Computationally Optimized Broadly Reactive Antigens For Influenza
Ross; Ted M. ; et al.
U.S. patent application number 16/924032 was filed with the patent office on 2020-10-22 for computationally optimized broadly reactive antigens for influenza. This patent application is currently assigned to University of Pittsburgh - Of the Commonwealth System of Higher Education. The applicant listed for this patent is University of Pittsburgh - Of the Commonwealth System of Higher Education. Invention is credited to Brendan M. Giles, Ted M. Ross.
| Application Number | 20200330584 16/924032 |
| Document ID | / |
| Family ID | 1000004939888 |
| Filed Date | 2020-10-22 |





View All Diagrams
| United States Patent Application | 20200330584 |
| Kind Code | A1 |
| Ross; Ted M. ; et al. | October 22, 2020 |
COMPUTATIONALLY OPTIMIZED BROADLY REACTIVE ANTIGENS FOR INFLUENZA
Abstract
The development of a computationally optimized influenza HA protein that elicits broadly reactive immune response to all H5N1 influenza virus isolates is described. The optimized HA protein was developed through a series of HA protein alignments, and subsequent generation of consensus sequences, for clade 2 H5N1 influenza virus isolates. The final consensus HA amino acid sequence was reverse translated and optimized for expression in mammalian cells. Influenza virus-like particles containing the optimized HA protein are an effective vaccine against H5N1 influenza virus infection in animals.
| Inventors: | Ross; Ted M.; (Athens, GA) ; Giles; Brendan M.; (Centennial, CO) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | University of Pittsburgh - Of the
Commonwealth System of Higher Education Pittsburgh PA |
||||||||||
| Family ID: | 1000004939888 | ||||||||||
| Appl. No.: | 16/924032 | ||||||||||
| Filed: | July 8, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 16128917 | Sep 12, 2018 | |||
| 16924032 | ||||
| 15463960 | Mar 20, 2017 | 10098946 | ||
| 16128917 | ||||
| 14511930 | Oct 10, 2014 | |||
| 15463960 | ||||
| 13822844 | Mar 13, 2013 | 8883171 | ||
| PCT/US2011/051072 | Sep 9, 2011 | |||
| 14511930 | ||||
| 61403407 | Sep 14, 2010 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 2039/5258 20130101; C12N 2760/16123 20130101; C12N 2760/16323 20130101; A61K 39/00 20130101; C07K 2319/00 20130101; C12N 2760/16122 20130101; A61K 2039/545 20130101; C07K 14/11 20130101; A61K 2039/55505 20130101; C12N 2800/22 20130101; A61K 2039/575 20130101; C12N 7/00 20130101; A61K 39/145 20130101; C12N 2760/16223 20130101; C07K 14/005 20130101; C12N 2760/16234 20130101; C12Y 302/01018 20130101; C12N 2760/16334 20130101; C12N 2760/16134 20130101; C12N 9/2402 20130101; C12N 2760/16143 20130101; A61K 2039/54 20130101; C12N 2760/16171 20130101; A61K 39/39 20130101; C12N 15/86 20130101; A61K 39/12 20130101; A61K 2039/552 20130101 |
| International Class: | A61K 39/145 20060101 A61K039/145; A61K 39/12 20060101 A61K039/12; C07K 14/005 20060101 C07K014/005; C07K 14/11 20060101 C07K014/11; A61K 39/39 20060101 A61K039/39; C12N 7/00 20060101 C12N007/00; C12N 9/24 20060101 C12N009/24; C12N 15/86 20060101 C12N015/86 |
Claims
1. A method of eliciting a broadly reactive immune response against influenza virus in a subject, comprising generating an optimized influenza virus polypeptide sequence comprising the steps of: (i) obtaining the amino acid sequences of the polypeptide from a group of influenza virus isolates, wherein the influenza virus isolates are from the same subtype; (ii) organizing the amino acid sequences of the polypeptide from the group of influenza virus isolates by clade or sub-clade and then by geographical region within each clade or sub-clade; (iii) aligning the amino acid sequences within each geographical region to generate primary consensus sequences, wherein each geographic region is represented by a primary consensus sequence; (iv) aligning the primary consensus sequences to generate secondary consensus sequences, wherein each clade or sub-clade is represented by a secondary consensus sequence; and (v) aligning the secondary consensus sequences, thereby generating the optimized influenza virus polypeptide sequence; and administering the optimized influenza virus polypeptide to the subject.
2. The method of claim 1, wherein generating the optimized influenza virus polypeptide sequence further comprises: (vi) reverse translating the optimized influenza virus polypeptide sequence to generate a coding sequence; and (vii) optimizing the coding sequence for expression in mammalian cells.
3. The method of claim 1, comprising administering a virus-like particle (VLP) comprising the optimized influenza virus polypeptide sequence to the subject.
4. The method of claim 3, wherein the influenza virus is an H5N1 virus and the VLP elicits a broadly reactive immune response against H5N1 influenza.
5. The method of claim 4, wherein the VLP elicits a protective immune response against at least 80% of known H5N1 isolates.
6. The method of claim 1, wherein the geographical region is a continent.
7. The method of claim 1, wherein the geographical region is a country.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This is a continuation of U.S. application Ser. No. 16/128,917, filed Sep. 12, 2018, which is a continuation of U.S. application Ser. No. 15/463,960, filed Mar. 20, 2017, issued as U.S. Pat. No. 10,098,946 on Oct. 16, 2018, which is a divisional of U.S. application Ser. No. 14/511,930, filed Oct. 10, 2014, now abandoned, which is a divisional of U.S. application Ser. No. 13/822,844, filed Mar. 13, 2013, issued as U.S. Pat. No. 8,883,171 on Nov. 11, 2014, which is the U.S. National Stage of International Application No. PCT/US2011/051072, filed Sep. 9, 2011, published in English under PCT Article 21(2), which claims the benefit of U.S. Provisional Application No. 61/403,407, filed Sep. 14, 2010. The above-listed applications are herein incorporated by reference in their entirety.
FIELD
[0002] This disclosure concerns an optimized influenza hemagglutinin protein that elicits broadly reactive immune responses to H5N1 virus isolates and its use as a vaccine.
BACKGROUND
[0003] Influenza virus is a member of Orthomyxoviridae family. There are three subtypes of influenza viruses, designated influenza A, influenza B, and influenza C. The influenza virion contains a segmented negative-sense RNA genome, which encodes the following proteins: hemagglutinin (HA), neuraminidase (NA), matrix (M1), proton ion-channel protein (M2), nucleoprotein (NP), polymerase basic protein 1 (PB1), polymerase basic protein 2 (PB2), polymerase acidic protein (PA), and nonstructural protein 2 (NS2). The HA, NA, M1, and M2 are membrane associated, whereas NP, PB1, PB2, PA, and NS2 are nucleocapsid associated proteins. The M1 protein is the most abundant protein in influenza particles. The HA and NA proteins are envelope glycoproteins, responsible for virus attachment and penetration of the viral particles into the cell, and the sources of the major immunodominant epitopes for virus neutralization and protective immunity. Both HA and NA proteins are considered the most important components for prophylactic influenza vaccines.
[0004] Each year, seasonal influenza causes over 300,000 hospitalizations and 36,000 deaths in the U.S. alone (Simonsen et al., Lancet Infect Dis 7:658-66, 2007). The emergence of the novel H1N1 influenza virus in 2009 demonstrated how quickly a new influenza pandemic can sweep across the world. The spread of highly pathogenic H5N1 viruses in birds and coincident infections in humans have raised the concerns that H5N1 viruses may cause a new pandemic in humans. Vaccination is an effective method to prevent influenza infection. There are two influenza vaccine approaches licensed in the United States; the inactivated, split vaccine and the live-attenuated virus vaccine. Inactivated vaccines can efficiently induce humoral immune responses but generally only poor cellular immune responses. Thus, a need exists for a broadly protective influenza virus vaccine.
SUMMARY
[0005] Disclosed herein is the development of an optimized influenza HA protein that elicits broadly reactive immune response to H5N1 influenza virus isolates. The optimized HA protein was developed through a series of HA protein alignments, and subsequent generation of consensus sequences for clade 2 H5N1 influenza virus isolates (FIG. 1). The final consensus HA amino acid sequence was reverse translated and optimized for expression in mammalian cells. The optimized HA coding sequence is set forth herein as SEQ ID NO: 1, and the optimized HA protein sequence is set forth herein as SEQ ID NO: 2.
[0006] Provided herein is an isolated nucleic acid molecule comprising a nucleotide sequence encoding an optimized influenza HA polypeptide, wherein the nucleotide sequence encoding the HA polypeptide is at least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 1. Optimized influenza HA polypeptides encoded by the nucleic acid molecule, vectors comprising the nucleic acid molecule, and host cells containing the disclosed vectors are also provided herein.
[0007] Further provided is an optimized influenza HA polypeptide, wherein the amino acid sequence of the polypeptide is at least 99% identical to SEQ ID NO: 2. Also provided are fusion proteins comprising the optimized HA polypeptide, virus-like particles (VLPs) containing the optimized HA polypeptides, and compositions comprising the optimized HA polypeptide.
[0008] Collections of plasmids are also provided herein. In some embodiments, the collections of plasmids include a plasmid encoding an influenza NA, a plasmid encoding an influenza MA, and a plasmid encoding the optimized HA protein disclosed herein.
[0009] Further provided is a method of eliciting an immune response to influenza virus in a subject by administering the optimized influenza HA protein, fusion proteins containing the optimized influenza HA, or VLPs containing the optimized influenza HA, as disclosed herein. Also provided is a method of immunizing a subject against influenza virus by administering to the subject VLPs containing the optimized influenza HA protein disclosed herein.
[0010] The foregoing and other objects and features of the disclosure will become more apparent from the following detailed description, which proceeds with reference to the accompanying figures.
BRIEF DESCRIPTION OF THE FIGURES
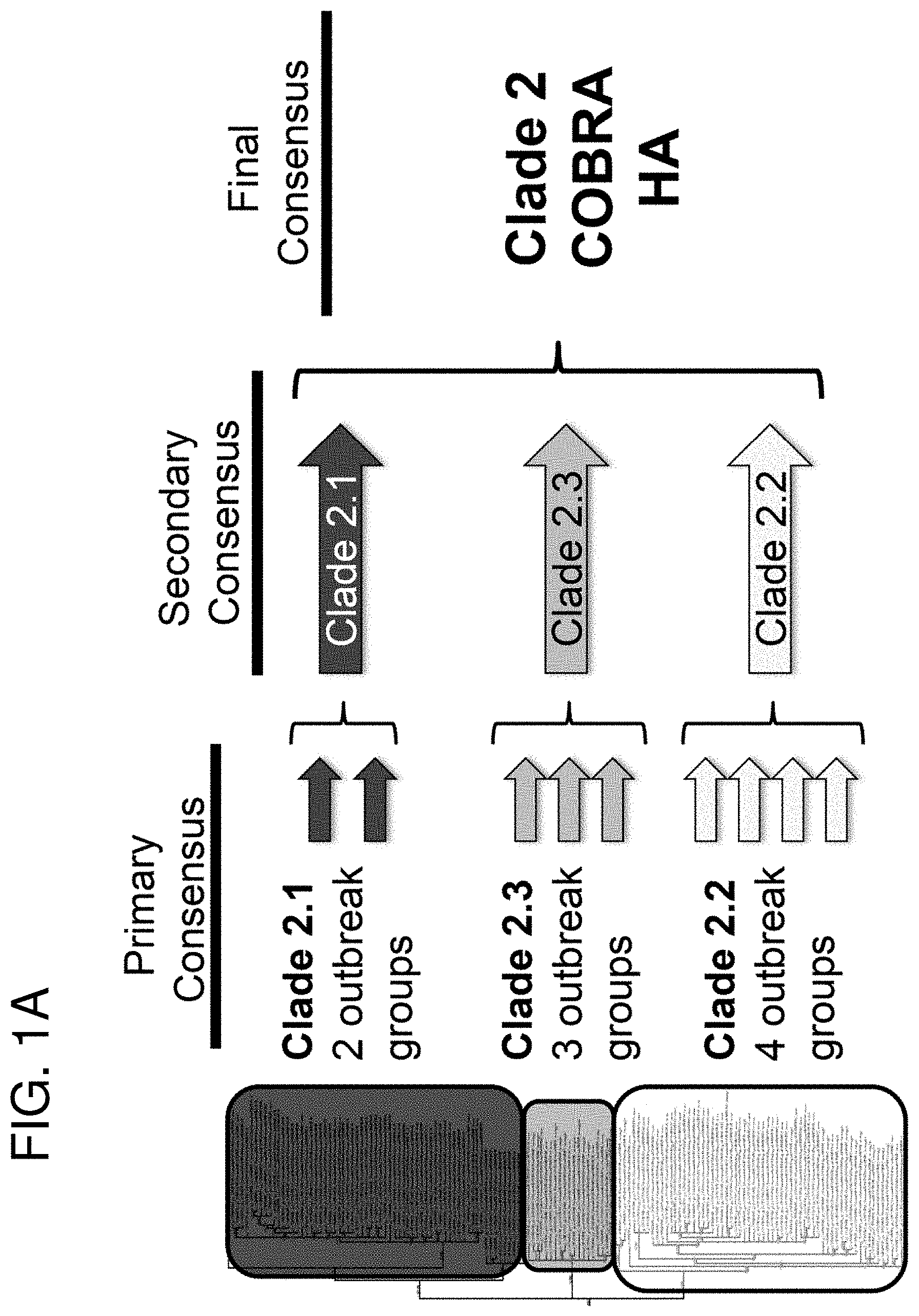
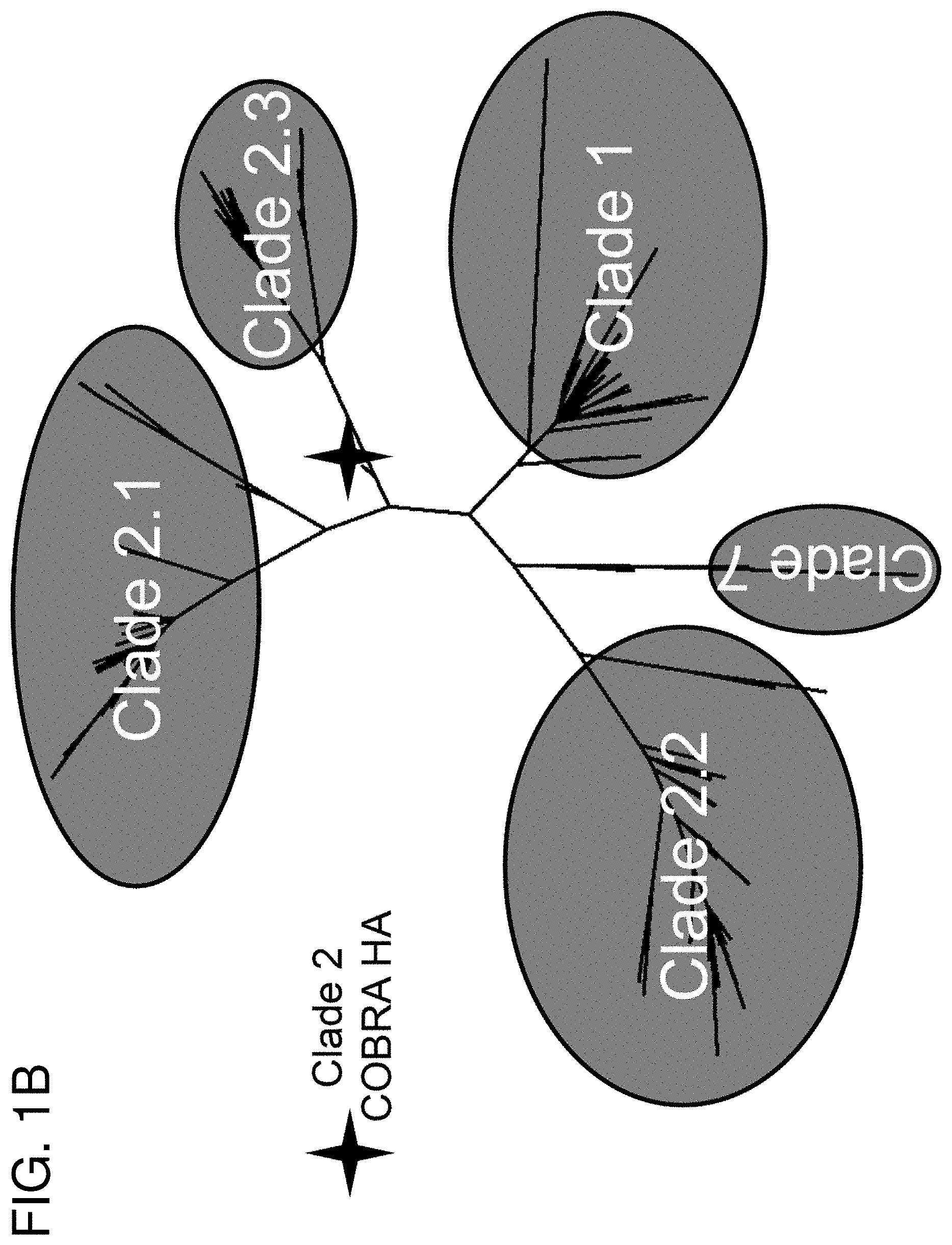
[0011] FIGS. 1A-1B: COBRA HA Design. (FIG. 1A) Schematic illustrating the design of the COBRA HA molecule. The phylogenetic tree was inferred from hemagglutinin amino acid sequences using the maximum likelihood method and clade/sub-clade groupings were identified. Primary consensus sequences were generated for each outbreak group. Secondary consensus sequences were then generated for each sub-clade using the primary sequences as input. The secondary consensus sequences were then aligned and the resulting consensus, designated COBRA, was generated. (FIG. 1B) Phylogenetic analysis of the COBRA HA. The unrooted phylogenetic tree was inferred from hemagglutinin amino acid sequences from human H5N1 infections isolated from 2004 to 2009 and the clade/sub-clade groupings are indicated. The star represents the COBRA HA sequence relative to human H5N1 infections.
[0012] FIGS. 2A-2C: COBRA HA Functional Characterization. COBRA HA was translated in vitro and the cell culture lysates were analyzed by SDS-PAGE (FIG. 2A). Lane designations: 1) H5N1 recombinant HA; 2) COBRA HA; 3) Expression vector; 4) H5N1 reassortant virus. The COBRA HA (lane 2) migrates at its expected molecular weight confirming expression of the synthetic protein. COBRA HA VLPs were prepared in various amounts, serially diluted, and incubated with 1% erythrocytes to evaluate receptor binding (FIG. 2B). HA titer was determined as the last well in which the RBCs remained suspended in a lattice structure. COBRA HA and control lentiviral pseudoparticles packaging a CMV-Luc gene were generated in HEK 293T cells and used to infect MDCK cells with or without trypsin (FIG. 2C). Particle fusion was determined by luciferase production by infected cells.
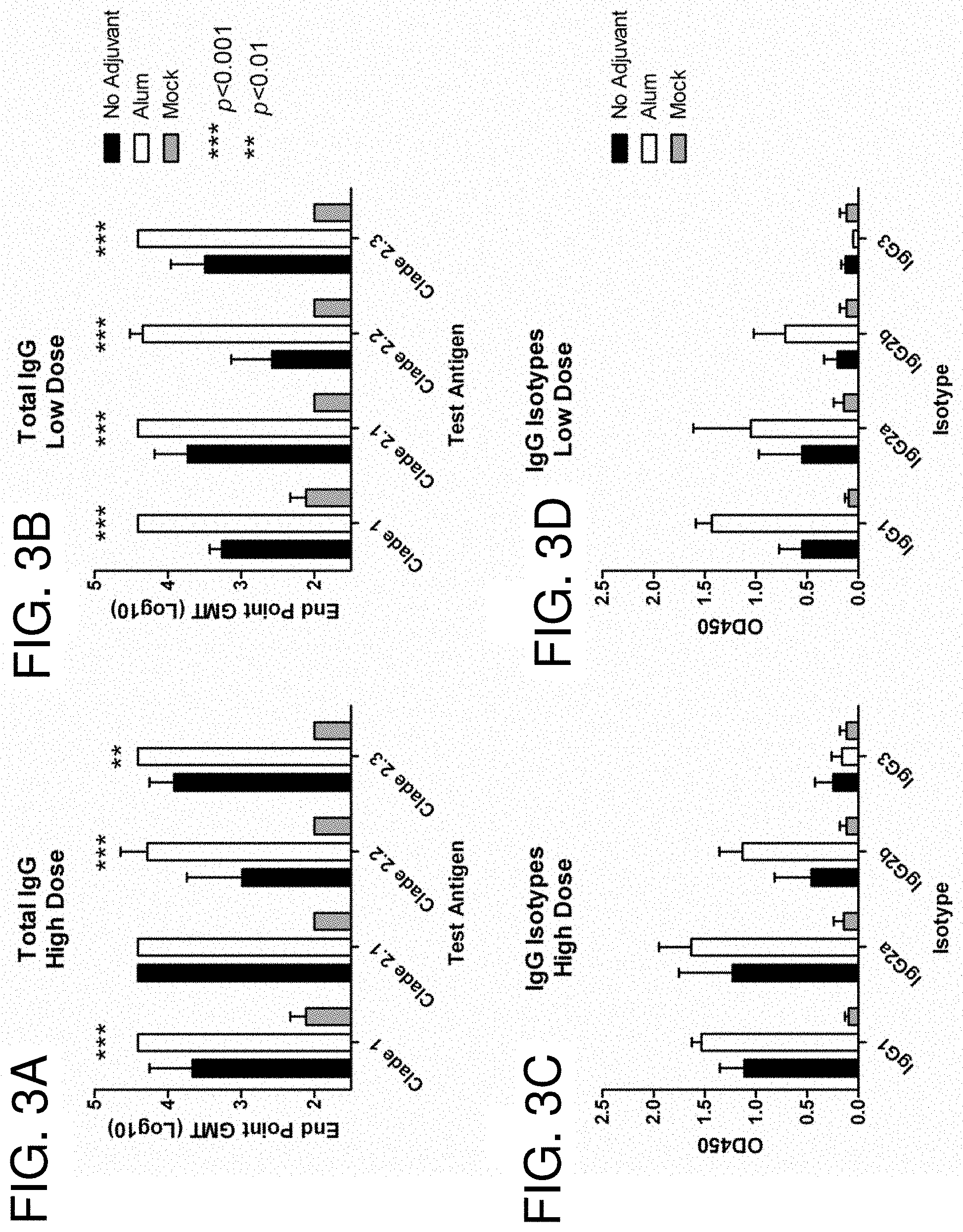
[0013] FIGS. 3A-3F: COBRA HA Mouse Dosing Immunogenicity. BALB/c mice (n=5/group) were vaccinated at 0 and 3 weeks with blood collected at 14 to 21 days after each vaccination. Vaccines were formulated at high (1.5 .mu.g HA), and low (0.03 .mu.g HA) doses, with and without Imject.RTM. alum, and delivered intramuscularly. Total IgG at week 5 was determined via ELISA for each vaccine group (FIGS. 3A and 3B). Values represent the geometric mean titer (.+-.95% confidence interval) of log.sub.10 transformed endpoint titers. IgG isotypes were evaluated via ELISA for each vaccine group (FIGS. 3C and 3D). Values represent the mean OD.sub.450 of a 1:200 dilution of serum. Hemagglutination inhibition (HAI) serum antibody titer for each vaccine group was determined at week 5 using representative reassortant viruses (FIGS. 3E and 3F). Values represent the geometric mean titer (.+-.95% confidence interval) of log 2 transformed titers. The dotted line represents the 1:40 titer. Significant differences were determined by two-way ANOVA with Bonferroni's post-test to evaluate differences between the vaccine formulations for each test antigen. A p value of less than 0.05 was considered significant.
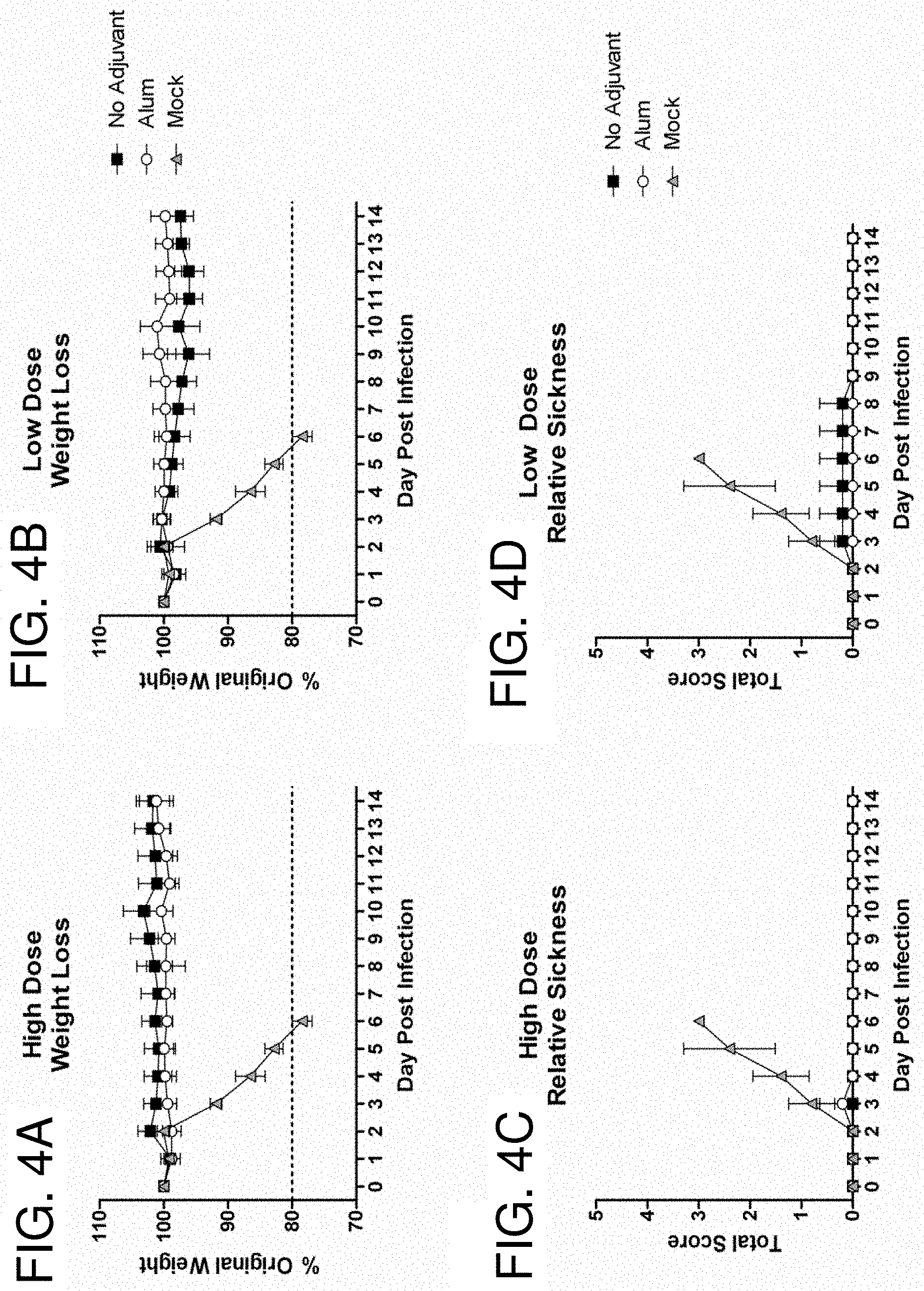
[0014] FIGS. 4A-4D: COBRA HA Mouse Dosing Efficacy. BALB/c mice (n=5/group) were vaccinated with COBRA HA VLPs with or without adjuvant. Mice were infected with 5.times.10.sup.3 PFU of the highly pathogenic clade 2.2 H5N1 virus A/Whooper Swan/Mongolia/244/2005. Mice were followed to monitor weight loss (FIGS. 4A and 4B) and sickness (FIGS. 4C and 4D). Sickness score was determined by evaluating activity (0=normal, 1=reduced, 2=severely reduced), hunched back (0=absent, 1=present) and ruffled fur (0=absent, 1=present). All mock vaccinated mice reached the experimental endpoint and required euthanasia by 6 days post infection.
[0015] FIGS. 5A-5B: Mouse Comparison Immunogenicity. BALB/c mice (n=20/group) were vaccinated at 0 and 3 weeks with blood collected at 14 to 21 days after each vaccination. Vaccines were formulated at a high dose (3 .mu.g HA) with Imject.RTM. alum and delivered intramuscularly. Total IgG at week 5 was determined via ELISA for each vaccine group (FIG. 5A). Values represent the geometric mean titer (.+-.95% confidence interval) of log.sub.10 transformed endpoint titers. Hemagglutination inhibition (HAI) serum antibody titer for each vaccine group was determined at week 5 using representative reassortant viruses (FIG. 5B). Values represent the geometric mean titer (.+-.95% confidence interval) of log 2 transformed titers. The dotted line represents the 1:40 titer. Significant differences were determined by two-way ANOVA with Bonferroni's post-test to evaluate differences between the vaccine formulations for each test antigen. A p value of less than 0.05 was considered significant.
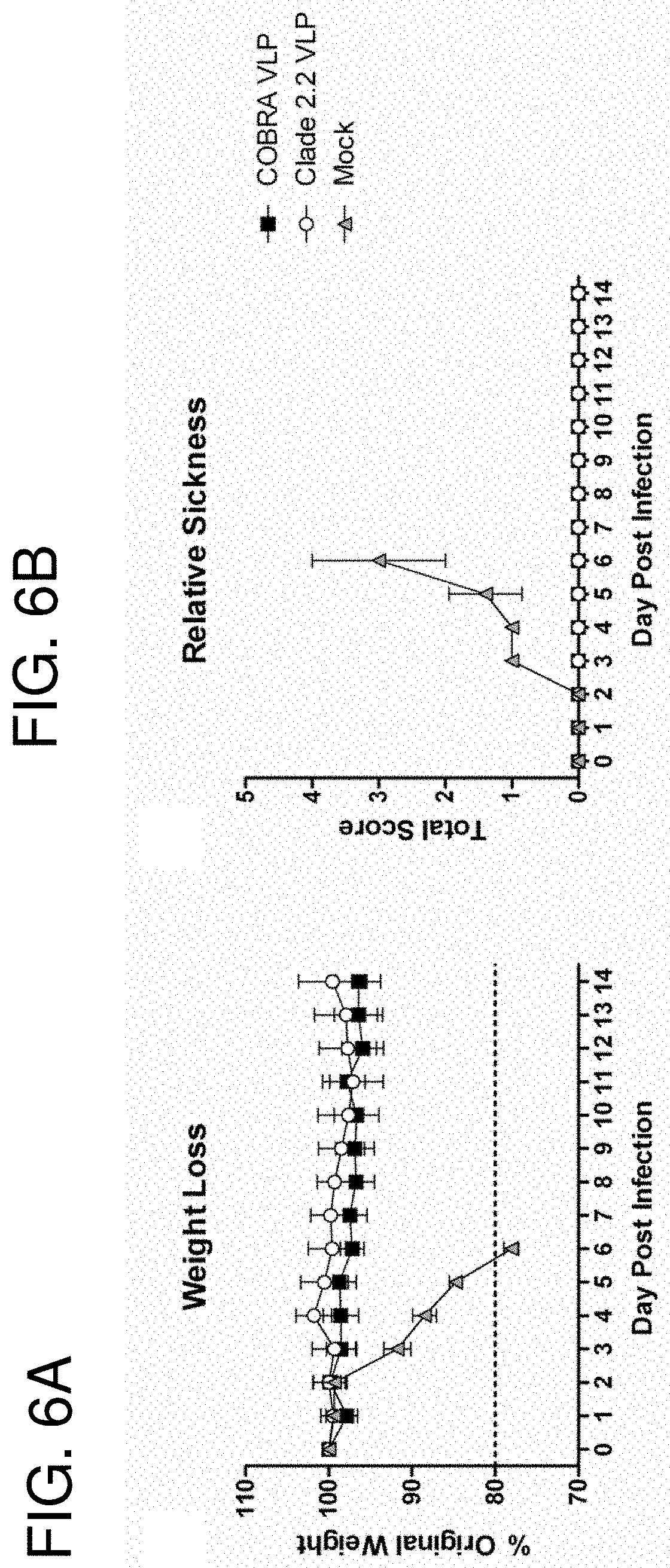
[0016] FIGS. 6A-6B: Mouse Comparison Efficacy. BALB/c mice (n=20/group) were vaccinated with VLPs and adjuvant. Mice were infected with 5.times.10.sup.3 PFU of the highly pathogenic clade 2.2 H5N1 virus A/Whooper Swan/Mongolia/244/2005. Mice were followed to monitor weight loss (FIG. 6A) and sickness (FIG. 6B). Sickness score was determined by evaluating activity (0=normal, 1=reduced, 2=severely reduced), hunched back (0=absent, 1=present) and ruffled fur (0=absent, 1=present). All mock (adjuvant-only) vaccinated mice reached the experimental endpoint and required euthanasia by 6 days post infection.
