Methods, Compounds, and Compositions For Treatment and Prophylaxis in the Respiratory Tract
Fang; Fang ; et al.
U.S. patent application number 16/893265 was filed with the patent office on 2020-09-24 for methods, compounds, and compositions for treatment and prophylaxis in the respiratory tract. This patent application is currently assigned to Ansun Biopharma, Inc.. The applicant listed for this patent is Ansun Biopharma, Inc.. Invention is credited to Fang Fang, Michael P. Malakhov, Ron Moss, David Wurtman.
| Application Number | 20200297822 16/893265 |
| Document ID | / |
| Family ID | 1000004870474 |
| Filed Date | 2020-09-24 |









View All Diagrams
| United States Patent Application | 20200297822 |
| Kind Code | A1 |
| Fang; Fang ; et al. | September 24, 2020 |
Methods, Compounds, and Compositions For Treatment and Prophylaxis in the Respiratory Tract
Abstract
The present invention provides a method of reducing the quantity of mucus in the respiratory tract of a subject with elevated levels of mucus in said respiratory tract. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a fusion protein comprising a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount comprises an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the compound or composition.
| Inventors: | Fang; Fang; (Rancho Santa Fe, CA) ; Wurtman; David; (San Diego, CA) ; Moss; Ron; (Encinitas, CA) ; Malakhov; Michael P.; (San Francisco, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Ansun Biopharma, Inc. San Diego CA |
||||||||||
| Family ID: | 1000004870474 | ||||||||||
| Appl. No.: | 16/893265 | ||||||||||
| Filed: | June 4, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15865217 | Jan 8, 2018 | |||
| 16893265 | ||||
| 15136751 | Apr 22, 2016 | |||
| 15865217 | ||||
| 14489428 | Sep 17, 2014 | |||
| 15136751 | ||||
| 13769095 | Feb 15, 2013 | |||
| 14489428 | ||||
| 12940742 | Nov 5, 2010 | 8398971 | ||
| 13769095 | ||||
| 61259033 | Nov 6, 2009 | |||
| 61259055 | Nov 6, 2009 | |||
| 61322813 | Apr 9, 2010 | |||
| 61332063 | May 6, 2010 | |||
| 61381420 | Sep 9, 2010 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 49/0008 20130101; C07K 14/485 20130101; A61K 33/14 20130101; C12N 9/2477 20130101; A61K 9/06 20130101; A61K 31/198 20130101; A61K 9/0014 20130101; A61K 45/06 20130101; A61K 38/1808 20130101; A61K 31/05 20130101; C12Y 302/01018 20130101; A61K 38/47 20130101; A61K 9/0073 20130101; G01N 2800/122 20130101; A61K 9/0043 20130101; G01N 2500/00 20130101; A61K 47/26 20130101; A61K 31/4172 20130101; A61K 31/7016 20130101; A61K 33/06 20130101; A61K 47/32 20130101; C07K 2319/00 20130101; A61K 9/0019 20130101; A61K 47/22 20130101; C12Q 1/34 20130101; A61K 47/34 20130101; A61K 9/0053 20130101; A61K 47/02 20130101; C12N 9/2402 20130101 |
| International Class: | A61K 38/47 20060101 A61K038/47; A61K 9/00 20060101 A61K009/00; A61K 9/06 20060101 A61K009/06; A61K 31/05 20060101 A61K031/05; A61K 31/198 20060101 A61K031/198; A61K 33/06 20060101 A61K033/06; A61K 33/14 20060101 A61K033/14; A61K 47/32 20060101 A61K047/32; A61K 47/34 20060101 A61K047/34; C12N 9/24 20060101 C12N009/24; A61K 31/4172 20060101 A61K031/4172; A61K 31/7016 20060101 A61K031/7016; A61K 45/06 20060101 A61K045/06; C12Q 1/34 20060101 C12Q001/34; A61K 38/18 20060101 A61K038/18; C07K 14/485 20060101 C07K014/485; A61K 47/02 20060101 A61K047/02; A61K 47/22 20060101 A61K047/22; A61K 47/26 20060101 A61K047/26; A61K 49/00 20060101 A61K049/00 |
Goverment Interests
FEDERALLY SPONSORED RESEARCH OR DEVELOPMENT
[0002] This invention was made with Government support under contract number HHSN266200600015C awarded by the United States Department of Health and Human Services, National Institutes of Health. The Government has certain rights in the invention.
Claims
1. A method of reducing the quantity or level of mucus or preventing an increase in the quantity or level of mucus in a respiratory tract of a subject, the method comprising: administering to the subject a compound or composition comprising a therapeutically effective amount of a fusion protein, wherein the fusion protein comprises at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase comprises the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12, inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin comprising the amino acid sequence of SEQ ID NO:7; and the therapeutically effective amount comprises an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the compound or composition.
Description
CLAIM OF PRIORITY
[0001] This application claims the benefit of U.S. patent application Ser. No. 15/865,217, filed on Jan. 8, 2018, which claims the benefit of U.S. patent application Ser. No. 15/136,751, filed on Apr. 22, 2016, which claims the benefit of U.S. patent application Ser. No. 14/489,428, filed on Sep. 17, 2014, which claims the benefit of U.S. patent application Ser. No. 13/769,095, filed on Feb. 15, 2013, which claims the benefit of U.S. patent application Ser. No. 12/940,742, filed on Nov. 5, 2010, (U.S. Pat. No. 8,398,971) which claims the benefit of U.S. Provisional Patent Application Ser. No. 61/259,033, filed on Nov. 6, 2009, and U.S. Provisional Patent Application Ser. No. 61/259,055, filed on Nov. 6, 2009, U.S. Provisional Patent Application Ser. No. 61/322,813, filed on Apr. 9, 2010, U.S. Provisional Patent Application Ser. No. 61/332,063, filed on May 6, 2010, and U.S. Provisional Patent Application Ser. No. 61/381,420 filed on Sep. 9, 2010, the entire contents of each of which are hereby incorporated by reference.
BACKGROUND
[0003] Respiratory tract infections (RTIs) are among the most common, and potentially most severe, types of infectious diseases. Examples of RTIs include influenza, parainfluenza, RSV, sinusitis, otitis, laryngitis, bronchitis and pneumonia.
[0004] One common feature of agents that cause RTIs, such as respiratory pathogenic bacteria, is that they establish commensal colonization on the mucosal surface of the upper airway; such colonization precedes an infection and generally is prerequisite for infections. Bacterial colonization in a neonate occurs shortly after birth. During one's lifetime, the upper airway, specifically the nasopharynx and oropharynx, remains a dynamic ecological reservoir of microbial species with bacteria being acquired, eliminated and re-acquired continually. In most cases, the bacterial flora in the pharynx are harmless. However, when the condition of the host is altered, some microorganisms may invade adjacent tissues or bloodstream to cause diseases.
[0005] In addition to serving as the port of entry for mucosal and invasive infections by both bacteria and viruses, the nasopharynx and oropharynx are also the major source of spreading the pathogenic microorganisms between individuals, as well as the reservoir where antibiotic-resistant bacteria are selected (Garcia-Rodriguez and Martinez, J Antimicrob Chemother, (2002) 50(Suppl S2), 59-73; Soriano and Rodriguez-Cerrato, J Antimicrob Chemother, (2002) 50 Suppl S2, 51-58). It is well established clinically that individuals who are prone to RTIs tend to be persistent and recurrent carriers of pathogenic bacteria (Garcia-Rodriguez and Martinez, J Antimicrob Chemother, (2002) 50(Suppl S2), 59-73; Mbaki et al., Tohoku J Exp. Med., (1987) 153(2), 111-121). For example, Helicobacter pylori is a human pathogen implicated in gastritis and peptic ulcer. The bacterium resides in the human stomach and binds to epithelial cells of the gastric antrum.
[0006] Other disorders of the respiratory tract (more broadly termed, RTDs) may not be caused by infectious agents, although they could arise as a consequence of infection. Examples of RTDs include a variety of obstructive lung diseases such as allergic and non-allergic asthma, COPD, bronchiectasis, vasculitis, mucous plugging, Wegener's granulomatosis and cystic fibrosis (CF). RTDs can have a genetic basis (for example, CF), can arise due to immunodeficiencies, can arise due to other deficiencies (for example, alpha-1-antitrypsin deficiency can make people more susceptible to bronchiectasis), can be caused by allergens and/or chemical pollutants, or can present as complications of other infectious diseases such as the RTIs described above or inflammatory diseases such as inflammatory bowel syndrome or Crohn's disease.
[0007] Common indications of RTIs and RTDs include inflammation and elevated levels of mucous in the respiratory tract. However, currently available drugs that are used to treat RTIs and RTDs often are unable to ameliorate these associated conditions. For example, Relenza.RTM. is a well-known treatment for influenza, but it is not recommended for patients who suffer from underlying airway disease, such as asthma and COPD. Thus, in addition to the need for drugs that reduce inflammation and/or reduce mucus in the respiratory tract or limit its increase are drugs that are capable of treating respiratory infectious diseases, such as influenza, parainfluenza and RSV, without aggravating underlying respiratory conditions, such as asthma, bronchitis, bronchiectasis, and COPD, of patients.
[0008] The present invention recognizes that drugs currently available for medical use have limited efficacy with respect to reducing inflammation, and/or reducing mucus in the respiratory tract or limiting its increase in the respiratory tract, and those that are available are associated with side effects. The present invention also recognizes that there is a need for drugs for treating respiratory infectious diseases in patients with underlying airway disease, such as asthma, bronchitis, bronchiectasis and COPD. Thus, there is a need for new drugs that are able to reduce inflammation, and/or drugs that reduce mucus in the respiratory tract or limit its increase in the respiratory tract. There is also a need for drugs that can treat respiratory infectious diseases while reducing inflammation, and/or while reducing mucus in the respiratory tract or limiting its increase in the respiratory tract.
SUMMARY
[0009] The compositions, components of compostions and methods provided below are characterized by a variety of component ingredients, steps of preparation, and biophysical, physical, biochemical or chemical parameters. As would be apparent to those of skill in the art, the compositions and methods provided herein include any and all permutations and combinations of the ingredients, steps and/or parameters described below.
[0010] The invention relates to the use of therapeutic compounds and compositions that have anti-inflammatory effects in the respiratory tract and to methods of treating respiratory inflammation and prophylaxis against respiratory inflammation. The invention also relates to therapeutic compounds and compositions that can be used to prevent or treat diseases that are caused by, cause, or are exacerbated by respiratory inflammation, including, but not limited to, inflammation not caused by allergies or allergic reactions.
[0011] The invention also relates to the use of therapeutic compounds and compositions to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, and to corresponding methods of treatment. The invention also relates to the use of therapeutic compounds and compositions to limit an increase in the quantity of mucus in the respiratory tract of subjects above a baseline level of mucus in their respiratory tract and to corresponding methods of treatment. The invention also relates to therapeutic compounds and compositions that can be used to prevent or treat conditions and/or diseases that are caused by, cause, or are exacerbated by increased mucus in the respiratory tract, such as, both allergic and non-allergic asthma, chronic obstructive pulmonary disease (COPD), bronchitis (both acute and non-acute), bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancers involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infections, alpha 1-antitrypsin deficiency, primary immunodeficiencies, acquired immune deficiency syndrome, opportunistic infections, infectious and post infectious states, common cold, exercise-induced asthma, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, allergic reactions to inhaled fungus spores, respiratory infections, respiratory obstructions, inhalation or aspiration of ammonia and other toxic gases, pulmonary aspiration, alcoholism, various allergies, and any other disorder that causes increased mucus production in the respiratory tract or is caused by or exacerbated by increased mucus production in the respiratory tract. In some embodiments, the subject has more than one of the aforementioned conditions and/or diseases. In other embodiments, the subject having one or more of the aforementioned conditions and/or diseases does not have an accompanying infectious disease (RTI), such as influenza, parainfluenza or RSV. In other embodiments, the subject having one or more of the aforementioned conditions and/or diseases has one or more accompanying infectious diseases, such as influenza, parainfluenza or RSV. Thus, provided herein are methods, compounds and compositions for treating inflammatory and/or allergic responses associated with an RTI, an RTD, or combinations thereof.
[0012] The compounds and compositions provided herein can reduce mucus production in the respiratory tract and/or reduce the levels of inflammatory cells that cause allergic or non-allergic types of inflammation, including, without limitation, monocytes, macrophages, dendritic cells, histiocytes, Kuppfer cells, mastocytes and neutrophiles. The compounds and compositions provided herein include a sialidase or active portion thereof. Without being bound by any theory, sialic acids have been implicated in allergic and/or inflammatory responses associated with RTIs and RTDs. For example, siglecs (sialic acid binding Ig-like lectins) are members of the immunoglobulin (Ig) superfamily that bind to sialic acid and are mainly expressed by cells of the hematopoietic system. At least 11 siglecs have been discovered and they seem to exclusively recognize cell surface sialic acid as the ligand. It is believed that the binding of siglecs to sialic acid mediates cell-cell adhesion and interactions (Crocker and Varki, Trends Immunol., (2001) 22(6), 337-342; Angata and Brinkman-Van der Linden, Biochim. Biophys. Acta, (2002) 1572(2-3), 294-316). Siglec-8 (SAF-2) is an adhesion molecule that is highly restricted to the surface of eosinophils, basophils, and mast cells, which are the central effector cells in allergic conditions including allergic rhinitis, asthma and eczema. Siglec-8 (homologous to Siglec-F in mice) is considered to be responsible for mediating the recruitment of the three allergic cell types to the airway, the lungs and other sites of allergy. Siglec-1 (sialoadhesion) and siglec-2 (CD22) are the adhesion molecules on macrophages and B cells, both types of cells play central roles in immune reactions that lead to inflammation. Siglec-9 is predominantly expressed on neutrophils, which are known to be important effector cells in inflammation (von Gunten, Yousefi, Seitz, Jakob, Schaffner, Seger, Takala, Villiger, and Simon (2005) Blood 106:1423-1431). Further, without being bound by any particular theory, sialic acid residues have been implicated in the interaction of muscaranic receptors with agonists; thus, sialidases can affect the interecation of muscarinic receptors with their agonists.
[0013] The present invention provides a method of reducing the quantity of mucus in the respiratory tract of a subject with elevated levels of mucus in said respiratory tract. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a fusion protein having a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount includes an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the composition.
[0014] In another embodiment, another method of reducing the quantity of mucus in the respiratory tract of a subject with elevated levels of mucus in said respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a fusion protein. The fusion protein has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12, inclusive, and at least one anchoring domain. The anchoring domain can be a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7. The therapeutically effective amount includes an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the composition.
[0015] In another embodiment, another method of reducing the quantity of mucus in the respiratory tract of a subject with elevated levels of mucus in said respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a protein or peptide having a sialidase or an active portion thereof. The therapeutically effective amount includes an amount of the protein or peptide that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the compound or composition.
[0016] In another embodiment, a method of treating or ameliorating the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in a subject with an elevated level of mucus in his or her respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a fusion protein. The fusion protein has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12, inclusive, and at least one anchoring domain. The anchoring domain can be a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7. The therapeutically effective amount includes an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the compound or composition.
[0017] In another embodiment, another method of treating or ameliorating the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in a subject with an elevated level of mucus in his or her respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a fusion protein. The fusion protein has a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount includes an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the compound or composition.
[0018] In another embodiment, another method of treating or ameliorating the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in a subject with an elevated level of mucus in his or her respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a protein or peptide having a sialidase or an active portion thereof. The therapeutically effective amount includes an amount of the protein or peptide that results in a reduction of the quantity of mucus in the respiratory tract after administration of the compound or composition when compared to the quantity of mucus present prior to administration of the compound or composition.
[0019] In another embodiment, a method of limiting an increase in the quantity of mucus in the respiratory tract of a subject above a baseline level of mucus in said subject's respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a fusion protein. The fusion protein has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12, inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin comprising the amino acid sequence of SEQ ID NO:7. The therapeutically effective amount includes an amount of the fusion protein that limits an increase in the quantity of mucus in the respiratory tract of said subject above a baseline level after administration of the compound or composition.
[0020] In another embodiment, another method of limiting an increase in the quantity of mucus in the respiratory tract of a subject above a baseline level of mucus in said subject's respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a fusion protein having a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount includes an amount of the fusion protein that limits an increase in the quantity of mucus in the respiratory tract of said subject above a baseline level after administration of the compound or composition.
[0021] In yet another embodiment, another method of limiting an increase in the quantity of mucus in the respiratory tract of a subject above a baseline level of mucus in said subject's respiratory tract is provided. The method includes administering to the subject a compound or composition containing a therapeutically effective amount of a protein or peptide having a sialidase or an active portion thereof. The therapeutically effective amount includes an amount of the protein or peptide that limits an increase in the quantity of mucus in the respiratory tract of the subject above a baseline level after administration of the compound or composition.
[0022] Also contemplated herein are methods of identifying sialidases or active portions thereof according to the compounds or compositions provided herein, where the sialidases or active portions thereof are effective at reducing the quantity of mucus in the respiratory tract of subjects. The reduction in mucus can be measured directly in standard assays known to those of skill in the art. For example, in some embodiments, a single compound or a library or collection of compounds or compositions comprising sialidase(s) and/or catalytically active portion(s) thereof are administered to an animal model of asthma having an associated inflammatory response, such as the guinea pig and the mouse as described in Example 1 and Example 2, respectively. An asthmatic or other inflammatory condition is created in the animal whereby the accumulation of mucus in the lung or respitatory tract is increased. The level of mucus is then quantitated and compared to the level after treatment with a sialidase or active portion thereof. If there is a reduction of the mucus level in the presence of the sialidase or active portion thereof, the sialidase or active portion thereof is identified or selected as one that can be used in the methods provided herein for treating inflammation, allergies and/or associated inflammatory/allergic responses, such as the overproduction of mucus.
[0023] In some embodiments, a sialidase or active portion thereof according to the compounds and compositions provided herein is identified as being suitable for treating inflammation, allergies or associated responses by measuring its ability to disrupt muscarinic receptor-agonist interactions according to standard methods known to those of skill in the art. For example, provided herein is a method of assessing whether a compound or composition comprising a sialidase and/or catalytically active portion thereof reduces the quantity of mucus in the respiratory tract of a subject, by
[0024] (a) contacting the muscarinic receptors of an animal subject with a compound or composition that includes a sialidase and/or a catalytically active portion thereof;
[0025] (b) administering a muscarinic receptor agonist to the subject;
[0026] (c) quantitating the airway resistance in the subject;
[0027] (d) comparing the airway resistance level measured in (c) with the airway resistance in the absence of contact with the compound or composition;
[0028] (e) identifying whether the compound or composition reduces the airway resistance relative to the airway resistance in the absence of contact with the compound or composition; and
[0029] (f) if the compound or composition reduces the airway resistance as determined in (e), assessing the compound or composition as one that reduces the quantity of mucus in the respiratory tract of the subject. Such a method is exemplified in Example 3.
[0030] Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. Methods and materials are described herein for use in the present invention; other, suitable methods and materials known in the art can also be used. The materials, methods, and examples are illustrative only and not intended to be limiting. All publications, patent applications, patents, sequences, database entries, and other references mentioned herein are incorporated by reference in their entirety. In case of conflict, the present specification, including definitions, will control.
[0031] Other features and advantages of the invention will be apparent from the following detailed description and figures, and from the claims.
BRIEF DESCRIPTION OF THE DRAWINGS
[0032] FIG. 1 shows GAG-binding sequences of four human genes: PF4, human platelet factor 4; IL8, human interleukin 8; AT III, human antithrombin III; ApoE, human apolipoprotein E; AAMP, human angio-associated migratory cell protein; human amphiregulin.
[0033] FIG. 2 is a sequence comparison between human sialidases NEU2 and NEU4 (SEQ ID NOs: 8 & 9).
[0034] FIG. 3 is a table comparing substrate specificity of bacterial and fungal sialidases.
[0035] FIG. 4 depicts the nucleotide and amino acid sequence (SEQ ID NOs: 28 & 29) of a construct of the present invention encoding His6-AvCD. NcoI and HindIII sites used for cloning into pTrc99a are shown in bold.
[0036] FIG. 5 depicts the nucleotide and amino acid sequences (SEQ ID NOs: 18 & 19) of another construct of the present invention encoding AR-AvCD. NcoI and HindIII sites used for cloning into pTrc99a are shown in bold.
[0037] FIG. 6 depicts the nucleotide and amino acid sequences (SEQ ID NO: 36 & 37) of another construct of the present invention encoding AR-G.sub.4S-AvCD. NcoI and HindIII sites used for cloning into pTrc99a are shown in bold.
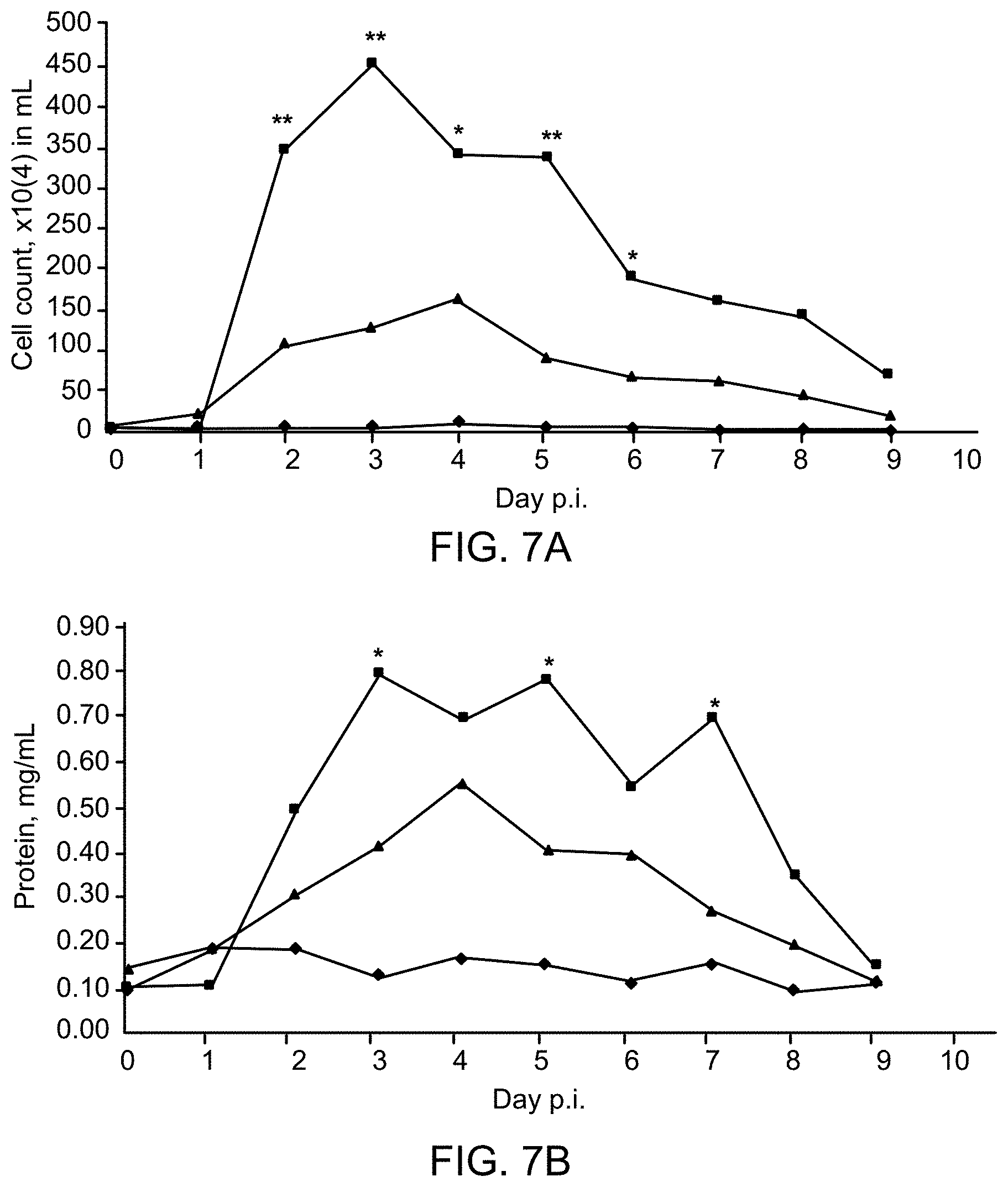
[0038] FIGS. 7A-B are graphs showing that topical administration of recombinant AR-AvCD sialidase fusion protein reduces the inflammatory responses of ferrets infected with an influenza A (H1N1) virus. FIG. 7A shows the total number of inflammatory cells from nasal wash samples obtained from infected animals at the indicated times after infection. he protein concentration was determined in cell-free nasal wash samples of infected ferrets. Infected ferrets were vehicle-treated (squares) or were treated with recombinant AR-AvCD sialidase fusion protein made from Construct #2 (triangles). Uninfected animals were also treated with recombinant AR-AvCD sialidase fusion protein (diamonds). Statistically significant values are labeled with * (p<0.05) and ** (p<0.01).
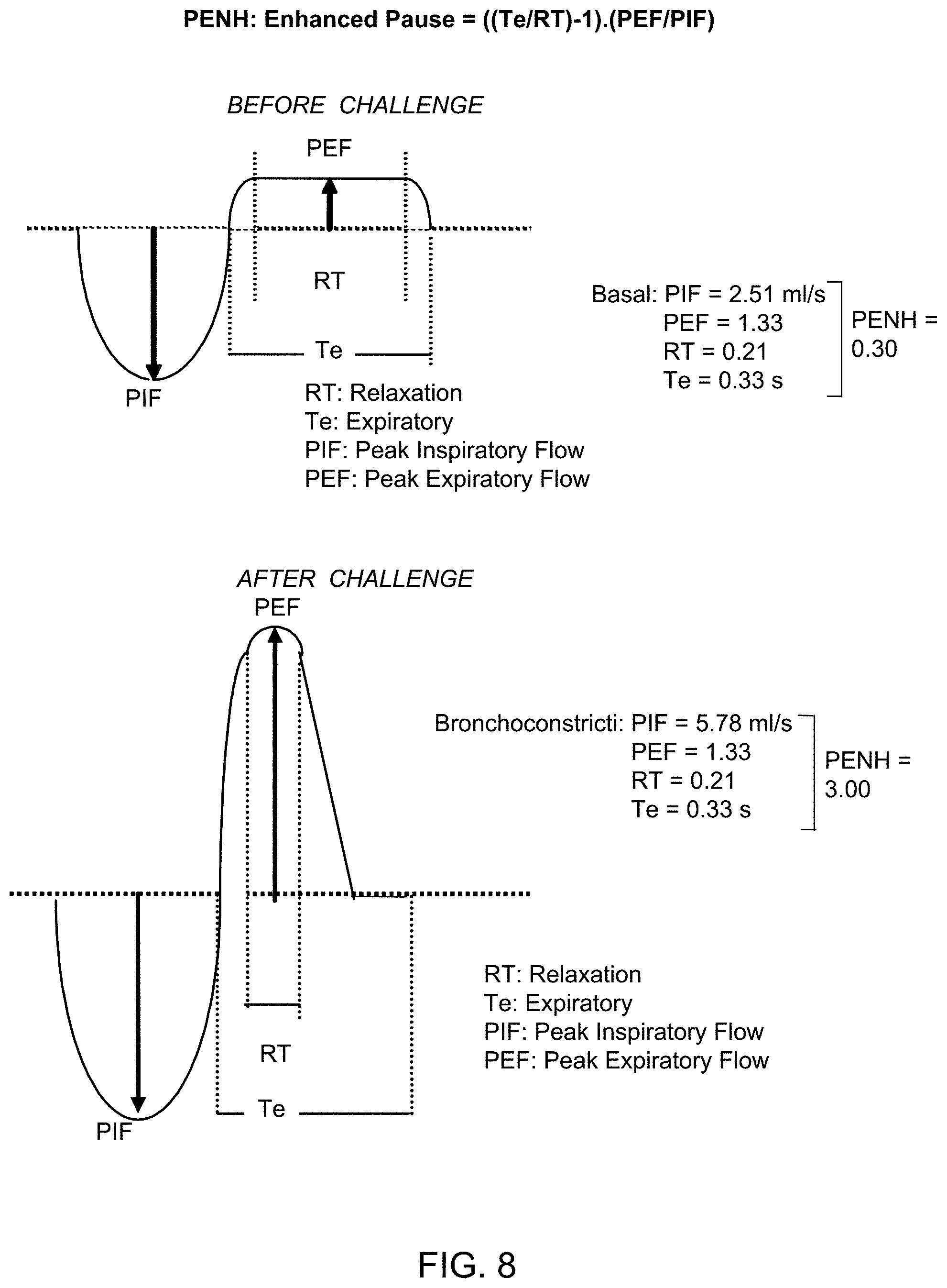
[0039] FIG. 8 provides graphs showing formula and explanation of the Enhanced Pause (PENH), the parameter used for measuring bronchoconstriction in conscious unrestrained animals.
[0040] FIG. 9 provides a graph showing early asthmatic reaction in response to an OVA-aerosol. Results are expressed as arithmetic average .+-.SEM. *p<0.05, ***p<0.001 using student's t-test.
[0041] FIG. 10 provides a graph showing the total number of cells in guinea pigs on the day of section. Results are expressed as arithmetic average .+-.SEM. **p<0.01, ***p<0.001 using student's t-test.
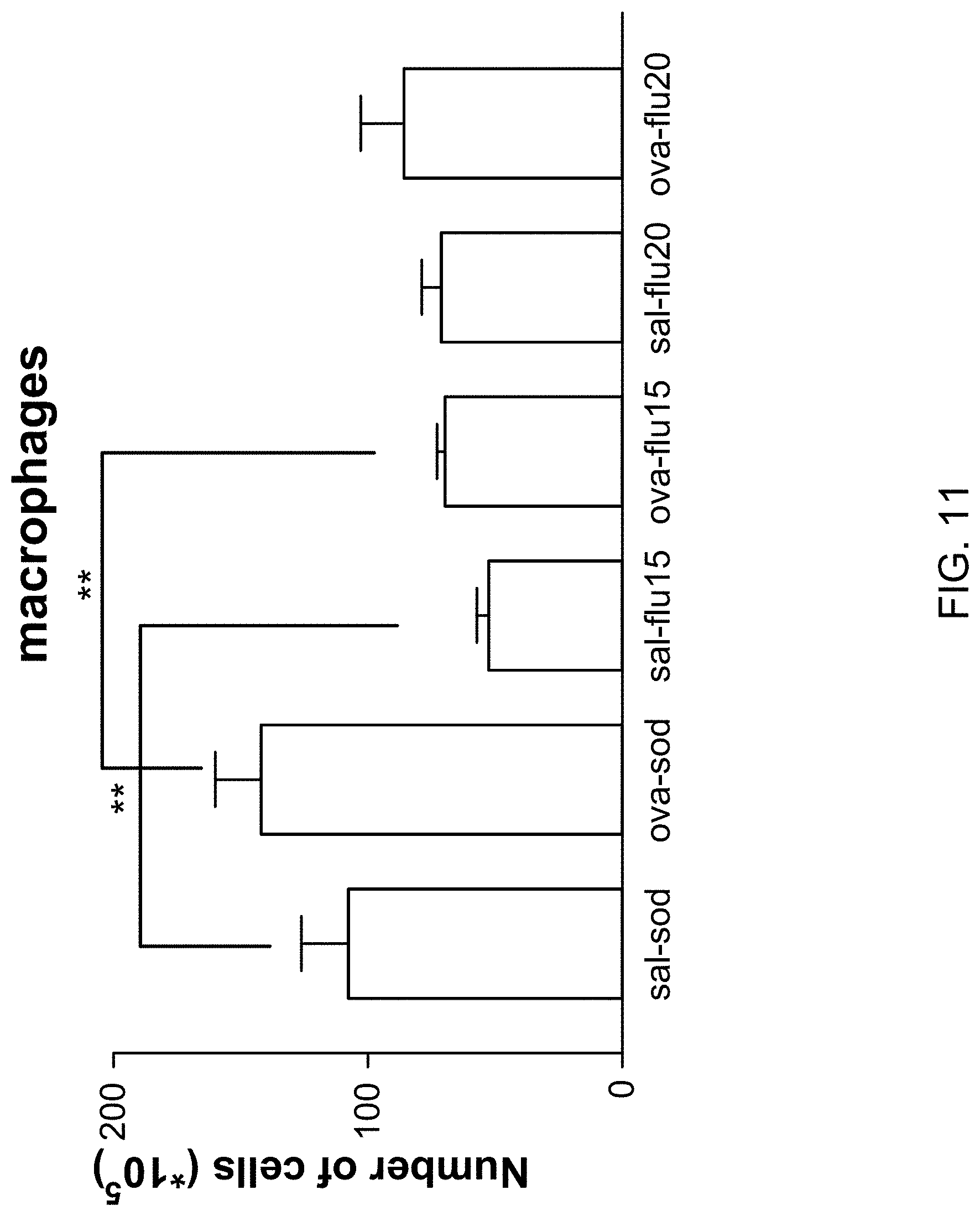
[0042] FIG. 11 provides a graph showing the total number of macrophages recovered in guinea pig BAL fluid on the day of section. Results are expressed as arithmetic average .+-.SEM. **p<0.01.
