Modulation Of T Cells With Bispecific Antibodies And Fc Fusions
Chu; Seung ; et al.
U.S. patent application number 16/721356 was filed with the patent office on 2020-10-29 for modulation of t cells with bispecific antibodies and fc fusions. The applicant listed for this patent is Xencor, Inc.. Invention is credited to Matthew Bernett, Seung Chu, John Desjarlais, Dilki Wickramarachichi.
| Application Number | 20200339624 16/721356 |
| Document ID | / |
| Family ID | 1000004942432 |
| Filed Date | 2020-10-29 |








View All Diagrams
| United States Patent Application | 20200339624 |
| Kind Code | A1 |
| Chu; Seung ; et al. | October 29, 2020 |
MODULATION OF T CELLS WITH BISPECIFIC ANTIBODIES AND FC FUSIONS
Abstract
The present invention relates to methods and compositions for modulating T cells. The modulation includes suppressing or inducing regulatory T cells or cytotoxic T cells.
| Inventors: | Chu; Seung; (Upland, CA) ; Bernett; Matthew; (Monrovia, CA) ; Wickramarachichi; Dilki; (Pasadena, CA) ; Desjarlais; John; (Pasadena, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004942432 | ||||||||||
| Appl. No.: | 16/721356 | ||||||||||
| Filed: | December 19, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14217166 | Mar 17, 2014 | 10544187 | ||
| 16721356 | ||||
| 61911438 | Dec 3, 2013 | |||
| 61800743 | Mar 15, 2013 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 2317/73 20130101; C07K 2319/00 20130101; C07K 2317/74 20130101; C07K 1/18 20130101; C07K 2317/31 20130101; A61K 2039/505 20130101; C07K 2317/60 20130101; C07K 2317/515 20130101; C07K 16/2866 20130101; C07K 2317/52 20130101; C07K 14/55 20130101; C07K 16/2815 20130101; C07K 16/2812 20130101 |
| International Class: | C07K 1/18 20060101 C07K001/18; C07K 16/28 20060101 C07K016/28; C07K 14/55 20060101 C07K014/55 |
Claims
1.-69. (canceled)
70. A heterodimeric protein comprising: (a) a first monomer comprising: (i) a first Fc domain; (ii) an IL-2 protein; and (b) a second monomer comprising: (i) a second Fc domain.
71. The heterodimeric protein according to claim 70, wherein the first and second Fc domains are variant Fc domains comprising amino acid variants selected from the group consisting of: L368D/K370S and S364K; L368D/K370S and S364K/E357L; L368D/K370S and S364K/E357Q; T411E/K360E/Q362E and D401K; L368E/K370S and S364K; and K370S and S364K/E357Q, wherein numbering is according to EU index as in Kabat.
72. The heterodimeric protein according to claim 71, wherein the first and/or second variant Fc domain further comprises an amino acid variant independently selected from the group consisting of: 236R, 328R, 330L, 236R/328R, 239D/332E, E233P/L234V/L235A/G236del/S239K, E233P/L234V/L235A/G236del/S239K/A327G, E233P/L234V/L235A/G236del/S267K/A327G, E233P/L234V/L235A/G236del, and E233P/L234V/L235A/G236del/S267K, wherein numbering is according to EU index as in Kabat.
73. The heterodimeric protein according to claim 70, wherein the first Fc domain is a variant Fc domain comprising amino acid variants E233P/L234V/L235A/G236del/S267K and S364K/E357Q, and wherein the second Fc domain is a variant Fc domain comprising amino acid variants E233P/L234V/L235A/G236del/S267K and L368D/K370S, wherein numbering is according to EU index as in Kabat.
74. The heterodimeric protein according to claim 70, wherein the IL-2 protein is an IL-2 variant having reduced ability to bind to IL-2R.beta., IL-2R.gamma., and/or IL-2R.alpha..
75. The heterodimeric protein according to claim 70, wherein the IL-2 protein is an IL-2 variant having increased ability to bind to IL-2R.alpha..
76. The heterodimeric protein according to claim 70, wherein the IL-2 protein is an IL-2 variant having reduced ability to bind to IL-2R.beta. and/or IL-2R.gamma. and increased ability to bind to IL-2R.alpha..
77. A method of inducing T cells, the method comprising contacting the T cells with a composition comprising a heterodimeric protein according to claim 70.
78. The method according to claim 77, wherein the T cells are regulatory T cells (Tregs).
79. A method of suppressing T cells, the method comprising contacting the T cells with a composition comprising a heterodimeric protein according to claim 70.
80. A method A method of treating an autoimmune disease in a subject, the method comprising administering to the subject a composition comprising a heterodimeric protein according to claim 70.
81. A method A method of treating a cancer in a subject, the method comprising administering to the subject a composition comprising a heterodimeric protein according to claim 70.
82. A nucleic acid composition encoding a heterodimeric protein, the nucleic acid composition comprising: a) a first nucleic acid encoding the first monomer of claim 70; and b) a second nucleic acid encoding the second monomer of claim 70.
83. A host cell comprising the nucleic acid composition of claim 82.
84. A method of making a heterodimeric protein comprising culturing a host cell according to claim 83 under conditions whereby the heterodimeric protein is produced.
85. A method of purifying a heterodimeric protein according to claim 70, the method comprising: (a) providing a composition comprising the heterodimeric protein; (b) loading the composition onto an ion exchange column; and (c) collecting a fraction containing the heterodimeric protein.
86. An IL-2 Fc fusion comprising: (a) a first monomer comprising: (i) a first Fc domain; (ii) a first IL-2 protein; and (b) a second monomer comprising (i) a second Fc domain; and (ii) a second IL-2 protein.
87. The IL-2 Fc fusion according to claim 86, wherein the first and/or second IL-2 protein is an IL-2 variant engineered to have reduced ability to bind to IL-2R.beta., IL-2R.gamma., and/or IL-2R.alpha..
88. A method of inducing T cells, the method comprising contacting the T cells with an IL-2 Fc fusion according to claim 86.
89. The method according to claim 88, wherein the T cells are regulatory T cells (Tregs).
90. A method of suppressing T cells, the method comprising contacting the T cells with an IL-2 Fc fusion according to claim 86.
91. A method A method of treating an autoimmune disease in a subject, the method comprising administering to the subject an IL-2 Fc fusion according to claim 86.
92. A method A method of treating a cancer in a subject, the method comprising administering to the subject an IL-2 Fc fusion according to claim 86.
93. A nucleic acid composition encoding an IL-2 fusion, the nucleic acid composition comprising: a) a first nucleic acid encoding the first monomer of claim 86; and b) a second nucleic acid encoding the second monomer of claim 86.
94. A host cell comprising the one or more nucleic acids of claim 86.
95. A method of making an IL-2 Fc fusion, the method comprising culturing a host cell according to claim 94 under conditions whereby the IL-2 Fc fusion is produced.
96. A method of purifying an IL-2 Fc fusion according to claim 86, the method comprising: (a) providing a composition comprising the IL-2 Fc fusion; (b) loading the composition onto an ion exchange column; and (c) collecting a fraction containing the IL-2 Fc fusion.
Description
PRIORITY CLAIM
[0001] This application is a continuation of U.S. patent application Ser. No. 14/217,166, filed Mar. 17, 2014, which claims priority to U.S. Provisional Patent Application Nos. 61/800,743, filed Mar. 15, 2013 and 61/911,438, filed Dec. 3, 2013, each of which is expressly incorporated by reference in the entirety.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Dec. 19, 2019, is named 067461-5163-US01_ST25.txt and is 831,510 bytes in size.
RELATED APPLICATIONS
[0003] U.S. Ser. No. 14/216,705, filed Mar. 17, 2014; Ser. No. 13/194,904, filed Jul. 29, 2011; Ser. No. 14/205,248, filed Mar. 11, 2014; Ser. No. 12/875,015, filed Sep. 2, 2010; Ser. No. 13/568,028, filed Aug. 6, 2012; Ser. No. 13/887,234, filed May 3, 2013; Ser. No. 13/648,951, filed Oct. 10, 2012; 61/913,832, filed Dec. 9, 2013, and 61/938,095, filed Feb. 10, 2014 are all expressly incorporated by reference in their entirety, particularly for the recitation of amino acid positions and substitutions, and all data, figures and legends relating thereto.
TECHNICAL FIELD
[0004] The present disclosure relates to methods and compositions for modulating T cells.
BACKGROUND OF THE INVENTION
[0005] Immune system homeostasis relies on a fine balance between a variety of T cell populations, including effector CD8 and CD4 T cells and regulatory T cells. In disease states however, such as cancer and autoimmune disease, this balance can be perturbed. In cancer, infiltrating anti-tumor cytotoxic T cells can be prevented from attacking cancer cells by tumor-resident regulatory T cells. This can be seen from analysis of most human tumors, in which there is a significant correlation between immune infiltration by cytotoxic T cells and improved outcome, whereas infiltration by regulatory T cells is instead associated with a poor outcome. Indeed, several studies have demonstrated prognostic significance of the CD8/Treg tumor ratio. Numerous mouse models have shown that depletion of Treg with anti-CD25 antibody before tumor implantation can have a dramatic impact on prevention of tumor growth. In autoimmune diseases, effector T cells remain unregulated and attack the body's own tissues. A major premise in this regard is that defects in Treg cell number or function are a contributing factor. Therefore, the ability to alter the balance between cytotoxicity and regulation by fine-tuning the T cell response has great potential for the treatment of cancer, autoimmune, and other diseases.
[0006] One approach to controlling the balance of effector to regulatory T cells is to target the Treg population for direct modulation. However, despite years of effort, the discovery of a single Treg-specific surface marker has been elusive, frustrating efforts to deplete them specifically with monoclonal antibodies.
[0007] Effector versus regulatory T cells can be loosely identified by their surface markers, which can change based on their activation state. Cytotoxic T cells express CD8, which interacts with class I MHC. Effector helper T cells express CD4, which interacts with class II MHC on antigen-presenting cells. The hallmark of Treg cells is constitutive expression of both CD4 and CD25. CD25 is the alpha component of the IL2 receptor (IL2R.alpha.), which, together with CD122 (IL2R.beta.) and the common cytokine receptor .gamma.-chain(y.sub.c) (CD132) form the trimeric high-affinity receptor for IL2. Several approaches have attempted Treg-specific depletion by targeting the high-affinity IL2 receptor CD25 (IL2R.alpha.) with anti-CD25 antibodies such as daclizumab, or with IL2-diptheria toxin (IL2-DT) fusions. However, CD25 alone is not an ideal target because it also expressed on CD8 and CD4 effector T cells upon activation. Thus, approaches that target Treg CD25 by itself might defeat their own purpose by also depleting the activated effector cells that could potentially attack the tumor.
[0008] Because of the importance of IL2 for T cell proliferation and homeostasis, a variety of approaches to T cell modulation have utilized IL2 itself or blocking of its high-affinity receptor component CD25. Systemic IL2 administration (Proleukin) is an approved therapy for metastatic melanoma and metastatic renal cell carcinoma based on its ability to promote expansion of effector T cells. However, systemic IL2 administration is also expected to promote the suppressive Treg population, potentially diminishing or confounding the desired enhancement of cytotoxic T cells. Furthermore, systemic IL2 administration is also associated with a variety of toxicities. Patients receiving systemic IL2 treatment frequently experience severe cardiovascular, pulmonary, renal, hepatic, gastrointestinal, neurological, cutaneous, haematological and systemic adverse events. The majority of these side effects can be explained by the development of so-called vascular leak syndrome (VLS), a pathological increase in vascular permeability leading to pulmonary edema and other issues. There is no treatment of VLS other than withdrawal of IL2. These problems have led to the pursuit of IL2 variants that perturb its affinity for one or more of its receptor subunits. Alternatively, anti-CD25 antibodies that block IL2-mediated T cell expansion have been utilized to treat various diseases. Zenapax (daclizumab) is an approved immunosuppressant for organ transplantation and is being investigated for the treatment of autoimmune diseases such as multiple sclerosis. These uses were developed based on daclizumab's presumed ability to reduce effector T cell responses. However, due to the more recently recognized dependence of Treg on IL2 for survival, daclizumab is now--somewhat paradoxically--being utilized in efforts to reduce Treg numbers in oncology. Because of the strong potential of either IL2 or anti-CD25 agents to promote or reduce both effector T cells and Treg with limited selectivity, there is a strong need in the field to create more selective Treg modulators.
SUMMARY OF THE INVENTION
[0009] Accordingly, the present invention provides methods and compositions for suppressing and inducing T cells. In preferred aspects, the methods and compositions of the invention suppress or induce specific T cell types with little or no impact on other T cell types. In further embodiments, the methods and compositions of the invention suppress or induce regulatory T cells with little or no impact on other T cell types, including cytotoxic T cells. In other embodiments, the methods and compositions of the invention suppress or induce cytotoxic T cells with little or no impact on other T cell types, including regulatory T cells.
[0010] In one aspect, the present invention provides a method for suppressing T cells that includes the step of administering a composition comprising a bispecific antibody, wherein that bispecific antibody includes: (a) a first monomer that has (i) a first heavy chain constant region with a first variant Fc domain; and (ii) an anti-CD25 binding moiety; and (b) a second monomer that has (i) a second heavy chain constant region with a second variant Fc domain; and (ii) a member selected from the group: an anti-CD4 binding moiety, an anti-CD8 binding moiety, an anti-CCR4 moiety, an anti-GITR binding moiety, and an anti-PD-1 binding moiety. In specific embodiments, the first variant Fc domain has a different amino acid sequence than the second variant Fc domain. The administration of such a bispecific antibody serves to suppress the T cells. Suppression can be measured using assays known in the art, including cell proliferation assays. Suppression can be shown in such assays by a decrease in cell proliferation and/or general T cell number as compared to the proliferation and/or numbers seen in the absence of the bispecific antibody of the invention.
[0011] In further embodiments and in accordance with the above, the T cells suppressed by the methods of the invention are regulatory T cells. In still further embodiments, the second monomer comprises the anti-CD4 binding moiety, and the bispecific antibody specifically targets regulatory T cells with limited to no impact on other T cell types.
[0012] In still further embodiments and in accordance with any of the above, the anti-CD25 binding moiety is an anti-CD25 scFV sequence that is covalently attached to the first heavy chain sequence.
[0013] In still further embodiments and in accordance with any of the above, the T cells suppressed by the methods and compositions of the invention are cytotoxic T-cells. In yet further embodiments, the second monomer comprises said anti-CD8 binding moiety, and the bispecific antibody specifically targets cytotoxic T cells with limited to no impact on other T cell types. In yet further embodiments, the anti-CD8 binding moiety comprises all or a portion of an antigen binding region of an antibody selected from the group consisting of MCD8, 3B5, Sk1, OKT-8, and DK-25.
[0014] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains include a set of amino acid substitutions selected from those sets depicted in FIG. 33A-33C.
[0015] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains comprise a set of amino acid substitutions selected from the group consisting of those sets depicted in FIG. 34A-34C.
[0016] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains comprise a set of amino acid substitutions selected from the group consisting of those sets depicted in FIG. 35.
[0017] In still further embodiments and in accordance with any of the above, the first and/or second variant Fc domain comprises an amino acid variant selected from the group consisting of: 236R, 239D, 239E, 243L, M252Y, V259I, 267D, 267E, 298A, V308F, 328F, 328R, 330L, 332D, 332E, M428L, N434A, N434S, 236R/328R, 239D/332E, M428L, 236R/328F, V259I/V308F, 267E/328F, M428L/N434S, Y436I/M428L, Y436V/M428L, Y436I/N434S, Y436V/N434S, 239D/332E/330L, M252Y/S254T/T256E, V259I/V308F/M428L, and E233P/L234V/L235A/G236del/S267K.
[0018] In still further embodiments and in accordance with any of the above, the bispecific antibody comprises a sequence selected from the sequences depicted in FIGS. 30-31.
[0019] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 11209-OKT4A_H0L0_scFv_Anti-TAC_H1L1_scFv_GDQ-Fc(216)_IgG1_pI_ISO(-)/pI_IS- O(+RR)_IgG1, Heavy chain 2 (Heavy chain 2 (Anti-TAC_H1L1_scFv_GDQ-Fc(216)_IgG1_pI_ISO(+RR)) in FIG. 30A-30FF.
[0020] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 11209-OKT4A_HOL0_scFv_Anti-TAC_H1L1_scFv_GDQ-Fc(216)_IgG1_pI_ISO(-)/pI_IS- O(+RR)_IgG1, Heavy chain 1 (OKT4A_HOL0_scFv_GDQ-Fc(216)_IgG1_pI_ISO(-)) in FIG. 30A-30FF.
[0021] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 12143-OKT4A_HOL0_scFv_Anti-TAC_H1L1_scFv_-Fc(216)_IgG1_pI_ISO(-)/pI_ISO(+- RR)_C220S/FcKO, Heavy chain 2 (Anti-TAC_H1L1_scFv_Fc(216)_IgG1_pI_ISO(+RR)_G236R/L328R) in FIG. 30A-30FF.
[0022] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 12143-OKT4A_H0L0_scFv_Anti-TAC_H1L1_scFv_-Fc(216)_IgG1_pI_ISO(-)/pI_ISO(+- RR)_C220S/FcKO, Heavy chain 1 (OKT4A_HOL0_scFv_Fc(216)_IgG1_pI_ISO(-)_G236R/L328R) in FIG. 30A-30FF.
[0023] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 13531-OKT4A_H1L1_Fab-Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO (-)-pI_ISO(+RR)_C220S_IgG1_G236R/L328R, Heavy chain 2 (Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO(+RR)_G236R/L328R) in FIG. 30A-30FF.
[0024] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 13531-OKT4A_H1L1_Fab-Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO (-)-pI_ISO(+RR)_C220S_IgG1_G236R/L328R, Heavy chain 1 (OKT4A_H1_IgG1_pI_ISO(-)_G236R/L328R) in FIG. 30A-30FF.
[0025] In still further embodiments and in accordance with any of the above, the second monomer further comprises a sequence according to the sequence designated as 13531-OKT4A_H1L1_Fab-Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO (-)-pI_ISO(+RR)_C220S_IgG1_G236R/L328R, Light chain (OKT4A_L1) in FIG. 30A-30FF.
[0026] In a further aspect, the present invention provides a method for stimulating T cells that includes administering a heterodimeric protein, where the heterodimeric protein includes: (a) a first monomer with (i) a first heavy chain constant region comprising a first variant Fc domain; (ii) an IL-2 protein; and (b) a second monomer with: (i) a second heavy chain constant region comprising a second variant Fc domain; (ii) a member selected from the group consisting of: an anti-CD4 binding moiety, an anti-CD8 binding moiety, an anti-CTLA4 binding moiety, an anti-CCR4 binding moiety, an anti-PD-1 binding moiety, and an anti-GITR binding moiety. In further embodiments, the first variant Fc domain has a different amino acid sequence than the second variant Fc domain. Administration of this heterodimeric protein stimulates the T cells. As will be appreciated, the IL2 protein may comprise a full length protein or a portion of the full length IL2 protein. In further embodiments, the full or portion of the IL2 protein that is part of the heterodimeric protein comprises a human IL2 protein sequence.
[0027] In a further embodiment and in accordance with the above, the T cells are regulatory T cells and the second monomer is the anti-CD4 binding moiety.
[0028] In still further embodiments and in accordance with any of the above, the second monomer further includes: (a) the second heavy chain constant region further having a heavy chain variable domain, and (b) a light chain sequence, where the heavy chain variable domain and the light chain sequence together form antigen binding moiety, including without limitation the anti-CD4 binding moiety.
[0029] In still further embodiments and in accordance with any of the above, the stimulated T cells are cytotoxic T cells and the second monomer comprises the anti-CD8 binding moiety. In yet further embodiments, the anti-CD8 binding moiety comprises all or a portion of an antigen binding region of an antibody selected from the group consisting of MCD8, 3B5, Sk1, OKT-8, and DK-25.
[0030] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains include a set of amino acid substitutions selected from the group consisting of those sets depicted in FIG. 33, 34 or 35.
[0031] In still further embodiments and in accordance with any of the above, the first and/or second variant Fc domain comprises an amino acid variant selected from the group consisting of: 236R, 239D, 239E, 243L, M252Y, V259I, 267D, 267E, 298A, V308F, 328F, 328R, 330L, 332D, 332E, M428L, N434A, N434S, 236R/328R, 239D/332E, M428L, 236R/328F, V259I/V308F, 267E/328F, M428L/N434S, Y436I/M428L, Y436V/M428L, Y436I/N434S, Y436V/N434S, 239D/332E/330L, M252Y/S254T/T256E, V259I/V308F/M428L, and E233P/L234V/L235A/G236del/S267K.
[0032] In still further embodiments and in accordance with any of the above, the heterodimeric protein comprises a sequence selected from the sequences depicted in FIGS. 30-31.
[0033] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 13027-hIL2_OKT4A_H1L1_IgG1_pI_ISO(-/+RR)_C220S_G236R/L328R, Heavy chain 1 (hIL2_IgG1_pI_ISO(-)_C220S/G236R/L328R) in FIG. 30A-30FF.
[0034] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 13027-hIL2_OKT4A_H1L1_IgG1_pI_ISO(-/+RR)_C220S_G236R/L328R, Heavy chain 2 (OKT4A_H1_IgG1_pI_ISO(+RR)_G236R/L328R) in FIG. 30A-30FF.
[0035] In still further embodiments and in accordance with any of the above, the second monomer further comprises a sequence according to the sequence designated as 13027-hIL2_OKT4A_H1L1_IgG1_pI_ISO(-/+RR)_C220S_G236R/L328R, Light chain (OKT4A_L1) in FIG. 30A-30FF.
[0036] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 13038-OKT4A_H1L1_IgG1_G236R/L328R_hIL2(2), Heavy chain (OKT4A_H1_IgG1_G236R/L328R_hIL2) in FIG. 30A-30FF.
[0037] In still further embodiments and in accordance with any of the above, the second monomer further comprises a sequence according to the sequence designated as 13038-OKT4A_H1L1_IgG1_G236R/L328R_hIL2(2), Light chain (OKT4A_L1) in FIG. 30A-30FF.
[0038] In further aspects and in accordance with any of the above, the present invention provides a composition comprising a heterodimeric antibody, where the heterodimeric antibody includes: (a) a first monomer having (i) a first antigen-binding domain, which is an anti-CD25 binding domain; (ii) a first heavy chain sequence comprising a first variant Fc domain as compared to a human Fc domain; and (b) a second monomer having (i) a second antigen-binding domain that binds to a member selected from the group consisting of: CD4, CD8, CCR4, GITR, and PD-1, and (ii) a second heavy chain sequence comprising a second variant Fc domain as compared to a human Fc domain. In further embodiments, the first and second variant Fc domains have different amino acid sequences.
[0039] In further embodiments and in accordance with the above, the antigen-binding domain binds to CD4.
[0040] In still further embodiments and in accordance with any of the above, the first and second variant Fc domain includes an amino acid variant independently selected from the variants listed in FIG. 33, 34, or 35.
[0041] In still further embodiments and in accordance with any of the above, the first and second variant Fc domain comprises an amino acid variant selected from the group consisting of: L368D/K370S and S364K; L368D/K370S and S364K/E357L; L368D/K370S and S364K/E357Q; T411E/K360E/Q362E and D401K; L368E/K370S and S364K; K370S and S364K/E357Q; and K370S and S364K/E357Q.
[0042] In still further embodiments and in accordance with any of the above, the first and/or second variant Fc domain further comprises an amino acid variant selected from the group consisting of: 236R, 239D, 239E, 243L, M252Y, V259I, 267D, 267E, 298A, V308F, 328F, 328R, 330L, 332D, 332E, M428L, N434A, N434S, 236R/328R, 239D/332E, M428L, 236R/328F, V259I/V308F, 267E/328F, M428L/N434S, Y436I/M428L, Y436V/M428L, Y436I/N434S, Y436V/N434S, 239D/332E/330L, M252Y/S254T/T256E, V259I/V308F/M428L, and E233P/L234V/L235A/G236del/S267K.
[0043] In still further embodiments and in accordance with any of the above, the anti-CD25 binding domain is an anti-CD25 scFv sequence and is covalently attached to said first heavy chain sequence.
[0044] In still further embodiments and in accordance with any of the above, the second antigen-binding domain comprises an scFv sequence.
[0045] In still further embodiments and in accordance with any of the above, the second monomer further has the second heavy chain sequence further comprising a heavy chain variable domain, and a light chain sequence, where the heavy chain variable domain and the light chain sequence form said second antigen-binding domain.
[0046] In still further embodiments and in accordance with any of the above, the composition comprises a format in accordance with a format as depicted in FIG. 3 or FIGS. 36A-37U.
[0047] In a further aspect, the present invention provides a composition comprising a heterodimeric protein that has: (a) a first monomer comprising: (i) a first protein comprising a cell marker; (ii) a first heavy chain sequence with a first variant Fc domain as compared to a human Fc domain; and (b) a second monomer comprising: (i) an antigen-binding domain that binds to a member selected from the group consisting of: CD4, CD8, CTLA-4, CCR4, and PD-1, and (ii) a second heavy chain sequence comprising a second variant Fc domain as compared to a human Fc domain. In further embodiments, the first and second variant Fc domains have different amino acid sequences.
[0048] In still further embodiments and in accordance with any of the above, the protein of the first monomer comprises a regulatory T cell marker selected from the group listed in FIG. 32. In other embodiments, the protein of the first monomer comprises a cytokine. In yet further embodiments, the cytokine is IL2.
[0049] In still further embodiments and in accordance with any of the above, the first and second variant Fc domain comprises an amino acid variant independently selected from the variants listed in FIG. 33, 34 or 35.
[0050] In still further embodiments and in accordance with any of the above, the first and second variant Fc domain includes an amino acid variant independently selected from the group consisting of: L368D/K370S and S364K; L368D/K370S and S364K/E357L; L368D/K370S and S364K/E357Q; T411E/K360E/Q362E and D401K; L368E/K370S and S364K; K370S and S364K/E357Q; and K370S and S364K/E357Q.
[0051] In still further embodiments and in accordance with any of the above, the first and/or second variant Fc domain further includes an amino acid variant selected from the group consisting of: 236R, 239D, 239E, 243L, M252Y, V259I, 267D, 267E, 298A, V308F, 328F, 328R, 330L, 332D, 332E, M428L, N434A, N434S, 236R/328R, 239D/332E, M428L, 236R/328F, V259I/V308F, 267E/328F, M428L/N434S, Y436I/M428L, Y436V/M428L, Y436I/N434S, Y436V/N434S, 239D/332E/330L, M252Y/S254T/T256E, V259I/V308F/M428L, and E233P/L234V/L235A/G236del/S267K.
[0052] In still further embodiments and in accordance with any of the above, the antigen-binding domain is an scFv sequence that is covalently attached to said second heavy chain sequence.
[0053] In still further embodiments and in accordance with any of the above, the second monomer further includes: (a) the second heavy chain sequence further comprising a heavy chain variable domain, and (b) a light chain sequence, wherein said heavy chain variable domain and said light chain sequence form said second antigen-binding domain.
[0054] In still further embodiments and in accordance with any of the above, the heterodimeric protein comprises a sequence as listed in FIGS. 30 and 31.
[0055] In still further embodiments and in accordance with any of the above, the invention provides one or more nucleic acids encoding a composition according to any of the compositions described above. In yet further embodiments, the invention includes a host cell expressing those one or more nucleic acids. In yet further embodiments, the present invention provides a method of making any of the compositions described herein, the method including the step of culturing a host cell or more nucleic acids encoding a composition according to any of the compositions described above under conditions whereby the composition is produced.
[0056] In further aspects, the present invention provides a method of purifying a heterodimeric protein or bispecific antibody in accordance with any of the above, the method including: (a) providing a composition in accordance with any of the above, (b) loading the composition onto an ion exchange column; and (c) collecting a fraction containing the heterodimeric protein or bispecific antibody, thus purifying the protein or antibody.
[0057] In a further aspect, the present invention provides a method of treating cancer in a subject, the method comprising administering to said subject a composition comprising a bispecific antibody, where the bispecific antibody includes: (a) a first monomer that has (i) a first heavy chain constant region with a first variant Fc domain; and (ii) an anti-CD25 binding moiety; and (b) a second monomer that has (i) a second heavy chain constant region with a second variant Fc domain; and (ii) a member selected from the group: an anti-CD4 binding moiety, an anti-CD8 binding moiety, an anti-CCR4 moiety, an anti-GITR binding moiety, and an anti-PD-1 binding moiety. In specific embodiments, the first variant Fc domain has a different amino acid sequence than the second variant Fc domain. The administration of such a bispecific antibody serves to suppress the T cells. Suppression can be measured using assays known in the art, including cell proliferation assays. Suppression can be shown in such assays by a decrease in cell proliferation and/or general T cell number as compared to the proliferation and/or numbers seen in the absence of the bispecific antibody of the invention.
[0058] In further embodiments and in accordance with the above, the T cells suppressed by the methods of the invention are regulatory T cells. In still further embodiments, the second monomer comprises the anti-CD4 binding moiety, and the bispecific antibody specifically targets regulatory T cells with limited to no impact on other T cell types.
[0059] In still further embodiments and in accordance with any of the above, the anti-CD25 binding moiety is an anti-CD25 scFV sequence that is covalently attached to the first heavy chain sequence.
[0060] In still further embodiments and in accordance with any of the above, the T cells suppressed by the methods and compositions of the invention are cytotoxic T-cells. In yet further embodiments, the second monomer comprises said anti-CD8 binding moiety, and the bispecific antibody specifically targets cytotoxic T cells with limited to no impact on other T cell types. In yet further embodiments, the anti-CD8 binding moiety comprises all or a portion of an antigen binding region of an antibody selected from the group consisting of MCD8, 3B5, Sk1, OKT-8, and DK-25.
[0061] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains include a set of amino acid substitutions selected from those sets depicted in FIG. 33.
[0062] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains comprise a set of amino acid substitutions selected from the group consisting of those sets depicted in FIG. 34A-34C.
[0063] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains comprise a set of amino acid substitutions selected from the group consisting of those sets depicted in FIG. 35.
[0064] In still further embodiments and in accordance with any of the above, the first and/or second variant Fc domain comprises an amino acid variant selected from the group consisting of: 236R, 239D, 239E, 243L, M252Y, V259I, 267D, 267E, 298A, V308F, 328F, 328R, 330L, 332D, 332E, M428L, N434A, N434S, 236R/328R, 239D/332E, M428L, 236R/328F, V259I/V308F, 267E/328F, M428L/N434S, Y436I/M428L, Y436V/M428L, Y436I/N434S, Y436V/N434S, 239D/332E/330L, M252Y/S254T/T256E, V259I/V308F/M428L, and E233P/L234V/L235A/G236del/S267K.
[0065] In still further embodiments and in accordance with any of the above, the bispecific antibody comprises a sequence selected from the sequences depicted in FIGS. 30-31.
[0066] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 11209-OKT4A_HOL0_scFv_Anti-TAC_H1L1_scFv_GDQ-Fc(216)_IgG1_pI_ISO(-)/pI_IS- O(+RR)_IgG1, Heavy chain 2 (Heavy chain 2 (Anti-TAC_H1L1_scFv_GDQ-Fc(216)_IgG1_pI_ISO(+RR)) in FIG. 30A-30FF.
[0067] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 11209-OKT4A_HOL0_scFv_Anti-TAC_H1L1_scFv_GDQ-Fc(216)_IgG1_pI_ISO(-)/pI_IS- O(+RR)_IgG1, Heavy chain 1 (OKT4A_HOL0_scFv_GDQ-Fc(216)_IgG1_pI_ISO(-)) in FIG. 30A-30FF.
[0068] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 12143-OKT4A_HOL0_scFv_Anti-TAC_H1L1_scFv_-Fc(216)_IgG1_pI_ISO(-)/pI_ISO(+- RR)_C220S/FcKO, Heavy chain 2 (Anti-TAC_H1L1_scFv_Fc(216)_IgG1_pI_ISO(+RR)_G236R/L328R) in FIG. 30A-30FF.
[0069] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 12143-OKT4A_HOL0_scFv_Anti-TAC_H1L1_scFv_-Fc(216)_IgG1_pI_ISO(-)/pI_ISO(+- RR)_C220S/FcKO, Heavy chain 1 (OKT4A_HOL0_scFv_Fc(216)_IgG1_pI_ISO(-)_G236R/L328R) in FIG. 30A-30FF.
[0070] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 13531-OKT4A_H1L1_Fab-Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO (-)-pI_ISO(+RR)_C220S_IgG1_G236R/L328R, Heavy chain 2 (Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO(+RR)_G236R/L328R) in FIG. 30A-30FF.
[0071] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 13531-OKT4A_H1L1_Fab-Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO (-)-pI_ISO(+RR)_C220S_IgG1_G236R/L328R, Heavy chain 1 (OKT4A_H1_IgG1_pI_ISO(-)_G236R/L328R) in FIG. 30A-30FF.
[0072] In still further embodiments and in accordance with any of the above, the second monomer further comprises a sequence according to the sequence designated as 13531-OKT4A_H1L1_Fab-Anti-TAC_H1.8L1_scFv_Fc(216)_IgG1_pI_ISO (-)-pI_ISO(+RR)_C220S_IgG1_G236R/L328R, Light chain (OKT4A_L1) in FIG. 30A-30FF.
[0073] In a further aspect, the present invention provides a method for treating autoimmune disease in a subject that includes administering a heterodimeric protein to the subject, where the heterodimeric protein includes: (a) a first monomer with (i) a first heavy chain constant region comprising a first variant Fc domain; (ii) an IL-2 protein; and (b) a second monomer with: (i) a second heavy chain constant region comprising a second variant Fc domain; (ii) a member selected from the group consisting of: an anti-CD4 binding moiety, an anti-CD8 binding moiety, an anti-CTLA4 binding moiety, an anti-CCR4 binding moiety, an anti-PD-1 binding moiety, and an anti-GITR binding moiety. In further embodiments, the first variant Fc domain has a different amino acid sequence than the second variant Fc domain. Administration of this heterodimeric protein stimulates the T cells. As will be appreciated, the IL2 protein may comprise a full length protein or a portion of the full length IL2 protein. In further embodiments, the full or portion of the IL2 protein that is part of the heterodimeric protein comprises a human IL2 protein sequence.
[0074] In a further embodiment and in accordance with the above, the T cells are regulatory T cells and the second monomer is the anti-CD4 binding moiety.
[0075] In still further embodiments and in accordance with any of the above, the second monomer further includes: (a) the second heavy chain constant region further having a heavy chain variable domain, and (b) a light chain sequence, where the heavy chain variable domain and the light chain sequence together form antigen binding moiety, including without limitation the anti-CD4 binding moiety.
[0076] In still further embodiments and in accordance with any of the above, the stimulated T cells are cytotoxic T cells and the second monomer comprises the anti-CD8 binding moiety. In yet further embodiments, the anti-CD8 binding moiety comprises all or a portion of an antigen binding region of an antibody selected from the group consisting of MCD8, 3B5, Sk1, OKT-8, and DK-25.
[0077] In still further embodiments and in accordance with any of the above, the first and second variant Fc domains include a set of amino acid substitutions selected from the group consisting of those sets depicted in FIG. 33, 34 or 35.
[0078] In still further embodiments and in accordance with any of the above, the first and/or second variant Fc domain comprises an amino acid variant selected from the group consisting of: 236R, 239D, 239E, 243L, M252Y, V259I, 267D, 267E, 298A, V308F, 328F, 328R, 330L, 332D, 332E, M428L, N434A, N434S, 236R/328R, 239D/332E, M428L, 236R/328F, V259I/V308F, 267E/328F, M428L/N434S, Y436I/M428L, Y436V/M428L, Y436I/N434S, Y436V/N434S, 239D/332E/330L, M252Y/S254T/T256E, V259I/V308F/M428L, and E233P/L234V/L235A/G236del/S267K.
[0079] In still further embodiments and in accordance with any of the above, the heterodimeric protein comprise a sequence selected from the sequences depicted in FIGS. 30A-31OOO.
[0080] In still further embodiments and in accordance with any of the above, the first monomer comprises a sequence according to the sequence designated as 13027-hIL2_OKT4A_H1L1_IgG1_pI_ISO(-/+RR)_C220S_G236R/L328R, Heavy chain 1 (hIL2_IgG1_pI_ISO(-)_C220S/G236R/L328R) in FIG. 30A-30FF.
[0081] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 13027-hIL2_OKT4A_H1L1_IgG1_pI_ISO(-/+RR)_C220S_G236R/L328R, Heavy chain 2 (OKT4A_H1_IgG1_pI_ISO(+RR)_G236R/L328R) in FIG. 30A-30FF.
[0082] In still further embodiments and in accordance with any of the above, the second monomer further comprises a sequence according to the sequence designated as 13027-hIL2_OKT4A_H1L1_IgG1_pI_ISO(-/+RR)_C220S_G236R/L328R, Light chain (OKT4A_L1) in FIG. 30A-30FF.
[0083] In still further embodiments and in accordance with any of the above, the second monomer comprises a sequence according to the sequence designated as 13038-OKT4A_H1L1_IgG1_G236R/L328R_hIL2(2), Heavy chain (OKT4A_H1_IgG1_G236R/L328R_hIL2) in FIG. 30A-30FF.
[0084] In still further embodiments and in accordance with any of the above, the second monomer further comprises a sequence according to the sequence designated as 13038-OKT4A_H1L1_IgG1_G236R/L328R_hIL2(2), Light chain (OKT4A_L1) in FIG. 30A-30FF.
BRIEF DESCRIPTION OF THE DRAWINGS
[0085] FIG. 1. Diagram illustrating suppression of Treg cells with anti-CD4.times.anti-CD25 bispecifics.
[0086] FIG. 2. Evaluation of the ability of various anti-CD25 heavy chains to pair with anti-CD4 light chains and anti-CD4 heavy chains to pair with anti-CD25 light chains. Biacore was used to examine binding of the various pairs to both CD4 and CD25 and the results tabulated. The HuMax-TAC anti-CD25 heavy chain has the unique ability to pair with the anti-CD4 lights chains of OKT4A and zanolimumab, with the HuMax-TAC/OKT4A pair showing the strongest binding.
[0087] FIG. 3. Diagram showing three exemplary anti-CD4.times.anti-CD25 bispecific formats. Common light-chain, dual scFv, and Fab/scFv-Fc formats are shown. Purification of heterodimer formats is accomplished utilizing Protein A and IEX chromatography. IgG1, FcR enhanced, and/or FcR knockout Fc regions may be used in further embodiments of the invention.
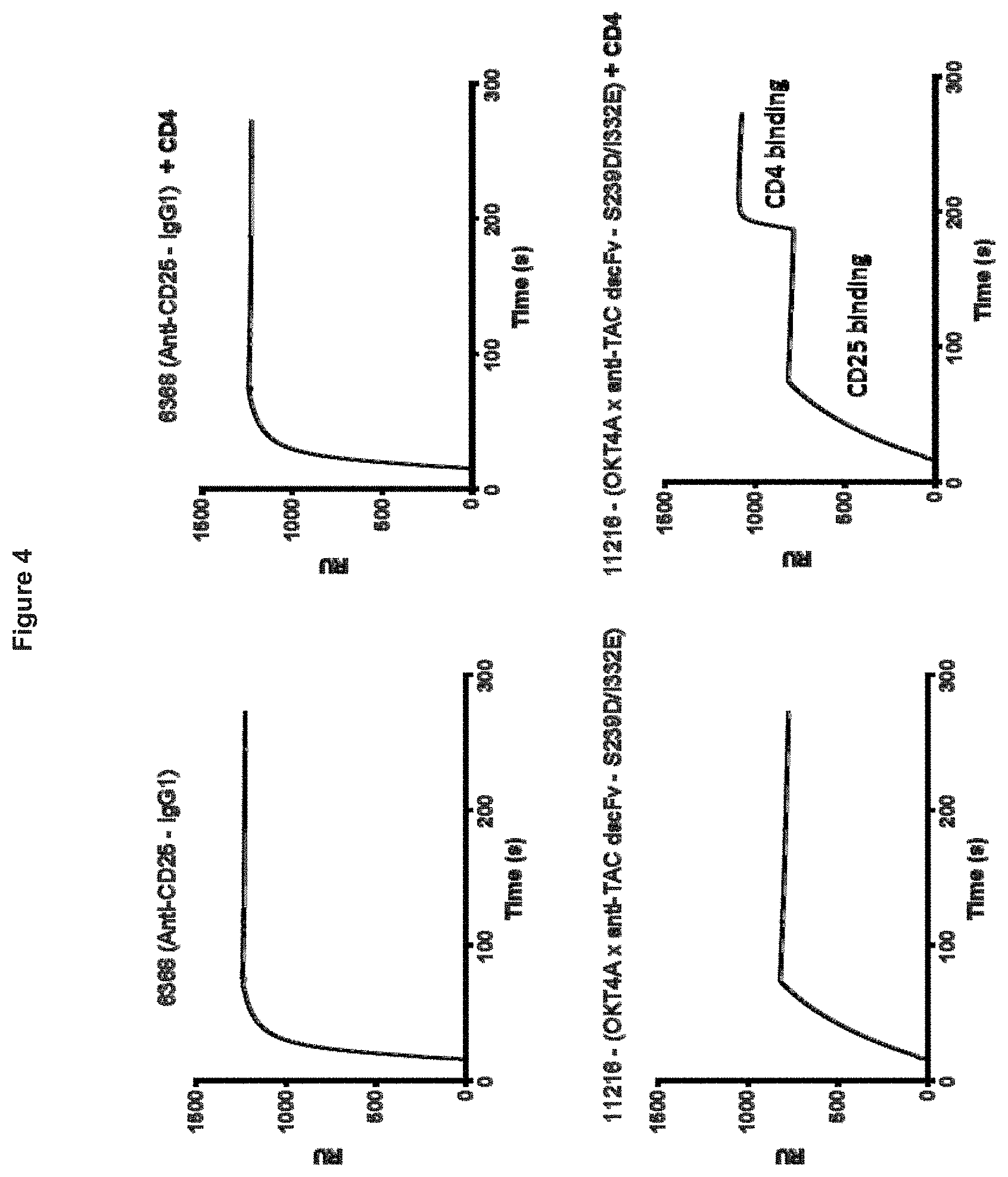
[0088] FIG. 4. Dual scFv-Fc bispecific antibody OKT4A_HOL0_scFv_Anti-TAC_H1L1_scFv_GDQ-Fc(216)_IgG1_pI_ISO(-)/pI_ISO(+RR)- _IgG1_S239D/1332E can bind to CD25 and CD4 simultaneously. The antibody was bound to a CD25 chip on Biacore followed by binding of CD4. As a control, anti-CD25 antibody Anti-TAC_H1L1_IgG1 does not bind CD4.
[0089] FIG. 5. Suppression of Treg cells by anti-CD4.times.anti-CD25 bispecifics. Proliferation of Tregs was assayed using CFSE in the presence of bispecific or control antibodies with 15 U/mL IL2.
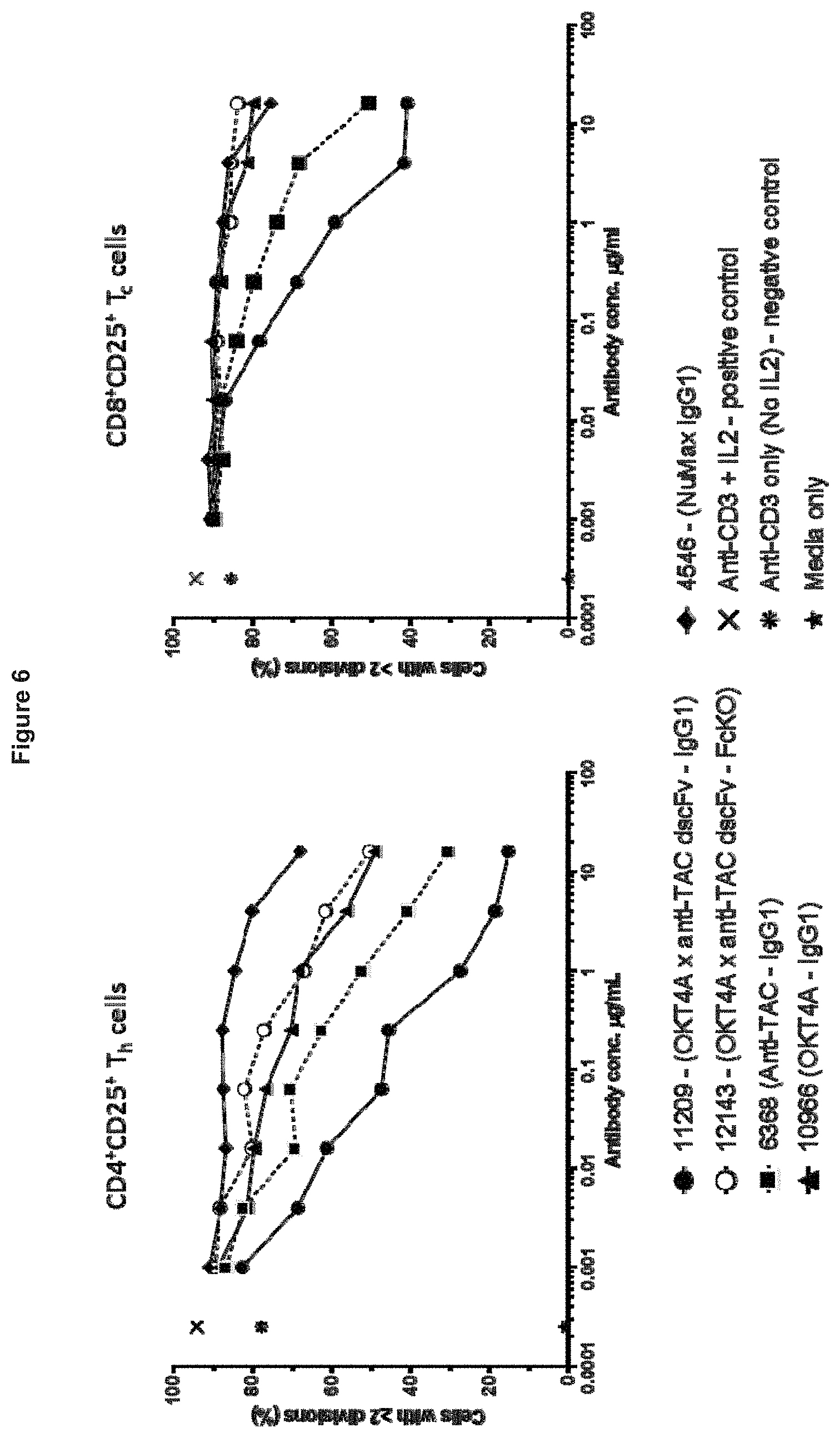
[0090] FIG. 6. Effect of anti-CD4.times.anti-CD25 bispecifics on helper (CD4.sup.+CD25.sup.+) and cytotoxic (CD8.sup.+CD25.sup.+) T cell populations.
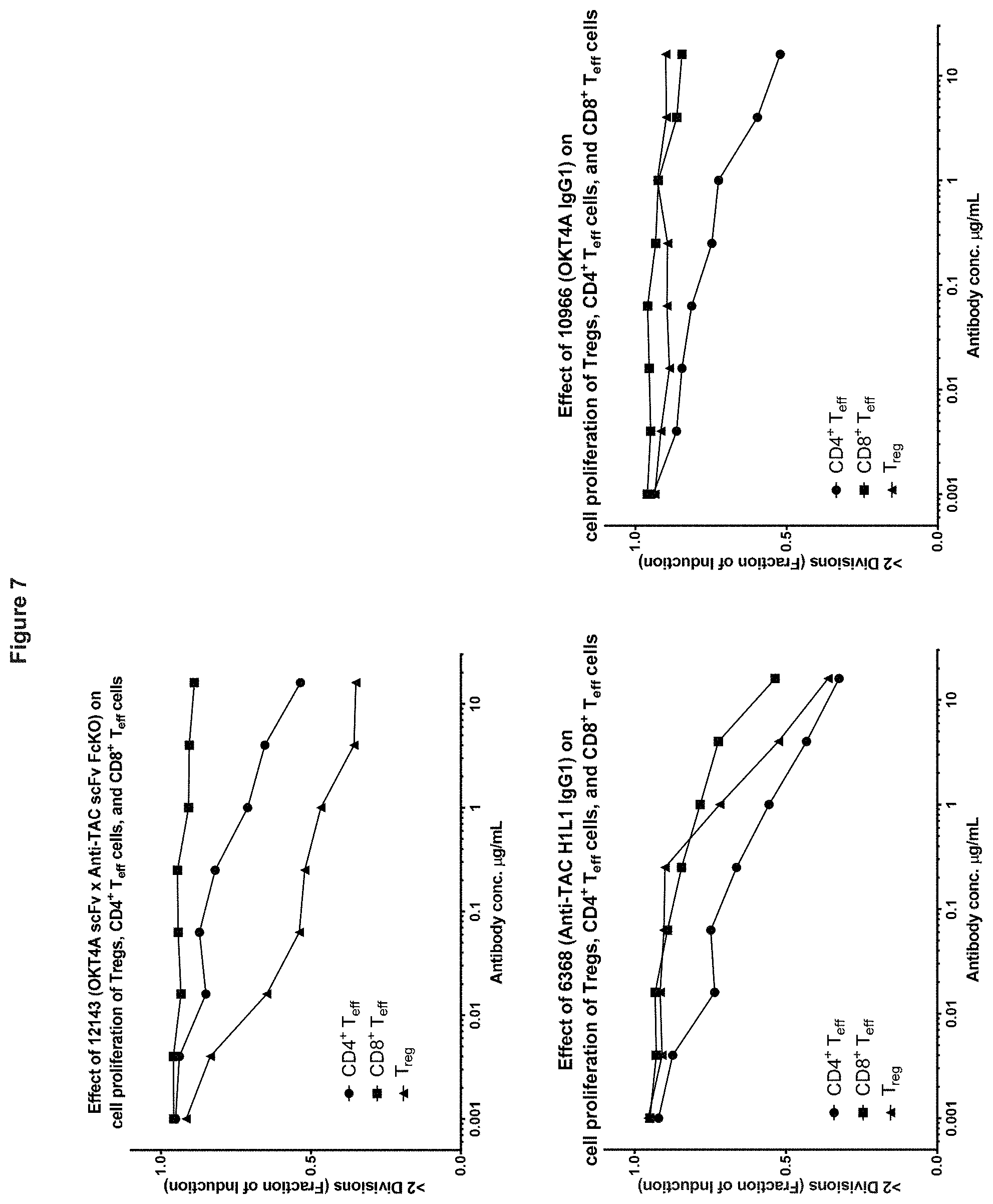
[0091] FIG. 7. Effect of bispecific antibodies and control anti-CD25 and anti-CD4 antibodies on cell proliferation of Tregs, CD4+ T-effectors, and CD8+ T-effectors. Bispecific antibody 12143 has higher potency on Tregs compared to controls as well as lower potency compared to controls on unwanted suppression of T effector cells.
[0092] FIG. 8. Direct binding of anti-CD4.times.anti-CD25 bispecifics and control antibodies on Treg cells.
[0093] FIG. 9. Direct binding of anti-CD4.times.anti-CD25 bispecifics and control antibodies on Treg cells. Variants engineered for altered CD25 affinity are shown.
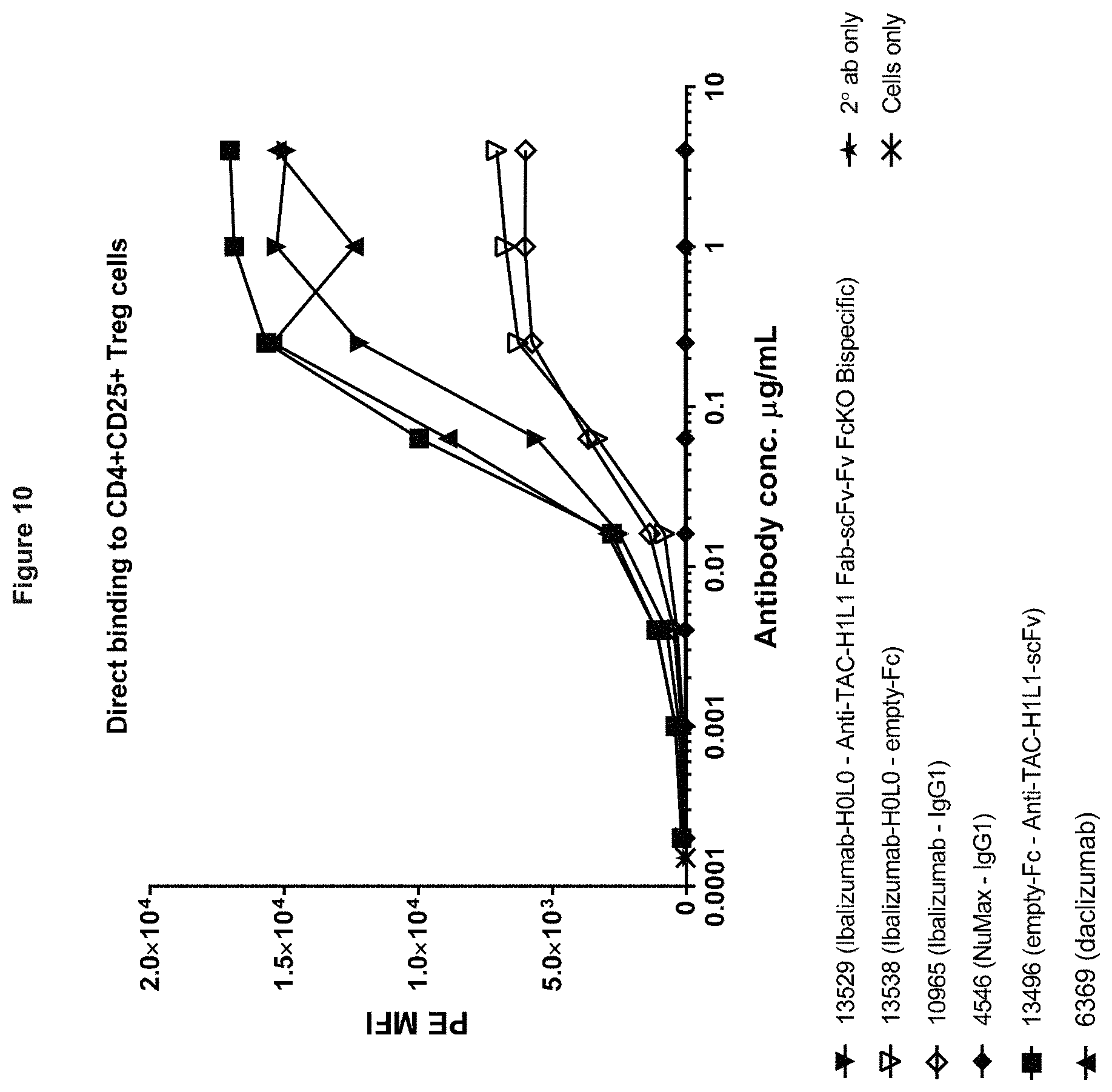
[0094] FIG. 10. Direct binding of anti-CD4.times.anti-CD25 bispecifics and control antibodies on Treg cells. Variants containing the ibalizumab anti-CD4 Fv are shown.
[0095] FIG. 11. Direct binding of anti-CD4.times.anti-CD25 bispecifics and control antibodies on Treg cells.
[0096] FIG. 12. Suppression of Treg cells by anti-CD4.times.anti-CD25 bispecifics. Proliferation of Tregs was assayed using CFSE in the presence of bispecific or control antibodies with 15 U/mL IL2.
[0097] FIG. 13. Suppression of Treg cells by anti-CD4.times.anti-CD25 bispecifics. Proliferation of Tregs was assayed using Alamar Blue in the presence of bispecific or control antibodies with 15 U/mL IL2.
[0098] FIG. 14. Binding of anti-CD4.times.anti-CD25 bispecifics and control antibodies to purified naive human CD4+ T cells.
[0099] FIG. 15. Effect of altering CD25 binding affinity on suppression of Treg cells by anti-CD4.times.anti-CD25 bispecifics. Proliferation of Tregs was assayed using CFSE in the presence of bispecific or control antibodies with 15 U/mL IL2.
[0100] FIG. 16. Direct binding of altered CD25 affinity anti-CD4.times.anti-CD25 bispecifics to purified naive human CD4+ T cells.
[0101] FIG. 17. Direct binding of anti-CD4.times.anti-CD25 bispecifics and controls to activated T effector cells (CD4+CD25+).
[0102] FIG. 18. Direct binding of altered CD25 affinity anti-CD4.times.anti-CD25 bispecifics to activated T effector cells (CD4+CD25+).
[0103] FIG. 19. Direct binding of anti-CD4.times.anti-CD25 bispecifics and controls to activated T effector cells (CD4+CD25+).
[0104] FIG. 20. Direct binding of anti-CD4.times.anti-CD25 bispecifics and controls to activated T effector cells (CD8+CD25+).
[0105] FIG. 21. Direct binding of altered CD25 affinity anti-CD4.times.anti-CD25 bispecifics to activated T effector cells (CD8+CD25+).
[0106] FIG. 22. Direct binding of anti-CD4.times.anti-CD25 bispecifics and controls to activated T effector cells (CD8+CD25+).
[0107] FIG. 23. Summary of IL2 variants that can be used for suppression or induction of Tregs.
[0108] FIG. 24. Diagram illustrating induction of Treg cells with anti-CD4.times.IL2 Fc-fusions. An example construct is also shown.
[0109] FIG. 25. Exemplary IL2 Fc-fusions and bispecific antibody-IL2 Fc-fusions for induction of Tregs.
[0110] FIG. 26. Purification and analysis of anti-CD4.times.IL2 Fc-fusions. Fc-fusions are purified by Protein A and IEX chromatography, and purity assessed by SEC and SDS-PAGE.
[0111] FIG. 27. Induction of regulatory T cells (Tregs) by anti-CD4.times.IL2 Fc-fusions. Induction of Tregs was assayed using the alamar blue cell viability assay in the presence of anti-CD4.times.IL2 Fc-fusions or control antibodies.
[0112] FIG. 28. Diagram illustrating suppression of activated cytotoxic (CD8+CD25+) T cells by anti-CD8.times.anti-CD25 bispecific antibodies.
[0113] FIG. 29. Diagram illustrating induction of naive and activated cytotoxic (CD8.sup.+CD25.sup.+) T cells by anti-CD8.times.IL2 Fc-fusions.
