Delivering And/or Receiving Material With Respect To A Subject Surface
Bernstein; Howard ; et al.
U.S. patent application number 16/705286 was filed with the patent office on 2020-11-12 for delivering and/or receiving material with respect to a subject surface. This patent application is currently assigned to Seventh Sense Biosystems, Inc.. The applicant listed for this patent is Seventh Sense Biosystems, Inc.. Invention is credited to Howard Bernstein, Donald E. Chickering, III, Shawn Davis, Ping Gong.
| Application Number | 20200353155 16/705286 |
| Document ID | / |
| Family ID | 1000004974642 |
| Filed Date | 2020-11-12 |












View All Diagrams
| United States Patent Application | 20200353155 |
| Kind Code | A1 |
| Bernstein; Howard ; et al. | November 12, 2020 |
DELIVERING AND/OR RECEIVING MATERIAL WITH RESPECT TO A SUBJECT SURFACE
Abstract
The present invention generally relates to receiving bodily fluid through a device opening. In one aspect, the device includes a flow activator arranged to cause fluid to be released from a subject. The flow activator may be actuated in a deployment direction by a deployment actuator, which may in turn cause fluid release from a subject. The flow activator may also be moved in a retraction direction by a retraction actuator. In one aspect, the device may include a vacuum source that may help facilitate fluid flow into the opening of the device and/or may help facilitate fluid flow from the opening to a storage chamber. In one aspect, an effector may enable fluid communication between the opening and the vacuum source and may do so in response to actuation of the flow activator.
| Inventors: | Bernstein; Howard; (Cambridge, MA) ; Chickering, III; Donald E.; (Framingham, MA) ; Gong; Ping; (Belmont, MA) ; Davis; Shawn; (Santa Monica, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Seventh Sense Biosystems,
Inc. Medford MA |
||||||||||
| Family ID: | 1000004974642 | ||||||||||
| Appl. No.: | 16/705286 | ||||||||||
| Filed: | December 6, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15349090 | Nov 11, 2016 | 10543310 | ||
| 16705286 | ||||
| 13718196 | Dec 18, 2012 | |||
| 15349090 | ||||
| 61577399 | Dec 19, 2011 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61M 2037/0023 20130101; A61B 5/150969 20130101; A61B 5/150984 20130101; A61B 5/150412 20130101; A61M 37/0015 20130101; A61M 1/38 20130101; A61B 5/150099 20130101; A61B 5/15188 20130101; A61B 5/150022 20130101; A61M 5/00 20130101; A61B 5/15113 20130101 |
| International Class: | A61M 5/00 20060101 A61M005/00; A61M 1/38 20060101 A61M001/38; A61B 5/15 20060101 A61B005/15; A61B 5/151 20060101 A61B005/151; A61M 37/00 20060101 A61M037/00 |
Claims
1-8. (canceled)
9. A material delivery/receiving device, including: a device actuator; a plurality of needles configured to deploy in response to actuation of the device actuator to form a plurality of openings in a subject's skin; a material source configured to provide a drug or other material to the subject through the plurality of openings in the subject's skin formed by the plurality of needles; and an adhesive configured to adhere the device to the subject's skin.
10. The device of claim 9, wherein the drug or other material of the material source is configured to move through the plurality of openings by diffusion.
11. The device of claim 9, wherein the adhesive is configured to adhere the device to the subject's skin for hours.
12. The device of claim 9, wherein the adhesive is configured to adhere the device to the subject's skin for days.
13. The device of claim 9, wherein the adhesive is configured to adhere the device to the subject's skin for months.
14. The device of claim 9, further comprising mechanical elements configured to mechanically hold the device to the subject's skin.
15. The device of claim 9, wherein the material source comprises a drug eluting hydrogel arranged to release a drug for delivery to the plurality of openings in the subject's skin.
16. The device of claim 9, wherein the material source comprises a reservoir including the drug or other material, and the device further comprises a positive pressure source configured to force the drug or other material from the reservoir to the subject's skin.
17. The device of claim 16, further comprising a device cover, wherein the positive pressure source comprises positive pressure stored in space under the device cover.
18. A method comprising: providing a material delivery/receiving device including a device actuator, a plurality of needles, and a material source; affixing the device to a subject's skin; and actuating the device actuator, causing the plurality of needles to deploy to form a plurality of openings in the subject's skin, wherein a drug or other material from the material source enters the subject's skin through the plurality of openings.
19. The method of claim 18, wherein the device is affixed to the subject's skin for hours.
20. The method of claim 18, wherein the device is affixed to the subject's skin for days.
21. The method of claim 18, wherein the device is affixed to the subject's skin for months.
22. The method of claim 18, wherein the device is affixed to the subject's skin via an adhesive.
23. The method of claim 18, wherein the device is affixed to the subject's skin via mechanical elements.
24. The method of claim 18, wherein the drug or other material from the material source enters the subject's skin through the plurality of openings by diffusion.
25. The method of claim 18, wherein actuating the device actuator exposes the material source to a positive pressure, which drives the drug or other material into the plurality of openings.
26. The method of claim 25, wherein actuating the device actuator causes a membrane to be pierced, which exposes the material source to the positive pressure.
27. The method of claim 18, further comprising receiving blood or other material from the plurality of openings in the subject's skin into the device.
28. The method of claim 27, wherein the blood or other material from the plurality of openings in the subject's skin is received into the device prior to the drug or other material from the material source entering the subject's skin.
Description
RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Patent Application Ser. No. 61/577,399, filed Dec. 19, 2011, entitled "Delivering and/or Receiving Material with Respect to a Subject Surface," by Bernstein, et al., which is incorporated herein by reference.
FIELD OF INVENTION
[0002] The present invention generally relates to systems and methods for delivering to and/or receiving fluids or other materials, such as blood or interstitial fluid, from subjects, e.g., to or from the skin and/or beneath the skin.
SUMMARY OF INVENTION
[0003] In some embodiments, the present invention generally relates to devices and methods for receiving fluids from a subject, such as the reception and separation of blood to form plasma or serum. The subject matter of the present invention involves, in some cases, interrelated products, alternative solutions to a particular problem, and/or a plurality of different uses of one or more systems and/or articles.
[0004] In one aspect of the invention, the device includes a flow activator arranged to cause fluid to be released from a subject. The flow activator may be moved in a deployment direction by a deployment actuator. The flow activator may also be moved in a retraction direction by a retraction actuator. In one aspect, the flow activator may be at a distance from the opening before deployment that is different from its distance from the opening after retraction.
[0005] In another aspect of the invention, an effector that includes only mechanical components moves the flow activator for deployment and retraction. Deployment movement may occur substantially faster than retraction movement.
[0006] In another aspect of the invention, the device may include a vacuum source that provides a pressure less than ambient pressure. The device may also include a channel that is fluidly coupled between the opening and the vacuum source. In one aspect of the invention, fluid communication between the opening and the vacuum source along the channel is enabled in response to actuation of the flow activator. In another aspect, fluid communication between the opening and the vacuum source is enabled in response to retraction of the flow activator. In another aspect, an effector actuates the flow activator and enables fluid communication between the opening and vacuum source.
[0007] In another aspect of the invention, the device includes a seal that is capable of closing fluid communication between the opening and the vacuum source through the channel. The seal and the flow activator may be attached together.
[0008] In another aspect of the invention, the effector may have an initial stored potential energy prior to any deployment movement of the flow activator. The effector may be arranged to release the stored potential energy to retract the flow activator.
[0009] In another aspect of the invention, flow activator, retraction actuator, and deployment actuator may be concentrically aligned with one another. Additionally, the device may include a spacer element that is also concentrically aligned with the flow activator, retraction actuator, and deployment actuator.
[0010] In another aspect, the present invention encompasses methods of making one or more of the embodiments described herein, for example, a device for receiving fluid. In still another aspect, the present invention encompasses methods of using one or more of the embodiments described herein, for example, a device for receiving fluid.
[0011] Other advantages and novel features of the present invention will become apparent from the following detailed description of various non-limiting embodiments of the invention when considered in conjunction with the accompanying figures. In cases where the present specification and a document incorporated by reference include conflicting and/or inconsistent disclosure, the present specification shall control. If two or more documents incorporated by reference include conflicting and/or inconsistent disclosure with respect to each other, then the document having the later effective date shall control.
BRIEF DESCRIPTION OF THE DRAWINGS
[0012] Non-limiting embodiments that incorporate one or more aspects of the invention will be described by way of example with reference to the accompanying figures, which are schematic and are not necessarily intended to be drawn to scale. In the figures, each identical or nearly identical component illustrated is typically represented by a single numeral. For purposes of clarity, not every component is labeled in every figure, nor is every component of each embodiment of the invention shown where illustration is not necessary to allow those of ordinary skill in the art to understand the invention. In the figures:
[0013] FIG. 1 is a perspective view of a material delivery/receiving device in accordance with aspects of the invention;
[0014] FIG. 2 is a perspective view of the of a material delivery/receiving device of FIG. 1;
[0015] FIG. 3 is a perspective view of the device in shown FIG. 1 with the cover removed;
[0016] FIG. 4 is a cross-sectional view of the device shown in FIG. 1;
[0017] FIG. 5 is an exploded view of the device shown in FIG. 1;
[0018] FIGS. 6A-6C show a series of three states of a flow activator of the device of FIG. 1;
[0019] FIG. 7A is an enlarged view of an effector including a retraction actuator and deployment actuator in a specific arrangement;
[0020] FIG. 7B is an underside view of the arrangement shown in FIG. 7A;
[0021] FIG. 8 is a close up view of a release element for the retraction actuator of the device in FIG. 1;
[0022] FIG. 9 is an enlarged view of a portion of a retraction actuator;
[0023] FIG. 10 is an enlarged view of a region of the device shown in FIG. 1 that illustrates a relationship between a storage vessel and a vacuum source;
[0024] FIG. 11 is a perspective view of a device in yet another embodiment of the invention, having separate retractor and seal actuator portions;
[0025] FIG. 12 is an enlarged view of the retractor and seal actuator in the device shown in FIG. 11;
[0026] FIG. 13 is an exploded view of the device shown in FIG. 11;
[0027] FIG. 14 is a cross-sectional view of the device shown in FIG. 11;
[0028] FIG. 15 is a perspective view of a device in yet another embodiment of the invention, having a rotatable release element;
[0029] FIG. 16 is an enlargement of a ramp engagement region in the device shown in FIG. 15;
[0030] FIG. 17 is an exploded view of the device shown in FIG. 15;
[0031] FIG. 18 is a cross-sectional view of the device shown in FIG. 15;
[0032] FIG. 19 is a perspective view of a device in yet another embodiment of the invention, having a sliding trigger tip;
[0033] FIG. 20 is a perspective view of the underside of the device shown in FIG. 19;
[0034] FIG. 21 is a perspective view of the device shown in FIG. 19 with the cover removed;
[0035] FIG. 22 is a perspective view of the device shown in FIG. 19 with the cover removed and at a different angle than the view shown in FIG. 21;
[0036] FIG. 23A is an enlargement of a trigger bridge from the device shown in FIG. 22;
[0037] FIG. 23B is an underside of the enlargement shown in FIG. 23A;
[0038] FIG. 24 is an exploded view of the device shown in FIG. 19;
[0039] FIG. 25 is a cross-sectional view of the device shown in FIG. 19;
[0040] FIGS. 26A-26D show alternative arrangements for connecting a flow activator to a deployment actuator;
[0041] FIG. 27 is a cross sectional view of a material delivery/receiving device in another embodiment; and
[0042] FIG. 28 shows perspective views of two configurations for the FIG. 27 embodiment.
DETAILED DESCRIPTION
[0043] Aspects of the invention are not limited in application to the details of construction and the arrangement of components set forth in the following description or illustrated in the drawings. For example, illustrative embodiments relating to piercing skin and receiving blood released from the pierced skin are discussed below, but aspects of the invention are not limited to use with devices that pierce skin and/or receive blood. Other embodiments may be employed, such as devices that receive other bodily fluids without piercing, devices that deliver drugs and/or other materials with or without piercing, and aspects of the inventions may be practiced or be carried out in various ways. Also, the phraseology and terminology used herein is for the purpose of description and should not be regarded as limiting.
