Tetravalent Anti-psgl-1 Antibodies And Uses Thereof
LIN; Rong-Hwa ; et al.
U.S. patent application number 16/584356 was filed with the patent office on 2020-09-03 for tetravalent anti-psgl-1 antibodies and uses thereof. The applicant listed for this patent is AbGenomics International Inc., BioAlliance C.V.. Invention is credited to Rong-Hwa LIN, Shih-Yao LIN, Yu-Ying TSAI.
| Application Number | 20200277395 16/584356 |
| Document ID | / |
| Family ID | 1000004842807 |
| Filed Date | 2020-09-03 |





| United States Patent Application | 20200277395 |
| Kind Code | A1 |
| LIN; Rong-Hwa ; et al. | September 3, 2020 |
TETRAVALENT ANTI-PSGL-1 ANTIBODIES AND USES THEREOF
Abstract
Provided herein are tetravalent antibodies that specifically bind to human PSGL-1. Unlike bivalent antibodies, these tetravalent antibodies contain a dimer of two monomers, with each monomer comprising two light chain variable (VL) domains and two heavy chain variable (VH) domains. This format allows for cross-linker/FcR-expressing cell-independent tetravalent antibodies against PSGL-1 that show enhanced efficacy as compared to bivalent PSGL-1 antibodies. These tetravalent antibodies can be used in a variety of diagnostic and therapeutic methods, including without limitation treating T-cell mediated inflammatory diseases, transplantations, and transfusions.
| Inventors: | LIN; Rong-Hwa; (Palo Alto, CA) ; LIN; Shih-Yao; (Taipei, TW) ; TSAI; Yu-Ying; (Taipei City, TW) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 1000004842807 | ||||||||||
| Appl. No.: | 16/584356 | ||||||||||
| Filed: | September 26, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15400888 | Jan 6, 2017 | 10472422 | ||
| 16584356 | ||||
| 62276806 | Jan 8, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 2317/622 20130101; C07K 2317/53 20130101; C07K 2317/76 20130101; C07K 2317/626 20130101; C07K 2317/56 20130101; C07K 2317/35 20130101; C07K 2317/21 20130101; C07K 2317/24 20130101; C07K 2317/732 20130101; C07K 2317/52 20130101; C07K 16/2896 20130101; C07K 2317/565 20130101; A61P 29/00 20180101 |
| International Class: | C07K 16/28 20060101 C07K016/28; A61P 29/00 20060101 A61P029/00 |
Claims
1. A tetravalent antibody that specifically binds to human PSGL-1, the tetravalent antibody comprising a dimer of two monomers, wherein each monomer of the dimer comprises a single-chain polypeptide comprising, from N-terminus to C-terminus: (a) a first light chain variable (VL) domain; (b) a first linker sequence; (c) a first heavy chain variable (VH) domain; (d) a second linker sequence; (e) a second VL domain; (f) a third linker sequence; (g) a second VH domain; (h) a fourth linker sequence; and (i) an antibody Fc domain, wherein each of the first and the second VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3; wherein each of the first and the second VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3; and wherein each of the first and the second VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and the second VH domains, and wherein each of the two VH-VL binding units is specific for human PSGL-1.
2. The tetravalent antibody of claim 1, wherein at least one of the two VH domains comprises: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19; and/or wherein at least one of the two VL domains comprises: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22.
3. (canceled)
4. The tetravalent antibody of claim 2, wherein each of the two VH domains comprises the amino acid sequence of SEQ ID NO:23; an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:23; the amino acid sequence of SEQ ID NO:29; or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:29; and/or wherein each of the two VL domains comprises the amino acid sequence of SEQ ID NO:24; an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:24; the amino acid sequence of SEQ ID NO:30; or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:30.
5-9. (canceled)
10. The tetravalent antibody of claim 1, wherein the first, second and third linker sequences each comprise two or more repeats of the amino acid sequence of SEQ ID NO:25, or the first, second or third linker sequence comprises the amino acid sequence of SEQ ID NO:33, 34, 35, or 36.
11. (canceled)
12. (canceled)
13. The tetravalent antibody of claim 1, wherein the fourth linker sequence comprises the amino acid sequence of SEQ ID NO:26.
14. The tetravalent antibody of claim 1, wherein each of the two single-chain polypeptides comprises the amino acid sequence of SEQ ID NO:1, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:1.
15. (canceled)
16. A tetravalent antibody that specifically binds to human PSGL-1, the tetravalent antibody comprising a dimer of two monomers, wherein each monomer of the dimer comprises a single-chain polypeptide comprising, from N-terminus to C-terminus: (a) a first heavy chain variable (VH) domain; (b) a first linker sequence; (c) a first light chain variable (VL) domain; (d) a second linker sequence; (e) a second VL domain; (f) a third linker sequence; (g) a second VH domain; (h) a fourth linker sequence; and (i) an antibody Fc domain, wherein each of the first and the second VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3; wherein each of the first and the second VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3; and wherein each of the first and the second VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and the second VH domains, and wherein each of the two VH-VL binding units is specific for human PSGL-1.
17. The tetravalent antibody of claim 16, wherein at least one of the two VH domains comprises: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19; and/or wherein at least one of the two VL domains comprises: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22.
18. (canceled)
19. The tetravalent antibody of claim 17, wherein each of the two VH domains comprises the amino acid sequence of SEQ ID NO:23; an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:23; the amino acid sequence of SEQ ID NO:29; or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:29; and/or wherein each of the two VL domains comprises the amino acid sequence of SEQ ID NO:24; an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:24; the amino acid sequence of SEQ ID NO:30; or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:30.
20-24. (canceled)
25. The tetravalent antibody of claim 16, wherein the first and the third linker sequences have the same sequence comprising five repeats of SEQ ID NO:25.
26. (canceled)
27. The tetravalent antibody of claim 16, wherein the fourth linker sequence comprises the amino acid sequence of SEQ ID NO:26.
28. The tetravalent antibody of claim 16, wherein each of the two single-chain polypeptides comprises the amino acid sequence of SEQ ID NO:3, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:3.
29. (canceled)
30. A tetravalent antibody that specifically binds to human PSGL-1, the tetravalent antibody comprising a dimer of two monomers, wherein each monomer of the dimer comprises an antibody heavy chain and an antibody light chain; wherein the antibody light chain comprises, from N-terminus to C-terminus: (i) a first heavy chain variable (VH) domain, (ii) a first linker sequence, (iii) a first light chain variable (VL) domain, (iv) a second linker sequence, (v) a second VL domain, and (vi) a light chain constant (CL) domain; wherein the antibody heavy chain comprises: (i) a second VH domain, and (ii) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain; wherein each of the first and the second VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3; wherein each of the first and the second VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3; and wherein each of the first and the second VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and the second VH domains, and wherein each of the two VH-VL binding units is specific for human PSGL-1.
31. The tetravalent antibody of claim 30, wherein at least one of the first and the second VH domains comprises: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19; and/or wherein at least one of the first and the second VL domains comprises: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22.
32. (canceled)
33. The tetravalent antibody of claim 31, wherein the first and the second VH domains each comprise the amino acid sequence of SEQ ID NO:23; an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:23; the amino acid sequence of SEQ ID NO:29; or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:29; and/or wherein each of the two VL domains comprises the amino acid sequence of SEQ ID NO:24; an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:24; the amino acid sequence of SEQ ID NO:30; or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:30.
34-38. (canceled)
39. The tetravalent antibody of claim 30, wherein the CL domain is a kappa CL domain.
40. The tetravalent antibody of claim 30, wherein the first linker sequence comprises five repeats of SEQ ID NO:25; and/or wherein the second linker sequence comprises the amino acid sequence of SEQ ID NO:28.
41. (canceled)
42. The tetravalent antibody of claim 30, wherein the antibody light chain comprises the amino acid sequence of SEQ ID NO:7, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:7; and/or wherein the antibody heavy chain comprises the amino acid sequence of SEQ ID NO:11, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:11.
43-45. (canceled)
46. The tetravalent antibody of claim 1, wherein the antibody Fc domain is a human antibody Fc domain.
47-49. (canceled)
50. An isolated polynucleotide encoding the tetravalent antibody of claim 1.
51. (canceled)
52. A vector comprising the isolated polynucleotide of claim 50.
53. A host cell comprising the polynucleotide of claim 50.
54. A method of producing a tetravalent antibody comprising culturing the host cell of claim 53 so that the tetravalent antibody is produced.
55. (canceled)
56. A pharmaceutical composition comprising the tetravalent antibody of claim 1 and a pharmaceutically acceptable carrier.
57-63. (canceled)
64. A method of treating a T-cell mediated inflammatory disease, the method comprising administering to a subject in need thereof a therapeutically effective amount of the tetravalent antibody of claim 1.
65. A method for treating an individual in need of a transfusion or transplantation, comprising administering to the individual a therapeutically effective amount of the tetravalent antibody of claim 1 before, concurrently with, and/or after the transfusion or transplantation.
66-70. (canceled)
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the priority benefit of U.S. Provisional Application Ser. No. 62/276,806, filed Jan. 8, 2016, which is incorporated herein by reference in its entirety.
SUBMISSION OF SEQUENCE LISTING ON ASCII TEXT FILE
[0002] The content of the following submission on ASCII text file is incorporated herein by reference in its entirety: a computer readable form (CRF) of the Sequence Listing (file name: 606592001300SEQLIST.TXT, date recorded: Jan. 3, 2017, size: 70 KB).
FIELD
[0003] Provided herein are tetravalent antibodies that specifically bind to human P-selectin glycoprotein ligand-1 (PSGL-1), as well as polynucleotides, vectors, host cells, methods, pharmaceutical compositions, kits, and uses related thereto. These tetravalent antibodies may find use in a variety of diagnostic and therapeutic methods, including without limitation treating T-cell mediated inflammatory diseases, transplantations, and transfusions.
BACKGROUND
[0004] Inflammatory responses to infection or injury are initiated by the adherence of leukocytes to the vascular wall (McEver et al, 1997, J. Clin. Invest., 100 (3): 485-492). Selectin represents a family of glycoproteins which mediate the first leukocyte-endothelial cell and leukocyte-platelet interactions during inflammation. The selectin family, which consists of L-selectin, E-selectin, and P-selectin, comprises an NH2-terminal lectin domain, followed by an EGF-like domain, a series of consensus repeats, a transmembrane domain, and a short cytoplasmic tail. The lectin domains of selectins interact with specific glycoconjugate ligands in order to facilitate cell adhesion. L-selectin, expressed on most leukocytes, binds to ligands on some endothelial cells and other leukocytes. E-selectin, expressed on cytokine activated endothelial cells, binds to ligands on most leukocytes. P-selectin, expressed on activated platelets and endothelial cells, also binds to ligands on most leukocytes.
[0005] P-selectin glycoprotein ligand-1 ("PSGL-1"), also known as SELPLG or CD162 (cluster of differentiation 162) is a human mucin-type glycoprotein ligand for all three selectins (Constantin, Gabriela, 2004, Drug News Perspect., 17(9): 579-585; McEver et al., 1997, J. Clin. Invest., 100 (3): 485-492). PSGL-1 is a disulfide-bonded homodimer with two 120-kD subunits and is expressed on the surface of monocytes, lymphocytes, granulocytes, and in some CD34.sup.+ stem cells. PSGL-1 is likely to contribute to pathological leukocyte recruitment in many inflammatory disorders since it facilitates the adhesive interactions of selectins. In addition, PSGL-1 is shown to have a unique regulatory role in T cells. Mice deficient in PSGL-1 show enhanced proliferative responses and autoimmunity, suggesting that PSGL-1 plays an important role in down-regulating T cell responses (Krystle M. et al. J. Immunol. 2012; 188:1638-1646. Urzainqui et al. Ann Rheum Dis 2013; 71:650; Perez-Frias A, et al. Arthritis Rheumatol. 2014 November; 66(11):3178-89.; Angiari et al. J Immunol. 2013; 191(11):5489-500).
[0006] Several anti-PSGL-1 antibodies have been developed (see, e.g., International Application Pub. Nos. WO 2005/110475, WO 2003/013603, and WO 2012/174001; Constantin, Gabriela, 2004, Drug News Perspect., 17(9): 579-585, Chen et al. Blood. 2004; 104(10):3233-42, Huang et al, Eur J Immunol. 2005; 35(7):2239-49; and U.S. Pat. No. 7,604,800). Some of the existing agonistic PSGL-1 antibodies preferentially induce apoptosis of late-stage activated T cells but not other PSGL-1-expressing cells; such antibodies may therefore be useful as anti-inflammatory therapeutics, or for use in transplantations and/or transfusions. However, a need exists for improved anti-PSGL-1 antibodies with greater in vivo efficacy than existing antibodies.
[0007] All publications, patents, and patent applications cited herein are hereby incorporated by reference in their entirety for all purposes.
BRIEF SUMMARY
[0008] To meet this need, provided herein are tetravalent antibodies that specifically bind to human PSGL-1, as well as polynucleotides, vectors, host cells, methods, pharmaceutical compositions, kits, and uses related thereto. The present disclosure demonstrates that tetravalent antibodies that specifically bind to human PSGL-1 have greater potency and efficacy than conventional (e.g., bivalent) anti-PSGL-1 antibodies. As such, these tetravalent antibodies may find use, inter alia, in diagnostic and/or therapeutic methods, uses, and compositions related to T-cell function, such as in treating T-cell mediated inflammatory diseases, transfusions, and/or transplantations.
[0009] Accordingly, in one aspect, provided herein is a tetravalent antibody that specifically binds to human PSGL-1, the tetravalent antibody comprising a dimer of two monomers, wherein each monomer of the dimer comprises a single-chain polypeptide comprising: (a) two light chain variable (VL) domains, wherein each of the two VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3; (b) two heavy chain variable (VH) domains, wherein each of the two VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3; and (c) an antibody Fc domain, wherein each of the two VL domains forms a VH-VL binding unit with a corresponding VH domain of the two VH domains, and wherein each of the two VH-VL binding units is specific for human PSGL-1. In some embodiments, at least one of the two VH domains comprises: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19. In some embodiments, each of the two VH domains comprises: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19. In some embodiments, one or both of the two VH domains comprises the amino acid sequence of SEQ ID NO:23, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:23. In some embodiments, one or both of the two VH domains comprises the amino acid sequence of SEQ ID NO:29, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:29. In some embodiments, at least one of the two VL domains comprises: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22. In some embodiments, each of the two VL domains comprises: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22. In some embodiments, one or both of the two VL domains comprises the amino acid sequence of SEQ ID NO:24, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:24. In some embodiments, one or both of the two VL domains comprises the amino acid sequence of SEQ ID NO:30, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:30. In some embodiments, each of the two single-chain polypeptides comprises, from N-terminus to C-terminus: (a) a first VL domain of the two VL domains; (b) a first linker sequence; (c) a first VH domain of the two VH domains; (d) a second linker sequence; (e) a second VL domain of the two VL domains; (f) a third linker sequence; (g) a second VH domain of the two VH domains; (h) a fourth linker sequence; and (i) the antibody Fc domain. In some embodiments, the first, second and third linker sequences each comprise two or more repeats of the amino acid sequence of SEQ ID NO:25. In some embodiments, the first and the third linker sequences have the same sequence and comprise two repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises five repeats of SEQ ID NO:25. In some embodiments, the fourth linker sequence comprises the amino acid sequence of SEQ ID NO:26. In some embodiments, each of the two single-chain polypeptides comprises the amino acid sequence of SEQ ID NO:1, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:1. In some embodiments, each of the two single-chain polypeptides is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:2. In some embodiments, each of the two single-chain polypeptides comprises, from N-terminus to C-terminus: (a) a first VH domain of the two VH domains; (b) a first linker sequence; (c) a first VL domain of the two VL domains; (d) a second linker sequence; (e) a second VL domain of the two VL domains; (f) a third linker sequence; (g) a second VH domain of the two VH domains; (h) a fourth linker sequence; and (i) the antibody Fc domain. In some embodiments, each of the two single-chain polypeptides comprises, from N-terminus to C-terminus: (a) a first VL domain of the two VL domains; (b) a first linker sequence; (c) a first VH domain of the two VH domains; (d) a second linker sequence; (e) a second VH domain of the two VH domains; (f) a third linker sequence; (g) a second VL domain of the two VL domains; (h) a fourth linker sequence; and (i) the antibody Fc domain. In some embodiments, the first, second or third linker sequence comprises two or more repeats of the amino acid sequence of SEQ IN NO:25. In some embodiments, the first, second or third linker sequence comprises the amino acid sequence of SEQ ID NO:33, 34, 35, or 36. In some embodiments, the first and the third linker sequences have the same sequence comprising five repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises the amino acid sequence of SEQ ID NO:27. In some embodiments, the fourth linker sequence comprises the amino acid sequence of SEQ ID NO:26. In some embodiments, each of the two single-chain polypeptides comprises the amino acid sequence of SEQ ID NO:3, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:3. In some embodiments, each of the two single-chain polypeptides is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:4. In some embodiments, each of the two single-chain polypeptides comprises the amino acid sequence of SEQ ID NO:5. In some embodiments, each of the two single-chain polypeptides is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:6.
[0010] In another aspect, provided herein is a tetravalent antibody that specifically binds to human PSGL-1, the tetravalent antibody comprising a dimer of two monomers, wherein each monomer of the dimer comprises an antibody heavy chain and an antibody light chain; wherein the antibody light chain comprises: (i) two light chain variable (VL) domains, wherein each of the two VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3, (ii) a first heavy chain variable (VH) domain, and (iii) a light chain constant (CL) domain; wherein the antibody heavy chain comprises: (i) a second heavy chain variable (VH) domain, and (ii) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain; wherein the first and the second VH domains each comprise a CDR-H1, a CDR-H2, and a CDR-H3, wherein each of the two VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and the second VH domains, and wherein each of the two VH-VL binding units is specific for human PSGL-1. In some embodiments, at least one of the first and the second VH domains comprises: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19. In some embodiments, the first and the second VH domains each comprise: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19. In some embodiments, the first and/or the second VH domains comprise the amino acid sequence of SEQ ID NO:23, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:23. In some embodiments, the first and/or the second VH domains comprise the amino acid sequence of SEQ ID NO:29, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:29. In some embodiments, at least one of the first and the second VL domains comprises: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22. In some embodiments, the first and the second VL domains each comprise: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22. In some embodiments, the first and/or the second VL domains comprise the amino acid sequence of SEQ ID NO:24, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:24. In some embodiments, the first and/or the second VL domains comprise the amino acid sequence of SEQ ID NO:30, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:30. In some embodiments, the antibody light chain comprises, from N-terminus to C-terminus: (a) the first VH domain; (b) a first linker sequence; (c) a first VL domain of the two or more VL domains; (d) a second linker sequence; (e) a second VL domain of the two or more VL domains; and (f) the CL domain. In some embodiments, the CL domain is a kappa CL domain. In some embodiments, the first linker sequence comprises five repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises the amino acid sequence of SEQ ID NO:28. In some embodiments, the antibody light chain comprises the amino acid sequence of SEQ ID NO:7, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:7. In some embodiments, the antibody light chain is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:8. In some embodiments, the antibody light chain comprises, from N-terminus to C-terminus: (a) a first VL domain of the two VL domains; (b) the CL domain; (c) a first linker sequence; (d) the first VH domain; (e) a second linker sequence; and (f) a second VL domain of the two VL domains. In some embodiments, the CL domain is a kappa CL domain. In some embodiments, the first linker sequence comprises two repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises five repeats of SEQ ID NO:25. In some embodiments, the antibody light chain comprises the amino acid sequence of SEQ ID NO:9. In some embodiments, the antibody light chain is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:10. In some embodiments, the antibody heavy chain comprises, from N-terminus to C-terminus: (a) the second VH domain; and (b) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain. In some embodiments, the antibody heavy chain comprises the amino acid sequence of SEQ ID NO:11, or an amino acid sequence having at least 90%, at least 95%, or at least 99% sequence identity to SEQ ID NO:11. In some embodiments, the antibody heavy chain is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:12.
[0011] In another aspect, provided herein is a tetravalent antibody that specifically binds to human PSGL-1, the tetravalent antibody comprising a dimer of two monomers, wherein each monomer of the dimer comprises an antibody heavy chain and an antibody light chain; wherein the antibody light chain comprises: (i) a first heavy chain variable (VH) domain, (ii) a first light chain variable (VL) domain, and (iii) a light chain constant (CL) domain; wherein the antibody heavy chain comprises: (i) a second heavy chain variable (VH) domain, (ii) a second light chain variable (VL) domain, and (iii) a heavy chain constant domain comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain; wherein each of the first and second VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3; wherein each of the first and second VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3; wherein each of the first and second VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and second VH domains; and wherein each of the two VH-VL binding units is specific for human PSGL-1. In some embodiments, at least one of the first and second VH domains comprises: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19. In some embodiments, the first and the second VH domains each comprise: (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19. In some embodiments, the first and/or the second VH domains comprise the amino acid sequence of SEQ ID NO:23. In some embodiments, the first and/or the second VH domains comprise the amino acid sequence of SEQ ID NO:29. In some embodiments, at least one of the first and second VL domains comprises: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22. In some embodiments, the first and the second VL domains each comprise: (i) a CDR-L1 comprising the amino acid sequence of SEQ ID NO:20; (ii) a CDR-L2 comprising the amino acid sequence of SEQ ID NO:21; and (iii) a CDR-L3 comprising the amino acid sequence of SEQ ID NO:22. In some embodiments, the first and/or the second VL domains comprise the amino acid sequence of SEQ ID NO:24. In some embodiments, the first and/or the second VL domains comprise the amino acid sequence of SEQ ID NO:30. In some embodiments, the antibody light chain comprises, from N-terminus to C-terminus: (a) the first VH domain; (b) a first linker sequence; (c) the first VL domain; and (d) the CL domain. In some embodiments, the CL domain is a kappa CL domain. In some embodiments, the first linker sequence comprises five repeats of SEQ ID NO:25. In some embodiments, the antibody light chain comprises the amino acid sequence of SEQ ID NO:13. In some embodiments, the antibody light chain is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:14. In some embodiments, the antibody heavy chain comprises, from N-terminus to C-terminus: (a) the second VH domain; (b) a second linker sequence; (c) the second VL domain; and (d) the heavy chain constant region comprising the first heavy chain constant region (CH1) domain, the antibody hinge region, the second heavy chain constant region (CH2) domain, and the third heavy chain constant region (CH3) domain. In some embodiments, the second linker sequence comprises five repeats of SEQ ID NO:25. In some embodiments, the antibody heavy chain comprises the amino acid sequence of SEQ ID NO:15. In some embodiments, the antibody heavy chain is encoded by a polynucleotide comprising the polynucleotide sequence of SEQ ID NO:16.
[0012] In some embodiments of any of the above embodiments, the antibody Fc domain is a human antibody Fc domain. In some embodiments, the antibody Fc domain is a human IgG4 Fc domain. In some embodiments, the human IgG4 Fc domain comprises a hinge region sequence comprising one or more amino acid substitutions that result in reduced IgG4 shuffling, as compared to an IgG4 hinge region lacking the one or more amino acid substitutions. In some embodiments, the human IgG4 Fc domain comprises a hinge region sequence comprising a serine to proline substitution at amino acid 228, numbering according to EU index. In some embodiments of any of the above embodiments, the antibody hinge region comprises a serine to proline substitution at amino acid 228, numbering according to EU index. In some embodiments, a tetravalent antibody of the present disclosure displays enhanced induction of apoptosis in a target cell (e.g., a cell expressing human PSGL-1 or an epitope thereof) as compared to a conventional (e.g., bivalent) antibody having one or more VH or VL domains in common with the tetravalent antibody. In some embodiments, a tetravalent antibody of the present disclosure displays enhanced inhibition of DTH (e.g., in a trans vivo animal model) as compared to a conventional (e.g., bivalent) antibody having one or more VH or VL domains in common with the tetravalent antibody.
[0013] In another aspect, provided herein is an isolated polynucleotide encoding the tetravalent antibody of any one of the above embodiments. In some embodiments, the isolated polynucleotide comprises a polynucleotide sequence selected from the group consisting of SEQ ID NOs:2, 4, 6, 8, 10, 12, 14, and 16. In another aspect, provided herein is a vector comprising the isolated polynucleotide of any of the above embodiments. In another aspect, provided herein is a host cell comprising the polynucleotide of any of the above embodiments and/or the vector of any of the above embodiments. In another aspect, provided herein is a method of producing a tetravalent antibody comprising culturing the host cell of any of the above embodiments so that the tetravalent antibody is produced. In some embodiments, the method further comprises recovering the tetravalent antibody from the host cell.
[0014] In another aspect, provided herein is a pharmaceutical composition comprising the tetravalent antibody of any one of the above embodiments and a pharmaceutically acceptable carrier. In another aspect, provided herein is a kit comprising the tetravalent antibody of any one of the above embodiments and an optional pharmaceutically acceptable carrier. In some embodiments, the kit further comprises a package insert comprising instructions for administration of the tetravalent antibody to treat a T-cell mediated inflammatory disease or condition. In some embodiments, the kit further comprises a package insert comprising instructions for administration of the tetravalent antibody before, concurrently with, and/or after a transfusion or transplantation. In another aspect, provided herein is the tetravalent antibody of any one of the above embodiments for use in treating a T-cell mediated inflammatory disease or condition. In another aspect, provided herein is the tetravalent antibody of any one of the above embodiments for use in treating an individual in need of a transfusion or transplantation. In another aspect, provided herein is a use of the tetravalent antibody of any one of the above embodiments in the manufacture of a medicament for treating a T-cell mediated inflammatory disease or condition. In another aspect, provided herein is a use of the tetravalent antibody of any one of the above embodiments in the manufacture of a medicament for treating an individual in need of a transfusion or transplantation. In another aspect, provided herein is a method of treating a T-cell mediated inflammatory disease or condition, the method comprising administering to a subject in need thereof a therapeutically effective amount of the tetravalent antibody of any one of the above embodiments. In another aspect, provided herein is a method for treating an individual in need of a transfusion or transplantation, comprising administering to the individual a therapeutically effective amount of the tetravalent antibody of any one of the above embodiments before, concurrently with, and/or after the transfusion or transplantation. In some embodiments, the T-cell mediated inflammatory disease is an autoimmune disease. In some embodiments, the T-cell mediated inflammatory disease is selected from the group consisting of psoriasis, psoriatic arthritis, rheumatoid arthritis, Crohn's disease, ankylosing spondylitis, type I diabetes, ulcerative colitis, multiple sclerosis, and graft versus host disease (GVHD). In some embodiments, the psoriasis is plaque psoriasis, chronic plaque psoriasis, guttate psoriasis, inverse psoriasis, pustular psoriasis, or erythrodermic psoriasis. In some embodiments, the transplantation is a transplantation of a tissue selected from the group consisting of bone marrow, kidney, heart, liver, neuronal tissue, lung, pancreas, skin, and intestine. In some embodiments, the transfusion is a transfusion comprising one or more of white blood cells, red blood cells, and platelets.
[0015] It is to be understood that one, some, or all of the properties of the various embodiments described herein may be combined to form other embodiments of the present invention. These and other aspects of the invention will become apparent to one of skill in the art.
BRIEF DESCRIPTION OF THE DRAWINGS
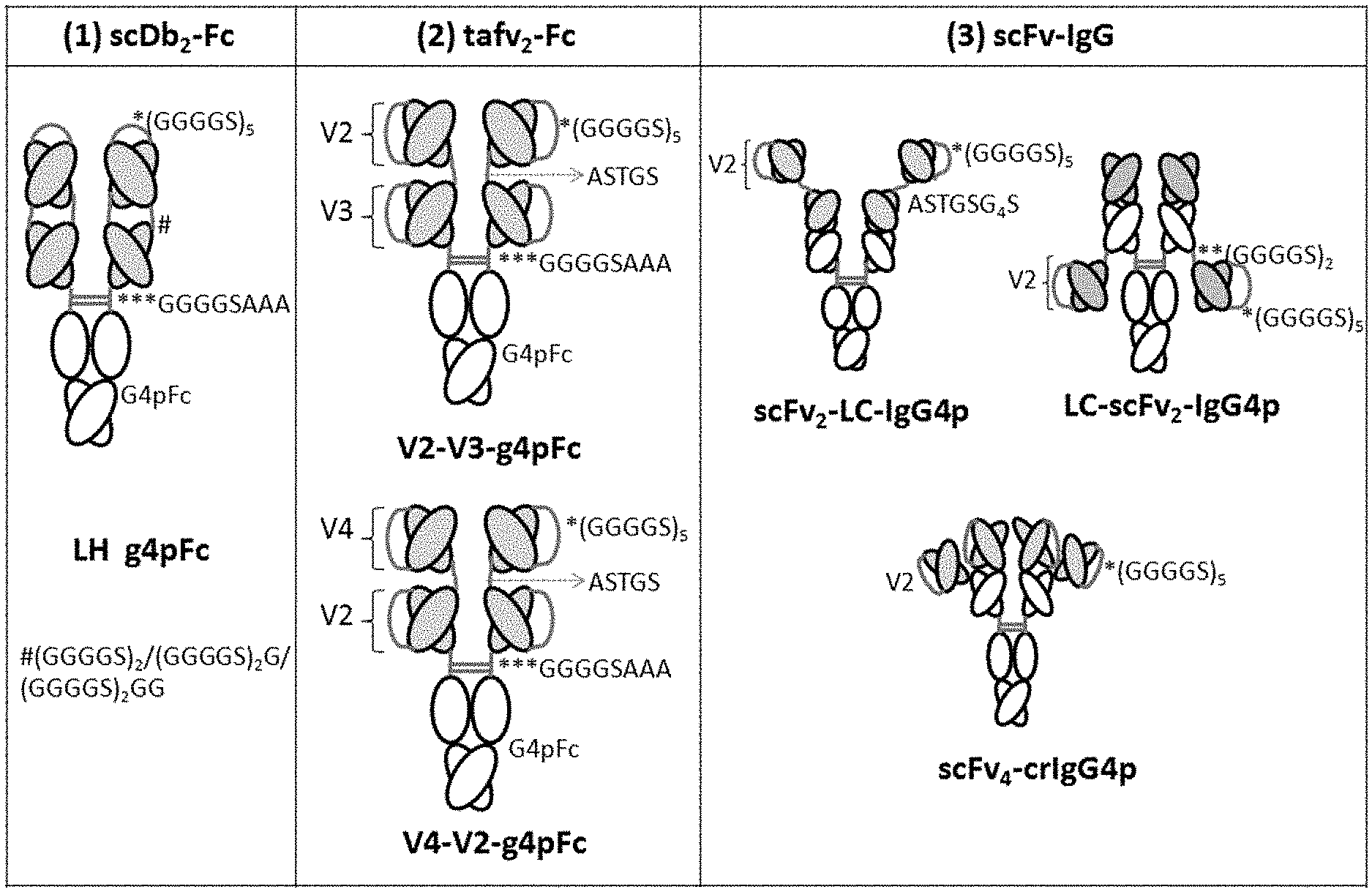
[0016] FIGS. 1A & 1B provide schematics illustrating exemplary tetravalent antibodies in accordance with some embodiments. FIG. 1A illustrates the following exemplary formats: (1) a dimer composed of two single-chain diabodies fused to an Fc domain (scDb.sub.2-Fc), showing linker sequences: (GGGGS).sub.5 (SEQ ID NO:33), GGGGSAAA (SEQ ID NO:26) and (GGGGS).sub.2 (SEQ ID NO:34)/(GGGGS).sub.2G (SEQ ID NO:35)/(GGGGS).sub.2GG (SEQ ID NO:36); (2) two different formats, each having a dimer of two tandem single-chain variable fragment units (taFv.sub.2-Fc), showing identical linker sequences for both formats: (GGGGS).sub.5 (SEQ ID NO:33), ASTGS (SEQ ID NO:27), GGGGSAAA (SEQ ID NO:26); and (3) three different formats based on single-chain variable fragments (scFv-IgG), showing: scFv.sub.2-LC-IgG4p linker sequences (GGGGS).sub.5 (SEQ ID NO:33) and ASTGSG.sub.4S (SEQ ID NO:28), LC-scFv.sub.2-IgG4p linker sequences (GGGGS).sub.2 (SEQ ID NO:34) and (GGGGS).sub.5 (SEQ ID NO:33), scFv.sub.4-crlG4p linker sequences (GGGGS).sub.5 (SEQ ID NO:33). FIG. 1B provides another illustration of the three scFv-based formats, with the variable fragments shaded and V2 scFvs indicated.
[0017] FIGS. 2A-2C show the verification of the molecular weights and basic structures of exemplary tetravalent antibodies by SDS-PAGE followed by Coomassie blue staining. Non-reducing (FIGS. 2A & 2B) and reducing (FIG. 2C) conditions are shown.
DETAILED DESCRIPTION
[0018] Provided herein are tetravalent antibodies that specifically bind to human PSGL-1. The present disclosure is based at least in part on the finding described herein that certain tetravalent anti-PSGL-1 antibodies show enhanced efficacy compared to the parental anti-PSGL-1 antibody both in vitro and trans vivo. These tetravalent antibodies displayed higher potency for apoptosis induction and enhanced efficacy in a trans vivo model for delayed type hypersensitivity (DTH) than the parental anti-PSGL-1 antibody. Further provided herein are isolated polynucleotides, vectors, host cells, pharmaceutical compositions, kits, uses, and methods related to the tetravalent antibodies. For example, the tetravalent antibodies of the present disclosure may find use in treating a T-cell mediated inflammatory disease, or administration before, concurrently with, and/or after a transfusion or transplantation.