[0114] FIG. 30A-30FF. Sequences of anti-CD4.times.anti-CD25 bispecifics, anti-CD4.times.IL2 Fc-fusions, and control antibodies.
[0115] FIG. 31A-31OOO. Sequences of T cell modulating bispecifics, Fc-fusions, and control antibodies.
[0116] FIG. 32. Table of exemplary Treg markers for use in embodiments of the invention.
[0117] FIG. 33A-33C. Table of exemplary amino acid variants for embodiments of heterodimeric proteins of the invention.
[0118] FIG. 34A-34C. Table of exemplary amino acid variants for embodiments of heterodimeric proteins of the invention.
[0119] FIG. 35. Table of exemplary amino acid variants for embodiments of heterodimeric proteins of the invention.
[0120] FIG. 36A-36M. Illustration of a number of heterodimeric protein formats, including heterodimeric Fc fusion proteins as well as heterodimeric antibodies. FIG. 36A shows the basic concept of a dimeric Fc region with four possible fusion partners A, B, C and D. A, B, C and D are optionally and independently selected from immunoglobulin domain(s) (e.g. Fab, vH, vL, scFv, scFv2, scFab, dAb, etc.), peptide(s), cytokines (e.g. IL-2, IL-10, IL-12, GCSF, GM-CSF, etc.), chemokine(s) (e.g. RANTES, CXCL9, CXCL10, CXCL12, etc.), hormone(s) (e.g. FSH, growth hormone), immune receptor(s) (e.g. CTLA-4, TNFR1, TNFRII, other TNFSF, other TNFRSF, etc.) and blood factor(s) (e.g. Factor VII, Factor VIII, Factor IX, etc.). Domains filled with solid white or solid black are engineered with heterodimerization variants as outlined herein. FIG. 36B depicts the "triple F" format (sometimes also referred to as the "bottle-opener" configuration as discussed below). FIG. 36C shows a "triple F" configuration with another scFv attached to the Fab monomer (this one, along with FIG. 36F, has a greater molecular weight differential as well). FIG. 36D depicts a "triple F" with another scFv attached to the scFv monomer. FIG. 36E depicts a "three scFv" format. FIG. 36F depicts an additional Fab attached to the Fab monomer. FIG. 36G depicts a Fab hooked to one of the scFv monomers. FIGS. 1H-1L show additional varieties of "higher multispecificity" embodiments of the "triple F" format, all with one monomer comprising an scFv (and all of which have molecular weight differentials which can be exploited for purification of the heterodimers). FIG. 36H shows a "Fab-Fv" format with binding to two different antigens, with FIG. 361 depicting the "Fab-Fv" format with binding to a single antigen (e.g. bivalent binding to antigen 1). FIGS. 36J and 36K depicts a "Fv-Fab" format with similar bivalent or monovalent additional antigen binding. FIG. 36L depicts one monomer with a CH1-CL attached to the second scFv. FIG. 36M depicts a dual scFv format.
[0121] FIG. 37A-37U. Depicts a wide variety of the multispecific (e.g. heterodimerization) formats and the combinations of different types of heterodimerization variants that can be used in the present invention (these are sometimes referred to herein as "heterodimeric scaffolds"). Note in addition that all of these formats can include addition variants in the Fc region, as more fully discussed below, including "ablation" or "knock out" variants (FIG. 39), Fc variants to alter Fc.gamma.R binding (Fc.gamma.RIIb, Fc.gamma.RIIIa, etc.), Fc variants to alter binding to FcRn receptor, etc. FIG. 37A shows a dual scFv-Fc format, that, as for all heterodimerization formats herein can include heterodimerization variants such as pI variants, knobs in holes (KIH, also referred to herein as steric variants or "skew" variants), charge pairs (a subset of steric variants), isosteric variants, and SEED body ("strand-exchange engineered domain"; see Klein et al., mAbs 4:6 653-663 (2012) and Davis et al, Protein Eng Des Sel 2010 23:195-202) which rely on the fact that the CH3 domains of human IgG and IgA do not bind to each other. FIG. 37B depicts a bispecific IgG, again with the option of a variety of heterodimerization variants. FIG. 37C depicts the "one armed" version of DVD-Ig which utilizes two different variable heavy and variable light domains. FIG. 37D is similar, except that rather than an "empty arm", the variable heavy and light chains are on opposite heavy chains. FIG. 37E is generally referred to as "mAb-Fv". FIG. 37F depicts a multi-scFv format; as will be appreciated by those in the art, similar to the "A, B, C, D" formats discussed herein, there may be any number of associated scFvs (or, for that matter, any other binding ligands or functionalities). Thus, FIG. 37F could have 1, 2, 3 or 4 scFvs (e.g. for bispecifics, the scFv could be "cis" or "trans", or both on one "end" of the molecule). FIG. 37G depicts a heterodimeric FabFc with the Fab being formed by two different heavy chains one containing heavy chain Fab sequences and the other containing light chain Fab sequences. FIG. 37H depicts the "one armed Fab-Fc", where one heavy chain comprises the Fab. FIG. 37I depicts a "one armed scFv-Fc", wherein one heavy chain Fc comprises an scFv and the other heavy chain is "empty". FIG. 37J shows a scFv-CH3, wherein only heavy chain CH3 regions are used, each with their own scFv. FIG. 37K depicts a mAb-scFv, wherein one end of the molecule engages an antigen bivalently with a monovalent engagement using an scFv on one of the heavy chains. FIG. 37L depicts the same structure except that both heavy chains comprise an additional scFv, which can either bind the same antigen or different antigens. FIG. 37M shows the "CrossMab" structure, where the problem of multiplex formation due to two different light chains is addressed by switching sequences in the Fab portion. FIG. 37N depicts an scFv, FIG. 370 is a "BITE" or scFv-scFv linked by a linker as outlined herein, FIG. 37P depicts a DART, FIG. 37Q depicts a TandAb, and FIG. 37R shows a diabody. FIGS. 37S, 37T and 37U depict additional alternative scaffold formats that find use in the present invention.
[0122] FIG. 38. Depicts a list of isotypic and isosteric variant antibody constant regions and their respective substitutions. pI_(-) indicates lower pI variants, while pI_(+) indicates higher pI variants. These can be optionally and independently combined with other heterodimerization variants of the invention.
[0123] FIG. 39. Depicts a number of suitable "knock out" ("KO") variants to reduce binding to some or all of the Fc.gamma.R receptors. As is true for many if not all variants herein, these KO variants can be independently and optionally combined, both within the set described in FIG. 39 and with any heterodimerization variants outlined herein, including steric and pI variants. For example, E233P/L234V/L235A/G236del can be combined with any other single or double variant from the list. In addition, while it is preferred in some embodiments that both monomers contain the same KO variants, it is possible to combine different KO variants on different monomers, as well as have only one monomer comprise the KO variant(s). Reference is also made to the Figures and Legends of U.S. Ser. No. 61/913,870, all of which is expressly incorporated by reference in its entirety as it relates to "knock out" or "ablation" variants.
[0124] FIG. 40. Depicts a number of charged scFv linkers that find use in increasing or decreasing the pI of heterodimeric proteins that utilize one or more scFv as a component. A single prior art scFv linker with a single charge is referenced as "Whitlow", from Whitlow et al., Protein Engineering 6(8):989-995 (1993). It should be noted that this linker was used for reducing aggregation and enhancing proteolytic stability in scFvs.
[0125] FIG. 41A-41B. FIGS. 41A and 41B provides an additional list of potential heterodimerization variants for use in the present invention, including isotypic variants.
[0126] FIG. 42A-42J. Depicts additional exemplary heterodimerization variant pairs for use in heterodimeric proteins of the invention.
[0127] FIG. 43. Depicts amino acid sequences of wild-type constant regions used in the invention.
[0128] FIG. 44. Depicts two different Triple F embodiments for bispecific antibodies of the invention.
[0129] FIG. 45. Literature pIs of the 20 amino acids. It should be noted that the listed pIs are calculated as free amino acids; the actual pI of any side chain in the context of a protein is different, and thus this list is used to show pI trends and not absolute numbers for the purposes of the invention.
[0130] FIG. 46A-46C. List of all possible reduced pI variants created from isotypic substitutions of IgG1-4. Shown are the pI values for the three expected species as well as the average delta pI between the heterodimer and the two homodimer species present when the variant heavy chain is transfected with IgG1-WT heavy chain.
[0131] FIG. 47. List of all possible increased pI variants created from isotypic substitutions of IgG1-4. Shown are the pI values for the three expected species as well as the average delta pI between the heterodimer and the two homodimer species present when the variant heavy chain is transfected with IgG1-WT heavy chain.
[0132] FIG. 48A-48B. Matrix of possible combinations of first and second monomers for heterodimeric proteins of the invention, FcRn variants, Scaffolds, Fc variants and combinations, with each variant being independently and optionally combined from the appropriate source. Note that the target antigens for the first and second monomer are each independently selected from the list provided in the first column. Legend: Legend A are suitable FcRn variants: 434A, 434S, 428L, 308F, 259I, 428L/434S, 259I/308F, 436I/428L, 436I or V/434S, 436V/428L, 252Y, 252Y/254T/256E, 259I/308F/428L. Legend B are suitable scaffolds and include IgG1, IgG2, IgG3, IgG4, and IgG1/2. Sequences for such scaffolds can be found for example in US Patent Publication No. 2012/0128663, published on May 24, 2012, which is hereby incorporated by reference in its entirety for all purposes and in particular for all teachings, figures and legends related to scaffolds and their sequences. Legend C are suitable Fc variants: 236A, 239D, 239E, 332E, 332D, 239D/332E, 267D, 267E, 328F, 267E/328F, 236A/332E, 239D/332E/330Y, 239D, 332E/330L, 236R, 328R, 236R/328R, 236N/267E, 243L, 298A and 299T. (Note, additional suitable Fc variants are found in FIG. 41 of US 2006/0024298, the figure and legend of which are hereby incorporated by reference in their entirety). Legend D reflects the following possible combinations, again, with each variant being independently and optionally combined from the appropriate source Legend: 1) Monomer targets (each independently selected from the list in the first column) plus FcRn variants; 2) Monomer targets (each independently selected from the list in the first column) plus FcRn variants plus Scaffold; 3) Monomer targets (each independently selected from the list in the first column) plus FcRn variants plus Scaffold plus Fc variants; 4) Monomer targets (each independently selected from the list in the first column) plus Scaffold 5) Monomer targets (each independently selected from the list in the first column) plus Fc variants; 6) FcRn variants plus Scaffold; 7) Monomer targets (each independently selected from the list in the first column) plus Fc variants; 8) Scaffold plus Fc variants; 9) Monomer targets (each independently selected from the list in the first column) plus Scaffold plus Fc variants; and 10) Monomer targets (each independently selected from the list in the first column) plus FcRn variants plus Fc variants.
DETAILED DESCRIPTION OF EXEMPLARY EMBODIMENTS
Overview of Invention
[0133] The present invention provides methods and compositions for modulating T cells by administering heterodimeric proteins. Heterodimeric proteins include without limitation heterodimeric antibodies (such as bispecific antibodies) and heterodimeric fusion proteins. By "modulating T cells" as discussed herein is meant suppressing or stimulating T cells. In general, the heterodimeric proteins of the invention are specific for their target T cell, meaning that the heterodimeric proteins primarily affect one type of T cell with little or no impact on other T cell types. For example (and as is described in further detail herein), methods and compositions of the present invention for suppression or induction of regulatory T cells (also referred to herein as "Tregs") have little or no impact on other T cell types. Similarly, methods and compositions of the invention for suppressing or inducing other T cell types, such as cytotoxic T cells, primarily affect the cytotoxic T cells with little or no impact on other T cell types.
[0134] By "suppressing T cells" as used herein refers to decreasing any aspect of T cell expression or function as compared to expression or function in the absence of the administered heterodimeric protein. For example, suppression of regulatory T cells by admiinistering a heterodimeric protein includes suppression of the proliferation of regulatory T cells as compared to proliferation in the absence of the administered heterodimeric protein. "Inducing T cells" as used herein refers to increasing any aspect of T cell expression or function as compared to expression or function in the absence of the administered heterodimeric protein, including stimulation of the proliferation of the target T cell.
[0135] As discussed in further detail herein, suppression or induction of T cells can be measured with assays to quantify T cell numbers. For example, cell proliferation assays can be used to detect and quantitate T cells. Other methods of quantifying T cells, particularly Tregs, may also be used, including methods utilizing qPCR to measure the amount of demethylated FOXP3 (a Treg marker) that is present. Such assays are described for example in Wieczorek et al., 2009, Cancer Res, 69(2): 599-608, Vries et al., 2011, Clin Cancer Res, 17:841-848, and Baron et al., 2007, Eur. J. Immunol., 37:2378-2389, each of which is hereby incorporated by reference in its entirety for all purposes and in particular for all teachings, figures and legends related to assays for FOXP3, demethylated FOXP3, and quantification of Tregs.
[0136] Suppression of T cells of a particular type is generally accomplished by administering a heterodimeric protein that targets antigens specific for that T cell type. For example, for regulatory T cells, administering a heterodimeric protein that targets both CD4 and CD25 reduces regulatory T cell proliferation with minimal to no effect on other T cells, such as CD4+CD25- T effector cells or CD8+CD25+ cyotoxic T cells. For suppression of T cells, such a heterodimeric protein is generally a bispecific antibody, although other multispecific antibodies and other heterodimeric proteins such as fusion proteins are also contemplated. In certain instances, suppression of regulatory T cells is accomplished by administering a bispecific antibody that targets both CD4 and CD25; in other words, the bispecific antibody comprises two monomers in which one monomer comprises an anti-CD4 binding domain and the other monomer comprises an anti-CD25 binding domain ("binding domain" and "binding moiety" are used interchangeably herein). "Anti-X binding domain" refers to a domain of the monomer that binds to X (i.e., an anti-CD25 binding domain is a part of the monomer that binds to CD25).
[0137] Other bispecific antibodies that suppress Tregs include without limitation anti-CD25.times.anti-CTLA4, anti-CD25.times.anti-PD-1, anti-CD25.times.anti-CCR4, and anti-CD25.times.anti-G ITR antibodies.
[0138] Bispecific antibodies can also be used to suppress other T cell types, such as cytotoxic T cells. In some instances, one monomer of the bispecific antibody comprises an anti-CD8 domain, including without limitation domains from anti-CD8 antibodies such as MCD8, 3B5, SK1, OKT-8, 51.1 and DK-25. The monomer with the anti-CD8 domain can be combined with a monomer comprising an anti-CD25 binding domain to produce a bispecific antibody for suppression of cytoxic T cells.
[0139] Generally, the antigen binding domains of bispecific antibodies of the invention are part of monomers that further comprise at least a heavy chain constant region that contains a variant Fc domain as compared to a parent Fc domain.
[0140] In some situations, anti-CD4 and anti-CD8 targeting agents may further be utilized in combination with T cell cytokines, including without limitation IL-7, IL-12, IL-15, and IL-17.
[0141] Fc fusion proteins may also be used in accordance with the invention to suppress T cells. For example, a fusion protein comprising an IL2 protein on one arm can be engineered to have reduced ability to bind to IL2R.beta., IL2R.gamma., and or IL2R.alpha. in order to ablate IL2 receptor signaling. When coupled with an anti-CD4 antibody (or any other Treg surface marker antibody), this results in an anti-CD4.times.IL2 Fc-fusion capable of suppressing Treg cells through targeted binding to CD4 and CD25, but without the ability to induce Treg proliferation. In one non-limiting theory, the mechanism of action for this fusion may be that it blocks endogenous IL2 from binding to receptor, thus preventing Treg proliferation. Exemplary embodiments of such fusion proteins are provided in FIG. 23.
[0142] Heterodimeric proteins may also be used to induce T cells. As with methods and compositions for suppressing T cells, induction of T cells in accordance with the present invention is generally accomplished by administering a heterodimeric protein that targets antigens and proteins specific for that T cell type. In specific instances, the present invention provides Fc fusion proteins comprising one monomer with a binding domain that targets a T cell marker and a second monomer comprising an IL2 protein. Examples of fusion proteins of use in the present invention for inducing T cells include without limitation fusion proteins that comprise IL2 on one monomer and one of the following binding domains on the other monomer: anti-CD4, anti-CCR4, anti-PD-1, anti-CD8, LAG3, and anti-CTLA4. In some situations, potency of the fusion proteins is increased by engineering the IL2 arm to increase the affinity of IL2 for IL2R.alpha.. Exemplary variants of IL2 of use in the present invention are listed in FIG. 23. Other cytokines that may be used in Fc fusion proteins of the invention include without limitation IL-7, IL-12, IL-15, and IL-17.
[0143] As will be appreciated and as is described in further detail herein, the heterodimeric proteins discussed herein may comprise a variety of formats, including those described herein (see for example FIGS. 3, 25, 36 and 37) and those described in the art (see for example Kontermann et al., 2012, Landes Bioscience, which is incorporated herein by reference for all purposes and in particular for all teachings related to heterodimeric proteins such as bispecific antibodies). In some situations, bispecific antibodies may have one heavy chain containing a single chain Fv ("scFv", as defined herein) and the other heavy chain is a "regular" FAb format, comprising a variable heavy chain and a light chain. This structure is sometimes referred to herein as "triple F" format (scFv-FAb-Fc) or the "bottle-opener" format, due to a rough visual similarity to a bottle-opener, as described for example in U.S. Ser. No. 14/205,248, filed Mar. 11, 2014, which is hereby incorporated by reference for all purposes and in particular for all teachings related to the triple F or bottle opener format. In some situations, both of the heavy chains of the bispecific antibodies described herein contain scFvs. Similarly, for any of the fusion proteins described herein, the antibody arm of the fusion protein may be in the scFv or regular FAb format.
[0144] As is discussed in further herein, the heterodimeric proteins of the present invention may further include one or more amino acid substitutions in the Fc region that have the effect of increasing serum half-life, ablating binding to Fc.quadrature.R, and/or increasing ADCC. The heterodimeric proteins of the invention may also further include "heterodimerization variants" that, as is also described in further detail herein, promote heterodimeric formation and/or allow for ease of purification of heterodimers over the homodimers. In certain situations, the heterodimeric proteins of the invention comprise one or more variant Fc domains comprising an amino acid variant selected from among the variants listed in FIGS. 33 and 34. In some situations, the amino acid variants may further comprise variants selected from the group: 236R; 239D; 239E; 243L; M252Y; V259I; 267D; 267E; 298A; V308F; 328F; 328R; 330L; 332D; 332E; M428L; N434A; N434S; 236R/328R; 239D/332E; M428L; 236R/328F; V259I/V308F; 267E/328F; M428L/N434S; Y436I/M428L; Y436V/M428L; Y436I/N434S; Y436V/N434S; 239D/332E/330L; M252Y/S254T/T256E; V259I/V308F/M428L; and E233P/L234V/L235A/G236del/S267K.
[0145] The methods and compositions of the present invention further include methods for treating and/or alleviating the symptoms of diseases and disorders affected by T cells, including without limitation cancer and autoimmune disease. In particular, methods and compositions of the present invention for the suppression of T cells, particularly Tregs, can be used to treat cancer. In addition, methods and compositions of the present invention for stimulation of T cells can be used to treat autoimmune disease.
[0146] As will be appreciated, suppression of T cells in accordance with the present invention may be used to treat any type of cancer. Bispecific antibodies targeting both CD4 and CD25 (or any other combination of T cell markers as described herein and listed in FIG. 32) may in certain further embodiments be beneficial for the treatment of adult T cell leukemia (ATL), a rare disease associated with human T cell lymphotrophic virus (HTLV-1). Diseased cells from ATL patients function as regulatory cells and may arise from Treg cells, since these cells display a CD4.sup.+CD25.sup.+ phenotype consistent with that of Treg cells. Depletion of tumor cells with anti-CD4/CD25 bispecific antibodies coupled to an enhanced effector function Fc domain may be a viable treatment option.
[0147] As discussed above, the balance of Treg versus effector T cells can be disregulated in autoimmune disease, and therapeutic approaches to favor higher Treg ratios utilizing methods and compositions of the invention can be of use for treating such diseases. Induction and promotion of T cells in accordance with the methods described herein can also be used to treat (i.e., suppress) anti-graft responses in organ transplant and graft-vs-host disease after allogeneic stem cell or bone marrow transplant. Fc-fusion molecules, which in one non-limiting mechanism may selectively `feed` IL2 to Treg, promote the survival and expansion. Such agents, which should alter the balance in favor of Treg vs effector T cells, may provide a viable treatment option for controlling autoimmune disease, organ transplant rejection, and graft-vs-host disease. In general, such treatments include the use of antibody-IL2 fusion proteins, in particular wherein a single IL2 protein is coupled with an anti-CD4 (or other Treg marker) moiety to provide selectivity for Treg versus effector T cells through the requirement for simultaneous engagement of CD4 and the high-affinity IL-2 receptor CD25.
[0148] Treatment of cancer, autoimmune disease or any other T cell associated disease or disorder in accordance with the present invention generally involves administering a composition containing a heterodimeric protein of the invention (antibody or Fc fusion) to a patient in need thereof.
Definitions
[0149] In order that the application may be more completely understood, several definitions are set forth below. Such definitions are meant to encompass grammatical equivalents.
[0150] By "ablation" herein is meant a decrease or removal of activity. Thus for example, "ablating Fc.gamma.R binding" means the Fc region amino acid variant has less than 50% starting binding as compared to an Fc region not containing the specific variant, with less than 70-80-90-95-98% loss of activity being preferred, and in general, with the activity being below the level of detectable binding in a Biacore assay. Of particular use in the ablation of Fc.gamma.R binding are those shown in FIG. 7.
[0151] By "ADCC" or "antibody dependent cell-mediated cytotoxicity" as used herein is meant the cell-mediated reaction wherein nonspecific cytotoxic cells that express Fc.gamma.Rs recognize bound antibody on a target cell and subsequently cause lysis of the target cell. ADCC is correlated with binding to Fc.gamma.RIIIa; increased binding to Fc.gamma.RIIIa leads to an increase in ADCC activity.
[0152] By "ADCP" or antibody dependent cell-mediated phagocytosis as used herein is meant the cell-mediated reaction wherein nonspecific cytotoxic cells that express Fc.gamma.Rs recognize bound antibody on a target cell and subsequently cause phagocytosis of the target cell.
[0153] By "modification" herein is meant an amino acid substitution, insertion, and/or deletion in a polypeptide sequence or an alteration to a moiety chemically linked to a protein. For example, a modification may be an altered carbohydrate or PEG structure attached to a protein. By "amino acid modification" herein is meant an amino acid substitution, insertion, and/or deletion in a polypeptide sequence. For clarity, unless otherwise noted, the amino acid modification is always to an amino acid coded for by DNA, e.g. the 20 amino acids that have codons in DNA and RNA.
[0154] By "amino acid substitution" or "substitution" herein is meant the replacement of an amino acid at a particular position in a parent polypeptide sequence with a different amino acid. In particular, in some embodiments, the substitution is to an amino acid that is not naturally occurring at the particular position, either not naturally occurring within the organism or in any organism. For example, the substitution E272Y refers to a variant polypeptide, in this case an Fc variant, in which the glutamic acid at position 272 is replaced with tyrosine. For clarity, a protein which has been engineered to change the nucleic acid coding sequence but not change the starting amino acid (for example exchanging CGG (encoding arginine) to CGA (still encoding arginine) to increase host organism expression levels) is not an "amino acid substitution"; that is, despite the creation of a new gene encoding the same protein, if the protein has the same amino acid at the particular position that it started with, it is not an amino acid substitution.
[0155] By "amino acid insertion" or "insertion" as used herein is meant the addition of an amino acid sequence at a particular position in a parent polypeptide sequence. For example, -233E or 233E designates an insertion of glutamic acid after position 233 and before position 234. Additionally, -233ADE or A233ADE designates an insertion of AlaAspGlu after position 233 and before position 234.
[0156] By "amino acid deletion" or "deletion" as used herein is meant the removal of an amino acid sequence at a particular position in a parent polypeptide sequence. For example, E233- or E233# or E233( ) designates a deletion of glutamic acid at position 233. Additionally, EDA233- or EDA233# designates a deletion of the sequence GluAspAla that begins at position 233.
[0157] By "variant protein" or "protein variant", or "variant" as used herein is meant a protein that differs from that of a parent protein by virtue of at least one amino acid modification. Protein variant may refer to the protein itself, a composition comprising the protein, or the amino sequence that encodes it. Preferably, the protein variant has at least one amino acid modification compared to the parent protein, e.g. from about one to about seventy amino acid modifications, and preferably from about one to about five amino acid modifications compared to the parent. As described below, in some embodiments the parent polypeptide, for example an Fc parent polypeptide, is a human wild type sequence, such as the Fc region from IgG1, IgG2, IgG3 or IgG4, although human sequences with variants can also serve as "parent polypeptides", for example the IgG1/2 hybrid of FIG. 13. The protein variant sequence herein will preferably possess at least about 80% identity with a parent protein sequence, and most preferably at least about 90% identity, more preferably at least about 95-98-99% identity. Variant protein can refer to the variant protein itself, compositions comprising the protein variant, or the DNA sequence that encodes it. Accordingly, by "antibody variant" or "variant antibody" as used herein is meant an antibody that differs from a parent antibody by virtue of at least one amino acid modification, "IgG variant" or "variant IgG" as used herein is meant an antibody that differs from a parent IgG (again, in many cases, from a human IgG sequence) by virtue of at least one amino acid modification, and "immunoglobulin variant" or "variant immunoglobulin" as used herein is meant an immunoglobulin sequence that differs from that of a parent immunoglobulin sequence by virtue of at least one amino acid modification. "Fc variant" or "variant Fc" as used herein is meant a protein comprising an amino acid modification in an Fc domain. The Fc variants of the present invention are defined according to the amino acid modifications that compose them. Thus, for example, N434S or 434S is an Fc variant with the substitution serine at position 434 relative to the parent Fc polypeptide, wherein the numbering is according to the EU index. Likewise, M428L/N434S defines an Fc variant with the substitutions M428L and N434S relative to the parent Fc polypeptide. The identity of the WT amino acid may be unspecified, in which case the aforementioned variant is referred to as 428L/434S. It is noted that the order in which substitutions are provided is arbitrary, that is to say that, for example, 428L/434S is the same Fc variant as M428L/N434S, and so on. For all positions discussed in the present invention that relate to antibodies, unless otherwise noted, amino acid position numbering is according to the EU index. The EU index or EU index as in Kabat or EU numbering scheme refers to the numbering of the EU antibody (Edelman et al., 1969, Proc Natl Acad Sci USA 63:78-85, hereby entirely incorporated by reference.) The modification can be an addition, deletion, or substitution. Substitutions can include naturally occurring amino acids and, in some cases, synthetic amino acids. Examples include U.S. Pat. No. 6,586,207; WO 98/48032; WO 03/073238; US2004-0214988A1; WO 05/35727A2; WO 05/74524A2; J. W. Chin et al., (2002), Journal of the American Chemical Society 124:9026-9027; J. W. Chin, & P. G. Schultz, (2002), ChemBioChem 11:1135-1137; J. W. Chin, et al., (2002), PICAS United States of America 99:11020-11024; and, L. Wang, & P. G. Schultz, (2002), Chem. 1-10, all entirely incorporated by reference.
[0158] As used herein, "protein" herein is meant at least two covalently attached amino acids, which includes proteins, polypeptides, oligopeptides and peptides. The peptidyl group may comprise naturally occurring amino acids and peptide bonds, or synthetic peptidomimetic structures, i.e. "analogs", such as peptoids (see Simon et al., PNAS USA 89(20):9367 (1992), entirely incorporated by reference). The amino acids may either be naturally occurring or synthetic (e.g. not an amino acid that is coded for by DNA); as will be appreciated by those in the art. For example, homo-phenylalanine, citrulline, ornithine and noreleucine are considered synthetic amino acids for the purposes of the invention, and both D- and L-(R or S) configured amino acids may be utilized. The variants of the present invention may comprise modifications that include the use of synthetic amino acids incorporated using, for example, the technologies developed by Schultz and colleagues, including but not limited to methods described by Cropp & Shultz, 2004, Trends Genet. 20(12):625-30, Anderson et al., 2004, Proc Natl Acad Sci USA 101 (2):7566-71, Zhang et al., 2003, 303(5656):371-3, and Chin et al., 2003, Science 301(5635):964-7, all entirely incorporated by reference. In addition, polypeptides may include synthetic derivatization of one or more side chains or termini, glycosylation, PEGylation, circular permutation, cyclization, linkers to other molecules, fusion to proteins or protein domains, and addition of peptide tags or labels.
[0159] By "residue" as used herein is meant a position in a protein and its associated amino acid identity. For example, Asparagine 297 (also referred to as Asn297 or N297) is a residue at position 297 in the human antibody IgG1.
[0160] By "Fab" or "Fab region" as used herein is meant the polypeptide that comprises the VH, CH1, VL, and CL immunoglobulin domains. Fab may refer to this region in isolation, or this region in the context of a full length antibody, antibody fragment or Fab fusion protein. By "Fv" or "Fv fragment" or "Fv region" as used herein is meant a polypeptide that comprises the VL and VH domains of a single antibody.
[0161] By "IgG subclass modification" or "isotype modification" as used herein is meant an amino acid modification that converts one amino acid of one IgG isotype to the corresponding amino acid in a different, aligned IgG isotype. For example, because IgG1 comprises a tyrosine and IgG2 a phenylalanine at EU position 296, a F296Y substitution in IgG2 is considered an IgG subclass modification.
[0162] By "non-naturally occurring modification" as used herein is meant an amino acid modification that is not isotypic. For example, because none of the IgGs comprise a serine at position 434, the substitution 434S in IgG1, IgG2, IgG3, or IgG4 (or hybrids thereof) is considered a non-naturally occurring modification.
[0163] By "amino acid" and "amino acid identity" as used herein is meant one of the 20 naturally occurring amino acids that are coded for by DNA and RNA.
[0164] By "effector function" as used herein is meant a biochemical event that results from the interaction of an antibody Fc region with an Fc receptor or ligand. Effector functions include but are not limited to ADCC, ADCP, and CDC.
[0165] By "IgG Fc ligand" as used herein is meant a molecule, preferably a polypeptide, from any organism that binds to the Fc region of an IgG antibody to form an Fc/Fc ligand complex. Fc ligands include but are not limited to Fc.gamma.RIs, Fc.gamma.RIIs, Fc.gamma.RIIIs, FcRn, C1q, C3, mannan binding lectin, mannose receptor, staphylococcal protein A, streptococcal protein G, and viral Fc.gamma.R. Fc ligands also include Fc receptor homologs (FcRH), which are a family of Fc receptors that are homologous to the Fc.gamma.Rs (Davis et al., 2002, Immunological Reviews 190:123-136, entirely incorporated by reference). Fc ligands may include undiscovered molecules that bind Fc. Particular IgG Fc ligands are FcRn and Fc gamma receptors. By "Fc ligand" as used herein is meant a molecule, preferably a polypeptide, from any organism that binds to the Fc region of an antibody to form an Fc/Fc ligand complex.
[0166] By "Fc gamma receptor, "Fc.gamma.R" or "FcqammaR" as used herein is meant any member of the family of proteins that bind the IgG antibody Fc region and is encoded by an Fc.gamma.R gene. In humans this family includes but is not limited to Fc.gamma.RI (CD64), including isoforms Fc.gamma.RIa, Fc.gamma.RIb, and Fc.gamma.RIc; Fc.gamma.RII (CD32), including isoforms Fc.gamma.RIIa (including allotypes H131 and R131), Fc.gamma.RIIb (including Fc.gamma.RIIb-1 and Fc.gamma.RIIb-2), and Fc.gamma.RIIc; and Fc.gamma.RIII (CD16), including isoforms Fc.gamma.RIIIa (including allotypes V158 and F158) and Fc.gamma.RIIIb (including allotypes Fc.gamma.RIIb-NA1 and Fc.gamma.RIIb-NA2) (Jefferis et al., 2002, Immunol Lett 82:57-65, entirely incorporated by reference), as well as any undiscovered human Fc.gamma.Rs or Fc.gamma.R isoforms or allotypes. An Fc.gamma.R may be from any organism, including but not limited to humans, mice, rats, rabbits, and monkeys. Mouse Fc.gamma.Rs include but are not limited to Fc.gamma.RI (CD64), Fc.gamma.RII (CD32), Fc.gamma.RIII (CD16), and Fc.gamma.RIII-2 (CD16-2), as well as any undiscovered mouse Fc.gamma.Rs or Fc.gamma.R isoforms or allotypes.
[0167] By "FcRn" or "neonatal Fc Receptor" as used herein is meant a protein that binds the IgG antibody Fc region and is encoded at least in part by an FcRn gene. The FcRn may be from any organism, including but not limited to humans, mice, rats, rabbits, and monkeys. As is known in the art, the functional FcRn protein comprises two polypeptides, often referred to as the heavy chain and light chain. The light chain is beta-2-microglobulin and the heavy chain is encoded by the FcRn gene. Unless otherwise noted herein, FcRn or an FcRn protein refers to the complex of FcRn heavy chain with beta-2-microglobulin. A variety of FcRn variants used to increase binding to the FcRn receptor, and in some cases, to increase serum half-life, are shown in paragraph [0320] of this specification.
[0168] By "parent polypeptide" as used herein is meant a starting polypeptide that is subsequently modified to generate a variant. The parent polypeptide may be a naturally occurring polypeptide, or a variant or engineered version of a naturally occurring polypeptide. Parent polypeptide may refer to the polypeptide itself, compositions that comprise the parent polypeptide, or the amino acid sequence that encodes it. Accordingly, by "parent immunoglobulin" as used herein is meant an unmodified immunoglobulin polypeptide that is modified to generate a variant, and by "parent antibody" as used herein is meant an unmodified antibody that is modified to generate a variant antibody. It should be noted that "parent antibody" includes known commercial, recombinantly produced antibodies as outlined below.
[0169] By "Fc fusion protein" or "immunoadhesin" herein is meant a protein comprising an Fc region, generally linked (optionally through a linker moiety, as described herein) to a different protein, such as a binding moiety to a target protein, as described herein. In some cases, one monomer of the heterodimeric protein comprises an antibody heavy chain (either including an scFv or further including a light chain) and the other monomer is a Fc fusion, comprising a variant Fc domain and a ligand. In some embodiments, these "half antibody-half fusion proteins" are referred to as "Fusionbodies".
[0170] By "position" as used herein is meant a location in the sequence of a protein. Positions may be numbered sequentially, or according to an established format, for example the EU index for antibody numbering.
[0171] By "target antigen" as used herein is meant the molecule that is bound specifically by the variable region of a given antibody. A target antigen may be a protein, carbohydrate, lipid, or other chemical compound. A wide number of suitable target antigens are described below.
[0172] By "strandedness" in the context of the monomers of the heterodimeric proteins of the invention herein is meant that, similar to the two strands of DNA that "match", heterodimerization variants are incorporated into each monomer so as to preserve the ability to "match" to form heterodimers. For example, if some pI variants are engineered into monomer A (e.g. making the pI higher) then steric variants that are "charge pairs" that can be utilized as well do not interfere with the pI variants, e.g. the charge variants that make a pI higher are put on the same "strand" or "monomer" to preserve both functionalities.
[0173] By "target cell" as used herein is meant a cell that expresses a target antigen.
[0174] By "variable region" as used herein is meant the region of an immunoglobulin that comprises one or more Ig domains substantially encoded by any of the V.kappa., V.lamda., and/or VH genes that make up the kappa, lambda, and heavy chain immunoglobulin genetic loci respectively.
[0175] By "wild type or WT" herein is meant an amino acid sequence or a nucleotide sequence that is found in nature, including allelic variations. A WT protein has an amino acid sequence or a nucleotide sequence that has not been intentionally modified.
[0176] The antibodies of the present invention are generally isolated or recombinant. "Isolated," when used to describe the various polypeptides disclosed herein, means a polypeptide that has been identified and separated and/or recovered from a cell or cell culture from which it was expressed. Ordinarily, an isolated polypeptide will be prepared by at least one purification step. An "isolated antibody," refers to an antibody which is substantially free of other antibodies having different antigenic specificities.
[0177] "Specific binding" or "specifically binds to" or is "specific for" a particular antigen or an epitope means binding that is measurably different from a non-specific interaction. Specific binding can be measured, for example, by determining binding of a molecule compared to binding of a control molecule, which generally is a molecule of similar structure that does not have binding activity. For example, specific binding can be determined by competition with a control molecule that is similar to the target.
[0178] Specific binding for a particular antigen or an epitope can be exhibited, for example, by an antibody having a KD for an antigen or epitope of at least about 10-4 M, at least about 10-5 M, at least about 10-6 M, at least about 10-7 M, at least about 10-8 M, at least about 10-9 M, alternatively at least about 10-10 M, at least about 10-11 M, at least about 10-12 M, or greater, where KD refers to a dissociation rate of a particular antibody-antigen interaction. Typically, an antibody that specifically binds an antigen will have a KD that is 20-, 50-, 100-, 500-, 1000-, 5,000-, 10,000- or more times greater for a control molecule relative to the antigen or epitope.
[0179] Also, specific binding for a particular antigen or an epitope can be exhibited, for example, by an antibody having a KA or Ka for an antigen or epitope of at least 20-, 50-, 100-, 500-, 1000-, 5,000-, 10,000- or more times greater for the epitope relative to a control, where KA or Ka refers to an association rate of a particular antibody-antigen interaction.
Methods and Compositions for Suppressing T Cells
[0180] In one aspect, the present invention provides methods and compositions for suppressing T cells. In preferred embodiments, the methods and compositions for suppressing T cells are specific for one type of T cell with limited to no impact on other T cells. In further embodiments, the methods and compositions of the present invention suppress Tregs with limited to no impact on other T cell types. In other embodiments, the methods and compositions of the present invention suppress cytotoxic T cells with limited to no impact on other T cell types.
[0181] In one aspect, the methods and compositions of the present invention suppress T cells by administration of heterodimeric proteins. Such heterodimeric proteins include without limitation bispecific (although trispecific, tetraspecific and higher order specificities are also contemplated) antibodies and fusion proteins.
[0182] In certain embodiments, suppression of T cells by methods and compositions of the invention serve to increase the numbers and/or proliferation as compared to T cells that were not treated in accordance with the present invention. In further embodiments, administration of any of the heterodimeric proteins discussed herein serves to increase the numbers and/or proliferation of T cells over that seen without the administration of the heterodimeric protein (or that seen with administration of a control protein) by at least 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 150%, 200%. In yet further embodiments, the increase in the is about 10-20%, 10-50%, 20-90%, 30-80%, 40-70%, 50-60%. In further embodiments, an increase in numbers and/or proliferation is measured in comparison for the targeted T cell type against the non-targeted type. For example, in embodiments in which the administered heterodimeric protein suppresses regulatory T cells, the increase in cell number and/or proliferation of regulatory T cells is measured in comparison to that of other T cell types. In still further embodiments, this comparative increase is at least 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 150%, 200% as compared to the non-targeted T cell type. In yet further embodiments, the comparative increase in the targeted T cell type is about 10-20%, 10-50%, 20-90%, 30-80%, 40-70%, 50-60% over that of the non-targeted T cell type.