[0017] FIGS. 7A-7B: Ferret Immunogenicity. Ferrets (n=9/group) were vaccinated with VLPs (15 .mu.g HA) with Imject.RTM. alum at weeks 0 and 3 and serum collected at week 5. Total IgG at week 5 was determined via ELISA for each vaccine group (FIG. 7A). Values represent the geometric mean titer (.+-.95% confidence interval) of login transformed endpoint titers. Hemagglutination inhibition (HAI) serum antibody titer for each vaccine group was determined at week 5 using representative reassortant viruses (FIG. 7B). Values represent the geometric mean titer (.+-.95% confidence interval) of log 2 transformed titers. The dotted line represents the 1:40 titer. Significant differences were determined by two-way ANOVA with Bonferroni's post-test to evaluate differences between the vaccine formulations for each test antigen. A p value of less than 0.05 was considered significant.
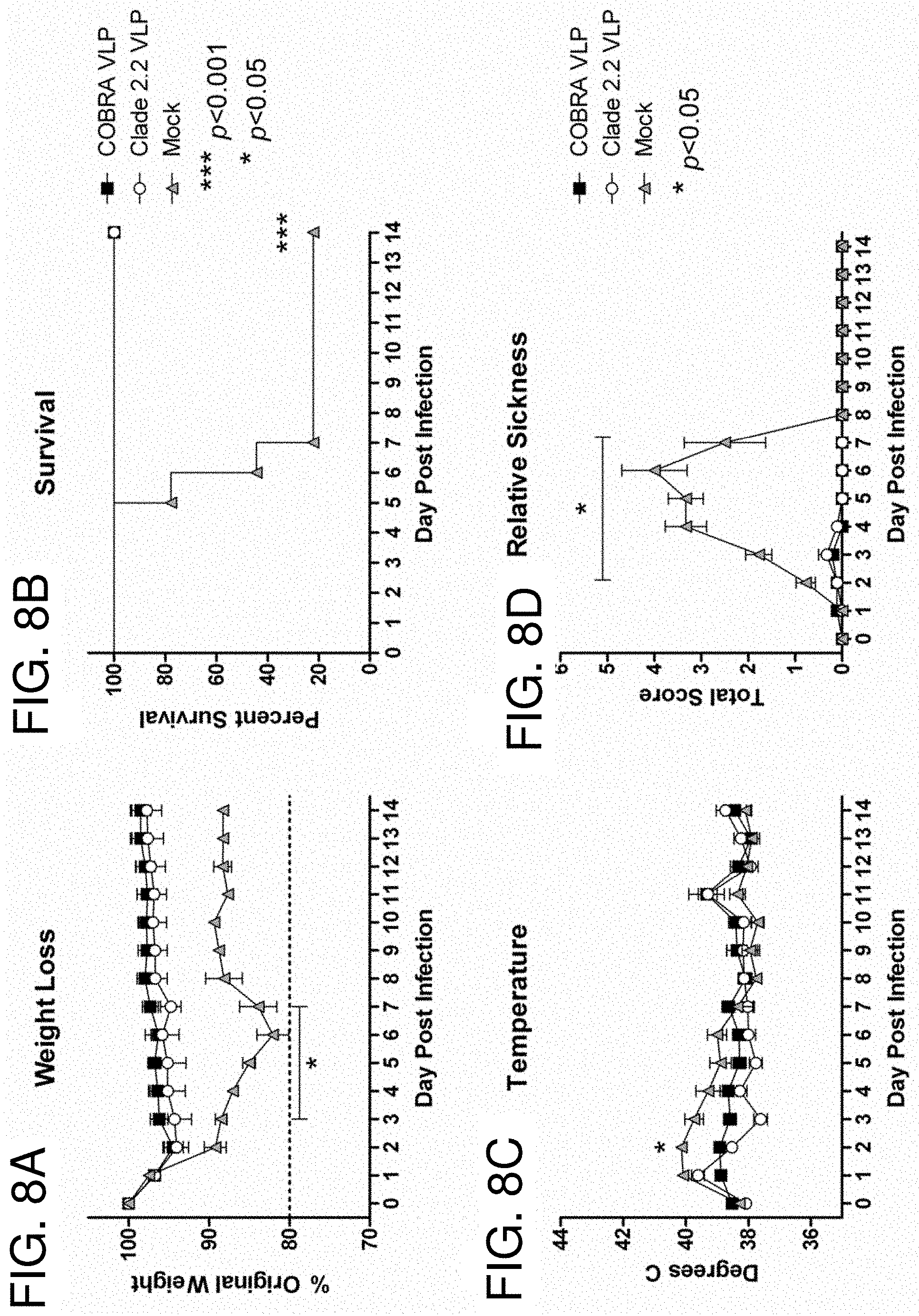
[0018] FIGS. 8A-8E: Ferret Efficacy. Ferrets (n=9/group) were vaccinated with VLPs formulated with adjuvant. Ferrets were challenged with 1.times.10.sup.6 PFU of the highly pathogenic clade 2.2 H5N1 virus A/Whooper Swan/Mongolia/244/2005. Animals were monitored daily for weight loss (FIG. 8A), survival (FIG. 8B), temperature (FIG. 8C) and clinical symptoms (FIG. 8D). Relative sickness scores were determined by measuring lethargy (0-3), runny nose (0-1), sneezing (0-1), loss of appetite (0-1) and diarrhea (0-1). Animals reaching experimental endpoint were euthanized according to institutional guidelines. Nasal washes were collected serially post infection and virus titers determined via plaque assay (FIG. 8E). Statistical significance was determined using a two-way ANOVA with Bonferroni's post test. A p value of less than 0.05 was considered significant.
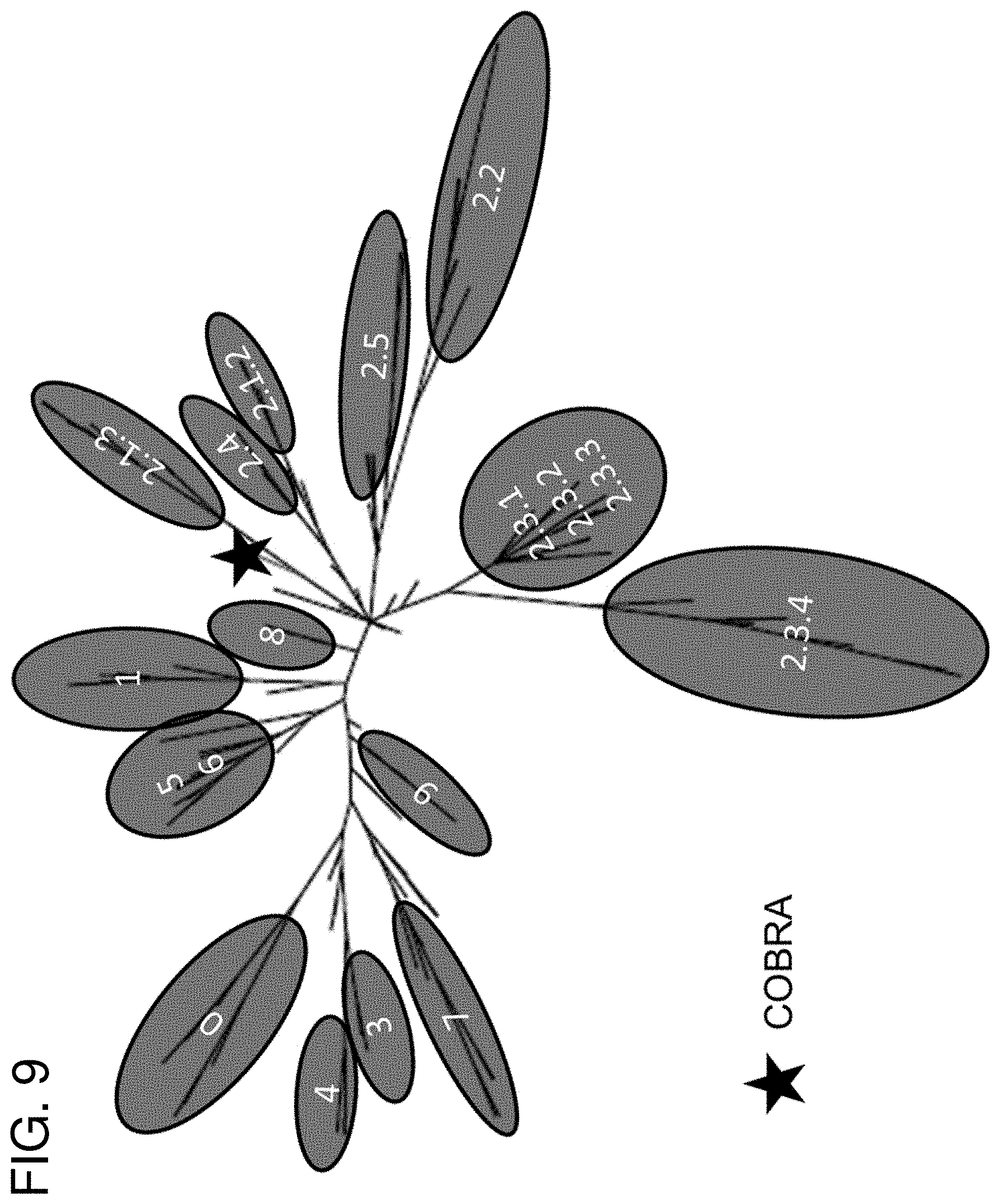
[0019] FIG. 9: Phylogenetic diversity of H5N1 influenza. The unrooted phylogenetic tree was inferred from HA amino acid sequences derived from 8 to 10 representative isolates in all clades and sub-clades and the COBRA HA using the maximum likelihood method. Clade/sub-clade clusters were identified and are indicated in the shaded ovals. The star identifies where the COBRA antigen is located relative to the various representative isolates. Sequences were aligned with MUSCLE 3.7 software and the alignment was refined by Gblocks 0.91b software. Phylogeny was determined using the maximum likelihood method with PhyML software. Trees were rendered using TreeDyn 198.3 software (Dereeper et al., Nucleic Acids Res 36:W465-W469, 2008). The NCBI accession numbers for the HA sequences used in phylogeny inference were obtained through the Influenza Virus Resource (Bao et al., J Virol 82:596-601, 2008).
[0020] FIGS. 10A-10F: Serology. Total IgG at week 3 (FIG. 10A) and week 6 (FIG. 10B) was determined via ELISA for each vaccine group. Each collected antiserum was assayed for antibody binding to representative HA molecules from clade 2.1 (A/Indonesia/5/2005), clade 2.2 (A/Whooper Swan/Mongolia/244/2005), and clade 2.3 (A/Anhui/1/2005). Values represent the geometric mean titer (.+-.95% confidence interval) of log.sub.10 transformed endpoint titers. Statistical significance of the antibody titer data was determined using a two-way analysis of variance (ANOVA) followed by Bonferroni's post-test to analyze differences between each vaccine group for each of the different antigens that were tested (multiparametric). Significance was defined as p<0.05. Statistical analyses were performed with GraphPad Prism software. HAI titer for each vaccine group was determined at week 3 (FIG. 10C) and week 6 (FIG. 10D) using representative H5N1 influenza viruses: clade 2.1 (A/Indonesia/5/2005), clade 2.2 (A/Whooper swan/Mongolia/244/2005), and clade 2.3 (A/Anhui/1/2005). Values represent the geometric mean titer (.+-.95% confidence interval) of log 2 transformed titers. The dotted line represents the 1:40 titer. Significant differences were determined by two-way ANOVA with Bonferroni's post-test to evaluate differences between the vaccine formulations for each test antigen. A p value of less than 0.05 was considered significant. The number of monkeys that responded with a titer greater than 1:40 is listed above each bar. Neutralizing antibody at week 3 (FIG. 10E) and week 6 (FIG. 10F) was determined via microneutralization assay for each vaccine group. Values represent the geometric mean titer (.+-.95% confidence interval).
[0021] FIG. 11: HAI serum antibody titers from vaccinated monkeys against a panel of clade 0, 1, 2, 4, and 7 isolates. HAI titer for each vaccine group was determined at week 9 using H5N1 influenza viruses. Values represent the geometric mean titer (.+-.95% confidence interval) of log 2 transformed titers. Significant differences were determined by two-way ANOVA with Bonferroni's post-test to evaluate differences between the vaccine formulations for each test antigen. A p value of less than 0.05 was considered significant as described in FIG. 10.
[0022] FIGS. 12A-12D: Vaccine induced serum antibody responses. BALB/c mice (n=30/group) or Fitch ferrets (n=6/group) were vaccinated at 0 and 3 weeks with blood collected 14 to 21 days after each vaccination. Total IgG after the second vaccination was determined via ELISA for each vaccine group (FIGS. 12A and 12C). Receptor blocking antibody titers after the second vaccination were determined via hemagglutination inhibition (HAI) for each vaccine group (FIGS. 12B and 12D). Values represent the geometric mean of the reciprocal dilution (+/-95% confidence interval) of the last positive well. Significant differences between COBRA and polyvalent vaccines were determined by a two-tailed Student's T test and a p value of less than 0.05 was considered significant (*).
[0023] FIGS. 13A-13D: Highly pathogenic Clade 2.2 challenge. Vaccinated BALB/c mice (n=5/group) were infected with 5.times.10.sup.3 PFU of the highly pathogenic clade 2.2 H5N1 virus A/Whooper Swan/Mongolia/244/2005 (WS/05). Mice were monitored daily for weight loss (FIG. 13A) and sickness (FIG. 13B). Vaccinated Fitch ferrets (n=6/group) were infected with 1.times.10.sup.6 PFU of the highly pathogenic clade WS/05 virus. Ferrets were monitored daily for weight loss (FIG. 13C) and sickness (FIG. 13D). Values represent mean (+/-SEM) for each group.
[0024] FIGS. 14A-14B: Clade 2.2 viral loads. Vaccinated BALB/c mice (n=15/group) were infected with 5.times.10.sup.3 PFU of the highly pathogenic clade 2.2 H5N1 virus A/Whooper Swan/Mongolia/244/2005 (WS/05). Cohorts of mice (n=5/group) were sacrificed at 1, 3 and 5 days post infection, lungs harvested, and viral load determined by plaque assay (FIG. 14A). Vaccinated Fitch ferrets (n=6/group) were infected with 1.times.10.sup.6 PFU of the highly pathogenic WS/05 virus. Nasal washes were collected and viral load determined by plaque assay (FIG. 14B). Values represent mean (+/-SEM) viral titer for each group. Significant differences between COBRA and polyvalent vaccines were determined by a two-tailed Student's T test and a p value of less than 0.05 was considered significant (*).
[0025] FIGS. 15A-15B: Histopathology of infected lungs. Vaccinated BALB/c mice (n=15/group) were infected with 5.times.10.sup.3 PFU of the highly pathogenic clade 2.2 H5N1 virus A/Whooper Swan/Mongolia/244/2005 (WS/05). Cohorts of mice (n=5/group) were sacrificed at 3 days post infection and in situ hybridization (ISH) for influenza matrix protein (MP) was performed on sections from paraffin embedded lung tissue (FIG. 15A). Severity of influenza ISH foci was accessed in the bronchi (FIG. 15B). Scoring: 0=no definitive signal; 1=occasional focus; 2=focus in most fields; 3=more than one focus per field.
[0026] FIGS. 16A-16C: Clade 1 challenge. Vaccinated BALB/c mice (n=4/group) were infected with 5.times.10.sup.3 PFU of reassortant virus containing the HA and NA genes from the clade 1 H5N1 virus A/Vietnam/1203/2004 (VN/04). Mice were monitored daily for weight loss (FIG. 16A) and sickness (FIG. 16B). Values represent mean (+/-SD) for each group. An additional cohort of vaccinated mice (n=3/group) were infected and lungs were harvested 3 days post infection for analysis of viral burden (FIG. 16C). Values represent mean (+/-SEM) viral titer for each group.
[0027] FIGS. 17A-17B: Post-challenge cellular immune responses. Vaccinated BALB/c mice (n=3/group) were infected with 5.times.10.sup.3 PFU of reassortant virus containing the HA and NA genes from the clade 1 H5N1 virus A/Vietnam/1203/2004 (VN/04). Mice were sacrificed 6 days post infection, lungs were harvested and the numbers of antibody secreting cells (FIG. 17A) and IFN-.gamma. producing cells (FIG. 17B) were determined by ELISpot assay. Values represent the mean (+/-SEM) spots for each group.
[0028] FIGS. 18A-18B: Passive transfer clade 1 challenge. BALB/c mice (n=10/group) were vaccinated at 0 and 3 weeks with blood collected 14 to 21 days after each vaccination. Serum collected after the second vaccination was pooled for each vaccine group and administered to naive recipient mice (n=5/group). One day after passive transfer, recipient mice were infected with 5.times.10.sup.3 PFU of reassortant virus containing the HA and NA genes from the clade 1 H5N1 virus A/Vietnam/1203/2004 (VN/04). Mice were monitored daily for weight loss (FIG. 18A) and sickness (FIG. 18B). Values represent mean (+/-SD) for each group. Significant differences were determined by two-way ANOVA with Bonferroni's post-test to evaluate differences between vaccines at each day. A p value of less than 0.05 was considered significant (*).
SEQUENCE LISTING
[0029] The nucleic and amino acid sequences listed in the accompanying sequence listing are shown using standard letter abbreviations for nucleotide bases, and three letter code for amino acids, as defined in 37 C.F.R. 1.822. Only one strand of each nucleic acid sequence is shown, but the complementary strand is understood as included by any reference to the displayed strand. The Sequence Listing is submitted as an ASCII text file, created on Jul. 7, 2020, 51.0 KB, which is incorporated by reference herein. In the accompanying sequence listing:
[0030] SEQ ID NOs: 1 and 2 are the nucleotide and amino acid sequences, respectively, of a codon-optimized influenza HA (designated COBRA).
[0031] SEQ ID NOs: 3 and 4 are the nucleotide and amino acid sequences, respectively, of a codon-optimized influenza NA.
[0032] SEQ ID NOs: 5 and 6 are the nucleotide and amino acid sequences, respectively, of a codon-optimized influenza M1.
[0033] SEQ ID NO: 7 is the nucleotide sequence of a plasmid encoding a codon-optimized influenza HA.
[0034] SEQ ID NO: 8 is the nucleotide sequence of a plasmid encoding a codon-optimized influenza NA.
[0035] SEQ ID NO: 9 is the nucleotide sequence of a plasmid encoding a codon-optimized influenza M1.
[0036] SEQ ID NO: 10 is the amino acid sequence of a T cell epitope in H5 HA (HAs33).
[0037] SEQ ID NO: 11 is the amino acid sequence of an ovalbumin T cell epitope (Ova257).
DETAILED DESCRIPTION
I. Abbreviations
[0038] ASC: antibody secreting cell
[0039] DPI: days post infection
[0040] HA: hemagglutinin or hemagglutination assay
[0041] HAI: hemagglutination inhibition
[0042] hRBC: horse red blood cell
[0043] IFU: infectious unit
[0044] LD.sub.50: lethal dose 50
[0045] M1: matrix protein 1
[0046] MN: microneutralization
[0047] MOI: multiplicity of infection
[0048] NA: neuraminidase
[0049] PFU: plaque form unit
[0050] RDE: receptor destroying enzyme
[0051] TCID: tissue culture infectious dose
[0052] tRBC: turkey red blood cell
[0053] VLP: virus-like particle
II. Terms and Methods
[0054] Unless otherwise noted, technical terms are used according to conventional usage. Definitions of common terms in molecular biology may be found in Benjamin Lewin, Genes V, published by Oxford University Press, 1994 (ISBN 0-19-854287-9); Kendrew et al. (eds.), The Encyclopedia of Molecular Biology, published by Blackwell Science Ltd., 1994 (ISBN 0-632-02182-9); and Robert A. Meyers (ed.), Molecular Biology and Biotechnology: a Comprehensive Desk Reference, published by VCH Publishers, Inc., 1995 (ISBN 1-56081-569-8).
[0055] In order to facilitate review of the various embodiments of the disclosure, the following explanations of specific terms are provided:
[0056] Adjuvant: A substance or vehicle that non-specifically enhances the immune response to an antigen. Adjuvants can include a suspension of minerals (alum, aluminum hydroxide, or phosphate) on which antigen is adsorbed; or water-in-oil emulsion in which antigen solution is emulsified in mineral oil (for example, Freund's incomplete adjuvant), sometimes with the inclusion of killed mycobacteria (Freund's complete adjuvant) to further enhance antigenicity.
[0057] Immunostimulatory oligonucleotides (such as those including a CpG motif) can also be used as adjuvants (for example, see U.S. Pat. Nos. 6,194,388; 6,207,646; 6,214,806; 6,218,371; 6,239,116; 6,339,068; 6,406,705; and 6,429,199). Adjuvants also include biological molecules, such as costimulatory molecules. Exemplary biological adjuvants include IL-2, RANTES, GM-CSF, TNF-.alpha., IFN-.gamma., G-CSF, LFA-3, CD72, B7-1, B7-2, OX-40L and 41 BBL.
[0058] Administer: As used herein, administering a composition to a subject means to give, apply or bring the composition into contact with the subject. Administration can be accomplished by any of a number of routes, such as, for example, topical, oral, subcutaneous, intramuscular, intraperitoneal, intravenous, intrathecal and intramuscular.
[0059] Antibody: An immunoglobulin molecule produced by B lymphoid cells with a specific amino acid sequence. Antibodies are evoked in humans or other animals by a specific antigen (immunogen). Antibodies are characterized by reacting specifically with the antigen in some demonstrable way, antibody and antigen each being defined in terms of the other. "Eliciting an antibody response" refers to the ability of an antigen or other molecule to induce the production of antibodies.
[0060] Antigen: A compound, composition, or substance that can stimulate the production of antibodies or a T-cell response in an animal, including compositions that are injected or absorbed into an animal. An antigen reacts with the products of specific humoral or cellular immunity, including those induced by heterologous immunogens. In some embodiments of the disclosed compositions and methods, the antigen is an influenza HA protein.
[0061] Attenuated: In the context of a live virus, the virus is attenuated if its ability to infect a cell or subject and/or its ability to produce disease is reduced (for example, eliminated) compared to a wild-type virus. Typically, an attenuated virus retains at least some capacity to elicit an immune response following administration to an immunocompetent subject. In some cases, an attenuated virus is capable of eliciting a protective immune response without causing any signs or symptoms of infection. In some embodiments, the ability of an attenuated virus to cause disease in a subject is reduced at least about 10%, at least about 25%, at least about 50%, at least about 75% or at least about 90% relative to wild-type virus.
[0062] Clade: Refers to the different categorizations of the known influenza viruses, such as influenza A H5N1 viruses. Viruses in an H5N1 clade are genetically related, but do not share the exact viral genome. There are at least ten different clades of H5N1 subtypes designated in the art: clade 0 clade 1, clade 2, clade 3, clade 4, clade 5, clade 6, clade 7, clade 8 and clade 9 (Abdel-Ghafar et al., N Engl J Med 358:261-273, 2008). Clade 2 is further divided into sub-clades (including clade 2.1, clade 2.2, clade 2.3, clade 2.4 and clade 2.5).
[0063] Codon-optimized: A "codon-optimized" nucleic acid refers to a nucleic acid sequence that has been altered such that the codons are optimal for expression in a particular system (such as a particular species of group of species). For example, a nucleic acid sequence can be optimized for expression in mammalian cells. Codon optimization does not alter the amino acid sequence of the encoded protein.
[0064] Fusion protein: A protein generated by expression of a nucleic acid sequence engineered from nucleic acid sequences encoding at least a portion of two different (heterologous) proteins. To create a fusion protein, the nucleic acid sequences must be in the same reading frame and contain to internal stop codons. For example, a fusion protein includes an influenza HA fused to a heterologous protein.
[0065] Geographical location or geographical region: Refers to preselected divisions of geographical areas of the earth, for example, by continent or other preselected territory or subdivision (e.g., the Middle East, which spans more than one continent). Examples of different geographical regions include countries (e.g., Turkey, Egypt, Iraq, Azerbaijan, China, United States), continents (e.g., Asia, Europe, North America, South America, Oceania, Africa), and recognized geopolitical subdivisions (such as the Middle East).
[0066] Hemagglutinin (HA): An influenza virus surface glycoprotein. HA mediates binding of the virus particle to a host cells and subsequent entry of the virus into the host cell. The nucleotide and amino acid sequences of numerous influenza HA proteins are known in the art and are publically available, such as those deposited with GenBank (see Table 1 for a list of GenBank Accession Nos. of H5N1 HA sequences). HA (along with NA) is one of the two major influenza virus antigenic determinants.
[0067] Immune response: A response of a cell of the immune system, such as a B-cell, T-cell, macrophage or polymorphonucleocyte, to a stimulus such as an antigen or vaccine. An immune response can include any cell of the body involved in a host defense response, including for example, an epithelial cell that secretes an interferon or a cytokine. An immune response includes, but is not limited to, an innate immune response or inflammation. As used herein, a protective immune response refers to an immune response that protects a subject from infection (prevents infection or prevents the development of disease associated with infection). Methods of measuring immune responses are well known in the art and include, for example, measuring proliferation and/or activity of lymphocytes (such as B or T cells), secretion of cytokines or chemokines, inflammation, antibody production and the like.
[0068] Immunogen: A compound, composition, or substance which is capable, under appropriate conditions, of stimulating an immune response, such as the production of antibodies or a T-cell response in an animal, including compositions that are injected or absorbed into an animal. As used herein, as "immunogenic composition" is a composition comprising an immunogen (such as an HA polypeptide).
[0069] Immunize: To render a subject protected from an infectious disease, such as by vaccination.
[0070] Influenza virus: A segmented negative-strand RNA virus that belongs to the Orthomyxoviridae family. There are three types of Influenza viruses, A, B and C. Influenza A viruses infect a wide variety of birds and mammals, including humans, horses, marine mammals, pigs, ferrets, and chickens. In animals, most influenza A viruses cause mild localized infections of the respiratory and intestinal tract. However, highly pathogenic influenza A strains, such as H5N1, cause systemic infections in poultry in which mortality may reach 100%. H5N1 is also referred to as "avian influenza."
[0071] Isolated: An "isolated" biological component (such as a nucleic acid, protein or virus) has been substantially separated or purified away from other biological components (such as cell debris, or other proteins or nucleic acids). Biological components that have been "isolated" include those components purified by standard purification methods. The term also embraces recombinant nucleic acids, proteins or viruses, as well as chemically synthesized nucleic acids or peptides.
[0072] Linker: One or more amino acids that serve as a spacer between two polypeptides of a fusion protein.
[0073] Matrix (M1) protein: An influenza virus structural protein found within the viral envelope. M1 is thought to function in assembly and budding.
[0074] Neuraminidase (NA): An influenza virus membrane glycoprotein. NA is involved in the destruction of the cellular receptor for the viral HA by cleaving terminal sialic acid residues from carbohydrate moieties on the surfaces of infected cells. NA also cleaves sialic acid residues from viral proteins, preventing aggregation of viruses. NA (along with HA) is one of the two major influenza virus antigenic determinants.
[0075] Operably linked: A first nucleic acid sequence is operably linked with a second nucleic acid sequence when the first nucleic acid sequence is placed in a functional relationship with the second nucleic acid sequence. For instance, a promoter is operably linked to a coding sequence if the promoter affects the transcription or expression of the coding sequence. Generally, operably linked DNA sequences are contiguous and, where necessary to join two protein-coding regions, in the same reading frame.
[0076] Optimized influenza HA protein: As used herein, "optimized influenza HA protein" refers to the HA protein consensus sequence generated by sequence alignments of clade 2 H5N1 influenza viruses (as described in Example 1 below). The nucleotide sequence encoding the optimized HA protein was further optimized for expression in mammalian cells via codon-optimization and RNA optimization (such as to increase RNA stability). The optimized influenza HA protein disclosed herein (and set forth herein as SEQ ID NO: 2) is also referred to as "COBRA."
[0077] ORF (open reading frame): A series of nucleotide triplets (codons) coding for amino acids without any termination codons. These sequences are usually translatable into a peptide.
[0078] Outbreak: As used herein, an influenza virus "outbreak" refers to a collection of virus isolates from within a single country in a given year.
[0079] Pharmaceutically acceptable vehicles: The pharmaceutically acceptable carriers (vehicles) useful in this disclosure are conventional. Remington's Pharmaceutical Sciences, by E. W. Martin, Mack Publishing Co., Easton, Pa., 15th Edition (1975), describes compositions and formulations suitable for pharmaceutical delivery of one or more therapeutic compositions, such as one or more influenza vaccines, and additional pharmaceutical agents.
[0080] In general, the nature of the carrier will depend on the particular mode of administration being employed. For instance, parenteral formulations usually comprise injectable fluids that include pharmaceutically and physiologically acceptable fluids such as water, physiological saline, balanced salt solutions, aqueous dextrose, glycerol or the like as a vehicle. For solid compositions (for example, powder, pill, tablet, or capsule forms), conventional non-toxic solid carriers can include, for example, pharmaceutical grades of mannitol, lactose, starch, or magnesium stearate. In addition to biologically-neutral carriers, pharmaceutical compositions to be administered can contain minor amounts of non-toxic auxiliary substances, such as wetting or emulsifying agents, preservatives, and pH buffering agents and the like, for example sodium acetate or sorbitan monolaurate.
[0081] Plasmid: A circular nucleic acid molecule capable of autonomous replication in a host cell.
[0082] Polypeptide: A polymer in which the monomers are amino acid residues which are joined together through amide bonds. When the amino acids are alpha-amino acids, either the L-optical isomer or the D-optical isomer can be used. The terms "polypeptide" or "protein" as used herein are intended to encompass any amino acid sequence and include modified sequences such as glycoproteins. The term "polypeptide" is specifically intended to cover naturally occurring proteins, as well as those which are recombinantly or synthetically produced. The term "residue" or "amino acid residue" includes reference to an amino acid that is incorporated into a protein, polypeptide, or peptide.
[0083] Conservative amino acid substitutions are those substitutions that, when made, least interfere with the properties of the original protein, that is, the structure and especially the function of the protein is conserved and not significantly changed by such substitutions. Examples of conservative substitutions are shown below.
TABLE-US-00001 Original Residue Conservative Substitutions Ala Ser Arg Lys Asn Gln, His Asp Glu Cys Ser Gln Asn Glu Asp His Asn; Gln Ile Leu, Val Leu Ile; Val Lys Arg; Gln; Glu Met Leu; Ile Phe Met; Leu; Tyr Ser Thr Thr Ser Trp Tyr Tyr Trp; Phe Val Ile; Leu
[0084] Conservative substitutions generally maintain (a) the structure of the polypeptide backbone in the area of the substitution, for example, as a sheet or helical conformation, (b) the charge or hydrophobicity of the molecule at the target site, or (c) the bulk of the side chain.
[0085] The substitutions which in general are expected to produce the greatest changes in protein properties will be non-conservative, for instance changes in which (a) a hydrophilic residue, for example, seryl or threonyl, is substituted for (or by) a hydrophobic residue, for example, leucyl, isoleucyl, phenylalanyl, valyl or alanyl; (b) a cysteine or proline is substituted for (or by) any other residue; (c) a residue having an electropositive side chain, for example, lysyl, arginyl, or histadyl, is substituted for (or by) an electronegative residue, for example, glutamyl or aspartyl; or (d) a residue having a bulky side chain, for example, phenylalanine, is substituted for (or by) one not having a side chain, for example, glycine.
[0086] Preventing, treating or ameliorating a disease: "Preventing" a disease refers to inhibiting the full development of a disease. "Treating" refers to a therapeutic intervention that ameliorates a sign or symptom of a disease or pathological condition after it has begun to develop. "Ameliorating" refers to the reduction in the number or severity of signs or symptoms of a disease.
[0087] Promoter: An array of nucleic acid control sequences which direct transcription of a nucleic acid. A promoter includes necessary nucleic acid sequences near the start site of transcription. A promoter also optionally includes distal enhancer or repressor elements. A "constitutive promoter" is a promoter that is continuously active and is not subject to regulation by external signals or molecules. In contrast, the activity of an "inducible promoter" is regulated by an external signal or molecule (for example, a transcription factor). In some embodiments herein, the promoter is a CMV promoter.
[0088] Purified: The term "purified" does not require absolute purity; rather, it is intended as a relative term. Thus, for example, a purified peptide, protein, virus, or other active compound is one that is isolated in whole or in part from naturally associated proteins and other contaminants. In certain embodiments, the term "substantially purified" refers to a peptide, protein, virus or other active compound that has been isolated from a cell, cell culture medium, or other crude preparation and subjected to fractionation to remove various components of the initial preparation, such as proteins, cellular debris, and other components.
[0089] Recombinant: A recombinant nucleic acid, protein or virus is one that has a sequence that is not naturally occurring or has a sequence that is made by an artificial combination of two otherwise separated segments of sequence. This artificial combination is often accomplished by chemical synthesis or by the artificial manipulation of isolated segments of nucleic acids, for example, by genetic engineering techniques.