[0019] In some embodiments, the tetravalent antibodies of the present disclosure comprise a dimer of two monomers, wherein each monomer of the dimer comprises a single-chain polypeptide comprising: (a) two light chain variable (VL) domains, wherein each of the two VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3; (b) two heavy chain variable (VH) domains, wherein each of the two VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3; and (c) an antibody Fc domain, wherein each of the two VL domains forms a VH-VL binding unit with a corresponding VH domain of the two VH domains, and wherein each of the two VH-VL binding units is specific for human PSGL-1. In other embodiments, the tetravalent antibodies of the present disclosure comprise a dimer of two monomers, wherein each monomer of the dimer comprises an antibody heavy chain and an antibody light chain; wherein the antibody light chain comprises: (i) two light chain variable (VL) domains, wherein each of the two VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3, (ii) a first heavy chain variable (VH) domain, and (iii) a light chain constant (CL) domain; wherein the antibody heavy chain comprises: (i) a second heavy chain variable (VH) domain, and (ii) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain; wherein the first and the second VH domains each comprise a CDR-H1, a CDR-H2, and a CDR-H3, wherein each of the two VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and the second VH domains, and wherein each of the two VH-VL binding units is specific for human PSGL-1. In other embodiments, the tetravalent antibodies of the present disclosure comprise a dimer of two monomers, wherein each monomer of the dimer comprises an antibody heavy chain and an antibody light chain; wherein the antibody light chain comprises: (i) a first heavy chain variable (VH) domain, (ii) a first light chain variable (VL) domain, and (iii) a light chain constant (CL) domain; wherein the antibody heavy chain comprises: (i) a second heavy chain variable (VH) domain, (ii) a second light chain variable (VL) domain, and (iii) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain; wherein each of the first and second VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3; wherein each of the first and second VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3; wherein each of the first and second VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and second VH domains; and wherein each of the two VH-VL binding units is specific for human PSGL-1.
I. Definitions
[0020] An "antibody" is an immunoglobulin molecule capable of specific binding to a target, such as a carbohydrate, polynucleotide, lipid, polypeptide, etc., through at least one antigen recognition site, located in the variable region of the immunoglobulin molecule. As used herein, the term encompasses not only intact polyclonal or monoclonal antibodies, but also polypeptides comprising fragments thereof (such as Fab, Fab', F(ab').sub.2, Fv); single-chain variable fragments (scFv), single-chain diabodies (scDbs), tandem single-chain variable fragment (scFv) units (termed taFv for tandem scFv), and mutants or other configurations thereof; fusion proteins comprising an antibody portion; and any other modified configuration of the immunoglobulin molecule that comprises an antigen recognition site.
[0021] As used herein, a "tetravalent" antibody may refer to an antibody that comprises four antibody VH-VL binding units, with each VH-VL binding unit comprising an antibody VH domain and an antibody VL domain. As used herein, references to a "monomer" of a tetravalent antibody may include both single-chain polypeptides and multiple-chain polypeptides. For example, a monomer may refer to a single-chain polypeptide, or it may refer to an antibody heavy chain-light chain unit, where the heavy chain and light chain are encoded by separate polynucleotides and/or are formed from the association of separate polypeptides.
[0022] An antibody includes an antibody of any class, such as IgG, IgA, or IgM (or sub-class thereof), and the antibody need not be of any particular class. Depending on the antibody amino acid sequence of the constant domain of its heavy chains, immunoglobulins can be assigned to different classes. There are five major classes of immunoglobulins: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into subclasses (isotypes), e.g., IgG1, IgG2, IgG3, IgG4, IgA1, and IgA2. The heavy chain constant domains that correspond to the different classes of immunoglobulins are called alpha, delta, epsilon, gamma, and mu, respectively. The subunit structures and three-dimensional configurations of different classes of immunoglobulins are well known.
[0023] The antibodies of the present disclosure are further intended to include bispecific, multispecific, chimeric, humanized, and recombinantly constructed molecules having affinity for a polypeptide conferred by at least one CDR region of the antibody. Single domain antibodies which are either the variable domain of an antibody heavy chain or the variable domain of an antibody light chain are known in the art. See, e.g., Holt et al., Trends Biotechnol. 21:484-490, 2003. Methods of making antibodies comprising either the variable domain of an antibody heavy chain or the variable domain of an antibody light chain, containing three of the six naturally occurring complementarity determining regions from an antibody, are also known in the art. See, e.g., Muyldermans, Rev. Mol. Biotechnol. 74:277-302, 2001.
[0024] As used herein, "monoclonal antibody" refers to an antibody of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical except for possible naturally-occurring mutations that may be present in minor amounts. Monoclonal antibodies are generally highly specific, being directed against a single antigenic site. Furthermore, in contrast to polyclonal antibody preparations, which typically include different antibodies directed against different determinants (epitopes), each monoclonal antibody is directed against a single determinant on the antigen. The modifier "monoclonal" indicates the character of the antibody as being obtained from a substantially homogeneous population of antibodies, and is not to be construed as requiring production of the antibody by any particular method. For example, the monoclonal antibodies to be used in accordance with the present disclosure may be made by the hybridoma method first described by Kohler and Milstein, 1975, Nature, 256:495, or may be made by recombinant DNA methods such as described in U.S. Pat. No. 4,816,567. The monoclonal antibodies may also be isolated from phage libraries generated using the techniques described in McCafferty et al., 1990, Nature, 348:552-554, for example.
[0025] As used herein, a "chimeric antibody" refers to an antibody having a variable region or part of a variable region from a first species and a constant region from a second species. An intact chimeric antibody comprises two copies of a chimeric light chain and two copies of a chimeric heavy chain. The production of chimeric antibodies is known in the art (Cabilly et al. (1984), Proc. Natl. Acad. Sci. USA, 81:3273-3277; Harlow and Lane (1988), Antibodies: a Laboratory Manual, Cold Spring Harbor Laboratory). Typically, in these chimeric antibodies, the variable region of both light and heavy chains mimics the variable regions of antibodies derived from one species of mammal, while the constant portions are homologous to the sequences in antibodies derived from another. One clear advantage to such chimeric forms is that, for example, the variable regions can conveniently be derived from presently known sources using readily available hybridomas or B-cells from non-human host organisms in combination with constant regions derived from, for example, human cell preparations. While the variable region has the advantage of ease of preparation, and the specificity is not affected by its source, the constant region being human is less likely to elicit an immune response from a human subject when the antibodies are injected than would the constant region from a non-human source. However, the definition is not limited to this particular example. In some embodiments, amino acid modifications are made in the variable and/or constant region.
[0026] As used herein, "humanized" antibodies refer to forms of non-human (e.g., murine) antibodies that are specific chimeric immunoglobulins, immunoglobulin chains, or fragments thereof (such as Fv, Fab, Fab', F(ab').sub.2, or other antigen-binding subsequences of antibodies) that contain minimal sequence derived from non-human immunoglobulin. For the most part, humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a complementary determining region (CDR) of the recipient are replaced by residues from a CDR of a non-human species (donor antibody) such as mouse, rat, or rabbit having the desired specificity, affinity, and capacity. In some instances, Fv framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues. Furthermore, the humanized antibody may comprise residues that are found neither in the recipient antibody nor in the imported CDR or framework sequences, but are included to further refine and optimize antibody performance. In general, the humanized antibody will comprise substantially all of at least one, and typically two, variable domains in which all or substantially all of the CDR regions correspond to those of a non-human immunoglobulin and all or substantially all of the FR regions are those of a human immunoglobulin consensus sequence. The humanized antibody optimally also will comprise at least a portion of an immunoglobulin constant region or domain (e.g., an Fc domain), typically that of a human immunoglobulin. Antibodies may have Fc regions modified as described in WO 99/58572. Other forms of humanized antibodies have one or more CDRs (one, two, three, four, five, or six) which are altered with respect to the original antibody, which are also termed one or more CDRs "derived from" one or more CDRs from the original antibody.
[0027] As used herein, "human antibody" means an antibody having an amino acid sequence corresponding to that of an antibody produced by a human and/or has been made using any of the techniques for making human antibodies known in the art or disclosed herein. This definition of a human antibody includes antibodies comprising at least one human heavy chain polypeptide or at least one human light chain polypeptide. One such example is an antibody comprising murine light chain and human heavy chain polypeptides. Human antibodies can be produced using various techniques known in the art. In one embodiment, the human antibody is selected from a phage library, where that phage library expresses human antibodies (Vaughan et al., 1996, Nature Biotechnology, 14:309-314; Sheets et al., 1998, PNAS, (USA) 95:6157-6162; Hoogenboom and Winter, 1991, J. Mol. Biol., 227:381; Marks et al., 1991, J. Mol. Biol., 222:581). Human antibodies can also be made by introducing human immunoglobulin loci into transgenic animals, e.g., mice in which the endogenous immunoglobulin genes have been partially or completely inactivated. This approach is described in U.S. Pat. Nos. 5,545,807; 5,545,806; 5,569,825; 5,625,126; 5,633,425; and 5,661,016. Alternatively, the human antibody may be prepared by immortalizing human B-lymphocytes that produce an antibody directed against a target antigen (such B-lymphocytes may be recovered from an individual or may have been immunized in vitro). See, e.g., Cole et al., Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, p. 77 (1985); Boerner et al., 1991, J. Immunol., 147 (1):86-95; and U.S. Pat. No. 5,750,373.
[0028] A "variable region" (the term "variable domain" may be used interchangeably herein) of an antibody refers to the variable region of the antibody light chain (VL) or the variable region of the antibody heavy chain (VH), either alone or in combination. The variable regions of the heavy and light chain (VH and VL domains, respectively) each consist of four framework regions (FR) connected by three complementarity determining regions (CDRs) also known as hypervariable regions. The CDRs in each chain are held together in close proximity by the FRs and, with the CDRs from the other chain, contribute to the formation of the antigen-binding site of antibodies. There are at least two techniques for determining CDRs: (1) an approach based on cross-species sequence variability (i.e., Kabat et al. Sequences of Proteins of Immunological Interest, (5th ed., 1991, National Institutes of Health, Bethesda Md.)); and (2) an approach based on crystallographic studies of antigen-antibody complexes (Al-lazikani et al (1997) J. Molec. Biol. 273:927-948)). As used herein, a CDR may refer to CDRs defined by either approach or by a combination of both approaches.
[0029] A number of HVR delineations are in use and are encompassed herein. The Kabat Complementarity Determining Regions (CDRs) are based on sequence variability and are the most commonly used (Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991)). Chothia refers instead to the location of the structural loops (Chothia and Lesk, J. Mol. Biol. 196:901-917 (1987)). The AbM HVRs represent a compromise between the Kabat HVRs and Chothia structural loops, and are used by Oxford Molecular's AbM antibody modeling software. The "contact" HVRs are based on an analysis of the available complex crystal structures. The residues from each of these HVRs are noted below.
TABLE-US-00001 Loop Kabat AbM Chothia Contact L1 L24-L34 L24-L34 L26-L32 L30-L36 L2 L50-L56 L50-L56 L50-L52 L46-L55 L3 L89-L97 L89-L97 L91-L96 L89-L96 H1 H31-H35B H26-H35B H26-H32 H30-H35B (Kabat numbering) H1 H31-H35 H26-H35 H26-H32 H30-H35 (Chothia numbering) H2 H50-H65 H50-H58 H53-H55 H47-H58 H3 H95-H102 H95-H102 H96-H101 H93-H101
[0030] The Kabat numbering system is generally used when referring to a residue in the variable domain (approximately residues 1-107 of the light chain and residues 1-113 of the heavy chain) (e.g., Kabat et al., Sequences of Immunological Interest. 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991)). The "EU numbering system" or "EU index" is generally used when referring to a residue in an immunoglobulin heavy chain constant region (e.g., the EU index reported in Kabat et al., supra, or and Edelman, G. M. et al. (1969) Proc. Natl. Acad. Sci. USA 63:78-85).
[0031] "Fv" as used herein may refer to the minimum antibody fragment which contains a complete antigen-recognition and -binding site. This fragment typically consists of a dimer of one heavy- and one light-chain variable region domain in tight, non-covalent association. From the folding of these two domains emanate six hypervariable loops (3 loops each from the H and L chain) that contribute the amino acid residues for antigen binding and confer antigen binding specificity to the antibody. However, even a single variable domain (or half of an Fv comprising only three HVRs specific for an antigen) has the ability to recognize and bind antigen, although at a lower affinity than the entire binding site.
[0032] A "constant region" (the term "constant domain" may be used interchangeably herein) of an antibody refers to the constant region of the antibody light chain (CL) or the constant region of the antibody heavy chain (CH), either alone or in combination. A constant region of an antibody generally provides structural stability and other biological functions such as antibody chain association, secretion, transplacental mobility, and complement binding, but is not involved with binding to the antigen. The amino acid sequence and corresponding exon sequences in the genes of the constant region is dependent upon the species from which it is derived; however, variations in the amino acid sequence leading to allotypes is relatively limited for particular constant regions within a species. The variable region of each chain is joined to the constant region by a linking polypeptide sequence. The linkage sequence is coded by a "J" sequence in the light chain gene, and a combination of a "D" sequence and a "J" sequence in the heavy chain gene. Depending on the antibody isotype, a heavy chain constant region may include a CH1 domain, a hinge region, a CH2 domain, a CH3 domain, and/or a CH4 domain. In certain embodiments, a heavy chain constant region comprises a CH1 domain, a hinge region, a CH2 domain, and a CH3 domain.
[0033] The term "Fc region" (the term "Fc domain" may be used interchangeably herein) herein is used to define a C-terminal region of an immunoglobulin heavy chain, including native-sequence Fc regions and variant Fc regions. The boundaries of the Fc region of an immunoglobulin heavy chain might vary; in some embodiments, the Fc region may include one or more amino acids of the hinge region. In some embodiments, the human IgG heavy-chain Fc region is defined to stretch from an amino acid residue at EU position 216 to the carboxyl-terminus thereof. Suitable native-sequence Fc regions for use in the antibodies of the present disclosure include human IgG1, IgG2 (IgG2A, IgG2B), IgG3 and IgG4.
[0034] "Single-chain Fv" also abbreviated as "sFv" or "scFv" are antibody fragments that comprise the V.sub.H and V.sub.L antibody domains connected into a single polypeptide chain. Preferably, the sFv polypeptide further comprises a polypeptide linker between the V.sub.H and V.sub.L domains which enables the sFv to form the desired structure for antigen binding. For a review of the sFv, see Pluckthun in The Pharmacology of Monoclonal Antibodies, vol. 113, Rosenburg and Moore eds., Springer-Verlag, New York, pp. 269-315 (1994).
[0035] The term "diabodies" refers to antibody fragments prepared by constructing sFv fragments (see preceding paragraph) with short linkers (e.g., about 5-12 residues) between the V.sub.H and V.sub.L domains such that inter-chain but not intra-chain pairing of the V domains is achieved, thereby resulting in a bivalent fragment, i.e., a fragment having two antigen-binding sites. Bispecific diabodies are heterodimers of two "crossover" sFv fragments in which the V.sub.H and V.sub.L domains of the two antibodies are present on different polypeptide chains. Diabodies are described in greater detail in, for example, EP 404,097; WO 93/11161; Hollinger et al., Proc. Natl. Acad. Sci. USA 90: 6444-6448 (1993).
[0036] "Percent (%) amino acid sequence identity" with respect to a reference polypeptide sequence is defined as the percentage of amino acid residues in a candidate sequence that are identical with the amino acid residues in the reference polypeptide sequence, after aligning the sequences and introducing gaps, if necessary, to achieve the maximum percent sequence identity, and not considering any conservative substitutions as part of the sequence identity. Alignment for purposes of determining percent amino acid sequence identity can be achieved in various ways that are within the skill in the art, for instance, using publicly available computer software such as BLAST, BLAST-2, ALIGN or Megalign (DNASTAR) software. Those skilled in the art can determine appropriate parameters for aligning sequences, including any algorithms needed to achieve maximal alignment over the full length of the sequences being compared.
[0037] As used herein, "antibody-dependent cell-mediated cytotoxicity" and "ADCC" refer to a cell-mediated reaction in which nonspecific cytotoxic cells that express Fc receptors (FcRs) (e.g., natural killer (NK) cells, neutrophils, or macrophages) recognize bound antibody on a target cell and subsequently cause lysis of the target cell. ADCC activity of a molecule of interest can be assessed using an in vitro ADCC assay, such as that described in U.S. Pat. No. 5,500,362 or 5,821,337. Useful effector cells for such assays include peripheral blood mononuclear cells (PBMC) and NK cells. Alternatively, or additionally, ADCC activity of the molecule of interest may be assessed in vivo, e.g., in a animal model such as that disclosed in Clynes et al., 1998, PNAS (USA), 95:652-656.
[0038] "Complement dependent cytotoxicity" and "CDC" refer to the lysing of a target in the presence of complement. The complement activation pathway is initiated by the binding of the first component of the complement system (C1q) to a molecule (e.g., an antibody) complexed with a cognate antigen. To assess complement activation, a CDC assay, e.g., as described in Gazzano-Santoro et al., J. Immunol. Methods, 202:163 (1996), may be performed.
[0039] The terms "polypeptide," "oligopeptide," "peptide," and "protein" are used interchangeably herein to refer to polymers of amino acids of any length. The polymer may be linear or branched, it may comprise modified amino acids, and it may be interrupted by non-amino acids. The terms also encompass an amino acid polymer that has been modified naturally or by intervention; for example, disulfide bond formation, glycosylation, lipidation, acetylation, phosphorylation, or any other manipulation or modification, such as conjugation with a labeling component. Also included within the definition are, for example, polypeptides containing one or more analogs of an amino acid (including, for example, unnatural amino acids, etc.), as well as other modifications known in the art. It is understood that, because the polypeptides of the present disclosure are based upon a tetravalent antibody, the polypeptides can occur as single chains or associated chains.
[0040] "Polynucleotide," or "nucleic acid," as used interchangeably herein, refer to polymers of nucleotides of any length, and include DNA and/or RNA. The nucleotides can be deoxyribonucleotides, ribonucleotides, modified nucleotides or bases, and/or their analogs, or any substrate that can be incorporated into a polymer by DNA or RNA polymerase. A polynucleotide may comprise modified nucleotides, such as methylated nucleotides and their analogs. If present, modification to the nucleotide structure may be imparted before or after assembly of the polymer. The sequence of nucleotides may be interrupted by non-nucleotide components. A polynucleotide may be further modified after polymerization, such as by conjugation with a labeling component. Other types of modifications include, for example, "caps," substitution of one or more of the naturally occurring nucleotides with an analog, internucleotide modifications such as, for example, those with uncharged linkages (e.g., methyl phosphonates, phosphotriesters, phosphoamidates, cabamates, etc.) and with charged linkages (e.g., phosphorothioates, phosphorodithioates, etc.), those containing pendant moieties, such as, for example, proteins (e.g., nucleases, toxins, antibodies, signal peptides, ply-L-lysine, etc.), those with intercalators (e.g., acridine, psoralen, etc.), those containing chelators (e.g., metals, radioactive metals, boron, oxidative metals, etc.), those containing alkylators, those with modified linkages (e.g., alpha anomeric nucleic acids, etc.), as well as unmodified forms of the polynucleotide(s). Further, any of the hydroxyl groups ordinarily present in the sugars may be replaced, for example, by phosphonate groups, phosphate groups, protected by standard protecting groups, or activated to prepare additional linkages to additional nucleotides, or may be conjugated to solid supports. The 5' and 3' terminal OH can be phosphorylated or substituted with amines or organic capping group moieties of from 1 to 20 carbon atoms. Other hydroxyls may also be derivatized to standard protecting groups. Polynucleotides can also contain analogous forms of ribose or deoxyribose sugars that are generally known in the art, including, for example, 2'-O-methyl-, 2'-O-allyl, 2'-fluoro- or 2'-azido-ribose, carbocyclic sugar analogs, .alpha.-anomeric sugars, epimeric sugars such as arabinose, xyloses, lyxoses, pyranose sugars, furanose sugars, sedoheptuloses, acyclic analogs, and abasic nucleoside analogs such as methyl ribosides. One or more phosphodiester linkages may be replaced by alternative linking groups. These alternative linking groups include, but are not limited to, embodiments wherein phosphate is replaced by P(O)S("thioate"), P(S)S ("dithioate"), "(O)NR.sub.2 ("amidate"), P(O)R, P(O)OR', CO, or CH.sub.2 ("formacetal"), in which each R or R' is independently H or substituted or unsubstituted alkyl (1-20 C) optionally containing an ether (--O--) linkage, aryl, alkenyl, cycloalkyl, cycloalkenyl, or araldyl. Not all linkages in a polynucleotide need be identical. The preceding description applies to all polynucleotides referred to herein, including RNA and DNA.
[0041] As used herein, "vector" means a construct that is capable of delivering and desirably expressing one or more gene(s) or sequence(s) of interest in a host cell. Examples of vectors include, but are not limited to, viral vectors, naked DNA or RNA expression vectors, plasmid, cosmid or phage vectors, DNA or RNA expression vectors associated with cationic condensing agents, DNA or RNA expression vectors encapsulated in liposomes, and certain eukaryotic cells, such as producer cells.
[0042] As used herein, "expression control sequence" means a nucleic acid sequence that directs transcription of a nucleic acid. An expression control sequence can be a promoter, such as a constitutive or an inducible promoter, or an enhancer. The expression control sequence is operably linked to the nucleic acid sequence to be transcribed.
[0043] As used herein, an "effective dosage" or "therapeutically effective amount" of drug, compound, or pharmaceutical composition is an amount sufficient to effect beneficial, desired, and/or therapeutic results. For prophylactic use, beneficial or desired results include results such as eliminating or reducing the risk, lessening the severity, or delaying the onset of the disease, including biochemical, histological and/or behavioral symptoms of the disease, its complications and intermediate pathological phenotypes presenting during development of the disease. For therapeutic use, beneficial or desired results include clinical results such as decreasing one or more symptoms resulting from the disease, increasing the quality of life of those suffering from the disease, decreasing the dose of other medications required to treat the disease, enhancing effect of another medication such as via targeting, delaying the progression of the disease, and/or prolonging survival. In the case of treating an individual awaiting a transplantation, for example, an effective amount of the drug may reduce to some extent the level of alloantibodies and/or PRA in the individual. In the case of treating an individual receiving a transplantation or transfusion, an effective amount of the drug may have the effect in and/or relieving to some extent one or more of the symptoms or conditions (such as graft rejection) associated with the transplantation or transfusion. An effective amount can be administered in one or more administrations. For purposes of the present disclosure, an effective amount of drug, compound, or pharmaceutical composition is an amount sufficient to accomplish prophylactic or therapeutic treatment either directly or indirectly. An effective dosage can be administered in one or more administrations. For purposes of the present disclosure, an effective dosage of drug, compound, or pharmaceutical composition is an amount sufficient to accomplish prophylactic or therapeutic treatment either directly or indirectly. As is understood in the clinical context, an effective dosage of a drug, compound, or pharmaceutical composition may or may not be achieved in conjunction with another drug, compound, or pharmaceutical composition. Thus, an "effective dosage" may be considered in the context of administering one or more therapeutic agents, and a single agent may be considered to be given in an effective amount if, in conjunction with one or more other agents, a desirable result may be or is achieved.
[0044] As used herein, "in conjunction with" refers to administration of one treatment modality in addition to another treatment modality. As such, "in conjunction with" refers to administration of one treatment modality before, during, or after administration of the other treatment modality to the individual.
[0045] As used herein, "treatment" or "treating" is an approach for obtaining beneficial or desired results, including desirably clinical results. Beneficial, desired, and/or therapeutic clinical results include, but are not limited to, one or more of the following: reducing or abrogating one or more symptoms of inflammation or autoimmunity (e.g., stemming from a T-cell mediated inflammatory disease), increasing the likelihood of a successful patient outcome and/or mitigating one or more contraindications or detrimental outcomes related to a medical treatment (e.g., related to a transplantation or transfusion), decreasing symptoms resulting from the disease, increasing the quality of life of those suffering from the disease, decreasing the dose of other medications required to treat the disease, delaying the progression of the disease, and/or prolonging survival of individuals.
[0046] As used herein, "delaying development of a disease" means to defer, hinder, slow, retard, stabilize, and/or postpone development of the disease (such as cancer). This delay can be of varying lengths of time, depending on the history of the disease and/or individual being treated. As is evident to one skilled in the art, a sufficient or significant delay can, in effect, encompass prevention, in that the individual does not develop the disease. For example, a symptom of an inflammatory disease, such as a T-cell mediated inflammatory disease, may be delayed.
[0047] An "individual" or a "subject" is a mammal, more desirably a human. Mammals also include, but are not limited to, farm animals, sport animals, pets (such as cats, dogs, or horses), primates, mice, and rats.
[0048] As used herein, the term "specifically recognizes" or "specifically binds" refers to measurable and reproducible interactions such as attraction or binding between a target and an antibody (e.g., a full-length antibody, an antibody fragment, or an antibody VH-VL binding unit) that is determinative of the presence of the target in the presence of a heterogeneous population of molecules including biological molecules. For example, an antibody, antibody fragment, or antibody VH-VL binding unit that specifically or preferentially binds to an epitope is an antibody that binds this epitope with greater affinity, avidity, more readily, and/or with greater duration than it binds to other epitopes of the target or non-target epitopes. It is also understood by reading this definition that, for example, an antibody, antibody fragment, or antibody VH-VL binding unit that specifically or preferentially binds to a first target may or may not specifically or preferentially bind to a second target. As such, "specific binding" or "preferential binding" does not necessarily require (although it can include) exclusive binding. An antibody, antibody fragment, or antibody VH-VL binding unit that specifically binds to a target may have an association constant of greater than or about 10.sup.3M.sup.-1 or about 10.sup.4M.sup.-1, sometimes about 10.sup.5 M.sup.-1 or about 10.sup.6M.sup.-1, in other instances about 10.sup.6M.sup.-1 or about 10.sup.7M.sup.-1, about 10.sup.8M.sup.-1 to about 10.sup.9M.sup.-1, or about 10.sup.10 M.sup.-1 to about 10.sup.11 M.sup.-1 or higher. A variety of immunoassay formats can be used to select antibodies, antibody fragments, or antibody VH-VL binding units that are specifically immunoreactive with a particular protein. For example, solid-phase ELISA immunoassays are routinely used to select monoclonal antibodies specifically immunoreactive with a protein. See, e.g., Harlow and Lane (1988) Antibodies, A Laboratory Manual, Cold Spring Harbor Publications, New York, for a description of immunoassay formats and conditions that can be used to determine specific immunoreactivity.
[0049] A "package insert" refers to instructions customarily included in commercial packages of medicaments that contain information about the indications customarily included in commercial packages of medicaments that contain information about the indications, usage, dosage, administration, contraindications, other medicaments to be combined with the packaged product, and/or warnings concerning the use of such medicaments, etc.
[0050] As used herein and in the appended claims, the singular forms "a," "an," and "the" include plural reference unless the context clearly indicates otherwise. For example, reference to an "antibody" is a reference to from one to many antibodies, such as molar amounts, and includes equivalents thereof known to those skilled in the art, and so forth.
[0051] Reference to "about" a value or parameter herein includes (and describes) embodiments that are directed to that value or parameter per se. For example, description referring to "about X" includes description of "X."
[0052] It is understood that aspect and variations of the present disclosure described herein include "consisting" and/or "consisting essentially of" aspects and variations.
II. Tetravalent Antibodies
[0053] Certain aspects of the present disclosure relate to tetravalent antibodies that specifically bind to human PSGL-1. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers. As described infra, the monomers may be coupled using any means known in the art, including without limitation wild-type interactions between antibody Fc domains or regions, altered or mutated interactions between antibody Fc domains or regions (e.g., using a hinge region mutation described herein), or other artificial covalent or non-covalent interactions (e.g., cross-linking or a linker). Exemplary tetravalent antibodies and antibody formats are described below and illustrated in FIGS. 1A & 1B.
[0054] Human PSGL-1 may also be referred to as selectin P ligand, SELPG, CLA, CD162, or PSGL1. In some embodiments, a tetravalent antibody of the present disclosure binds to a polypeptide encoded by the human SELPG gene, e.g., as described by NCBI RefSeq Gene ID No. 6404. In some embodiments, a tetravalent antibody of the present disclosure binds to a human PSGL-1 polypeptide containing 15 or 16 decamer repeats. In some embodiments, a tetravalent antibody of the present disclosure binds to a polypeptide comprising the amino acid sequence of SEQ ID NO:31. In some embodiments, a tetravalent antibody of the present disclosure binds to a polypeptide comprising the amino acid sequence of SEQ ID NO:32. In some embodiments, a tetravalent antibody of the present disclosure binds to a polypeptide comprising the amino acid sequence of SEQ ID NO:31 and binds to a polypeptide comprising the amino acid sequence of SEQ ID NO:32. The amino acid sequence of SEQ ID NO:31 depicts full length human PSGL-1, GenBank.TM. accession number AAA74577.1, GL902797, and the amino acid sequence of SEQ ID NO:32 depicts the shorter 402 amino acid human PSGL-1 protein (GenBank.TM. accession number XP_005269133). In specific embodiments, a tetravalent antibody described herein specifically binds to human PSGL-1 as determined, e.g., by ELISA or other antigen-binding assay known in the art, or described herein.
[0055] In some embodiments, a VH domain and a VL domain of the present disclosure form a VH-VL binding unit (e.g., that specifically binds an epitope, such as an epitope of human PSGL-1). As described herein, a VH-VL binding unit may be formed between a VH domain and a VL domain using wild-type VH-VL interactions, or a VH-VL binding unit may be further stabilized using one or more mutations or chemical bonds (e.g., a disulfide bond, such as the vH44-vL100 disulfide bond introduced by cysteine substitutions in the VH and VL domain of SEQ ID NOs: 29 and 30, respectively).
[0056] In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, where each monomer of the dimer comprises a single-chain polypeptide.
[0057] In some embodiments, a single-chain, heavy chain, and/or light chain polypeptide of the present disclosure comprises a linker sequence. A variety of linker sequences may suitably be used, e.g., to link VH and VL domains of a VH-VL binding unit, to link a VH or VL domain of a VH-VL binding unit to a VH or VL domain of another VH-VL binding unit, or to link a VH or VL domain of a VH-VL binding unit to an antibody constant region, such as an Fc domain or region. In some embodiments, a linker of the present disclosure may be present between domains or regions. In some embodiments, two domains or regions of the present disclosure may be joined without a linker, or the linker joining two domains or regions may be removed. Coupling of such single-chain fragments using various linkers is described in Kortt et al., 1997, Protein Engineering, 10:423-433. In some embodiments, a linker sequence of the present disclosure comprises 1-50 amino acids. In certain embodiments, a linker sequence of the present disclosure comprises 5-12 amino acids. Exemplary linker sequences are described herein and illustrated in FIG. 1A. In some embodiments, a linker sequence of the present disclosure comprises one or more repeats of the amino acid sequence of GGGGS (SEQ ID NO:25). In some embodiments, a linker sequence of the present disclosure comprises two, three, four, or five repeats of the amino acid sequence of GGGGS (SEQ ID NO:25). In some embodiments, a linker sequence of the present disclosure comprises the amino acid sequence of SEQ ID NO:33, 34, 35, or 36. In some embodiments, a linker sequence of the present disclosure comprises the amino acid sequence of GGGGSAAA (SEQ ID NO:26). In some embodiments, a linker sequence of the present disclosure comprises the amino acid sequence of ASTGS (SEQ ID NO:27). In some embodiments, a linker sequence of the present disclosure comprises the amino acid sequence of ASTGSGGGGS (SEQ ID NO:28).
[0058] In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two single-chain diabodies (scDbs), which may optionally be fused to an antibody constant region, such as an Fc domain.
[0059] In some embodiments, each monomer of the dimer comprises a single-chain polypeptide comprising (a) two light chain variable (VL) domains, wherein each of the two VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3, and wherein the two VL domains are specific for human PSGL-1; (b) two heavy chain variable (VH) domains, wherein each of the two VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3, and wherein the two VH domains are specific for human PSGL-1; and (c) an antibody Fc domain. In some embodiments, each of the two VL domains forms a VH-VL binding unit with a corresponding VH domain of the two VH domains.
[0060] In certain embodiments, each of the two single-chain polypeptides comprises, from N-terminus to C-terminus: (a) a first VL domain of two VL domains; (b) a first linker sequence; (c) a first VH domain of two VH domains; (d) a second linker sequence; (e) a second VL domain of two VL domains; (f) a third linker sequence; (g) a second VH domain of two VH domains; (h) a fourth linker sequence; and (i) an antibody Fc domain. In some embodiments, the first VL domain forms a VH-VL binding unit with the second VH domain, and the first VH domain forms a VH-VL binding unit with the second VL domain.
[0061] In some embodiments, the first, second and third linker sequences each comprise two or more repeats of the amino acid sequence of SEQ ID NO:25. In some embodiments, the first, second or third linker sequence comprises the amino acid sequence of SEQ ID NO:33, 34, 35, or 36. In some embodiments, the first and the third linker sequences have the same sequence and comprise two repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises five repeats of SEQ ID NO:25. In some embodiments, the fourth linker sequence comprises the amino acid sequence of SEQ ID NO:26.
[0062] In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two tandem single-chain variable fragment (scFv) units (termed taFv for tandem scFv), which may optionally be fused to an antibody constant domain, such as an Fc domain of a heavy chain constant domain.
[0063] In certain embodiments, each of the two single-chain polypeptides comprises, from N-terminus to C-terminus: (a) a first VH domain of the two VH domains; (b) a first linker sequence; (c) a first VL domain of the two VL domains; (d) a second linker sequence; (e) a second VL domain of the two VL domains; (f) a third linker sequence; (g) a second VH domain of the two VH domains; (h) a fourth linker sequence; and (i) an antibody Fc domain. In some embodiments, the first VL domain forms a VH-VL binding unit with the first VH domain, and the second VH domain forms a VH-VL binding unit with the second VL domain. In other embodiments, each of the two single-chain polypeptides comprises, from N-terminus to C-terminus: (a) a first VL domain of the two VL domains; (b) a first linker sequence; (c) a first VH domain of the two VH domains; (d) a second linker sequence; (e) a second VH domain of the two VH domains; (f) a third linker sequence; (g) a second VL domain of the two VL domains; (h) a fourth linker sequence; and (i) the heavy chain constant domain comprising an antibody Fc domain.