[0183] In general, the heterodimeric proteins of use for suppression of T cells in accordance with the present invention comprise two monomers, and each monomer comprises a heavy chain constant region with a variant Fc domain as compared to a parent Fc domain and an antigen binding moiety. In certain embodiments, the variant Fc domain of one of the heavy chain constant region of one of the monomers is different than the heavy chain constant region of the other monomer.
[0184] In certain aspects, the heterodimeric proteins of the invention for suppression of T cells comprise bispecific antibodies or Fc fusion proteins. The bispecific antibodies of the invention can take any format described herein and known in the art, including those pictured in FIG. 3 and FIGS. 36 and 37. The antigen binding domains of these bispecific antibodies will generally comprise an anti-CD25 binding domain on one monomer and a binding domain for a T cell marker on another arm. As will be appreciated, however, any combination of proteins on T cells, including those listed in FIG. 32 can be targets in any combination for bispecific antibodies of the invention. In other words, bispecific antibodies of the invention for suppression of T cells may target any two T cell markers, including any two of those listed in FIG. 32.
[0185] In further exemplary embodiments, bispecific antibodies of the invention comprise an anti-CD25 binding domain on one monomer and an anti-CD4 binding domain on the other monomer (such antibodies are also designated herein as anti-CD25.times.anti-CD4 bispecific antibodies). In further embodiments, the bispecific antibodies of the invention comprise the following combinations of antigen binding domains: anti-CD25.times.anti-CTLA4, anti-CD25.times.anti-PD-1, anti-CD25.times.anti-CCR4, Anti-CD4.times.Anti-CTLA4 and anti-CD4.times.Anti-CCR4 and anti-CD25.times.anti-GITR antibodies.
[0186] Treg cells express CD4 and CD25 simultaneously, and targeting both antigens with a bispecific antibody could in one non-limiting theory be a powerful mechanism to selectively suppress Treg cells and allow the immune system to mount a response against tumor cells. Thus, a bispecific antibody allowing for simultaneous avid targeting of CD4 and CD25 (or any other combination of antigens as discussed above) may in certain embodiments reduce Treg cell proliferation, either via cytotoxic depletion or by interfering with IL2-dependent proliferation. Such an approach will in further embodiments have little or no effect on unactivated CD4+CD25.sup.- T effector cells or CD8+CD25.sup.- cytotoxic T cells. Although it may exhibit some suppression of activated CD4+CD25+ effector T cells, Tregs are reported to have significantly higher dependence on IL-2 for survival (Malek and Bayer Nature 2004, hereby incorporated by reference in its entirety for all purposes and in particular for all teachings related to T cells), providing additional selectivity of this approach for Treg vs CD4 effector T cells.
[0187] Bispecific antibodies can also be used to suppress other T cell types, such as cytotoxic T cells. In some instances, one monomer of the bispecific antibody comprises an anti-CD8 domain, including without limitation domains from anti-CD8 antibodies such as MCD8, 3B5, SK1, OKT-8, 51.1 and DK-25. The monomer with the anti-CD8 domain can be combined with a monomer comprising an anti-CD25 binding domain to produce a bispecific antibody for suppression of cytoxic T cells.
[0188] Generally, the antigen binding domains of bispecific antibodies of the invention are part of monomers that further comprise at least a heavy chain constant region that contains a variant Fc domain as compared to a parent Fc domain.
[0189] Fc fusion proteins may also be used in accordance with the invention to suppress T cells. For example, a fusion protein comprising an IL2 protein on one arm can be engineered to have reduced ability to bind to IL2R.beta., IL2R.gamma., and or IL2R.alpha. in order to ablate IL2 receptor signaling. When coupled with an anti-CD4 antibody (or any other Treg surface marker antibody), this results in an anti-CD4.times.IL2 Fc-fusion capable of suppressing Treg cells through targeted binding to CD4 and CD25, but without the ability to induce Treg proliferation. In one non-limiting theory, the mechanism of action for this fusion may be that it blocks endogenous IL2 from binding to receptor, thus preventing Treg proliferation. Exemplary embodiments of such fusion proteins are provided in FIG. 23.
[0190] Any of the above described heterodimeric antibodies and fusion proteins for suppressing T cells may further include additional amino acid substitutions in the Fc domain. Such substitutions may include one or any combination of substitutions that affect heterodimer formation, serum half-life and/or binding to FcRn (also referred to herein as "Fc variants"), binding to Fc receptors, or ADCC. Exemplary further substitutions of use in any of the heterodimeric proteins discussed herein for suppression of T cells are listed in FIGS. 38-42 and 48.
Methods and Compositions for Inducing T Cells
[0191] In one aspect, the present invention provides methods and compositions for inducing T cells. In preferred embodiments, the methods and compositions for inducing T cells are specific for one type of T cell with limited to no impact on other T cells. In further embodiments, the methods and compositions of the present invention induce Tregs with limited to no impact on other T cell types. In other embodiments, the methods and compositions of the present invention induce cytotoxic T cells with limited to no impact on other T cell types.
[0192] "Inducing T cells" as used herein refers to increasing any aspect of T cell expression or function as compared to expression or function in the absence of the administered heterodimeric protein, including stimulation of the proliferation of the target T cell.
[0193] As discussed herein and understood in the art, induction (as well as suppression) of T cells can be measured with assays to quantify T cell numbers. For example, cell proliferation assays can be used to detect and quantitate T cells. Other methods of quantifying T cells, particularly Tregs, may also be used, including methods utilizing qPCR to measure the amount of demethylated FOXP3 (a Treg marker) that is present. Such assays are described for example in Wieczorek et al., 2009, Cancer Res, 69(2): 599-608, Vries et al., 2011, Clin Cancer Res, 17:841-848, and Baron et al., 2007, Eur. J. Immunol., 37:2378-2389, each of which is hereby incorporated by reference in its entirety for all purposes and in particular for all teachings, figures and legends related to assays for FOXP3, demethylated FOXP3, and quantification of Tregs. Such assays can also be used to quantify the specificity of induction by providing quantitative measurements of numbers of T cells of one type that are induced as compared to other types of T cells (for example, numbers of Tregs induced as compared to cytotoxic T cells).
[0194] In certain embodiments, induction of T cells by methods and compositions of the invention serve to increase the numbers and/or proliferation as compared to T cells that were not treated in accordance with the present invention. In further embodiments, administration of any of the heterodimeric proteins discussed herein serves to increase the numbers and/or proliferation of T cells over that seen without the administration of the heterodimeric protein (or that seen with administration of a control protein) by at least 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 150%, 200%. In yet further embodiments, the increase in the is about 10-20%, 10-50%, 20-90%, 30-80%, 40-70%, 50-60%. In further embodiments, an increase in numbers and/or proliferation is measured in comparison for the targeted T cell type against the non-targeted type. For example, in embodiments in which the administered heterodimeric protein induces regulatory T cells, the increase in cell number and/or proliferation of regulatory T cells is measured in comparison to that of other T cell types. In still further embodiments, this comparative increase is at least 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 150%, 200% as compared to the non-targeted T cell type. In yet further embodiments, the comparative increase in the targeted T cell type is about 10-20%, 10-50%, 20-90%, 30-80%, 40-70%, 50-60% over that of the non-targeted T cell type.
[0195] In further aspects, induction of T cells is accomplished in accordance with the present invention using heterodimeric Fc fusion proteins. Such fusion proteins are also referred to herein as "fusionbodies" because they generally comprise two monomers, in which one monomer is an Fc domain fused to a ligand, such as IL2, and the other monomer is a FAb monomer comprising a heavy chain and a light chain.
[0196] In certain embodiments, Fc fusion proteins of the invention include one monomer that comprises a T cell protein, including without limitation those proteins listed in FIG. 32. In further embodiments, the monomer comprises all or a portion of an IL2 protein. As will be appreciated, the IL2 protein may comprise an IL2 protein from any source, including any mammalian species. In preferred embodiments, the IL2 portion of the monomer comprises a sequence from human IL2. Variants of IL2 may also be used in Fc fusion proteins of the invention, including without limitation variants such as those listed in FIG. 23.
[0197] In further embodiments, the Fc fusion proteins comprise a second monomer that comprises a T cell protein, including without limitation any of the proteins listed in FIG. 32. In exemplary embodiments, the fusionbodies of the present invention for induction of T cells have one monomer that is an Fc domain fused to all or part of an IL2 protein and the second monomer comprises an antigen binding domain that targets one of the following: CD4, CD8, CTLA-4, CCR4, and PD-1. In still further embodiments, the second monomer comprises both a heavy chain and a light chain sequence, and the variable domains of those heavy and light chain sequences form the antigen-binding domain.
Methods of Making Compositions of the Invention
[0198] Any of the heterodimeric proteins discussed herein, including bispecific antibodies and heterodimeric Fc fusion proteins, can be made using methods known in the art and methods described in further detail herein.
[0199] In certain aspects, the invention provides one or more nucleic acids encoding a composition according to any of the compositions described herein. As will be appreciated, different monomers of the heterodimeric proteins of the invention may be expressed using nucleic acids encoding all or a portion of one or more of the monomers of the protein. Thus, for example, for a bispecific antibody in which one monomer targets CD4 and the other monomer target CD25, the present invention further provides a nucleic acid encoding the first and second monomers as separate molecules that are then assembled together by co-expression in the same host cell. In other embodiments, the two monomers may be encoded in the same nucleic acid, in some embodiments within the same vector. In embodiments in which one or both of the monomers comprise both heavy and light chain sequences, those sequences may also be encoded by one or by multiple nucleic acids.
[0200] In further embodiments, the invention further provides host cells expressing the one or more nucleic acids encoding the one or more monomers of heterodimeric proteins of the invention. As will be appreciated and as is discussed above, the heterodimeric proteins of the present invention may be encoded by one or more nucleic acids. These one or more nucleic acids may be expressed in a single host cell or in separate host cells. For example, for heterodimeric proteins that are in the bottle-opener format in which one of the monomers is an scFv and the other monomer is a FAb, there may be three nucleic acids encoding this protein: one for the scFv, one for the heavy chain sequence of the FAb, and one for the light chain sequence of the FAb. These three nucleic acids will in general be expressed in the same host cell in order to produce the heterodimeric protein, although expression in separate host cells is also contemplated.
[0201] In yet further embodiments, and in accordance with any of the above, the present invention provides a method of making any of the compositions described herein, the method including the step of culturing a host cell or more nucleic acids encoding a heterodimeric protein of the invention, including any of the bispecific antibodies or Fc fusion proteins described herein.
[0202] In further aspects, the present invention provides a method of purifying a heterodimeric protein or bispecific antibody in accordance with any of the above, the method including: (a) providing a composition in accordance with any of the heterodimeric proteins described herein, (b) loading the composition onto an ion exchange column; and (c) collecting a fraction containing the heterodimeric protein or bispecific antibody, thus purifying the protein or antibody.
Heterodimeric Proteins Overview
[0203] The present invention is directed to methods of modulating T cells using novel constructs to provide heterodimeric proteins that allow binding to more than one antigen or ligand, e.g. to allow for multispecific binding. The heterodimeric protein constructs are based on the self-assembling nature of the two Fc domains of the heavy chains of antibodies, e.g. two "monomers" that assemble into a "dimer". Heterodimeric proteins are made by altering the amino acid sequence of each monomer as more fully discussed below. Thus, the present invention is generally directed to the creation of heterodimeric proteins including antibodies, which can co-engage antigens in several ways, relying on amino acid variants in the constant regions that are different on each chain to promote heterodimeric formation and/or allow for ease of purification of heterodimers over the homodimers. As discussed more fully below, the heterodimeric proteins can be antibody variants or based on Fc fusion proteins. Although much of the following discussion is in terms of heterodimeric antibodies, it will be appreciated by those in the art and more fully described below, the discussion applies equally to heterodimeric proteins that are based on Fc fusion proteins (also referred to herein as fusionbodies).
[0204] Thus, the present invention provides bispecific antibodies (or, as discussed below, trispecific or tetraspecific antibodies can also be made). An ongoing problem in antibody technologies is the desire for "bispecific" (and/or multispecific) antibodies that bind to two (or more) different antigens simultaneously, in general thus allowing the different antigens to be brought into proximity and resulting in new functionalities and new therapies. In general, these antibodies are made by including genes for each heavy and light chain into the host cells. This generally results in the formation of the desired heterodimer (A-B), as well as the two homodimers (A-A and B-B). However, a major obstacle in the formation of multispecific antibodies is the difficulty in purifying the heterodimeric antibodies away from the homodimeric antibodies and/or biasing the formation of the heterodimer over the formation of the homodimers.
[0205] There are a number of mechanisms that can be used to generate the heterodimers of the present invention. In addition, as will be appreciated by those in the art, these mechanisms can be combined to ensure high heterodimerization. Thus, amino acid variants that lead to the production of heterodimers are referred to as "heterodimerization variants". As discussed below, heterodimerization variants can include steric variants (e.g. the "knobs and holes" or "skew" variants described below and the "charge pairs" variants described below) as well as "pI variants", which allows purification of homodimers away from heterodimers.
[0206] One mechanism is generally referred to in the art as "knobs and holes" ("KIH") or sometimes herein as "skew" variants, referring to amino acid engineering that creates steric influences to favor heterodimeric formation and disfavor homodimeric formation can also optionally be used; this is sometimes referred to as "knobs and holes"; as described in U.S. Ser. No. 61/596,846 and U.S. Ser. No. 12/875,0015, Ridgway et al., Protein Engineering 9(7):617 (1996); Atwell et al., J. Mol. Biol. 1997 270:26; U.S. Pat. No. 8,216,805, US 2012/0149876, all of which are hereby incorporated by reference in their entirety. The Figures identify a number of "monomer A-monomer B" pairs that include "knobs and holes" amino acid substitutions. In addition, as described in Merchant et al., Nature Biotech. 16:677 (1998), these "knobs and hole" mutations can be combined with disulfide bonds to skew formation to heterodimerization.
[0207] An additional mechanism that finds use in the generation of heterodimers is sometimes referred to as "electrostatic steering" or "charge pairs" as described in Gunasekaran et al., J. Biol. Chem. 285(25):19637 (2010), hereby incorporated by reference in its entirety. This is sometimes referred to herein as "charge pairs". In this embodiment, electrostatics are used to skew the formation towards heterodimerization. As those in the art will appreciate, these may also have an effect on pI, and thus on purification, and thus could in some cases also be considered pI variants. However, as these were generated to force heterodimerization and were not used as purification tools, they are classified as "steric variants". These include, but are not limited to, D221E/P228E/L368E paired with D221R/P228R/K409R (e.g. these are "monomer corresponding sets) and C220E/P228E/368E paired with C220R/E224R/P228R/K409R and others shown in the Figures.
[0208] In the present invention, in some embodiments, pI variants are used to alter the pI of one or both of the monomers and thus allowing the isoelectric purification of A-A, A-B and B-B dimeric proteins.
[0209] In the present invention, there are several basic mechanisms that can lead to ease of purifying heterodimeric proteins; one relies on the use of pI variants, such that each monomer has a different pI, thus allowing the isoelectric purification of A-A, A-B and B-B dimeric proteins. Alternatively, some scaffold formats, such as the "triple F" format, also allows separation on the basis of size. As is further outlined below, it is also possible to "skew" the formation of heterodimers over homodimers. Thus, a combination of steric heterodimerization variants and pI or charge pair variants find particular use in the invention. Additionally, as more fully outlined below, scaffolds that utilize scFv(s) such as the Triple F format can include charged scFv linkers (either positive or negative), that give a further pI boost for purification purposes. As will be appreciated by those in the art, some Triple F formats are useful with just charged scFv linkers and no additional pI adjustments, although the invention does provide the use of skew variants with charged scFv linkers as well (and combinations of Fc, FcRn and KO variants).
[0210] In the present invention that utilizes pI as a separation mechanism to allow the purification of heterodimeric proteins, amino acid variants can be introduced into one or both of the monomer polypeptides; that is, the pI of one of the monomers (referred to herein for simplicity as "monomer A") can be engineered away from monomer B, or both monomer A and B change be changed, with the pI of monomer A increasing and the pI of monomer B decreasing. As is outlined more fully below, the pI changes of either or both monomers can be done by removing or adding a charged residue (e.g. a neutral amino acid is replaced by a positively or negatively charged amino acid residue, e.g. glycine to glutamic acid), changing a charged residue from positive or negative to the opposite charge (aspartic acid to lysine) or changing a charged residue to a neutral residue (e.g. loss of a charge; lysine to serine.). A number of these variants are shown in the Figures.
[0211] Accordingly, in this embodiment of the present invention provides for creating a sufficient change in pI in at least one of the monomers such that heterodimers can be separated from homodimers. As will be appreciated by those in the art, and as discussed further below, this can be done by using a "wild type" heavy chain constant region and a variant region that has been engineered to either increase or decrease its pI (wt A-+B or wt A--B), or by increasing one region and decreasing the other region (A+-B- or A-B+).
[0212] Thus, in general, a component of some embodiments of the present invention are amino acid variants in the constant regions of antibodies that are directed to altering the isoelectric point (pI) of at least one, if not both, of the monomers of a dimeric protein to form "pI heterodimers" (when the protein is an antibody, these are referred to as "pI antibodies") by incorporating amino acid substitutions ("pI variants" or "pI substitutions") into one or both of the monomers. As shown herein, the separation of the heterodimers from the two homodimers can be accomplished if the pIs of the two monomers differ by as little as 0.1 pH unit, with 0.2, 0.3, 0.4 and 0.5 or greater all finding use in the present invention.
[0213] As will be appreciated by those in the art, the number of pI variants to be included on each or both monomer(s) to get good separation will depend in part on the starting pI of the scFv and Fab of interest. That is, to determine which monomer to engineer or in which "direction" (e.g. more positive or more negative), the Fv sequences of the two target antigens are calculated and a decision is made from there. As is known in the art, different Fvs will have different starting pIs which are exploited in the present invention. In general, as outlined herein, the pIs are engineered to result in a total pI difference of each monomer of at least about 0.1 logs, with 0.2 to 0.5 being preferred as outlined herein.
[0214] Furthermore, as will be appreciated by those in the art and outlined herein, heterodimers can be separated from homodimers on the basis of size. For example, as shown in FIGS. 36 and 37, heterodimers with two scFvs can be separated by those of the "triple F" format and a bispecific mAb. This can be further exploited in higher valency with additional antigen binding sites being utilized. For example, as additionally shown, one monomer will have two Fab fragments and the other will have one scFv, resulting in a differential in size and thus molecular weight.
[0215] In addition, as will be appreciated by those in the art and outlined herein, the format outlined herein can be expanded to provide trispecific and tetraspecific antibodies as well. In this embodiment, some variations of which are depicted in the FIG. 36A36M, it will be recognized that it is possible that some antigens are bound divalently (e.g. two antigen binding sites to a single antigen; for example, A and B could be part of a typical bivalent association and C and D can be optionally present and optionally the same or different). As will be appreciated, any combination of Fab and scFvs can be utilized to achieve the desired result and combinations.
[0216] In the case where pI variants are used to achieve heterodimerization, by using the constant region(s) of the heavy chain(s), a more modular approach to designing and purifying multispecific proteins, including antibodies, is provided. Thus, in some embodiments, heterodimerization variants (including skew and purification heterodimerization variants) are not included in the variable regions, such that each individual antibody must be engineered. In addition, in some embodiments, the possibility of immunogenicity resulting from the pI variants is significantly reduced by importing pI variants from different IgG isotypes such that pI is changed without introducing significant immunogenicity. Thus, an additional problem to be solved is the elucidation of low pI constant domains with high human sequence content, e.g. the minimization or avoidance of non-human residues at any particular position.
[0217] A side benefit that can occur with this pI engineering is also the extension of serum half-life and increased FcRn binding. That is, as described in U.S. Ser. No. 13/194,904 (incorporated by reference in its entirety), lowering the pI of antibody constant domains (including those found in antibodies and Fc fusions) can lead to longer serum retention in vivo. These pI variants for increased serum half life also facilitate pI changes for purification.
[0218] In addition, it should be noted that the pI variants of the heterodimerization variants give an additional benefit for the analytics and quality control process of bispecific antibodies, the ability to eliminate, minimize and/or distinguish when homodimers are present is significant. Similarly, the ability to reliably test the reproducibility of the heterodimeric protein production is important.
[0219] In addition to all or part of a variant heavy constant domain, one or both of the monomers may contain one or two fusion partners, such that the heterodimers form multivalent proteins. As is generally depicted in the Figures, and specifically FIG. 36A, the fusion partners are depicted as A, B, C and D, with all combinations possible. In general, A, B, C and D are selected such that the heterodimer is at least bispecific or bivalent in its ability to interact with additional proteins.
[0220] As will be appreciated by those in the art and discussed more fully below, the heterodimeric fusion proteins of the present invention can take on a wide variety of configurations, as are generally depicted in FIGS. 36 and 37. Some figures depict "single ended" configurations, where there is one type of specificity on one "arm" of the molecule and a different specificity on the other "arm". Other figures depict "dual ended" configurations, where there is at least one type of specificity at the "top" of the molecule and one or more different specificities at the "bottom" of the molecule. Furthermore as is shown, these two configurations can be combined, where there can be triple or quadruple specificities based on the particular combination. Thus, the present invention provides "multispecific" binding proteins, including multispecific antibodies. Thus, the present invention is directed to novel immunoglobulin compositions that co-engage at least a first and a second antigen. First and second antigens of the invention are herein referred to as antigen-1 and antigen-2 respectively.
[0221] One heterodimeric scaffold that finds particular use in the present invention is the "triple F" or "bottle opener" scaffold format. In this embodiment, one heavy chain of the antibody contains an single chain Fv ("scFv", as defined below) and the other heavy chain is a "regular" FAb format, comprising a variable heavy chain and a light chain. This structure is sometimes referred to herein as "triple F" format (scFv-FAb-Fc) or the "bottle-opener" format, due to a rough visual similarity to a bottle-opener (see FIG. 36B). The two chains are brought together by the use of amino acid variants in the constant regions (e.g. the Fc domain and/or the hinge region) that promote the formation of heterodimeric antibodies as is described more fully below.
[0222] There are several distinct advantages to the present "triple F" format. As is known in the art, antibody analogs relying on two scFv constructs often have stability and aggregation problems, which can be alleviated in the present invention by the addition of a "regular" heavy and light chain pairing. In addition, as opposed to formats that rely on two heavy chains and two light chains, there is no issue with the incorrect pairing of heavy and light chains (e.g. heavy 1 pairing with light 2, etc.)
[0223] In addition to all or part of a variant heavy constant domain, one or both of the monomers may contain one or two fusion partners, such that the heterodimers form multivalent proteins. As is generally depicted in the FIG. 64 of U.S. Ser. No. 13/648,951, hereby incorporated by reference with its accompanying legend, the fusion partners are depicted as A, B, C and D, with all combinations possible. In general, A, B, C and D are selected such that the heterodimer is at least bispecific or bivalent in its ability to interact with additional proteins. In the context of the present "triple F" format, generally A and B are an scFv and a Fv (as will be appreciated, either monomer can contain the scFv and the other the Fv/Fab) and then optionally one or two additional fusion partners.
[0224] Furthermore, as outlined herein, additional amino acid variants may be introduced into the bispecific antibodies of the invention, to add additional functionalities. For example, amino acid changes within the Fc region can be added (either to one monomer or both) to facilitate increased ADCC or CDC (e.g. altered binding to Fc.gamma. receptors); to allow or increase yield of the addition of toxins and drugs (e.g. for ADC), as well as to increase binding to FcRn and/or increase serum half-life of the resulting molecules. As is further described herein and as will be appreciated by those in the art, any and all of the variants outlined herein can be optionally and independently combined with other variants.
[0225] Similarly, another category of functional variants are "Fc.gamma. ablation variants" or "Fc knock out (FcKO or KO) variants. In these embodiments, for some therapeutic applications, it is desirable to reduce or remove the normal binding of the Fc domain to one or more or all of the Fc.gamma. receptors (e.g. Fc.gamma.RI, Fc.gamma.RIIa, Fc.gamma.RIIb, Fc.gamma.RIIIa, etc.) to avoid additional mechanisms of action. That is, for example, in many embodiments, particularly in the use of bispecific antibodies of the invention, it is generally desirable to ablate Fc.gamma.RIIIa binding to eliminate or significantly reduce ADCC activity.
Antibodies
[0226] The present invention relates to the generation of multispecific antibodies, generally therapeutic antibodies. As is discussed below, the term "antibody" is used generally. Antibodies that find use in the present invention can take on a number of formats as described herein, including traditional antibodies as well as antibody derivatives, fragments and mimetics, described below. In general, the term "antibody" includes any polypeptide that includes at least one constant domain, including, but not limited to, CH1, CH2, CH3 and CL.
[0227] Traditional antibody structural units typically comprise a tetramer. Each tetramer is typically composed of two identical pairs of polypeptide chains, each pair having one "light" (typically having a molecular weight of about 25 kDa) and one "heavy" chain (typically having a molecular weight of about 50-70 kDa). Human light chains are classified as kappa and lambda light chains. The present invention is directed to the IgG class, which has several subclasses, including, but not limited to IgG1, IgG2, IgG3, and IgG4. Thus, "isotype" as used herein is meant any of the subclasses of immunoglobulins defined by the chemical and antigenic characteristics of their constant regions. It should be understood that therapeutic antibodies can also comprise hybrids of isotypes and/or subclasses. For example, as shown in US Publication 2009/0163699, incorporated by reference, the present invention covers pI engineering of IgG1/G2 hybrids.
[0228] The amino-terminal portion of each chain includes a variable region of about 100 to 110 or more amino acids primarily responsible for antigen recognition, generally referred to in the art and herein as the "Fv domain" or "Fv region". In the variable region, three loops are gathered for each of the V domains of the heavy chain and light chain to form an antigen-binding site. Each of the loops is referred to as a complementarity-determining region (hereinafter referred to as a "CDR"), in which the variation in the amino acid sequence is most significant. "Variable" refers to the fact that certain segments of the variable region differ extensively in sequence among antibodies. Variability within the variable region is not evenly distributed. Instead, the V regions consist of relatively invariant stretches called framework regions (FRs) of 15-30 amino acids separated by shorter regions of extreme variability called "hypervariable regions" that are each 9-15 amino acids long or longer.
[0229] Each VH and VL is composed of three hypervariable regions ("complementary determining regions," "CDRs") and four FRs, arranged from amino-terminus to carboxy-terminus in the following order: FR1-CDR1-FR2-CDR2-FR3-CDR3-FR4.
[0230] The hypervariable region generally encompasses amino acid residues from about amino acid residues 24-34 (LCDR1; "L" denotes light chain), 50-56 (LCDR2) and 89-97 (LCDR3) in the light chain variable region and around about 31-35B (HCDR1; "H" denotes heavy chain), 50-65 (HCDR2), and 95-102 (HCDR3) in the heavy chain variable region; Kabat et al., SEQUENCES OF PROTEINS OF IMMUNOLOGICAL INTEREST, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991) and/or those residues forming a hypervariable loop (e.g. residues 26-32 (LCDR1), 50-52 (LCDR2) and 91-96 (LCDR3) in the light chain variable region and 26-32 (HCDR1), 53-55 (HCDR2) and 96-101 (HCDR3) in the heavy chain variable region; Chothia and Lesk (1987) J. Mol. Biol. 196:901-917. Specific CDRs of the invention are described below.
[0231] Throughout the present specification, the Kabat numbering system is generally used when referring to a residue in the variable domain (approximately, residues 1-107 of the light chain variable region and residues 1-113 of the heavy chain variable region) and the EU numbering system for Fc regions (e.g, Kabat et al., supra (1991)).
[0232] The CDRs contribute to the formation of the antigen-binding, or more specifically, epitope binding site of antibodies. "Epitope" refers to a determinant that interacts with a specific antigen binding site in the variable region of an antibody molecule known as a paratope. Epitopes are groupings of molecules such as amino acids or sugar side chains and usually have specific structural characteristics, as well as specific charge characteristics. A single antigen may have more than one epitope.
[0233] The epitope may comprise amino acid residues directly involved in the binding (also called immunodominant component of the epitope) and other amino acid residues, which are not directly involved in the binding, such as amino acid residues which are effectively blocked by the specifically antigen binding peptide; in other words, the amino acid residue is within the footprint of the specifically antigen binding peptide.
[0234] Epitopes may be either conformational or linear. A conformational epitope is produced by spatially juxtaposed amino acids from different segments of the linear polypeptide chain. A linear epitope is one produced by adjacent amino acid residues in a polypeptide chain. Conformational and nonconformational epitopes may be distinguished in that the binding to the former but not the latter is lost in the presence of denaturing solvents.
[0235] An epitope typically includes at least 3, and more usually, at least 5 or 8-10 amino acids in a unique spatial conformation. Antibodies that recognize the same epitope can be verified in a simple immunoassay showing the ability of one antibody to block the binding of another antibody to a target antigen, for example "binning."
[0236] The carboxy-terminal portion of each chain defines a constant region primarily responsible for effector function. Kabat et al. collected numerous primary sequences of the variable regions of heavy chains and light chains. Based on the degree of conservation of the sequences, they classified individual primary sequences into the CDR and the framework and made a list thereof (see SEQUENCES OF IMMUNOLOGICAL INTEREST, 5th edition, NIH publication, No. 91-3242, E. A. Kabat et al., entirely incorporated by reference).
[0237] In the IgG subclass of immunoglobulins, there are several immunoglobulin domains in the heavy chain. By "immunoglobulin (Ig) domain" herein is meant a region of an immunoglobulin having a distinct tertiary structure. Of interest in the present invention are the heavy chain domains, including, the constant heavy (CH) domains and the hinge domains. In the context of IgG antibodies, the IgG isotypes each have three CH regions. Accordingly, "CH" domains in the context of IgG are as follows: "CH1" refers to positions 118-220 according to the EU index as in Kabat. "CH2" refers to positions 237-340 according to the EU index as in Kabat, and "CH3" refers to positions 341-447 according to the EU index as in Kabat. As shown herein and described below, the pI variants can be in one or more of the CH regions, as well as the hinge region, discussed below.
[0238] It should be noted that for the IgG sequences depicted herein start at the CH1 region, position 118; the variable regions are not included except as noted. For example, the first amino acid, while designated as position "1" in the sequence listing, corresponds to position 118 of the CH1 region, according to EU numbering.
[0239] Another type of Ig domain of the heavy chain is the hinge region. By "hinge" or "hinge region" or "antibody hinge region" or "immunoglobulin hinge region" herein is meant the flexible polypeptide comprising the amino acids between the first and second constant domains of an antibody. Structurally, the IgG CH1 domain ends at EU position 220, and the IgG CH2 domain begins at residue EU position 237. Thus for IgG the antibody hinge is herein defined to include positions 221 (D221 in IgG1) to 236 (G236 in IgG1), wherein the numbering is according to the EU index as in Kabat. In some embodiments, for example in the context of an Fc region, the lower hinge is included, with the "lower hinge" generally referring to positions 226 or 230. As noted herein, pI variants can be made in the hinge region as well.
[0240] The light chain generally comprises two domains, the variable light domain (containing the light chain CDRs and together with the variable heavy domains forming the Fv region), and a constant light chain region (often referred to as CL or CK).
[0241] Another region of interest for additional substitutions, outlined below, is the Fc region. By "Fc" or "Fc region" or "Fc domain" as used herein is meant the polypeptide comprising the constant region of an antibody excluding the first constant region immunoglobulin domain and in some cases, part of the hinge. Thus Fc refers to the last two constant region immunoglobulin domains of IgA, IgD, and IgG, the last three constant region immunoglobulin domains of IgE and IgM, and the flexible hinge N-terminal to these domains. For IgA and IgM, Fc may include the J chain. For IgG, the Fc domain comprises immunoglobulin domains C.gamma.2 and C.gamma.3 (C.gamma.2 and C.gamma.3) and the lower hinge region between C.gamma.1 (C.gamma.1) and C.gamma.2 (C.gamma.2). Although the boundaries of the Fc region may vary, the human IgG heavy chain Fc region is usually defined to include residues C226 or P230 to its carboxyl-terminus, wherein the numbering is according to the EU index as in Kabat. In some embodiments, as is more fully described below, amino acid modifications are made to the Fc region, for example to alter binding to one or more Fc.gamma.R receptors or to the FcRn receptor.
[0242] Accordingly, in some embodiments the present invention provides heterodimeric antibodies that rely on the use of two different heavy chain variant Fc domains that will self-assemble to form heterodimeric antibodies.
[0243] In some embodiments, the antibodies are full length. By "full length antibody" herein is meant the structure that constitutes the natural biological form of an antibody, including variable and constant regions, including one or more modifications as outlined herein, particularly in the Fc domains to allow either heterodimerization formation or the purification of heterodimers away from homodimers. A full length heterodimeric antibody is two heavy chains with different Fc domains and either two light chains or a common light chain.
[0244] Alternatively, the antibodies can include a variety of structures as are generally shown in the Figures, including, but not limited to, antibody fragments, monoclonal antibodies, bispecific antibodies, minibodies, domain antibodies, synthetic antibodies (sometimes referred to herein as "antibody mimetics"), chimeric antibodies, humanized antibodies, antibody fusions (sometimes referred to as "antibody conjugates"), and fragments of each, respectively.
[0245] In one embodiment, the antibody is an antibody fragment, as long as it contains at least one constant domain which can be engineered to produce heterodimers, such as pI engineering. Other antibody fragments that can be used include fragments that contain one or more of the CH1, CH2, CH3, hinge and CL domains of the invention that have been pI engineered. For example, Fc fusions are fusions of the Fc region (CH2 and CH3, optionally with the hinge region) fused to another protein. A number of Fc fusions are known the art and can be improved by the addition of the heterodimerization variants of the invention. In the present case, antibody fusions can be made comprising CH1; CH1, CH2 and CH3; CH2; CH3; CH2 and CH3; CH1 and CH3, any or all of which can be made optionally with the hinge region, utilizing any combination of heterodimerization variants described herein.
scFv Embodiments
[0246] In some embodiments of the present invention, one monomer comprises a heavy chain comprises a scFV linked to an Fc domain, and the other monomer comprises a heavy chain comprising a Fab linked to an Fc domain, e.g. a "typical" heavy chain, and a light chain. By "Fab" or "Fab region" as used herein is meant the polypeptide that comprises the VH, CH1, VL, and CL immunoglobulin domains. Fab may refer to this region in isolation, or this region in the context of a full length antibody, antibody fragment or Fab fusion protein. By "Fv" or "Fv fragment" or "Fv region" as used herein is meant a polypeptide that comprises the VL and VH domains of a single antibody.
[0247] Several of the heterodimeric antibody embodiments described herein rely on the use of one or more scFv domains, comprising the variable heavy and variable light chains, covalently linked using a linker, forming an antigen binding domain. Some embodiments herein use "standard" linkers, usually linkers of glycine and serine, as is well known in the art.
[0248] The present invention further provides charged scFv linkers, to facilitate the separation in pI between a first and a second monomer. That is, by incorporating a charged scFv linker, either positive or negative (or both, in the case of scaffolds that use scFvs on different monomers), this allows the monomer comprising the charged linker to alter the pI without making further changes in the Fc domains. These charged linkers can be substituted into any scFv containing standard linkers. Again, as will be appreciated by those in the art, charged scFv linkers are used on the correct "strand" or monomer, according to the desired changes in pI. For example, as discussed herein, to make triple F format heterodimeric antibody, the original pI of the Fv region for each of the desired antigen binding domains are calculated, and one is chosen to make an scFv, and depending on the pI, either positive or negative linkers are chosen.
[0249] In addition, disulfide bonds can be engineered into the variable heavy and variable light chains to give additional stability.
Chimeric and Humanized Antibodies