[0043] FIG. 12 provides a graph showing the total number of lymphocytes recovered in guinea pig BAL fluid on the day of section. Results are expressed as arithmetic average .+-.SEM. *p<0.05.
[0044] FIG. 13 provides a graph showing the total number of neutrophils recovered in guinea pig BAL fluid on the day of section. Results are expressed as arithmetic average .+-.SEM. *p<0.05, ***p<0.001.
[0045] FIG. 14 provides a graph showing the total number of eosinophils recovered in guinea pig BAL fluid on the day of section. Results are expressed as arithmetic average .+-.SEM. *p<0.05, ***p<0.001.
[0046] FIG. 15 provides a graph showing the percent change in Penh at Mch 48 mg/mL in the effect of sialidase treatment on the early and late asthmatic reaction in guinea pigs.
[0047] FIG. 16 provides a graph showing the percent change in Pehn at a range of Mch concentrations in the effect of sialidase treatment on the early and late asthmatic reaction in guinea pigs.
[0048] FIG. 17 provides a graph showing blood Eosinophils in the effect of sialidase treatment on the early and late asthmatic reaction in guinea pigs.
[0049] FIG. 18 provides a graph showing PAS staining for lung mucus in the effect of sialidase treatment on the early and late asthmatic reaction in guinea pigs.
[0050] FIGS. 19A-F provide a PAS staining for lung mucus.
[0051] FIG. 20 provides a graph showing MBP immunostaining for eosinophils in the effect of sialidase treatment on the early and late asthmatic reaction in guinea pigs.
[0052] FIGS. 21A-B provide graphs showing reduced airway resistance in naive mice treated intranasally with low doses of DAS181 (methacholine challenged).
[0053] FIG. 22 provides a graph showing reduced airway resistance in naive mice treated intranasally with a low dose of DAS181 (methacholine challenged).
[0054] FIG. 23 provides a graph showing reduced airway resistance in naive mice treated intranasally with DAS181 (carbachol challenged).
[0055] FIG. 24 provides a graph showing airway resistance in naive mice treated intranasally with a low dose of DAS185 (methacholine challenged).
[0056] FIG. 25 provides graphsshowing time-course of DAS185 mediated reduction of airway resistance (methacholine challenged).
[0057] FIG. 26 provides a graph showing reduced airway resistance in naive mice treated intranasally with very lose doses of DAS181 (methacholine challenged).
[0058] Like reference symbols in the various drawings indicate like elements.
DETAILED DESCRIPTION
[0059] The present disclosure provides, inter alia, novel methods of use for compounds described in U.S. patent application Ser. Nos. 10/718,986 and 10/939,262 (both of which are hereby incorporated by reference in their entirety) to reduce mucus, e.g., in the respiratory tract of subjects with elevated levels of mucus in their respiratory tract. In some embodiments, the present disclosure provides compositions and methods for reducing mucus (e.g., mucus levels) in a subject in need of reduced mucus levels and that does not have influenza (e.g., is not infected with influenza at the time of treatment) or ashma.
[0060] In some embodiments, the compounds can include compounds made by NexBio, Inc. under the compound name DAS181 and under the trademark Fludase.RTM. (provided herein as SEQ ID NO:21). DAS181 is a fusion protein comprising a catalytic domain of a sialidase, and an anchoring domain. Several of the examples described herein use DAS181 or compositions containing DAS181.
Definitions
[0061] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. Generally, the nomenclature used herein and the manufacture or laboratory procedures described below are well known and commonly employed in the art. Conventional methods are used for these procedures, such as those provided in the art and various general references. Where a term is provided in the singular, the inventors also contemplate the plural of that term. Where there are discrepancies in terms and definitions used in references that are incorporated by reference, the terms used in this application shall have the definitions given herein. As employed throughout the disclosure, the following terms, unless otherwise indicated, shall be understood to have the following meanings:
[0062] As used herein, a "subject" includes any animal for whom diagnosis, screening, monitoring or treatment is contemplated. Animals include mammals such as primates and domesticated animals. An exemplary primate is human. A patient refers to a subject such as a mammal, primate, human or livestock subject afflicted with a disease condition or for which a disease condition is to be determined or risk of a disease condition is to be determined.
[0063] In some embodiments, the methods disclosed herein can include selecting a a subject in need of reduced mucus levels and that is not infected with one or more of influenza, parainfluenza, and/or respiratory syncytial virus (RSV). In some instances, the terms infected or infection can include the presence of a influenza and/or parainfluenza virus and/or RSV in a subject. In some instances, the terms infected or infection can include the presence of active or replicating influenza and/or parainfluenza virus and/or RSV in a subject. In some embodiments, a subject with an active or replicating influenza and/or parainfluenza virus and/or RSV infection can be selected based on the presence or detection of influenza and/or parainfluenza virus shedding and/or RSV shedding in the subject (e.g., in a sample from the subject). In some embodiments, the methods disclosed herein can include selecting a subject in need of reduced mucus levels, wherein the subject has a latent influenza, parainfluenza, and/or RSV infection.
[0064] An "animal model" as used herein means an animal that sufficiently mimics, resembles or reproduces a disease or condition of interest in its anatomy, physiology, or response (to a pathogen or allergen, e.g.) so as to be useful in medical research that can be extrapolated to the disease or condition of interest (e.g., to screen for diagnostic or therapeutic agents; to measure therapeutic efficacy of a compound or composition, etc.). For example, the guinea pig and the mouse can be animal models to mimic inflammatory and/or allergic responses associated with asthma, as demonstrated in Examples 1 and 2, respectively. The mouse also can be an animal model to study the interaction of muscarinic receptors with their agonists, and the disruption thereof by agents such as the compounds and compositions provided herein (see Example 3).
[0065] A "pathogen" can be any virus or microorganism that can infect a cell, a tissue or an organism. A pathogen can be a virus, bacterium, or protozoan.
[0066] A "target cell" is any cell that can be infected by a pathogen or any cell that can interact with inflammatory cells, or a host cell that is the intended destination for an exogenous gene transferred by a recombinant virus.
[0067] "Inflammatory cells" are the cells that carry out or participate in inflammatory responses of the immune system. Inflammatory cells include include B lymphocytes, T lymphocytes, macrophages, basophils, eosinophils, mast cells, NK cells, monocytes, and neutrophils.
[0068] An "extracellular activity that can inhibit adhesion or function of inflammatory cells" is any activity that can prevent inflammatory cells from contacting the target cell and affecting the normal physiological status of the target cell.
[0069] A "domain that can anchor said at least one therapeutic domain to the membrane of a target cell", also called an "extracellular anchoring domain" or simply, "anchoring domain" refers to a chemical entity can that can stably bind a moiety that is at or on the exterior of a cell surface or is in close proximity to the surface of a cell. An extracellular anchoring domain can be reversibly or irreversibly linked to one or more moieties, such as one or more therapeutic domains, and thereby cause the one or more attached therapeutic moieties to be retained at or in close proximity to the exterior surface of a eukaryotic cell. An extracellular anchoring domain can bind at least one molecule on the surface of a target cell or at least one molecule found in close association with the surface of a target cell. For example, an extracellular anchoring domain can bind a molecule covalently or noncovalently associated with the cell membrane of a target cell, or can bind a molecule present in the extracellular matrix surrounding a target cell. An extracellular anchoring domain can be a peptide, polypeptide, or protein, and can also comprise any additional type of chemical entity, including one or more additional proteins, polypeptides, or peptides, a nucleic acid, peptide nucleic acid, nucleic acid analogue, nucleotide, nucleotide analogue, small organic molecule, polymer, lipids, steroid, fatty acid, carbohydrate, or a combination of any of these.
[0070] As used herein, a protein or peptide sequences is "substantially homologous" to a reference sequence when it is either identical to a reference sequence, or comprises one or more amino acid deletions, one or more additional amino acids, or more one or more conservative amino acid substitutions, and retains the same or essentially the same activity as the reference sequence. Conservative substitutions may be defined as exchanges within one of the following five groups: [0071] I. Small, aliphatic, nonpolar or slightly polar residues: Ala, Ser, Thr, Pro, Gly [0072] II. Polar, negatively charged residues and their amides: Asp, Asn, Glu, Gln [0073] III. Polar, positively charged residues: His, Arg, Lys [0074] IV. Large, aliphatic nonpolar residues: Met, Leu, Ile, Val, Cys [0075] V. Large aromatic residues: Phe, Try, Trp
[0076] Within the foregoing groups, the following substitutions are considered to be "highly conservative": Asp/Glu, His/Arg/Lys, Phe/Tyr/Trp, and Met/Leu/Ile/Val. Semi-conservative substitutions are defined to be exchanges between two of groups (I)-(IV) above which are limited to supergroup (A), comprising (I), (II), and (III) above, or to supergroup (B), comprising (IV) and (V) above. In addition, where hydrophobic amino acids are specified in the application, they refer to the amino acids Ala, Gly, Pro, Met, Leu, Ile, Val, Cys, Phe, and Trp, whereas hydrophilic amino acids refer to Ser, Thr, Asp, Asn, Glu, Gln, His, Arg, Lys, and Tyr.
[0077] A "sialidase" is an enzyme that can remove a sialic acid residue from a substrate molecule. The sialidases (N-acylneuraminosylglycohydrolases, EC 3.2.1.18) are a group of enzymes that hydrolytically remove sialic acid residues from sialo-glycoconjugates. Sialic acids are alpha-keto acids with 9-carbon backbones that are usually found at the outermost positions of the oligosaccharide chains that are attached to glycoproteins and glycolipids. One of the major types of sialic acids is N-acetylneuraminic acid (Neu5Ac), which is the biosynthetic precursor for most of the other types. The substrate molecule can be, as nonlimiting examples, an oligosaccharide, a polysaccharide, a glycoprotein, a ganglioside, or a synthetic molecule. For example, a sialidase can cleave bonds having alpha(2,3)-Gal, alpha(2,6)-Gal, or alpha(2,8)-Gal linkages between a sialic acid residue and the remainder of a substrate molecule. A sialidase can also cleave any or all of the linkages between the sialic acid residue and the remainder of the substrate molecule. Two major linkages between Neu5Ac and the penultimate galactose residues of carbohydrate side chains are found in nature, Neu5Ac alpha (2,3)-Gal and Neu5Ac alpha (2,6)-Gal. Both Neu5Ac alpha (2,3)-Gal and Neu5Ac alpha (2,6)-Gal molecules can be recognized by influenza viruses as the receptor, although human viruses seem to prefer Neu5Ac alpha (2,6)-Gal, avian and equine viruses predominantly recognize Neu5Ac alpha (2,3)-Gal. A sialidase can be a naturally-occurring sialidase, an engineered sialidase (such as, but not limited to a sialidase whose amino acid sequence is based on the sequence of a naturally-occurring sialidase, including a sequence that is substantially homologous to the sequence of a naturally-occurring sialidase). As used herein, "sialidase" can also mean the active portion of a naturally-occurring sialidase, or a peptide or protein that comprises sequences based on the active portion of a naturally-occurring sialidase.
[0078] A "fusion protein" is a protein comprising amino acid sequences from at least two different sources. A fusion protein can comprise amino acid sequence that is derived from a naturally occurring protein or is substantially homologous to all or a portion of a naturally occurring protein, and in addition can comprise from one to a very large number of amino acids that are derived from or substantially homologous to all or a portion of a different naturally occurring protein. In the alternative, a fusion protein can comprise amino acid sequence that is derived from a naturally occurring protein or is substantially homologous to all or a portion of a naturally occurring protein, and in addition can comprise from one to a very large number of amino acids that are synthetic sequences.
[0079] A "sialidase catalytic domain protein" is a protein that comprises the catalytic domain of a sialidase, or an amino acid sequence that is substantially homologous to the catalytic domain of a sialidase, but does not comprises the entire amino acid sequence of the sialidase the catalytic domain is derived from, wherein the sialidase catalytic domain protein retains substantially the same activity as the intact sialidase the catalytic domain is derived from. A sialidase catalytic domain protein can comprise amino acid sequences that are not derived from a sialidase, but this is not required. A sialidase catalytic domain protein can comprise amino acid sequences that are derived from or substantially homologous to amino acid sequences of one or more other known proteins, or can comprise one or more amino acids that are not derived from or substantially homologous to amino acid sequences of other known proteins.
[0080] "Therapeutically effective amount" means an amount of a composition or compound that is needed for a desired therapeutic, prophylactic, or other biological effect or response when a composition or compound is administered to a subject in a single dosage form. The particular amount of the composition or compound will vary widely according to conditions such as the nature of the composition or compound, the nature of the condition being treated, the age and size of the subject.
[0081] "Treatment" means any manner in which one or more of the symptoms of a condition, disorder or disease are ameliorated or otherwise beneficially altered. Treatment also encompasses any pharmaceutical use of the composition or compound herein, such as for reducing mucus in the respiratory tract.
[0082] "Respiratory tract" means the air passages from the nose to the pulmonary alveoli, including the nose, throat, pharynx, larynx, trachea, and bronchi, and it also includes the lungs, and is sometimes referred to by medical practitioners as the respiratory system.
[0083] "Inhaler" means a device for giving medicines in the form of a spray or dry powder that is inhaled (breathed in either naturally or mechanically forced in to the lungs) through the nose or mouth, and includes without limitation, a passive or active ventilator (mechanical with or with an endotracheal tube), nebulizer, dry powder inhaler, metered dose inhaler, and pressureized metered dose inhaler.
[0084] "Inhalant" is any substance that is inhaled through the nose or mouth.
[0085] "Reducing the quantity of mucus" means diminishing all or some, generally more than by 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 85%, 90% 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% or more of the amount of mucus in the respiratory tract when compared with the amount prior to administration of the compositions or compounds described herein. "Reducing the quantity of mucus" can also mean reducing the amount of mucus in an amount that is observable by a healthcare practitioner using whatever medical implements are available for such observation, such as, e.g., by auscultation, by MM or other radiographic study, by direct visualization with a bronchoscope or other visualization device, or by measuring patient mucus over time. "Reducing the quantity of mucus" can also mean reducing the amount of mucus in an amount that is observable by the patient or subject himself or herself with self-reporting or self-observation, such as, e.g., monitoring the amount of expectorated or swallowed mucus over time, or by subjectively observing the sense of congestion in his or her lungs over time.
[0086] "Limiting an increase in the quantity of mucus" means that the amount of mucus in the respiratory tract after administration of the compositions and compounds described herein does not increase more than if they had not been administered. "Limiting an increase in the quantity of mucus" also means that the amount of mucus in the respiratory tract after administration of the compositions and compounds described herein does not increase after their administration of the compositions and compounds. "Limiting an increase in the quantity of mucus" can also mean limiting an increase over the patient's baseline at the time of administration of the compounds or compositions in an amount that is observable or ascertainable by a healthcare practitioner using whatever medical implements and analytical systems are available for such observations, such as, e.g., by ausculation, by MM or other radiographic study, by direct visualization with a bronchoscope or other visualization device, or by measuring patient sputum over time. "Limiting an increase in the quantity of mucus" can also mean limiting an increase over the patient's baseline at the time of administration of the compounds or compositions in an amount that is observable by the patient or subject himself or herself with self-reporting or self-observation, such as, e.g., monitoring the amount of expectorated or swallowed mucus over time, or by subjectively observing the sense of congestion in his or her lungs over time.
[0087] "Excipient" as used herein means one or more inactive substances or compounds that either alone or in combination are used as a carrier for the active ingredients of a medication. As used herein "excipient" can also mean one or more substances or compounds that are included in a pharmaceutical composition to improve its beneficial effects or that have a synergistic effect with the active ingredient.
Peptide or Protein Based Compounds
[0088] The present invention includes peptide or protein-based compounds that comprise at least one domain that can anchor the compound to the membrane of a eukaryotic cell and at least one additional domain that is a therapeutic domain. By "peptide or protein-based" compounds, it is meant that the two major domains of the compound have an amino acid framework, in which the amino acids are joined by peptide bonds. A peptide or protein-based compound can also have other chemical compounds or groups attached to the amino acid framework or backbone, including moieties that contribute to the anchoring activity of the anchoring domain, or moieties that contribute to the therapeutic activity of the therapeutic domain. For example, the protein-based therapeutics used in the present invention can comprise compounds and molecules such as but not limited to: carbohydrates, fatty acids, lipids, steroids, nucleotides, nucleotide analogues, nucleic acid molecules, nucleic acid analogues, peptide nucleic acid molecules, small organic molecules, or even polymers. The protein-based therapeutics of the present invention can also comprise modified or non-naturally occurring amino acids. Non-amino acid portions of the compounds can serve any purpose, including but not limited to: facilitating the purification of the compound, improving the solubility or distribution or the compound (such as in a therapeutic formulation), linking domains of the compound or linking chemical moieties to the compound, contributing to the two-dimensional or three-dimensional structure of the compound, increasing the overall size of the compound, increasing the stability of the compound, and contributing to the anchoring activity or therapeutic activity of the compound.
[0089] The peptide or protein-based compounds of the present invention can also include protein or peptide sequences in addition to those that comprise anchoring domains or therapeutic domains. The additional protein sequences can serve any purpose, including but not limited to any of the purposes outlined above (facilitating the purification of the compound, improving the solubility or distribution or the compound, linking domains of the compound or linking chemical moieties to the compound, contributing to the two-dimensional or three-dimensional structure of the compound, increasing the overall size of the compound, increasing the stability of the compound, or contributing to the anchoring activity or therapeutic activity of the compound). Any additional protein or amino acid sequences can be part of a single polypeptide or protein chain that includes the anchoring domain or domains and therapeutic domain or domains, but any feasible arrangement of protein sequences is within the scope of the present invention.
[0090] The anchoring domain and therapeutic domain can be arranged in any appropriate way that allows the compound to bind at or near a target cell membrane. The compound can have at least one protein or peptide-based anchoring domain and at least one peptide or protein-based therapeutic domain. In this case, the domains can be arranged linearly along the peptide backbone in any order. The anchoring domain can be N-terminal to the therapeutic domain, or can be C-terminal to the therapeutic domain. It is also possible to have one or more therapeutic domains flanked by at least one anchoring domain on each end. Alternatively, one or more anchoring domains can be flanked by at least one therapeutic domain on each end. Chemical or peptide linkers can optionally be used to join some or all of the domains of a compound.
[0091] It is also possible to have the domains in a nonlinear, branched arrangement. For example, the therapeutic domain can be attached to a derivatized side chain of an amino acid that is part of a polypeptide chain that also includes, or is linked to, the anchoring domain.
[0092] A compound of the present invention can have more than one anchoring domain. In cases in which a compound has more than one anchoring domain, the anchoring domains can be the same or different. A compound used in the present invention can have more than one therapeutic domain. In cases in which a compound has more than one therapeutic domain, the therapeutic domains can be the same or different. Where a compound comprises multiple anchoring domains, the anchoring domains can be arranged in tandem (with or without linkers) or on alternate sides of other domains, such as therapeutic domains. Where a compound comprises multiple therapeutic domains, the therapeutic domains can be arranged in tandem (with or without linkers) or on alternate sides of other domains, such as, but not limited to, anchoring domains.
[0093] A peptide or protein-based compound of the present invention can be made by any appropriate way, including purifying naturally occurring proteins, optionally proteolytically cleaving the proteins to obtain the desired functional domains, and conjugating the functional domains to other functional domains. Peptides can also be chemically synthesized, and optionally chemically conjugated to other peptides or chemical moieties. A peptide or protein-based compound of the present invention can be made by engineering a nucleic acid construct to encode at least one anchoring domain and at least one therapeutic domain together (with or without nucleic acid linkers) in a continuous polypeptide. The nucleic acid constructs, in some embodiments having appropriate expression sequences, can be transfected into prokaryotic or eukaryotic cells, and the therapeutic protein-based compound can be expressed by the cells and purified. Any desired chemical moieties can optionally be conjugated to the peptide or protein-based compound after purification. In some cases, cell lines can be chosen for expressing the protein-based therapeutic for their ability to perform desirable post-translational modifications (such as, but not limited to glycosylation).
[0094] A great variety of constructs can be designed and their protein products tested for desirable activities (such as, for example, binding activity of an anchoring domain, or a binding, catalytic, or inhibitory activity of a therapeutic domain).
Anchoring Domain
[0095] As used herein, an "extracellular anchoring domain" or "anchoring domain" is any moiety that can stably bind an entity that is at or on the exterior surface of a target cell or is in close proximity to the exterior surface of a target cell. An anchoring domain serves to retain a compound used in the present invention at or near the external surface of a target cell.
[0096] An extracellular anchoring domain can bind 1) a molecule expressed on the surface of a target cell, or a moiety, domain, or epitope of a molecule expressed on the surface of a target cell, 2) a chemical entity attached to a molecule expressed on the surface of a target cell, or 3) a molecule of the extracellular matrix surrounding a target cell.
[0097] An anchoring domain can be a peptide or protein domain (including a modified or derivatized peptide or protein domain), or comprises a moiety coupled to a peptide or protein. A moiety coupled to a peptide or protein can be any type of molecule that can contribute to the binding of the anchoring domain to an entity at or near the target cell surface, and in some embodiments is an organic molecule, such as, for example, nucleic acid, peptide nucleic acid, nucleic acid analogue, nucleotide, nucleotide analogue, small organic molecule, polymer, lipids, steroid, fatty acid, carbohydrate, or any combination of any of these.
[0098] A molecule, complex, domain, or epitope that is bound by an anchoring domain may or may not be specific for the target cell. For example, an anchoring domain may bind an epitope present on molecules on or in close proximity to the target cell and that occur at sites other than the vicinity of the target cell as well. In many cases, however, localized delivery of a therapeutic compound of the present invention will restrict its occurrence primarily to the surface of target cells. In other cases, a molecule, complex, moiety, domain, or epitope bound by an anchoring domain may be specific to a target tissue or target cell type.
[0099] Target tissue or target cell type includes the sites in an animal or human body where a pathogen invades or amplifies. For example, a target cell can be an endothelial cell that can be infected by a pathogen. A composition used in the present invention can comprise an anchoring domain that can bind a cell surface epitope, for example, that is specific for the endothelial cell type. In another example, a target cell can be an epithelial cell and a composition of the present invention can bind an epitope present on the cell surface of many epithelial cell types, or present in the extracellular matrix of different types of epithelial cells. In this case localized delivery of the composition can restrict its localization to the site of the epithelial cells that are targets of the pathogen.
[0100] Compounds used in the present invention can have one or more anchoring domains that can bind at or near the surface of epithelial cells. For example, heparan sulfate, closely related to heparin, is a type of glycosaminoglycan (GAG) that is ubiquitously present on cell membranes, including the surface of respiratory epithelium. Many proteins specifically bind to heparin/heparan sulfate, and the GAG-binding sequences in these proteins have been identified (Meyer, F A, King, M and Gelman, R A. (1975) Biochimica et Biophysica Acta 392: 223-232; Schauer, S. ed., pp 233. Sialic Acids Chemistry, Metabolism and Function. Springer-Verlag, 1982). For example, the GAG-binding sequences of human platelet factor 4 (PF4) (SEQ ID NO:2), human interleukin 8 (IL8) (SEQ ID NO:3), human antithrombin III (AT III) (SEQ ID NO:4), human apoprotein E (ApoE) (SEQ ID NO:5), human angio-associated migratory cell protein (AAMP) (SEQ ID NO:6), or human amphiregulin (SEQ ID NO:7) (FIG. 1) have been shown to have very high affinity (in the nanomolar range) towards heparin (Lee, M K and Lander, A D. (1991) Pro Natl Acad Sci USA 88:2768-2772; Goger, B, Halden, Y, Rek, A, Mosl, R, Pye, D. Gallagher, J and Kungl, A J. (2002) Biochem. 41:1640-1646; Witt, D P and Lander A D (1994) Curr Bio 4:394-400; Weisgraber, K H, Rall, S C, Mahley, R W, Milne, R W and Marcel, Y. (1986) J Bio Chem 261:2068-2076). The GAG-binding sequences of these proteins are distinct from their receptor-binding sequences, so they will not induce the biological activities associated with the full-length proteins or the receptor-binding domains. These sequences, or other sequences that have been identified or are identified in the future as heparin/heparan sulfate binding sequences, or sequences substantially homologous to identified heparin/heparan sulfate binding sequences that have heparin/heparan sulfate binding activity, can be used as epithelium-anchoring-domains in compounds used in the present invention.
[0101] An anchoring domain can bind a moiety that is specific to the target cell type of a particular species or can bind a moiety that is found in the target cell type of more than one species.
Therapeutic Domain
[0102] A compound used in the present invention includes at least one therapeutic domain or active portion, those terms being used interchangeable herein. The therapeutic activity can be, as nonlimiting examples, a binding activity, a catalytic activity, or an inhibitory activity. A therapeutic domain can modify or inhibit a function of the target cell or target organism. An active portion of a compound has therapeutic activity. For example, the catalytic domain or active portion of a sialidase can be its therapeutic domain.
[0103] The therapeutic domain can act extracellularly, meaning that its infection-preventing, inflammatory response-modulating, or transduction-enhancing activity takes place at the target cell surface or in the immediate area surrounding the target cell, including sites within the extracellular matrix, intracellular spaces, or luminal spaces of tissues.
[0104] A therapeutic domain can be a peptide or protein domain (including a modified or derivatized peptide or protein domain), or comprises a moiety coupled to a peptide or protein. A moiety coupled to a peptide or protein can be any type of molecule, and is in some embodiments an organic molecule, such as, for example, nucleic acid, peptide nucleic acid, nucleic acid analogue, nucleotide, nucleotide analogue, small organic molecule, polymer, lipids, steroid, fatty acid, carbohydrate, or any combination of any of these.
[0105] A therapeutic domain can be a synthetic peptide or polypeptide, or can comprise a synthetic molecule that can be conjugated to a peptide or polypeptide, can be a naturally-occurring peptide or protein, or a domain of naturally-occurring protein. A therapeutic domain can also be a peptide or protein that is substantially homologous to a naturally-occurring peptide or protein.
Linkers
[0106] A compound used in the present invention can optionally include one or more linkers that can join domains of the compound. Linkers can be used to provide optimal spacing or folding of the domains of a compound. The domains of a compound joined by linkers can be therapeutic domains, anchoring domains, or any other domains or moieties of the compound that provide additional functions such as enhancing compound stability, facilitating purification, etc. A linker used to join domains of compounds of the present invention can be a chemical linker or an amino acid or peptide linker. Where a compound comprises more than one linker, the linkers can be the same or different. Where a compound comprises more than one linker, the linkers can be of the same or different lengths.
[0107] Many chemical linkers of various compositions, polarity, reactivity, length, flexibility, and cleavability are known in the art of organic chemistry. Preferred linkers include amino acid or peptide linkers. Peptide linkers are well known in the art. Some embodiments of linkers are between one and about one hundred amino acids in length, and between one and about thirty amino acids in length, although length is not a limitation in the linkers of the compounds of the present invention. The linkder amino acid sequences can be selected such that they do not interfere with the mucus-reducing and/or anti-inflammatory activity of the compounds and compositions used in the present invention. Some embodiments of linkers are those that include the amino acid glycine. For example, linkers having the sequence:
(GGGGS (SEQ ID NO:10))n, where n is a whole number between 1 and 20, or between 1 and 12, can be used to link domains of therapeutic compounds used in the present invention.
Composition Comprising at Least One Anchoring Domain and at Least One Catalytic Activity
[0108] In some aspects, the present invention can use compounds that have a therapeutic domain that has an enzymatic activity. The enzymatic activity can be a catalytic activity that removes, degrades or modifies a host molecule or complex. In some embodiments the host molecule or complex can be removed, degraded, or modified by the enzymatic activity of a compound of the present invention is on, at, or near the surface of a target cell.
[0109] Compounds used in the present invention can have, for example, one of the following structures: [0110] (Anchoring Domain)n-[linker]-(Enzymatic Activity)n (n=1,2, 3 or more) or: [0111] (Enzymatic Activity)n (n=1,2, 3 or more)-[linker]-(Anchoring Domain)n, [0112] where the linkers are optional.
[0113] The enzymatic activity can be a monomeric form of a peptide or polypeptide or can be multiple copies of the same polypeptide that are either linked directly or with spacing sequence in between. The polypeptides or peptides can be linked directly or via a spacer composed of peptide linker sequence. The anchoring domain can be any peptide or polypeptide that can bind to or near the surface of target cells.
[0114] In one embodiment, a therapeutic domain comprises a sialidase that can eliminate or greatly reduce the level of sialic acid on the surface of epithelial cells. The therapeutic domain can comprise a complete sialidase protein, or an active portion thereof, wherein the active portion thereof retains the ability to perform the catalytic function(s) of the sialidase protein (e.g., cleaving sialic acid residues).
[0115] Sialic acid mediates cell adhesion and interactions between inflammatory cells and target cells. Therefore, treating the surface of respiratory epithelial cells with a sialidase can prevent the recruitment of inflammatory cells to the airway surface, and therefore can treat allergic reactions including asthma and allergic rhinitis. It also unexpectedly results in reducing the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tract, and limiting increase in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in the respiratory tract of those subjects.
[0116] Among the sialidases contemplated for use in the methods described herein are the large bacterial sialidases that can degrade the receptor sialic acids Neu5Ac alpha(2,6)-Gal and Neu5Ac alpha(2,3)-Gal. For example, the bacterial sialidase enzymes from Clostridium perfringens (Genbank Accession Number X87369), Actinomyces viscosus, Arthrobacter ureafaciens, or Micromonospora viridifaciens (Genbank Accession Number D01045) can be used. Therapeutic domains of compounds of the present invention can comprise all or a portion of the amino acid sequence of a large bacterial sialidase or can comprise amino acid sequences that are substantially homologous to all or a portion of the amino acid sequence of a large bacterial sialidase. In one preferred embodiment, a therapeutic domain comprises a sialidase encoded by Actinomyces viscosus, such as that of SEQ ID NO:12, or such as sialidase sequence substantially homologous to SEQ ID NO:12. In yet another preferred embodiment, a therapeutic domain comprises the catalytic domain of the Actinomyces viscosus sialidase extending from amino acids 274-667 of SEQ ID NO:12, or a substantially homologous sequence.
[0117] Other sialidases contemplated for use in the methods described herein are the human sialidases such as those encoded by the genes NEU2 (SEQ ID NO:8; Genbank Accession Number Y16535; Monti, E, Preti, Rossi, E., Ballabio, A and Borsani G. (1999) Genomics 57:137-143) and NEU4 (SEQ ID NO:9; Genbank Accession Number NM080741; Monti, E, Preti, A, Venerando, B and Borsani, G. (2002) Neurochem Res 27:646-663) (FIG. 2). Therapeutic domains of compounds used in the present invention can comprise all or a portion of the amino acid sequences of a human sialidase or can comprise amino acid sequences that are substantially homologous to all or a portion of the amino acid sequences of a human sialidase. Where a therapeutic domain comprises a portion of the amino acid sequences of a naturally occurring sialidase, or sequences substantially homologous to a portion of the amino acid sequences of a naturally occurring sialidase, the portion can have essentially the same activity as the human sialidase.
[0118] A compound for reducing elevated levels of mucus in the respiratory tract can in some embodiments have one or anchoring domains that can bind at or near the surface of epithelial cells. In some embodiments, the epithelium-anchoring domain is a GAG-binding sequence from a human protein, such as, for example, the GAG-binding amino acid sequences of human platelet factor 4 (PF4) (SEQ ID NO:2), human interleukin 8 (IL8) (SEQ ID NO:3), human antithrombin III (AT III) (SEQ ID NO:4), human apoprotein E (ApoE) (SEQ ID NO:5), human angio-associated migratory cell protein (AAMP) (SEQ ID NO:6), and human amphiregulin (SEQ ID NO:7) (FIG. 1). An epithelial anchoring domain can also be substantially homologous to a naturally occurring GAG-binding sequence, such as those listed in FIG. 1. Such compounds can be formulated for nasal, tracheal, bronchial, oral, or topical administration, or can be formulated as an injectable solution or as eyedrops, or formulated into a solution or dry powder and inhaled with inhalers.
[0119] A pharmaceutical composition comprising such compounds can be used to treat or prevent allergy or inflammatory response. In addition, such compounds have been shown herein to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, and to limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts. Therefore, such compounds can be used to as therapeutic treatments to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, or as prophylactic treatments to limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts. Due to their effect on mucus in the respiratory tract, these compounds can also be used to prevent, treat, or ameliorate the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in subjects with elevated levels of mucus in their respiratory tract or who are at risk of having increased levels of mucus in their respiratory tract.
[0120] It is also within the scope of the present invention to use compounds or compositions comprising a human sialidase, such as any of those described herein, or an active portion thereof, or a compound with substantial homology to a sialidase, in the absence of an anchoring domain (a) to treat or prevent allergic and inflammatory responses in the respiratory tract, (b) to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, (c) to limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts, and/or (d) to prevent, treat, or ameliorate the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in subjects with elevated levels of mucus in their respiratory tract or who are at risk of having increased levels of mucus in their respiratory tract. The present invention recognizes that elevated levels of mucus in the respiratory tract can be reduced by the use of a sialidase or an active portion of a sialidase, and that such sialidases or active portions thereof can optionally be adapted, by genetic or chemical engineering, or by pharmaceutical formulation, to improve their half life or retention at the respiratory epithelium.
