLM agg., no B/E + a solution slightly few B/E particles yellow clear view of B/E particles 24 hrs orange solids, PLM majority agg., small amount of no B/E + liquid left a few B/E particles clear view of B/E particles (75:25) 0 -- PLM agg., irr., no B/E PVP K-90 agg., irr., no B/E 6 hrs orange solids, PLM agg., no B/E solution slightly agg., no B/E yellow 24 hrs orange solids, PLM agg., no B/E small amount of a few B/E particles liquid left in view field (25:75) 0 -- PLM agg., irr., no B/E HPMC-P agg., irr., no B/E 6 hrs light orange solids, PLM agg., no B/E liquid turbid agg., no B/E 24 hrs orange solids, PLM agg., no B/E liquid turbid agg., no B/E (50:50) 0 -- PLM agg., irr., no B/E HPMC-P agg., irr., no B/E 6 hrs orange solids, PLM agg., no B/E liquid turbid agg., no B/E 24 hrs orange solids, PLM agg., no B/E solution cloudy agg., no B/E (75:25) 0 -- PLM agg., irr., no B/E HPMC-P agg., irr., no B/E 6 hrs orange solids, PLM agg., no liquid turbid B/E + some B/E particles clear view of B/E particles 24 hrs orange solids, PLM B/E particles small amount of observed liquid left clear view of B/E particles (25:75) 0 -- PLM agg., irr., no B/E HPMC-AS agg., irr., no B/E HG 6 hrs light orange PLM no B/E observed solids in no B/E observed cloudy liquid 24 hrs orange solids in PLM no B/E observed cloudy solution no B/E observed (50:50) 0 -- PLM agg., irr., no B/E HPMC-AS agg., irr., no B/E HG 6 hrs orange solids, PLM no B/E observed liquid cloudy no B/E observed 24 hrs orange solids in PLM no B/E observed cloudy solution (75:25) 0 -- PLM agg., irr., no B/E HPMC-AS agg., irr., no B/E HG 6 hrs orange solids, PLM no B/E observed liquid turbid no B/E observed 24 hrs orange solids + PLM agg., no B/E cloudy solution agg., no B/E (25:75) 0 -- PLM agg., irr., no B/E HPMC-AS agg., irr., no B/E MG 6 hrs light orange PLM no B/E observed solids in cloudy liquid 24 hrs orange solids in PLM majority no B/E, a cloudy liquid few B/E particles B/E particles seems fiber-like, may due to foreign materials (50:50) 0 -- PLM agg., irr., no B/E HPMC-AS agg., irr., no B/E MG 6 hrs orange solids, PLM agg., no B/E + liquid turbid a few B/E particles seems due to foreign material clear view of B/E particles 24 hrs orange solids in PLM no B/E observed cloudy solution no B/E observed (75:25) 0 -- PLM agg., irr., no B/E HPMC-AS agg., irr., no B/E MG 6 hrs orange solids, PLM no B/E observed liquid turbid no B/E observed 24 hrs orange solids in PLM no B/E observed cloudy liquid agg., no B/E (25:75) 0 -- PLM agg., irr., no B/E Eudragit agg., irr., no B/E L100-55 6 hrs light orange PLM no B/E observed solids in cloudy liquid 24 hrs orange solids in PLM no B/E observed cloudy solution (50:50) 0 -- PLM agg., irr., no B/E Eudragit agg., irr., no B/E L100-55 6 hrs orange solids in PLM no B/E observed cloudy liquid except 2 particles 24 hrs orange solids in PLM majority no B/E, a cloudy solurion few B/E particles in center clear view of B/E particles (75:25) 0 -- PLM agg., irr., no B/E Eudragit agg., irr., no B/E L100-55 6 hrs orange solids, PLM agg., no B/E liquid turbid agg., no B/E 24 hrs orange solids in PLM agg., no B/E cloudy liquid (a): approximate ratio of Rifaximin to polymer, by weight. (b): time is cumulative and approximate; 100 .mu.L of 0.1N HCl solution added into samples at t = 0. (c): 100 .mu.L of 0.1N HCl solution added into the sample after PLM analysis at 6 hrs.
TABLE-US-00007 TABLE 7 Physical Stability Assessment at 40.degree. C./75% RH/7 d Condition for Rifaximin and Rifaximin Dispersions Prepared in Methanol by Spray Drying Description (a) Habit/Description Analysis Results (100:0) orange solids, dry PLM agg., irr., no B/E Rifaximin-only (25:75) dark yellow solids, PLM agg., irr., no B/E PVP K-90 dry (50:50) orange solids, dry PLM agg., irr., no B/E PVP K-90 (75:25) orange solids, dry PLM agg., irr., no B/E PVP K-90 (25:75) light orange solids, PLM agg., irr., no B/E HPMC-P dry mDSC 148.degree. C. (T.sub.g, midpoint); 0.3 J/g .degree. C. (.DELTA.C.sub.p) (50:50) orange solids, dry PLM agg., irr., no B/E HPMC-P (75:25) orange solids, dry PLM agg., irr., no B/E HPMC-P (25:75) light orange solids, PLM agg., irr., no B/E HPMC-AS HG dry (50:50) orange solids, dry PLM agg., irr., no B/E HPMC-AS HG (75:25) orange solids, dry PLM agg., irr., no B/E HPMC-AS HG mDSC 177.degree. C. (T.sub.g, midpoint); 0.5 J/g .degree. C. (.DELTA.C.sub.p) (25:75) light orange solids, PLM agg., irr., no B/E HPMC-AS MG dry (50:50) orange solids, dry PLM agg., irr., no B/E HPMC-AS MG (75:25) orange solids, dry PLM agg., irr., no B/E HPMC-AS MG mDSC 152.degree. C. (T.sub.g, midpoint) (25:75) light orange solids, PLM agg., irr., no B/E Eudragit dry mDSC 140.degree. C. (T.sub.g, midpoint); L100-55 0.5 J/g .degree. C. (.DELTA.C.sub.p) (50:50) orange solids, dry PLM agg., irr., no B/E Eudragit L100-55 (75:25) orange solids, dry PLM agg., irr., no B/E Eudragit L100-55 (a): approximate ratio of Rifaximin to polymer, by weight. (b): analysis treated as non-cGMP.
TABLE-US-00008 TABLE 8 Physical Stability Assessment at 60.degree. C./Dry/7 d Condition for Rifaximin and Rifaximin Dispersions Prepared in Methanol by Spray Drying Description (a) Habit/Description Analysis Results (100:0) orange solids PLM agg., irr., no B/E Rifaximin-only (25:75) orange solids PLM agg., irr., no B/E PVP K-90 (50:50) orange solids PLM agg., irr., no B/E PVP K-90 (75:25) orange solids PLM agg., irr., no B/E PVP K-90 (25:75) light orange solids PLM agg., irr., no B/E HPMC-P (50:50) orange solids PLM agg., irr., no B/E HPMC-P (75:25) orange solids PLM agg., irr., no B/E HPMC-P (25:75) light orange solids PLM agg., irr., no B/E HPMC-AS HG (50:50) orange solids PLM agg., irr., no B/E HPMC-AS HG (75:25) orange solids PLM agg., irr., no B/E HPMC-AS HG (25:75) light orange solids PLM agg., irr., no B/E HPMC-AS MG (50:50) orange solids PLM agg., irr., no B/E HPMC-AS MG (75:25) orange solids PLM agg., irr., no B/E HPMC-AS MG (25:75) light orange solids PLM agg., irr., no B/E Eudragit L100-55 (50:50) orange solids PLM agg., irr., no B/E Eudragit L100-55 (75:25) orange solids PLM agg., irr., no B/E Eudragit L100-55 (a): approximate ratio of Rifaximin to polymer, by weight.
TABLE-US-00009 TABLE 9 Parameters for Rifaximin Solid Dispersions by Spray Drying Inlet temp. Aspirator Pump Inlet temp. Outlet temp. Spray rate (b) Description (a) (set, .degree. C.) % % (measured, .degree. C.) (measured, .degree. C.) mL/min (50:50) 120 95 40-30 120-119 60-45 9.6 HPMC-AS MG, ~10 g scale (25:75) 120 95 45-30 120-119 55-43 9.7 HPMC-P, ~10 g scale (a): approximate ratio of Rifaximin to polymer, by weight. (b): flow rates are estimated at 30% pump.
TABLE-US-00010 TABLE 10 Characterizations of 50:50 (w/w) Rifaximin/HPMC-AS MG Dispersion by Spray Drying Analysis Results XRPD x-ray amorphous mDSC 154.degree. C. (midpoint, T.sub.g) 0.4 J/g .degree. C. (.DELTA.Cp) TG-IR 0.5 wt % (loss up to 100.degree. C.) 199.degree. C. (onset, apparent decomp.) water, methanol and unknown volatiles
TABLE-US-00011 TABLE 11 Characterizations of 25:75 (w/w) Rifaximin/HPMC-P Dispersion by Spray Drying Analysis Results XRPD x-ray amorphous mDSC 152.degree. C. (midpoint, T.sub.g) 0.4 J/g .degree. C. (.DELTA.Cp) TG-IR 1.5 wt % (loss up to 100.degree. C.) 178.degree. C. (onset, apparent decomp.) water and methanol
TABLE-US-00012 TABLE 12 Sample Information of Rifaximin Dispersions for Dissolution Test in pH 6.52 FASSIF Buffer at 37.degree. C. Dissolution Solids Weight Volume of Description (a) Sample ID Vessel No (mg) Buffer (mL) (50:50) 4042-97-01 1 122.1 300 HPMC-AS MG 2 120.5 3 121.4 (25:75) 4103-01-01 4 242.5 300 HPMC-P 5 239.2 6 242.4 (a): approximate ratio of Rifaximin to polymer, by weight.
TABLE-US-00013 TABLE 13 Rifaximin Concentrations of 50:50 (w/w) Rifaximin/HPMC-AS MG Dispersion in pH 6.52 FASSIF Buffer at 37.degree. C. Dissolution Time Dilution Absorbance Concentration Vessel No (mm.) (c) (d) (.mu.g/mL) 1 5 -- 0.0159 0.34 10 -- 0.0346 2.53 15 -- 0.0569 5.13 30 -- 0.09655 9.75 60 -- 0.1626 17.46 90 -- 0.2216 24.35 120 -- 0.25625 28.39 1440 4 0.4093 184.99 2 5 2 0.02895 3.73 10 -- 0.0304 2.04 15 -- 0.04655 3.92 30 -- 0.104 10.62 60 -- 0.17755 19.21 90 -- 0.248 27.43 120 -- 0.3065 34.25 1440 4 0.3944 178.04 3 5 -- 0.0107 -0.26 10 -- 0.02555 1.47 15 -- 0.03975 3.13 30 -- 0.08735 8.68 60 -- 0.1766 19.10 90 -- 0.25815 28.61 120 -- 0.32055 35.89 1440 4 0.4202 190.08 (c): certain samples were diluted before analyzed to avoid the possibility of falling outside the linearity range of the instrument. (d): absorbance data less than 0.05 is below instrument detection limit and therefore concentration calculated from such absorbance is an approximate value.
TABLE-US-00014 TABL 14 Rifaximin Concentrations of 25:75 (w/w) Rifaximin/HPMC-P Dispersion in pH 6.52 FASSIF Buffer at 37.degree. C. Dissolution Time Dilution Absorbance Concentration Vessel No (mm.) (d) (e) (.mu.g/mL) 4 5 -- 0.01555 0.30 10 -- 0.03395 2.45 15 -- 0.0528 4.65 30 -- 0.12235 12.77 60 -- 0.2643 29.33 90 -- 0.37355 42.08 120 -- 0.455 51.58 1440 4 0.39465 178.16 5 5 -- 0.0329 2.33 10 -- 0.06805 6.43 15 -- 0.07905 7.71 30 -- 0.13745 14.53 60 -- 0.242 26.73 90 -- 0.32595 36.52 120 -- 0.40555 45.81 1440 4 0.38525 173.77 6 5 -- 0.0155 0.30 10 -- 0.057 5.14 15 -- 0.09415 9.47 30 -- 0.17145 18.49 60 -- 0.2724 30.27 90 -- 0.36815 41.45 120 -- 0.43155 48.84 1440 4 0.3838 173.09 (d): certain samples were diluted before analyzed to avoid the possibility of falling outside the linearity range of the instrument. (e): absorbance data less than 0.05 is below instrument detection limit and therefore concentration calculated from such absorbance is an approximate value.
TABLE-US-00015 TABLE 15 Averaged Concentrations of 50:50 (w/w) Rifaximin/HPMC-AS MG Dispersions in pH 6.52 FASSIF Buffer at 37.degree. C. Con- Average Description Dissolution Time centration Con- Standard (a) Vessel No (min.) (.mu.g/mL) centration Deviation (50:50) 1 5 0.34 1.27.sup.b 2.154 HPMC-AS 2 3.73 MG 3 -0.26 1 10 2.53 2.01.sup.b 0.5284 2 2.04 3 1.47 1 15 5.13 4.06.sup.b 1.008 2 3.92 3 3.13 1 30 9.75 9.69 0.970 2 10.62 3 8.68 1 60 17.46 18.59 0.977 2 19.21 3 19.10 1 90 24.35 26.80 2.202 2 27.43 3 28.61 1 120 28.39 32.85 3.945 2 34.25 3 35.89 1 1440 184.99 184.37 6.0455 2 178.04 3 190.08 .sup.aapproximate ratio of Rifaximin to polymer, by weight. .sup.babsorbance data less than 0.05 is below instrument detection limit and therefore concentration calculated from such absorbance is an approximate value.
TABLE-US-00016 TABLE 16 Averaged Concentrations of 25:75 (w/w) Rifaximin/HPMC-P Dispersions in pH 6.52 FASSIF Buffer at 37.degree. C. Con- Average Description Dissolution Time centration Con- Standard (a) Vessel No (min.) (.mu.g/mL) centration Deviation (25:75) 4 5 0.30 0.98.sup.b 1.171 HPMC-P 5 2.33 6 0.30 4 10 2.45 4.67.sup.b 2.030 5 6.43 6 5.14 4 15 4.65 7.28 2.442 5 7.71 6 9.47 4 30 12.77 15.26 2.935 5 14.53 6 18.49 4 60 29.33 28.78 1.840 5 26.73 6 30.27 4 90 42.08 40.02 3.041 5 36.52 6 41.45 4 120 51.58 48.75 2.886 5 45.81 6 48.84 4 1440 178.16 175.01 2.749 5 173.77 6 173.09 .sup.aapproximate ratio of Rifaximin to polymer, by weight. .sup.babsorbance data less than 0.05 is below instrument detection limit and therefore concentration calculated from such absorbance is an approximate value.
TABLE-US-00017 TABLe 17 Analysis of Rifaximin Dispersions after Dissolution Test in pH 6.52 FASSIF Buffer at 37.degree. C. Description Dissolution (a) Vessel No Analysis Results (50:50) 1 PLM no B/E observed HPMC-AS MG change view field, no B/E 2 PLM no B/E observed change view field, no B/E 3 PLM no B/E observed majority no B/E, only 1 B/E particle in view field (25:75) 4 PLM B/E flakes and blades HPMC-P 5 PLM no B/E material + B/E flakes 6 PLM no B/E material + B/E flakes & blades (a): approximate ratio of Rifaximin to polymer, by weight.
ABBREVIATIONS
TABLE-US-00018 [0231] Type Abbreviation Full Name/Description INSTRU- XRPD x-ray powder diffractometry MENTAL mDSC modulated differential scanning calorimetry TG-IR thermogravimetric infrared PLM polarized light microscopy UV ultraviolet spectroscopy POLYMER HPMC-AS hydroxypropylmethyl cellulose acetate succinate HPMC-P hydroxypropylmethyl cellulose phthalate Eudragit L100 anionic polymers with methacrylic acid as a functional group, dissolution at pH > 6.0 PVP K-90 polyvinylpyrrolidone, grade K-90 RESULTS T.sub.g glass transition temperature .DELTA.C.sub.p heat of capacity change amorph. amorphous agg. aggregates irr. irregular decomp. decomposition B birefringence E extinction
Example 2. Ternary Dispersion of 50:50 (w/w) Rifaximin:HPMC-AS MG
[0232] A ternary dispersion of 50:50 (w/w) Rifaximin:HPMC-AS MG with 5.9 wt % Pluronic F-127 was prepared in large quantity (containing approximately 110 g of Rifaximin) by spray drying. Disclosed herein are the analytical characterizations for Rifaximin ternary dispersion as-prepared and post-stress samples at 70.degree. C./75% RH for 1 week and 3 week, and post-stress sample at 40.degree. C./75% RH for 6 weeks and 12 weeks.
Characterization of Rifaximin Ternary Dispersion
[0233] Characterizations of the spray dried Rifaximin ternary dispersion (50:50 (w/w) rifaximin:HPMC-AS MG with 5.9 wt % Pluronic F-127) are described in Table 18.
TABLE-US-00019 TABLE 18 Characterizations of Combined Rifaximin Ternary Dispersion Solids-Spray Drying Sample ID Analysis Results (b) 4103- XRPD x-ray amorphous 74-01a mDSC 136.degree. C. (midpoint, T.sub.g) 0.4 J/g .degree. C. (.DELTA.Cp) TG-IR 0.7 wt % (loss up to 100.degree. C.) 202.degree. C. (onset, volatilization and apparent decomp.) methanol and possible acetic acid IR-ATR consistent with structure Raman consistent with structure SEM agglomerates of collapsed spheres PLM irregularly-shaped equant particles PSA d10 (.mu.m): 3.627, d50 (.mu.m): 8.233, d90 (.mu.m): 17.530 DVS 0.13 wt % (loss at 5% RH) 11.14 wt % (gain, 5-95% RH) 10.80 wt % (loss, 95-5% RH) 4074-89-01 XRPD x-ray amorphous (c) (b): temperatures are round to the nearest degree; .DELTA.Cp is rounded to one decimal places and wt % is rounded to one decimal place.
[0234] A high resolution XRPD pattern was acquired and material is x-ray amorphous (FIG. 46). By mDSC (FIG. 47), a single apparent T.sub.g is observed from the step change in the reversing heat flow signal at approximately 136.degree. C. with a heat capacity change at T.sub.g of approximately 0.4 J/g.degree. C.
[0235] Thermogravimetric analysis coupled with infra-red spectroscopy (TG-IR) was performed to analyze volatiles generated upon heating. The total weight loss of sample was approximately 0.7 wt % to 100.degree. C. and the dramatic change in the slope occurs at approximately 202.degree. C. (FIG. 48). The Gram-Schmidt plot corresponds to the overall IR intensity associated with volatiles released by a sample upon heating at 20.degree. C./min. By Gram-Schmidt, a negligible increase of intensity upon heating is observed before .about.7 minutes followed by a dramatic increase of intensity with the maximum at .about.11.8 min. The waterfall plot (data not shown) of this sample indicates volatile are released upon heating after .about.7 min (data is shown in FIG. 49 using the linked IR spectrum at different time points as an example) and volatiles were identified as residual methanol from the processing solvent in spray drying and possible acetic acid from HPMC-AS MG.
[0236] Vibrational spectroscopy techniques, including IR and Raman were employed to further characterize this ternary dispersion. The overlay of IR spectra for the dispersion and X-ray amorphous Rifaximin is shown in FIG. 50. Based on visual inspection, two spectra are very similar. Similar observations can be drawn from the comparison of Raman analysis (FIG. 51). The sample is composed of agglomerates of collapsed spheres. Particles sizes of spheres are not uniform, ranging from slightly larger to much less than 10 .mu.m.
[0237] PLM images (data not shown) of solids dispersed in mineral oil were collected, which indicate sample primarily is composed of irregularly-shaped equant particles approximately 5-15 .mu.m in length with some agglomerates 20-50 .mu.m in length. Particle size analysis (FIG. 52) indicates that 50% of particles have size less than 8.233 .mu.m and 90% of particles have size less than 17.530 .mu.m. Data was acquired in 2% (w/v) Lecithin in Isopar G.
[0238] The DVS isotherm of solids is shown in FIG. 53. The material exhibits a 0.13 wt % loss upon equilibration at 5% RH. Solids then gain 11.14 wt % between 5% and 95% RH and exhibits some hysteresis with 10.80 wt % loss upon desorption from 95% to 5% RH. XRPD analysis of the solids recovered after completion of the desorption step showed no evidence of sharp peaks indicative of a crystalline solid (FIG. 54).
[0239] Physical Stability Assessment on Rifaximin Ternary Dispersion
[0240] An assessment of physical stability of this rifaximin ternary dispersion is currently in progress by exposing solids to varied elevated temperature/relative humidity conditions, including 25.degree. C./60% RH, 40.degree. C./75% RH and 70.degree. C./75% RH for extended period of time. At designated time interval, such as at 1 week, 3 week, 6 week, and 12 weeks, selected samples were removed from stress conditions for characterization.
[0241] Table 19 summarized characterization results for the samples that stressed at 70.degree. C./75% RH condition 1 week and 3 weeks, and the sample that stressed at 40.degree. C./75% RH condition 6 weeks.
TABLE-US-00020 TABLE 19 Physical Stability Evaluation on Rifaximin Ternary Dispersion Habit/ Condition Time Description Analysis Results (a) 70.degree. C./75% RH 1 week orange solids, XRPD x-ray amorphous aggregates, no mDSC 134.degree. C. (midpoint, T.sub.g) B/E 0.4 J/g .degree. C. (.DELTA.Cp) SEM agglomerates of collapsed spheres KF 3.80% 70.degree. C./75% RH 3 weeks dark orange XRPD x-ray amorphous solids, mDSC 134.degree. C. (midpoint, T.sub.g) aggregates, no 0.4 J/g .degree. C. (.DELTA.Cp) B/E SEM agglomerates of collapsed spheres KF 3.19% 40.degree. C./75% RH 6 weeks orange solids, XRPD x-ray amorphous aggregates, no mDSC 133.degree. C. (midpoint, T.sub.g) B/E 0.4 J/g .degree. C. (.DELTA.Cp) SEM agglomerates of collapsed spheres KF 4.05% 40.degree. C./75% RH 12 weeks orange solids, XRPD x-ray amorphous aggregates, no mDSC 132.degree. C. (midpoint, T.sub.g) B/E 0.5 J/g .degree. C. (.DELTA.Cp) SEM agglomerates of collapsed spheres KF 3.37% (a): temperatures are round to the nearest degree; .DELTA.Cp is rounded to one decimal places.
[0242] For a sample that was stressed at 70.degree. C./75% RH for 1 week, solids are still x-ray amorphous according to XRPD (FIG. 55). A single T.sub.g at approximately 134.degree. C. was observed from the apparent step change in the reversing heat flow signal in mDSC with the change of heat capacity 0.4 J/g .degree. C., indicating the components of each dispersion remained intimately miscible after stress (FIG. 56). A non-reversible endotherm was observed at approximately 54.degree. C. which is likely due to the residual solvent from spray drying and moisture that materials absorbed during stress, which is confirmed by KF analysis that sample contains 3.80 wt % of water (KF analysis for Rifaximin ternary dispersion after 70.degree. C./75% RH 1 week; 1.2855 g--R1=3.72 and 0.988 g--R1=3.87%). The sample is composed of agglomerates of collapsed spheres and particles sizes of spheres are not uniform, which is similar to the as-prepared material.
[0243] For the sample that was stressed at 70.degree. C./75% RH for 3 weeks, although the color of the material appeared to be darker than the 1-week sample, characterization results for 3-week sample are similar to that for 1-week sample. Solids are also x-ray amorphous by XRPD (FIG. 55) and display a single T.sub.g at approximately 134.degree. C. by mDSC (FIG. 57). KF analysis indicates it contains 3.19 wt % of water (KF analysis for rifaximin ternary dispersion after 70.degree. C./75% RH 3 weeks; 1.2254 g--R1=3.45 and 1.1313 g--R1=2.93). By SEM (data not shown), the material has morphology similar to the as-prepared dispersion and 1-week stress sample, which is composed of agglomerates of collapsed spheres and particles sizes of spheres are not uniform.
[0244] For the sample that was stressed at 40.degree. C./75% RH for 6 weeks, solids are still x-ray amorphous according to XRPD (FIG. 55). It has a single T.sub.g at approximately 133.degree. C. by mDSC with the change of heat capacity 0.4 J/g .degree. C. (FIG. 58). It contains 4.05 wt % of water by KF (KF analysis for rifaximin ternary dispersion after 40.degree. C./75% RH 6 weeks; 1.0947 g--R1=3.47 and 1.2030--R1=4.63). By SEM (data not shown), the sample is composed of agglomerates of collapsed spheres and particles sizes of spheres are not uniform, which is similar to the as-prepared material.
[0245] For the sample that was stressed at 40.degree. C./75% RH for 12 weeks, solids are x-ray amorphous (FIG. 55) and display a single T.sub.g at approximately 132.degree. C. with the change of heat capacity 0.5 J/g .degree. C. (FIG. 59). It contains 3.37 wt % of water by KF (KF analysis for Rifaximin ternary dispersion after 40.degree. C./75% RH 12 weeks; 1.3687 g--R1=3.06 and 1.1630 g--R1=3.67). SEM analysis (data not shown) indicates that the sample is composed of agglomerates of collapsed spheres and particles sizes of spheres are not uniform, which is similar to the as-prepared material.
Example 3. Rifaximin Solid Dispersion Composition and Procedures
Rifaximin Ternary Dispersion Ingredients:
[0246] Rifaximin ternary dispersions (50:50 w/w Rifaximin:HPMC-AS MG with 5.9 wt % Pluronic F-127) were prepared from methanol using spray drying in closed mode suitable for processing organic solvents. Ingredients are listed as below in Table 20:
TABLE-US-00021 TABLE 20 Components of Rifaximin Solid Dispersion Component mg/g Purpose Rifaximin 472 active pharmaceutical ingredient Hydroxypropylmethyl 472 stabilizing agent cellulose acetate succinate (HPMC-AS), Type MG Pluronic F-127 56 wetting agent Methanol -- volatile; removed during process
Spray Drying Procedures:
[0247] Rifaximin ternary dispersions were prepared by spray drying in both small scale (.about.1 g API) and large scale (.gtoreq.34 g API in a single batch).
[0248] For the small-scale sample, rifaximin and then the methanol were added to a flask. The mixture was stirred at ambient temperature for .about.5 min to give a clear solution. HPMC-AS MG and Pluronic F-127 were added in succession and the sample was stirred for .about.1 hr. An orange solution was obtained.
[0249] For large-scale samples, a solution was prepared at .about.40.degree. C. Rifaximin and then methanol were added to a flask and the mixture was stirred at .about.40.degree. C. for .about.5 min until clear. HPMC-AS MG, and then Pluronic F-127 were added into the rifaximin solution under stirring at .about.40.degree. C. The sample continued to stir for .about.1.5 hr to 2 hr at this temperature. A dark red solution was obtained. The sample was removed from the hot plate and left at ambient to cool.
[0250] Experimental conditions to prepare Rifaximin ternary solutions are summarized in Table 21 below:
TABLE-US-00022 TABLE 21 Experimental Conditions to Prepare Rifaximin Ternary Solutions weight Concen- (API/HPMC AS tration Solvent MG/Pluronic F127, g) Temperature (g/L) methanol, 100 mL 1.0535/1.0529/0.1249 ambient 22.3 methanol, 1000 mL 34.07/34.07/4.02 ~40.degree. C. 72.2 methanol, 1250 mL 50.34/50.32/5.94 ~40.degree. C. 85.3 methanol, 1250 mL 50.16/50.14/5.92 ~40.degree. C. 85 methanol, 1250 mL 50.05/50.06/5.91 ~40.degree. C. 85
[0251] During the spray drying process, both the small and large scale rifaximin ternary solutions were kept at ambient temperature. The pump % was decreased during the process in an attempt to control outlet temperature above 40.degree. C. The operating parameters used for processing are presented in Table 22 below.
TABLE-US-00023 TABLE 22 Operating Parameters Used For Processing Rifaximin SD Inlet Spray rate temp. Aspirator Pump Inlet temp. Outlet temp. (b) Description (a) (set, .degree. C.) % % (measured, .degree. C.) (measured, .degree. C.) mL/min 50:50 120 95 35 120 60-55 10.4 Rifaximin:HPM 120 95 65-30 120-119 61-42 23 C-AS MG 120 95 50-30 120-119 67-43 16 5.9 wt % 120 95 50-30 120-119 65-43 16 Pluronic F-127 120 95 50-30 120-119 67-43 16 (a): 50:50 is approximate ratio of Rifaximin to polymer, by weight; 5.9 wt % Pluronic is weight fraction to 50:50 rifaximin:HPMC-AS MG dispersion. (b): Flow rates are estimated. Flow rate for 4103-41-01 was measured at pump 35%; for 4103-56-01 was measured at pump 65%, while for others were measured at pump 50%.
[0252] Solids recovered after spray drying were dried at 40.degree. C. under vacuum for 24 hours and then stored at sub-ambient temperatures over desiccant.
Spray Drying Process Parameters:
[0253] Spray Dryer--PSD 1 [0254] Two Fluid Niro Nozzle [0255] Nozzle orifice--1 mm [0256] Inlet gas temperature--125.+-.5 deg C. [0257] Process gas flow (mmH2O)--44 [0258] Atomizing gas pressure--0.7-1 bar [0259] Feed rate--4.7 kg/Hr [0260] Outlet temperature--55.+-.3 deg C. [0261] Solution temperature--36 deg C. [0262] Post spray drying vacuum dry at 40 deg C. for 48 hrs
Example 4
[0263] Exemplary formulations for micronized, API, amorphous, solid dispersion and micronized capsules are below in Table 23. These capsules were used in the dog study of Example 5.
TABLE-US-00024 TABLE 23 Capsule Formulation composition (Solid Dispersion (SD) Capsules) Micronized Amorphous Micronized Capsules API Capsules Capsules SD Capsules Tablets Ingredients % g/dose % g/dose % g/dose % g/dose % g/dose Rifaximin 95.5 2.2 47.2 2.2 51.7 2.2 42.47 2.2 50 2.2 Ac-di-sol 4.5 0.1 5 0.23 5 0.21 10.02 0.52 7.5 0.33 Mannitol 160C 47.8 2.23 43.3 1.84 Pluronic 188 5.04 0.26 HPMC AS 42.47 2.2 Avicel 113 26 1.14 Avicel 112 15 0.66 Magnesium 1 0.04 Stearate Cab-o-sil 0.5 0.02 Avicel CL-611 Mannitol 160C Total 100 2.3 100 4.66 100 4.26 100 5.18 100 4.4
TABLE-US-00025 TABLE 24 Manufacture of rifaximin/HPMC-AS/Pluronic 275 mg Capsules % mg/ Theo. Actual Component Formula caps Qty (g) Qty (g) Rifaximin 42.47 275 113.7 113.7 HPMC-AS 42.47 275 113.7 113.7 (type MG) Pluronic F-127 5.04 32.63 13.49 13.49 Sodium 10.02 64.87 26.82 26.82 Croscarmellose Hard Gelatin 1 N/A 300 300 Capsule (size 000) Clear Total 100 647.5 267.7 g
Blending/Encapsulation Procedure:
[0264] To form the capsules sodium croscarmellose was added to the bag of SD rifaximin dispersion and bag blend for 1 minute, and then the material was added to the V-blender and blended for 10 minutes at 24 rpm.
[0265] The material was then discharged into a stainless steel pan and record the height of material in the pan. Empty capsules were tared using an analytical balance, then the capsules were filled by depressing into the bed of material. The weight is adjusted within + or -5% of target fill weight of 647.5 mg (acceptable fill range 615.13-679.88 mg).
[0266] FIGS. 61-63 show the rifaximin solid dispersion (SD) capsules in various buffers; with and without SDS; and compared to amorphous rifaximin. FIG. 61 shows results of dissolution studies of rifaximin SD capsules in acid phase: 0.1 N HCl with variable exposure times in a buffer containing 0.45% SDS at pH 6.8. FIG. 62 shows results of dissolution studies of rifaximin SD capsules in acid phase for 2 hours buffered at pH 6.8 with and without SDS. FIG. 63 shows results of dissolution studies of rifaximin SD capsules in acid phase in a phosphate buffer at pH 6.8 with 0.45% SDS compared to amorphous rifaximin. As shown in the FIGS. 61-63 rifaximin SD near 100% dissolution is achieved in 0.45% SDS and the SD formulation dissolves more slowly than the amorphous rifaximin.
Example 5. Pharmacokinetic (PK) Studies of Solid Dispersion in Capsules
[0267] Presented herein are dog pharmacokinetics (PK) studies comparing various forms of rifaximin. PK following administration of rifaximin API in capsule, micronized API in capsule, nanocrystal API in capsule (containing surfactant), amorphous in capsule, and solid dispersion (SD) in capsule were tested.
[0268] In the SD dosage form, the polymer used was HPMC-AS at a drug to polymer ratio of 50:50. The formulation also comprised pluronic F127 and crosscarmellose sodium (see Example 4).
[0269] A brief study design: male beagle dogs (N=6, approximately 10 kg) received rifaximin 2200 mg in the dosage forms described above as a single dose (capsules, 275 mg, 8 capsules administered in rapid succession), in a cross-over design with one week washout between phases. Blood was collected at timed intervals for 24 h after dosage administration, and plasma was harvested for LC-MS/MS analysis. The mean concentrations are shown in FIG. 60.
[0270] Table 25 shows the PK parameters. From the table it can be seen that systemic exposure of the solid dispersion formulation is greater than that of amorphous or crystalline form (API) of rifaximin.
TABLE-US-00026 TABLE 25 PK Parameters of API, Amorphous and Solid Dispersion to Dogs Half-life* Tmax Cmax AUClast AUCINF_obs AUC_0-24 ID h h ng/mL h*ng/mL h*ng/mL h*ng/mL 901_API 16.76 0.5 65.5 101 118 101 902_API 9.41 1 3.83 25 29 25 903_API 10.03 1 197 344 360 344 904_API 3.56 1 1.21 5 6 6 905_API 2.94 1 1.53 5 6 6 906_API 24 0.52 7 7 mean 6.98 1 44.93 81 104 82 SD [0.5-24] 78.75 134 150 134 901_amorph 5.38 1 536 1407 1421 1407 902_amorph 5.93 2 4100 12258 12762 12258 903_amorph 6.25 2 1050 3375 3523 3375 904_amorph 4.77 2 763 2291 2306 2291 905_amorph 7.72 1 1200 2041 2059 2041 906_amorph 5.63 2 704 2076 2090 2076 mean 5.88 2 1392.17 3908 4027 3908 SD [1-2] 1348.24 4141 4334 4141 901_SD amorph 6.66 2 491 1354 1394 1354 902_SD amorph 2.04 2 6550 25140 25149 25140 903_SD amorph 2.8 4 2410 10490 10508 10490 904_SD amorph 2.24 1 1410 6343 6350 6343 905_SD amorph 3.97 2 2860 7885 7895 7885 906_SD amorph 4.89 2 1900 4532 4558 4532 mean 3.01 2 3026 10878 10892 10878 SD [1-4] 2043.58 8267 8264 8267 *geometric mean **median and range
[0271] API exposures were low, in keeping with what has been previously observed for rifaximin. In contrast, mean exposures (AUCinf) following amorphous and SD rifaximin administration were substantially higher, with .about.40- and .about.100-fold greater exposure, respectively, as compared with API. Variability was high in all three dose groups. In general, the shapes of all three profiles were similar, suggesting effects of the dosage forms on bioavailability without effects on clearance or volume of distribution.
Example 6. Human Clinical Studies
[0272] Rifaximin SDD with 10% CS formulation was used in human clinical studies. FIG. 65A-B show the kinetic solubility of rifaximin SD granules 10% wt CS FaSSIF or 10% wt CS FeSSIF (a) and the dissolution profiles of SDD tablet 10% CS in 0.2% SLS at pH 4.5, 5.5 and 7.4. As shown in FIG. 65A-B, rifaximin SDD 100%, or near 100%, dissolution is achieved in 0.2% SLS, pH 4.5, 5.5 and 7.4. FIG. 66A-B show that release can be delayed up to two hours and extended up to three hours.
Example 7. Effects of Media pH on Dissolution
[0273] FIG. 67A-B, FIG. 68A-B, FIG. 69A-B, and FIG. 70A-B show the effects of media pH on Rifaximin SDD tablet SDD tablet dissolution at various levels of CS: 0%, 2.5%, 5%, and 10% CS. FIG. 67A-B and FIG. 68A-B show dissolution profiles of SDD tablet with 0%, 2.5%, 5% or 10% CS in 0.2% SDS at 2 hours pH 2.0, pH 4.5, 0.2% SDS pH 5.5, or 0.2% SDS, pH 7.4. FIG. 69A-B and FIG. 70A-B show the dissolution profiles of SDD tablet 2.5% CS, 0% CS, 10% CS and 5% CS in 0.2% SLS, pH4.5, 0.2% SLS, pH 5.5 and 0.2% SLS, pH 7.4. FIG. 71A-B show CS release mechanism.
Example 8
[0274] Described herein are the preparation and characterization of rifaximin quaternary dispersions with antioxidants. Antioxidants used were butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT) and propyl gallate (PG).
Sample preparation and Characterization
[0275] Three rifaximin quaternary samples were prepared by spray drying from methanol. Spray drying parameters are summarized in Table 26. Table 2 Parameters for Samples Prepared by Spray Drying
TABLE-US-00027 TABLE 26 Inlet Outlet Spray temp. Inlet temp. temp. rate (set, Aspirator Pump (measured, (measured, (a) Sample ID .degree. C.) % % .degree. C.) .degree. C.) mL/min 0.063 wt % 120 95 45-35 120-124 61-49 19 of BHA in the dispersion 0.063 wt % 120 95 45-35 120-121 60-50 20 of BHT in the dispersion 0.094 wt % 120 95 45-35 119-120 60-48 20 of propyl gallate in the dispersion (a): flow rates are estimated based on initial pump % of 45%.
TABLE-US-00028 TABLE 27 Characterization of Rifaximin Quaternary Samples Habit/Description Analysis Results (b) orange solids, XRPD x-ray amorphous irregular mDSC 133.degree. C. (midpoint, T.sub.g) aggregates, no B/E 0.3 J/g .degree. C. (.DELTA.Cp) orange solids, XRPD x-ray amorphous irregular mDSC 133.degree. C. (midpoint, T.sub.g) aggregates, no B/E 0.4 J/g .degree. C. (.DELTA.Cp) orange solids, XRPD x-ray amorphous irregular mDSC 134.degree. C. (midpoint, T.sub.g) aggregates, no B/E 0.4 J/g .degree. C. (.DELTA.Cp)
[0276] A small sub-lot from each of spray dried materials was visually inspected by PLM and then characterized by XRPD and mDSC. Characterization results are summarized in Table 27. The prepared materials are x-ray amorphous, as shown in FIG. 72 the overlay of XRPD patterns, which agree with their PLM observations.
[0277] In the mDSC, each of material displays a single apparent T.sub.g in the reversing heat flow signal at approximately 133.degree. C. (FIG. 73, with 0.063 wt % BHA), 133.degree. C. (FIG. 74, with 0.063 wt % BHT), and 134.degree. C. (FIG. 75, with 0.094 wt % PG), which is consistent with the T.sub.g of the spray dried rifaximin ternary dispersion of 47.2:47.2:5.6 w/w/w/ rifaximin/HPMC-AS MG/Pluronic F-127 (135 or 136.degree. C.).
Example 9: Rifaximin Solid Dispersions
[0278] This example sets forth exemplary microgranules of rifaximin and pharmaceutical compositions comprising the same.
[0279] Spray dry dispersion (SDD), solid dispersion, amorphous solid dispersion are used interchangabley herein to refer to the rifaximin formulations.
[0280] The complete statement of the components and quantitative composition of Rifaximin Solid Dispersion Formulation (Intermediate) is given in Table 28
TABLE-US-00029 TABLE 28 Composition of Rifaximin Solid Dispersion Formulation Component Quantity (%) Function Rifaximin Drug 42.48 Active Ingredient Substance Hypromellose Acetate 42.48 Solubility Enhancer Succinate (HPMC-AS) Poloxamer 407 5.04 Surfactant Croscarmellose 10.00 Dissolution Enhancer Sodium
Composition of Rifaximin Solid Dispersion IR Capsule
TABLE-US-00030 [0281] TABLE 29 Composition of Rifaximin solid dispersion IR capsule Component Quantity Function Rifaximin solid dispersion 75 mg-275 mg* Active ingredient (amorphous) Hard Gelatin capsules 1 unit Capsule Coni-Snap, Size 000, Transparent *Rifaximin dose equivalent
Description of Manufacturing Process and Process Controls
Manufacturing Process for Rifaximin Solid Dispersion Formulation
Table 30 Sets Forth the Manufacture of Rifaximin Solid Dispersion Microgranules
TABLE-US-00031 [0282] TABLE 30 Component Process Methanol Rifaximin HPMC-AS Poloxamer 407 ##STR00001## Feed Solution ##STR00002## Croscarmellose Sodium ##STR00003##
Manufacturing Process for Rifaximin Solid Dispersion IR Capsules
[0283] The manufacturing process the Rifaximin solid dispersion IR capsules is given in Table 31.
TABLE-US-00032 TABLE 31 Manufacture of Rifaximin solid dispersion microgranules in IR capsules Component Process Rifaximin solid dispersion Formulation ##STR00004##
Exemplary spray drying processes are set forth in Table 32.
TABLE-US-00033 TABLE 32 Spray Drying Process: Spray Dryer--PSD 1 Two Fluid Niro Nozzle Nozzle orifice--1 mm Inlet gas temperature--125 .+-. 3 deg C. Process gas flow (mmH2O)--44 Atomizing gas pressure--1 bar Feed rate--4.7 kg/Hr Outlet temperature--55 .+-. 3 deg C. Solution temperature--36 deg C. Post spray drying vacuum dry at 40 deg C. for 48 hrs Micronized Amorphous Amorphous Micronized Caps API Caps Caps SD caps Tab Ingredients % g/dose % g/dose % g/dose % g/dose % g/dose Rifaximin 95.5 2.2 47.2 2.2 51.7 2.2 42.47 2.2 50 2.2 Ac-di-sol 4.5 0.1 5 0.23 5 0.21 10.02 0.52 7.5 0.33 Mannitol 47.8 2.23 43.3 1.84 160C Pluronic 5.04 0.26 188 HPMC AS 42.47 2.2 Avicel 113 26 1.14 Avicel 112 15 0.66 Magnesium 1 0.04 Stearate Cab-o-sil 0.5 0.02 Avicel CL- 611 Mannitol 160C Total 100 2.3 100 4.66 100 4.26 100 5.18 100 4.4
Example 10: Characterization of Drug Product Samples Containing Rifaximin Solid Dispersion
[0284] Disclosed herein is dissolution data for roller compacted materials of Solid Dispersion Rifaximin with varying levels (0, 2.5%, 5%, and 10%) of croscarmellose sodium.
[0285] Three roller compacted material of Amorphous Solid Dispersion Rifaximin with varying levels (0, 2.5%, 5%) of croscarmellose sodium were dissolution tested. Results are compared to dissolution of the rifaximin granules with 10% croscarmellose sodium.
Dissolution Studies with USP Paddle Method
[0286] Dissolution tests were performed on as received roller compacted materials of Solid Dispersion Rifaximin with 0, 2.5 wt %, and 5 wt % croscarmellose sodium. Powders of solids were directly added into pH 6.5 FaSSIF buffer with gentle agitation of the media (50 rpm paddle stirrer) at 37.degree. C. for 24 hrs.
[0287] At designated time points of 5, 10, 20, 30, 60, 90, 120, 240 and 1440 minutes, aliquots were removed from each of the samples. Analysis of the date indicates that an increase in rifaximin concentration is apparent with the rising croscarmellose sodium level in materials, particularly in the early stage of the dissolution. After 24 hrs, the rifaximin concentration from granules containing 5 wt % croscarmellose sodium is similar to granules with 10 wt % croscarmellose sodium.
Example 11: Characterization of Rifaximin Solid Dispersion Powder 42.48% w/w
[0288] Described herein is the characterization of Rifaximin Solid Dispersion Powder 42.48% w/w. Dissolution testing was also performed on the material at pH 6.5 in FaSSIF at 37.degree. C.
[0289] A sample of rifaximin ternary dispersion was characterized by XRPD, mDSC, TG-IR, SEM and KF.
[0290] X-ray powder diffraction (XRPD) analysis using a method for Rifaximin Solid Dispersion Powder 42.48% w/w was conducted. The XRPD pattern by visual inspection is x-ray amorphous with no sharp peaks (FIG. 76). By mDSC a single apparent T.sub.g is observed from the step change in the reversing heat flow signal at approximately 134.degree. C. with a heat capacity change at T.sub.g of approximately 0.36 J/g.degree. C.
[0291] Thermogravimetric analysis coupled with infra-red spectroscopy (TG-IR) was performed to analyze volatiles generated upon heating. The total weight loss of sample was approximately 0.4 wt % to 100.degree. C., and a dramatic change in the slope occurs at approximately 190.degree. C. which is likely due to decomposition. The Gram-Schmidt plot corresponds to the overall IR intensity associated with volatiles released by a sample upon heating at 20.degree. C./min. Gram-Schmidt indicates that volatiles are released upon heating after .about.8 min, and volatiles were identified as residual methanol from the processing solvent in spray drying and possible acetic acid from HPMC-AS MG.
[0292] KF analysis indicates that the material contains 1.07 wt % water [(1.00+1.13)/2=1.07%].
Example 12: Methods for Spray Drying Rifaximin Ternary Dispersion (50:50 w/w Rifaximin:HPMC-AS MG with 5.9 wt % Pluronic F-127)
[0293] Provided herein are procedures to spray dry Rifaximin ternary dispersion (50:50 w/w Rifaximin:HPMC-AS MG with 5.9 wt % Pluronic F-127).
[0294] Rifaximin ternary dispersions (50:50 w/w Rifaximin:HPMC-AS MG with 5.9 wt % Pluronic F-127) were prepared from methanol using Buchi B-290 Mini Spray Dryer in closed mode suitable for processing organic solvents. Ingredients are listed in Table 33 below:
TABLE-US-00034 TABLE 33 No. Component mg/g Purpose 1 Rifaximin 472 active pharmaceutical ingredient 2 Hydroxypropylmethyl cellulose 472 stabilizing agent acetate succinate (HPMC-AS), Type MG 3 Pluronic F-127 56 wetting agent 4 Methanol -- volatile; removed during process
[0295] Rifaximin ternary dispersions were prepared by spray drying in both small scale (.about.1 g API) and large scale (.gtoreq.34 g API in a single batch).
[0296] For a small-scale sample, rifaximin and then the methanol were added into a clean flask. The mixture was stirred at ambient for .about.5 min to give a clear solution. HPMC-AS MG and Pluronic F-127 were added in succession and the sample was stirred for .about.1 hr. An orange solution was obtained.
[0297] For a large-scale sample, a solution was prepared at .about.40.degree. C. Rifaximin and then methanol were added to a clean flask and the mixture was stirred at .about.40.degree. C. for .about.5 min until clear. HPMC-AS MG, and then Pluronic F-127 were added into the rifaximin solution under stirring at .about.40.degree. C. The sample continued to stir for .about.1.5 hr to 2 hr at this temperature. A dark red solution was obtained. The sample was removed from the hot plate and left at ambient to cool.
[0298] Experimental conditions to prepare Rifaximin ternary solutions are summarized in Table 34 below:
TABLE-US-00035 TABLE 34 weight Concen- (API/HPMC AS MG/ tration Solvent Pluronic F127, g) Temperature (g/L) methanol, 100 mL 1.0535/1.0529/0.1249 ambient 22.3 methanol, 1000 mL 34.07/34.07/4.02 ~40.degree. C. 72.2 methanol, 1250 mL 50.34/50.32/5.94 ~40.degree. C. 85.3 methanol, 1250 mL 50.16/50.14/5.92 ~40.degree. C. 85.0 methanol, 1250 mL 50.05/50.06/5.91 ~40.degree. C. 85.0
[0299] During spray drying process, both the small and large scale rifaximin ternary solutions were kept at ambient temperature. The Pump % was decreased during process in attempt to control outlet temperature above 40.degree. C. The operating parameters used for processing are presented in Table 35 below.
TABLE-US-00036 TABLE 35 Inlet temp. Aspirator Pump Inlet temp. Outlet temp. Spray rate (b) Description (a) (set, .degree. C.) % % (measured, .degree. C.) (measured, .degree. C.) mL/min 50:50 120 95 35 120 60-55 10.4 Rifaximin:HPMC-AS 120 95 65-30 120-119 61-42 23 MG 120 95 50-30 120-119 67-43 16 5.9 wt % Pluronic F-127 120 95 50-30 120-119 65-43 16 120 95 50-30 120-119 67-43 16 (a): 50:50 is approximate ratio of Rifaximin to polymer, by weight; 5.9 wt % Pluronic is weight fraction to 50:50 Rifaximin:HPMC-AS MG dispersion. (b): flow rates are estimated. Flow rate for 4103-41-01 was measured at pump 35%; for 4103-56-01 was measured at pump 65%, while for others were measured at pump 50%.
[0300] Solids recovered after spray drying were dried at 40.degree. C. under vacuum for 24 hours and then stored at sub-ambient (freezer) over desiccant.
Example 13. Non-Clinical Data-Form/Formulation Comparison and Dose Ranging in Dogs
[0301] Described herein is non-clinical data, form/formulation comparison in dogs and SDD dose ranging in dogs. FIG. 77 indicates the results of two studies conducted to characterize the pharmacokinetics of rifaximin following administration of varying forms and formulations following a single oral dose. Blood samples were collected at timed intervals over the 24 h after single dose administration (2200 mg total dose in each case) and processed to plasma for analysis of rifaximin concentrations. PK parameters were estimated by noncompartmental methods. The results are shown in FIG. 77. Of the forms/formulations shown, the spray-dried dispersion showed that the highest exposure, and therefore the highest bioavailability, resulted from administration of the SDD formulation (dosed as SDD powder in gelatin capsules). In order of decreasing exposure among forms dosed in gelatin capsule formulation, SDD>amorphous>iota>micronzed>eta>current crystalline API. Lower in systemic exposure than all of those are the micronized suspension formulation (reconstituted powder for oral suspension) and the current 550 mg Xifaxan tablet. Table 36, below, shows Pk parameters for dog forms.
TABLE-US-00037 TABLE 36 Tmax Cmax AUCall AUCINF_obs HL_Lambda_z h h ng/mL h*ng/mL h*ng/mL Eta 9.70 1.5 162.28 434.14 608.14 Iota 6.56 2 276.50 718.23 739.94 Amorphous 5.82 2 1392.17 3907.84 4026.86 API capsules 5.64 1 44.93 81.20 103.83 SDD 3.16 2 2603.50 9290.71 9308.83 Micronized capsules 8.10 1 473.43 894.65 905.97 Micronized suspension 5.22 3 0.68 5.11 8.41 Micronized tablets 4.77 5 0.83 6.81 10.20 Nanocrystal capsules 5.01 5 0.99 9.05 8.70
[0302] FIG. 78 shows the results of the dog dose escalation, in which dogs received single doses of the SDD formulation in capsules, at doses from 150 mg to 2200 mg. The results indicate an essentially linear dose escalation (increases in exposure that are approximately proportional to increase in dose) up to 550 mg, followed by a greater-than-proportional increase at 1100 mg and 2200 mg. This is quite unusual in the linear range in that the current crystalline form of rifaxmin does not dose escalate, generally, exposure does not increase substantially on increasing dose. The greater than dose proportional increase on increasing dose is also remarkable and suggests that, at the higher doses, rifaximin is saturating intestinal P-glycoprotein transport that would otherwise limit systemic absorption, thereby allowing increased absorption.
Example 14. Human Studies
[0303] Described herein are clinical studies carried in ten male human subjects. FIG. 79 sets out the quotient study design for rifaximin SDD dose escalation. FIG. 80 outlines the dose escalation/regional absorption study, dose escalation/dose selection. FIGS. 81 and 82 show representative subject data from an exemplary dose escalation study. Mean data (linear scale and log scale) is shown in FIGS. 83 and 84, respectively. Mean profiles, log scale. Terminal phases are parallel, in clearance mechanisms. A summary of rifaximin SDD dose escalation is shown indicating that it is likely that there is not saturation of any metabolic or other systemic FIG. 85. To summarize, there are roughly dose proportional increases in exposure (C.sub.max and AUC) with increases in dose, as shown by C.sub.max multiple and AUC multiple columns. T.sub.max is not delayed by dose increases, further indicating an early absorption window (corroborated by regional absorption data). The percent of dose in urine is remarkable in that it stays low, approximately 0.2% or less of the dose excreted over 24 h. This result is surprising in that this is quite low in spite of the significant increases in systemic exposure as compared with the crystalline formulation. Taken together, the results indicate a considerably increased solubility that presumably leads to increased local/lumenal soluble rifaximin, with accompanying increases in systemic exposure, but without significant increases in urinary excretion that are reflective of percent of rifaximin dose absorbed.
[0304] Dose/dosage form comparisons are shown in FIGS. 86 and 87. The tables compare SDD at increasing doses to the current crystalline formulation in terms of systemic PK. As noted in FIG. 87, as compared to the PK of rifaximin from the current formulation, the SDD formulation at the same dose shows an approximate 6.4-fold increase in C.sub.max and an approximate 8.9-fold increase in AUC. Nonetheless, these exposures are less than those observed in any hepatic impaired subject with the current tablet formulation.
Example 15. Exemplary Tablet Formulations
[0305] According to certain exemplary embodiments, microgranules, blends and tablets are formulated as set forth in Table 37, below
TABLE-US-00038 Rifaximin 5DD Granules % w/w % w/w % w/w % w/w Component Function (0% CS) (2.5% CS) (5% CS) (10% CSf Rifaximin Drug 47.2 46.02 44.84 42.48 HPMC-AS Polymer 47.2 46.02 44.84 42.48 Pluronic F-127 Wetting agent 5.6 5.46 5.32 5.04 Croscarmellose Rate 0 2.5 5 10 Na (CS) controlling Total 100 100 100 100
TABLE-US-00039 Granule Blend mg/Tab mg/Tab mg/Tab mg/Tab Roller Compacted Granules 635.59 652.34 669.05 706.21 Granules Avicel PH102 Filler 166 149.18 132.52 95.38 Croscarmellose Na Disintegrant 42.5 42.5 42.5 42.5 (Extra-granuler) Cab-O-Sil Glidant 1.7 1.7 1.7 1.7 Magnesium Stearate Lubricant 4.25 4.25 4.25 4.25 Total 850.04 849.97 850.02 850.04
TABLE-US-00040 Overall Rifaximin Tablet Composition % w/w % w/w % w/w % w/w Component Function (0% CS) (2.5% CS) (5% CS) (10% CS) Rifaximin Drug 35.29 35.32 35.29 35.29 HPMC-AS Polymer 35.29 35.32 35.29 35.29 Pluronic F-127 Wetting agent 4.19 4.19 4.19 4.19 Croscarmellose Rate 0.00 1.92 3.94 8.31 Na (intra- controlling granuler} Avicel PH102 Filler 19.53 17.55 15.59 11.22 Croscarmellose Disintegrant 5.00 5.00 5.00 5.00 Na (Extra- granuler) Cab-O-Sil Glidant 0.20 0.20 0.20 0.20 Magnesium Lubricant 0.50 0.50 0.50 0.50 Stearate Total 100 100 100 100
* * * * *




