Lubricating Compositions
Sampler; Edward P. ; et al.
U.S. patent application number 16/305083 was filed with the patent office on 2020-12-03 for lubricating compositions. The applicant listed for this patent is The Lubrizol Corporation. Invention is credited to Stephen J. Cook, Samuel M. Kilsby, Blayne M. McKenzie, Edward P. Sampler, Gary M. Walker.
| Application Number | 20200377817 16/305083 |
| Document ID | / |
| Family ID | 1000005060856 |
| Filed Date | 2020-12-03 |











View All Diagrams
| United States Patent Application | 20200377817 |
| Kind Code | A1 |
| Sampler; Edward P. ; et al. | December 3, 2020 |
Lubricating Compositions
Abstract
A lubricating composition suited for use in lubricating a driveline device includes an oil of lubricating viscosity and a compound comprising a polyolefin-substituted bridged hydroxyaromatic compound or metal salt thereof, wherein the polyolefin is derived from a isobutylene having a number average molecular weight of 150 to 800.
| Inventors: | Sampler; Edward P.; (Belper, Derby, GB) ; Walker; Gary M.; (Belper, Derby, GB) ; Cook; Stephen J.; (Belper, Derby, GB) ; Kilsby; Samuel M.; (Belper, Derby, GB) ; McKenzie; Blayne M.; (Willoughby Hills, OH) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000005060856 | ||||||||||
| Appl. No.: | 16/305083 | ||||||||||
| Filed: | June 14, 2017 | ||||||||||
| PCT Filed: | June 14, 2017 | ||||||||||
| PCT NO: | PCT/US2017/037458 | ||||||||||
| 371 Date: | November 28, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10M 2203/003 20130101; C10M 169/044 20130101; C10M 141/10 20130101; C10M 161/00 20130101; C10M 135/04 20130101; C10N 2040/044 20200501; C10N 2030/04 20130101; C10M 2219/022 20130101; C10M 143/18 20130101; C10N 2040/042 20200501; C10M 135/10 20130101; C10M 2205/12 20130101; C10M 2223/045 20130101; C10N 2020/04 20130101; C10M 2219/044 20130101; C10M 137/02 20130101; C10M 137/10 20130101; C10M 2223/049 20130101; C10N 2030/06 20130101 |
| International Class: | C10M 169/04 20060101 C10M169/04; C10M 143/18 20060101 C10M143/18; C10M 135/10 20060101 C10M135/10; C10M 137/10 20060101 C10M137/10; C10M 137/02 20060101 C10M137/02; C10M 135/04 20060101 C10M135/04; C10M 141/10 20060101 C10M141/10; C10M 161/00 20060101 C10M161/00 |
Claims
1. A lubricating composition comprising: an oil of lubricating viscosity; a dispersant; and a compound comprising a polyolefin-substituted bridged hydroxyaromatic compound or salt thereof, wherein the polyolefin is derived from a branched alkene having at least 4 carbon atoms and wherein the polyolefin has a number average molecular weight of 150 to 800.
2. The lubricating composition of claim 1, wherein the branched alkene comprises isobutylene.
3. The lubricating composition of claim 1, wherein the polyolefin includes a chain derived from at least 4, or at least 5, or up to 6, or up to 7, or up to 8, or up to 18 branched alkene units.
4. The lubricating composition claim 1, wherein the polyolefin has a number average molecular weight of 150 to 400.
5. The lubricating composition of claim 1, wherein the compound is a metal salt or pnictogen salt.
6. The lubricating composition claim 1, wherein the polyolefin is prepared in the absence of a chain transfer agent and has a polydispersity greater than 1.5.
7. The lubricating composition of claim 6, wherein the metal in the salt comprises calcium.
8. The lubricating composition of claim 1, wherein the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof is overbased.
9. The lubricating composition of claim 1, wherein the compound is bridged with a sulfide bridge.
10. The lubricating composition of claim 1, wherein the compound is bridged with an alkylene bridge.
11. The lubricating composition of claim 1, wherein the compound is a bridged metal phenate.
12. The lubricating composition of claim 1, wherein the compound is free of C.sub.8 and higher unbranched alkyl chains.
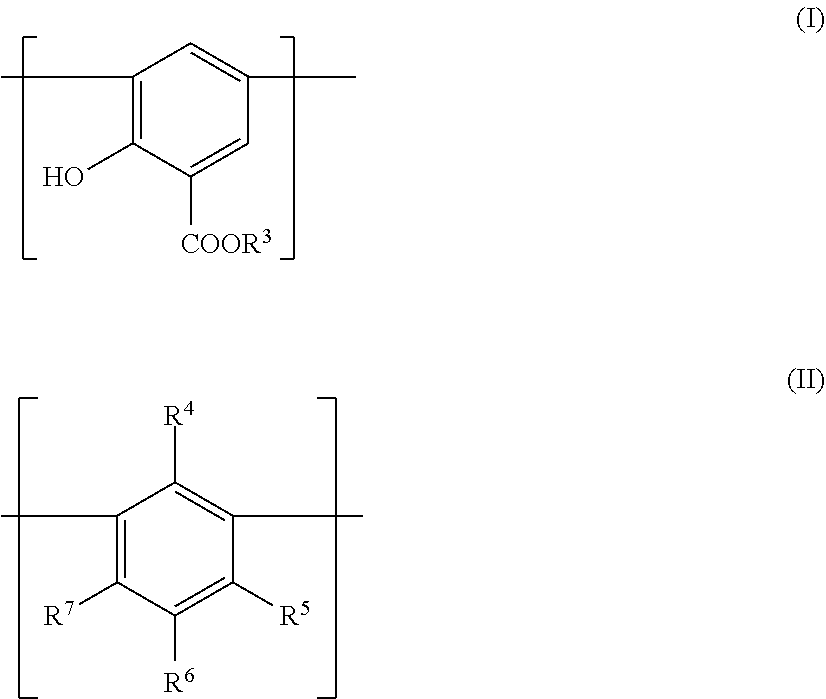
13. The lubricating composition of claim 1, wherein the compound includes a bridged phenate unit represented by the formula: ##STR00017## and salts thereof, where each R1 and each R2 represents a polyolefin group; X represents a bridging group; Y and Z each represent a terminal group; each n is at least 1; m is at least 1; and p is at least 1.
14. The lubricating composition of claim 13, wherein the compound is not coupled with cresol.
15. The lubricating composition of claim 13, wherein n is 1.
16. The lubricating composition of claim 1, wherein each R1 and R2 is independently a polyolefin of 8 to 50 carbon atoms, or at least 10 carbon atoms, or at least 10 carbon atoms, or at least 12 carbon atoms, or at least 14 carbon atoms, or at least 16 carbon atoms, or at least 18 carbon atoms, or at least 20 carbon atoms, or at least 24 carbon atoms, or up to 40 carbon atoms, or up to 35 carbon atoms, or up to 30 carbon atoms.
17. The lubricating composition of claim 1, wherein each R1 and each R2 is a polyisobutylene.
18. The lubricating composition of claim 1, wherein each R1 is a 300-400 MW polyisobutylene.
19. The lubricating composition of claim 1, wherein the compound has a weight-average molecular weight of 550-5600 in its unsalted form.
20. The lubricating composition of claim 1, wherein the compound has a weight average molecular weight of up to 2800 in its unsalted form.
21. The lubricating composition of claim 1, wherein the compound is at least 0.01 wt. % of the lubricating composition.
22. The lubricating composition of claim 21, wherein the compound is at least 0.1 wt. %, or at least 0.5 wt. %, or at least 1 wt. %, or at least 2 wt. % of the lubricating composition.
23. The lubricating composition of claim 21, wherein the compound is up to 20 wt. %, or up to 10 wt. %, or up to 5 wt. %, or up to 3 wt. %, or up to 2.5 wt. % of the lubricating composition.
24. The lubricating composition of claim 1, wherein the oil of lubricating viscosity comprises at least one of an API Group I, II, III, IV, and V base oil.
25. The lubricating composition of claim 1, wherein the oil of lubricating viscosity is at least 10 wt. % of the lubricating composition.
26. The lubricating composition of claim 25, wherein the oil of lubricating viscosity is at least 30 wt. % of the lubricating composition.
27. The lubricating composition of claim 1, wherein the oil of lubricating viscosity is up to 95 wt. % of the lubricating composition.
28. The lubricant claim 1 further comprising at least one of a phosphorus-containing antiwear agent comprising a non-ionic phosphorus compound; an amine salt of a phosphorus compound; a calcium-containing detergent; another phenate detergent; a sulphur-containing extreme pressure agent; a sulphur-containing corrosion inhibitor; or combinations thereof.
29. The lubricating composition claim 1 further comprising from about 0.05 to about 3 wt % of a C2-C18 di- or tri-hydrocarbyl phosphite, based on the total weight of said lubricant.
30. The lubricating composition claims 1 further comprising less than 2000 ppm zinc based on a total weight of said lubricant.
31. The lubricating composition of claim 1, wherein the lubricant is substantially free of to free of a phenol alkylated with oligomers of propylene.
32. A method of lubricating a manual transmission comprising supplying the lubricant composition of claim 1 to said manual transmission.
33. The method of claim 32, wherein the lubricating composition further comprises: a thiadiazole and a phosphorous containing antiwear agent.
34. A method of lubricating automatic transmission comprising supplying the lubricating composition of claim 1 to said automatic transmission.
35. The method of claim 34, wherein the lubricating composition further comprises: a thiadiazole and a phosphorous containing antiwear agent.
36. A method of lubricating an off-highway vehicle comprising supplying the lubricating composition of claim 1 to said off-highway vehicle.
37. The method of claim 36, wherein the lubricating composition further comprises: a zinc dialkyl dithiophosphate antiwear agent
38. The method of claim 37, wherein the vehicle is a farm-tractor vehicle or a construction vehicle.
39. A method of lubricating a highway vehicle comprising supplying the lubricating composition of claim 1 to said off-highway vehicle.
40. The method of claim 39, wherein the lubricating composition further comprises: a sulfurized olefin; a thiadiazole; and a phosphorous containing antiwear agent.
41. A lubricating composition comprising: an oil of lubricating viscosity; a dispersant; a sulfurized olefin; a thiadazole; a phosphorous-containing anti-wear agent; and a bridged phenate unit represented by the formula ##STR00018## and salts thereof, where each R.sup.1 and each R.sup.2 represents a polyolefin group having a number average molecular weight of 200 to 500, or 300 to 400; X represents a bridging group; Y and Z each represent a terminal group; each n is at least 1; m is at least 1; and p is at least 1.
Description
BACKGROUND
[0001] The invention relates generally to lubricating compositions (lubricants) and finds particular application in connection with an additive for a lubricating composition and to a lubricating method.
[0002] Phenol-based detergents are known. Among these are phenates based on phenolic monomers linked with sulfur bridges or alkylene bridges such as methylene linkages derived from formaldehyde. The phenolic monomers themselves are typically substituted with an aliphatic hydrocarbyl group to provide a measure of oil solubility. The hydrocarbyl groups may be alkyl groups, such as dodecylphenol (or propylene tetramer-substituted phenol). Basic sulfurized polyvalent metal phenates are described, for example, in U.S. Pat. No. 2,680,096, to Walker et al., issued Jun. 1, 1954; and U.S. Pat. No. 3,372,116, to Meinhardt, issued Mar. 6, 1968.
[0003] Recently, however, certain alkylphenols and products prepared from them have come under increased scrutiny due to their classification as potentially harmful materials. In particular, alkylphenol detergents which are based on oligomers of C.sub.12 alkyl phenols may contain residual monomeric C.sub.12 alkyl phenol species. These detergents include those derived from para-dodecyl phenol. There is interest, therefore, in developing alkyl-substituted phenate detergents, for uses in lubricants, fuels, and as industrial additives, which reduce or eliminate the need for dodecylphenol-based compounds.
[0004] There have been several efforts to prepare phenate detergents that do not contain C.sub.n alkyl phenols. U.S. Pat. No. 7,435,709, to Stonebraker, et al., issued Oct. 14, 2008, discloses a linear alkylphenol-derived detergent which is a salt of a reaction product of an olefin having at least 10 carbon atoms and a hydroxyaromatic compound. Greater than 90 mole % of the olefin is a linear C.sub.20-C.sub.30 n-alpha olefin, less than 10 mole % of the olefin is a linear olefin of less than 20 carbon atoms, and less than 5 mole % of the olefin is a branched chain olefin of 18 carbons or less.
[0005] U.S. Pub. No. 2011/0190185, to Sinquin, et al, published Aug. 4, 2011, discloses an overbased salt of an oligomerized alkylhydroxyaromatic compound. The alkyl group is derived from an olefin mixture comprising propylene oligomers having an initial boiling point of at least 195.degree. C. and a final boiling point of greater than 325.degree. C. The propylene oligomers may contain a distribution of carbon atoms that include at least 50 wt. % C.sub.14 to C.sub.20 carbon atoms.
[0006] U.S. Pub. No. 2011/0124539, to Sinquin, et al, published May 26, 2011, discloses an overbased, sulfurized salt of an alkylated hydroxyaromatic compound. The alkyl substituent is a residue of at least one isomerized olefin having from 15 to 99 wt. % branching. The hydroxyaromatic compound may be a phenol, cresol, xylenol, or mixture thereof.
[0007] U.S. Pub. No. 2011/0118160, to Campbell, et al., published May 19, 2011, discloses an alkylated hydroxyaromatic compound prepared by reacting a hydroxyaromatic compound with at least one branched olefinic propylene oligomer having from 20 to 80 carbon atoms. Suitable hydroxyaromatic compounds include phenol, catechol, resorcinol, hydroquinone, pyrogallol, cresol, and the like.
[0008] U.S. Pub. No. 2010/0029529, to Campbell, et al., published Feb. 4, 2010, discloses an overbased salt of an oligomerized alkylhydroxyaromatic compound. The alkyl group is derived from an olefin mixture comprising propylene oligomers having an initial boing point of at least 195.degree. C. and a final boiling point of no more than 325.degree. C.
[0009] U.S. Pub. No. 2008/0269351, to Campbell, et al., published Oct. 30, 2008, discloses an alkylated hydroxyaromatic compound prepared by reacting a hydroxyaromatic compound with a branched olefinic oligomer having from 20 to 80 carbon atoms.
[0010] U.S. Pat. No. 6,310,009, to Carrick, et al., issued Oct. 30, 2001, discloses bridged phenolic compounds, each phenol group being substituted with an alkyl group of 1 to 60 carbon atoms, e.g., 9 to 18 carbon atoms.
[0011] There remains a need for a phenolic material with appropriate oil solubility, viscosity performance, and detergency (characteristic of moderate chain length alkyl groups) but free from or substantially free from C.sub.12 alkyl phenol moieties.
BRIEF DESCRIPTION
[0012] In accordance with one aspect of the exemplary embodiment, a lubricating composition includes an oil of lubricating viscosity and a compound containing a polyolefin-substituted bridged hydroxyaromatic compound or metal salt thereof.
[0013] In accordance with another aspect of the exemplary embodiment, a method of lubricating a mechanical device with the lubricating composition is disclosed.
[0014] In accordance with another aspect of the exemplary embodiment, a method of forming a lubricating composition including an oil of lubricating viscosity and a compound containing a polyolefin-substituted bridged hydroxyaromatic compound or metal salt thereof is described.
[0015] In accordance with another aspect, a use of the composition for lubricating a mechanical device is also disclosed.
DETAILED DESCRIPTION
[0016] Aspects of the exemplary embodiment relate to a lubricating composition, method of use and method of manufacturing the composition. The lubricating composition contains an oil of lubricating viscosity and a polyolefin-substituted bridged hydroxyaromatic compound or salt thereof.
[0017] The exemplary compounds described herein are bridged polyolefin-substituted hydroxyaromatic (e.g., phenol) compounds and their salts. The compounds are free from or substantially free from unsubstituted phenol moieties. Such compounds and their salts are useful as lubricant additives.
[0018] Suitable hydroxyaromatic compounds include phenol, catechol, resorcinol, hydroquinone, pyrogallol, cresol, and the like. In one embodiment, the hydroxyaromatic compounds that are bridged include no more than one aromatic ring.
[0019] The term "substantially free" as it refers to the exemplary compound, means that less than 0.01 mol. %, or less than 0.001 mol. %, or less than 0.0001 mol. % of the exemplary polyolefin-substituted bridged hydroxyaromatic compound in the lubricating composition is substituted with one or more C.sub.12 alkyl groups. In one embodiment, the lubricating compound contains less than 0.5 wt. %, or less than 0.1 wt. %, or less than 0.01 wt. %, or less than 0.001 wt. %, of C.sub.12 alkyl hydroxyaromatic compounds.
[0020] The lubricating composition may also include a bridged phenate. In the exemplary embodiment, the compound is not coupled with cresol. The compound can serve as a dispersant or detergent in the lubricating composition.
[0021] A. The Polyolefin-Substituted Bridged Hydroxyaromatic Compound
[0022] The exemplary polyolefin-substituted, bridged hydroxyaromatic compound is a bridged phenol in which at least one hydroxyl group is directly bonded to an aromatic ring that is substituted with a polyolefin group.
[0023] The aromatic polyol on which the exemplary compound is based may be a substituted or unsubstituted compound that has at least one hydroxyl group directly bonded to an aromatic group (within the definition of HUckel Rule 47+2 electrons) such as phenol. An exemplary polyolefin-substituted aromatic polyol compound may be represented by the general structure shown in Formula I:
##STR00001##
[0024] and salts thereof,
[0025] where each R.sup.1 and each R.sup.2 represents a polyolefin group,
[0026] X represents a bridging group, such as a sulfur or an alkylene bridging group containing 1 to 8 carbon atoms (e.g., a bridge derived from one or more aldehyde and/or propanal monomer units);
[0027] Y and Z each represent a terminal group, such as --H, --OH, a C.sub.1-C.sub.6 alkyl group, or a group derived from the bridging monomer (e.g., --SH, an aldehyde-derived group, such as --C(H).dbd.O, or the like);
[0028] each n is at least 1, such as up to 3, on average, or up to 2, or 1;
[0029] m is at least 1, such as up to 5, or up to 4, or up to 3, on average, such as 1 or 2; and
[0030] p is at least 1, such as up to 5, on average, or 1 or 2. For example, when X is an alkylene bridge, p is 1, 2, or 3, for example, 1 or 2, further for example 1. Further for example, when X is sulfur, p is 1 or 2 or 3.
[0031] An exemplary salt may be represented by the general structure shown in Formula II:
##STR00002##
[0032] where M represents a cation which is linked to at least one of the O.sup.- groups; and x is at least 1, such as 2 in the case of a calcium ion.
[0033] The exemplary compound of Formula II may also be associated with a counter ion in the compound. For example, in an overbased compound, the metal ion to compound ratio may be raised above the stoichiometric amount, e.g., by bubbling CO.sub.2 through the mixture to provide a carbonate counterion. As will be appreciated, these aspects can also be used in combinations thereof.
[0034] Examples of branched polyolefin groups suitable for use as R.sup.1 and R.sup.2 include polyolefin groups which are derived from a branched alkene having at least 4 carbon atoms, or up to 12 carbon atoms, or up to 8 carbon atoms, or up to 6 carbon atoms, such as a C.sub.4-C.sub.6 branched alkene. Suitable branched alkenes include isobutylene (2-methylpropene), 2-methylbutene, 2-ethyl-1-butene, 2-methyl-1-pentene, 3-methyl-1-pentene, 4-methyl-l-pentene, 2-methyl, 3-methyl-1-pentene, 2-ethyl-1 -pentene, 3-ethyl-1 -pentene, 2-methyl-1-hexene, 3-methyl-1-hexene, 2-ethyl-1-hexene, 3-ethyl-1-hexene, 4-ethyl-1-hexene, 2-methyl-1-heptene, 3-methyl-1-heptene, 2-methyl-1-octene, 2-methyl-1-nonene, 2-methyl-1-decene, 2-methyl-1-undecene, and mixtures thereof. Each polyolefin group is derived from at least two or at least three, or at least four, or up to twenty, or up to eighteen, or up to twelve branched alkene monomer units to form a chain with at least two or at least three, or at least four branches from the main chain. In one embodiment, the polyolefin includes a chain derived from at least four, or at least five, or up to eighteen, or up to eight, or up to seven, or up to six branched alkene units. The branched alkene may be branched at the alpha or beta position, or further along the longest chain. In one embodiment, at least one or both of R.sup.1 and R.sup.2 are derived from a branched alkene which is solely or at least partially isobutylene and at least one or both of R.sup.1 and R.sup.2 is/are each polyisobutylene (PIB).
[0035] R.sup.1 and R.sup.2 may be independently a polyolefin of 8 to 50 carbon atoms, or at least 10 carbon atoms, or at least 12 carbon atoms, or at least 14 carbon atoms, or at least 16 carbon atoms, or at least 18 carbon atoms, or at least 20 carbon atoms, or at least 24 carbon atoms, or up to 40 carbon atoms, or up to 35 carbon atoms, or up to 30 carbon atoms.
[0036] The polyolefin group may have a number-average molecular weight Mn of at least 150, or at least 200, or at least 300, or up to 800, or up to 600, or up to 500, or up to 400, or up to 360. In some embodiments R.sub.1 and R.sub.2 may have different chain lengths. In the exemplary embodiment, R.sub.1 and R.sub.2 are of the same chain length (or have the same number of branches off the main chain or differ by no more than .+-.1 branch). A polyolefin group with M.sub.n of less than 500, e.g., up to about 400 is particularly suitable as it allows the compound to provide good detergent properties for deposit control and cleanliness without resulting in viscosity creep or undesirable thickening of the oil.
[0037] In one embodiment, the compound may be a mixture of polyolefin-substituted, bridged hydroxyaromatic compounds with high and low molecular weight polyolefin groups, respectively. The low molecular weight polyolefin may be up to a C.sub.30 or up to a C.sub.25 polyolefin. The high molecular weight polyolefin may be up to a C.sub.40 or up to a C.sub.35 polyolefin. A ratio of the compound with low molecular weight polyolefin to the compound with high molecular weight polyolefin may be at least 50:50 by weight, or at least 80:20, or at least 90:10.
