Novel Substituted 6,7-dihydro-5h-benzo[7]annulene Compounds, Processes For Their Preparation And Therapeutic Uses Thereof
BOUABOULA; Monsif ; et al.
U.S. patent application number 16/743504 was filed with the patent office on 2020-12-17 for novel substituted 6,7-dihydro-5h-benzo[7]annulene compounds, processes for their preparation and therapeutic uses thereof. The applicant listed for this patent is SANOFI. Invention is credited to Monsif BOUABOULA, Maurice BROLLO, Victor CERTAL, Youssef EL-AHMAD, Bruno FILOCHE-ROMME, Frank HALLEY, Gary MCCORT, Laurent SCHIO, Michel TABART, Corinne TERRIER, Fabienne THOMPSON.
| Application Number | 20200392081 16/743504 |
| Document ID | / |
| Family ID | 1000005051477 |
| Filed Date | 2020-12-17 |








View All Diagrams
| United States Patent Application | 20200392081 |
| Kind Code | A1 |
| BOUABOULA; Monsif ; et al. | December 17, 2020 |
NOVEL SUBSTITUTED 6,7-DIHYDRO-5H-BENZO[7]ANNULENE COMPOUNDS, PROCESSES FOR THEIR PREPARATION AND THERAPEUTIC USES THEREOF
Abstract
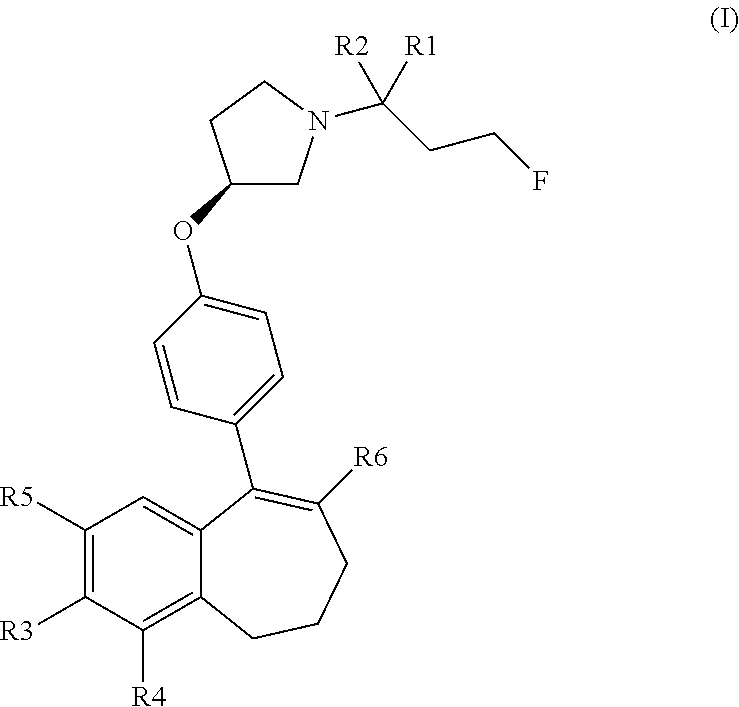
Compounds of formula (I): ##STR00001## wherein R1 and R2 represent hydrogen or deuterium atoms; R3 represents a hydrogen atom or a --COOH, a --OH or a --OPO(OH).sub.2 group; R4 represents a hydrogen atom or a fluorine atom; R5 represents a hydrogen atom or a --OH group; wherein at least one of R3 or R5 is different from a hydrogen atom; when R3 represents a --COOH, --OH or --OPO(OH).sub.2 group, then R5 represents a hydrogen atom; when R5 represents a --OH group, then R3 and R4 represent hydrogen atoms; and R6 is selected from an optionally substituted phenyl, heteroaryl, cycloalkyl and heterocycloalkyl group; and the preparation and the therapeutic uses of the compounds of formula (I) as inhibitors and degraders of estrogen receptors, useful especially in the treatment of cancer.
| Inventors: | BOUABOULA; Monsif; (Paris, FR) ; BROLLO; Maurice; (Paris, FR) ; CERTAL; Victor; (Paris, FR) ; EL-AHMAD; Youssef; (Paris, FR) ; FILOCHE-ROMME; Bruno; (Paris, FR) ; HALLEY; Frank; (Paris, FR) ; MCCORT; Gary; (Paris, FR) ; SCHIO; Laurent; (Paris, FR) ; TABART; Michel; (Paris, FR) ; TERRIER; Corinne; (Paris, FR) ; THOMPSON; Fabienne; (Paris, FR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005051477 | ||||||||||
| Appl. No.: | 16/743504 | ||||||||||
| Filed: | January 15, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15613689 | Jun 5, 2017 | 10570090 | ||
| 16743504 | ||||
| 15432470 | Feb 14, 2017 | 9714221 | ||
| 15613689 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07D 413/12 20130101; C07D 403/12 20130101; C07D 207/12 20130101; C07D 471/04 20130101; C07F 5/025 20130101; C07D 405/12 20130101; C07D 417/12 20130101; C07F 7/0812 20130101; C07D 401/12 20130101 |
| International Class: | C07D 207/12 20060101 C07D207/12; C07D 403/12 20060101 C07D403/12; C07D 401/12 20060101 C07D401/12; C07D 471/04 20060101 C07D471/04; C07D 405/12 20060101 C07D405/12; C07D 413/12 20060101 C07D413/12; C07F 7/08 20060101 C07F007/08; C07D 417/12 20060101 C07D417/12; C07F 5/02 20060101 C07F005/02 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Feb 15, 2016 | EP | 16305174.1 |
Claims
1.-10. (canceled)
11. A process for preparing a compound of formula (I): ##STR00338## wherein: R1 and R2 represent independently a hydrogen atom or a deuterium atom; R3 represents a hydrogen atom, a --COOH group, a --OH group, or a --OPO(OH).sub.2 group; R4 represents a hydrogen atom or a fluorine atom; R5 represents a hydrogen atom or a --OH group; wherein: at least one of R3 or R5 is different from a hydrogen atom; when R3 represents a --COOH group, a --OH group or a --OPO(OH).sub.2 group, then R5 represents a hydrogen atom; and when R5 represents a --OH group, then R3 and R4 represent hydrogen atoms; R6 is selected from: a phenyl group or a heteroaryl group comprising 3 to 9 carbon atoms and comprising from 1 to 3 heteroatoms independently selected from oxygen, nitrogen and sulphur, said phenyl and heteroaryl groups being unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group unsubstituted or substituted with one or more fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.6)-alkoxy group unsubstituted or substituted with one or more fluorine atoms; a cyano group; a sulphur group substituted with 5 fluorine atoms or (C.sub.1-C.sub.6)-alkyl groups substituted with two or more fluorine atoms; a sulfonyl-(C.sub.1-C.sub.6)-alkyl group wherein said (C.sub.1-C.sub.6)-alkyl group is unsubstituted or substituted with two or more fluorine atoms; a silane group substituted with 3 (C.sub.1-C.sub.6)-alkyl groups; an amine group unsubstituted or substituted with one or more (C.sub.1-C.sub.6)-alkyl groups; an amide group unsubstituted or substituted with one or more (C.sub.1-C.sub.6)-alkyl groups; a heterocycloalkyl group saturated or partially saturated, comprising 3 to 5 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur; and a heteroaryl group comprising 2 to 4 carbon atoms and comprising 1 to 3 heteroatoms selected from oxygen, nitrogen and sulphur and being unsubstituted or substituted with an oxo group; and a cycloalkyl group or a heterocycloalkyl group comprising 4 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said cycloalkyl or heterocycloalkyl groups being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: a fluorine atom; a --OH group; a (C.sub.1-C.sub.6)-alkyl group; a --COOR7 group wherein R7 is an (C.sub.1-C.sub.6)-alkyl group; and an oxo group or a pharmaceutically acceptable salt thereof; comprising submitting a compound of formula (D) below ##STR00339## wherein R1, R2, R3, R4 and R5 are as defined above for the compound of formula I, and wherein R3 and R5 are optionally in protected form, to a Suzuki coupling with a boronic reagent of Formula R6B(OR').sub.2 wherein (--B(OR').sub.2) is a boronic acid or a pinacolate ester and wherein R6 is as defined above for the compound of formula I; and optionally deprotecting a protected R3 and/or R5 group.
12. A compound selected from the group consisting of the following formulae: ##STR00340## ##STR00341## ##STR00342## wherein: R1 and R2 represent independently a hydrogen atom or a deuterium atom; R3 represents a hydrogen atom, a --COOH group, a --OH group, or a --OPO(OH).sub.2 group; R4 represents a hydrogen atom or a fluorine atom; R5 represents a hydrogen atom or a --OH group; wherein: at least one of R3 or R5 is different from a hydrogen atom; when R3 represents a --COOH group, a --OH group or a --OPO(OH).sub.2 group, then R5 represents a hydrogen atom; and when R5 represents a --OH group, then R3 and R4 represent hydrogen atoms; R6 is selected from: a phenyl group or a heteroaryl group comprising 3 to 9 carbon atoms and comprising from 1 to 3 heteroatoms independently selected from oxygen, nitrogen and sulphur, said phenyl and heteroaryl groups being unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group unsubstituted or substituted with one or more fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.6)-alkoxy group unsubstituted or substituted with one or more fluorine atoms; a cyano group; a sulphur group substituted with 5 fluorine atoms or (C.sub.1-C.sub.6)-alkyl groups substituted with two or more fluorine atoms; a sulfonyl-(C.sub.1-C.sub.6)-alkyl group wherein said (C.sub.1-C.sub.6)-alkyl group is unsubstituted or substituted with two or more fluorine atoms; a silane group substituted with 3 (C.sub.1-C.sub.6)-alkyl groups; an amine group unsubstituted or substituted with one or more (C.sub.1-C.sub.6)-alkyl groups; an amide group unsubstituted or substituted with one or more (C.sub.1-C.sub.6)-alkyl groups; a heterocycloalkyl group saturated or partially saturated, comprising 3 to 5 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur; and a heteroaryl group comprising 2 to 4 carbon atoms and comprising 1 to 3 heteroatoms selected from oxygen, nitrogen and sulphur and being unsubstituted or substituted with an oxo group; and a cycloalkyl group or a heterocycloalkyl group comprising 4 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said cycloalkyl or heterocycloalkyl groups being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: a fluorine atom; a --OH group; a (C.sub.1-C.sub.6)-alkyl group; a --COOR7 group wherein R7 is an (C.sub.1-C.sub.6)-alkyl group; and an oxo group; and when R3 represents a --COOH or a --OH group or when R5 represents a --OH group, said R3 or R5 group is optionally protected.
13.-27. (canceled)
28. The process according to claim 11, wherein when R3 represents a protected --COOH group, said group is protected as a methyl ester and when R3 or R5 represents a protected --OH group, said R3 or R5 group is protected as a pivaloyl ester.
29. The process according to claim 11, further comprising a step of brominating a compound for Formula (C): ##STR00343## to obtain a compound of Formula D, wherein R1, R2, R3, R4 and R5 are as defined in claim 11 for the compound of formula D.
30. The process according to claim 29, wherein when R3 represents a protected --COOH group, said group is protected as a methyl ester and when R3 or R5 represents a protected --OH group, said R3 or R5 group is protected as a pivaloyl ester.
31. The process according to claim 29, comprising a Suzuki coupling of a compound of formula (B): ##STR00344## with a reagent of formula (1): ##STR00345## to give the compound of formula (C), wherein R1, R2, R3, R4 and R5 are as defined in claim 29 and when R3 represents a --COOH or a --OH group or when R5 represents a --OH group, said R3 or R5 group is protected.
32. The process according to claim 29, wherein when R3 represents a protected --COOH group, said group is protected as a methyl ester and when R3 or R5 represents a protected --OH group, said R3 or R5 group is protected as a pivaloyl ester.
33. The process according to claim 11 for preparing a compound of formula 51 below or a pharmaceutically acceptable salt thereof, ##STR00346## wherein the compound of formula (D) is the compound of formula (D5): ##STR00347##
34. The process according to claim 33, comprising brominating a compound of formula (C3) below: ##STR00348## to obtain a compound of formula (D5) as defined in claim 33.
35. The process according to claim 11, comprising deprotecting a compound of formula (Ib): ##STR00349## wherein R1, R2, R4 and R6 are as defined in claim 11 for a compound of formula (I).
36. The process according to claim 35, wherein the compound of formula (Ib) is 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-- yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid methyl ester of the formula: ##STR00350##
37. The process according to claim 11, comprising deprotecting a compound of formula: ##STR00351## to obtain 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid.
38. A process for preparing a compound of formula 51 as defined in claim 33, comprising deprotecting 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid methyl ester of the formula: ##STR00352##
38. The compound according to claim 12, selected from the following formulae: ##STR00353## wherein R1, R2, R3, R4 and R5 are as defined in claim 12, wherein when R3 represents a --COOH or a --OH group or when R5 represents a --OH group, said R3 or R5 group is protected.
39. The compound according to claim 38 wherein when R3 represents a protected --COOH group, said group is protected as a methyl ester and when R3 or R5 represents a protected --OH group, said R3 or R5 group is protected as a pivaloyl ester.
40. The compound according to claim 39, wherein the compound is selected from the group consisting of the compound of formula (C3) and the compound of formula (D5): ##STR00354##
41. The compound of formula (1) according to claim 12, which is: ##STR00355##
Description
[0001] Provided herein are novel substituted 6,7-dihydro-5H-benzo[7]annulene compounds, the processes for their preparation, as well as the therapeutic uses thereof, in particular as anticancer agents via selective antagonism and degradation of estrogen receptors.
[0002] The Estrogen Receptors (ER) belong to the steroid/nuclear receptor superfamily involved in the regulation of eukaryotic gene expression, cellular proliferation and differentiation in target tissues. ERs are in two forms: the estrogen receptor alpha (ER.alpha.) and the estrogen receptor beta (ER.beta.) respectively encoded by the ESR1 and the ESR2 genes. ER.alpha. and ER.beta. are ligand-activated transcription factors which are activated by the hormone estrogen (the most potent estrogen produced in the body is 17.beta.-estradiol). In the absence of hormone, ERs are largely located in the cytosol of the cell. When the hormone estrogen binds to ERs, ERs migrate from the cytosol to the nucleus of the cell, form dimers and then bind to specific genomic sequences called Estrogen Response Elements (ERE). The DNA/ER complex interacts with co-regulators to modulate the transcription of target genes.
[0003] ER.alpha. is mainly expressed in reproductive tissues such as uterus, ovary, breast, bone and white adipose tissue. Abnormal ER.alpha. signaling leads to development of a variety of diseases, such as cancers, metabolic and cardiovascular diseases, neurodegenerative diseases, inflammation diseases and osteoporosis.
[0004] ER.alpha. is expressed in not more than 10% of normal breast epithelium but approximately 50-80% of breast tumors. Such breast tumors with high level of ER.alpha. are classified as ER.alpha.-positive breast tumors. The etiological role of estrogen in breast cancer is well established and modulation of ER.alpha. signaling remains the mainstay of breast cancer treatment for the majority ER.alpha.-positive breast tumors. Currently, several strategies for inhibiting the estrogen axis in breast cancer exist, including: 1-blocking estrogen synthesis by aromatase inhibitors that are used to treat early and advanced ER.alpha.-positive breast cancer patients; 2-antagonizing estrogen ligand binding to ER.alpha. by tamoxifen which is used to treat ER.alpha.-positive breast cancer patients in both pre- and post-menopausal setting; 3-antagonizing and downregulating ER.alpha. levels by fulvestrant, which is used to treat breast cancer in patients that have progressed despite endocrine therapies such as tamoxifen or aromatase inhibitors.
[0005] Although these endocrine therapies have contributed enormously to reduction in breast cancer development, about more than one-third of ER.alpha.-positive patients display de-novo resistance or develop resistance over time to such existing therapies. Several mechanisms have been described to explain resistance to such hormone therapies. For example, hypersensitivity of ER.alpha. to low estrogen level in treatment with aromatase inhibitors, the switch of tamoxifen effects from antagonist to agonist effects in tamoxifen treatments or multiple growth factor receptor signaling pathways. More recently, acquired mutations in ER.alpha. occurring after initiation of hormone therapies may play a role in treatment failure and cancer progression. Certain mutations in ER.alpha., particularly those identified in the Ligand Binding Domain (LBD), result in the ability to bind to DNA in the absence of ligand and confer hormone independence in cells harboring such mutant receptors.
[0006] Most of the endocrine therapy resistance mechanisms identified rely on ER.alpha.-dependent activity. One of the new strategies to counterforce such resistance is to shut down the ER.alpha. signaling by removing ER.alpha. from the tumor cells using Selective Estrogen Receptors degraders (SERDs). Clinical and preclinical data showed that a significant number of the resistance pathways can be circumvented by the use SERDs.
[0007] There is still a need to provide SERDs with good degradation efficacy.
[0008] G. M. Anstead et al. have described 2,3-diarylindenes and 2,3-diarylindenones as binders of estrogen receptors (Journal of Medicinal Chemistry, 1988, Vol. 31, No. 7, p. 1316-1326).
[0009] R. McCague et al. have described analogues of (Z)- and (E)-4-hydroxytamoxifen and have tested their binding affinities to estrogen receptors (Journal of Medicinal Chemistry, 1998, Vol. 31, No. 7, p. 1285-1290).
[0010] Provided herein are novel compounds able to selectively antagonize and degrade the estrogen receptors (SERDs compounds), for use in cancer treatment.
[0011] Provided herein are compounds of the formula (I):
##STR00002##
wherein: [0012] R1 and R2 represent independently a hydrogen atom or a deuterium atom; [0013] R3 represents a hydrogen atom, a --COOH group, a --OH group or a --OPO(OH).sub.2 group; [0014] R4 represents a hydrogen atom or a fluorine atom; [0015] R5 represents a hydrogen atom or a --OH group; [0016] wherein: [0017] at least one of R3 or R5 is different from a hydrogen atom; [0018] when R3 represents a --COOH group, a --OH group or a --OPO(OH).sub.2 group, then R5 represents a hydrogen atom; [0019] when R5 represents a --OH group, then R3 and R4 represent hydrogen atoms; [0020] R6 is selected from: [0021] a phenyl group or a heteroaryl group comprising 3 to 9 carbon atoms and comprising from 1 to 3 heteroatoms independently selected from oxygen, nitrogen and sulphur, said phenyl and heteroaryl groups being unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.6)-alkoxy group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a cyano group; a sulphur group substituted with 5 fluorine atoms or (C.sub.1-C.sub.6)-alkyl groups substituted with two or more (such as 2 or 3) fluorine atoms; a sulfonyl-(C.sub.1-C.sub.6)-alkyl group wherein said (C.sub.1-C.sub.6)-alkyl group are unsubstituted or substituted with two or more (such as 2 or 3) fluorine atoms; a silane group substituted with 3 (C.sub.1-C.sub.6)-alkyl groups; an amine group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups; an amide group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups; a heterocycloalkyl group saturated or partially saturated, comprising 3 to 5 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur; or a heteroaryl group comprising 2 to 4 carbon atoms and comprising 1 to 3 heteroatoms selected from oxygen, nitrogen and sulphur and being unsubstituted or substituted with an oxo group; [0022] a cycloalkyl group or a heterocycloalkyl group comprising 4 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said cycloalkyl or heterocycloalkyl groups being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: [0023] a fluorine atom; a --OH group; a (C.sub.1-C.sub.6)-alkyl group; a --COOR7 group wherein R7 is a (C.sub.1-C.sub.6)-alkyl group; and an oxo group.
[0024] The compounds of formula (I) contain one or more asymmetric carbon atoms, more particularly one asymmetric carbon atom on the pyrrolydinyl group. They may therefore exist in the form of enantiomers. The compounds of formula (I) include enantiomers, racemates, and mixtures thereof. (In particular, the carbon 3 of the pyrrolidinyl group linked to the oxygen atom of the formula (I) may be in the absolute configuration (R) or (S). The carbon 3 of the pyrrolidinyl group is advantageously in the absolute configuration (S).
[0025] The compounds of formula (I) also include tautomer forms thereof.
[0026] The compounds of formula (I) may exist in the form of bases, acids, or zwitterions.
[0027] The compounds of formula (I) can be in the form of addition salts with acids or bases. Hence, provided herein inter alia, are compounds of formula (I) or to pharmaceutically acceptable salts thereof.
[0028] These salts may be prepared with pharmaceutically acceptable acids or bases, although the salts of other acids or bases useful, for example, for purifying or isolating the compounds of formula (I) are provided.
[0029] As used herein, the terms below have the following definitions unless otherwise mentioned throughout the instant specification: [0030] a halogen atom: a fluorine, a chlorine, a bromine or an iodine atom; [0031] an oxo: a ".dbd.O" group; [0032] a cyano group: a "--C.ident.N" group; [0033] an amine group: a nitrogen atom unsubstituted or substituted with one or more (C.sub.1-C.sub.6)-alkyl groups; [0034] an amide group: a --C(O)NH.sub.2 group wherein the nitrogen atom can be unsubstituted or substituted with one or more (C.sub.1-C.sub.6)-alkyl groups; [0035] a silane group: a silicon atom substituted with 3 (C.sub.1-C.sub.6)-alkyl groups; [0036] an alkyl group: a linear or branched saturated hydrocarbon-based aliphatic group comprising, unless otherwise mentioned, from 1 to 6 carbon atoms (noted "(C.sub.1-C.sub.6)-alkyl"). By way of examples, mention may be made of, but not limited to: methyl, ethyl, propyl, n-propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, hexyl and isohexyl groups, and the like; [0037] an alkoxy group: an --O-alkyl group where the alkyl group is as previously defined. By way of examples, mention may be made of, but not limited to: methoxy, ethoxy, propoxy, isopropoxy, linear, secondary or tertiary butoxy, isobutoxy, pentoxy or hexoxy groups, and the like; [0038] a cycloalkyl group: a cyclic alkyl group comprising, unless otherwise mentioned, from 3 to 6 carbon atoms, saturated or partially unsaturated and unsubstituted or substituted. By way of examples, mention may be made of, but not limited to: cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexenyl groups, and the like; [0039] a heterocycloalkyl group: a cyclic alkyl group comprising, unless otherwise mentioned, from 3 to 6 carbon atoms and containing 1 or 2 heteroatoms such as oxygen, nitrogen or sulphur. Such nitrogen atom may be substituted by an oxygen atom in order to form a --N--O bond. Such --N--O bond can be in a form of a N-oxide (--N.sup.+--O.sup.-). Such heterocycloalkyl group may be saturated or partially saturated and unsubstituted or substituted and may be monocyclic or bicyclic.
[0040] By way of examples of monocyclic heterocycloalkyl groups, mention may be made of, but not limited to: tetrahydropyridinyl, dihydropyridinyl, dihydropyranyl, tetrahydropyranyl groups, and the like.
[0041] A bicyclic heterocycloalkyl group means: a phenyl group fused to a monocyclic heterocycloalkyl group as defined above. By way of examples of bicyclic heterocycloalkyl groups, mention may be made of, but not limited to: tetrahydroquinolinyl, indolinyl, benzodioxolyl, dihydrobenzodioxinyl, dihydrobenzoxazinyl, benzofuranyl groups, all optionally substituted as above indicated, and the like. [0042] A heteroaryl group: a cyclic aromatic group containing between 4 and 9 carbon atoms and containing between 1 and 3 heteroatoms, such as nitrogen, oxygen or sulphur. Such nitrogen atom may be substituted by an oxygen atom in order to form a --N--O bond. Such --N--O bond can be in a form of a N-oxide (--N.sup.+O.sup.-). Said heteroaryl group may be monocyclic or bicyclic. By way of examples of heteroaryl groups, mention may be made of, but not limited to: isoxazole, pyridine, pyrimidine, benzotriazole, benzoxazole, pyrrolo[2,3-b]pyridine, benzimidazole, benzoxadiazole, benzothiazole, benzothiadiazole, benzofuran, indole, quinolyl, indazole, benzisoxazole, benzisothiazole groups and the like; [0043] A zwitterion: a globally neutral molecule with a positive and a negative electrical charge and having an acid group and a basic group. By way of examples, mention may be made of, but not limited to compounds of formula (I) having R3 which represents a --COOH group or an --OPO(OH).sub.2 group.
[0044] In an embodiment, in the compounds of formula (I), R1 and R2 represent hydrogen atoms.
[0045] In another embodiment, in the compounds of formula (I), R1 and R2 represent deuterium atoms.
[0046] In another embodiment, in the compounds of formula (I), R3 represents a hydrogen atom.
[0047] In another embodiment, in the compounds of formula (I), R3 represents a --COOH group.
[0048] In another embodiment, in the compounds of formula (I), R3 represents a --OH group.
[0049] In another embodiment, in the compound of formula (I), R3 represents a --COOH group or a --OH group.
[0050] In another embodiment, in the compounds of formula (I), R3 represents a --OPO(OH).sub.2 group.
[0051] In another embodiment, in the compounds of formula (I), R4 represents a hydrogen atom.
[0052] In another embodiment, in the compounds of formula (I), R4 represents a fluorine atom.
[0053] In another embodiment, in the compounds of formula (I), R5 represents a hydrogen atom.
[0054] In another embodiment, in the compounds of formula (I), R5 represents a --OH group.
[0055] In another embodiment, in the compounds of formula (I) R6 is selected from a phenyl group unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.6)-alkoxy group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a cyano group; a sulphur group substituted with 5 fluorine atoms or (C.sub.1-C.sub.6)-alkyl groups substituted with two or more (such as 2 or 3) fluorine atoms; a sulfonyl-(C.sub.1-C.sub.6)-alkyl group wherein said (C.sub.1-C.sub.6)-alkyl group are unsubstituted or substituted with two or more (such as 2 or 3) fluorine atoms; a silane group substituted with 3 (C.sub.1-C.sub.6)-alkyl groups; an amine group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups; an amide group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups; a heterocycloalkyl group saturated or partially saturated, comprising 3 to 5 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur; and a heteroaryl group comprising 2 to 4 carbon atoms and comprising 1 to 3 heteroatoms selected from oxygen, nitrogen and sulphur and being unsubstituted or substituted with an oxo group.
[0056] In another embodiment, in the compounds of formula (I), R6 is selected from a phenyl group unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.6)-alkoxy group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a cyano group; a sulphur group substituted with 5 fluorine atoms or (C.sub.1-C.sub.6)-alkyl groups substituted with two or more (such as 2 or 3) fluorine atoms; a sulfonyl-(C.sub.1-C.sub.6)-alkyl group wherein said (C.sub.1-C.sub.6)-alkyl group are unsubstituted or substituted with two or more (such as 2 or 3) fluorine atoms; a silane group substituted with 3 (C.sub.1-C.sub.6)-alkyl groups; an amide group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups; a heterocycloalkyl group saturated or partially saturated, comprising 3 to 5 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, and a heteroaryl group comprising 2 to 4 carbon atoms and comprising 1 to 3 heteroatoms selected from oxygen, nitrogen and sulphur and being unsubstituted or substituted with an oxo group.
[0057] In another embodiment, in the compounds of formula (I), R6 is selected from a phenyl group unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.3)-alkyl group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.3)-alkoxy group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a cyano group; a sulphur group substituted with 5 fluorine atoms or (C.sub.1-C.sub.3)-alkyl groups substituted with two or more (such as 2 or 3) fluorine atoms; a sulfonyl-(C.sub.1-C.sub.3)-alkyl group wherein said (C.sub.1-C.sub.3)-alkyl group are unsubstituted or substituted with two or more (such as 2 or 3) fluorine atoms; a silane group substituted with 3 (C.sub.1-C.sub.3)-alkyl groups; an amide group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.3)-alkyl groups; a heterocycloalkyl group saturated or partially saturated, comprising 3 to 5 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, and a heteroaryl group comprising 2 to 4 carbon atoms and comprising 1 to 3 heteroatoms selected from oxygen, nitrogen and sulphur and being unsubstituted or substituted with an oxo group.
[0058] In another embodiment, in the compounds of formula (I), R6 is selected from a phenyl group unsubstituted or substituted with 1 to 3 substituents independently selected from: a methyl group; an ethyl group; an isopropyl group; a tert-butyl group; a --CHF.sub.2 group; a --CF.sub.3 group; a --CF.sub.2CH.sub.3 group; a chlorine atom; a fluorine atom; a --OH group; a --OCH.sub.3 group; a --OCH.sub.2CH.sub.3 group; a --OCH.sub.2CH.sub.2F group; a --OCHF.sub.2 group; a --OCH.sub.2CHF.sub.2 group; a --OCF.sub.3 group; a --OCH.sub.2CF.sub.3 group; a cyano group; a --SCHF.sub.2 group; a --SCF.sub.3 group; a --SF.sub.5 group; a --SO.sub.2CH.sub.3 group; a --SO.sub.2CF.sub.3 group; a --Si(CH.sub.3).sub.3 group; an oxetane group; a piperidine group; a morpholine group; a pyrrolidine group and a triazolone group.
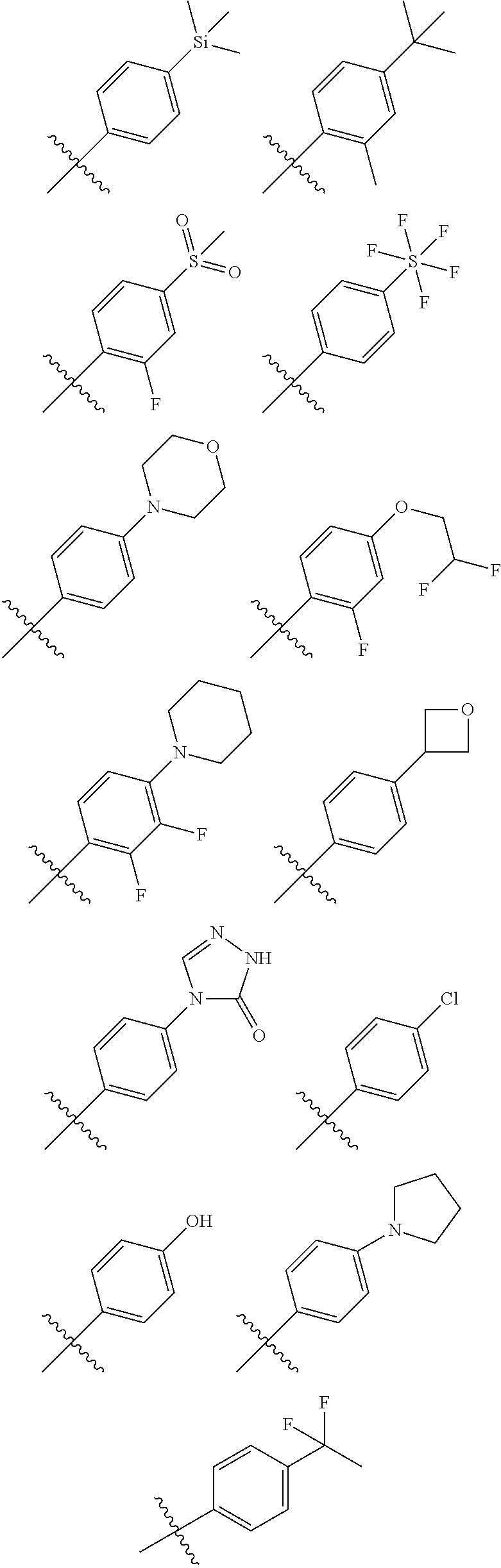
[0059] In another embodiment, in the compounds of formula (I), R6 is selected from an unsubstituted or substituted phenyl group selected from the following list:
##STR00003## ##STR00004## ##STR00005## ##STR00006## ##STR00007##
[0060] In another embodiment, in the compounds of formula (I), R6 is selected from a heteroaryl group comprising 3 to 9 carbon atoms and comprising from 1 to 3 heteroatoms independently selected from oxygen, nitrogen and sulphur, said heteroaryl group being unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a halogen atom; a --OH group; a N-oxide (--N.sup.+--O.sup.-), a (C.sub.1-C.sub.6)-alkoxy group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a cyano group; a sulphur group substituted with 5 fluorine atoms or (C.sub.1-C.sub.6)-alkyl groups substituted with two or more (such as 2 or 3) fluorine atoms; a sulfonyl-(C.sub.1-C.sub.6)-alkyl group wherein said (C.sub.1-C.sub.6)-alkyl group being unsubstituted or substituted with two or more (such as 2 or 3) fluorine atoms; a silane group substituted with 3 (C.sub.1-C.sub.6)-alkyl groups; an amine group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups; an amide group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups; a heterocycloalkyl group saturated or partially saturated, comprising 3 to 5 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur; and a heteroaryl group comprising 2 to 4 carbon atoms and comprising 1 to 3 heteroatoms selected from oxygen, nitrogen and sulphur and being unsubstituted or substituted with an oxo group.
[0061] In another embodiment, in the compounds of formula (I), R6 is selected from a heteroaryl group comprising 3 to 9 carbon atoms and comprising from 1 to 3 heteroatoms independently selected from oxygen, nitrogen and sulphur, said heteroaryl group being unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.6)-alkoxy group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; and an amine group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.6)-alkyl groups.
[0062] In another embodiment, the compounds of formula (I), R6 is selected from a heteroaryl group comprising 3 to 9 carbon atoms and comprising from 1 to 3 heteroatoms independently selected from oxygen, nitrogen and sulphur, said heteroaryl group being unsubstituted or substituted with 1 to 3 substituents independently selected from: a (C.sub.1-C.sub.3)-alkyl group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; a halogen atom; a --OH group; a (C.sub.1-C.sub.3)-alkoxy group unsubstituted or substituted with one or more (such as 1, 2 or 3) fluorine atoms; and an amine group unsubstituted or substituted with one or more (such as 1 or 2) (C.sub.1-C.sub.3)-alkyl groups.
[0063] In another embodiment, in the compounds of formula (I), R6 is selected from a heteroaryl group comprising 3 to 9 carbon atoms and comprising from 1 to 3 heteroatoms independently selected from oxygen, nitrogen and sulphur, said heteroaryl group being unsubstituted or substituted with 1 to 3 substituents independently selected from: a methyl group; a --CF.sub.3 group; a chlorine atom; a fluorine atom; a --OH group; a --OCH.sub.3 group; a --OCH.sub.2CH.sub.3 group; a --OCHF.sub.2 group; and a --NH.sub.2 group.
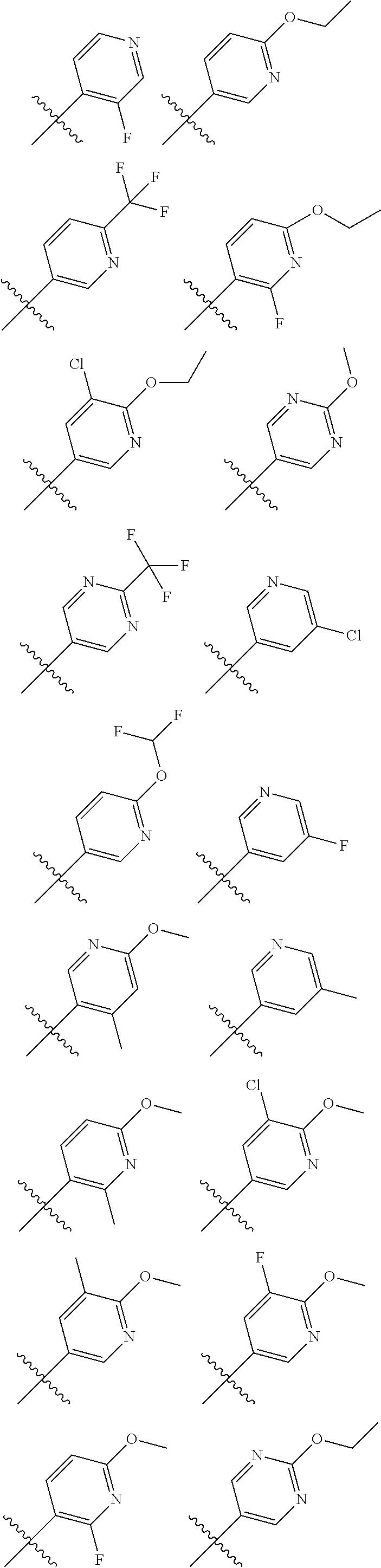
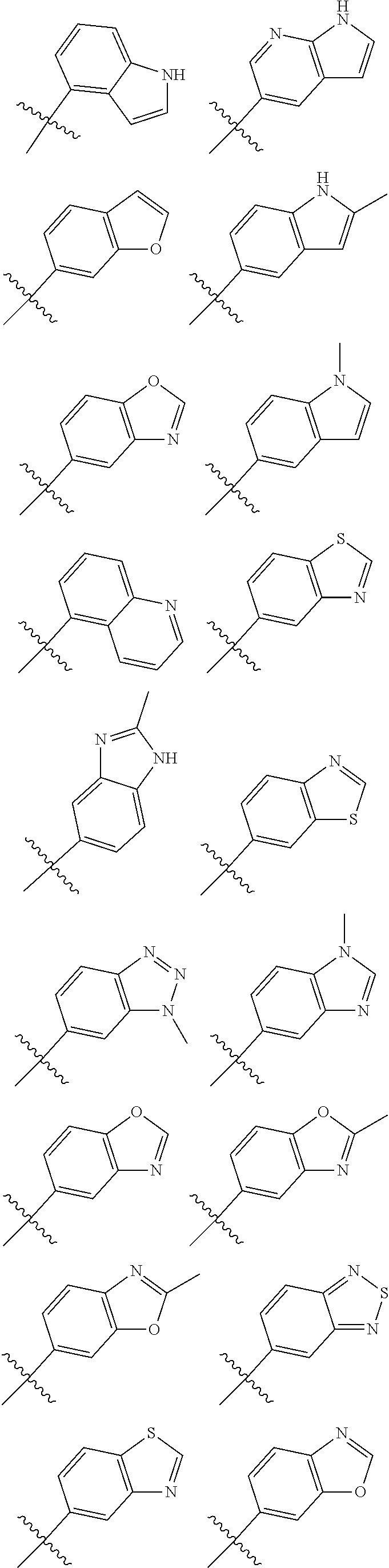
[0064] In another embodiment, in the compounds of formula (I), R6 is selected from an unsubstituted or substituted heteroaryl group selected from the following list:
##STR00008## ##STR00009## ##STR00010## ##STR00011##
[0065] In another embodiment, in the compounds of formula (I), R6 is selected from a (C.sub.1-C.sub.6)-cycloalkyl group saturated or partially saturated and unsubstituted or substituted with 1 or 2 substituents independently selected from: a fluorine atom; a --OH group; a (C.sub.1-C.sub.6)-alkyl group; a --COOR7 group wherein R7 is a (C.sub.1-C.sub.6)-alkyl group; and an oxo group.
[0066] In another embodiment, in the compounds of formula (I), R6 is selected from a (C.sub.1-C.sub.6)-cycloalkyl group saturated or partially saturated and unsubstituted or substituted with 1 or 2 substituents independently selected from: a fluorine atom; a --OH group; a (C.sub.1-C.sub.3)-alkyl group; a --COOR7 group wherein R7 is a (C.sub.1-C.sub.3)-alkyl group; and an oxo group.
[0067] In another embodiment, in the compounds of formula (I), R6 is selected from a (C.sub.1-C.sub.6)-cycloalkyl group saturated or partially saturated, unsubstituted or substituted with 1 or 2 substituents independently selected from: a fluorine atom or a --OH group.
[0068] In another embodiment, in the compounds of formula (I), R6 is selected from a substituted (C.sub.1-C.sub.6)-cycloalkyl group selected from the following list:
##STR00012##
[0069] In another embodiment, in the compounds of formula (I), R6 is selected from a heterocycloalkyl group comprising 4 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: a fluorine atom; a --OH group; a (C.sub.1-C.sub.6)-alkyl group; a --COOR7 group wherein R7 is an (C.sub.1-C.sub.6)-alkyl group; and an oxo group.
[0070] In another embodiment, in the compounds of formula (I), R6 is selected from: [0071] a monocyclic-(C.sub.1-C.sub.6)-heterocycloalkyl group comprising one heteroatom selected from oxygen, nitrogen and sulphur, said monocyclic heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 or 2 substituents independently selected from: a (C.sub.1-C.sub.6)-alkyl group and a --COOR7 group wherein R7 is an (C.sub.1-C.sub.6)-alkyl group or [0072] a bicyclic heterocycloalkyl group comprising 8 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said bicyclic heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: a fluorine atom; a (C.sub.1-C.sub.6)-alkyl group; a --COOR7 group wherein R7 is an (C.sub.1-C.sub.6)-alkyl group; and an oxo group.
[0073] In another embodiment, in the compounds of formula (I), R6 is selected from: [0074] a monocyclic-(C.sub.1-C.sub.6)-heterocycloalkyl group comprising one heteroatom selected from oxygen, nitrogen and sulphur, said monocyclic heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 or 2 substituents independently selected from: a (C.sub.1-C.sub.3)-alkyl group and a --COOR7 group wherein R7 is an (C.sub.1-C.sub.4)-alkyl group or [0075] a bicyclic heterocycloalkyl group comprising 8 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said bicyclic heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: a fluorine atom; a (C.sub.1-C.sub.3)-alkyl group; a --COOR7 group wherein R7 is an (C.sub.1-C.sub.4)-alkyl group; and an oxo group.
[0076] In another embodiment, in the compounds of formula (I), R6 is selected from: [0077] a monocyclic (C.sub.1-C.sub.6)-heterocycloalkyl group comprising one heteroatom selected from oxygen, nitrogen and sulphur, said monocyclic heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 or 2 substituents independently selected from: a methyl group and a --COO-tert butyl group or [0078] a bicyclic heterocycloalkyl group comprising 8 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said bicyclic heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: a fluorine atom; a methyl group; an ethyl group; a --COO-tert butyl group; and an oxo group.
[0079] In another embodiment, in the compounds of formula (I), R6 is selected from a monocyclic (C.sub.1-C.sub.6)-heterocycloalkyl group comprising one heteroatom selected from oxygen, nitrogen and sulphur, said monocyclic (C.sub.1-C.sub.6)-heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 or 2 substituents independently selected from: a methyl group and a --COO-tert butyl group.
[0080] In another embodiment, in the compounds of formula (I), R6 is selected from an unsubstituted or substituted monocyclic (C.sub.1-C.sub.6)-heterocycloalkyl group selected from the following list:
##STR00013##
[0081] In another embodiment, in the compounds of formula (I), R6 is selected from a bicyclic heterocycloalkyl group comprising 8 to 9 carbon atoms and comprising 1 or 2 heteroatoms independently selected from oxygen, nitrogen and sulphur, said bicyclic heterocycloalkyl group being saturated or partially saturated and being unsubstituted or substituted with 1 to 4 substituents independently selected from: a fluorine atom; a methyl group; an ethyl group; a --COO-tert butyl group; and an oxo group.
[0082] In another embodiment, in the compounds of formula (I), R6 is selected from an unsubstituted or substituted bicyclic heterocycloalkyl group selected from the following list:
##STR00014##
[0083] The compounds of formula (I) include the compounds having any combination of the above-defined embodiments for R1, R2, R3, R4, R5 and R6 with each other.
[0084] Among the compounds of formula (I), mention may be made in particular of the following compounds: [0085] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydroxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-3-ol; [0086] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(3-hydroxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-3-ol; [0087] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indol-5-yl)- -8,9-dihydro-7H-benzo[7]annulen-3-ol; [0088] 6-(2-chloro-4-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-3-ol; [0089] 6-(2-chloro-4-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0090] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(3-hydroxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0091] 6-(3-chloro-2-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0092] 6-(2-chloro-3-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0093] 6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0094] 6-(2-fluoro-4-hydroxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0095] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydroxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0096] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indol-5-yl)- -8,9-dihydro-7H-benzo[7]annulen-2-ol; [0097] 6-(4-chloro-3-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0098] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(3-fluoro-4-pyr- idyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0099] 6-(4-chloro-2-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0100] 6-(4-chloro-3-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0101] 6-(4-fluoro-2-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0102] 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0103] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indol-6-yl)- -8,9-dihydro-7H-benzo[7]annulen-2-ol; [0104] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indol-4-yl)- -8,9-dihydro-7H-benzo[7]annulen-2-ol; [0105] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-indolin-5-yl-8,- 9-dihydro-7H-benzo[7]annulen-2-ol; [0106] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-pyrrolo[2,3- -b]pyridin-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0107] 6-(2-chloro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0108] tert-butyl 4-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hydroxy-8,9-- dihydro-7H-benzo[7]annulen-6-yl]-3,6-dihydro-2H-pyridine-1-carboxylate; [0109] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-methy- l-3,6-dihydro-2H-pyridin-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0110] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1,2,3,6-tetrah- ydropyridin-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0111] 6-(4-ethoxy-2-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0112] 6-(benzofuran-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxypheny- l]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0113] 6-(2-fluoro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0114] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(2-methyl-1H-ind- ol-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0115] 6-(2,3-dimethylphenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0116] 6-(4-chloro-2-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0117] 6-(3-fluoro-2-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0118] 6-(6-ethoxy-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0119] 6-(3-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0120] 5-[4-[(3S)-1-(1,1-dideuterio-3-fluoro-propyl)pyrrolidin-3-yl]oxphenyl]-6-- (2-fluoro-4-methyl-phenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0121] 6-(3-chloro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0122] 6-(3,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0123] 6-(3-chloro-2-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0124] 6-(4-fluoro-2-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0125] 6-(3-fluoro-2-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0126] 6-(4-ethoxy-2-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0127] 6-(2-chloro-4-ethoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0128] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(4-methoxy-2-met- hyl-phenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0129] 1-fluoro-6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0130] 6-(4-ethoxy-2-methyl-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0131] 6-(2,4-dichlorophenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0132] 6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0133] 6-(2,3-dihydro-1,4-benzodioxin-6-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrroli- din-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0134] 6-(4-fluoro-3-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0135] 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0136] 6-(4-ethoxy-2,3-difluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0137] 6-(4-chloro-3-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0138] 6-(1,3-benzoxazol-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0139] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(4-hydroxyphenyl- )-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0140] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(2-isopropylphen- yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0141] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(o-tolyl)-8,9-di- hydro-7H-benzo[7]annulen-2-ol; [0142] 6-(2-chlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl- ]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0143] 2-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hydroxy-8,9-- dihydro-7H-benzo[7]annulen-6-yl]-5-methoxy-benzonitrile; [0144] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-[2(trifluorometh- yl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0145] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-[4-fluoro-2-(tri- fluoromethyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0146] 6-(4-ethoxy-2,5-difluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0147] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(4-methoxy-2-met- hyl-phenyl)-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride; [0148] 6-(2,4-dimethoxyphenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyp- henyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0149] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-methoxy-2-(t- rifluoromethyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0150] 6-[4-(difluoromethoxy)-3-fluoro-phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrr- olidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0151] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[2-methyl-4-(tr- ifluoromethyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0152] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[6-(trifluorome- thyl)-3-pyridyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0153] 6-[4-(difluoromethoxy)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0154] 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0155] 6-(6-ethoxy-2-fluoro-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0156] 6-(4-tert-butylphenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0157] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1,2,3,4-tetrah- ydroquinolin-6-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0158] 6-(3-ethoxyphenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl- ]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0159] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trifluorome- thoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0160] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-methoxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0161] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(3-methoxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0162] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trifluorome- thyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0163] 6-[4-(difluoromethoxy)-3-fluoro-phenyl]-1-fluoro-5-[4-[(3S)-1-(3-fluoropr- opyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0164] 6-(5-chloro-6-ethoxy-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrro- lidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0165] 6-(2-ethylphenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]- -8,9-dihydro-7H-benzo[7]annulen-2-ol; [0166] 6-(6-ethoxy-2-fluoro-3-pyridyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyr- rolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0167] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(2-methoxypyrimi- din-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0168] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-[2-(trifluoromet- hyl)pyrimidin-5-yl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0169] 2-fluoro-4-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-2-hydr- oxy-8,9-dihydro-7H-benzo[7]annulen-6-yl]benzonitrile; [0170] 6-(5-chloro-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphe- nyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0171] 6-[6-(difluoromethoxy)-3-pyridyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin- -3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0172] 6-(2,5-difluoro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin- -3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0173] 6-(2-chloro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0174] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(5-fluoro-3-pyri- dyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0175] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(6-methoxy-4-met- hyl-3-pyridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0176] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(4-methoxy-2,5-d- imethyl-phenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0177] 6-(2,3-difluoro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin- -3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0178] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-[4-(trifluoromet- hylsulfanyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0179] 6-(3-chloro-4-ethoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0180] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(5-methyl-3-pyri- dyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0181] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(6-methoxy-2-met- hyl-3-pyridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0182] 6-(2,2-dimethyl-3H-benzofuran-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0183] 6-(5-chloro-6-methoxy-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0184] 6-(4-ethoxy-2,5-dimethyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0185] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(6-methoxy-5-me- thyl-3-pyridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol [0186] 6-(5-fluoro-6-methoxy-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0187] 6-(3-chloro-4-ethoxy-2-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0188] 6-(2-fluoro-6-methoxy-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0189] 6-(3,5-difluoro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin- -3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0190] 6-(1-ethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyp- henyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0191] 6-(2-ethoxypyrimidin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]o- xyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0192] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(6-methoxy-3-py- ridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0193] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(2-methoxy-4-py- ridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0194] 6-(6-ethoxy-5-methyl-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0195] 6-(3-fluoro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0196] 6-(2,4-difluoro-3-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin- -3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0197] 6-(4-chloro-3-methyl-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0198] 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(tr- ifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0199] 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-flu- oro-2-(trifluoromethyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol;
[0200] 6-[4-(difluoromethoxy)-2-fluoro-phenyl]-5-[4-[(3S)-1-(3-fluoroprop- yl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0201] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[2-fluoro-4-(tr- ifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0202] 6-(2,6-dichloro-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]o- xyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0203] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(2,2,2-trifl- uoroethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0204] 6-(4-ethoxy-3,5-difluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0205] 6-(4-chloro-2-fluoro-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0206] 6-(2-chloro-3-fluoro-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0207] 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[2-met- hyl-4-(trifluoromethyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0208] 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(tr- ifluoromethyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0209] 6-(6-ethoxy-2-methyl-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0210] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-methylindol-- 5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0211] 6-(6-chloro-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0212] 2-fluoro-4-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hyd- roxy-8,9-dihydro-7H-benzo[7]annulen-6-yl]-N-methyl-benzamide; [0213] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[2-fluoro-6-(tr- ifluoromethyl)-3-pyridyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0214] 6-[4-(2-fluoroethoxy)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0215] 6-(4-ethoxy-2,3-dimethyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0216] 6-[6-ethoxy-5-(trifluoromethyl)-3-pyridyl]-5-[4-[(3S)-1-(3-fluoropropyl)p- yrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0217] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-methyl-2,3-d- ihydro-1,4-benzoxazin-7-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0218] 6-(2,2-difluoro-1,3-benzodioxol-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0219] 4-ethyl-6-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hydr- oxy-8,9-dihydro-7H-benzo[7]annulen-6-yl]-1,4-benzoxazin-3-one; [0220] 6-[2-chloro-4-(trifluoromethoxy)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrr- olidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0221] 6-[4-(difluoromethoxy)-3,5-difluoro-phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)- pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0222] 6-(4-tert-butylphenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0223] 6-(6-ethoxy-4-methyl-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0224] 6-(3-chloro-4-ethoxy-5-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0225] 6-(2-aminopyrimidin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]ox- yphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0226] 6-[4-(difluoromethyl)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0227] 6-[4-(difluoromethoxy)phenyl]-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrro- lidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0228] 6-[3,5-difluoro-4-(trifluoromethoxy)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)- pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0229] 6-[4-(difluoromethoxy)-2-methyl-phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrr- olidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0230] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[2-methyl-4-(tr- ifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0231] 6-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hydroxy-8,9-- dihydro-7H-benzo[7]annulen-6-yl]-4-methyl-1,4-benzoxazin-3-one; [0232] 6-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hydroxy-8,9-- dihydro-7H-benzo[7]annulen-6-yl]-4H-1,4-benzoxazin-3-one; [0233] 6-(2,3-dichloro-4-ethoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0234] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[3-methyl-4-(tr- ifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0235] 6-[3-chloro-4-(trifluoromethoxy)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrr- olidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0236] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(5-quinolyl)-8,- 9-dihydro-7H-benzo[7]annulen-2-ol; [0237] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-pyridyl)-8,9- -dihydro-7H-benzo[7]annulen-2-ol; [0238] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(3-pyridyl)-8,9- -dihydro-7H-benzo[7]annulen-2-ol; [0239] 6-[2-chloro-6-(trifluoromethyl)-3-pyridyl]-5-[4-[(3S)-1-(3-fluoropropyl)p- yrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0240] tert-butyl 6-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hydroxy-8,9-- dihydro-7H-benzo[7]annulen-6-yl]-2,3-dihydro-1,4-benzoxazine-4-carboxylate- ; [0241] 6-[4-(difluoromethylsulfanyl)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl- )pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0242] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1,2,3,4-tetrah- ydroquinolin-7-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0243] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trifluorome- thoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0244] 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[2-flu- oro-4-(trifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0245] 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6- -[4-(trifluoromethylsulfanyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0246] 6-(2,4-dichloro-5-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrr- olidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0247] [5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trifluorom- ethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-yl] dihydrogen phosphate; [0248] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(5-methy- lisoxazol-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0249] 6-[4-(difluoromethoxy)-2-fluoro-phenyl]-1-fluoro-5-[4-[(3S)-1-(3-fluoropr- opyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0250] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trif- luoromethylsulfonyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0251] 6-(3,4-dihydro-2H-1,4-benzoxazin-6-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrro- lidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol hydrochloride; [0252] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[2-fluor- o-4-(trifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0253] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-isoxazol-4-yl-8- ,9-dihydro-7H-benzo[7]annulen-2-ol; [0254] 6-(6-ethoxy-5-fluoro-3-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0255] 6-fluoro-5-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hyd- roxy-8,9-dihydro-7H-benzo[7]annulen-6-yl]pyridin-2-ol; [0256] 6-(6-tert-butyl-2-fluoro-4-pyridyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0257] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(4-trimethylsily- lphenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0258] 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride; [0259] 6-(1,3-benzothiazol-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]ox- phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0260] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(2-methyl-1H-be- nzimidazol-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0261] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-[4-(trifluoromet- hylsulfanyl)phenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0262] 6-(1,3-benzothiazol-6-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-- 3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0263] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(3-methylbenzotr- iazol-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0264] 6-[2-chloro-4-(trifluoromethoxy)phenyl]-1-fluoro-5-[4-[(3S)-1-(3-fluoropr- opyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0265] 6-(4-tert-butyl-2-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrr- olidin-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0266] 6-(2-fluoro-4-methylsulfonyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrroli- din-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0267] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(3-methylisoxazo- l-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0268] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-[4-(pentafluoro-- sulfanyl)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0269] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(4-morpholinophe- nyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0270] 6-[4-(2,2-difluoroethoxy)-2-fluoro-phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)p- yrrolidin-3-yl]oxphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0271] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxphenyl]-6-(1-methylbenzimi- dazol-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0272] 6-(1,2-benzoxazol-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxph- enyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0273] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-oxidopyridin- -1-ium-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0274] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-pyrrolidin-1- -ylphenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0275] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(2-methyl-1,3-b- enzoxazol-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0276] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(2-methyl-1,3-b- enzoxazol-6-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0277] 6-(2,1,3-benzoxadiazol-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0278] 6-(2,1,3-benzothiadiazol-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-- yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0279] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(oxetan-3-yl- )phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0280] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(2,2,3,3-tetraf- luoro-1,4-benzodioxin-6-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0281] 6-(1,2-benzothiazol-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]ox- yphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0282] 6-[2,3-difluoro-4-(1-piperidyl)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrro- lidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0283] 6-(1,3-benzoxazol-6-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyp- henyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0284] 6-(1,2-benzoxazol-6-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyp- henyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0285] 6-[4-(1,1-difluoroethyl)phenyl]-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0286] 6-(3,6-dihydro-2H-pyran-4-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0287] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-tetrahydropyran- -4-yl-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0288] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydroxycyclo- hexyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0289] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(3-methylbenzim- idazol-5-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0290] 4-[4-[5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-2-hydroxy-8- ,9-dihydro-7H-benzo[7]annulen-6-yl]phenyl]-1H-1,2,4-triazol-5-one; [0291] 6-(4,4-difluorocyclohexen-1-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0292] 6-(4,4-difluorocyclohexyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]o- xyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0293] 6-(4-chlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl- ]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0294] 6-(2-chlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl- ]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0295] 6-(2,4-dichlorophenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0296] 6-(4-chloro-2-fluoro-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0297] 6-(2-chloro-4-fluoro-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl- )pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid; [0298] 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-phenyl-6,7-di- hydro-5H-benzo[7]annulen-3-ol; [0299] 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indazol-5-y- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol; [0300] 6-(2-Chloro-3-fluoro-phenyl)-5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-y- loxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-carboxylic acid; [0301] 5-{4-[(S)-1-(3-Fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-6-phenyl-8,9-di- hydro-7H-benzocycloheptene-2-carboxylic acid; [0302] 6-Benzooxazol-5-yl-5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phen- yl}-8,9-dihydro-7H-benzocycloheptene-2-carboxylic acid; and [0303] 6-[4-(1,1-Difluoro-ethyl)-phenyl]-5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidi- n-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-carboxylic acid; [0304] or a pharmaceutically acceptable salt thereof.