[0121] These compounds and pharmaceutical compositions can be delivered to the upper respiratory tract as a nasal spray, or delivered to the respiratory tract as an inhalant with inhalers.
[0122] The compounds described herein can be formulated into pharmaceutical compositions that include various additional compounds either alone or in various combinations, such as, Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs. These additional compounds can be included in the pharmaceutical compositions to act as excipients or as active ingredients that provide additional beneficial effects.
Therapeutic Composition Comprising at Least One Sialidase Activity
[0123] The present invention includes methods that use therapeutic compounds and compositions that comprise at least one sialidase activity. The sialidase activity can be a sialidase isolated from any source, such as, for example, a bacterial or mammalian source, or can be a recombinant protein that is substantially homologous to at least a portion of a naturally occurring sialidase. In some embodiments sialidases are the large bacterial sialidases that can degrade the receptor sialic acids Neu5Ac alpha(2,6)-Gal and Neu5Ac alpha(2,3)-Gal. For example, the bacterial sialidase enzymes from Clostridium perfringens (Genbank Accession Number X87369), Actinomyces viscosus (Genbank Accession Number L06898), Arthrobacter ureafaciens, or Micromonospora viridifaciens (Genbank Accession Number D01045) or substantially homologous proteins can be used.
[0124] For example, therapeutic compounds and compositions used in the present invention can comprise a large bacterial sialidase or can comprise a protein with the amino acid sequence of a large bacterial sialidase or can comprise amino acid sequences that are substantially homologous to the amino acid sequence of a large bacterial sialidase. A pharmaceutical composition that can be used in the present invention comprises the A. viscosus sialidase (SEQ ID NO:12), or comprises a protein substantially homologous to the A. viscosus sialidase.
[0125] Other sialidases that can be used in the compositions, compounds and methods described herein are the human sialidases such as those encoded by the genes NEU2 (SEQ ID NO:8; Genbank Accession Number Y16535; Monti, E, Preti, Rossi, E., Ballabio, A and Borsani G. (1999) Genomics 57:137-143) and NEU4 (SEQ ID NO:9; Genbank Accession Number NM080741; Monti, E, Preti, A, Venerando, B and Borsani, G. (2002) Neurochem Res 27:646-663) (FIG. 2). Therapeutic domains of compounds of the present invention can comprise a human sialidase protein that is substantially homologous to the amino acid sequences of a human sialidase or can comprise amino acid sequences that are substantially homologous to all or a portion of the amino acid sequences of a human sialidase. Where a therapeutic domain comprises a portion of the amino acid sequences of a naturally occurring sialidase, or sequences substantially homologous to a portion of the amino acid sequences of a naturally occurring sialidase, the portion can have essentially the same activity as the human sialidase, e.g., an active portion of the sialidase.
[0126] Generally, sialidases that can effectively degrade on respiratory epithelial cells both receptor sialic acids Neu5Ac a(2,6)-Gal and Neu5Ac a(2,3)-Gal, can be used. Sialidases are found in higher eukaryotes, as well as in some mostly pathogenic microbes, including viruses, bacteria and protozoans. Viral and bacterial sialidases have been well characterized, and the three-dimensional structures of some of them have been determined (Crennell, S J, Garman, E, Laver, G, Vimr, E and Taylor, G. (1994) Structure 2:535-544; Janakiraman, M N, White, C L, Laver, W G, Air, G M and Luo, M. (1994) Biochemistry 33:8172-8179; Pshezhetsky, A, Richard, C, Michaud, L, Igdoura, S, Wang, S, Elsliger, M, Qu, J, Leclerc, D, Gravel, R, Dallaire, L and Potier, M. (1997) Nature Genet 15: 316-320). Several human sialidases have also been cloned in the recent years (Milner, C M, Smith, S V, Carrillo M B, Taylor, G L, Hollinshead, M and Campbell, R D. (1997) J Bio Chem 272:4549-4558; Monti, E, Preti, A, Nesti, C, Ballabio, A and Borsani G. 1999. Glycobiol 9:1313-1321; Wada, T, Yoshikawa, Y, Tokuyama, S, Kuwabara, M, Akita, H and Miyagi, T. (1999) Biochem Biophy Res Communi 261:21-27; Monti, E, Bassi, M T, Papini, N, Riboni, M, Manzoni, M, Veneranodo, B, Croci, G, Preti, A, Ballabio, A, Tettamanti, G and Borsani, G. (2000) Bichem J 349:343-351). DAS181, which contains an active portion of a sialidase, has also been cloned.
[0127] All the sialidases characterized share a four amino acid motif in the amino terminal portion followed by the Asp box motif which is repeated three to five times depending on the protein. (Monti, E, Bassi, M T, Papini, N, Riboni, M, Manzoni, M, Veneranodo, B, Croci, G, Preti, A, Ballabio, A, Tettamanti, G and Borsani, G. (2000) Bichem J349:343-351; Copley, R R, Russell, R B and Ponting, C P. (2001) Protein Sci 10:285-292). While the overall amino acid identity of the sialidase superfamily is relatively low at about 20-30%, the overall fold of the molecules, especially the catalytic amino acids, are remarkably similar (Wada, T, Yoshikawa, Y, Tokuyama, S, Kuwabara, M, Akita, H and Miyagi, T. (1999) Biochem Biophy Res Communi 261:21-27; Monti, E, Bassi, M T, Papini, N, Riboni, M, Manzoni, M, Veneranodo, B, Croci, G, Preti, A, Ballabio, A, Tettamanti, G and Borsani, G. (2000) Bichem J 349:343-351; Copley, R R, Russell, R B and Ponting, C P. (2001) Protein Sci 10:285-292).
[0128] The sialidases are generally divided into two families: "small" sialidases have molecular weight of about 42 kDa and do not require divalent metal ion for maximal activity; "large" sialidases have molecular weight above 65 kDa and may require divalent metal ion for activity (Wada, T, Yoshikawa, Y, Tokuyama, S, Kuwabara, M, Akita, H and Miyagi, T. (1999) Biochem Biophy Res Communi 261:21-27; Monti, E, Bassi, M T, Papini, N, Riboni, M, Manzoni, M, Veneranodo, B, Croci, G, Preti, A, Ballabio, A, Tettamanti, G and Borsani, G. (2000) Bichem J 349:343-351; Copley, R R, Russell, R B and Ponting, C P. (2001) Protein Sci 10:285-292).
[0129] Over fifteen sialidase proteins have been purified and they vary greatly from one another in substrate specificities and enzymatic kinetics. Large bacterial sialidases can effectively cleave sialic acid in both (.alpha.,2-6) linkage and (.alpha.,2-3) linkage in the context of most natural substrates (FIG. 4; Vimr, D R. (1994) Trends Microbiol 2: 271-277; Drzeniek, R. (1973) Histochem J5:271-290; Roggentin, P, Kleineidam, R G and Schauer, R. (1995) Biol Chem Hoppe-Seyler 376:569-575; Roggentin, P, Schauer, R, Hoyer, L L and Vimr, E R. (1993) Mol Microb 9:915-921). Because of their broad substrate specificities, large bacterial sialidases make good candidates.
[0130] FIG. 4 shows several of the large bacterial sialidases with known substrate specificity. These enzymes have high specific activity (600 U/mg protein for C. perfringens (Corfield, A P, Veh, R W, Wember, M, Michalski, J C and Schauer, R. (1981) Bichem J 197:293-299) and 680 U/mg protein for A. viscosus (Teufel, M, Roggentin, P. and Schauer, R. (1989) Biol Chem Hoppe Seyler 370:435-443)), are fully active without divalent metal iron, and have been cloned and purified as recombinant proteins from E. coli (Roggentin, P, Kleineidam, R G and Schauer, R. (1995) Biol Chem Hoppe-Seyler 376:569-575, Teufel, M, Roggentin, P. and Schauer, R. (1989) Biol Chem Hoppe Seyler 370:435-443, Sakurada, K, Ohta, T and Hasegawa, M. (1992) J Bacteriol 174: 6896-6903). In addition, C. perfringens is stable in solution at 2-8.degree. C. for several weeks, and at 4.degree. C. in the presence of albumin for more than two years (Wang, F Z, Akula, S M, Pramod, N P, Zeng, L and Chandran, B. (2001) J Virol 75:7517-27). A. viscosus is labile towards freezing and thawing, but is stable at 4.degree. C. in 0.1 M acetate buffer, pH 5 (Teufel, M, Roggentin, P. and Schauer, R. (1989) Biol Chem Hoppe Seyler 370:435-443).
[0131] A pharmaceutical composition comprising a sialidase can include other compounds, including but not limited to other proteins, that can also have therapeutic activity. A pharmaceutical composition comprising a sialidase can include other compounds that can enhance the stability, solubility, packaging, delivery, consistency, taste, or fragrance of the composition.
[0132] Compounds comprising a sialidase can be formulated for nasal, tracheal, bronchial, oral, or topical administration, or can be formulated as an injectable solution or as eyedrops, or formulated into a solution or dry powder and inhaled with inhalers. The sialidases described herein can be formulated into pharmaceutical compositions that include various additional compounds such as, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs.
[0133] These sialidases or pharmaceutical compositions containing them can be used (a) to treat or prevent allergic and inflammatory responses in the respiratory tract, (b) to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, (c) to limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts, and/or (d) to prevent, treat, or ameliorate the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in subjects with elevated levels of mucus in their respiratory tract or who are at risk of having increased levels of mucus in their respiratory tract. In some embodiments, subjects with elevated levels of mucus in their respiratory tract do not include subjects with one or more of influenza, parainfluenza, and/or respiratory syncytial virus (RSV).
Sialidase Catalytic Domain Proteins or Peptides
[0134] As used herein a "sialidase catalytic domain protein or peptide" comprises a catalytic domain of a sialidase but does not comprise the entire amino acid sequence of the sialidase from which the catalytic domain is derived. A sialidase catalytic domain protein or peptide has sialidase activity. A sialidase catalytic domain protein or peptide can have at least 10%, at least 20%, at least 50%, at least 70% of the activity of the sialidase from which the catalytic domain sequence is derived. A sialidase catalytic domain protein or peptide can have at least 90% of the activity of the sialidase from which the catalytic domain sequence is derived.
[0135] A sialidase catalytic domain protein or peptide can include other amino acid sequences, such as but not limited to additional sialidase sequences, sequences derived from other proteins, or sequences that are not derived from sequences of naturally-occurring proteins. Additional amino acid sequences can perform any of a number of functions, including contributing other activities to the catalytic domain protein, enhancing the expression, processing, folding, or stability of the sialidase catalytic domain protein, or even providing a desirable size or spacing of the protein or peptide.
[0136] A preferred sialidase catalytic domain protein or peptide is a protein that comprises the catalytic domain of the A. viscosus sialidase. An A. viscosus sialidase catalytic domain protein or peptide can include amino acids 270-667 of the A. viscosus sialidase sequence (SEQ ID NO:12). An A. viscosus sialidase catalytic domain protein or peptide can include amino acid sequence that begins at any of the amino acids from amino acid 270 to amino acid 290 of the A. viscosus sialidase sequence (SEQ ID NO:12) and ends at any of the amino acids from amino acid 665 to amino acid 901 of said A. viscosus sialidase sequence (SEQ ID NO:12), and lacks any A. viscosus sialidase protein sequence extending from amino acid 1 to amino acid 269. (As used herein "lacks any A. viscosus sialidase protein sequence extending from amino acid 1 to amino acid 269" means lacks any stretch of four or more consecutive amino acids as they appear in the designated protein or amino acid sequence.)
[0137] In some embodiments, an A. viscosus sialidase catalytic domain protein or peptide comprises amino acids 274-681 of the A. viscosus sialidase sequence (SEQ ID NO:12) and lacks other A. viscosus sialidase sequence. In other embodiments, an A. viscosus sialidase catalytic domain protein comprises amino acids 290-666 or 290-667 of the A. viscosus sialidase sequence (SEQ ID NO:12) and lacks any other A. viscosus sialidase sequence. In yet other embodiments, an A. viscosus sialidase catalytic domain protein or peptide comprises amino acids 274-666 of the A. viscosus sialidase sequence (SEQ ID NO:12) and lacks any other A. viscosus sialidase sequence. In yet other embodiments, an A. viscosus sialidase catalytic domain protein or peptide comprises amino acids 290-666 or 290-667 of the A. viscosus sialidase sequence (SEQ ID NO:12) and lacks any other A. viscosus sialidase sequence. In yet other embodiments, an A. viscosus sialidase catalytic domain protein or peptide comprises amino acids 290-681 of the A. viscosus sialidase sequence (SEQ ID NO:12) and lacks any other A. viscosus sialidase sequence.
[0138] Such sialidase catalytic domain proteins or peptides can be formulated for nasal, tracheal, bronchial, oral, or topical administration, or can be formulated as an injectable solution or as eyedrops, or formulated into a solution or dry powder and inhaled with an inhaler. The sialidase catalytic domain proteins or peptides described herein can be formulated into pharmaceutical compositions that include various additional compounds, such as, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs. These additional compounds can be included in the pharmaceutical compositions either alone or in various combinations, such as, Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs. These additional compounds can be included in the pharmaceutical compositions to act as excipients or as active ingredients that provide additional beneficial effects.
[0139] Such sialidase catalytic domain proteins or peptides or pharmaceutical compositions containing them can be used (a) to treat or prevent allergic and inflammatory responses in the respiratory tract, (b) to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, (c) to limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts, and/or (d) to prevent, treat, or ameliorate the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in subjects with elevated levels of mucus in their respiratory tract or who are at risk of having increased levels of mucus in their respiratory tract.
Fusion Proteins
[0140] Sialidase catalytic domain proteins can be fusion proteins, in which the fusion protein comprises at least one sialidase catalytic domain and at least one other protein domain, including but not limited to: a purification domain, a protein tag, a protein stability domain, a solubility domain, a protein size-increasing domain, a protein folding domain, a protein localization domain, an anchoring domain, an N-terminal domain, a C-terminal domain, a catalytic activity domain, a binding domain, or a catalytic activity-enhancing domain. The at least one other protein domain can be derived from another source, such as, but not limited to, sequences from another protein. The at least one other protein domain need not be based on any known protein sequence, but can be engineered and empirically tested to perform any function in the fusion protein.
[0141] Purification domains can include, as nonlimiting examples, one or more of a his tag, a calmodulin binding domain, a maltose binding protein domain, a streptavidin domain, a streptavidin binding domain, an intein domain, or a chitin binding domain. Protein tags can comprise sequences that can be used for antibody detection of proteins, such as, for example, the myc tag, the hemagglutinin tag, or the FLAG tag. Protein domains that enhance protein expression, modification, folding, stability, size, or localization can be based on sequences of know proteins or engineered. Other protein domains can have binding or catalytic activity or enhance the catalytic activity of the sialidase catalytic domain.
[0142] Fusion proteins used in the compositions, compounds and methods of the present invention comprise at least one sialidase catalytic domain and at least one anchoring domain. In some embodiments, anchoring domains include GAG-binding domains, such as the GAG-binding domain or human amphiregulin (SEQ ID NO:7).
[0143] Sialidase catalytic domains and other domains of a fusion protein used in the present invention can optionally be joined by linkers, such as but not limited to peptide linkers. A variety of peptide linkers are known in the art. In one embodiment a linker can be a peptide linker comprising glycine, such as G-G-G-G-S(SEQ ID NO:10).
[0144] Such fusion proteins can be formulated for nasal, tracheal, bronchial, oral, or topical administration, or can be formulated as an injectable solution or as eyedrops or formulated into a solution or dry powder and inhaled with an inhaler. These fusion proteins can be formulated into pharmaceutical compositions that include various additional compounds either alone or in various combinations, such as, Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs. These additional compounds can be included in the pharmaceutical compositions to act as excipients or as active ingredients that provide additional beneficial effects.
[0145] Such fusion proteins or pharmaceutical compositions containing them can be used (a) to treat or prevent allergic and inflammatory responses in the respiratory tract, (b) to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, (c) to limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts, and/or (d) to prevent, treat, or ameliorate the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in subjects with elevated levels of mucus in their respiratory tract or who are at risk of having increased levels of mucus in their respiratory tract.
[0146] Various constructs of fusion proteins are shown in FIGS. 4-6, as well as in the sequences provided in the sequence listing provided herein.
Methods for Testing the Compounds and Compositions and/or for Screening to Identify Sialidases and/or Active Portions Thereof to Treat Diseases Accompanied by Inflammation
[0147] The compounds and compositions provided herein can be tested for their activity in reducing inflammation, allergies or associated responses, such as mucus overproduction, using standard assays known to those of skill in the art. Several cell-based (e.g., tracheal cell cultures) and animal-based assays (mouse models, guinea pig models) for measuring inflammation or mucus overproduction are known (see, e.g., Nakao et al., J. Immunol., 180:6262-6269 (2008); Westerhof et al., Mediators Inflamm., 10(3):143-154 (2001); Miller et al., J. Immunol., 170:3348-3356 (2003); Nakanishi et al., Proc. Natl. Acad. Sci. USA, 98(9):5175-5180 (2001); and DuBuske, Allergy Proc., 16(2):55-58 (1995), the contents of each of which are incorporated in their entirety by reference herein). The compounds and compositions provided herein can be tested for their ability to reduce inflammation or mucus overproduction in any of these assays or other standard assays known to those of skill in the art. In addition, sialidases or active portions thereof can be identified and/or selected for their anti-inflammatory activity and/or ability to reduce associated responses, such as mucus overproduction, using such assays. Exemplary assays and protocols are described herein in Example 1 and Example 2.
[0148] In addition to assays that measure inflammation or associated responses, such as mucus overproduction, the compounds and compositions provided herein can be tested for their activity by assessing their ability to disrupt muscarinic receptor--mediated signaling in the presence of an agonist. Muscarinic receptors, or mAChRs, are G protein-coupled acetylcholine receptors found in the plasma membranes of certain neurons- and other cells. They play several roles, including acting as the main end-receptor stimulated by acetylcholine released from postganglionic fibers in the parasympathetic nervous system.
[0149] Muscarinic receptor-agonist interactions, and the resulting signaling, is believed to play a role in diseases that have associated inflammatory and/or allergic responses, such as asthma and COPD (see, e.g., "Muscarinic Receptors in Airways Diseases," Birkhauser-Verlag publ., Zangsma et al., Eds.).
[0150] More specifically, acetylcholinergic mechanisms are recognized to influence the following normal and pathogenic respiratory functions: [0151] 1. secretion of mucus, [0152] 2. active transport of ions across the respiratory epithelium and during mucociliary transport, [0153] 3. smooth muscle tone of the airways, [0154] 4. immunologic and inflammatory response of the airways, [0155] 5. reflex regulation of the airways, [0156] 6. respiratory responses of the airways in asthma and in other hypersensitivity states of the respiratory tract.
[0157] Consequently, certain anti-muscarinic agents have been effective against: (a) acetylcholinergically induced bronchoconstriction; (b) iatrogenic airway spasms induced by beta blockers; and (c) psychogenic bronchospasm. The two main pulmonary applications of anti-muscarinic agents has been chronic bronchitis and bronchial asthma (Pharmacology of Anti-Muscarinic Agents, Laszlo Gyermek (1998)).
[0158] There are five broad classes of muscarinic receptors, based on their physiological roles, and agonists for each of these receptors are known to those of skill in the art:
[0159] M1 receptor--exemplary agonists include acetylcholine, oxotremorine, muscarine, carbachol and McNA343
[0160] M2 receptor--exemplary agonists include acetylcholine, methacholine, carbachol, oxotremorine and muscarine
[0161] M3 receptor--exemplary agonists include acetylcholine, bethanechol, carbachol, oxotremorine and pilocarpine
[0162] M4 receptor--exemplary agonists include acetylcholine, carbachol and oxotremorine
[0163] M5 receptor--exemplary agonists include acetylcholine, carbachol and oxotremorine
[0164] In some embodiments, the compounds and compositions provided herein can be tested for the ability to reduce inflammation and/or allergic responses, including mucus overproduction, associated with RTIs or RTDs by assessing their ability to disrupt muscarinic receptor-agonist interactions. Further, sialidases and/or active portions thereof can be screened, identified and selected for their ability to reduce inflammation, allergies, and/or associated responses such as mucus overproduction by assessing their ability to disrupt muscarinic receptor--agonist interactions. These tests and screens can be performed using standard assays known to those of skill in the art (see, e.g, Armstrong et al., Curr. Protocols in Pharmacol., UNIT 12-13 (2010), the contents of which are incorporated in their entirety by reference herein). An exemplary assay and protocol is provided herein in Example 3.
Pharmaceutical Compositions
[0165] The present invention includes compounds of the present invention formulated as pharmaceutical compositions. The pharmaceutical compositions comprise a pharmaceutically acceptable carrier prepared for storage and subsequent administration, which have a pharmaceutically effective amount of the compound in a pharmaceutically acceptable carrier or diluent. Acceptable carriers or diluents for therapeutic use are well known in the pharmaceutical art, and are described, for example, in Remington's Pharmaceutical Sciences, 18th Ed., Mack Publishing Co., Easton, Pa. (1990)). Preservatives, stabilizers, dyes and even flavoring agents can be provided in the pharmaceutical composition. For example, sodium benzoate, sorbic acid and esters of p-hydroxybenzoic acid can be added as preservatives. In addition, antioxidants and suspending agents can be used.
[0166] Depending on the target cell, the compounds of the present invention can be formulated and used as tablets, capsules or elixirs for oral or inhaled administration; salves or ointments for topical application; suppositories for rectal administration; sterile solutions, suspensions, and encapsulated powders and the like for use as inhalants or nasal sprays. Injectables can also be prepared in conventional forms either as liquid solutions or suspensions, solid forms suitable for solution or suspension in liquid prior to injection, or as emulsions.
[0167] Suitable excipients are, for example, water, saline, dextrose, mannitol, lactose, lecithin, albumin, sodium glutamate, cysteine hydrochloride and the like. In addition to those excipients, additional compounds that can be included in the pharmaceutical compositions described herein either alone or in various combinations include Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs or Mg salts and/or Ca salts. These additional compounds can be included in the pharmaceutical compositions to act as excipients or as active ingredients that provide additional beneficial effects. In addition, if desired, the injectable pharmaceutical compositions can contain minor amounts of nontoxic auxiliary substances, such as wetting agents, pH buffering agents and the like.
[0168] The pharmaceutically effective amount of a test compound required as a dose will depend on the route of administration, the type of animal or patient being treated, and the physical characteristics of the specific animal under consideration. The dose can be tailored to achieve a desired effect, such as reduction of elevated levels of mucus in the respiratory tract, but will depend on such factors as weight, diet, concurrent medication and other factors which those skilled in the medical arts will recognize. In practicing the methods of the present invention, the pharmaceutical compositions can be used alone or in combination with one another, or in combination with other therapeutic or diagnostic agents. These products can be utilized in vivo in a non-human animal subject, in a mammalian subject, in a human subject, or in vitro. In employing them in vivo, the pharmaceutical compositions can be administered to the patient or subject in a variety of ways, including topically, parenterally, intravenously, subcutaneously, intramuscularly, colonically, rectally, nasally or intraperitoneally, employing a variety of dosage forms. Such methods can also be used in testing the activity of test compounds in vivo.
[0169] In some embodiments, these pharmaceutical compositions may be in the form of orally-administrable suspensions, solutions, tablets or lozenges; nasal sprays; inhalants; injectables, topical sprays, ointments, powders, or gels, or formulated into a solutions or dry powders and inhaled with an inhaler.
[0170] When administered orally as a suspension, compositions of the present invention are prepared according to techniques well-known in the art of pharmaceutical formulation and may contain microcrystalline cellulose for imparting bulk, alginic acid or sodium alginate as a suspending agent, methylcellulose as a viscosity enhancer, and sweeteners/flavoring agents known in the art. As immediate release tablets, these compositions may contain microcrystalline cellulose, dicalcium phosphate, starch, magnesium stearate and lactose and/or other excipients, binders, extenders, disintegrants, diluents and lubricants known in the art. Components in the formulation of a mouthwash or rinse include antimicrobials, surfactants, cosurfactants, oils, water and other additives such as sweeteners/flavoring agents known in the art.
[0171] When administered by a drinking solution, the composition comprises one or more of the compounds of the present invention, dissolved in water, with appropriate pH adjustment, and with carrier. The compound can be dissolved in distilled water, tap water, spring water, and the like. The pH can in some embodiments be adjusted to between about 3.5 and about 8.5. Sweeteners can be added, e.g., 1% (w/v) sucrose.
[0172] Lozenges can be prepared according to U.S. Pat. No. 3,439,089, herein incorporated by reference for these purposes.
[0173] When administered by nasal aerosol or inhalation, the pharmaceutical compositions are prepared according to techniques well-known in the art of pharmaceutical formulation and can be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art. See, for example, Ansel, H. C. et al., Pharmaceutical Dosage Forms and Drug Delivery Systems, Sixth Ed. (1995). Inhaled powders can also be prepared using techniques described in U.S. patent application Ser. Nos. 11/657,813 and 12/179,520, both of which are incorporated herein by reference in their entirety. These compositions and formulations can generally be prepared with suitable nontoxic pharmaceutically acceptable ingredients. These ingredients are known to those skilled in the preparation of nasal dosage forms and some of these can be found in Remington's Pharmaceutical Sciences, 18th Ed., Mack Publishing Co., Easton, Pa. (1990, a standard reference in the field. The choice of suitable carriers is highly dependent upon the exact nature of the nasal dosage form desired, e.g., solutions, suspensions, ointments, or gels. Nasal dosage forms generally contain large amounts of water in addition to the active ingredient. Minor amounts of other ingredients such as pH adjusters, emulsifiers or dispersing agents, preservatives, surfactants, jelling agents, or buffering and other stabilizing and solubilizing agents can also be present. Generally, the nasal dosage form can be isotonic with nasal secretions.
[0174] Nasal formulations can be administers as drops, sprays, aerosols or by any other intranasal dosage form. Optionally, the delivery system can be a unit dose delivery system. The volume of solution or suspension delivered per dose can be anywhere from about 5 to about 2000 microliters, from about 10 to about 1000 microliters, or from about 50 to about 500 microliters. Delivery systems for these various dosage forms can be dropper bottles, plastic squeeze units, atomizers, nebulizers or pharmaceutical aerosols in either unit dose or multiple dose packages.
[0175] The formulations of this invention can be varied to include; (1) other acids and bases to adjust the pH; (2) other tonicity imparting agents such as sorbitol, glycerin and dextrose; (3) other antimicrobial preservatives such as other parahydroxy benzoic acid esters, sorbate, benzoate, propionate, chlorbutanol, phenylethyl alcohol, benzalkonium chloride, and mercurials; (4) other viscosity imparting agents such as sodium carboxymethylcellulose, microcrystalline cellulose, polyvinylpyrrolidone, polyvinyl alcohol and other gums; (5) suitable absorption enhancers; (6) stabilizing agents such as antioxidants, like bisulfite and ascorbate, metal chelating agents such as sodium edetate and drug solubility enhancers such as polyethylene glycols.
[0176] One embodiment of the invention includes pharmaceutical compositions that at various dosage levels, such as dosage levels between about 0.01 mg and about 100 mg, reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts, and/or that limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts. Examples of such dosage levels include doses of about 0.05 mg, 0.06 mg, 0.1 mg, 0.5 mg, 1 mg, 5 mg, 10 mg, 20 mg, 50 mg, or 100 mg. Another embodiment of the invention includes pharameutical compositions that at various dosage levels, such as dosage levels between about 0.01 mg and about 100 mg, reduce inflammation in the respiratory tract or prevent worsening of inflammation in the respiratory tract. Examples of such dosage levels include doses of about 0.05 mg, 0.06 mg, 0.1 mg, 0.5 mg, 1 mg, 5 mg, 10 mg, 20 mg, 50 mg, or 100 mg. The foregoing doses can be administered one or more times per day, for one day, two days, three days, four days, five days, six days, seven days, eight days, nine days, ten days, eleven days, twelve days, thirteen days, or fourteen or more days. Higher doses or lower doses can also be administered. Typically, dosages can be between about 1 ng/kg and about 10 mg/kg, between about 10 ng/kg and about 1 mg/kg, and between about 100 ng/kg and about 100 micrograms/kg. In various examples described herein, mice were treated with various dosages of the compositions described herein, including dosages of 0.0008 mg/kg, 0.004 mg/kg, 0.02 mg/kg, 0.06 mg/kg, 0.1 mg/kg, 0.3 mg/kg, 0.6 mg/kg, and 1.0 gm/kg.
[0177] In one embodiment a pharmaceutical composition includes DAS181, MgSO.sub.4 1.446 mg/ml, CaCl.sub.2) 0.059 mg/ml, Histidine 1.427 mg/ml, Histidine-HCl 1.943 mg/ml, and Trehalose 3.000 mg/ml.
[0178] In another embodiment a pharameutical composition includes DAS181, MgSO.sub.4, CaCl.sub.2), Histidine, Histidine-HCl, and Trehalose.
[0179] In another embodiment a pharameutical composition includes DAS181, Na.sub.2SO.sub.4, and CaCl.sub.2).
[0180] In another embodiment a pharmaceutical composition includes DAS181 and any combination of one or more of the following: Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histidine-HCl, and Trehalose.
[0181] In another embodiment a pharmaceutical composition includes (a) a naturally occurring sialidase protein or peptide or an active portion thereof, or a recombinant protein substantially homologous to at least a portion of a naturally occurring sialidase, (b) MgSO.sub.4 1.446 mg/ml, (c) CaCl.sub.2) 0.059 mg/ml, (d) Histidine 1.427 mg/ml, (e) Histidine-HCl 1.943 mg/ml, and (f) Trehalose 3.000 mg/ml. In one embodiment, the protein or peptide is a sialidase with substantial homology to the A. viscosus sialidase (SEQ ID NO:12) or substantial homology to an active portion thereof, such as amino acids 274-666, 274-667, 270-667, 274-681, or 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis sialidase. In other embodiments, the protein or peptide is from one of the large bacterial sialidases that can degrade the receptor sialic acids Neu5Ac alpha(2,6)-Gal and Neu5Ac alpha(2,3)-Gal. For example, the bacterial sialidase enzymes from Clostridium perfringens (Genbank Accession Number X87369), Arthrobacter ureafaciens, or Micromonospora viridifaciens (Genbank Accession Number D01045) or proteins or peptides that are substantially homologous to those sialidases or their active portions. In other embodiments, the protein or peptide is from other sialidases, such as those encoded by the genes NEU2 (SEQ ID NO:8; Genbank Accession Number Y16535; Monti, E, Preti, Rossi, E., Ballabio, A and Borsani G. (1999) Genomics 57:137-143) and NEU4 (SEQ ID NO:9; Genbank Accession Number NM080741; Monti, E, Preti, A, Venerando, B and Borsani, G. (2002) Neurochem Res 27:646-663) (FIG. 2), or active portions of those sialidases.
[0182] In another embodiment a pharmaceutical composition includes (a) a naturally occurring sialidase protein or peptide or an active portion thereof, or a recombinant protein substantially homologous to at least a portion of a naturally occurring sialidase, (b) MgSO.sub.4, (c) CaCl.sub.2), (d) Histidine, (e) Histidine-HCl, and (f) Trehalose. In one embodiment, the protein or peptide is a sialidase with substantial homology to the A. viscosus sialidase (SEQ ID NO:12) or substantial homology to an active portion thereof, such as amino acids 274-666, 274-667, 270-667, 274-681, or 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis sialidase. In other embodiments, the protein or peptide is from one of the large bacterial sialidases that can degrade the receptor sialic acids Neu5Ac alpha(2,6)-Gal and Neu5Ac alpha(2,3)-Gal. For example, the bacterial sialidase enzymes from Clostridium perfringens (Genbank Accession Number X87369), Arthrobacter ureafaciens, or Micromonospora viridifaciens (Genbank Accession Number D01045) or proteins or peptides that are substantially homologous to those sialidases or their active portions. In other embodiments, the protein or peptide is from other sialidases, such as those encoded by the genes NEU2 (SEQ ID NO:8; Genbank Accession Number Y16535; Monti, E, Preti, Rossi, E., Ballabio, A and Borsani G. (1999) Genomics 57:137-143) and NEU4 (SEQ ID NO:9; Genbank Accession Number NM080741; Monti, E, Preti, A, Venerando, B and Borsani, G. (2002) Neurochem Res 27:646-663) (FIG. 2), or active portions of those sialidases.