[0250] In some embodiments, the antibody can be a mixture from different species, e.g. a chimeric antibody and/or a humanized antibody. In general, both "chimeric antibodies" and "humanized antibodies" refer to antibodies that combine regions from more than one species. For example, "chimeric antibodies" traditionally comprise variable region(s) from a mouse (or rat, in some cases) and the constant region(s) from a human. "Humanized antibodies" generally refer to non-human antibodies that have had the variable-domain framework regions swapped for sequences found in human antibodies. Generally, in a humanized antibody, the entire antibody, except the CDRs, is encoded by a polynucleotide of human origin or is identical to such an antibody except within its CDRs. The CDRs, some or all of which are encoded by nucleic acids originating in a non-human organism, are grafted into the beta-sheet framework of a human antibody variable region to create an antibody, the specificity of which is determined by the engrafted CDRs. The creation of such antibodies is described in, e.g., WO 92/11018, Jones, 1986, Nature 321:522-525, Verhoeyen et al., 1988, Science 239:1534-1536, all entirely incorporated by reference. "Backmutation" of selected acceptor framework residues to the corresponding donor residues is often required to regain affinity that is lost in the initial grafted construct (U.S. Pat. Nos. 5,530,101; 5,585,089; 5,693,761; 5,693,762; 6,180,370; 5,859,205; 5,821,337; 6,054,297; 6,407,213, all entirely incorporated by reference). The humanized antibody optimally also will comprise at least a portion of an immunoglobulin constant region, typically that of a human immunoglobulin, and thus will typically comprise a human Fc region. Humanized antibodies can also be generated using mice with a genetically engineered immune system. Roque et al., 2004, Biotechnol. Prog. 20:639-654, entirely incorporated by reference. A variety of techniques and methods for humanizing and reshaping non-human antibodies are well known in the art (See Tsurushita & Vasquez, 2004, Humanization of Monoclonal Antibodies, Molecular Biology of B Cells, 533-545, Elsevier Science (USA), and references cited therein, all entirely incorporated by reference). Humanization methods include but are not limited to methods described in Jones et al., 1986, Nature 321:522-525; Riechmann et al., 1988; Nature 332:323-329; Verhoeyen et al., 1988, Science, 239:1534-1536; Queen et al., 1989, Proc Natl Acad Sci, USA 86:10029-33; He et al., 1998, J. Immunol. 160: 1029-1035; Carter et al., 1992, Proc Natl Acad Sci USA 89:4285-9, Presta et al., 1997, Cancer Res. 57(20):4593-9; Gorman et al., 1991, Proc. Natl. Acad. Sci. USA 88:4181-4185; O'Connor et al., 1998, Protein Eng 11:321-8, all entirely incorporated by reference. Humanization or other methods of reducing the immunogenicity of nonhuman antibody variable regions may include resurfacing methods, as described for example in Roguska et al., 1994, Proc. Natl. Acad. Sci. USA 91:969-973, entirely incorporated by reference. In one embodiment, the parent antibody has been affinity matured, as is known in the art. Structure-based methods may be employed for humanization and affinity maturation, for example as described in U.S. Ser. No. 11/004,590. Selection based methods may be employed to humanize and/or affinity mature antibody variable regions, including but not limited to methods described in Wu et al., 1999, J. Mol. Biol. 294:151-162; Baca et al., 1997, J. Biol. Chem. 272(16):10678-10684; Rosok et al., 1996, J. Biol. Chem. 271(37): 22611-22618; Rader et al., 1998, Proc. Natl. Acad. Sci. USA 95: 8910-8915; Krauss et al., 2003, Protein Engineering 16(10):753-759, all entirely incorporated by reference. Other humanization methods may involve the grafting of only parts of the CDRs, including but not limited to methods described in U.S. Ser. No. 09/810,510; Tan et al., 2002, J. Immunol. 169:1119-1125; De Pascalis et al., 2002, J. Immunol. 169:3076-3084, all entirely incorporated by reference.
Heterodimeric Heavy Chain Constant Regions
[0251] Accordingly, the present invention provides heterodimeric proteins based on the use of monomers containing variant heavy chain constant regions, and specifically the Fc domains, as a first domain. By "monomer" herein is meant one half of the heterodimeric protein. It should be noted that traditional antibodies are actually tetrameric (two heavy chains and two light chains). In the context of the present invention, one pair of heavy-light chains (if applicable, e.g. if the monomer comprises an Fab) is considered a "monomer". Similarly, a heavy chain region comprising the scFv is considered a monomer. In the case where an Fv region is one fusion partner (e.g. heavy and light variable domains) and a non-antibody protein is another fusion partner, each "half" is considered a monomer. Essentially, each monomer comprises sufficient heavy chain constant region to allow heterodimerization engineering, whether that be all the constant region, e.g. Ch1-hinge-CH2-CH3, the Fc region (CH2-CH3), or just the CH3 domain.
[0252] The variant heavy chain constant regions can comprise all or part of the heavy chain constant region, including the full length construct, CH1-hinge-CH2-CH3, or portions thereof, including for example CH2-CH3 or CH3 alone. In addition, the heavy chain region of each monomer can be the same backbone (CH1-hinge-CH2-CH3 or CH2-CH3) or different. N- and C-terminal truncations and additions are also included within the definition; for example, some pI variants include the addition of charged amino acids to the C-terminus of the heavy chain domain.
[0253] Thus, in general, one monomer of the present "triple F" construct is a scFv region-hinge-Fc domain) and the other is (VH-CH1-hinge-CH2-CH3 plus associated light chain), with heterodimerization variants, including steric, isotypic, charge steering, and pI variants, Fc and FcRn variants, ablation variants, and additional antigen binding domains (with optional linkers) included in these regions.
[0254] In addition to the heterodimerization variants (e.g. steric and pI variants) outlined herein, the heavy chain regions may also contain additional amino acid substitutions, including changes for altering Fc.gamma.R and FcRn binding as discussed below.
[0255] In addition, some monomers can utilize linkers between the variant heavy chain constant region and the fusion partner. For the scFv portion of the "bottle-opener", standard linkers as are known in the art can be used, or the charged scFv linkers described herein. In the case where additional fusion partners are made (e.g. FIGS. 1 and 2), traditional peptide linkers can be used, including flexible linkers of glycine and serine, or the charged linkers of FIG. 9. In some cases, the linkers for use as components of the monomer are different from those defined below for the ADC constructs, and are in many embodiments not cleavable linkers (such as those susceptible to proteases), although cleavable linkers may find use in some embodiments.
[0256] The heterodimerization variants include a number of different types of variants, including, but not limited to, steric variants (including charge variants) and pI variants, that can be optionally and independently combined with any other variants. In these embodiments, it is important to match "monomer A" with "monomer B"; that is, if a heterodimeric protein relies on both steric variants and pI variants, these need to be correctly matched to each monomer: e.g. the set of steric variants that work (1 set on monomer A, 1 set on monomer B) is combined with pI variant sets (1 set on monomer A, 1 set on monomer B), such that the variants on each monomer are designed to achieve the desired function, keeping in mind the pI "strandedness" such that steric variants that may alter pI are put on the appropriate monomer.
[0257] It is important to note that the heterodimerization variants outlined herein (for example, including but not limited to those variants shown in FIGS. 3 and 12), can be optionally and independently combined with any other variants, and on any other monomer. That is, what is important for the heterodimerization is that there are "sets" of variants, one set for one monomer and one set for the other. Whether these are combined from the FIGS. 1 to 1 (e.g. monomer 1 listings can go together) or switched (monomer 1 pI variants with monomer 2 steric variants) is irrelevant. However, as noted herein, "strandedness" should be preserved when combinations are made as outlined above. Furthermore, for the additional Fc variants (such as for Fc.gamma.R binding, FcRn binding, etc.), either monomer, or both monomers, can include any of the listed variants, independently and optionally. In some cases, both monomers have the additional variants and in some only one monomer has the additional variants, or they can be combined.
Heterodimerization Variants
[0258] The present invention provides heterodimeric proteins, including heterodimeric antibodies in a variety of formats, which utilize heterodimeric variants to allow for heterodimeric formation and/or purification away from homodimers.
Steric Variants
[0259] In some embodiments, the formation of heterodimers can be facilitated by the addition of steric variants. That is, by changing amino acids in each heavy chain, different heavy chains are more likely to associate to form the heterodimeric structure than to form homodimers with the same Fc amino acid sequences. Suitable steric variants are included in FIG. 41A-41B.
[0260] One mechanism is generally referred to in the art as "knobs and holes", referring to amino acid engineering that creates steric influences to favor heterodimeric formation and disfavor homodimeric formation can also optionally be used; this is sometimes referred to as "knobs and holes", as described in U.S. Ser. No. 61/596,846, Ridgway et al., Protein Engineering 9(7):617 (1996); Atwell et al., J. Mol. Biol. 1997 270:26; U.S. Pat. No. 8,216,805, all of which are hereby incorporated by reference in their entirety. The Figures identify a number of "monomer A-monomer B" pairs that rely on "knobs and holes". In addition, as described in Merchant et al., Nature Biotech. 16:677 (1998), these "knobs and hole" mutations can be combined with disulfide bonds to skew formation to heterodimerization.
[0261] An additional mechanism that finds use in the generation of heterodimers is sometimes referred to as "electrostatic steering" as described in Gunasekaran et al., J. Biol. Chem. 285(25):19637 (2010), hereby incorporated by reference in its entirety. This is sometimes referred to herein as "charge pairs". In this embodiment, electrostatics are used to skew the formation towards heterodimerization. As those in the art will appreciate, these may also have an effect on pI, and thus on purification, and thus could in some cases also be considered pI variants. However, as these were generated to force heterodimerization and were not used as purification tools, they are classified as "steric variants". These include, but are not limited to, D221E/P228E/L368E paired with D221R/P228R/K409R (e.g. these are "monomer corresponding sets) and C220E/P228E/368E paired with C220R/E224R/P228R/K409R.
[0262] Additional monomer A and monomer B variants that can be combined with other variants, optionally and independently in any amount, such as pI variants outlined herein or other steric variants that are shown in FIG. 37 of US 2012/0149876, the figure and legend and SEQ ID NOs of which are incorporated expressly by reference herein.
[0263] In some embodiments, the steric variants outlined herein can be optionally and independently incorporated with any pI variant (or other variants such as Fc variants, FcRn variants, etc.) into one or both monomers, and can be independently and optionally included or excluded from the proteins of the invention.
pI (Isoelectric Point) Variants for Heterodimers
[0264] In general, as will be appreciated by those in the art, there are two general categories of pI variants: those that increase the pI of the protein (basic changes) and those that decrease the pI of the protein (acidic changes). As described herein, all combinations of these variants can be done: one monomer may be wild type, or a variant that does not display a significantly different pI from wild-type, and the other can be either more basic or more acidic. Alternatively, each monomer is changed, one to more basic and one to more acidic.
[0265] Combinations of pI variants are shown in the figures.
Heavy Chain pI Changes
[0266] As outlined herein and shown in the figures, PI variants are shown relative to IgG1, but all isotypes can be altered this way, as well as isotype hybrids. In the case where the heavy chain constant domain is from IgG2-4, R133E and R133Q can also be used.
Antibody Heterodimers Light Chain Variants
[0267] In the case of antibody based heterodimers, e.g. where at least one of the monomers comprises a light chain in addition to the heavy chain domain, pI variants can also be made in the light chain. Amino acid substitutions for lowering the pI of the light chain include, but are not limited to, K126E, K126Q, K145E, K145Q, N152D, S156E, K169E, S202E, K207E and adding peptide DEDE at the c-terminus of the light chain. Changes in this category based on the constant lambda light chain include one or more substitutions at R108Q, Q124E, K126Q, N138D, K145T and Q199E. In addition, increasing the pI of the light chains can also be done.
Isotypic Variants
[0268] In addition, many embodiments of the invention rely on the "importation" of pI amino acids at particular positions from one IgG isotype into another, thus reducing or eliminating the possibility of unwanted immunogenicity being introduced into the variants. A number of these are shown in FIGS. 10A and 10B. That is, IgG1 is a common isotype for therapeutic antibodies for a variety of reasons, including high effector function. However, the heavy constant region of IgG1 has a higher pI than that of IgG2 (8.10 versus 7.31). By introducing IgG2 residues at particular positions into the IgG1 backbone, the pI of the resulting monomer is lowered (or increased) and additionally exhibits longer serum half-life. For example, IgG1 has a glycine (pI 5.97) at position 137, and IgG2 has a glutamic acid (pI 3.22); importing the glutamic acid will affect the pI of the resulting protein. As is described below, a number of amino acid substitutions are generally required to significant affect the pI of the variant antibody. However, it should be noted as discussed below that even changes in IgG2 molecules allow for increased serum half-life.
[0269] In other embodiments, non-isotypic amino acid changes are made, either to reduce the overall charge state of the resulting protein (e.g. by changing a higher pI amino acid to a lower pI amino acid), or to allow accommodations in structure for stability, etc. as is more further described below.
[0270] In addition, by pI engineering both the heavy and light constant domains, significant changes in each monomer of the heterodimer can be seen. As discussed herein, having the pIs of the two monomers differ by at least 0.5 can allow separation by ion exchange chromatography or isoelectric focusing, or other methods sensitive to isoelectric point.
Calculating pI
[0271] The pI of each monomer can depend on the pI of the variant heavy chain constant domain and the pI of the total monomer, including the variant heavy chain constant domain and the fusion partner. Thus, in some embodiments, the change in pI is calculated on the basis of the variant heavy chain constant domain, using the chart in FIGS. 46A-46C and 47. As discussed herein, which monomer to engineer is generally decided by the inherent pI of the Fv and scaffold regions. Alternatively, the pI of each monomer can be compared.
[0272] Heterodimeric Fc Fusion Proteins
[0273] In addition to heterodimeric antibodies, the invention provides heterodimeric proteins that comprise a first monomer comprising a variant Fc region and a first fusion partner and a second monomer, also comprising a variant Fc region and a second fusion partner. The variant Fc regions are engineered as herein for antibodies, and are thus different, and in general the first and second fusion partners are different as well. In some cases, where one monomer is antibody based (e.g. either comprising a standard heavy and light chain or a Fc domain with an scFv) and the other is an Fc fusion protein, the resulting heterodimeric protein is called a "fusionbody".
pI Variants that Also Confer Better FcRn In Vivo Binding
[0274] In the case where the pI variant decreases the pI of the monomer, they can have the added benefit of improving serum retention in vivo.
[0275] Although still under examination, Fc regions are believed to have longer half-lives in vivo, because binding to FcRn at pH 6 in an endosome sequesters the Fc (Ghetie and Ward, 1997 Immunol Today. 18(12): 592-598, entirely incorporated by reference). The endosomal compartment then recycles the Fc to the cell surface. Once the compartment opens to the extracellular space, the higher pH, .about.7.4, induces the release of Fc back into the blood. In mice, Dall' Acqua et al. showed that Fc mutants with increased FcRn binding at pH 6 and pH 7.4 actually had reduced serum concentrations and the same half life as wild-type Fc (Dall' Acqua et al. 2002, J. Immunol. 169:5171-5180, entirely incorporated by reference). The increased affinity of Fc for FcRn at pH 7.4 is thought to forbid the release of the Fc back into the blood. Therefore, the Fc mutations that will increase Fc's half-life in vivo will ideally increase FcRn binding at the lower pH while still allowing release of Fc at higher pH. The amino acid histidine changes its charge state in the pH range of 6.0 to 7.4. Therefore, it is not surprising to find His residues at important positions in the Fc/FcRn complex.
[0276] Recently it has been suggested that antibodies with variable regions that have lower isoelectric points may also have longer serum half-lives (Igawa et al., 2010 PEDS. 23(5): 385-392, entirely incorporated by reference). However, the mechanism of this is still poorly understood. Moreover, variable regions differ from antibody to antibody. Constant region variants with reduced pI and extended half-life would provide a more modular approach to improving the pharmacokinetic properties of antibodies, as described herein.
[0277] pI variants that find use in this embodiment, as well as their use for purification optimization, are disclosed in FIG. 20.
Combination of Heterodimeric Variants
[0278] As will be appreciated by those in the art, all of the recited heterodimerization variants can be optionally and independently combined in any way, as long as they retain their "strandedness" or "monomer partition". In addition, all of these variants can be combined into any of the heterodimerization formats.
[0279] In the case of pI variants, while embodiments finding particular use are shown in the Figures, other combinations can be generated, following the basic rule of altering the pI difference between two monomers to facilitate purification.
Nucleic Acids of the Invention
[0280] As discussed above regarding methods of making compositions of the present invention, the invention further provides nucleic acid compositions encoding the heterodimeric proteins of the invention. As will be appreciated by those in the art, the nucleic acid compositions will depend on the format and scaffold of the heterodimeric protein. Thus, for example, when the format requires three amino acid sequences, such as for the triple F format (e.g. a first amino acid monomer comprising an Fc domain and a scFv, a second amino acid monomer comprising a heavy chain and a light chain), three nucleic acid sequences can be incorporated into one or more expression vectors for expression. Similarly, some formats (e.g. dual scFv formats such as disclosed in FIG. 36M) only two nucleic acids are needed; again, they can be put into one or two expression vectors.
Target Antigens
[0281] The heterodimeric proteins of the invention may target virtually any antigens. The "triple F" format is particularly beneficial for targeting two (or more) distinct antigens. (As outlined herein, this targeting can be any combination of monovalent and divalent binding, depending on the format). Thus the immunoglobulins herein preferably co-engage two target antigens, although in some cases, three or four antigens can be monovalently engaged. Each monomer's specificity can be selected from the lists below. While the triple F immunoglobulins described herein are particularly beneficial for targeting distinct antigens, in some cases it may be beneficial to target only one antigen. That is, each monomer may have specificity for the same antigen.
[0282] Particular suitable applications of the heterodimeric proteins herein are co-target pairs for which it is beneficial or critical to engage each target antigen monovalently. Such antigens may be, for example, immune receptors that are activated upon immune complexation. Cellular activation of many immune receptors occurs only by cross-linking, achieved typically by antibody/antigen immune complexes, or via effector cell to target cell engagement. For some immune receptors, activation only upon engagement with co-engaged target is critical, as nonspecific cross-linking in a clinical setting can elicit a cytokine storm and toxicity. Therapeutically, by engaging such antigens monovalently rather than multivalently, using the immunoglobulins herein, such activation occurs only in response to cross-linking only in the microenvironment of the primary target antigen. The ability to target two different antigens with different valencies is a novel and useful aspect of the present invention. Examples of target antigens for which it may be therapeutically beneficial or necessary to co-engage monovalently include but are not limited to immune activating receptors such as CD3, Fc.gamma.Rs, toll-like receptors (TLRs) such as TLR4 and TLR9, cytokine, chemokine, cytokine receptors, and chemokine receptors. In many embodiments, one of the antigen binding sites binds to CD3, and in some embodiments it is the scFv-containing monomer.
[0283] Virtually any antigen may be targeted by the immunoglobulins herein, including but not limited to proteins, subunits, domains, motifs, and/or epitopes belonging to the following list of target antigens, which includes both soluble factors such as cytokines and membrane-bound factors, including transmembrane receptors: 17-IA, 4-1BB, 4Dc, 6-keto-PGF1a, 8-iso-PGF2a, 8-oxo-dG, A1 Adenosine Receptor, A33, ACE, ACE-2, Activin, Activin A, Activin AB, Activin B, Activin C, Activin RIA, Activin RIA ALK-2, Activin RIB ALK-4, Activin RIIA, Activin RIIB, ADAM, ADAM10, ADAM12, ADAM15, ADAM17/TACE, ADAMS, ADAM9, ADAMTS, ADAMTS4, ADAMTS5, Addressins, aFGF, ALCAM, ALK, ALK-1, ALK-7, alpha-1-antitrypsin, alpha-V/beta-1 antagonist, ANG, Ang, APAF-1, APE, APJ, APP, APRIL, AR, ARC, ART, Artemin, anti-Id, ASPARTIC, Atrial natriuretic factor, av/b3 integrin, Axl, b2M, B7-1, B7-2, B7-H, B-lymphocyte Stimulator (BlyS), BACE, BACE-1, Bad, BAFF, BAFF-R, Bag-1, BAK, Bax, BCA-1, BCAM, Bcl, BCMA, BDNF, b-ECGF, bFGF, BID, Bik, BIM, BLC, BL-CAM, BLK, BMP, BMP-2 BMP-2a, BMP-3 Osteogenin, BMP-4 BMP-2b, BMP-5, BMP-6 Vgr-1, BMP-7 (OP-1), BMP-8 (BMP-8a, OP-2), BMPR, BMPR-IA (ALK-3), BMPR-IB (ALK-6), BRK-2, RPK-1, BMPR-II (BRK-3), BMPs, b-NGF, BOK, Bombesin, Bone-derived neurotrophic factor, BPDE, BPDE-DNA, BTC, complement factor 3 (C3), C3a, C4, C5, C5a, C10, CA125, CAD-8, Calcitonin, cAMP, carcinoembryonic antigen (CEA), carcinoma-associated antigen, Cathepsin A, Cathepsin B, Cathepsin C/DPPI, Cathepsin D, Cathepsin E, Cathepsin H, Cathepsin L, Cathepsin O, Cathepsin S, Cathepsin V, Cathepsin X/Z/P, CBL, CCI, CCK2, CCL, CCL1, CCL11, CCL12, CCL13, CCL14, CCL15, CCL16, CCL17, CCL18, CCL19, CCL2, CCL20, CCL21, CCL22, CCL23, CCL24, CCL25, CCL26, CCL27, CCL28, CCL3, CCL4, CCL5, CCL6, CCL7, CCL8, CCL9/10, CCR, CCR1, CCR10, CCR10, CCR2, CCR3, CCR4, CCR5, CCR6, CCR7, CCR8, CCR9, CD1, CD2, CD3, CD3E, CD4, CD5, CD6, CD7, CD8, CD10, CD11a, CD11b, CD11c, CD13, CD14, CD15, CD16, CD18, CD19, CD20, CD21, CD22, CD23, CD25, CD27L, CD28, CD29, CD30, CD30L, CD32, CD33 (p67 proteins), CD34, CD38, CD40, CD40L, CD44, CD45, CD46, CD49a, CD52, CD54, CD55, CD56, CD61, CD64, CD66e, CD74, CD80 (B7-1), CD89, CD95, CD123, CD137, CD138, CD140a, CD146, CD147, CD148, CD152, CD164, CEACAM5, CFTR, cGMP, CINC, Clostridium botulinum toxin, Clostridium perfringens toxin, CKb8-1, CLC, CMV, CMV UL, CNTF, CNTN-1, COX, C-Ret, CRG-2, CT-1, CTACK, CTGF, CTLA-4, CX3CL1, CX3CR1, CXCL, CXCL1, CXCL2, CXCL3, CXCL4, CXCL5, CXCL6, CXCL7, CXCL8, CXCL9, CXCL10, CXCL11, CXCL12, CXCL13, CXCL14, CXCL15, CXCL16, CXCR, CXCR1, CXCR2, CXCR3, CXCR4, CXCR5, CXCR6, cytokeratin tumor-associated antigen, DAN, DCC, DcR3, DC-SIGN, Decay accelerating factor, des(1-3)-IGF-1 (brain IGF-1), Dhh, digoxin, DNAM-1, Dnase, Dpp, DPPIV/CD26, Dtk, ECAD, EDA, EDA-A1, EDA-A2, EDAR, EGF, EGFR (ErbB-1), EMA, EMMPRIN, ENA, endothelin receptor, Enkephalinase, eNOS, Eot, eotaxin1, EpCAM, Ephrin B2/EphB4, EPO, ERCC, E-selectin, ET-1, Factor IIa, Factor VII, Factor VIIIc, Factor IX, fibroblast activation protein (FAP), Fas, FcR1, FEN-1, Ferritin, FGF, FGF-19, FGF-2, FGF3, FGF-8, FGFR, FGFR-3, Fibrin, FL, FLIP, Flt-3, Flt-4, Follicle stimulating hormone, Fractalkine, FZD1, FZD2, FZD3, FZD4, FZD5, FZD6, FZD7, FZD8, FZD9, FZD10, G250, Gas 6, GCP-2, GCSF, GD2, GD3, GDF, GDF-1, GDF-3 (Vgr-2), GDF-5 (BMP-14, CDMP-1), GDF-6 (BMP-13, CDMP-2), GDF-7 (BMP-12, CDMP-3), GDF-8 (Myostatin), GDF-9, GDF-15 (MIC-1), GDNF, GDNF, GFAP, GFRa-1, GFR-alpha1, GFR-alpha2, GFR-alpha3, GITR, Glucagon, Glut 4, glycoprotein IIb/IIIa (GP IIb/IIIa), GM-CSF, gp130, gp72, GRO, Growth hormone releasing factor, Hapten (NP-cap or NIP-cap), HB-EGF, HCC, HCMV gB envelope glycoprotein, HCMV) gH envelope glycoprotein, HCMV UL, Hemopoietic growth factor (HGF), Hep B gp120, heparanase, Her2, Her2/neu (ErbB-2), Her3 (ErbB-3), Her4 (ErbB-4), herpes simplex virus (HSV) gB glycoprotein, HSV gD glycoprotein, HGFA, High molecular weight melanoma-associated antigen (HMW-MAA), HIV gp120, HIV IIIB gp 120 V3 loop, HLA, HLA-DR, HM1.24, HMFG PEM, HRG, Hrk, human cardiac myosin, human cytomegalovirus (HCMV), human growth hormone (HGH), HVEM, I-309, IAP, ICAM, ICAM-1, ICAM-3, ICE, ICOS, IFNg, Ig, IgA receptor, IgE, IGF, IGF binding proteins, IGF-1R, IGFBP, IGF-I, IGF-II, IL, IL-1, IL-1R, IL-2, IL-2R, IL-4, IL-4R, IL-5, IL-5R, IL-6, IL-6R, IL-8, IL-9, IL-10, IL-12, IL-13, IL-15, IL-18, IL-18R, IL-23, interferon (INF)-alpha, INF-beta, INF-gamma, Inhibin, iNOS, Insulin A-chain, Insulin B-chain, Insulin-like growth factor 1, integrin alpha2, integrin alpha3, integrin alpha4, integrin alpha4/beta1, integrin alpha4/beta7, integrin alpha5 (alphaV), integrin alpha5/beta1, integrin alpha5/beta3, integrin alpha6, integrin beta1, integrin beta2, interferon gamma, IP-10, I-TAC, JE, Kallikrein 2, Kallikrein 5, Kallikrein 6, Kallikrein 11, Kallikrein 12, Kallikrein 14, Kallikrein 15, Kallikrein L1, Kallikrein L2, Kallikrein L3, Kallikrein L4, KC, KDR, Keratinocyte Growth Factor (KGF), laminin 5, LAMP, LAP, LAP (TGF-1), Latent TGF-1, Latent TGF-1 bp1, LBP, LDGF, LECT2, Lefty, Lewis-Y antigen, Lewis-Y related antigen, LFA-1, LFA-3, Lfo, LIF, LIGHT, lipoproteins, LIX, LKN, Lptn, L-Selectin, LT-a, LT-b, LTB4, LTBP-1, Lung surfactant, Luteinizing hormone, Lymphotoxin Beta Receptor, Mac-1, MAdCAM, MAG, MAP2, MARC, MCAM, MCAM, MCK-2, MCP, M-CSF, MDC, Mer, METALLOPROTEASES, MGDF receptor, MGMT, MHC (HLA-DR), MIF, MIG, MIP, MIP-1-alpha, MK, MMAC1, MMP, MMP-1, MMP-10, MMP-11, MMP-12, MMP-13, MMP-14, MMP-15, MMP-2, MMP-24, MMP-3, MMP-7, MMP-8, MMP-9, MPIF, Mpo, MSK, MSP, mucin (Muc1), MUC18, Muellerian-inhibitin substance, Mug, MuSK, NAIP, NAP, NCAD, N-Cadherin, NCA 90, NCAM, NCAM, Neprilysin, Neurotrophin-3,-4, or -6, Neurturin, Neuronal growth factor (NGF), NGFR, NGF-beta, nNOS, NO, NOS, Npn, NRG-3, NT, NTN, OB, OGG1, OPG, OPN, OSM, OX40L, OX40R, p150, p95, PADPr, Parathyroid hormone, PARC, PARP, PBR, PBSF, PCAD, P-Cadherin, PCNA, PDGF, PDGF, PDK-1, PECAM, PEM, PF4, PGE, PGF, PGI2, PGJ2, PIN, PLA2, placental alkaline phosphatase (PLAP), PIGF, PLP, PP14, Proinsulin, Prorelaxin, Protein C, PS, PSA, PSCA, prostate specific membrane antigen (PSMA), PTEN, PTHrp, Ptk, PTN, R51, RANK, RANKL, RANTES, RANTES, Relaxin A-chain, Relaxin B-chain, renin, respiratory syncytial virus (RSV) F, RSV Fgp, Ret, Rheumatoid factors, RLIP76, RPA2, RSK, S100, SCF/KL, SDF-1, SERINE, Serum albumin, sFRP-3, Shh, SIGIRR, SK-1, SLAM, SLPI, SMAC, SMDF, SMOH, SOD, SPARC, Stat, STEAP, STEAP-II, TACE, TACI, TAG-72 (tumor-associated glycoprotein-72), TARC, TCA-3, T-cell receptors (e.g., T-cell receptor alpha/beta), TdT, TECK, TEM1, TEM5, TEM7, TEM8, TERT, testicular PLAP-like alkaline phosphatase, TfR, TGF, TGF-alpha, TGF-beta, TGF-beta Pan Specific, TGF-beta RI (ALK-5), TGF-beta RII, TGF-beta RIIb, TGF-beta RIII, TGF-beta1, TGF-beta2, TGF-beta3, TGF-beta4, TGF-beta5, Thrombin, Thymus Ck-1, Thyroid stimulating hormone, Tie, TIMP, TIQ, Tissue Factor, TMEFF2, Tmpo, TMPRSS2, TNF, TNF-alpha, TNF-alpha beta, TNF-beta2, TNFc, TNF-RI, TNF-RII, TNFRSF10A (TRAIL R1 Apo-2, DR4), TNFRSF10B (TRAIL R2 DR5, KILLER, TRICK-2A, TRICK-B), TNFRSF10C (TRAIL R3 DcR1, LIT, TRID), TNFRSF10D (TRAIL R4 DcR2, TRUNDD), TNFRSF11A (RANK ODF R, TRANCE R), TNFRSF11B (OPG OCIF, TR1), TNFRSF12 (TWEAK R FN14), TNFRSF13B (TACI), TNFRSF13C (BAFF R), TNFRSF14 (HVEM ATAR, HveA, LIGHT R, TR2), TNFRSF16 (NGFR p75NTR), TNFRSF17 (BCMA), TNFRSF18 (GITR AITR), TNFRSF19 (TROY TAJ, TRADE), TNFRSF19L (RELT), TNFRSF1A (TNF RI CD120a, p55-60), TNFRSF1B (TNF RII CD120b, p75-80), TNFRSF26 (TNFRH3), TNFRSF3 (LTbR TNF RIII, TNFC R), TNFRSF4 (OX40 ACT35, TXGP1 R), TNFRSF5 (CD40 p50), TNFRSF6 (Fas Apo-1, APT1, CD95), TNFRSF6B (DcR3 M68, TR6), TNFRSF7 (CD27), TNFRSF8 (CD30), TNFRSF9 (4-1BB CD137, ILA), TNFRSF21 (DR6), TNFRSF22 (DcTRAIL R2 TNFRH2), TNFRST23 (DcTRAIL R1 TNFRH1), TNFRSF25 (DR3 Apo-3, LARD, TR-3, TRAMP, WSL-1), TNFSF10 (TRAIL Apo-2 Ligand, TL2), TNFSF11 (TRANCE/RANK Ligand ODF, OPG Ligand), TNFSF12 (TWEAK Apo-3 Ligand, DR3 Ligand), TNFSF13 (APRIL TALL2), TNFSF13B (BAFF BLYS, TALL1, THANK, TNFSF20), TNFSF14 (LIGHT HVEM Ligand, LTg), TNFSF15 (TL1A/VEGI), TNFSF18 (GITR Ligand AITR Ligand, TL6), TNFSF1A (TNF-.alpha. Conectin, DIF, TNFSF2), TNFSF1B (TNF-b LTa, TNFSF1), TNFSF3 (LTb TNFC, p33), TNFSF4 (OX40 Ligand gp34, TXGP1), TNFSF5 (CD40 Ligand CD154, gp39, HIGM1, IMD3, TRAP), TNFSF6 (Fas Ligand Apo-1 Ligand, APT1 Ligand), TNFSF7 (CD27 Ligand CD70), TNFSF8 (CD30 Ligand CD153), TNFSF9 (4-1BB Ligand CD137 Ligand), TP-1, t-PA, Tpo, TRAIL, TRAIL R, TRAIL-R1, TRAIL-R2, TRANCE, transferring receptor, TRF, Trk, TROP-2, TSG, TSLP, tumor-associated antigen CA 125, tumor-associated antigen expressing Lewis Y related carbohydrate, TWEAK, TXB2, Ung, uPAR, uPAR-1, Urokinase, VCAM, VCAM-1, VECAD, VE-Cadherin, VE-cadherin-2, VEFGR-1 (flt-1), VEGF, VEGFR, VEGFR-3 (flt-4), VEGI, VIM, Viral antigens, VLA, VLA-1, VLA-4, VNR integrin, von Willebrands factor, WIF-1, WNT1, WNT2, WNT2B/13, WNT3, WNT3A, WNT4, WNT5A, WNT5B, WNT6, WNT7A, WNT7B, WNT8A, WNT8B, WNT9A, WNT9A, WNT9B, WNT10A, WNT10B, WNT11, WNT16, XCL1, XCL2, XCR1, XCR1, XEDAR, XIAP, XPD, and receptors for hormones and growth factors. To form the bispecific or trispecific antibodies of the invention, antibodies to any combination of these antigens can be made; that is, each of these antigens can be optionally and independently included or excluded from a multispecific antibody according to the present invention.
[0284] Exemplary antigens that may be targeted specifically by the immunoglobulins of the invention include but are not limited to: CD20, CD19, Her2, EGFR, EpCAM, CD3, Fc.gamma.RIIIa (CD16), Fc.gamma.RIIa (CD32a), Fc.gamma.RIIb (CD32b), Fc.gamma.RI (CD64), Toll-like receptors (TLRs) such as TLR4 and TLR9, cytokines such as IL-2, IL-5, IL-13, IL-12, IL-23, and TNF.alpha., cytokine receptors such as IL-2R, chemokines, chemokine receptors, growth factors such as VEGF and HGF, and the like. To form the multispecific antibodies of the invention, antibodies to any combination of these antigens can be made; that is, each of these antigens can be optionally and independently included or excluded from a multispecific antibody according to the present invention.
[0285] The choice of suitable target antigens and co-targets depends on the desired therapeutic application. Some targets that have proven especially amenable to antibody therapy are those with signaling functions. Other therapeutic antibodies exert their effects by blocking signaling of the receptor by inhibiting the binding between a receptor and its cognate ligand. Another mechanism of action of therapeutic antibodies is to cause receptor down regulation. Other antibodies do not work by signaling through their target antigen. The choice of co-targets will depend on the detailed biology underlying the pathology of the indication that is being treated.
[0286] Monoclonal antibody therapy has emerged as an important therapeutic modality for cancer (Weiner et al., 2010, Nature Reviews Immunology 10:317-327; Reichert et al., 2005, Nature Biotechnology 23[9]:1073-1078; herein expressly incorporated by reference). For anti-cancer treatment it may be desirable to target one antigen (antigen-1) whose expression is restricted to the cancerous cells while co-targeting a second antigen (antigen-2) that mediates some immunological killing activity. For other treatments, it may be beneficial to co-target two antigens, for example two angiogenic factors or two growth factors, that are each known to play some role in proliferation of the tumor. Exemplary co-targets for oncology include but are not limited to HGF and VEGF, IGF-1R and VEGF, Her2 and VEGF, CD19 and CD3, CD20 and CD3, Her2 and CD3, CD19 and Fc.gamma.RIIIa, CD20 and Fc.gamma.RIIIa, Her2 and Fc.gamma.RIIIa. An immunoglobulin of the invention may be capable of binding VEGF and phosphatidylserine; VEGF and ErbB3; VEGF and PLGF; VEGF and ROBO4; VEGF and BSG2; VEGF and CDCP1; VEGF and ANPEP; VEGF and c-MET; HER-2 and ERB3; HER-2 and BSG2; HER-2 and CDCP1; HER-2 and ANPEP; EGFR and CD64; EGFR and BSG2; EGFR and CDCP1; EGFR and ANPEP; IGF1R and PDGFR; IGF1R and VEGF; IGF1R and CD20; CD20 and CD74; CD20 and CD30; CD20 and DR4; CD20 and VEGFR2; CD20 and CD52; CD20 and CD4; HGF and c-MET; HGF and NRP1; HGF and phosphatidylserine; ErbB3 and IGF1R; ErbB3 and IGF1,2; c-Met and Her-2; c-Met and NRP1; c-Met and IGF1R; IGF1,2 and PDGFR; IGF1,2 and CD20; IGF1,2 and IGF1R; IGF2 and EGFR; IGF2 and HER2; IGF2 and CD20; IGF2 and VEGF; IGF2 and IGF1R; IGF1 and IGF2; PDGFRa and VEGFR2; PDGFRa and PLGF; PDGFRa and VEGF; PDGFRa and c-Met; PDGFRa and EGFR; PDGFRb and VEGFR2; PDGFRb and c-Met; PDGFRb and EGFR; RON and c-Met; RON and MTSP1; RON and MSP; RON and CDCP1; VGFR1 and PLGF; VGFR1 and RON; VGFR1 and EGFR; VEGFR2 and PLGF; VEGFR2 and NRP1; VEGFR2 and RON; VEGFR2 and DLL4; VEGFR2 and EGFR; VEGFR2 and ROBO4; VEGFR2 and CD55; LPA and S1P; EPHB2 and RON; CTLA4 and VEGF; CD3 and EPCAM; CD40 and IL6; CD40 and IGF; CD40 and CD56; CD40 and CD70; CD40 and VEGFR1; CD40 and DR5; CD40 and DR4; CD40 and APRIL; CD40 and BCMA; CD40 and RANKL; CD28 and MAPG; CD80 and CD40; CD80 and CD30; CD80 and CD33; CD80 and CD74; CD80 and CD2; CD80 and CD3; CD80 and CD19; CD80 and CD4; CD80 and CD52; CD80 and VEGF; CD80 and DR5; CD80 and VEGFR2; CD22 and CD20; CD22 and CD80; CD22 and CD40; CD22 and CD23; CD22 and CD33; CD22 and CD74; CD22 and CD19; CD22 and DR5; CD22 and DR4; CD22 and VEGF; CD22 and CD52; CD30 and CD20; CD30 and CD22; CD30 and CD23; CD30 and CD40; CD30 and VEGF; CD30 and CD74; CD30 and CD19; CD30 and DR5; CD30 and DR4; CD30 and VEGFR2; CD30 and CD52; CD30 and CD4; CD138 and RANKL; CD33 and FTL3; CD33 and VEGF; CD33 and VEGFR2; CD33 and CD44; CD33 and DR4; CD33 and DR5; DR4 and CD137; DR4 and IGF1,2; DR4 and IGF1R; DR4 and DR5; DR5 and CD40; DR5 and CD137; DR5 and CD20; DR5 and EGFR; DR5 and IGF1,2; DR5 and IGFR, DR5 and HER-2, and EGFR and DLL4. Other target combinations include one or more members of the EGF/erb-2/erb-3 family.
[0287] Other targets (one or more) involved in oncological diseases that the immunoglobulins herein may bind include, but are not limited to those selected from the group consisting of: CD52, CD20, CD19, CD3, CD4, CD8, BMP6, IL12A, IL1A, IL1B, 1L2, IL24, INHA, TNF, TNFSF10, BMP6, EGF, FGF1, FGF10, FGF11, FGF12, FGF13, FGF14, FGF16, FGF17, FGF18, FGF19, FGF2, FGF20, FGF21, FGF22, FGF23, FGF3, FGF4, FGF5, FGF6, FGF7, FGF8, FGF9, GRP, IGF1, IGF2, IL12A, IL1A, IL1B, 1L2, INHA, TGFA, TGFB1, TGFB2, TGFB3, VEGF, CDK2, FGF10, FGF18, FGF2, FGF4, FGF7, IGF1R, IL2, BCL2, CD164, CDKN1A, CDKN1B, CDKN1C, CDKN2A, CDKN2B, CDKN2C, CDKN3, GNRH1, IGFBP6, IL1A, IL1B, ODZ1, PAWR, PLG, TGFB1I1, AR, BRCA1, CDK3, CDK4, CDK5, CDK6, CDK7, CDK9, E2F1, EGFR, ENO1, ERBB2, ESR1, ESR2, IGFBP3, IGFBP6, IL2, INSL4, MYC, NOX5, NR6A1, PAP, PCNA, PRKCQ, PRKD1, PRL, TP53, FGF22, FGF23, FGF9, IGFBP3, IL2, INHA, KLK6, TP53, CHGB, GNRH1, IGF1, IGF2, INHA, INSL3, INSL4, PRL, KLK6, SHBG, NR1D1, NR1H3, NR113, NR2F6, NR4A3, ESR1, ESR2, NROB1, NROB2, NR1D2, NR1H2, NR1H4, NR112, NR2C1, NR2C2, NR2E1, NR2E3, NR2F1, NR2F2, NR3C1, NR3C2, NR4A1, NR4A2, NR5A1, NR5A2, NR6 p1, PGR, RARB, FGF1, FGF2, FGF6, KLK3, KRT1, APOC1, BRCA1, CHGA, CHGB, CLU, COL1A1, COL6A1, EGF, ERBB2, ERK8, FGF1, FGF10, FGF11, FGF13, FGF14, FGF16, FGF17, FGF18, FGF2, FGF20, FGF21, FGF22, FGF23, FGF3, FGF4, FGF5, FGF6, FGF7, FGF8, FGF9, GNRH1, IGF1, IGF2, IGFBP3, IGFBP6, IL12A, IL1A, IL1B, 1L2, IL24, INHA, INSL3, INSL4, KLK10, KLK12, KLK13, KLK14, KLK15, KLK3, KLK4, KLK5, KLK6, KLK9, MMP2, MMP9, MSMB, NTN4, ODZ1, PAP, PLAU, PRL, PSAP, SERPINA3, SHBG, TGFA, TIMP3, CD44, CDH1, CDH10, CDH19, CDH20, CDH7, CDH9, CDH1, CDH10, CDH13, CDH18, CDH19, CDH2O, CDH7, CDH8, CDH9, ROBO2, CD44, ILK, ITGA1, APC, CD164, COL6A1, MTSS1, PAP, TGFB111, AGR2, AIG1, AKAP1, AKAP2, CANT1, CAV1, CDH12, CLDN3, CLN3, CYB5, CYC1, DAB21P, DES, DNCL1, ELAC2, ENO2, ENO3, FASN, FLJ12584, FLJ25530, GAGEB1, GAGEC1, GGT1, GSTP1, HIP1, HUMCYT2A, IL29, K6HF, KAI1, KRT2A, MIB1, PART1, PATE, PCA3, PIAS2, PIK3CG, PPID, PR1, PSCA, SLC2A2, SLC33 pI, SLC43 pI, STEAP, STEAP2, TPM1, TPM2, TRPC6, ANGPT1, ANGPT2, ANPEP, ECGF1, EREG, FGF1, FGF2, FIGF, FLT1, JAG1, KDR, LAMA5, NRP1, NRP2, PGF, PLXDC1, STAB 1, VEGF, VEGFC, ANGPTL3, BAI1, COL4A3, IL8, LAMA5, NRP1, NRP2, STAB 1, ANGPTL4, PECAM1, PF4, PROK2, SERPINF1, TNFAIP2, CCL11, CCL2, CXCL1, CXCL10, CXCL3, CXCL5, CXCL6, CXCL9, IFNA1, IFNB1, IFNG, IL1B, IL6, MDK, EDG1, EFNA1, EFNA3, EFNB2, EGF, EPHB4, FGFR3, HGF, IGF1, ITGB3, PDGFA, TEK, TGFA, TGFB1, TGFB2, TGFBR1, CCL2, CDH5, COL1A1, EDG1, ENG, ITGAV, ITGB3, THBS1, THBS2, BAD, BAG1, BCL2, CCNA1, CCNA2, CCND1, CCNE1, CCNE2, CDH1 (E-cadherin), CDKN1B (p27Kip1), CDKN2A (p161NK4a), COL6A1, CTNNB1 (b-catenin), CTSB (cathepsin B), ERBB2 (Her-2), ESR1, ESR2, F3 (TF), FOSL1 (FRA-1), GATA3, GSN (Gelsolin), IGFBP2, IL2RA, IL6, IL6R, IL6ST (glycoprotein 130), ITGA6 (a6 integrin), JUN, KLK5, KRT19, MAP2K7 (c-Jun), MKI67 (Ki-67), NGFB (GF), NGFR, NME1 (M23A), PGR, PLAU (uPA), PTEN, SERPINB5 (maspin), SERPINE1 (PAI-1), TGFA, THBS1 (thrombospondin-1), TIE (Tie-1), TNFRSF6 (Fas), TNFSF6 (FasL), TOP2A (topoisomerase Iia), TP53, AZGP1 (zinc-a-glycoprotein), BPAG1 (plectin), CDKN1A (p21Wap1/Cip1), CLDN7 (claudin-7), CLU (clusterin), ERBB2 (Her-2), FGF1, FLRT1 (fibronectin), GABRP (GABAa), GNAS1, ID2, ITGA6 (a6 integrin), ITGB4 (b 4 integrin), KLF5 (GC Box BP), KRT19 (Keratin 19), KRTHB6 (hair-specific type II keratin), MACMARCKS, MT3 (metallothionectin-III), MUC1 (mucin), PTGS2 (COX-2), RAC2 (p21Rac2), S100A2, SCGB1D2 (lipophilin B), SCGB2A1 (mammaglobin 2), SCGB2A2 (mammaglobin 1), SPRR1B (Spr1), THBS1, THBS2, THBS4, and TNFAIP2 (B94), RON, c-Met, CD64, DLL4, PLGF, CTLA4, phophatidylserine, ROBO4, CD80, CD22, CD40, CD23, CD28, CD80, CD55, CD38, CD70, CD74, CD30, CD138, CD56, CD33, CD2, CD137, DR4, DR5, RANKL, VEGFR2, PDGFR, VEGFR1, MTSP1, MSP, EPHB2, EPHA1, EPHA2, EpCAM, PGE2, NKG2D, LPA, SIP, APRIL, BCMA, MAPG, FLT3, PDGFR alpha, PDGFR beta, ROR1, PSMA, PSCA, SCD1, and CD59. To form the bispecific or trispecific antibodies of the invention, antibodies to any combination of these antigens can be made; that is, each of these antigens can be optionally and independently included or excluded from a multispecific antibody according to the present invention.
[0288] Monoclonal antibody therapy has become an important therapeutic modality for treating autoimmune and inflammatory disorders (Chan & Carter, 2010, Nature Reviews Immunology 10:301-316; Reichert et al., 2005, Nature Biotechnology 23[9]:1073-1078; herein expressly incorporated by reference). Many proteins have been implicated in general autoimmune and inflammatory responses, and thus may be targeted by the immunoglobulins of the invention. Autoimmune and inflammatory targets include but are not limited to C5, CCL1 (1-309), CCL11 (eotaxin), CCL13 (mcp-4), CCL15 (MIP-1d), CCL16 (HCC-4), CCL17 (TARC), CCL18 (PARC), CCL19, CCL2 (mcp-1), CCL20 (MIP-3a), CCL21 (MIP-2), CCL23 (MPIF-1), CCL24 (MPIF-2/eotaxin-2), CCL25 (TECK), CCL26, CCL3 (MIP-1a), CCL4 (MIP-1b), CCL5 (RANTES), CCL7 (mcp-3), CCL8 (mcp-2), CXCL1, CXCL10 (IP-10), CXCL11 (1-TAC/IP-9), CXCL12 (SDF1), CXCL13, CXCL14, CXCL2, CXCL3, CXCL5 (ENA-78/LIX), CXCL6 (GCP-2), CXCL9, 1L13, IL8, CCL13 (mcp-4), CCR1, CCR2, CCR3, CCR4, CCR5, CCR6, CCR7, CCR8, CCR9, CX3CR1, IL8RA, XCR1 (CCXCR1), IFNA2, IL10, 1L13, IL17C, IL1A, IL1B, IL1F10, IL1F5, IL1F6, IL1F7, IL1F8, IL1F9, IL22, IL5, IL8, IL9, LTA, LTB, MIF, SCYE1 (endothelial Monocyte-activating cytokine), SPP1, TNF, TNFSF5, IFNA2, IL10RA, IL10RB, IL13, IL13RA1, IL5RA, IL9, IL9R, ABCF1, BCL6, C3, C4A, CEBPB, CRP, ICEBERG, IL1R1, IL1RN, IL8RB, LTB4R, TOLLIP, FADD, IRAK1, IRAK2, MYD88, NCK2, TNFAIP3, TRADD, TRAF1, TRAF2, TRAF3, TRAF4, TRAF5, TRAF6, ACVR1, ACVR1B, ACVR2, ACVR2B, ACVRL1, CD28, CD3E, CD3G, CD3Z, CD69, CD80, CD86, CNR1, CTLA4, CYSLTR1, FCER1A, FCER2, FCGR3A, GPR44, HAVCR2, OPRD1, P2RX7, TLR2, TLR3, TLR4, TLR5, TLR6, TLR7, TLR8, TLR9, TLR10, BLR1, CCL1, CCL2, CCL3, CCL4, CCL5, CCL7, CCL8, CCL11, CCL13, CCL15, CCL16, CCL17, CCL18, CCL19, CCL20, CCL21, CCL22, CCL23, CCL24, CCL25, CCR1, CCR2, CCR3, CCR4, CCR5, CCR6, CCR7, CCR8, CCR9, CX3CL1, CX3CR1, CXCL1, CXCL2, CXCL3, CXCL5, CXCL6, CXCL10, CXCL11, CXCL12, CXCL13, CXCR4, GPR2, SCYE1, SDF2, XCL1, XCL2, XCR1, AMH, AMHR2, BMPR1A, BMPR1B, BMPR2, C19orf10 (IL27w), CER1, CSF1, CSF2, CSF3, DKFZp451J0118, FGF2, GFI1, IFNA1, IFNB1, IFNG, IGF1, IL1A, IL1B, IL1R1, IL1R2, IL2, IL2RA, IL2RB, IL2RG, IL3, IL4, IL4R, IL5, IL5RA, IL6, IL6R, IL6ST, IL7, IL8, IL8RA, IL8RB, IL9, IL9R, IL10, MORA, IL10RB, IL11, IL12RA, IL12A, IL12B, IL12RB1, IL12RB2, 1L13, IL13RA1, IL13RA2, 1L15, IL15RA, IL16, 1L17, IL17R, IL18, IL18R1, 1L19, IL20, KITLG, LEP, LTA, LTB, LTB4R, LTB4R2, LTBR, MIF, NPPB, PDGFB, TBX21, TDGF1, TGFA, TGFB1, TGFB111, TGFB2, TGFB3, TGFB1, TGFBR1, TGFBR2, TGFBR3, TH1L, TNF, TNFRSF1A, TNFRSF1B, TNFRSF7, TNFRSF8, TNFRSF9, TNFRSF11A, TNFRSF21, TNFSF4, TNFSF5, TNFSF6, TNFSF11, VEGF, ZFPM2, and RNF110 (ZNF144). To form the bispecific or trispecific antibodies of the invention, antibodies to any combination of these antigens can be made; that is, each of these antigens can be optionally and independently included or excluded from a multispecific antibody according to the present invention.
[0289] Exemplary co-targets for autoimmune and inflammatory disorders include but are not limited to IL-1 and TNFalpha, IL-6 and TNFalpha, IL-6 and IL-1, IgE and IL-13, IL-1 and IL-13, IL-4 and IL-13, IL-5 and IL-13, IL-9 and IL-13, CD19 and Fc.gamma.RIIb, and CD79 and Fc.gamma.RIIb.
[0290] Immunoglobulins of the invention with specificity for the following pairs of targets to treat inflammatory disease are contemplated: TNF and IL-17A; TNF and RANKL; TNF and VEGF; TNF and SOST; TNF and DKK; TNF and alphaVbeta3; TNF and NGF; TNF and IL-23p19; TNF and IL-6; TNF and SOST; TNF and IL-6R; TNF and CD-20; IgE and IL-13; IL-13 and IL23p19; IgE and IL-4; IgE and IL-9; IgE and IL-9; IgE and IL-13; IL-13 and IL-9; IL-13 and IL-4; IL-13 and IL-9; IL-13 and IL-9; IL-13 and IL-4; IL-13 and IL-23p19; IL-13 and IL-9; IL-6R and VEGF; IL-6R and IL-17A; IL-6R and RANKL; IL-17A and IL-1beta; IL-1beta and RANKL; IL-1beta and VEGF; RANKL and CD-20; IL-1alpha and IL-1beta; IL-1alpha and IL-1beta.