[0305] Another embodiment is a compound selected from the above list, or a pharmaceutically acceptable salt thereof, for use in therapy, especially as an inhibitor and degrader of estrogen receptors.
[0306] Another embodiment is a compound selected from the above list, or a pharmaceutically acceptable salt thereof, for use in the treatment of cancer, especially breast cancer.
[0307] Another embodiment is a method of treating cancer, comprising administering to a subject in need thereof, in particular a human, a therapeutically effective amount of a compound selected from the above list, or a pharmaceutically acceptable salt thereof.
[0308] Another embodiment is a pharmaceutical composition comprising as active principle an effective dose of a compound selected from the above list, or a pharmaceutically acceptable salt thereof, and also at least one pharmaceutically acceptable excipient.
[0309] The compounds of the formula (I) can be prepared by the following processes.
[0310] The compounds of the formula (I) and other related compounds having different substituents are synthesized using techniques and materials described below or otherwise known by the skilled person in the art. In addition, solvents, temperatures and other reaction conditions presented below may vary as deemed appropriate to the skilled person in the art.
[0311] General below methods for the preparation of compounds of formula (I) are optionally modified by the use of appropriate reagents and conditions for the introduction of the various moieties found in the compound of formula (I) as described below.
[0312] The following abbreviations and empirical formulae are used: [0313] AcOEt ethyl acetate [0314] AlCl.sub.3 aluminium trichloride [0315] Boc tert-butyloxycarbonyl [0316] P(Ph).sub.2-(CH.sub.2).sub.3--P(Ph).sub.2 1,3-bis(diphenylphosphino)propane [0317] Ph.sub.3P.dbd.O triphenylphosphine oxide [0318] Cs.sub.2CO.sub.3 cesium carbonate [0319] CO carbon monoxide [0320] DCM dichloromethane [0321] DMF N,N-dimethylformamide [0322] DMSO dimethyl sulfoxide [0323] EDCl 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide [0324] Et.sub.3N triethylamine [0325] EtOH ethanol [0326] Et.sub.2O diethyl ether [0327] Hal halogen atom [0328] HCl hydrogen chloride [0329] HPLC high-performance liquid chromatography [0330] K.sub.2CO.sub.3 potassium carbonate [0331] LCMS liquid chromatography/mass spectrometry [0332] LiAlD.sub.4 lithium aluminum deuteride [0333] Lutidine 2,6-dimethyl-pyridine [0334] MeOH methanol [0335] MgSO.sub.4 magnesium sulfate [0336] NaOH sodium hydroxide [0337] NaCl sodium chloride [0338] NaHCO.sub.3 sodium bicarbonate [0339] Na.sub.2SO.sub.4 sodium sulfate [0340] NH.sub.4H.sub.2PO.sub.4 ammonium dihydrogen phosphate [0341] NH.sub.4Cl ammonium chloride [0342] NH.sub.4OH ammonium hydroxide [0343] Pd(OAc).sub.2 palladium acetate [0344] Pd(dppf)Cl.sub.2 1,1'bis(diphenylphosphino)ferrocene] dichloropalladium(II) [0345] Tf.sub.2O triflic anhydride [0346] THF tetrahydrofuran [0347] .degree. C. degrees Celsius [0348] RT room temperature [0349] min minute(s) [0350] mL millilitre(s) [0351] mmol millimole(s) [0352] .mu.mol micromole(s) [0353] .mu.M micromolar [0354] nM nanomolar [0355] ppm parts per million [0356] SCX strong cation exchange [0357] HIC hydrophobic interaction column
##STR00015## ##STR00016##
[0358] According to SCHEME 1, in which R1, R2, R3, R4, R5 and R6 are defined as described above, a substituted 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene intermediate (A) is converted in STEP 1 into the corresponding enol triflate intermediate (B) by treatment for example with triflic anhydride (Tf.sub.2O) in solution in dichloromethane (DCM) in the presence of a base, for example pyridine, at room temperature. This intermediate (B) is subjected in STEP 2 to a Suzuki coupling with reagent (1) ((S)-1-(3-fluoropropyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2dioxaborolan2yl)ph- enoxy)pyrrolidine) using for example [1,1'-bis(diphenylphosphino) ferrocene]dichloropalladium(II) (Pd(dppf)Cl.sub.2), complex with DCM, as catalyst, in a mixture of dioxane and water and in the presence of a base, for example cesium carbonate (Cs.sub.2CO.sub.3), at room temperature or by heating up to reflux. The preparation of reagent (1) is described hereunder in SCHEME 2.
[0359] The intermediate (C) obtained is brominated in STEP 3 using for example pyridinium tribromide in DCM or tetrahydrofuran (THF) at room temperature. This bromo derivative intermediate (D) is then subjected in STEP 4 to a second Suzuki coupling with a suitable boronic reagent R6B(OR').sub.2, wherein --B(OR').sub.2 is a boronic acid or a pinacolate ester and R6 is as above defined, using for example Pd(dppf)Cl.sub.2, complex with DCM, as catalyst, in a mixture of dioxane and water as solvent and in the presence of a base, for example Cs.sub.2CO.sub.3, at room temperature or by heating up to reflux.
[0360] In the above-described reactions, it can be necessary to protect reactive functional groups, for example hydroxy, amino, thio or carboxy groups, where these groups are desired in the final product, to avoid their unwanted participation in the reactions. Conventional protecting groups can be used in accordance with standard practice, for example, see T. W. Greene and P. G. M. Wuts in "Protective Groups in Organic Chemistry', John Wiley and Sons, 2006.
[0361] When R3 or R5 represents a --OH group, this --OH group is protected, for example as a pivaloyl ester. Deprotection can be performed just after STEP 3 or after STEP 4 by treating, with an aqueous solution of sodium hydroxide 2N (NaOH), a solution of the pivaloyl ester in methanol (MeOH) at room temperature, followed by acidification with an aqueous solution of hydrogen chloride 2N (HCl).
[0362] When R3 represents a --COOH group, this --COOH group is protected, for example as a methyl ester. Deprotection is performed just after STEP 4 by treating, with an aqueous solution of sodium hydroxide (NaOH) 2N, a solution of the methyl ester in MeOH at room temperature, followed by acidification with an aqueous solution of HCl 2N.
[0363] In an embodiment, it can be advantageous to use a variation of SCHEME 1, called SCHEME 1a depicted below, that consists in transforming intermediate (D) into a boronate derivative which is engaged in a Suzuki coupling with an halogenated derivative R6-Hal, wherein R6 is as above defined and Hal represents a halogen atom selected from a chlorine, a bromine or an iodine atom. Deprotection of the --OH group or --COOH group of R3 or R5 can be performed before or after STEP 1 or STEP 2 of SCHEME 1a, as explained above.
##STR00017##
[0364] STEP 1 of the above SCHEME 1a consists of reacting intermediate (D) with 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) using for example Pd(dppf)Cl.sub.2, complex with DCM, as catalyst, in a mixture of dioxane and water and in the presence of a base, for example Cs.sub.2CO.sub.3, at about 80.degree. C. Intermediate (D') obtained is engaged in STEP 2 of the above SCHEME 1a in a Suzuki coupling with R6-Hal, wherein R6 and Hal are as above defined, using for example Pd(dppf)Cl.sub.2, complex with DCM, as catalyst, in a mixture of dioxane and water as solvent and in the presence of a base, for example Cs.sub.2CO.sub.3, at about 70.degree. C.
[0365] In another embodiment, it can be advantageous, when the compounds of formula (I) are such that R3 is a --COOH group, to use a variation of SCHEME 1, called SCHEME 1 b depicted below.
##STR00018##
[0366] Hereinabove SCHEME 1 b describes the synthesis of the compounds of the formula (Ib) wherein R3 represents a --COOH group, by generating the --COOH group from the --OH group of R3 of the compounds of formula (Ia) wherein R1, R2, R4 and R6 are as above defined in formula (I). In STEP 1 of SCHEME 1 b the --OH group of R3 in the compounds of formula (Ia) is transformed into a triflate group with, for example Tf.sub.2O in DCM with a base, for example pyridine, at room temperature.
[0367] The intermediate (K) obtained is then carbonylated in STEP 2 of SCHEME 1 b under 2 to 10 bars of carbon monoxide (CO) at about 70.degree. C. in a mixture of MeOH and N,N-dimethylformamide (DMF) using for example palladium acetate (Pd(OAc).sub.2) and 1,3-bis(diphenylphosphino)propane (P(Ph).sub.2-(CH.sub.2).sub.3--P(Ph).sub.2) as catalytic system.
[0368] The methyl ester of formula (Ib) then obtained is deprotected as defined above, so as to obtain compounds of formula (I) wherein R1, R2, R4 and R6 are as above defined in formula (I) and R3 is a --COOH group.
[0369] In another embodiment, it can be advantageous as well, when R3 represents a --COOH group, to use a variation of SCHEME 1, called SCHEME 1c depicted below. This SCHEME 1c is an alternative process to the above SCHEME 1 b.
##STR00019##
[0370] Hereinabove SCHEME 1c describes the synthesis of the intermediates (C''') as defined above by generating the --COOMe group from the --OH group of intermediates (C') wherein R1, R2 and R4 are as above defined in formula (I). In STEP 1 of SCHEME 1c, the --OH group is transformed into a triflate group with, for example, Tf.sub.2O in DCM with a base, for example pyridine, at room temperature.
[0371] The intermediate (C'') then obtained is carbonylated in STEP 2 of SCHEME 1c under 2 to 10 bars of CO at about 70.degree. C. in a mixture of MeOH and DMF using for example Pd(dppf)Cl.sub.2 or Pd(OAc).sub.2 and P(Ph).sub.2-(CH.sub.2).sub.3--P(Ph).sub.2 as catalytic system.
[0372] In another embodiment, when R1 and R2 represent simultaneously a deuterium atom and R3 is other than a --COOH group, a process of synthesis of compounds of formula (Ic) is described below in SCHEME 1d which is a variation of the general SCHEME 1.
##STR00020## ##STR00021##
[0373] According to SCHEME 1d, a substituted enol triflate intermediate (B), obtained in accordance to STEP 1 of SCHEME 1, is subjected in STEP 1 of SCHEME 1d to a Suzuki coupling with compound (c) (tert-butyl (3S)-3-[4-(tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidine-1-carb- oxylate) using for example Pd(dppf)Cl.sub.2, complex with DCM, as catalyst, in dioxane and in the presence of a base, for example Cs.sub.2CO.sub.3, at room temperature.
[0374] The intermediate (E) then obtained is brominated in STEP 2 of SCHEME 1d using for example pyridinium tribromide in DCM or THF at room temperature.
[0375] This bromo derivative intermediate (F) obtained is then subjected in STEP 3 of SCHEME 1d to a second Suzuki coupling with a suitable boronic reagent R6B(OR').sub.2, wherein the --B(OR').sub.2 group is a boronic acid or a pinacolate ester, and R6 is as above defined, using for example Pd(dppf)Cl.sub.2, complex with DCM, as catalyst, in a mixture of dioxane and water and in the presence of a base, for example Cs.sub.2CO.sub.3, at room temperature or by heating up to reflux.
[0376] This intermediate (G) obtained is N-deprotected in STEP 4 of SCHEME 1d using for example a 4N solution of HCl in dioxane, at room temperature.
[0377] The NH-pyrrolidine intermediate (H) obtained is amidified in STEP 5 of SCHEME 1d using for example 3-fluoropropionic acid in DMF at room temperature using for example 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDCl) as coupling agent.
[0378] Finally, the carbonyl of the amide intermediate (J) then obtained is reduced in STEP 6 of SCHEME 1d into the deuteriated amine of formula (Ic) by for example lithium aluminum deuteride (LiAlD.sub.4) in ether (Et.sub.2O) at room temperature.
[0379] When R3 or R5 is a --OH group, this --OH group is protected, for example as a pivaloyl ester. Deprotection is done at the final STEP 6 for example by reduction with LiAlD.sub.4.
##STR00022##
[0380] According to the above SCHEME 2, commercially available compound (a) (4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenol) is condensed in STEP 1 of SCHEME 2 in THF at room temperature on (R)-1-N-Boc-3-hydroxypyrrolidine using N,N,N',N'-tetramethylazodicarboxamide as coupling agent.
[0381] Compound (c) thus obtained is N-deprotected in STEP 2 of SCHEME 2 in MeOH at room temperature using an acidic agent for example a solution of HCl 4N in dioxane. Alkylation of the pyrrolidine nitrogen is then performed in STEP 3 of SCHEME 2 by reacting compound (d) with the corresponding 1,1-disubstituted 1-halogeno-3-fluoro propane, for example 1-iodo-3-fluoropropane in acetonitrile in presence of potassium carbonate (K.sub.2CO.sub.3) at about 40.degree. C.
[0382] The new intermediate (A10) wherein R3 represents a --COOMe group, R4 represents a fluorine atom and R5 represents a hydrogen atom can be prepared according to reaction SCHEME 3 highlighted below:
##STR00023##
[0383] According to SCHEME 3, intermediate (A6) (1-fluoro 2-methoxy 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene) is converted in STEP 1 into the corresponding phenol intermediate (A7) by treatment for example with aluminum trichloride (AlCl.sub.3) in toluene at about 90.degree. C.
[0384] The --OH group is then transformed in STEP 2 of SCHEME 3 into a triflate group by treatment with a base, for example 2,6-dimethyl-pyridine (lutidine), using for example Tf.sub.2O in DCM at room temperature to obtain intermediate (A9).
[0385] The intermediate (A9) then obtained is finally carbonylated in STEP 3 of SCHEME 3 in the presence of a base, for example triethylamine (Et.sub.3N) and a catalyst, for example Pd(OAc).sub.2, under 2 to 10 bars of CO in a mixture of DMF and MeOH at about 70.degree. C. to produce intermediate (A10) (1-fluoro 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene 2-carboxylic acid, methyl ester).
[0386] In the above SCHEMES 1, 1a, 1 b, 1c, 1d, 2 and 3 the starting compounds and the reactants, when their preparation is not herein described, are commercially available, for example by Sigma-Aldrich, Fluka, Acros Organics, Alfa Aesar, and the like, or are described in the literature, or may be prepared by methods which are known to a person skilled in the art.
[0387] In another aspect, provided are the compounds as defined below, wherein R1, R2, R3, R4, R5 and R6 are as defined in formula (I) above, which are useful as intermediates or reagents in the synthesis of the compounds of the formula (I) as above defined, or salts thereof:
##STR00024## ##STR00025## ##STR00026##
[0388] Some compounds of formula (I) are described, with their structure, name, method of preparation and analytical data, in the below Table 1, which is merely illustrative and does not limit the scope of the present invention.
[0389] The methods of preparation A, B and C mentioned in table 1 are respectively described in examples 1, 51 and 48 below.
[0390] The examples with numbers underlined in Table 1 are further detailed hereafter. The 1H NMR spectra at 400 and 500 MHz were performed on a Bruker Avance DRX-400 and Bruker Avance DPX-500 spectrometer, respectively, with the chemical shifts (.delta. in ppm) in the solvent dimethyl sulfoxide-d6 (d6-DMSO) referenced at 2.5 ppm at a temperature of 303 K. Coupling constants (J) are given in Hertz.
[0391] The liquid chromatography/mass spectra (LC/MS) were obtained on a UPLC Acquity Waters instrument, light scattering detector Sedere and SQD Waters mass spectrometer using UV detection DAD 210<1<400 nm and column Acquity UPLC CSH C18 1.7 .mu.m, dimension 2.1.times.30 mm, mobile phase H.sub.2O+0.1% HCO.sub.2H/CH.sub.3CN+0.1% HCO.sub.2H.
TABLE-US-00001 TABLE 1 MASS: LC/MS Exam- Me- (m/z, ples Structure Name thod NMR MH+): 1 ##STR00027## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- hydroxyphenyl)- 8,9-dihydro- 7H- benzo[7] annulen-3-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 1.98 (m, 2 H); 2.20 (m, 3 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.54 (m, 1 H); 2.63 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.19 (d, J = 2.6 Hz, 1 H); 6.55 (m, 3 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.92 (d, J = 8.6 Hz, 2 474 H); 7.04 (d, J = 8.6 Hz, 1 H); 8.98 (s, 1 H); 9.28 (s, 1 H) 2 ##STR00028## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- hydroxyphenyl)- 8,9-dihydro- 7H- benzo[7] annulen-3-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.99 (m, 2 H); 2.19 (m, 3 H); 2.35 to 2.68 (m, 7 H); 2.80 (m, 1 H); 4.47 (td, J = 6.0 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.20 (d, J = 2.5 Hz, 1H); 6.48 to 6.55 (m, 3 H); 6.58 (dd, J = 2.5 and 8.6 Hz, 1 H); 6.62 (d, J = 8.6 Hz, 2 H); 6.74 (d, J = 8.6 Hz, 2 H); 6.96 (t, J = 8.2 Hz, 1 H); 7.06 (d, 474 J = 8.2 Hz, 1 H); 9.00 (s, 1 H); 9.19 (s, 1 H) 3 ##STR00029## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1H- indol-5-yl)- 8,9- dihydro- 7H- benzo[7] annulen-3-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.00 (m, 2 H); 2.17 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.61 (m, 1 H); 2.69 (m, 2 H); 2.78 (dd, =6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.21 (d, J = 2.9 Hz, 1 H); 6.29 (t, J = 2.9 Hz, 1 H); 6.58 (m, 3 H); 6.74 (d, 497 J = 8.8 Hz, 2 H); 6.85 (dd, J = 1.7 and 8.5 Hz, 1 H); 7.07 (d, J = 8.3 Hz, 1 H); 7.16 (d, J = 8.5 Hz, 1 H); 7.25 (t, J = 2.9 Hz, 1 H); 7.34 (s, 1 H); 8.99 (s, 1 H); 10.97 (m, 1 H) 4 ##STR00030## 6-(2- chloro-4- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-3-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.15 (t, J = 7.0 Hz, 2 H); 2.20 (m, 1 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.52 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.62 (m, 1 H); 2.69 to 2.82 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.20 (d, J = 2.7 Hz, 1 H); 6.62 (dd, J = 2.7 and 8.3 Hz, 1 H; 6.64 (d, 510 J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.04 (dt, J = 2.7 and 9.0 Hz, 1 H); 7.10 (d, J = 8.3 Hz, 1 H); 7.17 (dd, J = 6.4 and 8.9 Hz, 1 H); 7.40 (dd J = 2.7 and 8.9 Hz, 1 H); 9.07 (s, 1 H) 5 ##STR00031## 6-(2- chloro-4- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.19 (m, 3 H); 2.37 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.61 to 2.83 (m, 4 H); 4.47 (td, J = 6.2 and 47.6 Hz, 2 H); 4.72 (m, 1 H); 6.57 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.71 (m, 3 H); 7.02 (dt, J = 2.6 and 9.0 Hz, 1 H); 7.17 (dd, J = 6.4 and 9.0 Hz, 1 H); 7.39 (dd, J = 2.6 508 and 9.0 Hz, 1 H); 9.43 (s, 1 H) 6 ##STR00032## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- hydroxyphenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.20 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.66 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.48 to 6.55 (m, 5 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 474 H); 6.94 (t, J = 8.0 Hz, 1 H); 9.15 (s, 1 H); 9.39 (s, 1 H) 7 ##STR00033## 6-(3- chloro-2- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.01 to 2.23 (m, 5 H); 2.20 (s, 3 H); 2.35 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.60 to 2.82 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.57 (d, J = 1.5 Hz, 2 H); 6.59 (d, J = 8.9 Hz, 2 H); 6.66 (d, J = 8.9 Hz, 2 H); 6.71 (t, J = 1.5 Hz, 1 H); 6.97 (d, J = 8.0 Hz, 1 506 H); 7.03 (t, J = 8.0 Hz, 1 H); 7.21 (d, J = 8.0 Hz, 1 H); 9.41 (s, 1 H) 8 ##STR00034## 6-(2- chloro-3- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.18 (m, 3 H); 2.37 (m, 1 H); 2.43 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.78 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.58 (s, 2 H); 6.60 (d, J = 8.9 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.9 Hz, 2 H); 6.99 (m, 1 H); 7.20 (m, 2 H); 9.46 (s, 1 H) 510 9 ##STR00035## 6-(2-fluoro- 4-methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.12 to 2.22 (m, 3 H); 2.23 (s, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.63 (m, 1 H); 2.49 (t, J = 7.2 Hz, 2 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.3 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.56 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.71 490 (d, J = 8.8 Hz, 2 H); 6.82 (d, J = 8.0 Hz, 1 H); 6.87 (d, J = 11.3 Hz, 1 H); 7.00 (t, J = 8.0 Hz, 1 H); 9.41 (s, 1 H) 10 ##STR00036## 6-(2-fluoro- 4-hydroxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.13 (t, J = 7.0 Hz, 2 H); 2.20 (m, 1 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.70 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.40 (dd, J = 2.6 and 11.9 Hz, 1H); 6.42 (dd, 492 J = 2.6 and 8.7 Hz, 1H); 6.54 (m, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.89 (t, J = 8.7 Hz, 1 H); 9.39 (s, 1 H); 9.70 (s, 1 H) 11 ##STR00037## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- hydroxyphenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.20 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.63 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.52 (m, 4 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.68 (s, 1 H); 6.71 (d, J = 8.8 Hz, 2 H); 474 6.91 (d, J = 8.8 Hz, 2 H); 9.23 (s, 1 H); 9.32 (s, 1 H) 12 ##STR00038## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1H- indol-5-yl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.15 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.37 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.51 (m, 1 H); 2.61 (m, 1 H); 2.70 (t, J = 7.0 Hz, 2 H); 2.77 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.69 (m, 1 H); 6.28 (t, J = 2.8 Hz, 1 H); 6.53 (d, J = 8.8 Hz, 2 H); 6.55 (m, 2 497 H); 6.68 (d, J = 1.5 Hz, 1 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.84 (dd, J = 1.8 and 8.5 Hz, 1 H); 7.13 (d, J = 8.5 Hz, 1 H); 7.23 (t, J = 2.8 Hz, 1 H); 7.32 (d, J = 1.8 Hz, 1 H); 9.38 (s, 1 H); 11.90 (t, J = 2.8 Hz, 1 H) 13 ##STR00039## 6-(4- chloro-3- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.00 to 2.23 (m, 4 H); 2.12 (s, 3 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.59 to 2.81 (m, 5 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.56 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 7.01 (d, J = 8.5 Hz, 1 H); 7.09 (dd, J = 2.4 506 and 8.5 Hz, 1 H); 7.18 (d, =2.4 Hz, 1 H); 9.39 (s, 1 H) 14 ##STR00040## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- fluoro-4- pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 3 H); 2.38 (m, 1 H); 2.45 (t, J = 7.3 Hz, 2 H); 2.53 (m, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.1 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.59 (m, 2 H); 6.63 (d, J = 8.9 Hz, 2 H); 6.73 (m, 3 H); 7.25 (dd, J = 5.0 and 6.4 Hz, 1 477 H); 8.24 (dd, J = 1.7 and 5.0 Hz, 1 H); 8.37 (d, J = 1.7 Hz, 1 H); 9.50 (s, 1 H) 15 ##STR00041## 6-(4- chloro-2- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.13 to 2.24 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.73 (m, 1 H); 2.70 (t, J = 7.2 Hz, 2 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.3 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.58 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 510 H); 7.11 (dd, J = 2.2 and 8.2 Hz, 1 H); 7.18 (t, J = 8.2 Hz, 1 H); 7.27 (dd, J = 2.2 and 9.9 Hz, 1 H); 9.41 (s, 1 H) 16 ##STR00042## 6-(4- chloro-3- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.15 to 2.19 (m, 3 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.55 (m, 1 H); 2.68 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.57 (s, 2 H); 6.67 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 6.94 (dd, J = 2.0 and 510 8.5 Hz, 1 H); 7.13 (dd, J = 2.0 and 11.0 Hz, 1 H); 7.34 (t, J = 8.5 Hz, 1 H); 9.42 (s, 1 H) 17 ##STR00043## 6-(4-fluoro- 2-methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.02 to 2.24 (m, 5 H); 2.12 (s, 3 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.59 to 2.82 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 (s, 2 H); 6.58 (d, J = 8.8 Hz, 2 H); 6.67 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.74 (dt, J = 3.1 and 8.9 490 Hz, 1 H); 6.93 (dd, J = 3.1 and 10.4 Hz, 1 H); 7.02 (dd, J = 6.6 and 8.9 Hz, 1 H); 9.34 (s, 1 H) 18 ##STR00044## 6-(2,4- dichlorophenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.18 (m, 3 H); 2.38 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.62 (m, 1 H); 2.69 to 2.82 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.57 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.14 (d, J = 8.8 Hz, 1 H); 7.23 (dd, J = 2.5 526 and 8.8 Hz, 1 H); 7.54 (d, J = 2.5 Hz, 1 H); 9.42 (s, 1 H) 19 ##STR00045## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3-
yl]oxyphenyl]- 6-(1H- indol-6-yl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.15 (m, 1 H); 2.25 to 2.40 (m, 3 H); 2.43 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.69 (t, J = 7.0 Hz, 2 H); 2.77 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.30 (t, J = 2.8 Hz, 1 H); 6.53 (d, J = 8.8 Hz, 2 H); 6.55 (s, 2 H); 6.70 (s, 497 1 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.79 (dd, J = 1.6 and 8.3 Hz, 1 H); 7.15 (s, 1 H); 7.24 (t, J = 2.8 Hz, 1 H); 7.31 (d, J = 8.3 Hz, 1 H); 9.35 (s, 1 H); 10.89 (t, J = 2.8 Hz, 1 H) 20 ##STR00046## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1H- indol-4-yl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.62 (m, 1 H); 1.77 (dm, J = 25.3 Hz, 2 H); 2.00 (m, 2 H); 2.13 (m, 1 H); 2.31 (m, 3 H); 2.41 (t, J = 7.2 Hz, 2 H); 2.57 (m, 2 H); 2.74 (m, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.65 (m, 1 H); 6.25 (d, J = 3.0 Hz, 1 H); 6.44 (d, J = 8.8 Hz, 2 H); 6.58 (s, 2 H); 6.72 (m, 3 H); 6.71 (s, 1 H); 6.89 (t, J = 8.3 Hz, 1 H); 7.15 (d, J = 8.3 Hz, 1 497 H); 7.22 (7, J = 3.0 Hz, 1 H); 9.38 (s, 1 H); 11.00 (m, 1 H) 21 ##STR00047## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6- indolin-5- yl-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.01 (m, 2 H); 2.19 (m, 3 H); 2.39 (m, 1 H); 2.42 to 2.57 (m, 3 H); 2.61 (m, 3 H); 2.82 to 2.92 (m, 3 H); 3.38 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 5.39 (s, 1 H); 6.24 (d, J = 8.0 Hz, 1 H); 6.51 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.64 (m, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 499 6.81 (s, 1 H); 9.30 (s, 1 H) 22 ##STR00048## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1H- pyrrolo[2,3- b]pyridin- 5-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.17 (m, 1 H); 2.29 to 2.40 (m, 3 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.32 (dd, J = 2.0 and 2.9 Hz, 1 H); 6.57 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 498 H); 6.70 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 7.38 (t, J = 2.9 Hz, 1 H); 7.77 (d, J = 2.9 Hz, 1 H); 7.89 (d, J = 2.9 Hz, 1 H); 9.39 (s, 1 H); 11.49 (t, J = 2.9 Hz, 1 H) 23 ##STR00049## 6-(2- chloro-4- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.11 to 2.22 (m, 3 H); 2.23 (s, 3 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.58 to 2.83 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.56 (s, 2 H); 6.58 (d, J = 8.8 Hz, 2 H); 6.72 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.92 (d, J = 8.3 Hz, 1 H); 6.99 (d, J = 8.3 506 Hz, 1 H); 7.21 (s, 1 H); 9.41 (s, 1 H) 24 ##STR00050## tert-butyl 4-[5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]-3,6- dihydro-2H- pyridine-1- carboxylate A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.38 (s, 9 H); 1.70 to 2.08 (m, 9 H); 2.24 (m, 1 H); 2.40 (m, 1 H); 2.45 to 2.57 (m, 4 H); 2.60 (dd, J = 3.01 and 10.4 Hz, 1 H); 2.67 (m, 1 H); 2.83 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.24 (m, 2 H); 3.71 (m, 2 H); 4.49 (td, J = 6.1 and 47.5 Hz, 2 H); 4.80 (m, 1 H); 5.48 (m, 1 H); 6.52 (m, 2 H); 6.64 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 6.91 563 (d, J = 8.8 Hz, 2 H); 9.34 (s, 1 H) 25 ##STR00051## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1- methyl- 3,6- dihydro- 2H-pyridin- 4-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 to 1.89 (m, 3 H); 1.93 to 2.08 (m, 6 H); 2.14 (s, 3 H); 2.20 to 2.32 (m, 3 H); 2.40 (m, 1 H); 2.46 to 2.55 (m, 4 H); 2.60 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.69 (m, 1 H); 2.75 (m, 2 H); 2.84 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.49 (td, J = 6.1 and 47.5 Hz, 2 H); 4.81 (m, 1 H); 5.42 (m, 1 H); 6.51 (s, 2 H); 6.63 (s, 1 H); 6.73 (d, J = 8.8 Hz, 2 477 H); 6.91 (d, J = 8.8 Hz, 2 H); 9.31 (s, 1 H) 26 ##STR00052## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6- (1,2,3,6- tetrahydropyridin- 4- yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 to 1.89 (m, 5 H); 1.93 to 2.08 (m, 4 H); 2.15 (m, 1 H); 2.25 (m, 1 H); 2.40 (m, 1 H); 2.45 to 2.55 (m, 4 H); 2.60 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.83 (dd, J = 6.3 and 10.4 Hz, 1 H); 3.11 (m, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.81 (m, 1 H); 5.44 (m, 1 H); 6.51 (s, 2 H); 6.53 (s, 1 H); 6.73 (d, J = 8.8 Hz, 2 434 H); 6.92 (d, J = 8.8 Hz, 2 H); 9.31 (s, 1 H) 27 ##STR00053## 6-(4- ethoxy-2- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.29 (t, J = 7.1 Hz, 3 H); 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.99 to 2.23 (m, 5 H); 2.08 (s, 3 H); 2.35 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.59 to 2.81 (m, 4 H); 3.94 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.55 (m, 4 H); 6.59 (dd, J = 2.5 and 8.7 Hz, 1 H); 6.66 (m, 3 H); 6.70 (s, 1 H); 516 6.90 (d, J = 8.7 Hz, 1 H); 9.36 (s, 1 H) 28 ##STR00054## 6- (benzofuran- 5-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.20 (t, J = 7.0 Hz, 2 H); 2.35 to 2.80 (m, 6 H); 2.70 (t, J = 7.0 Hz, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.58 (m, 4 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.82 (dd, J = 1.1 and 2.5 Hz, 1 H); 7.05 (d, J = 2.0 and 8.7 Hz, 1 H); 7.37 (d, J = 8.7 Hz, 498 1 H); 7.42 (d, J = 2.0 Hz, 1 H); 7.91 (d, J = 2.5 Hz, 1 H); 9.39 (s, 1 H) 29 ##STR00055## 6-(2-fluoro- 4- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.15 (t, J = 7.0 Hz, 2 H); 2.20 (m, 1 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.71 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.55 (s, 2 H); 6.60 (m, 3 H); 6.65 to 6.73 (m, 4 H); 7.02 (t, 506 J = 8.9 Hz, 1 H); 9.40 (s, 1 H) 30 ##STR00056## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- methyl-1H- indol-5-yl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.61 to 1.87 (m, 3 H); 2.02 (m, 2 H); 2.15 (m, 1 H); 2.22 to 2.80 (m, 13 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 5.95 (s, 1 H); 6.52 (m, 4 H); 6.65 to 6.79 (m, 4 H); 7.02 (d, J = 8.5 Hz, 1 H); 7.16 (s, 1 H); 9.31 (s, 1 H); 10.76 (m, 1 H) 511 31 ##STR00057## 6-(2,3- dimethylphenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.98 to 2.22 (m, 5 H); 2.10 (s, 3 H); 2.19 (s, 3 H); 2.36 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.59 to 2.70 (m, 2 H); 2.72 (m, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.54 (d, J = 8.8 Hz, 2 H); 6.57 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.79 (d, J = 7.8 Hz, 1 486 H); 6.77 (t, J = 7.8 Hz, 1 H); 6.92 (d, J = 7.8 Hz, 1 H); 9.38 (s, 1 H) 32 ##STR00058## 6-(4- chloro-2- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.00 to 2.24 (m, 5 H); 2.13 (s, 3 H); 2.36 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.59 to 2.81 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.55 (s, 2 H); 6.58 (d, J = 8.8 Hz, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 7.02 (d, J = 8.2 Hz, 1 H); 7.10 (d, J = 8.2 Hz, 506 1 H); 7.19 (s, 1 H); 9.40 (s, 1 H) 33 ##STR00059## 6-(3-fluoro- 2-methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.00 to 2.23 (m, 5 H); 2.03 (s, 3 H); 2.36 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.59 to 2.80 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 (s, 2 H); 6.58 (d, J = 8.8 Hz, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.88 (d, J = 7.8 Hz, 1 H); 6.92 (m, 1 H); 7.16 490 (m, 1 H); 9.41 (s, 1 H) 34 ##STR00060## 6-(6- ethoxy-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.26 (t, J = 7.1 Hz, 3 H); 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 to 2.28 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.1 Hz, 2 H); 2.55 (m, 1 H); 2.66 (m, 3 H); 2.80 (dd, J = 6.5 and 10.5 Hz, 1 H); 4.20 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.55 (s, 2 H); 6.60 (d, J = 8.7 Hz, 1 H); 6.64 503 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.76 (d, J = 8.8 Hz, 2 H); 7.43 (dd, J = 2.4 and 8.7 Hz, 1 H); 7.84 (d, J = 2.4 Hz, 1 H); 9.40 (s, 1 H) 35 ##STR00061## 6-(3-fluoro- 4-methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.13 (s, 3 H); 2.17 to 2.27 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.66 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.54 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.74 490 (d, J = 8.8 Hz, 2 H); 6.84 (m, 2 H); 7.04 (t, J = 8.2 Hz, 1 H); 9.41 (s, 1 H) 36 ##STR00062## 5-[4-[(3S)- 1-(1,1- dideuterio- 3-fluoro- propyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- fluoro-4- methyl- phenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.77 (td, J = 6.3 and 25.6 Hz, 2 H); 2.04 (m, 2 H); 2.12 to 2.23 (m, 3 H); 2.24 (s, 3 H); 2.38 (m, 1 H); 2.53 (m, 1 H); 2.63 (m, 1 H); 2.70 (t, J = 7.0 Hz, 2 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.3 and 47.7 Hz, 2 H); 4.72 (m, 1 H); 6.56 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.82 (d, J = 8.1 Hz, 1 H); 6.88 (d, J = 11.6 Hz, 492 1 H); 7.00 (t, J = 8.1 Hz, 1 H); 9.42 (s, 1 H) 37 ##STR00063## 6-(3- chloro-4- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.15 to 2.28 (m, 3 H); 2.21 (s, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.2 and 10.5 Hz, 1 H); 2.65 (m, 3 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.56 (s, 2 H); 6.64 (d, J = 9.0 Hz, 2 H); 6.70 (s, 1 H); 6.74 506 (d, J = 9.0 Hz, 2 H); 6.98 (dd, J = 1.9 and 8.0 Hz, 1 H); 7.10 (d, J = 1.9 Hz, 1 H); 7.12 (d, J = 8.0 Hz, 1 H); 9.41 (s, 1 H) 38 ##STR00064## 6-(3,4- dichlorophenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.4 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.26 (t, J = 7.2 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.5 Hz, 1 H); 2.67 (m, 3 H); 2.80 (dd, J = 6.4 and 10.5 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.57 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.70 s, 1 H); 6.76 526 (d, J = 8.8 Hz, 2 H); 7.09 (dd, J = 2.2 and 8.5 Hz, 1 H); 7.32 (d, J = 2.2 Hz, 1 H); 7.40 (d, J = 8.5 Hz, 1 H); 9.47 (s, 1 H) 39 ##STR00065## 6-(3- chloro-2- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.19 (m, 3 H); 2.38 to 2.58
(m, 4 H); 2.60 to 2.81 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.56 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.71 (m, 3 H); 7.05 (t, J = 8.2 Hz, 1 H); 7.13 (m, 1 H); 7.36 (m, 1 H); 9.48 (s, 1 H) 510 40 ##STR00066## 6-(4-fluoro- 2- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.95 to 2.23 (m, 5 H); 2.31 to 2.55 (m, 4 H); 2.60 to 2.72 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.72 (s, 3 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.48 to 6.61 (m, 5 H); 6.71 (m, 3 H); 6.83 (m, 2 H); 9.37 (s, 1 H) 506 41 ##STR00067## 6-(3-fluoro- 2- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.13 to 2.24 (m, 3 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.74 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.57 (m, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.73 506 (d, J = 8.8 Hz, 2 H); 6.78 (dd, J = 2.0 and 8.2 Hz, 1 H); 6.85 (td, J = 5.0 and 8.2 Hz, 1 H); 7.03 (ddd, J = 2.0 and 8.2 and 12.2 Hz, 1 H); 9.40 (s, 1 H) 42 ##STR00068## 6-(4- ethoxy-2- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.38 (t, J = 7.1 Hz, 3 H); 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.10 to 2.24 (m, 3 H); 2.38 (m, 1 H); 2.42 to 2.58 (m, 3 H); 2.59 to 2.71 (m, 3 H); 2.79 (m, 1 H); 6.98 (q, J = 7.1 Hz, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.62 to 6.76 (m, 9 H); 7.00 (t, J = 8.8 Hz, 1 H); 9.40 (s, 1 H) 520 43 ##STR00069## 6-(2- chloro-4- ethoxy- phenyl)-5- [4-[(3S)-1- (3- flouropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.26 (t, J = 7.3 Hz, 3 H); 1.66 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.02 to 2.25 (m, 5 H); 2.35 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.59 to 2.82 (m, 4 H); 3.98 (q, J = 7.3 Hz, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.54 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.70 (m, 4 H); 6.93 (d, J = 2.4 Hz, 1 H); 6.99 536 (d, J = 8.9 Hz, 1 H); 9.41 (s, 1 H) 44 ##STR00070## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- methoxy- 2-methyl- phenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.98 to 2.22 (m, 5 H); 2.08 (s, 3 H); 2.36 (m, 1 H); 2.43 (m, 2 H); 2.53 (m, 1 H); 2.59 to 2.82 (m, 4 H); 3.68 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.50 to 6.71 (m, 9 H); 6.91 (d, J = 8.3 Hz, 1 H); 9.36 (s, 1 H) 502 45 ##STR00071## 1-fluoro-6- (2-fluoro-4- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.19 (m, 3 H); 2,22 (s, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.63 (m, 1 H); 2.79 (m, 3 H); 4.47 (td, J = 6.2 and 47.6 Hz, 2 H); 4.72 (m, 1 H); 6.40 (d, J = 8.5 Hz, 1 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.73 d, J = 8.8 Hz, 2 H); 6.74 (t, J = 8.5 Hz, 1 H); 6.83 (d, J = 8.0 Hz, 1 H); 6.89 (d, J = 11.2 Hz, 1 H); 7.01 508 (t, J = 8.0 Hz, 1 H); 9.83 (s, 1 H) 46 ##STR00072## 6-(4- ethoxy-2- methyl- phenyl)-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.28 (t, J = 7.1 Hz, 3 H); 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.00 to 2.23 (m, 5 H); 2.10 (s, 3 H); 2.37 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.74 to 2.85 (m, 3 H); 3.93 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.49 (d, J = 9.0 Hz, 1 H); 6.56 (d, J = 8.9 Hz, 2 H); 6.59 (dd, J = 2.6 and 8.6 Hz, 1 H); 6.65 (d, J = 2.6 Hz, 1 H); 534 6.68 (d, J = 8.6 Hz, 2 H); 6.71 (t, J = 9.0 Hz, 1 H); 6.90 (d, J = 8.6 Hz, 1 H); 9.80 (s, 1 H) 47 ##STR00073## 6-(2,4- dichlorophenyl)- 1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.19 (m, 3 H); 2.38 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.64 (m, 1 H); 2.70 to 2.83 (m, 2 H); 2.98 (m, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.40 (d, J = 8.6 Hz, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.73 (m, 3 H); 7.17 (d, J = 8.6 Hz, 1 H); 7.25 (dd, J = 1.5 and 8.6 Hz, 1 H); 7.58 (d, J = 1.5 544 Hz, 1 H); 9.89 (s, 1 H) 48 ##STR00074## 6-(2-fluoro- 4-methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid C 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 to 2.22 (m, 5 H); 2.25 (s, 3 H); 2.37 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.64 (m, 1 H); 2.89 (dd, J = 6.3 and 10.5 Hz, 1 H); 2.83 (t, J = 6.8 Hz, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.62 (d, J = 8.9 Hz, 2 H); 6.72 (d, J = 8.9 Hz, 2 H); 6.80 to 6.89 (m, 2 H); 6.91 (dd, J = 1.7 and 11.4 Hz, 1 H); 518 7.05 (t, J = 7.8 Hz, 1 H); 7.72 (dd, J = 1.9 and 8.4 Hz, 1 H); 7.89 (d, J = 1.9 Hz, 1 H); 12.90 (m, 1 H) 49 ##STR00075## 6-(2,3- dihydro- 1,4- benzodioxin- 6-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.20 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.15 (s, 4 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.50 to 6.65 (m, 7 H); 6.67 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 9.37 (s, 1 H) 516 50 ##STR00076## 6-(4-fluoro- 3- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.61 to 2.70 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 3.59 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.57 (m, 2 H); 6.64 (d, J = 8.8 Hz, 2 506 H); 6.68 (m, 1 H); 6.70 (d, J = 2.4 Hz, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 6.83 (dd, J = 2.4 and 8.7 Hz, 1 H); 6.98 (dd, J = 8.7 and 11.8 Hz, 1 H); 9.39 (s, 1 H) 51 ##STR00077## 6-(2,4- dichlorophenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 to 2.23 (m, 5 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.55 to 2.89 (m, 3 H); 4.47 (td, J = 6.2 and 47.6 Hz, 2 H); 4.72 (m, 1 H); 6.63 (d, J = 8.9 Hz, 2 H); 6.71 (m, 3 H); 7.18 (d, J = 8.4 Hz, 1 H); 8.26 (dd, J = 2.0 and 8.4 Hz, 1 H); 7.58 (d, J = 2.0 Hz, 1 H); 7.63 (d, J = 8.4 Hz, 1 H); 7.79 (s, 1 H); 12.3 554 (m, 1 H) 52 ##STR00078## 6-(4- ethoxy- 2,3- difluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.31 (t, J = 7.1 Hz, 3 H); 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.18 (t, J = 7.0 Hz, 2 H); 2.20 (m, 1 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.09 (q, J = 7.1 Hz, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 538 6.57 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.80 to 6.91 (m, 2 H); 9.41 (s, 1 H) 53 ##STR00079## 6-(4- chloro-3- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.10 (m, 2 H); 2.17 to 2.28 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.2 and 10.5 Hz, 1 H); 2.66 (m, 1 H); 2.80 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.68 (d, J = 8.9 Hz, 2 H); 6.73 (m, 1 H); 6.76 (d, J = 8.9 Hz, 2 H); 6.99 (dd, J = 2.3 and 8.4 Hz, 1 H); 7.20 (dd, J = 2.3 and 10.8 Hz, 1 H); 538 7.39 (t, J = 8.4 Hz, 1 H); 7.66 (d, J = 8.5 Hz, 1 H); 7.80 (s, 1 H); 12.90 (m, 1 H) 54 ##STR00080## 6-(1,3- benzoxazol- 5-yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.17 (m, 1 H); 2.30 to 2.39 (m, 3 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.70 (t, J = 7.0 Hz, 2 H); 2.77 (dd, J = 6.2 and 10.5 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 (m, 4 H); 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.19 (dd, J = 1.6 499 and 8.8 Hz, 1 H); 7.54 (m, 2 H); 8.65 (s, 1 H); 9.39 (s, 1 H) 55 ##STR00081## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- hydroxyphenyl)- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.20 (m, 3 H); 2.38 (m, 1 H); 2.47 (m, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 1 H); 2.73 to 2.83 (m, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.56 (d, J = 8.8 Hz, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.77 (d, J = 8.3 Hz, 1 H); 6.95 (d, J = 8.8 Hz, 2 H); 7.68 (dd, J = 2.0 502 and 8.3 Hz, 1 H); 7.82 (d, J = 2.0 Hz, 1 H); 9.36 (s, 1 H); 12.80 (m, 1 H) 56 ##STR00082## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- isopropylphenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 0.70 (d, J = 6.8 Hz, 3 H); 1.09 (d, J = 6.8 Hz, 3 H); 1.63 (m, 1 H); 1.77 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.17 (m, 3 H); 2.35 (m, 1 H); 2.43 (t, J = 7.2 Hz, 2 H); 2.50 (m, 1 H); 2.61 (m, 1 H); 2.69 to 2.78 (m, 3 H); 3.11 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.52 to 6.60 (m, 4 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.70 (d, 500 J = 2.0 Hz, 1 H); 7.02 to 7.20 (m, 4 H); 9.36 (s, 1 H) 57 ##STR00083## 5-[4-[(3S)- 1-(3- fluoropropyl] oxyphenyl) pyrrolidin-3- yl]-6-(o- tolyl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.01 to 2.22 (m, 5 H); 2.12 (s, 3 H); 2.35 (m, 1 H); 2.44 (m, 2 H); 2.53 (m, 1 H); 2.59 to 2.82 (m, 4 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.54 (d, J = 8.8 Hz, 2 H); 6.57 (s, 2 H); 6.66 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.99 to 7.11 (m, 4 H); 9.37 (s, 1 H) 472 58 ##STR00084## 6-(2- chlorophenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.18 (m, 3 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.69 to 2.85 (m, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.57 (m, 4 H); 6.71 (m, 3 H); 7.08 to 7.20 (m, 3 H); 7.39 (d, J = 8.2 Hz, 1 H); 9.40 (s, 1 H) 492 59 ##STR00085## 2-[5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]-2- hydroxy- 8,9- dihydro- 7H- benzo[7] nulen-6- yl]-5- methoxy- benzonitrile A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.09 (m, 2 H); 2.14 to 2.26 (m, 3 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 1 H); 2.80 (m, 3 H); 3.76 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.57 (m, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.72 (s, 1 H); 513 7.17 (dd, J = 2.8 and 8.6 Hz, 1 H); 7.21 (d, J = 2.8 Hz, 1 H); 7.33 (d, J = 8.6 Hz, 1 H); 9.44 (s, 1 H) 60 ##STR00086## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.02 to 2.27 (m, 5 H); 2.36 (m, 1 H); 2.43 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.58 to 2.69 (m, 2 H); 2.77 (m, 1 H); 2.85 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.57 (m, 4 H); 6.70 (m, 3 H); 7.14 (d, J = 8.2 Hz, 1 H); 7.32 to 7.45 (m, 2 H); 7.69 (d, J = 8.2 Hz, 1 H); 9.40 526 (s, 1 H)
61 ##STR00087## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- fluoro-2- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 to 2.24 (m, 5 H); 2.37 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.63 (m, 2 H); 2.73 to 2.80 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 (m, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 7.20 (dd, J = 6.0 and 8.8 Hz, 1 H); 7.32 (dt, 544 J = 2.8 and 8.8 Hz, 1 H); 7.60 (dd, J = 2.8 and 9.6 Hz, 1 H); 9.41 (s, 1 H) 62 ##STR00088## 6-(4- ethoxy- 2,5- difluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.30 (t, J = 7.0 Hz, 3 H); 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.11 to 2.23 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.60 to 2.71 (m, 3 H); 2.80 (dd, J = 6.4 and 10.5 Hz, 1 H); 4.04 (q, J = 7.0 Hz, 2 H); 4.48 (td, J = 6.2 and 47.6 Hz, 2 H); 4.74 (m, 1 H); 6.57 (s, 2 H); 6.62 (d, J = 8.9 Hz, 2 H); 6.70 538 (s, 1 H); 6.73 (d, J = 8.9 Hz, 2 H); 6.92 (dd, J = 7.4 and 11.2 Hz, 1 H); 7.00 (dd, J = 7.0 and 11.8 Hz, 1 H); 9.41 (s, 1 H) 63 ##STR00089## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- methoxy- 2-methyl- phenyl)- 8,9-dihydro- 7H- benzo[7] annulene-2- carboxylic acid hydrochloride B 1H NMR (500 MHz, DMSO-d6, .delta. ppm): 1.92 to 2.30 (m, 8 H); 2.12 (s, 3 H); 2.89 (m, 2 H); 3.09 to 3.40 (m, 4 H); 3.67 (m, 2 H); 3.70 (s, 3 H); 4.52 (td, J = 5.7 and 47.0 Hz, 2 H); 4.95 to 5.08 (m, 1 H); 6.63 (dd, J = 2.6 and 8.5 Hz, 1 H); 6.68 (d, J = 9.0 Hz, 2 H); 6.70 (d, J = 2.6 Hz, 1 H); 6.73 (d, J = 9.0 Hz, 2 H); 6.84 (d, J = 8.0 Hz, 1 H); 6.97 (d, J = 8.5 Hz, 1 H); 7.84 (dd, J = 1.9 and 8.0 Hz, 1 H); 7.90 (d, J = 1.9 530 Hz, 1 H); 10.30 (s, 0.5 H); 10.64 (s, 0.5 H); 12.86 (s, 1 H) 64 ##STR00090## 6-(2,4- dimethoxyphenyl)- 5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.99 (m, 2 H); 2.09 (m, 2 H); 2.19 (m, 1 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.59 to 2.71 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.70 (s, 6 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.29 (dd, J = 2.5 and 8.5 Hz, 1 H); 6.49 (d, J = 2.5 Hz, 1 H); 6.54 518 (d, J = 1.5 Hz, 2 H); 6.56 (d, J = 8.8 Hz, 2 H); 6.68 (t, J = 1.5 Hz, 1 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.75 (d, J = 8.5 Hz, 1 H); 9.30 (s, 1 H) 65 ##STR00091## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- methoxy- 2- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.00 to 2.25 (m, 5 H); 2.37 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 2 H); 2.62 (m, 1 H); 2.72 to 2.81 (m, 2 H); 3.78 (s, 3 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.55 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.00 (dd, J = 2.8 and 8.7 Hz, 1 556 H); 7.17 (d, J = 8.7 Hz, 1 H); 7.27 (d J = 2.8 Hz, 1 H); 9.39 (s, 1 H) 66 ##STR00092## 6-[4- (difluoromethoxy)- 3- fluoro- phenyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 to 2.28 (m, 3 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.54 (m, 1 H); 2.68 (m, 3 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.56 (s, 2 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 6.96 (d, J = 8.6 Hz, 1 542 H); 7.07 to 7.18 (m, 2 H); 7.19 (t, J = 73.8 Hz, 1 H); 9.41 (s, 1 H) 67 ##STR00093## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- methyl-4- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.01 to 2.25 (m, 5 H); 2.21 (s, 3 H); 2.36 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.59 to 2.81 (m, 4 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.58 (m, 4 H); 6.66 (d, J = 8.8 Hz, 2 H); 6.72 (s, 1 H); 7.22 (dd, J = 3.0 and 8.6 Hz, 1 H); 7.38 (d, J = 8.6 Hz, 1 H); 7.47 (s, 1 H); 540 9.40 (s, 1 H) 68 ##STR00094## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[6- (trifluoromethyl)- 3- pyridyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.20 (m, 1 H); 2.31 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 1 H); 2.70 (t, J = 7.2 Hz, 2 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.59 (s, 2 H); 6.66 (d, J = 8.8 Hz, 2 527 H); 6.72 (s, 1 H); 6.76 (d, J = 8.8 Hz, 2 H); 7.72 (d, J = 8.2 Hz, 1 H); 7.83 (dd, J = 2.5 and 8.2 Hz, 1 H); 8.40 (d, J = 2.5 Hz, 1 H); 9.50 (s, 1 H) 69 ##STR00095## 6-[4- (difluoromethoxy) phenyl]- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.05 to 2.29 (m, 3 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.53 (m, 1 H); 2.65 (m, 3 H); 2.79 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.55 (d, J = 1.5 Hz, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.73 (s, 1 H); 6.97 (d, J = 8.5 Hz, 2 H); 7.25 524 (d, J = 8.5 Hz, 2 H); 7.29 (t, J = 74.5 Hz, 1 H); 9.38 (s, 1 H) 70 ##STR00096## 6-(2,2- dimethylindolin- 5-yl)- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.13 (m, 2 H); 1.18 (s, 6 H); 1.65 to 1.88 (m, 3 H); 2.01 (m, 2 H); 2.20 (m, 3 H); 2.33 to 2.70 (m, 7 H); 2.79 (m, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 5.34 (s, 1 H); 6.19 (d, J = 8.0 Hz, 1 H); 6.48 to 6.75 (m, 9 H); 9.28 (s, 1 H) 527 71 ##STR00097## 6-(6- ethoxy-2- fluoro-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 0.77 (t, J = 7.1 Hz, 3 H); 1.71 (m, 1 H); 1.81 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.17 (t, J = 7.0 Hz, 2 H); 2.22 (m, 1 H); 2.35 to 2.93 (m, 6 H); 2.68 (t, J = 7.0 Hz, 2 H); 4.18 (q, J = 7.1 Hz, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.55 (s, 2 H); 6.59 (d, J = 8.3 Hz, 1 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.74 (d, 521 J = 8.8 Hz, 2 H); 7.58 (dd, J = 8.3 and 10.2 Hz, 1 H); 9.42 (s, 1 H) 72 ##STR00098## 6-(4-tert- butylphenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.21 (s, 9 H); 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.20 (m, 1 H); 2.25 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.55 (m, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.69 514 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.03 (d, J = 8.6 Hz, 2 H); 7.18 (d, J = 8.6 Hz, 2 H); 9.35 (s, 1 H) 73 ##STR00099## 5-[4-[(3S)- 1-(3- fluoropropyl] pyrrolidin-3- yl]oxyphenyl]- 6- (1,2,3,4- tetrahydroquinolin- 6- yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 to 1.88 (m, 5 H); 2.00 (m, 2 H); 2.19 (m, 3 H); 2.39 (m, 1 H); 2.43 to 2.52 (m, 4 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.59 to 2.69 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.10 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 5.52 (s, 1 H); 6.17 (d, J = 8.3 Hz, 1 H); 6.52 (m, 2 H); 6.59 to 6.66 (m, 5 H); 6.74 513 (d, J = 8.8 Hz, 2 H); 9.28 (s, 1 H) 74 ##STR00100## 6-(3- ethoxyphenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.20 (t, J = 7.1 Hz, 3 H); 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.19 (m, 1 H); 2.25 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.66 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.82 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.55 (m, 2 H); 6.59 to 6.71 (m, 6 H); 502 6.73 (d, J = 8.8 Hz, 2 H); 7.07 (t, J = 8.0 Hz, 1 H); 9.37 (s, 1 H) 75 ##STR00101## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.28 (t, J = 7.0 Hz, 6 H); 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.10 (m, 2 H); 2.20 (m, 1 H); 2.27 (t, J = 7.0 Hz, 2 H); 2.41 (m, 1 H); 2.48 (m, 2 H); 2.57 (m, 1 H); 2.68 (m, 1 H); 2.78 (t, J = 7.0 Hz, 2 H); 2.82 (m, 1 H); 4.17 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.73 (d, J = 8.8 Hz, 2 H); 542 6.79 (d, J = 8.6 Hz, 1 H); 7.03 (ddd, J = 1.3 and 2.8 and 8.6 Hz, 1 H); 7.16 (d, J = 2.8 Hz, 1 H); 7.18 (d, J = 8.9 Hz, 2 H); 7.28 (d, J = 8.9 Hz, 2 H) 76 ##STR00102## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- methoxyphenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.17 (m, 1 H); 2.23 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.65 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.69 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.54 (s, 2 H); 6.60 (d, J = 8.7 Hz, 2 H); 6.68 (s, 1 H); 6.72 (m, 4 H); 7.04 (d, 488 J = 8.7 Hz, 2 H); 9.31 (s, 1 H) 77 ##STR00103## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- methoxyphenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.19 (m, 1 H); 2.26 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.49 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.55 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 488 H); 6.62 to 6.70 (m, 4 H); 6.73 (d, J = 8.8 Hz, 2 H); 7.08 (m, 1 H); 9.38 (s, 1 H) 78 ##STR00104## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.19 (m, 1 H); 2.29 (m, 2 H); 2.39 (m, 1 H); 2.48 to 2.55 (m, 3 H); 2.60 to 2.71 (m, 3 H); 2.79 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.57 (m, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.72 (m, 3 H); 7.32 (d, J = 8.3 Hz, 2 H); 7.52 (d, J = 8.3 Hz, 2 H); 9.43 (s, 1 H) 526 79 ##STR00105## 6-[4- (difluoromethoxy)- 3- fluoro- phenyl]-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 1 H); 2.79 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.40 (d, J = 8.6 Hz, 1 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.73 (t, J = 8.6 Hz, 1 H); 6.77 (d, J = 8.8 Hz, 2 H); 6.98 (dd, J = 2.5 and 8.5 Hz, 1 H); 7.10 to 7.18 (m, 2 H); 7.19 (t, J = 73.5 Hz, 1 H); 9.87 (m, 1 H) 560 80 ##STR00106## 6-(5- chloro-6- ethoxy-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.29 (t, J = 7.1 Hz, 3 H); 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.15 to 2.18 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.57 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.67 (m, 3 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.30 (q, J = 7.1 Hz, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.57 (s, 2 H); 6.68 (d, 537 J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.78 (d, J = 8.8 Hz, 2 H); 7.60 (d, J = 2.4 Hz, 1 H); 7.80 (d, J = 2.4 Hz, 1 H); 9.42 (s, 1 H) 81 ##STR00107## 6-(2- ethylphenyl)- 5-[4- [(3S)-1-(3- fluoropropyl)
pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.08 (t, J = 7.6 Hz, 3 H); 1.65 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.01 to 2.22 (m, 5 H); 2.35 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.58 to 2.82 (m, 6 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.54 (d, J = 8.8 Hz, 2 H); 6.57 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.98 to 7.05 (m, 2 H); 7.11 (m, 1 486 H); 7.18 (d, J = 8.2 Hz, 1 H); 9.35 (s, 1 H) 82 ##STR00108## 6-(6- ethoxy-2- fluoro-3- pyridyl)-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.28 (t, J = 7.1 Hz, 3 H); 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.13 to 2.25 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.79 (m, 3 H); 4.19 (q, J = 7.1 Hz, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.40 (d, J = 8.3 Hz, 1 H); 6.60 (dd, J = 1.4 and 8.3 Hz, 1 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.74 (m, 3 H); 7.59 (dd, J = 8.3 and 10.2 Hz, 1 H); 9.89 (m, 1 H) 539 83 ##STR00109## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- methoxypyrimidin- 5- yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.16 to 2.30 (m, 3 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.56 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.68 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.86 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.57 (s, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.80 (d, J = 8.8 Hz, 2 490 H); 8.31 (s, 2 H); 9.46 (s, 1 H) 84 ##STR00110## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- (trifluoromethyl) pyrimidin- 5-yl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 to 1.86 (m, 3 H); 2.10 (m, 2 H); 2.21 (m, 1 H); 2.30 to 2.58 (m, 6 H); 2.61 to 2.82 (m, 4 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.79 (m, 1 H); 6.59 (s, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.72 (s, 1 H); 6.80 (d, J = 8.8 Hz, 2 H); 8.74 (s, 2 H); 9.56 (s, 1 H) 528 85 ##STR00111## 2-fluoro-4- [5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]benzonitrile A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.55 (m, 1 H); 2.79 (m, 3 H); 2.80 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.58 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.73 (d, J = 8.8 Hz, 2 H); 7.10 (d, J = 8.1 Hz, 1 H); 7.28 (d, J = 11.5 501 Hz, 1 H); 7.68 (t, J = 8.1 Hz, 1 H); 9.50 (s, 1 H) 86 ##STR00112## 6-(5- chloro-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.20 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.61 to 2.72 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.59 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 493 H); 6.77 (d, J = 8.8 Hz, 2 H); 7.70 (d, J = 2.5 Hz, 1 H); 8.18 (d, J = 2.5 Hz, 1 H); 8.31 (d, J = 2.5 Hz, 1 H); 9.49 (s, 1 H) 87 ##STR00113## 6-[6- (difluoromethoxy)- 3- pyridyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.20 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.56 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.68 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.56 (s, 2 H); 6.67 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.78 525 (d, J = 8.8 Hz, 2 H); 6.91 (d, J = 8.6 Hz, 1 H); 7.62 (t, J = 73.1 Hz, 1 H); 7.68 (dd, J = 2.0 and 8.6 Hz, 1 H); 7.96 (d, J = 2.0 Hz, 1 H); 9.46 (s, 1 H) 88 ##STR00114## 6-(2,5- difluoro-4- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.15 (t, J = 7.0 Hz, 2 H); 2.20 (m, 1 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.72 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.79 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.55 (s, 2 H); 6.62 (d, J = 8.8 524 Hz, 2 H); 6.70 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 6.93 (dd, J = 7.3 and 11.2 Hz, 1 H); 6.99 (dd, J = 7.1 and 12.1 Hz, 1 H); 9.40 (s, 1 H) 89 ##STR00115## 6-(2- chloro-4- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.12 to 2.23 (m, 3 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.62 (m, 1 H); 2.70 to 2.82 (m, 3 H); 3.71 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.55 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (dd, J = 2.6 and 522 8.8 Hz, 1 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.97 (d, J = 2.6 Hz, 1 H); 7.00 (d, J = 8.8 Hz, 1 H); 9.38 (s, 1 H) 90 ##STR00116## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(5- fluoro-3- pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.09 (m, 2 H); 2.20 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.1 and 10.4 Hz, 1 H); 2.61 to 2.72 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.58 (m, 2 H); 6.66 (d, J = 8.8 Hz, 2 H); 6.71 477 (s, 1 H); 6.77 (d, J = 8.8 Hz, 2 H); 7.52 (td, J = 2.9 and 10.4 Hz, 1 H); 8.09 (t, J = 2.9 Hz, 1 H); 8.27 (d, J = 2.9 Hz, 1 H); 9.48 (s, 1 H) 91 ##STR00117## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(6- methoxy- 4-methyl- 3-pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 to 2.23 (m, 5 H); 2.12 (s, 3 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 2 H); 2.79 (m, 2 H); 3.75 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.55 (s, 2 H); 6.57 (s, 1 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 503 6.71 (s, 1 H); 7.71 (s, 1 H); 9.39 (s, 1 H) 92 ##STR00118## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- methoxy- 2,5- dimethyl- phenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.98 to 2.22 (m, 5 H); 2.01 (s, 3 H); 2.04 (s, 3 H); 2.37 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H, 1 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.71 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.54 (s, 2 H); 6.55 (d, J = 8.8 Hz, 2 H); 6.61 516 (s, 1 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.81 (s, 1 H); 9.34 (s, 1 H) 93 ##STR00119## 6-(2,3- difluoro-4- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.14 to 2.25 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.61 to 2.71 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.82 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.57 (m, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 524 6.72 (d, J = 8.8 Hz, 2 H); 6.80 to 6.94 (m, 2 H); 9.43 (s, 1 H) 94 ##STR00120## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethylsulfanyl) phenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.57 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 558 6.71 (d, J = 8.8 Hz, 2 H); 7.25 (d, J = 8.4 Hz, 2 H); 7.50 (d, J = 8.4 Hz, 2 H); 9.46 (s, 1 H) 95 ##STR00121## 6-(3- chloro-4- ethoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.31 (t, J = 7.1 Hz, 3 H); 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.15 to 2.27 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.1 and 10.4 Hz, 1 H); 2.66 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.03 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.54 (s, 2 H); 6.65 (d, 536 J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 6.92 (d, J = 8.6 Hz, 1 H); 7.03 (dd, J = 2.6 and 8.6 Hz, 1 H); 7.11 (d, J = 2.6 Hz, 1 H); 9.38 (s, 1 H) 96 ##STR00122## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(5- methyl-3- pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.20 (s, 3 H); 2.26 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.61 to 2.71 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.57 (m, 2 H); 6.63 (d, J = 8.8 Hz, 2 473 H); 6.70 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 7.42 (t, J = 2.3 Hz, 1 H); 8.01 (d, J = 2.3 Hz, 1 H); 8.10 (d, J = 2.3 Hz, 1 H); 9.43 (s, 1 H) 97 ##STR00123## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(6- methoxy- 2-methyl- 3-pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 to 2.24 (m, 5 H); 2.20 (s, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.71 (t, J = 7.2 Hz, 2 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 3.77 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.51 (d, J = 8.4 Hz, 1 H); 6.55 (s, 2 H); 6.60 (d, J = 9.0 Hz, 2 503 H); 6.69 (d, J = 9.0 Hz, 2 H); 6.71 (s, 1 H); 7.33 (d, J = 8.4 Hz, 1 H); 9.41 (s, 1 H) 98 ##STR00124## 6-(2,2- dimethyl- 3H- benzofuran- 5-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.35 (s, 6 H); 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.13 to 2.25 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 2.84 (s, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.47 (d, J = 8.3 Hz, 1 H); 6.54 (m, 2 H); 6.61 528 (d, J = 8.8 Hz, 2 H); 6.68 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.81 (dd, J = 2.5 and 8.3 Hz, 1 H); 6.91 (d, J = 2.5 Hz, 1 H); 9.33 (s, 1 H) 99 ##STR00125## 6-(5- chloro-6- methoxy- 3-pyridyl)- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.20 (m, 1 H); 2.33 (t, J = 7.2 Hz, 2 H); 2.39 (m, 1 H); 2.48 (t, J = 7.3 Hz, 2 H); 2.55 (dd, J = 3.2 and 10.5 Hz, 1 H); 2.61 to 2.70 (m, 3 H); 2.80 (dd, J = 6.1 and 10.5 Hz, 1 H); 3.86 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.79 (m, 1 H); 6.56 (s, 2 H); 6.69 (d, J = 8.9 Hz, 2 523 H); 6.70 (s, 1 H); 6.79 (d, J = 8.9 Hz, 2 H); 7.61 (dd, J = 2.3 Hz, 1 H); 7.82 (d, J = 2.3 Hz, 1 H); 9.43 (s, 1 H) 100 ##STR00126## 6-(4-ethoxy- 2,5- dimethyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.31 (t, J = 7.1
Hz, 3 H); 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.01 (s, 6 H); 2.03 (m, 2 H); 2.10 (m, 2 H); 2.19 (m, 1 H); 2.37 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.61 (m, 1 H); 2.70 (m, 2 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 3.95 (m, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.53 530 (s, 2 H); 6.55 (d, J = 8.8 Hz, 2 H); 6.60 (s, 1 H); 6.67 (d, J = 8.8 Hz, 2 H); 6.69 (t, J = 1.0 Hz, 1 H); 6.80 (s, 1 H); 9.33 (s, 1 H) 101 ##STR00127## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(6- methoxy- 5-methyl- 3-pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.03 (s, 3 H); 2.05 (m, 2 H); 2.15 to 2.25 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.79 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.54 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 503 6.76 (d, J = 8.8 Hz, 2 H); 7.32 (d, J = 3.0 Hz, 1 H); 7.67 (d, J = 3.0 Hz, 1 H); 9.39 (s, 1 H) 102 ##STR00128## 6-(5-fluoro- 6- methoxy- 3-pyridyl)- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.13 to 2.28 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.68 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.86 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.56 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.79 507 (d, J = 8.8 Hz, 2 H); 7.42 (dd, J = 2.0 and 11.9 Hz, 1 H); 7.68 (d, J = 2.0 Hz, 1 H); 9.42 (s, 1 H) 103 ##STR00129## 6-(3- chloro-4- ethoxy-2- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.33 (t, J = 7.2 Hz, 3 H); 1.68 (m, 1 H); 1.81 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.16 to 2.23 (m, 3 H); 2.40 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.68 (t, J = 7.0 Hz, 2 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.10 (q, J = 7.2 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 554 6.56 (m, 2 H); 6.63 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.83 (d, J = 9.0 Hz, 1 H); 7.03 (t, J = 9.0 Hz, 1 H); 9.40 (s, 1 H) 104 ##STR00130## 6-(2-fluoro- 6- methoxy- 3-pyridyl)- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.13 to 2.25 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.1 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.78 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.56 (s, 2 H); 6.61 (d, J = 8.2 Hz, 1 H); 6.64 (d, 507 J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 7.59 (dd, J = 8.2 and 10.1 Hz, 1 H); 9.44 (s, 1 H) 105 ##STR00131## 6-(3,5- difluoro-4- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.21 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.2 and 10.5 Hz, 1 H); 2.66 (m, 3 H); 2.80 (dd, J = 6.2 and 10.5 Hz, 1 H); 3.85 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.79 (m, 1 H); 6.55 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.76 (d, J = 8.8 Hz, 2 524 H); 6.83 (d, J = 10.0 Hz, 2 H); 9.44 (s, 1 H) 106 ##STR00132## 6-(1- ethylindolin- 5-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.07 (t, J = 7.4 Hz, 3 H); 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.00 (m, 2 H); 2.20 (m, 3 H); 2.38 (m, 1 H); 2.47 (m, 2 H); 2.54 (m, 1 H); 2.65 (m, 3 H); 2.74 (t, J = 7.2 Hz, 2 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.02 (q, J = 7.4 Hz, 2 H); 3.22 (t, J = 7.2 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.25 (d, J = 8.2 Hz, 1 527 H); 6.52 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.65 (s, 1 H); 6.73 (m, 3 H); 6.81 (s, 1 H); 9.29 (s, 1 H) 107 ##STR00133## 6-(2- ethoxypyrimidin- 5- yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.28 (t, J = 7.1 Hz, 3 H); 1.65 to 1.88 (m, 3 H); 2.08 (m, 2 H); 2.16 to 2.29 (m, 3 H); 2.35 to 2.58 (m, 4 H); 3.68 (m, 3 H); 2.80 (m, 1 H); 4.27 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.57 (s, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.80 (d, J = 8.8 Hz, 2 H); 8.29 (s, 2 H); 9.46 (s, 1 H) 504 108 ##STR00134## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(6- methoxy- 3-pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 to 2.27 (m, 3 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.80 (dd, J = 6.4 and 10.3 Hz, 1 H); 3.78 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.56 (s, 2 H); 6.63 (m, 3 H); 6.70 (s, 1 H); 6.75 (d, J = 8.8 489 Hz, 2 H); 7.45 (dd, J = 2.5 and 8.7 Hz, 1 H); 7.89 (d, J = 2.5 Hz. 1 H); 9.40 (s, 1 H) 109 ##STR00135## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- methoxy- 4-pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.15 to 2.27 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.5 Hz, 1 H); 2.64 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.76 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.51 (s, 1 H); 6.57 (m, 2 H); 6.67 (m, 3 H); 6.70 (s, 1 H); 489 6.76 (d, J = 8.8 Hz, 2 H); 7.92 (d, J = 5.4 Hz, 1 H); 9.47 (s, 1 H) 110 ##STR00136## 6-(6- ethoxy-5- methyl-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.36 (t, J = 7.3 Hz, 3 H); 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.02 (s, 3 H); 2.04 (m, 2 H); 2.15 to 2.25 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.1 and 10.4 Hz, 1 H); 2.66 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.22 (q, J = 7.3 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.55 (s, 2 H); 517 6.64 (d, J = 8.8 Hz, 2 H); 6.79 (s, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 7.32 (d, J = 2.6 Hz, 1 H); 7.62 (d, J = 2.6 Hz, 1 H); 9.39 (s, 1 H) 111 ##STR00137## 6-(3-fluoro- 4- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.15 to 2.27 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (m, dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.77 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.54 (s, 2 H); 6.63 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.74 506 (d, J = 8.8 Hz, 2 H); 6.86, 85 to 6.92 (m, 2 H); 6.95 (t, J = 8.7 Hz, 1 H); 9.38 (s, 1 H) 112 ##STR00138## 6-(2,4- difluoro-3- methoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.18 (m, 3 H); 2.39 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.69 (t, J = 7.0 Hz, 2 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.74 (s, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.58 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.71 (m, 3 H); 6.90 (ddd, J = 6.2 524 and 8.5 and 9.0 Hz, 1 H); 6.98 (ddd, J = 2.2 and 9.0 and 11.0 Hz, 1 H); 9.43 (s, 1 H) 113 ##STR00139## 6-(4- chloro-3- methyl- phenyl)-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.18 (m, 1 H); 2.20 (s, 3 H); 2.26 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 1 H); 2.73 to 2.82 (m, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.40 (d, J = 8.7 Hz, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.72 (t, J = 8.7 Hz, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 6.91 (dd, J = 2.5 and 8.5 Hz, 1 H); 7.13 (d, J = 2.5 Hz, 1 H); 7.18 (d, J = 8.5 Hz, 1 H); 524 9.80 (m, 1 H) 114 ##STR00140## 1-fluoro-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.79 (m, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.40 (d, J = 8.3 Hz, 1 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.72 (m, 3 H); 7.15 (d, J = 8.9 Hz, 2 H); 7.23 (d, J = 8.9 Hz, 2 H); 9.79 (s, 1 H) 560 115 ##STR00141## 1-fluoro-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- fluoro-2- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.18 (m, 3 H); 2.37 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.59 to 2.81 (m, 3 H); 2.99 (s, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.40 (d, J = 8.6 Hz, 1 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.74 (t, J = 8.6 Hz, 1 H); 7.21 (dd, J = 6.0 and 8.9 Hz, 1 H); 7.32 (dt, J = 3.0 and 8.9 Hz, 1 H); 7.60 (dd, J = 3.0 and 9.5 Hz, 1 H); 9.80 (s, 1 H) 562 116 ##STR00142## 6-[4- (difluoromethoxy)- 2- fluoro- phenyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.12 to 2.25 (m, 3 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.59 to 2.72 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.56 (m, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 542 Hz, 2 H); 6.87 (dd, J = 2.7 and 8.7 Hz, 1 H); 6.97 (dd, J = 2.7 and 10.7 Hz, 1 H); 7.20 (t, J = 8.7 Hz, 1 H); 7.23 (t, J = 73.8 Hz, 1 H); 9.43 (s, 1 H) 117 ##STR00143## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- fluoro-4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.19 (m, 3 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.63 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.58 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.72 (s, 1 H); 560 7.08 (d, J = 8.6 Hz, 1 H); 7.22 (d, J = 10.2 Hz, 1 H); 7.29 (t, J = 8.6 Hz, 1 H); 9.44 (s, 1 H) 118 ##STR00144## 6-(2,6- dichloro-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, d ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 to 2.17 (m, 5 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.63 (m, 1 H); 2.78 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.57 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.73 (d, J = 8.8 Hz, 2 H); 7.41 (d, J = 8.7 Hz, 1 H); 7.68 (d, J = 8.7 Hz, 1 H); 527 9.99 (s, 1 H) 119 ##STR00145## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (2,2,2- trifluoroethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.13 to 2.27 (m, 3 H); 2.40 (m, 1 H); 2.48 (m, 2 H); 2.55 (m, 1 H); 2.65 (m, 3 H); 2.80 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.68 (q, J = 8.9 Hz, 2 H); 4.74 (m, 1 H); 6.54 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.85 (d, J = 9.0 Hz, 2 556 H); 7.08 (d, J = 9.0 Hz, 2 H); 9.34 (s, 1 H)
120 ##STR00146## 6-(4- ethoxy- 3,5- difluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.34 (t, J = 7.2 Hz, 3 H); 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.27 to 2.37 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.56 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.67 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.09 (q, J = 7.2 Hz, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.55 (s, 2 H); 6.67 (d, 538 J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 6.82 (d, J = 9.9 Hz, 2 H); 9.41 (s, 1 H) 121 ##STR00147## 6-(4- chloro-2- fluoro- phenyl)-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.19 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.79 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.42 (d, J = 8.0 Hz, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.72 (m, 3 H); 7.12 (dd, J = 2.5 and 8.3 Hz, 1 H); 7.20 (t, J = 8.3 Hz, 1 H); 7.29 (dd, J = 2.2 and 9.8 Hz, 1 H); 9.87 (s, 1 H) 528 122 ##STR00148## 6-(2- chloro-3- fluoro- phenyl)-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.19 (m, 3 H); 2.36 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.62 (m, 1 H); 2.77 (m, 2 H); 2.99 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.41 (d, J = 8.6 Hz, 1 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.75 (t, J = 8.6 Hz, 1 H); 7.00 (m, 1 H); 7.20 (m, 2 H); 9.87 (s, 1 H) 528 123 ##STR00149## 1-fluoro-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- methyl-4- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.18 (m, 3 H); 2.22 (s, 3 H); 2.35 (m, 1 H); 2.43 (t, J = 7.2 Hz, 2 H); 2.51 (m, 1 H); 2.62 (m, 1 H); 2.77 (m, 1 H); 2.87 (m, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.41 (d, J = 8.8 Hz, 1 H); 6.58 (d, J = 8.8 Hz, 2 H); 6.67 (d, J = 8.8 Hz, 2 H); 6.75 (t, J = 8.8 Hz, 1 H); 7.22 (dd, J = 3.1 and 7.9 Hz, 1 H); 7.38 (d, J = 7.9 Hz, 1 H); 7.48 (s, 1 H); 9.82 (s, 1 H) 558 124 ##STR00150## 1-fluoro-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.19 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.79 (m, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.41 (d, J = 8.7 Hz, 1 H); 6.63 (d, J = 8.8 Hz, 2 H); 6.74 (t, J = 8.7 Hz, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 7.35 (d, J = 8.7 Hz, 2 H); 7.53 (d, J = 8.7 Hz, 2 H); 9.88 (s, 1 H) 544 125 ##STR00151## 6-(6- ethoxy-2- methyl-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.25 (t, J = 7.1 Hz, 3 H); 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 to 2.23 (m, 5 H); 2.15 (s, 3 H); 2.33 to 2.58 (m, 4 H); 2.60 to 2.73 (m, 4 H); 4.20 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.48 (d, J = 8.7 Hz, 1 H); 6.55 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 7.32 517 (d, J = 8.7 Hz, 1 H); 9.38 (s, 1 H) 126 ##STR00152## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1- methylindol- 5-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.17 (m, 1 H); 2.31 (t, J = 7.0 Hz, 2 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.69 (t, J = 7.0 Hz, 2 H); 2.76 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.71 (s, 3 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.69 (m, 1 H); 6.27 (d, J = 3.2 Hz, 1 H); 6.63 (d, J = 8.8 Hz, 2 511 H); 6.65 (s, 2 H); 6.69 (s, 1 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.93 (dd, J = 1.8 and 8.6 Hz, 1 H); 7.19 (d, J = 8.6 Hz, 1 H); 7.22 (d, J = 3.2 Hz, 1 H); 7.32 (d, J = 1.8 Hz, 1 H); 9.32 (s, 1 H) 127 ##STR00153## 6-(6- chloro-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.21 (m, 1 H); 2.27 (t, J = 7.0 Hz, 2 H); 2.35 to 2.59 (m, 4 H); 2.69 (m, 3 H); 2.81 (m, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.57 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.77 (d, J = 8.8 Hz, 2 H); 7.34 (d, J = 8.3 Hz, 1 H); 7.62 (dd, J = 2.6 and 8.3 Hz, 1 493 H); 8.08 (d, J = 2.6 Hz, 1 H); 9.46 (s, 1 H) 128 ##STR00154## 2-fluoro-4- [5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]-N- methyl- benzamide A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 to 2.30 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.67 (m, 3 H); 2.74 (d, J = 4.8 Hz, 3 H); 2.80 (dd, =6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.56 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 533 6.76 (d, J = 8.8 Hz, 2 H); 6.98 (dd, J = 1.6 and 4.8 Hz, 1 H); 7.01 (s, 1 H); 7.42 (t, J = 8.1 Hz, 1 H); 8.09 (m, 1 H); 9.43 (s, 1 H) 129 ##STR00155## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- fluoro-6- (trifluoromethyl)- 3- pyridyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.10 (m, 2 H); 2.20 (m, 1 H); 2.26 (t, J = 7.0 Hz, 2 H); 2.40 (m, 1 H); 2.48 (t, J = 7.0 Hz, 2 H); 2.54 (m, 1 H); 2.66 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H); 2.79 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.59 (s, 2 H); 6.66 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.74 (s, 1 H); 545 7.74 (dd, J = 17 and 8.2 Hz, 1 H); 8.02 (t, J = 8.2 Hz, 1 H); 9.50 (s, 1 H) 130 ##STR00156## 6-[4-(2- fluoroethoxy) phenyl]- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.15 to 2.27 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.16 (dm, J = 30.3 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.69 (dm, J = 48.6 Hz, 2 H); 4.74 (m, 1 H); 6.54 (s, 2 H); 6.61 520 (d, J = 8.8 Hz, 2 H); 6.68 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.76 (d, J = 8.9 Hz, 2 H); 7.04 (d, J = 8.9 Hz, 2 H); 9.31 (s, 1 H) 131 ##STR00157## 6-(4- ethoxy- 2,3- dimethyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 0.80 (t, J = 7.1 Hz, 3 H); 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.03 (s, 3 H); 2.10 (s, 3 H); 2.12 to 2.22 (m, 3 H); 2.25 to 2.58 (m, 4 H); 2.63 (m, 2 H); 2.79 (m, 2 H); 3.92 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.54 (d, J = 8.8 Hz, 2 H); 6.56 (s, 2 H); 6.59 (d, J = 8.6 Hz, 1 H); 6.65 (d, J = 8.8 Hz, 530 2 H); 6.69 (s, 1 H); 6.72 (d, J = 8.6 Hz, 1 H); 9.30 (s, 1 H) 132 ##STR00158## 6-[6- ethoxy-5- (trifluoromethyl)- 3- pyridyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.28 (t, J = 7.1 Hz, 3 H); 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.20 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.37 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.79 (m, 1 H); 571 6.58 (m, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.71 (m, 1 H); 6.78 (d, J = 8.8 Hz, 2 H); 7.68 (d, J = 2.7 Hz, 1 H); 8.15 (d, J = 2.7 Hz, 1 H); 9.43 (s, 1 H) 133 ##STR00159## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- methyl- 2,3- dihydro- 1,4- benzoxazin- 7-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 (m, 1 H); 1.81 (dm, J = 25.3 Hz, 2 H); 2.00 (m, 2 H); 2.15 to 2.25 (m, 3 H); 2.40 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.56 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.76 (s, 3 H); 2.81 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.17 (m, 2 H); 4.12 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.42 (d, J = 2.2 Hz, 1 H); 529 6.47 (d, J = 8.5 Hz, 1 H); 6.52 (s, 2 H); 6.53 (dd, J = 2.2 and 8.5 Hz, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.65 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 9.28 (s, 1 H) 134 ##STR00160## 6-(2,2- difluoro- 1,3- benzodioxol- 5-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.25 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.61 to 2.71 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.56 (s, 2 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 538 6.75 (d, J = 8.8 Hz, 2 H); 6.92 (dd, J = 2.0 and 8.5 Hz, 1 H); 7.25 (d, J = 2.0 Hz, 1 H); 7.28 (d, J = 8.5 Hz, 1 H); 9.39 (s, 1 H) 135 ##STR00161## 4-ethyl-6- [5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro-7H- benzo[7] annulen-6- yl]-1,4- benzoxazin- 3-one A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 0.80 (t, J = 7.1 Hz, 3 H); 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.60 to 2.72 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.67 (q, J = 7.1 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.55 (s, 2 H); 4.73 (m, 1 H); 6.55 (s, 557 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.78 (d, J = 8.8 Hz, 2 H); 6.82 (s, 1 H); 6.85 (s, 2 H); 9.38 (s, 1 H) 136 ##STR00162## 6-[2- chloro-4- (trifluoromethoxy) phenyl]-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.19 (m, 3 H); 2.36 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.78 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.57 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 7.19 (d, J = 8.3 Hz, 1 H); 7.25 (d, J = 8.3 Hz, 576 1 H); 7.50 (s, 1 H); 9.45 (s, 1 H) 137 ##STR00163## 6-[4- (difluoromethoxy)- 3,5- difluoro- phenyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.16 to 2.28 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.67 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.57 (s, 2 H); 6.68 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.78 (d, J = 8.8 Hz, 2 560 H); 6.99 (d, J = 8.4 Hz, 2 H); 7.19 (t, J = 72.5 Hz, 1 H); 9.38 (s, 1 H) 138 ##STR00164## 6-(4-tert- butylphenyl)- 1-fluoro- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.22 (s, 9 H); 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.19 (m, 1 H); 2.25 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.65 (m, 1 H); 2.77 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.39 (d, J = 8.5 Hz, 1 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.72 (m, 3 H); 7.04 (d, J = 8.5 Hz, 2 H); 7.19 (d, J = 8.5 Hz, 2 H); 9.74 (s, 1 H) 532 139 ##STR00165## 6-(6- ethoxy-4- methyl-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.25 (t, J = 7.2 Hz, 3 H); 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.01 to 2.25 (m, 5 H); 2.12 (s, 3 H); 2.35 to 2.88 (m, 8 H); 4.19 (m, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.54 (s, 2 H); 6.56 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 7.70 (s,
1 H); 9.38 (s, 1 H) 517 140 ##STR00166## 6-(3- chloro-4- ethoxy-5- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.25 (t, J = 7.2 Hz, 3 H); 1.70 (m, 1 H); 1.81 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 to 2.17 (m, 3 H); 2.39 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.05 (q, J = 7.2 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.56 (m, 2 H); 6.67 (d, 554 J = 8.8 Hz, 2 H); 6.68 (s, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 6.97 (dd, J = 2.5 and 12.5 Hz, 1 H); 6.99 (t, J = 2.5 Hz, 1 H); 9.42 (s, 1 H) 141 ##STR00167## 6-(2- aminopyrimidin- 5- yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.17 to 2.26 (m, 3 H); 2.40 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.57 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.82 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.43 (s, 2 H); 6.55 (s, 2 H); 6.68 (s, 1 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.81 475 (d, J = 8.8 Hz, 2 H); 7.95 (s, 2 H); 9.34 (s, 1 H) 142 ##STR00168## 6-[4- (difluoromethyl) phenyl]- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.19 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.55 (m, 1 H); 2.68 (m, 3 H); 2.80 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.56 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.92 (t, J = 56.1 Hz, 1 H); 7.24 (d, J = 8.3 508 Hz, 2 H); 7.36 (d, J = 8.3 Hz, 2 H); 9.40 (s, 1 H) 143 ##STR00169## 6-[4- (difluoromethoxy) phenyl]-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.20 (m, 1 H); 2.26 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 1 H); 2.78 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.40 (d, J = 8.6 Hz, 1 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.73 (d, J = 8.8 Hz, 3 H); 6.98 (d, J = 8.8 Hz, 2 H); 7.18 (d, J = 8.8 Hz, 2 H); 7.19 (t, J = 74.3 Hz, 1 H); 9.80 (s, 1 H) 542 144 ##STR00170## 6-[3,5- difluoro-4- (trifluoromethoxy) phenyl]-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.20 (m, 1 H); 2.25 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.67 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.79 (m, 1 H); 6.57 (s, 2 H); 6.69 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.76 578 (d, J = 8.8 Hz, 2 H); 7.10 (d, J = 9.8 Hz, 2 H); 9.48 (s, 1 H) 145 ##STR00171## 6-[4- (difluoromethoxy)- 2- methyl- phenyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.02 to 2.23 (m, 5 H); 2.12 (s, 3 H); 2.37 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.59 to 2.81 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.56 (s, 2 H); 6.58 (d, J = 8.8 Hz, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.83 (dd, J = 2.5 and 8.7 Hz, 1 H); 6.91 (d, 538 J = 2.5 Hz, 1 H); 7.05 (d, J = 8.7 Hz, 1 H); 7.15 (t, J = 74.5 Hz, 1 H); 9.38 (s, 1 H) 146 ##STR00172## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- methyl-4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.02 to 2.13 (m, 5 H); 2.16 (s, 3 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.59 to 2.81 (m, 4 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 (d, J = 8.8 Hz, 2 H); 6.58 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 7.01 (d, J = 9.1 Hz, 1 H); 7.10 (d, J = 2.8 Hz, 1 556 H); 7.12 (dd, J = 2.9 and 9.1 Hz, 1 H); 9.40 (s, 1 H) 147 ##STR00173## 6-[5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]-4- methyl- 1,4- benzoxazin- 3-one A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.20 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.68 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.00 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.59 (s, 2 H); 4.75 (m, 1 H); 6.58 (m, 2 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.69 (m, 1 H); 543 6.77 (d, J = 8.8 Hz, 2 H); 6.79 (m, 2 H); 6.82 (s, 1 H); 9.39 (s, 1 H) 148 ##STR00174## 6-[5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]-4H-1,4- benzoxazin- 3-one A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.20 (m, 3 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.80 (d, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.51 (s, 2 H); 4.75 (m, 1 H); 6.55 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.64 (s, 1 H); 6.68 to 6.76 529 (m, 5 H); 9.38 (s, 1 H); 10.51 (s, 1 H) 149 ##STR00175## 6-(2,3- dichloro-4- ethoxy- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.31 (t, J = 7.1 Hz, 3 H); 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.12 to 2.25 (m, 3 H); 2.38 (m, 1 H); 2.42 to 2.56 (m, 3 H); 2.60 to 2.85 (m, 4 H); 4.08 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.56 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.92 (d, J = 8.9 Hz, 1 570 H); 7.02 (d, J = 8.9 Hz, 1 H); 9.40 (s, 1 H) 150 ##STR00176## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[3- methyl-4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.13 (s, 3 H); 2.19 (m, 1 H); 2.26 (m, 2 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.57 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 556 6.72 (d, J = 8.8 Hz, 2 H); 7.00 (dd, J = 2.3 and 8.5 Hz, 1 H); 7.17 (d, J = 8.5 Hz, 1 H); 7.25 (d, J = 2.3 Hz, 1 H); 9.40 (s, 1 H) 151 ##STR00177## 6-[3- chloro-4- (trifluoromethoxy) phenyl]- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.19 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.67 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.58 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 576 7.19 (dd, J = 2.2 and 8.6 Hz, 1 H); 7.34 (qd, J = 1.6 and 8.6 Hz, 1 H); 7.36 (d, J = 2.2 Hz, 1 H); 9.44 (s, 1 H) 152 ##STR00178## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(5- quinolyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.57 (m, 1 H); 1.75 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 3 H); 2.21 (m, 1 H); 2.31 (m, 1 H); 2.36 to 2.52 (m, 4 H); 2.58 (m, 1 H); 2.69 (m, 1 H); 2.79 (m, 1 H); 2.90 (m, 1 H); 4.43 (td, J = 6.1 and 47.5 Hz, 2 H); 4.61 (m, 1 H); 6.43 (d, J = 8.8 Hz, 2 H); 6.61 (m, 4 H); 6.75 (d, J = 2.5 Hz, 1 H); 7.31 (d, J = 7.8 Hz, 1 H); 7.48 (dd, J = 4.5 and 509 8.5 Hz, 1 H); 7.59 (m, 1 H); 7.82 (d, J = 8.5 Hz, 1 H); 8.30 (d, J = 8.9 Hz, 1 H); 8.84 (dd, J = 2.0 and 4.5 Hz, 1 H); 9.42 (s, 1 H) 153 ##STR00179## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.20 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.63 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.55 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.73 459 (d, J = 8.8 Hz, 2 H); 7.08 (d, J = 6.3 Hz, 2 H); 8032 (d, J = 6.3 Hz, 2 H); 9.48 (s, 1 H) 154 ##STR00180## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- pyridyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.72 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.57 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 459 6.74 (d, J = 8.8 Hz, 2 H); 7.22 (dd, J = 5.0 and 8.3 Hz, 1 H); 7.57 (td, J = 2.3 and 8.3 Hz, 1 H); 8.23 (d, J = 2.3 Hz, 1 H); 8.27 (dd, J = 2.3 and 5.0 Hz, 1 H); 9.44 (s, 1 H) 155 ##STR00181## 6-[2- chloro-6- (trifluoromethyl)- 3- pyridyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.06 to 2.31 (m, 5 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.64 (m, 1 H); 2.79 (m, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.59 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.73 (m, 3 H); 7.78 (d, J = 9.0 Hz, 1 H); 7.89 (d, J = 9.0 Hz, 1 H); 561 9.49 (s, 1 H) 156 ##STR00182## tert-butyl 6-[5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]-2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]-2,3- dihydro- 1,4- benzoxazine-4- carboxylate A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.34 (s, 9 H); 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.13 to 2.27 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.65 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.71 (t, J = 4.5 Hz, 2 H); 4.15 (t, J = 4.5 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.54 (m, 2 H); 6.58 to 6.65 (m, 4 H); 615 6.68 (s, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 7.58 (s, 1 H); 9.32 (s, 1 H) 157 ##STR00183## 6-[4- (difluoromethylsulfanyl) phenyl]- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m. 2 H); 2.19 (m, 1 H); 2.27 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.66 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.57 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 540 7.20 (d, J = 8.6 Hz, 2 H); 7.36 (d, J = 8.6 Hz, 2 H); 7.42 (t, J = 56.1 Hz, 1 H); 9.41 (s, 1 H) 158 ##STR00184## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6- (1,2,3,4- tetrahydroquinolin- 7- yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 to 1.88 (m, 5 H); 2.01 (m, 2 H); 2.15 (t, J = 7.0 Hz, 2 H); 2.21 (m, 1 H); 2.39 (m, 1 H); 2.43 to 2.79 (m, 8 H); 2.80 (m, 1 H); 3.10 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 5.40 (s, 1 H); 6.15 (d, J = 8.3 Hz, 1 H); 6.25 (s, 1 H); 6.53 (s, 2 H); 6.55 to 6.62 (m, 3 H); 6.67 (s, 1 H); 6.78 (d, J = 8.8 Hz, 2 H); 9.31 (s, 1 H) 513 159 ##STR00185## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.12 (t, J = 7.0 Hz, 2 H); 2.20 (m, 1 H); 2.27 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.5 Hz, 1 H); 2.65 (m, 1 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 2.84 (t, J = 7.0 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.87 (d, J = 8.4 Hz, 570 1 H); 7.19 (d, J = 8.5
Hz, 2 H); 7.28 (d, J = 8.5 Hz, 2 H); 7.74 (dd. J = 1.9 and 8.4 Hz, 1 H); 7.90 (d, J = 1.9 Hz, 1 H); 12.84 (m, 1 H) 160 ##STR00186## 1-fluoro-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- fluoro-4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.03 to 2.24 (m, 3 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.1 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.79 (m, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.41 (d, J = 8.7 Hz, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.75 (t, J = 8.7 Hz, 1 H); 7.09 (dd, J = 3.0 and 8.6 Hz, 1 H); 7.23 (dd, J = 3.0 and 10.3 Hz, 1 H); 7.30 (t, J = 8.6 Hz, 1 H); 9.89 (m, 1 H) 578 161 ##STR00187## 1-fluoro-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethylsulfanyl) phenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.5 Hz, 1 H); 2.64 (m, 1 H); 2.79 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.41 (d, J = 8.7 Hz, 1 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.73 (t, J = 8.7 Hz, 1 H); 7.28 (d, J = 8.4 Hz, 2 576 H); 7.50 (d, J = 8.4 Hz, 2 H); 9.82 (m, 1 H) 162 ##STR00188## 6-(2,4- dichloro-5- fluoro- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.12 to 2.14 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.86 (m, 4 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.57 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 7.29 (d, J = 9.9 Hz, 1 544 H); 7.73 (d, J = 7.1 Hz, 1 H); 9.47 (s, 1 H) 163 ##STR00189## [5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-yl] dihydrogen phosphate 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.82 to 2.11 (m, 5 H); 2.27 (m, 3 H); 2.70 (t, J = 7.0 Hz, 2 H); 2.80 to 3.30 (m, 6 H); 4.50 (td, J = 6.1 and 47.5 Hz, 2 H); 4.83 (m, 1 H); 6.57 (d, J = 8.8 Hz, 1 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.90 (dd, J = 3.3 and 8.8 Hz, 1 H); 7.10 (d, J = 3.3 Hz, 1 H); 7.15 (d, J = 8.8 Hz, 2 H); 7.23 (d, J = 8.8 Hz, 2 H) 622 164 ##STR00190## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(5- methylisoxazol- 4-yl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 to 1.90 (m, 3 H); 1.81 (s, 3 H); 2.04 (m, 2 H); 2.12 (m, 2 H); 2.22 (m, 1 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.58 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.59 to 2.71 (m, 3 H); 2.81 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.80 (m, 1 H); 6.57 (s, 2 H); 6.70 (s, 1 H); 6.71 (d, J = 8.8 Hz, 2 H); 6.80 (d, J = 8.8 Hz, 463 2 H); 8.39 (s, 1 H); 9.42 (s, 1 H) 165 ##STR00191## 6-[4- (difluoromethoxy)- 2- fluoro- phenyl]-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 3 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.79 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.40 (d, J = 8.5 Hz, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.74 (t, J = 8.5 Hz, 1 H); 6.89 (dd, J = 2.5 and 8.7 Hz, 1 H); 6.99 (dd, 560 J = 2.5 and 10.8 Hz, 1 H); 7.22 (t, J = 8.7 Hz, 1 H); 7.25 (t, J = 73.8 Hz, 1 H); 9.83 (s, 1 H) 166 ##STR00192## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethylsulfonyl) phenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.17 (m, 1 H); 2.30 to 2.40 (m, 3 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.72 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.59 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.71 (d, J = 8.8 Hz, 2 H); 6.73 590 (s, 1 H); 7.54 (d, J = 8.8 Hz, 2 H); 7.90 (d, J = 8.8 Hz, 2 H); 9.55 (s, 1 H) 167 ##STR00193## 6-(3,4- dihydro- 2H-1,4- benzoxazin- 6-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol hydrochloride A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.92 to 2.60 (m, 8 H); 2.62 (t, J = 7.0 Hz, 2 H); 3.11 to 4.00 (m, 8 H); 4.11 (m, 2 H); 4.53 (td, J = 6.1 and 47.5 Hz, 2 H); 5.03 (m, 0.5 H); 5.09 (m, 0.5 H); 6.31 (d, J = 8.5 Hz, 1 H); 6.47 (d, J = 8.5 Hz, 1 H); 6.50 to 6.58 (m, 3 H); 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.80 (d, J = 8.8 Hz, 2 H); 9.38 (m, 1 H); 10.50 (m, 0.5 H); 515 11.03 (m, 0.5 H) 168 ##STR00194## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[2- fluoro-4- (trifluoromethoxy) phenyl]-8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.10 to 2.23 (m, 5 H); 2.38 (m, 1 H); 2.42 to 2.55 (m, 3 H); 2.62 (m, 1 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 2.85 (m, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.86 (d, J = 8.2 Hz, 1 H); 7.11 (d, J = 8.6 Hz, 1 H); 7.29 (d, J = 10.2 Hz, 1 H); 7.35 (t, J = 8.6 Hz, 1 H); 7.73 588 (dd, J = 2.0 and 8.2 Hz, 1 H); 7.89 (d, J = 2.0 Hz, 1 H); 12.96 (m, 1 H) 169 ##STR00195## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6- isoxazol-4- yl-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.75 to 1.90 (m, 3 H); 2.11 (m, 2 H); 2.27 (m, 3 H); 2.38 to 2.55 (m, 4 H); 2.60 (t, J = 7.0 Hz, 2 H); 2.62 to 2.75 (m, 2 H); 2.85 (m, 1 H); 4.49 (td, J = 6.1 and 47.5 Hz, 2 H); 4.85 (m, 1 H); 6.55 (m, 2 H); 6.69 (d, J = 1.5 Hz, 1 H); 6.82 (d, J = 8.8 Hz, 2 H); 6.92 (d, J = 8.8 Hz, 2 H); 7.55 (s, 1 H); 8.74 (s, 1 H); 9.43 (s, 1 H) 449 170 ##STR00196## 6-(6- ethoxy-5- fluoro-3- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.30 t, J = 7.1 Hz, 3 H); 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 to 2.18 (m, 3 H); 2.39 (m, 1 H); 2.48 (m, 2 H); 2.56 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.72 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.30 (q, J = 7.1 Hz, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.55 (s, 2 H); 6.68 (d, 521 J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.78 (d, J = 8.6 Hz, 2 H); 7.43 (dd, J = 2.1 and 11.9 Hz, 1 H); 7.65 (d, J = 2.1 Hz, 1 H); 9.44 (s, 1 H) 171 ##STR00197## 6-fluoro-5- [5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]pyridin- 2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 (t, J = 7.0 Hz, 2 H); 2.20 (m, 1 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.67 (m, 3 H); 2.80 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.40 (dd, J = 1.5 and 8.2 Hz, 1 H); 6.56 (m, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 7.72 (d, J = 8.8 493 Hz, 2 H); 7.48 (dd, J = 8.2 and 10.4 Hz, 1 H); 9.42 (s, 1 H); 11.11 (m, 1 H) 172 ##STR00198## 6-(6-tert- butyl-2- fluoro-4- pyridyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.01 (s, 9 H); 1.67 (m, 1 H); 1.77 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.19 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.51 (m, 1 H); 2.60 to 2.70 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.57 (m, 2 H); 6.67 (d, J = 8.8 Hz, 2 H); 6.71 (s, 1 H); 6.73 533 (d, J = 8.8 Hz, 2 H); 6.78 (s, 1 H); 6.81 (s, 1 H); 9.51 (s, 1 H) 173 ##STR00199## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- trimethylsilylphenyl)- 8,9- dihydro-7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 0.20 (s, 9 H); 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.19 (m, 1 H); 2.26 (t, J = 7.0 Hz, 2 H); 2.27 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.62 to 2.70 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.54 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.74 530 (d, J = 8.8 Hz, 2 H); 7.11 (d, J = 8.0 Hz, 2 H); 7.30 (d, J = 8.0 Hz, 2 H); 9.41 (s, 1 H) 174 ##STR00200## 6-(2,2- dimethylindolin- 5-yl)- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid hydrochloride B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.18 (s, 6 H); 1.70 to 3.00 (m, 12 H); 2.22 (t, J = 7.0 Hz, 2 H); 2.58 (s, 2 H); 2.79 (t, J = 7.0 Hz, 2 H); 4.50 (td, J = 6.1 and 47.5 Hz, 2 H); 4.89 (m, 1 H); 5.51 (s, 1 H); 6.20 (d, J = 8.2 Hz, 1 H); 6.65 to 3.73 (m, 3 H); 6.79 (m, 3 H); 6.82 (d, J = 8.2 Hz, 1 H); 7.70 (dd, J = 2.5 and 8.2 Hz, 1 H); 7.85 (d, J = 2.5 Hz, 1 H); 12.79 (s, 1 H) 555 175 ##STR00201## 6-(1,3- benzothiazol- 5-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (500 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.17 (m, 1 H); 2.34 (m, 3 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H); 2.76 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.58 (m, 4 H); 6.71 (s, 1 H); 6.78 (d, J = 8.8 Hz, 2 H); 7.23 (dd, J = 1.8 and 8.4 Hz, 515 1 H); 7.83 (d, J = 1.8 Hz, 1 H); 7.92 (d, J = 8.4 Hz, 1 H); 9.31 (s, 1 H); 9.41 (s, 1 H) 176 ##STR00202## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- methyl-1H- benzimidazol- 5-yl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.17 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.41 (s, 3 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.62 (m, 1 H); 2.69 (t, J = 7.0 Hz, 2 H); 2.78 (dd, J = 6.5 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.54 (d, J = 8.8 Hz, 2 H); 6.56 (s, 2 H); 512 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.89 (dd, J = 1.7 and 8.4 Hz, 1 H); 7.10 to 7.28 (m, 2 H); 9.38 (s, 1 H); 12.00 (m, 1 H) 177 ##STR00203## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (trifluoromethylsulfanyl) phenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.11 (m, 2 H); 2.20 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.65 (m, 1 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 2.83 (t, J = 7.0 Hz, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.83 (d, J = 8.1 Hz, 1 H); 7.31 (d, J = 8.3 Hz, 2 H); 7.53 (d, J = 8.3 Hz, 2 H); 7.72 (dd, J = 1.9 and 8.3 Hz, 1 H); 7.88 (d, J = 1.9 Hz, 1 H); 586 12.90 (m, 1 H) 178 ##STR00204## 6-(1,3- benzothiazol- 6-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.16 (m, 1 H); 2.35 (m, 3 H); 2.43 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.71 (t, J = 7.2 Hz, 2 H); 2.76 (dd, J = 6.3 and 10.5 Hz, 1 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 to 6.62 (m, 4 H); 6.71 (d, J = 2.5 Hz, 1 H); 6.73 (d, J = 8.9 Hz, 2 H); 7.25 515 (dd, J = 1.8 and 8.5 Hz, 1 H); 7.82 (d, J = 8.5 Hz, 1 H); 7.94 (d, J = 1.8 Hz, 1 H); 9.29 (s, 1 H); 9.31 (s, 1 H) 179 ##STR00205## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- methylbenzotriazol- 5- yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.17 (m, 1 H); 2.31 to 2.53 (m, 6 H); 2.62 (m, 1 H); 2.72 (t, J = 7.2 Hz, 2 H); 2.77 (dd, J = 6.1 and 10.4 Hz, 1 H); 4.20 (s, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.57 (d, J = 8.9 Hz, 2 H); 6.59 (s, 2 H); 6.71 (s, 1 H); 6.72 (d, J = 8.9 Hz, 2 H); 7.05 (dd, J = 1.5 513 and 8.7 Hz, 1 H); 7.68 (s, 1 H); 7.70 (d, J = 8.7 Hz, 1 H); 9.42 (s, 1 H) 180 ##STR00206## 6-[2- chloro-4- (trifluoromethoxy) phenyl]-1-
fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.03 to 2.28 (m, 5 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.70 to 2.81 (m, 2 H); 2.99 (m, 1 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.42 (d, J = 8.6 Hz, 1 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.75 (t, J = 8.6 Hz, 1 H); 7.20 (d, J = 8.5 Hz, 1 H); 7.27 (dd, J = 2.1 and 8.5 Hz, 1 H); 7.51 (d, J = 2.1 Hz, 1 H); 9.85 (s, 1 H) 594 181 ##STR00207## 6-(4-tert- butyl-2- methyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.21 (s, 9 H); 1.68 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.00 to 2.24 (m, 5 H); 2.11 (s, 3 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.69 to 2.80 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.53 (d, J = 8.8 Hz, 2 H); 6.56 (s, 2 H); 6.66 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.92 (dd, J = 3.3 and 8.1 Hz, 528 1 H); 7.04 (d, J = 8.1 Hz, 1 H); 7.09 (s, 1 H); 9.33 (s, 1 H) 182 ##STR00208## 6-(2-fluoro- 4- methylsulfonyl- phenyl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.12 to 2.26 (m, 3 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.62 (m, 1 H); 2.71 (t, J = 7.3 Hz, 2 H); 2.79 (dd, J = 6.1 and 10.4 Hz, 1 H); 3.22 (s, 3 H); 4.47 (td, J = 6.2 and 47.6 Hz, 2 H); 4.72 (m, 1 H); 6.59 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 554 H); 6.72 (m, 3 H); 7.47 (t, J = 8.0 Hz, 1 H); 7.59 (dd, J = 1.8 and 8.0 Hz, 1 H); 7.62 (dd, J = 1.8 and 9.1 Hz, 1 H); 9.49 (s, 1 H) 183 ##STR00209## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- methylisoxazol- 4-yl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.64 (s, 3 H); 1.71 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.15 (m, 2 H); 2.21 (m, 1 H); 2.40 (m, 1 H); 2.49 (m, 2 H); 2.53 to 2.71 (m, 4 H); 2.82 (m, 1 H); 4.47 (td, J = 6.0 and 47.5 Hz, 2 H); 4.80 (m, 1 H); 6.58 (m, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.9 Hz, 2 H); 6.81 (d, J = 8.9 Hz, 2 H); 8.72 (s, 1 H); 9.42 (s, 1 H) 463 184 ##STR00210## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (pentafluorosulfanyl) phenyl]-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.29 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.55 (m, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.56 (s, 2 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.73 (d, J = 8.8 Hz, 2 584 H); 7.31 (d, J = 9.0 Hz, 2 H); 7.68 (d, J = 9.0 Hz, 2 H); 9.46 (s, 1 H) 185 ##STR00211## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- morpholinophenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.01 (m, 2 H); 2.15 to 2.26 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.05 (m, 4 H); 3.70 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.54 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.67 (s, 1 H); 6.71 (d, J = 8.8 543 Hz, 2 H); 6.74 (d, J = 9.0 Hz, 2 H); 6.98 (d, J = 9.0 Hz, 2 H); 9.31 (s, 1 H) 186 ##STR00212## 6-[4-(2,2- difluoroethoxy)- 2- fluoro- phenyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.13 to 2.23 (m, 3 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.62 (m, 1 H); 2.69 (t, J = 7.0 Hz, 2 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.29 (dt, J = 3.7 and 14.8 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.35 (tt, J = 3.7 556 and 54.5 Hz, 1 H); 6.55 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (dd, J = 2.7 and 8.7 Hz, 1 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.79 (dd, J = 2.7 and 11.9 Hz, 1 H); 7.07 (t, J = 8.7 Hz, 1 H); 9.39 (s, 1 H) 187 ##STR00213## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1- methylbenzimidazol- 5-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.17 (m, 1 H); 2.32 (t, J = 7.2 Hz, 2 H); 2.48 (m, 1 H); 2.44 (t, J = 7.0 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.70 (t, J = 7.0 Hz, 2 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.78 (s, 3 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.54 (d, J = 8.8 Hz, 2 H); 6.57 (s, 2 H); 6.70 (s, 512 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 7.03 (d, J = 8.4 Hz, 1 H); 7.32 (d, J = 8.4 Hz, 1 H); 7.40 (s, 1 H); 8.07 (s, 1 H); 9.31 (s, 1 H) 188 ##STR00214## 6-(1,2- benzoxazol- 5-yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.21 (m, 3 H); 2.40 (m, 1 H); 2.48 (t, J = 7.3 Hz, 2 H); 2.56 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.80 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.78 (m, 1 H); 6.54 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 499 6.79 (d, J = 9.0 Hz, 1 H); 7.20 (dd, J = 2.0 and 9.0 Hz, 1 H); 7.27 (d, J = 2.0 Hz, 1 H); 9.35 (s, 1 H); 11.00 (m, 1 H) 189 ##STR00215## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1- oxidopyridin- 1-ium-4- yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.07 (m, 2 H); 2.20 (m, 1 H); 2.25 (t, J = 7.0 Hz, 2 H); 2.40 (m, 1 H); 2.48 (m, 2 H); 2.58 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.66 (t, J = 7.0 Hz, 2 H); 2.82 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.79 (m, 1 H); 6.57 (s, 2 H); 6.70 (m, 3 H); 6.80 (t, J = 8.8 Hz, 2 H); 7.08 475 (d, J = 7.3 Hz, 2 H); 7.97 (d, J = 7.3 Hz, 2 H); 9.45 (s, 1 H) 190 ##STR00216## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- pyrrolidin- 1- ylphenyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 1.90 (m, 4 H); 2.00 (m, 2 H); 2.14 to 2.25 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.62 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.14 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.32 (d, J = 8.8 Hz, 2 H); 6.52 (d, J = 1.5 Hz, 2 527 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.66 (t, J = 1.5 Hz, 1 H); 6.74 (d, J = 8.8 Hz, 2 H); 6.92 (d, J = 8.8 Hz, 2 H); 9.39 (s, 1 H) 191 ##STR00217## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- methyl- 1,3- benzoxazol- 5-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (t, J = 7.0 Hz, 2 H); 2.15 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.36 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.53 (s, 3 H); 2.62 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.45 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 (s, 2 H); 6.58 (d, J = 8.8 Hz, 2 513 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.10 (dd, J = 1.8 and 8.5 Hz, 1 H); 7.38 (d, J = 1.8 Hz, 1 H); 7.41 (d J = 8.5 Hz, 1 H); 9.38 (s, 1 H) 192 ##STR00218## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(2- methyl- 1,3- benzoxazol- 6-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.18 (m, 1 H); 2.30 (t, J = 7.0 Hz, 2 H); 2.37 (m, 1 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.54 (s, 3 H); 2.60 to 2.73 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.55 (s, 2 H); 6.58 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.72 513 (d, J = 8.8 Hz, 2 H); 7.08 (dd, J = 1.6 and 8.3 Hz, 1 H); 7.40 (m, 2 H); 9.39 (s, 1 H) 193 ##STR00219## 6-(2,1,3- benzoxadiazol- 5-yl)- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.09 (m, 2 H); 2.19 (m, 1 H); 2.31 to 2.40 (m, 3 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.54 (m, 1 H); 2.60 to 2.72 (m, 3 H); 2.78 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.75 (m, 1 H); 6.60 (s, 2 H); 6.66 (d, J = 8.8 Hz, 2 H); 6.73 (s, 1 H); 6.81 (d, J = 8.8 Hz, 2 H); 7.20 (dd, J = 1.3 500 and 9.4 Hz, 1 H); 7.72 (dd, J = 1.3 and 9.4 Hz, 1 H); 7.83 (t, J = 1.3 Hz, 1 H) ; 9.52 (s, 1 H) 194 ##STR00220## 6-(2,1,3- benzothiadiazol- 5-yl)- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.09 (m, 2 H); 2.18 (m, 1 H); 2.3 to 2.52 (m, 6 H); 2.62 (m, 1 H); 2.70 to 2.80 (m, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.72 (m, 1 H); 6.59 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.72 (s, 1 H); 6.80 (d, J = 8.8 Hz, 2 H); 7.40 (dd, J = 2.0 and 9.2 Hz, 1 H); 7.80 (d, J = 9.2 Hz, 1 H); 7.89 516 (d, J = 2.0 Hz, 1 H); 9.48 (s, 1 H) 195 ##STR00221## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-[4- (oxetan-3- yl)phenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.02 (m, 2 H); 2.19 (m, 1 H); 2.24 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.65 (m, 3 H); 2.79 (dd, J = 6.4 and 10.3 Hz, 1 H); 4.17 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz. 2 H); 4.56 (dd, J = 5.9 and 6.9 Hz, 2 H); 4.72 (m, 1 H); 4.89 (dd, 514 J = 5.9 and 8.5 Hz, 2 H); 6.55 (s, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.12 (d, J = 8.3 Hz, 2 H); 7.19 (d, J = 8.3 Hz, 2 H); 9.38 (s, 1 H) 196 ##STR00222## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6- (2,2,3,3- tetrafluoro- 1,4- benzodioxin- 6-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.27 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.66 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.57 (s, 2 H); 6.66 (d, J = 8.8 Hz, 2 H); 6.70 (s, 1 H); 6.74 588 (d, J = 8.8 Hz, 2 H); 7.07 (dd, J = 2.0 and 8.6 Hz, 1 H); 7.15 (d, J = 2.0 Hz, 1 H); 7.24 (d, J = 8.6 Hz, 1 H); 9.44 (s, 1 H) 197 ##STR00223## 6-(1,2- benzothiazol- 5-yl)-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.65 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.08 (m, 2 H); 2.17 (m, 1 H); 2.34 (m, 3 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.73 (m, 3 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.58 (m, 4 H); 6.71 (s, 1 H); 6.75 (d, J = 8.8 Hz, 2 H); 7.32 (dd, J = 1.8 and 8.6 Hz, 1 H); 7.97 (d, J = 8.6 Hz, 1 H); 7.99 (d, 515 J = 1.8 Hz, 1 H); 8.97 (s, 1 H); 9.40 (s, 1 H) 198 ##STR00224## 6-[2,3- difluoro-4- (1- piperidyl) phenyl]-5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.50 (m, 2 H); 1.60 (m, 4 H); 1.69 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.12 to 2.25 (m, 3 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.70 (m, 3 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 2.94 (m, 4 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.56 (s, 2 H); 6.61 (d, J = 8.8 Hz, 2 H); 6.66 (s, 1
