Compositions, Articles Of Manufacture And Methods For Treating Cancer
PELED; Amnon ; et al.
U.S. patent application number 17/006954 was filed with the patent office on 2020-12-17 for compositions, articles of manufacture and methods for treating cancer. This patent application is currently assigned to Biokine Therapeutics Ltd.. The applicant listed for this patent is Biokine Therapeutics Ltd., BioLineRx Ltd.. Invention is credited to Amnon PELED, Yaron PEREG.
| Application Number | 20200390855 17/006954 |
| Document ID | / |
| Family ID | 1000005059677 |
| Filed Date | 2020-12-17 |
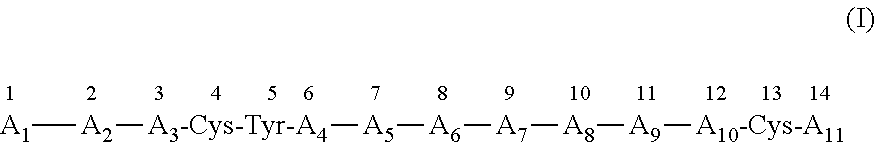

| United States Patent Application | 20200390855 |
| Kind Code | A1 |
| PELED; Amnon ; et al. | December 17, 2020 |
COMPOSITIONS, ARTICLES OF MANUFACTURE AND METHODS FOR TREATING CANCER
Abstract
A method of treating cancer in a subject in need thereof is provided. The method comprising administering to the subject a therapeutically effective amount of a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof; and an anti-cancer agent, thereby treating the cancer in the subject.
| Inventors: | PELED; Amnon; (Tel-Aviv, IL) ; PEREG; Yaron; (Shoham, IL) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Biokine Therapeutics Ltd. Nes Ziona IL BioLineRx Ltd. ModiIn IL |
||||||||||
| Family ID: | 1000005059677 | ||||||||||
| Appl. No.: | 17/006954 | ||||||||||
| Filed: | August 31, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15570342 | Oct 29, 2017 | 10786547 | ||
| PCT/IL2016/050765 | Jul 14, 2016 | |||
| 17006954 | ||||
| 62193201 | Jul 16, 2015 | |||
| 62259182 | Nov 24, 2015 | |||
| 62291039 | Feb 4, 2016 | |||
| 62291006 | Feb 4, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 38/395 20130101; A61K 39/3955 20130101; A61K 47/646 20170801; A61K 2039/55522 20130101; A61K 39/12 20130101; C07K 16/2827 20130101; A61P 35/00 20180101; C07K 16/30 20130101; C07K 2317/76 20130101; A61K 38/1761 20130101; C07K 16/2818 20130101; A61K 38/04 20130101; A61K 38/10 20130101 |
| International Class: | A61K 38/17 20060101 A61K038/17; A61K 38/04 20060101 A61K038/04; A61K 47/64 20060101 A61K047/64; A61P 35/00 20060101 A61P035/00; A61K 38/10 20060101 A61K038/10; A61K 39/12 20060101 A61K039/12; A61K 39/395 20060101 A61K039/395; C07K 16/28 20060101 C07K016/28; C07K 16/30 20060101 C07K016/30 |
Claims
1. An article of manufacture identified for use in treating cancer, comprising a packaging material, packaging a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 and an immune checkpoint regulator, wherein said immune checkpoint regulator is selected from the group consisting of B7H2, B7H3, B7H4, BTLA-4, HVEM, CD80, CD86, CD19, OX40L, 4-1BBL, CD70, CD40L, ICOSL and VISTA.
2. The article of manufacture of claim 1, wherein said peptide and said immune checkpoint regulator are in separate containers.
3. The article of manufacture of claim 1, wherein said peptide and said immune checkpoint regulator are in a co-formulation.
4. The article of manufacture of claim 1, wherein said cancer is a non-solid tumor.
5. The article of manufacture of claim 1, wherein said cancer is a solid tumor.
6. A method of treating cancer in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 and an immune checkpoint regulator, wherein said immune checkpoint regulator is selected from the group consisting of B7H2, B7H3, B7H4, BTLA-4, HVEM, CD80, CD86, CD19, OX40L, 4-1BBL, CD70, CD40L, ICOSL and VISTA.
7. The method of claim 6, wherein said peptide and said immune checkpoint regulator are in separate containers.
8. The method of claim 6, wherein said peptide and said immune checkpoint regulator are in a co-formulation.
9. The method of claim 6, wherein said cancer is a non-solid tumor.
10. The method of claim 6, wherein said cancer is a solid tumor.
Description
RELATED APPLICATIONS
[0001] This application is a Division of U.S. patent application Ser. No. 15/570,342 filed on Oct. 29, 2020, which is a National Phase of PCT Patent Application No. PCT/IL2016/050765 having International Filing Date of Jul. 14, 2016, which claims the benefit of priority under 35 USC .sctn. 119(e) of U.S. Provisional Patent Application Nos. 62/193,201 filed on Jul. 16, 2015; 62/259,182 filed on Nov. 24, 2015; 62/291,039 filed on Feb. 4, 2016; and 62/291,006 filed on Feb. 4, 2016. The contents of the above applications are all incorporated by reference as if fully set forth herein in their entirety.
SEQUENCE LISTING STATEMENT
[0002] The ASCII file, entitled 83822SequenceListing.txt, created on Aug. 27, 2020, comprising 39,930 bytes, submitted concurrently with the filing of this application is incorporated herein by reference. The sequence listing submitted herewith is identical to the sequence listing forming part of the international application.
FIELD AND BACKGROUND OF THE INVENTION
[0003] The present invention, in some embodiments thereof, relates to methods of treating cancer and, more particularly, but not exclusively, to the use of a CXCR4 antagonistic peptide and an anti-cancer agent in the treatment of cancer.
[0004] Cancer is the second leading cause of death in the US. The estimates for 2014 are that approximately 585,000 people will die of cancer and 1.6 million new cases will be diagnosed (American Cancer Society, Cancer Facts & FIGS. 2014). For early stage cancers, surgical removal is a very effective treatment. However, for more advanced cases and non-solid hematological malignancies, standard, non-specific cancer treatments such as chemotherapy and radiotherapy are typically used. These treatments affect many healthy cells and result in elevated toxicity and effective in only a minor percentage of treated individuals. Moreover, even individuals that initially respond to therapy are at risk for relapses, and often develop resistance.
[0005] Significant progress in understanding the underlying principles of tumor biology as well as the basic mechanisms of the immune response to cancer have led to the development of new immunotherapies aimed at employing the adaptive immune system to eradicate cancer with enhanced efficacy and reduced toxicity. Current immunotherapy strategies include cytokines, monoclonal antibodies against tumor cells or immune regulatory molecules, cancer vaccines and cell-based therapies such as adoptive transfer of ex-vivo activated T cells and natural killer (NK) cells.
[0006] Thus, for example, monoclonal antibodies have become part of the therapeutic repertoire for several type of cancers with the anti-CD20 mAb, Rituximab, and the anti-human HER2, Trastuzumab, routinely used for the treatment of breast cancer; and the anti-human EGFR, Cetuximab, routinely used for the treatment of head and neck cancer and colorectal carcinoma [Kirkwood et al. (2012) CA Cancer J Clin. 62(5): 309-335]. In addition, several recombinant IFN.alpha. [IFN-.alpha.2 (Intron-A, Merck); Roferon-A (Roche)] and IL-2 (aldesleukin, Proleukin; Prometheus Inc.) were approved by the United States Food and Drug Administration for the treatment of e.g. melanoma; and dendritic cell vaccines have shown safety and efficacy in several solid tumors, for example sipuleucel-T (Provenge; Dendreon Corporation) in prostate cancer and dendritic cells loaded with four melanoma peptides (gp100, melan-A/MART-1, tyrosinase and MAGE-3), KLH and flu matrix peptide in metastatic melanoma [Kirkwood et al. (2012) CA Cancer J Clin. 62(5): 309-335; and Banchereau (2001) Cancer Research 61: 6451-58].
[0007] 4F-benzoyl-TN14003 (also known as BKT140, hereinafter BL-8040), is a 14-residue bio stable synthetic peptide developed as a specific CXCR4 antagonist. It has been shown that BL-8040 binds the CXCR4 receptor with high affinity and long receptor occupancy. Studies in mice demonstrated that a single BL-8040 injection mobilized long term repopulating stem cells sufficient for transplantation. [Abraham M et al., Stem Cells (2007); 25:2158-66] Results from a study in multiple myeloma patients showed that combined treatment of BL-8040 and G-CSF enabled the collection of high number of CD34+ hematopoietic stem/progenitor cells (HSPC) in a single aphaeresis procedure.[Peled A et al. Clin Cancer Res; (2013) 20(2); 469-79] In addition, BL-8040 was found to be toxic against several tumors such as myeloid leukemia, hematopoietic tumors and non-small cell lung cancer (International Patent Application No. IL2014/050939 and International Patent Application Publication Nos. WO2013/160895 and WO2008/075370).
[0008] Additional background art includes:
[0009] International Patent Application Publication No. WO2014/155376;
[0010] International Patent Application Publication No. WO2012/095849;
[0011] International Patent Application Publication No. WO2002/020561;
[0012] International Patent Application Publication No. WO2004/020462;
[0013] International Patent Application Publication No. WO2008/075369;
[0014] International Patent Application Publication No. WO2008/075370;
[0015] International Patent Application Publication No. WO2008/075371;
[0016] International Patent Application Publication No. WO2010/146578;
[0017] International Patent Application Publication No. WO2010/146584;
[0018] International Patent Application Publication No. WO2003/072599;
[0019] International Patent Application Publication no. WO 2015/019284; and
[0020] U.S. Patent Application Publication No. US 2012/0082687.
SUMMARY OF THE INVENTION
[0021] According to an aspect of some embodiments of the present invention there is provided a method of treating cancer in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof; and an anti-cancer agent selected from the group consisting of:
[0022] (i) a vaccine selected from the group consisting of IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000;
[0023] (ii) anti-cancer reactive mononuclear blood cells (MNBCs);
[0024] (iii) a cytokine capable of inducing activation and/or proliferation of a T cell;
[0025] (iv) an immune-check point regulator, wherein the immune-check point regulator is not a PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist or ICOS agonist;
[0026] (v) an agent capable of binding an immune-check point protein expressed on a cancer cell;
[0027] (vi) a colony stimulating factor-1 receptor (CSF1R) antagonist;
[0028] (vii) a CXCR2 antagonist;
[0029] (viii) a STAT3 antagonist;
[0030] (ix) PV-10; and
[0031] (x) Cotara,
[0032] thereby treating the cancer in the subject.
[0033] According to some embodiments of the invention, the administering comprises multiple administrations of the peptide.
[0034] According to some embodiments of the invention, the administering comprises multiple administrations of the agent.
[0035] According to some embodiments of the invention, the administering the peptide and the administering the agent are effected sequentially.
[0036] According to some embodiments of the invention, the administering the peptide is effected prior to the administering the agent.
[0037] According to some embodiments of the invention, the administering the peptide is effected following the administering the agent.
[0038] According to some embodiments of the invention, the administering the peptide is effected concomitantly with the administering the agent.
[0039] According to some embodiments of the invention, the peptide is administered at a dose of 0.5-1 mg/kg.
[0040] According to some embodiments of the invention, the peptide is administered subcutaneously.
[0041] According to an aspect of some embodiments of the present invention there is provided an article of manufacture identified for use in treating cancer, comprising a packaging material packaging a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof and an anti-cancer agent selected from the group consisting of:
[0042] (i) a vaccine selected from the group consisting of IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623,
[0043] Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000;
[0044] (ii) anti-cancer reactive mononuclear blood cells (MNBCs);
[0045] (iii) a cytokine capable of inducing activation and/or proliferation of a T cell;
[0046] (iv) an immune-check point regulator, wherein the immune-check point regulator is not a PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist or ICOS agonist;
[0047] (v) an agent capable of binding an immune-check point protein expressed on a cancer cell;
[0048] (vi) a colony stimulating factor-1 receptor (CSF1R) antagonist;
[0049] (vii) a CXCR2 antagonist;
[0050] (viii) a STAT3 antagonist;
[0051] (ix) PV-10; and
[0052] (x) Cotara.
[0053] According to some embodiments of the invention, the peptide and the agent are in separate containers.
[0054] According to some embodiments of the invention, the peptide and the agent are in a co-formulation.
[0055] According to an aspect of some embodiments of the present invention there is provided a pharmaceutical composition comprising as active ingredients a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof and an anti-cancer agent selected from the group consisting of:
[0056] (i) a vaccine selected from the group consisting of IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000;
[0057] (ii) anti-cancer reactive mononuclear blood cells (MNBCs);
[0058] (iii) a cytokine capable of inducing activation and/or proliferation of a T cell;
[0059] (iv) an immune-check point regulator, wherein the immune-check point regulator is not a PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist or ICOS agonist;
[0060] (v) an agent capable of binding an immune-check point protein expressed on a cancer cell;
[0061] (vi) a colony stimulating factor-1 receptor (CSF1R) antagonist;
[0062] (vii) a CXCR2 antagonist;
[0063] (viii) a STAT3 antagonist;
[0064] (ix) PV-10; and
[0065] (x) Cotara,
[0066] and a pharmaceutically acceptable carrier or diluent.
[0067] According to some embodiments of the invention, the MNBCs are derived from a subject not treated with a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof.
[0068] According to some embodiments of the invention, the MNBCs are selected from the group consisting of T cells, NK cells and dendritic cells.
[0069] According to some embodiments of the invention, the MNBCs comprise T cells.
[0070] According to some embodiments of the invention, the MNBCs comprise dendritic cells.
[0071] According to some embodiments of the invention, the dendritic cells comprise a dendritic cells vaccine.
[0072] According to some embodiments of the invention, the cytokine is selected from the group consisting of IFN.alpha., IFN.gamma., IL-1, IL-2, IL-6, IL-12, IL-15, IL-21 and TNF.alpha..
[0073] According to some embodiments of the invention, the immune-check point regulator targets an immune check-point protein selected from the group consisting of B7-H3, CD19 and CD70.
[0074] According to some embodiments of the invention, the immune-check point regulator is selected from the group consisting of an antibody, a peptide and a small molecule.
[0075] According to some embodiments of the invention, the binding of the agent to the immune-check point protein expressed on the cancer cell results in at least one of:
[0076] (i) cell cycle arrest of the cancer cell;
[0077] (ii) apoptosis of the cancer cell;
[0078] (iii) sensitization of the cancer cell to a cytotoxic drug; and
[0079] (iv) activation of an immune response against the cancer cell.
[0080] According to some embodiments of the invention, the immune-check point protein expressed on the cancer cell is selected from the group consisting of LAG3, CD19 and CD70 and CEACAM1.
[0081] According to some embodiments of the invention, the agent capable of binding the immune-check point protein is an antibody or a T cell.
[0082] According to some embodiments of the invention, the T cell comprises a T cell transduced with a T cell receptor (TCR) or a chimeric antigen receptor (CAR).
[0083] According to some embodiments of the invention, the analog or derivative has an amino acid sequence as set forth in formula (I) or a salt thereof:
##STR00001##
[0084] wherein:
[0085] A.sub.1 is an arginine, lysine, ornithine, citrulline, alanine or glutamic acid residue or a N-.alpha.-substituted derivative of these amino acids, or A.sub.1 is absent;
[0086] A.sub.2 represents an arginine or glutamic acid residue if A.sub.1 is present, or A.sub.2 represents an arginine or glutamic acid residue or a N-.alpha.-substituted derivative of these amino acids if A.sub.1 is absent;
[0087] A.sub.3 represents an aromatic amino acid residue;
[0088] A.sub.4, A.sub.5 and A.sub.9 each independently represents an arginine, lysine, ornithine, citrulline, alanine or glutamic acid residue;
[0089] A.sub.6 represents a proline, glycine, ornithine, lysine, alanine, citrulline, arginine or glutamic acid residue;
[0090] A.sub.7 represents a proline, glycine, ornithine, lysine, alanine, citrulline or arginine residue;
[0091] A.sub.8 represents a tyrosine, phenylalanine, alanine, naphthylalanine, citrulline or glutamic acid residue;
[0092] A.sub.10 represents a citrulline, glutamic acid, arginine or lysine residue;
[0093] A.sub.11 represents an arginine, glutamic acid, lysine or citrulline residue wherein the C-terminal carboxyl may be derivatized;
[0094] and the cysteine residue of the 4-position or the 13-position can form a disulfide bond, and the amino acids can be of either L or D form.
[0095] According to some embodiments of the invention, the peptide is selected from the group consisting of SEQ ID NOs: 1-72.
[0096] According to some embodiments of the invention, the peptide is as set forth in SEQ ID NO: 1.
[0097] According to some embodiments of the invention, the cancer is a solid tumor cancer.
[0098] According to some embodiments of the invention, the solid tumor is selected from the group consisting of lung cancer, glioma, colon cancer, ovarian cancer, renal cancer, melanoma cancer, hepatocellular cancer, gastric or stomach cancer, glioblastoma, cervical cancer, bladder cancer, breast cancer, colorectal cancer, prostate cancer, thyroid cancer, head and neck and pancreatic cancer.
[0099] According to some embodiments of the invention, the cancer is a non-solid tumor cancer.
[0100] According to some embodiments of the invention, the non-solid tumor is selected from the group consisting of multiple myeloma and leukemia.
[0101] Unless otherwise defined, all technical and/or scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which the invention pertains. Although methods and materials similar or equivalent to those described herein can be used in the practice or testing of embodiments of the invention, exemplary methods and/or materials are described below. In case of conflict, the patent specification, including definitions, will control. In addition, the materials, methods, and examples are illustrative only and are not intended to be necessarily limiting.
DESCRIPTION OF SPECIFIC EMBODIMENTS OF THE INVENTION
[0102] The present invention, in some embodiments thereof, relates to methods of treating cancer and, more particularly, but not exclusively, to the use of a CXCR4 antagonistic peptide and an anti-cancer agent in the treatment of cancer.
[0103] Before explaining at least one embodiment of the invention in detail, it is to be understood that the invention is not necessarily limited in its application to the details set forth in the following description or exemplified by the Examples. The invention is capable of other embodiments or of being practiced or carried out in various ways.
[0104] Treatment of most types of cancer involves cytotoxic treatments such as chemotherapy and radiotherapy that may at least in part affect many healthy cells and thus result in elevated toxicity.
[0105] In addition, these treatments are effective in only a small percentage of cancer affected patients. Immunotherapy strategies for cancer therapy, aiming at harnessing the immune system to fight cancer, include cytokines, monoclonal antibodies against tumor cells or immune regulatory molecules, cancer vaccines as well as cell-based therapies such as adoptive transfer of ex-vivo activated T cells and natural killer (NK) cells.
[0106] 4F-benzoyl-TN14003 (SEQ ID NO: 1, also known as BKT140, hereinafter BL-8040) is a CXCR4 peptide antagonist. It has been shown that BL-8040 induces mobilization of CD34+ hematopoietic stem/progenitor cells (HSPC) that can be further used for transplantation. In addition, BL-8040 was found to be toxic against several tumors such as myeloid leukemia, hematopoietic tumors and non-small cell lung cancer.
[0107] While reducing the present invention to practice, the present inventors have found that in-vivo administration of BL-8040 induces rapid mobilization of a variety of immune cells including immature stem/progenitor cells as well as fully differentiated T cells and NK cells. The present findings therefore can be harnessed to the use of BL-8040 to induce the mobilization and dissemination of ImDCs and T effector and memory cells into tumor sites and thus can augment the anti-tumor effect of anti-cancer agents such as immunotherapeutics.
[0108] Consequently, the present teachings and the protocols presented in Example 1, suggest the use of a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof in combination with several anti-cancer agents for the treatment of cancer.