[0044] FIG. 1 shows a material delivery/receiving device 1 that incorporates various aspects of the invention. Although FIG. 1 incorporates many of the aspects of the invention, any suitable number of aspects of the invention may be incorporated into a material delivery/receiving device. Thus, aspects of the invention may be used alone or in any suitable combination with each other. This illustrative embodiment includes a cover 20 and a base 100 that are joined together and may cooperate to enclose various parts of the device 1 and support one or more external features, such as a device actuator 10 that is used to cause the device 1 to receive fluid from a subject. The base 100 and the cover 20 may be formed from or otherwise include Polyester (PCTA or PETG) or other polymers with low gas permeability. Although the device actuator 10 in this embodiment is arranged to be actuated by a user (e.g., by the press of a finger), the device actuator 10 may be arranged in other ways, e.g., for actuation by a machine, an electrical signal, or other suitable arrangement to cause the material delivery/receiving device 1 to receive fluid from a subject. Actuation of the device actuator 10 may occur automatically, e.g., in response to an elapsed timer or other stimulus or condition, or manually. In some embodiments, the device actuator 10 may include a push-button as shown, a sliding button discussed more below, a touch-screen interface, a switch, or other user-actuatable arrangement, etc. In some cases, the device actuator 10 may allow for actuation of the device 1 only once, e.g., the device actuator 10 may become locked in a position that prevents further actuation, or may allow the device 1 to be actuated multiple times.
[0045] According to one aspect of the invention, the device 1 may include a fluid transporter that receives fluid from a subject and/or delivers fluid to a subject. The fluid transporter may include an applicator region where bodily fluids from the body may accumulate. In some embodiments, the applicator region may be a recess or an indentation within the base of the device, which can receive a fluid from the surface of the skin or deliver a fluid to the skin. The applicator region may have any suitable shape. For example, the applicator region can be generally hemispherical, semi-oval, rectangular, irregular, etc.
[0046] The fluid transporter may include an opening of any size and/or geometry that is constructed to receive fluid into the device. For example, the opening may lie in a two-dimensional plane or the opening may include a three-dimensional cavity, hole, groove, slit, etc. In some embodiments, the fluid transporter may also include a flow activator, such as one or more microneedles, arranged to cause fluid to be released from or delivered to the subject, e.g., by piercing the skin of a subject. In some embodiments, if fluid may partially or fully fill an enclosure surrounding a flow activator, then the enclosure can define at least part of a fluid transporter.
[0047] It should be noted that a flow activator need not be included with all embodiments as the device may not necessarily employ a mechanism for causing fluid release from/delivery to the subject. For instance, the device may receive fluid that has already been released due to another cause, such as a cut or an abrasion, fluid release due to a separate and independent device, such as a separate lancet, an open fluid access such as during a surgical operation, and so on. Additionally, fluid may be introduced into the device via urination, spitting, pouring fluid into the device, etc. If included, a flow activator may physically penetrate, pierce, and/or or abrade, chemically peel, corrode and/or irritate, release and/or produce electromagnetic, acoustic or other waves, other otherwise operate to cause fluid release from/material delivery to a subject. The flow activator may include a moveable mechanism, e.g., to move a needle, or may not require movement to function. For example, the flow activator may include a jet injector or a "hypospray" that delivers fluid under pressure to a subject, a pneumatic system that delivers and/or receives fluid, a hygroscopic agent that adsorbs or absorbs fluid, a reverse iontophoresis system, a transducer that emits ultrasonic waves, or thermal, radiofrequency and/or laser energy, and so on, any of which need not necessarily require movement of a flow activator to cause fluid release from a subject.
[0048] FIG. 2 shows an underside of the material delivery/receiving device 1 of FIG. 1 with a fluid transporter 120 that includes an opening 130, an applicator region 131, and a flow activator 90. In this embodiment, the flow activator 90 includes one or more needles. As described in more detail below, the needles may be extended from the opening 130 to pierce a subject's skin, and then retracted back into the opening to allow blood or other fluid to enter the opening 130. That is, to use the device 1 to receive blood from a subject, the base 100 may be placed on the skin so that the opening 130 is adjacent the skin. Thereafter, the device actuator 10 may be depressed to cause the needles to be deployed, piercing the skin and causing blood to be released. Blood may enter the opening and be collected in the storage chamber 140. In one embodiment, blood may flow into the storage chamber 140 as a result of a relatively low pressure (vacuum) in the device 1 that draws blood from the opening 130 and into the storage chamber 140 (see FIG. 4). In other embodiments discussed in more detail below, a drug or other material may be delivered to the skin (below and/or on the skin surface) by needles piercing the skin and carrying the material into the skin and/or functioning as a conduit to carry the material into the skin (e.g., by a relatively high pressure in the device 1 that forces the material from the storage chamber 140 and into the skin by way of the needles or openings in the skin formed by the needles).
[0049] The needles may be of any suitable width, length and/or other size, and the needles may each be solid or hollow. Hollow needles or needles that otherwise have a flow channel may be used to transport material, such as a liquid carrier and drug, into the skin. The needles may have any suitable cross-section (e.g., perpendicular to the direction of penetration), such as circular, square, oval, elliptical, rectangular, rounded rectangle, triangular, polygonal, hexagonal, irregular, etc. In some embodiments, the needles may have a length of about 5 mm or less. Additional information regarding alternative needle arrangements is provided below.
[0050] In this embodiment (FIG. 4), activation of the device actuator 10 causes the flow activator 90 to release blood or other fluid from a subject, which is then received at the opening 130. The blood or other fluid may then be collected in one or more chambers 140. Collection of the blood or other fluid may be done in any suitable way, such as by absorption, capillary action, suction, or other means. In this illustrative embodiment, activation of the device actuator 10 causes a seal 76 to open so that blood or other fluid may flow from the opening 130, through a channel (see FIG. 4, element 110) to a chamber 140. As is explained more below, the device 1 may include a vacuum source that draws the blood or other fluid from the opening 130 and into the chamber 140 upon opening of the seal 76. That is, opening of the seal 76 may introduce a relatively low pressure to the chamber 140, which causes blood or other fluid to be drawn from the opening 130 and into the chamber 140.
[0051] In one aspect of the invention, the flow activator may be actuated by a deployment actuator and a retraction actuator. For example, the flow activator may be moveable and movement of the flow activator may be caused by a deployment actuator and a retraction actuator. The deployment actuator may cause the flow activator to move in a deployment direction towards the skin and/or other surface of a subject, and the retraction actuator may cause the flow activator to move in a retraction direction away from the skin and/or body of a subject. As discussed in more detail below, providing separate actuators for deployment and retraction movement may provide advantages in some cases, such as enabling the flow activator to be moved at different speeds for deployment and retraction, allowing the actuators to perform other additional functions such as opening a fluid flow path for blood or other fluid, enabling the flow activator to start and finish at different positions in the device before deployment and after retraction, and others. The deployment actuator and the retraction actuator may each include any suitable components, such as a button, a switch, a lever, a slider, a dial, a compression spring, a Belleville spring, a servo, rotary or linear electric motor, and/or a pneumatic apparatus, or other suitable device. Also, the deployment actuator and the retraction actuator may be of the same type, or may be different types of devices. Each actuator may operate manually, mechanically, electrically, pneumatically, electromagnetically, or other suitable mode of operation, and may or may not require user input for activation.
[0052] In accordance with an aspect of the invention, an effector may be arranged to cause deployment and/or retraction movement of a flow activator. For example, an effector may include both a deployment actuator and a retraction actuator. The effector may be formed from or otherwise include polyester (PETG or PCTA), or acetal resin, acrylonitrile butadiene styrene (ABS), etc. FIGS. 3, 4, and 5 illustrate a perspective view of device 1 of FIG. 1 with the cover 20 removed from the base 100, a partial cross sectional view of the device 1, and an exploded view of the device 1, respectively. In this embodiment, the device 1 includes an effector 50 that includes a retraction actuator 40 and a deployment actuator 60 and that is movable in up and down directions relative to the base 100 along effector guides 104. The deployment actuator 60 is attached to the flow activator 90 via a membrane 72 (see FIG. 4) so that downward movement of the deployment actuator 60 may cause the flow activator 90 to at least partially extend from the opening 130. (As discussed more below, the membrane 72 may separate a vacuum source 156 in the device 1 from the opening 130 so that a relatively low pressure is maintained in the vacuum source 156 until controllably opened to cause flow into the storage chamber 140.) The vacuum source 156 may be in the form of a sealed vacuum chamber. In this embodiment, the deployment actuator 60 has a generally domed shape (e.g., as in a Belleville spring) with a central hole that receives a part of the membrane 72 which attaches the deployment actuator 60 to the flow activator 90. (Although in this embodiment the flow activator 90 is attached to the deployment actuator 60 via the membrane 72, the flow activator 90 may be directly connected to the deployment actuator 60, e.g., via a vertical post or other structure that extends from the flow activator 90 to the deployment actuator 60.) The deployment actuator 60 may initially be arranged in a concave-down configuration shown in FIG. 4 and moved to a concave-up configuration, e.g., by a user pressing the device actuator 10 to cause a release element 30 to push a center portion of the deployment actuator 60 downwardly. The deployment actuator 60 may be made of a suitable material and configuration to rapidly move from the concave-down to concave-up configurations so as to rapidly extend the flow activator 90 from the opening 130 and pierce a subject's skin or other surface. While the deployment actuator 60 in this embodiment is arranged as a flexible spring with a dome shape, the deployment actuator 60 may be of any suitable shape and/or size. For example, the deployment actuator 60 may be circular (having no "legs" unlike the four legs shown in FIG. 5), oblong, triangular (have 3 legs), square (4 legs with straight sides between each leg), pentagonal (5 legs), hexagonal (6 legs), spider-legged, star-like, clover-shaped (with any number of lobes, e.g., 2, 3, 4, 5, etc.), a serrated disc or a wave shape, or the like. The deployment actuator 60 may have, in some embodiments, a central hole as shown or another feature, such as a dimple, or button in the center or other location. The deployment actuator 60 may be formed from or otherwise include any suitable material, for example, a metal such as stainless steel (e.g., 301, 301LN, 304, 304L, 304LN, 304H, 305, 312, 321, 321H, 316, 316L, 316LN, 316Ti, 317L, 409, 410, 430, 440A, 440B, 440C, 440F, 904L), carbon steel, spring steel, spring brass, phosphor bronze, beryllium copper, titanium, titanium alloy steels, chrome vanadium, nickel alloy steels (e.g., Monel 400, Monel K 500, Inconel 600, Inconel 718, Inconel x 750, etc.), a polymer (e.g., polyvinylchloride, polypropylene, polycarbonate, etc.), a composite or a laminate (e.g., comprising fiberglass, carbon fiber, bamboo, Kevlar, etc.), or the like.
[0053] In some embodiments, all portions of the deployment actuator may move less than a certain distance when the deployment actuator moves in a deployment direction towards opening 130. In some embodiments, all portions of the deployment actuator may move less than about 10 mm, less than about 5 mm, less than about 3 mm, less than about 2 mm, or less than about 1 mm. The retraction actuator 40 in this embodiment includes a reversibly deformable structure in the form of a leaf spring, but, like the deployment actuator 60, other arrangements are possible such as a coil spring, foam, an elastic bladder, or the like. The retraction actuator may be formed from or otherwise include any suitable material, for example, 1095 spring steel or 301 stainless steel or other spring material such as 1074/1075, 5160, 9255 spring steel etc. The retraction actuator 40 is attached to the deployment actuator 60 via the effector body 50 so that when the retraction actuator 40 is released upon actuation of the device actuator 10, the retraction actuator 40 (and other portions of the effector 50) can move away from the opening 130 along the effector guides 104. This retraction motion draws the flow activator 90 and the deployment actuator 60 away from the opening as well. Specifically, and as shown at least in part in FIGS. 4 and 5, before actuation of the device 1, the retraction actuator 40 is in a compressed state, storing potential energy. That is, the center of the retraction actuator 40 is pressed downwardly during assembly so that four arms of the retraction actuator 40 are elastically deformed. The retraction actuator 40 is held in this depressed condition by ear portions 103 (see FIGS. 8 and 9) of the retraction actuator 40 engaging with the base 100 until the device 1 is actuated. However, when the device actuator 10 is pushed down during device actuation, arms 31 of the release element 30 engage with the tabs 41 to release the ear portions 103 from the base 100, allowing the center portion of the retraction actuator 40 to move in a retraction direction away from the opening 130. Since the deployment actuator 60 and flow activator 90 are attached to the retraction actuator 40, movement of the retraction actuator 40 upward away from the opening 130 retracts the flow activator 90 from the opening 130. Additionally, movement of the retraction actuator 40 upward away from the opening 130 may also move the deployment actuator 60 in a retraction direction away from the opening 130 as well. In some embodiments, all portions of the deployment actuator 60 may move less than a certain distance when the deployment actuator 60 moves in a retraction direction away from the opening 130. In some embodiments, all portions of the deployment actuator may move less than about 10 mm, less than about 5 mm, less than about 3 mm, less than about 2 mm, or less than about 1 mm.
