Tracking And Delivery Confirmation Of Pharmaceutical Products
THOMPSON; Todd ; et al.
U.S. patent application number 16/680554 was filed with the patent office on 2020-10-29 for tracking and delivery confirmation of pharmaceutical products. The applicant listed for this patent is Proteus Digital Health, Inc.. Invention is credited to Lawrence ARNE, Yashar BEHZADI, Lorenzo DICARLO, Robert DUCK, Gregory MOON, Fataneh OMIDVAR, Todd THOMPSON.
| Application Number | 20200342555 16/680554 |
| Document ID | / |
| Family ID | 1000004944545 |
| Filed Date | 2020-10-29 |



| United States Patent Application | 20200342555 |
| Kind Code | A1 |
| THOMPSON; Todd ; et al. | October 29, 2020 |
TRACKING AND DELIVERY CONFIRMATION OF PHARMACEUTICAL PRODUCTS
Abstract
A system and method are disclosed that track a deliverable to a user. The system includes an identifier or tag secured to the deliverable, a computer system for interrogating the identifier, and a personal device in communication with the computer system, wherein the personal device is held by the user at the time the user is administered the deliverable to detect the unique identity associated with the identifier device and confirms delivery of the deliverable to the user. The method includes attaching an identifiable tag that produces a unique signature to the deliverable, interrogating the tag at about the time of delivery to the user, and confirming that the user has been administered the deliverable through detecting the identifiable tag.
| Inventors: | THOMPSON; Todd; (San Jose, CA) ; ARNE; Lawrence; (Palo Alto, CA) ; OMIDVAR; Fataneh; (Danville, CA) ; BEHZADI; Yashar; (San Francisco, CA) ; DUCK; Robert; (San Francisco, CA) ; DICARLO; Lorenzo; (San Francisco, CA) ; MOON; Gregory; (Palo Alto, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004944545 | ||||||||||
| Appl. No.: | 16/680554 | ||||||||||
| Filed: | November 12, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 13698953 | Nov 19, 2012 | 10529044 | ||
| PCT/US2011/037236 | May 19, 2011 | |||
| 16680554 | ||||
| 61346035 | May 19, 2010 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61M 3/00 20130101; G16H 20/17 20180101; G16H 20/13 20180101; G08C 15/06 20130101; G06Q 50/22 20130101; H04N 7/18 20130101 |
| International Class: | G06Q 50/22 20060101 G06Q050/22; G16H 20/13 20060101 G16H020/13; A61M 3/00 20060101 A61M003/00; G08C 15/06 20060101 G08C015/06; H04N 7/18 20060101 H04N007/18 |
Claims
1. A computer system to track medication from manufacturer to a patient, to enable a care provider to determine the origin of a medication and to confirm that at least one of the right type and the right dosage of the medication was delivered to the patient, the computer system comprising: a processor operative to: receive a unique identity of an identifier device secured to a dosage of a medication, wherein the identifier device comprises the unique identity that is associated with at least one of the manufacturer and the medication; receive information from a patient interface device in communication with the computer and the patient, wherein at the time the patient takes the medication, the patient interface device is operative to detect the unique identity associated with the identifier device and confirm delivery of the medication to the patient; and receive the detected unique identity associated with the identifier device detected via the patient interface device and the confirmation of delivery of the medication to the patient via the patient interface device; wherein the processor is operative to confirm the origin of the medication and the type of the medication based at least on the unique identity of the identifier device.
2. The computer system of claim 1, wherein the processor is operative to interrogate the identifier device.
3. The computer system of claim 1, wherein the identifier device comprises a tag provided with circuitry, wherein the tag is secured to the dosage of the medication, and wherein the processor is operative to read the unique identity from the tag.
4. The computer system of claim 3, wherein the tag is secured to a dosage of medication in the form of a pill.
5. The computer system of claim 3, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
6. The computer system of claim 5, wherein the syringe comprises a needle and wherein the needle comprises at least one contact point coupled to the tag.
7. The computer system of claim 1, wherein the processor is operative to communicate with a datacenter database to at least one of retrieve medical information about the patient and provide information related to the medication to be administered to the patient.
8. The computer system of claim 7, wherein the processor is operative to: communicate with at least one of a database or a processing system at the datacenter; transmit information to the processing system; receive information from the processing system, wherein the processing system is operative to access the information in the database of the data center and provide the information to the patient through the computer.
9. The computer system of claim 1, wherein the processor is operative to validate at least one of the type and dosage selected by the care provider prior to dispensing the medication to the patient.
10. The computer system of claim 1, wherein the processor is operative to communicate with an interrogation unit coupled to the identifier device, wherein the interrogation unit is operative to interrogate the identifier device secured to the medication and receive the unique identity of the identifier device to confirm the origin of the medication and the type of the medication, wherein the computer is operative to interrogate the interrogation unit and receive the unique identity of the identifier from the interrogation unit.
11. The computer system of claim 10, wherein the processor is operative to: communicate with a memory of the interrogation unit; and receive the unique identity of the identifier stored in the memory of the interrogation unit.
12. The computer system of claim 1, wherein the processor is operative to receive biometric parameters from the patient interface device, wherein the biometric parameters can identify the patient.
13. The computer system of claim 1, wherein the processor is operative to receive an image of the patient captured by a camera on the patient interface device.
14. The computer system of claim 1, comprising a display to provide information associated with the patient, wherein the information comprises any of an image of the patient, medical history of the patient, and a next type and dose of medication to deliver to the patient.
15. A computer-implemented method to track medication from manufacturer to a patient, to enable a care provider to determine the origin of a medication and to confirm that at least one of the right type and the right dosage of the medication was delivered to the patient, the computer comprising a processor, the method comprising: receiving by the processor a unique identity of the identifier device secured to a dosage of medication, wherein the identifier device comprises the unique identity that is associated with at least one of the manufacturer and the medication; receiving by the processor information from a patient interface device in communication with the computer and the patient, wherein at the time the patient takes the medication, the patient interface device is operative to detect the unique identity associated with the identifier device and confirm delivery of the medication to the patient; receiving by the processor the detected unique identity associated with the identifier device detected via the patient interface device and the confirmation of delivery of the medication to the patient via the patient interface device; confirming the origin of the medication and the type of the medication based at least on the unique identity of the identifier device.
16. The computer-implemented method of claim 15, comprising interrogating by the processor the identifier device.
17. The computer-implemented method of claim 15, comprising receiving by the processor the unique identity from a tag, wherein the identifier device comprises the tag provided with circuitry, wherein the tag is secured to the dosage of the medication, the method.
18. The computer-implemented method of claim 17, comprising receiving by the processor the unique identity from the tag, wherein the tag is secured to a dosage of medication in the form of a pill.
19. The computer-implemented method of claim 17, comprising receiving by the processor the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
20. The computer-implemented method of claim 19, comprising receiving by the processor the unique identity from the tag, wherein the syringe comprises a needle, and wherein the needle comprises at least one contact point coupled to the tag.
21. The computer-implemented method of claim 15, comprising at least one of: retrieving by the processor medical information about the patient from a datacenter database; and providing by the processor information related to the medication to be administered to the patient.
22. The computer-implemented method of claim 21, comprising: transmitting by the processor information to the processing system; receiving by the processor information from the processing system, wherein the processing system is operative to access the information in the database of the data center and provide the information to the patient through the computer.
23. The computer-implemented method of claim 15, comprising: validating by the processor at least one of the type and dosage selected by the care provider prior to dispensing the medication to the patient.
24. The computer-implemented method of claim 15, comprising: interrogating by the processor operative an interrogation unit coupled to the identifier device, wherein the interrogation unit is operative to interrogate the identifier device secured to the medication and receive the unique identity of the identifier device to confirm the origin of the medication and the type of the medication; and receiving by the computer the unique identity of the identifier from the interrogation unit.
25. The computer-implemented method of claim 24, comprising: communicating by the processor with a memory of the interrogation unit; and receiving by the processor the unique identity of the identifier stored in the memory of the interrogation unit.
26. The computer-implemented method of claim 15, comprising: receiving by the processor biometric parameters from the patient interface device, wherein the biometric parameters can identify the patient.
27. The computer-implemented method of claim 15, comprising: receiving by the processor an image of the patient captured by a camera on the patient interface device.
28. The computer-implemented method of claim 15, comprising: displaying on a display coupled to the processor information associated with the patient, wherein the information comprises any of an image of the patient, medical history of the patient, and a next type and dose of medication to deliver to the patient.
29. An apparatus, comprising: at least one contact point configured to electrically engage an identifier device secured to a medication, wherein the identifier device comprises a unique identity that is associated with at least one of the manufacturer and the medication; an interrogation unit coupled to the at least one contact point, wherein the interrogation unit is operative to interrogate the identifier device to obtain the unique identity; and a communication module coupled to the interrogation unit, wherein the communication module is operative to transmit the unique identification.
30. The apparatus of claim 29, wherein the interrogation unit is operative to read the unique identity associated with the identifier device.
31. The apparatus of claim 30, wherein the communication module is operative to transmit the unique identity associated with the identifier device to a computer in communication with the communication module.
32. The apparatus of claim 31, comprising a memory coupled to the interrogation unit to store the unique identity associated with the identifier device.
33. The apparatus of claim 29, wherein the interrogation unit is operative to read the unique identity from a tag provided with circuitry, wherein the tag is secured to the dosage of the medication.
34. The apparatus of claim 33, wherein the interrogation unit is operative to read the unique identity from the tag, wherein the tag is secured to a dosage of medication in the form of a pill.
35. The apparatus of claim 33, wherein the interrogation unit is operative to read the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
36. The apparatus of claim 35, wherein the interrogation unit is operative to read the unique identity from the tag, wherein the syringe comprises a needle and wherein the needle comprises at least one contact point coupled to the tag.
37. A method, comprising: electrically engaging by an interrogation unit an identifier device secured to a medication with at least one contact point coupled to the interrogation unit, wherein the identifier device comprises a unique identity that is associated with at least one of the manufacturer and the medication; interrogating the identifier device for the unique identity; and transmitting the unique identification by a communication module coupled to the interrogation unit.
38. The method of claim 37, comprising reading by the interrogation unit the unique identity associated with the identifier device.
39. The method of claim 38, comprising transmitting by the communication module the unique identity associated with the identifier device to a computer in communication with the communication module.
40. The method claim 38, storing by the interrogation unit the unique identity associated with the identifier device in a memory, wherein the memory is coupled to the interrogation unit.
41. The method of claim 37, the method comprising receiving by the interrogation unit the unique identity from a tag, wherein the identifier device comprises the tag provided with circuitry, wherein the tag is secured to the dosage of the medication.