[0038] In one embodiment, the compound is free of C.sub.8 and higher unbranched alkyl groups.
[0039] In one embodiment, R.sub.1 and R.sub.2 are each composed solely of carbon and hydrogen.
[0040] In one embodiment, the compound consists solely of elements selected from the group consisting of C, H, O, S and the counterion(s).
[0041] The compound may have a weight average molecular weight Mw of at least 550, or at least 700, or at least 750, or at least 800 in its unsalted form. The weight average molecular weight of the compound may be up to 5600, or up to 2800, or up to 1200, or up to 1000, or up to 950, in its unsalted form.
[0042] In one embodiment X is an alkylene, e.g., a methylene bridge, or a sulfur bridge. In the case of an alkylene bridge, the bridge may be up to 4 carbons in length, or up to 3 carbons in length.
[0043] As will be appreciated, these aspects can also be used in combinations thereof.
[0044] The salt of the compound of Formula I may be formed by reacting a cation or source of the cation with the compound. The compound of Formula I thus serves as the anion (or "substrate") in the salt. The cation or source thereof reacts with one or more of the residual OH groups to form a neutral or overbased salt of the above-described coupled polyolefin-substituted aromatic polyol.
[0045] In one embodiment, the cation is a metallic cation. The metallic cation may be derived from an alkaline earth metal, such as calcium, barium or magnesium (typically calcium), or an alkali metal, such as sodium or potassium (typically sodium). The metal cation may have an atomic weight of at least 6 or at least 12.
[0046] Exemplary metal cations include alkali metal cations, alkaline earth metal cations, transition metal cations, and combinations thereof. Examples of metal cations include Li.sup.+, Na.sup.+, K.sup.+, Rb.sup.+, Cs.sup.+, Be.sup.2+, Mg.sup.2+, Ca.sup.2+, Sr.sup.2+, Ba.sup.2+, Sc.sup.3+, Sc.sup.2+, Sc.sup.+, Y.sup.3+, Y.sup.2+, Y.sup.+, Ti.sup.4+, Ti.sup.3+, Ti.sup.2+, Zr.sup.4+, Zr.sup.3+, Zr.sup.2+, Hf.sup.4+, Hf.sup.3+, V.sup.4+, V.sup.3+, V.sup.2+, Nb.sup.4+, Nb.sup.3+, Nb.sup.2+, Ta.sup.4+, Ta.sup.3+, Ta.sup.2+, Cr.sup.4+, Cr.sup.3+, Cr.sup.2+, Cr.sup.+, Mo.sup.4+, Mo.sup.3+, Mo.sup.2+, Mo.sup.+, W.sup.4+, W.sup.3+, W.sup.2+, W.sup.+, Mn.sup.4+, Mn.sup.3+, Mn.sup.2+, Mn.sup.+, Re.sup.4+, Re.sup.3+, Re.sup.2+, Re.sup.+, Fe.sup.6+, Fe.sup.4+, Fe.sup.3+, Fe.sup.2+, Fe.sup.+, Ru.sup.4+, Ru.sup.3+, Ru.sup.2+, Os.sup.4+, Os.sup.3+, Os.sup.2+, Os.sup.+, Co.sup.5+, Co.sup.4+, Co.sup.3+, Co.sup.2+, Co.sup.+Rh.sup.4+, Ru.sup.3+, Rh.sup.2+, Rh.sup.+, 10.sup.+, Ir.sup.3+, Ir.sup.2+, Ir.sup.+, Ni.sup.3+, Ni.sup.2+, Ni.sup.+, Pd.sup.4+, Pd.sup.2+, Pd.sup.+, Pt.sup.4+, Pt.sup.3+, Pt.sup.2+, Pt.sup.+, Cu.sup.4+, Cu.sup.3+, Cu.sup.2+, Cu.sup.+, Ag.sup.3+, Ag.sup.2+, Ag.sup.+, Au.sup.4+, Au.sup.3+, Au.sup.2+, Au.sup.+, Zn.sup.2+, Zn.sup.+, Cd.sup.2+, Cd.sup.+, Hg.sup.4+, Hg.sup.2+, Hg.sup.+, Al.sup.3+, Al.sup.2+, Al.sup.+, Ga.sup.3+, Ga.sup.+, ln.sup.3+, ln.sup.2+, Tl.sup.3+, Tl.sup.+, Si.sup.4+, Si.sup.3+, Si.sup.2+, Si.sup.+, Ge.sup.4+, Ge.sup.3+, Ge.sup.2+, Ge.sup.+, Sn.sup.4+, Sn.sup.2+, Pb.sup.4+, Pb.sup.2+, As.sup.3+, As.sup.2+, As.sup.+, Sb.sup.3+, Bi.sup.3+, Te.sup.4+, Te.sup.2+, La.sup.3+, La.sup.2+, Ce.sup.4+, Ce.sup.3+, Ce.sup.2+, Pr.sup.4+, Pr.sup.3+, Pr.sup.2+, Nd.sup.3+, Nd.sup.2+, Sm.sup.3+, Sm.sup.2+, Eu.sup.3+, Eu.sup.2+, Gd.sup.3+, Gd.sup.2+, Gd.sup.+, Tb.sup.4+, Tb.sup.3+, Tb.sup.2+, Tb.sup.+, Db.sup.3+, Db.sup.+.sup.+, Ho.sup.3+, Er.sup.3+, Tm.sup.4+, Tm.sup.3+, Tm.sup.2+, Yb.sup.3+, Yb.sup.2+, and Lu.sup.3+. Particularly useful are those which form stable salts, i.e., which do not decompose by more than a minor amount over the expected lifetime and operating conditions of the lubricating composition.
[0047] In one embodiment, the metallic cation is derived from a metal base such as a metal base of a hydroxide, an oxide, carbonate, or bicarbonate. The metal base may be a hydroxide or an oxide. For example the metallic cation may be derived from calcium hydroxide, calcium oxide, sodium hydroxide, sodium oxide, magnesium hydroxide, magnesium oxide, or mixture thereof.
[0048] In one embodiment, the cation is an ash-free cation. An ash-free (ashless) organic cation is an organic ion that does not contain ash-forming metals. In one embodiment, the compound in the salt form has a sulfated ash of up to 0.5 wt. %, or up to 0.4 wt. %, according to ASTM D874.
[0049] In one embodiment, the cation is a pnictogen cation. As used herein the term "pnictogens" includes the elements in column 15 of the periodic table. The non-metallic pnictogens include nitrogen and phosphorus (typically nitrogen). The pnictogen cation may be derived from a source of the cation containing a primary amine, a secondary amine, a tertiary amine, or mixture thereof. In one embodiment, the amine salt may be derived from a secondary or tertiary amine.
[0050] When the cation is pnictogen cation derived from an amine or ammonium compound, the pnictogen cation (or the amine from which it is derived) may have molecular weight of at least 260 g/mol, or at least 300 g/mol or at least 350 g/mol, or at least 500 g/mol.
[0051] The pnictogen cation may be derived from a mono-, di-, or tri-substituted amine. Specific examples include primary alkylamines, such as methylamine, ethylamine, n-propylamine, n-butylamine, n-hexylamine, n-octylam ine, 2-ethylhexylamine, benzylamine, 2-phenylethylamine, cocoamine, oleylamine, and tridecylamine (CAS #86089-17-0); secondary and tertiary alkylamines such as isopropylamine, sec-butylamine, t-butylamine, cyclopentylamine, cyclohexylamine, and 1-phenylethylamine; dialkylamines, such as dimethylamine, diethylamine, dipropylamine, diisopropylamine, dibutylamine, dicyclohexylamine, di-(2-ethylhexyl)amine, dihexylamine, ethylbutylamine, N-ethylcyclohexylamine, and N-methylcyclohexylamine; cycloalkylamines, such as piperidine, N-ethylpiperidine, N,N'-dimethylpiperazine, morpholine, N-methylmorpholine, N-ethylmorpholine, N-methylpiperidine, pyrrolidine, N-methylpyrrolidine, and N-ethylpyrrolidine; and trialkylamines, such as trimethylamine, triethylamine, tripropylamine, triisopropylamine, tri-n-butylamine, trihexylamine, N, N-dimethylbenzylamine, dimethylethylamine, dimethylisopropylamine, dimethylbutylamine, and N,N-dimethylcyclohexylamine.
[0052] When the pnictogen cation includes at least one hydrocarbyl group (a quaternary ammonium ion), the pnictogen cation may be an ashless organic cation. Example ammonium cations of this type include N-substituted long chain alkenyl succinimides and aliphatic polyamines. N-substituted long chain alkenyl succinimides useful herein may be derived from an aliphatic polyamine, or mixture thereof. The aliphatic polyamine may be aliphatic polyamine such as an ethylenepolyamine, a propylenepolyamine, a butylenepolyamine, or mixture thereof. Examples of N-substituted long chain alkenyl succinimides include polyisobutylene succinimide with number average molecular weight of the polyisobutylene substituent of at least 350, or at least 500, or at least 550, or at least 750, and can be up to 5000, or up to 3000, or up to 2500. Such succinim ides can be formed, for example, from high vinylidene polyisobutylene and maleic anhydride.
[0053] Example N-substituted long chain alkenyl succinimides useful herein as pnictogen cations include those derived from succinimide dispersants, which are more fully described in U.S. Pat. Nos. 3,172,892, 3,219,666, 3,316,177, 3,340,281, 3,351,552, 3,381,022, 3,433,744, 3,444,170, 3,467,668, 3,501,405, 3,542,680, 3,576,743, 3,632,511, 4,234,435, RE 26,433, 6,165,235, 7,238,650, and EP Patent Application 0 355 895 A.
[0054] Example aliphatic polyamines useful as the pnictogen cation include ethylenepolyamines, propylenepolyamines, butylenepolyamines, and mixtures thereof. Example ethylenepolyamines include ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, pentaethylene-hexamine, polyamine still bottoms, and mixtures thereof.
[0055] In one embodiment, the exemplary polyolefin-substituted bridged hydroxyaromatic compound salt may be overbased, i.e., contain an excess of the metal cation in relation to the number of hydroxyl groups present in the compound. There are two common measures of basicity that are commonly used in the field of lubricant additives: Total Base Number (TBN), as measured by ASTM D2896, is a titration that measures both strong and weak bases, while ASTM D4739-11 "Standard Test Method for Base Number Determination by Potentiometric Hydrochloric Acid Titration," (BN) is a titration that measures strong bases but does not readily titrate weak bases, such as certain amines. TBN and BN are expressed as an equivalent in milligrams of potash per gram of oil (mg of KOH/g).
[0056] Total base number (TBN), as used herein, is the quantity of acid, expressed in terms of the equivalent number of milligrams of potassium hydroxide (meq KOH), that is required to neutralize all basic constituents present in 1 gram of a sample of the lubricating oil. The TBN values reported herein are determined according to ASTM Standard D2896-15, "Standard Test Method for Base Number of Petroleum Products by Potentiometric Perchloric Acid Titration" (2015), ASTM International, West Conshohocken, PA, 2003 (hereinafter, "D2896"). In various aspects, the neutral salt compound has a TBN of at least 50 mg of KOH/g, or at least 60 mg of KOH/g on an oil-free basis. The TBN of the neutral salt may be up to 300 mg KOH/g, or up to 250 mg KOH/g, or up to 165 mg KOH/g, on an oil-free basis. In various aspects, the lubricating composition containing the compound has a TBN of at least 3 mg KOH/g, or at least 4 mg of KOH/g, or at least 6 mg of KOH/g.
[0057] The cation may serve as a basic component of the lubricating composition which, in combination with any other basic components of the lubricating composition, may provide the lubricating composition with a TBN of at least 5, or at least 8, or at least 10, or at least 15, or at least 25. The cation itself may have a TBN of at least 10 or at least or at least 15, or at least 25, or at least 50.
[0058] B. Method of Forming the Polyolefin-Substituted Bridged Hydroxyaromatic Compound
[0059] A bridged (e.g., sulfur-coupled or formaldehyde-coupled) polyolefin-substituted hydroxyaromatic compound of Formula I or II may be formed through well-known techniques.
[0060] In one embodiment, the salt of a polyolefin-substituted hydroxyaromatic compound may be obtained/obtainable by (i) reacting a hydroxy aromatic compound (e.g., phenol) with an branched alkene, such as isobutylene, optionally in the presence of a catalyst, to form a hydroxy-substituted intermediate compound, (ii) coupling the intermediate compound, e.g., with sulfur or formaldehyde, and (to form the salt) iii) reacting the bridged compound formed in step (ii) with a metal base or a pnictogen base.
[0061] (i) Formation of Polyisoalkylene-Substituted Phenol Compound
[0062] The intermediate compound may be formed by reaction of a phenol with a poly(branched alkene). Substitution occurs primarily at the para position, although minor amounts of ortho- and/or meta-substitution may occur. The ratio of poly(branched alkene) to phenol may be from 1:1 to 1:6, such as at least 1:2. An excess of phenol helps to ensure primarily mono-substitution of the poly(branched alkene).
[0063] For example, a polyisobutylene-substituted phenol:
##STR00003##
[0064] where q is, for example, from 5 to 10, or up to 7, or at least 6, on average, may be formed by reacting phenol with a polyolefin (e.g., polyisobutylene), optionally in the presence of a base catalyst. Typically the reaction occurs in the presence of a base catalyst.
[0065] Example catalysts include Lewis acid catalysts, solid acid catalysts, trifluoromethanesulfonic acid, and acidic molecular sieve catalysts. Suitable Lewis acid catalysts include aluminum trichloride, aluminum tribromide, aluminum triiodide, boron trifluoride, boron tribromide, boron triiodide and the like. Suitable solid acidic catalysts include zeolites, acid clays, and/or silica-alumina.
##STR00004##
[0066] The process to prepare the intermediate may be carried out a reaction temperature of 1.degree. C. to 52.degree. C., or 5.degree. C. to 20.degree. C., or 10.degree. C. to 15.degree. C. The formation of the intermediate may be performed in the presence or absence of solvent. The solvent may include a hydrocarbon such as hexane, toluene, xylene, diluent oil, cyclohexane, or mixture thereof. In one embodiment the process to prepare the intermediate is performed in the presence of a solvent. Optionally the solvent is removed before sulfurizing and/or reacting of the intermediate with the metal base. The reaction mixture may then be neutralized with calcium hydroxide, followed by addition of diatomaceous earth and ammonia hydroxide to remove residual catalyst. The reaction mixture is filtered and the filtrate heated under vacuum to remove volatiles by distillation.
[0067] In one embodiment, the polyisobutylene is prepared in the absence of a chain transfer agent and has a polydispersity greater than 1.5.
[0068] ii) Coupling the Intermediate Compound with Sulfur or Aldehyde and Formation of Salt
[0069] Sulfurization may be performed by contacting the intermediate compound with a sulfur source which introduces S.sub.x bridging groups between the polyolefin-substituted phenol compound in the presence of a base. Reaction Scheme 2 illustrates the reaction.
##STR00005##
[0070] Any suitable sulfur source can be used such as, for example, elemental sulfur or a halide thereof such as sulfur monochloride, sulfur dichloride, hydrogen sulfide, sulfur dioxide, or a sodium sulfide hydrate. The sulfur can be employed either as molten sulfur or as a solid (e.g., powder or particulate) or as a solid suspension in a compatible hydrocarbon liquid, such as ethylene glycol.
[0071] Sulfur may be employed at from 0.5 to 4 moles per mole of the intermediate compound in the reaction system. In one embodiment, sulfur is employed at from 0.8 to 2 moles per mole of the intermediate compound.
[0072] The temperature range in which the sulfurization reaction is carried out is generally 80-250.degree. C., e.g., 100-220.degree. C. The reaction can be conducted under atmospheric pressure (or slightly lower) or at elevated pressures. During sulfurization a significant amount of by-product hydrogen sulfide gas is evolved. In one embodiment the reaction is carried out under vacuum to facilitate the H.sub.2S elimination.
[0073] Other sulfurization techniques which may be adapted to use herein are described, for example, in U.S. Pat. No. 2,680,096, to Walker et al., issued Jun. 1, 1954; U.S. Pat. No. 3,372,116, to Meinhardt, issued Mar. 6, 1968; U.S. Pat. No. 3,036,971, to Otto, issued May 29, 1962, U.S. Pat. No. 7,435,709, to Stonebraker, et al., issued Oct. 14, 2008, U.S. Pat. No. 8,772,209 to Mahieux, et al., issued Jul. 8, 2014, U.S. Pat. No. 9,062,271 to Jukes, et al., issued Jun. 23, 2015, and U.S. Pub. No. U.S. Pub. No. 20150045269, published Feb. 12, 2015, to Walker, et al. The 20150045269 publication, for example, describes preparation of a sulfurized alkaline earth metal (e.g., calcium) dodecylphenate by reacting dodecylphenol with calcium hydroxide or calcium oxide and an alkylene glycol. The reaction product is reacted with sulfur.
[0074] The sulfurization reaction is carried out in the presence of a base, which in one embodiment is the cation source, as described below.
[0075] In other embodiments the intermediate compound is contacted with formaldehyde or other aldehyde, which introduces alkylene bridging groups between polyolefin-substituted phenols in the presence of a base.
[0076] In general, sulfur coupling produces a more acidic compound which makes over-basing easier.
[0077] Formation of the salt may be performed by reaction of the sulfurized polyolefin-substituted compound or other sulfurized intermediate compound or alkylene bridged polyolefin-substituted compound with a basic metal compound which serves as a cation source, such as lime (calcium hydroxide/oxide) or magnesium oxide, or with a pnictogen base, in approximately equimolar amounts, with respect to the OH groups in the intermediate compound, optionally in the presence of a solvent.
[0078] Suitable basic metal compounds include hydroxides, oxides and alkoxides of the metal such as (1) an alkali metal salt derived from a metal base selected from an alkali hydroxide, alkali oxide or an alkali alkoxide, or (2) an alkaline earth metal salt derived from a metal base selected from an alkaline earth hydroxide, alkaline earth oxide or alkaline earth alkoxide. Representative examples of metal basic compounds with hydroxide functionality include lithium hydroxide, potassium hydroxide, sodium hydroxide, magnesium hydroxide, calcium hydroxide, barium hydroxide, aluminum hydroxide and the like. Representative examples of metal basic compounds with oxide functionality include lithium oxide, magnesium oxide, calcium oxide, barium oxide and the like. In one embodiment, the alkaline earth metal base is slaked lime (calcium hydroxide).
[0079] The pnictogen cation may be derived/derivable from a compound with a primary amine, a secondary amine, a tertiary amine, or mixtures thereof. Typically the amine salt may be derived from a secondary or a tertiary amine.
[0080] The amine that can be used to prepare a pnictogen may be any amine capable of salting with a protic acid. The amine may be an alkyl amine, typically a di- or tri-alkyl amine. The alkyl amine may have alkyl groups having 1 to 30, or 2 to 20, or 3 to 10 carbon atoms. Examples of a dialkyl amines include diethylamine, dipropylamine, dibutylamine, dipentylamine, dihexylamine, di-(2-ethylhexyl)amine, di-decylamine, di-dodecylamine, di-stearylamine, di-oleylamine, di-eicosylamine, or mixtures thereof. Examples of a trialkyl amine include triethylamine, tripropylamine, tributylamine, tripentylamine, trihexylamine, tri-(2-ethylhexyl)amine, tri-decylamine, tri-dodecylamine, tri-stearylamine, tri-oleylamine, tri-eicosylamine, and mixtures thereof.
[0081] The amine may also be a tertiary-aliphatic primary amine. The aliphatic group in this case may be an alkyl group containing 2 to 30, or 6 to 26, or 8 to 24 carbon atoms. Tertiary alkyl amines include monoamines such as tert-butylamine, tert-hexylamine, 1-methyl-1-amino-cyclohexane, tert-octylamine, tert-decylamine, tert-dodecylamine, tert-tetradecylamine, tert-hexadecylamine, tert-octadecylamine, tert-tetracosanylamine, and tert-octacosanylamine.
[0082] In one embodiment, the pnictogen base includes a phosphorus acid amine salt which includes an amine with Cii to C.sub.22 tertiary alkyl primary groups, or mixtures thereof.
[0083] In one embodiment the amine salt may be in the form of a quaternary ammonium salt. Examples of quaternary ammonium salts containing a hydroxyalkyl group, and methods for their synthesis, are disclosed in U.S. Pat No. 3,962,104. In certain embodiments, the quaternary ammonium compound is derived from a monoamine by means of alkylation, i.e., from a tertiary amine having only a single amino group, that is, having no additional amine nitrogen atoms in any of the three hydrocarbyl groups or substituted hydrocarbyl groups attached to the tertiary amine nitrogen. In certain embodiments there are no additional amine nitrogen atoms in any of the hydrocarbyl groups or substituted hydrocarbyl groups attached to the central nitrogen in the quaternary ammonium ion. The tetraalkylammonium hydroxide may contain alkyl groups having 1 to 30, or 2 to 20, or 3 to 10 carbon atoms. The tetraalkylammonium hydroxide may include tetrapropylammonium hydroxide, tetrabutylammonium hydroxide, tetra-pentylammonium hydroxide, tetrahexylammonium hydroxide, tetra-2-ethylhexyl-ammonium hydroxide, tetradecylammonium hydroxide, or a mixture thereof.
[0084] The amine may be quaternized with a quaternizing agent, or mixture thereof.
[0085] The pnictogen base may include an aminoalkyl substituted heterocyclic compound, such as 1-(3-aminopropyl)imidazole, 4-(3-aminopropyl)morpholine, 1-(2-aminoethyl)piperidine, 3,3-diamino-N-methyldipropylamine, and 3,3-aminobis(N,N-dimethylpropylamine).
[0086] Other examples of quaternary ammonium salts and methods for preparing the same are described in U.S. Pat. Nos. 3,778,371, 4,171,959, 4,253,980, 4,326,973, 4,338,206, and 5,254,138.
[0087] When the amine salt is derived from an aromatic amine, the aromatic amine may form an ion such as a pyridinium ion, or an imidazolium ion. Certain quaternary phosphonium salts may be prepared by the reaction of phosphine with aldehydes and a halide e.g., tetrakis(hydroxymethyl)phosphonium halide (typically chloride).
