Methods For Making Reference Electrode And Lithium Ion Battery Having The Same
CHU; ZHENG-YU ; et al.
U.S. patent application number 16/875932 was filed with the patent office on 2020-09-03 for methods for making reference electrode and lithium ion battery having the same. This patent application is currently assigned to Tsinghua University. The applicant listed for this patent is Tsinghua University. Invention is credited to ZHENG-YU CHU, XU-NING FENG, JIAN-QIU LI, LAN-GUANG LU, MING-GAO OUYANG.
| Application Number | 20200280051 16/875932 |
| Document ID | / |
| Family ID | 1000004842080 |
| Filed Date | 2020-09-03 |









View All Diagrams
| United States Patent Application | 20200280051 |
| Kind Code | A1 |
| CHU; ZHENG-YU ; et al. | September 3, 2020 |
METHODS FOR MAKING REFERENCE ELECTRODE AND LITHIUM ION BATTERY HAVING THE SAME
Abstract
The present disclosure relates to methods for making a reference electrode and a lithium ion battery having the reference electrode. The reference electrode obtained has a long service life, moreover, the manufacturing process is simple and can meet the industrial production requirements, making the production and the application of the lithium-ion battery with the reference electrode possible.
| Inventors: | CHU; ZHENG-YU; (Beijing, CN) ; FENG; XU-NING; (Beijing, CN) ; LU; LAN-GUANG; (Beijing, CN) ; LI; JIAN-QIU; (Beijing, CN) ; OUYANG; MING-GAO; (Beijing, CN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Tsinghua University Beijing CN |
||||||||||
| Family ID: | 1000004842080 | ||||||||||
| Appl. No.: | 16/875932 | ||||||||||
| Filed: | May 15, 2020 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| PCT/CN2018/114516 | Nov 8, 2018 | |||
| 16875932 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | H01M 2/166 20130101; H01M 4/661 20130101; H01M 2004/021 20130101; H01M 4/382 20130101; H01M 10/0525 20130101; H01M 4/1395 20130101 |
| International Class: | H01M 4/1395 20060101 H01M004/1395; H01M 4/66 20060101 H01M004/66; H01M 4/38 20060101 H01M004/38; H01M 2/16 20060101 H01M002/16; H01M 10/0525 20060101 H01M010/0525 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Nov 17, 2017 | CN | 201711143955.2 |
Claims
1. A method for making a reference electrode, comprising: providing a reference electrode substrate and a current collecting metal sheet adhered with a tab binder; welding the reference electrode substrate to a lower portion of the current collecting metal sheet; melting lithium metal to liquid state, thereby obtaining a liquid lithium; immersing a lower portion of the reference electrode substrate welded with the current collecting metal sheet into the liquid lithium and standing to coat a lithium metal layer on the lower portion of the reference electrode substrate, thereby obtaining a reference electrode welded with the current collecting metal sheet; and taking out the reference electrode welded with the current collecting metal sheet from the liquid lithium, cooling, and warping a portion below the tab binder of the reference electrode with a separator, thereby obtaining a reference electrode wrapped with the separator.
2. The method of claim 1, wherein the reference electrode substrate is a porous copper foam, a porous nickel foam, a copper mesh, or a nickel mesh.
3. The method of claim 1, wherein before the welding, the reference electrode substrate is washed and then dried.
4. The method of claim 1, wherein after the welding, an upper portion of the reference electrode substrate is overlapped with the lower portion of the current collecting metal sheet, and an area of the current collecting metal sheet is smaller than an area of the reference electrode substrate.
5. The method of claim 1, after the welding, further comprising: vacuum drying the reference electrode substrate welded with the current collecting metal sheet at about 60.degree. C. to about 90.degree. C. under the water-free and oxygen-free condition for about 4 to about 7 hours, cooling, and then transferring into and preserving in a water-free and oxygen-free environment.
6. The method of claim 1, wherein before the immersing, the liquid lithium is heated to about 200.degree. C. to about 500.degree. C. to remove an impurity on the surface of the liquid lithium under the water-free and oxygen-free condition.
7. The method of claim 1, wherein the lower portion of the reference electrode substrate is immersed into the liquid lithium and rested for about 1 min to about 5 min to coat the lithium metal layer on the lower portion of the reference electrode substrate.
8. The method of claim 1, wherein after the wrapping, the separator is wrapped tightly around the current collecting metal sheet and the reference electrode substrate.
9. The method of claim 1, wherein a material of the current collecting metal sheet is nickel or aluminum.
10. The method of claim 1, wherein a material of the separator is porous polypropylene, porous polyethylene, porous polypropylene coated with ceramic, porous polyethylene coated with ceramic, or non-woven fabric.
11. The method of claim 1, wherein the reference electrode substrate has a pore size of about 50 .mu.m to about 500 .mu.m and a thickness of about 0.1 mm to about 1 mm.
12. The method of claim 1, wherein a thickness of the current collecting metal sheet is about 0.1 mm to about 1 mm, and a length of the current collecting metal sheet is about 10 mm to about 30 mm.
13. The method of claim 1, wherein the lithium metal layer has a thickness of about 10 .mu.m to about 100 .mu.m.
14. A method for making a lithium ion battery having a reference electrode, comprising: providing a reference electrode substrate and a current collecting metal sheet adhered with a tab binder; welding the reference electrode substrate to a lower portion of the current collecting metal sheet; melting lithium metal to liquid state, thereby obtaining a liquid lithium; immersing a lower portion of the reference electrode substrate welded with the current collecting metal sheet into the liquid lithium and standing to coat a lithium metal layer on the lower portion of the reference electrode substrate, thereby obtaining a reference electrode welded with the current collecting metal sheet; taking out the reference electrode welded with the current collecting metal sheet from the liquid lithium, cooling, and warping a portion below the tab binder of the reference electrode with a separator, thereby obtaining a reference electrode wrapped with the separator; interposing the reference electrode wrapped with the separator between a cell separator and an anode plate of a cell of the lithium ion battery and protrude an upper end of the reference electrode out from the cell of the lithium ion battery under the water-free and oxygen-free condition; and packaging the lithium ion battery implanted with the reference electrode under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery having the reference electrode.
15. The method of claim 14, wherein an area of the reference electrode substrate is about 1% to about 10% of an area of an electrode plate of the lithium ion battery.
16. The method of claim 14, wherein the upper end of the reference electrode is protruded out from the cell of the lithium ion battery for 1 mm to 2 mm.
17. The method of claim 14, wherein the lithium ion battery implanted with the reference electrode is packaged by an aluminum plastic film under the anhydrous and oxygen-free condition.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims all benefits accruing under 35 U.S.C. .sctn. 119 from China Patent Application No. 201711143955.2, filed on Nov. 17, 2017 in the State Intellectual Property Office of China, the content of which is hereby incorporated by reference. This application is a continuation under 35 U.S.C. .sctn. 120 of international patent application PCT/CN2018/114516 filed on Nov. 8, 2018, the content of which is also hereby incorporated by reference.
FIELD
[0002] The present disclosure relates to methods for making reference electrode and lithium ion battery having the same, and belongs to the technical fields of measurement of an electrode potential and manufacture of an electrode.