[0064] In some embodiments, the first and the third linker sequences have the same sequence comprising five repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises the amino acid sequence of SEQ ID NO:27. In some embodiments, the fourth linker sequence comprises the amino acid sequence of SEQ ID NO:26.
[0065] In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, where each monomer of the dimer comprises an antibody heavy chain and an antibody light chain.
[0066] In some embodiments, a tetravalent antibody of the present disclosure comprises a light chain comprising (i) two light chain variable (VL) domains, wherein each of the two VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3, and wherein the two VL domains are specific for human PSGL-1, (ii) a first heavy chain variable (VH) domain, and (iii) a light chain constant (CL) domain; and/or a heavy chain comprising (i) a second heavy chain variable (VH) domain, and (ii) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain. In some embodiments, the first and the second VH domains each comprise a CDR-H1, a CDR-H2, and a CDR-H3. In some embodiments, the first and the second VH domains are specific for human PSGL-1. In some embodiments, each of the two VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and the second VH domains.
[0067] In certain embodiments, the antibody light chain comprises, from N-terminus to C-terminus: (a) the first VH domain; (b) a first linker sequence; (c) a first VL domain of the two or more VL domains; (d) a second linker sequence; (e) a second VL domain of the two or more VL domains; and (f) the CL domain. In some embodiments, the CL domain is a kappa CL domain. In other embodiments, the CL domain is a lambda CL domain. In some embodiments, the first VL domain forms a VH-VL binding unit with the first VH domain, and the second VH domain forms a VH-VL binding unit with the second VL domain.
[0068] In some embodiments, the first linker sequence comprises five repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises the amino acid sequence of SEQ ID NO:28.
[0069] In some embodiments, a tetravalent antibody of the present disclosure comprises a light chain comprising, from N-terminus to C-terminus: (a) a first VL domain of the two VL domains; (b) the CL domain; (c) a first linker sequence; (d) the first VH domain; (e) a second linker sequence; and (f) a second VL domain of the two VL domains. In some embodiments, the CL domain is a kappa CL domain. In other embodiments, the CL domain is a lambda CL domain.
[0070] In some embodiments, the first linker sequence comprises two repeats of SEQ ID NO:25. In some embodiments, the second linker sequence comprises five repeats of SEQ ID NO:25.
[0071] In some embodiments, a tetravalent antibody of the present disclosure comprises a heavy chain comprising, from N-terminus to C-terminus: (a) the second of two VH domains; and (b) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain. In some embodiments, the antibody Fc domain comprises a heavy chain constant 2 (CH2) domain and a heavy chain constant 3 (CH3) domain. In some embodiments, the first VL domain forms a VH-VL binding unit with the first VH domain, and the second VH domain forms a VH-VL binding unit with the second VL domain.
[0072] In some embodiments, a tetravalent antibody of the present disclosure comprises a light chain comprising (i) a first heavy chain variable (VH) domain, (ii) a first light chain variable (VL) domain, and (iii) a light chain constant (CL) domain; and/or a heavy chain comprising (i) a second heavy chain variable (VH) domain, (ii) a second light chain variable (VL) domain, and (iii) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain. In some embodiments, each of the first and second VL domains comprises a CDR-L1, a CDR-L2, and a CDR-L3. In some embodiments, the first and second VL domains are specific for human PSGL-1. In some embodiments, each of the first and second VH domains comprises a CDR-H1, a CDR-H2, and a CDR-H3. In some embodiments, the first and second VH domains are specific for human PSGL-1. In some embodiments, each of the first and second VL domains forms a VH-VL binding unit with a corresponding VH domain of the first and second VH domains.
[0073] In some embodiments, the antibody light chain comprises, from N-terminus to C-terminus: (a) the first VH domain; (b) a first linker sequence; (c) the first VL domain; and (d) the CL domain. In some embodiments, the CL domain is a kappa CL domain. In other embodiments, the CL domain is a lambda CL domain.
[0074] In some embodiments, the first linker sequence comprises five repeats of SEQ ID NO:25.
[0075] In some embodiments, the antibody heavy chain comprises, from N-terminus to C-terminus: (a) the second of two VH domains; (b) a second linker sequence; (c) the second of two VL domains; and (d) a heavy chain constant region comprising a first heavy chain constant region (CH1) domain, an antibody hinge region, an second heavy chain constant region (CH2) domain, and a third heavy chain constant region (CH3) domain. In some embodiments, the antibody Fc domain comprises a heavy chain constant 2 (CH2) domain and a heavy chain constant 3 (CH3) domain.
[0076] In some embodiments, the second linker sequence comprises five repeats of SEQ ID NO:25.
[0077] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VH domains comprising one or more CDRs selected from (i) a CDR-H1 comprising the amino acid sequence of SFGMH (SEQ ID NO:17); (ii) a CDR-H2 comprising the amino acid sequence of YINGGSSTIFYANAVKG (SEQ ID NO:18); and (iii) a CDR-H3 comprising the amino acid sequence of YASYGGGAMDY (SEQ ID NO:19). In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VH domains comprising (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19. In some embodiments, a tetravalent antibody of the present disclosure is a dimer of two monomers, each monomer comprising two VH domains, each VH domain comprising one or more CDRs selected from (i) a CDR-H1 comprising the amino acid sequence of SFGMH (SEQ ID NO:17); (ii) a CDR-H2 comprising the amino acid sequence of YINGGSSTIFYANAVKG (SEQ ID NO:18); and (iii) a CDR-H3 comprising the amino acid sequence of YASYGGGAMDY (SEQ ID NO:19). In some embodiments, a tetravalent antibody of the present disclosure is a dimer of two monomers, each monomer comprising two VH domains, each VH domain comprising (i) a CDR-H1 comprising the amino acid sequence of SEQ ID NO:17; (ii) a CDR-H2 comprising the amino acid sequence of SEQ ID NO:18; and (iii) a CDR-H3 comprising the amino acid sequence of SEQ ID NO:19.
[0078] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VH domains comprising the amino acid sequence of SEQ ID NO:23. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VH domains, each VH domain comprising the amino acid sequence of SEQ ID NO:23. In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VH domains comprising the amino acid sequence of SEQ ID NO:29. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VH domains, each VH domain comprising the amino acid sequence of SEQ ID NO:29.
[0079] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VH domains comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:23. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VH domains, each VH domain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:23. In some embodiments, the VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:23.
[0080] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VH domains comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:29. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VH domains, each VH domain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:29. In some embodiments, the VH sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:29.
[0081] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VL domains comprising one or more CDRs selected from (i) a CDR-L1 comprising the amino acid sequence of RSSQSIVHNDGNTYFE (SEQ ID NO:20); (ii) a CDR-L2 comprising the amino acid sequence of KVSNRFS (SEQ ID NO:21); and (iii) a CDR-L3 comprising the amino acid sequence of FQGSYVPLT (SEQ ID NO:22). In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VL domains comprising (i) a CDR-L1 comprising the amino acid sequence of RSSQSIVHNDGNTYFE (SEQ ID NO:20); (ii) a CDR-L2 comprising the amino acid sequence of KVSNRFS (SEQ ID NO:21); and (iii) a CDR-L3 comprising the amino acid sequence of FQGSYVPLT (SEQ ID NO:22). In some embodiments, a tetravalent antibody of the present disclosure is a dimer of two monomers, each monomer comprising two VL domains, each VL domain comprising one or more CDRs selected from (i) a CDR-L1 comprising the amino acid sequence of RSSQSIVHNDGNTYFE (SEQ ID NO:20); (ii) a CDR-L2 comprising the amino acid sequence of KVSNRFS (SEQ ID NO:21); and (iii) a CDR-L3 comprising the amino acid sequence of FQGSYVPLT (SEQ ID NO:22). In some embodiments, a tetravalent antibody of the present disclosure is a dimer of two monomers, each monomer comprising two VL domains, each VL domain comprising (i) a CDR-L1 comprising the amino acid sequence of RSSQSIVHNDGNTYFE (SEQ ID NO:20); (ii) a CDR-L2 comprising the amino acid sequence of KVSNRFS (SEQ ID NO:21); and (iii) a CDR-L3 comprising the amino acid sequence of FQGSYVPLT (SEQ ID NO:22).
[0082] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VL domains comprising the amino acid sequence of SEQ ID NO:24. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VL domains, each VL domain comprising the amino acid sequence of SEQ ID NO:24. In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VL domains comprising the amino acid sequence of SEQ ID NO:30. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VL domains, each VL domain comprising the amino acid sequence of SEQ ID NO:30.
[0083] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VL domains comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:24. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VL domains, each VL domain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:24. In some embodiments, the VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:24.
[0084] In some embodiments, a tetravalent antibody of the present disclosure comprises one or more VL domains comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:30. In some embodiments, a tetravalent antibody of the present disclosure comprises a monomer comprising two VL domains, each VL domain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NO:30. In some embodiments, the VL sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NO:30.
[0085] In some embodiments, a tetravalent antibody of the present disclosure comprises a single-chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NOs:1, 3, or 5. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising two single-chain polypeptides, each single-chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NOs:1, 3, or 5. In some embodiments, the single-chain polypeptide sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NOs:1, 3, or 5. In some embodiments, a tetravalent antibody of the present disclosure comprises two single-chain polypeptides, each comprising the amino acid sequence of SEQ ID NOs:1, 3, or 5.
[0086] In some embodiments, a tetravalent antibody of the present disclosure comprises a single-chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to a single-chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:2, 4, or 6. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising two single-chain polypeptides, each single-chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to a single-chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:2, 4, or 6. In some embodiments, the single-chain polypeptide sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in the single-chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:2, 4, or 6. In some embodiments, a tetravalent antibody of the present disclosure comprises two single-chain polypeptides, each encoded by the polynucleotide sequence of SEQ ID NOs:2, 4, or 6.
[0087] In some embodiments, a tetravalent antibody of the present disclosure comprises a light chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NOs:7, 9, or 13. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each light chain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NOs:7, 9, or 13. In some embodiments, the light chain polypeptide sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NOs:7, 9, or 13. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each light chain comprising the amino acid sequence of SEQ ID NOs:7, 9, or 13.
[0088] In some embodiments, a tetravalent antibody of the present disclosure comprises a light chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to a light chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:8, 10, or 14. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each light chain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to a light chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:8, 10, or 14. In some embodiments, the light chain polypeptide sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in the light chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:8, 10, or 14. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each light chain comprising a light chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:8, 10, or 14.
[0089] In some embodiments, a tetravalent antibody of the present disclosure comprises a heavy chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NOs:11 or 15. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each heavy chain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence of SEQ ID NOs:11 or 15. In some embodiments, the heavy chain polypeptide sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in SEQ ID NOs:11 or 15. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each heavy chain comprising the amino acid sequence of SEQ ID NOs:11 or 15.
[0090] In some embodiments, a tetravalent antibody of the present disclosure comprises a heavy chain polypeptide comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to a heavy chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:12 or 16. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each heavy chain comprising a sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to a heavy chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:12 or 16. In some embodiments, the heavy chain polypeptide sequence having at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but an anti-human PSGL-1 antibody comprising that sequence retains the ability to bind to human PSGL-1. In some embodiments, total of 1 to 10 amino acids have been substituted, inserted and/or deleted in the heavy chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs:12 or 16. In some embodiments, a tetravalent antibody of the present disclosure comprises a dimer of two monomers, each monomer comprising a heavy chain and a light chain, and each heavy chain comprising a light chain polypeptide encoded by the polynucleotide sequence of SEQ ID NOs: 12 or 16.
[0091] The present disclosure encompasses modifications to antibodies or polypeptide described herein, including functionally equivalent antibodies which do not significantly affect their properties and variants which have enhanced or decreased activity and/or affinity. Modification of polypeptides is routine practice in the art and need not be described in detail herein. Examples of modified polypeptides include polypeptides with conservative substitutions of amino acid residues, one or more deletions or additions of amino acids which do not significantly deleteriously change the functional activity, or use of chemical analogs.
[0092] Amino acid sequence insertions include amino- and/or carboxyl-terminal fusions ranging in length from one residue to polypeptides containing a hundred or more residues, as well as intrasequence insertions of single or multiple amino acid residues. Examples of terminal insertions include an antibody with an N-terminal methionyl residue or the antibody fused to an epitope tag. Other insertional variants of the antibody molecule include the fusion to the N- or C-terminus of the antibody of an enzyme or a polypeptide which increases the serum half-life of the antibody.
[0093] Substitution variants have at least one amino acid residue in the antibody molecule removed and a different residue inserted in its place. The sites of greatest interest for substitutional mutagenesis include the hypervariable regions, but FR alterations are also contemplated. Conservative substitutions are shown in the table below under the heading of "conservative substitutions." If such substitutions result in a change in biological activity, then more substantial changes, denominated "exemplary substitutions" in the table below, or as further described below in reference to amino acid classes, may be introduced and the products screened.
Amino Acid Substitutions
TABLE-US-00002 [0094] Original Conservative Exemplary Residue Substitutions Substitutions Ala (A) Val Val; Leu; Ile Arg (R) Lys Lys; Gln; Asn Asn (N) Gln Gln; His; Asp, Lys; Arg Asp (D) Glu Glu; Asn Cys (C) Ser Ser; Ala Gln (Q) Asn Asn; Glu Glu (E) Asp Asp; Gln Gly (G) Ala Ala His (H) Arg Asn; Gln; Lys; Arg Ile (I) Leu Leu; Val; Met; Ala; Phe; Norleucine Leu (L) Ile Norleucine; Ile; Val; Met; Ala; Phe Lys (K) Arg Arg; Gln; Asn Met (M) Leu Leu; Phe; Ile Phe (F) Tyr Leu; Val; Ile; Ala; Tyr Pro (P) Ala Ala Ser (S) Thr Thr Thr (T) Ser Ser Trp (W) Tyr Tyr; Phe Tyr (Y) Phe Trp; Phe; Thr; Ser Val (V) Leu Ile; Leu; Met; Phe; Ala; Norleucine
[0095] Substantial modifications in the biological properties of the antibody are accomplished by selecting substitutions that differ significantly in their effect on maintaining (a) the structure of the polypeptide backbone in the area of the substitution, for example, as a sheet or helical conformation, (b) the charge or hydrophobicity of the molecule at the target site, or (c) the bulk of the side chain. Naturally occurring residues are divided into groups based on common side-chain properties: [0096] (1) Non-polar: Norleucine, Met, Ala, Val, Leu, Ile; [0097] (2) Polar without charge: Cys, Ser, Thr, Asn, Gln; [0098] (3) Acidic (negatively charged): Asp, Glu; [0099] (4) Basic (positively charged): Lys, Arg; [0100] (5) Residues that influence chain orientation: Gly, Pro; and [0101] (6) Aromatic: Trp, Tyr, Phe, His
[0102] Non-conservative substitutions are made by exchanging a member of one of these classes for another class.
[0103] Any cysteine residue not involved in maintaining the proper conformation of the antibody also may be substituted, generally with serine, to improve the oxidative stability of the molecule and prevent aberrant cross-linking. Conversely, cysteine bond(s) may be added to the antibody to improve its stability, particularly where the antibody is an antibody fragment such as an Fv fragment. Exemplary cysteine mutations are described herein (e.g., the G44C VH domain mutation of SEQ ID NO:29, or the Q100C VL domain mutation of SEQ ID NO:30).
[0104] In some embodiments, a tetravalent antibody of the present disclosure comprises an antibody Fc domain. In some embodiments, the antibody Fc domain is a human Fc domain. In certain embodiments, the antibody Fc domain is a human IgG4 Fc domain.
[0105] In some embodiments, one or more amino acid residues in the heavy chain constant region and/or the light chain constant region of the antibody are modified. For example, amino acid residues of antibodies described in the Examples may be modified. In some embodiments, the Fc region of antibodies is modified to enhance or reduce ADCC and/or CDC activities of the antibodies. See Shields et al., J. Biol. Chem. 276:6591-6604 (2001); Presta et al., Biochem. Soc. Trans. 30:487-490 (2002).
[0106] In some embodiments, the Fc region of antibodies is modified to enhance dimer formation and/or stability, or to reduce dimer heterogeneity (e.g., shuffling). It has been demonstrated that a Serine to Proline mutation at position 241 using Kabat numbering (Kabat et al. 1991, Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91-3242) or at position 228 using the EU index (Edelman et al, 1969, Proc. Natl. Acad. Sci. USA, 63(1): 78-85) in the hinge region of human IgG4 results in considerable reduction of intra-chain disulfide bond formation, resulting in the reduction of IgG4 "half-antibody" molecules and reduced heterogeneity/shuffling of IgG4 molecules (Bloom et al. 1997, Protein Sci, 6:407-415; Angal et al, 1993, Molecular Immunology, 30(1): 105-108)). There are also published reports that this hinge mutation may decrease IgG4 shuffling and increase the half-life of the IgG4 molecules in vivo (Labrijn, et al, 2009, Nat Biotechnol 27:767-771; Stubenrauch, et al, 2010, Drug Metab Dispos 38:84-91). Van der Neut Kolfschoten et al, reported that the C.sub.H3 domain of IgG4 and not the core hinge is predominantly involved in the Fab arm exchange reaction (see Van der Neut Kolfschoten et al, 2007, Science, 317: 1554-1557 ("Van der Neut Kolfschoten") at page 1555, col. 2). Van der Neut Kolfschaten reported that exchanging the C.sub.H3 domain of IgG1 for the C.sub.H3 domain of IgG4 activated Fab arm exchange for the IgG1, while exchanging the C.sub.H3 domain of IgG4 abrogated Fab arm exchange for the IgG4 (see, p. 1555 and FIG. 2D).
[0107] In a specific embodiment, provided herein are tetravalent antibodies, that specifically bind to PSGL-1, and that contain one or more amino acid substitutions in the IgG4 hinge region, wherein said antibody or antigen-binding fragment thereof retains specific binding to said PSGL-1 and wherein IgG4 shuffling is reduced relative to an antibody comprising an IgG4 hinge region not comprising said one or more amino acid substitutions. In a specific embodiment, the IgG4 hinge region only comprises a single amino acid substitution. An example of a "human IgG4 hinge region," is the region on the heavy chain of an IgG4 antibody between the C.sub.HI and C.sub.H2 domains, as set forth in Angal et al., 1993, Molecular Immunology, 30(1): 105-108.
[0108] In a specific embodiment, a reduction in IgG4 shuffling is determined by detecting of a lower amount of half antibody molecules or of arm exchange produced from an antibody described herein which contains one or more amino acid substitutions in the hinge region, as compared to the amount of half antibody molecules or of arm exchange produced from an IgG4 molecule containing an IgG4 hinge region not comprising said one or more amino acid substitutions. Any assay well-known in the art can be used to detect half antibody production and bispecific antibody molecules. See, e.g., Van der Neut Kolfschoten et al, 2007, Science, 317: 1554-1557, for examples of assays to detect production of bispecific antibodies.
[0109] In a specific embodiment, provided herein are tetravalent antibodies that specifically bind to PSGL-1 and include a human IgG4 Fc domain comprising a Serine to Proline amino acid substitution at amino acid position 228 of the heavy chain numbered according to the EU index (also known as position 241 using Kabat numbering).
[0110] Modifications also include glycosylated and nonglycosylated polypeptides, as well as polypeptides with other post-translational modifications, such as, for example, glycosylation with different sugars, acetylation, and phosphorylation. Antibodies are glycosylated at conserved positions in their constant regions (Jefferis and Lund, 1997, Chem. Immunol. 65:111-128; Wright and Morrison, 1997, TibTECH 15:26-32). The oligosaccharide side chains of the immunoglobulins affect the protein's function (Boyd et al., 1996, Mol. Immunol. 32:1311-1318; Wittwe and Howard, 1990, Biochem. 29:4175-4180) and the intramolecular interaction between portions of the glycoprotein, which can affect the conformation and presented three-dimensional surface of the glycoprotein (Hefferis and Lund, supra; Wyss and Wagner, 1996, Current Opin. Biotech. 7:409-416). Oligosaccharides may also serve to target a given glycoprotein to certain molecules based upon specific recognition structures. Glycosylation of antibodies has also been reported to affect antibody-dependent cellular cytotoxicity (ADCC). In particular, CHO cells with tetracycline-regulated expression of .beta.(1,4)-N-acetylglucosaminyltransferase III (GnTIII), a glycosyltransferase catalyzing formation of bisecting GlcNAc, was reported to have improved ADCC activity (Umana et al., 1999, Mature Biotech. 17:176-180).
[0111] Glycosylation of antibodies is typically either N-linked or O-linked. N-linked refers to the attachment of the carbohydrate moiety to the side chain of an asparagine residue. The tripeptide sequences asparagine-X-serine, asparagine-X-threonine, and asparagine-X-cysteine, where X is any amino acid except proline, are the recognition sequences for enzymatic attachment of the carbohydrate moiety to the asparagine side chain. Thus, the presence of either of these tripeptide sequences in a polypeptide creates a potential glycosylation site. O-linked glycosylation refers to the attachment of one of the sugars N-acetylgalactosamine, galactose, or xylose to a hydroxyamino acid, most commonly serine or threonine, although 5-hydroxyproline or 5-hydroxylysine may also be used.
[0112] Addition of glycosylation sites to the antibody is conveniently accomplished by altering the amino acid sequence such that it contains one or more of the above-described tripeptide sequences (for N-linked glycosylation sites). The alteration may also be made by the addition of, or substitution by, one or more serine or threonine residues to the sequence of the original antibody (for O-linked glycosylation sites).
[0113] The glycosylation pattern of antibodies may also be altered without altering the underlying nucleotide sequence. Glycosylation largely depends on the host cell used to express the antibody. Since the cell type used for expression of recombinant glycoproteins, e.g., antibodies, as potential therapeutics is rarely the native cell, variations in the glycosylation pattern of the antibodies can be expected (see, e.g., Hse et al., 1997, J. Biol. Chem. 272:9062-9070).
[0114] In addition to the choice of host cells, factors that affect glycosylation during recombinant production of antibodies include growth mode, media formulation, culture density, oxygenation, pH, purification schemes, and the like. Various methods have been proposed to alter the glycosylation pattern achieved in a particular host organism including introducing or overexpressing certain enzymes involved in oligosaccharide production (U.S. Pat. Nos. 5,047,335; 5,510,261; and 5,278,299). Glycosylation, or certain types of glycosylation, can be enzymatically removed from the glycoprotein, for example using endoglycosidase H (Endo H), N-glycosidase F, endoglycosidase F1, endoglycosidase F2, or endoglycosidase F3. In addition, the recombinant host cell can be genetically engineered to be defective in processing certain types of polysaccharides. These and similar techniques are well known in the art.
[0115] In some embodiments, an antibody of the present disclosure is modified using coupling techniques known in the art, including, but not limited to, enzymatic means, oxidative substitution, and chelation. Modifications can be used, for example, for attachment of labels for immunoassay.
[0116] The tetravalent antibody or polypeptide of the present disclosure may be conjugated (for example, linked) to an agent, such as a therapeutic agent or a label. Examples of therapeutic agents are radioactive moieties, cytotoxins, and chemotherapeutic molecules.
[0117] The tetravalent antibody (or polypeptide) of the present disclosure may be linked to a label such as a fluorescent molecule, a radioactive molecule, an enzyme, or any other labels known in the art. As used herein, the term "label" refers to any molecule that can be detected.
[0118] In a certain embodiment, an antibody may be labeled by incorporation of a radiolabeled amino acid. In a certain embodiment, biotin moieties that can be detected by marked avidin (e.g., streptavidin containing a fluorescent marker or enzymatic activity that can be detected by optical or colorimetric methods) may be attached to the antibody. In certain embodiments, a label may be incorporated into or attached to another reagent which in turn binds to the antibody of interest. For example, a label may be incorporated into or attached to an antibody that in turn specifically binds the antibody of interest. In certain embodiments, the label or marker can also be therapeutic. Various methods of labeling polypeptides and glycoproteins are known in the art and may be used. Certain general classes of labels include, but are not limited to, enzymatic, fluorescent, chemiluminescent, and radioactive labels. Examples of labels for polypeptides include, but are not limited to, the following: radioisotopes or radionucleoides (e.g., .sup.3H, .sup.14C, .sup.15N, .sup.35S, .sup.90Y, .sup.99Tc, .sup.111In, .sup.125I or .sup.131I) fluorescent labels (e.g., fluorescein isothocyanate (FITC), rhodamine, lanthanide phosphors, or phycoerythrin (PE)), enzymatic labels (e.g., horseradish peroxidase, .beta.-galactosidase, luciferase, alkaline phosphatase, glucose oxidase, glucose-6-phosphate dehydrogenase, alcohol dehydrogenase, malate dehydrogenase, penicillinase, or luciferase), chemiluminescent, biotinyl groups, predetermined polypeptide epitopes recognized by a secondary reporter (e.g., leucine zipper pair sequences, binding sites for secondary antibodies, metal binding domains, or epitope tags). In certain embodiments, labels are attached by spacer arms of various lengths to reduce potential steric hindrance.
[0119] Based on the description herein, a tetravalent antibody of the present disclosure may be tested according to a variety of in vitro and in vivo assays known in the art. Such assays may include, e.g., binding assays directed to the ability of a tetravalent antibody or fragment thereof to bind an epitope or polypeptide of interest (e.g., human PSGL-1 or an epitope thereof), or functional assays directed to one or more functional properties of a tetravalent antibody or fragment thereof.
[0120] In some embodiments, a tetravalent antibody of the present disclosure may be tested for binding activity against human PSGL-1. In some embodiments, binding of a tetravalent antibody to human PSGL-1 or an epitope thereof may be tested in an in vitro binding assay. A variety of binding assays are known in the art. Such binding assays may be cell-based assays (e.g., testing the ability of a tetravalent antibody to bind a cell expressing human PSGL-1 or an epitope thereof), or they may be polypeptide-based (e.g., testing the ability of a tetravalent antibody to bind human PSGL-1 or an epitope thereof). In some embodiments, a tetravalent antibody of the present disclosure is tested for binding to a cell expressing human PSGL-1 (e.g., an Sp2 cell, as exemplified infra) by flow cytometry, FRET, histochemical assays, and the like. Other suitable binding assays may include without limitation equilibrium methods (e.g., enzyme-linked immunoabsorbent assay (ELISA), radioimmunoassay (MA), Biacore.TM. analysis, indirect binding assay, competitive inhibition assay, fluorescence resonance energy transfer (FRET), immunoprecipitation, gel electrophoresis and chromatography (e.g., gel filtration).
[0121] In some embodiments, a tetravalent antibody of the present disclosure may be tested for one or more functional assays for PSGL-1 function. In some embodiments, a tetravalent antibody of the present disclosure may be tested for induction of apoptosis in cell(s) expressing human PSGL-1. In some embodiments, a tetravalent antibody of the present disclosure displays enhanced induction of apoptosis in a target cell (e.g., a cell expressing human PSGL-1 or an epitope thereof) as compared to a conventional (e.g., bivalent) antibody having one or more VH or VL domains in common with the tetravalent antibody (e.g., a parental antibody). As demonstrated herein, tetravalent antibodies of the present disclosure displayed greater potency in inducing apoptosis in target cells than parental antibodies having a common VH and/or VL domain. Apoptosis assays are described in the art and can be readily carried out by one of skill in the art (see, e.g., Muppidi, J., Porter, M. and Siegel, R. M. 2004. Measurement of Apoptosis and Other Forms of Cell Death. Current Protocols in Immunology. 59:3.17.1-3.17.36). Selected assays for detecting apoptosis (e.g., Annexin V or propidium iodide staining) are exemplified supra. The term "induce" or "inducing" means initiation of or an increase of apoptosis above a control level. Apoptosis of activated T cells can be induced by about 10%, about 20%, about 30%, about 40%, about 50%, about 60%, about 70%, about 80%, about 90%, about 100%, about 125%, about 150% or more compared to a control (e.g. Apoptosis of activated T cells in the absence of the antibodies describe herein or in the presence of a non-specific antibody).
[0122] T cells and T cell lines which are appropriate for use in the assays described herein relating to PSGL-1 activity are readily available (e.g., ARR, DU.528, Jurkat, H-SB2, RPMI 8402, CML-T1, Karpas 45, KE-37/SKW-3, SUP-T1, SUP-T3, MOLT 3/4, P12-Ichikawa, PF-382, CCRF-CEM, HPB-ALL, K-Tl, TALL-1, MOLT 16/17, TALL-104, DND-41, Loucy, MOLT 13, Peer/Bel3, HUT 78/H9, HUT 102, MT-1, DEL, JB6, Karpas 299, SU-DHL1, 12H5, 3D054.8, 3DO11.10, 8D051.15, or 3D018.3) or can be readily identified using methods known in the art (see, e.g., Thornton, A. M. 2003. Fractionation of T and B Cells Using Magnetic Beads. Current Protocols in Immunology. 55:3.5A. 1-3.5A. i 1 Hathcock, K. 2001. T Cell Enrichment by Cytotoxic Elimination of B Cells and Accessory Cells. Current Protocols in Immunology. 00:3.3.1-3.3.5., Horgan, K., Shaw, S. and Boirivant, M. 2009. Immunomagnetic Purification of T Cell Subpopulations. Current Protocols in Immunology. 85:7.4.1-7.4.9., and Kanof, M. E. 2001. Purification of T Cell Subpopulations. Current Protocols in Immunology. 00:7.3.1-7.3.5). In particular embodiments, cells or cell lines for use in cell proliferation assays can express PSGL-1, endogenously or recombinantly. Cells or cell lines for use in cell viability assays can express PSGL-1, endogenously or recombinantly, and exert changes in cell viability in response to PSGL-1 ligand or anti-PSGL-1 antibody binding. Cells or cell lines for use in apoptosis assays can express PSGL-1, endogenously or recombinantly, and exert changes in apoptosis in response to PSGL-1 ligand or anti-PSGL-1 antibody binding. Preferably the cells or cell lines are human (e.g. ARR, DU.528, Jurkat, H-SB2, RPMI 8402, CML-Tl, Karpas 45, KE-37/SKW-3, SUP-Tl, SUP-T3, MOLT 3/4, P12-Ichikawa, PF-382, CCRF-CEM, HPB-ALL, K-Tl, TALL-1, MOLT 16/17, TALL-104, DND-41, Loucy, MOLT 13, Peer/Bel3, HUT 78/H9, HUT 102, MT-1, DEL, JB6, Karpas 299, or SU-DHL1).
[0123] In some embodiments, a tetravalent antibody of the present disclosure may be tested for inhibition of delayed type hypersensitivity (DTH). In some embodiments, a tetravalent antibody of the present disclosure displays enhanced inhibition of DTH (e.g., in a trans vivo animal model) as compared to a conventional (e.g., bivalent) antibody having one or more VH or VL domains in common with the tetravalent antibody (e.g., a parental antibody). As demonstrated herein, tetravalent antibodies of the present disclosure displayed greater potency in inhibiting DTH in a trans vivo mouse footpad swelling model than parental antibodies having a common VH and/or VL domain. DTH assays are described in the art and exemplified infra and can be readily carried out by one of skill in the art. In some embodiments, a tetravalent antibody of the present disclosure may display a potency of DTH inhibition that may be increased by about 10%, about 20%, about 30%, about 40%, about 50%, about 60%, about 70%, about 80%, about 90%, about 100%, about 125%, about 150%, about 200%, about 300%, about 400%, about 500%, about 600%, or more compared to a control (e.g. inhibition of DTH by a conventional or bivalent antibody, such as the parental antibody).
III. Polynucleotides, Vectors, Host Cells, and Antibody Production
[0124] The present disclosure also provides polynucleotides comprising a polynucleotide encoding any of the tetravalent antibodies and/or polypeptides described herein. In some embodiments, the polypeptides comprise the sequences of light chain and heavy chain variable regions. In some embodiments, the polynucleotide is an isolated polynucleotide (e.g., isolated from a host cell or from one or more different polynucleotides).
[0125] Provided herein are polynucleotides encoding any of the tetravalent antibodies or polypeptide constituents (e.g., monomers such as single-chain polypeptides, antibody heavy chains, and/or antibody light chains) described herein. In some embodiments, a polynucleotide of the present disclosure encodes a polypeptide sequence selected from SEQ ID NOs:1, 3, 5, 7, 9, 11, 13, 15, and 17-31. In some embodiments, a polynucleotide of the present disclosure comprises a polynucleotide sequence selected from SEQ ID NOs:2, 4, 6, 8, 10, 12, 14, and 16. In some embodiments, a polynucleotide of the present disclosure comprises one or more introns. In other embodiments, a polynucleotide of the present disclosure does not comprise an intron, e.g., a cDNA or processed mRNA sequence.
[0126] It is appreciated by those of ordinary skill in the art that, as a result of the degeneracy of the genetic code, there are many nucleotide sequences that encode a polypeptide as described herein. Some of these polynucleotides bear minimal homology to the nucleotide sequence of any native gene. Thus, polynucleotides that vary due to differences in codon usage are specifically contemplated by the present disclosure. Further, alleles of the genes comprising the polynucleotide sequences provided herein are within the scope of the present disclosure. Alleles are endogenous genes that are altered as a result of one or more mutations, such as deletions, additions, and/or substitutions of nucleotides. The resulting mRNA and protein can, but need not, have an altered structure or function. Alleles can be identified using standard techniques (such as hybridization, amplification, and/or database sequence comparison).
