Hyaline Cartilage Shaping
DINGER; Fred ; et al.
U.S. patent application number 16/825591 was filed with the patent office on 2020-10-15 for hyaline cartilage shaping. This patent application is currently assigned to Aerin Medical Inc.. The applicant listed for this patent is Aerin Medical Inc.. Invention is credited to Fred DINGER, Andrew FRAZIER.
| Application Number | 20200323548 16/825591 |
| Document ID | / |
| Family ID | 1000004916731 |
| Filed Date | 2020-10-15 |







View All Diagrams
| United States Patent Application | 20200323548 |
| Kind Code | A1 |
| DINGER; Fred ; et al. | October 15, 2020 |
HYALINE CARTILAGE SHAPING
Abstract
Disclosed embodiments include devices and methods for shaping, bending, and/or volumetrically reducing rigid cartilaginous structures, such as hyaline cartilage in the septum. In the case of septal cartilage, shaping, bending, or reducing the cartilage would be useful for reducing nasal obstruction or to improve the cosmetic appearance of the nose.
| Inventors: | DINGER; Fred; (Austin, TX) ; FRAZIER; Andrew; (Sunnyvale, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Aerin Medical Inc. Sunnyvale CA |
||||||||||
| Family ID: | 1000004916731 | ||||||||||
| Appl. No.: | 16/825591 | ||||||||||
| Filed: | March 20, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15429947 | Feb 10, 2017 | 10603059 | ||
| 16825591 | ||||
| 62294724 | Feb 12, 2016 | |||
| 62335802 | May 13, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61B 2017/248 20130101; A61B 2018/126 20130101; C12Y 302/01035 20130101; A61B 2018/00327 20130101; A61B 2018/0212 20130101; C12Y 304/21004 20130101; A61B 2018/0022 20130101; A61B 2090/062 20160201; A61B 2018/00875 20130101; A61B 18/1485 20130101; A61B 2017/00407 20130101; A61K 38/47 20130101; A61B 1/0661 20130101; A61B 18/1206 20130101; A61B 2090/3945 20160201; A61B 18/1442 20130101; A61K 9/0043 20130101; A61B 2017/0088 20130101; A61B 2017/00867 20130101; A61B 2018/046 20130101; A61B 17/24 20130101; C12Y 304/24007 20130101; A61B 2017/00893 20130101; A61B 2018/00642 20130101; A61B 2018/00595 20130101; A61B 5/065 20130101; A61B 17/00491 20130101; A61K 38/4826 20130101; A61B 2018/0016 20130101; A61B 18/1445 20130101; A61B 18/0218 20130101; A61B 2017/00929 20130101; A61B 2018/1253 20130101; A61B 2017/00955 20130101; A61B 18/1402 20130101; A61B 2018/1467 20130101; A61B 2017/320094 20170801; A61B 2018/143 20130101; A61B 2018/00821 20130101; A61B 2090/306 20160201; A61B 2018/00101 20130101 |
| International Class: | A61B 17/24 20060101 A61B017/24; A61B 18/14 20060101 A61B018/14; A61K 9/00 20060101 A61K009/00; A61K 38/47 20060101 A61K038/47; A61B 5/06 20060101 A61B005/06; A61B 1/06 20060101 A61B001/06; A61B 18/02 20060101 A61B018/02; A61B 18/12 20060101 A61B018/12; A61K 38/48 20060101 A61K038/48 |
Claims
1. A method for modifying a nasal septum of a subject's nose without forming an incision or removing tissue, the method comprising: injecting a substance under nasal mucosa and into contact with cartilage of the nasal septum, wherein the substance is configured to modify a property of the cartilage, and wherein the substance is selected from the group consisting of collagenase, hyaluronidase, tosyl lysyl chloromethane, trypsin, and trypsin/EDTA; inserting an elongate treatment element of a bipolar radiofrequency energy delivery device into the subject's nose, wherein the elongate treatment element comprises two rows of bipolar electrodes; applying force to the nasal septum with the elongate treatment element, to change a shape of the cartilage of the nasal septum; applying radiofrequency energy to the cartilage of the nasal septum at a selected tissue depth by transmitting the radiofrequency energy from a first row of the two rows of bipolar electrodes to a second row of the two rows, while continuing to apply force to the nasal septum with the elongate treatment element; and removing the elongate treatment element from the subject's nose, wherein the shape of the cartilage of the nasal septum remains changed after the elongate treatment element is removed.
2. (canceled)
3. The method of claim 1, wherein the substance is configured to soften the cartilage of the nasal septum.
4. The method of claim 1, wherein the substance is configured to dissolve proteoglycan structures of the cartilage.
5. The method of claim 1, wherein the substance comprises about 0.5 ml to about 2.5 ml of collagenase at a concentration of about 1 mg/ml to about 10 mg/ml.
6. The method of claim 1, wherein the substance comprises between about 0.5 ml to about 2.5 ml of trypsin at a concentration of about 10 .mu.g/ml to about 100 .mu.g/ml.
7. The method of claim 1, further comprising allowing the substance to reside on the nasal septum for a period of time before applying the radiofrequency energy to the cartilage of the nasal septum.
8. The method of claim 7, wherein the period of time is between 15 minutes and 90 minutes.
9. The method of claim 7, wherein the period of time is selected to be sufficient for the substance to create a band of degraded cartilage ranging from 100 .mu.m to 1 mm from a surface of the cartilage.
10. (canceled)
11. The method of claim 1, wherein injecting the substance comprises injecting into a space between the nasal mucosa and the cartilage.
12. The method of claim 1, further comprising defining an area of application of the substance by providing a physical barrier to contain the substance.
13. The method of claim 1, wherein applying the radiofrequency energy to the cartilage of the nasal septum comprises heating the cartilage.
14. The method of claim 13, wherein heating the cartilage of the nasal septum comprises heating the cartilage to a temperature selected to denature or deactivate the substance.
15. (canceled)
16. The method of claim 1, wherein changing the shape of the cartilage comprises correcting a deviation of the nasal septum.
17.-19. (canceled)
20. The method of claim 17, further comprising: inserting a reshaping device into the patient's nose, such that a first treatment element of the reshaping device is positioned on one side of the nasal septum and a second treatment element of the reshaping device is positioned on an opposite side of the nasal septum; and reshaping the nasal septum using the first treatment element and the second treatment element at least one of before, during or after applying the radiofrequency energy.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 15/429,947, filed Feb. 10, 2017, which claims the benefit of U.S. Provisional Application No. 62/294,724, filed on Feb. 12, 2016, and U.S. Provisional Application No. 62/335,802, filed on May 13, 2016. The disclosures of these priority applications are hereby incorporated by reference in their entireties herein.
[0002] Embodiments described in this application may be used in combination or conjunction with the subject matter described in the following applications, which are hereby fully incorporated by reference for any and all purposes as if set forth herein in their entireties: U.S. patent application Ser. No. 14/026,922, filed Sep. 13, 2013, entitled "METHODS AND DEVICES TO TREAT NASAL AIRWAYS," issued Mar. 24, 2015, as U.S. Pat. No. 8,986,301; U.S. patent application Ser. No. 14/675,689, filed Mar. 31, 2015, entitled "POST NASAL DRIP TREATMENT," issued Aug. 16, 2016, as U.S. Pat. No. 9,415,194; and U.S. patent application Ser. No. 15/175,651, filed Jun. 7, 2016, entitled "PRESSURE SENSITIVE TISSUE TREATMENT DEVICE," issued on Feb. 13, 2018 as U.S. Pat. No. 9,888,957.
FIELD OF THE INVENTION
[0003] This application relates generally to the field of medical devices and treatments, and in particular to systems, devices and methods for treating tissue, such as the hyaline cartilage of structures within the nose and upper airway.
BACKGROUND
[0004] During respiration, the anatomy, shape, tissue composition, and properties of the human airway produce airflow resistance. The nose is responsible for almost two thirds of this resistance. Most of this resistance occurs in the anterior part of the nose, known as the internal nasal valve, which acts as a flow-limiter. The external nasal valve structure also causes resistance to nasal airflow. Effective physiological normal respiration occurs over a range of airflow resistances. However, excessive resistance to airflow can result in abnormalities of respiration that can significantly affect a patient's quality of life. Poor nasal breathing and/or nasal congestion has profound effects on a person's health and quality of life, which can be measured by validated questionnaires, such as the NOSE score, as described in Stewart M G, Witsell D L, Smith T L, Weaver E M, Yueh B, and Hannley M T., "Development and Validation of the Nasal Obstruction Symptom Evaluation (NOSE) Scale," Otolaryngol Head Neck Surg 2004; 130:157-63.
[0005] Inadequate nasal airflow can result from a number of conditions causing an inadequate cross-sectional area of the nasal airway in the absence of any collapse or movement of the cartilages and soft tissues of the nasal airway. A common cause of inadequate nasal airflow is deviation of the nasal septum. The nasal septum is a wall of tissue that separates the nasal cavity into two nostrils. The septum is made up of bone, hyaline cartilage, and nasal mucosa. The American Academy of Otolaryngology estimates that many as 80% of adults have a nasal septum that is slightly off center. A more severe shift away from the midline of the nose, known as a deviated septum, frequently results in difficulty breathing and can often precipitate chronic sinusitis. The most common means of correcting a deviation is partial or full removal of the nasal septum, known as a septoplasty. More than 250,000 septoplasties are performed in the United States each year. Although septoplasty can be an effective treatment, it is also quite invasive, can lead to a painful and difficult recovery, and is associated with a number of risks and potential side effects, as with any invasive surgical procedure. It is estimated that only about 10% of patients with a deviated septum will elect to have surgery, in part due to the risks and invasiveness of the procedure.
[0006] Therefore, it would be advantageous to have improved methods and devices for treating a deviated septum, to help improve breathing and/or alleviate other symptoms in a patient. Ideally, such methods and devices would provide a non-surgical, minimally invasive or less invasive approach for correcting deviated septa and thus would provide patients with a less painful alternative treatment, with fewer risks and side effects and easier recovery. At least some of these objectives are addressed by the embodiments described in this application.
SUMMARY
[0007] Embodiments of the present application are directed to devices, systems and methods for treating nasal airways. Such embodiments may be used to improve breathing by decreasing airflow resistance or perceived airflow resistance in the nasal airways. For example, the devices, systems and methods described herein may be used to reshape, remodel, strengthen, or change the properties of the tissues of the nose, including, but not limited to the skin, muscle, mucosa, submucosa and cartilage in the area of the nasal septum.
[0008] The nasal septum forms a portion of the nasal valve. The nasal valve is divided into external and internal portions. The external nasal valve is the external nasal opening formed by the columella at the base of the septum, the nasal floor, and the nasal rim (the lower region of the nasal wall, also known as the caudal border of the lower lateral cartilage). The nasalis muscle dilates the external nasal valve portion during inspiration. The internal nasal valve, which accounts for a large part of the nasal resistance, is located in the area of transition between the skin and respiratory epithelium. The internal nasal valve area is formed by the nasal septum, the caudal border of the upper lateral cartilage (ULC), the head of the inferior turbinate, and the pyriform aperture and the tissues that surround it. An angle formed between the caudal border of the ULC and the nasal septum is normally between about 10 degrees and about 15 degrees, as illustrated in FIG. 1.
[0009] In one aspect, a method for modifying a nasal septum of a subject's nose may involve: applying a solution to the nasal septum, where the solution is configured to modify cartilage of the nasal septum; providing a time period for the solution to modify the cartilage; inserting a device into the subject's nose; applying energy to the nasal septum using the device; reshaping the cartilage using the device; and removing the device.
[0010] In various embodiments, the solution may be collagenase, hyaluronidase, tosyl lysyl chloromethane, trypsin, trypsin/EDTA, or some combination thereof. In some embodiments, the solution is configured to soften the cartilage of the nasal septum. In some embodiments, the solution is configured to dissolve proteoglycan structures of the cartilage. In one embodiment, for example, the solution may include about 0.5 ml to 2.5 ml of collagenase at a concentration ranging from about 1 mg/ml to 10 mg/ml. In another embodiment, the solution may include between about 0.5 ml to 2.5 ml of trypsin at a concentration of about 10 .mu.g/ml to about 100 .mu.g/ml.
[0011] In some embodiments, the time period for the solution to modify the cartilage may be between about 15 minutes to about 90 minutes. In some embodiments, providing the time period for the solution to modify the cartilage may involve providing a time period for the solution to create a band of degraded cartilage ranging from about 100 .mu.m to about 1 mm from a surface of the cartilage. In some embodiments, applying the solution may involve injecting the solution into or near the cartilage. In some embodiments, injecting the solution into or near the cartilage may involve injecting the solution through nasal mucosal tissue to a space between the nasal mucosa and the cartilage.
[0012] Optionally, the method may also involve defining an area of application of the solution by providing a physical barrier to contain the applied solution. In some embodiments, applying energy to the nasal septum using the device may involve heating tissue of the nasal septum. In some embodiments, the method may include heating tissue of the nasal septum to a temperature selected to denature or deactivate the solution. In some embodiments, the device may include a radiofrequency electrode. In some embodiments, reshaping the cartilage using the device may involve correcting a septal deviation.
[0013] In another aspect, a method for treating a deviated nasal septum in a patient's nasal cavity may involve applying a cartilage modifying substance to cartilage of the deviated nasal septum and applying energy to the nasal septum via an energy delivery device to treat the deviated nasal septum. Optionally, the method may also involve allowing the substance to remain in the cartilage of the nasal septum for a predetermined time period before applying the energy. For example, in various embodiments, the predetermined time period may be between about 15 minutes and about 90 minutes.
[0014] In various embodiments, the substance may be any of collagenase, hyaluronidase, tosyl lysyl chloromethane, trypsin, trypsin/EDTA, or a combination thereof. In some embodiments, the substance may be a collagen softening substance. In some embodiments, the substance may be a proteoglycan dissolving substance. One embodiment of a substance may be about 0.5 ml to about 2.5 ml of collagenase at a concentration ranging from about 1 mg/ml to about 10 mg/ml. Another embodiment of a substance may be between about 0.5 ml to about 2.5 ml of trypsin at a concentration of about 10 .mu.g/ml to about 100 .mu.g/ml.
[0015] In some embodiments, applying the substance may involve injecting the substance into or near the cartilage. For example, some embodiments may involve injecting the substance through nasal mucosal tissue to a space between the nasal mucosa and the cartilage. Optionally, the method may also include forming a physical barrier within the patient's nasal cavity to contain the applied substance in or near the nasal septum.
[0016] The energy applied may be any suitable energy, such as but not limited to radiofrequency, heat, electrical, ultrasound, microwave and/or cryogenic energy. In some embodiments, applying energy to the nasal septum may involve heating tissue of the nasal septum to a temperature selected to denature or deactivate the substance. In some embodiments, the method may also involve applying pressure to the nasal septum, using the energy delivery device, to reshape the septum. In some embodiments, the substance may be applied via the energy delivery device. In alternative embodiments, it may be applied with a separate device, such as a needle and syringe. In various embodiments, the cartilage of the nasal septum may include hyaline cartilage.
[0017] In some embodiments, correcting a deviated septum of a subject's nose may involve applying a solution to a nasal septum of a subject's nose having a deviation. The solution may be configured to modify cartilage of the nasal septum. Correcting a deviated septum may further involve inserting a device into the subject's nose such that a first treatment element of the device is positioned on a side of septum and a second treatment element of the device is positioned on an opposite side of the septum. Correcting a deviated septum may further involve applying energy to the nasal septum using the first treatment element or the second treatment element. Correcting a deviated septum may further involve reshaping the cartilage using the first treatment element and the second treatment element, thereby correcting the deviated nasal septum.
[0018] In some embodiments, treating a deviated septum may involve inserting a device into a subject's nose having a deviation, the device having an elongate treatment element. Treating a deviated septum may also involve creating an air channel in the deviation using the elongate treatment element of the device, thereby reducing the deviation and improving airflow. Treating a deviated septum may further involve removing the device from the subject's nose. The air channel in the deviation may persist after the device is removed.
[0019] In some embodiments, treating a deviated septum may involve applying a solution to the nasal septum having a deviation. The solution may be configured to modify cartilage of the nasal septum. Treating a deviated septum may further involve providing a dwell time for the solution to modify cartilage of the nasal septum. Treating a deviated septum may further involve removing the modified cartilage of the nasal septum, thereby treating the deviated septum of the subject's nose.
[0020] In some embodiments, a device for treating a deviated septum of a subject's nose, may include a handle, an elongate shaft extending from the handle, and an elongate treatment element extending from the elongate shaft and configured to create channels in the deviated septum of the subject's nose. The elongate treatment element can be configured to apply energy to or remove energy from tissue of the deviated septum of the subject's nose. The device can include multiple pairs of bipolar electrodes arranged in a serial alignment along the treatment element. The pairs of bipolar electrodes can be arranged with the center of the electrodes along a longitudinal axis of the treatment element.
[0021] These and other aspects and embodiments will be described in further detail below, in reference to the attached drawing figures.
BRIEF DESCRIPTION OF DRAWINGS
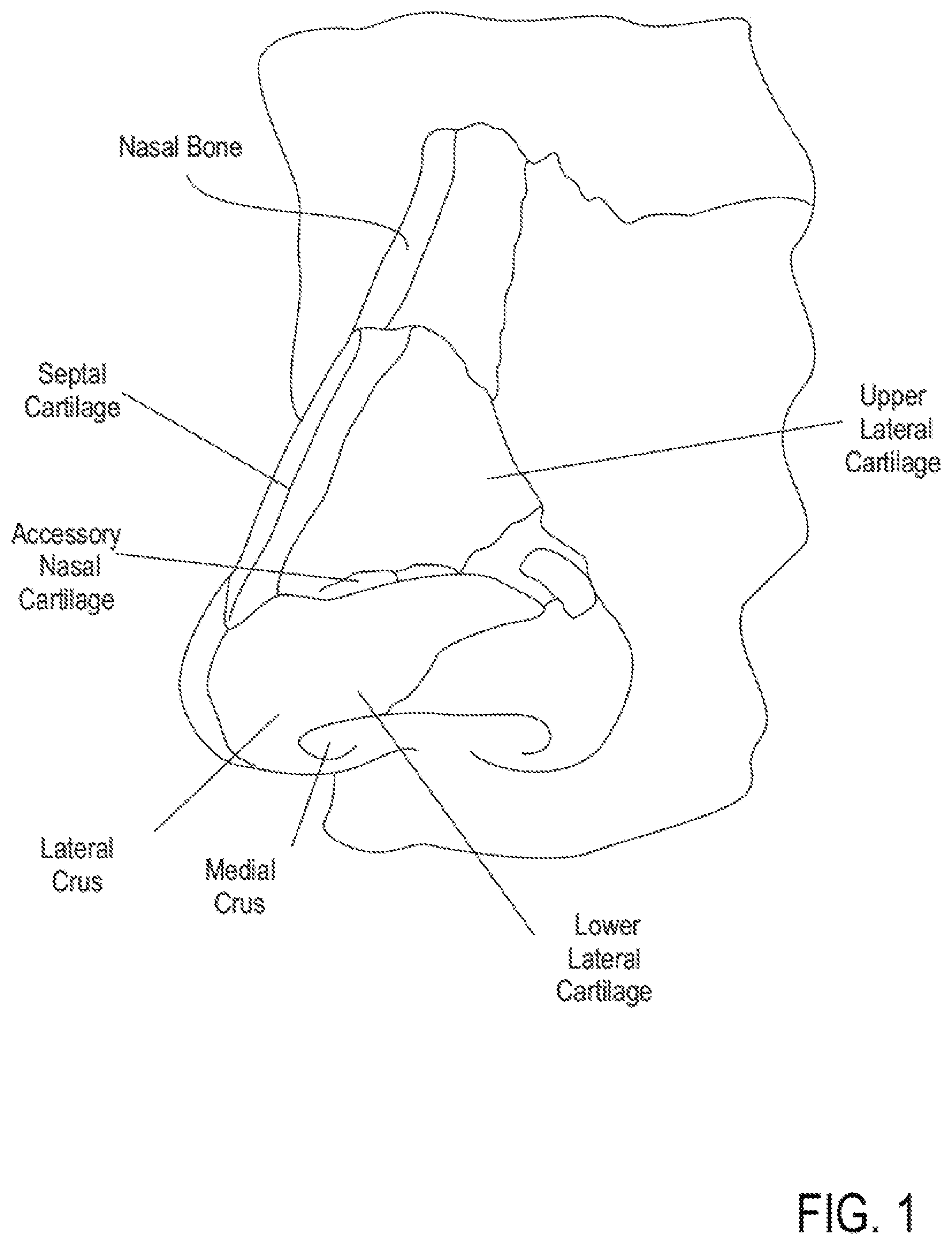
[0022] FIG. 1 depicts an illustration of bone and cartilage structures of a human nose.
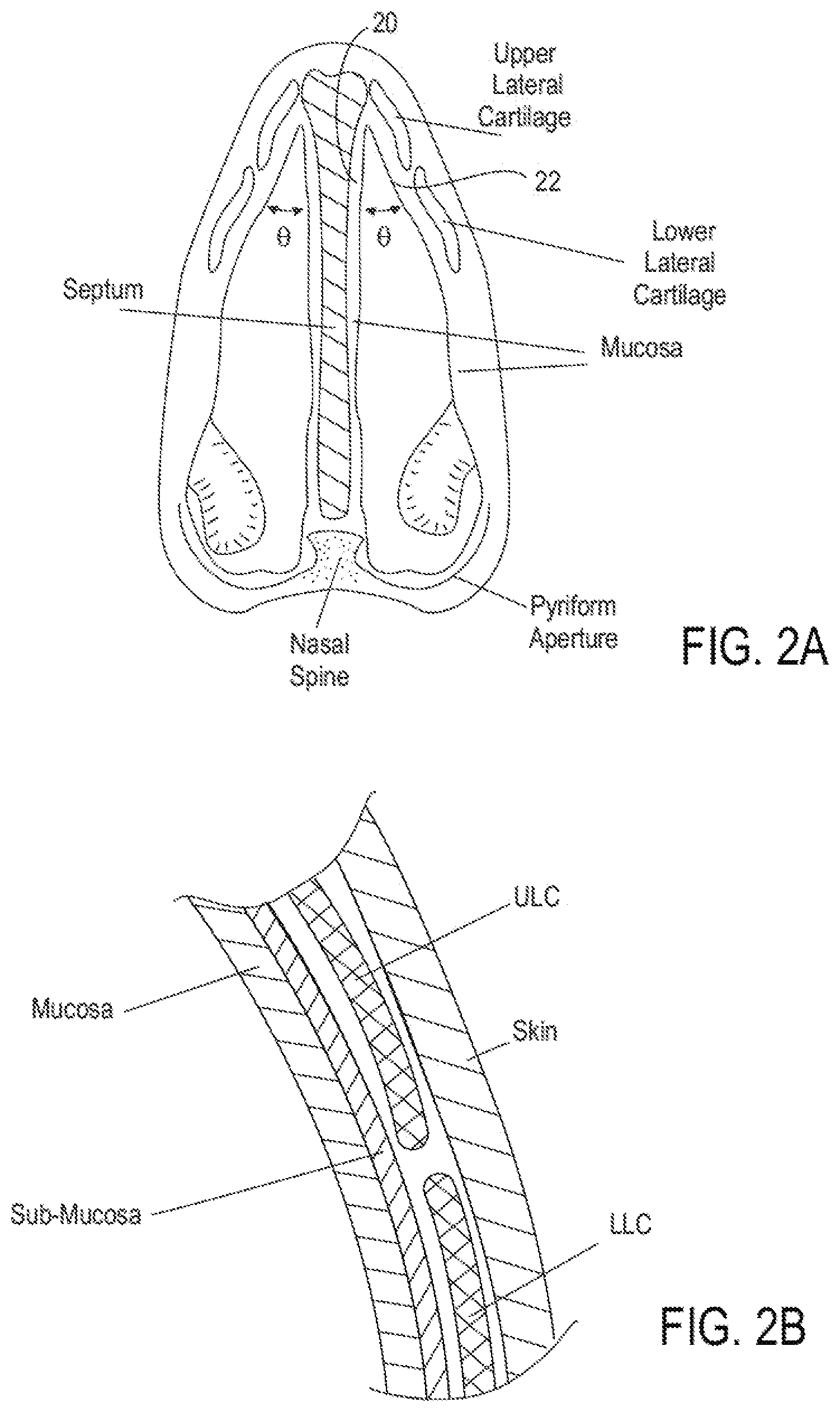
[0023] FIG. 2A shows a cross-sectional view, illustrating tissues and structures of a human nose.
[0024] FIG. 2B shows a detailed cross-sectional view, illustrating a detailed section of the structures of FIG. 2A.
[0025] FIG. 2C shows a view of the nostrils, illustrating tissues and structures of a human nose.
[0026] FIG. 3 depicts a schematic illustration of a nasal septum reshaping treatment device.
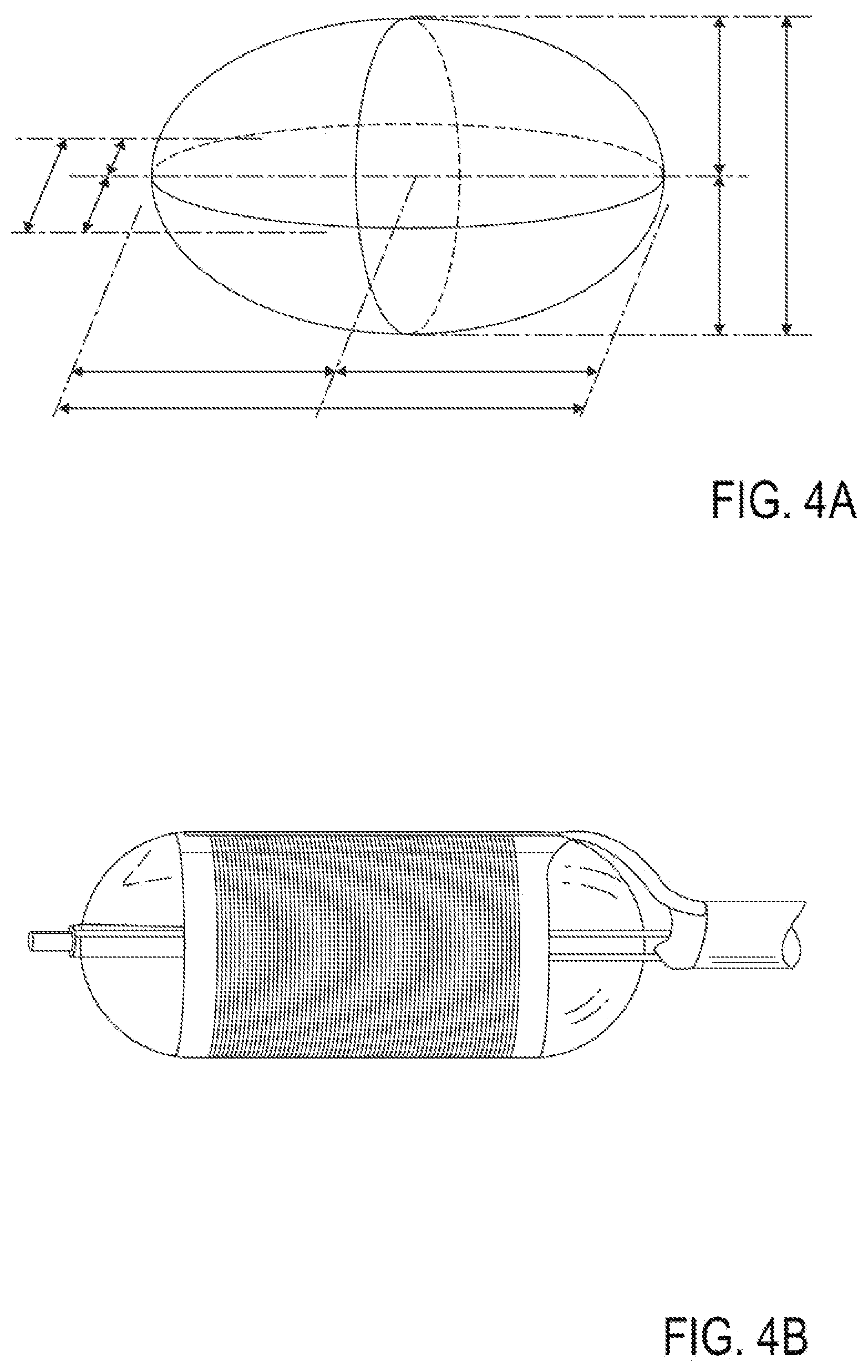
[0027] FIG. 4A is a perspective illustration of an embodiment of a treatment element shape.
[0028] FIG. 4B depicts a perspective illustration of another embodiment of a treatment element shape.
[0029] FIG. 4C shows a perspective illustration of another embodiment of a treatment element shape.
[0030] FIG. 4D depicts a cross-sectional view of a treatment device comprising a plurality of microneedles puncturing tissue in order to apply treatment at a desired tissue depth.
[0031] FIG. 5A illustrates one embodiment of a clamp-type nasal septum treatment device.
[0032] FIG. 5B illustrates another embodiment of a clamp-type nasal septum treatment device.
[0033] FIG. 6 depicts a partially-transparent perspective view, showing a stent implanted in a nose.
[0034] FIG. 7 depicts a perspective view, illustrating an energy delivery balloon being inserted into a nose.
[0035] FIGS. 8A-8J depict embodiments of various electrode arrangements for applying energy to the nasal septum area.
[0036] FIGS. 9A and 9B illustrate embodiments of devices for applying energy to the nasal septum area using a monopolar electrode.
[0037] FIGS. 10A and 10B illustrate an embodiment of a device for applying energy to the nasal septum area using a monopolar electrode and an external mold.
[0038] FIGS. 11A and 11B illustrate embodiments of devices for applying energy to the nasal septum area using electrode(s) and a counter-traction element.
[0039] FIGS. 12A and 12B illustrate embodiments of devices for applying energy to the nasal septum area and configured to be inserted into both nostrils simultaneously.
[0040] FIGS. 13A-13E illustrate embodiments of devices for applying energy to the nasal septum area configured to be inserted into both nostrils simultaneously, having a mold or counter-traction element for engaging the nose externally.
[0041] FIGS. 14A and 14B illustrate embodiments of devices for applying energy to the nasal septum area configured to be inserted into both nostrils simultaneously, having separate external molds.
[0042] FIGS. 15A-15C illustrate embodiments of devices for applying energy to the nasal septum area configured to be inserted into both nostrils simultaneously, having separate counter-traction elements.
[0043] FIG. 16 shows an embodiment of a system comprising a device for applying energy to the nasal septum area with an external electrode and a separate internal mold.
[0044] FIGS. 17A and 17B illustrate an embodiment of a device for applying energy to the nasal septum area comprising an external electrode and an internal mold.
[0045] FIG. 18 shows an embodiment of a device for applying energy to the nasal septum area comprising an array of non-penetrating electrodes.
[0046] FIGS. 19A and 19B illustrate an embodiment of a device for applying energy to the nasal septum area configured for use in only one nostril.
[0047] FIGS. 20A and 20B illustrate an embodiment of a device for applying energy to the nasal septum area configured for use in either nostril.
[0048] FIGS. 21A and 21B illustrate an embodiment of a device for applying energy to the nasal septum area having a symmetrical shape.
[0049] FIGS. 22A-22G illustrate an embodiment of a device for applying energy to the nasal septum area using a monopolar electrode.
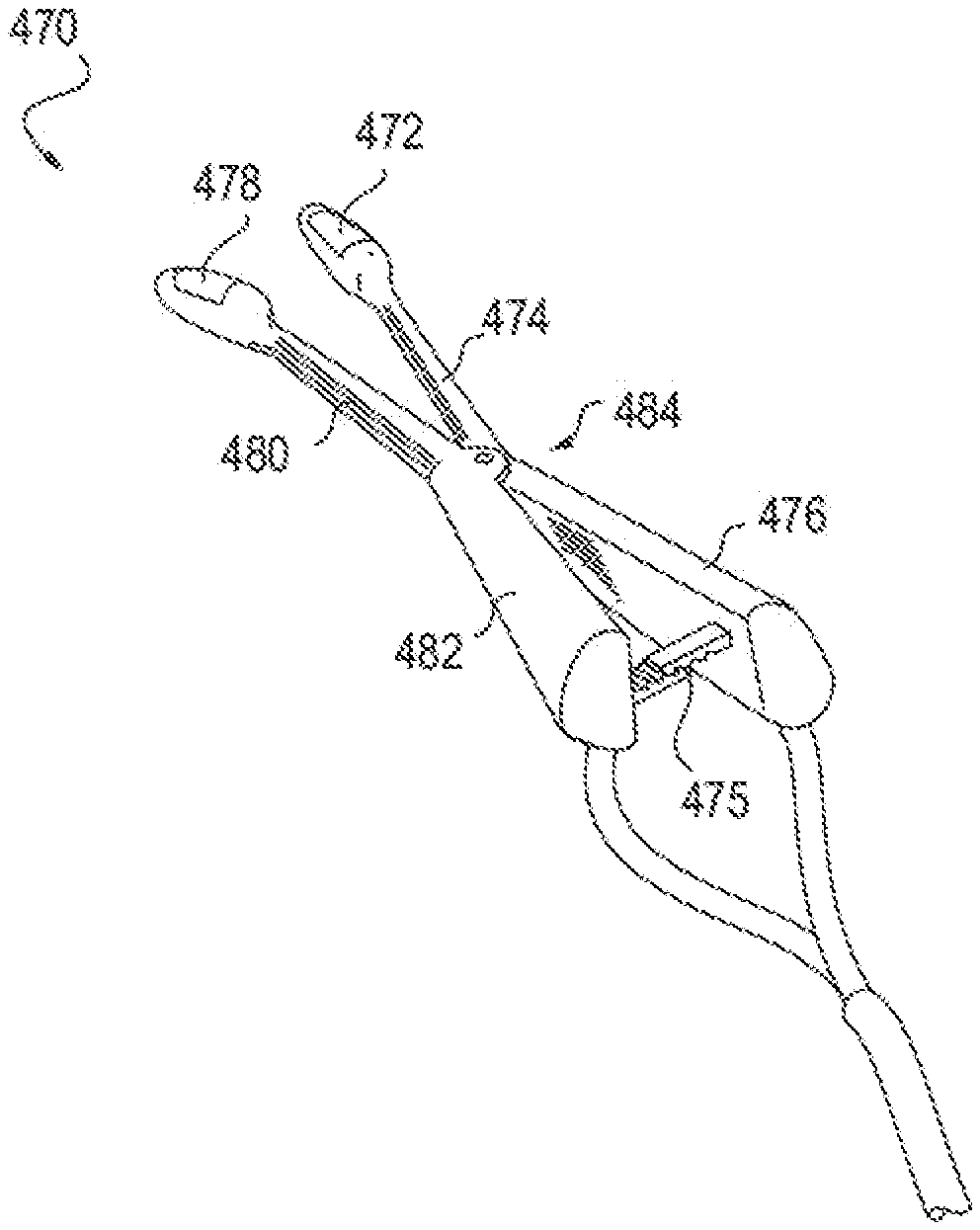
[0050] FIGS. 23A-23G illustrate an embodiment of a device for applying energy to the nasal septum area using an array of needle electrodes.
[0051] FIG. 24A depicts a cross-section of tissue at the nasal septum.
[0052] FIG. 24B depicts heat effects of RF treatment of tissue at the nasal septum.
[0053] FIGS. 25A and 25B illustrate embodiments of devices for applying energy to the nasal septum area incorporating cooling systems.
[0054] FIG. 26 shows an embodiment of a device for applying energy to the nasal septum area incorporating a heat pipe.
[0055] FIG. 27 depicts an embodiment of a device for applying energy to the nasal septum area incorporating heat pipes.
[0056] FIGS. 28A-28E depict embodiments of differential cooling mechanisms.
[0057] FIG. 29 shows an embodiment of a system comprising a device for applying energy to the nasal septum area with electrode needles and a separate cooling mechanism.
[0058] FIGS. 30A-30D show an embodiment of a method for modifying a nasal septum.
[0059] FIGS. 30E-30H show an embodiment of a method for applying energy to the nasal septum area using a device for applying energy to the nasal septum area.
[0060] FIGS. 31A and 31B are a perspective view and a side, cross-sectional view, respectively, of a device for applying energy to the nasal septum area, including an internal power source, according to one embodiment.
[0061] FIGS. 32A and 32B are a cross-sectional side view of facial skin and a front view of a face of a patient, respectively, illustrating use of various embodiments of sensors that may be part of a system for applying energy to the nasal septum area, according to one embodiment.
[0062] FIGS. 33A and 33B are a bottom view of a distal end of a treatment device and a cross-sectional view of a nasal passage, respectively, illustrating wings of the treatment device that may be used to help guide the distal end to a desired location in the nasal passage.
[0063] FIGS. 34A-34C are top views of various alternative embodiments of distal ends of treatment devices having different shapes for addressing differently shaped tissues.
[0064] FIG. 35 is a top view of a treatment device and a cross-sectional view of a portion of a nose, in which the treatment device includes an expandable member, according to one embodiment.
[0065] FIGS. 36A-36E illustrate a method and device for treating a septum having a deviation, according to some embodiments.
[0066] FIG. 37 illustrates a channel stylus device that may be used to treat a deviated septum, according to some embodiments.
[0067] FIGS. 38A-38C illustrate a method of treating a septum having a deviation using a channel stylus device, according to some embodiments.
[0068] FIGS. 39A-39D illustrate a method of treating a deviated septum by treating and evacuating cartilage, according to some embodiments.
DETAILED DESCRIPTION
[0069] The following disclosure provides embodiments of systems and methods for shaping, bending, and/or volumetrically reducing rigid cartilaginous structures, such as hyaline cartilage in a nasal septum. In the case of septal cartilage, shaping, bending, or reducing the cartilage would be useful for reducing nasal obstruction or to improve the cosmetic appearance of the nose. The treatment may improve breathing by correcting deviations in a subject's nasal septum, thereby decreasing airflow resistance or perceived airflow resistance.
[0070] Treatment of hyaline cartilage of the nasal septum presents several unique challenges, any or all of which may be addressed by the embodiments described in this application. For example, shaping and bending of hyaline cartilage may be difficult, due to its stiff and brittle nature. Further, some techniques that are effective at disrupting the matrix of elastic cartilage of certain portions of the nasal valve may not be effective at reshaping hyaline cartilage in the nasal septum, which may need more than the application of energy alone for modification. Disclosed embodiments may provide for modifying hyaline cartilage by, for example, providing methods and devices for softening the cartilage of the nasal septum (e.g., by applying a solution at or near the nasal septum) in addition to reshaping, remodeling, or changing the properties of the tissues.
[0071] While, in some instances, nasal dysfunction can lead to poor airflow, nasal breathing can also be improved in people with normal breathing and/or normal nasal anatomy by decreasing nasal airflow resistance in the nasal valve and associated nasal anatomy. Remodeling or changing the structure of the nasal septum can improve nasal airflow. Prior methods and systems generally involve invasive methods or unsightly devices that a person with normal breathing and/or anatomy may not necessarily be inclined to use or undergo. Thus, there remains an unmet need in the art for non-invasive and minimally invasive methods and devices to decrease nasal airflow resistance or perceived nasal airflow resistance and/or to improve nasal airflow or perceived nasal airflow and the resulting symptoms or sequella of poor nasal airflow--including but not limited to snoring, sleep disordered breathing, perceived nasal congestion and poor quality of life--through the change of structures within the nose that form the passageways for airflow. Methods and devices described herein may be used to treat nasal airways or other cartilaginous areas without the need for more invasive procedures (e.g., ablation or surgery).
[0072] Nasal breathing can be improved in people with normal breathing and/or normal nasal anatomy by decreasing nasal airflow resistance or perceived nasal airflow resistance in the nasal valve and associated nasal anatomy. Restructuring the shape, conformation, angle, strength, and cross sectional area of the nasal septum may improve nasal airflow. Changing the nasal septum can be performed alone or together with other procedures (e.g., surgical procedures), such as those described above. Such methods and devices can lead to improved nasal airflow and/or increased volume of nasal airflow in patients with normal or reduced nasal airflow.
[0073] FIGS. 1 and 2A-C illustrate anatomical elements of a human nose. The lower lateral cartilage (LLC) includes an external component referred to as the lateral crus and an internal component referred to as the medial crus. The medial crus and septal nasal cartilage create a nasal septum that separates the left and right nostrils. Upper lateral cartilage lies between the lower lateral cartilages and the nasal bone. The left ULC is separated from the right ULC by the septal cartilage extending down the bridge of the nose. The opposing edges of the LLC and ULC may move relative to one another. Disposed between the opposing edges is an accessory nasal cartilage. The septal nasal cartilage and the ULC form an angle (.theta.) called the nasal valve angle.
[0074] FIG. 2B illustrates a detailed cross-section of a segment of nose tissue in the area of the intersection of the ULC and the LLC. As shown in the detailed view of FIG. 2A, both inner and outer surfaces of the nasal cartilage are covered with soft tissue including mucosa, sub-mucosa and skin.
[0075] FIG. 2C illustrates a view of the nose as seen from the nostrils. FIG. 2 depicts the nasal valve 1 shown between the septum 2 and the ULC 3. FIG. 2A also depicts the position of the turbinate 4.
[0076] The internal nasal septum area of the nasal airway passage can be visualized prior to and/or during any treatment by any suitable method, including but not limited to direct visualization, endoscopic visualization, visualization by the use of a speculum, transillumination, ultrasound, MRI, x-ray or any other method. In some embodiments, treatments of the nasal septum area as described herein may be performed in conjunction with or following another procedure (e.g., a surgical procedure such as rhinoplasty and/or modification of the nasal valve). In such embodiments, the nasal septum area may be visualized and accessed during surgery. In some embodiments, it may be desirable to visualize the internal nasal septum with minimum disturbance, so as to avoid incorrect assessments due to altering the shape of the nasal septum during visualization. In some embodiments, visualization elements may be incorporated into or combined with treatment devices configured for treating internal and/or external nasal septum.
[0077] Airflow through the nasal passage can be measured prior to and/or during any treatment by any suitable method, including, but not limited to, a nasal cannula connected to a pressure measurement system, rhinomanometry, and rhino-hygrometer. Nasal airflow and resistance can also be evaluated by subjective evaluation before and after a manipulation to increase the cross-sectional area of the nasal passage, such as the Cottle maneuver. In some embodiments, it may be desirable to measure nasal airflow and/or resistance prior to, during and/or after a procedure.