D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012
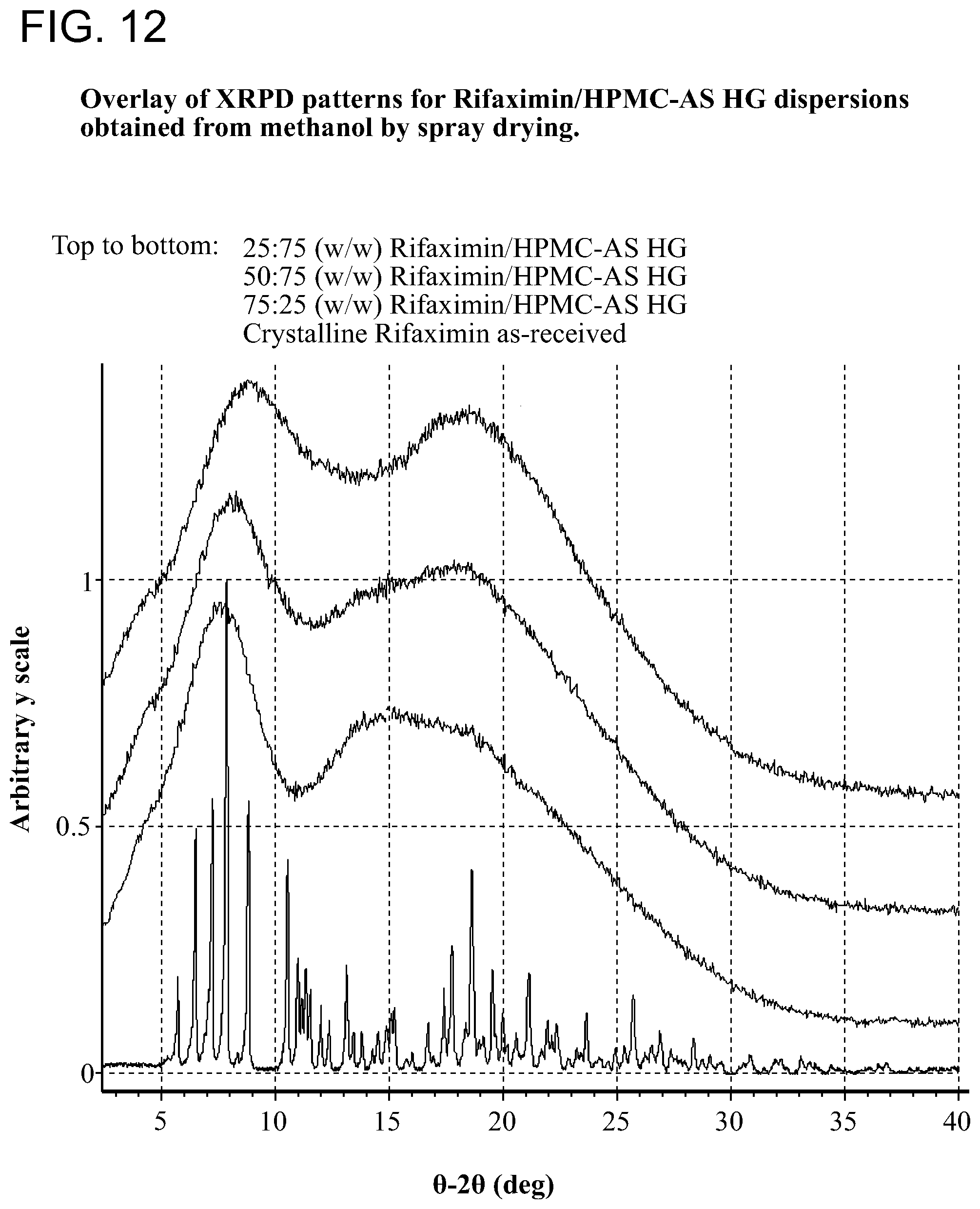
D00013
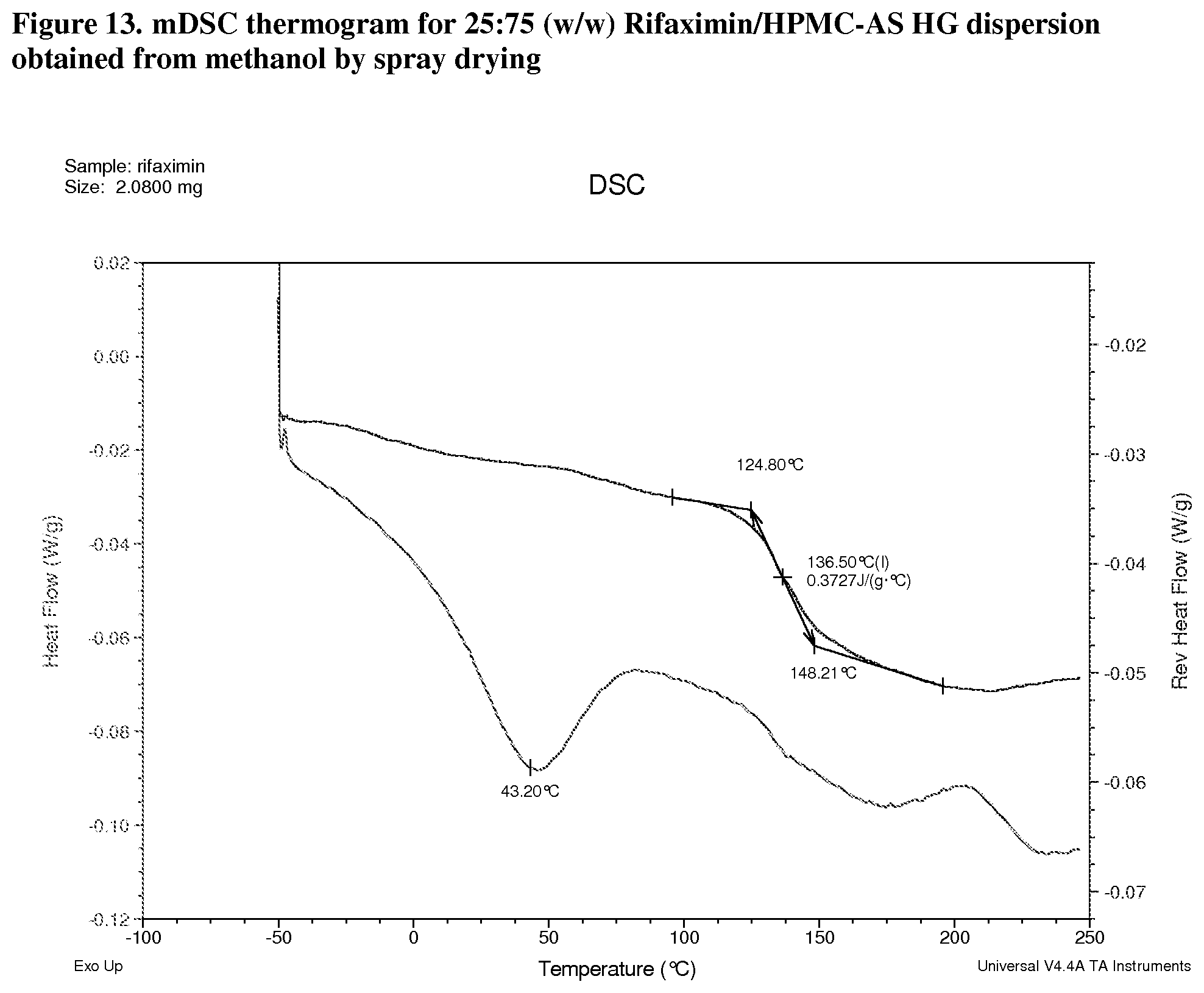
D00014
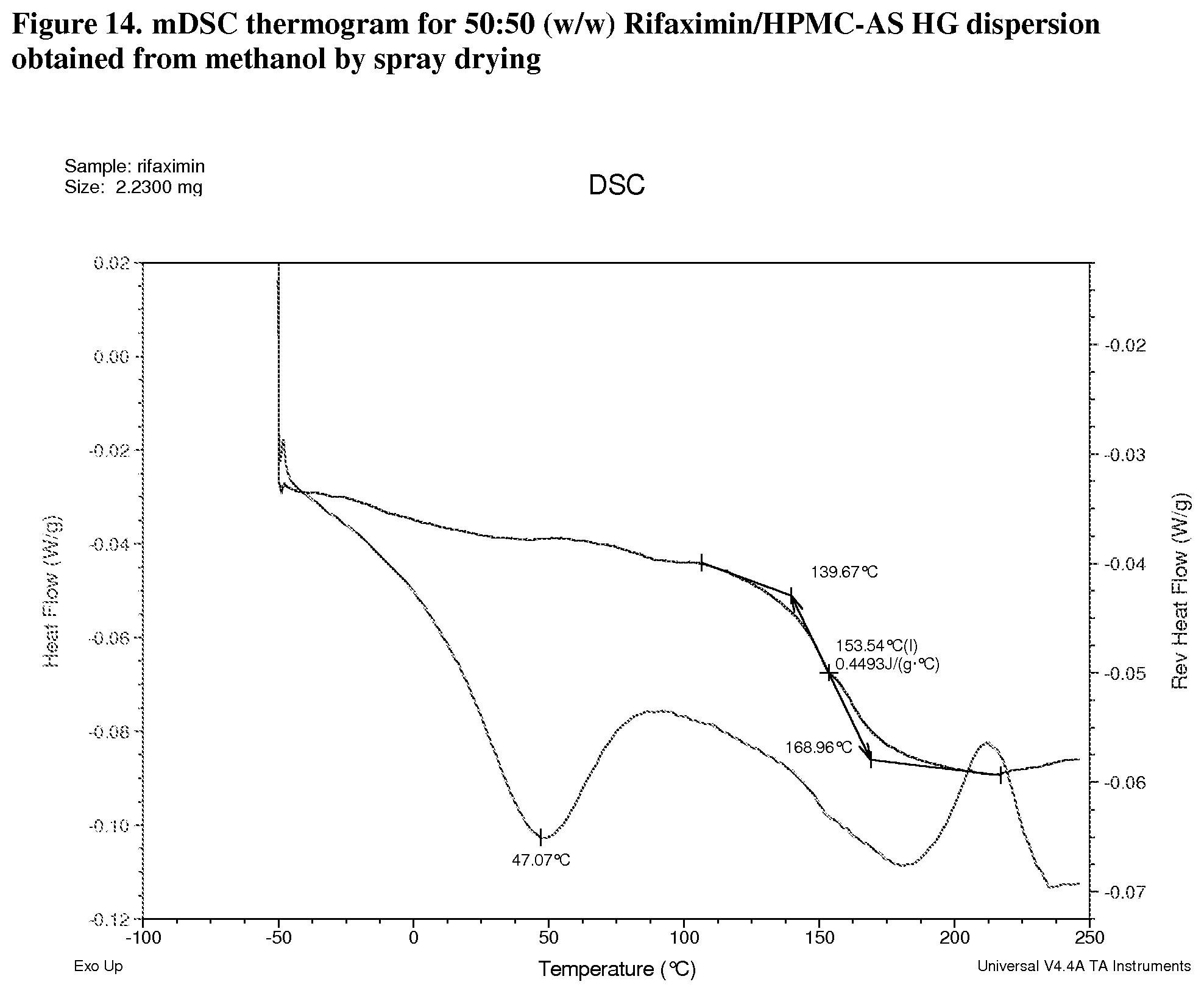
D00015