[0088] A quaternary pnictogen halide compound may be a commercially available material, or it may be prepared by reaction of a tertiary amine with a hydrocarbyl halide, by known techniques. This reaction may be performed in a separate vessel or in the same vessel in which it is subsequently (or simultaneously) reacted with the oil-soluble acidic compound, which may be converted previously (or simultaneously) into its metal neutralized form.
[0089] Neutralization of the sulfurized intermediate compound may be carried out in a continuous or batch process by any method known to a person skilled in the art. In general, neutralization can be carried out by contacting the sulfurized or intermediate compound with a metal or pnictogen base under reactive conditions, e.g., in an inert-compatible liquid hydrocarbon diluent. If desired, the reaction can be conducted under an inert gas, such as nitrogen. The metal or pnictogen base may be added either in a single addition or in a plurality of additions at intermediate points during the reaction.
[0090] Neutralization may be conducted in a suitable solvent or diluent oil, such as toluene, xylene and commonly with a promoter such as an alcohol, e.g., a C.sub.1 to C.sub.16 alcohol, such as methanol, decyl alcohol, or 2-ethylhexanol; a diol, e.g., C.sub.2 to C.sub.4 alkylene glycols, such as ethylene glycol; and/or carboxylic acids. Suitable diluent oils include naphthenic oils and mixed oils, e.g., paraffinic. The quantity of solvent or diluent oil used may be such that the amount of solvent or oil in the final product constitutes from 15% to 65% by weight of the final product, such as from 25% to 50%.
[0091] The neutralization reaction may be conducted at temperatures above room temperature (20.degree. C.). In general, neutralization can be carried out at a temperature of between 150-200.degree. C. The neutralization reaction itself may take place for over 5 to 60 minutes up to 9 hours, for example, 7 hours.
[0092] In another embodiment, the salt of the polyolefin-substituted bridged hydroxyaromatic compound can be prepared in a one-pot method. In this method, the intermediate compound is combined with diluent oil and ethylene glycol and heated while stirring. A metal or pnictogen base, such as hydrated lime, is added to the heated reaction mixture, e.g., in several portions. Sulfur is added to the reaction mixture, and optionally additional metal or pnictogen base is added and the mixture stirred. The reaction mixture may be vacuum stripped to remove excess solvent.
[0093] In one embodiment, the exemplary polyolefin-substituted bridged hydroxyaromatic compound may be overbased. Overbasing can be carried out either during or after one of the sulfurization and/or neutralization steps. Alternatively, sulfurization, neutralization and overbasing can be carried out simultaneously. In general, the overbasing is carried out by reaction of the salt of the sulfur-coupled polyolefin-substituted aromatic polyol with an acidic overbasing compound, such as carbon dioxide or boric acid. In one embodiment, an overbasing process is by way of carbonation, i.e., a reaction with carbon dioxide. Such carbonation can be conveniently effected by addition of solvents such as aromatic solvents, alcohols or a polyols, typically an alkylene diol, e.g., ethylene glycol. Conveniently, the reaction is conducted by bubbling of gaseous carbon dioxide through the reaction mixture, optionally in the presence of sulfonic acid. Excess solvents and any water formed during the overbasing reaction can be conveniently removed by distillation either during or after the reaction.
[0094] In one embodiment, the overbasing reaction is carried out in a reactor by reacting the salt of the polyolefin-substituted bridged hydroxyaromatic compound with a source of an alkaline earth metal such as lime (i.e., an alkaline earth metal hydroxide) in the presence of carbon dioxide, and optionally in the presence of an aromatic solvent (e.g., xylene), and/or a hydrocarbyl alcohol, such as methanol. The reaction may be conducted by bubbling gaseous carbon dioxide through the reaction mixture. The carbon dioxide is introduced over a period of 1 hour to 3 hours, at a temperature ranging from 150-200.degree. C. The degree of overbasing may be controlled by the quantity of the source of an alkaline earth metal, carbon dioxide and the reactants added to the reaction mixture and the reaction conditions used during the carbonation process.
[0095] In another embodiment, the overbasing reaction can be carried out at from 140-180.degree. C. in the presence of a polyol, typically an alkylene diol, e.g., ethylene glycol, and/or alkanols, e.g., C.sub.6 to C.sub.16 alkanol(s), such as decyl alcohols or 2-ethyl hexanol. Excess solvent and any water formed during the overbasing reaction can be conveniently removed by distillation either during or after the reaction.
[0096] Methods for forming overbased detergents useful herein are described, for example, in U.S. Pat. Nos. 5,259,966, 6,015,778, 5,534,168, and 6,268,318, and U.S. Pub. No. 2013/0203639.
[0097] The resulting overbased salt of the polyolefin-substituted bridged hydroxyaromatic compound may contain some amount of unsulfurized hydroxy-substituted intermediate compound and/or its unsulfurized metal salt.
[0098] The composition containing the overbased salt of the polyolefin-substituted bridged hydroxyaromatic compound may be sparged, e.g., by bubbling gas, such as air or nitrogen, at a temperature ranging from 190-250.degree. C. through the composition. The sparging results in removing substantially all of the unsulfurized hydroxy-substituted intermediate compound and salts thereof to provide a composition substantially free of the unsulfurized hydroxy-substituted intermediate compound and unsulfurized salts thereof. The term "substantially free" as used herein means less than 1.5 wt. %, or less than 1 wt. %, or less than 0.3 wt. % of these unsulfurized compounds, such as 0.1-0.3 wt. %, or less.
[0099] In one embodiment, the salt of the polyolefin-substituted bridged hydroxyaromatic compound does not contain any sulfonate functional groups.
[0100] In one embodiment, the salt of the polyolefin-substituted bridged hydroxyaromatic compound does not contain any phosphate functional groups.
[0101] In one embodiment, the salt of the polyolefin-substituted bridged hydroxyaromatic compound does not contain any borate functional groups.
[0102] In another embodiment, the salt of the polyolefin-substituted bridged hydroxyaromatic compound does contain a borate functional group.
[0103] The salts described above can be boronated by processes known to those skilled in the art. Boration can be accomplished either prior to, or after, the overbasing step. The boration can be accomplished by a number of boronating agents, such as boric acid, metaboric acid, orthoboric acid, alkyl borates, boron halides, polymers of boron, esters of boron and similar materials. When present, the boron content of the salt may be 0.1 wt. % to 5 wt. %, or 1 wt. % to 5 wt. %, or 2 wt. % to 4 wt. %.
[0104] The salt of the polyolefin-substituted bridged hydroxyaromatic compound, in one embodiment, may be formed from an anion composed of carbon, hydrogen, oxygen, boron and nitrogen; and a metallic cation.
[0105] In one embodiment, the salt of the polyolefin-substituted bridged hydroxyaromatic compound may comprise or consist of an anion composed of carbon, hydrogen, oxygen and optionally nitrogen; and a metallic cation, such as a calcium, magnesium or sodium cation.
[0106] Examples of suitable ethylenically unsaturated esters of boron include(meth)acrylates, fumarates and maleates which are derived from saturated alcohols, such as 2-ethylhexyl (meth)acrylate, heptyl (meth)acrylate, 2-tert-butylheptyl (meth)acrylate, octyl (meth)acrylate, 3-isopropylheptyl (meth)acrylate, nonyl (meth)acrylate, decyl (meth)acrylate, undecyl (meth)acrylate; and the corresponding fumarates and maleates. The expression "(meth)acrylates" encompasses methacrylates and acrylates and also mixtures of the two.
Lubricating Compositions
[0107] The exemplary polyolefin-substituted bridged hydroxyaromatic compound or salt thereof may be present in a lubricating composition at a concentration of at least 0.1 wt. % and may be up to 20 wt. %. For example, the concentration of the compound may be at least 0.25 wt. %, or at least 0.5 wt. %, or at least 1 wt. %, or at least 5 wt. %, or at least 10 wt. %, or at least 15 wt. % of the lubricating composition. The compound may also be present in a concentrate, alone or with other additives and with a lesser amount of oil. In a concentrate, the amount of the compound may be at least 2, or at least 3 times the concentration in the lubricating composition.
[0108] In addition to the polyolefin-substituted bridged hydroxyaromatic compound or metal salt thereof, the exemplary lubricating composition includes an oil of lubricating viscosity and optionally one or more additional performance additives suited to providing the performance properties of a fully formulated lubricating composition, e.g., a marine diesel cylinder lubricant. Examples of these additional performance additives include (overbased) detergents, viscosity modifiers, friction modifiers, antioxidants, dispersants, antiwear/antiscuffing agents, metal deactivators, extreme pressure agents, foam inhibitors, demulsifiers, pour point depressants, corrosion inhibitors, seal swelling agents, and the like, which may be used singly or in combination.
Oil of Lubricating Viscosity
[0109] The lubricating composition may include the oil of lubricating viscosity as a minor or major component thereof, such as at least 5 wt. %, or at least 10 wt. %, or at least 20 wt. %, or at least 30 wt. %, or at least 40 wt. %, or at least 60 wt. %, or at least 80 wt. % of the lubricating composition.
[0110] Suitable oils include natural and synthetic oils, oil derived from hydrocracking, hydrogenation, and hydrofinishing, unrefined, refined, re-refined oils or mixtures thereof. Unrefined, refined and re-refined oils, and natural and synthetic oils are described, for example, in WO2008/147704 and US Pub. No. 2010/197536. Synthetic oils may also be produced by Fischer-Tropsch reactions and typically may be hydroisomerized Fischer-Tropsch hydrocarbons or waxes. Oils may be prepared by a Fischer-Tropsch gas-to-liquid synthetic procedure as well as other gas-to-liquid procedures.
[0111] Oils of lubricating viscosity may also be defined as specified in April 2008 version of "Appendix E--API Base Oil Interchangeability Guidelines for Passenger Car Motor Oils and Diesel Engine Oils", section 1.3 Sub-heading 1.3. "Base Stock Categories". The API Guidelines are also summarized in U.S. Pat. No. 7,285,516. The five base oil groups are as follows: Group I (sulfur content >0.03 wt. %, and/or <90 wt. % saturates, viscosity index 80-120); Group II (sulfur content <0.03 wt. %, and >90 wt. % saturates, viscosity index 80-120); Group III (sulfur content <0.03 wt. %, and >90 wt. % saturates, viscosity index .gtoreq.120); Group IV (all polyalphaolefins (PAOs)); and Group V (all others not included in Groups I, II, III, or IV). The exemplary oil of lubricating viscosity includes an API Group I, Group II, Group III, Group IV, Group V oil, or mixtures thereof. In some embodiments, the oil of lubricating viscosity is an API Group I, Group II, Group III, or Group IV oil, or mixtures thereof. In some embodiments, the oil of lubricating viscosity is an API Group I, Group II, or Group III oil, or mixture thereof. In one embodiment the oil of lubricating viscosity may be an API Group II, Group III mineral oil, a Group IV synthetic oil, or mixture thereof. In some embodiments, at least 5 wt. %, or at least 10 wt. %, or at least 20 wt. %, or at least 40 wt. % of the lubricating composition is a polyalphaolefin (Group IV).
Engine Lubricating Compositions
[0112] For lubricating compositions useful in engines, the oil of lubricating viscosity may have a kinematic viscosity of up to 30 mm.sup.2/s or up to 25 mm.sup.2/s (cSt) at 100.degree. C. and can be at least 12 mm.sup.2/s at 100.degree. C., and in other embodiments at least 15 mm.sup.2/s. As used herein, kinematic viscosity is determined at 100.degree. C. by ASTM D445-14, "Standard Test Method for Kinematic Viscosity of Transparent and Opaque Liquids (and Calculation of Dynamic Viscosity)," ASTM International, West Conshohocken, Pa., 2003, DOI: 10.1520/D0445-14 and may be referred to as KV_100.
[0113] The viscosity grade of cylinder oils suited to use in 2-stroke marine diesel engines may be from SAE-40 to SAE-60, which corresponds to a KV_100 of 12.5 to 26 mm.sup.2/s. SAE-50 grade oils, for example, have a KV_100 of 16.3-21.9 mm.sup.2/s. Cylinder oils for 2-stroke marine diesel engines may be formulated to achieve a KV_100 of 19 to 21.5 mm.sup.2/s. This viscosity can be obtained by a mixture of additives and base oils, for example containing mineral bases of Group I such as Neutral Solvent (for example 500 NS or 600 NS) and Bright Stock bases. Any other combination of mineral or synthetic bases or bases of vegetable origin having, in mixture with the additives, a viscosity compatible with the grade SAE 50 can be used.
[0114] As an example, an oil formulation suited to use as a cylinder lubricant for low-speed 2-stroke marine diesel engines contains 18 to 25 wt. % of a Group I base oil of a BSS type (distillation residue, with a KV_100 of 28-32 mm.sup.2/s, with a density at 15.degree. C. of 895-915 kg/m.sup.3), and 50 to 60 wt. % of a Group I base oil of a SN 600 type (distillate, with a density at 15.degree. C. of 880-900 kg/m.sup.3, with a KV_100 of about 12 mm.sup.2/s).
[0115] In certain embodiments, the lubricating composition may contain synthetic ester base fluids. Synthetic esters may have a kinematic viscosity measured at 100.degree. C. of 2.5 mm.sup.2/s to 30 mm.sup.2/s. In one embodiment, the lubricating composition comprises less than 50 wt. % of a synthetic ester base fluid with a KV_100 of at least 5.5 mm.sup.2/s, or at least 6 mm.sup.2/s, or at least 8 mm.sup.2/s.
[0116] Exemplary synthetic oils include poly-alpha olefins, polyesters, poly-acrylates, and poly-methacrylates, and co-polymers thereof. Example synthetic esters include esters of a dicarboxylic acid (e.g., selected from phthalic acid, succinic acid, alkyl succinic acids, alkenyl succinic acids, maleic acid, azelaic acid, suberic acid, sebacic acid, fumaric acid, adipic acid, linoleic acid dimer, malonic acid, alkyl malonic acids, and alkenyl malonic acids) with an alcohol (e.g., selected from butyl alcohol, hexyl alcohol, dodecyl alcohol, 2-ethylhexyl alcohol, ethylene glycol, diethylene glycol monoether, and propylene glycol). Specific examples of these esters include dibutyl adipate, di(2-ethylhexyl) sebacate, di-n-hexyl fumarate, dioctyl sebacate, diisooctyl azelate, diisodecyl azelate, dioctyl phthalate, didecyl phthalate, dieicosyl sebacate, the 2-ethylhexyl diester of linoleic acid dimer, and the complex ester formed by reacting one mole of sebacic acid with two moles of tetraethylene glycol and two moles of 2-ethylhexanoic acid.
[0117] Esters useful as synthetic oils also include those made from C.sub.5 to C.sub.12 monocarboxylic acids and polyols and from polyol ethers such as neopentyl glycol, trimethylolpropane, pentaerythritol, dipentaerythritol, and tripentaerythritol. Esters can also be monoesters, such as are available under the trade name Priolube 1976.TM. (C.sub.18-alkyl-COO--C.sub.20 alkyl).
[0118] Synthetic ester base oils may be present in the lubricating composition of the invention in an amount less than 50 wt. % of the composition, or less than 40 weight %, or less than 35 weight %, or less than 28 weight %, or less than 21 weight %, or less than 17 weight %, or less than 10 weight %, or less than 5 weight % of the composition. In one embodiment, the lubricating composition of the invention is free of, or substantially free of, a synthetic ester base fluid having a KV_100 of at least 5.5 mm.sup.2/s.
[0119] Example natural oils include animal and vegetable oils, such as long chain fatty acid esters. Examples include linseed oil, sunflower oil, sesame seed oil, beef tallow oil, lard oil, palm oil, castor oil, cottonseed oil, corn oil, peanut oil, soybean oil, olive oil, whale oil, menhaden oil, sardine oil, coconut oil, palm kernel oil, babassu oil, rape oil, and soya oil.
[0120] The amount of the oil of lubricating viscosity present is typically the balance remaining after subtracting from 100 weight % the sum of the amount of the exemplary amino-carboxylate compound and the other performance additives.
Engine Lubricating Compositions--Other Additives
[0121] Engine lubricating compositions in accordance with the present invention may also contain other additives that provide particular performance benefits to the engine lubricant. These additional additives may include detergents, antioxidants, dispersants, anti-wear agents, oil soluble titanium compounds, extreme pressure agents, foam inhibitors, viscosity modifiers, corrosion inhibitors, metal deactivators, pour point depressants, friction modifiers, demulsifiers, and seal swell agents. These additional components are described in more detail below.
[0122] The lubricating composition optionally further includes at least one detergent, other than the exemplary compound. Exemplary detergents useful herein include overbased metal-containing detergents. The metal of the metal-containing detergent may be zinc, sodium, calcium, barium, or magnesium. The overbased metal-containing detergent may be chosen from sulfonates, non-sulfur containing phenates, sulfur containing phenates, salixarates, salicylates, and mixtures thereof, or borated equivalents thereof. The overbased detergent may be borated with a borating agent such as boric acid.
[0123] The overbased metal-containing detergent may also include "hybrid" detergents formed with mixed surfactant systems including phenate and/or sulfonate components, e.g., phenate/salicylates, sulfonate/phenates, sulfonate/salicylates, sulfonates/phenates/salicylates, as described, for example, in U.S. Pat. Nos. 6,429,178; 6,429,179; 6,153,565; and 6,281,179. Where a hybrid sulfonate/phenate detergent is employed, the hybrid detergent can be considered equivalent to amounts of distinct phenate and sulfonate detergents introducing like amounts of phenate and sulfonate soaps, respectively.
[0124] Example overbased metal-containing detergents include zinc, sodium, calcium and magnesium salts of sulfonates, phenates (including sulfur-containing and non-sulfur containing phenates), salixarates and salicylates. Such overbased sulfonates, salixarates, phenates and salicylates may have a total base number of 120 to 700, or 250 to 600, or 300 to 500 (on an oil free basis).
[0125] The overbased sulfonate detergent may have a metal ratio of 12 to less than 20, or 12 to 18, or 20 to 30, or 22 to 25.
[0126] Typically, an overbased metal-containing detergent may be a zinc, sodium, calcium or magnesium salt of a sulfonate, a phenate, sulfur containing phenate, salixarate or salicylate. Overbased sulfonates, salixarates, phenates and salicylates typically have a total base number of 120 to 700 TBN. Overbased sulfonates typically have a total base number of 120 to 700, or 250 to 600, or 300 to 500 (on an oil free basis).
[0127] The overbased sulfonate detergent may have a metal ratio of 12 to less than 20, or 12 to 18, or 20 to 30, or 22 to 25.
[0128] Example sulfonate detergents include linear and branched alkylbenzene sulfonate detergents, and mixtures thereof, which may have a metal ratio of at least 8, as described, for example, in U.S. Pub. No. 2005065045. Linear alkyl benzenes may have the benzene ring attached anywhere on the linear chain, usually at the 2, 3, or 4 position, or be mixtures thereof. Linear alkylbenzene sulfonate detergents may be particularly useful for assisting in improving fuel economy.
[0129] In one embodiment, the alkylbenzene sulfonate detergent may be a branched alkylbenzene sulfonate, a linear alkylbenzene sulfonate, or mixtures thereof.
[0130] In one embodiment, the lubricating composition may be free of linear alkylbenzene sulfonate detergent. The sulfonate detergent may be a metal salt of one or more oil-soluble alkyl toluene sulfonate compounds as disclosed in U.S. Pub. No. 20080119378.
[0131] The lubricating composition may include at least 0.01 wt. % or at least 0.1 wt. % of the detergent other than the exemplary compound, and in some embodiments, up to 2 wt. %, or up to 1 wt. % detergent.
[0132] The lubricating composition optionally further includes at least one antioxidant. Exemplary antioxidants useful herein include phenolic and aminic antioxidants, such as diarylamines, alkylated diarylamines, hindered phenols, and mixtures thereof. The diarylamine or alkylated diarylamine may be a phenyl-.alpha.-naphthylamine (PANA), an alkylated diphenylamine, an alkylated phenylnapthylamine, or mixture thereof. Example alkylated diphenylamines include dinonyl diphenylamine, nonyl diphenylamine, octyl diphenylamine, dioctyl diphenylamine, didecyl diphenylamine, decyl diphenylamine, and mixtures thereof. Example alkylated diarylamines include octyl, dioctyl, nonyl, dinonyl, decyl and didecyl phenylnapthylamines. Hindered phenol antioxidants often contain a secondary butyl and/or a tertiary butyl group as a steric hindering group. The phenol group may be further substituted with a hydrocarbyl group (e.g., a linear or branched alkyl) and/or a bridging group linking to a second aromatic group. Examples of suitable hindered phenol antioxidants include 2,6-di-tert-butylphenol, 4-methyl-2,6-di-tert-butylphenol, 4-ethyl-2,6-di-tert-butylphenol, 4-propyl-2,6-di-tert-butylphenol, 4-butyl-2,6-di-tert-butylphenol, and 4-dodecyl-2,6-di-tert-butylphenol. In one embodiment, the hindered phenol antioxidant may be an ester, such as those described in U.S. Pat. No. 6,559,105. One such hindered phenol ester is sold as Irganox.TM. L-135, obtainable from Ciba.
[0133] When present, the lubricating composition may include at least 0.1 wt. % or at least 0.5 wt. %, or at least 1 wt. % antioxidant, and in some embodiments, up to 3 wt. %, or up to 2.75 wt. %, or up to 2.5 wt. % antioxidant.
[0134] The lubricating composition optionally further includes at least one dispersant, other than the exemplary compound. Exemplary dispersants include succinimide dispersants, Mannich dispersants, succinimide dispersants, and polyolefin succinic acid esters, amides, and ester-amides, and mixtures thereof. The succinimide dispersant, where present, may be as described above for the succinim ides described as useful for cation M.
[0135] The succinimide dispersant may be derived from an aliphatic polyamine, or mixtures thereof. The aliphatic polyamine may be an ethylenepolyamine, a propylenepolyamine, a butylenepolyamine, or a mixture thereof. In one embodiment the aliphatic polyamine may be an ethylenepolyamine. In one embodiment the aliphatic polyamine may be chosen from ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, pentaethylenehexamine, polyamine still bottoms, and mixtures thereof.