H); 577 6.70 (s, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.83 (dt, J = 2.0 and 8.6 Hz, 1 H); 9.41 (s, 1 H) 199 ##STR00225## 6-(1,3- benzoxazol- 6-yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.67 (m, 1 H); 1.78 (dm, J = 25.4 Hz, 2 H); 2.06 (m, 2 H); 2.18 (m, 1 H); 2.30 to 2.40 (m, 3 H); 2.44 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.70 (t, J = 7.2 Hz, 2 H); 2.78 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.57 (d, J = 1.5 Hz, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.70 (t, J = 1.5 Hz, 1 499 H); 6.72 (d, J = 8.8 Hz, 2 H); 7.14 (dd, J = 1.7 and 8.3 Hz, 1 H); 7.51 (d, J = 1.7 Hz, 1 H); 7.55 (d, J = 8.3 Hz, 1 H); 8.62 (s, 1 H); 9.40 (s, 1 H) 200 ##STR00226## 6-(1,2- benzoxazol- 6-yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.03 (m, 2 H); 2.20 (m, 3 H); 2.39 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.64 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.57 (s, 2 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.67 (s, 1 H); 6.70 to 6.78 (m, 4 H); 7.37 499 (d, J = 8.3 Hz, 1 H); 9.43 (s, 1 H); 10.82 (m, 1 H) 201 ##STR00227## 6-[4-(1,1- difluoroethyl) phenyl]- 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.91 (t, J = 18.8 Hz, 3 H); 2.04 (m, 2 H); 2.20 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.79 (dd, J = 6.3 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.55 (s, 2 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 522 6.72 (d, J = 8.8 Hz, 2 H); 7.21 (d, J = 8.5 Hz, 2 H); 7.35 (d, J = 8.5 Hz, 2 H); 9.40 (s, 1 H) 202 ##STR00228## 6-(3,6- dihydro- 2H-pyran- 4-yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 to 1.89 (m, 3 H); 1.92 (m, 2 H); 1.98 to 2.09 (m, 4 H); 2.25 (m, 1 H); 2.41 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (t, J = 7.0 Hz, 2 H); 2.60 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.68 (m, 1 H); 2.83 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.53 (t, J = 5.4 Hz, 2 H); 3.98 (m, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.81 (m, 1 H); 5.51 (m, 1 H); 6.52 (m, 2 H); 6.64 (s, 1 H); 6.76 464 (d, J = 8.8 Hz, 2 H); 6.92 (d, J = 8.8 Hz, 2 H); 9.31 (s, 1 H) 203 ##STR00229## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6- tetrahydropyran- 4-yl- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.40 (m, 2 H); 1.65 (m, 2 H); 1.73 to 1.89 (m, 5 H); 2.05 (m, 2 H); 2.25 (m, 1 H); 2.40 to 2.72 (m, 8 H); 2.86 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.14 (m, 2 H); 3.84 (m, 2 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.82 (m, 1 H); 6.42 (d, J = 8.5 Hz, 1 H); 6.48 (dd, J = 2.6 and 8.5 Hz, 1 H); 6.62 (d, J = 2.6 Hz, 1 H); 6.81 (d, J = 8.8 Hz, 2 466 H); 6.92 (d, J = 8.8 Hz, 2 H); 9.22 (s, 1 H) 204 ##STR00230## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(4- hydroxycyclohexyl)- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): trans diaxialisomer: 0.97 (m, 2 H); 1.38 to 1.58 (m, 4 H); 1.72 to 1.89 (m, 5 H); 2.02 (m, 2 H); 2.28 (m, 2 H); 2.32 (m, 1 H); 2.48 to 2.55 (m, 6 H); 2.61 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.69 (m, 1 H); 2.87 (dd, J = 6.4 and 10.4 Hz, 1 H); 3.32 (m, 1 H); 4.40 (d, J = 4.9 Hz, 1 H); 4.49 (td, J = 6.1 and 47.5 Hz, 2 H); 4.83 (m, 1 H); 480 6.41 (d, J = 8.6 Hz, 1 H); 6.48 (dd, J = 2.7 and 8.6 Hz, 1 H); 6.61 (d, J = 2.7 Hz, 1 H); 6.81 (d, J = 8.9 Hz, 2 H); 6.89 (d, J = 8.9 Hz, 2 H); 9.20 (s, 1 H) 205 ##STR00231## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(3- methylbenzimidazol- 5-yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.17 (m, 1 H); 2.37 (m, 3 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.61 (m, 1 H); 2.71 (t, J = 7.0 Hz, 2 H); 2.77 (dd, J = 6.4 and 10.3 Hz, 1 H); 3.71 (s, 3 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.67 (m, 4 H); 6.70 (d, J = 1.5 Hz, 1 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.91 (dd, J = 1.6 and 510 8.6 Hz, 1 H); 7.35 (m, 2 H); 8.08 (s, 1 H); 9.36 (s, 1 H) 206 ##STR00232## 4-[4-[5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 2- hydroxy- 8,9- dihydro- 7H- benzo[7] annulen-6- yl]phenyl]- 1H-1,2,4- triazol-5- one A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.05 (m, 2 H); 2.20 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.38 (m, 1 H); 2.47 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.60 to 2.71 (m, 3 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.56 (s, 2 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.77 (d, J = 8.8 Hz, 2 541 H); 7.22 (d, J = 8.8 Hz, 2 H); 7.49 (t, J = 8.8 Hz, 2 H); 8.31 (s, 1 H); 9.40 (s, 1 H); 11.92 (s, 1 H) 207 ##STR00233## 6-(4,4- difluorocyclohexen- 1- yl)-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 to 1.95 (m, 5 H); 1.98 to 2.09 (m, 4 H); 2.15 (m, 2 H); 2.23 (m, 1 H); 2.37 to 2.58 (m, 7 H); 2.60 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.69 (m, 1 H); 2.82 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.81 (m, 1 H); 5.35 (m, 1 H); 6.53 (s, 2 H); 6.63 (s, 1 H); 6.73 (d, J = 8.9 Hz, 2 H); 6.92 (d, J = 8.9 Hz, 2 H); 9.31 (s, 1 H) 498 208 ##STR00234## 6-(4,4- difluorocyclohexyl)- 5- [4-[(3S)-1- (3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.55 to 1.90 (m, 11 H); 1.95 to 2.10 (m, 4 H); 2.25 (m, 1 H); 2.42 (m, 1 H); 2.46 to 2.52 (m, 3 H); 2.56 (t, J = 7.2 Hz, 2 H); 2.62 (dd, J = 3.0 and 10.3 Hz, 1 H); 2.69 (m, 1 H); 2.86 (dd, J = 6.3 and 10.6 Hz, 1 H); 4.49 (td, J = 6.1 and 47.5 Hz, 2 H); 4.83 (m, 1 H); 6.42 (d, J = 8.4 Hz, 1 H); 6.49 (dd, J = 2.6 and 8.4 Hz, 1 H); 6.62 (d, 500 J = 2.6 Hz, 1 H); 6.83 (d, J = 8.8 Hz, 2 H); 6.93 (d, J = 8.8 Hz, 2 H); 9.23 (s, 1 H) 209 ##STR00235## 6-(4- chlorophenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.10 (m, 2 H); 2.15 to 2.28 (m, 3 H); 2.38 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.2 and 10.4 Hz, 1 H); 2.65 (m, 1 H); 2.81 (m, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.77 (m, 1 H); 6.65 (d, J = 8.8 Hz, 2 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.81 (d, J = 8.1 Hz, 1 H); 7.18 (d, J = 8.8 Hz, 2 H); 7.25 (d, J = 8.8 Hz, 2 H); 7.71 (dd, 520 J = 1.9 and 8.1 Hz, 1 H); 7.87 (d, J = 1.9 Hz, 1 H); 12.05 (m, 1 H) 210 ##STR00236## 6-(2- chlorophenyl)- 5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.08 to 2.24 (m, 5 H); 2.38 (m, 1 H); 2.45 (t, J = 7.2 Hz, 2 H); 2.52 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.62 (m, 1 H); 2.79 (m, 1 H); 2.93 (m, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.71 (m, 1 H); 6.60 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.88 (d, J = 8.5 Hz, 1 H); 7.13 to 7.25 (m, 3 H); 7.41 (d, J = 8.3 Hz, 1 H); 7.75 (dd, 520 J = 2.0 and 8.5 Hz, 1 H); 7.90 (d, J = 2.0 Hz, 1 H); 12.67 (m, 1 H) 211 ##STR00237## 6-(2,4- dichlorophenyl)- 1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.09 to 2.27 (m, 5 H); 2.40 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.2 and 10.6 Hz, 1 H); 2.65 (m, 1 H); 2.81 (m, 2 H); 3.04 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.76 (m, 1 H); 6.67 (d, J = 8.8 Hz, 2 H); 6.69 (d, J = 7.8 Hz, 1 H); 6.76 (d, J = 8.8 Hz, 2 H); 7.21 (d, J = 8.3 Hz, 1 H); 7.30 (dd, J = 2.2 and 8.3 Hz, 1 572 H); 7.60 (d, J = 2.2 Hz, 1 H); 7.65 (t, J = 7.8 Hz, 1 H) 212 ##STR00238## 6-(4- chloro-2- fluoro- phenyl)-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.69 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.08 to 2.26 (m, 5 H); 2.39 (m, 1 H); 2.48 (t, J = 7.2 Hz, 2 H); 2.54 (dd, J = 2.8 and 10.4 Hz, 1 H); 2.64 (m, 1 H); 2.80 (dd, J = 6.4 and 10.4 Hz, 1 H); 2.87 (t, J = 7.2 Hz, 2 H); 4.48 (td, J = 6.2 and 47.6 Hz, 2 H); 4.76 (m, 1 H); 6.67 (d, J = 8.8 Hz, 3 H); 6.77 (d, J = 8.8 Hz, 2 H); 7.18 (dd, J = 2.1 and 8.3 Hz, 1 H); 7.24 (t, J = 8.3 Hz, 556 1 H); 7.32 (dd, J = 2.1 and 9.8 Hz, 1 H); 7.60 (t, J = 8.0 Hz, 1 H); 12.40 (m, 1 H) 213 ##STR00239## 6-(2- chloro-4- fluoro- phenyl)-1- fluoro-5-[4- [(3S)-1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 8,9- dihydro- 7H- benzo[7] annulene-2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.4 Hz, 2 H); 2.04 to 2.25 (m, 5 H); 2.38 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.52 (m, 1 H); 2.63 (m, 1 H); 2.79 (m, 2 H); 3.00 (m, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.51 (d, J = 8.2 Hz, 1 H); 6.62 (d, J = 8.9 Hz, 2 H); 6.73 (d, J = 8.9 Hz, 2 H); 7.08 (dt, J = 2.7 and 8.7 Hz, 1 H); 7.21 (dt, J = 6.4 and 8.7 Hz, 1 H); 7.32 (t, J = 8.2 Hz, 556 1 H); 7.40 (dd, J = 2.7 and 8.7 Hz, 1 H); 12.00 (m, 1 H) 214 ##STR00240## 9-(4-{[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxy} phenyl)-8- phenyl- 6,7- dihydro- 5H- benzo[7] annulen-3-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.04 (m, 2 H); 2.19 (m, 1 H); 2.27 (t, J = 7.0 Hz, 2 H); 2.40 (m, 1 H); 2.48 (m, 2 H); 2.55 (m, 1 H); 2.60 to 2.71 (m, 3 H); 2.80 (m, 1 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.56 (m, 2 H); 6.59 (d, J = 8.8 Hz, 2 H); 6.69 (s, 1 H); 6.71 (d, J = 8.8 Hz, 2 H); 7.05 to 7.19 (m, 5 H); 457 9.37 (s, 1 H) 215 ##STR00241## 5-[4-[(3S)- 1-(3- fluoropropyl) pyrrolidin-3- yl]oxyphenyl]- 6-(1H- indazol-5- yl)-8,9- dihydro- 7H- benzo[7] annulen-2-ol A 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.66 (m, 1 H); 1.78 (dm, J = 25.3 Hz, 2 H); 2.06 (m, 2 H); 2.17 (m, 1 H); 2.29 to 2.40 (m, 3 H); 2.43 (t, J = 7.2 Hz, 2 H); 2.53 (m, 1 H); 2.61 (m, 1 H); 2.70 (t, J = 7.0 Hz, 2 H); 2.77 (dd, J = 6.4 and 10.4 Hz, 1 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.70 (m, 1 H); 6.54 (d, J = 8.8 Hz, 2 H); 6.56 (s, 2 H); 6.70 (s, 1 H); 6.72 (d, J = 8.8 498 Hz, 2 H); 7.09 (d, J = 8.7 Hz, 1 H); 7.30 (d, J = 8.7 Hz, 1 H); 7.52 (s, 1 H); 7.91 (s, 1 H); 9.35 (s, 1 H); 12.90 (s, 1 H) 216 ##STR00242## 6-(2- Chloro-3- fluoro- phenyl)-5- {(4-[(S)-1- (3-fluoro- propyl)- pyrrolidin- 3-yloxy]- phenyl}- 8,9- dihydro- 7H- benzocycloheptene- 2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.68 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 2.10 to 2.25 (m, 5 H); 2.18 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.53 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.63 (m, 1 H); 2.79 (m, 1 H); 2.93 (t, J = 7.0 Hz, 2 H); 4.46 (td, J = 6.1 and 47.5 Hz, 2 H); 4.73 (m, 1 H); 6.63 (d, J = 8.8 Hz, 2 H); 6.73 (d, J = 8.8 Hz, 2 H); 6.89 (d, J = 8.1 Hz, 1 H); 7.05 (m, 1 H); 7.24 (m, 2 H); 7.77 (dd, J = 2.0 and 8.1 Hz, 538 1 H); 7.91 (d, J = 2.0 Hz, 1 H); 12.60 (m, 1 H) 217 ##STR00243## 5-{4-[(S)-1- (3-Fluoro- propyl)- pyrrolidin- 3-yloxy]- phenyl}-6- phenyl- 8,9- dihydro- 7H- benzocycloheptene- 2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.80 (dm, J = 25.3 Hz, 2 H); 2.11 (m, 2 H); 2.20 (m, 1 H); 2.27 (t, J = 7.0 Hz, 2 H); 2.41 (m, 1 H); 2.48 (m, 2 H); 2.58 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.67 (m, 1 H); 2.83 (m, 3 H); 4.48 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.62 (d, J = 8.8 Hz, 2 H); 6.72 (d, J = 8.8 Hz, 2 H); 6.89 (d, J = 8.1 Hz, 1 H); 7.10 to 7.25 (m, 5 H); 7.73 (dd, J = 2.0 and 8.1 Hz, 1 486 H); 7.89 (d, J = 2.0 Hz, 1 H); 12.84 (m, 1 H)
218 ##STR00244## 6- Benzooxazol- 5-yl-5- {4-[(S)-1- (3-fluoro- propyl)- pyrrolidin- 3-yloxy]- phenyl}- 8,9- dihydro- 7H- benzocycloheptene- 2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.90 to 2.08 (m, 4 H); 2.14 (m, 2 H); 2.32 (t, J = 7.0 Hz, 2 H); 2.90 (t, J = 7.0 Hz, 2 H); 3.00 to 3.40 (m, 6 H); 4.51 (td, J = 6.1 and 47.5 Hz, 2 H); 4.98 (m, 1 H); 6.70 (d, J = 8.8 Hz, 2 H); 6.81 (d, J = 8.8 Hz, 2 H); 6.88 (d, J = 8.1 Hz, 1 H); 7.25 (dd, J = 2.0 and 8.6 Hz, 1 H); 7.60 (m, 2 H); 7.77 (dd, J = 2.0 and 8.1 Hz, 1 H); 7.92 (d, J = 2.0 Hz, 1 H); 8.69 (s, 1 H); 9.92 (m, 1 H); 12.85 527 (m, 1 H) 219 ##STR00245## 6-[4-(1,1- Difluoro- ethyl)- phenyl]-5- {4-[(S)-1- (3-fluoro- propyl)- pyrrolidin- 3-yloxy]- phenyl}- 8,9- dihydro- 7H- benzocycloheptene- 2- carboxylic acid B 1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.70 (m, 1 H); 1.79 (dm, J = 25.3 Hz, 2 H); 1.92 (t, J = 19.0 Hz, 3 H); 2.11 (m, 2 H); 2.20 (m, 1 H); 2.28 (t, J = 7.0 Hz, 2 H); 2.39 (m, 1 H); 2.46 (t, J = 7.2 Hz, 2 H); 2.55 (dd, J = 3.0 and 10.4 Hz, 1 H); 2.65 (m, 1 H); 2.79 (dd, J = 6.4 and 10.4 Hz, 1 H); 2.84 (t, J = 7.0 Hz, 2 H); 4.47 (td, J = 6.1 and 47.5 Hz, 2 H); 4.74 (m, 1 H); 6.64 (d, J = 8.8 Hz, 2 H); 6.76 (d, J = 8.8 Hz, 2 550 H); 6.86 (d, J = 8.1 Hz, 1 H); 7.27 (d, J = 8.8 Hz, 2 H); 7.39 (d, J = 8.8 Hz, 2 H); 7.73 (dd, J = 2.0 and 8.1 Hz, 1 H); 7.90 (d, J = 2.0 Hz, 1 H); 12.48 (m, 1 H)
[0392] The examples which follow describe the preparation of some compounds of formula (I). The numbers of the compounds exemplified below match those given in the Table 1 above. All reactions are performed under inert atmosphere, unless otherwise stated.
Intermediates
Compound (c). Tert-butyl (3S)-3-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2yl)phenoxy]pyrrolidine- -1-carboxylate
##STR00246##
[0394] To a solution of 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenol (a) (82.7 g, 364.51 mmol) in THF (2 L) was added under argon (R)-1-N-Boc-3-hydroxypyrrolidine (b) (84.43 g, 437.41 mmol) followed by N,N,N',N'-tetramethylazodicarboxamide (99.1 g, 546.77 mmol). The clear reaction mixture turned orange and triphenylphosphine (143.41 g, 546.77 mmol) was added. The reaction mixture was stirred at room temperature for 24 hours, meanwhile a precipitate of triphenylphosphine oxide formed (Ph.sub.3P.dbd.O). The reaction mixture was poured in water (1.5 L) and extracted with ethyl acetate (AcOEt) (3.times.1.5 L). Gathered organic phases were dried over magnesium sulfate (MgSO.sub.4), filtered and concentrated under reduced pressure. The residue was taken up into diisopropylether (1.5 L) and the solid formed (Ph.sub.3P.dbd.O) was filtered. The solvent was concentrated under reduced pressure and the residue purified by column chromatography eluting with a mixture of heptane with AcOEt (90/10; v/v) to give 145 g (100%) of tert-butyl (3S)-3-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidin- e-1-carboxylate (c) as a colorless oil.
[0395] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.27 (s, 12H); 1.39 (s, 9H); 2.05 (m, 1H); 2.14 (m, 1H); 3.37 (3H); 3.55 (m, 1H); 5.05 (s, 1H); 6.94 (d, J=8.4 Hz, 2H); 7.61 (d, J=8.4 Hz, 2H)
Compound (d). (3S)-3-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-41)phenoxy]pyrrolidine, hydrochloride
##STR00247##
[0397] To a solution of (S)-tert-butyl 3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy)pyrrolidine-1-c- arboxylate (c) (80 g, 195.23 mmol) in MeOH (450 ml) was added slowly HCl 4N in dioxane (250 ml).
[0398] After 1.5 hours, the reaction mixture was concentrated under reduced pressure and the residue was taken up into Et.sub.2O with stirring to give a solid which then was filtered and dried under vacuum to give compound (d) 61.8 g (95%) as a white powder.
[0399] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.28 (s, 12H); 2.10 (m, 1H); 2.21 (m, 1H); 3.31 (3H); 3.48 (m, 1H); 5.19 (m, 1H); 6.97 (d, J=8.4 Hz, 2H); 7.63 (d, J=8.4 Hz, 2H); 9.48 (s, 1H); 9.71 (s, 1H).
[0400] LC/MS (m/z, MH.sup.+): 290
Reagent (1). (3S)-1-(3-fluoropropyl)-3-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl- )phenoxy]pyrrolidine
##STR00248##
[0402] To a suspension of (S)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy)pyrrolidine hydrochloride (d) (20 g, 61.42 mmol) in acetonitrile (100 ml), was added K.sub.2CO.sub.3 (21.22 g, 153.54 mmol) and 1-iodo-3-fluoropropane (12.15 g, 61.42 mmol), under argon. The reaction mixture was stirred at 40.degree. C. for 24 hours. After cooling to room temperature, the reaction mixture was filtered and washed with acetonitrile. The filtrate was concentrated under reduced pressure and the residue was taken up in DCM and the solid formed was filtered and washed with DCM. The filtrate was concentrated to give reagent (1) 21.5 g (100%) as a yellow foam.
[0403] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.27 (s, 12H); 1.77 (m, 2H); 1.84 (m, 1H); 2.27 (m, 1H); 2.41 (m, 1H); 2.49 (2H); 2.62 (dd, J=2.6 and 10.4 Hz, 1H); 2.69 (m, 1H); 2.83 (dd, J=6.2 and 10.4 Hz, 1H); 4.47 (td, J=6.2 and 47 Hz, 2H); 4.99 (m, 1H); 6.77 (d, J=8.4 Hz, 2H); 7.58 (d, J=8.4 Hz, 2H).
[0404] LC/MS (m/z, MH.sup.+): 350
Intermediate (A1). 9-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate
##STR00249##
[0406] To a solution of 3-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (2.42 g, 13.73 mmol), in acetone (100 ml), was added K.sub.2CO.sub.3 (1.90 g, 13.73 mmol) and pivaloyl chloride (1.69 ml, 13.73 mmol). The reaction mixture was stirred at room temperature for 18 hours, then was filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane in AcOEt (100/0 to 85/15, v/v) to give 2.62 g (73%) of 9-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (A1) which was used as such in the next step.
Intermediate (B1). 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate
##STR00250##
[0408] To a solution of 9-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (A1) (2.6 g, 10 mmol) in DCM (100 ml) was added under argon pyridine (1.26 ml, 14.98 mmol) and trifluoromethanesulfonic anhydride (3.39 ml, 19.97 mmol) dropwise. The reaction mixture was stirred at room temperature for 16 hours and ice (200 g) and DCM (200 ml) were added. The phases were separated, the aqueous phase was washed with DCM and the gathered organic phases were dried over MgSO.sub.4, filtered and evaporated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane in AcOEt (100/0 to 90/10, v/v) to give 3.65 g (93%) of 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (B1) as an orange oil.
[0409] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.30 (s, 9H); 1.98 (m, 2H); 2.26 (m, 2H); 2.72 (m, 2H); 6.46 (t, J=6.2 Hz, 1H); 7.10 to 7.14 (m, 2H); 7.38 (m, 1H)
Intermediate (C1). 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-2-yl 2,2-dimethylpropanoate
##STR00251##
[0411] To a solution of 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (B1) (600 mg, 1.53 mmol) and (S)-1-(3-fluoropropyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- phenoxy)pyrrolidine (1) (534 mg, 1.53 mmol) in dioxane (24 ml) and water (6 ml), Cs.sub.2CO.sub.3 (1.05 g, 3.21 mmol) was added under argon, followed by Pd(dppf)Cl.sub.2 (124.87 mg, 0.15 mmol). The reaction mixture was stirred for 20 minutes at 60.degree. C. After cooling to room temperature, water (40 ml) and DCM (200 ml) were added. After decantation, the organic phase was dried over MgSO.sub.4, then was filtered and concentrated under reduced pressure. The obtained residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0 to 4%; V/V) to give 0.7 g (98%) of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-2-yl 2,2-dimethylpropanoate (C1).
[0412] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.24 (s, 9H); 1.70 to 1.92 (m, 5H); 2.11 (m, 2H); 2.26 (m, 1H); 2.42 (m, 1H); 2.48 (t, J=7.2 Hz, 2H); 2.52 to 2.74 (m, 4H); 2.85 (dd, J=6.2 and 10.4 Hz, 1H); 4.49 (td, J=6.1 and 47.5 Hz, 2H); 4.85 (m, 1H); 6.39 (t, J=7.4 Hz, 1H); 6.59 (d, J=2.6 Hz, 1H); 6.84 (d, J=8.8 Hz, 2H); 6.97 (dd, J=2.6 and 8.2 Hz, 1H); 7.11 (d, J=8.8 Hz, 2H); 7.35 (d, J=8.2 Hz, 1H)
Intermediate (D1). 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-yl-2,2-dimethylpropanoate
##STR00252##
[0414] To a solution of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-2-yl 2,2-dimethylpropanoate (C1) (700 mg, 1.50 mmol) in THF (30 ml), was added pyridinium tribromide (481 mg, 1.50 mmol). The reaction mixture was stirred for 2.5 hours at room temperature. Water (20 ml) was added and pH was adjusted to 7 with concentrated solution of NaHCO.sub.3. DCM (60 ml) was added. The aqueous phase was washed with DCM, 3 times and the gathered organic phases were dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0 to 5%; V/V) to give 0.667 g (82%) of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (D1).
[0415] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.21 (s, 9H); 1.71 to 1.91 (m, 3H); 2.18 to 2.33 (m, 3H); 2.42 (m, 1H); 2.48 (t, J=7.2 Hz, 2H); 2.50 (m, 2H); 2.62 (dd, J=3.0 and 10.4 Hz, 1H); 2.65 to 2.77 (m, 3H); 2.86 (dd, J=6.2 and 10.4 Hz, 1H); 4.49 (td, J=6.1 and 47.5 Hz, 2H); 4.87 (m, 1H); 6.44 (d, J=2.6 Hz, 1H); 6.88 (d, J=8.8 Hz, 2H); 6.97 (dd, J=2.6 and 8.2 Hz, 1H); 7.10 (d, J=8.8 Hz, 2H); 7.34 (d, J=8.2 Hz, 1H)
Intermediate (D2). 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-ol
##STR00253##
[0417] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-yl-2,2-dimethylpropanoate (D1) (665 mg, 1.22 mmol) in methanol (30 ml), was added NaOH (5N, 2 ml, 10.00 mmol). The reaction mixture was stirred 15 minutes at room temperature and 2 ml of HCl 5N was added. The solvent was removed under reduced pressure. The residue was taken up into AcOEt. The phases were separated and the aqueous phase was washed with AcOEt. The organic phases were combined and dried over MgSO.sub.4, then were filtered and concentrated under reduced pressure and the residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0 to 3%; V/V) to give 0.4 g (72%) of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-ol (D2).
[0418] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 to 1.89 (m, 3H); 2.14 (m, 2H); 2.28 (m, 1H); 2.38 to 2.55 (m, 5H); 2.58 to 2.72 (m, 4H); 2.87 (dd, J=6.4 and 10.4 Hz, 1H); 4.49 (td, J=6.1 and 47.5 Hz, 2H); 4.85 (m, 1H); 6.20 (d, J=2.7 Hz, 1H); 6.60 (dd, J=2.7 and 8.2 Hz, 1H); 6.87 (d, J=8.8 Hz, 2H); 7.18 (d, J=8.8 Hz, 3H); 9.11 (s, 1H)
[0419] LC/MS (m/z, MH.sup.+): 460
Intermediate (A2). 2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one
##STR00254##
[0421] To a solution of 2-methoxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (15 g, 78.85 mmol) in toluene (400 ml) was added AlCl.sub.3 (25 g, 187.49 mmol). The reaction mixture was stirred at 91.degree. C. (bath temperature) for 45 minutes, cooled to room temperature and poured onto ice (900 g). The slurry was stirred for 20 minutes and the solid formed was filtered, washed with water (200 ml), and diisopropyl ether (200 ml), and then was dried to give 14.1 g (100%) of 2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A2) as a beige powder.
[0422] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 10.1 (s, 1H); 7.53 (d, 1H); 6.68 (dd, 1H); 6.62 (d, 1H); 2.84 (t, 2H); 2.52 (t, 2H); 1.65 (q, 2H); 1.55 (q, 2H).
[0423] LC/MS (m/z, MH.sup.+): 177
Intermediate (A3). 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate
##STR00255##
[0425] To a solution of 2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A2) (1.52 g, 8.63 mmol), in acetone (60 ml), was added K.sub.2CO.sub.3 (1.19 g, 8.63 mmol) and pivaloyl chloride (1.06 ml, 8.63 mmol). The reaction mixture was stirred at room temperature for 16 hours, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane in AcOEt (100/0 to 85/15, v/v) to give 1.55 g (69%) of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (A3) as a colorless oil.
[0426] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 7.65 (d, 1H); 7.10-7.04 (m, 2H); 2.95 (t, 2H); 2.68 (t, 2H); 1.85-1.65 (m, 4H).
[0427] LC/MS (m/z, MH.sup.+): 261
Intermediate (B2). 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate
##STR00256##
[0429] To a solution of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (A3) (15 g, 57.62 mmol) in DCM (500 ml) was added dropwise under argon pyridine (7.28 ml, 86.43 mmol) and trifluoromethanesulfonic anhydride (19.58 ml, 115.24 mmol). The reaction mixture was stirred at room temperature for 2 hours and ice (200 g) was added. The phases were separated, the aqueous phase was washed with DCM and the gathered organic phases were dried over MgSO.sub.4, filtered and evaporated under reduced pressure to give 22 g (97%) of 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (B2) as a white solid.
[0430] LC/MS (m/z, MH.sup.-): 391
Intermediate (C2). 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-yl-2,2-dimethylpropanoate
##STR00257##
[0432] To a solution of 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-d- imethylpropanoate (B2) (22 g, 56.07 mmol) and (3S)-1-(3-fluoropropyl)-3-[4-(tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy- ]pyrrolidine (1) (20.56 g, 58.87 mmol) in dioxane (420 ml) and water (120 ml) were added under argon Pd(dppf)Cl.sub.2 (2.75 g, 3.36 mmol) and Cs.sub.2CO.sub.3 (36.57 g, 112.13 mmol). The reaction mixture was stirred for 1 hour at room temperature and was partitioned between water and DCM. The aqueous phase was washed with DCM and the gathered organic phases dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0 to 5%; V/V) to give 31 g (100%) of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-yl-2,2-dimethylpropanoate (C2).
[0433] LC/MS (m/z, MH.sup.+): 466
Intermediate (D3). 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate
##STR00258##
[0435] To a solution of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-yl-2,2-dimethylpropanoate (C2) (11 g, 22.44 mmol) in THF (250 ml), was added pyridinium tribromide (7.98 g, 22.44 mmol). The reaction mixture was stirred for 1 hour at room temperature and 100 ml of water was added followed by a saturated solution of sodium bicarbonate (NaHCO.sub.3) until pH 7. The aqueous phase was washed with DCM, 3 times and the gathered organic phases were dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0 to 4%; V/V) to give 9.2 g (75%) of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (D3).
[0436] LC/MS (m/z, MH.sup.+): 545
Intermediate (D4). 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol
##STR00259##
[0438] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (D3) (9.2 g, 16.90 mmol) in MeOH (250 ml), was added NaOH (2N, 50 ml, 100 mmol). The reaction mixture was stirred 15 minutes at room temperature and 22 ml of aqueous HCl 5N was added. The solvent was removed under reduced pressure and the residue was taken up into DCM. The phases were separated and the aqueous phase was washed with DCM and AcOEt. The organic phase was dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0 to 05%; V/V) to give 6.03 g (78%) of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4).
[0439] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 to 1.89 (m, 3H); 2.19 (m, 2H); 2.28 (m, 1H); 2.39 to 2.52 (m, 5H); 2.59 to 2.72 (m, 4H); 2.87 (dd, J=6.4 and 10.4 Hz, 1H); 4.49 (td, J=6.1 and 47.5 Hz, 2H); 4.83 (m, 1H); 6.52 (s, 2H); 6.68 (s, 1H); 6.83 (d, J=8.8 Hz, 2H); 7.07 (d, J=8.8 Hz, 2H); 9.50 (s, 1H)
[0440] LC/MS (m/z, MH.sup.+): 461
Intermediate (A4). 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yltrifluoromethanesulfonate
##STR00260##
[0442] To a solution of 2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A2) (18.5 g, 105 mmol) in DCM (185 ml) and lutidine (13.35 ml, 113.505 mmol), cooled at 5.degree. C. under argon, was added dropwise trifluoromethanesulfonic anhydride (20.22 ml, 123.29 mmol) while keeping temperature between 10 and 20.degree. C. The reaction mixture was stirred for 1 hour at 5.degree. C. then at room temperature for 1 hour.
[0443] Then, ice (200 g) was added and the slurry partitioned between water and DCM. The organic phase was washed with aqueous NaHCO.sub.3 solution, dried over MgSO.sub.4, filtered off and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane/AcOEt from 100 to 90/10 to give 28.2 g (87%) of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl trifluoromethanesulfonate (A4) as an orange oil.
[0444] LC/MS (m/z, MH.sup.+): 309
Intermediate (A5). Methyl 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate
##STR00261##
[0446] To a solution of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl trifluoromethanesulfonate (A4) (5.03 g, 16.32 mmol) in DMF (24 ml) and MeOH (12 ml), were added Pd(dppf)Cl.sub.2 (754 mg, 0.98 mmol) and diisopropylethylamine (6 ml). The black suspension was carbonylated in an autoclave at 70.degree. C. under 5 bars of CO for 2.5 hours. The reaction mixture was filtered, then the filtrate was partially concentrated under reduced pressure, and the residue, was partitioned between AcOEt and water. The organic phase was washed with water (2.times.75 ml) and aqueous HCl 0.5 N, dried over MgSO.sub.4 and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane/AcOEt from 100/0 to 90/10 to give 3.4 g (95%) of methyl 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate (A5) as a colorless oil.
[0447] LC/MS (m/z, MH.sup.+): 219
Intermediate (B3). methyl 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-3-carboxy- late
##STR00262##
[0449] To a solution of methyl 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate (A5) (18.19 g, 83.34 mmol) in DCM (500 ml) and anhydrous pyridine (11 ml, 130.56 mmol), cooled at 5.degree. C. under argon, was added dropwise trifluoromethanesulfonic anhydride (30 ml, 176.54 mmol). The reaction mixture, a thick suspension, was stirred at room temperature for 24 hours, then ice was added and partitioned between water and DCM. The organic phase was dried over MgSO.sub.4, filtered off and concentrated under reduced pressure to give 29 g (100%) of methyl 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-3-carboxy- late (B3) as a yellow gum.
[0450] LC/MS (m/z, MH.sup.+): 351
Intermediate (C3). methyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulene-3-carboxylate
##STR00263##
[0452] To a solution of methyl 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-3-carboxy- late (B3) (29 g, 82.9 mmol), (3S)-1-(3-fluoropropyl)-3-[4-(tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy- ]pyrrolidine (1) (28.9 g, 82.9 mmol), in dioxane (225 ml) were added Pd(dppf)Cl.sub.2 under argon, complex with DCM (3.73 g, 4.57 mmol) and Cs.sub.2CO.sub.3 1.5 M aqueous solution (111.12 ml, 166.68 mmol). The reaction mixture was stirred at 60.degree. C. for 1 hour.
[0453] After cooling to room temperature, the reaction mixture was poured into a mixture of water (500 ml) and AcOEt (400 ml). The organic phase was washed with brine, dried over MgSO.sub.4, filtered on celite and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 23 g (65%) of methyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulene-3-carboxylate (C3) as a brown gum.
[0454] LC/MS (m/z, MH.sup.+): 424
Intermediate (D5). Methyl 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulene-3-carboxylate hydrobromide
##STR00264##
[0456] To a solution of methyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulene-3-carboxylate (C3) (13.93 g, 32.89 mmol), in DCM (150 ml) was added under argon pyridinium tribromide (15.78 g, 44.41 mmol). The reaction mixture was stirred for 1 hour at room temperature. Water (200 ml) was added, organic phase was then dried over MgSO.sub.4, and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 16.4 g (85%) of methyl 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulene-3-carboxylate hydrobromide (D5) as a yellow meringue.
[0457] LC/MS (m/z, MH.sup.+): 502
Intermediate (C4). 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-ol
##STR00265##
[0459] To a solution under argon of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-yl-2,2-dimethylpropanoate (C2) (24.8 g, 53.26 mmol) in MeOH (300 ml), was added NaOH 5M (23 ml, 115.00 mmol). The reaction mixture was stirred for 2 hours at room temperature. pH was then adjusted to 7 by addition of 6N aqueous HCl solution. The MeOH was concentrated under reduced pressure, then DCM was added. The organic phase was dried over MgSO.sub.4, and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 18.8 g (93%) of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-ol (C4) as a beige solid.
[0460] LC/MS (m/z, MH.sup.+): 382
Intermediate (C5). 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-yl trifluoromethanesulfonate
##STR00266##
[0462] To a solution of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-ol (C4) (20.6 g, 54.00 mmol) in DCM (200 ml) and pyridine (6.55 ml, 81.00 mmol), cooled to 5.degree. C. (ice bath), was added dropwise trifluoromethanesulfonic anhydride (18.93 ml, 108.00 mmol) under argon, and the reaction temperature was maintained <15.degree. C. The ice bath was removed, and the brown suspension was stirred at room temperature for 2 hours. Ice (200 g) and DCM (200 ml) were added and the phases separated. The organic phase was dried over MgSO.sub.4, and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 24.7 g (89.1%) of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-yl trifluoromethanesulfonate (C5) as a brown oil.
[0463] LC/MS (m/z, MH.sup.+): 514
Intermediate (C3). Methyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulene-3-carboxylate
##STR00267##
[0465] To a solution of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulen-3-yl trifluoromethanesulfonate (C5) (10.1 g, 19.67 mmol) in DMF (66 ml) and MeOH (33 ml), were added Pd(dppf)Cl.sub.2 (909 mg, 1.18 mmol) and diisopropylethylamine (7.21 ml). The black suspension was carbonylated in an autoclave at 70.degree. C. under 5 bars of CO for 5 hours. The reaction mixture was filtered, then the filtrate was partially concentrated under reduced pressure. The residue was partitioned between AcOEt and water. The organic phase was washed with water (2.times.100 ml), dried over MgSO.sub.4, and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 7.13 g (86%) of methyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-- benzo[7]annulene-3-carboxylate (C3) as a brown gum.
[0466] LC/MS (m/z, MH.sup.+): 424
Intermediate (A6). 1-fluoro-2-methoxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one
##STR00268##
[0467] Step 1. Ethyl 5-(2-fluoro-3-methoxyphenyl)pent-4-enoate
##STR00269##
[0469] To a solution of [3-(ethoxycarbonyl)propyl]triphenylphosphonium bromide (30 g, 65.5 mmol) in THF (300 ml) cooled at -78.degree. C., was added potassium bis(trimethylsilyl)amide (16 g, 80.45 mmol) in 5 minutes. The orange suspension was stirred 1 hour at -78.degree. C., and 2-fluoro-3-methoxybenzaldehyde (10 g, 65 mmol) was added. The reaction mixture was allowed to reach room temperature overnight under stirring. The solvent was concentrated under reduced pressure, the residue taken up in AcOEt (300 ml), washed twice with sodium bisulfite, 10% (w/v) aqueous solution (50 ml).
[0470] The organic phase was dried over anhydrous MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a mixture of AcOEt/cyclohexane 10/90 to give 9 g (55%) of (E)-ethyl 5-(2-fluoro-3-methoxyphenyl)pent-4-enoate as a yellow oil.
[0471] LC/MS (m/z, MH.sup.+): 253 mixture of E/Z isomers 69/31%
Step 2. Ethyl 5-(2-fluoro-3-methoxyphenyl)pentanoate
##STR00270##
[0473] To a solution of (E)-ethyl 5-(2-fluoro-3-methoxyphenyl)pent-4-enoate (9 g, 35.67 mmol) in ethanol (100 ml), was added Pd/C 10% (100 mg). The black suspension was hydrogenated in an autoclave, at room temperature under 10 bars of hydrogen during 24 hours. The slurry was filtered then the filtrate was concentrated under reduced pressure to give 8.9 g (98%) of ethyl 5-(2-fluoro-3-ethoxyphenyl)pentanoate as a colorless oil.
[0474] LC/MS (m/z, MH.sup.-): 255
Step 3. 5-(2-fluoro-3-methoxyphenyl)pentanoic acid
##STR00271##
[0476] To a solution of ethyl 5-(2-fluoro-3-methoxyphenyl)pentanoate (8.9 g, 35.00 mmol) in ethanol (60 ml), were added water (12 ml) and NaOH 32% (6 ml, 72 mmol). The white suspension was then stirred for 2 hours at 50.degree. C. After cooling to room temperature, 100 g of ice were added and the reaction mixture was acidified with aqueous HCl to pH 3. The solid obtained was filtered off and dried to give 7.9 g (100%) of 5-(2-fluoro-3-methoxyphenyl)pentanoic acid as a white solid.
[0477] LC/MS (m/z, MH.sup.+): 225
Step 4. 1-fluoro-2-methoxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A6)
##STR00272##
[0479] 5-(2-fluoro-3-methoxyphenyl)pentanoic acid (4.8 g, 21.22 mmol) was added to trifluoromethane sulfonic acid (19 ml, 212 mmol) cooled at 5.degree. C. The brown solution was stirred at 5.degree. C. during 1 hour. Ice (100 g) and AcOEt (100 ml) were added, followed by an aqueous solution of NaHCO.sub.3 until pH was 7. The organic phase was dried over MgSO.sub.4, filtered off and concentrated under reduced pressure to give 4.4 g (99%) of 1-fluoro-2-methoxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A6) as a brown oil.
[0480] LC/MS (m/z, MH.sup.-): 209
Intermediate (A7). 1-fluoro-2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one
##STR00273##
[0482] To a solution of 1-fluoro-2-methoxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A6) (6.2 g, 29.8 mmol) in toluene (100 ml) was added AlCl.sub.3 (4.76 g, 35.7 mmol). The brown suspension was stirred for 1 hour at 90.degree. C. After cooling to room temperature, the hot mixture was poured into 900 g of iced water. The solid obtained was filtered off, washed with water, aqueous HCl 0.1 N and dried to give 5.3 g (92%) of 1-fluoro-2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A7) as a beige solid.
[0483] LC/MS (m/z, MH.sup.+): 195
Intermediate (A8). 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate
##STR00274##
[0485] To a solution of 1-fluoro-2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A7) (5.3 g, 27.3 mmol) in acetone (150 ml) were added K.sub.2CO.sub.3 (3.77 g, 27.29 mmol) and pivaloyl chloride (2.29 g/3.36 ml, 27.3 mmol). The orange suspension was stirred for 2 hours at room temperature. The solids were filtered off and then washed with acetone (10 ml). The filtrate was concentrated under reduced pressure. AcOEt (100 ml) and water were added to the residue obtained. The organic phase was dried over MgSO.sub.4, filtered off and concentrated under reduced pressure to give 7.2 g (95%) of 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl-2,2-dimethyl- propanoate (A8) as a beige solid.
[0486] LC/MS (m/z, MH.sup.+): 279
Intermediate (B4). 4-fluoro-9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3- -yl 2,2-dimethylpropanoate
##STR00275##
[0488] To a solution of 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl-2,2-dimethylpro- panoate (A8) (2.05 g, 7.37 mmol) in DCM (50 ml) was added under argon pyridine (0.93 ml, 11.05 mmol) and trifluoromethanesulfonic anhydride (2.5 ml, 14.73 mmol) dropwise. The reaction mixture was stirred at room temperature for 2 hours and ice (100 g) was added. The phases were separated, the aqueous phase was washed with DCM and the gathered organic phases were dried over MgSO.sub.4, filtered and evaporated under pressure. The residue was purified by flash chromatography eluting with DCM to give 2.5 g (83%) of 4-fluoro-9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3- -yl-2,2-dimethylpropanoate (B4) as a yellow oil.
[0489] LC/MS (m/z, MH.sup.+): 411
Intermediate (C6). 4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-di- hydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate
##STR00276##
[0491] To a solution of 4-fluoro-9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3- -yl-2,2-dimethylpropanoate (B4) (700 mg, 1.71 mmol) and (S)-1-(3-fluoropropyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- phenoxy)pyrrolidine (1) (595.72 mg, 1.71 mmol) in dioxane (10 ml) and water (0.5 ml), were added Cs.sub.2CO.sub.3 (1.17 g, 3.58 mmol) and Pd(dppf)Cl.sub.2 (139 mg, 0.171 mmol). The reaction mixture was stirred for 1 hour at room temperature and partitioned between water and AcOEt. The aqueous phase was washed with AcOEt and the gathered organic phases were dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography eluting with DCM/MeOH/NH.sub.4OH 28% 93/6.3/0.07 to give 0.55 g (67%) of (4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-d- ihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (C6).
[0492] LC/MS (m/z, MH.sup.+): 484
Intermediate (D6). (8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}pheny- l)-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate
##STR00277##
[0494] To a solution of (4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-d- ihydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (C6) (550 mg, 1.14 mmol) in THF (30 ml), was added pyridinium tribromide (404 mg, 1.14 mmol). The reaction mixture was stirred for 1 hour at room temperature. A solution of ammonium dihydrogen phosphate (NH.sub.4H.sub.2PO.sub.4) and AcOEt was added. The aqueous phase was washed with AcOEt and the gathered organic phase dried over MgSO.sub.4 and filtered. The organic phase was concentrated under reduced pressure to give 0.63 g (98%) of 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (D6).
[0495] LC/MS (m/z, MH.sup.+): 562
Intermediate (D7). 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-ol
##STR00278##
[0497] To a solution of 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (D6) (640 mg, 1.14 mmol) in MeOH (15 ml), was added NaOH (2N, 2.84 ml, 5.69 mmol). The reaction mixture was stirred for 1 hour at room temperature and 2 ml of aqueous HCl 2N was added and the pH was adjusted to 5 with a solution of aqueous ammonium chloride (NH.sub.4Cl). The solvent was removed under reduced pressure and the residue taken up into AcOEt. The phases were separated and the aqueous phase was washed with AcOEt. The organic phases were combined and dried over MgSO.sub.4, filtered and concentrated under reduced pressure.
[0498] The residue was purified by column chromatography eluting with DCM/MeOH: 95/05 to give 0.47 g (86%) of 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-ol (D7) as a grey solid.
[0499] .sup.1H NMR (400 MHz, DMSO-d6, .delta. ppm): 1.71 to 2.00 (m, 3H); 2.20 (m, 2H); 2.25 to 3.15 (m, 11H); 4.50 (td, J=6.1 and 47.5 Hz, 2H); 4.92 (m, 1H); 6.38 (d, J=8.5 Hz, 1H); 6.71 (t, J=8.5 Hz, 1H); 6.88 (d, J=8.8 Hz, 2H); 7.10 (d, J=8.8 Hz, 2H); 9.93 (s, 1H); 10.03 (m, 1H)
[0500] LC/MS (m/z, MH.sup.+): 478
Intermediate (A9). 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl trifluoromethane sulfonate
##STR00279##
[0502] To a solution of 1-fluoro-2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A7) (5.5 g, 28.32 mmol), in DCM (35 ml) and lutidine (6.66 ml, 56.64 mmol), cooled at 5.degree. C. under argon, was added dropwise trifluoromethanesulfonic anhydride (9.30 ml, 56.64 mmol) while keeping temperature between 10 and 20.degree. C. The reaction mixture was stirred at 5.degree. C. for 1 hour and then at room temperature for 1 hour.
[0503] Ice (50 g) was added and the slurry partitioned between water and DCM. The organic phase was washed with aqueous NaHCO.sub.3 solution, dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with DCM to give 7.05 g (76%) of 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl trifluoromethanesulfonate (A9) as a brown oil.
[0504] LC/MS (m/z, MH.sup.+): 326
Intermediate (A10). Methyl 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate
##STR00280##
[0506] To a solution of 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl trifluoromethanesulfonate (A9) (7 g, 21.46 mmol) in DMF (20 ml) and MeOH (40 ml), were added Pd(dppf)Cl.sub.2 (991.51 mg, 1.29 mmol) and diisopropylethylamine (7.5 ml). The black suspension was carbonylated in an autoclave at 70.degree. C. under 5 bars of CO for 18 hours. The reaction mixture was filtered, then the filtrate was partially concentrated under reduced pressure. AcOEt and water were added to the residue obtained. The organic phase was washed with water and aqueous HCl 0.5 N, dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with DCM to give 3.4 g (67%) of methyl 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate (A10) as a colorless oil.
[0507] LC/MS (m/z, MH.sup.+): 237
Intermediate (B5). Methyl 4-fluoro-9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-- 3-carboxylate
##STR00281##
[0509] To a solution of methyl 1-fluoro-5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate (A10) (1.15 g, 4.87 mmol) in THF (25 ml) cooled at -10.degree. C., was added dropwise potassium bis(trimethylsilyl)amide (1.94 g, 9.74 mmol), followed by N,N-bis(trifluoromethylsulfonyl)aniline (1.95 g, 5.35 mmol). The reaction mixture was stirred for 30 minutes at -10.degree. C. and 20 hours at room temperature. The reaction mixture was cooled to 0.degree. C. and water (500 ml) and AcOEt (200 ml) were added. The organic phase was dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with DCM to give 1.25 g (69%) of methyl 4-fluoro-9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-- 3-carboxylate (B5) as an oil which was used as such in the following step.
[0510] LC/MS (m/z, MH.sup.+): 369
Intermediate (C7). Methyl 4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-di- hydro-5H-benzo[7]annulene-3-carboxylate
##STR00282##
[0512] To a solution under argon of methyl 4-fluoro-9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-- 3-carboxylate (B5) (1.53 g, 4.15 mmol), (S)-1-(3-fluoropropyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- phenoxy)pyrrolidine (1) (1.60 g, 4.57 mmol), in dioxane (10 ml) and water (0.5 ml) were added Pd(dppf)Cl.sub.2, complex with DCM (191.98 mg, 0.25 mmol) and Cs.sub.2CO.sub.3 (2.85 g, 8.72 mmol). The reaction mixture was stirred at 80.degree. C. for 1 hour. After cooling to room temperature, the reaction mixture was poured to a mixture of water (20 ml) and AcOEt (50 ml). The organic phase was washed with brine, dried over MgSO.sub.4, filtered on celite and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with isopropylether/MeOH 95/05 to give 0.7 g (39%) of methyl 4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-di- hydro-5H-benzo[7]annulene-3-carboxylate (C7) as a yellow oil.
[0513] LC/MS (m/z, MH.sup.+): 442
Intermediate (D8). Methyl 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate hydrobromide
##STR00283##
[0515] To a solution of methyl 4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-di- hydro-5H-benzo[7]annulene-3-carboxylate (C7) (900 mg, 2.04 mmol), in DCM (30 ml) was added pyridinium tribromide (880.11 mg, 2.75 mmol). The reaction mixture was stirred under argon for 30 minutes at room temperature. Water (30 ml) was added then organic phase was dried over MgSO.sub.4, and concentrated under reduced pressure. The obtained meringue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 0.8 g (63%) of methyl 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulene-3-carbon/late hydrobromide (D8) as an orange meringue.
[0516] LC/MS (m/z, MH.sup.+): 520
EXAMPLES
Example 1. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hy- droxyphenyl)-8,9-dihydro-7H-benzo[7]annulen-3-ol
##STR00284##
[0518] Method A:
[0519] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-ol (D2) (80 mg, 173.8 .mu.mol) in dioxane/water (80/20; V/V; 4 ml), were added 4-hydroxyphenyl-boronic acid (23.97 mg, 173.77 .mu.mol), Cs.sub.2CO.sub.3 (119.02 mg, 364.92 .mu.mol), and Pd(dppf)Cl.sub.2 (8.51 mg, 10.43 .mu.mol). The reaction mixture was microwaved at 90.degree. C. for 30 minutes, and purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 58 mg (71%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydroxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-3-ol.
Example 3. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-i- ndol-5-yl)-8,9-dihydro-7H-benzo[7]annulen-3-ol
##STR00285##
[0521] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-ol (D2) (80 mg, 173.8 .mu.mol) in dioxane/water (80/20; V/V; 4 ml), were added 5-indolylboronic acid (30.77 mg, 191.15 .mu.mol), Cs.sub.2CO.sub.3(119.02 mg, 364.92 .mu.mol), and Pd(dppf)Cl.sub.2 (8.51 mg, 10.43 .mu.mol). The reaction mixture was microwaved at 90.degree. C. for 30 minutes, and purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 12 mg (14%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indol-5-yl)- -8,9-dihydro-7H-benzo[7]annulen-3-01.
Example 4. 6-(2-chloro-4-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrro- lidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-3-ol
##STR00286##
[0523] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-2-ol (D2) (80 mg, 173.8 .mu.mol) in dioxane/water (80/20; V/V; 4 ml), were added 2-chloro-4-fluorophenylboronic acid (23.10 mg, 132.50 .mu.mol), Cs.sub.2CO.sub.3 (119.02 mg, 364.92 .mu.mol), and Pd(dppf)Cl.sub.2 (8.51 mg, 10.43 .mu.mol). The reaction mixture was microwaved at 90.degree. C. for 30 minutes, and purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 50 mg (74%) of 6-(2-chloro-4-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-3-ol.
Example 5. 6-(2-chloro-4-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrro- lidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00287##
[0525] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (60 mg, 130.33 .mu.mol) in dioxane/water (80/20; V/V; 3 ml), were added 2-chloro-4-fluorophenylboronic acid (23.43 mg, 130.33 .mu.mol), Cs.sub.2CO.sub.3 (89.26 mg, 273.69 .mu.mol), and Pd(dppf)Cl.sub.2 (6.39 mg, 7.82 .mu.mol). The reaction mixture was microwaved at 90.degree. C. for 1 hour and purified by column chromatography eluting with a gradient of methanol in dichloromethane (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 52 mg (78.2%) of 6-(2-chloro-4-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 9. 6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrro- lidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00288##
[0527] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (60 mg, 130.33 .mu.mol) in dioxane/water (80/20; V/V; 3 ml), were added 2-fluoro-4-methylphenylboronic acid (22.99 mg, 143.36 .mu.mol), Cs.sub.2CO.sub.3(89.26 mg, 273.69 .mu.mol), and Pd(dppf)Cl.sub.2 (6.39 mg, 7.82 .mu.mol). The reaction mixture was heated at 80.degree. C. for 1 hour and purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 52 mg (82%) of 6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 11. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydroxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00289##
[0529] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (60 mg, 130.33 .mu.mol), in dioxane/water (80/20; V/V; 3 ml), were added (4-hydroxyphenyl)boronic acid (17.98 mg, 130.33 .mu.mol), Cs.sub.2CO.sub.3 (89.26 mg, 273.69 .mu.mol), and Pd(dppf)Cl.sub.2 (6.39 mg, 7.82 .mu.mol). The reaction mixture was microwaved at 90.degree. C. for 40 minutes and poured in water. The aqueous phase was washed with DCM/MeOH solution (95/5; V/V) and the organic extracts dried over MgSO.sub.4 and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 52 mg (41%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hyd- roxyphenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 21. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-indolin-5-yl-8,- 9-dihydro-7H-benzo[7]annulen-2-ol
##STR00290##
[0531] To a solution of 8-bromo-9-(4-((3S)-1-(3-fluoropropyl)pyrrolidin-3-yl)oxy)phenyl)-6,7-dihy- dro-5H-benzo[7]annulen-3-ol (D4) (50 mg, 108.61 .mu.mol), in dioxane/water (80/20; V/V; 3 ml), were added 5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)indoline (26.62 mg, 108.61 .mu.mol), Cs.sub.2CO.sub.3 (74.39 mg, 228.07 .mu.mol), and Pd(dppf)Cl.sub.2 (5.32 mg, 6.52 .mu.mol).
[0532] The reaction mixture was microwaved at 90.degree. C. for 45 minutes and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 32 mg (59%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-indolin-5-yl-8,- 9-dihydro-7H-benzo[7]annulen-2-ol.
Example 25. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-methyl-3,6-d- ihydro-2H-pyridin-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00291##
[0534] To a solution of 8-bromo-9-(4-((3S)-1-(3-fluoropropyl)pyrrolidin-3-yl)oxy)phenyl)-6,7-dihy- dro-5H-benzo[7]annulen-3-ol (D4) (139.3 mg, 302.58 .mu.mol), in dioxane (2 ml) and water (1 ml), was added 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1,2,3,6-tetrahyd- ropyridine (81.01 mg, 363.09 .mu.mol), Cs.sub.2CO.sub.3 (197.17 mg, 605.15 .mu.mol), and Pd(dppf)Cl.sub.2 (13.28 mg, 18.15 .mu.mol). The reaction mixture was heated at 82.degree. C. for 1.5 hours and partitioned between water and DCM. The aqueous phase was washed with DCM and the organic phase was concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 63.7 mg (44.2%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-methyl-3,6-d- ihydro-2H-pyridin-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 26. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1,2,3,6-tetrah- ydropyridin-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00292##
[0536] To a solution of 4-(5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-hydroxy-8,- 9-dihydro-7H-benzocyclohepten-6-yl)-3,6-dihydro-2H-pyridine-1-carboxylic acid tert-butyl ester (Example 24, 78.2 mg, 138.97 .mu.mol), in MeOH (1.5 ml) was added HCl (120 .mu.l, 4N dioxane solution). The reaction mixture was stirred at room temperature for 2.5 hours and concentrated under reduced pressure. The residue was purified by strong cation exchange (SCX) column to give 60.9 mg (94.7%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1,2,3,6-tetrah- ydropyridin-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 29. 6-(2-fluoro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00293##
[0538] To a solution of 8-bromo-9-(4-((3S)-1-(3-fluoropropyl)pyrrolidin-3-yl)oxy)phenyl)-6,7-dihy- dro-5H-benzo[7]annulen-3-ol (D4) (125.5 mg, 272.60 .mu.mol), in dioxane (2 ml) and water (1 ml), were added 2-fluoro-4-methoxyphenylboronic acid (66.73 mg, 384.80 .mu.mol), Cs.sub.2CO.sub.3 (177.64 mg, 545.20 .mu.mol), and Pd(dppf)Cl.sub.2 (11.97 mg, 16.36 .mu.mol). The reaction mixture was heated at 90.degree. C. for 1 hour and partitioned between water and DCM. The aqueous phase was washed with DCM and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in diisopropyl ether (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 78 mg (56.6%) of 6-(2-fluoro-4-methoxy-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 36. 5-[4-[(3S)-1-(1,1-dideuterio-3-fluoro-propyl)pyrrolidin-3-yl]oxyphenyl]-6- -(2-fluoro-4-methyl-phenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
Step 1. Tert-butyl (3S)-3-(4-{3-[(2,2-dimethylpropanoyl)oxy]-6,7-dihydro-5H-benzo[7]annulen-- 9-yl}phenoxy)pyrrolidine-1-carboxylate (E1)
##STR00294##
[0540] To a solution of 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-d- imethylpropanoate (B2) (6.56 g, 16.72 mmol) in dioxane (45 ml), was added tert-butyl (3S)-3-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidin- e-1-carboxylate (c) (6.51 g, 16.72 mmol), Cs.sub.2CO.sub.3 (23 ml, 34.50 mmol), and Pd(dppf)Cl.sub.2 (1.44 g, 1.67 mmol). The reaction mixture was stirred at room temperature for 24 hours, and partitioned between water and AcOEt. The aqueous phase was extracted with AcOEt and the organic phase dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography, eluting with a mixture of heptane and DCM (60/40; V/V) to give 7.188 g (85%) of tert-butyl (3S)-3-(4-{3-[(2,2-dimethylpropanoyl)oxy]-6,7-dihydro-5H-benzo[7]annulen-- 9-yl}phenoxy)pyrrolidine-1-carboxylate (E1).
[0541] LC/MS (m/z, MH+): 507
Step 2. Tert-butyl (3S)-3-(4-{8-bromo-3-[(2,2-dimethylpropanoyl)oxy]-6,7-dihydro-5H-benzo[7]- annulen-9-yl}phenoxy)pyrrolidine-1-carboxylate (F1)
##STR00295##
[0543] To a solution of tert-butyl (3S)-3(4-{3-[(2,2-dimethylpropanoyl)oxy]-6,7-dihydro-5H-benzo[7]annulen-9- -yl}phenoxy)pyrrolidine-1-carboxylate (E1) (7.18 g, 14.20 mmol) in THF (60 ml), was added pyridinium tribromide (5.00 g, 15.62 mmol). The reaction mixture was stirred at room temperature for 1 hour, and partitioned between water and AcOEt. The aqueous phase was extracted with AcOEt and the organic phase dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography, eluting with a mixture of DCM and MeOH (96/4; V/V) to give 3.43 g (41.3%) of tert-butyl (3S)-3-(4-{8-bromo-3-[(2,2-dimethylpropanoyl)oxy]-6,7-dihydro-5H-benzo[7]- annulen-9-yl}phenoxy)pyrrolidine-1-carboxylate (F1).
[0544] LC/MS (m/z, MH+): 484 and 486 (M-BOC).
Step 3. Tert-butyl (3S)-3-(4-{3-[(2,2-dimethylpropanoyl)oxy]-8-(2-fluoro-4-methylphenyl)-6,7- -dihydro-5H-benzo[7]annulen-9-yl}phenoxy)pyrrolidine-1-carboxylate (G1)
##STR00296##
[0546] To a solution of (3S)-3-(4-{8-bromo-3-[(2,2-dimethylpropanoyl)oxy]-6,7-dihydro-5H-benzo[7]- annulen-9-yl}phenoxy)pyrrolidine-1-carboxylate (F1) (500 mg, 855.37 .mu.mol) in dioxane (5 ml), was added 2-fluoro-4-methylphenylboronic acid (150.89 mg, 940.91 .mu.mol), Cs.sub.2CO.sub.3 (2.5 ml, 3.75 mmol), and Pd(dppf)Cl.sub.2 (65.88 mg, 85.54 .mu.mol). The reaction mixture was heated at 80.degree. C. for 2 hours, and partitioned between water and AcOEt. The aqueous phase was extracted with AcOEt and the organic phase dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography, eluting with a mixture of heptane and DCM (50/50; V/V) to give 285 mg (54.3%) of tert-butyl (3S)-3-(4-{3-[(2,2-dimethylpropanoyl)oxy]-8-(2-fluoro-4-methylphenyl)-6,7- -dihydro-5H-benzo[7]annulen-9-yl}phenoxy)pyrrolidine-1-carboxylate (G1).
[0547] LC/MS (m/z, MH+): 614
Step 4. 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate hydrochloride salt (H1)
##STR00297##
[0549] To a solution of tert-butyl (3S)-3-(4-{3-[(2,2-dimethylpropanoyl)oxy]-8-(2-fluoro-4-methylphenyl)-6,7- -dihydro-5H-benzo[7]annulen-9-yl}phenoxy)pyrrolidine-1-carboxylate (G1) (295 mg, 480.65 .mu.mol) in MeOH (5 ml), was added hydrochloric acid in (4N, 1.20 ml, 4.80 mmol). The reaction mixture was stirred at room temperature for 2 hours and concentrated under reduced pressure to give a solid which was triturated with diisopropyl ether, filtered and dried to give 221 mg (59.5%) of 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-pyrrolidin-3-yl]oxy}phenyl)-6,7-d- ihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate, as an hydrochloride salt (H1).
[0550] LC/MS (m/z, MH+): 514
Step 5. 8-(2-Fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropanoyl)pyrro- lidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (J1)
##STR00298##
[0552] To a solution of 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-pyrrolidin-3-yl]oxy}phenyl)-6,7-d- ihydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (H1) (163 mg, 317.34 .mu.mol) in DMF (3 ml), was added 3-fluoropropanoic acid (30.76 mg, 317.34 .mu.mol), 4-dimethylaminopyridine (121.15 mg, 952.02 .mu.mol), and 1-(3-dimethylaminopropyl)-3-ethyl carbodiimide hydrochloride (76.84 mg, 380.81 .mu.mol). The reaction mixture was stirred at room temperature for 2 hours, and partitioned between water and AcOEt. The aqueous phase was extracted with AcOEt and the organic phase dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography, eluting with a mixture of DCM and MeOH (97/3; V/V) to give 180 mg (96.5%) of 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropanoyl)pyrrolidin-3- -yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (J1).
[0553] LC/MS (m/z, MH+): 588
Step 6. 5-[4-[(3S)-1-(1,1-dideuterio-3-fluoro-propyl)pyrrolidin-3-yl]oxyph- enyl]-6-(2-fluoro-4-methyl-phenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol (Ic)
##STR00299##
[0555] To a solution of 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropanoyl)pyrrolidin-3- -yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (J1) (180 mg, 306.28 .mu.mol) in diethylether (5 ml), was added lithium aluminum deuteride (39.36 mg, 918.84 .mu.mol). The reaction mixture was stirred at room temperature for 2 hours, diluted with DCM and a solution of sodium potassium bis-tartrate (1N) was added. The solid formed was filtered and the filtrate was dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography, eluting with a mixture of DCM and MeOH (97/3; V/V) to give 36 mg (23.9%) of 5-[4-[(3S)-1-(1,1-dideuterio-3-fluoro-propyl)pyrrolidin-3-yl]oxyphenyl]-6- -(2-fluoro-4-methyl-phenyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol (lc).
Example 39. 6-(3-chloro-2-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
Step 1: 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-(4,4,5- ,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-6,7-dihydro-5H-benzo[7]annulen-3-o- l (D')
##STR00300##
[0557] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (2.03 g, 4.41 mmol), in dioxane (25 ml) and water (10 ml), was added 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (1.34 g, 5.29 mmol), Cs.sub.2CO.sub.3 (2.88 g, 8.82 mmol), and Pd(dppf)Cl.sub.2 (203.77 mg, 264.56 .mu.mol). The reaction mixture was heated at 70.degree. C. for 45 minutes, and partitioned between DCM and water. The phases were separated and the organic phase concentrated under reduced pressure. The residue was first purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a crude solid, which was further separated on chiralpak AD 20 .mu.m, eluting with a mixture of heptane, ethanol and triethylamine (90/9.9/0.1; V/V/V) to give 967 mg (43%) of 5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-6-(4,4- ,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)-8,9-dihydro-7H-benzocyclohepten- -2-ol (D').
[0558] LC/MS (m/z, MH.sup.+): 509
Step 2: 6-(3-chloro-2-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00301##