[0090] Sequence identity: The similarity between amino acid or nucleic acid sequences is expressed in terms of the similarity between the sequences, otherwise referred to as sequence identity. Sequence identity is frequently measured in terms of percentage identity (or similarity or homology); the higher the percentage, the more similar the two sequences are. Homologs or variants of a given gene or protein will possess a relatively high degree of sequence identity when aligned using standard methods.
[0091] Methods of alignment of sequences for comparison are well known in the art. Various programs and alignment algorithms are described in: Smith and Waterman, Adv. Appl. Math. 2:482, 1981; Needleman and Wunsch, J. Mol. Biol. 48:443, 1970; Pearson and Lipman, Proc. Natl. Acad. Sci. U.S.A. 85:2444, 1988; Higgins and Sharp, Gene 73:237-244, 1988; Higgins and Sharp, CABIOS 5:151-153, 1989; Corpet et al., Nucleic Acids Research 16:10881-10890, 1988; and Pearson and Lipman, Proc. Natl. Acad. Sci. U.S.A. 85:2444, 1988. Altschul et al., Nature Genet. 6:119-129, 1994.
[0092] The NCBI Basic Local Alignment Search Tool (BLAST.TM.) (Altschul et al., J. Mol. Biol. 215:403-410, 1990) is available from several sources, including the National Center for Biotechnology Information (NCBI, Bethesda, Md.) and on the Internet, for use in connection with the sequence analysis programs blastp, blastn, blastx, tblastn and tblastx.
[0093] Subject: Living multi-cellular vertebrate organisms, a category that includes both human and non-human mammals, such as non-human primates. In one example, a subject is one who is infected with H5N1 or is susceptible to such infection.
[0094] Therapeutically effective amount: A quantity of a specified agent sufficient to achieve a desired effect in a subject being treated with that agent. For example, this may be the amount of an influenza virus vaccine useful for eliciting an immune response in a subject and/or for preventing infection by influenza virus. Ideally, in the context of the present disclosure, a therapeutically effective amount of an influenza vaccine is an amount sufficient to increase resistance to, prevent, ameliorate, and/or treat infection caused by influenza virus in a subject without causing a substantial cytotoxic effect in the subject. The effective amount of an influenza vaccine useful for increasing resistance to, preventing, ameliorating, and/or treating infection in a subject will be dependent on, for example, the subject being treated, the manner of administration of the therapeutic composition and other factors.
[0095] Transformed: A transformed cell is a cell into which has been introduced a nucleic acid molecule by molecular biology techniques. As used herein, the term transformation encompasses all techniques by which a nucleic acid molecule might be introduced into such a cell, including transfection with viral vectors, transformation with plasmid vectors, and introduction of naked DNA by electroporation, lipofection, and particle gun acceleration.
[0096] Vaccine: A preparation of immunogenic material capable of stimulating an immune response, administered for the prevention, amelioration, or treatment of disease, such as an infectious disease. The immunogenic material may include, for example, attenuated or killed microorganisms (such as attenuated viruses), or antigenic proteins, peptides or DNA derived from them. Vaccines may elicit both prophylactic (preventative) and therapeutic responses. Methods of administration vary according to the vaccine, but may include inoculation, ingestion, inhalation or other forms of administration. Inoculations can be delivered by any of a number of routes, including parenteral, such as intravenous, subcutaneous or intramuscular. Vaccines may be administered with an adjuvant to boost the immune response.
[0097] Vector: A vector is a nucleic acid molecule allowing insertion of foreign nucleic acid without disrupting the ability of the vector to replicate and/or integrate in a host cell. A vector can include nucleic acid sequences that permit it to replicate in a host cell, such as an origin of replication. An insertional vector is capable of inserting itself into a host nucleic acid. A vector can also include one or more selectable marker genes and other genetic elements. An expression vector is a vector that contains the necessary regulatory sequences to allow transcription and translation of inserted gene or genes. In some embodiments of the present disclosure, the vector encodes an influenza HA, NA or M1 protein. In some embodiments, the vector is the pTR600 expression vector (U.S. Patent Application Publication No. 2002/0106798; Ross et al., Nat Immunol. 1(2):102-103, 2000; Green et al., Vaccine 20:242-248, 2001).
[0098] Virus-like particle (VLP): Virus particles made up of one of more viral structural proteins, but lacking the viral genome. Because VLPs lack a viral genome, they are non-infectious. In addition, VLPs can often be produced by heterologous expression and can be easily purified. Most VLPs comprise at least a viral core protein that drives budding and release of particles from a host cell. One example of such a core protein is influenza M1. In some embodiments herein, an influenza VLP comprises the HA, NA and M1 proteins. As described herein, influenza VLPs can be produced by transfection of host cells with plasmids encoding the HA, NA and M1 proteins. After incubation of the transfected cells for an appropriate time to allow for protein expression (such as for approximately 72 hours), VLPs can be isolated from cell culture supernatants. Example 1 provides an exemplary protocol for purifying influenza VLPs from cell supernatants. In this example, VLPs are isolated by low speed centrifugation (to remove cell debris), vacuum filtration and ultracentrifugation through 20% glycerol.
[0099] Unless otherwise explained, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this disclosure belongs. The singular terms "a," "an," and "the" include plural referents unless context clearly indicates otherwise. Similarly, the word "or" is intended to include "and" unless the context clearly indicates otherwise. Hence "comprising A or B" means including A, or B, or A and B. It is further to be understood that all base sizes or amino acid sizes, and all molecular weight or molecular mass values, given for nucleic acids or polypeptides are approximate, and are provided for description. Although methods and materials similar or equivalent to those described herein can be used in the practice or testing of the present disclosure, suitable methods and materials are described below. All publications, patent applications, patents, and other references mentioned herein are incorporated by reference in their entirety. All GenBank Accession numbers are incorporated by reference herein as they appeared in the database on Sep. 9, 2010. In case of conflict, the present specification, including explanations of terms, will control. In addition, the materials, methods, and examples are illustrative only and not intended to be limiting.
III. Overview of Several Embodiments
[0100] Disclosed herein is the development of a computationally optimized influenza HA protein that elicits broadly reactive immune responses to H5N1 influenza virus isolates, such as the isolates listed in Table 1. The optimized HA protein was developed through a series of HA protein alignments, and subsequent generation of consensus sequences, for clade 2 H5N1 influenza virus isolates (described in detail in Example 1 below; see also FIG. 1). The final consensus HA amino acid sequence was reverse translated and optimized for expression in mammalian cells. Optimization of the nucleic acid sequence included optimization of the codons for expression in mammalian cells and RNA optimization (such as RNA stability). The optimized HA coding sequence is set forth herein as SEQ ID NO: 1, and the optimized HA protein sequence is set forth herein as SEQ ID NO: 2.
[0101] Thus, provided herein is an isolated nucleic acid molecule comprising a nucleotide sequence encoding an influenza HA polypeptide. In some embodiments, the nucleotide sequence encoding the HA polypeptide is at least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 1.
[0102] In some examples, the nucleotide sequence encoding the influenza HA polypeptide that is at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical to SEQ ID NO: 1 lacks the start codon (nucleotides 1-3 of SEQ ID NO: 1), encoding a N-terminal methionine. In particular examples, the nucleotide sequence encoding the influenza HA polypeptide is at least 94% identical to nucleotides 4-1707 of SEQ ID NO: 1. In other examples, the nucleotide sequence encoding the HA polypeptide comprises or consists of nucleotides 4-1707 of SEQ ID NO: 1.
[0103] In some examples, the nucleotide sequence encoding the HA polypeptide comprises SEQ ID NO: 1. In particular examples, the nucleotide sequence encoding the HA polypeptide consists of SEQ ID NO: 1. Also provided herein are influenza HA polypeptides encoded by the disclosed nucleic acid molecules.
[0104] Further provided are vectors containing a nucleotide sequence encoding an optimized HA polypeptide. In some embodiments of the vectors provided herein, the nucleotide sequence encoding the HA polypeptide is at least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 1. In some examples, the vector further includes a promoter operably linked to the nucleotide sequence encoding the HA polypeptide. In particular examples, the promoter is a cytomegalovirus (CMV) promoter. In some embodiments, the nucleotide sequence of the vector is at least 85%, at least 90%, at least 95%, at least 98% or at least 99% identical to the nucleotide sequence of SEQ ID NO: 7. In some examples, the nucleotide sequence of the vector comprises the nucleotide sequence of SEQ ID NO: 7. In particular examples, the nucleotide sequence of the vector consists of the nucleotide sequence of SEQ ID NO: 7.
[0105] Also provided herein are influenza HA polypeptides produced by transfecting a host cell with a vector provided herein under conditions sufficient to allow for expression of the HA polypeptide. Isolated cells containing the disclosed vectors are also provided.
[0106] Also provided herein are optimized influenza HA polypeptides. In some embodiments, the amino acid sequence of the polypeptide is at least 99% identical to SEQ ID NO: 2. In some examples, the amino acid sequence of the influenza HA polypeptide that is at least 99% identical to SEQ ID NO: 2 lacks the N-terminal methionine residue. In particular examples, the amino acid sequence of the influenza HA polypeptide is at least 99% identical to amino acids 2-568 of SEQ ID NO: 2. In other examples, the amino acid sequence of the HA polypeptides comprises or consists of amino acids 2-568 of SEQ ID NO: 2.
[0107] In some examples, the amino acid sequence of the polypeptide comprises the amino acid sequence of SEQ ID NO: 2. In particular examples, the amino acid sequence of the polypeptide consists of the amino acid sequence of SEQ ID NO: 2. Fusion proteins comprising the influenza HA polypeptides disclosed herein are also provided. The influenza HA polypeptide can be fused to any heterologous amino acid sequence to form the fusion protein.
[0108] Further provided herein are influenza virus-like particles (VLPs) containing an optimized influenza HA protein disclosed herein. In some embodiments, the HA protein of the VLP is at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99% or 100% identical to SEQ ID NO: 2. The influenza VLPs can further include any additional influenza proteins necessary to form the virus particle. In some embodiments, the influenza VLPs further include influenza neuraminidase (NA) protein, influenza matrix (M1) protein, or both.
[0109] In some embodiments of the influenza VLPs, the amino acid sequence of the influenza NA protein is at least 85%, at least 90%, at least 95%, at least 98% or at least 99% identical to SEQ ID NO: 4. In some examples, the amino acid sequence of the influenza NA protein comprises SEQ ID NO: 4. In particular examples, the amino acid sequence of the influenza NA protein consists of SEQ ID NO: 4. In some embodiments, the amino acid sequence of the influenza M1 protein is at least 85%, at least 90%, at least 95%, at least 98% or at least 99% identical to SEQ ID NO: 6. In some examples, the amino acid sequence of the influenza M1 protein comprises SEQ ID NO: 6. In particular examples, the amino acid sequence of the influenza M1 protein consists of SEQ ID NO: 6.
[0110] Also provided is an influenza VLP containing an influenza HA polypeptide as described herein, produced by transfecting a host cell with a vector encoding the HA polypeptide, a vector encoding an influenza NA protein and a vector encoding an influenza M1 protein, under conditions sufficient to allow for expression of the HA, M1 and NA proteins.
[0111] The vectors used to express the HA, NA and M1 proteins can be any suitable expression vectors known in the art. The vectors can be, for example, mammalian expression vectors, or viral vectors. In some embodiments, the vector is the pTR600 expression vector (U.S. Patent Application Publication No. 2002/0106798, herein incorporated by reference; Ross et al., Nat Immunol. 1(2):102-103, 2000; Green et al., Vaccine 20:242-248, 2001).
[0112] In some embodiments, the nucleotide sequence of the vector encoding the HA polypeptide is at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 7. In some examples, the nucleotide sequence of the vector encoding the HA polypeptide comprises SEQ ID NO: 7. In particular examples, the nucleotide sequence of the vector encoding the HA polypeptide consists of SEQ ID NO: 7.
[0113] In some embodiments, the nucleotide sequence of the vector encoding the NA protein is at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 8. In some examples, the nucleotide sequence of the vector encoding the NA protein comprises SEQ ID NO: 8. In particular examples, the nucleotide sequence of the vector encoding the NA protein consists of SEQ ID NO: 8.
[0114] In some embodiments, the nucleotide sequence of the vector encoding the M1 protein is at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 9. In some examples, the nucleotide sequence of the vector encoding the M1 protein comprises SEQ ID NO: 9. In particular examples, the nucleotide sequence of the vector encoding the M1 protein consists of SEQ ID NO: 9.
[0115] Collections of plasmids are also provided herein. In some embodiments, the collection of plasmids includes a plasmid encoding an influenza NA, a plasmid encoding an influenza MA, and a plasmid encoding the optimized HA protein disclosed herein. In some embodiments, the nucleotide sequence encoding the codon-optimized influenza HA of the HA-encoding plasmid is at least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 1. Also provided are kits comprising the collection of plasmids.
[0116] In some embodiments of the collections of plasmids, the influenza NA is codon-optimized and/or the influenza M1 is codon-optimized. In some examples, the nucleotide sequence encoding the codon-optimized influenza NA is at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 3. In particular examples, the nucleotide sequence encoding the codon-optimized influenza NA comprises, or consists of, SEQ ID NO: 3. In some examples, the nucleotide sequence encoding the codon-optimized influenza M1 is at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical to SEQ ID NO: 5. In particular examples, the nucleotide sequence encoding the codon-optimized influenza M1 comprises, or consists of, SEQ ID NO: 5.
[0117] In one non-limiting example, the plasmid encoding influenza NA comprises SEQ ID NO: 8; the plasmid encoding influenza M1 comprises SEQ ID NO: 9; and the plasmid encoding influenza HA comprises SEQ ID NO: 10.
[0118] In some embodiments, transfection of the collection of plasmids into host cells under conditions sufficient to allow for expression of the HA, NA and M1 proteins produces influenza VLPs comprising the HA, NA and M1 proteins.
[0119] Also provided herein are compositions comprising an optimized influenza HA protein as disclosed herein, or a fusion protein or VLP comprising the optimized influenza HA protein. In some embodiments, the compositions further comprise a pharmaceutically acceptable carrier and/or an adjuvant. For example, the adjuvant can be alum, Freund's complete adjuvant, a biological adjuvant or immunostimulatory oligonucleotides (such as CpG oligonucleotides).
[0120] Further provided is a method of eliciting an immune response to influenza virus in a subject by administering an influenza HA protein disclosed herein, fusion proteins containing the influenza HA, or VLPs containing the influenza HA, as disclosed herein. In some embodiments, the influenza virus is an H5N1 influenza virus. In some embodiments, the HA protein, HA fusion protein or VLP can be administered using any suitable route of administration, such as, for example, intramuscular. In some embodiments, the HA protein, fusion protein or VLP is administered as a composition further comprising a pharmaceutically acceptable carrier and/or an adjuvant. For example, the adjuvant can be alum, Freund's complete adjuvant, a biological adjuvant or immunostimulatory oligonucleotides (such as CpG oligonucleotides).
[0121] Also provided is a method of immunizing a subject against influenza virus by administering to the subject VLPs containing the optimized influenza HA protein disclosed herein, or administering a composition thereof. In some embodiments of the method, the composition further comprises a pharmaceutically acceptable carrier and/or an adjuvant. For example, the adjuvant can be alum, Freund's complete adjuvant, a biological adjuvant or immunostimulatory oligonucleotides (such as CpG oligonucleotides). In some embodiments, the VLPs (or compositions thereof) are administered intramuscularly.
[0122] In some embodiments of the methods of eliciting an immune response or immunizing a subject, the subject is administered at least 1 .mu.g of the VLPs containing the optimized HA protein, such as at least 5 .mu.g, at least 10 .mu.g, at least 15 .mu.g, at least 20 .mu.g, at least 25 .mu.g, at least 30 .mu.g, at least 40 .mu.g or at least 50 .mu.g of the VLPs containing the optimized HA protein, for example about 1 to about 50 .mu.g or about 1 to about 25 .mu.g of the VLPs containing the optimized HA protein. In particular examples, the subject is administered about 5 to about 20 .mu.g of the VLPs, or about 10 to about 15 .mu.g of the VLPs. In one specific non-limiting example, the subject is administered about 15 .mu.g of the VLPs. However, one of skill in the art is capable of determining a therapeutically effective amount (for example an amount that provides protection against H5N1 influenza virus infection) of VLPs to administer to a subject.
[0123] It is disclosed herein that administration of VLPs comprising the COBRA HA disclosed herein (SEQ ID NO: 2) elicits protective levels of HAI antibodies to a number of representative clade 2 isolates and provides complete protection against lethal challenge with a clade 2.2 H5N1 virus. In some embodiments, administration of VLPs containing an optimized influenza HA results in production of high HAI titers (.gtoreq.1:40) to H5N1 clade 1, clade 2.1, clade 2.2 and clade 2.3 isolates. In some examples, the VLPs containing an optimized influenza HA elicit high HAI titers against clade 1 and/or clade 7 viruses. The VLPs containing an optimized influenza HA disclosed herein elicit a broader immune response (e.g., elicit neutralizing antibodies to a broader range is H5N1 virus isolates, such as those from clade 1, sub-clades of clade 2, and clade 7) than a polyvalent influenza virus vaccine.
[0124] Also provided herein is a method of optimizing an influenza protein sequence to elicit broadly reactive immune responses in a subject. In the context of the present disclosure, "broadly reactive" means the protein sequence elicits an immune response in a subject that is sufficient to inhibit, neutralize or prevent infection of a broad range of influenza viruses (such as most or all influenza viruses within a specific subtype). In some embodiments, the influenza protein is influenza HA or influenza NA. In one example, the optimized influenza protein is capable of eliciting a protective immune response against most or all known H5N1 influenza virus isolates (such as those listed in Table 1), such as about 80%, about 85%, about 90% or about 95% of known H5N1 influenza virus isolates.
[0125] In some embodiments, the method of optimizing an influenza protein sequence includes obtaining the amino acid sequences of a group of influenza virus isolates. For example, the group can consist of influenza virus isolates from a specific subtype (such as, for example, H5N1 or H1N1), and/or from one or more clades/sub-clades of a specific influenza subtype (for example, one or more of clades/sub-clades 0, 1, 2.1, 2.2, 2.3, 2.4, 3, 4, 5, 6, 7, 8 and 9 of H5N1). Amino acid sequences of the group of influenza viruses are first organized by clade or sub-clade and then by geographic location within each clade or sub-clade. The amino acid sequences for each geographic location are aligned to generate a primary consensus sequence for each geographical region. Grouping virus isolates by geographical region controls for single outbreak dominance and incomplete reporting and sequencing. The primary consensus sequence can be generated, for example, by multiple alignment analysis using AlignX (Vector NTI), or by any other method known in the art. The primary geographically-based consensus sequences for each clade or sub-clade are then aligned, and a secondary consensus sequence is generated for each clade or sub-clade. The secondary consensus sequences for each clade or sub-clade are then aligned to generate the optimized, broadly reactive, consensus sequence (see FIG. 1). In some embodiments, the optimized influenza virus polypeptide sequence is further optimized for expression in mammalian cells. In some examples, optimization includes reverse translation of the optimized influenza virus polypeptide sequence to generate a coding sequence, followed by codon-optimization and/or optimization of the RNA (such as for stability).
[0126] In one non-limiting example, the method of optimization includes: (i) obtaining the amino acid sequences of the polypeptide from a group of influenza virus isolates, wherein the influenza virus isolates are from the same subtype; (ii) organizing the amino acid sequences of the polypeptide from the group of influenza virus isolates by clade or sub-clade and then by geographical region within each clade or sub-clade; (iii) aligning the amino acid sequences within each geographical region to generate primary consensus sequences, wherein each geographic region is represented by a primary consensus sequence; (iv) aligning the primary consensus sequences to generate secondary consensus sequences, wherein each clade or sub-clade is represented by a secondary consensus sequence; and (v) aligning the secondary consensus sequences to generate the optimized influenza virus polypeptide sequence. In some cases, the method further includes (i) reverse translating the optimized influenza virus polypeptide sequence to generate a coding sequence; and (ii) optimizing the coding sequence for expression in mammalian cells.
[0127] In an alternative embodiment, the primary consensus sequence is obtained by aligning influenza protein sequences (such as HA or NA sequences) from viral isolates from a single outbreak (a collection of influenza virus isolates within a single country within a given year). Thus, in one non-limiting example, the method of optimization includes: (i) obtaining the amino acid sequences of the polypeptide from a group of influenza virus isolates, wherein the influenza virus isolates are from the same subtype; (ii) organizing the amino acid sequences of the polypeptide from the group of influenza virus isolates by clade or sub-clade and then by outbreak; (iii) aligning the amino acid sequences within each outbreak to generate primary consensus sequences, wherein each outbreak is represented by a primary consensus sequence; (iv) aligning the primary consensus sequences to generate secondary consensus sequences, wherein each clade or sub-clade is represented by a secondary consensus sequence; and (v) aligning the secondary consensus sequences to generate the optimized influenza virus polypeptide sequence. In some cases, the method further includes (i) reverse translating the optimized influenza virus polypeptide sequence to generate a coding sequence; and (ii) optimizing the coding sequence for expression in mammalian cells.
VI. Influenza
[0128] Influenza viruses are segmented negative-strand RNA viruses that belong to the Orthomyxoviridae family. There are three types of Influenza viruses, A, B and C. Influenza A viruses infect a wide variety of birds and mammals, including humans, horses, marine mammals, pigs, ferrets, and chickens. In animals, most influenza A viruses cause mild localized infections of the respiratory and intestinal tract. However, highly pathogenic influenza A strains, such as H5N1, cause systemic infections in poultry in which mortality may reach 100%. Animals infected with influenza A often act as a reservoir for the influenza viruses and certain subtypes have been shown to cross the species barrier to humans.
[0129] Influenza A viruses can be classified into subtypes based on allelic variations in antigenic regions of two genes that encode surface glycoproteins, namely, hemagglutinin (HA) and neuraminidase (NA) which are required for viral attachment and cellular release. Currently, sixteen subtypes of HA (H1-H16) and nine NA (N1-N9) antigenic variants are known for influenza A virus. Previously, only three subtypes were known to circulate in humans (H1N1, H1N2, and H3N2). However, in recent years, the pathogenic H5N1 subtype of avian influenza A has been reported to cross the species barrier and infect humans as documented in Hong Kong in 1997 and 2003, leading to the death of several patients.
[0130] In humans, the avian influenza virus infects cells of the respiratory tract as well as the intestinal tract, liver, spleen, kidneys and other organs. Symptoms of avian influenza infection include fever, respiratory difficulties including shortness of breath and cough, lymphopenia, diarrhea and difficulties regulating blood sugar levels. In contrast to seasonal influenza, the group most at risk is healthy adults which make up the bulk of the population. Due to the high pathogenicity of certain avian influenza A subtypes, particularly H5N1, and their demonstrated ability to cross over to infect humans, there is a significant economic and public health risk associated with these viral strains, including a real epidemic and pandemic threat. Currently, no effective vaccines for H5N1 infection are available.
[0131] The influenza A virus genome encodes nine structural proteins and one nonstructural (NS1) protein with regulatory functions. The influenza virus segmented genome contains eight negative-sense RNA (nsRNA) gene segments (PB2, PB1, PA, NP, M, NS, HA and NA) that encode at least ten polypeptides, including RNA-directed RNA polymerase proteins (PB2, PB1 and PA), nucleoprotein (NP), neuraminidase (NA), hemagglutinin (subunits HA1 and HA2), the matrix proteins (M1 and M2) and the non-structural proteins (NS1 and NS2) (Krug et al., In "The Influenza Viruses," R. M. Krug, ed., Plenum Press, N.Y., 1989, pp. 89 152).
[0132] Influenza virus' ability to cause widespread disease is due to its ability to evade the immune system by undergoing antigenic change, which is believed to occur when a host is infected simultaneously with both an animal influenza virus and a human influenza virus. During mutation and reassortment in the host, the virus may incorporate an HA and/or NA surface protein gene from another virus into its genome, thereby producing a new influenza subtype and evading the immune system.
[0133] HA is a viral surface glycoprotein generally comprising approximately 560 amino acids and representing 25% of the total virus protein. It is responsible for adhesion of the viral particle to, and its penetration into, a host cell in the early stages of infection. Cleavage of the virus HA0 precursor into the HA1 and HA2 sub-fragments is a necessary step in order for the virus to infect a cell. Thus, cleavage is required in order to convert new virus particles in a host cell into virions capable of infecting new cells. Cleavage is known to occur during transport of the integral HA0 membrane protein from the endoplasmic reticulum of the infected cell to the plasma membrane. In the course of transport, hemagglutinin undergoes a series of co- and post-translational modifications including proteolytic cleavage of the precursor HA into the amino-terminal fragment HAI and the carboxy terminal HA2. One of the primary difficulties in growing influenza strains in primary tissue culture or established cell lines arises from the requirement for proteolytic cleavage activation of the influenza hemagglutinin in the host cell.
[0134] Although it is known that an uncleaved HA can mediate attachment of the virus to its neuraminic acid-containing receptors on a cell surface, it is not capable of the next step in the infectious cycle, which is fusion. It has been reported that exposure of the hydrophobic amino terminus of HA2 by cleavage is required so that it can be inserted into the target cell, thereby forming a bridge between virus and target cell membrane. This process is followed by fusion of the two membranes and entry of the virus into the target cell.
[0135] Proteolytic activation of HA involves cleavage at an arginine residue by a trypsin-like endoprotease, which is often an intracellular enzyme that is calcium dependent and has a neutral pH optimum. Since the activating proteases are cellular enzymes, the infected cell type determines whether the HA is cleaved. The HA of the mammalian influenza viruses and the nonpathogenic avian influenza viruses are susceptible to proteolytic cleavage only in a restricted number of cell types. On the other hand, HA of pathogenic avian viruses among the H5 and H7 subtypes are cleaved by proteases present in a broad range of different host cells. Thus, there are differences in host range resulting from differences in hemagglutinin cleavability which are correlated with the pathogenic properties of the virus.
[0136] Neuraminidase (NA) is a second membrane glycoprotein of the influenza viruses. The presence of viral NA has been shown to be important for generating a multi-faceted protective immune response against an infecting virus. For most influenza A viruses, NA is 413 amino acid in length, and is encoded by a gene of 1413 nucleotides. Nine different NA subtypes have been identified in influenza viruses (N1, N2, N3, N4, N5, N6, N7, N8 and N9), all of which have been found among wild birds. NA is involved in the destruction of the cellular receptor for the viral HA by cleaving terminal neuraminic acid (also called sialic acid) residues from carbohydrate moieties on the surfaces of infected cells. NA also cleaves sialic acid residues from viral proteins, preventing aggregation of viruses. Using this mechanism, it is hypothesized that NA facilitates release of viral progeny by preventing newly formed viral particles from accumulating along the cell membrane, as well as by promoting transportation of the virus through the mucus present on the mucosal surface. NA is an important antigenic determinant that is subject to antigenic variation.
[0137] In addition to the surface proteins HA and NA, influenza virus comprises six additional internal genes, which give rise to eight different proteins, including polymerase genes PB1, PB2 and PA, matrix proteins M1 and M2, nucleoprotein (NP), and non-structural proteins NS1 and NS2 (Horimoto et al., Clin Microbiol Rev. 14(1):129-149, 2001).
[0138] In order to be packaged into progeny virions, viral RNA is transported from the nucleus as a ribonucleoprotein (RNP) complex composed of the three influenza virus polymerase proteins, the nucleoprotein (NP), and the viral RNA, in association with the influenza virus matrix 1 (M1) protein and nuclear export protein (Marsh et al., J Virol, 82:2295-2304, 2008). The M1 protein that lies within the envelope is thought to function in assembly and budding. A limited number of M2 proteins are integrated into the virions (Zebedee, J. Virol. 62:2762-2772, 1988). They form tetramers having H+ ion channel activity, and when activated by the low pH in endosomes, acidify the inside of the virion, facilitating its uncoating (Pinto et al., Cell 69:517-528, 1992). Amantadine is an anti-influenza drug that prevents viral infection by interfering with M2 ion channel activity, thus inhibiting virus uncoating.
[0139] NS1, a nonstructural protein, has multiple functions, including regulation of splicing and nuclear export of cellular mRNAs as well as stimulation of translation. The major function of NS1 seems to be to counteract the interferon activity of the host, since an NS1 knockout virus was viable although it grew less efficiently than the parent virus in interferon-nondefective cells (Garcia-Sastre, Virology 252:324-330, 1998).
[0140] NS2 has been detected in virus particles (Richardson et al., Arch. Virol. 116:69-80, 1991; Yasuda et al., Virology 196:249-255, 1993). The average number of NS2 proteins in a virus particle was estimated to be 130-200 molecules. An in vitro binding assay shows direct protein-protein contact between M1 and NS2. NS2-M1 complexes have also been detected by immunoprecipitation in virus-infected cell lysates. The NS2 protein is thought to play a role in the export of RNP from the nucleus through interaction with M1 protein (Ward et al., Arch. Virol. 140:2067-2073, 1995).
V. Influenza Proteins, VLPs and Administration Thereof
[0141] Optimized influenza HA polypeptides and influenza VLPs comprising an optimized HA (such as the HA having the sequence set forth as SEQ ID NO: 2) are provided herein. The optimized HA polypeptides can be administered to elicit an immune response against influenza. In particular examples, the optimized HA polypeptides are administered as part of a VLP.
[0142] The influenza VLPs are made up of the HA, NA and M1 proteins. The production of influenza VLPs has been described in the art and is within the abilities of one of ordinary skill in the art. As described herein, influenza VLPs can be produced by transfection of host cells with plasmids encoding the HA, NA and M1 proteins. After incubation of the transfected cells for an appropriate time to allow for protein expression (such as for approximately 72 hours), VLPs can be isolated from cell culture supernatants. Example 1 below provides an exemplary protocol for purifying influenza VLPs from cell supernatants. In this example, VLPs are isolated by low speed centrifugation (to remove cell debris), vacuum filtration and ultracentrifugation through 20% glycerol.
[0143] The influenza VLPs disclosed herein can be used as influenza vaccines to elicit a protective immune response against H5N1 influenza viruses.
[0144] Influenza HA polypeptides and VLPs comprising HA polypeptides, or compositions thereof, can be administered to a subject by any of the routes normally used for introducing recombinant virus into a subject. Methods of administration include, but are not limited to, intradermal, intramuscular, intraperitoneal, parenteral, intravenous, subcutaneous, vaginal, rectal, intranasal, inhalation or oral. Parenteral administration, such as subcutaneous, intravenous or intramuscular administration, is generally achieved by injection. Injectables can be prepared in conventional forms, either as liquid solutions or suspensions, solid forms suitable for solution or suspension in liquid prior to injection, or as emulsions. Injection solutions and suspensions can be prepared from sterile powders, granules, and tablets of the kind previously described. Administration can be systemic or local.
[0145] Influenza VLPs, or compositions thereof, are administered in any suitable manner, such as with pharmaceutically acceptable carriers. Pharmaceutically acceptable carriers are determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions of the present disclosure.
[0146] Preparations for parenteral administration include sterile aqueous or non-aqueous solutions, suspensions, and emulsions. Examples of non-aqueous solvents are propylene glycol, polyethylene glycol, vegetable oils such as olive oil, and injectable organic esters such as ethyl oleate. Aqueous carriers include water, alcoholic/aqueous solutions, emulsions or suspensions, including saline and buffered media. Parenteral vehicles include sodium chloride solution, Ringer's dextrose, dextrose and sodium chloride, lactated Ringer's, or fixed oils. Intravenous vehicles include fluid and nutrient replenishers, electrolyte replenishers (such as those based on Ringer's dextrose), and the like. Preservatives and other additives may also be present such as, for example, antimicrobials, anti-oxidants, chelating agents, and inert gases and the like.
[0147] Some of the compositions may potentially be administered as a pharmaceutically acceptable acid- or base-addition salt, formed by reaction with inorganic acids such as hydrochloric acid, hydrobromic acid, perchloric acid, nitric acid, thiocyanic acid, sulfuric acid, and phosphoric acid, and organic acids such as formic acid, acetic acid, propionic acid, glycolic acid, lactic acid, pyruvic acid, oxalic acid, malonic acid, succinic acid, maleic acid, and fumaric acid, or by reaction with an inorganic base such as sodium hydroxide, ammonium hydroxide, potassium hydroxide, and organic bases such as mono-, di-, trialkyl and aryl amines and substituted ethanolamines.
[0148] Administration can be accomplished by single or multiple doses. The dose administered to a subject in the context of the present disclosure should be sufficient to induce a beneficial therapeutic response in a subject over time, or to inhibit or prevent H5N1 influenza virus infection. The dose required will vary from subject to subject depending on the species, age, weight and general condition of the subject, the severity of the infection being treated, the particular composition being used and its mode of administration. An appropriate dose can be determined by one of ordinary skill in the art using only routine experimentation.