[0183] In another embodiment a pharmaceutical composition includes (a) a naturally occurring sialidase protein or peptide or an active portion thereof, or a recombinant protein substantially homologous to at least a portion of a naturally occurring sialidase, (b) Na.sub.2SO.sub.4, and (c) CaCl.sub.2). In one embodiment, the protein or peptide is a sialidase with substantial homology to the A. viscosus sialidase (SEQ ID NO:12) or substantial homology to an active portion thereof, such as amino acids 274-666, 274-667, 270-667, 274-681, or 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis sialidase. In one embodiment, the protein or peptide is a sialidase with substantial homology to the A. viscosus sialidase (SEQ ID NO:12) or substantial homology to an active portion thereof, such as amino acids 274-666, 274-667, 270-667, 274-681, or 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis sialidase. In other embodiments, the protein or peptide is from one of the large bacterial sialidases that can degrade the receptor sialic acids Neu5Ac alpha(2,6)-Gal and Neu5Ac alpha(2,3)-Gal. For example, the bacterial sialidase enzymes from Clostridium perfringens (Genbank Accession Number X87369), Arthrobacter ureafaciens, or Micromonospora viridifaciens (Genbank Accession Number D01045) or proteins or peptides that are substantially homologous to those sialidases or their active portions. In other embodiments, the protein or peptide is from other sialidases, such as those encoded by the genes NEU2 (SEQ ID NO:8; Genbank Accession Number Y16535; Monti, E, Preti, Rossi, E., Ballabio, A and Borsani G. (1999) Genomics 57:137-143) and NEU4 (SEQ ID NO:9; Genbank Accession Number NM080741; Monti, E, Preti, A, Venerando, B and Borsani, G. (2002) Neurochem Res 27:646-663) (FIG. 2), or active portions of those sialidases.
[0184] In another embodiment a pharmaceutical composition includes (a) a naturally occurring sialidase protein or peptide or an active portion thereof, or a recombinant protein substantially homologous to at least a portion of a naturally occurring sialidase, and any combination of one or more of the following: Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histidine-HCl, and Trehalose. In one embodiment, the protein or peptide is a sialidase with substantial homology to the A. viscosus sialidase (SEQ ID NO:12) or substantial homology to an active portion thereof, such as amino acids 274-666, 274-667, 270-667, 274-681, or 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis sialidase. In other embodiments, the protein or peptide is from one of the large bacterial sialidases that can degrade the receptor sialic acids Neu5Ac alpha(2,6)-Gal and Neu5Ac alpha(2,3)-Gal. For example, the bacterial sialidase enzymes from Clostridium perfringens (Genbank Accession Number X87369), Arthrobacter ureafaciens, or Micromonospora viridifaciens (Genbank Accession Number D01045) or proteins or peptides that are substantially homologous to those sialidases or their active portions. In other embodiments, the protein or peptide is from other sialidases, such as those encoded by the genes NEU2 (SEQ ID NO:8; Genbank Accession Number Y16535; Monti, E,
[0185] Preti, Rossi, E., Ballabio, A and Borsani G. (1999) Genomics 57:137-143) and NEU4 (SEQ ID NO:9; Genbank Accession Number NM080741; Monti, E, Preti, A, Venerando, B and Borsani, G. (2002) Neurochem Res 27:646-663) (FIG. 2), or active portions of those sialidases.
[0186] In another embodiment a pharmaceutical composition includes (a) a fusion protein that has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7, (b) MgSO.sub.4 1.446 mg/ml, (c) CaCl.sub.2) 0.059 mg/ml, (d) Histidine 1.427 mg/ml, (e) Histidine-HCl 1.943 mg/ml, and (f) Trehalose 3.000 mg/ml.
[0187] In another embodiment a pharmaceutical composition includes (a) a fusion protein that has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7, (b) MgSO.sub.4, (c) CaCl.sub.2), (d) Histidine, (e) Histidine-HCl, and (f) Trehalose.
[0188] In another embodiment a pharmaceutical composition includes (a) a fusion protein that has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7, (b) Na.sub.2SO.sub.4, and (c) CaCl.sub.2).
[0189] In another embodiment a pharmaceutical composition includes (a) a fusion protein that has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7, and (b) any combination of one or more of the following: Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histidine-HCl, and Trehalose.
[0190] In another embodiment a pharmaceutical composition includes (a) a fusion protein having a sialidase or an active portion thereof and an anchoring domain, (b) MgSO.sub.4 1.446 mg/ml, (c) CaCl.sub.2) 0.059 mg/ml, (d) Histidine 1.427 mg/ml, (e) Histidine-HCl 1.943 mg/ml, and (f) Trehalose 3.000 mg/ml.
[0191] In another embodiment a pharmaceutical composition includes (a) a fusion protein having a sialidase or an active portion thereof and an anchoring domain, (b) MgSO.sub.4, (c) CaCl.sub.2), (d) Histidine, (e) Histidine-HCl, and (f) Trehalose.
[0192] In another embodiment a pharmaceutical composition includes (a) a fusion protein having a sialidase or an active portion thereof and an anchoring domain, (b) Na.sub.4SO.sub.4, and (c) CaCl.sub.2).
[0193] In another embodiment a pharmaceutical composition includes (a) a fusion protein having a sialidase or an active portion thereof and an anchoring domain, and (b) any combination of one or more of the following: Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histidine-HCl, and Trehalose.
[0194] Another representative example of a pharmaceutical composition of the present invention and that can be used in the methods described herein includes the following: DAS181, histidine, magnesium sulfate (or citrate salt), calcium chloride, trehalose, water, Na-Acetate, and acetic acid.
[0195] Yet another representative example of a pharmaceutical composition of the present invention and that can be used in the methods described herein includes DAS181 (in any concentration between about 0.01% and about 100% w/w, between about 1.00% and about 90.0% w/w, between about 5.00% and about 80.0% w/w, between about 10.0% and about 70.0% w/w, between about 20.0% and about 70% w/w, between about 30.0% and about 70.0% w/w, between about 40.0% and about 70.0% w/w, between about 50.0% and about 70% w/w, between about 60.0% and about 70.0% w/w) in combination with any of the following: histidine or histidine-HCl (in any concentration between about 0.00% and about 90.0% w/w, between about 0.01% and about 80.0% w/w, between about 1.00% and about 75.0% w/w, between about 2.00% and about 70.0% w/w, between about 3.00% and about 60% w/w, between about 4.00% and about 50.0% w/w, between about 5.00% and about 40.0% w/w, between about 6.00% and about 30% w/w, between about 7.00% and about 20.0% w/w), magnesium sulfate (or citrate salt or sodium sulfate)(in any concentration between about 0.00% and about 90.0% w/w, between about 0.01% and about 80.0% w/w, between about 1.00% and about 75.0% w/w, between about 2.00% and about 70.0% w/w, between about 3.00% and about 60% w/w, between about 4.00% and about 50.0% w/w, between about 5.00% and about 40.0% w/w, between about 6.00% and about 30% w/w, between about 7.00% and about 20.0% w/w), calcium chloride (in any concentration between about 0.00% and about 90.0% w/w, between about 0.01% and about 80.0% w/w, between about 0.01% and about 75.0% w/w, between about 0.01% and about 70.0% w/w, between about 0.01% and about 60% w/w, between about 0.01% and about 50.0% w/w, between about 0.01% and about 40.0% w/w, between about 0.01% and about 30% w/w, between about 0.10% and about 20.0% w/w), trehalose (in any concentration between about 0.00% and about 90.0% w/w, between about 0.01% and about 80.0% w/w, between about 1.00% and about 75.0% w/w, between about 2.00% and about 70.0% w/w, between about 3.00% and about 60% w/w, between about 4.00% and about 50.0% w/w, between about 5.00% and about 40.0% w/w, between about 6.00% and about 30% w/w, between about 7.00% and about 20.0% w/w), water (in any concentration between about 0.00% and about 90.0% w/w, between about 0.01% and about 80.0% w/w, between about 1.00% and about 75.0% w/w, between about 2.00% and about 70.0% w/w, between about 3.00% and about 60% w/w, between about 4.00% and about 50.0% w/w, between about 5.00% and about 40.0% w/w, between about 6.00% and about 30% w/w, between about 7.00% and about 20.0% w/w), Na-Acetate (in any concentration between about 0.00% and about 90.0% w/w, between about 0.01% and about 80.0% w/w, between about 0.01% and about 75.0% w/w, between about 0.01% and about 70.0% w/w, between about 0.01% and about 60% w/w, between about 0.01% and about 50.0% w/w, between about 0.01% and about 40.0% w/w, between about 0.01% and about 30% w/w, between about 0.10% and about 20.0% w/w), and acetic acid (in any concentration between about 0.00% and about 90.0% w/w, between about 0.01% and about 80.0% w/w, between about 0.01% and about 75.0% w/w, between about 0.01% and about 70.0% w/w, between about 0.01% and about 60% w/w, between about 0.01% and about 50.0% w/w, between about 0.01% and about 40.0% w/w, between about 0.01% and about 30% w/w, between about 0.10% and about 20.0% w/w).
[0196] Any of the above pharmaceutical compositions may in addition include MgCl.sub.2 in various concentrations ranging from about 0% to about 75% w/w.
Reducing mucus in the respiratory tract and limiting its increase
[0197] Accumulation or elevated levels of mucus in the respiratory airway tree can be caused by an increased volume of mucus produced, and also by decreased clearance due to defects in the ciliary clearance apparatus in the respiratory tract. Hypersecretion of mucus can be chronic, but increased volumes are produced in exacerbations of COPD, during attacks of asthma, and in bronchiectatic and cystic fibrosis patients (W. D. Kim, Eur Respir. J. 1997, 10:1914-1917). Intraluminal mucus accumulation (i.e., elevated levels of mucus) in the airways associated with hypersecretion of mucus or decreased clearance thereof creates a clinical problem in almost all pulmonary diseases and diseases that have an affect on the respiratory tract, including without limitation chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism. Elevated levels of mucus in the respiratory tract are an important determinant in the prognosis and clinical feastures of various pulmonary diseases, such as chronic bronchitis, bronchiectasis and bronchial asthma, in addition to cystic fibrosis and COPD (W. D. Kim, Eur Respir. J. 1997, 10:1914-1917). Accordingly, in some embodiments, the present disclosure include methods in which a subject with one or more of these conditions or diseases is selected for treatment. In some embodiments, the methods can include selecting a subject with one or more of the conditions or diseases provided herein and that is not infected with one or more of influenza, parainfluenza, and/or respiratory syncytial virus (RSV). Following selection, the subject can be treated by administration of one or more of the compositions disclosed herein.
[0198] Provided herein are methods that include the administration of the compounds described herein and in U.S. application Ser. Nos. 10/718,986 and 10/939,262, or compositions containing them, to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts and to limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts. Thus, the invention relates to method of using the therapeutic compounds and/or compositions described herein to prevent or treat diseases that are caused by, cause, or are exacerbated by respiratory inflammation or increased mucus production, such as, both allergic and non-allergic asthma, chronic obstructive pulmonary disease (COPD), bronchitis (both acute and non-acute), bronchiectasis, cystic fibrosis (CF), vasculitis, mucuous plugging, Wegener's granulomatosis, and any other disorder that causes inflammation or increased mucus production in the respiratory tract or is caused by or exacerbated by inflammation or increased mucus production in the respiratory tract. The invention also includes methods of using the therapeutic compounds and/or compositions described herein to reduce the quantity of mucus in the respiratory tract of subjects with elevated levels of mucus in their respiratory tracts and limit increases in the quantity of mucus in the respiratory tract of subjects above a baseline of mucus in their respiratory tracts.
[0199] In some embodiments, the methods include administering a composition or compound containing a therapeutically effective amount of a protein or peptide having a sialidase or an active portion thereof to a subject. The protein or peptide can be an isolated naturally occurring sialidase protein, or a recombinant protein substantially homologous to at least a portion of a naturally occurring sialidase. In one embodiment, a pharmaceutical composition or compound contains a sialidase with substantial homology to the A. viscosus sialidase (SEQ ID NO:12) or substantial homology to an active portion thereof, such as amino acids 274-666, 274-667, 270-667, 274-681, or 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis sialidase. The therapeutically effective amount includes an amount of the protein or peptide that results in a reduction of the quantity of mucus in the respiratory tract after administration of the composition or compound when compared to the quantity of mucus present prior to administration of the composition.
[0200] In other embodiments, the methods include administering a composition or compound containing a therapeutically effective amount of a fusion protein, wherein the fusion protein has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7. The therapeutically effective amount includes an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the composition or compound when compared to the quantity of mucus present prior to administration of the composition.
[0201] In yet other embodiments, the methods include administering a composition containing a therapeutically effective amount of a fusion protein having a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount includes an amount of the fusion protein that results in a reduction of the quantity of mucus in the respiratory tract after administration of the composition or compound when compared to the quantity of mucus present prior to administration of the composition.
[0202] Other embodiments include methods of preventing, treating or ameliorating the effects of chronic obstructive pulmonary disease (COPD), bronchitis, bronchiectasis, cystic fibrosis (CF), vasculitis, mucus plugging, Wegener's granulomatosis, pneumonia, tuberculosis, cancer involving the lungs or the respiratory tract, Kartagener syndrome, Young's syndrome, chronic sinopulmonary infection, alpha 1-antitrypsin deficiency, primary immunodeficiency, acquired immune deficiency syndrome, opportunistic infection, an infectious state, a post infectious state, common cold, exercise induced hypersecretion of mucus, inflammatory bowel disease, ulcerative colitis, Crohn's disease, respiratory infection, respiratory obstruction, inhalation or aspiration of a toxic gas, pulmonary aspiration, or alcoholism in a subject with an elevated level of mucus in his or her respiratory tract. The methods include administering (a) a composition containing a therapeutically effective amount of a protein or peptide having a sialidase or an active portion thereof to a subject, (b) a composition containing a therapeutically effective amount of a fusion protein, wherein the fusion protein has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7, or (c) a composition or compound containing a therapeutically effective amount of a fusion protein having a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount of these compositions or compounds includes an amount that results in a reduction of the quantity of mucus in the respiratory tract after administration of the composition when compared to the quantity of mucus present prior to administration of the composition or compound.
[0203] Yet other embodiments include methods of limiting an increase in the quantity of mucus in the respiratory tract of a subject above a baseline level of mucus in said subject's respiratory tract. The methods include administering (a) a composition or compound containing a therapeutically effective amount of a protein or peptide having a sialidase or an active portion thereof to a subject, (b) a composition or compound containing a therapeutically effective amount of a fusion protein, wherein the fusion protein has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of
[0204] SEQ ID NO:7, or (c) a composition or compound containing a therapeutically effective amount of a fusion protein having a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount of these compositions or compounds includes an amount that limits an increase in the quantity of mucus in the respiratory tract of the subject above a baseline level after administration of the composition.
[0205] In some embodiments, the compositions or compounds used can include additional compounds, including, without limitation, any one or more of the following either alone or in various combinations: Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs, Mg salts and/or Ca salts. These additional compounds can be included in the pharmaceutical compositions to act as excipients or as active ingredients that provide additional beneficial effects.
[0206] The subjects to be treated with the foregoing methods can be human subjects or non-human animal subjects. The compounds and compositions described herein can be administered to epithelial cells of the subject through various routes of administration, including, without limitation, by using inhalers to introduce the compounds or compositions into the respiratory tract of the subject.
[0207] In some preferred embodiments, compounds described herein can be delivered as an inhalant with an inhaler or as a nasal spray. They can also be administered as eye drops, ear drops, or sprays, ointments, lotions, or gels to be applied to the skin. They can also be administered intravenously or as a local injection.
Reducing or Preventing Inflammation in the Respiratory Tract
[0208] The present invention involves the unexpected discovery that administration of the compounds described in U.S. application Ser. Nos. 10/718,986 and 10/939,262, or compositions containing them, to reduce the amount of inflammatory cells in the respiratory tract. Thus, the invention relates to therapeutic compositions or compounds that can be used to reduce inflammation in the respiratory tract or prevent worsening of inflammation in the respiratory tract. The invention also includes methods of reducing inflammation in the respiratory tract or preventing worsening of inflammation in the respiratory tract. In addition, the invention relates to therapeutic compositions or compounds that can be used to prevent or treat diseases that are caused by, cause, or are exacerbated by respiratory inflammation, such as, both allergic and non-allergic asthma, allergic rhinitis, eczema, psoriasis, reactions to plant or animal toxins, autoimmune conditions, and any other disorder, disease or condition that causes inflammation in the respiratory tract or is caused by or exacerbated by inflammation in the respiratory tract.
[0209] In some preferred embodiments, the methods include administering a composition or compound containing a therapeutically effective amount of a protein or peptide having a sialidase or an active portion thereof to a subject. The protein or peptide can be an isolated naturally occurring sialidase protein, or a recombinant protein substantially homologous to at least a portion of a naturally occurring sialidase. A preferred pharmaceutical composition contains a sialidase with substantial homology to the A. viscosus sialidase (SEQ ID NO:12) or substantial homology to an active portion thereof, such as amino acids 274-666, 274-667, 270-667, 274-681, or 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis sialidase. The therapeutically effective amount includes an amount of the protein or peptide that prevents or reduces an allergic or inflammatory response in the respiratory tract the respiratory tract after administration of the composition or compound.
[0210] In other embodiments, the methods include administering a composition or compound containing a therapeutically effective amount of a fusion protein, wherein the fusion protein has at least one catalytic domain of a sialidase, wherein the catalytic domain of the sialidase includes the sequence of amino acids extending from amino acid 274 to amino acid 666 of SEQ ID NO:12 (alternatively, 274 to 666, 270-667, 274-681, 290-681 of SEQ ID NO:12, or any other catalytic domain of Actinomyces viscosis), inclusive, and at least one anchoring domain, wherein the anchoring domain is a glycosaminoglycan (GAG) binding domain of human amphiregulin including the amino acid sequence of SEQ ID NO:7. The therapeutically effective amount includes an amount of the fusion protein that prevents or reduces an allergic or inflammatory response in the respiratory tract the respiratory tract after administration of the composition or compound.
[0211] In yet other embodiments, the methods include administering a composition or compound containing a therapeutically effective amount of a fusion protein having a sialidase or an active portion thereof and an anchoring domain. The therapeutically effective amount includes an amount of the fusion protein that prevents or reduces an allergic or inflammatory response in the respiratory tract the respiratory tract after administration of the composition or compound.
[0212] In some embodiments, the compositions or compounds used can include additional compounds, including, without limitation, any one or more of the following either alone or in various combinations: Na.sub.2SO.sub.4, MgSO.sub.4, CaCl.sub.2), Histidine, Histine-HCl, and Trehalose or their analogs. These additional compounds can be included in the pharmaceutical compositions to act as excipients or as active ingredients that provide additional beneficial effects.
[0213] The subjects to be treated with the foregoing methods can be human subjects or non-human animal subjects. The compositions and compounds described herein can be administered to epithelial cells of the subject through various routes of administration, including, without limitation, by using inhalers to introduce the compounds or compositions into the respiratory tract of the subject.
[0214] In some preferred embodiments, compositions or compounds described herein can be delivered as an inhalant with an inhaler or as a nasal spray. They can also be administered as eye drops, ear drops, or sprays, ointments, lotions, or gels to be applied to the skin. They can also be administered intravenously or as a local injection. FIGS. 7A-B show the results of the effect of the use of one of the fusion protein construct depicted in FIG. 5 on inflammatory cells of ferrets infected with human unadapted influenza. In ferrets that shed the virus despite treatment with fusion protein (n=8), the inflammatory response was reduced and animals appeared to be more alert and active compared to the untreated ferrets that were invariably lethargic and feverish. For this group of 8 infected, fusion-protein treated animals, the mean AUC (area under the curve) value calculated for the nasal protein concentrations was reduced by approximately 40% (2.68 vs. 4.48, arbitrary units) compared to the vehicle-treated (phosphate buffer saline) infected animals (FIG. 7B). In vehicle-treated infected animals, the number of inflammatory cells in nasal washes was increased to approximately 100-fold above those in uninfected animals on day 2 post challenge. These levels were sustained for 4 additional days. The fusion protein-treated animals exhibited a significant reduction in the number of inflammatory cells in the nasal washes. Specifically, the AUC value for cell counts was reduced by approximately 3-fold in the fusion protein-treated animals compared to the vehicle-treated infected animals (1965 vs. 674, arbitrary units, (FIG. 7B). The observed reduction in the inflammatory response indicates the importance of inhibiting viral replication at the early stage of infection.
Dosage
[0215] As will be readily apparent to one skilled in the art, the useful in vivo dosage to be administered and the particular mode of administration will vary depending upon the age, weight and type of patient being treated, the particular pharmaceutical composition employed, and the specific use for which the pharmaceutical composition is employed. The determination of effective dosage levels, that is the dose levels necessary to achieve the desired result, can be accomplished by one skilled in the art using routine methods as discussed above. In non-human animal studies, applications of the pharmaceutical compositions are commenced at higher dose levels, with the dosage being decreased until the desired effect is no longer achieved or adverse side effects are reduced or disappear. The dosage for a compound of the present invention can range broadly depending upon the desired affects, the therapeutic indication, route of administration and purity and activity of the compound. Typically, human clinical applications of products are commenced at lower dosage levels, with dosage level being increased until the desired effect is achieved. Alternatively, acceptable in vitro studies can be used to establish useful doses and routes of administration of the test compound. Typically, dosages can be between about 1 ng/kg and about 10 mg/kg, between about 10 ng/kg and about 1 mg/kg, and between about 100 ng/kg and about 100 micrograms/kg. In various examples described herein, mice were treated with various dosages of the compositions described herein, including dosages of 0.0008 mg/kg, 0.004 mg/kg, 0.02 mg/kg, 0.06 mg/kg, 0.1 mg/kg, 0.3 mg/kg, 0.6 mg/kg, and 1.0 gm/kg. As nonlimiting examples, the compositions described herein can be administered to humans in doses of between about 0.01 mg and about 100 mg, such as about 0.05 mg, 0.06 mg, 0.1 mg, 0.5 mg, 1 mg, 5 mg, 10 mg, 20 mg, 50 mg, or 100 mg, and can be administered one or more times per day, for one day, two days, three days, four days, five days, six days, seven days, eight days, nine days, ten days, eleven days, twelve days, thirteen days, or fourteen or more days. Higher doses or lower doses can also be administered. In one embodiment, as shown in Example 3 below, a dose of 0.06 mg/kg of a sialidase compound is sufficient to desialylate muscarinic receptors resulting in reduced airway responsiveness to muscarinic receptor agonists, and thus potentially resulting in reducing airway constriction, airway hypersensitivity, inflammation, allergies or associated responses, such as bronchoconstriction, asthma, and mucus overproduction. Efficacy in low doses, such as 0.06 mg/kg (translating in adult humans into a dose of about 4 or 5 mg), or 0.02 mg/kg (translating in adult humans into a dose of about 1 or 2 mg), makes the sialidase-based compounds described herein good candidates for use in chronic diseases that require repeated long-term administration.
[0216] A treatment regimen can include administration of the compounds and compositions described herein from once per day to ten times per day, from once per day to six times per day, from once per day to five times per day, from once per day to four times per day, from once per day to three times per day, from once per day to twice per day, and just once per day. The treatment can last from just one day to daily, weekly, monthly, or other periodic use for a predetermined period of time or for the remainder of the subject's life.
[0217] The exact formulation, route of administration and dosage can be chosen by the individual physician in view of the patient's condition (see, Fingle et al., in The Pharmacological Basis of Therapeutics (1975)). It should be noted that the attending physician would know how to and when to terminate, interrupt or adjust administration due to toxicity, organ dysfunction or other adverse effects. Conversely, the attending physician would also know to adjust treatment to higher levels if the clinical response were not adequate. The magnitude of an administrated does in the management of the disorder of interest will vary with the severity of the condition to be treated and to the route of administration. The severity of the condition may, for example, be evaluated, in part, by standard prognostic evaluation methods. Further, the dose and perhaps dose frequency, will also vary according to the age, body weight and response of the individual patient, including those for veterinary applications.
[0218] In some preferred regimens, appropriate dosages are administered to each patient by either inhaler, nasal spray, or by topical application. It will be understood, however, that the specific dose level and frequency of dosage for any particular patient may be varied and will depend upon a variety of factors including the activity of the specific salt or other form employed, the metabolic stability and length of action of that compound, the age, body weight, general health, sex, diet, mode and time of administration, rate of excretion, drug combination, the severity of the particular condition, and the host undergoing therapy.
[0219] In some embodiments, the present disclosure provides methods for using any one or more of the compositions (indicated below as `X`) disclosed herein in the following methods:
[0220] Substance X for use as a medicament in the treatment of excess mucus or abnormal (e.g., above normal mucus levels as compared to one or more healthy subjects (e.g., of the same ethnicity and/or in the same or similar geographical location) and/or as indicated by a health care practitioner), elevated mucus production, and/or any one or more of the diseases/conditions disclosed herein; (each of which is collectively referred to in the following examples as `Y.`
[0221] Use of substance X for the manufacture of a medicament for the treatment of Y; and
[0222] Substance X for use in the treatment of Y.
EXAMPLES
[0223] The invention is further described in the following examples, which do not limit the scope of the invention described in the claims.
Example 1. Effect of Sialidase Treatment on the Early and Late Asthmatic Reaction in Guinea Pigs
[0224] 1. Overview
[0225] In this study Fludase.RTM. was tested in a guinea pig model of allergic asthma. Guinea pigs were sensitised with ovabumin (OVA) or saline and after 15 or 20 days they were treated with Fludase.RTM. or sodium sulfate. On day 21, all the animals were challenged with OVA to measure the early asthmatic reaction. Airway compliance/resistance were determined and broncho-alveolar lavage (BAL) fluid was taken from the left lung to count the total number of cells and to differentiate them.
[0226] 2. Introduction
[0227] The main purpose of this study was to achieve a characterization of the effect of Fludase.RTM. on the early and late reactions in a guinea pig model for asthma. The guinea pigs involved in the study were naive, and thus not infected with influenza or other infectious agent as part of the experiment. Asthma was induced by sensitising the guinea pigs on day 0 with OVA. After 15 or 20 days the guinea pigs were treated intatracheal with Fludase.RTM. (0.3 mg/kg) or sodium sulfate (0.143 mM, pH 5.0). On day 21 the guinea pigs received an OVA aerosol and the airway responsiveness (PenH) was measured. On day 22 the pulmonary resistance and compliance were determined. At time intervals of 2 minutes doses of histamine from 0.2-2 .mu.g/kg were administered by intravenously injection. At the end of the experiment guinea pigs were sacrificed and the left lung was lavaged and the isolated BAL cells were washed, counted and differentiated into macrophages, lymphocytes, neutrophils and eosinophils.
[0228] 3. Materials & Methods
[0229] Animals
[0230] Male Hartley-strain guinea pigs (HSD Poc: DH, weighing 400-500) of specific pathogen free quality were obtained from Harlan-CPB (Zeist, The Netherlands). They were used after 1 week of acclimatisation to their housing conditions. Water and commercial chow were allowed ad libitum. The experiments were approved by the Animal Ethics Committee of the Utrecht University (Utrecht, The Netherlands).
[0231] Sensitisation, Pre-Treatment & Challenge
[0232] Guinea pigs were sensitised with saline (solutions contains 100 mg/ml Al(OH).sub.3) or OVA (solution contains 20 .mu.g/ml OVA and 100 mg/ml Al(OH).sub.3), administered intraperitoneally 0.5 ml and subcutanously 5.times.0.1 ml, total injection volume 1 ml. After 15 or 20 days animals were treated once with 0.3 mg/kg Fludase.RTM. and the control animals were treated with sodium sulfate (0.143 mM, pH 5.0) on day 20 by tracheal instillation. A laryngoscope was used to facilitate the location of the epiglottis. Then the Fludase.RTM. or sodium sulfate was given with a liquid aerosol using the IA-1C MicroSprayer (Penn Century, Inc, Philadelphia, USA). The guinea pig was in an upright position during the tracheal instillation.
[0233] During this tracheal instillation the guinea pigs were anaesthetized with 150 .mu.l of a mixture of Ketamine.RTM., Xylazin.RTM., Atropin and saline (3.5:3:1:3), injected intra muscular in the hind paw.
[0234] On day 21 the guinea pigs were challenged by exposure to an aerosol OVA (0.1% wt/vol in sterile saline). The aerosol was generated into a 3 liter perspex chamber in which the guinea pigs were placed. First the basal bronchoconstriction (PenH) was measured. The guinea pigs were provoked with OVA aerosol for 10 seconds. Directly after the challenge the early asthmatic reaction was (PenH) was measured.
[0235] Allergen-Induced Early Asthmatic Reaction in Conscious Unrestrained Guinea Pigs
[0236] Airway function of the animals was measured directly after exposure to aerosolised OVA in a ventilated bias flow whole body plethysmograph (Buxco Electronics, Sharon Conn., USA). The plethysmograph consists of a reference chamber and an animal chamber. The animal chamber is attached to the outside via a pneumotachograph in the top of the plethysmograph. An aerosol inlet to the animal chamber is centrically located in the roof of the animal chamber. When an animal is placed in the animal chamber and is breathing quietly, it creates pressure between tidal volume and thoracic movement during respiration. The differential pressure transducer measures the changes in pressure between animal chamber and the reference chamber and brings these data to a preamplifier. Thereafter, data is sent to a computer where several parameters are calculated, which represents animal's lung function. All guinea pigs used were measured basal for 5 minutes and after the aerosol for 15 minutes in the whole body plethysmograph. Besides known lung function parameters as peak expiratory flow (PEF) and tidal volume (TV), the enhanced pause (PenH) was also measured. The formula and explanation of the PenH is shown in FIG. 8. During bronchoconstriction peak expiratory flow and peak inspiratory flow are increased, while relaxation time and expiratory time are decreased. This results in an increased PenH. Data from bronchoconstriction in conscious unrestrained guinea pigs are presented in PenH (FIG. 8).
Airway Responsiveness In Vivo
[0237] On day 22 the guinea pigs were anaesthetized with urethane 2 g/kg intra peritoneally. The animals were allowed to breathe spontaneously. An anaesthesia-induced fall in body temperature was avoided by placing the animals in a heated chamber, which kept the body temperature at 37.degree. C. The guinea pigs were prepared for the measurement of pulmonary resistance (R.sub.L) and compliance (C) as follows. A small polyethylene catheter (PE-50) was placed in the right jugular vein for intra venous administration of increasing doses of histamine (0.2-2 .mu.g/kg). First the basal R.sub.L and C were measured for 5 minutes. Thereafter an increasing dose of histamine was injected and R.sub.L and C were measured for 2 minutes. Airflow and tidal volume were determined by cannulating and connecting the trachea with Fleisch flow head (nr 000; Meijnhart, Bunnik, The Netherlands) to a pneumotachograph. A pressure transducer (model MP45-2; Validyne Engineering Corp., Northridge, Calif.) measured the transpulmonary pressure by determing pressure differences between the tracheal cannula and a cannula filled with saline inserted in the oesophagus. R.sub.L and C were determined breath by breath with a respiratory analyser. R.sub.L was yielded by dividing transpulmonary pressure by airflow at isovolume points. C was determined by dividing volume by transpulmonary pressure at isoflow points. Data are presented as maximal R.sub.L and minimal C in cm H.sub.2O/ml*sec.sup.-1 an ml/cm H.sub.2O, respectively.
[0238] Collection of Broncho-Alveolar Lung Lavage Cells
[0239] Broncho-alveolar lavage cells were obtained as follows. The trachea was trimmed free of connective tissue and blood vessels and a small incision was made for insertion of a cannula into the trachea. The right lung was tied up so only the left lung was lavaged. The left lung was filled with 5 ml saline (0.9% NaCl) of 37.degree. C. in situ. Fluid was withdrawn from the lung after gentle massage and collected in a plastic tube on ice (4.degree. C.). This procedure was repeated 3 times (total 15 ml) and the cell suspensions recovered from each animal were pooled. Thereafter, cells were sedimented by centrifugation at 1500 rpm for 5 minutes at 4.degree. C. The supernatant solution was thrown away and the pellet was resuspended in 1 ml saline. Only plastic tubes were used throughout the isolation procedure in order to minimize adherence of the cells to the walls of the tubes.
[0240] Cell Count and Differentiation
[0241] The cells were stained with Turk solution and counted in a Burker-Turk bright-line counting chamber (microscope, magnification 100.times.). For differential BAL cell counts cytospin preparations were made and stained with Diff-Quick (Merz & Dade A. G., Dudingen, Switzerland). After coding all cytospin preparations were evaluated by one observer using oil immersion microscopy (magnification 1000.times.). Cells were differentiated into macrophages, lymphocytes, neutrophils and eosinophils by standard morphology. At least 200 cells per cytospin preparation were counted and the absolute number of each cell type was calculated.
[0242] Statistical Analysis
[0243] Unless stated otherwise, data are expressed as arithmetic average .+-.standard error of mean and comparisons between groups were made using Student's t-test. A probability value p<0.05 was considered significant.
[0244] 4. Results
[0245] Airway Responsiveness in Conscious Unrestrained Guinea Pigs
[0246] As shown in FIG. 9, basal airway resistance was not different between the saline guinea pigs treated with sodium sulfate or DAS181 on day 15 or day 20 (PenH=0.24.+-.0.009 sal-sodium sulfate, 0.25.+-.0.01 saline-Flu day 15, 0.28.+-.0.01 sal-Flu day 20 treated guinea pigs). There was also no difference between the OVA guinea pigs treated with sodium sulfate or Fludase.RTM. on day 15 or day 20 at basal level (PenH=0.25.+-.0.01 OVA-sodium sulfate, 0.27.+-.0.02 OVA-Flu day 15, 0.31.+-.0.03 OVA-Flu day 20 treated guinea pigs).