[0054] In some embodiments, as shown in FIG. 4, a spacer element 32 is located between the deployment actuator 60 and the retraction actuator 40. The spacer element 32 may help to eliminate a gap between the deployment actuator 60 and the release element 30. Actuation of device actuator 10 may cause the release element 30 to push down on the spacer element 32, which may in turn push on the deployment actuator 60 and cause the deployment actuator 60 to move the flow activator 90 in a deployment direction. In some embodiments, the flow activator 90, deployment actuator 60, retraction actuator 40, and spacer element 32 are substantially concentrically aligned.
[0055] By providing both a deployment actuator 60 and a retraction actuator 40 for the flow activator 90, the flow activator 90 may be controlled to have any suitable movement for both deployment and retraction. For example, the flow activator 90 may be caused to move more rapidly in the deployment direction than in the retraction direction, which has been found to potentially reduce pain when piercing skin to release blood and/or deliver material to the skin. That is, the deployment actuator 60 may be arranged to relatively rapidly move from the concave-down to concave-up configuration, quickly inserting the flow activator 90 into skin or another surface. Thereafter, the flow activator 90 may be more slowly withdrawn from the skin by the retraction actuator 40, e.g., as controlled by a relatively lower force exerted by the retraction actuator 40 on the flow activator 90 than the deployment actuator 60, by damped motion of the retraction actuator 40, or other suitable arrangements. In other embodiments, having separate deployment and retraction actuators may allow for a shorter range of motion in one direction, such as in the deployment direction, than in another direction, such as the retraction direction. For example, by having the flow activator 90 move a relatively short distance for deployment, the deployment actuator 60 may be made relatively compact, yet generate suitably high force to insert the flow activator 90 into skin. In contrast, a relatively longer distance traveled by the flow activator 90 during retraction may withdraw the activator 90 suitably to allow a pool or other collection of blood to enter a cavity or other space for reception by the device 1. Additionally, a short deployment distance may minimize alignment errors inherent in long travel distances.
[0056] Accordingly, in one aspect of the invention, the flow activator may be located at an initial pre-deployment distance from skin or another surface that is different from a final post-retraction distance between the flow activator and the skin or other surface. While this aspect can be provided in many different ways, such as by a motor, servo, or automated device as part of an effector, the effector 50 of the FIGS. 1-5 embodiment may provide an arrangement in which flow activator 90 is relatively close to the opening 130 prior to deployment, and is located relatively further away from the opening 130 after retraction. FIGS. 6A-6C show a series of schematic representations of three states of the device 1 of FIGS. 1-5, including an initial state before deployment of the flow activator 90, an intermediate state where the flow activator is extended from the opening 130 or otherwise positioned to cause release of fluid from a target skin or other surface, and a final state where the flow activator 90 is retracted, respectively.
[0057] As can be seen in FIG. 6A, a pre-deployment distance 181 between the opening 130 and the flow activator 90 is relatively small, such as 1 mm or less. In this state, the retraction actuator 40 is compressed, and the deployment actuator 60 is in a concave-down arrangement. As shown in FIG. 6B, the deployment actuator 60 is inverted to a concave-up configuration so that the flow activator 90 is deployed. The retraction actuator 40 may also be further compressed, e.g., by the user pressing down on the release element 30, but in other embodiments, the retraction actuator 40 need not be further compressed or otherwise deformed. As shown in FIG. 6C, a post-retraction distance 183 between the opening 130 and the flow activator 90 may be larger, in some cases significantly larger, than the pre-deployment distance 181. For example, the post-retraction distance 183 in which the flow activator 90 is fully retracted from the opening 130 may be 2-3 mm or more. Retraction of the flow activator 90 from the opening 130 may provide a space into which blood or other fluid released from the subject may collect and/or otherwise be received by the device 1. However, other arrangements are possible in which the post-retraction distance is less than, or the same as, the pre-deployment distance, and all aspects of the invention are not necessarily limited in this regard.
[0058] FIGS. 7A and 7B show top perspective and bottom perspective views of the effector 50 of the FIGS. 1-5 embodiment, and help to better illustrate how the motion of the effector 50 is controlled. As shown in FIG. 7A, the retraction actuator 40 has eight legs radiating from a central body having a central hole. Two of the shorter legs attach the retraction actuator 40 to the effector body 50 via two posts 52 that extend through holes 46 of the retraction actuator 40. The diameter of the post heads 52 may be made larger than the holes 46 and thus fix the retraction actuator 40 to the effector body 50. The retraction actuator 40 may alternately be attached to the effector body by 50 by adhesive (e.g. tape, liquid), mechanical fastening (e.g. interference fit, slot/groove, screws) or thermal methods (e.g. heat staking), and is not limited in this regard. Other legs 48 of the retraction actuator 40 may remain free to flex relative to the effector body 50, e.g., to provide the retraction movement of the effector 50. Two of the legs 48 include ear portions 103 which serve to engage with the base 100 and hold the retraction actuator 40 in a compressed, initial position before deployment of the flow activator 90. A space or gap 43 is provided between the ear portions 103 and the effector body 50 to allow the ear portions 103 to move toward the body for engagement with the base 100. As described above and shown in FIG. 7B, the deployment actuator 60 includes a central hole 66 and lobes 62 that are held within the grooves 56 of the effector body 50. Although the deployment actuator 60 is attached to the effector body 50, a central portion 64 of the deployment actuator 60 remains displaceable relative to the effector body 50 so that the deployment actuator 60 may move to deploy the flow activator 90.
[0059] As discussed above, the effector 50 may be mounted to the base 100 and guided in motion via effector guides 104 that protrude from the base 100. FIG. 8 shows a close up view of the retraction actuator 40 illustrating how the retraction actuator 40 engages with the base 100 in a compressed, initial state, while FIG. 9 shows a close up view of the ear portions 103 on two of the legs 48 of the retraction actuator 40 that engage with the base 100 to hold the retraction actuator 40 in the compressed, initial state. With the effector 50 held suitably by the effector guides 104, the effector 50 is pressed downwardly so that ear portions 103 of the tabs 41 can be positioned under corresponding protrusions 101 on the base 100. With the ear portions 103 engaged with the protrusions 101, the effector 50 may be released so that the spring force of the legs 48 biases the effector 50 to move upwardly in the retraction direction. However, with the ear portions 103 engaged with the protrusions 101, the effector 50 is held in a compressed condition. In this pre-deployment arrangement, the flow activator 90 may be at the initial pre-deployment distance 181 (see FIG. 6) from the opening 130. In some embodiments, this pre-deployment distance 181 may be arranged such that actuation of the deployment actuator 60 will cause the flow activator 90 to reach the skin of a subject and allow the flow activator 90 to penetrate and/or pierce the skin to cause fluid flow. Thus, having the retraction actuator 40 pre-loaded in an initial semi-compressed state may hold the flow activator 90 at a pre-deployment distance 181 that enables the flow activator 90 to be ready for deployment upon actuation of the device actuator 10.
[0060] FIG. 8 also illustrates how the retraction actuator 40 may be released to retract the flow activator 90. Arms 31 of the release element 30 may engage with the tabs 41 so that sloped portions of the arms 31 push the tabs 41 outwardly and away from the effector body 50 when the device actuator 10 and the release element 30 are moved downwardly. This releases the ear portions 103 from the protrusions 101, allowing the effector 50 to move upwardly under the bias of the deformed legs of the retraction actuator 40. The release element 30 may be formed from or otherwise include polyester (PETG or PCTA), or acetal resin, acrylonitrile butadiene styrene (ABS), etc. While in this embodiment the retraction actuator 40 is shown to engage with the base 100 via a releasable latch arrangement that includes the ear portions 103 and the protrusions 101, other arrangements are possible, such as a releasable lever, a sliding release, a detent, magnets that are separable using a wedge or by flipping polarity, etc., as the invention is not limited in this regard.
[0061] In another aspect of the invention, the effector may have an initial stored potential energy prior to any deployment movement of the flow activator. That is, the effector may have stored spring energy or other mechanical energy stored, for example, in an elastically deformed element, stored chemical energy, stored electrical energy, etc., that is used to deploy and/or retract a flow activator or cause other motion of other parts of the fluid receiving device. As explained above, before deployment of the flow activator 90, the retraction actuator 40 may be held in a compressed state by engagement of the ear portions 103 of the legs 48 with protrusion elements 101 on the base 100. Compression of the retraction actuator 40 stores potential energy in the retraction actuator 40 that can be used for different actions, such as retracting the flow activator 90. Thus, having the retraction actuator 40 at an initial compressed state permits the retraction actuator 40 to store potential energy and be ready for actuation without requiring energy to be input to the system at the time of actuation of the device.
[0062] In another aspect of the invention, the flow activator may move faster in a deployment direction than in a retraction direction. In the embodiments discussed above, the deployment actuator 60 may be arranged to move from an initial, pre-deployment position to a deployment position in rapid fashion, e.g., in a bi-stable manner. In contrast, the retraction actuator 40 may be arranged, e.g., to have a relatively lower spring constant or other characteristic, to move the flow activator 90 at a slower rate during at least a part of the retraction motion. In one set of embodiments, the flow activator 90 can be deployed at a speed of at least about 0.1 cm/s, at least about 0.3 cm/s, about 1 cm/s, at least about 3 cm/s, at least about 10 cm/s, at least about 30 cm/s, at least about 1 m/s, at least about 2 m/s, at least about 3 m/s, at least about 4 m/s, at least about 5 m/s, at least about 6 m/s, at least about 7 m/s, at least about 8 m/s, at least about 9 m/s, at least about 10 m/s, at least about 12 m/s, etc., at the point where the flow activator 90 initially contacts the skin. Without wishing to be bound by any theory, it is believed that relatively faster deployment speeds may increase the ability of the flow activator to penetrate the skin (without deforming the skin or causing the skin to move in response), and/or decrease the amount of pain felt by the application of the flow activator to the skin. Any suitable method of controlling the penetration speed into the skin may be used, including those described herein.
[0063] Retraction of the flow activator 90 may occur at a slower speed than deployment, e.g., to help reduce any pain associated with withdrawal of the flow activator 90. Where the retraction actuator 40 includes only mechanical elements that are not electronically controlled, e.g., as in the case of a spring, an elastic member, collapsible foam, etc., the spring or other element may be designed or otherwise arranged to provide a desired retraction speed. Alternately, other mechanical elements, such as one or more dampers may be provided to control a withdrawal speed. Other, electronically controlled systems, such as some servos, pneumatic systems, or the like, may incorporate open or closed loop control to provide a desired retraction rate. In the case of a manually-operated retraction actuator, the user may be able to control the speed of retraction. For example, a retraction actuator in the form of a spring may retract more slowly if force is gradually eased off the device actuator. However, if the force is abruptly removed, (e.g. a user suddenly releases the device actuator), the retraction may occur more quickly, although the fastest possible retraction speed may still be slower than the deployment speed.
[0064] In some aspects, the fluid receiving device may contain one or more chambers or vessels 140 for holding fluid received from a subject. In some cases, the chambers may be in fluidic communication with one or more fluid transporters and/or one or more microfluidic channels. For instance, the fluid receiving device may include a chamber for collecting fluid withdrawn from a subject (e.g., for storage and/or later analysis), a chamber for containing a fluid for delivery to the subject (e.g., blood, saline, optionally containing drugs, hormones, vitamins, pharmaceutical agents, or the like), etc.
