Selective Conversion of Paraffinic Naphtha to Propylene in the Presence of Hydrogen
Nesterenko; Nikolai ; et al.
U.S. patent application number 16/971743 was filed with the patent office on 2020-12-17 for selective conversion of paraffinic naphtha to propylene in the presence of hydrogen. The applicant listed for this patent is Total Research & Technology Feluy. Invention is credited to Raoul Dethier, Wolfgang Garcia, Nikolai Nesterenko, Valerie Vanrysselberghe.
| Application Number | 20200392418 16/971743 |
| Document ID | / |
| Family ID | 1000005100977 |
| Filed Date | 2020-12-17 |

| United States Patent Application | 20200392418 |
| Kind Code | A1 |
| Nesterenko; Nikolai ; et al. | December 17, 2020 |
Selective Conversion of Paraffinic Naphtha to Propylene in the Presence of Hydrogen
Abstract
The invention relates to a process of catalytic conversion by hydrocracking of paraffinic and naphthenic hydrocarbons from a naphtha feedstock (1) to propylene, the process comprising the steps of providing a naphtha feedstock (1) containing one or more paraffins comprising 4 to 10 carbon atoms; and contacting said naphtha feedstock (1) with a catalyst composition in the presence of hydrogen in a reaction zone under hydrocracking conditions; wherein the catalyst composition consists of one or more zeolite catalysts comprising acid 10-membered ring channels.
| Inventors: | Nesterenko; Nikolai; (Nivelles (Thines), BE) ; Dethier; Raoul; (Schaerbeek, BE) ; Vanrysselberghe; Valerie; (Aalbeke, BE) ; Garcia; Wolfgang; (Braine-l'Alleud, BE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005100977 | ||||||||||
| Appl. No.: | 16/971743 | ||||||||||
| Filed: | February 21, 2019 | ||||||||||
| PCT Filed: | February 21, 2019 | ||||||||||
| PCT NO: | PCT/EP2019/054341 | ||||||||||
| 371 Date: | August 21, 2020 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 2400/28 20130101; C10G 2300/4018 20130101; C10G 65/043 20130101; C10G 2300/4081 20130101; C10G 2300/4012 20130101; C10G 47/16 20130101; C10G 2400/20 20130101; C10G 2300/4006 20130101 |
| International Class: | C10G 65/04 20060101 C10G065/04; C10G 47/16 20060101 C10G047/16 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Feb 22, 2018 | EP | 18158160.4 |
| Feb 22, 2018 | EP | 18158175.2 |
Claims
1.-15. (canceled)
16. A process of catalytic conversion by hydrocracking of paraffinic and naphthenic hydrocarbons from a naphtha feedstock to propylene, the process being characterized in that it comprises the following steps: a) providing a naphtha feedstock containing one or more paraffins comprising 4 to 10 carbon atoms; and b) submitting said naphtha feedstock to a hydrocracking step by contacting said naphtha feedstock with a catalyst composition in the presence of hydrogen in a reaction zone under hydrocracking conditions to produce an effluent; c) submitting the effluent to a separation step to recover propane; and d) submitting said propane to a step of dehydrogenation into propylene in a propane dehydrogenation reactor; e) collecting hydrogen produced in the step of dehydrogenation into propylene, and recycling said hydrogen back to the hydrocracking reaction zone in order to perform the hydrocracking step; and in that the catalyst composition of the hydrocracking step comprises one or more zeolite catalysts comprising one or more acid 10-membered ring channels.
17. The process according to claim 16, characterized in that the naphtha feedstock comprises at least 10 wt % of naphthenes as based on the total weight of the naphtha feedstock.
18. The process according to claim 16, characterized in that the one or more zeolite catalysts have a Si/Al molar ratio ranging from 10 to 100.
19. The process according to claim 16, characterized in that the hydrocracking conditions of the hydrocracking step comprise: a. the naphtha feedstock being contacted with the catalyst composition at a temperature ranging from 200 to 600.degree. C., and/or b. the naphtha feedstock being contacted with the catalyst composition at a pressure ranging from 1 to 10 MPa.
20. The process according to claim 16, characterized in that the catalyst composition contains no added noble metal and no added transition metals.
21. The process according to claim 16, characterized in that the catalyst composition contains at most 50 ppm wt of noble metal and less than 0.05 wt % of transition metals as based on the total weight of the catalyst composition.
22. The process according to claim 16, characterized in that the hydrocracking conditions of the hydrocracking step comprise the naphtha feedstock being contacted with the catalyst composition at a WHSV (feed) of at least 0.1 h.sup.-1
23. The process according to claim 16, characterized in that, in the hydrocracking step, hydrogen is provided to the naphtha feedstock at a molar ratio H.sub.2/Naphtha ranging from 1000:1 to 1:1.
24. The process according to claim 16, characterized in that the catalyst composition comprises at least 60 wt % of one or more zeolite catalysts comprising one or more acid 10-membered ring channels.
25. The process according to claim 16, characterized in that the catalyst composition comprises one or more zeolite catalysts selected from the list comprising ZSM-5, silicalite-1, ZSM-11, silicalite-2, SSZ-46, MCM-68, CIT-1, SSZ-33, ZSM-8, Ferrierite, FU-9, ZSM-35, ZSM-23, ZSM-22, Theta-1, NU-10, ZSM-50, EU-1, ZSM-57, SAPO-11 and ZSM-48.
26. The process according to claim 16, characterized in that the one or more zeolite catalysts comprising one or more acid 10-membered ring channels comprise or are zeolites catalysts of the MFI-type.
27. The process according to claim 16, characterized in that the catalyst composition comprises a binder selected from silica, alpha-alumina, gamma-alumina, clays, alumina phosphates, mullite, zirconia, titania, yttria, silicon nitride, silicon carbide, iron, bronze and stainless steel, glass, and carbon.
28. The process according to claim 16, characterized in that the step of dehydrogenation into propylene is performed at a temperature ranging from 500 to 800.degree. C. and a partial pressure of propylene below one atmosphere.
29. The process according to claim 16, characterized in that the separation step comprises recovering a C4 paraffins fraction from the effluent of step b) and a step of recycling back the C4 paraffins fraction to the hydrocracking reaction zone, and/or the separation step comprises recovering a C5+ hydrocarbon fraction from the effluent of step b) and a step of valorization of said C5+ hydrocarbon fraction as gasoline.
30. The process according to claim 16, characterized in that it comprises a step of recovering the unconverted propane after the step of dehydrogenation into propylene and recycling it to the propane dehydrogenation reactor to be submitted to a further step of dehydrogenation into propylene.
31. The use of one or more zeolite catalysts comprising the one or more acid 10-membered ring channels in a process according to claim 16 of catalytic conversion by hydrocracking of paraffinic and naphthenic hydrocarbons from a naphtha feedstock to propane, and further dehydrogenation of said propane to propylene, characterized in that the zeolite catalysts contains at most 50 ppm wt of noble metal and less than 0.05 wt % of transition metals as based on the total weight of the catalyst composition.
Description
FIELD OF THE INVENTION
[0001] The invention relates to a process for producing propane from a naphtha feedstock comprising paraffinic and naphthenic hydrocarbons, and to further produce propylene from the propane recovered. The invention also relates to the use of catalyst compositions to improve propane selectivity in a process for producing propane from a naphtha feedstock comprising paraffinic and naphthenic hydrocarbons.
BACKGROUND OF THE INVENTION
[0002] World demand for propylene is expected to continuously grow up at an average annual rate of 4-5%. Currently, propylene is mainly produced as a by-product of liquefied petroleum gas (LPG)/naphtha steam crackers and Fluid Catalytic Cracking (FCC) units. With the startup of additional ethane-based ethylene capacities (lighter fractions like ethane or LPG are considered very advantageous naphtha feedstock), the production of propylene and aromatics via the steam cracker declines. Furthermore, the demand for propylene is actually growing faster than that of ethylene. To some extent, the propylene production can be optimized by transforming an FCC unit into a petrochemical FCC after a major revamp. However, a significant amount of dry gases and low value of by-products will be produced. In addition, an important revamp of the existing FCC unit and significantly higher process severity would be required to crack the naphtha to propylene and ethylene.