BACKGROUND
[0003] The electrode potential is one of the most important parameters in electrochemistry and battery research. The open-circuit potential is an electrode potential in a thermodynamic equilibrium state. In practice, a voltage vs. SOC curve of a battery obtained during a charge-discharge process at a low current can be regarded as an open-circuit voltage curve. If there is a current flowing through an electrode, the electrode would be polarized and a potential of the electrode would be deviated due to an occurrence of overpotential. The detailed information of a reaction occurring inside the electrode can be obtained by measuring a potential varying curve of the electrode. Taking a lithium ion battery with a graphite anode as an example, overcharging or low-temperature charging may result in a Li plating at the graphite anode, characterized in that the anode potential is below to the equilibrium potential for the lithium-plating. Therefore, the Li plating side reaction can be detected by measuring the electrode potential. However, the battery includes two electrodes, and the polarization of the individual electrode cannot be detected directly because the overpotential gauged is composed of the polarized potential of positive and negative electrodes, respectively. More specifically, one or more reference electrodes are interposed between the cathode and the anode to measure the voltage difference between the electrode and the reference electrode. The reference electrode in the related art is mainly made of electrochemical deposition, lithium foil, lithium alloy, lithium-containing metal oxide, or lithium-containing phosphate, and so on.
[0004] The research article Development of reliable lithium micro-reference electrodes for long-term in-situ studies of lithium-based battery systems, Journal of The Electrochemical Society, 2004 (DOI: 10.6100/IR624713) discloses lithium reference electrodes made by electrochemical deposition on two surfaces of a micron sized copper wire positioned in the battery. The method can minimize the blocking to the lithium ion flow. However, since the reference electrode is too small, the amount of lithium loaded on the reference electrode is small, and the deposition layer is usually uneven. Consequently, a potential drift may occur after a long time measurement, which makes it difficult to apply in durability studies, and requires high input impedance of the measuring instrument.
[0005] Another method involves inserting a lithium foil directly into a battery. The lithium metal is connected to a current collector by a physical pressing method. In the research article Self-Discharge of LiMn.sub.2O.sub.4/C Li-Ion Cells in Their Discharged State, J. Electrochem. Soc., Vol. 145, No. 1, 1998, lithium is physically connected to, i.e., pressed against, a current collector (for example, a copper mesh). In this method, the copper mesh has a relatively large pore diameter which is at the millimeter-scale. However, the connection reliability of the lithium and the copper mesh cannot be ensured by this physical connection method. A poor contact can lead to an extremely large ohmic resistance of the reference electrode influencing the use thereof.
[0006] In view of the above, the difficulties in the development of the reference electrode can be contributed to the lithium amount in the electrode. The size of the reference electrode should be as small as possible to decrease the blocking effect on lithium ions in the electrolyte. However, this will reduce the lithium amount in the material, and thus weaken the signal. In addition, an electrode material loss or a potential drift tends to occur because of the micro-currents in the measurement.
SUMMARY
[0007] An object of the present disclosure is to provide a method for making a reference electrode and a method for making a lithium ion battery having the reference electrode.
[0008] The method for making the reference electrode in the present disclosure includes: [0009] S11, washing and then drying a reference electrode substrate; [0010] S12, welding the reference electrode substrate to a lower portion of a current collecting metal sheet, an upper portion of which is adhered with a tab binder; [0011] S13, melting lithium metal to liquid state and continuing heating to remove an impurity on a surface of the liquid state lithium metal under a water-free and oxygen-free condition; [0012] S14, immersing a lower portion of the reference electrode substrate welded with the current collecting metal sheet into the liquid lithium and standing to coat a lithium metal layer on the lower portion of the reference electrode substrate; [0013] S15, taking out a reference electrode welded with the current collecting metal sheet from the liquid lithium, cooling, and warping a portion below the tab binder of the reference electrode with a separator by a winding method, thereby obtaining the reference electrode wrapped with the separator.
[0014] The method for making the lithium ion battery having the reference electrode in the present disclosure includes: [0015] S10, making a reference electrode, which comprises: [0016] S11, washing and then drying a reference electrode substrate; [0017] S12, welding the reference electrode substrate to a lower portion of a current collecting metal sheet, an upper portion of which is adhered with a tab binder; [0018] S13, melting lithium metal to liquid state and continuing heating to remove an impurity on a surface of the liquid state lithium metal under a water-free and oxygen-free condition; [0019] S14, immersing a lower portion of the reference electrode substrate welded with the current collecting metal sheet into the liquid lithium and standing to coat a lithium metal layer on the lower portion of the reference electrode substrate; [0020] S15, taking out a reference electrode welded with the current collecting metal sheet from the liquid lithium, cooling, and warping a portion below the tab binder of the reference electrode with a separator by a winding method, thereby obtaining the reference electrode wrapped with the separator; [0021] S20, interposing the reference electrode between a separator and an anode plate of a cell of the lithium ion battery and protrude an upper end of the reference electrode out from the cell of the lithium ion battery under the water-free and oxygen-free condition; and [0022] S30, packaging the lithium ion battery implanted with the reference electrode under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery having the reference electrode.
[0023] Another method for making the lithium ion battery having the reference electrode in the present disclosure includes: [0024] S100, making a reference electrode, which comprises: [0025] S110, providing a porous copper foam, a porous nickel foam, a copper mesh, or a mesh as a reference electrode substrate; and washing the reference electrode substrate with acetone or deionized water and then drying for later use, wherein a material of the reference electrode substrate has a pore size of about 50 .mu.m to about 500 .mu.m, a thickness of the reference electrode substrate is about 0.1 mm to about 1 mm, and an area of the reference electrode substrate is about 1% to about 10% of an area of an electrode plate of the lithium ion battery; [0026] S120, welding the reference electrode substrate obtained from the step S110 to a lower portion of a current collecting metal sheet of which an upper portion is adhered with a tab binder, to allow an upper portion of the reference electrode substrate to be overlapped with the lower portion of the current collecting metal sheet, wherein an area of the current collecting metal sheet is smaller than the area of the reference electrode substrate; and vacuum drying at about 60.degree. C. to about 90.degree. C. under the water-free and oxygen-free condition for about 4 to about 7 hours, cooing, and then transferring into and preserving in a water-free and oxygen-free environment, wherein the current collecting metal sheet is configured for collecting an electric current, a material of the current collecting metal sheet is nickel or aluminum, a thickness of the current collecting metal sheet is about 0.1 mm to about 1 mm, and a length of the current collecting metal sheet is about 10 mm to about 30 mm; and [0027] S130, melting lithium metal to liquid state and continuing heating to about 200.degree. C. to about 500.degree. C. to remove an impurity on a surface of the liquid state lithium metal under the water-free and oxygen-free condition; immersing the lower portion of the reference electrode substrate whose upper portion is welded with the current collecting metal sheet obtained from the step S120 into the liquid lithium and standing for about 1 min to about 5 min to allow a lithium metal layer having a thickness of about 10 .mu.m to about 100 .mu.m to coat on the lower portion of the reference electrode substrate; and taking out, cooling, and wrapping a portion below the tab binder with a separator by a winding method to allow the separator to be wrapped tightly and entirely around the current collecting metal sheet and the reference electrode substrate, thereby obtaining the reference electrode wrapped with the separator, wherein a material of the separator is porous polypropylene, porous polyethylene, porous polypropylene coated with ceramic, porous polyethylene coated with ceramic, or non-woven fabric; [0028] S200, interposing the reference electrode obtained from the step S100 between a separator and an anode plate of a cell of the lithium ion battery under the water-free and oxygen-free condition, and protruding an upper end of the reference electrode out from the cell of the lithium ion battery for about 1 mm to about 2 mm; [0029] S300, packaging the lithium ion battery implanted with the reference electrode with an aluminum plastic film under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery having the reference electrode.