[0109] Thus, according to a first aspect of the present invention there is provided a method of treating cancer in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof; and an anti-cancer agent selected from the group consisting of:
[0110] (i) a vaccine selected from the group consisting of IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000;
[0111] (ii) anti-cancer reactive mononuclear blood cells (MNBCs);
[0112] (iii) a cytokine capable of inducing activation and/or proliferation of a T cell;
[0113] (iv) an immune-check point regulator, wherein said immune-check point regulator is not a PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist or ICOS agonist;
[0114] (v) an agent capable of binding an immune-check point protein expressed on a cancer cell;
[0115] (vi) a colony stimulating factor-1 receptor (CSF1R) antagonist;
[0116] (vii) a CXCR2 antagonist;
[0117] (viii) a STAT3 antagonist;
[0118] (ix) PV-10; and
[0119] (x) Cotara,
[0120] thereby treating the cancer in the subject.
[0121] As used herein, the terms "treating" or "treatment" refers to inhibiting, preventing or arresting the development of a pathology (e.g. cancer) and/or causing the reduction, remission, or regression of a pathology. Those of skill in the art will understand that various methodologies and assays can be used to assess the development of a pathology, and similarly, various methodologies and assays may be used to assess the reduction, remission or regression of a pathology.
[0122] As used herein the phrase "subject in need thereof" refers to a mammalian male or female subject (e.g., human being) who is diagnosed with cancer. In a specific embodiment, this term encompasses individuals who are at risk to develop cancer. Veterinary uses are also contemplated. The subject may be of any gender or at any age including neonatal, infant, juvenile, adolescent, adult and elderly adult.
[0123] The terms "cancer" and "cancerous" refer to or describe the physiological condition in mammals that is typically characterized by unregulated cell growth.
[0124] Cancers which can be treated by the method of this aspect of some embodiments of the invention can be any solid or non-solid cancer and/or cancer metastasis.
[0125] According to a specific embodiment, the cancer is a solid tumor.
[0126] According another specific embodiment, the cancer is a non-solid tumor.
[0127] Examples of cancer include but are not limited to, carcinoma, lymphoma, blastoma, sarcoma, and leukemia. More particular examples of such cancers include squamous cell cancer, lung cancer (including small-cell lung cancer, non-small-cell lung cancer, adenocarcinoma of the lung, and squamous carcinoma of the lung), melanoma cancer, cancer of the peritoneum, hepatocellular cancer, gastric or stomach cancer (including gastrointestinal cancer), pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, hepatoma, breast cancer, colon cancer, colorectal cancer, endometrial or uterine carcinoma, salivary gland carcinoma, kidney or renal cancer, liver cancer, prostate cancer, vulval cancer, thyroid cancer, hepatic carcinoma and various types of head and neck cancer, as well as B-cell lymphoma (including low grade/follicular non-Hodgkin's lymphoma (NHL); small lymphocytic (SL) NHL; intermediate grade/follicular NHL; intermediate grade diffuse NHL; high grade immunoblastic NHL; high grade lymphoblastic NHL; high-grade small non-cleaved cell NHL; bulky disease NHL; mantle cell lymphoma; AIDS-related lymphoma; and Waldenstrom's Macroglobulinemia); chronic lymphocytic leukemia (CLL); acute lymphoblastic leukemia (ALL); Hairy cell leukemia; chronic myeloblastic leukemia; acute myeloblastic leukemia; Multiple Myeloma; and post-transplant lymphoproliferative disorder (PTLD), as well as abnormal vascular proliferation associated with phakomatoses, edema (such as that associated with brain tumors), and Meigs' syndrome. Preferably, the cancer is selected from the group consisting of breast cancer, colorectal cancer, rectal cancer, non-small cell lung cancer, non-Hodgkins lymphoma (NHL), acute lymphoblastic leukemia (ALL); chronic myeloblastic leukemia (CML); acute myeloblastic leukemia (AML); renal cell cancer, prostate cancer, liver cancer, pancreatic cancer, soft-tissue sarcoma, Kaposi's sarcoma, carcinoid carcinoma, head and neck cancer, melanoma, ovarian cancer, mesothelioma, and multiple myeloma. The cancerous conditions amenable for treatment of the invention include metastatic cancers.
[0128] According to specific embodiments the cancer is selected from the group consisting of lung cancer, glioma, colon cancer, ovarian cancer, renal cancer, melanoma cancer, hepatocellular cancer, gastric or stomach cancer, glioblastoma, cervical cancer, bladder cancer, breast cancer, colorectal cancer, prostate cancer, thyroid cancer, head and neck and pancreatic cancer.
[0129] According to specific embodiments, the cancer is selected from the group consisting of lung cancer, glioma, colon cancer and pancreatic cancer.
[0130] According to specific embodiments, the cancer is selected from the group consisting of multiple myeloma, Lymphoma and leukemia.
[0131] According to other specific embodiments, the cancer is selected from the group consisting of multiple myeloma and leukemia.
[0132] As used herein, the term "peptide" encompasses native peptides (either degradation products, synthetically synthesized peptides or recombinant peptides) and peptidomimetics (typically, synthetically synthesized peptides), as well as peptoids and semipeptoids which are peptide analogs, which may have, for example, modifications rendering the peptides more stable while in a body or more capable of penetrating into cells.
[0133] According to a specific embodiment, the peptide is no more than 100 amino acids in length. According to a specific embodiment, the peptide is 5-100 amino acids in length. According to a specific embodiment, the peptide is 5-50 amino acids in length. According to a specific embodiment, the peptide is 5-20 amino acids in length. According to a specific embodiment, the peptide is 5-15 amino acids in length. According to a specific embodiment, the peptide is 10-20 amino acids in length. According to a specific embodiment, the peptide is 10-15 amino acids in length.
[0134] As used herein the term "peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof" refers to 4F-benzoyl-TN14003 (SEQ ID NO: 1, also known as BKT140, hereinafter BL-8040) peptide and functional analogs or derivatives thereof. The peptides of the present invention are structurally and functionally related to the peptides disclosed in patent applications WO2002/020561 and WO2004/020462, also known as "T-140 analogs", as detailed hereinbelow. The peptide of the present invention is a CXCR4-antagonistic peptide i.e. it reduces CXCR-4 activation by at least 10% as compared to same in the absence of the peptide antagonist. According to a specific embodiment the peptide antagonist is a competitive inhibitor. According to a specific embodiment the peptide antagonist is a non-competitive inhibitor.
[0135] According to specific embodiments, a functional CXCR4 antagonistic peptide, as used herein, is capable of inducing mobilization and dissemination of ImDCs, NK cells, B cells, monocytes/macrophages and T effector and memory cells into a tumor of a subject upon administration.
[0136] According to other specific embodiments, a functional CXCR4 antagonistic peptide, as used herein, is capable of enhancing an immune-response to a tumor.
[0137] In various particular embodiments, the peptide analog or derivative has an amino acid sequence as set forth in the following formula (I) or a salt thereof:
##STR00002##
[0138] wherein:
[0139] A.sub.1 is an arginine, lysine, ornithine, citrulline, alanine or glutamic acid residue or a N-.alpha.-substituted derivative of these amino acids, or A.sub.1 is absent;
[0140] A.sub.2 represents an arginine or glutamic acid residue if A.sub.1 is present, or A.sub.2 represents an arginine or glutamic acid residue or a N-.alpha.-substituted derivative of these amino acids if A.sub.1 is absent;
[0141] A.sub.3 represents an aromatic amino acid residue;
[0142] A.sub.4, A.sub.5 and A.sub.9 each independently represents an arginine, lysine, ornithine, citrulline, alanine or glutamic acid residue;
[0143] A.sub.6 represents a proline, glycine, ornithine, lysine, alanine, citrulline, arginine or glutamic acid residue;
[0144] A.sub.7 represents a proline, glycine, ornithine, lysine, alanine, citrulline or arginine residue;
[0145] A.sub.8 represents a tyrosine, phenylalanine, alanine, naphthylalanine, citrulline or glutamic acid residue;
[0146] A.sub.10 represents a citrulline, glutamic acid, arginine or lysine residue;
[0147] A.sub.11 represents an arginine, glutamic acid, lysine or citrulline residue wherein the C-terminal carboxyl may be derivatized;
[0148] and the cysteine residue of the 4-position or the 13-position can form a disulfide bond, and the amino acids can be of either L or D form.
[0149] Exemplary peptides according to formula (I) are peptides having an amino acid sequence as set forth in any one of SEQ ID NOs: 1-72, as presented in Table 1 hereinbelow.
TABLE-US-00001 TABLE 1 T-140 and currently preferred T-140 analogs SEQ ID Analog NO: Amino acid sequence 4F-benzoyl- 1 4F-benzoyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg- TN14003 NH.sub.2 AcTC14003 2 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH AcTC14005 3 Ac-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-OH AcTC14011 4 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-OH AcTC14013 5 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Cit-Cit-Cys-Arg-OH AcTC14015 6 Ac-Cit-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH AcTC14017 7 Ac-Cit-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-OH AcTC14019 8 Ac-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Cit-Cit-Cys-Arg-OH AcTC14021 9 Ac-Cit-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Cit-Cit-Cys-Arg-OH AcTC14012 10 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 AcTC14014 11 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Cit-Cit-Cys-Arg-NH.sub.2 AcTC14016 12 Ac-Cit-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 AcTC14018 13 Ac-Cit-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 AcTC14020 14 Ac-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Cit-Cit-Cys-Arg-NH.sub.2 AcTC14022 15 Ac-Cit-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Cit-Cit-Cys-Arg-NH.sub.2 TE14001 16 H-DGlu-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TE14002 17 H-Arg-Glu-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TE14003 18 H-Arg-Arg-Nal-Cys-Tyr-Glu-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TE14004 19 H-Arg-Arg-Nal-Cys-Tyr-Arg-Glu-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TE14005 20 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-OH TE14006 21 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Glu-Cit-Cys-Arg-OH TE14007 22 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Glu-OH TE14011 23 H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.s- ub.2 TE14012 24 H-Arg-Arg-Nal-Cys-Tyr-DGlu-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-NH.- sub.2 TE14013 25 H-Arg-Arg-Nal-Cys-Tyr-DGlu-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.- sub.2 TE14014 26 H-DGlu-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.- sub.2 TE14015 27 H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-DGlu-Arg-Cit-Cys-Arg-NH.- sub.2 TE14016 28 H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-DGlu-Cys-Arg-NH.- sub.2 AcTE14014 29 Ac-DGlu-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 AcTE14015 30 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-DGlu-Arg-Cit-Cys-Arg-NH.sub.2 AcTE14016 31 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-DGlu-Cys-Arg-NH.sub.2 TF1: 32 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub- .2 AcTE14011 TF2: guanyl- 33 guanyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 TE14011 TF3: TMguanyl- 34 TMguanyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.- 2 TE14011 TF4: TMguanyl- 35 TMguanyl-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 TE14011 (2-14) TF5: 4F- 36 4F-benzoyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg- benzoyl- NH.sub.2 TE14011 TF6: 2F- 37 2F-benzoyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg- benzoyl- NH.sub.2 TE14011 TF7: APA- 38 APA-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 TE14011 (2-14) TF8: desamino- 39 desamino-R-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 R-TE14011 (2-14) TF9: guanyl- 40 Guanyl-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 TE14011 (2-14) TF10: succinyl- 41 succinyl-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 TE14011 (2-14) TF11: glutaryl- 42 glutaryl-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 TE14011 (2-14) TF12: 43 deaminoTMG-APA-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-A- rg- deaminoTMG- NH.sub.2 APA-TE14011 (2-14) TF15: H-Arg- 44 R-CH2-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 CH2NH- RTE14011 (2- 14) TF17: TE14011 45 H-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 (2-14) TF18: 46 TMguanyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg- -NH.sub.2 TMguanyl- TC14012 TF19: ACA- 47 ACA-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 TC14012 TF20: ACA- 48 ACA-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH T140 TZ14011 49 H-Arg-Arg-Nal-Cys-Tyr-Cit-Arg-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-NH.s- ub.2 AcTZ14011 50 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Arg-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 AcTN14003 51 Ac-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 AcTN14005 52 Ac-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-NH.sub.2 4F-benzoyl- 53 4F-benzoyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg- TN14011-Me NHMe 4F-benzoyl- 54 4F-benzoyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg- TN14011-Et NHEt 4F-benzoyl- 55 4F-benzoyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg- TN14011-iPr NHiPr 4F-benzoyl- 56 4F-benzoyl-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DGlu-Pro-Tyr-Arg-Cit-Cys-Arg- TN14011- tyramine tyramine TA14001 57 H-Ala-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TA14005 58 H-Arg-Arg-Nal-Cys-Tyr-Ala-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TA14006 59 H-Arg-Arg-Nal-Cys-Tyr-Arg-Ala-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TA14007 60 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DAla-Pro-Tyr-Arg-Cit-Cys-Arg-OH TA14008 61 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Ala-Tyr-Arg-Cit-Cys-Arg-OH TA14009 62 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Ala-Arg-Cit-Cys-Arg-OH TA14010 63 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Ala-Cit-Cys-Arg-OH TC14001 64 H-Cit-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TC14003 65 H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TN14003 66 H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-NH.s- ub.2 TC14004 67 H-Arg-Arg-Nal-Cys-Tyr-Arg-Cit-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TC14012 68 H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-NH.s- ub.2 T-140 69 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH TC14011 70 H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-OH TC14005 71 H-Arg-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-OH TC14018 72 H-Cit-Arg-Nal-Cys-Tyr-Arg-Lys-DCit-Pro-Tyr-Arg-Cit-Cys-Arg-NH.s- ub.2
[0150] According to a specific embodiment, in each one of SEQ ID NOs: 1-72, two cysteine residues are coupled in a disulfide bond.
[0151] In another embodiment, the analog or derivative has an amino acid sequence as set forth in SEQ ID NO: 65 (H-Arg-Arg-Nal-Cys-Tyr-Cit-Lys-DLys-Pro-Tyr-Arg-Cit-Cys-Arg-OH; TC14003).
[0152] In another embodiment, the peptide used in the compositions and methods of the invention consists essentially of an amino acid sequence as set forth in SEQ ID NO: 1. In another embodiment, the peptide used in the compositions and methods of the invention comprises an amino acid sequence as set forth in SEQ ID NO: 1. In another embodiment, the peptide is at least 60%, at least 70% or at least 80% homologous to SEQ ID NO: 1. In another embodiment, the peptide is at least 90% homologous to SEQ ID NO: 1. In another embodiment, the peptide is at least about 95% homologous to SEQ ID NO: 1. Each possibility represents a separate embodiment of the present invention.
[0153] In various other embodiments, the peptide is selected from SEQ ID NOs: 1-72, wherein each possibility represents a separate embodiment of the present invention.
[0154] In another embodiment, the peptide has an amino acid sequence as set forth in any one of SEQ ID NOs: 1-4, 10, 46, 47, 51-56, 65, 66, 68, 70 and 71. In another embodiment, the peptide has an amino acid sequence as set forth in any one of SEQ ID NOs: 4, 10, 46, 47, 68 and 70. In another embodiment, the peptide has an amino acid sequence as set forth in any one of SEQ ID NOs: 1, 2, 51, 65 and 66. In another embodiment, the peptide has an amino acid sequence as set forth in any one of SEQ ID NOs: 53-56.
[0155] In an embodiment, the peptide has an amino acid sequence as set forth in SEQ ID NO: 1. According to a specific embodiment, the peptide is as set forth in SEQ ID NO: 1. In another embodiment, the peptide has an amino acid sequence as set forth in SEQ ID NO: 2. In another embodiment, the peptide has an amino acid sequence as set forth in SEQ ID NO: 51. In another embodiment, the peptide has an amino acid sequence as set forth in SEQ ID NO: 66.
[0156] According to a specific embodiment the peptide is as set forth in SEQ ID NO: 1 and any embodiment described herein should be read as if specifically reading over this peptide.
[0157] The peptides of some embodiments of the invention may be synthesized by any techniques that are known to those skilled in the art of peptide synthesis. For solid phase peptide synthesis, a summary of the many techniques may be found in J. M. Stewart and J. D. Young, Solid Phase Peptide Synthesis, W. H. Freeman Co. (San Francisco), 1963 and J. Meienhofer, Hormonal Proteins and Peptides, vol. 2, p. 46, Academic Press (New York), 1973. For classical solution synthesis see G. Schroder and K. Lupke, The Peptides, vol. 1, Academic Press (New York), 1965.
[0158] In general, these methods comprise the sequential addition of one or more amino acids or suitably protected amino acids to a growing peptide chain. Normally, either the amino or carboxyl group of the first amino acid is protected by a suitable protecting group. The protected or derivatized amino acid can then either be attached to an inert solid support or utilized in solution by adding the next amino acid in the sequence having the complimentary (amino or carboxyl) group suitably protected, under conditions suitable for forming the amide linkage. The protecting group is then removed from this newly added amino acid residue and the next amino acid (suitably protected) is then added, and so forth. After all the desired amino acids have been linked in the proper sequence, any remaining protecting groups (and any solid support) are removed sequentially or concurrently, to afford the final peptide compound. By simple modification of this general procedure, it is possible to add more than one amino acid at a time to a growing chain, for example, by coupling (under conditions which do not racemize chiral centers) a protected tripeptide with a properly protected dipeptide to form, after deprotection, a pentapeptide and so forth. Further description of peptide synthesis is disclosed in U.S. Pat. No. 6,472,505.
[0159] Large scale peptide synthesis is described by Andersson Biopolymers 2000; 55(3):227-50.
[0160] According to specific embodiments, the CXCR4 antagonistic peptide is administered to the subject in combination with one or more white blood cell mobilizing agents. For example, the peptide may be administered in sequential or concomitant combination with one or more other growth factors or cytokines that affect mobilization such as, but not limited to, G-CSF, GM-CSF and SCF.
[0161] As mentioned, the peptide of the present invention is administered in combination with an anti-cancer agent.
[0162] As used herein, the term "anti-cancer agent" refers to an agent effective in inhibiting, slowing or arresting the growth or metastasis of a cancerous cell or which exhibits a cytotoxic effect on a cancerous cell. According to specific embodiments, the anti-cancer agent is an anti-cancer immune modulator agent which is capable of eliciting an immune response (e.g. T cell, NK cell, B cell, complement) against a cancerous cell.
[0163] The anti-cancer agent of the present invention is selected from the group consisting of:
[0164] (i) a vaccine selected from the group consisting of IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000;
[0165] (ii) anti-cancer reactive mononuclear blood cells (MNBCs);
[0166] (iii) a cytokine capable of inducing activation and/or proliferation of a T cell;
[0167] (iv) an immune-check point regulator, wherein said immune-check point regulator is not a PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist or ICOS agonist;
[0168] (v) an agent capable of binding an immune-check point protein expressed on a cancer cell;
[0169] (vi) a colony stimulating factor-1 receptor (CSF1R) antagonist;
[0170] (vii) a CXCR2 antagonist;
[0171] (viii) a STAT3 antagonist;
[0172] (ix) PV-10; and
[0173] (x) Cotara,
[0174] wherein each possibility represents a separate embodiment of the present invention.
[0175] As used herein, the term "vaccine" refers to a composition used to provoke a specific immune response against a cancerous cell (e.g. the production of anti-cancer antibodies, eliciting a cell mediated immune-response) following administration.
[0176] According to specific embodiments of the present invention the vaccine includes: IMCgp100 [a soluble affinity enhanced T cell receptor (TCR) specific for the melanoma-associated antigen gp100, fused to an anti-CD3 specific antibody fragment (scFv), e.g., produced by Immunocore]; the antigen based vaccines Prophage G-100 & G-200 (HSPPC-96, e.g., produced by Agenus), GV-1001 (e.g., produced by KAEL-GemVax), IMA-950 (e.g., produced by Immatics Biotechnologies), CV-9201 (e.g., produced by CureVac) and CV-9104 (CV-9103, e.g., produced by CureVac); the viral vectors vaccines Ad-RTS-hIL-12 (INXN-2001, e.g., produced by Ziopharm), ETBX-011 (Ad5 [E1-, E2b-]-CEA(6D), e.g., produced by Etubics), Cavatak (Coxsackievirus A21, e.g., produced by Viralytics), JX-594 (e.g., produced by Jennerex Biopharmaceuticals/Transgene), ColoAd1 (e.g., produced by PsiOxus Therapeutics), GL-ONC1 (e.g., produced by Genelux) and ONCOS-102 (CGTG-102, e.g., produced by Oncos Therapeutics); the bacterial vectors vaccines CRS-207 (an attenuated form of Listeria monocytogenes genetically modified to express the tumor associated antigen mesothelin, e.g., produced by Aduro BioTech/BioSante Pharmaceuticals) and ADU-623 (a live-attenuated, double-deleted strain of the Gram-positive bacterium Listeria monocytogenes encoding a mutant form of the tumor-associated antigens, epidermal growth factor receptor (EGFRvIII) and the cancer/testis antigen NY-ESO-1, e.g., produced by Aduro BioTech); the tumor cell vaccines Dorgenmeltucel-L (e.g., produced by NewLink Genetics), HyperAcute Prostate (e.g., produced by NewLink Genetics), FANG vaccine (e.g., produced by Gradalis), HPV vaccine and MGN-1601 (e.g., produced by Mologen AG); and Tarmogens such as GI-4000 (e.g., produced by Globelmmune) heat-inactivated S. cerevisiae yeast expressing a unique combination of three Ras mutations, collectively targeting seven of the most common Ras mutations observed in human cancers.