[0003] All the aforementioned factors have created an imbalance of supply and demand for propylene: a gap is being established between the available propylene supplies to meet ongoing demand growth. While the markets have evolved to the point where modes of propylene by-product production can no longer satisfy the demand for propylene, the traditional classification of propylene as a "by-product" begun to evolve into more of classification as a "coproduct" or even a "primary product".
[0004] Propane dehydrogenation (PDH) is the fastest on-purpose growing technology today to bridge the gap. The technology offers a mean of producing propylene as a single product near consumer markets. PDH system is suitable as stand-alone facilities not requiring integration with a nearby olefin or refinery unit but requires a secured supply of a big amount of propane (>0.5 Mt/y).
[0005] The PDH units used to be only appropriate for a limited amount of geographical regions, where propane was highly available. Today, the majority of the PDH projects are based on the imported propane from the United States of America (US). A direct propane import from the producers in the US or the Middle East has many limits on volume and requires sophisticated logistics. On the contrary, the market expects an important surplus in the availability of naphtha due to a massive shift to ethane as feedstock to steam-crakers and low oil prices. Therefore, there is a need for the development of a technology to transform naphtha to propylene with significant yield advantages versus naphtha steam cracker and petro FCC.
[0006] The majority of the catalytic processes to crack paraffinic naphtha is dedicated to direct production of propylene in absence of hydrogen at a temperature higher than 550.degree. C. and low partial pressure of hydrocarbon below atmospheric. These conditions reduce hydrogen transfer reactions and favor propylene and aromatics vs propane (D5943, Applied Catalysis A: General 398 (2011) 1-17). Propane is formed but as an undesirable side product. One should mention also that under high-temperature conditions, a lot of ethylene is produced which, in the context of growing ethylene supply from ethane crackers, reduces significantly attractiveness of the process.
[0007] The isomerization and hydrotreating processes of naphtha are well known to produce clean fuel from linear paraffinic species existing in light naphtha. Both processes occur in the presence of hydrogen at moderate pressure on the metal-containing catalysts with a purpose to increase fuel qualities. Light formation is big drawback for both reactions. A post-reforming process known as selectoforming was commercialized in the 1960s for raising the octane rating of reformates while producing propane as a by-product (Chen et al., 1968; Burd and Maziuk, 1972).
[0008] Some articles report naphtha hydrocracking, for example: "High Yields of LPG Via n-Hexane Hydrocracking Using Unloaded Acidic Zeolite Catalysts" in Journal Petroleum Science and Technology, Volume 33, 2015--Issue 12, includes investigating the hydrocracking of n-hexane, as a low-octane naphtha component to high-octane gaseous motor fuel (LPG) in a pulse flow atmospheric microreactor using untreated and steam-treated H-MOR, H-BEA, or H-ZSM-5 zeolite catalysts. All zeolites catalysts were metal-free and their bi-functionality depended only on the Bronsted zeolitic acid sites. Zeolites catalysts H-ZSM-5 and St-H-ZSM-5 acquire very low catalytic activities mainly due to their narrow pore structure, as well as due to their partial pore filling by Al debris in case of St-H-ZSM-5.
[0009] In "Conversion of light naphthas over sulfided nickel erionite" in Industrial and Engineering Chemistry Research Volume 32, Issue 6, 1993, Pages 1003-1006, a natural zeolite erionite has been exchanged with ammonium and nickel salts to yield a Ni/H erionite catalyst that is active and stable for selectively hydrocracking only the n-paraffins from light straight-run naphthas. The primary product is a C5+ liquid that has 15-20 octane numbers higher than the feed and a propane- and butane-rich gas by-product. Results from a 110-day pilot plant run demonstrated that a catalyst life of more than 1 year should be possible. Naphthenes, aromatics, and isoparaffins are neither produced nor consumed in this process, resulting in a C5+ liquid product that is lower in benzene and total aromatics than attainable by catalytic reforming of these feeds. Although no further work is planned with this catalyst, a naphtha-upgrading process based on shape-selective zeolitic hydrocracking could provide an attractive alternative to catalytic reforming or isomerization for these hard-to-upgrade naphthas. It should be particularly attractive in areas where the propane and butane by-products have good value.
[0010] WO2012/071137 describes the use of a catalyst comprising at least one zeolite having 10-membered ring channels and at least one group VIb, VIIb and/or VIII metal, in the presence of hydrogen at elevated temperature and elevated pressure. The process allowed to convert at least 40 wt % of the paraffins comprising from 4 to 12 carbon atoms based on the total weight of paraffins in the feed to ethane and/or propane to obtain a hydrocracked gas cracker naphtha feedstock comprising ethane and/or propane. The process was focussed on maximizing the production of ethane. The drawback of the invention is an important formation of methane due to the presence of the noble metals in the catalyst at about 6-20 wt %.
[0011] US 2017/058210 discloses a process for producing BTX comprising pyrolysis, aromatic ring opening and BTX recovery.
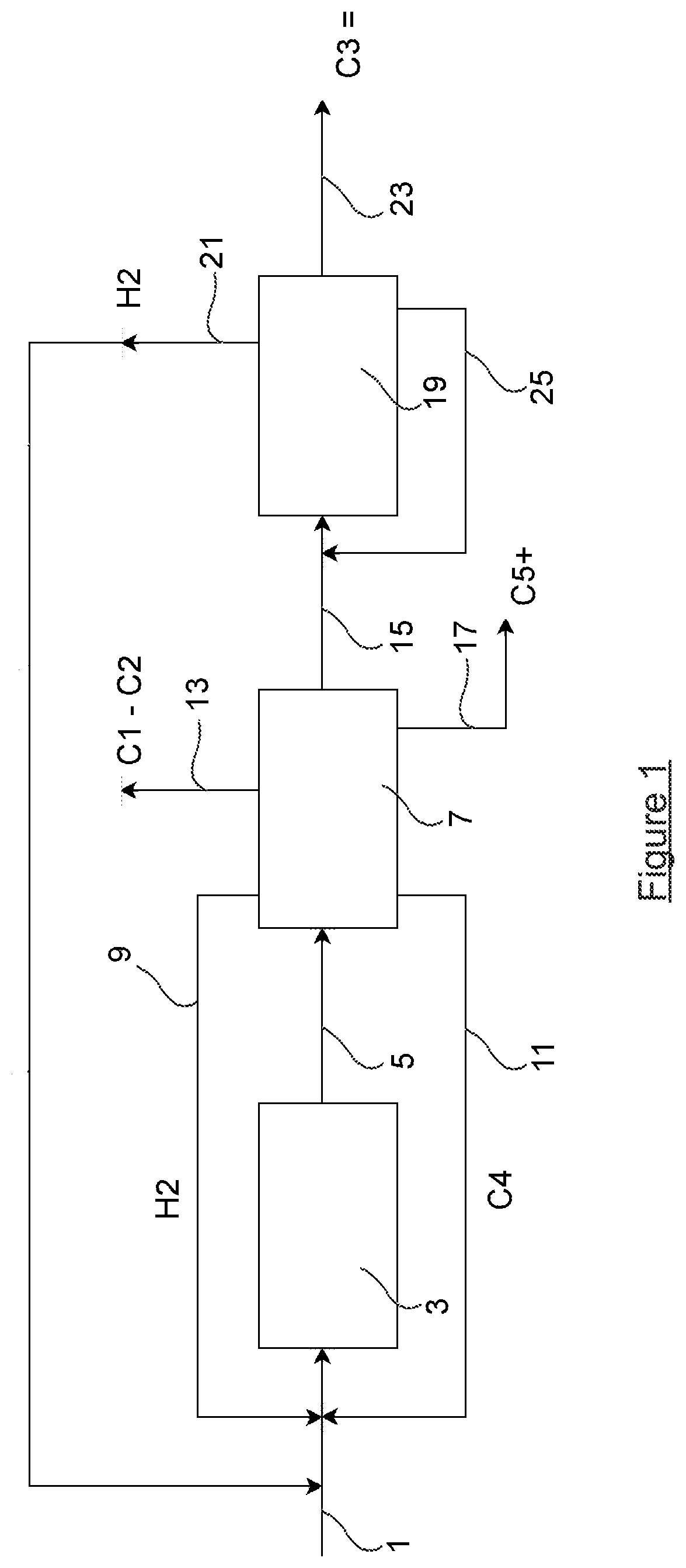
[0012] US 2016/369191 discloses a process for cracking a hydrocarbon feedstock in a steam cracker unit where a liquid hydrocarbon feedstock is fed to a hydrocracking unit, the stream hydrocracked are then separated into a high content aromatics stream and a gaseous stream comprising C2-C4 paraffins, hydrogen and methane, the C2-C4 paraffins are then separated form said gaseous stream and fed to a steam cracker unit.