[0149] Provided herein are pharmaceutical compositions which include a therapeutically effective amount of the influenza VLPs alone or in combination with a pharmaceutically acceptable carrier. Pharmaceutically acceptable carriers include, but are not limited to, saline, buffered saline, dextrose, water, glycerol, ethanol, and combinations thereof. The carrier and composition can be sterile, and the formulation suits the mode of administration. The composition can also contain minor amounts of wetting or emulsifying agents, or pH buffering agents. The composition can be a liquid solution, suspension, emulsion, tablet, pill, capsule, sustained release formulation, or powder. The composition can be formulated as a suppository, with traditional binders and carriers such as triglycerides. Oral formulations can include standard carriers such as pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharine, cellulose, and magnesium carbonate. Any of the common pharmaceutical carriers, such as sterile saline solution or sesame oil, can be used. The medium can also contain conventional pharmaceutical adjunct materials such as, for example, pharmaceutically acceptable salts to adjust the osmotic pressure, buffers, preservatives and the like. Other media that can be used with the compositions and methods provided herein are normal saline and sesame oil.
[0150] The influenza VLPs described herein can be administered alone or in combination with other therapeutic agents to enhance antigenicity. For example, the influenza VLPs can be administered with an adjuvant, such as Freund incomplete adjuvant or Freund's complete adjuvant.
[0151] Optionally, one or more cytokines, such as IL-2, IL-6, IL-12, RANTES, GM-CSF, TNF-.alpha., or IFN-.gamma., one or more growth factors, such as GM-CSF or G-CSF; one or more molecules such as OX-40L or 41 BBL, or combinations of these molecules, can be used as biological adjuvants (see, for example, Salgaller et al., 1998, J. Surg. Oncol. 68(2):122-38; Lotze et al., 2000, Cancer J. Sci. Am. 6(Suppl 1):S61-6; Cao et al., 1998, Stem Cells 16(Suppl 1):251-60; Kuiper et al., 2000, Adv. Exp. Med. Biol. 465:381-90). These molecules can be administered systemically (or locally) to the host.
[0152] A number of means for inducing cellular responses, both in vitro and in vivo, are known. Lipids have been identified as agents capable of assisting in priming CTL in vivo against various antigens. For example, as described in U.S. Pat. No. 5,662,907, palmitic acid residues can be attached to the alpha and epsilon amino groups of a lysine residue and then linked (for example, via one or more linking residues, such as glycine, glycine-glycine, serine, serine-serine, or the like) to an immunogenic peptide. The lipidated peptide can then be injected directly in a micellar form, incorporated in a liposome, or emulsified in an adjuvant. As another example, E. coli lipoproteins, such as tripalmitoyl-S-glycerylcysteinlyseryl-serine can be used to prime tumor specific CTL when covalently attached to an appropriate peptide (see, Deres et al., Nature 342:561, 1989). Further, as the induction of neutralizing antibodies can also be primed with the same molecule conjugated to a peptide which displays an appropriate epitope, two compositions can be combined to elicit both humoral and cell-mediated responses where that is deemed desirable.
[0153] Although administration of VLPs containing the optimized HA protein, one of skill in the art would understand that it is also possible to administer the optimized influenza HA protein itself (in the absence of a viral particle) or as a fusion protein to elicit an immune response in a subject.
[0154] The following examples are provided to illustrate certain particular features and/or embodiments. These examples should not be construed to limit the disclosure to the particular features or embodiments described.
EXAMPLES
Example 1: A Computationally Optimized Broadly Reactive Antigen (COBRA) Based H5N1 VLP Vaccine Elicits Broadly Reactive Antibodies in Mice and Ferrets
[0155] This example describes the finding that mice and ferrets vaccinated with COBRA hemagglutinin (HA) H5N1 VLPs exhibited protective levels of HAI antibodies to representative isolates from each sub-clade of clade 2 and were completely protected from lethal challenge with a clade 2.2 H5N1 virus.
Materials and Methods
COBRA Hemagglutinin (HA) Construction and Synthesis
[0156] Influenza A HA amino acid sequences isolated from human H5N1 infections were downloaded from the NCBI Influenza Virus Resource database (Bao et al., J Virol 82:596-601, 2008; see Table 1 for a complete list of accession numbers and isolate descriptions). Nucleotide sequences were translated into protein sequences using the standard genetic code. All available full length sequences from H5N1 clade 2 human infections from 2004 to 2006 were acquired and used for subsequent consensus generations. For each round of consensus generation, multiple alignment analysis was applied and the consensus sequence was generated using AlignX (Vector NTI). The final amino acid sequence, termed computationally optimized broadly reactive antigen (COBRA), was reverse translated and optimized for expression in mammalian cells, including codon usage and RNA optimization (GeneArt; Regensburg, Germany). This construct was then synthesized and inserted into the pTR600 expression vector (U.S. Patent Application Publication No. 2002/0106798; Ross et al., Nat Immunol. 1(2):102-103, 2000; Green et al., Vaccine 20:242-248, 2001).
TABLE-US-00002 TABLE 1 COBRA input sequences Strain Clade Accession Host Country Year A/Indonesia/534H/2006 2.1.2 EU146737 Human Indonesia 2006 A/Indonesia/535H/2006 2.1.2 EU146753 Human Indonesia 2006 A/Indonesia/536H/2006 2.1.2 EU146754 Human Indonesia 2006 A/Indonesia/538H/2006 2.1.2 EU146745 Human Indonesia 2006 A/Indonesia/546bH/2006 2.1.2 EU146793 Human Indonesia 2006 A/Indonesia/546H/2006 2.1.2 EU146755 Human Indonesia 2006 A/Indonesia/560H/2006 2.1.2 EU146785 Human Indonesia 2006 A/Indonesia/CDC594/2006 2.1.2 CY014272 Human Indonesia 2006 A/Indonesia/CDC595/2006 2.1.2 CY014280 Human Indonesia 2006 A/Indonesia/CDC596/2006 2.1.2 CY014288 Human Indonesia 2006 A/Indonesia/CDC597/2006 2.1.2 CY014296 Human Indonesia 2006 A/Indonesia/CDC599/2006 2.1.2 CY014303 Human Indonesia 2006 A/Indonesia/CDC599N/2006 2.1.2 CY014477 Human Indonesia 2006 A/Indonesia/CDC625/2006 2.1.2 CY014433 Human Indonesia 2006 A/Indonesia/CDC625L/2006 2.1.2 CY014465 Human Indonesia 2006 A/Indonesia/160H/2005 2.1.3 EU146648 Human Indonesia 2005 A/Indonesia/175H/2005 2.1.3 EU146640 Human Indonesia 2005 A/Indonesia/177H/2005 2.1.3 EU146680 Human Indonesia 2005 A/Indonesia/195H/2005 2.1.3 EU146656 Human Indonesia 2005 A/Indonesia/239H/2005 2.1.3 EU146664 Human Indonesia 2005 A/Indonesia/245H/2005 2.1.3 EU146672 Human Indonesia 2005 A/Indonesia/283H/2006 2.1.3 EU146681 Human Indonesia 2006 A/Indonesia/286H/2006 2.1.3 EU146688 Human Indonesia 2006 A/Indonesia/292H/2006 2.1.3 EU146713 Human Indonesia 2006 A/Indonesia/298H/2006 2.1.3 EU146697 Human Indonesia 2006 A/Indonesia/304H/2006 2.1.3 EU146705 Human Indonesia 2006 A/Indonesia/321H/2006 2.1.3 EU146721 Human Indonesia 2006 A/Indonesia/341H/2006 2.1.3 EU146729 Human Indonesia 2006 A/Indonesia/5/2005 2.1.3 EF541394 Human Indonesia 2005 A/Indonesia/542H/2006 2.1.3 EU146777 Human Indonesia 2006 A/Indonesia/567H/2006 2.1.3 EU146801 Human Indonesia 2006 A/Indonesia/569H/2006 2.1.3 EU146809 Human Indonesia 2006 A/Indonesia/583H/2006 2.1.3 EU146817 Human Indonesia 2006 A/Indonesia/604H/2006 2.1.3 EU146825 Human Indonesia 2006 A/Indonesia/7/2005 2.1.3 EU146632 Human Indonesia 2005 A/Indonesia/CDC184/2005 2.1.3 CY014197 Human Indonesia 2005 A/Indonesia/CDC194P/2005 2.1.3 CY014168 Human Indonesia 2005 A/Indonesia/CDC287E/2005 2.1.3 CY014198 Human Indonesia 2005 A/Indonesia/CDC287T/2005 2.1.3 CY014199 Human Indonesia 2005 A/Indonesia/CDC292N/2005 2.1.3 CY014200 Human Indonesia 2005 A/Indonesia/CDC292T/2005 2.1.3 CY014201 Human Indonesia 2005 A/Indonesia/CDC326/2006 2.1.3 CY014204 Human Indonesia 2006 A/Indonesia/CDC326N/2006 2.1.3 CY014202 Human Indonesia 2006 A/Indonesia/CDC326N2/2006 2.1.3 CY014203 Human Indonesia 2006 A/Indonesia/CDC326T/2006 2.1.3 CY014205 Human Indonesia 2006 A/Indonesia/CDC329/2006 2.1.3 CY014206 Human Indonesia 2006 A/Indonesia/CDC357/2006 2.1.3 CY014207 Human Indonesia 2006 A/Indonesia/CDC370/2006 2.1.3 CY014209 Human Indonesia 2006 A/Indonesia/CDC370E/2006 2.1.3 CY014210 Human Indonesia 2006 A/Indonesia/CDC370P/2006 2.1.3 CY014211 Human Indonesia 2006 A/Indonesia/CDC370T/2006 2.1.3 CY014212 Human Indonesia 2006 A/Indonesia/CDC390/2006 2.1.3 CY014213 Human Indonesia 2006 A/Indonesia/CDC523/2006 2.1.3 CY014311 Human Indonesia 2006 A/Indonesia/CDC523E/2006 2.1.3 CY014368 Human Indonesia 2006 A/Indonesia/CDC523T/2006 2.1.3 CY014376 Human Indonesia 2006 A/Indonesia/CDC582/2006 2.1.3 CY014384 Human Indonesia 2006 A/Indonesia/CDC610/2006 2.1.3 CY014393 Human Indonesia 2006 A/Indonesia/CDC623/2006 2.1.3 CY014401 Human Indonesia 2006 A/Indonesia/CDC623E/2006 2.1.3 CY014409 Human Indonesia 2006 A/Indonesia/CDC624/2006 2.1.3 CY014417 Human Indonesia 2006 A/Indonesia/CDC624E/2006 2.1.3 CY014425 Human Indonesia 2006 A/Indonesia/CDC634/2006 2.1.3 CY014441 Human Indonesia 2006 A/Indonesia/CDC634P/2006 2.1.3 CY014449 Human Indonesia 2006 A/Indonesia/CDC634T/2006 2.1.3 CY014457 Human Indonesia 2006 A/Indonesia/CDC644/2006 2.1.3 CY014518 Human Indonesia 2006 A/Indonesia/CDC644T/2006 2.1.3 CY014510 Human Indonesia 2006 A/Indonesia/CDC669/2006 2.1.3 CY014481 Human Indonesia 2006 A/Indonesia/CDC669P/2006 2.1.3 CY014489 Human Indonesia 2006 A/Indonesia/CDC699/2006 2.1.3 CY014497 Human Indonesia 2006 A/Indonesia/CDC7/2005 2.1.3 CY014177 Human Indonesia 2005 A/Indonesia/CDC739/2006 2.1.3 CY014529 Human Indonesia 2006 A/Indonesia/CDC759/2006 2.1.3 CY014543 Human Indonesia 2006 A/Indonesia/CDC835/2006 2.1.3 CY017662 Human Indonesia 2006 A/Indonesia/CDC836/2006 2.1.3 CY017670 Human Indonesia 2006 A/Indonesia/CDC836T/2006 2.1.3 CY017678 Human Indonesia 2006 A/Indonesia/CDC887/2006 2.1.3 CY017688 Human Indonesia 2006 A/Indonesia/CDC938/2006 2.1.3 CY017638 Human Indonesia 2006 A/Indonesia/CDC938E/2006 2.1.3 CY017646 Human Indonesia 2006 A/Indonesia/CDC940/2006 2.1.3 CY017654 Human Indonesia 2006 A/Indonesia/TLL001/2006 2.1.3 EU015403 Human Indonesia 2006 A/Indonesia/TLL002/2006 2.1.3 EU015404 Human Indonesia 2006 A/Indonesia/TLL003/2006 2.1.3 EU015405 Human Indonesia 2006 A/Indonesia/TLL004/2006 2.1.3 EU015406 Human Indonesia 2006 A/Indonesia/TLL005/2006 2.1.3 EU015407 Human Indonesia 2006 A/Indonesia/TLL006/2006 2.1.3 EU015408 Human Indonesia 2006 A/Indonesia/TLL007/2006 2.1.3 EU015409 Human Indonesia 2006 A/Indonesia/TLL008/2006 2.1.3 EU015410 Human Indonesia 2006 A/Indonesia/TLL009/2006 2.1.3 EU015411 Human Indonesia 2006 A/Indonesia/TLL010/2006 2.1.3 EU015412 Human Indonesia 2006 A/Indonesia/TLL011/2006 2.1.3 EU015413 Human Indonesia 2006 A/Indonesia/TLL012/2006 2.1.3 EU015414 Human Indonesia 2006 A/Indonesia/TLL013/2006 2.1.3 EU015415 Human Indonesia 2006 A/Indonesia/TLL014/2006 2.1.3 EU015416 Human Indonesia 2006 A/Djibouti/5691NAMRU3/2006 2.2 DQ666146 Human Djibouti 2006 A/Egypt/7021-NAMRU3/2006 2.2 CY062439 Human Egypt 2006 A/human/Iraq/207-NAMRU3/2006 2.2 DQ435202 Human Iraq 2006 A/Iraq/1/2006 2.2 EU146870 Human Iraq 2006 A/Iraq/659/2006 2.2 EU146876 Human Iraq 2006 A/Iraq/754/2006 2.2 EU146877 Human Iraq 2006 A/Iraq/755/2006 2.2 EU146869 Human Iraq 2006 A/Iraq/756/2006 2.2 EU146878 Human Iraq 2006 A/Turkey/12/2006 2.2 EF619982 Human Turkey 2006 A/Turkey/15/2006 2.2 EF619989 Human Turkey 2006 A/Turkey/651242/2006 2.2 EF619990 Human Turkey 2006 A/Turkey/65596/2006 2.2 EF619998 Human Turkey 2006 A/Xinjiang/1/2006 2.2 FJ492886 Human China 2006 A/Egypt/14724-NAMRU3/2006 2.2.1 EF200512 Human Egypt 2006 A/Egypt/14725-NAMRU3/2006 2.2.1 EF200513 Human Egypt 2006 A/Egypt/2782-NAMRU3/2006 2.2.1 DQ464377 Human Egypt 2006 A/Egypt/2991-NAMRU3/2006 2.2.1 EU095023 Human Egypt 2006 A/Egypt/2992-NAMRU3/2006 2.2.1 EU095024 Human Egypt 2006 A/Egypt/902782/2006 2.2.1 EU146867 Human Egypt 2006 A/Egypt/902786/2006 2.2.1 EU146868 Human Egypt 2006 A/Anhui/1/2005 2.3.4 DQ371928 Human China 2005 A/Anhui/2/2005 2.3.4 DQ371929 Human China 2005 A/China/2006 2.3.4 EF624256 Human China 2006 A/China/GD01/2006 2.3.4 DQ835313 Human China 2006 A/Fujian/1/2005 2.3.4 FJ492882 Human China 2005 A/Guangdong/1/2006 2.3.4 FJ492884 Human China 2006 A/Guangxi/1/2005 2.3.4 DQ371930 Human China 2005 A/human/China/GD02/2006 2.3.4 EU263981 Human China 2006 A/Hunan/1/2006 2.3.4 FJ492879 Human China 2006 A/Jiangxi/1/2005 2.3.4 FJ492885 Human China 2005 A/Shanghai/1/2006 2.3.4 AB462295 Human China 2006 A/Shenzhen/406H/2006 2.3.4 EF137706 Human China 2006 A/Sichuan/1/2006 2.3.4 FJ492881 Human China 2006 A/Vietnam/UT30850/2005 2.3.4 HM114537 Human Viet Nam 2005 A/Zhejiang/1/2006 2.3.4 FJ492880 Human China 2006 A/Zhejiang/16/2006 2.3.4 DQ643809 Human China 2006
COBRA HA Antigenic Modeling
[0157] Influenza hemagglutinin (HA) protein sequences representing COBRA, A/Indonesia/5/2005 (Clade 2.1), A/Whooper Swan/Mongolia/244/2005 (Clade 2.2) and A/Anhui/1/2005 (Clade 2.3) were submitted to the 3D-JIGSAW Protein Comparative Modeling website for rendering (Bates et al., Proteins 45(55):39-46, 2001; Bates and Sternberg, Proteins 37(53):47-54, 1999; Contreras-Moreira and Bates, Bioinformatics 18(8):1141-1142, 2002). Structures were overlaid and analyzed using Swiss-Pdb viewer software (Guex and Peitsch, Electrophoresis 18(15):2714-23, 1998). Antigenic regions and designations are based on the original description of the antigenic structure of the H3N2 virus A/Hong Kong/1/1968 (Wiley et al., Nature 289(5796):373-378, 1981). No significant alterations were observed in region B of the COBRA HA relative to the primary influenza isolates; however, some divergent structures in HA regions A and C were identified in primary isolates.
In Vitro Expression
[0158] COBRA HA protein expression was confirmed by transfecting mammalian cells. Human embryonic kidney (HEK) 293T cells (1.times.10.sup.6) were transiently transfected with 3 .mu.g of DNA expressing COBRA. Cells were incubated for 72 hours at 37.degree. C., supernatants were removed, the cells were lysed with 1% Triton-X 100 and cell lysates were collected. Cell lysates were electrophoresed on a 10% SDS-PAGE gel and transferred to a PVDF membrane. The blot was probed with mouse polyclonal antisera pooled from mice infected with 6:2 reassortant H5N1 viruses with the surface glycoproteins derived from either A/Vietnam/1203/2004 or A/Whooper Swan/244/2005, and the HA-antibody complexes were detected using a goat anti-mouse IgG conjugated to horse radish peroxidase (HRP) (Southern Biotech; Birmingham, Ala., USA). HRP was detected by chemiluminescent substrate (Pierce Biotechnology; Rockford Ill., USA) and exposed to X-ray film (ThermoFisher; Pittsburgh, Pa., USA).
COBRA HA Functional Characterization
[0159] To determine receptor binding characteristics, virus-like particles (VLPs) containing COBRA HA proteins were purified from the supernatants of mammalian cell lines. HEK 293T cells were transiently transfected with plasmids expressing HIV Gag, COBRA HA and neuraminidase (NA, A/Thailand/1(KAN-1)/2004) and incubated for 72 hours at 37.degree. C. Supernatants were collected and VLPs were purified via ultracentrifugation (100,000.times.g through 20% glycerol, weight per volume) for 4 hours at 4.degree. C. The pellets were subsequently resuspended in phosphate buffered saline PBS, pH 7.2 and stored at -80.degree. C. until use. Protein concentration was determined by Micro BCA.TM. Protein Assay Reagent Kit (Pierce Biotechnology, Rockford, Ill., USA). COBRA HA VLPs were prepared in various amounts as measured by total protein and each individual preparation was two-fold serially diluted in v-bottom microtiter plates. An equal volume of either 1% turkey or 1% horse erythrocytes (RBC) (Lampire; Pipersville, Pa., USA) in PBS was added to the diluted VLPs and incubated for 30-60 minutes at room temperature. The HA titer was determined by the reciprocal dilution of the last well which contained agglutinated RBC.
[0160] To determine endosomal fusion characteristics, COBRA-pseudotyped lentiviral vectors expressing a luciferase reporter gene were produced as described (Yang et al., J Virol 78(8):4029-4036). Briefly, 293T cells were co-transfected by using the following plasmids: 7 .mu.g of pCMVdeltaR8.2, 7 .mu.g of pHRCMV-Luc, 3 .mu.g pCMV/R N1(Kan-1) and 3 .mu.g pTR600 COBRA. Cells were transiently transfected and incubated for 72 hours at 37.degree. C. Supernatants were harvested, centrifuged to clear cell debris, filtered through a 0.22 .mu.m syringe filter, aliquotted, and used immediately or frozen at -80.degree. C. For fusion assays, 100 .mu.l of pseudoviruses were added to confluent MDCK cells in 48-well plates (30,000 cells per well). Plates were incubated at room temperature for 30 minutes, washed, and fresh medium added. Forty-eight hours after infection, cells were lysed in mammalian cell lysis buffer (Promega; Madison, Wis., USA). A standard quantity of cell lysate was used in a luciferase assay with luciferase assay reagent (Promega; Madison, Wis., USA) according to the manufacturer's protocol.
Vaccine Preparation and Dose Determination HEK 293T cells were transiently transfected with plasmids expressing M1 (A/Puerto Rico/8/1934, optimized for expression in mammalian cells; SEQ ID NO: 9), NA (A/Thailand/1(KAN-1)/2004, optimized for expression in mammalian cells; SEQ ID NO: 8) and COBRA HA (SEQ ID NO: 7) and incubated for 72 hours at 37.degree. C. Supernatants were collected and cell debris removed by low speed centrifugation followed by vacuum filtration through a 0.22 .mu.m sterile filter. VLPs were purified via ultracentrifugation (100,000.times.g through 20% glycerol, weight per volume) for 4 hours at 4.degree. C. The pellets were subsequently resuspended in PBS pH 7.2 and stored in single use aliquots at -80.degree. C. until use. Total protein concentration was determined by Micro BCA.TM. Protein Assay Reagent Kit (Pierce Biotechnology, Rockford, Ill., USA).
[0161] HA specific content was determined by western blot and densitometry. Purified recombinant COBRA HA and purified VLPs were prepared in standard total protein amounts and were electrophoresed on a 10% SDS-PAGE gel and transferred to a PVDF membrane. The blot was probed with mouse polyclonal antisera pooled from mice infected with 6:2 reassortant H5N1 viruses with the surface glycoproteins derived from either A/Vietnam/1203/2004 or A/Whooper Swan/244/2005 and the HA-antibody complexes were detected using a goat anti-mouse IgG conjugated to horse radish peroxidase (HRP) (Southern Biotech; Birmingham, Ala., USA). HRP was detected by chemiluminescent substrate (Pierce Biotechnology; Rockford Ill., USA) and exposed to X-ray film (ThermoFisher; Pittsburgh, Pa., USA). Density of bands was determined using ImageJ software (NIH) (Abramoff et al., Biophotonics International 11(7):36-42, 2004). Density of recombinant HA bands were used to calculate a standard curve and the density of the purified VLPs was interpolated using the results from the recombinant HA. Experiments were performed in triplicate and multiple exposure times were analyzed for all iterations.
Codon-Optimized Influenza HA, NA and M1 Genes
[0162] The nucleotide sequences of the codon-optimized HA (SEQ ID NO: 1), codon-optimized NA (SEQ ID NO: 3) and codon-optimized M1 (SEQ ID NO: 5) genes are set forth in the Sequence Listing. The corresponding amino acid sequences of the encoded HA, NA and M1 proteins are set forth in the Sequence Listing as SEQ ID NO: 2, SEQ ID NO: 4 and SEQ ID NO: 6, respectively.
Mouse Studies
[0163] BALB/c mice (Mus musculis, females, 6-8 weeks old) were purchased from Harlan Sprague Dawley (Indianapolis, Ind., USA) and housed in microisolator units and allowed free access to food and water and were cared for under USDA guidelines for laboratory animals. Mice (5 mice per group) were vaccinated with one of three doses of purified COBRA HA VLPs (1.5 .mu.g, 0.3 .mu.g or 0.06 .mu.g), based upon HA content from a densitometry assay, via intramuscular injection at week 0 and then boosted with the same dose at week 3. For comparison studies, mice (20 mice per group) were vaccinated with purified VLPs (3 .mu.g) via intramuscular injection at week 0 and then boosted with the same dose at week 3. Vaccines at each dose were formulated with Imject.RTM. alum adjuvant (Imject.RTM. Alum, Pierce Biotechnology; Rockford, Ill., USA) according to the manufacturer's protocol or vehicle alone. Fourteen to twenty-one days after each vaccination, blood was collected from anesthetized mice via the retro-orbital plexus and transferred to a microfuge tube. Tubes were centrifuged and sera was removed and frozen at -20.+-.5.degree. C.
[0164] Three weeks after final vaccination, mice were challenged intranasally with 5.times.10.sup.3 plaque forming units (PFU) of the highly pathogenic H5N1 virus A/Whooper Swan/Mongolia/244/2005 (clade 2.2) in a volume of 50 .mu.l. The challenge dose represents approximately 50LD.sub.50 in mice. After infection, mice were monitored daily for weight loss, disease signs and death for 14 days after infection. Individual body weights, sickness scores and death were recorded for each group on each day after inoculation. Sickness score was determined by evaluating activity (0=normal, 1=reduced, 2=severely reduced), hunched back (0=absent, 1=present) and ruffled fur (0=absent, 1=present) (Toapanta and Ross, Respiratory Res 10(1):112, 2009). Experimental endpoint was defined as >20% weight loss or display of neurological disease such as hind limb paralysis. All H5N1 influenza virus studies were performed under high-containment biosafety level 3 enhanced conditions (BSL3+).
Ferret Studies
[0165] Fitch ferrets (Mustela putorius faro, female, 6-12-months of age), influenza naive and de-scented, were purchased from Marshall Farms (Sayre, Pa., USA). Ferrets were pair housed in stainless steel cages (Shor-line, Kansas City, Kans., USA) containing Sani-chips Laboratory Animal Bedding (P.J. Murphy Forest Products, Montville, N.J., USA). Ferrets were provided with Teklad Global Ferret Diet (Harlan Teklad, Madison, Wis., USA) and fresh water ad libitum. The COBRA HA VLPs were diluted in PBS, pH 7.2 to achieve final concentration. Ferrets (n=3) were vaccinated with 15 .mu.g of purified COBRA VLPs, based upon HA content as determined by densitometry assay, via intramuscular injection in the quadriceps muscle in a volume of 0.25 ml at week 0 and then boosted with the same dose at week 3. Vaccines were stored at -80.degree. C. prior to use and formulated with Imject.RTM. alum adjuvant (Imject.RTM. Alum; Pierce Biotechnology, Rockford, Ill., USA) immediately prior to use. Animals were monitored for adverse events including weight loss, temperature, loss of activity, nasal discharge, sneezing and diarrhea weekly during the vaccination regimen. Prior to vaccination, animals were confirmed by HAI assay to be seronegative for circulating influenza A (H1N1 and H3N2) and influenza B viruses. Fourteen to twenty-one days after each vaccination, blood was collected from anesthetized ferrets via the anterior vena cava and transferred to a microfuge tube. Tubes were centrifuged and sera was removed and frozen at -20.+-.5.degree. C.
[0166] Three weeks after final vaccination, ferrets were challenged intranasally with 1.times.10.sup.6 plaque forming units (PFU) of the highly pathogenic H5N1 virus A/Whooper Swan/Mongolia/244/2005 (clade 2.2) in a volume of 0.5 ml in each nostril for a total infection volume of 1 ml. After infection, ferrets were monitored daily for weight loss, disease signs and death for 14 days after infection. Individual body weights, sickness scores, and death were recorded for each group on each day after inoculation. Sickness score was determined by evaluating activity (0=normal, 1=alert and active with stimulation, 2=alert but not active after stimulation, 3=not alert or active after stimulation), nasal discharge (0=absent, 1=present), sneezing (0=absent, 1=present), decreased food intake (0=absent, 1=present), diarrhea (0=absent, 1=present), dyspnea (0=absent, 1=present) and neurological symptoms (0=absent, 1=present). Nasal washes were performed by instilling 3 ml of PBS into the nares of anesthetized ferrets each day for 14 days after inoculation. Washes were collected and stored at -80.degree. C. until use. Experimental endpoint was defined as >20% weight loss, development of neurological symptoms, or an activity score of 3 (not active or alert after stimulation). All H5N1 influenza virus studies were performed under high-containment biosafety level 3 enhanced conditions (BSL3+).
Elisa
[0167] The ELISA assay was used to assess total antibody titer and IgG isotype titer to the HA. High binding, 96-well polystyrene plates (Costar; Lowell, Mass., USA) were coated overnight with 50 ng/well of recombinant HA. Coating antigens were derived from the following representative viral isolates: A/Vietnam/1203/2004 (clade 1), A/Indonesia/5/2005 (clade 2.1), A/Whooper Swan/244/2005 (clade 2.2) and A/Anhui/1/2005 (clade 2.3). Plates were blocked with 5% milk diluted in PBS with 0.05% Tween 20. Serum samples were diluted in blocking buffer and added to plates. Serum was two-fold serially diluted and allowed to incubate for 1 hour at room temperature. Plates were washed and species specific antibody against IgG, IgG1, IgG2a, IgG2b or IgG3 and linked to horseradish peroxidase (HRP) (Southern Biotech; Birmingham, Ala., USA) were diluted in blocking buffer and added to plates. Plates were incubated for 1 hour at room temperature. Plates were washed and HRP was developed with TMB substrate (Sigma-Aldrich; St. Louis, Mo., USA). Plates were incubated in the dark for 15 minutes and then the reaction was stopped with 2N H2SO.sub.4. Optical densities at a wavelength of 450 nm (OD.sub.450) were read by a spectrophotometer (BioTek; Winooski, Vt., USA) and end point dilution titers were determined. End point titers were determined as the reciprocal dilution of the last well which had an OD.sub.450 above the mean OD.sub.450 plus two standard deviations of naive animal sera.
Hemagglutination Inhibition (HAI)
[0168] The HAI assay was used to assess functional antibodies to HA able to inhibit agglutination of horse erythrocytes. The protocol was adapted from the CDC laboratory-based influenza surveillance manual (Gillim-Ross and Subbarao, Clin Microbiol Rev 19(4):614-636, 2006). To inactivate non-specific inhibitors, sera were treated with receptor destroying enzyme (RDE; Denka Seiken, Co., Japan) prior to being tested (Bright et al., Lancet 366(9492):1175-1181, 2005; Bright et al., Virology 308(2):270-278, 2003; Bright et al., JAMA 295(8):891-894, 2006; Mitchell et al., Vaccine 21(9-10):902-914, 2004; Ross et al., Nat Immunol 1(2):127-131, 2000). Briefly, three parts RDE was added to one part sera and incubated overnight at 37.degree. C. RDE was inactivated by incubation at 56.degree. C. for .about.30 minutes. RDE-treated sera was two-fold serially diluted in v-bottom microtiter plates. An equal volume of reassortant virus, adjusted to approximately 8 HAU/50 .mu.l, was added to each well. The reassortant viruses contained the internal genes from the mouse adapted strain A/Puerto Rico/8/1934 and the surface proteins HA and NA from the following representative viral isolates: A/Vietnam/1203/2004 (clade 1), A/Indonesia/5/2005 (clade 2.1), A/Whooper Swan/244/2005 (clade 2.2) and A/Anhui/1/2005 (clade 2.3). The plates were covered and incubated at room temperature for 20 minutes followed by the addition of 1% horse erythrocytes (hRBC) (Lampire Biologicals, Pipersville, Pa., USA) in PBS. Red blood cells were stored at 4.degree. C. and used within 72 hours of preparation. The plates were mixed by agitation, covered, and the RBCs were allowed to settle for 1 hour at room temperature (Askonas B, McMichael A, Webster R. The immune response to influenza viruses and the problem of protection against infection. In: Beare AS, editor. Basic and applied influenza research: CRC Press 1982: 159-188). The HAI titer was determined by the reciprocal dilution of the last row which contained non-agglutinated RBCs. Positive and negative serum controls were included for each plate. All mice were negative (HAI.ltoreq.1:10) for pre-existing antibodies to currently circulating human influenza viruses prior to vaccination.
Plaque Assay
[0169] Madin-Darby Canine Kidney (MDCK) cells were plated (5.times.10.sup.5) in each well of a 6-well plate. Samples were diluted (final dilution factors of 10.degree. to 10.sup.-6) and overlayed onto the cells in 100 .mu.l of DMEM supplemented with penicillin-streptomycin and incubated for 1 hour. Samples were removed, cells were washed twice and media was replaced with 2 ml of L15 medium plus 0.8% agarose (Cambrex; East Rutherford, N.J., USA) and incubated for 72 hours at 37.degree. C. with 5% CO2. Agarose was removed and discarded. Cells were fixed with 10% buffered formalin, and then stained with 1% crystal violet for 15 minutes. Following thorough washing in dH.sub.2O to remove excess crystal violet, plates were allowed to dry, plaques counted, and the plaque forming units (PFU)/m1 were calculated.
Statistical Analysis
[0170] Statistical significance of the antibody data was determined using a two-way analysis of variance (ANOVA) with Bonferroni's post-test to analyze differences between each vaccine group for the different test antigens (multiparametric). Differences in weight loss, sickness score, and viral titers were analyzed by two-way ANOVA, followed by Bonferroni's post test for each vaccine group at multiple time points. Significance was defined as p<0.05. Statistical analyses were done using GraphPad Prism software.