[0177] According to a specific embodiment, the vaccine is a human papiloma virus (HPV, e.g., HPV 16 vaccine) typically targeting E6 and/or E7. The vaccine may be a preventive vaccine or a therapeutic vaccine. Detailed examples of HPV vaccines which can be used along with the present teachings can be found in Lin et al. J Formos Med Assoc. 2010 January; 109(1): 4-24; and Rice et al. Cancer Gene Therapy 22, 454-462.
[0178] According to a specific embodiment, the preventive vaccines utilize the capsid proteins L1 and L2 as target antigens, inducing antibodies to neutralize and prevent entry of HPV into cells. Expression of recombinant L1, the major component of the capsid, in various cell types results in spontaneous assembly of virus-like particles (VLPs), which are immunologically and morphologically similar to HPV virions.
[0179] According to another specific embodiment, the vaccines is Gardasil.TM. or Cervarix.TM. Gardasil is a quadrivalent vaccine containing recombinant L1 VLPs for HPV genotypes 6, 11, 16 and 18 whereas the bivalent vaccine Cervarix contains L1 VLPs for HPV-16 and 18.
[0180] According to another specific embodiment, the vaccine is a monovalent HPV-16 L1 vaccine with an aluminium hydroxyphosphate sulfate adjuvant.
[0181] Exemplary, non-limiting, therapeutic vaccines comprise HPV E6 and E7 antigens. These represent ideal targets for therapeutic vaccines since these are constitutively expressed in HPV-infected cells and not healthy cells. E6 and E7 are essential to the induction and maintenance of cellular transformation, and thus are unlikely to be lost in an attempt to evade the immune system.
[0182] According to a specific embodiment, the therapeutic vaccines target E6 and/or E7.
[0183] Therapeutic vaccines typically include:
[0184] Live vector vaccines--Vector-based vaccines can deliver the antigens E6 and E7 to the dendritic cells (DCs), stimulating antigen expression through MHC class I (to CD8+ cytotoxic T cells) and MHC class II (to CD4+ helper T cells). Viral vectors used adenovirus, adeno-associated virus, vaccinia virus and alphaviruses, such as the Venezuelan equine encephalitis (VEE) virus;
[0185] Peptide/protein-based vaccines--Administered peptides and proteins derived from HPV antigens (e.g., E6 and/or E7) are taken up by DCs, processed and expressed via MHC II and/or I to the appropriate CD4+/CD8+ T cells;
[0186] Cell-based vaccines--dendritic cell-based or tumor cell based vaccines; and
[0187] Nucleic acid-based vaccines e.g., naked DNA based vaccines (e.g., ZYC-101 and ZYC-101a), naked RNA replicon vaccines.
[0188] The vaccine may be administered with an adjuvant, such as, but not limited to, incomplete Freund's Adjuvant, aluminum salts, oil-in-water emulsion (MF59), and nontoxic derivatives from Salmonella (MPL), water-in-oil emulsions (e.g. Montanide ISA 51 and ISA 720), saponins (e.g. ISCOM, QS-21, ASO1 and AS02), SRL-172, histamine dihydrochloride, thymocartin, Tio-TEPA, monophosphoryl-lipid A/micobacteria compositions, alum, Montanide ISA, Ribi Adjuvant System, TiterMax adjuvant, syntex adjuvant formulations, immune-stimulating complexes (ISCOMs), Gerbu.sup.R adjuvant, CpG oligodeoxynucleotides, lipopolysaccharide, and polyinosinic:polycytidylic acid.
[0189] As used herein, the phrase "anti-cancer reactive mononuclear blood cells (MNBCs)" refers to blood cells having a single nucleus. According to a specific embodiment MNBCs include lymphocytes, monocytes and dendritic cells (DCs) which are capable of eliciting an immune response (e.g. T cell, NK cell) against a cancerous cell.
[0190] According to specific embodiments the MNBCs are selected from the group consisting of dendritic cells (DCs), T cells, B cells, NK cells and NKT cells.
[0191] As used herein, the term "T cell" refers to a differentiated lymphocyte with a CD3.sup.+, T cell receptor (TCR).sup.+ having either CD4.sup.+ or CD8.sup.+ phenotype. The T cell may be either an effector or a regulatory T cells.
[0192] As used herein, the term "effector T cells" refers to a T cell that activates or directs other immune cells e.g. by producing cytokines or has a cytotoxic activity e.g., CD4+, Th1/Th2, CD8+ cytotoxic T lymphocyte.
[0193] As used herein, the term "regulatory T cell" or "Treg" refers to a T cell that negatively regulates the activation of other T cells, including effector T cells, as well as innate immune system cells. Treg cells are characterized by sustained suppression of effector T cell responses. According to a specific embodiment, the Treg is a CD4+CD25+Foxp3+ T cell.
[0194] According to specific embodiments, MNBCs comprise T cells.
[0195] As used herein the term "B cell" refers to a lymphocyte with a B cell receptor (BCR)+, CD19+ and or B220+ phenotype. B cells are characterized by their ability to bind a specific antigen and elicit a humoral response.
[0196] As used herein the term "natural killer cell (NK cell)" refers to a differentiated lymphocyte with a CD16+ CD56+ and/or CD57+ TCR- phenotype. NKs are characterized by their ability to bind to and kill cells that fail to express "self" MHC/HLA antigens by the activation of specific cytolytic enzymes, the ability to kill tumor cells or other diseased cells that express a ligand for NK activating receptors, and the ability to release protein molecules called cytokines that stimulate or inhibit the immune response.
[0197] As used herein the term "NKT cells" refers to a specialized population of T cells that express a semi-invariant .alpha..beta. T-cell receptor, but also express a variety of molecular markers that are typically associated with NK cells, such as NK1.1. NKT cells include NK1.1.sup.+ and NK1.1.sup.-, as well as CD4.sup.+, CD4.sup.-, CD8.sup.+ and CD8.sup.- cells. The TCR on NKT cells is unique in that it recognizes glycolipid antigens presented by the MHC I-like molecule CD1d. NKT cells can have either protective or deleterious effects due to their abilities to produce cytokines that promote either inflammation or immune tolerance.
[0198] As used herein the term "dendritic cell (DC)" refers to an antigen presenting cell capable of sensitizing HLA-restricted T cells. DCs include DCs derived from bone marrow hematopoietic cells such as plasmacytoid dendritic cells, myeloid dendritic cells, Langerhans cells and interdigitating cells; and follicular DCs. Dendritic cells may be recognized by function, or by phenotype, particularly by cell surface phenotype. These cells are characterized by their distinctive morphology having veil-like projections on the cell surface, intermediate to high levels of surface HLA-class II expression and ability to present antigen to T cells, particularly to naive T cells (See Steinman R, et al., Ann. Rev. Immunol. 1991; 9:271-196.). Typically, cell surface phenotype of DCs include CD1a+, CD4+, CD86+, or HLA-DR. The term DCs encompasses both immature and mature DCs.
[0199] According to specific embodiments, the MNBCs comprise dendritic cells.
[0200] According to specific embodiments the MNBCs do not comprise CD34+ hematopoietic stem/progenitor cells.
[0201] The MNBCs used according to specific embodiments of the present invention may be autologous or non-autologous; they can be syngeneic or non-syngeneic: allogeneic or xenogeneic. Each possibility represents a separate embodiment of the present invention.
[0202] As used herein, the term "autologous" means that the donor subject is the recipient subject. Thus, in autologous transplantation the cells have been removed and re-introduced e.g., re-infused to the subject.
[0203] As used herein, the term "non-autologous" means that the donor subject is not the recipient subject.
[0204] As used herein, the term "syngeneic" means that the donor subject is essentially genetically identical with the recipient subject. Examples of syngeneic transplantation include transplantation of cells derived from the subject (also referred to in the art as "autologous"), a clone of the subject, or a homozygotic twin of the subject.
[0205] As used herein, the term "allogeneic" means that the donor is of the same species as the recipient, but which is substantially non-clonal with the recipient. Typically, outbred, non-zygotic twin mammals of the same species are allogeneic with each other. It will be appreciated that an allogeneic donor may be HLA identical or HLA non-identical with respect to the subject.
[0206] As used herein, the term "xenogeneic" means that the donor subject is from a different species relative to the recipient subject.
[0207] Methods of obtaining and/or developing anti-cancer reactive MNBCs that can be used according to specific embodiments of the present invention are well known in the art (see e.g. Hildebrandt et al. Cytotherapy. 2014 16(40): S120-S129; Leen et al. Immunol Rev. 2014; 258(1): 12-29; Qian et al. Journal of Immunology Research Volume 2014, Article ID 525913, 9 pages; Martelli et al. Blood. 2014; 123(7):967-973; Ophir and Reisner Front Immunol. 2012 3:93; Lask et al. Blood. 2013; 121(15):3033-3040; Galluzzi et al. Oncoimmunology. 2012; 1(3):306-315; Itzhaki et al. Immunotherapy. 2013; 5(1):79-90; Rosenberg et al. Clinical cancer research: an official journal of the American Association for Cancer Research. 2011; 17(13):4550-4557; Bouquie, et al. Cancer Immunol Immunother. 2009; 58:553-66; Lu et al. Journal of immunology. 2013; 190(12):6034-6042; Robbins et al. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2011; 29(7):917-924; June J. Clin Invest. (2007) 117(6): 1466-1476; Greenberg, R. & Riddell, S. (1999) Science 285: 546-51); and Shtivelman et al. Oncotarget. 2014 Apr. 15; 5(7):1701-52; the contents of each of which are fully incorporated herein by reference).
[0208] Thus, for example, a peripheral blood sample is collected from a subject by methods well known in the art such as drawing whole blood from the subject and collection in a container containing an anti-coagulant (e.g. heparin or citrate); and apheresis. Following, at least one type of MNBCs is purified from the peripheral blood and enriched for at least one type of MNBCs effective for the treatment of cancer. There are several methods and reagents known to those skilled in the art for purifying MNBCs from whole blood such as leukapheresis, sedimentation, density gradient centrifugation (e.g. ficoll), centrifugal elutriation, fractionation, chemical lysis of e.g. red blood cells (e.g. by ACK), selection of specific cell types using cell surface markers (using e.g. FACS sorter or magnetic cell separation techniques), and depletion of specific cell types by methods such as eradication (e.g. killing) with specific antibodies or by affinity based purification based on negative selection (using e.g. magnetic cell separation techniques, FACS sorter and/or capture ELISA labeling). Such methods are described for example in THE HANDBOOK OF EXPERIMENTAL IMMUNOLOGY, Volumes 1 to 4, (D. N. Weir, editor) and FLOW CYTOMETRY AND CELL SORTING (A. Radbruch, editor, Springer Verlag, 2000).
[0209] Enriching for an anti-cancer reactive MNBCs can be effected by any method known in the art (see e.g. Hildebrandt et al. Cytotherapy. 2014 16(40): S120-S129; Leen et al. Immunol Rev. 2014; 258(1): 12-29; Qian et al. Journal of Immunology Research Volume 2014, Article ID 525913, 9 pages; Palucka and Banchereau Immunity. 2013; 39(1): 38-48; and Pizzurro and Barrio Front Immunol. 2015 Mar. 3; 6:91) and include for example, activating the specifically desired population of cells; promoting presentation of a specific antigen leading to enrichment of a population of antigen presenting cells presenting the specific antigen and/or activation and/or proliferation of antigen-specific population of cells (e.g. T cells); promoting presentation of a specific receptor leading to enrichment of a population of antigen-specific population of cells (e.g. T cells, B cells).
[0210] As a non-limiting example, for the generation of T cells that can be used for adoptive T cells transfer for cancer therapy the peripheral blood or purified population thereof comprising CD3+, CD4+ or CD8+ T cells or tumor-associated lymphocytes (TALs) selected for T-cell receptor (TCR) specificity (see e.g. Cancer Immunol Immunother. 2009; 58: 553-66) is activated and expanded ex-vivo by incubation with mature DCs preloaded with tumor antigens or transfected with mRNA coding for cancer antigens. Alternatively, the peripheral blood or a purified population thereof comprising both APCs (e.g. DCs) and T cells (e.g. CD3+, CD4+ or CD8+ T cells or TALs selected for TCR specificity) is contacted with tumor antigens or transfected with mRNA coding for cancer antigens under conditions which allow capturing and presentation of the cancer antigen by the APC leading to activation and expansion of anti-cancer antigen-specific T cells.
[0211] A specific non-limiting example of an autologous T cell therapy that can be used according to some embodiments of the present invention is C ntego tumor-infiltrating lymphocytes (TILs, produced by Lion Biotechnologies).
[0212] Other methods of expanding and processing T cells for anti-cancer adoptive cell transfer include redirecting T cell specificity by promoting presentation of an anti-cancer receptor by way of transducing with a T cell receptor (TCR) or a chimeric antigen receptor (CAR).
[0213] According to specific embodiments, the MNBCs comprise T cells transduced with a T cell receptor (TCR) or a chimeric antigen receptor (CAR).
[0214] As used herein "transduction with a TCR" refers to cloning of variable .alpha.- and .beta.-chains are from T cells with specificity against a cancer antigen presented in the context of MHC. Method of transducing with a TCR are known in the art and are disclosed e.g. in Nicholson et al. Adv Hematol. 2012; 2012:404081; Wang and Riviere Cancer Gene Ther. 2015 March; 22(2):85-94); and Lamers et al., Cancer Gene Therapy (2002) 9, 613-623.
[0215] As used herein "transducing with a CAR" refers to cloning of a nucleic acid sequence encoding a chimeric antigen receptor (CAR), wherein the CAR comprises an antigen recognition moiety and a T-cell activation moiety. A chimeric antigen receptor (CAR) is an artificially constructed hybrid protein or polypeptide containing an antigen binding domain of an antibody (e.g., a single chain variable fragment (scFv)) linked to T-cell signaling or T-cell activation domains. Method of transducing with a CAR are known in the art and are disclosed e.g. in Davila et al. Oncoimmunology. 2012 Dec. 1; 1(9):1577-1583; Wang and Riviere Cancer Gene Ther. 2015 March; 22(2):85-94); and Maus et al. Blood. 2014 Apr. 24; 123(17):2625-35.
[0216] Another non-limiting example is the generation of dendritic cells vaccine that can be used according to some embodiments of the present invention.
[0217] Typically, for the generation of an anti-cancer DC-based vaccine DCs are expanded ex vivo and contacted with a cancer antigen or a cancer cell lysate to thereby induce presentation of the cancer antigen (see e.g. Nestle, F. et al. (1998) Nature Medicine 4: 328-332). Alternatively, promoting presentation of a cancer antigen by a DC comprises transfecting the DC with a DNA, cDNA or an mRNA coding for a cancer antigen. Non-limiting examples of cancer antigens include MAGE-AI, MAGE-A2, MAGE-A3, MAGE-A4, MAGE-AS, MAGE-A6, MAGE-A7, MAGE-AS, MAGE-A9, MAGE-AIO, MAGE-All, MAGE-A12, GAGE-I, GAGE-2, GAGE-3, GAGE-4, GAGE-5, GAGE-6, GAGE-7, GAGE-8, BAGE-1, RAGE-1, LB33/MUM-1, PRAME, NAG, MAGE-Xp2 (MAGE-B2), MAGE-Xp3 (MAGE-B3), MAGE-Xp4 (MAGE-B4), MAGE-Cl/CT7, MAGE-C2, NY-ESO-1, LAGE-1, SSX-1, SSX-2(HOM-MEL-40), SSX-3, SSX-4, SSX-5, SCP-1 and XAGE, melanocyte differentiation antigens, p53, ras, CEA, MUCI, PMSA, PSA, tyrosinase, Melan-A, MART-I, gplOO, gp75, alphaactinin-4, Bcr-Abl fusion protein, Casp-8, beta-catenin, cdc27, cdk4, cdkn2a, coa-1, dek-can fusion protein, EF2, ETV6-AML1 fusion protein, LDLR-fucosyltransferaseAS fusion protein, HLA-A2, HLA-All, hsp70-2, KIAA0205, Mart2, Mum-2, and 3, neo-PAP, myosin class I, OS-9, pml-RAR alpha fusion protein, PTPRK, K-ras, N-ras, Triosephosphate isomerase, GnTV, Herv-K-mel, NA-88, SP17, and TRP2-Int2, (MART-I), E2A-PRL, H4-RET, IGH-IGK, MYL-RAR, Epstein Barr virus antigens, EBNA, human papillomavirus (HPV) antigens E6 and E7, TSP-180, MAGE-4, MAGE-5, MAGE-6, p185erbB2, p1SOerbB-3, c-met, nm-23H1, PSA, TAG-72-4, CA 19-9, CA 72-4, CAM 17.1, NuMa, K-ras, alpha.-fetoprotein, 13HCG, BCA225, BTAA, CA 125, CA 15-3 (CA 27.29\BCAA), CA 195, CA 242, CA-50, CAM43, CD68\KP1, CO-029, FGF-5, 0250, Ga733 (EpCAM), HTgp-175, M344, MA-50, MG7-Ag, MOV18, NB\170K, NYCO-I, RCASI, SDCCAG16, TA-90 (Mac-2 binding protein\cyclophilin C-associated protein), TAAL6, TAG72, TLP, TPS, tyrosinase related proteins, TRP-1, or TRP-2 and tumor derived heat shock proteins.
[0218] Other tumor antigens that may be expressed include out-of-frame peptide-MHC complexes generated by the non-AUG translation initiation mechanisms employed by "stressed" cancer cells (Malarkannan et al. Immunity 1999).
[0219] Other tumor antigens that may be expressed are well-known in the art (see for example WO00/20581; Cancer Vaccines and Immunotherapy (2000) Eds Stern, Beverley and Carroll, Cambridge University Press, Cambridge). The sequences of these tumor antigens are readily available from public databases but are also found in WO 1992/020356, WO 1994/005304, WO 1994/023031, WO 1995/020974, WO 1995/023874 & WO 1996/026214.
[0220] For EBV-associated lymphoma, EBV-specific antigens can be used as the cancer antigen. Other cancer antigens may include the proteins from viruses implicated in human cancers such a Human Papilloma Viruses (HPV), Hepatitis Viruses (HBV and HCV) and Kaposi's Herpes Sarcoma Virus (KHSV).
[0221] Alternatively or additionally a tumor antigen may be identified using cancer cells obtained from the subject by e.g. biopsy.
[0222] DCs can also be fused directly to tumor cells for the purposes of immunization (see e.g. Kugler, A. et al. (2000) Nature Medicine 6:332-336).
[0223] Specific non-limiting examples of DC-based vaccines that can be used according to specific embodiments of the present invention include: BPX-201 (produced by Bellicum Pharmaceuticals), AV-0113 (Trivax, produced by Activartis Biotech), ICT-107 (produced by ImmunoCellular Therapeutics) and Ad.p53-DC vaccine (produced by Medvax).
[0224] The anti-cancer reactive MNBCs used according to some embodiments of the present invention can be freshly isolated cells; stored e.g., cryopreserved (i.e. frozen) at e.g. liquid nitrogen temperature cells; and cell lines.