[0291] Pairs of targets that the immunoglobulins described herein can bind and be useful to treat asthma may be determined. In an embodiment, such targets include, but are not limited to, IL-13 and IL-1beta, since IL-1beta is also implicated in inflammatory response in asthma; IL-13 and cytokines and chemokines that are involved in inflammation, such as IL-13 and IL-9; IL-13 and IL-4; IL-13 and IL-5; IL-13 and IL-25; IL-13 and TARC; IL-13 and MDC; IL-13 and MIF; IL-13 and TGF-.beta.; IL-13 and LHR agonist; IL-13 and CL25; IL-13 and SPRR2a; IL-13 and SPRR2b; and IL-13 and ADAMS. The immunoglobulins herein may have specificity for one or more targets involved in asthma selected from the group consisting of CSF1 (MCSF), CSF2 (GM-CSF), CSF3 (GCSF), FGF2, IFNA1, IFNB1, IFNG, histamine and histamine receptors, IL1A, IL1B, IL2, IL3, IL4, IL5, IL6, IL7, IL8, IL9, IL10, IL11, IL12A, IL12B, IL13, IL14, IL15, IL16, IL17, IL18, IL19, KITLG, PDGFB, IL2RA, IL4R, IL5RA, IL8RA, IL8RB, IL12RB1, IL12RB2, IL13RA1, IL13RA2, IL18R1, TSLP, CCLi, CCL2, CCL3, CCL4, CCL5, CCL7, CCL8, CCL13, CCL17, CCL18, CCL19, CCL20, CCL22, CCL24, CX3CL1, CXCL1, CXCL2, CXCL3, XCLi, CCR2, CCR3, CCR4, CCR5, CCR6, CCR7, CCR8, CX3CR1, GPR2, XCR1, FOS, GATA3, JAK1, JAK3, STATE, TBX21, TGFB1, TNF, TNFSF6, YY1, CYSLTR1, FCER1A, FCER2, LTB4R, TB4R2, LTBR, and Chitinase. To form the bispecific or trispecific antibodies of the invention, antibodies to any combination of these antigens can be made; that is, each of these antigens can be optionally and independently included or excluded from a multispecific antibody according to the present invention.
[0292] Pairs of targets involved in rheumatoid arthritis (RA) may be co-targeted by the invention, including but not limited to TNF and IL-18; TNF and IL-12; TNF and IL-23; TNF and IL-1beta; TNF and MIF; TNF and IL-17; and TNF and IL-15.
[0293] Antigens that may be targeted in order to treat systemic lupus erythematosus (SLE) by the immunoglobulins herein include but are not limited to CD-20, CD-22, CD-19, CD28, CD4, CD80, HLA-DRA, IL10, IL2, IL4, TNFRSF5, TNFRSF6, TNFSF5, TNFSF6, BLR1, HDAC4, HDAC5, HDAC7A, HDAC9, ICOSL, IGBP1, MS4A1, RGSI, SLA2, CD81, IFNB1, IL10, TNFRSF5, TNFRSF7, TNFSF5, AICDA, BLNK, GALNAC4S-6ST, HDAC4, HDAC5, HDAC7A, HDAC9, IL10, IL11, IL4, INHA, INHBA, KLF6, TNFRSF7, CD28, CD38, CD69, CD80, CD83, CD86, DPP4, FCER2, IL2RA, TNFRSF8, TNFSF7, CD24, CD37, CD40, CD72, CD74, CD79A, CD79B, CR2, ILIR2, ITGA2, ITGA3, MS4A1, ST6GALI, CDIC, CHSTIO, HLA-A, HLA-DRA, and NT5E.; CTLA4, B7.1, B7.2, BlyS, BAFF, C5, IL-4, IL-6, IL-10, IFN-.alpha., and TNF-.alpha.. To form the bispecific or trispecific antibodies of the invention, antibodies to any combination of these antigens can be made; that is, each of these antigens can be optionally and independently included or excluded from a multispecific antibody according to the present invention.
[0294] The immunoglobulins herein may target antigens for the treatment of multiple sclerosis (MS), including but not limited to IL-12, TWEAK, IL-23, CXCL13, CD40, CD40L, IL-18, VEGF, VLA-4, TNF, CD45RB, CD200, IFNgamma, GM-CSF, FGF, C5, CD52, and CCR2. An embodiment includes co-engagement of anti-IL-12 and TWEAK for the treatment of MS.
[0295] One aspect of the invention pertains to immunoglobulins capable of binding one or more targets involved in sepsis, in an embodiment two targets, selected from the group consisting TNF, IL-1, MIF, IL-6, IL-8, IL-18, IL-12, IL-23, FasL, LPS, Toll-like receptors, TLR-4, tissue factor, MIP-2, ADORA2A, CASP1, CASP4, IL-10, IL-1B, NFKB1, PROC, TNFRSFIA, CSF3, CCR3, ILIRN, MIF, NFKB1, PTAFR, TLR2, TLR4, GPR44, HMOX1, midkine, IRAK1, NFKB2, SERPINA1, SERPINE1, and TREM1. To form the bispecific or trispecific antibodies of the invention, antibodies to any combination of these antigens can be made; that is, each of these antigens can be optionally and independently included or excluded from a multispecific antibody according to the present invention.
[0296] In some cases, immunoglobulins herein may be directed against antigens for the treatment of infectious diseases.
Antigen Binding Domains
[0297] As will be appreciated by those in the art, there are two basic types of antigen binding domains, those that resemble antibody antigen binding domains (e.g. comprising a set of 6 CDRs) and those that can be ligands or receptors, for example, that bind to targets without the use of CDRs.
Modified Antibodies
[0298] In addition to the modifications outlined above, other modifications can be made. For example, the molecules may be stabilized by the incorporation of disulphide bridges linking the VH and VL domains (Reiter et al., 1996, Nature Biotech. 14:1239-1245, entirely incorporated by reference). In addition, there are a variety of covalent modifications of antibodies that can be made as outlined below.
[0299] Covalent modifications of antibodies are included within the scope of this invention, and are generally, but not always, done post-translationally. For example, several types of covalent modifications of the antibody are introduced into the molecule by reacting specific amino acid residues of the antibody with an organic derivatizing agent that is capable of reacting with selected side chains or the N- or C-terminal residues.
[0300] Cysteinyl residues most commonly are reacted with .alpha.-haloacetates (and corresponding amines), such as chloroacetic acid or chloroacetamide, to give carboxymethyl or carboxyamidomethyl derivatives. Cysteinyl residues may also be derivatized by reaction with bromotrifluoroacetone, .alpha.-bromo-.beta.-(5-imidozoyl)propionic acid, chloroacetyl phosphate, N-alkylmaleimides, 3-nitro-2-pyridyl disulfide, methyl 2-pyridyl disulfide, p-chloromercuribenzoate, 2-chloromercuri-4-nitrophenol, or chloro-7-nitrobenzo-2-oxa-1,3-diazole and the like.
[0301] In addition, modifications at cysteines are particularly useful in antibody-drug conjugate (ADC) applications, further described below. In some embodiments, the constant region of the antibodies can be engineered to contain one or more cysteines that are particularly "thiol reactive", so as to allow more specific and controlled placement of the drug moiety. See for example U.S. Pat. No. 7,521,541, incorporated by reference in its entirety herein.
[0302] Histidyl residues are derivatized by reaction with diethylpyrocarbonate at pH 5.5-7.0 because this agent is relatively specific for the histidyl side chain. Para-bromophenacyl bromide also is useful; the reaction is preferably performed in 0.1M sodium cacodylate at pH 6.0.
[0303] Lysinyl and amino terminal residues are reacted with succinic or other carboxylic acid anhydrides. Derivatization with these agents has the effect of reversing the charge of the lysinyl residues. Other suitable reagents for derivatizing alpha-amino-containing residues include imidoesters such as methyl picolinimidate; pyridoxal phosphate; pyridoxal; chloroborohydride; trinitrobenzenesulfonic acid; O-methylisourea; 2,4-pentanedione; and transaminase-catalyzed reaction with glyoxylate.
[0304] Arginyl residues are modified by reaction with one or several conventional reagents, among them phenylglyoxal, 2,3-butanedione, 1,2-cyclohexanedione, and ninhydrin. Derivatization of arginine residues requires that the reaction be performed in alkaline conditions because of the high pKa of the guanidine functional group. Furthermore, these reagents may react with the groups of lysine as well as the arginine epsilon-amino group.
[0305] The specific modification of tyrosyl residues may be made, with particular interest in introducing spectral labels into tyrosyl residues by reaction with aromatic diazonium compounds or tetranitromethane. Most commonly, N-acetylimidizole and tetranitromethane are used to form O-acetyl tyrosyl species and 3-nitro derivatives, respectively. Tyrosyl residues are iodinated using 125I or 131I to prepare labeled proteins for use in radioimmunoassay, the chloramine T method described above being suitable.
[0306] Carboxyl side groups (aspartyl or glutamyl) are selectively modified by reaction with carbodiimides (R'--N.dbd.C.dbd.N--R'), where R and R' are optionally different alkyl groups, such as 1-cyclohexyl-3-(2-morpholinyl-4-ethyl) carbodiimide or 1-ethyl-3-(4-azonia-4,4-dimethylpentyl) carbodiimide. Furthermore, aspartyl and glutamyl residues are converted to asparaginyl and glutaminyl residues by reaction with ammonium ions.
[0307] Derivatization with bifunctional agents is useful for crosslinking antibodies to a water-insoluble support matrix or surface for use in a variety of methods, in addition to methods described below. Commonly used crosslinking agents include, e.g., 1,1-bis(diazoacetyl)-2-phenylethane, glutaraldehyde, N-hydroxysuccinimide esters, for example, esters with 4-azidosalicylic acid, homobifunctional imidoesters, including disuccinimidyl esters such as 3,3'-dithiobis (succinimidylpropionate), and bifunctional maleimides such as bis-N-maleimido-1,8-octane. Derivatizing agents such as methyl-3-[(p-azidophenyl)dithio]propioimidate yield photoactivatable intermediates that are capable of forming crosslinks in the presence of light. Alternatively, reactive water-insoluble matrices such as cynomolgusogen bromide-activated carbohydrates and the reactive substrates described in U.S. Pat. Nos. 3,969,287; 3,691,016; 4,195,128; 4,247,642; 4,229,537; and 4,330,440, all entirely incorporated by reference, are employed for protein immobilization.
[0308] Glutaminyl and asparaginyl residues are frequently deamidated to the corresponding glutamyl and aspartyl residues, respectively. Alternatively, these residues are deamidated under mildly acidic conditions. Either form of these residues falls within the scope of this invention.
[0309] Other modifications include hydroxylation of proline and lysine, phosphorylation of hydroxyl groups of seryl or threonyl residues, methylation of the .alpha.-amino groups of lysine, arginine, and histidine side chains (T. E. Creighton, Proteins: Structure and Molecular Properties, W. H. Freeman & Co., San Francisco, pp. 79-86 [1983], entirely incorporated by reference), acetylation of the N-terminal amine, and amidation of any C-terminal carboxyl group.
[0310] In addition, as will be appreciated by those in the art, labels (including fluorescent, enzymatic, magnetic, radioactive, etc. can all be added to the antibodies (as well as the other compositions of the invention).
Glycosylation
[0311] Another type of covalent modification is alterations in glycosylation. In another embodiment, the antibodies disclosed herein can be modified to include one or more engineered glycoforms. By "engineered glycoform" as used herein is meant a carbohydrate composition that is covalently attached to the antibody, wherein said carbohydrate composition differs chemically from that of a parent antibody. Engineered glycoforms may be useful for a variety of purposes, including but not limited to enhancing or reducing effector function. A preferred form of engineered glycoform is afucosylation, which has been shown to be correlated to an increase in ADCC function, presumably through tighter binding to the Fc.gamma.RIIIa receptor. In this context, "afucosylation" means that the majority of the antibody produced in the host cells is substantially devoid of fucose, e.g. 90-95-98% of the generated antibodies do not have appreciable fucose as a component of the carbohydrate moiety of the antibody (generally attached at N297 in the Fc region). Defined functionally, afucosylated antibodies generally exhibit at least a 50% or higher affinity to the Fc.gamma.RIIIa receptor.
[0312] Engineered glycoforms may be generated by a variety of methods known in the art (Uma a et al., 1999, Nat Biotechnol 17:176-180; Davies et al., 2001, Biotechnol Bioeng 74:288-294; Shields et al., 2002, J Biol Chem 277:26733-26740; Shinkawa et al., 2003, J Biol Chem 278:3466-3473; U.S. Pat. No. 6,602,684; U.S. Ser. No. 10/277,370; U.S. Ser. No. 10/113,929; PCT WO 00/61739A1; PCT WO 01/29246A1; PCT WO 02/31140A1; PCT WO 02/30954A1, all entirely incorporated by reference; (Potelligent.RTM. technology [Biowa, Inc., Princeton, N.J.]; GlycoMAb.RTM. glycosylation engineering technology [Glycart Biotechnology AG, Zurich, Switzerland]). Many of these techniques are based on controlling the level of fucosylated and/or bisecting oligosaccharides that are covalently attached to the Fc region, for example by expressing an IgG in various organisms or cell lines, engineered or otherwise (for example Lec-13 CHO cells or rat hybridoma YB2/0 cells, by regulating enzymes involved in the glycosylation pathway (for example FUT8 [.alpha.1,6-fucosyltranserase] and/or .beta.1-4-N-acetylglucosaminyltransferase III [GnTIII]), or by modifying carbohydrate(s) after the IgG has been expressed. For example, the "sugar engineered antibody" or "SEA technology" of Seattle Genetics functions by adding modified saccharides that inhibit fucosylation during production; see for example 20090317869, hereby incorporated by reference in its entirety. Engineered glycoform typically refers to the different carbohydrate or oligosaccharide; thus an antibody can include an engineered glycoform.
[0313] Alternatively, engineered glycoform may refer to the IgG variant that comprises the different carbohydrate or oligosaccharide. As is known in the art, glycosylation patterns can depend on both the sequence of the protein (e.g., the presence or absence of particular glycosylation amino acid residues, discussed below), or the host cell or organism in which the protein is produced. Particular expression systems are discussed below.
[0314] Glycosylation of polypeptides is typically either N-linked or O-linked. N-linked refers to the attachment of the carbohydrate moiety to the side chain of an asparagine residue. The tri-peptide sequences asparagine-X-serine and asparagine-X-threonine, where X is any amino acid except proline, are the recognition sequences for enzymatic attachment of the carbohydrate moiety to the asparagine side chain. Thus, the presence of either of these tri-peptide sequences in a polypeptide creates a potential glycosylation site. O-linked glycosylation refers to the attachment of one of the sugars N-acetylgalactosamine, galactose, or xylose, to a hydroxyamino acid, most commonly serine or threonine, although 5-hydroxyproline or 5-hydroxylysine may also be used.
[0315] Addition of glycosylation sites to the antibody is conveniently accomplished by altering the amino acid sequence such that it contains one or more of the above-described tri-peptide sequences (for N-linked glycosylation sites). The alteration may also be made by the addition of, or substitution by, one or more serine or threonine residues to the starting sequence (for O-linked glycosylation sites). For ease, the antibody amino acid sequence is preferably altered through changes at the DNA level, particularly by mutating the DNA encoding the target polypeptide at preselected bases such that codons are generated that will translate into the desired amino acids.
[0316] Another means of increasing the number of carbohydrate moieties on the antibody is by chemical or enzymatic coupling of glycosides to the protein. These procedures are advantageous in that they do not require production of the protein in a host cell that has glycosylation capabilities for N- and O-linked glycosylation. Depending on the coupling mode used, the sugar(s) may be attached to (a) arginine and histidine, (b) free carboxyl groups, (c) free sulfhydryl groups such as those of cysteine, (d) free hydroxyl groups such as those of serine, threonine, or hydroxyproline, (e) aromatic residues such as those of phenylalanine, tyrosine, or tryptophan, or (f) the amide group of glutamine. These methods are described in WO 87/05330 and in Aplin and Wriston, 1981, CRC Crit. Rev. Biochem., pp. 259-306, both entirely incorporated by reference.
[0317] Removal of carbohydrate moieties present on the starting antibody (e.g. post-translationally) may be accomplished chemically or enzymatically. Chemical deglycosylation requires exposure of the protein to the compound trifluoromethanesulfonic acid, or an equivalent compound. This treatment results in the cleavage of most or all sugars except the linking sugar (N-acetylglucosamine or N-acetylgalactosamine), while leaving the polypeptide intact. Chemical deglycosylation is described by Hakimuddin et al., 1987, Arch. Biochem. Biophys. 259:52 and by Edge et al., 1981, Anal. Biochem. 118:131, both entirely incorporated by reference. Enzymatic cleavage of carbohydrate moieties on polypeptides can be achieved by the use of a variety of endo- and exo-glycosidases as described by Thotakura et al., 1987, Meth. Enzymol. 138:350, entirely incorporated by reference. Glycosylation at potential glycosylation sites may be prevented by the use of the compound tunicamycin as described by Duskin et al., 1982, J. Biol. Chem. 257:3105, entirely incorporated by reference. Tunicamycin blocks the formation of protein-N-glycoside linkages.
[0318] Another type of covalent modification of the antibody comprises linking the antibody to various nonproteinaceous polymers, including, but not limited to, various polyols such as polyethylene glycol, polypropylene glycol or polyoxyalkylenes, in the manner set forth in, for example, 2005-2006 PEG Catalog from Nektar Therapeutics (available at the Nektar website) U.S. Pat. Nos. 4,640,835; 4,496,689; 4,301,144; 4,670,417; 4,791,192 or 4,179,337, all entirely incorporated by reference. In addition, as is known in the art, amino acid substitutions may be made in various positions within the antibody to facilitate the addition of polymers such as PEG. See for example, U.S. Publication No. 2005/0114037A1, entirely incorporated by reference.
Additional Fc Variants for Additional Functionality
[0319] In addition to pI amino acid variants, there are a number of useful Fc amino acid modification that can be made for a variety of reasons, including, but not limited to, altering binding to one or more Fc.gamma.R receptors, altered binding to FcRn receptors, etc.
[0320] Accordingly, the proteins of the invention can include amino acid modifications, including the heterodimerization variants outlined herein, which includes the pI variants and steric variants. Each set of variants can be independently and optionally included or excluded from any particular heterodimeric protein.
Fc.gamma.R Variants
[0321] Accordingly, there are a number of useful Fc substitutions that can be made to alter binding to one or more of the Fc.gamma.R receptors. Substitutions that result in increased binding as well as decreased binding can be useful. For example, it is known that increased binding to Fc.quadrature.RIIIa generally results in increased ADCC (antibody dependent cell-mediated cytotoxicity; the cell-mediated reaction wherein nonspecific cytotoxic cells that express Fc.gamma.Rs recognize bound antibody on a target cell and subsequently cause lysis of the target cell). Similarly, decreased binding to Fc.gamma.RIIb (an inhibitory receptor) can be beneficial as well in some circumstances. Amino acid substitutions that find use in the present invention include those listed in U.S. Ser. No. 11/124,620 (particularly FIG. 41), Ser. Nos. 11/174,287, 11/396,495, 11/538,406, all of which are expressly incorporated herein by reference in their entirety and specifically for the variants disclosed therein. Particular variants that find use include, but are not limited to, 236A, 239D, 239E, 332E, 332D, 239D/332E, 267D, 267E, 328F, 267E/328F, 236A/332E, 239D/332E/330Y, 239D, 332E/330L, 243A, 243L, 264A, 264V and 299T.
[0322] In addition, there are additional Fc substitutions that find use in increased binding to the FcRn receptor and increased serum half life, as specifically disclosed in U.S. Ser. No. 12/341,769, hereby incorporated by reference in its entirety, including, but not limited to, 434S, 434A, 428L, 308F, 259I, 428L/434S, 259I/308F, 436I/428L, 436I or V/434S, 436V/428L and 259I/308F/428L.
Linkers
[0323] The present invention optionally provides linkers as needed, for example in the addition of additional antigen binding sites, as depicted for example in FIG. 2, where "the other end" of the molecule contains additional antigen binding components. In addition, as outlined below, linkers are optionally also used in antibody drug conjugate (ADC) systems. When used to join the components of the central mAb-Fv constructs, the linker is generally a polypeptide comprising two or more amino acid residues joined by peptide bonds and are used to link one or more of the components of the present invention. Such linker polypeptides are well known in the art (see e.g., Holliger, P., et al. (1993) Proc. Natl. Acad. Sci. USA 90:6444-6448; Poljak, R. J., et al. (1994) Structure 2:1121-1123). A variety of linkers may find use in some embodiments described herein. As will be appreciated by those in the art, there are at least three different linker types used in the present invention.
[0324] "Linker" herein is also referred to as "linker sequence", "spacer", "tethering sequence" or grammatical equivalents thereof. Homo- or hetero-bifunctional linkers as are well known (see, 1994 Pierce Chemical Company catalog, technical section on cross-linkers, pages 155-200, incorporated entirely by reference). A number of strategies may be used to covalently link molecules together. These include, but are not limited to polypeptide linkages between N- and C-termini of proteins or protein domains, linkage via disulfide bonds, and linkage via chemical cross-linking reagents. In one aspect of this embodiment, the linker is a peptide bond, generated by recombinant techniques or peptide synthesis. The linker peptide may predominantly include the following amino acid residues: Gly, Ser, Ala, or Thr. The linker peptide should have a length that is adequate to link two molecules in such a way that they assume the correct conformation relative to one another so that they retain the desired activity. In one embodiment, the linker is from about 1 to 50 amino acids in length, preferably about 1 to 30 amino acids in length. In one embodiment, linkers of 1 to 20 amino acids in length may be used. Useful linkers include glycine-serine polymers, including for example (GS)n, (GSGGS)n, (GGGGS)n, and (GGGS)n, where n is an integer of at least one, glycine-alanine polymers, alanine-serine polymers, and other flexible linkers. Alternatively, a variety of nonproteinaceous polymers, including but not limited to polyethylene glycol (PEG), polypropylene glycol, polyoxyalkylenes, or copolymers of polyethylene glycol and polypropylene glycol, may find use as linkers, that is may find use as linkers.
[0325] Other linker sequences may include any sequence of any length of CL/CH1 domain but not all residues of CL/CH1 domain; for example the first 5-12 amino acid residues of the CL/CH1 domains. Linkers can be derived from immunoglobulin light chain, for example C.kappa. or C.lamda. Linkers can be derived from immunoglobulin heavy chains of any isotype, including for example C.gamma.1, C.gamma.2, C.gamma.3, C.gamma.4, C.alpha.1, C.alpha.2, C.delta., C.epsilon., and C.mu.. Linker sequences may also be derived from other proteins such as Ig-like proteins (e.g. TCR, FcR, KIR), hinge region-derived sequences, and other natural sequences from other proteins.
Antibody-Drug Conjugates
[0326] In some embodiments, the multispecific antibodies of the invention are conjugated with drugs to form antibody-drug conjugates (ADCs). In general, ADCs are used in oncology applications, where the use of antibody-drug conjugates for the local delivery of cytotoxic or cytostatic agents allows for the targeted delivery of the drug moiety to tumors, which can allow higher efficacy, lower toxicity, etc. An overview of this technology is provided in Ducry et al., Bioconjugate Chem., 21:5-13 (2010), Carter et al., Cancer J. 14(3):154 (2008) and Senter, Current Opin. Chem. Biol. 13:235-244 (2009), all of which are hereby incorporated by reference in their entirety.
[0327] Thus the invention provides multispecific antibodies conjugated to drugs. Generally, conjugation is done by covalent attachment to the antibody, as further described below, and generally relies on a linker, often a peptide linkage (which, as described below, may be designed to be sensitive to cleavage by proteases at the target site or not). In addition, as described above, linkage of the linker-drug unit (LU-D) can be done by attachment to cysteines within the antibody. As will be appreciated by those in the art, the number of drug moieties per antibody can change, depending on the conditions of the reaction, and can vary from 1:1 to 10:1 drug:antibody. As will be appreciated by those in the art, the actual number is an average.
[0328] Thus the invention provides multispecific antibodies conjugated to drugs. As described below, the drug of the ADC can be any number of agents, including but not limited to cytotoxic agents such as chemotherapeutic agents, growth inhibitory agents, toxins (for example, an enzymatically active toxin of bacterial, fungal, plant, or animal origin, or fragments thereof), or a radioactive isotope (that is, a radioconjugate) are provided. In other embodiments, the invention further provides methods of using the ADCs.
[0329] Drugs for use in the present invention include cytotoxic drugs, particularly those which are used for cancer therapy. Such drugs include, in general, DNA damaging agents, anti-metabolites, natural products and their analogs. Exemplary classes of cytotoxic agents include the enzyme inhibitors such as dihydrofolate reductase inhibitors, and thymidylate synthase inhibitors, DNA intercalators, DNA cleavers, topoisomerase inhibitors, the anthracycline family of drugs, the vinca drugs, the mitomycins, the bleomycins, the cytotoxic nucleosides, the pteridine family of drugs, diynenes, the podophyllotoxins, dolastatins, maytansinoids, differentiation inducers, and taxols.
[0330] Members of these classes include, for example, methotrexate, methopterin, dichloromethotrexate, 5-fluorouracil, 6-mercaptopurine, cytosine arabinoside, melphalan, leurosine, leurosideine, actinomycin, daunorubicin, doxorubicin, mitomycin C, mitomycin A, caminomycin, aminopterin, tallysomycin, podophyllotoxin and podophyllotoxin derivatives such as etoposide or etoposide phosphate, vinblastine, vincristine, vindesine, taxanes including taxol, taxotere retinoic acid, butyric acid, N8-acetyl spermidine, camptothecin, calicheamicin, esperamicin, ene-diynes, duocarmycin A, duocarmycin SA, calicheamicin, camptothecin, maytansinoids (including DM1), monomethylauristatin E (MMAE), monomethylauristatin F (MMAF), and maytansinoids (DM4) and their analogues.
[0331] Toxins may be used as antibody-toxin conjugates and include bacterial toxins such as diphtheria toxin, plant toxins such as ricin, small molecule toxins such as geldanamycin (Mandler et al (2000) J. Nat. Cancer Inst. 92(19):1573-1581; Mandler et al (2000) Bioorganic & Med. Chem. Letters 10:1025-1028; Mandler et al (2002) Bioconjugate Chem. 13:786-791), maytansinoids (EP 1391213; Liu et al., (1996) Proc. Natl. Acad. Sci. USA 93:8618-8623), and calicheamicin (Lode et al (1998) Cancer Res. 58:2928; Hinman et al (1993) Cancer Res. 53:3336-3342). Toxins may exert their cytotoxic and cytostatic effects by mechanisms including tubulin binding, DNA binding, or topoisomerase inhibition.
[0332] Conjugates of a multispecific antibody and one or more small molecule toxins, such as a maytansinoids, dolastatins, auristatins, a trichothecene, calicheamicin, and CC1065, and the derivatives of these toxins that have toxin activity, are contemplated.
Maytansinoids
[0333] Maytansine compounds suitable for use as maytansinoid drug moieties are well known in the art, and can be isolated from natural sources according to known methods, produced using genetic engineering techniques (see Yu et al (2002) PNAS 99:7968-7973), or maytansinol and maytansinol analogues prepared synthetically according to known methods. As described below, drugs may be modified by the incorporation of a functionally active group such as a thiol or amine group for conjugation to the antibody.
[0334] Exemplary maytansinoid drug moieties include those having a modified aromatic ring, such as: C-19-dechloro (U.S. Pat. No. 4,256,746) (prepared by lithium aluminum hydride reduction of ansamytocin P2); C-20-hydroxy (or C-20-demethyl)+/-C-19-dechloro (U.S. Pat. Nos. 4,361,650 and 4,307,016) (prepared by demethylation using Streptomyces or Actinomyces or dechlorination using LAH); and C-20-demethoxy, C-20-acyloxy (--OCOR), +/-dechloro (U.S. Pat. No. 4,294,757) (prepared by acylation using acyl chlorides) and those having modifications at other positions
[0335] Exemplary maytansinoid drug moieties also include those having modifications such as: C-9-SH (U.S. Pat. No. 4,424,219) (prepared by the reaction of maytansinol with H2S or P2S5); C-14-alkoxymethyl(demethoxy/CH2OR) (U.S. Pat. No. 4,331,598); C-14-hydroxymethyl or acyloxymethyl (CH2OH or CH2OAc) (U.S. Pat. No. 4,450,254) (prepared from Nocardia); C-15-hydroxy/acyloxy (U.S. Pat. No. 4,364,866) (prepared by the conversion of maytansinol by Streptomyces); C-15-methoxy (U.S. Pat. Nos. 4,313,946 and 4,315,929) (isolated from Trewia nudlflora); C-18-N-demethyl (U.S. Pat. Nos. 4,362,663 and 4,322,348) (prepared by the demethylation of maytansinol by Streptomyces); and 4,5-deoxy (U.S. Pat. No. 4,371,533) (prepared by the titanium trichloride/LAH reduction of maytansinol).
[0336] Of particular use are DM1 (disclosed in U.S. Pat. No. 5,208,020, incorporated by reference) and DM4 (disclosed in U.S. Pat. No. 7,276,497, incorporated by reference). See also a number of additional maytansinoid derivatives and methods in 5,416,064, WO/01/24763, 7,303,749, 7,601,354, U.S. Ser. No. 12/631,508, WO02/098883, 6,441,163, 7,368,565, WO02/16368 and WO04/1033272, all of which are expressly incorporated by reference in their entirety.
[0337] ADCs containing maytansinoids, methods of making same, and their therapeutic use are disclosed, for example, in U.S. Pat. Nos. 5,208,020; 5,416,064; 6,441,163 and European Patent EP 0 425 235 B1, the disclosures of which are hereby expressly incorporated by reference. Liu et al., Proc. Natl. Acad. Sci. USA 93:8618-8623 (1996) described ADCs comprising a maytansinoid designated DM1 linked to the monoclonal antibody C242 directed against human colorectal cancer. The conjugate was found to be highly cytotoxic towards cultured colon cancer cells, and showed antitumor activity in an in vivo tumor growth assay.
[0338] Chari et al., Cancer Research 52:127-131 (1992) describe ADCs in which a maytansinoid was conjugated via a disulfide linker to the murine antibody A7 binding to an antigen on human colon cancer cell lines, or to another murine monoclonal antibody TA.1 that binds the HER-2/neu oncogene. The cytotoxicity of the TA.1-maytansonoid conjugate was tested in vitro on the human breast cancer cell line SK-BR-3, which expresses 3.times.105 HER-2 surface antigens per cell. The drug conjugate achieved a degree of cytotoxicity similar to the free maytansinoid drug, which could be increased by increasing the number of maytansinoid molecules per antibody molecule. The A7-maytansinoid conjugate showed low systemic cytotoxicity in mice.
Auristatins and Dolastatins
[0339] In some embodiments, the ADC comprises a multispecific antibody conjugated to dolastatins or dolostatin peptidic analogs and derivatives, the auristatins (U.S. Pat. Nos. 5,635,483; 5,780,588). Dolastatins and auristatins have been shown to interfere with microtubule dynamics, GTP hydrolysis, and nuclear and cellular division (Woyke et al (2001) Antimicrob. Agents and Chemother. 45(12):3580-3584) and have anticancer (U.S. Pat. No. 5,663,149) and antifungal activity (Pettit et al (1998) Antimicrob. Agents Chemother. 42:2961-2965). The dolastatin or auristatin drug moiety may be attached to the antibody through the N (amino) terminus or the C (carboxyl) terminus of the peptidic drug moiety (WO 02/088172).
[0340] Exemplary auristatin embodiments include the N-terminus linked monomethylauristatin drug moieties DE and DF, disclosed in "Senter et al, Proceedings of the American Association for Cancer Research, Volume 45, Abstract Number 623, presented Mar. 28, 2004 and described in United States Patent Publication No. 2005/0238648, the disclosure of which is expressly incorporated by reference in its entirety.
[0341] An exemplary auristatin embodiment is MMAE (see U.S. Pat. No. 6,884,869 expressly incorporated by reference in its entirety).
[0342] Another exemplary auristatin embodiment is MMAF (see US 2005/0238649, 5,767,237 and 6,124,431, expressly incorporated by reference in their entirety).
[0343] Additional exemplary embodiments comprising MMAE or MMAF and various linker components (described further herein) have the following structures and abbreviations (wherein Ab means antibody and p is 1 to about 8):
[0344] Typically, peptide-based drug moieties can be prepared by forming a peptide bond between two or more amino acids and/or peptide fragments. Such peptide bonds can be prepared, for example, according to the liquid phase synthesis method (see E. Schroder and K. Lubke, "The Peptides", volume 1, pp 76-136, 1965, Academic Press) that is well known in the field of peptide chemistry. The auristatin/dolastatin drug moieties may be prepared according to the methods of: U.S. Pat. Nos. 5,635,483; 5,780,588; Pettit et al (1989) J. Am. Chem. Soc. 111:5463-5465; Pettit et al (1998) Anti-Cancer Drug Design 13:243-277; Pettit, G. R., et al. Synthesis, 1996, 719-725; Pettit et al (1996) J. Chem. Soc. Perkin Trans. 1 5:859-863; and Doronina (2003) Nat Biotechnol 21(7):778-784.
Calicheamicin
[0345] In other embodiments, the ADC comprises an antibody of the invention conjugated to one or more calicheamicin molecules. For example, Mylotarg is the first commercial ADC drug and utilizes calicheamicin .gamma.1 as the payload (see U.S. Pat. No. 4,970,198, incorporated by reference in its entirety). Additional calicheamicin derivatives are described in U.S. Pat. Nos. 5,264,586, 5,384,412, 5,550,246, 5,739,116, 5,773,001, 5,767,285 and 5,877,296, all expressly incorporated by reference. The calicheamicin family of antibiotics are capable of producing double-stranded DNA breaks at sub-picomolar concentrations. For the preparation of conjugates of the calicheamicin family, see U.S. Pat. Nos. 5,712,374, 5,714,586, 5,739,116, 5,767,285, 5,770,701, 5,770,710, 5,773,001, 5,877,296 (all to American Cyanamid Company). Structural analogues of calicheamicin which may be used include, but are not limited to, .gamma.1I, .alpha.2I, .alpha.2I, N-acetyl-.gamma.1I, PSAG and .theta.I1 (Hinman et al., Cancer Research 53:3336-3342 (1993), Lode et al., Cancer Research 58:2925-2928 (1998) and the aforementioned U.S. patents to American Cyanamid). Another anti-tumor drug that the antibody can be conjugated is QFA which is an antifolate. Both calicheamicin and QFA have intracellular sites of action and do not readily cross the plasma membrane. Therefore, cellular uptake of these agents through antibody mediated internalization greatly enhances their cytotoxic effects.
Duocarmycins
[0346] CC-1065 (see 4,169,888, incorporated by reference) and duocarmycins are members of a family of antitumor antibiotics utilized in ADCs. These antibiotics appear to work through sequence-selectively alkylating DNA at the N3 of adenine in the minor groove, which initiates a cascade of events that result in apoptosis.
[0347] Important members of the duocarmycins include duocarmycin A (U.S. Pat. No. 4,923,990, incorporated by reference) and duocarmycin SA (U.S. Pat. No. 5,101,038, incorporated by reference), and a large number of analogues as described in U.S. Pat. Nos. 7,517,903, 7,691,962, 5,101,038; 5,641,780; 5,187,186; 5,070,092; 5,070,092; 5,641,780; 5,101,038; 5,084,468, 5,475,092, 5,585,499, 5,846,545, WO2007/089149, WO2009/017394A1, 5,703,080, 6,989,452, 7,087,600, 7,129,261, 7,498,302, and 7,507,420, all of which are expressly incorporated by reference.
Other Cytotoxic Agents
[0348] Other antitumor agents that can be conjugated to the antibodies of the invention include BCNU, streptozoicin, vincristine and 5-fluorouracil, the family of agents known collectively LL-E33288 complex described in U.S. Pat. Nos. 5,053,394, 5,770,710, as well as esperamicins (U.S. Pat. No. 5,877,296).
[0349] Enzymatically active toxins and fragments thereof which can be used include diphtheria A chain, nonbinding active fragments of diphtheria toxin, exotoxin A chain (from Pseudomonas aeruginosa), ricin A chain, abrin A chain, modeccin A chain, alpha-sarcin, Aleurites fordii proteins, dianthin proteins, Phytolaca americana proteins (PAPI, PAPII, and PAP-S), Momordica charantia inhibitor, curcin, crotin, Sapaonaria officinalis inhibitor, gelonin, mitogellin, restrictocin, phenomycin, enomycin and the tricothecenes. See, for example, WO 93/21232 published Oct. 28, 1993.
[0350] The present invention further contemplates an ADC formed between an antibody and a compound with nucleolytic activity (e.g., a ribonuclease or a DNA endonuclease such as a deoxyribonuclease; DNase).
[0351] For selective destruction of the tumor, the antibody may comprise a highly radioactive atom. A variety of radioactive isotopes are available for the production of radioconjugated antibodies. Examples include At211, I131, I125, Y90, Re186, Re188, Sm153, Bi212, P32, Pb212 and radioactive isotopes of Lu.
[0352] The radio- or other labels may be incorporated in the conjugate in known ways. For example, the peptide may be biosynthesized or may be synthesized by chemical amino acid synthesis using suitable amino acid precursors involving, for example, fluorine-19 in place of hydrogen. Labels such as Tc99m or I123, Re186, Re188 and In111 can be attached via a cysteine residue in the peptide. Yttrium-90 can be attached via a lysine residue. The IODOGEN method (Fraker et al (1978) Biochem. Biophys. Res. Commun. 80: 49-57 can be used to incorporate Iodine-123. "Monoclonal Antibodies in Immunoscintigraphy" (Chatal, CRC Press 1989) describes other methods in detail.
[0353] For compositions comprising a plurality of antibodies, the drug loading is represented by p, the average number of drug molecules per Antibody. Drug loading may range from 1 to 20 drugs (D) per Antibody. The average number of drugs per antibody in preparation of conjugation reactions may be characterized by conventional means such as mass spectroscopy, ELISA assay, and HPLC. The quantitative distribution of Antibody-Drug-Conjugates in terms of p may also be determined.
[0354] In some instances, separation, purification, and characterization of homogeneous Antibody-Drug-conjugates where p is a certain value from Antibody-Drug-Conjugates with other drug loadings may be achieved by means such as reverse phase HPLC or electrophoresis. In exemplary embodiments, p is 2, 3, 4, 5, 6, 7, or 8 or a fraction thereof.
[0355] The generation of Antibody-drug conjugate compounds can be accomplished by any technique known to the skilled artisan. Briefly, the Antibody-drug conjugate compounds can include a multispecific antibody as the Antibody unit, a drug, and optionally a linker that joins the drug and the binding agent.
[0356] A number of different reactions are available for covalent attachment of drugs and/or linkers to binding agents. This is can be accomplished by reaction of the amino acid residues of the binding agent, for example, antibody molecule, including the amine groups of lysine, the free carboxylic acid groups of glutamic and aspartic acid, the sulfhydryl groups of cysteine and the various moieties of the aromatic amino acids. A commonly used non-specific methods of covalent attachment is the carbodiimide reaction to link a carboxy (or amino) group of a compound to amino (or carboxy) groups of the antibody. Additionally, bifunctional agents such as dialdehydes or imidoesters have been used to link the amino group of a compound to amino groups of an antibody molecule.
[0357] Also available for attachment of drugs to binding agents is the Schiff base reaction. This method involves the periodate oxidation of a drug that contains glycol or hydroxy groups, thus forming an aldehyde which is then reacted with the binding agent. Attachment occurs via formation of a Schiff base with amino groups of the binding agent. Isothiocyanates can also be used as coupling agents for covalently attaching drugs to binding agents. Other techniques are known to the skilled artisan and within the scope of the present invention.
[0358] In some embodiments, an intermediate, which is the precursor of the linker, is reacted with the drug under appropriate conditions. In other embodiments, reactive groups are used on the drug and/or the intermediate. The product of the reaction between the drug and the intermediate, or the derivatized drug, is subsequently reacted with an multispecific antibody of the invention under appropriate conditions.
[0359] It will be understood that chemical modifications may also be made to the desired compound in order to make reactions of that compound more convenient for purposes of preparing conjugates of the invention. For example a functional group e.g. amine, hydroxyl, or sulfhydryl, may be appended to the drug at a position which has minimal or an acceptable effect on the activity or other properties of the drug
ADC Linker Units
[0360] Typically, the antibody-drug conjugate compounds comprise a Linker unit between the drug unit and the antibody unit. In some embodiments, the linker is cleavable under intracellular or extracellular conditions, such that cleavage of the linker releases the drug unit from the antibody in the appropriate environment. For example, solid tumors that secrete certain proteases may serve as the target of the cleavable linker; in other embodiments, it is the intracellular proteases that are utilized. In yet other embodiments, the linker unit is not cleavable and the drug is released, for example, by antibody degradation in lysosomes.
[0361] In some embodiments, the linker is cleavable by a cleaving agent that is present in the intracellular environment (for example, within a lysosome or endosome or caveolea). The linker can be, for example, a peptidyl linker that is cleaved by an intracellular peptidase or protease enzyme, including, but not limited to, a lysosomal or endosomal protease. In some embodiments, the peptidyl linker is at least two amino acids long or at least three amino acids long or more.
[0362] Cleaving agents can include, without limitation, cathepsins B and D and plasmin, all of which are known to hydrolyze dipeptide drug derivatives resulting in the release of active drug inside target cells (see, e.g., Dubowchik and Walker, 1999, Pharm. Therapeutics 83:67-123). Peptidyl linkers that are cleavable by enzymes that are present in CD38-expressing cells. For example, a peptidyl linker that is cleavable by the thiol-dependent protease cathepsin-B, which is highly expressed in cancerous tissue, can be used (e.g., a Phe-Leu or a Gly-Phe-Leu-Gly linker (SEQ ID NO: 287)). Other examples of such linkers are described, e.g., in U.S. Pat. No. 6,214,345, incorporated herein by reference in its entirety and for all purposes.
[0363] In some embodiments, the peptidyl linker cleavable by an intracellular protease is a Val-Cit linker or a Phe-Lys linker (see, e.g., U.S. Pat. No. 6,214,345, which describes the synthesis of doxorubicin with the val-cit linker).
[0364] In other embodiments, the cleavable linker is pH-sensitive, that is, sensitive to hydrolysis at certain pH values. Typically, the pH-sensitive linker hydrolyzable under acidic conditions. For example, an acid-labile linker that is hydrolyzable in the lysosome (for example, a hydrazone, semicarbazone, thiosemicarbazone, cis-aconitic amide, orthoester, acetal, ketal, or the like) may be used. (See, e.g., U.S. Pat. Nos. 5,122,368; 5,824,805; 5,622,929; Dubowchik and Walker, 1999, Pharm. Therapeutics 83:67-123; Neville et al., 1989, Biol. Chem. 264:14653-14661.) Such linkers are relatively stable under neutral pH conditions, such as those in the blood, but are unstable at below pH 5.5 or 5.0, the approximate pH of the lysosome. In certain embodiments, the hydrolyzable linker is a thioether linker (such as, e.g., a thioether attached to the therapeutic agent via an acylhydrazone bond (see, e.g., U.S. Pat. No. 5,622,929).