[0065] In one aspect of the invention, the device may include a vacuum source. Vacuum (a pressure below ambient) may help facilitate fluid flow into the opening 130 of the device, and/or may help draw skin into the opening 130 for contact with the flow activator 90, and/or may help facilitate fluid flow from the opening 130 to a chamber 140. In some cases, the vacuum source may be one that is self-contained within the device, i.e., the device need not be connected to an external vacuum source (e.g., a house vacuum) during use of the device to withdraw blood or interstitial fluid from the skin and/or from beneath the skin. For example, as shown in FIG. 4, in one set of embodiments, the vacuum source may include a vacuum source 156 having a pressure less than ambient pressure before blood (or other fluid) is withdrawn into the device, i.e., the vacuum source 156 may be at a "negative pressure" (that is, negative relative to ambient pressure) or a "vacuum pressure" (or just having a "vacuum"). For example, if ambient pressure is at atmospheric pressure, the vacuum in the vacuum source may be at least about 50 mmHg, at least about 100 mmHg, at least about 150 mmHg, at least about 200 mmHg, at least about 250 mmHg, at least about 300 mmHg, at least about 350 mmHg, at least about 400 mmHg, at least about 450 mmHg, at least about 500 mmHg, at least 550 mmHg, at least 600 mmHg, at least 650 mmHg, at least about 700 mmHg, or at least about 750 mmHg, i.e., below the ambient atmospheric pressure. However, in other embodiments, it should be understood that other pressures may be used and/or that different methods may be used to produce other pressures (greater than or less than atmospheric pressure). As non-limiting examples, an external vacuum or a mechanical device may be used as the vacuum source. For example, the device may comprise an internal vacuum source, and/or be connectable to a vacuum source that is external to the device, such as a vacuum pump or an external (line) vacuum source. In some cases, vacuum may be created manually, e.g., by manipulating a syringe pump, a plunger, or the like, or the low pressure may be created mechanically or automatically, e.g., using a piston pump, a syringe, a bulb, a Venturi tube, manual (mouth) suction, etc., or the like.
[0066] Thus, in some cases, the device may be "pre-packaged" with a suitable vacuum source (e.g., a pre-evacuated vacuum source 156); for instance, in one embodiment, the device may be applied to the skin and activated in some fashion to create and/or access the vacuum source. In some embodiments, the self-contained vacuum source may be actuated in some fashion to create a vacuum within the device. For instance, the self-contained vacuum source may include a piston, a syringe, a mechanical device such as a vacuum pump able to create a vacuum within the device, and/or chemicals or other reactants that can react to increase or decrease pressure which, with the assistance of mechanical or other means driven by the reaction, can form a pressure differential associated with a pressure regulator. Chemical reaction can also drive mechanical actuation with or without a change in pressure based on the chemical reaction itself. A self-contained vacuum source can also include an expandable foam, a shape memory material, or the like.
[0067] In some cases, the device includes an interface 105 (see FIGS. 2, 4 and 5) that is able to help the device apply a vacuum to the skin and/or at the opening 130. The interface 105 may be, for example, a suction cup, a layer of a hydrogel material, such as Katecho 10G or other suitable hydrogel, or a circular bowl that is placed on the surface of the skin, and vacuum may be applied to the portion of skin exposed to the device 1 by the interface 105. In one set of embodiments, the interface is part of a support structure, e.g., the base 100. The interface 105 may be formed from any suitable material, e.g., glass, rubber, polymers such as silicone, polyurethane, nitrile rubber, EPDM rubber, neoprene, or the like. In some cases, the seal between the interface 105 and the skin may be enhanced (e.g., reducing leakage), for instance, using vacuum grease, petroleum jelly, a gel, an adhesive or the like. In some cases, the interface 105 may be relatively small, for example, having a diameter of less than about 5 cm, less than about 4 cm, less than about 3 cm, less than about 2 cm, less than about 1 cm, less than about 5 mm, less than about 4 mm, less than about 3 mm, less than about 2 mm, or less than about 1 mm. The interface 105 may be circular, although other shapes are also possible, for example, square, star-shaped (having 5, 6, 7, 8, 9, 10, 11, etc. points), tear-drop, oval, rectangular, or the like. In some embodiments, a portion of the interface 5 may extend across the opening 130, or at least a portion of the opening 130, and be arranged so that the flow activator 90 (e.g., one or more needles) passes through the interface portion 105 before entering the skin. In this way, the flow activator 90 may not only open the interface 105 to expose the opening 130 to the skin, but the interface portion 105 through which the flow activator passes may carry a drug or other material to be delivered to the skin. For example, the interface portion penetrated by the flow activator 90 may include a drug-loaded matrix, such as a drug eluting hydrogel, arranged so that as needles pass through the interface portion 105, the needles pick up material from the interface portion and carry the material into the skin. In other arrangements, the flow activator 90 may remain engaged with the skin after deployment through the interface portion and into the skin. In this way, the needles or other parts of the flow activator 90 may function as conduits or otherwise facilitate passage of the material into the skin, whether through channels or other flowpaths in or on the needles, dispersion through the needle material itself (e.g., in the case of a porous needle material), and so on. The needles may also be made dissolvable or otherwise degradable so that the needles help to maintain a flow pathway for drug or other material into the skin. Further, resorption of the needles may itself deliver drug or other material as is known in the art.
[0068] In some embodiments, vacuum from a vacuum source may facilitate the movement of blood or other fluids from an opening of a fluid transporter to a storage vessel. Alternately, pressure in a pressure source may help facilitate movement of drug or other material from a storage vessel to an opening of a fluid transporter and delivery to skin or other subject portion. In the FIGS. 1-5 embodiment, vacuum may be stored in a vacuum source 156, e.g., a majority of space enclosed by the cover 20 and the base 100. Vacuum in the vacuum source 156 may be selectively coupled to the storage chamber 140 so as to cause fluid at the opening 130 to be drawn into a channel 110 and to the chamber 140. For example, and as can be seen in FIG. 5, one or more channels 110 may be formed into the base 100 or otherwise provided between the opening 130 and the storage chamber 140. The channel 110 may be covered at an upper side by a lower surface of a channel plate 80. In some embodiments, the channel plate 80, membrane 72 and seal 76 could form a single part. (Additional configuration options for the channel 110 are discussed below.) The channel plate 80 may not only help to define the channel 110, but also define at least a portion of the cavity at the fluid transporter 120, part of the storage chamber 140, a vacuum inlet 154 and flow path 150 used for control of flow between the vacuum source 156 and the storage chamber 140, and a flow path between the channel 110 and the storage chamber 140. That is, as shown in FIGS. 4 and 10, the channel plate 80 helps to define a flow path between the opening 130 and the vacuum source 156 such that flow from the opening 130 may pass through the channel 110 and to an opening 144 in the channel plate 80 that connects the channel 110 and the storage chamber 140. The opening 144 may include a filter, a hydrophobic element (e.g., to help prevent aqueous fluid in the storage chamber 140 from later exiting the chamber 140), a one-way valve, or may be completely unobstructed. As can be seen in FIG. 10, flow may also occur from the storage chamber 140 through a passage 150 in the channel plate 80 to the vacuum inlet 154. The vacuum inlet 154 is normally closed by a seal 76, which may be part of the membrane 72, which also helps to isolate the vacuum source 156 from the opening 130 and other potential outlets for the low pressure in the vacuum source 156. As can be seen in FIG. 4, the seal 76 is engaged with one of the legs 48 of the retraction actuator 40 (a seal leg 49) so that when the retraction actuator 40 is in a compressed, initial state, the seal leg 49 presses the seal 76 into contact with the vacuum inlet 154 so as to close the passage 150 and prevent communication between the vacuum source 156 and the storage chamber 140. However, once the retraction actuator 40 is released, the seal leg 49 may move upwardly and/or the force of the seal leg 49 on the seal 76 may be reduced to a point at which the vacuum inlet 154 is open for flow from the storage chamber 140 to the vacuum source 156. Thus, once the seal 76 opens the vacuum inlet 154, the vacuum source 156 may draw fluid (e.g., air and/or liquid) from the storage chamber 140 so that fluid in the channel 110 is drawn into the storage chamber 140. Although not shown, a hydrophobic membrane or other suitable element may be provided at the vacuum inlet 154 or other suitable location (such as in the passage 150) to prevent liquid from flowing from the storage chamber 140 into the vacuum source 156. As will be appreciated, if the vacuum source 156 is actually a positive pressure source, opening of the seal 76 may cause the delivery of material in the storage chamber 140 to the flow activator 90, e.g., to effectively cause injection of the material into skin through needles of the flow activator 90.
[0069] In accordance with one aspect of the invention, fluid communication between the fluid transporter opening and the vacuum source may be enabled in response to actuation of the flow activator. For example, depression of the device actuator 10 may permit communication between the vacuum source 156 and the storage chamber 140/opening 130. While other arrangements are possible, in the illustrative embodiment of FIGS. 1-10, the seal 76 may be coupled to the seal leg 49 of the retraction actuator 40 so that once the flow activator 90 is actuated, e.g., deployment and retraction are initiated, the seal 76 may be released from the vacuum inlet 154 to permit fluid communication between the vacuum source 156 and the storage chamber 140. This may allow a vacuum to be exerted on skin at the opening 130, causing the skin to be drawn toward and/or into the opening 130 prior to the flow activator 90 interaction with the skin. In some cases, it has been found that drawing skin into the opening 130 before needles of a flow activator 90 penetrate the skin has aided in the withdrawal of blood from the skin, whether in terms of speed and/or volume of blood drawn. Although in this embodiment, the seal leg 49 of the retraction actuator 40 moves away from the vacuum inlet 154 (or at least reduces a pressure on the seal 76) as the flow activator 90 is retracted, it is possible to arrange the opening of the seal 76 upon deployment of the flow activator 90 or at any other point in the movement of the flow activator 90, as well as before movement begins or after movement is completed. For example, flow between the vacuum source 156 and the storage chamber 140 may be enabled by piercing a membrane or foil, e.g., with deployment of the flow activator 90 or upon full retraction of the flow activator 90. In one embodiment, a membrane seal could be located at the opening 130, and the flow activator 90 itself could serve to puncture the membrane, allowing flow from the opening 130 to the vacuum source 156. Thus, this puncture could serve to expose fluid at the opening 130 to vacuum to draw the fluid into a storage chamber 140. Of course, a membrane seal may be positioned at locations other than the opening 130, such as at the vacuum inlet 154, and a separate piercing element, such as a spike on the release element 30, could be used to puncture the membrane. Other arrangements are possible as well, such as actuating a vacuum source (such as a chemical vacuum source or vacuum pump) in response to flow activator actuation. For example, the retraction actuator 40 may be coupled to a syringe piston so that as the retraction actuator 40 moves in the retraction direction, the piston is moved to generate suction at the storage chamber 140.
[0070] As will be appreciated from the description above, in another aspect of the invention, the flow activator may be moved in a deployment direction to deploy the flow activator, and moved in a retraction direction to both retract the flow activator and enable fluid communication between the vacuum source and a fluid transporter opening. In the illustrative embodiment described above, the seal 76 may be released from the vacuum inlet 154 as the flow activator 90 is retracted. Opening of the flow path at the seal 76 may occur at the start of retraction, during retraction, and/or after retraction is complete. In some embodiments, the seal 76 and flow activator 90 may be both moved in the same retraction direction by the retraction actuator. That is, during retraction, the flow activator 90 may be retracted and the seal 76 lifted to enable fluid communication between the vacuum source 156 and the device opening 130 through a channel 110. The seal 76 may be formed from or otherwise include latex or other flexible material such as a thermoplastic elastomer (TPE) or polyurethane. In other embodiments, a force on the seal 76 may be sufficiently released to allow the relatively low pressure in the vacuum source 156 to cause flow from the storage chamber 140 to the vacuum source 156 to occur. Thus, the seal 76 need not necessarily be lifted from the vacuum inlet 154, but instead may act as a kind of check valve with a desired crack pressure that permits flow from the storage chamber 140 to the vacuum source 156 while a suitable pressure differential is present across the seal 76, but otherwise inhibits flow through the inlet 154. Other arrangements for opening fluid communication during retraction of the flow activator are possible, such as a spike on the retraction actuator 40 that pierces a membrane to open the fluid communication. In another embodiment, an electrical switch may be opened or closed by the retraction actuator, causing a vacuum source (such as a pump) to be activated. In another embodiment, movement of the retraction actuator may release a latch or other device, which allows a spring-loaded syringe piston or other device to move, creating a desired vacuum. In another embodiment, retraction movement of the retraction actuator 40 itself may move a syringe piston or other device to provide a desired vacuum. Thus, enabling of fluid communication between a vacuum source and a fluid transporter opening need not necessarily involve the opening of a valve or other device that blocks flow, but instead may involve the creation of suitable vacuum to cause flow. Other arrangements are possible as well.