42. The method of claim 37, comprising receiving by the interrogation unit the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
43. The method of claim 37, comprising receiving by the interrogation unit the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
44. The method of claim 43, comprising receiving by the interrogation unit the unique identity from the tag, wherein the syringe comprises a needle, and wherein the needle comprises at least one contact point coupled to the tag.
45. A patient interface device, comprising: a housing defining an outer surface, wherein the housing is configured to held by a patient; at least one contact point exposed on the outer surface portion of the housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient; a control module coupled to the at least one contact point, wherein the at least one contact point is coupled to the control module to capture the at least one physiological parameter associated with the patient; and a communication module coupled to the control unit to transmit the at least one physiological parameter to a computer in communication with the communication module.
46. The patient interface device of claim 45, comprising a power source coupled to the control module.
47. The patient interface device of claim 45, comprising an image generation unit to capture an image of the patient, wherein the image generation unit is coupled to the control module and is configured to capture the image of the patient and to transmit the captured image to the control module.
48. The patient interface device of claim 47, wherein the control module is operative to activate the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient.
49. A method, comprising: contacting by a patient at least one contact point exposed on an outer surface portion of a housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient; capturing by a control module coupled to the at least one contact point at least one physiological parameter associated with the patient; and transmitting by a communication module coupled to the control unit the at least one physiological parameter to a computer in communication with the communication module.
50. The method of claim 49, comprising: scanning by the at least contact point a fingerprint associated with the patient; confirming by the control module fingerprint information.
51. The method of claim 49, comprising: capturing an image of the patient by an image generation unit, wherein the image generation unit is coupled to the control module; and transmitting by the image generation unit the captured image of the patient to the control module.
52. The method of claim 51, comprising activating by the control module the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient.
Description
CROSS REFERENCE To RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Patent Application No. 61/346,035 entitled "Tracking and Delivery Confirmation of Pharmaceutical Products" and filed on May 19, 2010, which is herein entirely incorporated by reference.
FIELD OF THE INVENTION
[0002] The present invention is related to electronic systems for tracking products from manufacturer to consumer and, more specifically, to multiple devices including a mobile communication unit and an identifiable tag that is associated with a pharmaceutical product to confirm source of the dosage, the proper type and amount of dose, and delivery to the patient.
INTRODUCTION
[0003] In many instances there is a need for tracking pharmaceutical products from the manufacturer to the patient. In so doing, the provider or dispenser of the pharmaceutical products is able to confirm that the pharmaceutical products come from a reliable source and that the pharmaceutical products are not counterfeit. Additionally, in many remote areas, where access to medical facilities is limited, it is often the case wherein the provider is not able to read or understand information provided on the label. In such situations, the provider needs a simple and effective way to identify the patient and determine what medication should be delivered, especially when there are various medications from which to choose.
[0004] Thus, what is needed is a system and method that can track pharmaceutical products from the manufacturer along the supply chain to the patient as well as confirm actual delivery of the pharmaceutical products to the patient.
SUMMARY
[0005] The present disclosure includes a system for tracking pharmaceutical products from manufacturer to the patient. The present invention also includes systems for confirming delivery of the pharmaceutical products to the patient; the delivery confirmation includes systems and methods for confirming delivery of the right dose and the correct quantity. The present invention includes systems that include circuitry and components that can function within certain environments that include a conducting fluid. One example of such an environment is outside or inside a container that houses the conducting fluid, such as a sealed bag with a solution, which includes an IV bag. Another example is within the body of a living organism, such as an animal or a human. According to various aspects of the present invention, part of the system is ingestible or injectible and, hence, can be taken by or injected to or inhaled by the patient.
[0006] In one aspect, a computer system is provided to track a medication from manufacturer to a patient, to enable a care provider to determine the origin of the medication and to confirm that at least one of the right type and the right dosage of the medication was delivered to the patient. The computer system comprising: a processor operative to: receive a unique identity of an identifier device secured to a dosage of medication, wherein the identifier device comprises the unique identity that is associated with at least one of the manufacturer and the medication; receive information from a patient interface device in communication with the computer and the patient, wherein at the time the patient takes the medication, the patient interface device is operative to detect the unique identity associated with the identifier device and confirm delivery of the medication to the patient; and receive the detected unique identity associated with the identifier device detected via the patient interface device and the confirmation of delivery of the medication to the patient via the patient interface device; wherein the processor is operative to confirm the origin of the medication and the type of the medication based at least on the unique identity of the identifier device.
[0007] In another aspect of the computer system, the processor is operative to interrogate the identifier device.
[0008] In another aspect of the computer system, the identifier device comprises a tag provided with circuitry, wherein the tag is secured to the dosage of the medication, and wherein the processor is operative to read the unique identity from the tag.
[0009] In another aspect of the computer system, the tag is secured to a dosage of medication in the form of a pill.
[0010] In another aspect of the computer system, the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
[0011] In another aspect of the computer system, the syringe comprises a needle and wherein the needle comprises at least one contact point coupled to the tag.
[0012] In another aspect of the computer system, the processor is operative to communicate with a datacenter database to at least one of retrieve medical information about the patient and provide information related to the medication to be administered to the patient.
[0013] In another aspect of the computer system, the processor is operative to: communicate with at least one of a database or a processing system at the datacenter; transmit information to the processing system; receive information from the processing system, wherein the processing system is operative to access the information in the database of the data center and provide the information to the patient through the computer.
[0014] In another aspect of the computer system, the processor is operative to validate at least one of the type and dosage selected by the care provider prior to dispensing the medication to the patient.
[0015] In another aspect of the computer system, the processor is operative to communicate with an interrogation unit coupled to the identifier device, wherein the interrogation unit is operative to interrogate the identifier device secured to the medication and receive the unique identity of the identifier device to confirm the origin of the medication and the type of the medication, wherein the computer is operative to interrogate the interrogation unit and receive the unique identity of the identifier from the interrogation unit.
[0016] In another aspect of the computer system, the processor is operative to: communicate with a memory of the interrogation unit; and receive the unique identity of the identifier stored in the memory of the interrogation unit.
[0017] In another aspect of the computer system, the processor is operative to receive biometric parameters from the patient interface device, wherein the biometric parameters can identify the patient.
[0018] In another aspect of the computer system, the processor is operative to receive an image of the patient captured by a camera on the patient interface device.
[0019] In another aspect of the computer system, a display provides information associated with the patient, wherein the information comprises any of an image of the patient, medical history of the patient, and a next type and dose of medication to deliver to the patient.
[0020] In one aspect, a computer-implemented method is provided to track a medication from manufacturer to a patient, to enable a care provider to determine the origin of the medication and to confirm that at least one of the right type and the right dosage of medication was delivered to the patient, the computer comprising a processor. The method comprising: receiving by the processor a unique identity of the identifier device secured to a dosage of medication, wherein the identifier device comprises the unique identity that is associated with at least one of the manufacturer and the medication; receiving by the processor information from a patient interface device in communication with the computer and the patient, wherein at the time the patient takes the medication, the patient interface device is operative to detect the unique identity associated with the identifier device and confirm delivery of the medication to the patient; receiving by the processor the detected unique identity associated with the identifier device detected from the patient interface device and the confirmation of delivery of the medication to the patient from the patient interface device; and confirming the origin of the medication and the type of the medication based at least on the unique identity of the identifier device.
[0021] In another aspect, the computer-implemented method comprises interrogating by the processor the identifier device.
[0022] In another, the computer-implemented method comprises receiving by the processor the unique identity from a tag, wherein the identifier device comprises the tag provided with circuitry, wherein the tag is secured to the dosage of the medication, the method.
[0023] In another aspect, the computer-implemented method comprises receiving by the processor the unique identity from the tag, wherein the tag is secured to a dosage of medication in the form of a pill.
[0024] In another aspect, the computer-implemented method comprises receiving by the processor the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
[0025] In another aspect, the computer-implemented method comprises receiving by the processor the unique identity from the tag, wherein the syringe comprises a needle, and wherein the needle comprises at least one contact point coupled to the tag.
[0026] In another aspect, the computer-implemented method comprises at least one of: retrieving by the processor medical information about the patient from a datacenter database; and providing by the processor information related to the medication to be administered to the patient.
[0027] In another aspect, the computer-implemented method comprises transmitting by the processor information to the processing system; receiving by the processor information from the processing system, wherein the processing system is operative to access the information in the database of the data center and provide the information to the patient through the computer.
[0028] In another aspect, the computer-implemented method comprises validating by the processor at least one of the type and dosage selected by the care provider prior to dispensing the medication to the patient.
[0029] In another aspect, the computer-implemented method comprises interrogating by the processor operative an interrogation unit coupled to the identifier device, wherein the interrogation unit is operative to interrogate the identifier device secured to a medication and receive the unique identity of the identifier device to confirm the origin of the medication and the type of the medication; and receiving by the computer the unique identity of the identifier from the interrogation unit.
[0030] In another aspect, the computer-implemented method comprises communicating by the processor with a memory of the interrogation unit; and receiving by the processor the unique identity of the identifier stored in the memory of the interrogation unit.
[0031] In another aspect, the computer-implemented method comprises receiving by the processor biometric parameters from the patient interface device, wherein the biometric parameters can identify the patient.
[0032] In another aspect, the computer-implemented method comprises receiving by the processor an image of the patient captured by a camera on the patient interface device.
[0033] In another aspect, the computer-implemented method comprises displaying on a display coupled to the processor information associated with the patient, wherein the information comprises any of an image of the patient, medical history of the patient, and a next type and dose of medication to deliver to the patient.
[0034] In one aspect, an apparatus is provided. The apparatus comprising: at least one contact point configured to electrically engage an identifier device secured to a medication, wherein the identifier device comprises a unique identity that is associated with at least one of the manufacturer and the medication; an interrogation unit coupled to the at least one contact point, wherein the interrogation unit is operative to interrogate the identifier device to obtain the unique identity; and a communication module coupled to the interrogation unit, wherein the communication module is operative to transmit the unique identification.
[0035] In another aspect of the apparatus, the interrogation unit is operative to read the unique identity associated with the identifier device.
[0036] In another aspect, the communication module is operative to transmit the unique identity associated with the identifier device to a computer in communication with the communication module.
[0037] In another aspect of the apparatus, the apparatus comprises a memory coupled to the interrogation unit to store the unique identity associated with the identifier device.
[0038] In another aspect, the interrogation unit is operative to read the unique identity from a tag provided with circuitry, wherein the tag is secured to the dosage of the medication.
[0039] In another aspect of the apparatus, the interrogation unit is operative to read the unique identity from the tag, wherein the tag is secured to a dosage of medication in the form of a pill.
[0040] In another aspect of the apparatus, the interrogation unit is operative to read the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
[0041] In another aspect of the apparatus, the interrogation unit is operative to read the unique identity from the tag, wherein the syringe comprises a needle and wherein the needle comprises at least one contact point coupled to the tag.