[0030] In the method for making the reference electrode and the method for making the lithium ion battery having the reference electrode according to the present disclosure, the lithium metal can be formed on the surface of the substrate material, while the porous property of the substrate material can be maintained, so that small molecules in the liquid electrolyte can pass through the pores, and the normal operation of the battery will not be influenced. Moreover, the thickness of the lithium metal layer formed on the substrate can be controlled by regulating parameters in the making process to ensure that the porous property of the substrate material can be retained while sufficient lithium can be loaded on the substrate to meet measurement requirements. Therefore, the reference electrode made by the method according to the present disclosure can have sufficient micro-structures as well as long service life. In addition, the manufacturing process is simple and can meet the industrial production requirements, making the industrial production and the application of the lithium-ion battery with the reference electrode possible.
[0031] The methods provided in the present disclosure solve the unstable problem of the measurement using the traditional reference electrode and improve the service life of the reference electrode, thereby achieving a long term multi-cycle potential measurement of the individual electrode and increasing the accuracy of the measuring result.
BRIEF DESCRIPTION OF THE DRAWINGS
[0032] To describe the technical solutions of the embodiments of the present disclosure or the related technology more clearly, the following briefly introduces the accompanying drawings for describing the embodiments or the related technology. Obviously, the accompanying drawings in the following description are only embodiments of the present disclosure, and a person of ordinary skill in the art may still derive other drawings from the accompanying drawings of the present disclosure without creative effort.
[0033] FIG. 1 is a flowchart of a method for making a reference electrode according to an embodiment of the present disclosure.
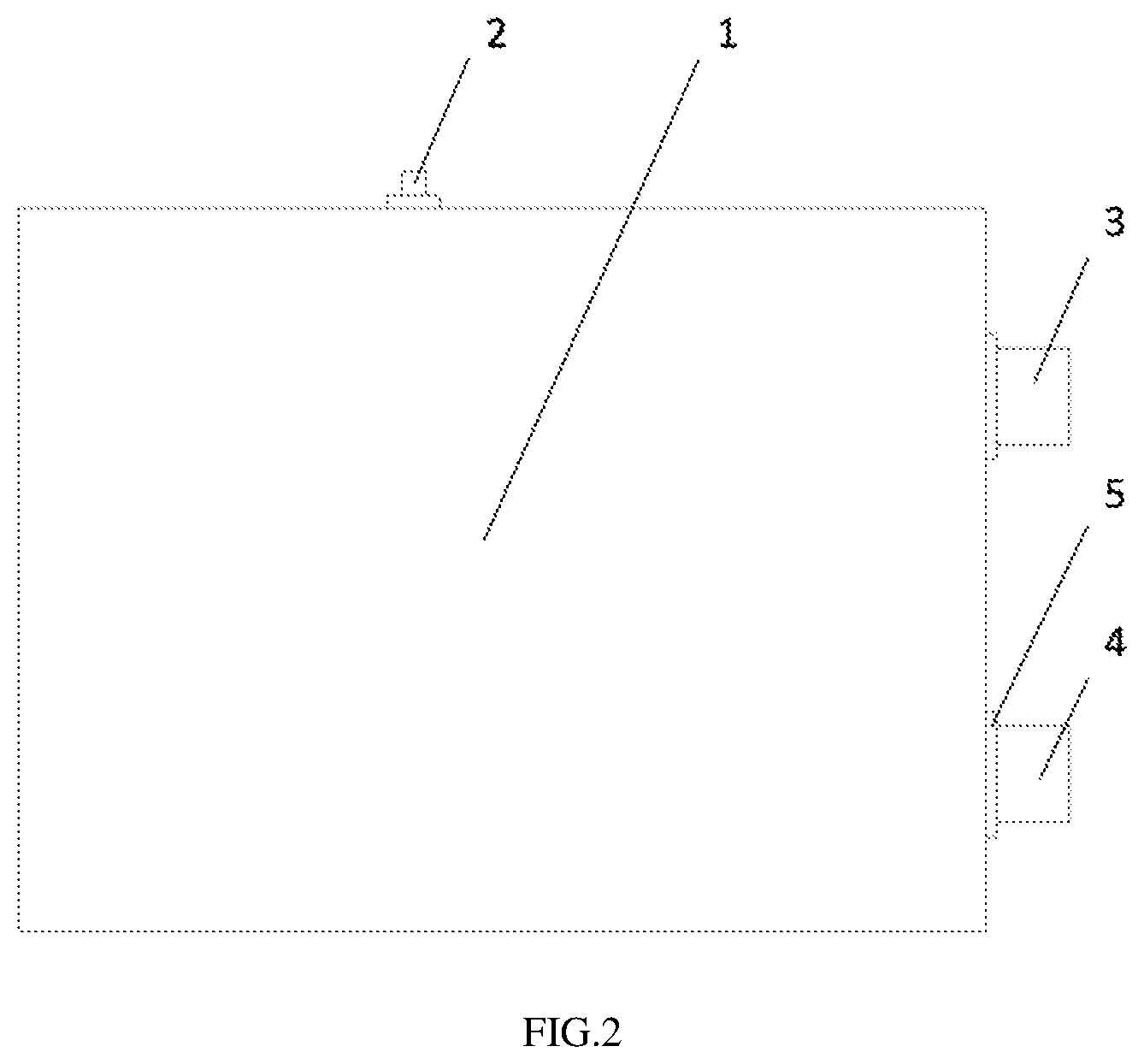
[0034] FIG. 2 is a schematic structural view of a lithium ion battery having the reference electrode according to an embodiment of the present disclosure.
[0035] FIG. 3 is a front view of the reference electrode according to an embodiment of the present disclosure.
[0036] FIG. 4 is a flowchart of another method for making the reference electrode according to an embodiment of the present disclosure.
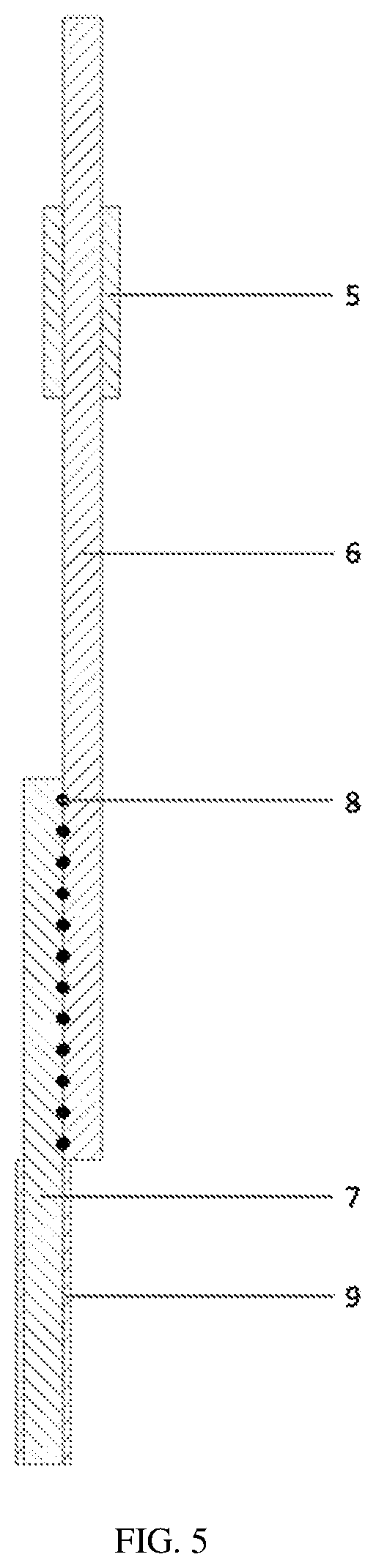
[0037] FIG. 5 is a side view of the reference electrode shown in the FIG. 3 according to an embodiment of the present disclosure.