[0136] In one embodiment, the dispersant may be a polyolefin succinic acid ester, amide, or ester-amide. A polyolefin succinic acid ester-amide may be a polyisobutylene succinic acid reacted with an alcohol (such as pentaerythritol) and a polyamine as described above. Example polyolefin succinic acid esters include polyisobutylene succinic acid esters of pentaerythritol and mixture thereof.
[0137] The dispersant may be an N-substituted long chain alkenyl succinimide. An example of an N-substituted long chain alkenyl succinimide is polyisobutylene succinimide. Typically the polyisobutylene from which polyisobutylene succinic anhydride is derived has a number average molecular weight of 350 to 5000, or 550 to 3000 or 750 to 2500. Succinimide dispersants and their preparation are disclosed, for example, in U.S. Pat. Nos. 3,172,892, 3,219,666, 3,316,177, 3,340,281, 3,351,552, 3,381,022, 3,433,744, 3,444,170, 3,467,668, 3,501,405, 3,542,680, 3,576,743, 3,632,511, 4,234,435, Re 26,433, and 6,165,235, and 7,238,650 and EP Patent Application 0 355 895 A.
[0138] The succinimide dispersant may comprise a polyisobutylene succinimide, wherein the polyisobutylene from which polyisobutylene succinimide is derived has a number average molecular weight of 350 to 5000, or 750 to 2500.
[0139] The exemplary dispersants may also be post-treated by conventional methods by a reaction with any of a variety of agents. Among these are boron compounds (such as boric acid), urea, thiourea, dimercaptothiadiazoles, carbon disulfide, aldehydes, ketones, carboxylic acids, such as terephthalic acid, hydrocarbon-substituted succinic anhydrides, maleic anhydride, nitriles, epoxides, and phosphorus compounds. In one embodiment the post-treated dispersant is borated. In one embodiment the post-treated dispersant is reacted with dimercaptothiadiazoles. In one embodiment the post-treated dispersant is reacted with phosphoric or phosphorous acid. In one embodiment the post-treated dispersant is reacted with terephthalic acid and boric acid (as described in U.S. Pub. No. 2009/0054278.
[0140] When present, the lubricating composition may include at least 0.01 wt. %, or at least 0.1 wt. %, or at least 0.5 wt. %, or at least 1 wt. % of other dispersant(s), and in some embodiments, up to 20 wt. %, or up to 15 wt. %, or up to 10 wt. %, or up to 6 wt. % or up to 3 wt. % dispersant.
[0141] The lubricating composition optionally further includes at least one antiwear agent. Examples of suitable antiwear agents suitable for use herein include titanium compounds, tartrates, tartrimides, oil soluble amine salts of phosphorus compounds, sulfurized olefins, metal dihydrocarbyldithiophosphates (such as zinc dialkyldithiophosphates), phosphites (such as dibutyl phosphite), phosphonates, thiocarbamate-containing compounds, such as thiocarbamate esters, thiocarbamate amides, thiocarbamic ethers, alkylene-coupled thiocarbamates, and bis(S-alkyldithiocarbamyl) disulfides. The antiwear agent may in one embodiment include a tartrate, or tartrimide as described in U.S. Pub. Nos. 2006/0079413; 2006/0183647; and 2010/0081592. The tartrate or tartrimide may contain alkyl-ester groups, where the sum of carbon atoms on the alkyl groups is at least 8. The antiwear agent may, in one embodiment, include a citrate as is disclosed in US Pub. No. 20050198894.
[0142] The lubricating composition may in one embodiment further include a phosphorus-containing antiwear agent. Example phosphorus-containing antiwear agents include zinc dialkyldithiophosphates, phosphites, phosphates, phosphonates, and ammonium phosphate salts, and mixtures thereof.
[0143] When present, the lubricating composition may include at least 0.01 wt. %, or at least 0.1 wt. %, or at least 0.5 wt. % antiwear agent, and in some embodiments, up to 3 wt. %, or up to 1.5 wt. %, or up to 0.9 wt. antiwear agent.
[0144] The lubricating composition may include one or more oil-soluble titanium compounds, which may function as antiwear agents, friction modifiers, antioxidants, deposit control additives, or more than one of these functions. Example oil-soluble titanium compounds are disclosed in U.S. Pat. No. 7,727,943 and U.S. Pub. No. 2006/0014651. Example oil soluble titanium compounds include titanium (IV) alkoxides, such as titanium (IV) isopropoxide and titanium (IV) 2 ethylhexoxide. Such alkoxides may be formed from a monohydric alcohol, a vicinal 1,2-diol, a polyol, or mixture thereof. The monohydric alkoxides may have 2 to 16, or 3 to 10 carbon atoms. In one embodiment, the titanium compound comprises the alkoxide of a vicinal 1,2-diol or polyol. 1,2-vicinal diols include fatty acid mono-esters of glycerol, where the fatty acid may be, for example, oleic acid. Other example oil soluble titanium compounds include titanium carboxylates, such as titanium neodecanoate.
[0145] When present in the lubricating composition, the amount of oil-soluble titanium compounds is included as part of the antiwear agent.
[0146] The lubricating composition may include an extreme pressure agent. Example extreme pressure agents that are soluble in the oil include sulfur- and chlorosulfur-containing EP agents, dimercaptothiadiazole or CS.sub.2 derivatives of dispersants (typically succinimide dispersants), derivative of chlorinated hydrocarbon EP agents and phosphorus EP agents. Examples of such EP agents include chlorinated wax; sulfurized olefins (such as sulfurized isobutylene), hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazoles and oligomers thereof, organic sulfides and polysulfides, such as dibenzyldisulfide, bis-(chlorobenzyl) disulfide, dibutyl tetrasulfide, sulfurized methyl ester of oleic acid, sulfurized alkylphenol, sulfurized dipentene, sulfurized terpene, and sulfurized Diels-Alder adducts; phosphosulfurized hydrocarbons such as the reaction product of phosphorus sulfide with turpentine or methyl oleate; phosphorus esters, such as dihydrocarbon and trihydrocarbon phosphites, e.g., dibutyl phosphite, diheptyl phosphite, dicyclohexyl phosphite, pentylphenyl phosphite; dipentylphenyl phosphite, tridecyl phosphite, distearyl phosphite and polypropylene substituted phenol phosphite; metal thiocarbamates, such as zinc dioctyldithiocarbamate and barium heptylphenol diacid; amine salts of alkyl and dialkylphosphoric acids or derivatives including, for example, the amine salt of a reaction product of a dialkyldithiophosphoric acid with propylene oxide and subsequently followed by a further reaction with P205; and mixtures thereof. Some useful extreme pressure agents are described in U.S. Pat. No. 3,197,405.
[0147] When present, the lubricating composition may include at least 0.01 wt. %, or at least 0.1 wt. %, or at least 0.5 wt. % extreme pressure agent, and in some embodiments, up to 3 wt. %, or up to 1.5 wt. %, or up to 0.9 wt. % of the extreme pressure agent.
[0148] The lubricating composition may include a foam inhibitor. Foam inhibitors that may be useful in the lubricant composition include polysiloxanes; copolymers of ethyl acrylate and 2-ethylhexylacrylate and optionally vinyl acetate; demulsifiers including fluorinated polysiloxanes, trialkyl phosphates, polyethylene glycols, polyethylene oxides, polypropylene oxides and (ethylene oxide-propylene oxide) polymers.
[0149] The lubricating composition may include a viscosity modifier. Viscosity modifiers (also sometimes referred to as viscosity index improvers or viscosity improvers) useful in the lubricant composition are usually polymers, including polyisobutenes, polymethacrylates (PMA) and polymethacrylic acid esters, diene polymers, polyalkylstyrenes, esterified styrene-maleic anhydride copolymers, hydrogenated alkenylarene-conjugated diene copolymers and polyolefins also referred to as olefin copolymer or OCP. PMA's are prepared from mixtures of methacrylate monomers having different alkyl groups. The alkyl groups may be either straight chain or branched chain groups containing from 1 to 18 carbon atoms. Most PMA's are viscosity modifiers as well as pour point depressants. In one embodiment, the viscosity modifier is a polyolefin comprising ethylene and one or more higher olefin, such as propylene.
[0150] When present, the lubricating composition may include at least 0.01 wt. %, or at least 0.1 wt. %, or at least 0.3 wt. %, or at least 0.5 wt. % polymeric viscosity modifiers, and in some embodiments, up to 10 wt. %, or up to 5 wt. %, or up to 2.5 wt. % polymeric viscosity modifiers.
[0151] The lubricating composition may include a corrosion inhibitor. Corrosion inhibitors/metal deactivators that may be useful in the exemplary lubricating composition include fatty amines, octylamine octanoate, condensation products of dodecenyl succinic acid or anhydride, and a fatty acid such as oleic acid with a polyamine, derivatives of benzotriazoles (e.g., tolyltriazole), 1,2,4-triazoles, benzimidazoles, 2-alkyldithiobenzimidazoles and 2-alkyldithiobenzothiazoles.
[0152] The lubricating composition may include a pour point depressant. Pour point depressants that may be useful in the exemplary lubricating composition include polyalphaolefins, esters of maleic anhydride-styrene copolymers, polymethacrylates, polyacrylates, and polyacrylam ides.
[0153] The lubricating composition may include a friction modifier. Friction modifiers that may be useful in the exemplary lubricating composition include fatty acid derivatives such as amines, esters, epoxides, fatty imidazolines, condensation products of carboxylic acids and polyalkylene-polyamines and amine salts of alkylphosphoric acids. The friction modifier may be an ash-free friction modifier. Such friction modifiers are those which typically not produce any sulfated ash when subjected to the conditions of ASTM D 874. An additive is referred to as "non-metal containing" if it does not contribute metal content to the lubricant composition. As used herein the term "fatty alkyl" or "fatty" in relation to friction modifiers means a carbon chain having 8 to 30 carbon atoms, typically a straight carbon chain.
[0154] In one embodiment, the ash-free friction modifier may be represented by the formula:
##STR00006##
[0155] where, D and D' are independently selected from --O--, >NH, >NR.sup.23, an imide group formed by taking together both D and D' groups and forming a R.sup.21--N< group between two >C.dbd.O groups; E is selected from --R.sup.24--O--R.sup.25--, >CH.sub.2, >CHR.sup.26, >CR.sup.26R.sup.27, >C(OH)(CO.sub.2R.sup.22), >C(CO.sub.2R.sup.22).sup.2, and >CHOR.sup.28; where R.sup.24 and R.sup.25 are independently selected from >CH.sub.2, >CHR.sup.26, >CR.sup.26R.sup.27, >C(OH)(CO.sub.2R.sup.22), and >CHOR.sup.28; q is 0 to 10, with the proviso that when q=1, E is not >CH.sub.2, and when n=2, both Es are not >CH.sub.2; p is 0 or 1; R.sup.21 is independently hydrogen or a hydrocarbyl group, typically containing 1 to 150 carbon atoms, with the proviso that when R.sup.21 is hydrogen, p is 0, and q is more than or equal to 1; R.sup.22 is a hydrocarbyl group, typically containing 1 to 150 carbon atoms; R.sup.23, R.sup.24, R.sup.25, R.sup.26 and R.sup.27 are independently hydrocarbyl groups; and R.sup.28 is hydrogen or a hydrocarbyl group, typically containing 1 to 150 carbon atoms, or 4 to 32 carbon atoms, or 8 to 24 carbon atoms. In certain embodiments, the hydrocarbyl groups R.sup.23, R.sup.24, and R.sup.25, may be linear or predominantly linear alkyl groups.
[0156] In certain embodiments, the ash-free friction modifier is a fatty ester, amide, or imide of various hydroxy-carboxylic acids, such as tartaric acid, malic acid lactic acid, glycolic acid, and mandelic acid. Examples of suitable materials include tartaric acid di(2-ethylhexyl) ester (i.e., di(2-ethylhexyl)tartrate), di(C.sub.8-C.sub.10)tartrate, di(C.sub.12-15)tartrate, di-oleyltartrate, oleyltartrimide, and oleyl maleim ide.
[0157] In certain embodiments, the ash-free friction modifier may be chosen from long chain fatty acid derivatives of amines, fatty esters, or fatty epoxides; fatty imidazolines such as condensation products of carboxylic acids and polyalkylene-polyamines; amine salts of alkylphosphoric acids; fatty alkyl tartrates; fatty alkyl tartrimides; fatty alkyl tartramides; fatty phosphonates; fatty phosphites; borated phospholipids, borated fatty epoxides; glycerol esters; borated glycerol esters; fatty amines; alkoxylated fatty amines; borated alkoxylated fatty amines; hydroxyl and polyhydroxy fatty amines including tertiary hydroxy fatty amines; hydroxy alkyl amides; metal salts of fatty acids; metal salts of alkyl salicylates; fatty oxazolines; fatty ethoxylated alcohols; condensation products of carboxylic acids and polyalkylene polyamines; or reaction products from fatty carboxylic acids with guanidine, aminoguanidine, urea, or thiourea and salts thereof.
[0158] Friction modifiers may also encompass materials such as sulfurized fatty compounds and olefins, sunflower oil or soybean oil monoester of a polyol and an aliphatic carboxylic acid.
[0159] In another embodiment the friction modifier may be a long chain fatty acid ester. In another embodiment the long chain fatty acid ester may be a mono-ester and in another embodiment the long chain fatty acid ester may be a triglyceride.
[0160] The amount of the ash-free friction modifier in a lubricant may be 0.1 to 3 percent by weight (or 0.12 to 1.2 or 0.15 to 0.8 percent by weight). The material may also be present in a concentrate, alone or with other additives and with a lesser amount of oil. In a concentrate, the amount of material may be two to ten times the above concentration amounts.
[0161] Molybdenum compounds are also known as friction modifiers. The exemplary molybdenum compound does not contain dithiocarbamate moieties or ligands.
[0162] Nitrogen-containing molybdenum materials include molybdenum-amine compounds, as described in U.S. Pat. No. 6,329,327, and organomolybdenum compounds made from the reaction of a molybdenum source, fatty oil, and a diamine as described in U.S. Pat. No. 6,914,037. Other molybdenum compounds are disclosed in U.S. Pub. No. 20080280795. Molybdenum amine compounds may be obtained by reacting a compound containing a hexavalent molybdenum atom with a primary, secondary or tertiary amine represented by the formula NR.sup.29R.sup.30R.sup.31, where each of R.sup.29, R.sup.30 and R.sup.31 is independently hydrogen or a hydrocarbyl group of 1 to 32 carbon atoms and wherein at least one of R.sup.29, R.sup.30 and R.sup.31 is a hydrocarbyl group of 4 or more carbon atoms or represented by the formula:
##STR00007##
where R.sup.32 represents a chain hydrocarbyl group having 10 or more carbon atoms, s is 0 or 1, R.sup.33 and/or R.sup.34 represents a hydrogen atom, a hydrocarbyl group, an alkanol group or an alkyl amino group having 2 to 4 carbon atoms, and when s=0, both R.sup.33 and R.sup.34 are not hydrogen atoms or hydrocarbon groups.
[0163] Specific examples of suitable amines include monoalkyl (or alkenyl) amines such as tetradecylamine, stearylamine, oleylamine, beef tallow alkylamine, hardened beef tallow alkylamine, and soybean oil alkylamine; dialkyl(or alkenyl)amines such as N-tetradecylmethylamine, N-pentadecylmethylamine, N-hexadecylmethylamine, N-stearylmethylamine, N-oleylmethylamine, N-dococylmethylamine, N-beef tallow alkyl methylamine, N-hardened beef tallow alkyl methylamine, N-soybean oil alkyl methylamine, ditetradecylamine, dipentadecylamine, dihexadecylamine, distearylamine, dioleylamine, didococylamine, bis(2-hexyldecyl)amine, bis(2-octyldodecyl)amine, bis(2-decyltetradecyl)amine, beef tallow dialkylamine, hardened beef tallow dialkylamine, and soybean oil dialkylamine; and trialk(en)ylamines such as tetradecyldimethylamine, hexadecyldimethylamine, octadecyldimethylamine, beef tallow alkyldimethylamine, hardened beef tallow alkyldimethylamine, soybean oil alkyldimethylamine, dioleylmethylamine, tritetradecylamine, tristearylamine, and trioleylamine. Suitable secondary amines have two alkyl (or alkenyl) groups with 14 to 18 carbon atoms.
[0164] Examples of the compound containing the hexavalent molybdenum atom include molybdenum trioxides or hydrates thereof (MoO.sub.3.nH.sub.2O), molybdenum acid (H.sub.2MoO.sub.4), alkali metal molybdates (Q.sub.2MoO.sub.4) wherein Q represents an alkali metal such as sodium and potassium, ammonium molybdates {(NH.sub.4).sub.2MoO.sub.4 or heptamolybdate (NH.sub.4).sub.6[Mo.sub.7O.sub.24].4H.sub.2O}, MoOCl.sub.4, MoO.sub.2Cl.sub.2, MoO.sub.2Br.sub.2, Mo.sub.2O.sub.3Cl.sub.6 and the like. Molybdenum trioxides or hydrates thereof, molybdenum acid, alkali metal molybdates and ammonium molybdates are often suitable because of their availability. In one embodiment, the lubricating composition comprises molybdenum amine compound.
[0165] Other organomolybdenum compounds of the invention may be the reaction products of fatty oils, mono-alkylated alkylene diamines and a molybdenum source. Materials of this sort are generally made in two steps, a first step involving the preparation of an aminoamide/glyceride mixture at high temperature, and a second step involving incorporation of the molybdenum.
[0166] Examples of fatty oils that may be used include cottonseed oil, groundnut oil, coconut oil, linseed oil, palm kernel oil, olive oil, corn oil, palm oil, castor oil, rapeseed oil (low or high erucic acids), soyabean oil, sunflower oil, herring oil, sardine oil, and tallow. These fatty oils are generally known as glyceryl esters of fatty acids, triacylglycerols or triglycerides.
[0167] Examples of some mono-alkylated alkylene diamines that may be used include methylam inopropylamine, methylam inoethylamine, butylam inopropylamine, butylam ino-ethylamine, octylam inopropylamine, octylam inoethylamine, dodecylaam inopropylaamine, dodecylam inoethylamine, hexadecylam inopropylamine, hexadecylam inoethylamine, octadecyl-aminopropylamine, octadecylaminoethylamine, isopropyloxypropyl-1,3-diaminopropane, and octyloxypropyl-1,3-diaminopropane. Mono-alkylated alkylene diamines derived from fatty acids may also be used. Examples include N-coco alkyl-1,3-propanediamine (Duomeen.RTM.C.), N-tall oil alkyl-1,3-propanediamine (Duomeen.RTM.T) and N-oleyl-1,3-propanediamine (Duomeen.RTM.O), all commercially available from Akzo Nobel.
[0168] Sources of molybdenum for incorporation into the fatty oil/diamine complex are generally oxygen-containing molybdenum compounds include, similar to those above, ammonium molybdates, sodium molybdate, molybdenum oxides and mixtures thereof. One suitable molybdenum source comprises molybdenum trioxide (MoO.sub.3).
[0169] Nitrogen-containing molybdenum compounds which are commercially available include, for example, Sakuralube.RTM.710 available from Adeka which is a molybdenum amine compound, and Molyvan.RTM. 855, available from R.T. Vanderbilt.
[0170] The nitrogen-containing molybdenum compound may be present in the lubricant composition at 0.005 to 2 wt. % of the composition, or 0.01 to 1.3 wt. %, or 0.02 to 1.0 wt. % of the composition. The molybdenum compound may provide the lubricant composition with 0 to 1000 ppm, or 5 to 1000 ppm, or 10 to 750 ppm 5 ppm to 300 ppm, or 20 ppm to 250 ppm of molybdenum.
[0171] Demulsifiers useful herein include trialkyl phosphates, and various polymers and copolymers of ethylene glycol, ethylene oxide, propylene oxide, and mixtures thereof.
[0172] Seal swell agents useful herein include sulfolene derivatives such as Exxon Necton-37.TM. (FN 1380) and Exxon Mineral Seal Oil.TM. (FN 3200).
Example Engine Lubricating Compositions
[0173] An engine lubricant in different embodiments may have a composition as illustrated in Table 1. All additives are expressed on an oil-free basis.
TABLE-US-00001 TABLE 1 Example Lubricating Compositions Embodiments (wt. %) Additive A B C polyolefin-substituted bridged 0.1 to 20.sup. 0.5 to 10 1 to 5 hydroxyaromatic compound or salt thereof Overbased Sulfonate Detergent 0 to 9 0.3 to 8 1 to 5 Phenol-based detergent 0 to 10 0.5 to 7 0.75 to 5 (Borated) Dispersant 0 to 12 0.5 to 8 1 to 5 Antioxidant 0 to 13 0.1 to 10 0.5 to 5.sup. Antiwear Agent 0 to 15 0.1 to 10 0.3 to 5.sup. Corrosion Inhibitor 0 to 2 0.1 to 1 0.2 to 0.5 Friction Modifier 0 to 6 0.05 to 4 0.1 to 2.sup. Viscosity Modifier 0 to 10 0.5 to 8 1 to 6 Other Performance Additives 0 to 10 .sup. 0 to 8 0 to 6 Oil of Lubricating Viscosity Balance to 100% .sup.
Use of the Lubricating Composition
[0174] The end use of the engine lubricating composition described herein includes use as a cylinder lubricant for an internal combustion engine, such as a 2-stroke marine diesel engine, but may also find use as an engine oil for a two or 4-stroke engine in a passenger car, heavy, medium and light duty diesel vehicles, small engines such as motorcycle and 2-stroke oil engines, as a driveline lubricant, including gear and automatic transmission oils, and for other industrial oils, such as hydraulic lubricants.
[0175] An exemplary method of lubricating a mechanical device, such as a cylinder of an internal combustion engine, includes supplying the exemplary lubricating composition to the device.
[0176] Generally, the lubricating composition is added to the lubricating system of an internal combustion engine, which then delivers the lubricating composition to the cylinder of the engine, during its operation, where it may be combusted with the fuel.