[0078] The nasal septum area of the nasal airway passage can be accessed through the nares. In some embodiments, one or more devices may be used to pull the tip of the nose caudally and increase the diameter of the nares in order to further facilitate access to the nasal septum for treatment. Such devices may include speculum type devices and retractors. In other embodiments, access to the nasal septum may also be achieved endoscopically via the nares, or via the mouth and throat. In some embodiments, visualization devices may be incorporated or combined with treatment devices for treating internal and/or external nasal valves. These and any other access and/or visualization devices may be used with any of the methods and devices below.
[0079] Some embodiments below provide apparatus and methods for modifying cartilage, such as hyaline cartilage. Some embodiments below provide apparatus and methods for modifying the nasal septum and/or modifying the structure and/or structural properties of tissues at or adjacent to the nasal septum.
[0080] In some embodiments, airflow restrictions to the internal nasal valve may be the result of a smaller-than-optimal internal nasal valve angle, shown as .theta. in FIG. 2A. An internal nasal valve angle (i.e., the angle formed between the caudal border of the ULC and the nasal septum) of less than the normally optimal range of between about 10 degrees and about 15 degrees can result in airflow restrictions. Thus, in some embodiments, treatments may be designed to reshape structures at or adjacent to the nasal septum in order to increase the internal nasal valve angle sufficiently that after such treatments, the nasal valve angle falls within the optimal range of about 10-15 degrees. In some embodiments, the internal valve angle may also be increased to be greater than 15 degrees.
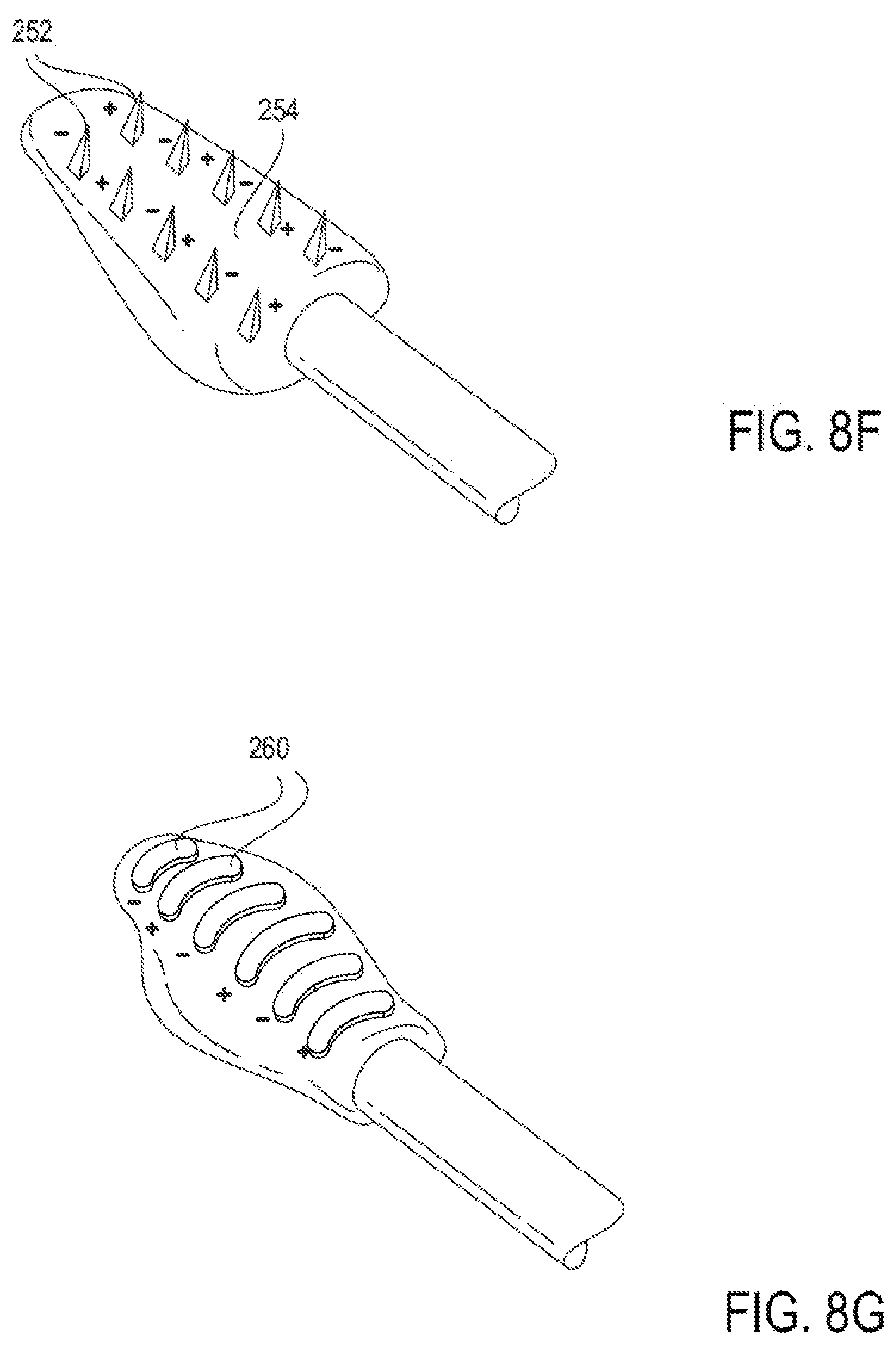
[0081] In some embodiments, airflow restrictions to the internal nasal valve may be the result of a smaller-than-optimal area of the internal nasal valve. An internal nasal valve with a less than optimal area can result in airflow restrictions. Thus, in some embodiments, treatments may be designed to reshape structures at or adjacent to the nasal septum in order to increase the internal nasal valve angle sufficiently that after such treatments, the area of the nasal valve falls within an optimal range. In some embodiments, modifying the nasal septum without increasing the angle of the nasal valve may improve airflow. In some embodiments, increasing the angle of the nasal valve without increasing the area of the opening at the nasal valve may improve airflow. In some embodiments, both the opening at the area of the nasal valve and the angle of the nasal valve may be increased to improve airflow.
[0082] In some embodiments, nasal airflow can be increased in the presence of normal nasal anatomy and/or normal or enlarged nasal valve angle or area.
[0083] With reference to FIG. 2A, in some embodiments, the internal valve angle .theta. or area may be increased by mechanically pressing against the nasal septum. In some embodiments, this pressing may be performed by an inflatable balloon (such as those discussed below with reference to FIGS. 4A-4B), which may be positioned between the upper portion of the nasal septum 20 and the outer lateral wall 22 and then inflated, pressing against the nasal septum until the nasal valve angle reaches a desired size. Similarly, other mechanical devices such as spreaders or retractors (such as those discussed below with reference to FIGS. 5A and 5B) or molds may be used. In alternative embodiments, short-term removable implants may be used to reshape the nasal septum. Some examples of short-term implants may include stents, molds or plugs. In further alternative embodiments, external reshaping elements, such as adhesive strips or face masks may be used to modify the shape of a nasal valve and/or septum. In some embodiments, energy application or other treatments as described below may be applied to substantially fix the reshaped tissue in a desired conformational shape before, during or after applying a mechanical reshaping force (e.g., with the balloon, mechanical devices, molds, short-term implants, or external reshaping elements described above or any of the mechanical devices described below).
[0084] In some embodiments, before, during or after reshaping the nasal tissue, the cartilage of the septum may be softened or otherwise modified. A cartilage softening or dissolving agent may be used to pre-treat cartilage, allowing it to be subsequently bent, shaped and/or reduced by energy application(s) and/or mechanical force. The cartilage may be softened at a surface of the tissue, so as to maintain the mechanical integrity of the nose as a whole but still enable sufficient reshaping, to alleviate the symptoms of a deviation. In some embodiments, the pre-treatment may substantially soften and/or dissolve the cartilage, such that the mechanical integrity of the cartilage is compromised.
[0085] In one embodiment, the cartilage may be treated with solutions via injections, topical applications, or direct infusions from the surface or surfaces of a device. The solutions may be selected to modify properties of the cartilage for subsequent modification. The solutions may include, but are not limited to collagenase, hyaluronidase, tosyl lysyl chloromethane, trypsin, trypsin/EDTA, and/or combinations thereof. Collagenase and trypsin are naturally occurring enzymes in the human body.
[0086] In the case of trypsin, proteoglycan structures of cartilage may be dissolved, leaving a compliant collagen matrix that may be subsequently heated, such as with radiofrequency energy. Heating the compliant matrix may denature, and thus shrink or bend, the cartilage into a more favorable position. Moreover, the heating can be used to stop the propagation of proteoglycan digestion by rendering the trypsin inactive.
[0087] In another method, collagenase may be used to digest both the proteoglycan and collagen matrix of the cartilage. Remaining cartilage and other tissues may then be shaped or further reduced as necessary using radiofrequency heating or by other energy modalities.
[0088] In one embodiment, a device may be configured to inject the solution into or near the tissue to be treated. The device may define the area of application to the cartilage by amount and number of sites injected and/or by providing a physical barrier, such as a ring pressed against the nasal mucosa that corrals/contains the injected enzyme between the mucosa and the cartilage.
[0089] As described above, the solution (e.g., a tissue dissolving agent) may be applied to the tissue (e.g., cartilage) via injections, topical applications, transmucosal delivery devices, and/or other manners. Once the agent has affected the tissue, a heating device is used to reduce and/or shape the tissue by applying energy/heat to the target area. The device is inserted into the nose and held against the target tissue, or alternately applied via a transmucosally inserted device. The energy is applied, and then the device is removed (e.g., once all target tissue has been treated).
[0090] In one embodiment, the heating device may be an RF Stylus. The stylus may include a single- or multi-electrode head, which is configured to apply RF energy to the septal cartilage through the nasal mucosa. The device may also be capable of measuring tissue temperature, impedance, wattage, and/or other properties to effectively alter power to the stylus to maintain desired RF energy and temperature.
[0091] In another embodiment, a reshaping device may be used to expand the diameter of the nasal passage at the nasal septum. The expansion device can be a balloon, a user controlled mechanical device, a self-expanding mechanical device, a fixed shape device, or any combination thereof. The expansion can increase the diameter over the normal range, in order for the diameter to remain expanded after removal of the device and healing of the tissue.
[0092] In some embodiments, a reshaping device may be used to conformationally change the structure of the nasal septum anatomy to allow greater airflow, without necessarily expanding the diameter of the nasal passage.
[0093] In some embodiments, a reshaping or remodeling device can be used to conformationally change the structure of areas of the nasal septum that causes the cross sectional or three dimensional structure of the nasal airway to assume a shape less restrictive to airflow without widening the nasal valve angle.
[0094] In some embodiments, energy may be applied in the form of heat, radiofrequency (RF), laser, light, ultrasound (e.g. high intensity focused ultrasound), microwave energy, electromechanical, mechanical force, cooling, alternating or direct electrical current (DC current), chemical, electrochemical, or others. In some embodiments, the nasal septum, nasal valve, and/or surrounding tissues may be strengthened through the application of cryogenic therapy, or through the injection or application of bulking agents, glues, polymers, collagen and/or other allogenic or autogenic tissues, or growth agents.
[0095] Any one or more of the above energy-application mechanisms may also be used to reshape, remodel, or change mechanical or physiologic properties of structures of a nasal septum or surrounding tissues. For example, in some embodiments, energy may be applied to a targeted region of tissue such that the tissue modification results in a tightening, shrinking or enlarging of such targeted tissues resulting in a change of shape. In some such embodiments, reshaping of a nasal septum section may be achieved by applying energy without necessarily applying a mechanical reshaping force. For example energy can be used to selectively shrink tissue in specific locations of the nasal airway that will lead to a controlled conformational change.
[0096] In some embodiments, strengthening and/or conformation change (i.e., reshaping) of nasal septum tissue to reduce negative pressure during inspiration may include modification of tissue growth and/or the healing and fibrogenic process. For example, in some embodiments energy may be applied to a targeted tissue in the region of the nasal septum in such a way that the healing process causes a change to the shape of the nasal septum and/or a change in the structural properties of the tissue. In some embodiments, such targeted energy application and subsequent healing may be further controlled through the use of temporary implants or reshaping devices (e.g., internal stents or molds, or external adhesive strips).
[0097] In some embodiments, energy may be delivered into the cartilage tissue to cause a conformational change and/or a change in the physical properties of the cartilage. Energy delivery may be accomplished by transferring the energy through the tissue covering the cartilage such as the epithelium, mucosa, sub-mucosa, muscle, ligaments, tendon and/or skin. In some embodiments, energy may also be delivered to the cartilage using needles, probes or microneedles that pass through the epithelium, mucosa, submucosa, muscle, ligaments, tendon and/or skin (as illustrated for example in FIG. 4D).
[0098] In some embodiments, energy may be delivered into the submucosal tissue to cause a conformational change and/or a change in the physical properties of the submucosal tissue. Energy delivery may be accomplished by transferring the energy through the tissue covering the submucosa such as the epithelium, mucosa, muscle, ligaments, cartilage, tendon and/or skin. In some embodiments, energy may also be delivered to the submucosa using needles, probes, microneedles, micro blades, or other non-round needles that pass through the epithelium, mucosa, muscle, ligaments, tendon and/or skin.
[0099] FIG. 3 illustrates an embodiment of a nasal septum treatment device 30. The device 30 comprises a treatment element 32 which may be configured to be placed inside the nasal cavity, nasal passage, and/or nasal airway to deliver the desired treatment. In some embodiments, the device 30 may further comprise a handle section 34 which may be sized and configured for easy handheld operation by a clinician. In some embodiments, a display 36 may be provided for displaying information to a clinician during treatment.
[0100] In some embodiments, the information provided on the display 36 may include treatment delivery information (e.g. quantitative information describing the energy being delivered to the treatment element) and/or feedback information from sensors within the device and/or within the treatment element. In some embodiments, the display may provide information on physician selected parameters of treatment, including time, power level, temperature, electric impedance, electric current, depth of treatment and/or other selectable parameters.
[0101] In some embodiments, the handle section 34 may also comprise input controls 38, such as buttons, knobs, dials, touchpad, joystick, etc. In some embodiments, controls may be incorporated into the display, such as by the use of a touch screen. In further embodiments, controls may be located on an auxiliary device which may be configured to communicate with the treatment device 30 via analog or digital signals sent over a cable 40 or wirelessly, such as via BLUETOOTH, WI-FI (or other 802.11 standard wireless protocol), infrared or any other wired or wireless communication method.
[0102] In some embodiments the treatment system may comprise an electronic control system 42 configured to control the timing, location, intensity and/or other properties and characteristics of energy or other treatment applied to targeted regions of a nasal passageway. In some embodiments, a control system 42 may be integrally incorporated into the handle section 34. Alternatively, the control system 42 may be located in an external device which may be configured to communicate with electronics within the handle section 34. A control system may include a closed-loop control system having any number of sensors, such as thermocouples, electric resistance or impedance sensors, ultrasound transducers, or any other sensors configured to detect treatment variables or other control parameters.
[0103] The treatment system may also comprise a power supply 44. In some embodiments, a power supply may be integrally incorporated within the handle section 34. In alternative embodiments, a power supply 44 may be external to the handle section 34. An external power supply 44 may be configured to deliver power to the handle section 34 and/or the treatment element 32 by a cable or other suitable connection. In some embodiments, a power supply 44 may include a battery or other electrical energy storage or energy generation device. In other embodiments, a power supply may be configured to draw electrical power from a standard wall outlet. In some embodiments, a power supply 44 may also include a system configured for driving a specific energy delivery technology in the treatment element 32. For example, the power supply 44 may be configured to deliver a radio frequency alternating current signal to an RF energy delivery element. Alternatively, the power supply may be configured to deliver a signal suitable for delivering ultrasound or microwave energy via suitable transducers. In further alternative embodiments, the power supply 44 may be configured to deliver a high-temperature or low-temperature fluid (e.g. air, water, steam, saline, or other gas or liquid) to the treatment element 32 by way of a fluid conduit.
[0104] In some embodiments, the treatment element 32 may have a substantially rigid or minimally elastic shape sized and shaped such that it substantially conforms to an ideal shape and size of a patient's nasal passageway, including the nasal septum. In some embodiments, the treatment element 32 may have a curved shape, either concave or convex with respect to the interior of the lateral wall of the nasal passage. In some embodiments, the shape of a fixed-shape treatment element may be substantially in a shape to be imparted to the cartilage or other structures of the nasal septum area.
[0105] In some embodiments, the treatment element 32 and control system 42 may be configured to modify the properties of cartilage and/or create specific localized tissue damage or ablation without the application of energy. For example, the treatment element 32 may be configured to apply (e.g., inject) a solution that modifies the properties of cartilage (e.g., collagenase, hyaluronidase, tosyllysylchloromethane, trypsin, trypsin/EDTA, and/or combinations thereof) at or near the tissue to be treated. In one embodiment, for example, the treatment element 32 may include one or more needles to inject the solution (e.g., into the space between the septal cartilage and the mucosal layer). In another embodiment, the treatment element 32 may be configured to chemically cauterize tissue around a nasal septum by delivering a cauterizing solution (e.g., silver nitrate, trichloroacetic acid, cantharidin, etc.) to the tissue. The treatment element 32 may include apertures configured to permit the solution to pass through to the nose. In some embodiments, the treatment element 32 may aerosolize the solution. Other delivery methods are also contemplated. The treatment element 32 may comprise a lumen, through which the solution passes. The lumen may be fluidly connected to a reservoir or container holding the solution. The device may include an input control (e.g., a button or switch) configured to control the delivery of the solution. In some embodiments, the treatment element 32 may include an applicator that can be coated in the solution (e.g., dipped in a reservoir of solution, swabbed with the solution, etc.) and the coated treatment element applicator may be applied to tissue to be treated. In some embodiments, the treatment element may be configured to apply the solution to the patient over a prolonged period of time (e.g., 30 seconds, 1 minute, 2 minutes, etc.).
[0106] In some embodiments, the treatment element 32 comprises shields or rings configured to protect tissue surrounding the tissue to be treated from coming into contact with the solution. In some embodiments, a separate element is used to shield tissue surrounding the tissue to be treated from coming into contact with the solution. While such treatments may be performed without the application of energy, in some embodiments, they may be performed in conjunction with energy treatments. In one embodiment, one or more shields may be configured to be placed on the nasal septum mucosa around the site of the deviation. In one embodiment, for example, there may be two rings--one for each side of the septum. The design of the device may be "U" shaped to fit the two arms of device into the two nostrils of the patient. The device may be spring loaded, such that when a user releases the device, the device is held in place by its spring force.
[0107] In some embodiments, as shown for example in FIG. 3, the treatment element 32 may comprise a substantially cylindrical central portion with a semi-spherical or semi-ellipsoid or another shaped end-cap section at proximal and/or distal ends of the treatment element 32. In alternative embodiments, the treatment element may comprise a substantially ellipsoid shape as shown, for example in FIGS. 4A-4D. In some embodiments, an ellipsoid balloon as shown in FIG. 4A may have an asymmetrical shape. In alternative embodiments, the treatment element 32 may have an asymmetrical "egg-shape" with a large-diameter proximal end and a smaller-diameter distal end. In some embodiments, the element 32 can be shaped so as to impart a shape to the tissue treated that is conducive to optimal nasal airflow. Any suitable solid or expandable medical balloon material and construction available to the skilled artisan may be used.
[0108] FIG. 4B illustrates an embodiment of a treatment element configured to deliver energy to an interior of a nose. In some embodiments, the treatment element of FIG. 4B also includes an expandable balloon.
[0109] FIG. 4C illustrates an embodiment of a bifurcated treatment element 70 having a pair of semi-ellipsoid elements 72, 74 sized and configured to be inserted into the nose with one element 72, 74 on either side of the septum. The elements may each have a medial surface 75a & 75b which may be substantially flat, curved or otherwise shaped and configured to lie adjacent to (and possibly in contact with) the nasal septum. In some embodiments, the elements 72, 74 may include expandable balloons with independent inflation lumens 76, 78. In alternative embodiments, the elements 72, 74 have substantially fixed non-expandable shapes. In still further embodiments, the elements 72, 74 may include substantially self-expandable sections. In some embodiments, the bifurcated treatment element halves 72, 74 may also carry energy delivery structures as described elsewhere herein. In some embodiments, the shape of the elements 72, 74 may be modified by the operator to impart an optimal configuration to the treated tissue. The shape modification of elements 72, 74 can be accomplished pre-procedure or during the procedure and can be either fixed after modification or capable of continuous modification.
[0110] In some embodiments, a nasal septum treatment system may also comprise a reshaping device configured to mechanically alter a shape of soft tissue and/or cartilage in a region of the nasal septum in order to impart a desired shape and mechanical properties to the tissue of the walls of the nasal airway. In some embodiments the reshaping device may be configured to reshape the nasal septum and/or nasal valve into a shape that improves the patency of one or both nasal valve sections at rest and during inspiration and/or expiration. In some embodiments, the reshaping device may comprise balloons, stents, mechanical devices, molds, external nasal strips, spreader forceps or any other suitable structure. In some embodiments, a reshaping device may be integrally formed with the treatment element 32. In alternative embodiments, a reshaping device may be provided as a separate device that may be used independently of the treatment element 32. As described in more detail below, such reshaping may be performed before, during or after treatment of the nose tissue with energy, injectable compositions or cryo-therapy.
[0111] With reference to FIGS. 4A-4C, some embodiments of treatment elements 32 may comprise one or more inflatable or expandable sections configured to expand from a collapsed configuration for insertion into the nasal passageway, to an expanded configuration in which some portion of the treatment element 32 contacts and engages an internal surface of a nasal passageway. In some embodiments, an expandable treatment element may comprise an inflation lumen configured to facilitate injection of an inflation medium into an expandable portion of the treatment element. In alternative embodiments, an expandable treatment element may comprise one or more segments comprising a shape-memory alloy material which may be configured to expand to a desired size and shape in response to a change of temperature past a transition temperature. In some embodiments, such a temperature change may be brought about by activating an energy-delivery (or removal) element in the treatment element 32.
[0112] In some embodiments, the treatment element 32 may expand with various locations on the element expanding to different configurations or not expanding at all to achieve a desired shape of the treatment element. In some embodiments, such expandable treatment elements or sections may be elastic, inelastic, or preshaped. In some embodiments, expandable treatment elements or sections thereof may be made from shape-memory metals such as nickel-cobalt or nickel-titanium, shape memory polymers, biodegradable polymers or other metals or polymers. Expandable balloon elements may be made of any elastic or inelastic expandable balloon material.
[0113] In alternative embodiments, the treatment element 32 can act to change the properties of the internal soft tissue of the nasal airway in conjunction with an external treatment device of fixed or variable shape to provide additional force to change the shape of the nasal septum. In some embodiments, an external mold element can be combined with an internal element.
[0114] FIGS. 5A and 5B illustrate reshaping treatment devices 80 and 90, respectively. The treatment devices 80 and 90 are structured as clamp devices configured to engage a targeted section of the nasal airway with either a clamping force or a spreading force. In some embodiments, the treatment devices of FIGS. 5A and 5B may include energy delivery elements (of any type described herein) which may be powered by a fluid lumen or cable 86.
[0115] The treatment device of FIG. 5A includes an outer clamp member 82 and an inner clamp member 84 joined at a hinge point 85. In some embodiments, the outer clamp member 82 may include an outwardly-bent section 83 sized and configured to extend around the bulk of a patient's nose when the inner clamp member may be positioned inside the patient's nose. The inner and outer tissue-engaging tips at the distal ends of the inner and outer clamp members may be configured to impart a desired shape to the nasal septum. In some embodiments, the tissue-engaging tips may be removable to allow for sterilization and/or to allow for tips of a wide range of shapes and sizes to be used with a single clamp handle.
[0116] The treatment device of FIG. 5B includes an outer clamp member 92 and an inner clamp member 94 joined at a hinge point 95. The inner and outer tissue-engaging tips at the distal ends of the inner and outer clamp members may be configured to impart a desired shape to the nasal cartilage. In the illustrated embodiment, the outer clamp member 92 includes a concave inner surface, and the inner clamp member includes a mating convex inner surface. The shape and dimensions of the mating surfaces may be designed to impart a desired shape to the structures of a patient's nose. In some embodiments, the shape of the mating surfaces may be modified by the operator to impart an optimal configuration to the treated tissue. The shape modification of the mating surfaces can be accomplished pre-procedure or during the procedure and can be either fixed after modification or capable of continuous modification.
[0117] In some embodiments, the tissue-engaging tips may be removable to allow for sterilization and/or to allow for tips of a wide range of shapes and sizes to be used with a single clamp handle.
[0118] In alternative embodiments, the devices of FIGS. 5A and 5B may be used as spreader devices by placing both clamp tips in a nasal passage and separating the handles, thereby separating the distal tips. In alternative embodiments, the handles may be configured to expand in response to a squeezing force. The shapes of the distal tips may be designed to impart a desired shape when used as a spreading device.
[0119] The reshaping elements of FIGS. 3-5B are generally configured to be used once and removed from a patient's nose once a treatment is delivered. In some embodiments, treatments may further involve placing longer term treatment elements, such as stents, molds, external strips, etc. for a period of time after treatment. An example of such a stent placed within a patient's nose after treatment is shown in FIG. 6. In some embodiments, the stent may be configured to be removed after a therapeutically effective period of time following the treatment. In some embodiments, such a therapeutically effective period of time may be on the order of days, weeks or more.
[0120] In some embodiments, the treatment element 32 may be configured to deliver heat energy to the nasal septum. In such embodiments, the treatment element may comprise any suitable heating element available to the skilled artisan. For example, the treatment element 32 may comprise electrical resistance heating elements. In alternative embodiments, the heating element may comprise conduits for delivering high-temperature fluids (e.g. hot water or steam) onto the nasal tissue. In some embodiments, a high-temperature fluid heating element may comprise flow channels which place high-temperature fluids into conductive contact with nasal tissues (e.g. through a membrane wall) without injecting such fluids into the patients nose. In further embodiments, any other suitable heating element may be provided. In further embodiments, the treatment element 32 may comprise elements for delivering energy in other forms such as light, laser, RF, microwave, cryogenic cooling, DC current and/or ultrasound in addition to or in place of heating elements.
[0121] U.S. Pat. No. 6,551,310 describes embodiments of endoscopic treatment devices configured to ablate tissue at a controlled depth from within a body lumen by applying radio frequency spectrum energy, non-ionizing ultraviolet radiation, warm fluid or microwave radiation. U.S. Pat. No. 6,451,013 and related applications referenced therein describe devices for ablating tissue at a targeted depth from within a body lumen. Embodiments of laser-treatment elements are described for example in U.S. Pat. No. 4,887,605, among others. U.S. Pat. No. 6,589,235 teaches methods and device for cartilage reshaping by radiofrequency heating. U.S. Pat. No. 7,416,550 also teaches methods and devices for controlling and monitoring shape change in tissues, such as cartilage. The devices described in these and other patents and publications available to the skilled artisan may be adapted for use in treating portions of a nasal septum or adjacent tissue as described herein. U.S. Pat. Nos. 7,416,550, 6,589,235, 6,551,310, 6,451,013 and 4,887,605 are hereby incorporated by reference in their entireties for any and all purposes.
[0122] In alternative embodiments, similar effects can be achieved through the use of energy removal devices, such as cryogenic therapies configured to transfer heat energy out of selected tissues, thereby lowering the temperature of targeted tissues until a desired level of tissue modification is achieved. Examples of suitable cryogenic therapy delivery elements are shown and described for example in U.S. Pat. Nos. 6,383,181 and 5,846,235, the entirety of each of which is hereby incorporated by reference for any and all purposes.
[0123] In some embodiments, the treatment element 32 may be configured to deliver energy (e.g. heat, RF, ultrasound, microwave) or cryo-therapy uniformly over an entire outer surface of the treatment element 32, thereby treating all nasal tissues in contact with the treatment element 32. Alternatively, the treatment element 32 may be configured to deliver energy at only selective locations on the outer surface of the treatment element 32 in order to treat selected regions of nasal tissues. In such embodiments, the treatment element 32 may be configured so that energy being delivered to selected regions of the treatment element can be individually controlled. In some embodiments, portions of the treatment element 32 are inert and do not deliver energy to the tissue. In further alternative embodiments, the treatment element 32 may be configured with energy-delivery (or removal) elements distributed over an entire outer surface of the treatment element 32. The control system 42 may be configured to engage such distributed elements individually or in selected groups so as to treat only targeted areas of the nasal passageway.