[0127] Also provided herein are polynucleotides that are optimized, e.g., by codon/RNA optimization, replacement with heterologous signal sequences, and elimination of mRNA instability elements. Methods to generate optimized nucleic acids encoding a tetravalent antibody or polypeptide thereof for recombinant expression by introducing codon changes and/or eliminating inhibitory regions in the mRNA can be carried out by adapting the optimization methods described in, e.g., U.S. Pat. Nos. 5,965,726; 6,174,666; 6,291,664; 6,414,132; and 6,794,498, accordingly. For example, potential splice sites and instability elements (e.g., A/T or A/U rich elements) within the RNA can be mutated without altering the amino acids encoded by the nucleic acid sequences to increase stability of the RNA for recombinant expression. The alterations utilize the degeneracy of the genetic code, e.g., using an alternative codon for an identical amino acid. In some embodiments, it can be desirable to alter one or more codons to encode a conservative mutation, e.g., a similar amino acid with similar chemical structure and properties and/or function as the original amino acid. Such methods can increase expression of an anti-PSGL-1 tetravalent antibody or polypeptide thereof relative to the expression of an anti-PSGL-1 tetravalent antibody or polypeptide thereof encoded by polynucleotides that have not been optimized. Furthermore, the polynucleotide sequences can be designed to match the preferred codon usage in the host cell, e.g. E. coli codon usage or CHO codon usage.
[0128] An optimized polynucleotide sequence encoding a tetravalent antibody or polypeptide thereof described herein can hybridize to an unoptimized polynucleotide sequence encoding a tetravalent antibody or polypeptide thereof described herein. In specific embodiments, an optimized nucleotide sequence encoding a tetravalent antibody or polypeptide thereof described herein hybridizes under high stringency conditions to an unoptimized polynucleotide sequence encoding a tetravalent antibody or polypeptide thereof described herein. In a specific embodiment, an optimized nucleotide sequence encoding a tetravalent antibody or polypeptide thereof described herein hybridizes under high stringency, intermediate or lower stringency hybridization conditions to an unoptimized nucleotide sequence encoding a tetravalent antibody or polypeptide thereof described herein. Information regarding hybridization conditions have been described, see, e.g., U.S. Patent Application Publication No. US 2005/0048549 (e.g., paragraphs 72-73), which is incorporated herein by reference in its entirety.
[0129] The polynucleotides of the present disclosure can be obtained using chemical synthesis, recombinant methods, or PCR. Methods of chemical polynucleotide synthesis are well known in the art and need not be described in detail herein. One of skill in the art can use the sequences provided herein and a commercial DNA synthesizer to produce a desired DNA sequence.
[0130] For preparing polynucleotides using recombinant methods, a polynucleotide comprising a desired sequence can be inserted into a suitable vector, and the vector in turn can be introduced into a suitable host cell for replication and amplification, as further discussed herein. Polynucleotides can be inserted into host cells by any means known in the art. Cells are transformed by introducing an exogenous polynucleotide by direct uptake, endocytosis, transfection, F-mating, or electroporation. Once introduced, the exogenous polynucleotide can be maintained within the cell as a non-integrated vector (such as a plasmid) or integrated into the host cell genome. The polynucleotide so amplified can be isolated from the host cell by methods well known within the art. See, e.g., Sambrook et al. (1989).
[0131] Alternatively, PCR allows reproduction of DNA sequences. PCR technology is well known in the art and is described in U.S. Pat. Nos. 4,683,195; 4,800,159; 4,754,065; and 4,683,202, as well as PCR: The Polymerase Chain Reaction, Mullis et al. eds., Birkauswer Press, Boston (1994).
[0132] The present disclosure also provides vectors (e.g., cloning vectors or expression vectors) comprising a nucleic acid sequence encoding any of the polypeptides (including antibodies) described herein. Suitable cloning vectors can be constructed according to standard techniques or may be selected from a large number of cloning vectors available in the art. While the cloning vector selected may vary according to the host cell intended to be used, useful cloning vectors generally have the ability to self-replicate, may possess a single target for a particular restriction endonuclease, and/or may carry genes for a marker that can be used in selecting clones containing the vector. Suitable examples include plasmids and bacterial viruses, e.g., pUC18, pUC19, Bluescript (e.g., pBS SK+) and its derivatives, mp18, mp19, pBR322, pMB9, ColE1, pCR1, RP4, phage DNAs, and shuttle vectors such as pSA3 and pAT28. These and many other cloning vectors are available from commercial vendors such as BioRad, Strategene, and Invitrogen.
[0133] Expression vectors generally are replicable polynucleotide constructs that contain a polynucleotide according to the present disclosure. The expression vector may replicable in the host cells either as episomes or as an integral part of the chromosomal DNA. Suitable expression vectors include but are not limited to plasmids, viral vectors, including adenoviruses, adeno-associated viruses, retroviruses, cosmids, and expression vector(s) disclosed in PCT Publication No. WO 87/04462. Vector components may generally include, but are not limited to, one or more of the following: a signal sequence; an origin of replication; one or more marker genes; and suitable transcriptional controlling elements (such as promoters, enhancers, or terminator). For expression (i.e., translation), one or more translational controlling elements are also usually required, such as ribosome binding sites, translation initiation sites, or stop codons.
[0134] Methods of making antibodies and polypeptides derived from the antibodies are known in the art and are disclosed herein. Well-established methods may be used to identify anti-PSGL antibodies (e.g., antibodies that specifically bind to human PSGL-1), from which variable domains (e.g., VH and/or VL domains) may be used in the tetravalent antibodies of the present disclosure. Exemplary anti-human PSGL-1 antibodies, as well as methods for screening, producing, and purifying such antibodies, are described in International Application Pub. No. WO 2012/174001.
[0135] Additional anti-human PSGL-1 antibodies may be identified using methods known in the art, such as those described in International Application Pub. No. WO 2012/174001 and supra. For example, the monoclonal antibodies can be prepared using hybridoma technology, such as those described by Kohler and Milstein (1975), Nature, 256:495. In a hybridoma method, a mouse, a hamster, or other appropriate host animal, is typically immunized with an immunizing agent (e.g., a cell expressing human PSGL-1 or a fragment thereof) to elicit lymphocytes that produce or are capable of producing antibodies that specifically bind to the immunizing agent. Alternatively, the lymphocytes may be immunized in vitro. The lymphocytes are then fused with an immortalized cell line using a suitable fusing agent, such as polyethylene glycol, to form a hybridoma cell (Goding, Monoclonal Antibodies: Principles and Practice, Academic Press, (1986) pp. 59-1031). Immortalized cell lines are usually transformed mammalian cells, particularly myeloma cells of rodent, rabbit, bovine, or human origin. Usually, rat or mouse myeloma cell lines are employed. The hybridoma cells may be cultured in a suitable culture medium that desirably contains one or more substances that inhibit the growth or survival of the unfused, immortalized cells. For example, if the parental cells lack the enzyme hypoxanthine guanine phosphoribosyl transferase (HGPRT or HPRT), the culture medium for the hybridomas typically includes hypoxanthine, aminopterin, and thymidine ("HAT medium"), which substances prevent the growth of HGPRT-deficient cells.
[0136] Desired immortalized cell lines are those that fuse efficiently, support stable high level expression of antibody by the selected antibody-producing cells, and are sensitive to a medium such as HAT medium. More desirable immortalized cell lines are murine myeloma lines, which can be obtained, for instance, from the Salk Institute Cell Distribution Center, San Diego, Calif. and the American Type Culture Collection, Manassas, Va. Human myeloma and mouse-human heteromyeloma cell lines also have been described for the production of human monoclonal antibodies (Kozbor, J. Immunol. (1984), 133:3001; Brodeur et al., Monoclonal Antibody Production Techniques and Applications, Marcel Dekker, Inc., New York, (1987) pp. 51-63).
[0137] The culture medium in which the hybridoma cells are cultured can then be assayed for the presence of monoclonal antibodies. The antibody may be screened for having specific binding to an ORP150 polypeptide (such as binding to an epitope in an extracellular domain of the ORP150 polypeptide) obtained from or expressed on the cell surface of plasmacytoma, multiple myeloma, colorectal, gastric, or esophageal cancer or tumor cells. Cancer cells or an ORP150 polypeptide (or a fragment thereof containing an extracellular domain of an ORP150 polypeptide) may be used for screening. For example, RPMI8226, U266, NCI-H929, L363, Colo205, DLD-1, HT29, SNU-1, Kato-III, or CE146T cells may be used for screening. A polypeptide comprising amino acids 673-800, 701-800, 673-752, or 723-732 of SEQ ID NO:17 may also be used for screening.
[0138] In some embodiments, the binding specificity of monoclonal antibodies produced by the hybridoma cells is determined by immunoprecipitation or by an in vitro binding assay, such as radioimmunoassay (MA) or enzyme-linked immunoabsorbent assay (ELISA). Such techniques and assays are known in the art. The binding affinity of the monoclonal antibody can, for example, be determined by the Scatchard analysis of Munson and Pollard (1980), Anal. Biochem., 107:220.
[0139] After the desired hybridoma cells are identified, the clones may be subcloned by limiting dilution procedures and grown by standard methods (Goding, supra). Suitable culture media for this purpose include, for example, Dulbecco's Modified Eagle's Medium or RPMI-1640 medium. Alternatively, the hybridoma cells may be grown in vivo as ascites in a mammal.
[0140] The monoclonal antibodies can be generated by culturing the hybridoma cells, and the antibodies secreted by the hybridoma cells may further be isolated or purified. Antibodies may be isolated or purified from the culture medium or ascites fluid by conventional immunoglobulin purification procedures such as, for example, protein A-Sepharose, hydroxylapatite chromatography, gel electrophoresis, dialysis, or affinity chromatography.
[0141] The tetravalent antibodies or polypeptides of the present disclosure may be generated by screening a library of antibodies or polypeptides to select antibodies or polypeptides that bind to human PSGL-1, e.g., expressed on the cell surface of a cell. Antibody phage display libraries known in the art may be used. In some embodiments, the antibodies in the library (e.g., displayed on phage) are single-chain Fv (scFv) fragments or Fab fragment. In some embodiments, the antibodies in the library (e.g., displayed on phage) are single-domain antibodies. For example, a single-domain antibody may comprise all or a portion of the heavy chain variable domain or all or a portion of the light chain variable domain of an antibody. In some embodiments, the antibodies in the library are human antibodies. The antibodies identified may further be tested for their capabilities to induce cell death (e.g., apoptosis) and/or bind human PSGL-1 using methods known in the art and described herein.
[0142] The tetravalent antibodies of the present disclosure can be made by recombinant DNA methods, such as those described in U.S. Pat. Nos. 4,816,567 and 6,331,415. For example, DNA encoding the variable or constant region of any of the tetravalent antibodies of the present disclosure (or single, heavy, or light chain polypeptides that are constituents thereof) can be readily isolated and sequenced using conventional procedures (e.g., by using oligonucleotide probes that are capable of binding specifically to genes encoding the heavy and light chains of murine antibodies). The hybridoma cells of the present disclosure serve as a preferred source of such DNA. Once isolated, the DNA can be placed into expression vectors, which are then transfected into host cells such as simian COS cells, Chinese hamster ovary (CHO) cells, or myeloma cells that do not otherwise produce immunoglobulin protein to synthesize monoclonal antibodies in the recombinant host cells. The DNA also can be modified, for example, by substituting the coding sequence for human heavy and light chain constant domains in place of the homologous murine sequences (U.S. Pat. No. 4,816,567) or by covalently joining to the immunoglobulin coding sequence all or part of the coding sequence for a non-immunoglobulin polypeptide. Such a non-immunoglobulin polypeptide can be substituted for the constant domains of an antibody of the present disclosure, or can be substituted for the variable domains of one antigen-combining site of an antibody of the present disclosure to create a chimeric bivalent antibody.
[0143] In some embodiments, the tetravalent antibodies of the present disclosure are expressed from two expression vectors. For example, each expression vector may express one monomer of a dimer of the present disclosure (e.g., a single-chain polypeptide or antibody heavy or light chain polypeptide). Alternatively, both monomers of a dimer of the present disclosure are expressed from a single expression vector.
[0144] Normally the expression vector has transcriptional and translational regulatory sequences which are derived from a species compatible with a host cell. In addition, the vector ordinarily carries a specific gene(s) which is (are) capable of providing phenotypic selection in transformed cells.
[0145] A wide variety of recombinant host-vector expression systems for eukaryotic cells are known and can be used in the present disclosure. For example, Saccharomyces cerevisiae, or common baker's yeast, is the most commonly used among eukaryotic microorganisms, although a number of other strains, such as Pichia pastoris, are available. Cell lines derived from multicellular organisms such as Sp2/0 or Chinese Hamster Ovary (CHO), which are available from the ATCC, may also be used as hosts. Typical vector plasmids suitable for eukaryotic cell transformations are, for example, pSV2neo and pSV2gpt (ATCC), pSVL and pSVK3 (Pharmacia), and pBPV-1/pML2d (International Biotechnology, Inc.).
[0146] The eukaryotic host cells useful in the present disclosure are, for example, hybridoma, myeloma, plasmacytoma, or lymphoma cells. However, other eukaryotic host cells may be suitably utilized provided the mammalian host cells are capable of recognizing transcriptional and translational DNA sequences for expression of the proteins; processing the leader peptide by cleavage of the leader sequence and secretion of the proteins; and providing post-translational modifications of the proteins, e.g., glycosylation.
[0147] Accordingly, the present disclosure provides host cells (e.g., eukaryotic host cells) which are transformed by recombinant expression vectors comprising DNA constructs disclosed herein and which are capable of expressing the tetravalent antibodies or polypeptides of the present disclosure. In some embodiments, the transformed host cells of the present disclosure comprise at least one DNA construct comprising a polynucleotide of the present disclosure, or a polynucleotide expressing a monomer, dimer, or tetravalent antibody of the present disclosure, and transcriptional and translational regulatory sequences which are positioned in relation to the coding DNA sequences to direct expression of antibodies or polypeptides.
[0148] Any host cells capable of over-expressing heterologous DNAs can be used for the purpose of isolating the genes encoding the antibody, polypeptide, or protein of interest. Non-limiting examples of mammalian host cells include but not limited to COS, HeLa, and CHO cells. See also PCT Publication No. WO 87/04462. Suitable non-mammalian host cells include prokaryotes (such as E. coli or B. subtillis) and yeast (such as S. cerevisae, S. pombe, or K. lactis).
[0149] The host cells used in the present disclosure may be transformed in a variety of ways by standard transfection procedures well known in the art. Among the standard transfection procedures which may be used are electroporation techniques, protoplast fusion and calcium-phosphate precipitation techniques. Such techniques are generally described by F. Toneguzzo et al. (1986), Mol. Cell. Biol., 6:703-706; G. Chu et al., Nucleic Acid Res. (1987), 15:1311-1325; D. Rice et al., Proc. Natl. Acad. Sci. USA (1979), 79:7862-7865; and V. Oi et al., Proc. Natl. Acad. Sci. USA (1983), 80:825-829. The vectors containing the polynucleotides of interest can be introduced into the host cell by any of a number of appropriate means, including electroporation, transfection employing calcium chloride, rubidium chloride, calcium phosphate, DEAE-dextran, or other substances; microprojectile bombardment; lipofection; and infection (e.g., where the vector is an infectious agent such as vaccinia virus). The choice of introducing vectors or polynucleotides often depends on features of the host cell.
[0150] In the case of two expression vectors, the two expression vectors can be transferred into a host cell one by one separately or together (co-transfer or co-transfect).
[0151] The present disclosure also provides a method for producing the antibodies or polypeptides that comprises culturing a host cell comprising an expression vector(s) encoding the antibodies or the polypeptides, and recovering the antibodies or polypeptides from the culture by ways well known to one skilled in the art.
[0152] Furthermore, the desired antibodies can be produced in a transgenic animal. A suitable transgenic animal can be obtained according to standard methods which include micro-injecting into eggs the appropriate expression vectors, transferring the eggs into pseudo-pregnant females, and selecting a descendant expressing the desired antibody.
[0153] The present disclosure also provides chimeric tetravalent antibodies that specifically bind human PSGL-1. For example, the variable and constant regions of the tetravalent antibody are from separate species. In some embodiments, the variable regions of both heavy chain and light chain are from the murine antibodies described herein. The chimeric antibody of the present disclosure can be prepared by techniques well-established in the art. See for example, U.S. Pat. Nos. 6,808,901; 6,652,852; 6,329,508; 6,120,767; and 5,677,427, each of which is hereby incorporated by reference. In general, the chimeric antibody can be prepared by obtaining cDNAs encoding the heavy and light chain variable regions of the antibodies, inserting the cDNAs into an expression vector, which upon being introduced into eukaryotic host cells, expresses the chimeric antibody of the present disclosure. Desirably, the expression vector carries a functionally complete constant heavy or light chain sequence so that any variable heavy or light chain sequence can be easily inserted into the expression vector.
[0154] The present disclosure provides a humanized tetravalent antibody that specifically binds to human PSGL-1. The humanized antibody is typically a human antibody in which residues from CDRs are replaced with residues from CDRs of a non-human species such as mouse, rat, or rabbit having the desired specificity, affinity and capacity. In some instances, Fv framework residues of the human antibody are replaced by corresponding non-human residues.
[0155] There are four general steps to humanize a monoclonal antibody. These are: (1) determining the nucleotide and predicted amino acid sequence of the starting antibody light and heavy variable domains, (2) designing the humanized antibody, i.e., deciding which antibody framework region to use during the humanizing process, (3) the actual humanizing methodologies/techniques, and (4) the transfection and expression of the humanized antibody. See, for example, U.S. Pat. Nos. 4,816,567; 5,807,715; 5,866,692; 6,331,415; 5,530,101; 5,693,761; 5,693,762; 5,585,089; 6,180,370; and 6,548,640. For example, the constant region may be engineered to more resemble human constant regions to avoid immune response if the antibody is used in clinical trials and treatments in humans. See, for example, U.S. Pat. Nos. 5,997,867 and 5,866,692.
[0156] It is important that antibodies be humanized with retention of high affinity for the antigen and other favorable biological properties. To achieve this goal, humanized antibodies can be prepared by a process of analysis of the parental sequences and various conceptual humanized products using three dimensional models of the parental and humanized sequences. Three dimensional immunoglobulin models are commonly available and are familiar to those skilled in the art. Computer programs are available which illustrate and display probable three-dimensional conformational structures of selected candidate immunoglobulin sequences. Inspection of these displays permits analysis of the likely role of the residues in the functioning of the candidate immunoglobulin sequence, i.e., the analysis of residues that influence the ability of the candidate immunoglobulin to bind its antigen. In this way, FR residues can be selected and combined from the consensus and import sequence so that the desired antibody characteristic, such as increased affinity for the target antigen(s), is achieved. In general, the CDR residues are directly and most substantially involved in influencing antigen binding. The humanized antibodies may also contain modifications in the hinge region to improve one or more characteristics of the antibody.
[0157] In another alternative, antibodies may be screened and made recombinantly by phage display technology. See, for example, U.S. Pat. Nos. 5,565,332; 5,580,717; 5,733,743 and 6,265,150; and Winter et al., Annu. Rev. Immunol. 12:433-455 (1994). Alternatively, the phage display technology (McCafferty et al., Nature 348:552-553 (1990)) can be used to produce human antibodies and antibody fragments in vitro, from immunoglobulin variable (V) domain gene repertoires from unimmunized donors. According to this technique, antibody V domain genes are cloned in-frame into either a major or minor coat protein gene of a filamentous bacteriophage, such as M13 or fd, and displayed as functional antibody fragments on the surface of the phage particle. Because the filamentous particle contains a single-stranded DNA copy of the phage genome, selections based on the functional properties of the antibody also result in selection of the gene encoding the antibody exhibiting those properties. Thus, the phage mimics some of the properties of the B-cell. Phage display can be performed in a variety of formats; for review see, e.g., Johnson, Kevin S. and Chiswell, David J., Current Opinion in Structural Biology 3, 564-571 (1993). Several sources of V-gene segments can be used for phage display. Clackson et al., Nature 352:624-628 (1991) isolated a diverse array of anti-oxazolone antibodies from a small random combinatorial library of V genes derived from the spleens of immunized mice. A repertoire of V genes from unimmunized human donors can be constructed, and antibodies to a diverse array of antigens (including self-antigens) can be isolated essentially following the techniques described by Mark et al., J. Mol. Biol. 222:581-597 (1991), or Griffith et al., EMBO J. 12:725-734 (1993). In a natural immune response, antibody genes accumulate mutations at a high rate (somatic hypermutation). Some of the changes introduced will confer higher affinity, and B-cells displaying high-affinity surface immunoglobulin are preferentially replicated and differentiated during subsequent antigen challenge. This natural process can be mimicked by employing the technique known as "chain shuffling." Marks et al., Bio/Technol. 10:779-783 (1992)). In this method, the affinity of "primary" human antibodies obtained by phage display can be improved by sequentially replacing the heavy and light chain V region genes with repertoires of naturally occurring variants (repertoires) of V domain genes obtained from unimmunized donors. This technique allows the production of antibodies and antibody fragments with affinities in the pM-nM range. A strategy for making very large phage antibody repertoires (also known as "the mother-of-all libraries") has been described by Waterhouse et al., Nucl. Acids Res. 21:2265-2266 (1993). Gene shuffling can also be used to derive human antibodies from rodent antibodies, where the human antibody has similar affinities and specificities to the starting rodent antibody. According to this method, which is also referred to as "epitope imprinting," the heavy or light chain V domain gene of rodent antibodies obtained by phage display technique is replaced with a repertoire of human V domain genes, creating rodent-human chimeras. Selection on antigen results in isolation of human variable regions capable of restoring a functional antigen-binding site, i.e., the epitope governs (imprints) the choice of partner. When the process is repeated in order to replace the remaining rodent V domain, a human antibody is obtained (see PCT Publication No. WO 93/06213, published Apr. 1, 1993). Unlike traditional humanization of rodent antibodies by CDR grafting, this technique provides completely human antibodies, which have no framework or CDR residues of rodent origin. It is apparent that although the above discussion pertains to humanized antibodies, the general principles discussed are applicable to customizing antibodies for use, for example, in dogs, cats, primates, equines, and bovines.
[0158] In certain embodiments, the antibody is a fully human antibody. Non-human antibodies that specifically bind an antigen can be used to produce a fully human antibody that binds to that antigen. For example, the skilled artisan can employ a chain swapping technique, in which the heavy chain of a non-human antibody is co-expressed with an expression library expressing different human light chains. The resulting hybrid antibodies, containing one human light chain and one non-human heavy chain, are then screened for antigen binding. The light chains that participate in antigen binding are then co-expressed with a library of human antibody heavy chains. The resulting human antibodies are screened once more for antigen binding. Techniques such as this one are further described in U.S. Pat. No. 5,565,332. In addition, an antigen can be used to inoculate an animal that is transgenic for human immunoglobulin genes. See, e.g., U.S. Pat. No. 5,661,016.
[0159] The present disclosure also provides bispecific antibodies. A bispecific antibody has binding specificities for at least two different antigens (including different epitopes). In some embodiments, a bispecific antibody of the present disclosure includes two or more different VH and/or VL domains that specifically bind PSGL-1. In some embodiments, the two or more different VH and/or VL domains specifically bind the same epitope of PSGL-1. In some embodiments, the two or more different VH and/or VL domains specifically bind different epitopes of PSGL-1, which may or may not be overlapping epitopes.
[0160] A bispecific antibody (a monoclonal antibody that has binding specificities for at least two different antigens) can be prepared using the antibodies disclosed herein. Methods for making bispecific antibodies are known in the art (see, e.g., Suresh et al., 1986, Methods in Enzymology 121:210). Traditionally, the recombinant production of bispecific antibodies was based on the coexpression of two immunoglobulin heavy chain-light chain pairs, with the two heavy chains having different specificities (Millstein and Cuello, 1983, Nature 305, 537-539). In some embodiments, a bispecific tetravalent antibody may be produced using the methods exemplified supra.
[0161] According to one approach to making bispecific antibodies, antibody variable domains with the desired binding specificities (antibody-antigen combining sites) are fused to immunoglobulin constant domain sequences. In some embodiments, the fusion is with an immunoglobulin heavy chain constant domain, comprising at least part of the hinge, CH2, and CH3 regions. In some embodiments, the first heavy chain constant region (CH1), containing the site necessary for light chain binding, is present in at least one of the fusions. DNAs encoding the immunoglobulin heavy chain fusions and, if desired, the immunoglobulin light chain, are inserted into separate expression vectors, and are cotransfected into a suitable host organism. This provides for great flexibility in adjusting the mutual proportions of the three polypeptide fragments in embodiments when unequal ratios of the three polypeptide chains used in the construction provide the optimum yields. It is, however, possible to insert the coding sequences for two or all three polypeptide chains in one expression vector when the expression of at least two polypeptide chains in equal ratios results in high yields or when the ratios are of no particular significance.
[0162] Heteroconjugate antibodies, comprising two covalently joined monomers or antibodies, are also within the scope of the present disclosure. Such antibodies have been used to target immune system cells to unwanted cells (U.S. Pat. No. 4,676,980), and for treatment of HIV infection (PCT Publication Nos. WO 91/00360 and WO 92/200373; and EP 03089). Heteroconjugate antibodies may be made using any convenient cross-linking methods. Suitable cross-linking agents and techniques are well known in the art, and are described in U.S. Pat. No. 4,676,980.
[0163] Certain aspects of the present disclosure relate to antibody variable domains and/or antibody fragments, e.g., that may be used as a constituent of a tetravalent antibody described herein. Antibody fragments may contain the active binding region of the antibodies, such as Fab, F(ab').sub.2, scFv, Fv fragments, and the like. Various methods known in the art may be used to produce and/or isolate antibody fragments, which may be incorporated into a tetravalent antibody of the present disclosure, e.g., by standard recombinant techniques known in the art based on the concepts described herein.
[0164] Single-chain Fv fragments may be produced, such as described in Iliades et al., 1997, FEBS Letters, 409:437-441. Coupling of such single-chain fragments using various linkers is described in Kortt et al., 1997, Protein Engineering, 10:423-433. A variety of techniques for the recombinant production and manipulation of antibodies are well known in the art. Such fragments can be produced from the monoclonal antibodies described herein using techniques well established in the art (Rousseaux et al. (1986), in Methods Enzymol., 121:663-69 Academic Press).
[0165] Methods of preparing antibody fragments are well known in the art. For example, an antibody fragment can be produced by enzymatic cleavage of antibodies with pepsin to provide a 100 Kd fragment denoted F(ab').sub.2. This fragment can be further cleaved using a thiol reducing agent, and optionally a blocking group for the sulfhydryl groups resulting from cleavage of disulfide linkages, to produce 50 Kd Fab' monovalent fragments. Alternatively, an enzymatic cleavage using papain produces two monovalent Fab fragments and an Fc fragment directly. These methods are described, for example, by U.S. Pat. Nos. 4,036,945 and 4,331,647 and references contained therein, which patents are incorporated herein by reference. Also, see Nisonoff et al. (1960), Arch Biochem. Biophys. 89: 230; Porter (1959), Biochem. J. 73: 119; Smyth (1967), Methods in Enzymology 11: 421-426. Alternatively, the Fab can be produced by inserting DNA encoding Fab of the antibody into an expression vector for prokaryote or an expression vector for eukaryote, and introducing the vector into a prokaryote or eukaryote to express the Fab.
IV. Methods and Uses
[0166] Certain aspects of the present disclosure relate to methods and uses for the tetravalent antibodies described herein. These methods and uses are based at least in part on the properties of the tetravalent antibodies as described herein, including without limitation their increased number of epitope binding domains, potential for lesser dependence upon cross-linking in vitro and/or in vivo, differential potency for inducing apoptosis (e.g., of human PSGL-1 expressing cells), and/or enhanced in vivo or trans vivo efficacy.
[0167] As described herein, PSGL-1 is known to be involved in inflammation and T cell biology. The tetravalent antibodies of the present disclosure that specifically bind human PSGL-1 may find use, inter alia, in treating individuals with diseases related to T cell function (e.g., a T-cell mediated inflammatory disease), or individuals in need of medical procedures that may result in inflammatory conditions such as immunological reactions, or for which such conditions are managed beforehand (e.g., a transplantation or transfusion).
[0168] In some embodiments, a disorder or disease treated by the methods described herein may be a T-cell mediated inflammatory disease. Non-limiting examples of disorders and diseases that can be treated, or one or more of whose symptoms may be ameliorated or prevented using the tetravalent antibodies described herein described herein include psoriasis, Crohn's disease, ankylosing spondylitis, arthritis (including rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, and psoriatic arthritis), diabetes mellitus, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, autoimmune thyroiditis, dermatitis (including atopic dermatitis and eczematous dermatitis), Sjogren's Syndrome, aphthous ulcer, iritis, conjunctivitis, keratoconjunctivitis, type I diabetes, inflammatory bowel diseases, ulcerative colitis, asthma, allergic asthma, cutaneous lupus erythematosus, scleroderma, vaginitis, proctitis, drug eruptions, leprosy reversal reactions, erythema nodosum leprosum, autoimmune uveitis, allergic encephalomyelitis, acute necrotizing hemorrhagic encephalopathy, idiopathic bilateral progressive sensorineural hearing loss, aplastic anemia, pure red cell anemia, idiopathic thrombocytopenia, polychondritis, Wegener's granulomatosis, chronic active hepatitis, Stevens-Johnson syndrome, idiopathic sprue, lichen planus, Graves' disease, graft versus host disease (GVHD), sarcoidosis, primary biliary cirrhosis, uveitis posterior, interstitial lung fibrosis, allergies such as atopic allergy, AIDS, and T cell neoplasms such as leukemias or lymphomas. In some embodiments, the disease is an autoimmune disease.
[0169] In another embodiment, the disease or disorder treated in accordance with the methods described herein is plaque psoriasis. Plaque psoriasis or psoriasis vulgaris is the most common form of psoriasis and is characterized by sharply demarcated, raised erythematous skin plaques covered by silvery scale. There is a predilection of the lesions to involve the extensor surfaces of the extremities, the lumbosacral area, and the scalp. The corresponding histopathological findings include significant inflammatory cellular infiltration of the dermis and epidermis, increased numbers of dilated vessels, and a substantial thickening of the epidermis with disordered differentiation of keratinocytes and hyperkeratosis. Approximately one third of patients with plaque psoriasis are categorized as having moderate or severe disease and are consequently candidates for therapy beyond just topical treatment.
[0170] In another embodiment, the disorder treated in accordance with the methods described herein is chronic plaque psoriasis. Symptoms of plaque chronic psoriasis include, but are not limited to, single or multiple raised reddened patches of skin, ranging from coin-sized to larger, on any part of the body, including but not limited to the knees, elbows, lumbosacral regions, scalp, and nails.
[0171] In another embodiment, the disorder treated in accordance with the methods described herein is guttate psoriasis. Symptoms of guttate psoriasis include, but are not limited to, flares of water drop shaped scaly plaques on the skin, followed by an infection, such as a streptococcal throat infection.
[0172] In another embodiment, the disease or disorder treated in accordance with the methods described herein is inverse psoriasis. Symptoms of inverse psoriasis include, but are not limited to, smooth, usually moist areas of skin that are red and inflamed, unlike the scaling associated with plaque psoriasis, on one or more of the following body parts: armpits, groin, under the breasts, and in other skin folds around the genitals and buttocks.
[0173] In another embodiment, the disease or disorder treated in accordance with the methods described herein is pustular psoriasis. Symptoms of pustular psoriasis include, but are not limited to, pus-filled blisters that vary in size and location, but mostly on the hands and feet.
[0174] In another embodiment, the disease or disorder treated in accordance with the methods described herein is erythodermic psoriasis. Symptoms of erythodermic psoriasis include, but are not limited to, periodic, widespread, fiery redness of the skin and the shedding of scales in sheets, rather than smaller flakes. The reddening and shedding of the skin are often accompanied by severe itching and pain, heart rate increase, and fluctuating body temperature.
[0175] In another embodiment, the disease or disorder treated in accordance with the methods described herein is rheumatoid arthritis. Symptoms of rheumatoid arthritis, include, but are not limited to, fatigue, loss of appetite, low fever, swollen glands, weakness, joint pain in wrists, elbows, shoulders, hips, knees, ankles, toes, jaw, hands, feet, fingers, and/or neck, morning stiffness, chest pain when taking a breath (pleurisy), eye burning, itching, and discharge, nodules under the skin, numbness, tingling, or burning in the hands and feet.
[0176] In another embodiment, the disease or disorder treated in accordance with the methods described herein is Crohn's disease. Symptoms of Crohn's disease, but are not limited to, crampy abdominal (belly area) pain, fever, fatigue, loss of appetite, pain with passing stool (tenesmus), persistent, watery diarrhea, unintentional weight loss, constipation, eye inflammation, fistulas (usually around the rectal area, may cause draining of pus, mucus, or stools), joint pain, liver inflammation, mouth ulcers, rectal bleeding and bloody stools, skin lumps or sores (ulcers), and swollen gums.
[0177] In another embodiment, the disease or disorder treated in accordance with the methods described herein is ankylosing spondylitis. Symptoms of ankylosing spondylitis include, but are not limited to, frequent pain and stiffness in the lower back and buttocks, spine, and/or neck; and pain and tenderness spreading to the ribs, shoulder blades, hips, thighs and heels; inflammation of the eye (iridocyclitis and uveitis), causing redness, eye pain, vision loss, floaters and photophobia; fatigue; and nausea.
[0178] In another embodiment, the disease or disorder treated in accordance with the methods described herein is diabetes mellitus. Symptoms of diabetes mellitus include, but are not limited to, loss of weight, polyuria (frequent urination), polydipsia (increased thirst), polyphagia (increased hunger), cardiovascular disease, diabetic retinopathy, diabetic neuropathy, hyperosmolar nonketotic state, and diabetic ketoacidosis.