D00016
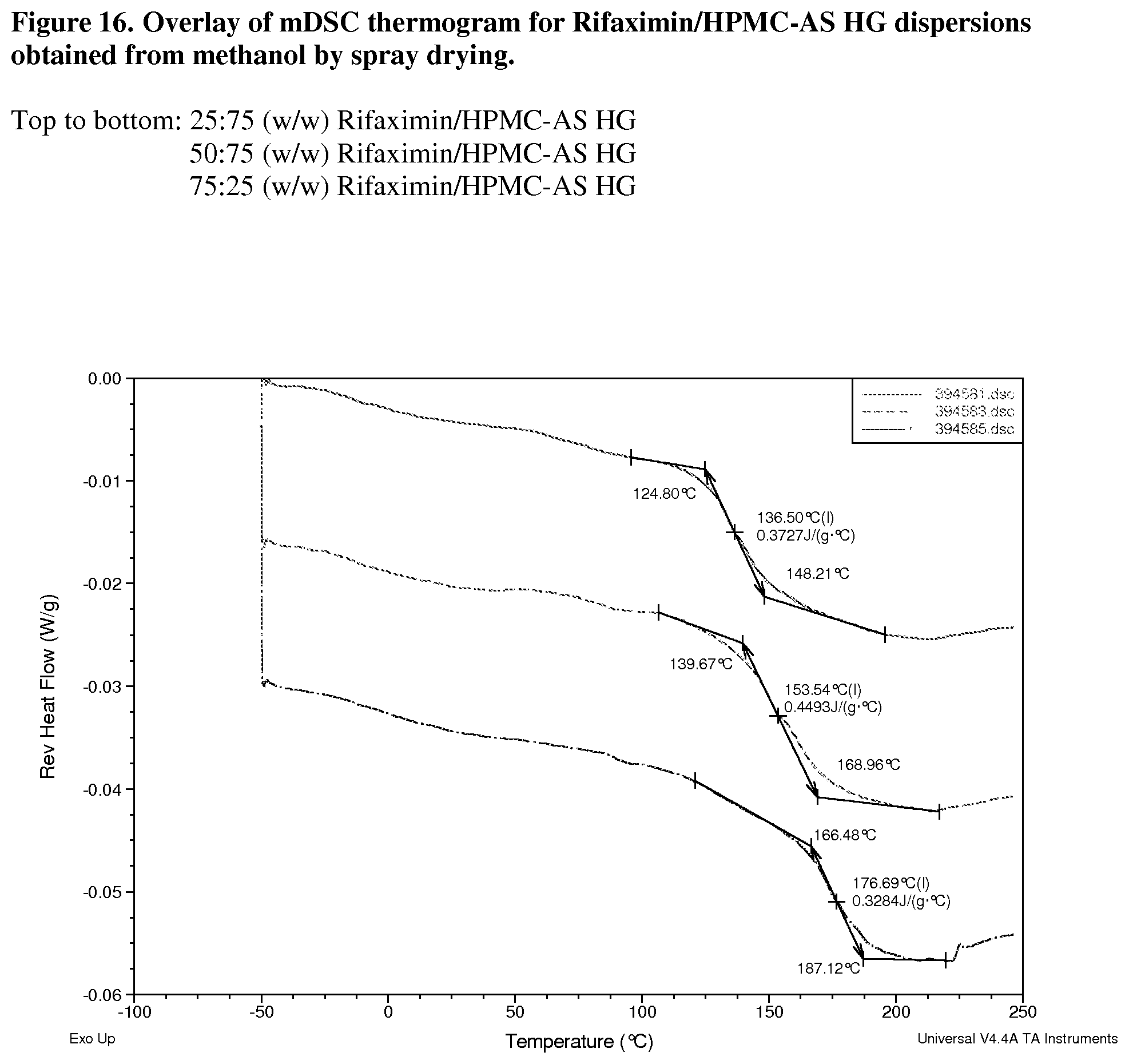
D00017

D00018

D00019

D00020

D00021

D00022
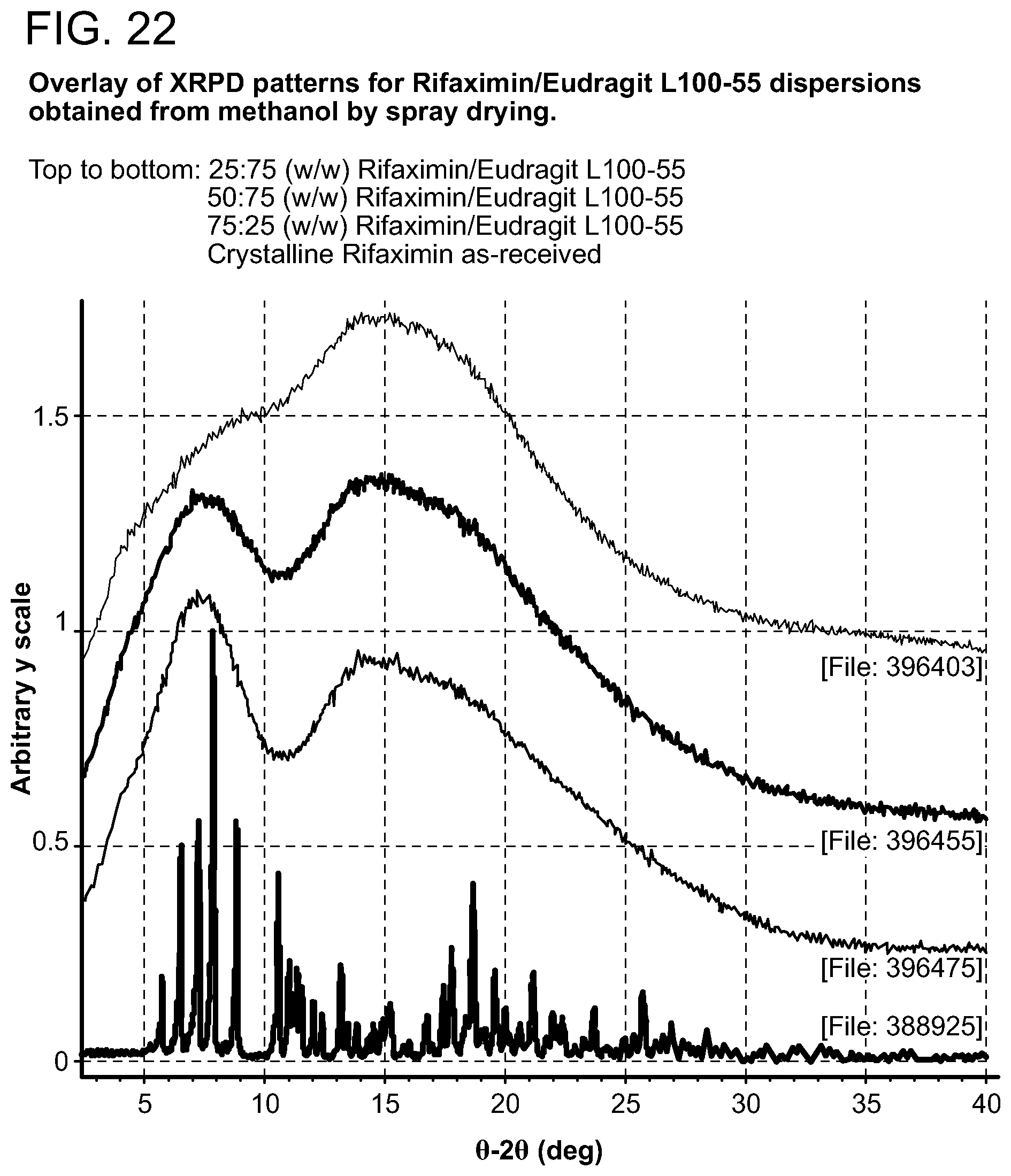
D00023
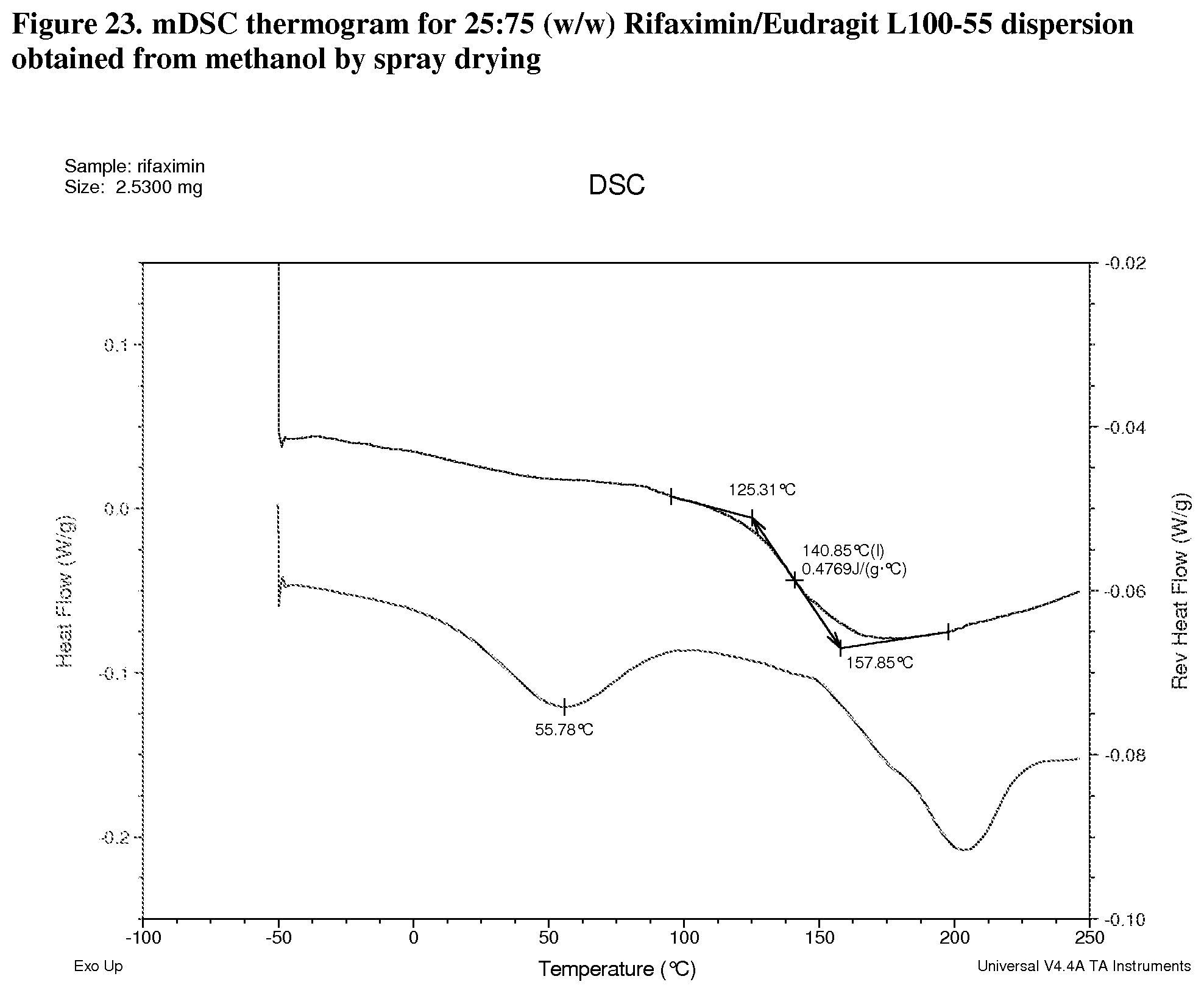
D00024
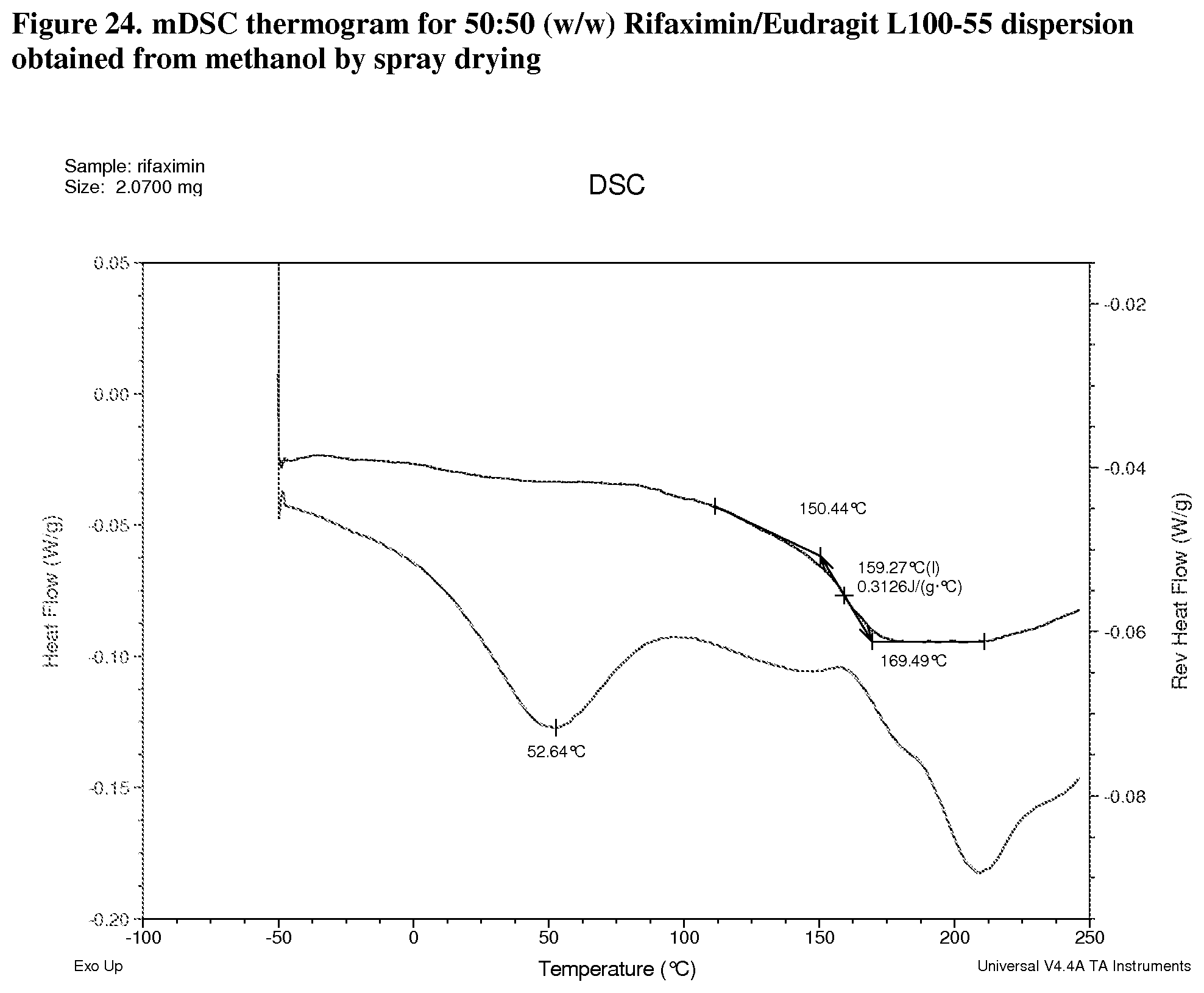
D00025
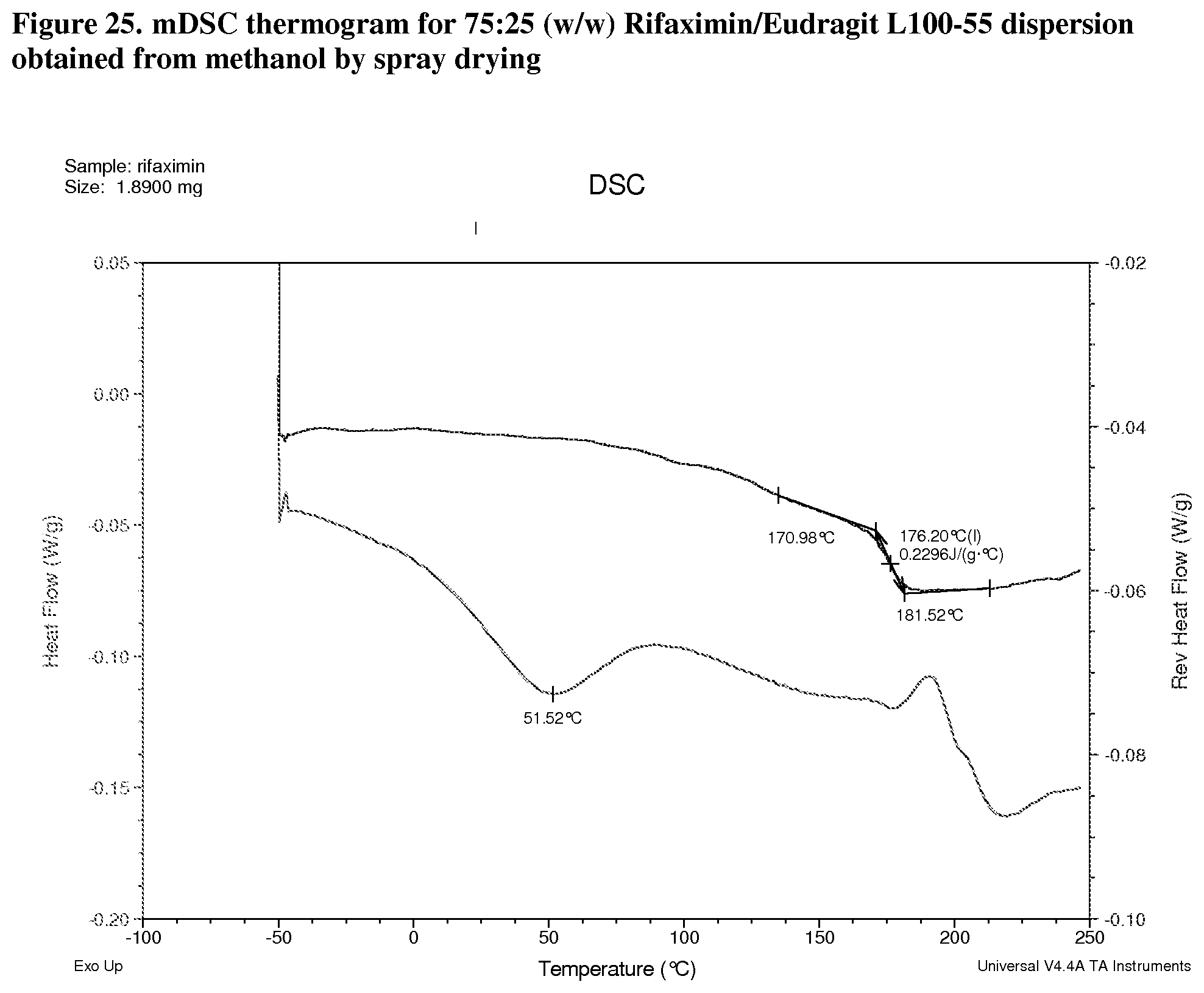
D00026