[0124] In some embodiments, the treatment element 32 may be a balloon with energy delivery elements positioned at locations where energy transfer is sufficient or optimal to effect change in breathing. Such a balloon may be configured to deliver energy while the balloon is in an inflated state, thereby providing a dual effect of repositioning tissue and delivering energy to effect a change the nasal septum. In other embodiments, a balloon may also deliver heat by circulating a fluid of elevated temperature though the balloon during treatment. The balloon can also deliver cryotherapy (e.g., by circulating a low-temperature liquid such as liquid nitrogen) while it is enlarged to increase the nasal valve diameter or otherwise alter the shape of a nasal septum. FIG. 7 illustrates an example of an energy-delivery balloon being inserted into a patient's nose for treatment. Several embodiments may be employed for delivering energy treatment over a desired target area. For example, in some embodiments, a laser treatment system may treat a large surface area by scanning a desired treatment pattern over an area to be treated. In the case of microwave or ultrasound, suitably configured transducers may be positioned adjacent to a target area and desired transducer elements may be activated under suitable depth focus and power controls to treat a desired tissue depth and region. In some embodiments, ultrasound and/or microwave treatment devices may also make use of lenses or other beam shaping of focusing devices or controls. In some embodiments, one or more electrical resistance heating elements may be positioned adjacent to a target region, and activated at a desired power level for a therapeutically effective duration. In some embodiments, such heating elements may be operated in a cyclical fashion to repeatedly heat and cool a target tissue. In other embodiments, RF electrodes may be positioned adjacent to and in contact with a targeted tissue region. The RF electrodes may then be activated at some frequency and power level therapeutically effective duration. In some embodiments, the depth of treatment may be controlled by controlling a spacing between electrodes. In alternative embodiments, RF electrodes may include needles which may puncture a nasal septum tissue to a desired depth (as shown for example in FIG. 4D and in other embodiments below).
[0125] In some embodiments, the treatment element 32 and control system 42 may be configured to deliver treatment energy or cryotherapy to a selected tissue depth in order to target treatment at specific tissues. For example, in some embodiments, treatments may be targeted at tightening sections of the epithelium of the nasal septum. In other embodiments, treatments may be targeted at strengthening soft tissues underlying the epithelium. In further embodiments, treatments may be targeted at strengthening cartilage in the area of the nasal valve and/or upper lateral cartilage. In still further embodiments, treatments may be targeted at stimulating or modifying the tissue of muscles of the nose or face in order to modify the nasal septum.
[0126] In some embodiments, the treatment element 32 and control system 42 may be configured to deliver treatment energy to create specific localized tissue damage or ablation, stimulating the body's healing response to create desired conformational or structural changes in the nasal tissue.
[0127] In some embodiments, a treatment element may be configured to treat a patient's nasal septum by applying treatment (e.g., energy, cryotherapy, or other treatments) from a position outside the patient's nose. For example, in some embodiments, the devices of FIGS. 5A and 5B may be configured to apply energy from an element positioned outside a patient's nose, such as on the skin. In another embodiment, a device may be placed on the external surface of the nose that would pull skin to effect a change in the nasal airway. Treatment may then be applied to the internal or external nasal tissue to achieve a desired nasal septum configuration.
[0128] In some embodiments, the device is configured to position tissue to be reshaped. In some embodiments, the device comprises features and mechanisms to pull, push or position the nasal tissue into a mold for reshaping. For example, suction, counter traction, or compression between two parts of the device may be used.
[0129] In some embodiments, the treatment device comprises one, two, three, four, or more molds configured to reshape tissue. The mold or reshaping element may be fixed in size or may vary in size. The mold may also be fixed in shape or may vary in shape. For example, the size or shape of the element may be varied or adjusted to better conform to a nasal septum of a patient. Adjustability may be accomplished using a variety of means, including, for example, mechanically moving the mold by way of joints, arms, guidewires, balloons, screws, stents, and scissoring arms, among other means. The mold may be adjusted manually or automatically. The mold is configured to impart a shape to the tissues of the nasal septum area to improve airflow or perceived airflow.
[0130] In some embodiments, the mold or reshaping element comprises a separate or integrated energy delivery or treatment element (e.g., an electrode such as those described below with respect to FIGS. 8A-8J). The treatment element may be fixed or adjustable in size. For example, the treatment element may be adjusted to better conform to the nasal septum of a patient. In the case of a separate reshaping element and treatment element, a distance between the two elements may either be fixed or adjustable. Adjustability may be accomplished using a variety of means, including, for example, mechanically moving the mold by way of joints, arms, guidewires, balloons, screws, stents, and scissoring arms, among other means.
[0131] In some embodiments, the mold or another part of the device is configured to deliver cooling (discussed in more detail below). In some embodiments, the mold or reshaping element comprises a balloon configured to reshape and/or deform tissue. A balloon may also be configured to deliver energy such as heat using hot liquid or gas.
Examples of Various Electrode Arrangements
[0132] Described below are embodiments of various treatment devices and, more particularly, electrode arrangements that may be used for applying energy to tissue, such as hyaline cartilage of the nasal septum area. These electrodes may, for example, deliver RF energy to preferentially shape the tissue to provide improved nasal breathing. In some embodiments, one or more electrodes may be used alone or in combination with a tissue shaping device or mold. In other embodiments, one or more electrodes may be integrally formed with a tissue shaping device or mold, so that the electrodes themselves create the shape for the tissue. In some embodiments, the energy delivery devices may use alternating current. In some embodiments, the energy delivery devices may use direct current. In certain such embodiments, the energy delivery device may comprise a configuration utilizing a grounding pad.
[0133] In some embodiments, the term "electrode" refers to any conductive or semi-conductive element that may be used to treat the tissue. This includes, but is not limited to metallic plates, needles, and various intermediate shapes such as dimpled plates, rods, domed plates, etc. Electrodes may also be configured to provide tissue deformation in addition to energy delivery. Unless specified otherwise, electrodes described can be monopolar (e.g., used in conjunction with a grounding pad) or bipolar (e.g., alternate polarities within the electrode body, used in conjunction with other tissue-applied electrodes).
[0134] In some embodiments, "mold", "tissue shaper", "reshaping element" and the like refer to any electrode or non-electrode surface or structure used to shape, configure or deflect tissue during treatment.
[0135] In some embodiments, "counter-traction" refers to applying a force opposite the electrode's primary force on the tissue to increase stability, adjustability, or for creating a specific shape.
[0136] As shown in FIG. 8A, in some embodiments, bipolar electrodes may be used to deliver energy, with one electrode 202 placed internally in the nose, for example against the nasal septum, and one electrode 204 placed externally on the outside of the nose. This embodiment may advantageously provide direct current flow through the tissue with no physical trauma from needles (as shown in some embodiments below). As shown in FIG. 8B, in some embodiments, bipolar electrodes may be used to deliver energy, with both electrodes 210, 212 placed internally. An insulating spacer 214 may be placed between them. This embodiment may be simple and may advantageously minimize current flow through the skin layer. In some embodiments, bipolar electrodes 220, 222 may be both placed externally and may be connected to a passive molding element 224 placed inside the nasal septum adjacent to tissue to be treated, as shown in FIG. 8C. This embodiment may advantageously minimize the potential for mucosal damage. In some embodiments, electrodes placed internally may be shaped to function as a mold or may comprise an additional structure that may function as a mold.
[0137] In some embodiments, a monopolar electrode may be used to deliver energy. As shown in FIG. 8D, the electrode 230 may be placed internally and may be connected to an external, remote grounding pad 232. The grounding pad 232 may, for example, be placed on the abdomen of a patient or in other desired locations. This embodiment may advantageously be simple to manufacture and may minimize current flow through the skin. In some embodiments, a monopolar electrode may be placed externally and may be connected to a molding element placed at the nasal septum as well as a remote grounding pad. This embodiment may also advantageously be simple to manufacture, may minimize mucosal current flow, and may also be simple to position. In some embodiments, electrodes placed internally may be shaped to function as a mold or may comprise an additional structure that may function as a mold.
[0138] In some embodiments, monopolar transmucosal needles may be used to deliver energy. The needle electrodes 240 may be placed internally, as shown in FIG. 8E penetrating through the mucosa to the cartilage, and a remote grounding pad 242 or element may be placed externally. In some embodiments, monopolar transmucosal needles may be used in conjunction with one or more molding elements which may be disposed on or around the needles. In some embodiments, monopolar transdermal needles may be used to deliver energy. In other embodiments (not shown), the needles may be placed external to the nose, and penetrate through to tissue to be treated. Needle configurations may advantageously target the cartilage tissue to be treated specifically. The monopolar transdermal needles may be used in conjunction with an internal molding device (not shown).
[0139] In some embodiments, bipolar transmucosal needles may be used to deliver energy to tissue to be treated. The needles may be placed internally, with an insulating spacer between them and may penetrate through the mucosa to the cartilage to be treated. In some embodiments, the bipolar transmucosal needles may be used in combination with one or more internal molding elements. The one or more molding elements may be placed on or near the needles. In some embodiments, bipolar transdermal needles may be used to deliver energy. In other embodiments, the transdermal needles may be placed externally and penetrate through to tissue to be treated. Needle configurations may advantageously target the cartilage tissue to be treated specifically. The transdermal bipolar needles may be used in conjunction with an internal molding element.
[0140] As shown in FIG. 8F, in some embodiments, an array of electrodes comprising one, two, or many pairs of bipolar needles 252 are located on a treatment element configured to be placed into contact with the cartilage. An insulator 254 may be disposed between the bipolar needles 252. An insulator may also be used on part of the needle's length to allow energy to be delivered only to certain tissue structures, such as cartilage. The electrodes may be placed either internally or transmucosally or they may be placed externally or transdermally. In the embodiment illustrated in FIG. 8F, the insulator 254 may also function as a mold or molding element. In other embodiments (not shown), the array of electrodes is used in conjunction with a separate tissue reshaping element.
[0141] FIG. 8G illustrates another embodiment of a treatment element comprises one, two or many pairs of bipolar electrodes 260. As opposed to FIG. 8F, where the pairs of electrodes are arranged side-by-side, the embodiment of FIG. 8G arranges the pairs of electrodes along the length of the treatment element. The electrodes of FIG. 8G are also non-penetrating, in contrast to the needles of FIG. 8F. The electrodes 260 may be placed against either the skin, externally, or the mucosa, internally as a means of delivering energy to target tissue such as cartilage.
[0142] In some embodiments of treatment devices comprising an array or multiple pairs of electrodes, each pair of electrodes (bipolar) or each electrode (monopolar) may have a separate, controlled electrical channel to allow for different regions of the treatment element to be activated separately. For example, the needles or needle pairs of FIG. 8F may be individually controlled to produce an optimal treatment effect. For another example, the separate electrodes of FIGS. 8B and 8C may be individually controlled to produce an optimal treatment effect. Other examples are also contemplated. The channels may also comprise separate or integrated feedback. This may advantageously allow for more accurate temperature control and more precise targeting of tissue. Separate control may also allow energy to be focused and/or intensified on a desired region of the treatment element in cases where the anatomy of the nasal tissue/structures does not allow the entire electrode region of the treatment element to engage the tissue. In such embodiments, the nasal tissue that is in contact with the treatment element may receive sufficient energy to treat the tissue.
[0143] Combinations of the described electrode configurations may also be used to deliver energy to tissue to be treated (e.g., by being reshaped). For example, transmucosal needles 264 may be placed internally, penetrating through to the tissue to be treated, and an electrode 266 may be placed externally or on an opposite side of the tissue to be treated, as shown in FIG. 8H. This embodiment may advantageously target the cartilage tissue specifically and be biased for mucosal preservation. For another example, transdermal needles 268 may be inserted on an opposite side of the nasal septum from an electrode 270, as shown in FIG. 8I. This embodiment may advantageously target the cartilage tissue specifically and be biased towards mucosal preservation. For another example bipolar needle electrodes 272, 274 can be placed on both sides of tissue to be treated, as shown in FIG. 8J. This embodiment may advantageously target the cartilage tissue specifically. Some embodiments of treatment elements may include inert areas which do not delivery energy to the tissue. Other combinations of electrode configuration are also possible.
Examples of Treatment Devices Including Electrodes
[0144] Embodiments of treatment devices incorporating treatment elements such as the electrodes described above are illustrated in FIGS. 9A-21B. The instrument designs described in these embodiments may be used in a device such as the device 30, described above, and in the system of FIG. 3. In some embodiments, the devices provide tissue reshaping or molding in addition to energy delivery. Applying energy to the nasal tissue may require properly positioning the electrode(s) at the nasal septum, deflecting or deforming the tissue into a more functional shape, and delivering or applying energy consistently prior to device removal. Embodiments described herein may advantageously provide adjustability, visualization of effect, ease of use, ease of manufacturability and component cost. Molding and reshaping of the tissues of the nose may allow for non-surgical nasal breathing improvement without the use of implants.
[0145] FIG. 9A depicts a device 300 comprising a single inter-nasal monopolar electrode 301 located at the end of a shaft 302. The shaft is attached to a handle 303. The electrode configuration may be similar to that described with respect to FIG. 8D. FIG. 9B depicts another device 304 comprising a single inter-nasal, monopolar electrode 305. The electrode 305 is located at the distal end of a shaft 306, which is attached to a handle 307. The handle comprises a power button 308 that may be used to activate and deactivate the electrode. As stated above, the device 304 may either comprise a generator or be connected to a remote generator. The electrode 305 may be provided on an enlarged, distal end of the shaft 306, and in the embodiment illustrated has a convex shape configured to press against and create a concavity in the nasal cartilage.
[0146] FIG. 10A depicts a side view of a device 310 comprising a single inter-nasal electrode 312 located at the end of a shaft 314. The shaft is attached to a handle 316. An external mold 318 is attached to the handle 316 and can be moved relative to the electrode shaft 314. The external mold 318 has a curved shape with an inner concave surface that may be moved in order to press against an external surface of a patient's nose to compress tissue between the external mold 318 and the electrode 312. FIG. 10B provides a front view of the device 310.
[0147] FIG. 11A depicts a device 320 comprising a single inter-nasal electrode 322 attached to the end of a shaft 324. The shaft 324 is attached to a handle 326. An internal shaft 328 comprising a tissue-contacting surface is attached to the handle 326. The internal shaft 328 can be moved relative to the electrode shaft 324 and may provide counter-traction and/or positioning. For example, when the electrode 322 is placed against a patient's nasal septum, the counter-traction element 328 may be pressed against the patient's upper or lower lateral cartilage.
[0148] FIG. 11B depicts a device 450 similar to device 320 of FIG. 11A comprising an inter-nasal electrode 451 located at a distal end of a shaft 452 connected to a handle 454. The device 450 further comprises a counter-traction element 456 connected to a handle 458. Like the device 320 depicted in FIG. 11A, the connection 460 between the two handles 454, 458 is such that squeezing the two handles 454, 458 together causes the electrode 451 and the counter-traction element 456 to move away from each other, spreading the tissue they are contacting.
[0149] FIG. 12A depicts a device 330 comprising a single inter-nasal electrode 332 located at the end of a shaft 334. The shaft 334 is attached to a handle 336. The device 330 comprises another single inter-nasal electrode 338 attached to the end of a shaft 340. The shaft 340 is attached to a handle 342. The device comprises a connection 344 between the two handles 336, 342 that allows simultaneous deformation and treatment of both nostrils.
[0150] FIG. 12B depicts a device 470 similar to device 330 of FIG. 12A comprising a first inter-nasal electrode 472 located at a distal end of a shaft 474 connected to a handle 476. The device 470 comprises a second inter-nasal electrode 478 located at a distal end of a second shaft 480 connected to a second handle 482. The connection 484 between the two handles 476, 482 is such that squeezing the handles 476, 482 together causes the electrodes 472, 478 to move together, compressing tissue therebetween. The device 470 comprises a ratcheting mechanism 475 between the two handles 476, 482 that allows the relative positions of the electrodes 472, 478 to be locked during treatment.
[0151] FIG. 13A depicts a side view of a device 350 also used for treating two nostrils comprising an inter-nasal electrode 352 attached to the end of a shaft 354. The shaft 354 is attached to a handle 356. As seen in the front view provided in FIG. 13B, the device 350 comprises a second inter-nasal electrode 358. The second inter-nasal electrode 358 is attached to the end of a shaft which is attached to a handle. A connection between the two handles allows simultaneous deformation and treatment of the nostrils. An external mold 366 is attached to the handles. The mold 366 may be moved relative to the electrode shafts 354, 360 and may provide counter-traction (e.g., against the bridge of the nose) and positioning.
[0152] FIGS. 13C-E depicts a device 490 similar to the device 350 shown in FIG. 13A and FIG. 13B. FIGS. 13C and 13D depict side and top views of a device 490 comprising a handle 492. The handle 492 bifurcates into a first shaft 494 with a first inter-nasal electrode 496 located at a distal end of the shaft 494 and a second shaft 498 with a second inter-nasal electrode 500 located at a distal end of the shaft 498. The device 490 comprises a mold 502 configured to provide counter-traction or compression of the bridge of the nose. The mold 502 comprises a handle 504. The connection 506 between the handles 492, 504 is such that squeezing the two handles 492, 504 causes the electrodes 496, 500 and the mold 502 to be compressed together. FIG. 13E depicts the device 490 being used on a patient. The arrows indicate the directions in which the handles 492, 504 are configured to be squeezed.
[0153] FIG. 14A depicts a front view of a device 370 comprising an inter-nasal electrode 372 attached to the end of a shaft 374 (shown in top view of FIG. 14B). The shaft 374 is attached to a handle 376. The device 370 comprises a second inter-nasal electrode 378 attached to the end of a second shaft 380. The second shaft 380 is attached to a second handle 382. A connection 384 between the two handles 376, 382 may allow simultaneous deformation and treatment of the nostrils. External molds 386, 388 are attached to the handles and can be moved relative to each electrode shaft 374, 380. The molds 386, 388 may provide counter-traction, compression of tissue, positioning, and external tissue deformation.
[0154] FIG. 15A depicts a front view of device 390 comprising a first inter-nasal electrode 392 and a second inter-nasal electrode 398. As shown in the side view of FIG. 15B, the device 390 comprises a first inter-nasal electrode 392 attached to the end of a shaft 394. The shaft is attached to a handle 396. A second inter-nasal electrode 398 is attached to the end of a second shaft 400, as shown in the top view of FIG. 15C. The second shaft 400 is attached to a second handle 402. A connection 404 between the two handles 396, 402 may allow simultaneous deformation and treatment of the nostrils. Additional internal shafts 406, 408 comprise tissue-contacting surfaces and are attached to the handles 396,402. The internal shafts 406, 408 may be moved relative to each electrode shaft 394, 400 (shown in FIG. 15B) and may provide counter-traction and positioning.
[0155] FIG. 16 depicts a system 410 comprising a first device having an extra-nasal electrode 412 along a concave surface configured to be positioned against an external surface of a patient's nose, the electrode 412 being attached to the end of a shaft 414. The shaft 414 is attached to a handle 416. A separate device 417 comprising an internal tissue mold 418 is attached to a shaft 420. The internal tissue mold is configured to be positioned inside the patient's nose. The shaft 420 is attached to a handle 422. Each handle 422, 416 may be manipulated individually and may apply energy and deformation to create a desired tissue effect.
[0156] FIG. 17A depicts a side view of a device 430 comprising an extra-nasal electrode 431 attached to the end of a shaft 432. The shaft 432 is attached to a handle 434. The device 430 also comprises an internal tissue mold 436 attached to a shaft 438 which is attached to a handle 440. The handles 434, 440 are attached together and may be moved relative to each other to simultaneously deliver energy and deform tissue. FIG. 17B depicts a front view of the device 430.
[0157] FIG. 18 depicts a device 390 comprising pairs of bipolar electrodes 392 located at the distal end of a shaft 394. The electrodes may be similar to the electrodes described with respect to the electrode configuration of FIG. 8G in that they are non-penetrating. The shaft 394 is connected to a handle 398 which comprises a button 395 configured to activate and deactivate the electrodes. As stated above, the device 390 may either comprise a generator or be connected to a remote generator.
[0158] FIG. 19A depicts the treatment element 503 of a treatment device (e.g., device 30). The treatment element 503 of the device comprises a monopolar electrode 505. A cross-section of the treatment element 503 is shown in FIG. 19B. It comprises an asymmetrical shape and has a convex surface where the electrode is positioned configured to conform to only one of a patient's nostrils (for example, a patient's right nostril). More specifically, the convex surface is configured such that when inserted into the particular nostril, the convex surface would be located adjacent the nasal septum. The treatment element 503 further comprises a light 507 configured to illuminate the treatment area. For example an LED or a visible laser may be used. The visible laser may experience less diffusion in the tissue. Furthermore, the light 507 can be situated such that light can be transmitted through the nasal tissue (including the skin) and can be visualized externally by the user. The user can then use the light to properly position the device in the desired location. Because the electrode 505 is not centered on the treatment element 503 of the device, a separate device having a mirror-image configuration may be required to treat the other nostril.
[0159] FIG. 20A depicts the treatment element 512 of a treatment device (e.g., device 30). The treatment element 512 of the device comprises two monopolar electrodes 514, 516 provided side-by-side on a convex surface of the treatment element. The cross section of the treatment element 512, shown in FIG. 20B, is configured to conform to the shape either nostril, depending on which side of the device (and accordingly, which of electrode 514 or 516) is placed in contact with the patient's nasal septum. Monopolar electrodes 514, 516 may allow the same treatment element 512 to be used for treatment in both nostrils, and each electrode may be activated separately depending on which side needs to be used. The treatment element 512 also comprises two lights 518, 520 (e.g., LEDs, lasers) configured to illuminate the treatment area for both nostrils. One or both of the lights 518, 520 can also be situated such that light can be transmitted through the nasal tissue (including the skin) and can be visualized externally by the user. The user can then use the light to properly position the device in the desired location.
[0160] FIG. 21A depicts a treatment element 522 of a treatment device (e.g., device 30). The tip 522 of the device comprises a monopolar electrode 524. The tip 522 comprises a symmetrical cross-section as shown in FIG. 21B. The tip 522 comprises a light 526 (e.g., LED) configured to illuminate the treatment area. The light 526 can also be situated such that light can be transmitted through the nasal tissue (including the skin) and can be visualized externally by the user. The symmetrical tip allows the user to treat either left or right nostril. The user can then use the light to properly position the device in the desired location.
[0161] FIGS. 22A-G depict a treatment device 530 similar to the embodiments of FIGS. 8D, 9A, and 9B. FIGS. 22A and 22F provide perspective views of the device 530. The device 530 comprises a treatment element 532 at its distal tip 534. The treatment element 532 comprises an electrode 535. The body of the treatment element 532, itself, may comprise an insulating material. The treatment element 532 may be provided on an enlarged distal tip 534 of an elongate shaft 536, and as in the embodiment illustrated, may have a convex shape configured to press against and create a concavity in the nasal cartilage (e.g., in the cartilage of the nasal septum). The distal tip 534 is located at the distal end of shaft 536. The shaft is attached at its proximal end to a handle 538. The handle 538 comprises an input control such as a power button 540 on its front side that may be used to activate and deactivate the electrode. The power button 540 may be positioned in a recess of the handle to allow for finger stability when activating and deactivating the electrode. In other embodiments, the input control is in the form of a switch or dial. Other configurations are also possible as described above.
[0162] The device 530 comprises a flexible wire or cable 542 electrically connected to an adaptor 544. The adaptor 544 can be used to connect the device 530 to a remote generator (not shown). The adaptor 544 may allow transmission of treatment energy between a remote generator and the device 530. The adaptor may also allow transmission of any sensor signals between the device 530 and a generator or control unit. The device 530 may either comprise an integrated generator or be connected to a remote generator. The treatment device 530 may be provided in a system or kit also including the remote generator. The system or kit (with or without the remote generator) may also include a grounding device and/or a cooling device as described above and further below. In some embodiments, the kit includes a positioning element (e.g., a "cottle" device) configured to help a user locate the optimal treatment area.
[0163] FIGS. 22B and 22C depict front and back views of the device. As shown in FIGS. 22B and 22C, the handle 538 of the device generally as a rounded elongate shape. Other shapes are also possible. For example the device 530 may have a square shaped cross section. In some embodiments, a circumference (or width or cross-sectional area) of the handle 538 may increase distally along the length of the handle 538.
[0164] FIGS. 22D and 22E depict side views of the device. As shown in FIGS. 22D and 22E, the handle 538 of the device 530 may comprise an indentation or recess around the middle of the handle 538. This may allow for enhanced grip and control when a user is holding the device. The indentation or recess may be near the input control or power button 540 to allow a user to easily activate and deactivate the device while holding it in a comfortable position.
[0165] In some embodiments, the shaft has a width or diameter of about 0.125 inches to about 0.25 inches. In some embodiments, the shaft is about 1.5 inches to about 4 inches long. In some embodiments, the shaft comprises a polymer such as polycarbonate or PEEK. In other embodiments, the shaft comprises stainless steel or other metals. The metals may be coated with an external and/or internal insulating coating (e.g., polyester, polyolefin, etc.). The handle may comprise the same material as the shaft, in some embodiments. In some embodiments, the shaft is rigid. This may allow a user of the device increased control over the deformation of nasal tissue. In some embodiments, the shaft comprises some amount of flexibility. This flexibility may allow a user adjust an angle of the distal tip by bending the distal end of the shaft.
[0166] FIG. 22G depicts a larger view of the distal tip 534 of the device 530. As shown best in FIG. 22G, the treatment element 532 comprises a generally elongate shape. The front of the treatment element 532 comprises a shallow, curved surface, providing a convex shape configured to deform the nasal tissue and create a concavity therein. In some embodiments, the front of the treatment element comprises a concave shape. The shape of the front surface of the treatment element may be selected to conform to the nasal tissue. The back of the treatment element 532 also comprises a shallow curved surface. As best seen in FIGS. 22D and 22E, the back surface varies in width along the length of the back surface of the treatment element 532. The back surface widens, moving distally along the tip until it is nearly in line with the proximal end of the electrode plate 535. The back surface then narrows towards the distal tip of the treatment element 532. This shape may maximize visualization of the area to be treated, while, at the same time, providing sufficient rigidity for treatment. Other shapes are also possible. For example, the treatment element may comprise a generally spherical or cylindrical shape. In some embodiments, the treatment element comprises an angular shape (e.g., triangular, conical) which may allow for close conformation to the tissue structures. The treatment element 532 comprises a monopolar electrode plate 535. The monopolar electrode plate 535 can be in the shape of a rectangle having a curved or convex tissue-facing surface. Other shapes are also possible (e.g., square, circular, ovular, etc.). The electrode 535 may protrude slightly from the treatment element 532. This may allow the electrode to itself provide a convex shape configured to create a concavity in tissue to be treated.