[0071] In another aspect of the invention, an effector that deploys and/or retracts the flow activator may also enable fluid communication between the fluid transporter opening and the vacuum source. Providing a single component or assembly to both deploy and/or retract a flow activator as well as open fluid communication between a fluid transporter and vacuum source may, in some embodiments, provide for a fluid receiving device that is simpler in operation or construction. For example, a single device, such as a retraction actuator 40 in the FIGS. 1-10 embodiment, may serve to both retract and open a flow path. This may reduce parts needed for construction of the fluid receiving device, reducing cost and/or assembly complexity. Of course, the effector need not necessarily perform both deployment and retraction functions, but instead may provide only deployment or retraction together with enabling fluid communication. For example, the effector may serve to only deploy a flow activator and enable fluid communication between the fluid transporter opening and vacuum source, e.g., in an embodiment where a flow activator is not retracted after deployment, but instead is permitted to remain embedded in skin to withdraw fluid as vacuum is applied to the flow activator. As discussed above, enabling of fluid communication between the fluid transporter opening and vacuum (or positive pressure) source may be provided in different ways, such as by opening a valve or similar structure (such as the seal 76), piercing a membrane, actuating a vacuum source (such as moving a syringe plunger or similar element), activating a chemically-operated vacuum source, and so on.
[0072] In another aspect of the invention, the flow activator and the vacuum seal may be attached together, e.g., as part of a single unitary structure or component. For example, as shown in FIGS. 4 and 5, the flow activator 90 may be attached to the membrane 72, e.g., by co-molding the flow activator 90 with the membrane, adhering the flow activator 90 to the membrane, etc., while the seal 76 is formed from part of the membrane 72 itself. Such an arrangement may ease assembly and reduce the number of components in the fluid receiving device 1.
[0073] As discussed above, flow enabled by movement of the seal 76 may cause flow along the channel 110 to the storage chamber 140. The channel 110 may be formed, at least in part, by a single component, e.g. an etched substrate or molded unit such as the base 100. The channel can have any cross-sectional shape, for example, circular, oval, triangular, irregular, square or rectangular (having any aspect ratio), or the like, and can be covered or uncovered (i.e., open to the external environment surrounding the channel). The channel 110 may be of any length. In some cases, the channel 110 can be a simple two-dimensional opening that creates a fluidic coupling between the opening 130 and another vessel such as a vacuum source or a storage vessel. In these cases, the channel may not have any length at all (e.g., as in a two-dimensional opening). In embodiments where the channel is completely covered, at least one portion of the channel can have a cross-section that is completely enclosed, and/or the entire channel may be completely enclosed along its entire length with the exception of its inlet and outlet.
[0074] A channel may have any aspect ratio (length to average cross-sectional dimension), e.g., an aspect ratio of at least about 2:1, more typically at least about 3:1, at least about 5:1, at least about 10:1, etc. As used herein, a "cross-sectional dimension," in reference to a fluidic or microfluidic channel, is measured in a direction generally perpendicular to fluid flow within the channel. A channel generally will include characteristics that facilitate control over fluid transport, e.g., structural characteristics and/or physical or chemical characteristics (hydrophobicity vs. hydrophilicity) and/or other characteristics that can exert a force (e.g., a containing force) on a fluid. The fluid within the channel may partially or completely fill the channel. In some cases the fluid may be held or confined within the channel or a portion of the channel in some fashion, for example, using surface tension (e.g., such that the fluid is held within the channel within a meniscus, such as a concave or convex meniscus). In an article or substrate, some (or all) of the channels may be of a particular size or less, for example, having a largest dimension perpendicular to fluid flow of less than about 5 mm, less than about 2 mm, less than about 1 mm, less than about 500 microns, less than about 200 microns, less than about 100 microns, less than about 60 microns, less than about 50 microns, less than about 40 microns, less than about 30 microns, less than about 25 microns, less than about 10 microns, less than about 3 microns, less than about 1 micron, less than about 300 nm, less than about 100 nm, less than about 30 nm, or less than about 10 nm or less in some cases. In one embodiment, the channel is a capillary.
[0075] In one set of embodiments, the device may include a microfluidic channel. As used herein, "microfluidic," "microscopic," "microscale," the "micro-" prefix (for example, as in "microchannel"), and the like generally refers to elements or articles having widths or diameters of less than about 1 mm, and less than about 100 microns (micrometers) in some cases. In some embodiments, larger channels may be used instead of, or in conjunction with, microfluidic channels for any of the embodiments discussed herein. For examples, channels having widths or diameters of less than about 10 mm, less than about 9 mm, less than about 8 mm, less than about 7 mm, less than about 6 mm, less than about 5 mm, less than about 4 mm, less than about 3 mm, or less than about 2 mm may be used in certain instances. In some cases, the element or article includes a channel through which a fluid can flow. In all embodiments, specified widths can be a smallest width (i.e. a width as specified where, at that location, the article can have a larger width in a different dimension), or a largest width (i.e. where, at that location, the article has a width that is no wider than as specified, but can have a length that is greater). Thus, for instance, the microfluidic channel may have an average cross-sectional dimension (e.g., perpendicular to the direction of flow of fluid in the microfluidic channel) of less than about 1 mm, less than about 500 microns, less than about 300 microns, or less than about 100 microns. In some cases, the microfluidic channel may have an average diameter of less than about 60 microns, less than about 50 microns, less than about 40 microns, less than about 30 microns, less than about 25 microns, less than about 10 microns, less than about 5 microns, less than about 3 microns, or less than about 1 micron.
[0076] Fluids received from the skin and/or from beneath the skin of the subject will often contain various analytes within the body that are important for diagnostic purposes, for example, markers for various disease states, such as glucose (e.g., for diabetics); other example analytes include ions such as sodium, potassium, chloride, calcium, magnesium, and/or bicarbonate (e.g., to determine dehydration); gases such as carbon dioxide or oxygen; H.sup.+ (i.e., pH); metabolites such as urea, blood urea nitrogen or creatinine; hormones such as estradiol, estrone, progesterone, progestin, testosterone, androstenedione, etc. (e.g., to determine pregnancy, illicit drug use, or the like); or cholesterol. Other examples include insulin, or hormone levels. Still other analytes include, but not limited to, high-density lipoprotein ("HDL"), low-density lipoprotein ("LDL"), albumin, alanine transaminase ("ALT"), aspartate transaminase ("AST"), alkaline phosphatase ("ALP"), bilirubin, lactate dehydrogenase, etc. (e.g., for liver function tests); luteinizing hormone or beta-human chorionic gonadotrophin (hCG) (e.g., for fertility tests); prothrombin (e.g., for coagulation tests); troponin, BNT or B-type natriuretic peptide, etc., (e.g., as cardiac markers); infectious disease markers for the flu, respiratory syncytial virus or RSV, etc.; or the like.
[0077] The fluid receiving device 1 may include one or more sensors for detecting one more characteristics of a fluid received from a subject. The sensor(s) may be located in any suitable way or location with respect to the device, such as at the storage chamber 140, at the channel 110, on the cover 20, etc. For example, the device 1 may include a pH sensor, an optical sensor, an oxygen sensor, a sensor able to detect the concentration of a substance, or the like. Non-limiting examples of sensors useful in the invention include dye-based detection systems, affinity-based detection systems, microfabricated gravimetric analyzers, CCD cameras, optical detectors, optical microscopy systems, electrical systems, thermocouples and thermistors, pressure sensors, etc. Those of ordinary skill in the art will be able to identify other suitable sensors. The sensor can include a colorimetric detection system in some cases, which may be external to the device, or microfabricated into the device in certain cases. As an example of a colorimetric detection system, if a dye or a fluorescent entity is used (e.g. in a particle), the colorimetric detection system may be able to detect a change or shift in the frequency and/or intensity of the dye or fluorescent entity.
[0078] In one set of embodiments, the sensor may be a test strip, for example, test strips that can be obtained commercially. Examples of test strips include, but are not limited to, glucose test strips, urine test strips, pregnancy test strips, or the like. A test strip will typically include a band, piece, or strip of paper or other material and contain one or more regions able to determine an analyte, e.g., via binding of the analyte to a diagnostic agent or a reaction entity able to interact with and/or associate with the analyte. For example, the test strip may include various enzymes or antibodies, glucose oxidase and/or ferricyanide, or the like. The test strip may be able to determine, for example, glucose, cholesterol, creatinine, ketones, blood, protein, nitrite, pH, urobilinogen, bilirubin, leucocytes, luteinizing hormone, etc., depending on the type of test strip. The test strip may be used in any number of different ways. In some cases, a test strip may be obtained commercially and inserted into the device, e.g., before or after receiving blood, interstitial fluid, or other fluids from a subject. At least a portion of the blood or other fluid may be exposed to the test strip to determine an analyte, e.g., in embodiments where the device uses the test strip as a sensor so that the device itself determines the analyte. In some cases, the device may be sold with a test strip pre-loaded, or a user may need to insert a test strip in a device (and optionally, withdraw and replace the test strip between uses). In certain cases, the test strip may form an integral part of the device that is not removable by a user. In some embodiments, after exposure to the blood or other fluid withdrawn from the subject, the test strip may be removed from the device and determined externally, e.g., using other apparatuses able to determine the test strip, for example, commercially-available test strip readers.
[0079] In some embodiments, the device may include a separation membrane that is impermeable to blood cells and other substances. Fluid received from the subject may flow through a separation membrane, and the received fluid may include components of various sizes. For example, the device may receive blood that includes blood cells, clotting factors, proteins, and blood plasma, among other components. Larger components such as blood cells and other larger substances may not be able to pass through the separation membrane while blood plasma is free to pass. In some embodiments, this blood plasma is collected into a storage chamber. If anticoagulant is not introduced to the blood plasma, the blood plasma, which contains clotting factors such as fibrinogen, may clot, thereby resulting in a solid clot component and a liquid component. This liquid component is known as serum, which is blood plasma without fibrinogen or other clotting factors. This serum can be collected via aspiration or other suitable method out of the storage chamber, leaving the blood clots in the storage chamber. If anticoagulant is introduced to the blood plasma, the blood plasma will not clot and blood plasma can be collected out of the storage chamber instead. Thus, the embodiments described throughout the specification may be used to produce plasma or serum. More details regarding plasma and serum production can be found in U.S. patent application Ser. No. 13/456,505, entitled "Plasma or Serum Production and Removal of Fluids Under Reduced Pressure," published as U.S. Pat. Apl. Pub. No. 2012/0275955 on Nov. 1, 2012, incorporated herein by reference in its entirety.
[0080] In some embodiments, the device may be connected to an external apparatus for determining at least a portion of the device, a fluid removed from the device, an analyte suspected of being present within the fluid, or the like. For example, the device may be connected to an external analytical apparatus, and fluid removed from the device for later analysis, or the fluid may be analyzed within the device in situ, e.g., by adding one or more reaction entities to the device, for instance, to a storage chamber, or to analytical chamber within the device. In some embodiments, assay disks 200 or membranes may be included in storage chamber 140, as shown in FIG. 4. In one embodiment, the external apparatus may have a port or other suitable surface for mating with a port or other suitable surface on the device, and blood, interstitial fluid, or other fluid can be removed from the device using any suitable technique, e.g., using vacuum or pressure, etc. The blood or other fluid may be removed by the external apparatus, and optionally, stored and/or analyzed in some fashion. For example, in one set of embodiments, the device may include an exit port for removing a fluid from the device (e.g., blood). In some embodiments, fluid contained within a storage chamber in the device may be removed from the device, and stored for later use or analyzed outside of the device. In some cases, the exit port may be separate from the fluid transporter. In some cases, an exit port can be in fluidic communication with a vacuum source, which can also serve as a fluid reservoir in some cases. Other methods for removing blood, interstitial fluid, or other fluids from the device include, but are not limited to, removal using a vacuum line, a pipette, extraction through a septum instead of an exit port, or the like. In some cases, the device may also be positioned in a centrifuge and subjected to various g forces (e.g., to a centripetal force of at least 50 g), e.g., to cause at separation of cells or other substances within a fluid within the device to occur.