D00027
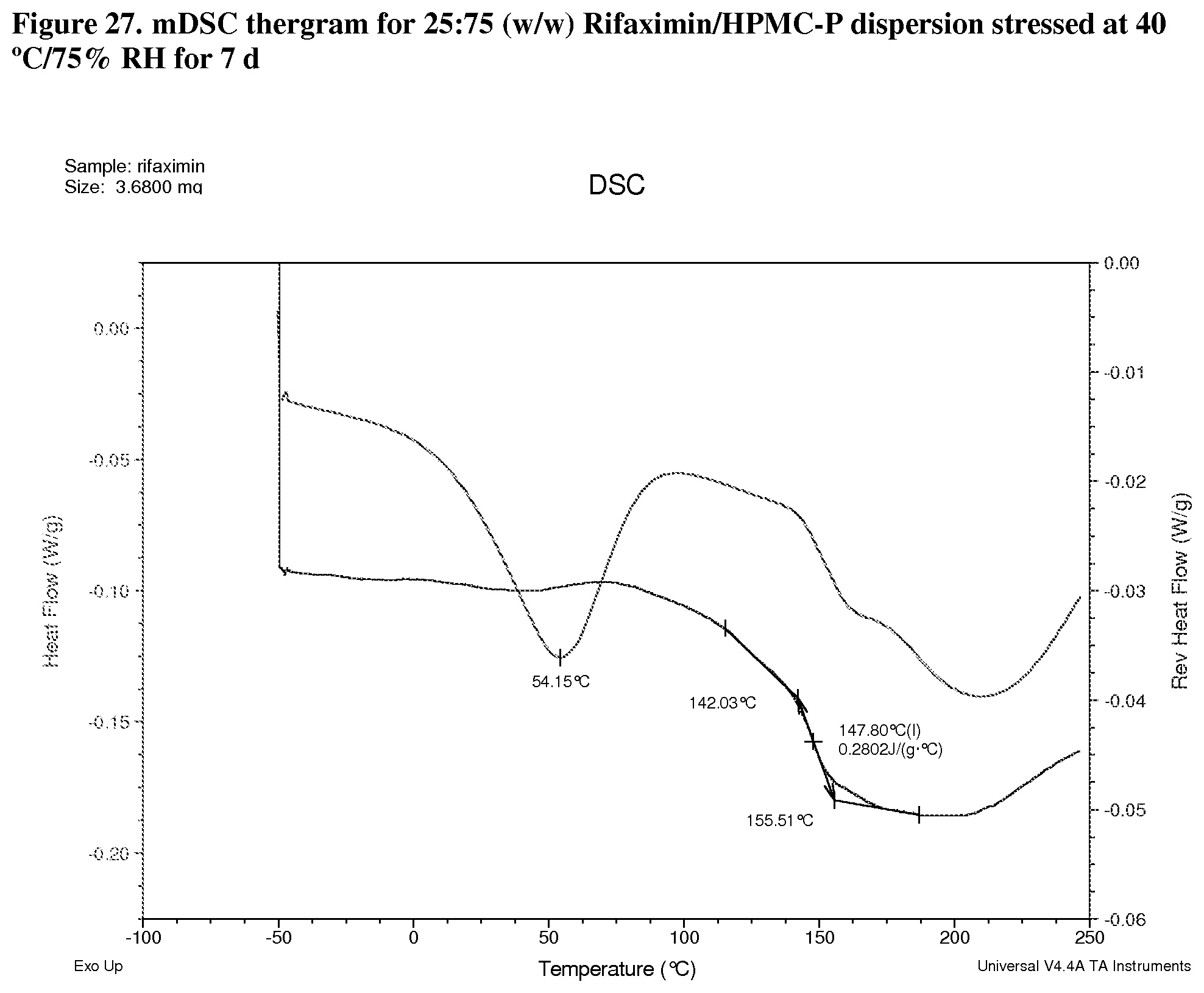
D00028
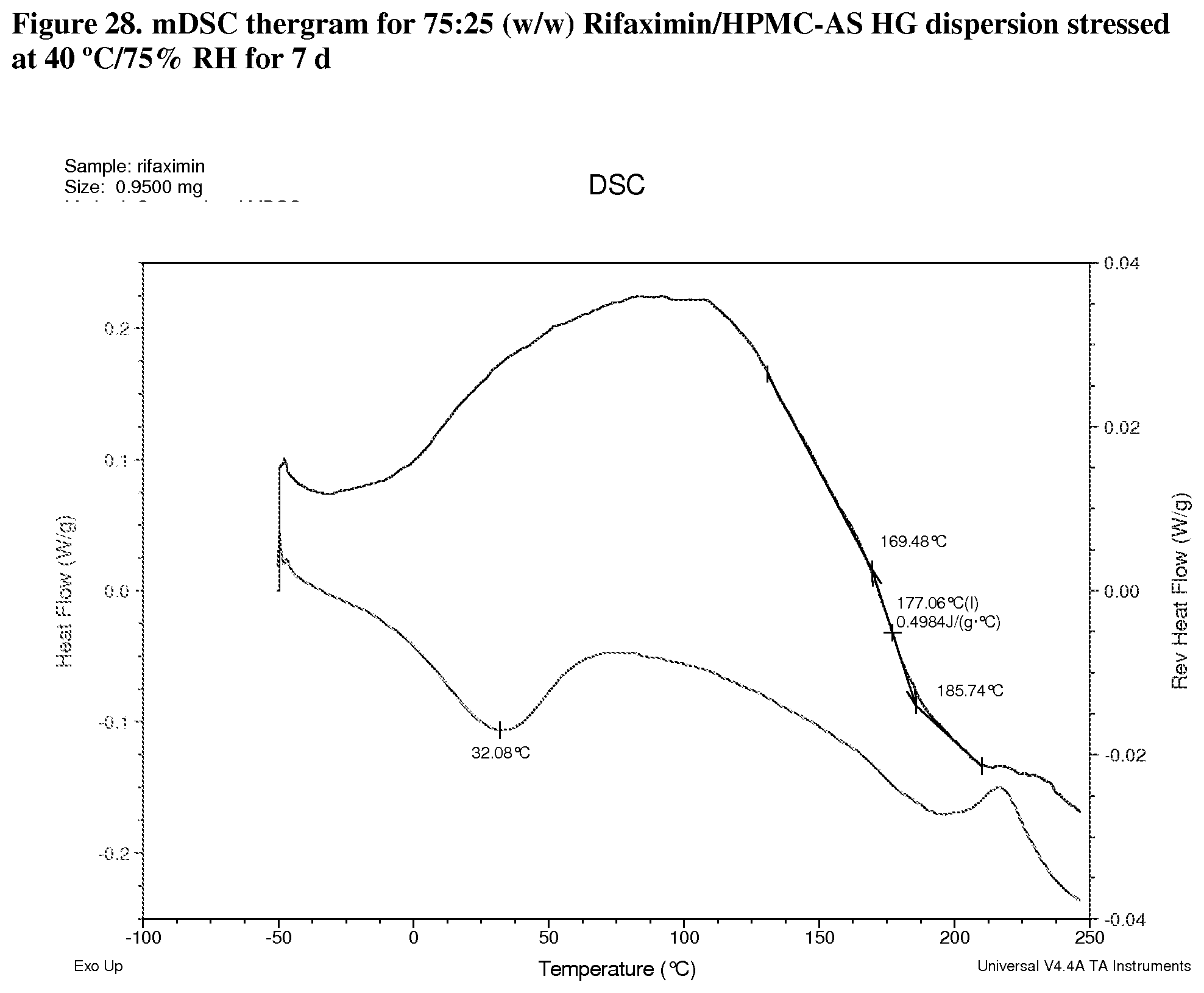
D00029
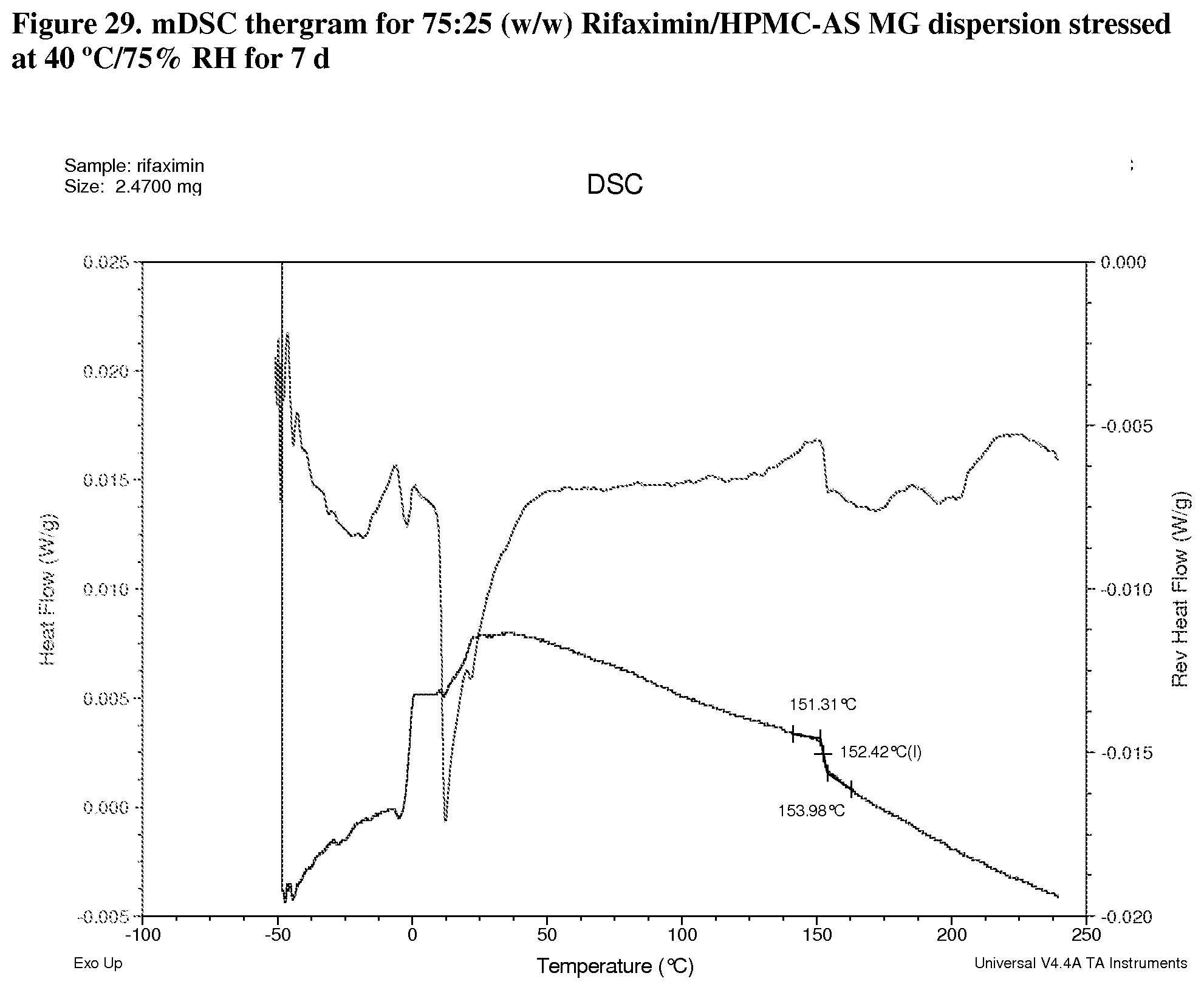
D00030
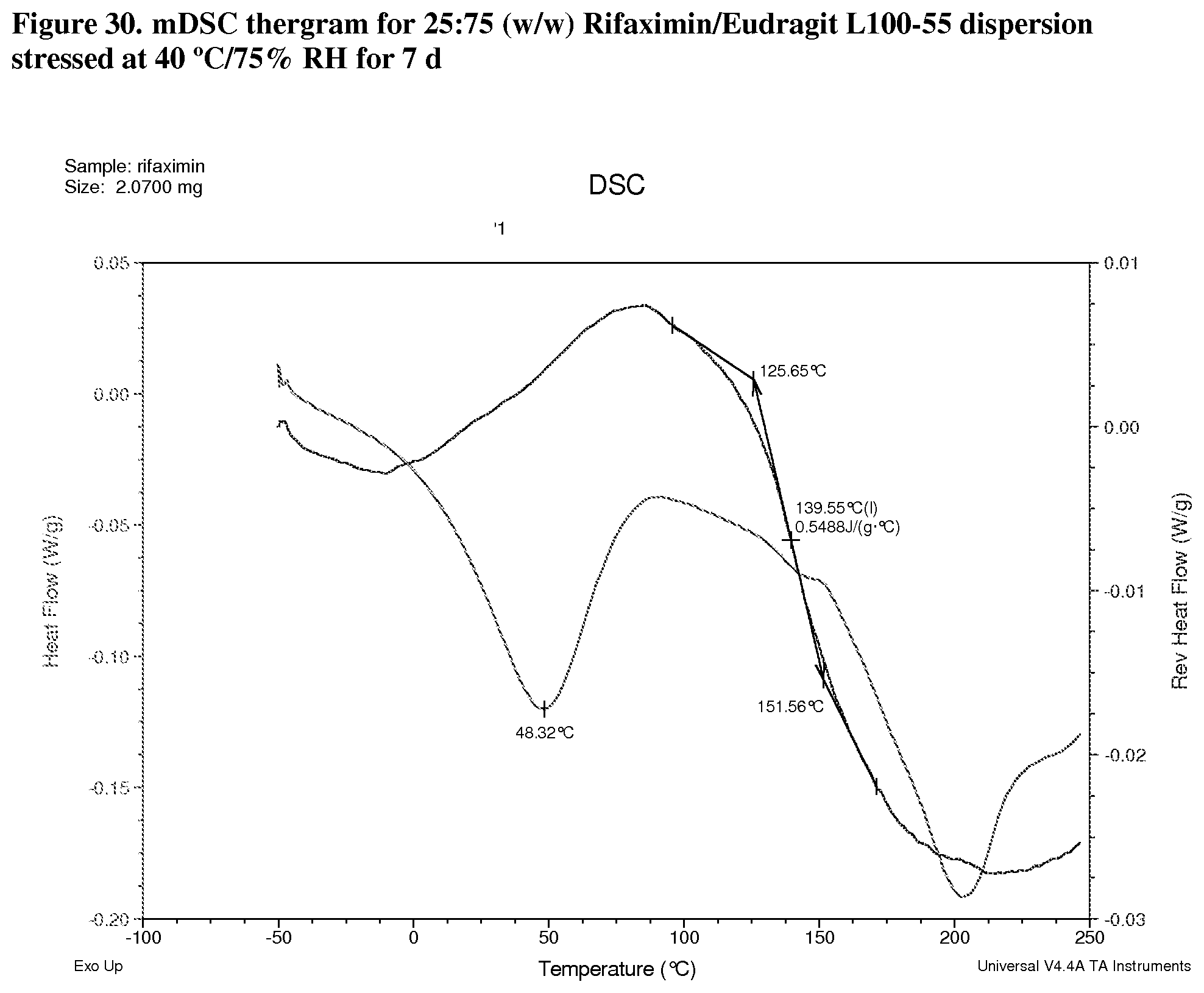
D00031
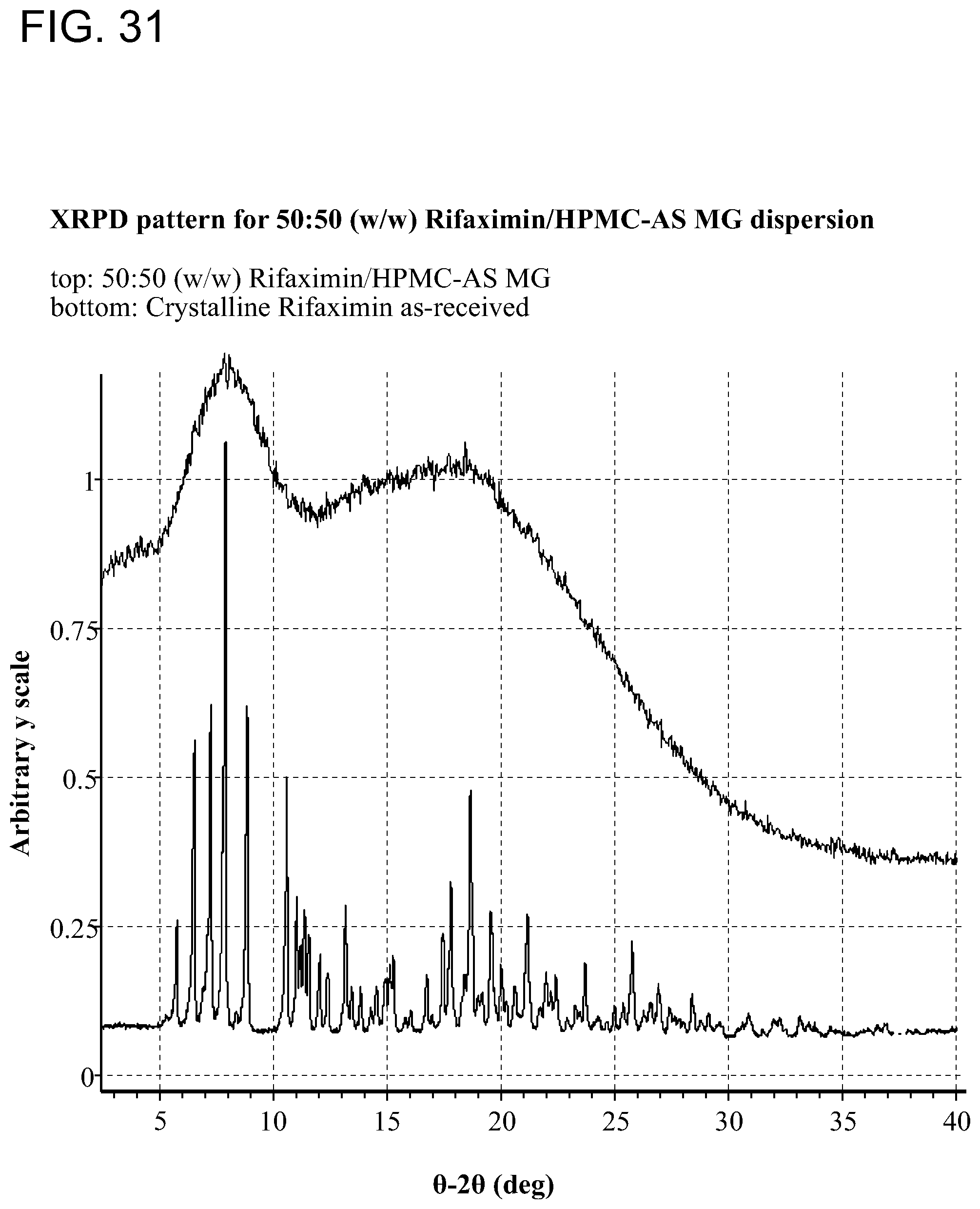
D00032

D00033
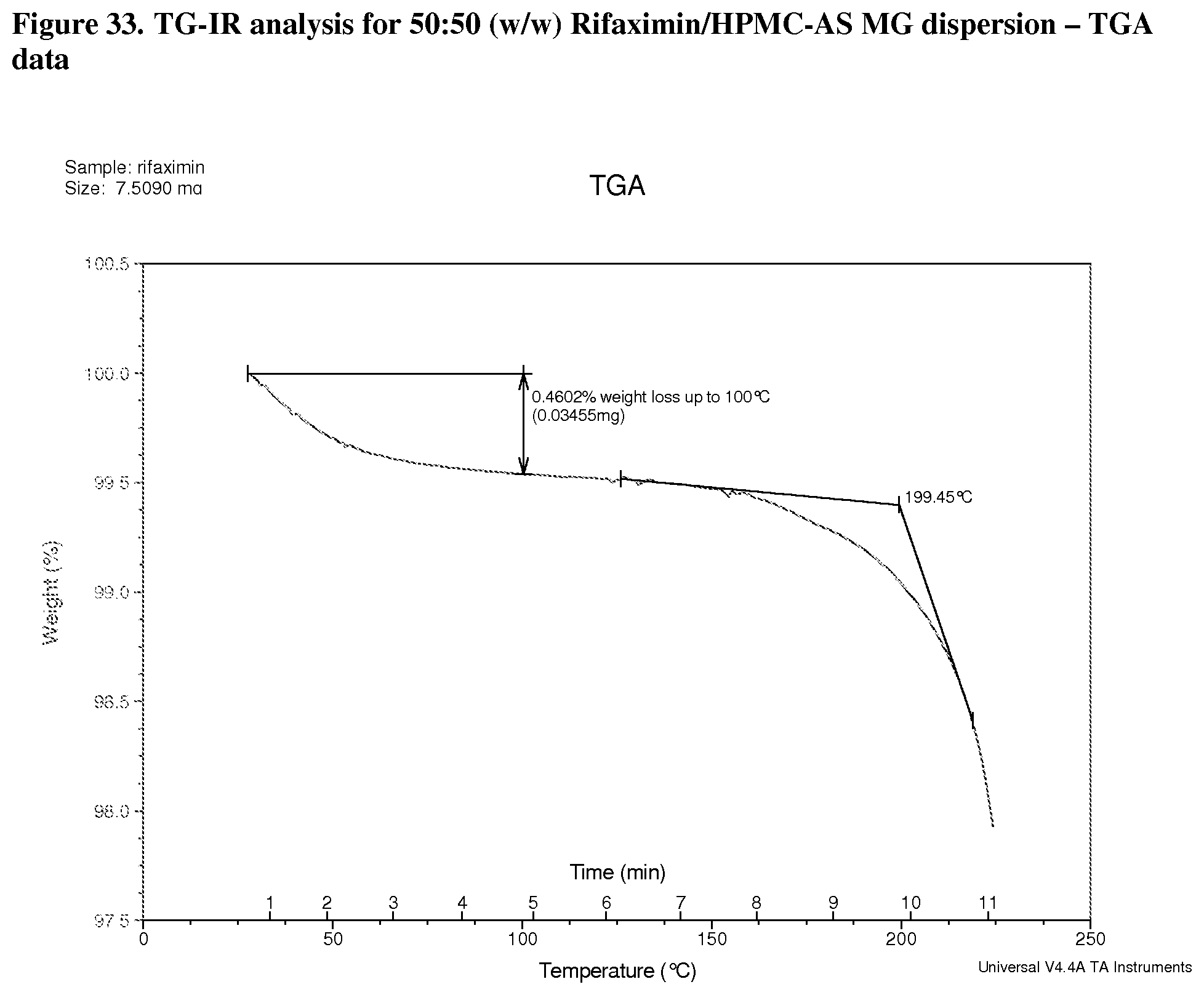
D00034
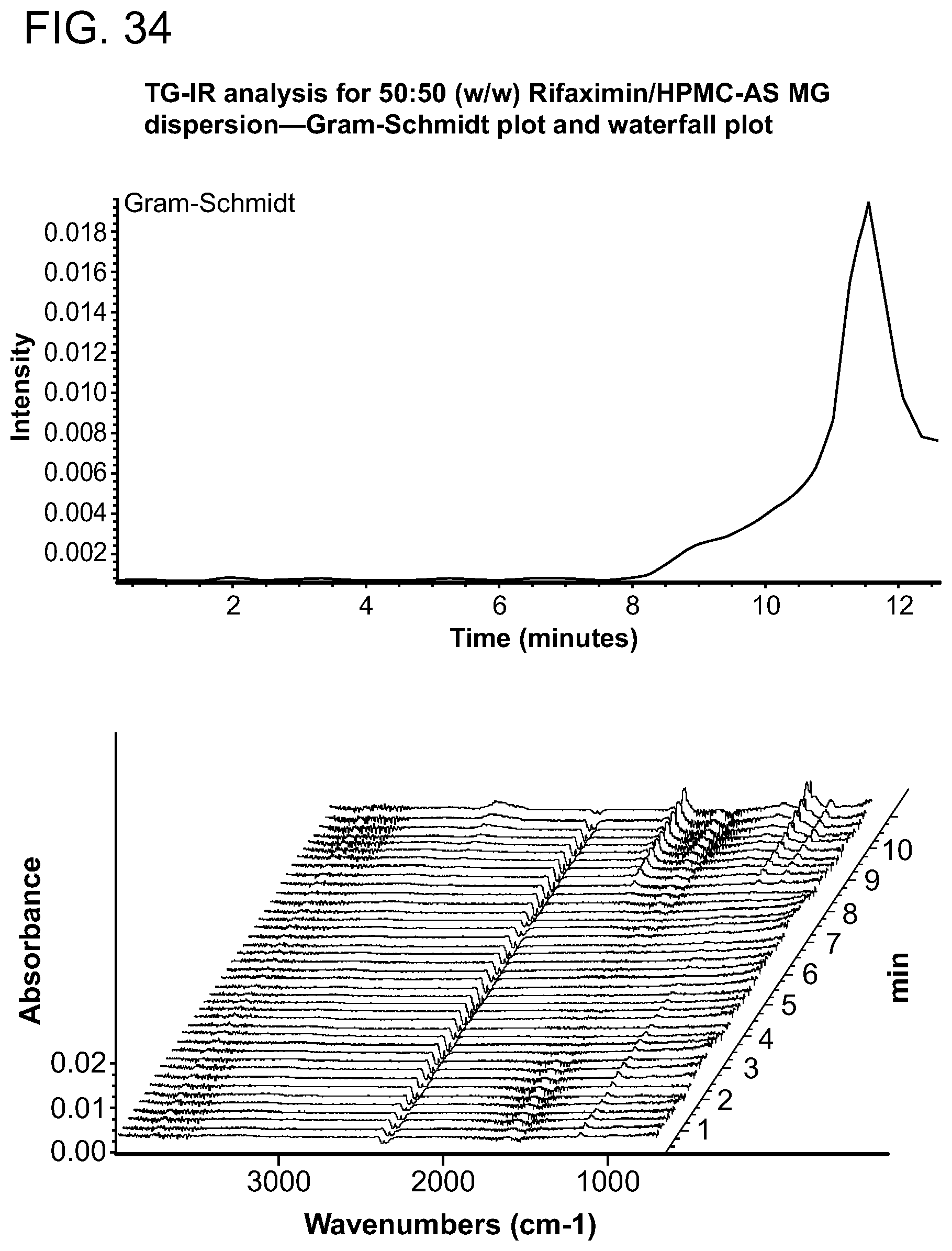
D00035
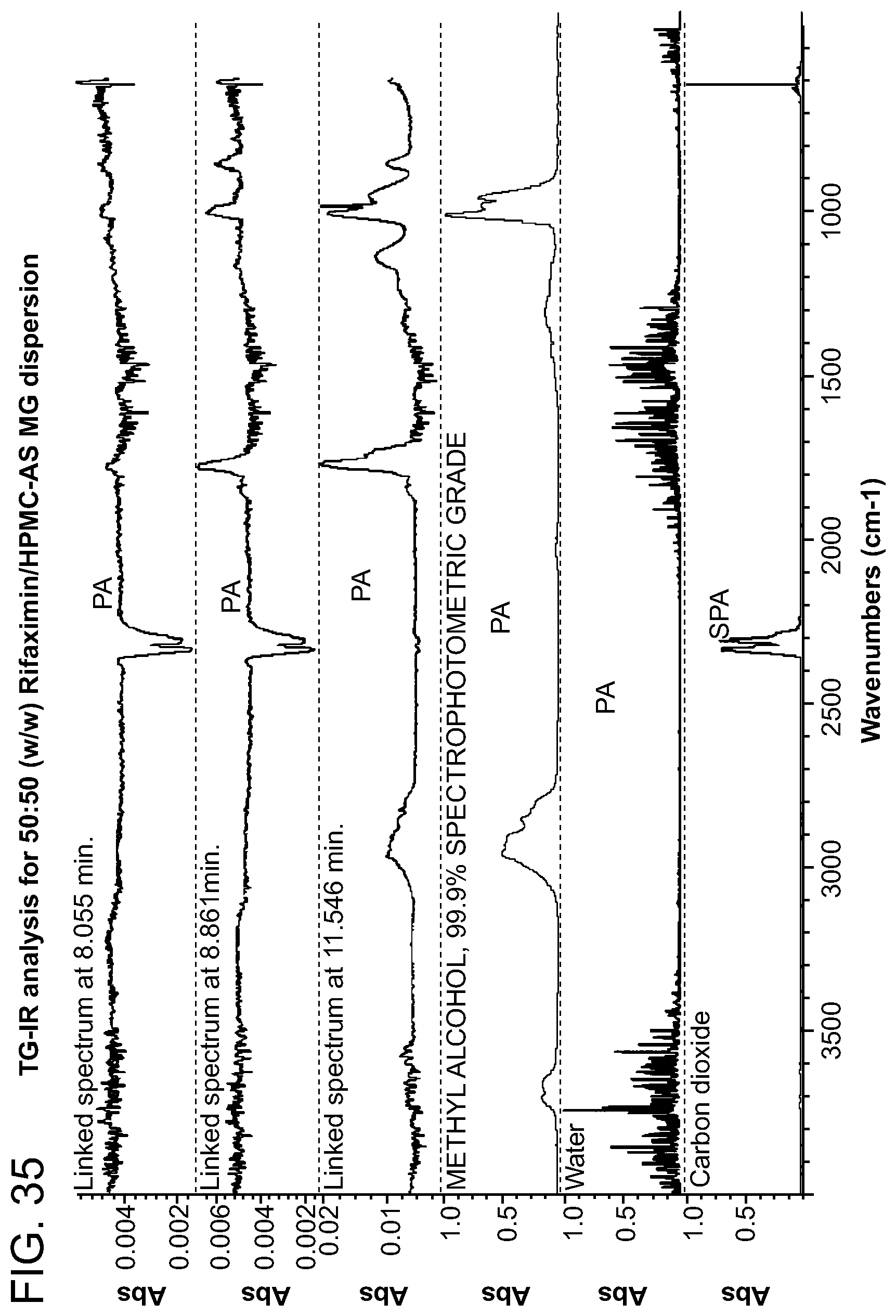
D00036