Results
Computationally Optimized Broadly Reactive Antigen Design
[0171] To address the challenge of antigenic diversity present in H5N1 influenza, a computationally optimized broadly reactive antigen (COBRA) was designed. For the first step of antigen generation, 129 unique hemagglutinin (HA) sequences were downloaded from the NCBI Influenza Virus Resource (IVR) sequence database (Bao et al., J Virol 82:596-601, 2008) representing clade 2 H5N1 viruses isolated from human infections between 2004 and 2006. The sequences were first grouped into phylogenetic sub-clades and then further divided into individual outbreak groups within each sub-clade based upon geographic location and time of isolation. HA amino acid sequences for each individual outbreak group were aligned and the most common amino acid at each position was determined resulting in primary consensus sequences representing each outbreak group within each sub-clade (FIG. 1A). Primary consensus sequences within each sub-clade were then aligned and the most common amino acid was chosen resulting in secondary consensus sequences representing each sub-clade (FIG. 1A). The secondary consensus sequences were aligned and the most common amino acid at each position was selected resulting in the final consensus sequence referred to as clade 2 COBRA HA (FIG. 1A). Phylogenetic analysis of the COBRA HA with all human isolates of H5N1 HA proteins indicated that COBRA retained a clade 2-like sequence without being grouped specifically within any clade 2 sub-clade cluster (FIG. 1B). Furthermore, a BLAST search using the COBRA HA sequence revealed that it is a unique sequence that has not been isolated from the environment.
Characterization of COBRA
[0172] Since COBRA is a fully synthetic protein, the retention of natural hemagglutinin function was confirmed. Initially, COBRA expression was verified by transient transfection of mammalian cells. Analysis of the total cell lysate demonstrated that the COBRA HA migrates at its predicted molecular weight of approximately 73 kDa (FIG. 2A). Because the poly-basic cleavage site was retained in the COBRA HA sequence, both HA0 and the HA1 subunits were detected by immunoblot at similar molecular weights as recombinant HA and the HA on the H5N1 virion (FIG. 2A). Virus-like particles (VLPs) with COBRA HA on the surface bound sialic acid in a dose-dependent manner and this binding was specific to COBRA, since empty lentiviral core alone did not bind to the receptor (FIG. 2B).
[0173] To determine if the COBRA HA was functional, the protein was pseudotyped onto lentiviral Gag.sub.p24 to generate pseudoparticles (Nefkens et al., J Clin Virol 39(1):27-33, 2007; Haynes et al., Vaccine 27(4):530-541, 2009). COBRA HA containing pseudoparticles mediated cell fusion as efficiently as H5N1 control pseudoparticles without the requirement for trypsin. In contrast, H1N1 pseudoparticles did require trypsin and pseudoparticles without surface HA produced luciferase at similar levels as the cell only controls (FIG. 2C). Taken together, these results demonstrate that although the COBRA HA is a synthetic protein not found in nature, it retains all of the functions of a natural hemagglutinin protein.
Mouse Dosing Immunizations
[0174] Mice (BALB/c; n=5) were vaccinated (week 0 and 3) via intramuscular injection with purified COBRA VLPs at either a high dose (1.5 .mu.g HA) or low dose (0.3 .mu.g HA) with and without Imject.RTM. alum adjuvant. At week 5, all COBRA VLP-vaccinated animals had anti-HA antibodies that recognized heterologous recombinant HA derived from both clade 1 and also sub-clades of clade 2 (FIGS. 3A and 3B). Imject.RTM. alum significantly increased anti-HA antibody titers in both low and high dose groups as compared to the non-adjuvanted groups (p<0.01). The IgG isotype subclasses elicited by the VLP vaccines against a clade 2.1 coating antigen consisted mainly of IgG1 and IgG2a, indicating a mixed T helper response (FIGS. 3C and 3D). Similar results were found for additional coating antigens representing clade 1, clade 2.2 and clade 2.3. In addition to recognizing HA, antibodies were also evaluated for the ability to block virus from binding its receptor via inhibition of viral-induced agglutination of horse erythrocytes (HAI). All mice administered Imject.RTM. alum adjuvanted vaccines, regardless of dose, had HAI titers.gtoreq.1:40 to viruses expressing HA from clades 2.1 and 2.2 and 90% of the mice had titers.gtoreq.1:40 to a clade 2.3 representative virus (FIGS. 3E and 3F). Non-adjuvanted vaccines elicited generally lower HAI antibody titers with 100% of high dose animals achieving titers.gtoreq.1:40 only against clade 2.1 viruses. Imject.RTM. alum adjuvanted vaccines elicited significantly higher HAI antibody titers to clade 2.2 and clade 2.3 viruses regardless of dose as compared to non-adjuvanted vaccines (p<0.05 for high dose and p<0.001 for low dose, respectively). None of the vaccines elicited high HAI titer antibodies to a clade 1 virus.
Mouse Dosing Challenge
[0175] Mice that received the COBRA VLP vaccines or mock vaccinated control mice were challenged intranasally with a lethal dose of clade 2.2 H5N1 highly pathogenic avian influenza (A/Mongolia/whooper swan/244/2005) to evaluate the protective efficacy of the different COBRA vaccine formulations. All COBRA vaccinated mice, regardless of dose or the presence of adjuvant, were protected from weight loss and death following lethal challenge, while all mock vaccinated animals rapidly lost weight and required euthanasia by day 6 post infection (FIGS. 4A and 4B). Additionally, COBRA VLP vaccinated mice had no signs of disease, while mock vaccinated animals developed such symptoms as ruffled fur, hunched back, and lethargy (FIGS. 4C and 4D).
Mouse Comparison Immunizations
[0176] To determine if the COBRA HA vaccine elicits a broader antibody response compared to a vaccine derived from a primary isolate, an additional set of mice were vaccinated with either COBRA VLPs or clade 2.2 (A/Mongolia/whooper swan/244/2005) VLPs. Mice (BALB/c; n=20) were vaccinated (week 0 and 3) via intramuscular injection with either COBRA VLPs or clade 2.2 VLPs at a high dose (3 .mu.g HA) with Imject.RTM. alum adjuvant. At week 5, all COBRA VLP-vaccinated mice and all clade 2.2 VLP-vaccinated mice had anti-HA antibodies that recognized heterologous recombinant HA derived from both clade 1 and various sub-clades of clade 2 (FIG. 5A). Although no significant differences were found in total IgG titers between vaccine groups, COBRA VLP-vaccinated animals had higher HAI antibody titers against all viruses tested as compared to clade 2.2 VLP-vaccinated animals (p<0.01; FIG. 5B). Furthermore, COBRA VLP-vaccinated animals had an increased frequency of HAI titers of .gtoreq.1:40 compared to clade 2.2 VLP-vaccinated animals (Table 2).
TABLE-US-00003 TABLE 2 Mouse seroconversion frequency Vaccine Antigen Clade 1.sup.a Clade 2.1.sup.b Clade 2.2.sup.c Clade 2.3.sup.d COBRA 45% (9/20) 100% (20/20) 100% (20/20) 100% (20/20) Clade 2.2.sup.c 0% (0/20) 0% (0/20) 0% (0/20) 0% (0/20) Percentage of VLP-vaccinated animals achieving an HAI titer of .gtoreq.1:40 to each test antigen. .sup.aA/Vietnam/1203/2004 .sup.bA/Indonesia/5/2005 .sup.cA/Whooper Swan/Mongolia/244/2005 .sup.dA/Anhui/1/2005
Mouse Comparison Challenge
[0177] Mice that received the COBRA VLP vaccine, clade 2.2 VLP vaccine or mock vaccinated control mice were challenged intranasally with a lethal dose of clade 2.2 H5N1 highly pathogenic avian influenza (A/Mongolia/whooper swan/244/2005) to evaluate the protective efficacy of the VLP vaccines. All VLP-vaccinated mice were protected from weight loss and death following lethal challenge while all mock vaccinated animals rapidly lost weight and required euthanasia by day 6 post infection (FIG. 6A). Additionally, VLP vaccinated mice did not show signs of disease, while mock vaccinated animals developed ruffled fur, hunched back, and lethargy (FIG. 6B). Even though the clade 2.2 VLP was matched to the challenge virus, no significant differences were found between COBRA VLP and clade 2.2 VLP vaccinated mice in any of the parameters analyzed indicating that the COBRA VLP vaccine protected animals as efficiently as the homologous vaccine.
Ferret Comparison Immunizations
[0178] Ferrets are the most relevant model for influenza disease and as such the COBRA vaccine was tested in this more rigorous animal model. Ferrets (Fitch; n=9) were vaccinated (week 0 and 3) via intramuscular injection with COBRA VLPs or clade 2.2 VLPs at a high dose (15 .mu.g HA) with Imject.RTM. alum adjuvant. Serum was collected from ferrets at week 5 and antibody responses to the COBRA vaccines were evaluated. All vaccinated ferrets had anti-HA antibodies that recognized heterologous recombinant HA derived from both clade 1 and also sub-clades of clade 2 (FIG. 7A). No significant difference in anti-HA antibody was found between the COBRA VLP vaccine and the clade 2.2 VLP vaccine for any of the antigens tested (p>0.05). In addition to recognizing HA, antibodies were also evaluated for HAI activity. COBRA VLP-vaccinated animals had higher HAI antibody titers against clade 2.1 and clade 2.3 viruses as compared to clade 2.2 VLP-vaccinated animals (p<0.01 FIG. 7B). Similar to the mice, COBRA VLP-vaccinated ferrets displayed an increased rate of achieving HAI titers.gtoreq.1:40 when compared to clade 2.2 VLP-vaccinated ferrets (Table 3).
TABLE-US-00004 TABLE 3 Ferret seroconversion frequency Vaccine Antigen Clade 1.sup.a Clade 2.1.sup.b Clade 2.2.sup.c Clade 2.3.sup.d COBRA 0% (0/9) 78% (7/9) 56% (5/9) 56% (5/9) Clade 2.2.sup.c 0% (0/9) 0% (0/9) 22% (2/9) 0% (0/9) Percentage of VLP-vaccinated animals achieving an HAI titer of .gtoreq.1:40 to each test antigen. .sup.aA/Vietnam/1203/2004 .sup.bA/Indonesia/5/2005 .sup.cA/Whooper Swan/Mongolia/244/2005 .sup.dA/Anhui/1/2005
Ferret Comparison Challenge
[0179] Ferrets that received the COBRA VLP vaccines, clade 2.2 VLP vaccines or mock vaccinated control animals were challenged intranasally with clade 2.2 H5N1 highly pathogenic avian influenza (A/Mongolia/whooper swan/244/2005) to evaluate the protective efficacy of the COBRA vaccine in the ferret model of influenza infection. All VLP vaccinated ferrets were protected from weight loss and death following viral challenge, while all mock vaccinated animals rapidly lost weight and 78% (7/9) of mock vaccinated animals required euthanasia by day 7 post-infection (FIGS. 8A and 8B). Additionally, both COBRA VLP-vaccinated and clade 2.2-vaccinated ferrets were protected from acute fever and failed to develop significant signs of disease while mock vaccinated animals had an elevated body temperature and developed such symptoms as lethargy, diarrhea and decreased food and water intake (FIGS. 8C and 8D). In addition to monitoring outward signs of disease progression, nasal washes were collected for determination of viral replication in the upper respiratory tract. Ferrets vaccinated with either COBRA VLPs or clade 2.2 VLPs did not have detectable virus at any point after infection, while mock vaccinated animals had high levels of viral replication for the first five days of the infection (FIG. 8E). No significant differences were found between COBRA VLP and clade 2.2 VLP vaccinated ferrets in any of the challenge parameters analyzed confirming the findings in mice that the COBRA VLP vaccine protected animals as efficiently as the homologous vaccine.
[0180] The percent identity of COBRA HA and the test antigens used in the mouse and ferret studies described above are shown in Table 4.
TABLE-US-00005 TABLE 4 Percent Identity of Test Antigens Vaccine Antigen Clade 1.sup.a Clade 2.1.sup.b Clade 2.2.sup.c Clade 2.3.sup.d COBRA 97% 97% 95% 97% Clade 2.2.sup.c 94% 97% 100% 94% HA amino acid sequences were aligned and percent identity across the entire protein was determined for the vaccine immunogens compared to the representative test antigens. .sup.aA/Vietnam/1203/2004 .sup.bA/Indonesia/5/2005 .sup.cA/Whooper Swan/Mongolia/244/2005 .sup.dA/Anhui/1/2005
Example 2: A Computationally-Optimized HA VLP Vaccines Elicits Broadly-Reactive Antibodies that Protect Monkeys from H5N1 Infection
[0181] This example describes the finding that a COBRA clade 2 HA H5N1 VLP elicits broad humoral immunity against multiple H5N1 isolates from different clades.
Materials and Methods
Expression and Purification of Virus-Like Particles
[0182] The COBRA HA sequence is described above in Example 1. 293T cells were transiently transfected with plasmids expressing HA, M1, and NA in low serum media, incubated for 72 h at 37.degree. C., and purified by ultracentrifugation through a 20% glycerol cushion as previously described (Giles and Ross, Vaccine 29:3043-3054, 2011). All VLP vaccines were engineered using the same NA from A/Thailand/1(KAN-1)/2004. HA content was quantified as previously described (Giles and Ross, Vaccine 29:3043-3054, 2011). Two different VLP preparations were purified, each containing one of the HA influenza gene products: WS/05 or the COBRA HA.
Primate Immunizations and H5N1 Challenges
[0183] Cynomolgus macaques (Macaca fascicularis, male, 3-5 years old) were vaccinated with 15 .mu.g (based upon HA content) of purified COBRA HA VLPs (n=7) or WS/05 VLPs (n=7), via intramuscular injection at weeks 0, 3 and 6. Vaccines at each dose were formulated with alum adjuvant (Imject.RTM. Alum, Pierce Biotechnology; Rockford, Ill., USA) immediately prior to use. Twenty-one days after each vaccination, blood was collected from anesthetized macaques. All procedures were in accordance with the NRC Guide for the Care and Use of Laboratory Animals.
[0184] Three weeks after final vaccination, macaques were placed into BSL3+ isolator units (Bioqual, Inc., Rockville, Md.) and then challenged by a multi-route of infection (ocular, nasal, tracheal) as previously described (Kobasa et al., Nature 445:319-323, 2007; Kuiken et al., Vet Pathol 40:304-310, 2003; Rimmelzwaan et al., Avian Dis 47:931-933, 2003) using 1.times.10.sup.6 plaque forming units (PFU) of the highly pathogenic H5N1 virus, A/Whooper Swan/Mongolia/244/2005 (clade 2.2), at each location. Monkeys were monitored daily for weight loss, signs of disease, and mortality until 7 days after infection. Individual body weights, sickness scores (based upon lethargy, temperature change, nasal discharge, lack of appetite, dehydration, lack of responsiveness), and death were recorded for each group.
[0185] Nasal and tracheal washes were performed at day 0, 1, 3, 5, and 7 post-infection. In addition, subsets of monkeys were sacrificed following administration of anesthesia and necropsies were performed according to standard procedures for assessment of gross pathologic and histopathologic changes, as well as the extent of virus replication.
Serological Assays
[0186] A quantitative ELISA was performed to assess anti-HA specific IgG in immune serum as previously described (Bright et al., PLoS One 3:e1501, 2008; Giles and Ross, Vaccine 29:3043-3054, 2011). The hemagglutination inhibition (HAI) assay was used on sera treated with receptor destroying enzyme (RDE; Denka Seiken, Co., Japan) prior to being tested (Bright et al., Vaccine 25:3871-3878, 2007; Mitchell et al., Vaccine 21:902-914, 2003; Bright et al., PLoS One 3:e1501, 2008) to assess functional antibodies to the HA able to inhibit agglutination of horse red blood (Askonas B, McMichael A, Webster R. The immune response to influenza viruses and the problem of protection against infection. In: Beare AS, editor. Basic and applied influenza research: CRC Press 1982: 159-188). The protocol was adapted from the CDC laboratory-based influenza surveillance manual and performed as previously described (Gillim-Ross and Subbarao, Clin Microbiol Rev 19:614-636, 2006; Bright et al., PLoS One 3:e1501, 2008). The HAI titer was determined by the reciprocal dilution of the last row which contained non-agglutinated RBC. Positive and negative serum controls were included for each plate. All monkeys were negative (HAI.ltoreq.1:10) for pre-existing antibodies to currently circulating human influenza viruses prior to vaccination. Serum neutralizing antibody titers were determined by microneutralization (MN) assays performed on Madin Darby Canine Kidney (MDCK) cells following the procedure until CPE was observed (Rowe et al., J Clin Microbiol 37:937-943, 1999). Cells were then fixed in 10% formalin and stained with 1% crystal violet to quantify CPE. The neutralizing antibody titers are expressed as the reciprocal of the highest dilution of serum that gave 50% neutralization of 100 TCID.sub.50 of virus in MDCK cells. Geometric mean neutralizing antibody titers were calculated for each group.
Histopathologic Evaluation and Immunohistochemical Analysis
[0187] Formalin-inflated lungs and trachea were fixed in 10% neutral buffered formalin. Cross-sections of upper and lower left and right lung lobes and trachea were made, concentrating on gross-lesions. Tissue was paraffin-embedded and 6 .mu.m sections were stained with hematoxylin and eosin for histologic evaluation. Sequential sections were processed for immunohistochemistry or in situ hybridization (ISH). Immunohistochemistry was performed as described previously (Bissel et al., Am J Pathol 160:927-941, 2002) using an immunoperoxidase method with a polyclonal antibody (Maine Biotechnology Services, Portland, Me.) directed against influenza A virus. A biotinylated donkey anti-goat IgG (Rockland Immunochemicals, Gilbertsville, Pa.) was used as the secondary antibody. ISH was performed as described previously (Fallert et al., J Virol Methods 99:23-32, 2002) using a 35S-labeled riboprobe synthesized using templates derived from 760 bp of influenza A/California/04/2009 matrix protein.
Results
Vaccine Induced Antibody Responses
[0188] Cynomolgus macaques were vaccinated with COBRA VLPs or WS/05 VLPs formulated with Imject.RTM. alum at 0, 3 and 6 weeks. At week 3 post-vaccination, all COBRA VLP-vaccinated animals had anti-HA antibodies that recognized recombinant HA derived from three sub-clades of clade 2, which were boosted at week 6 (FIGS. 10A and 10B). There was no statistical difference (p>0.05) in the anti-HA titers elicited against any of the HA proteins, except monkeys vaccinated with COBRA VLPs had a statistically higher titer against the Indo/05 HA (clade 2.1) compared with monkeys vaccinated with the WS/05 VLP (derived from clade 2.2) on week 6.
A Single COBRA VLP Vaccination Induced High Titer HAI and MN Antibodies to Clade 2 H5N1 Viruses
[0189] Monkeys vaccinated with COBRA VLPs (but not with WS/05 VLPs) had HAI activity against viruses representing all three clade 2 sub-clades after a single vaccination (FIG. 10C). Four to six monkeys responded to the COBRA VLP vaccine with an HAI titer.gtoreq.1:40 for the all of the various test antigens. In contrast, 4 of 7 monkeys vaccinated with the WS/05 VLP responded to the homologous clade 2.2 virus, but none of these vaccinated monkeys responded to the clade 2.1 or 2.3 virus. Following a second vaccination, almost all the monkeys vaccinated with either vaccine responded to all three viruses (FIG. 11D). These results were confirmed by microneutralization assay (FIGS. 11E and 11F). However, monkeys vaccinated with COBRA VLPs showed boosted HAI titers to all three clade 2 viruses (FIG. 11).
COBRA VLPs Induced HAI Antibodies that Recognize Broader Numbers of H5N1 Viruses
[0190] In order to determine if the COBRA HA elicited antibodies that recognized a broader number of H5N1 isolates, serum was collected and tested for the ability to inhibit influenza virus induced hemagglutination of red blood cells in vitro. Antisera collected from both vaccinated and unvaccinated monkeys were then tested against a broad panel of H5N1 viruses representing not only sub-clades of clade 2, but also non-clade 2 H5N1 virus strains (0, 1, 4, and 7) by HAI. Monkeys vaccinated with the COBRA VLP had high average HAI titers against all clade 2 isolates, regardless of sub-clade (FIG. 11). In general, all 7 monkeys responded to the COBRA VLP vaccine and seroconverted with an HAI titer.gtoreq.1:40 against all the clade 2 viruses. In contrast, monkeys vaccinated with the WS/05 VLP vaccine had lower HAI titers against clade 2 viruses (FIG. 10) and fewer number of monkeys responded to the vaccine. Of the 10 clade 2 viruses tested in the HAI assay, WS/05 VLP vaccinated monkeys responded more poorly (fewer than 4 of 7 monkeys) to 4 of the isolates and none of these monkeys had antibodies that responded to the Dk/HU/02 (clade 2.1.1) or Eg/3300/08 (clade 2.2.1) isolates. The COBRA VLPs elicited significantly higher HAI titers against almost all of the clade 2 viruses than the WS/05 VLPs (FIG. 11).
[0191] In addition to clade 2 isolates, a minimum of five COBRA VLP vaccinated monkeys had HAI antibodies against both clade 1 and 7 virus isolates (FIG. 11). In comparison, almost none of the WS/05 VLP vaccinated monkeys had HAI antibodies against clade 1 and clade 7 viruses. None of the monkeys, regardless of the vaccine, had antibodies that responded to the clade 0 or 4 isolates. All mock vaccinated monkeys did not recognize any of the H5N1 isolates.
Challenge of Vaccinated and Unvaccinated Primates with H5N1 Clade 2.2 Virus
[0192] Three weeks after final vaccination, both VLP vaccinated and mock-vaccinated monkeys were transferred to ABSL3+ isolator units and then challenged with highly pathogenic H5N1 virus, A/Whooper Swan/Mongolia/244/2005 (clade 2.2) (1.times.10.sup.6pfu), by a multi-route (ocular, nasal, tracheal, oral) of infection (Kobasa et al., Nature 445:319-323, 2007; Kuiken et al., Vet Pathol 40:304-310, 2003; Rimmelzwaan et al., Avian Dis 47:931-933, 2003). There was no significant weight loss or mortality in any of the monkeys over the 7 day period of observation. Unvaccinated monkeys had an elevated temperature of .about.2.degree. C. that was sustained for 5 days post-infection and higher gross pathology scores by day 3 post-infection (Table 5).
TABLE-US-00006 TABLE 5 Lung pathology, temperature and viral titer of vaccinated macaques Lung Pathology Elevated Score temperature Peak Viral Titer (pfu/ml) Vaccine (day 3) (days) (day) Mock 5.3 1.9.degree. C. Nasal wash: 2.2-2.5 (5 DPI) (1-5 DPI) Trachea wash: 2.0-4.4 (3 DPI) WS/05 VLP 3.3 1.1.degree. C.-1.3.degree. C. Nasal wash: <2 (1-5 DPI) Trachea wash: <2 COBRA 2.1 1.3.degree. C. Nasal wash: <2 VLP (2 DPI) Trachea wash: <2
[0193] The lungs of unvaccinated monkeys had mild to moderate acute pneumonia with alveolar pulmonary exudate by day 3 post-infection by H&E staining. ISH showed focal collections of H5N1 infected cells present at day 3 post-infection in alveolar spaces and to a lesser extent in bronchial epithelium. These results were similar to unvaccinated monkeys infected with the clade 1 H5N1 virus, A/Vietnam/1203/2004. In contrast, monkeys vaccinated with either the COBRA VLP or the WS/05 VLP vaccine had a reduced gross pathology scores of 2.1-3.3 at day 3 post-infection with a milder increase in body temperature (1.1-1.3.degree. C.) that spiked between days 2-3 post-infection and then returned to pre-infection temperatures. Vaccinated animals had fewer H5N1 infected cells that were detected primarily on day 1 post-infection (Table 6).
TABLE-US-00007 TABLE 6 H5N1 lung infection scores Alveolar Submucosal Vaccine infection score infection score 1 3 5 1 3 5 1 3 5 Mock 1.00 0.05 0 1.10 0.48 0.25 0 0 0 WS/05 VLP 0.05 0 0 0.55 0.10 0 0 0 0 COBRA 0 0 0 0.60 0.03 0.05 0 0 0 VLP
[0194] ISH for influenza was performed on tissue sections of from upper and lower left and right lung. A semi-quantitative scoring system was developed to evaluate the presence of influenza infected cells. Scores were then averaged: 0.2=rare or occasional cells but <5% of fields; 1=>1/2 to 1/4 low power fields; 2=>1/4 low power fields; 3=essentially all low power fields.
[0195] However, monkeys vaccinated with the COBRA VLP had little to no signs of lung inflammation by H&E staining, while animals vaccinated with the WS/05 VLP vaccine had similar signs of inflammation as non-vaccinated monkeys (Table 7). In addition, unvaccinated monkeys had high titers of virus in both the nasal and tracheal washes between days 3 and 5 post-infection. In contrast, no virus was detected in either vaccinated groups.
TABLE-US-00008 TABLE 7 Lung involvement and inflammation scores % lung Bronchial Alveolar involvement.sup.a inflammation.sup.b inflammation.sup.b Vaccine 1 3 5 1 3 5 1 3 5 Mock 0.38 1.13 1.25 0.63 0.75 1.25 0.63 1.00 1.25 (0-1) (0-2) (0-2) (0-1) (0-2) (0-2) (0-1) (0-2) (0-2) WS/05 VLP 0.75 1.50 0.88 1.00 1.42 0.63 1.00 1.25 1.00 (0-2) (0-3) (0-3) (1) (1-2) (0-2) (0-2) (0-2) (0-2) COBRA VLP 0.88 0.50 0.38 1.13 0.75 0.88 1.13 0.67 0.25 (0-2) (0-2) (0-2) (1-2) (0-2) (0-2) (0-2) (0-2) (0-1) .sup.a% Lung involvement. Tissue sections from upper and lower left and right lung were evaluated for percent area demonstrating pneumonia. Scores were then averaged. Range in parentheses. 0 = <10%, 1 = 10-24%, 2 = 25-50%, 3 = >50%. .sup.bBronchial and alveolar inflammation scores. Tissue sections from upper and lower left and right lung were evaluated for presence of bronchial inflammation and denudation and alveolar immune cell infiltration. Scores were then averages: 0 = absent, 1 = present, 2 = abundant.
Example 3: Comparison of Protective Efficacy by Vaccination with Computationally Optimized HA and Polyvalent HA Based H5N1 VLP Vaccines
[0196] This example describes a comparison of the COBRA HA vaccine to a polyvalent H5N1 vaccine. The results demonstrate that a single COBRA antigen elicits broader antibodies and is more effective than a polyvalent mixture of primary antigens.
Materials and Methods
Vaccine Antigens and Preparation
[0197] The design and characterization of the computationally optimized broadly reactive antigen (COBRA) is described in Example 1. Polyvalent vaccine HA antigens were derived via reverse transcription from the following 6:2 reassortant H5N1 viruses: A/Indonesia/5/2005 (clade 2.1; IN/05), A/Whooper Swan/Mongolia/244/2005 (clade 2.2; WS/05) and A/Anhui/1/2005 (clade 2.3; AN/05). All HA antigens were cloned into the pTR600 expression vector.
[0198] Virus-like particles (VLPs) were generated by transiently transfecting HEK 293T cells with plasmids expressing M1 (A/Puerto Rico/8/1934), NA (A/Thailand/1(KAN-1)/2004), and a single HA for each preparation. Cells were incubated for 72 h at 37.degree. C. after which supernatants were harvested. Cell debris was cleared by low speed centrifugation followed by vacuum filtration through a 0.22 .mu.m sterile filter. VLPs were purified by ultracentrifugation (100,000.times.g through 20% glycerol, weight to volume) for 4 hours at 4.degree. C. Pellets were then resuspended in PBS pH 7.2 and stored in single use aliquots at -80.degree. C. until use. Total protein concentration was determined by MicroBCA.TM. Protein Assay Reagent Kit (Pierce Biotechnology, Rockford, Ill., USA). HA specific content of each VLP was determined by scanning densitometry as described previously (Giles and Ross, Vaccine 29:3043-3054, 2011). Briefly, purified HA matched to each VLP was electrophoresed with purified VLPs, transferred to a PVDF membrane and probed by western blot with H5-specific antisera. The relative density of the HA band in the purified protein lanes was used to calculate a standard curve and the density of the HA in the VLP lanes was interpolated. In total, four different VLP preparations were purified and HA content quantified independently, each containing one of the three wild-type influenza gene products (IN/05, WS/05, AN/05) or the COBRA HA.
Mouse Studies
[0199] BALB/c mice (Mus musculis, females, 6-8 weeks) were purchased from Harlan Sprague Dawley, (Indianapolis, Ind., USA) and housed in microisolator units and allowed free access to food and water and were cared for under USDA guidelines for laboratory animals. Mice were vaccinated with purified COBRA VLPs (3 .mu.g HA) or a polyvalent formulation of VLPs consisting of 1 .mu.g HA each IN/05, WS/05 and AN/05 (3 .mu.g HA total) via intramuscular injection at week 0 and then boosted at week 3. Vaccines were formulated with Imject.RTM. alum adjuvant (Imject.RTM. Alum, Pierce Biotechnology; Rockford, Ill., USA) according to the manufacturer's protocol. Fourteen to twenty-one days after each vaccination, blood was collected from anesthetized mice via the retro-orbital plexus and transferred to a microfuge tube. Tubes were centrifuged and sera was removed and frozen at -20.+-.5.degree. C.
[0200] Three weeks after final vaccination, mice were challenged intranasally with 5.times.10.sup.3 plaque forming units (PFU) of either highly pathogenic wild type H5N1 virus A/Whooper Swan/Mongolia/244/2005 (n=20/group) or 6:2 reassortant virus with internal genes from the mouse adapted virus A/Puerto Rico/8/1934 and the surface proteins HA and NA from A/Vietnam/1203/2004 (n=10/group) in a total volume of 50 .mu.l. Challenge doses for both viruses were established independently and represent approximately 50LD.sub.50. After infection, mice were monitored daily for weight loss, disease signs and death for 14 days after infection. Individual body weights, sickness scores and death were recorded for each group on each day after inoculation. Sickness score was determined by evaluating activity (0=normal, 1=reduced, 2=severely reduced), hunched back (0=absent, 1=present) and ruffled fur (0=absent, 1=present) (Toapanta and Ross, Respiratory Res 10(1):112, 2009). Experimental endpoint was determined by >20% weight loss or display of neurological disease such as hind limb paralysis. All highly pathogenic wild type H5N1 influenza virus studies were performed under high-containment biosafety level 3 enhanced conditions (BSL3+).
Ferret Studies
[0201] Fitch ferrets (Mustela putorius faro, female, 6-12-months of age), influenza naive and descented, were purchased from Marshall Farms (Sayre, Pa., USA). Ferrets were pair housed in stainless steel cages (Shor-line, Kansas City, Kans., USA) containing Sani-chips Laboratory Animal Bedding (P.J. Murphy Forest Products, Montville, N.J., USA). Ferrets were provided with Teklad Global Ferret Diet (Harlan Teklad, Madison, Wis., USA) and fresh water ad libitum. The VLPs were diluted in PBS, pH 7.2 to achieve final concentration. Ferrets (n=6) were vaccinated with purified COBRA VLPs (15 .mu.g HA) or a polyvalent formulation of VLPs consisting of 5 .mu.g HA each IN/05, WS/05 and AN/05 (15 .mu.g HA total) via intramuscular injection at week 0 and then boosted at week 3. Vaccines were formulated with Imject.RTM. alum adjuvant (Imject.RTM. Alum, Pierce Biotechnology; Rockford, Ill., USA) immediately prior to use according to the manufacturer's protocol. Animals were monitored for adverse events including weight loss, temperature, loss of activity, nasal discharge, sneezing and diarrhea weekly during the vaccination regimen. Prior to vaccination, animals were confirmed by HAI assay to be seronegative for circulating influenza A (H1N1 and H3N2) and influenza B viruses. Fourteen to twenty-one days after each vaccination, blood was collected from anesthetized ferrets via the anterior vena cava and transferred to a microfuge tube. Tubes were centrifuged and sera was removed and frozen at -20.+-.5.degree. C.
[0202] Three weeks after final vaccination, ferrets were challenged intranasally with 1.times.10.sup.6 plaque forming units (PFU) of the highly pathogenic H5N1 virus A/Whooper Swan/Mongolia/244/2005 (clade 2.2) in a volume of 0.5 ml in each nostril for a total infection volume of 1 ml. After infection, ferrets were monitored daily for weight loss, disease signs and death for 14 days after infection. Individual body weights, sickness scores, and death were recorded for each group on each day after inoculation. Sickness score was determined by evaluating activity (0=normal, 1=alert and active after stimulation, 2=alert but not active after stimulation, 3=neither active nor alert after stimulation), nasal discharge (0=absent, 1=present), sneezing (0=absent, 1=present), decreased food intake (0=absent, 1=present), diarrhea (0=absent, 1=present), dyspnea (0=absent, 1=present) and neurological symptoms (0=absent, 1=present) as previously described (Giles and Ross, Vaccine 29:3043-3054, 2011). Experimental endpoint was defined as >20% weight loss, development of neurological disease or an activity score of 3 (neither active nor alert after stimulation). Nasal washes were performed by instilling 3 ml of PBS into the nares of anesthetized ferrets each day for 14 days after inoculation. Washes were collected and stored at -80.degree. C. until use. All highly pathogenic wild type H5N1 influenza virus studies were performed under high-containment biosafety level 3 enhanced conditions (BSL3+).