[0177] The internal combustion engine may be a diesel-fueled engine, such as a 2-stroke marine diesel engine, or a gasoline fueled engine, a natural gas fueled engine, a mixed gasoline/alcohol fueled engine, or a biodiesel fueled engine. The internal combustion engine may be a 2-stroke or 4-stroke engine.
[0178] The lubricating composition may be suitable for use as a cylinder lubricant irrespective of the sulfur, phosphorus or sulfated ash (ASTM D-874) content of the fuel. The sulfur content of the lubricating composition, which is particularly suited to use as an engine oil lubricant, may be 1 wt. % or less, or 0.8 wt. % or less, or 0.5 wt. % or less, or 0.3 wt. % or less. In one embodiment, the sulfur content may be in the range of 0.001 wt. % to 0.5 wt. %, or 0.01 wt. % to 0.3 wt. %. The phosphorus content may be 0.2 wt. % or less, or 0.12 wt. % or less, or 0.1 wt. % or less, or 0.085 wt. % or less, or 0.08 wt. % or less, or even 0.06 wt. % or less, 0.055 wt. % or less, or 0.05 wt. % or less. In one embodiment, the phosphorus content may be 100 ppm to 1000 ppm, or 200 ppm to 600 ppm. The total sulfated ash content may be 2 wt. % or less, or 1.5 wt. % or less, or 1.1 wt. % or less, or 1 wt. % or less, or 0.8 wt. % or less, or 0.5 wt. % or less, or 0.4 wt. % or less. In one embodiment, the sulfated ash content may be 0.05 wt. % to 0.9 wt. %, or 0.1 wt. % to 0.2 wt. % or to 0.45 wt. %.
Driveline Lubricating Compositions
[0179] In one embodiment, the lubricating composition in accordance with the present invention is for use in a driveline device. Lubricating compositions for driveline devices may include the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof as described herein in an oil of lubricating viscosisty, optionally in the presence of other performance additives as described below.
[0180] The lubricant composition may further include other additives. In one embodiment the invention provides a lubricant composition further comprising at least one of a dispersant, an antiwear agent, a dispersant viscosity modifier, a friction modifier, a viscosity modifier, an antioxidant, an overbased detergent (other than the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof as described above), a foam inhibitor, a demulsifier, a pour point depressant or mixtures thereof. In one embodiment the invention provides a lubricant composition further comprising at least one of a polyisobutylene succinimide dispersant, an antiwear agent, a corrosion inhibitor, a dispersant viscosity modifier, a friction modifier, a viscosity modifier (typically a polymethacrylate having linear, comb or star architecture), an antioxidant (including phenolic and aminic antioxidants), an overbased detergent (including overbased sulphonates, phenates, and salicylates other than the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described above), or mixtures thereof.
[0181] The amount of each other performance additive and chemistry of the other performance additive will depend on type of driveline device being lubricated. When present common additives across each driveline lubricant includes viscosity modifiers, dispersants, foam inhibitors, corrosion inhibitors, pour point depressants, demulsifiers, and seal swell agents.
[0182] Viscosity modifiers may be included in the lubricant composition. Viscosity modifiers are usually polymers, including polyisobutenes, polymethacrylic acid esters, diene polymers, polyalkylstyrenes, esterified styrene-maleic anhydride copolymers, alkenylarene-conjugated diene copolymers, and polyolefins. Multifunctional viscosity improvers, which also have dispersant and/or antioxidancy properties are known and may optionally be used. The amount of viscosity modifier may range from 0.1 to 70 wt %, or 1 to 50 wt %, or 2 to 40 wt %. Typically the viscosity modifier may be a polymethacrylate, or mixtures thereof.
[0183] The lubricant may comprise a dispersant, which may be a nitrogen-containing dispersants, for example a hydrocarbyl substituted nitrogen containing additive. Suitable hydrocarbyl substituted nitrogen containing additives include ashless dispersants and polymeric dispersants. Ashless dispersants are so-named because, as supplied, they do not contain metal and thus do not normally contribute to sulfated ash when added to a lubricant. However they may, of course, interact with ambient metals once they are added to a lubricant which includes metal-containing species. Ashless dispersants are characterized by a polar group attached to a relatively high molecular weight hydrocarbon chain. Examples of such materials include succinimide dispersants, Mannich dispersants.
[0184] The dispersant may be post-treated with other reagents, or not post-treated. The dispersant may be post-treated with urea, thiourea, dimercaptothiadiazoles, carbon disulphide, aldehydes, ketones, carboxylic acids, hydrocarbon-substituted succinic anhydrides, maleic anhydride, nitriles, epoxides, and phosphorus compounds. Typically a succinimide dispersant may optionally be post-treated by conventional methods by a reaction with any of a variety of other agents.
[0185] The polyamine may be an alkylenepolyamine. The alkylenepolyamine may include an ethylenepolyamine, a propylenepolyamine, a butylenepolyamine, or mixtures thereof. Examples of propylenepolyamine include propylenediamine, dipropylenetriamine and mixtures thereof.
[0186] In one embodiment the polyamine is chosen from ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, pentaethylenehexamine, polyamine still bottoms and mixtures thereof.
[0187] In another embodiment, the lubricating composition may have an antiwear additive comprising a phosphate amine salt. The C.sub.2-C.sub.18 (or C.sub.2 to C.sub.8 or C.sub.16-C.sub.18) di- or tri-hydrocarbyl phosphite, or mixtures thereof may be represented by the formula:
##STR00008##
wherein at least one of R.sup.6, R.sup.7 and R.sup.8 may be a hydrocarbyl group containing at least 4 carbon atoms and the other may be hydrogen or a hydrocarbyl group. In one embodiment R.sup.6, R.sup.7 and R.sup.8 are all hydrocarbyl groups. The hydrocarbyl groups may be alkyl, cycloalkyl, aryl, acyclic or mixtures thereof. In the formula with all three groups R.sup.6, R.sup.7 and R.sup.8, the compound may be a tri-hydrocarbyl substituted phosphite i.e., R.sup.6, R.sup.7 and R.sup.8 are all hydrocarbyl groups and in some embodiments may be alkyl groups. Typically the di- or tri-hydrocarbyl phosphite comprises dibutyl phosphite or oleyl phosphite.
[0188] Foam inhibitors that may be useful in the compositions include polysiloxanes, copolymers of ethyl acrylate and 2-ethylhexylacrylate and optionally vinyl acetate; demulsifiers including fluorinated polysiloxanes, trialkyl phosphates, polyethylene glycols, polyethylene oxides, polypropylene oxides and (ethylene oxide-propylene oxide) polymers.
[0189] Pour point depressants that may be useful in the compositions include polyalphaolefins, esters of maleic anhydride-styrene copolymers, poly(meth)acrylates, polyacrylates or polyacrylam ides.
[0190] Demulsifiers include trialkyl phosphates, and various polymers and copolymers of ethylene glycol, ethylene oxide, propylene oxide, or mixtures thereof.
[0191] Seal swell agents include sulpholene derivatives Exxon Necton-37.TM. (FN 1380) and Exxon Mineral Seal Oil.TM. (FN 3200).
Manual Transmission Lubricant
[0192] In one embodiment the invention provides a lubricant composition comprising:
[0193] an oil of lubricating viscosity,
[0194] a thiadiazole (typically present at 0.05 to 1 wt %, or 0.07 to 0.7 wt %, or 0.1 to 0.3 wt %, or 0.15 to 0.25 wt %),
[0195] a dispersant (typically present at 0.1 to 5 wt %, or 0.3 to 4 wt %, or 1 to 3 wt %, or 0.1 to 3 wt %),
[0196] a phosphorus-containing antiwear agent chosen from (i) a non-ionic phosphorus compound, which may be a hydrocarbyl phosphite; or (ii) an amine salt of a phosphorus compound,
[0197] from about 0.1 to about 2 wt % of a detergent, typically present in an amount to deliver 110 to 700 ppm, 130 to 600 ppm, 150 to 500 ppm or 160 to 400 ppm calcium; and wherein from about 0.01 to about 2 wt %, or from about 0.1 to about 1.75 wt %, or about 0.2 to about 1.5 wt % of the detergent comprises the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described herein.
[0198] The manual transmission may have synchromesh, or in another embodiment the manual transmission does not have a synchromesh. The synchromesh may be composed of aluminum, steel, bronze, molybdenum, brass (sintered or non-sintered), carbon in the form of fibers, graphitic material (optionally in combination with a cellulosic material), or a cellulosic material, or a phenolic resin.
[0199] In one embodiment the lubricant may comprise 0.03 to 1.0 wt %, or 0.1 to 0.6 wt %, or 0.2 to 0.5 wt % of calcium.
[0200] The detergent may be calcium or magnesium based, and the detergent may have at least 200 TBN, or 250 to 1000, or 450 to 900 or 650 to 800 mg KOH/g on an oil free basis. Typically the detergent is a calcium based detergent.
[0201] The lubricant may have 100 to 2000ppm, 150 to 1500ppm, 200 to 1000, or 250 to 800ppm, or 500 to 875 ppm of phosphorus delivered by an antiwear agent i.e., delivered by zinc dialkyl dithiophosphate or another phosphorus-containing antiwear agent.
[0202] In one embodiment the invention provides a method of lubricating a manual transmission comprising supplying to the manual transmission a lubricant composition comprising:
[0203] an oil of lubricating viscosity,
[0204] a thiadiazole (typically present at 0.05 to 1 wt %, or 0.07 to 0.7 wt %, or 0.1 to 0.3 wt %, or 0.15 to 0.25 wt %),
[0205] a dispersant (typically present at 0.1 to 5 wt %, or 0.3 to 4 wt %, or 1 to 3 wt %, or 0.1 to 3 wt %),
[0206] a phosphorus-containing antiwear agent chosen from (i) a non-ionic phosphorus compound, which may be a hydrocarbyl phosphite; or (ii) an amine salt of a phosphorus compound,
[0207] from about 0.1 to about 5 wt % of a detergent, typically present in an amount to deliver 110 to 700ppm, 130 to 600ppm, 150 to 500ppm or 160 to 400ppm calcium; and wherein from about 0.01 to about 2 wt %, or from about 0.1 to about 1.75 wt %, or about 0.2 to about 1.5 wt % of the detergent comprises the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described herein.
[0208] The thiadiazole compound may include mono- or di-hydrocarbyl substituted 2,5-dimercapto-1,3,4-thiadiazole compounds. Examples of a thiadiazole include 2,5-dimercapto-1,3,4-thiadiazole, or oligomers thereof, a hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole, a hydrocarbylthio-substituted 2,5-dimercapto-1,3,4-thiadiazole, or oligomers thereof. The oligomers of hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole typically form by forming a sulphur-sulphur bond between 2,5-dimercapto-1,3,4-thiadiazole units to form oligomers of two or more of said thiadiazole units. These thiadiazole compounds may also be used in the post treatment of dispersants as mentioned below in the formation of a dimercaptothiadiazole derivative of a polyisobutylene succinimide.
[0209] Examples of a suitable thiadiazole compound include at least one of a dimercaptothiadiazole, 2,5-dimercapto-[1,3,4]-thiadiazole, 3,5-dimercapto-[1,2,4]-thiadiazole, 3,4-dimercapto-[1,2,5]-thiadiazole, or 4-5-dimercapto-[1,2,3]-thiadiazole. Typically readily available materials such as 2,5-dimercapto-1,3,4-thiadiazole or a hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole or a hydrocarbylthio-substituted 2,5-dimercapto-1,3,4-thiadiazole are commonly utilised.
[0210] The lubricant may contain a detergent aside from the aklylphenol detergent described above. The detergent may be neutral or overbased, typically overbased. The detergent may be calcium or magnesium containing, typically calcium containing.
[0211] Another component of the disclosed lubricant can be an overbased, carbonated calcium or magnesium arylsulphonate detergent having a total base number (TBN) of at least 640 as calculated on an oil-free basis, or a mixture of such detergents. Detergents in general are typically overbased materials, otherwise referred to as overbased or superbased salts, which are generally homogeneous Newtonian systems having by a metal content in excess of that which would be present for neutralization according to the stoichiometry of the metal and the detergent anion. The amount of excess metal is commonly expressed in terms of metal ratio, that is, the ratio of the total equivalents of the metal to the equivalents of the acidic organic compound. Overbased materials are prepared by reacting an acidic material (such as carbon dioxide) with an acidic organic compound, an inert reaction medium (e.g., mineral oil), a stoichiometric excess of a metal base, and a promoter such as a phenol or alcohol. The acidic organic material will normally have a sufficient number of carbon atoms, to provide oil-solubility.
[0212] Overbased detergents may be characterized by Total Base Number (TBN), the amount of strong acid needed to neutralize all of the material's basicity, expressed as mg KOH per gram of sample. TBN is a very well-known parameter that is described in ASTM D 4739. Since overbased detergents are commonly provided in a form which contains diluent oil, for the purpose of this document, TBN is to be recalculated to an oil-free basis. Various detergents may have a TBN of 100 to 1000, or 150 to 800, or, 400 to 700. The detergents may have a TBN of at least 640, for instance, 650 to 1000, or even 680 to 800. In each case, the units are mg KOH/g.
[0213] Typically the detergent is an overbased calcium sulphonate detergent, but other metals may also be present, whether in a sulphonate detergent (for example, an overbased magnesium arylsulphonate detergent) or a different detergent substrate (for example, an overbased calcium phenate detergent). The metal compounds generally useful in making the basic metal salts are generally any Group 1 or Group 2 metal compounds (CAS version of the Periodic Table of the Elements). Examples include alkali metals such as sodium, potassium, lithium, copper, magnesium, calcium, barium, zinc, and cadmium. In one embodiment the metals are sodium, magnesium, or calcium. The anionic portion of the salt may be hydroxide, oxide, carbonate, borate, or nitrate. The detergents may be calcium or magnesium detergents, typically prepared using calcium or magnesium oxide or calcium or magnesium hydroxide. Since the detergents of particular interest are carbonated detergents, they will be materials that have been treated with carbon dioxide. Such treatment leads to more efficient incorporation of basic metal into the composition. Formation of high TBN detergents involving reaction with carbon dioxide is disclosed, for instance, in U.S. Pat. No. 7,238,651, Kocsis et al., Jul. 3, 2007, see, for instance, examples 10-13 and the claims. Other detergents, however, may also optionally be present, which need not be carbonated or need not be so highly overbased (i.e., of lower TBN). However, if multiple detergents are present, it is desirable that the overbased calcium or magnesium arylsulphonate detergent is present as the predominant amount by weight of the metal detergents, that is, at least 50 weight percent or at least 60 or 70 or 80 or 90 weight percent of the metal-containing detergents, on an oil free basis.
[0214] The lubricants may contain an overbased sulphonate detergent. Suitable sulphonic acids include sulphonic and thiosulphonic acids, including mono- or polynuclear aromatic or cycloaliphatic compounds. Certain oil-soluble sulphonates may be represented by R.sup.2-T-(SO.sup.-).sub.a or R.sup.3--(SO.sub.3.sup.-).sub.b, where a and b are each at least one; T is a cyclic nucleus such as benzene or toluene; R.sup.2 is an aliphatic group such as alkyl, alkenyl, alkoxy, or alkoxyalkyl; (R.sup.2)-T typically contains a total of at least 15 carbon atoms; and R.sup.3 is an aliphatic hydrocarbyl group typically containing at least 15 carbon atoms. The groups T, R.sup.2, and R.sup.3 may also contain other inorganic or organic substituents; they may also be described as hydrocarbyl groups. In one embodiment the sulphonate detergent may be a predominantly linear alkylbenzenesulphonate detergent as described in paragraphs [0026] to [0037] of US Patent Application 2005-065045. In some embodiments the linear alkyl (or hydrocarbyl) group may be attached to the benzene ring anywhere along the linear chain of the alkyl group, but often in the 2, 3, or 4 position of the linear chain, and in some instances predominantly in the 2 position. In other embodiments, the alkyl (or hydrocarbyl) group may be branched, that is, formed from a branched olefin such as propylene or 1-butene or isobutene. Sulphonate detergents having a mixture of linear and branched alkyl groups may also be used.
[0215] In certain embodiments the carbonated calcium or magnesium arylsulphonate detergent may be based on an alkylated and sulphonated benzene; in another embodiment, it may be based on an alkylated and sulphonated toluene. In either case there may be one or two or three, and in certain embodiments one, alkyl (or hydrocarbyl) group attached to the aromatic ring, in addition to the methyl group if toluene is used as the starting aromatic compound. In one embodiment, the detergent is a monoalkylbenzenemonosulphonate, and in another embodiment it is a monoalkyltoluenemonosulphonate. If there is one alkyl group, it may contain a sufficient number of carbon atoms to impart oil-solubility to the detergent, such as at least 8 carbon atoms, or 10 to 100 carbon atoms, or 10 to 50 carbon atoms, or 12 to 36 carbon atoms, or 14 to 24 or 16 to 20 or alternatively about 18 carbon atoms. If more than one alkyl group (other than methyl) is present, each alkyl group may have the afore-described number of carbon atoms, or all the alkyl groups together may have in total the afore-described number of carbon atoms, (e.g., two C.sub.12 alkyl groups for a total of 24 carbon atoms in the alkyl groups). Another type of overbased material that may additionally be present (that is, in addition to the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described above) in certain embodiments of the present invention is an overbased phenate detergent. Certain commercial grades of calcium or magnesium sulphonate detergents contain minor amounts of calcium or magnesium phenate detergents to aid in their processing or for other reasons and may contain, for instance, 4% phenate substrate content and 96% sulphonate substrate content. The phenols useful in making phenate detergents may be represented by (R.sup.1).sub.a--Ar--(OH).sub.b, where R.sup.1 is an aliphatic hydrocarbyl group of 4 to 400 or 6 to 80 or 6 to 30 or 8 to 25 or 8 to 15 carbon atoms; Ar is an aromatic group such as benzene, toluene or naphthalene; a and b are each at least one, the sum of a and b being up to the number of displaceable hydrogens on the aromatic nucleus of Ar, such as 1 to 4 or 1 to 2. There is typically an average of at least 7 or 8 aliphatic carbon atoms provided by the R.sup.1 groups for each phenol compound, and in some instances about 12 carbon atoms. Phenate detergents are also sometimes provided as sulphur-bridged species or as methylene-bridged species. Sulphur-bridged species may be prepared by reacting a hydrocarbyl phenol with sulphur. Methylene-bridged species may be prepared by reacting a hydrocarbyl phenol with formaldehyde (or a reactive equivalent such as paraformaldehyde). Examples include sulphur-bridged dodecylphenol (overbased Ca salt) and methylene-coupled heptylphenol.
[0216] In another embodiment, an optional, additional overbased material is an overbased saligenin detergent. Overbased saligenin detergents are commonly overbased magnesium salts which are based on saligenin derivatives. A general example of such a saligenin derivative may be represented by the formula:
##STR00009##
where X is --CHO or --CH.sub.2OH, Y is --CH.sub.2-- or --CH.sub.2OCH.sub.2--, and the --CHO groups typically comprise at least 10 mole percent of the X and Y groups; M is hydrogen, ammonium, or a valence of a metal ion (that is, if M is multivalent, one of the valences is satisfied by the illustrated structure and other valences are satisfied by other species such as anions or by another instance of the same structure), Ri is a hydrocarbyl group of 1 to 60 carbon atoms, m is 0 to typically 10, and each p is independently 0, 1, 2, or 3, provided that at least one aromatic ring contains an R.sup.1 substituent and that the total number of carbon atoms in all R.sup.1 groups is at least 7. When m is 1 or greater, one of the X groups may be hydrogen. In one embodiment, M is a valence (or equivalent) of a Mg ion or a mixture of Mg and hydrogen. Saligenin detergents are disclosed in greater detail in U.S. Pat. No. 6,310,009, with special reference to their methods of synthesis (Column 8 and Example 1) and preferred amounts of the various species of X and Y (Column 6).
[0217] Other optional detergents include salixarate detergents. Salixarate detergents are overbased materials that may be represented by a compound comprising at least one unit of formula (I) or formula (II):
##STR00010##
each end of the compound having a terminal group of formula (III) or (IV):
##STR00011##
such groups being linked by divalent bridging groups A, which may be the same or different. In formulas (I)-(IV) R.sup.3 is hydrogen, a hydrocarbyl group, or a valence of a metal ion; R.sup.2 is hydroxyl or a hydrocarbyl group, and j is 0, 1, or 2; R.sup.6 is hydrogen, a hydrocarbyl group, or a hetero-substituted hydrocarbyl group; either R.sup.4 is hydroxyl and R.sup.5 and R.sup.7 are independently either hydrogen, a hydrocarbyl group, or hetero-substituted hydrocarbyl group, or else R.sup.5 and R.sup.7 are both hydroxyl and R.sup.4 is hydrogen, a hydrocarbyl group, or a hetero-substituted hydrocarbyl group; provided that at least one of R.sup.4, R.sup.5, R.sup.6 and R.sup.7 is hydrocarbyl containing at least 8 carbon atoms; and wherein the molecules on average contain at least one of unit (I) or (III) and at least one of unit (II) or (IV) and the ratio of the total number of units (I) and (III) to the total number of units of (II) and (IV) in the composition is 0.1:1 to 2:1. The divalent bridging group "A," which may be the same or different in each occurrence, includes --CH.sub.2-- and --CH.sub.2OCH.sub.2--, either of which may be derived from formaldehyde or a formaldehyde equivalent (e.g., paraform, formalin).
[0218] Salixarate derivatives and methods of their preparation are described in greater detail in U.S. Pat. No. 6,200,936 and PCT Publication WO 01/56968. It is believed that the salixarate derivatives have a predominantly linear, rather than macrocyclic, structure, although both structures are intended to be encompassed by the term "salixarate." In one embodiment, a salixarate detergent may contain a portion of molecules represented (prior to neutralization) by the structure:
##STR00012##
where the R.sup.8 groups are independently hydrocarbyl groups containing at least 8 carbon atoms.