D00037
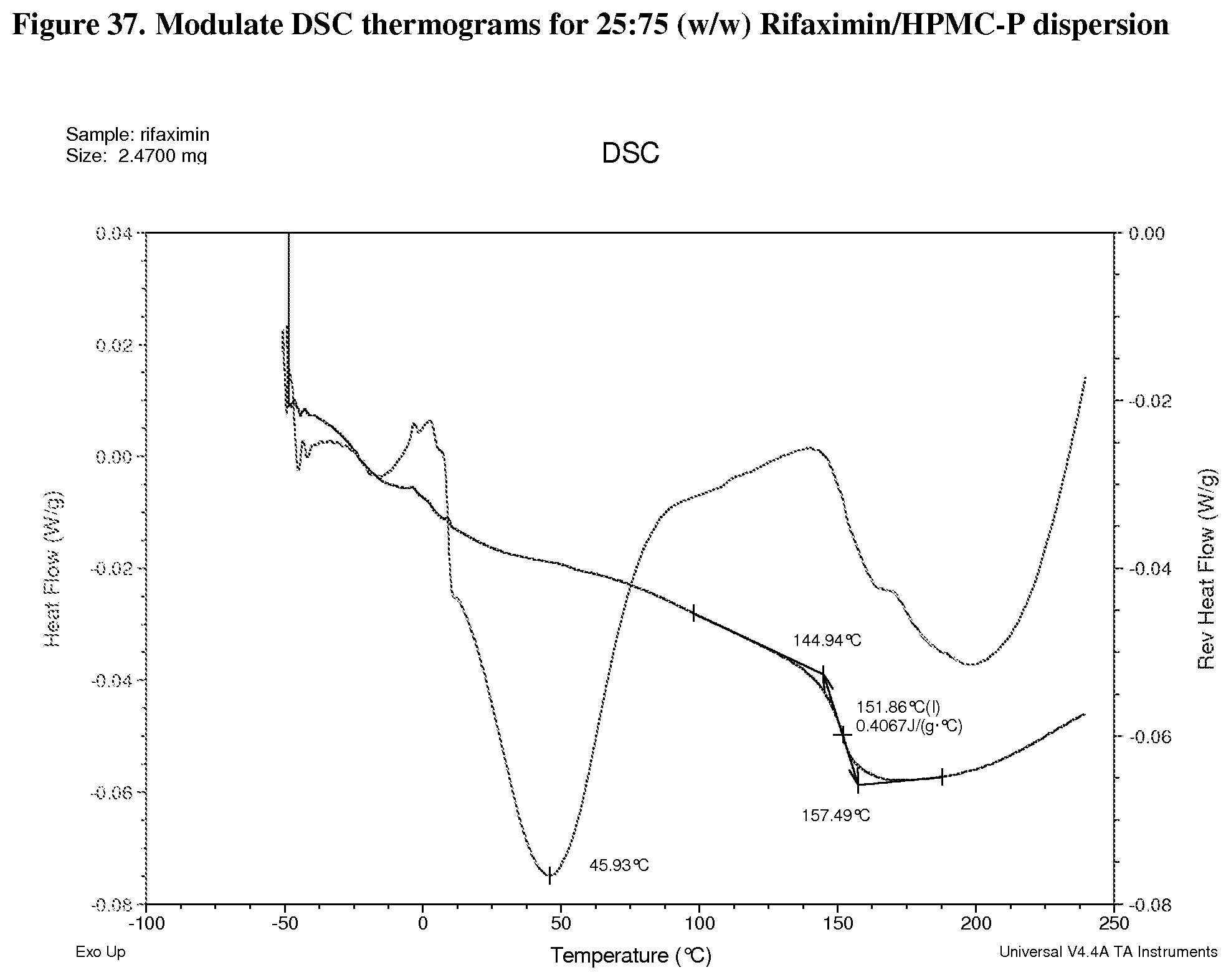
D00038

D00039

D00040
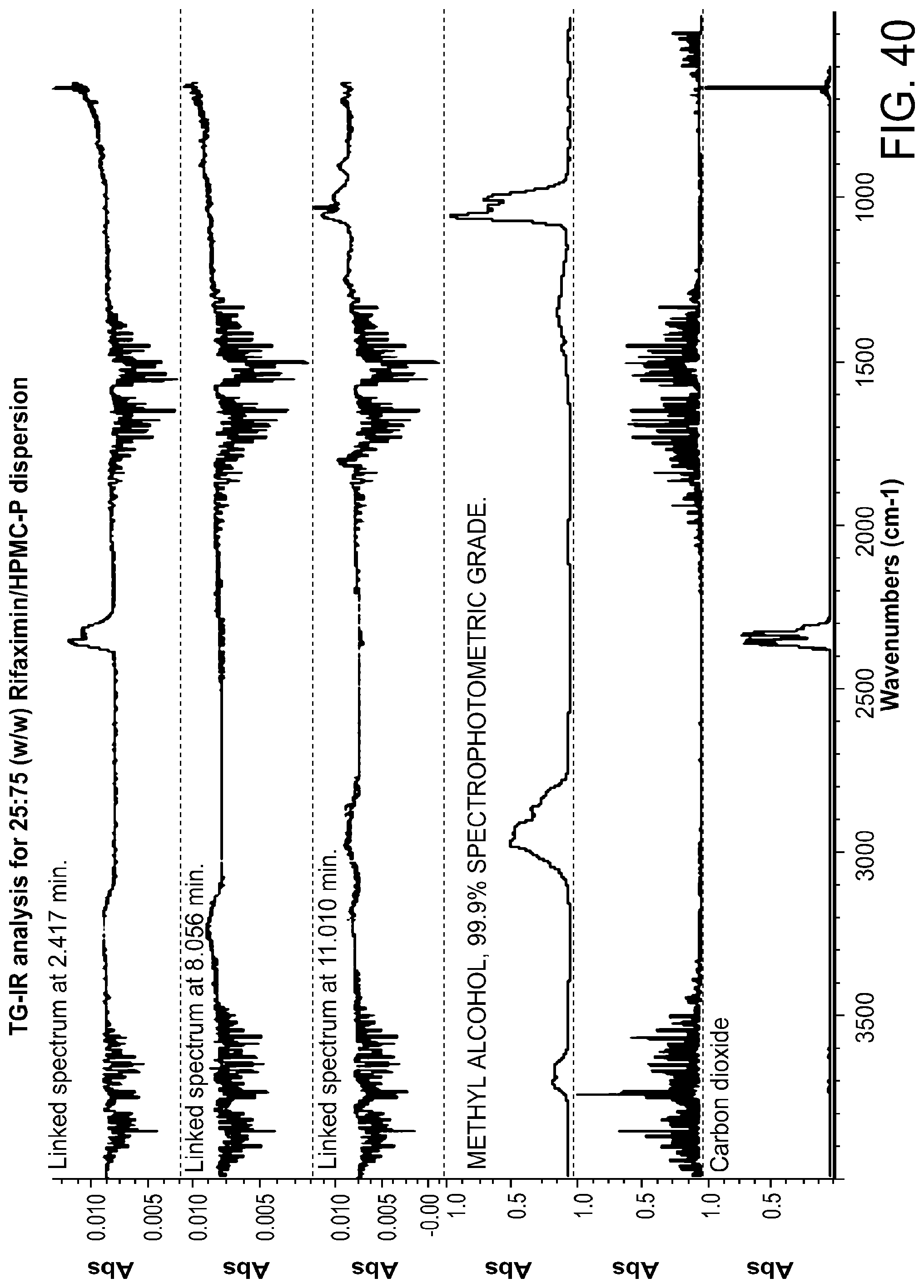
D00041

D00042
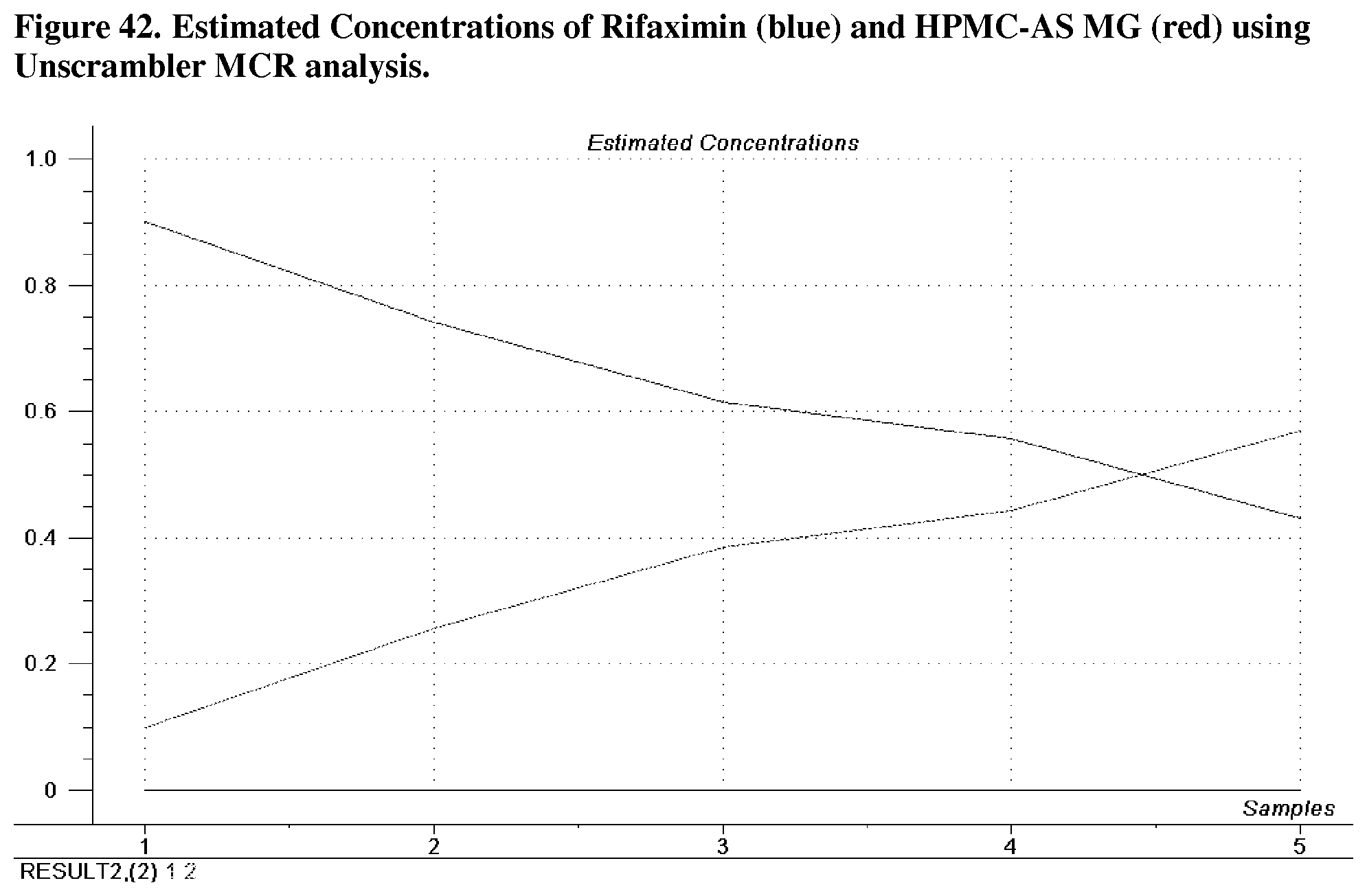
D00043
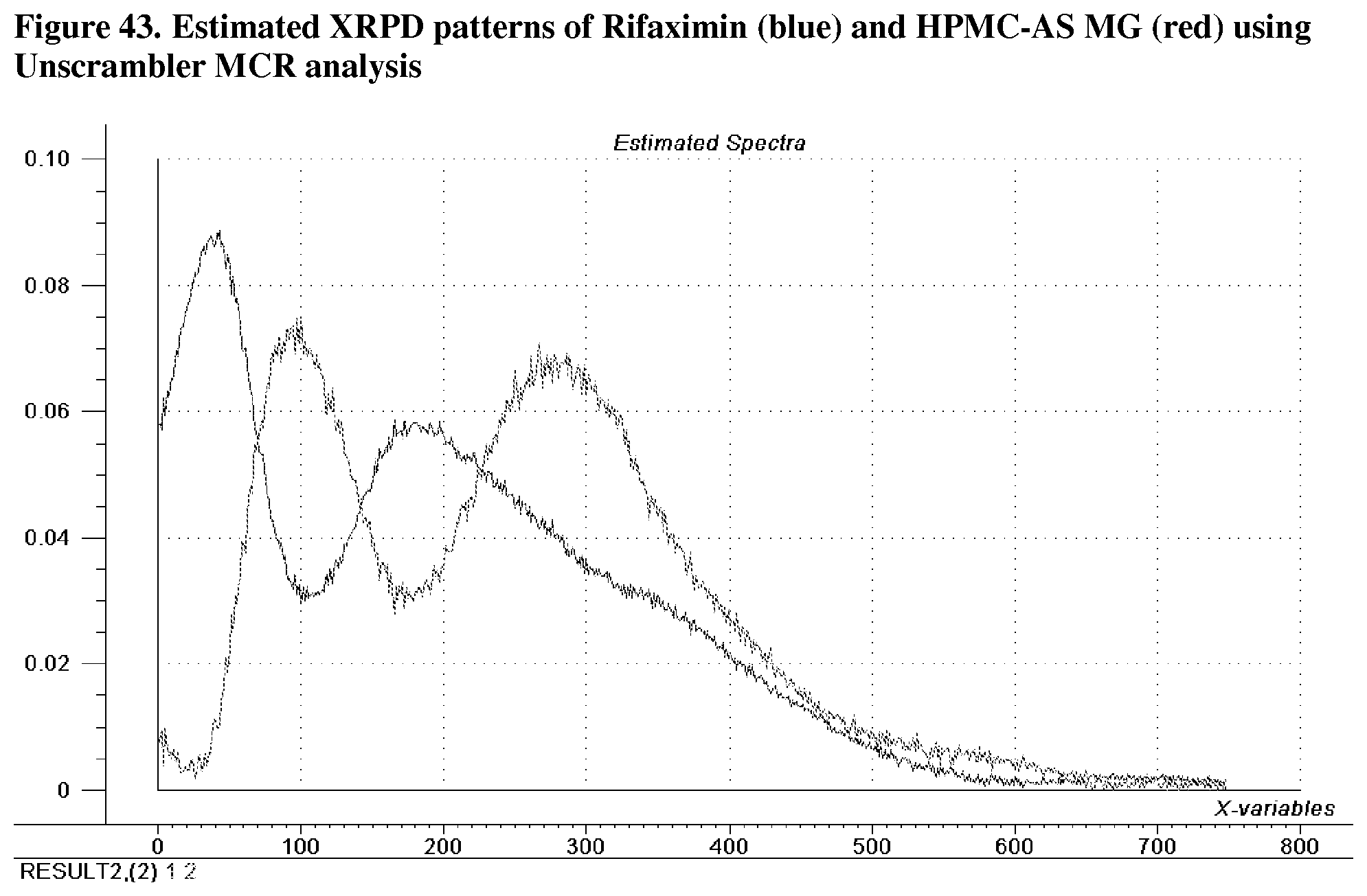
D00044
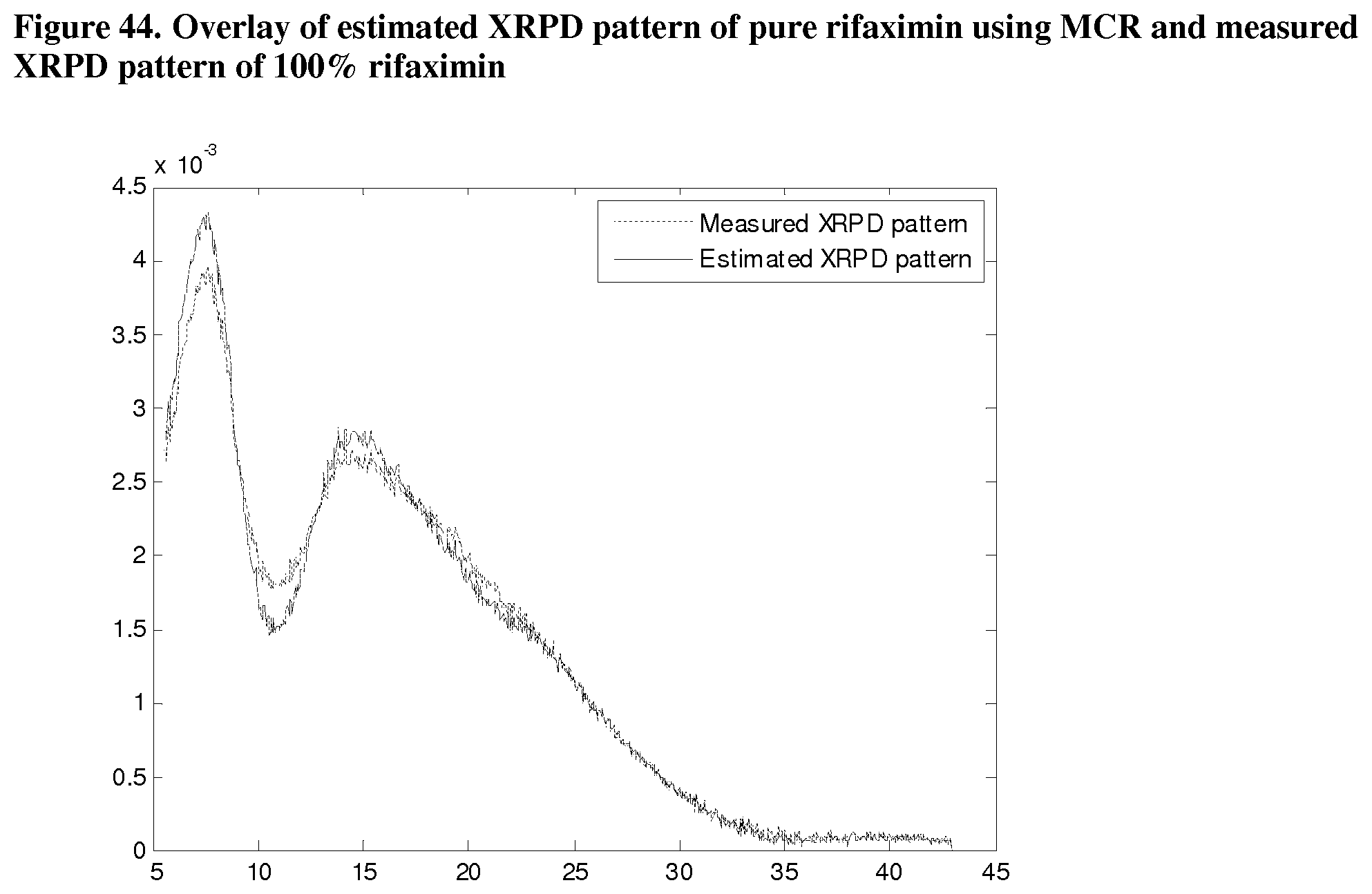
D00045
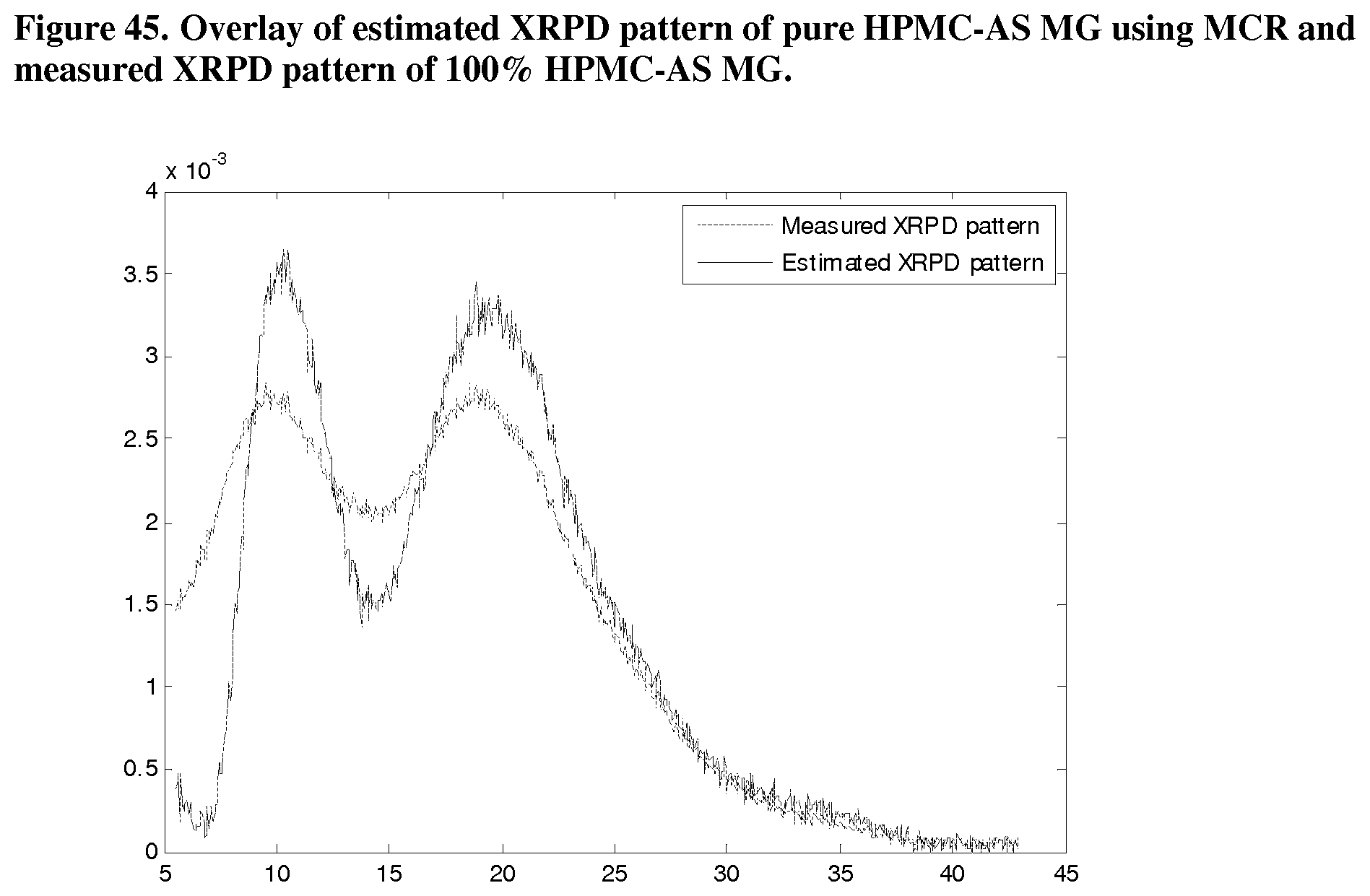
D00046
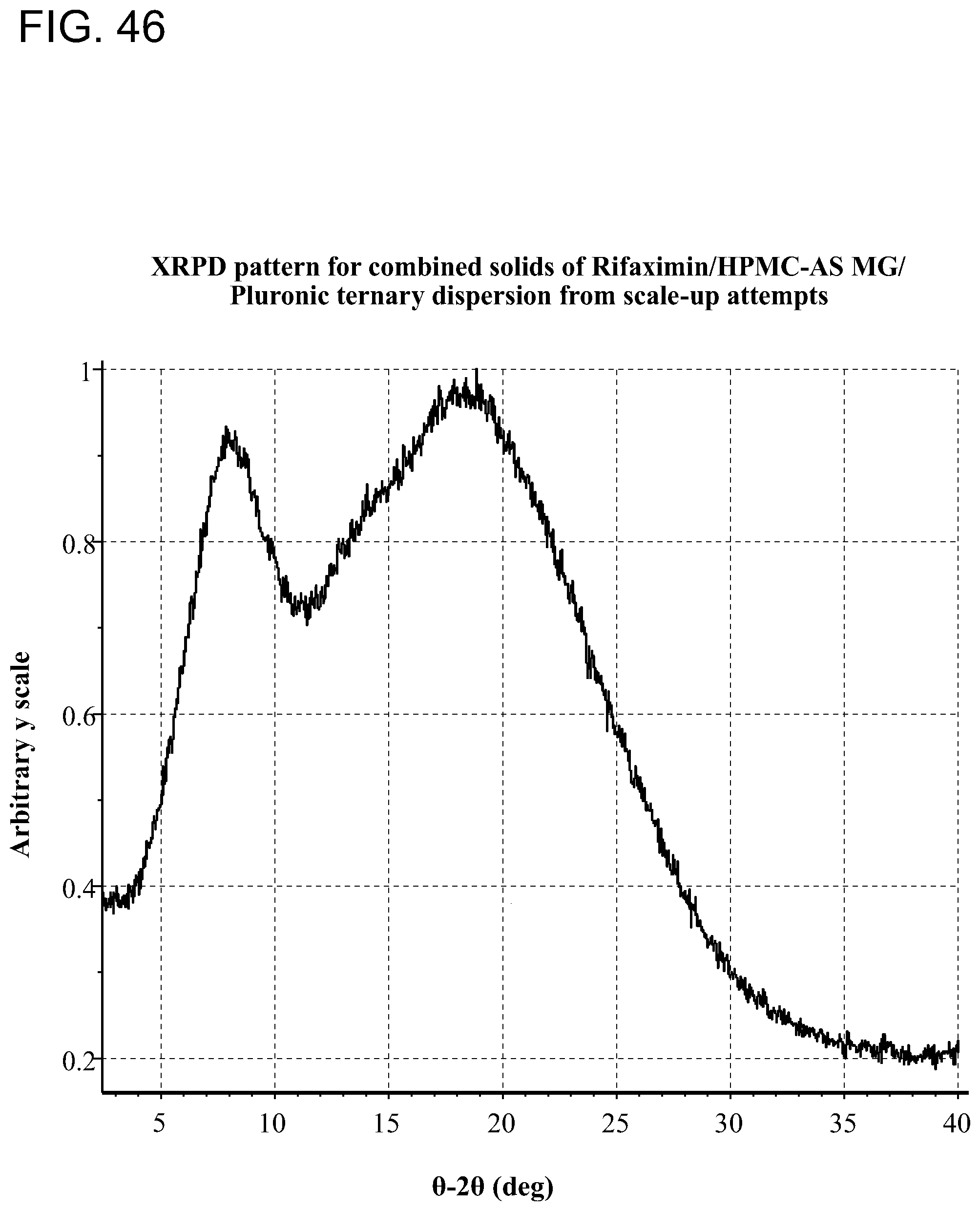
D00047