[0167] In some embodiments, the treatment element has a width or diameter of about 0.25 inches to about 0.45 inches. In some embodiments, the treatment element is about 0.4 inches to about 0.5 inches long. The treatment element can, in some embodiments, comprise a ceramic material (e.g., zirconium, alumina, silicon glass). Such ceramics may advantageously possess high dielectric strength and high temperature resistance. In some embodiments, the treatment element comprises polyimides or polyamides which may advantageously possess good dielectric strength and elasticity and be easy to manufacture. In some embodiments, the treatment element comprises thermoplastic polymers. Thermoplastic polymers may advantageously provide good dielectric strength and high elasticity. In some embodiments, the treatment element comprises thermoset polymers, which may advantageously provide good dielectric strength and good elasticity. In some embodiments, the treatment element comprises glass or ceramic infused polymers. Such polymers may advantageously provide good strength, good elasticity, and good dielectric strength.
[0168] In some embodiments, the electrode has a width of about 0.15 inches to about 0.25 inches. In some embodiments, the electrode is about 0.2 inches to about 0.5 inches long. In some embodiments, the treatment element comprises steel (e.g., stainless, carbon, alloy). Steel may advantageously provide high strength while being low in cost and minimally reactive. In some embodiments, the electrodes or energy delivery elements described herein comprise materials such as platinum, gold, or silver. Such materials may advantageously provide high conductivity while being minimally reactive. In some embodiments, the electrodes or energy delivery elements described herein comprise anodized aluminum. Anodized aluminum may advantageously be highly stiff and low in cost. In some embodiments, the electrodes or energy delivery elements described herein comprise titanium which may advantageously possess a high strength to weight ratio and be highly biocompatible. In some embodiments, the electrodes or energy delivery elements described herein comprise nickel titanium alloys. These alloys may advantageously provide high elasticity and be biocompatible. Other similar materials are also possible.
[0169] As shown in the embodiment of FIG. 22G, the treatment element 532 further comprises a pin-shaped structure comprising a thermocouple 533 within an insulating bushing extending through a middle portion of the plate 532. In some embodiments, different heat sensors (e.g., thermistors) may be used. In some embodiments, the thermocouple 533 is configured to measure a temperature of the surface or subsurface of tissue to be treated or tissue near the tissue to be treated. A pin-shape having a sharp point may allow the structure to penetrate the tissue to obtain temperature readings from below the surface. The thermocouple can also be configured to measure a temperature of the treatment element 532 itself. The temperature measurements taken by the thermocouple can be routed as feedback signals to a control unit (e.g., the control system 42 described with respect to FIG. 3) and the control unit can use the temperature measurements to adjust the intensity of energy being delivered through the electrode. In some embodiments, thermocouples or other sensing devices may be used to measure multiple tissue and device parameters. For example, multiple thermocouples or thermistors may be used to measure a temperature at different locations along the treatment element. In some embodiments, one of the sensors may be configured to penetrate deeper into the tissue to take a measurement of a more interior section of tissue. For example, a device may have multiple sensors configured to measure a temperature at the mucosa, the cartilage, and/or the treatment element itself. As described above, in some embodiments, the sensors described herein are configured to take a measurement of a different parameter. For example, tissue impedance can be measured. These measurements can be used to adjust the intensity and/or duration of energy being delivered through the treatment element. This type of feedback may be useful from both an efficacy and a safety perspective.
[0170] As shown in FIG. 22G, in some embodiments the thermocouple is within a pin shaped protrusion on the surface of the electrode 532. In other embodiments, the thermocouple can simply be on the surface of the electrode. In other embodiments, the thermocouple can protrude from the surface of the electrode in a rounded fashion. Rounded structures may be pressed into the tissue to obtain subsurface temperature readings. Other configurations and locations for the thermocouple are also possible. The use of thermocouples or temperature sensors may be applied not only to the embodiment of FIG. 22G, but also to any of the other embodiments described herein.
[0171] FIGS. 23A-G depict a treatment device 550 similar to the embodiments of FIGS. 8F and 18. FIGS. 23A and 23F are perspective views of the device 550 and show the device 550 comprising a treatment element 552 at the distal tip 556 of the device 550. The treatment element 552 may be provided on an enlarged distal tip 556 of an elongate shaft 558, and as in the embodiment illustrated, may have a convex shape configured to press against and create a concavity in the nasal cartilage (e.g., in cartilage of the nasal septum). The distal tip 556 is located at a distal end of shaft 558. The shaft is attached at its proximal end to a handle 560. The handle 560 comprises an input control, such as a power button 562, on its front side that may be used to activate and deactivate the electrode. The power button may be positioned in a recess of the handle to allow for finger stability when activating and deactivating the electrode. In other embodiments, the input control is in the form of a switch or dial. Other configurations are also possible as described above. The device 550 may either comprise a generator or be connected to a remote generator. The device 550 may comprise a flexible wire or cable 564 that connects to an adaptor 566 that is configured to be plugged into a remote generator (not shown). The adaptor 566 may allow transmission of treatment energy between a remote generator and the device 550. The adaptor 566 may also allow transmission of any sensor signals between the device 550 and a generator or control unit. The treatment device 550 may be provided in a system or kit also including the remote generator. The system or kit (with or without the remote generator) may also include a grounding device and/or a cooling device as described above and further below. In some embodiments, the kit includes a positioning element (e.g., a "cottle" device) configured to help a user locate the optimal treatment area.
[0172] In some embodiments, the shaft has a width or diameter or about 0.235 inches to about 0.25 inches. In some embodiments, the shaft is about 1.5 inches to about 4 inches long. In some embodiments, the shaft and/or handle comprises a polymer such as polycarbonate or PEEK. In other embodiments, the shaft comprises stainless steel or other metals. The metals may be coated with an external and/or internal insulating coating (e.g., polyester, polyolefin, etc.). The handle may comprise the same material as the shaft, in some embodiments. In some embodiments, the shaft is rigid. This may allow a user of the device increased control over the deformation of nasal tissue. In some embodiments, the shaft comprises some amount of flexibility. This flexibility may allow a user adjust an angle of the distal tip by bending the distal end of the shaft.
[0173] FIGS. 23B and 23C depict side views of the device. As shown in FIGS. 23B and 23C, the handle 560 of the device 550 may comprise an indentation or recess around the middle of the handle 560. This may allow for enhanced grip and control when a user is holding the device. The indentation or recess may be near the input control or power button 562 to allow a user to easily activate and deactivate the device while holding it in a comfortable position.
[0174] FIGS. 23D and 23E depict front and back views of the device. As shown in FIGS. 23D and 23E, the handle 560 of the device generally comprises a rounded elongate shape. Other shapes are also possible. For example the device 550 may have a square shaped cross section. In some embodiments, a circumference (or width or cross-sectional area) of the handle 560 may increase distally along the length of the handle 560.
[0175] FIG. 23G depicts a larger view of the distal tip 556 of the device 550. As shown best in FIG. 23G, the treatment element 552 comprises a generally elongate shape. The front of the treatment element 552 comprises a shallow curved surface, providing a convex shape configured to deform the nasal tissue and create a concavity therein. In some embodiments, the front of the treatment element comprises a concave shape. The shape of the front surface of the treatment element may be selected to conform to the nasal tissue. The back surface of the treatment element 552 comprises a shallow curved surface along most of its length. As best seen in FIGS. 23B and 23C, the back surface narrows distally along the length of the element 552 from approximately the distal end of the needle electrodes to the distal tip of the treatment element 552. This shape may maximize visualization of the area to be treated, while, at the same time, providing sufficient rigidity for treatment. Other shapes are also possible. For example, the treatment element may comprise a generally spherical or cylindrical shape. In some embodiments, the treatment element comprises an angular shape (e.g., triangular, conical) which may allow for close conformation to the tissue structures. The treatment element 552 comprises a monopolar or bipolar needle array comprising multiple needles 554. In some embodiments, the needles 554 are energized in between select needles to deliver bipolar energy. In other embodiments, the energy is delivered between the needles 554 and a remote grounding pad (not shown). In some embodiments, the electrode needle pairs are arranged horizontally across the treatment element 552. In some embodiments, the electrode needle pairs are arranged vertically across the treatment element 552, or along the direction of the shaft 558 and handle 560. Other configurations are also possible. For example, the needle pairs may be arranged diagonally across the treatment element 552. The treatment element 552 may be placed either internally, with the needle pairs 554 positioned transmucosally or the treatment element 552 may be placed externally with the needle pairs 554 positioned transdermally. The distal tip 556 of the device 550 may also function as a mold or molding element. In a monopolar embodiment, the energy may be selectively delivered between certain sets of needles, all needles, or even individual needles to optimize the treatment effect.
[0176] The treatment element 552 of the device 550 further comprises a pin-shaped structure comprising a thermocouple 555 within an insulating bushing extending through a middle portion of the front surface of the treatment element 552. In some embodiments, different heat sensors (e.g., thermistors) may be used. As described above, in some embodiments, the thermocouple 555 is configured to measure a temperature of the surface or subsurface of tissue to be treated or tissue near the tissue to be treated. A pin-shape having a sharp point may allow the structure to penetrate the tissue to obtain temperature readings from below the surface. The thermocouple can also be configured to measure a temperature of the treatment element 552 itself. The temperature measurements taken by the thermocouple can be routed as feedback signals to a control unit (e.g., the control system 42 described with respect to FIG. 3) and the control unit can use the temperature measurements to adjust the intensity of energy being delivered through the electrode. In some embodiments, thermocouples or other sensing devices may be used to measure multiple tissue and device parameters. For example, multiple thermocouples or thermistors may be used to measure a temperature at different locations along the treatment element. In some embodiments, one of the sensors may be configured to penetrate deeper into the tissue to take a measurement of a more interior section of tissue. For example, a device may have multiple sensors configured to measure a temperature at the mucosa, the cartilage, and/or the treatment element itself. As described above, in some embodiments, the sensors described herein are configured to take a measurement of a different parameter. For example, tissue impedance can be measured. These measurements can be used to adjust the intensity and/or duration of energy being delivered through the treatment element. This type of feedback may be useful from both an efficacy and a safety perspective.
[0177] In some embodiments, the treatment element has a width or diameter of about 0.25 inches to about 0.45 inches. In some embodiments, the treatment element is about 0.4 inches to about 0.5 inches long. The treatment element can, in some embodiments, comprise a ceramic material (e.g., zirconium, alumina, silicon glass). Such ceramics may advantageously possess high dielectric strength and high temperature resistance. In some embodiments, the treatment element comprises polyimides or polyamides which may advantageously possess good dielectric strength and elasticity and be easy to manufacture. In some embodiments, the treatment element comprises thermoplastic polymers. Thermoplastic polymers may advantageously provide good dielectric strength and high elasticity. In some embodiments, the treatment element comprises thermoset polymers, which may advantageously provide good dielectric strength and good elasticity. In some embodiments, the treatment element comprises glass or ceramic infused polymers. Such polymers may advantageously provide good strength, good elasticity, and good dielectric strength.
[0178] In some embodiments, the electrodes have a width or diameter of about 0.15 inches to about 0.25 inches. In some embodiments, the electrode is about 0.2 inches to about 0.5 inches long. In some embodiments, the treatment element comprises steel (e.g., stainless, carbon, alloy). Steel may advantageously provide high strength while being low in cost and minimally reactive. In some embodiments, the electrodes or energy delivery elements described herein comprise materials such as platinum, gold, or silver. Such materials may advantageously provide high conductivity while being minimally reactive. In some embodiments, the electrodes or energy delivery elements described herein comprise anodized aluminum. Anodized aluminum may advantageously be highly stiff and low in cost. In some embodiments, the electrodes or energy delivery elements described herein comprise titanium which may advantageously possess a high strength to weight ratio and be highly biocompatible. In some embodiments, the electrodes or energy delivery elements described herein comprise nickel titanium alloys. These alloys may advantageously provide high elasticity and be biocompatible. Other similar materials are also possible.
[0179] Energy applied to the tissue to be treated using any combination of the embodiments described in this application may be controlled by a variety of methods. In some embodiments, temperature or a combination of temperature and time may be used to control the amount of energy applied to the tissue. Tissue is particularly sensitive to temperature; so providing just enough energy to reach the target tissue may provide a specific tissue effect while minimizing damage resulting from energy causing excessive temperature readings. For example, a maximum temperature may be used to control the energy. In some embodiments, time at a specified maximum temperature may be used to control the energy. In some embodiments, thermocouples, such as those described above, are provided to monitor the temperature at the electrode and provide feedback to a control unit (e.g., control system 42 described with respect to FIG. 3). In some embodiments, tissue impedance may be used to control the energy. Impedance of tissue changes as it is affected by energy delivery. By determining the impedance reached when a tissue effect has been achieved, a maximum tissue impedance can be used to control energy applied.
[0180] In the embodiments described herein, energy may be produced and controlled via a generator that is either integrated into the electrode handpiece or as part of a separate assembly that delivers energy or control signals to the handpiece via a cable or other connection. In some embodiments, the generator is an RF energy source configured to communicate RF energy to the treatment element. For example, the generator may comprise a 460 KHz sinusoid wave generator. In some embodiments, the generator is configured to run between about 1 and 100 watts. In some embodiments, the generator is configured to run between about 5 and about 75 watts. In some embodiments, the generator is configured to run between about 10 and 50 watts.
[0181] In some embodiments, the energy delivery element comprises a monopolar electrode (e.g., electrode 535 of FIG. 22G). Monopolar electrodes are used in conjunction with a grounding pad. The grounding pad may be a rectangular, flat, metal pad. Other shapes are also possible. The grounding pad may comprise wires configured to electrically connect the grounding pad to an energy source (e.g., an RF energy source).
[0182] In some embodiments, the energy delivery element such as the electrodes described above can be flat. Other shapes are also possible. For example, the energy delivery element can be curved or comprise a complex shape. For example, a curved shape may be used to place pressure or deform the tissue to be treated. The energy delivery element may comprise needles or microneedles. The needles or microneedles may be partially or fully insulated. Such needles or microneedles may be configured to deliver energy or heat to specific tissues while avoiding tissues that should not receive energy delivery.
[0183] In some embodiments, the electrodes or energy delivery elements described herein comprise steel (e.g., stainless, carbon, alloy). Steel may advantageously provide high strength while being low in cost and minimally reactive. In some embodiments, the electrodes or energy delivery elements described herein comprise materials such as platinum, gold, or silver. Such materials may advantageously provide high conductivity while being minimally reactive. In some embodiments, the electrodes or energy delivery elements described herein comprise anodized aluminum. Anodized aluminum may advantageously be highly stiff and low in cost. In some embodiments, the electrodes or energy delivery elements described herein comprise titanium which may advantageously possess a high strength to weight ratio and be highly biocompatible. In some embodiments, the electrodes or energy delivery elements described herein comprise nickel titanium alloys. These alloys may advantageously provide high elasticity and be biocompatible. Other similar materials are also possible.
[0184] In some embodiments, the treatment elements (e.g., non-electrode portion of treatment element) of the devices described herein, including but not limited to FIGS. 8A-J, 9A-B, 10A-B, 11A-B, 12A-B, 13A-E, 14A-B, 15A-C, 16, 17A-B, 18, 19A-B, 20A-B, 21A-B, 22A-G, 23A-G, 25A-B, 26, 27, 28A-E, and 29, comprise an insulating material such as a ceramic material (e.g., zirconium, alumina, silicon glass). In some embodiments, the treatment elements comprise an insulating material interposed between multiple electrodes or electrode section. These insulating sections may provide an inert portion of the treatment element that does not delivery energy to the tissue. Such ceramics may advantageously possess high dielectric strength and high temperature resistance. In some embodiments, the insulators described herein comprise polyimides or polyamides which may advantageously possess good dielectric strength and elasticity and be easy to manufacture. In some embodiments, the insulators described herein comprise thermoplastic polymers. Thermoplastic polymers may advantageously provide good dielectric strength and high elasticity. In some embodiments, the insulators described herein comprise thermoset polymers, which may advantageously provide good dielectric strength and good elasticity. In some embodiments, the insulators described herein comprise glass or ceramic infused polymers. Such polymers may advantageously provide good strength, good elasticity, and good dielectric strength.
[0185] In some embodiments, the handle and/or shaft of the devices comprise the same materials as those described with respect to the insulators. In some embodiments, the handle and/or shaft of the device comprises a metal, such as stainless steel. In other embodiments, the handle and/or shaft of the device comprises a polymer, such as polycarbonate. Other metals and polymers are also contemplated.
[0186] In some embodiments, the device may be used in conjunction with a positioning element that can be used to aid in positioning of the device. The positioning element may be integrated into the device itself or can be separate. The positioning element may be used to determine the optimal placement of the device to achieve maximal increase in efficacy. In some embodiments, a positioning element is configured to be inserted and manipulated within the nose until the patient reports a desired improvement in breathing. The treatment device may then be used to treat while the positioning element is holding the nose in the desired configuration. In some embodiments, molds described herein may be used for the same purpose.
[0187] In some embodiments, a positioning element comprises a shaft comprising measurement marks indicating depth. For example, a physician may insert this element into the nose to manipulate the tissue to find the depth of treatment at which the patient reports the best breathing experience. The positioning element may comprise marks around the base of the shaft indicating which point of rotation of the device within the nostril provides the best breathing experience. The positioning element may also comprise marks indicating angle of insertion. The physician may then use the measurement marks to guide insertion of the treatment element to the same spot.
[0188] It will be appreciated that any combination of electrode configurations, molds, handles, connection between handles, and the like may be used to treat the nasal septum.
Examples of Treatment Devices Including Cooling Systems
[0189] Embodiments of devices configured to heat specific tissue while maintaining lower temperatures in other adjacent tissue are provided. These devices may be incorporated into any of the treatment apparatuses and methods described herein. The nasal septum is an example of a tissue complex that includes adjacent tissues that may benefit from being maintained at different temperatures. Other examples include the skin, which comprises the epidermis, dermis, and subcutaneous fat, the tonsils, which comprise mucosa, glandular tissue, and vessels. Treatment of other tissue complexes is also possible. For example, in some embodiments, the nasal septum may be heated while maintaining a lower temperature in the mucosal lining of the nose and/or skin. In other embodiments, the cartilage may be heated, while maintaining lower temperatures in the mucosa. Limiting unwanted heating of non-target tissues may allow trauma and pain to be reduced, may reduce scarring, may preserve tissue function, and may also decrease healing time. Combinations of heat transfer and/or heat isolation may allow directed treatment of specific tissue such as cartilage, while excluding another tissue, such as mucosa, without surgical dissection.
[0190] Generally, when using a device 570 with an electrode 572 (e.g., monopolar RF electrode) to heat nasal cartilage, the electrode 572 must be in contact with the mucosa. FIG. 24A shows a cross-section of tissue at the nasal septum. The cross-section shows that the nasal cartilage 704 sits in between layers of mucosa 702. When the electrode 572 is activated, both the mucosa and the cartilage are heated by the current flowing from the electrode to the return (e.g., ground pad), as shown in FIG. 24B. The tissue closest to the electrode 572 receives the highest current density, and thus, the highest heat. A surface cooling mechanism may allow the temperature of the electrode surface to be reduced. Such a cooling mechanism may maintain a lower temperature at the mucosa even though current flow will continue to heat the cartilage.
[0191] FIG. 25A depicts a device 580 configured to treat the nasal septum cartilage using an electrode while maintaining a reduced temperature at the mucosa. The device comprises a treatment element 582 comprising an electrode 584 at the distal tip of the device 580. The treatment element 582 is attached to a distal end of a shaft 586, which is attached to the distal end of a handle 588. Input and output coolant lines 590, 592 are attached to a pump and reservoir 594 and extend into the handle 588, through the distal end of the treatment element 582 to the electrode 582 and return back through the shaft 586 and handle 588 to the pump and reservoir 594. The coolant may be remotely cooled in the reservoir and may comprise a fluid or gas. The coolant flowing through the electrode 582 may allow the treatment element 582 to be maintained at a reduced temperature while still allowing current flow to heat the cartilage. Examples of coolant include air, saline, water, refrigerants, and the like. Water may advantageously provide moderate heat capacity and be non-reactive. Refrigerants may advantageously be able to transfer significant amounts of heat through phase change. The coolant may flow through internal or external cavities of the electrode or wand tip. For example, FIG. 25B depicts an embodiment of a device 600 comprising a treatment element 602 with an electrode 604 at the distal tip of the device 600. The treatment element 602 is attached to the distal end of a shaft 606 which is attached to the distal end of a handle 608. The handle may be attached to a cable comprises a lumen or channel 611 through which gas or fluid may flow. The lumen 611 may diverge, near the treatment element 602, into separate external channels flowing over the electrode 604. The lumen 611 and channels 610 or cavities may be attached to a fan or fluid pump 612. In some embodiments, the fan or fluid pump may remotely cool the gas or fluid.
[0192] FIG. 26 depicts another embodiment of a device 620 configured to treat the nasal septum using an electrode 624 while maintaining a reduced temperature at the mucosa and/or skin. The device comprises a treatment element 622 comprising an electrode 624 at its distal end. The treatment element 622 is connected to the distal end of a shaft 626 which is connected to the distal end of a handle 628. The device 620 comprises a heat pipe 630 attached to the electrode 624 or treatment element 622. The heat pipe 630 is configured to transfer heat to a remote heat sink 632. As shown in FIG. 26, the heat sink 632 may be placed in the handle of the device. In some embodiments, the heat sink may be placed remotely. The heat pipe 630 may comprise a sealed tube (e.g., a copper tube) filled with a material that evaporates at a given temperature. When one end of the heat pipe 630 is heated, the fluid may evaporate and flow to the opposite end where it may condense and subsequently transfer heat to the heat sink 632. Using a material such as copper for the heat pipe 630 and/or heat sink 632 may advantageously provide high heat and electrical conductivity.
[0193] FIG. 27 depicts another embodiment of a device 640 configured to treat the nasal septum using a bipolar electrode pair while maintaining a reduced temperature at the mucosa or the skin. The device 640 comprises a first treatment element 642 comprising a first electrode 644 of a bipolar electrode pair at the distal end of a shaft 646. The treatment element 642 comprises a thermocouple pin 650 like that described with respect to FIG. 22G. The shaft 646 is connected to the distal end of a handle 648. The handle 648 is connected to another handle 652 comprising a shaft 654 with a treatment element 656 at its distal tip. The treatment element 656 comprises a second electrode 657 of the bipolar electrode pair. The first and second treatment elements 642, 656 can be placed on either side of nasal tissue. For example, the first treatment element 642 may be in contact with the mucosa and the second treatment element 656 may be in contact with the skin. Similar to the device depicted in FIG. 26, the device of FIG. 27 comprises a heat pipe within both shafts 654, 646. Thus heat from the tissue is transferred from the treatment elements 642, 656 and is transported down the shafts 654, 646 into an integrated or a remote heat sink (not shown). This heat transfer may keep the skin and/or the mucosa relatively cool while still delivering sufficient treatment energy to the cartilage. The connection 658 and spring 647 between the two handles 648, 652 is configured to bias the two shafts 646, 654 and treatment elements 642, 656 towards a collapsed state. Squeezing the handles 648, 652 may separate the two shafts 646, 654 and treatment elements 642, 656. Thus, the handles 648, 652 can be squeezed to properly position the device 640 at the nasal tissue to be treated. Releasing the handles 648, 652 can cause the treatment element 642 and the cooling element 656 to contact the tissue. In some embodiments, the device 640 may only comprise one heat pipe. In some embodiments, the device 640 may comprise a treatment element with a monopolar electrode on one shaft and a molding element on the other shaft. Multiple configurations are contemplated. For example, the device may comprise one heat pipe and a bipolar electrode pair. For another example, the device may comprise one heat pipe and a monopolar electrode. For another example, the device may comprise two heat pipes and a monopolar electrode. Other device configurations are also possible.
[0194] The embodiments described with respect to FIGS. 25A-27 employ specific differential cooling mechanisms to maintain different and particular temperatures in adjacent tissues. FIGS. 28A-28E depicts various examples of more general mechanisms configured to maintain different temperatures in adjacent tissues. FIGS. 28A-28E depict examples of differential cooling mechanisms as applied to a cross-section of tissue at the nasal septum, like that shown in FIG. 24A. The nasal septum may include a middle cartilage layer 704 with mucosal tissue 702 on either side.