[0219] Glyoxylate detergents are also optional overbased materials. They are based on an anionic group which, in one embodiment, may have the structure:
##STR00013##
wherein each R is independently an alkyl group containing at least 4 or 8 carbon atoms, provided that the total number of carbon atoms in all such R groups is at least 12 or 16 or 24. Alternatively, each R may be an olefin polymer substituent. The acidic material upon from which the overbased glyoxylate detergent is prepared is the condensation product of a hydroxyaromatic material such as a hydrocarbyl-substituted phenol with a carboxylic reactant such as glyoxylic acid or another omega-oxoalkanoic acid. Overbased glyoxylic detergents and their methods of preparation are disclosed in greater detail in U.S. Pat. No. 6,310,011 and references cited therein.
[0220] Another optional overbased detergent is an overbased salicylate, e,g., an alkali metal or alkaline earth metal salt of a substituted salicylic acid. The salicylic acids may be hydrocarbyl-substituted wherein each substituent contains an average of at least 8 carbon atoms per substituent and 1 to 3 substituents per molecule. The substituents may be polyalkene substituents. In one embodiment, the hydrocarbyl substituent group contains 7 to 300 carbon atoms and may be an alkyl group having a molecular weight of 150 to 2000. Overbased salicylate detergents and their methods of preparation are disclosed in U.S. Pat. Nos. 4,719,023 and 3,372,116.
[0221] Other optional overbased detergents may include overbased detergents having a Mannich base structure, as disclosed in U.S. Pat. No. 6,569,818.
[0222] In certain embodiments, the hydrocarbyl substituents on hydroxy-substituted aromatic rings in the above detergents (e.g., phenate, saligenin, salixarate, glyoxylate, or salicylate) are free of or substantially free of C.sub.12 aliphatic hydrocarbyl groups (e.g., less than 1%, 0.1%, or 0.01% by weight of the substituents are C.sub.12 aliphatic hydrocarbyl groups). In some embodiments such hydrocarbyl substituents contain at least 14 or at least 18 carbon atoms.
[0223] Optionally the lubricant may further include an additional phosphorus containing material and may include a metal salt of a phosphorus acid. Metal salts may have the formula:
[(R.sup.8O)(R.sup.9O)P(.dbd.S)--S].sub.n-M
where R.sup.8 and R.sup.9 are independently hydrocarbyl groups containing 3 to 30 carbon atoms, are readily obtainable by heating phosphorus pentasulfide (P.sub.2S.sub.5) and an alcohol or phenol to form an O,O-dihydrocarbyl phosphorodithioic acid. The alcohol which reacts to provide the R.sup.8 and R.sup.9 groups may be a mixture of alcohols, for instance, a mixture of isopropanol and 4-methyl-2-pentanol, and in some embodiments a mixture of a secondary alcohol and a primary alcohol, such as isopropanol and 2-ethylhexanol. The resulting acid may be reacted with a basic metal compound to form the salt. The metal M, having a valence n, generally is aluminum, tin, manganese, cobalt, nickel, zinc, or copper, and in many cases, zinc, to form zinc dialkyldithiophosphates. Such materials are well known and readily available to those skilled in the art of lubricant formulation. Suitable variations to provide low phosphorus volatility are disclosed, for instance, in US published application 2008-0015129, see, e.g., claims.
[0224] Mixtures of amines may also be used in the invention. In one embodiment a useful mixture of amines is "Primene.RTM. 81R" and "Primene.RTM. JMT." Primene.RTM. 81R and Primene.RTM. JMT (both produced and sold by Rohm & Haas) are mixtures of C.sub.11 to C.sub.14 tertiary alkyl primary amines and C.sub.18 to C.sub.22 tertiary alkyl primary amines respectively.
[0225] In one embodiment the hydrocarbyl amine salt of an alkylphosphoric acid ester is the reaction product of a C.sub.14 to C.sub.18 alkylated phosphoric acid with Primene 81R.TM. (produced and sold by Rohm & Haas) which is a mixture of C.sub.11 to C.sub.14 tertiary alkyl primary amines.
[0226] Examples of hydrocarbyl amine salts of dialkyldithiophosphoric acid esters include the reaction product(s) of isopropyl, methyl-amyl (4-methyl-2-pentyl or mixtures thereof), 2-ethylhexyl, heptyl, octyl or nonyl dithiophosphoric acids with ethylene diamine, morpholine, or Primene 81R.TM., and mixtures thereof.
[0227] In one embodiment the dithiophosphoric acid may be reacted with an epoxide or a glycol. This reaction product is further reacted with a phosphorus acid, anhydride, or lower ester. The epoxide includes an aliphatic epoxide or a styrene oxide. Examples of useful epoxides include ethylene oxide, propylene oxide, butene oxide, octene oxide, dodecene oxide, styrene oxide and the like. In one embodiment the epoxide is Propylene oxide. The glycols may be aliphatic glycols having from 1 to about 12, or from about 2 to about 6, or about 2 to about 3 carbon atoms. The dithiophosphoric acids, glycols, epoxides, inorganic phosphorus reagents and methods of reacting the same are described in U.S. Pat. Nos. 3,197,405 and 3,544,465. The resulting acids may then be salted with amines. An example of suitable dithiophosphoric acid is prepared by adding phosphorus pentoxide (about 64 grams) at about 58.degree. C. over a period of about 45 minutes to about 514 grams of hydroxypropyl O,O-di(4-methyl-2-pentyl)phosphorodithioate (prepared by reacting di(4-methyl-2-pentyl)-phosphorodithioic acid with about 1.3 moles of propylene oxide at about 25.degree. C). . The mixture is heated at about 75.degree. C. for about 2.5 hours, mixed with a diatomaceous earth and filtered at about 70.degree. C. The filtrate contains about 11.8% by weight phosphorus, about 15.2% by weight sulfur, and an acid number of 87 (bromophenol blue).
[0228] If the additional phosphorus containing material is present it may provide 1% to 90%, or 10 to 80%, or 20 to 70% of the total amount of phosphorus to the lubricant. In one embodiment the additional phosphorus containing material is present, and in one embodiment the additional phosphorus containing material is present is absent.
[0229] The lubricant may further include an antioxidant, or mixtures thereof. The Antioxidant may include molybdenum compounds such as molybdenum dithiocarbamates, sulphurised olefins, hindered phenols, aminic compounds such as alkylated diphenylamines (typically di-nonyl diphenylamine, octyl diphenylamine, or di-octyl diphenylamine). When present, the antioxidant may be present at 0 to 3 wt %, or 0.1 to 2.5 wt %, or 0.2 to 1.5 wt %.
Automatic Transmission Lubricants
[0230] In one embodiment the invention provides a lubricant composition comprising:
[0231] an oil of lubricating viscosity,
[0232] a dispersant typically present at 0.01 to 5 wt %, or 0.05 to 3 wt %, or 0.1 to 3 wt %, or or 0.1 to 2 wt
[0233] from about 0.1 to about 1, typically present in an amount to deliver 40 to 1000ppm, 50 to 700ppm, 60 to 600ppm or 70 to 250ppm calcium; and wherein from about 0.01 to about 0.8 wt %, or from about 0.1 to about 0.5 wt %, or about 0.2 to about 0.15 wt % of the detergent comprises the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described herein, and
[0234] a friction modifier typically present at 0 to 4 wt %, or 0.1 to 4 wt %, 0.2 to 3 wt %, 0.3 to 3 wt %, 0.25 to 2.5 wt %. In one embodiment the friction modifier is present, and in an alternative embodiment the friction modifier is not present.
[0235] In one embodiment the invention provides a method of lubricating an automatic transmission comprising supplying to the automatic transmission a lubricant composition comprising:
[0236] an oil of lubricating viscosity,
[0237] a dispersant typically present at 0.5 to 3 wt %, or 1 to 2.5 wt %, or 1.5 to 4 wt %, or 1.5 to 3 wt
[0238] a phosphorus-containing antiwear agent chosen from (i) a non-ionic phosphorus compound, which may be a hydrocarbyl phosphite; or (ii) an amine salt of a phosphorus compound,
[0239] a thiadiazole, typically present at 0.1 to 0.5 wt %,or 0.2 to 0.4 wt %, or 0.25 to 0.35 wt %.
[0240] from about 0.1 to about 5 wt % of a detergent, typically present in an amount to deliver 40 to 700ppm, 50 to 600ppm, 60 to 500ppm or 70 to 250ppm calcium; and wherein from about 0.01 to about 2 wt %, or from about 0.1 to about 1.75 wt %, or about 0.2 to about 1.5 wt % of the detergent comprises the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described herein, and
[0241] a friction modifier typically present at 0 to 4 wt %, or 0.1 to 4 wt %, 0.2 to 3 wt %, 0.3 to 3 wt %, 0.25 to 2.5 wt %. In one embodiment the friction modifier is present, and in an alternative embodiment the friction modifier is not present.
[0242] The automatic transmission includes continuously variable transmissions (CVT), infinitely variable transmissions (IVT), Toroidal transmissions, continuously slipping torque converted clutches (CSTCC), stepped automatic transmissions or dual clutch transmissions (DCT).
[0243] The calcium-containing detergent may be an overbased detergent, a non-overbased detergent, or mixtures thereof. Typically the detergent may be overbased.
[0244] The preparation of the calcium-containing detergent is known in the art. Patents describing the preparation of overbased calcium-containing detergents include U.S. Pat. Nos. 2,501,731; 2,616,905; 2,616,911; 2,616,925; 2,777,874; 3,256,186; 3,384,585; 3,365,396; 3,320,162; 3,318,809; 3,488,284; and 3,629,109.
[0245] As used herein the TBN values quoted and associated range of TBN is on "an as is basis," i.e., containing conventional amounts of diluent oil. Conventional amounts of diluent oil typically range from 30 wt % to 60 wt % (often 40 wt % to 55 wt %) of the detergent component.
[0246] A more detailed description of the expressions "metal ratio", TBN and "soap content" are known to a person skilled in the art and explained in standard textbook entitled "Chemistry and Technology of Lubricants", Third Edition, Edited by R. M. Mortier and S. T. Orszulik, Copyright 2010, pages 219 to 220 under the sub-heading 7.2.5. Detergent Classification.
[0247] The calcium-containing detergent may be a non-overbased detergent (may also be referred to as a neutral detergent). The TBN of a non-overbased may be 20 to less than 200, or 30 to 100, or 35 to 50 mg KOH/g. The TBN of a non-overbased calcium-containing detergent may also be 20 to 175, or 30 to 100 mg KOH/g. When a non-overbased calcium-containing detergent is prepared from a strong acid such as a hydrocarbyl-substituted sulphonic acid, the TBN may be lower (for example 0 to 50 mg KOH/g, or 10 to 20 mg KOH/g).
[0248] The calcium-containing detergent may be an overbased detergent, which may have a TBN of greater than 200 mg KOH/g (typically 250 to 600, or 300 to 500 mg KOH/g).
[0249] The calcium-containing detergent may be formed by the reaction of a basic calcium compound and an acidic detergent substrate. The acidic detergent substrate may include an alkyl phenol, an aldehyde-coupled alkyl phenol, a sulphurised alkyl phenol, an alkyl aromatic sulphonic acid (such as, alkyl naphthalene sulphonic acid, alkyl toluene sulphonic acid or alkyl benzene sulphonic acid), an aliphatic carboxylic acid, a calixarene, a salixarene, an alkyl salicylic acid, or mixtures thereof.
[0250] The metal basic compound is used to supply basicity to the detergent. The basic calcium compound is a compound of a hydroxide or oxide of the metal.
[0251] The oxides and/or hydroxides may be used alone or in combination. The oxides or hydroxides may be hydrated or dehydrated, although hydrated is typical. In one embodiment the basic calcium compound may be calcium hydroxide, which may be used alone or mixtures thereof with other metal basic compounds. Calcium hydroxide is often referred to as lime. In one embodiment the metal basic compound may be calcium oxide which may be used alone or mixtures thereof with other metal basic compounds.
[0252] Collectively, when the alkyl phenol, the aldehyde-coupled alkyl phenol, and the sulphurised alkyl phenol are used to prepare a calcium-containing detergent, the detergent may be referred to as a calcium phenate. The calcium phenate may be an alkyl phenate, an aldehyde-coupled alkyl phenate, a sulphurised alkyl phenate, or mixtures thereof.
[0253] The TBN of a calcium phenate may vary from less 200, or 30 to 175 typically 150 to 175) mg KOH/g for a neutral phenate to 200 or more to 500, or 210 to 400 (typically 230 to 270) mg KOH/g for an overbased phenate.
[0254] The alkyl group of a phenate (i.e., an alkyl phenate) may contain 4 to 80, or 6 to 45, or 8 to 20, or 9 to 15 carbon atoms.
[0255] In one embodiment the calcium-containing detergent may be a sulphonate, or mixtures thereof. The sulphonate may be prepared from a mono- or di-hydrocarbyl-substituted benzene (or naphthalene, indenyl, indanyl, or bicyclopentadienyl) sulphonic acid, wherein the hydrocarbyl group may contain 6 to 40, or 8 to 35 or 9 to 30 carbon atoms.
[0256] The hydrocarbyl group may be derived from polypropylene or a linear or branched alkyl group containing at least 10 carbon atoms. Examples of a suitable alkyl group include branched and/or linear decyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, octadecenyl, nonodecyl, eicosyl, un-eicosyl, do-eicosyl, tri-eicosyl, tetra-eicosyl, penta-eicosyl, hexa-eicosyl or mixtures thereof.
[0257] In one embodiment the hydrocarbyl-substituted sulphonic acid may include polypropene benzenesulphonic acid and/or C.sub.16-C.sub.24 alkyl benzenesulphonic acid, or mixtures thereof.
[0258] In one embodiment a calcium sulphonate detergent may be a predominantly linear alkylbenzene sulphonate detergent having a metal ratio of at least 8 as is described in paragraphs [0026] to [0037] of US Patent Application 2005065045 (and granted as U.S. Pat. No. 7,407,919). In some embodiments the linear alkyl group may be attached to the benzene ring anywhere along the linear chain of the alkyl group, but often in the 2, 3 or 4 position of the linear chain, and in some instances predominantly in the 2 position.
[0259] When neutral or slightly basic, a calcium sulphonate detergent may have TBN of less than 100, or less than 75, typically 20 to 50 mg KOH/g, or 0 to 20 mg KOH/g.
[0260] When overbased, a calcium sulphonate detergent may have a TBN greater than 200, or 300 to 550, or 350 to 450 mg KOH/g.
[0261] The detergent may be borated or non-borated.
[0262] Chemical structures for sulphonates, and phenates detergents are known to a person skilled in the art. The standard textbook entitled "Chemistry and Technology of Lubricants", Third Edition, Edited by R. M. Mortier and S. T. Orszulik, Copyright 2010, pages 220 to 223 under the sub-heading 7.2.6 provide general disclosures of said detergents and their structures.
[0263] In one embodiment the calcium-containing detergent may be an overbased calcium sulphonate, an overbased calcium phenate, or mixtures thereof. Typically the detergent may be an overbased calcium sulphonate.
[0264] In one embodiment the calcium-containing detergent may be in a mixture with a having zinc-, barium-, sodium-, or magnesium-containing detergent. The zinc-, barium-, sodium-, or magnesium-containing detergent is also well known in the art and described in the same references describing a calcium-containing detergent. The TBN and metal ratios may however, differ slightly. The zinc-, barium-, sodium-, or magnesium-containing detergent may be a phenate, a sulphur-containing phenate, sulphonate, salixarate or salicylate. Typically a zinc-, barium-, sodium-, or magnesium-containing detergent may be a magnesium phenate, a magnesium sulphur-containing phenate, or a magnesium sulphonate.
[0265] The dispersant is described above.
[0266] The lubricant composition may include a friction modifier, typically at least two friction modifiers. Useful friction modifiers are described below.
[0267] In one embodiment the friction modifier may be formed by the condensation of the hydroxyalkyl compound with an acylating agent or an amine. A more detailed description of the hydroxyalkyl compound is described in U.S. Patent Application 60/725360 (filed on Oct. 11, 2005, inventors Bartley, Lahiri, Baker and Tipton) in paragraphs 8, 19-21. The friction modifier disclosed in U.S. Patent Application 60/725360 may be an amide represented by the formula R.sup.1R.sup.2N--C(O)R.sup.3, wherein R.sup.1 and R.sup.2 are each independently hydrocarbyl groups of at least 6 carbon atoms and R.sup.3 is a hydroxyalkyl group of 1 to 6 carbon atoms or a group formed by the condensation of said hydroxyalkyl group, through a hydroxyl group thereof, with an acylating agent. Preparative Examples are disclosed in Examples 1 and 2 (paragraphs 68 and 69). In one embodiment the amide of a hydroxylalkyl compound is prepared by reacting glycolic acid, that is, hydroxyacetic acid, HO--CH.sub.2--COOH with an amine.
[0268] In one embodiment the friction modifier may be a secondary or tertiary amine being represented by the formula R.sup.4R.sup.5NR.sup.6, wherein R.sup.4 and R.sup.5 are each independently an alkyl group of at least 6 carbon atoms and R.sup.6 is hydrogen, a hydrocarbyl group, a hydroxyl-containing alkyl group, or an amine-containing alkyl group. A more detailed description of the friction modifier is described in U.S. patent application Ser. No. 05/037897 in paragraphs 8 and 19 to 22.
[0269] In one embodiment the friction modifier may be derived from the reaction of a carboxylic acid or a reactive equivalent thereof with an aminoalcohol, wherein the friction modifier contains at least two hydrocarbyl groups, each containing at least 6 carbon atoms. An example of such a friction modifier includes the reaction product of isostearic acid or an alkyl succinic anhydride with tris-hydroxymethylaminomethane. A more detailed description of such a friction modifier is disclosed in International Publication WO04/007652) in paragraphs 8 and 9 to 14.
[0270] The friction modifier includes fatty amines, borated glycerol esters, fatty acid amides, non-borated fatty epoxides, borated fatty epoxides, alkoxylated fatty amines, borated alkoxylated fatty amines, metal salts of fatty acids, fatty imidazolines, metal salts of alkyl salicylates (may also be referred to as a detergent), metal salts of sulphonates (may also be referred to as a detergent), condensation products of carboxylic acids or polyalkylene-polyamines, or amides of hydroxyalkyl compounds.
[0271] In one embodiment the friction modifier includes a fatty acid ester of glycerol. The final product may be in the form of a metal salt, an amide, an imidazoline, or mixtures thereof. The fatty acids may contain 6 to 24, or 8 to 18 carbon atoms. The fatty acids may branched or straight-chain, saturated or unsaturated. Suitable acids include 2-ethylhexanoic, decanoic, oleic, stearic, isostearic, palmitic, myristic, palmitoleic, linoleic, lauric, and linolenic acids, and the acids from the natural products tallow, palm oil, olive oil, peanut oil, corn oil, and Neat's foot oil. In one embodiment the fatty acid is oleic acid. When in the form of a metal salt, typically the metal includes zinc or calcium; and the products include overbased and non-overbased products. Examples are overbased calcium salts and basic oleic acid-zinc salt complexes which may be represented by the general formula Zn.sub.4Oleate.sub.6O. When in the form of an amide, the condensation product includes those prepared with ammonia, or with primary or secondary amines such as diethylamine and diethanolamine. When in the form of an imidazoline, the condensation product of an acid with a diamine or polyamine such as a polyethylenepolyamine. In one embodiment the friction modifier is the condensation product of a fatty acid with C.sub.8 to C.sub.24 atoms, and a polyalkylene polyamine, and in particular, the product of isostearic acid with tetraethylenepentamine.
[0272] In one embodiment the friction modifier includes those formed by the condensation of the hydroxyalkyl compound with an acylating agent or an amine. A more detailed description of the hydroxyalkyl compound is described in WO 2007/0044820 paragraphs 9, and 20-22. The friction modifier disclosed in WO2007/044820 includes an amide represented by the formula R.sup.12R.sup.13N--C(O)R.sup.14, wherein R.sup.12 and R.sup.13 are each independently hydrocarbyl groups of at least 6 carbon atoms and R.sup.14 is a hydroxyalkyl group of 1 to 6 carbon atoms or a group formed by the condensation of said hydroxyalkyl group, through a hydroxyl group thereof, with an acylating agent. Preparative Examples are disclosed in Examples 1 and 2 (paragraphs 72 and 73 of WO2007/044820). In one embodiment the amide of a hydroxylalkyl compound is prepared by reacting glycolic acid, that is, hydroxyacetic acid, HO--CH.sub.2--COOH with an amine.
[0273] In one embodiment the friction modifier includes a reaction product of a di-cocoalkyl amine (or di-cocoamine) with glycolic acid. The friction modifier includes compounds prepared in Preparative Examples 1 and 2 of WO 2008/014319.
[0274] In one embodiment the friction modifier includes an alkoxylated alcohol. A detailed description of suitable alkoxylated alcohols is described in paragraphs 19 and 20 of US Patent Application 2005/0101497. The alkoxylated amines are also described in U.S. Pat. No. 5,641,732 in column 7, line 15 to column 9, line 25.
[0275] In one embodiment the friction modifier includes a hydroxyl amine compound as defined in column 37, line 19, to column 39, line 38 of U.S. Pat. No. 5,534,170. Optionally the hydroxyl amine includes borated as such products are described in column 39, line 39 to column 40 line 8 of U.S. Pat. No. 5,534,170.
[0276] In one embodiment the friction modifier includes an alkoxylated amine e.g., an ethoxylated amine derived from 1.8% Ethomeen.TM. T-12 and 0.90% Tomah.TM. PA-1 as described in Example E of U.S. Pat. No. 5,703,023, column 28, lines 30 to 46. Other suitable alkoxylated amine compounds include commercial alkoxylated fatty amines known by the trademark "ETHOMEEN" and available from Akzo Nobel. Representative examples of these ETHOMEEN.TM. materials is ETHOMEEN.TM. C/12 (bis[2-hydroxyethyl]-coco-amine); ETHOMEEN.TM. C/20 (polyoxyethylene-[10]cocoamine); ETHOMEEN.TM. S/12 (bis[2-hydroxyethyl]soyamine); ETHOMEEN.TM. T/12 (bis[2-hydroxyethyl]allow-amine); ETHOMEEN.TM. T/15 (polyoxyethylene-[5]tallowamine); ETHOMEEN.TM. 0/12 (bis[2-hydroxyethyl]oleyl-amine); ETHOMEEN.TM. 18/12 (bis[2-hydroxyethyl]octadecylamine); and ETHOMEEN.TM. 18/25 (polyoxyethylene[15]octadecylamine). Fatty amines and ethoxylated fatty amines are also described in U.S. Pat. No. 4,741,848.