D00048
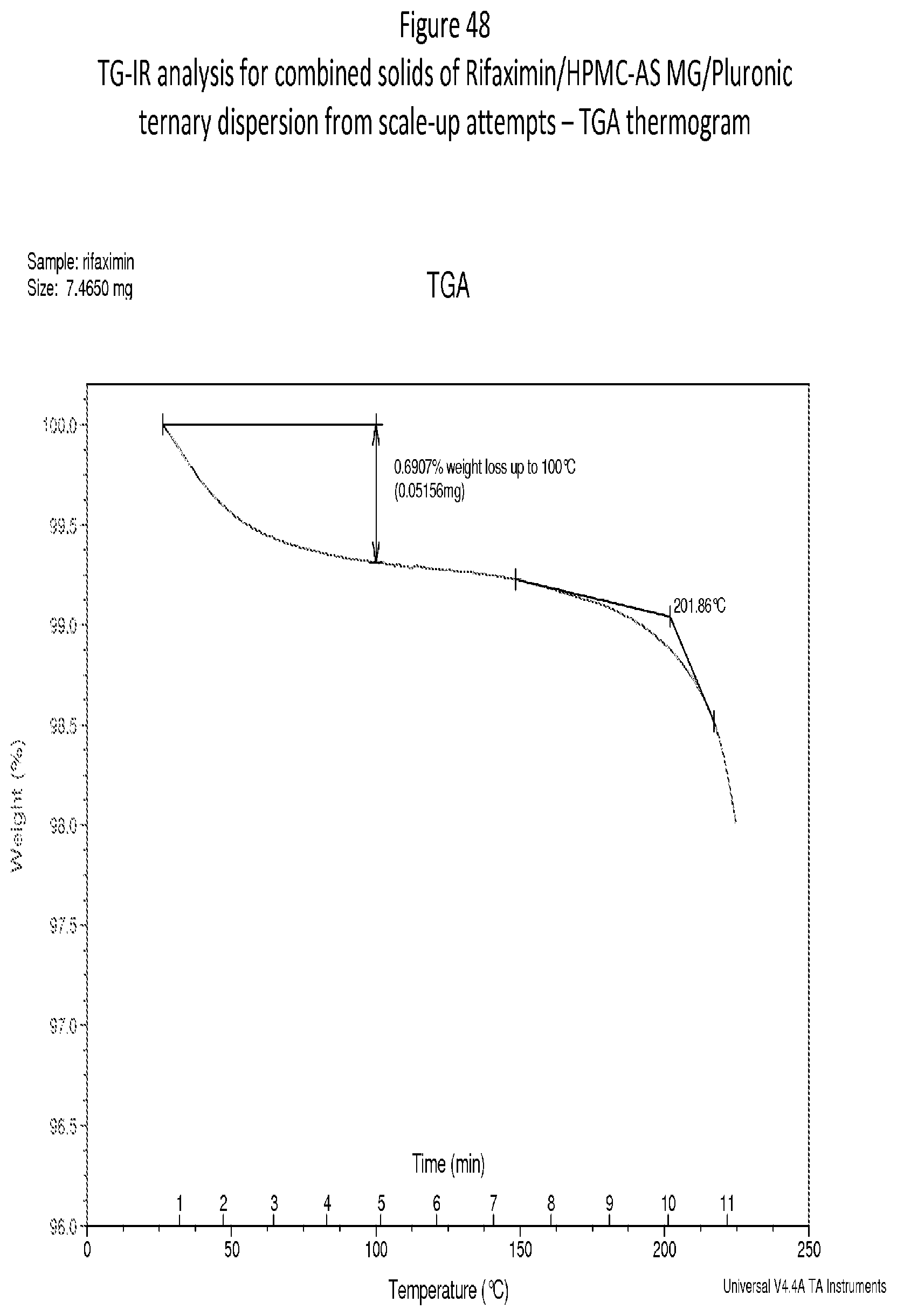
D00049
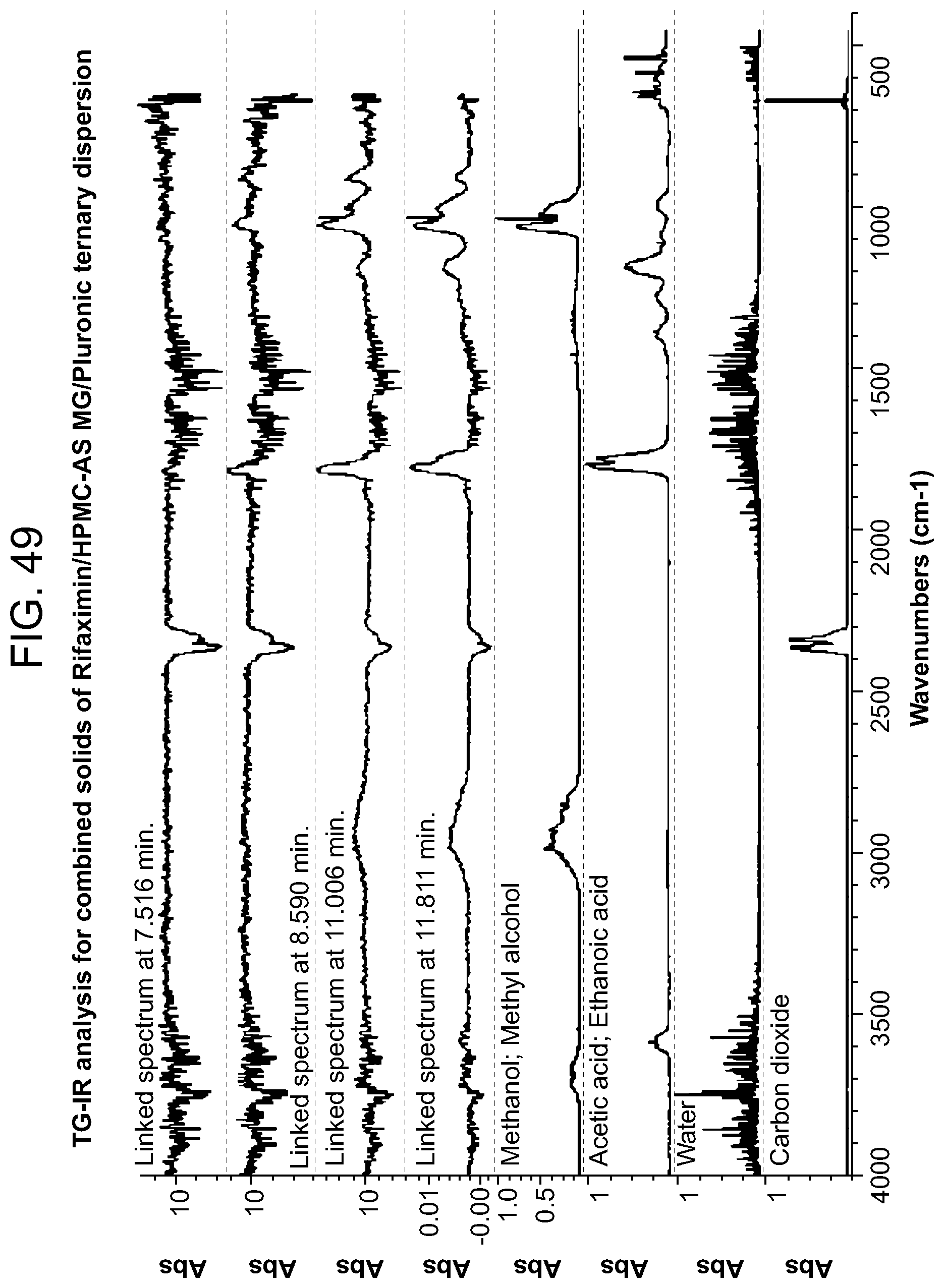
D00050
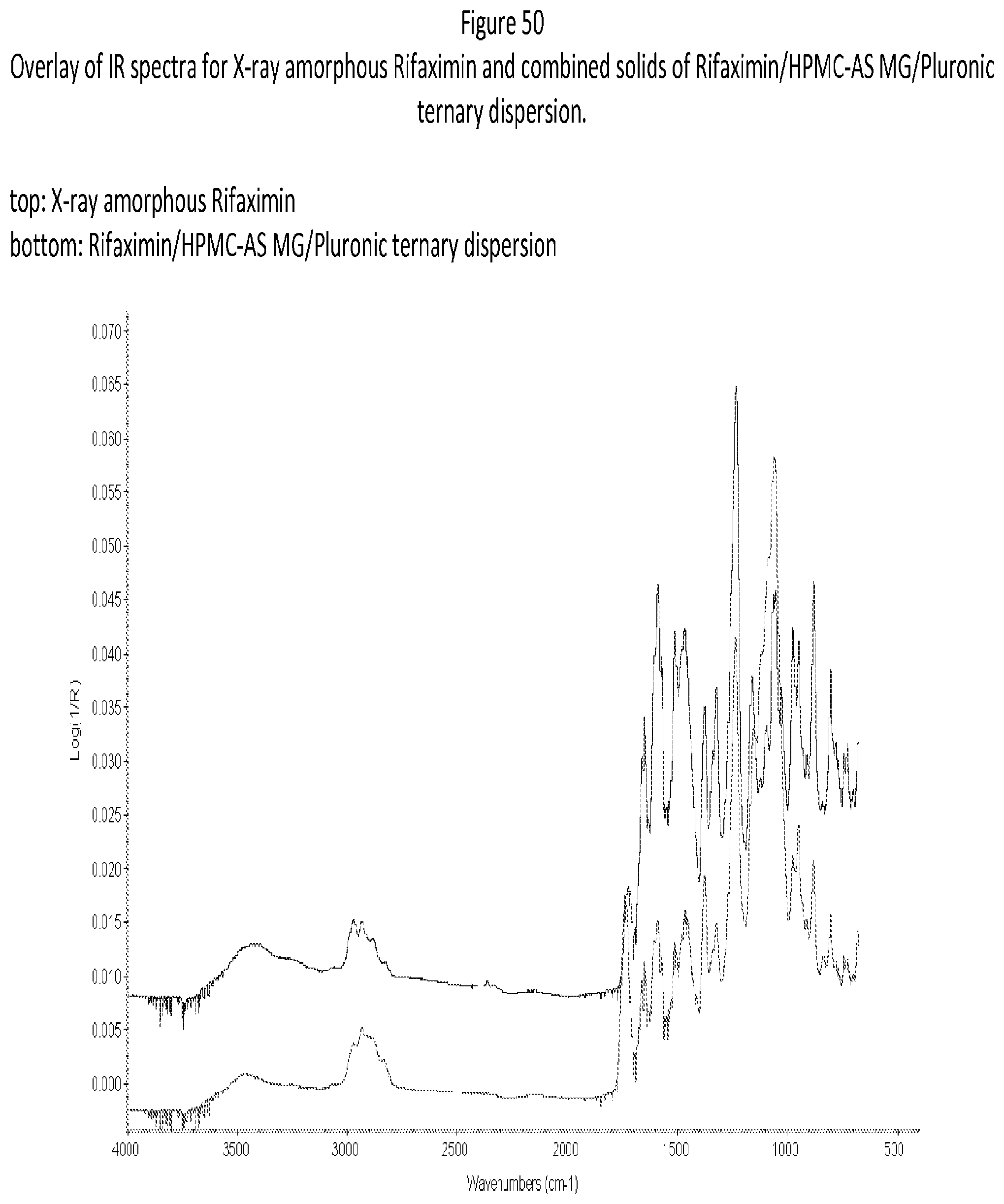
D00051
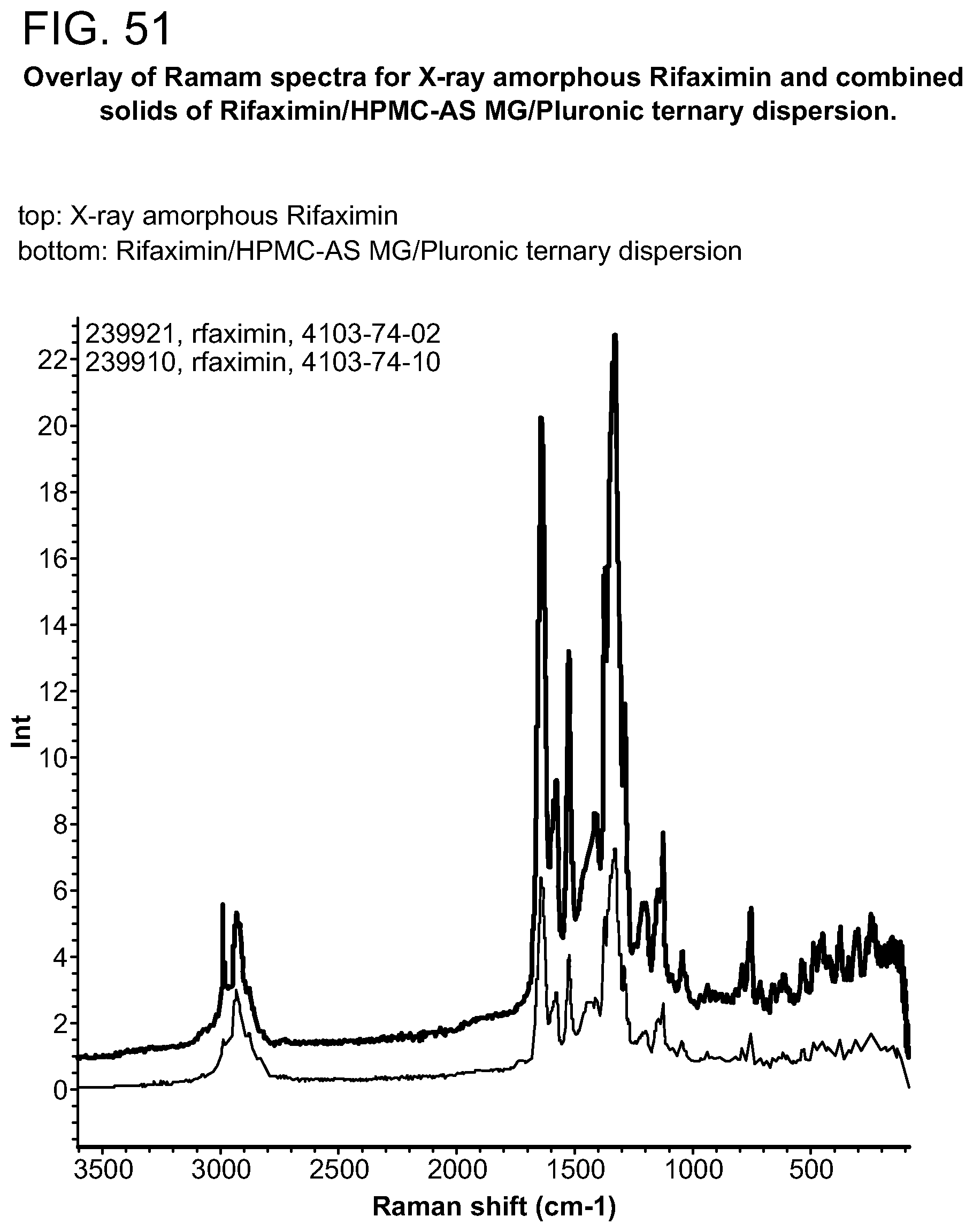
D00052
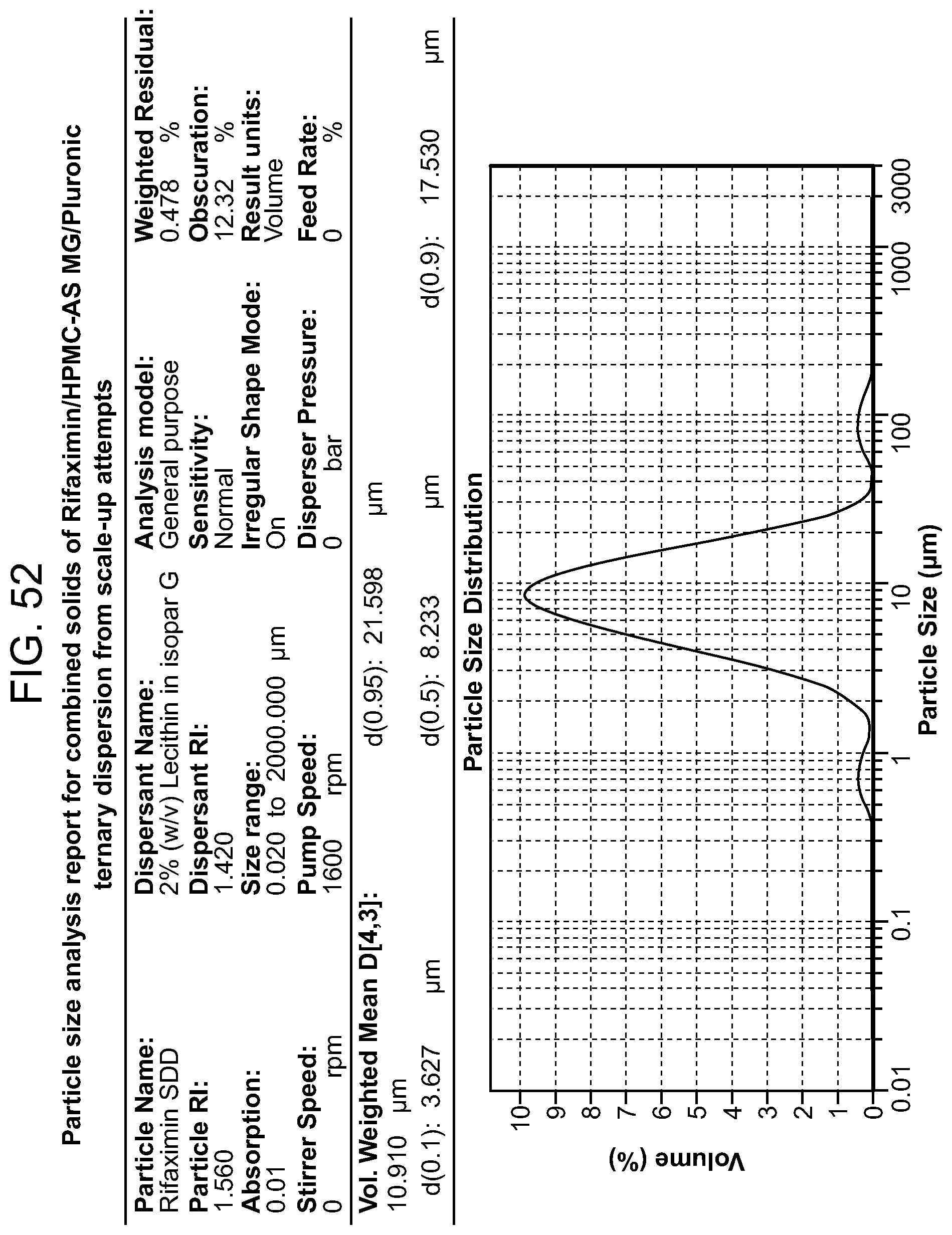
D00053

D00054

D00055
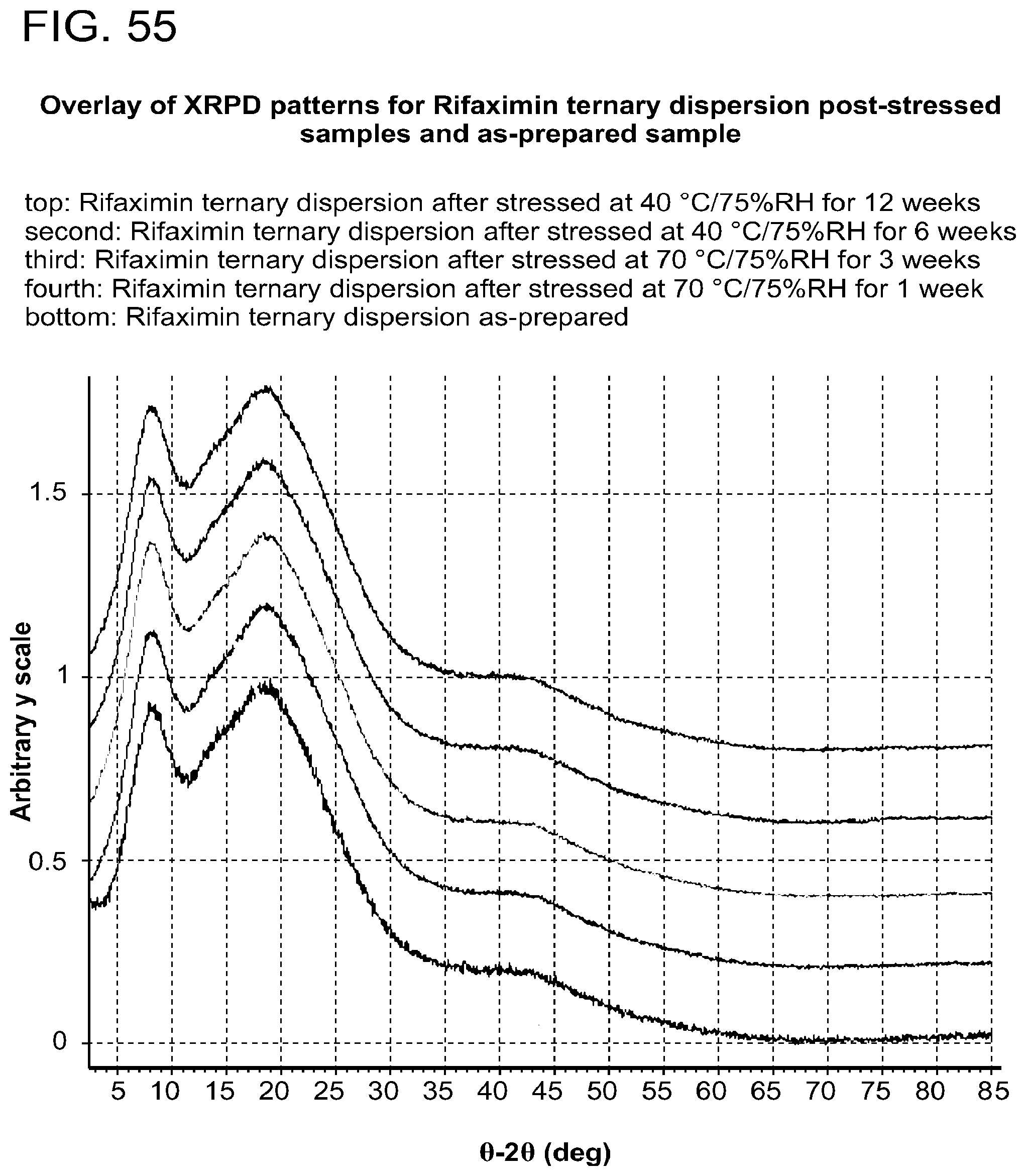
D00056
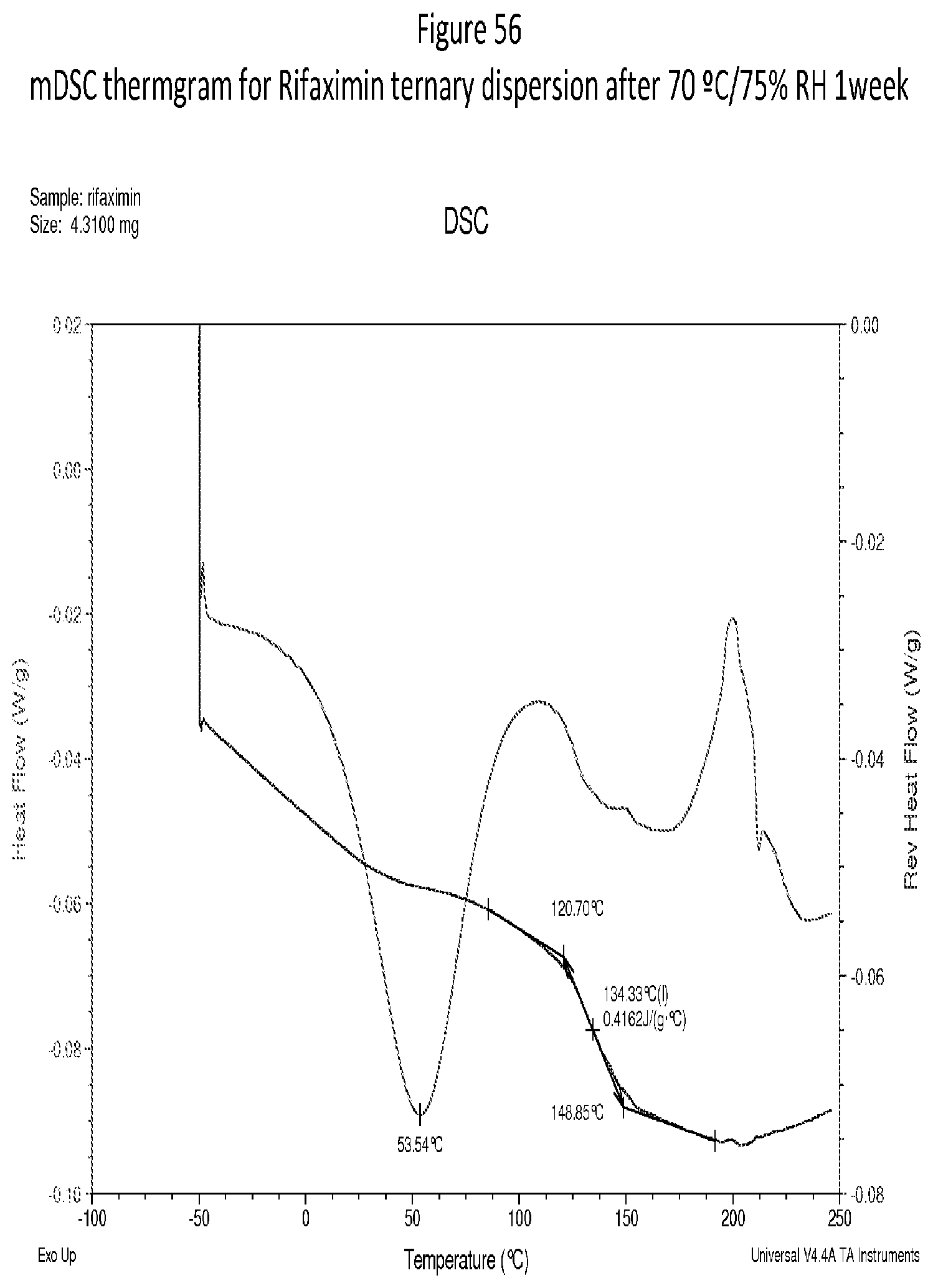
D00057
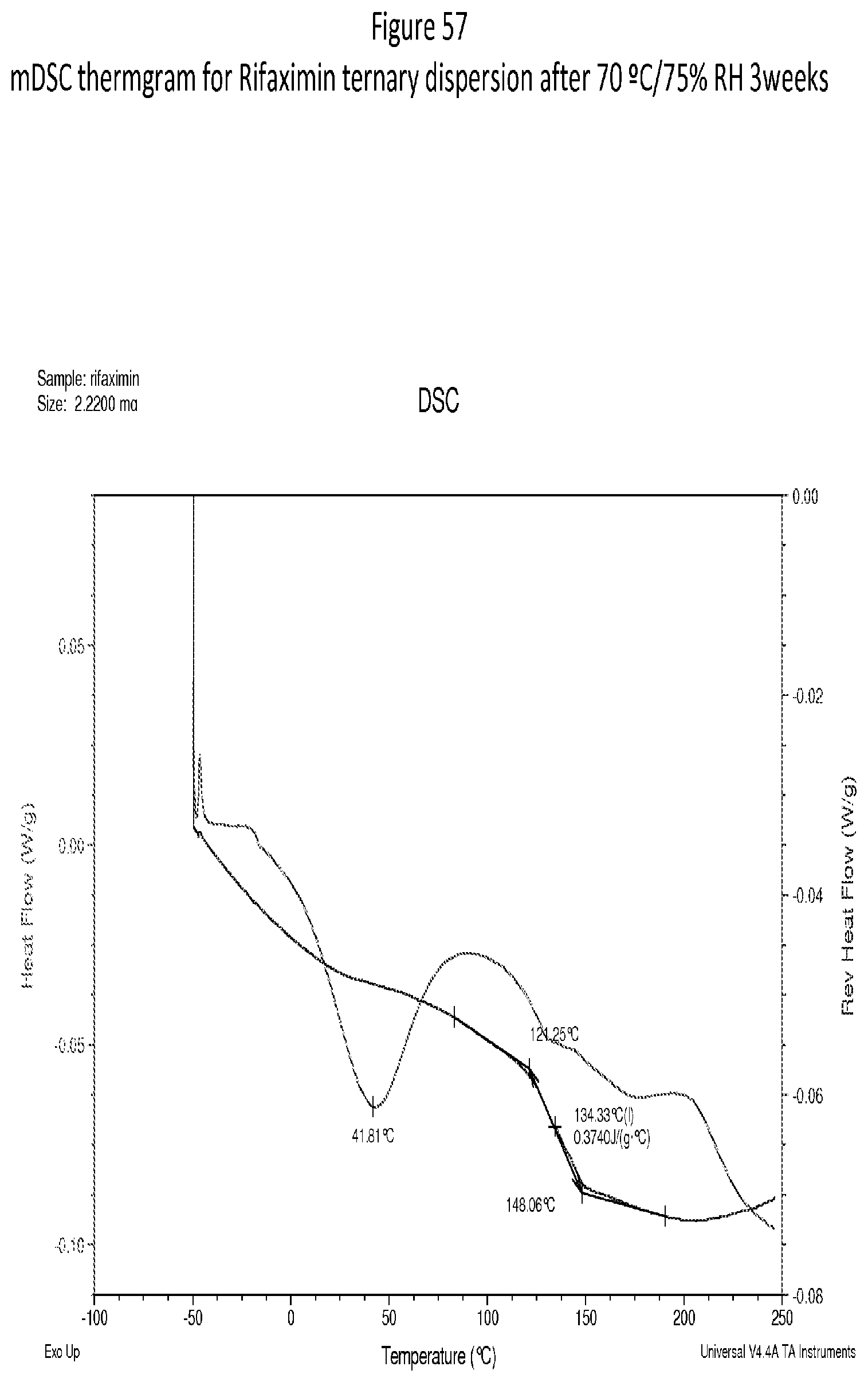
D00058