[0013] US 2017/369795 discloses a process for producing C2 and C3 hydrocarbons, comprising a) subjecting a mixed hydrocarbon stream to first hydrocracking in the presence of a first hydrocracking catalyst to produce a first hydrocracking product stream; and b) subjecting the first hydrocarbon product stream to C4 hydrocracking.
[0014] WO 98/56740 discloses a process for improving the conversion of a hydrocarbon feedstock to light olefins comprising the steps of thermally converting the hydrocarbon feedstock to produce an effluent; quenching the effluent to produce a quenched effluent; and contacting the quenched effluent with a light olefin-producing cracking catalyst.
[0015] It was discovered that the naphtha may selectively be transformed into propane on the metal free catalyst (i.e. noble and transition metal free catalyst) with very low selectivity to methane (from 0.3 to 1.5 wt % CH4). This opens an opportunity for a high efficient process of production of propylene from naphtha via a combination of hydrocracking of naphtha to propane followed by dehydrogenation of propane to propylene. The dehydrogenation process produces hydrogen, which could be used at least partially for the first step.
SUMMARY OF THE INVENTION
[0016] According to a first aspect, the invention provides a process of catalytic conversion by hydrocracking of a paraffinic and naphthenic hydrocarbons from a naphtha feedstock to propylene, wherein the process comprises the following steps: [0017] a) providing a naphtha feedstock containing one or more paraffins comprising 4 to 10 carbon atoms with preferably no olefins i.e. an olefin content of less than 1 wt % preferably less than 0.1 wt % even more preferably less than 0.01 wt % relating to the total weight of said naphtha feedstock; and [0018] b) submitting said naphtha feedstock to a hydrocracking step by contacting said naphtha feedstock with a catalyst composition in the presence of hydrogen in a reaction zone under hydrocracking conditions to produce an effluent; [0019] c) submitting the effluent to a separation step to recover propane; and [0020] d) submitting said propane to a step of dehydrogenation into propylene in a propane dehydrogenation reactor; [0021] e) collecting hydrogen (21) produced in the step of dehydrogenation into propylene (23), and recycling said hydrogen (21) back to the hydrocracking reaction zone in order to perform the hydrocracking step (3) and wherein the catalyst composition of the hydrocracking step comprises one or more zeolite catalysts comprising an acid 10-membered ring channels.
[0022] Surprisingly, it was found by the inventors that the catalyst composition comprising acid 10-membered ring channels was particularly resistant to deactivation. Indeed the hydrogen recycled from the dehydrogenation step to the hydrocracking reaction zone is not necessarily of the highest purity. This hydrogen can contain methane or other impurities like sulphur at relatively high concentration that deactivate traditional hydrocracking catalyst. The use of the catalyst composition comprising acid 10-membered ring channels is resistant to deactivation induced by the methane contained in hydrogen. Such catalyst allows avoiding expensive purification of the hydrogen. The hydrogen feed stream can for instance contain up to 5 wt % of methane, preferably up to 10 wt %, even more preferably up to 20 wt % of methane based on the total weight of the hydrogen feed stream. Other impurities may also be present such as sulphur compounds, such as H.sub.2S, at concentration for instance up to 0.1% wt, preferably 1% wt, even more preferably up to 5 wt % based on the total weight of the hydrogen feed stream. In a preferred embodiment, the one or more acid zeolite catalysts or catalyst composition are metal-free, containing less than 1000 ppm of noble metal and less than 1% of transition metals. The content of the noble metals is below 1000 ppm, preferably below 500 ppm, more preferably at most 250 ppm even more preferably at most 50 ppm wt, the most preferred being no noble metal at all, that is to say below the detection limit. In exceptional cases, traces of noble metals (Pt, Pd) may be introduced to increase the stability of the zeolite and hydrogenate the coke precursors. However, higher concentration of the metals may lead to changes of the reaction mechanism. Presence of a significant amount of noble metals (>0.2 wt %) on the catalyst leads to a different reaction mechanism, which involves activation of the hydrogen on the metal (hydrogen spillover) followed by subsequent cracking. This leads to a different selectivity pattern and formation of higher amount of ethane and methane. It is therefore preferred to have no noble metals at all.
[0023] As used herein the terms "metal-free" indicate that the one or more acid zeolite catalysts are also devoid of any transition metal selected from the groups of Fe, Ni, Co, W, Mo, Ga, Zn. The content of the transition metals is below 1.0 wt %, preferably below 0.5 wt % more preferably below 0.05 wt % even more preferably below 0.01 wt %. Traces of these metals may be present on the catalyst as impurities from the binder, e.g. a component of the clays. Surprisingly, it was found by the inventors that metal-free zeolite catalysts comprising acid 10-membered ring channels showed a high selectivity to propane and at the same time, stable performance in the presence of the aromatics presents in the naphtha feedstock. The use of said catalysts in catalyst compositions allows a direct cracking in presence of hydrogen of a naphtha feedstock comprising paraffinic and naphthenic hydrocarbons without deactivation of the catalyst.
[0024] It is noted that U.S. Pat. No. 4,061,690 discloses a method of catalytic conversion of a butane cut to propane by means of a catalyst consisting of acid mordenite in the presence of hydrogen, with a partial hydrogen pressure higher than 0.5 MPa. The results show that the use of a large pore mordenite exchanged by protons (Zeolons H) at 400.degree. C. and at a pressure of 3 MPa had a propane yield of 75.0 wt % for a rate of conversion of 92%, which corresponds to a propane selectivity of approximately 69%. The weighted analysis of the catalyst was SI: 40.6%; Al 6.2%; Na: 0.2%. The feedstock comprised a mixture of n-butane (60 wt %) and of iso-butane (40 wt %). These results were promising, however, it was found that MOR catalysts, having 12-membered ring channels, deactivate in the presence heavier feedstock with the number of atoms sufficient to form aromatics or even containing the aromatics (the naphtha feedstock). Thus, it was not possible to use MOR catalysts in direct hydrocracking of naphtha (DCN). By way of examples, it was shown that MOR deactivates very fast naphtha feedstock and is not suitable.