[0560] To a solution of 5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-6-(4,4,5,5-tetr- amethyl-[1,3,2]dioxaborolan-2-yl)-8,9-dihydro-7H-benzocyclohepten-2-ol (D') (100.3 mg, 197.66 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 3-chloro-2-fluoroiodobenzene (60.83 mg, 237.19 .mu.mol), Cs.sub.2CO.sub.3 (128.93 mg, 395.31 .mu.mol) and Pd(dppf)Cl.sub.2 (9.68 mg, 11.86 .mu.mol). The reaction mixture was heated at 70.degree. C. for 6 hours, and partitioned between DCM and water. The aqueous phase was washed with DCM and organic phases were dried and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 18 mg (18%) of 6-(3-chloro-2-fluoro-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 45. 1-fluoro-6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00302##
[0562] To a solution of 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-ol (D7) (60 mg, 125.43 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 2-fluoro-4-methylphenylboronic acid (22.12 mg, 137.97 .mu.mol), Cs.sub.2CO.sub.3 (81.73 mg, 250.85 .mu.mol), and Pd(dppf)Cl.sub.2 (6.15 mg, 7.53 .mu.mol). The reaction mixture was heated at 80.degree. C. for 30 minutes and the solid formed, filtered and washed with dioxane. The filtrate was concentrated under reduced pressure and the residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 45 mg (71%) of 1-fluoro-6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 48. 6-(2-fluoro-4-methyl-phenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
[0563] Method C
Step 1. 8-(2-Fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl trifluoromethanesulfonate
##STR00303##
[0565] To a solution of 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-ol (Example 9, 840 mg, 1.60 mmol), in DCM (30 ml), was added pyridine (387.4 .mu.l, 4.79 mmol), and trifluoromethanesulfonic anhydride (839.5 .mu.l, 4.79 mmol). The reaction mixture was stirred at room temperature for 16 hours, poured onto ice and partitioned between water and DCM. The aqueous phase was washed with DCM and the gathered organic phases, washed successively with a saturated solution of NaHCO.sub.3, and brine. The organic phase was dried over MgSO.sub.4, filtered, and the filtrate was concentrated under reduced pressure to give 860 mg (86.6%) of crude 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yltrifluoromethanesulfonate.
[0566] LC/MS (m/z, MH.sup.+): 622
Step 2. Methyl 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-carboxylate
##STR00304##
[0568] To a solution of 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl trifluoromethanesulfonate (860 mg, 1.35 mmol), in DMF (10 ml) and MeOH (5 ml), was added triethylamine (1 ml), Pd(OAc).sub.2 (60.52 mg, 269.54 .mu.mol), and 1,3-bis(diphenylphosphino)propane (dppp) (115.80 mg, 269.54 .mu.mol). The reaction mixture was heated at 40.degree. C., under an atmosphere of CO (2 bars), for 16 hours, and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of cyclohexane and AcOEt (80/20; V/V) to give 400 mg (55.8%) of methyl 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-carboxylate.
[0569] LC/MS (m/z, MH.sup.+): 532
Step 3. 8-(2-Fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylic acid
##STR00305##
[0571] To a solution of methyl 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-carboxylate (390 mg, 733.59 .mu.mol), MeOH (20 ml), was added NaOH solution (5N, 1.5 ml). The reaction mixture was heated at 60.degree. C., for 2 hours, and concentrated under reduced pressure. The residue was taken up into water (25 ml), and acidified with aqueous HCl (5N, 1.5 ml), and the solid formed was filtered, washed with water and dried under vacuum. The residue was purified by trituration in diisopropyl ether to give 180 mg (47.4%) of 8-(2-fluoro-4-methylphenyl)-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylic acid.
Example 51. 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
[0572] Method B:
Step 1: 6-(2,4-Dichloro-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-ylo- xy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester
##STR00306##
[0574] To a solution of methyl 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulene-3-carboxylate hydrobromide (D5) (150 mg, 298.56 .mu.mol), in dioxane (12 ml) and water (2 ml), was added 2,4-dichlorophenyl-boronic acid (62.67 mg, 328.41 .mu.mol), Cs.sub.2CO.sub.3 (204.48 mg, 626.97 .mu.mol), and Pd(dppf)Cl.sub.2 (14.63 mg, 17.91 .mu.mol). The reaction mixture was heated at 90.degree. C. for 3 hours, and partitioned between AcOEt and water. The phases were separated and the organic phase washed with brine, dried over MgSO.sub.4 and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (96/2/2; V/V/V) to give 80 mg (47%) of 6-(2,4-dichloro-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phe- nyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester.
[0575] LC/MS (m/z, MH.sup.+): 568
Step 2: 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
##STR00307##
[0577] To a solution of 6-(2,4-dichloro-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phe- nyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester (80 mg, 140.72 .mu.mol) in MeOH (5 ml) was added a solution of NaOH (562.88 .mu.l, 5 M) and the reaction mixture was heated at 60.degree. C. for 5 hours and the solvent removed under reduced pressure. The residue was taken up in water (10 ml) and aqueous HCl (5 M) added to pH 7. The slurry was extracted with DCM, dried over MgSO.sub.4 and concentrated under reduced pressure. The solid was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (90/5/5; V/V/V) to give 60 mg (77%) of 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyph- enyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid.
Example 63. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-methoxy-2-me- thyl-phenyl)-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride
Step 1: 6-(4-Methoxy-2-methyl-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin- -3-yloxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester
##STR00308##
[0579] To a solution of methyl 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulene-3-carboxylate hydrobromide (D5) (250 mg, 497.60 .mu.mol), in dioxane (12 ml) and water (2 ml), was added 4-methoxy-2-methylphenyl-boronic acid (90.85 mg, 547.36 .mu.mol), Cs.sub.2CO.sub.3 (340.81 mg, 1.04 mmol), and Pd(dppf)Cl.sub.2 (24.38 mg, 29.86 .mu.mol). The reaction mixture was heated at 90.degree. C. for 2 hours, and partitioned between AcOEt and water. The phases were separated and the organic phase washed with brine, dried over MgSO.sub.4 and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (96/2/2; V/V/V) to give 280 mg (100%) of crude 6-(4-methoxy-2-methyl-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-ylox- y]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester.
[0580] LC/MS (m/z, MH.sup.+): 544
Step 2: 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-metho- xy-2-methyl-phenyl)-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride
##STR00309##
[0582] To a solution of 6-(4-methoxy-2-methyl-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-ylox- y]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester (280 mg, 543.668 .mu.mol) in MeOH (10 ml) was added a solution of NaOH (5 M, 1.5 ml) and the reaction mixture was heated at 60.degree. C. for 6 hours and the solvent removed under reduced pressure. The residue was taken up in water (25 ml) and aqueous HCl (5 M) was added to pH 7. The slurry was extracted with DCM, dried over MgSO.sub.4 and concentrated under reduced pressure. The solid was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (90/5/5; V/V/V) to give a solid. This solid was triturated in diisopropyl ether with anhydrous HCl (2 M in diethyl ether) to give a solid which was filtered and dried to give 134 mg (46%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-methoxy-2-me- thyl-phenyl)-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride.
Example 70. 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
Step 1: 1-[5-(5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-- hydroxy-8,9-dihydro-7H-benzocyclohepten-6-yl)-2,2-dimethyl-2,3-dihydro-ind- ol-1-yl]-ethanone
##STR00310##
[0584] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (93.8 mg, 203.75 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 1-(2,2-dimethyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)indolin-1-- yl)ethanone (64.72 mg, 205.32 .mu.mol), Cs.sub.2CO.sub.3 (132.90 mg, 407.49 .mu.mol), and Pd(dppf)Cl.sub.2 (9.98 mg, 12.22 .mu.mol). The reaction mixture was heated at 72.degree. C. for 45 minutes, and partitioned between DCM and water. The phases were separated on hydrophobic interaction column and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 77 mg (67%) of 1-[5-(5-{4-[(S)-1-(3-Fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-- 2-hydroxy-8,9-dihydro-7H-benzocyclohepten-6-yl)-2,2-dimethyl-2,3-dihydro-i- ndol-1-yl]-ethanone.
[0585] LC/MS (m/z, MH.sup.+): 569
Step 2: 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00311##
[0587] To a solution of (S)-1-(5-(9-(4-((1-(3-fluoropropyl)pyrrolidin-3-yl)oxy)phenyl)-3-hydroxy-- 6,7-dihydro-5H-benzo[7]annulen-8-yl)-2,2-dimethylindolin-1-yl)ethanone (73 mg, 128.36 .mu.mol) in dioxane (1.9 ml), was added aqueous HCl (1N, 1.5 ml) and the reaction mixture heated in a microwave oven at 120.degree. C. for 2 hours. The reaction mixture was poured onto a saturated aqueous solution of NaHCO.sub.3, and extracted with DCM. The phases were separated on hydrophobic interaction column and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 39 mg (58%) of 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-01.
Example 73. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1,2,3,4-tetrah- ydroquinolin-6-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
Step 1: 6-(5-{4-[(S)-1-(3-Fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-hyd- roxy-8,9-dihydro-7H-benzocyclohepten-6-yl)-3,4-dihydro-2H-quinoline-1-carb- oxylic acid tert-butyl ester
##STR00312##
[0589] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (93.4 mg, 202.88 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added tert-butyl 6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-3,4-dihydroquinoline-1(2H- )-carboxylate (85.18 mg, 237.09 .mu.mol), Cs.sub.2CO.sub.3 (132.33 mg, 405.75 .mu.mol), and Pd(dppf)Cl.sub.2 (9 9.94 mg, 12.17 .mu.mol). The reaction mixture was heated at 72.degree. C. for 45 minutes, and partitioned between DCM and water. The phases were separated on hydrophobic interaction column and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 75 mg (60.3%) of 6-(5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-hydroxy- -8,9-dihydro-7H-benzocyclohepten-6-yl)-3,4-dihydro-2H-quinoline-1-carboxyl- ic acid tert-butyl ester.
[0590] LC/MS (m/z, MH.sup.+): 613
Step 2: 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00313##
[0592] To a solution of 6-(5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-hydroxy-8,- 9-dihydro-7H-benzocyclohepten-6-yl)-3,4-dihydro-2H-quinoline-1-carboxylic acid tert-butyl ester in DCM (2.4 ml) was added HCl (1M in diethylether, 1.17 ml) and the reaction mixture stirred at room temperature for 18 hours. A saturated aqueous NaHCO.sub.3 solution was added, and the aqueous phase was extracted with DCM. The phases were separated on hydrophobic interaction column and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 57.3 mg (95.1%) of 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 75. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trifluorome- thoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00314##
[0594] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (92.9 mg, 201.79 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 4-(trifluoromethoxy)phenylboronic acid (54.12 mg, 254.93 .mu.mol), Cs.sub.2CO.sub.3 (131.63 mg, 403.58 .mu.mol), and Pd(dppf)Cl.sub.2 (9.89 mg, 12.11 .mu.mol). The reaction mixture was heated at 72.degree. C. for 45 minutes, and partitioned between DCM and water. The phases were separated on hydrophobic interaction column and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 71.3 mg (61.4%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-trifluoromet- hoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 76. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-methoxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00315##
[0596] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (74.9 mg, 162.69 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 4-methoxyphenylboronic acid (34.49 mg, 222.45 .mu.mol), Cs.sub.2CO.sub.3 (131.91 mg, 404.45 .mu.mol), and Pd(dppf)Cl.sub.2 (9.91 mg, 12.13 .mu.mol). The reaction mixture was heated at 72.degree. C. for 45 minutes, and partitioned between DCM and water. The phases were separated on hydrophobic interaction column and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 63.9 mg (64.8%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-methoxypheny- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 82. 6-(6-ethoxy-2-fluoro-3-pyridyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyr- rolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00316##
[0598] To a solution of 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-ol (D7) (60 mg, 125.43 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 6-ethoxy-2-fluoropyridin-3-yl boronic acid (25.52 mg, 137.97 .mu.mol), Cs.sub.2CO.sub.3 (171.05 mg, 525.0 .mu.mol), and Pd(dppf)Cl.sub.2 (9.66 mg, 12.54 .mu.mol). The reaction mixture was heated at 60.degree. C. for 1 hour and partitioned between water and AcOEt. The aqueous phase was washed with AcOEt and the organic extracts dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified twice by column chromatography eluting first with a mixture of diisopropyl ether/MeOH (90/10; V/V) and with a mixture of DCM/MeOH (98/2) to give 38 mg (56.3%) of 6-(6-ethoxy-2-fluoro-3-pyridyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyr- rolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 107. 6-(2-ethoxypyrimidin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]o- xyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00317##
[0600] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (74.9 mg, 162.69 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 2-ethoxypyrimidin-5-yl boronic acid (30.06 mg, 178.96 .mu.mol), Cs.sub.2CO.sub.3(106.12 mg, 325.38 .mu.mol), and Pd(dppf)Cl.sub.2 (7.97 mg, 9.76 .mu.mol). The reaction mixture was heated at 72.degree. C. for 1 hour, partitioned between water and DCM and phases separated on hydrophobic partition column. The organic solvents were concentrated under reduced pressure and the residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 47.3 mg (57.7%) of 6-(2-ethoxypyrimidin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]o- xyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 108. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(6-methoxy-3-py- ridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00318##
[0602] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (90 mg, 195.49 .mu.mol), in dioxane/water (80/20; V/V; 4 ml), was added 2-methoxy-5-pyridineboronic acid (37.77 mg, 234.59 .mu.mol), Cs.sub.2CO.sub.3 (133.89 mg, 410.53 .mu.mol), and Pd(dppf)Cl.sub.2 (9.58 mg, 11.73 .mu.mol). The reaction mixture was heated in a microwave at 90.degree. C. for 30 minutes, and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 4%) to give a solid which was further purified on strong cation exchange (SCX) column to give 59 mg (61.8%) of 5-[4-[(3S)-1-(3-fluoropropyl) pyrrolidin-3-yl]oxyphenyl]-6-(6-methoxy-3-pyridyl)-8,9-dihydro-7H-benzo[7- ]annulen-2-ol.
Example 109. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(2-methoxy-4-py- ridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00319##
[0604] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (90 mg, 195.49 .mu.mol), in dioxane/water (80/20; V/V; 4 ml), was added 2-methoxypyridine-4-boronic acid (36.99 mg, 234.59 .mu.mol), Cs.sub.2CO.sub.3 (133.89 mg, 410.53 .mu.mol), and Pd(dppf)Cl.sub.2 (9.58 mg, 11.73 .mu.mol). The reaction mixture was heated in a microwave at 90.degree. C. for 30 minutes, and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 4%) to give a solid which was further purified on strong cation exchange (SCX) column to give 60 mg (62.8%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(2-methoxy-4-py- ridyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 114. 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(tr- ifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00320##
[0606] To a solution of 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulen-3-ol (D7) (60 mg, 125.43 .mu.mol), in dioxane (1 ml) and water (0.5 ml), was added 4-(trifluoromethoxy)phenylboronic acid (29 mg, 137.97 .mu.mol), Cs.sub.2CO.sub.3 (81.73 mg, 250.85 .mu.mol), and Pd(dppf)Cl.sub.2 (6.15 mg, 7.53 .mu.mol). The reaction mixture was heated at 80.degree. C. for 30 minutes and the solid filtered and washed with dioxane. The filtrate was concentrated under educed pressure and the residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 45 mg (71%) of 1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(tr- ifluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 163. [5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trifluorom- ethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-yl] dihydrogen phosphate
Step 1: Diethyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-[4-(trifluoro- methoxy)phenyl]-6,7-dihydro-5H-benzo[7]annulen-3-yl phosphate
##STR00321##
[0608] To a solution of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-trifluoromet- hoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol (Example 75, 312 mg, 576.10 .mu.mol), in acetonitrile (3 ml), was added triethylamine (353.1 .mu.l, 2.54 mmol), and diethyl chlorophosphate (249.76 .mu.l, 1.73 mmol). The reaction mixture was stirred at room temperature for 28 hours, and concentrated under reduced pressure. The residue was purified by strong cation exchange (SCX) column to give 256 mg (65.6%) of diethyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-[4-(trifluoro- methoxy)phenyl]-6,7-dihydro-5H-benzo[7]annulen-3-yl phosphate.
[0609] LC/MS (m/z, MH.sup.+): 678
Step 2: [5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(tri- fluoromethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-yl] dihydrogen phosphate
##STR00322##
[0611] To a solution of diethyl 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-[4-(trifluoro- methoxy)phenyl]-6,7-dihydro-5H-benzo[7]annulen-3-yl phosphate (256 mg, 377.77 .mu.mol), in acetonitrile (6 ml), was added iodotrimethylsilane (277.12 .mu.l, 1.89 mmol). The reaction mixture was stirred at room temperature for 1 hour, and concentrated under reduced pressure. The residue was purified by strong cation exchange (SCX) column and reverse phase column chromatography, eluting with a gradient of acetonitrile in water (20% to 80%) to give 167 mg (70.3%) of [5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-[4-(trifluorom- ethoxy)phenyl]-8,9-dihydro-7H-benzo[7]annulen-2-yl] dihydrogen phosphate.
Example 174. 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride
Step 1: Methyl 8-(2,2-dimethyl-2,3-dihydro-1H-indol-5-yl)-9-(4-{[(3S)-1-(3-fluoropropyl)- pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate
##STR00323##
[0613] To a solution of methyl 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulene-3-carboxylate hydrobromide (D5) (500 mg, 845.91 .mu.mol), in dioxane (12 ml) and water (2 ml), was added 1-(2,2-dimethyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)indolin-1-- yl)ethanone (279.98 mg, 888.21 .mu.mol), Cs.sub.2CO.sub.3 (744.91 mg, 2.28 mmol), and Pd(dppf)Cl.sub.2 (41.45 mg, 50.75 .mu.mol). The reaction mixture was heated in a microwave at 110.degree. C. for 1 hour, DCM was added and the organic phase washed with saturated NH.sub.4Cl solution. The organic phase was dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (96/2/2; V/V/V) to give 250 mg (48.4%) of methyl 8-(2,2-dimethyl-2,3-dihydro-1H-indol-5-yl)-9-(4-{[(3S)-1-(3-fluoropropyl)- pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate.
[0614] LC/MS (m/z, MH.sup.+): 611
Step 2: 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolid- in-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride
##STR00324##
[0616] To a solution of methyl 8-(2,2-dimethyl-2,3-dihydro-1H-indol-5-yl)-9-(4-{[(3S)-1-(3-fluoropropyl)- pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate (240 mg, 392.95 .mu.mol) in MeOH (20 ml) was added NaOH (15.72 mg, 392.95 .mu.mol) and the reaction mixture was heated under reflux for 3 hours and the solvent removed under reduced pressure. The residue was taken up in water (15 ml), HCl (5 M, 1 ml) was added and the reaction mixture was heated under reflux for 2 hours. NaOH solution was added to pH 7, and the aqueous phase extracted with DCM. The organic phase was washed with brine, dried over MgSO.sub.4, filtered and concentrated under reduced pressure. The residue was triturated with diisopropyl ether, filtered and dried to give 211 mg (90.8%) of 6-(2,2-dimethylindolin-5-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl- ]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid hydrochloride.
Example 189. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-oxidopyridin- -1-ium-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
Step 1: 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-(4,4,5- ,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-6,7-dihydro-5H-benzo[7]annulen-3-y- l 2,2-dimethylpropanoate
##STR00325##
[0618] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (D3) (1 g, 1.84 mmol), in dioxane (10 ml) and water (5 ml), was added 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (559.65 mg, 2.20 mmol), Cs.sub.2CO.sub.3 (1.20 g, 3.67 mmol), and Pd(dppf)Cl.sub.2 (84.87 mg, 110.19 .mu.mol). The reaction mixture was heated under reflux for 24 hours, and partitioned between DCM and water. The aqueous phase was washed with DCM and the gathered organic phase dried over hydrophobic partition column, and evaporated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of DCM and MeOH (98/2; V/V) to give 426 mg (39.2%) of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-(4,4,5,5-tetr- amethyl-1,3,2-dioxaborolan-2-yl)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate.
[0619] LC/MS (m/z, MH+): 592 (M+H).
Step 2: [9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-3-hydro- xy-6,7-dihydro-5H-benzo[7]annulen-8-yl]boronic acid
##STR00326##
[0621] To a solution of 9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-8-(4,4,5,5-tetr- amethyl-1,3,2-dioxaborolan-2-yl)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (426 mg, 720.13 .mu.mol), in MeOH (10 ml), was added NaOH (2N, 2.16 ml, 4.32 mmol). The reaction mixture was stirred at room temperature for 1.5 hour, and HCl (2N, 2.2 ml) was added. The aqueous phases was washed with DCM, and evaporated under reduced pressure. The residue was triturated with a mixture of DCM and MeOH (95/5, V/V), filtered and concentrated under reduced pressure to give 100 mg (32.7%) of crude [9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-3-hydroxy-6,7-- dihydro-5H-benzo[7]annulen-8-yl]boronic acid.
[0622] LC/MS (m/z, MH+): 426 (M+H).
Step 3: 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-oxido- pyridin-1-ium-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00327##
[0624] To a solution of [9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-3-hydroxy-6,7-- dihydro-5H-benzo[7]annulen-8-yl]boronic acid (100 mg, 235.12 .mu.mol), in dioxane (8 ml) and water (2 ml), 4-bromopyridine 1-oxide (57.28 mg, 329.17 .mu.mol), Cs.sub.2CO.sub.3 (161.04 mg, 493.76 .mu.mol) and Pd(dppf)Cl.sub.2 (11.52 mg, 14.11 .mu.mol). The reaction mixture was heated at 90.degree. C. for 2 hours, and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 4%) to give a solid which was further purified on strong cation exchange (SCX) column to give 21 mg (18.8%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1-oxidopyridin- -1-ium-4-yl)-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 203. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-tetrahydropyran- -4-yl-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00328##
[0626] To a solution of 6-(3,6-dihydro-2H-pyran-4-yl)-5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin-3-- yloxy]-phenyl}-8,9-dihydro-7H-benzocyclohepten-2-ol (Example 202, 110 mg, 237.28 .mu.mol) in AcOEt (3 ml) and ethanol (5 ml), was added palladium on carbon (10%, 2.53 mg, 23.73 .mu.mol). The reaction mixture was stirred at 50.degree. C. under hydrogen atmosphere (5 bars) for 2 hours. The reaction mixture was filtered on celite, washed with MeOH, and the filtrate was concentrated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of DCM and MeOH (90/10; V/V)) to give 61 mg (55%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-tetrahydropyran- -4-yl-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 204. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydroxycyclo- hexyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
Step 1: 6-(1,4-Dioxa-spiro[4.5]dec-7-en-8-yl)-5-{4-[(S)-1-(3-fluoro-propyl- )-pyrrolidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocyclohepten-2-ol
##STR00329##
[0628] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (500 mg, 1.09 mmol), in dioxane/water (80/20; V/V, 25 ml), was added 1,4-dioxa-spiro[4,5]dec-7-en-8-boronic acid pinacoyl ester (412.93 mg, 1.52 mmol), Cs.sub.2CO.sub.3 (743.85 mg, 2.28 mmol), and Pd(dppf)Cl.sub.2 (53.22 mg, 65.16 .mu.mol). The reaction mixture was heated at 80.degree. C. for 30 minutes, and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 4%) to give a solid which was further purified on strong cation exchange (SCX) column to give 485 mg (85.9%) of 6-(1,4-dioxa-spiro[4.5]dec-7-en-8-yl)-5-{4-[(S)-1-(3-fluoro-propyl)-pyrro- lidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocyclohepten-2-ol.
[0629] LC/MS (m/z, MH+): 520
Step 2: 6-(1,4-Dioxa-spiro[4.5]dec-8-yl)-5-{4-[(S)-1-(3-fluoro-propyl)-pyr- rolidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocyclohepten-2-ol
##STR00330##
[0631] To a solution of 6-(1,4-dioxa-spiro[4.5]dec-7-en-8-yl)-5-{4-[(S)-1-(3-fluoro-propyl)-pyrro- lidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocyclohepten-2-ol (485 mg, 933.31 .mu.mol), in AcOEt (10 ml) and ethanol (10 ml), was added palladium on carbon (10%, 9.93 mg, 93.33 .mu.mol). The reaction mixture was stirred at 50.degree. C. under hydrogen atmosphere (5 bars) for 24 hours. The reaction mixture was filtered over celite, rinsed with MeOH and the filtrate was concentrated under reduced pressure to give 487 mg (100%) of crude 6-(1,4-dioxa-spiro[4.5]dec-8-yl)-5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin- -3-yloxy]-phenyl}-8,9-dihydro-7H-benzocyclohepten-2-ol.
[0632] LC/MS (m/z, MH.sup.+): 522
Step 3: 4-(5-{4-[(S)-1-(3-Fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-hyd- roxy-8,9-dihydro-7H-benzocyclohepten-6-yl)-cyclohexanone
##STR00331##
[0634] To a solution of crude 6-(1,4-dioxa-spiro[4.5]dec-8-yl)-5-{4-[(S)-1-(3-fluoro-propyl)-pyrrolidin- -3-yloxy]-phenyl}-8,9-dihydro-7H-benzocyclohepten-2-ol (487 mg, 933.53 .mu.mol) in acetone (2 ml) was added concentrated aqueous HCl (1.4 ml) and the reaction mixture was stirred at room temperature for 4 days. A saturated aqueous solution of NaHCO.sub.3 and DCM was added and the phases were separated on hydrophobic interaction column. The organic phase was concentrated under reduced pressure and the residue purified by column chromatography eluting with a mixture of MeOH in DCM (3/97; V/V) to give 390 mg (87.5%) of 4-(5-{4-[(S)-1-(3-Fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-hydroxy-8,- 9-dihydro-7H-benzocyclohepten-6-yl)-cyclohexanone.
[0635] LC/MS (m/z, MH.sup.+): 478.
Step 4: 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydro- xycyclohexyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00332##
[0637] To a solution of 4-(5-{4-[(S)-1-(3-Fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-2-hydroxy-8,- 9-dihydro-7H-benzocyclohepten-6-yl)-cyclohexanone (200 mg, 418.74 .mu.mol), in MeOH (4 ml), was added sodium borohydride (147.3 mg, 3.89 mmol). The reaction mixture was stirred at room temperature for 4 days, and water was added. A solid forms which was filtered, rinsed with water and purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 28 mg (14%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(4-hydroxycyclo- hexyl)-8,9-dihydro-7H-benzo[7]annulen-2-ol
Example 207. 6-(4,4-difluorocyclohexen-1-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00333##
[0639] To a solution of 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dih- ydro-5H-benzo[7]annulen-3-ol (D4) (200 mg, 434.42 .mu.mol) in dioxane/water (80/20; V/V, 10 ml), was added 2-(4,4-difluorocyclohex-1-en-1-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolan- e (127.24 mg, 521.30 .mu.mol), Cs.sub.2CO.sub.3 (297.53 mg, 912.28 .mu.mol), and Pd(dppf)Cl.sub.2 (20.08 mg, 26.07 .mu.mol). The reaction mixture was heated at 90.degree. C. for 30 minutes, and the solvent was concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give a solid which was further purified on strong cation exchange (SCX) column to give 95 mg (44%) of 6-(4,4-difluorocyclohexen-1-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 208. 6-(4,4-difluorocyclohexyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]o- xyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00334##
[0641] To a solution of 6-(4,4-difluorocyclohexen-1-yl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- -yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol (95 mg, 190.92 .mu.mol), in AcOEt (3 ml) and ethanol (5 ml) was added palladium on carbon (10%, 2.03 mg, 19.09 .mu.mol). The reaction mixture was stirred at 50.degree. C. under hydrogen atmosphere (5 bars) for 2 hours and filtered on celite, rinsed with MeOH and the filtrate was concentrated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of MeOH in DCM (3/97; V/V) to give 70 mg (73.4%) of 6-(4,4-difluorocyclohexyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-y- l]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen-2-ol.
Example 213. 6-(2-chloro-4-fluoro-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
Step 1: Methyl 8-(2-chloro-4-fluorophenyl)-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate
##STR00335##
[0643] To a solution of methyl 8-bromo-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl- )-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate, hydrobromide (D8) (200 mg, 332.60 .mu.mol) in dioxane (15 ml) was added 2-chloro-4-fluorophenyl boronic acid (69.59 mg, 399.12 .mu.mol), Cs.sub.2CO.sub.3(465.64 .mu.l, 698.46 .mu.mol), and Pd(dppf)Cl.sub.2 (15.37 mg, 19.96 .mu.mol). The reaction mixture was heated at 70.degree. C. for 1 hour, and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 5%), to give a solid which was further purified on strong cation exchange (SCX) column to give 170 mg (89.7%) of methyl 8-(2-chloro-4-fluorophenyl)-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate.
[0644] LC/MS (m/z, MH.sup.+): 570
Step 2: 6-(2-chloro-4-fluoro-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl- )pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
##STR00336##
[0646] To a solution of methyl 8-(2-chloro-4-fluorophenyl)-4-fluoro-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate (170 mg, 298.22 .mu.mol) in MeOH (150 ml), was added NaOH 5 M (238.58 .mu.l, 1.19 mmol). The reaction mixture was heated at 90.degree. C. for 2 hours, aqueous HCl (5 N) was added, and purified on strong cation exchange (SCX) column to give 65 mg (39.2%) of 6-(2-chloro-4-fluoro-phenyl)-1-fluoro-5-[4-[(3S)-1-(3-fluoropropyl)pyrrol- idin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid.
Example 215. 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indazol-5-y- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol
##STR00337##
[0648] To a solution of 8-bromo-9-(4-((3S)-1-(3-fluoropropyl)pyrrolidin-3-yl)oxy)phenyl)-6,7-dihy- dro-5H-benzo[7]annulen-3-ol (D4) (306.6 mg, 665.97 .mu.mol), in dioxane (4 mL) and water (0.5 ml), was added 5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-indazole (201 mg, 823.43 .mu.mol), Cs.sub.2CO.sub.3 (557 mg, 1.71 mmol), and Pd(dppf)Cl.sub.2 (70 mg, 85.72 .mu.mol). The reaction mixture was heated at 72.degree. C. for 4 hours and partitioned between water and DCM. The phases were separated on hydrophobic interaction column and the organic phase concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0% to 10%) to give 26 mg (8%) of 5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-6-(1H-indazol-5-y- l)-8,9-dihydro-7H-benzo[7]annulen-2-ol.
[0649] Some compounds of formula (I) were subjected to pharmacological tests for determining their antagonist and degradation effects on estrogen receptors.
[0650] Test A: Biochemical Antagonist Activity on Wild Type (WT) and Mutants Estrogen Receptors
[0651] Test A involves measuring the in vitro antagonist activity of a compound of formula (I) on estrogen receptors.
[0652] The measurements of the antagonist activities were made using an estrogen receptor coactivator assay as described hereunder.
[0653] Antagonistic potency of compounds was evaluated using LanthaScreen.RTM. TR-FRET ER.alpha. Coactivator Assay (ThermoFisher) with modifications. It is a competition assay, where binding of a test compound to a complex comprised of (i) His6-ER.alpha.298-554 protein representing ER.alpha. ligand-binding domain, (ii) Tb-labeled His6 antibody, (iii) a fluorescein-labeled PGC1a coactivator peptide (EAEEPSLLKKLLLAPANTQ), and (iv) estradiol, results in a decrease of the TR-FRET signal due to dissociation of the coactivator peptide. His6-ER.alpha.298-554 proteins were expressed as VVT or D538G or Y537S mutants in E. coli and purified by affinity chromatography. The assay works in a homogeneous mix-and-read format. In a typical experiment, a 4 .mu.L mixture of 0.5 nM His6-ER.alpha.298-554, 0.5 nM Tb-labeled His6 antibody, 250 nM PGC1a peptide, and 3 nM estradiol in 100 mM potassium phosphate, pH 7.4, 0.01% Tween-20, 0.02% NaN.sub.3, 5 mM DTT, was added to 40 nL test compound in DMSO and incubated overnight at room temperature. The TR-FRET 520:495 nm emission ratio was calculated and used to determine the IC50 value from a dose response curve fit to the 4-parameter logistic equation.
[0654] The antagonist activity with respect to estrogen receptors in this test is given by the concentration which inhibits 50% of the estrogen receptor activity (or IC50) in nM.
[0655] The Table 2 below indicates the biochemical results of antagonist activity on VVT and mutants estrogen receptors for compounds of formula (I), and demonstrates that the compounds tested have an antagonist activity regarding estrogen receptors.
TABLE-US-00002 TABLE 2 Antagonism Antagonism Antagonism WT D538G Y537S Examples IC.sub.50 (nM) IC.sub.50 (nM) IC.sub.50 (nM) Example 1 7 32 22 Example 2 5 30 22 Example 3 37 268 139 Example 4 19 117 68 Example 5 7 21 21 Example 6 2 4 3 Example 7 5 36 19 Example 8 3 15 8 Example 9 3 20 10 Example 10 2 5 3 Example 11 2 4 3 Example 12 3 18 10 Example 13 4 19 11 Example 14 3 15 7 Example 15 5 37 24 Example 16 14 109 63 Example 17 3 24 15 Example 18 5 28 21 Example 19 8 49 26 Example 20 2 12 9 Example 21 7 30 26 Example 22 7 45 26 Example 23 3 9 4 Example 24 152 759 475 Example 25 284 937 726 Example 26 199 443 341 Example 27 3 10 6 Example 28 2 5 4 Example 29 2 4 2 Example 30 5 20 9 Example 31 2 19 12 Example 32 3 32 24 Example 33 3 41 24 Example 34 2 18 11 Example 35 12 244 134 Example 36 3 90 50 Example 37 14 309 241 Example 38 39 652 651 Example 39 4 76 53 Example 40 16 289 224 Example 41 10 177 133 Example 42 2 55 33 Example 43 0.8 14 11 Example 44 1 16 12 Example 45 2 40 23 Example 46 1 21 16 Example 47 2 31 23 Example 48 44 1119 549 Example 49 1 11 6 Example 50 10 208 113 Example 51 15 389 221 Example 52 2 49 29 Example 53 244 3541 2857 Example 54 1 8 5 Example 55 23 635 338 Example 56 15 389 204 Example 57 3 56 39 Example 58 2 37 24 Example 59 2 57 34 Example 60 3 68 39 Example 61 10 178 99 Example 62 20 337 178 Example 63 6 132 72 Example 64 6 149 88 Example 65 2 38 25 Example 66 12 217 127 Example 67 5 145 85 Example 68 12 184 109 Example 69 5 172 90 Example 70 2 36 22 Example 71 1 28 18 Example 72 31 895 498 Example 73 4 59 40 Example 74 2 28 26 Example 75 56 1295 679 Example 76 2 40 23 Example 77 1 19 13 Example 78 17 298 185 Example 79 9 182 113 Example 80 13 389 203 Example 81 7 128 93 Example 82 1 24 16 Example 83 51 842 648 Example 84 74 1464 964 Example 85 58 1111 735 Example 86 12 313 172 Example 87 9 150 93 Example 88 5 139 78 Example 89 1 12 8 Example 90 13 263 190 Example 91 2 35 18 Example 92 34 840 504 Example 93 2 29 24 Example 94 25 485 322 Example 95 12 243 150 Example 96 20 307 196 Example 97 9 127 78 Example 98 1 18 12 Example 99 18 804 280 Example 100 83 1329 1014 Example 101 14 269 163 Example 102 5 123 71 Example 103 4 86 55 Example 104 2 30 17 Example 105 8 129 81 Example 106 15 524 607 Example 107 42 977 507 Example 108 1 33 19 Example 109 1 29 17 Example 110 14 274 168 Example 111 4 120 67 Example 112 4 76 46 Example 113 34 633 434 Example 114 21 346 242 Example 115 6 86 71 Example 116 2 37 25 Example 117 12 238 163 Example 118 17 245 164 Example 119 62 953 714 Example 120 38 585 357 Example 121 4 76 56 Example 122 2 40 24 Example 123 4 72 46 Example 124 13 215 170 Example 125 8 154 106 Example 126 33 661 541 Example 127 30 603 347 Example 128 89 2046 1692 Example 129 23 474 290 Example 130 3 59 44 Example 131 15 387 253 Example 132 77 1371 1249 Example 133 20 384 190 Example 134 2 28 28 Example 135 59 1077 1226 Example 136 11 201 134 Example 137 28 595 363 Example 138 50 697 323 Example 139 2 43 28 Example 140 52 996 685 Example 141 27 475 436 Example 142 5 117 64 Example 143 4 100 57 Example 144 46 759 645 Example 145 2 44 28 Example 146 9 152 67 Example 147 54 982 583 Example 148 62 1037 916 Example 149 5 114 72 Example 150 91 1768 2148 Example 151 122 1641 >4000 Example 152 8 268 132 Example 153 4 74 47 Example 154 10 191 117 Example 155 11 231 141 Example 156 90 885 1006 Example 157 3 84 53 Example 158 5 106 63 Example 159 241 3816 2563 Example 160 15 266 110 Example 161 23 396 169 Example 162 16 290 159 Example 163 N/A* N/A* N/A* Example 164 8 191 121 Example 165 2 47 27 Example 166 13 560 299 Example 167 3 42 33 Example 168 228 3844 >4000 Example 169 46 868 787 Example 170 6 143 89 Example 171 2 33 20 Example 172 95 1632 1072 Example 173 154 2346 1495 Example 174 31 707 429 Example 175 1 20 14 Example 176 33 873 435 Example 177 289 >4000 2911 Example 178 2 52 29 Example 179 46 755 538 Example 180 7 133 78 Example 181 14 260 187 Example 182 10 176 120 Example 183 9 180 125 Example 184 49 991 459 Example 185 67 1545 981 Example 186 6 118 75 Example 187 28 476 321 Example 188 1 23 16 Example 189 183 2973 2414 Example 190 37 623 444 Example 191 0 6 4 Example 192 1 18 11 Example 193 57 939 516 Example 194 4 66 38 Example 195 2 56 34 Example 196 15 354 198 Example 197 11 169 124 Example 198 62 1039 586 Example 199 1 21 15 Example 200 195 3450 3097 Example 201 6 125 74 Example 202 5 94 67 Example 203 3 69 48 Example 204 2 50 29 Example 205 12 331 187 Example 206 27 606 374 Example 207 3 58 39 Example 208 1 32 19 Example 209 33 703 543 Example 210 11 283 162 Example 211 30 618 443 Example 212 84 2366 1111 Example 213 22 529 334 Example 214 2 39 23 Example 215 4 88 57 Example 216 31 743 431 Example 217 39 960 632 Example 218 61 1472 779 Example 219 84 1595 1094 N/A*: Not available. Prodrug of example 75
[0656] Test B: Cell Proliferation/Viability Assay on MCF7 (Breast Tumor Cells) WT and Mutants Cell Lines
[0657] Test B involves measuring the in vitro proliferation activity of a compound of formula (I) by analyzing the viability of the tumor cells.
[0658] The measurements of the viability were made using a breast cancer cell viability assay as described hereunder.
[0659] MCF7 cells expressing (and dependent) on mutants estrogen receptor Tyr 537 Ser or Asp 538 Gly were generated by transfection of MCF7 parental cells (ATCC) with expression vectors coding for different mutants of estrogen receptor Tyr 537 Ser or Asp 538 Gly. The cells were first selected by antibiotic (related to vector expression) and then selected for their growth dependence on estrogen receptor based on their ability to grow in vitro in absence of estradiol (parental cell line die in absence of estradiol).
[0660] MCF7 cells (ATCC) or MCF7 cells expressing (and dependent) on mutants estrogen receptor Tyr 537 Ser or Asp 538 Gly were seeded in 384 wells microplate at concentration of 1000 cells/30 .mu.L per well in red phenol free MEM medium containing 5% charcoal dextran striped FBS. The following day, 9 points serial 1:5 dilution of each compound were added to the cells in 20 .mu.L at final concentrations ranging from 3-0.000001 .mu.M. After 7 days of compound exposure, 50 .mu.L of CellTiter-Glo (Promega) was added to the cells and relative luminescence arbitrary units (RLUs) were determined in luminescence plate reader (Envision device). CellTiter-Glo was added to 50 .mu.L medium without cells to determine the background signal.
[0661] The percent of viability of each sample was determined as follows: (RLU sample -RLU background/RLU untreated -RLU background)*100=% viability.
[0662] The viability activity with respect to estrogen receptors in this test is given by the concentration which inhibits 50% of the viability activity (or IC50) in nM.
[0663] The Table 3 below indicates the cell proliferation/viability assay results on MCF7 (breast tumor cells) WT and mutants cell lines, for compounds of formula (I), and demonstrates that the compounds tested have a significant antiproliferative activity regarding estrogen receptors.
TABLE-US-00003 TABLE 3 proliferation proliferation proliferation MCF7 MCF7 MCF7 (WT) D538G Y537S Examples IC.sub.50 (nM) IC.sub.50 (nM) IC.sub.50 (nM) Example 1 0.6 0.1 3 Example 2 1 0.1 6 Example 3 9 0.2 17 Example 4 5 0.2 11 Example 5 0.3 0.2 2 Example 6 3 0.1 3 Example 7 0.2 0.3 1 Example 8 4 0.1 6 Example 9 0.4 1 5 Example 10 7 0.4 15 Example 11 3 0.2 7 Example 12 28 2 35 Example 13 0.5 0.4 5 Example 14 2 0.1 2 Example 15 0.2 0.4 4 Example 16 3 0.4 7 Example 17 0.2 0.3 2 Example 18 0.7 1 5 Example 19 5 0.4 19 Example 20 5 0.1 14 Example 21 23 2 36 Example 22 6 30 306 Example 23 0.2 1 4 Example 24 63 7 71 Example 25 48 5 67 Example 26 25 4 31 Example 27 0.2 1 8 Example 28 0.2 1 7 Example 29 0.5 1 3 Example 30 36 2 39 Example 31 4 0.3 6 Example 32 0.3 1 3 Example 33 0.3 1 3 Example 34 0.2 0.4 2 Example 35 0.2 1 5 Example 36 0.3 0.4 4 Example 37 0.5 1 7 Example 38 0.6 1 9 Example 39 0.2 1 4 Example 40 0.2 0.4 4 Example 41 0.2 1 4 Example 42 0.4 1 6 Example 43 0.4 1 5 Example 44 0.2 1 3 Example 45 0.7 2 14 Example 46 0.6 1 8 Example 47 0.3 0.3 3 Example 48 0.7 2 13 Example 49 0.4 1 5 Example 50 0.2 0.3 4 Example 51 0.4 1 10 Example 52 0.3 1 5 Example 53 10 21 112 Example 54 0.1 0.2 2 Example 55 2 4 39 Example 56 0.5 2 10 Example 57 0.2 0.3 3 Example 58 0.3 1 4 Example 59 0.1 0.5 3 Example 60 0.4 0.3 5 Example 61 0.3 1 4 Example 62 0.4 1 7 Example 63 0.4 1 5 Example 64 0.3 0.4 4 Example 65 0.1 1 2 Example 66 0.4 1 6 Example 67 0.2 0.4 3 Example 68 0.2 0.3 2 Example 69 0.3 1 5 Example 70 0.2 1 8 Example 71 0.2 0.4 4 Example 72 0.6 1 14 Example 73 0.9 2 15 Example 74 0.1 0.3 2 Example 75 0.5 1 7 Example 76 0.2 0.3 3 Example 77 0.1 0.3 2 Example 78 0.4 0.4 6 Example 79 6 0.6 12 Example 80 0.5 1 11 Example 81 0.2 0.2 4 Example 82 0.8 1 8 Example 83 1 1 14 Example 84 0.7 1 13 Example 85 0.8 1 9 Example 86 0.4 1 5 Example 87 0.1 0.4 2 Example 88 0.2 0.3 4 Example 89 0.2 0.3 3 Example 90 0.4 0.3 6 Example 91 0.1 0.2 1 Example 92 0.5 1 8 Example 93 0.2 1 4 Example 94 0.7 1 7 Example 95 0.5 1 10 Example 96 0.8 1 5 Example 97 0.2 0.1 1 Example 98 0.4 0.3 2 Example 99 0.4 1 3 Example 100 1 2 15 Example 101 0.8 1 6 Example 102 0.1 0.3 1 Example 103 0.5 1 5 Example 104 0.1 0.1 1 Example 105 0.3 0.3 4 Example 106 1 3 54 Example 107 0.5 1 12 Example 108 0.2 0.1 1 Example 109 0.2 0.1 2 Example 110 0.4 1 9 Example 111 0.2 0.2 3 Example 112 0.2 0.2 4 Example 113 0.6 2 11 Example 114 0.4 1 5 Example 115 0.3 1 5 Example 116 0.1 1 2 Example 117 0.5 1 6 Example 118 0.2 0.2 2 Example 119 0.7 2 8 Example 120 0.3 1 6 Example 121 0.3 1 2 Example 122 0.2 1 2 Example 123 0.2 1 5 Example 124 0.3 1 4 Example 125 0.2 1 3 Example 126 1 3 30 Example 127 0.1 0.4 1 Example 128 8 17 96 Example 129 0.1 0.3 5 Example 130 0.2 1 5 Example 131 0.4 1 11 Example 132 0.7 2 19 Example 133 1 2 29 Example 134 0.1 0.3 3 Example 135 1 3 21 Example 136 0.2 1 5 Example 137 0.2 1 7 Example 138 0.8 3 19 Example 139 0.1 1 3 Example 140 1 3 24 Example 141 0.3 1 5 Example 142 0.2 0.2 2 Example 143 0.7 1 5 Example 144 2 2 25 Example 145 0.3 0.4 3 Example 146 0.4 1 4 Example 147 2 2 20 Example 148 2 2 11 Example 149 0.7 1 6 Example 150 3 2 18 Example 151 0.9 1 16 Example 152 0.4 0.3 4 Example 153 0.1 0.1 1 Example 154 0.4 0.3 4 Example 155 0.1 0.2 2 Example 156 2 4 11 Example 157 0.6 0.2 2 Example 158 0.5 1 5 Example 159 8 11 112 Example 160 0.7 1 6 Example 161 0.8 1 7 Example 162 0.6 1 3 Example 163 N/A* N/A* N/A* Example 164 2 0.2 2 Example 165 0.4 0.3 4 Example 166 0.8 1 12 Example 167 0.4 1 3 Example 168 4 5 33 Example 169 10 1 1 Example 170 3 0.3 0.3 Example 171 0.9 0.1 0.1 Example 172 30 2 4 Example 173 5 11 177 Example 174 1 3 40 Example 175 0.2 1 10 Example 176 5 12 108 Example 177 11 18 187 Example 178 0.5 2 15 Example 179 1 3 35 Example 180 2 3 39 Example 181 2 4 52 Example 182 0.3 1 15 Example 183 1 1 9 Example 184 1 2 36 Example 185 41 77 1000 Example 186 0.9 2 26 Example 187 19 34 1000 Example 188 0.1 0.1 3 Example 189 12 27 246 Example 190 7 15 148 Example 191 0.5 2 19 Example 192 0.6 2 17 Example 193 0.6 1 20 Example 194 0.3 1 8 Example 195 0.2 0.2 4 Example 196 1 5 43 Example 197 0.2 1 6 Example 198 3 11 79 Example 199 0.2 1 2 Example 200 78 108 1000 Example 201 0.02 1 4 Example 202 0.3 1 6 Example 203 0.1 1 4 Example 204 0.1 0.3 1 Example 205 8 28 119 Example 206 1 8 26 Example 207 0.3 1 3 Example 208 0.1 0.3 1 Example 209 8 25 68 Example 210 0.5 2 8 Example 211 0.7 2 10 Example 212 4 14 112 Example 213 1 2 8 Example 214 0.1 1 2 Example 215 1 18 51 Example 216 1 6 19 Example 217 0.3 12 23 Example 218 1 13 29 Example 219 1 9 36 N/A*: Not available. Prodrug of example 75
[0664] Test C: Estrogen Receptor Depredation Activity
[0665] Test C involves measuring the in vitro degradation activity of a compound of formula (I).
[0666] The measurements of the degradation activities were made using a breast cancer cell ER.alpha. in cell western assay as described hereunder.
[0667] MCF7 cells (ATCC) were seeded in 384 wells microplate (collagen coated) at concentration of 10000 cells/30 .mu.L per well in red phenol free MEM alpha medium (Invitrogen) containing 5% charcoal dextran striped FBS. The following day, 9 points serial 1:5 dilution of each compound were added to the cells in 2.5 .mu.L at final concentrations ranging from 3-0.000018 .mu.M or 0.1 .mu.M for fulvestrant (using as positive control). At 4 hours post compounds addition the cells were fixed by adding 25 .mu.L of formalin (final concentration 5% formalin containing 0.1% triton) for 10 minutes at room temperature and then washed twice with PBS. Then, 50 .mu.L of LI-COR blocking buffer containing 0.1% Triton was added to plate for 30 minutes at room temperature. LI-COR blocking buffer was removed and cells were incubated overnight at cold room with 50 .mu.L anti-ER rabbit monoclonal antibody (Thermo scientific MA1-39540) diluted at 1:1000 in LI-COR blocking buffer containing 0.1% tween-20. Wells which were treated with blocking but no antibody were used as background control. Wells were washed twice with PBS (0.1% tween-20) and incubated at 37.degree. C. for 60 minutes in LI-COR (0.1% tween-20) containing goat anti-rabbit antibody Alexa 488 (1:1000) and Syto-64 a DNA dye (2 .mu.M final concentration). Cells were then washed 3 times in PBS and scanned in ACUMEN explorer (TTP-Labtech). Integrated intensities in the green fluorescence and red fluorescence were measured to determine the levels of ER.alpha. and DNA respectively.
[0668] The degradation activity with respect to estrogen receptors in this test is given by the concentration which degrades 50% of the estrogen receptor (or IC50) in nM.
[0669] The % of ER.alpha. levels decrease were determined as follows: % inhibition=100*(1-(sample -fulvestrant: DMSO -fulvestrant)).
[0670] The Table 4 below indicates the estrogen receptor degradation activity results for compounds of formula (I), and demonstrates that compounds tested have a significant degradation activity on estrogen receptors.
TABLE-US-00004 TABLE 4 Degradation % Degradation Examples IC.sub.50 (nM) At 3 .mu.M Example 1 0.4 88 Example 2 0.4 97 Example 3 3 82 Example 4 0.3 90 Example 5 0.3 93 Example 6 0.7 90 Example 7 0.5 97 Example 8 0.5 96 Example 9 0.7 95 Example 10 0.2 92 Example 11 0.7 89 Example 12 0.5 82 Example 13 0.5 91 Example 14 0.3 94 Example 15 0.2 95 Example 16 2 90 Example 17 0.8 83 Example 18 0.9 90 Example 19 1 82 Example 20 0.2 87 Example 21 0.2 83 Example 22 2 81 Example 23 2 82 Example 24 28 86 Example 25 38 91 Example 26 11 92 Example 27 2 91 Example 28 1 92 Example 29 0.4 88 Example 30 2 82 Example 31 1 91 Example 32 2 87 Example 33 0.6 86 Example 34 1 83 Example 35 0.8 89 Example 36 0.4 90 Example 37 1 95 Example 38 1 96 Example 39 0.5 91 Example 40 0.2 88 Example 41 0.3 85 Example 42 0.3 84 Example 43 0.2 83 Example 44 0.2 80 Example 45 0.2 93 Example 46 0.2 94 Example 47 0.4 90 Example 48 0.2 96 Example 49 0.2 94 Example 50 0.4 92 Example 51 0.2 98 Example 52 0.4 92 Example 53 8 95 Example 54 0.2 92 Example 55 5 87 Example 56 2 85 Example 57 0.5 86 Example 58 0.7 84 Example 59 0.4 86 Example 60 1 84 Example 61 1 86 Example 62 0.7 96 Example 63 0.3 95 Example 64 0.4 88 Example 65 0.8 89 Example 66 0.6 90 Example 67 2 92 Example 68 1 92 Example 69 0.2 87 Example 70 0.7 80 Example 71 0.6 83 Example 72 2 88 Example 73 0.4 86 Example 74 0.4 84 Example 75 2 84 Example 76 0.3 88 Example 77 0.3 83 Example 78 0.9 92 Example 79 0.9 84 Example 80 1 90 Example 81 0.6 83 Example 82 0.5 83 Example 83 2 80 Example 84 2 92 Example 85 1 92 Example 86 0.7 89 Example 87 0.4 90 Example 88 0.5 86 Example 89 0.6 84 Example 90 1 83 Example 91 0.2 86 Example 92 2 89 Example 93 0.3 86 Example 94 2 88 Example 95 1 84 Example 96 1 86 Example 97 0.7 95 Example 98 1 86 Example 99 0.9 94 Example 100 3 93 Example 101 1 87 Example 102 0.6 87 Example 103 1 86 Example 104 0.6 84 Example 105 0.3 93 Example 106 2 87 Example 107 2 89 Example 108 0.2 87 Example 109 0.5 80 Example 110 0.8 83 Example 111 0.3 84 Example 112 0.6 88 Example 113 0.2 90 Example 114 0.2 88 Example 115 0.2 89 Example 116 0.2 85 Example 117 0.2 85 Example 118 0.2 87 Example 119 0.2 81 Example 120 0.2 85 Example 121 0.2 87 Example 122 0.2 85 Example 123 0.2 83 Example 124 0.2 87 Example 125 0.2 93 Example 126 0.2 86 Example 127 0.2 89 Example 128 1 82 Example 129 0.2 90 Example 130 0.2 80 Example 131 0.2 84 Example 132 0.3 89 Example 133 0.2 88 Example 134 0.2 87 Example 135 0.2 85 Example 136 0.2 82 Example 137 0.2 85 Example 138 0.3 88 Example 139 0.2 80 Example 140 0.2 86 Example 141 0.2 84 Example 142 0.2 84 Example 143 0.2 81 Example 144 2 86 Example 145 0.2 83 Example 146 0.2 83 Example 147 0.2 88 Example 148 0.2 83 Example 149 0.2 82 Example 150 1 82 Example 151 1 81 Example 152 0.2 80 Example 153 0.2 91 Example 154 0.2 87 Example 155 0.2 89 Example 156 0.2 85 Example 157 0.2 81 Example 158 0.2 82 Example 159 0.7 84 Example 160 0.2 83 Example 161 0.2 81 Example 162 0.2 82 Example 163 N/A* N/A* Example 164 0.2 88 Example 165 0.2 82 Example 166 0.5 86 Example 167 0.2 81 Example 168 0.6 91 Example 169 1 87 Example 170 0.5 82 Example 171 0.3 83 Example 172 2 80 Example 173 9 85 Example 174 0.7 92 Example 175 0.2 88 Example 176 2 81 Example 177 8 92 Example 178 0.2 94 Example 179 0.9 94 Example 180 3 91 Example 181 3 94 Example 182 0.2 93 Example 183 0.7 98 Example 184 2 95 Example 185 9 80 Example 186 0.9 87 Example 187 15 90 Example 188 0.2 91 Example 189 22 90 Example 190 7 80 Example 191 0.5 89 Example 192 0.3 88 Example 193 0.2 88 Example 194 0.8 90 Example 195 0.2 87 Example 196 8 86 Example 197 0.6 92 Example 198 3 81 Example 199 0.2 95 Example 200 15 85 Example 201 0.3 90 Example 202 0.2 84 Example 203 0.2 81 Example 204 0.2 82 Example 205 4 88 Example 206 1 85 Example 207 0.2 86 Example 208 0.2 94 Example 209 8 91 Example 210 1 91 Example 211 0.3 95 Example 212 2 94 Example 213 0.2 96 Example 214 0.5 92 Example 215 1.5 80 Example 216 3 90 Example 217 2 90 Example 218 3 92 Example 219 1 89 N/A*: Not available. Prodrug of example 75
[0671] It is therefore apparent that the compounds of formula (I), or a pharmaceutically acceptable salt thereof, have antagonist and degradation activities for estrogen receptors, as well as antiproliferative activity. The compounds of formula (I), or a pharmaceutically acceptable salt thereof, can therefore be used for preparing medicaments, especially medicaments which are antagonists and degraders of estrogen receptors.
[0672] Accordingly, also provided are medicaments which comprise a compound of the formula (I), or a pharmaceutically acceptable salt thereof.
[0673] Another aspect is the compounds of formula (I) defined above, or a pharmaceutically acceptable salt thereof, for use in therapy, especially as inhibitors and degraders of estrogen receptors.
[0674] Another aspect is the compounds of formula (I) defined above, or a pharmaceutically acceptable salt thereof, for use in the treatment of ovulatory dysfunction, cancer, endometriosis, osteoporosis, benign prostatic hypertrophy or inflammation.
[0675] A particular aspect are the compounds of formula (I) defined above, or a pharmaceutically acceptable salt thereof, for use in the treatment of cancer.
[0676] In an embodiment, the cancer is a hormone dependent cancer.
[0677] In another embodiment, the cancer is an estrogen receptor dependent cancer, particularly the cancer is an estrogen receptor a dependent cancer.
[0678] In another embodiment, the cancer is a cancer with wild type estrogen receptors.
[0679] In another embodiment, the cancer is a cancer with deregulated function of estrogen receptors related to, but not limited to, at least one epigenetic and genetic alteration of estrogen receptors such us mutation, amplification, splice variant.
[0680] In another embodiment, the cancer is a cancer with mutated estrogen receptors. In another embodiment, the mutations of estrogen receptors can include, but not limited to, new or known mutations such us Leu536Arg, Tyr537Ser, Tyr537Asn, Asp538Gly.
[0681] In another embodiment, the cancer is an estrogen-sensitive cancer.
[0682] In another embodiment, the cancer is selected from breast, ovarian, endometrial, prostate, uterine, cervical and lung cancer, or a metastasis thereof.
[0683] In another embodiment, the metastasis is a cerebral metastasis.
[0684] In another embodiment, the cancer is breast cancer. Particularly, the breast cancer is an estrogen receptor positive breast cancer (ER.alpha. positive breast cancer).
[0685] In another embodiment, the cancer is resistant to anti-hormonal treatment. In a further embodiment, the anti-hormonal treatment is as single agent or in combination with other agents such as CDK4/6 or PI3K inhibitors.
[0686] In a further embodiment, the anti-hormonal treatment includes treatment with at least one agent selected from tamoxifen, fulvestrant, a steroidal aromatase inhibitor, and a non-steroidal aromatase inhibitor.
[0687] Another embodiment is a method of treating the pathological conditions indicated above, comprising administering to a subject in need thereof a therapeutically effective amount of a compound of formula (I), or a pharmaceutically acceptable salt thereof. In an embodiment of this method of treatment, the subject is a human.
[0688] Another aspect is the use of a compound of the formula (I), or a pharmaceutically acceptable salt thereof, for the manufacture of a medicament useful in treating any of the pathological conditions indicated above, more particularly the use in treating cancer.
[0689] Another aspect is a pharmaceutical composition comprising as active principle a compound of formula (I) or a pharmaceutically acceptable salt thereof. These pharmaceutical compositions comprise an effective dose of at least one compound of formula (I), or a pharmaceutically acceptable salt thereof, and also at least one pharmaceutically acceptable excipient.
[0690] The said excipients are selected, in accordance with the pharmaceutical form and method of administration desired, from the customary excipients, which are known to a person skilled in the art.
[0691] In the pharmaceutical compositions provided herein for oral, sublingual, subcutaneous, intramuscular, intravenous, topical, local, intra-tracheal, intranasal, transdermal or rectal administration, the active principle of formula (I) above, or its base, acid, zwitterion or salt thereof, may be administered in a unit administration form, in a mixture with conventional pharmaceutical excipients, to animals and to human beings for the treatment of the above disorders or diseases.
[0692] The unit administration forms appropriate include oral forms such as tablets, soft or hard gel capsules, powders, granules and oral solutions or suspensions, sublingual, buccal, intra-tracheal, intra-ocular and intra-nasal administration forms, forms for inhalative, topical, transdermal, subcutaneous, intra-muscular or intravenous administration, rectal administration forms and implants. For topical application it is possible to use the compounds of formula (I), or a pharmaceutically acceptable salt thereof, in creams, gels, ointments or lotions.
[0693] As an example, a unit administration form of a compound of formula (I) in tablet form may comprise the following components:
TABLE-US-00005 Compound of formula (I) 50.0 mg Mannitol 223.75 mg Sodium croscarmellose 6.0 mg Corn starch 15.0 mg Hydroxypropylmethylcellulose 2.25 mg Magnesium stearate 3.0 mg
[0694] There may be particular cases in which higher or lower dosages are appropriate. According to usual practice, the dosage that is appropriate for each patient is determined by the doctor according to the mode of administration and the weight and response of the said patient.
* * * * *