D00059

D00060

D00061
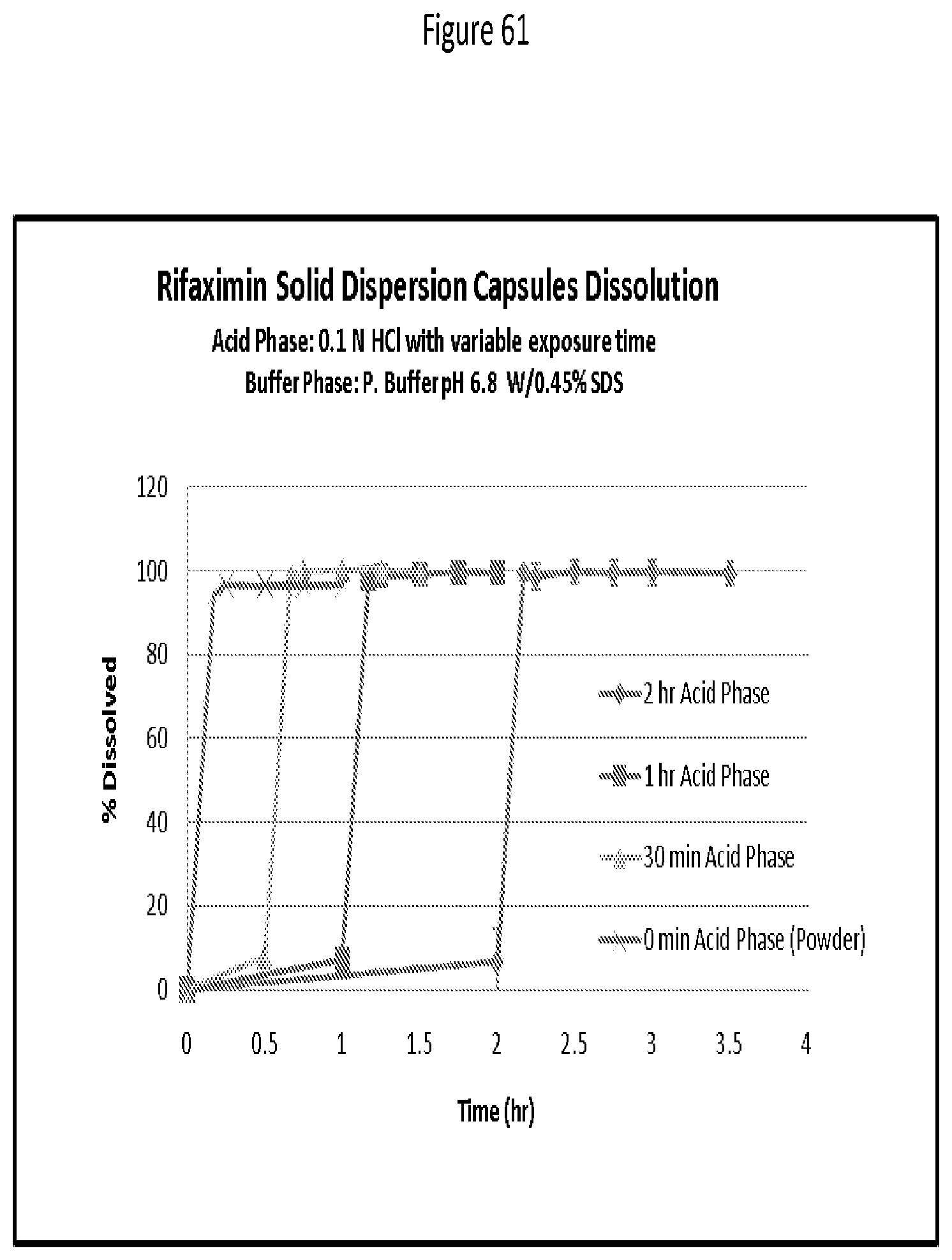
D00062
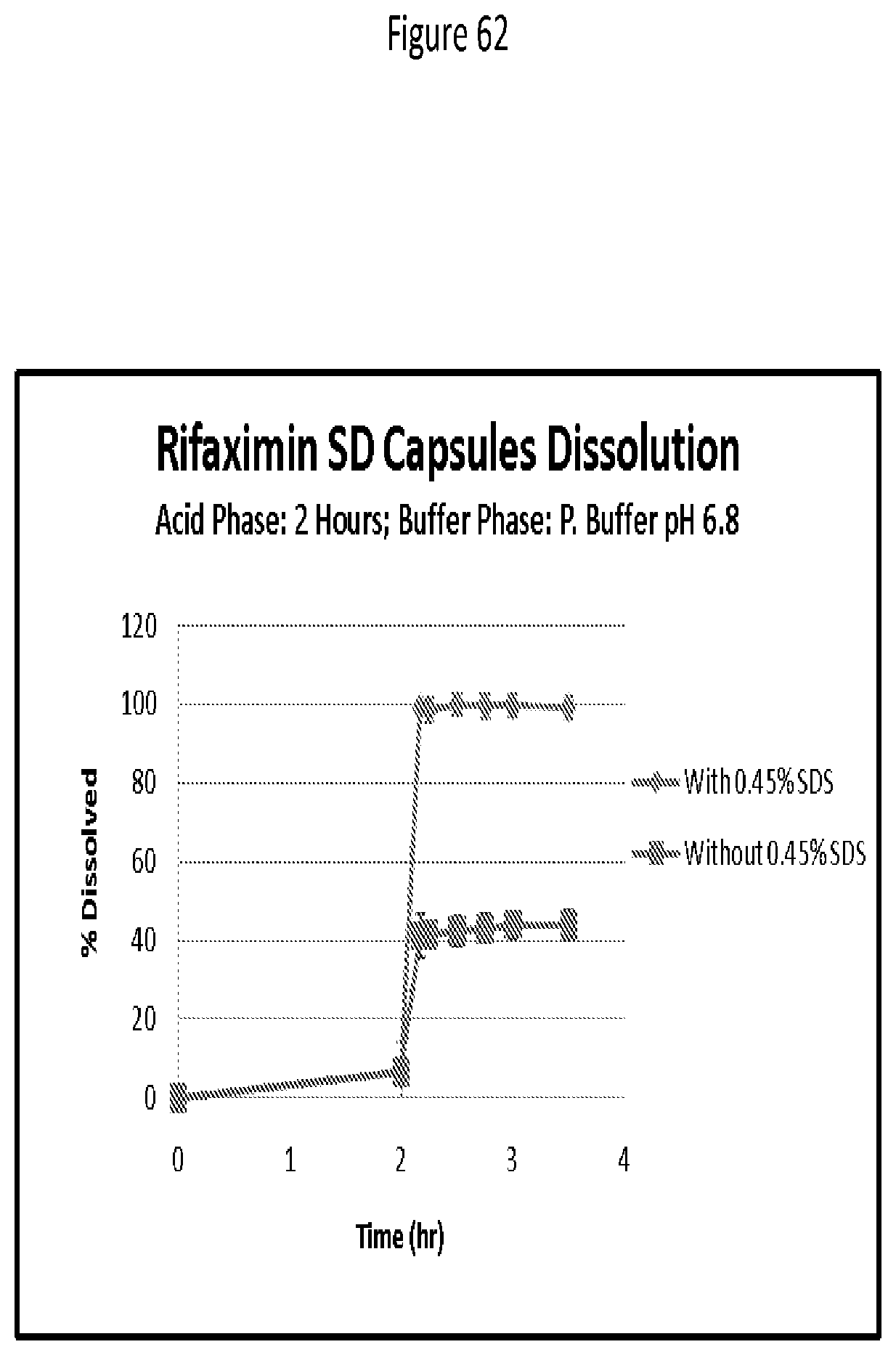
D00063

D00064

D00065
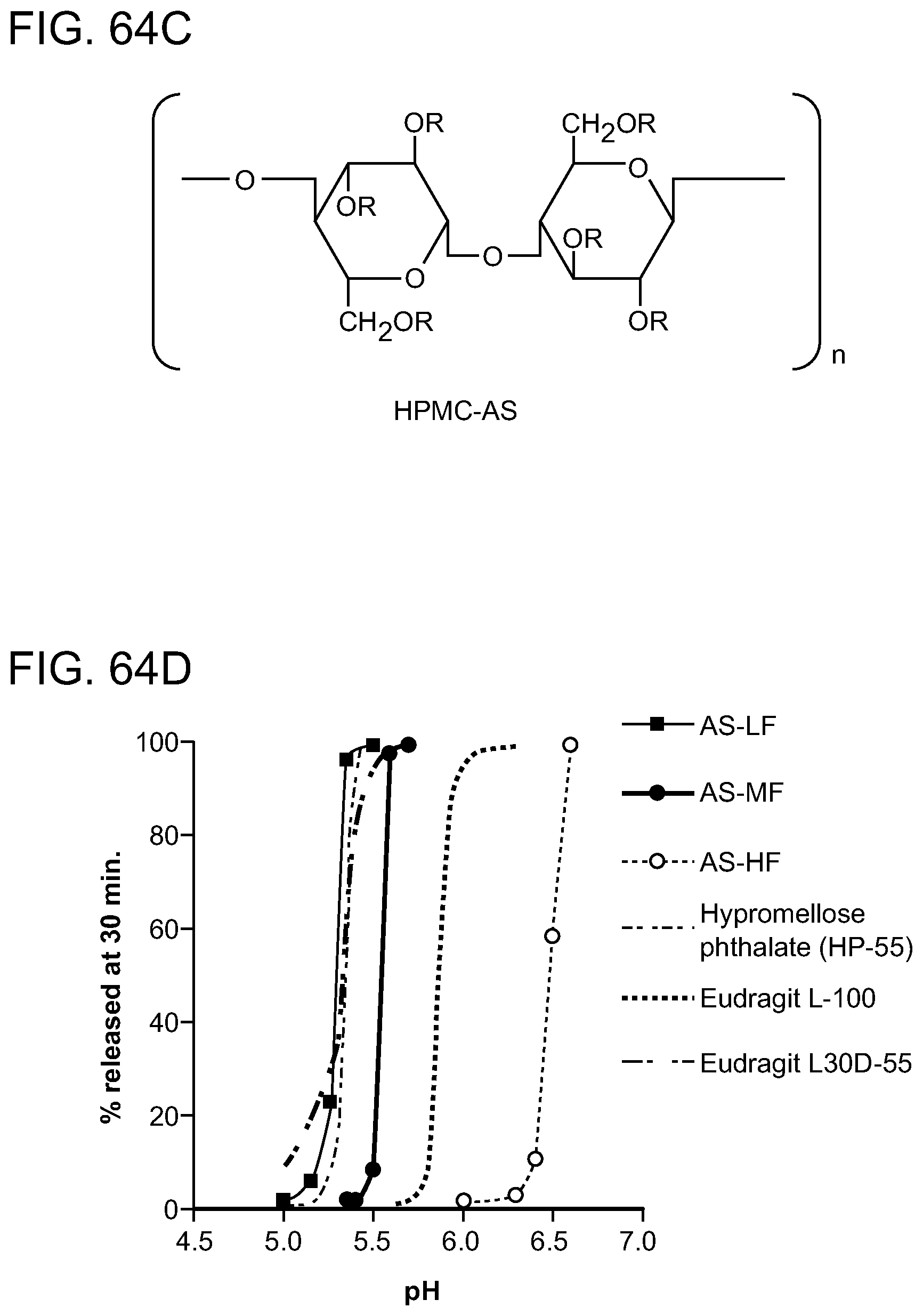
D00066
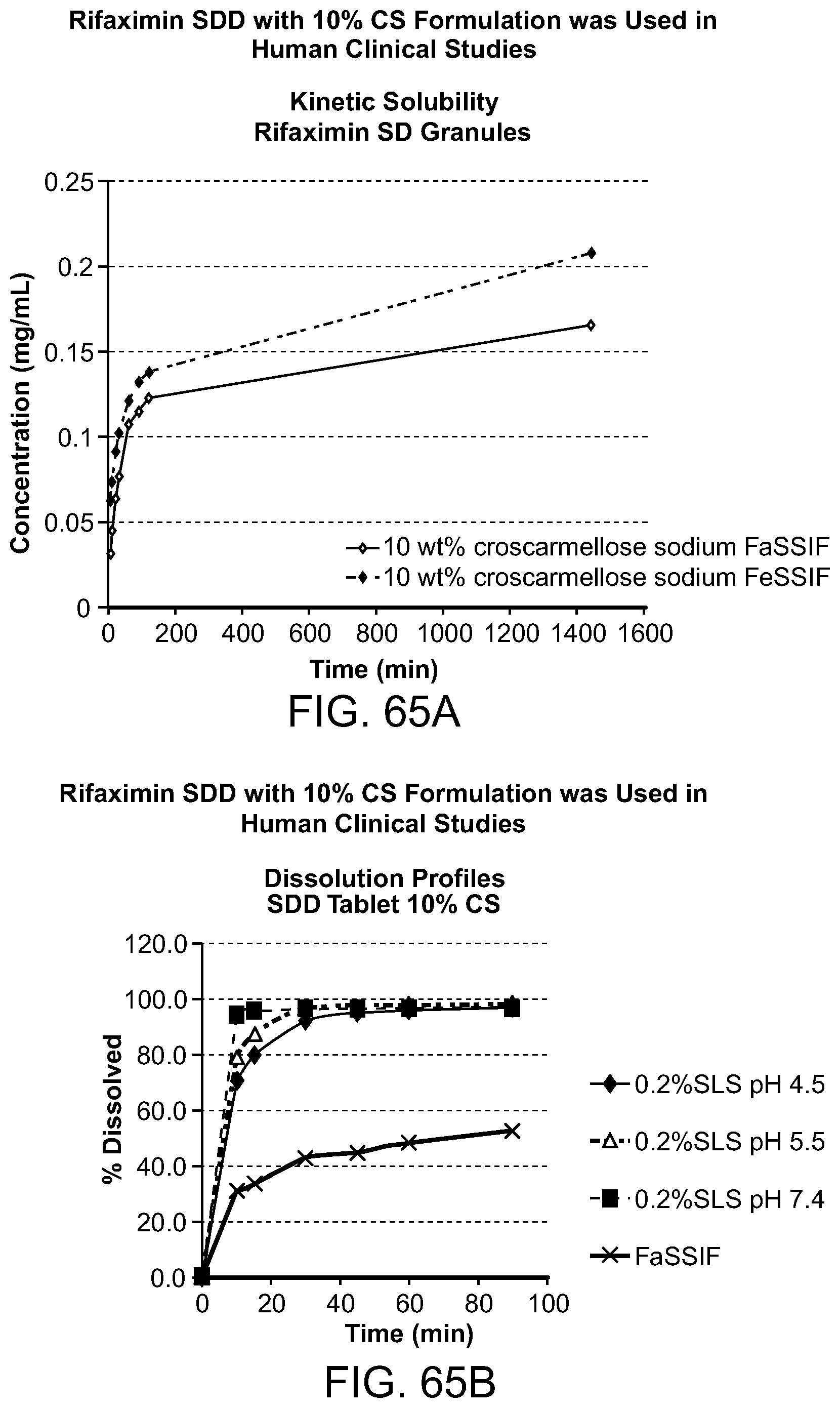
D00067
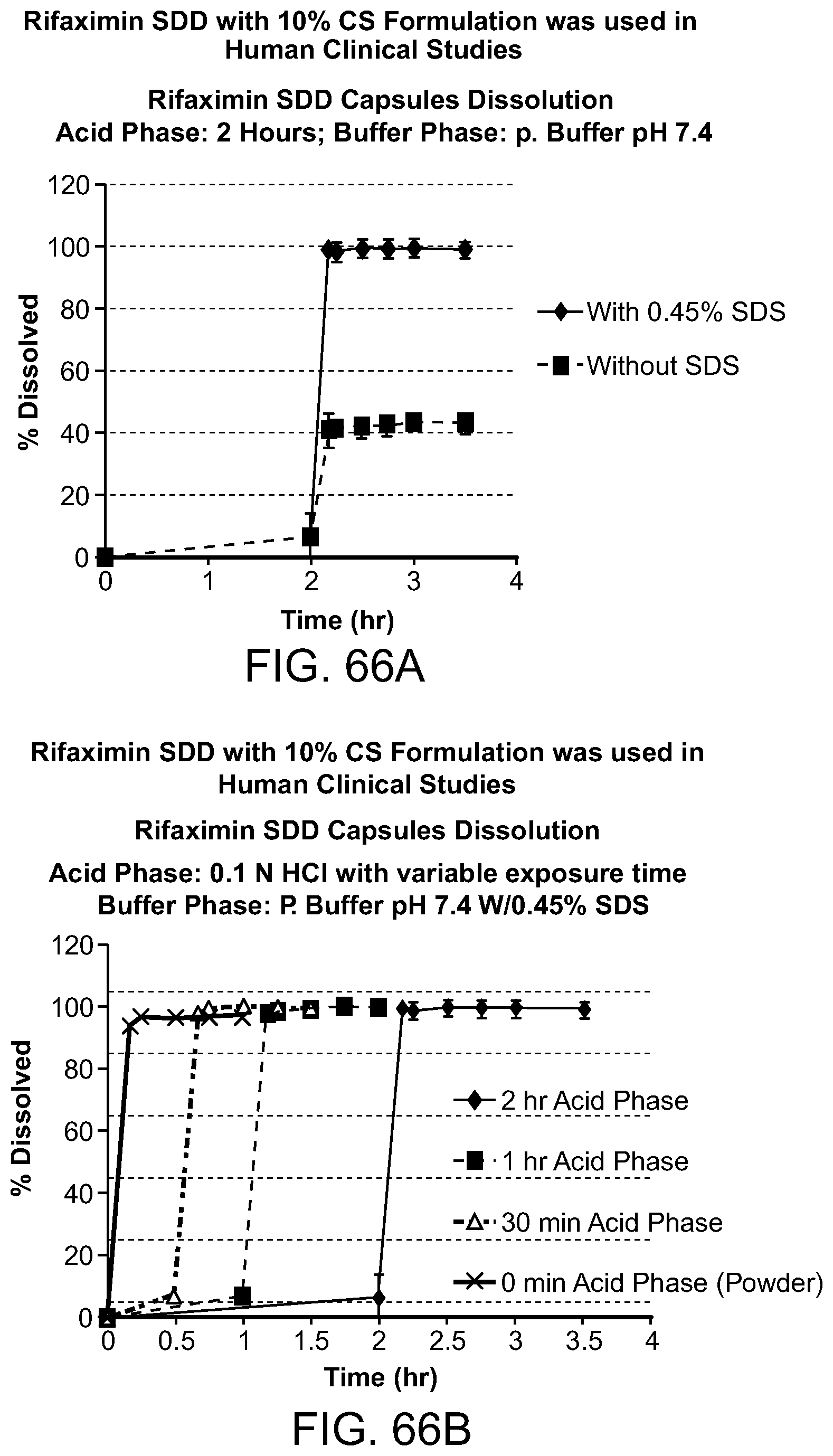
D00068

D00069

D00070
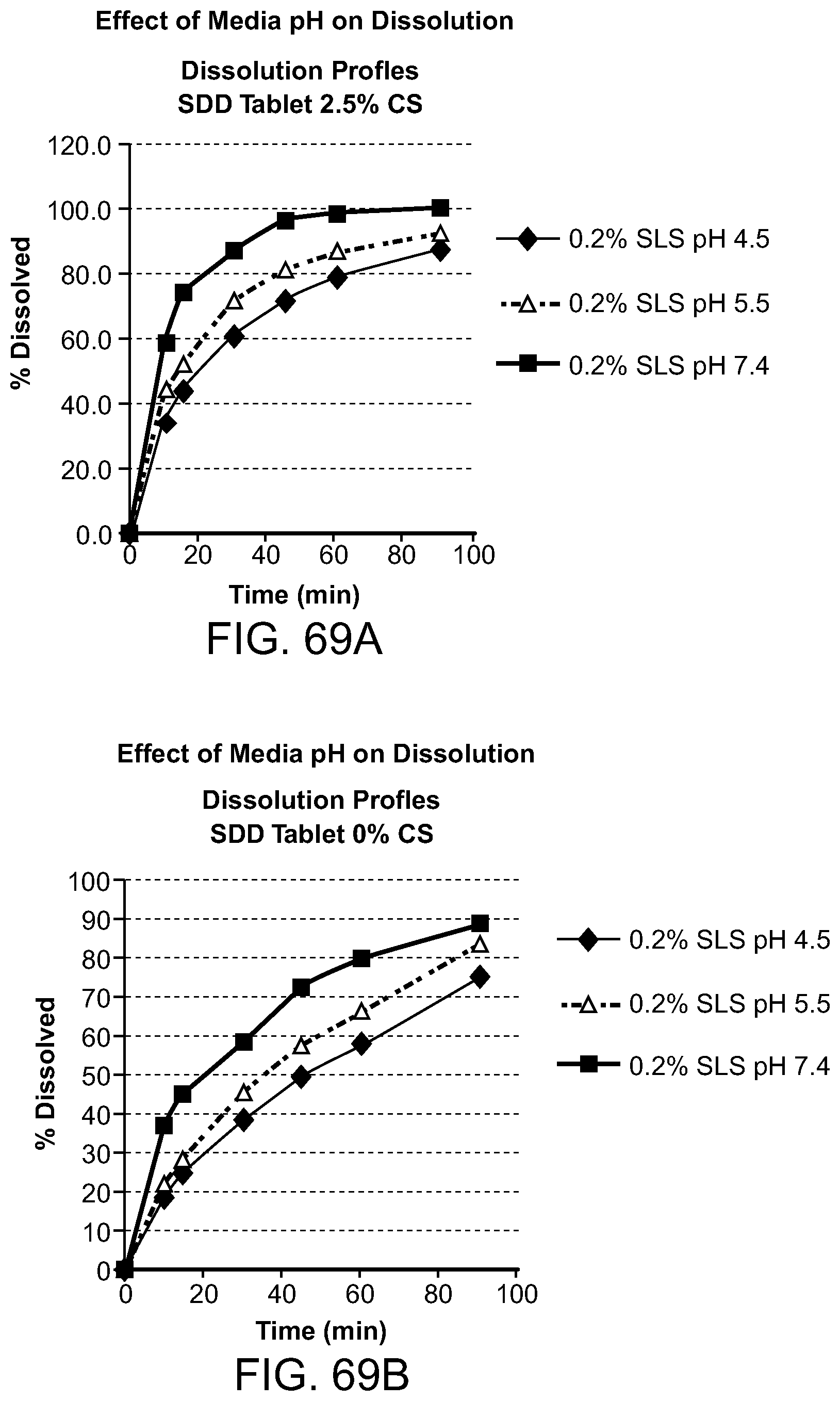
D00071
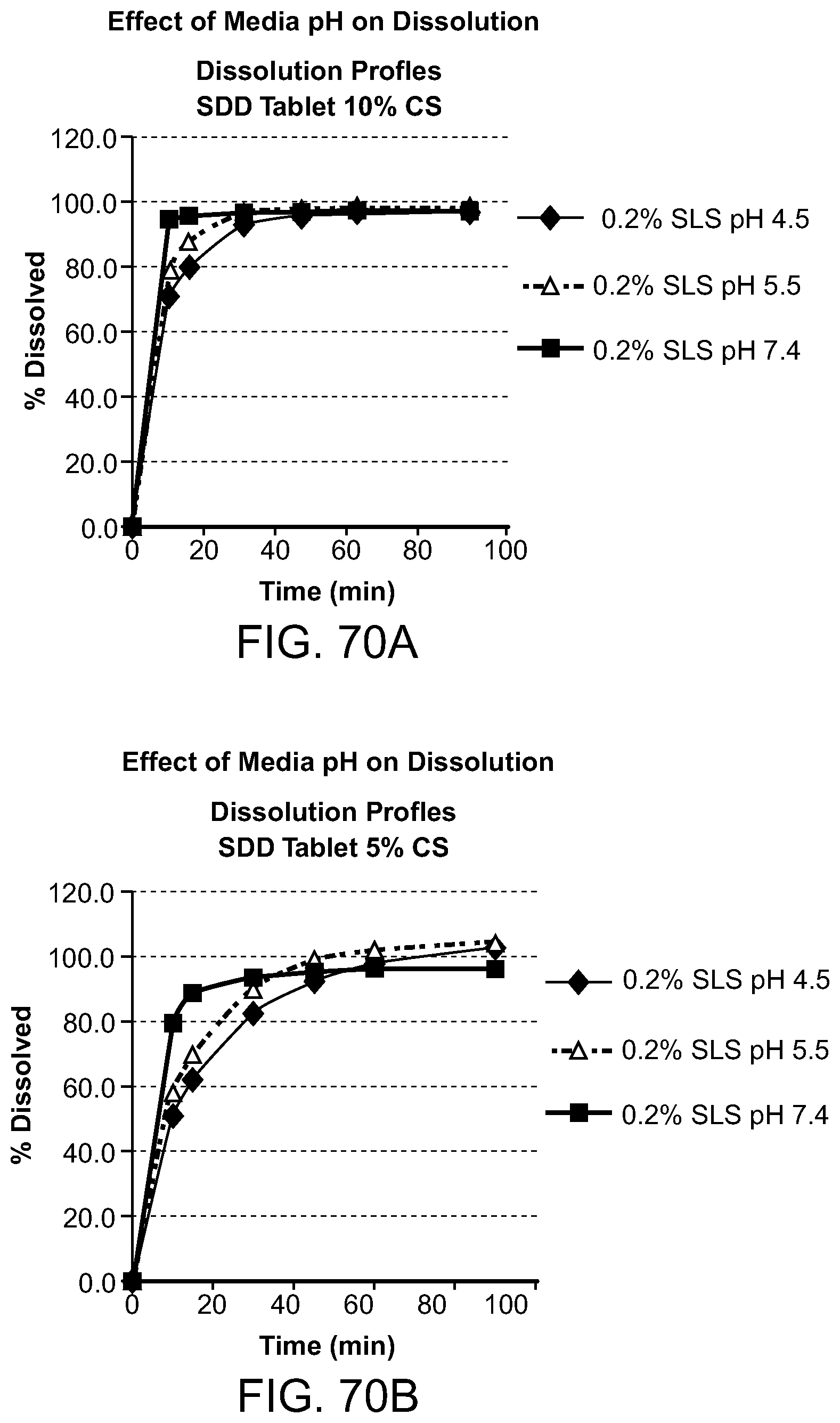
D00072