[0081] The device may include an anticoagulant or a stabilizing agent for stabilizing the fluid withdrawn from the skin and/or beneath the skin. As a specific non-limiting example, an anticoagulant may be used for blood withdrawn from the skin. Examples of anticoagulants include, but are not limited to, heparin, citrate, thrombin, oxalate, ethylenediaminetetraacetic acid (EDTA), sodium polyanethol sulfonate, acid citrate dextrose. Other agents may be used in conjunction with or instead of anticoagulants, for example, stabilizing agents such as solvents, diluents, buffers, chelating agents, enzyme inhibitors (i.e., Protease or Nuclease inhibitor), antioxidants, binding agents, preservatives, antimicrobials, or the like. Examples of preservatives include, for example, benzalkonium chloride, chlorobutanol, parabens, or thimerosal. Non-limiting examples of antioxidants include ascorbic acid, glutathione, lipoic acid, uric acid, carotenes, alpha-tocopherol, ubiquinol, or enzymes such as catalase, superoxide dismutase, or peroxidases. Examples of microbials include, but are not limited to, ethanol or isopropyl alcohol, azides, or the like. Examples of chelating agents include, but are not limited to, ethylene glycol tetraacetic acid or ethylenediaminetetraacetic acid. Examples of buffers include phosphate buffers such as those known to ordinary skill in the art.
[0082] In one set of embodiments, at least a portion of the device may be colored to indicate the anticoagulant(s) contained within the device. In some cases, the colors used may be identical or equivalent to that commercially used for Vacutainers.TM., Vacuettes.TM., or other commercially-available phlebotomy equipment. For example, lavender and/or purple may indicate ethylenediaminetetraacetic acid, light blue may indicate citrate, dark blue may indicate ethylenediaminetetraacetic acid, green may indicate heparin, gray may indicate a fluoride and/or an oxalate, orange may indicate a thrombin, yellow may indicate sodium polyanethol sulfonate and/or acid citrate dextrose, black may indicate citrate, brown may indicate heparin, etc. In other embodiments, however, other coloring systems may be used.
[0083] Other coloring systems may be used in other embodiments of the invention, not necessarily indicative of anti-coagulants. For example, in one set of embodiments, the device carries a color indicative of a recommended bodily use site for the device, e.g., a first color indicative of a device suitable for placement on the back, a second color indicative of a device suitable for placement on a leg, a third color indicative of a device suitable for placement on the arm, etc.
[0084] As mentioned, in one set of embodiments, a device of the invention as discussed herein may be shipped to another location for analysis. In some cases, the device may include an anticoagulant or a stabilizing agent contained within the device, e.g., within a storage chamber for the fluid. Thus, for example, fluid such as blood or interstitial fluid withdrawn from the skin and/or beneath the skin may be delivered to a chamber (e.g., a storage chamber) within the device, then the device, or a portion of the device (e.g., a module) may be shipped to another location for analysis. Any form of shipping may be used, e.g., via mail.
Alternative Embodiments
[0085] Alternative embodiments that may incorporate one or more aspects of the invention are discussed further below.
[0086] It should be understood that various components of a material delivery/receiving device may be modified in different ways, and that the embodiment discussed with respect to FIGS. 1-10 should not be used to limit aspects of the invention. For example, in one alternative embodiment, the retraction actuator 40 of a device 1 may include two separate elements. FIGS. 11-14 show an embodiment in which the retraction actuator 40 includes a retractor portion 42 and a seal actuator portion 44. As shown in FIGS. 11 and 12, the retractor portion 42 and the seal actuator portion 44 are stacked and coupled to the effector body 50 via five posts 52. Any number of posts may be used. The post 52 may be formed from or otherwise include Polyester (PCTA or PETG) or other polymer such as ABS, acetal resin, polystyrene, etc. Alternatively, the retractor portion 42 and the seal actuator portion 44 may be coupled to the effector body 50 via a single post, glue, tape, other adhesive, etc. FIG. 12 shows that the retractor portion 42 includes legs 48 that are free to flex relative to the effector 50. The seal actuator portion 44 includes tabs 41 and the seal leg 49 that is coupled to the seal 76. Both the retractor portion 42 and a seal actuator portion 44 otherwise have essentially the same features as the retraction actuator 40 described above. By separating the retraction actuator 40 into two portions, each may be designed and constructed to have desired features. For example, in some embodiments it may be desirable to have the legs 48 made of a highly elastic material, whereas the tabs 41 and seal leg 49 may be made of a less elastic material, e.g., to help release the seal 76 as the retraction actuator 40 moves upwardly. Additionally, as shown in FIG. 13, the membrane 72 may be made independent from the seal 76, e.g., the seal 76 may be formed as part of the seal leg 49 of the actuator 40. In some embodiments, the flow activator 90 may be mechanically coupled to the deployment actuator 60 via a transmission structure 94 such as a post, a rod, or other. As shown in FIG. 13, a post 94 is coupled to the membrane 72, the flow activator 90 and the deployment actuator 60, and may be made relatively stiff or non-compliant, e.g., to help transmit movement from the deployment actuator 60 to the flow activator 90 with little loss.
[0087] FIGS. 15-18 show yet another embodiment that is very similar to that of FIGS. 1-10, but in which the latch arrangement used to hold the retraction actuator 40 in an initial, compressed state is modified. In this illustrative embodiment, the device 1 contains a rotatable release element 170 that rotates relative to the base 100 during operation of the device. (The rotatable release element 170 and corresponding portions of the base 100 replace the release element 30 and the tabs 41 of the retraction actuator 40 of the FIGS. 1-10 embodiment.) A spinner ramp 174 of the release element 170 initially engages with a lock-out ramp 161 of an effector guide 104 and holds the rotatable release element 170 in place prior to actuation of the device 1. FIG. 16 shows a close-up of the initial engagement prior to actuation of the device 1. However, when the rotatable release element 170 is moved toward the base 100 during device actuation (e.g., depression of the device actuator 10), the release element 170 rotates slightly so that the spinner ramp 174 slides and clears the lock-out ramp 161 as the release element 170 moves towards the base 100. (Slight rotation of the release element 170 may be caused by a ramp or other angled surface on the element 170 contacting a corresponding ramp or other surface of the base 100 so that downward movement of the release element 170 upon actuation of the device actuator 10 causes the desired rotation.) Thereafter, when pressure on the release element 170 is released by the user, the spinner release ramp 175 engages the base release ramp 160 as the release element 170 moves upward so that as the rotatable release element 170 rotates so that the spinner release ramp 175 clears the base release ramp 160. This may allow the retraction actuator 40 to retract, e.g., to retract the flow activator 90.
[0088] In yet other embodiments, a material delivery/receiving device 10 may be arranged in other ways, as suggested above. For example, in one embodiment shown in FIGS. 19-25, a fluid receiving device 1 includes a horizontally sliding trigger 304 that can be actuated by a user or other by finger depression. Similar to the embodiments described above and as shown in FIGS. 19 and 20, the device 1 includes a cover 20 and a base 100, and fluid received at an opening 130 of a fluid transporter 120 may be conducted by a channel 110 to a storage chamber 140 (not shown). FIGS. 21 and 22 show internal components of the device 1. An O-ring seal 340 may be located on a trigger shaft 306 of the trigger 304. In another embodiment, a deformable membrane could form the seal. During use, sliding the trigger 304 rearwardly towards a trailing edge 102 of the base 100 causes the trigger shaft 306 to push the trigger pin 332 with a trigger pin cover 334 (see FIG. 22). This motion causes a carriage 330 to slide rearwardly along guides 360 (See FIG. 21) on the base 100 toward the trailing end 102 of the base 100. The guides 360 may be etched into the base 100, may be protruded from the base 100, or have any other suitable arrangement.
[0089] As the carriage 330 moves rearwardly, a trigger bridge 336 connected to the carriage 330 moves rearwardly relative to the effector body 50. The underside of the trigger bridge 336 includes a trigger tab 338, as can be seen in FIGS. 23A and 23B. The trigger tab 338 engages with a protrusion 339 (see FIG. 24) on the top of the effector body 50 so that as the trigger bridge 336 moves rearwardly, the trigger tab 338 moves the effector body 50 downwardly a sufficient amount to actuate a deployment actuator 60, which has a configuration like that in the embodiments described above. This causes the deployment actuator 60 to deploy the flow activator 90, e.g., to extend needles from the opening 130. Continued movement of the carriage 330 in the rearward direction causes a retraction actuator of the trigger (in the form of wedges 350) to slide beneath lifting struts 370 on the effector body 50. As the wedges 350 slide beneath the lifting struts 370, the effector 50 is lifted upwardly away from base 100, thereby retracting the flow activator 90, which is attached to the effector body 50 via the deployment actuator 60, and membrane 72 in a way similar to the embodiments above. The trigger tab 338 may be received in an opening 380 in the effector body 50, allowing a central portion of the effector body 50 to flex upwardly and allowing further retraction of the flow activator 90.
[0090] Connection of a flow actuator to a deployment actuator may be done in a variety of different ways, as suggested above. For example, FIG. 26A shows a schematic arrangement in which a post 94 used to connect a flow activator (not shown) to a membrane 72 and/or a deployment actuator 60 may be made by an adhesive 400. In another embodiment shown in FIG. 26B, the post 94 may be received into a cavity (or hole) in the membrane 72 as well as a hole in the deployment actuator 60. Engagement of the post 94 with the respective holes or cavities may be made in any suitable way, such as by interference or friction fit, adhesive, riveting, and so on. In this embodiment, the post 94 is engaged with a cavity of the membrane 72 by an adhesive 400 and has a rivet-type head that engages with the hole in the deployment actuator 60. The rivet head of the post 94 may be formed by plastically deforming part of the post 94, or the post 94 may include a flexible material arranged so that an upper portion of the rivet head may be resiliently deformed and forced through the hole of the actuator 60. FIG. 26C shows yet another embodiment in which a membrane 72 is joined to a deployment actuator by extending a portion of the membrane 72 through an opening in the actuator 60 and crimping or otherwise deforming the portion of the membrane 72 that extends through the opening. Alternately, a clip, band or other element may be clamped onto the membrane portion to maintain engagement of the membrane and actuator 60. The post 94 may be attached to both the membrane and actuator as part of the same process, e.g., part of the post may function as a clip or band. FIG. 26D shows an embodiment with a two part post 94 where the membrane 72 is trapped between the two parts of the post. The top part of the post extends through a hole in the deployment actuator 60 or is heat staked to create an interference fit between the post and the deployment actuator 60. A portion of the post 94 may be forced through an opening at the connection point, and thereby be engaged with the deployment actuator 60.
[0091] Further details regarding optional arrangements for needles, which may be included as part of a flow activator, are provided below.
[0092] As mentioned above, needles included with a flow activator may be arranged in a variety of different ways, depending on the intended application. For example, the needle(s) may have a length of less than about 5 mm, less than about 4 mm, less than about 3 mm, less than about 2 mm, less than about 1 mm, less than about 800 micrometers, less than 600 micrometers, less than 500 micrometers, less than 400 micrometers, less than about 300 micrometers, less than about 200 micrometers, less than about 175 micrometers, less than about 150 micrometers, less than about 125 micrometers, less than about 100 micrometers, less than about 75 micrometers, less than about 50 micrometers, less than about 10 micrometers, etc. The needle(s) may also have a largest cross-sectional dimension of less than about 5 mm, less than about 4 mm, less than about 3 mm, less than about 2 mm, less than about 1 mm, less than about 800 micrometers, less than 600 micrometers, less than 500 micrometers, less than 400 micrometers, less than about 300 micrometers, less than about 200 micrometers, less than about 175 micrometers, less than about 150 micrometers, less than about 125 micrometers, less than about 100 micrometers, less than about 75 micrometers, less than about 50 micrometers, less than about 10 micrometers, etc. For example, in one embodiment, the needle(s) may have a rectangular cross section having dimensions of 175 micrometers by 50 micrometers. In one set of embodiments, the needle(s) may have an aspect ratio of length to largest cross-sectional dimension of at least about 2:1, at least about 3:1, at least about 4:1, at least 5:1, at least about 7:1, at least about 10:1, at least about 15:1, at least about 20:1, at least about 25:1, at least about 30:1, etc.
[0093] In one embodiment, the needle(s) is(are) a microneedle(s). Typically, a microneedle will have an average cross-sectional dimension (e.g., diameter) of less than about a millimeter. It should be understood that references to "needle" or "microneedle" as discussed herein are by way of example and ease of presentation only, and that in other embodiments, more than one needle and/or microneedle may be present in any of the descriptions herein.