[0225] According to specific embodiments the MNBCs are derived from a subject not treated with a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof.
[0226] According to specific embodiments, the anti-cancer agent is a cytokine capable of inducing activation and/or proliferation of a T cell. Non-limiting examples of cytokines capable of inducing activation and/or proliferation of a T cell include, but are not limited to IL-2, IFN.alpha., IL-12, IFN-gamma, TNF-.alpha., IL-15, IL-6 and IL-1, IL-21.
[0227] According to specific embodiments, the cytokine is selected from the group consisting of IFN.alpha., IFN.gamma., IL-1, IL-2, IL-6, IL-12, IL-15, IL-21 and TNF.alpha..
[0228] According to other specific embodiments, the cytokine is selected from the group consisting of IFN.alpha., IL-2, IL-12, IL-21 and IL-15.
[0229] Specific non-limiting examples of cytokines and cytokines agonists that can be used according to some embodiments of the invention include:
[0230] IL-2 (produced by Roche); IL21 (produced by BMY); ALT-803 (IL15 superagonist combined with a soluble IL-15a receptor, produced by Altor Bioscience); Darleukin (L19-IL2, human IL-2 conjugated with an antibody (L19) that is specific to the EDB region of fibronectin, produced by Philogen); Denenicokin [BMS-982470, a recombinant human peptide homologous to IL-21, produced by Bristol-Myers Squibb (ZymoGenetics)]; and Immunopulse (delivery of DNA-based IL-12 leading to localized expression of IL-12 in the tumor microenvironment, produced by Oncosec Medical.
[0231] According to specific embodiments the anti-cancer agent is an immune-check point regulator.
[0232] As used herein the term "immune-check point regulator" refers to a molecule that modulates the activity of one or more immune-check point proteins in an agonistic or antagonistic manner resulting in recruitment of an immune cell to elicit an immune activity against a cancer cell. The immune-check point regulator may bind directly the immune-check point or through an intermediary molecule, it can modulate the activity of a specific immune-check point protein with no cross reactivity with other immune-check point proteins or modulate the activity of at least 2, at least 3, at least 4 immune-check point proteins; each possibility represents a separate embodiment of the present invention.
[0233] As used herein the term "activation" refers to the process of stimulating an immune cell (e.g. T cell, NK cell, B cell) that results in cellular proliferation, maturation, cytokine production and/or induction of regulatory or effector functions.
[0234] As used herein the term "immune-check point protein" refers to an antigen independent protein that modulates an immune cell response (i.e. activation or function). Immune-check point proteins can be either co-stimulatory proteins [i.e. positively regulating an immune cell activation or function by transmitting a co-stimulatory secondary signal resulting in activation of an immune cell] or inhibitory proteins (i.e. negatively regulating an immune cell activation or function by transmitting an inhibitory signal resulting in suppressing activity of an immune cell).
[0235] According to specific embodiments, the immune-check point protein regulates activation or function of a T cell.
[0236] Numerous check-point proteins are known in the art and include, but not limited to, PD1, PDL-1, B7H2, B7H3, B7H4, BTLA-4, HVEM, CTLA-4, CD80, CD86, LAG-3, TIM-3, KIR, IDO, CD19, OX40, OX40L, 4-1BB (CD137), 4-1BBL, CD27, CD70, CD40, CD40L, GITR, CD28, ICOS (CD278), ICOSL, VISTA and adenosine A2a receptor.
[0237] Methods of determining signaling of a stimulatory or inhibitory signal are well known in the art and include, but are not limited to, binding assay using e.g. BiaCore, HPLC or flow cytometry, enzymatic activity assays such as kinase activity assays, and expression of molecules involved in the signaling cascade using e.g. PCR, Western blot, immunoprecipitation and immunohistochemistry. Additionally or alternatively, determining transmission of a signal (co-stimulatory or inhibitory) can be effected by evaluating immune cell activation or function. Methods of evaluating immune cell activation or function are well known in the art and include, but are not limited to, proliferation assays such as BRDU and thymidine incorporation, cytotoxicity assays such as chromium release, cytokine secretion assays such as intracellular cytokine staining ELISPOT and ELISA, expression of activation markers such as CD25, CD69 and CD69 using flow cytometry.
[0238] According to specific embodiments, determining the signaling activity is effected in-vitro or ex-vivo e.g. in a mixed lymphocyte reaction (MLR).
[0239] For the same culture conditions the signaling activity or the immune cell activation or function are generally expressed in comparison to the signaling, activation or function in a cell of the same species but not contacted with the immune-check point regulator or contacted with a vehicle control, also referred to as control.
[0240] According to specific embodiments the immune-check point regulator is selected from the group consisting of an antibody, a peptide and a small molecule as further described in details hereinbelow.
[0241] Depending on the immune-check point protein (i.e. co-stimulatory or inhibitory) the immune-check point regulator can be an agonist or antagonist.
[0242] According to specific embodiment the immune-check point regulator is an antagonist.
[0243] As used herein the term "antagonist" refers to a molecule that prevents and/or inhibits the biological function and/or expression of an immune-check point protein.
[0244] According to specific embodiments, the antagonist prevents and/or inhibits the suppressive effect of an immune-check point protein on an immune cell (e.g. T cells).
[0245] According to specific embodiments, the antagonist prevents and/or inhibits signaling to an immune cell (e.g. T cell) by an immune-check point protein.
[0246] The molecule may be a reversible or an irreversible antagonist.
[0247] According to specific embodiments, the antagonist completely prevents the biological function (e.g. signal transduction) of the immune-check point protein.
[0248] According to other specific embodiments, the antagonist inhibits the biological function (e.g. signal transduction) of the immune-check point protein e.g., as detected by e.g. kinase activity, proliferation assay, cytotoxicity assay or cytokine secretion assay. The reduction may be by at least a 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95% or at least 99% as compared to same in the absence of the antagonist.
[0249] Preventing and/or inhibiting the biological function of an immune-check point protein can be effected at the protein level (e.g., antibodies, small molecules, inhibitory peptides, enzymes that cleave the polypeptide, aptamers and the like) but may also be effected at the genomic (e.g. homologous recombination and site specific endonucleases) and/or the transcript level using a variety of molecules which interfere with transcription and/or translation (e.g., RNA silencing agents) of an inhibitory immune-check point protein.
[0250] Non limiting examples of agents that can function as antagonists are described in details hereinbelow.
[0251] Suppressing Biological Function at the Polypeptide Level
[0252] According to specific embodiments, the antagonistic agent is an antibody.
[0253] According to specific embodiments the antagonistic antibody is capable of specifically binding an inhibitory immune-check point protein. According to specific embodiments, the antagonistic antibody specifically binds at least one epitope of an inhibitory immune-check point protein.
[0254] As used herein, the term "epitope" refers to any antigenic determinant on an antigen to which the paratope of an antibody binds. Epitopic determinants usually consist of chemically active surface groupings of molecules such as amino acids or carbohydrate side chains and usually have specific three dimensional structural characteristics, as well as specific charge characteristics.
[0255] The term "antibody" as used in this invention includes intact molecules as well as functional fragments thereof, such as Fab, F(ab')2, Fv, scFv, dsFv, or single domain molecules such as VH and VL that are capable of binding to an epitope of an antigen.
[0256] The antibody may be mono-specific (capable of recognizing one epitope or protein), bi-specific (capable of binding two epitopes or proteins) or multi-specific (capable of recognizing multiple epitopes or proteins).
[0257] Suitable antibody fragments for practicing some embodiments of the invention include a complementarity-determining region (CDR) of an immunoglobulin light chain (referred to herein as "light chain"), a complementarity-determining region of an immunoglobulin heavy chain (referred to herein as "heavy chain"), a variable region of a light chain, a variable region of a heavy chain, a light chain, a heavy chain, an Fd fragment, and antibody fragments comprising essentially whole variable regions of both light and heavy chains such as an Fv, a single chain Fv Fv (scFv), a disulfide-stabilized Fv (dsFv), an Fab, an Fab', and an F(ab')2.
[0258] As used herein, the terms "complementarity-determining region" or "CDR" are used interchangeably to refer to the antigen binding regions found within the variable region of the heavy and light chain polypeptides. Generally, antibodies comprise three CDRs in each of the VH (CDR HI or HI; CDR H2 or H2; and CDR H3 or H3) and three in each of the VL (CDR LI or LI; CDR L2 or L2; and CDR L3 or L3).
[0259] The identity of the amino acid residues in a particular antibody that make up a variable region or a CDR can be determined using methods well known in the art and include methods such as sequence variability as defined by Kabat et al. (See, e.g., Kabat et al., 1992, Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, NIH, Washington D.C.), location of the structural loop regions as defined by Chothia et al. (see, e.g., Chothia et al., Nature 342:877-883, 1989.), a compromise between Kabat and Chothia using Oxford Molecular's AbM antibody modeling software (now Accelrys.RTM., see, Martin et al., 1989, Proc. Natl Acad Sci USA. 86:9268; and world wide web site www(dot)bioinf-org(dot)uk/abs), available complex crystal structures as defined by the contact definition (see MacCallum et al., J. Mol. Biol. 262:732-745, 1996), the "conformational definition" (see, e.g., Makabe et al., Journal of Biological Chemistry, 283:1156-1166, 2008) and IMGT [Lefranc M P, et al. (2003) IMGT unique numbering for immunoglobulin and T cell receptor variable domains and Ig superfamily V-like domains. Dev Comp Immunol 27: 55-77].
[0260] As used herein, the "variable regions" and "CDRs" may refer to variable regions and CDRs defined by any approach known in the art, including combinations of approaches.
[0261] Functional antibody fragments comprising whole or essentially whole variable regions of both light and heavy chains are defined as follows:
[0262] (i) Fv, defined as a genetically engineered fragment consisting of the variable region of the light chain (VL) and the variable region of the heavy chain (VH) expressed as two chains;
[0263] (ii) single chain Fv ("scFv"), a genetically engineered single chain molecule including the variable region of the light chain and the variable region of the heavy chain, linked by a suitable polypeptide linker as a genetically fused single chain molecule;
[0264] (iii) disulfide-stabilized Fv ("dsFv"), a genetically engineered antibody including the variable region of the light chain and the variable region of the heavy chain, linked by a genetically engineered disulfide bond;
[0265] (iv) Fab, a fragment of an antibody molecule containing a monovalent antigen-binding portion of an antibody molecule which can be obtained by treating whole antibody with the enzyme papain to yield the intact light chain and the Fd fragment of the heavy chain which consists of the variable and CH1 domains thereof;
[0266] (v) Fab', a fragment of an antibody molecule containing a monovalent antigen-binding portion of an antibody molecule which can be obtained by treating whole antibody with the enzyme pepsin, followed by reduction (two Fab' fragments are obtained per antibody molecule);
[0267] (vi) F(ab') 2, a fragment of an antibody molecule containing a monovalent antigen-binding portion of an antibody molecule which can be obtained by treating whole antibody with the enzyme pepsin (i.e., a dimer of Fab' fragments held together by two disulfide bonds); and
[0268] (vii) Single domain antibodies or nanobodies are composed of a single VH or VL domains which exhibit sufficient affinity to the antigen.
[0269] The antibody may be monoclonal or polyclonal.
[0270] Methods of producing polyclonal and monoclonal antibodies as well as fragments thereof are well known in the art (See for example, Harlow and Lane, Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory, New York, 1988, incorporated herein by reference).
[0271] Antibody fragments according to some embodiments of the invention can be prepared by proteolytic hydrolysis of the antibody or by expression in E. coli or mammalian cells (e.g. Chinese hamster ovary cell culture or other protein expression systems) of DNA encoding the fragment. Antibody fragments can be obtained by pepsin or papain digestion of whole antibodies by conventional methods. For example, antibody fragments can be produced by enzymatic cleavage of antibodies with pepsin to provide a 5S fragment denoted F(ab')2. This fragment can be further cleaved using a thiol reducing agent, and optionally a blocking group for the sulfhydryl groups resulting from cleavage of disulfide linkages, to produce 3.5S Fab' monovalent fragments. Alternatively, an enzymatic cleavage using pepsin produces two monovalent Fab' fragments and an Fc fragment directly. These methods are described, for example, by Goldenberg, U.S. Pat. Nos. 4,036,945 and 4,331,647, and references contained therein, which patents are hereby incorporated by reference in their entirety. See also Porter, R. R. [Biochem. J. 73: 119-126 (1959)]. Other methods of cleaving antibodies, such as separation of heavy chains to form monovalent light-heavy chain fragments, further cleavage of fragments, or other enzymatic, chemical, or genetic techniques may also be used, so long as the fragments bind to the antigen that is recognized by the intact antibody.
[0272] Fv fragments comprise an association of VH and VL chains. This association may be noncovalent, as described in Inbar et al. [Proc. Nat'l Acad. Sci. USA 69:2659-62 (19720]. Alternatively, the variable chains can be linked by an intermolecular disulfide bond or cross-linked by chemicals such as glutaraldehyde. Preferably, the Fv fragments comprise VH and VL chains connected by a peptide linker. These single-chain antigen binding proteins (sFv) are prepared by constructing a structural gene comprising DNA sequences encoding the VH and VL domains connected by an oligonucleotide. The structural gene is inserted into an expression vector, which is subsequently introduced into a host cell such as E. coli. The recombinant host cells synthesize a single polypeptide chain with a linker peptide bridging the two V domains. Methods for producing sFvs are described, for example, by [Whitlow and Filpula, Methods 2: 97-105 (1991); Bird et al., Science 242:423-426 (1988); Pack et al., Bio/Technology 11:1271-77 (1993); and U.S. Pat. No. 4,946,778, which is hereby incorporated by reference in its entirety.
[0273] Another form of an antibody fragment is a peptide coding for a single complementarity-determining region (CDR). CDR peptides ("minimal recognition units") can be obtained by constructing genes encoding the CDR of an antibody of interest. Such genes are prepared, for example, by using the polymerase chain reaction to synthesize the variable region from RNA of antibody-producing cells. See, for example, Larrick and Fry [Methods, 2: 106-10 (1991)].
[0274] It will be appreciated that for human therapy or diagnostics, humanized antibodies are preferably used. Humanized forms of non-human (e.g., murine) antibodies are chimeric molecules of immunoglobulins, immunoglobulin chains or fragments thereof (such as Fv, Fab, Fab', F(ab').sub.2 or other antigen-binding subsequences of antibodies) which contain minimal sequence derived from non-human immunoglobulin. Humanized antibodies include human immunoglobulins (recipient antibody) in which residues form a complementary determining region (CDR) of the recipient are replaced by residues from a CDR of a non-human species (donor antibody) such as mouse, rat or rabbit having the desired specificity, affinity and capacity. In some instances, Fv framework residues of the human immunoglobulin are replaced by corresponding non-human residues. Humanized antibodies may also comprise residues which are found neither in the recipient antibody nor in the imported CDR or framework sequences. In general, the humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the CDR regions correspond to those of a non-human immunoglobulin and all or substantially all of the FR regions are those of a human immunoglobulin consensus sequence. The humanized antibody optimally also will comprise at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin [Jones et al., Nature, 321:522-525 (1986); Riechmann et al., Nature, 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol., 2:593-596 (1992)].
[0275] Methods for humanizing non-human antibodies are well known in the art.
[0276] Generally, a humanized antibody has one or more amino acid residues introduced into it from a source which is non-human. These non-human amino acid residues are often referred to as import residues, which are typically taken from an import variable domain. Humanization can be essentially performed following the method of Winter and co-workers [Jones et al., Nature, 321:522-525 (1986); Riechmann et al., Nature 332:323-327 (1988); Verhoeyen et al., Science, 239:1534-1536 (1988)], by substituting rodent CDRs or CDR sequences for the corresponding sequences of a human antibody. Accordingly, such humanized antibodies are chimeric antibodies (U.S. Pat. No. 4,816,567), wherein substantially less than an intact human variable domain has been substituted by the corresponding sequence from a non-human species. In practice, humanized antibodies are typically human antibodies in which some CDR residues and possibly some FR residues are substituted by residues from analogous sites in rodent antibodies.
[0277] Human antibodies can also be produced using various techniques known in the art, including phage display libraries [Hoogenboom and Winter, J. Mol. Biol., 227:381 (1991); Marks et al., J. Mol. Biol., 222:581 (1991)]. The techniques of Cole et al. and Boerner et al. are also available for the preparation of human monoclonal antibodies (Cole et al., Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, p. 77 (1985) and Boerner et al., J. Immunol., 147(1):86-95 (1991)]. Similarly, human antibodies can be made by introduction of human immunoglobulin loci into transgenic animals, e.g., mice in which the endogenous immunoglobulin genes have been partially or completely inactivated. Upon challenge, human antibody production is observed, which closely resembles that seen in humans in all respects, including gene rearrangement, assembly, and antibody repertoire. This approach is described, for example, in U.S. Pat. Nos. 5,545,807; 5,545,806; 5,569,825; 5,625,126; 5,633,425; 5,661,016, and in the following scientific publications: Marks et al., Bio/Technology 10: 779-783 (1992); Lonberg et al., Nature 368: 856-859 (1994); Morrison, Nature 368 812-13 (1994); Fishwild et al., Nature Biotechnology 14, 845-51 (1996); Neuberger, Nature Biotechnology 14: 826 (1996); and Lonberg and Huszar, Intern. Rev. Immunol. 13, 65-93 (1995).
[0278] Once antibodies are obtained, they may be tested for activity, for example via ELISA.
[0279] Another agent which can be used as antagonist with some embodiments of the invention is an aptamer. As used herein, the term "aptamer" refers to double stranded or single stranded RNA molecule that binds to specific molecular target, such as a protein. Various methods are known in the art which can be used to design protein specific aptamers. The skilled artisan can employ SELEX (Systematic Evolution of Ligands by Exponential Enrichment) for efficient selection as described in Stoltenburg R, Reinemann C, and Strehlitz B (Biomolecular engineering (2007) 24(4):381-403).
[0280] Another agent capable of being an antagonist would be any molecule which interferes with the immune-check point protein function (e.g. catalytic or interaction) by binding to and/or cleaving the immune-check point protein. Such molecules can be, but are not limited to, small molecules, inhibitory peptides, enzymes that cleave the immune-check point protein, adnectins, affibodies, avimers, anticalins, tetranectins, DARPins, and engineered Kunitz-type inhibitors wherein each possibility is a separate embodiment of the invention.
[0281] According to a specific embodiment, the antagonist is a small molecule.
[0282] According to a specific embodiment, the antagonist is a peptide molecule.
[0283] It will be appreciated that a non-functional analogue of at least a catalytic or binding portion of an inhibitory peptide can be also used as an antagonist.
[0284] Suppressing Biological Function at the Nucleic Acid Level
[0285] Down-regulation at the nucleic acid level is typically effected using a nucleic acid agent, having a nucleic acid backbone, DNA, RNA, mimetics thereof or a combination of same. The nucleic acid agent may be encoded from a DNA molecule or provided to the cell per se.
[0286] Thus, the antagonist of some embodiments of the invention can be an RNA silencing agent. As used herein, the phrase "RNA silencing" refers to a group of regulatory mechanisms [e.g. RNA interference (RNAi), transcriptional gene silencing (TGS), post-transcriptional gene silencing (PTGS), quelling, co-suppression, and translational repression] mediated by RNA molecules which result in the inhibition or "silencing" of the expression of a corresponding protein-coding gene. RNA silencing has been observed in many types of organisms, including plants, animals, and fungi.
[0287] As used herein, the term "RNA silencing agent" refers to an RNA which is capable of specifically inhibiting or "silencing" the expression of a target gene. In certain embodiments, the RNA silencing agent is capable of preventing complete processing (e.g., the full translation and/or expression) of an mRNA molecule through a post-transcriptional silencing mechanism. RNA silencing agents include non-coding RNA molecules, for example RNA duplexes comprising paired strands, as well as precursor RNAs from which such small non-coding RNAs can be generated. Exemplary RNA silencing agents include dsRNAs such as siRNAs, miRNAs and shRNAs.
[0288] In one embodiment, the RNA silencing agent is capable of inducing RNA interference.