D00073

D00074
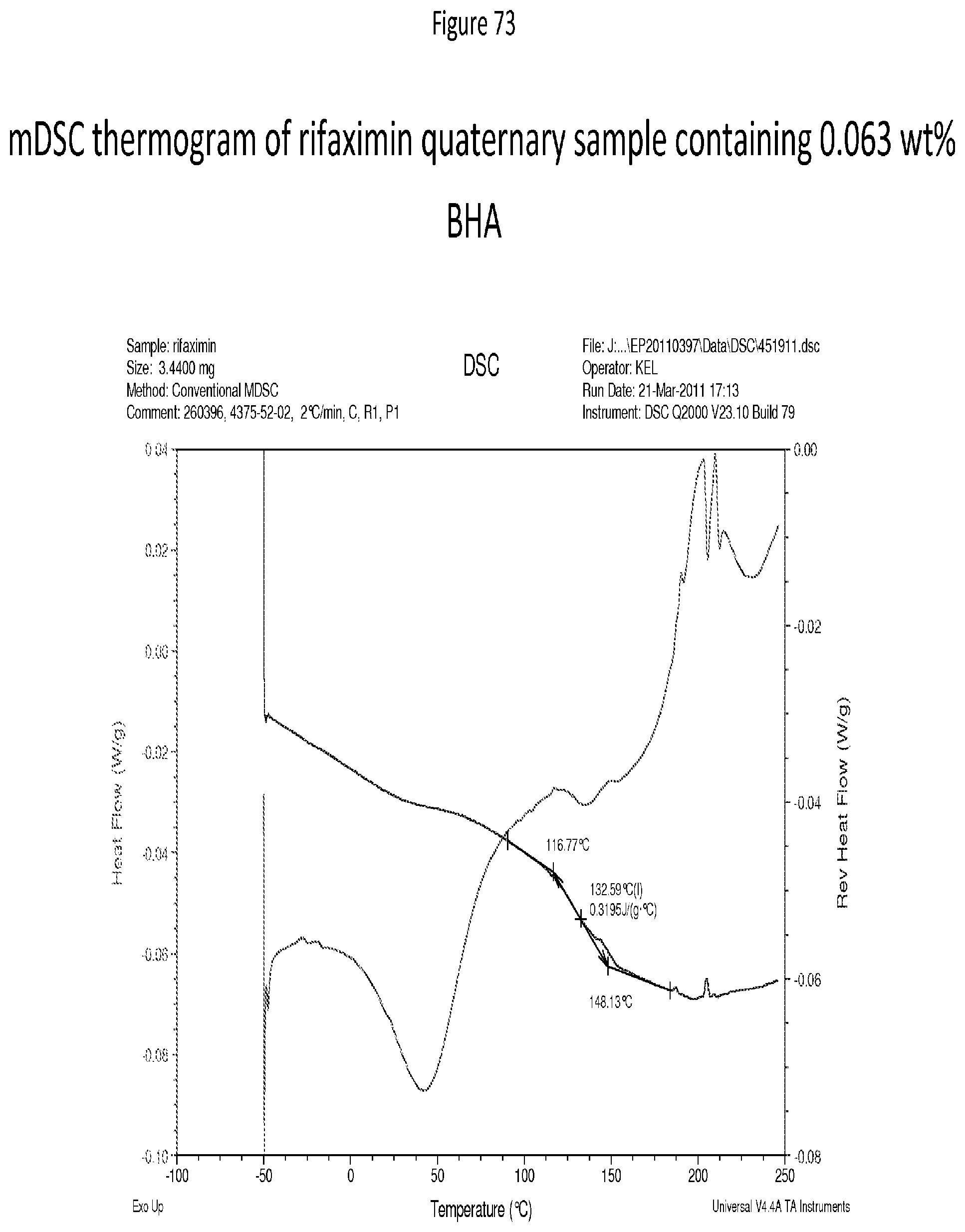
D00075

D00076
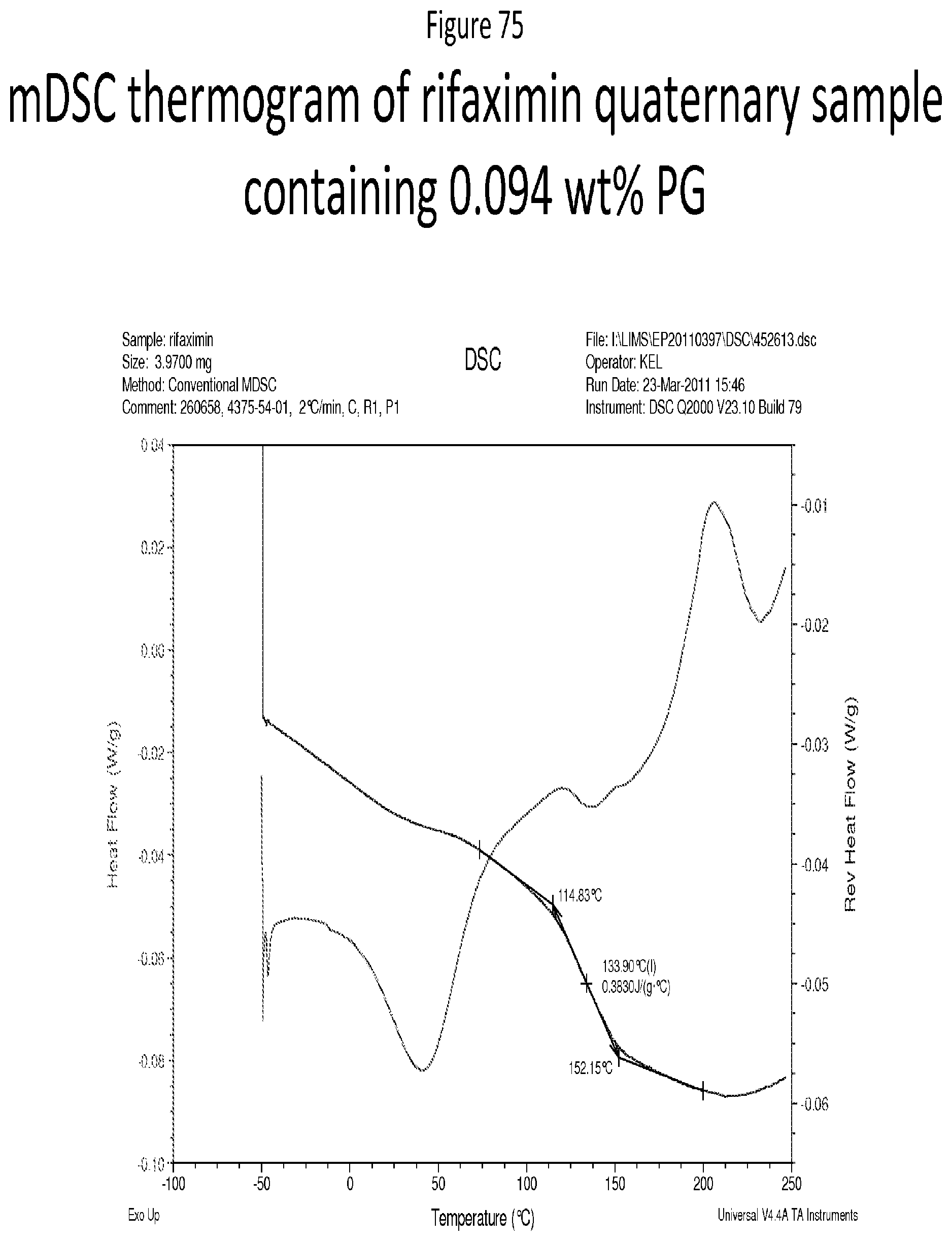
D00077

D00078

D00079
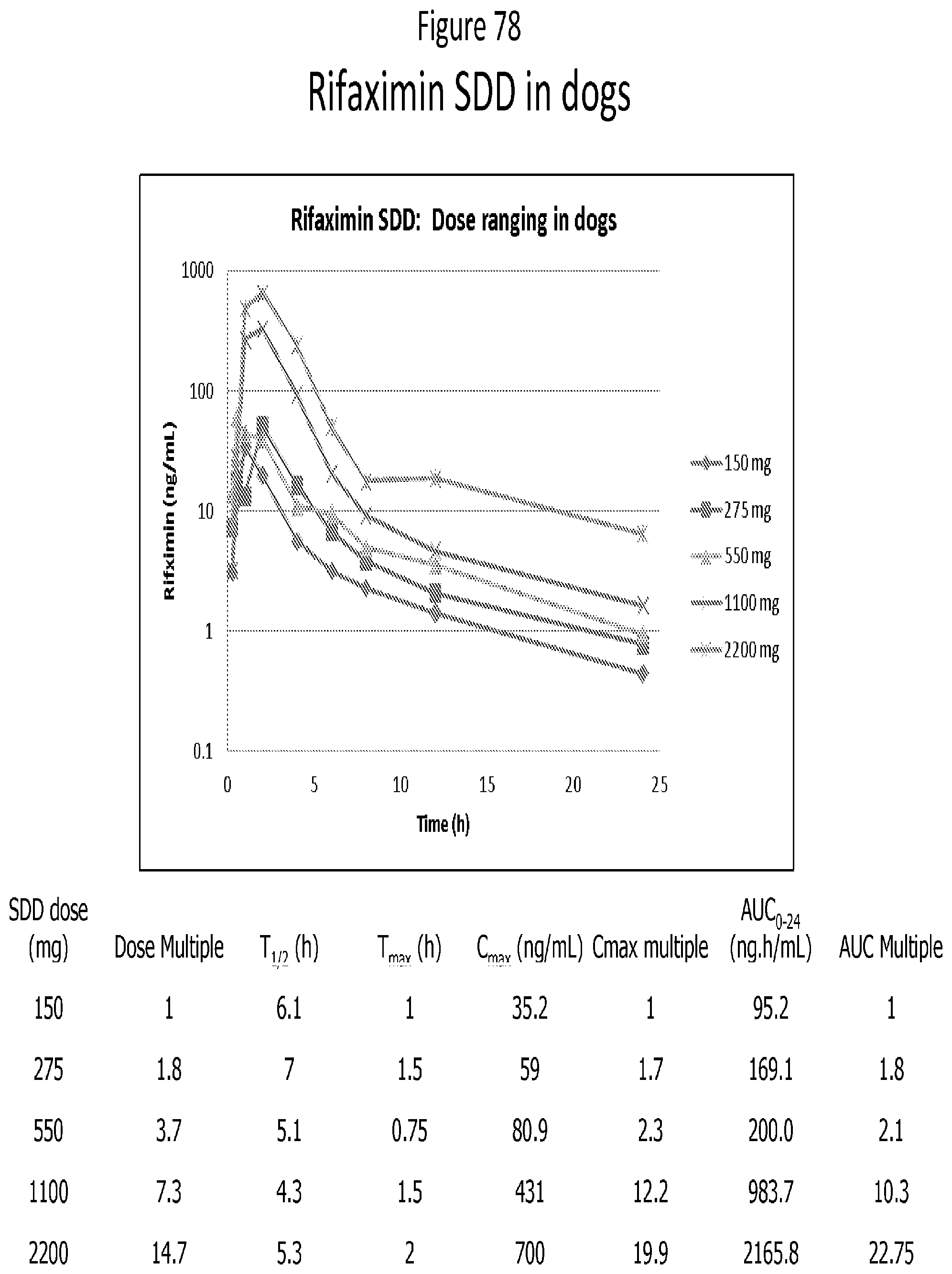
D00080

D00081

D00082
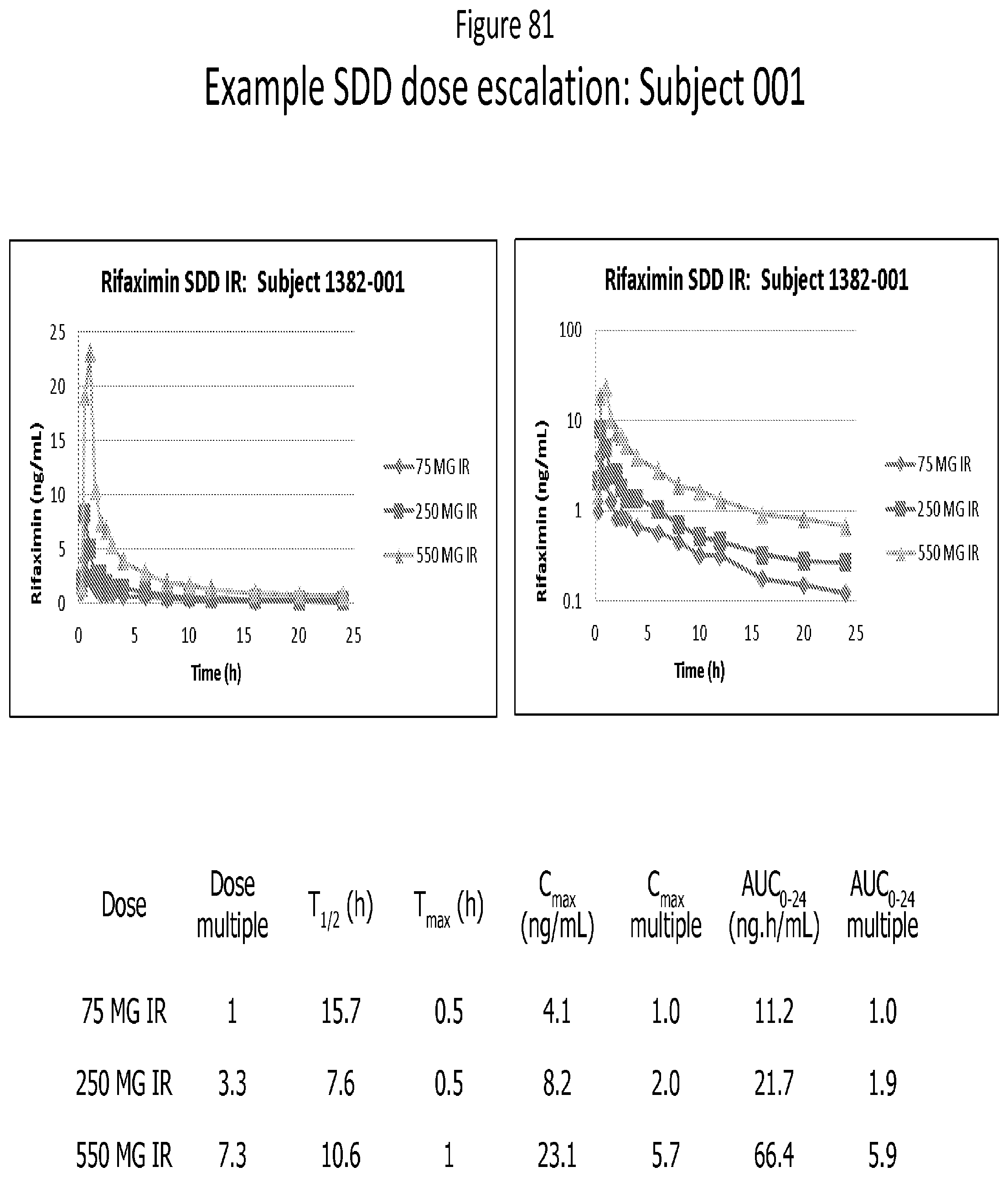
D00083

D00084
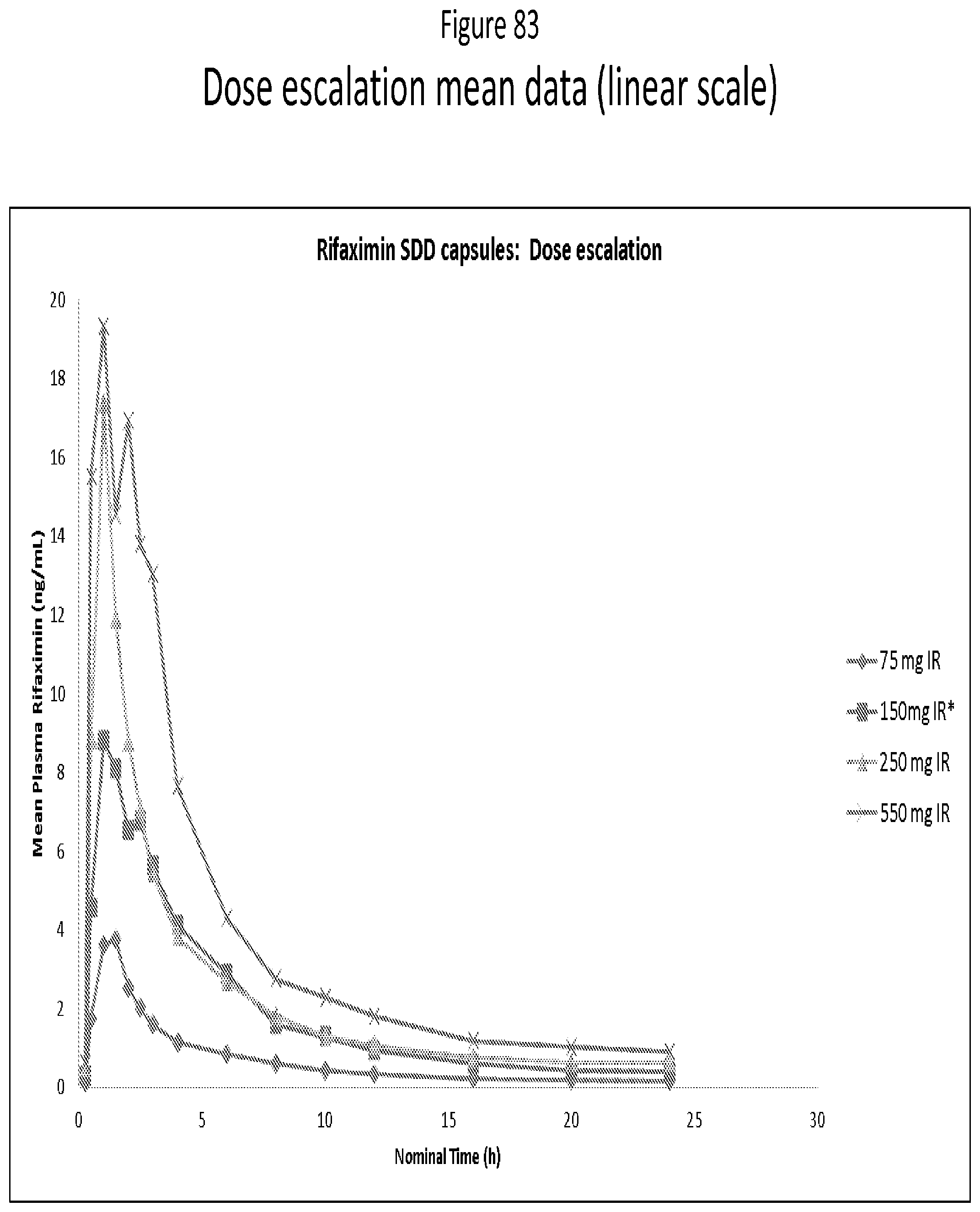
D00085
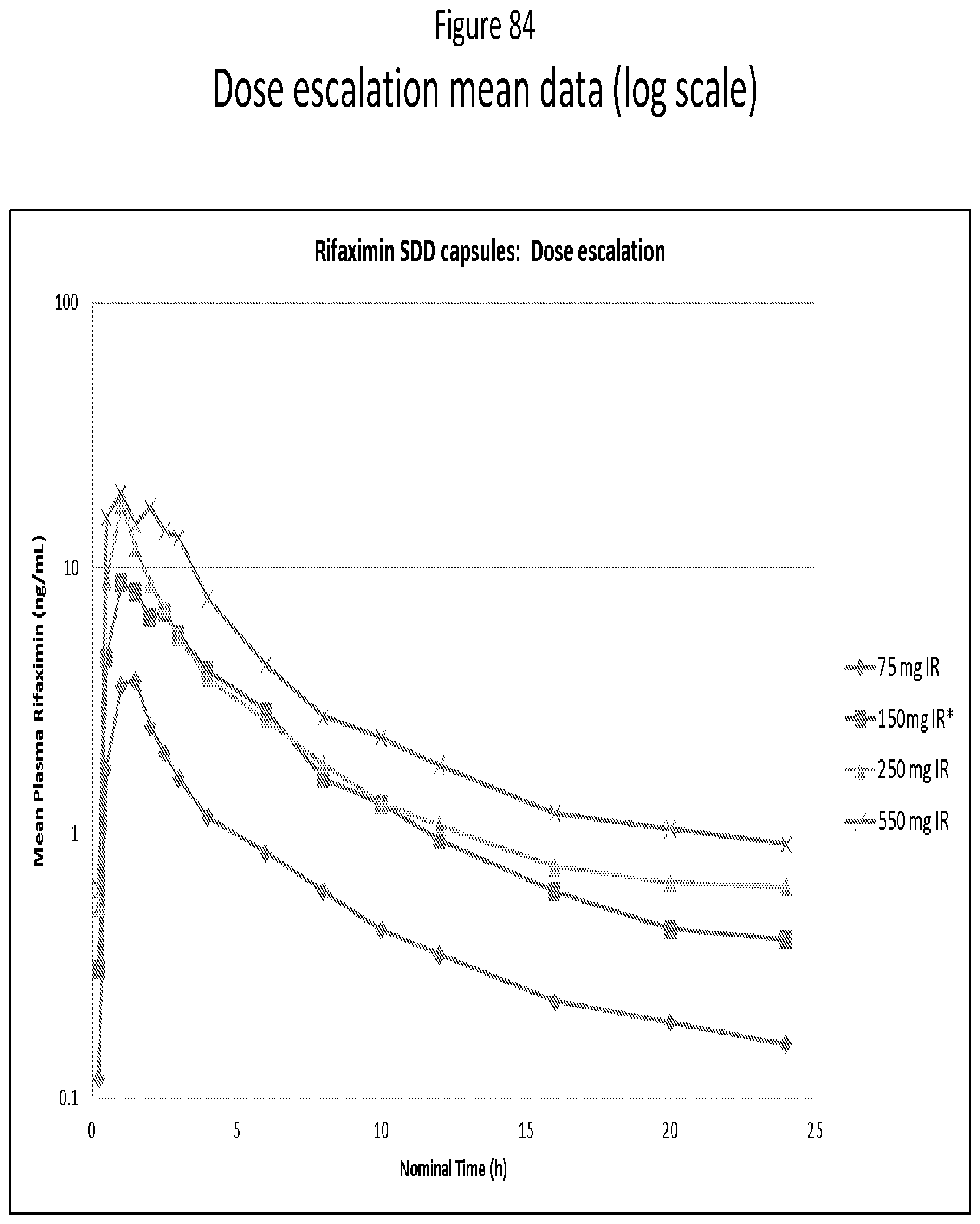
D00086
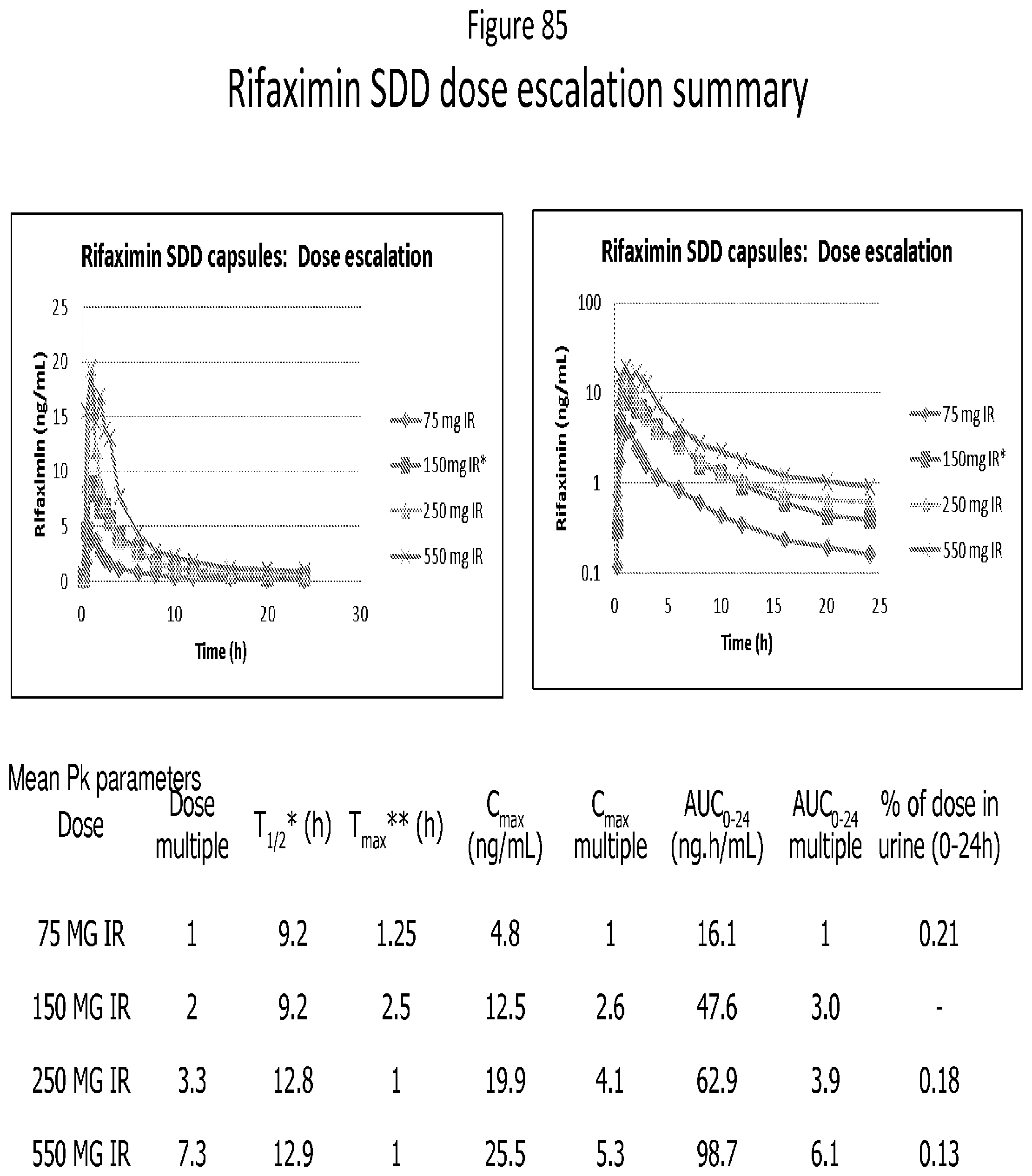
D00087
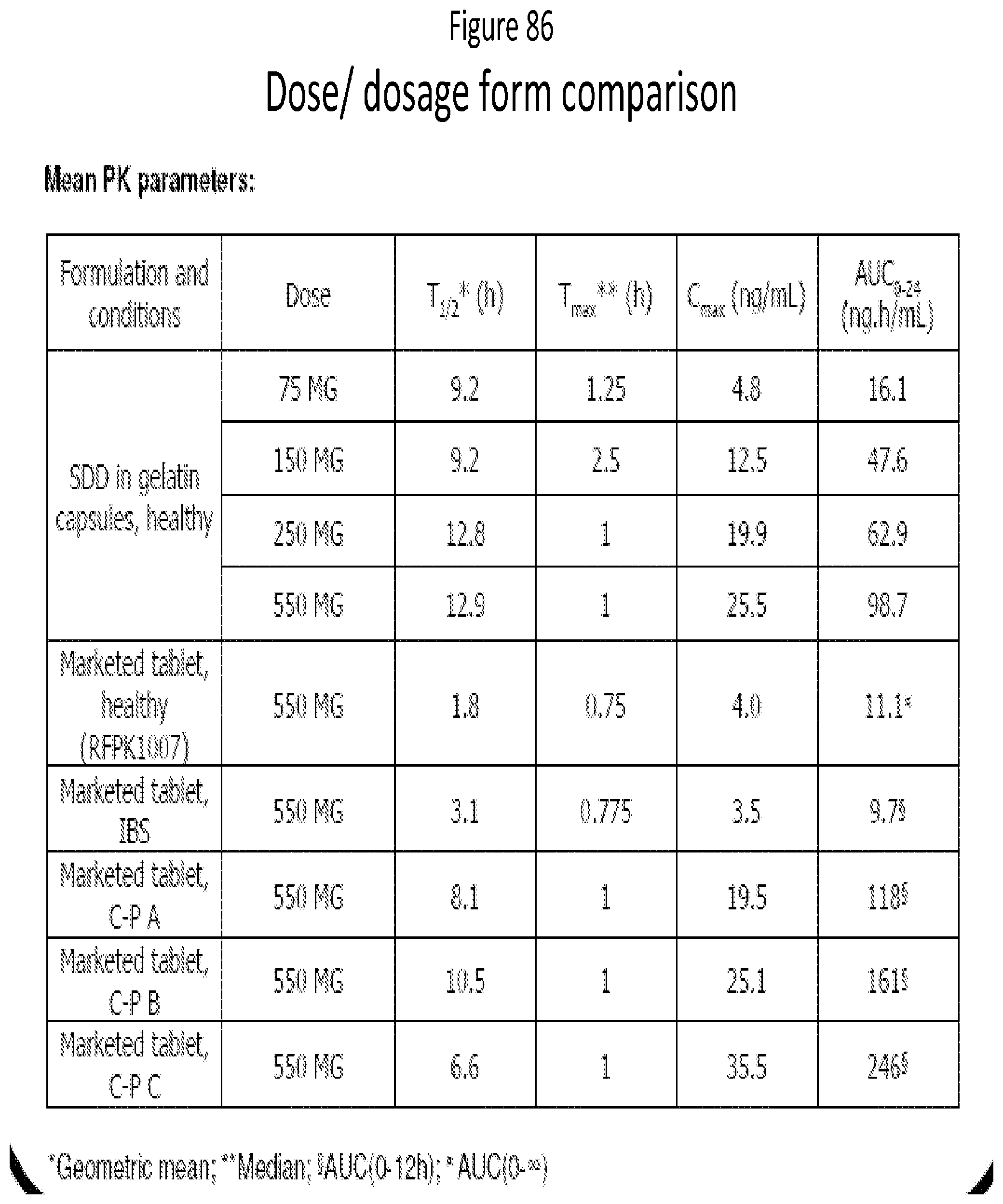
D00088

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.