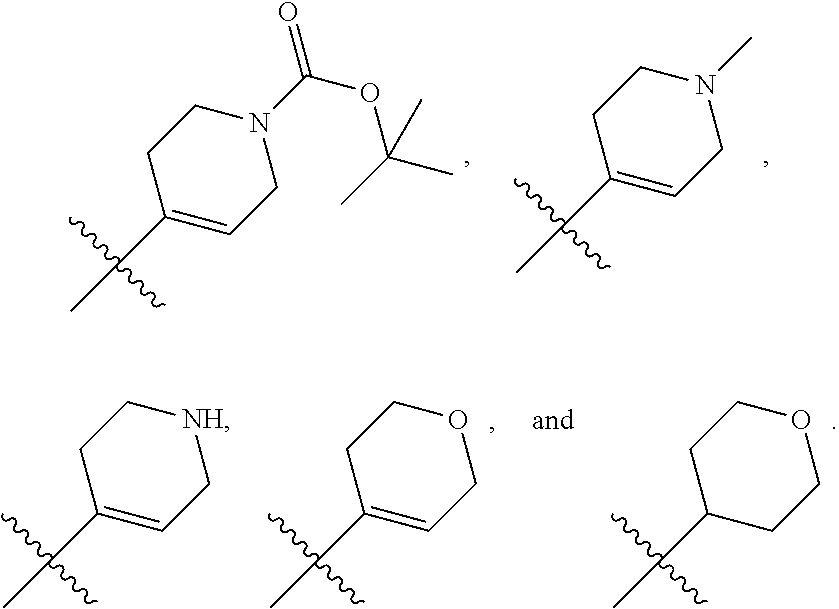








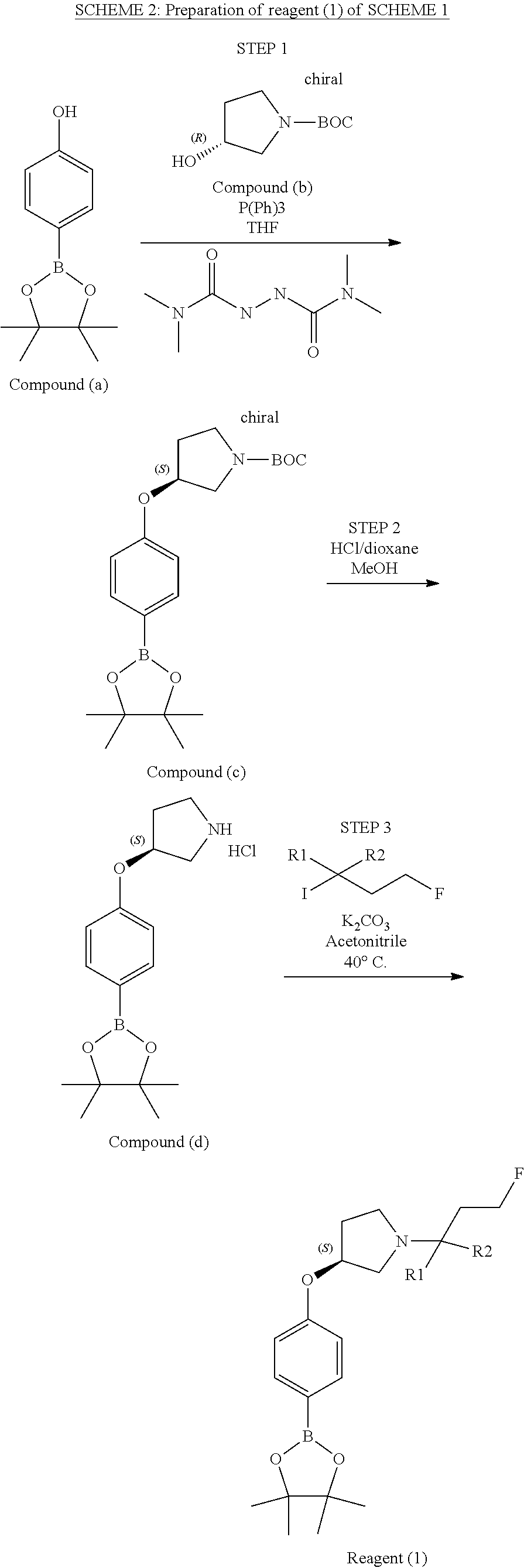
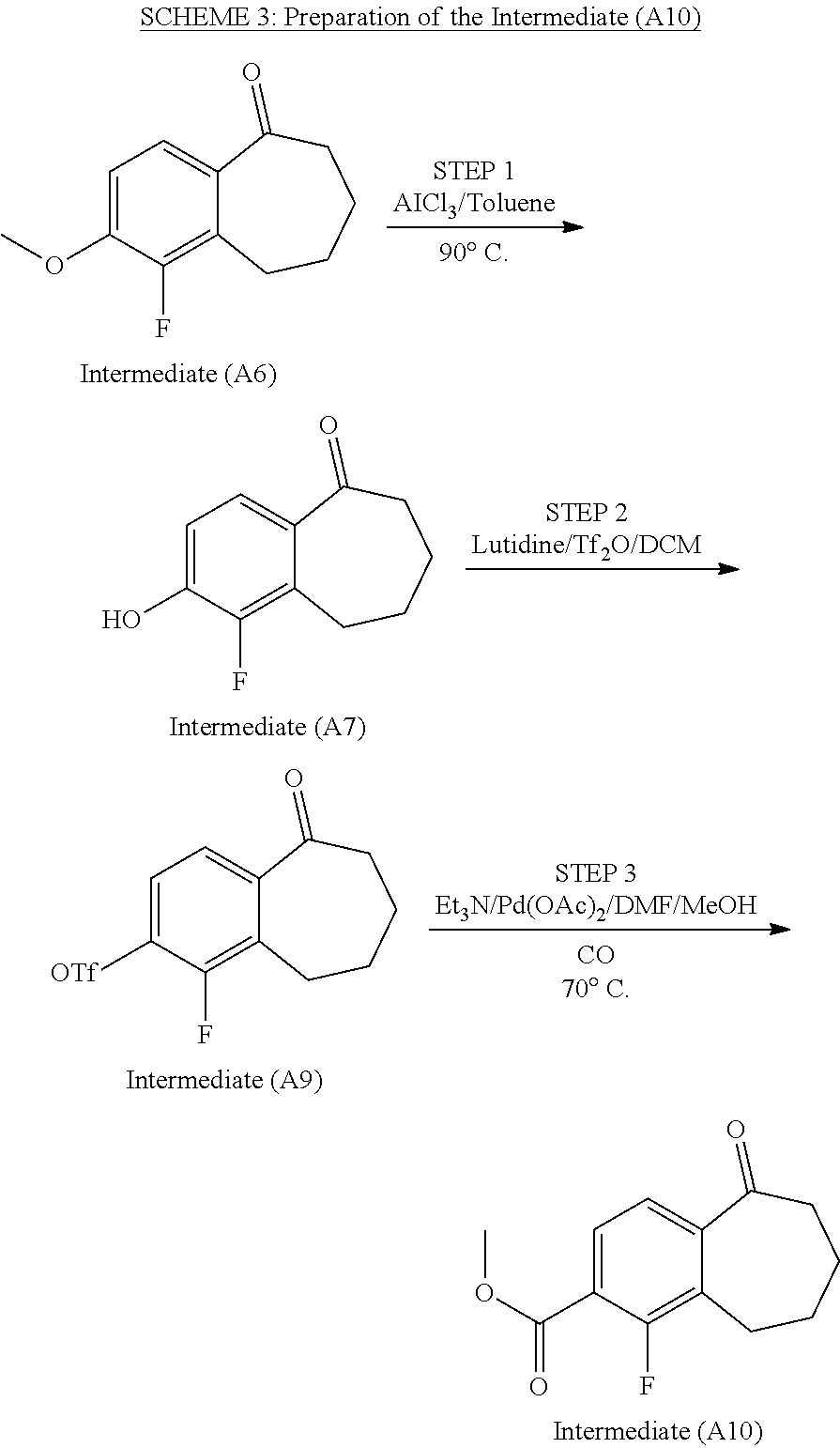