[0038] FIG. 6 is a flowchart of a method for making a lithium ion battery having the reference electrode according to an embodiment of the present disclosure.
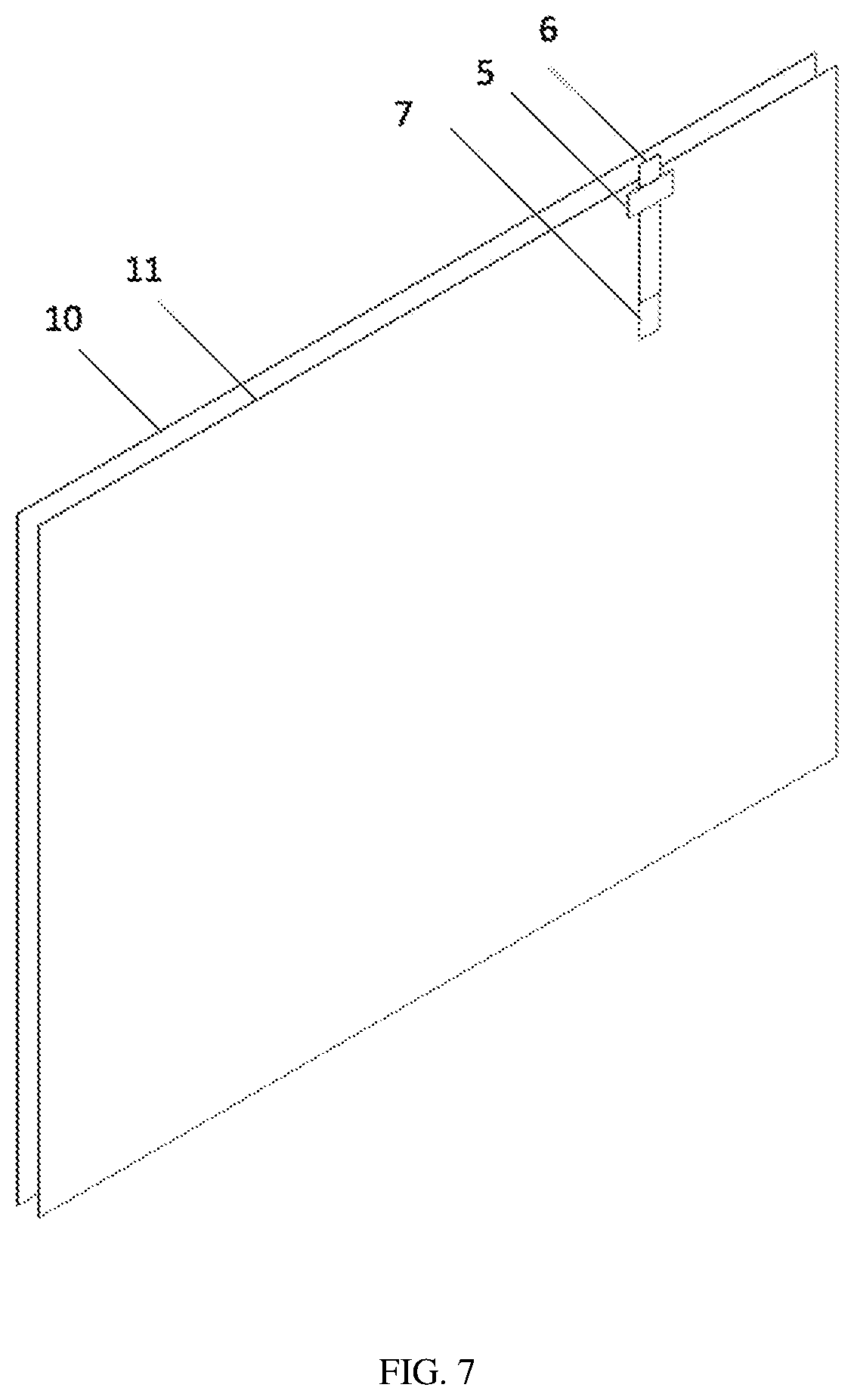
[0039] FIG. 7 is a schematic view of the lithium ion battery implanted with the reference electrode according to an embodiment of the present disclosure.
[0040] FIG. 8 is a flowchart of another method for making the lithium ion battery having the reference electrode according to an embodiment of the present disclosure.
[0041] FIG. 9 is a flowchart of yet another method for making the lithium ion battery having the reference electrode according to an embodiment of the present disclosure.
[0042] FIG. 10 is a schematic diagram showing a measuring circuit connecting the lithium ion battery having the reference electrode according to an embodiment of the present disclosure.
[0043] FIG. 11 is a diagram showing results of rate performance test for the lithium ion battery having the reference electrode according to an embodiment of the present disclosure.
[0044] FIG. 12 is a diagram showing results of capacity test for the lithium ion battery having the reference electrode according to an embodiment of the present disclosure.
DESCRIPTION OF REFERENCE NUMERAL
[0045] lithium ion battery 1; [0046] reference electrode 2; [0047] cathode 3; [0048] anode 4, [0049] tab binder 5, [0050] current collecting metal sheet 6, [0051] reference electrode substrate 7, [0052] welding spot 8, [0053] lithium metal layer 9, [0054] cathode plate 10, [0055] separator 11 between the cathode plate and the anode plate of the lithium ion battery
DETAILED DESCRIPTION
[0056] Referring to FIGS. 1 to 3, a method for making a reference electrode is provided in the present disclosure. The method for making the reference electrode includes:
[0057] S11, washing and then drying a reference electrode substrate;
[0058] S12, welding the reference electrode substrate 7 to a lower portion of a current collecting metal sheet 6, an upper portion of which is adhered with a tab binder 5;
[0059] S13, melting lithium metal to liquid state and continuing heating to remove an impurity on a surface of the liquid state lithium metal (i.e. liquid lithium) under a water-free and oxygen-free condition;
[0060] S14, immersing a lower portion of the reference electrode substrate 7 welded with the current collecting metal sheet 6 into the liquid lithium and standing to coat a lithium metal layer on the lower portion of the reference electrode substrate 7, thereby obtaining a reference electrode 2 welded with the current collecting metal sheet 6;
[0061] S15, taking out the reference electrode 2 welded with the current collecting metal sheet 6 from the liquid lithium, cooling, and warping a portion below the tab binder 5 of the reference electrode 2 with a separator by a winding method, thereby obtaining a reference electrode 2 wrapped with the separator.
[0062] Referring to FIG. 4, in an embodiment, prior to the step S11, S011, a porous copper foam, a porous nickel foam, a copper mesh, or a nickel mesh is adopted as the reference electrode substrate 7. In the step S11, the reference electrode substrate 7 is washed with acetone or deionized water and then dried for later use. In this embodiment, the material of the reference electrode substrate 7 can have a pore size of about 50 .mu.m to about 500 .mu.m, and a thickness of the reference electrode substrate 7 can be about 0.1 mm to about 1 mm.