[0025] With preference, one or more of the following embodiments can be used to better define the inventive process: [0026] The naphtha feedstock comprises C4-C10 hydrocarbons with a potential presence of the aromatics. The feedstock contains preferably no olefins i.e. an olefin content of less than 0.1 wt % relating to the total weight of said naphtha feedstock. The C5+ hydrocarbons lead to a significant deactivation of the 12-members ring zeolites catalysts. The small pore erionite could crack only a part of the feedstock. The solutions allow treating the total amount of the feedstock without significant deactivation of catalyst. [0027] The naphtha feedstock comprises at least 10 wt % of naphthenes as based on the total weight of the naphtha feedstock. [0028] The naphtha feedstock comprises at least 2.0 wt % of aromatics of five or more carbon atoms as based on the total weight of the naphtha feedstock, preferably at least 2.2 wt %. [0029] The naphtha feedstock comprises at most 10.0 wt % of aromatics of five or more carbon atoms as based on the total weight of the naphtha feedstock, preferably at most 9.0 wt %. [0030] The catalyst composition is metal-free, containing less than 1000 wt ppm, preferably less than 50 ppm wt, more preferably at most 5 ppm wt of noble metal and less than 1 wt %, preferably less than 0.05 wt % even more preferably below 0.01 wt % of transition metals as based on the total weight of the catalyst composition. The catalyst composition is not sensitive to the presence of sulfur and provides low C1-02 selectivity. [0031] The hydrocracking conditions of the hydrocracking step comprise the naphtha feedstock being contacted with the catalyst composition at a temperature ranging from 200.degree. C. to 600.degree. C., preferably ranging from 250.degree. C. to 550.degree. C., more preferably ranging from 300.degree. C. to 450.degree. C. [0032] The hydrocracking conditions of the hydrocracking step comprise the naphtha feedstock being contacted with the catalyst at a pressure ranging from 1 to 10 MPa, preferably in the range of 2 to 6 MPa, more preferably from 3 to 5 MPa. [0033] The hydrocracking conditions of the hydrocracking step comprise the naphtha feedstock being contacted with the catalyst at a WHSV (feed) of at least 0.1 h.sup.-1, preferably is ranging from 0.1 h.sup.-1 to 10.0 h.sup.-1, more preferably from 0.5 h.sup.-1 to 8.0 h.sup.-1, even more preferably from 1.0 h.sup.-1 to 6.0 h.sup.-1, and most preferably from 1.5 h.sup.-1 to 5.0 h.sup.-1. [0034] In the hydrocracking step, hydrogen is provided to the naphtha feedstock at a molar ratio H.sub.2/Naphtha ranging from 1000:1 to 1:1, preferably from 100:1 to 1:1, more preferably from 20:1 to 1:1. The hydrocracking step is particularly advantaging in that hydrogen with a low purity can preferably be used. That is to say that the hydrogen feed stream can for instance contain up to 5 wt % of methane, preferably up to 10 wt %, even more preferably up to 20 wt % of methane based on the total weight of the hydrogen feed stream. Other impurities may also be present such as sulphur compounds, such as H.sub.2S, at concentration up to 0.1% wt, preferably 1% wt, even more preferably up to 5 wt % based on the total weight of the hydrogen feed stream. [0035] The hydrocracking step is performed in a single reactor.
[0036] With preference, one or more of the following embodiments can be used to better define the catalyst used in the inventive process: [0037] The one or more zeolite catalysts have a Si/Al molar ratio ranging from 10 to 100, preferably from 20 to 80, more preferably from 30 to 60. [0038] At least 50 wt % of said one or more zeolite catalysts comprising an acid 10-membered ring channels are in their hydrogen form as based on the total weight of the zeolites catalysts. [0039] The catalyst composition comprises one or more zeolites catalysts comprising an acid 10-membered ring channels selected from the list comprising ZSM-5, silicalite-1, ZSM-11, silicalite-2, SSZ-46, MCM-68, CIT-1, SSZ-33, ZSM-8, Ferrierite, FU-9, ZSM-35, ZSM-23, ZSM-22, Theta-1, NU-10, ZSM-50, EU-1, ZSM-57, SAPO-11 and ZSM-48 being preferably under their acidic form or H-form. [0040] Optionally, the one or more zeolites catalysts comprising an acid 10-membered ring channels may be modified by phosphorous, steaming, leaching, mesoporization, dealumination or modification with alkali-earth or rare-earth metals. [0041] The preferred zeolite catalyst structure is MFI (H-ZSM-5). [0042] The catalyst composition comprises a binder selected from silica, alpha-alumina, gamma-alumina, clays, alumina phosphates, mullite, zirconia, titania, yttria, silicon nitride, silicon carbide, iron, bronze and stainless steel, glass, and carbon, preferably the binder is alumina.
[0043] In an embodiment, the step of dehydrogenation into propylene is performed at a temperature ranging from 500 to 800.degree. C. and a partial pressure of propylene below one atmosphere and/or the process further comprises a step of collecting hydrogen produced in the step of dehydrogenation into propylene, and a step of recycling said hydrogen back to the hydrocracking reaction zone in order to perform the hydrocracking step.
[0044] In an embodiment, the separation step comprises recovering a C4 paraffins fraction from the effluent of step b) and a step of recycling back the C4 paraffins fraction to the hydrocrakrting reaction zone, and/or the separation step comprises recovering a C5+ hydrocarbon fraction from the effluent of step b) and a step of valorization of said C5+ hydrocarbon fraction as gasoline.
[0045] In an embodiment, the process comprises a step of recovering the unconverted propane after the step of dehydrogenation into propylene and recycling it to the propane dehydrogenation reactor to be submitted to a further step of dehydrogenation into propylene.
[0046] According to a second aspect, the invention provides the use of one or more zeolite catalysts comprising acid 10-membered ring channels in a process as defined according to the first aspect of catalytic conversion by hydrocracking of paraffinic and naphthenic hydrocarbons from a naphtha feedstock to propane, and further dehydrogenation of said propane to propylene, wherein the one or more zeolites catalysts comprising an acid 10-membered ring channels are metal-free.
[0047] With preference, the one or more zeolites catalysts comprising an acid 10-membered ring channels comprise or are zeolites catalysts of the MFI-type, preferably the one or more zeolites catalysts comprising an acid 10-membered ring channels are or comprise H-ZSM-5.
DESCRIPTION OF THE FIGURES
[0048] FIG. 1 illustrates the process according to the invention.
DETAILED DESCRIPTION OF THE INVENTION
[0049] For the purpose of the invention the following definitions are given:
[0050] Naphtha is mainly a mixture of straight-chain, branched and cyclic aliphatic hydrocarbons. Naphtha is generally divided into light naphtha having from 4 to 10 carbon atoms per molecule and heavy naphtha having from 7 to 12 carbons per molecule. Typically, light naphtha contains naphthenes, such as cyclohexane and methyl-cyclopentane, and linear and branched paraffins or alkanes, such as hexane and pentane. Light naphtha typically contains 60% to 99% by weight of paraffins and cycloparaffins.
[0051] The term "metal free" as used herein means that in the course of the preparation of the catalyst composition, no metal is willingly added. It is always possible that some metals (noble or transition) are present as pollution or traces, but it shall be understood that they are present at a very low concentration if not below the detection limit. As a preferred embodiment, a metal-free catalyst composition contains at most 50 ppm wt, more preferably at most 5 ppm wt even more preferably at most 1 ppm wt of noble metal and less than 0.05 wt % even more preferably less than 0.01 wt % of transition metals as based on the total weight of the catalyst composition.
[0052] The term "alkane" or "alkanes" as used herein describes acyclic branched or unbranched hydrocarbons having the general formula C.sub.nH.sub.2n+2, and therefore consisting entirely of hydrogen atoms and saturated carbon atoms; see e.g. IUPAC. Compendium of Chemical Terminology, 2nd ed. (1997). The term "alkanes" accordingly describes unbranched alkanes ("normal-paraffins"or "n-paraffins" or "n-alkanes") and branched alkanes ("iso-paraffins" or "iso-alkanes") but excludes naphthenes (cycloalkanes).
[0053] The term "aromatic hydrocarbons" or "aromatics" relates to cyclically conjugated hydrocarbon with a stability (due to derealization) that is significantly greater than that of a hypothetical localized structure (e.g. Kekule structure). The most common method for determining aromaticity of a given hydrocarbon is the observation of diatropicity in the .sup.1H NMR spectrum.
[0054] The terms "naphthenic hydrocarbons" or "naphthenes" or "cycloalkanes" as used herein describes saturated cyclic hydrocarbons.
[0055] The term "olefin" as used herein relates to an unsaturated hydrocarbon compound containing at least one carbon-carbon double bond. Preferably, the term "olefins" relates to a mixture comprising two or more selected from ethylene, propylene, butadiene, butylene-1, isobutylene, isoprene, and cyclopentadiene.
[0056] The term "LPG" as used herein refers to the well-established acronym for the term "liquefied petroleum gas". LPG, as used herein, generally consists of a blend of C2-C4 hydrocarbons i.e. a mixture of C2, C3, and C4 hydrocarbons.