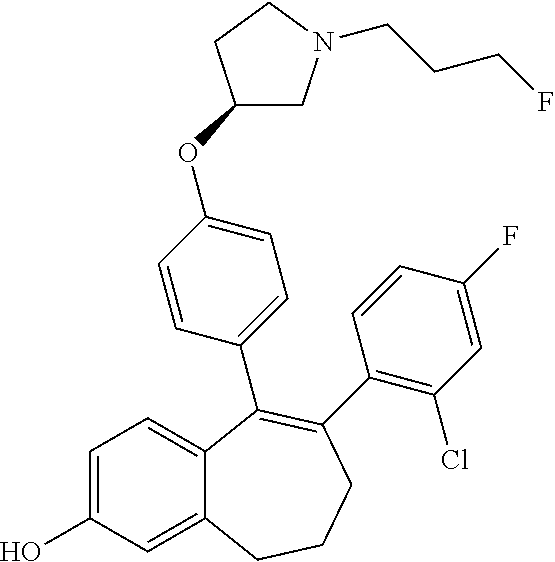




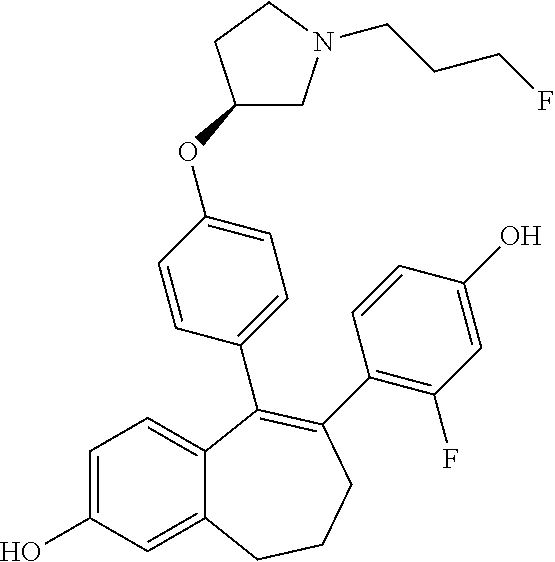

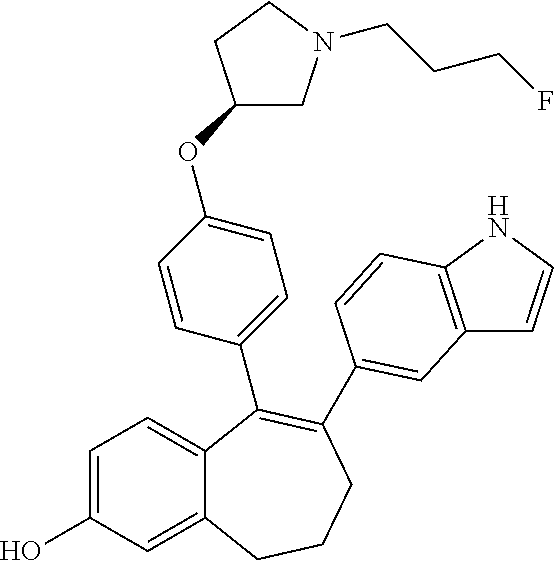

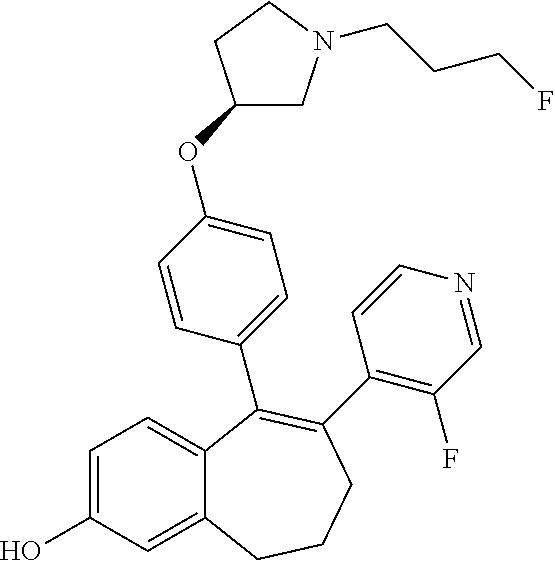
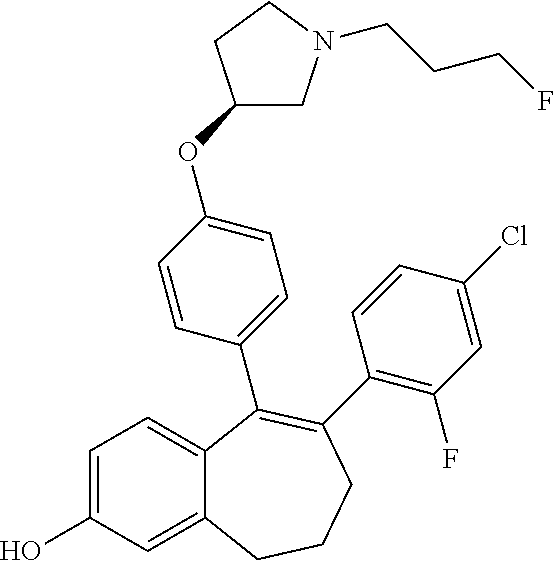
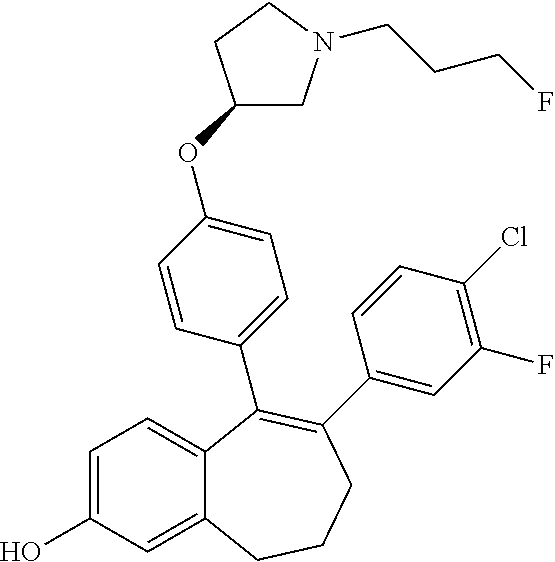


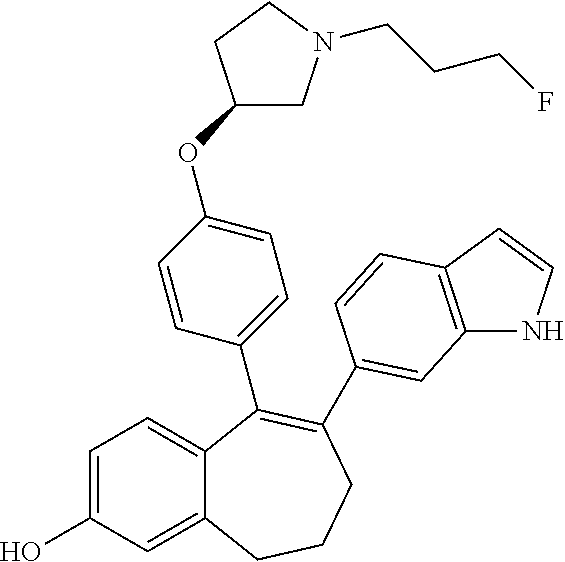
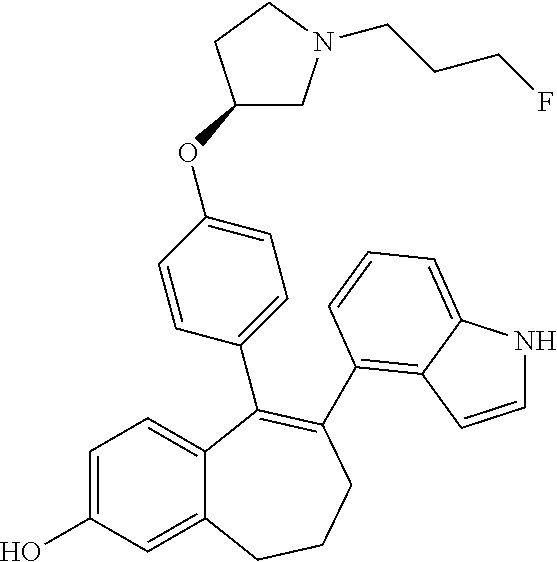

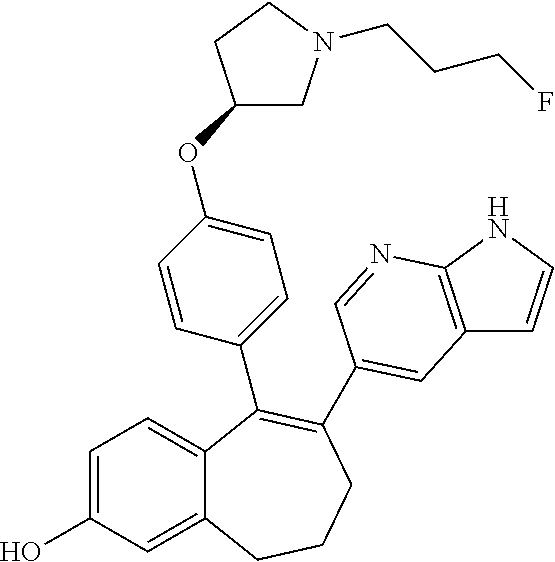


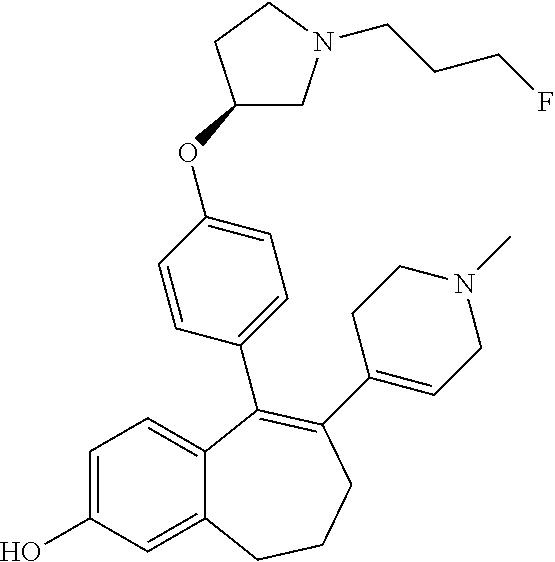

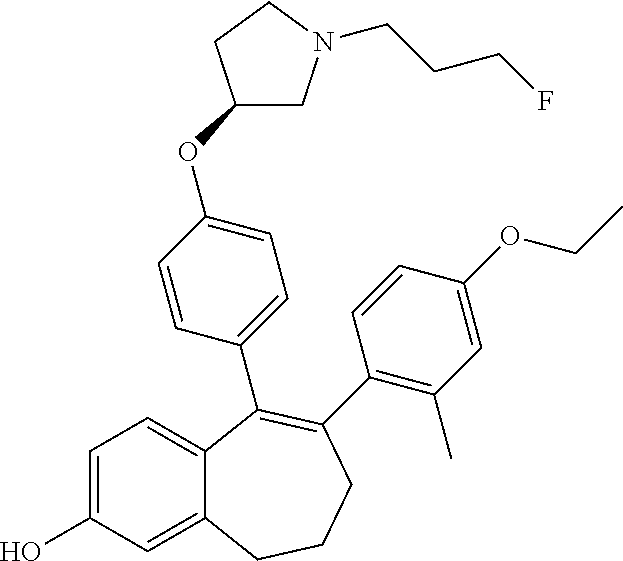

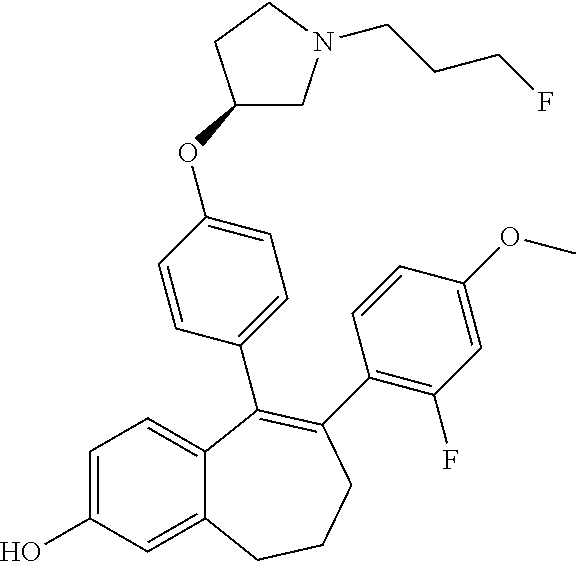


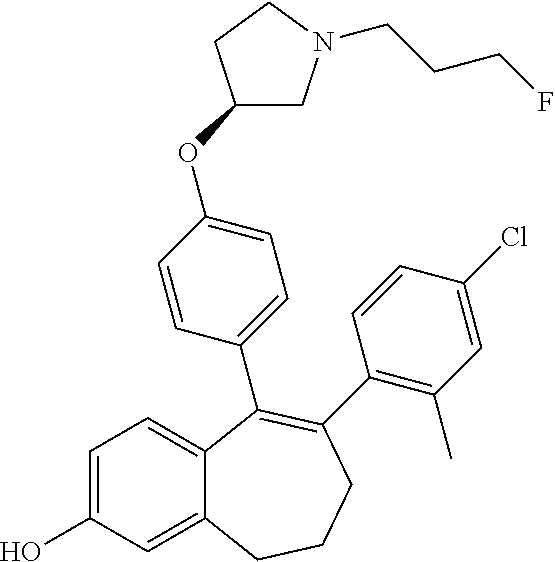





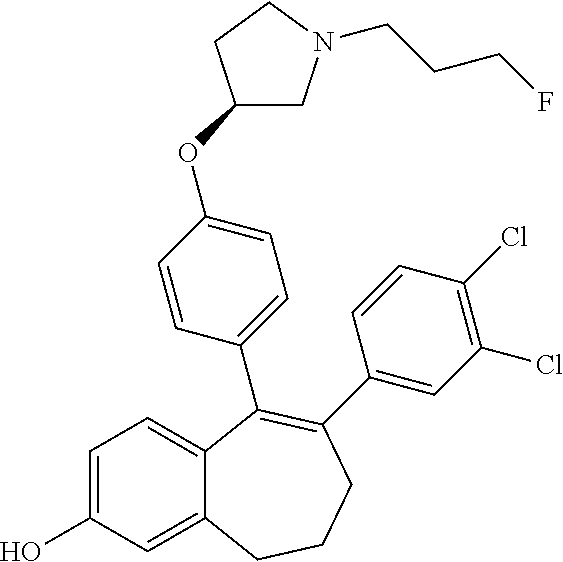




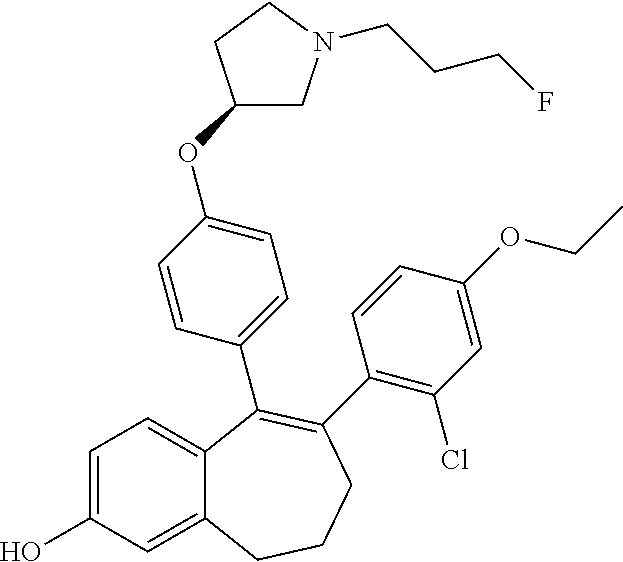

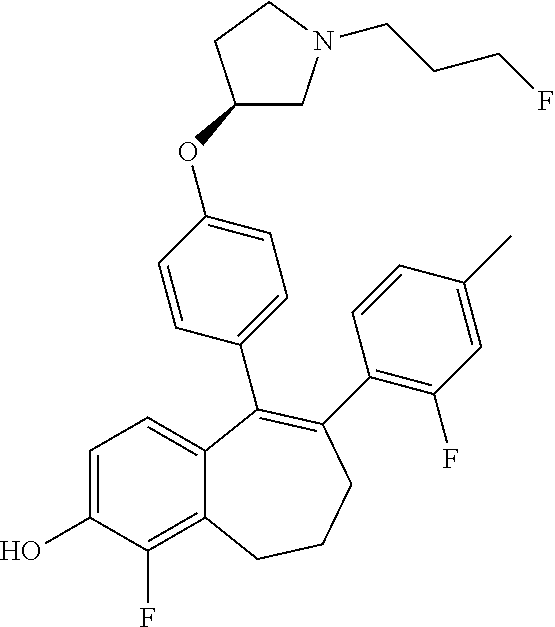

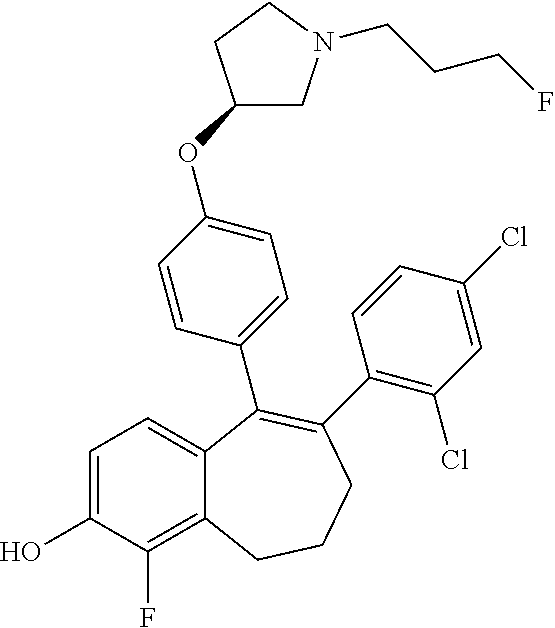

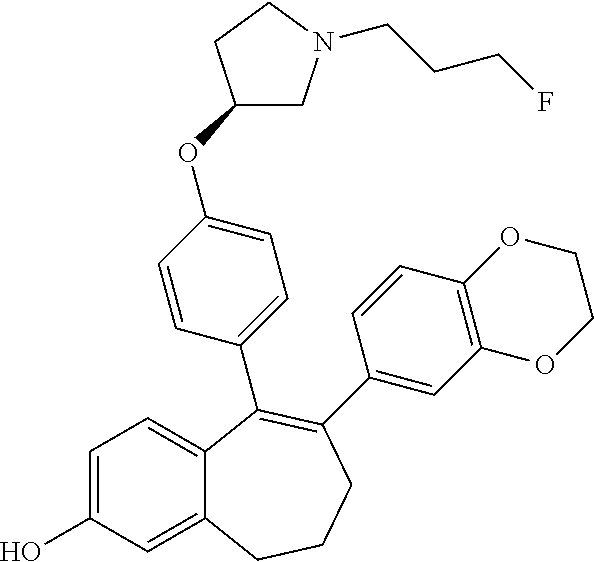


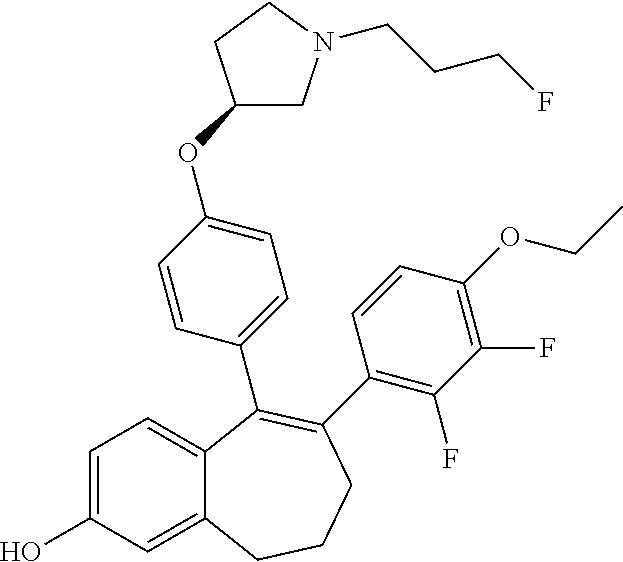
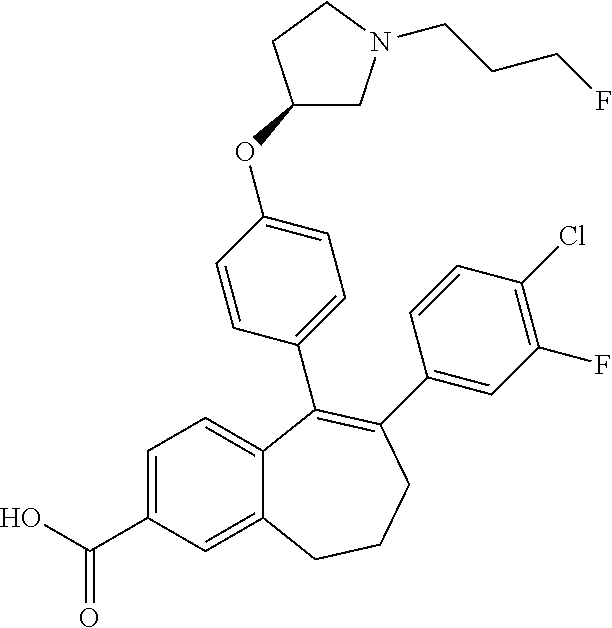



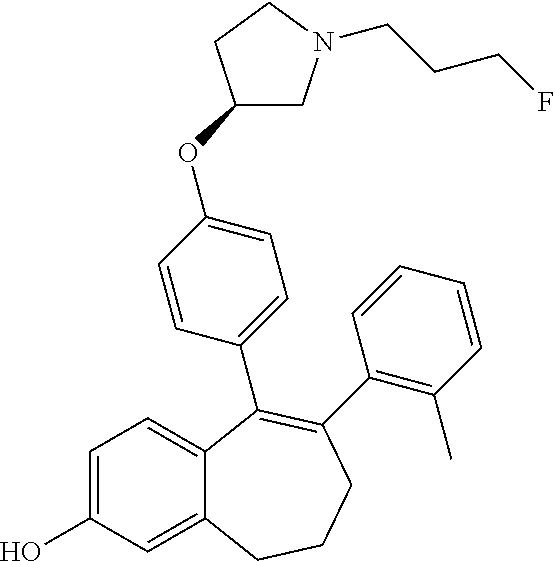


















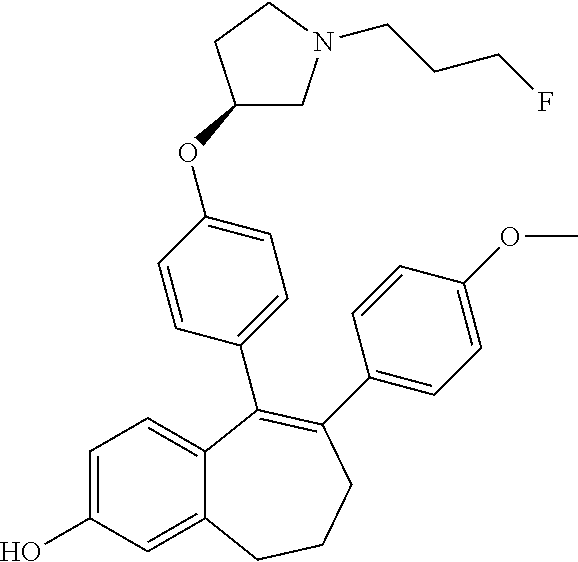


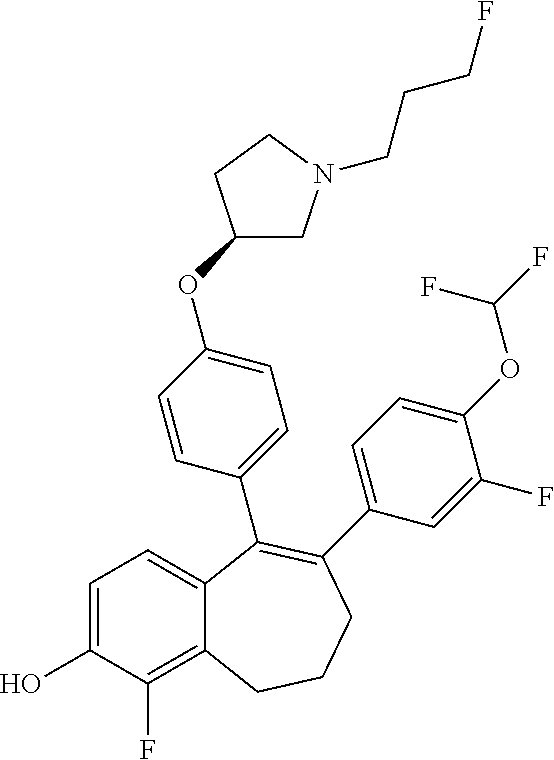

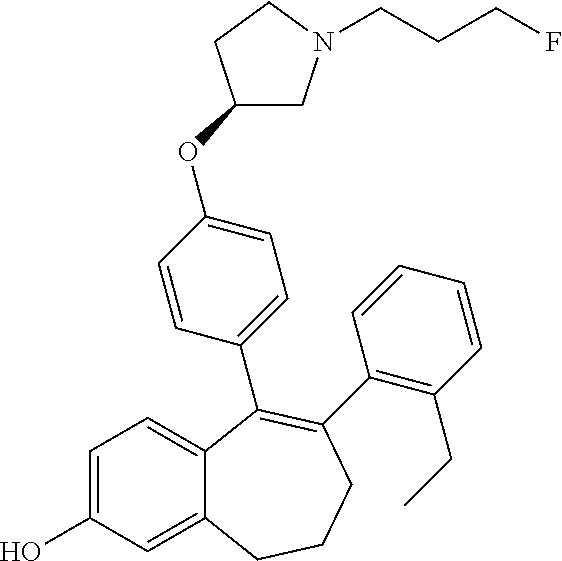







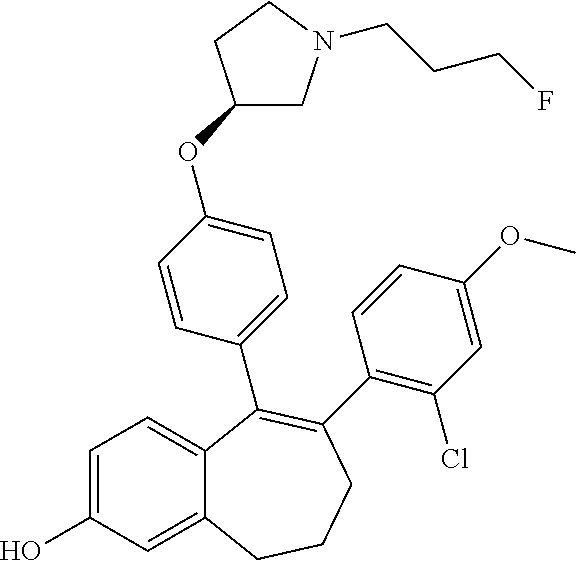

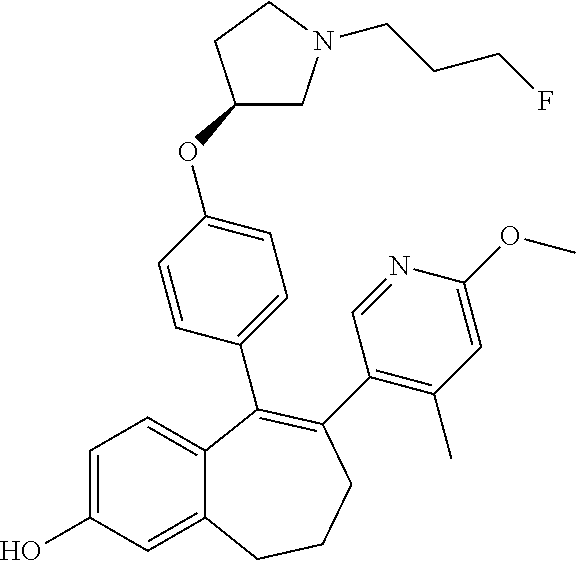

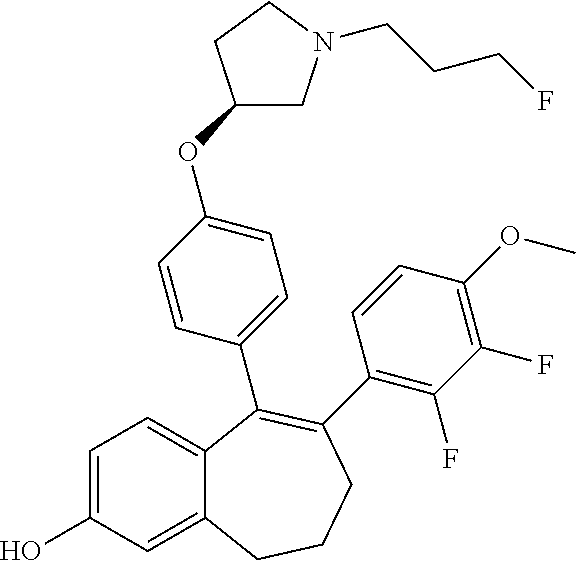







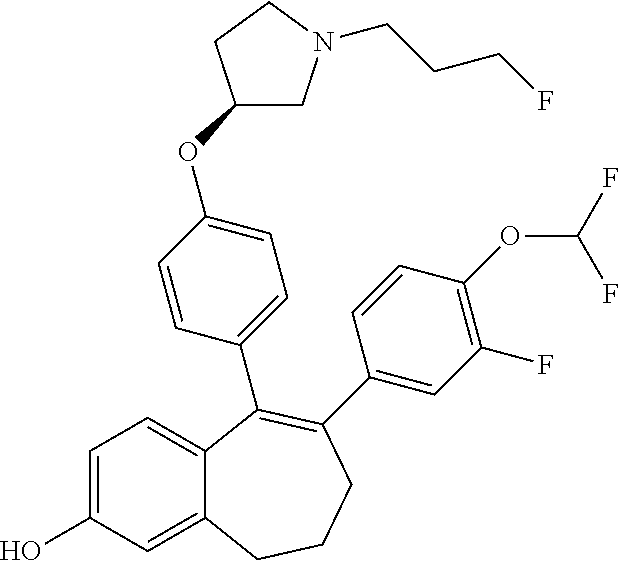
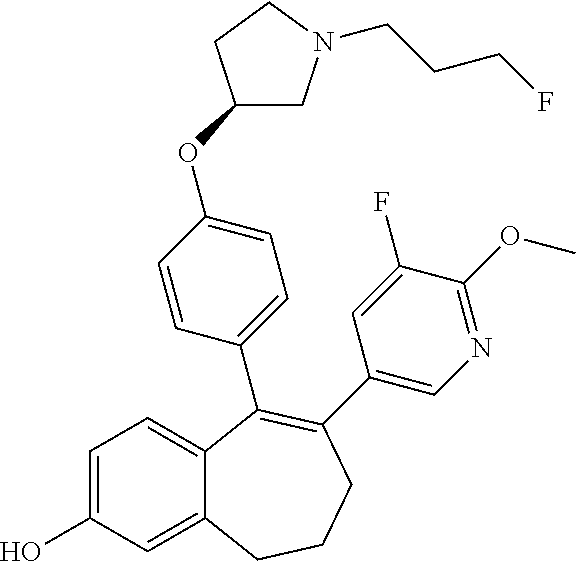






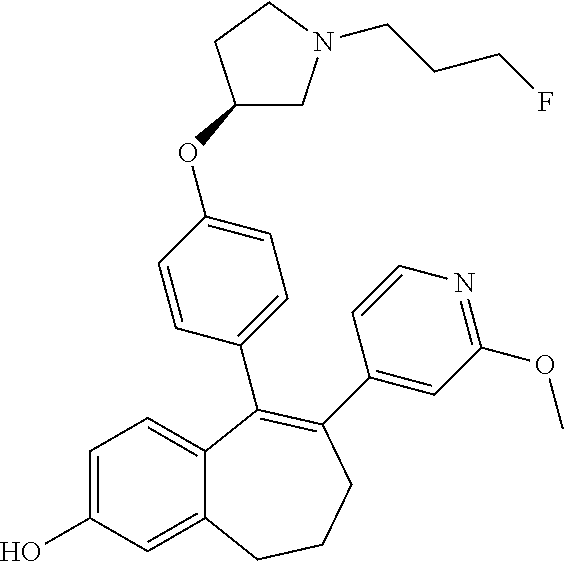




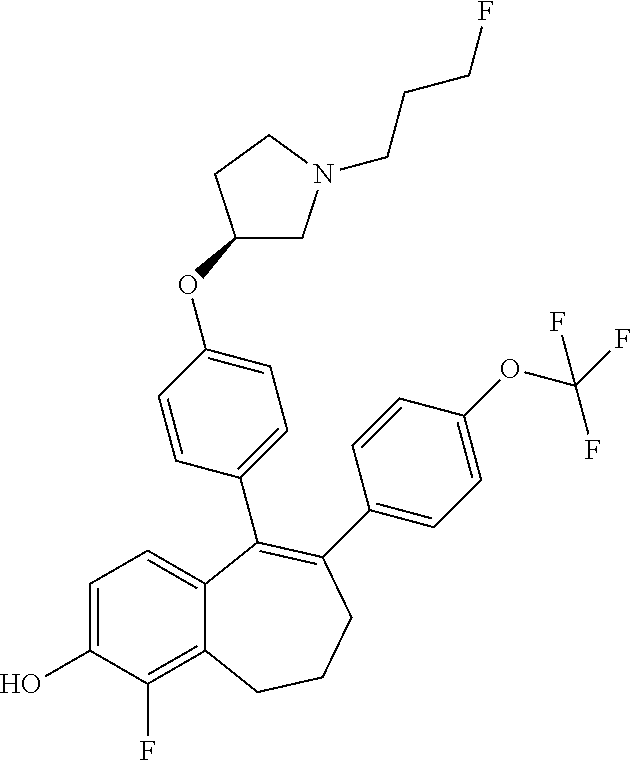




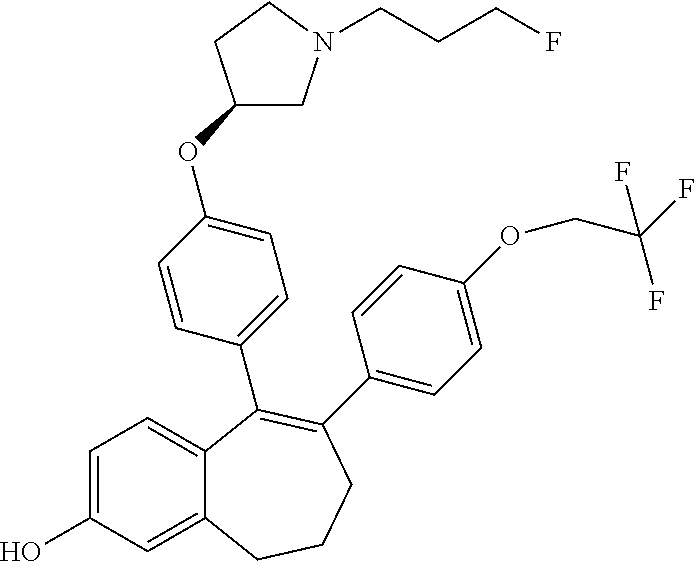



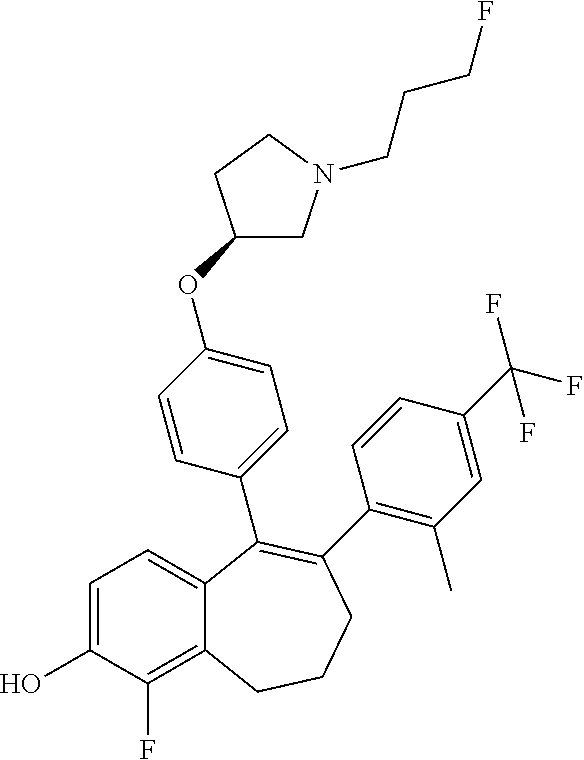

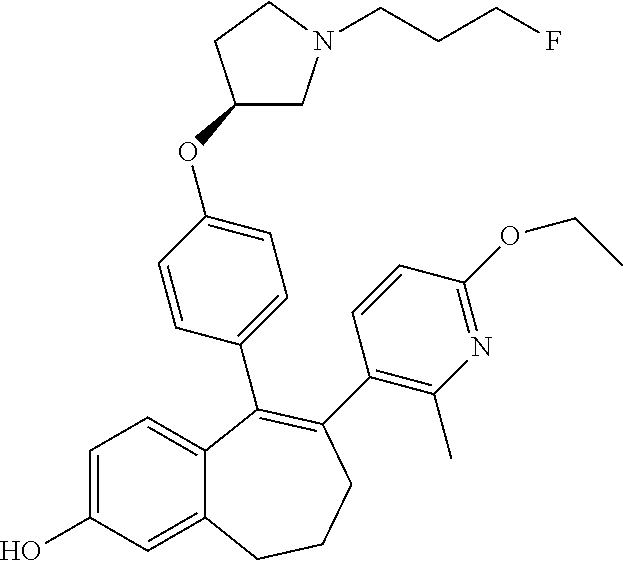


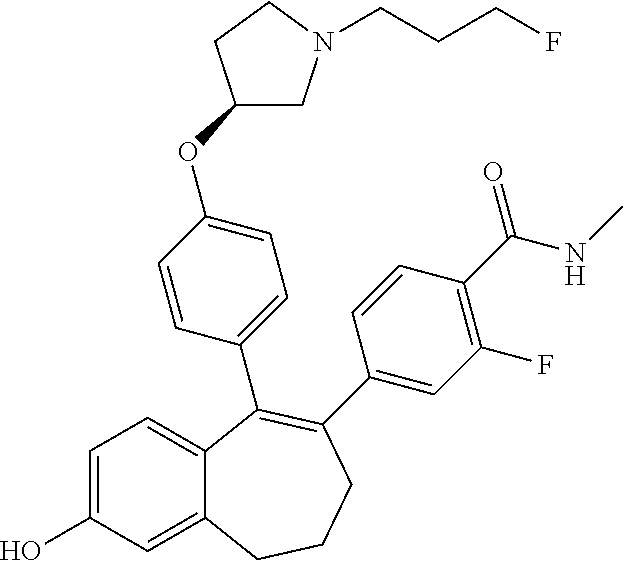


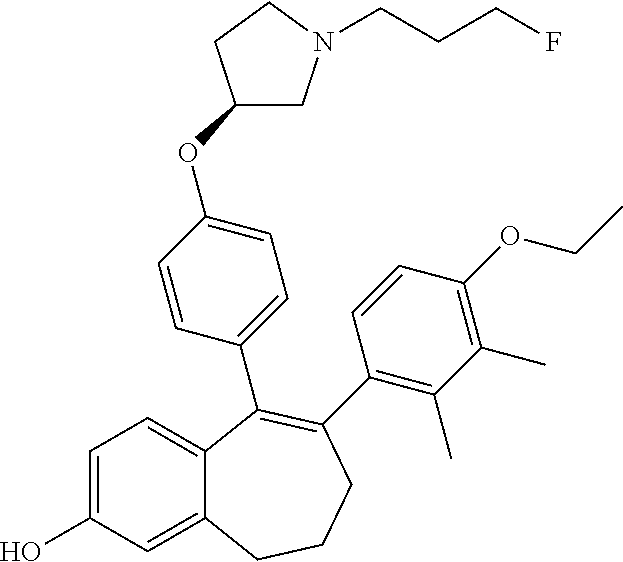
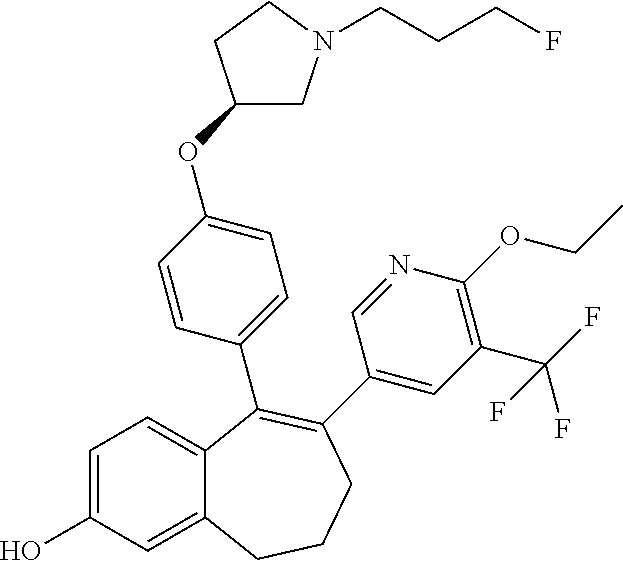
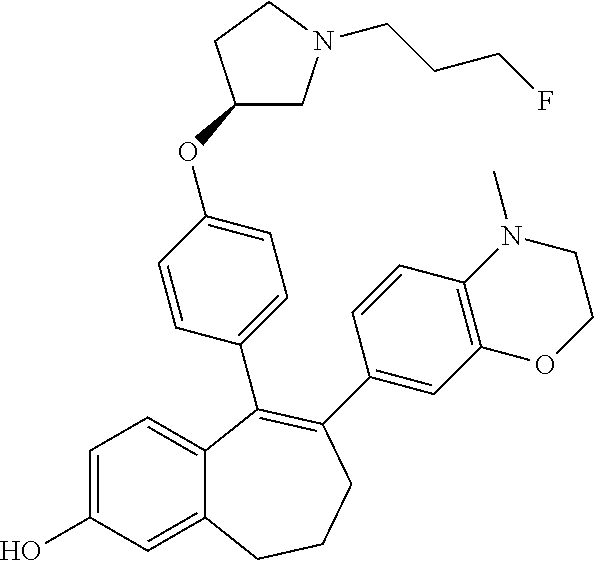