[0063] In an embodiment, in the step S12, the reference electrode substrate 7 is welded to the lower portion of the current collecting metal sheet 6 whose upper portion is adhered with the tab binder 5, to allow an upper portion of the reference electrode substrate 7 to be overlapped with the lower portion of the current collecting metal sheet 6. An area of the current collecting metal sheet 6 is smaller than an area of the reference electrode substrate 7. After the step S12, S012, the reference electrode substrate 7 welded with the current collecting metal sheet 6 is vacuum dried at about 60.degree. C. to about 90.degree. C. under the water-free and oxygen-free condition for about 4 to about 7 hours, cooled, and then transferred into and preserved in a water-free and oxygen-free environment. The current collecting metal sheet 6 is used for collecting an electric current. A material of the current collecting metal sheet 6 is nickel or aluminum. In this embodiment, a thickness of the current collecting metal sheet 6 can be about 0.1 mm to about 1 mm. A length of the current collecting metal sheet 6 can depend on a location of the reference electrode 2 disposed with respect to the battery, and can be about 10 mm to about 30 mm.
[0064] In an embodiment, in the step S13, the lithium metal is melted into the liquid state and is continuously heated to about 200.degree. C. to about 500.degree. C. to remove the impurity on the surface of the liquid state lithium metal under the water-free and oxygen-free condition.
[0065] Referring to FIG. 5, in an embodiment, in the step S14, the lower portion of the reference electrode substrate 7 whose upper portion is welded with the current collecting metal sheet 6 is immersed into the liquid lithium and rested for about 1 minute (min) to about 5 min to coat the lithium metal layer 9 on the lower portion of the reference electrode substrate 7. In this embodiment, the portion without any welding spot 8 of the substrate can be entirely immersed into the liquid lithium, and the lower portion of the reference electrode substrate 7 can be fully wetted with the lithium metal to allow the lithium metal layer 9 to be formed on the lower portion of the reference electrode substrate 7. The lithium metal layer 9 can have a thickness of about 10 .mu.m to about 100 .mu.m.
[0066] In an embodiment, in the step S15, the reference electrode 2 welded with the current collecting metal sheet 6 is taken out from the liquid lithium and cooled. The portion below the tab binder 5 of the reference electrode 2 is wrapped with the separator by the winding method. The separator can be wrapped tightly and entirely around the current collecting metal sheet 6 and the reference electrode substrate 7, thereby obtaining the reference electrode 2 wrapped with the separator. A material of the separator is porous polypropylene, porous polyethylene, porous polypropylene coated with ceramic, porous polyethylene coated with ceramic, or non-woven fabric.
[0067] Referring to FIGS. 6 and 7, a method for making a lithium ion battery having a reference electrode is provided in the present disclosure. The method for making the lithium ion battery having the reference electrode includes making two parts: a reference electrode and a lithium ion battery. The method includes:
[0068] S10, making a reference electrode 2, which includes:
[0069] S11, washing and then drying a reference electrode substrate 7;
[0070] S12, welding the reference electrode substrate 7 to a lower portion of a current collecting metal sheet 6, an upper portion of which is adhered with a tab binder 5;
[0071] S13, melting a lithium metal to liquid state and continuing heating to remove an impurity on a surface of the liquid state lithium metal (i.e. liquid lithium) under a water-free and oxygen-free condition;
[0072] S14, immersing a lower portion of the reference electrode substrate 7 welded with the current collecting metal sheet 6 into the liquid lithium and standing to coat a lithium metal layer on the lower portion of the reference electrode substrate 7, thereby obtaining a reference electrode 2 welded with the current collecting metal sheet 6; and
[0073] S15, taking out the reference electrode 2 welded with the current collecting metal sheet 6 from the liquid lithium, cooling, and warping a portion below the tab binder 5 of the reference electrode 2 with a separator by a winding method, thereby obtaining the reference electrode 2 wrapped with the separator;
[0074] The method further includes making the lithium ion battery 1 having the reference electrode 2, which includes:
[0075] S20, interposing the reference electrode 2 between a separator 11 and an anode plate of a cell of the lithium ion battery 1 and protruding an upper end of the reference electrode 2 out from the cell of the lithium ion battery 1 under the water-free and oxygen-free condition; and
[0076] S30, packaging the lithium ion battery 1 implanted with the reference electrode 2 under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery 1 having the reference electrode 2.
[0077] Referring to FIG. 8, in an embodiment, prior to the step S11, S011, a porous copper foam, a porous nickel foam, a copper mesh, or a nickel mesh is adopted as the reference electrode substrate 7. An area of the reference electrode substrate 7 is about 1% to about 10% of an area of an electrode plate of the lithium ion battery 1. In the step S11, the reference electrode substrate 7 is washed with acetone or deionized water and then dried for later use. In this embodiment, the material of the reference electrode substrate 7 can have a pore size of about 50 .mu.m to about 500 .mu.m, and a thickness of the reference electrode substrate 7 can be about 0.1 mm to about 1 mm.
[0078] In an embodiment, in the step S12, the reference electrode substrate 7 is welded to the lower portion of the current collecting metal sheet 6 whose upper portion is adhered with the tab binder 5, to allow an upper portion of the reference electrode substrate 7 to be overlapped with the lower portion of the current collecting metal sheet 6. An area of the current collecting metal sheet 6 is smaller than an area of the reference electrode substrate 7. After the step S12, S012, the reference electrode substrate 7 welded with the current collecting metal sheet 6 is vacuum dried at about 60.degree. C. to about 90.degree. C. under the water-free and oxygen-free condition for about 4 to about 7 hours, cooled, and then transferred into and preserved in a water-free and oxygen-free environment. The current collecting metal sheet 6 is used for collecting an electric current. A material of the current collecting metal sheet 6 is nickel or aluminum. In this embodiment, a thickness of the current collecting metal sheet 6 can be about 0.1 mm to about 1 mm. A length of the current collecting metal sheet 6 can depend on a location of the reference electrode 2 disposed with respect to the battery, and can be about 10 mm to about 30 mm.
[0079] In an embodiment, in the step S13, the lithium metal is melted into the liquid state and is continuously heated to about 200.degree. C. to about 500.degree. C. to remove the impurity on the surface of the liquid state lithium metal under the water-free and oxygen-free condition.
[0080] In an embodiment, in the step S14, the lower portion of the reference electrode substrate 7 whose upper portion is welded with the current collecting metal sheet 6 is immersed into the liquid lithium and rested for 1 min to 5 min to coat the lithium metal layer 9 on the lower portion of the reference electrode substrate 7. In this embodiment, the portion without any welding spot 8 of the substrate can be entirely immersed into the liquid lithium, and the lower portion of the reference electrode substrate 7 can be fully wetted with the lithium metal to allow the lithium metal layer 9 to be formed on the lower portion of the reference electrode substrate 7. The lithium metal layer 9 can have a thickness of about 10 .mu.m to about 100 .mu.m.