[0042] In one aspect, a method is provided. The method comprising: electrically engaging by an interrogation unit an identifier device secured to a medication with at least one contact point coupled to the interrogation unit, wherein the identifier device comprises a unique identity that is associated with at least one of the manufacturer and the medication; interrogating the identifier device for the unique identity; and transmitting the unique identification by a communication module coupled to the interrogation unit.
[0043] In another aspect, the method comprises reading by the interrogation unit the unique identity associated with the identifier device.
[0044] In another aspect, the method comprises transmitting by the communication module the unique identity associated with the identifier device to a computer in communication with the communication module.
[0045] In another aspect, the method comprises storing by the interrogation unit the unique identity associated with the identifier device in a memory, wherein the memory is coupled to the interrogation unit.
[0046] In another aspect, the method comprises receiving by the interrogation unit the unique identity from a tag, wherein the identifier device comprises the tag provided with circuitry, wherein the tag is secured to the dosage of the medication.
[0047] In another aspect, the method comprises receiving by the interrogation unit the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
[0048] In another aspect, the method comprises receiving by the interrogation unit the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air.
[0049] In another aspect, the method comprises receiving by the interrogation unit the unique identity from the tag, wherein the syringe comprises a needle, and wherein the needle comprises at least one contact point coupled to the tag.
[0050] In one aspect, a patient interface device is provided. The patient interface comprising: a housing defining an outer surface, wherein the housing is configured to held by a patient; at least one contact point exposed on the outer surface portion of the housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient; a control module coupled to the at least one contact point, wherein the at least one contact point is coupled to the control module to capture the at least one physiological parameter associated with the patient; and a communication module coupled to the control unit to transmit the at least one physiological parameter to a computer in communication with the communication module.
[0051] In another aspect of the patient interface device, the patient interface device comprises a power source coupled to the control module.
[0052] In another aspect of the patient interface device, the patient interface device comprises an image generation unit to capture an image of the patient, wherein the image generation unit is coupled to the control module and is configured to capture the image of the patient and to transmit the captured image to the control module.
[0053] In another aspect of the patient interface device, the control module is operative to activate the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient.
[0054] In one aspect, a method is provided. The method comprising: contacting by a patient at least one contact point exposed on an outer surface portion of a housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient; capturing by a control module coupled to the at least one contact point at least one physiological parameter associated with the patient; and transmitting by a communication module coupled to the control unit the at least one physiological parameter to a computer in communication with the communication module.
[0055] In another aspect, the method comprises scanning by the at least contact point a fingerprint associated with the patient; confirming by the control module fingerprint information.
[0056] In another aspect, the method comprises capturing an image of the patient by an image generation unit, wherein the image generation unit is coupled to the control module; and transmitting by the image generation unit the captured image of the patient to the control module.
[0057] In another aspect, the method comprises activating by the control module the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient.
[0058] In one aspect, a patient interface device is provided. The patient interface device comprising: a housing defining an outer surface, wherein the housing is configured to held by a patient; at least one contact point exposed on the outer surface portion of the housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient; a control module coupled to the at least one contact point, wherein the at least one contact point is coupled to the control module to capture the at least one physiological parameter associated with the patient; and a communication module coupled to the control unit to transmit the at least one physiological parameter to a computer in communication with the communication module.
[0059] In another aspect, the patient interface device comprises a power source coupled to the control module.
[0060] In another aspect, the patient interface device comprises an image generation unit to capture an image of the patient, wherein the image generation unit is coupled to the control module and is configured to capture the image of the patient and to transmit the captured image to the control module.
[0061] In another aspect of the patient interface device, the control module is operative to activate the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient.
[0062] In one aspect, a method is provided. The method comprising: contacting by a patient at least one contact point exposed on an outer surface portion of a housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient; capturing by a control module coupled to the at least one contact point at least one physiological parameter associated with the patient; and transmitting by a communication module coupled to the control unit the at least one physiological parameter to a computer in communication with the communication module.
[0063] In another aspect, the method comprises scanning by the at least contact point a fingerprint associated with the patient; confirming by the control module fingerprint information.
[0064] In another aspect, the method comprises capturing an image of the patient by an image generation unit, wherein the image generation unit is coupled to the control module; and transmitting by the image generation unit the captured image of the patient to the control module.
[0065] In another aspect, the method comprises activating by the control module the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient.
[0066] Notwithstanding the claims and the above aspects of the present invention, in various other aspects, the present invention also may be defined by the following clauses: [0067] 1. A computer system to track medication from manufacturer to a patient, to enable a care provider to determine the origin of the medication and to confirm that at least one of the right type and the right dosage of medication was delivered to the patient, the computer system comprising:
[0068] a processor operative to: [0069] receive a unique identity of an identifier device secured to a dosage of medication to confirm the origin of the medication and the type of the medication, wherein the identifier device comprises the unique identity that is associated with at least one of the manufacturer and the medication; [0070] receive information from a patient interface device in communication with the computer and the patient, wherein at the time the patient takes the medication, the patient interface device is operative to detect the unique identity associated with the identifier device and confirm delivery of the medication to the patient; and [0071] receive the detected unique identity associated with the identifier device detected from the patient interface device and the confirmation of delivery of the medication to the patient from the patient interface device. [0072] 2. The computer system of clause 1, wherein the processor is operative to interrogate the identifier device. [0073] 3. The computer system of clause 1 or 2, wherein the identifier device comprises a tag provided with circuitry, wherein the tag is secured to the dosage of the medication, and wherein the processor is operative to read the unique identity from the tag. [0074] 4. The computer system of any of the preceding clauses, wherein the tag is secured to a dosage of medication in the form of a pill. [0075] 5. The computer system of any of the preceding clauses 1-3, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air. [0076] 6. The computer system of clause 5, wherein the syringe comprises a needle and wherein the needle comprises at least one contact point coupled to the tag. [0077] 7. The computer system according to any of the preceding clauses wherein the processor is operative to communicate with a datacenter database to at least one of retrieve medical information about the patient and provide information related to the medication to be administered to the patient. [0078] 8. The computer system of clause 7, wherein the processor is operative to:
[0079] communicate with at least one of a database or a processing system at the datacenter;
[0080] transmit information to the processing system;
[0081] receive information from the processing system, wherein the processing system is operative to access the information in the database of the data center and provide the information to the patient through the computer. [0082] 9. The computer system according to any of the preceding clauses wherein the processor is operative to validate at least one of the type and dosage selected by the care provider prior to dispensing the medication to the patient. [0083] 10. The computer system according to any of the preceding clauses wherein the processor is operative to communicate with an interrogation unit coupled to the identifier device, wherein the interrogation unit is operative to interrogate the identifier device secured to the medication and receive the unique identity of the identifier device to confirm the origin of the medication and the type of the medication, wherein the computer is operative to interrogate the interrogation unit and receive the unique identity of the identifier from the interrogation unit. [0084] 11. The computer system of clause 10, wherein the processor is operative to:
[0085] communicate with a memory of the interrogation unit; and
[0086] receive the unique identity of the identifier stored in the memory of the interrogation unit. [0087] 12. The computer system according to any of the preceding clauses wherein the processor is operative to receive biometric parameters from the patient interface device, wherein the biometric parameters can identify the patient, or wherein the processor is operative to receive an image of the patient captured by a camera on the patient interface device. [0088] 13. The computer system according to any of the preceding clauses comprising a display to provide information associated with the patient, wherein the information comprises any of an image of the patient, medical history of the patient, and a next type and dose of medication to deliver to the patient. [0089] 14. A computer-implemented method to track medication from manufacturer to a patient, which preferably uses a computer system according to any of the preceding clauses, to enable a care provider to determine the origin of the medication and to confirm that at least one of the right type and the right dosage of medication was delivered to the patient, the computer comprising a processor, the method comprising:
[0090] receiving by the processor a unique identity of the identifier device secured to a dosage of medication to confirm the origin of the medication and the type of the medication, wherein the identifier device comprises the unique identity that is associated with at least one of the manufacturer and the medication;
[0091] receiving by the processor information from a patient interface device in communication with the computer and the patient, wherein at the time the patient takes the medication, the patient interface device is operative to detect the unique identity associated with the identifier device and confirm delivery of the medication to the patient; and
[0092] receiving by the processor the detected unique identity associated with the identifier device detected from the patient interface device and the confirmation of delivery of the medication to the patient from the patient interface device. [0093] 15. The computer-implemented method of clause 15, comprising interrogating by the processor the identifier device. [0094] 16. The computer-implemented method of clause 14 or 15, comprising receiving by the processor the unique identity from a tag, wherein the identifier device comprises the tag provided with circuitry, wherein the tag is secured to the dosage of the medication, the method. [0095] 17. The computer-implemented method of clause 16, comprising receiving by the processor the unique identity from the tag, wherein the tag is secured to a dosage of medication in the form of a pill or wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air, preferably comprising receiving by the processor the unique identity from the tag, wherein the syringe comprises a needle, and wherein the needle comprises at least one contact point coupled to the tag. [0096] 18. The computer-implemented according to any of the clauses 14-17 comprising at least one of:
[0097] retrieving by the processor medical information about the patient from a datacenter database; and
[0098] providing by the processor information related to the medication to be administered to the patient. [0099] 19. The computer-implemented method of clause 18, comprising:
[0100] transmitting by the processor information to the processing system;
[0101] receiving by the processor information from the processing system, wherein the processing system is operative to access the information in the database of the data center and provide the information to the patient through the computer. [0102] 20. The computer-implemented method according to any of the clauses 14-19, comprising:
[0103] validating by the processor at least one of the type and dosage selected by the care provider prior to dispensing the medication to the patient. [0104] 21. The computer-implemented method according to any of the clauses 14-20 comprising:
[0105] interrogating by the processor operative an interrogation unit coupled to the identifier device, wherein the interrogation unit is operative to interrogate the identifier device secured to the medication and receive the unique identity of the identifier device to confirm the origin of the medication and the type of the medication; and
[0106] receiving by the computer the unique identity of the identifier from the interrogation unit. [0107] 22. The computer-implemented method of clause 21, comprising:
[0108] communicating by the processor with a memory of the interrogation unit; and
[0109] receiving by the processor the unique identity of the identifier stored in the memory of the interrogation unit. [0110] 23. The computer-implemented method according to any of the clauses 14-22 comprising:
[0111] receiving by the processor biometric parameters from the patient interface device, wherein the biometric parameters can identify the patient. [0112] 24. The computer-implemented method according to any of the clauses 14-23 comprising:
[0113] receiving by the processor an image of the patient captured by a camera on the patient interface device. [0114] 25. The computer-implemented method according to any of the clauses 14-24 comprising:
[0115] displaying on a display coupled to the processor information associated with the patient, wherein the information comprises any of an image of the patient, medical history of the patient, and a next type and dose of medication to deliver to the patient. [0116] 26. An apparatus, comprising:
[0117] at least one contact point configured to electrically engage an identifier device secured to a medication, wherein the identifier device comprises a unique identity that is associated with at least one of the manufacturer and the medication;
[0118] an interrogation unit coupled to the at least one contact point, wherein the interrogation unit is operative to interrogate the identifier device to obtain the unique identity; and
[0119] a communication module coupled to the interrogation unit, wherein the communication module is operative to transmit the unique identification. [0120] 27. The apparatus of clause 26, wherein the interrogation unit is operative to read the unique identity associated with the identifier device. [0121] 28. The apparatus of clause 26 or 27 wherein the communication module is operative to transmit the unique identity associated with the identifier device to a computer in communication with the communication module. [0122] 29. The apparatus according to any of the clauses 26-28 comprising a memory coupled to the interrogation unit to store the unique identity associated with the identifier device. [0123] 30. The apparatus according to any of the clauses 26-29 wherein the interrogation unit is operative to read the unique identity from a tag provided with circuitry, wherein the tag is secured to the dosage of the medication, preferably wherein the interrogation unit is operative to read the unique identity from the tag, wherein the tag is secured to a dosage of medication in the form of a pill, or wherein the interrogation unit is operative to read the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air, preferably wherein the syringe comprises a needle and wherein the needle comprises at least one contact point coupled to the tag. [0124] 31. A method, comprising:
[0125] electrically engaging by an interrogation unit an identifier device secured to a medication with at least one contact point coupled to the interrogation unit, wherein the identifier device comprises a unique identity that is associated with at least one of the manufacturer and the medication;
[0126] interrogating the identifier device for the unique identity; and
[0127] transmitting the unique identification by a communication module coupled to the interrogation unit. [0128] 32. The method of clause 31, comprising reading by the interrogation unit the unique identity associated with the identifier device. [0129] 33. The method of clause 31 or 32 comprising transmitting by the communication module the unique identity associated with the identifier device to a computer in communication with the communication module. [0130] 34. The method according to any of the clauses 31-33 comprising storing by the interrogation unit the unique identity associated with the identifier device in a memory, wherein the memory is coupled to the interrogation unit. [0131] 35. The method according to any of the clauses 31-34 comprising receiving by the interrogation unit the unique identity from a tag, wherein the identifier device comprises the tag provided with circuitry, wherein the tag is secured to the dosage of the medication. [0132] 36. The method of clause 35, comprising receiving by the interrogation unit the unique identity from the tag, wherein the tag is secured to a syringe configured for delivery of the dosage of medication, wherein the syringe comprises a safety component which is activated upon exposure to air, preferably wherein the syringe comprises a needle, and wherein the needle comprises at least one contact point coupled to the tag. [0133] 37. A patient interface device, comprising:
[0134] a housing defining an outer surface, wherein the housing is configured to held by a patient;
[0135] at least one contact point exposed on the outer surface portion of the housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient;
[0136] a control module coupled to the at least one contact point, wherein the at least one contact point is coupled to the control module to capture the at least one physiological parameter associated with the patient; and
[0137] a communication module coupled to the control unit to transmit the at least one physiological parameter to a computer in communication with the communication module. [0138] 38. The patient interface device of clause 37, comprising a power source coupled to the control module. [0139] 39. The patient interface device of clause 37 or 38 comprising an image generation unit to capture an image of the patient, wherein the image generation unit is coupled to the control module and is configured to capture the image of the patient and to transmit the captured image to the control module. [0140] 40. The patient interface device according to any of the clauses 37-39 wherein the control module is operative to activate the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient. [0141] 41. A method, comprising:
[0142] contacting by a patient at least one contact point exposed on an outer surface portion of a housing, wherein the at least one contact point is positioned to be physically contacted by the patient to detect biometric information associated with the patient, wherein the at least one contact point is configured to detect at least one physiological parameter associated with the patient;
[0143] capturing by a control module coupled to the at least one contact point at least one physiological parameter associated with the patient; and
[0144] transmitting by a communication module coupled to the control unit the at least one physiological parameter to a computer in communication with the communication module. [0145] 42. The method of clause 41, comprising:
[0146] scanning by the at least contact point a fingerprint associated with the patient;
[0147] confirming by the control module fingerprint information. [0148] 43. The method of clause 41 or 42, comprising:
[0149] capturing an image of the patient by an image generation unit, wherein the image generation unit is coupled to the control module; and
[0150] transmitting by the image generation unit the captured image of the patient to the control module. [0151] 44. The method according to any of the clauses 41-43 comprising activating by the control module the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient. [0152] 45. System for tracking a product from a manufacturer along a supply chain to a user, the system comprising: [0153] Identifying means associated with the product for identifying the product, [0154] Interrogating means for interrogating the identifying means. [0155] 46. System according to clause 45 wherein the product is a medicine and the user is a patient, wherein the system enables a care provider to determine the origin of the medicine. [0156] 47. System according to clause 45 or 46, further comprising patient communication means for communicating identity of the medicine to the patient. [0157] 48. System according to any of the preceding clauses 45-47 wherein the identifying means comprises a device secured to the product. [0158] 49. System according to clause 48, wherein the identifying means has a unique identity associated with the manufacturer and/or the product. [0159] 50. System according to any of the preceding clauses 46-49 wherein the system confirms that a correct dosage and/or correct type of medication was delivered to the patient. [0160] 51. System according to any of the preceding clauses 45-50 wherein the interrogating means comprises a computer system which reads the unique identity of the identifying means. [0161] 52. System according to clause 51 wherein the computer system communicates with a database to retrieve user information and/or product information. [0162] 53. System according to clauses 52 wherein the user information is patient information, and wherein the product information is information related to the medicament to be administered to the patient. [0163] 54. System according to any of the clauses 51-53 wherein the computer system validates at least one of the type and dosage prior to dispensing. [0164] 55. System according to any of the preceding clauses 47-54, wherein the patient communication means is configured to be in physical communication with the patient at the time the patient takes the medication, and is in communication with the computer system, the patient communication means being a personal device which detects the unique identity of the identifying means and confirms delivery of the product. [0165] 56. System according to any of the preceding clauses 45-55 wherein the identifying means is a tag provided with circuitry, which tag is secured to a pill, and which circuitry is broken if the tag is removed from the pill. [0166] 57. System according to any of the preceding clauses 45-55 wherein the identifying means is a tag, which tag is secured to a syringe, wherein the tag and/or syringe comprises a safety component which is activated on exposure to air. [0167] 58. System according to clauses 56 or 57 wherein the tag cannot be interrogated if the medicament is not delivered within a pre-determined time period. [0168] 59. System according to clauses 57 or 58 wherein a needle of the syringe includes a contact point connected to the tag. [0169] 60. An interface device for confirming delivery of medication to a patient, preferably for use in a system according to any of the preceding clauses 45-59, the device comprising: a control module, wherein the control modules includes a communication module and a memory unit;
[0170] a contact point to detect at least one physiological parameter associated with the patient, wherein the contact point is connected to the control module and captures at least one physiological parameter associated with the patient and transmits that information to the patient; and
[0171] an image generation unit to capture the patient's image, wherein the image generation unit is connected to the control module and captures the image of the patient and transmits the image to the control module,
[0172] wherein the control module activates the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient. [0173] 61. System according to any of the preceding clauses 45-59, further comprising a device according to clause 60. [0174] 62. Method for tracking a product from a manufacturer along a supply chain to a user comprising the steps of attaching identifying means to a product, and interrogating the identifying means. [0175] 63. Method according to clause 62 further comprising the step of confirming receipt of the product to the user. [0176] 64. Method according to clause 62 or 63 using a system, device or apparatus according to any of the preceding. [0177] 65. A pill comprising a tag, which tag is provided with circuitry. [0178] 66. A syringe comprising a tag, wherein the tag and/or syringe comprises a safety component which is activated on exposure to air. [0179] 67. System to track medication from manufacturer to a patient, to enable a care provider to determine the origin of the medication and to confirm that at least one of the right type and the right dosage of medication was delivered to the patient, the system comprising: an identifier device secured to the medication, wherein the identifier device has a unique identity that is associated with at least one of the manufacturer and the medication;
[0180] a computer system for interrogating the identifier device,
[0181] wherein the computer system reads the unique identity of the identifier device to confirm the origin of the medication and the type of the medication and
[0182] wherein the computer system communicates with a database to at least one of retrieve medical information about the patient and provide information related to the medication to be administered to the patient and
[0183] wherein the computer system validates at least one of the type and dosage selected by the care provider prior to dispensing to the patient; and
[0184] a personal device in communication with the computer system, wherein the personal device is configured to be in physical communication with the patient at the time the patient takes the medication to detect the unique identity associated with the identifier device and confirm delivery of the medication to the patient. [0185] 68. Method to track and guide a care provider to deliver a proper type and dose of medication to a patient such that a care provider confirms that the medication is traceable to a known origin, the method comprising the steps of:
[0186] attaching an identifiable tag that produces a unique signature to the medication at the origin when the medication is created;
[0187] interrogating the tag at about the time of delivery to the patient to determine if the medication is the original medication and to confirm that that the correct medication has been selected for the patient; and
[0188] confirming that the patient has taken or received the medication through detecting the identifiable tag's unique signature through the patient's body based on communication of the identifiable tag with the patient. [0189] 69. System to track a deliverable to a user, the system comprising:
[0190] an identifier device secured to the deliverable, wherein the identifier device has a unique identity that is associated with the deliverable;
[0191] a computer system for interrogating the identifier device,
[0192] wherein the computer system identifies the identifier device to confirm information associated with the deliverable and
[0193] wherein the computer system communicates with a database to at least one of retrieve information associated with the user and provide a care provider with the information associated with the deliverable to be administered to the user and
[0194] wherein the computer system validates the information associated with the deliverable provided by the care provider prior to administration to the user; and
[0195] a personal device in communication with the computer system, wherein the personal device is configured to be held by the user at the time the user is administered the deliverable to detect the unique identity associated with the identifier device and to confirm delivery of the deliverable to the user. [0196] 70. Method to track and guide a care provider to deliver a deliverable to a user such that the care provider can confirm that the deliverable is traceable to a known origin, the method comprising the steps of:
[0197] providing an identifiable tag that produces a unique signature, the identifiable tag physically associated with the deliverable;
[0198] interrogating an identifiable tag associated with the deliverable at about the time of delivery to the user to determine if the deliverable is the intended deliverable and confirm that the care provider has selected the correct deliverable; and
[0199] confirming that the user has been administered the deliverable through detecting the identifiable tag's unique signature based on contact of the identifiable tag with the user. [0200] 71. Interface device for confirming delivery of medication to a patient, the device comprising:
[0201] a control module, wherein the control modules includes a communication module and a memory unit;
[0202] a contact point to detect at least one physiological parameter associated with the patient, wherein the contact point is connected to the control module and captures at least one physiological parameter associated with the patient and transmits that information to the patient; and
[0203] an image generation unit to capture the patient's image, wherein the image generation unit is connected to the control module and captures the image of the patient and transmits the image to the control module,
[0204] wherein the control module activates the image generation unit to capture the image of the patient when the contact point detects there is a change in the physiological parameter of the patient during the time the patient is in contact with the contact point and when the control module detects a current flow through the patient.