[0247] Ova challenge slightly increased the basal airway resistance in saline sensitized animals (SOD, Flu 15 and 20, FIG. 2). However, the early asthmatic reaction in response to the OVA aerosol was strongly increased in the OVA-sodium sulfate treated guinea pigs (PenH=1.57.+-.0.45). After treatment with Fludase.RTM. the early asthmatic reaction was decreased by nearly 30% on day 15, but not on day 20.
[0248] Cell Count in Broncho-Alveolar Lavage Fluid
[0249] As shown in FIG. 10, Fludase.RTM. decreases the total number of cells both in saline and OVA guinea pigs.
[0250] Differential Cell Count in the Broncho-Alveolar Lavage Fluid
[0251] As shown in FIG. 11, the number of macrophages was enhanced by (30%) in the OVA guinea pigs compared to the saline treated group. Total number of macrophages was strongly decreased after treatment with Fludase.RTM. on day 15 and 20, both in saline and OVA treated guinea pigs.
[0252] As shown in FIG. 12, a similar pattern was observed with the number of lymphocytes. The number of lymphocytes is decreased after Fludase.RTM. treatment.
[0253] As shown in FIG. 13, compared to historical controls, SOD induces a strong increase in the number of neutrophils into the lungs. Interestingly, Fludase.RTM. completely prevented this influx in both the saline and OVA groups.
[0254] As shown in FIG. 14, compared to historical controls SOD induces also an eosinophil influx into the airways, which is further increased by OVA challenge. Interestingly, Fludase.RTM. treatment can restore the number of eosinophils up to historical control levels.
[0255] 5. Discussion/Conclusion
[0256] OVA-sensitised and challenged animals demonstrate an early and late asthmatic reaction as measured by an increase in PENH up on OVA-challenge, and an increase in the number of inflammatory cells in the BAL-fluid.
[0257] Interestingly, Fludase.RTM. (Flu15) reduced the early asthmatic response by nearly 30% (FIG. 9), suggesting an effect of this compound on mast cell stimulation. Moreover, Fludase.RTM. had a tremendous effect on the inflammation caused by SOD or OVA. The number of all inflammatory cells (macrophages, lymphocytes, neutrophils and eosinophils) was significantly decreased by this compound.
[0258] In conclusion, Fludase.RTM. demonstrated to be effective in both the SOD-induced inflammation and the OVA-induced inflammation.
Example 2. The Effect of Fludase in a Mouse Model of Acute OVA Induced Asthma
[0259] 1. Introduction
[0260] The aim of the study was to investigate whether Fludase.RTM. (also referred to as DAS181) (1) inhibits allergen induced airway inflammation and airway hyperreactivity in an acute OVA challenge mouse model of asthma. In addition to studying the effect of DAS 181 alone as an intervention, three other interventions in the mouse asthma model were studied including a) DAS181+an excipient (used as the dry powder formulation to deliver DAS181 in vivo), b) the excipient alone, and c) dexamethasone (as a comparator).
[0261] 2. Materials and Methods
[0262] Mice
[0263] Female BALB/c mice age 12 weeks were purchased from Charles River and housed in the UCSD vivarium. They were used after approximately one week of acclimatization in the UCSD vivarium.
[0264] OVA Sensitization and OVA Challenge
[0265] Mice were immunized s.c. on days 0, 7, 14, and 21 with 25 ug of OVA adsorbed to 1 mg of alum in 200 ul normal saline to induce a predominant Th2 immune response. Intranasal OVA challenges (20 ug/50 ul) were started on day 26 and then repeated on day 28 and day 30. In the no OVA group, mice were sensitized to OVA but not challenged intranasally with OVA. Mice had airway responsiveness to methacholine (Mch) measurements performed 24 hours after the final OVA challenge by Penh on day 31. The mice involved in the study were naive and thus not infected with influenza or other infectious agent as part of the experiment. Mice were then immediately sacrificed. Bronchoalveolar lavage fluid (BAL), blood, and lung tissue were processed for outcomes detailed below.
[0266] Administration of Test Compounds to Mice
[0267] 1. Compounds Tested
[0268] The following compounds were studied in the mouse model of asthma [0269] a) DAS181 with excipient (0.6 mg/kg intranasal): DAS181-F02 (NexBio, Inc. part #43-071, lot #47-034) was prepared in PBS to 20 mgDAS/ml. Before each dosing, it was freshly diluted in PBS to 0.6 mgDAS181/kg with dosing volume of 50 ul, for mice with an average body weight of 21 g. [0270] b) DAS181 (0.6 mg/kg intranasal) [0271] c) Excipient (50 .mu.L/mouse): Excipient (416TL022A) was supplied by NexBio, Inc. as a solution, and the concentration of each of the excipient component is equivalent to that in 20 mgDAS181/ml (MgSO.sub.4 1.446 mg/ml, CaCl.sub.2) 0.059 mg/ml, Histidine 1.427 mg/ml, Histidine-HCl 1.943 mg/ml, and Trehalose 3.000 mg/ml). It was freshly diluted in PBS for dosing as in a). The final concentration of each excipient component is equivalent to that in 0.6 mgDAS181/kg, in 50 ul, for mice with average body weight of 21 g. [0272] d) Dexamethasone (1.0 mg/kg intraperitoneally)
[0273] Excipient solution was prepared in the following manner. The target final concentration for each excipient was calculated to reach equivalent concentration when DAS181-F02 bulk dry powder is reconstituted at a 20 mg protein/mL. The 10.times. stock solutions (100.times. for Calcium Chloride) for each excipient (10 mL each) were then prepared. Materials used to prepare the stock solutions are listed in Table 1. All materials are USP grade, or equivalent. Appropriate amounts of each excipient were weighed into a 15 mL conical tube according to Table 2, and water was added to bring the total weight to 10 grams and vortex to dissolve the material completely. The final 1.times. excipient solution was prepared by adding 1 mL of each stock solution, and then bringing the volume to 10 mL using water. All sample preparation was preformed gravimetrically, assuming solution density of 1 gram/mL.
[0274] Although referred to as "excipient" or "excipient" solution in these examples, these additional compounds can have additional beneficial effects with respect to reduction of mucus and reduction of inflammation and inflammatory cells. These excpients can also have a synergistic effect with DAS181.
TABLE-US-00001 TABLE 1 Material Information Description Manufacturer Mfg. Part # Mfg. Lot # Expiry L-Histidine Sigma H6034-100g 078K0179 September 2012 L-Histidine Sigma H4036-1kg 068K8310 January 2012 monohydrochloride monohydrate a,a - Trehalose, Dihydrate J. T. Baker 4226-04 G47596 November 2010 Magnesium Sulfate EMD 1.05882.0500 K38528682R February 2013 Heptahydrate Calcium Chloride, Dihydrate Mallinckrodt 4616-04 G24475 September 2009 WFI B. Braun S9200-SS J8K015 August 2010
TABLE-US-00002 TABLE 2 Stock Solution Preparation Sheet (Theoretical) Final conc. Dilution Stock conc. Wt. of Salt Composition (mg/mL) Factor (mg/mL) for 10 mL (mg) MgSO.sub.4 1.446 10 14.457 296 CaCl.sub.2 0.059 100 5.943 79 Histidine 1.427 10 14.266 143 Histidine.cndot.HCl 1.943 10 19.431 213 Trehalose 3.000 10 30.000 332
[0275] 2. Mouse Model of Asthma
[0276] The following groups of Balb/c mice (n=10 female mice/group) were studied.
[0277] a) No OVA
[0278] b) OVA
[0279] c) OVA+DAS181 with excipient
[0280] d) OVA+DAS181
[0281] e) OVA+excipient
[0282] f) OVA+dexamethasone
[0283] 3. Timing of Administration of Test Compounds
[0284] The test compounds were administered one hour prior to each of the three intranasal OVA challenges on days 26, 28, and 30.
[0285] Timing of End-Points Studied
[0286] Mice were sacrificed 24 hours after the final OVA challenge and blood, BAL, and lungs were analyzed (27).
[0287] 4. End-Points Studied
[0288] a) Penh
[0289] Airway responsiveness was assessed on day 31, twenty four hours after the final OVA inhalation, using a single chamber whole body plethysmograph obtained from Buxco (Troy, N.Y.). In this system, an unrestrained, spontaneously breathing mouse is placed into the main chamber of the plethysmograph, and pressure differences between this chamber and a reference chamber are recorded. The resulting box pressure signal is caused by volume and resultant pressure changes during the respiratory cycle of the mouse. A low pass filter in the wall of the main chamber allows thermal compensation. From these box pressure signals, the phases of the respiratory cycle, tidal volumes, and the enhanced pause (Penh) can be calculated. Penh is a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. It correlates closely with pulmonary resistance measured by conventional two-chamber plethysmography in ventilated mice. In the plethysmograph, mice were exposed for 3 min to nebulized PBS and subsequently to increasing concentrations of nebulized metacholine (MCh)(3, 6, 12, 24, 48 mg/ml Mch) (Sigma, St. Louis, Mo.) in PBS using an Aerosonic ultrasonic nebulizer (DeVilbiss). After each nebulization, recordings were taken for 3 min. The Penh values measured during each 3-min sequence were averaged and are expressed for each MCh concentration as the percentage of baseline Penh values following PBS exposure.
[0290] b) Blood Eosinophil Counts
[0291] Peripheral blood was collected from mice by cardiac puncture into EDTA-containing tubes. Erythrocytes were lysed using a 1:10 solution of 100 mM potassium carbonate-1.5 M ammonium chloride. The remaining cells were resuspended in 1 mL PBS. To perform differential cell counts, 200 .mu.L resuspended peripheral-blood leukocyte suspensions were cytospun onto microscope slides and air-dried. Slides were stained with Wright-Giemsa and the % of eosinophils in the total number of white blood cells were assessed under a light microscope.
[0292] c) PAS Staining for Lung Mucus
[0293] To quantitate the level of mucus expression in the airway, the number of periodic acid Schiff (PAS)-positive and PAS-negative epithelial cells in individual bronchioles were counted as previously described (Zhang, M., T. Angata, J. Y. Cho, M. Miller, D. H. Broide, A. Varki. 2007 Blood. 109:4280-4287). At least ten bronchioles were counted in each slide. Results are expressed as the percentage of PAS-positive cells per bronchiole, which is calculated from the number of PAS-positive epithelial cells per bronchus divided by the total number of epithelial cells of each bronchiole. Slides of lung tissue with no OVA, OVA and OVA+DAS181 were also taken and observed.
[0294] d) MBP Staining of Lungs for Peribronchial Eosinophils
[0295] Lungs from the different experimental groups were processed as a batch for either histologic staining or immunostaining under identical conditions as described in Zhang et al. Stained and immunostained slides were all quantified under identical light microscope conditions, including magnification (20 X), gain, camera position, and background illumination. Lung sections were processed for MBP immunohistochemistry as described above, using an anti-mouse MBP (Major Basic Protein) Ab (kindly provided by James Lee PhD, Mayo Clinic, Scottsdale, Ariz.) and the immunoperoxidase method as previously described in Zhang et al. Major Basic Protein is an eosinophil cytoplasmic granule protein which serves as a marker of eosinophils in tissues. The number of individual cells staining positive for MBP in the peribronchial space were counted using a light microscope. Results are expressed as the number of peribronchial cells staining positive for MBP per bronchiole with 150-200 .mu.m of internal diameter. At least ten bronchioles were counted in each slide.
[0296] 5. Statistical Analysis
[0297] Results in the different groups of mice were compared by Mann Whitney non-parametric T test. All results are presented as mean+SEM. A statistical software package (Graph Pad Prism, San Diego, Calif.) was used for the analysis. P values of <0.05 were considered statistically significant.
[0298] 6. Results
[0299] a) Penh
[0300] As shown in FIG. 15, OVA challenge induced a significant increase in airway responsiveness as assessed by changes in Penh (OVA vs no OVA; p<0.0001).
[0301] OVA challenged mice pre-treated with DAS181+excipient had a significant reduction in Penh compared to OVA challenged mice (OVA vs OVA+DAS181+excipient; p<0.01).
[0302] OVA challenged mice pre-treated with DAS181 resulted in a reduction in Penh compared to OVA challenged mice (OVA vs OVA+DAS181; p<0.005).
[0303] As shown in FIG. 16, measurement of Penh at 48 mg/ml Mch provides the largest difference between positive and negative controls (no OVA vs OVA) and is why this dose of Mch is used to assess the effect of an intervention such as DAS181.
[0304] b) Blood Eosinophils
[0305] As shown in FIG. 17, OVA challenge induced a significant increase in blood eosinophils (OVA vs no OVA; p<0.0005).
[0306] DAS 181 significantly reduced blood eosinophils (OVA vs OVA+DAS, p=0.04)
[0307] c) PAS Staining for Lung Mucus
[0308] As shown in FIGS. 18-19A-F, OVA challenge induced a significant increase in the % of airway epithelium staining positive for PAS (OVA vs no OVA; p<0.0001).
[0309] OVA challenged mice pre-treated with DAS181+excipient had a significant reduction in PAS staining compared to OVA challenged mice (OVA vs OVA+DAS181+excipient; p<0.0001).
[0310] OVA challenged mice pre-treated with DAS181 had a statistically significant reduction in PAS staining compared to OVA challenged mice (OVA vs OVA+DAS181; p<0.0001).
[0311] Effect on Mucus
[0312] DAS181 with excipient as well as DAS181 alone significantly reduced PAS staining showing that there is an inhibitory effect of DAS 181 on PAS staining. This shows that DAS181 with excipient or DAS181 alone reduces mucus in the respiratory tract.
[0313] d) MBP Immunostaining of Lungs
[0314] As shown in FIG. 20, OVA challenge induced a significant increase in the number of peribronchial MBP+eosinophils (OVA vs no OVA; p<0.0001).
[0315] OVA challenged mice pre-treated with DAS181+excipient had a significant reduction in the number of peribronchial MBP+eosinophils compared to OVA challenged mice (OVA vs OVA+DAS181+excipient; p=0.02).
Example 3. Reduced Airway Resistance in Naive Mice Treated Intranasally with Low Doses of DAS181 (Methacholine Challenged)
[0316] The objective of the study was to test the effect of different dose levels of DAS181 on muscarinic receptor mediated airway resistance in naive mice. BALB/c mice (N=4) were treated intranasally with PBS or DAS181 at 0.06, 0.1 or 0.6 mg/kg once daily for three days. Eight hours post the final treatment animals were challenged with increasing doses of muscarinic receptor agonist Methacholine (Mch). Airway responsiveness was assessed using whole body plethysmography. Changes in airway resistance were expressed as the enhanced pause (Penh), a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. Mice were exposed to nebulized PBS and subsequently to increasing concentrations of nebulized MCh (12, 24, 48 mg/ml Mch) for 2 min in PBS. Recordings were performed for 3 min following each exposure. The obtained Penh values were averaged and expressed as the percentage of baseline following PBS exposure.
[0317] Results: 24 and 48 mg/ml of the muscarinic receptor agonist methacholine increased airway resistance above baseline. All animals treated with DAS181 had significantly reduced airway resistance at 48 mg/ml of Mch (Fig). No difference was observed between the different dose groups.
[0318] Conclusions: Consistent with previous data, intranasal treatment with DAS181 reduced bronchoconstriction in response to the muscarinic receptor agonist Mch, further supporting the hypothesis that DAS181 dependent desialylation causes a reduction in muscarinic receptor signaling. Surprisingly, the two higher dose levels did not exert any greater effect than the lowest dose, suggesting that a dosage level as low as 0.06 mg/kg of intranasal DAS181 is sufficient to desialylate muscarinic receptors resulting in reduced airway responsiveness to muscarinic receptor agonists, and thus potentially resulting in reducing inflammation, allergies or acetylcholine-associated responses, such as bronchoconstriction, asthma, and mucus overproduction. These results are depicted in FIGS. 21A and 21B.
Example 4. Reduced Airway Resistance in Naive Mice Treated Intranasally with a Low Dose of DAS181 (Methacholine Challenged)
[0319] The objective of the study was to test the effect of a 0.6 mg/kg once daily dose of DAS181 on muscarinic receptor mediated airway resistance in naive mice. BALB/c mice (N=4) were treated intranasally with PBS or DAS181 at 0.6 mg/kg once daily for three days. Eight hours post the final treatment animals were challenged with increasing doses of muscarinic receptor agonist Methacholine (Mch). Airway responsiveness was assessed using whole body plethysmography. Changes in airway resistance were expressed as the enhanced pause (Penh), a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. Mice were exposed to nebulized PBS and subsequently to increasing concentrations of nebulized MCh (3, 6, 12, 24, 48 mg/ml Mch) for 2 min in PBS. Recordings were performed for 3 min following each exposure. The obtained Penh values were averaged and expressed as the percentage of baseline following PBS exposure.
[0320] Results: 3, 6, 12, 24 and 48 mg/ml of the muscarinic receptor agonist methacholine increased airway resistance above baseline in PBS treated animals, and 12, 24 and 48 mg/ml of methacholine increased airway resistance above baseline in DAS181 treated animals, while 3 and 6 mg/ml of methacholine did not increase airway resistance above baseline in DAS181 treated animals. All animals treated with DAS181 had significantly reduced airway resistance at 6, 12, 24 and 48 mg/ml of Mch compared to the control.
[0321] Conclusions: Consistent with previous data, intranasal treatment with DAS181 reduced bronchoconstriction in response to the muscarinic receptor agonist Mch, further supporting the hypothesis that DAS181 dependent desialylation causes a reduction in muscarinic receptor signaling. These results are depicted in FIG. 22.
Example 5. Reduced Airway Resistance in Naive Mice Treated Intranasally with DAS181 (Carbachol Challenged)
[0322] The objective of the study was to test the effect of a 0.6 mg/kg once daily dose of DAS181 on muscarinic receptor mediated airway resistance in naive mice. BALB/c mice (N=4) were treated intranasally with PBS or DAS181 at 0.6 mg/kg once daily for three days. Eight hours post the final treatment animals were challenged with increasing doses of muscarinic receptor agonist carbachol. Airway responsiveness was assessed using whole body plethysmography. Changes in airway resistance were expressed as the enhanced pause (Penh), a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. Mice were exposed to nebulized PBS and subsequently to increasing concentrations of nebulized Carbachol (1.25, 2.5, 5, 10, 20 mg/ml carbachol) for 2 min in PBS. Recordings were performed for 3 min following each exposure. The obtained Penh values were averaged and expressed as the percentage of baseline following PBS exposure.
[0323] Results: 5, 10, and 20 mg/ml of the muscarinic receptor agonist carbachol increased airway resistance above baseline in both PBS treated and DAS181 animals. All animals treated with DAS181 had significantly reduced airway resistance at 5, 10, and 20 mg/ml of carbachol.
[0324] Conclusions: Consistent with previous data, intranasal treatment with DAS181 reduced bronchoconstriction in response to the muscarinic receptor agonist carbachol, further supporting the hypothesis that DAS181 dependent desialylation causes a reduction in muscarinic receptor signaling. These results are depicted in FIG. 23.
Example 6. Airway Resistance in Naive Mice Treated Intranasally with a Low Dose of DAS185 (Methacholine Challenged)
[0325] The objective of the study was to test the effect of a 0.6 mg/kg once daily dose of DAS185 on muscarinic receptor mediated airway resistance in naive mice. DAS185 is an enzymatically inactive version of DAS181, in which a mutation in the sialidase portion renders the sialidase inactive. BALB/c mice (N=4) were treated intranasally with PBS or DAS185 at 0.6 mg/kg once daily for three days. Eight hours post the final treatment animals were challenged with increasing doses of muscarinic receptor agonist Methacholine (Mch). Airway responsiveness was assessed using whole body plethysmography. Changes in airway resistance were expressed as the enhanced pause (Penh), a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. Mice were exposed to nebulized PBS and subsequently to increasing concentrations of nebulized MCh (3, 6, 12, 24, 48 mg/ml Mch) for 2 min in PBS. Recordings were performed for 3 min following each exposure. The obtained Penh values were averaged and expressed as the percentage of baseline following PBS exposure.
[0326] Results: There was no difference in airway resistance in response to the Mch challenge between the DAS185 treated and PBS treated animals.
[0327] Conclusions: Whereas there was a difference in airway resistance between DAS181 and PBS treated animals in example 4 above, there was no difference when DAS181 was replaced with enzymatically inactive DAS185. This experiment shows that reduction in airway resistance in response to DAS181 is sialidase dependent. These results are depicted in FIG. 24.
Example 7. Time-Course of DAS181 Mediated Reduction of Airway Resistance (Methacholine Challenged)
[0328] The objective of the study was to test the effect of a 0.6 mg/kg once daily dose of DAS181 for one, two or three days on muscarinic receptor mediated airway resistance in naive mice. BALB/c mice (N=4) were treated intranasally with PBS or DAS181 at 0.6 mg/kg once daily for one, two or three days. Eight hours post the final treatment animals were challenged with increasing doses of muscarinic receptor agonist Methacholine (Mch). Airway responsiveness was assessed using whole body plethysmography. Changes in airway resistance were expressed as the enhanced pause (Penh), a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. Mice were exposed to nebulized PBS and subsequently to increasing concentrations of nebulized MCh (3, 6, 12, 24, 48 mg/ml Mch) for 2 min in PBS. Recordings were performed for 3 min following each exposure. The obtained Penh values were averaged and expressed as the percentage of baseline following PBS exposure.
[0329] Results: For one day of treatment, there was no difference in airway resistance in response to the Mch challenge between the DAS181 treated and PBS treated animals. For two days of treatment, DAS181 appears to reduce airway resistance relative to PBS at 12 and 24 mg/ml methacholine, but not at 3, 6 and 48 mg/ml of methacholine. At three days of treatment, DAS181 had significantly reduced airway resistance at 24 and 48 mg/ml of Mch.
[0330] Conclusions: Consistent with previous data, there was a difference in airway resistance between DAS181 and PBS treated animals on day 3. There was no difference when following one treatment dose, and partial reduction following two days of treatment with DAS181. This experiment shows that 2-3 days of treatment is optimal to achieve a reduction in airway resistance. These results are depicted in FIG. 25.
Example 8. Reduced Airway Resistance in Naive Mice Treated Intranasally with Very Low Doses of DAS181 (Methacholine Challenged)
[0331] The objective of the study was to test the effect of different low-dose levels of DAS181 on muscarinic receptor mediated airway resistance in naive mice. BALB/c mice (N=4) were treated intranasally with PBS or DAS181 at 0.0008, 0.004, 0.02, or 0.1 mg/kg once daily for three days. Eight hours post the final treatment animals were challenged with increasing doses of muscarinic receptor agonist Methacholine (Mch). Airway responsiveness was assessed using whole body plethysmography. Changes in airway resistance were expressed as the enhanced pause (Penh), a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. Mice were exposed to nebulized PBS and subsequently to increasing concentrations of nebulized MCh (12, 24, 48 mg/ml Mch) for 2 min in PBS. Recordings were performed for 3 min following each exposure. The obtained Penh values were averaged and expressed as the percentage of baseline following PBS exposure.
[0332] Results: 24 and 48 mg/ml of the muscarinic receptor agonist methacholine increased airway resistance above baseline. All animals treated with DAS181 had significantly reduced airway resistance at 24 and 48 mg/ml of Mch. No difference was observed between the different dose groups of DAS181.
[0333] Conclusions: Intranasal treatment with very low doses of DAS181 reduced bronchoconstriction in response to the muscarinic receptor agonist Mch, further supporting the hypothesis that DAS181 dependent desialylation causes a reduction in muscarinic receptor signaling even at very low doses of DAS181. This experiment shows that dosage levels as low as 0.0008 mg/kg of intranasal DAS181 is sufficient to desialylate muscarinic receptors resulting in reduced airway responsiveness to muscarinic receptor agonists, and thus potentially resulting in reducing inflammation, allergies or acetylcholine-associated responses, such as bronchoconstriction, asthma, and mucus overproduction. These results are depicted in FIG. 26.
Example 9. The Reduced Airway Resistance in Naive Mice Treated Intranasally with DAS181 is Dose-Dependent (Methacholine Challenged)
[0334] The objective of the study was to test the effect of different dose levels of DAS181 on muscarinic receptor mediated airway resistance in naive mice. BALB/c mice (N=4) were treated intranasally with PBS or DAS181 at 10 ng/kg, 0.1 .mu.g/kg, 1 .mu.g/kg, 10 .mu.g/kg or 0.1 mg/kg once daily for three days followed by increasing doses of muscarinic receptor agonist methacholine (Mch). Airway responsiveness was assessed using whole body plethysmography. Changes in airway resistance were expressed as the enhanced pause (Penh), a dimensionless value that represents a function of the proportion of maximal expiratory to maximal inspiratory box pressure signals and of the timing of expiration. Mice were exposed to nebulized PBS and subsequently to increasing concentrations of nebulized MCh (12, 24, 48 mg/ml Mch) for 2 min in PBS. Recordings were performed for 3 min following each exposure. The obtained Penh values were averaged and expressed as the percentage of baseline following PBS exposure.
[0335] Results: 24 mg/ml and higher concentrations of the muscarinic receptor agonist methacholine increased airway resistance above baseline. The airway responsiveness was assessed 8 hours post-treatment on Day 3. All animals treated with DAS181 showed reduced airway resistance at 24 mg/ml of Mch, with average percentage change Penh values (representing increasein airway resistance over baseline) as follows:
[0336] Control (PBS): 550-560%
[0337] 10 ng/kg DAS181: 525-530%
[0338] 0.1 .mu.g/kg DAS181: 450%
[0339] 1 .mu.g/kg DAS181: 525%
[0340] 10 .mu.g/kg DAS181: 225%
[0341] 0.1 mg/kg DAS181: 200%
[0342] As seen from the above results, dose levels of 0.01 or 0.1 mg/kg of DAS181 significantly reduced airway resistance in response to Mch.
[0343] Conclusion: The results demonstrate that reduced airway resistance in response to intranasal treatment with DAS181 is dose-dependent.
Example 10. Effect of DAS181 on M2 and M3 Muscarinic Receptor Signaling In Vitro
[0344] The objective of the study was to assess muscarinic receptor desialylation as a potential mechanism for airway protection, using an in vitro model. Human chem-1 cells (Millipore) stably transfected with M2 or M3 muscarinic receptor were treated with 0.4, 2 or 10 .mu.M of DAS181 for 30 min at 37.degree. C. prior to the addition of a receptor agonist (acetylcholine; ACh). Receptor signaling was determined using a fluorescence reporter for intracellular calcium.
[0345] Results:
[0346] At all doses of DAS181 tested, treatment of human chem-1 cells with DAS181 increased the potency of agonist-mediated M2 receptor signaling and decreased the potency of agonist-mediated M3 receptor signaling. The results are shown below:
[0347] A. M2 Receptor
TABLE-US-00003 Treatment Predicted EC50 Potency Value (M) ACh alone (no DAS181) 390 nM ACh + 10 .mu.M DAS181 140 nM ACh + 2 .mu.M DAS181 130 nM ACh + 0.4 .mu.M DAS181 140 nM
[0348] B. M3 Receptor
TABLE-US-00004 Treatment Predicted EC50 Potency Value (M) ACh alone (no DAS181) 6.3 nM ACh + 10 .mu.M DAS181 270 nM ACh + 2 .mu.M DAS181 51 nM ACh + 0.4 .mu.M DAS181 110 nM
[0349] Conclusion:
[0350] The results demonstrate that DAS181 increases signaling through the M2 muscarinic receptor and decreases signaling through the M3 muscarinic receptor. Thus, the DAS181-mediated responses could be indicative of positive allosteric modulation of the M2, and either antagonist or negative modulation M3. Desialylation may offer airway protection by reducing the stimulatory signal as well as enhancing the inhibitory signal mediated by muscarinic receptors.
Example 11. Therapeutic Efficacy of DAS181 Microparticle Formulations Against Parainfluenza
[0351] The efficacy of DAS181 was tested against parainfluenza virus (NV), which is an acute respiratory infection. A 63 year old female patient tested positive for PIV on Jul. 14, 2010 and Jul. 21, 2010; shedding of the PIV antigen in nasal swabs and sputum samples collected from the patient on those days was detected by PCR.
[0352] The patient was treated on Jul. 23, 24 and 25, 2010 with one capsule (10 mg delivered dose) a day of a dry powder formulation of DAS181 whose components and wt/wt % in the composition are as follows: DAS181: 64.54-64.69%; Histidine free base: 4.32-4.60%; Histidine HCl: 5.85-6.27%; Trehalose: 9.06-9.68%; Magnesium sulfate: 4.66-5.84%; Calcium chloride: 0.19%; Sodium acetate: 0.04-0.05%; Acetic acid: 0.02%; Water: 10%; Isopropanol: trace amounts. Each capsule contained 13 mg of the dry powder in a type 3 clear HPMC capsule (Capsugel), giving a delivered dose of 10 mg. The patient was administered one capsule a day for three days (Jul. 23, Jul. 24 and Jul. 25, 2010) by inhalation. The patient tested positive for PIV on the day after completion of the treatment (July 26), and tested negative for PIV on the fifth day following treatment (Jul. 30, 2010). The results demonstrate the effectiveness of DAS181 against parainfluenza, i.e., a respiratory infection of the upper respiratory tract.
Example 12. Therapeutic Efficacy of DAS181 Microparticle Formulations Against Asthma
[0353] The efficacy of the DAS181 microparticle formulation used in Example 9 above was tested against asthma. A 20 year old male Caucasian asthma patient was tested for changes in airflow prior to and 1 hour after oral administration of a 10 mg delivered dose (13 mg capsule) of Formulation A, as measured by FEV1 (forced expiratory volume of air in 1 second). Prior to administration of the drug, the FEV1 of the patient was at 82% of the predicted normal lung function. One hour after administration of the drug, the FEV1 of the patient indicated a clinically significant improvement in lung function, with a 10% increase in value to 92%. The results demonstrate that a DAS181 formulation can be effective against asthma, i.e., a non-infectious respiratory disorder affecting the central to upper respiratory tract.
OTHER EMBODIMENTS
[0354] It is to be understood that while the invention has been described in conjunction with the detailed description thereof, the foregoing description is intended to illustrate and not limit the scope of the invention, which is defined by the scope of the appended claims. Other aspects, advantages, and modifications are within the scope of the following claims.