[0179] In some embodiments, a tetravalent antibody or composition of the present disclosure may be administered to the individual before, concurrently with, and/or after a transplantation. For example, as described in greater detail below, a tetravalent antibody or composition of the present disclosure may be administered to increase the likelihood of a favorable treatment outcome, decrease the likelihood of an unfavorable outcome, and/or mitigate or prevent symptoms or unfavorable outcomes occurring before, concurrently with, or after the transplantation has been completed.
[0180] As used herein, treating an individual in need of a transplantation may refer to one or more of therapeutic treatment and prophylactic or preventative measures (e.g., increasing the likelihood of a favorable treatment outcome, such as graft survival, graft function, or decreasing the likelihood of an unfavorable outcome, such as an unfavorable response to treatment, or a condition that reduces the likelihood a favorable treatment, such as a transplantation, from occurring). Treating may include without limitation mitigating or preventing conditions and symptoms associated with a disorder or a condition, and/or problems or conditions that interfere with or limit an individual's access to treatment options of a disorder or a condition, such as sensitization, hypersensitization, high panel reactive antibodies (PRA) level and/or presence of pre-existing alloantibodies that limit availability of grafts to an individual awaiting a transplantation. Those in need of treatment include those already with the disorder or condition, as well as those in which the disorder or condition is to be prevented. Treatment of a disorder or condition may suppress immune-mediated events associated with the disorder or condition, ameliorate the symptoms of the disorder or condition, reduce the severity of the disorder or condition, alter the course of the disorder or condition progression, and/or ameliorate or cure the basic disorder or condition.
[0181] For example, successful treatment of an individual awaiting transplantation include, but is not limited to, reducing the level of alloantibodies, reducing panel reactive antibodies (PRA), enabling the individual to have more cross-match compatible donors, increasing the likelihood or probability of the individual to receive a graft, shortening the expected waiting period of the individual for a graft, desensitizing the individual, lowering risk of transplant-associated symptoms or conditions (such as immune-mediated events as described below), or any combination thereof.
[0182] For example, successful treatment of an individual receiving a transplantation includes, but is not limited to, protection and maintenance of the transplanted organ or tissue for a long term, which comprises controlling, reversing, mitigating, delaying, or preventing one or more symptoms or undesirable conditions associated with the organ transplant, such as immune-mediated events, including, but not limited to, production of donor-specific alloantibodies (DSA), GVHD, antibody-mediated rejection (AMR), hyperacute graft rejection, chronic graft rejection, graft failure, and graft loss, as measured by functional or histological signs of the symptom or condition. A treatment capable of controlling a disorder or condition (e.g., graft rejection) may include a treatment that slows the progression of the disease process, when initiated after functional or histological signs of the disorder or condition (e.g., graft rejection) are observed. Further, a treatment capable of reversing a disease or condition (e.g., graft rejection) may include a treatment that, when initiated after functional or histological signs of the disease or condition (e.g., graft rejection) have appeared, reverses the disease process and returns functional and histological findings closer to normal. A treatment capable of "delaying progression" of a disorder or condition (e.g., graft rejection) may include deferring, hindering, slowing, retarding, stabilizing, and/or postponing development of the disorder or condition (e.g., graft rejection). This delay can be of varying lengths of time, depending on the history of the disease and/or individual being treated. As is evident to one skilled in the art, a sufficient or significant delay can, in effect, encompass prevention, in that the individual, e.g., an individual at risk for developing the disorder or condition, does not develop the disorder or condition.
[0183] In some embodiments, a transplantation of the present disclosure may be transplantation of one or more tissues or organs including without limitation bone marrow, kidney, heart, liver, neuronal tissue, lung, pancreas, skin, and intestine (e.g., small and/or large intestine, as well as any sub-tissues thereof).
[0184] In addition, tetravalent antibodies are useful for preventing and/or treating certain disorders and diseases associated with or caused (in whole or in part) by increased proliferation and/or numbers of activated T cells relative to the proliferation and/or numbers of activated T cells found in healthy individuals or individuals not having the particular disorder or disease. Non-limiting examples of disorders and diseases that can be prevented and/or treated using the tetravalent antibodies described herein include graft-versus-host disease and cases of transplantation rejection (including transplantation rejection using allogeneic or xenogeneic tissues) such as bone marrow transplantation, liver transplantation, kidney transplant, or the transplantation of any organ or tissue.
[0185] In some embodiments, a tetravalent antibody or composition of the present disclosure may be administered to the individual before, concurrently with, and/or after a transfusion. For example, as described in greater detail below, a tetravalent antibody or composition of the present disclosure may be administered to increase the likelihood of a favorable treatment outcome, decrease the likelihood of an unfavorable outcome, and/or mitigate or prevent symptoms occurring before, concurrently with, or after the transfusion has been completed.
[0186] As used herein, treating an individual in need of a transfusion may refer to one or more of therapeutic treatment and prophylactic or preventative measures (e.g., increasing the likelihood of a favorable treatment outcome, such as replacement or supplementation of blood components/cells, or decreasing the likelihood of an unfavorable outcome, such as an unfavorable response to treatment, inefficacy of treatment, or immunological reaction, or a condition that reduces the likelihood a favorable treatment, such as a transfusion, from occurring). Treating may include without limitation mitigating or preventing conditions and symptoms associated with a disorder or a condition, and/or problems or conditions that interfere with or limit an individual's access to treatment options of a disorder or a condition. Those in need of treatment include those already with the disorder or condition, as well as those in which the disorder or condition is to be prevented. Treatment of a disorder or condition may suppress immune-mediated events associated with the disorder or condition, ameliorate the symptoms of the disorder or condition, reduce the severity of the disorder or condition, alter the course of the disorder or condition progression, and/or ameliorate or cure the basic disorder or condition.
[0187] In some embodiments, the transfusion is a transfusion comprising one or more of white blood cells, red blood cells, and platelets. In some embodiments, the transfusion comprises whole blood or one or more blood products, including without limitation white blood cells, red blood cells, platelets, fresh frozen plasma, cryoprecipitate or blood clotting factors, antibodies, and/or blood substitutes. Exemplary conditions that may be treated with a transfusion (e.g., transfusion of blood or a blood product) include without limitation hemorrhage or blood loss, reduced hematocrit or hemoglobin (e.g., anemia), sickle cell disease, thalassemia, blood supplementation during or after surgical procedures, cardiac disease, traumatic injury, deficiency of one or more blood factors (e.g., hemophilia, von Willebrand disease, hypofibrinogenemia, or a deficiency in factor II, V, VII, IX, X, or XI), conditions requiring fibrinogen supplementation (e.g., liver disease, blood transfusion, etc.), bone marrow failure, platelet function disorders, thrombocytopenia, immunodeficiency (e.g., from a therapy or disease), and the like. Descriptions of practices, dosing, responses, indications, and preparations related to transfusions may be found, e.g., in the American Red Cross Compendium of Transfusion Practice Guidelines.
[0188] Administration of a tetravalent antibody or polypeptide in accordance with the methods described herein can be continuous or intermittent, depending, for example, upon the recipient's physiological condition, whether the purpose of the administration is therapeutic or prophylactic, and other factors known to skilled practitioners. The administration of an antibody or a polypeptide may be essentially continuous over a preselected period of time or may be in a series of spaced dose.
[0189] The dosage and frequency of administration of a tetravalent antibody described herein or a pharmaceutical composition thereof is administered in accordance with the methods for preventing and/or treating while minimizing side effects. The exact dosage of a tetravalent antibody described herein to be administered to a particular subject or a pharmaceutical composition thereof can be determined by a practitioner, in light of factors related to the subject that requires treatment. Factors which can be taken into account include the severity of the disease state, general health of the subject, age, and weight of the subject, diet, time and frequency of administration, combination(s) with other therapeutic agents or drugs, reaction sensitivities, and tolerance/response to therapy. The dosage and frequency of administration of a tetravalent antibody described herein or a pharmaceutical composition thereof can be adjusted over time to provide sufficient levels of the antibody or an antibody derived antigen-binding fragment, or to maintain the desired effect.
[0190] The precise dose to be employed in the formulation will also depend on the route of administration, and the seriousness of an inflammatory disorder or disease, and should be decided according to the judgment of the practitioner and each patient's circumstances.
[0191] Effective doses can be extrapolated from dose-response curves derived from in vitro or animal model test systems.
[0192] In one embodiment, any of the compositions described herein is formulated for administration by intraperitoneal, intravenous, subcutaneous, or intramuscular injections, or other forms of administration such as oral, mucosal, via inhalation, sublingually, etc. Parenteral administration, in one embodiment, is characterized by injection, either subcutaneously, intramuscularly or intravenously is also contemplated herein. Injectables can be prepared in conventional forms, either as liquid solutions or suspensions, solid forms suitable for solution or suspension in liquid prior to injection, or as emulsions. The injectables, solutions and emulsions also contain one or more excipients. Suitable excipients are, for example, water, saline, dextrose, glycerol or ethanol. In addition, if desired, the pharmaceutical compositions to be administered can also contain minor amounts of non-toxic auxiliary substances such as wetting or emulsifying agents, pH buffering agents, stabilizers, solubility enhancers, and other such agents. Other routes of administration may include, enteric administration, intracerebral administration, nasal administration, intraarterial administration, intracardiac administration, intraosseous infusion, intrathecal administration, intravenous infusion, subcutaneous implantation or injection, intramuscular administration, intrarectal administration intravaginal administration, intragastrical administration, intratracheal administration, intrapulmonary administration and intraperitoneal administration. Preparations for parenteral administration include sterile solutions ready for injection, sterile dry soluble products, such as lyophilized powders, ready to be combined with a solvent just prior to use, including sterile suspensions ready for injection, sterile dry insoluble products ready to be combined with a vehicle just prior to use and sterile emulsions. The solutions can be either aqueous or nonaqueous. If administered intravenously, suitable carriers include physiological saline or phosphate buffered saline (PBS), water, and solutions containing thickening and solubilizing agents, such as glucose, polyethylene glycol, and polypropylene glycol and mixtures thereof.
[0193] In another embodiment, the present disclosure also contemplates administration of a composition comprising the antibodies or polypeptides of the present disclosure conjugated to other molecules, such as detectable labels, or therapeutic or cytotoxic agents. The agents may include, but are not limited to radioisotopes, toxins, toxoids, inflammatory agents, enzymes, antisense molecules, peptides, cytokines, and chemotherapeutic agents. Methods of conjugating the antibodies with such molecules are generally known to those of skilled in the art. See, e.g., PCT publications WO 92/08495; WO 91/14438; WO 89/12624; U.S. Pat. No. 5,314,995; and EP 396,387.
[0194] In one embodiment, the composition comprises an antibody or polypeptide conjugated to a cytotoxic agent. Cytotoxic agents can include any agents that are detrimental to cells. An exemplary class of cytotoxic agents that can be conjugated to the antibodies or fragments may include, but are not limited to, paclitaxol, cytochalasin B, gramicidin D, ethidium bromide, emetine, mitomycin, etoposide, teniposide, vincristine, vinblastine, colchicine, doxorubicin, daunorubicin, dihydroxy anthracin dione, mitoxantrone, mithramycin, actinomycin D, 1-dehydrotestosterone, glucocorticoids, procaine, tetracaine, lidocaine, propranolol, puromycin, and analogs or homologs thereof.
V. Pharmaceutical Compositions
[0195] The present disclosure also provides pharmaceutical compositions comprising tetravalent antibodies or polypeptides described herein, and a pharmaceutically acceptable carrier or excipients. The pharmaceutical compositions may find use, e.g., in the methods, uses, and/or kits of the present disclosure.
[0196] Pharmaceutically acceptable carriers or excipients are known in the art, and are relatively inert substances that facilitate administration of a pharmacologically effective substance. For example, an excipient can give form or consistency, or act as a diluent. Suitable excipients include but are not limited to stabilizing agents, wetting and emulsifying agents, salts for varying osmolarity, encapsulating agents, buffers, and skin penetration enhancers. In certain embodiments, a tetravalent antibody described herein is in a liquid pharmaceutical composition. Liquid pharmaceutically administrable compositions can, for example, be prepared by dissolving, dispersing, or otherwise mixing an antibody described herein in a carrier, such as, for example, water, saline, aqueous dextrose, glycerol, glycols, ethanol, and the like, to thereby form a solution or suspension. If desired, the pharmaceutical composition to be administered can also contain minor amounts of nontoxic auxiliary substances such as wetting agents, emulsifying agents, solubilizing agents, and pH buffering agents and the like. Excipients as well as formulations for parenteral and nonparenteral drug delivery are set forth in Remington, The Science and Practice of Pharmacy 20th Ed. Mack Publishing (2000).
[0197] The pharmaceutical compositions are provided for administration to humans and animals in unit dosage forms, such as sterile parenteral solutions or suspensions containing suitable quantities of a tetravalent antibody described herein. The tetravalent antibody is, in one embodiment, formulated and administered in unit-dosage forms or multiple-dosage forms. Unit-dose forms as used herein refers to physically discrete units suitable for human and animal subjects and packaged individually as is known in the art. Each unit-dose contains a predetermined quantity of the antibody or the antibody derived antigen-binding fragment sufficient to produce the desired therapeutic effect, in association with the required pharmaceutical carrier, vehicle or diluent. Examples of unit-dose forms include ampoules and syringes. Unit-dose forms can be administered in fractions or multiples thereof. A multiple-dose form is a plurality of identical unit-dosage forms packaged in a single container to be administered in segregated unit-dose form. Examples of multiple-dose forms include vials, or bottles of pints or gallons. Hence, multiple dose form is a multiple of unit-doses which are not segregated in packaging.
[0198] The concentration of tetravalent antibody in the pharmaceutical composition will depend on, e.g., the physicochemical characteristics of the antibody or the antibody derived antigen-binding fragment, the dosage schedule, and amount administered as well as other factors known to those of skill in the art. In some embodiments, the pharmaceutical compositions provide a dosage of from about 0.001 mg to about 100 mg of tetravalent antibody per kilogram of body weight per day. Pharmaceutical dosage unit forms can be prepared to provide from about 0.001 mg to about 100 mg, and/or a combination of other optional essential ingredients per dosage unit form.
[0199] In some embodiments, the present disclosure provides tetravalent antibodies and compositions (such as the pharmaceutical compositions described herein) for use in any of the methods described herein, whether in the context of use as a medicament and/or use for manufacture of a medicament.
VI. Kits
[0200] Certain aspects of the present disclosure are related to kits or articles of manufacture that comprise a tetravalent antibody of the present disclosure. Optionally, the kits described herein may contain one or more pharmaceutically acceptable carriers, such as the exemplary carriers described herein. In some embodiments, a kit of the present disclosure includes a pharmaceutical composition of the present disclosure. Kits described herein may find use, e.g., in the methods or uses of the present disclosure.
[0201] Kits may optionally provide additional components such as buffers and interpretive information. Normally, the kit comprises a container and a label or package insert(s) on or associated with the container. The containers may be unit doses, bulk packages (e.g., multi-dose packages) or sub-unit doses. Instructions supplied in the kits of the present disclosure are typically written instructions on a label or package insert (e.g., a paper sheet included in the kit), but machine-readable instructions (e.g., instructions carried on a magnetic or optical storage disk) are also acceptable.
[0202] In some embodiments, the kits further include a package insert comprising instructions for administration of the tetravalent antibody to treat a T-cell mediated inflammatory disease. In some embodiments, the kits further include a package insert comprising instructions for administration of the tetravalent antibody before, concurrently with, and/or after a transfusion or transplantation.
[0203] The kits of the present disclosure are in suitable packaging. Suitable packaging includes, but is not limited to, vials, bottles, jars, flexible packaging (e.g., sealed Mylar or plastic bags), and the like. Also contemplated are packages for use in combination with a specific device, such as an inhaler, nasal administration device (e.g., an atomizer), or an infusion device such as a minipump. A kit may have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle). The container may also have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle). At least one active agent in the composition is a tetravalent antibody or polypeptide described herein. The container may further comprise a second pharmaceutically active agent. In some embodiments, a kit may further include any other material or device useful in a treatment (e.g., a transfusion or transplantation), including without limitation one or more containers, tubing, sterilizing agents or equipment, cannulae, syringes, and the like.
EXAMPLES
[0204] The invention will be more fully understood by reference to the following examples. They should not, however, be construed as limiting the scope of the invention. It is understood that the examples and embodiments described herein are for illustrative purposes only and that various modifications or changes in light thereof will be suggested to persons skilled in the art and are to be included within the spirit and purview of this application and scope of the appended claims.
Example 1: Generation and Characterization of Anti-PSGL-1 Tetravalent Antibodies
[0205] P-selectin Glycoprotein Ligand-1 (PSGL-1) is expressed on a wide range of hematopoietic cells, including myeloid, lymphoid, dendritic, and CD34+ stem cell populations (see, e.g., Spertini et al. 1996, J Cell Biol. 135(2):523-31). Several mouse antibodies specific for PSGL-1 and capable of inducing apoptosis in T cells have previously been identified. Among these mouse antibodies, an antibody (h15A7) that did not interfere with the interaction between P-selectin and PSGL-1, which required for efficient localization of T cells and neutrophils to target inflammatory tissues, was chosen for clinical development and was modified to a humanized kappa-light-chain containing IgG4 monoclonal antibody to minimize ADCC and CDC on PSGL-1 expressing cells (see, e.g., U.S. Pat. No. 7,604,800). Subsequently, h15A7 was further engineered to produce h15A7H, which has a mutation of SER228PRO in hinge region of h15A7 (International Application Pub. No. WO 2012/174001). This mutation was introduced in order to reduce antibody shuffling, the intermolecular exchange among IgG4 antibodies in vivo. In vitro studies showed that h15A7/h15A7H preferentially induced apoptosis of late-stage activated T cells but not other PSGL-1-expressing cells. Without wishing to be bound to theory, it is thought that the mechanism of action of h15A7H appears to be dependent at least in part on cross-linking of human PSGL-1 molecules, which is mediated by antibody cross-linker in vitro and possibly FcR-expressing cells in vivo.
[0206] The Example presented below describes the development of several cross-linker/FcR-expressing cell-independent tetravalent antibodies derived from h15A7H (FIGS. 1A & 1B). Without wishing to be bound to theory, tetravalent antibodies may possess advantages over h15A7H for clinical development, e.g., treatment of T-cell mediated inflammatory diseases. These results demonstrate that tetravalent h15A7H antibodies show enhanced efficacy compared to the parental h15A7H antibody both in vitro and trans vivo.
[0207] Methods
Cells and Reagents
[0208] Sp2/0-Ag14 (ATCC.RTM.CRL-1581.TM.) and Sp2/0-hPSGL-1 were cultured in 90% DMEM (GIBC.RTM., Cat. No. 11965-092.TM.) supplemented with 10% FBS (GIBC.RTM., Cat. No. 26140-079), 100 U/mL penicillin/100 .mu.g/mL streptomycin (GIBCO.RTM., Cat. No. 15140) and 1 mM sodium pyruvate (GIBCO.RTM., Cat. No. 11360).
[0209] The h15A7H antibody was described in International Application Pub. No. WO 2012/174001. The h15A7H tetravalent antibodies used in the study were produced from a Flp-In CHO stable cell line, purified by protein A affinity chromatography, and maintained in Dulbecco's Phosphate-Buffered Saline (GIBCO.RTM. Cat. No. 21600-069)/0.02% Tween-20 (JT Baker.RTM. X251-07). Human IgG4p/K as irrelevant isotype control antibody was produced from Flp-In CHO cells. 12H5.5 is a murine IgG1 anti-idiotype antibody against h15A7/h15A7H.
Animals
[0210] Female B6 mice at 6-8 weeks of age were obtained from BioLASCO Taiwan Co., Ltd, Taipei, Taiwan. All mice were maintained under specific pathogen-free conditions. All animal studies were conducted following the guidelines of the Institutional Animal Care and Use Committee.
Construction of Anti-PSGL-1 Tetravalent Antibody Variants
[0211] scDb.sub.2-Fc
[0212] scDb.sub.2-Fc (FIG. 1A, left) included 2 domains of single-chain diabodies (scDbs) fused in parallel to the N-terminals of human IgG4 Fc with a mutation in the hinge region to minimize half-antibody exchange in vivo. Each scDb domain contained not only a domain sequence of VL-VH-VL-VH, but also a linker (G.sub.4S.sub.1).sub.5 (SEQ ID NO:33) between VH and VL and two identical linkers (e.g., SEQ ID NO:34) between VL and VH. Several scDb-Fcs with said linkers of different length were generated for optimization
[0213] taFv.sub.2-Fc
[0214] taFv.sub.2-Fc (FIG. 1A, middle) included 2 tandem single-chain variable fragment (scFv) units (termed taFv for tandem scFv) fused in parallel to the N-terminals of human IgG4 Fc with a mutation in the hinge region to minimize half-antibody exchange in vivo. There were three different kinds of scFvs used to construct taFv, including v2 (VH-VL), v3 (VL-VH), and v4 (VL-VH) versions, containing a linker (G.sub.4S.sub.1).sub.5 (SEQ ID NO:33) between VH and VL. Among them, v2 and v4 were structure-constrained by the formation of VH44-VL100 disulfide bond. The VH44-VL100 disulfide bond was introduced into scFv in both VL-VH and VH-VL orientations for increased conformational stability (see SEQ ID NOs:29 and 30). Each taFv had either sequential v2-v3 or sequential v4-v2 of anti-PSGL-1 scFv with a linker ASTGS (SEQ ID NO:27) between the two scFvs.
[0215] scFv-IgG
[0216] The disulfide-constrained v2 version of anti-PSGL-1 scFv was used to generate 3 scFv-IgG4p variants (FIG. 1A, right), including scFv.sub.4-crIgG4p, scFv.sub.2-LC-IgG4p, and LC-scFv.sub.2-IgG4p. scFv.sub.4-crIgG4p had 4 scFv units fused in parallel to the N-terminals of both constant regions of kappa light chain and heavy chain of IgG4p (crIgG) without a linker. scFv.sub.2-LC-IgG4p had 2 scFv units fused in parallel to the N-terminals of kappa light chains of h15A7H IgG with a linker ASTGSG.sub.4S (SEQ ID NO:28) in-between, whereas LC-scFv.sub.2-IgG4p had 2 scFv units fused in parallel to the C-terminals of kappa light chains of h15A7H IgG with a linker (G.sub.4S).sub.2 (SEQ ID NO:34) in-between. Light chains of LC-scFv.sub.2 IgG4p and scFv.sub.2-LC IgG4p formats were separately sub-cloned into a pcDNA5/FRT vector that encoded an intact h15A7H heavy chain sequence for antibody expression. FIG. 1B shows another diagram of these tetravalent antibody formats with the variable fragments shaded.
[0217] cDNAs of all tetravalent antibodies were cloned into the pcDNA5/FRT vector (Invitrogen.TM., Cat. No: V6010-20) for tetravalent antibody expression.
Production of Stable Cell Lines Expressing Anti-PSGL-1 Tetravalent Antibody Variants
[0218] Anti-PSGL1 tetravalent antibody variants were stably expressed and produced in Flp-In CHO cells (Invitrogen.TM., Cat. No: R708-07). The cDNA sequences of tetravalent antibody variants were inserted into the pcDNA5/FRT vector (Invitrogen.TM., Cat. No: V6010-20) and co-transfected with pOG44 (Invitrogen.TM., Cat. No V6005-20) following the standard procedure provided by the vendor. The culture supernatants of the established cell lines were collected and purified with protein A sepharose beads (GE Healthcare.TM., Cat. No: 17-5280-04). The purified proteins were analyzed with both SDS-PAGE and size exclusion chromatography to ensure the quality of antibodies.
Reducing and Non-Reducing SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis)
[0219] Purified anti-PSGL-1 tetravalent antibodies were electrophoresed in 10% reducing and non-reducing SDS polyacrylamide gels. For the reducing SDS polyacrylamide gels, 2 .mu.g of antibody were mixed with 5.times.SDS sample buffer (300 nM Tris, pH6.8, 10% SDS, 50% glycerol, 5% 2-mercaptoethanol and 0.06% bromophenol blue) and boiled for 10 min at 100.degree. C. before loading. For the non-reducing SDS polyacrylamide gels, 2 .mu.g of antibodies were mixed with 5.times. non-reducing sample buffer (300 nM Tris, pH6.8, 10% SDS, 50% glycerol and 0.06% bromophenol blue) and boiled for 10 min at 100.degree. C. before loading. The reducing and non-reducing protein samples were loaded onto the same SDS-polyacrylamide gels where electrophoresis was performed. Coomassie blue staining was used to detect proteins on the gel after electrophoresis.
Binding Assay of Anti-PSGL-1 Tetravalent Antibody Variants
[0220] Sp2/0 cells transfected with human PSGL-1(Sp2/0-hPSGL1) were used as the PSGL-1 expressing cell line. Sp2/0-hPSGL1 cells were centrifuged at 1200 rpm for 5 min. The cell pellets were resuspended in FACS buffer (PBS containing 1% FBS) and pipetted into 96 well plate (1.times.10.sup.5 cells/well). To each well was added 100 .mu.l of supernatants containing humanized 15A7H(h15A7H)/tetravalent antibodies, and these were incubated for 60 min at 4.degree. C. The cells were washed three times with cold FACS buffer and then incubated with 100 .mu.l of Mouse Anti-Human IgG.sub.4 pFc'-PE (SouthernBiotech Cat. no. 9190-09) at 1 .mu.g/ml concentration for 60 min at 4.degree. C. Subsequently, the cells were washed three times with cold FACS buffer and analyzed by FACS analysis. All flow cytometric analyses were performed on a BD-LSR flow cytometer (Becton Dickinson) using the Cell Quest software.
Apoptosis Assay of Anti-PSGL-1 Tetravalent Antibody Variants
[0221] 1.times.10.sup.5 Sp2/0-hPSGL1 cells were seeded into the wells of 96-well plates. Aliquots of purified anti-PSGL-1 tetravalent and control antibodies at titrated concentrations were prepared freshly and added to each well. The treated cells were kept at 37.degree. C. for 6 hr before FACS analysis for cellular apoptosis assay.
[0222] For the cellular apoptosis assay, an Annexin-V-FITC Apoptosis Detection Kit (Strong Biotech, Cat. No. AVK250) was used following the manufacturer's instructions. In brief, the treated cells were harvested and resuspended in 100 .mu.l Annexin V binding buffer containing 0.5 .mu.l Annexin V-FITC at room temperature. After 15 min incubation in the dark, the cells were washed twice with 200 .mu.l of Annexin V binding buffer. Before FACS analysis, 1 .mu.l of propidium iodide (PI) per sample was added. All flow cytometric analyses were performed on a BD-LSR flow cytometer (Becton Dickinson) using Cell Quest software. The Annexin V positive and/or PI positive cells are considered apoptotic cells.
Isolation of Human Peripheral Blood Mononuclear Cells (PBMCs)
[0223] 500 ml whole blood was collected from healthy donors that were previously tested as good tetanus responders. The blood was centrifuged at 1500 rpm for 6 min. The upper plasma layer was discarded, and the remnant blood was diluted with an equivalent volume of PBS. The diluted whole blood was carefully added over a Ficoll (GE, Ficoll Plaque Plus, Cat #17-1440-02) layer and centrifuged at 2400 rpm for 15 mins at room temperature. The buffy coat layer containing mononuclear cells was collected and washed with PBS 3 times to minimize platelet contamination. The cells were resuspended in PBS and kept on ice before use.
Trans-Vivo Delayed Type Hypersensitivity (DTH)
[0224] 8-10.times.10.sup.6 PBMC cells, along with 0.25LF unit of PBS-dialyzed Tetanus Toxoid (TT, Kuo Kwang, Cat # K4103-11) or PBS, were injected in a final volume of 50 .mu.l into the hind footpad of female B6 mice. Mice of 6.about.8 weeks were used in all experiments. Footpad thickness was measured before and 24 hrs post injection using a dial thickness gauge. The pre-injected value was subtracted from post-injection value to obtain the net paw thickness. All measurement values were recorded in millimeters (mm). h15A7H and h15A7H tetravalent antibodies titrated in PBS were intravenously administered at indicated doses into B6 mice one hour prior to PBMC and TT injection. PBS was used as the vehicle control. 2 or 4 paws (1 or 2 mice) per treatment were tested. The plasma samples were collected 24 hrs after Ab administration to check the concentrations of antibody variants. The percent inhibition of paw thickness was calculated as follows: 100.times.(.DELTA. paw thicknesss.sub.veh-.DELTA. paw thickness.sub.Ab)/(.DELTA. paw thickness.sub.veh-.DELTA. paw thickness.sub.PBMC only).
ELISA for Detecting Antibody Concentration in Mouse Plasma
[0225] 96-well microtiter plates were coated with anti-idiotype antibody 12H5.5 at 0.5 .mu.g/mL in ELISA coating buffer (30 mM Na.sub.2CO.sub.3/100 mM NaHCO.sub.3) at 4.degree. C. overnight. Plates were then blocked with 200 .mu.L/well of 0.5% BSA in PBS for 1 hour at room temperature, and washed 3 times with ELISA washing buffer (0.05% Tween20 in PBS), followed by addition of 50 .mu.L/well of calibration standard or samples. Calibration standards at a serial dilution were first prepared in the normal mouse plasma. Calibration standard or samples were pre-diluted 1000.times. in assay diluent (0.1% BSA and 0.05% Tween 20 in PBS), to make a final concentration of 0.1% mouse plasma in assay diluents, before dispensing onto the plates. Subsequent dilutions, if needed, were prepared using assay diluents containing 0.1% normal mouse plasma. After 1 hour incubation at room temperature and washing 5 times with ELISA washing buffer, the secondary antibody mouse anti-human IgG.sub.4 pFc'-HRP (SouthernBiotech Cat. no. 9190-05; dilution 1:15000) was added at 50 .mu.L/well and incubated at room temperature for 1 hour. The plates were then washed 5 times with ELISA washing buffer, followed by addition of TMB substrate for color development. Reactions were stopped by 0.5N H.sub.2SO.sub.4, and an absorbance value was measured at 450 nm in a microtiter plate reader (Molecular Device VERSAmax).
[0226] Results
Reducing and Non-Reducing SDS-PAGE of Humanized 15A711 Tetravalent Antibodies
[0227] As shown in FIGS. 2A-2C, SDS-PAGE followed by Coomassie blue staining was used to verify the molecular weight and basic structure of anti-PSGL-1 tetravalent antibodies under non-reducing and reducing conditions. h15A7H V2-V3, V4-V2 and LH10-g4pFc under non-reducing conditions yielded a major protein band with a molecular weight of around 150 kDa (FIG. 2A). In the same conditions, h15A7H scFv.sub.2-LC IgG4p, LC-scFv.sub.2IgG4p, and scFv.sub.4-crIgG4p yielded a major protein band with a molecular weight of around 200 kDa (FIG. 2B).
[0228] Under reducing conditions, h15A7H V2-V3, V4-V2 and LH10 g4pFc showed a single band with the expected molecular weight of around 75 kDa, whereas both h15A7H scFv.sub.2-LC and LC-scFv.sub.2 showed two major bands with similar molecular weight around 50 kDa (FIG. 2C). One band was the scFv-LC or LC-scFv fusion protein, and the other was the wild type h15A7H heavy chain. scFv.sub.4-crIgG4p also showed two major bands, one representing the scFv-CH1-hinge-CH2-CH3 (around 62.5 kDa) fusion protein, and the other the scFv-kappa-fusion (around 37.5 kDa) protein (FIG. 2C). As control, the h15A7H gave a single band with an expected molecular weight of 150 kDa in the non-reducing gels (FIGS. 2A & 2B) and two major bands (heavy chain: 50 kDa, light chain: 25 kDa) under reducing conditions (FIG. 2C).
Binding of Humanized 15A711 Tetravalent Antibody Variants to SP2/O-hPSGL-1 and SP2/O
[0229] The binding ability of h15A7H tetravalent antibodies was evaluated in human PSGL-1 SP2/O cells. The h15A7H tetravalent antibody bound positively to the SP2/O-hPSGL-1, but not to parental SP2/O cell lacking of hPSGL-1 antigen (Table A below). Additionally, wild type h15A7H and all of h15A7H tetravalent antibodies gave similar binding activity on SP2/O-hPSGL-1 (Table A). These results demonstrated that h15A7H tetravalent antibodies retained binding reactivity to hPSGL-1 molecule.
TABLE-US-00003 TABLE A Binding activity (measured by mean florescence intensity) of humanized 15A7H tetravalent antibodies to SP2/O-hPSGL-1 and SP2/O. SP2/O-hPSGL-1 SP2/O (.mu.g/mL) 3 1 0.3 0.1 3 1 0.3 0.1 h15A7H 4667 6400 5943 3410 17 26 8 8 h15A7H LH10-g4pFc 4627 6677 5410 2902 19 18 31 30 h15A7H V2-V3-g4pFc 4535 6260 5731 3156 33 22 26 17 h15A7H scFv.sub.2-LC-IgG4p 4382 5744 7060 5543 24 12 11 24 h15A7H V4-V2-g4pFc 4923 6779 6454 3953 23 20 21 14 h15A7H scFv.sub.4-crIgG4p 5938 6013 4637 2640 30 28 18 8 h15A7H LC-scFv.sub.2-IgG4p 6026 5822 3477 3042 24 23 25 3 hIgG4p (control) 28 ND ND ND 33 ND ND ND ND: not done
In Vitro Apoptosis of SP2/O-hPSGL-1 Cells Induced by Humanized 15A711 Tetravalent Antibodies
[0230] Induction of apoptosis was evaluated by staining of Annexin V and/or PI in SP2/O-hPSGL-1 cells after incubation with h15A7H or tetravalent antibody. As shown in Table B below, the parental antibody, h15A7H, did not induce apoptosis in SP2/O-hPSGL-1 cells at the tested concentration of 0.5 and 0.0625 .mu.g/mL in the absence of cross-linker. At the tested concentrations of 0.5 .mu.g/mL, all of the h15A7H tetravalent antibodies induced apoptosis (ranging from 18-36%). At the lowest tested concentration tested (0.0625 .mu.g/mL), 3 out of 6 tetravalent h15A7H antibodies, LH10-g4pFc, V2-V3-g4pFc, and scFv.sub.2-LC-IgG4p, induced apoptosis in 12-16% of cells, whereas h15A7H V4-V2-g4pFc, scFv.sub.4-crIgG4p, and LC-scFv.sub.2-IgG4p did not induce cell death in SP2/O-hPSGL-1 at this lower dose. These data clearly demonstrate that all of the h15A7H tetravalent antibodies possess apoptosis-inducing ability, but that some tetravalent antibodies do so with greater potency.