BRIEF DESCRIPTION OF THE FIGURES
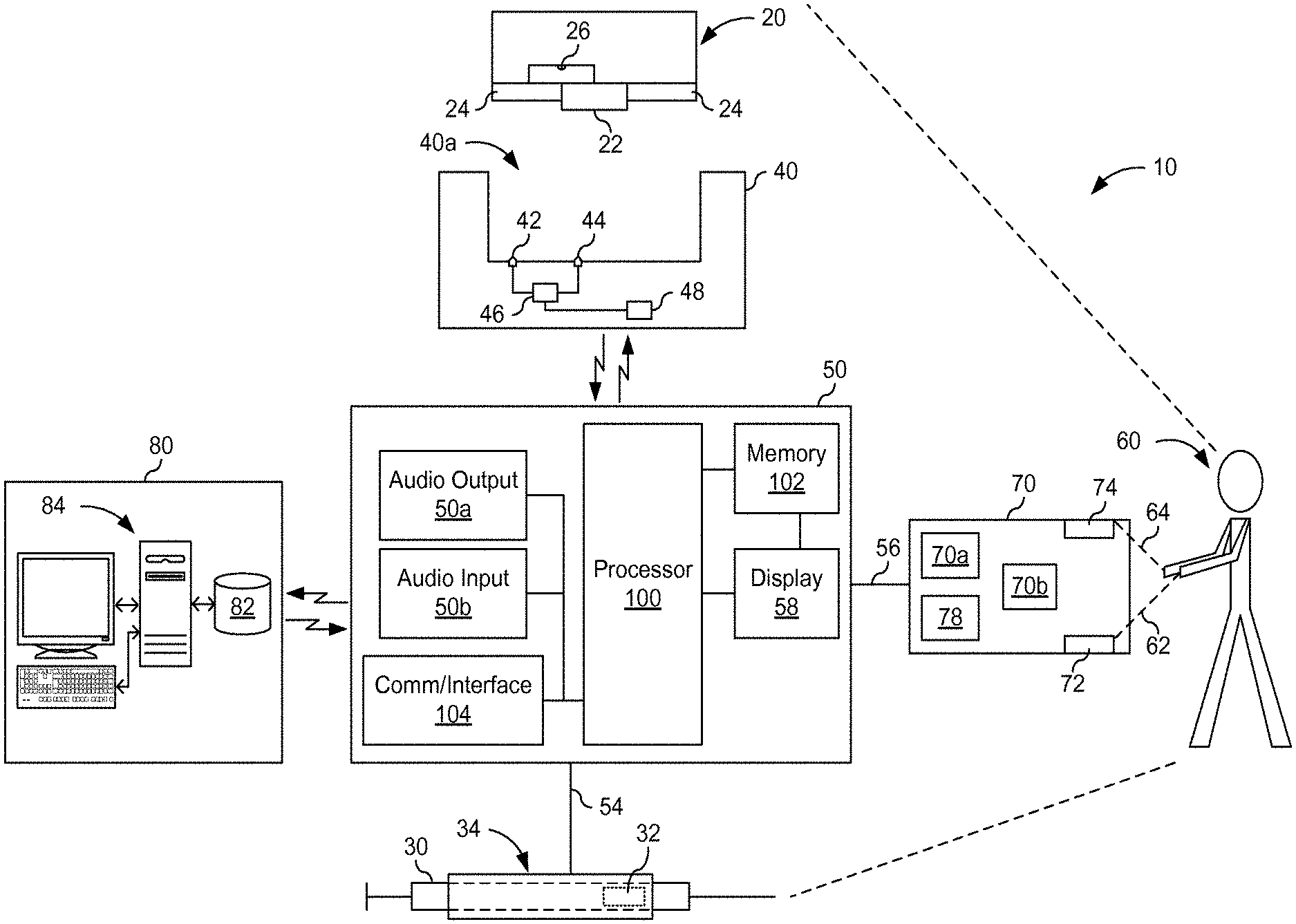
[0205] FIG. 1 shows a system for tracking medication from origin to patient as well as confirmation of delivery of the correct type and dose of medication.
[0206] FIG. 2A shows a needle of a syringe used for delivery of medication in accordance with one aspect of the present invention.
[0207] FIG. 2B shows a needle of a syringe used for delivery of medication in accordance with another aspect of the present invention.
[0208] FIG. 2C shows a cut-away section of a sleeve for holding the syringe of FIG. 2A and FIG. 2B in accordance with an aspect of the present invention.
[0209] FIG. 3 shows a functional block diagram representation of an identity confirmation device as used in FIG. 1 in accordance with one aspect of the present invention.
[0210] FIG. 4 shows one aspect of a tag.
DETAILED DESCRIPTION
[0211] Referring now to FIG. 1, a system 10 includes a device 40, a mobile computer 50, and a patient identity confirmation device 70. The system 10 communicates with a central data center 80. In accordance with various aspects of the present invention, there are at least two methods disclosed for delivery of medication: a pill 20 and a syringe 30. However, the scope of the present invention is not limited by delivery methods used. For example, the medication may be delivered by a patch, an inhaler, or an ointment. Further, the term "pill" includes various form factors of ingestible medicaments and carriers, e.g., capsules, gel caps, placebos, over capsulation carriers or vehicles, etc. In the event a patch is used, then the tag can be secured to the patch. If an ointment is used, then the tag may be included with ointment and delivery and tracking can be confirmed once the ointment is applied. At the origin or location of manufacturing of the medication, an identifiable tag is associated with the medication regardless of the type of medication or method of delivery/dispensing of the medication to the patient. The identifiable tag is similar to an ingestible event marker or an ionic emission module (IEM). The IEM can be ingested and is capable of providing an identifiable signature and is unique. The IEM is disclosed in greater detail in U.S. patent application Ser. No. 12/564,017 entitled COMMUNICATION SYSTEM WITH PARTIAL POWER SOURCE filed on Sep. 21, 2009, and published as 2010-0081894 A1 dated Apr. 1, 2010, which is incorporated herein by reference in its entirety.
[0212] In accordance with other aspects of the present invention, the identifiable tags may include bar codes on packaging that uniquely identify the medication in the package, which package includes tamper proof seals. Thus the care provider would use the mobile computer 50 to scan the bar code or capture an image of the bar code that is transmitted to the data center 80 for confirmation.
[0213] With respect to the pill 20, an identifiable tag 22 is shown secured to one side of the pill 20. Surrounding the tag 22 is a conducting surface 24. One surface of the tag 22 is exposed and the other surface of the tag 22 is electrically coupled to the surface 24 via a conduction path 26. The conducting surface 24 can be created using a variety of techniques, e.g., any ingestible conducting ink technology. To prevent removal of the tag 22 from the pill 20, the tag 22 is secured in such a way to cause destruction of the circuitry of the tag 22 if attempts are made to separate it from the medication. Additionally, including the conducting surface 24 around the tag 22 further prevents tampering with and removal of the tag 22. Specifically, if the tag 22 is removed from the pill 20, then the conducting surface 24 and the path 26 will be separated from the tag 22 thereby preventing interrogation of the tag 22 by a device 40.
[0214] The device 40 includes contact points 42 and 44, each of which is connected to an interrogation unit 46. The device 40 also includes a power source, which is not shown. The interrogation unit 46 is connected to a communication module 48. To confirm the origin of the pill 20 as well the type and dose, the pill 20 is placed in the opening 40a and comes into contact with contact points 42 and 44. The interrogation unit 46 can then read the identity of the tag 22. The identity of the tag 22 is then communicated to a mobile computer 50. The communication link 52 between the device 40 and the mobile computer 50 is any wireless communication link, such as Bluetooth.TM. technology or similar wireless technology. In another aspect, the device 40 may be part of the computer 50 as well.
[0215] According to another aspect of the present invention, the device 40 may include a memory for storing the information for later transmission or communication as well as confirmation in the event the communication link between the device 40 and the mobile computer 50 is interrupted or lost.
[0216] Additionally, in accordance with another aspect of the present invention, the device 40 may be directly connected to the mobile computer 50 with a physical link (not shown), such as a wired connection, such as a Universal Serial Bus (USB). The mobile computer 50 is then able to confirm the origin of the medication and other information associated with correct delivery of the type and dose of medication to a patient 60. The computer 50 is shown with a display 58. The display 58 can provide information such as an image of the patient, patient's medical history, the next type and dose of medication to deliver to the patient. In accordance with various aspects of the present invention, the mobile computer 50 may be any device that includes a display and wireless communication capabilities, such as a mobile communication device, a mobile phone, or a laptop computer with wireless communication unit. Furthermore, the mobile computer 50 includes audio output 50a and an audio input 50b, which can be used together to simulate a modem and create a data channel for communication. Also, the mobile computer 50 is capable of communication using various communications protocol(s), e.g., Short Message Service (SMS) such that information is transmitted to a destination through mobile phone communication channels.
[0217] With respect to the syringe 30, an identifiable tag 32 is secured to the syringe 30. To ensure that the tag 32 cannot be removed from the syringe 30 and reattached to a different syringe, the syringe and/or tag include(s) a safety component, e.g., the tag 32 includes a layer that is activated once exposed to air, such as a zinc battery. Thus, upon manufacture, each syringe 30 is secured within a hermetically sealed package to preserve the tag 32. Once the package is opened, the tag 32 is exposed to the air and, hence, activated. Thus, if the package is opened and the medication is not delivered within a short period of time, such a few minutes, then the tag 32 is deactivated and cannot be interrogated later. In accordance with one aspect of the present invention, the layer of the tag 32 is a zinc-based battery that is activated upon contact with the surrounding air and, hence, powers up the tag 32.
[0218] Once the syringe 30 is removed from the package and the care provider is ready to deliver the medication to the patient, the syringe 30 is placed inside the sleeve 34. The sleeve 34 can detect and capture the identity of the tag 32. The sleeve 34 transmits the information associated with the tag 32 secured to the syringe 30 via a physical connection 54 between the mobile computer 50 and the sleeve 34. In accordance with another aspect of the present invention, the sleeve 34 may be in communication with the mobile computer 50 via a wireless connection, not shown. As the needle of the syringe 30 comes into contact with the patient 60, then the circuit is completed as shown through the patient identity confirmation device 70.