[0195] As shown in FIG. 28A, in some embodiments, the differential cooling mechanism comprises two elements: a first element 708 and a second element 710. The two elements are on either side of the thickness of the nasal tissue. In one embodiment, the mechanism is configured to maintain normal temperatures in the cartilage 704 while cooling mucosal tissue 702 on a first side and mucosal tissue 702 on a second side. In such an embodiment, the first and second elements 708, 710 comprise a cooling apparatus such as those described above (e.g., heatsink, coolant lines, etc.). In some embodiments, the mucosal tissue 702 on the first and second sides is heated while normal temperatures are maintained in the cartilaginous middle layer 704. The cartilage 704 may be somewhat warmed, in such embodiments, but may be cooler than mucosal tissue 702 on the first and second sides. In such embodiments, the first and second elements 708, 710 comprise a heating apparatus, such as radio frequency electrodes or resistive heating elements. In some embodiments, mucosal tissue 702 on the first side is heated, the mucosal tissue 702 on second side is cooled, and normal temperatures are maintained in the cartilage 704. In such embodiments, the first element 708 comprises a heating apparatus and the second element 710 comprises a cooling apparatus. For example, the device 580, described with respect to FIG. 27, may use such a mechanism. In some embodiments, the second side is heated, the first side is cooled, and normal temperatures are maintained in the cartilage 704. In such embodiments, the first element 708 comprises a cooling apparatus and the second element 710 comprises a heating apparatus. Again, the device 580, described with respect to FIG. 27, is an example of a device that may use such a mechanism.
[0196] FIG. 28B shows an example of one of the embodiments described with respect to FIG. 28A. The first element 730 is on mucosal tissue 702 on a first side of the nasal septum. The second element 732 is an energy delivery element and is positioned on mucosal tissue 702 on a second side of the nasal septum. The first element 730 comprises a cooling apparatus and the second element 732 comprises an energy delivery element (e.g., an RF electrode). The a first side is cooled while a second side and cartilaginous areas 704 are heated. In other embodiments, the first element 730 can be positioned on mucosal tissue 702 on the second side and the second element 732 can be positioned on mucosal tissue 702 on the first side. In such embodiments, the mucosal tissue 702 on the second side is cooled while mucosal tissue 702 on the first side and the cartilage 704 are heated.
[0197] As shown in FIG. 28C, in some embodiments, the differential cooling mechanism comprises a first element 720 and a second element 722. Both elements 720, 722 are on mucosal tissue 702 on the first side of the nasal septum. In some embodiments, the mucosal tissue 702 on the first side is cooled while higher temperatures are maintained in the middle cartilaginous layer 704. In such embodiments, the first element 720 comprises a cooling apparatus, and the second element 722 comprises an energy delivery apparatus (e.g., a monopolar radiofrequency electrode). In some embodiments, the first element 720 is sufficiently efficient to maintain cool temperatures at mucosal tissue 702 on the first side despite the energy provided by the second element 722. In other embodiments, the first and second elements 720, 722 are both positioned on mucosal tissue 702 on the second side of the nasal septum. In such embodiments, mucosal tissue 702 on the second side is cooled while higher temperatures are maintained in the middle cartilaginous layer.
[0198] As shown in FIG. 28D, in some embodiments, the differential cooling mechanism comprises a first surface element 740 and a second surface element 742 on either side of the nasal septum. A third subsurface element 744 is engaged through the mucosal tissue 702 on the first side and into the cartilage area 704. In some embodiments, the mucosal tissue 702 on the first side and the second side are cooled while the middle cartilaginous layer 704 is heated. In such embodiments, the first and second elements 740, 742 comprise cooling apparatus while the third element 744 comprises a heating element (e.g., RF monopolar electrode, RF bipolar needles, etc.). In other embodiments, the third subsurface element 744 may be engaged through the mucosal tissue 702 on the second side and into the cartilage area 704.
[0199] As shown in FIG. 28E, in some embodiments, the differential cooling mechanism comprises a first surface element 750 and a second surface element 752 on either side of the nasal septum. The differential cooling mechanism further comprises a third surface element 754 and a fourth surface element 756 on either side of the nasal septum. In some embodiments, the cartilage layer 704 is heated while the mucosal tissue 702 on the first and second sides are cooled. In such embodiments, the first and second elements 750, 752 comprise cooling apparatus and the third and fourth elements 754, 756 comprise energy delivery apparatuses (e.g., bipolar plate electrodes). In some embodiments, the cartilage 704 and mucosal tissue 702 on the first side are heated while the mucosal tissue on the second side is cooled. In such embodiments, the first element 750 comprises a heating apparatus; the second element 752 comprises a cooling apparatus; and the third and fourth elements 754, 756 comprise energy delivery apparatuses. It will be appreciated that different differential temperature effects can be achieved by reconfiguring and adding or subtracting to the described configuration of elements.
[0200] Cooling occurring before, during, or after treatment may effect reduced temperature of the skin and/or mucosa. In some embodiments, attaching passive fins or other structures to the electrode or wand tip may allow for heat dissipation to the surrounding air. In some embodiments, the device may be configured to spray a cool material such as liquid nitrogen before, during, or after treatment. Using a material such as copper for the passive fins or other structure may advantageously provide high heat and electrical conductivity. In some embodiments, using metals with a high heat capacity in the device (e.g., in the energy delivery element, the reshaping element, or both) may advantageously provide the ability to resist temperature change during energy delivery. In some embodiments, pre-cooling the electrode (e.g., by refrigeration, submersion, spraying with a cool substance like liquid nitrogen, etc.) may maintain a reduced temperature at the mucosa. Any combination of the cooling methods described herein may be used in conjunction with any of the energy delivery methods described herein (e.g., bipolar RF electrodes, arrays needles, plates, etc.). For example, FIG. 29 depicts an embodiment of a device 800 comprising a treatment element 802 comprising electrode needles 804 at its distal tip. The device 800 may be used in conjunction with a separate cooling device 810 which may comprise channels 811 or cavities to circulate air or fluid. The independent cooling device 810 may, in other embodiments, employ a different cooling mechanism.
[0201] In embodiments using laser energy to heat cartilage, it is possible to use a combination of two or more lasers whose beams converge at a location within the target tissue. This convergence may cause more heat at that junction as compared to locations where only a single beam is acting. The junction may be controlled manually or via computer control. Specific treatment may be provided.
[0202] In some embodiments, insulating material may be used to protect non-target tissue during energy delivery. For example, an electrode needle may be preferentially insulated on a portion of the needle that is in contact with non-target tissue. For another example, flat electrode blades may be insulated on a portion of the blade that is in contact with non-target tissue. Other configurations for heat isolation are also possible.
[0203] Any of the cooling mechanisms or combinations of the cooling mechanisms described herein may be used in conjunction with any of the devices or combinations of devices described herein, or the like.
Examples of Methods of Treatment
[0204] Embodiments of methods for treating nasal airways are now described. Such methods may treat nasal airways by decreasing the airflow resistance or the perceived airflow resistance at the site of a nasal septum. Such treatments may also address related conditions, such as snoring. The methods may include using a device similar to those devices described above--including but not limited to FIGS. 8A, 9B, 18, 22A-G, and 23A-G--to provide tissue reshaping/molding and to impart energy to tissue near the nasal septum.
[0205] In one embodiment, a method of decreasing airflow resistance in a nose comprises the steps of inserting an energy-delivery or cryo-therapy device into a nasal passageway, and applying energy or cryo-therapy to a targeted region or tissue of the nasal passageway. For example, in some embodiments, the method may include delivering energy or cryo-therapy to a section of nasal septum cartilage. Energy and cryo-therapy can be applied to cartilage in the area of the upper lateral cartilage, or in the area of intersection of the upper and lower lateral cartilage. In alternative embodiments, the method may deliver energy to the epithelium, or underlying soft tissue adjacent to the nasal septum, the upper lateral cartilage and/or the intersection of the ULC and the LLC.
[0206] In another embodiment, a method comprises heating a section of nasal septum cartilage to be reshaped, applying a mechanical reshaping force, and then removing the heat. In some embodiments, the step of applying a mechanical reshaping force may occur before, during or after the step of applying heat.
[0207] In some embodiments, the method may further include the step of inserting a reshaping device into the nasal passageway after applying an energy or cryo-therapy treatment. In such embodiments, a reshaping device such as an external adhesive nasal strip (such as those described for example in U.S. Pat. No. 5,533,499 to Johnson or U.S. Pat. No. 7,114,495 to Lockwood, the entirety of each of which is hereby incorporated by reference) may be applied to the exterior of the nose after the treatment in order to allow for long-term reshaping of nasal septum structures as the treated tissues heal over time. In alternative embodiments, a temporary internal reshaping device (such as those taught in U.S. Pat. No. 7,055,523 to Brown or U.S. Pat. No. 6,978,781 to Jordan, the entirety of each of which is hereby incorporated by reference) may be placed in the nasal passageway after treatment in order to allow for long-term reshaping of nasal septum structures as the treated tissues heal over time. In some embodiments, the dilating nasal strips can be worn externally until healing occurs.
[0208] In alternative embodiments, internal and/or external reshaping devices may be used to reshape a nasal septum section prior to the step of applying energy or cryo-therapy treatments to targeted sections of the epithelial, soft tissue, mucosa, submucosa and/or cartilage of the nose. In some embodiments, the energy or cryo-therapy treatment may be configured to change the properties of treated tissues such that the tissues will retain the modified shape within a very short time of the treatment. In alternative embodiments, the treatment may be configured to reshape nasal septum structures over time as the tissue heals.
[0209] In some embodiments, a portion of the nose, the nasal septum and/or the soft tissue and cartilage of the nasal valve may be reshaped using a reshaping device and then fixed into place. In some embodiments, such fixation may be achieved by injecting a substance such as a glue, adhesive, bulking agent or a curable polymer into a region of the nasal tissue adjacent the target area. Alternatively, such a fixation substance may be applied to an external or internal surface of the nose.
[0210] In some embodiments, an injectable polymer may be injected into a region of the nose, either below the skin on the exterior of the nose, or under the epithelium of the interior of the nose. In some embodiments, an injectable polymer may include a two-part mixture configured to polymerize and solidify through a purely chemical process. One example of a suitable injectable two-part polymer material is described in U.S. Patent Application Publication 2010/0144996, the entirety of which is hereby incorporated by reference. In other embodiments, an injectable polymer may require application of energy in order to cure, polymerize or solidify. A reshaping device may be used to modify the shape of the nasal septum before or after or during injection of a polymer. In embodiments employing an energy-curable polymer, a reshaping device may include energy-delivery elements configured to deliver energy suitable for curing the polymer to a desired degree of rigidity.
[0211] In another embodiment, the soft tissue of the upper lip under the nares may be debulked or reshaped to reduce airflow resistance. In some embodiments, such reshaping of the upper lip soft tissue may be achieved by applying energy and/or cryotherapy from an external and/or internal treatment element. In some embodiments, the tissue of the upper lip under the nares may be compressed by an internal or external device prior to or during application of the energy or cryo-therapy. For example, devices such as those shown in FIGS. 5A and 5B may be adapted for this purpose by providing tissue-engaging clamp tips shaped for the purpose.
[0212] In another embodiment, the muscles of the nose and/or face are stimulated to affect (e.g., dilate) the nasal septum area prior to or during application of other treatments such as energy/cryo application or fixation treatments. In such embodiments, the muscles to be treated may include the nasal dilator muscles (nasalis) the levator labii, or other facial muscles affecting the internal and/or external nasal valves. In some embodiments, the targeted muscles may be stimulated by applying an electric current to contract the muscles, mentally by the patient, or manually by the clinician.
[0213] In some embodiments, the muscles of the nose and/or face may also be selectively deactivated through chemical, ablative, stimulatory, or mechanical means. For example, muscles may be deactivated by temporarily or permanently paralyzing or otherwise preventing the normal contraction of the muscle tissue. Chemical compounds for deactivating muscle tissues may include botulinum toxin (aka "Botox"), or others. Ablative mechanisms for deactivating muscle tissue may include RF ablation, laser ablation or others. Mechanical means of deactivating muscle tissues may include one or more surgical incisions to sever targeted muscle tissue.
[0214] In another embodiment, the tissue of the nasal septum may be reshaped by applying energy to the internal and external walls of the nose using a clamp like device as illustrated for example in FIGS. 5A and 5B. One arm of the clamp may provide inward pressure to the external, skin side tissue covering nasal tissue and the other side of the clamp may provide outward pressure to the mucosal tissue on nasal tissue (e.g., the lateral wall of the nasal airway above the ULC and LLC or both).
[0215] In some embodiments, energy may be applied to the skin of the nose to effect a shrinkage of the skin, epidermis, dermis, subdermal, subcutaneous, tendon, ligament, muscle, cartilage and/or cartilage tissue. The tissue shrinkage is intended to result in a change of forces acting on the tissues of the nasal septum.
[0216] In another embodiment, the nasal septum tissue may be damaged or stimulated by energy application, incisions, injections, compression, or other mechanical or chemical actions. Following such damage, a device may be used on the tissue to mold or shape the tissue of the septum during healing. In some embodiments, such a reshaping device may be temporarily placed or implanted inside or outside the patient's nose to hold a desired shape while the patient's healing process progresses.
[0217] In another embodiment, the aesthetic appearance of the nose may be adjusted by varying the device design and/or treatment procedure. The predicted post-procedure appearance of the nose may be shown to the patient through manipulating the nasal tissue to give a post procedure appearance approximation. The patient may then decide if the predicted post procedure appearance of the face and nose is acceptable or if the physician needs to change parameters of the device or procedure to produce an appearance more acceptable to the patient.
[0218] In another embodiment, reduction of the negative pressure in the nasal airway can be effected to reduce collapse of the structures of the nasal airway on inspiration without changing a shape of the nasal septum. For example, this may be accomplished by creating an air passage that allows flow of air directly into the site of negative pressure. One example of this is creating a hole through the lateral wall of the nose allowing airflow from the exterior of the nose through the nasal wall and into the nasal airway.
[0219] In another embodiment, energy, mechanical or chemical therapy may be applied to the tissue of the nasal airway with the express purpose of changing the properties of the extracellular matrix components to achieve a desired effect without damaging the chondrocytes or other cells of the nasal airway tissue.
[0220] In some embodiments, devices (e.g., devices like those described with respect to FIGS. 9A-21B) may be used to provide tissue reshaping/molding and to impart energy to the nasal septum. The electrode may be placed in contact with the target nasal septum tissue. The electrodes and molds may be moved to shape the tissue as necessary to achieve improvement in nasal airway. The electrodes may be activated while the tissue is deformed in the new shape to treat the tissue. The electrode may then be deactivated and the device may be removed from the nasal septum area.
[0221] FIGS. 30A-D show an embodiment of a method for modifying a nasal septum, viewed from the nares. FIG. 30A illustrates a view of a nose having a deviated nasal septum 2. FIG. 30B illustrates a substance delivery device 806 (shown as a syringe with needle, but alternatively a swab or any other device for applying a substance to the tissue) applying a solution 808 to tissue near the cartilage of the nasal septum 2. FIG. 30C illustrates an energy delivery device 800 with a treatment element applying energy to the nasal septum. FIG. 30D illustrates a view of the nose after treatment, having a corrected nasal septum 2.
[0222] The method may include identifying a patient who desires to improve the airflow through their nasal passageways and/or who may benefit from a modification to the nasal septum (e.g., the patient has a deviated septum as shown in FIG. 30A). The patient may be positioned either in an upright position (e.g., seated or standing) or lying down. Local anesthesia may be applied to an area near or surrounding the tissue to be treated. General anesthesia may also be used.
[0223] The physician (or other medical professional administering the treatment) may apply a chemical enzyme or other solution at the site of the deviation to be corrected (see, e.g., FIG. 30B). In one embodiment, for example, the physician injects the solution through mucosal tissue to the space between the nasal mucosa and the nasal septal cartilage. The solution may be contained in the general area of the septal deviation, using a shield, and may be left in the patient's septum for a designated amount of time, to allow the solution to effectively condition the septal cartilage.
[0224] In some embodiments, the solution is between about 0.5 ml to about 2.5 ml of collagenase at a concentration ranging from about 1 mg/ml to about 10 mg/ml. In an example, a solution can include collagenase at a concentration of approximately 2 mg/ml. In some embodiments, the solution is between about 0.5 ml to about 2.5 ml of trypsin at a concentration of about 10 .mu.g/ml to about 100 .mu.g/ml. In an example, a solution can include trypsin at a concentration of approximately 50 .mu.g/ml. The designated amount of time may be in the range of about 15 minutes to about 90 minutes, in various embodiments. The designated amount of time can vary based in part on the solution being used. In an example, the designated amount of time can be approximately 40 minutes where the solution includes trypsin at a concentration of approximately 50 .mu.g/ml. In an example, a designated amount of time can be approximately 20 minutes for a solution that includes collagenase at a concentration of approximately 2 mg/ml. The application of the solution may result in a narrow band of degraded cartilage ranging from about 100 .mu.m to about 1 mm from the cartilage surface in the area of application. Prior to the application of the solution, the solution may be prepared at a particular temperature (e.g., between about 20.degree. C. and about 80.degree. C., or at about 20.degree. C., 37.degree. C., 60.degree. C., or 80.degree. C.).
[0225] At the conclusion of the conditioning period, a device (e.g., a monopolar, bipolar, single electrode, or multi-electrode RF energy device) may be introduced to the area to be corrected, and energy (e.g., RF energy) may be applied to the site (see, e.g., FIG. 30C). The temperature and duration of the energy application may be selected to denature and/or deactivate the solution and/or reshape the septal cartilage to correct the septal deviation (see, e.g., FIG. 30D). In addition to energy, mechanical force can also be applied to aid in reshaping the cartilage. Reshaping the cartilage can include increasing a nasal cross-sectional area with or without changing a nasal valve angle. Reshaping the cartilage can include changing a nasal valve angle. Reshaping the cartilage can include other modifications. The cartilage can completely, substantially, or partially maintain its shape (e.g., a corrected deviation shape) after the device is removed and the affected tissue heals.
[0226] Optionally, a positioning element, like that described herein, may be used to measure a desired depth or angle of treatment. As described above, the positioning element may be be inserted to the desired depth of treatment and rotated to a desired angle of treatment. Marks along the positioning element can indicate the desired depth. Marks along the base of the shaft of the positioning element can indicate the desired angle. The physician or other medical professional administering the treatment can then insert the treatment device to the desired location. The physician may also assess any other characteristics relevant to the treatment of the patient's nose that may influence the manner of treatment. In some embodiments, a reshaping element may be used to manipulate the nasal tissue into a configuration allowing improved airflow; and treatment may be performed while such a reshaping element is maintaining the desired configuration of the nasal tissue.
[0227] If the treatment device includes a monopolar electrode or electrode needles, a ground pad may be attached to the patient. The ground pad may be attached at the patient's torso, for example the shoulder or abdomen. Other locations are also possible, such as the patient's buttocks. Preferably, the point of attachment is a large, fleshy area. After being attached, the ground pad may be plugged into a power source. If the device is powered by a remote generator (e.g., RF generator), the device may then be plugged into the generator.
[0228] FIGS. 30E-30H depict an embodiment of a method for treating nasal airways. FIG. 30E depicts the nose of a patient after the solution has been applied to tissue to be treated, but prior to insertion of a device for heating and reshaping tissue. As shown in FIG. 30F, a device 800 is then inserted into a nostril of the patient. The treatment element 802 of the device 800 may be positioned within the nasal airway, adjacent to the nasal tissue (e.g., nasal septum) to be treated. The treatment element 802 may be positioned so that the electrode is in contact with the tissue to be treated. The device 800 (as shown in FIG. 30G) includes multiple needle electrodes 804, although in alternative embodiments the electrodes 804 might not be needles. The needle electrodes 804 may be inserted so that they are penetrating or engaging tissue to be treated.
[0229] The treatment element 802 may be used to deform the nasal tissue into a desired shape by pressing a convex surface of the treatment element 802 against the nasal tissue to be treated. FIG. 30G shows an internal view, from the nares, of the treatment element 802 pushing against the nasal septum and deforming the nasal septum. FIG. 30H depicts an external view of the treatment element 802 deforming the nasal septum. In some embodiments, even from the outside, the nose appears to be bulging near the area to be treated. In some embodiments, the deformation required to treat the nose is not visually detectable. A control input, such as button 814 may be used to activate the electrode and deliver energy (e.g., RF energy) to the tissue to be treated.
[0230] In some embodiments, temperature of the area around the electrode during treating is from about 30.degree. C. to about 90.degree. C. In some embodiments, temperature of the area around the electrode during treating is from about 40.degree. C. to about 80.degree. C. In some embodiments, temperature of the area around the electrode during treating is from about 50.degree. C. to about 70.degree. C. In some embodiments, temperature of the area around the electrode during treating is about 60.degree. C. In some embodiments, for example during cryo-therapy, temperature of the area around the electrode may be lower. In some embodiments, the temperature is measured at the target tissue rather than the area around the electrode during treating.
[0231] In some embodiments, treating the target tissue includes treatment for about is to about 3 minutes. In some embodiments, treating the target tissue includes treatment for about 10 seconds to about 2 minutes. In some embodiments, treating the target tissue includes treatment for about 15 seconds to about 1 minute. In some embodiments, treating the target tissue includes treatment for about 20 seconds to about 45 seconds. In some embodiments, treating the target tissue includes treatment for about 30 seconds.
[0232] In some embodiments, treating the target tissue includes delivering between about 1 and about 100 watts to the tissue. In some embodiments, treating the target tissue includes delivering between about 5 and about 75 watts to the tissue. In some embodiments, treating the target tissue includes delivering between about 10 and about 50 watts to the tissue. In some embodiments, the wattage is selected based on an amount of wattage needed to produce a desired temperature at a particular location. In some embodiments, wattage is selected based on an amount of wattage used to produce a temperature of 60.degree. C. in the cartilage.
[0233] As shown in FIGS. 30F and 30H, a thermocouple 812 may be provided on the electrode (e.g., as described with reference to FIGS. 22G and 27). In some embodiments, more than one thermocouple may be provided. For example, in embodiments comprising more than one electrode or electrode pair, each electrode or electrode pair may include a thermocouple. The thermocouple 812 may monitor temperature of the electrode and provide feedback to a control unit (e.g., control system 42 described with respect to FIG. 3). The control unit may use the data from the thermocouple 812 to regulate temperature and auto-shutoff once treatment has been achieved or in the case of an overly high temperature.
[0234] After treating the tissue, the device 800 may be removed from the nostril. If a grounding pad is used, the grounding pad may be detached from the patient.
[0235] In some embodiments, differential cooling mechanisms may be used to treat the nasal septum using electrodes or other energy delivery elements while maintaining a reduced temperature at the skin and/or mucosa. For example, devices like those described with respect to FIGS. 25A-27 or devices employing the differential cooling mechanisms described with respect to FIGS. 28A-28E may be used. The cooling system may be activated. The device may then be inserted into the nose and placed in contact with the nasal septum. The device may then be activated. Activation of the device may cause an increase in the cartilage temperature while minimizing the temperature increase in the skin and/or mucosa. The device may then be deactivated and removed from the nose.
[0236] In some embodiments, devices may be used in which insulating material is used to protect non-target tissue during energy delivery. In an embodiment, a device includes an electrode needle preferentially insulated on a portion of the needle. The needle may be inserted into the cartilage so that the insulated portion is in contact with the mucosa and/or the skin and the non-insulated portion is in contact with the cartilage. The device may be activated, causing an increase in the cartilage temperature while minimizing temperature increase in the skin and/or mucosa. The device may be deactivated and removed from the nose.
Additional Embodiments and Optional Features
[0237] Referring now to FIGS. 31A and 31B, in one embodiment, a device 900 for treating a nasal valve may include an internal power source and thus be cordless. In the embodiment shown, the device 900 includes a handle 902 coupled with a shaft 904, which in turn is coupled with a treatment element 908. The handle 902 may include a power button 910 (or "on/off switch"), a circuit board (912, FIG. 31B) and a space and connections for insertion of batteries 914 as a power source. Treatment element 904 may include multiple needle electrodes 906 for applying RF energy to tissue.
[0238] Any suitable features, elements, materials or the like that have been described above may be applied to the device 900 in a similar way. In various alternative embodiments, the device 900 may include any number, size or type of batteries, depending on the size of the handle 902 and power requirements of the device 900. In some alternative embodiments, the device 900 may include an alternative power source. For example, the batteries 914 may be rechargeable in some embodiments. In other embodiments, it may be possible to plug the device 900 into a power generator for charging, and then unplug the device 900 for use. In yet other alternative embodiments, the device 900 may include a solar power collection member. The advantage of including an internal power source in the device 900 is that this eliminates the need for the device 900 to be connected, via power cord, to a large, table-top generator, as most energy delivery surgical/medical devices require. This allows a physician to perform a nasal valve procedure in any location or patient orientation without having to manage power cables and generators.
[0239] Referring now to FIGS. 32A and 32B, in some embodiments, a system for treating a nasal valve or a nasal septum may include one or more sensors. Such sensors may be used to sense any of a number of relevant tissue properties, such as temperature, impedance and the like. The sensors may be located on a treatment device in some embodiments, or alternatively they may be separate from the treatment device and positioned at or near the device during treatment. In some embodiments, the sensor(s) may provide feedback directly to the treatment device. For example if a particular tissue temperature threshold is reached, a sensor (or sensors) may send a signal to a power generator to shut down or decrease power delivered to a treatment device. In alternative embodiment, the sensor(s) may instead provide feedback to a physician or other user, so that the physician or other user can make treatment adjustments. For example, sensors may provide a warning signal when a particular tissue temperature or impedance is reached, which will help a physician know when to turn off or decrease power delivery to a treatment device. Additionally, sensor(s) may be used to sense one or more tissue properties in any suitable tissue or multiple tissues, such as but not limited to mucosa, cartilage, dermis, epidermis and other types of body soft tissue.