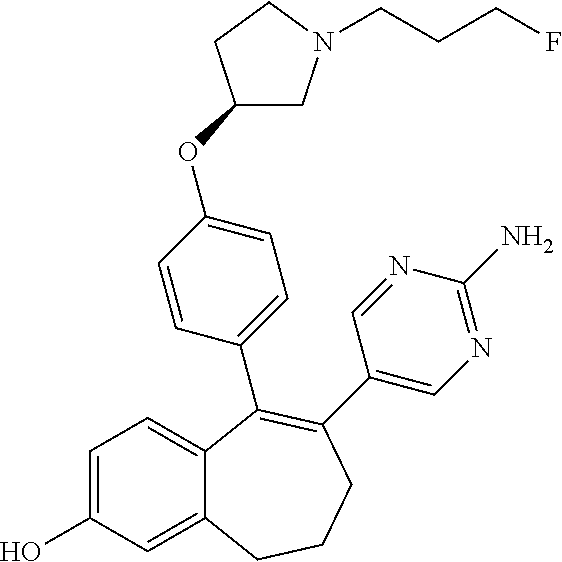






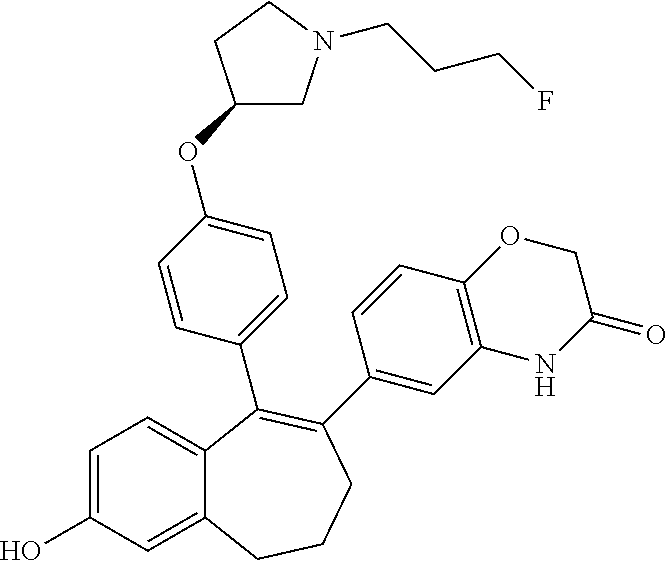

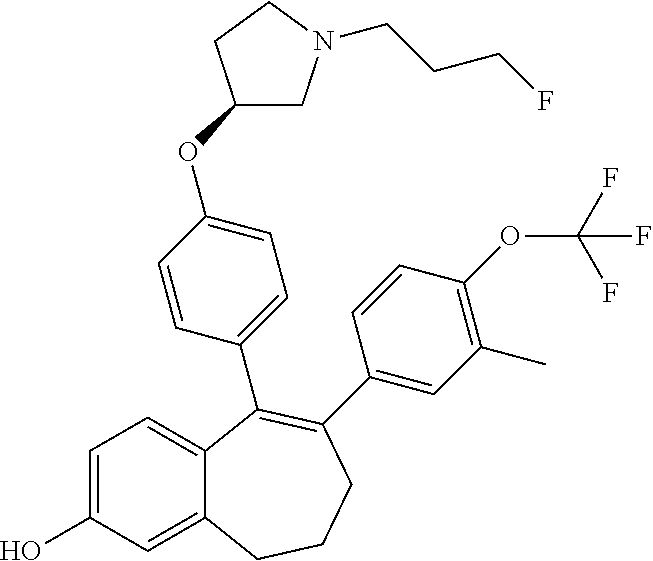

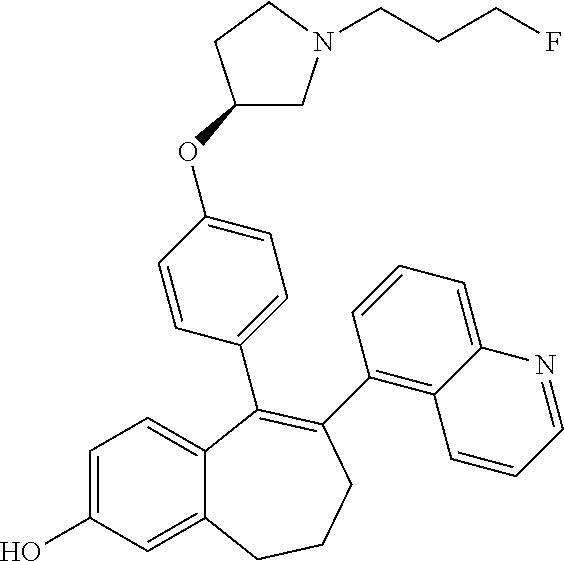
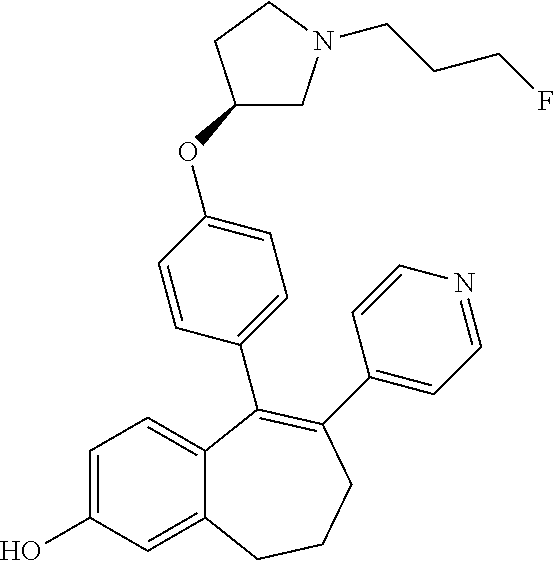


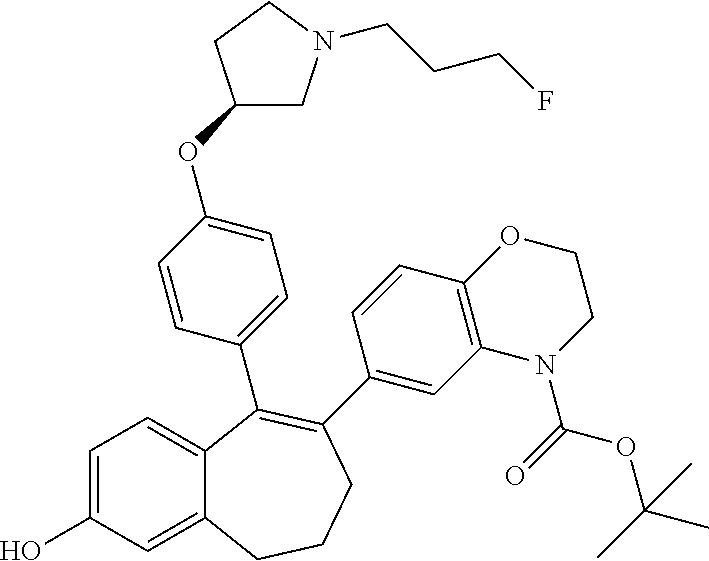
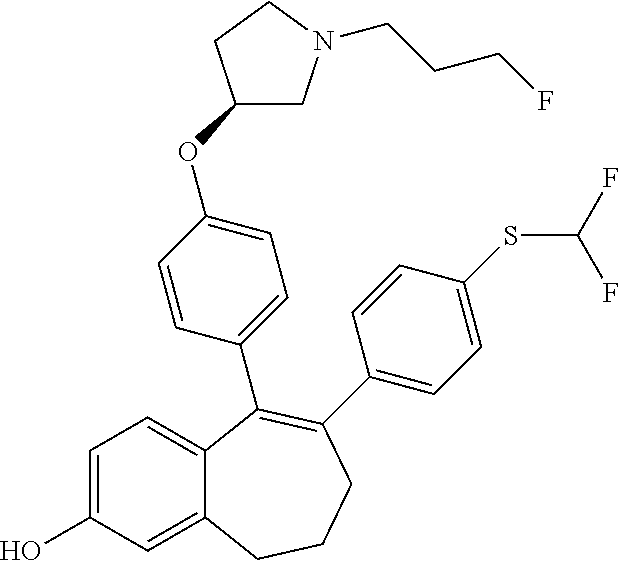






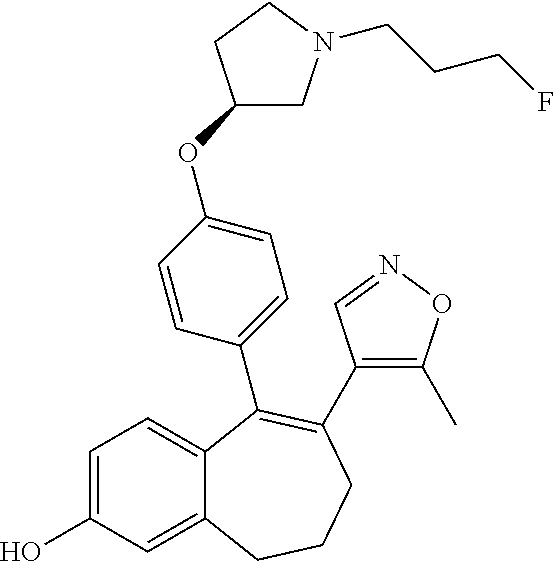
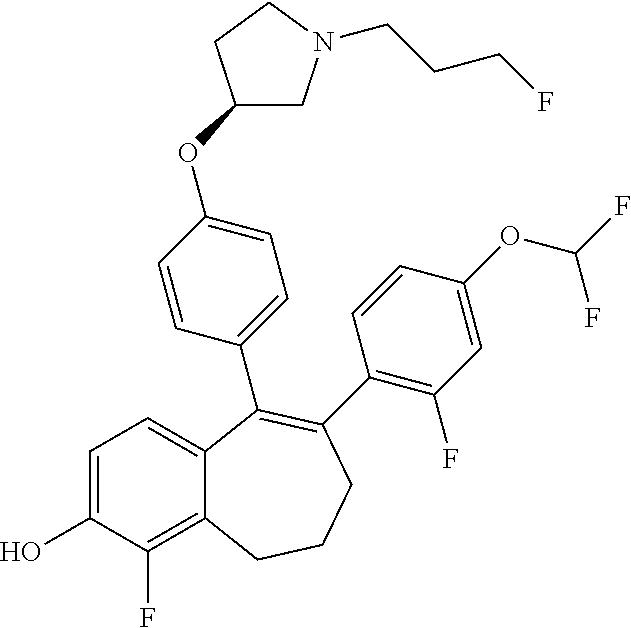
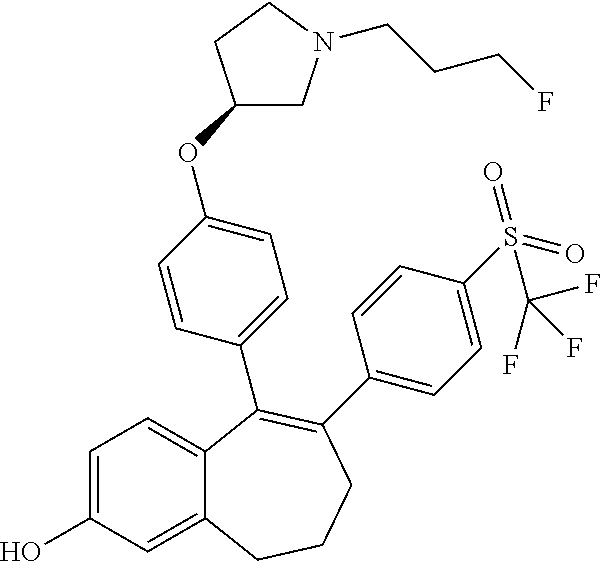
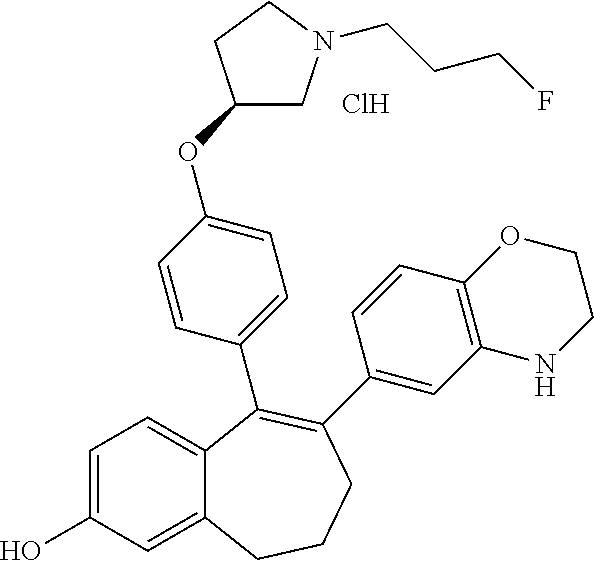
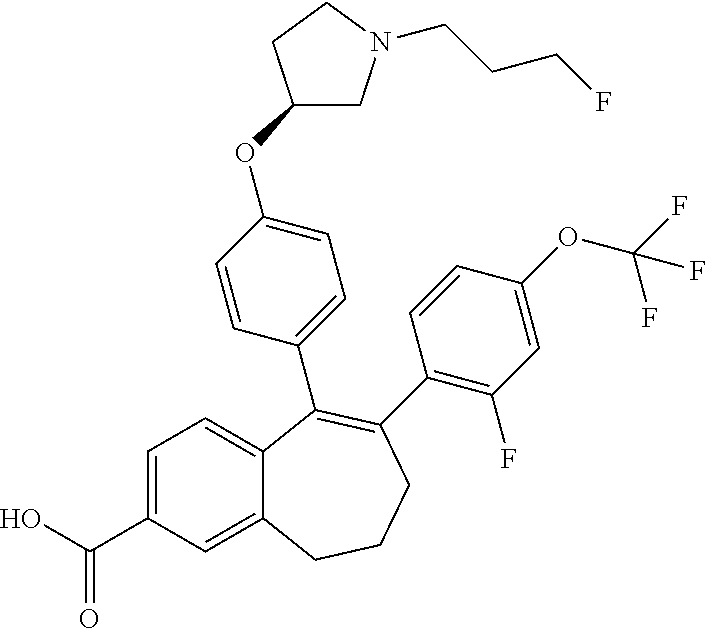











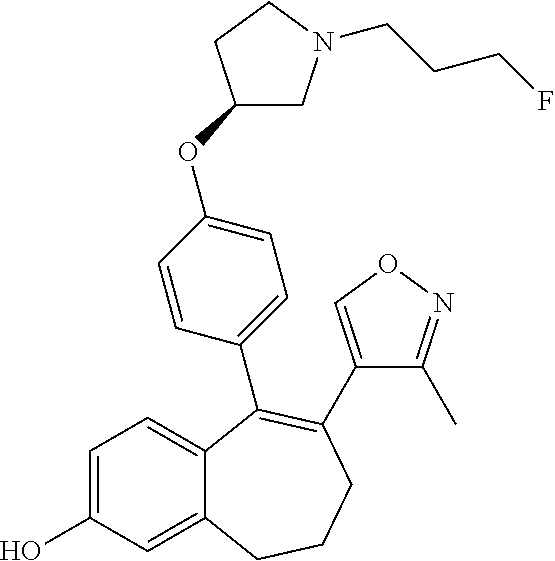
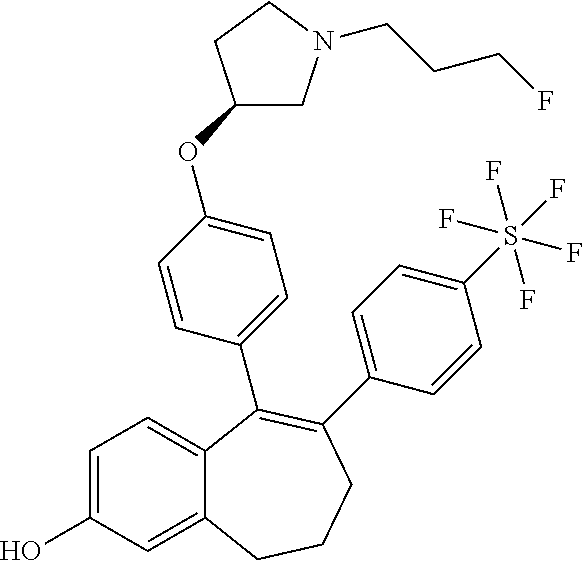


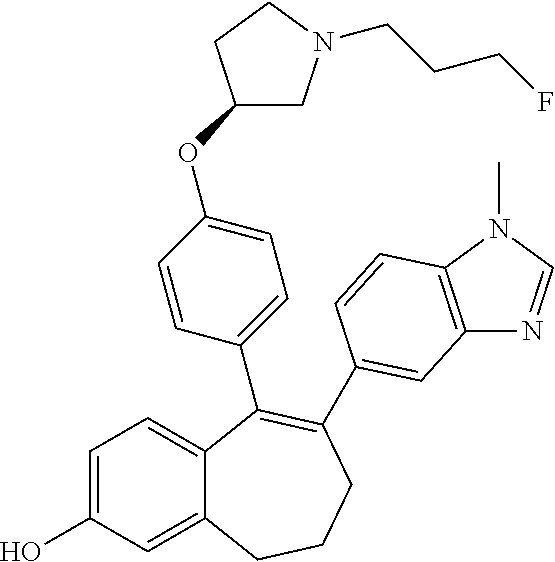












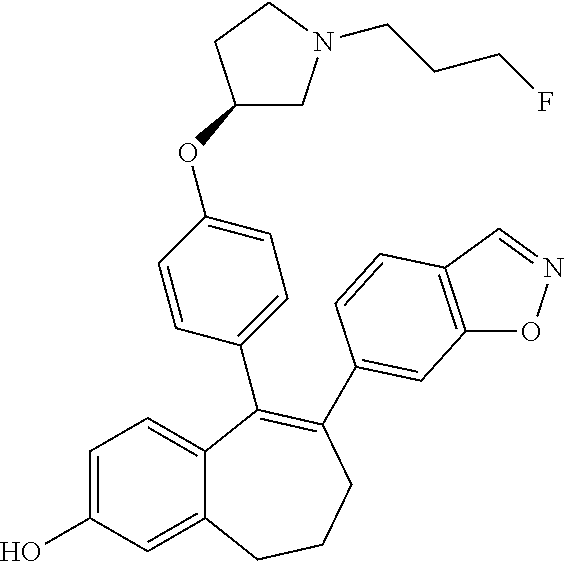
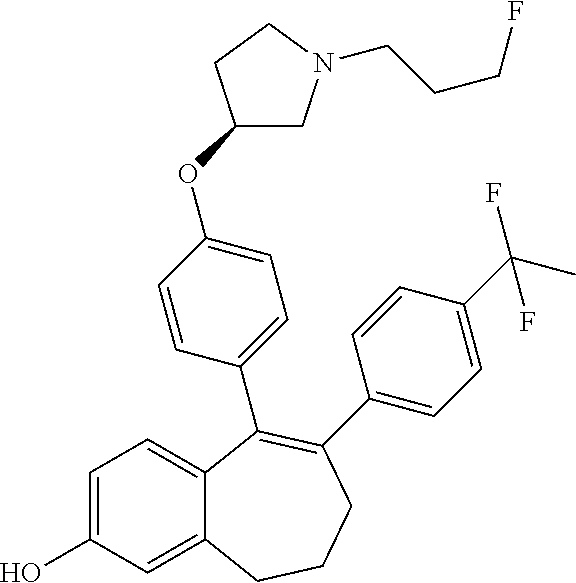
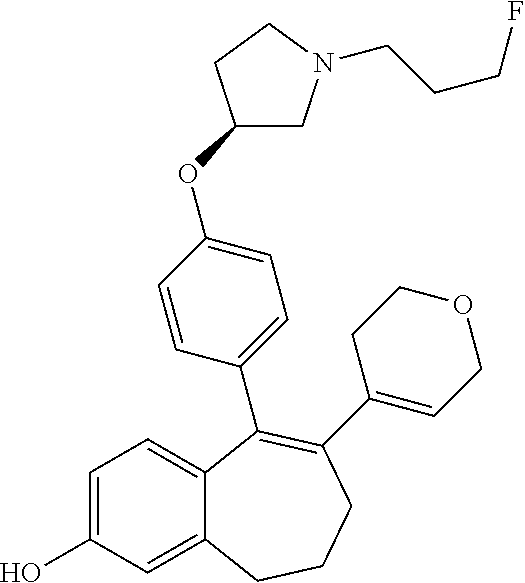


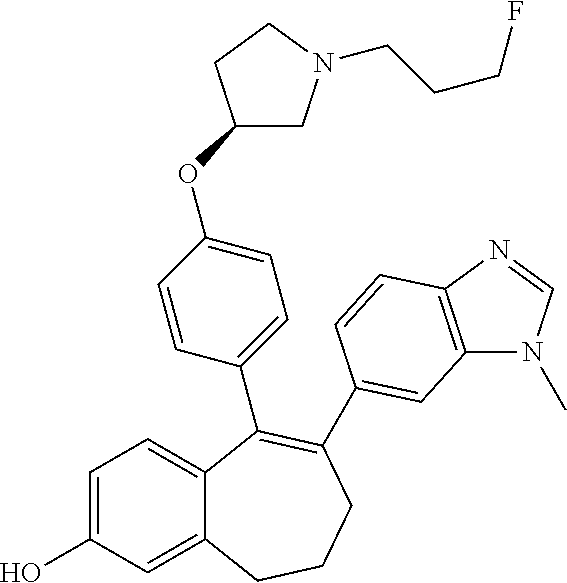
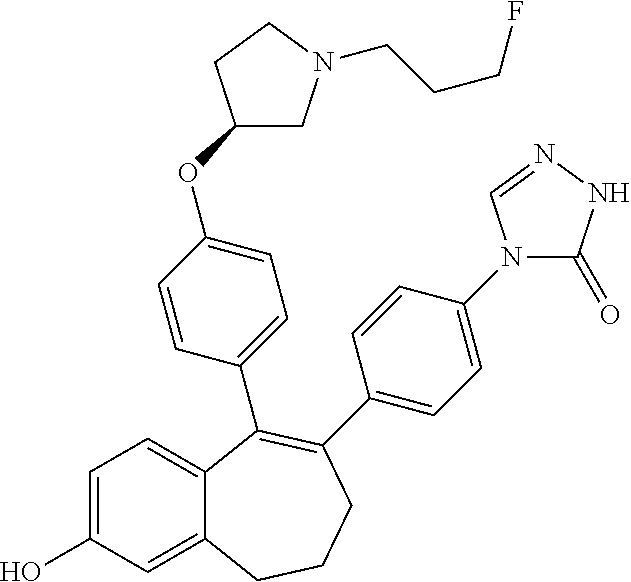



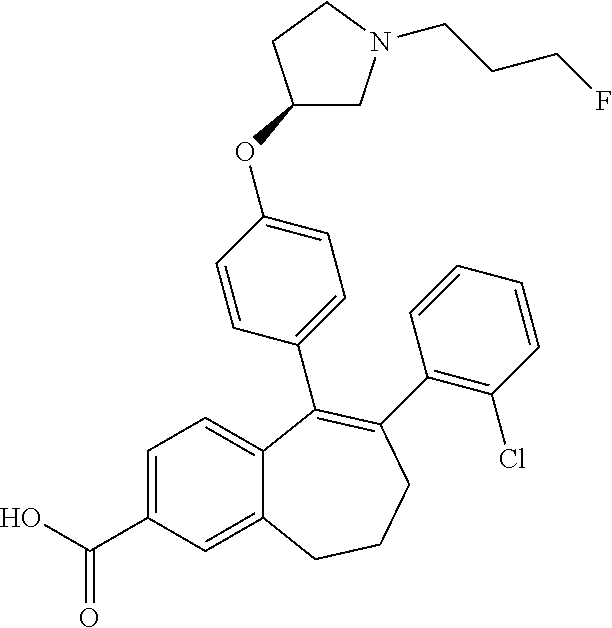







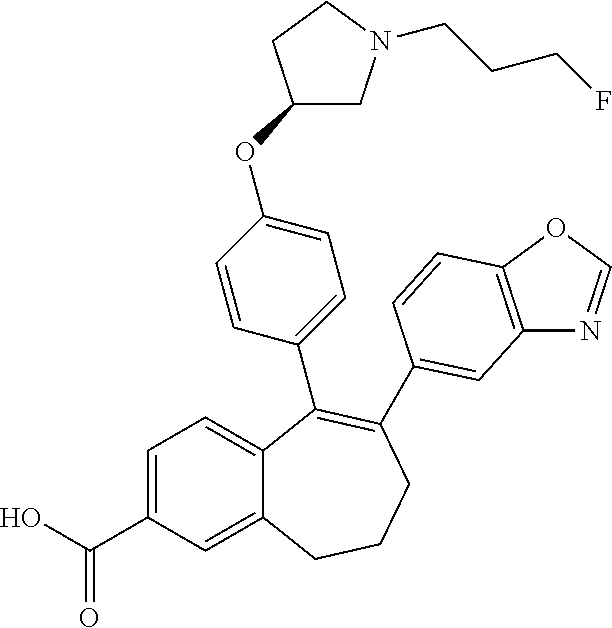


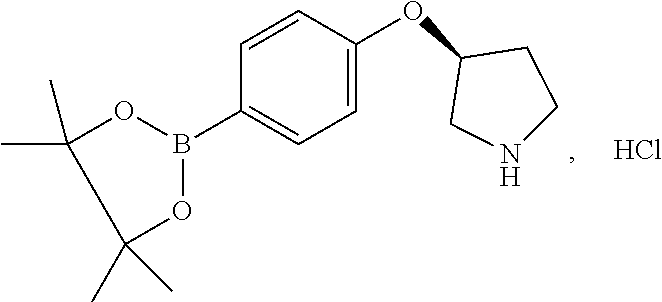

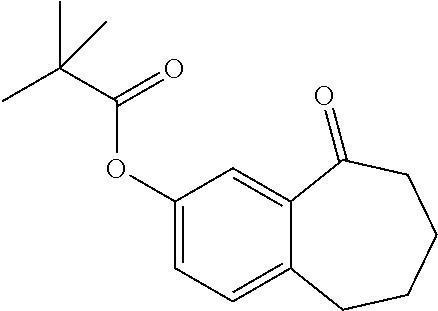
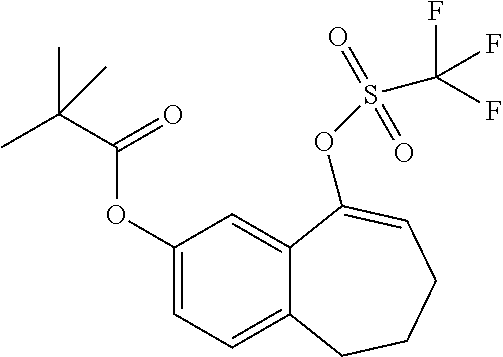
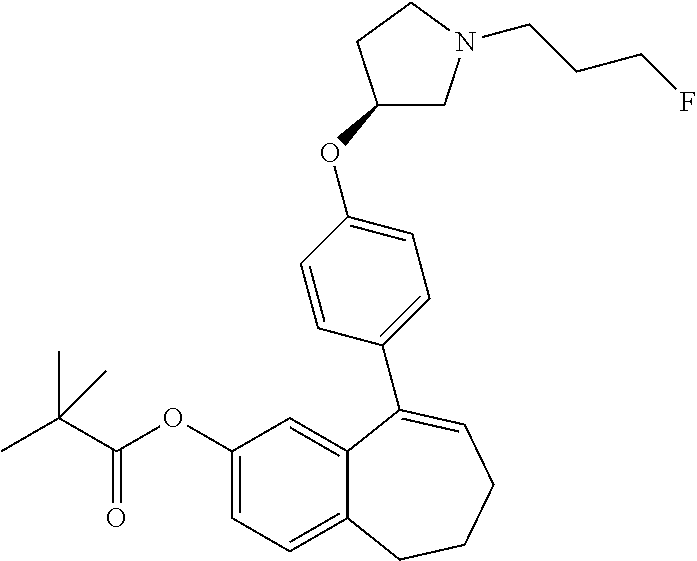






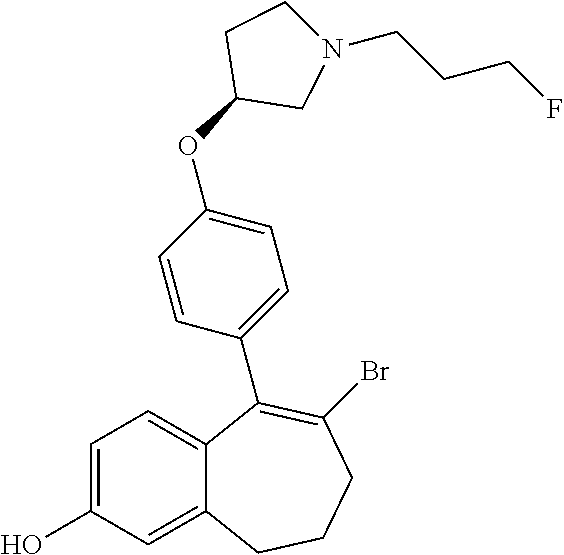






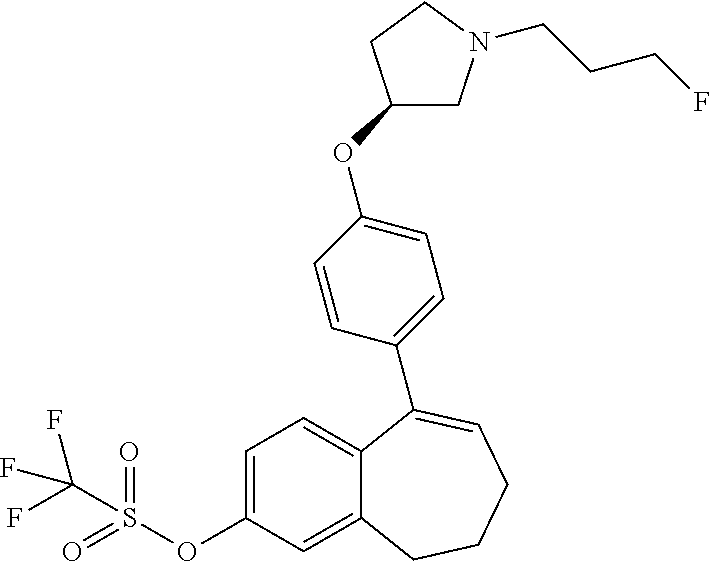

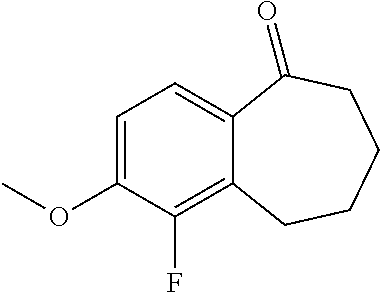








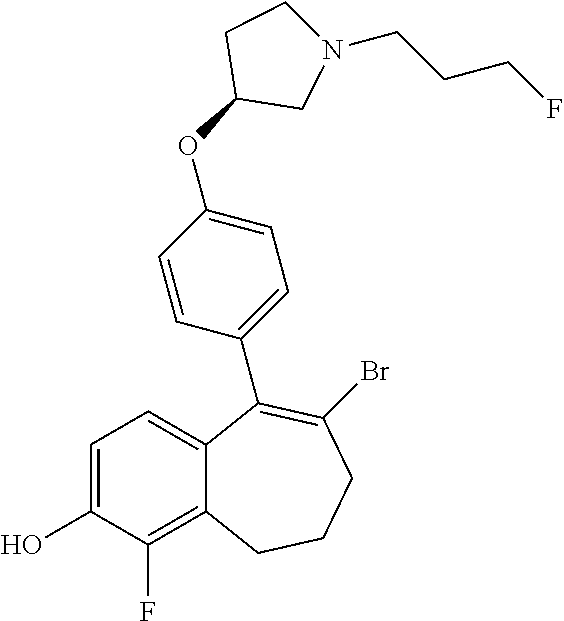

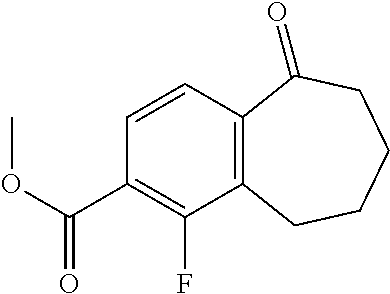





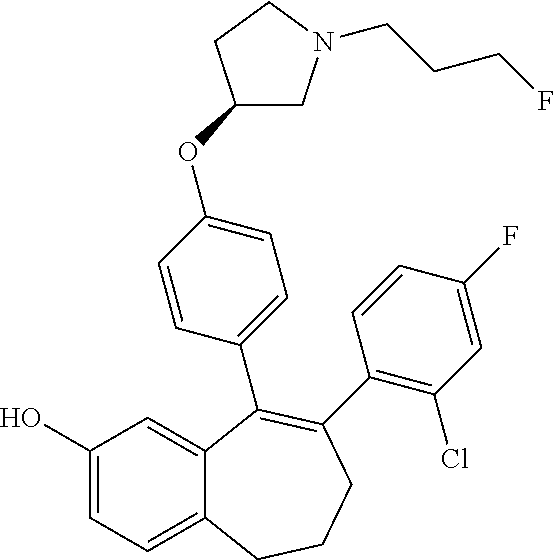
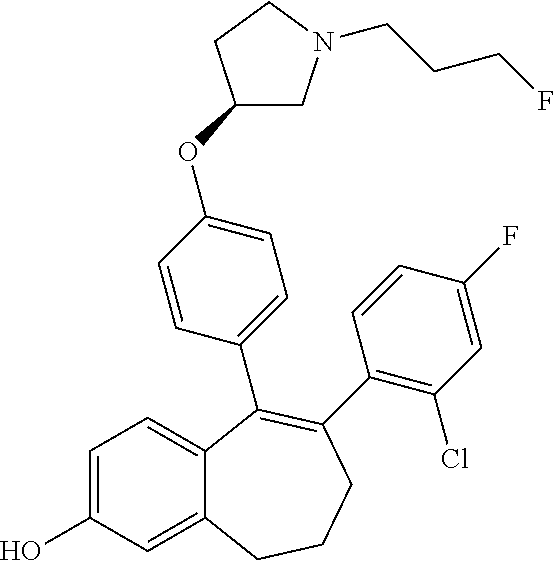



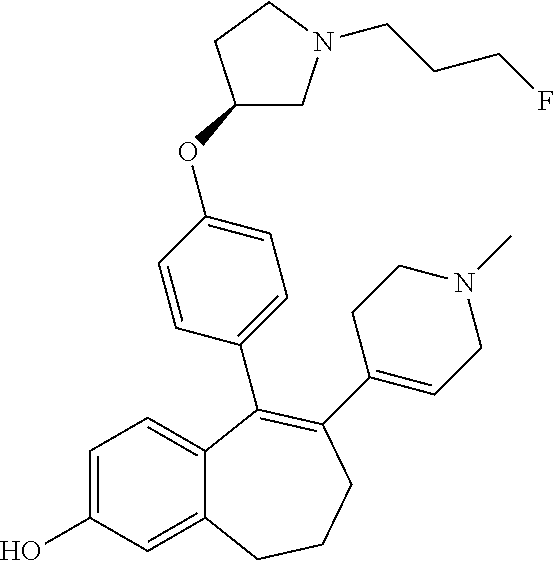


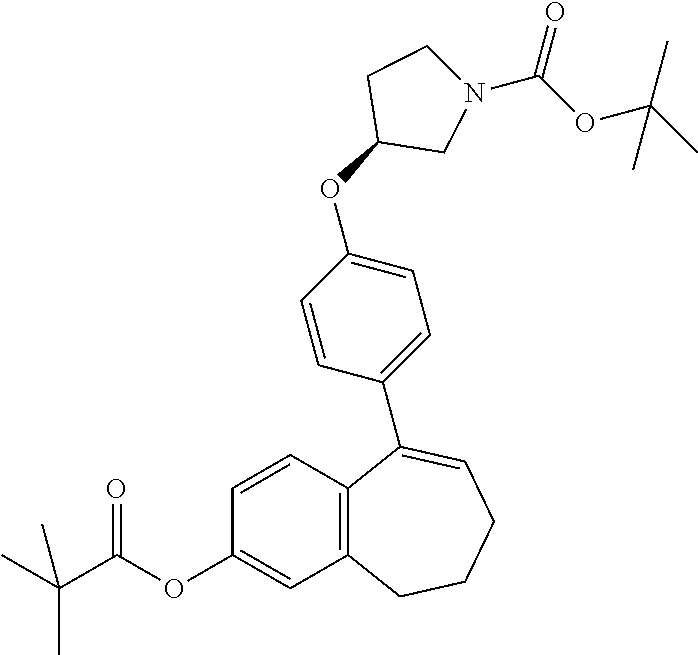

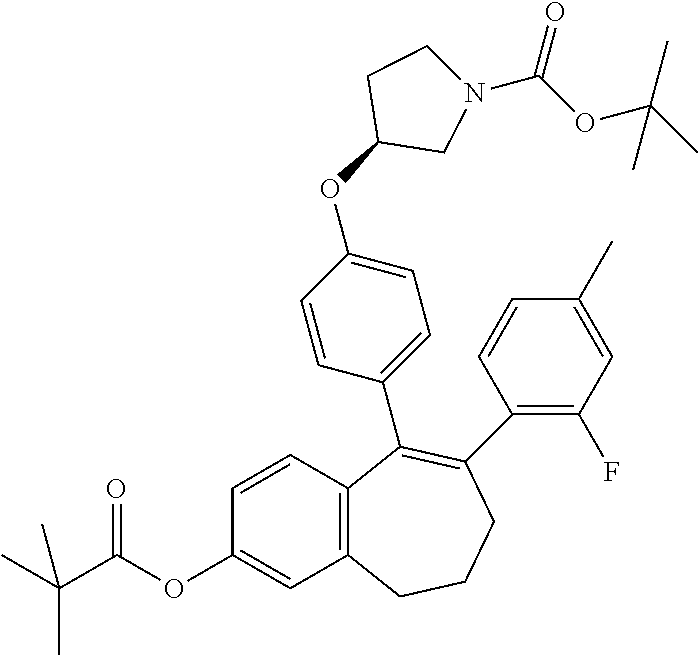
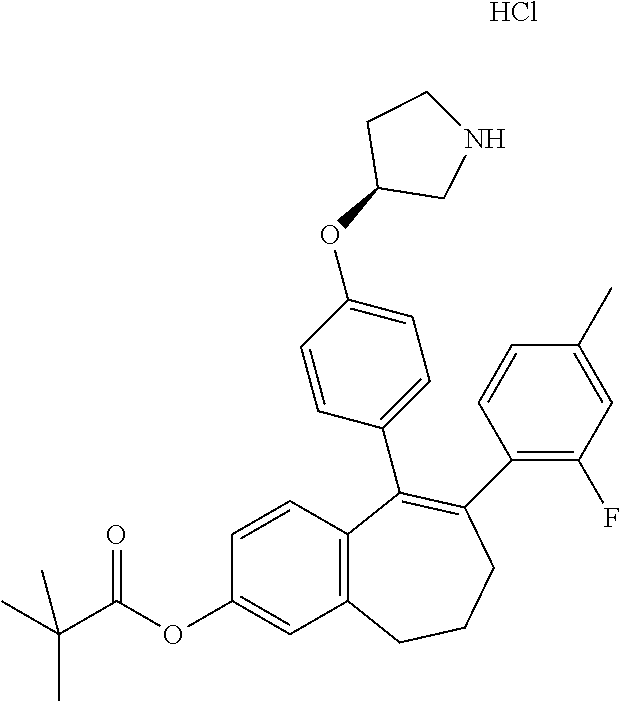

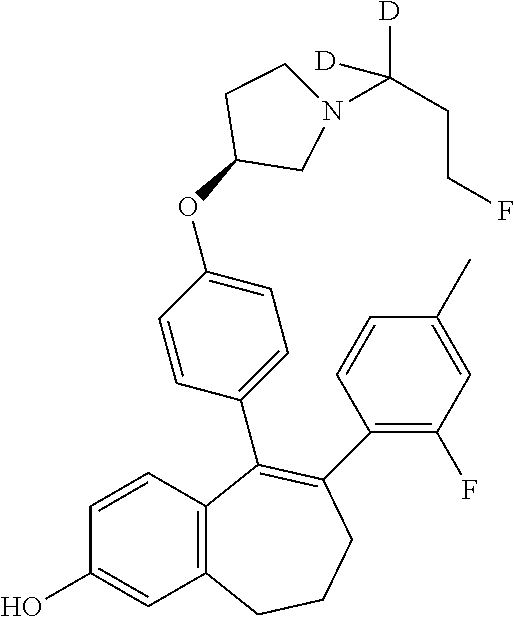












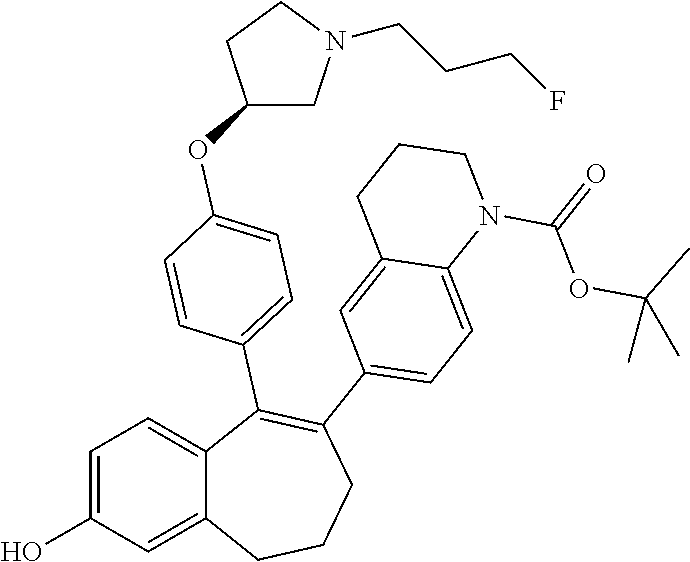








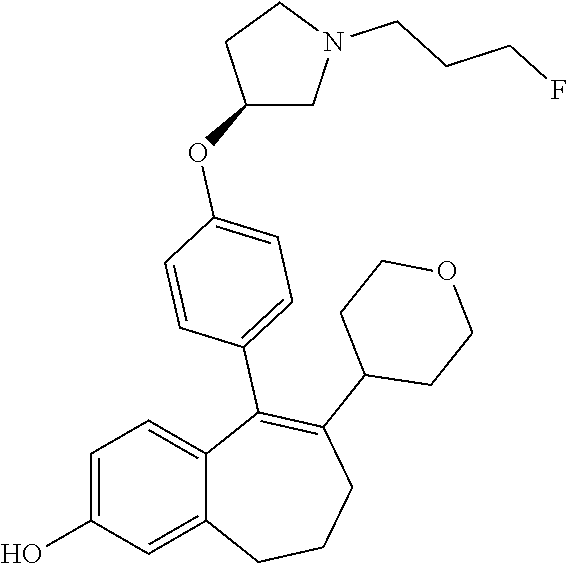










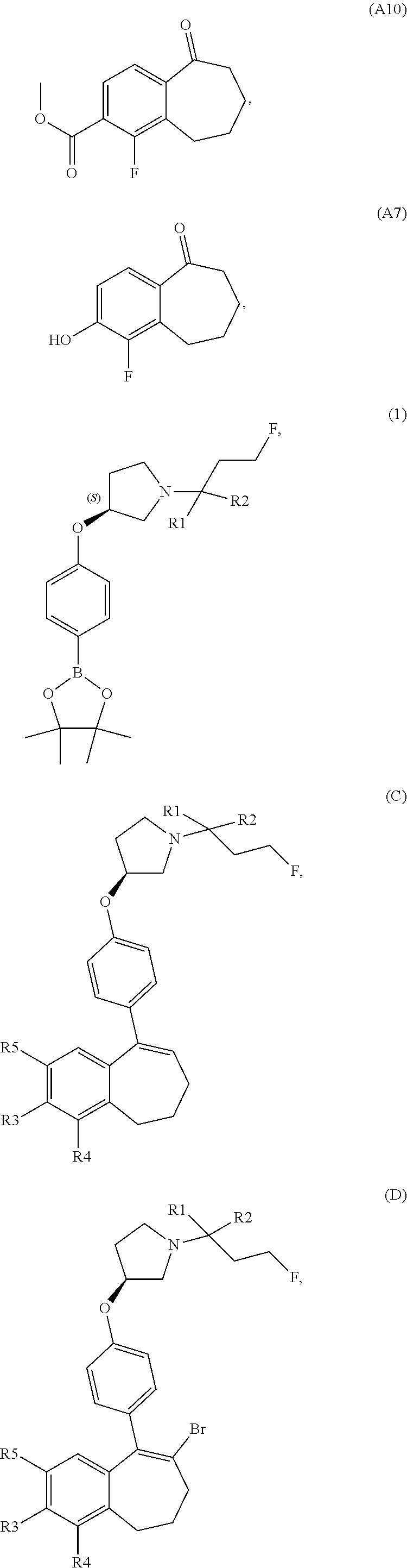


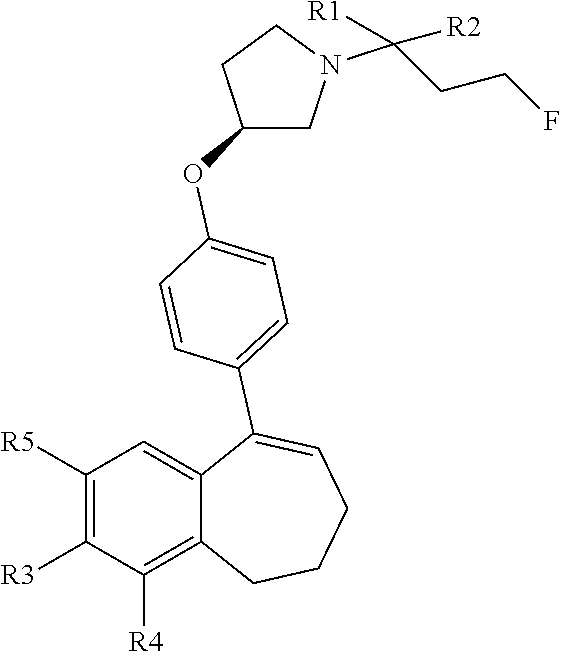
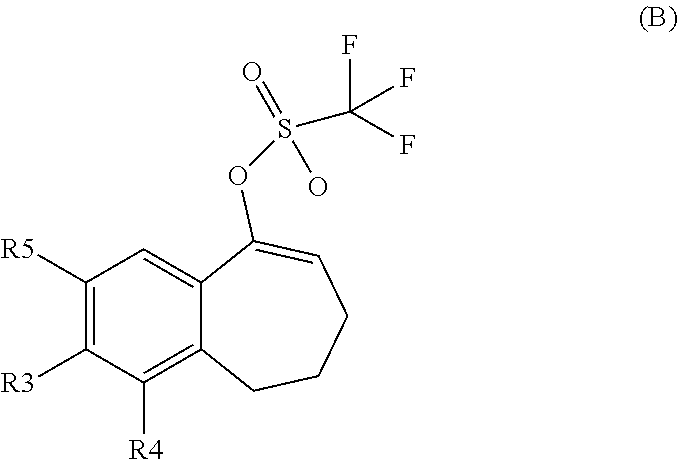

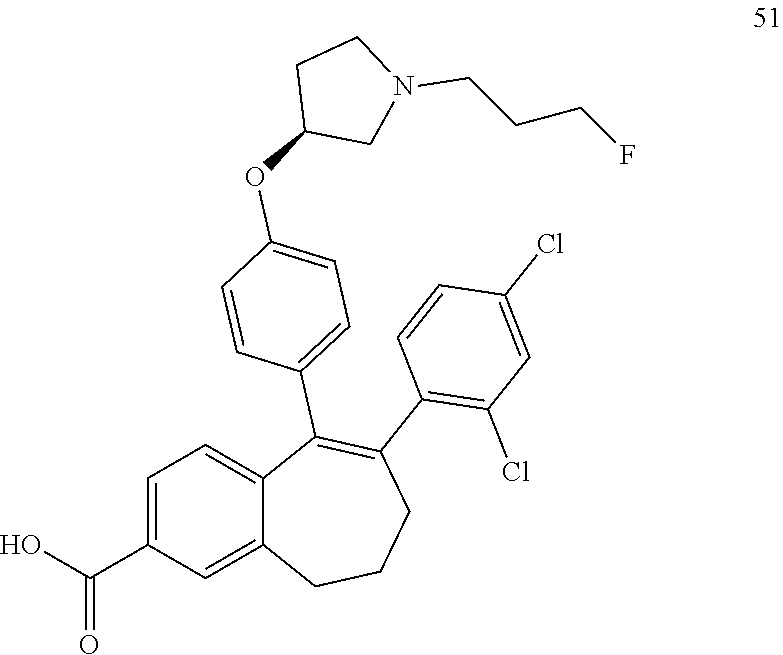


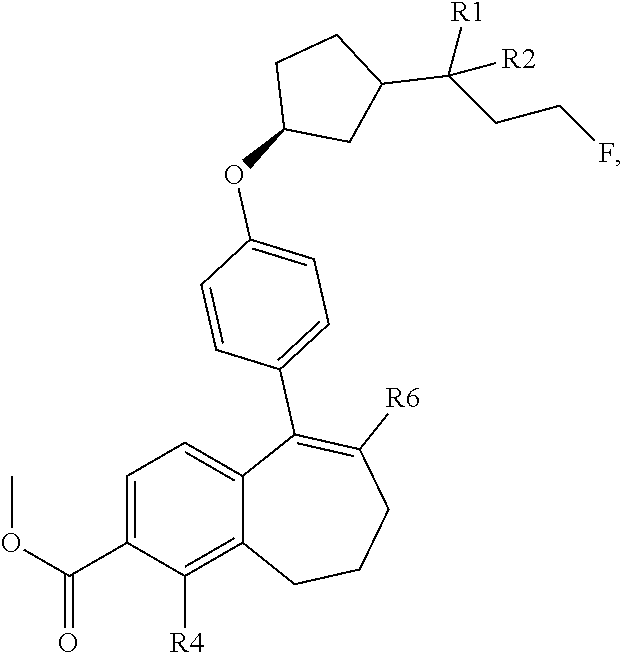



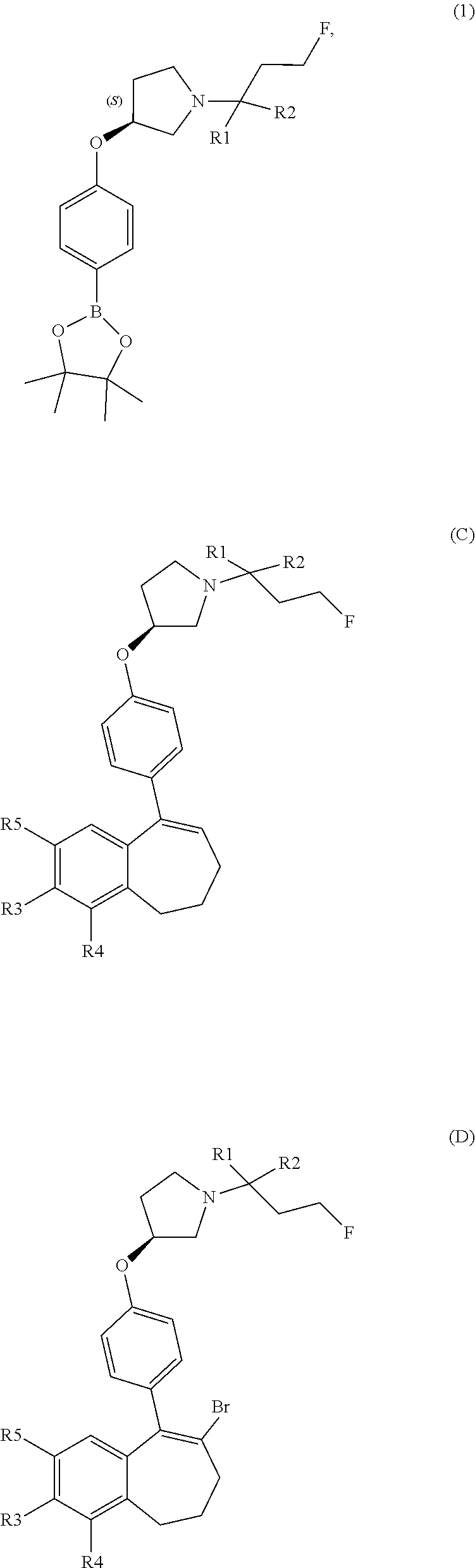


XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.