[0219] Referring now to FIG. 2A and FIG. 2B, in accordance with another aspect of the present invention, the needle of the syringe 30 may include specific contact points 36 that are connected to the tag 32 via connection 38. The contact points 36 are positioned along at the end of the syringe 30 or on the needle such that contact with the skin of the patient completes a circuit that is then used to activate the tag 32 to transmit a unique signature or identity that represents delivery of the medication to the patient 60.
[0220] Referring now to FIG. 2C, the contact points 36 are electrically coupled to the tag 32. The tag 32 is electrically coupled to the sleeve 34 as the syringe 30 is placed inside the sleeve 34 through a contact point 34a. The contact point 34a makes contact with the tag 32 to allow electrical signals to pass between the tag 32, through the connection 54, and the mobile computer 50. A stop tab 34b is positioned within the sleeve 34 to ensure proper alignment of the contact point 34a with the tag 32.
[0221] Referring now to FIG. 1 and FIG. 3, the patient identity confirmation device 70 is shown connected to the mobile computer 50 via a connection or link 56. The patient identity confirmation device 70 includes thumb pads or contact points 72 and 74 exposed on the surface of the patient identity confirmation device 70. The contact points 72 and 74 may be any form of biometric detection contacts in accordance with various aspects of the present invention. For example, the contact point may scan and confirm fingerprint information. In accordance with another aspect of the present invention, the contact points may detect the physiological parameters of the patient, such as temperature, or other biometric parameters that can identify the patient, including DNA. The contact points 72 and 74 are electrically coupled to a control unit 70a within the patient identity confirmation device 70. In accordance with one aspect of the present invention, a power source 70b is electrically coupled to the control unit 70a, so that the patient identity confirmation device 70 is self powered. In accordance with another aspect of the present invention, the patient identity confirmation device 70 can receive power from the mobile computer through the connection 56.
[0222] In accordance with one aspect of the present invention, the patient identity confirmation device 70 includes a camera 78 for capturing an image of the patient that is transmitted to the mobile computer 50. The camera 78 is electrically connected to the control unit 70a. The control unit 70a controls the activation of the camera 78 once the control unit 70a detected that a circuit path is complete, which indicates that the patient 60 is receiving the medication. More specifically, the patient 60 holds the patient identity confirmation device 70 using at least one of the contact points 72 and 74 to create a physical connection between the patient identity confirmation device 70 and the patient 60 at one of the limbs 62 and 64, e.g., right and left thumbs. As the patient 60 holds the patient identity confirmation device 70 and the syringe 30 comes into contact with the patient 60, then a circuit is completed. More specifically, the contact points 36 come into contact with the skin of the patient 60. The contact points 36 are electrically coupled to the tag 32 and the tag 32 is electrically coupled to the sleeve 34. The sleeve 34 is electrically coupled to the mobile computer 50 and the mobile computer 50 is electrically coupled to the patient identity confirmation device 70. Thus, a complete circuit is formed through the patient 60 once the patient hold the patient identity confirmation device 70 and the needle of the syringe 30 comes into contact with the skin of the patient 60.
[0223] As indicated above the contact points 72 and 74 can detect physiological parameters of the patient through the physical contact, including a change in heart rate, which is typical when the needle of the syringe 30 pierces the skin of patient 60. This change in physiological parameter of the patient 60 can be a trigger that is used by the control unit 70a to activate the camera to capture an image of the patient 60. The information associated with the time and date of delivery as well as the image of the patient 60 is sent to the mobile computer 50. The mobile computer 50 can record the delivery of the medication to the patient 60. Additionally, the mobile computer 50 can scan or interrogate the tag 32 to confirm the dose and type of medication delivered. Thereafter, the zinc-based layer of the tag 32 expires and the tag 32 cannot be used again. In accordance with another aspect of the present invention, the syringe 30 may be a single use syringe such that once the plunger is depressed, it cannot be retracted or extracted.
[0224] In various aspects the identifiable tags 22, 32, similar to an ingestible event marker or an ionic emission module (IEM), may be implemented in accordance with the system 120 shown in FIG. 4. The system 120 can be used in association with any medication product, as mentioned above, to determine the origin of the medication and to confirm that at least one of the right type and the right dosage of medication was delivered to the patient and in some aspects to determine when a patient takes the pharmaceutical product. The scope of the present disclosure, however, is not limited by the environment and the product that is used with the system 120. For example, the system may be activated either in wireless mode, in galvanic mode by placing the system 120 within a capsule and the placing the capsule within the conducting fluid, or a combination thereof, or exposing the system 120 to air. Once placed in a conducting fluid, for example, the capsule would dissolve over a period of time and release the system 120 into the conducting fluid. Thus, in one aspect, the capsule would contain the system 120 and no product. Such a capsule may then be used in any environment where a conducting fluid is present and with any product. For example, the capsule may be dropped into a container filled with jet fuel, salt water, tomato sauce, motor oil, or any similar product. Additionally, the capsule containing the system 120 may be ingested at the same time that any pharmaceutical product is ingested in order to record the occurrence of the event, such as when the product was taken.
[0225] In the specific example of the system 120 combined with a medication or pharmaceutical product, as the product or pill is ingested, or exposed to air, the system 120 is activated in galvanic mode. The system 120 controls conductance to produce a unique current signature that is detected, thereby signifying that the pharmaceutical product has been taken. When activated in wireless mode, the system controls modulation of capacitive plates to produce a unique voltage signature associated with the system 120 that is detected.
[0226] In one aspect, the system 120 includes a framework 122. The framework 122 is a chassis for the system 120 and multiple components are attached to, deposited upon, or secured to the framework 122. In this aspect of the system 120, a digestible material 124 is physically associated with the framework 122. The material 124 may be chemically deposited on, evaporated onto, secured to, or built-up on the framework all of which may be referred to herein as "deposit" with respect to the framework 122. The material 124 is deposited on one side of the framework 122. The materials of interest that can be used as material 124 include, but are not limited to: Cu, CuCl, or CuI. The material 124 is deposited by physical vapor deposition, electrodeposition, or plasma deposition, among other protocols. The material 124 may be from about 0.05 to about 500 .mu.m thick, such as from about 5 to about 100 .mu.m thick. The shape is controlled by shadow mask deposition, or photolithography and etching. Additionally, even though only one region is shown for depositing the material, each system 120 may contain two or more electrically unique regions where the material 124 may be deposited, as desired.
[0227] At a different side, which is the opposite side as shown in FIG. 4, another digestible material 126 is deposited, such that the materials 124 and 126 are dissimilar and insulated from each other. Although not shown, the different side selected may be the side next to the side selected for the material 124. The scope of the present disclosure is not limited by the side selected and the term "different side" can mean any of the multiple sides that are different from the first selected side. In various aspects, the dissimilar material may be located at different positions on a same side. Furthermore, although the shape of the system is shown as a square, the shape may be any geometrically suitable shape. The materials 124 and 126 are selected such that they produce a voltage potential difference when the system 120 is in contact with conducting liquid, such as body fluids. The materials of interest for material 126 include, but are not limited to: Mg, Zn, or other electronegative metals. As indicated above with respect to the material 124, the material 126 may be chemically deposited on, evaporated onto, secured to, or built-up on the framework. Also, an adhesion layer may be necessary to help the material 126 (as well as material 124 when needed) to adhere to the framework 122. Typical adhesion layers for the material 126 are Ti, TiW, Cr or similar material. Anode material and the adhesion layer may be deposited by physical vapor deposition, electrodeposition or plasma deposition. The material 126 may be from about 0.05 to about 500 .mu.m thick, such as from about 5 to about 100 .mu.m thick. However, the scope of the present disclosure is not limited by the thickness of any of the materials nor by the type of process used to deposit or secure the materials to the framework 122.
[0228] According to the disclosure set forth, the materials 124 and 126 can be any pair of materials with different electrochemical potentials. Additionally, in the embodiments wherein the system 120 is used in-vivo, the materials 124 and 126 may be vitamins that can be absorbed. More specifically, the materials 124 and 126 can be made of any two materials appropriate for the environment in which the system 120 will be operating. For example, when used with an ingestible product, the materials 124 and 126 are any pair of materials with different electrochemical potentials that are ingestible. An illustrative example includes the instance when the system 120 is in contact with an ionic solution, such as stomach acids. Suitable materials are not restricted to metals, and in certain embodiments the paired materials are chosen from metals and non-metals, e.g., a pair made up of a metal (such as Mg) and a salt (such as CuCl or CuI). With respect to the active electrode materials, any pairing of substances--metals, salts, or intercalation compounds--with suitably different electrochemical potentials (voltage) and low interfacial resistance are suitable.
[0229] Materials and pairings of interest include, but are not limited to, those reported in TABLE 1 below. In one embodiment, one or both of the metals may be doped with a non-metal, e.g., to enhance the voltage potential created between the materials as they come into contact with a conducting liquid. Non-metals that may be used as doping agents in certain embodiments include, but are not limited to: sulfur, iodine, and the like. In another embodiment, the materials are copper iodine (CuI) as the anode and magnesium (Mg) as the cathode. Aspects of the present disclosure use electrode materials that are not harmful to the human body.
TABLE-US-00001 TABLE 1 Anode Cathode Metals Magnesium, Zinc Sodium (.dagger.), Lithium (.dagger.) Iron Salts Copper salts: iodide, chloride, bromide, sulfate, formate, (other anions possible) Fe.sup.3+ salts: e.g. orthophosphate, pyrophosphate, (other anions possible) Oxygen (.dagger..dagger.) on platinum, gold or other catalytic surfaces Intercalation Graphite with Li, K, Vanadium oxide Manganese oxide compounds Ca, Na, Mg
[0230] Thus, when the system 120 is in contact with the conducting fluid, a current path is formed through the conducting fluid between the dissimilar materials 124 and 126. A control device 128 is secured to the framework 122 and electrically coupled to the materials 124 and 126. The control device 128 includes electronic circuitry, for example control logic that is capable of controlling and altering the conductance between the materials 124 and 126.
[0231] The voltage potential created between the dissimilar materials 124 and 126 provides the power for operating the system as well as produces the current flow through the conducting fluid and the system 120. In one aspect, the system 120 operates in direct current mode. In an alternative aspect, the system 120 controls the direction of the current so that the direction of current is reversed in a cyclic manner, similar to alternating current. As the system reaches the conducting fluid or the electrolyte, where the fluid or electrolyte component is provided by a physiological fluid, e.g., stomach acid, the path for current flow between the dissimilar materials 124 and 126 is completed external to the system 120; the current path through the system 120 is controlled by the control device 128. Completion of the current path allows for the current to flow and in turn a receiver, not shown, can detect the presence of the current and recognize that the system 120 has been activate and the desired event is occurring or has occurred.