[0240] FIG. 32A illustrates nasal skin in cross section, including mucosa, cartilage, dermis and epidermis. In one embodiment, a sensor device 920 may include an epidermal sensor 922 that is coupled to the epidermis via an adhesive 924. Any suitable sensor 922 (temperature, impedance, etc.) and any suitable adhesive 924 may be used. This embodiment of the sensor device 920 is also illustrated on a patient's face in FIG. 32B.
[0241] In an alternative embodiment, a sensor device 930 may include a transdermal needle sensor 932. In another alternative embodiment, a sensor device 942 may be attached directly to a treatment device 940. As illustrated by these various embodiments, sensors 922, 932 and 942 may be positioned either at or near a treatment location during a treatment. In some embodiments, for example, a sensor 922, 932 may be placed on or in epidermis while a treatment is being performed on mucosa and/or cartilage. Alternatively, a sensor 942 may be placed directly on mucosa or cartilage during a treatment of mucosa or cartilage. Additionally, in any given embodiment, multiple sensors may be placed at multiple different locations in and/or on tissue. As mentioned above, the sensor devices 920, 930 and 940 may, in various embodiments, provide any of a number of different types of feedback, such as feedback to a user, feedback to a power generator, or both.
[0242] Referring now to FIGS. 33A and 33B, in some embodiments, a treatment device 950 may include a treatment element 952 with wings 954 extending laterally from it. The wings 954 are configured to help direct the treatment element 952 into a particular treatment location/position and/or to prevent the treatment element 952 from contacting tissue that the physician does not want to treat. For example, FIG. 33B illustrates a top, cross-sectional view of a nose N, showing the lateral wall LW, nasal septum S and an intersection I between the lateral wall LW and nasal septum S. The wings 954 help prevent the treatment element 952 from being advanced far enough to reach an undesired treatment area. Alternative embodiments may include additional wings or other protrusions or shapes to prevent contact with particular structures. Some embodiments may include adjustable wings or wings that expand once the electrodes have been placed. Any other size, shape or configuration of one of more wings may be included, according to various embodiments.
[0243] Referring to FIGS. 34A-34C, in various alternative embodiments, treatment elements of nasal valve treatment devices may have different shapes and/or sizes for addressing different types and/or shapes of tissue. For example, as shown in FIG. 34A, in one embodiment, a treatment element 960 of a device may have a square or rectangular profile with a flat distal end, which may be ideal for addressing relatively flat tissue configurations. Two electrodes 962 (or two sets of electrodes) may be used to send an arc of current (e.g., RF current) through tissue in the pattern shown by the multi-headed arrow.
[0244] In another embodiment, as shown in FIG. 34B, a treatment element 970 may have an oval profile with a curved distal end. Two electrodes 972 or sets of electrodes send a current through tissue in an arc. This configuration may be advantageous for addressing tissue having a curved profile. In yet another embodiment, as shown in FIG. 34C, a treatment element 980 may have a flatter curved profile, for example for addressing tissue with a curved shape but not as sharp of an angle as the tissue shown in FIG. 34B. Again, the electrodes 982 send energy through the curved tissue in a curved arc.
[0245] As is evident from FIGS. 34A-34C, a treatment element of a treatment device may have any suitable configuration for advantageously addressing any tissue type and shape. In some embodiments, multiple different treatment devices, each having a differently shaped treatment element, may be provided, and a user may select a treatment device for a particular tissue type and/or shape, based at least in part on the shape of the treatment element of the device.
[0246] Referring to FIG. 35, in another embodiment, a treatment device 1000 for treating a nasal septum and/or tissues near the nasal septum may include an expandable member 1002, such as but not limited to an expandable polymeric balloon. For example, a non-compliant or semi-compliant balloon may be used in some embodiments to expand tissues in and/or around the nasal valve, to achieve one or more of the effects discussed above in relation to the various embodiments. Although expandable balloon devices (e.g., balloon catheters) have been described previously for use in expanding ostia (openings) of the sinus cavities, they have not been described for use in changing the shape and/or physical characteristics of the nasal valve. In various method embodiments, the expandable member 1002 may be positioned at any of a number of suitable locations at or near a nasal valve and then expanded to deform tissues that make up the nasal septum. In some cases, these tissues may be at least partially deformed permanently or at least for a period of time after the procedure. In other cases, the tissue may only be deformed during the procedure, but one or more properties of the tissue may be affected by the balloon expansion.
[0247] In various alternative embodiments, a treatment device for nasal septum tissue and/or other nasal tissue may use a treatment modality that does not involve delivery of energy to, or removal of energy from, tissue. For example, in some embodiments, the treatment device may create some kind of mechanical injury to one or more tissues to cause a change in shape and/or one or more properties of the tissue. The expandable balloon embodiment described above is one example. Other examples may include, but are not limited to, needles, micro-needles, blades or the like, any of which may be used to cause scar tissue formation and/or tissue contraction. Other embodiments may use sclerotherapy, involving injecting one or more substances (acid, coagulants, etc.) into the target tissue to induce scar tissue formation and/or other changes in the tissue properties. In some cases, one type of tissue (for example, mucosa or cartilage) may be transformed into a different type of tissue altogether (for example, scar tissue). In other examples, one or more properties of the tissue may be changed without changing the overall type of tissue. For example, the tissue may be caused to shrink, contract, stiffen and/or the like. One advantage of these non-energy-based embodiments is that they do not require a source of energy. This may make them easier to use and possibly to manufacture and supply.
[0248] In other embodiments, thermal energy may be applied to the nasal tissue by applying the energy from an external location on the nose, rather than an internal location within the nasal cavity. For example, in some embodiments, a treatment device may be positioned on the nose and used to deliver thermal energy through the epidermis to the nasal septum. In some embodiments, the treatment device may also be used to cool the superficial dermis and epidermis, for example. This delivery of energy may, in some embodiments, act to tighten tissues of the nasal valve, thus preventing collapse during breathing. In an alternative embodiment, instead of using thermal energy to change the tissues, a treatment device may use mechanical means, such as micro-needles, to create a subdermal tissue response, such as scarring, for a similar type of tissue tightening effect.
[0249] In yet other embodiments, some methods for treating a nasal valve may include applying a gel, paste, liquid or similar substance to a surface of the nose during an energy delivery treatment of the nasal valve. Such substances may be applied to target tissue, such as mucosa, non-target tissue, such as epidermis, or a combination of both. The substance (or substances) applied may serve any of a number of different purposes, such as but not limited to modifying conductivity of tissue and providing anesthetic effect. Conductivity enhancing substances may improve the efficiency and/or consistency of energy delivery (such as but not limited to RF energy). Alternatively or additionally, one or more substances may be injected into tissue. For example, saline, Lidocaine, other anesthetic agents, or any other suitable agents, may be injected. Some embodiments may involve applying one substance and injecting another substance.
[0250] In other alternative embodiments, it may be possible to achieve desired changes in tissue properties and/or shapes by injecting substance or applying substance only--in other words, without also applying energy. For example, injecting a sclerotherapy substance into tissue may, in some embodiments, achieve a desired tissue result. In additional alternative embodiments, a method of treating a nasal septum may include injecting a substance into nasal tissue and then curing the substance in order to change the substance's properties and, in turn, at least one of the nasal tissue's properties. A treatment device may be used to cure the substance. In some embodiments, the treatment device may be used to deform the target tissue and cure the substance, while the tissue is deformed, so that the tissue retains approximately the same, deformed shape after the substance is cured and the treatment device is removed. In another alternative embodiment, a surface-based biodegradable agent may be applied and cured to change the shape of the target nasal tissue.
[0251] FIGS. 36A-36E illustrate a method and device for treating a septum 1102 having a deviation 1104, according to some embodiments. The method and device of FIGS. 36A-36E may include one or more features of previously described treatment methods and devices, including but not limited to those described in relation to FIGS. 30A-30H.
[0252] FIG. 36A illustrates septum 1102 and deviation 1104 as though looking into a patient's left nostril. FIG. 36B illustrates an enlarged detail view of septum 1102 and deviation 1104.
[0253] FIG. 36C illustrates an applicator 1106 applying a treatment solution to deviation 1104. As illustrated, applicator 1106 is an injection device used to inject the treatment solution in an area of deviation 1104, but other application methods and applicators may be used. Other application methods include, but are not limited to, topical application, diffusion, current-driven application, electrophoresis, and any other method of applying a solution and/or enzymes to target tissue. The solution may be left to act for a designated dwell time to allow the solution to effectively condition the septal cartilage. The dwell time may be in the range of about 1 minute to about 60 minutes, about 1 minute to about 40 minutes, about 1 minute to about 20 minutes, about 1 minute to about 10 minutes, about 1 minute to about 5 minutes, or other ranges. In some embodiments, the dwell time is less than one minute. In some embodiments, there is approximately no dwell time at all.
[0254] FIG. 36D illustrates a treatment device 1110 being used to correct deviation 1104. As illustrated, treatment device 1110 includes a handle 1112, a first elongate shaft 1114 extending from handle 1112, a first treatment element 1116 disposed at a distal end of first shaft 1114, a second shaft 1118 extending from handle 1112, and a second treatment element 1120 disposed at a distal end of second shaft 1118. First treatment element 1116 may include one or more energy delivery or removal elements. First treatment element 1116 may be configured to be placed against the tissue of deviation 1104. Second treatment element 1120 may include one or more energy delivery or removal elements. Second treatment element 1120 may be configured to be placed on an opposite size of septum 1102 from first treatment element 1116. For example, as illustrated, first treatment element 1116 is placed against deviation 1104 in the left nostril, and second treatment element 1120 may be configured to be placed against septum 1102 in the right nostril. Second treatment element 1120 may act as a backstop, counter-traction element, guide, or otherwise facilitate treatment of deviation 1104 with device 1110. In some embodiments, treatment device 1110 includes applicator 1106. For example, first treatment element 1116 may include a needle configured as applicator 1106 adapted to apply the solution to deviation 1104. Other treatment devices may be used.
[0255] Using treatment device 1110 to correct deviation 1104 may include applying energy to or removing energy from septum 1102 using first treatment element 1116 and/or second treatment element 1120. In some embodiments, the application or removal of energy may enhance, inhibit, or otherwise modify the effect of the solution on the cartilage. In some embodiments, the application or removal of energy may deactivate the solution so it no longer substantially acts on the cartilage. In some embodiments applying energy to or removing energy from septum 1102 may further treat or modify cartilage of deviation 1104.
[0256] Using treatment device 1110 to correct deviation 1104 may include applying mechanical energy. In some embodiments, first treatment element 1116 and second treatment element 1120 may be used to apply mechanical energy to septum 1102. For example, a user may squeeze a portion of handle 1112, causing first treatment element 1116 and second treatment element 1120 to come together and pinch deviation 1104. In some embodiments, first treatment element 1116 moves towards second treatment element 1120, pushing deviation 1104 toward and/or against second treatment element 1120. Second treatment element 1120 may act as a backstop. In some embodiments, mechanical energy may be used to push deviation 1104 beyond flat, such that once device 1110 is removed and tissue of the cartilage heals, deviation 1104 is corrected. In some embodiments, second treatment element 1120 may be configured as a mold. For example, second treatment element 1120 may have a particular shape (e.g., concave or convex) and first treatment element 1116 may have a complimentary shape or may otherwise be configured to shape deviation 1104 against second treatment element 1120.
[0257] FIG. 36E illustrates septum 1102 having a corrected deviation 1108. Compared to deviation 1104, corrected deviation 1108 may be a slightly reduced deviation, a completely removed deviation, or otherwise corrected. A nose with corrected deviation 1108 may have improved airflow compared to a nose with deviation 1104.
[0258] FIG. 37 illustrates a channel stylus device 1200, which may be used to treat a deviated septum, according to some embodiments. Channel stylus device 1200 includes a handle 1202, an elongate shaft 1204 extending from handle 1202, and an elongate treatment element 1206. Elongate treatment element 1206 may be configured to create channels in a deviation, thereby reducing the deviation and improving airflow. Elongate treatment element 1206 may be further configured to apply energy to or remove energy from tissue. Elongate treatment element 1206 may include a plurality of pairs of bi-polar electrodes 1208. Electrodes 1208 may be arranged in a serial alignment along treatment element 1206 such that electrodes 1208 are in a line along treatment element 1206. For example, electrodes 1208 may be arranged with the center of each electrode 1208 along a longitudinal axis of treatment element 1206.
[0259] FIGS. 38A-38C illustrate a method of treating a septum 1210 having a deviation 1212 using channel stylus device 1200, according to some embodiments. FIG. 38A illustrates septum 1210 and deviation 1212 as though looking into a patient's left nostril. FIG. 38B illustrates treatment element 1206 of channel stylus device 1200 inserted into the nostril and pressing laterally against deviation 1212, thereby reshaping deviation 1212. As illustrated in FIG. 38B, treatment element 1206 may apply energy to or remove energy from the tissue of deviation 1212. This may facilitate the reshaping of deviation 1212. Before applying treatment element 1206 to deviation 1212, a treatment solution may be applied at or near deviation 1212 to facilitate treatment. By reshaping and applying energy to or removing energy from deviation 1212, the method may create a treated deviation that persists after treatment element 1206 is removed and the tissue of deviation 1212 heals. Reshaping deviation 1212 may include flattening deviation 1212. Reshaping deviation 1212 may include creating air channels, troughs, or other shapes in deviation 1212 to facilitate the flow of air. Such air channels may be particularly useful for treating posterior-running septal deviations. Reshaping deviation 1212 can include increasing a nasal cross-sectional area with or without changing a nasal valve angle. Reshaping deviation 1212 can include modifying a nasal valve angle. FIG. 38C illustrates a treated deviation 1214 after device 1200 is removed and the tissue of septum 1210 heals. As illustrated, air channels 1216 through which air may flow are formed in treated deviation 1214. Compared to deviation 1212, a patient having treated deviation 1214 may have improved airflow through the nostril.
[0260] FIGS. 39A-39D illustrate a method of treating a deviated septum 1302 by treating and evacuating cartilage 1304, according to some embodiments. FIG. 39A is a view of a nose having deviated septum 1302. FIG. 39B illustrates an applicator 1306 applying a solution 1308 to cartilage 1304. As illustrated, applicator 1306 is an injector injecting solution 1308 into or near cartilage 1304. Solution 1308 may be a solution that treats cartilage by, for example, softening or dissolving cartilage, such as those previously described herein. Other applicators and application methods may also be used. After solution 1308 has been applied to cartilage 1304, solution 1308 may be given a dwell time in which to act on cartilage 1304. After solution 1308 is applied, treated cartilage 1304 may be further treated, such as by a treatment element applying energy to or removing energy from treated cartilage 1304 or by a treatment element applying mechanical force to treated cartilage 1304. The further treatment may be selected to enhance, inhibit, guide, shape or otherwise modify treatment of cartilage 1304. In some embodiments, a portion of cartilage outside of the treatment area may be treated to denature or otherwise deactivate solution 1308 if it reaches that portion of cartilage. In some embodiments, the further treatment may include applying energy or force to treated cartilage 1304 to break or further weaken cartilage 1304 for ease of removal.
[0261] FIG. 39C illustrates an evacuator 1310 evacuating treated cartilage 1304. After the treatment or further treatment of cartilage 1304, cartilage 1304 may be in a condition ready for removal. As illustrated, evacuator 1310 may be inserted into septum 1302 and used to apply suction to remove treated cartilage 1304. Other evacuators 1310 and methods of evacuation may be used. The evacuation of softened cartilage 1304 may be performed with or without an incision. In some embodiments, an incision is made and cartilage 1304 is removed through the incision. In other embodiments, no incision is used and instead cartilage 1304 is removed through a needle inserted into septum 1302. In other embodiments, there is no active evacuation step. Instead, cartilage 1304 may be sufficiently treated that it does not need to be removed; for example, treated cartilage 1304 may be naturally resorbed by the patient's body or that cartilage 1304 is sufficiently weak that it no longer substantially negatively affects airflow. FIG. 39D illustrates a treated nose having a corrected deviation of septum 1302. With deviated cartilage 1304 removed, septum 1302 is no longer deviated.
[0262] Although emphasis has been placed on structure and function of the nasal septum in much of the foregoing description, modifications of other cartilage or tissue may also be performed based on the above disclosures. This may include, but need not be limited to hyaline or other cartilage located in a subject's nose, larynx, trachea, bronchi, airways, ribs, bones, joints, and other locations.
[0263] Although this invention has been disclosed in the context of certain preferred embodiments and examples, it will be understood by those skilled in the art that the present invention extends beyond the specifically disclosed embodiments to other alternative embodiments and/or uses of the invention and obvious modifications and equivalents thereof. Thus, it is intended that the scope of the present invention herein disclosed should not be limited by the particular disclosed embodiments described above, but should be determined only by a fair reading of the claims that follow.
EXPERIMENTS
[0264] Experiments may be carried out to explore ranges in concentration and volume of solutions applied to cartilage and duration of exposure in terms of their efficacy in degrading the cartilage surface. Additionally, the effect of temperature variation on solution activity may be explored. The aims of the experiments may include: comparing the effects of collagenase and trypsin (or other enzymes or solutions) exposure on the ability to enhance RF-induced reshaping of nasal septum cartilage and evaluate the effects of particular RF energy exposure to reshape treated nasal septum cartilage.
[0265] The efficacy of combined solution exposure and RF treatment may be assessed using bovine nasal septum cartilage obtained from a slaughterhouse. Nasal cartilage samples may be acquired within 24 hours of slaughter to ensure high cell viability and all protocols below may be carried out under sterile conditions to enable long term observation of cellular activity after enzyme and RF exposure.
[0266] Concentrations, volumes, and durations of exposures for solutions may be chosen based on previous work with articular cartilage. See, e.g., Griffin et al, Effects of Enzymatic Treatments on the Depth-Dependent Viscoelastic Shear Properties of Articular Cartilage, J Orthop Res Vol. 32, Issue 12, pp. 1652-1657 (2014), incorporated herein by reference for any and all purposes. A range of 0.5 ml to 2.5 ml of enzyme solutions may be applied to nasal septum cartilage at concentrations ranging from 1-10 mg/ml collagenase and/or 10-100 .mu.g/ml trypsin for times ranging from 15 to 90 minutes. In an example, a solution having a concentration of approximately 40-60 .mu.g/ml trypsin can be applied to nasal septum cartilage and given a dwell time of 30-60 minutes. In an example, a solution having a concentration of approximately 50 .mu.g/ml trypsin can be applied to nasal septum cartilage and given a dwell time of 40 minutes. In another example, a solution having a concentration of approximately 1-3 mg/ml collagenase can be applied to nasal septum cartilage and given a dwell time of approximately 15-25 minutes. In an example, a solution having a concentration of approximately 2 mg/ml collagenase can be applied to nasal septum cartilage and given a dwell time of approximately 20 minutes. Such treatments may be expected to result in a narrow band of degraded cartilage ranging from 100 .mu.m to 1 mm from the cartilage surface. Additionally, prior to application of the solution to cartilage, solutions may be prepared at 20.degree. C., 37.degree. C., 60.degree. C., and 80.degree. C. to document the efficiency of degradation at room temperature, body temperature, a temperature consistent with the current operation of the RF probe, and a temperature in excess of the current operation of the RF probe. Immediately after solution exposure, the thickness of all samples may be measured and subset of samples may be characterized by live/dead staining, histology and microscopy, and mechanical analysis by confocal elastography.
[0267] Remaining samples may be shaped using the RF probe, using standard operating conditions (e.g., sufficient RF wattage to produce a temperature of 60.degree. C. and application of 0.5 kg of force to the tissue for 30 seconds). Immediately after application of RF energy, the thickness of all samples may be measured and subset of samples may be characterized by live/dead staining, histology and microscopy, and mechanical analysis by confocal elastography (see below for descriptions).
[0268] The remaining samples may be maintained in sterile culture at 37.degree. C. in DMEM with 10% fetal bovine serum in a 5% CO2 atmosphere for times up to 1 week. After culture, these remaining samples may be characterized by live/dead staining, histology and microscopy, and mechanical analysis by confocal elastography (see below for descriptions).
[0269] Experimental Procedure
[0270] The experiment may explore the combination of controlled application of derivative solutions and RF energy to nasal cartilage to enable reshaping of a nasal septum.
[0271] Tissue Isolation, Live/Dead Staining
[0272] Bovine nasal septa are obtained from a local slaughterhouse within 24 hours of slaughter. Cartilage from the nasal septum is dissected under sterile conditions and cut to produce slabs of tissue 1 cm.times.1 cm.times.2 mm thick. Prior to use in experiments, tissue slabs are cultured in DMEM with 10% fetal bovine serum with 100 .mu.g/ml penicillin and 100 U/ml streptomycin.
[0273] Prior to experiments, sample thickness is measured with digital calipers. Digital photographs are recorded. After solution exposure, RF exposure, and post-exposure culture, a subset of samples are bisected and stained with a commercial live/dead kit (such as those provided by Invitrogen, Inc.). Samples are be exposed to 0.15 .mu.m calcein AM and 2 .mu.m ethidium homodimer-1 (EthD-1) for 60 minutes at room temperature. The stained samples are analyzed under a microscope equipped with an epifluorescence attachment (e.g., a Nikon TE2000-S) and a digital camera (e.g., a Spot RT digital camera).
[0274] Histology and Microscopy
[0275] To assess structural and compositional features of nasal septum cartilage after exposure to the solution, RF exposure, and post-exposure culture, a separate subset of samples are fixed in neutral formalin and processed for histology and Fourier Transform Infrared (FTIR) microscopy. To assess collagen structure in nasal cartilage samples, fixed samples are embedded in paraffin, cut into thin sections, and dewaxed in three xylene baths for 2 minutes each, rehydrated in three baths of ethyl alcohol (100%, 95%, and 70% ethanol, respectively, appropriately diluted with distilled water) for 2 minutes each, and dyed with Picrosirius red for 1 hours. This histochemical staining selectively binds to fibrillar collagen, enhancing tissue birefringence. Once prepared, samples are placed on a bright-field microscope with a 4.times. objective and two independently rotating linear polarizers set 90.degree. apart. Incident light is polarized, passed through the sample, and then passed through a second polarizer, producing a map of the local birefringence.
[0276] FTIR microscopy is used measure the local composition of proteoglycan and collagen within these samples, as described by our group previously. See, e.g., Silverberg et al, Structure-Function Relations and Rigidity Percolation in the Shear Properties of Articular Cartilage, Biophysical Journal, vol. 107, pp. 1721-1730 (2014), incorporated herein by reference for any and all purposes. Sections, 4 mm thick from each tissue sample, are be placed on 2-mm-thick mid-infrared (IR) transparent BaF.sub.2 disks that are 25 mm in diameter. Sections are dewaxed and rehydrated as described above. Samples are loaded into a Fourier transform infrared imaging (FTIR-I) microscope (e.g., a Hyperion 2000 FTIR-I microscope) in transmission mode set to acquire data on wavenumbers between 600 cm.sup.-1 and 4000 cm.sup.-1 with a resolution of 4 cm.sup.-1. A 15.times. objective is used with a slit aperture configured to acquire spectra over a rectangular region 25.times.200 mm.sup.2. Fifteen background-corrected scans are repeated at a given measurement point and averaged to generate a single IR spectra. The acquisition window is translated along the tissue thickness by a computer-controlled stage to acquire measurements at 80 points spaced 25 mm apart.
[0277] The two primary solid-matrix contributions to nasal septum come from type II collagen and aggrecan. Hence, pure compound spectra where both compounds were extracted from bovine articular cartilage is used. Each spectra is fit to a linear combination of a type II collagen spectrum, an aggrecan spectrum, and a linear baseline over the spectral window from 900 cm.sup.-1 to 1725 cm.sup.-1. The final product of this fit is a local map of collagen and proteoglycan concentration in solution- and RF-treated cartilage, which enables the determination of any compositional changes induced by these treatments.
[0278] Confocal Elasography
[0279] To assess the individual and combined effects of solution treatment and RF energy delivery to cartilage on the local mechanical properties of cartilage, grid-resolution automated tissue elastography (GRATE) is used to map local strains in repair cartilage. See, e.g., Buckley et al, High-Resolution Spatial Mapping of Shear Properties in Cartilage, J. Biomechanics, vol. 43, pp. 796-800 (2010); see also Buckley et al, Mapping the Depth Dependence of Shear Properties in Articular Cartilage, J Biomechanics vol. 41, pp. 2430-2437 (2008), each incorporated herein by reference for any and all purposes. For this analysis, samples of bovine septal cartilage obtained from experiments described above is cut longitudinally exposed to 7 .mu.g/mL 5-dichlorotriazinylaminofluorescein (5-DTAF) for 2 hours to uniformly stain the extracellular matrix. Samples are placed in a tissue deformation imaging stage (TDIS) and mounted on an inverted confocal microscope (e.g., the Zeiss LSM 510) where gridlines with a spacing of 50 .mu.m are photobleached on the sample using a 488 nm laser. A series of steps in compressive strain are imposed on the sample via the TDIS, and at each compressive strain, a series of sinusoidal shear displacements are imposed on the sample at frequencies ranging from 0.001 Hz to 1 Hz. During the application of compressive and shear displacements, load cells mounted on the TDIS will record the resultant forces. Simultaneous to this, images of the sample are acquired at 20 Hz. Using custom image analysis MATLAB code, the intensity minima corresponding to the location of the photobleached lines is tracked, and the local strains are determined from the slopes of these photobleached lines. At 20 .mu.m layers through the tissue the local modulus are calculated from the measured loads and local strains.
[0280] The result of these studies is a characterization of the treatment of nasal septum cartilage that change the shear properties of the tissue. This technique enables the description of the strain field on a length scale of 20 .mu.m.times.20 .mu.m, which is on the order of 1% of the tissue thickness. This data combined with that obtained from histological will gives insight into how cartilage reshaping techniques affects the local structure and properties of the tissue.
[0281] Although emphasis has been placed on structure and function of the nasal septum in much of the foregoing description, modifications of other cartilage or tissue may also be performed based on the above disclosures. This may include, but need not be limited to hyaline or other cartilage located in a subject's nose, larynx, trachea, bronchi, airways, ribs, bones, joints, and other locations.
[0282] Although this invention has been disclosed in the context of certain preferred embodiments and examples, it will be understood by those skilled in the art that the present invention extends beyond the specifically disclosed embodiments to other alternative embodiments and/or uses of the invention and obvious modifications and equivalents thereof. Thus, it is intended that the scope of the present invention herein disclosed should not be limited by the particular disclosed embodiments described above, but should be determined only by a fair reading of the claims that follow.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013
D00014