[0057] One of the petrochemical products which may be produced in the process of the present invention is BTX. The term "BTX" as used herein relates to a mixture of benzene, toluene, and xylenes.
[0058] As used herein, the term "C# hydrocarbons", wherein "#" is a positive integer, is meant to describe all hydrocarbons having # carbon atoms. C# hydrocarbons are sometimes indicated as just "C#". Moreover, the term "C#+ hydrocarbons" is meant to describe all hydrocarbon molecules having # or more carbon atoms. Accordingly, the term "C5+ hydrocarbons" is meant to describe a mixture of hydrocarbons having 5 or more carbon atoms. The term "C5+ alkanes" accordingly relates to alkanes having 5 or more carbon atoms.
[0059] The term "zeolite catalyst" refers to a molecular sieve aluminosilicate material. Reference herein to a zeolite catalyst having acid 10-membered ring channels is to a zeolite or aluminosilicate having 10-membered ring channels in one direction, optionally intersected with 8, 9 or 10-membered ring channels in another direction.
[0060] The terms "comprising", "comprises" and "comprised of" as used herein are synonymous with "including", "includes" or "containing", "contains", and are inclusive or open-ended and do not exclude additional, non-recited members, elements or method steps. The terms "comprising", "comprises" and "comprised of" also include the term "consisting of".
[0061] The recitation of numerical ranges by endpoints includes all integer numbers and, where appropriate, fractions subsumed within that range (e.g. 1 to 5 can include 1, 2, 3, 4 when referring to, for example, a number of elements, and can also include 1.5, 2, 2.75 and 3.80, when referring to, for example, measurements). The recitation of endpoints also includes the recited endpoint values themselves (e.g. from 1.0 to 5.0 includes both 1.0 and 5.0). Any numerical range recited herein is intended to include all sub-ranges subsumed therein.
[0062] The particular features, structures, characteristics or embodiments may be combined in any suitable manner, as would be apparent to a person skilled in the art from this disclosure, in one or more embodiments.
[0063] The process of the invention can be operated at high naphtha feedstock conversion, reducing the need for a further cracking process.
[0064] Reference is made to FIG. 1. The invention the invention provides a process of catalytic conversion by hydrocracking of a paraffinic and naphthenic hydrocarbons from a naphtha feedstock 1 to propylene, wherein the process comprises the following steps: [0065] a) providing a naphtha feedstock 1 containing one or more paraffins comprising 4 to 10 carbon atoms with preferably no olefins i.e. an olefin content of less than 1 wt % preferably less than 0.1 wt % even more preferably less than 0.01 wt % relating to the total weight of said naphtha feedstock; and [0066] b) submitting said naphtha feedstock 1 to a hydrocracking step 3 by contacting said naphtha feedstock 1 with a catalyst composition in the presence of hydrogen in a reaction zone under hydrocracking conditions to produce an effluent 5; [0067] c) submitting the effluent 5 to a separation step 7 to recover propane 15; and d) submitting said propane 15 to a step of dehydrogenation 19 into propylene 23 in a propane dehydrogenation reactor; [0068] e) collecting hydrogen 21 produced in the step of dehydrogenation into propylene 23, and recycling said hydrogen 21 back to the hydrocracking reaction zone in order to perform the hydrocracking step 3 and wherein the catalyst composition of the hydrocracking step 3 comprises one or more zeolite catalysts comprising an acid 10-membered ring channels.
[0069] The effluent 5 resulting from the hydrocracking step 3 can be submitted to a separation step 7, wherein: [0070] The C4 paraffins fraction 11 can be recycled back to the hydrocracking reactor to be submitted to a further hydrocracking step 3. [0071] The C5+ hydrocarbons fraction 17 can be valorized as gasoline or aromatics. Indeed, the C5+ fraction shows a significant improvement in Research Octane Number (RON) as compared to the naphtha feedstock 1. [0072] The hydrogen 9 can be recycled back to the hydrocracking reactor to be used in the hydrocracking step 3. [0073] The C1 and C2 fraction 13 (methane and ethane fraction) can be use as off-gas.
[0074] The process further comprises a step of collecting hydrogen 21 produced in the step of dehydrogenation into propylene 23, and a step of recycling said hydrogen 21 back to the hydrocracking reaction zone in order to perform the hydrocracking step 3.
[0075] With preference, the process comprises a step of recovering the unconverted propane 15 after the step of dehydrogenation 19 into propylene 23 and recycling it to the propane dehydrogenation reactor to be submitted to a further step of dehydrogenation 19 into propylene.
[0076] The naphtha feedstock 1 used in the invention comprises paraffinic and naphthenic hydrocarbons, preferably the naphtha feedstock 1 comprises one or more paraffins comprising 4 to 10 carbon atoms.
[0077] The naphtha feedstock 1 may comprise compounds other than paraffins. Preferably, the naphtha feedstock comprises at least 10 wt % of paraffins comprising 4 to 10 carbon atoms as based on the total weight of the naphtha feedstock, more preferably at least 50 wt %, more preferably at least 60 wt % of paraffins comprising 4 to 10 carbon atoms.
[0078] Preferably, the naphtha feedstock 1 comprises in the range of from 10 wt % to 100 wt % of paraffins comprising 4 to 10 carbon atoms as based on the total weight of the naphtha feedstock, more preferably of from 50 wt % to 99.5 wt %, more preferably of from 60 wt % to 95 wt % of paraffins comprising 4 to 10 carbon atoms.
[0079] The naphtha feedstock 1 may comprise straight run naphtha or naphtha fractions derived from natural gas, natural gas liquids or associated gas. The naphtha feedstock 1 may comprise naphtha fractions derived from pyrolysis gas. The naphtha feedstock 1 may also comprise naphthas or naphtha fractions obtained from a Fischer-Tropsch process for synthesising hydrocarbons from hydrogen and carbon monoxide. For example, the naphtha feedstock 1 is or comprises desalted crude oil.
[0080] The naphtha feedstock 1 may also comprise higher paraffins, i.e. paraffins comprising more than 10 carbon atoms. Cracking such higher paraffins typically requires the use of temperatures and pressures which are at the higher end of the preferred temperature and pressure ranges.
[0081] Preferably, the naphtha feedstock 1 comprises at least 10% of naphthenes. More preferably, the naphtha feedstock 1 comprises in the range of from 10 to 40 wt %, more preferably of from 50 to 90 wt % of naphthenes and paraffins C6+, based on the total weight of the naphtha feedstock.
[0082] The naphtha feedstock 1 may comprise olefins. However, as the olefins are hydrogenated during the hydrocracking process, the presence of olefins results in an undesired increased hydrogen consumption. Preferably, the naphtha feedstock 1 comprises in the range of from 0 to 20 wt % of olefins, based on the total weight of the naphtha feedstock, more preferably of from 0 to 10 wt % of olefins. Optionally, the naphtha feedstock 1 is subjected to a hydrogenation treatment prior to being supplied to a process according to the present invention.
[0083] In an embodiment, the naphtha feedstock 1 comprises at least 2.0 wt % of aromatics of five or more carbon atoms as based on the total weight of the naphtha feedstock, preferably at least 2.2 wt %, more preferably at least 2.5 wt %.
[0084] In an embodiment, the naphtha feedstock 1 comprises at most 10.0 wt % of aromatics of five or more carbon atoms as based on the total weight of the naphtha feedstock, preferably at most 9.0 wt %.
[0085] In a preferred embodiment, the hydrocracking step 3 is performed in a single reactor. Indeed, the invention provides a one-stage hydrocracking process.
[0086] The one or more zeolites catalysts having acid 10-membered ring channels that can be used for the invention can be selected from: [0087] one-dimensional zeolites catalysts having 10-membered ring channels in one direction, which are not intersected by others channels from another direction; [0088] three-dimensional zeolites catalysts having intersecting channels in at least two directions, whereby the channels in one direction are 10-membered ring channels, intersected by 8, 9 or 10-membered ring channels in another direction.