Sequence CWU 1
1
39158PRTBos taurus 1Arg Pro Asp Phe Cys Leu Glu Pro Pro Tyr Thr Gly
Pro Cys Lys Ala1 5 10 15Arg Ile Ile Arg Tyr Phe Tyr Asn Ala Lys Ala
Gly Leu Cys Gln Thr 20 25 30Phe Val Tyr Gly Gly Cys Arg Ala Lys Arg
Asn Asn Phe Lys Ser Ala 35 40 45Glu Asp Cys Met Arg Thr Cys Gly Gly
Ala 50 55224PRTHomo sapiens 2Asn Gly Arg Arg Ile Cys Leu Asp Leu
Gln Ala Pro Leu Tyr Lys Lys1 5 10 15Ile Ile Lys Lys Leu Leu Glu Ser
20327PRTHomo sapiens 3Gly Arg Glu Leu Cys Leu Asp Pro Lys Glu Asn
Trp Val Gln Arg Val1 5 10 15Val Glu Lys Phe Leu Lys Arg Ala Glu Asn
Ser 20 25434PRTHomo sapiens 4Gln Ile His Phe Phe Phe Ala Lys Leu
Asn Cys Arg Leu Tyr Arg Lys1 5 10 15Ala Asn Lys Ser Ser Lys Leu Val
Ser Ala Asn Arg Leu Phe Gly Asp 20 25 30Lys Ser534PRTHomo sapiens
5Glu Leu Arg Val Arg Leu Ala Ser His Leu Arg Lys Leu Arg Lys Arg1 5
10 15Leu Leu Arg Asp Ala Asp Asp Leu Gln Lys Arg Leu Ala Val Tyr
Gln 20 25 30Ala Gly612PRTHomo sapiens 6Arg Arg Leu Arg Arg Met Glu
Ser Glu Ser Glu Ser1 5 10721PRTHomo sapiens 7Lys Arg Lys Lys Lys
Gly Gly Lys Asn Gly Lys Asn Arg Arg Asn Arg1 5 10 15Lys Lys Lys Asn
Pro 208379PRTHomo sapiens 8Met Ala Ser Leu Pro Val Leu Gln Lys Glu
Ser Val Phe Gln Ser Gly1 5 10 15Ala His Ala Tyr Arg Ile Pro Ala Leu
Leu Tyr Leu Pro Gly Gln Gln 20 25 30Ser Leu Leu Ala Phe Ala Glu Gln
Arg Ala Ser Lys Lys Asp Glu His 35 40 45Ala Glu Leu Ile Val Leu Arg
Arg Gly Asp Tyr Asp Ala Pro Thr His 50 55 60Gln Val Gln Trp Gln Ala
Gln Glu Val Val Ala Gln Ala Arg Leu Asp65 70 75 80Gly His Arg Ser
Met Asn Pro Cys Pro Leu Tyr Asp Ala Gln Thr Gly 85 90 95Thr Leu Phe
Leu Phe Phe Ile Ala Ile Pro Gly Gln Val Thr Glu Gln 100 105 110Gln
Gln Leu Gln Thr Arg Ala Asn Val Thr Arg Leu Cys Gln Val Thr 115 120
125Ser Thr Asp His Gly Arg Thr Trp Ser Ser Pro Arg Asp Leu Thr Asp
130 135 140Ala Ala Ile Gly Pro Ala Tyr Arg Glu Trp Ser Thr Phe Ala
Val Gly145 150 155 160Pro Gly His Cys Leu Gln Leu Asn Asp Arg Ala
Arg Ser Leu Val Val 165 170 175Pro Ala Tyr Ala Tyr Arg Lys Leu His
Pro Ile Gln Arg Pro Ile Pro 180 185 190Ser Ala Phe Cys Phe Leu Ser
His Asp His Gly Arg Thr Trp Ala Arg 195 200 205Gly His Phe Val Ala
Gln Asp Thr Leu Glu Cys Gln Val Ala Glu Val 210 215 220Glu Thr Gly
Glu Gln Arg Val Val Thr Leu Asn Ala Arg Ser His Leu225 230 235
240Arg Ala Arg Val Gln Ala Gln Ser Thr Asn Asp Gly Leu Asp Phe Gln
245 250 255Glu Ser Gln Leu Val Lys Lys Leu Val Glu Pro Pro Pro Gln
Gly Cys 260 265 270Gln Gly Ser Val Ile Ser Phe Pro Ser Pro Arg Ser
Gly Pro Gly Ser 275 280 285Pro Gln Trp Leu Leu Tyr Thr His Pro Thr
His Ser Trp Gln Arg Ala 290 295 300Asp Leu Gly Ala Tyr Leu Asn Pro
Arg Pro Pro Ala Pro Glu Ala Trp305 310 315 320Ser Glu Pro Val Leu
Leu Ala Lys Gly Ser Cys Ala Tyr Ser Asp Leu 325 330 335Gln Ser Met
Gly Thr Gly Pro Asp Gly Ser Pro Leu Phe Gly Cys Leu 340 345 350Tyr
Glu Ala Asn Asp Tyr Glu Glu Ile Val Phe Leu Met Phe Thr Leu 355 360
365Lys Gln Ala Phe Pro Ala Glu Tyr Leu Pro Gln 370 3759424PRTHomo
sapiens 9Leu Ala Gly Gly Ser Val Arg Trp Gly Ala Leu His Val Leu
Gly Thr1 5 10 15Ala Ala Leu Ala Glu His Arg Ser Met Asn Pro Cys Pro
Val His Asp 20 25 30Ala Gly Thr Gly Thr Val Phe Leu Phe Phe Ile Ala
Val Leu Gly His 35 40 45Thr Pro Glu Ala Val Gln Ile Ala Thr Gly Arg
Asn Ala Ala Arg Leu 50 55 60Cys Cys Val Ala Ser Arg Asp Ala Gly Leu
Ser Trp Gly Ser Ala Arg65 70 75 80Asp Leu Thr Glu Glu Ala Ile Gly
Gly Ala Val Gln Asp Trp Ala Thr 85 90 95Phe Ala Val Gly Pro Gly His
Gly Val Gln Leu Pro Ser Gly Arg Leu 100 105 110Leu Val Pro Ala Tyr
Thr Tyr Arg Val Asp Arg Leu Glu Cys Phe Gly 115 120 125Lys Ile Cys
Arg Thr Ser Pro His Ser Phe Ala Phe Tyr Ser Asp Asp 130 135 140His
Gly Arg Thr Trp Arg Cys Gly Gly Leu Val Pro Asn Leu Arg Ser145 150
155 160Gly Glu Cys Gln Leu Ala Ala Val Asp Gly Gly Gln Ala Gly Ser
Phe 165 170 175Leu Tyr Cys Asn Ala Arg Ser Pro Leu Gly Ser Arg Val
Gln Ala Leu 180 185 190Ser Thr Asp Glu Gly Thr Ser Phe Leu Pro Ala
Glu Arg Val Ala Ser 195 200 205Leu Pro Glu Thr Ala Trp Gly Cys Gln
Gly Ser Ile Val Gly Phe Pro 210 215 220Ala Pro Ala Pro Asn Arg Pro
Arg Asp Asp Ser Trp Ser Val Gly Pro225 230 235 240Arg Ser Pro Leu
Gln Pro Pro Leu Leu Gly Pro Gly Val His Glu Pro 245 250 255Pro Glu
Glu Ala Ala Val Asp Pro Arg Gly Gly Gln Val Pro Gly Gly 260 265
270Pro Phe Ser Arg Leu Gln Pro Arg Gly Asp Gly Pro Arg Gln Pro Gly
275 280 285Pro Arg Pro Gly Val Ser Gly Asp Val Gly Ser Trp Thr Leu
Ala Leu 290 295 300Pro Met Pro Phe Ala Ala Pro Pro Gln Ser Pro Thr
Trp Leu Leu Tyr305 310 315 320Ser His Pro Val Gly Arg Arg Ala Arg
Leu His Met Gly Ile Arg Leu 325 330 335Ser Gln Ser Pro Leu Asp Pro
Arg Ser Trp Thr Glu Pro Trp Val Ile 340 345 350Tyr Glu Gly Pro Ser
Gly Tyr Ser Asp Leu Ala Ser Ile Gly Pro Ala 355 360 365Pro Glu Gly
Gly Leu Val Phe Ala Cys Leu Tyr Glu Ser Gly Ala Arg 370 375 380Thr
Ser Tyr Asp Glu Ile Ser Phe Cys Thr Phe Ser Leu Arg Glu Val385 390
395 400Leu Glu Asn Val Pro Ala Ser Pro Lys Pro Pro Asn Leu Gly Asp
Lys 405 410 415Pro Arg Gly Cys Cys Trp Pro Ser 420105PRTArtificial
SequenceSynthetic Construct 10Gly Gly Gly Gly Ser1
5112706DNAActinomyces viscosus 11atgacatcgc atagtccttt ctcccggagg
cgcctgccgg ccctcctggg ctccctgcca 60ctggccgcca ccggcctgat cgccgccgca
cccccggcgc acgccgtccc cacgtctgac 120ggcctggccg acgtcaccat
cacgcaggtg aacgcgcccg cggacggcct ctactccgtc 180ggcgatgtca
tgaccttcaa catcaccctg accaacacca gcggcgaggc ccactcctac
240gccccggcct cgacgaacct gtccgggaac gtctccaagt gccggtggcg
caacgtcccg 300gccgggacga ccaagaccga ctgcaccggc ctggccacgc
acacggtgac cgccgaggac 360ctcaaggccg gtggcttcac cccgcagatc
gcctacgagg tcaaggccgt ggagtacgcc 420gggaaggccc tgagcacccc
ggagacgatc aagggcgcga cgagcccagt caaggccaac 480tcgctgcggg
tcgagtcgat cacgccgtcg tcgagccagg agaactacaa gctgggcgac
540accgtcagct acacggtgcg cgtgcgctcg gtgtcggaca agacgatcaa
cgtcgccgcc 600accgaatcct ccttcgacga cctgggccgc cagtgccact
ggggcggcct caagccgggc 660aagggcgccg tctacaactg caagccgctc
acccacacga tcacgcaagc cgacgtcgac 720gccggccgct ggacgccatc
gatcaccctg acggccaccg gaaccgacgg cgccaccctc 780cagacgctca
ccgccaccgg caacccgatc aacgtcgtcg gcgaccaccc gcaggccacg
840cccgcaccgg cgcccgacgc gagcacggag ctgccggcct caatgagcca
ggcccagcac 900ctggccgcca acacggccac cgacaactac cgcatcccgg
cgatcaccac cgcccccaat 960ggggacctgc tcatctccta cgacgagcgc
ccgaaggaca acggcaacgg cggcagcgac 1020gcccccaacc cgaaccacat
cgtccagcgc cgctccaccg acggcggcaa gacctggtcg 1080gcgcccacct
acatccacca gggcacggag accggcaaga aggtcggcta ctccgacccg
1140agctacgtcg tcgatcacca gacgggcacg atcttcaact tccacgtcaa
gtcctacgac 1200cagggctggg gcggctcgcg cggcggcacc gacccggaga
accggggcat catccaggcc 1260gaggtgtcga cctccacgga caacggctgg
acctggacgc accgcacgat caccgcggac 1320atcacgaagg acaagccgtg
gaccgcgcgt ttcgcggcct cgggccaggg catccagatt 1380cagcacgggc
cccacgccgg gcgcctggtg cagcagtaca cgatcaggac cgccggcggc
1440gcggtgcagg ccgtctcggt ctactccgac gaccacggga agacgtggca
ggccggcacg 1500ccgatcggga ccggcatgga tgagaacaag gtcgttgagc
tctccgacgg ctccctcatg 1560ctcaactcgc gcgcctcgga tggctccggc
ttccgcaagg tggcccactc caccgacggt 1620gggcagacct ggagcgagcc
ggtgtccgac aagaacctgc ccgactcggt ggacaacgcc 1680cagatcatcc
gagccttccc gaacgccgcg ccggacgacc cgcgcgccaa ggtgctgctg
1740ctgagccact caccgaaccc gcggccgtgg tcgcgtgacc gcggcaccat
ctcgatgtcc 1800tgcgacgacg gcgcctcctg gacgaccagc aaggtcttcc
acgagccctt cgtcggatac 1860acgacgatcg cggtgcagtc cgacggcagc
atcgggctgc tcagcgagga cgcccacaac 1920ggcgccgact acggcggcat
ctggtaccgc aacttcacga tgaactggct cggcgagcag 1980tgcggccaga
agccggcgga gccgagcccg gcgccgtcgc cgacggcggc accctcagcg
2040gcaccgacgg agaagccggc cccgtcggcc gcgccgagcg ctgagcccac
gcaggcaccg 2100gcaccatcct ccgcgcccga gccgagcgct gcgcccgagc
cgagcagcgc cccggcgccg 2160gagcccacga ccgctccgag cacggagccc
acaccggctc ctgcgcccag ctccgcacct 2220gagcagaccg atgggccgac
cgctgcgccc gcaccggaga cgtcctctgc accggccgcc 2280gaaccgacgc
aggccccgac ggtggcgcct tctgttgagc ccacgcaggc tccgggtgcg
2340cagccgagct cagcacccaa gccgggggcg acgggtcggg ccccgtcggt
ggtgaacccg 2400aaggcgaccg gggcggcgac ggagcctggg acgccgtcat
cgagcgcgag cccggcaccg 2460agccggaacg cggcgccgac gccgaagccg
ggcatggagc ccgatgagat tgatcggccg 2520tctgacggca ccatggcgca
gccgaccggt ggcgccagcg cgccgagtgc cgcgccgacg 2580caggcggcga
aggccggcag caggctgtct cgcacgggga ccaacgcgct gctgatcctg
2640ggccttgcgg gtgtcgcggt tgtcggcggg tacctgctgc tgcgggctcg
ccgttcgaag 2700aactga 270612901PRTActinomyces viscosus 12Met Thr
Ser His Ser Pro Phe Ser Arg Arg Arg Leu Pro Ala Leu Leu1 5 10 15Gly
Ser Leu Pro Leu Ala Ala Thr Gly Leu Ile Ala Ala Ala Pro Pro 20 25
30Ala His Ala Val Pro Thr Ser Asp Gly Leu Ala Asp Val Thr Ile Thr
35 40 45Gln Val Asn Ala Pro Ala Asp Gly Leu Tyr Ser Val Gly Asp Val
Met 50 55 60Thr Phe Asn Ile Thr Leu Thr Asn Thr Ser Gly Glu Ala His
Ser Tyr65 70 75 80Ala Pro Ala Ser Thr Asn Leu Ser Gly Asn Val Ser
Lys Cys Arg Trp 85 90 95Arg Asn Val Pro Ala Gly Thr Thr Lys Thr Asp
Cys Thr Gly Leu Ala 100 105 110Thr His Thr Val Thr Ala Glu Asp Leu
Lys Ala Gly Gly Phe Thr Pro 115 120 125Gln Ile Ala Tyr Glu Val Lys
Ala Val Glu Tyr Ala Gly Lys Ala Leu 130 135 140Ser Thr Pro Glu Thr
Ile Lys Gly Ala Thr Ser Pro Val Lys Ala Asn145 150 155 160Ser Leu
Arg Val Glu Ser Ile Thr Pro Ser Ser Ser Gln Glu Asn Tyr 165 170
175Lys Leu Gly Asp Thr Val Ser Tyr Thr Val Arg Val Arg Ser Val Ser
180 185 190Asp Lys Thr Ile Asn Val Ala Ala Thr Glu Ser Ser Phe Asp
Asp Leu 195 200 205Gly Arg Gln Cys His Trp Gly Gly Leu Lys Pro Gly
Lys Gly Ala Val 210 215 220Tyr Asn Cys Lys Pro Leu Thr His Thr Ile
Thr Gln Ala Asp Val Asp225 230 235 240Ala Gly Arg Trp Thr Pro Ser
Ile Thr Leu Thr Ala Thr Gly Thr Asp 245 250 255Gly Ala Thr Leu Gln
Thr Leu Thr Ala Thr Gly Asn Pro Ile Asn Val 260 265 270Val Gly Asp
His Pro Gln Ala Thr Pro Ala Pro Ala Pro Asp Ala Ser 275 280 285Thr
Glu Leu Pro Ala Ser Met Ser Gln Ala Gln His Leu Ala Ala Asn 290 295
300Thr Ala Thr Asp Asn Tyr Arg Ile Pro Ala Ile Thr Thr Ala Pro
Asn305 310 315 320Gly Asp Leu Leu Ile Ser Tyr Asp Glu Arg Pro Lys
Asp Asn Gly Asn 325 330 335Gly Gly Ser Asp Ala Pro Asn Pro Asn His
Ile Val Gln Arg Arg Ser 340 345 350Thr Asp Gly Gly Lys Thr Trp Ser
Ala Pro Thr Tyr Ile His Gln Gly 355 360 365Thr Glu Thr Gly Lys Lys
Val Gly Tyr Ser Asp Pro Ser Tyr Val Val 370 375 380Asp His Gln Thr
Gly Thr Ile Phe Asn Phe His Val Lys Ser Tyr Asp385 390 395 400Gln
Gly Trp Gly Gly Ser Arg Gly Gly Thr Asp Pro Glu Asn Arg Gly 405 410
415Ile Ile Gln Ala Glu Val Ser Thr Ser Thr Asp Asn Gly Trp Thr Trp
420 425 430Thr His Arg Thr Ile Thr Ala Asp Ile Thr Lys Asp Lys Pro
Trp Thr 435 440 445Ala Arg Phe Ala Ala Ser Gly Gln Gly Ile Gln Ile
Gln His Gly Pro 450 455 460His Ala Gly Arg Leu Val Gln Gln Tyr Thr
Ile Arg Thr Ala Gly Gly465 470 475 480Ala Val Gln Ala Val Ser Val
Tyr Ser Asp Asp His Gly Lys Thr Trp 485 490 495Gln Ala Gly Thr Pro
Ile Gly Thr Gly Met Asp Glu Asn Lys Val Val 500 505 510Glu Leu Ser
Asp Gly Ser Leu Met Leu Asn Ser Arg Ala Ser Asp Gly 515 520 525Ser
Gly Phe Arg Lys Val Ala His Ser Thr Asp Gly Gly Gln Thr Trp 530 535
540Ser Glu Pro Val Ser Asp Lys Asn Leu Pro Asp Ser Val Asp Asn
Ala545 550 555 560Gln Ile Ile Arg Ala Phe Pro Asn Ala Ala Pro Asp
Asp Pro Arg Ala 565 570 575Lys Val Leu Leu Leu Ser His Ser Pro Asn
Pro Arg Pro Trp Ser Arg 580 585 590Asp Arg Gly Thr Ile Ser Met Ser
Cys Asp Asp Gly Ala Ser Trp Thr 595 600 605Thr Ser Lys Val Phe His
Glu Pro Phe Val Gly Tyr Thr Thr Ile Ala 610 615 620Val Gln Ser Asp
Gly Ser Ile Gly Leu Leu Ser Glu Asp Ala His Asn625 630 635 640Gly
Ala Asp Tyr Gly Gly Ile Trp Tyr Arg Asn Phe Thr Met Asn Trp 645 650
655Leu Gly Glu Gln Cys Gly Gln Lys Pro Ala Glu Pro Ser Pro Ala Pro
660 665 670Ser Pro Thr Ala Ala Pro Ser Ala Ala Pro Thr Glu Lys Pro
Ala Pro 675 680 685Ser Ala Ala Pro Ser Ala Glu Pro Thr Gln Ala Pro
Ala Pro Ser Ser 690 695 700Ala Pro Glu Pro Ser Ala Ala Pro Glu Pro
Ser Ser Ala Pro Ala Pro705 710 715 720Glu Pro Thr Thr Ala Pro Ser
Thr Glu Pro Thr Pro Ala Pro Ala Pro 725 730 735Ser Ser Ala Pro Glu
Gln Thr Asp Gly Pro Thr Ala Ala Pro Ala Pro 740 745 750Glu Thr Ser
Ser Ala Pro Ala Ala Glu Pro Thr Gln Ala Pro Thr Val 755 760 765Ala
Pro Ser Val Glu Pro Thr Gln Ala Pro Gly Ala Gln Pro Ser Ser 770 775
780Ala Pro Lys Pro Gly Ala Thr Gly Arg Ala Pro Ser Val Val Asn
Pro785 790 795 800Lys Ala Thr Gly Ala Ala Thr Glu Pro Gly Thr Pro
Ser Ser Ser Ala 805 810 815Ser Pro Ala Pro Ser Arg Asn Ala Ala Pro
Thr Pro Lys Pro Gly Met 820 825 830Glu Pro Asp Glu Ile Asp Arg Pro
Ser Asp Gly Thr Met Ala Gln Pro 835 840 845Thr Gly Gly Ala Ser Ala
Pro Ser Ala Ala Pro Thr Gln Ala Ala Lys 850 855 860Ala Gly Ser Arg
Leu Ser Arg Thr Gly Thr Asn Ala Leu Leu Ile Leu865 870 875 880Gly
Leu Ala Gly Val Ala Val Val Gly Gly Tyr Leu Leu Leu Arg Ala 885 890
895Arg Arg Ser Lys Asn 900131887DNAActinomyces viscosus
13ggcgaccacc cgcaggccac gcccgcaccg gcgcccgacg cgagcacgga gctgccggcc
60tcaatgagcc aggcccagca cctggccgcc aacacggcca ccgacaacta ccgcatcccg
120gcgatcacca ccgcccccaa tggggacctg ctcatctcct acgacgagcg
cccgaaggac 180aacggcaacg gcggcagcga cgcccccaac ccgaaccaca
tcgtccagcg ccgctccacc 240gacggcggca agacctggtc ggcgcccacc
tacatccacc agggcacgga gaccggcaag 300aaggtcggct actccgaccc
gagctacgtc gtcgatcacc agacgggcac gatcttcaac 360ttccacgtca
agtcctacga ccagggctgg
ggcggctcgc gcggcggcac cgacccggag 420aaccggggca tcatccaggc
cgaggtgtcg acctccacgg acaacggctg gacctggacg 480caccgcacga
tcaccgcgga catcacgaag gacaagccgt ggaccgcgcg tttcgcggcc
540tcgggccagg gcatccagat tcagcacggg ccccacgccg ggcgcctggt
gcagcagtac 600acgatcagga ccgccggcgg cgcggtgcag gccgtctcgg
tctactccga cgaccacggg 660aagacgtggc aggccggcac gccgatcggg
accggcatgg atgagaacaa ggtcgttgag 720ctctccgacg gctccctcat
gctcaactcg cgcgcctcgg atggctccgg cttccgcaag 780gtggcccact
ccaccgacgg tgggcagacc tggagcgagc cggtgtccga caagaacctg
840cccgactcgg tggacaacgc ccagatcatc cgagccttcc cgaacgccgc
gccggacgac 900ccgcgcgcca aggtgctgct gctgagccac tcaccgaacc
cgcggccgtg gtcgcgtgac 960cgcggcacca tctcgatgtc ctgcgacgac
ggcgcctcct ggacgaccag caaggtcttc 1020cacgagccct tcgtcggata
cacgacgatc gcggtgcagt ccgacggcag catcgggctg 1080ctcagcgagg
acgcccacaa cggcgccgac tacggcggca tctggtaccg caacttcacg
1140atgaactggc tcggcgagca gtgcggccag aagccggcgg agccgagccc
ggcgccgtcg 1200ccgacggcgg caccctcagc ggcaccgacg gagaagccgg
ccccgtcggc cgcgccgagc 1260gctgagccca cgcaggcacc ggcaccatcc
tccgcgcccg agccgagcgc tgcgcccgag 1320ccgagcagcg ccccggcgcc
ggagcccacg accgctccga gcacggagcc cacaccggct 1380cctgcgccca
gctccgcacc tgagcagacc gatgggccga ccgctgcgcc cgcaccggag
1440acgtcctctg caccggccgc cgaaccgacg caggccccga cggtggcgcc
ttctgttgag 1500cccacgcagg ctccgggtgc gcagccgagc tcagcaccca
agccgggggc gacgggtcgg 1560gccccgtcgg tggtgaaccc gaaggcgacc
ggggcggcga cggagcctgg gacgccgtca 1620tcgagcgcga gcccggcacc
gagccggaac gcggcgccga cgccgaagcc gggcatggag 1680cccgatgaga
ttgatcggcc gtctgacggc accatggcgc agccgaccgg tggcgccagc
1740gcgccgagtg ccgcgccgac gcaggcggcg aaggccggca gcaggctgtc
tcgcacgggg 1800accaacgcgc tgctgatcct gggccttgcg ggtgtcgcgg
ttgtcggcgg gtacctgctg 1860ctgcgggctc gccgttcgaa gaactga
188714628PRTActinomyces viscosus 14Gly Asp His Pro Gln Ala Thr Pro
Ala Pro Ala Pro Asp Ala Ser Thr1 5 10 15Glu Leu Pro Ala Ser Met Ser
Gln Ala Gln His Leu Ala Ala Asn Thr 20 25 30Ala Thr Asp Asn Tyr Arg
Ile Pro Ala Ile Thr Thr Ala Pro Asn Gly 35 40 45Asp Leu Leu Ile Ser
Tyr Asp Glu Arg Pro Lys Asp Asn Gly Asn Gly 50 55 60Gly Ser Asp Ala
Pro Asn Pro Asn His Ile Val Gln Arg Arg Ser Thr65 70 75 80Asp Gly
Gly Lys Thr Trp Ser Ala Pro Thr Tyr Ile His Gln Gly Thr 85 90 95Glu
Thr Gly Lys Lys Val Gly Tyr Ser Asp Pro Ser Tyr Val Val Asp 100 105
110His Gln Thr Gly Thr Ile Phe Asn Phe His Val Lys Ser Tyr Asp Gln
115 120 125Gly Trp Gly Gly Ser Arg Gly Gly Thr Asp Pro Glu Asn Arg
Gly Ile 130 135 140Ile Gln Ala Glu Val Ser Thr Ser Thr Asp Asn Gly
Trp Thr Trp Thr145 150 155 160His Arg Thr Ile Thr Ala Asp Ile Thr
Lys Asp Lys Pro Trp Thr Ala 165 170 175Arg Phe Ala Ala Ser Gly Gln
Gly Ile Gln Ile Gln His Gly Pro His 180 185 190Ala Gly Arg Leu Val
Gln Gln Tyr Thr Ile Arg Thr Ala Gly Gly Ala 195 200 205Val Gln Ala
Val Ser Val Tyr Ser Asp Asp His Gly Lys Thr Trp Gln 210 215 220Ala
Gly Thr Pro Ile Gly Thr Gly Met Asp Glu Asn Lys Val Val Glu225 230
235 240Leu Ser Asp Gly Ser Leu Met Leu Asn Ser Arg Ala Ser Asp Gly
Ser 245 250 255Gly Phe Arg Lys Val Ala His Ser Thr Asp Gly Gly Gln
Thr Trp Ser 260 265 270Glu Pro Val Ser Asp Lys Asn Leu Pro Asp Ser
Val Asp Asn Ala Gln 275 280 285Ile Ile Arg Ala Phe Pro Asn Ala Ala
Pro Asp Asp Pro Arg Ala Lys 290 295 300Val Leu Leu Leu Ser His Ser
Pro Asn Pro Arg Pro Trp Ser Arg Asp305 310 315 320Arg Gly Thr Ile
Ser Met Ser Cys Asp Asp Gly Ala Ser Trp Thr Thr 325 330 335Ser Lys
Val Phe His Glu Pro Phe Val Gly Tyr Thr Thr Ile Ala Val 340 345
350Gln Ser Asp Gly Ser Ile Gly Leu Leu Ser Glu Asp Ala His Asn Gly
355 360 365Ala Asp Tyr Gly Gly Ile Trp Tyr Arg Asn Phe Thr Met Asn
Trp Leu 370 375 380Gly Glu Gln Cys Gly Gln Lys Pro Ala Glu Pro Ser
Pro Ala Pro Ser385 390 395 400Pro Thr Ala Ala Pro Ser Ala Ala Pro
Thr Glu Lys Pro Ala Pro Ser 405 410 415Ala Ala Pro Ser Ala Glu Pro
Thr Gln Ala Pro Ala Pro Ser Ser Ala 420 425 430Pro Glu Pro Ser Ala
Ala Pro Glu Pro Ser Ser Ala Pro Ala Pro Glu 435 440 445Pro Thr Thr
Ala Pro Ser Thr Glu Pro Thr Pro Ala Pro Ala Pro Ser 450 455 460Ser
Ala Pro Glu Gln Thr Asp Gly Pro Thr Ala Ala Pro Ala Pro Glu465 470
475 480Thr Ser Ser Ala Pro Ala Ala Glu Pro Thr Gln Ala Pro Thr Val
Ala 485 490 495Pro Ser Val Glu Pro Thr Gln Ala Pro Gly Ala Gln Pro
Ser Ser Ala 500 505 510Pro Lys Pro Gly Ala Thr Gly Arg Ala Pro Ser
Val Val Asn Pro Lys 515 520 525Ala Thr Gly Ala Ala Thr Glu Pro Gly
Thr Pro Ser Ser Ser Ala Ser 530 535 540Pro Ala Pro Ser Arg Asn Ala
Ala Pro Thr Pro Lys Pro Gly Met Glu545 550 555 560Pro Asp Glu Ile
Asp Arg Pro Ser Asp Gly Thr Met Ala Gln Pro Thr 565 570 575Gly Gly
Ala Ser Ala Pro Ser Ala Ala Pro Thr Gln Ala Ala Lys Ala 580 585
590Gly Ser Arg Leu Ser Arg Thr Gly Thr Asn Ala Leu Leu Ile Leu Gly
595 600 605Leu Ala Gly Val Ala Val Val Gly Gly Tyr Leu Leu Leu Arg
Ala Arg 610 615 620Arg Ser Lys Asn625151182DNAArtificial
SequenceSynthetic Construct 15ggagatcatc cacaagctac accagcacct
gcaccagatg ctagcactga gctgccagca 60agcatgtctc aggctcagca tcttgcagca
aatacggcta ctgataatta tcgcattcca 120gcgattacaa ccgctccgaa
tggtgattta ctgattagct atgatgaacg gccgaaggac 180aatggaaatg
gtggttccga tgcccctaac ccgaatcata ttgttcagcg tcgctccaca
240gatggcggta aaacttggag cgcgccaacc tatattcatc agggtacgga
gactggcaag 300aaagtgggat attccgaccc ctcttatgtg gtggatcatc
aaaccggtac aatcttcaat 360tttcatgtga aatcatacga tcagggctgg
ggaggtagcc gtgggggaac agacccggaa 420aaccgcggga ttattcaggc
agaggtgtct acgagcacgg ataatggatg gacgtggaca 480catcgcacca
tcaccgcgga tattacgaaa gataaaccgt ggaccgcgcg ttttgcggcg
540tccggccaag gcattcagat ccagcatggg ccgcatgccg gccgtctggt
gcaacagtat 600accattcgta cggccggtgg agcggtgcag gctgtatcgg
tttattccga tgatcatggg 660aaaacgtggc aggctggcac cccgattggg
acgggtatgg atgaaaacaa agttgtagag 720ctgtctgacg gctctctgat
gctgaacagt cgtgcgtcgg acgggagcgg ctttcgtaag 780gttgcgcata
gcactgatgg tgggcagacc tggtccgaac cggtttcgga caaaaatttg
840ccggattcgg ttgataatgc ccagataatt cgtgcgtttc ctaatgctgc
ccccgatgac 900ccgcgcgcga aagtacttct tctgagtcat tccccaaatc
cacgtccgtg gtcccgggat 960cgtggtacga taagcatgtc atgtgatgac
ggggcctcat ggaccacttc caaagttttt 1020cacgaaccgt ttgtgggcta
cacgactatt gcagttcaga gtgatggaag catcggtctg 1080ctgtcggagg
acgcgcacaa tggcgctgat tatggtggca tctggtatcg taattttacg
1140atgaactggc tgggagaaca atgtggacaa aaacccgcgg aa