[0365] In yet other embodiments, the linker is cleavable under reducing conditions (for example, a disulfide linker). A variety of disulfide linkers are known in the art, including, for example, those that can be formed using SATA (N-succinimidyl-5-acetylthioacetate), SPDP (N-succinimidyl-3-(2-pyridyldithio)propionate), SPDB (N-succinimidyl-3-(2-pyridyldithio)butyrate) and SMPT (N-succinimidyl-oxycarbonyl-alpha-methyl-alpha-(2-pyridyl-dithio)toluene)- -, SPDB and SMPT. (See, e.g., Thorpe et al., 1987, Cancer Res. 47:5924-5931; Wawrzynczak et al., In Immunoconjugates: Antibody Conjugates in Radioimagery and Therapy of Cancer (C. W. Vogel ed., Oxford U. Press, 1987. See also U.S. Pat. No. 4,880,935.)
[0366] In other embodiments, the linker is a malonate linker (Johnson et al., 1995, Anticancer Res. 15:1387-93), a maleimidobenzoyl linker (Lau et al., 1995, Bioorg-Med-Chem. 3(10): 1299-1304), or a 3'-N-amide analog (Lau et al., 1995, Bioorg-Med-Chem. 3(10): 1305-12).
[0367] In yet other embodiments, the linker unit is not cleavable and the drug is released by antibody degradation. (See U.S. Publication No. 2005/0238649 incorporated by reference herein in its entirety and for all purposes).
[0368] In many embodiments, the linker is self-immolative. As used herein, the term "self-immolative Spacer" refers to a bifunctional chemical moiety that is capable of covalently linking together two spaced chemical moieties into a stable tripartite molecule. It will spontaneously separate from the second chemical moiety if its bond to the first moiety is cleaved. See for example, WO 2007059404A2, WO06110476A2, WO05112919A2, WO2010/062171, WO09/017394, WO07/089149, WO 07/018431, WO04/043493 and WO02/083180, which are directed to drug-cleavable substrate conjugates where the drug and cleavable substrate are optionally linked through a self-immolative linker and which are all expressly incorporated by reference.
[0369] Often the linker is not substantially sensitive to the extracellular environment. As used herein, "not substantially sensitive to the extracellular environment," in the context of a linker, means that no more than about 20%, 15%, 10%, 5%, 3%, or no more than about 1% of the linkers, in a sample of antibody-drug conjugate compound, are cleaved when the antibody-drug conjugate compound presents in an extracellular environment (for example, in plasma).
[0370] Whether a linker is not substantially sensitive to the extracellular environment can be determined, for example, by incubating with plasma the antibody-drug conjugate compound for a predetermined time period (for example, 2, 4, 8, 16, or 24 hours) and then quantitating the amount of free drug present in the plasma.
[0371] In other, non-mutually exclusive embodiments, the linker promotes cellular internalization. In certain embodiments, the linker promotes cellular internalization when conjugated to the therapeutic agent (that is, in the milieu of the linker-therapeutic agent moiety of the antibody-drug conjugate compound as described herein). In yet other embodiments, the linker promotes cellular internalization when conjugated to both the auristatin compound and the multispecific antibodies of the invention.
[0372] A variety of exemplary linkers that can be used with the present compositions and methods are described in WO 2004-010957, U.S. Publication No. 2006/0074008, U.S. Publication No. 20050238649, and U.S. Publication No. 2006/0024317 (each of which is incorporated by reference herein in its entirety and for all purposes).
Drug Loading
[0373] Drug loading is represented by p and is the average number of Drug moieties per antibody in a molecule. Drug loading ("p") may be 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 or more moieties (D) per antibody, although frequently the average number is a fraction or a decimal. Generally, drug loading of from 1 to 4 is frequently useful, and from 1 to 2 is also useful. ADCs of the invention include collections of antibodies conjugated with a range of drug moieties, from 1 to 20. The average number of drug moieties per antibody in preparations of ADC from conjugation reactions may be characterized by conventional means such as mass spectroscopy and, ELISA assay.
[0374] The quantitative distribution of ADC in terms of p may also be determined. In some instances, separation, purification, and characterization of homogeneous ADC where p is a certain value from ADC with other drug loadings may be achieved by means such as electrophoresis.
[0375] For some antibody-drug conjugates, p may be limited by the number of attachment sites on the antibody. For example, where the attachment is a cysteine thiol, as in the exemplary embodiments above, an antibody may have only one or several cysteine thiol groups, or may have only one or several sufficiently reactive thiol groups through which a linker may be attached. In certain embodiments, higher drug loading, e.g. p>5, may cause aggregation, insolubility, toxicity, or loss of cellular permeability of certain antibody-drug conjugates. In certain embodiments, the drug loading for an ADC of the invention ranges from 1 to about 8; from about 2 to about 6; from about 3 to about 5; from about 3 to about 4; from about 3.1 to about 3.9; from about 3.2 to about 3.8; from about 3.2 to about 3.7; from about 3.2 to about 3.6; from about 3.3 to about 3.8; or from about 3.3 to about 3.7. Indeed, it has been shown that for certain ADCs, the optimal ratio of drug moieties per antibody may be less than 8, and may be about 2 to about 5. See US 2005-0238649 A1 (herein incorporated by reference in its entirety).
[0376] In certain embodiments, fewer than the theoretical maximum of drug moieties are conjugated to an antibody during a conjugation reaction. An antibody may contain, for example, lysine residues that do not react with the drug-linker intermediate or linker reagent, as discussed below. Generally, antibodies do not contain many free and reactive cysteine thiol groups which may be linked to a drug moiety; indeed most cysteine thiol residues in antibodies exist as disulfide bridges. In certain embodiments, an antibody may be reduced with a reducing agent such as dithiothreitol (DTT) or tricarbonylethylphosphine (TCEP), under partial or total reducing conditions, to generate reactive cysteine thiol groups. In certain embodiments, an antibody is subjected to denaturing conditions to reveal reactive nucleophilic groups such as lysine or cysteine.
[0377] The loading (drug/antibody ratio) of an ADC may be controlled in different ways, e.g., by: (i) limiting the molar excess of drug-linker intermediate or linker reagent relative to antibody, (ii) limiting the conjugation reaction time or temperature, (iii) partial or limiting reductive conditions for cysteine thiol modification, (iv) engineering by recombinant techniques the amino acid sequence of the antibody such that the number and position of cysteine residues is modified for control of the number and/or position of linker-drug attachments (such as thioMab or thioFab prepared as disclosed herein and in WO2006/034488 (herein incorporated by reference in its entirety)).
[0378] It is to be understood that where more than one nucleophilic group reacts with a drug-linker intermediate or linker reagent followed by drug moiety reagent, then the resulting product is a mixture of ADC compounds with a distribution of one or more drug moieties attached to an antibody. The average number of drugs per antibody may be calculated from the mixture by a dual ELISA antibody assay, which is specific for antibody and specific for the drug. Individual ADC molecules may be identified in the mixture by mass spectroscopy and separated by HPLC, e.g. hydrophobic interaction chromatography.
[0379] In some embodiments, a homogeneous ADC with a single loading value may be isolated from the conjugation mixture by electrophoresis or chromatography.
Methods of Determining Cytotoxic Effect of ADCs
[0380] Methods of determining whether a Drug or Antibody-Drug conjugate exerts a cytostatic and/or cytotoxic effect on a cell are known. Generally, the cytotoxic or cytostatic activity of an Antibody Drug conjugate can be measured by: exposing mammalian cells expressing a target protein of the Antibody Drug conjugate in a cell culture medium; culturing the cells for a period from about 6 hours to about 5 days; and measuring cell viability. Cell-based in vitro assays can be used to measure viability (proliferation), cytotoxicity, and induction of apoptosis (caspase activation) of the Antibody Drug conjugate.
[0381] For determining whether an Antibody Drug conjugate exerts a cytostatic effect, a thymidine incorporation assay may be used. For example, cancer cells expressing a target antigen at a density of 5,000 cells/well of a 96-well plated can be cultured for a 72-hour period and exposed to 0.5 .mu.Ci of 3H-thymidine during the final 8 hours of the 72-hour period. The incorporation of 3H-thymidine into cells of the culture is measured in the presence and absence of the Antibody Drug conjugate.
[0382] For determining cytotoxicity, necrosis or apoptosis (programmed cell death) can be measured. Necrosis is typically accompanied by increased permeability of the plasma membrane; swelling of the cell, and rupture of the plasma membrane. Apoptosis is typically characterized by membrane blebbing, condensation of cytoplasm, and the activation of endogenous endonucleases. Determination of any of these effects on cancer cells indicates that an Antibody Drug conjugate is useful in the treatment of cancers.
[0383] Cell viability can be measured by determining in a cell the uptake of a dye such as neutral red, trypan blue, or ALAMAR.TM. blue (see, e.g., Page et al., 1993, Intl. J. Oncology 3:473-476). In such an assay, the cells are incubated in media containing the dye, the cells are washed, and the remaining dye, reflecting cellular uptake of the dye, is measured spectrophotometrically. The protein-binding dye sulforhodamine B (SRB) can also be used to measure cytotoxicity (Skehan et al., 1990, J. Natl. Cancer Inst. 82:1107-12).
[0384] Alternatively, a tetrazolium salt, such as MTT, is used in a quantitative colorimetric assay for mammalian cell survival and proliferation by detecting living, but not dead, cells (see, e.g., Mosmann, 1983, J. Immunol. Methods 65:55-63).
[0385] Apoptosis can be quantitated by measuring, for example, DNA fragmentation. Commercial photometric methods for the quantitative in vitro determination of DNA fragmentation are available. Examples of such assays, including TUNEL (which detects incorporation of labeled nucleotides in fragmented DNA) and ELISA-based assays, are described in Biochemica, 1999, no. 2, pp. 34-37 (Roche Molecular Biochemicals).
[0386] Apoptosis can also be determined by measuring morphological changes in a cell. For example, as with necrosis, loss of plasma membrane integrity can be determined by measuring uptake of certain dyes (e.g., a fluorescent dye such as, for example, acridine orange or ethidium bromide). A method for measuring apoptotic cell number has been described by Duke and Cohen, Current Protocols in Immunology (Coligan et al. eds., 1992, pp. 3.17.1-3.17.16). Cells also can be labeled with a DNA dye (e.g., acridine orange, ethidium bromide, or propidium iodide) and the cells observed for chromatin condensation and margination along the inner nuclear membrane. Other morphological changes that can be measured to determine apoptosis include, e.g., cytoplasmic condensation, increased membrane blebbing, and cellular shrinkage.
[0387] The presence of apoptotic cells can be measured in both the attached and "floating" compartments of the cultures. For example, both compartments can be collected by removing the supernatant, trypsinizing the attached cells, combining the preparations following a centrifugation wash step (e.g., 10 minutes at 2000 rpm), and detecting apoptosis (e.g., by measuring DNA fragmentation). (See, e.g., Piazza et al., 1995, Cancer Research 55:3110-16).
[0388] In vivo, the effect of a therapeutic composition of the multispecific antibody of the invention can be evaluated in a suitable animal model. For example, xenogenic cancer models can be used, wherein cancer explants or passaged xenograft tissues are introduced into immune compromised animals, such as nude or SCID mice (Klein et al., 1997, Nature Medicine 3: 402-408). Efficacy can be measured using assays that measure inhibition of tumor formation, tumor regression or metastasis, and the like.
[0389] The therapeutic compositions used in the practice of the foregoing methods can be formulated into pharmaceutical compositions comprising a carrier suitable for the desired delivery method. Suitable carriers include any material that when combined with the therapeutic composition retains the anti-tumor function of the therapeutic composition and is generally non-reactive with the patient's immune system. Examples include, but are not limited to, any of a number of standard pharmaceutical carriers such as sterile phosphate buffered saline solutions, bacteriostatic water, and the like (see, generally, Remington's Pharmaceutical Sciences 16th Edition, A. Osal., Ed., 1980).
Antibody Compositions for In Vivo Administration
[0390] Formulations of the antibodies used in accordance with the present invention are prepared for storage by mixing an antibody having the desired degree of purity with optional pharmaceutically acceptable carriers, excipients or stabilizers (Remington's Pharmaceutical Sciences 16th edition, Osol, A. Ed. [1980]), in the form of lyophilized formulations or aqueous solutions. Acceptable carriers, excipients, or stabilizers are nontoxic to recipients at the dosages and concentrations employed, and include buffers such as phosphate, citrate, and other organic acids; antioxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, histidine, arginine, or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugars such as sucrose, mannitol, trehalose or sorbitol; salt-forming counter-ions such as sodium; metal complexes (e.g. Zn-protein complexes); and/or non-ionic surfactants such as TWEEN.TM., PLURONICS.TM. or polyethylene glycol (PEG).
[0391] The formulation herein may also contain more than one active compound as necessary for the particular indication being treated, preferably those with complementary activities that do not adversely affect each other. For example, it may be desirable to provide antibodies with other specificities. Alternatively, or in addition, the composition may comprise a cytotoxic agent, cytokine, growth inhibitory agent and/or small molecule antagonist. Such molecules are suitably present in combination in amounts that are effective for the purpose intended.
[0392] The active ingredients may also be entrapped in microcapsules prepared, for example, by coacervation techniques or by interfacial polymerization, for example, hydroxymethylcellulose or gelatin-microcapsules and poly-(methylmethacylate) microcapsules, respectively, in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules) or in macroemulsions. Such techniques are disclosed in Remington's Pharmaceutical Sciences 16th edition, Osol, A. Ed. (1980).
[0393] The formulations to be used for in vivo administration should be sterile, or nearly so. This is readily accomplished by filtration through sterile filtration membranes.
[0394] Sustained-release preparations may be prepared. Suitable examples of sustained-release preparations include semipermeable matrices of solid hydrophobic polymers containing the antibody, which matrices are in the form of shaped articles, e.g. films, or microcapsules. Examples of sustained-release matrices include polyesters, hydrogels (for example, poly(2-hydroxyethyl-methacrylate), or poly(vinylalcohol)), polylactides (U.S. Pat. No. 3,773,919), copolymers of L-glutamic acid and .gamma. ethyl-L-glutamate, non-degradable ethylene-vinyl acetate, degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOT.TM. (injectable microspheres composed of lactic acid-glycolic acid copolymer and leuprolide acetate), and poly-D-(-)-3-hydroxybutyric acid. While polymers such as ethylene-vinyl acetate and lactic acid-glycolic acid enable release of molecules for over 100 days, certain hydrogels release proteins for shorter time periods.
[0395] When encapsulated antibodies remain in the body for a long time, they may denature or aggregate as a result of exposure to moisture at 37.degree. C., resulting in a loss of biological activity and possible changes in immunogenicity. Rational strategies can be devised for stabilization depending on the mechanism involved. For example, if the aggregation mechanism is discovered to be intermolecular S--S bond formation through thio-disulfide interchange, stabilization may be achieved by modifying sulfhydryl residues, lyophilizing from acidic solutions, controlling moisture content, using appropriate additives, and developing specific polymer matrix compositions.
Administrative Modalities
[0396] The antibodies and chemotherapeutic agents of the invention are administered to a subject, in accord with known methods, such as intravenous administration as a bolus or by continuous infusion over a period of time, by intramuscular, intraperitoneal, intracerobrospinal, subcutaneous, intra-articular, intrasynovial, intrathecal, oral, topical, or inhalation routes. Intravenous or subcutaneous administration of the antibody is preferred.
Treatment Modalities
[0397] In the methods of the invention, therapy is used to provide a positive therapeutic response with respect to a disease or condition. By "positive therapeutic response" is intended an improvement in the disease or condition, and/or an improvement in the symptoms associated with the disease or condition. For example, a positive therapeutic response would refer to one or more of the following improvements in the disease: (1) a reduction in the number of neoplastic cells; (2) an increase in neoplastic cell death; (3) inhibition of neoplastic cell survival; (5) inhibition (i.e., slowing to some extent, preferably halting) of tumor growth; (6) an increased patient survival rate; and (7) some relief from one or more symptoms associated with the disease or condition.
[0398] Positive therapeutic responses in any given disease or condition can be determined by standardized response criteria specific to that disease or condition. Tumor response can be assessed for changes in tumor morphology (i.e., overall tumor burden, tumor size, and the like) using screening techniques such as magnetic resonance imaging (MRI) scan, x-radiographic imaging, computed tomographic (CT) scan, bone scan imaging, endoscopy, and tumor biopsy sampling including bone marrow aspiration (BMA) and counting of tumor cells in the circulation.
[0399] In addition to these positive therapeutic responses, the subject undergoing therapy may experience the beneficial effect of an improvement in the symptoms associated with the disease.
[0400] Thus for B cell tumors, the subject may experience a decrease in the so-called B symptoms, i.e., night sweats, fever, weight loss, and/or urticaria. For pre-malignant conditions, therapy with an multispecific therapeutic agent may block and/or prolong the time before development of a related malignant condition, for example, development of multiple myeloma in subjects suffering from monoclonal gammopathy of undetermined significance (MGUS).
[0401] An improvement in the disease may be characterized as a complete response. By "complete response" is intended an absence of clinically detectable disease with normalization of any previously abnormal radiographic studies, bone marrow, and cerebrospinal fluid (CSF) or abnormal monoclonal protein in the case of myeloma.
[0402] Such a response may persist for at least 4 to 8 weeks, or sometimes 6 to 8 weeks, following treatment according to the methods of the invention. Alternatively, an improvement in the disease may be categorized as being a partial response. By "partial response" is intended at least about a 50% decrease in all measurable tumor burden (i.e., the number of malignant cells present in the subject, or the measured bulk of tumor masses or the quantity of abnormal monoclonal protein) in the absence of new lesions, which may persist for 4 to 8 weeks, or 6 to 8 weeks.
[0403] Treatment according to the present invention includes a "therapeutically effective amount" of the medicaments used. A "therapeutically effective amount" refers to an amount effective, at dosages and for periods of time necessary, to achieve a desired therapeutic result.
[0404] A therapeutically effective amount may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the medicaments to elicit a desired response in the individual. A therapeutically effective amount is also one in which any toxic or detrimental effects of the antibody or antibody portion are outweighed by the therapeutically beneficial effects.
[0405] A "therapeutically effective amount" for tumor therapy may also be measured by its ability to stabilize the progression of disease. The ability of a compound to inhibit cancer may be evaluated in an animal model system predictive of efficacy in human tumors.
[0406] Alternatively, this property of a composition may be evaluated by examining the ability of the compound to inhibit cell growth or to induce apoptosis by in vitro assays known to the skilled practitioner. A therapeutically effective amount of a therapeutic compound may decrease tumor size, or otherwise ameliorate symptoms in a subject. One of ordinary skill in the art would be able to determine such amounts based on such factors as the subject's size, the severity of the subject's symptoms, and the particular composition or route of administration selected.
[0407] Dosage regimens are adjusted to provide the optimum desired response (e.g., a therapeutic response). For example, a single bolus may be administered, several divided doses may be administered over time or the dose may be proportionally reduced or increased as indicated by the exigencies of the therapeutic situation. Parenteral compositions may be formulated in dosage unit form for ease of administration and uniformity of dosage. Dosage unit form as used herein refers to physically discrete units suited as unitary dosages for the subjects to be treated; each unit contains a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier.
[0408] The specification for the dosage unit forms of the present invention are dictated by and directly dependent on (a) the unique characteristics of the active compound and the particular therapeutic effect to be achieved, and (b) the limitations inherent in the art of compounding such an active compound for the treatment of sensitivity in individuals.
[0409] The efficient dosages and the dosage regimens for the multispecific antibodies used in the present invention depend on the disease or condition to be treated and may be determined by the persons skilled in the art.
[0410] An exemplary, non-limiting range for a therapeutically effective amount of an multispecific antibody used in the present invention is about 0.1-100 mg/kg, such as about 0.1-50 mg/kg, for example about 0.1-20 mg/kg, such as about 0.1-10 mg/kg, for instance about 0.5, about such as 0.3, about 1, or about 3 mg/kg. In another embodiment, he antibody is administered in a dose of 1 mg/kg or more, such as a dose of from 1 to 20 mg/kg, e.g. a dose of from 5 to 20 mg/kg, e.g. a dose of 8 mg/kg.
[0411] A medical professional having ordinary skill in the art may readily determine and prescribe the effective amount of the pharmaceutical composition required. For example, a physician or a veterinarian could start doses of the medicament employed in the pharmaceutical composition at levels lower than that required in order to achieve the desired therapeutic effect and gradually increase the dosage until the desired effect is achieved.
[0412] In one embodiment, the multispecific antibody is administered by infusion in a weekly dosage of from 10 to 500 mg/kg such as of from 200 to 400 mg/kg Such administration may be repeated, e.g., 1 to 8 times, such as 3 to 5 times. The administration may be performed by continuous infusion over a period of from 2 to 24 hours, such as of from 2 to 12 hours.
[0413] In one embodiment, the multispecific antibody is administered by slow continuous infusion over a long period, such as more than 24 hours, if required to reduce side effects including toxicity.
[0414] In one embodiment the multispecific antibody is administered in a weekly dosage of from 250 mg to 2000 mg, such as for example 300 mg, 500 mg, 700 mg, 1000 mg, 1500 mg or 2000 mg, for up to 8 times, such as from 4 to 6 times. The administration may be performed by continuous infusion over a period of from 2 to 24 hours, such as of from 2 to 12 hours. Such regimen may be repeated one or more times as necessary, for example, after 6 months or 12 months. The dosage may be determined or adjusted by measuring the amount of compound of the present invention in the blood upon administration by for instance taking out a biological sample and using anti-idiotypic antibodies which target the antigen binding region of the multispecific antibody.
[0415] In a further embodiment, the multispecific antibody is administered once weekly for 2 to 12 weeks, such as for 3 to 10 weeks, such as for 4 to 8 weeks.
[0416] In one embodiment, the multispecific antibody is administered by maintenance therapy, such as, e.g., once a week for a period of 6 months or more.
[0417] In one embodiment, the multispecific antibody is administered by a regimen including one infusion of an multispecific antibody followed by an infusion of an multispecific antibody conjugated to a radioisotope. The regimen may be repeated, e.g., 7 to 9 days later.
[0418] As non-limiting examples, treatment according to the present invention may be provided as a daily dosage of an antibody in an amount of about 0.1-100 mg/kg, such as 0.5, 0.9, 1.0, 1.1, 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 40, 45, 50, 60, 70, 80, 90 or 100 mg/kg, per day, on at least one of day 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, or 40, or alternatively, at least one of week 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19 or 20 after initiation of treatment, or any combination thereof, using single or divided doses of every 24, 12, 8, 6, 4, or 2 hours, or any combination thereof.
[0419] In some embodiments the multispecific antibody molecule thereof is used in combination with one or more additional therapeutic agents, e.g. a chemotherapeutic agent. Non-limiting examples of DNA damaging chemotherapeutic agents include topoisomerase I inhibitors (e.g., irinotecan, topotecan, camptothecin and analogs or metabolites thereof, and doxorubicin); topoisomerase II inhibitors (e.g., etoposide, teniposide, and daunorubicin); alkylating agents (e.g., melphalan, chlorambucil, busulfan, thiotepa, ifosfamide, carmustine, lomustine, semustine, streptozocin, decarbazine, methotrexate, mitomycin C, and cyclophosphamide); DNA intercalators (e.g., cisplatin, oxaliplatin, and carboplatin); DNA intercalators and free radical generators such as bleomycin; and nucleoside mimetics (e.g., 5-fluorouracil, capecitibine, gemcitabine, fludarabine, cytarabine, mercaptopurine, thioguanine, pentostatin, and hydroxyurea).
[0420] Chemotherapeutic agents that disrupt cell replication include: paclitaxel, docetaxel, and related analogs; vincristine, vinblastin, and related analogs; thalidomide, lenalidomide, and related analogs (e.g., CC-5013 and CC-4047); protein tyrosine kinase inhibitors (e.g., imatinib mesylate and gefitinib); proteasome inhibitors (e.g., bortezomib); NF-KB inhibitors, including inhibitors of IKB kinase; antibodies which bind to proteins overexpressed in cancers and thereby downregulate cell replication (e.g., trastuzumab, rituximab, cetuximab, and bevacizumab); and other inhibitors of proteins or enzymes known to be upregulated, overexpressed or activated in cancers, the inhibition of which downregulates cell replication.
[0421] In some embodiments, the antibodies of the invention can be used prior to, concurrent with, or after treatment with Velcade.RTM. (bortezomib).
[0422] All cited references are herein expressly incorporated by reference in their entirety.
[0423] Whereas particular embodiments of the invention have been described above for purposes of illustration, it will be appreciated by those skilled in the art that numerous variations of the details may be made without departing from the invention as described in the appended claims.
EXAMPLES
[0424] Examples are provided below are for illustrative purposes only. These examples are not meant to constrain any embodiment disclosed herein to any particular application or theory of operation.
Example 1. Constructing Anti-CD4.times.Anti-CD25 Bispecific Antibodies
[0425] A concept for suppressing Treg cells with anti-CD4.times.anti-CD25 bispecifics while not affecting other T cell types is shown schematically in FIG. 1.
[0426] The ability of various anti-CD25 heavy chains to pair with anti-CD4 light chains and anti-CD4 heavy chains to pair with anti-CD25 light chains in order to create a "common light-chain" anti-CD4.times.CD25 bispecific antibody was evaluated. Desired gene segments were synthesized by Blue Heron Biotechnologies (Bothell, Wash.) from synthetic oligonucleotides and PCR products by automated gene synthesis. Antibody constructs in the pTT5 vector were expressed in 293E cells and purified by standard Protein A, followed by IEX chromatography in order to isolate the desired heterodimeric bispecific. Biacore was used to examine binding of the various pairs to both CD4 and CD25 and the results tabulated (FIG. 2). 100 nM of each variant was immobilized on a Protein A chip for 1 min, followed by flowing antigen (CD4 or CD25) at 100 nM for 2 min dissociation. As can be seen from the data, the HuMax-TAC anti-CD25 heavy chain has the unique ability to pair with the anti-CD4 lights chains of OKT4A and zanolimumab, with the HuMax-TAC/OKT4A pair showing the strongest binding.
[0427] "Common light-chain" anti-CD4.times.CD25 bispecific antibodies were constructed by co-transfecting (in 293E cells) DNA encoding the heavy chain of anti-CD4 antibody OKT4A_H0L0 with the heavy chain of anti-CD25 antibody HuMAX-Tac and the light chain of anti-CD4 antibody OKTH0L0. These bispecific antibodies express well and have biophysical properties equivalent to normal monovalent IgG antibodies. Utilizing a heterodimeric Fc format, dual scFv-Fc anti-CD4.times.CD25 bispecific antibodies were also constructed and expressed. A third format, with a normal Fab-Fc on one side and scFv-Fc on the other side was also constructed. Control "one-armed" antibodies were also constructed to evaluate the effects of monovalent antigen binding (e.g. Anti-CD4.times.empty-Fc or Anti-CD25 by empty-Fc). For all three formats, variants with different Fc regions were produced: IgG1, high ADCC (S239D/I332E), and Fc knockout (G236R/L328R or PVA_/S267K). Bispecific formats are shown schematically in FIG. 3. These bispecific antibody variants were evaluated for the ability to simultaneously bind both CD4 and CD25 on Biacore. 100 nM of each variant was bound to a CD25 surface, followed by flowing of 100 nM of CD4 over the chip surface. An example of the data is shown in FIG. 4.
[0428] Although CD4 and CD25 antigens were initially targeted for suppressing Tregs, other combinations of Treg markers may also be used in accordance with the methods described herein, including combinations listed in FIG. 32. Anti-CTLA4.times.Anti-CD25, Anti-PD-1.times.Anti-CD25, and Anti-CCR4.times.Anti-CD25 bispecific antibodies were also constructed. Any of the formats shown in FIG. 3 can be made to bind to any combinations of the targets listed in FIG. 32.
Example 2. Suppression of Regulatory T Cells with Anti-CD4.times.Anti-CD25 Bispecific Antibodies
[0429] Treg cells were generated in vitro using the following method. CD4.sup.+ enriched T cells (isolated using the EasySep.TM. Human CD4.sup.+ T Cell Enrichment Kit from Stemcell Technologies) from PBMC were incubated with anti-CD3/anti-CD28 beads (20 .mu.l beads in 100 .mu.l volume, or 4:1 beads to cell ratio using Dynabeads.RTM. Regulatory CD4.sup.+CD25.sup.+ T Cell Kit) with 500 U/mL of IL2 in the presence of 0.1 .mu.g/ml rapamycin for a week. Cells were replaced with new culture with anti-CD3 (OKT3, eBiosciences) plate bound at 0.5 .mu.g/mL and soluble 0.5 .mu.g/mL anti-CD28 (clone 28.2, eBiosciences) with 100 U/mL of IL2 and 0.1 .mu.g/mL rapamycin.
[0430] Proliferation of Treg cells was assayed using CFSE cell proliferation assay or Alamar Blue cell viability assays in the presence of bispecific or control antibodies with 15 U/mL IL2. Results are shown in FIG. 5, FIG. 7, FIG. 12, FIG. 13, and FIG. 15. Anti-CD4.times.Anti-CD25 bispecifics 11209 and 12143 (IgG1 and FcKO Fc, respectively) were able to suppress proliferation of Treg cells more strongly compared to anti-CD25 (6368) antibody alone, and no effect was seen with anti-CD4 mAb (10966) alone. These results demonstrate the increased suppression of Treg cells with avid targeting using anti-CD4.times.anti-CD25 bispecific antibodies. The Fv of OKT4A was also humanized (OKT4A_H1L1) using the method of Lazar et al., 2007, Molecular Immunology, 44:1986-1998, hereby incorporated by reference in its entirety for all purposes and in particular for all teachings related to OKT4A, and this Fv was tested (FIG. 12 and FIG. 15), along with bispecifics containing the alternative anti-CD4 Fv Ibalizumab (FIG. 13). The epitopes of OKT4A and Ibalizumab differ, with binding of OKT4A to CD4 expected to block MHC II binding to CD4 whereas the epitope of Ibalizumab is away from the MHC II binding site on CD4 and its binding is not expected to be blocking. The precursor murine Fv of Ibalizumab (5A8) was also humanized to generate 5A8_H1L1. Bispecifics with the anti-CD4 Fv 5A8_H1L1 were also generated.
Example 3. Effect of Altering Antigen Binding Affinity of Anti-CD4.times.Anti-CD25 Bispecific Antibodies
[0431] Variant bispecific antibodies and one-armed antibody controls were constructed in which the CD25 binding affinity was altered. The Anti-TAC_H1.8L1 Fv (in 13531 and 13532) has 6-fold increased affinity for CD25. Conversely, the Anti-TAC_H1L1.12 Fv (in 13533 and 13534) has 17-fold lower CD25 affinity. These variants were assessed in cell proliferation assays (FIG. 15). A clear correlation between CD25 affinity and potency can be seen. 13531 with increased CD25 affinity has the strongest inhibition of cell proliferation, while lower affinity resulted in a reduced effect on cell proliferation. A similar pattern is also expected if CD4 affinity was altered. However, increasing the affinity for CD4 may result in even greater potency on Tregs due to its lower expression level compared to CD25. This can be shown by lower binding of anti-CD4 mAbs on Tregs compared to anti-CD25 mAbs (shown in FIG. 8).
Example 4. Direct Binding of Anti-CD4.times.Anti-CD25 Bispecific Antibodies to Tregs and Naive CD4+ T Cells
[0432] Binding of Anti-CD4.times.anti-CD25 bispecifics and control antibodies was measured to Tregs, naive CD4+ T cells, and activated CD4+ and CD8+ T cells. 200k Tregs were plated with antibodies at 4 .mu.g/mL (4.times. serial dilutions, 8 total dilutions). Cells and Abs were incubated at 45 min on ice and then washed and stained with secondary Ab anti-human F(ab)'2 Fcgamma specific PE labeled at 1 .mu.g/mL. Cells were washed and fixed with 1% PFA overnight and data acquired on a FACS Canto II. Results are shown in FIGS. 8-11. Bispecifics and anti-CD25 mAbs bound more strongly to Tregs compared to anti-CD4 mAbs, indicating that there may be a higher density of CD25 on Tregs compared to CD4. A clear avidity effect was seen with the bispecifics. Direct binding to purified naive human CD4+ T cells, activated CD4+, and activated CD8+ T cells was also assessed in a similar manner. Results for these binding assays are shown in FIG. 14 and FIGS. 16-22.
Example 5. Effect of Anti-CD4.times.Anti-CD25 Bispecific Antibodies on Cell Proliferation of CD4+CD25+(Helper T Cells) and CD8+CD25 (Cytotoxic T Cells)
[0433] For suppression of Tregs, it is desirable to suppress Treg cells and have little or no impact on other T cell types. To assess the impact of Anti-CD4.times.Anti-CD25 bispecific antibodies on other T cell types, CFSE labeled PBMC were incubated with 12.5 ng/mL anti-CD3 and 15 U/mL IL2 for 4 days in the presence of bispecific or control antibodies. Results are shown in FIG. 6 and FIG. 7. In this format, a clear dependence on Fc.gamma.R binding ability is seen. 11209 - Anti-CD4.times.Anti-CD25 IgG1 causes suppression of T-helper cells, while 12143 - Anti-CD4.times.Anti-CD25 FcKO has a much reduced level of suppression. Anti-CD25 (6368) antibody alone is also able to cause suppression of this T cell type, while anti-CD4 mAb (10966) alone shows limited activity (both are IgG1 Fc).
[0434] For cytotoxic T cells (CD8+CD25+), suppression was only seen with 11209 - Anti-CD4.times.CD25 IgG1 and Anti-CD25 (6368). No suppression was seen with 12143 - Anti-CD4.times.Anti-CD25 FcKO or anti-CD4 mAb (10966). Again, a clear dependence on Fc.gamma.R binding ability of the bispecifics is seen.
Example 6. Constructing Bispecific Anti-CD4.times.IL2 Fc-Fusions
[0435] The concept of inducing Treg cells with anti-CD4.times.IL2 Fc-fusions while not affecting other T cell types is shown schematically in FIG. 24.
[0436] Anti-CD4.times.IL2 Fc-fusions were designed and constructed from the sequences of human IL2 and the anti-CD4 antibody OKT4A (FIG. 25). Constructs in the pTT5 vector were expressed in 293E cells and purified using Protein A and IEX chromatography to isolate the desired heterodimeric Fc-fusion. SEC and SDS-PAGE analysis of the Protein A purified material as well as the final IEX purified material are shown in FIG. 26. Anti-CD4.times.IL2 Fc-fusions were homogeneous and obtained in high purity. All Fc-fusions were expressed with a Fc.gamma.R knocked out binding Fc region (IgG1 G236R/L328R or IgG1 PVA_/S267K). Anti-CD4.times.IL2 Fc-fusions using the Anti-CD4 mAbs Ibalizumab and 5A8_H1L1 were also constructed, as were Anti-CCR4.times.IL2, Anti-CTLA4.times.IL2, and Anti-PD1.times.IL2 antibody Fc-fusions. Bispecific IL2 Fc-fusions with antibodies against any of the Treg markers listed in Table 1 could also be constructed. Alternatively, similar variants may possess superior selectivity for Treg agonism versus other T cell types.
Example 7. Induction of Regulatory T Cells (Tregs) by Anti-CD4.times.IL2 Fc-Fusions
[0437] Treg cells were generated in vitro using the following method. CD4.sup.+ enriched T cells (isolated using the EasySep.TM. Human CD4+ T Cell Enrichment Kit from Stemcell Technologies) from PBMC were incubated with anti-CD3/anti-CD28 beads (20 .mu.l beads in 100 .mu.l volume, or 4:1 beads to cell ratio using Dynabeads.RTM. Regulatory CD4.sup.+CD25.sup.+ T Cell Kit) with 500 U/mL of IL2 in the presence of 0.1 .mu.g/mL rapamycin for a week. Cells were replaced with new culture with anti-CD3 (OKT3, eBiosciences) plate bound at 0.5 .mu.g/mL and soluble 0.5 .mu.g/mL anti-CD28 (clone 28.2, eBiosciences) with 100 U/mL of IL2 and 0.1 .mu.g/mL rapamycin.
[0438] Induction of Treg cells was assayed using the alamar blue cell viability assay in the presence of anti-CD4.times.IL2 Fc-fusions or control antibodies. Results are shown in FIG. 27. Increased viability of Treg cells was seen for the anti-CD4.times.IL2 Fc-fusions as well as IL2-only Fc-fusions. Anti-CD25 and anti-CD4 control antibodies showed no induction. The IL2-only Fc fusion (13044) served as a proxy for the reduced level of induction expected for cytotoxic T cells (CD8.sup.+CD25.sup.+).
Example 8. Suppression or Induction of Regulatory T Cells Using Anti-CD4.times.1L2 Fc-Fusions Engineered for Reduced or Increased IL2-Receptor Signaling
[0439] IL2 are engineered in order to alter the ratio of induction for Treg cells versus other types of IL2 receptor expressing cells (i.e. NK cells). For example, a dominant-negative IL2 Fc-fusion is created by engineering IL2 to have reduced ability to bind to IL2R.beta., IL2R.gamma., and or IL2R.alpha. in order to ablate IL2 receptor signaling. When coupled with an anti-CD4 antibody (or other Treg surface marker antibody), this results in an anti-CD4.times.IL2 Fc-fusion capable of suppressing Treg cells through targeted binding to CD4 and CD25, but without the ability to induce Treg proliferation. This Fc-fusion blocks endogenous IL2 from binding to receptor.
[0440] Likewise, more potent Anti-CD4.times.IL2 Fc-fusions inducers are engineered by increasing the affinity of IL2 for IL2R.alpha.. Exemplary variants of IL2 of use in the present invention are listed in FIG. 23.
Example 9. Suppression and Induction of Cytotoxic T Cells with Anti-CD8.times.Anti-CD25 Bispecific Antibodies or Anti-CD8.times.IL2 Fc-Fusions
[0441] Anti-CD8 antibodies including MCD8, 3B5, SK1, OKT-8, 51.1 or DK-25 are combined with an anti-CD25 antibody to make a bispecific antibody for suppression of cytotoxic T cells. Alternatively, in order to induce cytotoxic T cells, an Fc-fusion consisting of IL2 combined with an anti-CD8 antibody are used. Avidity may also drive IL-2 activation by binding the low affinity (beta/gamma) IL-2 receptor, circumventing the requirement for CD25, thus also being effective on non-activated CD8. Methods for suppression and induction are shown schematically in FIG. 28 and FIG. 29. These approaches are useful for treating cancer or autoimmune diseases, respectively.
Example 10. Evaluation of Treg Suppressor and Inducer Variants in a GVHD Mouse Model
[0442] Variants of the invention are evaluated in a Graft-versus-Host Disease model conducted in NSG SCID mice such as those conducted in Mutis et al., Clin Cancer Res (12), 2006. When NSG SCID mice are injected with human PBMCs they develop an autoimmune response against the human PBMCs, and this has been shown to be Treg dependent. NSG SCID mice injected with human PBMCs and then treated with a Treg suppression bispecific antibody such as 12143, 12462, 13025, or 13529 will have an exacerbation of disease and will die more quickly compared to untreated mice. Conversely, mice can be given a Treg inducing bispecific IL2-Fc fusion such as 13027 and they have a less severe disease and live longer than untreated mice.
Example 11. Evaluation of Treg Suppressor Mouse Surrogate Variants in Syngeneic Mouse Tumor Models
[0443] Mouse surrogate bispecific antibodies and IL2-Fc fusions can be made and studied in syngeneic mouse tumor models. The Treg suppressor bispecific Anti-mCD4.times.Anti-mCD25 can be made using the Anti-mouse CD4 antibody GK1.5 and the Anti-mouse CD25 antibody PC61. Tumors can be introduced in normal mice and then the mice treated with surrogate bispecific antibody. Suppression of the mouse Tregs should allow the mouse cytotoxic T cells to fight the tumor, resulting in a decreased tumor volume.
Example 12. Evaluation of Treg Inducer Mouse Surrogate Variants in an EAE Mouse Model
[0444] Mouse surrogate Treg inducer IL2-Fc fusion bispecifics can be created by using human IL2 with an anti-mouse CD4 antibody such as GK1.5. Human IL2 is known to bind to the mouse IL2 receptor. Experimental autoimmune encephalomyelitis (EAE) is a mouse model of autoimmunity. Mice can be induced for EAE and then treated with mouse surrogate Anti-mCD4.times.IL2 bispecific Fc-fusions. Induction of mouse Tregs should result in less severe disease.
[0445] All cited references are herein expressly incorporated by reference in their entirety.
[0446] Whereas particular embodiments have been described above for purposes of illustration, it will be appreciated by those skilled in the art that numerous variations of the details may be made without departing from the invention as described in the appended claims.
Sequence CWU 0 SQTB SEQUENCE LISTING The patent application
contains a lengthy "Sequence Listing" section. A copy of the
"Sequence Listing" is available in electronic form from the USPTO
web site
(https://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20200339624A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
0 SQTB SEQUENCE LISTING The patent application contains a lengthy
"Sequence Listing" section. A copy of the "Sequence Listing" is
available in electronic form from the USPTO web site
(https://seqdata.uspto.gov/?pageRequest=docDetail&DocID=US20200339624A1).
An electronic copy of the "Sequence Listing" will also be available
from the USPTO upon request and payment of the fee set forth in 37
CFR 1.19(b)(3).
References
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014
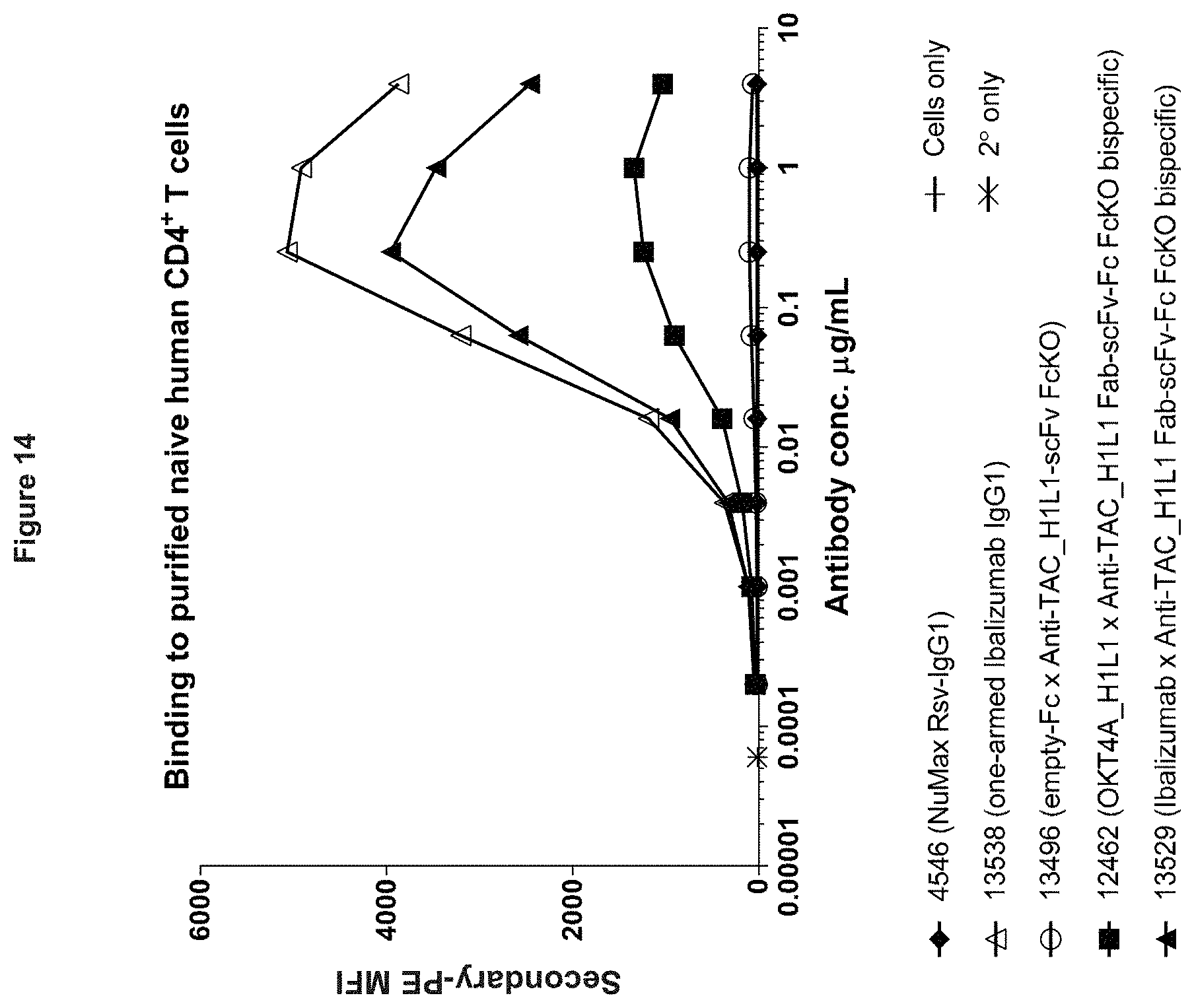
D00015