[0089] Examples of zeolites catalysts having acid 10-membered ring channels suitable for the process of the invention can be of, but not limited to, the MFI-type, the MEL-type, the MSE-type, the CON-type, the ZSM-8-type, the FER-type, the MTT-type, the TON-type, the EUO-type, the MFS-type, the AEL-type and the ZSM-48-type zeolites catalysts. Preferably, the catalyst is or comprises a zeolite of the MFI-type. The zeolite are preferably under their acidic form or H-form MFI-type have a three-dimensional structure, preferably the zeolites catalysts of the MFI-type are selected from ZSM-5, silicalite-1. The preferred MFI-type zeolite is ZSM-5. MEL-type have a three-dimensional structure, preferably the zeolite of the MEL-type is selected from ZSM-11, silicalite-2, and SSZ-46. The preferred zeolite of the MSE-type is MCM-68. The zeolite of the CON-type is selected from CIT-1 and SSZ-33. The zeolite of the FER-type is selected from Ferrierite, FU-9 and ZSM-35. The preferred zeolite of the MTT-type is ZSM-23. The zeolite of the TON-type is selected from ZSM-22, Theta-1 and NU-10. The zeolite of the EUO-type is selected from ZSM-50 and EU-1. The preferred zeolite of the MFS-type is ZSM-57. The preferred zeolite of the AEL-type is SAPO-11. ZSM-48 refers to the family of microporous materials consisting of silicon, aluminium, oxygen and optionally boron. All the zeolite are preferably under their acidic form or H-form
[0090] Preferably, the catalyst composition comprises one or more zeolites catalysts having an acid 10-membered ring channels selected from the list comprising ZSM-5, silicalite-1, ZSM-11, silicalite-2, SSZ-46, MCM-68, CIT-1, SSZ-33, ZSM-8, Ferrierite, FU-9, ZSM-35, ZSM-23, ZSM-22, Theta-1, NU-10, ZSM-50, EU-1, ZSM-57, SAPO-11 and ZSM-48. More preferably the catalyst is or comprises ZSM-5 zeolite. All the zeolite are preferably under their acidic form or H-form
[0091] In a preferred embodiment, the catalyst composition comprises 3D zeolite without cages (cavities) and containing at least one 10-member rings.
[0092] Preferably, the catalyst composition comprises at least 60 wt % of one or more zeolite catalysts having an acid 10-membered ring channels, more preferably at least 70 wt %, even more preferably at least 80 wt % and most preferably at least 90 wt %.
[0093] Several mesoporisation approaches may be used to create zeolites crystals that contain both mesopores and micropores, including destructive approaches such as demetallation (desilication and dealumination) and recrystallization; and constructive approaches such as using hard templates, supramolecular templates, and surface silanization.
[0094] To provide sufficient acidity for the hydrocracking reaction, it is preferred that the zeolites catalysts are at least partly in their hydrogen form, e.g. H-ZSM-5 or H-ZSM-11. Preferably at least 50 wt % of the total amount of the zeolites catalysts used are in their hydrogen form, preferably at least 80 wt %, more preferably at least 90 wt %, and even more preferably 100 wt % of the zeolites catalysts are in their hydrogen form.
[0095] When the zeolites catalysts are prepared in the presence of an organic cation, they may be activated by heating them in an inert or oxidative atmosphere to remove the organic cation. For example, they may be activated at a temperature over 500.degree. C. for at least 1 hour.
[0096] In a preferred embodiment, the one or more zeolite catalysts have a Si/Al molar ratio ranging from 10 to 100, preferably from 20 to 80, more preferably from 30 to 60.
[0097] In an embodiment, the zeolite is shaped with a binder. The binder is an inorganic material.
[0098] Preferably, the binder is selected from silica, alpha-alumina, gamma-alumina, clays, alumina phosphates, mullite, zirconia, titania, yttria, silicon nitride, silicon carbide, iron, bronze and stainless steel, glass, and carbon, preferably the binder is alumina.
[0099] The zeolite shaped with the binder forms a catalyst composition, and the catalyst composition of the present invention preferably comprises at least 10 wt % of a binder as based on the total weight of the catalyst composition, most preferably at least 20 wt % of a binder and preferably comprises up to 40 wt % of a binder.
[0100] Optionally, the zeolite may be modified by phosphorous, steaming, leaching, mesoporization, dealumination or modification with alkali-earth or rare-earth metals.
[0101] In a further aspect, the catalyst composition used in the process of the present invention is prepared by the method comprising the steps of: [0102] steaming acid zeolite catalyst at a temperature between 500.degree. C.-750.degree. C. for a period from 0.1 to 24 h under steam pressure from 0.1 to 10 bars; [0103] optionally, contacting the steamed zeolite with the one or more source of phosphorus; [0104] optionally, introducing to the phosphate sample at least 0.1 wt % of Mg, Ca, Sr, Ba, Ce, La, Fe, Ga; [0105] drying and steaming of the one or more modified acid zeolite catalysts at a temperature between 500.degree. C.-750.degree. C. for a period from 0.1 to 24 h under steam pressure from 0.1 to 10 bars.
[0106] When at least 0.1 wt % of Mg, Ca, Sr, Ba, Ce, La, Fe, Ga is introduced to the phosphate sample, the said metals may be further presented on catalyst in form of oxides, silicates, aluminates or phosphates.
[0107] Accordingly, the one or more acid zeolite catalysts are contacted with a solution in which one or more basic compounds are dissolved, and wherein, with preference, one or more redox elements are dissolved as well. Preferably, the solution is an aqueous solution. The preferred source of phosphorous is the phosphoric acid. The preferred soluble salts of the basic compounds and of the redox elements are nitrate salts. The preferred soluble salts of the basic compounds are selected from the list consisting of Mg(NO.sub.3).sub.2, Ca(NO.sub.3).sub.2, Sr(NO.sub.3).sub.2, La(NO.sub.3).sub.3, Ga(NO.sub.3).sub.3, Fe(NO.sub.3).sub.3.
[0108] In an embodiment, the phosphorous modified acid zeolite catalyst is preferably obtained by the process described in WO2009/016156, which is incorporated herein by reference. The process comprises the following steps in this order: [0109] selecting a zeolite with low Si/Al molar ratio (advantageously lower than 30) among H.sup.+ or NH.sub.4.sup.+-form of MFI, MEL, FER, MOR, clinoptilolite, said zeolite having been made preferably without direct addition of organic template; [0110] steaming at a temperature ranging from 400 to 870.degree. C. for 0.01 to 200 h; [0111] leaching with an aqueous acid solution containing the source of P at conditions effective to remove a substantial part of Al from the zeolite and to introduce at least 0.3 wt % of P; [0112] separation of the solid from the liquid; [0113] an optional washing step or an optional drying step or an optional drying step followed by a washing step; [0114] a calcination step.
[0115] The basic compounds and the optional redox element(s) and phosphorus (P), may be deposited by contacting the one or more acid zeolite catalysts with a single solution in which the soluble salts of the basic compounds, soluble salts of the redox elements and phosphoric acid are dissolved.
[0116] Alternatively, the basic compounds and the optional redox element(s) and phosphorus (P) may be deposited by subsequently contacting the one or more acid zeolite catalysts with the different elements and/or phosphorus, whereby the composition is dried to evaporate the solvent before contacting the composition with the following element. After depositing all the required elements, the resulting composition (catalyst precursor) is dried.
[0117] In one embodiment of the present invention, the catalyst precursor is air-dried, preferably for about 8 hours at a temperature ranging from 60.degree. C. to 80.degree. C. while stirring.
[0118] After drying, the zeolite-comprising composition, on which the basic compounds and the optional redox element(s) and the phosphorus (P) are deposited, is calcined in an oxygen-comprising atmosphere, preferably in a moisture-free atmospheric air. Preferably, the catalyst precursor is calcined at a temperature ranging from 450.degree. C. to 550.degree. C. to remove the residual amount of nitrates and carbons.