[0094] As an example, microneedles such as those disclosed in U.S. Pat. No. 6,334,856, issued Jan. 1, 2002, entitled "Microneedle Devices and Methods of Manufacture and Use Thereof," by Allen, et al., may be used to deliver to and/or withdraw fluids (or other materials) from a subject. The microneedles may be hollow or solid, and may be formed from any suitable material, e.g., metals, ceramics, semiconductors, organics, polymers, and/or composites. Examples include, but are not limited to, medical grade stainless steel, titanium, nickel, iron, gold, tin, chromium, copper, alloys of these or other metals, silicon, silicon dioxide, and polymers, including polymers of hydroxy acids such as lactic acid and glycolic acid polylactide, polyglycolide, polylactide-co-glycolide, and copolymers with polyethylene glycol, polyanhydrides, polyorthoesters, polyurethanes, polybutyric acid, polyvaleric acid, polylactide-co-caprolactone, polycarbonate, polymethacrylic acid, polyethylenevinyl acetate, polytetrafluorethylene, polymethyl methacrylate, polyacrylic acid, or polyesters.
[0095] In some cases, more than one needle or microneedle may be used. For example, arrays of needles or microneedles may be used, and the needles or microneedles may be arranged in the array in any suitable configuration, e.g., periodic, random, etc. In some cases, the array may have 3 or more, 4 or more, 5 or more, 6 or more, 10 or more, 15 or more, 20 or more, 35 or more, 50 or more, 100 or more, or any other suitable number of needles or microneedles. Typically, a microneedle will have an average cross-sectional dimension (e.g., diameter) of less than about a micron.
[0096] Those of ordinary skill in the art can arrange needles relative to the skin or other surface for these purposes including, in one embodiment, introducing needles into the skin at an angle, relative to the skin's surface, other than 90.degree., i.e., to introduce a needle or needles into the skin in a slanting fashion so as to limit the depth of penetration. In another embodiment, however, the needles may enter the skin or other surface at approximately 90.degree..
[0097] In some cases, the needles (or microneedles) may be present in an array selected such that the density of needles within the array is between about 0.5 needles/mm.sup.2 and about 10 needles/mm.sup.2, and in some cases, the density may be between about 0.6 needles/mm.sup.2 and about 5 needles/mm.sup.2, between about 0.8 needles/mm.sup.2 and about 3 needles/mm.sup.2, between about 1 needles/mm.sup.2 and about 2.5 needles/mm.sup.2, or the like. In some cases, the needles may be positioned within the array such that no two needles are closer than about 1 mm, about 0.9 mm, about 0.8 mm, about 0.7 mm, about 0.6 mm, about 0.5 mm, about 0.4 mm, about 0.3 mm, about 0.2 mm, about 0.1 mm, about 0.05 mm, about 0.03 mm, about 0.01 mm, etc.
[0098] In another set of embodiments, the needles (or microneedles) may be chosen such that the area of the needles (determined by determining the area of penetration or perforation on the surface of the skin of the subject by the needles) allows for adequate flow of fluid to or from the skin and/or beneath the skin of the subject. The needles may be chosen to have smaller or larger areas (or smaller or large diameters), so long as the area of contact for the needles to the skin is sufficient to allow adequate blood flow from the skin of the subject to the device. For example, in certain embodiments, the needles may be selected to have a combined skin-penetration area of at least about 500 nm.sup.2, at least about 1,000 nm.sup.2, at least about 3,000 nm.sup.2, at least about 10,000 nm.sup.2, at least about 30,000 nm.sup.2, at least about 100,000 nm.sup.2, at least about 300,000 nm.sup.2, at least about 1 microns.sup.2, at least about 3 microns.sup.2, at least about 10 microns.sup.2, at least about 30 microns.sup.2, at least about 100 microns.sup.2, at least about 300 microns.sup.2, at least about 500 microns.sup.2, at least about 1,000 microns.sup.2, at least about 2,000 microns.sup.2, at least about 2,500 microns.sup.2, at least about 3,000 microns.sup.2, at least about 5,000 microns.sup.2, at least about 8,000 microns.sup.2, at least about 10,000 microns.sup.2, at least about 35,000 microns.sup.2, at least about 100,000 microns.sup.2, at least about 300,000 microns.sup.2, at least about 500,000 microns.sup.2, at least about 800,000 microns.sup.2, at least about 8,000,000 microns.sup.2, etc., depending on the application.
[0099] The needles or microneedles may have any suitable length, and the length may be, in some cases, dependent on the application. For example, needles designed to only penetrate the epidermis may be shorter than needles designed to also penetrate the dermis, or to extend beneath the dermis or the skin. In certain embodiments, the needles or microneedles may have a maximum penetration into the skin of no more than about 3 mm, no more than about 2 mm, no more than about 1.75 mm, no more than about 1.5 mm, no more than about 1.25 mm, no more than about 1 mm, no more than about 900 microns, no more than about 800 microns, no more than about 750 microns, no more than about 600 microns, no more than about 500 microns, no more than about 400 microns, no more than about 300 microns, no more than about 200 microns, no more than about 175 micrometers, no more than about 150 micrometers, no more than about 125 micrometers, no more than about 100 micrometers, no more than about 75 micrometers, no more than about 50 micrometers, etc. In certain embodiments, the needles or microneedles may be selected so as to have a maximum penetration into the skin of at least about 50 micrometers, at least about 100 micrometers, at least about 300 micrometers, at least about 500 micrometers, at least about 1 mm, at least about 2 mm, at least about 3 mm, etc.
[0100] In one set of embodiments, the needles (or microneedles) may be coated. For example, the needles may be coated with a substance that is delivered when the needles are inserted into the skin. For instance, the coating may comprise heparin, an anticoagulant, an anti-inflammatory compound, an analgesic, an anti-histamine compound, etc. to assist with the flow of blood from the skin of the subject, or the coating may comprise a drug or other therapeutic agent such as those described herein. The drug or other therapeutic agent may be one used for localized delivery (e.g., of or proximate the region to which the coated needles or microneedles are applied), and/or the drug or other therapeutic agent may be one intended for systemic delivery within the subject.
[0101] FIG. 27 shows an illustrative embodiment of a material delivery/receiving device 1 that is arranged to deliver a drug or other material to a user's skin. In this illustrative embodiment, the device 1 is arranged as a relatively low profile assembly, having a total height of about 10 mm or less and a diameter of 50 mm or less. Of course, other dimensions may be used for the height and diameter of the device 1. The device 1 may be deployed as a patch or other similar arrangement, e.g., where the interface 105 includes an adhesive so that the device 1 can be adhered to a user's skin. FIG. 28 shows perspective views of two possible arrangements for a device 1, one with a circular shape, the other with a circular central portion and two tabs extending outwardly, e.g., in a way similar to some adhesive bandage arrangements. After adhering the device 1 to a user's skin or other body part, a device actuator 10 may be depressed or otherwise actuated to cause the device 1 to deliver drug or other material to the skin. In this embodiment, depression of the actuator 10 causes an effector 50 (which may include a snap dome actuator like that discussed above) to deploy a flow activator 90 (e.g., including a plurality of needles) to move downwardly toward the user's skin. In their downward movement, the needles or other components of the flow activator 90 may penetrate a portion 105a of the interface 105 that is positioned at an opening 130 of the device 1. After penetrating the portion 105a, the needles or other components may penetrate the user's skin. The interface portion 105a may be arranged in any suitable way, but in this embodiment may include a drug eluting hydrogel or other material that is arranged to deliver a drug or other material to the needles or other flow activator components. The material(s) included in an interface portion 105a may be, for example, an absorbent material, a sponge, gauze, a swab, a membrane, a filter, a pad, or the like, and may depend on the drug or other material to be delivered. For example, needles passing through the activator portion 105a may pick up drug from the portion 105a, and carry the drug into the skin. In other arrangements, drug or other material may be delivered to the needles or otherwise to the skin on an extended basis. For example, the needles may remain deployed in the skin after activation, and drug may be carried from the portion 105a to the skin by the needles. Alternately, openings in the skin formed by the needles (whether the needles are withdrawn or not) may provide a pathway for the drug or other material to move from the portion 105a to the skin, e.g., by diffusion or other transport. As discussed above, the needles may be hollow, include surface flow channels, be porous, be resorbable, or otherwise configured to aid in the delivery of material to the skin. Thus, the device may remain in place on a user for minutes, hours, days, months, etc.
[0102] Also, the device 1 may include other features to help with drug or other material delivery, such as a positive pressure reservoir that opens or otherwise helps deliver a drug or other material to the skin. For example, the portion 105a could include an impermeable membrane that is pierced by the flow activator 90. A positive pressure may be held inside the device 1 (e.g., in the space under the cover 20), and piercing of the membrane may allow the positive internal pressure to push drug or other material carried by the portion 105a toward the skin. In other arrangements discussed above, the device 1 may include a drug or other material reservoir that is fluidly coupled to the opening 130, e.g., by a conduit that is fluidly coupled to the flow activator 90 which may include a plurality of hollow needles. Activation of the device may expose the reservoir to a positive pressure, which drives the drug or other material to/through the needles and into the skin.
[0103] Drug or other material delivery could be coupled with blood or other material withdrawal from the skin or other user body portion. For example, a flow activator may be deployed to pierce the skin and cause withdrawal of blood, e.g., due in part to exposure to a vacuum. The received blood may be conducted to a storage chamber, an absorbent pad or other material, etc. Thereafter (or simultaneous therewith), a drug or other material may be delivered in a manner like that described above or otherwise. For example, a portion 105a of an interface 105 may carry a blood-thinning agent, pain-reducer or other component to aid in blood removal, pain reduction, or other aspect of blood reception. By passing through the portion 105a, needles or other flow activator components may pickup the drug and deliver the drug upon penetration into the skin. Drug or other material may continue to be delivered, with different drugs or other materials being delivered at different times in the use of the device. For example, some drugs or other material may be encapsulated in a material that dissolves and releases the drug after being exposed to blood. In this way, the drug may be delivered only after having been exposed to blood for a period of time. This may allow for the collection of a drug-free blood sample, followed by delivery of the drug. For example, in one embodiment, a blood sample may be taken by the device for use in determining a glucose level of the user. Thereafter, insulin or other suitable drug may be delivered by the device, possibly in an amount dependent on the glucose level determined. The device 1 may include some sort of indicator that drug or other material has been delivered. For example, a color change material may change its color indication to indicate that the drug has been delivered, e.g., based on exposure of the material to blood, passage of the drug to the flow activator, etc.
[0104] In some cases, the device may be an electrical and/or a mechanical device applicable or affixable to the surface of the skin, e.g., using adhesive, or other techniques such as those described herein. For example, in one set of embodiments, the device may include a support structure that contains an adhesive that can be used to immobilize the device to the skin. The adhesive may be permanent or temporary, and may be used to affix the device to the surface of the skin. The adhesive may be any suitable adhesive, for example, a pressure sensitive adhesive, a contact adhesive, a permanent adhesive, a cyanoacrylate, glue, gum, hot melts, an epoxy, a hydrogel, a hydrocolloid, or the like. In some cases, the adhesive is chosen to be biocompatible or hypoallergenic.
[0105] In another set of embodiments, the device may be mechanically held to the skin, For example, the device may include mechanical elements such as straps, belts, buckles, strings, ties, elastic bands, or the like. For example, a strap may be worn around the device to hold the device in place against the skin of the subject. In yet another set of embodiments, a combination of these and/or other techniques may be used. As one non-limiting example, the device may be affixed to a subject's arm or leg using adhesive and a strap.
[0106] Any or all of the arrangements described herein can be provided proximate a subject, for example on or proximate the skin of the subject, in various aspects. Activation of the devices can be carried out in a variety of ways, e.g., as described herein. For example, an on-skin device can be in the form of a patch or the like, optionally including multiple layers for activation, sensing, fluid flow, etc. In one embodiment, a patch or a device can be applied to a subject and a region of the patch or device activated (e.g., pushed, pressed, or tapped by a user) to inject a needle or a microneedle, or other fluid transporter, so as to access interstitial fluid or blood. The same or a different activation action, e.g., tapping or pushing action, can activate a vacuum source, open and/or close one or more of a variety of valves, or the like. The device can be a simple one in which it is applied to the skin and operates automatically (where e.g., application to the skin of the device allows access to interstitial fluid or blood, and delivers and/or withdraws fluid) or the patch or other device can be applied to the skin and one tapping or other activation action can cause fluid to flow through administration of a needle or a microneedle (or other fluid transporter), opening of a valve, activation of vacuum, etc., or any combination thereof. Any number of activation protocols can be carried out by a user repeatedly pushing, tapping, etc. a location or selectively, sequentially, and/or periodically activating a variety of switches (e.g., tapping regions of a patch or device).