ELISA Assay
[0203] The ELISA assay was used to assess total antibody titer to the HA. High binding, 96-well polystyrene plates (Costar; Lowell, Mass., USA) were coated overnight with 50 ng/well of recombinant HA. Coating antigens were derived from the following representative viral isolates: A/Vietnam/1203/2004 (clade 1), A/Indonesia/5/2005 (clade 2.1), A/Whooper Swan/Mongolia/244/2005 (clade 2.2) and A/Anhui/1/2005 (clade 2.3). Plates were blocked with 5% milk diluted in PBS with 0.05% Tween 20. Serum samples were diluted in blocking buffer and added to plates. Serum was two-fold serially diluted and allowed to incubate for 1 hour at room temperature. Plates were washed and species specific antibody against IgG linked to horseradish peroxidase (HRP) was diluted in blocking buffer and added to plates. Plates were incubated for 1 hour at room temperature. Plates were washed and HRP was developed with TMB substrate (Sigma-Aldrich; St. Louis, Mo., USA). Plates were incubated in the dark for 15 minutes and then the reaction was stopped with 2N H2504. Optical densities at a wavelength of 450 nm (OD.sub.450) were read by a spectrophotometer (BioTek; Winooski, Vt., USA) and end point dilution titers were determined as the reciprocal dilution of the last well which had an OD.sub.450 above the mean OD.sub.450 plus two standard deviations of naive animal sera.
Hemagglutination Inhibition (HAI) Assay
[0204] The HAI assay was used to assess functional antibodies to the HA able to inhibit agglutination of horse erythrocytes. The protocol was adapted from the CDC laboratory-based influenza surveillance manual (Gillim-Ross and Subbarao, Clin Microbiol Rev 19(4):614-636, 2006). To inactivate non-specific inhibitors, sera were treated with receptor destroying enzyme (RDE; Denka Seiken, Co., Japan) prior to being tested. Briefly, three parts RDE was added to one part sera and incubated overnight at 37.degree. C. RDE was inactivated by incubation at 56.degree. C. for .about.30 min. RDE treated sera was two-fold serially diluted in v-bottom microtiter plates. An equal volume of reassortant virus, adjusted to approximately 8 HAU/50.sub.111, was added to each well. The reassortant viruses contained the internal genes from the mouse adapted strain A/Puerto Rico/8/1934 and the surface proteins HA and NA from the following representative viral isolates: A/Vietnam/1203/2004 (clade 1), A/Indonesia/5/2005 (clade 2.1), A/Whooper Swan/Mongolia/244/2005 (clade 2.2) and A/Anhui/1/2005 (clade 2.3). The plates were covered and incubated at room temperature for 20 minutes followed by the addition of 1% horse erythrocytes (HRBC) (Lampire Biologicals, Pipersville, Pa., USA) in PBS. Red blood cells were stored at 4.degree. C. and used within 72 h of preparation. The plates were mixed by agitation, covered, and the RBCs were allowed to settle for 1 h at room temperature (Askonas B, McMichael A, Webster R. The immune response to influenza viruses and the problem of protection against infection. In: Beare AS, editor. Basic and applied influenza research: CRC Press 1982: 159-188). The HAI titer was determined by the reciprocal dilution of the last well which contained non-agglutinated RBC. Positive and negative serum controls were included for each plate. All mice and ferrets were negative (HAI.ltoreq.1:10) for pre-existing antibodies to currently circulating human influenza viruses prior to vaccination.
Plaque Assay
[0205] For mouse infections, lung virus titers were evaluated. For ferret infections, nasal wash virus titers were used to assess viral burden. Both lungs and nasal wash virus titers were determined using a plaque assay (Tobita et al., Med Microbiol Immunol 162:23-27, 1975; Tobita et al., Med Microbiol Immunol 162:9-14, 1975). Briefly, lungs from mice infected with virus were harvested post infection, snap-frozen and stored at -80.degree. C. until use. Samples were thawed, weighed and single cell suspensions were prepared via passage through a 70 .mu.m mesh (BD Falcon, Bedford, Mass., USA) in an appropriate volume of DMEM supplemented with penicillin-streptomycin (iDEME) as to achieve 100 mg/ml final concentration. Cell suspensions were centrifuged at 2000 rpm for 5 minutes and the supernatants were collected.
[0206] Madin-Darby Canine Kidney (MDCK) cells were plated (5.times.10.sup.5) in each well of a 6 well plate. Samples (lung supernatants for mice and nasal washes for ferrets) were diluted (dilution factors of 1.times.10.sup.1 to 10.sup.6) and overlayed onto the cells in 100 .mu.l of iDMEM and incubated for 1 hour. Virus-containing medium was removed and replaced with 2 ml of L15 medium plus 0.8% agarose (Cambrex, East Rutherford, N.J., USA) and incubated for 96 hours at 37.degree. C. with 5% CO2. Agarose was removed and discarded. Cells were fixed with 10% buffered formalin, and then stained with 1% crystal violet for 15 minutes. Following thorough washing in dH2O to remove excess crystal violet, plates were allowed to dry, plaques counted, and the plaque forming units (PFU)/g for or PFU/ml for nasal washes were calculated.
Histopathological Analysis
[0207] Left lobes of lungs from infected mice were collected 4 days post-infection and placed into 10% buffered formalin. After fixation, lungs were paraffin embedded and 6 .mu.m sections were prepared for histopathological analysis. For in situ hybridization (ISH), vectors containing 760 bp of Influenza/California/04/2009 matrix protein were linearized to create antisense and sense templates. .sup.35S-labeled riboprobes were generated using MAXIscript in vitro transcription kit (Ambion, Austin, Tex.). ISH was performed as described before (Bissel et al., Brain Pathol, Accepted Article doi: 10.1111/j.1750-3639.2010.00514.x). Control riboprobes did not hybridize to lung tissue at any time point post-infection and non-infected tissue did not show hybridization with viral probes. Hybridized slides were assessed and scored for abundance of foci.
Cellular Assays
[0208] The number of anti-influenza specific cells secreting interferon gamma (IFN-.gamma.) was determined by enzyme-linked immunospot (ELISpot) assay (R&D systems, Minneapolis, Minn., USA) following the manufacturer's protocol. Mice were sacrificed at 6 days post infection (DPI) and spleens and lungs were harvested and prepared in single cell suspensions. Briefly, pre-coated anti-IFN.gamma. plates were blocked with RPMI plus 10% FCS and antibiotics (cRPMI) for 30 minutes at room temperature. Media was removed from wells and 10.sup.5 cells were added to each well. Cells were stimulated with purified recombinant HA from ANietnam/1203/2004 (truncated at residue 530; 1 .mu.g/well), inactivated 6:2 reassortant virus A/Vietnam/1203/2004 (1:100 dilution of inactivated stock; 100 .mu.l/well) or the immunodominant H2-K.sup.d CD8.sup.+ T cell epitope in H5 HA: HA.sub.533 (IYSTVASSL; SEQ ID NO: 10; 1 .mu.g/well) (Pepscan Presto, Leystad, Netherlands). Additional wells were stimulated with PMA (50 ng/well) and ionomycin (500 ng/well) as positive controls or Ova257 (SIINFEKL; SEQ ID NO: 11; 1 .mu.g/well) (Pepscan Presto, Leystad, Netherlands) as negative controls. Additionally, IL-2 (10 U/ml) was added to each well. Plates were incubated at 37.degree. C. for 48 hours. After incubation, plates were washed four times with R&D wash buffer and were incubated at 4.degree. C. overnight with biotinylated anti-mouse IFN.gamma.. Plates were washed as before and incubated at room temperature for 2 hours with streptavidin conjugated to alkaline phosphatase. Plates were washed as before and spots were developed by incubating at room temperature for 1 hour in the dark with BCIP/NBT chromogen substrate. The plates were washed extensively with DI H.sub.2O and allowed to dry overnight prior to spots being counted using an ImmunoSpot ELISpot reader (Cellular Technology Ltd., Cleveland, Ohio, USA).
[0209] The number of anti-HA and anti-NA specific antibody secreting cells was determined by B cell ELISpot assay as previously described (Joo et al., Vaccine 28:2186-2194, 2009; Sasaki et al., PLoS ONE 3:e2975, 2008; Sasaki et al., J Virol 81:215-228, 2007). Mice were sacrificed at 6 DPI and spleens and lungs were harvested and prepared in single cell suspensions. Briefly, 0.45 .mu.m PVDF membrane plates (Millipore, Billerica, Mass., USA) were coated with either purified recombinant HA from A/Vietnam/1203/2004 or purified recombinant NA from A/Thailand/1(KAN-1)/2004 (250 ng/well) and incubated at 4.degree. C. overnight. Plates were washed three times with PBS and blocked with cRPMI for at 37.degree. C. for 3-4 hours. Media was removed from wells and 10.sup.5 cells were added to each well. Plates were incubated at 37.degree. C. for 48 hours. After incubation, plates were washed as before and incubated at room temperature for 2 hours with horse radish peroxidase conjugated anti-mouse IgG or IgA (Southern Biotech, Birmingham, Ala., USA). Plates were washed as before and spots were developed at room temperature for 1 hour in the dark with detection substrate (NovaRED.TM.; Vector Labs, Burlingame, Calif., USA). The plates were washed extensively with DI H.sub.2O and allowed to dry overnight prior to spots being counted using an ImmunoSpot ELISpot reader (Cellular Technology Ltd., Cleveland, Ohio, USA).
Passive Transfer of Sera
[0210] Serum from vaccinated mice was pooled and passively transferred into 9 week old recipient BALB/c mice (n=5/group). Equal amounts of serum from each mouse in a particular vaccine group were pooled and heat inactivated for 30 minutes at 56.degree. C. 200 .mu.l of pooled and inactivated serum was transferred to recipient mice via IP injection. 24 hours post transfer, mice were infected with 6:2 reassortant virus with internal genes from the mouse adapted virus A/Puerto Rico/8/1934 and surface antigens from A/Vietnam/1203/2004.
Statistical Analysis
[0211] Statistical significance of the antibody and cellular immunology data was determined using a two-tailed Student's T test to analyze differences between COBRA and polyvalent vaccine groups for each of the different test antigens. Differences in weight loss and sickness score were analyzed by two-way ANOVA, followed by Bonferroni's post test for each vaccine group at multiple time points (multiparametric). Statistical significance of viral titer data was evaluated using a two-tailed Student's T test on Log.sub.10 transformed values. Significance was defined as p<0.05. Statistical analyses were done using GraphPad Prism software.
Results
Immunogenicity in Mice and Ferrets
[0212] BALB/c mice were vaccinated twice via intramuscular injection with either purified COBRA or polyvalent VLPs and two weeks after the second vaccination serum was analyzed for antibody responses. All vaccinated mice had high titer anti-HA antibodies that bound to recombinant HA derived from both clade 1 and various sub-clades of clade 2 (FIG. 12A). Although both COBRA and polyvalent vaccines elicited similar total IgG, COBRA vaccinated animals had higher HAI antibody titers for all viruses tested (p<0.001; FIG. 12B). In addition to higher HAI titer, COBRA vaccinated mice had an increased frequency of HAI titers .gtoreq.1:40 for all viruses tested, including those which were components of the polyvalent formulation (Table 8).
[0213] To confirm the results from mice in a more rigorous animal model, ferrets were vaccinated twice via intramuscular injection with either COBRA or polyvalent vaccines. Serum was collected two weeks after the second vaccination and antibody responses were evaluated. Similar to the mice, all vaccinated ferrets had anti-HA antibodies that bound to diverse recombinant HA and the relative total IgG titers were equivalent for both COBRA and polyvalent vaccines (FIG. 12C). COBRA vaccinated ferrets demonstrated increased HAI antibody titers compared to polyvalent vaccinated animals against all viruses tested, however only the antibodies to the clade 2.1 virus were significantly different (p<0.05; FIG. 12D). Furthermore, COBRA vaccinated animals displayed an increased rate of achieving an HAI titer of .gtoreq.1:40 in comparison to the polyvalent vaccinated ferrets for all test antigens (Table 8).
TABLE-US-00009 TABLE 8 Seroconversion frequency Species Vaccine antigen Clade 1 Clade 2.1 Clade 2.2 Clade 2.3 Mouse COBRA 60% (18/30) 100% (30/30) 100% (30/30) 100% (30/30) Polyvalent 3.3% (1/30) 70% (21/30) 50% (15/30) 53% (16/30) Ferret COBRA 33% (2/6) 67% (4/6) 50% (3/6) 50% (3/6) Polyvalent 0% (0/6) 33% (2/6) 0% (0/6) 0% (0/6)
Wild Type Clade 2.2 Challenge
[0214] To confirm protective efficacy against highly pathogenic H5N1 infection, vaccinated animals were challenged with a lethal dose of the wild-type clade 2.2 isolate A/Whooper Swan/Mongolia/244/2005. All VLP vaccinated mice were protected from weight loss and death while mock vaccinated animals rapidly lost weight and reached experimental end-point by 6 days post infection (DPI; FIG. 13A). COBRA and polyvalent vaccinated mice both had a mean maximum weight loss of 4% at 12 and 13 DPI, respectively. Additionally, all VLP vaccinated mice failed to develop any overt signs of disease while mock vaccinated mice developed visible illness (FIG. 13B).
[0215] Similar to the mice, all VLP vaccinated ferrets were protected from death following a lethal challenge. Vaccinated ferrets demonstrated mild weight loss in response to the infection with COBRA vaccinated animals having mean maximum weight loss of 5.5% at 2 DPI and polyvalent vaccinated animals losing 6.8% at 3 DPI (FIG. 13C). Both groups rapidly recovered weight and failed to develop any significant signs of disease (FIG. 13D). Furthermore, VLP vaccinated animals did not demonstrate any temperature spikes while mock vaccinated animals had an elevated temperature of .about.3.degree. C. for 1-3 DPI.
[0216] To evaluate vaccine efficacy with a more sensitive output than morbidity and mortality, the viral burden of infected animals was also determined. Both COBRA and polyvalent vaccinated mice had reduced lung viral titers as quickly as 1 DPI when compared to mock vaccinated animals. Furthermore, COBRA vaccinated mice did not have detectable virus by 3 DPI while polyvalent vaccinated mice demonstrated prolonged viral replication with 1.8.times.10.sup.3 PFU/g at 3 DPI (p<0.05; FIG. 14A). Additionally, both VLP vaccines prevented extra-pulmonary spread of the virus while mock vaccinated animals had detectable virus in both kidney and liver by 3 DPI. Control of virus replication in ferrets was similar to that observed in mice, although complete clearance of the virus was delayed (FIG. 14B). All VLP vaccinated animals had decreased recovery of virus in nasal washes compared to mock vaccinated ferrets at all timepoints tested (p<0.05). COBRA vaccinated animals did not have detectable virus by 5 DPI. In contrast, virus replication did not reach undetectable levels until 9 DPI in polyvalent vaccinated ferrets.
Histopathology of Infected Lungs
[0217] To evaluate the location and severity of influenza viral antigen and viral replication, ISH for influenza A MP was scored on 3 DPI lung sections. COBRA vaccinated animals had rare bronchial epithelium infection (FIGS. 15A and 15B). Animals receiving polyvalent vaccines had occasional bronchial epithelium infection that was comparable to the COBRA vaccinated animals (FIGS. 15A and 15B). This was in contrast to significant bronchial epithelium infection and replication observed in mock animals (FIGS. 15A and 15B).
Reassortant Clade 1 Challenge
[0218] Having established the clade 2.2 protective profile of both the COBRA and polyvalent vaccines, the efficacy of these vaccines against a more divergent clade 1 challenge in mice was evaluated. COBRA and polyvalent vaccinated mice were challenged with 6:2 reassortant virus containing the HA and NA proteins from the clade 1 virus A/Vietnam/1203/2004. All VLP vaccinated animals were protected from weight loss and death while mock vaccinated animals rapidly lost weight and reached experimental endpoint by 7 DPI (FIG. 16A). Furthermore, vaccinated mice also did not develop any signs of disease throughout the course of the study (FIG. 16B). Lungs were harvested at 3 DPI for determination of viral burden (FIG. 16C). COBRA vaccinated animals did not have detectable virus while polyvalent animals had 1.1.times.10.sup.3 PFU/g virus (p=0.12). Importantly, both vaccines had significantly less recoverable virus than mock vaccinated animals at 3 DPI (p<0.01).
Post-Challenge Cellular Immune Responses
[0219] The magnitude of influenza specific cellular immune responses in the lungs post-infection was evaluated via ELISpot assay for both antibody secreting cells (ASC) and IFN-.gamma. producing cells. Vaccinated mice were infected with reassortant A/Vietnam/1203/2994 virus as before and lungs were harvested at 6 DPI. COBRA and polyvalent vaccinated animals had statistically equivalent numbers of both IgG and IgA ASC specific for HA from the challenge virus (p>0.05; FIG. 17A). No ASC were detected in mock vaccinated animals indicating that the 6 DPI time point is likely representative of a recall response. Additionally, the majority of the ASC response to infection was specific for HA as lower numbers of cells were detected for the NA component of the vaccines.
[0220] VLP vaccine primed IFN-.gamma. secreting cells were also evaluated after infection. IFN-.gamma. responses were equivalent between VLP vaccine groups regardless of stimulating antigen (p>0.05; FIG. 17B). Recombinant HA and inactivated virus were inefficient stimulators of IFN-.gamma. production compared to the HA533 peptide. HA533 is the immunodominant CD8.sup.+ T cell epitope in BALB/c mice and is conserved in all HA vaccine antigens used in this study. Overlapping peptide pools spanning the entire HA molecule were also used to stimulate cells and no differences were observed between COBRA and polyvalent vaccines for any of the pools. Similar to the ASC data, no IFN-.gamma. responses were detectable above background in mock vaccinated animals at 6 DPI.
Passive Transfer of Immune Sera
[0221] The contribution of serum factors to protection from clade 1 challenge was evaluated using a passive transfer model. Nine-week old recipient mice were administered pooled sera via IP injection from COBRA, polyvalent and mock vaccinated mice. The next day, recipient mice were challenged with the clade 1 reassortant A/Vietnam/1203/2004 virus as before. Regardless of transferred serum, all recipient mice lost weight and became visibly ill (FIGS. 18A and 18B). COBRA serum recipient mice lost less weight than polyvalent recipient mice with maximum losses of 5.2% (6 DPI) and 11.8% (7 DPI), respectively (p<0.05 at 7 DPI). COBRA serum recipient mice also began to resolve the clinical symptoms more rapidly than polyvalent recipient mice (p<0.05 at 7 DPI). Although COBRA serum prevented recipient mice from developing illness more efficiently than polyvalent serum, both COBRA and polyvalent serum protected all recipient mice from death. Conversely, all mice receiving serum from mock vaccinated mice rapidly lost weight, became visibly ill and reached experimental endpoint by 7 DPI.