TABLE-US-00004 TABLE B In vitro apoptosis of SP2/O-hPSGL-1 cells induced by humanized 15A7H tetravalent antibodies. Apoptosis % 0.5 .mu.g/mL 0.0625 .mu.g/mL (substrate background, n = 4) mean SD mean SD h15A7H 2.75 3.95 1.5 3.32 h15A7H LH10-g4pFc 26.75 11.32 11.75* 5.50 h15A7H V2-V3-g4pFc 26.5 5.69 13.5* 5.45 h15A7H scFv.sub.2-LC-IgG4p 23.75 9.00 15.5* 5.69 h15A7H V4-V2-g4pFc 30 4.55 0.5 3.00 h15A7H scFv.sub.4-crIgG4p 35.75 7.63 1.75 2.87 h15A7H LC-scFv.sub.2-IgG4p 18 9.83 0.5 4.20 SD: standard deviation *T-test P value < 0.05 (compared to treatment with V4-V2-g4pFc, scFv.sub.4-crIgG4p and LC-scFv.sub.2-IgG4p).
Efficacy of h15A7H and h15A7H Tetravalent Antibodies in the Inhibition of Trans-Vivo DTH Response in B6 Mice
[0231] The h15A7H and h15A7H tetravalent antibodies described above were tested for their efficacy in the inhibition of trans vivo DTH response in B6 mice. h15A7H antibody was intravenously injected into mice at the doses of 10 and 1 mg/kg, whereas tetravalent antibodies were intravenously injected into mice at the doses of 1 and 0.3 mg/kg. Experiments were conducted using PBMCs from four different donors, and % inhibition was calculated to evaluate the in vivo inhibitory efficacy.
[0232] As shown in Table C below, h15A7H antibody could inhibit footpad swelling by a mean of 93% at the dose of 10 mg/kg. The inhibition effect was reduced to 23% at the low dose of 1 mg/kg. As for 15A7H tetravalent antibodies, variants such as h15A7H LH10-g4p Fc, V2-V3-g4pFc and scFv.sub.2-LC-IgG4p remained effective in inhibition even at doses of 1 or 0.3 mg/kg (with 59-76% inhibition).
TABLE-US-00005 TABLE C Effect of h15A7H and h15A7H tetravalent antibodies on Trans-vivo DTH. Exp. 1 Exp. 2 Exp. 3 Exp. 4 Mean SEM % inhibition at 10 mg/kg h15A7H 71 104 82 116 93 10.2 % inhibition at 1 mg/kg h15A7H 15 28 18 32 23 4.0 h15A7H LH10-g4pFc 75 51 109 69 76 12.1 h15A7H V2-V3-g4pFc 29 33 100 91 63 18.7 h15A7H scFv.sub.2-LC-IgG4p 53 34 104 93 71 16.3 h15A7H V4-V2-g4pFc ND ND 11 44 27 16.5 h15A7H scFv.sub.4-crIgG4p 14 2 -18 6 1 6.8 h15A7H LC-scFv.sub.2-IgG4p -17 16 14 29 10 9.8 % inhibition at 0.3 mg/kg h15A7H LH10-g4pFc 28 73 109 50 65 17.2 h15A7H V2-V3-g4pFc 24 47 127 80 69 22.3 h15A7H scFv.sub.2-LC-IgG4p 74 24 66 73 59 11.8 h15A7H V4-V2-g4pFc ND ND -16 11 -2 13.7 h15A7H scFv.sub.4-crIgG4p 2 9 -7 6 3 3.6 h15A7H LC-scFv.sub.2-IgG4p -4 19 5 15 9 5.2 ND: not done.; SEM: the standard error of the mean
[0233] Plasma levels of h15A7H and h15A7H tetravalent antibodies were also measured 24 hrs after i.v. administration (Table D). All of the antibodies showed plasma levels around 6513-9025 ng/mL at 1 mg/kg except for V4-V2-g4pFc, which was undetectable after 24 hrs circulation in vivo. Without wishing to be bound by theory, it is thought that these results may indicate that the difference in efficacy among h15A7H and tetravalent variants could be mainly due to the differences in apoptosis-inducing ability, as demonstrated in Table B.
TABLE-US-00006 TABLE D Plasma concentrations of h15A7H and h15A7H tetravalent antibodies in B6 mice. Exp. 1 Exp. 2 Exp. 3 Exp. 4 Mean SEM Conc. (ng/mL) at 10 mg/kg h15A7H 103411 100189 104471 110820 104723 2227 Conc. (ng/mL) at 1 mg/kg h15A7H 9087 8578 8316 10118 9025 398 h15A7H LH10-g4pFc 5698 7333 6335 6686 6513 508 h15A7H V2-V3-g4pFc 6488 8173 6982 6478 7030 576 h15A7H scFv.sub.2-LC-IgG4p 5766 7082 7452 5979 6570 786 h15A7H V4-V2-g4pFc -- -- BLQ BLQ -- -- (<100) (<100) h15A7H scFv.sub.4-crIgG4p 6156 6924 5997 6353 6358 292 h15A7H LC-scFv.sub.2-IgG4p 7323 8432 9014 9006 8444 535 Conc. (ng/mL) at 0.3 mg/kg h15A7H LH10-g4pFc 1419 1853 1906 1793 1743 160 h15A7H V2-V3-g4pFc 1567 2284 2202 2065 2029 239 h15A7H scFv.sub.2-LC-IgG4p 1344 1968 2112 1632 1764 241 h15A7H V4-V2-g4pFc -- -- BLQ BLQ -- -- (<100) (<100) h15A7H scFv.sub.4-crIgG4p 1256 1909 1772 1668 1651 205 h15A7H LC-scFv.sub.2-IgG4p 1765 2493 2325 2356 2235 225 BLQ: beneath limit of quantification; SEM: the standard error of the mean
[0234] In summary, these data demonstrate that various h15A7H tetravalent antibodies possess differential abilities in induction of apoptosis in vitro, which correlate with differential abilities in the inhibition of a DTH response in trans vivo DTH murine model. Those tetravalent antibodies with higher potency for apoptosis induction showed enhanced efficacy compared to h15A7H in the trans-vivo DTH model. These results suggest that some of these h15A7H tetravalent variants may have potential advantages over h15A7H for further clinical development.
[0235] Although the foregoing embodiments have been described in some detail by way of illustration and example for purposes of clarity of understanding, the descriptions and examples should not be construed as limiting the scope of the present disclosure.
TABLE-US-00007 SEQUENCES All polypeptide sequences are presented N-terminal to C-terminal unless otherwise noted. All polynucleotide sequences are presented 5' to 3' unless otherwise noted. The three CDRs in each chain are underlined, and the linker regions are shown in lower case letters. Amino acid sequence of h15A7H LH10-g4pFc (SEQ ID NO: 1) DIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGT- HFT LTISSLQPEDFATYYCFQGSYVPLTFGQGTKVEIKggggsggggsEVQLVESGGGLVQPGGSLRLSCAASGFTF- SSF GMHWVRQAPGKGLEWVAYINGGSSTIFYANAVKGRFTISRDNAKNTLYLQMNSLRAEDTAVYYCARYASYGGGA- MDY WGQGTLVTVSSggggsggggsggggsggggsggggsDIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYF- EWY QQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSLQPEDFATYYCFQGSYVPLTFGQGTKVEIKggg- gsg gggsEVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKGLEWVAYINGGSSTIFYANAVKGRFTI- SRD NAKNTLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSggggsaaaESKYGPPCPPCPAPEFLGG- PSV FLFPPKPKDTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPREEQFNSTYRVVSVLTVLHQDWL- NGK EYKCKVSNKGLPSSIEKTISKAKGQPREPQVYTLPPSQEEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNY- KTT PPVLDSDGSFFLYSRLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK cDNA sequence of h15A7H LH10-g4pFc (SEQ ID NO: 2) GACATTCAGATGACCCAATCTCCGAGCTCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATC- TAG TCAGAGCATTGTACATAATGATGGAAACACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGC- TTC TCATCTATAAAGTTTCCAATCGATTTTCTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTC- ACC CTCACCATCTCTTCTCTGCAGCCGGAGGATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCAC- GTT CGGTCAAGGCACCAAGGTGGAAATCAAAggtggaggcggttcaggcggaggtggctctGAAGTGCAACTGGTGG- AGT CTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTCTGGATTCACTTTCAGTAGC- TTT GGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGGGACTCGAGTGGGTCGCATACATTAATGGTGGCAGTAGTAC- CAT CTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAGAACACCCTGTACCTGCAAA- TGA ATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTACGGAGGGGGTGCTATGGAC- TAT TGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggtggaggcggttcaggcggaggtggctctggcggtggcgg- atc cggaggcggaggttccggaggtggcggaagtGACATTCAGATGACCCAATCTCCGAGCTCTTTGTCTGCGTCTG- TAG GGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGGAAACACCTATTTTGAATGG- TAC CAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGATTTTCTGGTGTCCCATCCAG- GTT TAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCGGAGGATTTCGCAACCTATT- ACT GTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTCAAGGCACCAAGGTGGAAATCAAAggtggaggcggttca- ggc ggaggtggctctGAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTC- CTG TGCAGCCTCTGGATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGGGACTCGAGT- GGG TCGCATACATTAATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGA- GAT AATGCCAAGAACACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAG- ATA TGCTAGTTACGGAGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggaggcggag- gtt ccgcggccgcaGAGTCCAAATATGGTCCCCCATGCCCACCATGCCCAGCACCTGAGTTCCTGGGGGGACCATCA- GTC TTCCTGTTCCCCCCAAAACCCAAGGACACTCTCATGATCTCCCGGACCCCTGAGGTCACGTGCGTGGTGGTGGA- CGT GAGCCAGGAAGACCCCGAGGTCCAGTTCAACTGGTACGTGGATGGCGTGGAGGTGCATAATGCCAAGACAAAGC- CGC GGGAGGAGCAGTTCAACAGCACGTACCGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAACGGC- AAG GAGTACAAGTGCAAGGTCTCCAACAAAGGCCTCCCGTCCTCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCA- GCC CCGAGAGCCACAGGTGTACACCCTGCCCCCATCCCAGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCC- TGG TCAAAGGCTTCTACCCCAGCGACATCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACC- ACG CCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTACAGCAGGCTAACCGTGGACAAGAGCAGGTGGCAGGA- GGG GAATGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACACAGAAGAGCCTCTCCCTGTCTC- TGG GTAAATGA Amino acid sequence of h15A7H V2-V3-g4pFc (SEQ ID NO: 3) EVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKCLEWVAYINGGSSTIFYANAVKGRFTISRDN- AKN TLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSggggsggggsggggsggggsggggsDIQMTQ- SPS SLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSL- QPE DFATYYCFQGSYVPLTFGCGTKVEIKastgsDIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQ- KPG KAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSLQPEDFATYYCFQGSYVPLTFGQGTKVEIKggggsggg- gsg gggsggggsggggsEVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKGLEWVAYINGGSSTIFY- ANA VKGRFTISRDNAKNTLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSggggsaaaESKYGPPCP- PCP APEFLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPREEQFNSTYRVVS- VLT VLHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQVYTLPPSQEEMTKNQVSLTCLVKGFYPSDIAVEW- ESN GQPENNYKTTPPVLDSDGSFFLYSRLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK cDNA sequence of h15A7H V2-V3-g4pFc (SEQ ID NO: 4) GAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTC- TGG ATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGTGTCTCGAGTGGGTCGCATACA- TTA ATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAG- AAC ACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTA- CGG AGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggtggaggcggttcaggcggag- gtg gctctggcggtggcggatccggaggcggaggttccggaggtggcggaagtGACATTCAGATGACCCAATCTCCG- AGC TCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGG- AAA CACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGAT- TTT CTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCG- GAG GATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTTGTGGCACCAAGGTGGAAAT- CAA AgcttcaaccggttcaGACATTCAGATGACCCAATCTCCGAGCTCTTTGTCTGCGTCTGTAGGGGATAGGGTCA- CTA TCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGGAAACACCTATTTTGAATGGTACCAACAGAAACCA- GGA AAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGATTTTCTGGTGTCCCATCCAGGTTTAGTGGCAGTGG- GTC TGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCGGAGGATTTCGCAACCTATTACTGTTTTCAAGGTT- CAT ATGTTCCTCTCACGTTCGGTCAAGGCACCAAGGTGGAAATCAAAggtggaggcggttcaggcggaggtggctct- ggc ggtggcggatccggaggcggaggttccggaggtggcggaagtGAAGTGCAACTGGTGGAGTCTGGGGGAGGCTT- AGT GCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTCTGGATTCACTTTCAGTAGCTTTGGAATGCACTGGG- TTC GCCAGGCTCCAGGGAAGGGACTCGAGTGGGTCGCATACATTAATGGTGGCAGTAGTACCATCTTCTATGCAAAC- GCA GTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAGAACACCCTGTACCTGCAAATGAATTCTCTGAGGGC- TGA GGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTACGGAGGGGGTGCTATGGACTATTGGGGCCAAGGCA- CCC TGGTCACAGTCTCCTCAggaggcggaggttccgcggccgcaGAGTCCAAATATGGTCCCCCATGCCCACCATGC- CCA GCACCTGAGTTCCTGGGGGGACCATCAGTCTTCCTGTTCCCCCCAAAACCCAAGGACACTCTCATGATCTCCCG- GAC CCCTGAGGTCACGTGCGTGGTGGTGGACGTGAGCCAGGAAGACCCCGAGGTCCAGTTCAACTGGTACGTGGATG- GCG TGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTTCAACAGCACGTACCGTGTGGTCAGCGTCCTC- ACC GTCCTGCACCAGGACTGGCTGAACGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGGCCTCCCGTCCTCCAT- CGA GAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAGCCACAGGTGTACACCCTGCCCCCATCCCAGGAGGAGA- TGA CCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTACCCCAGCGACATCGCCGTGGAGTGGGAGAGC- AAT GGGCAGCCGGAGAACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTACAGCAG- GCT AACCGTGGACAAGAGCAGGTGGCAGGAGGGGAATGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACC- ACT ACACACAGAAGAGCCTCTCCCTGTCTCTGGGTAAATGA Amino acid sequence of h15A7H V4-V2-g4pFc (SEQ ID NO: 5) DIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGT- HFT LTISSLQPEDFATYYCFQGSYVPLTFGCGTKVEIKggggsggggsggggsggggsggggsEVQLVESGGGLVQP- GGS LRLSCAASGFTFSSFGMHWVRQAPGKCLEWVAYINGGSSTIFYANAVKGRFTISRDNAKNTLYLQMNSLRAEDT- AVY YCARYASYGGGAMDYWGQGTLVTVSSastgsEVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGK- CLE WVAYINGGSSTIFYANAVKGRFTISRDNAKNTLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSS- ggg gsggggsggggsggggsggggsDIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLL- IYK VSNRFSGVPSRFSGSGSGTHFTLTISSLQPEDFATYYCFQGSYVPLTFGCGTKVEIKggggsaaaESKYGPPCP- PCP APEFLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPREEQFNSTYRVVS- VLT VLHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQVYTLPPSQEEMTKNQVSLTCLVKGFYPSDIAVEW- ESN GQPENNYKTTPPVLDSDGSFFLYSRLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK cDNA sequence of h15A7H V4-V2-g4pFc (SEQ ID NO: 6) GACATTCAGATGACCCAATCTCCGAGCTCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATC- TAG TCAGAGCATTGTACATAATGATGGAAACACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGC- TTC TCATCTATAAAGTTTCCAATCGATTTTCTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTC- ACC CTCACCATCTCTTCTCTGCAGCCGGAGGATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCAC- GTT CGGTTGTGGCACCAAGGTGGAAATCAAAggtggaggcggttcaggcggaggtggctctggcggtggcggatccg- gag gcggaggttccggaggtggcggaagtGAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGA- AGC TTGAGACTCTCCTGTGCAGCCTCTGGATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGG- GAA GTGTCTCGAGTGGGTCGCATACATTAATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGAT- TCA CCATCTCCAGAGATAATGCCAAGAACACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTG- TAT TACTGTGCAAGATATGCTAGTTACGGAGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTC- CTC AgcttcaaccggttcaGAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGAC- TCT CCTGTGCAGCCTCTGGATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGTGTCTC- GAG TGGGTCGCATACATTAATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTC- CAG AGATAATGCCAAGAACACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTG- CAA GATATGCTAGTTACGGAGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggtgga- ggc ggttcaggcggaggtggctctggcggtggcggatccggaggcggaggttccggaggtggcggaagtGACATTCA- GAT GACCCAATCTCCGAGCTCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCA- TTG TACATAATGATGGAAACACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTAT- AAA GTTTCCAATCGATTTTCTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCAT- CTC TTCTCTGCAGCCGGAGGATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTTGTG- GCA CCAAGGTGGAAATCAAAggaggcggaggttccgcggccgcaGAGTCCAAATATGGTCCCCCATGCCCACCATGC- CCA GCACCTGAGTTCCTGGGGGGACCATCAGTCTTCCTGTTCCCCCCAAAACCCAAGGACACTCTCATGATCTCCCG- GAC CCCTGAGGTCACGTGCGTGGTGGTGGACGTGAGCCAGGAAGACCCCGAGGTCCAGTTCAACTGGTACGTGGATG- GCG TGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTTCAACAGCACGTACCGTGTGGTCAGCGTCCTC- ACC GTCCTGCACCAGGACTGGCTGAACGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGGCCTCCCGTCCTCCAT- CGA GAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAGCCACAGGTGTACACCCTGCCCCCATCCCAGGAGGAGA- TGA CCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTACCCCAGCGACATCGCCGTGGAGTGGGAGAGC- AAT
GGGCAGCCGGAGAACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTACAGCAG- GCT AACCGTGGACAAGAGCAGGTGGCAGGAGGGGAATGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACC- ACT ACACACAGAAGAGCCTCTCCCTGTCTCTGGGTAAATGA Amino acid sequence of h15A7H scFv.sub.2-LC-IgG4p Light chain (SEQ ID NO: 7) EVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKCLEWVAYINGGSSTIFYANAVKGRFTISRDN- AKN TLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSggggsggggsggggsggggsggggsDIQMTQ- SPS SLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSL- QPE DFATYYCFQGSYVPLTFGCGTKVEIKastgsggggsDIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYF- EWY QQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSLQPEDFATYYCFQGSYVPLTFGQGTKVEIKRTV- AAP SVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYE- KHK VYACEVTHQGLSSPVTKSFNRGEC cDNA sequence of h15A7H scFv.sub.2-LC-IgG4p Light chain (SEQ ID NO: 8) GAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTC- TGG ATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGTGTCTCGAGTGGGTCGCATACA- TTA ATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAG- AAC ACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTA- CGG AGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggtggaggcggttcaggcggag- gtg gctctggcggtggcggatccggaggcggaggttccggaggtggcggaagtGACATTCAGATGACCCAATCTCCG- AGC TCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGG- AAA CACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGAT- TTT CTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCG- GAG GATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTTGTGGCACCAAGGTGGAAAT- CAA AgcttcaaccggttcaggaggtggcggaagtGACATTCAGATGACCCAATCTCCGAGCTCTTTGTCTGCGTCTG- TAG GGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGGAAACACCTATTTTGAATGG- TAC CAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGATTTTCTGGTGTCCCATCCAG- GTT TAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCGGAGGATTTCGCAACCTATT- ACT GTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTCAAGGCACCAAGGTGGAAATCAAACGAACTGTGGCTGCA- CCA TCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGAACTGCCTCTGTTGTGTGCCTGCTGAATAA- CTT CTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCCCTCCAATCGGGTAACTCCCAGGAGAGTGTCA- CAG AGCAGGACAGCAAGGACAGCACCTACAGCCTCAGCAGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACAC- AAA GTCTACGCCTGCGAAGTCACCCATCAGGGCCTGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTTA- G Amino acid sequence of h15A7H LC- scFv.sub.2-IgG4p light chain (SEQ ID NO: 9) DIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGT- HFT LTISSLQPEDFATYYCFQGSYVPLTFGQGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKV- QWK VDNALQSGNSQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGECggggsgggg- sEV QLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKCLEWVAYINGGSSTIFYANAVKGRFTISRDNAK- NTL YLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSggggsggggsggggsggggsggggsDIQMTQSP- SSL SASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSLQP- EDF ATYYCFQGSYVPLTFGCGTKVEIKAAAHHHHHHHHHH cDNA sequence of h15A7H LC- scFv.sub.2-IgG4p light chain (SEQ ID NO: 10) GACATTCAGATGACCCAATCTCCGAGCTCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATC- TAG TCAGAGCATTGTACATAATGATGGAAACACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGC- TTC TCATCTATAAAGTTTCCAATCGATTTTCTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTC- ACC CTCACCATCTCTTCTCTGCAGCCGGAGGATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCAC- GTT CGGTCAAGGCACCAAGGTGGAAATCAAACGAACTGTGGCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATG- AGC AGTTGAAATCTGGAACTGCCTCTGTTGTGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGG- AAG GTGGATAACGCCCTCCAATCGGGTAACTCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAG- CCT CAGCAGCACCCTGACGCTGAGCAAAGCAGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACCCATCAGG- GCC TGAGCTCGCCCGTCACAAAGAGCTTCAACAGGGGAGAGTGTggtggaggcggttcaggcggaggtggctctGAA- GTG CAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTCTGGATT- CAC TTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGTGTCTCGAGTGGGTCGCATACATTAATG- GTG GCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAGAACACC- CTG TACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTACGGAGG- GGG TGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggtggaggcggttcaggcggaggtggct- ctg gcggtggcggatccggaggcggaggttccggaggtggcggaagtGACATTCAGATGACCCAATCTCCGAGCTCT- TTG TCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGGAAACAC- CTA TTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGATTTTCTG- GTG TCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCGGAGGAT- TTC GCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTTGTGGCACCAAGGTGGAAATCAAAGC- GGC CGCACATCATCATCATCATCACCACCACCACCACTAG Amino acid sequence of h15A7H scFv.sub.2-LC-IgG4p and h15A7 LC- ScFv.sub.2 -IgG4p heavy chain (SEQ ID NO: 11) EVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKGLEWVAYINGGSSTIFYANAVKGRFTISRDN- AKN TLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSASTKGPSVFPLAPCSRSTSESTAALGCLVKD- YFP EPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTKTYTCNVDHKPSNTKVDKRVESKYGPPCPP- CPA PEFLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPREEQFNSTYRVVSV- LTV LHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQVYTLPPSQEEMTKNQVSLTCLVKGFYPSDIAVEWE- SNG QPENNYKTTPPVLDSDGSFFLYSRLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK cDNA sequence of h15A7H scFv.sub.2-LC-IgG4p and h15A7 LC- scFv.sub.2 -IgG4p heavy chain (SEQ ID NO: 12) GAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTC- TGG ATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGGGACTCGAGTGGGTCGCATACA- TTA ATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAG- AAC ACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTA- CGG AGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAGCTTCCACCAAGGGCCCATCCG- TCT TCCCCCTGGCGCCCTGCTCCAGGAGCACCTCCGAGAGCACAGCCGCCCTGGGCTGCCTGGTCAAGGACTACTTC- CCC GAACCGGTGACGGTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTGCACACCTTCCCGGCTGTCCTACAGTC- CTC AGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTTGGGCACGAAGACCTACACCTGCAACG- TAG ATCACAAGCCCAGCAACACCAAGGTGGACAAGAGAGTTGAGTCCAAATATGGTCCCCCATGCCCACCATGCCCA- GCA CCTGAGTTCCTGGGGGGACCATCAGTCTTCCTGTTCCCCCCAAAACCCAAGGACACTCTCATGATCTCCCGGAC- CCC TGAGGTCACGTGCGTGGTGGTGGACGTGAGCCAGGAAGACCCCGAGGTCCAGTTCAACTGGTACGTGGATGGCG- TGG AGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTTCAACAGCACGTACCGTGTGGTCAGCGTCCTCACC- GTC CTGCACCAGGACTGGCTGAACGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGGCCTCCCGTCCTCCATCGA- GAA AACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAGCCACAGGTGTACACCCTGCCCCCATCCCAGGAGGAGATGA- CCA AGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTACCCCAGCGACATCGCCGTGGAGTGGGAGAGCAAT- GGG CAGCCGGAGAACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTACAGCAGGCT- AAC CGTGGACAAGAGCAGGTGGCAGGAGGGGAATGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACT- ACA CACAGAAGAGCCTCTCCCTGTCTCTGGGTAAATGA Amino acid sequence of h15A7H scFv.sub.4-crIgG4p light chain (SEQ ID NO: 13) EVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKCLEWVAYINGGSSTIFYANAVKGRFTISRDN- AKN TLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSggggsggggsggggsggggsggggsDIQMTQ- SPS SLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSL- QPE DFATYYCFQGSYVPLTFGCGTKVEIKRTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQ- SGN SQESVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC cDNA sequence of h15A7H scFv.sub.4-crIgG4p light chain (SEQ ID NO: 14) GAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTC- TGG ATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGTGTCTCGAGTGGGTCGCATACA- TTA ATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAG- AAC ACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTA- CGG AGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggtggaggcggttcaggcggag- gtg gctctggcggtggcggatccggaggcggaggttccggaggtggcggaagtGACATTCAGATGACCCAATCTCCG- AGC TCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGG- AAA CACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGAT- TTT CTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCG- GAG GATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTTGTGGCACCAAGGTGGAAAT- CAA ACGAACTGTGGCTGCACCATCTGTCTTCATCTTCCCGCCATCTGATGAGCAGTTGAAATCTGGAACTGCCTCTG- TTG TGTGCCTGCTGAATAACTTCTATCCCAGAGAGGCCAAAGTACAGTGGAAGGTGGATAACGCCCTCCAATCGGGT- AAC TCCCAGGAGAGTGTCACAGAGCAGGACAGCAAGGACAGCACCTACAGCCTCAGCAGCACCCTGACGCTGAGCAA- AGC AGACTACGAGAAACACAAAGTCTACGCCTGCGAAGTCACCCATCAGGGCCTGAGCTCGCCCGTCACAAAGAGCT- TCA ACAGGGGAGAGTGTTAG Amino acid sequence of h15A7H scFv.sub.4-crIgG4p heavy chain (SEQ ID NO: 15) EVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKCLEWVAYINGGSSTIFYANAVKGRFTISRDN- AKN TLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSSggggsggggsggggsggggsggggsDIQMTQ- SPS SLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGTHFTLTISSL- QPE DFATYYCFQGSYVPLTFGCGTKVEIKASTKGPSVFPLAPCSRSTSESTAALGCLVKDYFPEPVTVSWNSGALTS- GVH TFPAVLQSSGLYSLSSVVTVPSSSLGTKTYTCNVDHKPSNTKVDKRVESKYGPPCPPCPAPEFLGGPSVFLFPP- KPK DTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPREEQFNSTYRVVSVLTVLHQDWLNGKEYKCK- VSN KGLPSSIEKTISKAKGQPREPQVYTLPPSQEEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLD- SDG SFFLYSRLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK cDNA sequence of h15A7H scFv.sub.4-crIgG4p heavy chain (SEQ ID NO: 16) GAAGTGCAACTGGTGGAGTCTGGGGGAGGCTTAGTGCAGCCTGGAGGAAGCTTGAGACTCTCCTGTGCAGCCTC- TGG ATTCACTTTCAGTAGCTTTGGAATGCACTGGGTTCGCCAGGCTCCAGGGAAGTGTCTCGAGTGGGTCGCATACA- TTA ATGGTGGCAGTAGTACCATCTTCTATGCAAACGCAGTGAAGGGCCGATTCACCATCTCCAGAGATAATGCCAAG- AAC ACCCTGTACCTGCAAATGAATTCTCTGAGGGCTGAGGACACGGCCGTGTATTACTGTGCAAGATATGCTAGTTA- CGG AGGGGGTGCTATGGACTATTGGGGCCAAGGCACCCTGGTCACAGTCTCCTCAggtggaggcggttcaggcggag- gtg gctctggcggtggcggatccggaggcggaggttccggaggtggcggaagtGACATTCAGATGACCCAATCTCCG- AGC TCTTTGTCTGCGTCTGTAGGGGATAGGGTCACTATCACCTGCAGATCTAGTCAGAGCATTGTACATAATGATGG- AAA CACCTATTTTGAATGGTACCAACAGAAACCAGGAAAGGCACCCAAGCTTCTCATCTATAAAGTTTCCAATCGAT- TTT