D00016

D00017
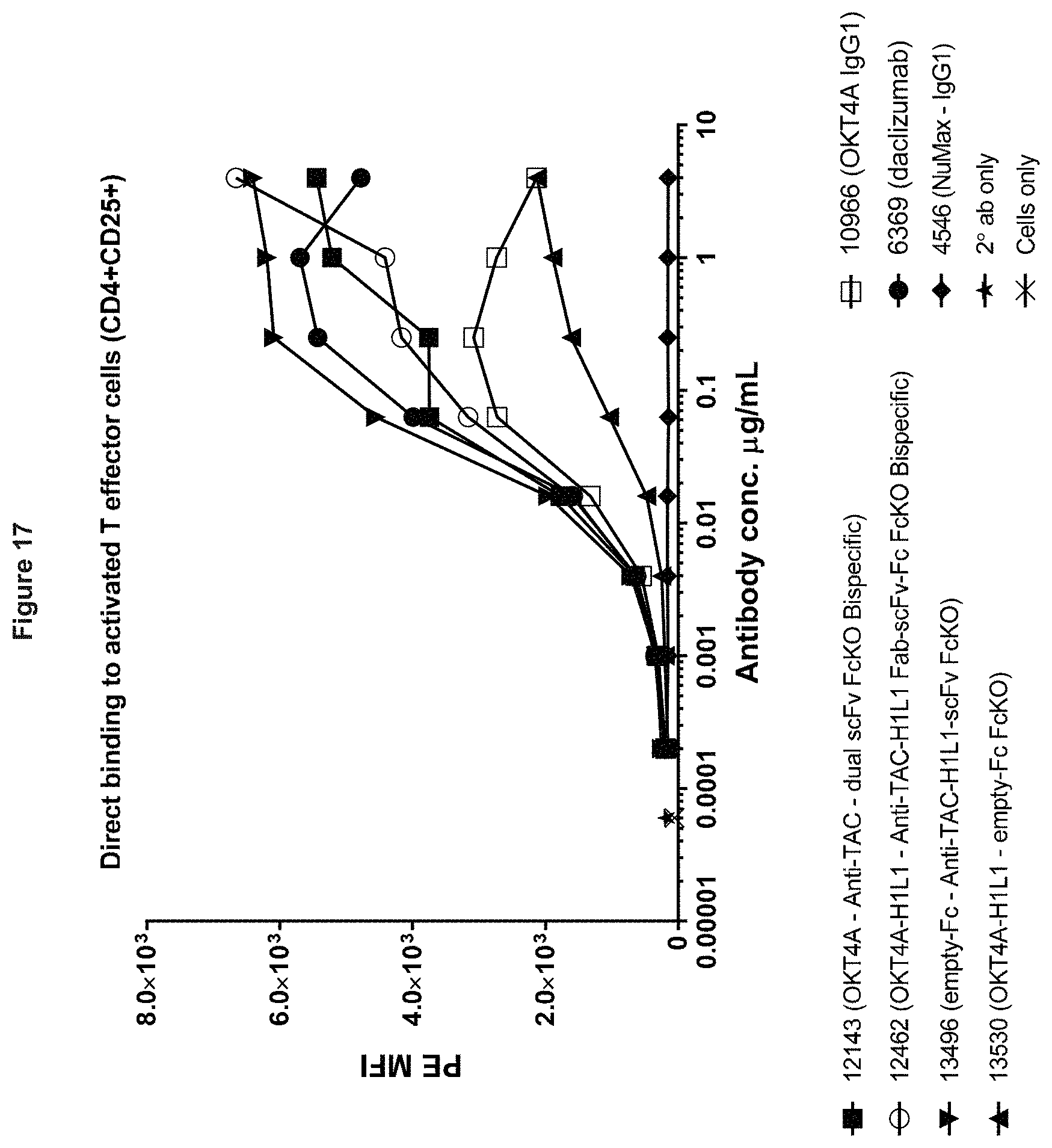
D00018
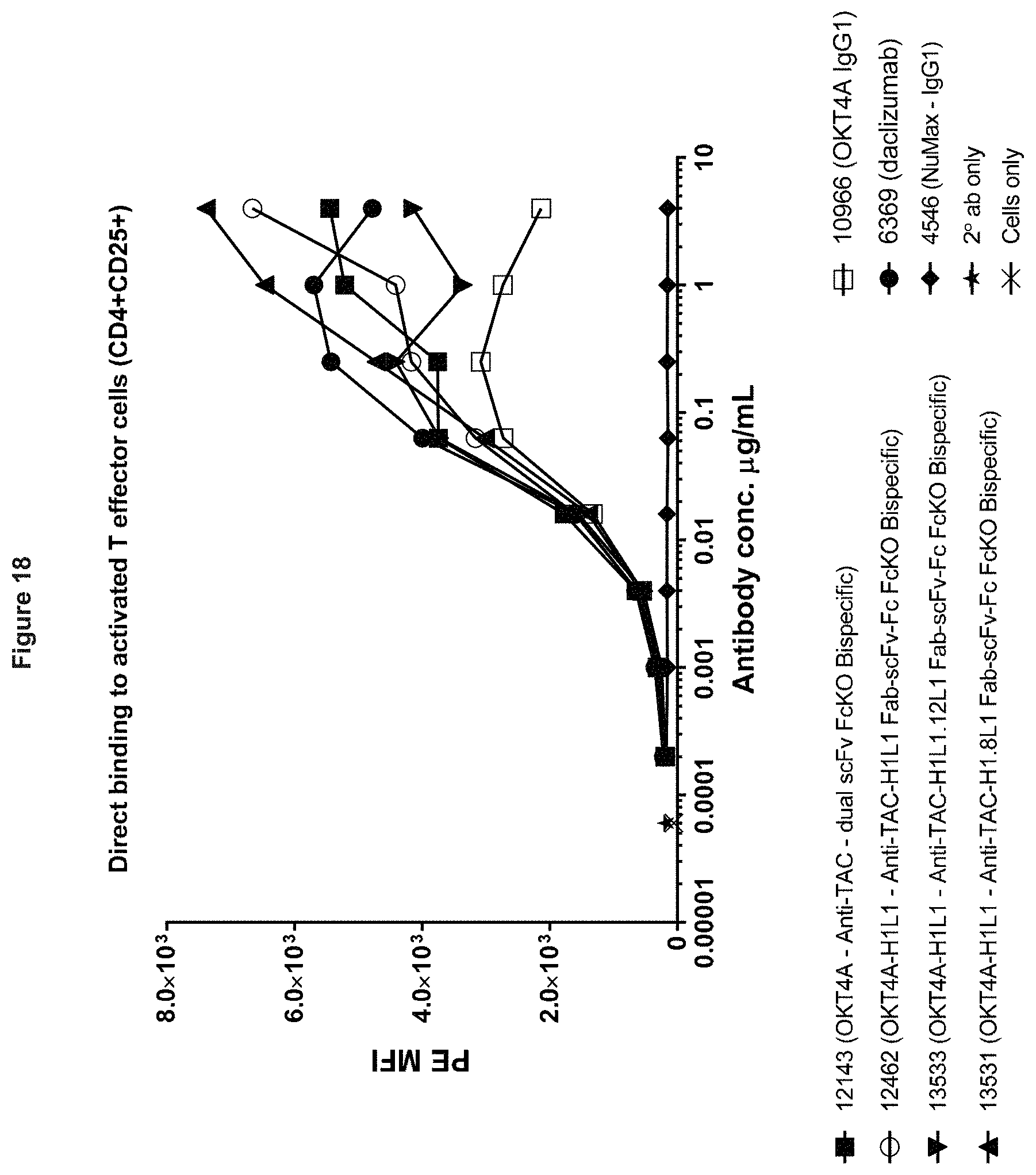
D00019