D00015
D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025
D00026
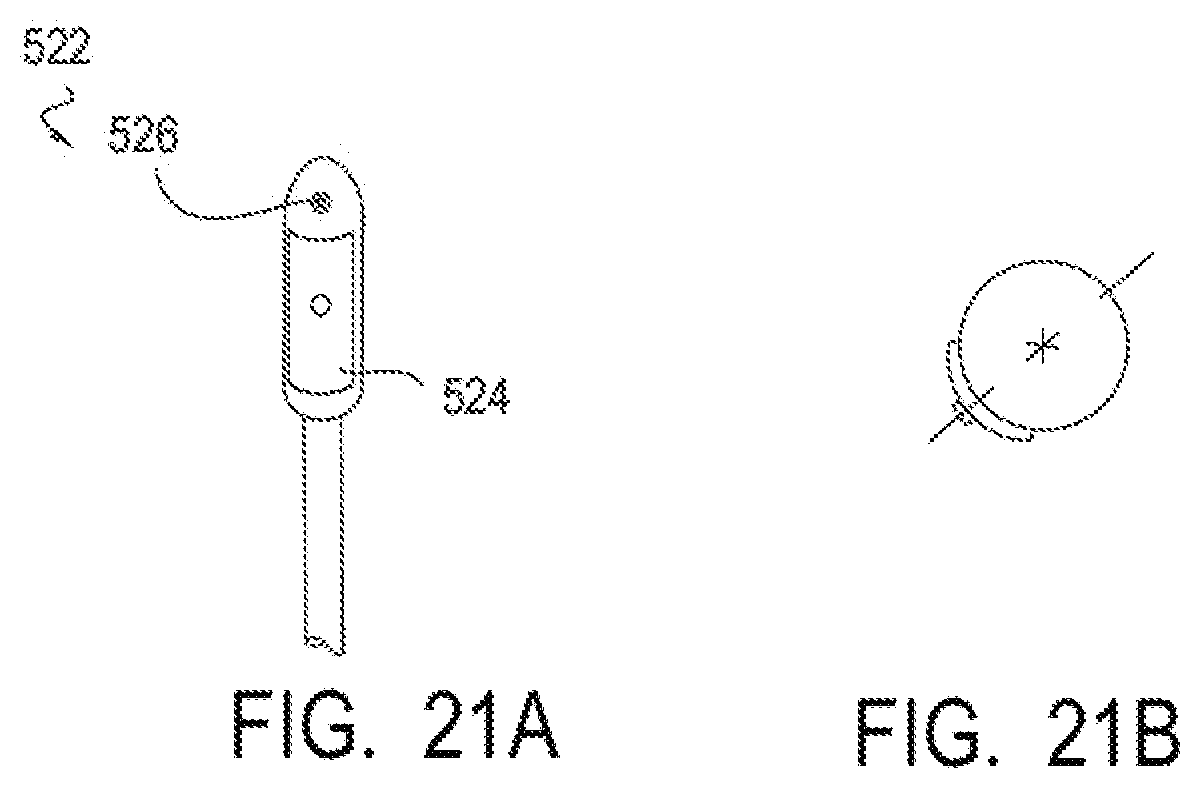
D00027
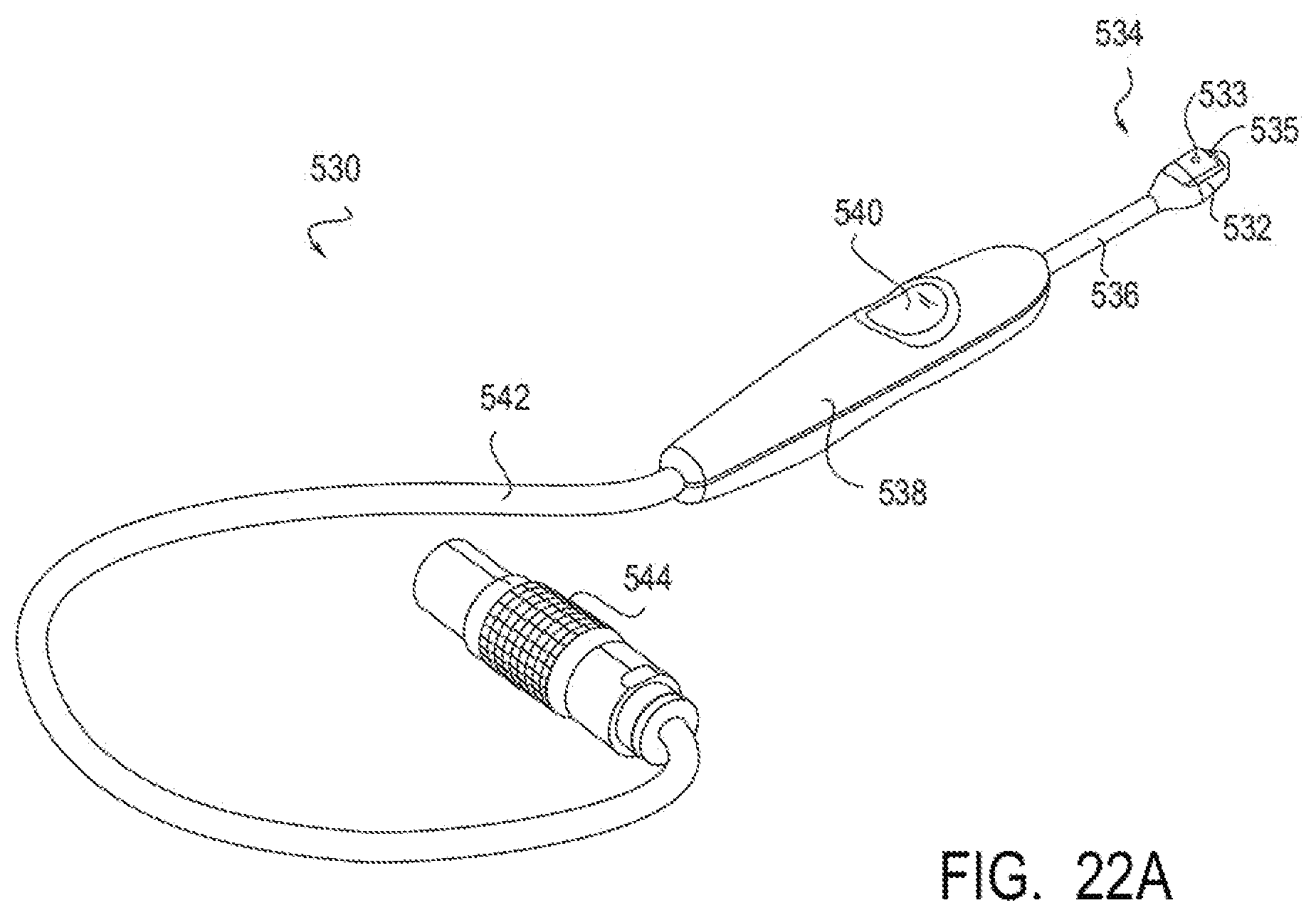
D00028
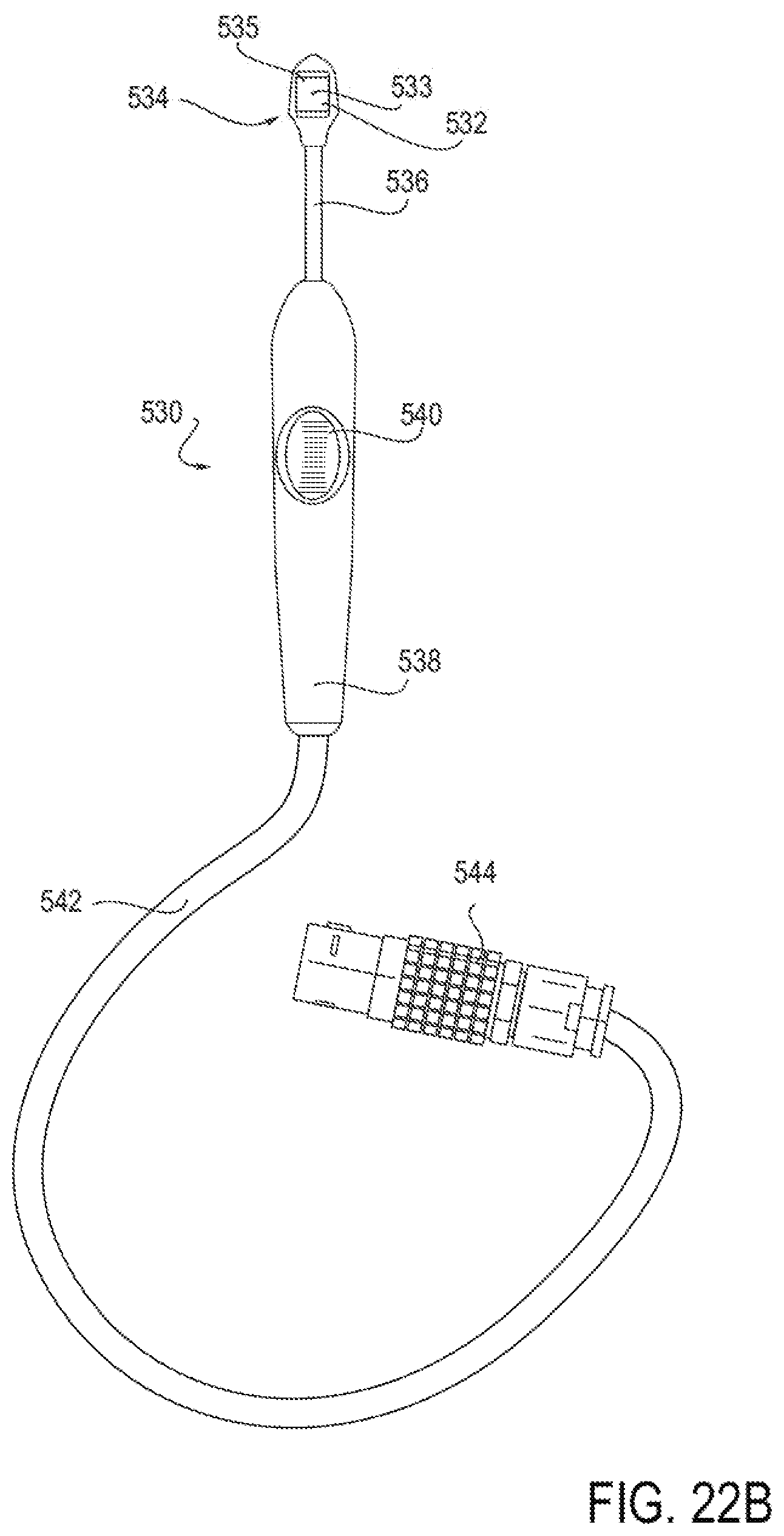
D00029