[0119] Most preferably, the catalyst precursor is calcined at about 500.degree. C. for about 4 hours. When a binder is present, it is preferred that the one or more acid zeolite catalysts are mixed with the binder prior to contacting the one or more acid zeolite catalysts with one or more solutions comprising soluble salts of basic compounds and the optional soluble salts of redox elements and phosphoric acid.
[0120] In the process of the invention, the naphtha feedstock is contacted with the catalyst composition at elevated temperatures and elevated pressures in hydrocracking conditions to perform a hydrocracking step.
[0121] In an embodiment, the hydrocracking conditions comprise the naphtha feedstock being contacted with the catalyst composition at a temperature ranging from 200.degree. C. to 600.degree. C., preferably ranging from 250.degree. C. to 550.degree. C., more preferably ranging from 300.degree. C. to 450.degree. C. Preferably, the hydrocracking conditions comprise the naphtha feedstock being contacted with the catalyst composition at a pressure ranging from 1 to 10 MPa, preferably in the range of 2 to 6 MPa, more preferably from 3 to 5 MPa.
[0122] In an embodiment, the hydrocracking conditions comprise the naphtha feedstock being contacted with the catalyst composition at a weight hourly space velocity of the naphtha feedstock (VVHSV) of at least 0.1 h.sup.-1, preferably is ranging from 0.1 h.sup.-1 to 10.0 h.sup.-1, more preferably from 0.5 h.sup.-1 to 8.0 h.sup.-1, even more preferably from 1.0 h.sup.-1 to 6.0 h.sup.-1, and most preferably from 1.5 h.sup.-1 to 5.0 h.sup.-1.
[0123] Hydrogen may be provided at any suitable ratio to the paraffins contained in the naphtha feedstock. Preferably, the hydrogen is provided in a molar ratio hydrogen to the paraffins in the naphtha feedstock ranging from 1000:1 to 1:1, more preferably from 100:1 to 1:1, even more preferably from 20:1 to 1:1; wherein the number of moles of the paraffins in the naphtha feedstock is based on the average molecular weight of the naphtha feedstock. The process according to the invention is to achieve a set conversion of the hydrocarbon naphtha feedstock. Preferably, the ratio of hydrogen to the paraffins in the naphtha feedstock is chosen such that the process conditions enable to achieve the desired conversion.
[0124] PDH is a catalytic dehydrogenation process that converts paraffins (in this case propane) to their corresponding light olefins (propylene) dating back from the 1930s. In the late 1980s, the chromia-alumina catalyst was specifically applied in the dehydrogenation of propane to propylene. The conversion process is favored under high temperature and low partial pressure of propane. The reaction is run optimally at 500.degree. C.-700.degree. C. to minimize thermal cracking, while the reaction pressure is typically atmospheric. The product mixture goes through a deethaniser to remove light hydrocarbons and traces of hydrogen. The last separation step involves a propane-propylene splitter to achieve polymer-grade propylene. It features high yields of 90 wt % propylene, low generation of by-products and relatively low investment and operating costs compared to steam cracking. Lummus CATOFIN and UOP OLEFLEX are the most commonly licensed and proven technology. Less common, at the global level, are the ThyssenKrupp Udhe STAR process and Linde/BASF's PDH technology. Recently, Dow Chemicals introduced a new fluidized bed propane dehydrogenation technology to the market.
[0125] The catalytic dehydrogenation of propane is an endothermic reaction, which produces propane and hydrogen. Hydrogen can be reused for naphtha cracking. The extent of conversion is limited by the thermodynamics of the reaction with higher temperature favouring higher conversion. The selectivity decreases as the conversion increases.
[0126] In U.S. Pat. No. 6,392,113 B1 assigned to ABB Lummus, the performance and economics of a catalytic dehydrogenation process are improved by using two pre-reactors before two main dehydrogenation reactors (one in operation and the other in regeneration). In this process, propane is preheated and then passed to the pre-reactor, wherein the catalyst bed is not heated, and propane is partially dehydrogenated with conversion at about 10-25% with the effluent leaves the reactor at 100.degree. C. The pre-reactor can be operated for hours before it is subjected to regeneration. The partially dehydrogenated reactor effluent is then reheated by passing through a fired unit, where the heat is supplied by a portion of the effluent air from the regeneration of the main dehydrogenation reactor. The remainder of the effluent air is used to regenerate the catalyst in the pre-reactor. The heated effluent from the pre-reactor is then dehydrogenated in the main dehydrogenation reactor. The operation of the pre-reactor is comparatively steady than that of the main reactor, which permits an extended cycle of about 24 hours before any catalyst regeneration is required. In WO95/23123, the overall performance of the dehydrogenation process can be improved with respect to the dehydrogenation cycle and heating cycle by the change from co-current to counter-current flow through the catalyst bed. In the process, the regeneration gas is introduced at the opposite of the reactor from the feed hydrocarbon. This provides the highest temperature at the end of the reactor, thus creating the most favourable conditions for the dehydrogenation reaction with respect to equilibrium. The reaction is carried at 590.degree. C. and 0.05 MPa in the presence of chromium catalyst supported on alumina with a conversion of propane at 47% and selectivity to propylene at 87%.
[0127] In the UOP Oleflex process, the dehydrogenation reaction is carried out at about 600.degree. C. and 0.1 MPa in four sequential moving-bed reactors with interheaters to reheat the reactor effluent to the desired reaction temperature before passing it to the next catalyst bed (U.S. Pat. No. 5,321,192). Hydrogen and other inert compounds may be added to the feed stream to the dehydrogenation reactor. The catalyst comprises 0.7-0.75 wt % Pt, 0.5 wt % Sn, and 3.5-4.4 wt % alkali metal supported on .gamma.-alumina (EP 0448858 B1, U.S. Pat. No. 5,457,256). It passes through annular bed via gravity flow (U.S. Pat. Nos. 5,227,567, 5,177,293). The reactor effluent is compressed and dried before it passes to a cryogenic operation system (a cold box) where hydrogen-methane gas is removed from C2+ hydrocarbon compounds. The gas is subjected to adsorption with an adsorbent, such as alumina. Silica gel, active carbon, or molecular sieves are used to remove methane from the hydrogen gas (U.S. Pat. No. 5,457,256). In U.S. Pat. No. 6,293,999 assigned to UOP, a pressure swing adsorption (PSA) process is used for the separation of a hydrocarbon feed gas comprising propylene and propane into a fraction, which comprises predominantly propylene and a fraction comprising propane. The process uses a small pore aluminophosphate molecular sieve, ALPO-14, to selectively adsorb propylene while essentially excluding propane at an adsorption temperature of 70-120.degree. C. and at a propylene partial pressure of 0.05-0.2 MPa. The desorption conditions are conducted under a temperature of 70-120.degree. C. and a propylene partial pressure of 0.001 and 0.05 MPa. Overall, the process comprises the adsorption step, the series of connected co-purge steps, and the counter-current depressurization and repressurization step. In another UOP patent (U.S. Pat. No. 6,218,589), a selective hydrogenation is employed to treat a mixture of the reactor effluent from a dehydrogenation and a recycle stream from a downstream propane-propylene splitter, and to convert the majority of highly unsaturated impurities, such as methylacetylene and propadiene, to propane. The effluent from the selective hydrogenation reactor is sent to a deethanizer before being purified within a propylene-propane splitter.
[0128] In a preferred embodiment the step 19 of dehydrogenation of propane 15 to propylene 23 (i.e. the PDH step) is performed with a catalyst selected from: [0129] 0.7-0.75 wt % Pt, 0.5 wt % Sn, and 3.5-4.4 wt % alkali metal supported on .gamma.-alumina; [0130] Cr.sub.2O.sub.3/Al.sub.2O.sub.3; [0131] Pt--Ga/Al.sub.2O.sub.3.
[0132] In a preferred embodiment, the PDH step 19 is performed at a temperature ranging from 500 to 800.degree. C. and a partial pressure of propylene below one atmosphere.