[0232] In one embodiment, the two dissimilar materials 124 and 126 are similar in function to the two electrodes needed for a direct current power source, such as a battery. The conducting liquid acts as the electrolyte needed to complete the power source. The completed power source described is defined by the physical chemical reaction between the dissimilar materials 124 and 126 of the system 120 and the surrounding fluids of the body. The completed power source may be viewed as a power source that exploits reverse electrolysis in an ionic or a conduction solution such as gastric fluid, blood, or other bodily fluids and some tissues. Additionally, the environment may be something other than a body and the liquid may be any conducting liquid. For example, the conducting fluid may be salt water or a metallic based paint.
[0233] In certain aspects, the two dissimilar materials 124 and 126 are shielded from the surrounding environment by an additional layer of material. Accordingly, when the shield is dissolved and the two dissimilar materials 124, 126 are exposed to the target site, a voltage potential is generated.
[0234] In certain aspects, the complete power source or supply is one that is made up of active electrode materials, electrolytes, and inactive materials, such as current collectors, packaging. The active materials are any pair of materials with different electrochemical potentials. Suitable materials are not restricted to metals, and in certain embodiments the paired materials are chosen from metals and non-metals, e.g., a pair made up of a metal (such as Mg) and a salt (such as CuI). With respect to the active electrode materials, any pairing of substances--metals, salts, or intercalation compounds--with suitably different electrochemical potentials (voltage) and low interfacial resistance are suitable.
[0235] A variety of different materials may be employed as the materials that form the electrodes. In certain embodiments, electrode materials are chosen to provide for a voltage upon contact with the target physiological site, e.g., the stomach, sufficient to drive the system of the identifier. In certain embodiments, the voltage provided by the electrode materials upon contact of the metals of the power source with the target physiological site is 0.001 V or higher, including 0.01 V or higher, such as 0.1 V or higher, e.g., 0.3 V or higher, including 0.5 volts or higher, and including 1.0 volts or higher, where in certain embodiments, the voltage ranges from about 0.001 to about 10 volts, such as from about 0.01 to about 10 V.
[0236] Referring still to FIG. 4, the dissimilar materials 124 and 126 provide the voltage potential to activate the control device 128. Once the control device 128 is activated or powered up, the control device 128 can alter conductance between the first and second materials 124 and 126 in a unique manner. By altering the conductance between the first and second materials 124 and 126, the control device 128 is capable of controlling the magnitude of the current through the conducting liquid that surrounds the system 120. This produces a unique current signature that can be detected and measured by a receiver (not shown), which can be positioned internal or external to the body. The receiver is disclosed in greater detail in U.S. patent application Ser. No. 12/673,326 entitled BODY-ASSOCIATED RECEIVER AND METHOD filed on Dec. 15, 2009, and published as 2010-0312188 A1 dated Dec. 9, 2010 which is incorporated herein by reference in its entirety. In addition to controlling the magnitude of the current path between the materials, non-conducting materials, membrane, or "skirt" are used to increase the "length" of the current path and, hence, act to boost the conductance path, as disclosed in the U.S. patent application Ser. No. 12/238,345 entitled, "In-Body Device with Virtual Dipole Signal Amplification" filed Sep. 25, 2008, the entire content of which is incorporated herein by reference. Alternatively, throughout the disclosure herein, the terms "non-conducting material," "membrane," and "skirt" are interchangeably used with the term "current path extender" without impacting the scope or the present embodiments and the claims herein. The skirt, shown in portion at 125 and 127, respectively, may be associated with, e.g., secured to, the framework 122. Various shapes and configurations for the skirt are contemplated as within the scope of the various aspects of the present invention. For example, the system 120 may be surrounded entirely or partially by the skirt and the skirt maybe positioned along a central axis of the system 120 or off-center relative to a central axis. Thus, the scope of the present invention as claimed herein is not limited by the shape or size of the skirt. Furthermore, in other embodiments, the dissimilar materials 124 and 126 may be separated by one skirt that is positioned in any defined region between the dissimilar materials 124 and 126.
[0237] The system 120 may be grounded through a ground contact. The system 120 also may include a sensor module. In operation, ion or current paths are established between the first material 124 to the second material 126 and through a conducting fluid in contact with the system 120. The voltage potential created between the first and second materials 124 and 126 is created through chemical reactions between the first and second materials 124/126 and the conducting fluid. In one aspect, the surface of the first material 124 is not planar, but rather an irregular surface. The irregular surface increases the surface area of the material and, hence, the area that comes in contact with the conducting fluid.
[0238] In one aspect, at the surface of the first material 124, there is chemical reaction between the material 124 and the surrounding conducting fluid such that mass is released into the conducting fluid. The term mass as used herein refers to protons and neutrons that form a substance. One example includes the instant where the material is CuCl and when in contact with the conducting fluid, CuCl becomes Cu (solid) and Cl-- in solution. The flow of ions into the conduction fluid is via ion paths. In a similar manner, there is a chemical reaction between the second material 126 and the surrounding conducting fluid and ions are captured by the second material 126. The release of ions at the first material 124 and capture of ion by the second material 126 is collectively referred to as the ionic exchange. The rate of ionic exchange and, hence the ionic emission rate or flow, is controlled by the control device 128. The control device 128 can increase or decrease the rate of ion flow by altering the conductance, which alters the impedance, between the first and second materials 124 and 126. Through controlling the ion exchange, the system 120 can encode information in the ionic exchange process. Thus, the system 120 uses ionic emission to encode information in the ionic exchange.
[0239] The control device 128 can vary the duration of a fixed ionic exchange rate or current flow magnitude while keeping the rate or magnitude near constant, similar to when the frequency is modulated and the amplitude is constant. Also, the control device 128 can vary the level of the ionic exchange rate or the magnitude of the current flow while keeping the duration near constant. Thus, using various combinations of changes in duration and altering the rate or magnitude, the control device 128 encodes information in the current flow or the ionic exchange. For example, the control device 128 may use, but is not limited to any of the following techniques namely, Binary Phase-Shift Keying (PSK), Frequency Modulation (FM), Amplitude Modulation (AM), On-Off Keying, and PSK with On-Off Keying.
[0240] Various aspects of the system 120 may comprise electronic components as part of the control device 128. Components that may be present include but are not limited to: logic and/or memory elements, an integrated circuit, an inductor, a resistor, and sensors for measuring various parameters. Each component may be secured to the framework and/or to another component. The components on the surface of the support may be laid out in any convenient configuration. Where two or more components are present on the surface of the solid support, interconnects may be provided.
[0241] The system 120 controls the conductance between the dissimilar materials and, hence, the rate of ionic exchange or the current flow. Through altering the conductance in a specific manner the system is capable of encoding information in the ionic exchange and the current signature. The ionic exchange or the current signature is used to uniquely identify the specific system. Additionally, the system 120 is capable of producing various different unique exchanges or signatures and, thus, provides additional information. For example, a second current signature based on a second conductance alteration pattern may be used to provide additional information, which information may be related to the physical environment. To further illustrate, a first current signature may be a very low current state that maintains an oscillator on the chip and a second current signature may be a current state at least a factor of ten higher than the current state associated with the first current signature.
[0242] Referring now back to FIG. 1, the mobile computer 50 is in communication with the data center 80. The data center 80 includes a database 82 and processing system 84. Information associated with all patients, including identity and medication types and doses, are stored in the database 82. The processing system 84 receives information from the mobile computer 50 and accesses the information in the database 82 of the data center 80 to provide information to the care provider through the mobile computer 50. The mobile computer 50 can communicate information including a photo of the patient for identification, the type of medication available to the care provider, as well as confirmation of the type and dose of medication that the care provider selects and delivers to the patient 60. The mobile computer 50 can communicate with the data center 80 using any mode and frequency of communication that is available in at the site, such as wireless, G2, G3, real-time, periodically based on predetermined time delays, as well as store and forward at later time.
[0243] FIG. 1 illustrates one aspect of the mobile computer 50. In various aspects, the mobile computer 50 may comprise or be implemented by a wireless device.
[0244] The mobile computer 50 generally may comprise various physical or logical elements implemented as hardware, software, or any combination thereof, as desired for a given set of design parameters or performance constraints. In various aspects, the physical or logical elements may be connected by one or more communications media. For example, communication media may comprise wired communication media, wireless communication media, or a combination of both, as desired for a given implementation.
[0245] As shown, the mobile computer 50 may comprise a display 58. The display 58 may be implemented using any type of visual interface such as a liquid crystal display (LCD).
[0246] As shown, the mobile computer 50 may comprise a memory 102. In various aspects, the memory 102 may comprise any machine-readable or computer-readable media capable of storing data, including both volatile and non-volatile memory. For example, memory may include read-only memory (ROM), random-access memory (RAM), dynamic RAM (DRAM), Double-Data-Rate DRAM (DDR-RAM), synchronous DRAM (SDRAM), static RAM (SRAM), programmable ROM (PROM), erasable programmable ROM (EPROM), electrically erasable programmable ROM (EEPROM), flash memory (e.g., NOR or NAND flash memory), content addressable memory (CAM), polymer memory (e.g., ferroelectric polymer memory), phase-change memory (e.g., ovonic memory), ferroelectric memory, silicon-oxide-nitride-oxide-silicon (SONOS) memory, disk memory (e.g., floppy disk, hard drive, optical disk, magnetic disk), or card (e.g., magnetic card, optical card), or any other type of media suitable for storing information.
[0247] The mobile computer 50 may comprise a processor 100 such as a central processing unit (CPU). In various aspects, the processor 100 may be implemented as a general purpose processor, a chip multiprocessor (CMP), a dedicated processor, an embedded processor, a digital signal processor (DSP), a network processor, a media processor, an input/output (I/O) processor, a media access control (MAC) processor, a radio baseband processor, a co-processor, a microprocessor such as a complex instruction set computer (CISC) microprocessor, a reduced instruction set computing (RISC) microprocessor, and/or a very long instruction word (VLIW) microprocessor, or other processing device. The processor 510 also may be implemented by a controller, a microcontroller, an application specific integrated circuit (ASIC), a field programmable gate array (FPGA), a programmable logic device (PLD), and so forth.
[0248] In various aspects, the processor 100 may be arranged to run an operating system (OS) and various mobile applications. Examples of an OS include, for example, operating systems generally known under the trade name of Microsoft Windows OS, and any other proprietary or open source OS. Examples of mobile applications include, for example, a telephone application, a camera (e.g., digital camera, video camera) application, a browser application, a multimedia player application, a gaming application, a messaging application (e.g., e-mail, short message, multimedia), a viewer application, and so forth.