D00030

D00031

D00032

D00033
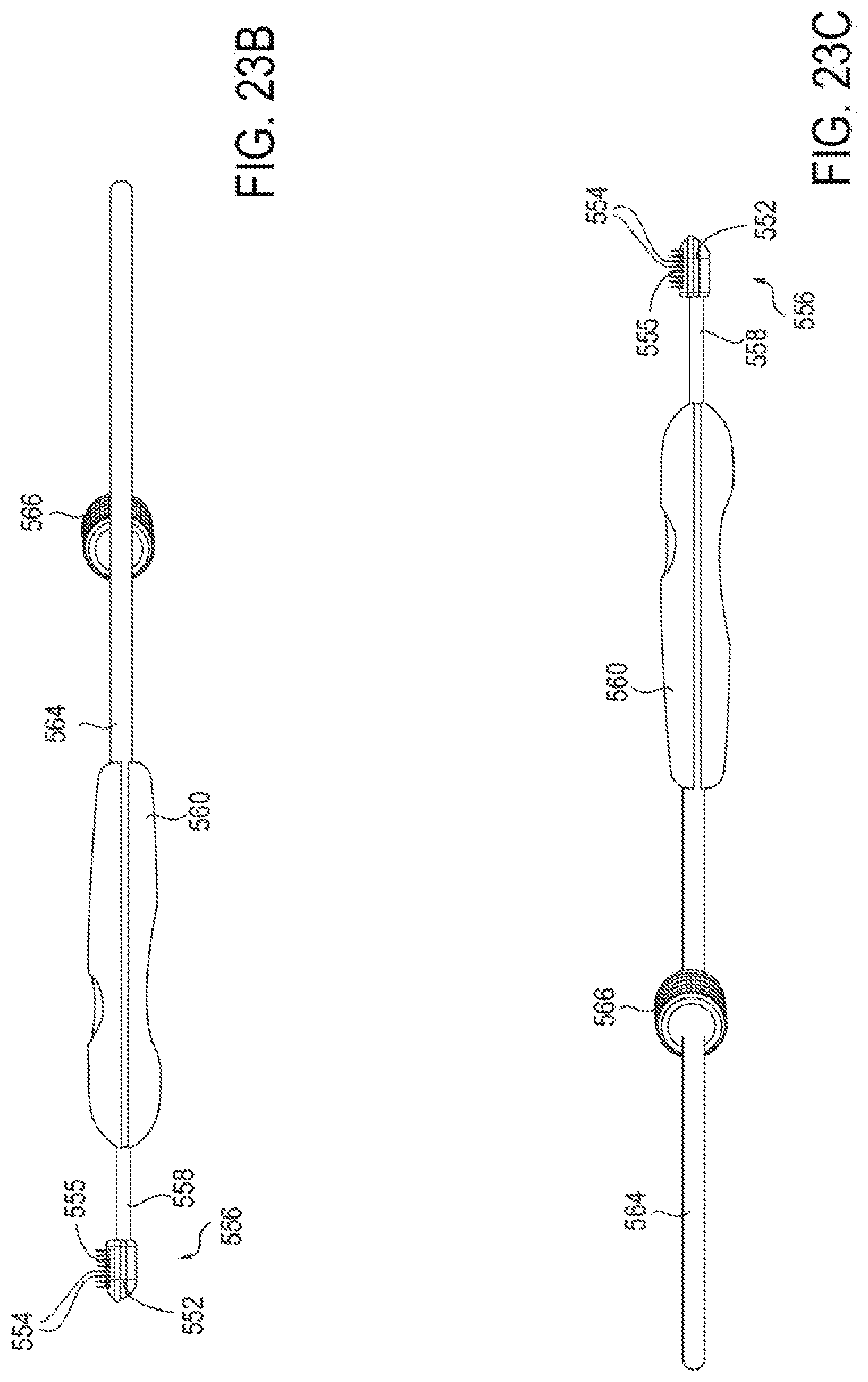
D00034

D00035
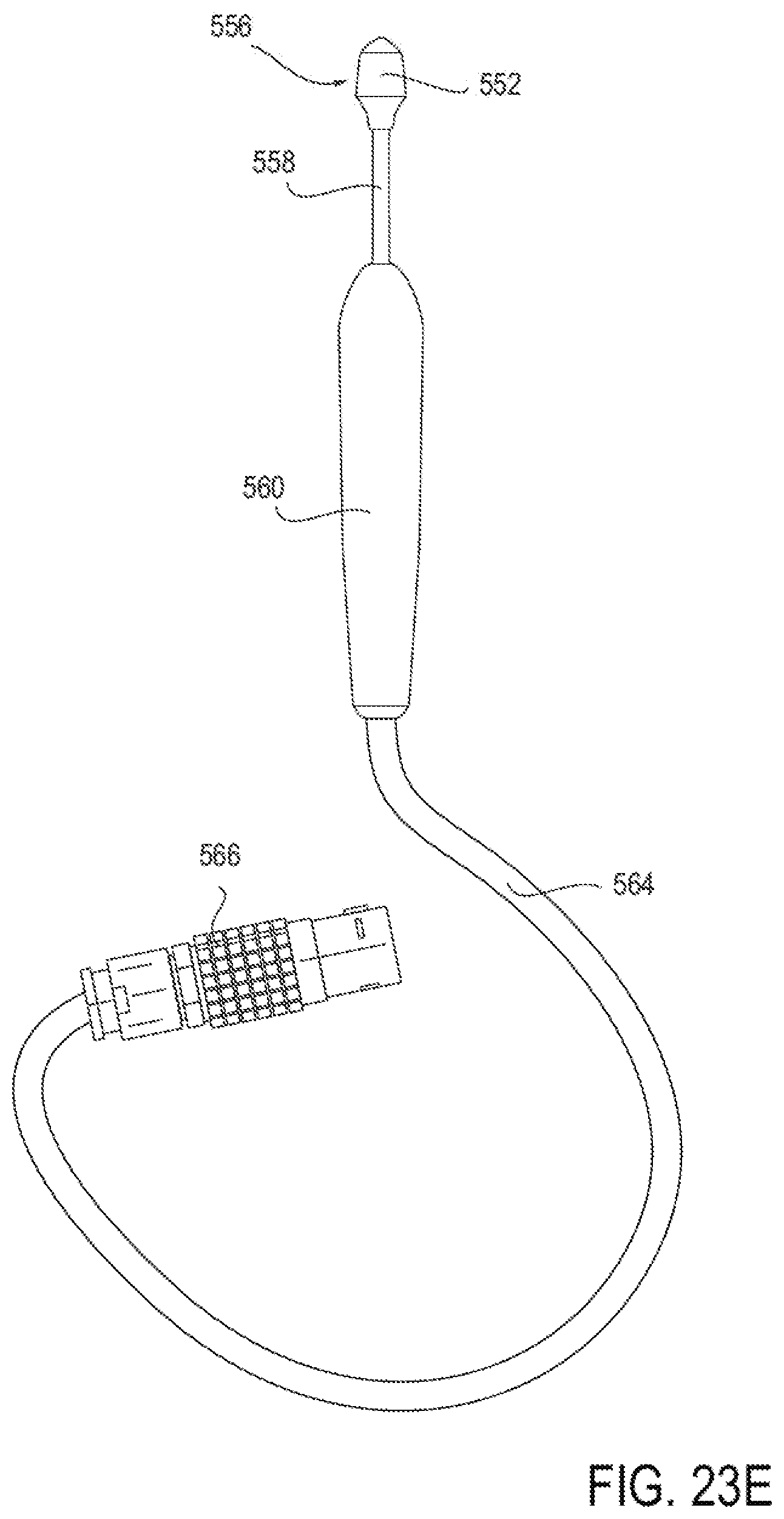
D00036

D00037

D00038

D00039

D00040

D00041

D00042

D00043
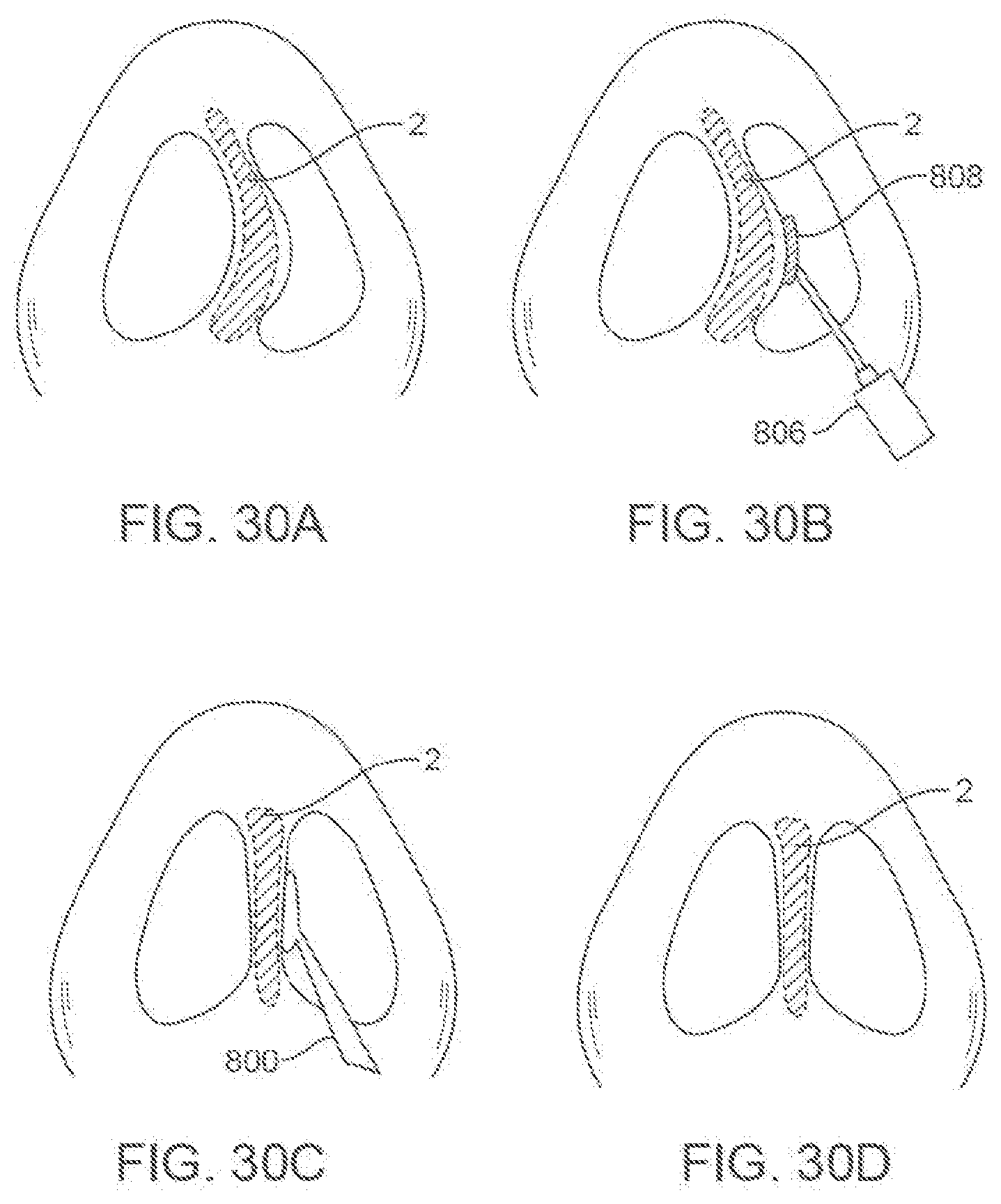
D00044

D00045

D00046

D00047

D00048

D00049

D00050

D00051

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.