[0133] In a preferred embodiment, the PDH step is performed as described in US2010/0236985. In an embodiment, the catalyst used in the dehydrogenation step of propane to propylene comprises: [0134] i. from 0.25 to 5.0 wt %, preferably 0.3 to 3.0 wt % of the first component, preferably gallium, or a compound thereof; [0135] ii. from 0.0005 to 0.05 wt %, preferably 0.0007 to 0.04 wt % of the second component, preferably platinum, or a compound thereof; [0136] iii. from 0.0 to 2.0 wt %, preferably 0.1 to 1.0 wt % of an alkali metal or alkaline earth metal, preferably potassium; and [0137] iv. a support comprising alumina in the gamma crystalline form.
[0138] Methods
[0139] Gas chromatography was performed on Columns: DB1 (40 m, 0.1 mm, 0.4 .mu.m) and Al.sub.2O.sub.3 (50 m, 0.32 mm, 5 .mu.m) using Agilent operated by ChemStation software.
[0140] The elemental composition (i.e. the metal content) of catalyst composition can preferably be determined by ICP-OES according for instance to the method UOP Method 961-12. The platinum content can preferably be determined according to the method ASTM D4642.
EXAMPLES
[0141] The following examples illustrate the invention.
Example 1
[0142] The process was conducted in a fixed bed reactor loaded with a ZSM-5 (Si/Al-40, CBV8014 from Zeolyst INT) containing catalyst extruded with an Al.sub.2O.sub.3 binder (80 wt % zeolite, 20 wt % Al.sub.2O.sub.3) in form of cylinders 1.6 mm. After the extrusion, the catalyst composition was dried at room temperature for 24 hours followed by calcination at 550.degree. C. for 6 h. The demonstration of the invention was performed in both micro pilots.
[0143] A stainless-steel reactor tube having an internal diameter of 10 mm is used. 10 ml of the catalyst composition, as pellets of 35-45 mesh, is loaded in the tubular reactor. The void spaces, before and after the catalyst composition, are filled with SiC granulates of 2 mm. The temperature profile is monitored with the aid of a thermocouple placed inside the reactor at the top of the catalyst bed. Prior to the reaction, the catalyst composition was pretreated with an hydrogen flow at 400.degree. C. for 6 h (heating rate 60.degree. C./h) followed by cooling down to the reaction temperature. The performance test is performed down-flow at 4 MPa of total pressure, molar H.sub.2/Naphtha of 13.2; WHSV (naphtha) of 2.5 h.sup.-1, temperature of 400.degree. C. Analysis of the products is performed by using an on-line gas chromatography. The results are provided in tables 1 to 3.
[0144] Feedstock Characteristics
TABLE-US-00001 TABLE 1 Naphtha feedstock, POINA analysis nPar iPar Napht Arom Total 36.38 38.06 23.28 2.29 C1 0 0 0 0 C2 0 0 0 0 C3 0 0 0 0 C4 8.1 0.58 0 0 C5 15.51 14.38 3.9 0 C6 11.43 17.75 14.97 1.98 C7 1.34 5.35 4.41 0.3 C8 0 0 0 0 C9 0 0 0 0 C10 0 0 0 0 C11 0 0 0 0 >200 C. 0 0 0 0 >200 C. 0 0 0 0 >200 C. 0 0 0 0 P + A
TABLE-US-00002 TABLE 2 The distillation cut (according to ASTM D 86) is given in below: DIST_86: Distillation ASTM D86 T.degree. C. at IBP 28.9.degree. C. DIST_86: T.degree. C. at 5% vol 38.1.degree. C. DIST_86: T.degree. C. at 10% vol 40.3.degree. C. DIST_86: T.degree. C. at 20% vol 44.2.degree. C. DIST_86: T.degree. C. at 30% vol 48.4.degree. C. DIST_86: T.degree. C. at 40% vol 52.6.degree. C. DIST_86: T.degree. C. at 50% vol 57.1.degree. C. DIST_86: T.degree. C. at 60% vol 61.6.degree. C. DIST_86: T.degree. C. at 70% vol 66.6.degree. C. DIST_86: T.degree. C. at 80% vol 72.1.degree. C. DIST_86: T.degree. C. at 90% vol 79.4.degree. C. DIST_86: T.degree. C. at 95% vol 89.9.degree. C. DIST_86: T.degree. C. at FBP 91.3.degree. C. DIST_86: % Recovered at 76.2% vol 70.degree. C. (*) DIST_86: Recovered % vol 97.0% vol DIST_86: Residue % vol 0.5% vol DIST_86: Loss % vol 2.5% vol DIST_86: (*) % Loss included N Yes/No
[0145] The density was determined according to ISO 12185 at 15.degree. C. and was found to be 0.6702 g/ml.
TABLE-US-00003 TABLE 3 FEED EFFLUENT Effluent with C4 (in wt %) (in wt %) recycling (in wt %) Paraffins 74.4 92.8 91.4 Cy-Paraffins 23.3 0.3 0.1 Olefins 0.00 0.28 0.4 Aromatics 2.3 6.6 8.1 Breakdown Methane 0 1.7 2.3 Ethane 0 4.7 6.3 Propane 0 38.5 51.6 i-Butane 0.58 13.0 0.2 n-Butane 8.1 12.1 0.2 C5+ 91.3 30.0 37.1
[0146] The process allows valorizing about 50% naphtha as propane and about 37 wt % as gasoline, which shows a very high carbon efficiency.
[0147] Taking into account an overall propylene yield in propane dehydrogenation of about 82%, the process allows converting of about 41 wt % of naphtha to propylene as opposed to 17 wt % in steam cracking. This offers about 2.4 times higher propylene yield from the same feedstock.
TABLE-US-00004 TABLE 4 RON of the C5+ fractions FEED Effluent RON 74 (71-77) 92 (89-93) Aromatic content, wt % 2.5 21.8 Olefins content, wt % <0.1 <1.0
[0148] The C5+ produced in the reaction demonstrates very valuable properties for the use of gasoline.
[0149] The catalyst composition was two times regenerated in the air at 550.degree. C. and demonstrated full recovering of the activity.
[0150] The process shows a low H.sub.2 consumption. Indeed, the naphtha feedstock contained 15.7 wt % of hydrogen as based on the total weight of the feed, whereas the weight content of hydrogen in the effluent was 16.4 wt %.
Example 2
[0151] The experiment was repeated with compositions comprising zeolites catalysts comprising 12-membered ring channels. They showed fast deactivation and significantly lower yield of propane, as shown by the results given in table 5.
[0152] Conditions: pressure: 4 MPa [0153] Molar H.sub.2/Naphtha: 13.2 [0154] WHSV (naphtha): 2.5 h-1 [0155] Temperature: 450.degree. C.
TABLE-US-00005 [0155] TABLE 5 FEED Composition Catalyst of the feed MOR ZSM-5 BETA ZSM-12 SI/AI 10 40 40 40 Structure 1D 3D 3D 1D Members ring 8, 12 10 12 12 Composition of the effluent Paraffins 84 78 92.5 79 83 Cy-paraffins 13.23 1.15 0.05 6.76 0.30 Olefins 0.00 8.61 0.92 2.69 0.93 aromatics 2.9 11.8 6.5 11.2 16.0 Conversion to C5- Total -- 59.8 83.9 15.6 60.0 Propane 0.0 27.4 43.9 5.8 32.0 Ethane 0.0 7.4 12.7 0.6 3.4 Iso-butane 0.1 11.8 11.6 5.3 12.4 N-butane 2.3 7.6 9.0 2.8 9.4 Conversion to C5+ Total 100 40.2 16.1 84.4 40.0 % aromatics 2.9 26.9 40.6 13.3 39.1 in C5+ BTX (in arom) 100 81.3 89.0 75.9 76.5
[0156] Many zeolites catalysts deactivate in the presence of aromatics in the feed. ZSM-5 shows stable performances.
[0157] The results demonstrate a higher selectivity for propane than for ethane for all the zeolite catalysts tested. However, best results were achieved with ZSM-5.
* * * * *
D00000

D00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.