[0249] In various aspects, the processor 100 may be arranged to receive information through a communications interface 104. The communications interface 104 may comprises any suitable hardware, software, or combination of hardware and software that is capable of coupling the mobile computer 50 to one or more networks and/or devices, such as the pill 20, the tag 22, the device 40, interrogation unit 46, the patient identity confirmation device 70, the tag 32, the sleeve 34, and/or the data center 80, among other devices. The communications interface 104 may be arranged to operate with any suitable technique for controlling information signals using a desired set of communications protocols, services or operating procedures. The communications interface 104 may include the appropriate physical connectors to connect with a corresponding communications medium, whether wired or wireless.
[0250] Vehicles of communication include a network. In various aspects, the network may comprise local area networks (LAN) as well as wide area networks (WAN) including without limitation Internet, wired channels, wireless channels, communication devices including telephones, computers, wire, radio, optical or other electromagnetic channels, and combinations thereof, including other devices and/or components capable of/associated with communicating data. For example, the communication environments include in-body communications, various devices, various modes of communications such as wireless communications, wired communications, and combinations of the same.
[0251] Wireless communication modes include any mode of communication between points that utilizes, at least in part, wireless technology including various protocols and combinations of protocols associated with wireless transmission, data, and devices. The points include, for example, wireless devices such as wireless headsets, audio and multimedia devices and equipment, such as audio players and multimedia players, telephones, including mobile telephones and cordless telephones, and computers and computer-related devices and components, such as printers.
[0252] Wired communication modes include any mode of communication between points that utilizes wired technology including various protocols and combinations of protocols associated with wired transmission, data, and devices. The points include, for example, devices such as audio and multimedia devices and equipment, such as audio players and multimedia players, telephones, including mobile telephones and cordless telephones, and computers and computer-related devices and components, such as printers.
[0253] Accordingly, in various aspects, the communications interface 104 may comprise one or more interfaces such as, for example, a wireless communications interface, a wired communications interface, a network interface, a transmit interface, a receive interface, a media interface, a system interface, a component interface, a switching interface, a chip interface, a controller, and so forth. When implemented by a wireless device or within wireless system, for example, the mobile computer 50 may include a wireless interface comprising one or more antennas, transmitters, receivers, transceivers, amplifiers, filters, control logic, and so forth.
[0254] In various implementations, the described aspects may communicate over wireless shared media in accordance with a number of wireless protocols. Examples of wireless protocols may include various wireless local area network (WLAN) protocols, including the Institute of Electrical and Electronics Engineers (IEEE) 802.xx series of protocols, such as IEEE 802.11a/b/g/n, IEEE 802.16, IEEE 802.20, and so forth. Other examples of wireless protocols may include various wireless wide area network (WWAN) protocols, such as GSM cellular radiotelephone system protocols with GPRS, CDMA cellular radiotelephone communication systems with 1.times.RTT, EDGE systems, EV-DO systems, EV-DV systems, HSDPA systems, and so forth. Further examples of wireless protocols may include wireless personal area network (PAN) protocols, such as an Infrared protocol, a protocol from the Bluetooth Special Interest Group (SIG) series of protocols, including Bluetooth Specification versions v1.0, v1.1, v1.2, v2.0, v2.0 with Enhanced Data Rate (EDR), as well as one or more Bluetooth Profiles, and so forth. Yet another example of wireless protocols may include near-field communication techniques and protocols, such as electro-magnetic induction (EMI) techniques. An example of EMI techniques may include passive or active radio-frequency identification (RFID) protocols and devices. Other suitable protocols may include Ultra Wide Band (UWB), Digital Office (DO), Digital Home, Trusted Platform Module (TPM), ZigBee, and so forth.
[0255] In various implementations, the described aspects may comprise part of a cellular communication system. Examples of cellular communication systems may include CDMA cellular radiotelephone communication systems, GSM cellular radiotelephone systems, North American Digital Cellular (NADC) cellular radiotelephone systems, Time Division Multiple Access (TDMA) cellular radiotelephone systems, Extended-TDMA (E-TDMA) cellular radiotelephone systems, Narrowband Advanced Mobile Phone Service (NAMPS) cellular radiotelephone systems, third generation (3G) systems such as WCDMA, CDMA-2000, UMTS cellular radiotelephone systems compliant with the Third-Generation Partnership Project (3GPP), and so forth.
[0256] In various aspects, the mobile computer 50 includes the functionality to wirelessly receive and/or wirelessly transmit data, e.g., physiologic data.
[0257] Further, in various aspects, the mobile computer 50 may incorporate and/or be associated with, e.g., communicate with, various devices. Such devices may generate, receive, and/or communicate data, e.g., physiologic data. The devices include, for example, "intelligent" devices such as gaming devices, e.g., electronic slot machines, handheld electronic games, electronic components associated with games and recreational activities.
[0258] The mobile computer 50 may be implemented as a mobile telephone. For example, the mobile computer 50 may be implemented as a short-range, portable electronic device used for mobile voice or data communication over a network of specialized cell site base stations. The mobile telephone is sometimes known as or referred to as "mobile," "wireless," "cellular phone," "cell phone," or "hand phone (HP)."
[0259] In addition to the standard voice function of a telephone, various aspects of mobile telephones may support many additional services and accessories such as short message service (SMS) for text messaging, email, packet switching for access to the Internet, java gaming, wireless, e.g., short range data/voice communications, infrared, camera with video recorder, and multimedia messaging system (MMS) for sending and receiving photos and video. Some aspects of mobile telephones connect to a cellular network of base stations (cell sites), which is, in turn, interconnected to the public switched telephone network (PSTN) or satellite communications in the case of satellite phones. Various aspects of mobile telephones can connect to the Internet, at least a portion of which can be navigated using the mobile telephones.
[0260] Some embodiments may be implemented, for example, using a machine-readable medium or article which may store an instruction or a set of instructions that, if executed by a machine, may cause the machine to perform a method and/or operations in accordance with the embodiments. Such a machine may include, for example, any suitable processing platform, computing platform, computing device, processing device, computing system, processing system, computer, processor, or the like, and may be implemented using any suitable combination of hardware and/or software. The machine-readable medium or article may include, for example, any suitable type of memory unit, memory device, memory article, memory medium, storage device, storage article, storage medium and/or storage unit, for example, memory, removable or non-removable media, erasable or non-erasable media, writeable or re-writeable media, digital or analog media, hard disk, floppy disk, Compact Disk Read Only Memory (CD-ROM), Compact Disk Recordable (CD-R), Compact Disk Rewriteable (CD-RW), optical disk, magnetic media, magneto-optical media, removable memory cards or disks, various types of Digital Versatile Disk (DVD), a tape, a cassette, or the like. The instructions may include any suitable type of code, such as source code, compiled code, interpreted code, executable code, static code, dynamic code, and the like. The instructions may be implemented using any suitable high-level, low-level, object-oriented, visual, compiled and/or interpreted programming language, such as C, C++, Java, BASIC, Perl, Matlab, Pascal, Visual BASIC, assembly language, machine code, and so forth.
[0261] It is to be understood that this invention is not limited to particular embodiments or aspects described, as such may vary. It is also to be understood that the terminology used herein is for the purpose of describing particular embodiments only, and is not intended to be limiting, since the scope of the present invention will be limited only by the appended claims.
[0262] Where a range of values is provided, it is understood that each intervening value, to the tenth of the unit of the lower limit unless the context clearly dictates otherwise, between the upper and lower limit of that range and any other stated or intervening value in that stated range, is encompassed within the invention. The upper and lower limits of these smaller ranges may independently be included in the smaller ranges and are also encompassed within the invention, subject to any specifically excluded limit in the stated range. Where the stated range includes one or both of the limits, ranges excluding either or both of those included limits are also included in the invention.
[0263] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. Although any methods and materials similar or equivalent to those described herein can also be used in the practice or testing of the present invention, representative illustrative methods and materials are now described.
[0264] All publications and patents cited in this specification are herein incorporated by reference as if each individual publication or patent were specifically and individually indicated to be incorporated by reference and are incorporated herein by reference to disclose and describe the methods and/or materials in connection with which the publications are cited. The citation of any publication is for its disclosure prior to the filing date and should not be construed as an admission that the present invention is not entitled to antedate such publication by virtue of prior invention. Further, the dates of publication provided may be different from the actual publication dates which may need to be independently confirmed.
[0265] It is noted that, as used herein and in the appended claims, the singular forms "a", "an", and "the" include plural referents unless the context clearly dictates otherwise. It is further noted that the claims may be drafted to exclude any optional element. As such, this statement is intended to serve as antecedent basis for use of such exclusive terminology as "solely," "only" and the like in connection with the recitation of claim elements, or use of a "negative" limitation.
[0266] As will be apparent to those of skill in the art upon reading this disclosure, each of the individual embodiments described and illustrated herein has discrete components and features which may be readily separated from or combined with the features of any of the other several embodiments without departing from the scope or spirit of the present invention. Any recited method can be carried out in the order of events recited or in any other order which is logically possible.
[0267] Unless specifically stated otherwise, it may be appreciated that terms such as "processing," "computing," "calculating," "determining," or the like, refer to the action and/or processes of a computer or computing system, or similar electronic computing device, that manipulates and/or transforms data represented as physical quantities (e.g., electronic) within the computing system's registers and/or memories into other data similarly represented as physical quantities within the computing system's memories, registers or other such information storage, transmission or display devices.
[0268] Numerous specific details have been set forth herein to provide a thorough understanding of the embodiments. It will be understood by those skilled in the art, however, that the embodiments may be practiced without these specific details. In other instances, well-known operations, components and circuits have not been described in detail so as not to obscure the embodiments. It can be appreciated that the specific structural and functional details disclosed herein may be representative and do not necessarily limit the scope of the embodiments.
[0269] Although the foregoing invention has been described in some detail by way of illustration and example for purposes of clarity of understanding, it is readily apparent to those of ordinary skill in the art in light of the teachings of this invention that certain changes and modifications may be made thereto without departing from the scope of the appended claims.
[0270] Accordingly, the preceding merely illustrates the principles of the invention. It will be appreciated that those skilled in the art will be able to devise various arrangements which, although not explicitly described or shown herein, embody the principles of the invention and are included within its spirit and scope. Furthermore, all examples and conditional language recited herein are principally intended to aid the reader in understanding the principles of the invention and the concepts contributed by the inventors to furthering the art, and are to be construed as being without limitation to such specifically recited examples and conditions. Moreover, all statements herein reciting principles, aspects, and embodiments of the invention as well as specific examples thereof, are intended to encompass both structural and functional equivalents thereof. Additionally, it is intended that such equivalents include both currently known equivalents and equivalents developed in the future, i.e., any elements developed that perform the same function, regardless of structure. The scope of the present invention, therefore, is not intended to be limited to the exemplary embodiments shown and described herein. Rather, the scope and spirit of present invention is embodied by the appended claims.
* * * * *
D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.