[0277] In one embodiment the friction modifier includes a polyol ester as described in U.S. Pat. No. 5,750,476 column 8, line 40 to column 9, line 28.
[0278] In one embodiment the friction modifier includes a low potency friction modifier as described in U.S. Pat. No. 5,840,662 in column 2, line 28 to column 3, line 26. U.S. Pat. No. 5,840,662 further discloses in column 3, line 48 to column 6, line 25 specific materials and methods of preparing the low potency friction modifier.
[0279] In one embodiment the friction modifier includes a reaction product of an isomerised alkenyl substituted succinic anhydride and a polyamine as described in U.S. Pat. No. 5,840,663 in column 2, lines 18 to 43. Specific embodiments of the friction modifier described in U.S. Pat. No. 5,840,663 are further disclosed in column 3, line 23 to column 4, line 35. Preparative examples are further disclosed in column 4, line 45 to column 5, line 37 of U.S. Pat. No. 5,840,663.
[0280] In one embodiment the friction modifier includes an alkylphosphonate mono- or di-ester sold commercially by Rhodia under the trademark Duraphos.RTM. DMODP.
[0281] The condensation of a fatty acid and a polyamine typically result in the formation of at least one compound chosen from hydrocarbyl amides, hydrocarbyl imidazolines and mixtures thereof. In one embodiment the condensation products are hydrocarbyl imidazolines. In one embodiment the condensation products are hydrocarbyl amides. In one embodiment the condensation products are mixtures of hydrocarbyl imidazolines and hydrocarbyl amides. Typically the condensation product is a mixture of hydrocarbyl imidazolines and hydrocarbyl amides.
[0282] The fatty acid may be derived from a hydrocarbyl carboxylic acid. The hydrocarbyl group may be alkyl, cycloalkyl, or aryl, although alkyl is typical, and the hydrocarbyl groups may be linear or branched. Typically the fatty acid contains 8 or more, 10 or more, more 13 or 14 or more carbon atoms (including the carbon of the carboxy group). Typically the fatty acid contains 8 to 30, 12 to 24, or 16 to 18 carbon atoms. Other suitable carboxylic acids may include the polycarboxylic acids or carboxylic acids or anhydrides having from 2 to 4 carbonyl groups, typically 2. The polycarboxylic acids may include succinic acids and anhydrides and Diels-Alder reaction products of unsaturated monocarboxylic acids with unsaturated carboxylic acids (such as acrylic, methacrylic, maleic, fumaric, crotonic and itaconic acids). The fatty carboxylic acids include fatty monocarboxylic acids containing 8 to 30, 10 to 26, or 12 to 24 carbon atoms.
[0283] Examples of suitable fatty acids may include caprylic acid, capric acid, lauric acid, myristic acid, palmitic acid, stearic acid, eicosic acid and, tall oil acids. In one embodiment the fatty acid is stearic acid, which may be used alone or in combination with other fatty acids.
[0284] One or both friction modifiers may in one embodiment be nitrogen-containing compounds, typically both friction modifiers are nitrogen-containing.
[0285] In one embodiment one of friction modifiers is the condensation product of a fatty acid with C.sub.8 to C.sub.24 atoms, and a polyalkylene polyamine, and in particular, the product of isostearic acid with tetraethylenepentamine.
[0286] The phosphorus-containing compound may be a non-ionic phosphorus compound.
[0287] In one embodiment the phosphorus-containing compounds comprise two or more (possibly up to four) non-ionic phosphorus compounds. Typically the non-ionic phosphorus compound may have an oxidation of +3 or +5. The different embodiments comprise phosphite ester, phosphate esters, or mixtures thereof.
[0288] In one embodiment the phosphorus-containing compound comprises a non-ionic phosphorus compound (a C.sub.4-6 hydrocarbyl phosphite) and an amine salt of a phosphorus acid or ester.
[0289] The phosphorus-containing compound comprises a non-ionic phosphorus compound that is a C.sub.4-6 hydrocarbyl phosphite, or mixtures thereof. The C.sub.4-6 hydrocarbyl phosphite includes those represented by the formula:
##STR00014##
[0290] wherein each R''' may be independently hydrogen or a hydrocarbyl group having 4 to 6 carbon atoms, typically 4 carbon atoms, with the proviso that at least one of the R''' groups is hydrocarbyl. Typically the C.sub.4-6 hydrocarbyl phosphite comprises dibutyl phosphite.
[0291] The C.sub.4-6 hydrocarbyl phosphite may deliver at least 175 ppm, or at least 200 ppm of the total amount of phosphorus delivered by the phosphorus-containing compounds.
[0292] The C.sub.4-6 hydrocarbyl phosphite may deliver at least 45 wt %, or 50 wt % to 100 wt %, or 50 wt % to 90 wt % or 60 wt % to 80 wt % of the total amount of phosphorus from the phosphorus-containing compound.
[0293] The phosphorus-containing compounds may comprise a second phosphite whose formula is similar to that disclosed above, except R''' may contain 2 to 40, 8 to 24 or 11 to 20 carbon atoms, with the proviso that the second phosphite is not a C.sub.4-6 hydrocarbyl phosphite. Examples of suitable hydrocarbyl groups include propyl, dodecyl, butadecyl, hexadecyl, octadecyl, propenyl, dodecenyl, butadecenyl, hexadeencyl, or octadecenylgroups.
[0294] As used herein the term "alk(en)yl" is intended to include moieties that have an alkyl and/or alkenyl group.
[0295] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and a C.sub.12-18 alk(en)yl hydrogen phosphite and optionally phosphoric acid. In different embodiments the phosphoric acid is present or absent.
[0296] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and a C.sub.16-18 alk(en)yl hydrogen phosphite. The alk(en)yl hydrogen phosphite be may an alkyl hydrogen phosphite, and alkenyl hydrogen phosphite, or a mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite. In one embodiment the alk(en)yl hydrogen phosphite be may a mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite and optionally phosphoric acid. The phosphoric acid may be present or absent.
[0297] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and a C.sub.11-14 alk(en)yl hydrogen phosphite. The alk(en)yl hydrogen phosphite be may an alkyl hydrogen phosphite, and alkenyl hydrogen phosphite, ora mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite. In one embodiment the alk(en)yl hydrogen phosphite may be a mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite and optionally phosphoric acid.
[0298] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and phosphoric acid.
[0299] The lubricant composition in one embodiment includes a package that comprises a phosphorus-containing compound and a non-ionic phosphorus compound that is a hydrocarbyl phosphite.
[0300] In one embodiment the lubricant composition further comprises a C.sub.8-20 hydrocarbyl phosphite, or a C.sub.12-18 hydrocarbyl phosphite, or C.sub.6-18 hydrocarbyl phosphite.
[0301] In one embodiment the lubricant composition includes an amine antioxidant. The amine antioxidant may be a phenyl-a-naphthylamine (PANA) or a hydrocarbyl substituted diphenylamine, or mixtures thereof. The hydrocarbyl substituted diphenylamine may include mono- or di-C.sub.4 to C.sub.16-, or C.sub.6 to C.sub.12-, or C.sub.9-alkyl diphenylamine. For example the hydrocarbyl substituted diphenylamine may be octyl diphenylamine, or di-octyl diphenylamine, dinonyl diphenylamine, typically dinonyl diphenylamine.
[0302] When present the amine antioxidant may be present at 0.2 wt % to 1.2 wt %, or 0.3 wt % to 1.0 wt %, or 0.4 wt % to 0.9 wt % or 0.5 wt % to 0.8 wt %, of the lubricant composition.
[0303] The lubricant composition be optionally include at least one other antixodiant that is known and includes sulphurised olefins, hindered phenols, molybdenum dithiocarbamates, and mixtures thereof.
[0304] The hindered phenol antioxidant often contains a secondary butyl and/or a tertiary butyl group as a sterically hindering group. The phenol group is often further substituted with a hydrocarbyl group and/or a bridging group linking to a second aromatic group. Examples of suitable hindered phenol antioxidants include 2,6-di-tert-butylphenol, 4-methyl-2,6-di-tert-butylphenol, 4-ethyl-2,6-di-tert-butylphenol, 4-propyl-2,6-di-tert-butylphenol or 4-butyl-2,6-di-tert-butylphenol, or 4-dodecyl-2,6-di-tert-butylphenol. In one embodiment the hindered phenol antioxidant may be an ester and may include, e.g., Irganox.TM. L-135 from Ciba, or butyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propanoate.
[0305] If present, the secondary antioxidant may be present at 0.1 wt % to 1 wt %, or 0.2 wt % to 0.9 wt % or 0.1 wt % to 0.4 wt %, or 0.4 wt % to 1.0 wt %, of the lubricant composition.
Off-Highway Vehicle
[0306] The polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described herein can be employed in off-highway vehicles, such as, for example, farm tractors and construction vehicles. Such vehicles often have a common sump that lubricates not only the transmission but also the gears, axles, and hydraulics in the vehicle. In one embodiment the invention includes a lubricant composition comprising:
[0307] an oil of lubricating viscosity,
[0308] a dispersant typically present at 0.1 to 3 wt %, or 0.1 to 2.5 wt %, or 0.2 to 2 wt %,
[0309] from about 0.1 to about 5 wt % of a detergent, typically present in an amount to deliver 100 to 5000 ppm, 500 to 4500 ppm, 500 to 4250 ppm, 650 to 4200 ppm calcium; and wherein from about 0.01 to about 2 wt %, or from about 0.1 to about 1.75 wt %, or about 0.2 to about 1.5 wt % of the detergent comprises the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described herein.
[0310] In one embodiment the invention includes a method of lubricating an off-highway vehicle comprising supplying to the vehicle a lubricant composition comprising:
[0311] an oil of lubricating viscosity,
[0312] a dispersant typically present at 0.1 to 3 wt %, or 0.1 to 2.5 wt %, or 0.2 to 2 wt %,
[0313] a phosphorus-containing antiwear agent chosen from (i) a non-ionic phosphorus compound, which may be a hydrocarbyl phosphite, such as a zinc dialkyldithiophosphate; or (ii) an amine salt of a phosphorus compound,
[0314] from about 0.1 to about 5 wt % of a detergent, typically present in an amount to deliver 10 to 5000ppm, 500 to 4500ppm, 500 to 4250ppm, 650 to 4200ppm calcium; and wherein from about 0.01 to about 2 wt %, or from about 0.1 to about 1.75 wt %, or about 0.2 to about 1.5 wt % of the detergent comprises the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof described herein.
[0315] The off-highway vehicle lubricated typically has a wet-brake, a transmission, a hydraulic, a final drive, a power take-off system. These parts are typically lubricated by a single lubricant supplied from a common sump. The transmission may be a manual transmission or an automatic transmission.
[0316] The calcium-containing detergent may be an overbased detergent, a non-overbased detergent, or mixtures thereof. Typically the detergent may be overbased.
[0317] The preparation of the calcium-containing detergent is known in the art. Patents describing the preparation of overbased calcium-containing detergents include U.S. Pat. Nos. 2,501,731; 2,616,905; 2,616,911; 2,616,925; 2,777,874; 3,256,186; 3,384,585; 3,365,396; 3,320,162; 3,318,809; 3,488,284; and 3,629,109.
[0318] The other detergent (other than the alkylphenol) and dispersant is described above.
[0319] The phosphorus-containing antiwear agent may include zinc dialkyldithiophosphate, a non-ionic phosphorus compound, which may be a hydrocarbyl phosphite; (i) a non-ionic phosphorus compound, which may be a hydrocarbyl phosphite; or (ii) an amine salt of a phosphorus compound, or mixtures thereof.
[0320] In one embodiment the lubricant composition disclosed herein contains no zinc dialkyldithiophosphate.
[0321] In one embodiment the lubricant composition disclosed herein contains zinc dialkyldithiophosphate.
[0322] The phosphorus-containing compound may be a non-ionic phosphorus compound.
[0323] In one embodiment the phosphorus-containing compounds comprise two or more (possibly up to four) non-ionic phosphorus compounds. Typically the non-ionic phosphorus compound may have an oxidation of +3 or +5. The different embodiments comprise phosphite ester, phosphate esters, or mixtures thereof.
[0324] In one embodiment the phosphorus-containing compound comprises a non-ionic phosphorus compound (a C.sub.4-6 hydrocarbyl phosphite) and an amine salt of a phosphorus acid or ester.
[0325] The phosphorus-containing compound comprises a non-ionic phosphorus compound that is a C.sub.4-6 hydrocarbyl phosphite, or mixtures thereof. The C.sub.4-6 hydrocarbyl phosphite includes those represented by the formula:
##STR00015##
wherein each R''' may be independently hydrogen or a hydrocarbyl group having 4 to 6 carbon atoms, typically 4 carbon atoms, with the proviso that at least one of the R''' groups is hydrocarbyl. Typically the C.sub.4-6 hydrocarbyl phosphite comprises dibutyl phosphite.
[0326] The C.sub.4-6 hydrocarbyl phosphite may deliver at least 175 ppm, or at least 200 ppm of the total amount of phosphorus delivered by the phosphorus-containing compounds.
[0327] The C.sub.4-6 hydrocarbyl phosphite may deliver at least 45 wt %, or 50 wt % to 100 wt %, or 50 wt % to 90 wt % or 60 wt % to 80 wt % of the total amount of phosphorus from the phosphorus-containing compound.
[0328] The phosphorus-containing compounds may comprise a second phosphite whose formula is similar to that disclosed above, except R''' may contain 2 to 40, 8 to 24 or 11 to 20 carbon atoms, with the proviso that the second phosphite is not a C.sub.4-6 hydrocarbyl phosphite. Examples of suitable hydrocarbyl groups include propyl, dodecyl, butadecyl, hexadecyl, octadecyl, propenyl, dodecenyl, butadecenyl, hexadeencyl, or octadecenylgroups.
[0329] As used herein the term "alk(en)yl" is intended to include moieties that have an alkyl and/or alkenyl group.
[0330] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and a C.sub.12-18 alk(en)yl hydrogen phosphite and optionally phosphoric acid.In different embodiments the phosphoric acid is present or absent.
[0331] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and a C.sub.16-18 alk(en)yl hydrogen phosphite. The alk(en)yl hydrogen phosphite be may an alkyl hydrogen phosphite, and alkenyl hydrogen phosphite, ora mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite. In one embodiment the alk(en)yl hydrogen phosphite be may a mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite and optionally phosphoric acid. The phosphoric acid may be present or absent.
[0332] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and a C.sub.11-14 alk(en)yl hydrogen phosphite. The alk(en)yl hydrogen phosphite be may an alkyl hydrogen phosphite, and alkenyl hydrogen phosphite, ora mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite. In one embodiment the alk(en)yl hydrogen phosphite may be a mixture of alkenyl hydrogen phosphite and alkyl hydrogen phosphite and optionally phosphoric acid.
[0333] In one embodiment the phosphorus-containing compounds include a mixture of a C.sub.4-6 hydrocarbyl phosphite (typically dibutyl phosphite) and phosphoric acid.
[0334] The lubricant composition in one embodiment includes a package that comprises a phosphorus-containing compound and a non-ionic phosphorus compound that is a hydrocarbyl phosphite.
[0335] In one embodiment the lubricant composition further comprises a C.sub.8-20 hydrocarbyl phosphite, or a C.sub.12-18 hydrocarbyl phosphite, or C.sub.16-18 hydrocarbyl phosphite, as described above.
[0336] In on embodiment the amine salt of a phosphorus acid may be derived from an amine salt of a phosphate. The amine salt of the phosphorus acidmay be represented by the formula:
##STR00016##
wherein [0337] R.sup.3 and R.sup.4 may be independently hydrogen or hydrocarbon typically containing 4 to 40, or 6 to 30, or 6 to 18, or 8 to 18 carbon atoms, with the proviso that at least one is a hydrocarbon group; and [0338] R.sup.5, R.sup.6, R.sup.7 and R.sup.8 may be independently hydrogen or a hydrocarbyl group, with the proviso that at least one is a hydrocarbyl group.
[0339] The hydrocarbon groups of R.sup.3 and/or R.sup.4 may be linear, branched, or cyclic.
[0340] Examples of a hydrocarbon group for R.sup.3 and/or R.sup.4 include straight-chain or branched alkyl groups include methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl and octadecyl.
[0341] Examples of a cyclic hydrocarbon group for R.sup.3 and/or R.sup.4 include cyclopentyl, cyclohexyl, cycloheptyl, methylcyclopentyl, dimethylcyclopentyl, methyl-cyclopentyl, dimethylcyclopentyl, methylethylcyclopentyl, diethylcyclopentyl, methylcyclohexyl, dimethylcyclohexyl, methylethylcyclohexyl, diethylcyclohexyl, methylcycloheptyl, dimethylcycloheptyl, methylethylcycloheptyl, and diethylcycloheptyl.
[0342] In one embodiment the phosphate may be an amine salt of a mixture of monoalkyl and dialkyl phosphoric acid esters. The monoalkyl and dialkyl groups may be linear or branched.
[0343] The amine salt of a phosphorus acid may be derived from an amine such as a primary amine, a secondary amine, a tertiary amine, or mixtures thereof. The amine may be aliphatic, or cyclic, aromatic or non-aromatic, typically aliphatic. In one embodiment the amine includes an aliphatic amine such as a tertiary-aliphatic primary amine.
[0344] Examples of suitable primary amines include ethylamine, propylamine, butylamine, 2-ethylhexylamine, bis-(2-ethylhexyl)amine, octylamine, and dodecyl-amine, as well as such fatty amines as n-octylamine, n-decylamine, n-dodecylamine, n-tetradecylamine, n-hexadecylamine, n-octadecylamine and oleyamine. Other useful fatty amines include commercially available fatty amines such as "Armeen.RTM." amines (products available from Akzo Chemicals, Chicago, Illinois), such as Armeen C, Armeen O, Armeen OL, Armeen T, Armeen HT, Armeen S and Armeen SD, wherein the letter designation relates to the fatty group, such as coco, oleyl, tallow, or stearyl groups.
[0345] Examples of suitable secondary amines include dimethylamine, diethylamine, dipropylamine, dibutylamine, diamylamine, dihexylamine, diheptylamine, methylethylamine, ethylbutylamine, N-methyl-1-amino-cyclohexane, Armeen.RTM. 2C and ethylamylamine. The secondary amines may be cyclic amines such as piperidine, piperazine and morpholine.
[0346] Examples of tertiary amines include tri-n-butylamine, tri-n-octylamine, tri-decylamine, tri-laurylamine, tri-hexadecylamine, and dimethyloleylamine (Armeen.RTM. DMOD).
[0347] In one embodiment the amines are in the form of a mixture. Examples of suitable mixtures of amines include (i) a tertiary alkyl primary amine with 11 to 14 carbon atoms, (ii) a tertiary alkyl primary amine with 14 to 18 carbon atoms, or (iii) a tertiary alkyl primary amine with 18 to 22 carbon atoms. Other examples of tertiary alkyl primary amines include tert-butylamine, tert-hexylamine, tert-octylamine (such as 1,1-dimethylhexylamine), tert-decylamine (such as 1,1-dimethyloctylamine), tertdodecylamine, tert-tetradecylamine, tert-hexadecylamine, tert-octadecylamine, tert-tetracosanylamine, and tert-octacosanylamine.
[0348] In one embodiment a useful mixture of amines is "Primene.RTM. 81R" or "Primene.RTM. JMT." Primene.RTM. 81R and Primene.RTM. JMT (both produced and sold by Rohm & Haas) are mixtures of C.sub.11 to C.sub.14 tertiary alkyl primary amines and C.sub.18 to C.sub.22 tertiary alkyl primary amines respectively.
[0349] The amine salt of a phosphorus acid may be prepared as is described in US Patent 6,468,946. Column 10, lines 15 to 63 describes phosphoric acid esters formed by reaction of phosphorus compounds, followed by reaction with an amine to form an amine salt of a phosphate hydrocarbon ester. Column 10, line 64, to column 12, line 23, describes preparative examples of reactions between phosphorus pentoxide with an alcohol (having 4 to 13 carbon atoms), followed by a reaction with an amine (typically Primene.RTM.81-R) to form an amine salt of a phosphate hydrocarbon ester.
[0350] The sulphur-containing extreme pressure agent may be an olefin sulphide, or mixtures thereof. The olefin sulphide may include a polysulphide or a sulphurised olefin such as sulphurised isobutylene, or mixtures thereof.
[0351] In one embodiment the olefin sulphide includes a polysulphide.
[0352] In one embodiment the olefin sulphide includes sulphurized isobutylene.
[0353] In one embodiment the olefin sulphide includes a mixture of a sulphurised isobutylene and a polysulphide.
[0354] In one embodiment at least 50 wt % of the polysulphide molecules are a mixture of tri- or tetra-sulphides. In other embodiments at least 55 wt %, or at least 60 wt % of the polysulphide molecules are a mixture of tri- or tetra-sulphides.
[0355] The polysulphide includes a sulphurised organic polysulphide from oils, fatty acids or ester, olefins or polyolefins.
[0356] Oils which may be sulfurized include natural or synthetic oils such as mineral oils, lard oil, carboxylate esters derived from aliphatic alcohols and fatty acids or aliphatic carboxylic acids (e.g., myristyl oleate and oleyl oleate), and synthetic unsaturated esters or glycerides.
[0357] Fatty acids include those that contain 8 to 30, or 12 to 24 carbon atoms. Examples of fatty acids include oleic, linoleic, linolenic, and tall oil. Sulphurised fatty acid esters prepared from mixed unsaturated fatty acid esters such as are obtained from animal fats and vegetable oils, including tall oil, linseed oil, soybean oil, rapeseed oil, and fish oil.