[0222] In view of the many possible embodiments to which the principles of the disclosed invention may be applied, it should be recognized that the illustrated embodiments are only examples of the disclosure and should not be taken as limiting the scope of the invention. Rather, the scope of the invention is defined by the following claims. We therefore claim as our invention all that comes within the scope and spirit of these claims.
Sequence CWU 1
1
1111707DNAArtificial SequenceSynthetic polynucleotideCDS(1)..(1707)
1atg gaa aag atc gtg ctg ctg ctg gct atc gtg agc ctg gtg aag agc
48Met Glu Lys Ile Val Leu Leu Leu Ala Ile Val Ser Leu Val Lys Ser1
5 10 15gac cag att tgc atc ggc tac cac gcc aac aac agc acc gag cag
gtg 96Asp Gln Ile Cys Ile Gly Tyr His Ala Asn Asn Ser Thr Glu Gln
Val 20 25 30gac acc atc atg gaa aag aac gtc acc gtg acc cac gcc cag
gac atc 144Asp Thr Ile Met Glu Lys Asn Val Thr Val Thr His Ala Gln
Asp Ile 35 40 45ctg gaa aag acc cac aac ggc aag ctg tgc gac ctg gac
ggc gtg aag 192Leu Glu Lys Thr His Asn Gly Lys Leu Cys Asp Leu Asp
Gly Val Lys 50 55 60ccc ctg atc ctg agg gac tgc agc gtg gcc ggc tgg
ctg ctg ggc aac 240Pro Leu Ile Leu Arg Asp Cys Ser Val Ala Gly Trp
Leu Leu Gly Asn65 70 75 80ccc atg tgc gac gag ttc atc aac gtg ccc
gag tgg agc tac atc gtg 288Pro Met Cys Asp Glu Phe Ile Asn Val Pro
Glu Trp Ser Tyr Ile Val 85 90 95gag aag gcc aac ccc gcc aac gac ctg
tgc tac ccc ggc aac ttc aac 336Glu Lys Ala Asn Pro Ala Asn Asp Leu
Cys Tyr Pro Gly Asn Phe Asn 100 105 110gac tac gag gaa ctg aag cac
ctg ctg tcc agg atc aac cac ttc gag 384Asp Tyr Glu Glu Leu Lys His
Leu Leu Ser Arg Ile Asn His Phe Glu 115 120 125aag atc cag atc atc
ccc aag agc agc tgg tcc gac cac gag gcc agc 432Lys Ile Gln Ile Ile
Pro Lys Ser Ser Trp Ser Asp His Glu Ala Ser 130 135 140agc ggc gtg
agc agc gcc tgc cca tac cag ggc agc ccc agc ttc ttc 480Ser Gly Val
Ser Ser Ala Cys Pro Tyr Gln Gly Ser Pro Ser Phe Phe145 150 155
160aga aac gtg gtg tgg ctg atc aag aag aac aac acc tac ccc acc atc
528Arg Asn Val Val Trp Leu Ile Lys Lys Asn Asn Thr Tyr Pro Thr Ile
165 170 175aag agg tcc tac aac aac acc aac cag gaa gat ctg ctg gtc
ctg tgg 576Lys Arg Ser Tyr Asn Asn Thr Asn Gln Glu Asp Leu Leu Val
Leu Trp 180 185 190ggc atc cac cac cct aat gac gcc gcc gaa cag acc
agg ctg tac cag 624Gly Ile His His Pro Asn Asp Ala Ala Glu Gln Thr
Arg Leu Tyr Gln 195 200 205aac ccc acc acc tac atc agc gtg ggc aca
agc acc ctg aac cag agg 672Asn Pro Thr Thr Tyr Ile Ser Val Gly Thr
Ser Thr Leu Asn Gln Arg 210 215 220ctg gtg ccc aag atc gcc acc agg
tcc aag gtg aac gga cag tcc ggc 720Leu Val Pro Lys Ile Ala Thr Arg
Ser Lys Val Asn Gly Gln Ser Gly225 230 235 240agg atg gaa ttc ttc
tgg acc atc ctg aag cct aac gac gcc atc aac 768Arg Met Glu Phe Phe
Trp Thr Ile Leu Lys Pro Asn Asp Ala Ile Asn 245 250 255ttc gag agc
aac ggc aac ttt atc gcc ccc gag tac gcc tac aag atc 816Phe Glu Ser
Asn Gly Asn Phe Ile Ala Pro Glu Tyr Ala Tyr Lys Ile 260 265 270gtg
aag aag ggc gac agc gcc atc atg aag agc gag ctg gaa tac ggc 864Val
Lys Lys Gly Asp Ser Ala Ile Met Lys Ser Glu Leu Glu Tyr Gly 275 280
285aac tgc aac acc aag tgc cag acc ccc atc ggc gcc atc aac agc agc
912Asn Cys Asn Thr Lys Cys Gln Thr Pro Ile Gly Ala Ile Asn Ser Ser
290 295 300atg ccc ttc cac aac atc cac ccc ctg acc atc ggc gag tgc
ccc aag 960Met Pro Phe His Asn Ile His Pro Leu Thr Ile Gly Glu Cys
Pro Lys305 310 315 320tac gtg aag agc aac agg ctg gtg ctg gcc acc
ggc ctg agg aac agc 1008Tyr Val Lys Ser Asn Arg Leu Val Leu Ala Thr
Gly Leu Arg Asn Ser 325 330 335ccc cag aga gag agc aga aga aag aag
agg ggc ctg ttc ggc gct atc 1056Pro Gln Arg Glu Ser Arg Arg Lys Lys
Arg Gly Leu Phe Gly Ala Ile 340 345 350gcc ggc ttc atc gag ggc ggc
tgg cag ggc atg gtg gac ggg tgg tac 1104Ala Gly Phe Ile Glu Gly Gly
Trp Gln Gly Met Val Asp Gly Trp Tyr 355 360 365ggc tac cac cac tct
aac gag cag ggc agc ggc tac gcc gcc gac aaa 1152Gly Tyr His His Ser
Asn Glu Gln Gly Ser Gly Tyr Ala Ala Asp Lys 370 375 380gag agc acc
cag aag gcc atc gac ggc gtc acc aac aag gtg aac agc 1200Glu Ser Thr
Gln Lys Ala Ile Asp Gly Val Thr Asn Lys Val Asn Ser385 390 395
400atc atc gac aag atg aac acc cag ttc gag gcc gtg ggc aga gag ttc
1248Ile Ile Asp Lys Met Asn Thr Gln Phe Glu Ala Val Gly Arg Glu Phe
405 410 415aac aac ctg gaa agg cgg atc gag aac ctg aac aag aaa atg
gaa gat 1296Asn Asn Leu Glu Arg Arg Ile Glu Asn Leu Asn Lys Lys Met
Glu Asp 420 425 430ggc ttc ctg gac gtg tgg acc tac aac gcc gag ctg
ctg gtg ctg atg 1344Gly Phe Leu Asp Val Trp Thr Tyr Asn Ala Glu Leu
Leu Val Leu Met 435 440 445gaa aac gag agg acc ctg gac ttc cac gac
agc aac gtg aag aac ctg 1392Glu Asn Glu Arg Thr Leu Asp Phe His Asp
Ser Asn Val Lys Asn Leu 450 455 460tac gac aaa gtg cgg ctg cag ctg
agg gac aac gcc aaa gag ctg ggc 1440Tyr Asp Lys Val Arg Leu Gln Leu
Arg Asp Asn Ala Lys Glu Leu Gly465 470 475 480aac ggc tgc ttc gag
ttc tac cac aag tgc gac aac gag tgc atg gaa 1488Asn Gly Cys Phe Glu
Phe Tyr His Lys Cys Asp Asn Glu Cys Met Glu 485 490 495agc gtg agg
aac ggc acc tac gac tac ccc cag tac agc gag gaa gcc 1536Ser Val Arg
Asn Gly Thr Tyr Asp Tyr Pro Gln Tyr Ser Glu Glu Ala 500 505 510agg
ctg aag agg gaa gag atc agc gga gtg aag ctg gaa agc atc ggc 1584Arg
Leu Lys Arg Glu Glu Ile Ser Gly Val Lys Leu Glu Ser Ile Gly 515 520
525acc tac cag atc ctg agc atc tac agc acc gtc gcc agc agc ctg gcc
1632Thr Tyr Gln Ile Leu Ser Ile Tyr Ser Thr Val Ala Ser Ser Leu Ala
530 535 540ctg gct atc atg gtg gcc gga ctg agc ctg tgg atg tgc agc
aac ggc 1680Leu Ala Ile Met Val Ala Gly Leu Ser Leu Trp Met Cys Ser
Asn Gly545 550 555 560agc ctg cag tgc agg atc tgc atc tga 1707Ser
Leu Gln Cys Arg Ile Cys Ile 5652568PRTArtificial SequenceSynthetic
Construct 2Met Glu Lys Ile Val Leu Leu Leu Ala Ile Val Ser Leu Val
Lys Ser1 5 10 15Asp Gln Ile Cys Ile Gly Tyr His Ala Asn Asn Ser Thr
Glu Gln Val 20 25 30Asp Thr Ile Met Glu Lys Asn Val Thr Val Thr His
Ala Gln Asp Ile 35 40 45Leu Glu Lys Thr His Asn Gly Lys Leu Cys Asp
Leu Asp Gly Val Lys 50 55 60Pro Leu Ile Leu Arg Asp Cys Ser Val Ala
Gly Trp Leu Leu Gly Asn65 70 75 80Pro Met Cys Asp Glu Phe Ile Asn
Val Pro Glu Trp Ser Tyr Ile Val 85 90 95Glu Lys Ala Asn Pro Ala Asn
Asp Leu Cys Tyr Pro Gly Asn Phe Asn 100 105 110Asp Tyr Glu Glu Leu
Lys His Leu Leu Ser Arg Ile Asn His Phe Glu 115 120 125Lys Ile Gln
Ile Ile Pro Lys Ser Ser Trp Ser Asp His Glu Ala Ser 130 135 140Ser
Gly Val Ser Ser Ala Cys Pro Tyr Gln Gly Ser Pro Ser Phe Phe145 150
155 160Arg Asn Val Val Trp Leu Ile Lys Lys Asn Asn Thr Tyr Pro Thr
Ile 165 170 175Lys Arg Ser Tyr Asn Asn Thr Asn Gln Glu Asp Leu Leu
Val Leu Trp 180 185 190Gly Ile His His Pro Asn Asp Ala Ala Glu Gln
Thr Arg Leu Tyr Gln 195 200 205Asn Pro Thr Thr Tyr Ile Ser Val Gly
Thr Ser Thr Leu Asn Gln Arg 210 215 220Leu Val Pro Lys Ile Ala Thr
Arg Ser Lys Val Asn Gly Gln Ser Gly225 230 235 240Arg Met Glu Phe
Phe Trp Thr Ile Leu Lys Pro Asn Asp Ala Ile Asn 245 250 255Phe Glu
Ser Asn Gly Asn Phe Ile Ala Pro Glu Tyr Ala Tyr Lys Ile 260 265
270Val Lys Lys Gly Asp Ser Ala Ile Met Lys Ser Glu Leu Glu Tyr Gly
275 280 285Asn Cys Asn Thr Lys Cys Gln Thr Pro Ile Gly Ala Ile Asn
Ser Ser 290 295 300Met Pro Phe His Asn Ile His Pro Leu Thr Ile Gly
Glu Cys Pro Lys305 310 315 320Tyr Val Lys Ser Asn Arg Leu Val Leu
Ala Thr Gly Leu Arg Asn Ser 325 330 335Pro Gln Arg Glu Ser Arg Arg
Lys Lys Arg Gly Leu Phe Gly Ala Ile 340 345 350Ala Gly Phe Ile Glu
Gly Gly Trp Gln Gly Met Val Asp Gly Trp Tyr 355 360 365Gly Tyr His
His Ser Asn Glu Gln Gly Ser Gly Tyr Ala Ala Asp Lys 370 375 380Glu
Ser Thr Gln Lys Ala Ile Asp Gly Val Thr Asn Lys Val Asn Ser385 390
395 400Ile Ile Asp Lys Met Asn Thr Gln Phe Glu Ala Val Gly Arg Glu
Phe 405 410 415Asn Asn Leu Glu Arg Arg Ile Glu Asn Leu Asn Lys Lys
Met Glu Asp 420 425 430Gly Phe Leu Asp Val Trp Thr Tyr Asn Ala Glu
Leu Leu Val Leu Met 435 440 445Glu Asn Glu Arg Thr Leu Asp Phe His
Asp Ser Asn Val Lys Asn Leu 450 455 460Tyr Asp Lys Val Arg Leu Gln
Leu Arg Asp Asn Ala Lys Glu Leu Gly465 470 475 480Asn Gly Cys Phe
Glu Phe Tyr His Lys Cys Asp Asn Glu Cys Met Glu 485 490 495Ser Val
Arg Asn Gly Thr Tyr Asp Tyr Pro Gln Tyr Ser Glu Glu Ala 500 505
510Arg Leu Lys Arg Glu Glu Ile Ser Gly Val Lys Leu Glu Ser Ile Gly
515 520 525Thr Tyr Gln Ile Leu Ser Ile Tyr Ser Thr Val Ala Ser Ser
Leu Ala 530 535 540Leu Ala Ile Met Val Ala Gly Leu Ser Leu Trp Met
Cys Ser Asn Gly545 550 555 560Ser Leu Gln Cys Arg Ile Cys Ile
56531350DNAArtificial SequenceSynthetic
polynucleotideCDS(1)..(1350) 3atg aat cct aat aag aag atc atc aca
atc gga agc atc tgc atg gtg 48Met Asn Pro Asn Lys Lys Ile Ile Thr
Ile Gly Ser Ile Cys Met Val1 5 10 15aca gga atg gtg agc ctg atg ctg
cag atc gga aat ctg atc agc atc 96Thr Gly Met Val Ser Leu Met Leu
Gln Ile Gly Asn Leu Ile Ser Ile 20 25 30tgg gtg agc cac agc atc cac
aca gga aat cag cac aag gcc gag cct 144Trp Val Ser His Ser Ile His
Thr Gly Asn Gln His Lys Ala Glu Pro 35 40 45atc agc aat aca aat ttt
ctg aca gag aag gcc gtg gcc agc gtg aag 192Ile Ser Asn Thr Asn Phe
Leu Thr Glu Lys Ala Val Ala Ser Val Lys 50 55 60ctg gcc gga aat agc
agc ctg tgc cct atc aat gga tgg gcc gtg tac 240Leu Ala Gly Asn Ser
Ser Leu Cys Pro Ile Asn Gly Trp Ala Val Tyr65 70 75 80agc aag gat
aat agc atc aga atc gga agc aag gga gat gtg ttt gtg 288Ser Lys Asp
Asn Ser Ile Arg Ile Gly Ser Lys Gly Asp Val Phe Val 85 90 95atc aga
gag cct ttt atc agc tgc agc cac ctg gag tgc aga aca ttt 336Ile Arg
Glu Pro Phe Ile Ser Cys Ser His Leu Glu Cys Arg Thr Phe 100 105
110ttt ctg aca cag gga gcc ctg ctg aat gat aag cac agc aat gga aca
384Phe Leu Thr Gln Gly Ala Leu Leu Asn Asp Lys His Ser Asn Gly Thr
115 120 125gtg aag gat aga agc cct cac aga aca ctg atg agc tgc cct
gtg gga 432Val Lys Asp Arg Ser Pro His Arg Thr Leu Met Ser Cys Pro
Val Gly 130 135 140gag gcc cct agc cct tac aat agc aga ttt gag agc
gtg gcc tgg agc 480Glu Ala Pro Ser Pro Tyr Asn Ser Arg Phe Glu Ser
Val Ala Trp Ser145 150 155 160gcc agc gcc tgc cac gat gga aca agc
tgg ctg aca atc gga atc agc 528Ala Ser Ala Cys His Asp Gly Thr Ser
Trp Leu Thr Ile Gly Ile Ser 165 170 175gga cct gat aat gga gcc gtg
gcc gtg ctg aag tac aat gga atc atc 576Gly Pro Asp Asn Gly Ala Val
Ala Val Leu Lys Tyr Asn Gly Ile Ile 180 185 190aca gat aca atc aag
agc tgg aga aat aat atc ctg aga aca cag gag 624Thr Asp Thr Ile Lys
Ser Trp Arg Asn Asn Ile Leu Arg Thr Gln Glu 195 200 205agc gag tgc
gcc tgc gtg aat gga agc tgc ttt aca gtg atg aca gat 672Ser Glu Cys
Ala Cys Val Asn Gly Ser Cys Phe Thr Val Met Thr Asp 210 215 220gga
cct agc aat gga cag gcc agc cac aag atc ttt aag atg gag aag 720Gly
Pro Ser Asn Gly Gln Ala Ser His Lys Ile Phe Lys Met Glu Lys225 230
235 240gga aag gtg gtg aag agc gtg gag ctg gat gcc cct aat tac cac
tac 768Gly Lys Val Val Lys Ser Val Glu Leu Asp Ala Pro Asn Tyr His
Tyr 245 250 255gag gag tgc agc tgc tac cct gat gcc gga gag atc aca
tgc gtg tgc 816Glu Glu Cys Ser Cys Tyr Pro Asp Ala Gly Glu Ile Thr
Cys Val Cys 260 265 270aga gat aat tgg cac gga agc aat aga cct tgg
gtg agc ttt aat cag 864Arg Asp Asn Trp His Gly Ser Asn Arg Pro Trp
Val Ser Phe Asn Gln 275 280 285aat ctg gag tac cag atc gga tac atc
tgc agc gga gtg ttt gga gat 912Asn Leu Glu Tyr Gln Ile Gly Tyr Ile
Cys Ser Gly Val Phe Gly Asp 290 295 300aat cct aga cct aat gat gga
aca gga agc tgc gga cct gtg agc agc 960Asn Pro Arg Pro Asn Asp Gly
Thr Gly Ser Cys Gly Pro Val Ser Ser305 310 315 320aat gga gcc tac
gga gtg aag gga ttt agc ttt aag tac gga aat gga 1008Asn Gly Ala Tyr
Gly Val Lys Gly Phe Ser Phe Lys Tyr Gly Asn Gly 325 330 335gtg tgg
atc gga aga aca aag agc aca aat agc aga agc gga ttt gag 1056Val Trp
Ile Gly Arg Thr Lys Ser Thr Asn Ser Arg Ser Gly Phe Glu 340 345
350atg atc tgg gac cct aat gga tgg aca gag aca gat agc agc ttt agc
1104Met Ile Trp Asp Pro Asn Gly Trp Thr Glu Thr Asp Ser Ser Phe Ser
355 360 365gtg aag cag gat atc gtg gcc atc aca gat tgg agc gga tac
agc gga 1152Val Lys Gln Asp Ile Val Ala Ile Thr Asp Trp Ser Gly Tyr
Ser Gly 370 375 380agc ttt gtg cag cac cct gag ctg aca gga ctg gat
tgc atc aga cct 1200Ser Phe Val Gln His Pro Glu Leu Thr Gly Leu Asp
Cys Ile Arg Pro385 390 395 400tgc ttt tgg gtg gag ctg atc aga gga
aga cct aag gag agc aca atc 1248Cys Phe Trp Val Glu Leu Ile Arg Gly
Arg Pro Lys Glu Ser Thr Ile 405 410 415tgg aca agc gga agc agc atc
agc ttt tgc gga gtg aat agc gat aca 1296Trp Thr Ser Gly Ser Ser Ile
Ser Phe Cys Gly Val Asn Ser Asp Thr 420 425 430gtg gga tgg agc tgg
cct gat gga gcc gag ctg cct ttt aca atc gat 1344Val Gly Trp Ser Trp
Pro Asp Gly Ala Glu Leu Pro Phe Thr Ile Asp 435 440 445aag tga
1350Lys4449PRTArtificial SequenceSynthetic Construct 4Met Asn Pro
Asn Lys Lys Ile Ile Thr Ile Gly Ser Ile Cys Met Val1 5 10 15Thr Gly
Met Val Ser Leu Met Leu Gln Ile Gly Asn Leu Ile Ser Ile 20 25 30Trp
Val Ser His Ser Ile His Thr Gly Asn Gln His Lys Ala Glu Pro 35 40
45Ile Ser Asn Thr Asn Phe Leu Thr Glu Lys Ala Val Ala Ser Val Lys
50 55 60Leu Ala Gly Asn Ser Ser Leu Cys Pro Ile Asn Gly Trp Ala Val
Tyr65 70 75 80Ser Lys Asp Asn Ser Ile Arg Ile Gly Ser Lys Gly Asp
Val Phe Val 85 90 95Ile Arg Glu Pro Phe Ile Ser Cys Ser His Leu Glu
Cys Arg Thr Phe 100 105 110Phe Leu Thr Gln Gly Ala Leu Leu Asn Asp
Lys His Ser Asn Gly Thr 115 120 125Val Lys Asp Arg Ser Pro His Arg
Thr Leu Met Ser Cys Pro Val Gly 130 135 140Glu Ala Pro Ser Pro Tyr
Asn Ser Arg Phe Glu Ser Val Ala Trp Ser145 150 155 160Ala Ser Ala
Cys His Asp Gly Thr Ser Trp Leu Thr Ile Gly Ile Ser 165 170 175Gly
Pro Asp Asn Gly Ala Val Ala Val Leu Lys Tyr Asn Gly Ile Ile 180 185
190Thr Asp Thr Ile Lys Ser Trp Arg Asn Asn Ile Leu Arg Thr Gln Glu
195 200 205Ser Glu Cys Ala Cys Val Asn Gly Ser Cys Phe Thr Val Met
Thr Asp 210 215 220Gly Pro Ser Asn Gly Gln Ala Ser His Lys Ile
Phe
Lys Met Glu Lys225 230 235 240Gly Lys Val Val Lys Ser Val Glu Leu
Asp Ala Pro Asn Tyr His Tyr 245 250 255Glu Glu Cys Ser Cys Tyr Pro
Asp Ala Gly Glu Ile Thr Cys Val Cys 260 265 270Arg Asp Asn Trp His
Gly Ser Asn Arg Pro Trp Val Ser Phe Asn Gln 275 280 285Asn Leu Glu
Tyr Gln Ile Gly Tyr Ile Cys Ser Gly Val Phe Gly Asp 290 295 300Asn
Pro Arg Pro Asn Asp Gly Thr Gly Ser Cys Gly Pro Val Ser Ser305 310
315 320Asn Gly Ala Tyr Gly Val Lys Gly Phe Ser Phe Lys Tyr Gly Asn
Gly 325 330 335Val Trp Ile Gly Arg Thr Lys Ser Thr Asn Ser Arg Ser
Gly Phe Glu 340 345 350Met Ile Trp Asp Pro Asn Gly Trp Thr Glu Thr
Asp Ser Ser Phe Ser 355 360 365Val Lys Gln Asp Ile Val Ala Ile Thr
Asp Trp Ser Gly Tyr Ser Gly 370 375 380Ser Phe Val Gln His Pro Glu
Leu Thr Gly Leu Asp Cys Ile Arg Pro385 390 395 400Cys Phe Trp Val
Glu Leu Ile Arg Gly Arg Pro Lys Glu Ser Thr Ile 405 410 415Trp Thr
Ser Gly Ser Ser Ile Ser Phe Cys Gly Val Asn Ser Asp Thr 420 425
430Val Gly Trp Ser Trp Pro Asp Gly Ala Glu Leu Pro Phe Thr Ile Asp
435 440 445Lys5759DNAArtificial SequenceSynthetic
polynucleotideCDS(1)..(759) 5atg agc ctg ctg acc gag gtg gag aca
tac gtg ctg tcc atc atc ccc 48Met Ser Leu Leu Thr Glu Val Glu Thr
Tyr Val Leu Ser Ile Ile Pro1 5 10 15agc ggc cct ctg aag gcc gag atc
gcc cag aga ctg gaa gat gtg ttc 96Ser Gly Pro Leu Lys Ala Glu Ile
Ala Gln Arg Leu Glu Asp Val Phe 20 25 30gcc ggc aag aac acc gac ctg
gaa gtg ctg atg gaa tgg ctg aaa acc 144Ala Gly Lys Asn Thr Asp Leu
Glu Val Leu Met Glu Trp Leu Lys Thr 35 40 45aga ccc atc ctg agc cct
ctg acc aag ggc atc ctg ggc ttc gtg ttc 192Arg Pro Ile Leu Ser Pro
Leu Thr Lys Gly Ile Leu Gly Phe Val Phe 50 55 60acc ctg acc gtg ccc
agc gag aga ggc ctg cag agg cgg aga ttc gtg 240Thr Leu Thr Val Pro
Ser Glu Arg Gly Leu Gln Arg Arg Arg Phe Val65 70 75 80cag aac gcc
ctg aac ggc aac ggc gac ccc aac aac atg gac aag gcc 288Gln Asn Ala
Leu Asn Gly Asn Gly Asp Pro Asn Asn Met Asp Lys Ala 85 90 95gtg aag
ctg tac aga aag ctg aag cgg gag atc acc ttc cac ggc gcc 336Val Lys
Leu Tyr Arg Lys Leu Lys Arg Glu Ile Thr Phe His Gly Ala 100 105
110aaa gag atc agc ctg agc tac agc gct ggc gcc ctg gcc agc tgc atg
384Lys Glu Ile Ser Leu Ser Tyr Ser Ala Gly Ala Leu Ala Ser Cys Met
115 120 125ggc ctg atc tac aac aga atg ggc gcc gtg acc acc gag gtg
gcc ttc 432Gly Leu Ile Tyr Asn Arg Met Gly Ala Val Thr Thr Glu Val
Ala Phe 130 135 140ggc ctg gtc tgc gcc acc tgc gag cag atc gcc gac
agc cag cac aga 480Gly Leu Val Cys Ala Thr Cys Glu Gln Ile Ala Asp
Ser Gln His Arg145 150 155 160tcc cac aga cag atg gtc acc acc acc
aac ccc ctg atc aga cac gag 528Ser His Arg Gln Met Val Thr Thr Thr
Asn Pro Leu Ile Arg His Glu 165 170 175aac aga atg gtg ctg gcc tct
acc acc gcc aag gcc atg gaa cag atg 576Asn Arg Met Val Leu Ala Ser
Thr Thr Ala Lys Ala Met Glu Gln Met 180 185 190gcc ggc agc agc gag
cag gcc gcc gag gct atg gaa gtc gcc tct cag 624Ala Gly Ser Ser Glu
Gln Ala Ala Glu Ala Met Glu Val Ala Ser Gln 195 200 205gct agg cag
atg gtc cag gcc atg aga acc atc ggc acc cac ccc agc 672Ala Arg Gln
Met Val Gln Ala Met Arg Thr Ile Gly Thr His Pro Ser 210 215 220agc
tct gct ggc ctg aag aac gac ctg ctg gaa aac ctg cag gcc tac 720Ser
Ser Ala Gly Leu Lys Asn Asp Leu Leu Glu Asn Leu Gln Ala Tyr225 230
235 240cag aaa aga atg ggc gtc cag atg cag aga ttc aag tga 759Gln
Lys Arg Met Gly Val Gln Met Gln Arg Phe Lys 245
2506252PRTArtificial SequenceSynthetic Construct 6Met Ser Leu Leu
Thr Glu Val Glu Thr Tyr Val Leu Ser Ile Ile Pro1 5 10 15Ser Gly Pro
Leu Lys Ala Glu Ile Ala Gln Arg Leu Glu Asp Val Phe 20 25 30Ala Gly
Lys Asn Thr Asp Leu Glu Val Leu Met Glu Trp Leu Lys Thr 35 40 45Arg
Pro Ile Leu Ser Pro Leu Thr Lys Gly Ile Leu Gly Phe Val Phe 50 55
60Thr Leu Thr Val Pro Ser Glu Arg Gly Leu Gln Arg Arg Arg Phe Val65
70 75 80Gln Asn Ala Leu Asn Gly Asn Gly Asp Pro Asn Asn Met Asp Lys
Ala 85 90 95Val Lys Leu Tyr Arg Lys Leu Lys Arg Glu Ile Thr Phe His
Gly Ala 100 105 110Lys Glu Ile Ser Leu Ser Tyr Ser Ala Gly Ala Leu
Ala Ser Cys Met 115 120 125Gly Leu Ile Tyr Asn Arg Met Gly Ala Val
Thr Thr Glu Val Ala Phe 130 135 140Gly Leu Val Cys Ala Thr Cys Glu
Gln Ile Ala Asp Ser Gln His Arg145 150 155 160Ser His Arg Gln Met
Val Thr Thr Thr Asn Pro Leu Ile Arg His Glu 165 170 175Asn Arg Met
Val Leu Ala Ser Thr Thr Ala Lys Ala Met Glu Gln Met 180 185 190Ala
Gly Ser Ser Glu Gln Ala Ala Glu Ala Met Glu Val Ala Ser Gln 195 200
205Ala Arg Gln Met Val Gln Ala Met Arg Thr Ile Gly Thr His Pro Ser
210 215 220Ser Ser Ala Gly Leu Lys Asn Asp Leu Leu Glu Asn Leu Gln
Ala Tyr225 230 235 240Gln Lys Arg Met Gly Val Gln Met Gln Arg Phe
Lys 245 25075511DNAArtificial SequenceSynthetic construct
7cgacaatatt ggctattggc cattgcatac gttgtatcta tatcataata tgtacattta
60tattggctca tgtccaatat gaccgccatg ttgacattga ttattgacta gttattaata
120gtaatcaatt acggggtcat tagttcatag cccatatatg gagttccgcg
ttacataact 180tacggtaaat ggcccgcctc gtgaccgccc aacgaccccc
gcccattgac gtcaataatg 240acgtatgttc ccatagtaac gccaataggg
actttccatt gacgtcaatg ggtggagtat 300ttacggtaaa ctgcccactt