D00020

D00021

D00022
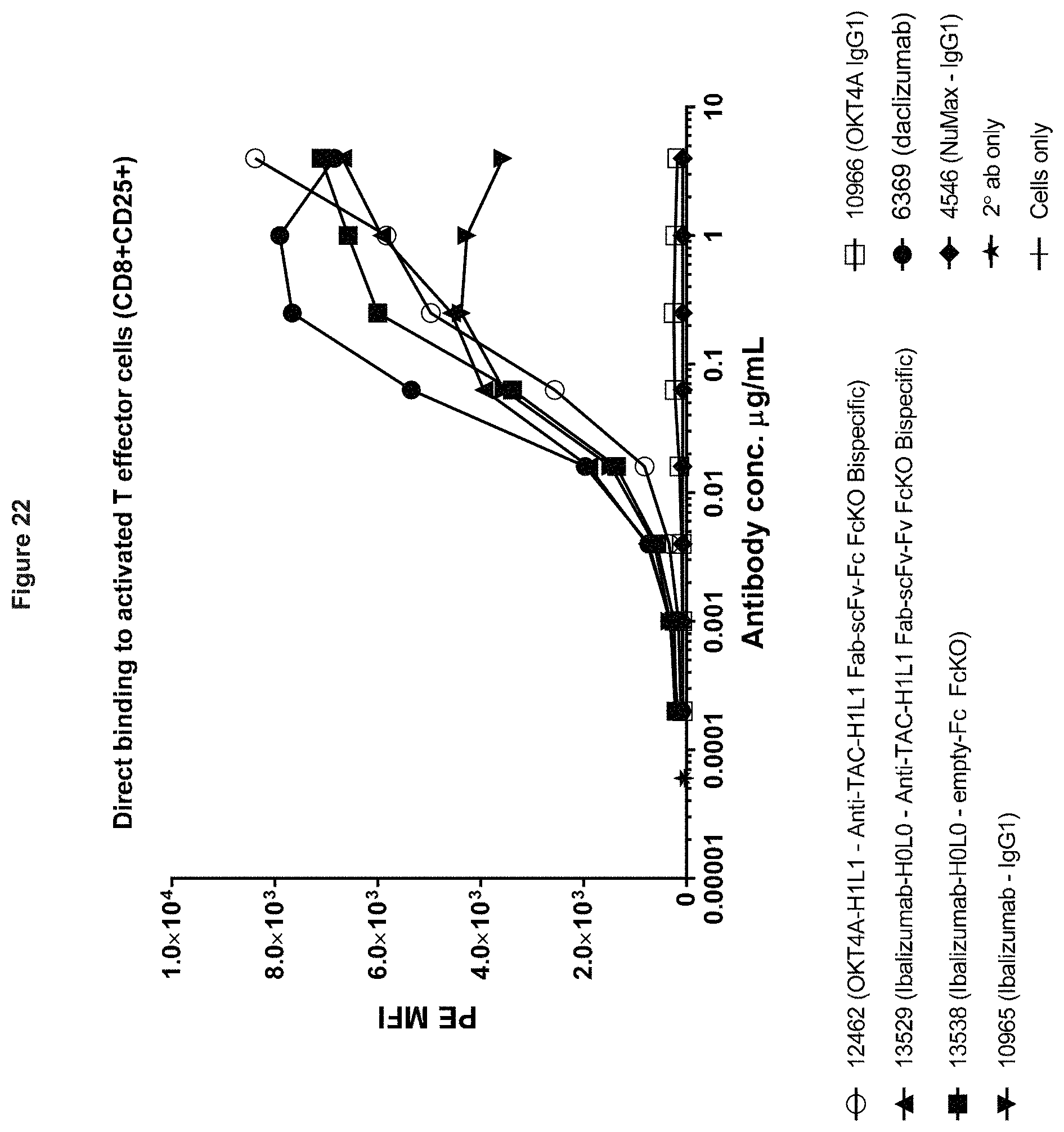
D00023

D00024

D00025

D00026

D00027

D00028
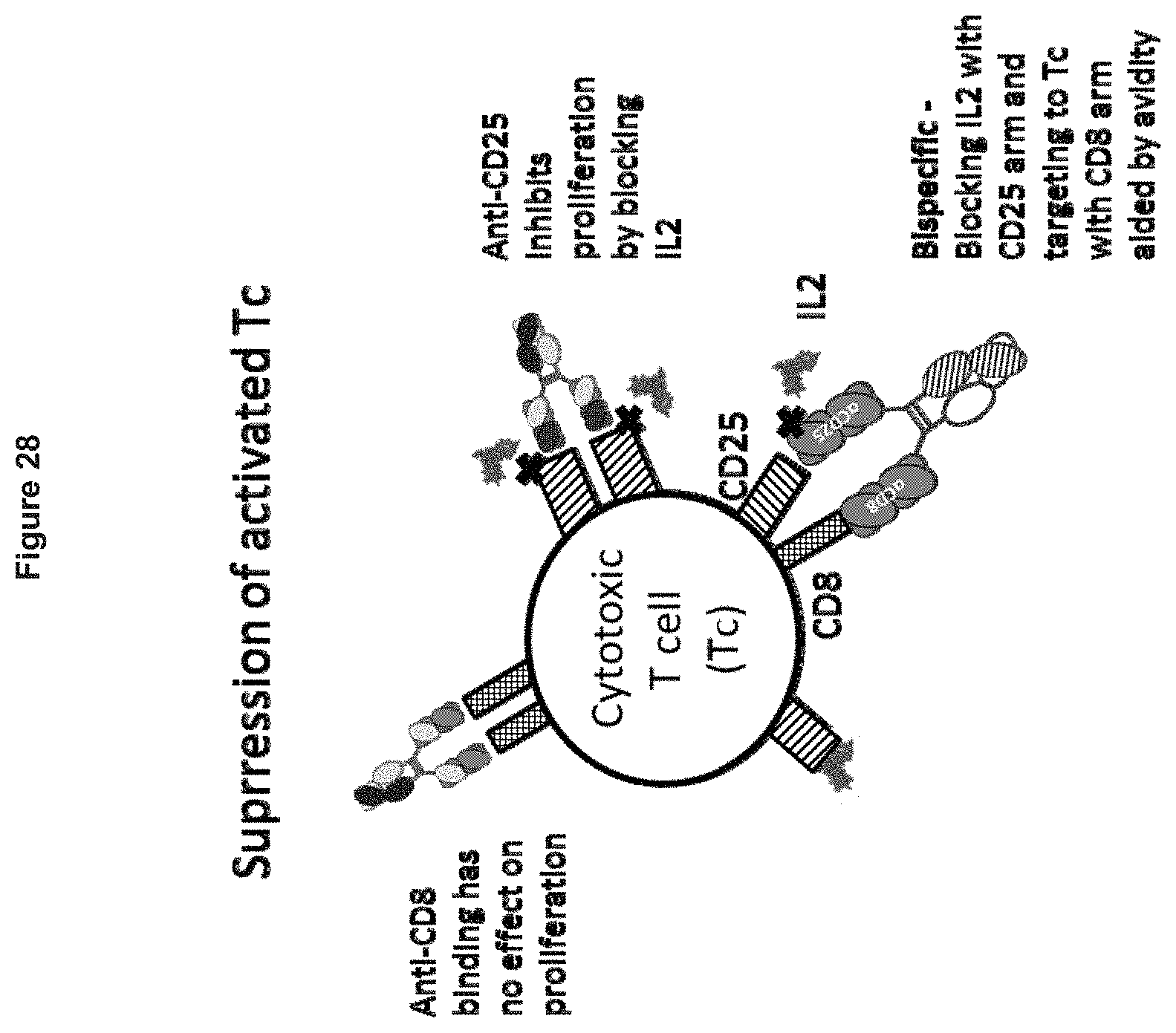
D00029
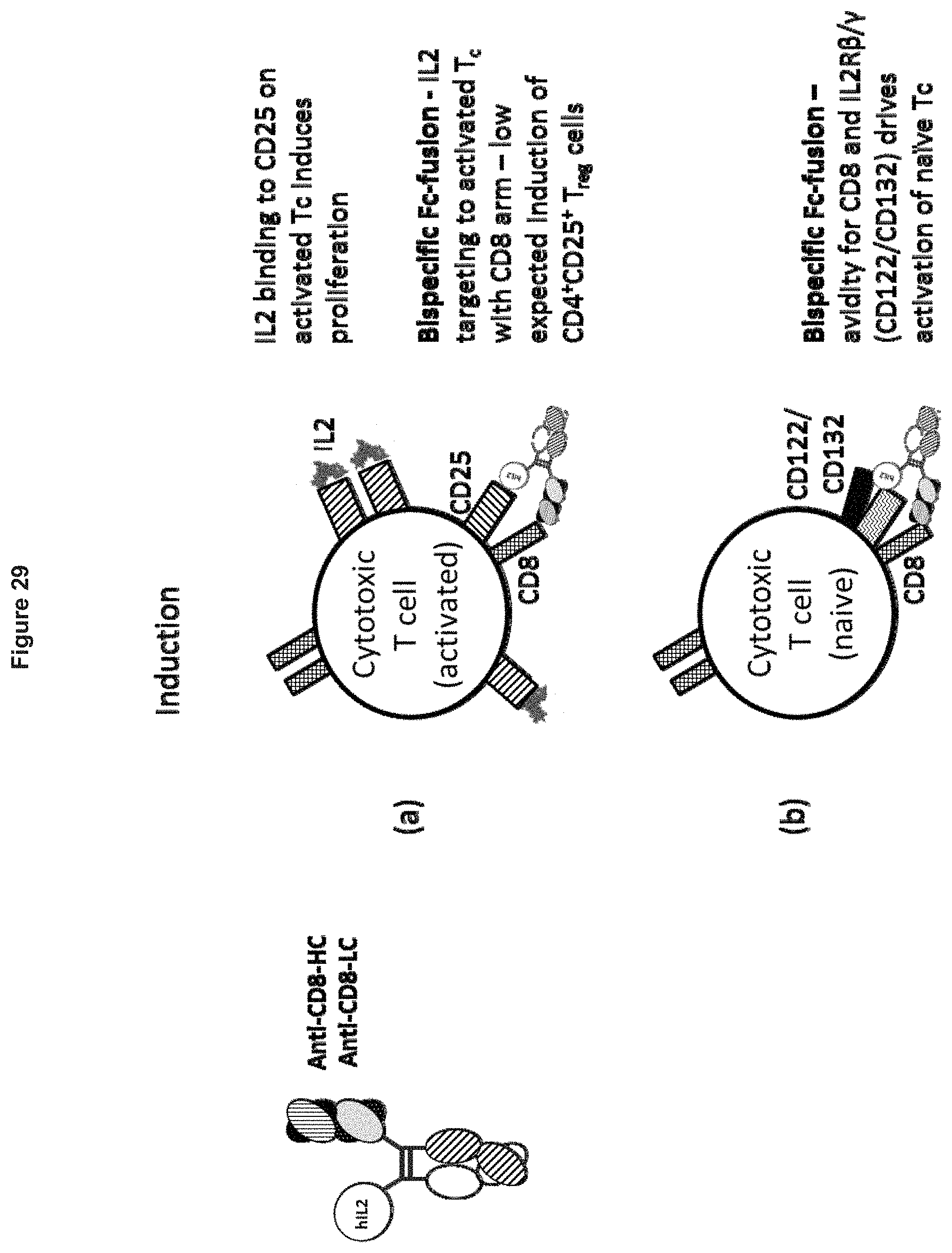
D00030

D00031

D00032

D00033

D00034

D00035

D00036

D00037

D00038

D00039

D00040

D00041

D00042

D00043

D00044

D00045

D00046

D00047

D00048

D00049

D00050

D00051

D00052

D00053

D00054

D00055

D00056

D00057

D00058

D00059

D00060

D00061

D00062

D00063

D00064

D00065

D00066

D00067

D00068

D00069

D00070

D00071

D00072

D00073

D00074

D00075

D00076

D00077

D00078

D00079

D00080

D00081

D00082

D00083

D00084

D00085

D00086

D00087

D00088

D00089

D00090

D00091

D00092

D00093

D00094

D00095

D00096

D00097

D00098

D00099

D00100

D00101

D00102

D00103

D00104

D00105

D00106

D00107

D00108

D00109

D00110

D00111

D00112

D00113

D00114

D00115

D00116

D00117

D00118

D00119

D00120

D00121

D00122

D00123

D00124

D00125

D00126

D00127

D00128

D00129

D00130
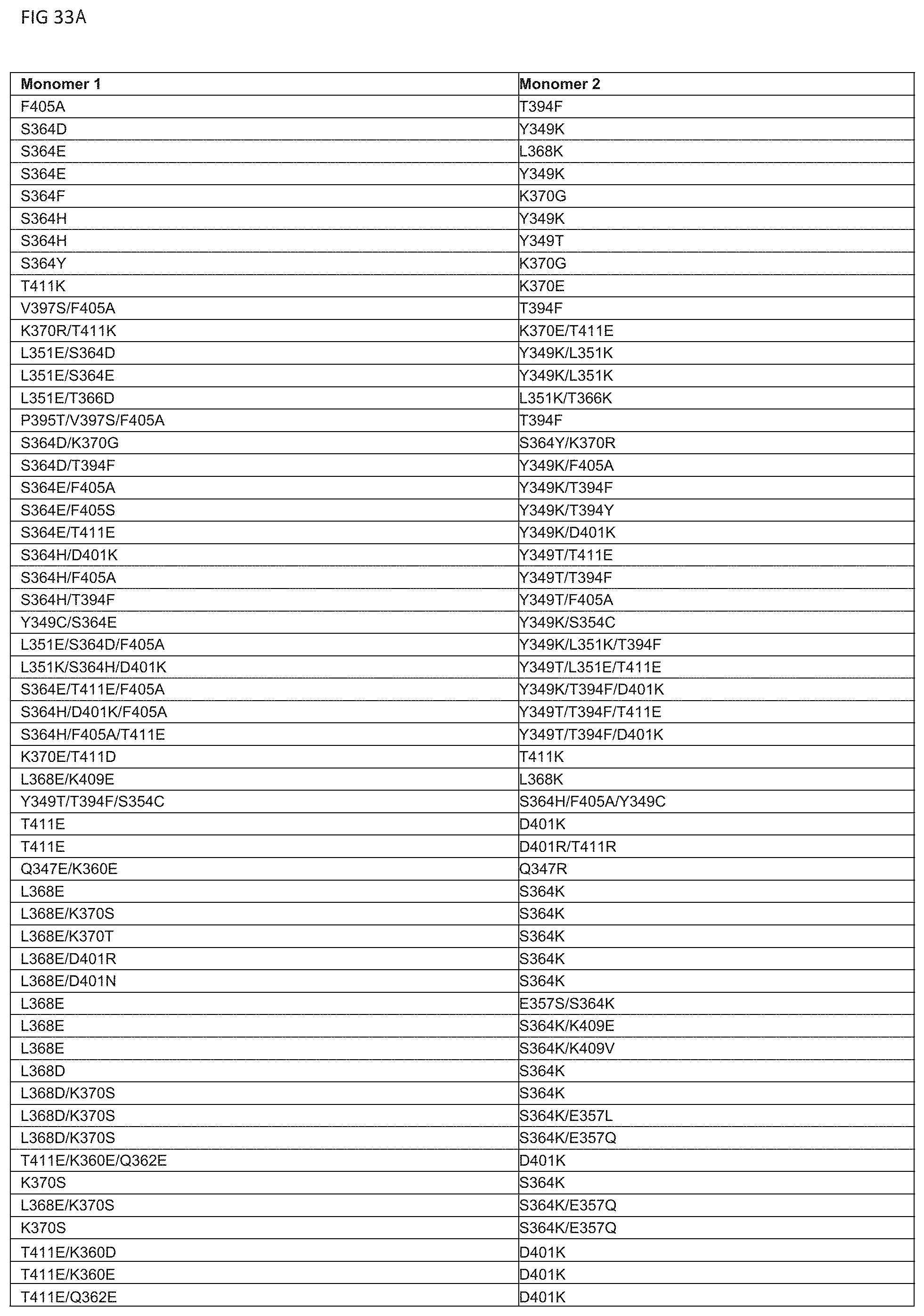
D00131

D00132

D00133
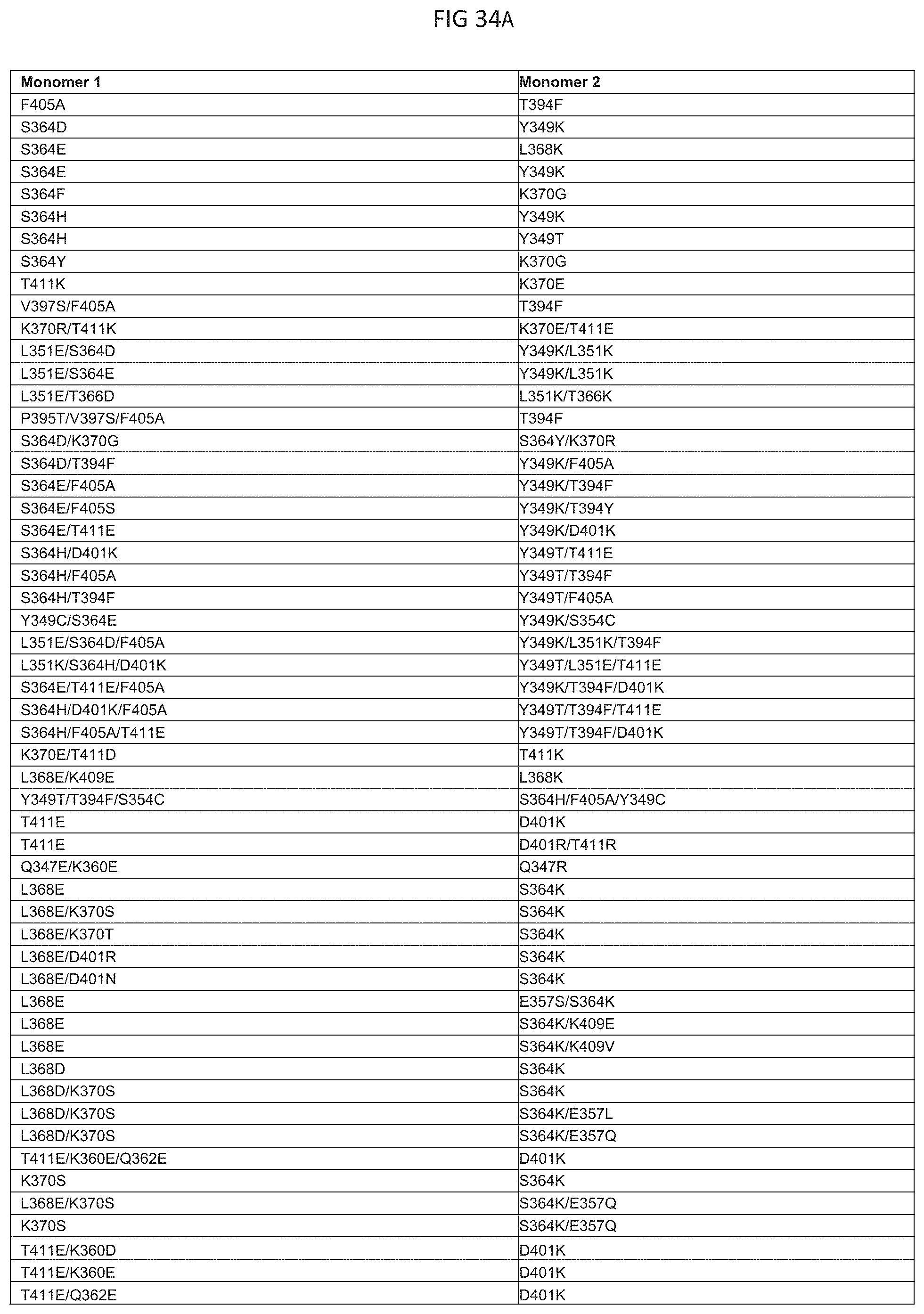
D00134

D00135

D00136

D00137

D00138
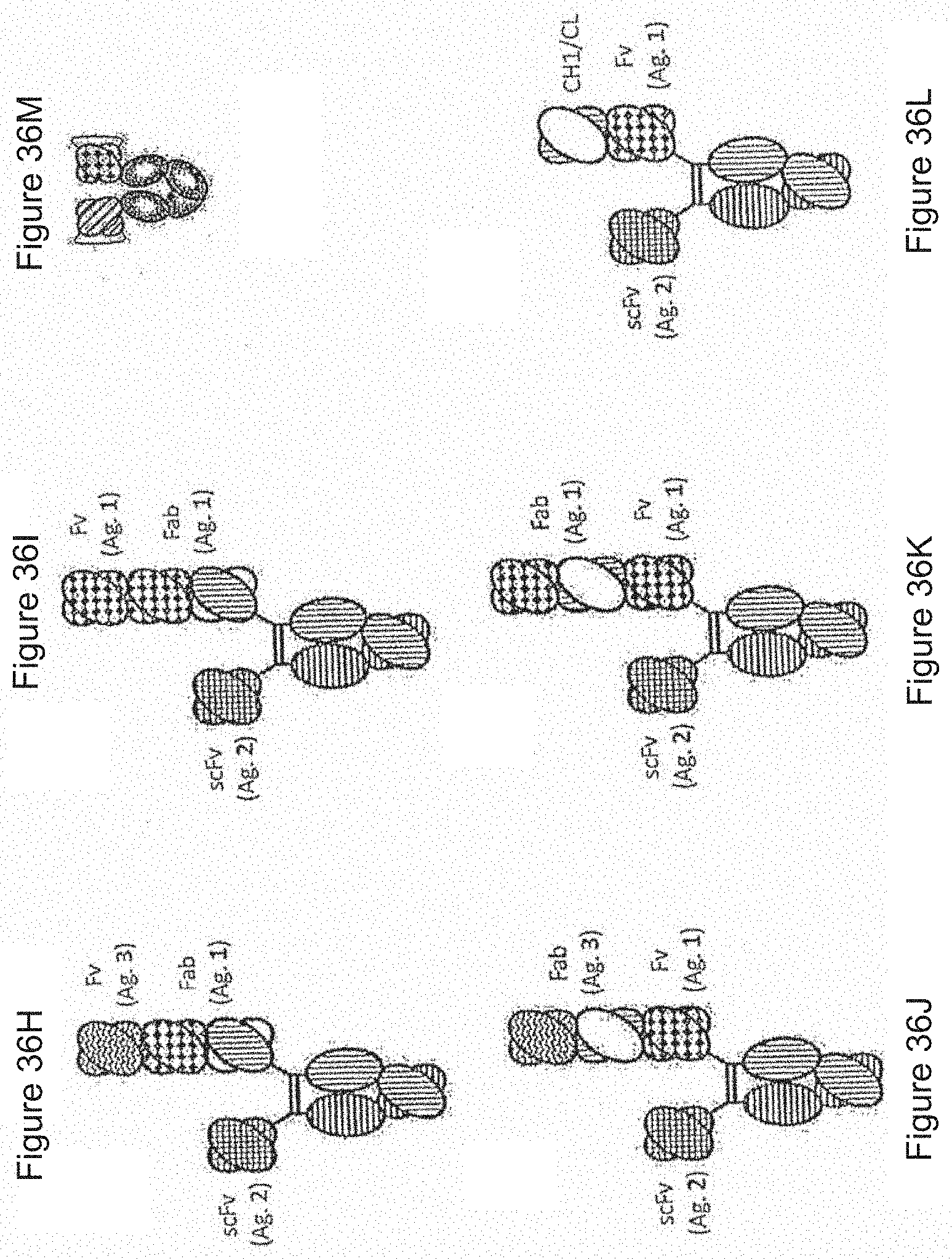
D00139

D00140

D00141

D00142

D00143

D00144
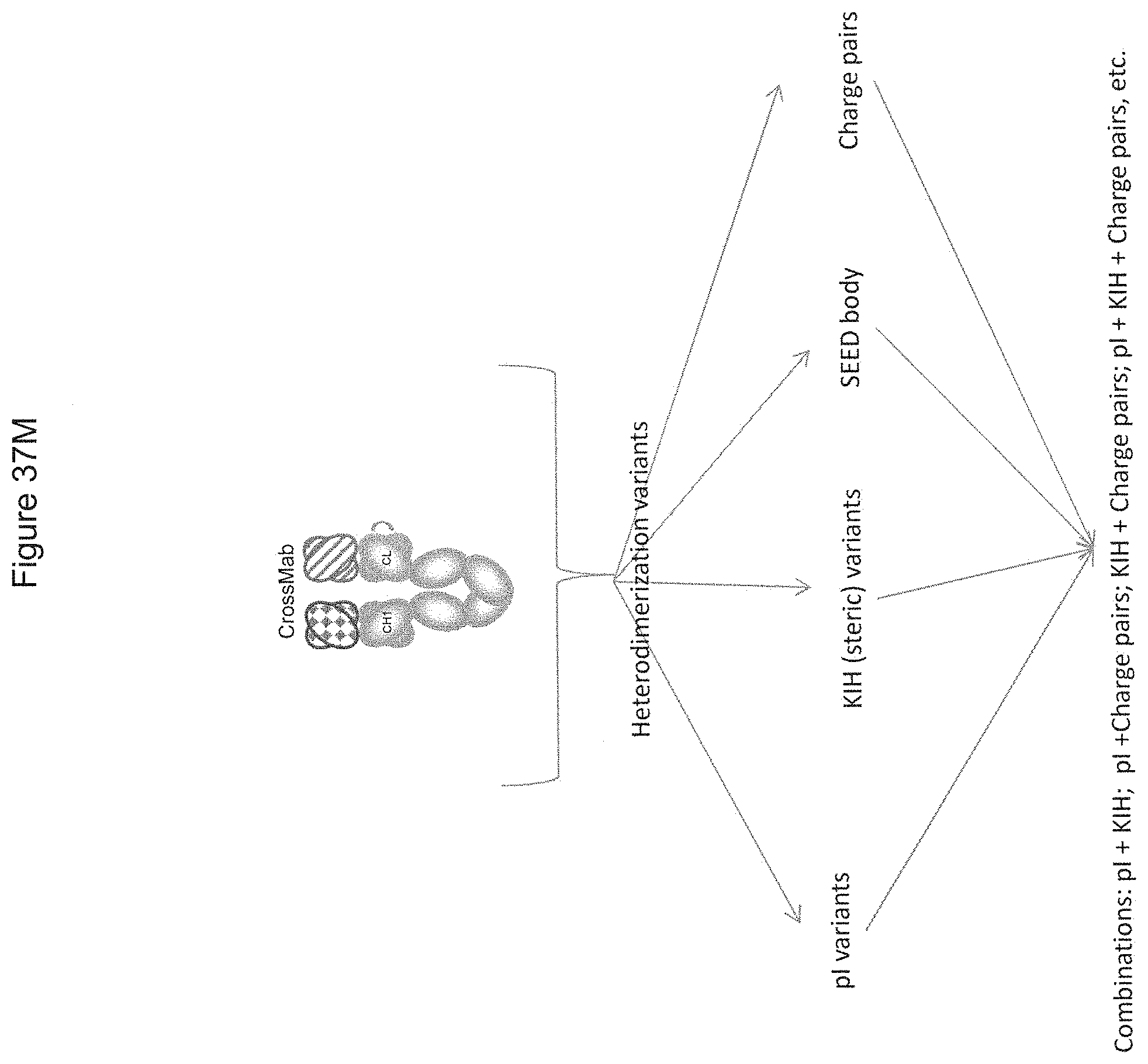
D00145

D00146

D00147

D00148

D00149

D00150

D00151

D00152

D00153

D00154

D00155

D00156
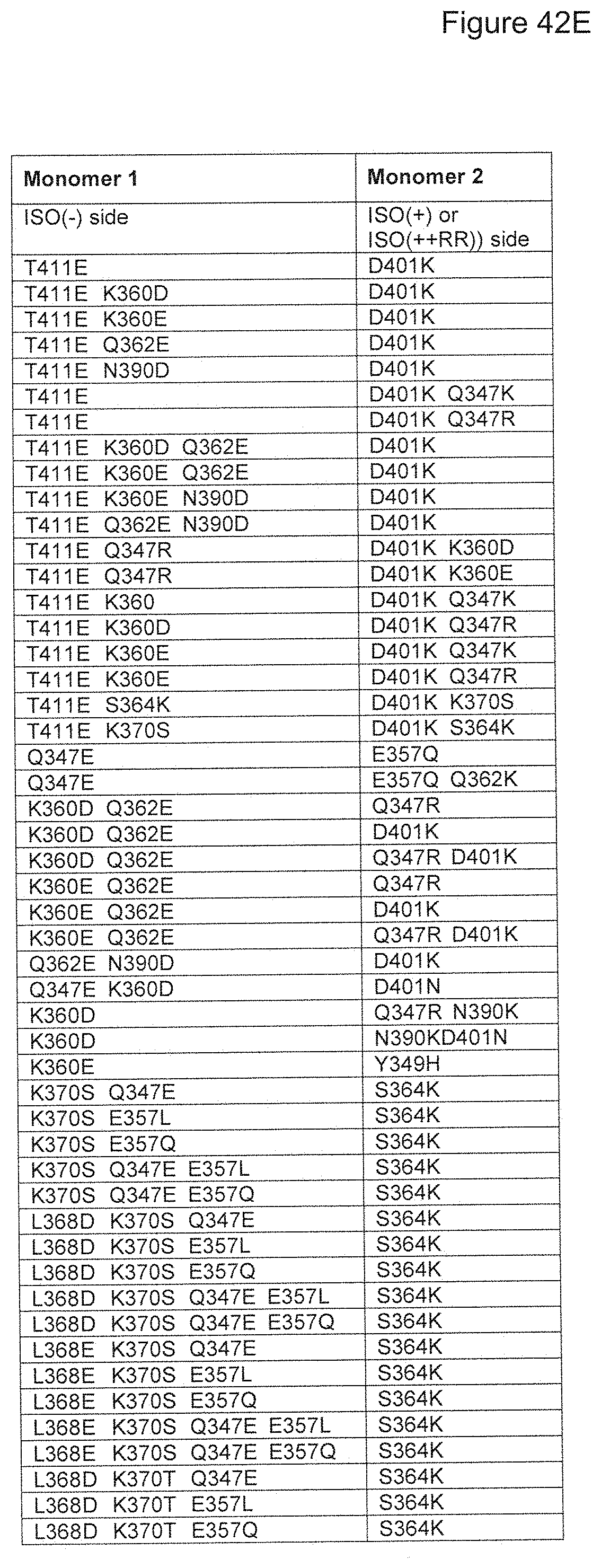
D00157

D00158

D00159

D00160
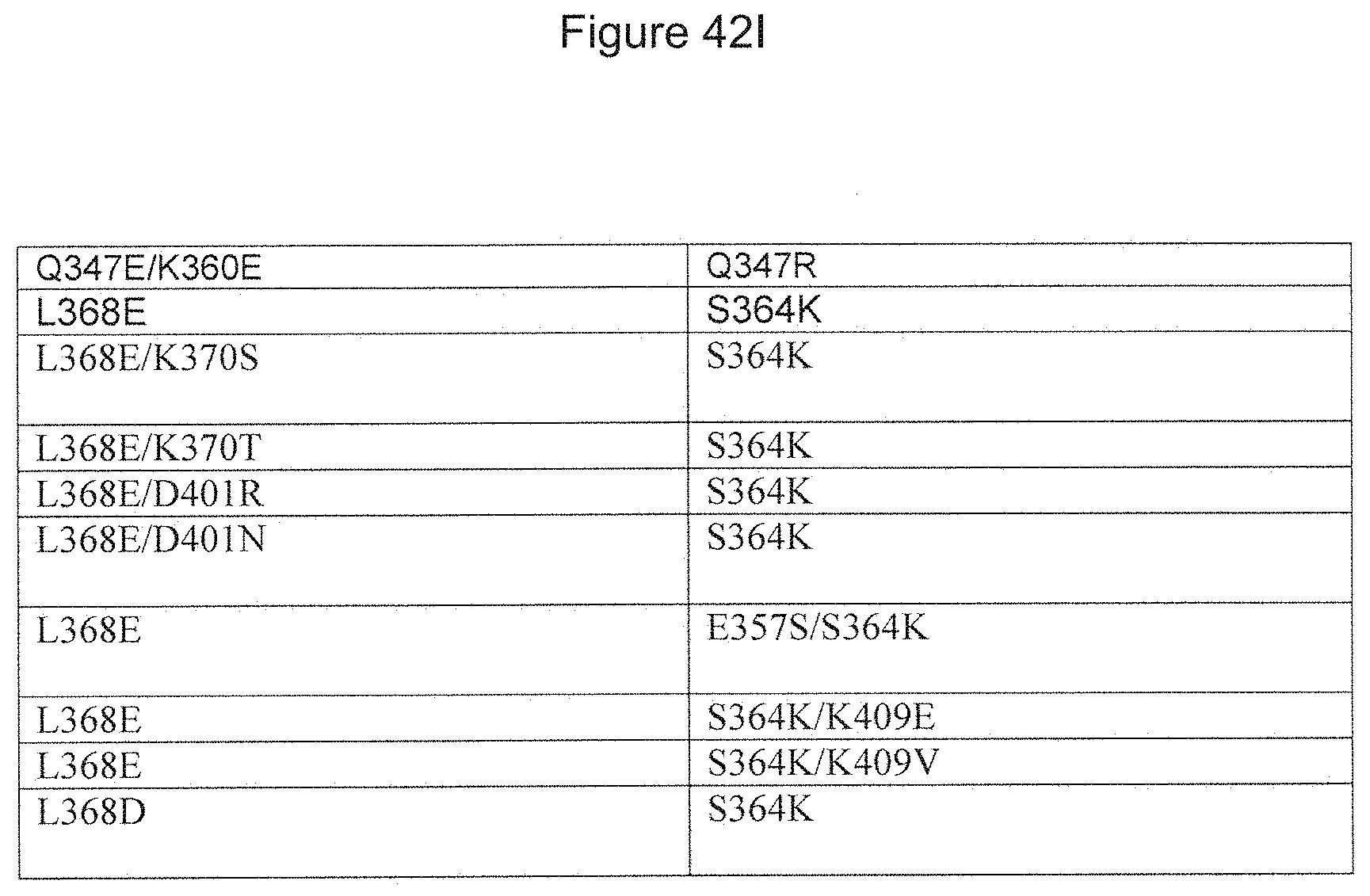
D00161
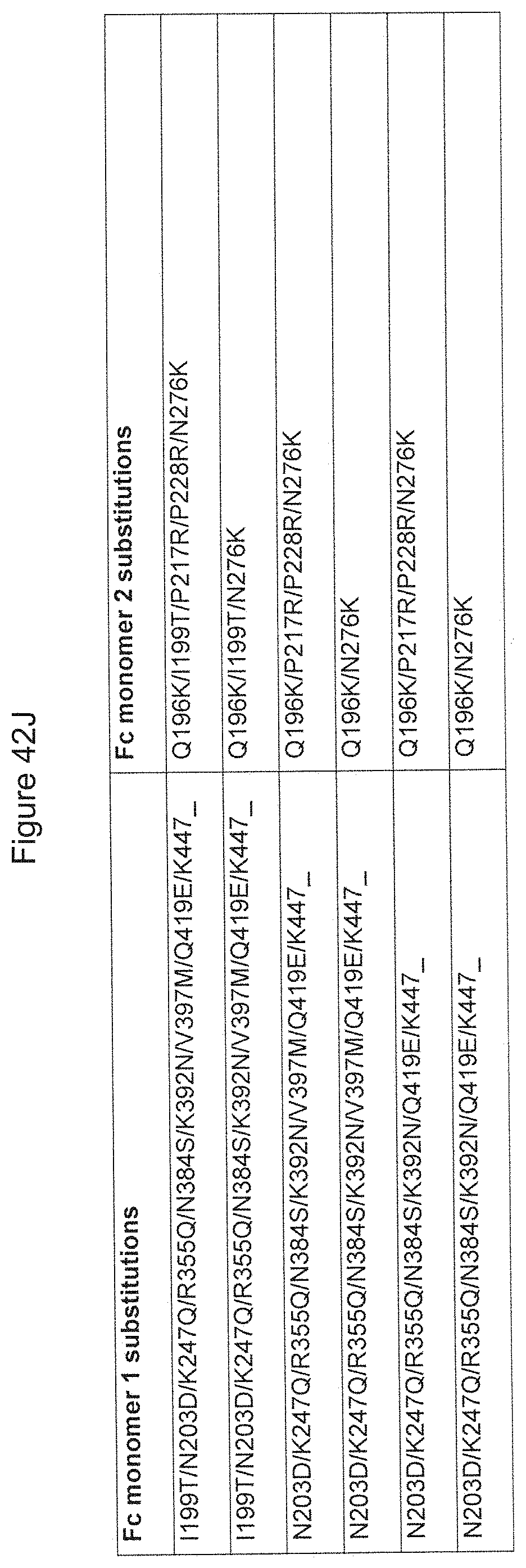
D00162

D00163

D00164

D00165

D00166
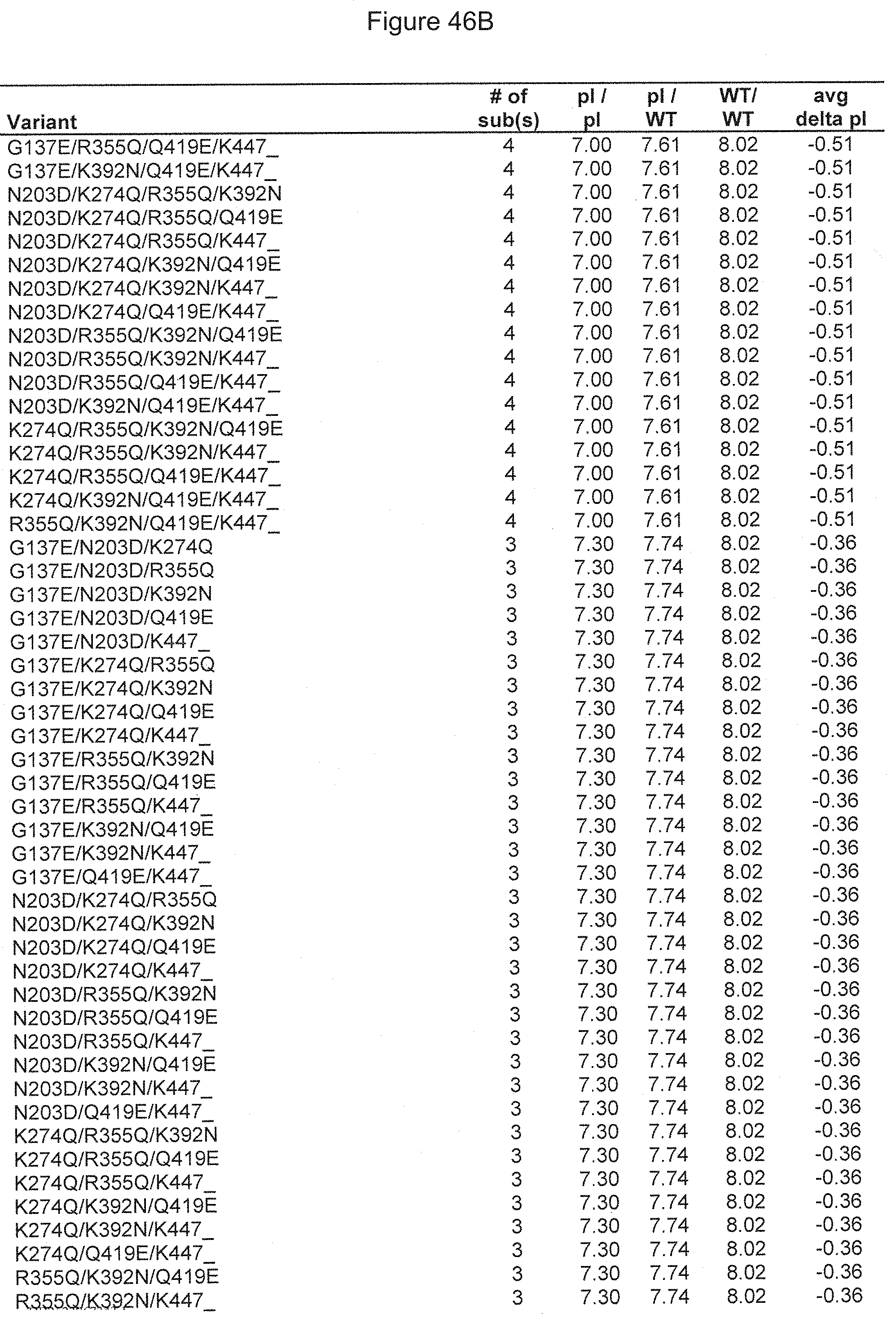
D00167

D00168

D00169

D00170
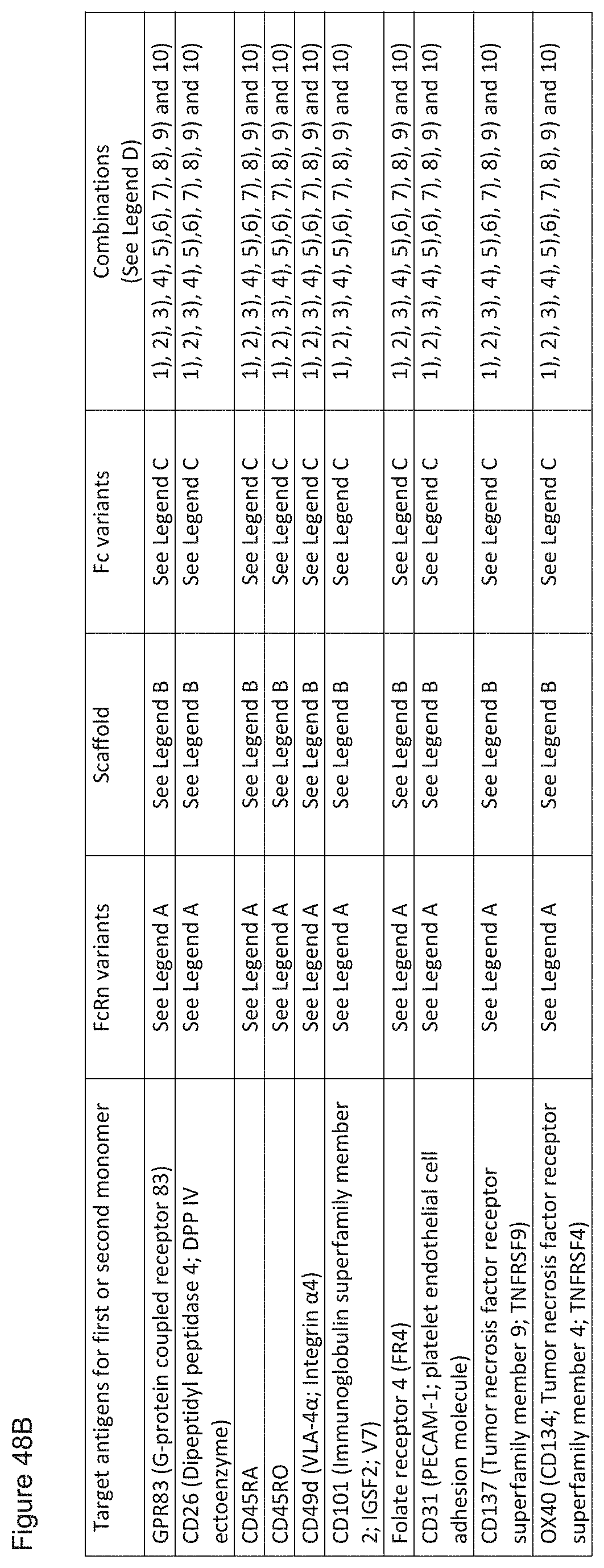
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.