CTGGTGTCCCATCCAGGTTTAGTGGCAGTGGGTCTGGGACACACTTCACCCTCACCATCTCTTCTCTGCAGCCG- GAG GATTTCGCAACCTATTACTGTTTTCAAGGTTCATATGTTCCTCTCACGTTCGGTTGTGGCACCAAGGTGGAAAT- CAA AGCTTCCACCAAGGGCCCATCCGTCTTCCCCCTGGCGCCCTGCTCCAGGAGCACCTCCGAGAGCACAGCCGCCC- TGG GCTGCCTGGTCAAGGACTACTTCCCCGAACCGGTGACGGTGTCGTGGAACTCAGGCGCCCTGACCAGCGGCGTG- CAC ACCTTCCCGGCTGTCCTACAGTCCTCAGGACTCTACTCCCTCAGCAGCGTGGTGACCGTGCCCTCCAGCAGCTT- GGG CACGAAGACCTACACCTGCAACGTAGATCACAAGCCCAGCAACACCAAGGTGGACAAGAGAGTTGAGTCCAAAT- ATG GTCCCCCATGCCCACCATGCCCAGCACCTGAGTTCCTGGGGGGACCATCAGTCTTCCTGTTCCCCCCAAAACCC- AAG GACACTCTCATGATCTCCCGGACCCCTGAGGTCACGTGCGTGGTGGTGGACGTGAGCCAGGAAGACCCCGAGGT- CCA GTTCAACTGGTACGTGGATGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTTCAACAGCA- CGT ACCGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAACGGCAAGGAGTACAAGTGCAAGGTCTCC- AAC AAAGGCCTCCCGTCCTCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAGCCACAGGTGTACAC- CCT GCCCCCATCCCAGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTACCCCAGCG- ACA TCGCCGTGGAGTGGGAGAGCAATGGGCAGCCGGAGAACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGAC- GGC TCCTTCTTCCTCTACAGCAGGCTAACCGTGGACAAGAGCAGGTGGCAGGAGGGGAATGTCTTCTCATGCTCCGT- GAT GCATGAGGCTCTGCACAACCACTACACACAGAAGAGCCTCTCCCTGTCTCTGGGTAAATGA Amino acid sequence of h15A7H CDR-H1 (SEQ ID NO: 17) SFGMH Amino acid sequence of h15A7H CDR-H2 (SEQ ID NO: 18) YINGGSSTIFYANAVKG Amino acid sequence of h15A7H CDR-H3 (SEQ ID NO: 19) YASYGGGAMDY Amino acid sequence of h15A7H CDR-L1 (SEQ ID NO: 20) RSSQSIVHNDGNTYFE Amino acid sequence of h15A7H CDR-L2 (SEQ ID NO: 21) KVSNRFS Amino acid sequence of h15A7H CDR-L3 (SEQ ID NO: 22) FQGSYVPLT Amino acid sequence of h15A7H VH (SEQ ID NO: 23) EVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKGLEWVAYINGGSSTIFYANAVKGRFTISRDN- AKN TLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSS Amino acid sequence of h15A7H VL (SEQ ID NO: 24) DIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGT- HFT LTISSLQPEDFATYYCFQGSYVPLTFGQGTKVEIK Amino acid sequence of linker sequence repeat (SEQ ID NO: 25) ggggs Amino acid sequence of linker with Fc (SEQ ID NO: 26) ggggsaaa Amino acid sequence of taFv linker (SEQ ID NO: 27) astgs Amino acid sequence of scFv light chain linker (SEQ ID NO: 28) astgsggggs Amino acid sequence of h15A7H VH G44C(SEQ ID NO: 29) EVQLVESGGGLVQPGGSLRLSCAASGFTFSSFGMHWVRQAPGKCLEWVAYINGGSSTIFYANAVKGRFTISRDN- AKN TLYLQMNSLRAEDTAVYYCARYASYGGGAMDYWGQGTLVTVSS Amino acid sequence of h15A7H VL Q100C(SEQ ID NO: 30) DIQMTQSPSSLSASVGDRVTITCRSSQSIVHNDGNTYFEWYQQKPGKAPKLLIYKVSNRFSGVPSRFSGSGSGT- HFT LTISSLQPEDFATYYCFQGSYVPLTFGCGTKVEIK Amino acid sequence of human PSGL-1 (SEQ ID NO: 31) MPLQLLLLLILLGPGNSLQLWDTWADEAEKALGPLLARDRRQATEYEYLDYDFLPETEPPEMLRNSTDTT PLTGPGTPESTTVEPAARRSTGLDAGGAVTELTTELANMGNLSTDSAAMEIQTTQPAATEAQTTQPVPTE AQTTPLAATEAQTTRLTATEAQTTPLAATEAQTTPPAATEAQTTQPTGLEAQTTAPAAMEAQTTAPAAME AQTTPPAAMEAQTTQTTAMEAQTTAPEATEAQTTQPTATEAQTTPLAAMEALSTEPSATEALSMEPTTKR GLFIPFSVSSVTHKGIPMAASNLSVNYPVGAPDHISVKQCLLAILILALVATIFFVCTVVLAVRLSRKGH MYPVRNYSPTEMVCISSLLPDGGEGPSATANGGLSKAKSPGLTPEPREDREGDDLTLHSFLP Amino acid sequence of shorter human PSGL-1 variant (SEQ ID NO: 32) MPLQLLLLLILLGPGNSLQLWDTWADEAEKALGPLLARDRRQATEYEYLDYDFLPETEPPEMLRNSTDTT PLTGPGTPESTTVEPAARRSTGLDAGGAVTELTTELANMGNLSTDSAAMEIQTTQPAATEAQTTPLAATE AQTTRLTATEAQTTPLAATEAQTTPPAATEAQTTQPTGLEAQTTAPAAMEAQTTAPAAMEAQTTPPAAME AQTTQTTAMEAQTTAPEATEAQTTQPTATEAQTTPLAAMEALSTEPSATEALSMEPTTKRGLFIPFSVSS VTHKGIPMAASNLSVNYPVGAPDHISVKQCLLAILILALVATIFFVCTVVLAVRLSRKGHMYPVRNYSPT EMVCISSLLPDGGEGPSATANGGLSKAKSPGLTPEPREDREGDDLTLHSFLP
Sequence CWU 1
1
361746PRTArtificial SequenceSynthetic Construct 1Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ser Ser Gln Ser Ile Val His Asn 20 25 30Asp Gly
Asn Thr Tyr Phe Glu Trp Tyr Gln Gln Lys Pro Gly Lys Ala 35 40 45Pro
Lys Leu Leu Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val Pro 50 55
60Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr His Phe Thr Leu Thr Ile65
70 75 80Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr Cys Phe Gln
Gly 85 90 95Ser Tyr Val Pro Leu Thr Phe Gly Gln Gly Thr Lys Val Glu
Ile Lys 100 105 110Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Glu Val
Gln Leu Val Glu 115 120 125Ser Gly Gly Gly Leu Val Gln Pro Gly Gly
Ser Leu Arg Leu Ser Cys 130 135 140Ala Ala Ser Gly Phe Thr Phe Ser
Ser Phe Gly Met His Trp Val Arg145 150 155 160Gln Ala Pro Gly Lys
Gly Leu Glu Trp Val Ala Tyr Ile Asn Gly Gly 165 170 175Ser Ser Thr
Ile Phe Tyr Ala Asn Ala Val Lys Gly Arg Phe Thr Ile 180 185 190Ser
Arg Asp Asn Ala Lys Asn Thr Leu Tyr Leu Gln Met Asn Ser Leu 195 200
205Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys Ala Arg Tyr Ala Ser Tyr
210 215 220Gly Gly Gly Ala Met Asp Tyr Trp Gly Gln Gly Thr Leu Val
Thr Val225 230 235 240Ser Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly
Ser Gly Gly Gly Gly 245 250 255Ser Gly Gly Gly Gly Ser Gly Gly Gly
Gly Ser Asp Ile Gln Met Thr 260 265 270Gln Ser Pro Ser Ser Leu Ser
Ala Ser Val Gly Asp Arg Val Thr Ile 275 280 285Thr Cys Arg Ser Ser
Gln Ser Ile Val His Asn Asp Gly Asn Thr Tyr 290 295 300Phe Glu Trp
Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile305 310 315
320Tyr Lys Val Ser Asn Arg Phe Ser Gly Val Pro Ser Arg Phe Ser Gly
325 330 335Ser Gly Ser Gly Thr His Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro 340 345 350Glu Asp Phe Ala Thr Tyr Tyr Cys Phe Gln Gly Ser
Tyr Val Pro Leu 355 360 365Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys Gly Gly Gly Gly Ser 370 375 380Gly Gly Gly Gly Ser Glu Val Gln
Leu Val Glu Ser Gly Gly Gly Leu385 390 395 400Val Gln Pro Gly Gly
Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe 405 410 415Thr Phe Ser
Ser Phe Gly Met His Trp Val Arg Gln Ala Pro Gly Lys 420 425 430Gly
Leu Glu Trp Val Ala Tyr Ile Asn Gly Gly Ser Ser Thr Ile Phe 435 440
445Tyr Ala Asn Ala Val Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala
450 455 460Lys Asn Thr Leu Tyr Leu Gln Met Asn Ser Leu Arg Ala Glu
Asp Thr465 470 475 480Ala Val Tyr Tyr Cys Ala Arg Tyr Ala Ser Tyr
Gly Gly Gly Ala Met 485 490 495Asp Tyr Trp Gly Gln Gly Thr Leu Val
Thr Val Ser Ser Gly Gly Gly 500 505 510Gly Ser Ala Ala Ala Glu Ser
Lys Tyr Gly Pro Pro Cys Pro Pro Cys 515 520 525Pro Ala Pro Glu Phe
Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro 530 535 540Lys Pro Lys
Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys545 550 555
560Val Val Val Asp Val Ser Gln Glu Asp Pro Glu Val Gln Phe Asn Trp
565 570 575Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro
Arg Glu 580 585 590Glu Gln Phe Asn Ser Thr Tyr Arg Val Val Ser Val
Leu Thr Val Leu 595 600 605His Gln Asp Trp Leu Asn Gly Lys Glu Tyr
Lys Cys Lys Val Ser Asn 610 615 620Lys Gly Leu Pro Ser Ser Ile Glu
Lys Thr Ile Ser Lys Ala Lys Gly625 630 635 640Gln Pro Arg Glu Pro
Gln Val Tyr Thr Leu Pro Pro Ser Gln Glu Glu 645 650 655Met Thr Lys
Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr 660 665 670Pro
Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn 675 680
685Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe
690 695 700Leu Tyr Ser Arg Leu Thr Val Asp Lys Ser Arg Trp Gln Glu
Gly Asn705 710 715 720Val Phe Ser Cys Ser Val Met His Glu Ala Leu
His Asn His Tyr Thr 725 730 735Gln Lys Ser Leu Ser Leu Ser Leu Gly
Lys 740 74522241DNAArtificial SequenceSynthetic Construct
2gacattcaga tgacccaatc tccgagctct ttgtctgcgt ctgtagggga tagggtcact
60atcacctgca gatctagtca gagcattgta cataatgatg gaaacaccta ttttgaatgg
120taccaacaga aaccaggaaa ggcacccaag cttctcatct ataaagtttc
caatcgattt 180tctggtgtcc catccaggtt tagtggcagt gggtctggga
cacacttcac cctcaccatc 240tcttctctgc agccggagga tttcgcaacc
tattactgtt ttcaaggttc atatgttcct 300ctcacgttcg gtcaaggcac
caaggtggaa atcaaaggtg gaggcggttc aggcggaggt 360ggctctgaag
tgcaactggt ggagtctggg ggaggcttag tgcagcctgg aggaagcttg
420agactctcct gtgcagcctc tggattcact ttcagtagct ttggaatgca
ctgggttcgc 480caggctccag ggaagggact cgagtgggtc gcatacatta
atggtggcag tagtaccatc 540ttctatgcaa acgcagtgaa gggccgattc
accatctcca gagataatgc caagaacacc 600ctgtacctgc aaatgaattc
tctgagggct gaggacacgg ccgtgtatta ctgtgcaaga 660tatgctagtt
acggaggggg tgctatggac tattggggcc aaggcaccct ggtcacagtc
720tcctcaggtg gaggcggttc aggcggaggt ggctctggcg gtggcggatc
cggaggcgga 780ggttccggag gtggcggaag tgacattcag atgacccaat
ctccgagctc tttgtctgcg 840tctgtagggg atagggtcac tatcacctgc
agatctagtc agagcattgt acataatgat 900ggaaacacct attttgaatg
gtaccaacag aaaccaggaa aggcacccaa gcttctcatc 960tataaagttt
ccaatcgatt ttctggtgtc ccatccaggt ttagtggcag tgggtctggg
1020acacacttca ccctcaccat ctcttctctg cagccggagg atttcgcaac
ctattactgt 1080tttcaaggtt catatgttcc tctcacgttc ggtcaaggca
ccaaggtgga aatcaaaggt 1140ggaggcggtt caggcggagg tggctctgaa
gtgcaactgg tggagtctgg gggaggctta 1200gtgcagcctg gaggaagctt
gagactctcc tgtgcagcct ctggattcac tttcagtagc 1260tttggaatgc
actgggttcg ccaggctcca gggaagggac tcgagtgggt cgcatacatt
1320aatggtggca gtagtaccat cttctatgca aacgcagtga agggccgatt
caccatctcc 1380agagataatg ccaagaacac cctgtacctg caaatgaatt
ctctgagggc tgaggacacg 1440gccgtgtatt actgtgcaag atatgctagt
tacggagggg gtgctatgga ctattggggc 1500caaggcaccc tggtcacagt
ctcctcagga ggcggaggtt ccgcggccgc agagtccaaa 1560tatggtcccc
catgcccacc atgcccagca cctgagttcc tggggggacc atcagtcttc
1620ctgttccccc caaaacccaa ggacactctc atgatctccc ggacccctga
ggtcacgtgc 1680gtggtggtgg acgtgagcca ggaagacccc gaggtccagt
tcaactggta cgtggatggc 1740gtggaggtgc ataatgccaa gacaaagccg
cgggaggagc agttcaacag cacgtaccgt 1800gtggtcagcg tcctcaccgt
cctgcaccag gactggctga acggcaagga gtacaagtgc 1860aaggtctcca
acaaaggcct cccgtcctcc atcgagaaaa ccatctccaa agccaaaggg
1920cagccccgag agccacaggt gtacaccctg cccccatccc aggaggagat
gaccaagaac 1980caggtcagcc tgacctgcct ggtcaaaggc ttctacccca
gcgacatcgc cgtggagtgg 2040gagagcaatg ggcagccgga gaacaactac
aagaccacgc ctcccgtgct ggactccgac 2100ggctccttct tcctctacag
caggctaacc gtggacaaga gcaggtggca ggaggggaat 2160gtcttctcat
gctccgtgat gcatgaggct ctgcacaacc actacacaca gaagagcctc
2220tccctgtctc tgggtaaatg a 22413756PRTArtificial SequenceSynthetic
Construct 3Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro
Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe
Ser Ser Phe 20 25 30Gly Met His Trp Val Arg Gln Ala Pro Gly Lys Cys
Leu Glu Trp Val 35 40 45Ala Tyr Ile Asn Gly Gly Ser Ser Thr Ile Phe
Tyr Ala Asn Ala Val 50 55 60Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn
Ala Lys Asn Thr Leu Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala
Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg Tyr Ala Ser Tyr Gly
Gly Gly Ala Met Asp Tyr Trp Gly Gln 100 105 110Gly Thr Leu Val Thr
Val Ser Ser Gly Gly Gly Gly Ser Gly Gly Gly 115 120 125Gly Ser Gly
Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly 130 135 140Ser
Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val145 150
155 160Gly Asp Arg Val Thr Ile Thr Cys Arg Ser Ser Gln Ser Ile Val
His 165 170 175Asn Asp Gly Asn Thr Tyr Phe Glu Trp Tyr Gln Gln Lys
Pro Gly Lys 180 185 190Ala Pro Lys Leu Leu Ile Tyr Lys Val Ser Asn
Arg Phe Ser Gly Val 195 200 205Pro Ser Arg Phe Ser Gly Ser Gly Ser
Gly Thr His Phe Thr Leu Thr 210 215 220Ile Ser Ser Leu Gln Pro Glu
Asp Phe Ala Thr Tyr Tyr Cys Phe Gln225 230 235 240Gly Ser Tyr Val
Pro Leu Thr Phe Gly Cys Gly Thr Lys Val Glu Ile 245 250 255Lys Ala
Ser Thr Gly Ser Asp Ile Gln Met Thr Gln Ser Pro Ser Ser 260 265
270Leu Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr Cys Arg Ser Ser
275 280 285Gln Ser Ile Val His Asn Asp Gly Asn Thr Tyr Phe Glu Trp
Tyr Gln 290 295 300Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile Tyr
Lys Val Ser Asn305 310 315 320Arg Phe Ser Gly Val Pro Ser Arg Phe
Ser Gly Ser Gly Ser Gly Thr 325 330 335His Phe Thr Leu Thr Ile Ser
Ser Leu Gln Pro Glu Asp Phe Ala Thr 340 345 350Tyr Tyr Cys Phe Gln
Gly Ser Tyr Val Pro Leu Thr Phe Gly Gln Gly 355 360 365Thr Lys Val
Glu Ile Lys Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser 370 375 380Gly
Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Glu385 390
395 400Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly
Ser 405 410 415Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser
Ser Phe Gly 420 425 430Met His Trp Val Arg Gln Ala Pro Gly Lys Gly
Leu Glu Trp Val Ala 435 440 445Tyr Ile Asn Gly Gly Ser Ser Thr Ile
Phe Tyr Ala Asn Ala Val Lys 450 455 460Gly Arg Phe Thr Ile Ser Arg
Asp Asn Ala Lys Asn Thr Leu Tyr Leu465 470 475 480Gln Met Asn Ser
Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys Ala 485 490 495Arg Tyr
Ala Ser Tyr Gly Gly Gly Ala Met Asp Tyr Trp Gly Gln Gly 500 505
510Thr Leu Val Thr Val Ser Ser Gly Gly Gly Gly Ser Ala Ala Ala Glu
515 520 525Ser Lys Tyr Gly Pro Pro Cys Pro Pro Cys Pro Ala Pro Glu
Phe Leu 530 535 540Gly Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro
Lys Asp Thr Leu545 550 555 560Met Ile Ser Arg Thr Pro Glu Val Thr
Cys Val Val Val Asp Val Ser 565 570 575Gln Glu Asp Pro Glu Val Gln
Phe Asn Trp Tyr Val Asp Gly Val Glu 580 585 590Val His Asn Ala Lys
Thr Lys Pro Arg Glu Glu Gln Phe Asn Ser Thr 595 600 605Tyr Arg Val
Val Ser Val Leu Thr Val Leu His Gln Asp Trp Leu Asn 610 615 620Gly
Lys Glu Tyr Lys Cys Lys Val Ser Asn Lys Gly Leu Pro Ser Ser625 630
635 640Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro
Gln 645 650 655Val Tyr Thr Leu Pro Pro Ser Gln Glu Glu Met Thr Lys
Asn Gln Val 660 665 670Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr Pro
Ser Asp Ile Ala Val 675 680 685Glu Trp Glu Ser Asn Gly Gln Pro Glu
Asn Asn Tyr Lys Thr Thr Pro 690 695 700Pro Val Leu Asp Ser Asp Gly
Ser Phe Phe Leu Tyr Ser Arg Leu Thr705 710 715 720Val Asp Lys Ser
Arg Trp Gln Glu Gly Asn Val Phe Ser Cys Ser Val 725 730 735Met His
Glu Ala Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu 740 745
750Ser Leu Gly Lys 75542271DNAArtificial SequenceSynthetic
Construct 4gaagtgcaac tggtggagtc tgggggaggc ttagtgcagc ctggaggaag
cttgagactc 60tcctgtgcag cctctggatt cactttcagt agctttggaa tgcactgggt
tcgccaggct 120ccagggaagt gtctcgagtg ggtcgcatac attaatggtg
gcagtagtac catcttctat 180gcaaacgcag tgaagggccg attcaccatc
tccagagata atgccaagaa caccctgtac 240ctgcaaatga attctctgag
ggctgaggac acggccgtgt attactgtgc aagatatgct 300agttacggag
ggggtgctat ggactattgg ggccaaggca ccctggtcac agtctcctca
360ggtggaggcg gttcaggcgg aggtggctct ggcggtggcg gatccggagg
cggaggttcc 420ggaggtggcg gaagtgacat tcagatgacc caatctccga
gctctttgtc tgcgtctgta 480ggggataggg tcactatcac ctgcagatct
agtcagagca ttgtacataa tgatggaaac 540acctattttg aatggtacca
acagaaacca ggaaaggcac ccaagcttct catctataaa 600gtttccaatc
gattttctgg tgtcccatcc aggtttagtg gcagtgggtc tgggacacac
660ttcaccctca ccatctcttc tctgcagccg gaggatttcg caacctatta
ctgttttcaa 720ggttcatatg ttcctctcac gttcggttgt ggcaccaagg
tggaaatcaa agcttcaacc 780ggttcagaca ttcagatgac ccaatctccg
agctctttgt ctgcgtctgt aggggatagg 840gtcactatca cctgcagatc
tagtcagagc attgtacata atgatggaaa cacctatttt 900gaatggtacc
aacagaaacc aggaaaggca cccaagcttc tcatctataa agtttccaat
960cgattttctg gtgtcccatc caggtttagt ggcagtgggt ctgggacaca
cttcaccctc 1020accatctctt ctctgcagcc ggaggatttc gcaacctatt
actgttttca aggttcatat 1080gttcctctca cgttcggtca aggcaccaag
gtggaaatca aaggtggagg cggttcaggc 1140ggaggtggct ctggcggtgg
cggatccgga ggcggaggtt ccggaggtgg cggaagtgaa 1200gtgcaactgg
tggagtctgg gggaggctta gtgcagcctg gaggaagctt gagactctcc
1260tgtgcagcct ctggattcac tttcagtagc tttggaatgc actgggttcg
ccaggctcca 1320gggaagggac tcgagtgggt cgcatacatt aatggtggca
gtagtaccat cttctatgca 1380aacgcagtga agggccgatt caccatctcc
agagataatg ccaagaacac cctgtacctg 1440caaatgaatt ctctgagggc
tgaggacacg gccgtgtatt actgtgcaag atatgctagt 1500tacggagggg
gtgctatgga ctattggggc caaggcaccc tggtcacagt ctcctcagga
1560ggcggaggtt ccgcggccgc agagtccaaa tatggtcccc catgcccacc
atgcccagca 1620cctgagttcc tggggggacc atcagtcttc ctgttccccc
caaaacccaa ggacactctc 1680atgatctccc ggacccctga ggtcacgtgc
gtggtggtgg acgtgagcca ggaagacccc 1740gaggtccagt tcaactggta
cgtggatggc gtggaggtgc ataatgccaa gacaaagccg 1800cgggaggagc
agttcaacag cacgtaccgt gtggtcagcg tcctcaccgt cctgcaccag
1860gactggctga acggcaagga gtacaagtgc aaggtctcca acaaaggcct
cccgtcctcc 1920atcgagaaaa ccatctccaa agccaaaggg cagccccgag
agccacaggt gtacaccctg 1980cccccatccc aggaggagat gaccaagaac
caggtcagcc tgacctgcct ggtcaaaggc 2040ttctacccca gcgacatcgc
cgtggagtgg gagagcaatg ggcagccgga gaacaactac 2100aagaccacgc
ctcccgtgct ggactccgac ggctccttct tcctctacag caggctaacc
2160gtggacaaga gcaggtggca ggaggggaat gtcttctcat gctccgtgat
gcatgaggct 2220ctgcacaacc actacacaca gaagagcctc tccctgtctc
tgggtaaatg a 22715756PRTArtificial SequenceSynthetic Construct 5Asp
Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10
15Asp Arg Val Thr Ile Thr Cys Arg Ser Ser Gln Ser Ile Val His Asn
20 25 30Asp Gly Asn Thr Tyr Phe Glu Trp Tyr Gln Gln Lys Pro Gly Lys
Ala 35 40 45Pro Lys Leu Leu Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly
Val Pro 50 55 60Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr His Phe Thr
Leu Thr Ile65 70 75 80Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr
Tyr Cys Phe Gln Gly 85 90 95Ser Tyr Val Pro Leu Thr Phe Gly Cys Gly
Thr Lys Val Glu Ile Lys 100 105 110Gly Gly Gly Gly Ser Gly Gly Gly
Gly Ser Gly Gly Gly Gly Ser Gly 115 120 125Gly Gly Gly Ser Gly Gly
Gly Gly Ser Glu Val Gln Leu Val Glu Ser 130 135 140Gly Gly Gly Leu
Val Gln Pro Gly Gly Ser Leu Arg Leu Ser Cys Ala145 150 155 160Ala
Ser Gly Phe Thr Phe Ser Ser Phe Gly Met His Trp Val Arg Gln 165 170
175Ala Pro Gly Lys Cys Leu Glu Trp Val Ala Tyr Ile Asn Gly Gly Ser
180 185
190Ser Thr Ile Phe Tyr Ala Asn Ala Val Lys Gly Arg Phe Thr Ile Ser
195 200 205Arg Asp Asn Ala Lys Asn Thr Leu Tyr Leu Gln Met Asn Ser
Leu Arg 210 215 220Ala Glu Asp Thr Ala Val Tyr Tyr Cys Ala Arg Tyr
Ala Ser Tyr Gly225 230 235 240Gly Gly Ala Met Asp Tyr Trp Gly Gln
Gly Thr Leu Val Thr Val Ser 245 250 255Ser Ala Ser Thr Gly Ser Glu
Val Gln Leu Val Glu Ser Gly Gly Gly 260 265 270Leu Val Gln Pro Gly
Gly Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly 275 280 285Phe Thr Phe
Ser Ser Phe Gly Met His Trp Val Arg Gln Ala Pro Gly 290 295 300Lys
Cys Leu Glu Trp Val Ala Tyr Ile Asn Gly Gly Ser Ser Thr Ile305 310
315 320Phe Tyr Ala Asn Ala Val Lys Gly Arg Phe Thr Ile Ser Arg Asp
Asn 325 330 335Ala Lys Asn Thr Leu Tyr Leu Gln Met Asn Ser Leu Arg
Ala Glu Asp 340 345 350Thr Ala Val Tyr Tyr Cys Ala Arg Tyr Ala Ser
Tyr Gly Gly Gly Ala 355 360 365Met Asp Tyr Trp Gly Gln Gly Thr Leu
Val Thr Val Ser Ser Gly Gly 370 375 380Gly Gly Ser Gly Gly Gly Gly
Ser Gly Gly Gly Gly Ser Gly Gly Gly385 390 395 400Gly Ser Gly Gly
Gly Gly Ser Asp Ile Gln Met Thr Gln Ser Pro Ser 405 410 415Ser Leu
Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr Cys Arg Ser 420 425
430Ser Gln Ser Ile Val His Asn Asp Gly Asn Thr Tyr Phe Glu Trp Tyr
435 440 445Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile Tyr Lys
Val Ser 450 455 460Asn Arg Phe Ser Gly Val Pro Ser Arg Phe Ser Gly
Ser Gly Ser Gly465 470 475 480Thr His Phe Thr Leu Thr Ile Ser Ser
Leu Gln Pro Glu Asp Phe Ala 485 490 495Thr Tyr Tyr Cys Phe Gln Gly
Ser Tyr Val Pro Leu Thr Phe Gly Cys 500 505 510Gly Thr Lys Val Glu
Ile Lys Gly Gly Gly Gly Ser Ala Ala Ala Glu 515 520 525Ser Lys Tyr
Gly Pro Pro Cys Pro Pro Cys Pro Ala Pro Glu Phe Leu 530 535 540Gly
Gly Pro Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr Leu545 550
555 560Met Ile Ser Arg Thr Pro Glu Val Thr Cys Val Val Val Asp Val
Ser 565 570 575Gln Glu Asp Pro Glu Val Gln Phe Asn Trp Tyr Val Asp
Gly Val Glu 580 585 590Val His Asn Ala Lys Thr Lys Pro Arg Glu Glu
Gln Phe Asn Ser Thr 595 600 605Tyr Arg Val Val Ser Val Leu Thr Val
Leu His Gln Asp Trp Leu Asn 610 615 620Gly Lys Glu Tyr Lys Cys Lys
Val Ser Asn Lys Gly Leu Pro Ser Ser625 630 635 640Ile Glu Lys Thr
Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln 645 650 655Val Tyr
Thr Leu Pro Pro Ser Gln Glu Glu Met Thr Lys Asn Gln Val 660 665
670Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala Val
675 680 685Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn Asn Tyr Lys Thr
Thr Pro 690 695 700Pro Val Leu Asp Ser Asp Gly Ser Phe Phe Leu Tyr
Ser Arg Leu Thr705 710 715 720Val Asp Lys Ser Arg Trp Gln Glu Gly
Asn Val Phe Ser Cys Ser Val 725 730 735Met His Glu Ala Leu His Asn
His Tyr Thr Gln Lys Ser Leu Ser Leu 740 745 750Ser Leu Gly Lys
75562271DNAArtificial SequenceSynthetic Construct 6gacattcaga
tgacccaatc tccgagctct ttgtctgcgt ctgtagggga tagggtcact 60atcacctgca
gatctagtca gagcattgta cataatgatg gaaacaccta ttttgaatgg
120taccaacaga aaccaggaaa ggcacccaag cttctcatct ataaagtttc
caatcgattt 180tctggtgtcc catccaggtt tagtggcagt gggtctggga
cacacttcac cctcaccatc 240tcttctctgc agccggagga tttcgcaacc
tattactgtt ttcaaggttc atatgttcct 300ctcacgttcg gttgtggcac
caaggtggaa atcaaaggtg gaggcggttc aggcggaggt 360ggctctggcg
gtggcggatc cggaggcgga ggttccggag gtggcggaag tgaagtgcaa
420ctggtggagt ctgggggagg cttagtgcag cctggaggaa gcttgagact
ctcctgtgca 480gcctctggat tcactttcag tagctttgga atgcactggg
ttcgccaggc tccagggaag 540tgtctcgagt gggtcgcata cattaatggt
ggcagtagta ccatcttcta tgcaaacgca 600gtgaagggcc gattcaccat
ctccagagat aatgccaaga acaccctgta cctgcaaatg 660aattctctga
gggctgagga cacggccgtg tattactgtg caagatatgc tagttacgga
720gggggtgcta tggactattg gggccaaggc accctggtca cagtctcctc
agcttcaacc 780ggttcagaag tgcaactggt ggagtctggg ggaggcttag
tgcagcctgg aggaagcttg 840agactctcct gtgcagcctc tggattcact
ttcagtagct ttggaatgca ctgggttcgc 900caggctccag ggaagtgtct
cgagtgggtc gcatacatta atggtggcag tagtaccatc 960ttctatgcaa
acgcagtgaa gggccgattc accatctcca gagataatgc caagaacacc
1020ctgtacctgc aaatgaattc tctgagggct gaggacacgg ccgtgtatta
ctgtgcaaga 1080tatgctagtt acggaggggg tgctatggac tattggggcc
aaggcaccct ggtcacagtc 1140tcctcaggtg gaggcggttc aggcggaggt
ggctctggcg gtggcggatc cggaggcgga 1200ggttccggag gtggcggaag
tgacattcag atgacccaat ctccgagctc tttgtctgcg 1260tctgtagggg
atagggtcac tatcacctgc agatctagtc agagcattgt acataatgat
1320ggaaacacct attttgaatg gtaccaacag aaaccaggaa aggcacccaa
gcttctcatc 1380tataaagttt ccaatcgatt ttctggtgtc ccatccaggt
ttagtggcag tgggtctggg 1440acacacttca ccctcaccat ctcttctctg
cagccggagg atttcgcaac ctattactgt 1500tttcaaggtt catatgttcc
tctcacgttc ggttgtggca ccaaggtgga aatcaaagga 1560ggcggaggtt
ccgcggccgc agagtccaaa tatggtcccc catgcccacc atgcccagca
1620cctgagttcc tggggggacc atcagtcttc ctgttccccc caaaacccaa
ggacactctc 1680atgatctccc ggacccctga ggtcacgtgc gtggtggtgg
acgtgagcca ggaagacccc 1740gaggtccagt tcaactggta cgtggatggc
gtggaggtgc ataatgccaa gacaaagccg 1800cgggaggagc agttcaacag
cacgtaccgt gtggtcagcg tcctcaccgt cctgcaccag 1860gactggctga
acggcaagga gtacaagtgc aaggtctcca acaaaggcct cccgtcctcc
1920atcgagaaaa ccatctccaa agccaaaggg cagccccgag agccacaggt
gtacaccctg 1980cccccatccc aggaggagat gaccaagaac caggtcagcc
tgacctgcct ggtcaaaggc 2040ttctacccca gcgacatcgc cgtggagtgg
gagagcaatg ggcagccgga gaacaactac 2100aagaccacgc ctcccgtgct
ggactccgac ggctccttct tcctctacag caggctaacc 2160gtggacaaga
gcaggtggca ggaggggaat gtcttctcat gctccgtgat gcatgaggct
2220ctgcacaacc actacacaca gaagagcctc tccctgtctc tgggtaaatg a
22717486PRTArtificial SequenceSynthetic Construct 7Glu Val Gln Leu
Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg
Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser Phe 20 25 30Gly Met
His Trp Val Arg Gln Ala Pro Gly Lys Cys Leu Glu Trp Val 35 40 45Ala
Tyr Ile Asn Gly Gly Ser Ser Thr Ile Phe Tyr Ala Asn Ala Val 50 55
60Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Thr Leu Tyr65
70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Ala Arg Tyr Ala Ser Tyr Gly Gly Gly Ala Met Asp Tyr Trp
Gly Gln 100 105 110Gly Thr Leu Val Thr Val Ser Ser Gly Gly Gly Gly
Ser Gly Gly Gly 115 120 125Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly
Gly Ser Gly Gly Gly Gly 130 135 140Ser Asp Ile Gln Met Thr Gln Ser
Pro Ser Ser Leu Ser Ala Ser Val145 150 155 160Gly Asp Arg Val Thr
Ile Thr Cys Arg Ser Ser Gln Ser Ile Val His 165 170 175Asn Asp Gly
Asn Thr Tyr Phe Glu Trp Tyr Gln Gln Lys Pro Gly Lys 180 185 190Ala
Pro Lys Leu Leu Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val 195 200
205Pro Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr His Phe Thr Leu Thr
210 215 220Ile Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr Cys
Phe Gln225 230 235 240Gly Ser Tyr Val Pro Leu Thr Phe Gly Cys Gly
Thr Lys Val Glu Ile 245 250 255Lys Ala Ser Thr Gly Ser Gly Gly Gly
Gly Ser Asp Ile Gln Met Thr 260 265 270Gln Ser Pro Ser Ser Leu Ser
Ala Ser Val Gly Asp Arg Val Thr Ile 275 280 285Thr Cys Arg Ser Ser
Gln Ser Ile Val His Asn Asp Gly Asn Thr Tyr 290 295 300Phe Glu Trp
Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile305 310 315
320Tyr Lys Val Ser Asn Arg Phe Ser Gly Val Pro Ser Arg Phe Ser Gly
325 330 335Ser Gly Ser Gly Thr His Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro 340 345 350Glu Asp Phe Ala Thr Tyr Tyr Cys Phe Gln Gly Ser
Tyr Val Pro Leu 355 360 365Thr Phe Gly Gln Gly Thr Lys Val Glu Ile
Lys Arg Thr Val Ala Ala 370 375 380Pro Ser Val Phe Ile Phe Pro Pro
Ser Asp Glu Gln Leu Lys Ser Gly385 390 395 400Thr Ala Ser Val Val
Cys Leu Leu Asn Asn Phe Tyr Pro Arg Glu Ala 405 410 415Lys Val Gln
Trp Lys Val Asp Asn Ala Leu Gln Ser Gly Asn Ser Gln 420 425 430Glu
Ser Val Thr Glu Gln Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser 435 440
445Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr
450 455 460Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser Pro Val Thr
Lys Ser465 470 475 480Phe Asn Arg Gly Glu Cys 48581461DNAArtificial
SequenceSynthetic Construct 8gaagtgcaac tggtggagtc tgggggaggc
ttagtgcagc ctggaggaag cttgagactc 60tcctgtgcag cctctggatt cactttcagt
agctttggaa tgcactgggt tcgccaggct 120ccagggaagt gtctcgagtg
ggtcgcatac attaatggtg gcagtagtac catcttctat 180gcaaacgcag
tgaagggccg attcaccatc tccagagata atgccaagaa caccctgtac