118216394PRTActinomyces viscosus 16Gly Asp His Pro Gln Ala Thr Pro
Ala Pro Ala Pro Asp Ala Ser Thr1 5 10 15Glu Leu Pro Ala Ser Met Ser
Gln Ala Gln His Leu Ala Ala Asn Thr 20 25 30Ala Thr Asp Asn Tyr Arg
Ile Pro Ala Ile Thr Thr Ala Pro Asn Gly 35 40 45Asp Leu Leu Ile Ser
Tyr Asp Glu Arg Pro Lys Asp Asn Gly Asn Gly 50 55 60Gly Ser Asp Ala
Pro Asn Pro Asn His Ile Val Gln Arg Arg Ser Thr65 70 75 80Asp Gly
Gly Lys Thr Trp Ser Ala Pro Thr Tyr Ile His Gln Gly Thr 85 90 95Glu
Thr Gly Lys Lys Val Gly Tyr Ser Asp Pro Ser Tyr Val Val Asp 100 105
110His Gln Thr Gly Thr Ile Phe Asn Phe His Val Lys Ser Tyr Asp Gln
115 120 125Gly Trp Gly Gly Ser Arg Gly Gly Thr Asp Pro Glu Asn Arg
Gly Ile 130 135 140Ile Gln Ala Glu Val Ser Thr Ser Thr Asp Asn Gly
Trp Thr Trp Thr145 150 155 160His Arg Thr Ile Thr Ala Asp Ile Thr
Lys Asp Lys Pro Trp Thr Ala 165 170 175Arg Phe Ala Ala Ser Gly Gln
Gly Ile Gln Ile Gln His Gly Pro His 180 185 190Ala Gly Arg Leu Val
Gln Gln Tyr Thr Ile Arg Thr Ala Gly Gly Ala 195 200 205Val Gln Ala
Val Ser Val Tyr Ser Asp Asp His Gly Lys Thr Trp Gln 210 215 220Ala
Gly Thr Pro Ile Gly Thr Gly Met Asp Glu Asn Lys Val Val Glu225 230
235 240Leu Ser Asp Gly Ser Leu Met Leu Asn Ser Arg Ala Ser Asp Gly
Ser 245 250 255Gly Phe Arg Lys Val Ala His Ser Thr Asp Gly Gly Gln
Thr Trp Ser 260 265 270Glu Pro Val Ser Asp Lys Asn Leu Pro Asp Ser
Val Asp Asn Ala Gln 275 280 285Ile Ile Arg Ala Phe Pro Asn Ala Ala
Pro Asp Asp Pro Arg Ala Lys 290 295 300Val Leu Leu Leu Ser His Ser
Pro Asn Pro Arg Pro Trp Ser Arg Asp305 310 315 320Arg Gly Thr Ile
Ser Met Ser Cys Asp Asp Gly Ala Ser Trp Thr Thr 325 330 335Ser Lys
Val Phe His Glu Pro Phe Val Gly Tyr Thr Thr Ile Ala Val 340 345
350Gln Ser Asp Gly Ser Ile Gly Leu Leu Ser Glu Asp Ala His Asn Gly
355 360 365Ala Asp Tyr Gly Gly Ile Trp Tyr Arg Asn Phe Thr Met Asn
Trp Leu 370 375 380Gly Glu Gln Cys Gly Gln Lys Pro Ala Glu385
390176PRTArtificial SequenceSynthetic Construct 17Met Val Lys Arg
Lys Lys1 5181281DNAArtificial SequenceSynthetic Construct
18ccatggttaa gcgcaaaaaa aaaggcggca aaaacggtaa aaatcgtcgt aaccgtaaga
60aaaaaaatcc tggagatcat ccacaagcta caccagcacc tgcaccagat gctagcactg
120agctgccagc aagcatgtct caggctcagc atcttgcagc aaatacggct
actgataatt 180atcgcattcc agcgattaca accgctccga atggtgattt
actgattagc tatgatgaac 240ggccgaagga caatggaaat ggtggttccg
atgcccctaa cccgaatcat attgttcagc 300gtcgctccac agatggcggt
aaaacttgga gcgcgccaac ctatattcat cagggtacgg 360agactggcaa
gaaagtggga tattccgacc cctcttatgt ggtggatcat caaaccggta
420caatcttcaa ttttcatgtg aaatcatacg atcagggctg gggaggtagc
cgtgggggaa 480cagacccgga aaaccgcggg attattcagg cagaggtgtc
tacgagcacg gataatggat 540ggacgtggac acatcgcacc atcaccgcgg
atattacgaa agataaaccg tggaccgcgc 600gttttgcggc gtccggccaa
ggcattcaga tccagcatgg gccgcatgcc ggccgtctgg 660tgcaacagta
taccattcgt acggccggtg gagcggtgca ggctgtatcg gtttattccg
720atgatcatgg gaaaacgtgg caggctggca ccccgattgg gacgggtatg
gatgaaaaca 780aagttgtaga gctgtctgac ggctctctga tgctgaacag
tcgtgcgtcg gacgggagcg 840gctttcgtaa ggttgcgcat agcactgatg
gtgggcagac ctggtccgaa ccggtttcgg 900acaaaaattt gccggattcg
gttgataatg cccagataat tcgtgcgttt cctaatgctg 960cccccgatga
cccgcgcgcg aaagtacttc ttctgagtca ttccccaaat ccacgtccgt
1020ggtcccggga tcgtggtacg ataagcatgt catgtgatga cggggcctca
tggaccactt 1080ccaaagtttt tcacgaaccg tttgtgggct acacgactat
tgcagttcag agtgatggaa 1140gcatcggtct gctgtcggag gacgcgcaca
atggcgctga ttatggtggc atctggtatc 1200gtaattttac gatgaactgg
ctgggagaac aatgtggaca aaaacccgcg gaataagctt 1260aaaaacccgc
ggaataagct t 128119444PRTArtificial SequenceSynthetic Construct
19Met Val Lys Arg Lys Lys Lys Gly Gly Lys Asn Gly Lys Asn Arg Arg1
5 10 15Asn Arg Lys Lys Lys Asn Pro Gly Asp His Pro Gln Ala Thr Pro
Ala 20 25 30Pro Ala Pro Asp Ala Ser Thr Glu Leu Pro Ala Ser Met Ser
Gln Ala 35 40 45Gln His Leu Ala Ala Asn Thr Ala Thr Asp Asn Tyr Arg
Ile Pro Ala 50 55 60Ile Thr Thr Ala Pro Asn Gly Asp Leu Leu Ile Ser
Tyr Asp Glu Arg65 70 75 80Pro Lys Asp Asn Gly Asn Gly Gly Ser Asp
Ala Pro Asn Pro Asn His 85 90 95Ile Val Gln Arg Arg Ser Thr Asp Gly
Gly Lys Thr Trp Ser Ala Pro 100 105 110Thr Tyr Ile His Gln Gly Thr
Glu Thr Gly Lys Lys Val Gly Tyr Ser 115 120 125Asp Pro Ser Tyr Val
Val Asp His Gln Thr Gly Thr Ile Phe Asn Phe 130 135 140His Val Lys
Ser Tyr Asp Gln Gly Trp Gly Gly Ser Arg Gly Gly Thr145 150 155
160Asp Pro Glu Asn Arg Gly Ile Ile Gln Ala Glu Val Ser Thr Ser Thr
165 170 175Asp Asn Gly Trp Thr Trp Thr His Arg Thr Ile Thr Ala Asp
Ile Thr 180 185 190Lys Asp Lys Pro Trp Thr Ala Arg Phe Ala Ala Ser
Gly Gln Gly Ile 195 200 205Gln Ile Gln His Gly Pro His Ala Gly Arg
Leu Val Gln Gln Tyr Thr 210 215 220Ile Arg Thr Ala Gly Gly Ala Val
Gln Ala Val Ser Val Tyr Ser Asp225 230 235 240Asp His Gly Lys Thr
Trp Gln Ala Gly Thr Pro Ile Gly Thr Gly Met 245 250 255Asp Glu Asn
Lys Val Val Glu Leu Ser Asp Gly Ser Leu Met Leu Asn 260 265 270Ser
Arg Ala Ser Asp Gly Ser Gly Phe Arg Lys Val Ala His Ser Thr 275 280
285Asp Gly Gly Gln Thr Trp Ser Glu Pro Val Ser Asp Lys Asn Leu Pro
290 295 300Asp Ser Val Asp Asn Ala Gln Ile Ile Arg Ala Phe Pro Asn
Ala Ala305 310 315 320Pro Asp Asp Pro Arg Ala Lys Val Leu Leu Leu
Ser His Ser Pro Asn 325 330 335Pro Arg Pro Trp Ser Arg Asp Arg Gly
Thr Ile Ser Met Ser Cys Asp 340 345 350Asp Gly Ala Ser Trp Thr Thr
Ser Lys Val Phe His Glu Pro Phe Val 355 360 365Gly Tyr Thr Thr Ile
Ala Val Gln Ser Asp Gly Ser Ile Gly Leu Leu 370 375 380Ser Glu Asp
Ala His Asn Gly Ala Asp Tyr Gly Gly Ile Trp Tyr Arg385 390 395
400Asn Phe Thr Met Asn Trp Leu Gly Glu Gln Cys Gly Gln Lys Pro Ala
405 410 415Glu Gly Ala Asp Tyr Gly Gly Ile Trp Tyr Arg Asn Phe Thr
Met Asn 420 425 430Trp Leu Gly Glu Gln Cys Gly Gln Lys Pro Ala Glu
435 440201248DNAArtificial SequenceSynthetic Construct 20atgggagatc
atccacaagc tacaccagca cctgcaccag atgctagcac tgagctgcca 60gcaagcatgt
ctcaggctca gcatcttgca gcaaatacgg ctactgataa ttatcgcatt
120ccagcgatta caaccgctcc gaatggtgat ttactgatta gctatgatga
acggccgaag 180gacaatggaa atggtggttc cgatgcccct aacccgaatc
atattgttca gcgtcgctcc 240acagatggcg gtaaaacttg gagcgcgcca
acctatattc atcagggtac ggagactggc 300aagaaagtgg gatattccga
cccctcttat gtggtggatc atcaaaccgg tacaatcttc 360aattttcatg
tgaaatcata cgatcagggc tggggaggta gccgtggggg aacagacccg
420gaaaaccgcg ggattattca ggcagaggtg tctacgagca cggataatgg
atggacgtgg 480acacatcgca ccatcaccgc ggatattacg aaagataaac
cgtggaccgc gcgttttgcg 540gcgtccggcc aaggcattca gatccagcat
gggccgcatg ccggccgtct ggtgcaacag 600tataccattc gtacggccgg
tggagcggtg caggctgtat cggtttattc cgatgatcat 660gggaaaacgt
ggcaggctgg caccccgatt gggacgggta tggatgaaaa caaagttgta
720gagctgtctg acggctctct gatgctgaac agtcgtgcgt cggacgggag
cggctttcgt 780aaggttgcgc atagcactga tggtgggcag acctggtccg
aaccggtttc ggacaaaaat 840ttgccggatt cggttgataa tgcccagata
attcgtgcgt ttcctaatgc tgcccccgat 900gacccgcgcg cgaaagtact
tcttctgagt cattccccaa atccacgtcc gtggtcccgg 960gatcgtggta
cgataagcat gtcatgtgat gacggggcct catggaccac ttccaaagtt
1020tttcacgaac cgtttgtggg ctacacgact attgcagttc agagtgatgg
aagcatcggt 1080ctgctgtcgg aggacgcgca caatggcgct gattatggtg
gcatctggta tcgtaatttt 1140acgatgaact ggctgggaga acaatgtgga
caaaaacccg cgaagcgcaa aaaaaaaggc 1200ggcaaaaacg gtaaaaatcg
tcgtaaccgt aagaaaaaaa atccttga 124821415PRTArtificial
SequenceSynthetic Construct 21Met Gly Asp His Pro Gln Ala Thr Pro
Ala Pro Ala Pro Asp Ala Ser1 5 10 15Thr Glu Leu Pro Ala Ser Met Ser
Gln Ala Gln His Leu Ala Ala Asn 20 25 30Thr Ala Thr Asp Asn Tyr Arg
Ile Pro Ala Ile Thr Thr Ala Pro Asn 35 40 45Gly Asp Leu Leu Ile Ser
Tyr Asp Glu Arg Pro Lys Asp Asn Gly Asn 50 55 60Gly Gly Ser Asp Ala
Pro Asn Pro Asn His Ile Val Gln Arg Arg Ser65 70 75 80Thr Asp Gly
Gly Lys Thr Trp Ser Ala Pro Thr Tyr Ile His
Gln Gly 85 90 95Thr Glu Thr Gly Lys Lys Val Gly Tyr Ser Asp Pro Ser
Tyr Val Val 100 105 110Asp His Gln Thr Gly Thr Ile Phe Asn Phe His
Val Lys Ser Tyr Asp 115 120 125Gln Gly Trp Gly Gly Ser Arg Gly Gly
Thr Asp Pro Glu Asn Arg Gly 130 135 140Ile Ile Gln Ala Glu Val Ser
Thr Ser Thr Asp Asn Gly Trp Thr Trp145 150 155 160Thr His Arg Thr
Ile Thr Ala Asp Ile Thr Lys Asp Lys Pro Trp Thr 165 170 175Ala Arg
Phe Ala Ala Ser Gly Gln Gly Ile Gln Ile Gln His Gly Pro 180 185
190His Ala Gly Arg Leu Val Gln Gln Tyr Thr Ile Arg Thr Ala Gly Gly
195 200 205Ala Val Gln Ala Val Ser Val Tyr Ser Asp Asp His Gly Lys
Thr Trp 210 215 220Gln Ala Gly Thr Pro Ile Gly Thr Gly Met Asp Glu
Asn Lys Val Val225 230 235 240Glu Leu Ser Asp Gly Ser Leu Met Leu
Asn Ser Arg Ala Ser Asp Gly 245 250 255Ser Gly Phe Arg Lys Val Ala
His Ser Thr Asp Gly Gly Gln Thr Trp 260 265 270Ser Glu Pro Val Ser
Asp Lys Asn Leu Pro Asp Ser Val Asp Asn Ala 275 280 285Gln Ile Ile
Arg Ala Phe Pro Asn Ala Ala Pro Asp Asp Pro Arg Ala 290 295 300Lys
Val Leu Leu Leu Ser His Ser Pro Asn Pro Arg Pro Trp Ser Arg305 310
315 320Asp Arg Gly Thr Ile Ser Met Ser Cys Asp Asp Gly Ala Ser Trp
Thr 325 330 335Thr Ser Lys Val Phe His Glu Pro Phe Val Gly Tyr Thr
Thr Ile Ala 340 345 350Val Gln Ser Asp Gly Ser Ile Gly Leu Leu Ser
Glu Asp Ala His Asn 355 360 365Gly Ala Asp Tyr Gly Gly Ile Trp Tyr
Arg Asn Phe Thr Met Asn Trp 370 375 380Leu Gly Glu Gln Cys Gly Gln
Lys Pro Ala Lys Arg Lys Lys Lys Gly385 390 395 400Gly Lys Asn Gly
Lys Asn Arg Arg Asn Arg Lys Lys Lys Asn Pro 405 410
415221293DNAArtificial SequenceSynthetic Construct 22atgggagatc
atccacaagc tacaccagca cctgcaccag atgctagcac tgagctgcca 60gcaagcatgt
ctcaggctca gcatcttgca gcaaatacgg ctactgataa ttatcgcatt
120ccagcgatta caaccgctcc gaatggtgat ttactgatta gctatgatga
acggccgaag 180gacaatggaa atggtggttc cgatgcccct aacccgaatc
atattgttca gcgtcgctcc 240acagatggcg gtaaaacttg gagcgcgcca
acctatattc atcagggtac ggagactggc 300aagaaagtgg gatattccga
cccctcttat gtggtggatc atcaaaccgg tacaatcttc 360aattttcatg
tgaaatcata cgatcagggc tggggaggta gccgtggggg aacagacccg
420gaaaaccgcg ggattattca ggcagaggtg tctacgagca cggataatgg
atggacgtgg 480acacatcgca ccatcaccgc ggatattacg aaagataaac
cgtggaccgc gcgttttgcg 540gcgtccggcc aaggcattca gatccagcat
gggccgcatg ccggccgtct ggtgcaacag 600tataccattc gtacggccgg
tggagcggtg caggctgtat cggtttattc cgatgatcat 660gggaaaacgt
ggcaggctgg caccccgatt gggacgggta tggatgaaaa caaagttgta
720gagctgtctg acggctctct gatgctgaac agtcgtgcgt cggacgggag
cggctttcgt 780aaggttgcgc atagcactga tggtgggcag acctggtccg
aaccggtttc ggacaaaaat 840ttgccggatt cggttgataa tgcccagata
attcgtgcgt ttcctaatgc tgcccccgat 900gacccgcgcg cgaaagtact
tcttctgagt cattccccaa atccacgtcc gtggtcccgg 960gatcgtggta
cgataagcat gtcatgtgat gacggggcct catggaccac ttccaaagtt
1020tttcacgaac cgtttgtggg ctacacgact attgcagttc agagtgatgg
aagcatcggt 1080ctgctgtcgg aggacgcgca caatggcgct gattatggtg
gcatctggta tcgtaatttt 1140acgatgaact ggctgggaga acaatgtgga
caaaaacccg cggaaccgag cccagcccct 1200agccctactg cagcaccgtc
cgctgcaaag cgcaaaaaaa aaggcggcaa aaacggtaaa 1260aatcgtcgta
accgtaagaa aaaaaatcct tga 129323430PRTArtificial SequenceSynthetic
Construct 23Met Gly Asp His Pro Gln Ala Thr Pro Ala Pro Ala Pro Asp
Ala Ser1 5 10 15Thr Glu Leu Pro Ala Ser Met Ser Gln Ala Gln His Leu
Ala Ala Asn 20 25 30Thr Ala Thr Asp Asn Tyr Arg Ile Pro Ala Ile Thr
Thr Ala Pro Asn 35 40 45Gly Asp Leu Leu Ile Ser Tyr Asp Glu Arg Pro
Lys Asp Asn Gly Asn 50 55 60Gly Gly Ser Asp Ala Pro Asn Pro Asn His
Ile Val Gln Arg Arg Ser65 70 75 80Thr Asp Gly Gly Lys Thr Trp Ser
Ala Pro Thr Tyr Ile His Gln Gly 85 90 95Thr Glu Thr Gly Lys Lys Val
Gly Tyr Ser Asp Pro Ser Tyr Val Val 100 105 110Asp His Gln Thr Gly
Thr Ile Phe Asn Phe His Val Lys Ser Tyr Asp 115 120 125Gln Gly Trp
Gly Gly Ser Arg Gly Gly Thr Asp Pro Glu Asn Arg Gly 130 135 140Ile
Ile Gln Ala Glu Val Ser Thr Ser Thr Asp Asn Gly Trp Thr Trp145 150
155 160Thr His Arg Thr Ile Thr Ala Asp Ile Thr Lys Asp Lys Pro Trp
Thr 165 170 175Ala Arg Phe Ala Ala Ser Gly Gln Gly Ile Gln Ile Gln
His Gly Pro 180 185 190His Ala Gly Arg Leu Val Gln Gln Tyr Thr Ile
Arg Thr Ala Gly Gly 195 200 205Ala Val Gln Ala Val Ser Val Tyr Ser
Asp Asp His Gly Lys Thr Trp 210 215 220Gln Ala Gly Thr Pro Ile Gly
Thr Gly Met Asp Glu Asn Lys Val Val225 230 235 240Glu Leu Ser Asp
Gly Ser Leu Met Leu Asn Ser Arg Ala Ser Asp Gly 245 250 255Ser Gly
Phe Arg Lys Val Ala His Ser Thr Asp Gly Gly Gln Thr Trp 260 265
270Ser Glu Pro Val Ser Asp Lys Asn Leu Pro Asp Ser Val Asp Asn Ala
275 280 285Gln Ile Ile Arg Ala Phe Pro Asn Ala Ala Pro Asp Asp Pro
Arg Ala 290 295 300Lys Val Leu Leu Leu Ser His Ser Pro Asn Pro Arg
Pro Trp Ser Arg305 310 315 320Asp Arg Gly Thr Ile Ser Met Ser Cys
Asp Asp Gly Ala Ser Trp Thr 325 330 335Thr Ser Lys Val Phe His Glu
Pro Phe Val Gly Tyr Thr Thr Ile Ala 340 345 350Val Gln Ser Asp Gly
Ser Ile Gly Leu Leu Ser Glu Asp Ala His Asn 355 360 365Gly Ala Asp
Tyr Gly Gly Ile Trp Tyr Arg Asn Phe Thr Met Asn Trp 370 375 380Leu
Gly Glu Gln Cys Gly Gln Lys Pro Ala Glu Pro Ser Pro Ala Pro385 390
395 400Ser Pro Thr Ala Ala Pro Ser Ala Ala Lys Arg Lys Lys Lys Gly
Gly 405 410 415Lys Asn Gly Lys Asn Arg Arg Asn Arg Lys Lys Lys Asn
Pro 420 425 430241203DNAArtificial SequenceSynthetic Construct
24atgggagagc tgccagcaag catgtctcag gctcagcatc ttgcagcaaa tacggctact
60gataattatc gcattccagc gattacaacc gctccgaatg gtgatttact gattagctat
120gatgaacggc cgaaggacaa tggaaatggt ggttccgatg cccctaaccc
gaatcatatt 180gttcagcgtc gctccacaga tggcggtaaa acttggagcg
cgccaaccta tattcatcag 240ggtacggaga ctggcaagaa agtgggatat
tccgacccct cttatgtggt ggatcatcaa 300accggtacaa tcttcaattt
tcatgtgaaa tcatacgatc agggctgggg aggtagccgt 360gggggaacag
acccggaaaa ccgcgggatt attcaggcag aggtgtctac gagcacggat
420aatggatgga cgtggacaca tcgcaccatc accgcggata ttacgaaaga
taaaccgtgg 480accgcgcgtt ttgcggcgtc cggccaaggc attcagatcc
agcatgggcc gcatgccggc 540cgtctggtgc aacagtatac cattcgtacg
gccggtggag cggtgcaggc tgtatcggtt 600tattccgatg atcatgggaa
aacgtggcag gctggcaccc cgattgggac gggtatggat 660gaaaacaaag
ttgtagagct gtctgacggc tctctgatgc tgaacagtcg tgcgtcggac
720gggagcggct ttcgtaaggt tgcgcatagc actgatggtg ggcagacctg
gtccgaaccg 780gtttcggaca aaaatttgcc ggattcggtt gataatgccc
agataattcg tgcgtttcct 840aatgctgccc ccgatgaccc gcgcgcgaaa
gtacttcttc tgagtcattc cccaaatcca 900cgtccgtggt cccgggatcg
tggtacgata agcatgtcat gtgatgacgg ggcctcatgg 960accacttcca
aagtttttca cgaaccgttt gtgggctaca cgactattgc agttcagagt
1020gatggaagca tcggtctgct gtcggaggac gcgcacaatg gcgctgatta
tggtggcatc 1080tggtatcgta attttacgat gaactggctg ggagaacaat
gtggacaaaa acccgcgaag 1140cgcaaaaaaa aaggcggcaa aaacggtaaa
aatcgtcgta accgtaagaa aaaaaatcct 1200tga 120325400PRTArtificial
SequenceSynthetic Construct 25Met Gly Glu Leu Pro Ala Ser Met Ser
Gln Ala Gln His Leu Ala Ala1 5 10 15Asn Thr Ala Thr Asp Asn Tyr Arg
Ile Pro Ala Ile Thr Thr Ala Pro 20 25 30Asn Gly Asp Leu Leu Ile Ser
Tyr Asp Glu Arg Pro Lys Asp Asn Gly 35 40 45Asn Gly Gly Ser Asp Ala
Pro Asn Pro Asn His Ile Val Gln Arg Arg 50 55 60Ser Thr Asp Gly Gly
Lys Thr Trp Ser Ala Pro Thr Tyr Ile His Gln65 70 75 80Gly Thr Glu
Thr Gly Lys Lys Val Gly Tyr Ser Asp Pro Ser Tyr Val 85 90 95Val Asp
His Gln Thr Gly Thr Ile Phe Asn Phe His Val Lys Ser Tyr 100 105
110Asp Gln Gly Trp Gly Gly Ser Arg Gly Gly Thr Asp Pro Glu Asn Arg
115 120 125Gly Ile Ile Gln Ala Glu Val Ser Thr Ser Thr Asp Asn Gly
Trp Thr 130 135 140Trp Thr His Arg Thr Ile Thr Ala Asp Ile Thr Lys
Asp Lys Pro Trp145 150 155 160Thr Ala Arg Phe Ala Ala Ser Gly Gln
Gly Ile Gln Ile Gln His Gly 165 170 175Pro His Ala Gly Arg Leu Val
Gln Gln Tyr Thr Ile Arg Thr Ala Gly 180 185 190Gly Ala Val Gln Ala
Val Ser Val Tyr Ser Asp Asp His Gly Lys Thr 195 200 205Trp Gln Ala
Gly Thr Pro Ile Gly Thr Gly Met Asp Glu Asn Lys Val 210 215 220Val
Glu Leu Ser Asp Gly Ser Leu Met Leu Asn Ser Arg Ala Ser Asp225 230
235 240Gly Ser Gly Phe Arg Lys Val Ala His Ser Thr Asp Gly Gly Gln
Thr 245 250 255Trp Ser Glu Pro Val Ser Asp Lys Asn Leu Pro Asp Ser
Val Asp Asn 260 265 270Ala Gln Ile Ile Arg Ala Phe Pro Asn Ala Ala
Pro Asp Asp Pro Arg 275 280 285Ala Lys Val Leu Leu Leu Ser His Ser
Pro Asn Pro Arg Pro Trp Ser 290 295 300Arg Asp Arg Gly Thr Ile Ser
Met Ser Cys Asp Asp Gly Ala Ser Trp305 310 315 320Thr Thr Ser Lys
Val Phe His Glu Pro Phe Val Gly Tyr Thr Thr Ile 325 330 335Ala Val
Gln Ser Asp Gly Ser Ile Gly Leu Leu Ser Glu Asp Ala His 340 345
350Asn Gly Ala Asp Tyr Gly Gly Ile Trp Tyr Arg Asn Phe Thr Met Asn
355 360 365Trp Leu Gly Glu Gln Cys Gly Gln Lys Pro Ala Lys Arg Lys
Lys Lys 370 375 380Gly Gly Lys Asn Gly Lys Asn Arg Arg Asn Arg Lys
Lys Lys Asn Pro385 390 395 400261248DNAArtificial SequenceSynthetic
Construct 26atgggagagc tgccagcaag catgtctcag gctcagcatc ttgcagcaaa
tacggctact 60gataattatc gcattccagc gattacaacc gctccgaatg gtgatttact
gattagctat 120gatgaacggc cgaaggacaa tggaaatggt ggttccgatg
cccctaaccc gaatcatatt 180gttcagcgtc gctccacaga tggcggtaaa
acttggagcg cgccaaccta tattcatcag 240ggtacggaga ctggcaagaa
agtgggatat tccgacccct cttatgtggt ggatcatcaa 300accggtacaa
tcttcaattt tcatgtgaaa tcatacgatc agggctgggg aggtagccgt
360gggggaacag acccggaaaa ccgcgggatt attcaggcag aggtgtctac
gagcacggat 420aatggatgga cgtggacaca tcgcaccatc accgcggata
ttacgaaaga taaaccgtgg 480accgcgcgtt ttgcggcgtc cggccaaggc
attcagatcc agcatgggcc gcatgccggc 540cgtctggtgc aacagtatac
cattcgtacg gccggtggag cggtgcaggc tgtatcggtt 600tattccgatg
atcatgggaa aacgtggcag gctggcaccc cgattgggac gggtatggat
660gaaaacaaag ttgtagagct gtctgacggc tctctgatgc tgaacagtcg
tgcgtcggac 720gggagcggct ttcgtaaggt tgcgcatagc actgatggtg
ggcagacctg gtccgaaccg 780gtttcggaca aaaatttgcc ggattcggtt
gataatgccc agataattcg tgcgtttcct 840aatgctgccc ccgatgaccc
gcgcgcgaaa gtacttcttc tgagtcattc cccaaatcca 900cgtccgtggt
cccgggatcg tggtacgata agcatgtcat gtgatgacgg ggcctcatgg
960accacttcca aagtttttca cgaaccgttt gtgggctaca cgactattgc
agttcagagt 1020gatggaagca tcggtctgct gtcggaggac gcgcacaatg
gcgctgatta tggtggcatc 1080tggtatcgta attttacgat gaactggctg
ggagaacaat gtggacaaaa acccgcggaa 1140ccgagcccag cccctagccc
tactgcagca ccgtccgctg caaagcgcaa aaaaaaaggc 1200ggcaaaaacg
gtaaaaatcg tcgtaaccgt aagaaaaaaa atccttga 124827415PRTArtificial
SequenceSynthetic Construct 27Met Gly Glu Leu Pro Ala Ser Met Ser
Gln Ala Gln His Leu Ala Ala1 5 10 15Asn Thr Ala Thr Asp Asn Tyr Arg
Ile Pro Ala Ile Thr Thr Ala Pro 20 25 30Asn Gly Asp Leu Leu Ile Ser
Tyr Asp Glu Arg Pro Lys Asp Asn Gly 35 40 45Asn Gly Gly Ser Asp Ala
Pro Asn Pro Asn His Ile Val Gln Arg Arg 50 55 60Ser Thr Asp Gly Gly
Lys Thr Trp Ser Ala Pro Thr Tyr Ile His Gln65 70 75 80Gly Thr Glu
Thr Gly Lys Lys Val Gly Tyr Ser Asp Pro Ser Tyr Val 85 90 95Val Asp
His Gln Thr Gly Thr Ile Phe Asn Phe His Val Lys Ser Tyr 100 105
110Asp Gln Gly Trp Gly Gly Ser Arg Gly Gly Thr Asp Pro Glu Asn Arg
115 120 125Gly Ile Ile Gln Ala Glu Val Ser Thr Ser Thr Asp Asn Gly
Trp Thr 130 135 140Trp Thr His Arg Thr Ile Thr Ala Asp Ile Thr Lys
Asp Lys Pro Trp145 150 155 160Thr Ala Arg Phe Ala Ala Ser Gly Gln
Gly Ile Gln Ile Gln His Gly 165 170 175Pro His Ala Gly Arg Leu Val
Gln Gln Tyr Thr Ile Arg Thr Ala Gly 180 185 190Gly Ala Val Gln Ala
Val Ser Val Tyr Ser Asp Asp His Gly Lys Thr 195 200 205Trp Gln Ala
Gly Thr Pro Ile Gly Thr Gly Met Asp Glu Asn Lys Val 210 215 220Val
Glu Leu Ser Asp Gly Ser Leu Met Leu Asn Ser Arg Ala Ser Asp225 230
235 240Gly Ser Gly Phe Arg Lys Val Ala His Ser Thr Asp Gly Gly Gln
Thr 245 250 255Trp Ser Glu Pro Val Ser Asp Lys Asn Leu Pro Asp Ser
Val Asp Asn 260 265 270Ala Gln Ile Ile Arg Ala Phe Pro Asn Ala Ala
Pro Asp Asp Pro Arg 275 280 285Ala Lys Val Leu Leu Leu Ser His Ser