[0081] In an embodiment, in the step S15, the reference electrode 2 welded with the current collecting metal sheet 6 is taken out from the liquid lithium and cooled. The portion below the tab binder 5 of the reference electrode 2 is wrapped with the separator by the winding method. The separator can be wrapped tightly and entirely around the current collecting metal sheet 6 and the reference electrode substrate 7, thereby obtaining the reference electrode 2 wrapped with the separator. A material of the separator is porous polypropylene, porous polyethylene, porous polypropylene coated with ceramic, porous polyethylene coated with ceramic, or non-woven fabric.
[0082] In an embodiment, in the step 20, the reference electrode 2 is interposed between the separator 11 and the anode plate of the cell of the lithium ion battery 1. The upper end of the reference electrode 2 is protruded out from the cell of the lithium ion battery 1 for 1 mm to 2 mm under the water-free and oxygen-free condition.
[0083] In an embodiment, in the step S30, the lithium ion battery 1 implanted with the reference electrode 2 is packaged with an aluminum plastic film under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery 1 having the reference electrode 2.
[0084] Referring to FIG. 9, a method for making a lithium ion battery having a reference electrode is provided in the present disclosure. The method including the following steps:
[0085] S100, a reference electrode 2 is made as follows:
[0086] S110, a porous copper foam, a porous nickel foam, a copper mesh, or a nickel mesh is adopted as the reference electrode substrate 7. The reference electrode substrate 7 is washed with acetone or deionized water and then dried for later use. The material of the reference electrode substrate 7 has a pore size of about 50 .mu.m to about 500 .mu.m. A thickness of the reference electrode substrate 7 is about 0.1 mm to about 1 mm. An area of the reference electrode substrate 7 is about 1% to about 10% of an area of an electrode plate of the lithium ion battery 1.
[0087] S120, the reference electrode substrate 7 obtained from the step S110 is welded to a lower portion of a current collecting metal sheet 6, an upper portion of which is adhered with a tab binder 5, to allow an upper portion of the reference electrode substrate 7 to be overlapped with the lower portion of the current collecting metal sheet 6, vacuum dried at about 60.degree. C. to about 90.degree. C. under a water-free and oxygen-free condition for about 4 to about 7 hours, cooled, and then transferred into and preserved in a water-free and oxygen-free environment. The current collecting metal sheet 6 is used for collecting an electric current. A material of the current collecting metal sheet 6 is nickel or aluminum. A thickness of the current collecting metal sheet 6 is about 0.1 mm to about 1 mm. A length of the current collecting metal sheet 6 depends on a location of the reference electrode 2 disposed with respect to the battery and can be about 10 mm to about 30 mm. The tab binder 5 disposed on the upper portion of the current collecting metal sheet 6 is configured to fix the reference electrode 2 relative to the cell of the lithium ion battery 1 when the reference electrode 2 is inserted into the cell, as shown in FIGS. 3 and 5.
[0088] S130, lithium metal is melted to liquid state and continued to heat to about 200.degree. C. to about 500.degree. C. to remove an impurity on a surface of the liquid state lithium metal under the water-free and oxygen-free condition. The lower portion of the reference electrode substrate 7 whose upper portion is welded with the current collecting metal sheet 6 obtained from the step S120 is immersed into the liquid lithium for about 1 min to about 5 min. The portion without any welding spot 8 of the substrate should be entirely immersed into the liquid lithium. The lower portion of the reference electrode substrate 7 is fully wetted by the lithium metal, so as to allow a lithium metal layer 9 having a thickness of about 10 .mu.m to about 100 .mu.m to be formed on the lower portion of the reference electrode substrate 7. The electrode is taken out, cooled, and a portion below the tab binder 5 is wrapped with a separator by a winding method to allow the separator to be wrapped tightly and entirely around the current collecting metal sheet 6 and the reference electrode substrate 7, thereby obtaining a reference electrode 2 wrapped with the separator. A material of the separator is porous polypropylene, porous polyethylene, porous polypropylene coated with ceramic, porous polyethylene coated with ceramic, or non-woven fabric. The separator can prevent the reference electrode 2 from directly contacting with a cathode 3 and an anode 4 of the lithium ion battery 1 and can allow lithium ions to pass therethrough.
[0089] S200, the reference electrode 2 obtained from the step S100 is interposed between a separator 11 and an anode plate of a cell of the lithium ion battery 1 under the water-free and oxygen-free condition, as shown in FIG. 7. An upper end of the reference electrode 2 is protruded out from the cell of the lithium ion battery 1 for 1 mm to 2 mm. In order to show the location of the reference electrode 2 disposed with respect to the cell of the lithium ion battery 1, only the cathode plate 10 in the cell of the lithium ion battery 1 is shown while the anode plate of the lithium ion battery 1 is not shown.
[0090] S300, the lithium ion battery 1 implanted with the reference electrode 2 is packed with an aluminum plastic film under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery 1 having the reference electrode 2.
[0091] Examples of making the lithium ion battery 1 having the reference electrode 2 according to the present disclosure are described as below.
EXAMPLE 1
[0092] S100, a reference electrode 2 is made, and a specific process is as below.
[0093] S110, a porous copper foam is provided as a reference electrode substrate 7. A material of the reference electrode substrate 7 has a pore size of 50 .mu.m. A thickness of the reference electrode substrate 7 is 0.2 mm. An area of the reference electrode substrate 7 is 2% of an area of an electrode plate of a lithium ion battery 1. The reference electrode substrate 7 is a rectangle sheet with a length of 10 mm and a width of 5 mm. The reference electrode substrate 7 is washed with acetone or deionized water and then dried for use.
[0094] S120, the reference electrode substrate 7 obtained from the step S110 is welded to a lower portion of a current collecting metal sheet 6 to allow an upper portion of the reference electrode substrate 7 to be overlapped with the lower portion of the current collecting metal sheet 6. An upper portion of the current collecting metal sheet 6 is adhered with a tab binder 5. In this embodiment, preferably, the current collecting nickel sheet 6 having a same width as the reference electrode substrate 7 and a length of 20 mm, provided with the tab binder 5, is used for welding. The reference electrode substrate 7 is vacuum dried at 80.degree. C. under the water-free and oxygen-free condition for 5 hours, cooled, and transferred into and preserved in a water-free and oxygen-free environment.
[0095] S130, lithium metal is melted into liquid state and continued to heat to 400.degree. C. to remove an impurity on a surface of the liquid state lithium metal under the water-free and oxygen-free condition. The lower portion of the reference electrode substrate 7 whose upper portion is welded with the current collecting metal sheet 6 obtained from the step S120 is immersed into the liquid lithium for 3 min. The portion without any welding spot 8 of the substrate is entirely immersed into the liquid lithium. The lower portion of the reference electrode substrate 7 is fully wetted by the lithium metal, so as to allow a lithium metal layer to coat on the lower portion of the reference electrode substrate 7. The reference electrode 2 is taken out from the liquid lithium, a surface of the reference electrode 2 is immediately blown with hot air at 400.degree. C. to remove the lithium filled in pores to expose the pores. In this embodiment, a thickness of the lithium metal layer 9 grown is about 10 .mu.m. After being cooled, a portion below the tab binder 5 of the reference electrode 2 is wrapped with a separator by a winding method. The current collecting metal sheet 6 and the entire reference electrode substrate 7 are wrapped tightly, thereby obtaining the reference electrode 2 wrapped with the separator. A material of the separator is porous polypropylene. The separator can prevent the reference electrode 2 from directly contacting with a cathode 3 and an anode 4 of the lithium ion battery 1 and can allow lithium ions to pass therethrough;
[0096] S200, the reference electrode 2 obtained from the step S100 is interposed between a separator 11 and an anode plate of a cell of the lithium ion battery 1 under the water-free and oxygen-free condition. In this embodiment, preferably, a soft package battery is an object to be tested. The cell of the battery is placed into the water-free and oxygen-free environment. The reference electrode 2 is interposed between a cathode plate 10 and an anode plate of the cell, more specifically, located at an interface between a separator 11 and the anode plate. The reference electrode 2 can be interposed between any two plates except the outermost electrode plate. The reference electrode 2 can be located at anywhere on the plate, including but not limited to a center, a corner, and an edge of the plate according to needs. In this embodiment, the reference electrode 2 is interposed from a central area of an edge, while an upper end of the reference electrode 2 is protruded out from the cell of the lithium ion battery for 2 mm.