[0289] In another embodiment, the RNA silencing agent is capable of mediating translational repression.
[0290] According to an embodiment of the invention, the RNA silencing agent is specific to the target RNA (i.e. an immune-check point protein) and does not cross inhibit or silence other targets or a splice variant which exhibits 99% or less global homology to the target gene, e.g., less than 98%, 97%, 96%, 95%, 94%, 93%, 92%, 91%, 90%, 89%, 88%, 87%, 86%, 85%, 84%, 83%, 82%, 81% global homology to the target gene; as determined by PCR, Western blot, Immunohistochemistry and/or flow cytometry.
[0291] RNA interference refers to the process of sequence-specific post-transcriptional gene silencing in animals mediated by short interfering RNAs (siRNAs).
[0292] Following is a detailed description on RNA silencing agents that can be used according to specific embodiments of the present invention.
[0293] DsRNA, siRNA and shRNA--
[0294] The presence of long dsRNAs in cells stimulates the activity of a ribonuclease III enzyme referred to as dicer. Dicer is involved in the processing of the dsRNA into short pieces of dsRNA known as short interfering RNAs (siRNAs). Short interfering RNAs derived from dicer activity are typically about 21 to about 23 nucleotides in length and comprise about 19 base pair duplexes.
[0295] The RNAi response also features an endonuclease complex, commonly referred to as an RNA-induced silencing complex (RISC), which mediates cleavage of single-stranded RNA having sequence complementary to the antisense strand of the siRNA duplex. Cleavage of the target RNA takes place in the middle of the region complementary to the antisense strand of the siRNA duplex.
[0296] Accordingly, some embodiments of the invention contemplate use of dsRNA to downregulate protein expression from mRNA.
[0297] According to one embodiment dsRNA longer than 30 bp are used. Various studies demonstrate that long dsRNAs can be used to silence gene expression without inducing the stress response or causing significant off-target effects--see for example [Strat et al., Nucleic Acids Research, 2006, Vol. 34, No. 13 3803-3810; Bhargava A et al. Brain Res. Protoc. 2004; 13:115-125; Diallo M., et al., Oligonucleotides. 2003; 13:381-392; Paddison P. J., et al., Proc. Natl Acad. Sci. USA. 2002; 99:1443-1448; Tran N., et al., FEBS Lett. 2004; 573:127-134].
[0298] According to some embodiments of the invention, dsRNA is provided in cells where the interferon pathway is not activated, see for example Billy et al., PNAS 2001, Vol. 98, pages 14428-14433. and Diallo et al., Oligonucleotides, Oct. 1, 2003, 13(5): 381-392. doi:10.1089/154545703322617069.
[0299] According to an embodiment of the invention, the long dsRNA are specifically designed not to induce the interferon and PKR pathways for down-regulating gene expression. For example, Shinagwa and Ishii [Genes & Dev. 17 (11): 1340-1345, 2003] have developed a vector, named pDECAP, to express long double-strand RNA from an RNA polymerase II (Pol II) promoter. Because the transcripts from pDECAP lack both the 5'-cap structure and the 3'-poly(A) tail that facilitate ds-RNA export to the cytoplasm, long ds-RNA from pDECAP does not induce the interferon response.
[0300] Another method of evading the interferon and PKR pathways in mammalian systems is by introduction of small inhibitory RNAs (siRNAs) either via transfection or endogenous expression.
[0301] The term "siRNA" refers to small inhibitory RNA duplexes (generally between 18-30 base pairs) that induce the RNA interference (RNAi) pathway.
[0302] Typically, siRNAs are chemically synthesized as 21mers with a central 19 bp duplex region and symmetric 2-base 3'-overhangs on the termini, although it has been recently described that chemically synthesized RNA duplexes of 25-30 base length can have as much as a 100-fold increase in potency compared with 21mers at the same location. The observed increased potency obtained using longer RNAs in triggering RNAi is suggested to result from providing Dicer with a substrate (27mer) instead of a product (21mer) and that this improves the rate or efficiency of entry of the siRNA duplex into RISC.
[0303] It has been found that position of the 3'-overhang influences potency of a siRNA and asymmetric duplexes having a 3'-overhang on the antisense strand are generally more potent than those with the 3'-overhang on the sense strand (Rose et al., 2005). This can be attributed to asymmetrical strand loading into RISC, as the opposite efficacy patterns are observed when targeting the antisense transcript.
[0304] The strands of a double-stranded interfering RNA (e.g., a siRNA) may be connected to form a hairpin or stem-loop structure (e.g., a shRNA). Thus, as mentioned, the RNA silencing agent of some embodiments of the invention may also be a short hairpin RNA (shRNA).
[0305] The term "shRNA", as used herein, refers to an RNA agent having a stem-loop structure, comprising a first and second region of complementary sequence, the degree of complementarity and orientation of the regions being sufficient such that base pairing occurs between the regions, the first and second regions being joined by a loop region, the loop resulting from a lack of base pairing between nucleotides (or nucleotide analogs) within the loop region. The number of nucleotides in the loop is a number between and including 3 to 23, or 5 to 15, or 7 to 13, or 4 to 9, or 9 to 11.
[0306] Some of the nucleotides in the loop can be involved in base-pair interactions with other nucleotides in the loop. Examples of oligonucleotide sequences that can be used to form the loop include 5'-CAAGAGA-3' and 5'-UUACAA-3' (International Patent Application Nos. WO2013126963 and WO2014107763). It will be recognized by one of skill in the art that the resulting single chain oligonucleotide forms a stem-loop or hairpin structure comprising a double-stranded region capable of interacting with the RNAi machinery.
[0307] Synthesis of RNA silencing agents suitable for use with some embodiments of the invention can be effected as follows. First, the inhibitory-check point mRNA sequence is scanned downstream of the AUG start codon for AA dinucleotide sequences. Occurrence of each AA and the 3' adjacent 19 nucleotides is recorded as potential siRNA target sites. Preferably, siRNA target sites are selected from the open reading frame, as untranslated regions (UTRs) are richer in regulatory protein binding sites. UTR-binding proteins and/or translation initiation complexes may interfere with binding of the siRNA endonuclease complex [Tuschl Chem Bio chem. 2:239-245]. It will be appreciated though, that siRNAs directed at untranslated regions may also be effective, as demonstrated for GAPDH wherein siRNA directed at the 5' UTR mediated about 90% decrease in cellular GAPDH mRNA and completely abolished protein level (www(dot)ambion(dot)com/techlib/tn/91/912(dot)html).
[0308] Second, potential target sites are compared to an appropriate genomic database (e.g., human, mouse, rat etc.) using any sequence alignment software, such as the BLAST software available from the NCBI server (www(dot)ncbi(dot)nlm(dot)nih(dot)gov/BLAST/). Putative target sites which exhibit significant homology to other coding sequences are filtered out.
[0309] Qualifying target sequences are selected as template for siRNA synthesis.
[0310] Preferred sequences are those including low G/C content as these have proven to be more effective in mediating gene silencing as compared to those with G/C content higher than 55%. Several target sites are preferably selected along the length of the target gene for evaluation. For better evaluation of the selected siRNAs, a negative control is preferably used in conjunction. Negative control siRNA preferably include the same nucleotide composition as the siRNAs but lack significant homology to the genome. Thus, a scrambled nucleotide sequence of the siRNA is preferably used, provided it does not display any significant homology to any other gene.
[0311] It will be appreciated that, and as mentioned hereinabove, the RNA silencing agent of some embodiments of the invention need not be limited to those molecules containing only RNA, but further encompasses chemically-modified nucleotides and non-nucleotides.
[0312] According to another embodiment the RNA silencing agent may be a miRNA.
[0313] The term "microRNA", "miRNA", and "miR" are synonymous and refer to a collection of non-coding single-stranded RNA molecules of about 19-28 nucleotides in length, which regulate gene expression. miRNAs are found in a wide range of organisms (viruses.fwdarw.humans) and have been shown to play a role in development, homeostasis, and disease etiology.
[0314] Below is a brief description of the mechanism of miRNA activity.
[0315] Genes coding for miRNAs are transcribed leading to production of a miRNA precursor known as the pri-miRNA. The pri-miRNA may form a hairpin with a stem and loop.
[0316] The hairpin structure of the pri-miRNA is recognized by Drosha, which is a RNase III endonuclease. Drosha typically recognizes terminal loops in the pri-miRNA and cleaves the pri-miRNA with a staggered cut typical of RNase III endonucleases yielding a pre-miRNA stem loop with a 5' phosphate and .about.2 nucleotide 3' overhang. The pre-miRNA is then actively transported from the nucleus to the cytoplasm by Ran-GTP and the export receptor Ex-portin-5.
[0317] The double-stranded stem or the 5' phosphate and 3' overhang at the base of the stem loop of the pre-miRNA is then recognized by Dicer, which is also an RNase III endonuclease. Dicer then cleaves off the terminal loop two helical turns away from the base of the stem loop leaving an additional 5' phosphate and .about.2 nucleotide 3' overhang. The resulting siRNA-like duplex, which may comprise mismatches, comprises the mature miRNA and a similar-sized fragment known as the miRNA*. miRNA* sequences may be found in libraries of cloned miRNAs but typically at lower frequency than the miRNAs.
[0318] Although initially present as a double-stranded species with miRNA*, the miRNA eventually becomes incorporated as a single-stranded RNA into a ribonucleoprotein complex known as the RNA-induced silencing complex (RISC) while the miRNA* is removed and degraded.
[0319] The RISC identifies target nucleic acids based on high levels of complementarity between the miRNA and the mRNA, especially by nucleotides 2-7 of the miRNA.
[0320] A number of studies have looked at the base-pairing requirement between miRNA and its mRNA target for achieving efficient inhibition of translation (reviewed by Bartel 2004, Cell 116-281). In mammalian cells, the first 8 nucleotides of the miRNA may be important (Doench & Sharp 2004 Genes Dev 2004-504). However, other parts of the microRNA may also participate in mRNA binding. Moreover, sufficient base pairing at the 3' can compensate for insufficient pairing at the 5' (Brennecke et al., 2005 PLoS 3-e85). Computation studies, analyzing miRNA binding on whole genomes have suggested a specific role for bases 2-7 at the 5' of the miRNA in target binding but the role of the first nucleotide, found usually to be "A" was also recognized (Lewis et at 2005 Cell 120-15). Similarly, nucleotides 1-7 or 2-8 were used to identify and validate targets by Krek et al. (2005, Nat Genet 37-495).
[0321] The target sites in the mRNA may be in the 5' UTR, the 3' UTR or in the coding region.
[0322] miRNAs may direct the RISC to downregulate gene expression by either of two mechanisms: mRNA cleavage or translational repression. The miRNA may specify cleavage of the mRNA if the mRNA has a certain degree of complementarity to the miRNA. When a miRNA guides cleavage, the cut is typically between the nucleotides pairing to residues 10 and 11 of the miRNA. Alternatively, the miRNA may repress translation if the miRNA does not have the requisite degree of complementarity to the miRNA.
[0323] It will be appreciated from the description provided herein above that contacting cells with a miRNA may be effected by transfecting/loading the cells with e.g. the mature double stranded miRNA, the pre-miRNA or the pri-miRNA.
[0324] The pre-miRNA sequence may comprise from 45-90, 60-80 or 60-70 nucleotides.
[0325] The pri-miRNA sequence may comprise from 45-30,000, 50-25,000, 100-20,000, 1,000-1,500 or 80-100 nucleotides.
[0326] Antisense--
[0327] Antisense is a single stranded RNA designed to prevent or inhibit expression of a gene by specifically hybridizing to its mRNA. Downregulation of an immune-check point can be effected using an antisense polynucleotide capable of specifically hybridizing with an mRNA transcript encoding the immune-check point protein.
[0328] Design of antisense molecules which can be used to efficiently downregulate an immune-check point must be effected while considering two aspects important to the antisense approach. The first aspect is delivery of the oligonucleotide into the cytoplasm of the appropriate cells, while the second aspect is design of an oligonucleotide which specifically binds the designated mRNA within cells in a way which inhibits translation thereof.
[0329] The prior art teaches of a number of delivery strategies which can be used to efficiently deliver oligonucleotides into a wide variety of cell types [see, for example, Jaaskelainen et al. Cell Mol Biol Lett. (2002) 7(2):236-7; Gait, Cell Mol Life Sci. (2003) 60(5):844-53; Martino et al. J Biomed Biotechnol. (2009) 2009:410260; Grijalvo et al. Expert Opin Ther Pat. (2014) 24(7):801-19; Falzarano et al., Nucleic Acid Ther. (2014) 24(1):87-100; Shilakari et al. Biomed Res Int. (2014) 2014: 526391; Prakash et al. Nucleic Acids Res. (2014) 42(13):8796-807 and Asseline et al. J Gene Med. (2014) 16(7-8):157-65].
[0330] In addition, algorithms for identifying those sequences with the highest predicted binding affinity for their target mRNA based on a thermodynamic cycle that accounts for the energetics of structural alterations in both the target mRNA and the oligonucleotide are also available [see, for example, Walton et al. Biotechnol Bioeng 65: 1-9 (1999)]. Such algorithms have been successfully used to implement an antisense approach in cells.
[0331] In addition, several approaches for designing and predicting efficiency of specific oligonucleotides using an in vitro system were also published [(Matveeva et al., Nature Biotechnology 16: 1374-1375 (1998)].
[0332] Thus, the generation of highly accurate antisense design algorithms and a wide variety of oligonucleotide delivery systems, enable an ordinarily skilled artisan to design and implement antisense approaches suitable for downregulating expression of known sequences without having to resort to undue trial and error experimentation.
[0333] Nucleic acid agents can also operate at the DNA level as summarized infra.
[0334] Suppressing the biological function of an immune-check point can also be achieved by inactivating the gene via introducing targeted mutations involving loss-of function alterations (e.g. point mutations, deletions and insertions) in the gene structure.
[0335] As used herein, the phrase "loss-of-function alterations" refers to any mutation in the DNA sequence of a gene which results in downregulation of the expression level and/or activity of the expressed product, i.e., the mRNA transcript and/or the translated protein. Non-limiting examples of such loss-of-function alterations include a missense mutation, i.e., a mutation which changes an amino acid residue in the protein with another amino acid residue and thereby abolishes the enzymatic activity of the protein; a nonsense mutation, i.e., a mutation which introduces a stop codon in a protein, e.g., an early stop codon which results in a shorter protein devoid of the enzymatic activity; a frame-shift mutation, i.e., a mutation, usually, deletion or insertion of nucleic acid(s) which changes the reading frame of the protein, and may result in an early termination by introducing a stop codon into a reading frame (e.g., a truncated protein, devoid of the enzymatic activity), or in a longer amino acid sequence (e.g., a readthrough protein) which affects the secondary or tertiary structure of the protein and results in a non-functional protein, devoid of the enzymatic activity of the non-mutated polypeptide; a readthrough mutation due to a frame-shift mutation or a modified stop codon mutation (i.e., when the stop codon is mutated into an amino acid codon), with an abolished enzymatic activity; a promoter mutation, i.e., a mutation in a promoter sequence, usually 5' to the transcription start site of a gene, which results in down-regulation of a specific gene product; a regulatory mutation, i.e., a mutation in a region upstream or downstream, or within a gene, which affects the expression of the gene product; a deletion mutation, i.e., a mutation which deletes coding nucleic acids in a gene sequence and which may result in a frame-shift mutation or an in-frame mutation (within the coding sequence, deletion of one or more amino acid codons); an insertion mutation, i.e., a mutation which inserts coding or non-coding nucleic acids into a gene sequence, and which may result in a frame-shift mutation or an in-frame insertion of one or more amino acid codons; an inversion, i.e., a mutation which results in an inverted coding or non-coding sequence; a splice mutation i.e., a mutation which results in abnormal splicing or poor splicing; and a duplication mutation, i.e., a mutation which results in a duplicated coding or non-coding sequence, which can be in-frame or can cause a frame-shift.
[0336] According to specific embodiments loss-of-function alteration of a gene may comprise at least one allele of the gene.
[0337] The term "allele" as used herein, refers to any of one or more alternative forms of a gene locus, all of which alleles relate to a trait or characteristic. In a diploid cell or organism, the two alleles of a given gene occupy corresponding loci on a pair of homologous chromosomes.
[0338] According to other specific embodiments loss-of-function alteration of a gene comprises both alleles of the gene.
[0339] Methods of introducing nucleic acid alterations to a gene of interest are well known in the art [see for example Menke D. Genesis (2013) 51: -618; Capecchi, Science (1989) 244:1288-1292; Santiago et al. Proc Natl Acad Sci USA (2008) 105:5809-5814; International Patent Application Nos. WO2014085593, WO2009071334 and WO2011146121; U.S. Pat. Nos. 8,771,945, 8,586,526, 6,774,279 and U.S. Patent Application Publication Nos. 20030232410, 20050026157, US20060014264; the contents of which are incorporated by reference in their entireties] and include targeted homologous recombination (e.g. "Hit and run", "double-replacement"), site specific recombinases (e.g. the Cre recombinase and the Flp recombinase), PB transposases (e.g. Sleeping Beauty, piggyBac, To12 or Frog Prince), genome editing by engineered nucleases (e.g. meganucleases, Zinc finger nucleases (ZFNs), transcription-activator like effector nucleases (TALENs) and CRISPR/Cas system) and genome editing using recombinant adeno-associated virus (rAAV) platform. Agents for introducing nucleic acid alterations to a gene of interest can be designed publically available sources or obtained commercially from Transposagen, Addgene and Sangamo Biosciences.
[0340] Methods for qualifying efficacy and detecting sequence alteration are well known in the art and include, but not limited to, DNA sequencing, electrophoresis, an enzyme-based mismatch detection assay and a hybridization assay such as PCR, RT-PCR, RNase protection, in-situ hybridization, primer extension, Southern blot, Northern Blot and dot blot analysis.
[0341] Sequence alterations in a specific gene can also be determined at the protein level using e.g. chromatography, electrophoretic methods, immunodetection assays such as ELISA and western blot analysis and immunohistochemistry.
[0342] As mentioned, depending on the immune-check point protein (i.e. co-stimulatory or inhibitory) the immune-check point regulator can be an agonist or antagonist. Thus, according to specific embodiments, the immune-check point regulator is an agonist.
[0343] As used herein the term "agonist" refers to a molecule that induces and/or increases the biological function and/or expression of an immune-check point protein.
[0344] According to specific embodiments, the agonist induces and/or increases the co-stimulatory effect of an immune-check point protein on an immune cell (e.g. T cells).
[0345] According to specific embodiments, the agonist induces and/or increases signaling to an immune cell (e.g. T cell) by an immune-check point protein. The agonist can be a naturally occurring activator or a functional derivative thereof; or non-naturally occurring activator.
[0346] According to specific embodiments, the agonist is a full agonist, that is, the effect of the agonist is equivalent to the effect of the naturally occurring activator (i.e. ligand).
[0347] According to other specific embodiments, the agonist is a partial agonist, that is, the effect of the agonist is lower than the maximal effect of the naturally occurring activator (i.e. ligand). The effect of the agonist may be lower by at least 5%, at least 10%, at least 20%, at least 30%, at least 40% at least 50%, at least 60%, at least 70%, at least 80% or at least 90% as compared to the maximal effect of the naturally occurring activator.
[0348] According to yet other specific embodiments, the agonist is a super agonist, that is, the effect of the agonist is higher than the maximal effect of the naturally occurring activator (i.e. ligand). The effect of the agonist may be higher by at least 5%, at least 10%, at least 20%, at least 30%, at least 40% at least 50%, at least 60%, at least 70%, at least 80%, at least 90% or at least 2 fold, at least 4 fold, at least 5 fold or at least 10 fold as compared to the maximal effect of the naturally occurring activator.
[0349] According to specific embodiments, the agonist induces complete activation the biological function (e.g. signal transduction) of the immune-check point protein.
[0350] According to other specific embodiments, the agonist increases the biological function (e.g. signal transduction) of the immune-check point protein e.g., as detected by e.g. kinase activity, proliferation assay, cytotoxicity assay or cytokine secretion assay. The increase may be by at least a 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95% or at least 99% as compared to same in the absence of the agonist.