Pro Asn Pro Arg Pro Trp Ser 290 295 300Arg Asp Arg Gly Thr Ile Ser
Met Ser Cys Asp Asp Gly Ala Ser Trp305 310 315 320Thr Thr Ser Lys
Val Phe His Glu Pro Phe Val Gly Tyr Thr Thr Ile 325 330 335Ala Val
Gln Ser Asp Gly Ser Ile Gly Leu Leu Ser Glu Asp Ala His 340 345
350Asn Gly Ala Asp Tyr Gly Gly Ile Trp Tyr Arg Asn Phe Thr Met Asn
355 360 365Trp Leu Gly Glu Gln Cys Gly Gln Lys Pro Ala Glu Pro Ser
Pro Ala 370 375 380Pro Ser Pro Thr Ala Ala Pro Ser Ala Ala Lys Arg
Lys Lys Lys Gly385 390 395 400Gly Lys Asn Gly Lys Asn Arg Arg Asn
Arg Lys Lys Lys Asn Pro 405 410 415281221DNAArtificial
SequenceSynthetic Construct 28ccatggggca tcaccatcac catcatctag
agggagatca tccacaagct acaccagcac 60ctgcaccaga tgctagcact gagctgccag
caagcatgtc tcaggctcag catcttgcag 120caaatacggc tactgataat
tatcgcattc cagcgattac aaccgctccg aatggtgatt 180tactgattag
ctatgatgaa cggccgaagg acaatggaaa tggtggttcc gatgccccta
240acccgaatca tattgttcag cgtcgctcca cagatggcgg taaaacttgg
agcgcgccaa 300cctatattca tcagggtacg gagactggca agaaagtggg
atattccgac ccctcttatg 360tggtggatca tcaaaccggt acaatcttca
attttcatgt gaaatcatac gatcagggct 420ggggaggtag ccgtggggga
acagacccgg aaaaccgcgg gattattcag gcagaggtgt 480ctacgagcac
ggataatgga tggacgtgga cacatcgcac catcaccgcg gatattacga
540aagataaacc gtggaccgcg cgttttgcgg cgtccggcca aggcattcag
atccagcatg 600ggccgcatgc cggccgtctg gtgcaacagt ataccattcg
tacggccggt ggagcggtgc 660aggctgtatc ggtttattcc gatgatcatg
ggaaaacgtg gcaggctggc accccgattg 720ggacgggtat ggatgaaaac
aaagttgtag agctgtctga cggctctctg atgctgaaca 780gtcgtgcgtc
ggacgggagc ggctttcgta aggttgcgca tagcactgat ggtgggcaga
840cctggtccga accggtttcg gacaaaaatt tgccggattc ggttgataat
gcccagataa 900ttcgtgcgtt tcctaatgct gcccccgatg acccgcgcgc
gaaagtactt cttctgagtc 960attccccaaa tccacgtccg tggtcccggg
atcgtggtac gataagcatg tcatgtgatg 1020acggggcctc atggaccact
tccaaagttt ttcacgaacc gtttgtgggc tacacgacta 1080ttgcagttca
gagtgatgga agcatcggtc tgctgtcgga ggacgcgcac aatggcgctg
1140attatggtgg catctggtat cgtaatttta cgatgaactg gctgggagaa
caatgtggac 1200aaaaacccgc ggaataagct t 122129404PRTArtificial
SequenceSynthetic Construct 29Met Gly His His His
His His His Leu Glu Gly Asp His Pro Gln Ala1 5 10 15Thr Pro Ala Pro
Ala Pro Asp Ala Ser Thr Glu Leu Pro Ala Ser Met 20 25 30Ser Gln Ala
Gln His Leu Ala Ala Asn Thr Ala Thr Asp Asn Tyr Arg 35 40 45Ile Pro
Ala Ile Thr Thr Ala Pro Asn Gly Asp Leu Leu Ile Ser Tyr 50 55 60Asp
Glu Arg Pro Lys Asp Asn Gly Asn Gly Gly Ser Asp Ala Pro Asn65 70 75
80Pro Asn His Ile Val Gln Arg Arg Ser Thr Asp Gly Gly Lys Thr Trp
85 90 95Ser Ala Pro Thr Tyr Ile His Gln Gly Thr Glu Thr Gly Lys Lys
Val 100 105 110Gly Tyr Ser Asp Pro Ser Tyr Val Val Asp His Gln Thr
Gly Thr Ile 115 120 125Phe Asn Phe His Val Lys Ser Tyr Asp Gln Gly
Trp Gly Gly Ser Arg 130 135 140Gly Gly Thr Asp Pro Glu Asn Arg Gly
Ile Ile Gln Ala Glu Val Ser145 150 155 160Thr Ser Thr Asp Asn Gly
Trp Thr Trp Thr His Arg Thr Ile Thr Ala 165 170 175Asp Ile Thr Lys
Asp Lys Pro Trp Thr Ala Arg Phe Ala Ala Ser Gly 180 185 190Gln Gly
Ile Gln Ile Gln His Gly Pro His Ala Gly Arg Leu Val Gln 195 200
205Gln Tyr Thr Ile Arg Thr Ala Gly Gly Ala Val Gln Ala Val Ser Val
210 215 220Tyr Ser Asp Asp His Gly Lys Thr Trp Gln Ala Gly Thr Pro
Ile Gly225 230 235 240Thr Gly Met Asp Glu Asn Lys Val Val Glu Leu
Ser Asp Gly Ser Leu 245 250 255Met Leu Asn Ser Arg Ala Ser Asp Gly
Ser Gly Phe Arg Lys Val Ala 260 265 270His Ser Thr Asp Gly Gly Gln
Thr Trp Ser Glu Pro Val Ser Asp Lys 275 280 285Asn Leu Pro Asp Ser
Val Asp Asn Ala Gln Ile Ile Arg Ala Phe Pro 290 295 300Asn Ala Ala
Pro Asp Asp Pro Arg Ala Lys Val Leu Leu Leu Ser His305 310 315
320Ser Pro Asn Pro Arg Pro Trp Ser Arg Asp Arg Gly Thr Ile Ser Met
325 330 335Ser Cys Asp Asp Gly Ala Ser Trp Thr Thr Ser Lys Val Phe
His Glu 340 345 350Pro Phe Val Gly Tyr Thr Thr Ile Ala Val Gln Ser
Asp Gly Ser Ile 355 360 365Gly Leu Leu Ser Glu Asp Ala His Asn Gly
Ala Asp Tyr Gly Gly Ile 370 375 380Trp Tyr Arg Asn Phe Thr Met Asn
Trp Leu Gly Glu Gln Cys Gly Gln385 390 395 400Lys Pro Ala
Glu301257DNAArtificial SequenceSynthetic Construct 30ccatgaagcg
caaaaaaaaa ggcggcaaaa acggtaaaaa tcgtcgtaac cgtaagaaaa 60aaaatcctgg
agatcatcca caagctacac cagcacctgc accagatgct agcactgagc
120tgccagcaag catgtctcag gctcagcatc ttgcagcaaa tacggctact
gataattatc 180gcattccagc gattacaacc gctccgaatg gtgatttact
gattagctat gatgaacggc 240cgaaggacaa tggaaatggt ggttccgatg
cccctaaccc gaatcatatt gttcagcgtc 300gctccacaga tggcggtaaa
acttggagcg cgccaaccta tattcatcag ggtacggaga 360ctggcaagaa
agtgggatat tccgacccct cttatgtggt ggatcatcaa accggtacaa
420tcttcaattt tcatgtgaaa tcatacgatc agggctgggg aggtagccgt
gggggaacag 480acccggaaaa ccgcgggatt attcaggcag aggtgtctac
gagcacggat aatggatgga 540cgtggacaca tcgcaccatc accgcggata
ttacgaaaga taaaccgtgg accgcgcgtt 600ttgcggcgtc cggccaaggc
attcagatcc agcatgggcc gcatgccggc cgtctggtgc 660aacagtatac
cattcgtacg gccggtggag cggtgcaggc tgtatcggtt tattccgatg
720atcatgggaa aacgtggcag gctggcaccc cgattgggac gggtatggat
gaaaacaaag 780ttgtagagct gtctgacggc tctctgatgc tgaacagtcg
tgcgtcggac gggagcggct 840ttcgtaaggt tgcgcatagc actgatggtg
ggcagacctg gtccgaaccg gtttcggaca 900aaaatttgcc ggattcggtt
gataatgccc agataattcg tgcgtttcct aatgctgccc 960ccgatgaccc
gcgcgcgaaa gtacttcttc tgagtcattc cccaaatcca cgtccgtggt
1020cccgggatcg tggtacgata agcatgtcat gtgatgacgg ggcctcatgg
accacttcca 1080aagtttttca cgaaccgttt gtgggctaca cgactattgc
agttcagagt gatggaagca 1140tcggtctgct gtcggaggac gcgcacaatg
gcgctgatta tggtggcatc tggtatcgta 1200attttacgat gaactggctg
ggagaacaat gtggacaaaa acccgcggaa taagctt 125731416PRTArtificial
SequenceSynthetic Construct 31Met Lys Arg Lys Lys Lys Gly Gly Lys
Asn Gly Lys Asn Arg Arg Asn1 5 10 15Arg Lys Lys Lys Asn Pro Gly Asp
His Pro Gln Ala Thr Pro Ala Pro 20 25 30Ala Pro Asp Ala Ser Thr Glu
Leu Pro Ala Ser Met Ser Gln Ala Gln 35 40 45His Leu Ala Ala Asn Thr
Ala Thr Asp Asn Tyr Arg Ile Pro Ala Ile 50 55 60Thr Thr Ala Pro Asn
Gly Asp Leu Leu Ile Ser Tyr Asp Glu Arg Pro65 70 75 80Lys Asp Asn
Gly Asn Gly Gly Ser Asp Ala Pro Asn Pro Asn His Ile 85 90 95Val Gln
Arg Arg Ser Thr Asp Gly Gly Lys Thr Trp Ser Ala Pro Thr 100 105
110Tyr Ile His Gln Gly Thr Glu Thr Gly Lys Lys Val Gly Tyr Ser Asp
115 120 125Pro Ser Tyr Val Val Asp His Gln Thr Gly Thr Ile Phe Asn
Phe His 130 135 140Val Lys Ser Tyr Asp Gln Gly Trp Gly Gly Ser Arg
Gly Gly Thr Asp145 150 155 160Pro Glu Asn Arg Gly Ile Ile Gln Ala
Glu Val Ser Thr Ser Thr Asp 165 170 175Asn Gly Trp Thr Trp Thr His
Arg Thr Ile Thr Ala Asp Ile Thr Lys 180 185 190Asp Lys Pro Trp Thr
Ala Arg Phe Ala Ala Ser Gly Gln Gly Ile Gln 195 200 205Ile Gln His
Gly Pro His Ala Gly Arg Leu Val Gln Gln Tyr Thr Ile 210 215 220Arg
Thr Ala Gly Gly Ala Val Gln Ala Val Ser Val Tyr Ser Asp Asp225 230
235 240His Gly Lys Thr Trp Gln Ala Gly Thr Pro Ile Gly Thr Gly Met
Asp 245 250 255Glu Asn Lys Val Val Glu Leu Ser Asp Gly Ser Leu Met
Leu Asn Ser 260 265 270Arg Ala Ser Asp Gly Ser Gly Phe Arg Lys Val
Ala His Ser Thr Asp 275 280 285Gly Gly Gln Thr Trp Ser Glu Pro Val
Ser Asp Lys Asn Leu Pro Asp 290 295 300Ser Val Asp Asn Ala Gln Ile
Ile Arg Ala Phe Pro Asn Ala Ala Pro305 310 315 320Asp Asp Pro Arg
Ala Lys Val Leu Leu Leu Ser His Ser Pro Asn Pro 325 330 335Arg Pro
Trp Ser Arg Asp Arg Gly Thr Ile Ser Met Ser Cys Asp Asp 340 345
350Gly Ala Ser Trp Thr Thr Ser Lys Val Phe His Glu Pro Phe Val Gly
355 360 365Tyr Thr Thr Ile Ala Val Gln Ser Asp Gly Ser Ile Gly Leu
Leu Ser 370 375 380Glu Asp Ala His Asn Gly Ala Asp Tyr Gly Gly Ile
Trp Tyr Arg Asn385 390 395 400Phe Thr Met Asn Trp Leu Gly Glu Gln
Cys Gly Gln Lys Pro Ala Glu 405 410 4153243DNAArtificial
SequenceSynthetic Constructmisc_feature17, 18, 20, 21n = A,T,C or G
32ttttcgtctc ccatgvnnvn naagcgcaaa aaaaaaggcg gca
433310PRTArtificial SequenceSynthetic ConstructVARIANT2, 3Xaa = Any
Amino Acid 33Met Xaa Xaa Lys Arg Lys Lys Lys Gly Gly1 5
10341272DNAArtificial SequenceSynthetic Construct 34ccatgaagcg
caaaaaaaaa ggcggcaaaa acggtaaaaa tcgtcgtaac cgtaagaaaa 60aaaatcctgg
tggtggtggt tctggagatc atccacaagc tacaccagca cctgcaccag
120atgctagcac tgagctgcca gcaagcatgt ctcaggctca gcatcttgca
gcaaatacgg 180ctactgataa ttatcgcatt ccagcgatta caaccgctcc
gaatggtgat ttactgatta 240gctatgatga acggccgaag gacaatggaa
atggtggttc cgatgcccct aacccgaatc 300atattgttca gcgtcgctcc
acagatggcg gtaaaacttg gagcgcgcca acctatattc 360atcagggtac
ggagactggc aagaaagtgg gatattccga cccctcttat gtggtggatc
420atcaaaccgg tacaatcttc aattttcatg tgaaatcata cgatcagggc
tggggaggta 480gccgtggggg aacagacccg gaaaaccgcg ggattattca
ggcagaggtg tctacgagca 540cggataatgg atggacgtgg acacatcgca
ccatcaccgc ggatattacg aaagataaac 600cgtggaccgc gcgttttgcg
gcgtccggcc aaggcattca gatccagcat gggccgcatg 660ccggccgtct
ggtgcaacag tataccattc gtacggccgg tggagcggtg caggctgtat
720cggtttattc cgatgatcat gggaaaacgt ggcaggctgg caccccgatt
gggacgggta 780tggatgaaaa caaagttgta gagctgtctg acggctctct
gatgctgaac agtcgtgcgt 840cggacgggag cggctttcgt aaggttgcgc
atagcactga tggtgggcag acctggtccg 900aaccggtttc ggacaaaaat
ttgccggatt cggttgataa tgcccagata attcgtgcgt 960ttcctaatgc
tgcccccgat gacccgcgcg cgaaagtact tcttctgagt cattccccaa
1020atccacgtcc gtggtcccgg gatcgtggta cgataagcat gtcatgtgat
gacggggcct 1080catggaccac ttccaaagtt tttcacgaac cgtttgtggg
ctacacgact attgcagttc 1140agagtgatgg aagcatcggt ctgctgtcgg
aggacgcgca caatggcgct gattatggtg 1200gcatctggta tcgtaatttt
acgatgaact ggctgggaga acaatgtgga caaaaacccg 1260cggaataagc tt
127235421PRTArtificial SequenceSynthetic Construct 35Met Lys Arg
Lys Lys Lys Gly Gly Lys Asn Gly Lys Asn Arg Arg Asn1 5 10 15Arg Lys
Lys Lys Asn Pro Gly Gly Gly Gly Ser Gly Asp His Pro Gln 20 25 30Ala
Thr Pro Ala Pro Ala Pro Asp Ala Ser Thr Glu Leu Pro Ala Ser 35 40
45Met Ser Gln Ala Gln His Leu Ala Ala Asn Thr Ala Thr Asp Asn Tyr
50 55 60Arg Ile Pro Ala Ile Thr Thr Ala Pro Asn Gly Asp Leu Leu Ile
Ser65 70 75 80Tyr Asp Glu Arg Pro Lys Asp Asn Gly Asn Gly Gly Ser
Asp Ala Pro 85 90 95Asn Pro Asn His Ile Val Gln Arg Arg Ser Thr Asp
Gly Gly Lys Thr 100 105 110Trp Ser Ala Pro Thr Tyr Ile His Gln Gly
Thr Glu Thr Gly Lys Lys 115 120 125Val Gly Tyr Ser Asp Pro Ser Tyr
Val Val Asp His Gln Thr Gly Thr 130 135 140Ile Phe Asn Phe His Val
Lys Ser Tyr Asp Gln Gly Trp Gly Gly Ser145 150 155 160Arg Gly Gly
Thr Asp Pro Glu Asn Arg Gly Ile Ile Gln Ala Glu Val 165 170 175Ser
Thr Ser Thr Asp Asn Gly Trp Thr Trp Thr His Arg Thr Ile Thr 180 185
190Ala Asp Ile Thr Lys Asp Lys Pro Trp Thr Ala Arg Phe Ala Ala Ser
195 200 205Gly Gln Gly Ile Gln Ile Gln His Gly Pro His Ala Gly Arg
Leu Val 210 215 220Gln Gln Tyr Thr Ile Arg Thr Ala Gly Gly Ala Val
Gln Ala Val Ser225 230 235 240Val Tyr Ser Asp Asp His Gly Lys Thr
Trp Gln Ala Gly Thr Pro Ile 245 250 255Gly Thr Gly Met Asp Glu Asn
Lys Val Val Glu Leu Ser Asp Gly Ser 260 265 270Leu Met Leu Asn Ser
Arg Ala Ser Asp Gly Ser Gly Phe Arg Lys Val 275 280 285Ala His Ser
Thr Asp Gly Gly Gln Thr Trp Ser Glu Pro Val Ser Asp 290 295 300Lys
Asn Leu Pro Asp Ser Val Asp Asn Ala Gln Ile Ile Arg Ala Phe305 310
315 320Pro Asn Ala Ala Pro Asp Asp Pro Arg Ala Lys Val Leu Leu Leu
Ser 325 330 335His Ser Pro Asn Pro Arg Pro Trp Ser Arg Asp Arg Gly
Thr Ile Ser 340 345 350Met Ser Cys Asp Asp Gly Ala Ser Trp Thr Thr
Ser Lys Val Phe His 355 360 365Glu Pro Phe Val Gly Tyr Thr Thr Ile
Ala Val Gln Ser Asp Gly Ser 370 375 380Ile Gly Leu Leu Ser Glu Asp
Ala His Asn Gly Ala Asp Tyr Gly Gly385 390 395 400Ile Trp Tyr Arg
Asn Phe Thr Met Asn Trp Leu Gly Glu Gln Cys Gly 405 410 415Gln Lys
Pro Ala Glu 420361275DNAArtificial SequenceSynthetic Construct
36ccatggttaa gcgcaaaaaa aaaggcggca aaaacggtaa aaatcgtcgt aaccgtaaga
60aaaaaaatcc tggtggtggt ggttctggag atcatccaca agctacacca gcacctgcac
120cagatgctag cactgagctg ccagcaagca tgtctcaggc tcagcatctt
gcagcaaata 180cggctactga taattatcgc attccagcga ttacaaccgc
tccgaatggt gatttactga 240ttagctatga tgaacggccg aaggacaatg
gaaatggtgg ttccgatgcc cctaacccga 300atcatattgt tcagcgtcgc
tccacagatg gcggtaaaac ttggagcgcg ccaacctata 360ttcatcaggg
tacggagact ggcaagaaag tgggatattc cgacccctct tatgtggtgg
420atcatcaaac cggtacaatc ttcaattttc atgtgaaatc atacgatcag
ggctggggag 480gtagccgtgg gggaacagac ccggaaaacc gcgggattat
tcaggcagag gtgtctacga 540gcacggataa tggatggacg tggacacatc
gcaccatcac cgcggatatt acgaaagata 600aaccgtggac cgcgcgtttt
gcggcgtccg gccaaggcat tcagatccag catgggccgc 660atgccggccg
tctggtgcaa cagtatacca ttcgtacggc cggtggagcg gtgcaggctg
720tatcggttta ttccgatgat catgggaaaa cgtggcaggc tggcaccccg
attgggacgg 780gtatggatga aaacaaagtt gtagagctgt ctgacggctc
tctgatgctg aacagtcgtg 840cgtcggacgg gagcggcttt cgtaaggttg
cgcatagcac tgatggtggg cagacctggt 900ccgaaccggt ttcggacaaa
aatttgccgg attcggttga taatgcccag ataattcgtg 960cgtttcctaa
tgctgccccc gatgacccgc gcgcgaaagt acttcttctg agtcattccc
1020caaatccacg tccgtggtcc cgggatcgtg gtacgataag catgtcatgt
gatgacgggg 1080cctcatggac cacttccaaa gtttttcacg aaccgtttgt
gggctacacg actattgcag 1140ttcagagtga tggaagcatc ggtctgctgt
cggaggacgc gcacaatggc gctgattatg 1200gtggcatctg gtatcgtaat
tttacgatga actggctggg agaacaatgt ggacaaaaac 1260ccgcggaata agctt
127537422PRTArtificial SequenceSynthetic Construct 37Met Val Lys
Arg Lys Lys Lys Gly Gly Lys Asn Gly Lys Asn Arg Arg1 5 10 15Asn Arg
Lys Lys Lys Asn Pro Gly Gly Gly Gly Ser Gly Asp His Pro 20 25 30Gln
Ala Thr Pro Ala Pro Ala Pro Asp Ala Ser Thr Glu Leu Pro Ala 35 40
45Ser Met Ser Gln Ala Gln His Leu Ala Ala Asn Thr Ala Thr Asp Asn
50 55 60Tyr Arg Ile Pro Ala Ile Thr Thr Ala Pro Asn Gly Asp Leu Leu
Ile65 70 75 80Ser Tyr Asp Glu Arg Pro Lys Asp Asn Gly Asn Gly Gly
Ser Asp Ala 85 90 95Pro Asn Pro Asn His Ile Val Gln Arg Arg Ser Thr
Asp Gly Gly Lys 100 105 110Thr Trp Ser Ala Pro Thr Tyr Ile His Gln
Gly Thr Glu Thr Gly Lys 115 120 125Lys Val Gly Tyr Ser Asp Pro Ser
Tyr Val Val Asp His Gln Thr Gly 130 135 140Thr Ile Phe Asn Phe His
Val Lys Ser Tyr Asp Gln Gly Trp Gly Gly145 150 155 160Ser Arg Gly
Gly Thr Asp Pro Glu Asn Arg Gly Ile Ile Gln Ala Glu 165 170 175Val
Ser Thr Ser Thr Asp Asn Gly Trp Thr Trp Thr His Arg Thr Ile 180 185
190Thr Ala Asp Ile Thr Lys Asp Lys Pro Trp Thr Ala Arg Phe Ala Ala
195 200 205Ser Gly Gln Gly Ile Gln Ile Gln His Gly Pro His Ala Gly
Arg Leu 210 215 220Val Gln Gln Tyr Thr Ile Arg Thr Ala Gly Gly Ala
Val Gln Ala Val225 230 235 240Ser Val Tyr Ser Asp Asp His Gly Lys
Thr Trp Gln Ala Gly Thr Pro 245 250 255Ile Gly Thr Gly Met Asp Glu
Asn Lys Val Val Glu Leu Ser Asp Gly 260 265 270Ser Leu Met Leu Asn
Ser Arg Ala Ser Asp Gly Ser Gly Phe Arg Lys 275 280 285Val Ala His
Ser Thr Asp Gly Gly Gln Thr Trp Ser Glu Pro Val Ser 290 295 300Asp
Lys Asn Leu Pro Asp Ser Val Asp Asn Ala Gln Ile Ile Arg Ala305 310
315 320Phe Pro Asn Ala Ala Pro Asp Asp Pro Arg Ala Lys Val Leu Leu
Leu 325 330 335Ser His Ser Pro Asn Pro Arg Pro Trp Ser Arg Asp Arg
Gly Thr Ile 340 345 350Ser Met Ser Cys Asp Asp Gly Ala Ser Trp Thr
Thr Ser Lys Val Phe 355 360 365His Glu Pro Phe Val Gly Tyr Thr Thr
Ile Ala Val Gln Ser Asp Gly 370 375 380Ser Ile Gly Leu Leu Ser Glu
Asp Ala His Asn Gly Ala Asp Tyr Gly385 390 395 400Gly Ile Trp Tyr
Arg Asn Phe Thr Met Asn Trp Leu Gly Glu Gln Cys 405 410 415Gly Gln
Lys Pro Ala Glu 42038416PRTArtificial SequenceSynthetic Construct
38Met Lys Arg Lys Lys Lys Gly Gly Lys Asn Gly Lys Asn Arg Arg Asn1
5 10 15Arg Lys Lys Lys Asn Pro Gly Asp His Pro Gln Ala Thr Pro Ala
Pro 20 25 30Ala Pro Asp Ala Ser Thr Glu Leu Pro Ala Ser Met Ser Gln
Ala Gln 35 40 45His Leu Ala Ala Asn Thr Ala Thr Asp Asn Tyr Arg Ile
Pro Ala Ile 50 55 60Thr Thr Ala Pro Asn Gly Asp Leu Leu Ile Ser Tyr
Asp Glu Arg Pro65 70 75 80Lys Asp Asn Gly Asn Gly Gly Ser Asp Ala
Pro
Asn Pro Asn His Ile 85 90 95Val Gln Arg Arg Ser Thr Asp Gly Gly Lys
Thr Trp Ser Ala Pro Thr 100 105 110Tyr Ile His Gln Gly Thr Glu Thr
Gly Lys Lys Val Gly Tyr Ser Asp 115 120 125Pro Ser Tyr Val Val Asp
His Gln Thr Gly Thr Ile Phe Asn Phe His 130 135 140Val Lys Ser Tyr
Asp Gln Gly Trp Gly Gly Ser Arg Gly Gly Thr Asp145 150 155 160Pro
Glu Asn Arg Gly Ile Ile Gln Ala Glu Val Ser Thr Ser Thr Asp 165 170
175Asn Gly Trp Thr Trp Thr His Arg Thr Ile Thr Ala Asp Ile Thr Lys
180 185 190Asp Lys Pro Trp Thr Ala Arg Phe Ala Ala Ser Gly Gln Gly
Ile Gln 195 200 205Ile Gln His Gly Pro His Ala Gly Arg Leu Val Gln
Gln Tyr Thr Ile 210 215 220Arg Thr Ala Gly Gly Ala Val Gln Ala Val
Ser Val Tyr Ser Asp Asp225 230 235 240His Gly Lys Thr Trp Gln Ala
Gly Thr Pro Ile Gly Thr Gly Met Asp 245 250 255Glu Asn Lys Val Val
Glu Leu Ser Asp Gly Ser Leu Met Leu Asn Ser 260 265 270Arg Ala Ser
Asp Gly Ser Gly Phe Arg Lys Val Ala His Ser Thr Asp 275 280 285Gly
Gly Gln Thr Trp Ser Glu Pro Val Ser Asp Lys Asn Leu Pro Asp 290 295
300Ser Val Asp Asn Ala Gln Ile Ile Arg Ala Phe Pro Asn Ala Ala
Pro305 310 315 320Asp Asp Pro Arg Ala Lys Val Leu Leu Leu Ser His
Ser Pro Asn Pro 325 330 335Arg Pro Trp Ser Arg Asp Arg Gly Thr Ile
Ser Met Ser Cys Asp Asp 340 345 350Gly Ala Ser Trp Thr Thr Ser Lys
Val Phe His Glu Pro Phe Val Gly 355 360 365Tyr Thr Thr Ile Ala Val
Gln Ser Asp Gly Ser Ile Gly Leu Leu Ser 370 375 380Glu Asp Ala His
Asn Gly Ala Asp Tyr Gly Gly Ile Trp Tyr Arg Asn385 390 395 400Phe
Thr Met Asn Trp Leu Gly Glu Gln Cys Gly Gln Lys Pro Ala Glu 405 410
41539421PRTArtificial SequenceSynthetic Construct 39Val Lys Arg Lys
Lys Lys Gly Gly Lys Asn Gly Lys Asn Arg Arg Asn1 5 10 15Arg Lys Lys
Lys Asn Pro Gly Gly Gly Gly Ser Gly Asp His Pro Gln 20 25 30Ala Thr
Pro Ala Pro Ala Pro Asp Ala Ser Thr Glu Leu Pro Ala Ser 35 40 45Met
Ser Gln Ala Gln His Leu Ala Ala Asn Thr Ala Thr Asp Asn Tyr 50 55
60Arg Ile Pro Ala Ile Thr Thr Ala Pro Asn Gly Asp Leu Leu Ile Ser65
70 75 80Tyr Asp Glu Arg Pro Lys Asp Asn Gly Asn Gly Gly Ser Asp Ala
Pro 85 90 95Asn Pro Asn His Ile Val Gln Arg Arg Ser Thr Asp Gly Gly
Lys Thr 100 105 110Trp Ser Ala Pro Thr Tyr Ile His Gln Gly Thr Glu
Thr Gly Lys Lys 115 120 125Val Gly Tyr Ser Asp Pro Ser Tyr Val Val
Asp His Gln Thr Gly Thr 130 135 140Ile Phe Asn Phe His Val Lys Ser
Tyr Asp Gln Gly Trp Gly Gly Ser145 150 155 160Arg Gly Gly Thr Asp
Pro Glu Asn Arg Gly Ile Ile Gln Ala Glu Val 165 170 175Ser Thr Ser
Thr Asp Asn Gly Trp Thr Trp Thr His Arg Thr Ile Thr 180 185 190Ala
Asp Ile Thr Lys Asp Lys Pro Trp Thr Ala Arg Phe Ala Ala Ser 195 200
205Gly Gln Gly Ile Gln Ile Gln His Gly Pro His Ala Gly Arg Leu Val
210 215 220Gln Gln Tyr Thr Ile Arg Thr Ala Gly Gly Ala Val Gln Ala
Val Ser225 230 235 240Val Tyr Ser Asp Asp His Gly Lys Thr Trp Gln
Ala Gly Thr Pro Ile 245 250 255Gly Thr Gly Met Asp Glu Asn Lys Val
Val Glu Leu Ser Asp Gly Ser 260 265 270Leu Met Leu Asn Ser Arg Ala
Ser Asp Gly Ser Gly Phe Arg Lys Val 275 280 285Ala His Ser Thr Asp
Gly Gly Gln Thr Trp Ser Glu Pro Val Ser Asp 290 295 300Lys Asn Leu
Pro Asp Ser Val Asp Asn Ala Gln Ile Ile Arg Ala Phe305 310 315
320Pro Asn Ala Ala Pro Asp Asp Pro Arg Ala Lys Val Leu Leu Leu Ser
325 330 335His Ser Pro Asn Pro Arg Pro Trp Ser Arg Asp Arg Gly Thr
Ile Ser 340 345 350Met Ser Cys Asp Asp Gly Ala Ser Trp Thr Thr Ser
Lys Val Phe His 355 360 365Glu Pro Phe Val Gly Tyr Thr Thr Ile Ala
Val Gln Ser Asp Gly Ser 370 375 380Ile Gly Leu Leu Ser Glu Asp Ala
His Asn Gly Ala Asp Tyr Gly Gly385 390 395 400Ile Trp Tyr Arg Asn
Phe Thr Met Asn Trp Leu Gly Glu Gln Cys Gly 405 410 415Gln Lys Pro
Ala Glu 420
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023
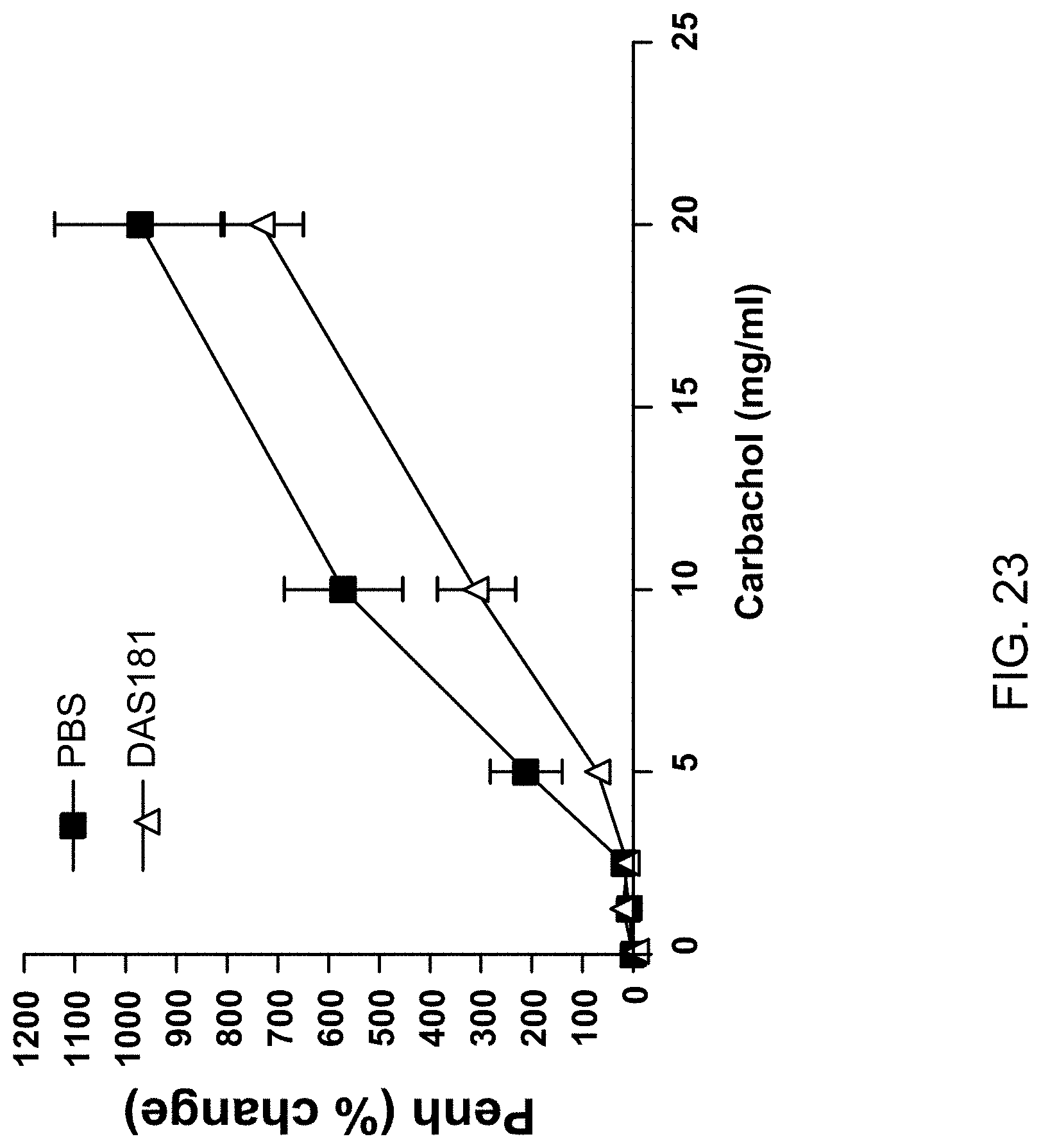
D00024

D00025

D00026
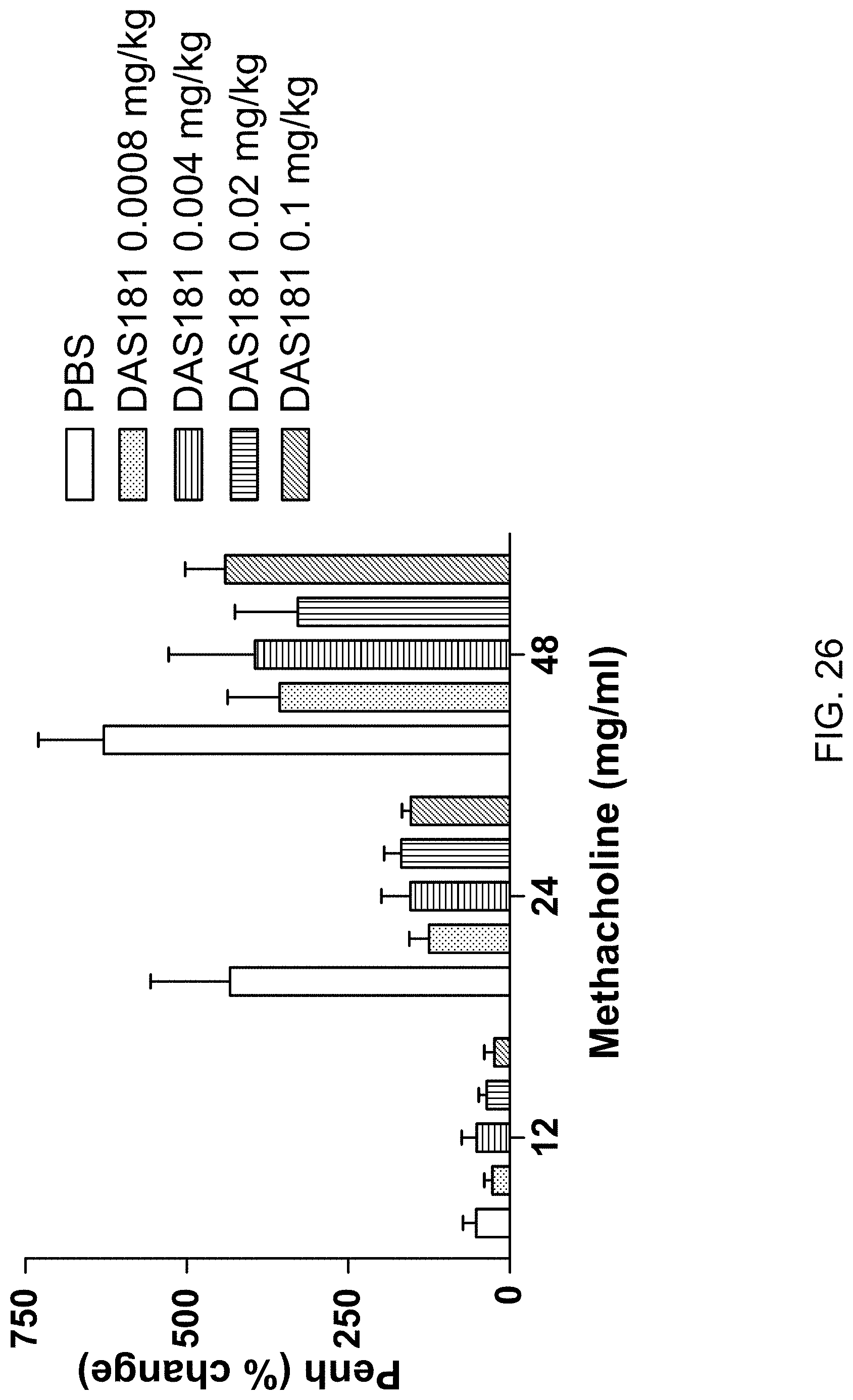
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.