[0358] The polysulphide includes olefins derived from a wide range of alkenes. The alkenes typically have one or more double bonds. The olefins in one embodiment contain 3 to 30 carbon atoms. In other embodiments, olefins contain 3 to 16, or 3 to 9 carbon atoms. In one embodiment the sulphurised olefin includes an olefin derived from propylene, isobutylene, pentene or mixtures thereof.
[0359] In one embodiment the polysulphide comprises a polyolefin derived from polymerising by known techniques, an olefin as described above.
[0360] In one embodiment the polysulphide includes dibutyl tetrasulphide, sulphurised methyl ester of oleic acid, sulphurised alkylphenol, sulphurised dipentene, sulphurised dicyclopentadiene, sulphurised terpene, and sulphurised Diels-Alder adducts.
[0361] The lubricant may also include a sulphur-containing corrosion inhibitor. The sulphur-containing corrosion inhibitor may be a thiadiazole compound, or mixtures thereof. The thiadiazole compound may include mono- or di-hydrocarbyl substituted 2,5-dimercapto-1,3,4-thiadiazole compounds. Examples of a thiadiazole include 2,5-dimercapto-1,3,4-thiadiazole, or oligomers thereof, a hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole, a hydrocarbylthio-substituted 2,5-dimercapto-1,3,4-thiadiazole, or oligomers thereof. The oligomers of hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole typically form by forming a sulphur-sulphur bond between 2,5-dimercapto-1,3,4-thiadiazole units to form oligomers of two or more of said thiadiazole units. These thiadiazole compounds may also be used in the post treatment of dispersants as mentioned below in the formation of a dimercaptothiadiazole derivative of a polyisobutylene succinimide.
[0362] Examples of a suitable thiadiazole compound include at least one of a dimercaptothiadiazole, 2,5-dimercapto-[1,3,4]-thiadiazole, 3,5-dimercapto-[1,2,4]-thiadiazole, 3,4-dimercapto-[1,2,5]-thiadiazole, or 4-5-dimercapto-[1,2,3]-thiadiazole. Typically readily available materials such as 2,5-dimercapto-1,3,4-thiadiazole or a hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole or a hydrocarbylthio-substituted 2,5-dimercapto-1,3,4-thiadiazole are commonly utilised.
[0363] Without intending to limit the scope of the exemplary embodiment, the following examples illustrate preparation and evaluation of example compounds.
EXAMPLES
Example 1
Preparation of Isobutylene-Substituted Phenol:
[0364] Phenol is alkylated with a 350 Mn high vinylidene polyisobutylene (obtained from Texas Petroleum Products) under standard conditions using boron trifluoride as a catalyst, as shown in reaction scheme 1 above.
[0365] Phenol (271.5 g) and toluene (131.2 g) are first mixed in a nitrogen blanketed vessel followed by the dropwise addition of BF.sub.3 phenol complex (43.7g) over 20 mins. To this mixture, a solution of 350Mn polyisobutylene (404.3 g, Texas Petroleum Products) diluted in toluene (132.3 g) is added dropwise over 4 hrs. The addition rate is controlled to maintain a temperature between 13-21.degree. C. and never exceeded 28.degree. C.
[0366] The reaction mixture is then neutralized slowly with calcium hydroxide (84.0 g) with the temperature being held below 28.degree. C. The neutralized reaction mixture is held for 3 hrs following which diatomaceous earth (100 g) is charged to the reaction and the neutralization completed with ammonia hydroxide (10 g). The reaction mixture is filtered and the filtrate heated under vacuum to a temperature of 185.degree. C. to remove volatiles by distillation. The resulting material is a 350 Mn polyisobutylene-alkylated phenol.
[0367] NMR analysis shows the product to be predominately the para-substituted isomer with <3.0% free polyisobutylene present.
Example 2
Bridging of Polyisobutylene-Alkylated Phenol
[0368] The material of Example 1 is used to synthesize a neutral calcium phenate using the method shown in scheme 2, above.
[0369] The 350 Mn polyisobutylene-alkylated phenol (250.2 g) is heated to 100.degree. C. while stirring in a vessel under a nitrogen blanket. Calcium hydroxide (22.2 g) and ethylene glycol (17.4 g) are added and the mixture is heated to 124.degree. C. Sulfur (45.4 g) is added and the mixture is heated to 185.degree. C. The reaction is held at this temperature for 7 hours. Diluent oil (52.7 g) is added and the mixture is then cooled to room temperature.
[0370] Ethylene glycol (13.7 g) and n-decanol (31.9 g) are added to the reaction mixture with stirring, and the mixture is then heated to 168.degree. C. under a nitrogen blanket. Calcium hydroxide (17.3 g) is then added and the mixture is heated to 168.degree. C. while removing volatiles by distillation. Additional diluent oil (39.3 g) is added to the reaction mixture which is heated to 220.degree. C. with vacuum (13 kPa pressure, 27'' Hg vacuum) for 3 hours. The mixture is cooled to 100.degree. C. and then filtered to yield the final product, in diluent oil.
Example 3
Polyisobutylene-Substituted Bridged Calcium Phenate Having an Oil Free TBN of about 400
[0371] A polyisobutylene-substituted bridged calcium phenate having an oil free TBN of about 400 was prepared using the following procedure: The material of Example 1 (400 g) is heated to 100.degree. C. while stirring in a vessel under a nitrogen blanket. Calcium hydroxide (23.6 g) and ethylene glycol (8.9 g) are added and the mixture is heated to 124.degree. C. Sulfur (68.5 g) is added and the mixture is heated to 185.degree. C. The reaction is held at this temperature for 5 hours. Diluent oil (98.2 g) is added and the mixture is then cooled to room temperature.
[0372] Ethylene glycol (110.2 g) and n-decanol (140 g) are added to the reaction mixture with stirring, followed by the addition of alkylbenzene sulfonic acid (34 g) and calcium hydroxide (186.1 g) and the mixture is then heated to 168.degree. C. under a nitrogen blanket. Carbon dioxide (65.3 g) is then introduced via subline over 2 hours. Volatile components are then removed by heating the reaction mixture to 220.degree. C., applying vacuum to the system (27'' Hg) and holding for 1 hour. The mixture is cooled to 100.degree. C., with the addition of diluent oil (349.2 g) and polyisobutylene-substituted succinic anhydride (59.4 g), and then filtered to yield the final product, in diluent oil.
Example 4
Polyisobutylene-Substituted Bridged Calcium Phenate Having an Oil Free TBN of about 200
[0373] A polyisobutylene-substituted bridged calcium phenate having an oil free TBN of about 200 was prepared using the following procedure: The material of Example 1 (500 g) is heated to 100.degree. C. while stirring in a vessel under a nitrogen blanket. Calcium hydroxide (44.4 g) and ethylene glycol (34.8 g) are added and the mixture is heated to 124.degree. C. Sulfur (90.7 g) is added and the mixture is heated to 185.degree. C. The reaction is held at this temperature for 7 hours. Diluent oil (103 g) is added and the mixture is then cooled to room temperature.
[0374] Ethylene glycol (27.4 g) and n-decanol (64.2 g) are added to the reaction mixture with stirring, followed by the addition of calcium hydroxide (34.9 g) and the mixture is then heated to 168.degree. C. under a nitrogen blanket and held for 3 hours. Volatile components are then removed by heating the reaction mixture to 220.degree. C., applying vacuum to the system (27'' Hg) and holding for 1 hour. The mixture is cooled to 100.degree. C., with the addition of diluent oil (112.8 g) and then filtered to yield the final product, in diluent oil.
Example 5
Polyisobutylene-Substituted Bridged Calcium Phenate Having an Oil Free TBN of about 550
[0375] A polyisobutylene-substituted bridged calcium phenate having an oil free TBN of about 550 could be prepared using the following procedure: The material of Example 1 (1000 g) is heated to 107.degree. C. while stirring in a vessel under a nitrogen blanket. Calcium hydroxide (306.9 g) is added and stirred for 5 minutes, followed by the addition of acetic acid (11.5 g) while maintaining the temperature below 110.degree. C. The reaction mixture is the heated to 141.degree. C. and stearic acid (276.1 g) and n-decanol (535 g), followed by increasing the temperature to 154.degree. C. and addition of ethylene glycol (168 g). Carbon dioxide (131.1 g) is then added as fast as possible maintaining the temperature below 168.degree. C. The temperature is reduced to 154.degree. C. and volatiles removed under vacuum (150 mm Hg) and held for 4 hours. The reaction temperature is increased to 220.degree. C. and the vacuum increased to 40 mm Hg. Diluent oil (156.4 g) and diatomaceous earth (104.2 g) is added, and then filtered to yield the final product, in diluent oil.
Example 6
Polyisobutylene-Substituted Bridged Calcium Phenate Having an Oil Free TBN of about 300
[0376] A polyisobutylene-substituted bridged calcium phenate having an oil free TBN of about 300 could be prepared using the following procedure: The material of Example 1 (555 g) is heated to 100.degree. C. while stirring in a vessel under a nitrogen blanket. Calcium hydroxide (23.6 g) and ethylene glycol (8.9 g) are added and the mixture is heated to 124.degree. C. Sulfur (92 g) is added and the mixture is heated to 185.degree. C. The reaction is held at this temperature for 5 hours. Diluent oil (98.2 g) is added and the mixture is then cooled to room temperature.
[0377] Ethylene glycol (110.2 g) and n-decanol (140 g) are added to the reaction mixture with stirring, followed by the addition calcium hydroxide (130 g) and the mixture is then heated to 168.degree. C. under a nitrogen blanket. Carbon dioxide (63.2 g) is then introduced via subline over 2 hours. Volatile components are then removed by heating the reaction mixture to 220.degree. C., applying vacuum to the system (27'' Hg) and holding for 1 hour. The mixture is cooled to 100.degree. C., with the addition of diluent oil (280 g), and then filtered to yield the final product, in diluent oil.
Analysis
[0378] Inductively Coupled Plasma (ICP) analysis of the polyisobutylene-substituted bridged calcium phenate of Example 2 confirms that the elemental sulfur and calcium contents (measured as wt. % of the product, which includes some diluent in addition to the compound) agree with the theoretical values, as shown in TABLE 2.
[0379] TBN is measured according to ASTM D2896 on an oil free basis.
TABLE-US-00002 TABLE 2 Analytical Results Wt. % S by ICP Wt. % Ca by ICP TBN Theoretical composition 4.9 5.1 190 Example 2 compound 4.2 5.0 135 Example 3 compound 4.6 14.7 391 Comparative overbased 4.7 5.2 199 calcium phenate
[0380] A GPC of the experimental polyisobutylene-substituted calcium phenate shows a number of peaks of increasing molecular weight indicating a number of oligomers including monomers, dimers, trimers, and tetramers, primarily dimers and trimers (i.e., m is 1-2 in Formula 2 above).
Preparation of Lubricating Composition
[0381] The sulfur bridged calcium phenate of Example 3 was blended into a driveline lubricating composition to provide Example 7 below.
Example 7
TABLE-US-00003 [0382] Component wt % .sup.1 11.6 cSt Gp I base oil 77.7 4.2 cSt Gp I base oil 13.7 PiB Succinic ester type dispersant 0.3 520TBN Ca Sulfonate detergent 0.8 Neutral Ca Sulfonate detergent 0.5 ZDDP Antiwear 2.6 Phenate of Example 3 0.7 Additive components (seal swell, 0.9 antifoams, PPD, solubilzers etc) Additional diluent oil Balance to 100 .sup.1 Oil free basis
[0383] A polyolefin-substituted bridged hydroxyaromatic compound of the present invention may be blended into driveline lubricating compositions as shown in the examples below. All wt % for components are on an oil free basis. Example 8
TABLE-US-00004 Component Wt % PiB succinic ester type dispersant 3.99 480TBN Calcium sulfonate 9.06 Neutral Calcium sulfonate 4.59 ZDDP Antiwear 29.77 Friction Modifier 1.37 Additional additives (seal swell agent, 8.17 antifoams and rust inhibitor) Compound of the invention 6.08 Base oil Balance to 100
Example 9
TABLE-US-00005 [0384] Component Wt % PiB succinimide dispersant 16.41 520TBN Calcium sulfonate 5.93 Neutral Ca Sulfonate detergent 3.13 ZDDP Antiwear 12.97 Alkylphosphite antiwear 1.56 Friction modifier package 4.68 (Alkenyl amide & Borate ester) Antifoams 0.03 Compound of the invention 10.94 Base Oil Balance to 100
Example 10
TABLE-US-00006 [0385] Component Wt % Mixed PiB succinic ester/amide 3.06 dispersant ZDDP antiwear 20.59 Alkyl phosphite antiwear 6.86 520 TBN Mg sulfonate detergent 3.31 Addititional additives (solubulizer, 47.74 friction modifier, antifoam) Compound of the invention 9.05 Base oil Balance to 100
Example 11
TABLE-US-00007 [0386] Component Wt % 520TBN detergent 7.60 170TBN Ca sulfonate detergent 10.35 PiB succinimide dispersant 1.73 ZDDP antiwear 6.29 Additional additives (antioxidant, rust 7.61 inhibitor, friction modifier) Compound of the invention 25.56 Base Oil Balance to 100
[0387] The lubricating oil of Example 7 (100.+-.2 ml) was heated (160.degree. C., 192hours) in accordance with the oxidation procedure outlined in CEC test L-48-00 (test method available from Coordinating European Council, Kellen Europe, Avenue Jules Bordet 142-1140 Brussels, Belgium (August 2015) by following the sections outlining the glassware and heating apparatus (Section 4), test preparation (Section 6), test operation (Section 7) and sludge rating (Section 8).
[0388] The dispersancy assessment was made using the following `Spot Rating` method. (1) 25 .mu.l of the oxidised oil sample is placed onto #1 chromatography paper (Whatman 3001-917 Grade 1 Cellulose Chromatography Paper Sheet) using a 25 .mu.L pipette with disposable tip; (2) The chromatography paper is dried in an oven (60.degree. C., 24 hours); (3) The vertical and horizontal diameters of the innermost spot is measured in millimetres. In the case of a non-circular spot, the largest measurement of the vertical and horizontal diameters are used; (4) The vertical and horizontal diameters of the outermost spot is measured in millimetres. In the case of a non-circular spot, the largest measurement of the vertical and horizontal diameters are used; (5) Spot rating =(The average of the innermost diameter spot/the average of the outermost diameter spot, this is usually the corona oil).times.100; (6) The spot rating can range from 100% (a clean, fully dispersed spot) to 1% (a very heavily sludged-out spot or a spot that is not fully dried).
[0389] The viscosity, viscosity index, total acid number, corrected peak area increase, dispersancy assessment, and tube assessment were measured. The results of the evaluation are summarized in Table 3:
TABLE-US-00008 TABLE 3 Pre- Post % Test Oxidation Oxidation Change Viscosity at 40.degree. C. D445 97.61 144.1 47.63 (cSt) Viscosity at 100.degree. C. D445 11.15 14.53 30.31 (cSt) Viscosity Index D2270 99 99 Total Acid Number, Buffer D664* 2.89 7.19 point (mg KOH/g) Corrected Peak Area D7214 Not 464.29 Increase (A cm.sup.-1/mm) Applicable Dispersancy See above Not 94 Assessment (%) Applicable Tube assessment CEC Not 2 L-48-00 Applicable *A volume of 60 ml rather than 125 ml of titrant was used (Section 12.1 of test method).
[0390] Other test methods which may be used to evaluate the efficacy of a lubricating composition made in accordance with the present invention include: FZG Scuffing: ASTM D5182, FZG Tractor: ASTM D4998, Copper Strip: ASTM D130, Foam testing: ASTM D892, and Compatibility with Caterpillar Elastomers (as outlined in the TO-4 specification). In addition, certain lubricating compositions may be evaluated to determine if they meet the requirements of various application requirements, such as the Caterpillar Transmission and Drive Train Fluid Requirements (CAT STM SEQ 1222). In addition, the toxicity of the components, in particular the polyolefin-substituted bridged hydroxyaromatic compound or salt thereof, or the alkyl-phenol used to form the compound, may be evaluated using tests such as the PolarScreen.TM. Estrogen Receptor Competitor Assay, as well as transactivation assays, or any other tests designed to assess the potential ability of compounds to bind to the estrogen receptor. Other tests may be used to evaluate the toxicity of the compounds of this invention to show the lower toxicity effect.
[0391] The singular forms "a," "an," and "the" include plural referents unless the context clearly dictates otherwise.
[0392] Unless otherwise stated, molecular weight herein is reported as number average molecular weight, in Daltons, and is measured by Gel Permeation Chromatography (GPC) using a polyisobutylene standard.
[0393] As used herein, the term "hydrocarbyl substituent" or "hydrocarbyl group" is used in its ordinary sense, which is well-known to those skilled in the art. Specifically, it refers to a group having a carbon atom directly attached to the remainder of the molecule and having predominantly hydrocarbon character. By predominantly hydrocarbon character, it is meant that at least 70% or at least 80% of the atoms in the substituent are hydrogen or carbon. Hydrocarbylene groups are the bivalent equivalents of hydrocarbyl groups, i.e., are attached at each end to two parts of the remainder of the molecule.
[0394] Examples of hydrocarbyl groups include:
[0395] (i) hydrocarbon substituents, that is, aliphatic (e.g., alkyl or alkenyl), alicyclic (e.g., cycloalkyl, cycloalkenyl) substituents, aryl, and aromatic-, aliphatic-, and alicyclic-substituted aromatic substituents, as well as cyclic substituents wherein the ring is completed through another portion of the molecule (e.g., two substituents together form a ring);
[0396] (ii) substituted hydrocarbon substituents, that is, substituents containing non-hydrocarbon groups which, in the context of this invention, do not alter the predominantly hydrocarbon nature of the substituent (e.g., halo (especially chloro and fluoro), hydroxy, alkoxy, mercapto, alkylmercapto, nitro, nitroso, and sulfoxy);
[0397] (iii) hetero substituents, that is, substituents which, while having a predominantly hydrocarbon character, may contain other than carbon in a ring or chain otherwise composed of carbon atoms.
[0398] Representative alkyl groups useful as hydrocarbyl groups may include at least 1, or at least 2, or at least 3, or at least 4 carbon atoms, and in some embodiments, up to 8, or up to 10, or up to 12, or up to 14, or up to 16, or up to 18 carbon atoms. Illustrative examples include methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, octyl, 2-ethylhexyl, nonyl, decyl, undecyl, dodecyl, tridecyl, tetradecyl, hexadecyl, stearyl, icosyl, docosyl, tetracosyl, 2-butyloctyl, 2-butyldecyl, 2-hexyloctyl, 2-hexydecyl, 2-octyldecyl, 2-hexyldodecyl, 2-octyldodecyl, 2-decyltetradecyl, 2-dodecylhexadecyl, 2-hexyldecyloctyldecyl, 2-tetradecyloctyldecyl, 4-methyl-2-pentyl, 2-propylheptyl, monomethyl branched-isostearyl, isomers thereof, mixtures thereof, and the like.
[0399] Representative alkenyl groups useful as hydrocarbyl groups include C.sub.2-C.sub.18 alkenyl groups, such as ethynyl, 2-propenyl, 1-methylene ethyl, 2-butenyl, 3-butenyl, pentenyl, hexenyl, heptenyl, octenyl, 2-ethylhexenyl, nonenyl, decenyl, undecenyl, dodecenyl, tridecenyl, tetradecenyl, hexadecenyl, isomers thereof, mixtures thereof, and the like.
[0400] Representative alicyclic groups useful as hydrocarbyl groups include cyclobutyl, cyclopentyl, and cyclohexyl groups.
[0401] Representative aryl groups include phenyl, toluyl, xylyl, cumenyl, mesityl, benzyl, phenethyl, styryl, cinnamyl, benzhydryl, trityl, ethylphenyl, propylphenyl, butylphenyl, pentylphenyl, hexylphenyl, heptylphenyl, octylphenyl, nonylphenyl, decylphenyl, undecylphenyl, dodecylphenyl, benzylphenyl, styrenated phenyl, p-cumylphenyl, .alpha.-naphthyl, .beta.-naphthyl groups, and mixtures thereof.
[0402] Representative heteroatoms include sulfur, oxygen, nitrogen, and encompass substituents, such as pyridyl, furyl, thienyl and imidazolyl. In general, no more than two, and in one embodiment, no more than one, non-hydrocarbon substituent will be present for every ten carbon atoms in the hydrocarbyl group. In some embodiments, there are no non-hydrocarbon substituents in the hydrocarbyl group.
[0403] Numerical values in the specification and claims of this application should be understood to include numerical values which are the same when reduced to the same number of significant figures and numerical values which differ from the stated value by less than the experimental error of conventional measurement technique of the type described in the present application to determine the value.
[0404] As used herein, the term "comprising" is inclusive and does not exclude additional, un-recited elements or method steps. However, in each recitation of "comprising" herein, it is intended that the term also encompasses, as alternative embodiments, the phrases "consisting essentially of" and "consisting of," where "consisting of" excludes any element or steps not specified and "consisting essentially of" permits the inclusion of additional un-recited elements or steps that do not materially affect the basic and novel, and essential characteristics of the composition or method under consideration.
[0405] It will be appreciated that variants of the above-disclosed and other features and functions, or alternatives thereof, may be combined into many other different systems or applications. Various presently unforeseen or unanticipated alternatives, modifications, variations or improvements therein may be subsequently made by those skilled in the art which are also intended to be encompassed by the following claims.
* * * * *

















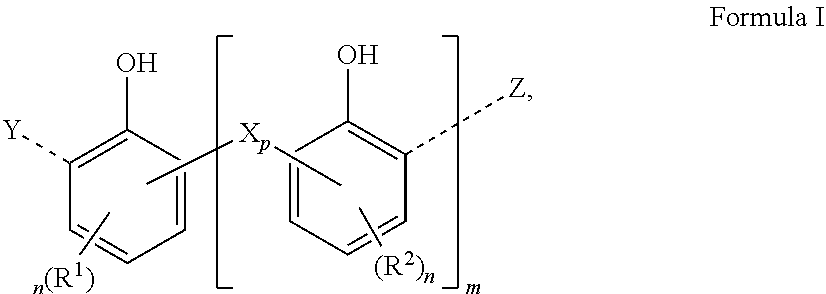
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.