ggcagtacat caagtgtatc atatgccaag tccgccccta 360ttgacgtcaa
tgacggtaaa tggcccgcct ggcattatgc ccagtacatg accttacggg
420actttcctac ttggcagtac atctacgtat tagtcatcgc tattaccatg
gtgatgcggt 480tttggcagta caccaatggg cgtggatagc ggtttgactc
acggggattt ccaagtctcc 540accccattga cgtcaatggg agtttgtttt
ggcaccaaaa tcaacgggac tttccaaaat 600gtcgtaataa ccccgccccg
ttgacgcaaa tgggcggtag gcgtgtacgg tgggaggtct 660atataagcag
agctcgttta gtgaaccgtc agatcgcctg gagacgccat ccacgctgtt
720ttgacctcca tagaagacac cgggaccgat ccagcctccg cggccgggaa
cggtgcattg 780gaacgcggat tccccgtgcc aagagtgacg taagtaccgc
ctatagactc tataggcaca 840cccctttggc tcttatgcat gctatactgt
ttttggcttg gggcctatac acccccgctc 900cttatgctat aggtgatggt
atagcttagc ctataggtgt gggttattga ccattattga 960ccactcccct
attggtgacg atactttcca ttactaatcc ataacatggc tctttgccac
1020aactatctct attggctata tgccaatact ctgtccttca gagactgaca
cggactctgt 1080atttttacag gatggggtcc catttattat ttacaaattc
acatatacaa caacgccgtc 1140ccccgtgccc gcagttttta ttaaacatag
cgtgggatct ccacgcgaat ctcgggtacg 1200tgttccggac atgggctctt
ctccggtagc ggcggagctt ccacatccga gccctggtcc 1260catgcctcca
gcggctcatg gtcgctcggc agctccttgc tcctaacagt ggaggccaga
1320cttaggcaca gcacaatgcc caccaccacc agtgtgccgc acaaggccgt
ggcggtaggg 1380tatgtgtctg aaaatgagct cggagattgg gctcgcaccg
tgacgcagat ggaagactta 1440aggcagcggc agaagaagat gcaggcagct
gagttgttgt attctgataa gagtcagagg 1500taactcccgt tgcggtgctg
ttaacggtgg agggcagtgt agtctgagca gtactcgttg 1560ctgccgcgcg
cgccaccaga cataatagct gacagactaa cagactgttc ctttccatgg
1620gtcttttctg cagtcaccgt ccaagcttat ggaaaagatc gtgctgctgc
tggctatcgt 1680gagcctggtg aagagcgacc agatttgcat cggctaccac
gccaacaaca gcaccgagca 1740ggtggacacc atcatggaaa agaacgtcac
cgtgacccac gcccaggaca tcctggaaaa 1800gacccacaac ggcaagctgt
gcgacctgga cggcgtgaag cccctgatcc tgagggactg 1860cagcgtggcc
ggctggctgc tgggcaaccc catgtgcgac gagttcatca acgtgcccga
1920gtggagctac atcgtggaga aggccaaccc cgccaacgac ctgtgctacc
ccggcaactt 1980caacgactac gaggaactga agcacctgct gtccaggatc
aaccacttcg agaagatcca 2040gatcatcccc aagagcagct ggtccgacca
cgaggccagc agcggcgtga gcagcgcctg 2100cccataccag ggcagcccca
gcttcttcag aaacgtggtg tggctgatca agaagaacaa 2160cacctacccc
accatcaaga ggtcctacaa caacaccaac caggaagatc tgctggtcct
2220gtggggcatc caccacccta atgacgccgc cgaacagacc aggctgtacc
agaaccccac 2280cacctacatc agcgtgggca caagcaccct gaaccagagg
ctggtgccca agatcgccac 2340caggtccaag gtgaacggac agtccggcag
gatggaattc ttctggacca tcctgaagcc 2400taacgacgcc atcaacttcg
agagcaacgg caactttatc gcccccgagt acgcctacaa 2460gatcgtgaag
aagggcgaca gcgccatcat gaagagcgag ctggaatacg gcaactgcaa
2520caccaagtgc cagaccccca tcggcgccat caacagcagc atgcccttcc
acaacatcca 2580ccccctgacc atcggcgagt gccccaagta cgtgaagagc
aacaggctgg tgctggccac 2640cggcctgagg aacagccccc agagagagag
cagaagaaag aagaggggcc tgttcggcgc 2700tatcgccggc ttcatcgagg
gcggctggca gggcatggtg gacgggtggt acggctacca 2760ccactctaac
gagcagggca gcggctacgc cgccgacaaa gagagcaccc agaaggccat
2820cgacggcgtc accaacaagg tgaacagcat catcgacaag atgaacaccc
agttcgaggc 2880cgtgggcaga gagttcaaca acctggaaag gcggatcgag
aacctgaaca agaaaatgga 2940agatggcttc ctggacgtgt ggacctacaa
cgccgagctg ctggtgctga tggaaaacga 3000gaggaccctg gacttccacg
acagcaacgt gaagaacctg tacgacaaag tgcggctgca 3060gctgagggac
aacgccaaag agctgggcaa cggctgcttc gagttctacc acaagtgcga
3120caacgagtgc atggaaagcg tgaggaacgg cacctacgac tacccccagt
acagcgagga 3180agccaggctg aagagggaag agatcagcgg agtgaagctg
gaaagcatcg gcacctacca 3240gatcctgagc atctacagca ccgtcgccag
cagcctggcc ctggctatca tggtggccgg 3300actgagcctg tggatgtgca
gcaacggcag cctgcagtgc aggatctgca tcggatcctc 3360gcaatcccta
gggctgtgcc ttctagttgc cagccaaact gttgtttgcc cctcccccgt
3420gccttccttg accctggaag gtgccactcc cactgtcctt tcctaataaa
atgaggaaat 3480tgcatcgcat tgtctgagta ggtgtcattc tattctgggg
ggtggggtgg ggcaggacag 3540caagggggag gattgggaag acaatagcag
gcatgctggg gatgcggtgg gctctatgtt 3600cagaacgctc ggttgccgcc
gggcgttttt tatctagagt cgacaaattc agaagaactc 3660gtcaagaagg
cgatagaagg cgatgcgctg cgaatcggga gcggcgatac cgtaaagcac
3720gaggaagcgg tcagcccatt cgccgccaag ctcttcagca atatcacggg
tagccaacgc 3780tatgtcctga tagcggtccg ccacacccag ccggccacag
tcgatgaatc cagaaaagcg 3840gccattttcc accatgatat tcggcaagca
ggcatcgcca tgggtcacga cgagatcctc 3900gccgtcgggc atgctcgcct
tgagcctggc gaacagttcg gctggcgcga gcccctgatg 3960ctcttcgtcc
agatcatcct gatcgacaag accggcttcc atccgagtac gtgctcgctc
4020gatgcgatgt ttcgcttggt ggtcgaatgg gcaggtagcc ggatcaagcg
tatgcagccg 4080ccgcattgca tcagccatga tggatacttt ctcggcagga
gcaaggtgag atgacaggag 4140atcctgcccc ggcacttcgc ccaatagcag
ccagtccctt cccgcttcag tgacaacgtc 4200gagcacagct gcgcaaggaa
cgcccgtcgt ggccagccac gatagccgcg ctgcctcgtc 4260ttgcagttca
ttcagggcac cggacaggtc ggtcttgaca aaaagaaccg ggcgcccctg
4320cgctgacagc cggaacacgg cggcatcaga gcagccgatt gtctgttgtg
cccagtcata 4380gccgaatagc ctctccaccc aagcggccgg agaacctgcg
tgcaatccat cttgttcaat 4440catgcgaaac gatcctcatc ctgtctcttg
atcagatctt gatcccctgc gccatcagat 4500ccttggcggc gagaaagcca
tccagtttac tttgcagggc ttcccaacct taccagaggg 4560cgccccagct
ggcaattccg gttcgcttgc tgtccataaa accgcccagt ctagctatcg
4620ccatgtaagc ccactgcaag ctacctgctt tctctttgcg cttgcgtttt
cccttgtcca 4680gatagcccag tagctgacat tcatccgggg tcagcaccgt
ttctgcggac tggctttcta 4740cgtgaaaagg atctaggtga agatcctttt
tgataatctc atgaccaaaa tcccttaacg 4800tgagttttcg ttccactgag
cgtcagaccc cgtagaaaag atcaaaggat cttcttgaga 4860tccttttttt
ctgcgcgtaa tctgctgctt gcaaacaaaa aaaccaccgc taccagcggt
4920ggtttgtttg ccggatcaag agctaccaac tctttttccg aaggtaactg
gcttcagcag 4980agcgcagata ccaaatactg tccttctagt gtagccgtag
ttaggccacc acttcaagaa 5040ctctgtagca ccgcctacat acctcgctct
gctaatcctg ttaccagtgg ctgctgccag 5100tggcgataag tcgtgtctta
ccgggttgga ctcaagacga tagttaccgg ataaggcgca 5160gcggtcgggc
tgaacggggg gttcgtgcac acagcccagc ttggagcgaa cgacctacac
5220cgaactgaga tacctacagc gtgagctatg agaaagcgcc acgcttcccg
aagggagaaa 5280ggcggacagg tatccggtaa gcggcagggt cggaacagga
gagcgcacga gggagcttcc 5340agggggaaac gcctggtatc tttatagtcc
tgtcgggttt cgccacctct gacttgagcg 5400tcgatttttg tgatgctcgt
caggggggcg gagcctatgg aaaaacgcca gcaacgcggc 5460ctttttacgg
ttcctgggct tttgctggcc ttttgctcac atgttgtcga c
551185769DNAArtificial SequenceSynthetic construct 8tcgcgcgttt
cggtgatgac ggtgaaaacc tctgacacat gcagctcccg gagacggtca 60cagcttgtct
gtaagcggat gccgggagca gacaagcccg tcagggcgcg tcagcgggtg
120ttggcgggtg tcggggctgg cttaactatg cggcatcaga gcagattgta
ctgagagtgc 180accatatgcg gtgtgaaata ccgcacagat gcgtaaggag
aaaataccgc atcagattgg 240ctattggcca ttgcatacgt tgtatccata
tcataatatg tacatttata ttggctcatg 300tccaacatta ccgccatgtt
gacattgatt attgactagt tattaatagt aatcaattac 360ggggtcatta
gttcatagcc catatatgga gttccgcgtt acataactta cggtaaatgg
420cccgcctggc tgaccgccca acgacccccg cccattgacg tcaataatga
cgtatgttcc 480catagtaacg ccaataggga ctttccattg acgtcaatgg
gtggagtatt tacggtaaac 540tgcccacttg gcagtacatc aagtgtatca
tatgccaagt acgcccccta ttgacgtcaa 600tgacggtaaa tggcccgcct
ggcattatgc ccagtacatg accttatggg actttcctac 660ttggcagtac
atctacgtat tagtcatcgc tattaccatg gtgatgcggt tttggcagta
720catcaatggg cgtggatagc ggtttgactc acggggattt ccaagtctcc
accccattga 780cgtcaatggg agtttgtttt ggcaccaaaa tcaacgggac
tttccaaaat gtcgtaacaa 840ctccgcccca ttgacgcaaa tgggcggtag
gcgtgtacgg tgggaggtct atataagcag 900agctcgttta gtgaaccgtc
agatcgcctg gagacgccat ccacgctgtt ttgacctcca 960tagaagacac
cgggaccgat ccagcctcca tcggctcgca tctctccttc acgcgcccgc
1020cgccctacct gaggccgcca tccacgccgg ttgagtcgcg ttctgccgcc
tcccgcctgt 1080ggtgcctcct gaactgcgtc cgccgtctag gtaagtttaa
agctcaggtc gagaccgggc 1140ctttgtccgg cgctcccttg gagcctacct
agactcagcc ggctctccac gctttgcctg 1200accctgcttg ctcaactcta
gttaacggtg gagggcagtg tagtctgagc agtactcgtt 1260gctgccgcgc
gcgccaccag acataatagc tgacagacta acagactgtt cctttccatg
1320ggtcttttct gcagtcaccg tcgtcgacac gatccgatat cgccgccacc
atgaatccta 1380ataagaagat catcacaatc ggaagcatct gcatggtgac
aggaatggtg agcctgatgc 1440tgcagatcgg aaatctgatc agcatctggg
tgagccacag catccacaca ggaaatcagc 1500acaaggccga gcctatcagc
aatacaaatt ttctgacaga gaaggccgtg gccagcgtga 1560agctggccgg
aaatagcagc ctgtgcccta tcaatggatg ggccgtgtac agcaaggata
1620atagcatcag aatcggaagc aagggagatg tgtttgtgat cagagagcct
tttatcagct 1680gcagccacct ggagtgcaga acattttttc tgacacaggg
agccctgctg aatgataagc 1740acagcaatgg aacagtgaag gatagaagcc
ctcacagaac actgatgagc tgccctgtgg 1800gagaggcccc tagcccttac
aatagcagat ttgagagcgt ggcctggagc gccagcgcct 1860gccacgatgg
aacaagctgg ctgacaatcg gaatcagcgg acctgataat ggagccgtgg
1920ccgtgctgaa gtacaatgga atcatcacag atacaatcaa gagctggaga
aataatatcc 1980tgagaacaca ggagagcgag tgcgcctgcg tgaatggaag
ctgctttaca gtgatgacag 2040atggacctag caatggacag gccagccaca
agatctttaa gatggagaag ggaaaggtgg 2100tgaagagcgt ggagctggat
gcccctaatt accactacga ggagtgcagc tgctaccctg 2160atgccggaga
gatcacatgc gtgtgcagag ataattggca cggaagcaat agaccttggg
2220tgagctttaa tcagaatctg gagtaccaga tcggatacat ctgcagcgga
gtgtttggag 2280ataatcctag acctaatgat ggaacaggaa gctgcggacc
tgtgagcagc aatggagcct 2340acggagtgaa gggatttagc tttaagtacg
gaaatggagt gtggatcgga agaacaaaga 2400gcacaaatag cagaagcgga
tttgagatga tctgggaccc taatggatgg acagagacag 2460atagcagctt
tagcgtgaag caggatatcg tggccatcac agattggagc ggatacagcg
2520gaagctttgt gcagcaccct gagctgacag gactggattg catcagacct
tgcttttggg 2580tggagctgat cagaggaaga cctaaggaga gcacaatctg
gacaagcgga agcagcatca 2640gcttttgcgg agtgaatagc gatacagtgg
gatggagctg gcctgatgga gccgagctgc 2700cttttacaat cgataagtga
gcggccgctc tagaccaggc cctggatcca gatctgctgt 2760gccttctagt
tgccagccat ctgttgtttg cccctccccc gtgccttcct tgaccctgga
2820aggtgccact cccactgtcc tttcctaata aaatgaggaa attgcatcgc
attgtctgag 2880taggtgtcat tctattctgg ggggtggggt ggggcaggac
agcaaggggg aggattggga 2940agacaatagc aggcatgctg gggatgcggt
gggctctatg ggtacccagg tgctgaagaa 3000ttgacccggt tcctcctggg
ccagaaagaa gcaggcacat ccccttctct gtgacacacc 3060ctgtccacgc
ccctggttct tagttccagc cccactcata ggacactcat agctcaggag
3120ggctccgcct tcaatcccac ccgctaaagt acttggagcg gtctctccct
ccctcatcag 3180cccaccaaac caaacctagc ctccaagagt gggaagaaat
taaagcaaga taggctatta 3240agtgcagagg gagagaaaat gcctccaaca
tgtgaggaag taatgagaga aatcatagaa 3300ttttaaggcc atgatttaag
gccatcatgg ccttaatctt ccgcttcctc gctcactgac 3360tcgctgcgct
cggtcgttcg gctgcggcga gcggtatcag ctcactcaaa ggcggtaata
3420cggttatcca cagaatcagg ggataacgca ggaaagaaca tgtgagcaaa
aggccagcaa 3480aaggccagga accgtaaaaa ggccgcgttg ctggcgtttt
tccataggct ccgcccccct 3540gacgagcatc acaaaaatcg acgctcaagt
cagaggtggc gaaacccgac aggactataa 3600agataccagg cgtttccccc
tggaagctcc ctcgtgcgct ctcctgttcc gaccctgccg 3660cttaccggat
acctgtccgc ctttctccct tcgggaagcg tggcgctttc tcatagctca
3720cgctgtaggt atctcagttc ggtgtaggtc gttcgctcca agctgggctg
tgtgcacgaa 3780ccccccgttc agcccgaccg ctgcgcctta tccggtaact
atcgtcttga gtccaacccg 3840gtaagacacg acttatcgcc actggcagca
gccactggta acaggattag cagagcgagg 3900tatgtaggcg gtgctacaga
gttcttgaag tggtggccta actacggcta cactagaaga 3960acagtatttg
gtatctgcgc tctgctgaag ccagttacct tcggaaaaag agttggtagc
4020tcttgatccg gcaaacaaac
caccgctggt agcggtggtt tttttgtttg caagcagcag 4080attacgcgca
gaaaaaaagg atctcaagaa gatcctttga tcttttctac ggggtctgac
4140gctcagtgga acgaaaactc acgttaaggg attttggtca tgagattatc
aaaaaggatc 4200ttcacctaga tccttttaaa ttaaaaatga agttttaaat
caatctaaag tatatatgag 4260taaacttggt ctgacagtta ccaatgctta
atcagtgagg cacctatctc agcgatctgt 4320ctatttcgtt catccatagt
tgcctgactc gggggggggg ggcgctgagg tctgcctcgt 4380gaagaaggtg
ttgctgactc ataccaggcc tgaatcgccc catcatccag ccagaaagtg
4440agggagccac ggttgatgag agctttgttg taggtggacc agttggtgat
tttgaacttt 4500tgctttgcca cggaacggtc tgcgttgtcg ggaagatgcg
tgatctgatc cttcaactca 4560gcaaaagttc gatttattca acaaagccgc
cgtcccgtca agtcagcgta atgctctgcc 4620agtgttacaa ccaattaacc
aattctgatt agaaaaactc atcgagcatc aaatgaaact 4680gcaatttatt
catatcagga ttatcaatac catatttttg aaaaagccgt ttctgtaatg
4740aaggagaaaa ctcaccgagg cagttccata ggatggcaag atcctggtat
cggtctgcga 4800ttccgactcg tccaacatca atacaaccta ttaatttccc
ctcgtcaaaa ataaggttat 4860caagtgagaa atcaccatga gtgacgactg
aatccggtga gaatggcaaa agcttatgca 4920tttctttcca gacttgttca
acaggccagc cattacgctc gtcatcaaaa tcactcgcat 4980caaccaaacc
gttattcatt cgtgattgcg cctgagcgag acgaaatacg cgatcgctgt
5040taaaaggaca attacaaaca ggaatcgaat gcaaccggcg caggaacact
gccagcgcat 5100caacaatatt ttcacctgaa tcaggatatt cttctaatac
ctggaatgct gttttcccgg 5160ggatcgcagt ggtgagtaac catgcatcat
caggagtacg gataaaatgc ttgatggtcg 5220gaagaggcat aaattccgtc
agccagttta gtctgaccat ctcatctgta acatcattgg 5280caacgctacc
tttgccatgt ttcagaaaca actctggcgc atcgggcttc ccatacaatc
5340gatagattgt cgcacctgat tgcccgacat tatcgcgagc ccatttatac
ccatataaat 5400cagcatccat gttggaattt aatcgcggcc tcgagcaaga
cgtttcccgt tgaatatggc 5460tcataacacc ccttgtatta ctgtttatgt
aagcagacag ttttattgtt catgatgata 5520tatttttatc ttgtgcaatg
taacatcaga gattttgaga cacaacgtgg ctttcccccc 5580ccccccatta
ttgaagcatt tatcagggtt attgtctcat gagcggatac atatttgaat
5640gtatttagaa aaataaacaa ataggggttc cgcgcacatt tccccgaaaa
gtgccacctg 5700acgtctaaga aaccattatt atcatgacat taacctataa
aaataggcgt atcacgaggc 5760cctttcgtc 576994598DNAArtificial
SequenceArtificial construct 9cgacaatatt ggctattggc cattgcatac
gttgtatcta tatcataata tgtacattta 60tattggctca tgtccaatat gaccgccatg
ttgacattga ttattgacta gttattaata 120gtaatcaatt acggggtcat
tagttcatag cccatatatg gagttccgcg ttacataact 180tacggtaaat
ggcccgcctc gtgaccgccc aacgaccccc gcccattgac gtcaataatg
240acgtatgttc ccatagtaac gccaataggg actttccatt gacgtcaatg
ggtggagtat 300ttacggtaaa ctgcccactt ggcagtacat caagtgtatc
atatgccaag tccgccccta 360ttgacgtcaa tgacggtaaa tggcccgcct
ggcattatgc ccagtacatg accttacggg 420actttcctac ttggcagtac
atctacgtat tagtcatcgc tattaccatg gtgatgcggt 480tttggcagta
caccaatggg cgtggatagc ggtttgactc acggggattt ccaagtctcc
540accccattga cgtcaatggg agtttgtttt ggcaccaaaa tcaacgggac
tttccaaaat 600gtcgtaataa ccccgccccg ttgacgcaaa tgggcggtag
gcgtgtacgg tgggaggtct 660atataagcag agctcgttta gtgaaccgtc
agatcgcctg gagacgccat ccacgctgtt 720ttgacctcca tagaagacac
cgggaccgat ccagcctccg cggccgggaa cggtgcattg 780gaacgcggat
tccccgtgcc aagagtgacg taagtaccgc ctatagactc tataggcaca
840cccctttggc tcttatgcat gctatactgt ttttggcttg gggcctatac
acccccgctc 900cttatgctat aggtgatggt atagcttagc ctataggtgt
gggttattga ccattattga 960ccactcccct attggtgacg atactttcca
ttactaatcc ataacatggc tctttgccac 1020aactatctct attggctata
tgccaatact ctgtccttca gagactgaca cggactctgt 1080atttttacag
gatggggtcc catttattat ttacaaattc acatatacaa caacgccgtc
1140ccccgtgccc gcagttttta ttaaacatag cgtgggatct ccacgcgaat
ctcgggtacg 1200tgttccggac atgggctctt ctccggtagc ggcggagctt
ccacatccga gccctggtcc 1260catgcctcca gcggctcatg gtcgctcggc
agctccttgc tcctaacagt ggaggccaga 1320cttaggcaca gcacaatgcc
caccaccacc agtgtgccgc acaaggccgt ggcggtaggg 1380tatgtgtctg
aaaatgagct cggagattgg gctcgcaccg tgacgcagat ggaagactta
1440aggcagcggc agaagaagat gcaggcagct gagttgttgt attctgataa
gagtcagagg 1500taactcccgt tgcggtgctg ttaacggtgg agggcagtgt
agtctgagca gtactcgttg 1560ctgccgcgcg cgccaccaga cataatagct
gacagactaa cagactgttc ctttccatgg 1620gtcttttctg cagtcaccgt
ccaagcttag atctgccacc atgagcctgc tgaccgaggt 1680ggagacatac
gtgctgtcca tcatccccag cggccctctg aaggccgaga tcgcccagag
1740actggaagat gtgttcgccg gcaagaacac cgacctggaa gtgctgatgg
aatggctgaa 1800aaccagaccc atcctgagcc ctctgaccaa gggcatcctg
ggcttcgtgt tcaccctgac 1860cgtgcccagc gagagaggcc tgcagaggcg
gagattcgtg cagaacgccc tgaacggcaa 1920cggcgacccc aacaacatgg
acaaggccgt gaagctgtac agaaagctga agcgggagat 1980caccttccac
ggcgccaaag agatcagcct gagctacagc gctggcgccc tggccagctg
2040catgggcctg atctacaaca gaatgggcgc cgtgaccacc gaggtggcct
tcggcctggt 2100ctgcgccacc tgcgagcaga tcgccgacag ccagcacaga
tcccacagac agatggtcac 2160caccaccaac cccctgatca gacacgagaa
cagaatggtg ctggcctcta ccaccgccaa 2220ggccatggaa cagatggccg
gcagcagcga gcaggccgcc gaggctatgg aagtcgcctc 2280tcaggctagg
cagatggtcc aggccatgag aaccatcggc acccacccca gcagctctgc
2340tggcctgaag aacgacctgc tggaaaacct gcaggcctac cagaaaagaa
tgggcgtcca 2400gatgcagaga ttcaagtgat gagatatccc tcagagaggg
gatcctcgca atccctaggg 2460ctgtgccttc tagttgccag ccaaactgtt
gtttgcccct cccccgtgcc ttccttgacc 2520ctggaaggtg ccactcccac
tgtcctttcc taataaaatg aggaaattgc atcgcattgt 2580ctgagtaggt
gtcattctat tctggggggt ggggtggggc aggacagcaa gggggaggat
2640tgggaagaca atagcaggca tgctggggat gcggtgggct ctatgttcag
aacgctcggt 2700tgccgccggg cgttttttat ctagagtcga caaattcaga
agaactcgtc aagaaggcga 2760tagaaggcga tgcgctgcga atcgggagcg
gcgataccgt aaagcacgag gaagcggtca 2820gcccattcgc cgccaagctc
ttcagcaata tcacgggtag ccaacgctat gtcctgatag 2880cggtccgcca
cacccagccg gccacagtcg atgaatccag aaaagcggcc attttccacc
2940atgatattcg gcaagcaggc atcgccatgg gtcacgacga gatcctcgcc
gtcgggcatg 3000ctcgccttga gcctggcgaa cagttcggct ggcgcgagcc
cctgatgctc ttcgtccaga 3060tcatcctgat cgacaagacc ggcttccatc
cgagtacgtg ctcgctcgat gcgatgtttc 3120gcttggtggt cgaatgggca
ggtagccgga tcaagcgtat gcagccgccg cattgcatca 3180gccatgatgg
atactttctc ggcaggagca aggtgagatg acaggagatc ctgccccggc
3240acttcgccca atagcagcca gtcccttccc gcttcagtga caacgtcgag
cacagctgcg 3300caaggaacgc ccgtcgtggc cagccacgat agccgcgctg
cctcgtcttg cagttcattc 3360agggcaccgg acaggtcggt cttgacaaaa
agaaccgggc gcccctgcgc tgacagccgg 3420aacacggcgg catcagagca
gccgattgtc tgttgtgccc agtcatagcc gaatagcctc 3480tccacccaag
cggccggaga acctgcgtgc aatccatctt gttcaatcat gcgaaacgat
3540cctcatcctg tctcttgatc agatcttgat cccctgcgcc atcagatcct
tggcggcgag 3600aaagccatcc agtttacttt gcagggcttc ccaaccttac
cagagggcgc cccagctggc 3660aattccggtt cgcttgctgt ccataaaacc
gcccagtcta gctatcgcca tgtaagccca 3720ctgcaagcta cctgctttct
ctttgcgctt gcgttttccc ttgtccagat agcccagtag 3780ctgacattca
tccggggtca gcaccgtttc tgcggactgg ctttctacgt gaaaaggatc
3840taggtgaaga tcctttttga taatctcatg accaaaatcc cttaacgtga
gttttcgttc 3900cactgagcgt cagaccccgt agaaaagatc aaaggatctt
cttgagatcc tttttttctg 3960cgcgtaatct gctgcttgca aacaaaaaaa
ccaccgctac cagcggtggt ttgtttgccg 4020gatcaagagc taccaactct
ttttccgaag gtaactggct tcagcagagc gcagatacca 4080aatactgtcc
ttctagtgta gccgtagtta ggccaccact tcaagaactc tgtagcaccg
4140cctacatacc tcgctctgct aatcctgtta ccagtggctg ctgccagtgg
cgataagtcg 4200tgtcttaccg ggttggactc aagacgatag ttaccggata
aggcgcagcg gtcgggctga 4260acggggggtt cgtgcacaca gcccagcttg
gagcgaacga cctacaccga actgagatac 4320ctacagcgtg agctatgaga
aagcgccacg cttcccgaag ggagaaaggc ggacaggtat 4380ccggtaagcg
gcagggtcgg aacaggagag cgcacgaggg agcttccagg gggaaacgcc
4440tggtatcttt atagtcctgt cgggtttcgc cacctctgac ttgagcgtcg
atttttgtga 4500tgctcgtcag gggggcggag cctatggaaa aacgccagca
acgcggcctt tttacggttc 4560ctgggctttt gctggccttt tgctcacatg ttgtcgac
4598109PRTArtificial SequenceSynthetic peptide 10Ile Tyr Ser Thr
Val Ala Ser Ser Leu1 5118PRTArtificial SequenceSynthetic peptide
11Ser Ile Ile Asn Phe Glu Lys Leu1 5
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013
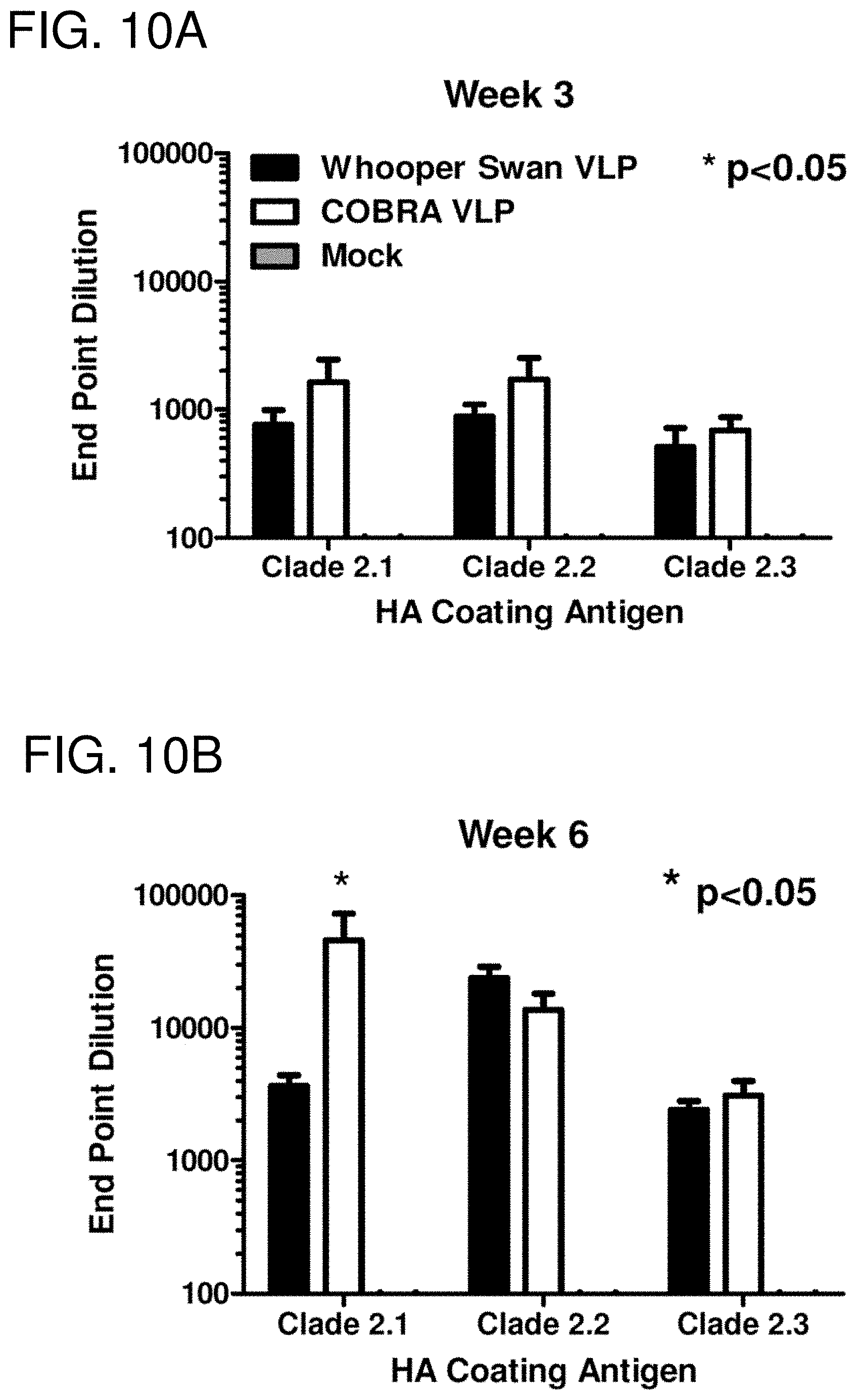
D00014
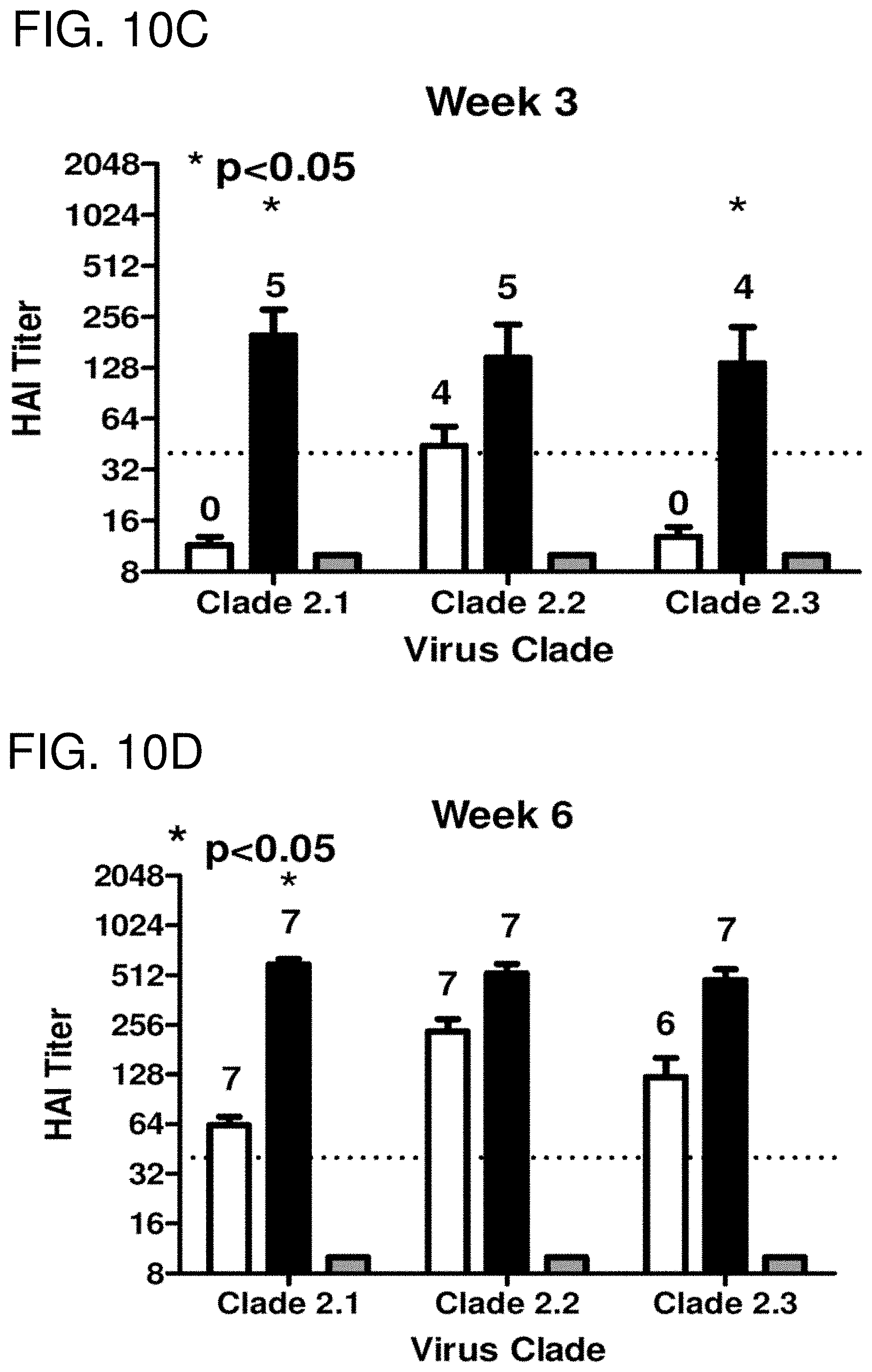
D00015

D00016

D00017
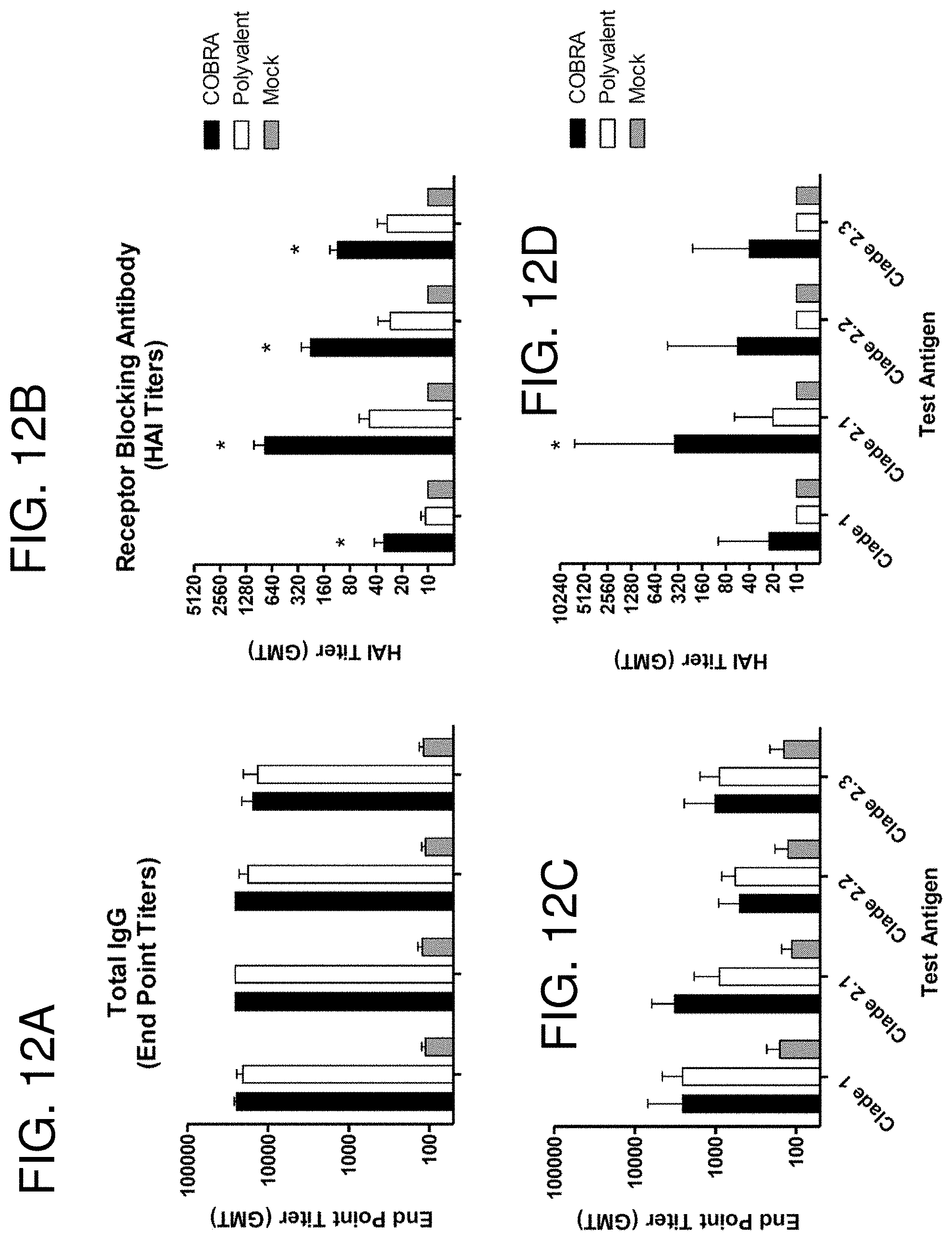
D00018

D00019

D00020

D00021
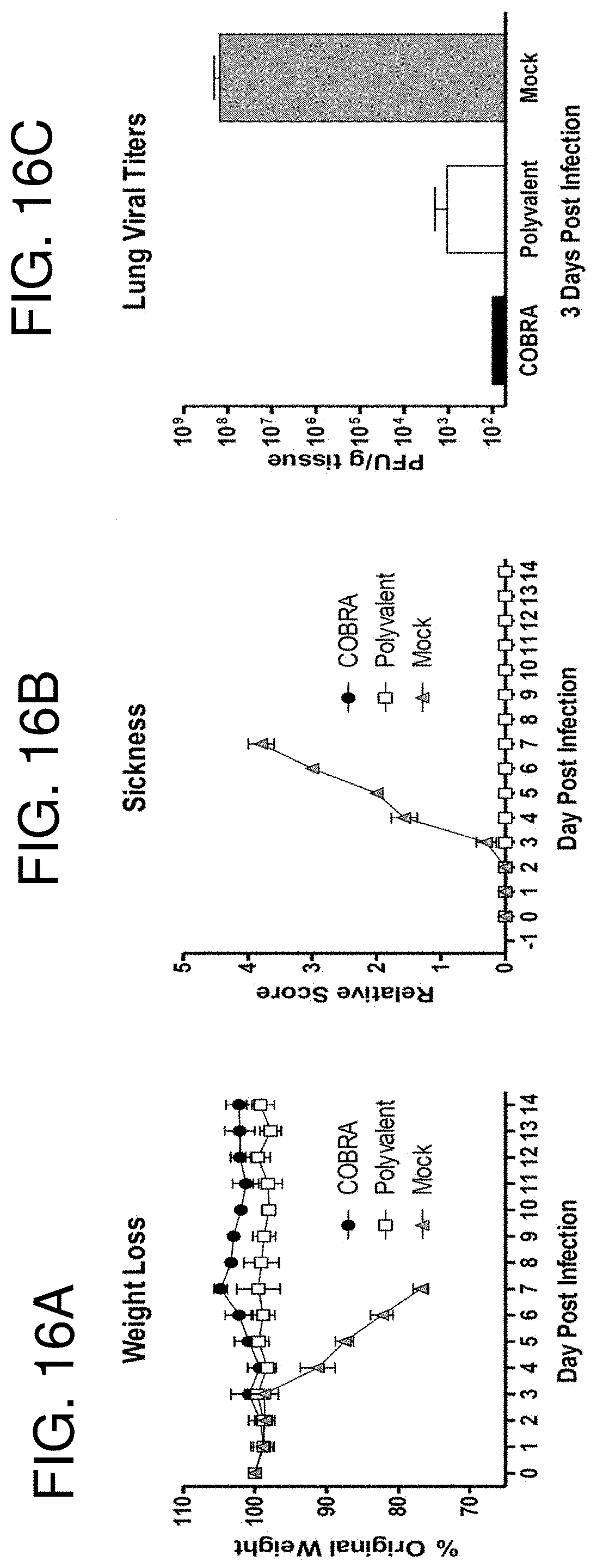
D00022
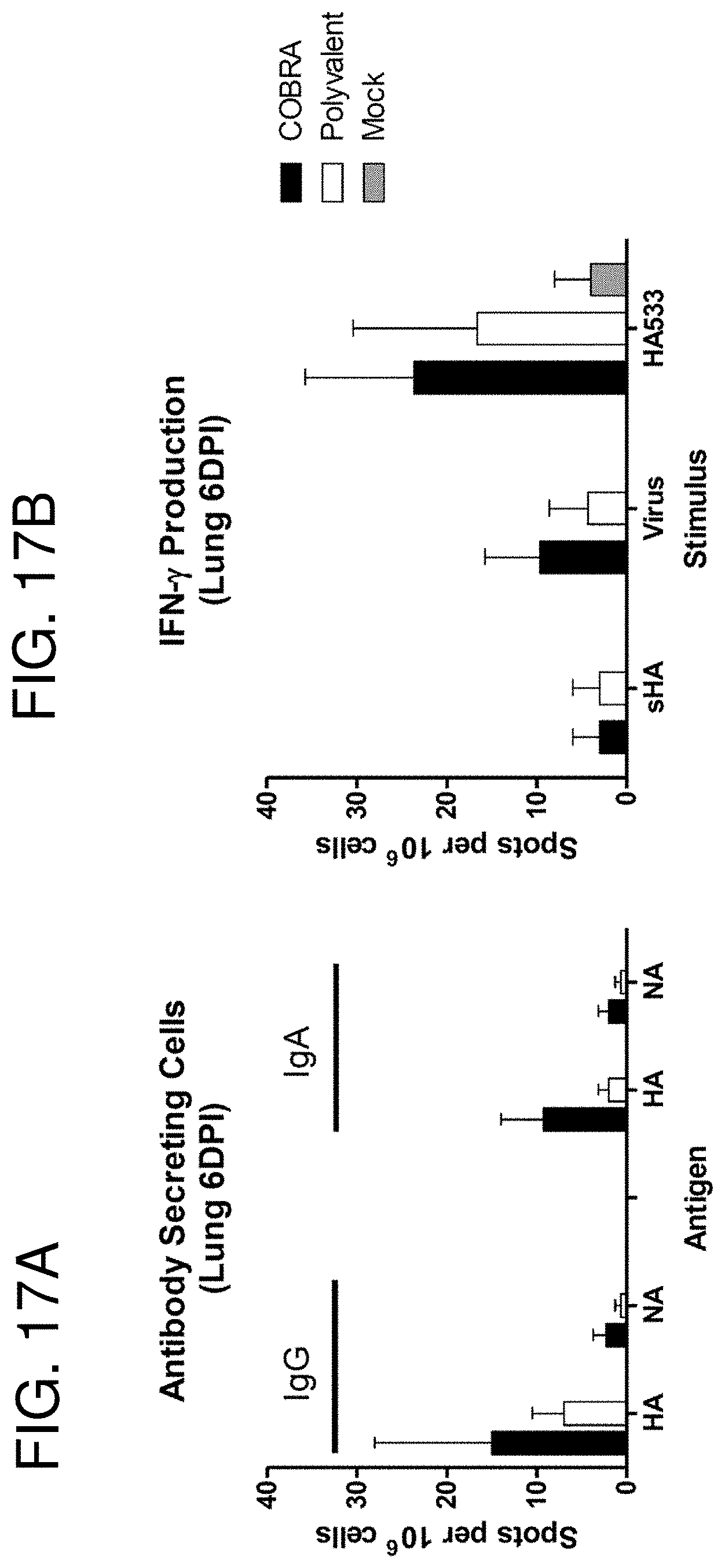
D00023
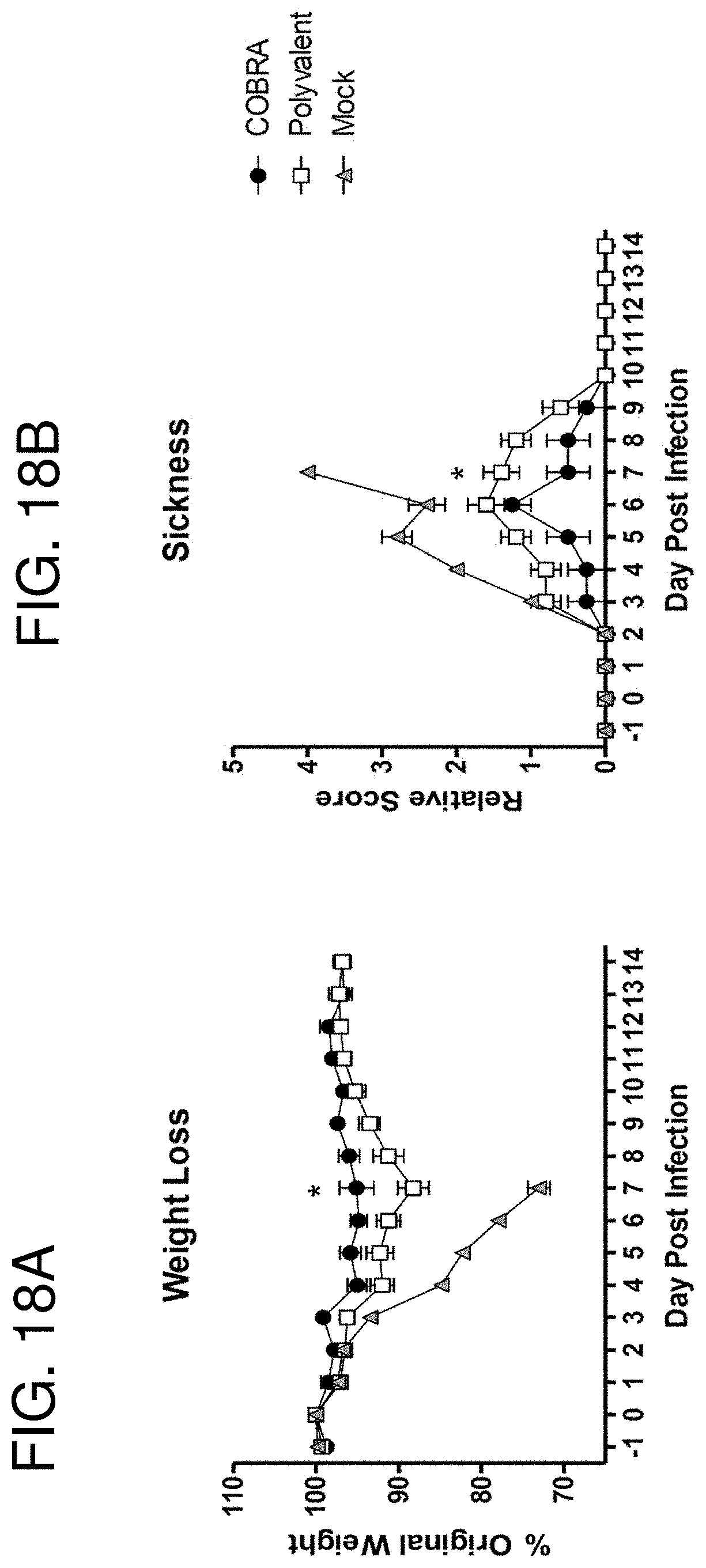
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.