[0097] S300, the lithium ion battery 1 provided with the reference electrode 2 obtained from the step S200 is packaged by an aluminum plastic film under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery 1 having the reference electrode 2.
EXAMPLE 2
[0098] S100, a reference electrode 2 is made, and a specific process is as below.
[0099] S110, a porous nickel foam is provided as a reference electrode substrate 7. A material of the reference electrode substrate 7 has a pore size of 100 .mu.m. A thickness of the reference electrode substrate 7 is 0.2 mm. An area of the reference electrode substrate 7 is 5% of an area of an electrode plate of a lithium ion battery 1. The reference electrode substrate 7 is sized to a rectangle sheet with a length of 12 mm and a width of 7 mm. The reference electrode substrate 7 is washed with acetone or deionized water and then dried for use.
[0100] S120, the reference electrode substrate 7 obtained from the step S110 is welded to a lower portion of a current collecting metal sheet 6 to allow an upper portion of the reference electrode substrate 7 to be overlapped with the lower portion of the current collecting metal sheet 6. An upper portion of the current collecting metal sheet 6 is adhered with a tab binder 5. In this embodiment, preferably, the current collecting nickel sheet 6 having a same width as the reference electrode substrate 7 and a length of 20 mm and provided with the tab binder 5 is used for welding. The reference electrode substrate 7 is vacuum dried at 90.degree. C. under the water-free and oxygen-free condition for 5 hours, cooled, and transferred into and preserved in a water-free and oxygen-free environment;
[0101] S130, lithium metal is melted into liquid state and continued to heat to 400.degree. C. to remove an impurity on a surface of the liquid state lithium metal under the water-free and oxygen-free condition. The lower portion of the reference electrode substrate 7 whose the upper portion is welded with the current collecting metal sheet 6 obtained from the step S120 is immersed into the liquid lithium for 4 min. The portion without any welding spot 8 of the substrate is entirely immersed into the liquid lithium. The lower portion of the reference electrode substrate 7 is fully wetted by the lithium metal, so as to allow a lithium metal layer to coat on the lower portion of the reference electrode substrate 7. The reference electrode 2 is taken out from the liquid lithium, a surface of the reference electrode 2 is immediately blown with hot air at 400.degree. C. to remove the lithium filled in pores to expose the pores. In this embodiment, a thickness of the lithium metal layer 9 grown is about 30 .mu.m. After being cooled, a portion, below the tab binder 5 of the reference electrode 2 is wrapped with a separator by a winding method. The current collecting metal sheet 6 and the entire reference electrode substrate 7 are wrapped tightly, thereby obtaining the reference electrode 2 wrapped with the separator. A material of the separator is porous polypropylene. The separator can prevent the reference electrode 2 from directly contacting with a cathode 3 and an anode 4 of the lithium ion battery 1 and can allow lithium ions to pass therethrough.
[0102] S200, the reference electrode 2 obtained from the step S100 is interposed between a separator 11 and an anode plate of a cell of the lithium ion battery 1 under the water-free and oxygen-free condition. In this embodiment, preferably, a soft package battery is an object to be tested. The cell of the battery is placed into the water-free and oxygen-free environment. The reference electrode 2 is interposed between a cathode plate 10 and an anode plate of the cell, more specifically, disposed at an interface between a separator 11 and the anode plate. The reference electrode 2 can be interposed between any two plates except the outermost electrode plate. The reference electrode 2 can be located at anywhere on the plate, including but not limited to a center, a corner, and an edge of the plate according to needs. In this embodiment, the reference electrode 2 is interposed from a central area of an edge, while an upper end of the reference electrode 2 is protruded out from the cell of the lithium ion battery for 2 mm.
[0103] S300, the lithium ion battery 1 provided with the reference electrode 2 obtained from the step S200 is packaged by an aluminum plastic film under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery 1 having the reference electrode 2.
EXAMPLE 3
[0104] S100, a reference electrode 2 is made, and a specific process is as below.
[0105] S110, a porous copper mesh is provided as a reference electrode substrate 7. A material of the reference electrode substrate 7 has a pore size of 300 .mu.m. A thickness of the reference electrode substrate 7 is 0.2 mm. An area of the reference electrode substrate 7 is 2% of an area of an electrode plate of a lithium ion battery 1. The reference electrode substrate 7 is a rectangle sheet with a length of 20 mm and a width of 10 mm. The reference electrode substrate 7 is washed with acetone or deionized water and then dried for use.
[0106] S120, the reference electrode substrate 7 obtained from the step S110 is welded to a lower portion of a current collecting metal sheet 6 to allow an upper portion of the reference electrode substrate 7 to be overlapped with the lower portion of the current collecting metal sheet 6. An upper portion of the current collecting metal sheet 6 is adhered with a tab binder 5. In this embodiment, preferably, the current collecting nickel sheet 6 having a same width as the reference electrode substrate 7 and a length of 20 mm and provided with the tab binder 5 is used for welding. The reference electrode substrate 7 is vacuum dried at 80.degree. C. under the water-free and oxygen-free condition for 5 hours, cooled, and transferred into and preserved in a water-free and oxygen-free environment;
[0107] S130, a lithium metal is melted into liquid state and continued to heat to 400.degree. C. to remove an impurity on a surface of the liquid state lithium metal under the water free and oxygen-free condition. The lower portion of the reference electrode substrate 7 whose upper portion is welded with the current collecting metal sheet 6 obtained from the step S120 is immersed into the liquid lithium for 3 min. The portion without any welding spot 8 of the substrate is entirely immersed into the liquid lithium. The lower portion of the reference electrode substrate 7 is fully wetted by the lithium metal, so as to allow a lithium metal layer to coat on the lower portion of the reference electrode substrate 7. The reference electrode 2 is taken out from the liquid lithium, and a surface of the reference electrode 2 is immediately blown with hot air at 400.degree. C. to remove the lithium filled in pores to expose the pores. In this embodiment, a thickness of the lithium metal layer 9 grown is about 100 .mu.m. After being cooled, a portion below the tab binder 5 of the reference electrode 2 is wrapped with a separator by a winding method. The current collecting metal sheet 6 and the entire reference electrode substrate 7 are wrapped tightly, thereby obtaining the reference electrode 2 wrapped with the separator. A material of the separator is porous polypropylene. The separator can prevent the reference electrode 2 from directly contacting with a cathode 3 and an anode 4 of the lithium ion battery 1 and can allow lithium ions to pass therethrough.