240ctgcaaatga attctctgag ggctgaggac acggccgtgt attactgtgc
aagatatgct 300agttacggag ggggtgctat ggactattgg ggccaaggca
ccctggtcac agtctcctca 360ggtggaggcg gttcaggcgg aggtggctct
ggcggtggcg gatccggagg cggaggttcc 420ggaggtggcg gaagtgacat
tcagatgacc caatctccga gctctttgtc tgcgtctgta 480ggggataggg
tcactatcac ctgcagatct agtcagagca ttgtacataa tgatggaaac
540acctattttg aatggtacca acagaaacca ggaaaggcac ccaagcttct
catctataaa 600gtttccaatc gattttctgg tgtcccatcc aggtttagtg
gcagtgggtc tgggacacac 660ttcaccctca ccatctcttc tctgcagccg
gaggatttcg caacctatta ctgttttcaa 720ggttcatatg ttcctctcac
gttcggttgt ggcaccaagg tggaaatcaa agcttcaacc 780ggttcaggag
gtggcggaag tgacattcag atgacccaat ctccgagctc tttgtctgcg
840tctgtagggg atagggtcac tatcacctgc agatctagtc agagcattgt
acataatgat 900ggaaacacct attttgaatg gtaccaacag aaaccaggaa
aggcacccaa gcttctcatc 960tataaagttt ccaatcgatt ttctggtgtc
ccatccaggt ttagtggcag tgggtctggg 1020acacacttca ccctcaccat
ctcttctctg cagccggagg atttcgcaac ctattactgt 1080tttcaaggtt
catatgttcc tctcacgttc ggtcaaggca ccaaggtgga aatcaaacga
1140actgtggctg caccatctgt cttcatcttc ccgccatctg atgagcagtt
gaaatctgga 1200actgcctctg ttgtgtgcct gctgaataac ttctatccca
gagaggccaa agtacagtgg 1260aaggtggata acgccctcca atcgggtaac
tcccaggaga gtgtcacaga gcaggacagc 1320aaggacagca cctacagcct
cagcagcacc ctgacgctga gcaaagcaga ctacgagaaa 1380cacaaagtct
acgcctgcga agtcacccat cagggcctga gctcgcccgt cacaaagagc
1440ttcaacaggg gagagtgtta g 14619499PRTArtificial SequenceSynthetic
Construct 9Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser
Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ser Ser Gln Ser Ile
Val His Asn 20 25 30Asp Gly Asn Thr Tyr Phe Glu Trp Tyr Gln Gln Lys
Pro Gly Lys Ala 35 40 45Pro Lys Leu Leu Ile Tyr Lys Val Ser Asn Arg
Phe Ser Gly Val Pro 50 55 60Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr
His Phe Thr Leu Thr Ile65 70 75 80Ser Ser Leu Gln Pro Glu Asp Phe
Ala Thr Tyr Tyr Cys Phe Gln Gly 85 90 95Ser Tyr Val Pro Leu Thr Phe
Gly Gln Gly Thr Lys Val Glu Ile Lys 100 105 110Arg Thr Val Ala Ala
Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu 115 120 125Gln Leu Lys
Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe 130 135 140Tyr
Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln145 150
155 160Ser Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp
Ser 165 170 175Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala
Asp Tyr Glu 180 185 190Lys His Lys Val Tyr Ala Cys Glu Val Thr His
Gln Gly Leu Ser Ser 195 200 205Pro Val Thr Lys Ser Phe Asn Arg Gly
Glu Cys Gly Gly Gly Gly Ser 210 215 220Gly Gly Gly Gly Ser Glu Val
Gln Leu Val Glu Ser Gly Gly Gly Leu225 230 235 240Val Gln Pro Gly
Gly Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe 245 250 255Thr Phe
Ser Ser Phe Gly Met His Trp Val Arg Gln Ala Pro Gly Lys 260 265
270Cys Leu Glu Trp Val Ala Tyr Ile Asn Gly Gly Ser Ser Thr Ile Phe
275 280 285Tyr Ala Asn Ala Val Lys Gly Arg Phe Thr Ile Ser Arg Asp
Asn Ala 290 295 300Lys Asn Thr Leu Tyr Leu Gln Met Asn Ser Leu Arg
Ala Glu Asp Thr305 310 315 320Ala Val Tyr Tyr Cys Ala Arg Tyr Ala
Ser Tyr Gly Gly Gly Ala Met 325 330 335Asp Tyr Trp Gly Gln Gly Thr
Leu Val Thr Val Ser Ser Gly Gly Gly 340 345 350Gly Ser Gly Gly Gly
Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly 355 360 365Ser Gly Gly
Gly Gly Ser Asp Ile Gln Met Thr Gln Ser Pro Ser Ser 370 375 380Leu
Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr Cys Arg Ser Ser385 390
395 400Gln Ser Ile Val His Asn Asp Gly Asn Thr Tyr Phe Glu Trp Tyr
Gln 405 410 415Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile Tyr Lys
Val Ser Asn 420 425 430Arg Phe Ser Gly Val Pro Ser Arg Phe Ser Gly
Ser Gly Ser Gly Thr 435 440 445His Phe Thr Leu Thr Ile Ser Ser Leu
Gln Pro Glu Asp Phe Ala Thr 450 455 460Tyr Tyr Cys Phe Gln Gly Ser
Tyr Val Pro Leu Thr Phe Gly Cys Gly465 470 475 480Thr Lys Val Glu
Ile Lys Ala Ala Ala His His His His His His His 485 490 495His His
His101500DNAArtificial SequenceSynthetic Construct 10gacattcaga
tgacccaatc tccgagctct ttgtctgcgt ctgtagggga tagggtcact 60atcacctgca
gatctagtca gagcattgta cataatgatg gaaacaccta ttttgaatgg
120taccaacaga aaccaggaaa ggcacccaag cttctcatct ataaagtttc
caatcgattt 180tctggtgtcc catccaggtt tagtggcagt gggtctggga
cacacttcac cctcaccatc 240tcttctctgc agccggagga tttcgcaacc
tattactgtt ttcaaggttc atatgttcct 300ctcacgttcg gtcaaggcac
caaggtggaa atcaaacgaa ctgtggctgc accatctgtc 360ttcatcttcc
cgccatctga tgagcagttg aaatctggaa ctgcctctgt tgtgtgcctg
420ctgaataact tctatcccag agaggccaaa gtacagtgga aggtggataa
cgccctccaa 480tcgggtaact cccaggagag tgtcacagag caggacagca
aggacagcac ctacagcctc 540agcagcaccc tgacgctgag caaagcagac
tacgagaaac acaaagtcta cgcctgcgaa 600gtcacccatc agggcctgag
ctcgcccgtc acaaagagct tcaacagggg agagtgtggt 660ggaggcggtt
caggcggagg tggctctgaa gtgcaactgg tggagtctgg gggaggctta
720gtgcagcctg gaggaagctt gagactctcc tgtgcagcct ctggattcac
tttcagtagc 780tttggaatgc actgggttcg ccaggctcca gggaagtgtc
tcgagtgggt cgcatacatt 840aatggtggca gtagtaccat cttctatgca
aacgcagtga agggccgatt caccatctcc 900agagataatg ccaagaacac
cctgtacctg caaatgaatt ctctgagggc tgaggacacg 960gccgtgtatt
actgtgcaag atatgctagt tacggagggg gtgctatgga ctattggggc
1020caaggcaccc tggtcacagt ctcctcaggt ggaggcggtt caggcggagg
tggctctggc 1080ggtggcggat ccggaggcgg aggttccgga ggtggcggaa
gtgacattca gatgacccaa 1140tctccgagct ctttgtctgc gtctgtaggg
gatagggtca ctatcacctg cagatctagt 1200cagagcattg tacataatga
tggaaacacc tattttgaat ggtaccaaca gaaaccagga 1260aaggcaccca
agcttctcat ctataaagtt tccaatcgat tttctggtgt cccatccagg
1320tttagtggca gtgggtctgg gacacacttc accctcacca tctcttctct
gcagccggag 1380gatttcgcaa cctattactg ttttcaaggt tcatatgttc
ctctcacgtt cggttgtggc 1440accaaggtgg aaatcaaagc ggccgcacat
catcatcatc atcaccacca ccaccactag 150011447PRTArtificial
SequenceSynthetic Construct 11Glu Val Gln Leu Val Glu Ser Gly Gly
Gly Leu Val Gln Pro Gly Gly1 5
10 15Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser
Phe 20 25 30Gly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu
Trp Val 35 40 45Ala Tyr Ile Asn Gly Gly Ser Ser Thr Ile Phe Tyr Ala
Asn Ala Val 50 55 60Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys
Asn Thr Leu Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp
Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg Tyr Ala Ser Tyr Gly Gly Gly
Ala Met Asp Tyr Trp Gly Gln 100 105 110Gly Thr Leu Val Thr Val Ser
Ser Ala Ser Thr Lys Gly Pro Ser Val 115 120 125Phe Pro Leu Ala Pro
Cys Ser Arg Ser Thr Ser Glu Ser Thr Ala Ala 130 135 140Leu Gly Cys
Leu Val Lys Asp Tyr Phe Pro Glu Pro Val Thr Val Ser145 150 155
160Trp Asn Ser Gly Ala Leu Thr Ser Gly Val His Thr Phe Pro Ala Val
165 170 175Leu Gln Ser Ser Gly Leu Tyr Ser Leu Ser Ser Val Val Thr
Val Pro 180 185 190Ser Ser Ser Leu Gly Thr Lys Thr Tyr Thr Cys Asn
Val Asp His Lys 195 200 205Pro Ser Asn Thr Lys Val Asp Lys Arg Val
Glu Ser Lys Tyr Gly Pro 210 215 220Pro Cys Pro Pro Cys Pro Ala Pro
Glu Phe Leu Gly Gly Pro Ser Val225 230 235 240Phe Leu Phe Pro Pro
Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr 245 250 255Pro Glu Val
Thr Cys Val Val Val Asp Val Ser Gln Glu Asp Pro Glu 260 265 270Val
Gln Phe Asn Trp Tyr Val Asp Gly Val Glu Val His Asn Ala Lys 275 280
285Thr Lys Pro Arg Glu Glu Gln Phe Asn Ser Thr Tyr Arg Val Val Ser
290 295 300Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly Lys Glu
Tyr Lys305 310 315 320Cys Lys Val Ser Asn Lys Gly Leu Pro Ser Ser
Ile Glu Lys Thr Ile 325 330 335Ser Lys Ala Lys Gly Gln Pro Arg Glu
Pro Gln Val Tyr Thr Leu Pro 340 345 350Pro Ser Gln Glu Glu Met Thr
Lys Asn Gln Val Ser Leu Thr Cys Leu 355 360 365Val Lys Gly Phe Tyr
Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn 370 375 380Gly Gln Pro
Glu Asn Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser385 390 395
400Asp Gly Ser Phe Phe Leu Tyr Ser Arg Leu Thr Val Asp Lys Ser Arg
405 410 415Trp Gln Glu Gly Asn Val Phe Ser Cys Ser Val Met His Glu
Ala Leu 420 425 430His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser
Leu Gly Lys 435 440 445121344DNAArtificial SequenceSynthetic
Construct 12gaagtgcaac tggtggagtc tgggggaggc ttagtgcagc ctggaggaag
cttgagactc 60tcctgtgcag cctctggatt cactttcagt agctttggaa tgcactgggt
tcgccaggct 120ccagggaagg gactcgagtg ggtcgcatac attaatggtg
gcagtagtac catcttctat 180gcaaacgcag tgaagggccg attcaccatc
tccagagata atgccaagaa caccctgtac 240ctgcaaatga attctctgag
ggctgaggac acggccgtgt attactgtgc aagatatgct 300agttacggag
ggggtgctat ggactattgg ggccaaggca ccctggtcac agtctcctca
360gcttccacca agggcccatc cgtcttcccc ctggcgccct gctccaggag
cacctccgag 420agcacagccg ccctgggctg cctggtcaag gactacttcc
ccgaaccggt gacggtgtcg 480tggaactcag gcgccctgac cagcggcgtg
cacaccttcc cggctgtcct acagtcctca 540ggactctact ccctcagcag
cgtggtgacc gtgccctcca gcagcttggg cacgaagacc 600tacacctgca
acgtagatca caagcccagc aacaccaagg tggacaagag agttgagtcc
660aaatatggtc ccccatgccc accatgccca gcacctgagt tcctgggggg
accatcagtc 720ttcctgttcc ccccaaaacc caaggacact ctcatgatct
cccggacccc tgaggtcacg 780tgcgtggtgg tggacgtgag ccaggaagac
cccgaggtcc agttcaactg gtacgtggat 840ggcgtggagg tgcataatgc
caagacaaag ccgcgggagg agcagttcaa cagcacgtac 900cgtgtggtca
gcgtcctcac cgtcctgcac caggactggc tgaacggcaa ggagtacaag
960tgcaaggtct ccaacaaagg cctcccgtcc tccatcgaga aaaccatctc
caaagccaaa 1020gggcagcccc gagagccaca ggtgtacacc ctgcccccat
cccaggagga gatgaccaag 1080aaccaggtca gcctgacctg cctggtcaaa
ggcttctacc ccagcgacat cgccgtggag 1140tgggagagca atgggcagcc
ggagaacaac tacaagacca cgcctcccgt gctggactcc 1200gacggctcct
tcttcctcta cagcaggcta accgtggaca agagcaggtg gcaggagggg
1260aatgtcttct catgctccgt gatgcatgag gctctgcaca accactacac
acagaagagc 1320ctctccctgt ctctgggtaa atga 134413364PRTArtificial
SequenceSynthetic Construct 13Glu Val Gln Leu Val Glu Ser Gly Gly
Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala
Ser Gly Phe Thr Phe Ser Ser Phe 20 25 30Gly Met His Trp Val Arg Gln
Ala Pro Gly Lys Cys Leu Glu Trp Val 35 40 45Ala Tyr Ile Asn Gly Gly
Ser Ser Thr Ile Phe Tyr Ala Asn Ala Val 50 55 60Lys Gly Arg Phe Thr
Ile Ser Arg Asp Asn Ala Lys Asn Thr Leu Tyr65 70 75 80Leu Gln Met
Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Tyr Ala Ser Tyr Gly Gly Gly Ala Met Asp Tyr Trp Gly Gln 100 105
110Gly Thr Leu Val Thr Val Ser Ser Gly Gly Gly Gly Ser Gly Gly Gly
115 120 125Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly Gly
Gly Gly 130 135 140Ser Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu
Ser Ala Ser Val145 150 155 160Gly Asp Arg Val Thr Ile Thr Cys Arg
Ser Ser Gln Ser Ile Val His 165 170 175Asn Asp Gly Asn Thr Tyr Phe
Glu Trp Tyr Gln Gln Lys Pro Gly Lys 180 185 190Ala Pro Lys Leu Leu
Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val 195 200 205Pro Ser Arg
Phe Ser Gly Ser Gly Ser Gly Thr His Phe Thr Leu Thr 210 215 220Ile
Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr Cys Phe Gln225 230
235 240Gly Ser Tyr Val Pro Leu Thr Phe Gly Cys Gly Thr Lys Val Glu
Ile 245 250 255Lys Arg Thr Val Ala Ala Pro Ser Val Phe Ile Phe Pro
Pro Ser Asp 260 265 270Glu Gln Leu Lys Ser Gly Thr Ala Ser Val Val
Cys Leu Leu Asn Asn 275 280 285Phe Tyr Pro Arg Glu Ala Lys Val Gln
Trp Lys Val Asp Asn Ala Leu 290 295 300Gln Ser Gly Asn Ser Gln Glu
Ser Val Thr Glu Gln Asp Ser Lys Asp305 310 315 320Ser Thr Tyr Ser
Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr 325 330 335Glu Lys
His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser 340 345
350Ser Pro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys 355
360141095DNAArtificial SequenceSynthetic Construct 14gaagtgcaac
tggtggagtc tgggggaggc ttagtgcagc ctggaggaag cttgagactc 60tcctgtgcag
cctctggatt cactttcagt agctttggaa tgcactgggt tcgccaggct
120ccagggaagt gtctcgagtg ggtcgcatac attaatggtg gcagtagtac
catcttctat 180gcaaacgcag tgaagggccg attcaccatc tccagagata
atgccaagaa caccctgtac 240ctgcaaatga attctctgag ggctgaggac
acggccgtgt attactgtgc aagatatgct 300agttacggag ggggtgctat
ggactattgg ggccaaggca ccctggtcac agtctcctca 360ggtggaggcg
gttcaggcgg aggtggctct ggcggtggcg gatccggagg cggaggttcc
420ggaggtggcg gaagtgacat tcagatgacc caatctccga gctctttgtc
tgcgtctgta 480ggggataggg tcactatcac ctgcagatct agtcagagca
ttgtacataa tgatggaaac 540acctattttg aatggtacca acagaaacca
ggaaaggcac ccaagcttct catctataaa 600gtttccaatc gattttctgg
tgtcccatcc aggtttagtg gcagtgggtc tgggacacac 660ttcaccctca
ccatctcttc tctgcagccg gaggatttcg caacctatta ctgttttcaa
720ggttcatatg ttcctctcac gttcggttgt ggcaccaagg tggaaatcaa
acgaactgtg 780gctgcaccat ctgtcttcat cttcccgcca tctgatgagc
agttgaaatc tggaactgcc 840tctgttgtgt gcctgctgaa taacttctat
cccagagagg ccaaagtaca gtggaaggtg 900gataacgccc tccaatcggg
taactcccag gagagtgtca cagagcagga cagcaaggac 960agcacctaca
gcctcagcag caccctgacg ctgagcaaag cagactacga gaaacacaaa
1020gtctacgcct gcgaagtcac ccatcagggc ctgagctcgc ccgtcacaaa
gagcttcaac 1080aggggagagt gttag 109515584PRTArtificial
SequenceSynthetic Construct 15Glu Val Gln Leu Val Glu Ser Gly Gly
Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser Leu Arg Leu Ser Cys Ala Ala
Ser Gly Phe Thr Phe Ser Ser Phe 20 25 30Gly Met His Trp Val Arg Gln
Ala Pro Gly Lys Cys Leu Glu Trp Val 35 40 45Ala Tyr Ile Asn Gly Gly
Ser Ser Thr Ile Phe Tyr Ala Asn Ala Val 50 55 60Lys Gly Arg Phe Thr
Ile Ser Arg Asp Asn Ala Lys Asn Thr Leu Tyr65 70 75 80Leu Gln Met
Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg
Tyr Ala Ser Tyr Gly Gly Gly Ala Met Asp Tyr Trp Gly Gln 100 105
110Gly Thr Leu Val Thr Val Ser Ser Gly Gly Gly Gly Ser Gly Gly Gly
115 120 125Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly Gly
Gly Gly 130 135 140Ser Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu
Ser Ala Ser Val145 150 155 160Gly Asp Arg Val Thr Ile Thr Cys Arg
Ser Ser Gln Ser Ile Val His 165 170 175Asn Asp Gly Asn Thr Tyr Phe
Glu Trp Tyr Gln Gln Lys Pro Gly Lys 180 185 190Ala Pro Lys Leu Leu
Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val 195 200 205Pro Ser Arg
Phe Ser Gly Ser Gly Ser Gly Thr His Phe Thr Leu Thr 210 215 220Ile
Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr Cys Phe Gln225 230
235 240Gly Ser Tyr Val Pro Leu Thr Phe Gly Cys Gly Thr Lys Val Glu
Ile 245 250 255Lys Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala
Pro Cys Ser 260 265 270Arg Ser Thr Ser Glu Ser Thr Ala Ala Leu Gly
Cys Leu Val Lys Asp 275 280 285Tyr Phe Pro Glu Pro Val Thr Val Ser
Trp Asn Ser Gly Ala Leu Thr 290 295 300Ser Gly Val His Thr Phe Pro
Ala Val Leu Gln Ser Ser Gly Leu Tyr305 310 315 320Ser Leu Ser Ser
Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Lys 325 330 335Thr Tyr
Thr Cys Asn Val Asp His Lys Pro Ser Asn Thr Lys Val Asp 340 345
350Lys Arg Val Glu Ser Lys Tyr Gly Pro Pro Cys Pro Pro Cys Pro Ala
355 360 365Pro Glu Phe Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro
Lys Pro 370 375 380Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val
Thr Cys Val Val385 390 395 400Val Asp Val Ser Gln Glu Asp Pro Glu
Val Gln Phe Asn Trp Tyr Val 405 410 415Asp Gly Val Glu Val His Asn
Ala Lys Thr Lys Pro Arg Glu Glu Gln 420 425 430Phe Asn Ser Thr Tyr
Arg Val Val Ser Val Leu Thr Val Leu His Gln 435 440 445Asp Trp Leu
Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn Lys Gly 450 455 460Leu
Pro Ser Ser Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly Gln Pro465 470
475 480Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Gln Glu Glu Met
Thr 485 490 495Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe
Tyr Pro Ser 500 505 510Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln
Pro Glu Asn Asn Tyr 515 520 525Lys Thr Thr Pro Pro Val Leu Asp Ser
Asp Gly Ser Phe Phe Leu Tyr 530 535 540Ser Arg Leu Thr Val Asp Lys
Ser Arg Trp Gln Glu Gly Asn Val Phe545 550 555 560Ser Cys Ser Val
Met His Glu Ala Leu His Asn His Tyr Thr Gln Lys 565 570 575Ser Leu
Ser Leu Ser Leu Gly Lys 580161755DNAArtificial SequenceSynthetic
Construct 16gaagtgcaac tggtggagtc tgggggaggc ttagtgcagc ctggaggaag
cttgagactc 60tcctgtgcag cctctggatt cactttcagt agctttggaa tgcactgggt
tcgccaggct 120ccagggaagt gtctcgagtg ggtcgcatac attaatggtg
gcagtagtac catcttctat 180gcaaacgcag tgaagggccg attcaccatc
tccagagata atgccaagaa caccctgtac 240ctgcaaatga attctctgag
ggctgaggac acggccgtgt attactgtgc aagatatgct 300agttacggag
ggggtgctat ggactattgg ggccaaggca ccctggtcac agtctcctca
360ggtggaggcg gttcaggcgg aggtggctct ggcggtggcg gatccggagg
cggaggttcc 420ggaggtggcg gaagtgacat tcagatgacc caatctccga
gctctttgtc tgcgtctgta 480ggggataggg tcactatcac ctgcagatct
agtcagagca ttgtacataa tgatggaaac 540acctattttg aatggtacca
acagaaacca ggaaaggcac ccaagcttct catctataaa 600gtttccaatc
gattttctgg tgtcccatcc aggtttagtg gcagtgggtc tgggacacac
660ttcaccctca ccatctcttc tctgcagccg gaggatttcg caacctatta
ctgttttcaa 720ggttcatatg ttcctctcac gttcggttgt ggcaccaagg
tggaaatcaa agcttccacc 780aagggcccat ccgtcttccc cctggcgccc
tgctccagga gcacctccga gagcacagcc 840gccctgggct gcctggtcaa
ggactacttc cccgaaccgg tgacggtgtc gtggaactca 900ggcgccctga
ccagcggcgt gcacaccttc ccggctgtcc tacagtcctc aggactctac
960tccctcagca gcgtggtgac cgtgccctcc agcagcttgg gcacgaagac
ctacacctgc 1020aacgtagatc acaagcccag caacaccaag gtggacaaga
gagttgagtc caaatatggt 1080cccccatgcc caccatgccc agcacctgag
ttcctggggg gaccatcagt cttcctgttc 1140cccccaaaac ccaaggacac
tctcatgatc tcccggaccc ctgaggtcac gtgcgtggtg 1200gtggacgtga
gccaggaaga ccccgaggtc cagttcaact ggtacgtgga tggcgtggag
1260gtgcataatg ccaagacaaa gccgcgggag gagcagttca acagcacgta
ccgtgtggtc 1320agcgtcctca ccgtcctgca ccaggactgg ctgaacggca
aggagtacaa gtgcaaggtc 1380tccaacaaag gcctcccgtc ctccatcgag
aaaaccatct ccaaagccaa agggcagccc 1440cgagagccac aggtgtacac
cctgccccca tcccaggagg agatgaccaa gaaccaggtc 1500agcctgacct
gcctggtcaa aggcttctac cccagcgaca tcgccgtgga gtgggagagc
1560aatgggcagc cggagaacaa ctacaagacc acgcctcccg tgctggactc
cgacggctcc 1620ttcttcctct acagcaggct aaccgtggac aagagcaggt
ggcaggaggg gaatgtcttc 1680tcatgctccg tgatgcatga ggctctgcac
aaccactaca cacagaagag cctctccctg 1740tctctgggta aatga
1755175PRTArtificial SequenceSynthetic Construct 17Ser Phe Gly Met
His1 51817PRTArtificial SequenceSynthetic Construct 18Tyr Ile Asn
Gly Gly Ser Ser Thr Ile Phe Tyr Ala Asn Ala Val Lys1 5 10
15Gly1911PRTArtificial SequenceSynthetic Construct 19Tyr Ala Ser
Tyr Gly Gly Gly Ala Met Asp Tyr1 5 102016PRTArtificial
SequenceSynthetic Construct 20Arg Ser Ser Gln Ser Ile Val His Asn
Asp Gly Asn Thr Tyr Phe Glu1 5 10 15217PRTArtificial
SequenceSynthetic Construct 21Lys Val Ser Asn Arg Phe Ser1
5229PRTArtificial SequenceSynthetic Construct 22Phe Gln Gly Ser Tyr
Val Pro Leu Thr1 523120PRTArtificial SequenceSynthetic Construct
23Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1
5 10 15Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser
Phe 20 25 30Gly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu
Trp Val 35 40 45Ala Tyr Ile Asn Gly Gly Ser Ser Thr Ile Phe Tyr Ala
Asn Ala Val 50 55 60Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys
Asn Thr Leu Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp
Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg Tyr Ala Ser Tyr Gly Gly Gly
Ala Met Asp Tyr Trp Gly Gln 100 105 110Gly Thr Leu Val Thr Val Ser
Ser 115 12024112PRTArtificial SequenceSynthetic Construct 24Asp Ile
Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp
Arg Val Thr Ile Thr Cys Arg Ser Ser Gln Ser Ile Val His Asn 20 25
30Asp Gly Asn Thr Tyr Phe Glu Trp Tyr Gln Gln Lys Pro Gly Lys Ala
35 40 45Pro Lys Leu Leu Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val
Pro 50 55 60Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr His Phe Thr Leu
Thr Ile65 70 75 80Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr
Cys Phe Gln Gly 85 90 95Ser Tyr Val Pro Leu Thr Phe Gly Gln Gly Thr
Lys Val Glu Ile Lys 100
105 110255PRTArtificial SequenceSynthetic Construct 25Gly Gly Gly
Gly Ser1 5268PRTArtificial SequenceSynthetic Construct 26Gly Gly
Gly Gly Ser Ala Ala Ala1 5275PRTArtificial SequenceSynthetic
Construct 27Ala Ser Thr Gly Ser1 52810PRTArtificial
SequenceSynthetic Construct 28Ala Ser Thr Gly Ser Gly Gly Gly Gly
Ser1 5 1029120PRTArtificial SequenceSynthetic Construct 29Glu Val
Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly1 5 10 15Ser
Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser Phe 20 25
30Gly Met His Trp Val Arg Gln Ala Pro Gly Lys Cys Leu Glu Trp Val
35 40 45Ala Tyr Ile Asn Gly Gly Ser Ser Thr Ile Phe Tyr Ala Asn Ala
Val 50 55 60Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Thr
Leu Tyr65 70 75 80Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala
Val Tyr Tyr Cys 85 90 95Ala Arg Tyr Ala Ser Tyr Gly Gly Gly Ala Met
Asp Tyr Trp Gly Gln 100 105 110Gly Thr Leu Val Thr Val Ser Ser 115
12030112PRTArtificial SequenceSynthetic Construct 30Asp Ile Gln Met
Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val
Thr Ile Thr Cys Arg Ser Ser Gln Ser Ile Val His Asn 20 25 30Asp Gly
Asn Thr Tyr Phe Glu Trp Tyr Gln Gln Lys Pro Gly Lys Ala 35 40 45Pro
Lys Leu Leu Ile Tyr Lys Val Ser Asn Arg Phe Ser Gly Val Pro 50 55
60Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr His Phe Thr Leu Thr Ile65
70 75 80Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr Cys Phe Gln
Gly 85 90 95Ser Tyr Val Pro Leu Thr Phe Gly Cys Gly Thr Lys Val Glu
Ile Lys 100 105 11031412PRTHomo Sapiens 31Met Pro Leu Gln Leu Leu
Leu Leu Leu Ile Leu Leu Gly Pro Gly Asn1 5 10 15Ser Leu Gln Leu Trp
Asp Thr Trp Ala Asp Glu Ala Glu Lys Ala Leu 20 25 30Gly Pro Leu Leu
Ala Arg Asp Arg Arg Gln Ala Thr Glu Tyr Glu Tyr 35 40 45Leu Asp Tyr
Asp Phe Leu Pro Glu Thr Glu Pro Pro Glu Met Leu Arg 50 55 60Asn Ser
Thr Asp Thr Thr Pro Leu Thr Gly Pro Gly Thr Pro Glu Ser65 70 75
80Thr Thr Val Glu Pro Ala Ala Arg Arg Ser Thr Gly Leu Asp Ala Gly
85 90 95Gly Ala Val Thr Glu Leu Thr Thr Glu Leu Ala Asn Met Gly Asn
Leu 100 105 110Ser Thr Asp Ser Ala Ala Met Glu Ile Gln Thr Thr Gln
Pro Ala Ala 115 120 125Thr Glu Ala Gln Thr Thr Gln Pro Val Pro Thr
Glu Ala Gln Thr Thr 130 135 140Pro Leu Ala Ala Thr Glu Ala Gln Thr
Thr Arg Leu Thr Ala Thr Glu145 150 155 160Ala Gln Thr Thr Pro Leu
Ala Ala Thr Glu Ala Gln Thr Thr Pro Pro 165 170 175Ala Ala Thr Glu
Ala Gln Thr Thr Gln Pro Thr Gly Leu Glu Ala Gln 180 185 190Thr Thr
Ala Pro Ala Ala Met Glu Ala Gln Thr Thr Ala Pro Ala Ala 195 200
205Met Glu Ala Gln Thr Thr Pro Pro Ala Ala Met Glu Ala Gln Thr Thr
210 215 220Gln Thr Thr Ala Met Glu Ala Gln Thr Thr Ala Pro Glu Ala
Thr Glu225 230 235 240Ala Gln Thr Thr Gln Pro Thr Ala Thr Glu Ala
Gln Thr Thr Pro Leu 245 250 255Ala Ala Met Glu Ala Leu Ser Thr Glu
Pro Ser Ala Thr Glu Ala Leu 260 265 270Ser Met Glu Pro Thr Thr Lys
Arg Gly Leu Phe Ile Pro Phe Ser Val 275 280 285Ser Ser Val Thr His
Lys Gly Ile Pro Met Ala Ala Ser Asn Leu Ser 290 295 300Val Asn Tyr
Pro Val Gly Ala Pro Asp His Ile Ser Val Lys Gln Cys305 310 315
320Leu Leu Ala Ile Leu Ile Leu Ala Leu Val Ala Thr Ile Phe Phe Val
325 330 335Cys Thr Val Val Leu Ala Val Arg Leu Ser Arg Lys Gly His
Met Tyr 340 345 350Pro Val Arg Asn Tyr Ser Pro Thr Glu Met Val Cys
Ile Ser Ser Leu 355 360 365Leu Pro Asp Gly Gly Glu Gly Pro Ser Ala
Thr Ala Asn Gly Gly Leu 370 375 380Ser Lys Ala Lys Ser Pro Gly Leu
Thr Pro Glu Pro Arg Glu Asp Arg385 390 395 400Glu Gly Asp Asp Leu
Thr Leu His Ser Phe Leu Pro 405 41032402PRTHomo Sapiens 32Met Pro
Leu Gln Leu Leu Leu Leu Leu Ile Leu Leu Gly Pro Gly Asn1 5 10 15Ser
Leu Gln Leu Trp Asp Thr Trp Ala Asp Glu Ala Glu Lys Ala Leu 20 25
30Gly Pro Leu Leu Ala Arg Asp Arg Arg Gln Ala Thr Glu Tyr Glu Tyr
35 40 45Leu Asp Tyr Asp Phe Leu Pro Glu Thr Glu Pro Pro Glu Met Leu
Arg 50 55 60Asn Ser Thr Asp Thr Thr Pro Leu Thr Gly Pro Gly Thr Pro
Glu Ser65 70 75 80Thr Thr Val Glu Pro Ala Ala Arg Arg Ser Thr Gly
Leu Asp Ala Gly 85 90 95Gly Ala Val Thr Glu Leu Thr Thr Glu Leu Ala
Asn Met Gly Asn Leu 100 105 110Ser Thr Asp Ser Ala Ala Met Glu Ile
Gln Thr Thr Gln Pro Ala Ala 115 120 125Thr Glu Ala Gln Thr Thr Pro
Leu Ala Ala Thr Glu Ala Gln Thr Thr 130 135 140Arg Leu Thr Ala Thr
Glu Ala Gln Thr Thr Pro Leu Ala Ala Thr Glu145 150 155 160Ala Gln
Thr Thr Pro Pro Ala Ala Thr Glu Ala Gln Thr Thr Gln Pro 165 170
175Thr Gly Leu Glu Ala Gln Thr Thr Ala Pro Ala Ala Met Glu Ala Gln
180 185 190Thr Thr Ala Pro Ala Ala Met Glu Ala Gln Thr Thr Pro Pro
Ala Ala 195 200 205Met Glu Ala Gln Thr Thr Gln Thr Thr Ala Met Glu
Ala Gln Thr Thr 210 215 220Ala Pro Glu Ala Thr Glu Ala Gln Thr Thr
Gln Pro Thr Ala Thr Glu225 230 235 240Ala Gln Thr Thr Pro Leu Ala
Ala Met Glu Ala Leu Ser Thr Glu Pro 245 250 255Ser Ala Thr Glu Ala
Leu Ser Met Glu Pro Thr Thr Lys Arg Gly Leu 260 265 270Phe Ile Pro
Phe Ser Val Ser Ser Val Thr His Lys Gly Ile Pro Met 275 280 285Ala
Ala Ser Asn Leu Ser Val Asn Tyr Pro Val Gly Ala Pro Asp His 290 295
300Ile Ser Val Lys Gln Cys Leu Leu Ala Ile Leu Ile Leu Ala Leu
Val305 310 315 320Ala Thr Ile Phe Phe Val Cys Thr Val Val Leu Ala
Val Arg Leu Ser 325 330 335Arg Lys Gly His Met Tyr Pro Val Arg Asn
Tyr Ser Pro Thr Glu Met 340 345 350Val Cys Ile Ser Ser Leu Leu Pro
Asp Gly Gly Glu Gly Pro Ser Ala 355 360 365Thr Ala Asn Gly Gly Leu
Ser Lys Ala Lys Ser Pro Gly Leu Thr Pro 370 375 380Glu Pro Arg Glu
Asp Arg Glu Gly Asp Asp Leu Thr Leu His Ser Phe385 390 395 400Leu
Pro3325PRTArtificial SequenceSynthetic Construct 33Gly Gly Gly Gly
Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly1 5 10 15Gly Gly Gly
Ser Gly Gly Gly Gly Ser 20 253410PRTArtificial SequenceSynthetic
Construct 34Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser1 5
103511PRTArtificial SequenceSynthetic Construct 35Gly Gly Gly Gly
Ser Gly Gly Gly Gly Ser Gly1 5 103612PRTArtificial
SequenceSynthetic Construct 36Gly Gly Gly Gly Ser Gly Gly Gly Gly
Ser Gly Gly1 5 10
D00000

D00001

D00002

D00003

D00004

D00005

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.