[0351] According to specific embodiments, the agonist binds directly the immune-check point protein.
[0352] According to other specific embodiments, the agonist indirectly binds the immune-check point protein by acting through an intermediary molecule, for example the agonist binds to or modulates a molecule that in turn binds to or modulates the immune-check point protein.
[0353] Activating and/or increasing the biological function of an immune-check point protein can be effected at the protein level (e.g., antibodies, small molecules, peptides and the like) but may also be effected at the genomic level (e.g., activation of transcription via promoters, enhancers, regulatory elements) and/or the transcript level using a variety of molecules which promote transcription and/or translation (e.g., correct splicing, polyadenylation, activation of translation) of a co-stimulatory immune-check point protein.
[0354] Non limiting examples of agents that can function as agonists are described in details hereinbelow.
[0355] Activating and/or Increasing Biological Function at the Polypeptide Level
[0356] According to specific embodiments, the agonist is the naturally occurring activator or a functional derivative or variant thereof which retain the ability to specifically bind to the immune-check point protein.
[0357] It will be appreciated that a functional analogue of at least a catalytic or binding portion of a co-stimulatory peptide can be also used as an agonist. Thus, according to specific embodiments, the agonist is an exogenous polypeptide including at least a functional portion (e.g. catalytic or interaction) of the co-stimulatory immune-check point protein. Thus, for example, the polypeptide can be a ligand capable of binding and activating the co-stimulatory immune-check point protein receptor.
[0358] According to specific embodiments, the agonist is an antibody.
[0359] According to specific embodiments the agonistic antibody is capable of specifically binding a co-stimulatory immune-check point protein. According to specific embodiments, the agonistic antibody specifically binds at least one epitope of a co-stimulatory immune-check point protein. A detailed description on antibodies that can be used according to specific embodiments of the present invention is provided hereinabove.
[0360] Another agent capable of being an agonist would be a molecule which promotes and/or increases the co-stimulatory immune-check point protein function (e.g. catalytic or interaction) by binding to the immune-check point protein or an intermediate thereof. Such molecules can be, but are not limited to, small molecules, peptides, aptamers, adnectins, affibodies, avimers, anticalins, tetranectins and DARPins, wherein each possibility is a separate embodiment of the invention.
[0361] According to specific embodiments, the agonist is a small molecule.
[0362] According to specific embodiments, the agonist is a peptide.
[0363] Activating and/or Increasing Biological Function at the Nucleic Acid Level
[0364] An agonist can also be a molecule which is capable of increasing the transcription and/or translation of an endogenous DNA or mRNA encoding the co-stimulatory immune-check point protein and thus increasing endogenous co-stimulatory immune-check point protein activity.
[0365] Another agonistic agent may be an exogenous polynucleotide (DNA or RNA) sequence designed and constructed to express at least a functional portion of the co-stimulatory immune-check point protein.
[0366] Several co-stimulatory immune-check points have been cloned from human, rat and mouse sources. Thus, coding sequences information is available from several databases including the GenBank database available through www(dot)ncbi(dot)nlm(dot)nih(dot)gov/.
[0367] To express an exogenous co-stimulatory immune-check point protein in mammalian cells, a polynucleotide sequence encoding a specific co-stimulatory immune-check point protein or a homologue thereof which exhibit the desired activity is preferably ligated into a nucleic acid construct suitable for mammalian cell expression. Such a nucleic acid construct includes a promoter sequence for directing transcription of the polynucleotide sequence in the cell in a constitutive [e.g. cytomegalovirus (CMV) and Rous sarcoma virus (RSV)] or inducible (e.g. the tetracycline-inducible promoter) manner.
[0368] According to specific embodiments, the promoter utilized by the nucleic acid construct of some embodiments of the invention is active in a specific cell population. Examples of cell type-specific and/or tissue-specific promoters include promoters such as, but not limited to lymphoid specific promoters [Calame et al., (1988) Adv. Immunol. 43:235-275]; in particular promoters of T-cell receptors [Winoto et al., (1989) EMBO J. 8:729-733] and immunoglobulins [Banerji et al. (1983) Cell 33729-740].
[0369] The nucleic acid construct (also referred to herein as an "expression vector") of some embodiments of the invention includes additional sequences which render this vector suitable for replication and integration in prokaryotes, eukaryotes, or preferably both (e.g., shuttle vectors). In addition, a typical cloning vectors may also contain a transcription and translation initiation sequence, transcription and translation terminator and a polyadenylation signal. By way of example, such constructs will typically include a 5' LTR, a tRNA binding site, a packaging signal, an origin of second-strand DNA synthesis, and a 3' LTR or a portion thereof. The construct may also include an enhancer element which can stimulate transcription up to 1,000 fold from linked homologous or heterologous promoters. The vector may or may not include a eukaryotic replicon.
[0370] The nucleic acid construct of some embodiments of the invention can also include a signal sequence for secretion of the peptide from a host cell in which it is placed. Preferably the signal sequence for this purpose is a mammalian signal sequence or the signal sequence of the polypeptide variants of some embodiments of the invention.
[0371] Polyadenylation sequences can also be added to the expression vector in order to increase the efficiency of a co-stimulatory immune-check point mRNA translation. Two distinct sequence elements are required for accurate and efficient polyadenylation: GU or U rich sequences located downstream from the polyadenylation site and a highly conserved sequence of six nucleotides, AAUAAA, located 11-30 nucleotides upstream. Termination and polyadenylation signals that are suitable for some embodiments of the invention include those derived from SV40.
[0372] The expression vector of some embodiments of the invention can further include additional polynucleotide sequences that allow, for example, the translation of several proteins from a single mRNA such as an internal ribosome entry site (IRES) and sequences for genomic integration of the promoter-chimeric polypeptide.
[0373] Other than containing the necessary elements for the transcription and translation of the inserted coding sequence, the expression construct of some embodiments of the invention can also include sequences engineered to enhance stability, production, or yield of the expressed peptide.
[0374] It will be appreciated that the individual elements comprised in the expression vector can be arranged in a variety of configurations.
[0375] The type of vector used by some embodiments of the invention will depend on the cell type transformed. The ability to select suitable vectors according to the cell type transformed is well within the capabilities of the ordinary skilled artisan and as such no general description of selection consideration is provided herein.
[0376] Recombinant viral vectors are useful for in vivo expression of an immune-check point protein since they offer advantages such as lateral infection and targeting specificity. Viral vectors can also be produced that are unable to spread laterally.
[0377] Various methods can be used to introduce the expression vector of some embodiments of the invention into cells. Such methods are generally described in Sambrook et al., Molecular Cloning: A Laboratory Manual, Cold Springs Harbor Laboratory, New York (1989, 1992), in Ausubel et al., Current Protocols in Molecular Biology, John Wiley and Sons, Baltimore, Md. (1989), Chang et al., Somatic Gene Therapy, CRC Press, Ann Arbor, Mich. (1995), Vega et al., Gene Targeting, CRC Press, Ann Arbor Mich. (1995), Vectors: A Survey of Molecular Cloning Vectors and Their Uses, Butterworths, Boston Mass. (1988) and Gilboa et at. [Biotechniques 4 (6): 504-512, 1986].
[0378] Currently preferred in vivo nucleic acid transfer techniques include transfection with viral or non-viral constructs, such as adenovirus, lentivirus, Herpes simplex I virus, or adeno-associated virus (AAV) and lipid-based systems. Useful lipids for lipid-mediated transfer of the gene are, for example, DOTMA, DOPE, and DC-Chol [Tonkinson et al., Cancer Investigation, 14(1): 54-65 (1996)]. The most preferred constructs for use in gene therapy are viruses, most preferably adenoviruses, AAV, lentiviruses, or retroviruses. Other vectors can be used that are non-viral, such as cationic lipids, polylysine, and dendrimers.
[0379] According to specific embodiments, the immune-check point regulator targets an immune check-point protein selected from the group consisting of B7-H3, CD19 and CD70.
[0380] B7-H3 (gene symbol CD276, also known as CD276), is a B7 family protein. The gene is over-expressed in a variety of solid tumors, including prostate, pancreatic, melanoma, renal cell, ovarian, colorectal, gastric, bladder and non-small cell lung cancers has been reported (e.g. Immune Netw. 2014 December; 14(6): 265-276). According to a specific embodiment, the B7-H3 protein refers to the human protein, such as provided in the following GenBank Number NP_001019907.
[0381] CD19 (gene symbol CD19, also known as B-lymphocyte antigen CD19), is a cell surface molecule that assembles with the antigen receptor of B lymphocytes in order to decrease the threshold for antigen receptor-dependent stimulation. CD19 is expressed on follicular dendritic cells and B cells. Typically, it is present on B cells from earliest recognizable B-lineage cells during development to B-cell blasts but is lost on maturation to plasma cells. CD19 is also expressed on several hematologic e.g. B cell tumors. According to a specific embodiment, the CD19 protein refers to the human protein, such as provided in the following GenBank Number NP_001171569.
[0382] CD70 is the ligand for CD27, which is expressed on activated lymphocytes. CD70 expression is found in different types of cancers including renal cell carcinomas, metastatic breast cancers, brain tumours, leukemias, lymphomas and nasopharyngeal carcinomas. According to a specific embodiment, the CD70 protein refers to the human protein, such as provided in the following GenBank Number NP_001243.
[0383] It should be acknowledged that the present invention does not contemplate the use of an immune-check point regulator selected from the group consisting of PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist and ICOS agonist.
[0384] PD1 (Programmed Death 1), gene symbol PDCD1, is also known as CD279. According to a specific embodiment, the PD1 protein refers to the human protein, such as provided in the following GenBank Number NP_005009.
[0385] PDL-1, gene symbol CD274, is also known as CD274 and B7-H1. According to a specific embodiment the PDL-1 protein refers to the human protein, such as provided in the following GenBank accession Numbers NP_054862 and NP_054862.
[0386] CTLA4, gene symbol Ctla4 is also known as CD152. According to a specific embodiment the CTLA-4 protein refers to the human protein, such as provided in the following GenBank Number NP_001032720.
[0387] LAG-3 (Lymphocyte-activation gene 3), gene symbol LAG3, is also known as CD223. According to a specific embodiment the LAG-3 protein refers to the human protein, such as provided in the following GenBank Number NP_002277.
[0388] TIM-3, gene symbol HAVCR2, is also known as Hepatitis A Virus Cellular Receptor 2, T Cell Immunoglobulin Mucin 3, T-Cell Immunoglobulin And Mucin Domain-Containing Protein, T-Cell Membrane Protein 3 and KIM-3. According to a specific embodiment the TIM-3 protein refers to the human protein, such as provided in the following GenBank Number NP_116171.
[0389] KIRs (killer cell Ig-like receptors) are cell surface glycoproteins, comprising one to three extracellular immunoglobulin-like domains, which are expressed by some T cells as well as most human NK cells. As used herein, the term "KIR" refers to a KIR that delivers an inhibitory signal to the cell. A number of KIRs are well characterized (see, e.g., Carrington and Norman, The KIR Gene Cluster, May 28, 2003, available through the National Center for Biotechnology Information (NCBI) web site at www(dot)ncbi(dot)nlm(dot)nih(dot)gov/books/bookres(dot)fcgi/mono_003/ch1d- 1(dot)pdf). The sequences of human KIR genes and cDNAs, as well as their protein products, are available in public databases, including GenBank. Non-limiting exemplary GenBank entries of human KIRs have the following accession numbers: KIR2DL1: Genbank accession number U24076, NM_014218, AAR16197, L41267 or NP_055033; KIR2DL2: Genbank accession number U24075, L76669 or NP_055034; KIR2DL3: Genbank accession number U24074, L41268 or NP_056952; KIR2DL4: Genbank accession number X97229; and KIR3DL1: Genbank accession number L41269.
[0390] IDO (indoleamine 2,3-dioxygenase), EC 1.13.11.52, is a heme-containing intracellular enzyme that catalyzes the first and rate-determining step in the degradation of the essential amino acid L-tryptophan to N-formyl-kynurenine. According to a specific embodiment, the IDO protein refers to the human protein, such as provided in the following GenBank Number NP_002155.
[0391] OX40, gene symbol TNFRSF4, is also known as CD134, Tumor necrosis factor receptor superfamily, member 4, TNFRSF4, TXGP1L, ACT35 and IMD16. According to a specific embodiment, the OX40 protein refers to the human protein, such as provided in the following GenBank Number NP_003318.
[0392] CD137, gene symbol TNFRSF9, is also known as 4-1BB and Tumor Necrosis Factor Receptor Superfamily, Member 9 and TNFRSF9. According to a specific embodiment the CD137 protein refers to the human protein, such as provided in the following GenBank Number NP_001552.
[0393] CD27, gene symbol CD27, is also known as Tumor Necrosis Factor Receptor Superfamily, Member 7, TNFRSF7 and S152. According to a specific embodiment, the CD27 protein refers to the human protein, such as provided in the following GenBank Number NP_001233.
[0394] CD40, gene symbol CD40, is also known as Tumor Necrosis Factor Receptor Superfamily, Member 5 and TNFRSF5. According to a specific embodiment the CD40 protein refers to the human protein, such as provided in the following GenBank Number NP_001241.
[0395] GITR (glucocorticoid-induced tumor necrosis factor receptor), gene symbol TNFRSF18 is also known as TNF receptor superfamily 18, TNFRSF18, AITR and CD357. According to a specific embodiment the GITR protein refers to the human protein, such as provided in the following GenBank Numbers NP_004186, NP_683699, NP_683700.
[0396] CD28, gene symbol CD28, is also known as Tp44. According to a specific embodiment the CD28 protein refers to the human protein, such as provided in the following GenBank Numbers NP_001230006, NP_001230007 and NP_006130.
[0397] ICOS (Inducible T-cell co-stimulator), gene symbol ICOS, is also known as CD278, AILIM and CVID1. According to a specific embodiment the ICOS protein refers to the human protein, such as provided in the following GenBank Number NP_036224.
[0398] It has been demonstrated that multiple cancer cells overexpress certain types of immune-regulatory proteins. Hence, according to specific embodiments, the anti-cancer agent may be an agent capable of binding an immune-check point protein expressed on a cancer cell.
[0399] According to specific embodiments, the immune-check point protein expressed on said cancer cell is selected from the group consisting of PDL-1, CD27, LAG3, CD19, CD70 and CEACAM1.
[0400] According to specific embodiments, binding of the anti-cancer agent to the immune-check point protein expressed on the cancer cell results in at least one of:
[0401] (i) cell cycle arrest of said cancer cell;
[0402] (ii) apoptosis of said cancer cell;
[0403] (iii) sensitization of said cancer cell to a cytotoxic drug; and
[0404] (iv) activation of an immune response against said cancer cell.
[0405] Examples of agents capable of binding a cancer cell expressing a specific antigen that can be used according to specific embodiments of the present invention are described in details hereinabove.
[0406] According to specific embodiments, the agent capable of binding said immune-check point protein is an antibody or a T cell.
[0407] According to specific embodiments, the T cell comprises a T cell transduced with a T cell receptor (TCR) or a chimeric antigen receptor (CAR).
[0408] Specific non-limiting examples of immune-check point regulators and/or agents capable of binding an immune-check point protein expressed on a cancer cell that can be used according to some embodiments of the invention include:
[0409] IMP321, a Ig fusion protein comprising a soluble dimeric recombinant form of LAG-3) designed to activate antigen presenting cells, induce maturation and migration of DCs to the lymph nodes and enhanced cross-presentation of antigens to CD8+ T cells [see e.g. Brignone et al., (2007) J. Immunol. 179:4202-4211].
[0410] An agent targeting B7-H3, such as, MGA271 (produced by MacroGenics), a humanized IgG1.kappa. monoclonal antibody that recognizes human B7-H3, B7-H3 antagonist such as disclosed e.g. in US Application Publication No. US 20130122021 and International Application Publication Nos. WO 2011109400 and WO 2012004410, or a B7-H3 agonist such as disclosed e.g. in International Application Publication No. WO 2004093894, the contents of each of which are fully incorporated herein by reference.
[0411] An agent capable of binding CD19, such as, MEDI-551 (produced by MedImmune) and SAR3419 (produced by Sanofi), monoclonal antibodies that target CD19 and CART cells directed at CD19 such as CTL-019 (produced by Novartis) or such as disclosed e.g. in US Application Publication No: US 20140271635.
[0412] An agent capable of binding CD70, such as ARGX-110 (produced by arGEN-X BVBA) a monoclonal antibody that targets CD70 and induces cytotoxic T cell response and ADCC to cells expressing CD70.
[0413] As disclosed above the anti-cancer agent can also be selected from the group consisting of a colony stimulating factor-1 receptor (CSF1R) antagonist, a CXCR2 antagonist, a STAT3 antagonist, PV-10 and Cotara.
[0414] CSF1R (also known as macrophage colony-stimulating factor receptor, M-CSFR and CD115) is a cell single pass type I membrane protein acting as the receptor for colony stimulating factor 1, a cytokine which controls the production, differentiation, and function of macrophages. According to a specific embodiment, the CSF1R protein refers to the human protein, such as provided in the following GenBank Number NP_001275634.
[0415] Specific non-limiting examples of CSF1R antagonists that can be used according to some embodiments of the invention include the antibodies FPA008 (produced by Five Prime Therapeutics) and RG7155 (produced by Roche). Tumor associated macrophages (TAM), are dependent on CSF1R signaling. TAMs are myeloid lineage-derived cells that are part of the tumor microenvironmental support system. TAMs are potently angiogenic, remodel the stroma (extracellular matrix and related components) and are immunosuppressive. The plethora of critical factors produced by TAM include hypoxia response proteins and growth factors that drive angiogenesis, tissue remodeling and immunosuppression, i.e. HIF2a, MMP-9, EGF, VEGF and TGF-beta, cytokines that can maintain this response in a chronic state (IL-10, IL-4) and chemokines that attract myeloid cells and regulatory T cells (CCL22, CCXL8). The TAM population can be directly regulated by tumor cell secretion of CSF-1, thus the importance of the CSF1R target.
[0416] CXCR2 (also known as IL-8 receptor, beta) is a G-protein-coupled chemokine receptor which functions as a receptor for e.g. IL-8, CXCL1, CXCL2, CXCL3 and CXCL5. According to a specific embodiment, the CXCR2 protein refers to the human protein, such as provided in the following GenBank Number NP_001161770.
[0417] A specific non-limiting example of a CXCR2 antagonist that can be used according to some embodiments of the invention include AZD5069 (produced by AstraZeneca). Cancer initiation and progression also depends on escape from host immunosurveillance. In several solid tumors, immune evasion involves a shift of immune responses, including imbalance in Th1/Th2 responses and enhancement of immunosuppressive cells such as myeloid-derived suppressor cells (MDSCs) and regulatory T cells. The number of MDSCs in the blood correlates well with clinical cancer stage and metastatic tumor burden in patients (Diaz-Montero et al., 2009; Cancer Immunology, Immunotherapy, January 2009, Volume 58, Issue 1, pp 49-59). It is widely accepted that MDSCs contribute to cancer immune evasion via suppressing functions of T and natural killer (NK) cells (Gabrilovich and Nagaraj, 2009 Nature Reviews Immunology 9, 162-174). Accumulation of Myeloid Derived Suppressor Cells (MDSCs) in the tumor bed limits the efficacy of checkpoint blockade in cancer. Expansion of MDSCs is a major mechanism of tumor immune escape. CXCR2 is essential for MDSC infiltration into tumors, which promotes tumor growth and progression via inhibition of CD8+ T cell cytotoxic activity.
[0418] Signal transducer and activator of transcription 3 (STAT3) is a transcription factor encoded by the STAT3 gene. STAT3, is a member of the STAT protein family of signal transducers and activators of transcription, which represent a family of proteins that, when activated by protein tyrosine kinases in the cytoplasm of the cell, migrate to the nucleus and activate gene transcription.
[0419] According to a specific embodiment, the STAT3 protein refers to the human protein, such as provided in the following GenBank Number NP_003141.