[0108] S200, the reference electrode 2 obtained from the step S100 is interposed between a separator 11 and an anode plate of a cell of the lithium ion battery 1 under the water-free and oxygen-free condition. In this embodiment, preferably, a soft package battery is an object to be tested. The cell of the battery is placed into the water-free and oxygen-free environment. The reference electrode 2 is interposed between a cathode plate 10 and an anode plate of the cell, more specifically, disposed at an interface between a separator 11 and the anode plate. The reference electrode 2 can be interposed between any two plates except the outermost electrode plate. The reference electrode 2 can be located at anywhere on the plate, including but not limited to a center, a corner, and an edge according to needs. In this embodiment, the reference electrode 2 is interposed from a central area of an edge, while an upper end of the reference electrode 2 is protruded out from the cell of the lithium ion battery for 2 mm.
[0109] S300, the lithium ion battery 1 provided with the reference electrode 2 obtained from the step S200 is packaged by an aluminum plastic film under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery 1 having the reference electrode 2.
EXAMPLE 4
[0110] S100, a reference electrode 2 is made, and a specific process is as below.
[0111] S110, a porous nickel mesh is provided as a reference electrode substrate 7. A material of the reference electrode substrate 7 has a pore size of 500 .mu.m. A thickness of the reference electrode substrate 7 is 0.2 mm. An area of the reference electrode substrate 7 is 1% of an area of an electrode plate of a lithium ion battery 1. The reference electrode substrate 7 is a rectangle sheet with a length of 20 mm and a width of 8 mm. The reference electrode substrate 7 is washed with acetone or deionized water and then dried for use.
[0112] S120, the reference electrode substrate 7 obtained from the step S110 is welded to a lower portion of a current collecting metal sheet 6 to allow an upper portion of the reference electrode substrate 7 to be overlapped with the lower portion of the current collecting metal sheet 6. An upper portion of the current collecting metal sheet 6 is adhered with a tab binder 5. In this embodiment, preferably, a current collecting nickel sheet 6 having a same width as the reference electrode substrate 7 and a length of 20 mm and provided with the tab binder 5 is used for welding. The reference electrode substrate 7 is vacuum dried at 80.degree. C. under the water-free and oxygen-free condition for 5 hours, cooled, and transferred into and preserved in a water-free and oxygen-free environment.
[0113] S130, lithium metal is melted into liquid state and continued to heat to 400.degree. C. to remove an impurity on a surface of the liquid state lithium metal under the water-free and oxygen-free condition. The lower portion of the reference electrode substrate 7 whose upper portion is welded with the current collecting metal sheet 6 obtained from the step S120 is immersed into the liquid lithium for 3 min. The portion without any welding spot 8 of the substrate is entirely immersed into the liquid lithium, and the lower portion of the reference electrode substrate 7 is fully wetted by the lithium metal, so as to allow a lithium metal layer to coat on the lower portion of the reference electrode substrate 7. A reference electrode 2 is taken out from the liquid lithium and a surface of the reference electrode 2 is immediately blown with hot air at 400.degree. C. to remove the lithium filled in pores to expose the pores. In this embodiment, a thickness of the lithium metal layer 9 grown is about 100 .mu.m. After being cooled, a portion below the tab binder 5 of the reference electrode 2 is wrapped with a separator by a winding method. The current collecting metal sheet 6 and the entire reference electrode substrate 7 are wrapped tightly, thereby obtaining the reference electrode 2 wrapped with the separator. A material of the separator is porous polypropylene. The separator can prevent the reference electrode 2 from directly contacting with a cathode 3 and an anode 4 of the lithium ion battery 1 and can allow lithium ions to pass therethrough.
[0114] S200, the reference electrode 2 obtained from the step S100 is interposed between a separator 11 and an anode plate of a cell of the lithium ion battery 1 under the water-free and oxygen-free condition. In this embodiment, preferably, a soft package battery is an object to be tested. The cell of the battery is placed into the water-free and oxygen-free environment. The reference electrode 2 is interposed between a cathode plate 10 and an anode plate of the cell, more specifically, disposed at an interface between a separator 11 and the anode plate. The reference electrode 2 can be interposed between any two plates except the outermost electrode plate. The reference electrode 2 can be located at anywhere on the plate, including but not limited to a center, a corner, and an edge according to needs. In this embodiment, the reference electrode 2 is interposed from a central area of an edge, while an upper end of the reference electrode 2 is protruded out from the cell of the lithium ion battery for 2 mm.
[0115] S300, the lithium ion battery 1 provided with the reference electrode 2 obtained from the step S200 is packaged by an aluminum plastic film under the water-free and oxygen-free condition, thereby obtaining the lithium ion battery 1 having the reference electrode 2.
[0116] A charge-discharge test is performed by connecting the cathode 3 and the anode 4 of the lithium ion battery 1 having the reference electrode 2 to an instrument such as a charge-discharge tester or other power supply. The performance of assembled lithium ion battery 1 having the reference electrode 2 is measured by the measuring instrument. As shown in FIG. 10 which is a schematic measuring circuit diagram, a high input impedance voltage synchronous acquisition equipment having three testing channels is used, an external voltage U1 of the lithium ion battery 1 having the reference electrode 2 is measured and acquired via the channel 1, an anode-reference voltage U2 is measured and acquired via the channel 2, and a cathode-reference voltage U3 is measured and acquired via the channel 3. Moreover, different types of charge-discharge cycling tests with a higher cutoff voltage of 4.2 V and a lower cutoff voltage of 2.5 V are performed on the lithium ion battery 1 having the reference electrode 2 via the channel 1 during which charge and discharge currents are recorded. Result analyses for two types of charge-discharge cycling tests are described as below.
[0117] FIG. 11 is a diagram showing results of a charge-discharge rate performance test for the battery having the reference electrode 2, in which a horizontal axis denotes a time during the test and a vertical axis denotes a tested voltage. External voltage is shown by the solid line. Voltage of the individual electrode, which is the anode 4, with respect to the reference electrode 2 is shown by the dotted line. During the test, the charge-discharge current rate gradually increased from 0.5 C to 1.5 C. More specifically, the battery is charged and discharged at 0.5 C in the first charge-discharge cycle, charged and discharged at 1 C in the second charge-discharge cycle, and charged and discharged at 1.5 C in the third charge-discharge cycle, as is shown in the FIG. 11. It can be found upon this test that the variations of the external voltage in the three different charge-discharge cycles using different charge-discharge rates are consistent, while the variations of the voltage of the anode 4 tested with respect to the reference electrode 2 clearly tend to increase with the increase of the charge-discharge rate. Such feature that the voltage of the anode 4 is changed with the change of the charge-discharge rate, cannot be tested by only measuring the external voltage. By using the reference electrode 2, this voltage variation for an individual electrode can be directly and conveniently tested, which demonstrates the superiority of the testing using the reference electrode 2.
[0118] FIG. 12 is a diagram showing results of a capacity test for the battery having the reference electrode 2, in which a horizontal axis denotes a time during the test and a vertical axis denotes a tested voltage. External voltage of the battery is shown by the solid line. Voltage of the individual electrode, which is the anode 4, with respect to the reference electrode 2 of the battery is shown by the dotted line. During the test, the charge-discharge current rate is kept at 1 C. It can be seen that the voltage of the anode 4 as the individual electrode with respect to the reference electrode is stable in each charge-discharge cycle, while the voltage of the whole battery is also stable in each charge-discharge cycle, suggesting that the reference electrode 2 has a satisfactory service time and does not interfere with the normal operation of the battery.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.