[0107] As mentioned, the device may include an anticoagulant or a stabilizing agent for stabilizing the fluid withdrawn from the skin. As a specific non-limiting example, an anticoagulant may be used for blood withdrawn from the skin. Examples of anticoagulants include, but are not limited to, heparin, citrate, oxalate, or ethylenediaminetetraacetic acid (EDTA). Other agents may be used in conjunction or instead of anticoagulants, for example, stabilizing agents such as solvents, diluents, buffers, chelating agents, antioxidants, binding agents, preservatives, antimicrobials, or the like. Examples of preservatives include, for example, benzalkonium chloride, chlorobutanol, parabens, or thimerosal. Non-limiting examples of antioxidants include ascorbic acid, glutathione, lipoic acid, uric acid, carotenes, alpha-tocopherol, ubiquinol, or enzymes such as catalase, superoxide dismutase, or peroxidases. Examples of microbials include, but are not limited to, ethanol or isopropyl alcohol, azides, or the like. Examples of chelating agents include, but are not limited to, ethylene glycol tetraacetic acid or ethylenediaminetetraacetic acid. Examples of buffers include phosphate buffers such as those known to ordinary skill in the art.
[0108] As yet another example, the device may include a therapeutic agent such as an anti-inflammatory compound, an analgesic, or an anti-histamine compound. Examples of anti-inflammatory compounds include, but are not limited to, NSAIDs (non-steroidal anti-inflammatory drugs) such as aspirin, ibuprofen, or naproxen. Examples of analgesics include, but are not limited to, benzocaine, butamben, dibucaine, lidocaine, oxybuprocaine, pramoxine, proparacaine, proxymetacaine, tetracaine, acetaminophen, NSAIDs such as acetylsalicylic acid, salicylic acid, diclofenac, ibuprofen, etc., or opioid drugs such as morphine or opium, etc. Examples of anti-histamine compounds include, but are not limited to, clemastine, diphenhydramine, doxylamine, loratadine, desloratadine, fexofenadine, pheniramine, cetirizine, ebastine, promethazine, chlorpheniramine, levocetirizine, olopatadine, quetiapine, meclizine, dimenhydrinate, embramine, dimethindene, dexchlorpheniramine, vitamin C, cimetidine, famotidine, ranitidine, nizatidine, roxatidine, or lafutidine. Other specific non-limiting examples of therapeutic agents that could be used include, but are not limited to biological agents such as erythropoietin ("EPO"), alpha-interferon, beta-interferon, gamma-interferon, insulin, morphine or other pain medications, antibodies such as monoclonal antibodies, or the like. In short, the type of drug(s) or other material(s) delivered by the device 1 need not be limited in any particular way or method of delivery.
[0109] U.S. Provisional Patent Application Ser. No. 61/577,399, filed Dec. 19, 2011, entitled "Delivering and/or Receiving Material with Respect to a Subject Surface," by Bernstein, et al., is incorporated herein by reference in its entirety. In addition, each of the following is incorporated herein by reference in its entirety: U.S. patent application Ser. No. 12/716,229, filed Mar. 2, 2010; U.S. patent application Ser. No. 12/716,226, filed Mar. 2, 2010; U.S. patent application Ser. No. 12/915,735, filed Oct. 29, 2010; U.S. patent application Ser. No. 12/915,789, filed Oct. 29, 2010; U.S. patent application Ser. No. 12/915,820, filed Oct. 29, 2010; U.S. patent application Ser. No. 12/953,744, filed Nov. 24, 2010; U.S. patent application Ser. No. 13/006,165, filed Jan. 13, 2011; U.S. patent application Ser. No. 13/006,177, filed Jan. 13, 2011; U.S. patent application Ser. No. 13/016,575, filed Jan. 28, 2011; PCT Apl. No. PCT/US2011/043698, filed Jul. 12, 2011; PCT Apl. No. PCT/US2011/047565, filed Aug. 12, 2011; U.S. patent application Ser. No. 13/456,570, filed Apr. 26, 2012; U.S. patent application Ser. No. 13/456,394, filed Apr. 26, 2012; U.S. patent application Ser. No. 13/456,505, filed Apr. 26, 2012; U.S. patent application Ser. No. 13/456,546, filed Apr. 26, 2012; and U.S. Prov. Pat. Apl. Ser. No. 61/577,399, filed Dec. 19, 2011.
[0110] While aspects of the invention have been described with reference to various illustrative embodiments, such aspects are not limited to the embodiments described. Thus, it is evident that many alternatives, modifications, and variations of the embodiments described will be apparent to those skilled in the art. Accordingly, embodiments as set forth herein are intended to be illustrative, not limiting. Various changes may be made without departing from the spirit of aspects of the invention.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018
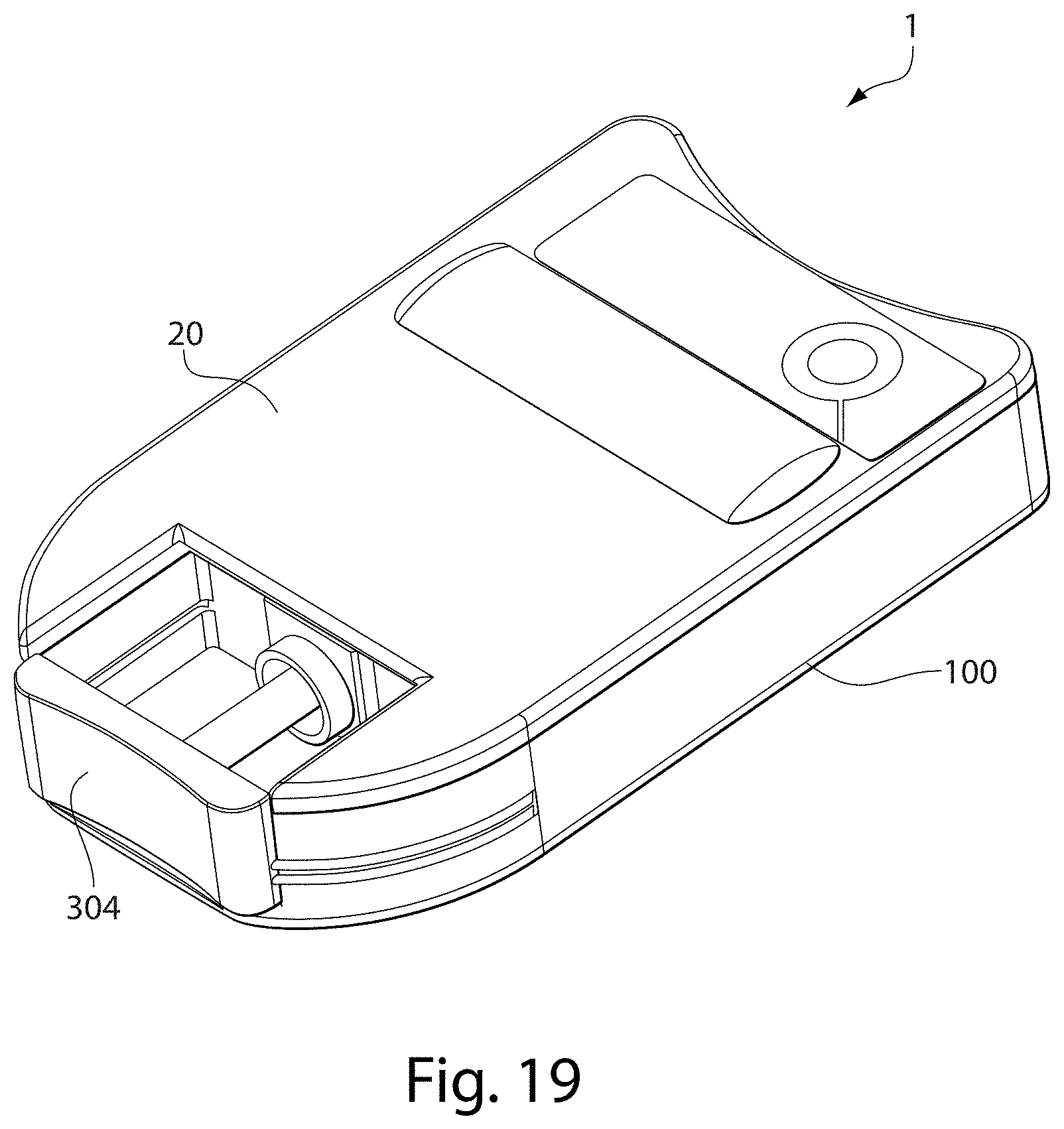
D00019

D00020

D00021

D00022
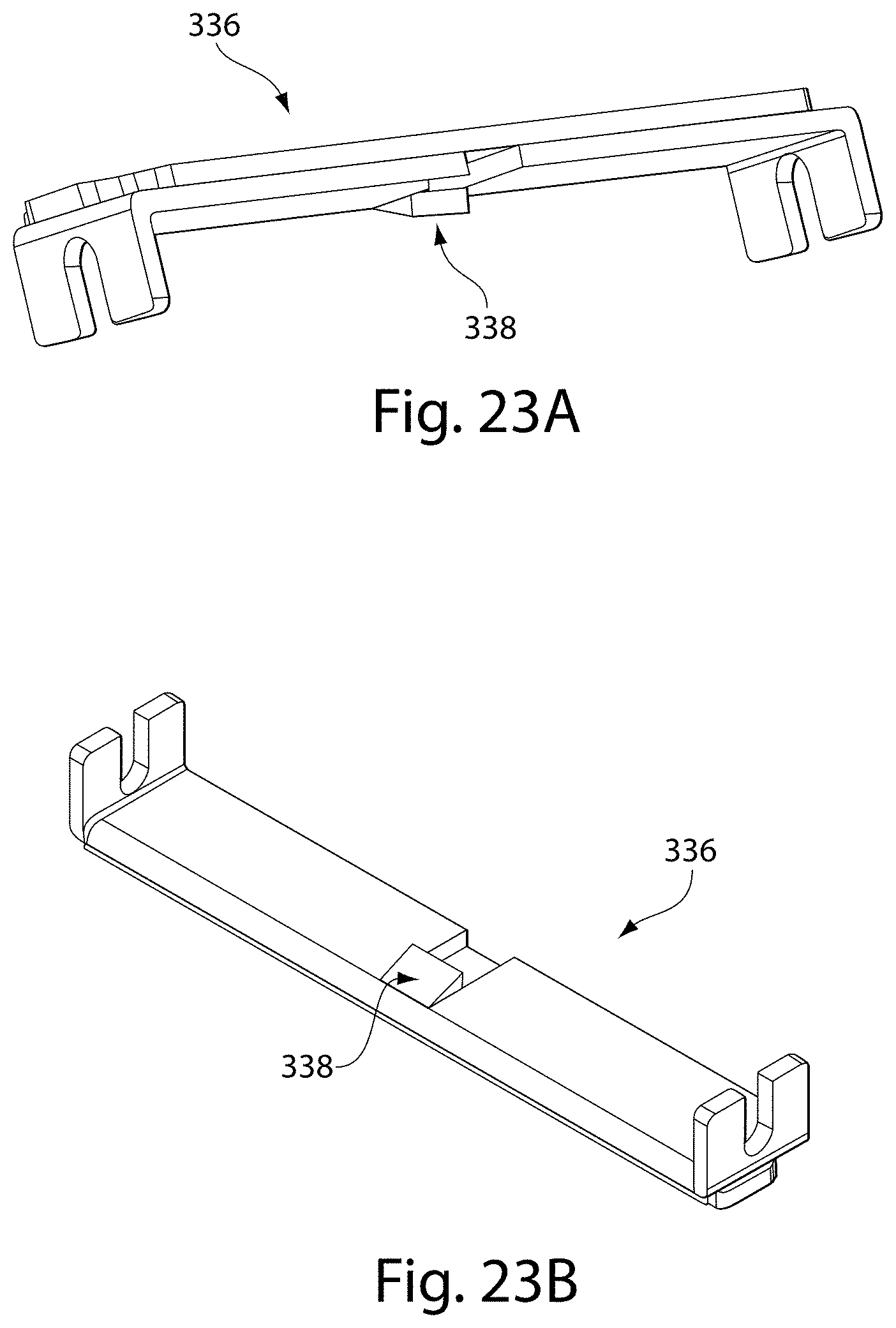
D00023

D00024

D00025

D00026

D00027

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.