[0420] Specific non-limiting examples of STAT3 antagonists that can be used according to some embodiments of the invention include AZD9150 (produced by AstraZeneca), a STAT3 antisense; and STAT3 siRNA such as disclosed e.g. in Herrmann A, et al. J Clin Invest. (2014) 124(7): 2977-87.
[0421] PV-10 (produced by Provectus Biopharmaceuticals Inc.) is a composition containing Rose Bengal shown to eradicate tumors and activate anti-tumor T cell response.
[0422] Cotara (131I-chTNT-1/B Mab, produced by Peregrine Pharmaceuticals Inc.), is an antibody which is combined with radioactive iodine. Typically, Cotara binds to proteins within the nucleus of necrotic (i.e. dead and dying) cells which are present in most malignant tumors and are primarily found at the center of the tumor. When the Ab is delivered directly to the tumor, it remains within the tumor and the attached radioactive iodine bombards the neighboring living tumor cells with radiation.
[0423] According to specific embodiments, the CXCR4 antagonistic peptide can be administered to a subject in combination with several of the anti-cancer agents described hereinabove.
[0424] The order in which the CXCR4 antagonistic peptide and the anti-cancer agent are administered to the subject can vary according to the method of treating.
[0425] Thus, according to a specific embodiment, administering the peptide is effected prior to administering the agent.
[0426] According to another specific embodiment, administering the peptide is effected following administering the agent.
[0427] According to yet another specific embodiment, administering the peptide is effected concomitantly with administering the agent.
[0428] Multiple rounds of administration according to the methods of the present invention and multiple doses of the CXCR4 antagonistic peptide and the anti-cancer agent can be administered. According to specific embodiments, administering comprises multiple administrations of the peptide. Thus, according to specific embodiments, administration of the anti-cancer agent is effected following at least one administration of the CXCR4 antagonistic peptide. According to specific embodiments, administering comprises multiple administrations of the anti-cancer agent. Thus, according to specific embodiments, administering the CXCR4 antagonistic peptide of the present invention is effected following at least one administration of the agent. According to specific embodiments, administering the peptide and administering the agent are effected sequentially.
[0429] According to specific embodiments, the CXCR4 antagonistic peptide and the anti-cancer agent of the invention can be administered to a subject in combination with other established or experimental therapeutic regimen to treat cancer including analgetics, chemotherapeutic agents, radiotherapeutic agents, hormonal therapy, immune modulators and other treatment regimens (e.g., surgery, cell transplantation e.g. hematopoietic stem cell transplantation) which are well known in the art.
[0430] The CXCR4 antagonistic peptides and/or the anti-cancer agent described hereinabove can be administered to the subject per se, or in a pharmaceutical composition where it is mixed with suitable carriers or excipients.
[0431] Thus, according to an aspect of the present invention there is provided a pharmaceutical composition comprising as active ingredients a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof and an anti-cancer agent selected from the group consisting of:
[0432] (i) a vaccine selected from the group consisting of IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000;
[0433] (ii) anti-cancer reactive mononuclear blood cells (MNBCs);
[0434] (iii) a cytokine capable of inducing activation and/or proliferation of a T cell;
[0435] (iv) an immune-check point regulator, wherein said immune-check point regulator is not a PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist or ICOS agonist;
[0436] (v) an agent capable of binding an immune-check point protein expressed on a cancer cell;
[0437] (vi) a colony stimulating factor-1 receptor (CSF1R) antagonist;
[0438] (vii) a CXCR2 antagonist;
[0439] (viii) a STAT3 antagonist;
[0440] (ix) PV-10; and
[0441] (x) Cotara,
and a pharmaceutically acceptable carrier or diluent.
[0442] As used herein a "pharmaceutical composition" refers to a preparation of one or more of the active ingredients described herein with other chemical components such as physiologically suitable carriers and excipients. The purpose of a pharmaceutical composition is to facilitate administration of a compound to an organism.
[0443] Herein the term "active ingredient" refers to the CXCR4 antagonistic peptides and/or the anti-cancer agent accountable for the biological effect.
[0444] Hereinafter, the phrases "physiologically acceptable carrier" and "pharmaceutically acceptable carrier" which may be interchangeably used refer to a carrier or a diluent that does not cause significant irritation to an organism and does not abrogate the biological activity and properties of the administered compound. An adjuvant is included under these phrases.
[0445] Herein the term "excipient" refers to an inert substance added to a pharmaceutical composition to further facilitate administration of an active ingredient. Examples, without limitation, of excipients include calcium carbonate, calcium phosphate, various sugars and types of starch, cellulose derivatives, gelatin, vegetable oils and polyethylene glycols.
[0446] Techniques for formulation and administration of drugs may be found in "Remington's Pharmaceutical Sciences," Mack Publishing Co., Easton, Pa., latest edition, which is incorporated herein by reference.
[0447] Suitable routes of administration may, for example, include oral, rectal, transmucosal, especially transnasal, intestinal or parenteral delivery, including intramuscular, intradermal, subcutaneous and intramedullary injections as well as intrathecal, direct intraventricular, intracardiac, e.g., into the right or left ventricular cavity, into the common coronary artery, intravenous, intraperitoneal, intranasal, or intraocular injections.
[0448] Conventional approaches for drug delivery to the central nervous system (CNS) include: neurosurgical strategies (e.g., intracerebral injection or intracerebroventricular infusion); molecular manipulation of the agent (e.g., production of a chimeric fusion protein that comprises a transport peptide that has an affinity for an endothelial cell surface molecule in combination with an agent that is itself incapable of crossing the BBB) in an attempt to exploit one of the endogenous transport pathways of the BBB; pharmacological strategies designed to increase the lipid solubility of an agent (e.g., conjugation of water-soluble agents to lipid or cholesterol carriers); and the transitory disruption of the integrity of the BBB by hyperosmotic disruption (resulting from the infusion of a mannitol solution into the carotid artery or the use of a biologically active agent such as an angiotensin peptide). However, each of these strategies has limitations, such as the inherent risks associated with an invasive surgical procedure, a size limitation imposed by a limitation inherent in the endogenous transport systems, potentially undesirable biological side effects associated with the systemic administration of a chimeric molecule comprised of a carrier motif that could be active outside of the CNS, and the possible risk of brain damage within regions of the brain where the BBB is disrupted, which renders it a suboptimal delivery method.
[0449] Alternately, one may administer the pharmaceutical composition in a local rather than systemic manner, for example, via injection of the pharmaceutical composition directly into a tissue region of a patient.
[0450] The CXCR4 antagonistic peptide of the invention, the anti-cancer agent or the pharmaceutical composition comprising same can be administered in the same route or in separate routes.
[0451] According to a specific embodiment, the CXCR4 antagonistic peptide of the invention or the pharmaceutical composition comprising same is administered subcutaneously.
[0452] According to another specific embodiment, the CXCR4 antagonistic peptide of the invention or the pharmaceutical composition comprising same is administered intravenously.
[0453] According to a specific embodiment, the anti-cancer agent or the pharmaceutical composition comprising same is administered intravenously.
[0454] According to a specific embodiment, the anti-cancer agent or the pharmaceutical composition comprising same is administered via a subcutaneous route.
[0455] Pharmaceutical compositions of some embodiments of the invention may be manufactured by processes well known in the art, e.g., by means of conventional mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping or lyophilizing processes.
[0456] Pharmaceutical compositions for use in accordance with some embodiments of the invention thus may be formulated in conventional manner using one or more physiologically acceptable carriers comprising excipients and auxiliaries, which facilitate processing of the active ingredients into preparations which, can be used pharmaceutically. Proper formulation is dependent upon the route of administration chosen.
[0457] For injection, the active ingredients of the pharmaceutical composition may be formulated in aqueous solutions, preferably in physiologically compatible buffers such as Hank's solution, Ringer's solution, or physiological salt buffer. For transmucosal administration, penetrants appropriate to the barrier to be permeated are used in the formulation. Such penetrants are generally known in the art.
[0458] For oral administration, the pharmaceutical composition can be formulated readily by combining the active compounds with pharmaceutically acceptable carriers well known in the art. Such carriers enable the pharmaceutical composition to be formulated as tablets, pills, dragees, capsules, liquids, gels, syrups, slurries, suspensions, and the like, for oral ingestion by a patient. Pharmacological preparations for oral use can be made using a solid excipient, optionally grinding the resulting mixture, and processing the mixture of granules, after adding suitable auxiliaries if desired, to obtain tablets or dragee cores. Suitable excipients are, in particular, fillers such as sugars, including lactose, sucrose, mannitol, or sorbitol; cellulose preparations such as, for example, maize starch, wheat starch, rice starch, potato starch, gelatin, gum tragacanth, methyl cellulose, hydroxypropylmethyl-cellulose, sodium carbomethylcellulose; and/or physiologically acceptable polymers such as polyvinylpyrrolidone (PVP). If desired, disintegrating agents may be added, such as cross-linked polyvinyl pyrrolidone, agar, or alginic acid or a salt thereof such as sodium alginate.
[0459] Dragee cores are provided with suitable coatings. For this purpose, concentrated sugar solutions may be used which may optionally contain gum arabic, talc, polyvinyl pyrrolidone, carbopol gel, polyethylene glycol, titanium dioxide, lacquer solutions and suitable organic solvents or solvent mixtures. Dyestuffs or pigments may be added to the tablets or dragee coatings for identification or to characterize different combinations of active compound doses.
[0460] Pharmaceutical compositions which can be used orally include push-fit capsules made of gelatin as well as soft, sealed capsules made of gelatin and a plasticizer, such as glycerol or sorbitol. The push-fit capsules may contain the active ingredients in admixture with filler such as lactose, binders such as starches, lubricants such as talc or magnesium stearate and, optionally, stabilizers. In soft capsules, the active ingredients may be dissolved or suspended in suitable liquids, such as fatty oils, liquid paraffin, or liquid polyethylene glycols. In addition, stabilizers may be added. All formulations for oral administration should be in dosages suitable for the chosen route of administration.
[0461] For buccal administration, the compositions may take the form of tablets or lozenges formulated in conventional manner.
[0462] For administration by nasal inhalation, the active ingredients for use according to some embodiments of the invention are conveniently delivered in the form of an aerosol spray presentation from a pressurized pack or a nebulizer with the use of a suitable propellant, e.g., dichlorodifluoromethane, trichlorofluoromethane, dichloro-tetrafluoroethane or carbon dioxide. In the case of a pressurized aerosol, the dosage unit may be determined by providing a valve to deliver a metered amount. Capsules and cartridges of, e.g., gelatin for use in a dispenser may be formulated containing a powder mix of the compound and a suitable powder base such as lactose or starch.
[0463] The pharmaceutical composition described herein may be formulated for parenteral administration, e.g., by bolus injection or continuous infusion. Formulations for injection may be presented in unit dosage form, e.g., in ampoules or in multidose containers with optionally, an added preservative. The compositions may be suspensions, solutions or emulsions in oily or aqueous vehicles, and may contain formulatory agents such as suspending, stabilizing and/or dispersing agents.
[0464] Pharmaceutical compositions for parenteral administration include aqueous solutions of the active preparation in water-soluble form. Additionally, suspensions of the active ingredients may be prepared as appropriate oily or water based injection suspensions. Suitable lipophilic solvents or vehicles include fatty oils such as sesame oil, or synthetic fatty acids esters such as ethyl oleate, triglycerides or liposomes.
[0465] Aqueous injection suspensions may contain substances, which increase the viscosity of the suspension, such as sodium carboxymethyl cellulose, sorbitol or dextran. Optionally, the suspension may also contain suitable stabilizers or agents which increase the solubility of the active ingredients to allow for the preparation of highly concentrated solutions.
[0466] Alternatively, the active ingredient may be in powder form for constitution with a suitable vehicle, e.g., sterile, pyrogen-free water based solution, before use.
[0467] The pharmaceutical composition of some embodiments of the invention may also be formulated in rectal compositions such as suppositories or retention enemas, using, e.g., conventional suppository bases such as cocoa butter or other glycerides.
[0468] Alternative embodiments include depots providing sustained release or prolonged duration of activity of the active ingredient in the subject, as are well known in the art.
[0469] Pharmaceutical compositions suitable for use in context of some embodiments of the invention include compositions wherein the active ingredients are contained in an amount effective to achieve the intended purpose. More specifically, according to specific embodiments, a therapeutically effective amount means an amount of active ingredients effective to prevent, alleviate or ameliorate symptoms of a disorder (e.g., cancer) or prolong the survival of the subject being treated.
[0470] Determination of a therapeutically effective amount is well within the capability of those skilled in the art, especially in light of the detailed disclosure provided herein.
[0471] For any preparation used in the methods of the invention, the therapeutically effective amount or dose can be estimated initially from in vitro and cell culture assays. For example, a dose can be formulated in animal models to achieve a desired concentration or titer. Such information can be used to more accurately determine useful doses in humans.
[0472] Toxicity and therapeutic efficacy of the active ingredients described herein can be determined by standard pharmaceutical procedures in vitro, in cell cultures or experimental animals. The data obtained from these in vitro and cell culture assays and animal studies can be used in formulating a range of dosage for use in human.
[0473] The dosage may vary depending upon the dosage form employed and the route of administration utilized.
[0474] The exact formulation, route of administration and dosage can be chosen by the individual physician in view of the patient's condition. (See e.g., Fingl, et al., 1975, in "The Pharmacological Basis of Therapeutics", Ch. 1 p. 1).
[0475] Dosage amount and interval may be adjusted individually to provide levels of the active ingredient are sufficient to induce or suppress the biological effect (minimal effective concentration, MEC). The MEC will vary for each preparation, but can be estimated from in vitro data. Dosages necessary to achieve the MEC will depend on individual characteristics and route of administration. Detection assays can be used to determine plasma concentrations.
[0476] Depending on the severity and responsiveness of the condition to be treated, dosing can be of a single or a plurality of administrations, with course of treatment lasting from several days to several weeks or until cure is effected or diminution of the disease state is achieved.
[0477] The amount of a composition to be administered will, of course, be dependent on the subject being treated, the severity of the affliction, the manner of administration, the judgment of the prescribing physician, etc.
[0478] According to specific embodiments the CXCR4 antagonistic peptide of the invention or the pharmaceutical composition comprising same is administered in a dose ranging between 0.1 to 10 mg/kg of body weight, between 0.1 to 2 mg/kg of body weight, between 0.1 to 1 mg/kg of body weight, between 0.3 to 10 mg/kg of body weight, between 0.3 to 2 mg/kg of body weight, between 0.3 to 1 mg/kg of body weight or between 0.3 to 0.9 mg/kg of body weight.
[0479] According to a specific embodiment, the CXCR4 antagonistic peptide of the invention or the pharmaceutical composition comprising same is administered in a dose ranging between 0.5-2 mg/kg.
[0480] According to another specific embodiment the CXCR4 antagonistic peptide of the invention or the pharmaceutical composition comprising same is administered at a dose of 0.5-1 mg/kg.
[0481] According to specific embodiments, the anti-cancer antibody is administered in a dose ranging between 0.001 to 30 mg/kg body weight, between 0.001 to 20 mg/kg body weight, between 0.001 to 10 mg/kg body weight, between 0.001 to 1 mg/kg body weight, between 0.01 to 30 mg/kg body weight, between 0.01 to 20 mg/kg body weight, between 0.01 to 10 mg/kg body weight, between 0.01 to 1 mg/kg body weight, between 0.1 to 30 mg/kg body weight, between 0.1 to 20 mg/kg body weight, between 0.1 to 10 mg/kg body weight, between 0.1 to 1 mg/kg body weight, between 1 to about 30 mg/kg, between 1 to about 20 mg/kg or between 1 to about 10 mg/kg.
[0482] The desired dose can be administered at one time or divided into sub-doses, e.g., 2-4 sub-doses and administered over a period of time, e.g., at appropriate intervals through the day or other appropriate schedule.
[0483] According to specific embodiments, the CXCR4 antagonistic peptide of the invention, the anti-cancer agent or the pharmaceutical composition comprising same is administered multiple times e.g. 2-10, over a period of time e.g. for several days to several weeks at appropriate intervals e.g. once a day, twice a week, once a week, once every two weeks, once a month, once every 3 to 6 months.
[0484] Compositions of some embodiments of the invention may, if desired, be presented in a pack or dispenser device, such as an FDA approved kit, which may contain one or more unit dosage forms containing the active ingredient. The pack may, for example, comprise metal or plastic foil, such as a blister pack. The pack or dispenser device may be accompanied by instructions for administration. The pack or dispenser may also be accommodated by a notice associated with the container in a form prescribed by a governmental agency regulating the manufacture, use or sale of pharmaceuticals, which notice is reflective of approval by the agency of the form of the compositions or human or veterinary administration. Such notice, for example, may be of labeling approved by the U.S. Food and Drug Administration for prescription drugs or of an approved product insert. Compositions comprising a preparation of the invention formulated in a compatible pharmaceutical carrier may also be prepared, placed in an appropriate container, and labeled for treatment of an indicated condition, as is further detailed above.
[0485] According an aspect of the present invention there is provided an article of manufacture identified for use in treating cancer, comprising a packaging material packaging a peptide having an amino acid sequence as set forth in SEQ ID NO: 1 or an analog or derivative thereof and an anti-cancer agent selected from the group consisting of:
[0486] (i) a vaccine selected from the group consisting of IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000;
[0487] (ii) anti-cancer reactive mononuclear blood cells (MNBCs);
[0488] (iii) a cytokine capable of inducing activation and/or proliferation of a T cell;
[0489] (iv) an immune-check point regulator, wherein said immune-check point regulator is not a PD1 antagonist, PDL-1 antagonist, CTLA-4 antagonist, LAG-3 antagonist, TIM-3 antagonist, KIR antagonist, IDO antagonist, OX40 agonist, CD137 agonist, CD27 agonist, CD40 agonist, GITR agonist, CD28 agonist or ICOS agonist;
[0490] (v) an agent capable of binding an immune-check point protein expressed on a cancer cell;
[0491] (vi) a colony stimulating factor-1 receptor (CSF1R) antagonist;
[0492] (vii) a CXCR2 antagonist;
[0493] (viii) a STAT3 antagonist;
[0494] (ix) PV-10; and
[0495] (x) Cotara.
[0496] The peptide and the agent may be packaged in the same container or in separate containers; each possibility represents a separate embodiment of the present invention.
[0497] According to specific embodiments, the peptide and the agent are in separate containers.
[0498] According to specific embodiments, the peptide and the agent are in separate formulations.
[0499] According to other specific embodiments, the peptide and the agent are in a co-formulation.
[0500] It is expected that during the life of a patent maturing from this application many relevant anti-cancer agents will be developed and the scope of the term "anti-cancer agent" is intended to include all such new technologies a priori.
[0501] As used herein the term "about" refers to .+-.10%.
[0502] The terms "comprises", "comprising", "includes", "including", "having" and their conjugates mean "including but not limited to".
[0503] The term "consisting of" means "including and limited to".
[0504] The term "consisting essentially of" means that the composition, method or structure may include additional ingredients, steps and/or parts, but only if the additional ingredients, steps and/or parts do not materially alter the basic and novel characteristics of the claimed composition, method or structure.
[0505] As used herein, the singular form "a", "an" and "the" include plural references unless the context clearly dictates otherwise. For example, the term "a compound" or "at least one compound" may include a plurality of compounds, including mixtures thereof.
[0506] Throughout this application, various embodiments of this invention may be presented in a range format. It should be understood that the description in range format is merely for convenience and brevity and should not be construed as an inflexible limitation on the scope of the invention. Accordingly, the description of a range should be considered to have specifically disclosed all the possible subranges as well as individual numerical values within that range. For example, description of a range such as from 1 to 6 should be considered to have specifically disclosed subranges such as from 1 to 3, from 1 to 4, from 1 to 5, from 2 to 4, from 2 to 6, from 3 to 6 etc., as well as individual numbers within that range, for example, 1, 2, 3, 4, 5, and 6. This applies regardless of the breadth of the range.
[0507] Whenever a numerical range is indicated herein, it is meant to include any cited numeral (fractional or integral) within the indicated range. The phrases "ranging/ranges between" a first indicate number and a second indicate number and "ranging/ranges from" a first indicate number "to" a second indicate number are used herein interchangeably and are meant to include the first and second indicated numbers and all the fractional and integral numerals therebetween.
[0508] As used herein the term "method" refers to manners, means, techniques and procedures for accomplishing a given task including, but not limited to, those manners, means, techniques and procedures either known to, or readily developed from known manners, means, techniques and procedures by practitioners of the chemical, pharmacological, biological, biochemical and medical arts.
[0509] When reference is made to particular sequence listings, such reference is to be understood to also encompass sequences that substantially correspond to its complementary sequence as including minor sequence variations, resulting from, e.g., sequencing errors, cloning errors, or other alterations resulting in base substitution, base deletion or base addition, provided that the frequency of such variations is less than 1 in 50 nucleotides, alternatively, less than 1 in 100 nucleotides, alternatively, less than 1 in 200 nucleotides, alternatively, less than 1 in 500 nucleotides, alternatively, less than 1 in 1000 nucleotides, alternatively, less than 1 in 5,000 nucleotides, alternatively, less than 1 in 10,000 nucleotides.
[0510] It is appreciated that certain features of the invention, which are, for clarity, described in the context of separate embodiments, may also be provided in combination in a single embodiment. Conversely, various features of the invention, which are, for brevity, described in the context of a single embodiment, may also be provided separately or in any suitable subcombination or as suitable in any other described embodiment of the invention. Certain features described in the context of various embodiments are not to be considered essential features of those embodiments, unless the embodiment is inoperative without those elements.
[0511] Various embodiments and aspects of the present invention as delineated hereinabove and as claimed in the claims section below find experimental support in the following examples.
EXAMPLES
[0512] Reference is now made to the following examples, which together with the above descriptions illustrate some embodiments of the invention in a non limiting fashion.
[0513] Generally, the nomenclature used herein and the laboratory procedures utilized in the present invention include molecular, biochemical, microbiological and recombinant DNA techniques. Such techniques are thoroughly explained in the literature. See, for example, "Molecular Cloning: A laboratory Manual" Sambrook et al., (1989); "Current Protocols in Molecular Biology" Volumes I-III Ausubel, R. M., ed. (1994); Ausubel et al., "Current Protocols in Molecular Biology", John Wiley and Sons, Baltimore, Md. (1989); Perbal, "A Practical Guide to Molecular Cloning", John Wiley & Sons, New York (1988); Watson et al., "Recombinant DNA", Scientific American Books, New York; Birren et al. (eds) "Genome Analysis: A Laboratory Manual Series", Vols. 1-4, Cold Spring Harbor Laboratory Press, New York (1998); methodologies as set forth in U.S. Pat. Nos. 4,666,828; 4,683,202; 4,801,531; 5,192,659 and 5,272,057; "Cell Biology: A Laboratory Handbook", Volumes I-III Cellis, J. E., ed. (1994); "Culture of Animal Cells--A Manual of Basic Technique" by Freshney, Wiley-Liss, N. Y. (1994), Third Edition; "Current Protocols in Immunology" Volumes I-III Coligan J. E., ed. (1994); Stites et al. (eds), "Basic and Clinical Immunology" (8th Edition), Appleton & Lange, Norwalk, Conn. (1994); Mishell and Shiigi (eds), "Selected Methods in Cellular Immunology", W. H. Freeman and Co., New York (1980); available immunoassays are extensively described in the patent and scientific literature, see, for example, U.S. Pat. Nos. 3,791,932; 3,839,153; 3,850,752; 3,850,578; 3,853,987; 3,867,517; 3,879,262; 3,901,654; 3,935,074; 3,984,533; 3,996,345; 4,034,074; 4,098,876; 4,879,219; 5,011,771 and 5,281,521; "Oligonucleotide Synthesis" Gait, M. J., ed. (1984); "Nucleic Acid Hybridization" Hames, B. D., and Higgins S. J., eds. (1985); "Transcription and Translation" Hames, B. D., and Higgins S. J., eds. (1984); "Animal Cell Culture" Freshney, R. I., ed. (1986); "Immobilized Cells and Enzymes" IRL Press, (1986); "A Practical Guide to Molecular Cloning" Perbal, B., (1984) and "Methods in Enzymology" Vol. 1-317, Academic Press; "PCR Protocols: A Guide To Methods And Applications", Academic Press, San Diego, Calif. (1990); Marshak et al., "Strategies for Protein Purification and Characterization--A Laboratory Course Manual" CSHL Press (1996); all of which are incorporated by reference as if fully set forth herein. Other general references are provided throughout this document. The procedures therein are believed to be well known in the art and are provided for the convenience of the reader. All the information contained therein is incorporated herein by reference.
Example 1
Use of BL-8040 for Treating Cancer
[0514] BL-8040 is safe and well tolerated drug that was shown to induce rapid mobilization of hematopoietic stem/progenitor cells and mesenchymal stem cells as well as T cells, B cells, NK cells, NKT cells and ImDC to the peripheral blood. Therefore, BL-8040 can be used to induce the mobilization and dissemination of immature DC and T effector and memory cells into tumors.
[0515] Experimental Procedures
[0516] According to one protocol, BL-8040 is injected into a cancer patient at a dose of 0.5-1 mg/kg for 3-10 days and then 1-3 times a week in combination with a tumor vaccine such as a dendritic cells vaccine, IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000.
[0517] According to another protocol, BL-8040 is injected into a cancer patient at a dose of 0.5-1 mg/kg for 3-10 days and then 1-3 times a week in combination with immunomodulatory cytokines such as IL-2, IFN-.alpha., IL-12.
[0518] According to another protocol, BL-8040 is injected into a cancer patient at a dose of 0.5-1 mg/kg for 3-10 days and then 1-3 times a week in combination with a tumor vaccine such as a dendritic cells vaccine, IMCgp100, Prophage G-100 & G-200, GV-1001, IMA-950, CV-9201, CV-9104, Ad-RTS-hIL-12, ETBX-011, Cavatak, JX-594, ColoAd1, GL-ONC1, ONCOS-102, CRS-207, ADU-623, Dorgenmeltucel-L, HyperAcute Prostate, FANG vaccine, MGN-1601, HPV vaccine and Tarmogens such as GI-4000 and immunomodulatory cytokines such as IL-2, IFN-.alpha. and IL-12.
[0519] According to another protocol, BL-8040 is injected into a cancer patient at a dose of 0.5-1 mg/kg for 3-10 days and then 1-3 times a week in combination with anti-cancer adoptive T cell transfer such as T cells transduced with an anti-cancer T cell receptor (TCR) or a chimeric antigen receptor (CAR).
[0520] According to another protocol, BL-8040 is injected into a cancer patient at a dose of 0.5-1 mg/kg for 3-10 days and then 1-3 times a week in combination with an immune-check point regulator targeting e.g. B7-H3, CD19 and CD70.
[0521] According to another protocol, BL-8040 is injected into a cancer patient at a dose of 0.5-1 mg/kg for 3-10 days and then 1-3 times a week in combination with an anti-cancer agent (e.g. antibody or T cell) capable of binding a cancer cell expressing an immune-check point protein.
[0522] Although the invention has been described in conjunction with specific embodiments thereof, it is evident that many alternatives, modifications and variations will be apparent to those skilled in the art. Accordingly, it is intended to embrace all such alternatives, modifications and variations that fall within the spirit and broad scope of the appended claims.
[0523] All publications, patents and patent applications mentioned in this specification are herein incorporated in their entirety by reference into the specification, to the same extent as if each individual publication, patent or patent application was specifically and individually indicated to be incorporated herein by reference. In addition, citation or identification of any reference in this application shall not be construed as an admission that such reference is available as prior art to the present invention. To the extent that section headings are used, they should not be construed as necessarily limiting.
Sequence CWU 1
1
72114PRTArtificialsynthetic
peptideMOD_RES(1)..(1)4-fluorobenzoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(1-
2)citrullineMOD_RES(14)..(14)C' AMIDATED 1Xaa Arg Xaa Cys Tyr Xaa
Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5 10214PRTArtificialSYNTHETIC
PEPTIDEMOD_RES(1)..(1)N' ACETYLATIONMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrulline 2Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1
5 10314PRTArtificialSYNTHETIC PEPTIDEMOD_RES(1)..(1)N'
ACETYLATIONMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(12)citrulline 3Arg
Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
10414PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(-
12)citrulline 4Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys
Arg1 5 10514PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(11)..(12)c-
itrulline 5Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Xaa Xaa Cys Arg1
5 10614PRTArtificialsynthetic peptideMOD_RES(1)..(1)N' acetylated
citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrulline 6Xaa Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1
5 10714PRTArtificialsynthetic peptideMOD_RES(1)..(1)N' acetylated
citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(12)citrulline 7Xaa
Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
10814PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(11)..(12)citrulline 8Arg
Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Xaa Xaa Cys Arg1 5
10914PRTArtificialsynthetic peptideMOD_RES(1)..(1)N' ACETYLATED
citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(11)..(12)citrulline 9Xaa Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Xaa Xaa Cys Arg1 5
101014PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(-
12)citrullineMOD_RES(14)..(14)C' AMIDATED 10Arg Arg Xaa Cys Tyr Xaa
Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5 101114PRTArtificialsynthetic
peptideMOD_RES(1)..(1)N' ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(11)..(12)c-
itrullineMOD_RES(14)..(14)C' AMIDATED 11Arg Arg Xaa Cys Tyr Xaa Lys
Xaa Pro Tyr Xaa Xaa Cys Arg1 5 101214PRTArtificialsynthetic
peptideMOD_RES(1)..(1)N' ACETYLATED
citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrullineMOD_RES(14)..(14)AMIDATED 12Xaa Arg Xaa Cys Tyr Xaa Lys
Xaa Pro Tyr Arg Xaa Cys Arg1 5 101314PRTArtificialsynthetic
peptideMOD_RES(1)..(1)N' ACETYLATED
citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(12)citrullineMOD_RES(14).-
.(14)C' AMIDATED 13Xaa Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa
Cys Arg1 5 101414PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(11)..(12)citrullineMOD_RES(14).-
.(14)C' AMIDATED 14Arg Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Xaa Xaa
Cys Arg1 5 101514PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATED citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(11)..(12)citrullineMOD_RES(14)..(14-
)C' AMIDATED 15Xaa Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Xaa Xaa Cys
Arg1 5 101614PRTArtificialsynthetic
peptideMOD_RES(1)..(1)D-glutamic acidMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 16Xaa Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
101714PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 17Arg Glu
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
101814PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 18Arg Arg
Xaa Cys Tyr Glu Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
101914PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 19Arg Arg
Xaa Cys Tyr Arg Glu Xaa Pro Tyr Arg Xaa Cys Arg1 5
102014PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-glutamic acidMOD_RES(12)..(12)citrulline
20Arg Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
102114PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 21Arg Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Glu Xaa Cys Arg1 5
102214PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 22Arg Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Glu1 5
102314PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)C' amidated 23Arg
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
102414PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)D-glutamic
acidMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(12)citrullineMOD_RES(14)..(1-
4)c' amidated 24Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys
Arg1 5 102514PRTArtificialsynthetic
peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)D-glutamic acidMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 25Arg
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
102614PRTArtificialsynthetic peptideMOD_RES(1)..(1)D-glutamic
acidMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 26Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
102714PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(10)..(10)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 27Arg
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Xaa Arg Xaa Cys Arg1 5
102814PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)D-glutamic acidMOD_RES(14)..(14)c' amidated
28Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
102914PRTArtificialsynthetic peptideMOD_RES(1)..(1)N' ACETYLATED
D-glutamic acidMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 29Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103014PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(10)..(10)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 30Arg
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Xaa Arg Xaa Cys Arg1 5
103114PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)D-glutamic acidMOD_RES(14)..(14)c' amidated
31Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103214PRTArtificialsynthetic peptideMOD_RES(1)..(1)N'
ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 32Arg
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103314PRTArtificialsynthetic
peptideMOD_RES(1)..(1)Guanyl-arginineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 33Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103414PRTArtificialsynthetic
peptideMOD_RES(1)..(1)Tetramethylguanyl-arginineMOD_RES(3)..(3)3-((2-naph-
thyl) alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 34Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103513PRTArtificialsynthetic
peptideMOD_RES(1)..(1)Tetramethylguanyl-arginineMOD_RES(2)..(2)3-((2-naph-
thyl) alanineMOD_RES(5)..(5)citrullineMOD_RES(7)..(7)D-glutamic
acidMOD_RES(11)..(11)citrullineMOD_RES(13)..(13)c' amidated 35Xaa
Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103614PRTArtificialsynthetic
peptideMOD_RES(1)..(1)4-fluorobenzoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl) alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)amidated 36Xaa Arg
Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103714PRTArtificialsynthetic
peptideMOD_RES(1)..(1)2-fluorobenzoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl) alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 37Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103813PRTArtificialsynthetic
peptideMOD_RES(1)..(1)5-aminopentanoyl-arginineMOD_RES(2)..(2)3-((2-napht-
hyl) alanineMOD_RES(5)..(5)citrullineMOD_RES(7)..(7)D-glutamic
acidMOD_RES(11)..(11)citrullineMOD_RES(13)..(13)c' amidated 38Xaa
Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
103914PRTArtificialsynthetic
peptideMOD_RES(1)..(1)2-desamino-arginylMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 39Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104013PRTArtificialsynthetic
peptideMOD_RES(1)..(1)Guanyl-arginineMOD_RES(2)..(2)3-((2-naphthyl)
alanineMOD_RES(5)..(5)citrullineMOD_RES(7)..(7)D-glutamic
acidMOD_RES(11)..(11)citrullineMOD_RES(13)..(13)c' amidated 40Xaa
Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104113PRTArtificialsynthetic
peptideMOD_RES(1)..(1)Succinyl-arginineMOD_RES(2)..(2)3-((2-naphthyl)
alanineMOD_RES(5)..(5)citrullineMOD_RES(7)..(7)D-glutamic
acidMOD_RES(11)..(11)citrullineMOD_RES(13)..(13)c' amidated 41Xaa
Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104213PRTArtificialsynthetic
peptideMOD_RES(1)..(1)Glutaryl-arginineMOD_RES(2)..(2)3-((2-naphthyl)
alanineMOD_RES(5)..(5)citrullineMOD_RES(7)..(7)D-glutamic
acidMOD_RES(11)..(11)citrullineMOD_RES(13)..(13)c' amidated 42Xaa
Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104314PRTArtificialsynthetic
peptideMISC_FEATURE(1)..(1)desaminoTMG-APA (formula IV in the
specification)MOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 43Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104414PRTArtificialsynthetic peptideMISC_FEATURE(1)..(1)R-CH2 -
formula (V) in the specificationMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMOD_RES(14)..(14)c' amidated 44Xaa
Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104513PRTArtificialsynthetic peptideMOD_RES(2)..(2)3-((2-naphthyl)
alanineMOD_RES(5)..(5)citrullineMOD_RES(7)..(7)D-glutamic
acidMOD_RES(11)..(11)citrullineMOD_RES(13)..(13)c' amidated 45Arg
Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104614PRTArtificialsynthetic
peptideMOD_RES(1)..(1)tetramethylguanyl-arginineMOD_RES(3)..(3)3-((2-naph-
thyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-citrullineMOD_RES(1-
2)..(12)citrullineMOD_RES(14)..(14)C' AMIDATED 46Xaa Arg Xaa Cys
Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104714PRTArtificialsynthetic
peptideMOD_RES(1)..(1)6-aminohexanoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-citrullineMOD_RES(12)-
..(12)citrullineMOD_RES(14)..(14)C' AMIDATED 47Xaa Arg Xaa Cys Tyr
Xaa Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5 104814PRTArtificialsynthetic
peptideMOD_RES(1)..(1)6-aminohexanoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl) alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 48Xaa
Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
104914PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrullineMOD_RES(14)..(14)C' amidated 49Arg Arg Xaa Cys Tyr Xaa Arg
Xaa Pro Tyr Arg Xaa Cys Arg1 5 105014PRTArtificialsynthetic
peptideMOD_RES(1)..(1)N' ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrullineMOD_RES(14)..(14)C' amidated 50Arg Arg Xaa Cys Tyr Xaa Arg
Xaa Pro Tyr Arg Xaa Cys Arg1 5 105114PRTArtificialSYNTHETIC
PEPTIDEMOD_RES(1)..(1)N' ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrullineMOD_RES(14)..(14)C' AMIDATED 51Arg Arg Xaa Cys Tyr Xaa Lys
Xaa Pro Tyr Arg Xaa Cys Arg1 5 105214PRTArtificialsynthetic
peptideMOD_RES(1)..(1)N' ACETYLATEDMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(12)citrullineMOD_RES(14).-
.(14)C' AMIDATED 52Arg Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa
Cys Arg1 5 105314PRTArtificialsynthetic
peptideMOD_RES(1)..(1)4-fluorobenzoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl) alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMISC_FEATURE(14)..(14)derivatization
by a NH-methyl group 53Xaa Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg
Xaa Cys Arg1 5 105414PRTArtificialsynthetic
peptideMOD_RES(1)..(1)4-fluorobenzoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl) alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMISC_FEATURE(14)..(14)derivatization
by a NH-ethyl group 54Xaa Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg
Xaa Cys Arg1 5 105514PRTArtificialsynthetic
peptideMOD_RES(1)..(1)4-fluorobenzoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl) alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMISC_FEATURE(14)..(14)derivatization
by NH-isopropyl 55Xaa Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa
Cys Arg1 5 105614PRTArtificialsynthetic
peptideMOD_RES(1)..(1)4-fluorobenzoyl-arginineMOD_RES(3)..(3)3-((2-naphth-
yl) alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-glutamic
acidMOD_RES(12)..(12)citrullineMISC_FEATURE(14)..(14)derivatization
with a tyramine residue 56Xaa Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr
Arg Xaa Cys Arg1 5 105714PRTArtificialsynthetic
peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 57Ala Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
105814PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 58Arg Arg
Xaa Cys Tyr Ala Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
105914PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 59Arg Arg
Xaa Cys Tyr Arg Ala Xaa Pro Tyr Arg Xaa Cys Arg1 5
106014PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-alanineMOD_RES(12)..(12)citrulline 60Arg
Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
106114PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 61Arg Arg
Xaa Cys Tyr Arg Lys Xaa Ala Tyr Arg Xaa Cys Arg1 5
106214PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 62Arg Arg
Xaa Cys Tyr Arg Lys Xaa Pro Ala Arg Xaa Cys Arg1 5
106314PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 63Arg Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Ala Xaa Cys Arg1 5
106414PRTArtificialsynthetic
peptideMOD_RES(1)..(1)citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 64Xaa Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
106514PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrulline 65Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys
Arg1 5 106614PRTArtificialsynthetic
peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrullineMOD_RES(14)..(14)C' AMIDATED 66Arg Arg Xaa Cys Tyr Xaa Lys
Xaa Pro Tyr Arg Xaa Cys Arg1 5 106714PRTArtificialsynthetic
peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(7)..(7)citrullineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)c-
itrulline 67Arg Arg Xaa Cys Tyr Arg Xaa Xaa Pro Tyr Arg Xaa Cys
Arg1 5 106814PRTArtificialsynthetic
peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(-
12)citrullineMOD_RES(14)..(14)C' AMIDATED 68Arg Arg Xaa Cys Tyr Xaa
Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5 106914PRTArtificialsynthetic
peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-lysineMOD_RES(12)..(12)citrulline 69Arg Arg
Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
107014PRTArtificialsynthetic peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(6)..(6)citrullineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(-
12)citrulline 70Arg Arg Xaa Cys Tyr Xaa Lys Xaa Pro Tyr Arg Xaa Cys
Arg1 5 107114PRTArtificialsynthetic
peptideMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(12)citrulline 71Arg
Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa Cys Arg1 5
107214PRTArtificialsynthetic
peptideMOD_RES(1)..(1)citrullineMOD_RES(3)..(3)3-((2-naphthyl)
alanineMOD_RES(8)..(8)D-citrullineMOD_RES(12)..(12)citrullineMOD_RES(14).-
.(14)C' Amidated 72Xaa Arg Xaa Cys Tyr Arg Lys Xaa Pro Tyr Arg Xaa
Cys Arg1 5 10
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.